User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
nav[contains(@class, 'nav-ce-stack nav-ce-stack__large-screen')]
header[@id='header']
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'main-prefix')]
footer[@id='footer']
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
div[contains(@class, 'view-medstat-quiz-listing-panes')]
Prazosin for PTSD: Sorting out the evidence
Mr. H, age 43, presents to your clinic for management of posttraumatic stress disorder (PTSD). At his last appointment 8 weeks ago, he was continued on fluoxetine, 60 mg/d; he had been stable on this medication for 6 months. Today, Mr. H reports an increase in the frequency and severity of nightmares. He states that he wakes at least 3 times every week with “disturbing dreams” about his time in the military and does not feel rested even when he sleeps through the night. His Clinician-Administered PTSD Scale (CAPS) score is 95 on this visit, suggesting extreme PTSD symptomatology. Mr. H asks if anything can be done to reduce the frequency and intensity of his nightmares.
PTSD is the development of characteristic symptoms following exposure to ≥1 traumatic events. According to DSM-5, PTSD symptoms include the presence of ≥1 intrusion symptoms (recurrent, intrusive memories of the traumatic event; recurrent distressing dreams; dissociative reactions), persistent avoidance of stimuli, negative alterations in cognition and mood, and marked alterations in arousal and reactivity associated with the traumatic event(s).1 The symptoms must be present for >1 month, cause clinically significant distress or impairment in functioning, and not be attributable to the psychologic effects of a substance or medical conditions.1 This article focuses specifically on the hyperarousal symptoms, and the clinical controversies surrounding the use of prazosin for PTSD.
Prazosin for PTSD treatment
Sleep disorders are extremely common in patients with PTSD. Up to 90% of patients report sleep disturbances, and up to 70% report nightmares.2 Prazosin has been widely used in the treatment of PTSD-related sleep disorders and nightmares.The American Psychiatric Association3 and the British Association of Psychopharmacology4 guidelines in-clude prazosin as a first-line recommendation for treatment of PTSD. However, updated 2017 guidelines from the Veterans Affairs/Department of Defense (VA/DoD)5 and data from the 2018 Prazosin and Combat Trauma PTSD (PACT) trial6 contradict these original recommendations. Previously, the 2010 VA/DoD guideline said prazosin had insufficient evidence for monotherapy, but recommended it as adjunctive treatment for sleep and nightmares.7 The updated 2017 VA/DoD guideline recommends “weak against” prazosin use for global symptoms of PTSD, and says there is insufficient evidence for its use in nightmares.5 Below we summarize the findings of studies that contributed to those original recommendations, along with results of the PACT trial.
Raskind et al8,9 conducted 2 studies of prazosin use in combat veterans with PTSD. In both studies, prazosin had significant positive effects on the Clinician-Administered PTSD Scale (CAPS) and Clinical Global Impression of Change (CGIC) scores.8,9 The 2007 study also found significant effects of prazosin on Pittsburgh Sleep Quality Index (PSQI) scores.9
Raskind et al10 conducted another study in 2013 of prazosin use for active-duty soldiers who had combat trauma PTSD with nightmares. Prazosin had positive effects for nightmares, sleep quality, and CAPS scores.10
Germain et al11 reviewed prazosin for treating sleep disturbances in US military veterans. Prazosin was associated with significant improvements in insomnia and daytime PTSD symptom severity as demonstrated by changes in PSQI and CAPS scores.11
Taylor et al12 examined the effects of prazosin on sleep measures and clinical symptoms in civilians with PTSD. Prazosin significantly increased total sleep time, rapid eye movement sleep time, and CGIC scores while significantly decreasing trauma-related nightmares.12
Continue to: Overall, these trials...
Overall, these trials found efficacy for the use of prazosin for patients diagnosed with PTSD; however, the population size in each of these studies was small.
Results of the PACT trial
The PACT trial was a 26-week, multicenter, double-blind, randomized, placebo-controlled trial conducted across 12 VA medical centers.6 During the first 5 weeks, participants were randomized to receive placebo or prazosin, which could be titrated up to 20 mg/d in men and 12 mg/d in women. Participants remained on that dose from the end of Week 5 through Week 10. At that time, other pharmacologic therapies and psychotherapy could be added, discontinued, or adjusted. The mean maintenance total daily dose of prazosin was 14.8 mg.
A total of 413 patients were screened, 304 were randomized (152 per group), and 271 completed the 10-week primary outcome assessment. The population was almost entirely male (96.1% in the prazosin group and 99.3% in the placebo group), and most participants were White (64.5% in the prazosin group and 69.1% in the placebo group), with an average age of approximately 50 years. Primary outcomes included change from baseline to Week 10 in both CAPS item B2 (“recurrent distressing dreams”) and PSQI scores. CGIC score was evaluated at Week 10.
At Week 10, none of the primary outcomes were found to be statistically significant. The mean difference in change from baseline to Week 10 in CAPS item B2 score and PSQI score were 0.2 (P = .38) and 0.1 (P = .80), respectively. There was no significant difference in mean CGIC scores (P = .96). Repeated measures of CAPS item B2, PSQI, and CGIC scores were conducted through Week 26 as secondary outcomes. No significant differences were found. This study concluded that prazosin did not alleviate distressing dreams, improve sleep quality, or improve overall clinical symptoms.6
The PACT trial: Strengths and weaknesses
The PACT trial is the largest placebo-controlled trial for prazosin use in PTSD to date. It failed to show efficacy of prazosin for PTSD-associated nightmares, which contradicts previous studies. Although the mean total daily dose of prazosin was adequate and primary outcomes were measured with appropriate scales, the study failed to enroll the desired number of patients, which increased the possibility of false-negative results. Furthermore, participant recruitment may have led to selection bias because all participants were clinically stable, which could explain the lack of efficacy. However, the average CAPS scores were 80.7 in the prazosin group and 81.9 in the placebo group, which indicates that these patients had significant symptomatology at baseline and before entering the study.
Continue to: A major theme...
A major theme of studies evaluating prazosin treatment for PTSD is a focus on a military population and military-related trauma. Other than Taylor et al12 (N=13), none of these trials included patients who were diagnosed with PTSD due to other traumas, such as sexual trauma, which limits the generalizability of the results. Furthermore, apart from the PACT trial, none of these studies had >100 participants, which further reduces external validity. Current guidelines have not been updated to include the results of the PACT trial, and it is unclear if the results of this trial are strong enough to change clinical practice.
CASE CONTINUED
To ensure patient-centered care, the treating clinicians conduct a risk/benefit discussion with the patient regarding starting prazosin. Mr. H opts to try prazosin, so the clinicians initiate a low dose (1 mg/d) to mitigate adverse effects, and plan to titrate to clinical effect or intolerability. Per evidence from the trials discussed, it is likely Mr. H will need to be titrated to at least 5 to 6 mg/d to see a clinical effect.
Related Resource
North CS, Hong BA, Downs DL. PTSD: A systematic approach to diagnosis and treatment. Current Psychiatry 2018;17(4):35-43.
Drug Brand Names
Fluoxetine • Prozac
Prazosin • Minipress
1. Diagnostic and statistical manual of mental disorders, 5th ed. American Psychiatric Association; 2013.
2. Maher MJ, Rego SA, Asnis, GM. Sleep disturbances in patients with post-traumatic stress disorder: epidemiology, impact and approaches to management. CNS Drugs. 2006;20(7):567-590.
3. Benedek DM, Friedman MJ, Zatzick D, et al. Guideline watch (March 2009): Practice guideline for the treatment of patients with acute stress disorder and posttraumatic stress disorder. APA Practice Guidelines. Published 2010. Accessed March 14, 2021. https://psychiatryonline.org/pb/assets/raw/sitewide/practice_guidelines/guidelines/acutestressdisorderptsd-watch.pdf
4. Baldwin DS, Anderson IM, Nutt DJ, et al. Evidence-based pharmacological treatment of anxiety disorders, post-traumatic stress disorder and obsessive-compulsive disorder: a revision of the 2005 guidelines from the British Association for Psychopharmacology. J Psychopharmacol. 2014;28(5):403-439. doi: 10.1177/0269881114525674
5. Department of Veterans Affairs, Department of Defense. VA/DoD clinical practice guideline for the management of posttraumatic stress disorder and acute stress disorder. Version 3.0. Published 2017. Accessed February 5, 2021. https://www.healthquality.va.gov/guidelines/MH/ptsd/VADoDPTSDCPGFinal012418.pdf
6. Raskind MA, Peskind ER, Chow B, et al. Trial of prazosin for post-traumatic stress disorder in military veterans. N Engl J Med. 2018;378(6):507-517.
7. Department of Veterans Affairs, Department of Defense. VA/DoD clinical practice guideline: management of post-traumatic stress. Version 2.0. Published 2010. Accessed February 5, 2021. https://www.healthquality.va.gov/guidelines/MH/ptsd/cpg_PTSD-full-201011612.PDF
8. Raskind MA, Peskind ER, Katner ED, et al. Reduction of nightmares and other PTSD symptoms in combat veterans by prazosin: a placebo-controlled study. Am J Psychiatry. 2003;160(2):371-373.
9. Raskind MA, Peskind ER, Hoff DJ, et al. A parallel group placebo-controlled study of prazosin for trauma nightmares and sleep disturbance in combat veterans with post-traumatic stress disorder. Biol Psychiatry. 2007;61(8):928-934.
10. Raskind MA, Peterson K, Williams T, et al. A trial of prazosin for combat trauma PTSD with nightmares in active-duty soldiers returned from Iraq and Afghanistan. Am J Psychiatry. 2013;170(9):1003-1010.
11. Germain A, Richardson R, Moul DE, et al. Placebo-controlled comparison of prazosin and cognitive-behavioral treatments for sleep disturbances in US military veterans. J Psychosom Res. 2012;72(2):89-96.
12. Taylor FB, Martin P, Thompson C, et al. Prazosin effects on objective sleep measures and clinical symptoms in civilian trauma posttraumatic stress disorder: a placebo-controlled study. Biol Psychiatry. 2008;63(6):629-632.
Mr. H, age 43, presents to your clinic for management of posttraumatic stress disorder (PTSD). At his last appointment 8 weeks ago, he was continued on fluoxetine, 60 mg/d; he had been stable on this medication for 6 months. Today, Mr. H reports an increase in the frequency and severity of nightmares. He states that he wakes at least 3 times every week with “disturbing dreams” about his time in the military and does not feel rested even when he sleeps through the night. His Clinician-Administered PTSD Scale (CAPS) score is 95 on this visit, suggesting extreme PTSD symptomatology. Mr. H asks if anything can be done to reduce the frequency and intensity of his nightmares.
PTSD is the development of characteristic symptoms following exposure to ≥1 traumatic events. According to DSM-5, PTSD symptoms include the presence of ≥1 intrusion symptoms (recurrent, intrusive memories of the traumatic event; recurrent distressing dreams; dissociative reactions), persistent avoidance of stimuli, negative alterations in cognition and mood, and marked alterations in arousal and reactivity associated with the traumatic event(s).1 The symptoms must be present for >1 month, cause clinically significant distress or impairment in functioning, and not be attributable to the psychologic effects of a substance or medical conditions.1 This article focuses specifically on the hyperarousal symptoms, and the clinical controversies surrounding the use of prazosin for PTSD.
Prazosin for PTSD treatment
Sleep disorders are extremely common in patients with PTSD. Up to 90% of patients report sleep disturbances, and up to 70% report nightmares.2 Prazosin has been widely used in the treatment of PTSD-related sleep disorders and nightmares.The American Psychiatric Association3 and the British Association of Psychopharmacology4 guidelines in-clude prazosin as a first-line recommendation for treatment of PTSD. However, updated 2017 guidelines from the Veterans Affairs/Department of Defense (VA/DoD)5 and data from the 2018 Prazosin and Combat Trauma PTSD (PACT) trial6 contradict these original recommendations. Previously, the 2010 VA/DoD guideline said prazosin had insufficient evidence for monotherapy, but recommended it as adjunctive treatment for sleep and nightmares.7 The updated 2017 VA/DoD guideline recommends “weak against” prazosin use for global symptoms of PTSD, and says there is insufficient evidence for its use in nightmares.5 Below we summarize the findings of studies that contributed to those original recommendations, along with results of the PACT trial.
Raskind et al8,9 conducted 2 studies of prazosin use in combat veterans with PTSD. In both studies, prazosin had significant positive effects on the Clinician-Administered PTSD Scale (CAPS) and Clinical Global Impression of Change (CGIC) scores.8,9 The 2007 study also found significant effects of prazosin on Pittsburgh Sleep Quality Index (PSQI) scores.9
Raskind et al10 conducted another study in 2013 of prazosin use for active-duty soldiers who had combat trauma PTSD with nightmares. Prazosin had positive effects for nightmares, sleep quality, and CAPS scores.10
Germain et al11 reviewed prazosin for treating sleep disturbances in US military veterans. Prazosin was associated with significant improvements in insomnia and daytime PTSD symptom severity as demonstrated by changes in PSQI and CAPS scores.11
Taylor et al12 examined the effects of prazosin on sleep measures and clinical symptoms in civilians with PTSD. Prazosin significantly increased total sleep time, rapid eye movement sleep time, and CGIC scores while significantly decreasing trauma-related nightmares.12
Continue to: Overall, these trials...
Overall, these trials found efficacy for the use of prazosin for patients diagnosed with PTSD; however, the population size in each of these studies was small.
Results of the PACT trial
The PACT trial was a 26-week, multicenter, double-blind, randomized, placebo-controlled trial conducted across 12 VA medical centers.6 During the first 5 weeks, participants were randomized to receive placebo or prazosin, which could be titrated up to 20 mg/d in men and 12 mg/d in women. Participants remained on that dose from the end of Week 5 through Week 10. At that time, other pharmacologic therapies and psychotherapy could be added, discontinued, or adjusted. The mean maintenance total daily dose of prazosin was 14.8 mg.
A total of 413 patients were screened, 304 were randomized (152 per group), and 271 completed the 10-week primary outcome assessment. The population was almost entirely male (96.1% in the prazosin group and 99.3% in the placebo group), and most participants were White (64.5% in the prazosin group and 69.1% in the placebo group), with an average age of approximately 50 years. Primary outcomes included change from baseline to Week 10 in both CAPS item B2 (“recurrent distressing dreams”) and PSQI scores. CGIC score was evaluated at Week 10.
At Week 10, none of the primary outcomes were found to be statistically significant. The mean difference in change from baseline to Week 10 in CAPS item B2 score and PSQI score were 0.2 (P = .38) and 0.1 (P = .80), respectively. There was no significant difference in mean CGIC scores (P = .96). Repeated measures of CAPS item B2, PSQI, and CGIC scores were conducted through Week 26 as secondary outcomes. No significant differences were found. This study concluded that prazosin did not alleviate distressing dreams, improve sleep quality, or improve overall clinical symptoms.6
The PACT trial: Strengths and weaknesses
The PACT trial is the largest placebo-controlled trial for prazosin use in PTSD to date. It failed to show efficacy of prazosin for PTSD-associated nightmares, which contradicts previous studies. Although the mean total daily dose of prazosin was adequate and primary outcomes were measured with appropriate scales, the study failed to enroll the desired number of patients, which increased the possibility of false-negative results. Furthermore, participant recruitment may have led to selection bias because all participants were clinically stable, which could explain the lack of efficacy. However, the average CAPS scores were 80.7 in the prazosin group and 81.9 in the placebo group, which indicates that these patients had significant symptomatology at baseline and before entering the study.
Continue to: A major theme...
A major theme of studies evaluating prazosin treatment for PTSD is a focus on a military population and military-related trauma. Other than Taylor et al12 (N=13), none of these trials included patients who were diagnosed with PTSD due to other traumas, such as sexual trauma, which limits the generalizability of the results. Furthermore, apart from the PACT trial, none of these studies had >100 participants, which further reduces external validity. Current guidelines have not been updated to include the results of the PACT trial, and it is unclear if the results of this trial are strong enough to change clinical practice.
CASE CONTINUED
To ensure patient-centered care, the treating clinicians conduct a risk/benefit discussion with the patient regarding starting prazosin. Mr. H opts to try prazosin, so the clinicians initiate a low dose (1 mg/d) to mitigate adverse effects, and plan to titrate to clinical effect or intolerability. Per evidence from the trials discussed, it is likely Mr. H will need to be titrated to at least 5 to 6 mg/d to see a clinical effect.
Related Resource
North CS, Hong BA, Downs DL. PTSD: A systematic approach to diagnosis and treatment. Current Psychiatry 2018;17(4):35-43.
Drug Brand Names
Fluoxetine • Prozac
Prazosin • Minipress
Mr. H, age 43, presents to your clinic for management of posttraumatic stress disorder (PTSD). At his last appointment 8 weeks ago, he was continued on fluoxetine, 60 mg/d; he had been stable on this medication for 6 months. Today, Mr. H reports an increase in the frequency and severity of nightmares. He states that he wakes at least 3 times every week with “disturbing dreams” about his time in the military and does not feel rested even when he sleeps through the night. His Clinician-Administered PTSD Scale (CAPS) score is 95 on this visit, suggesting extreme PTSD symptomatology. Mr. H asks if anything can be done to reduce the frequency and intensity of his nightmares.
PTSD is the development of characteristic symptoms following exposure to ≥1 traumatic events. According to DSM-5, PTSD symptoms include the presence of ≥1 intrusion symptoms (recurrent, intrusive memories of the traumatic event; recurrent distressing dreams; dissociative reactions), persistent avoidance of stimuli, negative alterations in cognition and mood, and marked alterations in arousal and reactivity associated with the traumatic event(s).1 The symptoms must be present for >1 month, cause clinically significant distress or impairment in functioning, and not be attributable to the psychologic effects of a substance or medical conditions.1 This article focuses specifically on the hyperarousal symptoms, and the clinical controversies surrounding the use of prazosin for PTSD.
Prazosin for PTSD treatment
Sleep disorders are extremely common in patients with PTSD. Up to 90% of patients report sleep disturbances, and up to 70% report nightmares.2 Prazosin has been widely used in the treatment of PTSD-related sleep disorders and nightmares.The American Psychiatric Association3 and the British Association of Psychopharmacology4 guidelines in-clude prazosin as a first-line recommendation for treatment of PTSD. However, updated 2017 guidelines from the Veterans Affairs/Department of Defense (VA/DoD)5 and data from the 2018 Prazosin and Combat Trauma PTSD (PACT) trial6 contradict these original recommendations. Previously, the 2010 VA/DoD guideline said prazosin had insufficient evidence for monotherapy, but recommended it as adjunctive treatment for sleep and nightmares.7 The updated 2017 VA/DoD guideline recommends “weak against” prazosin use for global symptoms of PTSD, and says there is insufficient evidence for its use in nightmares.5 Below we summarize the findings of studies that contributed to those original recommendations, along with results of the PACT trial.
Raskind et al8,9 conducted 2 studies of prazosin use in combat veterans with PTSD. In both studies, prazosin had significant positive effects on the Clinician-Administered PTSD Scale (CAPS) and Clinical Global Impression of Change (CGIC) scores.8,9 The 2007 study also found significant effects of prazosin on Pittsburgh Sleep Quality Index (PSQI) scores.9
Raskind et al10 conducted another study in 2013 of prazosin use for active-duty soldiers who had combat trauma PTSD with nightmares. Prazosin had positive effects for nightmares, sleep quality, and CAPS scores.10
Germain et al11 reviewed prazosin for treating sleep disturbances in US military veterans. Prazosin was associated with significant improvements in insomnia and daytime PTSD symptom severity as demonstrated by changes in PSQI and CAPS scores.11
Taylor et al12 examined the effects of prazosin on sleep measures and clinical symptoms in civilians with PTSD. Prazosin significantly increased total sleep time, rapid eye movement sleep time, and CGIC scores while significantly decreasing trauma-related nightmares.12
Continue to: Overall, these trials...
Overall, these trials found efficacy for the use of prazosin for patients diagnosed with PTSD; however, the population size in each of these studies was small.
Results of the PACT trial
The PACT trial was a 26-week, multicenter, double-blind, randomized, placebo-controlled trial conducted across 12 VA medical centers.6 During the first 5 weeks, participants were randomized to receive placebo or prazosin, which could be titrated up to 20 mg/d in men and 12 mg/d in women. Participants remained on that dose from the end of Week 5 through Week 10. At that time, other pharmacologic therapies and psychotherapy could be added, discontinued, or adjusted. The mean maintenance total daily dose of prazosin was 14.8 mg.
A total of 413 patients were screened, 304 were randomized (152 per group), and 271 completed the 10-week primary outcome assessment. The population was almost entirely male (96.1% in the prazosin group and 99.3% in the placebo group), and most participants were White (64.5% in the prazosin group and 69.1% in the placebo group), with an average age of approximately 50 years. Primary outcomes included change from baseline to Week 10 in both CAPS item B2 (“recurrent distressing dreams”) and PSQI scores. CGIC score was evaluated at Week 10.
At Week 10, none of the primary outcomes were found to be statistically significant. The mean difference in change from baseline to Week 10 in CAPS item B2 score and PSQI score were 0.2 (P = .38) and 0.1 (P = .80), respectively. There was no significant difference in mean CGIC scores (P = .96). Repeated measures of CAPS item B2, PSQI, and CGIC scores were conducted through Week 26 as secondary outcomes. No significant differences were found. This study concluded that prazosin did not alleviate distressing dreams, improve sleep quality, or improve overall clinical symptoms.6
The PACT trial: Strengths and weaknesses
The PACT trial is the largest placebo-controlled trial for prazosin use in PTSD to date. It failed to show efficacy of prazosin for PTSD-associated nightmares, which contradicts previous studies. Although the mean total daily dose of prazosin was adequate and primary outcomes were measured with appropriate scales, the study failed to enroll the desired number of patients, which increased the possibility of false-negative results. Furthermore, participant recruitment may have led to selection bias because all participants were clinically stable, which could explain the lack of efficacy. However, the average CAPS scores were 80.7 in the prazosin group and 81.9 in the placebo group, which indicates that these patients had significant symptomatology at baseline and before entering the study.
Continue to: A major theme...
A major theme of studies evaluating prazosin treatment for PTSD is a focus on a military population and military-related trauma. Other than Taylor et al12 (N=13), none of these trials included patients who were diagnosed with PTSD due to other traumas, such as sexual trauma, which limits the generalizability of the results. Furthermore, apart from the PACT trial, none of these studies had >100 participants, which further reduces external validity. Current guidelines have not been updated to include the results of the PACT trial, and it is unclear if the results of this trial are strong enough to change clinical practice.
CASE CONTINUED
To ensure patient-centered care, the treating clinicians conduct a risk/benefit discussion with the patient regarding starting prazosin. Mr. H opts to try prazosin, so the clinicians initiate a low dose (1 mg/d) to mitigate adverse effects, and plan to titrate to clinical effect or intolerability. Per evidence from the trials discussed, it is likely Mr. H will need to be titrated to at least 5 to 6 mg/d to see a clinical effect.
Related Resource
North CS, Hong BA, Downs DL. PTSD: A systematic approach to diagnosis and treatment. Current Psychiatry 2018;17(4):35-43.
Drug Brand Names
Fluoxetine • Prozac
Prazosin • Minipress
1. Diagnostic and statistical manual of mental disorders, 5th ed. American Psychiatric Association; 2013.
2. Maher MJ, Rego SA, Asnis, GM. Sleep disturbances in patients with post-traumatic stress disorder: epidemiology, impact and approaches to management. CNS Drugs. 2006;20(7):567-590.
3. Benedek DM, Friedman MJ, Zatzick D, et al. Guideline watch (March 2009): Practice guideline for the treatment of patients with acute stress disorder and posttraumatic stress disorder. APA Practice Guidelines. Published 2010. Accessed March 14, 2021. https://psychiatryonline.org/pb/assets/raw/sitewide/practice_guidelines/guidelines/acutestressdisorderptsd-watch.pdf
4. Baldwin DS, Anderson IM, Nutt DJ, et al. Evidence-based pharmacological treatment of anxiety disorders, post-traumatic stress disorder and obsessive-compulsive disorder: a revision of the 2005 guidelines from the British Association for Psychopharmacology. J Psychopharmacol. 2014;28(5):403-439. doi: 10.1177/0269881114525674
5. Department of Veterans Affairs, Department of Defense. VA/DoD clinical practice guideline for the management of posttraumatic stress disorder and acute stress disorder. Version 3.0. Published 2017. Accessed February 5, 2021. https://www.healthquality.va.gov/guidelines/MH/ptsd/VADoDPTSDCPGFinal012418.pdf
6. Raskind MA, Peskind ER, Chow B, et al. Trial of prazosin for post-traumatic stress disorder in military veterans. N Engl J Med. 2018;378(6):507-517.
7. Department of Veterans Affairs, Department of Defense. VA/DoD clinical practice guideline: management of post-traumatic stress. Version 2.0. Published 2010. Accessed February 5, 2021. https://www.healthquality.va.gov/guidelines/MH/ptsd/cpg_PTSD-full-201011612.PDF
8. Raskind MA, Peskind ER, Katner ED, et al. Reduction of nightmares and other PTSD symptoms in combat veterans by prazosin: a placebo-controlled study. Am J Psychiatry. 2003;160(2):371-373.
9. Raskind MA, Peskind ER, Hoff DJ, et al. A parallel group placebo-controlled study of prazosin for trauma nightmares and sleep disturbance in combat veterans with post-traumatic stress disorder. Biol Psychiatry. 2007;61(8):928-934.
10. Raskind MA, Peterson K, Williams T, et al. A trial of prazosin for combat trauma PTSD with nightmares in active-duty soldiers returned from Iraq and Afghanistan. Am J Psychiatry. 2013;170(9):1003-1010.
11. Germain A, Richardson R, Moul DE, et al. Placebo-controlled comparison of prazosin and cognitive-behavioral treatments for sleep disturbances in US military veterans. J Psychosom Res. 2012;72(2):89-96.
12. Taylor FB, Martin P, Thompson C, et al. Prazosin effects on objective sleep measures and clinical symptoms in civilian trauma posttraumatic stress disorder: a placebo-controlled study. Biol Psychiatry. 2008;63(6):629-632.
1. Diagnostic and statistical manual of mental disorders, 5th ed. American Psychiatric Association; 2013.
2. Maher MJ, Rego SA, Asnis, GM. Sleep disturbances in patients with post-traumatic stress disorder: epidemiology, impact and approaches to management. CNS Drugs. 2006;20(7):567-590.
3. Benedek DM, Friedman MJ, Zatzick D, et al. Guideline watch (March 2009): Practice guideline for the treatment of patients with acute stress disorder and posttraumatic stress disorder. APA Practice Guidelines. Published 2010. Accessed March 14, 2021. https://psychiatryonline.org/pb/assets/raw/sitewide/practice_guidelines/guidelines/acutestressdisorderptsd-watch.pdf
4. Baldwin DS, Anderson IM, Nutt DJ, et al. Evidence-based pharmacological treatment of anxiety disorders, post-traumatic stress disorder and obsessive-compulsive disorder: a revision of the 2005 guidelines from the British Association for Psychopharmacology. J Psychopharmacol. 2014;28(5):403-439. doi: 10.1177/0269881114525674
5. Department of Veterans Affairs, Department of Defense. VA/DoD clinical practice guideline for the management of posttraumatic stress disorder and acute stress disorder. Version 3.0. Published 2017. Accessed February 5, 2021. https://www.healthquality.va.gov/guidelines/MH/ptsd/VADoDPTSDCPGFinal012418.pdf
6. Raskind MA, Peskind ER, Chow B, et al. Trial of prazosin for post-traumatic stress disorder in military veterans. N Engl J Med. 2018;378(6):507-517.
7. Department of Veterans Affairs, Department of Defense. VA/DoD clinical practice guideline: management of post-traumatic stress. Version 2.0. Published 2010. Accessed February 5, 2021. https://www.healthquality.va.gov/guidelines/MH/ptsd/cpg_PTSD-full-201011612.PDF
8. Raskind MA, Peskind ER, Katner ED, et al. Reduction of nightmares and other PTSD symptoms in combat veterans by prazosin: a placebo-controlled study. Am J Psychiatry. 2003;160(2):371-373.
9. Raskind MA, Peskind ER, Hoff DJ, et al. A parallel group placebo-controlled study of prazosin for trauma nightmares and sleep disturbance in combat veterans with post-traumatic stress disorder. Biol Psychiatry. 2007;61(8):928-934.
10. Raskind MA, Peterson K, Williams T, et al. A trial of prazosin for combat trauma PTSD with nightmares in active-duty soldiers returned from Iraq and Afghanistan. Am J Psychiatry. 2013;170(9):1003-1010.
11. Germain A, Richardson R, Moul DE, et al. Placebo-controlled comparison of prazosin and cognitive-behavioral treatments for sleep disturbances in US military veterans. J Psychosom Res. 2012;72(2):89-96.
12. Taylor FB, Martin P, Thompson C, et al. Prazosin effects on objective sleep measures and clinical symptoms in civilian trauma posttraumatic stress disorder: a placebo-controlled study. Biol Psychiatry. 2008;63(6):629-632.
High-dose lumateperone: A case report
Lumateperone is a novel antipsychotic that possesses a variety of unique receptor affinities. The recommended dose of lumateperone is 42 mg/d. In clinical trials, reductions in Positive and Negative Syndrome Scale scores observed with lumateperone, 28 mg/d and 84 mg/d, failed to separate from placebo.1 However, in these trials, safety profiles were similar for all 3 doses.
Despite the popular understanding of lumateperone’s “unexplained narrow therapeutic window,”2 we report the case of a patient with schizophrenia who responded well to lumateperone, 84 mg/d, without adverse effects or EKG changes.
Case report. Mr. W, age 26, has treatment-resistant schizophrenia (paranoid type). He failed to achieve remission on fluphenazine (10 to 25 mg/d), perphenazine (4 to 24 mg/d), risperidone (started at 4 mg/d and increased to 8 mg/d), and olanzapine (15, 20, and 25 mg/d). None of these medications eliminated his auditory or visual hallucinations. His response was most robust to perphenazine, as he reported a 50% reduction in the frequency of auditory hallucinations and a near-complete resolution of visual hallucinations (once or twice per week), but he never achieved full remission.
We started lumateperone, 42 mg/d, without a cross-taper. After 4 weeks of partial response, the patient escalated his dose to 84 mg/d on his own. At a follow-up visit 3.5 weeks after this self-directed dose increase, Mr. W reported a complete resolution of his auditory and visual hallucinations.
Six months later, Mr. W continued to receive lumateperone, 84 mg/d, without extrapyramidal symptoms, tardive dyskinesia, or other adverse effects. His QTc showed no significant change (410 ms vs 412 ms).
Although some studies indicate a possible “therapeutic window” for lumateperone dosing, clinicians should not deprive patients who partially respond to the recommended 42 mg/d dose of the opportunity for additional benefit through dose escalation. Due to the vagaries of psychiatric pathology, and unique profiles of metabolism and receptor sensitivity, there will always be patients who may require higher-than-recommended doses of lumateperone, as with all other agents.
1. Lieberman JA, Davis RE, Correll CU, et al. ITI-007 for the treatment of schizophrenia: a 4-week randomized, double-blind, controlled trial. Biol Psychiatry. 2016;79(12):952-961. doi: 10.1016/j.biopsych.2015.08.026
2. Kantrowitz JT. The potential role of lumateperone—something borrowed? something new? JAMA Psychiatry. 2020;77(4):343-344. doi:10.1001/jamapsychiatry.2019.4265
Lumateperone is a novel antipsychotic that possesses a variety of unique receptor affinities. The recommended dose of lumateperone is 42 mg/d. In clinical trials, reductions in Positive and Negative Syndrome Scale scores observed with lumateperone, 28 mg/d and 84 mg/d, failed to separate from placebo.1 However, in these trials, safety profiles were similar for all 3 doses.
Despite the popular understanding of lumateperone’s “unexplained narrow therapeutic window,”2 we report the case of a patient with schizophrenia who responded well to lumateperone, 84 mg/d, without adverse effects or EKG changes.
Case report. Mr. W, age 26, has treatment-resistant schizophrenia (paranoid type). He failed to achieve remission on fluphenazine (10 to 25 mg/d), perphenazine (4 to 24 mg/d), risperidone (started at 4 mg/d and increased to 8 mg/d), and olanzapine (15, 20, and 25 mg/d). None of these medications eliminated his auditory or visual hallucinations. His response was most robust to perphenazine, as he reported a 50% reduction in the frequency of auditory hallucinations and a near-complete resolution of visual hallucinations (once or twice per week), but he never achieved full remission.
We started lumateperone, 42 mg/d, without a cross-taper. After 4 weeks of partial response, the patient escalated his dose to 84 mg/d on his own. At a follow-up visit 3.5 weeks after this self-directed dose increase, Mr. W reported a complete resolution of his auditory and visual hallucinations.
Six months later, Mr. W continued to receive lumateperone, 84 mg/d, without extrapyramidal symptoms, tardive dyskinesia, or other adverse effects. His QTc showed no significant change (410 ms vs 412 ms).
Although some studies indicate a possible “therapeutic window” for lumateperone dosing, clinicians should not deprive patients who partially respond to the recommended 42 mg/d dose of the opportunity for additional benefit through dose escalation. Due to the vagaries of psychiatric pathology, and unique profiles of metabolism and receptor sensitivity, there will always be patients who may require higher-than-recommended doses of lumateperone, as with all other agents.
Lumateperone is a novel antipsychotic that possesses a variety of unique receptor affinities. The recommended dose of lumateperone is 42 mg/d. In clinical trials, reductions in Positive and Negative Syndrome Scale scores observed with lumateperone, 28 mg/d and 84 mg/d, failed to separate from placebo.1 However, in these trials, safety profiles were similar for all 3 doses.
Despite the popular understanding of lumateperone’s “unexplained narrow therapeutic window,”2 we report the case of a patient with schizophrenia who responded well to lumateperone, 84 mg/d, without adverse effects or EKG changes.
Case report. Mr. W, age 26, has treatment-resistant schizophrenia (paranoid type). He failed to achieve remission on fluphenazine (10 to 25 mg/d), perphenazine (4 to 24 mg/d), risperidone (started at 4 mg/d and increased to 8 mg/d), and olanzapine (15, 20, and 25 mg/d). None of these medications eliminated his auditory or visual hallucinations. His response was most robust to perphenazine, as he reported a 50% reduction in the frequency of auditory hallucinations and a near-complete resolution of visual hallucinations (once or twice per week), but he never achieved full remission.
We started lumateperone, 42 mg/d, without a cross-taper. After 4 weeks of partial response, the patient escalated his dose to 84 mg/d on his own. At a follow-up visit 3.5 weeks after this self-directed dose increase, Mr. W reported a complete resolution of his auditory and visual hallucinations.
Six months later, Mr. W continued to receive lumateperone, 84 mg/d, without extrapyramidal symptoms, tardive dyskinesia, or other adverse effects. His QTc showed no significant change (410 ms vs 412 ms).
Although some studies indicate a possible “therapeutic window” for lumateperone dosing, clinicians should not deprive patients who partially respond to the recommended 42 mg/d dose of the opportunity for additional benefit through dose escalation. Due to the vagaries of psychiatric pathology, and unique profiles of metabolism and receptor sensitivity, there will always be patients who may require higher-than-recommended doses of lumateperone, as with all other agents.
1. Lieberman JA, Davis RE, Correll CU, et al. ITI-007 for the treatment of schizophrenia: a 4-week randomized, double-blind, controlled trial. Biol Psychiatry. 2016;79(12):952-961. doi: 10.1016/j.biopsych.2015.08.026
2. Kantrowitz JT. The potential role of lumateperone—something borrowed? something new? JAMA Psychiatry. 2020;77(4):343-344. doi:10.1001/jamapsychiatry.2019.4265
1. Lieberman JA, Davis RE, Correll CU, et al. ITI-007 for the treatment of schizophrenia: a 4-week randomized, double-blind, controlled trial. Biol Psychiatry. 2016;79(12):952-961. doi: 10.1016/j.biopsych.2015.08.026
2. Kantrowitz JT. The potential role of lumateperone—something borrowed? something new? JAMA Psychiatry. 2020;77(4):343-344. doi:10.1001/jamapsychiatry.2019.4265
Switching antipsychotics: A guide to dose equivalents
Chlorpromazine (CPZ), a low-potency first-generation antipsychotic (FGA), was the first medication approved for the management of schizophrenia. Since its approval, some psychiatrists have prescribed subsequent antipsychotics based on CPZ’s efficacy and dosing. Comparing dosages of newer antipsychotics using a CPZ equivalent as a baseline remains a relevant method of determining which agent to prescribe, and at what dose.1,2
Psychiatrists frequently care for patients who are treatment-refractory or older adults with poor medication tolerance and age-related medical illness. Quick access to the comparative potency of different antipsychotics can help guide titration to the approximate equivalent dose of CPZ when initiating a medication, switching from 1 antipsychotic to another, or augmenting or combining antipsychotics. Fortunately, many authors, such as Woods2and Davis,3 have codified the dosing ratio equivalences of FGAs and second-generation antipsychotics (SGAs) using CPZ, 100 mg. To help psychiatrists use CPZ dosages as a point of comparison for prescribing other antipsychotics, the Table1,2,4 (page 14) lists dose equivalents for oral FGAs and SGAs based on CPZ, 100 mg. (For information on dose equivalents for injectable antipsychotics, see “Second-generation long-acting injectable antipsychotics: A practical guide,”
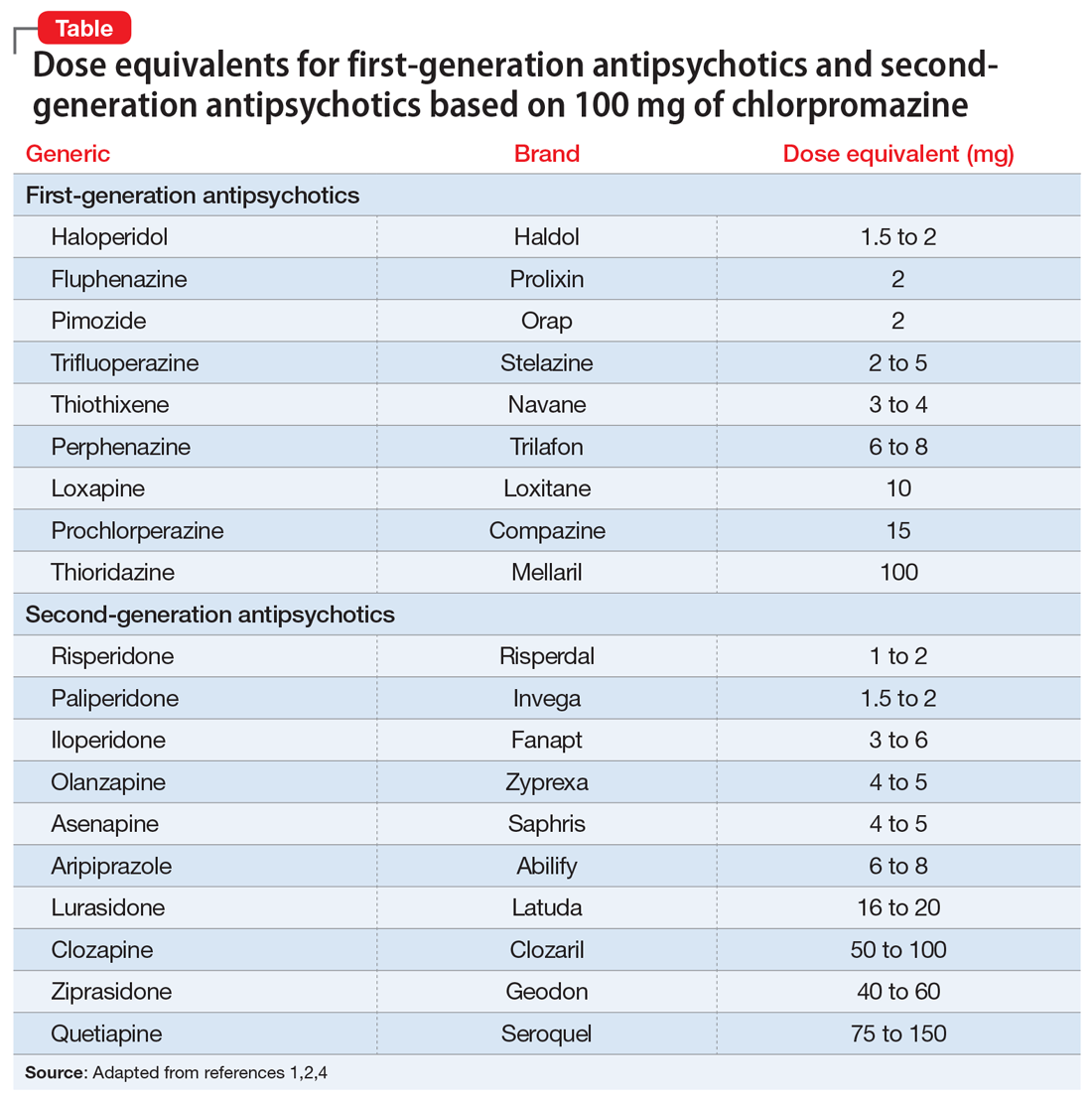
While this information cannot replace a psychiatrist’s clinical judgment, it can serve as a clinically useful prescribing tool. In addition to providing this Table, we discuss what you should consider when using these equivalents to switch antipsychotics and estimate the ultimate dose target for effective management of psychotic disorders.
A few caveats
Bioactive equivalent dosages should be targeted as a rough guide when switching from one FGA or SGA to another. Common indications for switching antipsychotics include an inadequate therapeutic response after a medication trial of an adequate dose and duration; relapse of psychosis despite medication adherence; intolerable adverse effects; cost; a new-onset, contraindicating medical illness; and lapses in medication compliance that necessitate a change to IM formulations.5 Keep in mind that medication changes should be tailored to the patient’s specific clinical characteristics.
Several other clinical and pharmacologic variabilities should be kept in mind when switching antipsychotics using CPZ dosage equivalents5,6:
- The therapeutic CPZ equivalent doses may be less precise for SGAs than for FGAs because the equivalents are largely based on dopaminergic blockade instead of cholinergic, serotonergic, or histaminergic systems
- For some antipsychotics, the relationship between dose and potency is nonlinear. For example, as the dosage of haloperidol increases, its relative antipsychotic potency decreases
- Differences in half-lives between 2 agents can add complexity to calculating the dosage equivalent
- Regardless of comparative dosing, before initiating a new antipsychotic, psychiatrists should read the dosing instructions in the FDA-approved package insert, and exercise caution before titrating a new medication to the maximum recommended dose.
1. Danivas V, Venkatasubramanian G. Current perspectives on chlorpromazine equivalents: comparing apples and oranges! Indian J Psychiatry. 2013;55(2):207-208.
2. Woods SW. Chlorpromazine equivalent doses for the newer atypical antipsychotics. J Clin Psychiatry. 2003;64(6):663-667.
3. Davis JM. Dose equivalence of the anti-psychotic drugs. J Psych Res. 1974;11:65-69.
4. Psychiatric pharmacy essentials: antipsychotic dose equivalents. College of Psychiatric and Neurologic Pharmacists. Accessed February 2, 2021. https://cpnp.org/guideline/essentials/antipsychotic-dose-equivalents
5. Guidelines for antipsychotic medication switches. Humber NHS. Last Reviewed September 2012. Accessed February 2, 2021. https://www.psychdb.com/_media/meds/antipsychotics/nhs_guidelines_antipsychotic_switch.pdf
6. Bobo WV. Switching antipsychotics: why, when, and how? Psychiatric Times. Published March 14, 2013. Accessed February 2, 2021. https://www.psychiatrictimes.com/view/switching-antipsychotics-why-when-and-how
Chlorpromazine (CPZ), a low-potency first-generation antipsychotic (FGA), was the first medication approved for the management of schizophrenia. Since its approval, some psychiatrists have prescribed subsequent antipsychotics based on CPZ’s efficacy and dosing. Comparing dosages of newer antipsychotics using a CPZ equivalent as a baseline remains a relevant method of determining which agent to prescribe, and at what dose.1,2
Psychiatrists frequently care for patients who are treatment-refractory or older adults with poor medication tolerance and age-related medical illness. Quick access to the comparative potency of different antipsychotics can help guide titration to the approximate equivalent dose of CPZ when initiating a medication, switching from 1 antipsychotic to another, or augmenting or combining antipsychotics. Fortunately, many authors, such as Woods2and Davis,3 have codified the dosing ratio equivalences of FGAs and second-generation antipsychotics (SGAs) using CPZ, 100 mg. To help psychiatrists use CPZ dosages as a point of comparison for prescribing other antipsychotics, the Table1,2,4 (page 14) lists dose equivalents for oral FGAs and SGAs based on CPZ, 100 mg. (For information on dose equivalents for injectable antipsychotics, see “Second-generation long-acting injectable antipsychotics: A practical guide,”

While this information cannot replace a psychiatrist’s clinical judgment, it can serve as a clinically useful prescribing tool. In addition to providing this Table, we discuss what you should consider when using these equivalents to switch antipsychotics and estimate the ultimate dose target for effective management of psychotic disorders.
A few caveats
Bioactive equivalent dosages should be targeted as a rough guide when switching from one FGA or SGA to another. Common indications for switching antipsychotics include an inadequate therapeutic response after a medication trial of an adequate dose and duration; relapse of psychosis despite medication adherence; intolerable adverse effects; cost; a new-onset, contraindicating medical illness; and lapses in medication compliance that necessitate a change to IM formulations.5 Keep in mind that medication changes should be tailored to the patient’s specific clinical characteristics.
Several other clinical and pharmacologic variabilities should be kept in mind when switching antipsychotics using CPZ dosage equivalents5,6:
- The therapeutic CPZ equivalent doses may be less precise for SGAs than for FGAs because the equivalents are largely based on dopaminergic blockade instead of cholinergic, serotonergic, or histaminergic systems
- For some antipsychotics, the relationship between dose and potency is nonlinear. For example, as the dosage of haloperidol increases, its relative antipsychotic potency decreases
- Differences in half-lives between 2 agents can add complexity to calculating the dosage equivalent
- Regardless of comparative dosing, before initiating a new antipsychotic, psychiatrists should read the dosing instructions in the FDA-approved package insert, and exercise caution before titrating a new medication to the maximum recommended dose.
Chlorpromazine (CPZ), a low-potency first-generation antipsychotic (FGA), was the first medication approved for the management of schizophrenia. Since its approval, some psychiatrists have prescribed subsequent antipsychotics based on CPZ’s efficacy and dosing. Comparing dosages of newer antipsychotics using a CPZ equivalent as a baseline remains a relevant method of determining which agent to prescribe, and at what dose.1,2
Psychiatrists frequently care for patients who are treatment-refractory or older adults with poor medication tolerance and age-related medical illness. Quick access to the comparative potency of different antipsychotics can help guide titration to the approximate equivalent dose of CPZ when initiating a medication, switching from 1 antipsychotic to another, or augmenting or combining antipsychotics. Fortunately, many authors, such as Woods2and Davis,3 have codified the dosing ratio equivalences of FGAs and second-generation antipsychotics (SGAs) using CPZ, 100 mg. To help psychiatrists use CPZ dosages as a point of comparison for prescribing other antipsychotics, the Table1,2,4 (page 14) lists dose equivalents for oral FGAs and SGAs based on CPZ, 100 mg. (For information on dose equivalents for injectable antipsychotics, see “Second-generation long-acting injectable antipsychotics: A practical guide,”

While this information cannot replace a psychiatrist’s clinical judgment, it can serve as a clinically useful prescribing tool. In addition to providing this Table, we discuss what you should consider when using these equivalents to switch antipsychotics and estimate the ultimate dose target for effective management of psychotic disorders.
A few caveats
Bioactive equivalent dosages should be targeted as a rough guide when switching from one FGA or SGA to another. Common indications for switching antipsychotics include an inadequate therapeutic response after a medication trial of an adequate dose and duration; relapse of psychosis despite medication adherence; intolerable adverse effects; cost; a new-onset, contraindicating medical illness; and lapses in medication compliance that necessitate a change to IM formulations.5 Keep in mind that medication changes should be tailored to the patient’s specific clinical characteristics.
Several other clinical and pharmacologic variabilities should be kept in mind when switching antipsychotics using CPZ dosage equivalents5,6:
- The therapeutic CPZ equivalent doses may be less precise for SGAs than for FGAs because the equivalents are largely based on dopaminergic blockade instead of cholinergic, serotonergic, or histaminergic systems
- For some antipsychotics, the relationship between dose and potency is nonlinear. For example, as the dosage of haloperidol increases, its relative antipsychotic potency decreases
- Differences in half-lives between 2 agents can add complexity to calculating the dosage equivalent
- Regardless of comparative dosing, before initiating a new antipsychotic, psychiatrists should read the dosing instructions in the FDA-approved package insert, and exercise caution before titrating a new medication to the maximum recommended dose.
1. Danivas V, Venkatasubramanian G. Current perspectives on chlorpromazine equivalents: comparing apples and oranges! Indian J Psychiatry. 2013;55(2):207-208.
2. Woods SW. Chlorpromazine equivalent doses for the newer atypical antipsychotics. J Clin Psychiatry. 2003;64(6):663-667.
3. Davis JM. Dose equivalence of the anti-psychotic drugs. J Psych Res. 1974;11:65-69.
4. Psychiatric pharmacy essentials: antipsychotic dose equivalents. College of Psychiatric and Neurologic Pharmacists. Accessed February 2, 2021. https://cpnp.org/guideline/essentials/antipsychotic-dose-equivalents
5. Guidelines for antipsychotic medication switches. Humber NHS. Last Reviewed September 2012. Accessed February 2, 2021. https://www.psychdb.com/_media/meds/antipsychotics/nhs_guidelines_antipsychotic_switch.pdf
6. Bobo WV. Switching antipsychotics: why, when, and how? Psychiatric Times. Published March 14, 2013. Accessed February 2, 2021. https://www.psychiatrictimes.com/view/switching-antipsychotics-why-when-and-how
1. Danivas V, Venkatasubramanian G. Current perspectives on chlorpromazine equivalents: comparing apples and oranges! Indian J Psychiatry. 2013;55(2):207-208.
2. Woods SW. Chlorpromazine equivalent doses for the newer atypical antipsychotics. J Clin Psychiatry. 2003;64(6):663-667.
3. Davis JM. Dose equivalence of the anti-psychotic drugs. J Psych Res. 1974;11:65-69.
4. Psychiatric pharmacy essentials: antipsychotic dose equivalents. College of Psychiatric and Neurologic Pharmacists. Accessed February 2, 2021. https://cpnp.org/guideline/essentials/antipsychotic-dose-equivalents
5. Guidelines for antipsychotic medication switches. Humber NHS. Last Reviewed September 2012. Accessed February 2, 2021. https://www.psychdb.com/_media/meds/antipsychotics/nhs_guidelines_antipsychotic_switch.pdf
6. Bobo WV. Switching antipsychotics: why, when, and how? Psychiatric Times. Published March 14, 2013. Accessed February 2, 2021. https://www.psychiatrictimes.com/view/switching-antipsychotics-why-when-and-how
Your patient refuses a suicide risk assessment. Now what?
On occasion, a patient may refuse to cooperate with a suicide risk assessment or is unable to participate due to the severity of a psychiatric or medical condition. In such situations, how can we conduct an assessment that meets our ethical, professional, and legal obligations?
First, skipping a suicide risk assessment is never an option. A patient’s refusal or inability to cooperate does not release us from our duty of care. We are obligated to gather information about suicide risk to anticipate the likelihood and severity of harm.1 Furthermore, collecting information helps us evaluate what types of precautions are necessary to reduce or eliminate suicide risk.
Some clinicians may believe that a suicide risk assessment is only possible when they can ask patients about ideation, intent, plans, and past suicidal behavior. While the patient’s self-report is valuable, it is only one data point, and in some cases, it may not be reliable or credible.2 So how should you handle such situations? Here I describe 3 steps to take to estimate a patient’s suicide risk without their participation.
1. Obtain information from other sources.
These can include:
- your recent contacts with the patient
- the patient’s responses to previous inquiries about suicidality
- collateral reports from staff
- the patient’s chart and past medical records
- past suicide attempts (including the precipitants, the patient’s reasons for the attempt, details of the actions taken and methods used, any medical outcome, and the patient’s reaction to surviving)3
- past nonsuicidal self-injury
- past episodes of suicidal thinking
- treatment progress to date
- mental status.
Documenting your sources of information will indicate that you made reasonable efforts to appreciate the risk despite imperfect circumstances. Furthermore, these sources of data can support your work to assess the severity of the patient’s current suicidality, to clinically formulate why the patient is susceptible to suicidal thoughts and behavior, and to anticipate circumstances that could constitute a high-risk period for your patient to attempt suicide.
2. Document the reasons you were unable to interview the patient. For patients who are competent to refuse services, document the efforts you made to gain the patient’s cooperation. If the patient’s psychiatric condition (eg, florid psychosis) was the main impediment, note this.
3. Explain the limitations of your assessment. This might include acknowledging that your estimation of the patient’s suicide risk is missing important information but is the best possible estimate at the time. Explain how you determined the level of risk with a statement such as, “Because the patient was unable to participate, I estimated risk based on….” If the patient’s lack of participation lowers your confidence in your risk estimate, this also should be documented. Reduced confidence may indicate the need for additional steps to assure the patient’s safety (eg, admission, delaying discharge, initiating continuous observation).
1. Obegi JH. Probable standards of care for suicide risk assessment. J Am Acad Psychiatry Law. 2017;45(4):452-459.
2. Hom MA, Stanley IH, Duffy ME, et al. Investigating the reliability of suicide attempt history reporting across five measures: a study of US military service members at risk of suicide. J Clin Psychol. 2019;75(7):1332-1349.
3. Rudd MD. Core competencies, warning signs, and a framework for suicide risk assessment in clinical practice. In: Nock MK, ed. The Oxford handbook of suicide and self-injury. Oxford University Press; 2014:323-336.
On occasion, a patient may refuse to cooperate with a suicide risk assessment or is unable to participate due to the severity of a psychiatric or medical condition. In such situations, how can we conduct an assessment that meets our ethical, professional, and legal obligations?
First, skipping a suicide risk assessment is never an option. A patient’s refusal or inability to cooperate does not release us from our duty of care. We are obligated to gather information about suicide risk to anticipate the likelihood and severity of harm.1 Furthermore, collecting information helps us evaluate what types of precautions are necessary to reduce or eliminate suicide risk.
Some clinicians may believe that a suicide risk assessment is only possible when they can ask patients about ideation, intent, plans, and past suicidal behavior. While the patient’s self-report is valuable, it is only one data point, and in some cases, it may not be reliable or credible.2 So how should you handle such situations? Here I describe 3 steps to take to estimate a patient’s suicide risk without their participation.
1. Obtain information from other sources.
These can include:
- your recent contacts with the patient
- the patient’s responses to previous inquiries about suicidality
- collateral reports from staff
- the patient’s chart and past medical records
- past suicide attempts (including the precipitants, the patient’s reasons for the attempt, details of the actions taken and methods used, any medical outcome, and the patient’s reaction to surviving)3
- past nonsuicidal self-injury
- past episodes of suicidal thinking
- treatment progress to date
- mental status.
Documenting your sources of information will indicate that you made reasonable efforts to appreciate the risk despite imperfect circumstances. Furthermore, these sources of data can support your work to assess the severity of the patient’s current suicidality, to clinically formulate why the patient is susceptible to suicidal thoughts and behavior, and to anticipate circumstances that could constitute a high-risk period for your patient to attempt suicide.
2. Document the reasons you were unable to interview the patient. For patients who are competent to refuse services, document the efforts you made to gain the patient’s cooperation. If the patient’s psychiatric condition (eg, florid psychosis) was the main impediment, note this.
3. Explain the limitations of your assessment. This might include acknowledging that your estimation of the patient’s suicide risk is missing important information but is the best possible estimate at the time. Explain how you determined the level of risk with a statement such as, “Because the patient was unable to participate, I estimated risk based on….” If the patient’s lack of participation lowers your confidence in your risk estimate, this also should be documented. Reduced confidence may indicate the need for additional steps to assure the patient’s safety (eg, admission, delaying discharge, initiating continuous observation).
On occasion, a patient may refuse to cooperate with a suicide risk assessment or is unable to participate due to the severity of a psychiatric or medical condition. In such situations, how can we conduct an assessment that meets our ethical, professional, and legal obligations?
First, skipping a suicide risk assessment is never an option. A patient’s refusal or inability to cooperate does not release us from our duty of care. We are obligated to gather information about suicide risk to anticipate the likelihood and severity of harm.1 Furthermore, collecting information helps us evaluate what types of precautions are necessary to reduce or eliminate suicide risk.
Some clinicians may believe that a suicide risk assessment is only possible when they can ask patients about ideation, intent, plans, and past suicidal behavior. While the patient’s self-report is valuable, it is only one data point, and in some cases, it may not be reliable or credible.2 So how should you handle such situations? Here I describe 3 steps to take to estimate a patient’s suicide risk without their participation.
1. Obtain information from other sources.
These can include:
- your recent contacts with the patient
- the patient’s responses to previous inquiries about suicidality
- collateral reports from staff
- the patient’s chart and past medical records
- past suicide attempts (including the precipitants, the patient’s reasons for the attempt, details of the actions taken and methods used, any medical outcome, and the patient’s reaction to surviving)3
- past nonsuicidal self-injury
- past episodes of suicidal thinking
- treatment progress to date
- mental status.
Documenting your sources of information will indicate that you made reasonable efforts to appreciate the risk despite imperfect circumstances. Furthermore, these sources of data can support your work to assess the severity of the patient’s current suicidality, to clinically formulate why the patient is susceptible to suicidal thoughts and behavior, and to anticipate circumstances that could constitute a high-risk period for your patient to attempt suicide.
2. Document the reasons you were unable to interview the patient. For patients who are competent to refuse services, document the efforts you made to gain the patient’s cooperation. If the patient’s psychiatric condition (eg, florid psychosis) was the main impediment, note this.
3. Explain the limitations of your assessment. This might include acknowledging that your estimation of the patient’s suicide risk is missing important information but is the best possible estimate at the time. Explain how you determined the level of risk with a statement such as, “Because the patient was unable to participate, I estimated risk based on….” If the patient’s lack of participation lowers your confidence in your risk estimate, this also should be documented. Reduced confidence may indicate the need for additional steps to assure the patient’s safety (eg, admission, delaying discharge, initiating continuous observation).
1. Obegi JH. Probable standards of care for suicide risk assessment. J Am Acad Psychiatry Law. 2017;45(4):452-459.
2. Hom MA, Stanley IH, Duffy ME, et al. Investigating the reliability of suicide attempt history reporting across five measures: a study of US military service members at risk of suicide. J Clin Psychol. 2019;75(7):1332-1349.
3. Rudd MD. Core competencies, warning signs, and a framework for suicide risk assessment in clinical practice. In: Nock MK, ed. The Oxford handbook of suicide and self-injury. Oxford University Press; 2014:323-336.
1. Obegi JH. Probable standards of care for suicide risk assessment. J Am Acad Psychiatry Law. 2017;45(4):452-459.
2. Hom MA, Stanley IH, Duffy ME, et al. Investigating the reliability of suicide attempt history reporting across five measures: a study of US military service members at risk of suicide. J Clin Psychol. 2019;75(7):1332-1349.
3. Rudd MD. Core competencies, warning signs, and a framework for suicide risk assessment in clinical practice. In: Nock MK, ed. The Oxford handbook of suicide and self-injury. Oxford University Press; 2014:323-336.
CDC adds new medical conditions to COVID-19 high-risk list
The Centers for Disease Control and Prevention has added several new medical conditions to its list of those that predispose adults to more severe COVID-19 illness.
Conditions that had previously been categorized as “might be” placing individuals at increased risk – but now are listed as high risk – include type 1 diabetes (in addition to type 2), moderate-to-severe asthma, liver disease, dementia or other neurologic conditions, stroke/cerebrovascular disease, HIV infection, cystic fibrosis, and overweight (in addition to obesity).
Substance use disorders, which hadn’t been previously listed, are now also considered high risk.
The new list groups together certain categories, such as chronic lung diseases (chronic obstructive pulmonary disease, asthma, cystic fibrosis, etc) and heart conditions (heart failure, coronary artery disease, hypertension, etc).
Both diabetes types are now grouped under “diabetes.”
The added medical conditions were posted on the CDC website’s COVID-19 page on March 29.
Type 1 diabetes and other conditions now priority for vaccination
The CDC refers to the medical conditions list as phase 1c in regard to COVID-19 vaccine prioritization, which means that anyone with any of these conditions can now be prioritized for vaccination, following those in groups 1a (frontline essential workers and those in long-term care facilities) and 1b (people aged 65-74 years; other essential workers; and people aged 16-64 years with underlying conditions that increase the risk of serious, life-threatening complications from COVID-19).
But in many cases, multiple states have already either fully opened up vaccine eligibility to all adults or have created their own lists of underlying high-risk medical conditions, CDC spokeswoman Kristen Nordlund told this news organization.
No conditions have been removed from the list.
In January, the American Diabetes Association and 18 other organizations sent a letter to the CDC requesting that type 1 diabetes be prioritized along with type 2, based on data from studies showing people with both types to be at high risk for severe COVID-19 illness.
Now, ADA says, “this updated guidance will help to address the fact that in many states, millions of people with type 1 diabetes have not been prioritized equally, slowing their access to critical vaccines.”
While awaiting this latest CDC move, ADA had been urging state governors to prioritize type 1 and type 2 diabetes equally. As of now, 38 states and the District of Columbia had either done so or announced that they would.
A version of this article first appeared on Medscape.com.
The Centers for Disease Control and Prevention has added several new medical conditions to its list of those that predispose adults to more severe COVID-19 illness.
Conditions that had previously been categorized as “might be” placing individuals at increased risk – but now are listed as high risk – include type 1 diabetes (in addition to type 2), moderate-to-severe asthma, liver disease, dementia or other neurologic conditions, stroke/cerebrovascular disease, HIV infection, cystic fibrosis, and overweight (in addition to obesity).
Substance use disorders, which hadn’t been previously listed, are now also considered high risk.
The new list groups together certain categories, such as chronic lung diseases (chronic obstructive pulmonary disease, asthma, cystic fibrosis, etc) and heart conditions (heart failure, coronary artery disease, hypertension, etc).
Both diabetes types are now grouped under “diabetes.”
The added medical conditions were posted on the CDC website’s COVID-19 page on March 29.
Type 1 diabetes and other conditions now priority for vaccination
The CDC refers to the medical conditions list as phase 1c in regard to COVID-19 vaccine prioritization, which means that anyone with any of these conditions can now be prioritized for vaccination, following those in groups 1a (frontline essential workers and those in long-term care facilities) and 1b (people aged 65-74 years; other essential workers; and people aged 16-64 years with underlying conditions that increase the risk of serious, life-threatening complications from COVID-19).
But in many cases, multiple states have already either fully opened up vaccine eligibility to all adults or have created their own lists of underlying high-risk medical conditions, CDC spokeswoman Kristen Nordlund told this news organization.
No conditions have been removed from the list.
In January, the American Diabetes Association and 18 other organizations sent a letter to the CDC requesting that type 1 diabetes be prioritized along with type 2, based on data from studies showing people with both types to be at high risk for severe COVID-19 illness.
Now, ADA says, “this updated guidance will help to address the fact that in many states, millions of people with type 1 diabetes have not been prioritized equally, slowing their access to critical vaccines.”
While awaiting this latest CDC move, ADA had been urging state governors to prioritize type 1 and type 2 diabetes equally. As of now, 38 states and the District of Columbia had either done so or announced that they would.
A version of this article first appeared on Medscape.com.
The Centers for Disease Control and Prevention has added several new medical conditions to its list of those that predispose adults to more severe COVID-19 illness.
Conditions that had previously been categorized as “might be” placing individuals at increased risk – but now are listed as high risk – include type 1 diabetes (in addition to type 2), moderate-to-severe asthma, liver disease, dementia or other neurologic conditions, stroke/cerebrovascular disease, HIV infection, cystic fibrosis, and overweight (in addition to obesity).
Substance use disorders, which hadn’t been previously listed, are now also considered high risk.
The new list groups together certain categories, such as chronic lung diseases (chronic obstructive pulmonary disease, asthma, cystic fibrosis, etc) and heart conditions (heart failure, coronary artery disease, hypertension, etc).
Both diabetes types are now grouped under “diabetes.”
The added medical conditions were posted on the CDC website’s COVID-19 page on March 29.
Type 1 diabetes and other conditions now priority for vaccination
The CDC refers to the medical conditions list as phase 1c in regard to COVID-19 vaccine prioritization, which means that anyone with any of these conditions can now be prioritized for vaccination, following those in groups 1a (frontline essential workers and those in long-term care facilities) and 1b (people aged 65-74 years; other essential workers; and people aged 16-64 years with underlying conditions that increase the risk of serious, life-threatening complications from COVID-19).
But in many cases, multiple states have already either fully opened up vaccine eligibility to all adults or have created their own lists of underlying high-risk medical conditions, CDC spokeswoman Kristen Nordlund told this news organization.
No conditions have been removed from the list.
In January, the American Diabetes Association and 18 other organizations sent a letter to the CDC requesting that type 1 diabetes be prioritized along with type 2, based on data from studies showing people with both types to be at high risk for severe COVID-19 illness.
Now, ADA says, “this updated guidance will help to address the fact that in many states, millions of people with type 1 diabetes have not been prioritized equally, slowing their access to critical vaccines.”
While awaiting this latest CDC move, ADA had been urging state governors to prioritize type 1 and type 2 diabetes equally. As of now, 38 states and the District of Columbia had either done so or announced that they would.
A version of this article first appeared on Medscape.com.
Vaccine mismatch: What to do after dose 1 when plans change
Ideally, Americans receiving their Pfizer/BioNTech or Moderna COVID-19 vaccines will get both doses from the same manufacturer, said Gregory Poland, MD, a vaccinologist at the Mayo Clinic in Rochester, Minn.
After all, that’s how they were tested for efficacy and safety, and it was results from those studies that led to emergency use authorization (EUA) being granted by the Food and Drug Administration.
But states and countries have struggled to keep up with the demand for vaccine, and more flexible vaccination schedules could help.
So researchers are exploring whether it is safe and effective to get the first and second doses from different manufacturers. And they are even wondering whether mixing doses from different manufacturers could increase effectiveness, particularly in light of emerging variants.
It’s called the “interchangeability issue,” said Dr. Poland, who has gotten a steady stream of questions about it.
For example, a patient recently asked about options for his father, who had gotten his first dose of the AstraZeneca vaccine in Ecuador, but had since moved to the United States, where that product has not been approved for use.
Dr. Poland said in an interview that he prefaces each answer with: “I’ve got no science for what I’m about to tell you.”
In this particular case, he recommended that the man’s father talk with his doctor about his level of COVID-19 risk and consider whether he should gamble on the AstraZeneca vaccine getting approved in the United States soon, or whether he should ask for a second dose from one of the three vaccines currently approved.
On March 22, 2021, AstraZeneca released positive results from its phase 3 trial, which will likely speed its path toward use in the United States.
Although clinical trials have started to test combinations and boosters, there’s currently no definitive evidence from human trials on mixing COVID vaccines, Dr. Poland pointed out.
But a study of a mixed-vaccine regimen is currently underway in the United Kingdom.
Participants in that 13-month trial will be given the Oxford/AstraZeneca and Pfizer/BioNTech vaccines in different combinations and at different intervals. The first results from that trial are expected this summer.
And interim results from a trial combining Russia’s Sputnik V and the AstraZeneca vaccines are expected in 2 months, according to a Reuters report.
Mix only in ‘exceptional situations’
The Centers for Disease Control and Prevention has been hesitant to open the door to mixing Pfizer and Moderna vaccinations, noting that the two “are not interchangeable.” But CDC guidance has changed slightly. Now, instead of saying the two vaccines should not be mixed, CDC guidance says they can be mixed in “exceptional situations,” and that the second dose can be administered up to 6 weeks after the first dose.
It is reasonable to assume that mixing COVID-19 vaccines that use the same platform – such as the mRNA platform used by both the Pfizer and Moderna vaccines – will be acceptable, Dr. Poland said, although human trials have not proven that.
However, it is unclear whether vaccines that use different platforms can be mixed. Can the first dose of an mRNA vaccine be followed by an adenovirus-based vaccine, like the Johnson & Johnson product or Novavax, if that vaccine is granted an EUA?
Ross Kedl, PhD, a vaccine researcher and professor of immunology at the University of Colorado at Denver, Aurora, said matching vaccine platforms might not be the preferred vaccination strategy.
He disagreed that there’s a lack of science surrounding the issue, and said all signs point to mixing as not only a good option, but probably a better one.
Researcher says science backs mixing
A mix of two different vaccine platforms likely enhances immunity, Dr. Kedl said. The heterologous prime-boost strategy has been used in animal studies for decades, “and it is well known that this promotes a much better immune response than when immunizing with the same vaccine twice.
“If you think about it in a Venn diagram sort of way, it makes sense,” he said in an interview. “Each vaccine has a number of components in it that influence immunity in various ways, but between the two of them, they only have one component that is similar. In the case of the coronavirus vaccines, the one thing both have in common is the spike protein from SARS-CoV-2. In essence, this gives you two shots at generating immunity against the one thing in each vaccine you care most about, but only one shot for the other vaccine components in each platform, resulting in an amplified response against the common target.”
In fact, the heterologous prime-boost vaccination strategy has proven to be effective in humans in early studies.
For example, an Ebola regimen that consisted of an adenovirus vector, similar to the AstraZeneca COVID vaccine, and a modified vaccinia virus vector showed promise in a phase 1 study. And an HIV regimen that consisted of the combination of a DNA vaccine, similar to the Pfizer and Moderna mRNA vaccines, and another viral vector showed encouraging results in a proof-of-concept study.
In both these cases, the heterologous prime-boost strategy was far better than single-vaccine prime-boost regimens, Dr. Kedl pointed out. And neither study reported any safety issues with the combinations.
For now, it’s best to stick with the same manufacturer for both shots, as the CDC guidance suggests, he said, agreeing with Dr. Poland.
But “I would be very surprised if we didn’t move to a mixing of vaccine platforms for the population,” Dr. Kedl said.
A version of this article first appeared on Medscape.com.
Ideally, Americans receiving their Pfizer/BioNTech or Moderna COVID-19 vaccines will get both doses from the same manufacturer, said Gregory Poland, MD, a vaccinologist at the Mayo Clinic in Rochester, Minn.
After all, that’s how they were tested for efficacy and safety, and it was results from those studies that led to emergency use authorization (EUA) being granted by the Food and Drug Administration.
But states and countries have struggled to keep up with the demand for vaccine, and more flexible vaccination schedules could help.
So researchers are exploring whether it is safe and effective to get the first and second doses from different manufacturers. And they are even wondering whether mixing doses from different manufacturers could increase effectiveness, particularly in light of emerging variants.
It’s called the “interchangeability issue,” said Dr. Poland, who has gotten a steady stream of questions about it.
For example, a patient recently asked about options for his father, who had gotten his first dose of the AstraZeneca vaccine in Ecuador, but had since moved to the United States, where that product has not been approved for use.
Dr. Poland said in an interview that he prefaces each answer with: “I’ve got no science for what I’m about to tell you.”
In this particular case, he recommended that the man’s father talk with his doctor about his level of COVID-19 risk and consider whether he should gamble on the AstraZeneca vaccine getting approved in the United States soon, or whether he should ask for a second dose from one of the three vaccines currently approved.
On March 22, 2021, AstraZeneca released positive results from its phase 3 trial, which will likely speed its path toward use in the United States.
Although clinical trials have started to test combinations and boosters, there’s currently no definitive evidence from human trials on mixing COVID vaccines, Dr. Poland pointed out.
But a study of a mixed-vaccine regimen is currently underway in the United Kingdom.
Participants in that 13-month trial will be given the Oxford/AstraZeneca and Pfizer/BioNTech vaccines in different combinations and at different intervals. The first results from that trial are expected this summer.
And interim results from a trial combining Russia’s Sputnik V and the AstraZeneca vaccines are expected in 2 months, according to a Reuters report.
Mix only in ‘exceptional situations’
The Centers for Disease Control and Prevention has been hesitant to open the door to mixing Pfizer and Moderna vaccinations, noting that the two “are not interchangeable.” But CDC guidance has changed slightly. Now, instead of saying the two vaccines should not be mixed, CDC guidance says they can be mixed in “exceptional situations,” and that the second dose can be administered up to 6 weeks after the first dose.
It is reasonable to assume that mixing COVID-19 vaccines that use the same platform – such as the mRNA platform used by both the Pfizer and Moderna vaccines – will be acceptable, Dr. Poland said, although human trials have not proven that.
However, it is unclear whether vaccines that use different platforms can be mixed. Can the first dose of an mRNA vaccine be followed by an adenovirus-based vaccine, like the Johnson & Johnson product or Novavax, if that vaccine is granted an EUA?
Ross Kedl, PhD, a vaccine researcher and professor of immunology at the University of Colorado at Denver, Aurora, said matching vaccine platforms might not be the preferred vaccination strategy.
He disagreed that there’s a lack of science surrounding the issue, and said all signs point to mixing as not only a good option, but probably a better one.
Researcher says science backs mixing
A mix of two different vaccine platforms likely enhances immunity, Dr. Kedl said. The heterologous prime-boost strategy has been used in animal studies for decades, “and it is well known that this promotes a much better immune response than when immunizing with the same vaccine twice.
“If you think about it in a Venn diagram sort of way, it makes sense,” he said in an interview. “Each vaccine has a number of components in it that influence immunity in various ways, but between the two of them, they only have one component that is similar. In the case of the coronavirus vaccines, the one thing both have in common is the spike protein from SARS-CoV-2. In essence, this gives you two shots at generating immunity against the one thing in each vaccine you care most about, but only one shot for the other vaccine components in each platform, resulting in an amplified response against the common target.”
In fact, the heterologous prime-boost vaccination strategy has proven to be effective in humans in early studies.
For example, an Ebola regimen that consisted of an adenovirus vector, similar to the AstraZeneca COVID vaccine, and a modified vaccinia virus vector showed promise in a phase 1 study. And an HIV regimen that consisted of the combination of a DNA vaccine, similar to the Pfizer and Moderna mRNA vaccines, and another viral vector showed encouraging results in a proof-of-concept study.
In both these cases, the heterologous prime-boost strategy was far better than single-vaccine prime-boost regimens, Dr. Kedl pointed out. And neither study reported any safety issues with the combinations.
For now, it’s best to stick with the same manufacturer for both shots, as the CDC guidance suggests, he said, agreeing with Dr. Poland.
But “I would be very surprised if we didn’t move to a mixing of vaccine platforms for the population,” Dr. Kedl said.
A version of this article first appeared on Medscape.com.
Ideally, Americans receiving their Pfizer/BioNTech or Moderna COVID-19 vaccines will get both doses from the same manufacturer, said Gregory Poland, MD, a vaccinologist at the Mayo Clinic in Rochester, Minn.
After all, that’s how they were tested for efficacy and safety, and it was results from those studies that led to emergency use authorization (EUA) being granted by the Food and Drug Administration.
But states and countries have struggled to keep up with the demand for vaccine, and more flexible vaccination schedules could help.
So researchers are exploring whether it is safe and effective to get the first and second doses from different manufacturers. And they are even wondering whether mixing doses from different manufacturers could increase effectiveness, particularly in light of emerging variants.
It’s called the “interchangeability issue,” said Dr. Poland, who has gotten a steady stream of questions about it.
For example, a patient recently asked about options for his father, who had gotten his first dose of the AstraZeneca vaccine in Ecuador, but had since moved to the United States, where that product has not been approved for use.
Dr. Poland said in an interview that he prefaces each answer with: “I’ve got no science for what I’m about to tell you.”
In this particular case, he recommended that the man’s father talk with his doctor about his level of COVID-19 risk and consider whether he should gamble on the AstraZeneca vaccine getting approved in the United States soon, or whether he should ask for a second dose from one of the three vaccines currently approved.
On March 22, 2021, AstraZeneca released positive results from its phase 3 trial, which will likely speed its path toward use in the United States.
Although clinical trials have started to test combinations and boosters, there’s currently no definitive evidence from human trials on mixing COVID vaccines, Dr. Poland pointed out.
But a study of a mixed-vaccine regimen is currently underway in the United Kingdom.
Participants in that 13-month trial will be given the Oxford/AstraZeneca and Pfizer/BioNTech vaccines in different combinations and at different intervals. The first results from that trial are expected this summer.
And interim results from a trial combining Russia’s Sputnik V and the AstraZeneca vaccines are expected in 2 months, according to a Reuters report.
Mix only in ‘exceptional situations’
The Centers for Disease Control and Prevention has been hesitant to open the door to mixing Pfizer and Moderna vaccinations, noting that the two “are not interchangeable.” But CDC guidance has changed slightly. Now, instead of saying the two vaccines should not be mixed, CDC guidance says they can be mixed in “exceptional situations,” and that the second dose can be administered up to 6 weeks after the first dose.
It is reasonable to assume that mixing COVID-19 vaccines that use the same platform – such as the mRNA platform used by both the Pfizer and Moderna vaccines – will be acceptable, Dr. Poland said, although human trials have not proven that.
However, it is unclear whether vaccines that use different platforms can be mixed. Can the first dose of an mRNA vaccine be followed by an adenovirus-based vaccine, like the Johnson & Johnson product or Novavax, if that vaccine is granted an EUA?
Ross Kedl, PhD, a vaccine researcher and professor of immunology at the University of Colorado at Denver, Aurora, said matching vaccine platforms might not be the preferred vaccination strategy.
He disagreed that there’s a lack of science surrounding the issue, and said all signs point to mixing as not only a good option, but probably a better one.
Researcher says science backs mixing
A mix of two different vaccine platforms likely enhances immunity, Dr. Kedl said. The heterologous prime-boost strategy has been used in animal studies for decades, “and it is well known that this promotes a much better immune response than when immunizing with the same vaccine twice.
“If you think about it in a Venn diagram sort of way, it makes sense,” he said in an interview. “Each vaccine has a number of components in it that influence immunity in various ways, but between the two of them, they only have one component that is similar. In the case of the coronavirus vaccines, the one thing both have in common is the spike protein from SARS-CoV-2. In essence, this gives you two shots at generating immunity against the one thing in each vaccine you care most about, but only one shot for the other vaccine components in each platform, resulting in an amplified response against the common target.”
In fact, the heterologous prime-boost vaccination strategy has proven to be effective in humans in early studies.
For example, an Ebola regimen that consisted of an adenovirus vector, similar to the AstraZeneca COVID vaccine, and a modified vaccinia virus vector showed promise in a phase 1 study. And an HIV regimen that consisted of the combination of a DNA vaccine, similar to the Pfizer and Moderna mRNA vaccines, and another viral vector showed encouraging results in a proof-of-concept study.
In both these cases, the heterologous prime-boost strategy was far better than single-vaccine prime-boost regimens, Dr. Kedl pointed out. And neither study reported any safety issues with the combinations.
For now, it’s best to stick with the same manufacturer for both shots, as the CDC guidance suggests, he said, agreeing with Dr. Poland.
But “I would be very surprised if we didn’t move to a mixing of vaccine platforms for the population,” Dr. Kedl said.
A version of this article first appeared on Medscape.com.
Rhythm and blues: Using heart rate to diagnose depression
Depression might be a disorder of the brain, but its harms aren’t confined to the cranium. Prolonged depression has been linked with a slew of health problems, from impaired immune function to gastrointestinal dysfunction. It’s also been linked with cardiovascular disease (CVD), even increasing the risk for heart attack and a disrupted heart rate. Now, researchers are exploring whether heart function could be a valuable biomarker in informing depression diagnosis and treatment.
Major depressive disorder has proved difficult to diagnose and treat, and biomarkers that indicate a depressive episode or suggest specific interventions would be an attractive solution to its clinically nebulous nature.
Currently, diagnosing depression relies on the patients effectively communicating their symptoms. If the patient does receive a diagnosis, treating it remains a matter of trial and error. It takes weeks to know whether a treatment is working, and in only one-third of cases does the condition go into remission after the patient is initially prescribed an antidepressant. Even after successful treatment, it’s challenging to identify who might be at risk for relapse, and when. Research now shows that cardiac biomarkers may be a way improve this picture. Clinicians could use changes in heart rate to both inform depression diagnosis and gauge a patient’s predicted response to treatment.
Given the increased risk for CVD among people with depression and the link between heart rate changes and CVD risk, these biomarkers could have implications for heart health, too. “We need more than just the current toolkit,” said Amit Shah, MD, a cardiologist and assistant professor of epidemiology at Emory University, Atlanta. “Ultimately, what we’re trying to do is develop interventions not only for depression but also for the associated physical health problems related to depression, in particular, cardiovascular disease,” he said. These overlapping interests – and the prospect of clinically considering both conditions in tandem – mean this research is “really worth its weight in gold,” added Dr. Shah.
The data on heart rate biomarkers
Patients with depression are often found to have lower heart rate variability (HRV) and higher heart rates. Scientists don’t know the mechanisms underpinning this relationship but think changes in the autonomic nervous system during depression, as well as stress generally, have a role.
Rébecca Robillard, PhD, is the head scientist of the Clinical Sleep Research Platform at the Royal’s Institute of Mental Health Research, Ottawa, Ont. In a 2019 study published in BMC Psychiatry, Dr. Robillard’s team used electrocardiogram recordings from sleep studies to see whether heart rate abnormalities were associated with depression. Using a profiling algorithm to analyze heart rate and HRV data, the team identified persons with depression with 80% accuracy among 174 people with sleep complaints.
Dr. Robillard said.
In another study, Stephan Claes, MD, PhD, psychiatrist and professor of psychiatry at Katholieke Universiteit Leuven, Belgium, and his group tested the biomarker potential of heart rate and HRV data that were continuously recorded over several days. They too used an algorithm to distinguish 16 people with treatment-resistant depression from 16 without depression. Within the depression group, they used the algorithm to distinguish patients who had received ketamine treatment from those who had not.
The algorithm could differentiate between the depressed and nondepressed groups with 90% accuracy. Those with depression had higher overall heart rates, particularly at night, and lower HRV. Dr. Claes noted that, unlike in other studies, “the most reliable parameter that we had for this prediction was the heart rate, not the HRV.” After treatment, heart rates improved, but HRV remained the same.
Although their study has not yet been peer reviewed and more research is needed, Dr. Claes said that increased heart rate, especially during the night, could eventually serve as a warning sign of depression relapse. “That would allow a quicker referral to care and better care because of earlier intervention,” he said.
Finding a signal amid the noise
But heart rate and HRV aren’t foolproof biomarkers. Some studies have found that antidepressant use lowers HRV and that HRV changes aren’t unique to depression. There’s the added complication that depression often overlaps with other mental disorders.
“I think we’ve been very disappointed about the success of using particular biomarkers for particular disorders, because the majority of mental disorders are very heterogeneous,” said Andrew Kemp, PhD, psychology professor at Swansea University, Swansea, Wales. “A particular biomarker will, at the end of the day, be just one particular aspect of the overall profile that clinicians will have on particular individuals.”
The clinical utility of a heart rate–depression connection may go both ways.
For instance, depression could serve as a warning sign for atrial fibrillation, according to research from Parveen K. Garg, MD, associate professor of clinical medicine at the University of Southern California, Los Angeles. In a study involving more than 6,000 people, Dr. Garg showed that higher scores on depression scales correlated with a higher risk for the occurrence of atrial fibrillation over a follow-up period of about 13 years.
Depression is associated with other heart conditions as well. “A lot of data seem to suggest that just the presence of depression can increase risk for a whole range of cardiovascular problems,” said Dr. Garg. Epidemiologic studies have found associations between depression and the development of coronary heart disease and a modest increased risk for stroke.
“Things going on in your brain also have effects on the rest of your body,” said Dr. Garg. “Just recognizing this link, that maybe mental illness has an effect on other illnesses or diseases that can affect other parts of your body – I think that’s something we can share now.”
A version of this article first appeared on Medscape.com.
Depression might be a disorder of the brain, but its harms aren’t confined to the cranium. Prolonged depression has been linked with a slew of health problems, from impaired immune function to gastrointestinal dysfunction. It’s also been linked with cardiovascular disease (CVD), even increasing the risk for heart attack and a disrupted heart rate. Now, researchers are exploring whether heart function could be a valuable biomarker in informing depression diagnosis and treatment.
Major depressive disorder has proved difficult to diagnose and treat, and biomarkers that indicate a depressive episode or suggest specific interventions would be an attractive solution to its clinically nebulous nature.
Currently, diagnosing depression relies on the patients effectively communicating their symptoms. If the patient does receive a diagnosis, treating it remains a matter of trial and error. It takes weeks to know whether a treatment is working, and in only one-third of cases does the condition go into remission after the patient is initially prescribed an antidepressant. Even after successful treatment, it’s challenging to identify who might be at risk for relapse, and when. Research now shows that cardiac biomarkers may be a way improve this picture. Clinicians could use changes in heart rate to both inform depression diagnosis and gauge a patient’s predicted response to treatment.
Given the increased risk for CVD among people with depression and the link between heart rate changes and CVD risk, these biomarkers could have implications for heart health, too. “We need more than just the current toolkit,” said Amit Shah, MD, a cardiologist and assistant professor of epidemiology at Emory University, Atlanta. “Ultimately, what we’re trying to do is develop interventions not only for depression but also for the associated physical health problems related to depression, in particular, cardiovascular disease,” he said. These overlapping interests – and the prospect of clinically considering both conditions in tandem – mean this research is “really worth its weight in gold,” added Dr. Shah.
The data on heart rate biomarkers
Patients with depression are often found to have lower heart rate variability (HRV) and higher heart rates. Scientists don’t know the mechanisms underpinning this relationship but think changes in the autonomic nervous system during depression, as well as stress generally, have a role.
Rébecca Robillard, PhD, is the head scientist of the Clinical Sleep Research Platform at the Royal’s Institute of Mental Health Research, Ottawa, Ont. In a 2019 study published in BMC Psychiatry, Dr. Robillard’s team used electrocardiogram recordings from sleep studies to see whether heart rate abnormalities were associated with depression. Using a profiling algorithm to analyze heart rate and HRV data, the team identified persons with depression with 80% accuracy among 174 people with sleep complaints.
Dr. Robillard said.
In another study, Stephan Claes, MD, PhD, psychiatrist and professor of psychiatry at Katholieke Universiteit Leuven, Belgium, and his group tested the biomarker potential of heart rate and HRV data that were continuously recorded over several days. They too used an algorithm to distinguish 16 people with treatment-resistant depression from 16 without depression. Within the depression group, they used the algorithm to distinguish patients who had received ketamine treatment from those who had not.
The algorithm could differentiate between the depressed and nondepressed groups with 90% accuracy. Those with depression had higher overall heart rates, particularly at night, and lower HRV. Dr. Claes noted that, unlike in other studies, “the most reliable parameter that we had for this prediction was the heart rate, not the HRV.” After treatment, heart rates improved, but HRV remained the same.
Although their study has not yet been peer reviewed and more research is needed, Dr. Claes said that increased heart rate, especially during the night, could eventually serve as a warning sign of depression relapse. “That would allow a quicker referral to care and better care because of earlier intervention,” he said.
Finding a signal amid the noise
But heart rate and HRV aren’t foolproof biomarkers. Some studies have found that antidepressant use lowers HRV and that HRV changes aren’t unique to depression. There’s the added complication that depression often overlaps with other mental disorders.
“I think we’ve been very disappointed about the success of using particular biomarkers for particular disorders, because the majority of mental disorders are very heterogeneous,” said Andrew Kemp, PhD, psychology professor at Swansea University, Swansea, Wales. “A particular biomarker will, at the end of the day, be just one particular aspect of the overall profile that clinicians will have on particular individuals.”
The clinical utility of a heart rate–depression connection may go both ways.
For instance, depression could serve as a warning sign for atrial fibrillation, according to research from Parveen K. Garg, MD, associate professor of clinical medicine at the University of Southern California, Los Angeles. In a study involving more than 6,000 people, Dr. Garg showed that higher scores on depression scales correlated with a higher risk for the occurrence of atrial fibrillation over a follow-up period of about 13 years.
Depression is associated with other heart conditions as well. “A lot of data seem to suggest that just the presence of depression can increase risk for a whole range of cardiovascular problems,” said Dr. Garg. Epidemiologic studies have found associations between depression and the development of coronary heart disease and a modest increased risk for stroke.
“Things going on in your brain also have effects on the rest of your body,” said Dr. Garg. “Just recognizing this link, that maybe mental illness has an effect on other illnesses or diseases that can affect other parts of your body – I think that’s something we can share now.”
A version of this article first appeared on Medscape.com.
Depression might be a disorder of the brain, but its harms aren’t confined to the cranium. Prolonged depression has been linked with a slew of health problems, from impaired immune function to gastrointestinal dysfunction. It’s also been linked with cardiovascular disease (CVD), even increasing the risk for heart attack and a disrupted heart rate. Now, researchers are exploring whether heart function could be a valuable biomarker in informing depression diagnosis and treatment.
Major depressive disorder has proved difficult to diagnose and treat, and biomarkers that indicate a depressive episode or suggest specific interventions would be an attractive solution to its clinically nebulous nature.
Currently, diagnosing depression relies on the patients effectively communicating their symptoms. If the patient does receive a diagnosis, treating it remains a matter of trial and error. It takes weeks to know whether a treatment is working, and in only one-third of cases does the condition go into remission after the patient is initially prescribed an antidepressant. Even after successful treatment, it’s challenging to identify who might be at risk for relapse, and when. Research now shows that cardiac biomarkers may be a way improve this picture. Clinicians could use changes in heart rate to both inform depression diagnosis and gauge a patient’s predicted response to treatment.
Given the increased risk for CVD among people with depression and the link between heart rate changes and CVD risk, these biomarkers could have implications for heart health, too. “We need more than just the current toolkit,” said Amit Shah, MD, a cardiologist and assistant professor of epidemiology at Emory University, Atlanta. “Ultimately, what we’re trying to do is develop interventions not only for depression but also for the associated physical health problems related to depression, in particular, cardiovascular disease,” he said. These overlapping interests – and the prospect of clinically considering both conditions in tandem – mean this research is “really worth its weight in gold,” added Dr. Shah.
The data on heart rate biomarkers
Patients with depression are often found to have lower heart rate variability (HRV) and higher heart rates. Scientists don’t know the mechanisms underpinning this relationship but think changes in the autonomic nervous system during depression, as well as stress generally, have a role.
Rébecca Robillard, PhD, is the head scientist of the Clinical Sleep Research Platform at the Royal’s Institute of Mental Health Research, Ottawa, Ont. In a 2019 study published in BMC Psychiatry, Dr. Robillard’s team used electrocardiogram recordings from sleep studies to see whether heart rate abnormalities were associated with depression. Using a profiling algorithm to analyze heart rate and HRV data, the team identified persons with depression with 80% accuracy among 174 people with sleep complaints.
Dr. Robillard said.
In another study, Stephan Claes, MD, PhD, psychiatrist and professor of psychiatry at Katholieke Universiteit Leuven, Belgium, and his group tested the biomarker potential of heart rate and HRV data that were continuously recorded over several days. They too used an algorithm to distinguish 16 people with treatment-resistant depression from 16 without depression. Within the depression group, they used the algorithm to distinguish patients who had received ketamine treatment from those who had not.
The algorithm could differentiate between the depressed and nondepressed groups with 90% accuracy. Those with depression had higher overall heart rates, particularly at night, and lower HRV. Dr. Claes noted that, unlike in other studies, “the most reliable parameter that we had for this prediction was the heart rate, not the HRV.” After treatment, heart rates improved, but HRV remained the same.
Although their study has not yet been peer reviewed and more research is needed, Dr. Claes said that increased heart rate, especially during the night, could eventually serve as a warning sign of depression relapse. “That would allow a quicker referral to care and better care because of earlier intervention,” he said.
Finding a signal amid the noise
But heart rate and HRV aren’t foolproof biomarkers. Some studies have found that antidepressant use lowers HRV and that HRV changes aren’t unique to depression. There’s the added complication that depression often overlaps with other mental disorders.
“I think we’ve been very disappointed about the success of using particular biomarkers for particular disorders, because the majority of mental disorders are very heterogeneous,” said Andrew Kemp, PhD, psychology professor at Swansea University, Swansea, Wales. “A particular biomarker will, at the end of the day, be just one particular aspect of the overall profile that clinicians will have on particular individuals.”
The clinical utility of a heart rate–depression connection may go both ways.
For instance, depression could serve as a warning sign for atrial fibrillation, according to research from Parveen K. Garg, MD, associate professor of clinical medicine at the University of Southern California, Los Angeles. In a study involving more than 6,000 people, Dr. Garg showed that higher scores on depression scales correlated with a higher risk for the occurrence of atrial fibrillation over a follow-up period of about 13 years.
Depression is associated with other heart conditions as well. “A lot of data seem to suggest that just the presence of depression can increase risk for a whole range of cardiovascular problems,” said Dr. Garg. Epidemiologic studies have found associations between depression and the development of coronary heart disease and a modest increased risk for stroke.
“Things going on in your brain also have effects on the rest of your body,” said Dr. Garg. “Just recognizing this link, that maybe mental illness has an effect on other illnesses or diseases that can affect other parts of your body – I think that’s something we can share now.”
A version of this article first appeared on Medscape.com.
National Psoriasis Foundation recommends some stop methotrexate for 2 weeks after J&J vaccine
The , Joel M. Gelfand, MD, said at Innovations in Dermatology: Virtual Spring Conference 2021.
The new guidance states: “Patients 60 or older who have at least one comorbidity associated with an increased risk for poor COVID-19 outcomes, and who are taking methotrexate with well-controlled psoriatic disease, may, in consultation with their prescriber, consider holding it for 2 weeks after receiving the Ad26.COV2.S [Johnson & Johnson] vaccine in order to potentially improve vaccine response.”
The key word here is “potentially.” There is no hard evidence that a 2-week hold on methotrexate after receiving the killed adenovirus vaccine will actually provide a clinically meaningful benefit. But it’s a hypothetical possibility. The rationale stems from a small randomized trial conducted in South Korea several years ago in which patients with rheumatoid arthritis were assigned to hold or continue their methotrexate for the first 2 weeks after receiving an inactivated-virus influenza vaccine. The antibody response to the vaccine was better in those who temporarily halted their methotrexate, explained Dr. Gelfand, cochair of the NPF COVID-19 Task Force and professor of dermatology and of epidemiology at the University of Pennsylvania, Philadelphia.
“If you have a patient on methotrexate who’s 60 or older and whose psoriasis is completely controlled and quiescent and the patient is concerned about how well the vaccine is going to work, this is a reasonable thing to consider in someone who’s at higher risk for poor outcomes if they get infected,” he said.
If the informed patient wants to continue on methotrexate without interruption, that’s fine, too, in light of the lack of compelling evidence on this issue, the dermatologist added at the conference, sponsored by MedscapeLIVE! and the producers of the Hawaii Dermatology Seminar and Caribbean Dermatology Symposium.
The NPF task force does not extend the recommendation to consider holding methotrexate in recipients of the mRNA-based Moderna and Pfizer vaccines because of their very different mechanisms of action. Nor is it recommended to hold biologic agents after receiving any of the available COVID-19 vaccines. Studies have shown no altered immunologic response to influenza or pneumococcal vaccines in patients who continued on tumor necrosis factor inhibitors or interleukin-17 inhibitors. The interleukin-23 inhibitors haven’t been studied in this regard.
The task force recommends that most psoriasis patients should continue on treatment throughout the pandemic, and newly diagnosed patients should commence appropriate therapy as if there was no pandemic.
“We’ve learned that many patients who stopped their treatment for psoriatic disease early in the pandemic came to regret that decision because their psoriasis flared and got worse and required reinstitution of therapy,” Dr. Gelfand said. “The current data is largely reassuring that if there is an effect of our therapies on the risk of COVID, it must be rather small and therefore unlikely to be clinically meaningful for our patients.”
Dr. Gelfand reported serving as a consultant to and recipient of institutional research grants from Pfizer and numerous other pharmaceutical companies.
MedscapeLIVE and this news organization are owned by the same parent company.
The , Joel M. Gelfand, MD, said at Innovations in Dermatology: Virtual Spring Conference 2021.
The new guidance states: “Patients 60 or older who have at least one comorbidity associated with an increased risk for poor COVID-19 outcomes, and who are taking methotrexate with well-controlled psoriatic disease, may, in consultation with their prescriber, consider holding it for 2 weeks after receiving the Ad26.COV2.S [Johnson & Johnson] vaccine in order to potentially improve vaccine response.”
The key word here is “potentially.” There is no hard evidence that a 2-week hold on methotrexate after receiving the killed adenovirus vaccine will actually provide a clinically meaningful benefit. But it’s a hypothetical possibility. The rationale stems from a small randomized trial conducted in South Korea several years ago in which patients with rheumatoid arthritis were assigned to hold or continue their methotrexate for the first 2 weeks after receiving an inactivated-virus influenza vaccine. The antibody response to the vaccine was better in those who temporarily halted their methotrexate, explained Dr. Gelfand, cochair of the NPF COVID-19 Task Force and professor of dermatology and of epidemiology at the University of Pennsylvania, Philadelphia.
“If you have a patient on methotrexate who’s 60 or older and whose psoriasis is completely controlled and quiescent and the patient is concerned about how well the vaccine is going to work, this is a reasonable thing to consider in someone who’s at higher risk for poor outcomes if they get infected,” he said.
If the informed patient wants to continue on methotrexate without interruption, that’s fine, too, in light of the lack of compelling evidence on this issue, the dermatologist added at the conference, sponsored by MedscapeLIVE! and the producers of the Hawaii Dermatology Seminar and Caribbean Dermatology Symposium.
The NPF task force does not extend the recommendation to consider holding methotrexate in recipients of the mRNA-based Moderna and Pfizer vaccines because of their very different mechanisms of action. Nor is it recommended to hold biologic agents after receiving any of the available COVID-19 vaccines. Studies have shown no altered immunologic response to influenza or pneumococcal vaccines in patients who continued on tumor necrosis factor inhibitors or interleukin-17 inhibitors. The interleukin-23 inhibitors haven’t been studied in this regard.
The task force recommends that most psoriasis patients should continue on treatment throughout the pandemic, and newly diagnosed patients should commence appropriate therapy as if there was no pandemic.
“We’ve learned that many patients who stopped their treatment for psoriatic disease early in the pandemic came to regret that decision because their psoriasis flared and got worse and required reinstitution of therapy,” Dr. Gelfand said. “The current data is largely reassuring that if there is an effect of our therapies on the risk of COVID, it must be rather small and therefore unlikely to be clinically meaningful for our patients.”
Dr. Gelfand reported serving as a consultant to and recipient of institutional research grants from Pfizer and numerous other pharmaceutical companies.
MedscapeLIVE and this news organization are owned by the same parent company.
The , Joel M. Gelfand, MD, said at Innovations in Dermatology: Virtual Spring Conference 2021.
The new guidance states: “Patients 60 or older who have at least one comorbidity associated with an increased risk for poor COVID-19 outcomes, and who are taking methotrexate with well-controlled psoriatic disease, may, in consultation with their prescriber, consider holding it for 2 weeks after receiving the Ad26.COV2.S [Johnson & Johnson] vaccine in order to potentially improve vaccine response.”
The key word here is “potentially.” There is no hard evidence that a 2-week hold on methotrexate after receiving the killed adenovirus vaccine will actually provide a clinically meaningful benefit. But it’s a hypothetical possibility. The rationale stems from a small randomized trial conducted in South Korea several years ago in which patients with rheumatoid arthritis were assigned to hold or continue their methotrexate for the first 2 weeks after receiving an inactivated-virus influenza vaccine. The antibody response to the vaccine was better in those who temporarily halted their methotrexate, explained Dr. Gelfand, cochair of the NPF COVID-19 Task Force and professor of dermatology and of epidemiology at the University of Pennsylvania, Philadelphia.
“If you have a patient on methotrexate who’s 60 or older and whose psoriasis is completely controlled and quiescent and the patient is concerned about how well the vaccine is going to work, this is a reasonable thing to consider in someone who’s at higher risk for poor outcomes if they get infected,” he said.
If the informed patient wants to continue on methotrexate without interruption, that’s fine, too, in light of the lack of compelling evidence on this issue, the dermatologist added at the conference, sponsored by MedscapeLIVE! and the producers of the Hawaii Dermatology Seminar and Caribbean Dermatology Symposium.
The NPF task force does not extend the recommendation to consider holding methotrexate in recipients of the mRNA-based Moderna and Pfizer vaccines because of their very different mechanisms of action. Nor is it recommended to hold biologic agents after receiving any of the available COVID-19 vaccines. Studies have shown no altered immunologic response to influenza or pneumococcal vaccines in patients who continued on tumor necrosis factor inhibitors or interleukin-17 inhibitors. The interleukin-23 inhibitors haven’t been studied in this regard.
The task force recommends that most psoriasis patients should continue on treatment throughout the pandemic, and newly diagnosed patients should commence appropriate therapy as if there was no pandemic.
“We’ve learned that many patients who stopped their treatment for psoriatic disease early in the pandemic came to regret that decision because their psoriasis flared and got worse and required reinstitution of therapy,” Dr. Gelfand said. “The current data is largely reassuring that if there is an effect of our therapies on the risk of COVID, it must be rather small and therefore unlikely to be clinically meaningful for our patients.”
Dr. Gelfand reported serving as a consultant to and recipient of institutional research grants from Pfizer and numerous other pharmaceutical companies.
MedscapeLIVE and this news organization are owned by the same parent company.
FROM INNOVATIONS IN DERMATOLOGY
Child abuse tied to persistent inflammation in later life
People who suffer abuse as children continue to have higher levels of inflammatory biomarkers as adults, likely placing them at increased risk for chronic health problems, new research shows.
In a study assessing trajectories of inflammation over a 3-year period in healthy adults, those who reported higher rates of physical, sexual, or emotional abuse had higher levels of bioinflammatory blood markers versus those who reported lower rates of abuse. These individuals also had significantly higher rates of loneliness and depression.
“These adverse experiences that people can have in childhood and adolescence really can continue to influence our health at a biological level well into adulthood,” investigator Megan E. Renna, PhD, postdoctoral fellow at Ohio State University, Columbus, said in an interview.
“There may be a somewhat invisible biological effect of that abuse later on in life, in addition to all of the psychological distress that could go along with those experiences,” she added.
The findings were presented at the virtual Anxiety and Depression Association of America Conference 2021.
Need for intervention
Ages ranged from 47 to 67 years (mean age, 57 years), 81% were women, all were relatively healthy, and there were low rates of medical comorbidities.
Participant data on physical, emotional, and sexual abuse prior to age 18 were ascertained using the Childhood Experiences Questionnaire. Blood samples were assayed for cytokines interleukin-6, IL-8, IL-1 beta and tumor necrosis factor–alpha at each visit.
After controlling for age, sex, body mass index, and medical comorbidities, results showed markers of inflammation increased at a greater rate over time in participants with higher rates of physical (P = .05) and sexual abuse (P = .02), compared with those with no history of childhood abuse.
“So, inflammation was increasing at a faster rate across those three visits for people with versus without an abuse history. And this was abuse experienced before age 18, but the mean age of our participants was about 57,” said Dr. Renna.
“It is likely that emotional abuse plays a role in inflammation but we did not have a big enough sample to show significance,” she added.
Participants who had reported childhood abuse also demonstrated significantly higher rates of loneliness and depression across all visits than those without a history of abuse.
“One of the things this work really highlights is the need for intervention for these children and adolescents who are experiencing abuse. This may have a helpful impact on their psychological health as they age, as well as their physical health,” Dr. Renna said.
‘Considerable interest’
In a comment, ADAA President Luana Marques, PhD, Harvard Medical School, Boston, said research is “consistently indicating that childhood adversity puts individuals at risk for a host of problems, including inflammatory concerns, which are precursors for other physical illnesses.”
Such results “demonstrate the importance of early identification and intervention of possible traumatic experiences for youth, and how early intervention at the parent level might also be helpful,” said Dr. Marques, who was not involved with the research.
Also commenting on the study, Charles B. Nemeroff, MD, PhD, professor and chair in the department of psychiatry and behavioral science at the University of Texas at Austin, and president-elect of the ADAA, said in an interview that the findings are pertinent for the field.
“The investigators demonstrated that a history of childhood physical or sexual abuse was associated with a greater inflammatory response, and this is of considerable interest because this increased inflammatory response very likely contributes to the well-documented increased prevalence of serious medical disorders such as heart disease, diabetes, and cancer in victims of child abuse and neglect,” said Dr. Nemeroff, who was not associated with the research.
Dr. Renna, Dr. Marques, and Dr. Nemeroff disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
People who suffer abuse as children continue to have higher levels of inflammatory biomarkers as adults, likely placing them at increased risk for chronic health problems, new research shows.
In a study assessing trajectories of inflammation over a 3-year period in healthy adults, those who reported higher rates of physical, sexual, or emotional abuse had higher levels of bioinflammatory blood markers versus those who reported lower rates of abuse. These individuals also had significantly higher rates of loneliness and depression.
“These adverse experiences that people can have in childhood and adolescence really can continue to influence our health at a biological level well into adulthood,” investigator Megan E. Renna, PhD, postdoctoral fellow at Ohio State University, Columbus, said in an interview.
“There may be a somewhat invisible biological effect of that abuse later on in life, in addition to all of the psychological distress that could go along with those experiences,” she added.
The findings were presented at the virtual Anxiety and Depression Association of America Conference 2021.
Need for intervention
Ages ranged from 47 to 67 years (mean age, 57 years), 81% were women, all were relatively healthy, and there were low rates of medical comorbidities.
Participant data on physical, emotional, and sexual abuse prior to age 18 were ascertained using the Childhood Experiences Questionnaire. Blood samples were assayed for cytokines interleukin-6, IL-8, IL-1 beta and tumor necrosis factor–alpha at each visit.
After controlling for age, sex, body mass index, and medical comorbidities, results showed markers of inflammation increased at a greater rate over time in participants with higher rates of physical (P = .05) and sexual abuse (P = .02), compared with those with no history of childhood abuse.
“So, inflammation was increasing at a faster rate across those three visits for people with versus without an abuse history. And this was abuse experienced before age 18, but the mean age of our participants was about 57,” said Dr. Renna.
“It is likely that emotional abuse plays a role in inflammation but we did not have a big enough sample to show significance,” she added.
Participants who had reported childhood abuse also demonstrated significantly higher rates of loneliness and depression across all visits than those without a history of abuse.
“One of the things this work really highlights is the need for intervention for these children and adolescents who are experiencing abuse. This may have a helpful impact on their psychological health as they age, as well as their physical health,” Dr. Renna said.
‘Considerable interest’
In a comment, ADAA President Luana Marques, PhD, Harvard Medical School, Boston, said research is “consistently indicating that childhood adversity puts individuals at risk for a host of problems, including inflammatory concerns, which are precursors for other physical illnesses.”
Such results “demonstrate the importance of early identification and intervention of possible traumatic experiences for youth, and how early intervention at the parent level might also be helpful,” said Dr. Marques, who was not involved with the research.
Also commenting on the study, Charles B. Nemeroff, MD, PhD, professor and chair in the department of psychiatry and behavioral science at the University of Texas at Austin, and president-elect of the ADAA, said in an interview that the findings are pertinent for the field.
“The investigators demonstrated that a history of childhood physical or sexual abuse was associated with a greater inflammatory response, and this is of considerable interest because this increased inflammatory response very likely contributes to the well-documented increased prevalence of serious medical disorders such as heart disease, diabetes, and cancer in victims of child abuse and neglect,” said Dr. Nemeroff, who was not associated with the research.
Dr. Renna, Dr. Marques, and Dr. Nemeroff disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
People who suffer abuse as children continue to have higher levels of inflammatory biomarkers as adults, likely placing them at increased risk for chronic health problems, new research shows.
In a study assessing trajectories of inflammation over a 3-year period in healthy adults, those who reported higher rates of physical, sexual, or emotional abuse had higher levels of bioinflammatory blood markers versus those who reported lower rates of abuse. These individuals also had significantly higher rates of loneliness and depression.
“These adverse experiences that people can have in childhood and adolescence really can continue to influence our health at a biological level well into adulthood,” investigator Megan E. Renna, PhD, postdoctoral fellow at Ohio State University, Columbus, said in an interview.
“There may be a somewhat invisible biological effect of that abuse later on in life, in addition to all of the psychological distress that could go along with those experiences,” she added.
The findings were presented at the virtual Anxiety and Depression Association of America Conference 2021.
Need for intervention
Ages ranged from 47 to 67 years (mean age, 57 years), 81% were women, all were relatively healthy, and there were low rates of medical comorbidities.
Participant data on physical, emotional, and sexual abuse prior to age 18 were ascertained using the Childhood Experiences Questionnaire. Blood samples were assayed for cytokines interleukin-6, IL-8, IL-1 beta and tumor necrosis factor–alpha at each visit.
After controlling for age, sex, body mass index, and medical comorbidities, results showed markers of inflammation increased at a greater rate over time in participants with higher rates of physical (P = .05) and sexual abuse (P = .02), compared with those with no history of childhood abuse.
“So, inflammation was increasing at a faster rate across those three visits for people with versus without an abuse history. And this was abuse experienced before age 18, but the mean age of our participants was about 57,” said Dr. Renna.
“It is likely that emotional abuse plays a role in inflammation but we did not have a big enough sample to show significance,” she added.
Participants who had reported childhood abuse also demonstrated significantly higher rates of loneliness and depression across all visits than those without a history of abuse.
“One of the things this work really highlights is the need for intervention for these children and adolescents who are experiencing abuse. This may have a helpful impact on their psychological health as they age, as well as their physical health,” Dr. Renna said.
‘Considerable interest’
In a comment, ADAA President Luana Marques, PhD, Harvard Medical School, Boston, said research is “consistently indicating that childhood adversity puts individuals at risk for a host of problems, including inflammatory concerns, which are precursors for other physical illnesses.”
Such results “demonstrate the importance of early identification and intervention of possible traumatic experiences for youth, and how early intervention at the parent level might also be helpful,” said Dr. Marques, who was not involved with the research.
Also commenting on the study, Charles B. Nemeroff, MD, PhD, professor and chair in the department of psychiatry and behavioral science at the University of Texas at Austin, and president-elect of the ADAA, said in an interview that the findings are pertinent for the field.
“The investigators demonstrated that a history of childhood physical or sexual abuse was associated with a greater inflammatory response, and this is of considerable interest because this increased inflammatory response very likely contributes to the well-documented increased prevalence of serious medical disorders such as heart disease, diabetes, and cancer in victims of child abuse and neglect,” said Dr. Nemeroff, who was not associated with the research.
Dr. Renna, Dr. Marques, and Dr. Nemeroff disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
COVID-19 ‘long-haul’ symptoms overlap with ME/CFS
People experiencing long-term symptoms following acute COVID-19 infection are increasingly meeting criteria for myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS), a phenomenon that highlights the need for unified research and clinical approaches, speakers said at a press briefing March 25 held by the advocacy group MEAction.
“Post-COVID lingering illness was predictable. Similar lingering fatigue syndromes have been reported in the scientific literature for nearly 100 years, following a variety of well-documented infections with viruses, bacteria, fungi, and even protozoa,” said Anthony Komaroff, MD, professor of medicine at Harvard Medical School, Boston.
Core criteria for ME/CFS established by the Institute of Medicine in 2015 include substantial decrement in functioning for at least 6 months, postexertional malaise (PEM), or a worsening of symptoms following even minor exertion (often described as “crashes”), unrefreshing sleep, and cognitive impairment and/or orthostatic intolerance.
Patients with ME/CFS also commonly experience painful headaches, muscle or joint aches, and allergies/other sensitivities. Although many patients can trace their symptoms to an initiating infection, “the cause is often unclear because the diagnosis is often delayed for months or years after symptom onset,” said Lucinda Bateman, MD, founder of the Bateman Horne Center, Salt Lake City, who leads a clinician coalition that aims to improve ME/CFS management.
In an international survey of 3762 COVID-19 “long-haulers” published in a preprint in December of 2020, the most frequent symptoms reported at least 6 months after illness onset were fatigue in 78%, PEM in 72%, and cognitive dysfunction (“brain fog”) in 55%. At the time of the survey, 45% reported requiring reduced work schedules because of their illness, and 22% reported being unable to work at all.
Dr. Bateman said those findings align with her experience so far with 12 COVID-19 “long haulers” who self-referred to her ME/CFS and fibromyalgia specialty clinic. Nine of the 12 met criteria for postural orthostatic tachycardia syndrome (POTS) based on the 10-minute NASA Lean Test, she said, and half also met the 2016 American College of Rheumatology criteria for fibromyalgia.
“Some were severely impaired. We suspect a small fiber polyneuropathy in about half, and mast cell activation syndrome in more than half. We look forward to doing more testing,” Dr. Bateman said.
To be sure, Dr. Komaroff noted, there are some differences. “Long COVID” patients will often experience breathlessness and ongoing anosmia (loss of taste and smell), which aren’t typical of ME/CFS.
But, he said, “many of the symptoms are quite similar ... My guess is that ME/CFS is an illness with a final common pathway that can be triggered by different things,” said Dr. Komaroff, a senior physician at Brigham and Women’s Hospital in Boston, and editor-in-chief of the Harvard Health Letter.
Based on previous data about CFS suggesting a 10% rate of symptoms persisting at least a year following a variety of infectious agents and the predicted 200 million COVID-19 cases globally by the end of 2021, Dr. Komaroff estimated that about 20 million cases of “long COVID” would be expected in the next year.
‘A huge investment’
On the research side, the National Institutes of Health recently appropriated $1.15 billion dollars over the next 4 years to investigate “the heterogeneity in the recovery process after COVID and to develop treatments for those suffering from [postacute COVID-19 syndrome]” according to a Feb. 5, 2021, blog from the National Institute of Neurological Disorders and Stroke (NINDS).
That same day, another NINDS blog announced “new resources for large-scale ME/CFS research” and emphasized the tie-in with long–COVID-19 syndrome.
“That’s a huge investment. In my opinion, there will be several lingering illnesses following COVID,” Dr. Komaroff said, adding, “It’s my bet that long COVID will prove to be caused by certain kinds of abnormalities in the brain, some of the same abnormalities already identified in ME/CFS. Research will determine whether that’s right or wrong.”
In 2017, NINDS had announced a large increase in funding for ME/CFS research, including the creation of four dedicated research centers. In April 2019, NINDS held a 2-day conference highlighting that ongoing work, as reported by Medscape Medical News.
During the briefing, NINDS clinical director Avindra Nath, MD, described a comprehensive ongoing ME/CFS intramural study he’s been leading since 2016.
He’s now also overseeing two long–COVID-19 studies, one of which has a protocol similar to that of the ME/CFS study and will include individuals who are still experiencing long-term symptoms following confirmed cases of COVID-19. The aim is to screen about 1,300 patients. Several task forces are now examining all of these data together.
“Each aspect is now being analyzed … What we learn from one applies to the other,” Dr. Nath said.
Advice for clinicians
In interviews, Dr. Bateman and Dr. Nath offered clinical advice for managing patients who meet ME/CFS criteria, whether they had confirmed or suspected COVID-19, a different infection, or unknown trigger(s).
Dr. Bateman advised that clinicians assess patients for each of the symptoms individually. “Besides exercise intolerance and PEM, the most commonly missed is orthostatic intolerance. It really doesn’t matter what the cause is, it’s amenable to supportive treatment. It’s one aspect of the illness that contributes to severely impaired function. My plea to all physicians would be for sure to assess for [orthostatic intolerance], and gain an understanding about activity management and avoiding PEM symptoms.”
Dr. Nath noted that an often-challenging situation is when tests for the infectious agent and other blood work come back negative, yet the patient still reports multiple debilitating symptoms. This has been a particular issue with long COVID-19, since many patients became ill early in the pandemic before the polymerase chain reaction (PCR) tests for SARS-CoV-2 were widely available.
“The physician can only order tests that are available at their labs. I think what the physician should do is handle symptoms symptomatically but also refer patients to specialists who are taking care of these patients or to research studies,” he said.
Dr. Bateman added, “Whether they had a documented COVID infection – we just have to let go of that in 2020. Way too many people didn’t have access to a test or the timing wasn’t amenable. If people meet criteria for ME/CFS, it’s irrelevant … It’s mainly a clinical diagnosis. It’s not reliant on identifying the infectious trigger.”
Dr. Komaroff, who began caring for then-termed “chronic fatigue syndrome” patients and researching the condition more than 30 years ago, said that “every cloud has its silver lining. The increased focus on postinfectious fatigue syndrome is a silver lining in my mind around the terrible dark cloud that is the pandemic of COVID.”
Dr. Komaroff has received personal fees from Serimmune Inc., Ono Pharma, and Deallus, and grants from the NIH. Dr. Bateman is employed by the Bateman Horne Center, which receives grants from the NIH, and fees from Exagen Inc., and Teva Pharmaceutical. Dr. Nath is an NIH employee.
A version of this article first appeared on Medscape.com.
People experiencing long-term symptoms following acute COVID-19 infection are increasingly meeting criteria for myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS), a phenomenon that highlights the need for unified research and clinical approaches, speakers said at a press briefing March 25 held by the advocacy group MEAction.
“Post-COVID lingering illness was predictable. Similar lingering fatigue syndromes have been reported in the scientific literature for nearly 100 years, following a variety of well-documented infections with viruses, bacteria, fungi, and even protozoa,” said Anthony Komaroff, MD, professor of medicine at Harvard Medical School, Boston.
Core criteria for ME/CFS established by the Institute of Medicine in 2015 include substantial decrement in functioning for at least 6 months, postexertional malaise (PEM), or a worsening of symptoms following even minor exertion (often described as “crashes”), unrefreshing sleep, and cognitive impairment and/or orthostatic intolerance.
Patients with ME/CFS also commonly experience painful headaches, muscle or joint aches, and allergies/other sensitivities. Although many patients can trace their symptoms to an initiating infection, “the cause is often unclear because the diagnosis is often delayed for months or years after symptom onset,” said Lucinda Bateman, MD, founder of the Bateman Horne Center, Salt Lake City, who leads a clinician coalition that aims to improve ME/CFS management.
In an international survey of 3762 COVID-19 “long-haulers” published in a preprint in December of 2020, the most frequent symptoms reported at least 6 months after illness onset were fatigue in 78%, PEM in 72%, and cognitive dysfunction (“brain fog”) in 55%. At the time of the survey, 45% reported requiring reduced work schedules because of their illness, and 22% reported being unable to work at all.
Dr. Bateman said those findings align with her experience so far with 12 COVID-19 “long haulers” who self-referred to her ME/CFS and fibromyalgia specialty clinic. Nine of the 12 met criteria for postural orthostatic tachycardia syndrome (POTS) based on the 10-minute NASA Lean Test, she said, and half also met the 2016 American College of Rheumatology criteria for fibromyalgia.
“Some were severely impaired. We suspect a small fiber polyneuropathy in about half, and mast cell activation syndrome in more than half. We look forward to doing more testing,” Dr. Bateman said.
To be sure, Dr. Komaroff noted, there are some differences. “Long COVID” patients will often experience breathlessness and ongoing anosmia (loss of taste and smell), which aren’t typical of ME/CFS.
But, he said, “many of the symptoms are quite similar ... My guess is that ME/CFS is an illness with a final common pathway that can be triggered by different things,” said Dr. Komaroff, a senior physician at Brigham and Women’s Hospital in Boston, and editor-in-chief of the Harvard Health Letter.
Based on previous data about CFS suggesting a 10% rate of symptoms persisting at least a year following a variety of infectious agents and the predicted 200 million COVID-19 cases globally by the end of 2021, Dr. Komaroff estimated that about 20 million cases of “long COVID” would be expected in the next year.
‘A huge investment’
On the research side, the National Institutes of Health recently appropriated $1.15 billion dollars over the next 4 years to investigate “the heterogeneity in the recovery process after COVID and to develop treatments for those suffering from [postacute COVID-19 syndrome]” according to a Feb. 5, 2021, blog from the National Institute of Neurological Disorders and Stroke (NINDS).
That same day, another NINDS blog announced “new resources for large-scale ME/CFS research” and emphasized the tie-in with long–COVID-19 syndrome.
“That’s a huge investment. In my opinion, there will be several lingering illnesses following COVID,” Dr. Komaroff said, adding, “It’s my bet that long COVID will prove to be caused by certain kinds of abnormalities in the brain, some of the same abnormalities already identified in ME/CFS. Research will determine whether that’s right or wrong.”
In 2017, NINDS had announced a large increase in funding for ME/CFS research, including the creation of four dedicated research centers. In April 2019, NINDS held a 2-day conference highlighting that ongoing work, as reported by Medscape Medical News.
During the briefing, NINDS clinical director Avindra Nath, MD, described a comprehensive ongoing ME/CFS intramural study he’s been leading since 2016.
He’s now also overseeing two long–COVID-19 studies, one of which has a protocol similar to that of the ME/CFS study and will include individuals who are still experiencing long-term symptoms following confirmed cases of COVID-19. The aim is to screen about 1,300 patients. Several task forces are now examining all of these data together.
“Each aspect is now being analyzed … What we learn from one applies to the other,” Dr. Nath said.
Advice for clinicians
In interviews, Dr. Bateman and Dr. Nath offered clinical advice for managing patients who meet ME/CFS criteria, whether they had confirmed or suspected COVID-19, a different infection, or unknown trigger(s).
Dr. Bateman advised that clinicians assess patients for each of the symptoms individually. “Besides exercise intolerance and PEM, the most commonly missed is orthostatic intolerance. It really doesn’t matter what the cause is, it’s amenable to supportive treatment. It’s one aspect of the illness that contributes to severely impaired function. My plea to all physicians would be for sure to assess for [orthostatic intolerance], and gain an understanding about activity management and avoiding PEM symptoms.”
Dr. Nath noted that an often-challenging situation is when tests for the infectious agent and other blood work come back negative, yet the patient still reports multiple debilitating symptoms. This has been a particular issue with long COVID-19, since many patients became ill early in the pandemic before the polymerase chain reaction (PCR) tests for SARS-CoV-2 were widely available.
“The physician can only order tests that are available at their labs. I think what the physician should do is handle symptoms symptomatically but also refer patients to specialists who are taking care of these patients or to research studies,” he said.
Dr. Bateman added, “Whether they had a documented COVID infection – we just have to let go of that in 2020. Way too many people didn’t have access to a test or the timing wasn’t amenable. If people meet criteria for ME/CFS, it’s irrelevant … It’s mainly a clinical diagnosis. It’s not reliant on identifying the infectious trigger.”
Dr. Komaroff, who began caring for then-termed “chronic fatigue syndrome” patients and researching the condition more than 30 years ago, said that “every cloud has its silver lining. The increased focus on postinfectious fatigue syndrome is a silver lining in my mind around the terrible dark cloud that is the pandemic of COVID.”
Dr. Komaroff has received personal fees from Serimmune Inc., Ono Pharma, and Deallus, and grants from the NIH. Dr. Bateman is employed by the Bateman Horne Center, which receives grants from the NIH, and fees from Exagen Inc., and Teva Pharmaceutical. Dr. Nath is an NIH employee.
A version of this article first appeared on Medscape.com.
People experiencing long-term symptoms following acute COVID-19 infection are increasingly meeting criteria for myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS), a phenomenon that highlights the need for unified research and clinical approaches, speakers said at a press briefing March 25 held by the advocacy group MEAction.
“Post-COVID lingering illness was predictable. Similar lingering fatigue syndromes have been reported in the scientific literature for nearly 100 years, following a variety of well-documented infections with viruses, bacteria, fungi, and even protozoa,” said Anthony Komaroff, MD, professor of medicine at Harvard Medical School, Boston.
Core criteria for ME/CFS established by the Institute of Medicine in 2015 include substantial decrement in functioning for at least 6 months, postexertional malaise (PEM), or a worsening of symptoms following even minor exertion (often described as “crashes”), unrefreshing sleep, and cognitive impairment and/or orthostatic intolerance.
Patients with ME/CFS also commonly experience painful headaches, muscle or joint aches, and allergies/other sensitivities. Although many patients can trace their symptoms to an initiating infection, “the cause is often unclear because the diagnosis is often delayed for months or years after symptom onset,” said Lucinda Bateman, MD, founder of the Bateman Horne Center, Salt Lake City, who leads a clinician coalition that aims to improve ME/CFS management.
In an international survey of 3762 COVID-19 “long-haulers” published in a preprint in December of 2020, the most frequent symptoms reported at least 6 months after illness onset were fatigue in 78%, PEM in 72%, and cognitive dysfunction (“brain fog”) in 55%. At the time of the survey, 45% reported requiring reduced work schedules because of their illness, and 22% reported being unable to work at all.
Dr. Bateman said those findings align with her experience so far with 12 COVID-19 “long haulers” who self-referred to her ME/CFS and fibromyalgia specialty clinic. Nine of the 12 met criteria for postural orthostatic tachycardia syndrome (POTS) based on the 10-minute NASA Lean Test, she said, and half also met the 2016 American College of Rheumatology criteria for fibromyalgia.
“Some were severely impaired. We suspect a small fiber polyneuropathy in about half, and mast cell activation syndrome in more than half. We look forward to doing more testing,” Dr. Bateman said.
To be sure, Dr. Komaroff noted, there are some differences. “Long COVID” patients will often experience breathlessness and ongoing anosmia (loss of taste and smell), which aren’t typical of ME/CFS.
But, he said, “many of the symptoms are quite similar ... My guess is that ME/CFS is an illness with a final common pathway that can be triggered by different things,” said Dr. Komaroff, a senior physician at Brigham and Women’s Hospital in Boston, and editor-in-chief of the Harvard Health Letter.
Based on previous data about CFS suggesting a 10% rate of symptoms persisting at least a year following a variety of infectious agents and the predicted 200 million COVID-19 cases globally by the end of 2021, Dr. Komaroff estimated that about 20 million cases of “long COVID” would be expected in the next year.
‘A huge investment’
On the research side, the National Institutes of Health recently appropriated $1.15 billion dollars over the next 4 years to investigate “the heterogeneity in the recovery process after COVID and to develop treatments for those suffering from [postacute COVID-19 syndrome]” according to a Feb. 5, 2021, blog from the National Institute of Neurological Disorders and Stroke (NINDS).
That same day, another NINDS blog announced “new resources for large-scale ME/CFS research” and emphasized the tie-in with long–COVID-19 syndrome.
“That’s a huge investment. In my opinion, there will be several lingering illnesses following COVID,” Dr. Komaroff said, adding, “It’s my bet that long COVID will prove to be caused by certain kinds of abnormalities in the brain, some of the same abnormalities already identified in ME/CFS. Research will determine whether that’s right or wrong.”
In 2017, NINDS had announced a large increase in funding for ME/CFS research, including the creation of four dedicated research centers. In April 2019, NINDS held a 2-day conference highlighting that ongoing work, as reported by Medscape Medical News.
During the briefing, NINDS clinical director Avindra Nath, MD, described a comprehensive ongoing ME/CFS intramural study he’s been leading since 2016.
He’s now also overseeing two long–COVID-19 studies, one of which has a protocol similar to that of the ME/CFS study and will include individuals who are still experiencing long-term symptoms following confirmed cases of COVID-19. The aim is to screen about 1,300 patients. Several task forces are now examining all of these data together.
“Each aspect is now being analyzed … What we learn from one applies to the other,” Dr. Nath said.
Advice for clinicians
In interviews, Dr. Bateman and Dr. Nath offered clinical advice for managing patients who meet ME/CFS criteria, whether they had confirmed or suspected COVID-19, a different infection, or unknown trigger(s).
Dr. Bateman advised that clinicians assess patients for each of the symptoms individually. “Besides exercise intolerance and PEM, the most commonly missed is orthostatic intolerance. It really doesn’t matter what the cause is, it’s amenable to supportive treatment. It’s one aspect of the illness that contributes to severely impaired function. My plea to all physicians would be for sure to assess for [orthostatic intolerance], and gain an understanding about activity management and avoiding PEM symptoms.”
Dr. Nath noted that an often-challenging situation is when tests for the infectious agent and other blood work come back negative, yet the patient still reports multiple debilitating symptoms. This has been a particular issue with long COVID-19, since many patients became ill early in the pandemic before the polymerase chain reaction (PCR) tests for SARS-CoV-2 were widely available.
“The physician can only order tests that are available at their labs. I think what the physician should do is handle symptoms symptomatically but also refer patients to specialists who are taking care of these patients or to research studies,” he said.
Dr. Bateman added, “Whether they had a documented COVID infection – we just have to let go of that in 2020. Way too many people didn’t have access to a test or the timing wasn’t amenable. If people meet criteria for ME/CFS, it’s irrelevant … It’s mainly a clinical diagnosis. It’s not reliant on identifying the infectious trigger.”
Dr. Komaroff, who began caring for then-termed “chronic fatigue syndrome” patients and researching the condition more than 30 years ago, said that “every cloud has its silver lining. The increased focus on postinfectious fatigue syndrome is a silver lining in my mind around the terrible dark cloud that is the pandemic of COVID.”
Dr. Komaroff has received personal fees from Serimmune Inc., Ono Pharma, and Deallus, and grants from the NIH. Dr. Bateman is employed by the Bateman Horne Center, which receives grants from the NIH, and fees from Exagen Inc., and Teva Pharmaceutical. Dr. Nath is an NIH employee.
A version of this article first appeared on Medscape.com.












