User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
nav[contains(@class, 'nav-ce-stack nav-ce-stack__large-screen')]
header[@id='header']
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'main-prefix')]
footer[@id='footer']
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
div[contains(@class, 'view-medstat-quiz-listing-panes')]
Step therapy: Inside the fight against insurance companies and fail-first medicine
Every day Melissa Fulton, RN, MSN, FNP, APRN-C, shows up to work, she’s ready for another fight. An advanced practice nurse who specializes in multiple sclerosis care, Ms. Fulton said she typically spends more than a third of her time battling it out with insurance companies over drugs she knows her patients need but that insurers don’t want to cover. Instead, they want the patient to first receive less expensive and often less efficacious drugs, even if that goes against recommendations and, in some cases, against the patient’s medical history.
The maddening protocol – familiar to health care providers everywhere – is known as “step therapy.” It forces patients to try alternative medications – medications that often fail – before receiving the one initially prescribed. The process can take weeks or months, which is time that some patients don’t have. Step therapy was sold as a way to lower costs. However, beyond the ethically problematic notion of forcing sick patients to receiver cheaper alternatives that are ineffective, research has also shown it may actually be more costly in the long run.
Ms. Fulton, who works at Saunders Medical Center in Wahoo, Neb., is a veteran in the war against step therapy. She is used to pushing her appeals up the insurance company chain of command, past nonmedical reviewers, until her patient’s case finally lands on the desk of someone with a neurology background. She said that can take three or four appeals – a judge might even get involved – and the patient could still lose. “This happens constantly,” she said, “but we fight like hell.”
Fortunately, life may soon get a little easier for Ms. Fulton. In late March, a bill to restrict step therapy made it through the Nebraska state legislature and is on its way to the governor’s desk. The Step Therapy Reform Act doesn’t outright ban the practice; however, it will put guardrails in place. It requires that insurers respond to appeals within certain time frames, and it creates key exemptions.
When the governor signs off, Nebraska will join more than two dozen other states that already have step therapy restrictions on the books, according to Hannah Lynch, MPS, associate director of federal government relations and health policy at the National Psoriasis Foundation, a leading advocate to reform and protect against the insurance practice. “There’s a lot of frustration out there,” Ms. Lynch said. “It really hinders providers’ ability to make decisions they think will have the best outcomes.”
Driven by coalitions of doctors, nurses, and patients, laws reining in step therapy have been adopted at a relatively quick clip, mostly within the past 5 years. Recent additions include South Dakota and North Carolina, which adopted step therapy laws in 2020, and Arkansas, which passed a law earlier this year.
Ms. Lynch attributed growing support to rising out-of-pocket drug costs and the introduction of biologic drugs, which are often more effective but also more expensive. Like Nebraska’s law, most step therapy reform legislation carves out exemptions and requires timely appeals processes; however, many of the laws still have significant gaps, such as not including certain types of insurance plans.
Ideally, Ms. Lynch said, the protections would apply to all types of health plans that are regulated at the state level, such as Medicaid, state employee health plans, and coverage sold through state insurance exchanges. Closing loopholes in the laws is a top priority for advocates, she added, pointing to work currently underway in Arkansas to extend its new protections to Medicaid expansion patients.
“With so many outside stakeholders, you have to compromise – it’s a give and take,” Ms. Lynch said. Still, when it comes to fighting step therapy, she says, “Any protection on the books is always our first goal when we go into a state.”
Putting patients first
Lisa Arkin, MD, a pediatric dermatologist at the University of Wisconsin–Madison, said she finds herself “swimming upstream every day in the fight with insurance.” Her patients are typically on their second or third stop and have more complex disorders. Dr. Arkin said that the problem with step therapy is that it tries to squeeze all patients into the same box, even if the circumstances don’t fit.
Her state passed restrictions on step therapy in 2019, but the measures only went into effect last year. Under the Wisconsin law, patients can be granted an exemption if an alternative treatment is contraindicated, likely to cause harm, or expected to be ineffective. Patients can also be exempt if their current treatment is working.
Dr. Arkin, an outspoken advocate for curbing step therapy, says the Wisconsin law is “very strong.” However, because it only applies to certain health plans – state employee health plans and those purchased in the state’s health insurance exchange – fewer than half the state’s patients benefit from its protections. She notes that some of the most severe presentations she treats occur in patients who rely on Medicaid coverage and already face barriers to care.
“I’m a doctor who puts up a fuss [with insurers], but that’s not fair – we shouldn’t have to do that,” Dr. Arkin said. “To me, it’s really critical to make this an even playing field so this law affords protection to everyone I see in the clinic.”
Major medical associations caution against step therapy as well. The American Society of Clinical Oncology and the American Medical Association have called out the risks to patient safety and health. In fact, in 2019, after the Centers for Medicare & Medicaid Services gave new authority to Medicare Advantage plans to start using step therapy, dozens of national medical groups called out the agency for allowing a practice that could potentially hurt patients and undercut the physician-patient decision-making process.
Last year, in a new position paper from the American College of Physicians, authors laid out recommendations for combating step therapy’s side effects. These recommendations included making related data transparent to the public and minimizing the policy’s disruptions to care. Jacqueline W. Fincher, MD, MACP, a member of the committee that issued the position paper and who is a primary care physician in Georgia, said such insurance practices need to be designed with “strong input from frontline physicians, not clipboard physicians.
“What we want from insurers is understanding, transparency, and the least burdensome protocol to provide patients the care they need at a cost-effective price they can afford,” said Dr. Fincher, who is also the current president of the ACP. “The focus needs to be on what’s in the patient’s best interest.”
Every year a new fight
“We all dread January,” said Dr. Fincher. That is the worst month, she added, because new health benefits go into effect, which means patients who are responding well to certain treatments may suddenly face new restrictions.
Another aggravating aspect of step therapy? It is often difficult – if not impossible – to access information on specific step therapy protocols in a patient’s health plan in real time in the exam room, where treatment conversations actually take place. In a more patient-centered world, Dr. Fincher said, she would be able to use the electronic health record system to quickly identify whether a patient’s plan covers a particular treatment and, if not, what the alternatives are.
Georgia’s new step therapy law went into effect last year. Like laws in other states, it spells out step therapy exemptions and sets time frames in which insurers must respond to exceptions and appeals. Dr. Fincher, who spoke in favor of the new law, said she’s “happy for any step forward.” Still, the growing burden of prior authorization rules are an utter “time sink” for her and her staff.
“I have to justify my decisions to nondoctors before I even get to a doctor, and that’s really frustrating,” she said. “We’re talking about people here, not widgets.”
Advocates in Nevada are hoping this is the year a step therapy bill will make it into law in their state. As of March, one had yet to be introduced in the state legislature. Tom McCoy, director of state government affairs at the Nevada Chronic Care Collaborative, said existing Nevada law already prohibits nonmedical drug switching during a policy year; however, insurers can still make changes the following year.
A bill to rein in step therapy was proposed previously, Mr. McCoy said, but it never got off the ground. The collaborative, as well as about two dozen organizations representing Nevada providers and patients, are now calling on state lawmakers to make the issue a priority in the current session.
“The health plans have a lot of power – a lot,” Mr. McCoy said. “We’re hoping to get a [legislative] sponsor in 2021 ... but it’s also been a really hard year to connect legislators with patients and doctors, and being able to hear their stories really does make a difference.”
In Nebraska, Marcus Snow, MD, a rheumatologist at Nebraska Medicine, in Omaha, said that the state’s new step therapy law will be a “great first step in helping to provide some guardrails” around the practice. He noted that turnaround requirements for insurer responses are “sorely needed.” However, he said that, because the bill doesn’t apply to all health plans, many Nebraskans still won’t benefit.
Dealing with step therapy is a daily “headache” for Dr. Snow, who says navigating the bureaucracy of prior authorization seems to be getting worse every year. Like his peers around the country, he spends an inordinate amount of time pushing appeals up the insurance company ranks to get access to treatments he believes will be most effective. But Snow says that, more than just being a mountain of tiresome red tape, these practices also intrude on the patient-provider relationship, casting an unsettling sense of uncertainty that the ultimate decision about the best course of action isn’t up to the doctor and patient at all.
“In the end, the insurance company is the judge and jury of my prescription,” Dr. Snow said. “They’d argue I can still prescribe it, but if it costs $70,000 a year – I don’t know who can afford that.”
Ms. Lynch, at the National Psoriasis Foundation, said their step therapy advocacy will continue to take a two-pronged approach. They will push for new and expanded protections at both state and federal levels. Protections are needed at both levels to make sure that all health plans regulated by all entities are covered. In the U.S. Senate and the House, step therapy bills were reintroduced this year. They would apply to health plans subject to the federal Employee Retirement Income Security Act, which governs employer-sponsored health coverage, and could close a big gap in existing protections. Oregon, New Jersey, and Arizona are at the top of the foundation’s advocacy list this year, according to Ms. Lynch.
“Folks are really starting to pay more attention to this issue,” she said. “And hearing those real-world stories and frustrations is definitely one of the most effective tools we have.”
A version of this article first appeared on Medscape.com.
Every day Melissa Fulton, RN, MSN, FNP, APRN-C, shows up to work, she’s ready for another fight. An advanced practice nurse who specializes in multiple sclerosis care, Ms. Fulton said she typically spends more than a third of her time battling it out with insurance companies over drugs she knows her patients need but that insurers don’t want to cover. Instead, they want the patient to first receive less expensive and often less efficacious drugs, even if that goes against recommendations and, in some cases, against the patient’s medical history.
The maddening protocol – familiar to health care providers everywhere – is known as “step therapy.” It forces patients to try alternative medications – medications that often fail – before receiving the one initially prescribed. The process can take weeks or months, which is time that some patients don’t have. Step therapy was sold as a way to lower costs. However, beyond the ethically problematic notion of forcing sick patients to receiver cheaper alternatives that are ineffective, research has also shown it may actually be more costly in the long run.
Ms. Fulton, who works at Saunders Medical Center in Wahoo, Neb., is a veteran in the war against step therapy. She is used to pushing her appeals up the insurance company chain of command, past nonmedical reviewers, until her patient’s case finally lands on the desk of someone with a neurology background. She said that can take three or four appeals – a judge might even get involved – and the patient could still lose. “This happens constantly,” she said, “but we fight like hell.”
Fortunately, life may soon get a little easier for Ms. Fulton. In late March, a bill to restrict step therapy made it through the Nebraska state legislature and is on its way to the governor’s desk. The Step Therapy Reform Act doesn’t outright ban the practice; however, it will put guardrails in place. It requires that insurers respond to appeals within certain time frames, and it creates key exemptions.
When the governor signs off, Nebraska will join more than two dozen other states that already have step therapy restrictions on the books, according to Hannah Lynch, MPS, associate director of federal government relations and health policy at the National Psoriasis Foundation, a leading advocate to reform and protect against the insurance practice. “There’s a lot of frustration out there,” Ms. Lynch said. “It really hinders providers’ ability to make decisions they think will have the best outcomes.”
Driven by coalitions of doctors, nurses, and patients, laws reining in step therapy have been adopted at a relatively quick clip, mostly within the past 5 years. Recent additions include South Dakota and North Carolina, which adopted step therapy laws in 2020, and Arkansas, which passed a law earlier this year.
Ms. Lynch attributed growing support to rising out-of-pocket drug costs and the introduction of biologic drugs, which are often more effective but also more expensive. Like Nebraska’s law, most step therapy reform legislation carves out exemptions and requires timely appeals processes; however, many of the laws still have significant gaps, such as not including certain types of insurance plans.
Ideally, Ms. Lynch said, the protections would apply to all types of health plans that are regulated at the state level, such as Medicaid, state employee health plans, and coverage sold through state insurance exchanges. Closing loopholes in the laws is a top priority for advocates, she added, pointing to work currently underway in Arkansas to extend its new protections to Medicaid expansion patients.
“With so many outside stakeholders, you have to compromise – it’s a give and take,” Ms. Lynch said. Still, when it comes to fighting step therapy, she says, “Any protection on the books is always our first goal when we go into a state.”
Putting patients first
Lisa Arkin, MD, a pediatric dermatologist at the University of Wisconsin–Madison, said she finds herself “swimming upstream every day in the fight with insurance.” Her patients are typically on their second or third stop and have more complex disorders. Dr. Arkin said that the problem with step therapy is that it tries to squeeze all patients into the same box, even if the circumstances don’t fit.
Her state passed restrictions on step therapy in 2019, but the measures only went into effect last year. Under the Wisconsin law, patients can be granted an exemption if an alternative treatment is contraindicated, likely to cause harm, or expected to be ineffective. Patients can also be exempt if their current treatment is working.
Dr. Arkin, an outspoken advocate for curbing step therapy, says the Wisconsin law is “very strong.” However, because it only applies to certain health plans – state employee health plans and those purchased in the state’s health insurance exchange – fewer than half the state’s patients benefit from its protections. She notes that some of the most severe presentations she treats occur in patients who rely on Medicaid coverage and already face barriers to care.
“I’m a doctor who puts up a fuss [with insurers], but that’s not fair – we shouldn’t have to do that,” Dr. Arkin said. “To me, it’s really critical to make this an even playing field so this law affords protection to everyone I see in the clinic.”
Major medical associations caution against step therapy as well. The American Society of Clinical Oncology and the American Medical Association have called out the risks to patient safety and health. In fact, in 2019, after the Centers for Medicare & Medicaid Services gave new authority to Medicare Advantage plans to start using step therapy, dozens of national medical groups called out the agency for allowing a practice that could potentially hurt patients and undercut the physician-patient decision-making process.
Last year, in a new position paper from the American College of Physicians, authors laid out recommendations for combating step therapy’s side effects. These recommendations included making related data transparent to the public and minimizing the policy’s disruptions to care. Jacqueline W. Fincher, MD, MACP, a member of the committee that issued the position paper and who is a primary care physician in Georgia, said such insurance practices need to be designed with “strong input from frontline physicians, not clipboard physicians.
“What we want from insurers is understanding, transparency, and the least burdensome protocol to provide patients the care they need at a cost-effective price they can afford,” said Dr. Fincher, who is also the current president of the ACP. “The focus needs to be on what’s in the patient’s best interest.”
Every year a new fight
“We all dread January,” said Dr. Fincher. That is the worst month, she added, because new health benefits go into effect, which means patients who are responding well to certain treatments may suddenly face new restrictions.
Another aggravating aspect of step therapy? It is often difficult – if not impossible – to access information on specific step therapy protocols in a patient’s health plan in real time in the exam room, where treatment conversations actually take place. In a more patient-centered world, Dr. Fincher said, she would be able to use the electronic health record system to quickly identify whether a patient’s plan covers a particular treatment and, if not, what the alternatives are.
Georgia’s new step therapy law went into effect last year. Like laws in other states, it spells out step therapy exemptions and sets time frames in which insurers must respond to exceptions and appeals. Dr. Fincher, who spoke in favor of the new law, said she’s “happy for any step forward.” Still, the growing burden of prior authorization rules are an utter “time sink” for her and her staff.
“I have to justify my decisions to nondoctors before I even get to a doctor, and that’s really frustrating,” she said. “We’re talking about people here, not widgets.”
Advocates in Nevada are hoping this is the year a step therapy bill will make it into law in their state. As of March, one had yet to be introduced in the state legislature. Tom McCoy, director of state government affairs at the Nevada Chronic Care Collaborative, said existing Nevada law already prohibits nonmedical drug switching during a policy year; however, insurers can still make changes the following year.
A bill to rein in step therapy was proposed previously, Mr. McCoy said, but it never got off the ground. The collaborative, as well as about two dozen organizations representing Nevada providers and patients, are now calling on state lawmakers to make the issue a priority in the current session.
“The health plans have a lot of power – a lot,” Mr. McCoy said. “We’re hoping to get a [legislative] sponsor in 2021 ... but it’s also been a really hard year to connect legislators with patients and doctors, and being able to hear their stories really does make a difference.”
In Nebraska, Marcus Snow, MD, a rheumatologist at Nebraska Medicine, in Omaha, said that the state’s new step therapy law will be a “great first step in helping to provide some guardrails” around the practice. He noted that turnaround requirements for insurer responses are “sorely needed.” However, he said that, because the bill doesn’t apply to all health plans, many Nebraskans still won’t benefit.
Dealing with step therapy is a daily “headache” for Dr. Snow, who says navigating the bureaucracy of prior authorization seems to be getting worse every year. Like his peers around the country, he spends an inordinate amount of time pushing appeals up the insurance company ranks to get access to treatments he believes will be most effective. But Snow says that, more than just being a mountain of tiresome red tape, these practices also intrude on the patient-provider relationship, casting an unsettling sense of uncertainty that the ultimate decision about the best course of action isn’t up to the doctor and patient at all.
“In the end, the insurance company is the judge and jury of my prescription,” Dr. Snow said. “They’d argue I can still prescribe it, but if it costs $70,000 a year – I don’t know who can afford that.”
Ms. Lynch, at the National Psoriasis Foundation, said their step therapy advocacy will continue to take a two-pronged approach. They will push for new and expanded protections at both state and federal levels. Protections are needed at both levels to make sure that all health plans regulated by all entities are covered. In the U.S. Senate and the House, step therapy bills were reintroduced this year. They would apply to health plans subject to the federal Employee Retirement Income Security Act, which governs employer-sponsored health coverage, and could close a big gap in existing protections. Oregon, New Jersey, and Arizona are at the top of the foundation’s advocacy list this year, according to Ms. Lynch.
“Folks are really starting to pay more attention to this issue,” she said. “And hearing those real-world stories and frustrations is definitely one of the most effective tools we have.”
A version of this article first appeared on Medscape.com.
Every day Melissa Fulton, RN, MSN, FNP, APRN-C, shows up to work, she’s ready for another fight. An advanced practice nurse who specializes in multiple sclerosis care, Ms. Fulton said she typically spends more than a third of her time battling it out with insurance companies over drugs she knows her patients need but that insurers don’t want to cover. Instead, they want the patient to first receive less expensive and often less efficacious drugs, even if that goes against recommendations and, in some cases, against the patient’s medical history.
The maddening protocol – familiar to health care providers everywhere – is known as “step therapy.” It forces patients to try alternative medications – medications that often fail – before receiving the one initially prescribed. The process can take weeks or months, which is time that some patients don’t have. Step therapy was sold as a way to lower costs. However, beyond the ethically problematic notion of forcing sick patients to receiver cheaper alternatives that are ineffective, research has also shown it may actually be more costly in the long run.
Ms. Fulton, who works at Saunders Medical Center in Wahoo, Neb., is a veteran in the war against step therapy. She is used to pushing her appeals up the insurance company chain of command, past nonmedical reviewers, until her patient’s case finally lands on the desk of someone with a neurology background. She said that can take three or four appeals – a judge might even get involved – and the patient could still lose. “This happens constantly,” she said, “but we fight like hell.”
Fortunately, life may soon get a little easier for Ms. Fulton. In late March, a bill to restrict step therapy made it through the Nebraska state legislature and is on its way to the governor’s desk. The Step Therapy Reform Act doesn’t outright ban the practice; however, it will put guardrails in place. It requires that insurers respond to appeals within certain time frames, and it creates key exemptions.
When the governor signs off, Nebraska will join more than two dozen other states that already have step therapy restrictions on the books, according to Hannah Lynch, MPS, associate director of federal government relations and health policy at the National Psoriasis Foundation, a leading advocate to reform and protect against the insurance practice. “There’s a lot of frustration out there,” Ms. Lynch said. “It really hinders providers’ ability to make decisions they think will have the best outcomes.”
Driven by coalitions of doctors, nurses, and patients, laws reining in step therapy have been adopted at a relatively quick clip, mostly within the past 5 years. Recent additions include South Dakota and North Carolina, which adopted step therapy laws in 2020, and Arkansas, which passed a law earlier this year.
Ms. Lynch attributed growing support to rising out-of-pocket drug costs and the introduction of biologic drugs, which are often more effective but also more expensive. Like Nebraska’s law, most step therapy reform legislation carves out exemptions and requires timely appeals processes; however, many of the laws still have significant gaps, such as not including certain types of insurance plans.
Ideally, Ms. Lynch said, the protections would apply to all types of health plans that are regulated at the state level, such as Medicaid, state employee health plans, and coverage sold through state insurance exchanges. Closing loopholes in the laws is a top priority for advocates, she added, pointing to work currently underway in Arkansas to extend its new protections to Medicaid expansion patients.
“With so many outside stakeholders, you have to compromise – it’s a give and take,” Ms. Lynch said. Still, when it comes to fighting step therapy, she says, “Any protection on the books is always our first goal when we go into a state.”
Putting patients first
Lisa Arkin, MD, a pediatric dermatologist at the University of Wisconsin–Madison, said she finds herself “swimming upstream every day in the fight with insurance.” Her patients are typically on their second or third stop and have more complex disorders. Dr. Arkin said that the problem with step therapy is that it tries to squeeze all patients into the same box, even if the circumstances don’t fit.
Her state passed restrictions on step therapy in 2019, but the measures only went into effect last year. Under the Wisconsin law, patients can be granted an exemption if an alternative treatment is contraindicated, likely to cause harm, or expected to be ineffective. Patients can also be exempt if their current treatment is working.
Dr. Arkin, an outspoken advocate for curbing step therapy, says the Wisconsin law is “very strong.” However, because it only applies to certain health plans – state employee health plans and those purchased in the state’s health insurance exchange – fewer than half the state’s patients benefit from its protections. She notes that some of the most severe presentations she treats occur in patients who rely on Medicaid coverage and already face barriers to care.
“I’m a doctor who puts up a fuss [with insurers], but that’s not fair – we shouldn’t have to do that,” Dr. Arkin said. “To me, it’s really critical to make this an even playing field so this law affords protection to everyone I see in the clinic.”
Major medical associations caution against step therapy as well. The American Society of Clinical Oncology and the American Medical Association have called out the risks to patient safety and health. In fact, in 2019, after the Centers for Medicare & Medicaid Services gave new authority to Medicare Advantage plans to start using step therapy, dozens of national medical groups called out the agency for allowing a practice that could potentially hurt patients and undercut the physician-patient decision-making process.
Last year, in a new position paper from the American College of Physicians, authors laid out recommendations for combating step therapy’s side effects. These recommendations included making related data transparent to the public and minimizing the policy’s disruptions to care. Jacqueline W. Fincher, MD, MACP, a member of the committee that issued the position paper and who is a primary care physician in Georgia, said such insurance practices need to be designed with “strong input from frontline physicians, not clipboard physicians.
“What we want from insurers is understanding, transparency, and the least burdensome protocol to provide patients the care they need at a cost-effective price they can afford,” said Dr. Fincher, who is also the current president of the ACP. “The focus needs to be on what’s in the patient’s best interest.”
Every year a new fight
“We all dread January,” said Dr. Fincher. That is the worst month, she added, because new health benefits go into effect, which means patients who are responding well to certain treatments may suddenly face new restrictions.
Another aggravating aspect of step therapy? It is often difficult – if not impossible – to access information on specific step therapy protocols in a patient’s health plan in real time in the exam room, where treatment conversations actually take place. In a more patient-centered world, Dr. Fincher said, she would be able to use the electronic health record system to quickly identify whether a patient’s plan covers a particular treatment and, if not, what the alternatives are.
Georgia’s new step therapy law went into effect last year. Like laws in other states, it spells out step therapy exemptions and sets time frames in which insurers must respond to exceptions and appeals. Dr. Fincher, who spoke in favor of the new law, said she’s “happy for any step forward.” Still, the growing burden of prior authorization rules are an utter “time sink” for her and her staff.
“I have to justify my decisions to nondoctors before I even get to a doctor, and that’s really frustrating,” she said. “We’re talking about people here, not widgets.”
Advocates in Nevada are hoping this is the year a step therapy bill will make it into law in their state. As of March, one had yet to be introduced in the state legislature. Tom McCoy, director of state government affairs at the Nevada Chronic Care Collaborative, said existing Nevada law already prohibits nonmedical drug switching during a policy year; however, insurers can still make changes the following year.
A bill to rein in step therapy was proposed previously, Mr. McCoy said, but it never got off the ground. The collaborative, as well as about two dozen organizations representing Nevada providers and patients, are now calling on state lawmakers to make the issue a priority in the current session.
“The health plans have a lot of power – a lot,” Mr. McCoy said. “We’re hoping to get a [legislative] sponsor in 2021 ... but it’s also been a really hard year to connect legislators with patients and doctors, and being able to hear their stories really does make a difference.”
In Nebraska, Marcus Snow, MD, a rheumatologist at Nebraska Medicine, in Omaha, said that the state’s new step therapy law will be a “great first step in helping to provide some guardrails” around the practice. He noted that turnaround requirements for insurer responses are “sorely needed.” However, he said that, because the bill doesn’t apply to all health plans, many Nebraskans still won’t benefit.
Dealing with step therapy is a daily “headache” for Dr. Snow, who says navigating the bureaucracy of prior authorization seems to be getting worse every year. Like his peers around the country, he spends an inordinate amount of time pushing appeals up the insurance company ranks to get access to treatments he believes will be most effective. But Snow says that, more than just being a mountain of tiresome red tape, these practices also intrude on the patient-provider relationship, casting an unsettling sense of uncertainty that the ultimate decision about the best course of action isn’t up to the doctor and patient at all.
“In the end, the insurance company is the judge and jury of my prescription,” Dr. Snow said. “They’d argue I can still prescribe it, but if it costs $70,000 a year – I don’t know who can afford that.”
Ms. Lynch, at the National Psoriasis Foundation, said their step therapy advocacy will continue to take a two-pronged approach. They will push for new and expanded protections at both state and federal levels. Protections are needed at both levels to make sure that all health plans regulated by all entities are covered. In the U.S. Senate and the House, step therapy bills were reintroduced this year. They would apply to health plans subject to the federal Employee Retirement Income Security Act, which governs employer-sponsored health coverage, and could close a big gap in existing protections. Oregon, New Jersey, and Arizona are at the top of the foundation’s advocacy list this year, according to Ms. Lynch.
“Folks are really starting to pay more attention to this issue,” she said. “And hearing those real-world stories and frustrations is definitely one of the most effective tools we have.”
A version of this article first appeared on Medscape.com.
Encephalopathy common, often lethal in hospitalized patients with COVID-19
, new research shows. Results of a retrospective study show that of almost 4,500 patients with COVID-19, 12% were diagnosed with TME. Of these, 78% developed encephalopathy immediately prior to hospital admission. Septic encephalopathy, hypoxic-ischemic encephalopathy (HIE), and uremia were the most common causes, although multiple causes were present in close to 80% of patients. TME was also associated with a 24% higher risk of in-hospital death.
“We found that close to one in eight patients who were hospitalized with COVID-19 had TME that was not attributed to the effects of sedatives, and that this is incredibly common among these patients who are critically ill” said lead author Jennifer A. Frontera, MD, New York University.
“The general principle of our findings is to be more aggressive in TME; and from a neurologist perspective, the way to do this is to eliminate the effects of sedation, which is a confounder,” she said.
The study was published online March 16 in Neurocritical Care.
Drilling down
“Many neurological complications of COVID-19 are sequelae of severe illness or secondary effects of multisystem organ failure, but our previous work identified TME as the most common neurological complication,” Dr. Frontera said.
Previous research investigating encephalopathy among patients with COVID-19 included patients who may have been sedated or have had a positive Confusion Assessment Method (CAM) result.
“A lot of the delirium literature is effectively heterogeneous because there are a number of patients who are on sedative medication that, if you could turn it off, these patients would return to normal. Some may have underlying neurological issues that can be addressed, but you can›t get to the bottom of this unless you turn off the sedation,” Dr. Frontera noted.
“We wanted to be specific and try to drill down to see what the underlying cause of the encephalopathy was,” she said.
The researchers retrospectively analyzed data on 4,491 patients (≥ 18 years old) with COVID-19 who were admitted to four New York City hospitals between March 1, 2020, and May 20, 2020. Of these, 559 (12%) with TME were compared with 3,932 patients without TME.
The researchers looked at index admissions and included patients who had:
- New changes in mental status or significant worsening of mental status (in patients with baseline abnormal mental status).
- Hyperglycemia or with transient focal neurologic deficits that resolved with glucose correction.
- An adequate washout of sedating medications (when relevant) prior to mental status assessment.
Potential etiologies included electrolyte abnormalities, organ failure, hypertensive encephalopathy, sepsis or active infection, fever, nutritional deficiency, and environmental injury.
Foreign environment
Most (78%) of the 559 patients diagnosed with TME had already developed encephalopathy immediately prior to hospital admission, the authors report. The most common etiologies of TME among hospitalized patients with COVID-19 are listed below.
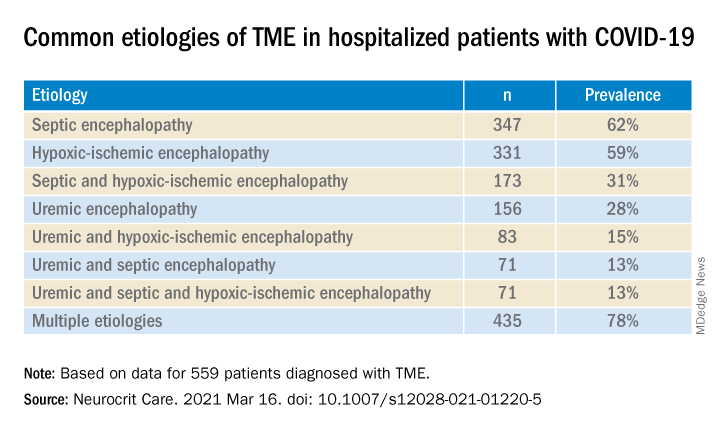
Compared with patients without TME, those with TME – (all Ps < .001):
- Were older (76 vs. 62 years).
- Had higher rates of dementia (27% vs. 3%).
- Had higher rates of psychiatric history (20% vs. 10%).
- Were more often intubated (37% vs. 20%).
- Had a longer length of hospital stay (7.9 vs. 6.0 days).
- Were less often discharged home (25% vs. 66%).
“It’s no surprise that older patients and people with dementia or psychiatric illness are predisposed to becoming encephalopathic,” said Dr. Frontera. “Being in a foreign environment, such as a hospital, or being sleep-deprived in the ICU is likely to make them more confused during their hospital stay.”
Delirium as a symptom
In-hospital mortality or discharge to hospice was considerably higher in the TME versus non-TME patients (44% vs. 18%, respectively).
When the researchers adjusted for confounders (age, sex, race, worse Sequential Organ Failure Assessment score during hospitalization, ventilator status, study week, hospital location, and ICU care level) and excluded patients receiving only comfort care, they found that TME was associated with a 24% increased risk of in-hospital death (30% in patients with TME vs. 16% in those without TME).
The highest mortality risk was associated with hypoxemia, with 42% of patients with HIE dying during hospitalization, compared with 16% of patients without HIE (adjusted hazard ratio 1.56; 95% confidence interval, 1.21-2.00; P = .001).
“Not all patients who are intubated require sedation, but there’s generally a lot of hesitation in reducing or stopping sedation in some patients,” Dr. Frontera observed.
She acknowledged there are “many extremely sick patients whom you can’t ventilate without sedation.”
Nevertheless, “delirium in and of itself does not cause death. It’s a symptom, not a disease, and we have to figure out what causes it. Delirium might not need to be sedated, and it’s more important to see what the causal problem is.”
Independent predictor of death
Commenting on the study, Panayiotis N. Varelas, MD, PhD, vice president of the Neurocritical Care Society, said the study “approached the TME issue better than previously, namely allowing time for sedatives to wear off to have a better sample of patients with this syndrome.”
Dr. Varelas, who is chairman of the department of neurology and professor of neurology at Albany (N.Y.) Medical College, emphasized that TME “is not benign and, in patients with COVID-19, it is an independent predictor of in-hospital mortality.”
“One should take all possible measures … to avoid desaturation and hypotensive episodes and also aggressively treat SAE and uremic encephalopathy in hopes of improving the outcomes,” added Dr. Varelas, who was not involved with the study.
Also commenting on the study, Mitchell Elkind, MD, professor of neurology and epidemiology at Columbia University in New York, who was not associated with the research, said it “nicely distinguishes among the different causes of encephalopathy, including sepsis, hypoxia, and kidney failure … emphasizing just how sick these patients are.”
The study received no direct funding. Individual investigators were supported by grants from the National Institute on Aging and the National Institute of Neurological Disorders and Stroke. The investigators, Dr. Varelas, and Dr. Elkind have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, new research shows. Results of a retrospective study show that of almost 4,500 patients with COVID-19, 12% were diagnosed with TME. Of these, 78% developed encephalopathy immediately prior to hospital admission. Septic encephalopathy, hypoxic-ischemic encephalopathy (HIE), and uremia were the most common causes, although multiple causes were present in close to 80% of patients. TME was also associated with a 24% higher risk of in-hospital death.
“We found that close to one in eight patients who were hospitalized with COVID-19 had TME that was not attributed to the effects of sedatives, and that this is incredibly common among these patients who are critically ill” said lead author Jennifer A. Frontera, MD, New York University.
“The general principle of our findings is to be more aggressive in TME; and from a neurologist perspective, the way to do this is to eliminate the effects of sedation, which is a confounder,” she said.
The study was published online March 16 in Neurocritical Care.
Drilling down
“Many neurological complications of COVID-19 are sequelae of severe illness or secondary effects of multisystem organ failure, but our previous work identified TME as the most common neurological complication,” Dr. Frontera said.
Previous research investigating encephalopathy among patients with COVID-19 included patients who may have been sedated or have had a positive Confusion Assessment Method (CAM) result.
“A lot of the delirium literature is effectively heterogeneous because there are a number of patients who are on sedative medication that, if you could turn it off, these patients would return to normal. Some may have underlying neurological issues that can be addressed, but you can›t get to the bottom of this unless you turn off the sedation,” Dr. Frontera noted.
“We wanted to be specific and try to drill down to see what the underlying cause of the encephalopathy was,” she said.
The researchers retrospectively analyzed data on 4,491 patients (≥ 18 years old) with COVID-19 who were admitted to four New York City hospitals between March 1, 2020, and May 20, 2020. Of these, 559 (12%) with TME were compared with 3,932 patients without TME.
The researchers looked at index admissions and included patients who had:
- New changes in mental status or significant worsening of mental status (in patients with baseline abnormal mental status).
- Hyperglycemia or with transient focal neurologic deficits that resolved with glucose correction.
- An adequate washout of sedating medications (when relevant) prior to mental status assessment.
Potential etiologies included electrolyte abnormalities, organ failure, hypertensive encephalopathy, sepsis or active infection, fever, nutritional deficiency, and environmental injury.
Foreign environment
Most (78%) of the 559 patients diagnosed with TME had already developed encephalopathy immediately prior to hospital admission, the authors report. The most common etiologies of TME among hospitalized patients with COVID-19 are listed below.

Compared with patients without TME, those with TME – (all Ps < .001):
- Were older (76 vs. 62 years).
- Had higher rates of dementia (27% vs. 3%).
- Had higher rates of psychiatric history (20% vs. 10%).
- Were more often intubated (37% vs. 20%).
- Had a longer length of hospital stay (7.9 vs. 6.0 days).
- Were less often discharged home (25% vs. 66%).
“It’s no surprise that older patients and people with dementia or psychiatric illness are predisposed to becoming encephalopathic,” said Dr. Frontera. “Being in a foreign environment, such as a hospital, or being sleep-deprived in the ICU is likely to make them more confused during their hospital stay.”
Delirium as a symptom
In-hospital mortality or discharge to hospice was considerably higher in the TME versus non-TME patients (44% vs. 18%, respectively).
When the researchers adjusted for confounders (age, sex, race, worse Sequential Organ Failure Assessment score during hospitalization, ventilator status, study week, hospital location, and ICU care level) and excluded patients receiving only comfort care, they found that TME was associated with a 24% increased risk of in-hospital death (30% in patients with TME vs. 16% in those without TME).
The highest mortality risk was associated with hypoxemia, with 42% of patients with HIE dying during hospitalization, compared with 16% of patients without HIE (adjusted hazard ratio 1.56; 95% confidence interval, 1.21-2.00; P = .001).
“Not all patients who are intubated require sedation, but there’s generally a lot of hesitation in reducing or stopping sedation in some patients,” Dr. Frontera observed.
She acknowledged there are “many extremely sick patients whom you can’t ventilate without sedation.”
Nevertheless, “delirium in and of itself does not cause death. It’s a symptom, not a disease, and we have to figure out what causes it. Delirium might not need to be sedated, and it’s more important to see what the causal problem is.”
Independent predictor of death
Commenting on the study, Panayiotis N. Varelas, MD, PhD, vice president of the Neurocritical Care Society, said the study “approached the TME issue better than previously, namely allowing time for sedatives to wear off to have a better sample of patients with this syndrome.”
Dr. Varelas, who is chairman of the department of neurology and professor of neurology at Albany (N.Y.) Medical College, emphasized that TME “is not benign and, in patients with COVID-19, it is an independent predictor of in-hospital mortality.”
“One should take all possible measures … to avoid desaturation and hypotensive episodes and also aggressively treat SAE and uremic encephalopathy in hopes of improving the outcomes,” added Dr. Varelas, who was not involved with the study.
Also commenting on the study, Mitchell Elkind, MD, professor of neurology and epidemiology at Columbia University in New York, who was not associated with the research, said it “nicely distinguishes among the different causes of encephalopathy, including sepsis, hypoxia, and kidney failure … emphasizing just how sick these patients are.”
The study received no direct funding. Individual investigators were supported by grants from the National Institute on Aging and the National Institute of Neurological Disorders and Stroke. The investigators, Dr. Varelas, and Dr. Elkind have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, new research shows. Results of a retrospective study show that of almost 4,500 patients with COVID-19, 12% were diagnosed with TME. Of these, 78% developed encephalopathy immediately prior to hospital admission. Septic encephalopathy, hypoxic-ischemic encephalopathy (HIE), and uremia were the most common causes, although multiple causes were present in close to 80% of patients. TME was also associated with a 24% higher risk of in-hospital death.
“We found that close to one in eight patients who were hospitalized with COVID-19 had TME that was not attributed to the effects of sedatives, and that this is incredibly common among these patients who are critically ill” said lead author Jennifer A. Frontera, MD, New York University.
“The general principle of our findings is to be more aggressive in TME; and from a neurologist perspective, the way to do this is to eliminate the effects of sedation, which is a confounder,” she said.
The study was published online March 16 in Neurocritical Care.
Drilling down
“Many neurological complications of COVID-19 are sequelae of severe illness or secondary effects of multisystem organ failure, but our previous work identified TME as the most common neurological complication,” Dr. Frontera said.
Previous research investigating encephalopathy among patients with COVID-19 included patients who may have been sedated or have had a positive Confusion Assessment Method (CAM) result.
“A lot of the delirium literature is effectively heterogeneous because there are a number of patients who are on sedative medication that, if you could turn it off, these patients would return to normal. Some may have underlying neurological issues that can be addressed, but you can›t get to the bottom of this unless you turn off the sedation,” Dr. Frontera noted.
“We wanted to be specific and try to drill down to see what the underlying cause of the encephalopathy was,” she said.
The researchers retrospectively analyzed data on 4,491 patients (≥ 18 years old) with COVID-19 who were admitted to four New York City hospitals between March 1, 2020, and May 20, 2020. Of these, 559 (12%) with TME were compared with 3,932 patients without TME.
The researchers looked at index admissions and included patients who had:
- New changes in mental status or significant worsening of mental status (in patients with baseline abnormal mental status).
- Hyperglycemia or with transient focal neurologic deficits that resolved with glucose correction.
- An adequate washout of sedating medications (when relevant) prior to mental status assessment.
Potential etiologies included electrolyte abnormalities, organ failure, hypertensive encephalopathy, sepsis or active infection, fever, nutritional deficiency, and environmental injury.
Foreign environment
Most (78%) of the 559 patients diagnosed with TME had already developed encephalopathy immediately prior to hospital admission, the authors report. The most common etiologies of TME among hospitalized patients with COVID-19 are listed below.

Compared with patients without TME, those with TME – (all Ps < .001):
- Were older (76 vs. 62 years).
- Had higher rates of dementia (27% vs. 3%).
- Had higher rates of psychiatric history (20% vs. 10%).
- Were more often intubated (37% vs. 20%).
- Had a longer length of hospital stay (7.9 vs. 6.0 days).
- Were less often discharged home (25% vs. 66%).
“It’s no surprise that older patients and people with dementia or psychiatric illness are predisposed to becoming encephalopathic,” said Dr. Frontera. “Being in a foreign environment, such as a hospital, or being sleep-deprived in the ICU is likely to make them more confused during their hospital stay.”
Delirium as a symptom
In-hospital mortality or discharge to hospice was considerably higher in the TME versus non-TME patients (44% vs. 18%, respectively).
When the researchers adjusted for confounders (age, sex, race, worse Sequential Organ Failure Assessment score during hospitalization, ventilator status, study week, hospital location, and ICU care level) and excluded patients receiving only comfort care, they found that TME was associated with a 24% increased risk of in-hospital death (30% in patients with TME vs. 16% in those without TME).
The highest mortality risk was associated with hypoxemia, with 42% of patients with HIE dying during hospitalization, compared with 16% of patients without HIE (adjusted hazard ratio 1.56; 95% confidence interval, 1.21-2.00; P = .001).
“Not all patients who are intubated require sedation, but there’s generally a lot of hesitation in reducing or stopping sedation in some patients,” Dr. Frontera observed.
She acknowledged there are “many extremely sick patients whom you can’t ventilate without sedation.”
Nevertheless, “delirium in and of itself does not cause death. It’s a symptom, not a disease, and we have to figure out what causes it. Delirium might not need to be sedated, and it’s more important to see what the causal problem is.”
Independent predictor of death
Commenting on the study, Panayiotis N. Varelas, MD, PhD, vice president of the Neurocritical Care Society, said the study “approached the TME issue better than previously, namely allowing time for sedatives to wear off to have a better sample of patients with this syndrome.”
Dr. Varelas, who is chairman of the department of neurology and professor of neurology at Albany (N.Y.) Medical College, emphasized that TME “is not benign and, in patients with COVID-19, it is an independent predictor of in-hospital mortality.”
“One should take all possible measures … to avoid desaturation and hypotensive episodes and also aggressively treat SAE and uremic encephalopathy in hopes of improving the outcomes,” added Dr. Varelas, who was not involved with the study.
Also commenting on the study, Mitchell Elkind, MD, professor of neurology and epidemiology at Columbia University in New York, who was not associated with the research, said it “nicely distinguishes among the different causes of encephalopathy, including sepsis, hypoxia, and kidney failure … emphasizing just how sick these patients are.”
The study received no direct funding. Individual investigators were supported by grants from the National Institute on Aging and the National Institute of Neurological Disorders and Stroke. The investigators, Dr. Varelas, and Dr. Elkind have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM NEUROCRITICAL CARE
New expert guidance on ketamine for resistant depression
An international panel of mood disorder experts has published guidance on how to safely and effectively use ketamine and esketamine to treat adults with treatment-resistant depression (TRD).
“Ketamine and esketamine are the first rapid-onset treatments for adults with TRD, and there was an international need for best-practice guidance on the deft and safe implementation of ketamine and esketamine at the point of care, as none previously existed,” first author Roger McIntyre, MD, professor of psychiatry and pharmacology, University of Toronto, said in an interview.
“This need has only been amplified by the significant increase in the number of clinics and centers providing this treatment,” added Dr. McIntyre, head of the mood disorders psychopharmacology unit.
Their article was published online March 17 in the American Journal of Psychiatry.
Insufficient evidence of long-term efficacy
As reported by this news organization, the U.S. Food and Drug Administration (FDA) approved esketamine nasal spray (Spravato) for TRD in March 2019.
In August 2020, the FDA updated the approval to include adults with major depression and suicidal thoughts or actions.
To provide clinical guidance, Dr. McIntyre and colleagues synthesized the available literature on the efficacy, safety, and tolerability of ketamine and esketamine for TRD.
The evidence, they note, supports the rapid-onset (within 1-2 days) efficacy of esketamine and ketamine in TRD.
The strongest evidence of efficacy is for intranasal esketamine and intravenous ketamine. There is insufficient evidence for oral, subcutaneous, or intramuscular ketamine for TRD, they report.
Intranasal esketamine demonstrates efficacy, safety, and tolerability for up to 1 year in adults with TRD. Evidence for long-term efficacy, safety, and tolerability of intravenous ketamine for patients with TRD is insufficient, the group notes.
They also note that esketamine is approved in the United States for major depression in association with suicidal ideation or behavior and that it has been proven to reduce suicide completion.
Safety concerns with ketamine and esketamine identified in the literature include, but are not limited to, psychiatric, neurologic/cognitive, genitourinary, and hemodynamic effects.
Implementation checklist
The group has developed an “implementation checklist” for use of ketamine/esketamine in clinical practice.
Starting with patient selection, they note that appropriate patients are those with a confirmed diagnosis of TRD for whom psychosis and other conditions that would significantly affect the risk-benefit ratio have been ruled out.
They suggest that a physical examination and monitoring of vital signs be undertaken during treatment and during posttreatment surveillance. A urine drug screen should be considered if appropriate.
The group advises that esketamine and ketamine be administered only in settings with multidisciplinary personnel, including, but not limited to, those with expertise in the assessment of mood disorders.
Clinics should be equipped with appropriate cardiorespiratory monitoring and be capable of psychiatric assessment of dissociation and psychotomimetic effects.
Depressive symptoms should be measured, and the authors suggest assessing for anxiety, cognitive function, well-being, and psychosocial function.
Patients should be monitored immediately after treatment to ensure cardiorespiratory stability, clear sensorium, and attenuation of dissociative and psychotomimetic effects.
The United States and some other countries require a risk evaluation and mitigation strategy (REMS) when administering esketamine. Regarding the REMS, it is advised that all patients be monitored for a minimum of 2 hours before discharge.
Patients should arrange for reliable transportation for each appointment, and they should be advised not to operate motor vehicles or hazardous machinery without at least one night of sleep.
, only worsened by COVID-19,” said Dr. McIntyre.
“Clinicians of different professional backgrounds have been interested in ketamine/esketamine, and we are extraordinarily pleased to see our international guidelines published,” he added.
‘Extremely useful’
Reached for comment, Alan Schatzberg, MD, professor of psychiatry and behavioral sciences at Stanford (Calif.) University, said this document “puts a lot of information in one place as far as what we know and what we don’t know right now, and that’s helpful. I think it’s an attempt to have a kind of a somewhat objective review of the literature, and it’s in a good journal.”
The article, Dr. Schatzberg added, “could be extremely useful for someone who is considering whether ketamine is useful for a patient or what they can tell a patient about ketamine, that is, about how long they might need, is it going to work, will it continue to work, and the level of data we have either on benefits or side effects.”
The research had no specific funding. The original article contains a complete list of author disclosures. Dr. Schatzberg has received grant support from Janssen; has served as a consultant for Alkermes, Avanir, Brain Resource, Bracket, Compass, Delpor, Epiodyne, GLG, Jazz, Janssen Pharmaceuticals, Lundbeck/Takeda, McKinsey and Company, Merck, Myriad Genetics, Neuronetics, Owl Analytics, Pfizer, Sage, Sunovion, and Xhale; holds equity in Corcept (cofounder), Delpor, Dermira, Epiodyne, Gilead, Incyte Genetics, Intersect ENT, Madrigal, Merck, Owl Analytics, Seattle Genetics, Titan, and Xhale; and is listed as an inventor on patents for pharmacogenetics and antiglucocorticoid use in the prediction of antidepressant response.
A version of this article first appeared on Medscape.com.
An international panel of mood disorder experts has published guidance on how to safely and effectively use ketamine and esketamine to treat adults with treatment-resistant depression (TRD).
“Ketamine and esketamine are the first rapid-onset treatments for adults with TRD, and there was an international need for best-practice guidance on the deft and safe implementation of ketamine and esketamine at the point of care, as none previously existed,” first author Roger McIntyre, MD, professor of psychiatry and pharmacology, University of Toronto, said in an interview.
“This need has only been amplified by the significant increase in the number of clinics and centers providing this treatment,” added Dr. McIntyre, head of the mood disorders psychopharmacology unit.
Their article was published online March 17 in the American Journal of Psychiatry.
Insufficient evidence of long-term efficacy
As reported by this news organization, the U.S. Food and Drug Administration (FDA) approved esketamine nasal spray (Spravato) for TRD in March 2019.
In August 2020, the FDA updated the approval to include adults with major depression and suicidal thoughts or actions.
To provide clinical guidance, Dr. McIntyre and colleagues synthesized the available literature on the efficacy, safety, and tolerability of ketamine and esketamine for TRD.
The evidence, they note, supports the rapid-onset (within 1-2 days) efficacy of esketamine and ketamine in TRD.
The strongest evidence of efficacy is for intranasal esketamine and intravenous ketamine. There is insufficient evidence for oral, subcutaneous, or intramuscular ketamine for TRD, they report.
Intranasal esketamine demonstrates efficacy, safety, and tolerability for up to 1 year in adults with TRD. Evidence for long-term efficacy, safety, and tolerability of intravenous ketamine for patients with TRD is insufficient, the group notes.
They also note that esketamine is approved in the United States for major depression in association with suicidal ideation or behavior and that it has been proven to reduce suicide completion.
Safety concerns with ketamine and esketamine identified in the literature include, but are not limited to, psychiatric, neurologic/cognitive, genitourinary, and hemodynamic effects.
Implementation checklist
The group has developed an “implementation checklist” for use of ketamine/esketamine in clinical practice.
Starting with patient selection, they note that appropriate patients are those with a confirmed diagnosis of TRD for whom psychosis and other conditions that would significantly affect the risk-benefit ratio have been ruled out.
They suggest that a physical examination and monitoring of vital signs be undertaken during treatment and during posttreatment surveillance. A urine drug screen should be considered if appropriate.
The group advises that esketamine and ketamine be administered only in settings with multidisciplinary personnel, including, but not limited to, those with expertise in the assessment of mood disorders.
Clinics should be equipped with appropriate cardiorespiratory monitoring and be capable of psychiatric assessment of dissociation and psychotomimetic effects.
Depressive symptoms should be measured, and the authors suggest assessing for anxiety, cognitive function, well-being, and psychosocial function.
Patients should be monitored immediately after treatment to ensure cardiorespiratory stability, clear sensorium, and attenuation of dissociative and psychotomimetic effects.
The United States and some other countries require a risk evaluation and mitigation strategy (REMS) when administering esketamine. Regarding the REMS, it is advised that all patients be monitored for a minimum of 2 hours before discharge.
Patients should arrange for reliable transportation for each appointment, and they should be advised not to operate motor vehicles or hazardous machinery without at least one night of sleep.
, only worsened by COVID-19,” said Dr. McIntyre.
“Clinicians of different professional backgrounds have been interested in ketamine/esketamine, and we are extraordinarily pleased to see our international guidelines published,” he added.
‘Extremely useful’
Reached for comment, Alan Schatzberg, MD, professor of psychiatry and behavioral sciences at Stanford (Calif.) University, said this document “puts a lot of information in one place as far as what we know and what we don’t know right now, and that’s helpful. I think it’s an attempt to have a kind of a somewhat objective review of the literature, and it’s in a good journal.”
The article, Dr. Schatzberg added, “could be extremely useful for someone who is considering whether ketamine is useful for a patient or what they can tell a patient about ketamine, that is, about how long they might need, is it going to work, will it continue to work, and the level of data we have either on benefits or side effects.”
The research had no specific funding. The original article contains a complete list of author disclosures. Dr. Schatzberg has received grant support from Janssen; has served as a consultant for Alkermes, Avanir, Brain Resource, Bracket, Compass, Delpor, Epiodyne, GLG, Jazz, Janssen Pharmaceuticals, Lundbeck/Takeda, McKinsey and Company, Merck, Myriad Genetics, Neuronetics, Owl Analytics, Pfizer, Sage, Sunovion, and Xhale; holds equity in Corcept (cofounder), Delpor, Dermira, Epiodyne, Gilead, Incyte Genetics, Intersect ENT, Madrigal, Merck, Owl Analytics, Seattle Genetics, Titan, and Xhale; and is listed as an inventor on patents for pharmacogenetics and antiglucocorticoid use in the prediction of antidepressant response.
A version of this article first appeared on Medscape.com.
An international panel of mood disorder experts has published guidance on how to safely and effectively use ketamine and esketamine to treat adults with treatment-resistant depression (TRD).
“Ketamine and esketamine are the first rapid-onset treatments for adults with TRD, and there was an international need for best-practice guidance on the deft and safe implementation of ketamine and esketamine at the point of care, as none previously existed,” first author Roger McIntyre, MD, professor of psychiatry and pharmacology, University of Toronto, said in an interview.
“This need has only been amplified by the significant increase in the number of clinics and centers providing this treatment,” added Dr. McIntyre, head of the mood disorders psychopharmacology unit.
Their article was published online March 17 in the American Journal of Psychiatry.
Insufficient evidence of long-term efficacy
As reported by this news organization, the U.S. Food and Drug Administration (FDA) approved esketamine nasal spray (Spravato) for TRD in March 2019.
In August 2020, the FDA updated the approval to include adults with major depression and suicidal thoughts or actions.
To provide clinical guidance, Dr. McIntyre and colleagues synthesized the available literature on the efficacy, safety, and tolerability of ketamine and esketamine for TRD.
The evidence, they note, supports the rapid-onset (within 1-2 days) efficacy of esketamine and ketamine in TRD.
The strongest evidence of efficacy is for intranasal esketamine and intravenous ketamine. There is insufficient evidence for oral, subcutaneous, or intramuscular ketamine for TRD, they report.
Intranasal esketamine demonstrates efficacy, safety, and tolerability for up to 1 year in adults with TRD. Evidence for long-term efficacy, safety, and tolerability of intravenous ketamine for patients with TRD is insufficient, the group notes.
They also note that esketamine is approved in the United States for major depression in association with suicidal ideation or behavior and that it has been proven to reduce suicide completion.
Safety concerns with ketamine and esketamine identified in the literature include, but are not limited to, psychiatric, neurologic/cognitive, genitourinary, and hemodynamic effects.
Implementation checklist
The group has developed an “implementation checklist” for use of ketamine/esketamine in clinical practice.
Starting with patient selection, they note that appropriate patients are those with a confirmed diagnosis of TRD for whom psychosis and other conditions that would significantly affect the risk-benefit ratio have been ruled out.
They suggest that a physical examination and monitoring of vital signs be undertaken during treatment and during posttreatment surveillance. A urine drug screen should be considered if appropriate.
The group advises that esketamine and ketamine be administered only in settings with multidisciplinary personnel, including, but not limited to, those with expertise in the assessment of mood disorders.
Clinics should be equipped with appropriate cardiorespiratory monitoring and be capable of psychiatric assessment of dissociation and psychotomimetic effects.
Depressive symptoms should be measured, and the authors suggest assessing for anxiety, cognitive function, well-being, and psychosocial function.
Patients should be monitored immediately after treatment to ensure cardiorespiratory stability, clear sensorium, and attenuation of dissociative and psychotomimetic effects.
The United States and some other countries require a risk evaluation and mitigation strategy (REMS) when administering esketamine. Regarding the REMS, it is advised that all patients be monitored for a minimum of 2 hours before discharge.
Patients should arrange for reliable transportation for each appointment, and they should be advised not to operate motor vehicles or hazardous machinery without at least one night of sleep.
, only worsened by COVID-19,” said Dr. McIntyre.
“Clinicians of different professional backgrounds have been interested in ketamine/esketamine, and we are extraordinarily pleased to see our international guidelines published,” he added.
‘Extremely useful’
Reached for comment, Alan Schatzberg, MD, professor of psychiatry and behavioral sciences at Stanford (Calif.) University, said this document “puts a lot of information in one place as far as what we know and what we don’t know right now, and that’s helpful. I think it’s an attempt to have a kind of a somewhat objective review of the literature, and it’s in a good journal.”
The article, Dr. Schatzberg added, “could be extremely useful for someone who is considering whether ketamine is useful for a patient or what they can tell a patient about ketamine, that is, about how long they might need, is it going to work, will it continue to work, and the level of data we have either on benefits or side effects.”
The research had no specific funding. The original article contains a complete list of author disclosures. Dr. Schatzberg has received grant support from Janssen; has served as a consultant for Alkermes, Avanir, Brain Resource, Bracket, Compass, Delpor, Epiodyne, GLG, Jazz, Janssen Pharmaceuticals, Lundbeck/Takeda, McKinsey and Company, Merck, Myriad Genetics, Neuronetics, Owl Analytics, Pfizer, Sage, Sunovion, and Xhale; holds equity in Corcept (cofounder), Delpor, Dermira, Epiodyne, Gilead, Incyte Genetics, Intersect ENT, Madrigal, Merck, Owl Analytics, Seattle Genetics, Titan, and Xhale; and is listed as an inventor on patents for pharmacogenetics and antiglucocorticoid use in the prediction of antidepressant response.
A version of this article first appeared on Medscape.com.
A paleolithic raw bar, and the human brush with extinction
This essay is adapted from the newly released book, “A History of the Human Brain: From the Sea Sponge to CRISPR, How Our Brain Evolved.”
“He was a bold man that first ate an oyster.” – Jonathan Swift
That man or, just as likely, that woman, may have done so out of necessity. It was either eat this glistening, gray blob of briny goo or perish.
Beginning 190,000 years ago, a glacial age we identify today as Marine Isotope Stage 6, or MIS6, had set in, cooling and drying out much of the planet. There was widespread drought, leaving the African plains a harsher, more barren substrate for survival – an arena of competition, desperation, and starvation for many species, including ours. Some estimates have the sapiens population dipping to just a few hundred people during MIS6. Like other apes today, we were an endangered species. But through some nexus of intelligence, ecological exploitation, and luck, we managed. Anthropologists argue over what part of Africa would’ve been hospitable enough to rescue sapiens from Darwinian oblivion. Arizona State University archaeologist Curtis Marean, PhD, believes the continent’s southern shore is a good candidate.
For 2 decades, Dr. Marean has overseen excavations at a site called Pinnacle Point on the South African coast. The region has over 9,000 plant species, including the world’s most diverse population of geophytes, plants with underground energy-storage organs such as bulbs, tubers, and rhizomes. These subterranean stores are rich in calories and carbohydrates, and, by virtue of being buried, are protected from most other species (save the occasional tool-wielding chimpanzee). They are also adapted to cold climates and, when cooked, easily digested. All in all, a coup for hunter-gatherers.
The other enticement at Pinnacle Point could be found with a few easy steps toward the sea. Mollusks. Geological samples from MIS6 show South Africa’s shores were packed with mussels, oysters, clams, and a variety of sea snails. We almost certainly turned to them for nutrition.
Dr. Marean’s research suggests that, sometime around 160,000 years ago, at least one group of sapiens began supplementing their terrestrial diet by exploiting the region’s rich shellfish beds. This is the oldest evidence to date of humans consistently feasting on seafood – easy, predictable, immobile calories. No hunting required. As inland Africa dried up, learning to shuck mussels and oysters was a key adaptation to coastal living, one that supported our later migration out of the continent.
Dr. Marean believes the change in behavior was possible thanks to our already keen brains, which supported an ability to track tides, especially spring tides. Spring tides occur twice a month with each new and full moon and result in the greatest difference between high and low tidewaters. The people of Pinnacle Point learned to exploit this cycle. “By tracking tides, we would have had easy, reliable access to high-quality proteins and fats from shellfish every 2 weeks as the ocean receded,” he says. “Whereas you can’t rely on land animals to always be in the same place at the same time.” Work by Jan De Vynck, PhD, a professor at Nelson Mandela University in South Africa, supports this idea, showing that foraging shellfish beds under optimal tidal conditions can yield a staggering 3,500 calories per hour!
“I don’t know if we owe our existence to seafood, but it was certainly important for the population [that Dr.] Curtis studies. That place is full of mussels,” said Ian Tattersall, PhD, curator emeritus with the American Museum of Natural History in New York.
“And I like the idea that during a population bottleneck we got creative and learned how to focus on marine resources.” Innovations, Dr. Tattersall explained, typically occur in small, fixed populations. Large populations have too much genetic inertia to support radical innovation; the status quo is enough to survive. “If you’re looking for evolutionary innovation, you have to look at smaller groups.”
MIS6 wasn’t the only near-extinction in our past. During the Pleistocene epoch, roughly 2.5 million to 12,000 years ago, humans tended to maintain a small population, hovering around a million and later growing to maybe 8 million at most. Periodically, our numbers dipped as climate shifts, natural disasters, and food shortages brought us dangerously close to extinction. Modern humans are descended from the hearty survivors of these bottlenecks.
One especially dire stretch occurred around 1 million years ago. Our effective population (the number of breeding individuals) shriveled to around 18,000, smaller than that of other apes at the time. Worse, our genetic diversity – the insurance policy on evolutionary success and the ability to adapt – plummeted. A similar near extinction may have occurred around 75,000 years ago, the result of a massive volcanic eruption in Sumatra.
Our smarts and adaptability helped us endure these tough times – omnivorism helped us weather scarcity.
A sea of vitamins
Both Dr. Marean and Dr. Tattersall agree that the sapiens hanging on in southern Africa couldn’t have lived entirely on shellfish.
Most likely they also spent time hunting and foraging roots inland, making pilgrimages to the sea during spring tides. Dr. Marean believes coastal cuisine may have allowed a paltry human population to hang on until climate change led to more hospitable terrain. He’s not entirely sold on the idea that marine life was necessarily a driver of human brain evolution.
By the time we incorporated seafood into our diets we were already smart, our brains shaped through millennia of selection for intelligence. “Being a marine forager requires a certain degree of sophisticated smarts,” he said. It requires tracking the lunar cycle and planning excursions to the coast at the right times. Shellfish were simply another source of calories.
Unless you ask Michael Crawford.
Dr. Crawford is a professor at Imperial College London and a strident believer that our brains are those of sea creatures. Sort of.
In 1972, he copublished a paper concluding that the brain is structurally and functionally dependent on an omega-3 fatty acid called docosahexaenoic acid, or DHA. The human brain is composed of nearly 60% fat, so it’s not surprising that certain fats are important to brain health. Nearly 50 years after Dr. Crawford’s study, omega-3 supplements are now a multi-billion-dollar business.
Omega-3s, or more formally, omega-3 polyunsaturated fatty acids (PUFAs), are essential fats, meaning they aren’t produced by the body and must be obtained through diet. We get them from vegetable oils, nuts, seeds, and animals that eat such things. But take an informal poll, and you’ll find most people probably associate omega fatty acids with fish and other seafood.
In the 1970s and 1980s, scientists took notice of the low rates of heart disease in Eskimo communities. Research linked their cardiovascular health to a high-fish diet (though fish cannot produce omega-3s, they source them from algae), and eventually the medical and scientific communities began to rethink fat. Study after study found omega-3 fatty acids to be healthy. They were linked with a lower risk for heart disease and overall mortality. All those decades of parents forcing various fish oils on their grimacing children now had some science behind them. There is such a thing as a good fat.
, especially DHA and eicosapentaenoic acid, or EPA. Omega fats provide structure to neuronal cell membranes and are crucial in neuron-to-neuron communication. They increase levels of a protein called brain-derived neurotrophic factor (BDNF), which supports neuronal growth and survival. A growing body of evidence shows omega-3 supplementation may slow down the process of neurodegeneration, the gradual deterioration of the brain that results in Alzheimer’s disease and other forms of dementia.
Popping a daily omega-3 supplement or, better still, eating a seafood-rich diet, may increase blood flow to the brain. In 2019, the International Society for Nutritional Psychiatry Research recommended omega-3s as an adjunct therapy for major depressive disorder. PUFAs appear to reduce the risk for and severity of mood disorders such as depression and to boost attention in children with ADHD as effectively as drug therapies.
Many researchers claim there would’ve been plenty of DHA available on land to support early humans, and marine foods were just one of many sources.
Not Dr. Crawford.
He believes that brain development and function are not only dependent on DHA but, in fact, DHA sourced from the sea was critical to mammalian brain evolution. “The animal brain evolved 600 million years ago in the ocean and was dependent on DHA, as well as compounds such as iodine, which is also in short supply on land,” he said. “To build a brain, you need these building blocks, which were rich at sea and on rocky shores.”
Dr. Crawford cites his early biochemical work showing DHA isn’t readily accessible from the muscle tissue of land animals. Using DHA tagged with a radioactive isotope, he and his colleagues in the 1970s found that “ready-made” DHA, like that found in shellfish, is incorporated into the developing rat brain with 10-fold greater efficiency than plant- and land animal–sourced DHA, where it exists as its metabolic precursor alpha-linolenic acid. “I’m afraid the idea that ample DHA was available from the fats of animals on the savanna is just not true,” he disputes. According to Dr. Crawford, our tiny, wormlike ancestors were able to evolve primitive nervous systems and flit through the silt thanks to the abundance of healthy fat to be had by living in the ocean and consuming algae.
For over 40 years, Dr. Crawford has argued that rising rates of mental illness are a result of post–World War II dietary changes, especially the move toward land-sourced food and the medical community’s subsequent support of low-fat diets. He feels that omega-3s from seafood were critical to humans’ rapid neural march toward higher cognition, and are therefore critical to brain health. “The continued rise in mental illness is an incredibly important threat to mankind and society, and moving away from marine foods is a major contributor,” said Dr. Crawford.
University of Sherbrooke (Que.) physiology professor Stephen Cunnane, PhD, tends to agree that aquatically sourced nutrients were crucial to human evolution. It’s the importance of coastal living he’s not sure about. He believes hominins would’ve incorporated fish from lakes and rivers into their diet for millions of years. In his view, it wasn’t just omega-3s that contributed to our big brains, but a cluster of nutrients found in fish: iodine, iron, zinc, copper, and selenium among them. “I think DHA was hugely important to our evolution and brain health but I don’t think it was a magic bullet all by itself,” he said. “Numerous other nutrients found in fish and shellfish were very probably important, too, and are now known to be good for the brain.”
Dr. Marean agrees. “Accessing the marine food chain could have had a huge impact on fertility, survival, and overall health, including brain health, in part, due to the high return on omega-3 fatty acids and other nutrients.” But, he speculates, before MIS6, hominins would have had access to plenty of brain-healthy terrestrial nutrition, including meat from animals that consumed omega-3–rich plants and grains.
Dr. Cunnane agrees with Dr. Marean to a degree. He’s confident that higher intelligence evolved gradually over millions of years as mutations inching the cognitive needle forward conferred survival and reproductive advantages – but he maintains that certain advantages like, say, being able to shuck an oyster, allowed an already intelligent brain to thrive.
Foraging marine life in the waters off of Africa likely played an important role in keeping some of our ancestors alive and supported our subsequent propagation throughout the world. By this point, the human brain was already a marvel of consciousness and computing, not too dissimilar to the one we carry around today.
In all likelihood, Pleistocene humans probably got their nutrients and calories wherever they could. If we lived inland, we hunted. Maybe we speared the occasional catfish. We sourced nutrients from fruits, leaves, and nuts. A few times a month, those of us near the coast enjoyed a feast of mussels and oysters.
Dr. Stetka is an editorial director at Medscape.com, a former neuroscience researcher, and a nonpracticing physician. A version of this article first appeared on Medscape.
This essay is adapted from the newly released book, “A History of the Human Brain: From the Sea Sponge to CRISPR, How Our Brain Evolved.”
“He was a bold man that first ate an oyster.” – Jonathan Swift
That man or, just as likely, that woman, may have done so out of necessity. It was either eat this glistening, gray blob of briny goo or perish.
Beginning 190,000 years ago, a glacial age we identify today as Marine Isotope Stage 6, or MIS6, had set in, cooling and drying out much of the planet. There was widespread drought, leaving the African plains a harsher, more barren substrate for survival – an arena of competition, desperation, and starvation for many species, including ours. Some estimates have the sapiens population dipping to just a few hundred people during MIS6. Like other apes today, we were an endangered species. But through some nexus of intelligence, ecological exploitation, and luck, we managed. Anthropologists argue over what part of Africa would’ve been hospitable enough to rescue sapiens from Darwinian oblivion. Arizona State University archaeologist Curtis Marean, PhD, believes the continent’s southern shore is a good candidate.
For 2 decades, Dr. Marean has overseen excavations at a site called Pinnacle Point on the South African coast. The region has over 9,000 plant species, including the world’s most diverse population of geophytes, plants with underground energy-storage organs such as bulbs, tubers, and rhizomes. These subterranean stores are rich in calories and carbohydrates, and, by virtue of being buried, are protected from most other species (save the occasional tool-wielding chimpanzee). They are also adapted to cold climates and, when cooked, easily digested. All in all, a coup for hunter-gatherers.
The other enticement at Pinnacle Point could be found with a few easy steps toward the sea. Mollusks. Geological samples from MIS6 show South Africa’s shores were packed with mussels, oysters, clams, and a variety of sea snails. We almost certainly turned to them for nutrition.
Dr. Marean’s research suggests that, sometime around 160,000 years ago, at least one group of sapiens began supplementing their terrestrial diet by exploiting the region’s rich shellfish beds. This is the oldest evidence to date of humans consistently feasting on seafood – easy, predictable, immobile calories. No hunting required. As inland Africa dried up, learning to shuck mussels and oysters was a key adaptation to coastal living, one that supported our later migration out of the continent.
Dr. Marean believes the change in behavior was possible thanks to our already keen brains, which supported an ability to track tides, especially spring tides. Spring tides occur twice a month with each new and full moon and result in the greatest difference between high and low tidewaters. The people of Pinnacle Point learned to exploit this cycle. “By tracking tides, we would have had easy, reliable access to high-quality proteins and fats from shellfish every 2 weeks as the ocean receded,” he says. “Whereas you can’t rely on land animals to always be in the same place at the same time.” Work by Jan De Vynck, PhD, a professor at Nelson Mandela University in South Africa, supports this idea, showing that foraging shellfish beds under optimal tidal conditions can yield a staggering 3,500 calories per hour!
“I don’t know if we owe our existence to seafood, but it was certainly important for the population [that Dr.] Curtis studies. That place is full of mussels,” said Ian Tattersall, PhD, curator emeritus with the American Museum of Natural History in New York.
“And I like the idea that during a population bottleneck we got creative and learned how to focus on marine resources.” Innovations, Dr. Tattersall explained, typically occur in small, fixed populations. Large populations have too much genetic inertia to support radical innovation; the status quo is enough to survive. “If you’re looking for evolutionary innovation, you have to look at smaller groups.”
MIS6 wasn’t the only near-extinction in our past. During the Pleistocene epoch, roughly 2.5 million to 12,000 years ago, humans tended to maintain a small population, hovering around a million and later growing to maybe 8 million at most. Periodically, our numbers dipped as climate shifts, natural disasters, and food shortages brought us dangerously close to extinction. Modern humans are descended from the hearty survivors of these bottlenecks.
One especially dire stretch occurred around 1 million years ago. Our effective population (the number of breeding individuals) shriveled to around 18,000, smaller than that of other apes at the time. Worse, our genetic diversity – the insurance policy on evolutionary success and the ability to adapt – plummeted. A similar near extinction may have occurred around 75,000 years ago, the result of a massive volcanic eruption in Sumatra.
Our smarts and adaptability helped us endure these tough times – omnivorism helped us weather scarcity.
A sea of vitamins
Both Dr. Marean and Dr. Tattersall agree that the sapiens hanging on in southern Africa couldn’t have lived entirely on shellfish.
Most likely they also spent time hunting and foraging roots inland, making pilgrimages to the sea during spring tides. Dr. Marean believes coastal cuisine may have allowed a paltry human population to hang on until climate change led to more hospitable terrain. He’s not entirely sold on the idea that marine life was necessarily a driver of human brain evolution.
By the time we incorporated seafood into our diets we were already smart, our brains shaped through millennia of selection for intelligence. “Being a marine forager requires a certain degree of sophisticated smarts,” he said. It requires tracking the lunar cycle and planning excursions to the coast at the right times. Shellfish were simply another source of calories.
Unless you ask Michael Crawford.
Dr. Crawford is a professor at Imperial College London and a strident believer that our brains are those of sea creatures. Sort of.
In 1972, he copublished a paper concluding that the brain is structurally and functionally dependent on an omega-3 fatty acid called docosahexaenoic acid, or DHA. The human brain is composed of nearly 60% fat, so it’s not surprising that certain fats are important to brain health. Nearly 50 years after Dr. Crawford’s study, omega-3 supplements are now a multi-billion-dollar business.
Omega-3s, or more formally, omega-3 polyunsaturated fatty acids (PUFAs), are essential fats, meaning they aren’t produced by the body and must be obtained through diet. We get them from vegetable oils, nuts, seeds, and animals that eat such things. But take an informal poll, and you’ll find most people probably associate omega fatty acids with fish and other seafood.
In the 1970s and 1980s, scientists took notice of the low rates of heart disease in Eskimo communities. Research linked their cardiovascular health to a high-fish diet (though fish cannot produce omega-3s, they source them from algae), and eventually the medical and scientific communities began to rethink fat. Study after study found omega-3 fatty acids to be healthy. They were linked with a lower risk for heart disease and overall mortality. All those decades of parents forcing various fish oils on their grimacing children now had some science behind them. There is such a thing as a good fat.
, especially DHA and eicosapentaenoic acid, or EPA. Omega fats provide structure to neuronal cell membranes and are crucial in neuron-to-neuron communication. They increase levels of a protein called brain-derived neurotrophic factor (BDNF), which supports neuronal growth and survival. A growing body of evidence shows omega-3 supplementation may slow down the process of neurodegeneration, the gradual deterioration of the brain that results in Alzheimer’s disease and other forms of dementia.
Popping a daily omega-3 supplement or, better still, eating a seafood-rich diet, may increase blood flow to the brain. In 2019, the International Society for Nutritional Psychiatry Research recommended omega-3s as an adjunct therapy for major depressive disorder. PUFAs appear to reduce the risk for and severity of mood disorders such as depression and to boost attention in children with ADHD as effectively as drug therapies.
Many researchers claim there would’ve been plenty of DHA available on land to support early humans, and marine foods were just one of many sources.
Not Dr. Crawford.
He believes that brain development and function are not only dependent on DHA but, in fact, DHA sourced from the sea was critical to mammalian brain evolution. “The animal brain evolved 600 million years ago in the ocean and was dependent on DHA, as well as compounds such as iodine, which is also in short supply on land,” he said. “To build a brain, you need these building blocks, which were rich at sea and on rocky shores.”
Dr. Crawford cites his early biochemical work showing DHA isn’t readily accessible from the muscle tissue of land animals. Using DHA tagged with a radioactive isotope, he and his colleagues in the 1970s found that “ready-made” DHA, like that found in shellfish, is incorporated into the developing rat brain with 10-fold greater efficiency than plant- and land animal–sourced DHA, where it exists as its metabolic precursor alpha-linolenic acid. “I’m afraid the idea that ample DHA was available from the fats of animals on the savanna is just not true,” he disputes. According to Dr. Crawford, our tiny, wormlike ancestors were able to evolve primitive nervous systems and flit through the silt thanks to the abundance of healthy fat to be had by living in the ocean and consuming algae.
For over 40 years, Dr. Crawford has argued that rising rates of mental illness are a result of post–World War II dietary changes, especially the move toward land-sourced food and the medical community’s subsequent support of low-fat diets. He feels that omega-3s from seafood were critical to humans’ rapid neural march toward higher cognition, and are therefore critical to brain health. “The continued rise in mental illness is an incredibly important threat to mankind and society, and moving away from marine foods is a major contributor,” said Dr. Crawford.
University of Sherbrooke (Que.) physiology professor Stephen Cunnane, PhD, tends to agree that aquatically sourced nutrients were crucial to human evolution. It’s the importance of coastal living he’s not sure about. He believes hominins would’ve incorporated fish from lakes and rivers into their diet for millions of years. In his view, it wasn’t just omega-3s that contributed to our big brains, but a cluster of nutrients found in fish: iodine, iron, zinc, copper, and selenium among them. “I think DHA was hugely important to our evolution and brain health but I don’t think it was a magic bullet all by itself,” he said. “Numerous other nutrients found in fish and shellfish were very probably important, too, and are now known to be good for the brain.”
Dr. Marean agrees. “Accessing the marine food chain could have had a huge impact on fertility, survival, and overall health, including brain health, in part, due to the high return on omega-3 fatty acids and other nutrients.” But, he speculates, before MIS6, hominins would have had access to plenty of brain-healthy terrestrial nutrition, including meat from animals that consumed omega-3–rich plants and grains.
Dr. Cunnane agrees with Dr. Marean to a degree. He’s confident that higher intelligence evolved gradually over millions of years as mutations inching the cognitive needle forward conferred survival and reproductive advantages – but he maintains that certain advantages like, say, being able to shuck an oyster, allowed an already intelligent brain to thrive.
Foraging marine life in the waters off of Africa likely played an important role in keeping some of our ancestors alive and supported our subsequent propagation throughout the world. By this point, the human brain was already a marvel of consciousness and computing, not too dissimilar to the one we carry around today.
In all likelihood, Pleistocene humans probably got their nutrients and calories wherever they could. If we lived inland, we hunted. Maybe we speared the occasional catfish. We sourced nutrients from fruits, leaves, and nuts. A few times a month, those of us near the coast enjoyed a feast of mussels and oysters.
Dr. Stetka is an editorial director at Medscape.com, a former neuroscience researcher, and a nonpracticing physician. A version of this article first appeared on Medscape.
This essay is adapted from the newly released book, “A History of the Human Brain: From the Sea Sponge to CRISPR, How Our Brain Evolved.”
“He was a bold man that first ate an oyster.” – Jonathan Swift
That man or, just as likely, that woman, may have done so out of necessity. It was either eat this glistening, gray blob of briny goo or perish.
Beginning 190,000 years ago, a glacial age we identify today as Marine Isotope Stage 6, or MIS6, had set in, cooling and drying out much of the planet. There was widespread drought, leaving the African plains a harsher, more barren substrate for survival – an arena of competition, desperation, and starvation for many species, including ours. Some estimates have the sapiens population dipping to just a few hundred people during MIS6. Like other apes today, we were an endangered species. But through some nexus of intelligence, ecological exploitation, and luck, we managed. Anthropologists argue over what part of Africa would’ve been hospitable enough to rescue sapiens from Darwinian oblivion. Arizona State University archaeologist Curtis Marean, PhD, believes the continent’s southern shore is a good candidate.
For 2 decades, Dr. Marean has overseen excavations at a site called Pinnacle Point on the South African coast. The region has over 9,000 plant species, including the world’s most diverse population of geophytes, plants with underground energy-storage organs such as bulbs, tubers, and rhizomes. These subterranean stores are rich in calories and carbohydrates, and, by virtue of being buried, are protected from most other species (save the occasional tool-wielding chimpanzee). They are also adapted to cold climates and, when cooked, easily digested. All in all, a coup for hunter-gatherers.
The other enticement at Pinnacle Point could be found with a few easy steps toward the sea. Mollusks. Geological samples from MIS6 show South Africa’s shores were packed with mussels, oysters, clams, and a variety of sea snails. We almost certainly turned to them for nutrition.
Dr. Marean’s research suggests that, sometime around 160,000 years ago, at least one group of sapiens began supplementing their terrestrial diet by exploiting the region’s rich shellfish beds. This is the oldest evidence to date of humans consistently feasting on seafood – easy, predictable, immobile calories. No hunting required. As inland Africa dried up, learning to shuck mussels and oysters was a key adaptation to coastal living, one that supported our later migration out of the continent.
Dr. Marean believes the change in behavior was possible thanks to our already keen brains, which supported an ability to track tides, especially spring tides. Spring tides occur twice a month with each new and full moon and result in the greatest difference between high and low tidewaters. The people of Pinnacle Point learned to exploit this cycle. “By tracking tides, we would have had easy, reliable access to high-quality proteins and fats from shellfish every 2 weeks as the ocean receded,” he says. “Whereas you can’t rely on land animals to always be in the same place at the same time.” Work by Jan De Vynck, PhD, a professor at Nelson Mandela University in South Africa, supports this idea, showing that foraging shellfish beds under optimal tidal conditions can yield a staggering 3,500 calories per hour!
“I don’t know if we owe our existence to seafood, but it was certainly important for the population [that Dr.] Curtis studies. That place is full of mussels,” said Ian Tattersall, PhD, curator emeritus with the American Museum of Natural History in New York.
“And I like the idea that during a population bottleneck we got creative and learned how to focus on marine resources.” Innovations, Dr. Tattersall explained, typically occur in small, fixed populations. Large populations have too much genetic inertia to support radical innovation; the status quo is enough to survive. “If you’re looking for evolutionary innovation, you have to look at smaller groups.”
MIS6 wasn’t the only near-extinction in our past. During the Pleistocene epoch, roughly 2.5 million to 12,000 years ago, humans tended to maintain a small population, hovering around a million and later growing to maybe 8 million at most. Periodically, our numbers dipped as climate shifts, natural disasters, and food shortages brought us dangerously close to extinction. Modern humans are descended from the hearty survivors of these bottlenecks.
One especially dire stretch occurred around 1 million years ago. Our effective population (the number of breeding individuals) shriveled to around 18,000, smaller than that of other apes at the time. Worse, our genetic diversity – the insurance policy on evolutionary success and the ability to adapt – plummeted. A similar near extinction may have occurred around 75,000 years ago, the result of a massive volcanic eruption in Sumatra.
Our smarts and adaptability helped us endure these tough times – omnivorism helped us weather scarcity.
A sea of vitamins
Both Dr. Marean and Dr. Tattersall agree that the sapiens hanging on in southern Africa couldn’t have lived entirely on shellfish.
Most likely they also spent time hunting and foraging roots inland, making pilgrimages to the sea during spring tides. Dr. Marean believes coastal cuisine may have allowed a paltry human population to hang on until climate change led to more hospitable terrain. He’s not entirely sold on the idea that marine life was necessarily a driver of human brain evolution.
By the time we incorporated seafood into our diets we were already smart, our brains shaped through millennia of selection for intelligence. “Being a marine forager requires a certain degree of sophisticated smarts,” he said. It requires tracking the lunar cycle and planning excursions to the coast at the right times. Shellfish were simply another source of calories.
Unless you ask Michael Crawford.
Dr. Crawford is a professor at Imperial College London and a strident believer that our brains are those of sea creatures. Sort of.
In 1972, he copublished a paper concluding that the brain is structurally and functionally dependent on an omega-3 fatty acid called docosahexaenoic acid, or DHA. The human brain is composed of nearly 60% fat, so it’s not surprising that certain fats are important to brain health. Nearly 50 years after Dr. Crawford’s study, omega-3 supplements are now a multi-billion-dollar business.
Omega-3s, or more formally, omega-3 polyunsaturated fatty acids (PUFAs), are essential fats, meaning they aren’t produced by the body and must be obtained through diet. We get them from vegetable oils, nuts, seeds, and animals that eat such things. But take an informal poll, and you’ll find most people probably associate omega fatty acids with fish and other seafood.
In the 1970s and 1980s, scientists took notice of the low rates of heart disease in Eskimo communities. Research linked their cardiovascular health to a high-fish diet (though fish cannot produce omega-3s, they source them from algae), and eventually the medical and scientific communities began to rethink fat. Study after study found omega-3 fatty acids to be healthy. They were linked with a lower risk for heart disease and overall mortality. All those decades of parents forcing various fish oils on their grimacing children now had some science behind them. There is such a thing as a good fat.
, especially DHA and eicosapentaenoic acid, or EPA. Omega fats provide structure to neuronal cell membranes and are crucial in neuron-to-neuron communication. They increase levels of a protein called brain-derived neurotrophic factor (BDNF), which supports neuronal growth and survival. A growing body of evidence shows omega-3 supplementation may slow down the process of neurodegeneration, the gradual deterioration of the brain that results in Alzheimer’s disease and other forms of dementia.
Popping a daily omega-3 supplement or, better still, eating a seafood-rich diet, may increase blood flow to the brain. In 2019, the International Society for Nutritional Psychiatry Research recommended omega-3s as an adjunct therapy for major depressive disorder. PUFAs appear to reduce the risk for and severity of mood disorders such as depression and to boost attention in children with ADHD as effectively as drug therapies.
Many researchers claim there would’ve been plenty of DHA available on land to support early humans, and marine foods were just one of many sources.
Not Dr. Crawford.
He believes that brain development and function are not only dependent on DHA but, in fact, DHA sourced from the sea was critical to mammalian brain evolution. “The animal brain evolved 600 million years ago in the ocean and was dependent on DHA, as well as compounds such as iodine, which is also in short supply on land,” he said. “To build a brain, you need these building blocks, which were rich at sea and on rocky shores.”
Dr. Crawford cites his early biochemical work showing DHA isn’t readily accessible from the muscle tissue of land animals. Using DHA tagged with a radioactive isotope, he and his colleagues in the 1970s found that “ready-made” DHA, like that found in shellfish, is incorporated into the developing rat brain with 10-fold greater efficiency than plant- and land animal–sourced DHA, where it exists as its metabolic precursor alpha-linolenic acid. “I’m afraid the idea that ample DHA was available from the fats of animals on the savanna is just not true,” he disputes. According to Dr. Crawford, our tiny, wormlike ancestors were able to evolve primitive nervous systems and flit through the silt thanks to the abundance of healthy fat to be had by living in the ocean and consuming algae.
For over 40 years, Dr. Crawford has argued that rising rates of mental illness are a result of post–World War II dietary changes, especially the move toward land-sourced food and the medical community’s subsequent support of low-fat diets. He feels that omega-3s from seafood were critical to humans’ rapid neural march toward higher cognition, and are therefore critical to brain health. “The continued rise in mental illness is an incredibly important threat to mankind and society, and moving away from marine foods is a major contributor,” said Dr. Crawford.
University of Sherbrooke (Que.) physiology professor Stephen Cunnane, PhD, tends to agree that aquatically sourced nutrients were crucial to human evolution. It’s the importance of coastal living he’s not sure about. He believes hominins would’ve incorporated fish from lakes and rivers into their diet for millions of years. In his view, it wasn’t just omega-3s that contributed to our big brains, but a cluster of nutrients found in fish: iodine, iron, zinc, copper, and selenium among them. “I think DHA was hugely important to our evolution and brain health but I don’t think it was a magic bullet all by itself,” he said. “Numerous other nutrients found in fish and shellfish were very probably important, too, and are now known to be good for the brain.”
Dr. Marean agrees. “Accessing the marine food chain could have had a huge impact on fertility, survival, and overall health, including brain health, in part, due to the high return on omega-3 fatty acids and other nutrients.” But, he speculates, before MIS6, hominins would have had access to plenty of brain-healthy terrestrial nutrition, including meat from animals that consumed omega-3–rich plants and grains.
Dr. Cunnane agrees with Dr. Marean to a degree. He’s confident that higher intelligence evolved gradually over millions of years as mutations inching the cognitive needle forward conferred survival and reproductive advantages – but he maintains that certain advantages like, say, being able to shuck an oyster, allowed an already intelligent brain to thrive.
Foraging marine life in the waters off of Africa likely played an important role in keeping some of our ancestors alive and supported our subsequent propagation throughout the world. By this point, the human brain was already a marvel of consciousness and computing, not too dissimilar to the one we carry around today.
In all likelihood, Pleistocene humans probably got their nutrients and calories wherever they could. If we lived inland, we hunted. Maybe we speared the occasional catfish. We sourced nutrients from fruits, leaves, and nuts. A few times a month, those of us near the coast enjoyed a feast of mussels and oysters.
Dr. Stetka is an editorial director at Medscape.com, a former neuroscience researcher, and a nonpracticing physician. A version of this article first appeared on Medscape.
In U.S., lockdowns added 2 pounds per month
Americans gained nearly 2 pounds per month under COVID-19 shelter-in-place orders in 2020, according to a new study published March 22, 2021, in JAMA Network Open.
Those who kept the same lockdown habits could have gained 20 pounds during the past year, the study authors said.
“We know that weight gain is a public health problem in the U.S. already, so anything making it worse is definitely concerning, and shelter-in-place orders are so ubiquitous that the sheer number of people affected by this makes it extremely relevant,” Gregory Marcus, MD, the senior author and a cardiologist at the University of California, San Francisco, told the New York Times.
Dr. Marcus and colleagues analyzed more than 7,000 weight measurements from 269 people in 37 states who used Bluetooth-connected scales from Feb. 1 to June 1, 2020. Among the participants, about 52% were women, 77% were White, and they had an average age of 52 years.
The research team found that participants had a steady weight gain of more than half a pound every 10 days. That equals about 1.5-2 pounds per month.
Many of the participants were losing weight before the shelter-in-place orders went into effect, Dr. Marcus said. The lockdown effects could be even greater for those who weren’t losing weight before.
“It’s reasonable to assume these individuals are more engaged with their health in general, and more disciplined and on top of things,” he said. “That suggests we could be underestimating – that this is the tip of the iceberg.”
The small study doesn’t represent all of the nation and can’t be generalized to the U.S. population, the study authors noted, but it’s an indicator of what happened during the pandemic. The participants’ weight increased regardless of their location and chronic medical conditions.
Overall, people don’t move around as much during lockdowns, the UCSF researchers reported in another study published in Annals of Internal Medicine in November 2020. According to smartphone data, daily step counts decreased by 27% in March 2020. The step counts increased again throughout the summer but still remained lower than before the COVID-19 pandemic.
“The detrimental health outcomes suggested by these data demonstrate a need to identify concurrent strategies to mitigate weight gain,” the authors wrote in the JAMA Network Open study, “such as encouraging healthy diets and exploring ways to enhance physical activity, as local governments consider new constraints in response to SARS-CoV-2 and potential future pandemics.”
A version of this article first appeared on WebMD.com.
Americans gained nearly 2 pounds per month under COVID-19 shelter-in-place orders in 2020, according to a new study published March 22, 2021, in JAMA Network Open.
Those who kept the same lockdown habits could have gained 20 pounds during the past year, the study authors said.
“We know that weight gain is a public health problem in the U.S. already, so anything making it worse is definitely concerning, and shelter-in-place orders are so ubiquitous that the sheer number of people affected by this makes it extremely relevant,” Gregory Marcus, MD, the senior author and a cardiologist at the University of California, San Francisco, told the New York Times.
Dr. Marcus and colleagues analyzed more than 7,000 weight measurements from 269 people in 37 states who used Bluetooth-connected scales from Feb. 1 to June 1, 2020. Among the participants, about 52% were women, 77% were White, and they had an average age of 52 years.
The research team found that participants had a steady weight gain of more than half a pound every 10 days. That equals about 1.5-2 pounds per month.
Many of the participants were losing weight before the shelter-in-place orders went into effect, Dr. Marcus said. The lockdown effects could be even greater for those who weren’t losing weight before.
“It’s reasonable to assume these individuals are more engaged with their health in general, and more disciplined and on top of things,” he said. “That suggests we could be underestimating – that this is the tip of the iceberg.”
The small study doesn’t represent all of the nation and can’t be generalized to the U.S. population, the study authors noted, but it’s an indicator of what happened during the pandemic. The participants’ weight increased regardless of their location and chronic medical conditions.
Overall, people don’t move around as much during lockdowns, the UCSF researchers reported in another study published in Annals of Internal Medicine in November 2020. According to smartphone data, daily step counts decreased by 27% in March 2020. The step counts increased again throughout the summer but still remained lower than before the COVID-19 pandemic.
“The detrimental health outcomes suggested by these data demonstrate a need to identify concurrent strategies to mitigate weight gain,” the authors wrote in the JAMA Network Open study, “such as encouraging healthy diets and exploring ways to enhance physical activity, as local governments consider new constraints in response to SARS-CoV-2 and potential future pandemics.”
A version of this article first appeared on WebMD.com.
Americans gained nearly 2 pounds per month under COVID-19 shelter-in-place orders in 2020, according to a new study published March 22, 2021, in JAMA Network Open.
Those who kept the same lockdown habits could have gained 20 pounds during the past year, the study authors said.
“We know that weight gain is a public health problem in the U.S. already, so anything making it worse is definitely concerning, and shelter-in-place orders are so ubiquitous that the sheer number of people affected by this makes it extremely relevant,” Gregory Marcus, MD, the senior author and a cardiologist at the University of California, San Francisco, told the New York Times.
Dr. Marcus and colleagues analyzed more than 7,000 weight measurements from 269 people in 37 states who used Bluetooth-connected scales from Feb. 1 to June 1, 2020. Among the participants, about 52% were women, 77% were White, and they had an average age of 52 years.
The research team found that participants had a steady weight gain of more than half a pound every 10 days. That equals about 1.5-2 pounds per month.
Many of the participants were losing weight before the shelter-in-place orders went into effect, Dr. Marcus said. The lockdown effects could be even greater for those who weren’t losing weight before.
“It’s reasonable to assume these individuals are more engaged with their health in general, and more disciplined and on top of things,” he said. “That suggests we could be underestimating – that this is the tip of the iceberg.”
The small study doesn’t represent all of the nation and can’t be generalized to the U.S. population, the study authors noted, but it’s an indicator of what happened during the pandemic. The participants’ weight increased regardless of their location and chronic medical conditions.
Overall, people don’t move around as much during lockdowns, the UCSF researchers reported in another study published in Annals of Internal Medicine in November 2020. According to smartphone data, daily step counts decreased by 27% in March 2020. The step counts increased again throughout the summer but still remained lower than before the COVID-19 pandemic.
“The detrimental health outcomes suggested by these data demonstrate a need to identify concurrent strategies to mitigate weight gain,” the authors wrote in the JAMA Network Open study, “such as encouraging healthy diets and exploring ways to enhance physical activity, as local governments consider new constraints in response to SARS-CoV-2 and potential future pandemics.”
A version of this article first appeared on WebMD.com.
Vitamin D may protect against COVID-19, especially in Black patients
Higher levels of vitamin D than traditionally considered sufficient may help prevent COVID-19 infection – particularly in Black patients, shows a new single-center, retrospective study looking at the role of vitamin D in prevention of infection.
The study, published recently in JAMA Network Open, noted that expert opinion varies as to what “sufficient” levels of vitamin D are, some define this as 30 ng/mL, while others cite 40 ng/mL or greater.
In their discussion, the authors also noted that their results showed the “risk of positive COVID-19 test results decreased significantly with increased vitamin D level of 30 ng/mL or greater when measured as a continuous variable.”
“These new results tell us that having vitamin D levels above those normally considered sufficient is associated with decreased risk of testing positive for COVID-19, at least in Black individuals,” lead author, David Meltzer, MD, chief of hospital medicine at the University of Chicago, said in a press release from his institution.
“These findings suggest that randomized clinical trials to determine whether increasing vitamin D levels to greater than 30-40 ng/mL affect COVID-19 risk are warranted, especially in Black individuals,” he and his coauthors said.
Vitamin D at time of testing most strongly associated with COVID risk
An earlier study by the same researchers found that vitamin D deficiency (less than 20 ng/mL) may raise the risk of testing positive for COVID-19 in people from various ethnicities, as reported by this news organization.
Data for this latest study were drawn from electronic health records for 4,638 individuals at the University of Chicago Medicine and were used to examine whether the likelihood of a positive COVID-19 test was associated with a person’s most recent vitamin D level (within the previous year), and whether there was any effect of ethnicity on this outcome.
Mean age was 52.8 years, 69% were women, 49% were Black, 43% White, and 8% were another race/ethnicity. A total of 27% of the individuals were deficient in vitamin D (less than 20 ng/mL), 27% had insufficient levels (20-30 ng/mL), 22% had sufficient levels (30-40 ng/mL), and the remaining 24% had levels of 40 ng/mL or greater.
In total, 333 (7%) of people tested positive for COVID-19, including 102 (5%) Whites and 211 (9%) Blacks. And 36% of Black individuals who tested positive for COVID-19 were classified as vitamin D deficient, compared with 16% of Whites.
A positive test result for COVID-19 was not significantly associated with vitamin D levels in white individuals but was in Black individuals.
In Black people, compared with levels of at least 40 ng/mL, vitamin D levels of 30-40 ng/mL were associated with an incidence rate ratio of 2.64 for COVID-19 positivity (P = .01). For levels of 20-30 ng/mL, the IRR was 1.69 (P = 0.21); and for less than 20 ng/mL the IRR was 2.55 (P = .009).
The researchers also found that the risk of positive test results with lower vitamin D levels increased when those levels were lower just prior to the positive COVID-19 test, lending “support [to] the idea that vitamin D level at the time of testing is most strongly associated with COVID-19 risk,” they wrote.
Try upping vitamin D levels to 40 ng/mL or greater to prevent COVID?
In their discussion, the authors noted that significant association of vitamin D levels with COVID-19 risk in Blacks but not in Whites, “could reflect their higher COVID-19 risk, to which socioeconomic factors and structural inequities clearly contribute.
“Biological susceptibility to vitamin D deficiency may also be less frequent in White than Black individuals, since lighter skin increases vitamin D production in response to sunlight, and vitamin D binding proteins may vary by race and affect vitamin D bioavailability.”
Given less than 10% of U.S. adults have a vitamin D level greater than 40 ng/mL, the study findings increase the urgency to consider whether increased sun exposure or supplementation could reduce COVID-19 risk, according to the authors.
“When increased sun exposure is impractical, achieving vitamin D levels of 40 ng/mL or greater typically requires greater supplementation than currently recommended for most individuals of 600-800 IU/d vitamin D3,” they added.
However, Dr. Meltzer also acknowledged that “this is an observational study. We can see that there’s an association between vitamin D levels and likelihood of a COVID-19 diagnosis, but we don’t know exactly why that is, or whether these results are due to the vitamin D directly or other related biological factors.”
All in all, the authors suggested that randomized clinical trials are needed to understand if vitamin D can reduce COVID-19 risk, and as such they should include doses of supplements likely to increase vitamin D to at least 40 ng/mL, and perhaps even higher, although they pointed out that the latter must be achieved safely.
“Studies should also consider the role of vitamin D testing, loading doses, dose adjustments for individuals who are obese or overweight, risks for hypercalcemia, and strategies to monitor for and mitigate hypercalcemia, and that non-White populations, such as Black individuals, may have greater needs for supplementation,” they outlined.
They are now recruiting participants for two separate clinical trials testing the efficacy of vitamin D supplements for preventing COVID-19.
The authors disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Higher levels of vitamin D than traditionally considered sufficient may help prevent COVID-19 infection – particularly in Black patients, shows a new single-center, retrospective study looking at the role of vitamin D in prevention of infection.
The study, published recently in JAMA Network Open, noted that expert opinion varies as to what “sufficient” levels of vitamin D are, some define this as 30 ng/mL, while others cite 40 ng/mL or greater.
In their discussion, the authors also noted that their results showed the “risk of positive COVID-19 test results decreased significantly with increased vitamin D level of 30 ng/mL or greater when measured as a continuous variable.”
“These new results tell us that having vitamin D levels above those normally considered sufficient is associated with decreased risk of testing positive for COVID-19, at least in Black individuals,” lead author, David Meltzer, MD, chief of hospital medicine at the University of Chicago, said in a press release from his institution.
“These findings suggest that randomized clinical trials to determine whether increasing vitamin D levels to greater than 30-40 ng/mL affect COVID-19 risk are warranted, especially in Black individuals,” he and his coauthors said.
Vitamin D at time of testing most strongly associated with COVID risk
An earlier study by the same researchers found that vitamin D deficiency (less than 20 ng/mL) may raise the risk of testing positive for COVID-19 in people from various ethnicities, as reported by this news organization.
Data for this latest study were drawn from electronic health records for 4,638 individuals at the University of Chicago Medicine and were used to examine whether the likelihood of a positive COVID-19 test was associated with a person’s most recent vitamin D level (within the previous year), and whether there was any effect of ethnicity on this outcome.
Mean age was 52.8 years, 69% were women, 49% were Black, 43% White, and 8% were another race/ethnicity. A total of 27% of the individuals were deficient in vitamin D (less than 20 ng/mL), 27% had insufficient levels (20-30 ng/mL), 22% had sufficient levels (30-40 ng/mL), and the remaining 24% had levels of 40 ng/mL or greater.
In total, 333 (7%) of people tested positive for COVID-19, including 102 (5%) Whites and 211 (9%) Blacks. And 36% of Black individuals who tested positive for COVID-19 were classified as vitamin D deficient, compared with 16% of Whites.
A positive test result for COVID-19 was not significantly associated with vitamin D levels in white individuals but was in Black individuals.
In Black people, compared with levels of at least 40 ng/mL, vitamin D levels of 30-40 ng/mL were associated with an incidence rate ratio of 2.64 for COVID-19 positivity (P = .01). For levels of 20-30 ng/mL, the IRR was 1.69 (P = 0.21); and for less than 20 ng/mL the IRR was 2.55 (P = .009).
The researchers also found that the risk of positive test results with lower vitamin D levels increased when those levels were lower just prior to the positive COVID-19 test, lending “support [to] the idea that vitamin D level at the time of testing is most strongly associated with COVID-19 risk,” they wrote.
Try upping vitamin D levels to 40 ng/mL or greater to prevent COVID?
In their discussion, the authors noted that significant association of vitamin D levels with COVID-19 risk in Blacks but not in Whites, “could reflect their higher COVID-19 risk, to which socioeconomic factors and structural inequities clearly contribute.
“Biological susceptibility to vitamin D deficiency may also be less frequent in White than Black individuals, since lighter skin increases vitamin D production in response to sunlight, and vitamin D binding proteins may vary by race and affect vitamin D bioavailability.”
Given less than 10% of U.S. adults have a vitamin D level greater than 40 ng/mL, the study findings increase the urgency to consider whether increased sun exposure or supplementation could reduce COVID-19 risk, according to the authors.
“When increased sun exposure is impractical, achieving vitamin D levels of 40 ng/mL or greater typically requires greater supplementation than currently recommended for most individuals of 600-800 IU/d vitamin D3,” they added.
However, Dr. Meltzer also acknowledged that “this is an observational study. We can see that there’s an association between vitamin D levels and likelihood of a COVID-19 diagnosis, but we don’t know exactly why that is, or whether these results are due to the vitamin D directly or other related biological factors.”
All in all, the authors suggested that randomized clinical trials are needed to understand if vitamin D can reduce COVID-19 risk, and as such they should include doses of supplements likely to increase vitamin D to at least 40 ng/mL, and perhaps even higher, although they pointed out that the latter must be achieved safely.
“Studies should also consider the role of vitamin D testing, loading doses, dose adjustments for individuals who are obese or overweight, risks for hypercalcemia, and strategies to monitor for and mitigate hypercalcemia, and that non-White populations, such as Black individuals, may have greater needs for supplementation,” they outlined.
They are now recruiting participants for two separate clinical trials testing the efficacy of vitamin D supplements for preventing COVID-19.
The authors disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Higher levels of vitamin D than traditionally considered sufficient may help prevent COVID-19 infection – particularly in Black patients, shows a new single-center, retrospective study looking at the role of vitamin D in prevention of infection.
The study, published recently in JAMA Network Open, noted that expert opinion varies as to what “sufficient” levels of vitamin D are, some define this as 30 ng/mL, while others cite 40 ng/mL or greater.
In their discussion, the authors also noted that their results showed the “risk of positive COVID-19 test results decreased significantly with increased vitamin D level of 30 ng/mL or greater when measured as a continuous variable.”
“These new results tell us that having vitamin D levels above those normally considered sufficient is associated with decreased risk of testing positive for COVID-19, at least in Black individuals,” lead author, David Meltzer, MD, chief of hospital medicine at the University of Chicago, said in a press release from his institution.
“These findings suggest that randomized clinical trials to determine whether increasing vitamin D levels to greater than 30-40 ng/mL affect COVID-19 risk are warranted, especially in Black individuals,” he and his coauthors said.
Vitamin D at time of testing most strongly associated with COVID risk
An earlier study by the same researchers found that vitamin D deficiency (less than 20 ng/mL) may raise the risk of testing positive for COVID-19 in people from various ethnicities, as reported by this news organization.
Data for this latest study were drawn from electronic health records for 4,638 individuals at the University of Chicago Medicine and were used to examine whether the likelihood of a positive COVID-19 test was associated with a person’s most recent vitamin D level (within the previous year), and whether there was any effect of ethnicity on this outcome.
Mean age was 52.8 years, 69% were women, 49% were Black, 43% White, and 8% were another race/ethnicity. A total of 27% of the individuals were deficient in vitamin D (less than 20 ng/mL), 27% had insufficient levels (20-30 ng/mL), 22% had sufficient levels (30-40 ng/mL), and the remaining 24% had levels of 40 ng/mL or greater.
In total, 333 (7%) of people tested positive for COVID-19, including 102 (5%) Whites and 211 (9%) Blacks. And 36% of Black individuals who tested positive for COVID-19 were classified as vitamin D deficient, compared with 16% of Whites.
A positive test result for COVID-19 was not significantly associated with vitamin D levels in white individuals but was in Black individuals.
In Black people, compared with levels of at least 40 ng/mL, vitamin D levels of 30-40 ng/mL were associated with an incidence rate ratio of 2.64 for COVID-19 positivity (P = .01). For levels of 20-30 ng/mL, the IRR was 1.69 (P = 0.21); and for less than 20 ng/mL the IRR was 2.55 (P = .009).
The researchers also found that the risk of positive test results with lower vitamin D levels increased when those levels were lower just prior to the positive COVID-19 test, lending “support [to] the idea that vitamin D level at the time of testing is most strongly associated with COVID-19 risk,” they wrote.
Try upping vitamin D levels to 40 ng/mL or greater to prevent COVID?
In their discussion, the authors noted that significant association of vitamin D levels with COVID-19 risk in Blacks but not in Whites, “could reflect their higher COVID-19 risk, to which socioeconomic factors and structural inequities clearly contribute.
“Biological susceptibility to vitamin D deficiency may also be less frequent in White than Black individuals, since lighter skin increases vitamin D production in response to sunlight, and vitamin D binding proteins may vary by race and affect vitamin D bioavailability.”
Given less than 10% of U.S. adults have a vitamin D level greater than 40 ng/mL, the study findings increase the urgency to consider whether increased sun exposure or supplementation could reduce COVID-19 risk, according to the authors.
“When increased sun exposure is impractical, achieving vitamin D levels of 40 ng/mL or greater typically requires greater supplementation than currently recommended for most individuals of 600-800 IU/d vitamin D3,” they added.
However, Dr. Meltzer also acknowledged that “this is an observational study. We can see that there’s an association between vitamin D levels and likelihood of a COVID-19 diagnosis, but we don’t know exactly why that is, or whether these results are due to the vitamin D directly or other related biological factors.”
All in all, the authors suggested that randomized clinical trials are needed to understand if vitamin D can reduce COVID-19 risk, and as such they should include doses of supplements likely to increase vitamin D to at least 40 ng/mL, and perhaps even higher, although they pointed out that the latter must be achieved safely.
“Studies should also consider the role of vitamin D testing, loading doses, dose adjustments for individuals who are obese or overweight, risks for hypercalcemia, and strategies to monitor for and mitigate hypercalcemia, and that non-White populations, such as Black individuals, may have greater needs for supplementation,” they outlined.
They are now recruiting participants for two separate clinical trials testing the efficacy of vitamin D supplements for preventing COVID-19.
The authors disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Senate confirms Murthy as Surgeon General
Seven Republicans – Bill Cassidy (La.), Susan Collins (Maine), Roger Marshall (Kan.), Susan Murkowski (Alaska), Rob Portman (Ohio), Mitt Romney (Utah), and Dan Sullivan (Alaska) – joined all the Democrats and independents in the 57-43 vote approving Dr. Murthy’s nomination.
Dr. Murthy, 43, previously served as the 19th Surgeon General, from December 2014 to April 2017, when he was asked to step down by President Donald J. Trump.
Surgeons General serve 4-year terms.
During his first tenure, Dr. Murthy issued the first-ever Surgeon General’s report on the crisis of addiction and issued a call to action to doctors to help battle the opioid crisis.
When Dr. Murthy was nominated by President-elect Joseph R. Biden Jr. in December, he was acting as cochair of the incoming administration’s COVID-19 transition advisory board.
Early in 2020, before the COVID-19 pandemic hit, Dr. Murthy published a timely book: “Together: The Healing Power of Human Connection in a Sometimes Lonely World”.
He earned his bachelor’s degree from Harvard and his MD and MBA degrees from Yale. He completed his internal medicine residency at Brigham and Women’s Hospital in Boston, where he also served as a hospitalist, and later joined Harvard Medical School as a faculty member in internal medicine.
He is married to Alice Chen, MD. The couple have two children.
A version of this article first appeared on WebMD.com.
Seven Republicans – Bill Cassidy (La.), Susan Collins (Maine), Roger Marshall (Kan.), Susan Murkowski (Alaska), Rob Portman (Ohio), Mitt Romney (Utah), and Dan Sullivan (Alaska) – joined all the Democrats and independents in the 57-43 vote approving Dr. Murthy’s nomination.
Dr. Murthy, 43, previously served as the 19th Surgeon General, from December 2014 to April 2017, when he was asked to step down by President Donald J. Trump.
Surgeons General serve 4-year terms.
During his first tenure, Dr. Murthy issued the first-ever Surgeon General’s report on the crisis of addiction and issued a call to action to doctors to help battle the opioid crisis.
When Dr. Murthy was nominated by President-elect Joseph R. Biden Jr. in December, he was acting as cochair of the incoming administration’s COVID-19 transition advisory board.
Early in 2020, before the COVID-19 pandemic hit, Dr. Murthy published a timely book: “Together: The Healing Power of Human Connection in a Sometimes Lonely World”.
He earned his bachelor’s degree from Harvard and his MD and MBA degrees from Yale. He completed his internal medicine residency at Brigham and Women’s Hospital in Boston, where he also served as a hospitalist, and later joined Harvard Medical School as a faculty member in internal medicine.
He is married to Alice Chen, MD. The couple have two children.
A version of this article first appeared on WebMD.com.
Seven Republicans – Bill Cassidy (La.), Susan Collins (Maine), Roger Marshall (Kan.), Susan Murkowski (Alaska), Rob Portman (Ohio), Mitt Romney (Utah), and Dan Sullivan (Alaska) – joined all the Democrats and independents in the 57-43 vote approving Dr. Murthy’s nomination.
Dr. Murthy, 43, previously served as the 19th Surgeon General, from December 2014 to April 2017, when he was asked to step down by President Donald J. Trump.
Surgeons General serve 4-year terms.
During his first tenure, Dr. Murthy issued the first-ever Surgeon General’s report on the crisis of addiction and issued a call to action to doctors to help battle the opioid crisis.
When Dr. Murthy was nominated by President-elect Joseph R. Biden Jr. in December, he was acting as cochair of the incoming administration’s COVID-19 transition advisory board.
Early in 2020, before the COVID-19 pandemic hit, Dr. Murthy published a timely book: “Together: The Healing Power of Human Connection in a Sometimes Lonely World”.
He earned his bachelor’s degree from Harvard and his MD and MBA degrees from Yale. He completed his internal medicine residency at Brigham and Women’s Hospital in Boston, where he also served as a hospitalist, and later joined Harvard Medical School as a faculty member in internal medicine.
He is married to Alice Chen, MD. The couple have two children.
A version of this article first appeared on WebMD.com.
Discovery of schizophrenia gene could advance research, therapies
A new genetic mutation in schizophrenia that blocks neuron communication in the brain may lead to novel treatment strategies and improve understanding of the mechanics of this disease.
The discovery of this new gene, PCDHA3, could enhance the development of genetic-risk calculators “that may help us understand vulnerability to schizophrenia in high-risk individuals and identify individuals with schizophrenia who have a greater risk for poor outcomes,” said Todd Lencz, PhD, a professor at the Feinstein Institutes for Medical Research in New York, and lead author of this research. Dr. Lencz and associates reported on this new finding in the journal Neuron.
Schizophrenia affects 20 million people worldwide. Previous research has identified the important role genes play in the disease, but isolating individual genes to better understand schizophrenia has proven to be a challenge. This is a very heterogeneous disorder, with many hundreds if not thousands of genes involved, Dr. Lencz explained in an interview. “It is very different from single-gene disorders like Huntington disease, for example. For this reason, we need very large sample sizes to find any one gene that seems to be common to many cases in a sample.”
Study focused on homogeneous population
To enhance the power of finding rare variants in a heterogeneous disease with large numbers of genes, Dr. Lencz and colleagues chose a homogeneous “founder” population, a cohort of Ashkenazi Jews, to examine genomes from schizophrenia patients and controls. “As we have reported in prior work over the last decade, the 10 million or so Ashkenazi Jews living worldwide today all are descended from just a few hundred people who lived approximately 750 years ago, and moved into Central and Eastern Europe,” said Dr. Lencz. The study included 786 cases of schizophrenia and 463 controls from this Ashkenazi population. This is considered to be an extremely small sample for a genetic study. However, because this population evolved from a few hundred individuals to a massive explosion in a historically short period of time, it had enhanced statistical power, said Dr. Lencz.
“We showed that just a few thousand Ashkenazi Jewish cases would have the statistical power of a regular population that was 5-10 times larger, from a genetic discovery perspective,” he added.
Search for ultrarare variants
The investigators used whole-genome sequencing to conduct their analysis, using public databases to filter out any variants that had been previously observed in healthy individuals worldwide. “We were looking for ultrarare variants that might have a very powerful effect on the disease,” Dr. Lencz said. Such individual mutations are very rarely seen in the general population.
Because of the disease’s ultraheterogeneity, it’s extremely unusual to find a recurrent, ultrarare variant. “In some ways, the genetics of schizophrenia is so complex that every patient worldwide is unique in the genetics that led to his or her disorder.” The goal was to find individual mutations that might be observed multiple times across the schizophrenia group, Dr. Lencz said.
Rare gene found in five cases
Dr. Lencz and colleagues accomplished this with their unique Ashkenazi Jewish population. “We identified one particular mutation that was repeatedly observed in our cases that has not been observed in healthy individuals that we’re aware of,” he said. The PCDHA3 mutation was identified in 3 out of the 786 schizophrenia cases.
In another dataset, they examined from the Schizophrenia Exome Sequencing Meta-analysis (SCHEMA) consortium, they found it two additional times, bringing the total to five cases. SCHEMA is a large international consortium of genetics studies in schizophrenia that contains thousands of cases and controls, some of which are Ashkenazi Jewish cases.
“Importantly, the mutation was not observed in any controls, in either our Ashkenazi dataset, the SCHEMA dataset, or more than 100,000 other controls reported in several publicly available genetics databases,” Dr. Lencz said.
How the gene leads to schizophrenia
PCDHA3 derives from the protocadherin gene family, which generates a unique bar code that enables neurons to recognize and communicate with other neurons. This communication creates a scaffolding of sorts that enables normal brain function. Dr. Lencz and colleagues discovered that the PCDHA3 variant blocks this normal protocadherin function.
Among the 786 cases, the investigators found several other genes in the broad cadherin family that had implications in schizophrenia development.
Much of the genetics of schizophrenia in recent years has focused on the synapse as the point of abnormality underlying the disorder. “We think our paper demonstrates in multiple ways the synaptic scaffolding role the cadherins superfamily of genes play in schizophrenia pathophysiology. The discovery of the PCDHA3 variant adds a level of detail and resolution to this process, pointing researchers toward a specific aspect of synaptic formation that may be aberrant. “So the hope is we’re not just learning about these five individuals and their synapses. This result is perhaps telling us to look very carefully at this aspect of synaptic formation.”
Implications for clinical practice
Dr. Lencz and colleagues plan to expand upon and enhance their existing Ashkenazi sample to take advantage of the founder effect in this population. “Of course, there are many large-scale efforts to recruit ethnically diverse patients with schizophrenia to study around the world. We encourage that. Our expectation is that the biology is not in any way unique to Ashkenazi individuals. This is just the approach we took to enhance our power,” he said.
The PCDHA3 discovery won’t have an immediate impact on clinical practice. In the longer term, “we are aware of certain pharmacologic approaches that might be able to manipulate the cadherins. That would be a worthy focus for future research,” Dr. Lencz said.
Additional studies will be critical to see how current medications in schizophrenia treatment could mitigate and improve any changes caused by this genetic mutation, noted Anthony T. Ng, MD, who was not involved with the study. More specifically, studies would help assess the impact of a schizophrenia patient with this mutation in areas of functioning, “so that psychosocial and rehabilitation treatment models of schizophrenia can provide more targeted treatment,” said Dr. Ng, medical director of community services and director of neuromodulation services at Northern Light Acadia Hospital in Bangor, Maine.
The work of Dr. Lencz and associates is significant in that “it started to identify a very specific genetic change that can help focus treatment of schizophrenia,” Dr. Ng said.
Neither Dr. Lencz nor his associates had any conflicts of interest. Dr. Ng had no disclosures.
A new genetic mutation in schizophrenia that blocks neuron communication in the brain may lead to novel treatment strategies and improve understanding of the mechanics of this disease.
The discovery of this new gene, PCDHA3, could enhance the development of genetic-risk calculators “that may help us understand vulnerability to schizophrenia in high-risk individuals and identify individuals with schizophrenia who have a greater risk for poor outcomes,” said Todd Lencz, PhD, a professor at the Feinstein Institutes for Medical Research in New York, and lead author of this research. Dr. Lencz and associates reported on this new finding in the journal Neuron.
Schizophrenia affects 20 million people worldwide. Previous research has identified the important role genes play in the disease, but isolating individual genes to better understand schizophrenia has proven to be a challenge. This is a very heterogeneous disorder, with many hundreds if not thousands of genes involved, Dr. Lencz explained in an interview. “It is very different from single-gene disorders like Huntington disease, for example. For this reason, we need very large sample sizes to find any one gene that seems to be common to many cases in a sample.”
Study focused on homogeneous population
To enhance the power of finding rare variants in a heterogeneous disease with large numbers of genes, Dr. Lencz and colleagues chose a homogeneous “founder” population, a cohort of Ashkenazi Jews, to examine genomes from schizophrenia patients and controls. “As we have reported in prior work over the last decade, the 10 million or so Ashkenazi Jews living worldwide today all are descended from just a few hundred people who lived approximately 750 years ago, and moved into Central and Eastern Europe,” said Dr. Lencz. The study included 786 cases of schizophrenia and 463 controls from this Ashkenazi population. This is considered to be an extremely small sample for a genetic study. However, because this population evolved from a few hundred individuals to a massive explosion in a historically short period of time, it had enhanced statistical power, said Dr. Lencz.
“We showed that just a few thousand Ashkenazi Jewish cases would have the statistical power of a regular population that was 5-10 times larger, from a genetic discovery perspective,” he added.
Search for ultrarare variants
The investigators used whole-genome sequencing to conduct their analysis, using public databases to filter out any variants that had been previously observed in healthy individuals worldwide. “We were looking for ultrarare variants that might have a very powerful effect on the disease,” Dr. Lencz said. Such individual mutations are very rarely seen in the general population.
Because of the disease’s ultraheterogeneity, it’s extremely unusual to find a recurrent, ultrarare variant. “In some ways, the genetics of schizophrenia is so complex that every patient worldwide is unique in the genetics that led to his or her disorder.” The goal was to find individual mutations that might be observed multiple times across the schizophrenia group, Dr. Lencz said.
Rare gene found in five cases
Dr. Lencz and colleagues accomplished this with their unique Ashkenazi Jewish population. “We identified one particular mutation that was repeatedly observed in our cases that has not been observed in healthy individuals that we’re aware of,” he said. The PCDHA3 mutation was identified in 3 out of the 786 schizophrenia cases.
In another dataset, they examined from the Schizophrenia Exome Sequencing Meta-analysis (SCHEMA) consortium, they found it two additional times, bringing the total to five cases. SCHEMA is a large international consortium of genetics studies in schizophrenia that contains thousands of cases and controls, some of which are Ashkenazi Jewish cases.
“Importantly, the mutation was not observed in any controls, in either our Ashkenazi dataset, the SCHEMA dataset, or more than 100,000 other controls reported in several publicly available genetics databases,” Dr. Lencz said.
How the gene leads to schizophrenia
PCDHA3 derives from the protocadherin gene family, which generates a unique bar code that enables neurons to recognize and communicate with other neurons. This communication creates a scaffolding of sorts that enables normal brain function. Dr. Lencz and colleagues discovered that the PCDHA3 variant blocks this normal protocadherin function.
Among the 786 cases, the investigators found several other genes in the broad cadherin family that had implications in schizophrenia development.
Much of the genetics of schizophrenia in recent years has focused on the synapse as the point of abnormality underlying the disorder. “We think our paper demonstrates in multiple ways the synaptic scaffolding role the cadherins superfamily of genes play in schizophrenia pathophysiology. The discovery of the PCDHA3 variant adds a level of detail and resolution to this process, pointing researchers toward a specific aspect of synaptic formation that may be aberrant. “So the hope is we’re not just learning about these five individuals and their synapses. This result is perhaps telling us to look very carefully at this aspect of synaptic formation.”
Implications for clinical practice
Dr. Lencz and colleagues plan to expand upon and enhance their existing Ashkenazi sample to take advantage of the founder effect in this population. “Of course, there are many large-scale efforts to recruit ethnically diverse patients with schizophrenia to study around the world. We encourage that. Our expectation is that the biology is not in any way unique to Ashkenazi individuals. This is just the approach we took to enhance our power,” he said.
The PCDHA3 discovery won’t have an immediate impact on clinical practice. In the longer term, “we are aware of certain pharmacologic approaches that might be able to manipulate the cadherins. That would be a worthy focus for future research,” Dr. Lencz said.
Additional studies will be critical to see how current medications in schizophrenia treatment could mitigate and improve any changes caused by this genetic mutation, noted Anthony T. Ng, MD, who was not involved with the study. More specifically, studies would help assess the impact of a schizophrenia patient with this mutation in areas of functioning, “so that psychosocial and rehabilitation treatment models of schizophrenia can provide more targeted treatment,” said Dr. Ng, medical director of community services and director of neuromodulation services at Northern Light Acadia Hospital in Bangor, Maine.
The work of Dr. Lencz and associates is significant in that “it started to identify a very specific genetic change that can help focus treatment of schizophrenia,” Dr. Ng said.
Neither Dr. Lencz nor his associates had any conflicts of interest. Dr. Ng had no disclosures.
A new genetic mutation in schizophrenia that blocks neuron communication in the brain may lead to novel treatment strategies and improve understanding of the mechanics of this disease.
The discovery of this new gene, PCDHA3, could enhance the development of genetic-risk calculators “that may help us understand vulnerability to schizophrenia in high-risk individuals and identify individuals with schizophrenia who have a greater risk for poor outcomes,” said Todd Lencz, PhD, a professor at the Feinstein Institutes for Medical Research in New York, and lead author of this research. Dr. Lencz and associates reported on this new finding in the journal Neuron.
Schizophrenia affects 20 million people worldwide. Previous research has identified the important role genes play in the disease, but isolating individual genes to better understand schizophrenia has proven to be a challenge. This is a very heterogeneous disorder, with many hundreds if not thousands of genes involved, Dr. Lencz explained in an interview. “It is very different from single-gene disorders like Huntington disease, for example. For this reason, we need very large sample sizes to find any one gene that seems to be common to many cases in a sample.”
Study focused on homogeneous population
To enhance the power of finding rare variants in a heterogeneous disease with large numbers of genes, Dr. Lencz and colleagues chose a homogeneous “founder” population, a cohort of Ashkenazi Jews, to examine genomes from schizophrenia patients and controls. “As we have reported in prior work over the last decade, the 10 million or so Ashkenazi Jews living worldwide today all are descended from just a few hundred people who lived approximately 750 years ago, and moved into Central and Eastern Europe,” said Dr. Lencz. The study included 786 cases of schizophrenia and 463 controls from this Ashkenazi population. This is considered to be an extremely small sample for a genetic study. However, because this population evolved from a few hundred individuals to a massive explosion in a historically short period of time, it had enhanced statistical power, said Dr. Lencz.
“We showed that just a few thousand Ashkenazi Jewish cases would have the statistical power of a regular population that was 5-10 times larger, from a genetic discovery perspective,” he added.
Search for ultrarare variants
The investigators used whole-genome sequencing to conduct their analysis, using public databases to filter out any variants that had been previously observed in healthy individuals worldwide. “We were looking for ultrarare variants that might have a very powerful effect on the disease,” Dr. Lencz said. Such individual mutations are very rarely seen in the general population.
Because of the disease’s ultraheterogeneity, it’s extremely unusual to find a recurrent, ultrarare variant. “In some ways, the genetics of schizophrenia is so complex that every patient worldwide is unique in the genetics that led to his or her disorder.” The goal was to find individual mutations that might be observed multiple times across the schizophrenia group, Dr. Lencz said.
Rare gene found in five cases
Dr. Lencz and colleagues accomplished this with their unique Ashkenazi Jewish population. “We identified one particular mutation that was repeatedly observed in our cases that has not been observed in healthy individuals that we’re aware of,” he said. The PCDHA3 mutation was identified in 3 out of the 786 schizophrenia cases.
In another dataset, they examined from the Schizophrenia Exome Sequencing Meta-analysis (SCHEMA) consortium, they found it two additional times, bringing the total to five cases. SCHEMA is a large international consortium of genetics studies in schizophrenia that contains thousands of cases and controls, some of which are Ashkenazi Jewish cases.
“Importantly, the mutation was not observed in any controls, in either our Ashkenazi dataset, the SCHEMA dataset, or more than 100,000 other controls reported in several publicly available genetics databases,” Dr. Lencz said.
How the gene leads to schizophrenia
PCDHA3 derives from the protocadherin gene family, which generates a unique bar code that enables neurons to recognize and communicate with other neurons. This communication creates a scaffolding of sorts that enables normal brain function. Dr. Lencz and colleagues discovered that the PCDHA3 variant blocks this normal protocadherin function.
Among the 786 cases, the investigators found several other genes in the broad cadherin family that had implications in schizophrenia development.
Much of the genetics of schizophrenia in recent years has focused on the synapse as the point of abnormality underlying the disorder. “We think our paper demonstrates in multiple ways the synaptic scaffolding role the cadherins superfamily of genes play in schizophrenia pathophysiology. The discovery of the PCDHA3 variant adds a level of detail and resolution to this process, pointing researchers toward a specific aspect of synaptic formation that may be aberrant. “So the hope is we’re not just learning about these five individuals and their synapses. This result is perhaps telling us to look very carefully at this aspect of synaptic formation.”
Implications for clinical practice
Dr. Lencz and colleagues plan to expand upon and enhance their existing Ashkenazi sample to take advantage of the founder effect in this population. “Of course, there are many large-scale efforts to recruit ethnically diverse patients with schizophrenia to study around the world. We encourage that. Our expectation is that the biology is not in any way unique to Ashkenazi individuals. This is just the approach we took to enhance our power,” he said.
The PCDHA3 discovery won’t have an immediate impact on clinical practice. In the longer term, “we are aware of certain pharmacologic approaches that might be able to manipulate the cadherins. That would be a worthy focus for future research,” Dr. Lencz said.
Additional studies will be critical to see how current medications in schizophrenia treatment could mitigate and improve any changes caused by this genetic mutation, noted Anthony T. Ng, MD, who was not involved with the study. More specifically, studies would help assess the impact of a schizophrenia patient with this mutation in areas of functioning, “so that psychosocial and rehabilitation treatment models of schizophrenia can provide more targeted treatment,” said Dr. Ng, medical director of community services and director of neuromodulation services at Northern Light Acadia Hospital in Bangor, Maine.
The work of Dr. Lencz and associates is significant in that “it started to identify a very specific genetic change that can help focus treatment of schizophrenia,” Dr. Ng said.
Neither Dr. Lencz nor his associates had any conflicts of interest. Dr. Ng had no disclosures.
FROM NEURON
Change is hard: Lessons from an EHR conversion
During this “go-live,” 5 hospitals and approximately 300 ambulatory service and physician practice locations made the transition, consolidating over 100 disparate electronic systems and dozens of interfaces into one world-class medical record.
If you’ve ever been part of such an event, you know it is anything but simple. On the contrary, it requires an enormous financial investment along with years of planning, hours of meetings, and months of training. No matter how much preparation goes into it, there are sure to be bumps along the way. It is a traumatic and stressful time for all involved, but the end result is well worth the effort. Still, there are lessons to be learned and wisdom to be gleaned, and this month we’d like to share a few that we found most important. We believe that many of these are useful lessons even to those who will never live through a go-live.
Safety always comes first
Patient safety is a term so often used that it has a tendency to be taken for granted. Health systems build processes and procedures to ensure safety – some even win awards and recognition for their efforts. But the best (and safest) health care institutions build patient safety into their cultures. More than just being taught to use checklists or buzzwords, the staff at these institutions are encouraged to put the welfare of patients first, making all other activities secondary to this pursuit. We had the opportunity to witness the benefits of such a culture during this go-live and were incredibly impressed with the results.
To be successful in an EHR transition of any magnitude, an organization needs to hold patient safety as a core value and provide its employees with the tools to execute on that value. This enables staff to prepare adequately and to identify risks and opportunities before the conversion takes place. Once go-live occurs, staff also must feel empowered to speak up when they identify problem areas that might jeopardize patients’ care. They also must be given a clear escalation path to ensure their voices can be heard. Most importantly, everyone must understand that the electronic health record itself is just one piece of a major operational change.
As workflows are modified to adapt to the new technology, unsafe processes should be called out and fixed quickly. While the EHR may offer the latest in decision support and system integration, no advancement in technology can make up for bad outcomes, nor justify processes that lead to patient harm.
Training is no substitute for good support
It takes a long time to train thousands of employees, especially when that training must occur during the era of social distancing in the midst of a pandemic. Still, even in the best of times, education should be married to hands-on experience in order to have a real impact. Unfortunately, this is extremely challenging.
Trainees forget much of what they’ve learned in the weeks or months between education and go-live, so they must be given immediately accessible support to bridge the gap. This is known as “at-the-elbow” (ATE) support, and as the name implies, it consists of individuals who are familiar with the new system and are always available to end users, answering their questions and helping them navigate. Since health care never sleeps, this support needs to be offered 24/7, and it should also be flexible and plentiful.
There are many areas that will require more support than anticipated to accommodate the number of clinical and other staff who will use the system, so support staff must be nimble and available for redeployment. In addition, ensuring high-quality support is essential. As many ATE experts are hired contractors, their knowledge base and communications skills can vary widely. Accountability is key, and end users should feel empowered to identify gaps in coverage and deficits in knowledge base in the ATE.
As employees become more familiar with the new system, the need for ATE will wane, but there will still be questions that arise for many weeks to months, and new EHR users will also be added all the time. A good after–go-live support system should remain available so clinical and clerical employees can get just-in-time assistance whenever they need it.
Users should be given clear expectations
Clinicians going through an EHR conversion may be frustrated to discover that the data transferred from their old system into the new one is not quite what they expected. While structured elements such as allergies and immunizations may transfer, unstructured patient histories may not come over at all.
There may be gaps in data, or the opposite may even be true: an overabundance of useless information may transfer over, leaving doctors with dozens of meaningless data points to sift through and eliminate to clean up the chart. This can be extremely time-consuming and discouraging and may jeopardize the success of the go-live.
Providers deserve clear expectations prior to conversion. They should be told what will and will not transfer and be informed that there will be extra work required for documentation at the outset. They may also want the option to preemptively reduce patient volumes to accommodate the additional effort involved in preparing charts. No matter what, this will be a heavy lift, and physicians should understand the implications long before go-live to prepare accordingly.
Old habits die hard
One of the most common complaints we’ve heard following EHR conversions is that “things just worked better in the old system.” We always respond with a question: “Were things better, or just different?” The truth may lie somewhere in the middle, but there is no question that muscle memory develops over many years, and change is difficult no matter how much better the new system is. Still, appropriate expectations, access to just-in-time support, and a continual focus on safety will ensure that the long-term benefits of a patient-centered and integrated electronic record will far outweigh the initial challenges of go-live.
Dr. Notte is a family physician and chief medical officer of Abington (Pa.) Hospital–Jefferson Health. Dr. Skolnik is professor of family and community medicine at Sidney Kimmel Medical College, Philadelphia, and associate director of the family medicine residency program at Abington Hospital–Jefferson Health. They have no conflicts related to the content of this piece.
During this “go-live,” 5 hospitals and approximately 300 ambulatory service and physician practice locations made the transition, consolidating over 100 disparate electronic systems and dozens of interfaces into one world-class medical record.
If you’ve ever been part of such an event, you know it is anything but simple. On the contrary, it requires an enormous financial investment along with years of planning, hours of meetings, and months of training. No matter how much preparation goes into it, there are sure to be bumps along the way. It is a traumatic and stressful time for all involved, but the end result is well worth the effort. Still, there are lessons to be learned and wisdom to be gleaned, and this month we’d like to share a few that we found most important. We believe that many of these are useful lessons even to those who will never live through a go-live.
Safety always comes first
Patient safety is a term so often used that it has a tendency to be taken for granted. Health systems build processes and procedures to ensure safety – some even win awards and recognition for their efforts. But the best (and safest) health care institutions build patient safety into their cultures. More than just being taught to use checklists or buzzwords, the staff at these institutions are encouraged to put the welfare of patients first, making all other activities secondary to this pursuit. We had the opportunity to witness the benefits of such a culture during this go-live and were incredibly impressed with the results.
To be successful in an EHR transition of any magnitude, an organization needs to hold patient safety as a core value and provide its employees with the tools to execute on that value. This enables staff to prepare adequately and to identify risks and opportunities before the conversion takes place. Once go-live occurs, staff also must feel empowered to speak up when they identify problem areas that might jeopardize patients’ care. They also must be given a clear escalation path to ensure their voices can be heard. Most importantly, everyone must understand that the electronic health record itself is just one piece of a major operational change.
As workflows are modified to adapt to the new technology, unsafe processes should be called out and fixed quickly. While the EHR may offer the latest in decision support and system integration, no advancement in technology can make up for bad outcomes, nor justify processes that lead to patient harm.
Training is no substitute for good support
It takes a long time to train thousands of employees, especially when that training must occur during the era of social distancing in the midst of a pandemic. Still, even in the best of times, education should be married to hands-on experience in order to have a real impact. Unfortunately, this is extremely challenging.
Trainees forget much of what they’ve learned in the weeks or months between education and go-live, so they must be given immediately accessible support to bridge the gap. This is known as “at-the-elbow” (ATE) support, and as the name implies, it consists of individuals who are familiar with the new system and are always available to end users, answering their questions and helping them navigate. Since health care never sleeps, this support needs to be offered 24/7, and it should also be flexible and plentiful.
There are many areas that will require more support than anticipated to accommodate the number of clinical and other staff who will use the system, so support staff must be nimble and available for redeployment. In addition, ensuring high-quality support is essential. As many ATE experts are hired contractors, their knowledge base and communications skills can vary widely. Accountability is key, and end users should feel empowered to identify gaps in coverage and deficits in knowledge base in the ATE.
As employees become more familiar with the new system, the need for ATE will wane, but there will still be questions that arise for many weeks to months, and new EHR users will also be added all the time. A good after–go-live support system should remain available so clinical and clerical employees can get just-in-time assistance whenever they need it.
Users should be given clear expectations
Clinicians going through an EHR conversion may be frustrated to discover that the data transferred from their old system into the new one is not quite what they expected. While structured elements such as allergies and immunizations may transfer, unstructured patient histories may not come over at all.
There may be gaps in data, or the opposite may even be true: an overabundance of useless information may transfer over, leaving doctors with dozens of meaningless data points to sift through and eliminate to clean up the chart. This can be extremely time-consuming and discouraging and may jeopardize the success of the go-live.
Providers deserve clear expectations prior to conversion. They should be told what will and will not transfer and be informed that there will be extra work required for documentation at the outset. They may also want the option to preemptively reduce patient volumes to accommodate the additional effort involved in preparing charts. No matter what, this will be a heavy lift, and physicians should understand the implications long before go-live to prepare accordingly.
Old habits die hard
One of the most common complaints we’ve heard following EHR conversions is that “things just worked better in the old system.” We always respond with a question: “Were things better, or just different?” The truth may lie somewhere in the middle, but there is no question that muscle memory develops over many years, and change is difficult no matter how much better the new system is. Still, appropriate expectations, access to just-in-time support, and a continual focus on safety will ensure that the long-term benefits of a patient-centered and integrated electronic record will far outweigh the initial challenges of go-live.
Dr. Notte is a family physician and chief medical officer of Abington (Pa.) Hospital–Jefferson Health. Dr. Skolnik is professor of family and community medicine at Sidney Kimmel Medical College, Philadelphia, and associate director of the family medicine residency program at Abington Hospital–Jefferson Health. They have no conflicts related to the content of this piece.
During this “go-live,” 5 hospitals and approximately 300 ambulatory service and physician practice locations made the transition, consolidating over 100 disparate electronic systems and dozens of interfaces into one world-class medical record.
If you’ve ever been part of such an event, you know it is anything but simple. On the contrary, it requires an enormous financial investment along with years of planning, hours of meetings, and months of training. No matter how much preparation goes into it, there are sure to be bumps along the way. It is a traumatic and stressful time for all involved, but the end result is well worth the effort. Still, there are lessons to be learned and wisdom to be gleaned, and this month we’d like to share a few that we found most important. We believe that many of these are useful lessons even to those who will never live through a go-live.
Safety always comes first
Patient safety is a term so often used that it has a tendency to be taken for granted. Health systems build processes and procedures to ensure safety – some even win awards and recognition for their efforts. But the best (and safest) health care institutions build patient safety into their cultures. More than just being taught to use checklists or buzzwords, the staff at these institutions are encouraged to put the welfare of patients first, making all other activities secondary to this pursuit. We had the opportunity to witness the benefits of such a culture during this go-live and were incredibly impressed with the results.
To be successful in an EHR transition of any magnitude, an organization needs to hold patient safety as a core value and provide its employees with the tools to execute on that value. This enables staff to prepare adequately and to identify risks and opportunities before the conversion takes place. Once go-live occurs, staff also must feel empowered to speak up when they identify problem areas that might jeopardize patients’ care. They also must be given a clear escalation path to ensure their voices can be heard. Most importantly, everyone must understand that the electronic health record itself is just one piece of a major operational change.
As workflows are modified to adapt to the new technology, unsafe processes should be called out and fixed quickly. While the EHR may offer the latest in decision support and system integration, no advancement in technology can make up for bad outcomes, nor justify processes that lead to patient harm.
Training is no substitute for good support
It takes a long time to train thousands of employees, especially when that training must occur during the era of social distancing in the midst of a pandemic. Still, even in the best of times, education should be married to hands-on experience in order to have a real impact. Unfortunately, this is extremely challenging.
Trainees forget much of what they’ve learned in the weeks or months between education and go-live, so they must be given immediately accessible support to bridge the gap. This is known as “at-the-elbow” (ATE) support, and as the name implies, it consists of individuals who are familiar with the new system and are always available to end users, answering their questions and helping them navigate. Since health care never sleeps, this support needs to be offered 24/7, and it should also be flexible and plentiful.
There are many areas that will require more support than anticipated to accommodate the number of clinical and other staff who will use the system, so support staff must be nimble and available for redeployment. In addition, ensuring high-quality support is essential. As many ATE experts are hired contractors, their knowledge base and communications skills can vary widely. Accountability is key, and end users should feel empowered to identify gaps in coverage and deficits in knowledge base in the ATE.
As employees become more familiar with the new system, the need for ATE will wane, but there will still be questions that arise for many weeks to months, and new EHR users will also be added all the time. A good after–go-live support system should remain available so clinical and clerical employees can get just-in-time assistance whenever they need it.
Users should be given clear expectations
Clinicians going through an EHR conversion may be frustrated to discover that the data transferred from their old system into the new one is not quite what they expected. While structured elements such as allergies and immunizations may transfer, unstructured patient histories may not come over at all.
There may be gaps in data, or the opposite may even be true: an overabundance of useless information may transfer over, leaving doctors with dozens of meaningless data points to sift through and eliminate to clean up the chart. This can be extremely time-consuming and discouraging and may jeopardize the success of the go-live.
Providers deserve clear expectations prior to conversion. They should be told what will and will not transfer and be informed that there will be extra work required for documentation at the outset. They may also want the option to preemptively reduce patient volumes to accommodate the additional effort involved in preparing charts. No matter what, this will be a heavy lift, and physicians should understand the implications long before go-live to prepare accordingly.
Old habits die hard
One of the most common complaints we’ve heard following EHR conversions is that “things just worked better in the old system.” We always respond with a question: “Were things better, or just different?” The truth may lie somewhere in the middle, but there is no question that muscle memory develops over many years, and change is difficult no matter how much better the new system is. Still, appropriate expectations, access to just-in-time support, and a continual focus on safety will ensure that the long-term benefits of a patient-centered and integrated electronic record will far outweigh the initial challenges of go-live.
Dr. Notte is a family physician and chief medical officer of Abington (Pa.) Hospital–Jefferson Health. Dr. Skolnik is professor of family and community medicine at Sidney Kimmel Medical College, Philadelphia, and associate director of the family medicine residency program at Abington Hospital–Jefferson Health. They have no conflicts related to the content of this piece.
FDA warning letters target OTC cannabidiol product claims for pain relief
The Food and Drug Administration has warned two manufacturers about illegal marketing of drugs containing cannabidiol (CBD) for over-the-counter use without an approved new drug application, for using substandard manufacturing processes, and for failure to comply with current good manufacturing practices. These warnings add to 51 previous warning letters issued by the FDA since 2015 to other manufacturers of products containing CBD who were violating the Federal Food, Drug, and Cosmetic Act.
In a news release, the agency explained that its two most recent letters, sent to Honest Globe Inc. on March 15 and BioLyte Laboratories LLC on March 18, were issued because CBD has “known pharmacologic effects on humans, with demonstrated risks, it cannot be legally marketed as an inactive ingredient in OTC drug products that are not reviewed and approved by the FDA.” They also describe the companies’ failures to comply with current good manufacturing practices.
“The FDA continues to alert the public to potential safety and efficacy concerns with unapproved CBD products sold online and in stores across the country,” FDA Principal Deputy Commissioner Amy P. Abernethy, MD, PhD, said in the release. “It’s important that consumers understand that the FDA has only approved one drug containing CBD as an ingredient [Epidiolex]. These other, unapproved, CBD products may have dangerous health impacts and side effects. We remain focused on exploring potential pathways for CBD products to be lawfully marketed while also educating the public about these outstanding questions of CBD’s safety. Meanwhile, we will continue to monitor and take action, as needed, against companies that unlawfully market their products – prioritizing those that pose a risk to public health.”
The specific products from Santa Ana, Calif.–based Honest Globe that the FDA called unapproved new drugs and misbranded under the Federal Food, Drug, and Cosmetic Act included Elixicure Original Pain Relief and Elixicure Lavender Pain Relief, both of which were described as containing CBD. Products from Grand Rapids, Mich.–based BioLyte Laboratories LLC that the FDA similarly cited for violations included Silver Gel, Silver Gel with Aloe, Silver Liquid Supplement, Therapeutic Pain Gel, Pain Relief Cream, and Magnesium Oil Spray.
The agency has asked the two companies to respond to its letters within 15 working days, “stating how they will address these violations or providing their reasoning and supporting information as to why they believe these products are not in violation of the law. Failure to adequately address the violations promptly may result in legal action, including product seizure and/or injunction.”
The Food and Drug Administration has warned two manufacturers about illegal marketing of drugs containing cannabidiol (CBD) for over-the-counter use without an approved new drug application, for using substandard manufacturing processes, and for failure to comply with current good manufacturing practices. These warnings add to 51 previous warning letters issued by the FDA since 2015 to other manufacturers of products containing CBD who were violating the Federal Food, Drug, and Cosmetic Act.
In a news release, the agency explained that its two most recent letters, sent to Honest Globe Inc. on March 15 and BioLyte Laboratories LLC on March 18, were issued because CBD has “known pharmacologic effects on humans, with demonstrated risks, it cannot be legally marketed as an inactive ingredient in OTC drug products that are not reviewed and approved by the FDA.” They also describe the companies’ failures to comply with current good manufacturing practices.
“The FDA continues to alert the public to potential safety and efficacy concerns with unapproved CBD products sold online and in stores across the country,” FDA Principal Deputy Commissioner Amy P. Abernethy, MD, PhD, said in the release. “It’s important that consumers understand that the FDA has only approved one drug containing CBD as an ingredient [Epidiolex]. These other, unapproved, CBD products may have dangerous health impacts and side effects. We remain focused on exploring potential pathways for CBD products to be lawfully marketed while also educating the public about these outstanding questions of CBD’s safety. Meanwhile, we will continue to monitor and take action, as needed, against companies that unlawfully market their products – prioritizing those that pose a risk to public health.”
The specific products from Santa Ana, Calif.–based Honest Globe that the FDA called unapproved new drugs and misbranded under the Federal Food, Drug, and Cosmetic Act included Elixicure Original Pain Relief and Elixicure Lavender Pain Relief, both of which were described as containing CBD. Products from Grand Rapids, Mich.–based BioLyte Laboratories LLC that the FDA similarly cited for violations included Silver Gel, Silver Gel with Aloe, Silver Liquid Supplement, Therapeutic Pain Gel, Pain Relief Cream, and Magnesium Oil Spray.
The agency has asked the two companies to respond to its letters within 15 working days, “stating how they will address these violations or providing their reasoning and supporting information as to why they believe these products are not in violation of the law. Failure to adequately address the violations promptly may result in legal action, including product seizure and/or injunction.”
The Food and Drug Administration has warned two manufacturers about illegal marketing of drugs containing cannabidiol (CBD) for over-the-counter use without an approved new drug application, for using substandard manufacturing processes, and for failure to comply with current good manufacturing practices. These warnings add to 51 previous warning letters issued by the FDA since 2015 to other manufacturers of products containing CBD who were violating the Federal Food, Drug, and Cosmetic Act.
In a news release, the agency explained that its two most recent letters, sent to Honest Globe Inc. on March 15 and BioLyte Laboratories LLC on March 18, were issued because CBD has “known pharmacologic effects on humans, with demonstrated risks, it cannot be legally marketed as an inactive ingredient in OTC drug products that are not reviewed and approved by the FDA.” They also describe the companies’ failures to comply with current good manufacturing practices.
“The FDA continues to alert the public to potential safety and efficacy concerns with unapproved CBD products sold online and in stores across the country,” FDA Principal Deputy Commissioner Amy P. Abernethy, MD, PhD, said in the release. “It’s important that consumers understand that the FDA has only approved one drug containing CBD as an ingredient [Epidiolex]. These other, unapproved, CBD products may have dangerous health impacts and side effects. We remain focused on exploring potential pathways for CBD products to be lawfully marketed while also educating the public about these outstanding questions of CBD’s safety. Meanwhile, we will continue to monitor and take action, as needed, against companies that unlawfully market their products – prioritizing those that pose a risk to public health.”
The specific products from Santa Ana, Calif.–based Honest Globe that the FDA called unapproved new drugs and misbranded under the Federal Food, Drug, and Cosmetic Act included Elixicure Original Pain Relief and Elixicure Lavender Pain Relief, both of which were described as containing CBD. Products from Grand Rapids, Mich.–based BioLyte Laboratories LLC that the FDA similarly cited for violations included Silver Gel, Silver Gel with Aloe, Silver Liquid Supplement, Therapeutic Pain Gel, Pain Relief Cream, and Magnesium Oil Spray.
The agency has asked the two companies to respond to its letters within 15 working days, “stating how they will address these violations or providing their reasoning and supporting information as to why they believe these products are not in violation of the law. Failure to adequately address the violations promptly may result in legal action, including product seizure and/or injunction.”