User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'main-prefix')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
div[contains(@class, 'view-medstat-quiz-listing-panes')]
div[contains(@class, 'pane-article-sidebar-latest-news')]
div[contains(@class, 'medstat-accordion-set article-series')]
A tiny patch may someday do your patients’ lab work
A smartwatch can tell a lot about a person’s health, but for guarding against big threats like diabetes and heart disease, blood tests remain the gold standard – for now.
Someday, a wearable patch could give patients and doctors the same information, minus the poke in the arm and the schlep to the medical lab.
The patch will track markers in interstitial fluid.
Continuous glucose monitors have already provided this glimpse into the future, by using interstitial fluid to track blood glucose levels in real time.
Now scientists are asking: What else could this tech help us measure?
“The vision is eventually to develop a lab under the skin,” said Joseph Wang, PhD, professor of nanoengineering at the University of California San Diego.
The result:
How does it work?
Sweat and saliva may be easier to get to, but interstitial fluid is a better mirror for blood. It leaks from tiny blood vessels (capillaries), and it carries nutrients to and removes waste from your skin.
To capture this fluid, each monitor has either a tiny wire or an array of less-than-a-millimeter-long microneedles that penetrate the skin for days, weeks, or however long you wear it. “You don’t feel it,” Dr. Wang said. “Once you place it on the skin, you forget about it.”
The microneedles or wires are made from a polymer that sucks up the fluid, which flows to a biochemical sensor targeting the marker you want to measure.
The earliest patents for this technology date back to the 1990s (the first wearable glucose monitors for home use rolled out in the 2000s), but sensors have come a long way since then, becoming smaller, more accurate, and more sophisticated.
Glucose sensors use an enzyme that reacts to glucose to reveal its concentration in the blood. Researcher Jason Heikenfeld, PhD, and his team at the University of Cincinnati focus on “aptamers,” short single strands of DNA that bind to target molecules. “You can leverage the body’s own ability to generate stuff to grab a needle in a haystack,” he said.
The bigger picture
As our population ages and health care costs spiral, and our medical infrastructure and labor force are stretched thin, we’re seeing a push for decentralized medicine, Dr. Heikenfeld said. Like other at-home monitoring technologies, interstitial fluid sensing promises convenience and better access to care.
“There’s a lot you can do over telemedicine, over the phone,” said Justin T. Baca, MD, PhD, associate professor at the University of New Mexico, Albuquerque. “But we still haven’t figured out how to collect reliable biosamples and analyze them remotely.”
Unlike a traditional blood test, which gives a health snapshot for a single point in time, these devices track data continuously, revealing trends and helping you spot oncoming threats earlier.
Take ketones, for example. Dr. Baca and others are using interstitial fluid to continuously detect ketone levels in the blood, potentially enabling us to catch diabetic ketoacidosis sooner.
“It’s potentially like an early warning sign that somebody needs to get either checked out or get rehydrated or get some insulin; kind of an early diagnostic to avoid hospital visits later on,” Dr. Baca said.
Here’s what else this tech could help us do:
Chronic disease management
Seeing the health impact of medication and diet in real time could motivate patients to stick to their treatment plans, Dr. Heikenfeld said. Researchers in Taiwan are developing a test that could help people with chronic kidney disease track levels of cystatin C, a protein that goes up as kidney function declines. Heart disease patients could watch their cholesterol levels drop over time, and of course, diabetes patients can already track glucose.
Prescription drug monitoring
Providers could monitor drug levels in a patient’s body – like antibiotics for an infection – to see how it’s being metabolized, and adjust the dose as needed, Dr. Heikenfeld said.
Stress and hormone therapy
Interstitial fluid could help us measure hormone levels, such as the stress hormone cortisol.
Scientists in the United Kingdom and Norway developed a waist-worn device that collects interstitial fluid samples continuously for up to 3 days. In their study, samples were sent out for analysis, but someday the device could be equipped with a sensor to monitor a single hormone in real time, said study author Thomas Upton, PhD, a clinical research fellow at the University of Bristol in England. “There is a lot of interest in real-time cortisol monitoring,” he said.
Among those who could benefit: patients with hormone deficiencies, night shift workers with disturbed circadian rhythms, or anyone who wants to keep tabs on their stress response.
Human performance and wellness
Athletes could use glucose and lactate monitors to optimize training, recovery time, and diet. For those on the keto diet, a monitor could help them adjust their carb intake based on their ketone levels. Abbott’s Analyte Ventures group is working on blood alcohol sensors, helpful to anyone who wants to avoid overindulging.
When will this be ready for clinical use?
Early research has been promising, but much more is needed before interstitial fluid sensors can be verified and approved.
Manufacturing will be a challenge. Producing these sensors at scale, without sacrificing consistency or quality, won’t be cheap, said Dr. Heikenfeld. Today’s continuous glucose monitors took decades and hundreds of millions of dollars to develop.
Still, the groundwork has been laid.
“As we all pivot more towards interstitial fluid, there’s a proven roadmap of success that the big diagnostic companies over decades have cut their teeth on,” said Dr. Heikenfeld.
For now, scientists are refining sensors and figuring out how to protect them from other body fluids while in use, Dr. Wang said. But if it all comes together, the result could be game-changing.
Dr. Wang’s lab is developing a system that can monitor glucose and lactate or glucose and alcohol – which could become available in as little as 2 years, he said.
In the next decade, Dr. Wang predicted, we’ll be able to measure a dozen markers with one simple patch.
A version of this article originally appeared on WebMD.com.
A smartwatch can tell a lot about a person’s health, but for guarding against big threats like diabetes and heart disease, blood tests remain the gold standard – for now.
Someday, a wearable patch could give patients and doctors the same information, minus the poke in the arm and the schlep to the medical lab.
The patch will track markers in interstitial fluid.
Continuous glucose monitors have already provided this glimpse into the future, by using interstitial fluid to track blood glucose levels in real time.
Now scientists are asking: What else could this tech help us measure?
“The vision is eventually to develop a lab under the skin,” said Joseph Wang, PhD, professor of nanoengineering at the University of California San Diego.
The result:
How does it work?
Sweat and saliva may be easier to get to, but interstitial fluid is a better mirror for blood. It leaks from tiny blood vessels (capillaries), and it carries nutrients to and removes waste from your skin.
To capture this fluid, each monitor has either a tiny wire or an array of less-than-a-millimeter-long microneedles that penetrate the skin for days, weeks, or however long you wear it. “You don’t feel it,” Dr. Wang said. “Once you place it on the skin, you forget about it.”
The microneedles or wires are made from a polymer that sucks up the fluid, which flows to a biochemical sensor targeting the marker you want to measure.
The earliest patents for this technology date back to the 1990s (the first wearable glucose monitors for home use rolled out in the 2000s), but sensors have come a long way since then, becoming smaller, more accurate, and more sophisticated.
Glucose sensors use an enzyme that reacts to glucose to reveal its concentration in the blood. Researcher Jason Heikenfeld, PhD, and his team at the University of Cincinnati focus on “aptamers,” short single strands of DNA that bind to target molecules. “You can leverage the body’s own ability to generate stuff to grab a needle in a haystack,” he said.
The bigger picture
As our population ages and health care costs spiral, and our medical infrastructure and labor force are stretched thin, we’re seeing a push for decentralized medicine, Dr. Heikenfeld said. Like other at-home monitoring technologies, interstitial fluid sensing promises convenience and better access to care.
“There’s a lot you can do over telemedicine, over the phone,” said Justin T. Baca, MD, PhD, associate professor at the University of New Mexico, Albuquerque. “But we still haven’t figured out how to collect reliable biosamples and analyze them remotely.”
Unlike a traditional blood test, which gives a health snapshot for a single point in time, these devices track data continuously, revealing trends and helping you spot oncoming threats earlier.
Take ketones, for example. Dr. Baca and others are using interstitial fluid to continuously detect ketone levels in the blood, potentially enabling us to catch diabetic ketoacidosis sooner.
“It’s potentially like an early warning sign that somebody needs to get either checked out or get rehydrated or get some insulin; kind of an early diagnostic to avoid hospital visits later on,” Dr. Baca said.
Here’s what else this tech could help us do:
Chronic disease management
Seeing the health impact of medication and diet in real time could motivate patients to stick to their treatment plans, Dr. Heikenfeld said. Researchers in Taiwan are developing a test that could help people with chronic kidney disease track levels of cystatin C, a protein that goes up as kidney function declines. Heart disease patients could watch their cholesterol levels drop over time, and of course, diabetes patients can already track glucose.
Prescription drug monitoring
Providers could monitor drug levels in a patient’s body – like antibiotics for an infection – to see how it’s being metabolized, and adjust the dose as needed, Dr. Heikenfeld said.
Stress and hormone therapy
Interstitial fluid could help us measure hormone levels, such as the stress hormone cortisol.
Scientists in the United Kingdom and Norway developed a waist-worn device that collects interstitial fluid samples continuously for up to 3 days. In their study, samples were sent out for analysis, but someday the device could be equipped with a sensor to monitor a single hormone in real time, said study author Thomas Upton, PhD, a clinical research fellow at the University of Bristol in England. “There is a lot of interest in real-time cortisol monitoring,” he said.
Among those who could benefit: patients with hormone deficiencies, night shift workers with disturbed circadian rhythms, or anyone who wants to keep tabs on their stress response.
Human performance and wellness
Athletes could use glucose and lactate monitors to optimize training, recovery time, and diet. For those on the keto diet, a monitor could help them adjust their carb intake based on their ketone levels. Abbott’s Analyte Ventures group is working on blood alcohol sensors, helpful to anyone who wants to avoid overindulging.
When will this be ready for clinical use?
Early research has been promising, but much more is needed before interstitial fluid sensors can be verified and approved.
Manufacturing will be a challenge. Producing these sensors at scale, without sacrificing consistency or quality, won’t be cheap, said Dr. Heikenfeld. Today’s continuous glucose monitors took decades and hundreds of millions of dollars to develop.
Still, the groundwork has been laid.
“As we all pivot more towards interstitial fluid, there’s a proven roadmap of success that the big diagnostic companies over decades have cut their teeth on,” said Dr. Heikenfeld.
For now, scientists are refining sensors and figuring out how to protect them from other body fluids while in use, Dr. Wang said. But if it all comes together, the result could be game-changing.
Dr. Wang’s lab is developing a system that can monitor glucose and lactate or glucose and alcohol – which could become available in as little as 2 years, he said.
In the next decade, Dr. Wang predicted, we’ll be able to measure a dozen markers with one simple patch.
A version of this article originally appeared on WebMD.com.
A smartwatch can tell a lot about a person’s health, but for guarding against big threats like diabetes and heart disease, blood tests remain the gold standard – for now.
Someday, a wearable patch could give patients and doctors the same information, minus the poke in the arm and the schlep to the medical lab.
The patch will track markers in interstitial fluid.
Continuous glucose monitors have already provided this glimpse into the future, by using interstitial fluid to track blood glucose levels in real time.
Now scientists are asking: What else could this tech help us measure?
“The vision is eventually to develop a lab under the skin,” said Joseph Wang, PhD, professor of nanoengineering at the University of California San Diego.
The result:
How does it work?
Sweat and saliva may be easier to get to, but interstitial fluid is a better mirror for blood. It leaks from tiny blood vessels (capillaries), and it carries nutrients to and removes waste from your skin.
To capture this fluid, each monitor has either a tiny wire or an array of less-than-a-millimeter-long microneedles that penetrate the skin for days, weeks, or however long you wear it. “You don’t feel it,” Dr. Wang said. “Once you place it on the skin, you forget about it.”
The microneedles or wires are made from a polymer that sucks up the fluid, which flows to a biochemical sensor targeting the marker you want to measure.
The earliest patents for this technology date back to the 1990s (the first wearable glucose monitors for home use rolled out in the 2000s), but sensors have come a long way since then, becoming smaller, more accurate, and more sophisticated.
Glucose sensors use an enzyme that reacts to glucose to reveal its concentration in the blood. Researcher Jason Heikenfeld, PhD, and his team at the University of Cincinnati focus on “aptamers,” short single strands of DNA that bind to target molecules. “You can leverage the body’s own ability to generate stuff to grab a needle in a haystack,” he said.
The bigger picture
As our population ages and health care costs spiral, and our medical infrastructure and labor force are stretched thin, we’re seeing a push for decentralized medicine, Dr. Heikenfeld said. Like other at-home monitoring technologies, interstitial fluid sensing promises convenience and better access to care.
“There’s a lot you can do over telemedicine, over the phone,” said Justin T. Baca, MD, PhD, associate professor at the University of New Mexico, Albuquerque. “But we still haven’t figured out how to collect reliable biosamples and analyze them remotely.”
Unlike a traditional blood test, which gives a health snapshot for a single point in time, these devices track data continuously, revealing trends and helping you spot oncoming threats earlier.
Take ketones, for example. Dr. Baca and others are using interstitial fluid to continuously detect ketone levels in the blood, potentially enabling us to catch diabetic ketoacidosis sooner.
“It’s potentially like an early warning sign that somebody needs to get either checked out or get rehydrated or get some insulin; kind of an early diagnostic to avoid hospital visits later on,” Dr. Baca said.
Here’s what else this tech could help us do:
Chronic disease management
Seeing the health impact of medication and diet in real time could motivate patients to stick to their treatment plans, Dr. Heikenfeld said. Researchers in Taiwan are developing a test that could help people with chronic kidney disease track levels of cystatin C, a protein that goes up as kidney function declines. Heart disease patients could watch their cholesterol levels drop over time, and of course, diabetes patients can already track glucose.
Prescription drug monitoring
Providers could monitor drug levels in a patient’s body – like antibiotics for an infection – to see how it’s being metabolized, and adjust the dose as needed, Dr. Heikenfeld said.
Stress and hormone therapy
Interstitial fluid could help us measure hormone levels, such as the stress hormone cortisol.
Scientists in the United Kingdom and Norway developed a waist-worn device that collects interstitial fluid samples continuously for up to 3 days. In their study, samples were sent out for analysis, but someday the device could be equipped with a sensor to monitor a single hormone in real time, said study author Thomas Upton, PhD, a clinical research fellow at the University of Bristol in England. “There is a lot of interest in real-time cortisol monitoring,” he said.
Among those who could benefit: patients with hormone deficiencies, night shift workers with disturbed circadian rhythms, or anyone who wants to keep tabs on their stress response.
Human performance and wellness
Athletes could use glucose and lactate monitors to optimize training, recovery time, and diet. For those on the keto diet, a monitor could help them adjust their carb intake based on their ketone levels. Abbott’s Analyte Ventures group is working on blood alcohol sensors, helpful to anyone who wants to avoid overindulging.
When will this be ready for clinical use?
Early research has been promising, but much more is needed before interstitial fluid sensors can be verified and approved.
Manufacturing will be a challenge. Producing these sensors at scale, without sacrificing consistency or quality, won’t be cheap, said Dr. Heikenfeld. Today’s continuous glucose monitors took decades and hundreds of millions of dollars to develop.
Still, the groundwork has been laid.
“As we all pivot more towards interstitial fluid, there’s a proven roadmap of success that the big diagnostic companies over decades have cut their teeth on,” said Dr. Heikenfeld.
For now, scientists are refining sensors and figuring out how to protect them from other body fluids while in use, Dr. Wang said. But if it all comes together, the result could be game-changing.
Dr. Wang’s lab is developing a system that can monitor glucose and lactate or glucose and alcohol – which could become available in as little as 2 years, he said.
In the next decade, Dr. Wang predicted, we’ll be able to measure a dozen markers with one simple patch.
A version of this article originally appeared on WebMD.com.
Med students, doctor groups react to SCOTUS affirmative action ban
The U.S. Supreme Court ruled on June 29 that using race as a factor in college admissions is unconstitutional, rolling back more than 40 years of affirmative action standards and changing how medical schools evaluate applicants to attract students from diverse backgrounds.
Jesse M. Ehrenfeld, MD, MPH, president of the American Medical Association, said in a prepared statement that the Supreme Court ruling will result in a less diverse physician workforce, which is “bad for health care, bad for medicine, and undermines the health of our nation.” He cited the AMA’s recent adoption of a policy advising medical schools to increase enrollment of people from racial and ethnic groups traditionally underrepresented in medicine – even if that means considering race as a factor in admissions criteria.
“Supporting racial and ethnic diversity in the health professions – spanning classrooms, labs, and clinical settings – enriches the educational experiences of all medical and health professions students and the teaching experiences of faculty, and it is essential to improving the overall health of our nation,” the Association of American Medical Colleges (AAMC) said in a prepared statement.
The American Medical Student Association also denounced the Supreme Court decision. “As future physicians committed to justice and equality, we are profoundly outraged ... We strongly support increased representation of minority students in all levels of education, including colleges and medical schools. By fostering diversity and inclusion, institutions have the power to create more empathetic and inclusive learning environments,” the organization said in a press release.
“Diversity in the health care workforce not only benefits underserved patients but improves care for all patients” by increasing understanding and empathy for people of various cultures, Omar T. Atiq, MD, president of the American College of Physicians, said in a press release.
The Supreme Court ruling stems from a lawsuit by the Students for Fair Admissions against Harvard University and the University of North Carolina. The lawsuit alleges that considering race in the college admission process constitutes discrimination and violates the Equal Protection Clause.
Chief Justice John Roberts, who delivered the court’s decision, stated that an applicant’s personal experiences should carry the most weight in admission decisions and that historically, universities have “wrongly concluded that the touchstone of an individual’s identity is not challenges bested, skills built, or lessons learned, but the color of their skin. Our constitutional history does not tolerate that choice.”
Still, Justice Roberts said the opinion does not prohibit universities from considering how race has affected an applicant’s life, “be it through discrimination, inspiration, or otherwise.”
Diversity in medical schools increased last year, with more Black, Hispanic, and female students applying and enrolling. But continued diversity efforts were expected to prove challenging with affirmative action off the table, according to an amicus brief filed last year by the AMA, the AAMC, and dozens of other professional health care organizations.
The brief supported continued use of race in college admissions, stating that eliminating that factor could slow efforts to achieve greater health equity because fewer doctors would be training and working with colleagues from diverse backgrounds.
Several universities with medical programs, such as Yale and Johns Hopkins universities, filed a separate brief citing similar concerns. After the June 29 decision, Harvard and the University of North Carolina released statements stating they would comply with the ruling.
A version of this article first appeared on Medscape.com.
The U.S. Supreme Court ruled on June 29 that using race as a factor in college admissions is unconstitutional, rolling back more than 40 years of affirmative action standards and changing how medical schools evaluate applicants to attract students from diverse backgrounds.
Jesse M. Ehrenfeld, MD, MPH, president of the American Medical Association, said in a prepared statement that the Supreme Court ruling will result in a less diverse physician workforce, which is “bad for health care, bad for medicine, and undermines the health of our nation.” He cited the AMA’s recent adoption of a policy advising medical schools to increase enrollment of people from racial and ethnic groups traditionally underrepresented in medicine – even if that means considering race as a factor in admissions criteria.
“Supporting racial and ethnic diversity in the health professions – spanning classrooms, labs, and clinical settings – enriches the educational experiences of all medical and health professions students and the teaching experiences of faculty, and it is essential to improving the overall health of our nation,” the Association of American Medical Colleges (AAMC) said in a prepared statement.
The American Medical Student Association also denounced the Supreme Court decision. “As future physicians committed to justice and equality, we are profoundly outraged ... We strongly support increased representation of minority students in all levels of education, including colleges and medical schools. By fostering diversity and inclusion, institutions have the power to create more empathetic and inclusive learning environments,” the organization said in a press release.
“Diversity in the health care workforce not only benefits underserved patients but improves care for all patients” by increasing understanding and empathy for people of various cultures, Omar T. Atiq, MD, president of the American College of Physicians, said in a press release.
The Supreme Court ruling stems from a lawsuit by the Students for Fair Admissions against Harvard University and the University of North Carolina. The lawsuit alleges that considering race in the college admission process constitutes discrimination and violates the Equal Protection Clause.
Chief Justice John Roberts, who delivered the court’s decision, stated that an applicant’s personal experiences should carry the most weight in admission decisions and that historically, universities have “wrongly concluded that the touchstone of an individual’s identity is not challenges bested, skills built, or lessons learned, but the color of their skin. Our constitutional history does not tolerate that choice.”
Still, Justice Roberts said the opinion does not prohibit universities from considering how race has affected an applicant’s life, “be it through discrimination, inspiration, or otherwise.”
Diversity in medical schools increased last year, with more Black, Hispanic, and female students applying and enrolling. But continued diversity efforts were expected to prove challenging with affirmative action off the table, according to an amicus brief filed last year by the AMA, the AAMC, and dozens of other professional health care organizations.
The brief supported continued use of race in college admissions, stating that eliminating that factor could slow efforts to achieve greater health equity because fewer doctors would be training and working with colleagues from diverse backgrounds.
Several universities with medical programs, such as Yale and Johns Hopkins universities, filed a separate brief citing similar concerns. After the June 29 decision, Harvard and the University of North Carolina released statements stating they would comply with the ruling.
A version of this article first appeared on Medscape.com.
The U.S. Supreme Court ruled on June 29 that using race as a factor in college admissions is unconstitutional, rolling back more than 40 years of affirmative action standards and changing how medical schools evaluate applicants to attract students from diverse backgrounds.
Jesse M. Ehrenfeld, MD, MPH, president of the American Medical Association, said in a prepared statement that the Supreme Court ruling will result in a less diverse physician workforce, which is “bad for health care, bad for medicine, and undermines the health of our nation.” He cited the AMA’s recent adoption of a policy advising medical schools to increase enrollment of people from racial and ethnic groups traditionally underrepresented in medicine – even if that means considering race as a factor in admissions criteria.
“Supporting racial and ethnic diversity in the health professions – spanning classrooms, labs, and clinical settings – enriches the educational experiences of all medical and health professions students and the teaching experiences of faculty, and it is essential to improving the overall health of our nation,” the Association of American Medical Colleges (AAMC) said in a prepared statement.
The American Medical Student Association also denounced the Supreme Court decision. “As future physicians committed to justice and equality, we are profoundly outraged ... We strongly support increased representation of minority students in all levels of education, including colleges and medical schools. By fostering diversity and inclusion, institutions have the power to create more empathetic and inclusive learning environments,” the organization said in a press release.
“Diversity in the health care workforce not only benefits underserved patients but improves care for all patients” by increasing understanding and empathy for people of various cultures, Omar T. Atiq, MD, president of the American College of Physicians, said in a press release.
The Supreme Court ruling stems from a lawsuit by the Students for Fair Admissions against Harvard University and the University of North Carolina. The lawsuit alleges that considering race in the college admission process constitutes discrimination and violates the Equal Protection Clause.
Chief Justice John Roberts, who delivered the court’s decision, stated that an applicant’s personal experiences should carry the most weight in admission decisions and that historically, universities have “wrongly concluded that the touchstone of an individual’s identity is not challenges bested, skills built, or lessons learned, but the color of their skin. Our constitutional history does not tolerate that choice.”
Still, Justice Roberts said the opinion does not prohibit universities from considering how race has affected an applicant’s life, “be it through discrimination, inspiration, or otherwise.”
Diversity in medical schools increased last year, with more Black, Hispanic, and female students applying and enrolling. But continued diversity efforts were expected to prove challenging with affirmative action off the table, according to an amicus brief filed last year by the AMA, the AAMC, and dozens of other professional health care organizations.
The brief supported continued use of race in college admissions, stating that eliminating that factor could slow efforts to achieve greater health equity because fewer doctors would be training and working with colleagues from diverse backgrounds.
Several universities with medical programs, such as Yale and Johns Hopkins universities, filed a separate brief citing similar concerns. After the June 29 decision, Harvard and the University of North Carolina released statements stating they would comply with the ruling.
A version of this article first appeared on Medscape.com.
For psoriasis, review finds several biosimilars as safe and effective as biologics
The effectiveness and safety of biosimilars for psoriasis appear to be similar to the originator biologics, reported the authors of a review of studies comparing the two.
“This systematic review found that there was no clinically or statistically significant difference in the efficacy and safety between biosimilars and originators of adalimumab, etanercept, infliximab, and ustekinumab for the treatment of psoriasis,” senior study author and clinical lecturer Zenas Z. N. Yiu, MBChB, PhD, and his colleagues at the University of Manchester, England, wrote in JAMA Dermatology.“The biosimilars evaluated in this study could be considered alongside originators for biologic-naive patients to improve the accessibility of biological treatments,” they added. “Switching patients currently on originators to biosimilars could be considered where clinically appropriate to reduce treatment costs.”
Biologics versus biosimilars
In contrast to most chemically synthesized drugs, biologics are created from living organisms, and they have complex structures that can vary slightly from batch to batch, Luigi Naldi, MD, director of the department of dermatology of Ospedale San Bortolo, Vicenza, Italy, and Antonio Addis, PharmD, researcher in the department of epidemiology, Regione Lazio, in Rome, wrote in an accompanying editorial.
Once the patent on the “originator” biologic expires, U.S. and European regulators allow other manufacturers to develop similar molecules – biosimilars – through an abbreviated approval process. If the results of a limited number of equivalence or noninferiority clinical trials are acceptable, registration for all the indications of the originator is allowed for its biosimilars. Referring to the expense of biologics, Dr. Naldi and Dr. Addis noted that in the United States, “biologics comprise less than 3% of the volume of drugs on the market, but account for more than one-third of all drug spending.”
Systematic review
Dr. Yiu and his colleagues queried standard medical research databases in August 2022, and included 14 randomized clinical trials (10 adalimumab, 2 etanercept, 1 infliximab, and 1 ustekinumab) and 3 cohort studies (1 adalimumab, 1 etanercept, 1 infliximab and etanercept) in their review.
Twelve trials compared biosimilars vs. originators in originator-naive patients, and 11 trials compared switching from originators to biosimilars vs. continuous treatment with the originator.
The researchers found the following:
At week 16, mean PASI75 (Psoriasis Area and Severity Index) response rates ranges from 60.7% to 90.6% for adalimumab biosimilars, vs. 61.5% to 91.7% for the originator. Mean PASI75 responses for the two etanercept biosimilars were 56.1% and 76.7% vs. 55.5% and 73.4% for the originator. In the ustekinumab study, mean PASI75 responses were 86.1% for the biosimilar vs. 84.0% for the originator.
At week 52, mean PASI75 responses were between 86.3% and 92.8% for adalimumab biosimilars vs. 84.9% and 93.9% for the originator. In the one comparison of an etanercept biosimilar, mean PAS175 responses were 80.9% for the biosimilar vs. 82.9% for the originator.
In studies involving patients switching from the originator to a biosimilar vs. continuing treatment with the originator, 32-week response rates ranged from 87.0% to 91.3% for adalimumab biosimilars and from 88.2% to 93.2% for the originator. In the one ustekinumab study, the 32-week mean PASI75 response was 92.6% after switching from the originator to a biosimilar vs. 92.9% with continuous treatment with the originator.
At week 52, mean PASI75 responses to adalimumab were between 84.2% and 94.8% for patients who switched to biosimilars and between 88.1% and 93.9% for those who stayed on the originator.
At week 52, in all the randomized trials, the incidence of adverse events and serious adverse events among those who switched to the biosimilar and those who continued with the originator were similar. Two cohort studies showed similar safety outcomes between originators and biosimilars, but one reported more adverse events in patients who switched to adalimumab biosimilars (P = .04).
Three clinical trials showed low risk for bias, 11 had moderate risk, and all cohort studies had moderate to high risk for bias.
Experts weigh in
Asked to comment on the study, Steven R. Feldman, MD, PhD, professor of dermatology at Wake Forest University, Winston-Salem, N.C., told this news organization that he expects that the results will affect patient care.
However, he added, “I believe the decision of whether to use a biosimilar instead of the originator biologic may be more in the hands of the insurers than in the hands of physicians and patients.
“Biologics for psoriasis are so complicated that even the originator products vary from batch to batch. A biosimilar is basically like another batch of the innovative product,” explained Dr. Feldman, who was not involved in the study. “If we’re comfortable with patients being on different batches of the innovator product, we probably should be comfortable with them being on a biosimilar, as we have more evidence for the similarity of the biosimilar than we do for the current batch of the originator product.”
Aída Lugo-Somolinos, MD, professor of dermatology and director of the Contact Dermatitis Clinic at the University of North Carolina, Chapel Hill, said that “biologics have become the treatment of choice for moderate to severe psoriasis, and the use of biosimilars may be an alternative to reduce psoriasis treatment costs.
“Unfortunately, this study included a comparison of the existing biosimilars, which are drugs that are not the first line of treatment for psoriasis any longer,” added Dr. Lugo-Somolinos, who was not involved in the study.
Neil J. Korman, MD, PhD, professor of dermatology and codirector of the Skin Study Center at Case Western Reserve University, Cleveland, said the study was an important systematic review.
“This is a very timely publication because in the United States, several biosimilars are reaching the market in 2023,” he said. “The costs of the originator biologics are extraordinarily high, and the promise of biosimilars is that their costs will be significantly lower.”
Because all the studies were short term, Dr. Korman, who was not involved in the study, joins the study authors in recommending further related research into the long-term safety and efficacy of these agents.
Dr. Feldman, as well as one study author and one editorial author, reported relevant relationships with various pharmaceutical companies, including those that develop biosimilars. The remaining study authors, as well as Dr. Lugo-Somolinos and Dr. Korman, reported no relevant relationships. The study was funded by the Psoriasis Association and supported by the NIHR (National Institute for Health and Care Research) Manchester Biomedical Research Centre. All outside experts commented by email.
The effectiveness and safety of biosimilars for psoriasis appear to be similar to the originator biologics, reported the authors of a review of studies comparing the two.
“This systematic review found that there was no clinically or statistically significant difference in the efficacy and safety between biosimilars and originators of adalimumab, etanercept, infliximab, and ustekinumab for the treatment of psoriasis,” senior study author and clinical lecturer Zenas Z. N. Yiu, MBChB, PhD, and his colleagues at the University of Manchester, England, wrote in JAMA Dermatology.“The biosimilars evaluated in this study could be considered alongside originators for biologic-naive patients to improve the accessibility of biological treatments,” they added. “Switching patients currently on originators to biosimilars could be considered where clinically appropriate to reduce treatment costs.”
Biologics versus biosimilars
In contrast to most chemically synthesized drugs, biologics are created from living organisms, and they have complex structures that can vary slightly from batch to batch, Luigi Naldi, MD, director of the department of dermatology of Ospedale San Bortolo, Vicenza, Italy, and Antonio Addis, PharmD, researcher in the department of epidemiology, Regione Lazio, in Rome, wrote in an accompanying editorial.
Once the patent on the “originator” biologic expires, U.S. and European regulators allow other manufacturers to develop similar molecules – biosimilars – through an abbreviated approval process. If the results of a limited number of equivalence or noninferiority clinical trials are acceptable, registration for all the indications of the originator is allowed for its biosimilars. Referring to the expense of biologics, Dr. Naldi and Dr. Addis noted that in the United States, “biologics comprise less than 3% of the volume of drugs on the market, but account for more than one-third of all drug spending.”
Systematic review
Dr. Yiu and his colleagues queried standard medical research databases in August 2022, and included 14 randomized clinical trials (10 adalimumab, 2 etanercept, 1 infliximab, and 1 ustekinumab) and 3 cohort studies (1 adalimumab, 1 etanercept, 1 infliximab and etanercept) in their review.
Twelve trials compared biosimilars vs. originators in originator-naive patients, and 11 trials compared switching from originators to biosimilars vs. continuous treatment with the originator.
The researchers found the following:
At week 16, mean PASI75 (Psoriasis Area and Severity Index) response rates ranges from 60.7% to 90.6% for adalimumab biosimilars, vs. 61.5% to 91.7% for the originator. Mean PASI75 responses for the two etanercept biosimilars were 56.1% and 76.7% vs. 55.5% and 73.4% for the originator. In the ustekinumab study, mean PASI75 responses were 86.1% for the biosimilar vs. 84.0% for the originator.
At week 52, mean PASI75 responses were between 86.3% and 92.8% for adalimumab biosimilars vs. 84.9% and 93.9% for the originator. In the one comparison of an etanercept biosimilar, mean PAS175 responses were 80.9% for the biosimilar vs. 82.9% for the originator.
In studies involving patients switching from the originator to a biosimilar vs. continuing treatment with the originator, 32-week response rates ranged from 87.0% to 91.3% for adalimumab biosimilars and from 88.2% to 93.2% for the originator. In the one ustekinumab study, the 32-week mean PASI75 response was 92.6% after switching from the originator to a biosimilar vs. 92.9% with continuous treatment with the originator.
At week 52, mean PASI75 responses to adalimumab were between 84.2% and 94.8% for patients who switched to biosimilars and between 88.1% and 93.9% for those who stayed on the originator.
At week 52, in all the randomized trials, the incidence of adverse events and serious adverse events among those who switched to the biosimilar and those who continued with the originator were similar. Two cohort studies showed similar safety outcomes between originators and biosimilars, but one reported more adverse events in patients who switched to adalimumab biosimilars (P = .04).
Three clinical trials showed low risk for bias, 11 had moderate risk, and all cohort studies had moderate to high risk for bias.
Experts weigh in
Asked to comment on the study, Steven R. Feldman, MD, PhD, professor of dermatology at Wake Forest University, Winston-Salem, N.C., told this news organization that he expects that the results will affect patient care.
However, he added, “I believe the decision of whether to use a biosimilar instead of the originator biologic may be more in the hands of the insurers than in the hands of physicians and patients.
“Biologics for psoriasis are so complicated that even the originator products vary from batch to batch. A biosimilar is basically like another batch of the innovative product,” explained Dr. Feldman, who was not involved in the study. “If we’re comfortable with patients being on different batches of the innovator product, we probably should be comfortable with them being on a biosimilar, as we have more evidence for the similarity of the biosimilar than we do for the current batch of the originator product.”
Aída Lugo-Somolinos, MD, professor of dermatology and director of the Contact Dermatitis Clinic at the University of North Carolina, Chapel Hill, said that “biologics have become the treatment of choice for moderate to severe psoriasis, and the use of biosimilars may be an alternative to reduce psoriasis treatment costs.
“Unfortunately, this study included a comparison of the existing biosimilars, which are drugs that are not the first line of treatment for psoriasis any longer,” added Dr. Lugo-Somolinos, who was not involved in the study.
Neil J. Korman, MD, PhD, professor of dermatology and codirector of the Skin Study Center at Case Western Reserve University, Cleveland, said the study was an important systematic review.
“This is a very timely publication because in the United States, several biosimilars are reaching the market in 2023,” he said. “The costs of the originator biologics are extraordinarily high, and the promise of biosimilars is that their costs will be significantly lower.”
Because all the studies were short term, Dr. Korman, who was not involved in the study, joins the study authors in recommending further related research into the long-term safety and efficacy of these agents.
Dr. Feldman, as well as one study author and one editorial author, reported relevant relationships with various pharmaceutical companies, including those that develop biosimilars. The remaining study authors, as well as Dr. Lugo-Somolinos and Dr. Korman, reported no relevant relationships. The study was funded by the Psoriasis Association and supported by the NIHR (National Institute for Health and Care Research) Manchester Biomedical Research Centre. All outside experts commented by email.
The effectiveness and safety of biosimilars for psoriasis appear to be similar to the originator biologics, reported the authors of a review of studies comparing the two.
“This systematic review found that there was no clinically or statistically significant difference in the efficacy and safety between biosimilars and originators of adalimumab, etanercept, infliximab, and ustekinumab for the treatment of psoriasis,” senior study author and clinical lecturer Zenas Z. N. Yiu, MBChB, PhD, and his colleagues at the University of Manchester, England, wrote in JAMA Dermatology.“The biosimilars evaluated in this study could be considered alongside originators for biologic-naive patients to improve the accessibility of biological treatments,” they added. “Switching patients currently on originators to biosimilars could be considered where clinically appropriate to reduce treatment costs.”
Biologics versus biosimilars
In contrast to most chemically synthesized drugs, biologics are created from living organisms, and they have complex structures that can vary slightly from batch to batch, Luigi Naldi, MD, director of the department of dermatology of Ospedale San Bortolo, Vicenza, Italy, and Antonio Addis, PharmD, researcher in the department of epidemiology, Regione Lazio, in Rome, wrote in an accompanying editorial.
Once the patent on the “originator” biologic expires, U.S. and European regulators allow other manufacturers to develop similar molecules – biosimilars – through an abbreviated approval process. If the results of a limited number of equivalence or noninferiority clinical trials are acceptable, registration for all the indications of the originator is allowed for its biosimilars. Referring to the expense of biologics, Dr. Naldi and Dr. Addis noted that in the United States, “biologics comprise less than 3% of the volume of drugs on the market, but account for more than one-third of all drug spending.”
Systematic review
Dr. Yiu and his colleagues queried standard medical research databases in August 2022, and included 14 randomized clinical trials (10 adalimumab, 2 etanercept, 1 infliximab, and 1 ustekinumab) and 3 cohort studies (1 adalimumab, 1 etanercept, 1 infliximab and etanercept) in their review.
Twelve trials compared biosimilars vs. originators in originator-naive patients, and 11 trials compared switching from originators to biosimilars vs. continuous treatment with the originator.
The researchers found the following:
At week 16, mean PASI75 (Psoriasis Area and Severity Index) response rates ranges from 60.7% to 90.6% for adalimumab biosimilars, vs. 61.5% to 91.7% for the originator. Mean PASI75 responses for the two etanercept biosimilars were 56.1% and 76.7% vs. 55.5% and 73.4% for the originator. In the ustekinumab study, mean PASI75 responses were 86.1% for the biosimilar vs. 84.0% for the originator.
At week 52, mean PASI75 responses were between 86.3% and 92.8% for adalimumab biosimilars vs. 84.9% and 93.9% for the originator. In the one comparison of an etanercept biosimilar, mean PAS175 responses were 80.9% for the biosimilar vs. 82.9% for the originator.
In studies involving patients switching from the originator to a biosimilar vs. continuing treatment with the originator, 32-week response rates ranged from 87.0% to 91.3% for adalimumab biosimilars and from 88.2% to 93.2% for the originator. In the one ustekinumab study, the 32-week mean PASI75 response was 92.6% after switching from the originator to a biosimilar vs. 92.9% with continuous treatment with the originator.
At week 52, mean PASI75 responses to adalimumab were between 84.2% and 94.8% for patients who switched to biosimilars and between 88.1% and 93.9% for those who stayed on the originator.
At week 52, in all the randomized trials, the incidence of adverse events and serious adverse events among those who switched to the biosimilar and those who continued with the originator were similar. Two cohort studies showed similar safety outcomes between originators and biosimilars, but one reported more adverse events in patients who switched to adalimumab biosimilars (P = .04).
Three clinical trials showed low risk for bias, 11 had moderate risk, and all cohort studies had moderate to high risk for bias.
Experts weigh in
Asked to comment on the study, Steven R. Feldman, MD, PhD, professor of dermatology at Wake Forest University, Winston-Salem, N.C., told this news organization that he expects that the results will affect patient care.
However, he added, “I believe the decision of whether to use a biosimilar instead of the originator biologic may be more in the hands of the insurers than in the hands of physicians and patients.
“Biologics for psoriasis are so complicated that even the originator products vary from batch to batch. A biosimilar is basically like another batch of the innovative product,” explained Dr. Feldman, who was not involved in the study. “If we’re comfortable with patients being on different batches of the innovator product, we probably should be comfortable with them being on a biosimilar, as we have more evidence for the similarity of the biosimilar than we do for the current batch of the originator product.”
Aída Lugo-Somolinos, MD, professor of dermatology and director of the Contact Dermatitis Clinic at the University of North Carolina, Chapel Hill, said that “biologics have become the treatment of choice for moderate to severe psoriasis, and the use of biosimilars may be an alternative to reduce psoriasis treatment costs.
“Unfortunately, this study included a comparison of the existing biosimilars, which are drugs that are not the first line of treatment for psoriasis any longer,” added Dr. Lugo-Somolinos, who was not involved in the study.
Neil J. Korman, MD, PhD, professor of dermatology and codirector of the Skin Study Center at Case Western Reserve University, Cleveland, said the study was an important systematic review.
“This is a very timely publication because in the United States, several biosimilars are reaching the market in 2023,” he said. “The costs of the originator biologics are extraordinarily high, and the promise of biosimilars is that their costs will be significantly lower.”
Because all the studies were short term, Dr. Korman, who was not involved in the study, joins the study authors in recommending further related research into the long-term safety and efficacy of these agents.
Dr. Feldman, as well as one study author and one editorial author, reported relevant relationships with various pharmaceutical companies, including those that develop biosimilars. The remaining study authors, as well as Dr. Lugo-Somolinos and Dr. Korman, reported no relevant relationships. The study was funded by the Psoriasis Association and supported by the NIHR (National Institute for Health and Care Research) Manchester Biomedical Research Centre. All outside experts commented by email.
FROM JAMA DERMATOLOGY
Does colchicine have a role in treating excess ASCVD risk in patients with chronic inflammatory conditions?
The recent Food and Drug Administration approval of colchicine 0.5 mg (Lodoco) for use in atherosclerotic cardiovascular disease (ASCVD) prevention will possibly create opportunities to use the drug to treat residual risk for ASCVD in some patients with immune-mediated inflammatory diseases, particularly in rheumatology.
Potential in rheumatology
The 0.5-mg dose is just a shade under the 0.6-mg, twice daily dosing rheumatologists typically prescribe for gout, Christie Bartels, MD, MS, chief of rheumatology at the University of Wisconsin–Madison, said in an interview. Clinicians also use the 0.6-mg dose off-label for pseudogout or calcium pyrophosphate deposition disease (CPPD), Dr. Bartels noted.
The new formulation opens the consideration for using colchicine more in patients with psoriatic arthritis, lupus, and rheumatoid arthritis, she said. “I think we could certainly discuss it, particularly, in secondary prevention patients who already had an event or who are at the highest risk and already on optimal traditional agents,” she said.
She cited previous comments by Paul Ridker, MD, director of the center for cardiovascular disease prevention at Brigham and Women’s Hospital in Boston, and developer of the high-sensitivity C-reactive protein (hsCRP) test for measuring inflammatory markers. “We might not know the answer because Dr. Ridker pointed out he used colchicine 0.5 mg in patients that had a high-sensitivity CRP that was high; we need patients who have had inflammation of unknown origin, so those patients presumably weren’t already on another anti-inflammatory,” she said, noting that hydroxychloroquine, methotrexate, and some biologics provide some protection from cardiovascular risks.
However, a potential role for long-term colchicine 0.5 mg in ASCVD prevention may cause consideration for changing the drug’s role in gout treatment, Dr. Bartels said. “In gout, where we do have an FDA-approved indication for colchicine, we used to use it only for the first 6 months while we were getting patients to goal on allopurinol, which was usually then monotherapy after the first 6 months,” she said. “I think this will likely change how I treat gout patients in that I may also offer to continue both medications [colchicine and allopurinol] if they are tolerating them well.
“And then in patients where I’m using it off-label in CPPD, I might again share with them that in addition to possibly helping their CPPD, there may be this added benefit to reduce inflammation just in discussing the risks and benefits of the medicine.”
However, rheumatologists must be careful in using colchicine beyond the typical 6-month cycle, Dr. Bartels said. “One of the tricky things with colchicine, and part of the reason we did not traditionally continue it specifically past the first 6 months, was that it can cause myopathies or cytopenias, so we still have to counsel patients regarding these risks and monitor that,” she said.
Additionally, colchicine can have drug interactions with statins or calcium channel blockers that can change colchicine levels. “I think the dose here is so low, the 0.5 mg, that it’s probably still safe, but again, it’s something that we have to take a look at in the patient’s whole picture and the rest of their burden of their meds in order to make a decision with them,” Dr. Bartels said.
Possibilities in dermatology
The LoDoCo2 trial one of two major randomized trials that supported approval of colchicine 0.5 mg, reported that treated patients had a 60% lower rate of gout than the placebo group (1.4% vs. 3.4%). Joel Gelfand, MD, MSCE, the James J. Leyden professor of dermatology and epidemiology at the University of Pennsylvania, Philadelphia, pointed to this in evaluating the dermatologic implications of the drug’s approval. “This may be of particular interest as people with psoriasis have an increased risk of gout,” he said in emailed comments.
Colchicine’s mechanism of action to reduce inflammation parallels that of tumor necrosis factor (TNF) inhibitors used for dermatologic indications, namely by inhibiting leukocyte adhesion to disrupt the downregulation of TNF receptors, Dr. Gelfand said.
“Interestingly, observational data suggests biologics that target TNF such as adalimumab, etanercept, etc., are associated with a reduction in CV events, and in placebo-controlled trials we conducted in psoriasis patients, it reduced key inflammatory mediators of cardiovascular disease, including IL [interleukin]-6,” he said. “Randomized clinical trials to evaluate the ability of TNF inhibitors, which are now available as biosimilars, to prevent cardiovascular events in high-risk patients, should be conducted, and more work is needed to identify which additional immune-targeted treatments may lower CV risk with an acceptable safety profile.”
Colchicine currently has few indications for rare conditions in dermatology, Dr. Gelfand said, including Sweets syndrome, subcorneal pustular dermatosis, and cutaneous vasculitis. “There are some reports to suggest it may help psoriatic disease, but current data are limited and insufficient to recommend its use for psoriasis and/or psoriatic arthritis,” he said.
The approval of colchicine 0.5 mg for ASCVD could be meaningful for people with psoriasis who are also being treated for CV risk factors, Dr. Gelfand said. “Additional considerations such as signs of residual inflammation (elevated hsCRP) and CV imaging findings may be used to further guide shared decision-making for optimal use,” he said.
Another consideration he noted: “This is also a novel 0.5-mg formulation, and thus cost may be an issue.”
Would side effects bar use in gastroenterology?
Colchicine 0.5 mg may not move the needle much for expanding treatment of ASCVD in patients with inflammatory bowel disease (IBD) and potentially other gastrointestinal conditions, Edward Loftus Jr., MD, the Maxine and Jack Zarrow Family professor of gastroenterology specifically for IBD at the Mayo Clinic in Rochester, Minn., told MDEdge in emailed comments. “Given the GI side effect profile [of colchicine], I am not sure I would go there,” he said.
“Hopefully, the prescribers of this low-dose formulation are aware of the gastrointestinal side effects, such as diarrhea and nausea, and educate patients about these side effects so that a proper risk-benefit discussion can ensue,” he said.
Dr. Bartels reporting a previous financial relationship with Pfizer. Dr. Gelfand said he has financial relationships with AbbVie, Amgen, Bristol-Myers Squibb, Boehringer Ingelheim, Celldex, GlaxoSmithKline, Twill, Lilly, Leo, Moonlake, Janssen Biologics, Novartis, Pfizer, UCB, Neuroderm, and Veolia North America. Dr. Loftus disclosed relationships with AbbVie, Alvotech, Amgen, Arena, Avalo, Boehringer Ingelheim, Bristol-Myers Squibb, Celgene/Receptos, Celltrion Healthcare, Eli Lilly, Fresenius Kabi, Genentech, Gilead, GlaxoSmithKline, Gossamer Bio, Iterative Health, Janssen, KSL Diagnostics, Morphic, Ono, Pfizer, Sun, Surrozen, Takeda, Theravance, and UCB.
The recent Food and Drug Administration approval of colchicine 0.5 mg (Lodoco) for use in atherosclerotic cardiovascular disease (ASCVD) prevention will possibly create opportunities to use the drug to treat residual risk for ASCVD in some patients with immune-mediated inflammatory diseases, particularly in rheumatology.
Potential in rheumatology
The 0.5-mg dose is just a shade under the 0.6-mg, twice daily dosing rheumatologists typically prescribe for gout, Christie Bartels, MD, MS, chief of rheumatology at the University of Wisconsin–Madison, said in an interview. Clinicians also use the 0.6-mg dose off-label for pseudogout or calcium pyrophosphate deposition disease (CPPD), Dr. Bartels noted.
The new formulation opens the consideration for using colchicine more in patients with psoriatic arthritis, lupus, and rheumatoid arthritis, she said. “I think we could certainly discuss it, particularly, in secondary prevention patients who already had an event or who are at the highest risk and already on optimal traditional agents,” she said.
She cited previous comments by Paul Ridker, MD, director of the center for cardiovascular disease prevention at Brigham and Women’s Hospital in Boston, and developer of the high-sensitivity C-reactive protein (hsCRP) test for measuring inflammatory markers. “We might not know the answer because Dr. Ridker pointed out he used colchicine 0.5 mg in patients that had a high-sensitivity CRP that was high; we need patients who have had inflammation of unknown origin, so those patients presumably weren’t already on another anti-inflammatory,” she said, noting that hydroxychloroquine, methotrexate, and some biologics provide some protection from cardiovascular risks.
However, a potential role for long-term colchicine 0.5 mg in ASCVD prevention may cause consideration for changing the drug’s role in gout treatment, Dr. Bartels said. “In gout, where we do have an FDA-approved indication for colchicine, we used to use it only for the first 6 months while we were getting patients to goal on allopurinol, which was usually then monotherapy after the first 6 months,” she said. “I think this will likely change how I treat gout patients in that I may also offer to continue both medications [colchicine and allopurinol] if they are tolerating them well.
“And then in patients where I’m using it off-label in CPPD, I might again share with them that in addition to possibly helping their CPPD, there may be this added benefit to reduce inflammation just in discussing the risks and benefits of the medicine.”
However, rheumatologists must be careful in using colchicine beyond the typical 6-month cycle, Dr. Bartels said. “One of the tricky things with colchicine, and part of the reason we did not traditionally continue it specifically past the first 6 months, was that it can cause myopathies or cytopenias, so we still have to counsel patients regarding these risks and monitor that,” she said.
Additionally, colchicine can have drug interactions with statins or calcium channel blockers that can change colchicine levels. “I think the dose here is so low, the 0.5 mg, that it’s probably still safe, but again, it’s something that we have to take a look at in the patient’s whole picture and the rest of their burden of their meds in order to make a decision with them,” Dr. Bartels said.
Possibilities in dermatology
The LoDoCo2 trial one of two major randomized trials that supported approval of colchicine 0.5 mg, reported that treated patients had a 60% lower rate of gout than the placebo group (1.4% vs. 3.4%). Joel Gelfand, MD, MSCE, the James J. Leyden professor of dermatology and epidemiology at the University of Pennsylvania, Philadelphia, pointed to this in evaluating the dermatologic implications of the drug’s approval. “This may be of particular interest as people with psoriasis have an increased risk of gout,” he said in emailed comments.
Colchicine’s mechanism of action to reduce inflammation parallels that of tumor necrosis factor (TNF) inhibitors used for dermatologic indications, namely by inhibiting leukocyte adhesion to disrupt the downregulation of TNF receptors, Dr. Gelfand said.
“Interestingly, observational data suggests biologics that target TNF such as adalimumab, etanercept, etc., are associated with a reduction in CV events, and in placebo-controlled trials we conducted in psoriasis patients, it reduced key inflammatory mediators of cardiovascular disease, including IL [interleukin]-6,” he said. “Randomized clinical trials to evaluate the ability of TNF inhibitors, which are now available as biosimilars, to prevent cardiovascular events in high-risk patients, should be conducted, and more work is needed to identify which additional immune-targeted treatments may lower CV risk with an acceptable safety profile.”
Colchicine currently has few indications for rare conditions in dermatology, Dr. Gelfand said, including Sweets syndrome, subcorneal pustular dermatosis, and cutaneous vasculitis. “There are some reports to suggest it may help psoriatic disease, but current data are limited and insufficient to recommend its use for psoriasis and/or psoriatic arthritis,” he said.
The approval of colchicine 0.5 mg for ASCVD could be meaningful for people with psoriasis who are also being treated for CV risk factors, Dr. Gelfand said. “Additional considerations such as signs of residual inflammation (elevated hsCRP) and CV imaging findings may be used to further guide shared decision-making for optimal use,” he said.
Another consideration he noted: “This is also a novel 0.5-mg formulation, and thus cost may be an issue.”
Would side effects bar use in gastroenterology?
Colchicine 0.5 mg may not move the needle much for expanding treatment of ASCVD in patients with inflammatory bowel disease (IBD) and potentially other gastrointestinal conditions, Edward Loftus Jr., MD, the Maxine and Jack Zarrow Family professor of gastroenterology specifically for IBD at the Mayo Clinic in Rochester, Minn., told MDEdge in emailed comments. “Given the GI side effect profile [of colchicine], I am not sure I would go there,” he said.
“Hopefully, the prescribers of this low-dose formulation are aware of the gastrointestinal side effects, such as diarrhea and nausea, and educate patients about these side effects so that a proper risk-benefit discussion can ensue,” he said.
Dr. Bartels reporting a previous financial relationship with Pfizer. Dr. Gelfand said he has financial relationships with AbbVie, Amgen, Bristol-Myers Squibb, Boehringer Ingelheim, Celldex, GlaxoSmithKline, Twill, Lilly, Leo, Moonlake, Janssen Biologics, Novartis, Pfizer, UCB, Neuroderm, and Veolia North America. Dr. Loftus disclosed relationships with AbbVie, Alvotech, Amgen, Arena, Avalo, Boehringer Ingelheim, Bristol-Myers Squibb, Celgene/Receptos, Celltrion Healthcare, Eli Lilly, Fresenius Kabi, Genentech, Gilead, GlaxoSmithKline, Gossamer Bio, Iterative Health, Janssen, KSL Diagnostics, Morphic, Ono, Pfizer, Sun, Surrozen, Takeda, Theravance, and UCB.
The recent Food and Drug Administration approval of colchicine 0.5 mg (Lodoco) for use in atherosclerotic cardiovascular disease (ASCVD) prevention will possibly create opportunities to use the drug to treat residual risk for ASCVD in some patients with immune-mediated inflammatory diseases, particularly in rheumatology.
Potential in rheumatology
The 0.5-mg dose is just a shade under the 0.6-mg, twice daily dosing rheumatologists typically prescribe for gout, Christie Bartels, MD, MS, chief of rheumatology at the University of Wisconsin–Madison, said in an interview. Clinicians also use the 0.6-mg dose off-label for pseudogout or calcium pyrophosphate deposition disease (CPPD), Dr. Bartels noted.
The new formulation opens the consideration for using colchicine more in patients with psoriatic arthritis, lupus, and rheumatoid arthritis, she said. “I think we could certainly discuss it, particularly, in secondary prevention patients who already had an event or who are at the highest risk and already on optimal traditional agents,” she said.
She cited previous comments by Paul Ridker, MD, director of the center for cardiovascular disease prevention at Brigham and Women’s Hospital in Boston, and developer of the high-sensitivity C-reactive protein (hsCRP) test for measuring inflammatory markers. “We might not know the answer because Dr. Ridker pointed out he used colchicine 0.5 mg in patients that had a high-sensitivity CRP that was high; we need patients who have had inflammation of unknown origin, so those patients presumably weren’t already on another anti-inflammatory,” she said, noting that hydroxychloroquine, methotrexate, and some biologics provide some protection from cardiovascular risks.
However, a potential role for long-term colchicine 0.5 mg in ASCVD prevention may cause consideration for changing the drug’s role in gout treatment, Dr. Bartels said. “In gout, where we do have an FDA-approved indication for colchicine, we used to use it only for the first 6 months while we were getting patients to goal on allopurinol, which was usually then monotherapy after the first 6 months,” she said. “I think this will likely change how I treat gout patients in that I may also offer to continue both medications [colchicine and allopurinol] if they are tolerating them well.
“And then in patients where I’m using it off-label in CPPD, I might again share with them that in addition to possibly helping their CPPD, there may be this added benefit to reduce inflammation just in discussing the risks and benefits of the medicine.”
However, rheumatologists must be careful in using colchicine beyond the typical 6-month cycle, Dr. Bartels said. “One of the tricky things with colchicine, and part of the reason we did not traditionally continue it specifically past the first 6 months, was that it can cause myopathies or cytopenias, so we still have to counsel patients regarding these risks and monitor that,” she said.
Additionally, colchicine can have drug interactions with statins or calcium channel blockers that can change colchicine levels. “I think the dose here is so low, the 0.5 mg, that it’s probably still safe, but again, it’s something that we have to take a look at in the patient’s whole picture and the rest of their burden of their meds in order to make a decision with them,” Dr. Bartels said.
Possibilities in dermatology
The LoDoCo2 trial one of two major randomized trials that supported approval of colchicine 0.5 mg, reported that treated patients had a 60% lower rate of gout than the placebo group (1.4% vs. 3.4%). Joel Gelfand, MD, MSCE, the James J. Leyden professor of dermatology and epidemiology at the University of Pennsylvania, Philadelphia, pointed to this in evaluating the dermatologic implications of the drug’s approval. “This may be of particular interest as people with psoriasis have an increased risk of gout,” he said in emailed comments.
Colchicine’s mechanism of action to reduce inflammation parallels that of tumor necrosis factor (TNF) inhibitors used for dermatologic indications, namely by inhibiting leukocyte adhesion to disrupt the downregulation of TNF receptors, Dr. Gelfand said.
“Interestingly, observational data suggests biologics that target TNF such as adalimumab, etanercept, etc., are associated with a reduction in CV events, and in placebo-controlled trials we conducted in psoriasis patients, it reduced key inflammatory mediators of cardiovascular disease, including IL [interleukin]-6,” he said. “Randomized clinical trials to evaluate the ability of TNF inhibitors, which are now available as biosimilars, to prevent cardiovascular events in high-risk patients, should be conducted, and more work is needed to identify which additional immune-targeted treatments may lower CV risk with an acceptable safety profile.”
Colchicine currently has few indications for rare conditions in dermatology, Dr. Gelfand said, including Sweets syndrome, subcorneal pustular dermatosis, and cutaneous vasculitis. “There are some reports to suggest it may help psoriatic disease, but current data are limited and insufficient to recommend its use for psoriasis and/or psoriatic arthritis,” he said.
The approval of colchicine 0.5 mg for ASCVD could be meaningful for people with psoriasis who are also being treated for CV risk factors, Dr. Gelfand said. “Additional considerations such as signs of residual inflammation (elevated hsCRP) and CV imaging findings may be used to further guide shared decision-making for optimal use,” he said.
Another consideration he noted: “This is also a novel 0.5-mg formulation, and thus cost may be an issue.”
Would side effects bar use in gastroenterology?
Colchicine 0.5 mg may not move the needle much for expanding treatment of ASCVD in patients with inflammatory bowel disease (IBD) and potentially other gastrointestinal conditions, Edward Loftus Jr., MD, the Maxine and Jack Zarrow Family professor of gastroenterology specifically for IBD at the Mayo Clinic in Rochester, Minn., told MDEdge in emailed comments. “Given the GI side effect profile [of colchicine], I am not sure I would go there,” he said.
“Hopefully, the prescribers of this low-dose formulation are aware of the gastrointestinal side effects, such as diarrhea and nausea, and educate patients about these side effects so that a proper risk-benefit discussion can ensue,” he said.
Dr. Bartels reporting a previous financial relationship with Pfizer. Dr. Gelfand said he has financial relationships with AbbVie, Amgen, Bristol-Myers Squibb, Boehringer Ingelheim, Celldex, GlaxoSmithKline, Twill, Lilly, Leo, Moonlake, Janssen Biologics, Novartis, Pfizer, UCB, Neuroderm, and Veolia North America. Dr. Loftus disclosed relationships with AbbVie, Alvotech, Amgen, Arena, Avalo, Boehringer Ingelheim, Bristol-Myers Squibb, Celgene/Receptos, Celltrion Healthcare, Eli Lilly, Fresenius Kabi, Genentech, Gilead, GlaxoSmithKline, Gossamer Bio, Iterative Health, Janssen, KSL Diagnostics, Morphic, Ono, Pfizer, Sun, Surrozen, Takeda, Theravance, and UCB.
Commentary: DMARD and HCQ in RA, July 2023
Despite multiple existing conventional synthetic disease-modifying antirheumatic drug (csDMARD) and biologic DMARD (bDMARD) options, many patients with rheumatoid arthritis (RA) do not respond adequately to treatment. In an exciting development, a recent phase 2 study by Tuttle and colleagues examined a novel treatment approach in RA: stimulation of the programmed cell death protein 1 (PD-1) inhibitor pathway. PD-1 is a checkpoint inhibitor receptor whose activation reflects T-cell activation and may play a role in synovitis and extra-articular inflammation. Blocking PD-1 in cancer therapy has been associated with an increase in inflammatory arthritis. In this 12-week study, RA disease activity was analyzed in patients randomly assigned to two different monthly intravenous doses of peresolimab or placebo. Of note, a large majority of participants were seropositive for rheumatoid factor (RF) or cyclic citrullinated peptide (CCP). Patients receiving the 700-mg dose of peresolimab had a better American College of Rheumatology (ACR) 20 response than did those receiving placebo (71% vs 42%), but not a better ACR50 or ACR70 response; the 300-mg dose was not better than placebo. Although reported adverse events were similar in all three groups, with a short timeframe it would be difficult to address concerns about cancer risk. Though this novel treatment is exciting, a larger and longer-term trial is necessary to address this concern as well as potentially tease out risk factors (including age or other immunosuppression) in this susceptible group.
Two other studies examined use of a much older csDMARD therapy, hydroxychloroquine (HCQ), in Brazilian patients with RA. Bredemeier and colleagues looked at the effects of HCQ on adverse events as well as the persistence of bDMARD/targeted synthetic DMARD (tsDMARD) therapy in over 1300 patients with RA. Using the BiobadaBrasil registry of patients starting their first bDMARD or Janus kinase (JAK) inhibitor, they looked at effects of combination therapy with HCQ during the treatment course of up to six bDMARD or JAK inhibitors. At baseline, patients prescribed antimalarial therapy had shorter RA duration and began treatment earlier, perhaps due to patient or physician preferences regarding starting "milder" antimalarial medication earlier or due to use of "triple therapy" with methotrexate and sulfasalazine. Of interest, patients receiving antimalarial therapy had a lower incidence of adverse events, especially serious infections, but no effect on cardiovascular events was seen despite HCQ's perceived beneficial effects on thrombotic risk and cholesterol profile. Patients receiving HCQ were also more likely to persist in their course of bDMARD or JAK inhibitor therapy, though the effect size seems relatively small. As the focus in this study was on adverse effects, the authors' analysis of the effects on antimalarials on the persistence of therapy was not detailed.
Lin and colleagues also looked at the effects of HCQ in patients with older-onset RA with respect to mortality risk. Using data from the electronic health records of a hospital in Taiwan, mortality-associated risk factors were evaluated in 980 patients with RA diagnosed at >60 years. Male sex, current smoking status, and cancer status were all associated with mortality, whereas HCQ use was associated with reduced mortality (hazard ratio 0.30). In contrast to the registry study mentioned above, patients receiving HCQ had a lower risk for cardiovascular events, hyperlipidemia, diabetes, and chronic kidney disease. Interaction with cancer was less clear due to lower number of patients. Of interest, use of cyclosporine, leflunomide, and a bDMARD was associated with higher mortality risk. The source and true relevance of the potential risk reduction in this study is not clear because of the lack of prospective data, but combined with the information above, this study suggests that the benefits of HCQ use should not be discounted in patients with RA.
Despite multiple existing conventional synthetic disease-modifying antirheumatic drug (csDMARD) and biologic DMARD (bDMARD) options, many patients with rheumatoid arthritis (RA) do not respond adequately to treatment. In an exciting development, a recent phase 2 study by Tuttle and colleagues examined a novel treatment approach in RA: stimulation of the programmed cell death protein 1 (PD-1) inhibitor pathway. PD-1 is a checkpoint inhibitor receptor whose activation reflects T-cell activation and may play a role in synovitis and extra-articular inflammation. Blocking PD-1 in cancer therapy has been associated with an increase in inflammatory arthritis. In this 12-week study, RA disease activity was analyzed in patients randomly assigned to two different monthly intravenous doses of peresolimab or placebo. Of note, a large majority of participants were seropositive for rheumatoid factor (RF) or cyclic citrullinated peptide (CCP). Patients receiving the 700-mg dose of peresolimab had a better American College of Rheumatology (ACR) 20 response than did those receiving placebo (71% vs 42%), but not a better ACR50 or ACR70 response; the 300-mg dose was not better than placebo. Although reported adverse events were similar in all three groups, with a short timeframe it would be difficult to address concerns about cancer risk. Though this novel treatment is exciting, a larger and longer-term trial is necessary to address this concern as well as potentially tease out risk factors (including age or other immunosuppression) in this susceptible group.
Two other studies examined use of a much older csDMARD therapy, hydroxychloroquine (HCQ), in Brazilian patients with RA. Bredemeier and colleagues looked at the effects of HCQ on adverse events as well as the persistence of bDMARD/targeted synthetic DMARD (tsDMARD) therapy in over 1300 patients with RA. Using the BiobadaBrasil registry of patients starting their first bDMARD or Janus kinase (JAK) inhibitor, they looked at effects of combination therapy with HCQ during the treatment course of up to six bDMARD or JAK inhibitors. At baseline, patients prescribed antimalarial therapy had shorter RA duration and began treatment earlier, perhaps due to patient or physician preferences regarding starting "milder" antimalarial medication earlier or due to use of "triple therapy" with methotrexate and sulfasalazine. Of interest, patients receiving antimalarial therapy had a lower incidence of adverse events, especially serious infections, but no effect on cardiovascular events was seen despite HCQ's perceived beneficial effects on thrombotic risk and cholesterol profile. Patients receiving HCQ were also more likely to persist in their course of bDMARD or JAK inhibitor therapy, though the effect size seems relatively small. As the focus in this study was on adverse effects, the authors' analysis of the effects on antimalarials on the persistence of therapy was not detailed.
Lin and colleagues also looked at the effects of HCQ in patients with older-onset RA with respect to mortality risk. Using data from the electronic health records of a hospital in Taiwan, mortality-associated risk factors were evaluated in 980 patients with RA diagnosed at >60 years. Male sex, current smoking status, and cancer status were all associated with mortality, whereas HCQ use was associated with reduced mortality (hazard ratio 0.30). In contrast to the registry study mentioned above, patients receiving HCQ had a lower risk for cardiovascular events, hyperlipidemia, diabetes, and chronic kidney disease. Interaction with cancer was less clear due to lower number of patients. Of interest, use of cyclosporine, leflunomide, and a bDMARD was associated with higher mortality risk. The source and true relevance of the potential risk reduction in this study is not clear because of the lack of prospective data, but combined with the information above, this study suggests that the benefits of HCQ use should not be discounted in patients with RA.
Despite multiple existing conventional synthetic disease-modifying antirheumatic drug (csDMARD) and biologic DMARD (bDMARD) options, many patients with rheumatoid arthritis (RA) do not respond adequately to treatment. In an exciting development, a recent phase 2 study by Tuttle and colleagues examined a novel treatment approach in RA: stimulation of the programmed cell death protein 1 (PD-1) inhibitor pathway. PD-1 is a checkpoint inhibitor receptor whose activation reflects T-cell activation and may play a role in synovitis and extra-articular inflammation. Blocking PD-1 in cancer therapy has been associated with an increase in inflammatory arthritis. In this 12-week study, RA disease activity was analyzed in patients randomly assigned to two different monthly intravenous doses of peresolimab or placebo. Of note, a large majority of participants were seropositive for rheumatoid factor (RF) or cyclic citrullinated peptide (CCP). Patients receiving the 700-mg dose of peresolimab had a better American College of Rheumatology (ACR) 20 response than did those receiving placebo (71% vs 42%), but not a better ACR50 or ACR70 response; the 300-mg dose was not better than placebo. Although reported adverse events were similar in all three groups, with a short timeframe it would be difficult to address concerns about cancer risk. Though this novel treatment is exciting, a larger and longer-term trial is necessary to address this concern as well as potentially tease out risk factors (including age or other immunosuppression) in this susceptible group.
Two other studies examined use of a much older csDMARD therapy, hydroxychloroquine (HCQ), in Brazilian patients with RA. Bredemeier and colleagues looked at the effects of HCQ on adverse events as well as the persistence of bDMARD/targeted synthetic DMARD (tsDMARD) therapy in over 1300 patients with RA. Using the BiobadaBrasil registry of patients starting their first bDMARD or Janus kinase (JAK) inhibitor, they looked at effects of combination therapy with HCQ during the treatment course of up to six bDMARD or JAK inhibitors. At baseline, patients prescribed antimalarial therapy had shorter RA duration and began treatment earlier, perhaps due to patient or physician preferences regarding starting "milder" antimalarial medication earlier or due to use of "triple therapy" with methotrexate and sulfasalazine. Of interest, patients receiving antimalarial therapy had a lower incidence of adverse events, especially serious infections, but no effect on cardiovascular events was seen despite HCQ's perceived beneficial effects on thrombotic risk and cholesterol profile. Patients receiving HCQ were also more likely to persist in their course of bDMARD or JAK inhibitor therapy, though the effect size seems relatively small. As the focus in this study was on adverse effects, the authors' analysis of the effects on antimalarials on the persistence of therapy was not detailed.
Lin and colleagues also looked at the effects of HCQ in patients with older-onset RA with respect to mortality risk. Using data from the electronic health records of a hospital in Taiwan, mortality-associated risk factors were evaluated in 980 patients with RA diagnosed at >60 years. Male sex, current smoking status, and cancer status were all associated with mortality, whereas HCQ use was associated with reduced mortality (hazard ratio 0.30). In contrast to the registry study mentioned above, patients receiving HCQ had a lower risk for cardiovascular events, hyperlipidemia, diabetes, and chronic kidney disease. Interaction with cancer was less clear due to lower number of patients. Of interest, use of cyclosporine, leflunomide, and a bDMARD was associated with higher mortality risk. The source and true relevance of the potential risk reduction in this study is not clear because of the lack of prospective data, but combined with the information above, this study suggests that the benefits of HCQ use should not be discounted in patients with RA.
Placebo effect can be found in a cup of coffee
The best part of waking up is placebo in your cup
Coffee makes the world go round. It’s impossible to picture any workplace without a cast of forlorn characters huddled around the office coffee maker on a Monday morning, imbibing their beverage du jour until they’ve been lifted out of their semi-zombified stupor.
Millions upon millions of people swear by their morning coffee. And if they don’t get that sweet, sweet caffeine boost, they’ll make Garfield and the Boomtown Rats’ opinions of Mondays look tame. And it only makes sense that they’d believe that. After all, caffeine is a stimulant. It helps your brain focus and kicks it into overdrive. Of course drinking a beverage full of caffeine wakes you up. Right?
Not so fast, a group of Portuguese researchers say. That morning cup of coffee? It may actually be a placebo. Cue the dramatic sound effect.
Here’s the scoop: After recruiting a group of coffee drinkers (at least one cup a day), the researchers kept their test subjects off of coffee for at least 3 hours, then performed a brief functional MRI scan on all test subjects. Half an hour later, study participants received either a standard cup of coffee or pure caffeine. Half an hour after consuming their respective study product, the subjects underwent a second MRI.
As expected, both people who consumed coffee and those who consumed pure caffeine showed decreased connectivity in the default mode network after consumption, indicating preparation in the brain to move from resting to working on tasks. However, those who had pure caffeine did not show increased connectivity in the visual and executive control networks, while those who had coffee did. Simply put, caffeine may wake you up, but it doesn’t make you any sharper. Only coffee gets you in shape for that oh-so-important Monday meeting.
This doesn’t make a lot of sense. How can the drug part of coffee not be responsible for every effect the drink gives you? That’s where the placebo comes in, according to the scientists. It’s possible the effect they saw was caused by withdrawal – after just 3 hours? Yikes, hope not – but it’s more likely it comes down to psychology. We expect coffee to wake us up and make us ready for the day, so that’s exactly what it does. Hey, if that’s all it takes, time to convince ourselves that eating an entire pizza is actually an incredibly effective weight loss tool. Don’t let us down now, placebo effect.
Bread, milk, toilet paper, AFib diagnosis
Now consider the shopping cart. It does its job of carrying stuff around the store well enough, but can it lift you out of a semi-zombified stupor in the morning? No. Can it identify undiagnosed atrial fibrillation? Again, no.
Not so fast, say the investigators conducting the SHOPS-AF (Supermarket/Hypermarket Opportunistic Screening for Atrial Fibrillation) study. They built a better shopping cart. Except they call it a trolley, not a cart, since the study was conducted in England, where they sometimes have funny names for things.
Their improved shopping trolley – we’re just going to call it a cart from here on – has an electrocardiogram sensor embedded into the handlebar, so it can effectively detect AFib in shoppers who held it for at least 60 seconds. The sensor lights up red if it detects an irregular heartbeat and green if it does not. Let’s see a cup of coffee do that.
They put 10 of these modified carts in four supermarkets in Liverpool to see what would happen. Would shoppers be able to tell that we secretly replaced the fine coffee they usually serve with Folger’s crystals? Oops. Sorry about that. Coffee on the brain, apparently. Back to the carts.
A total of 2,155 adult shoppers used one of the carts over 2 months, and electrocardiogram data were available for 220 participants who either had a red light on the sensor and/or an irregular pulse that suggested atrial fibrillation. After further review by the SHOPS-AF cardiologist, AFib was diagnosed in 59 shoppers, of whom 39 were previously undiagnosed.
They’re already working to cut the scan time to 30 seconds for SHOPS-AF II, but we’re wondering about a possible flaw in the whole health-care-delivery-through-shopping-cart scenario. When we go to the local super/hyper/megamart, it seems like half of the people trundling up and down the aisles are store employees filling orders for customers who won’t even set foot inside. Is the shopping cart on its way out? Maybe. Who wants to tell the SHOPS-AF II team? Not us.
Put pneumonia where your mouth is
Getting dentures does not mean the end of dental care. If anything, new research reveals a huge reason for staying on top of one’s denture care: pneumonia.
It all started with swabs. Scientists in the United Kingdom took mouth, tongue, and denture specimens from frail elderly hospital patients who had pneumonia and wore dentures and from similar patients in care homes who wore dentures and did not have pneumonia. When they compared the microbial populations of the two groups, the investigators found about 20 times the number of respiratory pathogens on the dentures of those with pneumonia.
The research team suggested that dentures may play a role in causing pneumonia, but lead author Josh Twigg, BDS, PhD, also noted that “you certainly couldn’t say that people got pneumonia because they were wearing dentures. It’s just showing that there is an association there.” Improper cleaning, though, could lead to microbial colonization of the dentures, and patients could be inhaling those microbes into their lungs, thereby turning a dental issue into a respiratory issue.
More research needs to be done on the association between dentures and pneumonia, but Dr. Twigg hoped that the results of this study could be presented to the public. The message? “It is important to clean dentures thoroughly” and visit the dentist regularly, he said, but the best way to prevent denture-related infections is to avoid needing to wear dentures entirely.
The best part of waking up is placebo in your cup
Coffee makes the world go round. It’s impossible to picture any workplace without a cast of forlorn characters huddled around the office coffee maker on a Monday morning, imbibing their beverage du jour until they’ve been lifted out of their semi-zombified stupor.
Millions upon millions of people swear by their morning coffee. And if they don’t get that sweet, sweet caffeine boost, they’ll make Garfield and the Boomtown Rats’ opinions of Mondays look tame. And it only makes sense that they’d believe that. After all, caffeine is a stimulant. It helps your brain focus and kicks it into overdrive. Of course drinking a beverage full of caffeine wakes you up. Right?
Not so fast, a group of Portuguese researchers say. That morning cup of coffee? It may actually be a placebo. Cue the dramatic sound effect.
Here’s the scoop: After recruiting a group of coffee drinkers (at least one cup a day), the researchers kept their test subjects off of coffee for at least 3 hours, then performed a brief functional MRI scan on all test subjects. Half an hour later, study participants received either a standard cup of coffee or pure caffeine. Half an hour after consuming their respective study product, the subjects underwent a second MRI.
As expected, both people who consumed coffee and those who consumed pure caffeine showed decreased connectivity in the default mode network after consumption, indicating preparation in the brain to move from resting to working on tasks. However, those who had pure caffeine did not show increased connectivity in the visual and executive control networks, while those who had coffee did. Simply put, caffeine may wake you up, but it doesn’t make you any sharper. Only coffee gets you in shape for that oh-so-important Monday meeting.
This doesn’t make a lot of sense. How can the drug part of coffee not be responsible for every effect the drink gives you? That’s where the placebo comes in, according to the scientists. It’s possible the effect they saw was caused by withdrawal – after just 3 hours? Yikes, hope not – but it’s more likely it comes down to psychology. We expect coffee to wake us up and make us ready for the day, so that’s exactly what it does. Hey, if that’s all it takes, time to convince ourselves that eating an entire pizza is actually an incredibly effective weight loss tool. Don’t let us down now, placebo effect.
Bread, milk, toilet paper, AFib diagnosis
Now consider the shopping cart. It does its job of carrying stuff around the store well enough, but can it lift you out of a semi-zombified stupor in the morning? No. Can it identify undiagnosed atrial fibrillation? Again, no.
Not so fast, say the investigators conducting the SHOPS-AF (Supermarket/Hypermarket Opportunistic Screening for Atrial Fibrillation) study. They built a better shopping cart. Except they call it a trolley, not a cart, since the study was conducted in England, where they sometimes have funny names for things.
Their improved shopping trolley – we’re just going to call it a cart from here on – has an electrocardiogram sensor embedded into the handlebar, so it can effectively detect AFib in shoppers who held it for at least 60 seconds. The sensor lights up red if it detects an irregular heartbeat and green if it does not. Let’s see a cup of coffee do that.
They put 10 of these modified carts in four supermarkets in Liverpool to see what would happen. Would shoppers be able to tell that we secretly replaced the fine coffee they usually serve with Folger’s crystals? Oops. Sorry about that. Coffee on the brain, apparently. Back to the carts.
A total of 2,155 adult shoppers used one of the carts over 2 months, and electrocardiogram data were available for 220 participants who either had a red light on the sensor and/or an irregular pulse that suggested atrial fibrillation. After further review by the SHOPS-AF cardiologist, AFib was diagnosed in 59 shoppers, of whom 39 were previously undiagnosed.
They’re already working to cut the scan time to 30 seconds for SHOPS-AF II, but we’re wondering about a possible flaw in the whole health-care-delivery-through-shopping-cart scenario. When we go to the local super/hyper/megamart, it seems like half of the people trundling up and down the aisles are store employees filling orders for customers who won’t even set foot inside. Is the shopping cart on its way out? Maybe. Who wants to tell the SHOPS-AF II team? Not us.
Put pneumonia where your mouth is
Getting dentures does not mean the end of dental care. If anything, new research reveals a huge reason for staying on top of one’s denture care: pneumonia.
It all started with swabs. Scientists in the United Kingdom took mouth, tongue, and denture specimens from frail elderly hospital patients who had pneumonia and wore dentures and from similar patients in care homes who wore dentures and did not have pneumonia. When they compared the microbial populations of the two groups, the investigators found about 20 times the number of respiratory pathogens on the dentures of those with pneumonia.
The research team suggested that dentures may play a role in causing pneumonia, but lead author Josh Twigg, BDS, PhD, also noted that “you certainly couldn’t say that people got pneumonia because they were wearing dentures. It’s just showing that there is an association there.” Improper cleaning, though, could lead to microbial colonization of the dentures, and patients could be inhaling those microbes into their lungs, thereby turning a dental issue into a respiratory issue.
More research needs to be done on the association between dentures and pneumonia, but Dr. Twigg hoped that the results of this study could be presented to the public. The message? “It is important to clean dentures thoroughly” and visit the dentist regularly, he said, but the best way to prevent denture-related infections is to avoid needing to wear dentures entirely.
The best part of waking up is placebo in your cup
Coffee makes the world go round. It’s impossible to picture any workplace without a cast of forlorn characters huddled around the office coffee maker on a Monday morning, imbibing their beverage du jour until they’ve been lifted out of their semi-zombified stupor.
Millions upon millions of people swear by their morning coffee. And if they don’t get that sweet, sweet caffeine boost, they’ll make Garfield and the Boomtown Rats’ opinions of Mondays look tame. And it only makes sense that they’d believe that. After all, caffeine is a stimulant. It helps your brain focus and kicks it into overdrive. Of course drinking a beverage full of caffeine wakes you up. Right?
Not so fast, a group of Portuguese researchers say. That morning cup of coffee? It may actually be a placebo. Cue the dramatic sound effect.
Here’s the scoop: After recruiting a group of coffee drinkers (at least one cup a day), the researchers kept their test subjects off of coffee for at least 3 hours, then performed a brief functional MRI scan on all test subjects. Half an hour later, study participants received either a standard cup of coffee or pure caffeine. Half an hour after consuming their respective study product, the subjects underwent a second MRI.
As expected, both people who consumed coffee and those who consumed pure caffeine showed decreased connectivity in the default mode network after consumption, indicating preparation in the brain to move from resting to working on tasks. However, those who had pure caffeine did not show increased connectivity in the visual and executive control networks, while those who had coffee did. Simply put, caffeine may wake you up, but it doesn’t make you any sharper. Only coffee gets you in shape for that oh-so-important Monday meeting.
This doesn’t make a lot of sense. How can the drug part of coffee not be responsible for every effect the drink gives you? That’s where the placebo comes in, according to the scientists. It’s possible the effect they saw was caused by withdrawal – after just 3 hours? Yikes, hope not – but it’s more likely it comes down to psychology. We expect coffee to wake us up and make us ready for the day, so that’s exactly what it does. Hey, if that’s all it takes, time to convince ourselves that eating an entire pizza is actually an incredibly effective weight loss tool. Don’t let us down now, placebo effect.
Bread, milk, toilet paper, AFib diagnosis
Now consider the shopping cart. It does its job of carrying stuff around the store well enough, but can it lift you out of a semi-zombified stupor in the morning? No. Can it identify undiagnosed atrial fibrillation? Again, no.
Not so fast, say the investigators conducting the SHOPS-AF (Supermarket/Hypermarket Opportunistic Screening for Atrial Fibrillation) study. They built a better shopping cart. Except they call it a trolley, not a cart, since the study was conducted in England, where they sometimes have funny names for things.
Their improved shopping trolley – we’re just going to call it a cart from here on – has an electrocardiogram sensor embedded into the handlebar, so it can effectively detect AFib in shoppers who held it for at least 60 seconds. The sensor lights up red if it detects an irregular heartbeat and green if it does not. Let’s see a cup of coffee do that.
They put 10 of these modified carts in four supermarkets in Liverpool to see what would happen. Would shoppers be able to tell that we secretly replaced the fine coffee they usually serve with Folger’s crystals? Oops. Sorry about that. Coffee on the brain, apparently. Back to the carts.
A total of 2,155 adult shoppers used one of the carts over 2 months, and electrocardiogram data were available for 220 participants who either had a red light on the sensor and/or an irregular pulse that suggested atrial fibrillation. After further review by the SHOPS-AF cardiologist, AFib was diagnosed in 59 shoppers, of whom 39 were previously undiagnosed.
They’re already working to cut the scan time to 30 seconds for SHOPS-AF II, but we’re wondering about a possible flaw in the whole health-care-delivery-through-shopping-cart scenario. When we go to the local super/hyper/megamart, it seems like half of the people trundling up and down the aisles are store employees filling orders for customers who won’t even set foot inside. Is the shopping cart on its way out? Maybe. Who wants to tell the SHOPS-AF II team? Not us.
Put pneumonia where your mouth is
Getting dentures does not mean the end of dental care. If anything, new research reveals a huge reason for staying on top of one’s denture care: pneumonia.
It all started with swabs. Scientists in the United Kingdom took mouth, tongue, and denture specimens from frail elderly hospital patients who had pneumonia and wore dentures and from similar patients in care homes who wore dentures and did not have pneumonia. When they compared the microbial populations of the two groups, the investigators found about 20 times the number of respiratory pathogens on the dentures of those with pneumonia.
The research team suggested that dentures may play a role in causing pneumonia, but lead author Josh Twigg, BDS, PhD, also noted that “you certainly couldn’t say that people got pneumonia because they were wearing dentures. It’s just showing that there is an association there.” Improper cleaning, though, could lead to microbial colonization of the dentures, and patients could be inhaling those microbes into their lungs, thereby turning a dental issue into a respiratory issue.
More research needs to be done on the association between dentures and pneumonia, but Dr. Twigg hoped that the results of this study could be presented to the public. The message? “It is important to clean dentures thoroughly” and visit the dentist regularly, he said, but the best way to prevent denture-related infections is to avoid needing to wear dentures entirely.
New international guidelines on opioid deprescribing
An expert panel of pain management clinicians has released what they say are the first international guidelines for general practitioners on opioid analgesic deprescribing in adults.
The recommendations describe best practices for stopping opioid therapy and emphasize slow tapering and individualized deprescribing plans tailored to each patient.
Developed by general practitioners, pain specialists, addiction specialists, pharmacists, registered nurses, consumers, and physiotherapists, the guidelines note that deprescribing may not be appropriate for every patient and that stopping abruptly can be associated with an increased risk of overdose.
“Internationally, we were seeing significant harms from opioids but also significant harms from unsolicited and abrupt opioid cessation,” said lead author Aili Langford, PhD, who conducted the study as a doctoral student at the University of Sydney. “It was clear that recommendations to support safe and person-centered opioid deprescribing were required.”
The findings were published online in the Medical Journal of Australia.
Deprescribing plan
The consensus guidelines include 11 recommendations for deprescribing in adult patients who take at least one opioid for any type of pain.
Recommendations include implementing a deprescribing plan when opioids are first prescribed and gradual and individualized deprescribing, with regular monitoring and review.
Clinicians should consider opioid deprescribing in patients who experience no clinically meaningful improvement in function, quality of life, or pain at high risk with opioid therapy, they note. Patients who are at high risk for opioid-related harm are also good candidates for deprescribing.
Stopping opioid therapy is not recommended for patients with severe opioid use disorder (OUD). In those patients, medication-assisted OUD treatment and other evidence-based interventions are recommended.
“Opioids can be effective in pain management,” co-author Carl Schneider, PhD, an associate professor of pharmacy at the University of Sydney, said in a press release. “However, over the longer term, the risk of harms may outweigh the benefits.”
A ‘global problem’
Commenting on the guidelines, Orman Trent Hall, DO, assistant professor of addiction medicine, department of psychiatry and behavioral health at the Ohio State University Wexner Medical Center, Columbus, said they are similar to recommendations published by the U.S. Centers for Disease Control and Prevention in 2016 and 2022 but offer additional information that could be helpful.
“This new guideline provides more explicit advice about tapering and withdrawal management, which may be useful to practitioners. The opioid crisis is a global problem, and while individual countries may require local solutions, the new international guideline may offer a framework for approaching this issue,” he said.
The guideline’s emphasis on the potential risks of deprescribing in some patients is also key, Dr. Hall added. Patients who are tapering off opioid therapy may have worsening pain and loss of function that can affect their quality of life.
“Patients may also experience psychological harm and increased risk of opioid use disorder and death by suicide following opioid deprescribing,” Dr. Hall said. “Therefore, it is important for providers to carefully weigh the risks of prescribing and deprescribing and engage patients with person-centered communication and shared decision-making.”
The work was funded by grants from the University of Sydney and the National Health and Medical Research Council. Full disclosures are available in the original article. Dr. Hall has provided expert opinion to the health care consultancy firm Lumanity and Emergent BioSolutions regarding the overdose crisis.
A version of this article originally appeared on Medscape.com.
An expert panel of pain management clinicians has released what they say are the first international guidelines for general practitioners on opioid analgesic deprescribing in adults.
The recommendations describe best practices for stopping opioid therapy and emphasize slow tapering and individualized deprescribing plans tailored to each patient.
Developed by general practitioners, pain specialists, addiction specialists, pharmacists, registered nurses, consumers, and physiotherapists, the guidelines note that deprescribing may not be appropriate for every patient and that stopping abruptly can be associated with an increased risk of overdose.
“Internationally, we were seeing significant harms from opioids but also significant harms from unsolicited and abrupt opioid cessation,” said lead author Aili Langford, PhD, who conducted the study as a doctoral student at the University of Sydney. “It was clear that recommendations to support safe and person-centered opioid deprescribing were required.”
The findings were published online in the Medical Journal of Australia.
Deprescribing plan
The consensus guidelines include 11 recommendations for deprescribing in adult patients who take at least one opioid for any type of pain.
Recommendations include implementing a deprescribing plan when opioids are first prescribed and gradual and individualized deprescribing, with regular monitoring and review.
Clinicians should consider opioid deprescribing in patients who experience no clinically meaningful improvement in function, quality of life, or pain at high risk with opioid therapy, they note. Patients who are at high risk for opioid-related harm are also good candidates for deprescribing.
Stopping opioid therapy is not recommended for patients with severe opioid use disorder (OUD). In those patients, medication-assisted OUD treatment and other evidence-based interventions are recommended.
“Opioids can be effective in pain management,” co-author Carl Schneider, PhD, an associate professor of pharmacy at the University of Sydney, said in a press release. “However, over the longer term, the risk of harms may outweigh the benefits.”
A ‘global problem’
Commenting on the guidelines, Orman Trent Hall, DO, assistant professor of addiction medicine, department of psychiatry and behavioral health at the Ohio State University Wexner Medical Center, Columbus, said they are similar to recommendations published by the U.S. Centers for Disease Control and Prevention in 2016 and 2022 but offer additional information that could be helpful.
“This new guideline provides more explicit advice about tapering and withdrawal management, which may be useful to practitioners. The opioid crisis is a global problem, and while individual countries may require local solutions, the new international guideline may offer a framework for approaching this issue,” he said.
The guideline’s emphasis on the potential risks of deprescribing in some patients is also key, Dr. Hall added. Patients who are tapering off opioid therapy may have worsening pain and loss of function that can affect their quality of life.
“Patients may also experience psychological harm and increased risk of opioid use disorder and death by suicide following opioid deprescribing,” Dr. Hall said. “Therefore, it is important for providers to carefully weigh the risks of prescribing and deprescribing and engage patients with person-centered communication and shared decision-making.”
The work was funded by grants from the University of Sydney and the National Health and Medical Research Council. Full disclosures are available in the original article. Dr. Hall has provided expert opinion to the health care consultancy firm Lumanity and Emergent BioSolutions regarding the overdose crisis.
A version of this article originally appeared on Medscape.com.
An expert panel of pain management clinicians has released what they say are the first international guidelines for general practitioners on opioid analgesic deprescribing in adults.
The recommendations describe best practices for stopping opioid therapy and emphasize slow tapering and individualized deprescribing plans tailored to each patient.
Developed by general practitioners, pain specialists, addiction specialists, pharmacists, registered nurses, consumers, and physiotherapists, the guidelines note that deprescribing may not be appropriate for every patient and that stopping abruptly can be associated with an increased risk of overdose.
“Internationally, we were seeing significant harms from opioids but also significant harms from unsolicited and abrupt opioid cessation,” said lead author Aili Langford, PhD, who conducted the study as a doctoral student at the University of Sydney. “It was clear that recommendations to support safe and person-centered opioid deprescribing were required.”
The findings were published online in the Medical Journal of Australia.
Deprescribing plan
The consensus guidelines include 11 recommendations for deprescribing in adult patients who take at least one opioid for any type of pain.
Recommendations include implementing a deprescribing plan when opioids are first prescribed and gradual and individualized deprescribing, with regular monitoring and review.
Clinicians should consider opioid deprescribing in patients who experience no clinically meaningful improvement in function, quality of life, or pain at high risk with opioid therapy, they note. Patients who are at high risk for opioid-related harm are also good candidates for deprescribing.
Stopping opioid therapy is not recommended for patients with severe opioid use disorder (OUD). In those patients, medication-assisted OUD treatment and other evidence-based interventions are recommended.
“Opioids can be effective in pain management,” co-author Carl Schneider, PhD, an associate professor of pharmacy at the University of Sydney, said in a press release. “However, over the longer term, the risk of harms may outweigh the benefits.”
A ‘global problem’
Commenting on the guidelines, Orman Trent Hall, DO, assistant professor of addiction medicine, department of psychiatry and behavioral health at the Ohio State University Wexner Medical Center, Columbus, said they are similar to recommendations published by the U.S. Centers for Disease Control and Prevention in 2016 and 2022 but offer additional information that could be helpful.
“This new guideline provides more explicit advice about tapering and withdrawal management, which may be useful to practitioners. The opioid crisis is a global problem, and while individual countries may require local solutions, the new international guideline may offer a framework for approaching this issue,” he said.
The guideline’s emphasis on the potential risks of deprescribing in some patients is also key, Dr. Hall added. Patients who are tapering off opioid therapy may have worsening pain and loss of function that can affect their quality of life.
“Patients may also experience psychological harm and increased risk of opioid use disorder and death by suicide following opioid deprescribing,” Dr. Hall said. “Therefore, it is important for providers to carefully weigh the risks of prescribing and deprescribing and engage patients with person-centered communication and shared decision-making.”
The work was funded by grants from the University of Sydney and the National Health and Medical Research Council. Full disclosures are available in the original article. Dr. Hall has provided expert opinion to the health care consultancy firm Lumanity and Emergent BioSolutions regarding the overdose crisis.
A version of this article originally appeared on Medscape.com.
Commentary: Evaluating new and established treatments for PsA, July 2023
Despite using methotrexate (MTX) in the management of psoriasis and PsA for more than 50 years, there is paucity of data on its effectiveness in PsA. It is also largely assumed that MTX is more effective for rheumatoid arthritis (RA) than for PsA. To evaluate MTX effectiveness vis-à-vis RA, Lindström and colleagues conducted an observational study with disease-modifying antirheumatic drug (DMARD)–naive patients with newly diagnosed PsA (n = 3642) who initiated MTX and matched comparator patients with RA (n = 3642) from national Swedish registers. At 6 months, although both groups of patients had improvement in pain, global health, and remission, the rates were higher in RA compared with PsA. Overall, 71% of patients with PsA and 76% of patients with RA were still taking MTX 2 years after initiating MTX. The time to starting another DMARD was shorter in RA compared with PsA. Thus, in early PsA, MTX treatment seems to provide significant benefits. However, the improvements are less impressive than they are for RA.
Biologic agents are generally safe and effective in the treatment of PsA. However, there are limited data on the safety and effectiveness in older and younger populations with PsA. Gossec and colleagues conducted a post hoc analysis of the PsABio trial that included patients with PsA who received ustekinumab, an interleukin 12/23 inhibitor, and were subgrouped into those < 60 years (n = 336) and ≥ 60 years (n = 103). At 6 months, a numerically higher proportion of patients < 60 years achieved clinical Disease Activity Index for Psoriatic Arthritis low disease activity compared with those ≥ 60 years. The proportions of patients reporting at least one (32.7% vs 40.9%) or serious (5.3% vs 9.6%) adverse events were numerically higher in the older population. However, treatment persistence was numerically higher in the older subgroup. These differences were not clinically meaningful; thus, ustekinumab may be safely used in older populations.
There are also limited data on the effectiveness of biologics in patients with juvenile PsA (jPsA). To address this, Correll and colleagues evaluated the safety and effectiveness of etanercept, an anti–tumor necrosis factor agent, in patients with jPsA using data from the Childhood Arthritis and Rheumatology Research Alliance Registry. In 226 patients, the overall incidence of adverse events of special interest and serious adverse events were low and included three cases of uveitis (incidence rate [IR]/100 person-years 0.55; 95% CI 0.18-1.69), one of neuropathy (IR/100 person-years 0.18; 95% CI 0.03-1.29), and one of cancer (IR/100 person-years 0.13; 95% CI 0.02-0.90). The ACR provisional criteria for inactive disease were achieved by 51.9% and 43.8% of patients at 6 and 12 months. Thus, etanercept is safe and effective in jPsA.
Despite using methotrexate (MTX) in the management of psoriasis and PsA for more than 50 years, there is paucity of data on its effectiveness in PsA. It is also largely assumed that MTX is more effective for rheumatoid arthritis (RA) than for PsA. To evaluate MTX effectiveness vis-à-vis RA, Lindström and colleagues conducted an observational study with disease-modifying antirheumatic drug (DMARD)–naive patients with newly diagnosed PsA (n = 3642) who initiated MTX and matched comparator patients with RA (n = 3642) from national Swedish registers. At 6 months, although both groups of patients had improvement in pain, global health, and remission, the rates were higher in RA compared with PsA. Overall, 71% of patients with PsA and 76% of patients with RA were still taking MTX 2 years after initiating MTX. The time to starting another DMARD was shorter in RA compared with PsA. Thus, in early PsA, MTX treatment seems to provide significant benefits. However, the improvements are less impressive than they are for RA.
Biologic agents are generally safe and effective in the treatment of PsA. However, there are limited data on the safety and effectiveness in older and younger populations with PsA. Gossec and colleagues conducted a post hoc analysis of the PsABio trial that included patients with PsA who received ustekinumab, an interleukin 12/23 inhibitor, and were subgrouped into those < 60 years (n = 336) and ≥ 60 years (n = 103). At 6 months, a numerically higher proportion of patients < 60 years achieved clinical Disease Activity Index for Psoriatic Arthritis low disease activity compared with those ≥ 60 years. The proportions of patients reporting at least one (32.7% vs 40.9%) or serious (5.3% vs 9.6%) adverse events were numerically higher in the older population. However, treatment persistence was numerically higher in the older subgroup. These differences were not clinically meaningful; thus, ustekinumab may be safely used in older populations.
There are also limited data on the effectiveness of biologics in patients with juvenile PsA (jPsA). To address this, Correll and colleagues evaluated the safety and effectiveness of etanercept, an anti–tumor necrosis factor agent, in patients with jPsA using data from the Childhood Arthritis and Rheumatology Research Alliance Registry. In 226 patients, the overall incidence of adverse events of special interest and serious adverse events were low and included three cases of uveitis (incidence rate [IR]/100 person-years 0.55; 95% CI 0.18-1.69), one of neuropathy (IR/100 person-years 0.18; 95% CI 0.03-1.29), and one of cancer (IR/100 person-years 0.13; 95% CI 0.02-0.90). The ACR provisional criteria for inactive disease were achieved by 51.9% and 43.8% of patients at 6 and 12 months. Thus, etanercept is safe and effective in jPsA.
Despite using methotrexate (MTX) in the management of psoriasis and PsA for more than 50 years, there is paucity of data on its effectiveness in PsA. It is also largely assumed that MTX is more effective for rheumatoid arthritis (RA) than for PsA. To evaluate MTX effectiveness vis-à-vis RA, Lindström and colleagues conducted an observational study with disease-modifying antirheumatic drug (DMARD)–naive patients with newly diagnosed PsA (n = 3642) who initiated MTX and matched comparator patients with RA (n = 3642) from national Swedish registers. At 6 months, although both groups of patients had improvement in pain, global health, and remission, the rates were higher in RA compared with PsA. Overall, 71% of patients with PsA and 76% of patients with RA were still taking MTX 2 years after initiating MTX. The time to starting another DMARD was shorter in RA compared with PsA. Thus, in early PsA, MTX treatment seems to provide significant benefits. However, the improvements are less impressive than they are for RA.
Biologic agents are generally safe and effective in the treatment of PsA. However, there are limited data on the safety and effectiveness in older and younger populations with PsA. Gossec and colleagues conducted a post hoc analysis of the PsABio trial that included patients with PsA who received ustekinumab, an interleukin 12/23 inhibitor, and were subgrouped into those < 60 years (n = 336) and ≥ 60 years (n = 103). At 6 months, a numerically higher proportion of patients < 60 years achieved clinical Disease Activity Index for Psoriatic Arthritis low disease activity compared with those ≥ 60 years. The proportions of patients reporting at least one (32.7% vs 40.9%) or serious (5.3% vs 9.6%) adverse events were numerically higher in the older population. However, treatment persistence was numerically higher in the older subgroup. These differences were not clinically meaningful; thus, ustekinumab may be safely used in older populations.
There are also limited data on the effectiveness of biologics in patients with juvenile PsA (jPsA). To address this, Correll and colleagues evaluated the safety and effectiveness of etanercept, an anti–tumor necrosis factor agent, in patients with jPsA using data from the Childhood Arthritis and Rheumatology Research Alliance Registry. In 226 patients, the overall incidence of adverse events of special interest and serious adverse events were low and included three cases of uveitis (incidence rate [IR]/100 person-years 0.55; 95% CI 0.18-1.69), one of neuropathy (IR/100 person-years 0.18; 95% CI 0.03-1.29), and one of cancer (IR/100 person-years 0.13; 95% CI 0.02-0.90). The ACR provisional criteria for inactive disease were achieved by 51.9% and 43.8% of patients at 6 and 12 months. Thus, etanercept is safe and effective in jPsA.
Physician suicide roundtable: 8 important initiatives that can help
Physician suicide continues to be a challenging problem in the United States. Each year, 1 in 10 doctors think about or attempt suicide, and 400 die by suicide each year. More than half of the doctors reading this know a colleague who has attempted or died by suicide.
These are part of a public health suicide prevention strategy, the preferred method for prevention, in hospitals and institutions around the country. A public health model for preventing suicide is a multifaceted approach that includes universal education, health promotion, selective and targeted prevention, and treatment and recovery.
These physicians hope to continue creating and implementing these and other risk-reduction measures across all health care organizations.
Our physician experts for this discussion
Mary Moffit, PhD, is an associate professor in the department of psychiatry at Oregon Health & Science University, Portland. She directs the resident and faculty wellness program and is director of the OHSU peer support program. She helped design and developed a comprehensive wellness program that is now a national model for academic medical centers.
Christine Yu Moutier, MD, is the chief medical officer of the American Foundation for Suicide Prevention. She is the author of “Suicide Prevention,” a Cambridge University Press clinical handbook. She has been a practicing psychiatrist, professor of psychiatry, dean in the medical school at the University of California, San Diego, and medical director of the inpatient psychiatric unit at the VA Medical Center in La Jolla, Calif.
Michael F. Myers, MD, is a professor of clinical psychiatry in the department of psychiatry & behavioral sciences at the State University of New York, Brooklyn. He is recent past vice-chair of education and director of training in the department of psychiatry & behavioral sciences at the university. He is the author of several books, including “Why Physicians Die By Suicide,” “The Physician as Patient,” and “Touched by Suicide.”
The participants discussed these risk-reduction initiatives as having much potential for helping physicians at risk for suicide and suicidal ideations.
The importance of peer support programs
Peer support program models may differ across institutions but typically describe colleagues providing some degree of emotional first aid to peers who may be at risk.
Dr. Moffit: The Pew support program that we have in place at OHSU, similar to what’s available in many hospitals and systems nationwide, trains individual physicians across multiple specialties in a peer support model. It’s not specifically emotional first aid, although that’s integral to it. It’s also for adverse events: Having a tragic patient death, having learned that you will be named in a lawsuit, and exposure to trauma in the medical role.
Peer to peer is not where we anticipate physicians seeking someone to talk to about their marital relationship not going well. However, the peer supporter will know about resources throughout the university and the community for what is needed. We’ve got 20-30 peer supporters. We try to match them – for example, a surgeon with a surgeon, a primary care doc with a primary care doc. Physicians who use peer support aren’t tracked, and no notes are taken or documented. It takes place informally but has changed the culture and lowered a barrier. We have a waiting list of people who want to be peer supporters.
Dr. Moutier: Peer-to-peer support is usually part of a multi-pronged program and is usually not the only effort going on. Depending on how they’re set up, the goals may be slightly different for each program. Peer-to-peer can be one of the most powerful ways to augment awareness raising and education, which is almost always a basic first step.
Dr. Myers: It doesn’t feel as threatening when people start in a peer-to-peer support group. Users may have been afraid of getting a mental health diagnosis, but with peers, many of whom are often not psychiatrists, that eases distress. Peer support can break down that sense of isolation and loneliness so that someone can take the next step.
Dr. Moutier: To be connected to family, to any community resource, frankly, is a protective factor that mitigates suicide risk. So that’s the logic model from a suicide prevention standpoint. It may be the only opportunity for someone to start disclosing what they’re experiencing, receive validation and support, and not a judgmental response. It can open up the avenue toward help-seeking.
Opt-in/opt-out support for medical residents
This initiative matches residents with a counselor as part of their orientation.
Dr. Moffit: Each resident has a meet and greet with a counselor when they arrive or in their first 6 months at their university. The resident can opt out and cancel the meeting, but they’re scheduled for it as part of their “curriculum.” Institutions like Michigan, Columbia, Montefiore, Mount Sinai, and the University of California, San Diego, have this in place. It starts something like: ‘Hello. Good afternoon. How’s it going? I’m Dr. Moffitt, and here are the services available in this program.’
Dr. Myers: It’s another excellent example of normalizing the stress in the rigors of training and making it part of the wellness initiative.
Dr. Moutier: It’s just a normal part of orientation. Again, as a universal strategy, one thing that I was doing at UCSD with a particular group of medical students, who were at higher risk, was a postbaccalaureate program that found students from underrepresented, under-resourced backgrounds and brought them into this post-bacc year. I was directing it and mentoring these students.
So, I could afford a lot more intensive time and attention to them because it was a small group, but every one of them had regular meetings with me every 2 weeks. My approach was to help them uncover their unique strengths and vulnerabilities as they started this program. They all made it into med school.
It was a very intensive and more concierge-personalized approach. It’s like personalized medicine. What specific self-care, mentoring, and mental health care plan would each student codesign with me to stay on track?
And it would involve very holistic things, like if part of their vulnerability was that leaving their Chicano family was creating stress because their father had said: ‘You’re leaving our culture and our family by going into the profession of medicine,’ then we had specific plans around how to care for that aspect of their struggle. It was a much more informed, customized mentoring approach called the UCSD CAP (Conditional Acceptance Post-Baccalaureate Program). It could be a feature in a more specialized opt-in/opt-out program.
One-question survey: How full is your gas tank?
This initiative is a one-question survey emailed/texted to residents to check in on their wellness. We ask, how full is your gas tank? Select 1 to 5 (Empty to Full). If they flag low, they receive a follow-up.
Dr. Moffit: It’s certainly a metaphor that we use. It’s the idea of being depleted in combination with being extremely sleep deprived and the inability to access the usual sources of support or outlets, and how that can create a perfect storm of a level of distress that can put physicians at risk.
Dr. Moutier: It is a way to help people realize that there are things they can do proactively to keep that tank at least somewhat full enough.
Dr. Myers: Using colloquial or figurative language can get better buy-in than “Here’s a PHQ-9.” It also has a caring or intimate tone to it. Somebody could feel they’re a 1 in this rotation but a 4-5 the next. We know from a lot of the literature that when residents get a good, welcoming orientation, their satisfaction with that rotation is uniformly better than if they’re thrown to the wolves. And we know trial by fire can put trainees at risk.
A buddy to check in with
This initiative is when you’re assigned a buddy in or out of residency that you regularly check in with about how you’re doing.
Dr. Myers: Not to be cynical, but there has been some mentor/mentee research that if you’re assigned a mentor, the results are not nearly as good. And if it’s left to the individual to find a mentor, results could be marginal as well. You need a guide to say, ‘Here are some potential mentors for you, but you decide.’ We do a lot of that at (SUNY) Downstate instead of assigning a person. So, it may require some oversight. Picking a check-in buddy from a list provided rather than having one assigned may be more beneficial.
A lot of what we’re talking about are universal strategies that allow for increased interpersonal connection, which is a protective factor that normalizes help-seeking.
A platform or social media forum to share experiences
An online forum or platform where medical students, residents, and physicians can discuss mental health and suicide prevention. Physicians with personal experience could provide testimonials.
Dr. Myers: I’ve recently signed a book contract, and the working title is “Physicians With Lived Experience: How Their Stories Give Clinical Guidance.” When I talk with doctors who have published their personal stories in the New England Journal of Medicine, JAMA, or sometimes The Washington Post or The New York Times, many of them have said they had no idea at the beginning of their journey that they would do something like this: be transparent about their story. It’s a measure of their health, growth, and grace.
Dr. Moutier: The current president of the Academic Association of Surgeons, Carrie Cunningham, MD, MPH, used her platform at the annual AAS conference in 2022 to focus on suicide prevention. She told her own recent story of having gotten into recovery after having been near suicide and struggling with addiction. It was a groundbreaking moment for the field of surgery and produced a ripple effect. She risked everything to tell her story, which was highly emotional since it was still raw. It got everyone engaged, a real turning point for that field. Storytelling and a place for trainees to discuss suicide prevention, and physicians to recall their lived experiences can be highly beneficial.
Interactive Screening Program
The Interactive Screening Program (ISP) is used in higher education to allow physicians to take a safe, confidential screening test and receive a personalized response that can connect them to mental health services before a crisis emerges.
Dr. Moutier: ISP is a tool within a public health model that can afford anonymity to the user so they can safely have their needs addressed. It’s a way for high-risk individuals to sync up with treatment and support. It’s sometimes used in the universal approach because it can be offered to everyone within the health system community of physicians and staff.
It can produce a ripple effect of normalizing that we all have mental health to take care of. Its intended value is in identifying those with a higher risk for suicide, but it doesn’t stop at identifying those at risk. It helps physicians move past a stage of suffering in silence.
Our data show that 86% of a very high-risk group (currently having suicidal ideation, a recent attempt, or other high-risk factors for suicide) aren’t in any form of treatment and have not disclosed their situation to anyone. A fairly high percentage of those going through ISP request a referral to treatment. It’s a unique, very niche tool, and because users remain anonymous, that affords safety around confidentiality.
It’s usually part of a multipronged approach with education, stigma reduction, storytelling, peer support, and other modalities. In my experience with the UCSD HEAR (Healer Assessment Education and Recovery) program, which is still going strong in about its 15th year, the program went from seeing 13 physicians die by suicide in the years leading up to its launch and in the 15 years since it’s been going, one suicide. We all believe that the ISP is the heart of prevention.
Even though all of the universal strategies are important, they probably wouldn’t be sufficient by themselves because the risk [for suicide] is dynamic, and you have to catch people when they are suffering and ready to seek treatment. Suicide prevention is challenging and must be strategic, multifaceted, and sustained over time.
The importance of confidentiality for physicians
In the past, physicians may have been hesitant to seek treatment when struggling with mental health, substance use disorder and suicidal ideations because they heard stories from doctors who said they had to disclose mental health treatment to medical and state licensing boards.
Dr. Myers: There is so much dated stuff out there, and it gets propagated by people who have had a bad experience. I’m not challenging the authenticity of that, but I feel like those are in the minority. The vast majority of people are seeking help. The Federation of State Physician Health Programs is working with state boards to update and get rid of antiquated questions, and they’re working with credentialing groups.
When I was in practice and my patient was petrified of having to come into the hospital [because of confidentiality] I would just be their physician and say: “Look, I know that this is a worry for you [licensing and credentialing issues] but trust me, I’m going to help you get well; that’s my job. And I’m going to help you sort all that out afterward.” It was part of my work as their physician that if they were going to have to jump through hurdles to get their license reinstated, etc., I could help.
The Dr. Lorna Breen Heroes’ Foundation is also doing so much good work in this area, especially with their toolkits to audit, change, remove, and communicate the changes about intrusive language in licensing applications and credentialing. (Dr. Breen was a New York City ED physician who died by suicide in April 2020 during the early days and height of the COVID-19 pandemic. Her father was quoted as saying: “She was in the trenches. She was a hero.”)
Dr. Moutier: We’re seeing hundreds of physicians get therapy and psychiatric treatment annually. And the advocacy effort is incredibly important, and I think we are witnessing a swifter pace to eliminate those inappropriate and illegal questions about mental health and mental health treatment for physicians and nurses.
Dr. Moffit: We have lowered barriers, not only in individual institutions but also with programming. We have also worked with the Federation of State Medical Boards and The Lorna Breen Foundation to change the legislation. The Foundation has audited and changed 20 state medical boards to remove intrusive language from licensing applications.
Support for colleagues working to help each other
Dr. Myers: One final note for those physicians who need to take time out for medical leave: In my clinical experience, I find that they felt lonely as they were getting well. I can’t tell you how much it made a difference for those who received a phone call, a card, or an email from their colleagues at work. It doesn’t take long for a vibrant, active physician to feel out of the loop when ill.
We know from suicide literature that when somebody’s discharged from the hospital or the emergency department, caring communications, brief expressions of care and concern by email, letter, card, text message, etc., can make all the difference to their recovery. Reaching out to those struggling and those in recovery can help your fellow physician.
A version of this article originally appeared on Medscape.com.
Physician suicide continues to be a challenging problem in the United States. Each year, 1 in 10 doctors think about or attempt suicide, and 400 die by suicide each year. More than half of the doctors reading this know a colleague who has attempted or died by suicide.
These are part of a public health suicide prevention strategy, the preferred method for prevention, in hospitals and institutions around the country. A public health model for preventing suicide is a multifaceted approach that includes universal education, health promotion, selective and targeted prevention, and treatment and recovery.
These physicians hope to continue creating and implementing these and other risk-reduction measures across all health care organizations.
Our physician experts for this discussion
Mary Moffit, PhD, is an associate professor in the department of psychiatry at Oregon Health & Science University, Portland. She directs the resident and faculty wellness program and is director of the OHSU peer support program. She helped design and developed a comprehensive wellness program that is now a national model for academic medical centers.
Christine Yu Moutier, MD, is the chief medical officer of the American Foundation for Suicide Prevention. She is the author of “Suicide Prevention,” a Cambridge University Press clinical handbook. She has been a practicing psychiatrist, professor of psychiatry, dean in the medical school at the University of California, San Diego, and medical director of the inpatient psychiatric unit at the VA Medical Center in La Jolla, Calif.
Michael F. Myers, MD, is a professor of clinical psychiatry in the department of psychiatry & behavioral sciences at the State University of New York, Brooklyn. He is recent past vice-chair of education and director of training in the department of psychiatry & behavioral sciences at the university. He is the author of several books, including “Why Physicians Die By Suicide,” “The Physician as Patient,” and “Touched by Suicide.”
The participants discussed these risk-reduction initiatives as having much potential for helping physicians at risk for suicide and suicidal ideations.
The importance of peer support programs
Peer support program models may differ across institutions but typically describe colleagues providing some degree of emotional first aid to peers who may be at risk.
Dr. Moffit: The Pew support program that we have in place at OHSU, similar to what’s available in many hospitals and systems nationwide, trains individual physicians across multiple specialties in a peer support model. It’s not specifically emotional first aid, although that’s integral to it. It’s also for adverse events: Having a tragic patient death, having learned that you will be named in a lawsuit, and exposure to trauma in the medical role.
Peer to peer is not where we anticipate physicians seeking someone to talk to about their marital relationship not going well. However, the peer supporter will know about resources throughout the university and the community for what is needed. We’ve got 20-30 peer supporters. We try to match them – for example, a surgeon with a surgeon, a primary care doc with a primary care doc. Physicians who use peer support aren’t tracked, and no notes are taken or documented. It takes place informally but has changed the culture and lowered a barrier. We have a waiting list of people who want to be peer supporters.
Dr. Moutier: Peer-to-peer support is usually part of a multi-pronged program and is usually not the only effort going on. Depending on how they’re set up, the goals may be slightly different for each program. Peer-to-peer can be one of the most powerful ways to augment awareness raising and education, which is almost always a basic first step.
Dr. Myers: It doesn’t feel as threatening when people start in a peer-to-peer support group. Users may have been afraid of getting a mental health diagnosis, but with peers, many of whom are often not psychiatrists, that eases distress. Peer support can break down that sense of isolation and loneliness so that someone can take the next step.
Dr. Moutier: To be connected to family, to any community resource, frankly, is a protective factor that mitigates suicide risk. So that’s the logic model from a suicide prevention standpoint. It may be the only opportunity for someone to start disclosing what they’re experiencing, receive validation and support, and not a judgmental response. It can open up the avenue toward help-seeking.
Opt-in/opt-out support for medical residents
This initiative matches residents with a counselor as part of their orientation.
Dr. Moffit: Each resident has a meet and greet with a counselor when they arrive or in their first 6 months at their university. The resident can opt out and cancel the meeting, but they’re scheduled for it as part of their “curriculum.” Institutions like Michigan, Columbia, Montefiore, Mount Sinai, and the University of California, San Diego, have this in place. It starts something like: ‘Hello. Good afternoon. How’s it going? I’m Dr. Moffitt, and here are the services available in this program.’
Dr. Myers: It’s another excellent example of normalizing the stress in the rigors of training and making it part of the wellness initiative.
Dr. Moutier: It’s just a normal part of orientation. Again, as a universal strategy, one thing that I was doing at UCSD with a particular group of medical students, who were at higher risk, was a postbaccalaureate program that found students from underrepresented, under-resourced backgrounds and brought them into this post-bacc year. I was directing it and mentoring these students.
So, I could afford a lot more intensive time and attention to them because it was a small group, but every one of them had regular meetings with me every 2 weeks. My approach was to help them uncover their unique strengths and vulnerabilities as they started this program. They all made it into med school.
It was a very intensive and more concierge-personalized approach. It’s like personalized medicine. What specific self-care, mentoring, and mental health care plan would each student codesign with me to stay on track?
And it would involve very holistic things, like if part of their vulnerability was that leaving their Chicano family was creating stress because their father had said: ‘You’re leaving our culture and our family by going into the profession of medicine,’ then we had specific plans around how to care for that aspect of their struggle. It was a much more informed, customized mentoring approach called the UCSD CAP (Conditional Acceptance Post-Baccalaureate Program). It could be a feature in a more specialized opt-in/opt-out program.
One-question survey: How full is your gas tank?
This initiative is a one-question survey emailed/texted to residents to check in on their wellness. We ask, how full is your gas tank? Select 1 to 5 (Empty to Full). If they flag low, they receive a follow-up.
Dr. Moffit: It’s certainly a metaphor that we use. It’s the idea of being depleted in combination with being extremely sleep deprived and the inability to access the usual sources of support or outlets, and how that can create a perfect storm of a level of distress that can put physicians at risk.
Dr. Moutier: It is a way to help people realize that there are things they can do proactively to keep that tank at least somewhat full enough.
Dr. Myers: Using colloquial or figurative language can get better buy-in than “Here’s a PHQ-9.” It also has a caring or intimate tone to it. Somebody could feel they’re a 1 in this rotation but a 4-5 the next. We know from a lot of the literature that when residents get a good, welcoming orientation, their satisfaction with that rotation is uniformly better than if they’re thrown to the wolves. And we know trial by fire can put trainees at risk.
A buddy to check in with
This initiative is when you’re assigned a buddy in or out of residency that you regularly check in with about how you’re doing.
Dr. Myers: Not to be cynical, but there has been some mentor/mentee research that if you’re assigned a mentor, the results are not nearly as good. And if it’s left to the individual to find a mentor, results could be marginal as well. You need a guide to say, ‘Here are some potential mentors for you, but you decide.’ We do a lot of that at (SUNY) Downstate instead of assigning a person. So, it may require some oversight. Picking a check-in buddy from a list provided rather than having one assigned may be more beneficial.
A lot of what we’re talking about are universal strategies that allow for increased interpersonal connection, which is a protective factor that normalizes help-seeking.
A platform or social media forum to share experiences
An online forum or platform where medical students, residents, and physicians can discuss mental health and suicide prevention. Physicians with personal experience could provide testimonials.
Dr. Myers: I’ve recently signed a book contract, and the working title is “Physicians With Lived Experience: How Their Stories Give Clinical Guidance.” When I talk with doctors who have published their personal stories in the New England Journal of Medicine, JAMA, or sometimes The Washington Post or The New York Times, many of them have said they had no idea at the beginning of their journey that they would do something like this: be transparent about their story. It’s a measure of their health, growth, and grace.
Dr. Moutier: The current president of the Academic Association of Surgeons, Carrie Cunningham, MD, MPH, used her platform at the annual AAS conference in 2022 to focus on suicide prevention. She told her own recent story of having gotten into recovery after having been near suicide and struggling with addiction. It was a groundbreaking moment for the field of surgery and produced a ripple effect. She risked everything to tell her story, which was highly emotional since it was still raw. It got everyone engaged, a real turning point for that field. Storytelling and a place for trainees to discuss suicide prevention, and physicians to recall their lived experiences can be highly beneficial.
Interactive Screening Program
The Interactive Screening Program (ISP) is used in higher education to allow physicians to take a safe, confidential screening test and receive a personalized response that can connect them to mental health services before a crisis emerges.
Dr. Moutier: ISP is a tool within a public health model that can afford anonymity to the user so they can safely have their needs addressed. It’s a way for high-risk individuals to sync up with treatment and support. It’s sometimes used in the universal approach because it can be offered to everyone within the health system community of physicians and staff.
It can produce a ripple effect of normalizing that we all have mental health to take care of. Its intended value is in identifying those with a higher risk for suicide, but it doesn’t stop at identifying those at risk. It helps physicians move past a stage of suffering in silence.
Our data show that 86% of a very high-risk group (currently having suicidal ideation, a recent attempt, or other high-risk factors for suicide) aren’t in any form of treatment and have not disclosed their situation to anyone. A fairly high percentage of those going through ISP request a referral to treatment. It’s a unique, very niche tool, and because users remain anonymous, that affords safety around confidentiality.
It’s usually part of a multipronged approach with education, stigma reduction, storytelling, peer support, and other modalities. In my experience with the UCSD HEAR (Healer Assessment Education and Recovery) program, which is still going strong in about its 15th year, the program went from seeing 13 physicians die by suicide in the years leading up to its launch and in the 15 years since it’s been going, one suicide. We all believe that the ISP is the heart of prevention.
Even though all of the universal strategies are important, they probably wouldn’t be sufficient by themselves because the risk [for suicide] is dynamic, and you have to catch people when they are suffering and ready to seek treatment. Suicide prevention is challenging and must be strategic, multifaceted, and sustained over time.
The importance of confidentiality for physicians
In the past, physicians may have been hesitant to seek treatment when struggling with mental health, substance use disorder and suicidal ideations because they heard stories from doctors who said they had to disclose mental health treatment to medical and state licensing boards.
Dr. Myers: There is so much dated stuff out there, and it gets propagated by people who have had a bad experience. I’m not challenging the authenticity of that, but I feel like those are in the minority. The vast majority of people are seeking help. The Federation of State Physician Health Programs is working with state boards to update and get rid of antiquated questions, and they’re working with credentialing groups.
When I was in practice and my patient was petrified of having to come into the hospital [because of confidentiality] I would just be their physician and say: “Look, I know that this is a worry for you [licensing and credentialing issues] but trust me, I’m going to help you get well; that’s my job. And I’m going to help you sort all that out afterward.” It was part of my work as their physician that if they were going to have to jump through hurdles to get their license reinstated, etc., I could help.
The Dr. Lorna Breen Heroes’ Foundation is also doing so much good work in this area, especially with their toolkits to audit, change, remove, and communicate the changes about intrusive language in licensing applications and credentialing. (Dr. Breen was a New York City ED physician who died by suicide in April 2020 during the early days and height of the COVID-19 pandemic. Her father was quoted as saying: “She was in the trenches. She was a hero.”)
Dr. Moutier: We’re seeing hundreds of physicians get therapy and psychiatric treatment annually. And the advocacy effort is incredibly important, and I think we are witnessing a swifter pace to eliminate those inappropriate and illegal questions about mental health and mental health treatment for physicians and nurses.
Dr. Moffit: We have lowered barriers, not only in individual institutions but also with programming. We have also worked with the Federation of State Medical Boards and The Lorna Breen Foundation to change the legislation. The Foundation has audited and changed 20 state medical boards to remove intrusive language from licensing applications.
Support for colleagues working to help each other
Dr. Myers: One final note for those physicians who need to take time out for medical leave: In my clinical experience, I find that they felt lonely as they were getting well. I can’t tell you how much it made a difference for those who received a phone call, a card, or an email from their colleagues at work. It doesn’t take long for a vibrant, active physician to feel out of the loop when ill.
We know from suicide literature that when somebody’s discharged from the hospital or the emergency department, caring communications, brief expressions of care and concern by email, letter, card, text message, etc., can make all the difference to their recovery. Reaching out to those struggling and those in recovery can help your fellow physician.
A version of this article originally appeared on Medscape.com.
Physician suicide continues to be a challenging problem in the United States. Each year, 1 in 10 doctors think about or attempt suicide, and 400 die by suicide each year. More than half of the doctors reading this know a colleague who has attempted or died by suicide.
These are part of a public health suicide prevention strategy, the preferred method for prevention, in hospitals and institutions around the country. A public health model for preventing suicide is a multifaceted approach that includes universal education, health promotion, selective and targeted prevention, and treatment and recovery.
These physicians hope to continue creating and implementing these and other risk-reduction measures across all health care organizations.
Our physician experts for this discussion
Mary Moffit, PhD, is an associate professor in the department of psychiatry at Oregon Health & Science University, Portland. She directs the resident and faculty wellness program and is director of the OHSU peer support program. She helped design and developed a comprehensive wellness program that is now a national model for academic medical centers.
Christine Yu Moutier, MD, is the chief medical officer of the American Foundation for Suicide Prevention. She is the author of “Suicide Prevention,” a Cambridge University Press clinical handbook. She has been a practicing psychiatrist, professor of psychiatry, dean in the medical school at the University of California, San Diego, and medical director of the inpatient psychiatric unit at the VA Medical Center in La Jolla, Calif.
Michael F. Myers, MD, is a professor of clinical psychiatry in the department of psychiatry & behavioral sciences at the State University of New York, Brooklyn. He is recent past vice-chair of education and director of training in the department of psychiatry & behavioral sciences at the university. He is the author of several books, including “Why Physicians Die By Suicide,” “The Physician as Patient,” and “Touched by Suicide.”
The participants discussed these risk-reduction initiatives as having much potential for helping physicians at risk for suicide and suicidal ideations.
The importance of peer support programs
Peer support program models may differ across institutions but typically describe colleagues providing some degree of emotional first aid to peers who may be at risk.
Dr. Moffit: The Pew support program that we have in place at OHSU, similar to what’s available in many hospitals and systems nationwide, trains individual physicians across multiple specialties in a peer support model. It’s not specifically emotional first aid, although that’s integral to it. It’s also for adverse events: Having a tragic patient death, having learned that you will be named in a lawsuit, and exposure to trauma in the medical role.
Peer to peer is not where we anticipate physicians seeking someone to talk to about their marital relationship not going well. However, the peer supporter will know about resources throughout the university and the community for what is needed. We’ve got 20-30 peer supporters. We try to match them – for example, a surgeon with a surgeon, a primary care doc with a primary care doc. Physicians who use peer support aren’t tracked, and no notes are taken or documented. It takes place informally but has changed the culture and lowered a barrier. We have a waiting list of people who want to be peer supporters.
Dr. Moutier: Peer-to-peer support is usually part of a multi-pronged program and is usually not the only effort going on. Depending on how they’re set up, the goals may be slightly different for each program. Peer-to-peer can be one of the most powerful ways to augment awareness raising and education, which is almost always a basic first step.
Dr. Myers: It doesn’t feel as threatening when people start in a peer-to-peer support group. Users may have been afraid of getting a mental health diagnosis, but with peers, many of whom are often not psychiatrists, that eases distress. Peer support can break down that sense of isolation and loneliness so that someone can take the next step.
Dr. Moutier: To be connected to family, to any community resource, frankly, is a protective factor that mitigates suicide risk. So that’s the logic model from a suicide prevention standpoint. It may be the only opportunity for someone to start disclosing what they’re experiencing, receive validation and support, and not a judgmental response. It can open up the avenue toward help-seeking.
Opt-in/opt-out support for medical residents
This initiative matches residents with a counselor as part of their orientation.
Dr. Moffit: Each resident has a meet and greet with a counselor when they arrive or in their first 6 months at their university. The resident can opt out and cancel the meeting, but they’re scheduled for it as part of their “curriculum.” Institutions like Michigan, Columbia, Montefiore, Mount Sinai, and the University of California, San Diego, have this in place. It starts something like: ‘Hello. Good afternoon. How’s it going? I’m Dr. Moffitt, and here are the services available in this program.’
Dr. Myers: It’s another excellent example of normalizing the stress in the rigors of training and making it part of the wellness initiative.
Dr. Moutier: It’s just a normal part of orientation. Again, as a universal strategy, one thing that I was doing at UCSD with a particular group of medical students, who were at higher risk, was a postbaccalaureate program that found students from underrepresented, under-resourced backgrounds and brought them into this post-bacc year. I was directing it and mentoring these students.
So, I could afford a lot more intensive time and attention to them because it was a small group, but every one of them had regular meetings with me every 2 weeks. My approach was to help them uncover their unique strengths and vulnerabilities as they started this program. They all made it into med school.
It was a very intensive and more concierge-personalized approach. It’s like personalized medicine. What specific self-care, mentoring, and mental health care plan would each student codesign with me to stay on track?
And it would involve very holistic things, like if part of their vulnerability was that leaving their Chicano family was creating stress because their father had said: ‘You’re leaving our culture and our family by going into the profession of medicine,’ then we had specific plans around how to care for that aspect of their struggle. It was a much more informed, customized mentoring approach called the UCSD CAP (Conditional Acceptance Post-Baccalaureate Program). It could be a feature in a more specialized opt-in/opt-out program.
One-question survey: How full is your gas tank?
This initiative is a one-question survey emailed/texted to residents to check in on their wellness. We ask, how full is your gas tank? Select 1 to 5 (Empty to Full). If they flag low, they receive a follow-up.
Dr. Moffit: It’s certainly a metaphor that we use. It’s the idea of being depleted in combination with being extremely sleep deprived and the inability to access the usual sources of support or outlets, and how that can create a perfect storm of a level of distress that can put physicians at risk.
Dr. Moutier: It is a way to help people realize that there are things they can do proactively to keep that tank at least somewhat full enough.
Dr. Myers: Using colloquial or figurative language can get better buy-in than “Here’s a PHQ-9.” It also has a caring or intimate tone to it. Somebody could feel they’re a 1 in this rotation but a 4-5 the next. We know from a lot of the literature that when residents get a good, welcoming orientation, their satisfaction with that rotation is uniformly better than if they’re thrown to the wolves. And we know trial by fire can put trainees at risk.
A buddy to check in with
This initiative is when you’re assigned a buddy in or out of residency that you regularly check in with about how you’re doing.
Dr. Myers: Not to be cynical, but there has been some mentor/mentee research that if you’re assigned a mentor, the results are not nearly as good. And if it’s left to the individual to find a mentor, results could be marginal as well. You need a guide to say, ‘Here are some potential mentors for you, but you decide.’ We do a lot of that at (SUNY) Downstate instead of assigning a person. So, it may require some oversight. Picking a check-in buddy from a list provided rather than having one assigned may be more beneficial.
A lot of what we’re talking about are universal strategies that allow for increased interpersonal connection, which is a protective factor that normalizes help-seeking.
A platform or social media forum to share experiences
An online forum or platform where medical students, residents, and physicians can discuss mental health and suicide prevention. Physicians with personal experience could provide testimonials.
Dr. Myers: I’ve recently signed a book contract, and the working title is “Physicians With Lived Experience: How Their Stories Give Clinical Guidance.” When I talk with doctors who have published their personal stories in the New England Journal of Medicine, JAMA, or sometimes The Washington Post or The New York Times, many of them have said they had no idea at the beginning of their journey that they would do something like this: be transparent about their story. It’s a measure of their health, growth, and grace.
Dr. Moutier: The current president of the Academic Association of Surgeons, Carrie Cunningham, MD, MPH, used her platform at the annual AAS conference in 2022 to focus on suicide prevention. She told her own recent story of having gotten into recovery after having been near suicide and struggling with addiction. It was a groundbreaking moment for the field of surgery and produced a ripple effect. She risked everything to tell her story, which was highly emotional since it was still raw. It got everyone engaged, a real turning point for that field. Storytelling and a place for trainees to discuss suicide prevention, and physicians to recall their lived experiences can be highly beneficial.
Interactive Screening Program
The Interactive Screening Program (ISP) is used in higher education to allow physicians to take a safe, confidential screening test and receive a personalized response that can connect them to mental health services before a crisis emerges.
Dr. Moutier: ISP is a tool within a public health model that can afford anonymity to the user so they can safely have their needs addressed. It’s a way for high-risk individuals to sync up with treatment and support. It’s sometimes used in the universal approach because it can be offered to everyone within the health system community of physicians and staff.
It can produce a ripple effect of normalizing that we all have mental health to take care of. Its intended value is in identifying those with a higher risk for suicide, but it doesn’t stop at identifying those at risk. It helps physicians move past a stage of suffering in silence.
Our data show that 86% of a very high-risk group (currently having suicidal ideation, a recent attempt, or other high-risk factors for suicide) aren’t in any form of treatment and have not disclosed their situation to anyone. A fairly high percentage of those going through ISP request a referral to treatment. It’s a unique, very niche tool, and because users remain anonymous, that affords safety around confidentiality.
It’s usually part of a multipronged approach with education, stigma reduction, storytelling, peer support, and other modalities. In my experience with the UCSD HEAR (Healer Assessment Education and Recovery) program, which is still going strong in about its 15th year, the program went from seeing 13 physicians die by suicide in the years leading up to its launch and in the 15 years since it’s been going, one suicide. We all believe that the ISP is the heart of prevention.
Even though all of the universal strategies are important, they probably wouldn’t be sufficient by themselves because the risk [for suicide] is dynamic, and you have to catch people when they are suffering and ready to seek treatment. Suicide prevention is challenging and must be strategic, multifaceted, and sustained over time.
The importance of confidentiality for physicians
In the past, physicians may have been hesitant to seek treatment when struggling with mental health, substance use disorder and suicidal ideations because they heard stories from doctors who said they had to disclose mental health treatment to medical and state licensing boards.
Dr. Myers: There is so much dated stuff out there, and it gets propagated by people who have had a bad experience. I’m not challenging the authenticity of that, but I feel like those are in the minority. The vast majority of people are seeking help. The Federation of State Physician Health Programs is working with state boards to update and get rid of antiquated questions, and they’re working with credentialing groups.
When I was in practice and my patient was petrified of having to come into the hospital [because of confidentiality] I would just be their physician and say: “Look, I know that this is a worry for you [licensing and credentialing issues] but trust me, I’m going to help you get well; that’s my job. And I’m going to help you sort all that out afterward.” It was part of my work as their physician that if they were going to have to jump through hurdles to get their license reinstated, etc., I could help.
The Dr. Lorna Breen Heroes’ Foundation is also doing so much good work in this area, especially with their toolkits to audit, change, remove, and communicate the changes about intrusive language in licensing applications and credentialing. (Dr. Breen was a New York City ED physician who died by suicide in April 2020 during the early days and height of the COVID-19 pandemic. Her father was quoted as saying: “She was in the trenches. She was a hero.”)
Dr. Moutier: We’re seeing hundreds of physicians get therapy and psychiatric treatment annually. And the advocacy effort is incredibly important, and I think we are witnessing a swifter pace to eliminate those inappropriate and illegal questions about mental health and mental health treatment for physicians and nurses.
Dr. Moffit: We have lowered barriers, not only in individual institutions but also with programming. We have also worked with the Federation of State Medical Boards and The Lorna Breen Foundation to change the legislation. The Foundation has audited and changed 20 state medical boards to remove intrusive language from licensing applications.
Support for colleagues working to help each other
Dr. Myers: One final note for those physicians who need to take time out for medical leave: In my clinical experience, I find that they felt lonely as they were getting well. I can’t tell you how much it made a difference for those who received a phone call, a card, or an email from their colleagues at work. It doesn’t take long for a vibrant, active physician to feel out of the loop when ill.
We know from suicide literature that when somebody’s discharged from the hospital or the emergency department, caring communications, brief expressions of care and concern by email, letter, card, text message, etc., can make all the difference to their recovery. Reaching out to those struggling and those in recovery can help your fellow physician.
A version of this article originally appeared on Medscape.com.
New DEA CME mandate affects 2 million prescribers
The Consolidated Appropriations Act of 2023 mandates that all Drug Enforcement Administration–registered physicians and health care providers complete a one-time, 8-hour CME training on managing and treating opioid and other substance abuse disorders. This requirement goes into effect on June 27, 2023. New DEA registrants must also comply. Veterinarians are exempt.
A DEA registration is required to prescribe any controlled substance. The DEA categorizes these as Schedule I-V, with V being the least likely to be abused (Table 1). For example, opioids like fentanyl, oxycodone, and morphine are Schedule II. Medications without abuse potential are not scheduled.
Will 16 million hours of opioid education save lives?
One should not underestimate the sweeping scope of this new federal requirement. DEA registrants include physicians and other health care providers such as nurse practitioners, physician assistants, and dentists. That is 8 hours per provider x 2 million providers: 16 million hours of CME!
Many states already require 1 or more hours of opioid training and pain management as part of their relicensure requirements (Table 2). To avoid redundancy, the DEA-mandated 8-hour training satisfies the various states’ requirements. 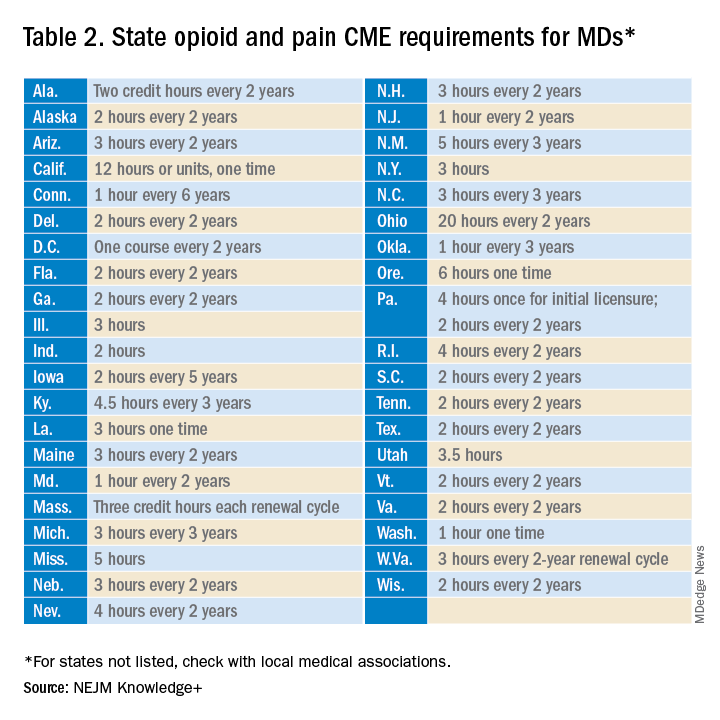
An uncompensated mandate
Physicians are no strangers to lifelong learning and most eagerly pursue educational opportunities. Though some physicians may have CME time and stipends allocated by their employers, many others, such as the approximately 50,000 locum tenens doctors, do not. However, as enthusiastic as these physicians may be about this new CME course, they will likely lose a day of seeing patients (and income) to comply with this new obligation.
Not just pain doctors
The mandate’s broad brush includes many health care providers who hold DEA certificates but do not prescribe opioids. For example, as a general neurologist and epileptologist, I do not treat patients with chronic pain and cannot remember the last time I wrote an opioid prescription. However, I frequently prescribe lacosamide, a Schedule V drug. A surprisingly large number of antiseizure drugs are Schedule III, IV, or V drugs (Table 3).
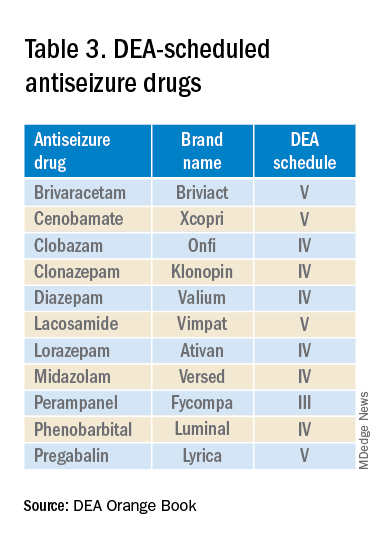
Real-world abuse?
How often scheduled antiseizure drugs are diverted or abused in an epilepsy population is unknown but appears to be infrequent. For example, perampanel abuse has not been reported despite its classification as a Schedule III drug. Anecdotally, in more than 40 years of clinical practice, I have never known a patient with epilepsy to abuse their antiseizure medications.
Take the course
Many organizations are happy to charge for the new 8-hour course. For example, the Tennessee Medical Association offers the training for $299 online or $400 in person. Materials from Elite Learning satisfy the 8-hour requirement for $80. However, NEJM Knowledge+ provides a complimentary 10-hour DEA-compliant course.
I recently completed the NEJM course. The information was thorough and took the whole 10 hours to finish. As excellent as it was, the content was only tangentially relevant to my clinical practice.
Conclusions
To obtain or renew a DEA certificate, neurologists, epilepsy specialists, and many other health care providers must comply with the new 8-hour CME opioid training mandate. Because the course requires 1 day to complete, health care providers would be prudent to obtain their CME well before their DEA certificate expires.
Though efforts to control the morbidity and mortality of the opioid epidemic are laudatory, perhaps the training should be more targeted to physicians who actually prescribe opioids rather than every DEA registrant. In the meantime, whether 16 million CME hours will save lives remains to be seen.
Dr. Wilner is professor of neurology at the University of Tennessee Health Science Center, Memphis. He reported a conflict of interest with Accordant Health Services.
A version of this article first appeared on Medscape.com.
The Consolidated Appropriations Act of 2023 mandates that all Drug Enforcement Administration–registered physicians and health care providers complete a one-time, 8-hour CME training on managing and treating opioid and other substance abuse disorders. This requirement goes into effect on June 27, 2023. New DEA registrants must also comply. Veterinarians are exempt.
A DEA registration is required to prescribe any controlled substance. The DEA categorizes these as Schedule I-V, with V being the least likely to be abused (Table 1). For example, opioids like fentanyl, oxycodone, and morphine are Schedule II. Medications without abuse potential are not scheduled.
Will 16 million hours of opioid education save lives?
One should not underestimate the sweeping scope of this new federal requirement. DEA registrants include physicians and other health care providers such as nurse practitioners, physician assistants, and dentists. That is 8 hours per provider x 2 million providers: 16 million hours of CME!
Many states already require 1 or more hours of opioid training and pain management as part of their relicensure requirements (Table 2). To avoid redundancy, the DEA-mandated 8-hour training satisfies the various states’ requirements. 
An uncompensated mandate
Physicians are no strangers to lifelong learning and most eagerly pursue educational opportunities. Though some physicians may have CME time and stipends allocated by their employers, many others, such as the approximately 50,000 locum tenens doctors, do not. However, as enthusiastic as these physicians may be about this new CME course, they will likely lose a day of seeing patients (and income) to comply with this new obligation.
Not just pain doctors
The mandate’s broad brush includes many health care providers who hold DEA certificates but do not prescribe opioids. For example, as a general neurologist and epileptologist, I do not treat patients with chronic pain and cannot remember the last time I wrote an opioid prescription. However, I frequently prescribe lacosamide, a Schedule V drug. A surprisingly large number of antiseizure drugs are Schedule III, IV, or V drugs (Table 3).

Real-world abuse?
How often scheduled antiseizure drugs are diverted or abused in an epilepsy population is unknown but appears to be infrequent. For example, perampanel abuse has not been reported despite its classification as a Schedule III drug. Anecdotally, in more than 40 years of clinical practice, I have never known a patient with epilepsy to abuse their antiseizure medications.
Take the course
Many organizations are happy to charge for the new 8-hour course. For example, the Tennessee Medical Association offers the training for $299 online or $400 in person. Materials from Elite Learning satisfy the 8-hour requirement for $80. However, NEJM Knowledge+ provides a complimentary 10-hour DEA-compliant course.
I recently completed the NEJM course. The information was thorough and took the whole 10 hours to finish. As excellent as it was, the content was only tangentially relevant to my clinical practice.
Conclusions
To obtain or renew a DEA certificate, neurologists, epilepsy specialists, and many other health care providers must comply with the new 8-hour CME opioid training mandate. Because the course requires 1 day to complete, health care providers would be prudent to obtain their CME well before their DEA certificate expires.
Though efforts to control the morbidity and mortality of the opioid epidemic are laudatory, perhaps the training should be more targeted to physicians who actually prescribe opioids rather than every DEA registrant. In the meantime, whether 16 million CME hours will save lives remains to be seen.
Dr. Wilner is professor of neurology at the University of Tennessee Health Science Center, Memphis. He reported a conflict of interest with Accordant Health Services.
A version of this article first appeared on Medscape.com.
The Consolidated Appropriations Act of 2023 mandates that all Drug Enforcement Administration–registered physicians and health care providers complete a one-time, 8-hour CME training on managing and treating opioid and other substance abuse disorders. This requirement goes into effect on June 27, 2023. New DEA registrants must also comply. Veterinarians are exempt.
A DEA registration is required to prescribe any controlled substance. The DEA categorizes these as Schedule I-V, with V being the least likely to be abused (Table 1). For example, opioids like fentanyl, oxycodone, and morphine are Schedule II. Medications without abuse potential are not scheduled.
Will 16 million hours of opioid education save lives?
One should not underestimate the sweeping scope of this new federal requirement. DEA registrants include physicians and other health care providers such as nurse practitioners, physician assistants, and dentists. That is 8 hours per provider x 2 million providers: 16 million hours of CME!
Many states already require 1 or more hours of opioid training and pain management as part of their relicensure requirements (Table 2). To avoid redundancy, the DEA-mandated 8-hour training satisfies the various states’ requirements. 
An uncompensated mandate
Physicians are no strangers to lifelong learning and most eagerly pursue educational opportunities. Though some physicians may have CME time and stipends allocated by their employers, many others, such as the approximately 50,000 locum tenens doctors, do not. However, as enthusiastic as these physicians may be about this new CME course, they will likely lose a day of seeing patients (and income) to comply with this new obligation.
Not just pain doctors
The mandate’s broad brush includes many health care providers who hold DEA certificates but do not prescribe opioids. For example, as a general neurologist and epileptologist, I do not treat patients with chronic pain and cannot remember the last time I wrote an opioid prescription. However, I frequently prescribe lacosamide, a Schedule V drug. A surprisingly large number of antiseizure drugs are Schedule III, IV, or V drugs (Table 3).

Real-world abuse?
How often scheduled antiseizure drugs are diverted or abused in an epilepsy population is unknown but appears to be infrequent. For example, perampanel abuse has not been reported despite its classification as a Schedule III drug. Anecdotally, in more than 40 years of clinical practice, I have never known a patient with epilepsy to abuse their antiseizure medications.
Take the course
Many organizations are happy to charge for the new 8-hour course. For example, the Tennessee Medical Association offers the training for $299 online or $400 in person. Materials from Elite Learning satisfy the 8-hour requirement for $80. However, NEJM Knowledge+ provides a complimentary 10-hour DEA-compliant course.
I recently completed the NEJM course. The information was thorough and took the whole 10 hours to finish. As excellent as it was, the content was only tangentially relevant to my clinical practice.
Conclusions
To obtain or renew a DEA certificate, neurologists, epilepsy specialists, and many other health care providers must comply with the new 8-hour CME opioid training mandate. Because the course requires 1 day to complete, health care providers would be prudent to obtain their CME well before their DEA certificate expires.
Though efforts to control the morbidity and mortality of the opioid epidemic are laudatory, perhaps the training should be more targeted to physicians who actually prescribe opioids rather than every DEA registrant. In the meantime, whether 16 million CME hours will save lives remains to be seen.
Dr. Wilner is professor of neurology at the University of Tennessee Health Science Center, Memphis. He reported a conflict of interest with Accordant Health Services.
A version of this article first appeared on Medscape.com.