User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'main-prefix')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
div[contains(@class, 'view-medstat-quiz-listing-panes')]
div[contains(@class, 'pane-article-sidebar-latest-news')]
div[contains(@class, 'medstat-accordion-set article-series')]
Five ways docs may qualify for discounts on medical malpractice premiums
Getting a better deal might simply mean taking advantage of incentives and discounts your insurer may already offer. These include claims-free, new-to-practice, and working part-time discounts.
However, if you decide to shop around, keep in mind that discounts are just one factor that can affect your premium price – insurers look at your specialty, location, and claims history.
One of the most common ways physicians can earn discounts is by participating in risk management programs. With this type of program, physicians evaluate elements of their practice and documentation practices and identify areas that might leave them at risk for a lawsuit. While they save money, physician risk management programs also are designed to reduce malpractice claims, which ultimately minimizes the potential for bigger financial losses, insurance experts say.
“It’s a win-win situation when liability insurers and physicians work together to minimize risk, and it’s a win for patients,” said Gary Price, MD, president of The Physicians Foundation.
Doctors in private practice or employed by small hospitals that are not self-insured can qualify for these discounts, said David Zetter, president of Zetter HealthCare Management Consultants.
“I do a lot of work with medical malpractice companies trying to find clients policies. All the carriers are transparent about what physicians have to do to lower their premiums. Physicians can receive the discounts if they follow through and meet the insurer’s requirements,” said Mr. Zetter.
State insurance departments regulate medical malpractice insurance, including the premium credits insurers offer. Most states cap discounts at 25%, but some go as high as 70%, according to The Doctors Company, a national physician-owned medical malpractice insurer.
Insurers typically offer doctors several ways to earn discounts. The size of the discount also can depend on whether a doctor is new to a practice, remains claims free, or takes risk management courses.
In addition to the premium discount, some online risk management classes and webinars are eligible for CME credits.
“The credits can add up and they can be used for recertification or relicensure,” said Susan Boisvert, senior patient safety risk manager at The Doctors Company.
Here are five ways you may qualify for discounts with your insurer.
1. Make use of discounts available to new doctors
Doctors can earn hefty discounts on their premiums when they are no longer interns or residents and start practicing medicine. The Doctors Company usually gives a 50% discount on member premiums the first year they’re in practice and a 25% discount credit in their second year. The discounts end after that.
Other insurance carriers offer similar discounts to doctors starting to practice medicine. The deepest one is offered in the first year (at least 50%) and a smaller one (20%-25%) the second year, according to medical malpractice brokers.
“The new-to-practice discount is based solely on when the physician left their formal training to begin their practice for the first time; it is not based on claim-free history,” explained Mr. Zetter.
This is a very common discount used by different insurer carriers, said Dr. Price. “New physicians don’t have the same amount of risk of a lawsuit when they’re starting out. It’s unlikely they will have a claim and most liability actions have a 2-year time limit from the date of injury to be filed.”
2. Take advantage of being claims free
If you’ve been claims free for at least a few years, you may be eligible for a large discount.
“Doctors without claims are a better risk. Once a doctor has one claim, they’re likely to have a second, which the research shows,” said Mr. Zetter.
The most common credit The Doctors Company offers is 3 years of being claim free – this earns doctors up to 25%, he said. Mr. Zetter explained that the criteria and size of The Doctors Company credit may depend on the state where physicians practice.
“We allowed insurance carriers that we acquired to continue with their own claim-free discount program such as Florida’s First Professionals Insurance Company we acquired in 2011,” he said.
Doctors with other medical malpractice insurers may also be eligible for a credit up to 25%. In some instances, they may have to be claims free for 5 or 10 years, say insurance experts.
It pays to shop around before purchasing insurance.
3. If you work part time, make sure your premium reflects that
Physicians who see patients part time can receive up to a 75% discount on their medical liability insurance premiums.
The discounts are based on the hours the physician works per week. The fewer hours worked, the larger the discount. This type of discount does not vary by specialty.
According to The Doctors Company, working 10 hours or less per week may entitle doctors to a 75% discount; working 11-20 hours per week may entitle them to a 50% discount, and working 21-30 hours per week may entitle them to a 25% discount. If you are in this situation, it pays to ask your insurer if there is a discount available to you.
4. Look into your professional medical society insurance company
“I would look at your state medical association [or] state specialty society and talk to your colleagues to learn what premiums they’re paying and about any discounts they’re getting,” advised Mr. Zetter.
Some state medical societies have formed their own liability companies and offer lower premiums to their members because “they’re organized and managed by doctors, which makes their premiums more competitive,” Dr. Price said.
Other state medical societies endorse specific insurance carriers and offer their members a 5% discount for enrolling with them.
5. Enroll in a risk management program
Most insurers offer online educational activities designed to improve patient safety and reduce the risk of a lawsuit. Physicians may be eligible for both premium discounts and CME credits.
Medical Liability Mutual Insurance Company, owned by Berkshire Hathaway, operates in New York and offers physicians a premium discount of up to 5%, CME credit, and maintenance of certification credit for successfully completing its risk management program every other year.
ProAssurance members nationwide can earn 5% in premium discounts if they complete a 2-hour video series called “Back to Basics: Loss Prevention and Navigating Everyday Risks: Using Data to Drive Change.”
They can earn one credit for completing each webinar on topics such as “Medication Management: Minimizing Errors and Improving Safety” and “Opioid Prescribing: Keeping Patients Safe.”
MagMutual offers its insured physicians 1 CME credit for completing their specialty’s risk assessment and courses, which may be applied toward their premium discounts.
The Doctors Company offers its members a 5% premium discount if they complete 4 CME credits. One of its most popular courses is “How To Get Rid of a Difficult Patient.”
“Busy residents like the shorter case studies worth one-quarter credit that they can complete in 15 minutes,” said Ms. Boisvert.
“This is a good bargain from the physician’s standpoint and the fact that risk management education is offered online makes it a lot easier than going to a seminar in person,” said Dr. Price.
A version of this article first appeared on Medscape.com.
Getting a better deal might simply mean taking advantage of incentives and discounts your insurer may already offer. These include claims-free, new-to-practice, and working part-time discounts.
However, if you decide to shop around, keep in mind that discounts are just one factor that can affect your premium price – insurers look at your specialty, location, and claims history.
One of the most common ways physicians can earn discounts is by participating in risk management programs. With this type of program, physicians evaluate elements of their practice and documentation practices and identify areas that might leave them at risk for a lawsuit. While they save money, physician risk management programs also are designed to reduce malpractice claims, which ultimately minimizes the potential for bigger financial losses, insurance experts say.
“It’s a win-win situation when liability insurers and physicians work together to minimize risk, and it’s a win for patients,” said Gary Price, MD, president of The Physicians Foundation.
Doctors in private practice or employed by small hospitals that are not self-insured can qualify for these discounts, said David Zetter, president of Zetter HealthCare Management Consultants.
“I do a lot of work with medical malpractice companies trying to find clients policies. All the carriers are transparent about what physicians have to do to lower their premiums. Physicians can receive the discounts if they follow through and meet the insurer’s requirements,” said Mr. Zetter.
State insurance departments regulate medical malpractice insurance, including the premium credits insurers offer. Most states cap discounts at 25%, but some go as high as 70%, according to The Doctors Company, a national physician-owned medical malpractice insurer.
Insurers typically offer doctors several ways to earn discounts. The size of the discount also can depend on whether a doctor is new to a practice, remains claims free, or takes risk management courses.
In addition to the premium discount, some online risk management classes and webinars are eligible for CME credits.
“The credits can add up and they can be used for recertification or relicensure,” said Susan Boisvert, senior patient safety risk manager at The Doctors Company.
Here are five ways you may qualify for discounts with your insurer.
1. Make use of discounts available to new doctors
Doctors can earn hefty discounts on their premiums when they are no longer interns or residents and start practicing medicine. The Doctors Company usually gives a 50% discount on member premiums the first year they’re in practice and a 25% discount credit in their second year. The discounts end after that.
Other insurance carriers offer similar discounts to doctors starting to practice medicine. The deepest one is offered in the first year (at least 50%) and a smaller one (20%-25%) the second year, according to medical malpractice brokers.
“The new-to-practice discount is based solely on when the physician left their formal training to begin their practice for the first time; it is not based on claim-free history,” explained Mr. Zetter.
This is a very common discount used by different insurer carriers, said Dr. Price. “New physicians don’t have the same amount of risk of a lawsuit when they’re starting out. It’s unlikely they will have a claim and most liability actions have a 2-year time limit from the date of injury to be filed.”
2. Take advantage of being claims free
If you’ve been claims free for at least a few years, you may be eligible for a large discount.
“Doctors without claims are a better risk. Once a doctor has one claim, they’re likely to have a second, which the research shows,” said Mr. Zetter.
The most common credit The Doctors Company offers is 3 years of being claim free – this earns doctors up to 25%, he said. Mr. Zetter explained that the criteria and size of The Doctors Company credit may depend on the state where physicians practice.
“We allowed insurance carriers that we acquired to continue with their own claim-free discount program such as Florida’s First Professionals Insurance Company we acquired in 2011,” he said.
Doctors with other medical malpractice insurers may also be eligible for a credit up to 25%. In some instances, they may have to be claims free for 5 or 10 years, say insurance experts.
It pays to shop around before purchasing insurance.
3. If you work part time, make sure your premium reflects that
Physicians who see patients part time can receive up to a 75% discount on their medical liability insurance premiums.
The discounts are based on the hours the physician works per week. The fewer hours worked, the larger the discount. This type of discount does not vary by specialty.
According to The Doctors Company, working 10 hours or less per week may entitle doctors to a 75% discount; working 11-20 hours per week may entitle them to a 50% discount, and working 21-30 hours per week may entitle them to a 25% discount. If you are in this situation, it pays to ask your insurer if there is a discount available to you.
4. Look into your professional medical society insurance company
“I would look at your state medical association [or] state specialty society and talk to your colleagues to learn what premiums they’re paying and about any discounts they’re getting,” advised Mr. Zetter.
Some state medical societies have formed their own liability companies and offer lower premiums to their members because “they’re organized and managed by doctors, which makes their premiums more competitive,” Dr. Price said.
Other state medical societies endorse specific insurance carriers and offer their members a 5% discount for enrolling with them.
5. Enroll in a risk management program
Most insurers offer online educational activities designed to improve patient safety and reduce the risk of a lawsuit. Physicians may be eligible for both premium discounts and CME credits.
Medical Liability Mutual Insurance Company, owned by Berkshire Hathaway, operates in New York and offers physicians a premium discount of up to 5%, CME credit, and maintenance of certification credit for successfully completing its risk management program every other year.
ProAssurance members nationwide can earn 5% in premium discounts if they complete a 2-hour video series called “Back to Basics: Loss Prevention and Navigating Everyday Risks: Using Data to Drive Change.”
They can earn one credit for completing each webinar on topics such as “Medication Management: Minimizing Errors and Improving Safety” and “Opioid Prescribing: Keeping Patients Safe.”
MagMutual offers its insured physicians 1 CME credit for completing their specialty’s risk assessment and courses, which may be applied toward their premium discounts.
The Doctors Company offers its members a 5% premium discount if they complete 4 CME credits. One of its most popular courses is “How To Get Rid of a Difficult Patient.”
“Busy residents like the shorter case studies worth one-quarter credit that they can complete in 15 minutes,” said Ms. Boisvert.
“This is a good bargain from the physician’s standpoint and the fact that risk management education is offered online makes it a lot easier than going to a seminar in person,” said Dr. Price.
A version of this article first appeared on Medscape.com.
Getting a better deal might simply mean taking advantage of incentives and discounts your insurer may already offer. These include claims-free, new-to-practice, and working part-time discounts.
However, if you decide to shop around, keep in mind that discounts are just one factor that can affect your premium price – insurers look at your specialty, location, and claims history.
One of the most common ways physicians can earn discounts is by participating in risk management programs. With this type of program, physicians evaluate elements of their practice and documentation practices and identify areas that might leave them at risk for a lawsuit. While they save money, physician risk management programs also are designed to reduce malpractice claims, which ultimately minimizes the potential for bigger financial losses, insurance experts say.
“It’s a win-win situation when liability insurers and physicians work together to minimize risk, and it’s a win for patients,” said Gary Price, MD, president of The Physicians Foundation.
Doctors in private practice or employed by small hospitals that are not self-insured can qualify for these discounts, said David Zetter, president of Zetter HealthCare Management Consultants.
“I do a lot of work with medical malpractice companies trying to find clients policies. All the carriers are transparent about what physicians have to do to lower their premiums. Physicians can receive the discounts if they follow through and meet the insurer’s requirements,” said Mr. Zetter.
State insurance departments regulate medical malpractice insurance, including the premium credits insurers offer. Most states cap discounts at 25%, but some go as high as 70%, according to The Doctors Company, a national physician-owned medical malpractice insurer.
Insurers typically offer doctors several ways to earn discounts. The size of the discount also can depend on whether a doctor is new to a practice, remains claims free, or takes risk management courses.
In addition to the premium discount, some online risk management classes and webinars are eligible for CME credits.
“The credits can add up and they can be used for recertification or relicensure,” said Susan Boisvert, senior patient safety risk manager at The Doctors Company.
Here are five ways you may qualify for discounts with your insurer.
1. Make use of discounts available to new doctors
Doctors can earn hefty discounts on their premiums when they are no longer interns or residents and start practicing medicine. The Doctors Company usually gives a 50% discount on member premiums the first year they’re in practice and a 25% discount credit in their second year. The discounts end after that.
Other insurance carriers offer similar discounts to doctors starting to practice medicine. The deepest one is offered in the first year (at least 50%) and a smaller one (20%-25%) the second year, according to medical malpractice brokers.
“The new-to-practice discount is based solely on when the physician left their formal training to begin their practice for the first time; it is not based on claim-free history,” explained Mr. Zetter.
This is a very common discount used by different insurer carriers, said Dr. Price. “New physicians don’t have the same amount of risk of a lawsuit when they’re starting out. It’s unlikely they will have a claim and most liability actions have a 2-year time limit from the date of injury to be filed.”
2. Take advantage of being claims free
If you’ve been claims free for at least a few years, you may be eligible for a large discount.
“Doctors without claims are a better risk. Once a doctor has one claim, they’re likely to have a second, which the research shows,” said Mr. Zetter.
The most common credit The Doctors Company offers is 3 years of being claim free – this earns doctors up to 25%, he said. Mr. Zetter explained that the criteria and size of The Doctors Company credit may depend on the state where physicians practice.
“We allowed insurance carriers that we acquired to continue with their own claim-free discount program such as Florida’s First Professionals Insurance Company we acquired in 2011,” he said.
Doctors with other medical malpractice insurers may also be eligible for a credit up to 25%. In some instances, they may have to be claims free for 5 or 10 years, say insurance experts.
It pays to shop around before purchasing insurance.
3. If you work part time, make sure your premium reflects that
Physicians who see patients part time can receive up to a 75% discount on their medical liability insurance premiums.
The discounts are based on the hours the physician works per week. The fewer hours worked, the larger the discount. This type of discount does not vary by specialty.
According to The Doctors Company, working 10 hours or less per week may entitle doctors to a 75% discount; working 11-20 hours per week may entitle them to a 50% discount, and working 21-30 hours per week may entitle them to a 25% discount. If you are in this situation, it pays to ask your insurer if there is a discount available to you.
4. Look into your professional medical society insurance company
“I would look at your state medical association [or] state specialty society and talk to your colleagues to learn what premiums they’re paying and about any discounts they’re getting,” advised Mr. Zetter.
Some state medical societies have formed their own liability companies and offer lower premiums to their members because “they’re organized and managed by doctors, which makes their premiums more competitive,” Dr. Price said.
Other state medical societies endorse specific insurance carriers and offer their members a 5% discount for enrolling with them.
5. Enroll in a risk management program
Most insurers offer online educational activities designed to improve patient safety and reduce the risk of a lawsuit. Physicians may be eligible for both premium discounts and CME credits.
Medical Liability Mutual Insurance Company, owned by Berkshire Hathaway, operates in New York and offers physicians a premium discount of up to 5%, CME credit, and maintenance of certification credit for successfully completing its risk management program every other year.
ProAssurance members nationwide can earn 5% in premium discounts if they complete a 2-hour video series called “Back to Basics: Loss Prevention and Navigating Everyday Risks: Using Data to Drive Change.”
They can earn one credit for completing each webinar on topics such as “Medication Management: Minimizing Errors and Improving Safety” and “Opioid Prescribing: Keeping Patients Safe.”
MagMutual offers its insured physicians 1 CME credit for completing their specialty’s risk assessment and courses, which may be applied toward their premium discounts.
The Doctors Company offers its members a 5% premium discount if they complete 4 CME credits. One of its most popular courses is “How To Get Rid of a Difficult Patient.”
“Busy residents like the shorter case studies worth one-quarter credit that they can complete in 15 minutes,” said Ms. Boisvert.
“This is a good bargain from the physician’s standpoint and the fact that risk management education is offered online makes it a lot easier than going to a seminar in person,” said Dr. Price.
A version of this article first appeared on Medscape.com.
Boys may carry the weight, or overweight, of adults’ infertility
Overweight boy, infertile man?
When it comes to causes of infertility, history and science have generally focused on women. A lot of the research overlooks men, but some previous studies have suggested that male infertility contributes to about half of the cases of couple infertility. The reason for much of that male infertility, however, has been a mystery. Until now.
A group of Italian investigators looked at the declining trend in sperm counts over the past 40 years and the increase of childhood obesity. Is there a correlation? The researchers think so. Childhood obesity can be linked to multiple causes, but the researchers zeroed in on the effect that obesity has on metabolic rates and, therefore, testicular growth.
Collecting data on testicular volume, body mass index (BMI), and insulin resistance from 268 boys aged 2-18 years, the researchers discovered that those with normal weight and normal insulin levels had testicular volumes 1.5 times higher than their overweight counterparts and 1.5-2 times higher than those with hyperinsulinemia, building a case for obesity being a factor for infertility later in life.
Since low testicular volume is associated with lower sperm count and production as an adult, putting two and two together makes a compelling argument for childhood obesity being a major male infertility culprit. It also creates even more urgency for the health care industry and community decision makers to focus on childhood obesity.
It sure would be nice to be able to take one of the many risk factors for future human survival off the table. Maybe by taking something, like cake, off the table.
Fecal transplantation moves to the kitchen
Fecal microbiota transplantation is an effective way to treat Clostridioides difficile infection, but, in the end, it’s still a transplantation procedure involving a nasogastric or colorectal tube or rather large oral capsules with a demanding (30-40 capsules over 2 days) dosage. Please, Science, tell us there’s a better way.
Science, in the form of investigators at the University of Geneva and Lausanne University Hospital in Switzerland, has spoken, and there may be a better way. Presenting fecal beads: All the bacterial goodness of donor stool without the tubal insertions or massive quantities of giant capsules.
We know you’re scoffing out there, but it’s true. All you need is a little alginate, which is a “biocompatible polysaccharide isolated from brown algae” of the Phaeophyceae family. The donor feces is microencapsulated by mixing it with the alginate, dropping that mixture into water containing calcium chloride, turning it into a gel, and then freeze-drying the gel into small (just 2 mm), solid beads.
Sounds plausible enough, but what do you do with them? “These brownish beads can be easily dispersed in a liquid or food that is pleasant to eat. They also have no taste,” senior author Eric Allémann, PhD, said in a statement released by the University of Geneva.
Pleasant to eat? No taste? So which is it? If you really want to know, watch fecal beads week on the new season of “The Great British Baking Show,” when Paul and Prue judge poop baked into crumpets, crepes, and crostatas. Yum.
We’re on the low-oxygen diet
Nine out of ten doctors agree: Oxygen is more important to your continued well-being than food. After all, a human can go weeks without food, but just minutes without oxygen. However, ten out of ten doctors agree that the United States has an obesity problem. They all also agree that previous research has shown soldiers who train at high altitudes lose more weight than those training at lower altitudes.
So, on the one hand, we have a country full of overweight people, and on the other, we have low oxygen levels causing weight loss. The solution, then, is obvious: Stop breathing.
More specifically (and somewhat less facetiously), researchers from Louisiana have launched the Low Oxygen and Weight Status trial and are currently recruiting individuals with BMIs of 30-40 to, uh, suffocate themselves. No, no, it’s okay, it’s just when they’re sleeping.
Fine, straight face. Participants in the LOWS trial will undergo an 8-week period when they will consume a controlled weight-loss diet and spend their nights in a hypoxic sealed tent, where they will sleep in an environment with an oxygen level equivalent to 8,500 feet above sea level (roughly equivalent to Aspen, Colo.). They will be compared with people on the same diet who sleep in a normal, sea-level oxygen environment.
The study’s goal is to determine whether or not spending time in a low-oxygen environment will suppress appetite, increase energy expenditure, and improve weight loss and insulin sensitivity. Excessive weight loss in high-altitude environments isn’t a good thing for soldiers – they kind of need their muscles and body weight to do the whole soldiering thing – but it could be great for people struggling to lose those last few pounds. And it also may prove LOTME’s previous thesis: Air is not good.
Overweight boy, infertile man?
When it comes to causes of infertility, history and science have generally focused on women. A lot of the research overlooks men, but some previous studies have suggested that male infertility contributes to about half of the cases of couple infertility. The reason for much of that male infertility, however, has been a mystery. Until now.
A group of Italian investigators looked at the declining trend in sperm counts over the past 40 years and the increase of childhood obesity. Is there a correlation? The researchers think so. Childhood obesity can be linked to multiple causes, but the researchers zeroed in on the effect that obesity has on metabolic rates and, therefore, testicular growth.
Collecting data on testicular volume, body mass index (BMI), and insulin resistance from 268 boys aged 2-18 years, the researchers discovered that those with normal weight and normal insulin levels had testicular volumes 1.5 times higher than their overweight counterparts and 1.5-2 times higher than those with hyperinsulinemia, building a case for obesity being a factor for infertility later in life.
Since low testicular volume is associated with lower sperm count and production as an adult, putting two and two together makes a compelling argument for childhood obesity being a major male infertility culprit. It also creates even more urgency for the health care industry and community decision makers to focus on childhood obesity.
It sure would be nice to be able to take one of the many risk factors for future human survival off the table. Maybe by taking something, like cake, off the table.
Fecal transplantation moves to the kitchen
Fecal microbiota transplantation is an effective way to treat Clostridioides difficile infection, but, in the end, it’s still a transplantation procedure involving a nasogastric or colorectal tube or rather large oral capsules with a demanding (30-40 capsules over 2 days) dosage. Please, Science, tell us there’s a better way.
Science, in the form of investigators at the University of Geneva and Lausanne University Hospital in Switzerland, has spoken, and there may be a better way. Presenting fecal beads: All the bacterial goodness of donor stool without the tubal insertions or massive quantities of giant capsules.
We know you’re scoffing out there, but it’s true. All you need is a little alginate, which is a “biocompatible polysaccharide isolated from brown algae” of the Phaeophyceae family. The donor feces is microencapsulated by mixing it with the alginate, dropping that mixture into water containing calcium chloride, turning it into a gel, and then freeze-drying the gel into small (just 2 mm), solid beads.
Sounds plausible enough, but what do you do with them? “These brownish beads can be easily dispersed in a liquid or food that is pleasant to eat. They also have no taste,” senior author Eric Allémann, PhD, said in a statement released by the University of Geneva.
Pleasant to eat? No taste? So which is it? If you really want to know, watch fecal beads week on the new season of “The Great British Baking Show,” when Paul and Prue judge poop baked into crumpets, crepes, and crostatas. Yum.
We’re on the low-oxygen diet
Nine out of ten doctors agree: Oxygen is more important to your continued well-being than food. After all, a human can go weeks without food, but just minutes without oxygen. However, ten out of ten doctors agree that the United States has an obesity problem. They all also agree that previous research has shown soldiers who train at high altitudes lose more weight than those training at lower altitudes.
So, on the one hand, we have a country full of overweight people, and on the other, we have low oxygen levels causing weight loss. The solution, then, is obvious: Stop breathing.
More specifically (and somewhat less facetiously), researchers from Louisiana have launched the Low Oxygen and Weight Status trial and are currently recruiting individuals with BMIs of 30-40 to, uh, suffocate themselves. No, no, it’s okay, it’s just when they’re sleeping.
Fine, straight face. Participants in the LOWS trial will undergo an 8-week period when they will consume a controlled weight-loss diet and spend their nights in a hypoxic sealed tent, where they will sleep in an environment with an oxygen level equivalent to 8,500 feet above sea level (roughly equivalent to Aspen, Colo.). They will be compared with people on the same diet who sleep in a normal, sea-level oxygen environment.
The study’s goal is to determine whether or not spending time in a low-oxygen environment will suppress appetite, increase energy expenditure, and improve weight loss and insulin sensitivity. Excessive weight loss in high-altitude environments isn’t a good thing for soldiers – they kind of need their muscles and body weight to do the whole soldiering thing – but it could be great for people struggling to lose those last few pounds. And it also may prove LOTME’s previous thesis: Air is not good.
Overweight boy, infertile man?
When it comes to causes of infertility, history and science have generally focused on women. A lot of the research overlooks men, but some previous studies have suggested that male infertility contributes to about half of the cases of couple infertility. The reason for much of that male infertility, however, has been a mystery. Until now.
A group of Italian investigators looked at the declining trend in sperm counts over the past 40 years and the increase of childhood obesity. Is there a correlation? The researchers think so. Childhood obesity can be linked to multiple causes, but the researchers zeroed in on the effect that obesity has on metabolic rates and, therefore, testicular growth.
Collecting data on testicular volume, body mass index (BMI), and insulin resistance from 268 boys aged 2-18 years, the researchers discovered that those with normal weight and normal insulin levels had testicular volumes 1.5 times higher than their overweight counterparts and 1.5-2 times higher than those with hyperinsulinemia, building a case for obesity being a factor for infertility later in life.
Since low testicular volume is associated with lower sperm count and production as an adult, putting two and two together makes a compelling argument for childhood obesity being a major male infertility culprit. It also creates even more urgency for the health care industry and community decision makers to focus on childhood obesity.
It sure would be nice to be able to take one of the many risk factors for future human survival off the table. Maybe by taking something, like cake, off the table.
Fecal transplantation moves to the kitchen
Fecal microbiota transplantation is an effective way to treat Clostridioides difficile infection, but, in the end, it’s still a transplantation procedure involving a nasogastric or colorectal tube or rather large oral capsules with a demanding (30-40 capsules over 2 days) dosage. Please, Science, tell us there’s a better way.
Science, in the form of investigators at the University of Geneva and Lausanne University Hospital in Switzerland, has spoken, and there may be a better way. Presenting fecal beads: All the bacterial goodness of donor stool without the tubal insertions or massive quantities of giant capsules.
We know you’re scoffing out there, but it’s true. All you need is a little alginate, which is a “biocompatible polysaccharide isolated from brown algae” of the Phaeophyceae family. The donor feces is microencapsulated by mixing it with the alginate, dropping that mixture into water containing calcium chloride, turning it into a gel, and then freeze-drying the gel into small (just 2 mm), solid beads.
Sounds plausible enough, but what do you do with them? “These brownish beads can be easily dispersed in a liquid or food that is pleasant to eat. They also have no taste,” senior author Eric Allémann, PhD, said in a statement released by the University of Geneva.
Pleasant to eat? No taste? So which is it? If you really want to know, watch fecal beads week on the new season of “The Great British Baking Show,” when Paul and Prue judge poop baked into crumpets, crepes, and crostatas. Yum.
We’re on the low-oxygen diet
Nine out of ten doctors agree: Oxygen is more important to your continued well-being than food. After all, a human can go weeks without food, but just minutes without oxygen. However, ten out of ten doctors agree that the United States has an obesity problem. They all also agree that previous research has shown soldiers who train at high altitudes lose more weight than those training at lower altitudes.
So, on the one hand, we have a country full of overweight people, and on the other, we have low oxygen levels causing weight loss. The solution, then, is obvious: Stop breathing.
More specifically (and somewhat less facetiously), researchers from Louisiana have launched the Low Oxygen and Weight Status trial and are currently recruiting individuals with BMIs of 30-40 to, uh, suffocate themselves. No, no, it’s okay, it’s just when they’re sleeping.
Fine, straight face. Participants in the LOWS trial will undergo an 8-week period when they will consume a controlled weight-loss diet and spend their nights in a hypoxic sealed tent, where they will sleep in an environment with an oxygen level equivalent to 8,500 feet above sea level (roughly equivalent to Aspen, Colo.). They will be compared with people on the same diet who sleep in a normal, sea-level oxygen environment.
The study’s goal is to determine whether or not spending time in a low-oxygen environment will suppress appetite, increase energy expenditure, and improve weight loss and insulin sensitivity. Excessive weight loss in high-altitude environments isn’t a good thing for soldiers – they kind of need their muscles and body weight to do the whole soldiering thing – but it could be great for people struggling to lose those last few pounds. And it also may prove LOTME’s previous thesis: Air is not good.
Low disease state for childhood lupus approaches validation
MANCHESTER, ENGLAND – An age-appropriate version of the Lupus Low Disease Activity State (LLDAS) has been developed by an international task force that will hopefully enable childhood-onset systemic lupus erythematosus (cSLE) to be treated to target in the near future.
The new childhood LLDAS (cLLDAS) has been purposefully developed to align with that already used for adults, Eve Smith, MBChB, PhD, explained at the annual meeting of the British Society for Rheumatology.
“There’s a lot of compelling data that’s accumulating from adult lupus and increasingly from childhood lupus that [treat to target] might be a good idea,” said Dr. Smith, who is a senior clinical fellow and honorary consultant at the University of Liverpool (England) and Alder Hey Children’s NHS Foundation Trust Hospital, also in Liverpool.
Urgent need to improve childhood lupus outcomes
“We urgently need to do something to try and improve outcomes for children,” Dr. Smith said.
“We know that childhood lupus patients have got higher disease activity as compared to adults; they have a greater medication burden, particularly steroids; and they tend to have more severe organ manifestations,” she added.
Moreover, data show that one-fifth of pediatric patients with lupus have already accrued early damage, and there is much higher mortality associated with childhood lupus than there is with adult lupus.
“So, really we want to use treat to target as a way to try and improve on these aspects,” Dr. Smith said.
The treat-to-target (T2T) approach is not a new idea in lupus, with a lot of work already done in adult patients. One large study of more than 3,300 patients conducted in 13 countries has shown that patients who never achieve LLDAS are more likely to have high levels of damage, greater glucocorticoid use, worse quality of life, and higher mortality than are those who do.
Conversely, data have also shown that achieving a LLDAS is associated with a reduction in the risk for new damage, flares, and hospitalization, as well as reducing health care costs and improving patients’ overall health-related quality of life.
T2T is a recognized approach in European adult SLE guidelines, Dr. Smith said, although the approach has not really been fully realized as of yet, even in adult practice.
The cSLE T2T international task force and cLLDAS definition
With evidence accumulating on the benefits of getting children with SLE to a low disease activity state, Dr. Smith and colleague Michael Beresford, MBChB, PhD, Brough Chair, Professor of Child Health at the University of Liverpool, put out a call to develop a task force to look into the feasibility of a T2T approach.
“We had a really enthusiastic response internationally, which we were really encouraged by,” Dr. Smith said, “and we now lead a task force of 20 experts from across all five continents, and we have really strong patient involvement.”
Through a consensus process, an international cSLE T2T Task Force agreed on overarching principles and points to consider that will “lay the foundation for future T2T approaches in cSLE,” according to the recommendations statement, which was endorsed by the Paediatric Rheumatology European Society.
Next, they looked to develop an age-appropriate definition for low disease activity.
“We’re deliberately wanting to maintain sufficient unity with the adult definition, so that we could facilitate life-course studies,” said Dr. Smith, who presented the results of a literature review and series of Delphi surveys at the meeting.
The conceptual definition of cLLDAS is similar to adults in describing it as a sustained state that is associated with a low likelihood of adverse outcome, Dr. Smith said, but with the added wording of “considering disease activity, damage, and medication toxicity.”
The definition is achieved when the SLE Disease Activity Index-2K is ≤ 4 and there is no activity in major organ systems; there are no new features of lupus disease activity since the last assessment; there is a score of ≤ 1 on Physician Global Assessment; steroid doses are ≤ 0.15 mg/kg/day or a maximum of 7.5 mg/day (whichever is lower); and immunosuppressive treatment is stable, with any changes to medication only because of side effects, adherence, changes in weight, or when in the process of reaching a target dose.
“It’s all very well having a definition, but you need to think about how that will work in practice,” Dr. Smith said. This is something that the task force is thinking about very carefully.
The task force next aims to validate the cLLDAS definition, form an extensive research agenda to inform the T2T methods, and develop innovative methods to apply the approach in practice.
The work is supported by the Wellcome Trust, National Institutes for Health Research, Versus Arthritis, and the University of Liverpool, Alder Hey Children’s NHS Foundation Trust and the Alder Hey Charity. Dr. Smith reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
MANCHESTER, ENGLAND – An age-appropriate version of the Lupus Low Disease Activity State (LLDAS) has been developed by an international task force that will hopefully enable childhood-onset systemic lupus erythematosus (cSLE) to be treated to target in the near future.
The new childhood LLDAS (cLLDAS) has been purposefully developed to align with that already used for adults, Eve Smith, MBChB, PhD, explained at the annual meeting of the British Society for Rheumatology.
“There’s a lot of compelling data that’s accumulating from adult lupus and increasingly from childhood lupus that [treat to target] might be a good idea,” said Dr. Smith, who is a senior clinical fellow and honorary consultant at the University of Liverpool (England) and Alder Hey Children’s NHS Foundation Trust Hospital, also in Liverpool.
Urgent need to improve childhood lupus outcomes
“We urgently need to do something to try and improve outcomes for children,” Dr. Smith said.
“We know that childhood lupus patients have got higher disease activity as compared to adults; they have a greater medication burden, particularly steroids; and they tend to have more severe organ manifestations,” she added.
Moreover, data show that one-fifth of pediatric patients with lupus have already accrued early damage, and there is much higher mortality associated with childhood lupus than there is with adult lupus.
“So, really we want to use treat to target as a way to try and improve on these aspects,” Dr. Smith said.
The treat-to-target (T2T) approach is not a new idea in lupus, with a lot of work already done in adult patients. One large study of more than 3,300 patients conducted in 13 countries has shown that patients who never achieve LLDAS are more likely to have high levels of damage, greater glucocorticoid use, worse quality of life, and higher mortality than are those who do.
Conversely, data have also shown that achieving a LLDAS is associated with a reduction in the risk for new damage, flares, and hospitalization, as well as reducing health care costs and improving patients’ overall health-related quality of life.
T2T is a recognized approach in European adult SLE guidelines, Dr. Smith said, although the approach has not really been fully realized as of yet, even in adult practice.
The cSLE T2T international task force and cLLDAS definition
With evidence accumulating on the benefits of getting children with SLE to a low disease activity state, Dr. Smith and colleague Michael Beresford, MBChB, PhD, Brough Chair, Professor of Child Health at the University of Liverpool, put out a call to develop a task force to look into the feasibility of a T2T approach.
“We had a really enthusiastic response internationally, which we were really encouraged by,” Dr. Smith said, “and we now lead a task force of 20 experts from across all five continents, and we have really strong patient involvement.”
Through a consensus process, an international cSLE T2T Task Force agreed on overarching principles and points to consider that will “lay the foundation for future T2T approaches in cSLE,” according to the recommendations statement, which was endorsed by the Paediatric Rheumatology European Society.
Next, they looked to develop an age-appropriate definition for low disease activity.
“We’re deliberately wanting to maintain sufficient unity with the adult definition, so that we could facilitate life-course studies,” said Dr. Smith, who presented the results of a literature review and series of Delphi surveys at the meeting.
The conceptual definition of cLLDAS is similar to adults in describing it as a sustained state that is associated with a low likelihood of adverse outcome, Dr. Smith said, but with the added wording of “considering disease activity, damage, and medication toxicity.”
The definition is achieved when the SLE Disease Activity Index-2K is ≤ 4 and there is no activity in major organ systems; there are no new features of lupus disease activity since the last assessment; there is a score of ≤ 1 on Physician Global Assessment; steroid doses are ≤ 0.15 mg/kg/day or a maximum of 7.5 mg/day (whichever is lower); and immunosuppressive treatment is stable, with any changes to medication only because of side effects, adherence, changes in weight, or when in the process of reaching a target dose.
“It’s all very well having a definition, but you need to think about how that will work in practice,” Dr. Smith said. This is something that the task force is thinking about very carefully.
The task force next aims to validate the cLLDAS definition, form an extensive research agenda to inform the T2T methods, and develop innovative methods to apply the approach in practice.
The work is supported by the Wellcome Trust, National Institutes for Health Research, Versus Arthritis, and the University of Liverpool, Alder Hey Children’s NHS Foundation Trust and the Alder Hey Charity. Dr. Smith reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
MANCHESTER, ENGLAND – An age-appropriate version of the Lupus Low Disease Activity State (LLDAS) has been developed by an international task force that will hopefully enable childhood-onset systemic lupus erythematosus (cSLE) to be treated to target in the near future.
The new childhood LLDAS (cLLDAS) has been purposefully developed to align with that already used for adults, Eve Smith, MBChB, PhD, explained at the annual meeting of the British Society for Rheumatology.
“There’s a lot of compelling data that’s accumulating from adult lupus and increasingly from childhood lupus that [treat to target] might be a good idea,” said Dr. Smith, who is a senior clinical fellow and honorary consultant at the University of Liverpool (England) and Alder Hey Children’s NHS Foundation Trust Hospital, also in Liverpool.
Urgent need to improve childhood lupus outcomes
“We urgently need to do something to try and improve outcomes for children,” Dr. Smith said.
“We know that childhood lupus patients have got higher disease activity as compared to adults; they have a greater medication burden, particularly steroids; and they tend to have more severe organ manifestations,” she added.
Moreover, data show that one-fifth of pediatric patients with lupus have already accrued early damage, and there is much higher mortality associated with childhood lupus than there is with adult lupus.
“So, really we want to use treat to target as a way to try and improve on these aspects,” Dr. Smith said.
The treat-to-target (T2T) approach is not a new idea in lupus, with a lot of work already done in adult patients. One large study of more than 3,300 patients conducted in 13 countries has shown that patients who never achieve LLDAS are more likely to have high levels of damage, greater glucocorticoid use, worse quality of life, and higher mortality than are those who do.
Conversely, data have also shown that achieving a LLDAS is associated with a reduction in the risk for new damage, flares, and hospitalization, as well as reducing health care costs and improving patients’ overall health-related quality of life.
T2T is a recognized approach in European adult SLE guidelines, Dr. Smith said, although the approach has not really been fully realized as of yet, even in adult practice.
The cSLE T2T international task force and cLLDAS definition
With evidence accumulating on the benefits of getting children with SLE to a low disease activity state, Dr. Smith and colleague Michael Beresford, MBChB, PhD, Brough Chair, Professor of Child Health at the University of Liverpool, put out a call to develop a task force to look into the feasibility of a T2T approach.
“We had a really enthusiastic response internationally, which we were really encouraged by,” Dr. Smith said, “and we now lead a task force of 20 experts from across all five continents, and we have really strong patient involvement.”
Through a consensus process, an international cSLE T2T Task Force agreed on overarching principles and points to consider that will “lay the foundation for future T2T approaches in cSLE,” according to the recommendations statement, which was endorsed by the Paediatric Rheumatology European Society.
Next, they looked to develop an age-appropriate definition for low disease activity.
“We’re deliberately wanting to maintain sufficient unity with the adult definition, so that we could facilitate life-course studies,” said Dr. Smith, who presented the results of a literature review and series of Delphi surveys at the meeting.
The conceptual definition of cLLDAS is similar to adults in describing it as a sustained state that is associated with a low likelihood of adverse outcome, Dr. Smith said, but with the added wording of “considering disease activity, damage, and medication toxicity.”
The definition is achieved when the SLE Disease Activity Index-2K is ≤ 4 and there is no activity in major organ systems; there are no new features of lupus disease activity since the last assessment; there is a score of ≤ 1 on Physician Global Assessment; steroid doses are ≤ 0.15 mg/kg/day or a maximum of 7.5 mg/day (whichever is lower); and immunosuppressive treatment is stable, with any changes to medication only because of side effects, adherence, changes in weight, or when in the process of reaching a target dose.
“It’s all very well having a definition, but you need to think about how that will work in practice,” Dr. Smith said. This is something that the task force is thinking about very carefully.
The task force next aims to validate the cLLDAS definition, form an extensive research agenda to inform the T2T methods, and develop innovative methods to apply the approach in practice.
The work is supported by the Wellcome Trust, National Institutes for Health Research, Versus Arthritis, and the University of Liverpool, Alder Hey Children’s NHS Foundation Trust and the Alder Hey Charity. Dr. Smith reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
AT BSR 2023
Clinical trials: Top priority for long COVID
The Centers for Disease Control and Prevention and the U.S. Census Bureau estimate that 6.1% of the U.S. adult population is living with long COVID, with millions more debilitated worldwide. The demand for substantial treatment is enormous, but the urgency to fund and begin the necessary range of clinical trials has not met the severity of the problem.
While trials are slowly beginning to happen, the treatment choices and trial design require crucial nuances and understanding of viral-onset illnesses, and few research groups are creating strong trials that fully reflect the complexities of this landscape.
These recommendations recognize that roughly half of long COVID patients have new-onset myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS) and dysautonomia from COVID, which must be at the forefront of how trials are designed and conducted, and are additionally based on the current hypotheses about long COVID’s pathophysiologies.
1: Drugs proposed by experts in postviral fields should be prioritized
Upward of 50 drugs for viral-onset conditions like ME/CFS, dysautonomia, AIDS, and others have been waiting for years to go to trial, but have not had the funding to do so.
Treatments proposed by experts in viral-onset illnesses (such as ME/CFS and dysautonomia) should be prioritized (PM R. 2022 Oct;14[10]:1270-91), as outside researchers are not familiar with these fields and their potential treatment options.
2: Drugs targeting a wide range of mechanisms should be trialed
Treatments that should be trialed include anticoagulants/antiplatelets for clotting and vascular functioning, immunomodulators including JAK-STAT inhibitors, COVID-specific antivirals and antivirals against reactivated herpesviruses (Valcyte, Valacyclovir, EBV vaccine).
Other options include prescription mast cell stabilizers (ketotifen, cromolyn sodium), drugs that regulate microglial activation (low-dose naltrexone, low-dose aripiprazole), anti-CGRP medications, beta-blockers, and intravenous immunoglobulin.
Others include medications that target mitochondrial dysfunction; ivabradine; pyridostigmine;, DRP1 inhibitors; supplements showing success in patient communities including lactoferrin, ubiquinone, and nattokinase; and therapies targeting glymphatic/lymphatic dysfunction, microbiome therapies, and therapeutic peptides.
3: Use appropriate long COVID subtypes
Long COVID is an umbrella term that encompasses multiple new-onset and worsened conditions and symptoms after COVID. Roughly half of long COVID patients likely meet the criteria for ME/CFS and/or dysautonomia. Others may have new-onset diabetes, major clotting events, lung damage, neurological disorders, loss of smell or taste, and other manifestations.
Patients in different categories likely have different responses to treatments. It’s critical to identify appropriate subtypes for each trial, ideally performing detailed analyses to identify the treatments that work best, and don’t, for each subtype.
4: Behavioral treatments, especially those that have harmed similar populations, should not be trialed
Behavioral treatments including exercise, graded exercise therapy (GET), and cognitive-behavioral therapy (CBT) should not be trialed, let alone prioritized, for long COVID.
In patients with postexertional malaise (PEM), one of the most common long COVID symptoms, exercise is actively harmful and causes dysfunctional metabolic patterns, cardiac preload failure, impaired systemic oxygen extraction, and more. GET and CBT have failed similar populations , and exercise is explicitly contraindicated by the World Health Organization, the British National Institute for Health and Care Excellence, the CDC, and other organizations.
Resources should instead be put toward the wide range of medications that have not yet adequately undergone clinical trials.
5: PCR and antibody tests should not be used as inclusion criteria for trial participants
Only an estimated 1%-3% of cases in the first wave of COVID were documented, and the CDC estimates that only 25% of cases through September 2021 were documented. Similarly, antibody tests are unreliable to determine past infection, as roughly a third of patients don’t seroconvert, and a similar proportion serorevert within a few months. Using polymerase chain reaction (PCR) and antibody testing to determine who should be included in clinical trials limits who is eligible to participate in research, particularly those who have been ill for longer. Additionally, the majority of those who serorevert are women, so using antibody tests for inclusion introduces a selection bias and may miss mechanisms of immune system functioning that are part of long COVID.
PCR tests also have high false-negative rates and requiring them in research excludes people with lower viral loads with long COVID, which would confound findings.
These issues with testing also lead to COVID-infected people accidentally being included in control groups, which ruins the credibility of the research findings completely.
6: Include comparator groups
There are several common diagnoses that occur in people with long COVID, including ME/CFS, postural orthostatic tachycardia syndrome, small-fiber neuropathy, mast cell activation syndrome, and Ehlers-Danlos syndrome.
Identifying people with these conditions within the trial cohort improves research across all fields, benefiting all groups, and helps clarify what types of patients benefit most from certain medications.
7: Identify the right endpoints; avoid the wrong ones
Even though our understanding of the pathophysiology of long COVID is still evolving, it’s still possible to do clinical trials by identifying strong endpoints and outcome measures.
Several tools have been designed for viral-onset conditions and should be used alongside other endpoints. Postexertional malaise and autonomic symptoms, which are some of the most common symptoms of long COVID, can be measured with the validated DSQ-PEM and COMPASS-31, respectively. Tools for cognitive dysfunction trials should capture specific and common types of impairment, like processing speed.
Endpoints should be high-impact and aim for large improvements that have clinical significance over small improvements that do not have clinical significance.
Objective tests should be incorporated where possible; some to consider include natural killer cell functioning, cerebral blood flow, T-cell functioning, levels of reactivated herpesviruses, blood lactate levels, and microclots, as testing becomes available.
Mental health outcomes shouldn’t be primary endpoints, except where a trial is targeting a specific mental health condition because of COVID (for example, premenstrual dysphoric disorder).
If mental health conditions are tracked secondarily, it’s vital not to use questionnaires that include physical symptoms like fatigue, difficulty concentrating, difficulty sleeping, or palpitations, as these artificially increase depression and anxiety scores in chronically ill respondents. Tools that include physical symptoms (Patient Health Questionnaire–9, Beck Anxiety Inventory, Beck Depression Inventory) can be replaced with scales like the PHQ-2, General Anxiety Disorder–7, Hospital Anxiety and Depression Scale, or PROMIS-29 subscales.
Because certain cytokines and other inflammatory markers may naturally decrease over time without corresponding improvement in the ME/CFS subtype, caution should be taken when using cytokines as endpoints.
8: Consider enrollment and objectives carefully
A proportion of people with long COVID will recover in the early months after infection. Ideally, clinical trials will primarily study treatments in patients who have been ill 6 months or longer, as some natural recovery will happen before that can bias studies.
But where resources are abundant, it is ideal for trials to additionally look at whether the treatments can help patients in the early months recover and prevent progression to the later stage.
9: Tracking illness duration is crucial
Research from ME/CFS shows that there may be an immune change in the first few years of the illness, where cytokines decrease without any corresponding change in symptom improvement.
Because of this and the possibility that other markers follow the same pattern, disease duration should be a core feature of all analyses and trial designs. Trial outcomes should be designed to answer the question of whether the medication helps patients at different durations of illness.
10: Prioritize patient populations less likely to recover without intervention
Some long COVID phenotypes seem less likely to recover without intervention. Trials should take care to focus on these patient populations, which include those with neurologic symptoms and those meeting ME/CFS criteria.
11: Account for the relapsing/remitting nature
Outcome measures need to be assessed in a way that can distinguish a temporary remission, which is part of the natural course of the disease, from a permanent cure.
Factors that can contribute to the relapsing/remitting nature include physical and cognitive postexertional malaise, menstrual cycle changes, and seasonal changes.
12: Trial participants should reflect the diversity of the long COVID population
Certain demographics are more likely to be affected by acute and long COVID and need to be appropriately recruited and reflected in research, including in patient engagement.
Trials must include high numbers of Hispanic/Latinx, Black, and indigenous communities, queer and transgender populations, and women. Trial materials and design need to incorporate linguistic diversity in addition to racial/ethnic diversity.
Upward of 75% of long COVID cases happen after mild acute cases; clinical researchers should ensure that nonhospitalized patients make up the bulk of trial participants.
13: Utilize meaningful engagement of patients, especially in treatment selection and study design
Meaningful patient engagement means engaging multiple patients at every step of the trial process, from treatment selection to study design to analysis to communication of the results.
Patient experiences are extremely valuable and contain information that researchers may not be familiar with, including the nature and patterns of the illness, insights into possible treatments, and barriers to documentation and care that may also impact research. Tapping into those patient experiences will make trials stronger.
Overall, the landscape of long COVID clinical trials is ripe for discovery, and researchers choosing to go down this path will be deeply appreciated by the patient community.
Hannah Davis is a long COVID patient-researcher and cofounder of the Patient-Led Research Collaborative, an organization studying the long-term effects of COVID.
A version of this article first appeared on Medscape.com.
The Centers for Disease Control and Prevention and the U.S. Census Bureau estimate that 6.1% of the U.S. adult population is living with long COVID, with millions more debilitated worldwide. The demand for substantial treatment is enormous, but the urgency to fund and begin the necessary range of clinical trials has not met the severity of the problem.
While trials are slowly beginning to happen, the treatment choices and trial design require crucial nuances and understanding of viral-onset illnesses, and few research groups are creating strong trials that fully reflect the complexities of this landscape.
These recommendations recognize that roughly half of long COVID patients have new-onset myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS) and dysautonomia from COVID, which must be at the forefront of how trials are designed and conducted, and are additionally based on the current hypotheses about long COVID’s pathophysiologies.
1: Drugs proposed by experts in postviral fields should be prioritized
Upward of 50 drugs for viral-onset conditions like ME/CFS, dysautonomia, AIDS, and others have been waiting for years to go to trial, but have not had the funding to do so.
Treatments proposed by experts in viral-onset illnesses (such as ME/CFS and dysautonomia) should be prioritized (PM R. 2022 Oct;14[10]:1270-91), as outside researchers are not familiar with these fields and their potential treatment options.
2: Drugs targeting a wide range of mechanisms should be trialed
Treatments that should be trialed include anticoagulants/antiplatelets for clotting and vascular functioning, immunomodulators including JAK-STAT inhibitors, COVID-specific antivirals and antivirals against reactivated herpesviruses (Valcyte, Valacyclovir, EBV vaccine).
Other options include prescription mast cell stabilizers (ketotifen, cromolyn sodium), drugs that regulate microglial activation (low-dose naltrexone, low-dose aripiprazole), anti-CGRP medications, beta-blockers, and intravenous immunoglobulin.
Others include medications that target mitochondrial dysfunction; ivabradine; pyridostigmine;, DRP1 inhibitors; supplements showing success in patient communities including lactoferrin, ubiquinone, and nattokinase; and therapies targeting glymphatic/lymphatic dysfunction, microbiome therapies, and therapeutic peptides.
3: Use appropriate long COVID subtypes
Long COVID is an umbrella term that encompasses multiple new-onset and worsened conditions and symptoms after COVID. Roughly half of long COVID patients likely meet the criteria for ME/CFS and/or dysautonomia. Others may have new-onset diabetes, major clotting events, lung damage, neurological disorders, loss of smell or taste, and other manifestations.
Patients in different categories likely have different responses to treatments. It’s critical to identify appropriate subtypes for each trial, ideally performing detailed analyses to identify the treatments that work best, and don’t, for each subtype.
4: Behavioral treatments, especially those that have harmed similar populations, should not be trialed
Behavioral treatments including exercise, graded exercise therapy (GET), and cognitive-behavioral therapy (CBT) should not be trialed, let alone prioritized, for long COVID.
In patients with postexertional malaise (PEM), one of the most common long COVID symptoms, exercise is actively harmful and causes dysfunctional metabolic patterns, cardiac preload failure, impaired systemic oxygen extraction, and more. GET and CBT have failed similar populations , and exercise is explicitly contraindicated by the World Health Organization, the British National Institute for Health and Care Excellence, the CDC, and other organizations.
Resources should instead be put toward the wide range of medications that have not yet adequately undergone clinical trials.
5: PCR and antibody tests should not be used as inclusion criteria for trial participants
Only an estimated 1%-3% of cases in the first wave of COVID were documented, and the CDC estimates that only 25% of cases through September 2021 were documented. Similarly, antibody tests are unreliable to determine past infection, as roughly a third of patients don’t seroconvert, and a similar proportion serorevert within a few months. Using polymerase chain reaction (PCR) and antibody testing to determine who should be included in clinical trials limits who is eligible to participate in research, particularly those who have been ill for longer. Additionally, the majority of those who serorevert are women, so using antibody tests for inclusion introduces a selection bias and may miss mechanisms of immune system functioning that are part of long COVID.
PCR tests also have high false-negative rates and requiring them in research excludes people with lower viral loads with long COVID, which would confound findings.
These issues with testing also lead to COVID-infected people accidentally being included in control groups, which ruins the credibility of the research findings completely.
6: Include comparator groups
There are several common diagnoses that occur in people with long COVID, including ME/CFS, postural orthostatic tachycardia syndrome, small-fiber neuropathy, mast cell activation syndrome, and Ehlers-Danlos syndrome.
Identifying people with these conditions within the trial cohort improves research across all fields, benefiting all groups, and helps clarify what types of patients benefit most from certain medications.
7: Identify the right endpoints; avoid the wrong ones
Even though our understanding of the pathophysiology of long COVID is still evolving, it’s still possible to do clinical trials by identifying strong endpoints and outcome measures.
Several tools have been designed for viral-onset conditions and should be used alongside other endpoints. Postexertional malaise and autonomic symptoms, which are some of the most common symptoms of long COVID, can be measured with the validated DSQ-PEM and COMPASS-31, respectively. Tools for cognitive dysfunction trials should capture specific and common types of impairment, like processing speed.
Endpoints should be high-impact and aim for large improvements that have clinical significance over small improvements that do not have clinical significance.
Objective tests should be incorporated where possible; some to consider include natural killer cell functioning, cerebral blood flow, T-cell functioning, levels of reactivated herpesviruses, blood lactate levels, and microclots, as testing becomes available.
Mental health outcomes shouldn’t be primary endpoints, except where a trial is targeting a specific mental health condition because of COVID (for example, premenstrual dysphoric disorder).
If mental health conditions are tracked secondarily, it’s vital not to use questionnaires that include physical symptoms like fatigue, difficulty concentrating, difficulty sleeping, or palpitations, as these artificially increase depression and anxiety scores in chronically ill respondents. Tools that include physical symptoms (Patient Health Questionnaire–9, Beck Anxiety Inventory, Beck Depression Inventory) can be replaced with scales like the PHQ-2, General Anxiety Disorder–7, Hospital Anxiety and Depression Scale, or PROMIS-29 subscales.
Because certain cytokines and other inflammatory markers may naturally decrease over time without corresponding improvement in the ME/CFS subtype, caution should be taken when using cytokines as endpoints.
8: Consider enrollment and objectives carefully
A proportion of people with long COVID will recover in the early months after infection. Ideally, clinical trials will primarily study treatments in patients who have been ill 6 months or longer, as some natural recovery will happen before that can bias studies.
But where resources are abundant, it is ideal for trials to additionally look at whether the treatments can help patients in the early months recover and prevent progression to the later stage.
9: Tracking illness duration is crucial
Research from ME/CFS shows that there may be an immune change in the first few years of the illness, where cytokines decrease without any corresponding change in symptom improvement.
Because of this and the possibility that other markers follow the same pattern, disease duration should be a core feature of all analyses and trial designs. Trial outcomes should be designed to answer the question of whether the medication helps patients at different durations of illness.
10: Prioritize patient populations less likely to recover without intervention
Some long COVID phenotypes seem less likely to recover without intervention. Trials should take care to focus on these patient populations, which include those with neurologic symptoms and those meeting ME/CFS criteria.
11: Account for the relapsing/remitting nature
Outcome measures need to be assessed in a way that can distinguish a temporary remission, which is part of the natural course of the disease, from a permanent cure.
Factors that can contribute to the relapsing/remitting nature include physical and cognitive postexertional malaise, menstrual cycle changes, and seasonal changes.
12: Trial participants should reflect the diversity of the long COVID population
Certain demographics are more likely to be affected by acute and long COVID and need to be appropriately recruited and reflected in research, including in patient engagement.
Trials must include high numbers of Hispanic/Latinx, Black, and indigenous communities, queer and transgender populations, and women. Trial materials and design need to incorporate linguistic diversity in addition to racial/ethnic diversity.
Upward of 75% of long COVID cases happen after mild acute cases; clinical researchers should ensure that nonhospitalized patients make up the bulk of trial participants.
13: Utilize meaningful engagement of patients, especially in treatment selection and study design
Meaningful patient engagement means engaging multiple patients at every step of the trial process, from treatment selection to study design to analysis to communication of the results.
Patient experiences are extremely valuable and contain information that researchers may not be familiar with, including the nature and patterns of the illness, insights into possible treatments, and barriers to documentation and care that may also impact research. Tapping into those patient experiences will make trials stronger.
Overall, the landscape of long COVID clinical trials is ripe for discovery, and researchers choosing to go down this path will be deeply appreciated by the patient community.
Hannah Davis is a long COVID patient-researcher and cofounder of the Patient-Led Research Collaborative, an organization studying the long-term effects of COVID.
A version of this article first appeared on Medscape.com.
The Centers for Disease Control and Prevention and the U.S. Census Bureau estimate that 6.1% of the U.S. adult population is living with long COVID, with millions more debilitated worldwide. The demand for substantial treatment is enormous, but the urgency to fund and begin the necessary range of clinical trials has not met the severity of the problem.
While trials are slowly beginning to happen, the treatment choices and trial design require crucial nuances and understanding of viral-onset illnesses, and few research groups are creating strong trials that fully reflect the complexities of this landscape.
These recommendations recognize that roughly half of long COVID patients have new-onset myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS) and dysautonomia from COVID, which must be at the forefront of how trials are designed and conducted, and are additionally based on the current hypotheses about long COVID’s pathophysiologies.
1: Drugs proposed by experts in postviral fields should be prioritized
Upward of 50 drugs for viral-onset conditions like ME/CFS, dysautonomia, AIDS, and others have been waiting for years to go to trial, but have not had the funding to do so.
Treatments proposed by experts in viral-onset illnesses (such as ME/CFS and dysautonomia) should be prioritized (PM R. 2022 Oct;14[10]:1270-91), as outside researchers are not familiar with these fields and their potential treatment options.
2: Drugs targeting a wide range of mechanisms should be trialed
Treatments that should be trialed include anticoagulants/antiplatelets for clotting and vascular functioning, immunomodulators including JAK-STAT inhibitors, COVID-specific antivirals and antivirals against reactivated herpesviruses (Valcyte, Valacyclovir, EBV vaccine).
Other options include prescription mast cell stabilizers (ketotifen, cromolyn sodium), drugs that regulate microglial activation (low-dose naltrexone, low-dose aripiprazole), anti-CGRP medications, beta-blockers, and intravenous immunoglobulin.
Others include medications that target mitochondrial dysfunction; ivabradine; pyridostigmine;, DRP1 inhibitors; supplements showing success in patient communities including lactoferrin, ubiquinone, and nattokinase; and therapies targeting glymphatic/lymphatic dysfunction, microbiome therapies, and therapeutic peptides.
3: Use appropriate long COVID subtypes
Long COVID is an umbrella term that encompasses multiple new-onset and worsened conditions and symptoms after COVID. Roughly half of long COVID patients likely meet the criteria for ME/CFS and/or dysautonomia. Others may have new-onset diabetes, major clotting events, lung damage, neurological disorders, loss of smell or taste, and other manifestations.
Patients in different categories likely have different responses to treatments. It’s critical to identify appropriate subtypes for each trial, ideally performing detailed analyses to identify the treatments that work best, and don’t, for each subtype.
4: Behavioral treatments, especially those that have harmed similar populations, should not be trialed
Behavioral treatments including exercise, graded exercise therapy (GET), and cognitive-behavioral therapy (CBT) should not be trialed, let alone prioritized, for long COVID.
In patients with postexertional malaise (PEM), one of the most common long COVID symptoms, exercise is actively harmful and causes dysfunctional metabolic patterns, cardiac preload failure, impaired systemic oxygen extraction, and more. GET and CBT have failed similar populations , and exercise is explicitly contraindicated by the World Health Organization, the British National Institute for Health and Care Excellence, the CDC, and other organizations.
Resources should instead be put toward the wide range of medications that have not yet adequately undergone clinical trials.
5: PCR and antibody tests should not be used as inclusion criteria for trial participants
Only an estimated 1%-3% of cases in the first wave of COVID were documented, and the CDC estimates that only 25% of cases through September 2021 were documented. Similarly, antibody tests are unreliable to determine past infection, as roughly a third of patients don’t seroconvert, and a similar proportion serorevert within a few months. Using polymerase chain reaction (PCR) and antibody testing to determine who should be included in clinical trials limits who is eligible to participate in research, particularly those who have been ill for longer. Additionally, the majority of those who serorevert are women, so using antibody tests for inclusion introduces a selection bias and may miss mechanisms of immune system functioning that are part of long COVID.
PCR tests also have high false-negative rates and requiring them in research excludes people with lower viral loads with long COVID, which would confound findings.
These issues with testing also lead to COVID-infected people accidentally being included in control groups, which ruins the credibility of the research findings completely.
6: Include comparator groups
There are several common diagnoses that occur in people with long COVID, including ME/CFS, postural orthostatic tachycardia syndrome, small-fiber neuropathy, mast cell activation syndrome, and Ehlers-Danlos syndrome.
Identifying people with these conditions within the trial cohort improves research across all fields, benefiting all groups, and helps clarify what types of patients benefit most from certain medications.
7: Identify the right endpoints; avoid the wrong ones
Even though our understanding of the pathophysiology of long COVID is still evolving, it’s still possible to do clinical trials by identifying strong endpoints and outcome measures.
Several tools have been designed for viral-onset conditions and should be used alongside other endpoints. Postexertional malaise and autonomic symptoms, which are some of the most common symptoms of long COVID, can be measured with the validated DSQ-PEM and COMPASS-31, respectively. Tools for cognitive dysfunction trials should capture specific and common types of impairment, like processing speed.
Endpoints should be high-impact and aim for large improvements that have clinical significance over small improvements that do not have clinical significance.
Objective tests should be incorporated where possible; some to consider include natural killer cell functioning, cerebral blood flow, T-cell functioning, levels of reactivated herpesviruses, blood lactate levels, and microclots, as testing becomes available.
Mental health outcomes shouldn’t be primary endpoints, except where a trial is targeting a specific mental health condition because of COVID (for example, premenstrual dysphoric disorder).
If mental health conditions are tracked secondarily, it’s vital not to use questionnaires that include physical symptoms like fatigue, difficulty concentrating, difficulty sleeping, or palpitations, as these artificially increase depression and anxiety scores in chronically ill respondents. Tools that include physical symptoms (Patient Health Questionnaire–9, Beck Anxiety Inventory, Beck Depression Inventory) can be replaced with scales like the PHQ-2, General Anxiety Disorder–7, Hospital Anxiety and Depression Scale, or PROMIS-29 subscales.
Because certain cytokines and other inflammatory markers may naturally decrease over time without corresponding improvement in the ME/CFS subtype, caution should be taken when using cytokines as endpoints.
8: Consider enrollment and objectives carefully
A proportion of people with long COVID will recover in the early months after infection. Ideally, clinical trials will primarily study treatments in patients who have been ill 6 months or longer, as some natural recovery will happen before that can bias studies.
But where resources are abundant, it is ideal for trials to additionally look at whether the treatments can help patients in the early months recover and prevent progression to the later stage.
9: Tracking illness duration is crucial
Research from ME/CFS shows that there may be an immune change in the first few years of the illness, where cytokines decrease without any corresponding change in symptom improvement.
Because of this and the possibility that other markers follow the same pattern, disease duration should be a core feature of all analyses and trial designs. Trial outcomes should be designed to answer the question of whether the medication helps patients at different durations of illness.
10: Prioritize patient populations less likely to recover without intervention
Some long COVID phenotypes seem less likely to recover without intervention. Trials should take care to focus on these patient populations, which include those with neurologic symptoms and those meeting ME/CFS criteria.
11: Account for the relapsing/remitting nature
Outcome measures need to be assessed in a way that can distinguish a temporary remission, which is part of the natural course of the disease, from a permanent cure.
Factors that can contribute to the relapsing/remitting nature include physical and cognitive postexertional malaise, menstrual cycle changes, and seasonal changes.
12: Trial participants should reflect the diversity of the long COVID population
Certain demographics are more likely to be affected by acute and long COVID and need to be appropriately recruited and reflected in research, including in patient engagement.
Trials must include high numbers of Hispanic/Latinx, Black, and indigenous communities, queer and transgender populations, and women. Trial materials and design need to incorporate linguistic diversity in addition to racial/ethnic diversity.
Upward of 75% of long COVID cases happen after mild acute cases; clinical researchers should ensure that nonhospitalized patients make up the bulk of trial participants.
13: Utilize meaningful engagement of patients, especially in treatment selection and study design
Meaningful patient engagement means engaging multiple patients at every step of the trial process, from treatment selection to study design to analysis to communication of the results.
Patient experiences are extremely valuable and contain information that researchers may not be familiar with, including the nature and patterns of the illness, insights into possible treatments, and barriers to documentation and care that may also impact research. Tapping into those patient experiences will make trials stronger.
Overall, the landscape of long COVID clinical trials is ripe for discovery, and researchers choosing to go down this path will be deeply appreciated by the patient community.
Hannah Davis is a long COVID patient-researcher and cofounder of the Patient-Led Research Collaborative, an organization studying the long-term effects of COVID.
A version of this article first appeared on Medscape.com.
Colchicine’s 2010 price spike had major impact on gout care
A large price increase for colchicine in 2010 led to a significant falloff in its use for gout that persisted for the next decade while emergency and rheumatology visits for gout rose, suggesting poorer disease control, a retrospective cohort study reported.
The price of colchicine, commonly prescribed for acute gout attacks, climbed from $11.25 per prescription in 2009 to $190.49 in 2011, with the average out-of-pocket cost more than quadrupling, from $7.37 to $29.42, the study noted. Colchicine prescriptions for gout declined 27% over the next decade, according to adjusted analyses that the study authors performed.
“A roughly 16-fold increase in colchicine prices appeared to have lowered colchicine use over the next decade,” senior author Zirui Song, MD, PhD, an associate professor of health care policy and medicine at Harvard Medical School and an internist at Massachusetts General Hospital in Boston, told this news organization in written comments. “Over the same period, patients with gout used more of other medications that could treat gout. They also had more emergency department visits for gout and rheumatologist visits for gout, which potentially signals poorer disease control.”
The study, published online in JAMA Internal Medicine, examined MarketScan data from a longitudinal cohort of patients who had employer-sponsored health insurance and a diagnosis of gout from 2007 to 2019. MarketScan is an IBM database of medical and drug data from employers and health plans. The study examined more than 2.7 million patient-year observations over the 13-year period.
How the price increase happened
After 2011, a large percentage of patients shifted to less effective but more affordable drugs to treat gout. Prescriptions for allopurinol increased 32% (P < .001) and oral corticosteroids 8.3% over the decade. “These are imperfect substitutes,” Dr. Song said. “Allopurinol is used to prevent gout, while oral corticosteroids can be used to treat a gout flare.”
At the same time, visits for gout-related complaints to emergency departments and rheumatology offices increased through the ensuing years: 39.8% and 10.5% on an adjusted analysis, respectively (P < .001 for both).
Colchicine is actually a drug that predates the creation of the U.S. Food and Drug Administration in 1938 and had been grandfathered under its Unapproved Drug Initiative. Then in 2009, the FDA determined that colchicine was effective for treating arthritis-related gout flares after the manufacturer, URL Pharma, presented results of a randomized, controlled trial of 185 patients with gout.
The next year, the FDA granted URL Pharma 3 years of market exclusivity for the drug under the brand name Colcrys, now trademarked by Takeda Pharmaceuticals.
The latest study noted that longer-term analysis of the impact of the FDA’s decision had been lacking. The goal, said Dr. Song, was “to better understand the long-run implications of large drug price increases in the U.S. by studying the case of colchicine.”
He added, “For drugs that lack competition, large price increases can have large economic and clinical consequences over many years.”
Absorbing the cost
Lead author Dan P. Ly, MD, PhD, MPP, assistant professor at the University of California, Los Angeles, added, “Our study has large implications [for] when generic medications or other medications experience large price increases. Use of the medication in question drops or patients have to pay more out of pocket, and patient health can suffer as a result.”
The dropoff in colchicine use in this patient population could have been worse, Dr. Song said. “Despite colchicine use decreasing by 27% over nearly a decade, the fact that it did not decline more suggests that for patients with gout, the large price increase was mostly absorbed by their insurers, employers, or themselves – e.g., passed through to higher premiums, lower wages, or higher cost-sharing.”
Aaron Kesselheim, MD, JD, MPH, a professor at Harvard Medical School, Boston, reported previously on the price consequences of colchicine early on after the FDA granted the manufacturer market exclusivity.
“In our past research, we looked at how the massive increase in the price of colchicine increased spending on the drug and reduced use in a relatively short time period after the price hike,” said Dr. Kesselheim, who was not involved in this current study by Dr. Ly, Dr. Song, and Mia Giuriato, BBA, MA, from Harvard Medical School. “This study evaluated the experiences of patients with gout over multiple years and showed that the reductions in use persisted and were associated with increases in ED and rheumatology visits, suggesting worsening control of gout due to the relative inaccessibility of the drug at the new high price.”
The latest findings have public policy implications, Dr. Kesselheim said. “In the case of colchicine, the FDA made a bad pitch, leading to a home run for the manufacturer and a shutout for patients.”
“The FDA needs to make sure to take into account the quite predictable patient effects that can result from disruptions to competition when it considers taking steps like it did in the colchicine case to disrupt the market and create an artificial monopoly, even if the FDA acted in the best of intentions in this case,” Dr. Kesselheim added.
Dr. Song received funding for the study from the National Institutes of Health and Arnold Ventures. He also disclosed receiving personal fees from the Research Triangle Institute, Google Ventures, VBID Health, and the International Foundation of Employee Benefit Plans. Dr. Ly, Ms. Giuriato, and Dr. Kesselheim report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A large price increase for colchicine in 2010 led to a significant falloff in its use for gout that persisted for the next decade while emergency and rheumatology visits for gout rose, suggesting poorer disease control, a retrospective cohort study reported.
The price of colchicine, commonly prescribed for acute gout attacks, climbed from $11.25 per prescription in 2009 to $190.49 in 2011, with the average out-of-pocket cost more than quadrupling, from $7.37 to $29.42, the study noted. Colchicine prescriptions for gout declined 27% over the next decade, according to adjusted analyses that the study authors performed.
“A roughly 16-fold increase in colchicine prices appeared to have lowered colchicine use over the next decade,” senior author Zirui Song, MD, PhD, an associate professor of health care policy and medicine at Harvard Medical School and an internist at Massachusetts General Hospital in Boston, told this news organization in written comments. “Over the same period, patients with gout used more of other medications that could treat gout. They also had more emergency department visits for gout and rheumatologist visits for gout, which potentially signals poorer disease control.”
The study, published online in JAMA Internal Medicine, examined MarketScan data from a longitudinal cohort of patients who had employer-sponsored health insurance and a diagnosis of gout from 2007 to 2019. MarketScan is an IBM database of medical and drug data from employers and health plans. The study examined more than 2.7 million patient-year observations over the 13-year period.
How the price increase happened
After 2011, a large percentage of patients shifted to less effective but more affordable drugs to treat gout. Prescriptions for allopurinol increased 32% (P < .001) and oral corticosteroids 8.3% over the decade. “These are imperfect substitutes,” Dr. Song said. “Allopurinol is used to prevent gout, while oral corticosteroids can be used to treat a gout flare.”
At the same time, visits for gout-related complaints to emergency departments and rheumatology offices increased through the ensuing years: 39.8% and 10.5% on an adjusted analysis, respectively (P < .001 for both).
Colchicine is actually a drug that predates the creation of the U.S. Food and Drug Administration in 1938 and had been grandfathered under its Unapproved Drug Initiative. Then in 2009, the FDA determined that colchicine was effective for treating arthritis-related gout flares after the manufacturer, URL Pharma, presented results of a randomized, controlled trial of 185 patients with gout.
The next year, the FDA granted URL Pharma 3 years of market exclusivity for the drug under the brand name Colcrys, now trademarked by Takeda Pharmaceuticals.
The latest study noted that longer-term analysis of the impact of the FDA’s decision had been lacking. The goal, said Dr. Song, was “to better understand the long-run implications of large drug price increases in the U.S. by studying the case of colchicine.”
He added, “For drugs that lack competition, large price increases can have large economic and clinical consequences over many years.”
Absorbing the cost
Lead author Dan P. Ly, MD, PhD, MPP, assistant professor at the University of California, Los Angeles, added, “Our study has large implications [for] when generic medications or other medications experience large price increases. Use of the medication in question drops or patients have to pay more out of pocket, and patient health can suffer as a result.”
The dropoff in colchicine use in this patient population could have been worse, Dr. Song said. “Despite colchicine use decreasing by 27% over nearly a decade, the fact that it did not decline more suggests that for patients with gout, the large price increase was mostly absorbed by their insurers, employers, or themselves – e.g., passed through to higher premiums, lower wages, or higher cost-sharing.”
Aaron Kesselheim, MD, JD, MPH, a professor at Harvard Medical School, Boston, reported previously on the price consequences of colchicine early on after the FDA granted the manufacturer market exclusivity.
“In our past research, we looked at how the massive increase in the price of colchicine increased spending on the drug and reduced use in a relatively short time period after the price hike,” said Dr. Kesselheim, who was not involved in this current study by Dr. Ly, Dr. Song, and Mia Giuriato, BBA, MA, from Harvard Medical School. “This study evaluated the experiences of patients with gout over multiple years and showed that the reductions in use persisted and were associated with increases in ED and rheumatology visits, suggesting worsening control of gout due to the relative inaccessibility of the drug at the new high price.”
The latest findings have public policy implications, Dr. Kesselheim said. “In the case of colchicine, the FDA made a bad pitch, leading to a home run for the manufacturer and a shutout for patients.”
“The FDA needs to make sure to take into account the quite predictable patient effects that can result from disruptions to competition when it considers taking steps like it did in the colchicine case to disrupt the market and create an artificial monopoly, even if the FDA acted in the best of intentions in this case,” Dr. Kesselheim added.
Dr. Song received funding for the study from the National Institutes of Health and Arnold Ventures. He also disclosed receiving personal fees from the Research Triangle Institute, Google Ventures, VBID Health, and the International Foundation of Employee Benefit Plans. Dr. Ly, Ms. Giuriato, and Dr. Kesselheim report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A large price increase for colchicine in 2010 led to a significant falloff in its use for gout that persisted for the next decade while emergency and rheumatology visits for gout rose, suggesting poorer disease control, a retrospective cohort study reported.
The price of colchicine, commonly prescribed for acute gout attacks, climbed from $11.25 per prescription in 2009 to $190.49 in 2011, with the average out-of-pocket cost more than quadrupling, from $7.37 to $29.42, the study noted. Colchicine prescriptions for gout declined 27% over the next decade, according to adjusted analyses that the study authors performed.
“A roughly 16-fold increase in colchicine prices appeared to have lowered colchicine use over the next decade,” senior author Zirui Song, MD, PhD, an associate professor of health care policy and medicine at Harvard Medical School and an internist at Massachusetts General Hospital in Boston, told this news organization in written comments. “Over the same period, patients with gout used more of other medications that could treat gout. They also had more emergency department visits for gout and rheumatologist visits for gout, which potentially signals poorer disease control.”
The study, published online in JAMA Internal Medicine, examined MarketScan data from a longitudinal cohort of patients who had employer-sponsored health insurance and a diagnosis of gout from 2007 to 2019. MarketScan is an IBM database of medical and drug data from employers and health plans. The study examined more than 2.7 million patient-year observations over the 13-year period.
How the price increase happened
After 2011, a large percentage of patients shifted to less effective but more affordable drugs to treat gout. Prescriptions for allopurinol increased 32% (P < .001) and oral corticosteroids 8.3% over the decade. “These are imperfect substitutes,” Dr. Song said. “Allopurinol is used to prevent gout, while oral corticosteroids can be used to treat a gout flare.”
At the same time, visits for gout-related complaints to emergency departments and rheumatology offices increased through the ensuing years: 39.8% and 10.5% on an adjusted analysis, respectively (P < .001 for both).
Colchicine is actually a drug that predates the creation of the U.S. Food and Drug Administration in 1938 and had been grandfathered under its Unapproved Drug Initiative. Then in 2009, the FDA determined that colchicine was effective for treating arthritis-related gout flares after the manufacturer, URL Pharma, presented results of a randomized, controlled trial of 185 patients with gout.
The next year, the FDA granted URL Pharma 3 years of market exclusivity for the drug under the brand name Colcrys, now trademarked by Takeda Pharmaceuticals.
The latest study noted that longer-term analysis of the impact of the FDA’s decision had been lacking. The goal, said Dr. Song, was “to better understand the long-run implications of large drug price increases in the U.S. by studying the case of colchicine.”
He added, “For drugs that lack competition, large price increases can have large economic and clinical consequences over many years.”
Absorbing the cost
Lead author Dan P. Ly, MD, PhD, MPP, assistant professor at the University of California, Los Angeles, added, “Our study has large implications [for] when generic medications or other medications experience large price increases. Use of the medication in question drops or patients have to pay more out of pocket, and patient health can suffer as a result.”
The dropoff in colchicine use in this patient population could have been worse, Dr. Song said. “Despite colchicine use decreasing by 27% over nearly a decade, the fact that it did not decline more suggests that for patients with gout, the large price increase was mostly absorbed by their insurers, employers, or themselves – e.g., passed through to higher premiums, lower wages, or higher cost-sharing.”
Aaron Kesselheim, MD, JD, MPH, a professor at Harvard Medical School, Boston, reported previously on the price consequences of colchicine early on after the FDA granted the manufacturer market exclusivity.
“In our past research, we looked at how the massive increase in the price of colchicine increased spending on the drug and reduced use in a relatively short time period after the price hike,” said Dr. Kesselheim, who was not involved in this current study by Dr. Ly, Dr. Song, and Mia Giuriato, BBA, MA, from Harvard Medical School. “This study evaluated the experiences of patients with gout over multiple years and showed that the reductions in use persisted and were associated with increases in ED and rheumatology visits, suggesting worsening control of gout due to the relative inaccessibility of the drug at the new high price.”
The latest findings have public policy implications, Dr. Kesselheim said. “In the case of colchicine, the FDA made a bad pitch, leading to a home run for the manufacturer and a shutout for patients.”
“The FDA needs to make sure to take into account the quite predictable patient effects that can result from disruptions to competition when it considers taking steps like it did in the colchicine case to disrupt the market and create an artificial monopoly, even if the FDA acted in the best of intentions in this case,” Dr. Kesselheim added.
Dr. Song received funding for the study from the National Institutes of Health and Arnold Ventures. He also disclosed receiving personal fees from the Research Triangle Institute, Google Ventures, VBID Health, and the International Foundation of Employee Benefit Plans. Dr. Ly, Ms. Giuriato, and Dr. Kesselheim report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Study shifts burden of IgG4-related disease to women
The incidence and prevalence of IgG4-related disease each rose considerably from 2015 to 2019 in the United States, and the risk of death in those with the immune-mediated condition is about 2.5 times higher than those who are not affected, based on an analysis of claims data from commercially insured adults.
The first population-based study of IgG4-RD incidence, prevalence, and mortality establishes “key benchmarks for informing the diagnosis and management of patients” with a condition “that causes fibrosing inflammatory lesions at nearly any anatomic site,” and wasn’t initially described until 2001, Zachary S. Wallace, MD, and associates said in Annals of the Rheumatic Diseases.
The increases in incidence and prevalence likely reflected increased disease awareness, they suggested. Overall U.S. incidence was 1.2 per 100,000 person-years for the 5-year period of 2015-2019, rising 86% from 0.78 per 100,000 person-years to 1.45 in 2018 before dropping to 1.39 in 2019. The change in prevalence was even greater, increasing 122% from 2.41 per 100,000 persons in 2015 to 5.34 per 100,000 in 2019, the investigators said.
Previous studies had indicated that the majority of patients with IgG4-RD were male, but the current study, using Optum’s Clinformatics Data Mart, which includes commercial health plan and Medicare Advantage members in all 50 states, showed that both incidence and prevalence (see graph) were higher among women, noted Dr. Wallace of Massachusetts General Hospital, Boston, and associates. They identified 524 patients (57.6% female) in the database who met the criteria for IgG4-RD from Jan. 1, 2010, to Dec. 31, 2019.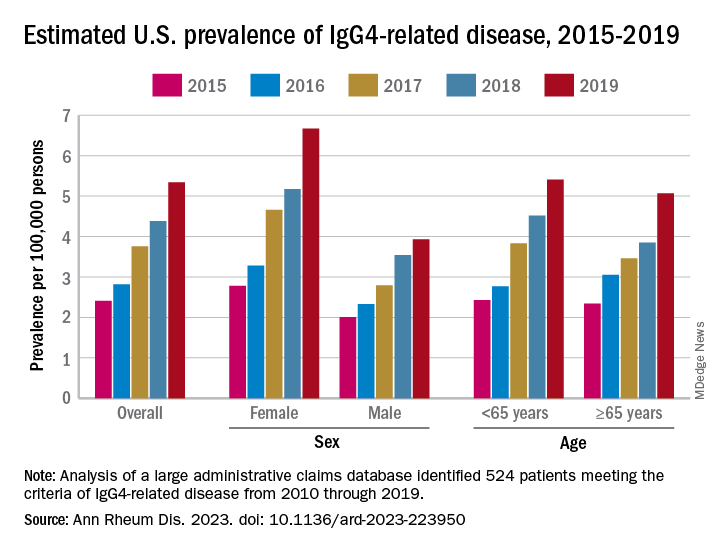
Incidence over the course of the study “was similar in patients identified as Asian or White but lower in those identified as Black or Hispanic,” they noted, adding that “the prevalence of IgG4-RD during this period reflected similar trends.” A jump in prevalence from 2018 to 2019, however, left White patients with a much higher rate (6.13 per 100,000 persons) than Asian patients (4.54 per 100,000), Black patients (3.42), and Hispanic patients (3.02).
For the mortality analysis, 516 patients with IgG4-RD were age-, sex-, and race-matched with 5,160 patients without IgG4-RD. Mortality was 3.42 and 1.46 per 100 person-years, respectively, over the 5.5 years of follow-up, so IgG4-RD was associated with a 2.5-fold higher risk of death. “The association of IgG4-RD with a higher risk of death was observed across the age spectrum and among both male and female patients,” the researchers said.
“Clinicians across specialties should be aware of IgG4-RD given the incidence, prevalence, and excess risk of death associated with this condition. ... Additional studies are urgently needed to define optimal management strategies to improve survival,” they wrote.
The study was supported by a grant to Massachusetts General Hospital from Sanofi, and Dr. Wallace received funding from the National Institutes of Health/National Institute of Arthritis and Musculoskeletal and Skin Diseases, and the Rheumatology Research Foundation. He has received research support and consulting fees from several companies, and four coinvestigators are employees of Sanofi.
The incidence and prevalence of IgG4-related disease each rose considerably from 2015 to 2019 in the United States, and the risk of death in those with the immune-mediated condition is about 2.5 times higher than those who are not affected, based on an analysis of claims data from commercially insured adults.
The first population-based study of IgG4-RD incidence, prevalence, and mortality establishes “key benchmarks for informing the diagnosis and management of patients” with a condition “that causes fibrosing inflammatory lesions at nearly any anatomic site,” and wasn’t initially described until 2001, Zachary S. Wallace, MD, and associates said in Annals of the Rheumatic Diseases.
The increases in incidence and prevalence likely reflected increased disease awareness, they suggested. Overall U.S. incidence was 1.2 per 100,000 person-years for the 5-year period of 2015-2019, rising 86% from 0.78 per 100,000 person-years to 1.45 in 2018 before dropping to 1.39 in 2019. The change in prevalence was even greater, increasing 122% from 2.41 per 100,000 persons in 2015 to 5.34 per 100,000 in 2019, the investigators said.
Previous studies had indicated that the majority of patients with IgG4-RD were male, but the current study, using Optum’s Clinformatics Data Mart, which includes commercial health plan and Medicare Advantage members in all 50 states, showed that both incidence and prevalence (see graph) were higher among women, noted Dr. Wallace of Massachusetts General Hospital, Boston, and associates. They identified 524 patients (57.6% female) in the database who met the criteria for IgG4-RD from Jan. 1, 2010, to Dec. 31, 2019.
Incidence over the course of the study “was similar in patients identified as Asian or White but lower in those identified as Black or Hispanic,” they noted, adding that “the prevalence of IgG4-RD during this period reflected similar trends.” A jump in prevalence from 2018 to 2019, however, left White patients with a much higher rate (6.13 per 100,000 persons) than Asian patients (4.54 per 100,000), Black patients (3.42), and Hispanic patients (3.02).
For the mortality analysis, 516 patients with IgG4-RD were age-, sex-, and race-matched with 5,160 patients without IgG4-RD. Mortality was 3.42 and 1.46 per 100 person-years, respectively, over the 5.5 years of follow-up, so IgG4-RD was associated with a 2.5-fold higher risk of death. “The association of IgG4-RD with a higher risk of death was observed across the age spectrum and among both male and female patients,” the researchers said.
“Clinicians across specialties should be aware of IgG4-RD given the incidence, prevalence, and excess risk of death associated with this condition. ... Additional studies are urgently needed to define optimal management strategies to improve survival,” they wrote.
The study was supported by a grant to Massachusetts General Hospital from Sanofi, and Dr. Wallace received funding from the National Institutes of Health/National Institute of Arthritis and Musculoskeletal and Skin Diseases, and the Rheumatology Research Foundation. He has received research support and consulting fees from several companies, and four coinvestigators are employees of Sanofi.
The incidence and prevalence of IgG4-related disease each rose considerably from 2015 to 2019 in the United States, and the risk of death in those with the immune-mediated condition is about 2.5 times higher than those who are not affected, based on an analysis of claims data from commercially insured adults.
The first population-based study of IgG4-RD incidence, prevalence, and mortality establishes “key benchmarks for informing the diagnosis and management of patients” with a condition “that causes fibrosing inflammatory lesions at nearly any anatomic site,” and wasn’t initially described until 2001, Zachary S. Wallace, MD, and associates said in Annals of the Rheumatic Diseases.
The increases in incidence and prevalence likely reflected increased disease awareness, they suggested. Overall U.S. incidence was 1.2 per 100,000 person-years for the 5-year period of 2015-2019, rising 86% from 0.78 per 100,000 person-years to 1.45 in 2018 before dropping to 1.39 in 2019. The change in prevalence was even greater, increasing 122% from 2.41 per 100,000 persons in 2015 to 5.34 per 100,000 in 2019, the investigators said.
Previous studies had indicated that the majority of patients with IgG4-RD were male, but the current study, using Optum’s Clinformatics Data Mart, which includes commercial health plan and Medicare Advantage members in all 50 states, showed that both incidence and prevalence (see graph) were higher among women, noted Dr. Wallace of Massachusetts General Hospital, Boston, and associates. They identified 524 patients (57.6% female) in the database who met the criteria for IgG4-RD from Jan. 1, 2010, to Dec. 31, 2019.
Incidence over the course of the study “was similar in patients identified as Asian or White but lower in those identified as Black or Hispanic,” they noted, adding that “the prevalence of IgG4-RD during this period reflected similar trends.” A jump in prevalence from 2018 to 2019, however, left White patients with a much higher rate (6.13 per 100,000 persons) than Asian patients (4.54 per 100,000), Black patients (3.42), and Hispanic patients (3.02).
For the mortality analysis, 516 patients with IgG4-RD were age-, sex-, and race-matched with 5,160 patients without IgG4-RD. Mortality was 3.42 and 1.46 per 100 person-years, respectively, over the 5.5 years of follow-up, so IgG4-RD was associated with a 2.5-fold higher risk of death. “The association of IgG4-RD with a higher risk of death was observed across the age spectrum and among both male and female patients,” the researchers said.
“Clinicians across specialties should be aware of IgG4-RD given the incidence, prevalence, and excess risk of death associated with this condition. ... Additional studies are urgently needed to define optimal management strategies to improve survival,” they wrote.
The study was supported by a grant to Massachusetts General Hospital from Sanofi, and Dr. Wallace received funding from the National Institutes of Health/National Institute of Arthritis and Musculoskeletal and Skin Diseases, and the Rheumatology Research Foundation. He has received research support and consulting fees from several companies, and four coinvestigators are employees of Sanofi.
FROM ANNALS OF THE RHEUMATIC DISEASES
Axial spondyloarthritis versus axial psoriatic arthritis: Different entities?
Are there clinically significant differences between axial spondyloarthritis with psoriasis and psoriatic arthritis with axial symptoms? Does it matter?
It all depends on whom you ask, but right now the evidence seems to be tipping in favor of the “splitters” who cite evidence supporting their contention that axial spondyloarthritis (axSpA)/ankylosing spondylitis (AS) with psoriasis and psoriatic arthritis (PsA) with axial symptoms are distinct clinical entities that require more precise diagnosis and treatment.
“Lumpers,” in contrast, argue that they are different points on the same clinical spectrum.
The debate is not just of academic interest, but has real consequences for patients, say specialists on both sides of the aisle.
Overlapping features, different presentations
“Axial SpA and axPsA have overlapping features but also meaningful differences in genetics, clinical presentation, imaging, and immunophenotype. Efforts are underway to develop classification criteria for axPsA to aid research efforts as well as clinical diagnosis and management,” Philip J. Mease, MD, director of rheumatology research at Swedish Medical Center/Providence–St. Joseph Health in Seattle, and colleagues contend.
In an editorial published in the International Journal of Rheumatic Diseases, Dr. Mease and colleagues noted that, although HLA-B*27 is a genetic risk factor for both axPsA and axSpA, some HLA-B alleles are significantly associated with axPsA, whereas other alleles are associated with axSpA.
In addition, while genes in the interleukin-23 and IL-17 pathway are associated with increased risk for axSpA, genes in the IL-13 pathway have been identified as risk markers for axPsA, they noted.
Two cohorts better than one?
Dafna Gladman, MD, professor of medicine at the University of Toronto and senior scientist at the Schroeder Arthritis Institute at Toronto Western Hospital, and colleagues have a unique perspective on the similarities and differences between the disease entities.
Her group’s research uses data on cohorts of patients treated in two separate clinics at Toronto Western Hospital: one for patients with PsA, and one for patients with axial spondyloarthritis, including those with ankylosing spondylitis, nonradiographic axSpA, and spondylitis associated with inflammatory bowel disease.
“Our work has shown that there are differences, and one of the reasons that it’s now important is that the anti–IL-23 medications, both the IL-12/23 inhibitor ustekinumab [Stelara] and the IL-23 inhibitor guselkumab [Tremfya] work for psoriatic arthritis, whereas IL-23 did not work in ankylosing spondylitis, so that provided further impetus to look into the distinction between the two groups,” Dr. Gladman said in an interview.
Dr. Gladman and colleagues published a study in Rheumatology in which they compared clinical presentations and features of patients with AS with or without psoriasis with patients with axPsA.
They found that patients with AS with or without psoriasis tended to be younger, had a higher proportion of males to females, and were more likely to be positive for HLA-B*27. Patients with AS also had more back pain at presentation, worse axial disease activity scores, worse global assessments by physicians, and higher grades of sacroiliitis, and they were more likely to be taking biologic agents.
“What that showed, right off the top, that whether we’re looking at the total group or we’re looking specifically at those patients who have psoriasis or don’t have psoriasis, they are different from those with psoriatic arthritis with axial disease,” she said.
They concluded that “axPsA seems to be a distinct entity.”
Two clinics, same presentation
Because the aforementioned study included all patients with PsA with or without peripheral disease, the investigators decided to filter out some of the background noise and conduct a second study in which they compared patients who presented to the two clinics with the same presentation, either with spinal disease and psoriasis to the spondylitis clinic, or with psoriasis and isolated axial disease to the PsA clinic.
The results, published in Annals of the Rheumatic Diseases, showed that just 2.03% of patients with PsA had isolated axial disease, and an additional 29.38% had axial and peripheral disease.
In this study, “you can see that even in that group there are distinct differences. The patients that are labeled psoriatic spondylitis are different from those that are labeled ankylosing spondylitis with psoriasis,” Dr. Gladman said.
Isolated axial disease in patients with PsA was associated with HLA-B*27 positivity and lower Health Assessment Questionnaire scores. In addition, patients who were HLA-B*27 positive also had a nearly eightfold higher risk for developing peripheral disease over time.
Patients with isolated axial PsA were significantly more likely to be diagnosed at an older age (mean, 37.44 vs. 29.65 years), had higher Psoriasis Area Severity Index scores and a higher likelihood of having psoriatic nail lesions than patients with AS with isolated axial disease and psoriasis.
In contrast, patients with isolated axSpA with psoriasis were more likely to have inflammatory back pain, spinal pain, joint pain/swelling, and areas of localized tenderness, and they had greater severity of morning stiffness.
Dr. Gladman noted that, although AS and PsA are associated with the same gene that encodes for the IL-23 receptor, each condition is associated with a different single-nucleotide polymorphism.
Same disease, different flavors?
But as Mark Twain said, it is difference of opinion that makes horse races, and some specialists in rheumatology say that axSpA amd axPsA are just two sides of the same coin.
“There are always different schools of thought. I believe that they are not different diseases, but a spectrum of diseases,” said Shailendra Singh, MD, a rheumatologist at Unity Health Medical Center in Searcy, Ark., and past president of the Arkansas Rheumatology Association.
In an interview, Dr. Singh said that the spectrum ranges from diseases with primarily axial involvement, such as AS, to those with primarily peripheral involvement, such as reactive arthritis.
He pointed out that these conditions have overlapping symptoms, including enthesitis, dactylitis, and uveitis, and inflammatory arthritis.
Daniel Wendling, MD, PhD, from the Centre Hospitalier Régional Universitaire de Besançon (France), Université de Franche-Comté, and colleagues agreed.
“The criteria currently available for both SpA [ASAS (Assessment of Spondyloarthritis International Society) criteria] and PsA [CASPAR (Classification for Psoriatic Arthritis) criteria] are classification criteria, not diagnostic criteria. They are not very stringent and are not exclusive. Thus, the same patient can easily be classified simultaneously in both entities, making the distinction between axSpA with psoriasis and axPsA theoretical,” they wrote in an editorial published in Joint Bone Spine.
They cited as an example of the allegedly fuzzy criteria a prospective study conducted by the investigators in Bath, England, in which modified New York criteria for AS were met by 24% of patients with AS, and CASPAR criteria for PsA were met by an equal number of patients with AS.
Therapeutic implications
Dr. Wendling and colleagues acknowledge the differences cited in studies by Dr. Gladman, Dr. Mease, and others between patients with axPsA and those with axSpA, but argue that the differences are not that great and not so clear.
“It should also be emphasized that, although some differences between axPsA and axSpA reach statistical significance, they are mostly at the margin, with low odd ratios,” they wrote.
“It is also important to consider the variability in the definition of axPsA, sometimes simply ‘physician reported’ and elsewhere based on the modified New York radiographic criteria; the latter are only present late in the course of the disease, and this may induce bias,” they continued.
Dr. Singh agreed that, as noted by Dr. Gladman, some patients will respond to anti–IL-17, anti–IL-23, and anti–IL-12/23 agents, whereas others will have better responses with tumor necrosis factor (TNF) inhibitors, and still others, such as those with peripheral involvement in the hands and feet may fare better with nonbiologic disease-modifying antirheumatic drugs such as methotrexate.
Answers to come?
Dr. Gladman noted that the information available to date about the efficacy of IL-23 inhibition in axPsA is based on a post hoc analysis of the PSUMMIT 1 and 2 controlled trials in PsA, and is not definitive.
The randomized, controlled STAR trial, currently recruiting patients, is designed to see whether guselkumab can reduce axial symptoms and inflammation in patients with active axPsA.
“What I say is, there is a rationale for [anti–IL-23] to work in psoriatic arthritis, and not work in ankylosing spondylitis,” she said.
In contrast, IL-17 inhibitors, anti-TNF agents, and Janus kinase inhibitors show efficacy against both axPsA and AS. Rituximab is ineffective against PsA, but has shown efficacy against AS, especially in patients with neurologic complications from anti-TNF agents.
“There may be other medications that would work more specifically in axial psoriatic arthritis that don’t work in ankylosing spondylitis, but at least recognizing that there may be some differences, and that therefore a correct diagnosis should be obtained, might be important,” she said.
Ideally, the picture will become clearer with results from the ongoing Axial Involvement in Psoriatic Arthritis cohort, a joint project of ASAS and the Group for Research and Assessment of Psoriasis and Psoriatic Arthritis. The multinational, cross-sectional study is designed “to systematically evaluate clinical and imaging manifestations indicative of axial involvement in patients with PsA and to develop classification criteria and a unified nomenclature for axial involvement in PsA that would allow defining a homogeneous subgroup of patients for research.”
Stay tuned.
Dr. Gladman’s research is supported by a grant from the Krembil Foundation. Dr. Singh disclosed research support from various companies. Funding sources and conflict of interest disclosures from other works cited are contained in their respective references.
Are there clinically significant differences between axial spondyloarthritis with psoriasis and psoriatic arthritis with axial symptoms? Does it matter?
It all depends on whom you ask, but right now the evidence seems to be tipping in favor of the “splitters” who cite evidence supporting their contention that axial spondyloarthritis (axSpA)/ankylosing spondylitis (AS) with psoriasis and psoriatic arthritis (PsA) with axial symptoms are distinct clinical entities that require more precise diagnosis and treatment.
“Lumpers,” in contrast, argue that they are different points on the same clinical spectrum.
The debate is not just of academic interest, but has real consequences for patients, say specialists on both sides of the aisle.
Overlapping features, different presentations
“Axial SpA and axPsA have overlapping features but also meaningful differences in genetics, clinical presentation, imaging, and immunophenotype. Efforts are underway to develop classification criteria for axPsA to aid research efforts as well as clinical diagnosis and management,” Philip J. Mease, MD, director of rheumatology research at Swedish Medical Center/Providence–St. Joseph Health in Seattle, and colleagues contend.
In an editorial published in the International Journal of Rheumatic Diseases, Dr. Mease and colleagues noted that, although HLA-B*27 is a genetic risk factor for both axPsA and axSpA, some HLA-B alleles are significantly associated with axPsA, whereas other alleles are associated with axSpA.
In addition, while genes in the interleukin-23 and IL-17 pathway are associated with increased risk for axSpA, genes in the IL-13 pathway have been identified as risk markers for axPsA, they noted.
Two cohorts better than one?
Dafna Gladman, MD, professor of medicine at the University of Toronto and senior scientist at the Schroeder Arthritis Institute at Toronto Western Hospital, and colleagues have a unique perspective on the similarities and differences between the disease entities.
Her group’s research uses data on cohorts of patients treated in two separate clinics at Toronto Western Hospital: one for patients with PsA, and one for patients with axial spondyloarthritis, including those with ankylosing spondylitis, nonradiographic axSpA, and spondylitis associated with inflammatory bowel disease.
“Our work has shown that there are differences, and one of the reasons that it’s now important is that the anti–IL-23 medications, both the IL-12/23 inhibitor ustekinumab [Stelara] and the IL-23 inhibitor guselkumab [Tremfya] work for psoriatic arthritis, whereas IL-23 did not work in ankylosing spondylitis, so that provided further impetus to look into the distinction between the two groups,” Dr. Gladman said in an interview.
Dr. Gladman and colleagues published a study in Rheumatology in which they compared clinical presentations and features of patients with AS with or without psoriasis with patients with axPsA.
They found that patients with AS with or without psoriasis tended to be younger, had a higher proportion of males to females, and were more likely to be positive for HLA-B*27. Patients with AS also had more back pain at presentation, worse axial disease activity scores, worse global assessments by physicians, and higher grades of sacroiliitis, and they were more likely to be taking biologic agents.
“What that showed, right off the top, that whether we’re looking at the total group or we’re looking specifically at those patients who have psoriasis or don’t have psoriasis, they are different from those with psoriatic arthritis with axial disease,” she said.
They concluded that “axPsA seems to be a distinct entity.”
Two clinics, same presentation
Because the aforementioned study included all patients with PsA with or without peripheral disease, the investigators decided to filter out some of the background noise and conduct a second study in which they compared patients who presented to the two clinics with the same presentation, either with spinal disease and psoriasis to the spondylitis clinic, or with psoriasis and isolated axial disease to the PsA clinic.
The results, published in Annals of the Rheumatic Diseases, showed that just 2.03% of patients with PsA had isolated axial disease, and an additional 29.38% had axial and peripheral disease.
In this study, “you can see that even in that group there are distinct differences. The patients that are labeled psoriatic spondylitis are different from those that are labeled ankylosing spondylitis with psoriasis,” Dr. Gladman said.
Isolated axial disease in patients with PsA was associated with HLA-B*27 positivity and lower Health Assessment Questionnaire scores. In addition, patients who were HLA-B*27 positive also had a nearly eightfold higher risk for developing peripheral disease over time.
Patients with isolated axial PsA were significantly more likely to be diagnosed at an older age (mean, 37.44 vs. 29.65 years), had higher Psoriasis Area Severity Index scores and a higher likelihood of having psoriatic nail lesions than patients with AS with isolated axial disease and psoriasis.
In contrast, patients with isolated axSpA with psoriasis were more likely to have inflammatory back pain, spinal pain, joint pain/swelling, and areas of localized tenderness, and they had greater severity of morning stiffness.
Dr. Gladman noted that, although AS and PsA are associated with the same gene that encodes for the IL-23 receptor, each condition is associated with a different single-nucleotide polymorphism.
Same disease, different flavors?
But as Mark Twain said, it is difference of opinion that makes horse races, and some specialists in rheumatology say that axSpA amd axPsA are just two sides of the same coin.
“There are always different schools of thought. I believe that they are not different diseases, but a spectrum of diseases,” said Shailendra Singh, MD, a rheumatologist at Unity Health Medical Center in Searcy, Ark., and past president of the Arkansas Rheumatology Association.
In an interview, Dr. Singh said that the spectrum ranges from diseases with primarily axial involvement, such as AS, to those with primarily peripheral involvement, such as reactive arthritis.
He pointed out that these conditions have overlapping symptoms, including enthesitis, dactylitis, and uveitis, and inflammatory arthritis.
Daniel Wendling, MD, PhD, from the Centre Hospitalier Régional Universitaire de Besançon (France), Université de Franche-Comté, and colleagues agreed.
“The criteria currently available for both SpA [ASAS (Assessment of Spondyloarthritis International Society) criteria] and PsA [CASPAR (Classification for Psoriatic Arthritis) criteria] are classification criteria, not diagnostic criteria. They are not very stringent and are not exclusive. Thus, the same patient can easily be classified simultaneously in both entities, making the distinction between axSpA with psoriasis and axPsA theoretical,” they wrote in an editorial published in Joint Bone Spine.
They cited as an example of the allegedly fuzzy criteria a prospective study conducted by the investigators in Bath, England, in which modified New York criteria for AS were met by 24% of patients with AS, and CASPAR criteria for PsA were met by an equal number of patients with AS.
Therapeutic implications
Dr. Wendling and colleagues acknowledge the differences cited in studies by Dr. Gladman, Dr. Mease, and others between patients with axPsA and those with axSpA, but argue that the differences are not that great and not so clear.
“It should also be emphasized that, although some differences between axPsA and axSpA reach statistical significance, they are mostly at the margin, with low odd ratios,” they wrote.
“It is also important to consider the variability in the definition of axPsA, sometimes simply ‘physician reported’ and elsewhere based on the modified New York radiographic criteria; the latter are only present late in the course of the disease, and this may induce bias,” they continued.
Dr. Singh agreed that, as noted by Dr. Gladman, some patients will respond to anti–IL-17, anti–IL-23, and anti–IL-12/23 agents, whereas others will have better responses with tumor necrosis factor (TNF) inhibitors, and still others, such as those with peripheral involvement in the hands and feet may fare better with nonbiologic disease-modifying antirheumatic drugs such as methotrexate.
Answers to come?
Dr. Gladman noted that the information available to date about the efficacy of IL-23 inhibition in axPsA is based on a post hoc analysis of the PSUMMIT 1 and 2 controlled trials in PsA, and is not definitive.
The randomized, controlled STAR trial, currently recruiting patients, is designed to see whether guselkumab can reduce axial symptoms and inflammation in patients with active axPsA.
“What I say is, there is a rationale for [anti–IL-23] to work in psoriatic arthritis, and not work in ankylosing spondylitis,” she said.
In contrast, IL-17 inhibitors, anti-TNF agents, and Janus kinase inhibitors show efficacy against both axPsA and AS. Rituximab is ineffective against PsA, but has shown efficacy against AS, especially in patients with neurologic complications from anti-TNF agents.
“There may be other medications that would work more specifically in axial psoriatic arthritis that don’t work in ankylosing spondylitis, but at least recognizing that there may be some differences, and that therefore a correct diagnosis should be obtained, might be important,” she said.
Ideally, the picture will become clearer with results from the ongoing Axial Involvement in Psoriatic Arthritis cohort, a joint project of ASAS and the Group for Research and Assessment of Psoriasis and Psoriatic Arthritis. The multinational, cross-sectional study is designed “to systematically evaluate clinical and imaging manifestations indicative of axial involvement in patients with PsA and to develop classification criteria and a unified nomenclature for axial involvement in PsA that would allow defining a homogeneous subgroup of patients for research.”
Stay tuned.
Dr. Gladman’s research is supported by a grant from the Krembil Foundation. Dr. Singh disclosed research support from various companies. Funding sources and conflict of interest disclosures from other works cited are contained in their respective references.
Are there clinically significant differences between axial spondyloarthritis with psoriasis and psoriatic arthritis with axial symptoms? Does it matter?
It all depends on whom you ask, but right now the evidence seems to be tipping in favor of the “splitters” who cite evidence supporting their contention that axial spondyloarthritis (axSpA)/ankylosing spondylitis (AS) with psoriasis and psoriatic arthritis (PsA) with axial symptoms are distinct clinical entities that require more precise diagnosis and treatment.
“Lumpers,” in contrast, argue that they are different points on the same clinical spectrum.
The debate is not just of academic interest, but has real consequences for patients, say specialists on both sides of the aisle.
Overlapping features, different presentations
“Axial SpA and axPsA have overlapping features but also meaningful differences in genetics, clinical presentation, imaging, and immunophenotype. Efforts are underway to develop classification criteria for axPsA to aid research efforts as well as clinical diagnosis and management,” Philip J. Mease, MD, director of rheumatology research at Swedish Medical Center/Providence–St. Joseph Health in Seattle, and colleagues contend.
In an editorial published in the International Journal of Rheumatic Diseases, Dr. Mease and colleagues noted that, although HLA-B*27 is a genetic risk factor for both axPsA and axSpA, some HLA-B alleles are significantly associated with axPsA, whereas other alleles are associated with axSpA.
In addition, while genes in the interleukin-23 and IL-17 pathway are associated with increased risk for axSpA, genes in the IL-13 pathway have been identified as risk markers for axPsA, they noted.
Two cohorts better than one?
Dafna Gladman, MD, professor of medicine at the University of Toronto and senior scientist at the Schroeder Arthritis Institute at Toronto Western Hospital, and colleagues have a unique perspective on the similarities and differences between the disease entities.
Her group’s research uses data on cohorts of patients treated in two separate clinics at Toronto Western Hospital: one for patients with PsA, and one for patients with axial spondyloarthritis, including those with ankylosing spondylitis, nonradiographic axSpA, and spondylitis associated with inflammatory bowel disease.
“Our work has shown that there are differences, and one of the reasons that it’s now important is that the anti–IL-23 medications, both the IL-12/23 inhibitor ustekinumab [Stelara] and the IL-23 inhibitor guselkumab [Tremfya] work for psoriatic arthritis, whereas IL-23 did not work in ankylosing spondylitis, so that provided further impetus to look into the distinction between the two groups,” Dr. Gladman said in an interview.
Dr. Gladman and colleagues published a study in Rheumatology in which they compared clinical presentations and features of patients with AS with or without psoriasis with patients with axPsA.
They found that patients with AS with or without psoriasis tended to be younger, had a higher proportion of males to females, and were more likely to be positive for HLA-B*27. Patients with AS also had more back pain at presentation, worse axial disease activity scores, worse global assessments by physicians, and higher grades of sacroiliitis, and they were more likely to be taking biologic agents.
“What that showed, right off the top, that whether we’re looking at the total group or we’re looking specifically at those patients who have psoriasis or don’t have psoriasis, they are different from those with psoriatic arthritis with axial disease,” she said.
They concluded that “axPsA seems to be a distinct entity.”
Two clinics, same presentation
Because the aforementioned study included all patients with PsA with or without peripheral disease, the investigators decided to filter out some of the background noise and conduct a second study in which they compared patients who presented to the two clinics with the same presentation, either with spinal disease and psoriasis to the spondylitis clinic, or with psoriasis and isolated axial disease to the PsA clinic.
The results, published in Annals of the Rheumatic Diseases, showed that just 2.03% of patients with PsA had isolated axial disease, and an additional 29.38% had axial and peripheral disease.
In this study, “you can see that even in that group there are distinct differences. The patients that are labeled psoriatic spondylitis are different from those that are labeled ankylosing spondylitis with psoriasis,” Dr. Gladman said.
Isolated axial disease in patients with PsA was associated with HLA-B*27 positivity and lower Health Assessment Questionnaire scores. In addition, patients who were HLA-B*27 positive also had a nearly eightfold higher risk for developing peripheral disease over time.
Patients with isolated axial PsA were significantly more likely to be diagnosed at an older age (mean, 37.44 vs. 29.65 years), had higher Psoriasis Area Severity Index scores and a higher likelihood of having psoriatic nail lesions than patients with AS with isolated axial disease and psoriasis.
In contrast, patients with isolated axSpA with psoriasis were more likely to have inflammatory back pain, spinal pain, joint pain/swelling, and areas of localized tenderness, and they had greater severity of morning stiffness.
Dr. Gladman noted that, although AS and PsA are associated with the same gene that encodes for the IL-23 receptor, each condition is associated with a different single-nucleotide polymorphism.
Same disease, different flavors?
But as Mark Twain said, it is difference of opinion that makes horse races, and some specialists in rheumatology say that axSpA amd axPsA are just two sides of the same coin.
“There are always different schools of thought. I believe that they are not different diseases, but a spectrum of diseases,” said Shailendra Singh, MD, a rheumatologist at Unity Health Medical Center in Searcy, Ark., and past president of the Arkansas Rheumatology Association.
In an interview, Dr. Singh said that the spectrum ranges from diseases with primarily axial involvement, such as AS, to those with primarily peripheral involvement, such as reactive arthritis.
He pointed out that these conditions have overlapping symptoms, including enthesitis, dactylitis, and uveitis, and inflammatory arthritis.
Daniel Wendling, MD, PhD, from the Centre Hospitalier Régional Universitaire de Besançon (France), Université de Franche-Comté, and colleagues agreed.
“The criteria currently available for both SpA [ASAS (Assessment of Spondyloarthritis International Society) criteria] and PsA [CASPAR (Classification for Psoriatic Arthritis) criteria] are classification criteria, not diagnostic criteria. They are not very stringent and are not exclusive. Thus, the same patient can easily be classified simultaneously in both entities, making the distinction between axSpA with psoriasis and axPsA theoretical,” they wrote in an editorial published in Joint Bone Spine.
They cited as an example of the allegedly fuzzy criteria a prospective study conducted by the investigators in Bath, England, in which modified New York criteria for AS were met by 24% of patients with AS, and CASPAR criteria for PsA were met by an equal number of patients with AS.
Therapeutic implications
Dr. Wendling and colleagues acknowledge the differences cited in studies by Dr. Gladman, Dr. Mease, and others between patients with axPsA and those with axSpA, but argue that the differences are not that great and not so clear.
“It should also be emphasized that, although some differences between axPsA and axSpA reach statistical significance, they are mostly at the margin, with low odd ratios,” they wrote.
“It is also important to consider the variability in the definition of axPsA, sometimes simply ‘physician reported’ and elsewhere based on the modified New York radiographic criteria; the latter are only present late in the course of the disease, and this may induce bias,” they continued.
Dr. Singh agreed that, as noted by Dr. Gladman, some patients will respond to anti–IL-17, anti–IL-23, and anti–IL-12/23 agents, whereas others will have better responses with tumor necrosis factor (TNF) inhibitors, and still others, such as those with peripheral involvement in the hands and feet may fare better with nonbiologic disease-modifying antirheumatic drugs such as methotrexate.
Answers to come?
Dr. Gladman noted that the information available to date about the efficacy of IL-23 inhibition in axPsA is based on a post hoc analysis of the PSUMMIT 1 and 2 controlled trials in PsA, and is not definitive.
The randomized, controlled STAR trial, currently recruiting patients, is designed to see whether guselkumab can reduce axial symptoms and inflammation in patients with active axPsA.
“What I say is, there is a rationale for [anti–IL-23] to work in psoriatic arthritis, and not work in ankylosing spondylitis,” she said.
In contrast, IL-17 inhibitors, anti-TNF agents, and Janus kinase inhibitors show efficacy against both axPsA and AS. Rituximab is ineffective against PsA, but has shown efficacy against AS, especially in patients with neurologic complications from anti-TNF agents.
“There may be other medications that would work more specifically in axial psoriatic arthritis that don’t work in ankylosing spondylitis, but at least recognizing that there may be some differences, and that therefore a correct diagnosis should be obtained, might be important,” she said.
Ideally, the picture will become clearer with results from the ongoing Axial Involvement in Psoriatic Arthritis cohort, a joint project of ASAS and the Group for Research and Assessment of Psoriasis and Psoriatic Arthritis. The multinational, cross-sectional study is designed “to systematically evaluate clinical and imaging manifestations indicative of axial involvement in patients with PsA and to develop classification criteria and a unified nomenclature for axial involvement in PsA that would allow defining a homogeneous subgroup of patients for research.”
Stay tuned.
Dr. Gladman’s research is supported by a grant from the Krembil Foundation. Dr. Singh disclosed research support from various companies. Funding sources and conflict of interest disclosures from other works cited are contained in their respective references.
Care for patients with gout needs improvement, says doctor
said a presenter at the annual meeting of the American College of Physicians. Failure to understand the disease process and goals of urate-lowering therapy (ULT) is a key barrier to achieving optimal gout therapy.
“There’s too much focus on the flare and too little focus on the urate burden of the disease. Regardless of the clinical setting, the goal should be to manage [high] serum uric acid levels,” said Lawrence Edwards, MD, professor at the University of Florida, Gainesville, during his talk.
Dr. Edwards, who specializes in treating patients with gout and rheumatoid arthritis, discussed the role of primary care providers in the treatment of gout. “We can and must do better,” he said.
Understanding the pathology of gout is key to effective treatment
Knowledge of the molecular pathology of gout has advanced drastically over the last few years. “The improved understanding of the molecules involved in disease initiation and progression can help us make better treatment decisions depending on the stage of the disease,” Dr. Edwards said.
Gout is caused by the deposition of monosodium urate (MSU) crystals, which starts as asymptomatic hyperuricemia, he said. Inflammatory responses to MSU crystals are responsible for gout flares, the frequency of which increases as the disease progresses.
Innate immune responses driven by macrophages and neutrophils play a crucial role in acute gout attacks. In the molecular pathway, proinflammatory cytokines IL-1 beta and IL-6 are the mediators of gout flares, whereas IL-8 accumulates over time and contributes to disease progression and systemic illness. If left untreated or undertreated, the repeated inflammatory reaction leads to advanced gout. The urate burden also increases with disease progression.
“Physicians need to better educate themselves on the destructive nature of this inflammatory arthritis and the need for effective urate-lowering therapy in the management of gout,” Dr. Edwards said.
Management of acute gout attacks
The management of gout flares involves the use of pharmacological agents to control pain and inflammation. The three most common anti-inflammatory therapies are colchicine, NSAIDs, and corticosteroids (either oral or intramuscular).
The choice of which of these should be used alone or in combination for a flare is based on previous tolerance of the medication or the presence of diabetes, kidney disease, heart disease, or a history of upper gastrointestinal bleeding. Dr. Edwards referred internists to the 2020 American College of Rheumatology gout management guideline.
“Regardless of which therapy is chosen, the more important consideration is how quickly the patients can start treatment after the flare begins,” said Dr. Edwards when asked about priorities in the management of gout flares. “This means that the patient should have ready access to whichever the chosen approach is. We call this the ‘pill-in-the-pocket’ approach,” he added.
Reducing the urate burden is also important for effective treatment. The serum urate level is the primary marker of how well a patient’s gout is being managed. ULT should be initiated in patients with subcutaneous tophi, gout-related radiographic damage, or frequent flares (≥ 2 per year). Allopurinol is typically the first-line ULT of choice.
Dr. Edwards noted that far too much focus is placed on flare treatment rather than addressing the underlying sources of gouty symptoms – the elevated serum levels of urate.
Management of advanced gout
“The management of advanced gout is challenging, and the dissolution of MSU is slow unless you take an aggressive approach,” Dr. Edwards said.
Switching to pegloticase is recommended for patients with frequent flares, nonresolving tophi, or high serum urate levels that persist despite treatment with xanthine oxidase inhibitors or other ULT agents.
“The frequency and severity of gout flares are what patients focus on, but if that’s the only focus of the treating physicians, then they are leaving the job less than halfway done. Getting the serum urate to below a level of 6.0 mg/dL is the most important aspect in the lifelong management of gout,” said Dr. Edwards.
Barriers to effective gout treatment
When asked during an interview after the session about the most important barriers to successful gout management, Allison M. Mays, MD, a geriatric medicine subspecialist at Cedars-Sinai Medical Center, Los Angeles, said that “the fact that gout mostly impacts quality of life and not necessarily mortality means that other things may take precedent.” She explained that gout typically coexists with other comorbidities, often multiple ones. Patients may also defer taking an additional medication for a disease like gout, which has only episodic discomfort.
She added that gout management involves shared decision-making between patients and the medical team – including the primary care physician, rheumatologist, orthopedist, and emergency physician. Following a visit to the urgent care or the ED for an acute flare of gout, the patient may not follow up with their primary care doctor or bring it up at their next visit for chronic management, she noted.
Dr. Edwards serves as a consultant to Horizon Pharmaceuticals, Atom Biosciences, Shanton Biosciences, and Aclaris Therapeutics. Horizon marketed pegloticase up until last month when Amgen bought the drug. Dr. Edwards is also president of Gout Education Society, and he has no financial agreement with any of the multiple companies that produce colchicine and allopurinol. Dr. Mays reported no conflicts.
said a presenter at the annual meeting of the American College of Physicians. Failure to understand the disease process and goals of urate-lowering therapy (ULT) is a key barrier to achieving optimal gout therapy.
“There’s too much focus on the flare and too little focus on the urate burden of the disease. Regardless of the clinical setting, the goal should be to manage [high] serum uric acid levels,” said Lawrence Edwards, MD, professor at the University of Florida, Gainesville, during his talk.
Dr. Edwards, who specializes in treating patients with gout and rheumatoid arthritis, discussed the role of primary care providers in the treatment of gout. “We can and must do better,” he said.
Understanding the pathology of gout is key to effective treatment
Knowledge of the molecular pathology of gout has advanced drastically over the last few years. “The improved understanding of the molecules involved in disease initiation and progression can help us make better treatment decisions depending on the stage of the disease,” Dr. Edwards said.
Gout is caused by the deposition of monosodium urate (MSU) crystals, which starts as asymptomatic hyperuricemia, he said. Inflammatory responses to MSU crystals are responsible for gout flares, the frequency of which increases as the disease progresses.
Innate immune responses driven by macrophages and neutrophils play a crucial role in acute gout attacks. In the molecular pathway, proinflammatory cytokines IL-1 beta and IL-6 are the mediators of gout flares, whereas IL-8 accumulates over time and contributes to disease progression and systemic illness. If left untreated or undertreated, the repeated inflammatory reaction leads to advanced gout. The urate burden also increases with disease progression.
“Physicians need to better educate themselves on the destructive nature of this inflammatory arthritis and the need for effective urate-lowering therapy in the management of gout,” Dr. Edwards said.
Management of acute gout attacks
The management of gout flares involves the use of pharmacological agents to control pain and inflammation. The three most common anti-inflammatory therapies are colchicine, NSAIDs, and corticosteroids (either oral or intramuscular).
The choice of which of these should be used alone or in combination for a flare is based on previous tolerance of the medication or the presence of diabetes, kidney disease, heart disease, or a history of upper gastrointestinal bleeding. Dr. Edwards referred internists to the 2020 American College of Rheumatology gout management guideline.
“Regardless of which therapy is chosen, the more important consideration is how quickly the patients can start treatment after the flare begins,” said Dr. Edwards when asked about priorities in the management of gout flares. “This means that the patient should have ready access to whichever the chosen approach is. We call this the ‘pill-in-the-pocket’ approach,” he added.
Reducing the urate burden is also important for effective treatment. The serum urate level is the primary marker of how well a patient’s gout is being managed. ULT should be initiated in patients with subcutaneous tophi, gout-related radiographic damage, or frequent flares (≥ 2 per year). Allopurinol is typically the first-line ULT of choice.
Dr. Edwards noted that far too much focus is placed on flare treatment rather than addressing the underlying sources of gouty symptoms – the elevated serum levels of urate.
Management of advanced gout
“The management of advanced gout is challenging, and the dissolution of MSU is slow unless you take an aggressive approach,” Dr. Edwards said.
Switching to pegloticase is recommended for patients with frequent flares, nonresolving tophi, or high serum urate levels that persist despite treatment with xanthine oxidase inhibitors or other ULT agents.
“The frequency and severity of gout flares are what patients focus on, but if that’s the only focus of the treating physicians, then they are leaving the job less than halfway done. Getting the serum urate to below a level of 6.0 mg/dL is the most important aspect in the lifelong management of gout,” said Dr. Edwards.
Barriers to effective gout treatment
When asked during an interview after the session about the most important barriers to successful gout management, Allison M. Mays, MD, a geriatric medicine subspecialist at Cedars-Sinai Medical Center, Los Angeles, said that “the fact that gout mostly impacts quality of life and not necessarily mortality means that other things may take precedent.” She explained that gout typically coexists with other comorbidities, often multiple ones. Patients may also defer taking an additional medication for a disease like gout, which has only episodic discomfort.
She added that gout management involves shared decision-making between patients and the medical team – including the primary care physician, rheumatologist, orthopedist, and emergency physician. Following a visit to the urgent care or the ED for an acute flare of gout, the patient may not follow up with their primary care doctor or bring it up at their next visit for chronic management, she noted.
Dr. Edwards serves as a consultant to Horizon Pharmaceuticals, Atom Biosciences, Shanton Biosciences, and Aclaris Therapeutics. Horizon marketed pegloticase up until last month when Amgen bought the drug. Dr. Edwards is also president of Gout Education Society, and he has no financial agreement with any of the multiple companies that produce colchicine and allopurinol. Dr. Mays reported no conflicts.
said a presenter at the annual meeting of the American College of Physicians. Failure to understand the disease process and goals of urate-lowering therapy (ULT) is a key barrier to achieving optimal gout therapy.
“There’s too much focus on the flare and too little focus on the urate burden of the disease. Regardless of the clinical setting, the goal should be to manage [high] serum uric acid levels,” said Lawrence Edwards, MD, professor at the University of Florida, Gainesville, during his talk.
Dr. Edwards, who specializes in treating patients with gout and rheumatoid arthritis, discussed the role of primary care providers in the treatment of gout. “We can and must do better,” he said.
Understanding the pathology of gout is key to effective treatment
Knowledge of the molecular pathology of gout has advanced drastically over the last few years. “The improved understanding of the molecules involved in disease initiation and progression can help us make better treatment decisions depending on the stage of the disease,” Dr. Edwards said.
Gout is caused by the deposition of monosodium urate (MSU) crystals, which starts as asymptomatic hyperuricemia, he said. Inflammatory responses to MSU crystals are responsible for gout flares, the frequency of which increases as the disease progresses.
Innate immune responses driven by macrophages and neutrophils play a crucial role in acute gout attacks. In the molecular pathway, proinflammatory cytokines IL-1 beta and IL-6 are the mediators of gout flares, whereas IL-8 accumulates over time and contributes to disease progression and systemic illness. If left untreated or undertreated, the repeated inflammatory reaction leads to advanced gout. The urate burden also increases with disease progression.
“Physicians need to better educate themselves on the destructive nature of this inflammatory arthritis and the need for effective urate-lowering therapy in the management of gout,” Dr. Edwards said.
Management of acute gout attacks
The management of gout flares involves the use of pharmacological agents to control pain and inflammation. The three most common anti-inflammatory therapies are colchicine, NSAIDs, and corticosteroids (either oral or intramuscular).
The choice of which of these should be used alone or in combination for a flare is based on previous tolerance of the medication or the presence of diabetes, kidney disease, heart disease, or a history of upper gastrointestinal bleeding. Dr. Edwards referred internists to the 2020 American College of Rheumatology gout management guideline.
“Regardless of which therapy is chosen, the more important consideration is how quickly the patients can start treatment after the flare begins,” said Dr. Edwards when asked about priorities in the management of gout flares. “This means that the patient should have ready access to whichever the chosen approach is. We call this the ‘pill-in-the-pocket’ approach,” he added.
Reducing the urate burden is also important for effective treatment. The serum urate level is the primary marker of how well a patient’s gout is being managed. ULT should be initiated in patients with subcutaneous tophi, gout-related radiographic damage, or frequent flares (≥ 2 per year). Allopurinol is typically the first-line ULT of choice.
Dr. Edwards noted that far too much focus is placed on flare treatment rather than addressing the underlying sources of gouty symptoms – the elevated serum levels of urate.
Management of advanced gout
“The management of advanced gout is challenging, and the dissolution of MSU is slow unless you take an aggressive approach,” Dr. Edwards said.
Switching to pegloticase is recommended for patients with frequent flares, nonresolving tophi, or high serum urate levels that persist despite treatment with xanthine oxidase inhibitors or other ULT agents.
“The frequency and severity of gout flares are what patients focus on, but if that’s the only focus of the treating physicians, then they are leaving the job less than halfway done. Getting the serum urate to below a level of 6.0 mg/dL is the most important aspect in the lifelong management of gout,” said Dr. Edwards.
Barriers to effective gout treatment
When asked during an interview after the session about the most important barriers to successful gout management, Allison M. Mays, MD, a geriatric medicine subspecialist at Cedars-Sinai Medical Center, Los Angeles, said that “the fact that gout mostly impacts quality of life and not necessarily mortality means that other things may take precedent.” She explained that gout typically coexists with other comorbidities, often multiple ones. Patients may also defer taking an additional medication for a disease like gout, which has only episodic discomfort.
She added that gout management involves shared decision-making between patients and the medical team – including the primary care physician, rheumatologist, orthopedist, and emergency physician. Following a visit to the urgent care or the ED for an acute flare of gout, the patient may not follow up with their primary care doctor or bring it up at their next visit for chronic management, she noted.
Dr. Edwards serves as a consultant to Horizon Pharmaceuticals, Atom Biosciences, Shanton Biosciences, and Aclaris Therapeutics. Horizon marketed pegloticase up until last month when Amgen bought the drug. Dr. Edwards is also president of Gout Education Society, and he has no financial agreement with any of the multiple companies that produce colchicine and allopurinol. Dr. Mays reported no conflicts.
AT INTERNAL MEDICINE 2023
Normal CRP during RA flares: An ‘underappreciated, persistent phenotype’
MANCHESTER, England – Even when C-reactive protein (CRP) levels are normal, patients with seropositive rheumatoid arthritis (RA) could still be experiencing significant disease that persists over time, researchers from University College London have found.
Similar levels of joint erosion and disease activity were observed over a 5-year period; researchers compared patients who had high CRP levels (> 5 mg/L)* with patients whose CRP levels were consistently normal (< 5 mg/L) at the time of an ultrasound-proven disease flare.
“Our data suggests that the phenotype of normal CRP represents at least 5% of our cohort,” Bhavika Sethi, MBChB, reported in a virtual poster presentation at the annual meeting of the British Society for Rheumatology.
“They are more likely to require biologic treatment, and this continues on even though they have equivalent DAS28 [disease activity score in 28 joints] and risk of joint damage” to high-CRP patients, she said.
These patients are a significant minority, Dr. Sethi added, and “we need to think about how we provide care for them and allocate resources.”
Diagnostic delay and poor outcomes previously seen
The study is a continuation of a larger project, the corresponding author for the poster, Matthew Hutchinson, MBChB, told this news organization.
A few years ago, Dr. Hutchinson explained, a subset of patients with normal CRP levels during RA flares were identified and were found to be more likely to have experienced diagnostic delay and worse outcomes than did those with high CRP levels.
The aim of the current study was to see whether those findings persisted by longitudinally assessing patient records and seeing what happened 1, 2, and 5 years later. They evaluated 312 patients with seropositive RA, of whom 28 had CRP < 5 mg/L as well as active disease, which was determined on the basis of a DAS28 > 4.5. Of those 28 patients, 16 had persistently low CRP (< 5 mg/L) despite active disease. All patients who were taking tocilizumab were excluded from the study because of its CRP-lowering properties.
“Our project was showing that this group of people exist, trying to characterize them a little better” and that the study serves as a “jumping-off point” for future research, Dr. Hutchinson said.
The study was also conducted to “make people more aware of [patients with normal CRP during flare], because treating clinicians could be falsely reassured by a normal CRP,” he added. “Patients in front of them could actually be undertreated and have worse outcomes if [it is] not picked up,” Dr. Hutchinson suggested.
In comparison with those with high CRP levels, those with normal CRP levels were more likely to be receiving biologic treatment at 5 years (76.6% vs. 44.4%; P = .0323).
At 5 years, DAS28 was similar (P = .9615) among patients with normal CRP levels and those with high CRP levels, at a median of 2.8 and 3.2, respectively. A similar percentage of patients in these two groups also had joint damage (63.3% vs. 71.4%; P = .7384).
Don’t rely only on CRP to diagnose and manage RA flares
“CRP is a generic inflammatory marker in most people,” Dr. Hutchinson said. “In the majority of situations when either there is inflammation or an infection, certainly if it’s systemic infection or inflammation, you will find CRP being elevated on the blood tests.”
For someone presenting with joint pain, high CRP can be a useful indicator that it’s more of an inflammatory process than physical injury, he added. CRP is also frequently used to calculate DAS28 to monitor disease activity.
“This study highlights that CRP may be normal during flares in some people with RA,” Jeffrey A. Sparks, MD, told this news organization.
“These patients may still require advanced therapies and can accrue damage,” the rheumatologist from Brigham and Women’s Hospital and Harvard University, Boston, added.
“Clinicians should not only rely on CRP to diagnose and manage RA flares,” said Dr. Sparks, who was not involved in the study.
The study was independently supported. Dr. Hutchinson and Dr. Sethi report no relevant financial relationships. Dr. Sparks is supported by the National Institute of Arthritis and Musculoskeletal and Skin Diseases, the R. Bruce and Joan M. Mickey Research Scholar Fund, and the Llura Gund Award for Rheumatoid Arthritis Research and Care; he has received research support from Bristol-Myers Squibb and has performed consultancy for AbbVie, Amgen, Boehringer Ingelheim, Bristol-Myers Squibb, Gilead, Inova Diagnostics, Janssen, Optum, and Pfizer.
*Correction, 5/9/2023: This article has been updated to correct the units for C-reactive protein from mg/dL to mg/L.
A version of this article first appeared on Medscape.com.
MANCHESTER, England – Even when C-reactive protein (CRP) levels are normal, patients with seropositive rheumatoid arthritis (RA) could still be experiencing significant disease that persists over time, researchers from University College London have found.
Similar levels of joint erosion and disease activity were observed over a 5-year period; researchers compared patients who had high CRP levels (> 5 mg/L)* with patients whose CRP levels were consistently normal (< 5 mg/L) at the time of an ultrasound-proven disease flare.
“Our data suggests that the phenotype of normal CRP represents at least 5% of our cohort,” Bhavika Sethi, MBChB, reported in a virtual poster presentation at the annual meeting of the British Society for Rheumatology.
“They are more likely to require biologic treatment, and this continues on even though they have equivalent DAS28 [disease activity score in 28 joints] and risk of joint damage” to high-CRP patients, she said.
These patients are a significant minority, Dr. Sethi added, and “we need to think about how we provide care for them and allocate resources.”
Diagnostic delay and poor outcomes previously seen
The study is a continuation of a larger project, the corresponding author for the poster, Matthew Hutchinson, MBChB, told this news organization.
A few years ago, Dr. Hutchinson explained, a subset of patients with normal CRP levels during RA flares were identified and were found to be more likely to have experienced diagnostic delay and worse outcomes than did those with high CRP levels.
The aim of the current study was to see whether those findings persisted by longitudinally assessing patient records and seeing what happened 1, 2, and 5 years later. They evaluated 312 patients with seropositive RA, of whom 28 had CRP < 5 mg/L as well as active disease, which was determined on the basis of a DAS28 > 4.5. Of those 28 patients, 16 had persistently low CRP (< 5 mg/L) despite active disease. All patients who were taking tocilizumab were excluded from the study because of its CRP-lowering properties.
“Our project was showing that this group of people exist, trying to characterize them a little better” and that the study serves as a “jumping-off point” for future research, Dr. Hutchinson said.
The study was also conducted to “make people more aware of [patients with normal CRP during flare], because treating clinicians could be falsely reassured by a normal CRP,” he added. “Patients in front of them could actually be undertreated and have worse outcomes if [it is] not picked up,” Dr. Hutchinson suggested.
In comparison with those with high CRP levels, those with normal CRP levels were more likely to be receiving biologic treatment at 5 years (76.6% vs. 44.4%; P = .0323).
At 5 years, DAS28 was similar (P = .9615) among patients with normal CRP levels and those with high CRP levels, at a median of 2.8 and 3.2, respectively. A similar percentage of patients in these two groups also had joint damage (63.3% vs. 71.4%; P = .7384).
Don’t rely only on CRP to diagnose and manage RA flares
“CRP is a generic inflammatory marker in most people,” Dr. Hutchinson said. “In the majority of situations when either there is inflammation or an infection, certainly if it’s systemic infection or inflammation, you will find CRP being elevated on the blood tests.”
For someone presenting with joint pain, high CRP can be a useful indicator that it’s more of an inflammatory process than physical injury, he added. CRP is also frequently used to calculate DAS28 to monitor disease activity.
“This study highlights that CRP may be normal during flares in some people with RA,” Jeffrey A. Sparks, MD, told this news organization.
“These patients may still require advanced therapies and can accrue damage,” the rheumatologist from Brigham and Women’s Hospital and Harvard University, Boston, added.
“Clinicians should not only rely on CRP to diagnose and manage RA flares,” said Dr. Sparks, who was not involved in the study.
The study was independently supported. Dr. Hutchinson and Dr. Sethi report no relevant financial relationships. Dr. Sparks is supported by the National Institute of Arthritis and Musculoskeletal and Skin Diseases, the R. Bruce and Joan M. Mickey Research Scholar Fund, and the Llura Gund Award for Rheumatoid Arthritis Research and Care; he has received research support from Bristol-Myers Squibb and has performed consultancy for AbbVie, Amgen, Boehringer Ingelheim, Bristol-Myers Squibb, Gilead, Inova Diagnostics, Janssen, Optum, and Pfizer.
*Correction, 5/9/2023: This article has been updated to correct the units for C-reactive protein from mg/dL to mg/L.
A version of this article first appeared on Medscape.com.
MANCHESTER, England – Even when C-reactive protein (CRP) levels are normal, patients with seropositive rheumatoid arthritis (RA) could still be experiencing significant disease that persists over time, researchers from University College London have found.
Similar levels of joint erosion and disease activity were observed over a 5-year period; researchers compared patients who had high CRP levels (> 5 mg/L)* with patients whose CRP levels were consistently normal (< 5 mg/L) at the time of an ultrasound-proven disease flare.
“Our data suggests that the phenotype of normal CRP represents at least 5% of our cohort,” Bhavika Sethi, MBChB, reported in a virtual poster presentation at the annual meeting of the British Society for Rheumatology.
“They are more likely to require biologic treatment, and this continues on even though they have equivalent DAS28 [disease activity score in 28 joints] and risk of joint damage” to high-CRP patients, she said.
These patients are a significant minority, Dr. Sethi added, and “we need to think about how we provide care for them and allocate resources.”
Diagnostic delay and poor outcomes previously seen
The study is a continuation of a larger project, the corresponding author for the poster, Matthew Hutchinson, MBChB, told this news organization.
A few years ago, Dr. Hutchinson explained, a subset of patients with normal CRP levels during RA flares were identified and were found to be more likely to have experienced diagnostic delay and worse outcomes than did those with high CRP levels.
The aim of the current study was to see whether those findings persisted by longitudinally assessing patient records and seeing what happened 1, 2, and 5 years later. They evaluated 312 patients with seropositive RA, of whom 28 had CRP < 5 mg/L as well as active disease, which was determined on the basis of a DAS28 > 4.5. Of those 28 patients, 16 had persistently low CRP (< 5 mg/L) despite active disease. All patients who were taking tocilizumab were excluded from the study because of its CRP-lowering properties.
“Our project was showing that this group of people exist, trying to characterize them a little better” and that the study serves as a “jumping-off point” for future research, Dr. Hutchinson said.
The study was also conducted to “make people more aware of [patients with normal CRP during flare], because treating clinicians could be falsely reassured by a normal CRP,” he added. “Patients in front of them could actually be undertreated and have worse outcomes if [it is] not picked up,” Dr. Hutchinson suggested.
In comparison with those with high CRP levels, those with normal CRP levels were more likely to be receiving biologic treatment at 5 years (76.6% vs. 44.4%; P = .0323).
At 5 years, DAS28 was similar (P = .9615) among patients with normal CRP levels and those with high CRP levels, at a median of 2.8 and 3.2, respectively. A similar percentage of patients in these two groups also had joint damage (63.3% vs. 71.4%; P = .7384).
Don’t rely only on CRP to diagnose and manage RA flares
“CRP is a generic inflammatory marker in most people,” Dr. Hutchinson said. “In the majority of situations when either there is inflammation or an infection, certainly if it’s systemic infection or inflammation, you will find CRP being elevated on the blood tests.”
For someone presenting with joint pain, high CRP can be a useful indicator that it’s more of an inflammatory process than physical injury, he added. CRP is also frequently used to calculate DAS28 to monitor disease activity.
“This study highlights that CRP may be normal during flares in some people with RA,” Jeffrey A. Sparks, MD, told this news organization.
“These patients may still require advanced therapies and can accrue damage,” the rheumatologist from Brigham and Women’s Hospital and Harvard University, Boston, added.
“Clinicians should not only rely on CRP to diagnose and manage RA flares,” said Dr. Sparks, who was not involved in the study.
The study was independently supported. Dr. Hutchinson and Dr. Sethi report no relevant financial relationships. Dr. Sparks is supported by the National Institute of Arthritis and Musculoskeletal and Skin Diseases, the R. Bruce and Joan M. Mickey Research Scholar Fund, and the Llura Gund Award for Rheumatoid Arthritis Research and Care; he has received research support from Bristol-Myers Squibb and has performed consultancy for AbbVie, Amgen, Boehringer Ingelheim, Bristol-Myers Squibb, Gilead, Inova Diagnostics, Janssen, Optum, and Pfizer.
*Correction, 5/9/2023: This article has been updated to correct the units for C-reactive protein from mg/dL to mg/L.
A version of this article first appeared on Medscape.com.
AT BSR 2023
COVID-19 and psoriasis: Is there a link?
.
Psoriasis has several well-established triggers, including stress, skin injury, cold or warm air, and allergies. Illnesses like strep throat can also cause a psoriasis flare in some people – and it appears COVID may also do so. “Psoriasis flares have long been associated with bacterial and viral infections, particularly a form of psoriasis called guttate, which is characterized by tons of tiny red scaly bumps all over the body,” said Joel M. Gelfand, MD, a professor of dermatology and epidemiology at the University of Pennsylvania, Philadelphia. “Infection with COVID-19 has been associated with flares of guttate and pustular psoriasis, and even psoriasis that affects 100% of the skin ... in many published case reports.”
Israeli researchers recently found that psoriasis patients have a slightly higher risk of getting COVID, although they are not at higher risk of hospitalization or death, which could be related to treatment with immune-modulating therapy, which can increase their risk of infections.
How could COVID cause psoriasis to flare?
Psoriasis is an autoimmune condition, and inflammation can cause symptoms.
Investigators for a study from Albany (N.Y.) Medical College and Weirton (Pa.) Medical Center found that people in the study who were already diagnosed with the skin condition had an unexpected flare within a week to a month after testing positive for COVID. New psoriasis after a COVID infection was also found. The researchers think this could be because COVID causes inflammation in the body, which negatively affects previously well-controlled psoriasis. They also think it’s possible that COVID-related inflammation could trigger a genetic tendency to have psoriasis, which may explain why it can appear for the first time after a positive test.
“A viral infection like COVID-19 can signal the release of proinflammatory factors that can appear as rashes, such as with psoriasis.” said Robert O. Carpenter, MD, director of wellness at Texas A&M University in Bryan.
What are the symptoms of COVID-related psoriasis?
The signs are the same as those of any form of psoriasis.
For a patient with psoriasis, will COVID automatically make it worse?
Not necessarily.
“Psoriasis is a common condition, so people should be aware that new psoriasis that develops may not be related to COVID-19,” said Esther Freeman MD, PhD, director of global health dermatology at Massachusetts General Hospital in Boston.
As with every aspect of COVID, doctors and scientists are still learning about how serious and widespread a problem psoriasis after COVID-19 may be. “We have seen case reports that psoriasis can flare after COVID-19,” said Dr. Freeman, who is also an associate professor of dermatology at Harvard Medical School. “I will say, this has not been a tidal wave – more like sporadic cases here and there. So I do not think psoriasis flares are a major post-COVID finding, nor do they necessarily mean you have long COVID. That being said, we know that many different infections can cause psoriasis flares, and so, in that respect, it’s not that surprising that SARS-CoV-2, like other infections, could trigger a psoriasis flare.”
Could getting COVID more than once cause psoriasis to flare? It’s possible.
“Your body can change after having COVID-19,” said Dr. Carpenter. “We don’t know the long-term implications, but having COVID-19 repeatedly can increase the risk of long COVID, which can cause many systemic changes in your body.”
Another important point: For patients who take biologics for treating psoriasis, getting vaccinated and boosted for COVID is an important step to take to help protect themselves.
Is psoriasis itself a potential symptom of COVID?
“Yes, but we don’t know the frequency at which this may occur, and a causal relationship is difficult to establish from just case reports,” said Dr. Gelfand, who’s also medical director of the clinical studies unit in the department of dermatology at his university. “Typically, if a patient presents with a flare of psoriasis, particularly guttate, pustular, or erythrodermic forms, an infectious trigger should be considered, and testing for strep and possibly COVID-19 may be appropriate.”
A version of this article first appeared on Medscape.com.
.
Psoriasis has several well-established triggers, including stress, skin injury, cold or warm air, and allergies. Illnesses like strep throat can also cause a psoriasis flare in some people – and it appears COVID may also do so. “Psoriasis flares have long been associated with bacterial and viral infections, particularly a form of psoriasis called guttate, which is characterized by tons of tiny red scaly bumps all over the body,” said Joel M. Gelfand, MD, a professor of dermatology and epidemiology at the University of Pennsylvania, Philadelphia. “Infection with COVID-19 has been associated with flares of guttate and pustular psoriasis, and even psoriasis that affects 100% of the skin ... in many published case reports.”
Israeli researchers recently found that psoriasis patients have a slightly higher risk of getting COVID, although they are not at higher risk of hospitalization or death, which could be related to treatment with immune-modulating therapy, which can increase their risk of infections.
How could COVID cause psoriasis to flare?
Psoriasis is an autoimmune condition, and inflammation can cause symptoms.
Investigators for a study from Albany (N.Y.) Medical College and Weirton (Pa.) Medical Center found that people in the study who were already diagnosed with the skin condition had an unexpected flare within a week to a month after testing positive for COVID. New psoriasis after a COVID infection was also found. The researchers think this could be because COVID causes inflammation in the body, which negatively affects previously well-controlled psoriasis. They also think it’s possible that COVID-related inflammation could trigger a genetic tendency to have psoriasis, which may explain why it can appear for the first time after a positive test.
“A viral infection like COVID-19 can signal the release of proinflammatory factors that can appear as rashes, such as with psoriasis.” said Robert O. Carpenter, MD, director of wellness at Texas A&M University in Bryan.
What are the symptoms of COVID-related psoriasis?
The signs are the same as those of any form of psoriasis.
For a patient with psoriasis, will COVID automatically make it worse?
Not necessarily.
“Psoriasis is a common condition, so people should be aware that new psoriasis that develops may not be related to COVID-19,” said Esther Freeman MD, PhD, director of global health dermatology at Massachusetts General Hospital in Boston.
As with every aspect of COVID, doctors and scientists are still learning about how serious and widespread a problem psoriasis after COVID-19 may be. “We have seen case reports that psoriasis can flare after COVID-19,” said Dr. Freeman, who is also an associate professor of dermatology at Harvard Medical School. “I will say, this has not been a tidal wave – more like sporadic cases here and there. So I do not think psoriasis flares are a major post-COVID finding, nor do they necessarily mean you have long COVID. That being said, we know that many different infections can cause psoriasis flares, and so, in that respect, it’s not that surprising that SARS-CoV-2, like other infections, could trigger a psoriasis flare.”
Could getting COVID more than once cause psoriasis to flare? It’s possible.
“Your body can change after having COVID-19,” said Dr. Carpenter. “We don’t know the long-term implications, but having COVID-19 repeatedly can increase the risk of long COVID, which can cause many systemic changes in your body.”
Another important point: For patients who take biologics for treating psoriasis, getting vaccinated and boosted for COVID is an important step to take to help protect themselves.
Is psoriasis itself a potential symptom of COVID?
“Yes, but we don’t know the frequency at which this may occur, and a causal relationship is difficult to establish from just case reports,” said Dr. Gelfand, who’s also medical director of the clinical studies unit in the department of dermatology at his university. “Typically, if a patient presents with a flare of psoriasis, particularly guttate, pustular, or erythrodermic forms, an infectious trigger should be considered, and testing for strep and possibly COVID-19 may be appropriate.”
A version of this article first appeared on Medscape.com.
.
Psoriasis has several well-established triggers, including stress, skin injury, cold or warm air, and allergies. Illnesses like strep throat can also cause a psoriasis flare in some people – and it appears COVID may also do so. “Psoriasis flares have long been associated with bacterial and viral infections, particularly a form of psoriasis called guttate, which is characterized by tons of tiny red scaly bumps all over the body,” said Joel M. Gelfand, MD, a professor of dermatology and epidemiology at the University of Pennsylvania, Philadelphia. “Infection with COVID-19 has been associated with flares of guttate and pustular psoriasis, and even psoriasis that affects 100% of the skin ... in many published case reports.”
Israeli researchers recently found that psoriasis patients have a slightly higher risk of getting COVID, although they are not at higher risk of hospitalization or death, which could be related to treatment with immune-modulating therapy, which can increase their risk of infections.
How could COVID cause psoriasis to flare?
Psoriasis is an autoimmune condition, and inflammation can cause symptoms.
Investigators for a study from Albany (N.Y.) Medical College and Weirton (Pa.) Medical Center found that people in the study who were already diagnosed with the skin condition had an unexpected flare within a week to a month after testing positive for COVID. New psoriasis after a COVID infection was also found. The researchers think this could be because COVID causes inflammation in the body, which negatively affects previously well-controlled psoriasis. They also think it’s possible that COVID-related inflammation could trigger a genetic tendency to have psoriasis, which may explain why it can appear for the first time after a positive test.
“A viral infection like COVID-19 can signal the release of proinflammatory factors that can appear as rashes, such as with psoriasis.” said Robert O. Carpenter, MD, director of wellness at Texas A&M University in Bryan.
What are the symptoms of COVID-related psoriasis?
The signs are the same as those of any form of psoriasis.
For a patient with psoriasis, will COVID automatically make it worse?
Not necessarily.
“Psoriasis is a common condition, so people should be aware that new psoriasis that develops may not be related to COVID-19,” said Esther Freeman MD, PhD, director of global health dermatology at Massachusetts General Hospital in Boston.
As with every aspect of COVID, doctors and scientists are still learning about how serious and widespread a problem psoriasis after COVID-19 may be. “We have seen case reports that psoriasis can flare after COVID-19,” said Dr. Freeman, who is also an associate professor of dermatology at Harvard Medical School. “I will say, this has not been a tidal wave – more like sporadic cases here and there. So I do not think psoriasis flares are a major post-COVID finding, nor do they necessarily mean you have long COVID. That being said, we know that many different infections can cause psoriasis flares, and so, in that respect, it’s not that surprising that SARS-CoV-2, like other infections, could trigger a psoriasis flare.”
Could getting COVID more than once cause psoriasis to flare? It’s possible.
“Your body can change after having COVID-19,” said Dr. Carpenter. “We don’t know the long-term implications, but having COVID-19 repeatedly can increase the risk of long COVID, which can cause many systemic changes in your body.”
Another important point: For patients who take biologics for treating psoriasis, getting vaccinated and boosted for COVID is an important step to take to help protect themselves.
Is psoriasis itself a potential symptom of COVID?
“Yes, but we don’t know the frequency at which this may occur, and a causal relationship is difficult to establish from just case reports,” said Dr. Gelfand, who’s also medical director of the clinical studies unit in the department of dermatology at his university. “Typically, if a patient presents with a flare of psoriasis, particularly guttate, pustular, or erythrodermic forms, an infectious trigger should be considered, and testing for strep and possibly COVID-19 may be appropriate.”
A version of this article first appeared on Medscape.com.