User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'main-prefix')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
div[contains(@class, 'view-medstat-quiz-listing-panes')]
div[contains(@class, 'pane-article-sidebar-latest-news')]
div[contains(@class, 'medstat-accordion-set article-series')]
How to recognize and treat hidden inflammation
“This then leads to inflammation, the healing of which the body is unable to keep under control,” explained Ulf Müller-Ladner, MD, PhD, chairperson of the German Society of Internal Medicine.
At the DGIM annual press conference, Dr. Müller-Ladner, who is also director of the department of rheumatology and clinical immunology at the Kerckhoff Clinic in Bad Nauheim, Germany, explained how IgG4 inflammation is triggered throughout the body and what therapeutic options are available.
Many manifestations
IgG4-associated inflammation can affect one or more organs or the surrounding connective tissue and cause fibrosis. As a result of fibrosis, the organ gradually loses function and is eventually transformed completely into scarred connective tissue.
“In the case of IgG4-associated inflammation, these fibroses have a histological structure, but extracting a sample is not possible from every affected organ,” said Dr. Müller-Ladner. Liver, bile ducts, blood vessels, skin, eyes, or even the central nervous system – practically every organ system can be affected by these inflammatory reactions.
IgG4-associated diseases have likely been around for some time, but it is only in the past 10 years that awareness has grown that, despite various manifestations, “they are all one and the same disease,” said Dr. Müller-Ladner.
IgG4-associated chronic, inflammatory, fibrosing diseases were only classified together as a single entity in the past few years. In terms of pathophysiology, B lymphocytes, IgG4-positive plasma cells, follicular T-helper cells, cytotoxic CD4-positive T cells, and macrophages work together and trigger an inflammatory reaction, which then encourages fibroblasts to overproduce connective tissue.
Beware inexplicable inflammation
It is estimated that 1 in 100,000 people suffer from the disease, but the number of incorrectly categorized patients may be significantly higher.
The diagnostic challenge lies in the fact that IgG4-associated inflammation occurs in almost every organ. It can cause different symptoms, depending on the organ affected.
Dr. Müller-Ladner provided the following take-home message: “Every inexplicable inflammation event and every organ dysfunction, especially if associated with an increase in connective tissue, could be an IgG4-associated disease. Keeping this in mind is the key to recovery.”
With most people, the inflammation persists for many years before any symptoms of the disease develop. Highly acute courses of progression are also possible.
Classic symptoms, such as fever, are not so characteristic of the latent inflammatory reaction, and according to classification criteria published by specialist rheumatology societies, they are an exclusion criterion. This is true with respect to the differential diagnosis for vasculitis, which also occurs throughout the body.
Histology is key
Blood levels of IgG4 and imaging are not always enough to confirm the diagnosis. In such cases, the histology is often a crucial factor in making a definitive diagnosis. Dominant organs in IgG4-associated diseases are the pancreas, the liver, the gallbladder, the intestines, the retroperitoneum, large blood vessels, the kidneys, the heart, the brain, saliva, tear ducts, as well all of the body’s connective tissue.
The kidneys play host to inflammation in the connective tissue and space-occupying masses in particular. “If the pancreas is affected, the signs can vary from diffuse swelling to the onset of diabetes mellitus. In contrast, if the aorta is affected, then the inflammation is characterized through a thickening of the vessel walls, aneurysms, and the corresponding circulation disorders,” said Dr. Müller-Ladner.
Because of the long period before the diagnosis is made, more than 50% of patients exhibit irreversible organ damage at the time of diagnosis, he added.
Glucocorticoids and immunosuppressants
Despite therapeutic intervention, the disease can have a fatal outcome, even if the patient is young, said Dr. Müller-Ladner. Glucocorticoids are the current therapy of choice. The dose is more than 0.5 mg of prednisolone equivalent per kg of body weight. “This usually leads to a rapid improvement in the inflammation. Subsequently, every organ is thoroughly diagnosed to assess the severity of the disease and to plan further treatment steps.”
In the long term, proven immunosuppressants, such as azathioprine, mycophenolate, leflunomide, and methotrexate, can be used, just as for many other chronic inflammatory diseases. Cyclophosphamide or cyclosporine is used more rarely, owing to their side effect profiles.
Because of the B-cell dominance, B-cell–depleting therapy with rituximab is currently a highly effective therapeutic option but one that must be applied for, because such use is off label. “If the body responds well to the medication, organ function often recovers,” said Dr. Müller-Ladner.
This article was translated from the Medscape German edition. A version appeared on Medscape.com.
“This then leads to inflammation, the healing of which the body is unable to keep under control,” explained Ulf Müller-Ladner, MD, PhD, chairperson of the German Society of Internal Medicine.
At the DGIM annual press conference, Dr. Müller-Ladner, who is also director of the department of rheumatology and clinical immunology at the Kerckhoff Clinic in Bad Nauheim, Germany, explained how IgG4 inflammation is triggered throughout the body and what therapeutic options are available.
Many manifestations
IgG4-associated inflammation can affect one or more organs or the surrounding connective tissue and cause fibrosis. As a result of fibrosis, the organ gradually loses function and is eventually transformed completely into scarred connective tissue.
“In the case of IgG4-associated inflammation, these fibroses have a histological structure, but extracting a sample is not possible from every affected organ,” said Dr. Müller-Ladner. Liver, bile ducts, blood vessels, skin, eyes, or even the central nervous system – practically every organ system can be affected by these inflammatory reactions.
IgG4-associated diseases have likely been around for some time, but it is only in the past 10 years that awareness has grown that, despite various manifestations, “they are all one and the same disease,” said Dr. Müller-Ladner.
IgG4-associated chronic, inflammatory, fibrosing diseases were only classified together as a single entity in the past few years. In terms of pathophysiology, B lymphocytes, IgG4-positive plasma cells, follicular T-helper cells, cytotoxic CD4-positive T cells, and macrophages work together and trigger an inflammatory reaction, which then encourages fibroblasts to overproduce connective tissue.
Beware inexplicable inflammation
It is estimated that 1 in 100,000 people suffer from the disease, but the number of incorrectly categorized patients may be significantly higher.
The diagnostic challenge lies in the fact that IgG4-associated inflammation occurs in almost every organ. It can cause different symptoms, depending on the organ affected.
Dr. Müller-Ladner provided the following take-home message: “Every inexplicable inflammation event and every organ dysfunction, especially if associated with an increase in connective tissue, could be an IgG4-associated disease. Keeping this in mind is the key to recovery.”
With most people, the inflammation persists for many years before any symptoms of the disease develop. Highly acute courses of progression are also possible.
Classic symptoms, such as fever, are not so characteristic of the latent inflammatory reaction, and according to classification criteria published by specialist rheumatology societies, they are an exclusion criterion. This is true with respect to the differential diagnosis for vasculitis, which also occurs throughout the body.
Histology is key
Blood levels of IgG4 and imaging are not always enough to confirm the diagnosis. In such cases, the histology is often a crucial factor in making a definitive diagnosis. Dominant organs in IgG4-associated diseases are the pancreas, the liver, the gallbladder, the intestines, the retroperitoneum, large blood vessels, the kidneys, the heart, the brain, saliva, tear ducts, as well all of the body’s connective tissue.
The kidneys play host to inflammation in the connective tissue and space-occupying masses in particular. “If the pancreas is affected, the signs can vary from diffuse swelling to the onset of diabetes mellitus. In contrast, if the aorta is affected, then the inflammation is characterized through a thickening of the vessel walls, aneurysms, and the corresponding circulation disorders,” said Dr. Müller-Ladner.
Because of the long period before the diagnosis is made, more than 50% of patients exhibit irreversible organ damage at the time of diagnosis, he added.
Glucocorticoids and immunosuppressants
Despite therapeutic intervention, the disease can have a fatal outcome, even if the patient is young, said Dr. Müller-Ladner. Glucocorticoids are the current therapy of choice. The dose is more than 0.5 mg of prednisolone equivalent per kg of body weight. “This usually leads to a rapid improvement in the inflammation. Subsequently, every organ is thoroughly diagnosed to assess the severity of the disease and to plan further treatment steps.”
In the long term, proven immunosuppressants, such as azathioprine, mycophenolate, leflunomide, and methotrexate, can be used, just as for many other chronic inflammatory diseases. Cyclophosphamide or cyclosporine is used more rarely, owing to their side effect profiles.
Because of the B-cell dominance, B-cell–depleting therapy with rituximab is currently a highly effective therapeutic option but one that must be applied for, because such use is off label. “If the body responds well to the medication, organ function often recovers,” said Dr. Müller-Ladner.
This article was translated from the Medscape German edition. A version appeared on Medscape.com.
“This then leads to inflammation, the healing of which the body is unable to keep under control,” explained Ulf Müller-Ladner, MD, PhD, chairperson of the German Society of Internal Medicine.
At the DGIM annual press conference, Dr. Müller-Ladner, who is also director of the department of rheumatology and clinical immunology at the Kerckhoff Clinic in Bad Nauheim, Germany, explained how IgG4 inflammation is triggered throughout the body and what therapeutic options are available.
Many manifestations
IgG4-associated inflammation can affect one or more organs or the surrounding connective tissue and cause fibrosis. As a result of fibrosis, the organ gradually loses function and is eventually transformed completely into scarred connective tissue.
“In the case of IgG4-associated inflammation, these fibroses have a histological structure, but extracting a sample is not possible from every affected organ,” said Dr. Müller-Ladner. Liver, bile ducts, blood vessels, skin, eyes, or even the central nervous system – practically every organ system can be affected by these inflammatory reactions.
IgG4-associated diseases have likely been around for some time, but it is only in the past 10 years that awareness has grown that, despite various manifestations, “they are all one and the same disease,” said Dr. Müller-Ladner.
IgG4-associated chronic, inflammatory, fibrosing diseases were only classified together as a single entity in the past few years. In terms of pathophysiology, B lymphocytes, IgG4-positive plasma cells, follicular T-helper cells, cytotoxic CD4-positive T cells, and macrophages work together and trigger an inflammatory reaction, which then encourages fibroblasts to overproduce connective tissue.
Beware inexplicable inflammation
It is estimated that 1 in 100,000 people suffer from the disease, but the number of incorrectly categorized patients may be significantly higher.
The diagnostic challenge lies in the fact that IgG4-associated inflammation occurs in almost every organ. It can cause different symptoms, depending on the organ affected.
Dr. Müller-Ladner provided the following take-home message: “Every inexplicable inflammation event and every organ dysfunction, especially if associated with an increase in connective tissue, could be an IgG4-associated disease. Keeping this in mind is the key to recovery.”
With most people, the inflammation persists for many years before any symptoms of the disease develop. Highly acute courses of progression are also possible.
Classic symptoms, such as fever, are not so characteristic of the latent inflammatory reaction, and according to classification criteria published by specialist rheumatology societies, they are an exclusion criterion. This is true with respect to the differential diagnosis for vasculitis, which also occurs throughout the body.
Histology is key
Blood levels of IgG4 and imaging are not always enough to confirm the diagnosis. In such cases, the histology is often a crucial factor in making a definitive diagnosis. Dominant organs in IgG4-associated diseases are the pancreas, the liver, the gallbladder, the intestines, the retroperitoneum, large blood vessels, the kidneys, the heart, the brain, saliva, tear ducts, as well all of the body’s connective tissue.
The kidneys play host to inflammation in the connective tissue and space-occupying masses in particular. “If the pancreas is affected, the signs can vary from diffuse swelling to the onset of diabetes mellitus. In contrast, if the aorta is affected, then the inflammation is characterized through a thickening of the vessel walls, aneurysms, and the corresponding circulation disorders,” said Dr. Müller-Ladner.
Because of the long period before the diagnosis is made, more than 50% of patients exhibit irreversible organ damage at the time of diagnosis, he added.
Glucocorticoids and immunosuppressants
Despite therapeutic intervention, the disease can have a fatal outcome, even if the patient is young, said Dr. Müller-Ladner. Glucocorticoids are the current therapy of choice. The dose is more than 0.5 mg of prednisolone equivalent per kg of body weight. “This usually leads to a rapid improvement in the inflammation. Subsequently, every organ is thoroughly diagnosed to assess the severity of the disease and to plan further treatment steps.”
In the long term, proven immunosuppressants, such as azathioprine, mycophenolate, leflunomide, and methotrexate, can be used, just as for many other chronic inflammatory diseases. Cyclophosphamide or cyclosporine is used more rarely, owing to their side effect profiles.
Because of the B-cell dominance, B-cell–depleting therapy with rituximab is currently a highly effective therapeutic option but one that must be applied for, because such use is off label. “If the body responds well to the medication, organ function often recovers,” said Dr. Müller-Ladner.
This article was translated from the Medscape German edition. A version appeared on Medscape.com.
Telemedicine usage still high among rheumatologists as interest wanes in other specialties
There was an explosion in the use of telemedicine during the COVID-19 pandemic, but usage has stabilized and varies between specialties. However, telemedicine use is still somewhat high among rheumatologists, according to speakers at the 2023 Rheumatology Winter Clinical Symposium.
Speaking in general about the future of rheumatology, Jack Cush, MD, a rheumatologist based in Dallas and executive editor of RheumNow.com, said it is up to rheumatologists to adapt to the changing winds in the specialty.
“The future is going to happen no matter what, so the question is, are you going to go along with it? Are you going to be a part of it? Are you going to be resistant to it?” Dr. Cush asked attendees. “Your recent experience with COVID would tell you maybe what your path is going to be if you’re dying to get back to the way it once was.”
Rheumatologists can expect changes in where they work, how they’re paid, increases in their workload, and new innovations in connecting with patients, he said.
“You’re going to be integrating a new style of medicine, you’re going to be digitally connected,” he explained. “All these networks are going to be working together to make you supposedly better at what you do, or maybe they’re working together to make you obsolete – and I think you better start protecting your space.”
One major area of change, telemedicine, already occurred as a result of the COVID-19 pandemic and will “begin to dominate” over the next decade, Dr. Cush said. An analysis conducted by consulting firm McKinsey & Company found telehealth usage increased 78-fold between February and April 2020 before leveling off at a 38-fold higher rate, compared with prepandemic levels. In the same analysis, rheumatology ranked third in terms of telehealth usage claims behind psychiatry and substance use disorder treatment, Dr. Cush observed, as other specialties have “fallen off quite a bit.”
“The common denominators are chronic care, cognitive care, nonprocedural care, pattern recognition, and monitoring, and this is what you do,” he said. “This is why, in many ways, for you to abandon telemedicine I think is a gigantic mistake.”
Changes to telemedicine
The most immediate change to telemedicine will come when the Biden administration officially ends the COVID-19 public health emergency in May 2023, and temporary telehealth services will be extended for approximately 5 months after the end of the public health emergency. Legislation passed by Congress will ensure some of the flexibilities in telemedicine will be extended until the end of December 2024.
Alvin Wells, MD, PhD, director of the department of rheumatology at Advocate Aurora Health in Franklin, Wisc., said he sees telemedicine as persisting even after the official COVID-19 public health emergency ends. “There’s a lot of push from the American Medical Association, from the American College of Physicians. You’re going to see people – this will not go away because [there’s] also going to be that demand.”
Despite decreased usage since April 2020, telehealth was estimated to be a $60 billion industry in 2022 and will likely increase over the next decade, Dr. Cush noted. “I question [the decline] because I think it still is a major part of your [future in] 2033.”
The number of physicians who have at least three licenses to practice in other U.S. states increased from 50,454 in 2010 to 72,752 in 2020, and that trend will continue, Dr. Wells explained. It is now becoming easier for physicians to become licensed in other states with companies like CompHealth that offer services to simplify obtaining medical licenses with states that participate in the Interstate Medical Licensure Compact.
“It’s a telemedicine easy pass,” Dr. Cush said.
Concerns in telemedicine
Commenting on the presentation, Allan Gibofsky, MD, JD, professor of medicine at Weill Cornell Medicine and codirector of the Clinic for Inflammatory Arthritis and Biologic Therapy at the Hospital for Special Surgery (HSS), both in New York, pointed out that because telemedicine is governed by U.S. states, rather than the federal government, a physician needs to be licensed in the state where the patient is located. While many states relaxed their restrictions during COVID-19, as states began tightening their restrictions later, “many physicians didn’t want to have three licenses,” he said.
“There’s an expense in getting three licenses. There’s an expense in obtaining it and maintaining it, and the reimbursement for the telemedicine visit did not reach that expectation,” Dr. Gibofsky explained. With the exception of the orthopedic surgeons at HSS who practice in New York and a satellite office in Florida, none of the surgeons at his center have obtained more than one license to practice telemedicine in other states.
“Our volume of telemedicine at HSS has remained about the same at 30%, but fewer physicians are doing it because they don’t want to maintain multiple licensures,” he said. “So don’t overlook the role of legal concerns in terms of who’s going to be allowed to do what where. Your talk was great in terms of an exuberance of what’s going to be available, but it’s not going to relieve the physician from the burden of being responsible for their use.”
Eric Ruderman, MD, professor of rheumatology at Northwestern University, Chicago, asked the presenters about the balance between seeing patients for virtual and in-person visits. “The question is what’s the sweet spot? Are there people you’re willing to see virtually forever?” he asked, noting that he has patients scheduling telemedicine visits that he hasn’t seen since before the COVID-19 pandemic.
“That’s not going to work for me. At some point, you have to lay hands on people,” he said.
Dr. Wells said his current practice is 40% virtual, and his staff converts potential no-shows into a telemedicine consultation over the phone. “My no-show rate has gone down to zero. Somebody’s scheduled for a visit, they don’t show up, my [medical assistants] get them on the phone, they put them on hold, tee up the refills. I turn them into a telephone call,” he said. “We don’t accept the no-show at all because we can do a telephone [consultation].”
In Dr. Cush’s practice, he alternates telemedicine visits with in-person visits. “If you come back for two videos in a row, you’re catching hell from me for that,” he said. Responding to how Dr. Wells incorporates telemedicine into his practice, Dr. Cush said many rheumatologists “don’t have the setups to support the care, and that’s why it’s hard to do and that’s why we’re not as great as we could be.”
“This is the way we were trained. We’re used to seeing these patients in the clinic that often. Not every single patient needs to be seen that frequently if they’re stable and doing fine,” Dr. Wells countered.
Dr. Cush and Dr. Wells reported having financial relationships with numerous pharmaceutical companies.
There was an explosion in the use of telemedicine during the COVID-19 pandemic, but usage has stabilized and varies between specialties. However, telemedicine use is still somewhat high among rheumatologists, according to speakers at the 2023 Rheumatology Winter Clinical Symposium.
Speaking in general about the future of rheumatology, Jack Cush, MD, a rheumatologist based in Dallas and executive editor of RheumNow.com, said it is up to rheumatologists to adapt to the changing winds in the specialty.
“The future is going to happen no matter what, so the question is, are you going to go along with it? Are you going to be a part of it? Are you going to be resistant to it?” Dr. Cush asked attendees. “Your recent experience with COVID would tell you maybe what your path is going to be if you’re dying to get back to the way it once was.”
Rheumatologists can expect changes in where they work, how they’re paid, increases in their workload, and new innovations in connecting with patients, he said.
“You’re going to be integrating a new style of medicine, you’re going to be digitally connected,” he explained. “All these networks are going to be working together to make you supposedly better at what you do, or maybe they’re working together to make you obsolete – and I think you better start protecting your space.”
One major area of change, telemedicine, already occurred as a result of the COVID-19 pandemic and will “begin to dominate” over the next decade, Dr. Cush said. An analysis conducted by consulting firm McKinsey & Company found telehealth usage increased 78-fold between February and April 2020 before leveling off at a 38-fold higher rate, compared with prepandemic levels. In the same analysis, rheumatology ranked third in terms of telehealth usage claims behind psychiatry and substance use disorder treatment, Dr. Cush observed, as other specialties have “fallen off quite a bit.”
“The common denominators are chronic care, cognitive care, nonprocedural care, pattern recognition, and monitoring, and this is what you do,” he said. “This is why, in many ways, for you to abandon telemedicine I think is a gigantic mistake.”
Changes to telemedicine
The most immediate change to telemedicine will come when the Biden administration officially ends the COVID-19 public health emergency in May 2023, and temporary telehealth services will be extended for approximately 5 months after the end of the public health emergency. Legislation passed by Congress will ensure some of the flexibilities in telemedicine will be extended until the end of December 2024.
Alvin Wells, MD, PhD, director of the department of rheumatology at Advocate Aurora Health in Franklin, Wisc., said he sees telemedicine as persisting even after the official COVID-19 public health emergency ends. “There’s a lot of push from the American Medical Association, from the American College of Physicians. You’re going to see people – this will not go away because [there’s] also going to be that demand.”
Despite decreased usage since April 2020, telehealth was estimated to be a $60 billion industry in 2022 and will likely increase over the next decade, Dr. Cush noted. “I question [the decline] because I think it still is a major part of your [future in] 2033.”
The number of physicians who have at least three licenses to practice in other U.S. states increased from 50,454 in 2010 to 72,752 in 2020, and that trend will continue, Dr. Wells explained. It is now becoming easier for physicians to become licensed in other states with companies like CompHealth that offer services to simplify obtaining medical licenses with states that participate in the Interstate Medical Licensure Compact.
“It’s a telemedicine easy pass,” Dr. Cush said.
Concerns in telemedicine
Commenting on the presentation, Allan Gibofsky, MD, JD, professor of medicine at Weill Cornell Medicine and codirector of the Clinic for Inflammatory Arthritis and Biologic Therapy at the Hospital for Special Surgery (HSS), both in New York, pointed out that because telemedicine is governed by U.S. states, rather than the federal government, a physician needs to be licensed in the state where the patient is located. While many states relaxed their restrictions during COVID-19, as states began tightening their restrictions later, “many physicians didn’t want to have three licenses,” he said.
“There’s an expense in getting three licenses. There’s an expense in obtaining it and maintaining it, and the reimbursement for the telemedicine visit did not reach that expectation,” Dr. Gibofsky explained. With the exception of the orthopedic surgeons at HSS who practice in New York and a satellite office in Florida, none of the surgeons at his center have obtained more than one license to practice telemedicine in other states.
“Our volume of telemedicine at HSS has remained about the same at 30%, but fewer physicians are doing it because they don’t want to maintain multiple licensures,” he said. “So don’t overlook the role of legal concerns in terms of who’s going to be allowed to do what where. Your talk was great in terms of an exuberance of what’s going to be available, but it’s not going to relieve the physician from the burden of being responsible for their use.”
Eric Ruderman, MD, professor of rheumatology at Northwestern University, Chicago, asked the presenters about the balance between seeing patients for virtual and in-person visits. “The question is what’s the sweet spot? Are there people you’re willing to see virtually forever?” he asked, noting that he has patients scheduling telemedicine visits that he hasn’t seen since before the COVID-19 pandemic.
“That’s not going to work for me. At some point, you have to lay hands on people,” he said.
Dr. Wells said his current practice is 40% virtual, and his staff converts potential no-shows into a telemedicine consultation over the phone. “My no-show rate has gone down to zero. Somebody’s scheduled for a visit, they don’t show up, my [medical assistants] get them on the phone, they put them on hold, tee up the refills. I turn them into a telephone call,” he said. “We don’t accept the no-show at all because we can do a telephone [consultation].”
In Dr. Cush’s practice, he alternates telemedicine visits with in-person visits. “If you come back for two videos in a row, you’re catching hell from me for that,” he said. Responding to how Dr. Wells incorporates telemedicine into his practice, Dr. Cush said many rheumatologists “don’t have the setups to support the care, and that’s why it’s hard to do and that’s why we’re not as great as we could be.”
“This is the way we were trained. We’re used to seeing these patients in the clinic that often. Not every single patient needs to be seen that frequently if they’re stable and doing fine,” Dr. Wells countered.
Dr. Cush and Dr. Wells reported having financial relationships with numerous pharmaceutical companies.
There was an explosion in the use of telemedicine during the COVID-19 pandemic, but usage has stabilized and varies between specialties. However, telemedicine use is still somewhat high among rheumatologists, according to speakers at the 2023 Rheumatology Winter Clinical Symposium.
Speaking in general about the future of rheumatology, Jack Cush, MD, a rheumatologist based in Dallas and executive editor of RheumNow.com, said it is up to rheumatologists to adapt to the changing winds in the specialty.
“The future is going to happen no matter what, so the question is, are you going to go along with it? Are you going to be a part of it? Are you going to be resistant to it?” Dr. Cush asked attendees. “Your recent experience with COVID would tell you maybe what your path is going to be if you’re dying to get back to the way it once was.”
Rheumatologists can expect changes in where they work, how they’re paid, increases in their workload, and new innovations in connecting with patients, he said.
“You’re going to be integrating a new style of medicine, you’re going to be digitally connected,” he explained. “All these networks are going to be working together to make you supposedly better at what you do, or maybe they’re working together to make you obsolete – and I think you better start protecting your space.”
One major area of change, telemedicine, already occurred as a result of the COVID-19 pandemic and will “begin to dominate” over the next decade, Dr. Cush said. An analysis conducted by consulting firm McKinsey & Company found telehealth usage increased 78-fold between February and April 2020 before leveling off at a 38-fold higher rate, compared with prepandemic levels. In the same analysis, rheumatology ranked third in terms of telehealth usage claims behind psychiatry and substance use disorder treatment, Dr. Cush observed, as other specialties have “fallen off quite a bit.”
“The common denominators are chronic care, cognitive care, nonprocedural care, pattern recognition, and monitoring, and this is what you do,” he said. “This is why, in many ways, for you to abandon telemedicine I think is a gigantic mistake.”
Changes to telemedicine
The most immediate change to telemedicine will come when the Biden administration officially ends the COVID-19 public health emergency in May 2023, and temporary telehealth services will be extended for approximately 5 months after the end of the public health emergency. Legislation passed by Congress will ensure some of the flexibilities in telemedicine will be extended until the end of December 2024.
Alvin Wells, MD, PhD, director of the department of rheumatology at Advocate Aurora Health in Franklin, Wisc., said he sees telemedicine as persisting even after the official COVID-19 public health emergency ends. “There’s a lot of push from the American Medical Association, from the American College of Physicians. You’re going to see people – this will not go away because [there’s] also going to be that demand.”
Despite decreased usage since April 2020, telehealth was estimated to be a $60 billion industry in 2022 and will likely increase over the next decade, Dr. Cush noted. “I question [the decline] because I think it still is a major part of your [future in] 2033.”
The number of physicians who have at least three licenses to practice in other U.S. states increased from 50,454 in 2010 to 72,752 in 2020, and that trend will continue, Dr. Wells explained. It is now becoming easier for physicians to become licensed in other states with companies like CompHealth that offer services to simplify obtaining medical licenses with states that participate in the Interstate Medical Licensure Compact.
“It’s a telemedicine easy pass,” Dr. Cush said.
Concerns in telemedicine
Commenting on the presentation, Allan Gibofsky, MD, JD, professor of medicine at Weill Cornell Medicine and codirector of the Clinic for Inflammatory Arthritis and Biologic Therapy at the Hospital for Special Surgery (HSS), both in New York, pointed out that because telemedicine is governed by U.S. states, rather than the federal government, a physician needs to be licensed in the state where the patient is located. While many states relaxed their restrictions during COVID-19, as states began tightening their restrictions later, “many physicians didn’t want to have three licenses,” he said.
“There’s an expense in getting three licenses. There’s an expense in obtaining it and maintaining it, and the reimbursement for the telemedicine visit did not reach that expectation,” Dr. Gibofsky explained. With the exception of the orthopedic surgeons at HSS who practice in New York and a satellite office in Florida, none of the surgeons at his center have obtained more than one license to practice telemedicine in other states.
“Our volume of telemedicine at HSS has remained about the same at 30%, but fewer physicians are doing it because they don’t want to maintain multiple licensures,” he said. “So don’t overlook the role of legal concerns in terms of who’s going to be allowed to do what where. Your talk was great in terms of an exuberance of what’s going to be available, but it’s not going to relieve the physician from the burden of being responsible for their use.”
Eric Ruderman, MD, professor of rheumatology at Northwestern University, Chicago, asked the presenters about the balance between seeing patients for virtual and in-person visits. “The question is what’s the sweet spot? Are there people you’re willing to see virtually forever?” he asked, noting that he has patients scheduling telemedicine visits that he hasn’t seen since before the COVID-19 pandemic.
“That’s not going to work for me. At some point, you have to lay hands on people,” he said.
Dr. Wells said his current practice is 40% virtual, and his staff converts potential no-shows into a telemedicine consultation over the phone. “My no-show rate has gone down to zero. Somebody’s scheduled for a visit, they don’t show up, my [medical assistants] get them on the phone, they put them on hold, tee up the refills. I turn them into a telephone call,” he said. “We don’t accept the no-show at all because we can do a telephone [consultation].”
In Dr. Cush’s practice, he alternates telemedicine visits with in-person visits. “If you come back for two videos in a row, you’re catching hell from me for that,” he said. Responding to how Dr. Wells incorporates telemedicine into his practice, Dr. Cush said many rheumatologists “don’t have the setups to support the care, and that’s why it’s hard to do and that’s why we’re not as great as we could be.”
“This is the way we were trained. We’re used to seeing these patients in the clinic that often. Not every single patient needs to be seen that frequently if they’re stable and doing fine,” Dr. Wells countered.
Dr. Cush and Dr. Wells reported having financial relationships with numerous pharmaceutical companies.
FROM RWCS 2023
‘Only a sociopath could work for a large health system,’ doc says sardonically
A frustrated physician recently voiced some strong words in Medscape’s US Physician Burnout & Depression Report: “Only a sociopath could work for a large health system and not be burned out. Anyone who cares about patients is doomed to burnout.”
Medscape’s report showed that 53% of physicians feel burned out by job requirements; 65% say that burnout has impacted their relationships, and other statistics say that physicians are leaving clinical medicine because of all this pressure.
What is it about being employed by large organizations that can be so negative? In another study, MEMO – Minimizing Error, Maximizing Outcomes – researchers at the University of Wisconsin surveyed more than 400 doctors to learn about how their working environments corresponded with medical errors. More than half of the physicians reported time pressures when conducting physical examinations. Nearly a third felt they needed at least 50% more time than was allotted for this patient care function, and nearly a quarter said they needed at least 50% more time for follow-up appointments.
Some have asked: Can anyone, then, thrive in today’s health care environment and avoid burnout?
Although the frustrated physician noted above may sardonically say that a doctor needs to be sociopathic to enjoy it – lacking in feelings for others – “It’s a very small number of doctors who get in it for the wrong reasons and therefore care about their own benefit and not their patients,” said psychiatrist Wendy Dean, MD, CEO and cofounder of Moral Injury of Healthcare, a nonprofit organization addressing workforce distress in health care. “Those are the outliers.”
The vast majority of physicians do care about their patients – deeply, said Dr. Dean. They struggle under the weight of the health care system and yet must find ways to get through. Today, thriving in an imperfect system requires honing new skills, asking for help when needed, and pushing for systemic and cultural change.
“We’ve been assessing and trying to address burnout for half a century,” said Dr. Dean. “Despite all the good intentions, and people dedicating their entire careers to solving the issue, we’ve barely made a dent.”
With the advent of new technological requirements on the job and more demands from increasingly larger health care organizations, the risk for burnout is higher than ever before. “There’s an increased burden of regulatory-mandated and cumbersome administrative workload per patient,” said Shomron Ben-Horin, MD, cofounder of Evinature. “Often the computer/paperwork before and after a procedure is much longer than the procedure itself.”
Meeting insurance requirements is increasingly cumbersome, too, and preauthorizations and debates with payers over medical approval may put physicians frustratingly in the middle.
“This increases the psychological burden for physicians who may feel responsible for wrongdoing no matter which option they deem better,” Dr. Ben-Horin said. “Add in physician accessibility around the clock via mobile phones, emails, and apps, and you end up on call even if you’re not officially on call.”
Why some physicians suffer more
Some physicians are more likely to suffer burnout than others, said Jessi Gold, MD, assistant professor in the department of psychiatry at Washington University in St. Louis. “The self-valuation concept comes into play here,” she said. “If you make a mistake, do you blame yourself or see it as a growth opportunity? If it’s the former, you’re more likely to burn out.”
Dr. Ben-Horin added that the most patient-centric doctors are the ones who struggle most. “These are the doctors we’d all love to have as a patient,” he said. “But they are burdened by the extra tasks of the job, and they are the most stressed by the environment.”
So too are those physicians who never master compartmentalizing their feelings and emotions. “We learn in training to compartmentalize our emotions,” said Dr. Dean. “You can’t allow yourself to get emotional while performing chest compressions on an 18-year-old kid. So you shut it all away; otherwise, you might lose the patient.”
This turn-off switch becomes automatic, but it also comes at a cost. “When doctors were interviewed about [Buffalo Bills player] Damar Hamlin going into cardiac arrest on the football field, they talked about how a life-and-death situation is so common that they have to put the emotions away, work on the patient, and move onto the next,” said Dr. Dean. “The next patient needs you just as much. We must lock away our feelings and manage the situation.”
Dr. Gold explained that burying feelings, however, is a symptom of burnout. “We have to remove ourselves from the situation to protect ourselves,” she said. “We can’t cry in these situations, but we can’t bury our feelings either.”
Instead, Dr. Gold suggested, a good medium may exist. “You may not be able to address them in the moment, but you should sometime after,” she said.
This is just a starting point on how to remain a dedicated, caring physician without burning out. “The system is pretty broken, and to survive it first means wanting to survive it,” Dr. Gold said. “There’s a lot of focus on resiliency and lack thereof if a physician expresses burnout, but that’s a false notion. Doctors are a resilient bunch but even they get burned out.”
Change for the better must come from several places. One is asking for help, something that can be hard for a group conditioned to keeping a stiff upper lip. “Just because your peers might look healthy (emotionally) doesn’t mean they are,” said Dr. Gold. “We’ve normalized this culture of burying feelings, but that doesn’t mean it’s right.”
Dr. Ben-Horin also advocates diversifying your work. This might include engaging in research and academics, for instance. “This not only makes you a better broad-perspective doctor but allows you to psychologically switch gears on research days,” he said.
The biggest place to make change, however, is within the health care system culture itself. The AMA created a series of recommendations to address burnout at the resident and fellow level, a good starting point to carry through into staff work. The steps include creating a well-being framework, gathering a team to support a well-being program, developing the program in a way to foster fun and connectivity among the staff, fostering individual well-being that addresses emotional and physical well-being, and confronting burnout and creating a sustainable culture of well-being.
On a personal level, it’s essential that physicians keep close tabs on themselves and peers. “Understand the signs and symptoms of burnout by taking stock of where you are emotionally,” said Dr. Gold. “Have a place and time at the end of a hard day to reflect or find a ritual that helps you and stay with it.”
You might also reach out to a therapist or a peer when you’re struggling. Having honest conversations with peers can go a long way. “Find a confidant that allows you to be vulnerable,” Dr. Gold recommended. “Acknowledge that this is hard and that you might need help taking care of yourself. The system needs to change, but we can also learn to survive in the meantime. You don’t have to be a sociopath to make it.”
A version of this article originally appeared on Medscape.com.
A frustrated physician recently voiced some strong words in Medscape’s US Physician Burnout & Depression Report: “Only a sociopath could work for a large health system and not be burned out. Anyone who cares about patients is doomed to burnout.”
Medscape’s report showed that 53% of physicians feel burned out by job requirements; 65% say that burnout has impacted their relationships, and other statistics say that physicians are leaving clinical medicine because of all this pressure.
What is it about being employed by large organizations that can be so negative? In another study, MEMO – Minimizing Error, Maximizing Outcomes – researchers at the University of Wisconsin surveyed more than 400 doctors to learn about how their working environments corresponded with medical errors. More than half of the physicians reported time pressures when conducting physical examinations. Nearly a third felt they needed at least 50% more time than was allotted for this patient care function, and nearly a quarter said they needed at least 50% more time for follow-up appointments.
Some have asked: Can anyone, then, thrive in today’s health care environment and avoid burnout?
Although the frustrated physician noted above may sardonically say that a doctor needs to be sociopathic to enjoy it – lacking in feelings for others – “It’s a very small number of doctors who get in it for the wrong reasons and therefore care about their own benefit and not their patients,” said psychiatrist Wendy Dean, MD, CEO and cofounder of Moral Injury of Healthcare, a nonprofit organization addressing workforce distress in health care. “Those are the outliers.”
The vast majority of physicians do care about their patients – deeply, said Dr. Dean. They struggle under the weight of the health care system and yet must find ways to get through. Today, thriving in an imperfect system requires honing new skills, asking for help when needed, and pushing for systemic and cultural change.
“We’ve been assessing and trying to address burnout for half a century,” said Dr. Dean. “Despite all the good intentions, and people dedicating their entire careers to solving the issue, we’ve barely made a dent.”
With the advent of new technological requirements on the job and more demands from increasingly larger health care organizations, the risk for burnout is higher than ever before. “There’s an increased burden of regulatory-mandated and cumbersome administrative workload per patient,” said Shomron Ben-Horin, MD, cofounder of Evinature. “Often the computer/paperwork before and after a procedure is much longer than the procedure itself.”
Meeting insurance requirements is increasingly cumbersome, too, and preauthorizations and debates with payers over medical approval may put physicians frustratingly in the middle.
“This increases the psychological burden for physicians who may feel responsible for wrongdoing no matter which option they deem better,” Dr. Ben-Horin said. “Add in physician accessibility around the clock via mobile phones, emails, and apps, and you end up on call even if you’re not officially on call.”
Why some physicians suffer more
Some physicians are more likely to suffer burnout than others, said Jessi Gold, MD, assistant professor in the department of psychiatry at Washington University in St. Louis. “The self-valuation concept comes into play here,” she said. “If you make a mistake, do you blame yourself or see it as a growth opportunity? If it’s the former, you’re more likely to burn out.”
Dr. Ben-Horin added that the most patient-centric doctors are the ones who struggle most. “These are the doctors we’d all love to have as a patient,” he said. “But they are burdened by the extra tasks of the job, and they are the most stressed by the environment.”
So too are those physicians who never master compartmentalizing their feelings and emotions. “We learn in training to compartmentalize our emotions,” said Dr. Dean. “You can’t allow yourself to get emotional while performing chest compressions on an 18-year-old kid. So you shut it all away; otherwise, you might lose the patient.”
This turn-off switch becomes automatic, but it also comes at a cost. “When doctors were interviewed about [Buffalo Bills player] Damar Hamlin going into cardiac arrest on the football field, they talked about how a life-and-death situation is so common that they have to put the emotions away, work on the patient, and move onto the next,” said Dr. Dean. “The next patient needs you just as much. We must lock away our feelings and manage the situation.”
Dr. Gold explained that burying feelings, however, is a symptom of burnout. “We have to remove ourselves from the situation to protect ourselves,” she said. “We can’t cry in these situations, but we can’t bury our feelings either.”
Instead, Dr. Gold suggested, a good medium may exist. “You may not be able to address them in the moment, but you should sometime after,” she said.
This is just a starting point on how to remain a dedicated, caring physician without burning out. “The system is pretty broken, and to survive it first means wanting to survive it,” Dr. Gold said. “There’s a lot of focus on resiliency and lack thereof if a physician expresses burnout, but that’s a false notion. Doctors are a resilient bunch but even they get burned out.”
Change for the better must come from several places. One is asking for help, something that can be hard for a group conditioned to keeping a stiff upper lip. “Just because your peers might look healthy (emotionally) doesn’t mean they are,” said Dr. Gold. “We’ve normalized this culture of burying feelings, but that doesn’t mean it’s right.”
Dr. Ben-Horin also advocates diversifying your work. This might include engaging in research and academics, for instance. “This not only makes you a better broad-perspective doctor but allows you to psychologically switch gears on research days,” he said.
The biggest place to make change, however, is within the health care system culture itself. The AMA created a series of recommendations to address burnout at the resident and fellow level, a good starting point to carry through into staff work. The steps include creating a well-being framework, gathering a team to support a well-being program, developing the program in a way to foster fun and connectivity among the staff, fostering individual well-being that addresses emotional and physical well-being, and confronting burnout and creating a sustainable culture of well-being.
On a personal level, it’s essential that physicians keep close tabs on themselves and peers. “Understand the signs and symptoms of burnout by taking stock of where you are emotionally,” said Dr. Gold. “Have a place and time at the end of a hard day to reflect or find a ritual that helps you and stay with it.”
You might also reach out to a therapist or a peer when you’re struggling. Having honest conversations with peers can go a long way. “Find a confidant that allows you to be vulnerable,” Dr. Gold recommended. “Acknowledge that this is hard and that you might need help taking care of yourself. The system needs to change, but we can also learn to survive in the meantime. You don’t have to be a sociopath to make it.”
A version of this article originally appeared on Medscape.com.
A frustrated physician recently voiced some strong words in Medscape’s US Physician Burnout & Depression Report: “Only a sociopath could work for a large health system and not be burned out. Anyone who cares about patients is doomed to burnout.”
Medscape’s report showed that 53% of physicians feel burned out by job requirements; 65% say that burnout has impacted their relationships, and other statistics say that physicians are leaving clinical medicine because of all this pressure.
What is it about being employed by large organizations that can be so negative? In another study, MEMO – Minimizing Error, Maximizing Outcomes – researchers at the University of Wisconsin surveyed more than 400 doctors to learn about how their working environments corresponded with medical errors. More than half of the physicians reported time pressures when conducting physical examinations. Nearly a third felt they needed at least 50% more time than was allotted for this patient care function, and nearly a quarter said they needed at least 50% more time for follow-up appointments.
Some have asked: Can anyone, then, thrive in today’s health care environment and avoid burnout?
Although the frustrated physician noted above may sardonically say that a doctor needs to be sociopathic to enjoy it – lacking in feelings for others – “It’s a very small number of doctors who get in it for the wrong reasons and therefore care about their own benefit and not their patients,” said psychiatrist Wendy Dean, MD, CEO and cofounder of Moral Injury of Healthcare, a nonprofit organization addressing workforce distress in health care. “Those are the outliers.”
The vast majority of physicians do care about their patients – deeply, said Dr. Dean. They struggle under the weight of the health care system and yet must find ways to get through. Today, thriving in an imperfect system requires honing new skills, asking for help when needed, and pushing for systemic and cultural change.
“We’ve been assessing and trying to address burnout for half a century,” said Dr. Dean. “Despite all the good intentions, and people dedicating their entire careers to solving the issue, we’ve barely made a dent.”
With the advent of new technological requirements on the job and more demands from increasingly larger health care organizations, the risk for burnout is higher than ever before. “There’s an increased burden of regulatory-mandated and cumbersome administrative workload per patient,” said Shomron Ben-Horin, MD, cofounder of Evinature. “Often the computer/paperwork before and after a procedure is much longer than the procedure itself.”
Meeting insurance requirements is increasingly cumbersome, too, and preauthorizations and debates with payers over medical approval may put physicians frustratingly in the middle.
“This increases the psychological burden for physicians who may feel responsible for wrongdoing no matter which option they deem better,” Dr. Ben-Horin said. “Add in physician accessibility around the clock via mobile phones, emails, and apps, and you end up on call even if you’re not officially on call.”
Why some physicians suffer more
Some physicians are more likely to suffer burnout than others, said Jessi Gold, MD, assistant professor in the department of psychiatry at Washington University in St. Louis. “The self-valuation concept comes into play here,” she said. “If you make a mistake, do you blame yourself or see it as a growth opportunity? If it’s the former, you’re more likely to burn out.”
Dr. Ben-Horin added that the most patient-centric doctors are the ones who struggle most. “These are the doctors we’d all love to have as a patient,” he said. “But they are burdened by the extra tasks of the job, and they are the most stressed by the environment.”
So too are those physicians who never master compartmentalizing their feelings and emotions. “We learn in training to compartmentalize our emotions,” said Dr. Dean. “You can’t allow yourself to get emotional while performing chest compressions on an 18-year-old kid. So you shut it all away; otherwise, you might lose the patient.”
This turn-off switch becomes automatic, but it also comes at a cost. “When doctors were interviewed about [Buffalo Bills player] Damar Hamlin going into cardiac arrest on the football field, they talked about how a life-and-death situation is so common that they have to put the emotions away, work on the patient, and move onto the next,” said Dr. Dean. “The next patient needs you just as much. We must lock away our feelings and manage the situation.”
Dr. Gold explained that burying feelings, however, is a symptom of burnout. “We have to remove ourselves from the situation to protect ourselves,” she said. “We can’t cry in these situations, but we can’t bury our feelings either.”
Instead, Dr. Gold suggested, a good medium may exist. “You may not be able to address them in the moment, but you should sometime after,” she said.
This is just a starting point on how to remain a dedicated, caring physician without burning out. “The system is pretty broken, and to survive it first means wanting to survive it,” Dr. Gold said. “There’s a lot of focus on resiliency and lack thereof if a physician expresses burnout, but that’s a false notion. Doctors are a resilient bunch but even they get burned out.”
Change for the better must come from several places. One is asking for help, something that can be hard for a group conditioned to keeping a stiff upper lip. “Just because your peers might look healthy (emotionally) doesn’t mean they are,” said Dr. Gold. “We’ve normalized this culture of burying feelings, but that doesn’t mean it’s right.”
Dr. Ben-Horin also advocates diversifying your work. This might include engaging in research and academics, for instance. “This not only makes you a better broad-perspective doctor but allows you to psychologically switch gears on research days,” he said.
The biggest place to make change, however, is within the health care system culture itself. The AMA created a series of recommendations to address burnout at the resident and fellow level, a good starting point to carry through into staff work. The steps include creating a well-being framework, gathering a team to support a well-being program, developing the program in a way to foster fun and connectivity among the staff, fostering individual well-being that addresses emotional and physical well-being, and confronting burnout and creating a sustainable culture of well-being.
On a personal level, it’s essential that physicians keep close tabs on themselves and peers. “Understand the signs and symptoms of burnout by taking stock of where you are emotionally,” said Dr. Gold. “Have a place and time at the end of a hard day to reflect or find a ritual that helps you and stay with it.”
You might also reach out to a therapist or a peer when you’re struggling. Having honest conversations with peers can go a long way. “Find a confidant that allows you to be vulnerable,” Dr. Gold recommended. “Acknowledge that this is hard and that you might need help taking care of yourself. The system needs to change, but we can also learn to survive in the meantime. You don’t have to be a sociopath to make it.”
A version of this article originally appeared on Medscape.com.
Not always implemented or enforced: Harassment policies at work
Many companies, government agencies, and organizations have implemented policies and procedures to shield employees from sexual and other forms of harassment. The U.S. Department of Health & Human Services and the American Medical Association are just two examples.
Employers can tap a rich lode of guidance and resources to craft these antiharassment policies. The National Institutes of Health’s resource page is a good site for hospitals to check out.
But how effective have official policies proved in deterring harassment in medical workplaces? After all, in a study by the American Association of Medical Colleges, 34% of female faculty said they had experienced sexual harassment irrespective of such policies. And in a recent Medscape survey of more than 3,000 physicians, 27% reported that they had either witnessed or been subjected to sexual harassment or misconduct at work during the past 4 years.
When policies are absent or unenforced
She believes employer rules and policies generally are helpful in establishing who fields harassment complaints and in creating at least some accountability.
On the other hand, policies that don’t recognize anonymous complaints effectively discourage harassment victims from coming forward, Dr. Rohr-Kirchgraber argues. Even those policies that do allow anonymous complaints may have limitations.
For example, the NIH policy on reporting harassment acknowledges that “officials must follow up on all allegations of harassment and cannot guarantee that your identity will not become apparent during the process. Please note that if you remain anonymous, key details about the allegation or concern [may] be omitted. This will limit the NIH’s ability to conduct an inquiry and take corrective action as warranted.”
Risks in pressing a harassment case
A complainant whose name becomes public risks getting a reputation as a problem employee or suffering workplace retaliation, according to Dr. Rohr-Kirchgraber. She recalls a colleague who was on a clinical education track until she lodged a harassment complaint. Abruptly, she was told she was needed on a service with fewer teaching opportunities.
With such risks in mind, respondents to the Medscape survey advised employees in medical workplaces to familiarize themselves with policies and procedures before pressing a case.
“Document everything,” an ophthalmologist urged, including time, place, offender, and witnesses. Present that information to your supervisor, and if nothing is done, hire a lawyer, a gastroenterologist suggested.
But taking the situation to the Equal Employment Opportunity Commission can be complicated, Roberta Gebhard, DO, past AMWA president and founder of its Gender Equity Task Force, told this news organization.
“They talk to the employer and get the employer’s side of the story and eventually render a decision about whether you have a case you can put through and file a lawsuit,” she said. “I don’t know of any other situation in which you need ‘permission’ to file a lawsuit.”
Nevertheless, an attorney can be helpful with cases, and when someone is terminated, a lawyer can possibly have it overturned or converted to a resignation, Dr. Gebhard said.
“And always have a lawyer review your contract before you take the job,” she advised. The lawyer might adjust the contract’s verbiage in ways that can protect one down the road in the event of a potential termination. “It’s money very well spent.”
More education needed
Dr. Rohr-Kirchgraber said that protection against harassment goes beyond the employer’s policies and procedures. Building an overall consciousness of what harassment is should begin with employee onboarding, she said.
“The harasser may not even recognize that what they’re doing or saying is a form of harassment, so we need better education,” Dr. Rohr-Kirchgraber emphasized.
A version of this article originally appeared on Medscape.com.
Many companies, government agencies, and organizations have implemented policies and procedures to shield employees from sexual and other forms of harassment. The U.S. Department of Health & Human Services and the American Medical Association are just two examples.
Employers can tap a rich lode of guidance and resources to craft these antiharassment policies. The National Institutes of Health’s resource page is a good site for hospitals to check out.
But how effective have official policies proved in deterring harassment in medical workplaces? After all, in a study by the American Association of Medical Colleges, 34% of female faculty said they had experienced sexual harassment irrespective of such policies. And in a recent Medscape survey of more than 3,000 physicians, 27% reported that they had either witnessed or been subjected to sexual harassment or misconduct at work during the past 4 years.
When policies are absent or unenforced
She believes employer rules and policies generally are helpful in establishing who fields harassment complaints and in creating at least some accountability.
On the other hand, policies that don’t recognize anonymous complaints effectively discourage harassment victims from coming forward, Dr. Rohr-Kirchgraber argues. Even those policies that do allow anonymous complaints may have limitations.
For example, the NIH policy on reporting harassment acknowledges that “officials must follow up on all allegations of harassment and cannot guarantee that your identity will not become apparent during the process. Please note that if you remain anonymous, key details about the allegation or concern [may] be omitted. This will limit the NIH’s ability to conduct an inquiry and take corrective action as warranted.”
Risks in pressing a harassment case
A complainant whose name becomes public risks getting a reputation as a problem employee or suffering workplace retaliation, according to Dr. Rohr-Kirchgraber. She recalls a colleague who was on a clinical education track until she lodged a harassment complaint. Abruptly, she was told she was needed on a service with fewer teaching opportunities.
With such risks in mind, respondents to the Medscape survey advised employees in medical workplaces to familiarize themselves with policies and procedures before pressing a case.
“Document everything,” an ophthalmologist urged, including time, place, offender, and witnesses. Present that information to your supervisor, and if nothing is done, hire a lawyer, a gastroenterologist suggested.
But taking the situation to the Equal Employment Opportunity Commission can be complicated, Roberta Gebhard, DO, past AMWA president and founder of its Gender Equity Task Force, told this news organization.
“They talk to the employer and get the employer’s side of the story and eventually render a decision about whether you have a case you can put through and file a lawsuit,” she said. “I don’t know of any other situation in which you need ‘permission’ to file a lawsuit.”
Nevertheless, an attorney can be helpful with cases, and when someone is terminated, a lawyer can possibly have it overturned or converted to a resignation, Dr. Gebhard said.
“And always have a lawyer review your contract before you take the job,” she advised. The lawyer might adjust the contract’s verbiage in ways that can protect one down the road in the event of a potential termination. “It’s money very well spent.”
More education needed
Dr. Rohr-Kirchgraber said that protection against harassment goes beyond the employer’s policies and procedures. Building an overall consciousness of what harassment is should begin with employee onboarding, she said.
“The harasser may not even recognize that what they’re doing or saying is a form of harassment, so we need better education,” Dr. Rohr-Kirchgraber emphasized.
A version of this article originally appeared on Medscape.com.
Many companies, government agencies, and organizations have implemented policies and procedures to shield employees from sexual and other forms of harassment. The U.S. Department of Health & Human Services and the American Medical Association are just two examples.
Employers can tap a rich lode of guidance and resources to craft these antiharassment policies. The National Institutes of Health’s resource page is a good site for hospitals to check out.
But how effective have official policies proved in deterring harassment in medical workplaces? After all, in a study by the American Association of Medical Colleges, 34% of female faculty said they had experienced sexual harassment irrespective of such policies. And in a recent Medscape survey of more than 3,000 physicians, 27% reported that they had either witnessed or been subjected to sexual harassment or misconduct at work during the past 4 years.
When policies are absent or unenforced
She believes employer rules and policies generally are helpful in establishing who fields harassment complaints and in creating at least some accountability.
On the other hand, policies that don’t recognize anonymous complaints effectively discourage harassment victims from coming forward, Dr. Rohr-Kirchgraber argues. Even those policies that do allow anonymous complaints may have limitations.
For example, the NIH policy on reporting harassment acknowledges that “officials must follow up on all allegations of harassment and cannot guarantee that your identity will not become apparent during the process. Please note that if you remain anonymous, key details about the allegation or concern [may] be omitted. This will limit the NIH’s ability to conduct an inquiry and take corrective action as warranted.”
Risks in pressing a harassment case
A complainant whose name becomes public risks getting a reputation as a problem employee or suffering workplace retaliation, according to Dr. Rohr-Kirchgraber. She recalls a colleague who was on a clinical education track until she lodged a harassment complaint. Abruptly, she was told she was needed on a service with fewer teaching opportunities.
With such risks in mind, respondents to the Medscape survey advised employees in medical workplaces to familiarize themselves with policies and procedures before pressing a case.
“Document everything,” an ophthalmologist urged, including time, place, offender, and witnesses. Present that information to your supervisor, and if nothing is done, hire a lawyer, a gastroenterologist suggested.
But taking the situation to the Equal Employment Opportunity Commission can be complicated, Roberta Gebhard, DO, past AMWA president and founder of its Gender Equity Task Force, told this news organization.
“They talk to the employer and get the employer’s side of the story and eventually render a decision about whether you have a case you can put through and file a lawsuit,” she said. “I don’t know of any other situation in which you need ‘permission’ to file a lawsuit.”
Nevertheless, an attorney can be helpful with cases, and when someone is terminated, a lawyer can possibly have it overturned or converted to a resignation, Dr. Gebhard said.
“And always have a lawyer review your contract before you take the job,” she advised. The lawyer might adjust the contract’s verbiage in ways that can protect one down the road in the event of a potential termination. “It’s money very well spent.”
More education needed
Dr. Rohr-Kirchgraber said that protection against harassment goes beyond the employer’s policies and procedures. Building an overall consciousness of what harassment is should begin with employee onboarding, she said.
“The harasser may not even recognize that what they’re doing or saying is a form of harassment, so we need better education,” Dr. Rohr-Kirchgraber emphasized.
A version of this article originally appeared on Medscape.com.
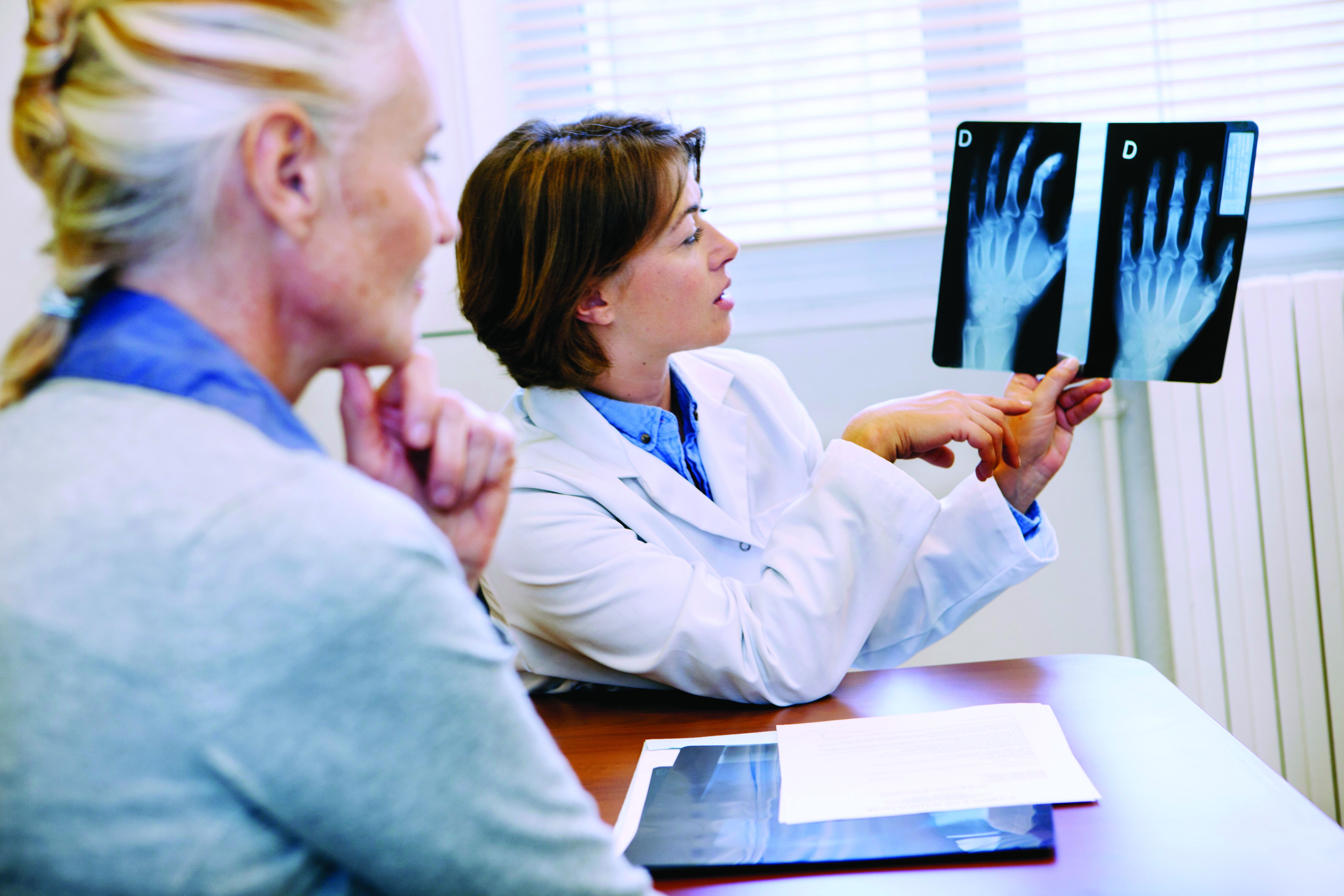
Are repeat radiographs necessary in rheumatoid and psoriatic arthritis?
Follow-up radiographs after an initial baseline reading in patients with rheumatoid arthritis or psoriatic arthritis may still show radiographic progression despite treatment with current therapies, but it’s unclear if they will affect treatment decisions between patients and doctors that may take place regardless of the radiographic information, according to arguments made for and against their usefulness in a point-counterpoint session at the 2023 Rheumatology Winter Clinical Symposium.
Alvin Wells, MD, PhD, director of the department of rheumatology at Advocate Aurora Health in Franklin, Wisc., said that x-rays “reflect the history of joint pathology” and can get worse over time, correlating with disease activity and severity.
While RA does not yet have the “holy grail” of complete or true remission, Dr. Wells argued, the combination of clinical remission, laboratory remission, and imaging remission gets patients with RA close to the ideal when measured over time. “You need to continue to monitor these patients as you follow them along,” he said.
The BARFOT study, which evaluated 1,938 patients with early RA in two cohorts during 1992-1999 and again between 2000 and 2006, showed that more active treatments in the 2000s did not result in improvements in Health Assessment Questionnaire (HAQ) and pain scores, compared with patients treated in the 1990s. “You can see in some of those patients those scores do increase, and that even despite aggressive therapies that we had in 2006, you can still see some of those patients still have progression of the disease,” Dr. Wells explained. “How did they know? Because they looked.”
He also cited a study from researchers at the Mayo Clinic who examined 586 patients with RA that showed a higher prevalence of functional disability in patients with RA who also had radiographic changes, compared with patients without RA. “Radiographic changes correlate with disease severity and functional disability as well,” Dr. Wells said.
Just as prostate-specific antigen levels are used in prostate cancer screening and hemoglobin A1c is measured in diabetes management, radiographs should be used to track progression of disease in RA and PsA, Dr. Wells argued. “[I]f you don’t know, you can’t treat,” he said.
Some patients near remission may have radiographic progression even though disease activity measurements such as C-reactive protein (CRP) values do not show presence of active disease. In a study analyzing 1,184 patients with RA in the ASPIRE, ERA, Leflunomide, PREMIER and TEMPO trials, swollen joint count (SJC) was a better predictor of radiographic progression than CRP in patients near remission.
“[E]ven where you don’t see smoke, there still could be fire,” Dr. Wells said. “Some of these patients still progress and these are outliers, and the way they saw that [was] because they followed those patients along. If you don’t look, you don’t know.”
Radiographic progression can also be seen among nonswollen joints in patients with RA and PsA. In a study of 1,207 joints in 55 patients with RA and 352 joints in 18 patients with PsA, researchers in Austria found tenderness in nonswollen joints was associated with radiographic progression.
Despite having effective treatments in RA and PsA, “none of our therapies show that they’re able to prevent progression,” Dr. Wells said.
When it comes to hitting the treatment target in RA, some rheumatologists may think they can accomplish it without use of repeated radiographs. “I have a different perspective on that – that you really do indeed need to do the x-rays today and follow those x-rays along, especially if it’s going to change your treatment paradigm and what your treatment decision would be for the patient,” he said.
Counterpoint: Repeat radiographs aren’t helpful
Almost all rheumatologists would likely order an initial radiograph for their patients with RA or PsA, Roy M. Fleischmann, MD, clinical professor of medicine at the University of Texas and codirector of the Metroplex Clinical Research Center, both in Dallas, said in his presentation.
“If you see erosions when you start, chances are you’re going to be much more aggressive,” Dr. Fleischmann said. “So it is justification for early, more aggressive treatment of disease.”
In recent decades, radiographic progression in RA has decreased as more effective antirheumatic treatments have come into use, Dr. Fleischmann argued.
“We had x-ray progression in virtually everybody, and it was consistent no matter what we treated with, which was gold or penicillamine or any of the NSAIDs or sulfasalazine,” he said. “With methotrexate ... about 60% of patients actually have no x-ray progression, and that was a major change, and that’s one of the reasons why methotrexate has become the keystone of therapy. But even with methotrexate, [we] still had many patients who progressed.”
After the introduction of tumor necrosis factor inhibitors and other mechanisms in the late 1990s, “all of a sudden, you don’t see x-ray progression – mean x-ray progression – in a group of patients,” he noted.
Many rheumatologists now use a treat-to-target strategy, and if the patient achieves true clinical remission or sustained very low disease activity as measured by Boolean remission, Simple Disease Activity Index, or Clinical Disease Activity Index, they have “very little chance of radiographic progression and functional decline,” he said.
“If a patient doesn’t achieve remission or very low disease activity, obtaining a radiograph doesn’t change what you do because the patient’s not where they want to be, where you want them to be; you’re going to make a change anyway,” Dr. Fleischmann explained. “The radiograph isn’t going to help you do that.”
If a patient is in sustained remission but a radiograph is ordered and shows disease progression, he questioned what the rheumatologist would do in that situation.
“Now the patient’s in, let’s say, a Boolean remission. They have no tender joints. They have no swollen joints ... their pain assessment is zero, their CRP is zero, and they do have some x-ray progression. Where are you going to change?” Dr. Fleischmann asked. “There’s no data that anything else would work. I don’t know what you would do. So, in conclusion, I would say you really don’t need to repeat an x-ray.”
AI reading x-rays?
Commenting on the point-counterpoint session, Arthur Kavanaugh, MD, professor of medicine at the University of California, San Diego, and director of RWCS, asked Dr. Fleischmann and Dr. Wells how they address the issue of how many radiologists seem to be unfamiliar with reading hand radiographs and RA progression.
Dr. Fleischmann said he was trained in how to read hand radiographs in medical school, but that training no longer appears to be occurring. “If you have a good bone radiologist, of which there are not a lot, you’re great. But if you don’t have a really good bone radiologist, it’s difficult,” he said.
Dr. Kavanaugh alluded to the advancement of artificial intelligence (AI) in radiology and posed the question of how both rheumatologists felt about AI reading and interpreting their radiographs. “If you could reliably submit x-rays and they would say what the Sharp score was and where the differences were, would that change anything?” he asked.
“I think having artificial intelligence read the x-ray or an MRI is really, really good. It’ll be better than the radiologists,” Dr. Fleischmann responded. “But I don’t think that you really need to repeat the x-ray. I mean, I really don’t think you need to repeat it. You need to treat the patient.”
Dr. Wells reported having financial relationships with numerous pharmaceutical companies. Dr. Fleischmann reported no relevant financial relationships.
Follow-up radiographs after an initial baseline reading in patients with rheumatoid arthritis or psoriatic arthritis may still show radiographic progression despite treatment with current therapies, but it’s unclear if they will affect treatment decisions between patients and doctors that may take place regardless of the radiographic information, according to arguments made for and against their usefulness in a point-counterpoint session at the 2023 Rheumatology Winter Clinical Symposium.
Alvin Wells, MD, PhD, director of the department of rheumatology at Advocate Aurora Health in Franklin, Wisc., said that x-rays “reflect the history of joint pathology” and can get worse over time, correlating with disease activity and severity.
While RA does not yet have the “holy grail” of complete or true remission, Dr. Wells argued, the combination of clinical remission, laboratory remission, and imaging remission gets patients with RA close to the ideal when measured over time. “You need to continue to monitor these patients as you follow them along,” he said.
The BARFOT study, which evaluated 1,938 patients with early RA in two cohorts during 1992-1999 and again between 2000 and 2006, showed that more active treatments in the 2000s did not result in improvements in Health Assessment Questionnaire (HAQ) and pain scores, compared with patients treated in the 1990s. “You can see in some of those patients those scores do increase, and that even despite aggressive therapies that we had in 2006, you can still see some of those patients still have progression of the disease,” Dr. Wells explained. “How did they know? Because they looked.”
He also cited a study from researchers at the Mayo Clinic who examined 586 patients with RA that showed a higher prevalence of functional disability in patients with RA who also had radiographic changes, compared with patients without RA. “Radiographic changes correlate with disease severity and functional disability as well,” Dr. Wells said.
Just as prostate-specific antigen levels are used in prostate cancer screening and hemoglobin A1c is measured in diabetes management, radiographs should be used to track progression of disease in RA and PsA, Dr. Wells argued. “[I]f you don’t know, you can’t treat,” he said.
Some patients near remission may have radiographic progression even though disease activity measurements such as C-reactive protein (CRP) values do not show presence of active disease. In a study analyzing 1,184 patients with RA in the ASPIRE, ERA, Leflunomide, PREMIER and TEMPO trials, swollen joint count (SJC) was a better predictor of radiographic progression than CRP in patients near remission.
“[E]ven where you don’t see smoke, there still could be fire,” Dr. Wells said. “Some of these patients still progress and these are outliers, and the way they saw that [was] because they followed those patients along. If you don’t look, you don’t know.”
Radiographic progression can also be seen among nonswollen joints in patients with RA and PsA. In a study of 1,207 joints in 55 patients with RA and 352 joints in 18 patients with PsA, researchers in Austria found tenderness in nonswollen joints was associated with radiographic progression.
Despite having effective treatments in RA and PsA, “none of our therapies show that they’re able to prevent progression,” Dr. Wells said.
When it comes to hitting the treatment target in RA, some rheumatologists may think they can accomplish it without use of repeated radiographs. “I have a different perspective on that – that you really do indeed need to do the x-rays today and follow those x-rays along, especially if it’s going to change your treatment paradigm and what your treatment decision would be for the patient,” he said.
Counterpoint: Repeat radiographs aren’t helpful
Almost all rheumatologists would likely order an initial radiograph for their patients with RA or PsA, Roy M. Fleischmann, MD, clinical professor of medicine at the University of Texas and codirector of the Metroplex Clinical Research Center, both in Dallas, said in his presentation.
“If you see erosions when you start, chances are you’re going to be much more aggressive,” Dr. Fleischmann said. “So it is justification for early, more aggressive treatment of disease.”
In recent decades, radiographic progression in RA has decreased as more effective antirheumatic treatments have come into use, Dr. Fleischmann argued.
“We had x-ray progression in virtually everybody, and it was consistent no matter what we treated with, which was gold or penicillamine or any of the NSAIDs or sulfasalazine,” he said. “With methotrexate ... about 60% of patients actually have no x-ray progression, and that was a major change, and that’s one of the reasons why methotrexate has become the keystone of therapy. But even with methotrexate, [we] still had many patients who progressed.”
After the introduction of tumor necrosis factor inhibitors and other mechanisms in the late 1990s, “all of a sudden, you don’t see x-ray progression – mean x-ray progression – in a group of patients,” he noted.
Many rheumatologists now use a treat-to-target strategy, and if the patient achieves true clinical remission or sustained very low disease activity as measured by Boolean remission, Simple Disease Activity Index, or Clinical Disease Activity Index, they have “very little chance of radiographic progression and functional decline,” he said.
“If a patient doesn’t achieve remission or very low disease activity, obtaining a radiograph doesn’t change what you do because the patient’s not where they want to be, where you want them to be; you’re going to make a change anyway,” Dr. Fleischmann explained. “The radiograph isn’t going to help you do that.”
If a patient is in sustained remission but a radiograph is ordered and shows disease progression, he questioned what the rheumatologist would do in that situation.
“Now the patient’s in, let’s say, a Boolean remission. They have no tender joints. They have no swollen joints ... their pain assessment is zero, their CRP is zero, and they do have some x-ray progression. Where are you going to change?” Dr. Fleischmann asked. “There’s no data that anything else would work. I don’t know what you would do. So, in conclusion, I would say you really don’t need to repeat an x-ray.”
AI reading x-rays?
Commenting on the point-counterpoint session, Arthur Kavanaugh, MD, professor of medicine at the University of California, San Diego, and director of RWCS, asked Dr. Fleischmann and Dr. Wells how they address the issue of how many radiologists seem to be unfamiliar with reading hand radiographs and RA progression.
Dr. Fleischmann said he was trained in how to read hand radiographs in medical school, but that training no longer appears to be occurring. “If you have a good bone radiologist, of which there are not a lot, you’re great. But if you don’t have a really good bone radiologist, it’s difficult,” he said.
Dr. Kavanaugh alluded to the advancement of artificial intelligence (AI) in radiology and posed the question of how both rheumatologists felt about AI reading and interpreting their radiographs. “If you could reliably submit x-rays and they would say what the Sharp score was and where the differences were, would that change anything?” he asked.
“I think having artificial intelligence read the x-ray or an MRI is really, really good. It’ll be better than the radiologists,” Dr. Fleischmann responded. “But I don’t think that you really need to repeat the x-ray. I mean, I really don’t think you need to repeat it. You need to treat the patient.”
Dr. Wells reported having financial relationships with numerous pharmaceutical companies. Dr. Fleischmann reported no relevant financial relationships.
Follow-up radiographs after an initial baseline reading in patients with rheumatoid arthritis or psoriatic arthritis may still show radiographic progression despite treatment with current therapies, but it’s unclear if they will affect treatment decisions between patients and doctors that may take place regardless of the radiographic information, according to arguments made for and against their usefulness in a point-counterpoint session at the 2023 Rheumatology Winter Clinical Symposium.
Alvin Wells, MD, PhD, director of the department of rheumatology at Advocate Aurora Health in Franklin, Wisc., said that x-rays “reflect the history of joint pathology” and can get worse over time, correlating with disease activity and severity.
While RA does not yet have the “holy grail” of complete or true remission, Dr. Wells argued, the combination of clinical remission, laboratory remission, and imaging remission gets patients with RA close to the ideal when measured over time. “You need to continue to monitor these patients as you follow them along,” he said.
The BARFOT study, which evaluated 1,938 patients with early RA in two cohorts during 1992-1999 and again between 2000 and 2006, showed that more active treatments in the 2000s did not result in improvements in Health Assessment Questionnaire (HAQ) and pain scores, compared with patients treated in the 1990s. “You can see in some of those patients those scores do increase, and that even despite aggressive therapies that we had in 2006, you can still see some of those patients still have progression of the disease,” Dr. Wells explained. “How did they know? Because they looked.”
He also cited a study from researchers at the Mayo Clinic who examined 586 patients with RA that showed a higher prevalence of functional disability in patients with RA who also had radiographic changes, compared with patients without RA. “Radiographic changes correlate with disease severity and functional disability as well,” Dr. Wells said.
Just as prostate-specific antigen levels are used in prostate cancer screening and hemoglobin A1c is measured in diabetes management, radiographs should be used to track progression of disease in RA and PsA, Dr. Wells argued. “[I]f you don’t know, you can’t treat,” he said.
Some patients near remission may have radiographic progression even though disease activity measurements such as C-reactive protein (CRP) values do not show presence of active disease. In a study analyzing 1,184 patients with RA in the ASPIRE, ERA, Leflunomide, PREMIER and TEMPO trials, swollen joint count (SJC) was a better predictor of radiographic progression than CRP in patients near remission.
“[E]ven where you don’t see smoke, there still could be fire,” Dr. Wells said. “Some of these patients still progress and these are outliers, and the way they saw that [was] because they followed those patients along. If you don’t look, you don’t know.”
Radiographic progression can also be seen among nonswollen joints in patients with RA and PsA. In a study of 1,207 joints in 55 patients with RA and 352 joints in 18 patients with PsA, researchers in Austria found tenderness in nonswollen joints was associated with radiographic progression.
Despite having effective treatments in RA and PsA, “none of our therapies show that they’re able to prevent progression,” Dr. Wells said.
When it comes to hitting the treatment target in RA, some rheumatologists may think they can accomplish it without use of repeated radiographs. “I have a different perspective on that – that you really do indeed need to do the x-rays today and follow those x-rays along, especially if it’s going to change your treatment paradigm and what your treatment decision would be for the patient,” he said.
Counterpoint: Repeat radiographs aren’t helpful
Almost all rheumatologists would likely order an initial radiograph for their patients with RA or PsA, Roy M. Fleischmann, MD, clinical professor of medicine at the University of Texas and codirector of the Metroplex Clinical Research Center, both in Dallas, said in his presentation.
“If you see erosions when you start, chances are you’re going to be much more aggressive,” Dr. Fleischmann said. “So it is justification for early, more aggressive treatment of disease.”
In recent decades, radiographic progression in RA has decreased as more effective antirheumatic treatments have come into use, Dr. Fleischmann argued.
“We had x-ray progression in virtually everybody, and it was consistent no matter what we treated with, which was gold or penicillamine or any of the NSAIDs or sulfasalazine,” he said. “With methotrexate ... about 60% of patients actually have no x-ray progression, and that was a major change, and that’s one of the reasons why methotrexate has become the keystone of therapy. But even with methotrexate, [we] still had many patients who progressed.”
After the introduction of tumor necrosis factor inhibitors and other mechanisms in the late 1990s, “all of a sudden, you don’t see x-ray progression – mean x-ray progression – in a group of patients,” he noted.
Many rheumatologists now use a treat-to-target strategy, and if the patient achieves true clinical remission or sustained very low disease activity as measured by Boolean remission, Simple Disease Activity Index, or Clinical Disease Activity Index, they have “very little chance of radiographic progression and functional decline,” he said.
“If a patient doesn’t achieve remission or very low disease activity, obtaining a radiograph doesn’t change what you do because the patient’s not where they want to be, where you want them to be; you’re going to make a change anyway,” Dr. Fleischmann explained. “The radiograph isn’t going to help you do that.”
If a patient is in sustained remission but a radiograph is ordered and shows disease progression, he questioned what the rheumatologist would do in that situation.
“Now the patient’s in, let’s say, a Boolean remission. They have no tender joints. They have no swollen joints ... their pain assessment is zero, their CRP is zero, and they do have some x-ray progression. Where are you going to change?” Dr. Fleischmann asked. “There’s no data that anything else would work. I don’t know what you would do. So, in conclusion, I would say you really don’t need to repeat an x-ray.”
AI reading x-rays?
Commenting on the point-counterpoint session, Arthur Kavanaugh, MD, professor of medicine at the University of California, San Diego, and director of RWCS, asked Dr. Fleischmann and Dr. Wells how they address the issue of how many radiologists seem to be unfamiliar with reading hand radiographs and RA progression.
Dr. Fleischmann said he was trained in how to read hand radiographs in medical school, but that training no longer appears to be occurring. “If you have a good bone radiologist, of which there are not a lot, you’re great. But if you don’t have a really good bone radiologist, it’s difficult,” he said.
Dr. Kavanaugh alluded to the advancement of artificial intelligence (AI) in radiology and posed the question of how both rheumatologists felt about AI reading and interpreting their radiographs. “If you could reliably submit x-rays and they would say what the Sharp score was and where the differences were, would that change anything?” he asked.
“I think having artificial intelligence read the x-ray or an MRI is really, really good. It’ll be better than the radiologists,” Dr. Fleischmann responded. “But I don’t think that you really need to repeat the x-ray. I mean, I really don’t think you need to repeat it. You need to treat the patient.”
Dr. Wells reported having financial relationships with numerous pharmaceutical companies. Dr. Fleischmann reported no relevant financial relationships.
FROM RWCS 2023
New tool better estimates cardiovascular risk in people with lupus
Current risk estimators are inaccurate
A tool that incorporates lupus-related variables with traditional risk factors provides a much more accurate assessment of cardiovascular (CV) risk in patients with systemic lupus erythematosus (SLE), according to data presented at the annual meeting of the Canadian Rheumatology Association.
In the initial clinical assessment of this tool, called the SLECRISK, “it identified high-risk lupus patients who would otherwise be missed by traditional methods of CV risk assessment,” reported May Y. Choi, MD, associate director of translational research at the University of Calgary’s (Alta.) Lupus Centre of Excellence.
It is well known that patients with SLE face an increased risk of CV events starting at an age long before risk begins climbing in the general population, according to Dr. Choi. She cited one study that showed women aged 35-44 years have a 50-fold greater risk of myocardial infarction than healthy individuals.
All major guidelines recognize this increased risk and recommend CV risk assessment in patients with SLE, even though Dr. Choi pointed out that traditional tools, such as the American College of Cardiology atherosclerotic cardiovascular disease (ASCVD) risk calculator or the Framingham Risk Score (FRS) have a limited ability to detect the patients with SLE who are most likely to have an event.
In SLE, current tools are inadequate
“These risk assessment tools perform poorly in SLE patients because they do not capture SLE-related inflammation,” Dr. Choi said. Of several examples, Dr. Choi cited a study showing “seven times more MIs and strokes observed than expected in SLE patients on the basis of the FRS.”
The disparity between expected and observed MIs and strokes is worse with increasing severity of SLE. In a study she presented 3 years ago, rates of CV events were 12 times higher in those with inactive or mild SLE, rising to a 16-fold increase among those with moderate disease and jumping to a 32-fold increase in those with severe SLE.
The SLECRISK tool was developed from the Brigham and Women’s Hospital SLE Registry, which was initiated in 1992. Patients without a history of CV disease were evaluated for traditional CV risk factors and for SLE-specific characteristics such as disease activity, levels of the complement proteins C3 and C4, kidney function, the presence of nephritis, and SLE duration. The value of these characteristics as predictors of CV events were then assessed over a 10-year follow-up period before being assembled into the SLECRISK tool.
In an example of the risk equation, Dr. Choi described a 50-year-old patient with SLE and a 5% 10-year ASCVD risk score, which is low. After adjustment for SLE risks, which included 10 years disease duration, high disease activity, elevated creatinine, and positive anti–double stranded DNA status, the 10-year CV risk score climbed to 16.2%, which is moderate.
The performance of the SLECRISK was evaluated in 1,243 patients providing 8,946.51 person-years of follow-up. During this period, there were 90 major adverse cardiac events (MACE), of which 82% were adjudicated by cardiologists, and 211 secondary events.
Relative to the ASCVD risk score, the SLECRISK identified about twice as many patients with SLE as having moderate risk and 3.5-fold more patients as having high risk. Among patients who experienced CV events, traditional CV risk factors were more common but so were SLE-specific risk factors, including greater disease severity, a greater likelihood of lupus nephritis, increased complement levels, and greater exposure to glucocorticoids, according to Dr. Choi.
Specificities for CV events higher on SLECRISK
In predicting CV events, the differences in specificities were in the same general range, although somewhat higher for the ASCVD risk score in regard to predicting MACE (83% vs. 72%) and MACE plus secondary events (90% vs. 79%). However, the sensitivities were much higher for SLECRISK relative to the ASCVD risk score for MACE alone (64% vs. 41%) and for MACE plus secondary events (58% vs. 35%).
When comparing those who had an MI or stroke, the ASCVD risk score identified 8 (7%) patients missed by SLECRISK, whereas SLECRISK identified 89 (73%) missed by the ASCVD risk score. The remaining 25 patients (20%) were identified by both. The advantage of SLECRISK was similar for MACE plus secondary outcomes.
Dr. Choi noted that all of the SLE-specific variables in SLECRISK are readily obtained and often already available in patient charts. She said that there is a plan to validate the tool in larger groups, but with a goal of creating a tool available online for clinicians and their patients to use. There is also an even more ambitious plan for the future.
“We have funding to look at machine learning to evaluate predictive variables in SLE patients,” Dr. Choi said. Rather than adding SLE-specific variables to traditional risks, the plan is to “start from scratch,” letting artificial intelligence assemble predictors without prejudice to what might or might not be relevant.
A SLE-specific tool for evaluating CV risk is an important “unmet need,” according to Karen H. Costenbader, MD, professor in the division of rheumatology, inflammation, and immunity at Brigham and Women’s Hospital and Harvard Medical School, both in Boston. In an interview, she reiterated that measuring CV risk in SLE is already guideline recommended, but conventional tools have been shown to be inaccurate.
“I can envision it being used in clinical encounters to help guide shared decision-making with patients,” explained Dr. Costenbader, who was not involved in the presentation at the CRA meeting but worked with Dr. Choi in developing SLECRISK. “It would give us more precise estimates, allowing us to risk stratify our patients and informing us as to which modifiable SLE-specific and nonspecific factors are contributing most to CV risk.’
The problem of using conventional risk assessments in SLE has been well recognized. Of those who have written on this subject, Maureen McMahon, MD, site director of the Lupus Clinical Trials Network at the University of California, Los Angeles, said: “There is a critical need for the development of SLE-specific risk assessment tools like SLECRISK.”
Author of several studies looking at alternatives for CV risk assessment in SLE, including a study looking at a panel of biomarkers that was published in ACR Open Rheumatology, Dr. McMahon said in an interview that CV risk in SLE is high but conventional risk assessments are flawed.
“Multiple previous studies have demonstrated that these currently available calculators are not adequate for identifying risk in the lupus patient population,” she said. According to Dr. McMahon, the fact that rheumatologists remain “dependent upon [these conventional] cardiovascular risk calculators” is a well-recognized problem that needs resolution.
Dr. Choi has financial relationships with AstraZeneca, GlaxoSmithKline, Mallinckrodt. MitogenDx, Organon, and Werfen International. Dr. Costenbader reports no potential conflicts of interest. Dr. McMahon has financial relationships with AstraZeneca, Aurinia Pharmaceuticals, Eli Lilly, and GlaxoSmithKline.
Current risk estimators are inaccurate
Current risk estimators are inaccurate
A tool that incorporates lupus-related variables with traditional risk factors provides a much more accurate assessment of cardiovascular (CV) risk in patients with systemic lupus erythematosus (SLE), according to data presented at the annual meeting of the Canadian Rheumatology Association.
In the initial clinical assessment of this tool, called the SLECRISK, “it identified high-risk lupus patients who would otherwise be missed by traditional methods of CV risk assessment,” reported May Y. Choi, MD, associate director of translational research at the University of Calgary’s (Alta.) Lupus Centre of Excellence.
It is well known that patients with SLE face an increased risk of CV events starting at an age long before risk begins climbing in the general population, according to Dr. Choi. She cited one study that showed women aged 35-44 years have a 50-fold greater risk of myocardial infarction than healthy individuals.
All major guidelines recognize this increased risk and recommend CV risk assessment in patients with SLE, even though Dr. Choi pointed out that traditional tools, such as the American College of Cardiology atherosclerotic cardiovascular disease (ASCVD) risk calculator or the Framingham Risk Score (FRS) have a limited ability to detect the patients with SLE who are most likely to have an event.
In SLE, current tools are inadequate
“These risk assessment tools perform poorly in SLE patients because they do not capture SLE-related inflammation,” Dr. Choi said. Of several examples, Dr. Choi cited a study showing “seven times more MIs and strokes observed than expected in SLE patients on the basis of the FRS.”
The disparity between expected and observed MIs and strokes is worse with increasing severity of SLE. In a study she presented 3 years ago, rates of CV events were 12 times higher in those with inactive or mild SLE, rising to a 16-fold increase among those with moderate disease and jumping to a 32-fold increase in those with severe SLE.
The SLECRISK tool was developed from the Brigham and Women’s Hospital SLE Registry, which was initiated in 1992. Patients without a history of CV disease were evaluated for traditional CV risk factors and for SLE-specific characteristics such as disease activity, levels of the complement proteins C3 and C4, kidney function, the presence of nephritis, and SLE duration. The value of these characteristics as predictors of CV events were then assessed over a 10-year follow-up period before being assembled into the SLECRISK tool.
In an example of the risk equation, Dr. Choi described a 50-year-old patient with SLE and a 5% 10-year ASCVD risk score, which is low. After adjustment for SLE risks, which included 10 years disease duration, high disease activity, elevated creatinine, and positive anti–double stranded DNA status, the 10-year CV risk score climbed to 16.2%, which is moderate.
The performance of the SLECRISK was evaluated in 1,243 patients providing 8,946.51 person-years of follow-up. During this period, there were 90 major adverse cardiac events (MACE), of which 82% were adjudicated by cardiologists, and 211 secondary events.
Relative to the ASCVD risk score, the SLECRISK identified about twice as many patients with SLE as having moderate risk and 3.5-fold more patients as having high risk. Among patients who experienced CV events, traditional CV risk factors were more common but so were SLE-specific risk factors, including greater disease severity, a greater likelihood of lupus nephritis, increased complement levels, and greater exposure to glucocorticoids, according to Dr. Choi.
Specificities for CV events higher on SLECRISK
In predicting CV events, the differences in specificities were in the same general range, although somewhat higher for the ASCVD risk score in regard to predicting MACE (83% vs. 72%) and MACE plus secondary events (90% vs. 79%). However, the sensitivities were much higher for SLECRISK relative to the ASCVD risk score for MACE alone (64% vs. 41%) and for MACE plus secondary events (58% vs. 35%).
When comparing those who had an MI or stroke, the ASCVD risk score identified 8 (7%) patients missed by SLECRISK, whereas SLECRISK identified 89 (73%) missed by the ASCVD risk score. The remaining 25 patients (20%) were identified by both. The advantage of SLECRISK was similar for MACE plus secondary outcomes.
Dr. Choi noted that all of the SLE-specific variables in SLECRISK are readily obtained and often already available in patient charts. She said that there is a plan to validate the tool in larger groups, but with a goal of creating a tool available online for clinicians and their patients to use. There is also an even more ambitious plan for the future.
“We have funding to look at machine learning to evaluate predictive variables in SLE patients,” Dr. Choi said. Rather than adding SLE-specific variables to traditional risks, the plan is to “start from scratch,” letting artificial intelligence assemble predictors without prejudice to what might or might not be relevant.
A SLE-specific tool for evaluating CV risk is an important “unmet need,” according to Karen H. Costenbader, MD, professor in the division of rheumatology, inflammation, and immunity at Brigham and Women’s Hospital and Harvard Medical School, both in Boston. In an interview, she reiterated that measuring CV risk in SLE is already guideline recommended, but conventional tools have been shown to be inaccurate.
“I can envision it being used in clinical encounters to help guide shared decision-making with patients,” explained Dr. Costenbader, who was not involved in the presentation at the CRA meeting but worked with Dr. Choi in developing SLECRISK. “It would give us more precise estimates, allowing us to risk stratify our patients and informing us as to which modifiable SLE-specific and nonspecific factors are contributing most to CV risk.’
The problem of using conventional risk assessments in SLE has been well recognized. Of those who have written on this subject, Maureen McMahon, MD, site director of the Lupus Clinical Trials Network at the University of California, Los Angeles, said: “There is a critical need for the development of SLE-specific risk assessment tools like SLECRISK.”
Author of several studies looking at alternatives for CV risk assessment in SLE, including a study looking at a panel of biomarkers that was published in ACR Open Rheumatology, Dr. McMahon said in an interview that CV risk in SLE is high but conventional risk assessments are flawed.
“Multiple previous studies have demonstrated that these currently available calculators are not adequate for identifying risk in the lupus patient population,” she said. According to Dr. McMahon, the fact that rheumatologists remain “dependent upon [these conventional] cardiovascular risk calculators” is a well-recognized problem that needs resolution.
Dr. Choi has financial relationships with AstraZeneca, GlaxoSmithKline, Mallinckrodt. MitogenDx, Organon, and Werfen International. Dr. Costenbader reports no potential conflicts of interest. Dr. McMahon has financial relationships with AstraZeneca, Aurinia Pharmaceuticals, Eli Lilly, and GlaxoSmithKline.
A tool that incorporates lupus-related variables with traditional risk factors provides a much more accurate assessment of cardiovascular (CV) risk in patients with systemic lupus erythematosus (SLE), according to data presented at the annual meeting of the Canadian Rheumatology Association.
In the initial clinical assessment of this tool, called the SLECRISK, “it identified high-risk lupus patients who would otherwise be missed by traditional methods of CV risk assessment,” reported May Y. Choi, MD, associate director of translational research at the University of Calgary’s (Alta.) Lupus Centre of Excellence.
It is well known that patients with SLE face an increased risk of CV events starting at an age long before risk begins climbing in the general population, according to Dr. Choi. She cited one study that showed women aged 35-44 years have a 50-fold greater risk of myocardial infarction than healthy individuals.
All major guidelines recognize this increased risk and recommend CV risk assessment in patients with SLE, even though Dr. Choi pointed out that traditional tools, such as the American College of Cardiology atherosclerotic cardiovascular disease (ASCVD) risk calculator or the Framingham Risk Score (FRS) have a limited ability to detect the patients with SLE who are most likely to have an event.
In SLE, current tools are inadequate
“These risk assessment tools perform poorly in SLE patients because they do not capture SLE-related inflammation,” Dr. Choi said. Of several examples, Dr. Choi cited a study showing “seven times more MIs and strokes observed than expected in SLE patients on the basis of the FRS.”
The disparity between expected and observed MIs and strokes is worse with increasing severity of SLE. In a study she presented 3 years ago, rates of CV events were 12 times higher in those with inactive or mild SLE, rising to a 16-fold increase among those with moderate disease and jumping to a 32-fold increase in those with severe SLE.
The SLECRISK tool was developed from the Brigham and Women’s Hospital SLE Registry, which was initiated in 1992. Patients without a history of CV disease were evaluated for traditional CV risk factors and for SLE-specific characteristics such as disease activity, levels of the complement proteins C3 and C4, kidney function, the presence of nephritis, and SLE duration. The value of these characteristics as predictors of CV events were then assessed over a 10-year follow-up period before being assembled into the SLECRISK tool.
In an example of the risk equation, Dr. Choi described a 50-year-old patient with SLE and a 5% 10-year ASCVD risk score, which is low. After adjustment for SLE risks, which included 10 years disease duration, high disease activity, elevated creatinine, and positive anti–double stranded DNA status, the 10-year CV risk score climbed to 16.2%, which is moderate.
The performance of the SLECRISK was evaluated in 1,243 patients providing 8,946.51 person-years of follow-up. During this period, there were 90 major adverse cardiac events (MACE), of which 82% were adjudicated by cardiologists, and 211 secondary events.
Relative to the ASCVD risk score, the SLECRISK identified about twice as many patients with SLE as having moderate risk and 3.5-fold more patients as having high risk. Among patients who experienced CV events, traditional CV risk factors were more common but so were SLE-specific risk factors, including greater disease severity, a greater likelihood of lupus nephritis, increased complement levels, and greater exposure to glucocorticoids, according to Dr. Choi.
Specificities for CV events higher on SLECRISK
In predicting CV events, the differences in specificities were in the same general range, although somewhat higher for the ASCVD risk score in regard to predicting MACE (83% vs. 72%) and MACE plus secondary events (90% vs. 79%). However, the sensitivities were much higher for SLECRISK relative to the ASCVD risk score for MACE alone (64% vs. 41%) and for MACE plus secondary events (58% vs. 35%).
When comparing those who had an MI or stroke, the ASCVD risk score identified 8 (7%) patients missed by SLECRISK, whereas SLECRISK identified 89 (73%) missed by the ASCVD risk score. The remaining 25 patients (20%) were identified by both. The advantage of SLECRISK was similar for MACE plus secondary outcomes.
Dr. Choi noted that all of the SLE-specific variables in SLECRISK are readily obtained and often already available in patient charts. She said that there is a plan to validate the tool in larger groups, but with a goal of creating a tool available online for clinicians and their patients to use. There is also an even more ambitious plan for the future.
“We have funding to look at machine learning to evaluate predictive variables in SLE patients,” Dr. Choi said. Rather than adding SLE-specific variables to traditional risks, the plan is to “start from scratch,” letting artificial intelligence assemble predictors without prejudice to what might or might not be relevant.
A SLE-specific tool for evaluating CV risk is an important “unmet need,” according to Karen H. Costenbader, MD, professor in the division of rheumatology, inflammation, and immunity at Brigham and Women’s Hospital and Harvard Medical School, both in Boston. In an interview, she reiterated that measuring CV risk in SLE is already guideline recommended, but conventional tools have been shown to be inaccurate.
“I can envision it being used in clinical encounters to help guide shared decision-making with patients,” explained Dr. Costenbader, who was not involved in the presentation at the CRA meeting but worked with Dr. Choi in developing SLECRISK. “It would give us more precise estimates, allowing us to risk stratify our patients and informing us as to which modifiable SLE-specific and nonspecific factors are contributing most to CV risk.’
The problem of using conventional risk assessments in SLE has been well recognized. Of those who have written on this subject, Maureen McMahon, MD, site director of the Lupus Clinical Trials Network at the University of California, Los Angeles, said: “There is a critical need for the development of SLE-specific risk assessment tools like SLECRISK.”
Author of several studies looking at alternatives for CV risk assessment in SLE, including a study looking at a panel of biomarkers that was published in ACR Open Rheumatology, Dr. McMahon said in an interview that CV risk in SLE is high but conventional risk assessments are flawed.
“Multiple previous studies have demonstrated that these currently available calculators are not adequate for identifying risk in the lupus patient population,” she said. According to Dr. McMahon, the fact that rheumatologists remain “dependent upon [these conventional] cardiovascular risk calculators” is a well-recognized problem that needs resolution.
Dr. Choi has financial relationships with AstraZeneca, GlaxoSmithKline, Mallinckrodt. MitogenDx, Organon, and Werfen International. Dr. Costenbader reports no potential conflicts of interest. Dr. McMahon has financial relationships with AstraZeneca, Aurinia Pharmaceuticals, Eli Lilly, and GlaxoSmithKline.
FROM CRA 2023
Medicare ‘offers’ cancer patient a choice: Less life or more debt
We’re gonna need a bigger meth lab
In case you’ve been living under a rock for the past 15 years, the TV show “Breaking Bad” details the spiraling rise and downfall of a high school chemistry teacher who, after developing a case of terminal lung cancer, starts producing methamphetamine to provide for his family in response to the steep cost of treatment for his cancer.
Meanwhile, here in 2023 in the real world, we have Paul Davis, a retired physician in Ohio, who’s being forced to choose between an expensive cancer treatment and bankrupting his family, since Medicare’s decided it doesn’t want to cover the cost. Hey, we’ve seen this one before!
A bit of backstory: In November 2019, Dr. Davis was diagnosed with uveal melanoma, a very rare type of cancer that affects eye tissue. The news got worse in 2022 when the cancer spread to his liver, a move which typically proves fatal within a year. However, in a stroke of great news, the Food and Drug Administration approved the drug Kimmtrak earlier that year, which could be used to treat his cancer. Not cure, of course, but it would give him more time.
His initial treatments with the drug went fine and were covered, but when he transferred his care from a hospital in Columbus to one closer to home, big problem. Medicare decided it didn’t like that hospital and abruptly cut off coverage, denying the local hospital’s claims. That leaves Dr. Davis on the hook for his cancer treatment, and it’s what you might call expensive. Expensive to the tune of $50,000.
A week.
Apparently the coding the local hospital submitted was wrong, indicating that Dr. Davis was receiving Kimmtrak for a type of cancer that the FDA hadn’t approved the drug for. So until the government bureaucracy works itself out, his treatment is on hold, leaving all his faith in Medicare working quickly to rectify its mistake. If it can rectify its mistake. We’re not hopeful.
And in case you were wondering, if Dr. Davis wanted to go full Walter White, the average street price of meth is about $20-$60 per gram, so to pay for his treatment, he’d need to make at least a kilogram of meth every week. That’s, uh, quite a lot of illegal drug, or what we here at the LOTME office would call a fun Saturday night.
When you give a mouse a movie
Researchers have been successfully testing Alzheimer drugs on mice for years, but none of the drugs has proved successful in humans. Recent work, however, might have found the missing link, and it’s a combination no one ever thought of before: mice and movies.
Turns out that Orson Welles’ 1958 film noir classic “Touch of Evil” tapped a part of the mouse brain that has been overlooked: the hippocampus, which is crucial for learning and memory. Previous researchers thought it was just used as a kind of GPS system, but that’s only partially true.
Not only did the mice choose to pay attention to the movie clip, but the hippocampus responded to the visual stimuli only when the rodents saw the scenes from the clip later in the order that they were presented and not in a scrambled order. These findings represent a “major paradigm shift” in studying mouse recall, Mayank Mehta, PhD, of the University of California, Los Angeles, said in a statement from the school.
This breakthrough could run parallel to Alzheimer’s patients struggling with similar defects. “Selective and episodic activation of the mouse hippocampus using a human movie opens up the possibility of directly testing human episodic memory disorders and therapies using mouse neurons, a major step forward,” said coauthor Chinmay Purandare, PhD, who is now at the University of California, San Francisco.
Who would have thought that a classic film would help advance Alzheimer research?
A less human way to study mosquitoes
We here at LOTME have a history with mosquitoes. We know they don’t like us, and they know that we don’t like them. Trust us, they know. So when humans gain a little ground in the war against the buzzy little bloodsuckers, we want to share the joy.
To know the enemy, scientists have to study the enemy, but there is a problem. “Many mosquito experiments still rely on human volunteers and animal subjects,” bioengineering graduate student Kevin Janson, said in a statement from Rice University. Most people don’t like being bitten by mosquitoes, so that kind of testing can be expensive.
Is there a way to automate the collection and processing of mosquito behavior data using inexpensive cameras and machine-learning software? We’re glad you asked, because Mr. Janson and the research team, which includes bioengineers from Rice and tropical medicine experts from Tulane University, have managed to eliminate the need for live volunteers by using patches of synthetic skin made with a 3D printer.
“Each patch of gelatin-like hydrogel comes complete with tiny passageways that can be filled with flowing blood” from a chicken, sheep, or cow, they explained, and proof-of-concept testing showed that mosquitoes would feed on hydrogels without any repellent and stay away from those treated with a repellent.
To conduct the feeding tests, the blood-infused hydrogels are placed in a clear plastic box that is surrounded by cameras.
A bunch of mosquitoes are then tossed in the box and the cameras record all their insect activities: how often they land at each location, how long they stay, whether or not they bite, how long they feed, etc. Humans don’t have to watch and don’t have to be food sources.
Humans don’t have to be food sources, and we just pictured the future of mosquito control. Imagine a dozen Arnold Schwarzenegger–style Terminators, covered in 3D-printed skin, walking through your neighborhood in the summer while wearing sweat-soaked, brightly colored clothing. The mosquitoes wouldn’t be able to stay away, but guess what? They’re feeding off robots with nonhuman skin and nonhuman blood, so we win. It’s good to have a cerebral cortex.
Getting medieval on brain surgery
Let’s get one thing clear: The so-called “Dark Ages” were not nearly as dark as they’re made out to be. For one thing, there’s a world beyond Western Europe. The Roman Empire didn’t collapse everywhere. But even in Western Europe, the centuries between the fall of Rome and the Renaissance were hardly lacking in cultural development.
That said, we wouldn’t want to be in the position of the seventh-century noblewoman whose remains were recently uncovered in a Byzantine fortress in central Italy with multiple cross-shaped incisions in her skull. Yes, this unfortunate woman underwent at least two brain surgeries.
Then again, maybe not. Nothing like it had been discovered at the site, and while the markings – signs of a procedure called trepanation – can be surgical in nature, there are other explanations. For example, the Avar people practiced ritual trepanation during the same time period, but they were hundreds of miles away in the Carpathian mountains, and there was no evidence to support that a different form of ritualistic trepanation ever took place in Byzantine-era Italy.
The investigators then moved on to a form of judicial punishment called decalvatio, which involves mutilation by scalping. Look, the Dark Ages weren’t dark, but no one said they were fun. Anyway, this was discarded, since decalvatio was only meted out to soldiers who deserted the battlefield.
That brings us back to surgery. While one of the trepanations was fully engraved into her skull, indicating that the woman died soon after the surgery, she also bore indications of a healed trepanation. A 50% success rate isn’t terrible for our medieval surgeon. Sure, the Incas managed 80%, but even during the Civil War brain surgery only had a 50% success rate. And that’s the end of the story, nothing more to say about our medieval Italian woman.
Nope. Nothing at all.
Fine. While a surgical procedure was deemed most likely, the study investigators found no direct evidence of a medical condition. No trauma, no tumor, nothing. Just a couple of suggestions of “a systemic pathological condition,” they said. Okay, we swear, it really wasn’t that bad in the Middle [Editor’s note: Approximately 5,000 more words on medieval culture not included. This is a medical column, thank you very much.]
We’re gonna need a bigger meth lab
In case you’ve been living under a rock for the past 15 years, the TV show “Breaking Bad” details the spiraling rise and downfall of a high school chemistry teacher who, after developing a case of terminal lung cancer, starts producing methamphetamine to provide for his family in response to the steep cost of treatment for his cancer.
Meanwhile, here in 2023 in the real world, we have Paul Davis, a retired physician in Ohio, who’s being forced to choose between an expensive cancer treatment and bankrupting his family, since Medicare’s decided it doesn’t want to cover the cost. Hey, we’ve seen this one before!
A bit of backstory: In November 2019, Dr. Davis was diagnosed with uveal melanoma, a very rare type of cancer that affects eye tissue. The news got worse in 2022 when the cancer spread to his liver, a move which typically proves fatal within a year. However, in a stroke of great news, the Food and Drug Administration approved the drug Kimmtrak earlier that year, which could be used to treat his cancer. Not cure, of course, but it would give him more time.
His initial treatments with the drug went fine and were covered, but when he transferred his care from a hospital in Columbus to one closer to home, big problem. Medicare decided it didn’t like that hospital and abruptly cut off coverage, denying the local hospital’s claims. That leaves Dr. Davis on the hook for his cancer treatment, and it’s what you might call expensive. Expensive to the tune of $50,000.
A week.
Apparently the coding the local hospital submitted was wrong, indicating that Dr. Davis was receiving Kimmtrak for a type of cancer that the FDA hadn’t approved the drug for. So until the government bureaucracy works itself out, his treatment is on hold, leaving all his faith in Medicare working quickly to rectify its mistake. If it can rectify its mistake. We’re not hopeful.
And in case you were wondering, if Dr. Davis wanted to go full Walter White, the average street price of meth is about $20-$60 per gram, so to pay for his treatment, he’d need to make at least a kilogram of meth every week. That’s, uh, quite a lot of illegal drug, or what we here at the LOTME office would call a fun Saturday night.
When you give a mouse a movie
Researchers have been successfully testing Alzheimer drugs on mice for years, but none of the drugs has proved successful in humans. Recent work, however, might have found the missing link, and it’s a combination no one ever thought of before: mice and movies.
Turns out that Orson Welles’ 1958 film noir classic “Touch of Evil” tapped a part of the mouse brain that has been overlooked: the hippocampus, which is crucial for learning and memory. Previous researchers thought it was just used as a kind of GPS system, but that’s only partially true.
Not only did the mice choose to pay attention to the movie clip, but the hippocampus responded to the visual stimuli only when the rodents saw the scenes from the clip later in the order that they were presented and not in a scrambled order. These findings represent a “major paradigm shift” in studying mouse recall, Mayank Mehta, PhD, of the University of California, Los Angeles, said in a statement from the school.
This breakthrough could run parallel to Alzheimer’s patients struggling with similar defects. “Selective and episodic activation of the mouse hippocampus using a human movie opens up the possibility of directly testing human episodic memory disorders and therapies using mouse neurons, a major step forward,” said coauthor Chinmay Purandare, PhD, who is now at the University of California, San Francisco.
Who would have thought that a classic film would help advance Alzheimer research?
A less human way to study mosquitoes
We here at LOTME have a history with mosquitoes. We know they don’t like us, and they know that we don’t like them. Trust us, they know. So when humans gain a little ground in the war against the buzzy little bloodsuckers, we want to share the joy.
To know the enemy, scientists have to study the enemy, but there is a problem. “Many mosquito experiments still rely on human volunteers and animal subjects,” bioengineering graduate student Kevin Janson, said in a statement from Rice University. Most people don’t like being bitten by mosquitoes, so that kind of testing can be expensive.
Is there a way to automate the collection and processing of mosquito behavior data using inexpensive cameras and machine-learning software? We’re glad you asked, because Mr. Janson and the research team, which includes bioengineers from Rice and tropical medicine experts from Tulane University, have managed to eliminate the need for live volunteers by using patches of synthetic skin made with a 3D printer.
“Each patch of gelatin-like hydrogel comes complete with tiny passageways that can be filled with flowing blood” from a chicken, sheep, or cow, they explained, and proof-of-concept testing showed that mosquitoes would feed on hydrogels without any repellent and stay away from those treated with a repellent.
To conduct the feeding tests, the blood-infused hydrogels are placed in a clear plastic box that is surrounded by cameras.
A bunch of mosquitoes are then tossed in the box and the cameras record all their insect activities: how often they land at each location, how long they stay, whether or not they bite, how long they feed, etc. Humans don’t have to watch and don’t have to be food sources.
Humans don’t have to be food sources, and we just pictured the future of mosquito control. Imagine a dozen Arnold Schwarzenegger–style Terminators, covered in 3D-printed skin, walking through your neighborhood in the summer while wearing sweat-soaked, brightly colored clothing. The mosquitoes wouldn’t be able to stay away, but guess what? They’re feeding off robots with nonhuman skin and nonhuman blood, so we win. It’s good to have a cerebral cortex.
Getting medieval on brain surgery
Let’s get one thing clear: The so-called “Dark Ages” were not nearly as dark as they’re made out to be. For one thing, there’s a world beyond Western Europe. The Roman Empire didn’t collapse everywhere. But even in Western Europe, the centuries between the fall of Rome and the Renaissance were hardly lacking in cultural development.
That said, we wouldn’t want to be in the position of the seventh-century noblewoman whose remains were recently uncovered in a Byzantine fortress in central Italy with multiple cross-shaped incisions in her skull. Yes, this unfortunate woman underwent at least two brain surgeries.
Then again, maybe not. Nothing like it had been discovered at the site, and while the markings – signs of a procedure called trepanation – can be surgical in nature, there are other explanations. For example, the Avar people practiced ritual trepanation during the same time period, but they were hundreds of miles away in the Carpathian mountains, and there was no evidence to support that a different form of ritualistic trepanation ever took place in Byzantine-era Italy.
The investigators then moved on to a form of judicial punishment called decalvatio, which involves mutilation by scalping. Look, the Dark Ages weren’t dark, but no one said they were fun. Anyway, this was discarded, since decalvatio was only meted out to soldiers who deserted the battlefield.
That brings us back to surgery. While one of the trepanations was fully engraved into her skull, indicating that the woman died soon after the surgery, she also bore indications of a healed trepanation. A 50% success rate isn’t terrible for our medieval surgeon. Sure, the Incas managed 80%, but even during the Civil War brain surgery only had a 50% success rate. And that’s the end of the story, nothing more to say about our medieval Italian woman.
Nope. Nothing at all.
Fine. While a surgical procedure was deemed most likely, the study investigators found no direct evidence of a medical condition. No trauma, no tumor, nothing. Just a couple of suggestions of “a systemic pathological condition,” they said. Okay, we swear, it really wasn’t that bad in the Middle [Editor’s note: Approximately 5,000 more words on medieval culture not included. This is a medical column, thank you very much.]
We’re gonna need a bigger meth lab
In case you’ve been living under a rock for the past 15 years, the TV show “Breaking Bad” details the spiraling rise and downfall of a high school chemistry teacher who, after developing a case of terminal lung cancer, starts producing methamphetamine to provide for his family in response to the steep cost of treatment for his cancer.
Meanwhile, here in 2023 in the real world, we have Paul Davis, a retired physician in Ohio, who’s being forced to choose between an expensive cancer treatment and bankrupting his family, since Medicare’s decided it doesn’t want to cover the cost. Hey, we’ve seen this one before!
A bit of backstory: In November 2019, Dr. Davis was diagnosed with uveal melanoma, a very rare type of cancer that affects eye tissue. The news got worse in 2022 when the cancer spread to his liver, a move which typically proves fatal within a year. However, in a stroke of great news, the Food and Drug Administration approved the drug Kimmtrak earlier that year, which could be used to treat his cancer. Not cure, of course, but it would give him more time.
His initial treatments with the drug went fine and were covered, but when he transferred his care from a hospital in Columbus to one closer to home, big problem. Medicare decided it didn’t like that hospital and abruptly cut off coverage, denying the local hospital’s claims. That leaves Dr. Davis on the hook for his cancer treatment, and it’s what you might call expensive. Expensive to the tune of $50,000.
A week.
Apparently the coding the local hospital submitted was wrong, indicating that Dr. Davis was receiving Kimmtrak for a type of cancer that the FDA hadn’t approved the drug for. So until the government bureaucracy works itself out, his treatment is on hold, leaving all his faith in Medicare working quickly to rectify its mistake. If it can rectify its mistake. We’re not hopeful.
And in case you were wondering, if Dr. Davis wanted to go full Walter White, the average street price of meth is about $20-$60 per gram, so to pay for his treatment, he’d need to make at least a kilogram of meth every week. That’s, uh, quite a lot of illegal drug, or what we here at the LOTME office would call a fun Saturday night.
When you give a mouse a movie
Researchers have been successfully testing Alzheimer drugs on mice for years, but none of the drugs has proved successful in humans. Recent work, however, might have found the missing link, and it’s a combination no one ever thought of before: mice and movies.
Turns out that Orson Welles’ 1958 film noir classic “Touch of Evil” tapped a part of the mouse brain that has been overlooked: the hippocampus, which is crucial for learning and memory. Previous researchers thought it was just used as a kind of GPS system, but that’s only partially true.
Not only did the mice choose to pay attention to the movie clip, but the hippocampus responded to the visual stimuli only when the rodents saw the scenes from the clip later in the order that they were presented and not in a scrambled order. These findings represent a “major paradigm shift” in studying mouse recall, Mayank Mehta, PhD, of the University of California, Los Angeles, said in a statement from the school.
This breakthrough could run parallel to Alzheimer’s patients struggling with similar defects. “Selective and episodic activation of the mouse hippocampus using a human movie opens up the possibility of directly testing human episodic memory disorders and therapies using mouse neurons, a major step forward,” said coauthor Chinmay Purandare, PhD, who is now at the University of California, San Francisco.
Who would have thought that a classic film would help advance Alzheimer research?
A less human way to study mosquitoes
We here at LOTME have a history with mosquitoes. We know they don’t like us, and they know that we don’t like them. Trust us, they know. So when humans gain a little ground in the war against the buzzy little bloodsuckers, we want to share the joy.
To know the enemy, scientists have to study the enemy, but there is a problem. “Many mosquito experiments still rely on human volunteers and animal subjects,” bioengineering graduate student Kevin Janson, said in a statement from Rice University. Most people don’t like being bitten by mosquitoes, so that kind of testing can be expensive.
Is there a way to automate the collection and processing of mosquito behavior data using inexpensive cameras and machine-learning software? We’re glad you asked, because Mr. Janson and the research team, which includes bioengineers from Rice and tropical medicine experts from Tulane University, have managed to eliminate the need for live volunteers by using patches of synthetic skin made with a 3D printer.
“Each patch of gelatin-like hydrogel comes complete with tiny passageways that can be filled with flowing blood” from a chicken, sheep, or cow, they explained, and proof-of-concept testing showed that mosquitoes would feed on hydrogels without any repellent and stay away from those treated with a repellent.
To conduct the feeding tests, the blood-infused hydrogels are placed in a clear plastic box that is surrounded by cameras.
A bunch of mosquitoes are then tossed in the box and the cameras record all their insect activities: how often they land at each location, how long they stay, whether or not they bite, how long they feed, etc. Humans don’t have to watch and don’t have to be food sources.
Humans don’t have to be food sources, and we just pictured the future of mosquito control. Imagine a dozen Arnold Schwarzenegger–style Terminators, covered in 3D-printed skin, walking through your neighborhood in the summer while wearing sweat-soaked, brightly colored clothing. The mosquitoes wouldn’t be able to stay away, but guess what? They’re feeding off robots with nonhuman skin and nonhuman blood, so we win. It’s good to have a cerebral cortex.
Getting medieval on brain surgery
Let’s get one thing clear: The so-called “Dark Ages” were not nearly as dark as they’re made out to be. For one thing, there’s a world beyond Western Europe. The Roman Empire didn’t collapse everywhere. But even in Western Europe, the centuries between the fall of Rome and the Renaissance were hardly lacking in cultural development.
That said, we wouldn’t want to be in the position of the seventh-century noblewoman whose remains were recently uncovered in a Byzantine fortress in central Italy with multiple cross-shaped incisions in her skull. Yes, this unfortunate woman underwent at least two brain surgeries.
Then again, maybe not. Nothing like it had been discovered at the site, and while the markings – signs of a procedure called trepanation – can be surgical in nature, there are other explanations. For example, the Avar people practiced ritual trepanation during the same time period, but they were hundreds of miles away in the Carpathian mountains, and there was no evidence to support that a different form of ritualistic trepanation ever took place in Byzantine-era Italy.
The investigators then moved on to a form of judicial punishment called decalvatio, which involves mutilation by scalping. Look, the Dark Ages weren’t dark, but no one said they were fun. Anyway, this was discarded, since decalvatio was only meted out to soldiers who deserted the battlefield.
That brings us back to surgery. While one of the trepanations was fully engraved into her skull, indicating that the woman died soon after the surgery, she also bore indications of a healed trepanation. A 50% success rate isn’t terrible for our medieval surgeon. Sure, the Incas managed 80%, but even during the Civil War brain surgery only had a 50% success rate. And that’s the end of the story, nothing more to say about our medieval Italian woman.
Nope. Nothing at all.
Fine. While a surgical procedure was deemed most likely, the study investigators found no direct evidence of a medical condition. No trauma, no tumor, nothing. Just a couple of suggestions of “a systemic pathological condition,” they said. Okay, we swear, it really wasn’t that bad in the Middle [Editor’s note: Approximately 5,000 more words on medieval culture not included. This is a medical column, thank you very much.]
PsA prediction tool approaches clinical utility
Easily collected variables establish risk
A new tool for predicting which patients with psoriasis will develop psoriatic arthritis (PsA) is showing promise for such clinical applications as early treatment in those at risk or trials to prevent PsA, according to a summary of progress at the annual meeting of the Canadian Rheumatology Association.
Based on current levels of sensitivity and specificity, psoriasis “can be predicted with reasonable accuracy,” reported Lihi Eder, MD, PhD, director of research in the rheumatology division at the University of Toronto.
The predictive method, called PRESTO (Prediction of Psoriatic Arthritis Tool), is based on variables readily available in clinical practice, according to Dr. Eder. Once values are assigned to the risk factors, the risk of PsA over a 1-year or 5-year time frame can be estimated with a calculator.
She called PRESTO the “first clinical tool for predicting PsA among psoriasis patients.”
The work on this tool began in 2006 when the International Psoriasis and Arthritis Research Team (IPART) initiated a prospectively collected cohort of psoriasis patients. To be enrolled, patients had to be free of signs and symptoms of arthritis upon examination by a rheumatologist. They were then invited to return annually for follow-up that again included screening for joint involvement by a rheumatologist.
At baseline and at follow-up evaluations, 13 predictors were evaluated. These involved psoriasis characteristics, such as nail pitting; symptoms, such as stiffness; comorbidities, such as additional inflammatory diseases; and laboratory values, such as upregulated markers of inflammation.
Symptoms and signs used to predict PsA
Dr. Eder and her colleagues applied regression models to select an optimal combination of variables weighted for predictive value. Variables offering predictive value included higher PASI (Psoriasis Area and Severity Index), greater fatigue score as measured by FACIT (Functional Assessment of Chronic Illness Therapy) score, greater morning stiffness, and greater pain.
When applied to 635 patients in the IPART cohort, in which there were 51 incident PsA cases over 1 year and 75 incident cases over 5 years, the area under the curve (AUC) for PRESTO at the cutoffs studied was 72% for the 1-year time window and 75% for the 5-year time window.
These levels are associated with adequate accuracy, according to Dr. Eder, who explained that “an AUC greater than 70% is considered reasonable” for clinical applicability.
Moreover, the cutoffs can be adjusted for the specific purpose of the predictive tool. For example, to screen patients for risk, lower cutoffs could be employed to increase sensitivity. In order to select patients for a clinical trial to prevent PsA, higher cutoffs could be employed to increase specificity.
But sensitivities and specificities move in opposite directions when cutoffs are adjusted. Showing data from the 5-year prediction model, Dr. Eder reported that specificities climbed from about 58% to 97% as cutoffs were increased. The sensitivities with these adjustments fell from 79% to 14%.
In general, Dr. Eder said there was “excellent calibration” for the cutoffs employed when they compared the predicted and observed rates of PsA according to quintile of predictive probability. The differences were particularly minor over a 1-year time period. Over the 5-year period, observed rates were somewhat higher than predicted in the fourth and fifth quintile, but, again, this discrepancy could be modified for specific applications with cutoff adjustments.
Validation studies are planned
Even though psoriasis patients in IPART represents one of the largest cohorts of prospectively collected psoriasis patients, Dr. Eder acknowledged that the sample size would be considered “moderate” for developing a predictive model. However, the fact that the data were collected prospectively using standardized methodology strengthens the findings and provides the basis for the next step.
“Validation studies are planned with external cohorts,” said Dr. Eder, who indicated that a viable tool for identifying psoriasis patients at risk for PsA is likely. Even if it is not employed routinely in its current form at the level of individual patient care, she predicted that it will have value at a research level for understanding the relationship of psoriasis to PsA.
Christopher T. Ritchlin, MD, a professor and researcher at the University of Rochester (N.Y.), agreed that PRESTO has important potential as a clinical tool. Dr. Ritchlin has been involved in the development of PRESTO but was not involved in the presentation made at the CRA annual meeting.
“The PRESTO tool has the ability to predict the 2- and 5-year risk of developing psoriatic arthritis, which is an important advance if confirmed,” he said in an interview. He pointed out that approximately 25%-30% who develop psoriasis will go on to develop PsA but until now there has been no way to identify them.
“This tool may provide a pathway to early intervention,” he said.
Dr. Eder has financial relationships with AbbVie, Eli Lilly, Fresenius Kabi, Janssen, Novartis, Pfizer, Sandoz, and UCB. Dr. Ritchlin has financial relationships with many of the same companies.
Easily collected variables establish risk
Easily collected variables establish risk
A new tool for predicting which patients with psoriasis will develop psoriatic arthritis (PsA) is showing promise for such clinical applications as early treatment in those at risk or trials to prevent PsA, according to a summary of progress at the annual meeting of the Canadian Rheumatology Association.
Based on current levels of sensitivity and specificity, psoriasis “can be predicted with reasonable accuracy,” reported Lihi Eder, MD, PhD, director of research in the rheumatology division at the University of Toronto.
The predictive method, called PRESTO (Prediction of Psoriatic Arthritis Tool), is based on variables readily available in clinical practice, according to Dr. Eder. Once values are assigned to the risk factors, the risk of PsA over a 1-year or 5-year time frame can be estimated with a calculator.
She called PRESTO the “first clinical tool for predicting PsA among psoriasis patients.”
The work on this tool began in 2006 when the International Psoriasis and Arthritis Research Team (IPART) initiated a prospectively collected cohort of psoriasis patients. To be enrolled, patients had to be free of signs and symptoms of arthritis upon examination by a rheumatologist. They were then invited to return annually for follow-up that again included screening for joint involvement by a rheumatologist.
At baseline and at follow-up evaluations, 13 predictors were evaluated. These involved psoriasis characteristics, such as nail pitting; symptoms, such as stiffness; comorbidities, such as additional inflammatory diseases; and laboratory values, such as upregulated markers of inflammation.
Symptoms and signs used to predict PsA
Dr. Eder and her colleagues applied regression models to select an optimal combination of variables weighted for predictive value. Variables offering predictive value included higher PASI (Psoriasis Area and Severity Index), greater fatigue score as measured by FACIT (Functional Assessment of Chronic Illness Therapy) score, greater morning stiffness, and greater pain.
When applied to 635 patients in the IPART cohort, in which there were 51 incident PsA cases over 1 year and 75 incident cases over 5 years, the area under the curve (AUC) for PRESTO at the cutoffs studied was 72% for the 1-year time window and 75% for the 5-year time window.
These levels are associated with adequate accuracy, according to Dr. Eder, who explained that “an AUC greater than 70% is considered reasonable” for clinical applicability.
Moreover, the cutoffs can be adjusted for the specific purpose of the predictive tool. For example, to screen patients for risk, lower cutoffs could be employed to increase sensitivity. In order to select patients for a clinical trial to prevent PsA, higher cutoffs could be employed to increase specificity.
But sensitivities and specificities move in opposite directions when cutoffs are adjusted. Showing data from the 5-year prediction model, Dr. Eder reported that specificities climbed from about 58% to 97% as cutoffs were increased. The sensitivities with these adjustments fell from 79% to 14%.
In general, Dr. Eder said there was “excellent calibration” for the cutoffs employed when they compared the predicted and observed rates of PsA according to quintile of predictive probability. The differences were particularly minor over a 1-year time period. Over the 5-year period, observed rates were somewhat higher than predicted in the fourth and fifth quintile, but, again, this discrepancy could be modified for specific applications with cutoff adjustments.
Validation studies are planned
Even though psoriasis patients in IPART represents one of the largest cohorts of prospectively collected psoriasis patients, Dr. Eder acknowledged that the sample size would be considered “moderate” for developing a predictive model. However, the fact that the data were collected prospectively using standardized methodology strengthens the findings and provides the basis for the next step.
“Validation studies are planned with external cohorts,” said Dr. Eder, who indicated that a viable tool for identifying psoriasis patients at risk for PsA is likely. Even if it is not employed routinely in its current form at the level of individual patient care, she predicted that it will have value at a research level for understanding the relationship of psoriasis to PsA.
Christopher T. Ritchlin, MD, a professor and researcher at the University of Rochester (N.Y.), agreed that PRESTO has important potential as a clinical tool. Dr. Ritchlin has been involved in the development of PRESTO but was not involved in the presentation made at the CRA annual meeting.
“The PRESTO tool has the ability to predict the 2- and 5-year risk of developing psoriatic arthritis, which is an important advance if confirmed,” he said in an interview. He pointed out that approximately 25%-30% who develop psoriasis will go on to develop PsA but until now there has been no way to identify them.
“This tool may provide a pathway to early intervention,” he said.
Dr. Eder has financial relationships with AbbVie, Eli Lilly, Fresenius Kabi, Janssen, Novartis, Pfizer, Sandoz, and UCB. Dr. Ritchlin has financial relationships with many of the same companies.
A new tool for predicting which patients with psoriasis will develop psoriatic arthritis (PsA) is showing promise for such clinical applications as early treatment in those at risk or trials to prevent PsA, according to a summary of progress at the annual meeting of the Canadian Rheumatology Association.
Based on current levels of sensitivity and specificity, psoriasis “can be predicted with reasonable accuracy,” reported Lihi Eder, MD, PhD, director of research in the rheumatology division at the University of Toronto.
The predictive method, called PRESTO (Prediction of Psoriatic Arthritis Tool), is based on variables readily available in clinical practice, according to Dr. Eder. Once values are assigned to the risk factors, the risk of PsA over a 1-year or 5-year time frame can be estimated with a calculator.
She called PRESTO the “first clinical tool for predicting PsA among psoriasis patients.”
The work on this tool began in 2006 when the International Psoriasis and Arthritis Research Team (IPART) initiated a prospectively collected cohort of psoriasis patients. To be enrolled, patients had to be free of signs and symptoms of arthritis upon examination by a rheumatologist. They were then invited to return annually for follow-up that again included screening for joint involvement by a rheumatologist.
At baseline and at follow-up evaluations, 13 predictors were evaluated. These involved psoriasis characteristics, such as nail pitting; symptoms, such as stiffness; comorbidities, such as additional inflammatory diseases; and laboratory values, such as upregulated markers of inflammation.
Symptoms and signs used to predict PsA
Dr. Eder and her colleagues applied regression models to select an optimal combination of variables weighted for predictive value. Variables offering predictive value included higher PASI (Psoriasis Area and Severity Index), greater fatigue score as measured by FACIT (Functional Assessment of Chronic Illness Therapy) score, greater morning stiffness, and greater pain.
When applied to 635 patients in the IPART cohort, in which there were 51 incident PsA cases over 1 year and 75 incident cases over 5 years, the area under the curve (AUC) for PRESTO at the cutoffs studied was 72% for the 1-year time window and 75% for the 5-year time window.
These levels are associated with adequate accuracy, according to Dr. Eder, who explained that “an AUC greater than 70% is considered reasonable” for clinical applicability.
Moreover, the cutoffs can be adjusted for the specific purpose of the predictive tool. For example, to screen patients for risk, lower cutoffs could be employed to increase sensitivity. In order to select patients for a clinical trial to prevent PsA, higher cutoffs could be employed to increase specificity.
But sensitivities and specificities move in opposite directions when cutoffs are adjusted. Showing data from the 5-year prediction model, Dr. Eder reported that specificities climbed from about 58% to 97% as cutoffs were increased. The sensitivities with these adjustments fell from 79% to 14%.
In general, Dr. Eder said there was “excellent calibration” for the cutoffs employed when they compared the predicted and observed rates of PsA according to quintile of predictive probability. The differences were particularly minor over a 1-year time period. Over the 5-year period, observed rates were somewhat higher than predicted in the fourth and fifth quintile, but, again, this discrepancy could be modified for specific applications with cutoff adjustments.
Validation studies are planned
Even though psoriasis patients in IPART represents one of the largest cohorts of prospectively collected psoriasis patients, Dr. Eder acknowledged that the sample size would be considered “moderate” for developing a predictive model. However, the fact that the data were collected prospectively using standardized methodology strengthens the findings and provides the basis for the next step.
“Validation studies are planned with external cohorts,” said Dr. Eder, who indicated that a viable tool for identifying psoriasis patients at risk for PsA is likely. Even if it is not employed routinely in its current form at the level of individual patient care, she predicted that it will have value at a research level for understanding the relationship of psoriasis to PsA.
Christopher T. Ritchlin, MD, a professor and researcher at the University of Rochester (N.Y.), agreed that PRESTO has important potential as a clinical tool. Dr. Ritchlin has been involved in the development of PRESTO but was not involved in the presentation made at the CRA annual meeting.
“The PRESTO tool has the ability to predict the 2- and 5-year risk of developing psoriatic arthritis, which is an important advance if confirmed,” he said in an interview. He pointed out that approximately 25%-30% who develop psoriasis will go on to develop PsA but until now there has been no way to identify them.
“This tool may provide a pathway to early intervention,” he said.
Dr. Eder has financial relationships with AbbVie, Eli Lilly, Fresenius Kabi, Janssen, Novartis, Pfizer, Sandoz, and UCB. Dr. Ritchlin has financial relationships with many of the same companies.
FROM CRA 2023
Saying goodbye: How to transition teens to adult medical care
However, many clinicians feel insufficiently prepared to provide comprehensive transition services. This can result in the actual handoff or transfer into adult care being abrupt, incomplete, or outright unsuccessful. By following the recommended best practices of transitions, providers of pediatric care can ensure that this challenging goodbye prepares everyone for the next steps ahead.
Using a structured transition process
In 2011, a health care transition clinical report based on expert opinion and practice consensus and endorsed by the American Academy of Pediatrics, American Academy of Family Physicians, and American College of Physicians – Society of Internal Medicine was released. This report provided a decision-making algorithm for “practice-based implementation of transition for all youth beginning in early adolescence.”
The Got Transition organization, funded by the Maternal Child Health Bureau and Health Resources and Services Administration, provides web-based information and materials for health care providers and families to establish a smooth and successful transition. At the center of these recommendations are the Six Core Elements of Health Care Transition – the essential components of a structured transition process: 1) transition policy/guide; 2) tracking and monitoring; 3) readiness; 4) planning; 5) transfer of care, and 6) transition completion.
This transition process should start early in adolescence, preferably by age 12-14 years, to give adequate time to progress successfully through these elements and improve the likelihood of a smooth, final transfer into the care of an adult clinician.
Preparing your patients for transfer
Despite the availability of these recommendations, national surveys show that the overwhelming majority of adolescents with and without special health care needs report not receiving transition services. Lack of time, resources, interest, and patients being lost to care during adolescence all contribute to this deficit in care. Without transition preparation, the actual handoff or transfer to adult care can be difficult for adolescents, caregivers, and clinicians alike. Adolescents and caregivers may feel a sense of abandonment or have inadequate health knowledge/literacy, pediatric clinicians may fear that the patient is not ready for the expected independence, and adult clinicians face numerous challenges integrating these young patients into their practice.
A structured transition process can help the family and clinicians know what to expect during the transfer of care. Pediatric clinicians can gradually move from a pediatric model of care, in which the caregiver is the center of communication, to an adult model, putting the patient at the center. By encouraging the adolescent to be the direct communicator, the pediatric clinician can promote independence and assess health knowledge, allowing for education where gaps exist.
Assisting the patient in identifying and even meeting the adult clinician well ahead of the final transfer date can also make the process less daunting for the adolescent.
Adult clinicians should consider allowing more time for the first visit with a new young adult patient and welcome caregiver input early in the transfer process, particularly for patients with a chronic disease. By engaging patients and families in an intentional, gradual transition process with an expected outcome, all those involved will be more prepared for the final handoff.
Utilizing transition tools and engaging the adolescent
Numerous tools can assist in the preparation for transfer to adult care. These include transition summaries and emergency plans, which contain essential information such as current medical problems, allergies, medications, prior procedures and treatments, and sick day plans. Such tools can also be built into electronic medical records for easy modification and updating. They can be used as methods to engage and teach adolescents about their disease history and current regimen and can contain essential components for information handoff at the time of transfer to adult care. If the patient carries a rare diagnosis, or one that has historically been associated with lower survival to adulthood, these transfer documents can also include summary information about disease states and contact information for pediatric specialty clinicians.
Adolescent engagement in their health care during the time of transition can also be prompted through the use of patient portals within an electronic health record. Such portals put health information directly at the adolescent’s fingertips, provide them with an outlet for communication with their clinicians, and give reminders regarding health maintenance.
Completing the transfer: The final handoff
The best and most recommended means of relaying information at the time of transfer to adult care is a direct, verbal handoff between clinicians. This direct handoff has several goals:
(1) To ensure the patient has scheduled or attended the first appointment with the adult clinician
(2) To ensure record transfer has occurred successfully
(3) To answer any questions the receiving clinician may have about prior or ongoing care.
(4) To offer the adult clinician ongoing access to the pediatric clinician as an “expert” resource for additional questions.
By remaining available as a resource, the pediatric clinician can alleviate concerns for both the patient and caregiver as well as the receiving adult clinician.
As valuable as verbal handoffs can be, they are not always possible due to patients not having selected an adult clinician prior to leaving the pediatric clinician, an inability to reach the receiving clinician, and/or time limitations. Many of these barriers can be alleviated by early discussions of transitions of care as well as utilization of structured documentation tools as noted above.
It is also recommended that the pediatric clinician follows up with the patient and/or caregiver several months after the transfer is complete. This allows for the adolescent and/or the caregiver to reflect on the transition process and provide feedback to the pediatric clinicians and their practice for ongoing process improvement.
Reflection as a pediatrician
Ideally, all transition steps occur for the adolescent; in our opinion, a crucial component is to prepare the adolescent patient for the change from a pediatric to adult model of care, in which they are independent in their health communication and decision-making. By engaging adolescents to understand their health, how to maintain it, and when to seek care, we empower them to advocate for their own health as young adults. With appropriate health knowledge and literacy, adolescents are more likely to actively engage with their health care providers and make healthy lifestyle choices. So though saying goodbye may still be difficult, it can be done with the confidence that the patients will continue to get the care they need as they transition into adulthood.
Dr. Kim is assistant clinical professor, department of pediatrics, University of California, San Diego. Dr. Mennito is associate professor of pediatrics and internal medicine, Medical University of South Carolina, Charleston, S.C. Dr. Kim and Dr. Mennito have disclosed no relevant financial relationships. A version of this article originally appeared on Medscape.com.
However, many clinicians feel insufficiently prepared to provide comprehensive transition services. This can result in the actual handoff or transfer into adult care being abrupt, incomplete, or outright unsuccessful. By following the recommended best practices of transitions, providers of pediatric care can ensure that this challenging goodbye prepares everyone for the next steps ahead.
Using a structured transition process
In 2011, a health care transition clinical report based on expert opinion and practice consensus and endorsed by the American Academy of Pediatrics, American Academy of Family Physicians, and American College of Physicians – Society of Internal Medicine was released. This report provided a decision-making algorithm for “practice-based implementation of transition for all youth beginning in early adolescence.”
The Got Transition organization, funded by the Maternal Child Health Bureau and Health Resources and Services Administration, provides web-based information and materials for health care providers and families to establish a smooth and successful transition. At the center of these recommendations are the Six Core Elements of Health Care Transition – the essential components of a structured transition process: 1) transition policy/guide; 2) tracking and monitoring; 3) readiness; 4) planning; 5) transfer of care, and 6) transition completion.
This transition process should start early in adolescence, preferably by age 12-14 years, to give adequate time to progress successfully through these elements and improve the likelihood of a smooth, final transfer into the care of an adult clinician.
Preparing your patients for transfer
Despite the availability of these recommendations, national surveys show that the overwhelming majority of adolescents with and without special health care needs report not receiving transition services. Lack of time, resources, interest, and patients being lost to care during adolescence all contribute to this deficit in care. Without transition preparation, the actual handoff or transfer to adult care can be difficult for adolescents, caregivers, and clinicians alike. Adolescents and caregivers may feel a sense of abandonment or have inadequate health knowledge/literacy, pediatric clinicians may fear that the patient is not ready for the expected independence, and adult clinicians face numerous challenges integrating these young patients into their practice.
A structured transition process can help the family and clinicians know what to expect during the transfer of care. Pediatric clinicians can gradually move from a pediatric model of care, in which the caregiver is the center of communication, to an adult model, putting the patient at the center. By encouraging the adolescent to be the direct communicator, the pediatric clinician can promote independence and assess health knowledge, allowing for education where gaps exist.
Assisting the patient in identifying and even meeting the adult clinician well ahead of the final transfer date can also make the process less daunting for the adolescent.
Adult clinicians should consider allowing more time for the first visit with a new young adult patient and welcome caregiver input early in the transfer process, particularly for patients with a chronic disease. By engaging patients and families in an intentional, gradual transition process with an expected outcome, all those involved will be more prepared for the final handoff.
Utilizing transition tools and engaging the adolescent
Numerous tools can assist in the preparation for transfer to adult care. These include transition summaries and emergency plans, which contain essential information such as current medical problems, allergies, medications, prior procedures and treatments, and sick day plans. Such tools can also be built into electronic medical records for easy modification and updating. They can be used as methods to engage and teach adolescents about their disease history and current regimen and can contain essential components for information handoff at the time of transfer to adult care. If the patient carries a rare diagnosis, or one that has historically been associated with lower survival to adulthood, these transfer documents can also include summary information about disease states and contact information for pediatric specialty clinicians.
Adolescent engagement in their health care during the time of transition can also be prompted through the use of patient portals within an electronic health record. Such portals put health information directly at the adolescent’s fingertips, provide them with an outlet for communication with their clinicians, and give reminders regarding health maintenance.
Completing the transfer: The final handoff
The best and most recommended means of relaying information at the time of transfer to adult care is a direct, verbal handoff between clinicians. This direct handoff has several goals:
(1) To ensure the patient has scheduled or attended the first appointment with the adult clinician
(2) To ensure record transfer has occurred successfully
(3) To answer any questions the receiving clinician may have about prior or ongoing care.
(4) To offer the adult clinician ongoing access to the pediatric clinician as an “expert” resource for additional questions.
By remaining available as a resource, the pediatric clinician can alleviate concerns for both the patient and caregiver as well as the receiving adult clinician.
As valuable as verbal handoffs can be, they are not always possible due to patients not having selected an adult clinician prior to leaving the pediatric clinician, an inability to reach the receiving clinician, and/or time limitations. Many of these barriers can be alleviated by early discussions of transitions of care as well as utilization of structured documentation tools as noted above.
It is also recommended that the pediatric clinician follows up with the patient and/or caregiver several months after the transfer is complete. This allows for the adolescent and/or the caregiver to reflect on the transition process and provide feedback to the pediatric clinicians and their practice for ongoing process improvement.
Reflection as a pediatrician
Ideally, all transition steps occur for the adolescent; in our opinion, a crucial component is to prepare the adolescent patient for the change from a pediatric to adult model of care, in which they are independent in their health communication and decision-making. By engaging adolescents to understand their health, how to maintain it, and when to seek care, we empower them to advocate for their own health as young adults. With appropriate health knowledge and literacy, adolescents are more likely to actively engage with their health care providers and make healthy lifestyle choices. So though saying goodbye may still be difficult, it can be done with the confidence that the patients will continue to get the care they need as they transition into adulthood.
Dr. Kim is assistant clinical professor, department of pediatrics, University of California, San Diego. Dr. Mennito is associate professor of pediatrics and internal medicine, Medical University of South Carolina, Charleston, S.C. Dr. Kim and Dr. Mennito have disclosed no relevant financial relationships. A version of this article originally appeared on Medscape.com.
However, many clinicians feel insufficiently prepared to provide comprehensive transition services. This can result in the actual handoff or transfer into adult care being abrupt, incomplete, or outright unsuccessful. By following the recommended best practices of transitions, providers of pediatric care can ensure that this challenging goodbye prepares everyone for the next steps ahead.
Using a structured transition process
In 2011, a health care transition clinical report based on expert opinion and practice consensus and endorsed by the American Academy of Pediatrics, American Academy of Family Physicians, and American College of Physicians – Society of Internal Medicine was released. This report provided a decision-making algorithm for “practice-based implementation of transition for all youth beginning in early adolescence.”
The Got Transition organization, funded by the Maternal Child Health Bureau and Health Resources and Services Administration, provides web-based information and materials for health care providers and families to establish a smooth and successful transition. At the center of these recommendations are the Six Core Elements of Health Care Transition – the essential components of a structured transition process: 1) transition policy/guide; 2) tracking and monitoring; 3) readiness; 4) planning; 5) transfer of care, and 6) transition completion.
This transition process should start early in adolescence, preferably by age 12-14 years, to give adequate time to progress successfully through these elements and improve the likelihood of a smooth, final transfer into the care of an adult clinician.
Preparing your patients for transfer
Despite the availability of these recommendations, national surveys show that the overwhelming majority of adolescents with and without special health care needs report not receiving transition services. Lack of time, resources, interest, and patients being lost to care during adolescence all contribute to this deficit in care. Without transition preparation, the actual handoff or transfer to adult care can be difficult for adolescents, caregivers, and clinicians alike. Adolescents and caregivers may feel a sense of abandonment or have inadequate health knowledge/literacy, pediatric clinicians may fear that the patient is not ready for the expected independence, and adult clinicians face numerous challenges integrating these young patients into their practice.
A structured transition process can help the family and clinicians know what to expect during the transfer of care. Pediatric clinicians can gradually move from a pediatric model of care, in which the caregiver is the center of communication, to an adult model, putting the patient at the center. By encouraging the adolescent to be the direct communicator, the pediatric clinician can promote independence and assess health knowledge, allowing for education where gaps exist.
Assisting the patient in identifying and even meeting the adult clinician well ahead of the final transfer date can also make the process less daunting for the adolescent.
Adult clinicians should consider allowing more time for the first visit with a new young adult patient and welcome caregiver input early in the transfer process, particularly for patients with a chronic disease. By engaging patients and families in an intentional, gradual transition process with an expected outcome, all those involved will be more prepared for the final handoff.
Utilizing transition tools and engaging the adolescent
Numerous tools can assist in the preparation for transfer to adult care. These include transition summaries and emergency plans, which contain essential information such as current medical problems, allergies, medications, prior procedures and treatments, and sick day plans. Such tools can also be built into electronic medical records for easy modification and updating. They can be used as methods to engage and teach adolescents about their disease history and current regimen and can contain essential components for information handoff at the time of transfer to adult care. If the patient carries a rare diagnosis, or one that has historically been associated with lower survival to adulthood, these transfer documents can also include summary information about disease states and contact information for pediatric specialty clinicians.
Adolescent engagement in their health care during the time of transition can also be prompted through the use of patient portals within an electronic health record. Such portals put health information directly at the adolescent’s fingertips, provide them with an outlet for communication with their clinicians, and give reminders regarding health maintenance.
Completing the transfer: The final handoff
The best and most recommended means of relaying information at the time of transfer to adult care is a direct, verbal handoff between clinicians. This direct handoff has several goals:
(1) To ensure the patient has scheduled or attended the first appointment with the adult clinician
(2) To ensure record transfer has occurred successfully
(3) To answer any questions the receiving clinician may have about prior or ongoing care.
(4) To offer the adult clinician ongoing access to the pediatric clinician as an “expert” resource for additional questions.
By remaining available as a resource, the pediatric clinician can alleviate concerns for both the patient and caregiver as well as the receiving adult clinician.
As valuable as verbal handoffs can be, they are not always possible due to patients not having selected an adult clinician prior to leaving the pediatric clinician, an inability to reach the receiving clinician, and/or time limitations. Many of these barriers can be alleviated by early discussions of transitions of care as well as utilization of structured documentation tools as noted above.
It is also recommended that the pediatric clinician follows up with the patient and/or caregiver several months after the transfer is complete. This allows for the adolescent and/or the caregiver to reflect on the transition process and provide feedback to the pediatric clinicians and their practice for ongoing process improvement.
Reflection as a pediatrician
Ideally, all transition steps occur for the adolescent; in our opinion, a crucial component is to prepare the adolescent patient for the change from a pediatric to adult model of care, in which they are independent in their health communication and decision-making. By engaging adolescents to understand their health, how to maintain it, and when to seek care, we empower them to advocate for their own health as young adults. With appropriate health knowledge and literacy, adolescents are more likely to actively engage with their health care providers and make healthy lifestyle choices. So though saying goodbye may still be difficult, it can be done with the confidence that the patients will continue to get the care they need as they transition into adulthood.
Dr. Kim is assistant clinical professor, department of pediatrics, University of California, San Diego. Dr. Mennito is associate professor of pediatrics and internal medicine, Medical University of South Carolina, Charleston, S.C. Dr. Kim and Dr. Mennito have disclosed no relevant financial relationships. A version of this article originally appeared on Medscape.com.
Physicians don’t feel safe with some patients: Here’s how to reduce the danger
“I talked to him about whether he was okay seeing me and he said yes,” Dr. Cheng said. “But I remained vigilant and conscious of what the patient was doing the whole time so he couldn’t take advantage of the situation.”
Dr. Cheng never turned his back to the patient and even backed out of the exam room. That encounter passed without incident. However, a urologist Dr. Cheng knew from residency wasn’t so fortunate. Ronald Gilbert, MD, of Newport Beach, Calif., was shot and killed by a patient in his office. The patient blamed him for complications following prostate surgery 25 years earlier.
In 2022, a gunman in Tulsa, Okla., blamed his physician for pain from a recent back surgery and shot and killed him, another physician, and two others in a medical building before taking his own life.
Nearly 9 in 10 physicians reported in a recent Medscape poll that they had experienced one or more violent or potentially violent incidents in the past year. The most common patient behaviors were verbal abuse, getting angry and leaving, and behaving erratically.
About one in three respondents said that the patients threatened to harm them, and about one in five said that the patients became violent.
Experts say that many factors contribute to this potentially lethal situation: Health care services have become more impersonal, patients experience longer wait times, some abuse prescription drugs, mental health services are lacking, and security is poor or nonexistent at some health care facilities.
Violence against hospital workers has become so common that a bill was introduced in 2022 in Congress to better protect them. The Safety From Violence for Healthcare Employees Act includes stiffer penalties for acts involving the use of a dangerous weapon or committed during a public emergency and would also provide $25 million in grants to hospitals for programs aimed at reducing violent incidents in health care settings, including de-escalation training. The American Hospital Association and American College of Emergency Physicians support the bill, which is now before the House Judiciary Subcommittee on Crime, Terrorism, and Homeland Security.
The worst day of their lives
“You have people who already are having the worst day of their lives and feeling on edge. If they already have a short fuse or substance abuse issues, that can translate into agitation, violence, or aggression,” said Scott Zeller, MD, vice president of acute psychiatry at Vituity, a physician-owned multispecialty group that operates in several states.
Health care workers in psychiatric and substance abuse hospitals were 10 times more likely to experience nonfatal injuries by others in 2018 than were health care workers in ambulatory settings, according to an April 2020 Bureau of Labor Statistics report. In addition, health care workers were five times more likely to suffer a workplace violence injury than were workers overall in 2018.
Psychiatrists who responded to the poll were the specialists most likely to report that they encountered violent patients and potentially violent patients. “Historically, inpatient psychiatry, which requires more acute care and monitoring, is considered the most dangerous profession outside of the police,” said Dr. Zeller.
Emergency physicians have reported an uptick in violence from patients; 85% said in a survey by ACEP in 2022 that they believed the rate of violence in emergency departments has increased over the past 5 years, whereas 45% indicated that it has greatly increased.
Some doctors have been threatened with violence or actually killed by family members. Alex Skog, MD, president-elect of ACEP’s Oregon chapter, told HealthCare Dive that “a patient’s family member with a gun holster on his hip threatened to kill me and kill my entire family after I told his father that he needed to be admitted because he had coronavirus.”
“I’ve been scared for my safety as well as the safety of my family,” Dr. Skog said. “That was just not something that we were seeing 3, 4, or 5 years ago.”
Many patients are already upset by the time they see doctors, according to the poll.
“The most common reason patients are upset is that they’re already in a lot of pain, which can be expressed as anger, hostility, or aggression. They’re very anxious and afraid of what’s happening and may be thinking about the worst-case scenario – that a bump or lump is cancer,” Dr. Zeller said.
Patients may also get upset if they disagree with their doctors’ diagnosis or treatment plan or the doctor refuses to prescribe them the drugs or tests they want.
“One doctor commented recently: ‘After over 30 years in this business, I can say patients are worse now than at any point in my career. Entitled, demanding, obnoxious. Any denial is met with outrage and indignity, whether it’s an opioid request or a demand for MRI of something because they ‘want to know.’ ”
An orthopedic surgeon in Indiana lost his life after he refused to prescribe opioids to a patient. Her angry husband shot and killed the doctor in the parking lot only 2 hours after confronting him in his office.
Decreased physician-patient trust
“When doctors experience something frightening, they become more apprehensive in the future. There’s no doubt that after the first violent experience, they think of things differently,” said Dr. Zeller.
More than half of the doctors who reported experiencing at least one violent or potentially violent incident in the poll said they trusted patients less.
This diminished trust can negatively impact the physician-patient relationship, said the authors of a recent Health Affairs article.
“The more patients harm their health care providers, intentionally or unintentionally, the more difficult it will be for those providers to trust them, leading to yet another unfortunate pattern: physicians pulling back on some of the behaviors thought to be most trust-building, for example, talking about their personal lives, building rapport, displaying compassion, or giving out their personal cell phone numbers,” the article stated.
What doctors can do
Most doctors who experienced a violent or potentially violent incident said they had tried to defuse the situation and that they succeeded at least some of the time, the poll results show.
One of the best ways to defuse a situation is to be empathetic and show the person that you’re on their side and not the enemy, said Dr. Cheng,.
“Rather than making general statements like ‘I understand that you’re upset,’ it’s better to be specific about the reason the person is upset. For example: ‘I understand that you’re upset that the pharmacy didn’t fill your prescription’ or ‘I understand how you’re feeling about Doctor So-and-so, who didn’t treat you right,’ ” Dr. Cheng stated.
Dr. Zeller urged physicians to talk to patients about why they’re upset and how they can help them. That approach worked with a patient who was having a psychotic episode.
“I told the staff, who wanted to forcibly restrain him and inject him with medication, that I would talk to him. I asked the patient, who was screaming ‘ya ya ya ya,’ whether he would take his medication if I gave it to him and he said yes. When he was calm, he explained that he was screaming to stop the voices telling him to kill his parents. He then got the help he needed,” said Dr. Zeller.
Dr. Cheng was trained in de-escalation techniques as an Orange County reserve deputy sheriff. He and Dr. Zeller recommended that physicians and staff receive training in how to spot potentially violent behavior and defuse these situations before they escalate.
Dr. Cheng suggests looking at the person’s body language for signs of increasing agitation or tension, such as clenched fists, tense posture, tight jaw, or fidgeting that may be accompanied by shouting and/or verbal abuse.
Physicians also need to consider where they are physically in relation to patients they see. “You don’t want to be too close to the patient or stand in front of them, which can be seen as confrontational. Instead, stand or sit off to the side, and never block the door if the patient’s upset,” said Dr. Cheng.
He recommended that physician practices prepare for violent incidents by developing detailed plans, including how and when to escape, how to protect patients, and how to cooperate with law enforcement.
“If a violent incident is inescapable, physicians and staff must be ready to fight back with whatever tools they have available, which may include fire extinguishers, chairs, or scalpels,” said Dr. Cheng.
A version of this article originally appeared on Medscape.com.
“I talked to him about whether he was okay seeing me and he said yes,” Dr. Cheng said. “But I remained vigilant and conscious of what the patient was doing the whole time so he couldn’t take advantage of the situation.”
Dr. Cheng never turned his back to the patient and even backed out of the exam room. That encounter passed without incident. However, a urologist Dr. Cheng knew from residency wasn’t so fortunate. Ronald Gilbert, MD, of Newport Beach, Calif., was shot and killed by a patient in his office. The patient blamed him for complications following prostate surgery 25 years earlier.
In 2022, a gunman in Tulsa, Okla., blamed his physician for pain from a recent back surgery and shot and killed him, another physician, and two others in a medical building before taking his own life.
Nearly 9 in 10 physicians reported in a recent Medscape poll that they had experienced one or more violent or potentially violent incidents in the past year. The most common patient behaviors were verbal abuse, getting angry and leaving, and behaving erratically.
About one in three respondents said that the patients threatened to harm them, and about one in five said that the patients became violent.
Experts say that many factors contribute to this potentially lethal situation: Health care services have become more impersonal, patients experience longer wait times, some abuse prescription drugs, mental health services are lacking, and security is poor or nonexistent at some health care facilities.
Violence against hospital workers has become so common that a bill was introduced in 2022 in Congress to better protect them. The Safety From Violence for Healthcare Employees Act includes stiffer penalties for acts involving the use of a dangerous weapon or committed during a public emergency and would also provide $25 million in grants to hospitals for programs aimed at reducing violent incidents in health care settings, including de-escalation training. The American Hospital Association and American College of Emergency Physicians support the bill, which is now before the House Judiciary Subcommittee on Crime, Terrorism, and Homeland Security.
The worst day of their lives
“You have people who already are having the worst day of their lives and feeling on edge. If they already have a short fuse or substance abuse issues, that can translate into agitation, violence, or aggression,” said Scott Zeller, MD, vice president of acute psychiatry at Vituity, a physician-owned multispecialty group that operates in several states.
Health care workers in psychiatric and substance abuse hospitals were 10 times more likely to experience nonfatal injuries by others in 2018 than were health care workers in ambulatory settings, according to an April 2020 Bureau of Labor Statistics report. In addition, health care workers were five times more likely to suffer a workplace violence injury than were workers overall in 2018.
Psychiatrists who responded to the poll were the specialists most likely to report that they encountered violent patients and potentially violent patients. “Historically, inpatient psychiatry, which requires more acute care and monitoring, is considered the most dangerous profession outside of the police,” said Dr. Zeller.
Emergency physicians have reported an uptick in violence from patients; 85% said in a survey by ACEP in 2022 that they believed the rate of violence in emergency departments has increased over the past 5 years, whereas 45% indicated that it has greatly increased.
Some doctors have been threatened with violence or actually killed by family members. Alex Skog, MD, president-elect of ACEP’s Oregon chapter, told HealthCare Dive that “a patient’s family member with a gun holster on his hip threatened to kill me and kill my entire family after I told his father that he needed to be admitted because he had coronavirus.”
“I’ve been scared for my safety as well as the safety of my family,” Dr. Skog said. “That was just not something that we were seeing 3, 4, or 5 years ago.”
Many patients are already upset by the time they see doctors, according to the poll.
“The most common reason patients are upset is that they’re already in a lot of pain, which can be expressed as anger, hostility, or aggression. They’re very anxious and afraid of what’s happening and may be thinking about the worst-case scenario – that a bump or lump is cancer,” Dr. Zeller said.
Patients may also get upset if they disagree with their doctors’ diagnosis or treatment plan or the doctor refuses to prescribe them the drugs or tests they want.
“One doctor commented recently: ‘After over 30 years in this business, I can say patients are worse now than at any point in my career. Entitled, demanding, obnoxious. Any denial is met with outrage and indignity, whether it’s an opioid request or a demand for MRI of something because they ‘want to know.’ ”
An orthopedic surgeon in Indiana lost his life after he refused to prescribe opioids to a patient. Her angry husband shot and killed the doctor in the parking lot only 2 hours after confronting him in his office.
Decreased physician-patient trust
“When doctors experience something frightening, they become more apprehensive in the future. There’s no doubt that after the first violent experience, they think of things differently,” said Dr. Zeller.
More than half of the doctors who reported experiencing at least one violent or potentially violent incident in the poll said they trusted patients less.
This diminished trust can negatively impact the physician-patient relationship, said the authors of a recent Health Affairs article.
“The more patients harm their health care providers, intentionally or unintentionally, the more difficult it will be for those providers to trust them, leading to yet another unfortunate pattern: physicians pulling back on some of the behaviors thought to be most trust-building, for example, talking about their personal lives, building rapport, displaying compassion, or giving out their personal cell phone numbers,” the article stated.
What doctors can do
Most doctors who experienced a violent or potentially violent incident said they had tried to defuse the situation and that they succeeded at least some of the time, the poll results show.
One of the best ways to defuse a situation is to be empathetic and show the person that you’re on their side and not the enemy, said Dr. Cheng,.
“Rather than making general statements like ‘I understand that you’re upset,’ it’s better to be specific about the reason the person is upset. For example: ‘I understand that you’re upset that the pharmacy didn’t fill your prescription’ or ‘I understand how you’re feeling about Doctor So-and-so, who didn’t treat you right,’ ” Dr. Cheng stated.
Dr. Zeller urged physicians to talk to patients about why they’re upset and how they can help them. That approach worked with a patient who was having a psychotic episode.
“I told the staff, who wanted to forcibly restrain him and inject him with medication, that I would talk to him. I asked the patient, who was screaming ‘ya ya ya ya,’ whether he would take his medication if I gave it to him and he said yes. When he was calm, he explained that he was screaming to stop the voices telling him to kill his parents. He then got the help he needed,” said Dr. Zeller.
Dr. Cheng was trained in de-escalation techniques as an Orange County reserve deputy sheriff. He and Dr. Zeller recommended that physicians and staff receive training in how to spot potentially violent behavior and defuse these situations before they escalate.
Dr. Cheng suggests looking at the person’s body language for signs of increasing agitation or tension, such as clenched fists, tense posture, tight jaw, or fidgeting that may be accompanied by shouting and/or verbal abuse.
Physicians also need to consider where they are physically in relation to patients they see. “You don’t want to be too close to the patient or stand in front of them, which can be seen as confrontational. Instead, stand or sit off to the side, and never block the door if the patient’s upset,” said Dr. Cheng.
He recommended that physician practices prepare for violent incidents by developing detailed plans, including how and when to escape, how to protect patients, and how to cooperate with law enforcement.
“If a violent incident is inescapable, physicians and staff must be ready to fight back with whatever tools they have available, which may include fire extinguishers, chairs, or scalpels,” said Dr. Cheng.
A version of this article originally appeared on Medscape.com.
“I talked to him about whether he was okay seeing me and he said yes,” Dr. Cheng said. “But I remained vigilant and conscious of what the patient was doing the whole time so he couldn’t take advantage of the situation.”
Dr. Cheng never turned his back to the patient and even backed out of the exam room. That encounter passed without incident. However, a urologist Dr. Cheng knew from residency wasn’t so fortunate. Ronald Gilbert, MD, of Newport Beach, Calif., was shot and killed by a patient in his office. The patient blamed him for complications following prostate surgery 25 years earlier.
In 2022, a gunman in Tulsa, Okla., blamed his physician for pain from a recent back surgery and shot and killed him, another physician, and two others in a medical building before taking his own life.
Nearly 9 in 10 physicians reported in a recent Medscape poll that they had experienced one or more violent or potentially violent incidents in the past year. The most common patient behaviors were verbal abuse, getting angry and leaving, and behaving erratically.
About one in three respondents said that the patients threatened to harm them, and about one in five said that the patients became violent.
Experts say that many factors contribute to this potentially lethal situation: Health care services have become more impersonal, patients experience longer wait times, some abuse prescription drugs, mental health services are lacking, and security is poor or nonexistent at some health care facilities.
Violence against hospital workers has become so common that a bill was introduced in 2022 in Congress to better protect them. The Safety From Violence for Healthcare Employees Act includes stiffer penalties for acts involving the use of a dangerous weapon or committed during a public emergency and would also provide $25 million in grants to hospitals for programs aimed at reducing violent incidents in health care settings, including de-escalation training. The American Hospital Association and American College of Emergency Physicians support the bill, which is now before the House Judiciary Subcommittee on Crime, Terrorism, and Homeland Security.
The worst day of their lives
“You have people who already are having the worst day of their lives and feeling on edge. If they already have a short fuse or substance abuse issues, that can translate into agitation, violence, or aggression,” said Scott Zeller, MD, vice president of acute psychiatry at Vituity, a physician-owned multispecialty group that operates in several states.
Health care workers in psychiatric and substance abuse hospitals were 10 times more likely to experience nonfatal injuries by others in 2018 than were health care workers in ambulatory settings, according to an April 2020 Bureau of Labor Statistics report. In addition, health care workers were five times more likely to suffer a workplace violence injury than were workers overall in 2018.
Psychiatrists who responded to the poll were the specialists most likely to report that they encountered violent patients and potentially violent patients. “Historically, inpatient psychiatry, which requires more acute care and monitoring, is considered the most dangerous profession outside of the police,” said Dr. Zeller.
Emergency physicians have reported an uptick in violence from patients; 85% said in a survey by ACEP in 2022 that they believed the rate of violence in emergency departments has increased over the past 5 years, whereas 45% indicated that it has greatly increased.
Some doctors have been threatened with violence or actually killed by family members. Alex Skog, MD, president-elect of ACEP’s Oregon chapter, told HealthCare Dive that “a patient’s family member with a gun holster on his hip threatened to kill me and kill my entire family after I told his father that he needed to be admitted because he had coronavirus.”
“I’ve been scared for my safety as well as the safety of my family,” Dr. Skog said. “That was just not something that we were seeing 3, 4, or 5 years ago.”
Many patients are already upset by the time they see doctors, according to the poll.
“The most common reason patients are upset is that they’re already in a lot of pain, which can be expressed as anger, hostility, or aggression. They’re very anxious and afraid of what’s happening and may be thinking about the worst-case scenario – that a bump or lump is cancer,” Dr. Zeller said.
Patients may also get upset if they disagree with their doctors’ diagnosis or treatment plan or the doctor refuses to prescribe them the drugs or tests they want.
“One doctor commented recently: ‘After over 30 years in this business, I can say patients are worse now than at any point in my career. Entitled, demanding, obnoxious. Any denial is met with outrage and indignity, whether it’s an opioid request or a demand for MRI of something because they ‘want to know.’ ”
An orthopedic surgeon in Indiana lost his life after he refused to prescribe opioids to a patient. Her angry husband shot and killed the doctor in the parking lot only 2 hours after confronting him in his office.
Decreased physician-patient trust
“When doctors experience something frightening, they become more apprehensive in the future. There’s no doubt that after the first violent experience, they think of things differently,” said Dr. Zeller.
More than half of the doctors who reported experiencing at least one violent or potentially violent incident in the poll said they trusted patients less.
This diminished trust can negatively impact the physician-patient relationship, said the authors of a recent Health Affairs article.
“The more patients harm their health care providers, intentionally or unintentionally, the more difficult it will be for those providers to trust them, leading to yet another unfortunate pattern: physicians pulling back on some of the behaviors thought to be most trust-building, for example, talking about their personal lives, building rapport, displaying compassion, or giving out their personal cell phone numbers,” the article stated.
What doctors can do
Most doctors who experienced a violent or potentially violent incident said they had tried to defuse the situation and that they succeeded at least some of the time, the poll results show.
One of the best ways to defuse a situation is to be empathetic and show the person that you’re on their side and not the enemy, said Dr. Cheng,.
“Rather than making general statements like ‘I understand that you’re upset,’ it’s better to be specific about the reason the person is upset. For example: ‘I understand that you’re upset that the pharmacy didn’t fill your prescription’ or ‘I understand how you’re feeling about Doctor So-and-so, who didn’t treat you right,’ ” Dr. Cheng stated.
Dr. Zeller urged physicians to talk to patients about why they’re upset and how they can help them. That approach worked with a patient who was having a psychotic episode.
“I told the staff, who wanted to forcibly restrain him and inject him with medication, that I would talk to him. I asked the patient, who was screaming ‘ya ya ya ya,’ whether he would take his medication if I gave it to him and he said yes. When he was calm, he explained that he was screaming to stop the voices telling him to kill his parents. He then got the help he needed,” said Dr. Zeller.
Dr. Cheng was trained in de-escalation techniques as an Orange County reserve deputy sheriff. He and Dr. Zeller recommended that physicians and staff receive training in how to spot potentially violent behavior and defuse these situations before they escalate.
Dr. Cheng suggests looking at the person’s body language for signs of increasing agitation or tension, such as clenched fists, tense posture, tight jaw, or fidgeting that may be accompanied by shouting and/or verbal abuse.
Physicians also need to consider where they are physically in relation to patients they see. “You don’t want to be too close to the patient or stand in front of them, which can be seen as confrontational. Instead, stand or sit off to the side, and never block the door if the patient’s upset,” said Dr. Cheng.
He recommended that physician practices prepare for violent incidents by developing detailed plans, including how and when to escape, how to protect patients, and how to cooperate with law enforcement.
“If a violent incident is inescapable, physicians and staff must be ready to fight back with whatever tools they have available, which may include fire extinguishers, chairs, or scalpels,” said Dr. Cheng.
A version of this article originally appeared on Medscape.com.