User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'main-prefix')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
div[contains(@class, 'view-medstat-quiz-listing-panes')]
div[contains(@class, 'pane-article-sidebar-latest-news')]
div[contains(@class, 'medstat-accordion-set article-series')]
Gout: Suboptimal management a continuing problem
The prevalence of gout is skyrocketing worldwide, and while drugs in the pipeline hold promise for improving the efficacy and safety of treatment, experts warn that “gout remains suboptimally managed.”
“For a really well-understood disease, gout is remarkably undertreated,” said Robert A. Terkeltaub, MD, professor of medicine emeritus at the University of California, San Diego. “This is amazing and depressing because allopurinol has been around for about 60 years or so.”
Randomized, controlled trials show that 80%-90% of patients with gout can be effectively treated to target with existing gout therapies. “Over a year or two, gout flares improve and patients do well,” Dr. Terkeltaub said.
By lowering excessive levels of serum urate, current therapies slow the formation of monosodium urate crystals that precipitate within joints and soft tissues, inducing a highly inflammatory local response with superimposed systemic inflammation. These therapies reduce the frequency of excruciatingly painful gout flares.
“Many patients with gout are not taking urate-lowering therapy at all,” Sara K. Tedeschi, MD, MPH, assistant professor of medicine at Harvard Medical School, Boston, and head of crystal-induced arthritis diseases at Brigham and Women’s Hospital, also in Boston, said in an interview.
“Unfortunately, a common problem in gout is treatment inertia,” said Tuhina Neogi, MD, PhD, chief of rheumatology at Boston Medical Center.
On a global scale, only one-third of patients with gout are started on urate-lowering therapy, and more than 50% abandon treatment after 1 year. As a result, the effectiveness of urate-lowering therapies in reality is well below 50%, Dr. Terkeltaub said.
“I think gout has been taken less seriously than it should be for quite some time,” he explained in an interview. Gout’s impact on health and well-being is no trivial matter. A recent study showed that a diagnosis of gout was associated with an increased risk of anxiety and depression, and there is new evidence suggesting that gout flares are associated with an increased risk of cardiovascular events, including fatal myocardial infarction and stroke.
“We need drugs that are not just effective but also safe, and we need to incorporate real-world data into our assessment of treatment effectiveness, especially in the presence of comorbidities,” Dr. Terkeltaub said.
The prevalence of what used to be thought of as the “disease of kings” has increased 100% over the last 30 years, outstripping world population growth and life expectancy. In the United States, an estimated 5% of adults, or 12 million, have gout. Globally, the number affected exceeds 50 million.
The patient demographics associated with gout have also expanded. Once seen primarily in fleshy, middle-aged men of privilege, gout affects more women, more adults at either end of the age spectrum, and more people in Third World countries than ever before.
Management
In the United States, the optimal management of gout remains the subject of debate, with differences in expert opinion reflected in evidence-based clinical guidelines. “We know that the perception of gout is different between primary care physicians, patients, and rheumatologists,” Dr. Terkeltaub said.
The 2017 American College of Physicians guidelines for the management of gout recommend a treat-to-symptom approach to urate-lowering therapy. However, the 2020 American College of Rheumatology guidelines reinforce a standard treat-to-target strategy to a serum urate target of < 6.0 mg/dL.
In their report, the ACR guidelines’ authors stated that the use of urate-lowering therapy for gout has not increased in the last decade. Research shows that adherence to treatment for gout continues to be the lowest among seven common chronic medical conditions, including hypertension and seizure disorders, they said.
Some physicians don’t recommend urate-lowering medication to their patients with gout, and others don’t up-titrate it sufficiently to meet the recommended serum urate target, said Dr. Tedeschi. The latter “can require increasing the dose of allopurinol well beyond the 300 mg that often seems the landing point for many patients with gout,” she pointed out.
In fact, it can take up to 800 mg a day of allopurinol – less in patients with moderate to severe kidney disease – to reduce the symptom burden in gout. And it can take a year or longer of drug testing and titration to reach the optimal serum urate target. Paradoxically, gout flares usually get worse during this time.
“We need to reduce the time it takes to get the patient to the serum urate target, and simplify regimens with once-a-day dosing,” Dr. Terkeltaub said. “We also need greater precision so that we can get a home run, hitting the serum urate target the first or second visit, with minimal dose titration.”
Clinician education is important, but education alone is not enough, Dr. Neogi emphasized. “Just as clinicians treat-to-target in other conditions such as hypertension and diabetes, or titrate warfarin to maintain a certain level of anticoagulation, gout must be monitored and treatments adjusted accordingly,” she said.
Practice changes, such as partnering with nursing or pharmacy, may help facilitate in-clinic dose titration, “much like a warfarin clinic,” Dr. Neogi suggested.
That’s exactly what Dr. Terkeltaub has done. Overwhelmed by the number of gout consults, Dr. Terkeltaub and his team set up a pharmacist-managed, rheumatology-supervised clinic to care for gout patients remotely. The model has been very successful, he said. Nurses and clinical pharmacists educate the patients and manage their lab testing and prescriptions, all according to ACR guidelines.
The treatment of gout has become more complex, with a greater risk of drug complications and interactions, particularly in older patients with comorbid diabetes, chronic kidney disease, and heart disease. Many of the patients he sees are already on “10, 15, or 20 other medications,” Dr. Terkeltaub noted.
The steps involved in the titration of urate-lowering therapy also complicate the treatment of gout, making it impractical for many patients and impossible for others whose access to primary care is limited to one or two visits a year. The process of drug titration, with steadily increasing doses, can make patients anxious about the possibility of being overmedicated. Taking a drug every day, even when joints feel “normal,” can also increase the risk of nonadherence.
“In our conversations with patients with gout, it’s extremely important that we counsel them about the need to take urate-lowering therapy on an ongoing basis to reduce the risk of a gout flare,” said Dr. Tedeschi. “Patients need to have prescription refills available and know to contact the doctor before they run out, so that the chances of having a gout flare are reduced.”
Current drugs
Although urate-lowering drugs form the cornerstone of gout therapy, there are only three oral medications available in the United States currently, and all have significant limitations. “We need more drugs, basically,” Dr. Terkeltaub said.
- Allopurinol (Zyloprim, Aloprim), an inexpensive xanthine oxidase inhibitor (XOI), is still considered a first-line treatment, but is associated with allopurinol hypersensitivity syndrome. In select patients of Asian, African, and Arab descent, this adverse drug reaction can be life-threatening, and is associated with a mortality rate of 20%-25%.
- Febuxostat (Uloric), another XOI, is considered a second-line drug in the treatment of gout, but has carried a boxed warning from the Food and Drug Administration since 2019. It is associated with a significantly increased risk of cardiovascular death.
- Probenecid (Probalan), a uricosuric agent that increases renal uric acid excretion, is associated with an increased risk of drug interactions and kidney stones, and is rarely used.
Drugs in the pipeline
New drugs in the pipeline offer treatment options that are not only effective but also safe. “This will be important in clinical practice, especially for patients in whom existing medications are contraindicated or there is an increased risk of side effects,” Dr. Neogi said.
Most of these investigational drugs are uricosuric agents that increase the renal excretion of uric acid, reducing serum levels. “The pipeline of new drugs is rich,” Dr. Terkeltaub said. “These drugs are very selective and really work well and they appear to be safe.”
AR882, an inhibitor of selective uric acid transporter 1 (URAT1), is shaping up to be one of them. In July, results from a phase 2b study of AR882 were presented at the annual European Congress of Rheumatology in Milan. They showed that in the intent-to-treat population, 73% of patients had serum uric acid levels < 5 mg/dL and 55% had < 4 mg/dL by week 12 of therapy. In the per-protocol analysis, 82% had serum uric acid levels < 5 mg/dL and 63% < 4 mg/dL.
“These efficacy results are not typically what you see with a once-daily oral medication, so it is really exciting,” said Robert Keenan, MD, chief medical officer of Arthrosi Therapeutics, San Diego, who presented the results.
“More efficacious URAT1 inhibitors that are safe and have a reduced pill burden will be useful additions to current urate lowering options,” Dr. Neogi said.
The recent phase 3 DISSOLVE I and II trials of the investigational uricase-based infusion therapy SEL-212 in refractory gout have also demonstrated encouraging results, particularly in older patients. In DISSOLVE I, a response rate of 65% was observed in patients 50 years of age and older at least 80% of the time during month 6 of treatment. In DISSOLVE II, a response rate of 47% was reported in older patients.
SEL-212, which is made up of PEGylated uricase (pegadricase) coadministered with sirolimus (Rapamycin), will be submitted for U.S. regulatory approval in the first half of 2024.
In the management of gout flares, interleukin (IL)-1beta and inflammasome inhibitors, both of which target specific inflammatory pathways, could also provide attractive additions to urate-lowering therapies. Other agents commonly used in the treatment of flares, such as NSAIDs, steroids, and colchicine (Colcrys), are not as specific, and have side effects that often limit their usability, Dr. Neogi said.
In the meantime, new research indicates that an inflammasome inhibitor that has already been approved for use in diabetes may provide distinct benefits for the management of gout. An analysis of data from 15,067 adults with both gout and type 2 diabetes showed that when a sodium-glucose cotransporter 2 (SGLT-2) inhibitor was added to urate-lowering therapy, the symptoms of gout, including flares, were significantly reduced, resulting in fewer emergency department visits and hospitalizations.
“SGLT-2 inhibitors have anti-inflammatory activity that limits the progression of kidney failure, heart failure, and will also lower the serum uric acid,” said Dr. Terkeltaub. “That’s a major development.”
Dr. Neogi disclosed relationships with Novartis, Pfizer/Lilly, and Regeneron, Dr. Terkeltaub reported relationships with Dyve, Fortress, and Atom, and Dr. Tedeschi reported a relationship with Novartis.
This story was updated on August 14, 2023.
The prevalence of gout is skyrocketing worldwide, and while drugs in the pipeline hold promise for improving the efficacy and safety of treatment, experts warn that “gout remains suboptimally managed.”
“For a really well-understood disease, gout is remarkably undertreated,” said Robert A. Terkeltaub, MD, professor of medicine emeritus at the University of California, San Diego. “This is amazing and depressing because allopurinol has been around for about 60 years or so.”
Randomized, controlled trials show that 80%-90% of patients with gout can be effectively treated to target with existing gout therapies. “Over a year or two, gout flares improve and patients do well,” Dr. Terkeltaub said.
By lowering excessive levels of serum urate, current therapies slow the formation of monosodium urate crystals that precipitate within joints and soft tissues, inducing a highly inflammatory local response with superimposed systemic inflammation. These therapies reduce the frequency of excruciatingly painful gout flares.
“Many patients with gout are not taking urate-lowering therapy at all,” Sara K. Tedeschi, MD, MPH, assistant professor of medicine at Harvard Medical School, Boston, and head of crystal-induced arthritis diseases at Brigham and Women’s Hospital, also in Boston, said in an interview.
“Unfortunately, a common problem in gout is treatment inertia,” said Tuhina Neogi, MD, PhD, chief of rheumatology at Boston Medical Center.
On a global scale, only one-third of patients with gout are started on urate-lowering therapy, and more than 50% abandon treatment after 1 year. As a result, the effectiveness of urate-lowering therapies in reality is well below 50%, Dr. Terkeltaub said.
“I think gout has been taken less seriously than it should be for quite some time,” he explained in an interview. Gout’s impact on health and well-being is no trivial matter. A recent study showed that a diagnosis of gout was associated with an increased risk of anxiety and depression, and there is new evidence suggesting that gout flares are associated with an increased risk of cardiovascular events, including fatal myocardial infarction and stroke.
“We need drugs that are not just effective but also safe, and we need to incorporate real-world data into our assessment of treatment effectiveness, especially in the presence of comorbidities,” Dr. Terkeltaub said.
The prevalence of what used to be thought of as the “disease of kings” has increased 100% over the last 30 years, outstripping world population growth and life expectancy. In the United States, an estimated 5% of adults, or 12 million, have gout. Globally, the number affected exceeds 50 million.
The patient demographics associated with gout have also expanded. Once seen primarily in fleshy, middle-aged men of privilege, gout affects more women, more adults at either end of the age spectrum, and more people in Third World countries than ever before.
Management
In the United States, the optimal management of gout remains the subject of debate, with differences in expert opinion reflected in evidence-based clinical guidelines. “We know that the perception of gout is different between primary care physicians, patients, and rheumatologists,” Dr. Terkeltaub said.
The 2017 American College of Physicians guidelines for the management of gout recommend a treat-to-symptom approach to urate-lowering therapy. However, the 2020 American College of Rheumatology guidelines reinforce a standard treat-to-target strategy to a serum urate target of < 6.0 mg/dL.
In their report, the ACR guidelines’ authors stated that the use of urate-lowering therapy for gout has not increased in the last decade. Research shows that adherence to treatment for gout continues to be the lowest among seven common chronic medical conditions, including hypertension and seizure disorders, they said.
Some physicians don’t recommend urate-lowering medication to their patients with gout, and others don’t up-titrate it sufficiently to meet the recommended serum urate target, said Dr. Tedeschi. The latter “can require increasing the dose of allopurinol well beyond the 300 mg that often seems the landing point for many patients with gout,” she pointed out.
In fact, it can take up to 800 mg a day of allopurinol – less in patients with moderate to severe kidney disease – to reduce the symptom burden in gout. And it can take a year or longer of drug testing and titration to reach the optimal serum urate target. Paradoxically, gout flares usually get worse during this time.
“We need to reduce the time it takes to get the patient to the serum urate target, and simplify regimens with once-a-day dosing,” Dr. Terkeltaub said. “We also need greater precision so that we can get a home run, hitting the serum urate target the first or second visit, with minimal dose titration.”
Clinician education is important, but education alone is not enough, Dr. Neogi emphasized. “Just as clinicians treat-to-target in other conditions such as hypertension and diabetes, or titrate warfarin to maintain a certain level of anticoagulation, gout must be monitored and treatments adjusted accordingly,” she said.
Practice changes, such as partnering with nursing or pharmacy, may help facilitate in-clinic dose titration, “much like a warfarin clinic,” Dr. Neogi suggested.
That’s exactly what Dr. Terkeltaub has done. Overwhelmed by the number of gout consults, Dr. Terkeltaub and his team set up a pharmacist-managed, rheumatology-supervised clinic to care for gout patients remotely. The model has been very successful, he said. Nurses and clinical pharmacists educate the patients and manage their lab testing and prescriptions, all according to ACR guidelines.
The treatment of gout has become more complex, with a greater risk of drug complications and interactions, particularly in older patients with comorbid diabetes, chronic kidney disease, and heart disease. Many of the patients he sees are already on “10, 15, or 20 other medications,” Dr. Terkeltaub noted.
The steps involved in the titration of urate-lowering therapy also complicate the treatment of gout, making it impractical for many patients and impossible for others whose access to primary care is limited to one or two visits a year. The process of drug titration, with steadily increasing doses, can make patients anxious about the possibility of being overmedicated. Taking a drug every day, even when joints feel “normal,” can also increase the risk of nonadherence.
“In our conversations with patients with gout, it’s extremely important that we counsel them about the need to take urate-lowering therapy on an ongoing basis to reduce the risk of a gout flare,” said Dr. Tedeschi. “Patients need to have prescription refills available and know to contact the doctor before they run out, so that the chances of having a gout flare are reduced.”
Current drugs
Although urate-lowering drugs form the cornerstone of gout therapy, there are only three oral medications available in the United States currently, and all have significant limitations. “We need more drugs, basically,” Dr. Terkeltaub said.
- Allopurinol (Zyloprim, Aloprim), an inexpensive xanthine oxidase inhibitor (XOI), is still considered a first-line treatment, but is associated with allopurinol hypersensitivity syndrome. In select patients of Asian, African, and Arab descent, this adverse drug reaction can be life-threatening, and is associated with a mortality rate of 20%-25%.
- Febuxostat (Uloric), another XOI, is considered a second-line drug in the treatment of gout, but has carried a boxed warning from the Food and Drug Administration since 2019. It is associated with a significantly increased risk of cardiovascular death.
- Probenecid (Probalan), a uricosuric agent that increases renal uric acid excretion, is associated with an increased risk of drug interactions and kidney stones, and is rarely used.
Drugs in the pipeline
New drugs in the pipeline offer treatment options that are not only effective but also safe. “This will be important in clinical practice, especially for patients in whom existing medications are contraindicated or there is an increased risk of side effects,” Dr. Neogi said.
Most of these investigational drugs are uricosuric agents that increase the renal excretion of uric acid, reducing serum levels. “The pipeline of new drugs is rich,” Dr. Terkeltaub said. “These drugs are very selective and really work well and they appear to be safe.”
AR882, an inhibitor of selective uric acid transporter 1 (URAT1), is shaping up to be one of them. In July, results from a phase 2b study of AR882 were presented at the annual European Congress of Rheumatology in Milan. They showed that in the intent-to-treat population, 73% of patients had serum uric acid levels < 5 mg/dL and 55% had < 4 mg/dL by week 12 of therapy. In the per-protocol analysis, 82% had serum uric acid levels < 5 mg/dL and 63% < 4 mg/dL.
“These efficacy results are not typically what you see with a once-daily oral medication, so it is really exciting,” said Robert Keenan, MD, chief medical officer of Arthrosi Therapeutics, San Diego, who presented the results.
“More efficacious URAT1 inhibitors that are safe and have a reduced pill burden will be useful additions to current urate lowering options,” Dr. Neogi said.
The recent phase 3 DISSOLVE I and II trials of the investigational uricase-based infusion therapy SEL-212 in refractory gout have also demonstrated encouraging results, particularly in older patients. In DISSOLVE I, a response rate of 65% was observed in patients 50 years of age and older at least 80% of the time during month 6 of treatment. In DISSOLVE II, a response rate of 47% was reported in older patients.
SEL-212, which is made up of PEGylated uricase (pegadricase) coadministered with sirolimus (Rapamycin), will be submitted for U.S. regulatory approval in the first half of 2024.
In the management of gout flares, interleukin (IL)-1beta and inflammasome inhibitors, both of which target specific inflammatory pathways, could also provide attractive additions to urate-lowering therapies. Other agents commonly used in the treatment of flares, such as NSAIDs, steroids, and colchicine (Colcrys), are not as specific, and have side effects that often limit their usability, Dr. Neogi said.
In the meantime, new research indicates that an inflammasome inhibitor that has already been approved for use in diabetes may provide distinct benefits for the management of gout. An analysis of data from 15,067 adults with both gout and type 2 diabetes showed that when a sodium-glucose cotransporter 2 (SGLT-2) inhibitor was added to urate-lowering therapy, the symptoms of gout, including flares, were significantly reduced, resulting in fewer emergency department visits and hospitalizations.
“SGLT-2 inhibitors have anti-inflammatory activity that limits the progression of kidney failure, heart failure, and will also lower the serum uric acid,” said Dr. Terkeltaub. “That’s a major development.”
Dr. Neogi disclosed relationships with Novartis, Pfizer/Lilly, and Regeneron, Dr. Terkeltaub reported relationships with Dyve, Fortress, and Atom, and Dr. Tedeschi reported a relationship with Novartis.
This story was updated on August 14, 2023.
The prevalence of gout is skyrocketing worldwide, and while drugs in the pipeline hold promise for improving the efficacy and safety of treatment, experts warn that “gout remains suboptimally managed.”
“For a really well-understood disease, gout is remarkably undertreated,” said Robert A. Terkeltaub, MD, professor of medicine emeritus at the University of California, San Diego. “This is amazing and depressing because allopurinol has been around for about 60 years or so.”
Randomized, controlled trials show that 80%-90% of patients with gout can be effectively treated to target with existing gout therapies. “Over a year or two, gout flares improve and patients do well,” Dr. Terkeltaub said.
By lowering excessive levels of serum urate, current therapies slow the formation of monosodium urate crystals that precipitate within joints and soft tissues, inducing a highly inflammatory local response with superimposed systemic inflammation. These therapies reduce the frequency of excruciatingly painful gout flares.
“Many patients with gout are not taking urate-lowering therapy at all,” Sara K. Tedeschi, MD, MPH, assistant professor of medicine at Harvard Medical School, Boston, and head of crystal-induced arthritis diseases at Brigham and Women’s Hospital, also in Boston, said in an interview.
“Unfortunately, a common problem in gout is treatment inertia,” said Tuhina Neogi, MD, PhD, chief of rheumatology at Boston Medical Center.
On a global scale, only one-third of patients with gout are started on urate-lowering therapy, and more than 50% abandon treatment after 1 year. As a result, the effectiveness of urate-lowering therapies in reality is well below 50%, Dr. Terkeltaub said.
“I think gout has been taken less seriously than it should be for quite some time,” he explained in an interview. Gout’s impact on health and well-being is no trivial matter. A recent study showed that a diagnosis of gout was associated with an increased risk of anxiety and depression, and there is new evidence suggesting that gout flares are associated with an increased risk of cardiovascular events, including fatal myocardial infarction and stroke.
“We need drugs that are not just effective but also safe, and we need to incorporate real-world data into our assessment of treatment effectiveness, especially in the presence of comorbidities,” Dr. Terkeltaub said.
The prevalence of what used to be thought of as the “disease of kings” has increased 100% over the last 30 years, outstripping world population growth and life expectancy. In the United States, an estimated 5% of adults, or 12 million, have gout. Globally, the number affected exceeds 50 million.
The patient demographics associated with gout have also expanded. Once seen primarily in fleshy, middle-aged men of privilege, gout affects more women, more adults at either end of the age spectrum, and more people in Third World countries than ever before.
Management
In the United States, the optimal management of gout remains the subject of debate, with differences in expert opinion reflected in evidence-based clinical guidelines. “We know that the perception of gout is different between primary care physicians, patients, and rheumatologists,” Dr. Terkeltaub said.
The 2017 American College of Physicians guidelines for the management of gout recommend a treat-to-symptom approach to urate-lowering therapy. However, the 2020 American College of Rheumatology guidelines reinforce a standard treat-to-target strategy to a serum urate target of < 6.0 mg/dL.
In their report, the ACR guidelines’ authors stated that the use of urate-lowering therapy for gout has not increased in the last decade. Research shows that adherence to treatment for gout continues to be the lowest among seven common chronic medical conditions, including hypertension and seizure disorders, they said.
Some physicians don’t recommend urate-lowering medication to their patients with gout, and others don’t up-titrate it sufficiently to meet the recommended serum urate target, said Dr. Tedeschi. The latter “can require increasing the dose of allopurinol well beyond the 300 mg that often seems the landing point for many patients with gout,” she pointed out.
In fact, it can take up to 800 mg a day of allopurinol – less in patients with moderate to severe kidney disease – to reduce the symptom burden in gout. And it can take a year or longer of drug testing and titration to reach the optimal serum urate target. Paradoxically, gout flares usually get worse during this time.
“We need to reduce the time it takes to get the patient to the serum urate target, and simplify regimens with once-a-day dosing,” Dr. Terkeltaub said. “We also need greater precision so that we can get a home run, hitting the serum urate target the first or second visit, with minimal dose titration.”
Clinician education is important, but education alone is not enough, Dr. Neogi emphasized. “Just as clinicians treat-to-target in other conditions such as hypertension and diabetes, or titrate warfarin to maintain a certain level of anticoagulation, gout must be monitored and treatments adjusted accordingly,” she said.
Practice changes, such as partnering with nursing or pharmacy, may help facilitate in-clinic dose titration, “much like a warfarin clinic,” Dr. Neogi suggested.
That’s exactly what Dr. Terkeltaub has done. Overwhelmed by the number of gout consults, Dr. Terkeltaub and his team set up a pharmacist-managed, rheumatology-supervised clinic to care for gout patients remotely. The model has been very successful, he said. Nurses and clinical pharmacists educate the patients and manage their lab testing and prescriptions, all according to ACR guidelines.
The treatment of gout has become more complex, with a greater risk of drug complications and interactions, particularly in older patients with comorbid diabetes, chronic kidney disease, and heart disease. Many of the patients he sees are already on “10, 15, or 20 other medications,” Dr. Terkeltaub noted.
The steps involved in the titration of urate-lowering therapy also complicate the treatment of gout, making it impractical for many patients and impossible for others whose access to primary care is limited to one or two visits a year. The process of drug titration, with steadily increasing doses, can make patients anxious about the possibility of being overmedicated. Taking a drug every day, even when joints feel “normal,” can also increase the risk of nonadherence.
“In our conversations with patients with gout, it’s extremely important that we counsel them about the need to take urate-lowering therapy on an ongoing basis to reduce the risk of a gout flare,” said Dr. Tedeschi. “Patients need to have prescription refills available and know to contact the doctor before they run out, so that the chances of having a gout flare are reduced.”
Current drugs
Although urate-lowering drugs form the cornerstone of gout therapy, there are only three oral medications available in the United States currently, and all have significant limitations. “We need more drugs, basically,” Dr. Terkeltaub said.
- Allopurinol (Zyloprim, Aloprim), an inexpensive xanthine oxidase inhibitor (XOI), is still considered a first-line treatment, but is associated with allopurinol hypersensitivity syndrome. In select patients of Asian, African, and Arab descent, this adverse drug reaction can be life-threatening, and is associated with a mortality rate of 20%-25%.
- Febuxostat (Uloric), another XOI, is considered a second-line drug in the treatment of gout, but has carried a boxed warning from the Food and Drug Administration since 2019. It is associated with a significantly increased risk of cardiovascular death.
- Probenecid (Probalan), a uricosuric agent that increases renal uric acid excretion, is associated with an increased risk of drug interactions and kidney stones, and is rarely used.
Drugs in the pipeline
New drugs in the pipeline offer treatment options that are not only effective but also safe. “This will be important in clinical practice, especially for patients in whom existing medications are contraindicated or there is an increased risk of side effects,” Dr. Neogi said.
Most of these investigational drugs are uricosuric agents that increase the renal excretion of uric acid, reducing serum levels. “The pipeline of new drugs is rich,” Dr. Terkeltaub said. “These drugs are very selective and really work well and they appear to be safe.”
AR882, an inhibitor of selective uric acid transporter 1 (URAT1), is shaping up to be one of them. In July, results from a phase 2b study of AR882 were presented at the annual European Congress of Rheumatology in Milan. They showed that in the intent-to-treat population, 73% of patients had serum uric acid levels < 5 mg/dL and 55% had < 4 mg/dL by week 12 of therapy. In the per-protocol analysis, 82% had serum uric acid levels < 5 mg/dL and 63% < 4 mg/dL.
“These efficacy results are not typically what you see with a once-daily oral medication, so it is really exciting,” said Robert Keenan, MD, chief medical officer of Arthrosi Therapeutics, San Diego, who presented the results.
“More efficacious URAT1 inhibitors that are safe and have a reduced pill burden will be useful additions to current urate lowering options,” Dr. Neogi said.
The recent phase 3 DISSOLVE I and II trials of the investigational uricase-based infusion therapy SEL-212 in refractory gout have also demonstrated encouraging results, particularly in older patients. In DISSOLVE I, a response rate of 65% was observed in patients 50 years of age and older at least 80% of the time during month 6 of treatment. In DISSOLVE II, a response rate of 47% was reported in older patients.
SEL-212, which is made up of PEGylated uricase (pegadricase) coadministered with sirolimus (Rapamycin), will be submitted for U.S. regulatory approval in the first half of 2024.
In the management of gout flares, interleukin (IL)-1beta and inflammasome inhibitors, both of which target specific inflammatory pathways, could also provide attractive additions to urate-lowering therapies. Other agents commonly used in the treatment of flares, such as NSAIDs, steroids, and colchicine (Colcrys), are not as specific, and have side effects that often limit their usability, Dr. Neogi said.
In the meantime, new research indicates that an inflammasome inhibitor that has already been approved for use in diabetes may provide distinct benefits for the management of gout. An analysis of data from 15,067 adults with both gout and type 2 diabetes showed that when a sodium-glucose cotransporter 2 (SGLT-2) inhibitor was added to urate-lowering therapy, the symptoms of gout, including flares, were significantly reduced, resulting in fewer emergency department visits and hospitalizations.
“SGLT-2 inhibitors have anti-inflammatory activity that limits the progression of kidney failure, heart failure, and will also lower the serum uric acid,” said Dr. Terkeltaub. “That’s a major development.”
Dr. Neogi disclosed relationships with Novartis, Pfizer/Lilly, and Regeneron, Dr. Terkeltaub reported relationships with Dyve, Fortress, and Atom, and Dr. Tedeschi reported a relationship with Novartis.
This story was updated on August 14, 2023.
We asked doctors using AI scribes: Just how good are they?
Andrea Partida, DO, an obstetrician and gynecologist in Enid, Okla., loves her new assistant.
The 15 or 20 minutes she used to spend on documentation for each patient visit is now 3. The 2-3 hours she’d spend charting outside clinic hours is maybe 1.
All that time saved allows her to see two to five more patients a day, provide better care to each patient, and get more involved in hospital leadership at Integris Health, where she works.
“I have a better work-life balance with my family,” Dr. Partida said. “I leave work at work and get home earlier.”
You’ve probably figured out the plot twist: Dr. Partida’s assistant is not a person – it’s artificial intelligence (AI).
Dr. Partida uses IRIS, a tool from OnPoint Healthcare Partners, part of a fast-growing niche of AI medical scribes designed to automate onerous data entry. The evolution of generative AI – specifically, large language models, such as ChatGPT – has led to a rapid explosion of these tools. Other companies in the space include Abridge, Ambience Healthcare, Augmedix, DeepScribe, Nuance (part of Microsoft), and Suki. The newest kid on the block, Amazon Web Services, announced the launch of HealthScribe in July.
These tools – some of which are already on the market, with more on the way – record patient visits and generate notes for treatment and billing. Earlier iterations combine AI with offsite human scribes who provide quality control. But more and more are fully automated, no human required. Some also offer video recording and foreign language translation.
The promise is alluring: Ease your workload and reclaim hours in your day so you can spend more time with patients or try that “work-life balance” thing you’ve heard so much about.
But do these tools fulfill that promise?
According to Dr. Partida and other doctors who spoke with this news organization, the answer is a resounding yes.
A tech solution for a tech problem
“I believe a lot of doctors see patients for free. They get paid to do paperwork,” said Anthony J. Mazzarelli, MD, JD, MBE, co-president and CEO of Cooper University Health Care, in Camden, N.J.
Indeed, for every hour U.S. clinicians spend with their patients, they may spend 2 more hours documenting in electronic health records (EHRs), estimates show. About half of doctors, especially those in primary care, report feeling burned out, and some 42% say they want to quit clinical practice.
Enter AI scribes.
“The holy grail in medicine right now is improving burnout while also maintaining or improving productivity and quality,” said Patricia Garcia, MD, associate clinical information officer for ambulatory care at Stanford (Calif.) Health Care. “These ambient digital scribes have the potential to do just that.”
While anyone can buy these products, their use has been mostly limited to pilot programs and early adopters so far, said Dr. Garcia, who has been helping to pilot Nuance’s digital scribe, DAX, at Stanford.
But that’s expected to change quickly. “I don’t think the time horizon is a decade,” Dr. Garcia said. “I think within a matter of 2 or 3 years, these tools will be pervasive throughout health care.”
Since introducing these tools at Cooper, “our doctors’ paperwork burden is significantly lighter,” said Dr. Mazzarelli, who decides which technologies Cooper should invest in and who monitors their results. In Cooper studies, physicians who used DAX more than half the time spent 43% less time working on notes.
“They spend more time connecting with their patients, talking with them, and looking them in the eye,” Dr. Mazzarelli said. That, in turn, seems to improve patient outcomes, reduce doctor burnout and turnover, and lower costs.
The AI scribes, by virtue of eliminating the distraction of note taking, also allow doctors to give their full attention to the patient. “The patient relationship is the most important aspect of medicine,” said Raul Ayala, MD, MHCM, a family medicine physician at Adventist Health, in Hanford, Calif., who uses Augmedix. The digital scribe “helps us strengthen that relationship.”
What’s it like to use an AI medical scribe?
The scribes feature hardware (typically a smartphone or tablet) and software built on automatic speech recognition, natural language processing, and machine learning. Download an app to your device, and you’re ready to go. Use it to record in-person or telehealth visits.
In the first week, a company may help train you to use the hardware and software. You’ll likely start by using it for a few patient visits per day, ramping up gradually. Dr. Partida said she was comfortable using the system for all her patients in 6 weeks.
Each day, Dr. Partida logs in to a dedicated smartphone or tablet, opens the app, and reviews her schedule, including details she needs to prepare for each patient.
At the start of each patient visit, Dr. Partida taps the app icon to begin recording and lays the device nearby. She can pause as needed. At the end of the visit, she taps the icon again to stop recording.
The AI listens, creates the note, and updates relevant data in the EHR. The note includes patient problems, assessment, treatment plan, patient history, orders, and tasks for staff, along with medications, referrals, and preauthorizations. A human scribe, who is also a physician, reviews the information for accuracy and edits it as needed. By the next morning, the data are ready for Dr. Partida to review.
Fully automated versions can generate notes much faster. Jack Shilling, MD, MBA, an orthopedic surgeon at Cooper University Health Care, in Voorhees, N.J., uses DAX. A new feature called DAX Express – which uses OpenAI’s GPT-4 but no humans – provides him with a draft of his clinical notes in just seconds.
How accurate are AI notes?
The accuracy of those notes remains an open question, Dr. Garcia said – mostly because accuracy can be hard to define.
“If you asked five docs to write a note based on the same patient encounter, you’d get five different notes,” Dr. Garcia said. “That makes it hard to assess these technologies in a scientifically rigorous way.”
Still, the onus is on the physician to review the notes and edit them as needed, Dr. Garcia said. How light or heavy those edits are can depend on your unique preferences.
Dr. Shilling said he may need to lightly edit transcripts of his conversations with patients. “When someone tells me how long their knee hurts, slight variability in their transcribed words is tolerable,” he said. But for some things – such as physical exam notes and x-ray readings – he dictates directly into the device, speaking at a closer range and being less conversational, more exact in his speech.
Should you let patients know they’re being recorded?
The federal Health Insurance Portability and Accountability Act (HIPAA) does not require providers to inform patients that their face-to-face conversations are being recorded, said Daniel Lebovic, JD, corporate legal counsel at Compliancy Group, in Greenlawn, N.Y., a company that helps providers adhere to HIPAA rules.
But make sure you know the laws in your state and the policies at your health care practice. State laws may require providers to inform patients and to get patients’ consent in advance of being recorded.
All the doctors who spoke to this news organization said their patients are informed that they’ll be recorded and that they can opt out if they wish.
How much do AI scribes cost?
As the marketplace for these tools expands, companies are offering more products and services at different price points that target a range of organizations, from large health care systems to small private practices.
Price models vary, said Dr. Garcia. Some are based on the number of users, others on the number of notes, and still others on minutes.
Amazon’s HealthScribe is priced at 10 cents per minute. For 1,000 consultation transcripts per month, with each call averaging 15 minutes, it would take 15,000 minutes at a total cost of $1,500 for the month.
In general, the rapidly growing competition in this space could mean prices become more affordable, Dr. Garcia said. “It’s good that so many are getting into this game, because that means the price will come down and it will be a lot more accessible to everybody.”
A version of this article appeared on Medscape.com.
Andrea Partida, DO, an obstetrician and gynecologist in Enid, Okla., loves her new assistant.
The 15 or 20 minutes she used to spend on documentation for each patient visit is now 3. The 2-3 hours she’d spend charting outside clinic hours is maybe 1.
All that time saved allows her to see two to five more patients a day, provide better care to each patient, and get more involved in hospital leadership at Integris Health, where she works.
“I have a better work-life balance with my family,” Dr. Partida said. “I leave work at work and get home earlier.”
You’ve probably figured out the plot twist: Dr. Partida’s assistant is not a person – it’s artificial intelligence (AI).
Dr. Partida uses IRIS, a tool from OnPoint Healthcare Partners, part of a fast-growing niche of AI medical scribes designed to automate onerous data entry. The evolution of generative AI – specifically, large language models, such as ChatGPT – has led to a rapid explosion of these tools. Other companies in the space include Abridge, Ambience Healthcare, Augmedix, DeepScribe, Nuance (part of Microsoft), and Suki. The newest kid on the block, Amazon Web Services, announced the launch of HealthScribe in July.
These tools – some of which are already on the market, with more on the way – record patient visits and generate notes for treatment and billing. Earlier iterations combine AI with offsite human scribes who provide quality control. But more and more are fully automated, no human required. Some also offer video recording and foreign language translation.
The promise is alluring: Ease your workload and reclaim hours in your day so you can spend more time with patients or try that “work-life balance” thing you’ve heard so much about.
But do these tools fulfill that promise?
According to Dr. Partida and other doctors who spoke with this news organization, the answer is a resounding yes.
A tech solution for a tech problem
“I believe a lot of doctors see patients for free. They get paid to do paperwork,” said Anthony J. Mazzarelli, MD, JD, MBE, co-president and CEO of Cooper University Health Care, in Camden, N.J.
Indeed, for every hour U.S. clinicians spend with their patients, they may spend 2 more hours documenting in electronic health records (EHRs), estimates show. About half of doctors, especially those in primary care, report feeling burned out, and some 42% say they want to quit clinical practice.
Enter AI scribes.
“The holy grail in medicine right now is improving burnout while also maintaining or improving productivity and quality,” said Patricia Garcia, MD, associate clinical information officer for ambulatory care at Stanford (Calif.) Health Care. “These ambient digital scribes have the potential to do just that.”
While anyone can buy these products, their use has been mostly limited to pilot programs and early adopters so far, said Dr. Garcia, who has been helping to pilot Nuance’s digital scribe, DAX, at Stanford.
But that’s expected to change quickly. “I don’t think the time horizon is a decade,” Dr. Garcia said. “I think within a matter of 2 or 3 years, these tools will be pervasive throughout health care.”
Since introducing these tools at Cooper, “our doctors’ paperwork burden is significantly lighter,” said Dr. Mazzarelli, who decides which technologies Cooper should invest in and who monitors their results. In Cooper studies, physicians who used DAX more than half the time spent 43% less time working on notes.
“They spend more time connecting with their patients, talking with them, and looking them in the eye,” Dr. Mazzarelli said. That, in turn, seems to improve patient outcomes, reduce doctor burnout and turnover, and lower costs.
The AI scribes, by virtue of eliminating the distraction of note taking, also allow doctors to give their full attention to the patient. “The patient relationship is the most important aspect of medicine,” said Raul Ayala, MD, MHCM, a family medicine physician at Adventist Health, in Hanford, Calif., who uses Augmedix. The digital scribe “helps us strengthen that relationship.”
What’s it like to use an AI medical scribe?
The scribes feature hardware (typically a smartphone or tablet) and software built on automatic speech recognition, natural language processing, and machine learning. Download an app to your device, and you’re ready to go. Use it to record in-person or telehealth visits.
In the first week, a company may help train you to use the hardware and software. You’ll likely start by using it for a few patient visits per day, ramping up gradually. Dr. Partida said she was comfortable using the system for all her patients in 6 weeks.
Each day, Dr. Partida logs in to a dedicated smartphone or tablet, opens the app, and reviews her schedule, including details she needs to prepare for each patient.
At the start of each patient visit, Dr. Partida taps the app icon to begin recording and lays the device nearby. She can pause as needed. At the end of the visit, she taps the icon again to stop recording.
The AI listens, creates the note, and updates relevant data in the EHR. The note includes patient problems, assessment, treatment plan, patient history, orders, and tasks for staff, along with medications, referrals, and preauthorizations. A human scribe, who is also a physician, reviews the information for accuracy and edits it as needed. By the next morning, the data are ready for Dr. Partida to review.
Fully automated versions can generate notes much faster. Jack Shilling, MD, MBA, an orthopedic surgeon at Cooper University Health Care, in Voorhees, N.J., uses DAX. A new feature called DAX Express – which uses OpenAI’s GPT-4 but no humans – provides him with a draft of his clinical notes in just seconds.
How accurate are AI notes?
The accuracy of those notes remains an open question, Dr. Garcia said – mostly because accuracy can be hard to define.
“If you asked five docs to write a note based on the same patient encounter, you’d get five different notes,” Dr. Garcia said. “That makes it hard to assess these technologies in a scientifically rigorous way.”
Still, the onus is on the physician to review the notes and edit them as needed, Dr. Garcia said. How light or heavy those edits are can depend on your unique preferences.
Dr. Shilling said he may need to lightly edit transcripts of his conversations with patients. “When someone tells me how long their knee hurts, slight variability in their transcribed words is tolerable,” he said. But for some things – such as physical exam notes and x-ray readings – he dictates directly into the device, speaking at a closer range and being less conversational, more exact in his speech.
Should you let patients know they’re being recorded?
The federal Health Insurance Portability and Accountability Act (HIPAA) does not require providers to inform patients that their face-to-face conversations are being recorded, said Daniel Lebovic, JD, corporate legal counsel at Compliancy Group, in Greenlawn, N.Y., a company that helps providers adhere to HIPAA rules.
But make sure you know the laws in your state and the policies at your health care practice. State laws may require providers to inform patients and to get patients’ consent in advance of being recorded.
All the doctors who spoke to this news organization said their patients are informed that they’ll be recorded and that they can opt out if they wish.
How much do AI scribes cost?
As the marketplace for these tools expands, companies are offering more products and services at different price points that target a range of organizations, from large health care systems to small private practices.
Price models vary, said Dr. Garcia. Some are based on the number of users, others on the number of notes, and still others on minutes.
Amazon’s HealthScribe is priced at 10 cents per minute. For 1,000 consultation transcripts per month, with each call averaging 15 minutes, it would take 15,000 minutes at a total cost of $1,500 for the month.
In general, the rapidly growing competition in this space could mean prices become more affordable, Dr. Garcia said. “It’s good that so many are getting into this game, because that means the price will come down and it will be a lot more accessible to everybody.”
A version of this article appeared on Medscape.com.
Andrea Partida, DO, an obstetrician and gynecologist in Enid, Okla., loves her new assistant.
The 15 or 20 minutes she used to spend on documentation for each patient visit is now 3. The 2-3 hours she’d spend charting outside clinic hours is maybe 1.
All that time saved allows her to see two to five more patients a day, provide better care to each patient, and get more involved in hospital leadership at Integris Health, where she works.
“I have a better work-life balance with my family,” Dr. Partida said. “I leave work at work and get home earlier.”
You’ve probably figured out the plot twist: Dr. Partida’s assistant is not a person – it’s artificial intelligence (AI).
Dr. Partida uses IRIS, a tool from OnPoint Healthcare Partners, part of a fast-growing niche of AI medical scribes designed to automate onerous data entry. The evolution of generative AI – specifically, large language models, such as ChatGPT – has led to a rapid explosion of these tools. Other companies in the space include Abridge, Ambience Healthcare, Augmedix, DeepScribe, Nuance (part of Microsoft), and Suki. The newest kid on the block, Amazon Web Services, announced the launch of HealthScribe in July.
These tools – some of which are already on the market, with more on the way – record patient visits and generate notes for treatment and billing. Earlier iterations combine AI with offsite human scribes who provide quality control. But more and more are fully automated, no human required. Some also offer video recording and foreign language translation.
The promise is alluring: Ease your workload and reclaim hours in your day so you can spend more time with patients or try that “work-life balance” thing you’ve heard so much about.
But do these tools fulfill that promise?
According to Dr. Partida and other doctors who spoke with this news organization, the answer is a resounding yes.
A tech solution for a tech problem
“I believe a lot of doctors see patients for free. They get paid to do paperwork,” said Anthony J. Mazzarelli, MD, JD, MBE, co-president and CEO of Cooper University Health Care, in Camden, N.J.
Indeed, for every hour U.S. clinicians spend with their patients, they may spend 2 more hours documenting in electronic health records (EHRs), estimates show. About half of doctors, especially those in primary care, report feeling burned out, and some 42% say they want to quit clinical practice.
Enter AI scribes.
“The holy grail in medicine right now is improving burnout while also maintaining or improving productivity and quality,” said Patricia Garcia, MD, associate clinical information officer for ambulatory care at Stanford (Calif.) Health Care. “These ambient digital scribes have the potential to do just that.”
While anyone can buy these products, their use has been mostly limited to pilot programs and early adopters so far, said Dr. Garcia, who has been helping to pilot Nuance’s digital scribe, DAX, at Stanford.
But that’s expected to change quickly. “I don’t think the time horizon is a decade,” Dr. Garcia said. “I think within a matter of 2 or 3 years, these tools will be pervasive throughout health care.”
Since introducing these tools at Cooper, “our doctors’ paperwork burden is significantly lighter,” said Dr. Mazzarelli, who decides which technologies Cooper should invest in and who monitors their results. In Cooper studies, physicians who used DAX more than half the time spent 43% less time working on notes.
“They spend more time connecting with their patients, talking with them, and looking them in the eye,” Dr. Mazzarelli said. That, in turn, seems to improve patient outcomes, reduce doctor burnout and turnover, and lower costs.
The AI scribes, by virtue of eliminating the distraction of note taking, also allow doctors to give their full attention to the patient. “The patient relationship is the most important aspect of medicine,” said Raul Ayala, MD, MHCM, a family medicine physician at Adventist Health, in Hanford, Calif., who uses Augmedix. The digital scribe “helps us strengthen that relationship.”
What’s it like to use an AI medical scribe?
The scribes feature hardware (typically a smartphone or tablet) and software built on automatic speech recognition, natural language processing, and machine learning. Download an app to your device, and you’re ready to go. Use it to record in-person or telehealth visits.
In the first week, a company may help train you to use the hardware and software. You’ll likely start by using it for a few patient visits per day, ramping up gradually. Dr. Partida said she was comfortable using the system for all her patients in 6 weeks.
Each day, Dr. Partida logs in to a dedicated smartphone or tablet, opens the app, and reviews her schedule, including details she needs to prepare for each patient.
At the start of each patient visit, Dr. Partida taps the app icon to begin recording and lays the device nearby. She can pause as needed. At the end of the visit, she taps the icon again to stop recording.
The AI listens, creates the note, and updates relevant data in the EHR. The note includes patient problems, assessment, treatment plan, patient history, orders, and tasks for staff, along with medications, referrals, and preauthorizations. A human scribe, who is also a physician, reviews the information for accuracy and edits it as needed. By the next morning, the data are ready for Dr. Partida to review.
Fully automated versions can generate notes much faster. Jack Shilling, MD, MBA, an orthopedic surgeon at Cooper University Health Care, in Voorhees, N.J., uses DAX. A new feature called DAX Express – which uses OpenAI’s GPT-4 but no humans – provides him with a draft of his clinical notes in just seconds.
How accurate are AI notes?
The accuracy of those notes remains an open question, Dr. Garcia said – mostly because accuracy can be hard to define.
“If you asked five docs to write a note based on the same patient encounter, you’d get five different notes,” Dr. Garcia said. “That makes it hard to assess these technologies in a scientifically rigorous way.”
Still, the onus is on the physician to review the notes and edit them as needed, Dr. Garcia said. How light or heavy those edits are can depend on your unique preferences.
Dr. Shilling said he may need to lightly edit transcripts of his conversations with patients. “When someone tells me how long their knee hurts, slight variability in their transcribed words is tolerable,” he said. But for some things – such as physical exam notes and x-ray readings – he dictates directly into the device, speaking at a closer range and being less conversational, more exact in his speech.
Should you let patients know they’re being recorded?
The federal Health Insurance Portability and Accountability Act (HIPAA) does not require providers to inform patients that their face-to-face conversations are being recorded, said Daniel Lebovic, JD, corporate legal counsel at Compliancy Group, in Greenlawn, N.Y., a company that helps providers adhere to HIPAA rules.
But make sure you know the laws in your state and the policies at your health care practice. State laws may require providers to inform patients and to get patients’ consent in advance of being recorded.
All the doctors who spoke to this news organization said their patients are informed that they’ll be recorded and that they can opt out if they wish.
How much do AI scribes cost?
As the marketplace for these tools expands, companies are offering more products and services at different price points that target a range of organizations, from large health care systems to small private practices.
Price models vary, said Dr. Garcia. Some are based on the number of users, others on the number of notes, and still others on minutes.
Amazon’s HealthScribe is priced at 10 cents per minute. For 1,000 consultation transcripts per month, with each call averaging 15 minutes, it would take 15,000 minutes at a total cost of $1,500 for the month.
In general, the rapidly growing competition in this space could mean prices become more affordable, Dr. Garcia said. “It’s good that so many are getting into this game, because that means the price will come down and it will be a lot more accessible to everybody.”
A version of this article appeared on Medscape.com.
Autoantibody against enteric nervous system protein linked to GI dysfunction in systemic sclerosis
(SSc), new research suggests. Researchers also found that gephyrin is expressed in the patient’s enteric nervous system (ENS), which regulates gut motility.
“While there are many antibodies that are helpful in identifying patients at risk for extraintestinal complications of this disease, markers that identify patients at higher risk for gastrointestinal complications are limited. Furthermore, the biological mechanisms that cause and perpetuate the progression of gastrointestinal disease in scleroderma are not well understood, making it challenging to distinguish between patients whose gastrointestinal disease will progress from those whose GI disease will remain stable/mild,” Zsuzsanna H. McMahan, MD, MHS, told this news organization in an email. Dr. McMahan is co–first author on the study along with Subhash Kulkarni, PhD. They conducted the research with colleagues when they both worked at Johns Hopkins University in Baltimore, Md.
When asked for comment, Kimberly Lakin, MD, MS, assistant professor of medicine at Weill Cornell Medicine and a rheumatologist at Hospital for Special Surgery, New York, called the study “interesting and novel.”
“Not only did [antigephyrin antibodies] correlate with the presence of lower GI symptoms, but also higher levels of antibodies correlated with worse lower GI symptoms. This suggests that not only could this antibody be used to predict who may have constipation and potentially need more aggressive GI interventions, but it may also be useful in quantifying GI severity in systemic sclerosis, although more research is still needed,” said Dr. Lakin, who was not involved with the research.
The study was published online in Arthritis & Rheumatology.
In the cross-sectional study, researchers identified gephyrin as an autoantigen in sera from a single patient with SSc by isolating it from immunoprecipitations performed with murine myenteric plexus neuron lysates, and then characterizing it by mass spectrometry and validating it in further assays. That patient had GI dysfunction but no defined SSc-associated autoantibodies.
Dr. McMahan and colleagues then investigated the prevalence of the autoantibody by screening the sera of 188 patients with SSc who presented consecutively to the Johns Hopkins Scleroderma Center between April 2016 and August 2017, as well as 40 controls, and compared GI symptom severity between antibody-positive and antibody-negative patients with SSc.
A total of 16 (8.5%) of the 188 patients with SSc had antigephyrin antibodies, compared with none of the controls. Of these 16 patients, 4 had no other defined SSc antibodies. In the SSc cohort, severe constipation was more common in antigephyrin antibody–positive patients, compared with antibody-negative patients (46% vs. 15%). Antibody-positive patients also had higher constipation scores, and severe distension and bloating occurred in the antibody-positive group more than twice as often (54% vs. 25%).
Patients with severe constipation, distention, and bloating had higher antigephyrin antibody levels. After adjusting for confounders such as disease duration, patients with severe constipation were nearly five times as likely (odds ratio, 4.74; P = .010) to be antigephyrin antibody–positive, and patients with severe distention and bloating were nearly four times as likely (OR, 3.71; P = .027) to be antibody-positive.
Last, the authors showed via immunohistochemistry that gephyrin is expressed in the myenteric ganglia of human GI tissue.
“Gastrointestinal function is highly regulated by the ENS, so it is interesting that antibodies that target a protein expressed by ENS cells (gephyrin) were identified in patients with scleroderma who have severe lower bowel dysfunction,” said Dr. McMahan, who is associate professor in the division of rheumatology and codirector of the scleroderma program at the University of Texas Health Science Center at Houston. “Gephyrin is a key mediator of normal communications between nerves in the gut, so it is tantalizing to speculate that autoimmune-mediated disruption (e.g., an inhibitory or blocking antibody) in neural (ENS) communications in the gut might lead to impaired bowel transit and prominent constipation.”
The study was supported by grants from the National Institute of Arthritis and Musculoskeletal and Skin Diseases and other NIH grants, as well as the Scleroderma Research Foundation, Rheumatology Research Foundation, Jerome L. Greene Foundation, Martha McCrory Professorship, and Chresanthe Stauraluakis Memorial Discovery Fund. The study authors and Dr. Lakin report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
(SSc), new research suggests. Researchers also found that gephyrin is expressed in the patient’s enteric nervous system (ENS), which regulates gut motility.
“While there are many antibodies that are helpful in identifying patients at risk for extraintestinal complications of this disease, markers that identify patients at higher risk for gastrointestinal complications are limited. Furthermore, the biological mechanisms that cause and perpetuate the progression of gastrointestinal disease in scleroderma are not well understood, making it challenging to distinguish between patients whose gastrointestinal disease will progress from those whose GI disease will remain stable/mild,” Zsuzsanna H. McMahan, MD, MHS, told this news organization in an email. Dr. McMahan is co–first author on the study along with Subhash Kulkarni, PhD. They conducted the research with colleagues when they both worked at Johns Hopkins University in Baltimore, Md.
When asked for comment, Kimberly Lakin, MD, MS, assistant professor of medicine at Weill Cornell Medicine and a rheumatologist at Hospital for Special Surgery, New York, called the study “interesting and novel.”
“Not only did [antigephyrin antibodies] correlate with the presence of lower GI symptoms, but also higher levels of antibodies correlated with worse lower GI symptoms. This suggests that not only could this antibody be used to predict who may have constipation and potentially need more aggressive GI interventions, but it may also be useful in quantifying GI severity in systemic sclerosis, although more research is still needed,” said Dr. Lakin, who was not involved with the research.
The study was published online in Arthritis & Rheumatology.
In the cross-sectional study, researchers identified gephyrin as an autoantigen in sera from a single patient with SSc by isolating it from immunoprecipitations performed with murine myenteric plexus neuron lysates, and then characterizing it by mass spectrometry and validating it in further assays. That patient had GI dysfunction but no defined SSc-associated autoantibodies.
Dr. McMahan and colleagues then investigated the prevalence of the autoantibody by screening the sera of 188 patients with SSc who presented consecutively to the Johns Hopkins Scleroderma Center between April 2016 and August 2017, as well as 40 controls, and compared GI symptom severity between antibody-positive and antibody-negative patients with SSc.
A total of 16 (8.5%) of the 188 patients with SSc had antigephyrin antibodies, compared with none of the controls. Of these 16 patients, 4 had no other defined SSc antibodies. In the SSc cohort, severe constipation was more common in antigephyrin antibody–positive patients, compared with antibody-negative patients (46% vs. 15%). Antibody-positive patients also had higher constipation scores, and severe distension and bloating occurred in the antibody-positive group more than twice as often (54% vs. 25%).
Patients with severe constipation, distention, and bloating had higher antigephyrin antibody levels. After adjusting for confounders such as disease duration, patients with severe constipation were nearly five times as likely (odds ratio, 4.74; P = .010) to be antigephyrin antibody–positive, and patients with severe distention and bloating were nearly four times as likely (OR, 3.71; P = .027) to be antibody-positive.
Last, the authors showed via immunohistochemistry that gephyrin is expressed in the myenteric ganglia of human GI tissue.
“Gastrointestinal function is highly regulated by the ENS, so it is interesting that antibodies that target a protein expressed by ENS cells (gephyrin) were identified in patients with scleroderma who have severe lower bowel dysfunction,” said Dr. McMahan, who is associate professor in the division of rheumatology and codirector of the scleroderma program at the University of Texas Health Science Center at Houston. “Gephyrin is a key mediator of normal communications between nerves in the gut, so it is tantalizing to speculate that autoimmune-mediated disruption (e.g., an inhibitory or blocking antibody) in neural (ENS) communications in the gut might lead to impaired bowel transit and prominent constipation.”
The study was supported by grants from the National Institute of Arthritis and Musculoskeletal and Skin Diseases and other NIH grants, as well as the Scleroderma Research Foundation, Rheumatology Research Foundation, Jerome L. Greene Foundation, Martha McCrory Professorship, and Chresanthe Stauraluakis Memorial Discovery Fund. The study authors and Dr. Lakin report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
(SSc), new research suggests. Researchers also found that gephyrin is expressed in the patient’s enteric nervous system (ENS), which regulates gut motility.
“While there are many antibodies that are helpful in identifying patients at risk for extraintestinal complications of this disease, markers that identify patients at higher risk for gastrointestinal complications are limited. Furthermore, the biological mechanisms that cause and perpetuate the progression of gastrointestinal disease in scleroderma are not well understood, making it challenging to distinguish between patients whose gastrointestinal disease will progress from those whose GI disease will remain stable/mild,” Zsuzsanna H. McMahan, MD, MHS, told this news organization in an email. Dr. McMahan is co–first author on the study along with Subhash Kulkarni, PhD. They conducted the research with colleagues when they both worked at Johns Hopkins University in Baltimore, Md.
When asked for comment, Kimberly Lakin, MD, MS, assistant professor of medicine at Weill Cornell Medicine and a rheumatologist at Hospital for Special Surgery, New York, called the study “interesting and novel.”
“Not only did [antigephyrin antibodies] correlate with the presence of lower GI symptoms, but also higher levels of antibodies correlated with worse lower GI symptoms. This suggests that not only could this antibody be used to predict who may have constipation and potentially need more aggressive GI interventions, but it may also be useful in quantifying GI severity in systemic sclerosis, although more research is still needed,” said Dr. Lakin, who was not involved with the research.
The study was published online in Arthritis & Rheumatology.
In the cross-sectional study, researchers identified gephyrin as an autoantigen in sera from a single patient with SSc by isolating it from immunoprecipitations performed with murine myenteric plexus neuron lysates, and then characterizing it by mass spectrometry and validating it in further assays. That patient had GI dysfunction but no defined SSc-associated autoantibodies.
Dr. McMahan and colleagues then investigated the prevalence of the autoantibody by screening the sera of 188 patients with SSc who presented consecutively to the Johns Hopkins Scleroderma Center between April 2016 and August 2017, as well as 40 controls, and compared GI symptom severity between antibody-positive and antibody-negative patients with SSc.
A total of 16 (8.5%) of the 188 patients with SSc had antigephyrin antibodies, compared with none of the controls. Of these 16 patients, 4 had no other defined SSc antibodies. In the SSc cohort, severe constipation was more common in antigephyrin antibody–positive patients, compared with antibody-negative patients (46% vs. 15%). Antibody-positive patients also had higher constipation scores, and severe distension and bloating occurred in the antibody-positive group more than twice as often (54% vs. 25%).
Patients with severe constipation, distention, and bloating had higher antigephyrin antibody levels. After adjusting for confounders such as disease duration, patients with severe constipation were nearly five times as likely (odds ratio, 4.74; P = .010) to be antigephyrin antibody–positive, and patients with severe distention and bloating were nearly four times as likely (OR, 3.71; P = .027) to be antibody-positive.
Last, the authors showed via immunohistochemistry that gephyrin is expressed in the myenteric ganglia of human GI tissue.
“Gastrointestinal function is highly regulated by the ENS, so it is interesting that antibodies that target a protein expressed by ENS cells (gephyrin) were identified in patients with scleroderma who have severe lower bowel dysfunction,” said Dr. McMahan, who is associate professor in the division of rheumatology and codirector of the scleroderma program at the University of Texas Health Science Center at Houston. “Gephyrin is a key mediator of normal communications between nerves in the gut, so it is tantalizing to speculate that autoimmune-mediated disruption (e.g., an inhibitory or blocking antibody) in neural (ENS) communications in the gut might lead to impaired bowel transit and prominent constipation.”
The study was supported by grants from the National Institute of Arthritis and Musculoskeletal and Skin Diseases and other NIH grants, as well as the Scleroderma Research Foundation, Rheumatology Research Foundation, Jerome L. Greene Foundation, Martha McCrory Professorship, and Chresanthe Stauraluakis Memorial Discovery Fund. The study authors and Dr. Lakin report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM ARTHRITIS & RHEUMATOLOGY
Drug name confusion: More than 80 new drug pairs added to the list
Zolpidem (Ambien) is a well-known sedative for sleep. Letairis (Ambrisentan) is a vasodilator for the treatment of pulmonary arterial hypertension. Citalopram (Celexa) is an antidepressant; escitalopram (Lexapro) is prescribed for anxiety and depression.
The aim is to increase awareness about the potential for a serious medication mistake when the wrong drug is given because of drug names that look and sound similar.
Awareness of these drug names, however, is just the first step in preventing medication mistakes. Health care providers should take a number of other steps as well, experts said.
ISMP launched its confusing drug names list, previously called look-alike, sound-alike (LASA) drugs, in 2008. The new list is an update of the 2019 version, said Michael J. Gaunt, PharmD, senior manager of error reporting programs for the ISMP, which focuses on the prevention of medication mistakes. The new entries were chosen on the basis of a number of factors, including ISMP’s analysis of recent medication mishap reports that were submitted to it.
The ISMP list now includes about 528 drug pairs, Dr. Gaunt said. The list is long, he said, partly because each pair is listed twice, so readers can cross reference. For instance, hydralazine and hydroxyzine are listed in one entry in the list, and hydroxyzine and hydralazine are listed in another.
Brand Institute in Miami has named, among other drugs, Entresto, Rybelsus, and Lunesta. The regulatory arm of the company, the Drug Safety Institute, “considers drug names that have been confused as an important part of our comprehensive drug name assessments,” Todd Bridges, global president of the institute, said in an emailed statement. Information on the confusing drug names are incorporated into the company’s proprietary algorithm and is used when developing brand names for drugs. “We continually update this algorithm as new drug names that are often confused are identified,” Mr. Bridges said.
Confusing drug names: Ongoing issue
The length of the list, as well as the latest additions, are not surprising, said Mary Ann Kliethermes, PharmD, director of medication safety and quality for the American Society of Health-System Pharmacists, a membership organization of about 60,000 pharmacists who practice in inpatient and outpatient settings.
“I’ve been in practice over 45 years,” she said, “and this has been a problem ever since I have been in practice.” The sheer volume of new drugs is one reason, she said. From 2013 through 2022, the U.S. Food and Drug Administration approved an average of 43 novel drugs per year, according to a report from its Center for Drug Evaluation and Research. “Since the 90s, this [confusion about similar drug names] has happened,” Dr. Kliethermes said.
According to a 2023 report, about 7,000-9,000 people die each year in the United States as the result of a medication error. However, it’s impossible to say for sure what percentage of those errors involve name confusion, Dr. Gaunt said.
Not all the mistakes are reported. Some that are reported are dramatic and deadly. In 2022, a Tennessee nurse was convicted of gross neglect and negligent homicide. She was sentenced to 3 years’ probation after she mistakenly gave vercuronium, an anesthetic agent, instead of the sedative Versed to a patient, and the woman died.
Updated list: A closer look
Many of the new drug pairs that are listed in the update are cephalosporins, said Dr. Kliethermes, who reviewed the new list for this news organization. In all, 20 of the latest 82 additions are cephalosporins. These include drugs such as cefazolin, which can be confused with cefotetan, and vice versa. These drugs have been around since the 1980s, she said, but “they needed to be on there.” Even in the 1980s, it was becoming difficult to differentiate them, and there were fewer drugs in that class then, she said.
Influenza vaccines made the new list, too. Fluzone High-Dose Quadrivalent can be confused with fluzone quadrivalent. Other new additions: hydrochlorothiazide and hydroxychloroquine, Lasik and Wakix, Pitressin and Pitocin, Remeron and Rozerem.
Beyond the list
While it’s not possible to pinpoint how big a problem name confusion is in causing medication mistakes, “it is certainly still an issue,” Dr. Gaunt said. A variety of practices can reduce that risk substantially, Dr. Gaunt and Dr. Kliethermes agreed.
Tall-man lettering. Both the FDA and the ISMP recommend the use of so-called tall-man lettering (TML), which involves the use of uppercase letters, sometimes in boldface, to distinguish similar names on product labels and elsewhere. Examples include vinBLAStine and vinCRIStine.
Electronic prescribing. “It eliminates the risk of handwriting confusion,” Dr. Gaunt said. However, electronic prescribing can have a downside, Dr. Kliethermes said. When ordering medication, a person may type in a few letters and may then be presented with a prompt that lists several drug names, and it can be easy to click the wrong one. For that reason, ISMP and other experts recommend typing at least five letters when searching for a medication in an electronic system.
Use both brand and generic names on labels and prescriptions.
Write the indication. That can serve as a double check. If a prescription for Ambien says “For sleep,” there’s probably less risk of filling a prescription for ambrisentan, the vasodilator.
Smart formulary additions. When hospitals add medications to their formularies, “part of that formulary assessment should include looking at the potential risk for errors,” Dr. Gaunt said. This involves keeping an eye out for confusing names and similar packaging. “Do that analysis up front and put in strategies to minimize that. Maybe you look for a different drug [for the same use] that has a different name.” Or choose a different manufacturer, so the medication would at least have a different container.
Use bar code scanning. Suppose a pharmacist goes to the shelf and pulls the wrong drug. “Bar code scanning provides the opportunity to catch the error,” Dr. Gaunt said. Many community pharmacies now have bar code scanning. ISMP just issued best practices for community pharmacies, Dr. Gaunt said, and these include the use of bar code scanning and other measures.
Educate consumers. Health care providers can educate consumers on how to minimize the risk of getting the wrong drug, Dr. Gaunt said. When patients are picking up a prescription, suggest they look at the container label; if it looks different from previous prescriptions of the same medicine, ask the pharmacist for an explanation. Some patients just pass it off, Dr. Gaunt said, figuring the pharmacist or health plan switched manufacturers of their medication.
Access the list. The entire list is on the ISMP site and is accessible after free registration.
Goal: Preventing confusion
The FDA has provided guidance for industry on naming drugs not yet approved so that the proposed names are not too similar in sound or appearance to those already on the market. Included in the lengthy document are checklists, such as, “Across a range of dialects, are the names consistently pronounced differently?” and “Are the lengths of the names dissimilar when scripted?” (Lengths are considered different if they differ by two or more letters.)
The FDA also offers the phonetic and orthographic computer analysis (POCA) program, a software tool that employs an advanced algorithm to evaluate similarities between two drug names. The data sources are updated regularly as new drugs are approved.
Liability update
The problem may be decreasing. In a 2020 report, researchers used pharmacists’ professional liability claim data from the Healthcare Providers Service Organization. They compared 2018 data on claims with 2013 data. The percentage of claims associated with wrong drug dispensing errors declined from 43.8% in 2013 to 36.8% in 2018. Wrong dose claims also declined, from 31.5% to 15.3%.
These researchers concluded that technology and automation have contributed to the prevention of medication errors caused by the use of the wrong drug and the wrong dose, but mistakes continue, owing to system and human errors.
A version of this article first appeared on Medscape.com.
Zolpidem (Ambien) is a well-known sedative for sleep. Letairis (Ambrisentan) is a vasodilator for the treatment of pulmonary arterial hypertension. Citalopram (Celexa) is an antidepressant; escitalopram (Lexapro) is prescribed for anxiety and depression.
The aim is to increase awareness about the potential for a serious medication mistake when the wrong drug is given because of drug names that look and sound similar.
Awareness of these drug names, however, is just the first step in preventing medication mistakes. Health care providers should take a number of other steps as well, experts said.
ISMP launched its confusing drug names list, previously called look-alike, sound-alike (LASA) drugs, in 2008. The new list is an update of the 2019 version, said Michael J. Gaunt, PharmD, senior manager of error reporting programs for the ISMP, which focuses on the prevention of medication mistakes. The new entries were chosen on the basis of a number of factors, including ISMP’s analysis of recent medication mishap reports that were submitted to it.
The ISMP list now includes about 528 drug pairs, Dr. Gaunt said. The list is long, he said, partly because each pair is listed twice, so readers can cross reference. For instance, hydralazine and hydroxyzine are listed in one entry in the list, and hydroxyzine and hydralazine are listed in another.
Brand Institute in Miami has named, among other drugs, Entresto, Rybelsus, and Lunesta. The regulatory arm of the company, the Drug Safety Institute, “considers drug names that have been confused as an important part of our comprehensive drug name assessments,” Todd Bridges, global president of the institute, said in an emailed statement. Information on the confusing drug names are incorporated into the company’s proprietary algorithm and is used when developing brand names for drugs. “We continually update this algorithm as new drug names that are often confused are identified,” Mr. Bridges said.
Confusing drug names: Ongoing issue
The length of the list, as well as the latest additions, are not surprising, said Mary Ann Kliethermes, PharmD, director of medication safety and quality for the American Society of Health-System Pharmacists, a membership organization of about 60,000 pharmacists who practice in inpatient and outpatient settings.
“I’ve been in practice over 45 years,” she said, “and this has been a problem ever since I have been in practice.” The sheer volume of new drugs is one reason, she said. From 2013 through 2022, the U.S. Food and Drug Administration approved an average of 43 novel drugs per year, according to a report from its Center for Drug Evaluation and Research. “Since the 90s, this [confusion about similar drug names] has happened,” Dr. Kliethermes said.
According to a 2023 report, about 7,000-9,000 people die each year in the United States as the result of a medication error. However, it’s impossible to say for sure what percentage of those errors involve name confusion, Dr. Gaunt said.
Not all the mistakes are reported. Some that are reported are dramatic and deadly. In 2022, a Tennessee nurse was convicted of gross neglect and negligent homicide. She was sentenced to 3 years’ probation after she mistakenly gave vercuronium, an anesthetic agent, instead of the sedative Versed to a patient, and the woman died.
Updated list: A closer look
Many of the new drug pairs that are listed in the update are cephalosporins, said Dr. Kliethermes, who reviewed the new list for this news organization. In all, 20 of the latest 82 additions are cephalosporins. These include drugs such as cefazolin, which can be confused with cefotetan, and vice versa. These drugs have been around since the 1980s, she said, but “they needed to be on there.” Even in the 1980s, it was becoming difficult to differentiate them, and there were fewer drugs in that class then, she said.
Influenza vaccines made the new list, too. Fluzone High-Dose Quadrivalent can be confused with fluzone quadrivalent. Other new additions: hydrochlorothiazide and hydroxychloroquine, Lasik and Wakix, Pitressin and Pitocin, Remeron and Rozerem.
Beyond the list
While it’s not possible to pinpoint how big a problem name confusion is in causing medication mistakes, “it is certainly still an issue,” Dr. Gaunt said. A variety of practices can reduce that risk substantially, Dr. Gaunt and Dr. Kliethermes agreed.
Tall-man lettering. Both the FDA and the ISMP recommend the use of so-called tall-man lettering (TML), which involves the use of uppercase letters, sometimes in boldface, to distinguish similar names on product labels and elsewhere. Examples include vinBLAStine and vinCRIStine.
Electronic prescribing. “It eliminates the risk of handwriting confusion,” Dr. Gaunt said. However, electronic prescribing can have a downside, Dr. Kliethermes said. When ordering medication, a person may type in a few letters and may then be presented with a prompt that lists several drug names, and it can be easy to click the wrong one. For that reason, ISMP and other experts recommend typing at least five letters when searching for a medication in an electronic system.
Use both brand and generic names on labels and prescriptions.
Write the indication. That can serve as a double check. If a prescription for Ambien says “For sleep,” there’s probably less risk of filling a prescription for ambrisentan, the vasodilator.
Smart formulary additions. When hospitals add medications to their formularies, “part of that formulary assessment should include looking at the potential risk for errors,” Dr. Gaunt said. This involves keeping an eye out for confusing names and similar packaging. “Do that analysis up front and put in strategies to minimize that. Maybe you look for a different drug [for the same use] that has a different name.” Or choose a different manufacturer, so the medication would at least have a different container.
Use bar code scanning. Suppose a pharmacist goes to the shelf and pulls the wrong drug. “Bar code scanning provides the opportunity to catch the error,” Dr. Gaunt said. Many community pharmacies now have bar code scanning. ISMP just issued best practices for community pharmacies, Dr. Gaunt said, and these include the use of bar code scanning and other measures.
Educate consumers. Health care providers can educate consumers on how to minimize the risk of getting the wrong drug, Dr. Gaunt said. When patients are picking up a prescription, suggest they look at the container label; if it looks different from previous prescriptions of the same medicine, ask the pharmacist for an explanation. Some patients just pass it off, Dr. Gaunt said, figuring the pharmacist or health plan switched manufacturers of their medication.
Access the list. The entire list is on the ISMP site and is accessible after free registration.
Goal: Preventing confusion
The FDA has provided guidance for industry on naming drugs not yet approved so that the proposed names are not too similar in sound or appearance to those already on the market. Included in the lengthy document are checklists, such as, “Across a range of dialects, are the names consistently pronounced differently?” and “Are the lengths of the names dissimilar when scripted?” (Lengths are considered different if they differ by two or more letters.)
The FDA also offers the phonetic and orthographic computer analysis (POCA) program, a software tool that employs an advanced algorithm to evaluate similarities between two drug names. The data sources are updated regularly as new drugs are approved.
Liability update
The problem may be decreasing. In a 2020 report, researchers used pharmacists’ professional liability claim data from the Healthcare Providers Service Organization. They compared 2018 data on claims with 2013 data. The percentage of claims associated with wrong drug dispensing errors declined from 43.8% in 2013 to 36.8% in 2018. Wrong dose claims also declined, from 31.5% to 15.3%.
These researchers concluded that technology and automation have contributed to the prevention of medication errors caused by the use of the wrong drug and the wrong dose, but mistakes continue, owing to system and human errors.
A version of this article first appeared on Medscape.com.
Zolpidem (Ambien) is a well-known sedative for sleep. Letairis (Ambrisentan) is a vasodilator for the treatment of pulmonary arterial hypertension. Citalopram (Celexa) is an antidepressant; escitalopram (Lexapro) is prescribed for anxiety and depression.
The aim is to increase awareness about the potential for a serious medication mistake when the wrong drug is given because of drug names that look and sound similar.
Awareness of these drug names, however, is just the first step in preventing medication mistakes. Health care providers should take a number of other steps as well, experts said.
ISMP launched its confusing drug names list, previously called look-alike, sound-alike (LASA) drugs, in 2008. The new list is an update of the 2019 version, said Michael J. Gaunt, PharmD, senior manager of error reporting programs for the ISMP, which focuses on the prevention of medication mistakes. The new entries were chosen on the basis of a number of factors, including ISMP’s analysis of recent medication mishap reports that were submitted to it.
The ISMP list now includes about 528 drug pairs, Dr. Gaunt said. The list is long, he said, partly because each pair is listed twice, so readers can cross reference. For instance, hydralazine and hydroxyzine are listed in one entry in the list, and hydroxyzine and hydralazine are listed in another.
Brand Institute in Miami has named, among other drugs, Entresto, Rybelsus, and Lunesta. The regulatory arm of the company, the Drug Safety Institute, “considers drug names that have been confused as an important part of our comprehensive drug name assessments,” Todd Bridges, global president of the institute, said in an emailed statement. Information on the confusing drug names are incorporated into the company’s proprietary algorithm and is used when developing brand names for drugs. “We continually update this algorithm as new drug names that are often confused are identified,” Mr. Bridges said.
Confusing drug names: Ongoing issue
The length of the list, as well as the latest additions, are not surprising, said Mary Ann Kliethermes, PharmD, director of medication safety and quality for the American Society of Health-System Pharmacists, a membership organization of about 60,000 pharmacists who practice in inpatient and outpatient settings.
“I’ve been in practice over 45 years,” she said, “and this has been a problem ever since I have been in practice.” The sheer volume of new drugs is one reason, she said. From 2013 through 2022, the U.S. Food and Drug Administration approved an average of 43 novel drugs per year, according to a report from its Center for Drug Evaluation and Research. “Since the 90s, this [confusion about similar drug names] has happened,” Dr. Kliethermes said.
According to a 2023 report, about 7,000-9,000 people die each year in the United States as the result of a medication error. However, it’s impossible to say for sure what percentage of those errors involve name confusion, Dr. Gaunt said.
Not all the mistakes are reported. Some that are reported are dramatic and deadly. In 2022, a Tennessee nurse was convicted of gross neglect and negligent homicide. She was sentenced to 3 years’ probation after she mistakenly gave vercuronium, an anesthetic agent, instead of the sedative Versed to a patient, and the woman died.
Updated list: A closer look
Many of the new drug pairs that are listed in the update are cephalosporins, said Dr. Kliethermes, who reviewed the new list for this news organization. In all, 20 of the latest 82 additions are cephalosporins. These include drugs such as cefazolin, which can be confused with cefotetan, and vice versa. These drugs have been around since the 1980s, she said, but “they needed to be on there.” Even in the 1980s, it was becoming difficult to differentiate them, and there were fewer drugs in that class then, she said.
Influenza vaccines made the new list, too. Fluzone High-Dose Quadrivalent can be confused with fluzone quadrivalent. Other new additions: hydrochlorothiazide and hydroxychloroquine, Lasik and Wakix, Pitressin and Pitocin, Remeron and Rozerem.
Beyond the list
While it’s not possible to pinpoint how big a problem name confusion is in causing medication mistakes, “it is certainly still an issue,” Dr. Gaunt said. A variety of practices can reduce that risk substantially, Dr. Gaunt and Dr. Kliethermes agreed.
Tall-man lettering. Both the FDA and the ISMP recommend the use of so-called tall-man lettering (TML), which involves the use of uppercase letters, sometimes in boldface, to distinguish similar names on product labels and elsewhere. Examples include vinBLAStine and vinCRIStine.
Electronic prescribing. “It eliminates the risk of handwriting confusion,” Dr. Gaunt said. However, electronic prescribing can have a downside, Dr. Kliethermes said. When ordering medication, a person may type in a few letters and may then be presented with a prompt that lists several drug names, and it can be easy to click the wrong one. For that reason, ISMP and other experts recommend typing at least five letters when searching for a medication in an electronic system.
Use both brand and generic names on labels and prescriptions.
Write the indication. That can serve as a double check. If a prescription for Ambien says “For sleep,” there’s probably less risk of filling a prescription for ambrisentan, the vasodilator.
Smart formulary additions. When hospitals add medications to their formularies, “part of that formulary assessment should include looking at the potential risk for errors,” Dr. Gaunt said. This involves keeping an eye out for confusing names and similar packaging. “Do that analysis up front and put in strategies to minimize that. Maybe you look for a different drug [for the same use] that has a different name.” Or choose a different manufacturer, so the medication would at least have a different container.
Use bar code scanning. Suppose a pharmacist goes to the shelf and pulls the wrong drug. “Bar code scanning provides the opportunity to catch the error,” Dr. Gaunt said. Many community pharmacies now have bar code scanning. ISMP just issued best practices for community pharmacies, Dr. Gaunt said, and these include the use of bar code scanning and other measures.
Educate consumers. Health care providers can educate consumers on how to minimize the risk of getting the wrong drug, Dr. Gaunt said. When patients are picking up a prescription, suggest they look at the container label; if it looks different from previous prescriptions of the same medicine, ask the pharmacist for an explanation. Some patients just pass it off, Dr. Gaunt said, figuring the pharmacist or health plan switched manufacturers of their medication.
Access the list. The entire list is on the ISMP site and is accessible after free registration.
Goal: Preventing confusion
The FDA has provided guidance for industry on naming drugs not yet approved so that the proposed names are not too similar in sound or appearance to those already on the market. Included in the lengthy document are checklists, such as, “Across a range of dialects, are the names consistently pronounced differently?” and “Are the lengths of the names dissimilar when scripted?” (Lengths are considered different if they differ by two or more letters.)
The FDA also offers the phonetic and orthographic computer analysis (POCA) program, a software tool that employs an advanced algorithm to evaluate similarities between two drug names. The data sources are updated regularly as new drugs are approved.
Liability update
The problem may be decreasing. In a 2020 report, researchers used pharmacists’ professional liability claim data from the Healthcare Providers Service Organization. They compared 2018 data on claims with 2013 data. The percentage of claims associated with wrong drug dispensing errors declined from 43.8% in 2013 to 36.8% in 2018. Wrong dose claims also declined, from 31.5% to 15.3%.
These researchers concluded that technology and automation have contributed to the prevention of medication errors caused by the use of the wrong drug and the wrong dose, but mistakes continue, owing to system and human errors.
A version of this article first appeared on Medscape.com.
Low-dose steroids may not increase cardiovascular risk in rheumatoid arthritis
A daily prednisolone dose of 5 mg or higher is associated with increased risk for major adverse cardiovascular events (MACE) among patients with rheumatoid arthritis (RA), data suggest. Patients taking daily doses below this threshold did not appear to have an increased risk of MACE, compared with those not taking glucocorticoids (GCs).
Other studies of GCs and CV risk among RA patients have yielded conflicting results, especially for low-dose GCs. Findings from a 2020 study published in PLOS Medicine suggested that patients who had several immune-mediated inflammatory diseases – including RA – and who took less than a 5-mg prednisolone-equivalent dose daily had 74% higher risk for all-cause CVD, compared with nonusers. But results from a 2021 study published in Annals of the Rheumatic Diseases suggested that a daily prednisone dose of 4 mg or less did not increase cardiovascular events over a period of 6 months to 1 year.
These contradictory results were “primarily due to incomplete control of confounding variables, such as failure to adjust for C-reactive protein (CRP) levels,” Dr. Tam said. “Our study aimed to use a big data analytical approach to determine the effect of systemic GC dose and duration on the risk of major adverse cardiovascular events in patients with RA, while controlling for systemic inflammation, traditional CV risk factors, and other therapies.”
Is there a ‘safe’ dose for glucocorticoids?
To analyze this relationship, Dr. Lam and colleagues used the Hospital Authority Data Collaboration Laboratory, a citywide health care database. The investigators recruited patients with RA who had no history of MACE from 2006 to 2015 and followed them until the end of 2018. The primary outcome was the first occurrence of a MACE, defined as a composite of myocardial infarction (MI), unstable angina, ischemic or hemorrhagic cerebrovascular accident, transient ischemic attack, and CV death.
The study was published in Annals of the Rheumatic Diseases.
The analysis included 12,233 patients with RA and had over 105,826 person-years of follow-up. The average follow-up time was 8.7 years. During the study period, 860 patients had their first MACE. After controlling for confounding factors, a daily prednisolone dose of 5 mg or higher doubled the risk for MACE, compared with GC nonusers. MACE risk increased by 7% per month.
Long-term glucocorticoid use discouraged
Daily doses of less than 5 mg were not associated with higher MACE risk, but more research is necessary to understand whether these low doses are clinically efficacious, Dr. Tam said. “The study results suggest that a very-low-dose GC (less than 5 mg prednisolone daily) may be cardiovascular risk–neutral. However, further evaluation is needed to determine whether this dose is therapeutic. Other potential side effects, such as bone loss, increased infection risk, dyslipidemia, and hyperglycemia, should also be considered.”
Both the American College of Rheumatology and the European Alliance of Associations for Rheumatology acknowledge that short-term GCs may be necessary for some RA patients, but they emphasize using the smallest necessary dose for the shortest period possible because of the known toxicity of GCs.
“We recommend stopping GCs as soon as it is clinically feasible, in line with previous recommendations, until these issues are investigated further,” Dr. Tam added.
Dr. Bartels agreed that long-term use of GCs should be avoided if possible, even at lower doses, because although CV risk may be less of an issue, studies have shown an increased risk for infection even at GC doses of less than 5 mg a day.
How might risk increase with dose?
While the study showed a distinct difference in risk with doses of prednisolone higher and lower than 5 mg, more information on how risk increases with dose could be useful, said Beth Wallace, MD, an assistant professor in internal medicine at the University of Michigan, Ann Arbor, and a staff rheumatologist at the VA Ann Arbor Healthcare Center. She was also unaffiliated with the research. “If someone is on 5-10 mg ... how much better is that than being on 10-20 mg or being on 20-30 mg?” she asked. While these study findings are “very important,” she said, it would be useful to know the risk associated with 7.5 mg vs. a higher dose.
But even in this relatively healthy population in Hong Kong, “taking more than 5 mg of prednisolone doubles the risk of cardiovascular disease,” Dr. Wallace added. This is important for clinicians to know, especially if they are more cautious about prescribing steroids to older or sicker patients but are “using [the drugs] a little more indiscriminately in younger people and healthier people.”
The study did not receive outside funding. Dr. Tam and Dr. Bartels report no relevant financial relationships. Dr. Wallace has received a grant from the Department of Veterans Affairs Administration to study steroid tapering in RA.
A version of this article first appeared on Medscape.com.
A daily prednisolone dose of 5 mg or higher is associated with increased risk for major adverse cardiovascular events (MACE) among patients with rheumatoid arthritis (RA), data suggest. Patients taking daily doses below this threshold did not appear to have an increased risk of MACE, compared with those not taking glucocorticoids (GCs).
Other studies of GCs and CV risk among RA patients have yielded conflicting results, especially for low-dose GCs. Findings from a 2020 study published in PLOS Medicine suggested that patients who had several immune-mediated inflammatory diseases – including RA – and who took less than a 5-mg prednisolone-equivalent dose daily had 74% higher risk for all-cause CVD, compared with nonusers. But results from a 2021 study published in Annals of the Rheumatic Diseases suggested that a daily prednisone dose of 4 mg or less did not increase cardiovascular events over a period of 6 months to 1 year.
These contradictory results were “primarily due to incomplete control of confounding variables, such as failure to adjust for C-reactive protein (CRP) levels,” Dr. Tam said. “Our study aimed to use a big data analytical approach to determine the effect of systemic GC dose and duration on the risk of major adverse cardiovascular events in patients with RA, while controlling for systemic inflammation, traditional CV risk factors, and other therapies.”
Is there a ‘safe’ dose for glucocorticoids?
To analyze this relationship, Dr. Lam and colleagues used the Hospital Authority Data Collaboration Laboratory, a citywide health care database. The investigators recruited patients with RA who had no history of MACE from 2006 to 2015 and followed them until the end of 2018. The primary outcome was the first occurrence of a MACE, defined as a composite of myocardial infarction (MI), unstable angina, ischemic or hemorrhagic cerebrovascular accident, transient ischemic attack, and CV death.
The study was published in Annals of the Rheumatic Diseases.
The analysis included 12,233 patients with RA and had over 105,826 person-years of follow-up. The average follow-up time was 8.7 years. During the study period, 860 patients had their first MACE. After controlling for confounding factors, a daily prednisolone dose of 5 mg or higher doubled the risk for MACE, compared with GC nonusers. MACE risk increased by 7% per month.
Long-term glucocorticoid use discouraged
Daily doses of less than 5 mg were not associated with higher MACE risk, but more research is necessary to understand whether these low doses are clinically efficacious, Dr. Tam said. “The study results suggest that a very-low-dose GC (less than 5 mg prednisolone daily) may be cardiovascular risk–neutral. However, further evaluation is needed to determine whether this dose is therapeutic. Other potential side effects, such as bone loss, increased infection risk, dyslipidemia, and hyperglycemia, should also be considered.”
Both the American College of Rheumatology and the European Alliance of Associations for Rheumatology acknowledge that short-term GCs may be necessary for some RA patients, but they emphasize using the smallest necessary dose for the shortest period possible because of the known toxicity of GCs.
“We recommend stopping GCs as soon as it is clinically feasible, in line with previous recommendations, until these issues are investigated further,” Dr. Tam added.
Dr. Bartels agreed that long-term use of GCs should be avoided if possible, even at lower doses, because although CV risk may be less of an issue, studies have shown an increased risk for infection even at GC doses of less than 5 mg a day.
How might risk increase with dose?
While the study showed a distinct difference in risk with doses of prednisolone higher and lower than 5 mg, more information on how risk increases with dose could be useful, said Beth Wallace, MD, an assistant professor in internal medicine at the University of Michigan, Ann Arbor, and a staff rheumatologist at the VA Ann Arbor Healthcare Center. She was also unaffiliated with the research. “If someone is on 5-10 mg ... how much better is that than being on 10-20 mg or being on 20-30 mg?” she asked. While these study findings are “very important,” she said, it would be useful to know the risk associated with 7.5 mg vs. a higher dose.
But even in this relatively healthy population in Hong Kong, “taking more than 5 mg of prednisolone doubles the risk of cardiovascular disease,” Dr. Wallace added. This is important for clinicians to know, especially if they are more cautious about prescribing steroids to older or sicker patients but are “using [the drugs] a little more indiscriminately in younger people and healthier people.”
The study did not receive outside funding. Dr. Tam and Dr. Bartels report no relevant financial relationships. Dr. Wallace has received a grant from the Department of Veterans Affairs Administration to study steroid tapering in RA.
A version of this article first appeared on Medscape.com.
A daily prednisolone dose of 5 mg or higher is associated with increased risk for major adverse cardiovascular events (MACE) among patients with rheumatoid arthritis (RA), data suggest. Patients taking daily doses below this threshold did not appear to have an increased risk of MACE, compared with those not taking glucocorticoids (GCs).
Other studies of GCs and CV risk among RA patients have yielded conflicting results, especially for low-dose GCs. Findings from a 2020 study published in PLOS Medicine suggested that patients who had several immune-mediated inflammatory diseases – including RA – and who took less than a 5-mg prednisolone-equivalent dose daily had 74% higher risk for all-cause CVD, compared with nonusers. But results from a 2021 study published in Annals of the Rheumatic Diseases suggested that a daily prednisone dose of 4 mg or less did not increase cardiovascular events over a period of 6 months to 1 year.
These contradictory results were “primarily due to incomplete control of confounding variables, such as failure to adjust for C-reactive protein (CRP) levels,” Dr. Tam said. “Our study aimed to use a big data analytical approach to determine the effect of systemic GC dose and duration on the risk of major adverse cardiovascular events in patients with RA, while controlling for systemic inflammation, traditional CV risk factors, and other therapies.”
Is there a ‘safe’ dose for glucocorticoids?
To analyze this relationship, Dr. Lam and colleagues used the Hospital Authority Data Collaboration Laboratory, a citywide health care database. The investigators recruited patients with RA who had no history of MACE from 2006 to 2015 and followed them until the end of 2018. The primary outcome was the first occurrence of a MACE, defined as a composite of myocardial infarction (MI), unstable angina, ischemic or hemorrhagic cerebrovascular accident, transient ischemic attack, and CV death.
The study was published in Annals of the Rheumatic Diseases.
The analysis included 12,233 patients with RA and had over 105,826 person-years of follow-up. The average follow-up time was 8.7 years. During the study period, 860 patients had their first MACE. After controlling for confounding factors, a daily prednisolone dose of 5 mg or higher doubled the risk for MACE, compared with GC nonusers. MACE risk increased by 7% per month.
Long-term glucocorticoid use discouraged
Daily doses of less than 5 mg were not associated with higher MACE risk, but more research is necessary to understand whether these low doses are clinically efficacious, Dr. Tam said. “The study results suggest that a very-low-dose GC (less than 5 mg prednisolone daily) may be cardiovascular risk–neutral. However, further evaluation is needed to determine whether this dose is therapeutic. Other potential side effects, such as bone loss, increased infection risk, dyslipidemia, and hyperglycemia, should also be considered.”
Both the American College of Rheumatology and the European Alliance of Associations for Rheumatology acknowledge that short-term GCs may be necessary for some RA patients, but they emphasize using the smallest necessary dose for the shortest period possible because of the known toxicity of GCs.
“We recommend stopping GCs as soon as it is clinically feasible, in line with previous recommendations, until these issues are investigated further,” Dr. Tam added.
Dr. Bartels agreed that long-term use of GCs should be avoided if possible, even at lower doses, because although CV risk may be less of an issue, studies have shown an increased risk for infection even at GC doses of less than 5 mg a day.
How might risk increase with dose?
While the study showed a distinct difference in risk with doses of prednisolone higher and lower than 5 mg, more information on how risk increases with dose could be useful, said Beth Wallace, MD, an assistant professor in internal medicine at the University of Michigan, Ann Arbor, and a staff rheumatologist at the VA Ann Arbor Healthcare Center. She was also unaffiliated with the research. “If someone is on 5-10 mg ... how much better is that than being on 10-20 mg or being on 20-30 mg?” she asked. While these study findings are “very important,” she said, it would be useful to know the risk associated with 7.5 mg vs. a higher dose.
But even in this relatively healthy population in Hong Kong, “taking more than 5 mg of prednisolone doubles the risk of cardiovascular disease,” Dr. Wallace added. This is important for clinicians to know, especially if they are more cautious about prescribing steroids to older or sicker patients but are “using [the drugs] a little more indiscriminately in younger people and healthier people.”
The study did not receive outside funding. Dr. Tam and Dr. Bartels report no relevant financial relationships. Dr. Wallace has received a grant from the Department of Veterans Affairs Administration to study steroid tapering in RA.
A version of this article first appeared on Medscape.com.
FROM ANNALS OF THE RHEUMATIC DISEASES
Cigna accused of using AI, not doctors, to deny claims: Lawsuit
and forcing providers to bill patients in full.
In a complaint filed recently in California’s eastern district court, plaintiffs and Cigna health plan members Suzanne Kisting-Leung and Ayesha Smiley and their attorneys say that Cigna violates state insurance regulations by failing to conduct a “thorough, fair, and objective” review of their and other members’ claims.
The lawsuit says that, instead, Cigna relies on an algorithm, PxDx, to review and frequently deny medically necessary claims. According to court records, the system allows Cigna’s doctors to “instantly reject claims on medical grounds without ever opening patient files.” With use of the system, the average claims processing time is 1.2 seconds.
Cigna says it uses technology to verify coding on standard, low-cost procedures and to expedite physician reimbursement. In a statement to CBS News, the company called the lawsuit “highly questionable.”
The case highlights growing concerns about AI and its ability to replace humans for tasks and interactions in health care, business, and beyond. Public advocacy law firm Clarkson, which is representing the plaintiffs, has previously sued tech giants Google and ChatGPT creator OpenAI for harvesting Internet users’ personal and professional data to train their AI systems.
According to the complaint, Cigna denied the plaintiffs medically necessary tests, including blood work to screen for vitamin D deficiency and ultrasounds for patients suspected of having ovarian cancer. The plaintiffs’ attempts to appeal were unfruitful, and they were forced to pay out of pocket.
The plaintiff’s attorneys argue that the claims do not undergo more detailed reviews by physicians and employees, as mandated by California insurance laws, and that Cigna benefits by saving on labor costs.
Clarkson is demanding a jury trial and has asked the court to certify the Cigna case as a federal class action, potentially allowing the insurer’s other 2 million health plan members in California to join the lawsuit.
I. Glenn Cohen, JD, deputy dean and professor at Harvard Law School, Cambridge, Mass., said in an interview that this is the first lawsuit he’s aware of in which AI was involved in denying health insurance claims and that it is probably an uphill battle for the plaintiffs.
“In the last 25 years, the U.S. Supreme Court’s decisions have made getting a class action approved more difficult. If allowed to go forward as a class action, which Cigna is likely to vigorously oppose, then the pressure on Cigna to settle the case becomes enormous,” he said.
The allegations come after a recent deep dive by the nonprofit ProPublica uncovered similar claim denial issues. One physician who worked for Cigna told the nonprofit that he and other company doctors essentially rubber-stamped the denials in batches, which took “all of 10 seconds to do 50 at a time.”
In 2022, the American Medical Association and two state physician groups joined another class action against Cigna stemming from allegations that the insurer’s intermediary, Multiplan, intentionally underpaid medical claims. And in March, Cigna’s pharmacy benefit manager, Express Scripts, was accused of conspiring with other PBMs to drive up prescription drug prices for Ohio consumers, violating state antitrust laws.
Mr. Cohen said he expects Cigna to push back in court about the California class size, which the plaintiff’s attorneys hope will encompass all Cigna health plan members in the state.
“The injury is primarily to those whose claims were denied by AI, presumably a much smaller set of individuals and harder to identify,” said Mr. Cohen.
A version of this article first appeared on Medscape.com.
and forcing providers to bill patients in full.
In a complaint filed recently in California’s eastern district court, plaintiffs and Cigna health plan members Suzanne Kisting-Leung and Ayesha Smiley and their attorneys say that Cigna violates state insurance regulations by failing to conduct a “thorough, fair, and objective” review of their and other members’ claims.
The lawsuit says that, instead, Cigna relies on an algorithm, PxDx, to review and frequently deny medically necessary claims. According to court records, the system allows Cigna’s doctors to “instantly reject claims on medical grounds without ever opening patient files.” With use of the system, the average claims processing time is 1.2 seconds.
Cigna says it uses technology to verify coding on standard, low-cost procedures and to expedite physician reimbursement. In a statement to CBS News, the company called the lawsuit “highly questionable.”
The case highlights growing concerns about AI and its ability to replace humans for tasks and interactions in health care, business, and beyond. Public advocacy law firm Clarkson, which is representing the plaintiffs, has previously sued tech giants Google and ChatGPT creator OpenAI for harvesting Internet users’ personal and professional data to train their AI systems.
According to the complaint, Cigna denied the plaintiffs medically necessary tests, including blood work to screen for vitamin D deficiency and ultrasounds for patients suspected of having ovarian cancer. The plaintiffs’ attempts to appeal were unfruitful, and they were forced to pay out of pocket.
The plaintiff’s attorneys argue that the claims do not undergo more detailed reviews by physicians and employees, as mandated by California insurance laws, and that Cigna benefits by saving on labor costs.
Clarkson is demanding a jury trial and has asked the court to certify the Cigna case as a federal class action, potentially allowing the insurer’s other 2 million health plan members in California to join the lawsuit.
I. Glenn Cohen, JD, deputy dean and professor at Harvard Law School, Cambridge, Mass., said in an interview that this is the first lawsuit he’s aware of in which AI was involved in denying health insurance claims and that it is probably an uphill battle for the plaintiffs.
“In the last 25 years, the U.S. Supreme Court’s decisions have made getting a class action approved more difficult. If allowed to go forward as a class action, which Cigna is likely to vigorously oppose, then the pressure on Cigna to settle the case becomes enormous,” he said.
The allegations come after a recent deep dive by the nonprofit ProPublica uncovered similar claim denial issues. One physician who worked for Cigna told the nonprofit that he and other company doctors essentially rubber-stamped the denials in batches, which took “all of 10 seconds to do 50 at a time.”
In 2022, the American Medical Association and two state physician groups joined another class action against Cigna stemming from allegations that the insurer’s intermediary, Multiplan, intentionally underpaid medical claims. And in March, Cigna’s pharmacy benefit manager, Express Scripts, was accused of conspiring with other PBMs to drive up prescription drug prices for Ohio consumers, violating state antitrust laws.
Mr. Cohen said he expects Cigna to push back in court about the California class size, which the plaintiff’s attorneys hope will encompass all Cigna health plan members in the state.
“The injury is primarily to those whose claims were denied by AI, presumably a much smaller set of individuals and harder to identify,” said Mr. Cohen.
A version of this article first appeared on Medscape.com.
and forcing providers to bill patients in full.
In a complaint filed recently in California’s eastern district court, plaintiffs and Cigna health plan members Suzanne Kisting-Leung and Ayesha Smiley and their attorneys say that Cigna violates state insurance regulations by failing to conduct a “thorough, fair, and objective” review of their and other members’ claims.
The lawsuit says that, instead, Cigna relies on an algorithm, PxDx, to review and frequently deny medically necessary claims. According to court records, the system allows Cigna’s doctors to “instantly reject claims on medical grounds without ever opening patient files.” With use of the system, the average claims processing time is 1.2 seconds.
Cigna says it uses technology to verify coding on standard, low-cost procedures and to expedite physician reimbursement. In a statement to CBS News, the company called the lawsuit “highly questionable.”
The case highlights growing concerns about AI and its ability to replace humans for tasks and interactions in health care, business, and beyond. Public advocacy law firm Clarkson, which is representing the plaintiffs, has previously sued tech giants Google and ChatGPT creator OpenAI for harvesting Internet users’ personal and professional data to train their AI systems.
According to the complaint, Cigna denied the plaintiffs medically necessary tests, including blood work to screen for vitamin D deficiency and ultrasounds for patients suspected of having ovarian cancer. The plaintiffs’ attempts to appeal were unfruitful, and they were forced to pay out of pocket.
The plaintiff’s attorneys argue that the claims do not undergo more detailed reviews by physicians and employees, as mandated by California insurance laws, and that Cigna benefits by saving on labor costs.
Clarkson is demanding a jury trial and has asked the court to certify the Cigna case as a federal class action, potentially allowing the insurer’s other 2 million health plan members in California to join the lawsuit.
I. Glenn Cohen, JD, deputy dean and professor at Harvard Law School, Cambridge, Mass., said in an interview that this is the first lawsuit he’s aware of in which AI was involved in denying health insurance claims and that it is probably an uphill battle for the plaintiffs.
“In the last 25 years, the U.S. Supreme Court’s decisions have made getting a class action approved more difficult. If allowed to go forward as a class action, which Cigna is likely to vigorously oppose, then the pressure on Cigna to settle the case becomes enormous,” he said.
The allegations come after a recent deep dive by the nonprofit ProPublica uncovered similar claim denial issues. One physician who worked for Cigna told the nonprofit that he and other company doctors essentially rubber-stamped the denials in batches, which took “all of 10 seconds to do 50 at a time.”
In 2022, the American Medical Association and two state physician groups joined another class action against Cigna stemming from allegations that the insurer’s intermediary, Multiplan, intentionally underpaid medical claims. And in March, Cigna’s pharmacy benefit manager, Express Scripts, was accused of conspiring with other PBMs to drive up prescription drug prices for Ohio consumers, violating state antitrust laws.
Mr. Cohen said he expects Cigna to push back in court about the California class size, which the plaintiff’s attorneys hope will encompass all Cigna health plan members in the state.
“The injury is primarily to those whose claims were denied by AI, presumably a much smaller set of individuals and harder to identify,” said Mr. Cohen.
A version of this article first appeared on Medscape.com.
Disclosure inaccuracies common in top rheumatology journals
TOPLINE:
Conflicts of interests in rheumatology research – particularly for clinical trials – are often incorrectly reported, according to a new analysis.
METHODOLOGY:
- Researchers reviewed the first 50 clinical research reports, reviews, and editorials published in 2019 by Arthritis & Rheumatology, Arthritis Care & Research, and Seminars in Arthritis & Rheumatism.
- They cross-checked disclosures from the first, second, and last authors of each paper (150 total) with payment reports from the Open Payments Database (OPD).
- Payment reports captured consulting fees, honoraria, and speaker/faculty compensation in the 36 months prior to an article’s publication.
TAKEAWAY:
- A total of 87% of the 135 authors with potential conflicts of interest (PCOI) inaccurately reported their disclosures.
- All authors of the included 14 clinical trial publications either did not report or underreported PCOI.
- The total nondisclosed dollar amount was $5,190,901, and the total underdisclosed amount was $4,135,126.
IN PRACTICE:
“Improved community education and firmer expectations would permit readers to better assess any possible impact of PCOI on publications,” the authors wrote.
STUDY DETAILS:
Mary Guan, MD, of the NYU Grossman School of Medicine, New York, led the research. The study was published online in Arthritis Care & Research on July 31, 2023.
LIMITATIONS:
The OPD does not include non–U.S.-based authors and authors without a medical degree, and there are no data on the accuracy of the database. The analysis does not provide insight into why these discrepancies occurred and if they were unintentional errors.
DISCLOSURES:
One coauthor reported consulting fees from Federation Bio, Fortress Biotech, Horizon, and Sobi and receiving grants or contracts paid to his institution from Horizon and Hikma. Dr. Guan and senior author Aryeh Abeles, MD, reported no relevant disclosures.
A version of this article appeared on Medscape.com.
TOPLINE:
Conflicts of interests in rheumatology research – particularly for clinical trials – are often incorrectly reported, according to a new analysis.
METHODOLOGY:
- Researchers reviewed the first 50 clinical research reports, reviews, and editorials published in 2019 by Arthritis & Rheumatology, Arthritis Care & Research, and Seminars in Arthritis & Rheumatism.
- They cross-checked disclosures from the first, second, and last authors of each paper (150 total) with payment reports from the Open Payments Database (OPD).
- Payment reports captured consulting fees, honoraria, and speaker/faculty compensation in the 36 months prior to an article’s publication.
TAKEAWAY:
- A total of 87% of the 135 authors with potential conflicts of interest (PCOI) inaccurately reported their disclosures.
- All authors of the included 14 clinical trial publications either did not report or underreported PCOI.
- The total nondisclosed dollar amount was $5,190,901, and the total underdisclosed amount was $4,135,126.
IN PRACTICE:
“Improved community education and firmer expectations would permit readers to better assess any possible impact of PCOI on publications,” the authors wrote.
STUDY DETAILS:
Mary Guan, MD, of the NYU Grossman School of Medicine, New York, led the research. The study was published online in Arthritis Care & Research on July 31, 2023.
LIMITATIONS:
The OPD does not include non–U.S.-based authors and authors without a medical degree, and there are no data on the accuracy of the database. The analysis does not provide insight into why these discrepancies occurred and if they were unintentional errors.
DISCLOSURES:
One coauthor reported consulting fees from Federation Bio, Fortress Biotech, Horizon, and Sobi and receiving grants or contracts paid to his institution from Horizon and Hikma. Dr. Guan and senior author Aryeh Abeles, MD, reported no relevant disclosures.
A version of this article appeared on Medscape.com.
TOPLINE:
Conflicts of interests in rheumatology research – particularly for clinical trials – are often incorrectly reported, according to a new analysis.
METHODOLOGY:
- Researchers reviewed the first 50 clinical research reports, reviews, and editorials published in 2019 by Arthritis & Rheumatology, Arthritis Care & Research, and Seminars in Arthritis & Rheumatism.
- They cross-checked disclosures from the first, second, and last authors of each paper (150 total) with payment reports from the Open Payments Database (OPD).
- Payment reports captured consulting fees, honoraria, and speaker/faculty compensation in the 36 months prior to an article’s publication.
TAKEAWAY:
- A total of 87% of the 135 authors with potential conflicts of interest (PCOI) inaccurately reported their disclosures.
- All authors of the included 14 clinical trial publications either did not report or underreported PCOI.
- The total nondisclosed dollar amount was $5,190,901, and the total underdisclosed amount was $4,135,126.
IN PRACTICE:
“Improved community education and firmer expectations would permit readers to better assess any possible impact of PCOI on publications,” the authors wrote.
STUDY DETAILS:
Mary Guan, MD, of the NYU Grossman School of Medicine, New York, led the research. The study was published online in Arthritis Care & Research on July 31, 2023.
LIMITATIONS:
The OPD does not include non–U.S.-based authors and authors without a medical degree, and there are no data on the accuracy of the database. The analysis does not provide insight into why these discrepancies occurred and if they were unintentional errors.
DISCLOSURES:
One coauthor reported consulting fees from Federation Bio, Fortress Biotech, Horizon, and Sobi and receiving grants or contracts paid to his institution from Horizon and Hikma. Dr. Guan and senior author Aryeh Abeles, MD, reported no relevant disclosures.
A version of this article appeared on Medscape.com.
FROM ARTHRITIS CARE & RESEARCH
Autoantibodies could help predict cancer risk in scleroderma
TOPLINE:
METHODOLOGY:
- Included patients from the Johns Hopkins Scleroderma Center Research Registry and the University of Pittsburgh Scleroderma Center, Pittsburgh.
- A total of 676 patients with scleroderma and a history of cancer were compared with 687 control patients with scleroderma but without a history of cancer.
- Serum tested via line blot and enzyme-linked immunosorbent assay for an array of scleroderma autoantibodies.
- Examined association between autoantibodies and overall cancer risk.
TAKEAWAYS:
- Anti-POLR3 and monospecific anti-Ro52 were associated with significantly increased overall cancer risk.
- Anti-centromere and anti-U1RNP were associated with a decreased cancer risk.
- These associations remained when looking specifically at cancer-associated scleroderma.
- Patients positive for anti-Ro52 in combination with either anti-U1RNP or anti-Th/To had a decreased risk of cancer, compared with those who had anti-Ro52 alone.
IN PRACTICE:
This study is too preliminary to have practice application.
SOURCE:
Ji Soo Kim, PhD, of John Hopkins University, Baltimore, was the first author of the study, published in Arthritis & Rheumatology on July 24, 2023. Fellow Johns Hopkins researchers Livia Casciola-Rosen, PhD, and Ami A. Shah, MD, were joint senior authors.
DISCLOSURES:
The study was supported by the National Institute of Arthritis and Musculoskeletal and Skin Diseases, the Donald B. and Dorothy L. Stabler Foundation, the Jerome L. Greene Foundation, the Chresanthe Staurulakis Memorial Discovery Fund, the Martha McCrory Professorship, and the Johns Hopkins inHealth initiative. The authors disclosed the following patents or patent applications: Autoimmune Antigens and Cancer, Materials and Methods for Assessing Cancer Risk and Treating Cancer.
A version of this article appeared on Medscape.com.
TOPLINE:
METHODOLOGY:
- Included patients from the Johns Hopkins Scleroderma Center Research Registry and the University of Pittsburgh Scleroderma Center, Pittsburgh.
- A total of 676 patients with scleroderma and a history of cancer were compared with 687 control patients with scleroderma but without a history of cancer.
- Serum tested via line blot and enzyme-linked immunosorbent assay for an array of scleroderma autoantibodies.
- Examined association between autoantibodies and overall cancer risk.
TAKEAWAYS:
- Anti-POLR3 and monospecific anti-Ro52 were associated with significantly increased overall cancer risk.
- Anti-centromere and anti-U1RNP were associated with a decreased cancer risk.
- These associations remained when looking specifically at cancer-associated scleroderma.
- Patients positive for anti-Ro52 in combination with either anti-U1RNP or anti-Th/To had a decreased risk of cancer, compared with those who had anti-Ro52 alone.
IN PRACTICE:
This study is too preliminary to have practice application.
SOURCE:
Ji Soo Kim, PhD, of John Hopkins University, Baltimore, was the first author of the study, published in Arthritis & Rheumatology on July 24, 2023. Fellow Johns Hopkins researchers Livia Casciola-Rosen, PhD, and Ami A. Shah, MD, were joint senior authors.
DISCLOSURES:
The study was supported by the National Institute of Arthritis and Musculoskeletal and Skin Diseases, the Donald B. and Dorothy L. Stabler Foundation, the Jerome L. Greene Foundation, the Chresanthe Staurulakis Memorial Discovery Fund, the Martha McCrory Professorship, and the Johns Hopkins inHealth initiative. The authors disclosed the following patents or patent applications: Autoimmune Antigens and Cancer, Materials and Methods for Assessing Cancer Risk and Treating Cancer.
A version of this article appeared on Medscape.com.
TOPLINE:
METHODOLOGY:
- Included patients from the Johns Hopkins Scleroderma Center Research Registry and the University of Pittsburgh Scleroderma Center, Pittsburgh.
- A total of 676 patients with scleroderma and a history of cancer were compared with 687 control patients with scleroderma but without a history of cancer.
- Serum tested via line blot and enzyme-linked immunosorbent assay for an array of scleroderma autoantibodies.
- Examined association between autoantibodies and overall cancer risk.
TAKEAWAYS:
- Anti-POLR3 and monospecific anti-Ro52 were associated with significantly increased overall cancer risk.
- Anti-centromere and anti-U1RNP were associated with a decreased cancer risk.
- These associations remained when looking specifically at cancer-associated scleroderma.
- Patients positive for anti-Ro52 in combination with either anti-U1RNP or anti-Th/To had a decreased risk of cancer, compared with those who had anti-Ro52 alone.
IN PRACTICE:
This study is too preliminary to have practice application.
SOURCE:
Ji Soo Kim, PhD, of John Hopkins University, Baltimore, was the first author of the study, published in Arthritis & Rheumatology on July 24, 2023. Fellow Johns Hopkins researchers Livia Casciola-Rosen, PhD, and Ami A. Shah, MD, were joint senior authors.
DISCLOSURES:
The study was supported by the National Institute of Arthritis and Musculoskeletal and Skin Diseases, the Donald B. and Dorothy L. Stabler Foundation, the Jerome L. Greene Foundation, the Chresanthe Staurulakis Memorial Discovery Fund, the Martha McCrory Professorship, and the Johns Hopkins inHealth initiative. The authors disclosed the following patents or patent applications: Autoimmune Antigens and Cancer, Materials and Methods for Assessing Cancer Risk and Treating Cancer.
A version of this article appeared on Medscape.com.
FROM ARTHRITIS & RHEUMATOLOGY
Medical students are skipping class lectures: Does it matter?
New technologies, including online lectures and guided-lesson websites, along with alternative teaching methods, such as the flipped classroom model, in which med students complete before-class assignments and participate in group projects, are helping to train future physicians for their medical careers.
So though students may not be attending in-person lectures like they did in the past, proponents of online learning say the education students receive and the subsequent care they deliver remains the same.
The Association of American Medical Colleges’ most recent annual survey of 2nd-year medical students found that 25% “almost never” attended their in-person lectures in 2022. The figure has steadily improved since 2020 but mirrors what AAMC recorded in 2017.
“The pandemic may have exacerbated the trend, but it’s a long-standing issue,” said Katherine McOwen, senior director of educational and student affairs at AAMC. She said in an interview that she’s witnessed the pattern for 24 years in her work with medical schools.
“I know it sounds alarming that students aren’t attending lectures. But that doesn’t mean they’re not learning,” said Ahmed Ahmed, MD, MPP, MSc, a recent graduate of Harvard Medical School and now a resident at Brigham and Women’s Hospital, Boston.
Today’s generation of medical students grew up in the age of technology. They are comfortable in front of the screen, so it makes sense for them to learn certain aspects of medical sciences and public health in the same way, Dr. Ahmed told this news organization.
Dr. Ahmed said that at Harvard he participated in one or two case-based classes per week that followed a flipped classroom model, which allows students to study topics on their own before discussing in a lecture format as a group. “We had to come up with a diagnostic plan and walk through the case slide by slide,” he said. “It got us to think like a clinician.”
The flipped classroom allows students to study at their own pace using their preferred learning style, leading to more collaboration in the classroom and between students, according to a 2022 article on the “new standard in medical education” published in Trends in Anaesthesia & Critical Care.
Students use online education tools to complete pre-class assignments such as watching short videos, listening to podcasts, or reading journal articles. In-class time can then be used to cement and create connections through discussions, interactive exercises, group learning, and case studies, the article stated.
Benefits of the flipped classroom include student satisfaction, learner motivation, and faculty interest in learning new teaching methods, according to the article: “Students are performing at least as well as those who attended traditional lectures, while some studies in select health care settings show increased retention in flipped classroom settings.”
Another study on the flipped classroom, published in 2018 in BMC Medical Education found that the teaching method was superior to traditional classrooms for health professions education. Researchers focused specifically on flipped classrooms that provided prerecorded videos to students.
Molly Cooke, MD, director of education for global health sciences at the University of California, San Francisco, School of Medicine, said that the school no longer requires attendance at lectures. “Personally, my position is that medical students are very busy people and make, by and large, rational decisions about how to spend their time. As learning and retention from 50-minute lectures has been shown for decades to be poor, I think it’s perfectly reasonable to watch lectures on their own time.”
Dr. Ahmed agrees. “By our standards, the old model is archaic. It’s passive, and instead we should be encouraging lifelong, self-directed learning.”
To that end, Dr. Ahmed and his fellow students also relied heavily during medical school on secondary educational sources such as Boards and Beyond and Sketchy. “There’s an entire community of medical school students across the country using them,” Dr. Ahmed explained. “You can learn what you need in a tenth of the time of lectures.”
Today lectures only provide a portion of the information delivered to students, Dr. McGowen said. “They also learn in small groups, in problem-solving sessions, and in clinical experiences, all of which make up the meat of their education.”
The purpose of medical school is to prepare students for residency, she added. “Medical school education is very different from other types of education. Students are examined in a variety of ways before they move on to residency and ultimately, practice.”
For example, every student must pass the three-part United States Medical Licensing Examination. Students complete the first two parts in medical school and the third part during residency. “The tests represent a combination of everything students have learned, from lectures, clinical time, and in self-directed learning,” Dr. McGowen said.
Post pandemic, the tools and styles of learning in medical education have changed, and they are likely to continue to evolve along with students and technology, according to the 2022 article on the flipped classroom. “The future of medical education will continue to move in ways that embrace digital technology, as this is what digital native learners are increasingly expecting for their health care education,” states the article.
A version of this article first appeared on Medscape.com.
New technologies, including online lectures and guided-lesson websites, along with alternative teaching methods, such as the flipped classroom model, in which med students complete before-class assignments and participate in group projects, are helping to train future physicians for their medical careers.
So though students may not be attending in-person lectures like they did in the past, proponents of online learning say the education students receive and the subsequent care they deliver remains the same.
The Association of American Medical Colleges’ most recent annual survey of 2nd-year medical students found that 25% “almost never” attended their in-person lectures in 2022. The figure has steadily improved since 2020 but mirrors what AAMC recorded in 2017.
“The pandemic may have exacerbated the trend, but it’s a long-standing issue,” said Katherine McOwen, senior director of educational and student affairs at AAMC. She said in an interview that she’s witnessed the pattern for 24 years in her work with medical schools.
“I know it sounds alarming that students aren’t attending lectures. But that doesn’t mean they’re not learning,” said Ahmed Ahmed, MD, MPP, MSc, a recent graduate of Harvard Medical School and now a resident at Brigham and Women’s Hospital, Boston.
Today’s generation of medical students grew up in the age of technology. They are comfortable in front of the screen, so it makes sense for them to learn certain aspects of medical sciences and public health in the same way, Dr. Ahmed told this news organization.
Dr. Ahmed said that at Harvard he participated in one or two case-based classes per week that followed a flipped classroom model, which allows students to study topics on their own before discussing in a lecture format as a group. “We had to come up with a diagnostic plan and walk through the case slide by slide,” he said. “It got us to think like a clinician.”
The flipped classroom allows students to study at their own pace using their preferred learning style, leading to more collaboration in the classroom and between students, according to a 2022 article on the “new standard in medical education” published in Trends in Anaesthesia & Critical Care.
Students use online education tools to complete pre-class assignments such as watching short videos, listening to podcasts, or reading journal articles. In-class time can then be used to cement and create connections through discussions, interactive exercises, group learning, and case studies, the article stated.
Benefits of the flipped classroom include student satisfaction, learner motivation, and faculty interest in learning new teaching methods, according to the article: “Students are performing at least as well as those who attended traditional lectures, while some studies in select health care settings show increased retention in flipped classroom settings.”
Another study on the flipped classroom, published in 2018 in BMC Medical Education found that the teaching method was superior to traditional classrooms for health professions education. Researchers focused specifically on flipped classrooms that provided prerecorded videos to students.
Molly Cooke, MD, director of education for global health sciences at the University of California, San Francisco, School of Medicine, said that the school no longer requires attendance at lectures. “Personally, my position is that medical students are very busy people and make, by and large, rational decisions about how to spend their time. As learning and retention from 50-minute lectures has been shown for decades to be poor, I think it’s perfectly reasonable to watch lectures on their own time.”
Dr. Ahmed agrees. “By our standards, the old model is archaic. It’s passive, and instead we should be encouraging lifelong, self-directed learning.”
To that end, Dr. Ahmed and his fellow students also relied heavily during medical school on secondary educational sources such as Boards and Beyond and Sketchy. “There’s an entire community of medical school students across the country using them,” Dr. Ahmed explained. “You can learn what you need in a tenth of the time of lectures.”
Today lectures only provide a portion of the information delivered to students, Dr. McGowen said. “They also learn in small groups, in problem-solving sessions, and in clinical experiences, all of which make up the meat of their education.”
The purpose of medical school is to prepare students for residency, she added. “Medical school education is very different from other types of education. Students are examined in a variety of ways before they move on to residency and ultimately, practice.”
For example, every student must pass the three-part United States Medical Licensing Examination. Students complete the first two parts in medical school and the third part during residency. “The tests represent a combination of everything students have learned, from lectures, clinical time, and in self-directed learning,” Dr. McGowen said.
Post pandemic, the tools and styles of learning in medical education have changed, and they are likely to continue to evolve along with students and technology, according to the 2022 article on the flipped classroom. “The future of medical education will continue to move in ways that embrace digital technology, as this is what digital native learners are increasingly expecting for their health care education,” states the article.
A version of this article first appeared on Medscape.com.
New technologies, including online lectures and guided-lesson websites, along with alternative teaching methods, such as the flipped classroom model, in which med students complete before-class assignments and participate in group projects, are helping to train future physicians for their medical careers.
So though students may not be attending in-person lectures like they did in the past, proponents of online learning say the education students receive and the subsequent care they deliver remains the same.
The Association of American Medical Colleges’ most recent annual survey of 2nd-year medical students found that 25% “almost never” attended their in-person lectures in 2022. The figure has steadily improved since 2020 but mirrors what AAMC recorded in 2017.
“The pandemic may have exacerbated the trend, but it’s a long-standing issue,” said Katherine McOwen, senior director of educational and student affairs at AAMC. She said in an interview that she’s witnessed the pattern for 24 years in her work with medical schools.
“I know it sounds alarming that students aren’t attending lectures. But that doesn’t mean they’re not learning,” said Ahmed Ahmed, MD, MPP, MSc, a recent graduate of Harvard Medical School and now a resident at Brigham and Women’s Hospital, Boston.
Today’s generation of medical students grew up in the age of technology. They are comfortable in front of the screen, so it makes sense for them to learn certain aspects of medical sciences and public health in the same way, Dr. Ahmed told this news organization.
Dr. Ahmed said that at Harvard he participated in one or two case-based classes per week that followed a flipped classroom model, which allows students to study topics on their own before discussing in a lecture format as a group. “We had to come up with a diagnostic plan and walk through the case slide by slide,” he said. “It got us to think like a clinician.”
The flipped classroom allows students to study at their own pace using their preferred learning style, leading to more collaboration in the classroom and between students, according to a 2022 article on the “new standard in medical education” published in Trends in Anaesthesia & Critical Care.
Students use online education tools to complete pre-class assignments such as watching short videos, listening to podcasts, or reading journal articles. In-class time can then be used to cement and create connections through discussions, interactive exercises, group learning, and case studies, the article stated.
Benefits of the flipped classroom include student satisfaction, learner motivation, and faculty interest in learning new teaching methods, according to the article: “Students are performing at least as well as those who attended traditional lectures, while some studies in select health care settings show increased retention in flipped classroom settings.”
Another study on the flipped classroom, published in 2018 in BMC Medical Education found that the teaching method was superior to traditional classrooms for health professions education. Researchers focused specifically on flipped classrooms that provided prerecorded videos to students.
Molly Cooke, MD, director of education for global health sciences at the University of California, San Francisco, School of Medicine, said that the school no longer requires attendance at lectures. “Personally, my position is that medical students are very busy people and make, by and large, rational decisions about how to spend their time. As learning and retention from 50-minute lectures has been shown for decades to be poor, I think it’s perfectly reasonable to watch lectures on their own time.”
Dr. Ahmed agrees. “By our standards, the old model is archaic. It’s passive, and instead we should be encouraging lifelong, self-directed learning.”
To that end, Dr. Ahmed and his fellow students also relied heavily during medical school on secondary educational sources such as Boards and Beyond and Sketchy. “There’s an entire community of medical school students across the country using them,” Dr. Ahmed explained. “You can learn what you need in a tenth of the time of lectures.”
Today lectures only provide a portion of the information delivered to students, Dr. McGowen said. “They also learn in small groups, in problem-solving sessions, and in clinical experiences, all of which make up the meat of their education.”
The purpose of medical school is to prepare students for residency, she added. “Medical school education is very different from other types of education. Students are examined in a variety of ways before they move on to residency and ultimately, practice.”
For example, every student must pass the three-part United States Medical Licensing Examination. Students complete the first two parts in medical school and the third part during residency. “The tests represent a combination of everything students have learned, from lectures, clinical time, and in self-directed learning,” Dr. McGowen said.
Post pandemic, the tools and styles of learning in medical education have changed, and they are likely to continue to evolve along with students and technology, according to the 2022 article on the flipped classroom. “The future of medical education will continue to move in ways that embrace digital technology, as this is what digital native learners are increasingly expecting for their health care education,” states the article.
A version of this article first appeared on Medscape.com.
A new and completely different pain medicine
This transcript has been edited for clarity.
When you stub your toe or get a paper cut on your finger, you feel the pain in that part of your body. It feels like the pain is coming from that place. But, of course, that’s not really what is happening. Pain doesn’t really happen in your toe or your finger. It happens in your brain.
It’s a game of telephone, really. The afferent nerve fiber detects the noxious stimulus, passing that signal to the second-order neuron in the dorsal root ganglia of the spinal cord, which runs it up to the thalamus to be passed to the third-order neuron which brings it to the cortex for localization and conscious perception. It’s not even a very good game of telephone. It takes about 100 ms for a pain signal to get from the hand to the brain – longer from the feet, given the greater distance. You see your foot hit the corner of the coffee table and have just enough time to think: “Oh no!” before the pain hits.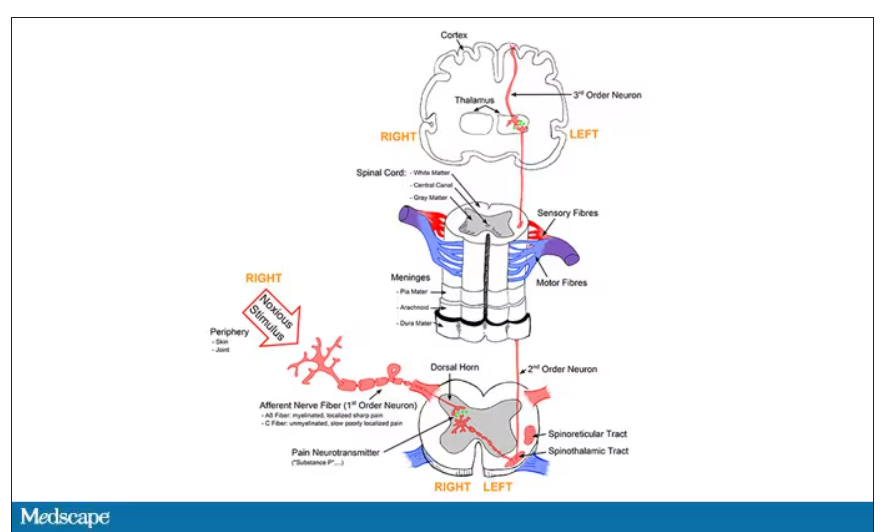
Given the Rube Goldberg nature of the process, it would seem like there are any number of places we could stop pain sensation. And sure, local anesthetics at the site of injury, or even spinal anesthetics, are powerful – if temporary and hard to administer – solutions to acute pain.
But in our everyday armamentarium, let’s be honest – we essentially have three options: opiates and opioids, which activate the mu-receptors in the brain to dull pain (and cause a host of other nasty side effects); NSAIDs, which block prostaglandin synthesis and thus limit the ability for pain-conducting neurons to get excited; and acetaminophen, which, despite being used for a century, is poorly understood.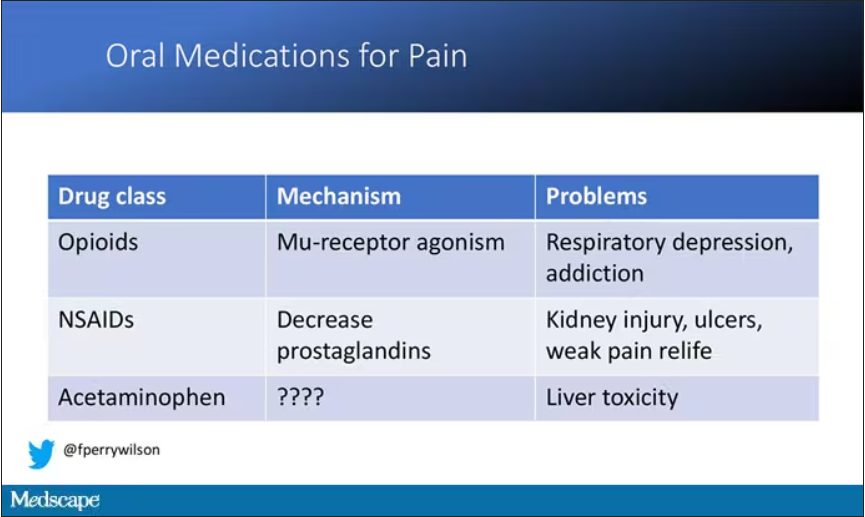
But
If you were to zoom in on the connection between that first afferent pain fiber and the secondary nerve in the spinal cord dorsal root ganglion, you would see a receptor called Nav1.8, a voltage-gated sodium channel.
This receptor is a key part of the apparatus that passes information from nerve 1 to nerve 2, but only for fibers that transmit pain signals. In fact, humans with mutations in this receptor that leave it always in the “open” state have a severe pain syndrome. Blocking the receptor, therefore, might reduce pain.
In preclinical work, researchers identified VX-548, which doesn’t have a brand name yet, as a potent blocker of that channel even in nanomolar concentrations. Importantly, the compound was highly selective for that particular channel – about 30,000 times more selective than it was for the other sodium channels in that family.
Of course, a highly selective and specific drug does not a blockbuster analgesic make. To determine how this drug would work on humans in pain, they turned to two populations: 303 individuals undergoing abdominoplasty and 274 undergoing bunionectomy, as reported in a new paper in the New England Journal of Medicine.
I know this seems a bit random, but abdominoplasty is quite painful and a good model for soft-tissue pain. Bunionectomy is also quite a painful procedure and a useful model of bone pain. After the surgeries, patients were randomized to several different doses of VX-548, hydrocodone plus acetaminophen, or placebo for 48 hours.
At 19 time points over that 48-hour period, participants were asked to rate their pain on a scale from 0 to 10. The primary outcome was the cumulative pain experienced over the 48 hours. So, higher pain would be worse here, but longer duration of pain would also be worse.
The story of the study is really told in this chart.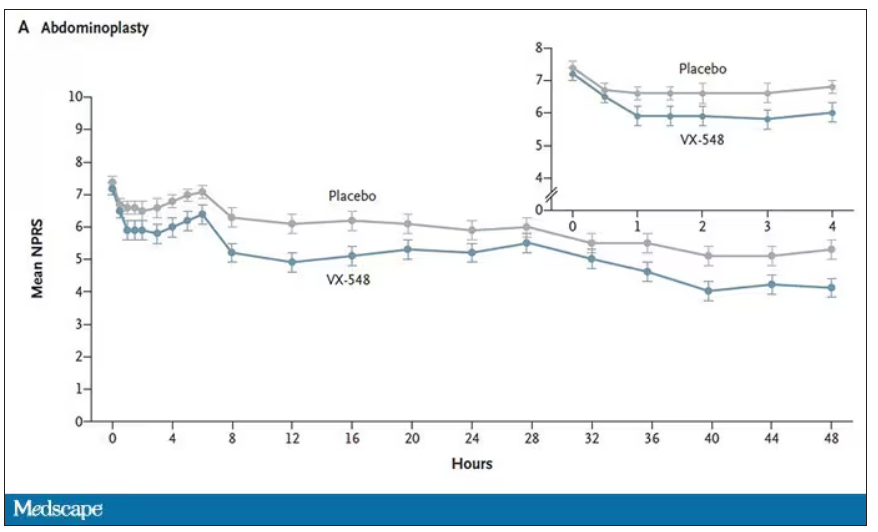
Yes, those assigned to the highest dose of VX-548 had a statistically significant lower cumulative amount of pain in the 48 hours after surgery. But the picture is really worth more than the stats here. You can see that the onset of pain relief was fairly quick, and that pain relief was sustained over time. You can also see that this is not a miracle drug. Pain scores were a bit better 48 hours out, but only by about a point and a half.
Placebo isn’t really the fair comparison here; few of us treat our postabdominoplasty patients with placebo, after all. The authors do not formally compare the effect of VX-548 with that of the opioid hydrocodone, for instance. But that doesn’t stop us.
This graph, which I put together from data in the paper, shows pain control across the four randomization categories, with higher numbers indicating more (cumulative) control. While all the active agents do a bit better than placebo, VX-548 at the higher dose appears to do the best. But I should note that 5 mg of hydrocodone may not be an adequate dose for most people.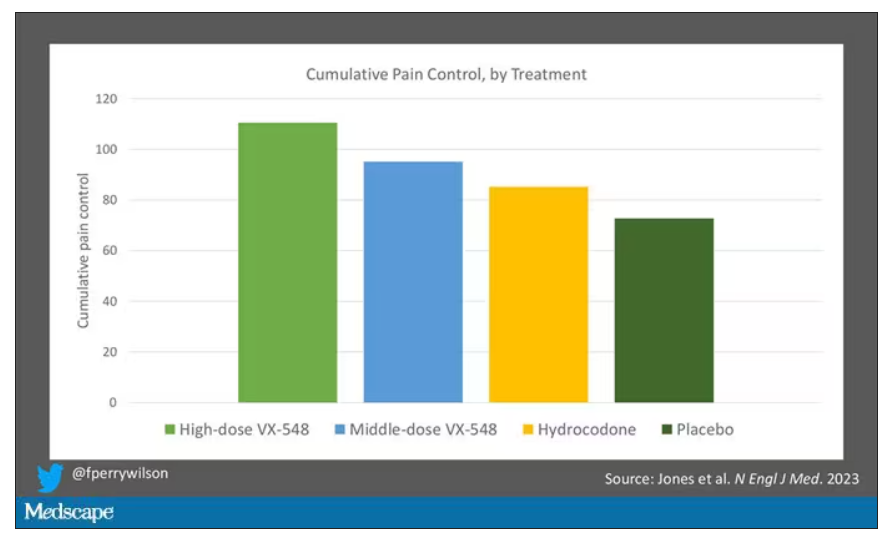
Yes, I would really have killed for an NSAID arm in this trial. Its absence, given that NSAIDs are a staple of postoperative care, is ... well, let’s just say, notable.
Although not a pain-destroying machine, VX-548 has some other things to recommend it. The receptor is really not found in the brain at all, which suggests that the drug should not carry much risk for dependency, though that has not been formally studied.
The side effects were generally mild – headache was the most common – and less prevalent than what you see even in the placebo arm.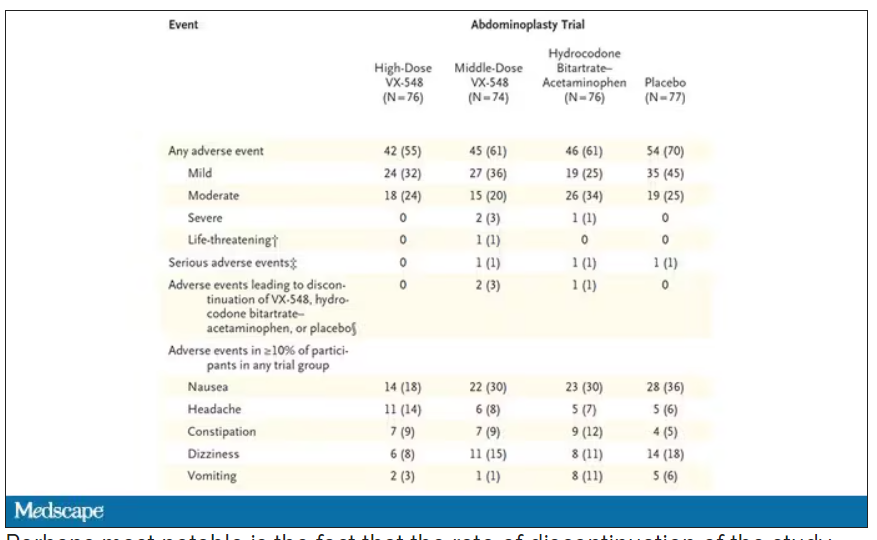
Perhaps most notable is the fact that the rate of discontinuation of the study drug was lowest in the VX-548 arm. Patients could stop taking the pill they were assigned for any reason, ranging from perceived lack of efficacy to side effects. A low discontinuation rate indicates to me a sort of “voting with your feet” that suggests this might be a well-tolerated and reasonably effective drug.
VX-548 isn’t on the market yet; phase 3 trials are ongoing. But whether it is this particular drug or another in this class, I’m happy to see researchers trying to find new ways to target that most primeval form of suffering: pain.
Dr. Wilson is an associate professor of medicine and public health and director of Yale’s Clinical and Translational Research Accelerator, New Haven, Conn. He disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
When you stub your toe or get a paper cut on your finger, you feel the pain in that part of your body. It feels like the pain is coming from that place. But, of course, that’s not really what is happening. Pain doesn’t really happen in your toe or your finger. It happens in your brain.
It’s a game of telephone, really. The afferent nerve fiber detects the noxious stimulus, passing that signal to the second-order neuron in the dorsal root ganglia of the spinal cord, which runs it up to the thalamus to be passed to the third-order neuron which brings it to the cortex for localization and conscious perception. It’s not even a very good game of telephone. It takes about 100 ms for a pain signal to get from the hand to the brain – longer from the feet, given the greater distance. You see your foot hit the corner of the coffee table and have just enough time to think: “Oh no!” before the pain hits.
Given the Rube Goldberg nature of the process, it would seem like there are any number of places we could stop pain sensation. And sure, local anesthetics at the site of injury, or even spinal anesthetics, are powerful – if temporary and hard to administer – solutions to acute pain.
But in our everyday armamentarium, let’s be honest – we essentially have three options: opiates and opioids, which activate the mu-receptors in the brain to dull pain (and cause a host of other nasty side effects); NSAIDs, which block prostaglandin synthesis and thus limit the ability for pain-conducting neurons to get excited; and acetaminophen, which, despite being used for a century, is poorly understood.
But
If you were to zoom in on the connection between that first afferent pain fiber and the secondary nerve in the spinal cord dorsal root ganglion, you would see a receptor called Nav1.8, a voltage-gated sodium channel.
This receptor is a key part of the apparatus that passes information from nerve 1 to nerve 2, but only for fibers that transmit pain signals. In fact, humans with mutations in this receptor that leave it always in the “open” state have a severe pain syndrome. Blocking the receptor, therefore, might reduce pain.
In preclinical work, researchers identified VX-548, which doesn’t have a brand name yet, as a potent blocker of that channel even in nanomolar concentrations. Importantly, the compound was highly selective for that particular channel – about 30,000 times more selective than it was for the other sodium channels in that family.
Of course, a highly selective and specific drug does not a blockbuster analgesic make. To determine how this drug would work on humans in pain, they turned to two populations: 303 individuals undergoing abdominoplasty and 274 undergoing bunionectomy, as reported in a new paper in the New England Journal of Medicine.
I know this seems a bit random, but abdominoplasty is quite painful and a good model for soft-tissue pain. Bunionectomy is also quite a painful procedure and a useful model of bone pain. After the surgeries, patients were randomized to several different doses of VX-548, hydrocodone plus acetaminophen, or placebo for 48 hours.
At 19 time points over that 48-hour period, participants were asked to rate their pain on a scale from 0 to 10. The primary outcome was the cumulative pain experienced over the 48 hours. So, higher pain would be worse here, but longer duration of pain would also be worse.
The story of the study is really told in this chart.
Yes, those assigned to the highest dose of VX-548 had a statistically significant lower cumulative amount of pain in the 48 hours after surgery. But the picture is really worth more than the stats here. You can see that the onset of pain relief was fairly quick, and that pain relief was sustained over time. You can also see that this is not a miracle drug. Pain scores were a bit better 48 hours out, but only by about a point and a half.
Placebo isn’t really the fair comparison here; few of us treat our postabdominoplasty patients with placebo, after all. The authors do not formally compare the effect of VX-548 with that of the opioid hydrocodone, for instance. But that doesn’t stop us.
This graph, which I put together from data in the paper, shows pain control across the four randomization categories, with higher numbers indicating more (cumulative) control. While all the active agents do a bit better than placebo, VX-548 at the higher dose appears to do the best. But I should note that 5 mg of hydrocodone may not be an adequate dose for most people.
Yes, I would really have killed for an NSAID arm in this trial. Its absence, given that NSAIDs are a staple of postoperative care, is ... well, let’s just say, notable.
Although not a pain-destroying machine, VX-548 has some other things to recommend it. The receptor is really not found in the brain at all, which suggests that the drug should not carry much risk for dependency, though that has not been formally studied.
The side effects were generally mild – headache was the most common – and less prevalent than what you see even in the placebo arm.
Perhaps most notable is the fact that the rate of discontinuation of the study drug was lowest in the VX-548 arm. Patients could stop taking the pill they were assigned for any reason, ranging from perceived lack of efficacy to side effects. A low discontinuation rate indicates to me a sort of “voting with your feet” that suggests this might be a well-tolerated and reasonably effective drug.
VX-548 isn’t on the market yet; phase 3 trials are ongoing. But whether it is this particular drug or another in this class, I’m happy to see researchers trying to find new ways to target that most primeval form of suffering: pain.
Dr. Wilson is an associate professor of medicine and public health and director of Yale’s Clinical and Translational Research Accelerator, New Haven, Conn. He disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
When you stub your toe or get a paper cut on your finger, you feel the pain in that part of your body. It feels like the pain is coming from that place. But, of course, that’s not really what is happening. Pain doesn’t really happen in your toe or your finger. It happens in your brain.
It’s a game of telephone, really. The afferent nerve fiber detects the noxious stimulus, passing that signal to the second-order neuron in the dorsal root ganglia of the spinal cord, which runs it up to the thalamus to be passed to the third-order neuron which brings it to the cortex for localization and conscious perception. It’s not even a very good game of telephone. It takes about 100 ms for a pain signal to get from the hand to the brain – longer from the feet, given the greater distance. You see your foot hit the corner of the coffee table and have just enough time to think: “Oh no!” before the pain hits.
Given the Rube Goldberg nature of the process, it would seem like there are any number of places we could stop pain sensation. And sure, local anesthetics at the site of injury, or even spinal anesthetics, are powerful – if temporary and hard to administer – solutions to acute pain.
But in our everyday armamentarium, let’s be honest – we essentially have three options: opiates and opioids, which activate the mu-receptors in the brain to dull pain (and cause a host of other nasty side effects); NSAIDs, which block prostaglandin synthesis and thus limit the ability for pain-conducting neurons to get excited; and acetaminophen, which, despite being used for a century, is poorly understood.
But
If you were to zoom in on the connection between that first afferent pain fiber and the secondary nerve in the spinal cord dorsal root ganglion, you would see a receptor called Nav1.8, a voltage-gated sodium channel.
This receptor is a key part of the apparatus that passes information from nerve 1 to nerve 2, but only for fibers that transmit pain signals. In fact, humans with mutations in this receptor that leave it always in the “open” state have a severe pain syndrome. Blocking the receptor, therefore, might reduce pain.
In preclinical work, researchers identified VX-548, which doesn’t have a brand name yet, as a potent blocker of that channel even in nanomolar concentrations. Importantly, the compound was highly selective for that particular channel – about 30,000 times more selective than it was for the other sodium channels in that family.
Of course, a highly selective and specific drug does not a blockbuster analgesic make. To determine how this drug would work on humans in pain, they turned to two populations: 303 individuals undergoing abdominoplasty and 274 undergoing bunionectomy, as reported in a new paper in the New England Journal of Medicine.
I know this seems a bit random, but abdominoplasty is quite painful and a good model for soft-tissue pain. Bunionectomy is also quite a painful procedure and a useful model of bone pain. After the surgeries, patients were randomized to several different doses of VX-548, hydrocodone plus acetaminophen, or placebo for 48 hours.
At 19 time points over that 48-hour period, participants were asked to rate their pain on a scale from 0 to 10. The primary outcome was the cumulative pain experienced over the 48 hours. So, higher pain would be worse here, but longer duration of pain would also be worse.
The story of the study is really told in this chart.
Yes, those assigned to the highest dose of VX-548 had a statistically significant lower cumulative amount of pain in the 48 hours after surgery. But the picture is really worth more than the stats here. You can see that the onset of pain relief was fairly quick, and that pain relief was sustained over time. You can also see that this is not a miracle drug. Pain scores were a bit better 48 hours out, but only by about a point and a half.
Placebo isn’t really the fair comparison here; few of us treat our postabdominoplasty patients with placebo, after all. The authors do not formally compare the effect of VX-548 with that of the opioid hydrocodone, for instance. But that doesn’t stop us.
This graph, which I put together from data in the paper, shows pain control across the four randomization categories, with higher numbers indicating more (cumulative) control. While all the active agents do a bit better than placebo, VX-548 at the higher dose appears to do the best. But I should note that 5 mg of hydrocodone may not be an adequate dose for most people.
Yes, I would really have killed for an NSAID arm in this trial. Its absence, given that NSAIDs are a staple of postoperative care, is ... well, let’s just say, notable.
Although not a pain-destroying machine, VX-548 has some other things to recommend it. The receptor is really not found in the brain at all, which suggests that the drug should not carry much risk for dependency, though that has not been formally studied.
The side effects were generally mild – headache was the most common – and less prevalent than what you see even in the placebo arm.
Perhaps most notable is the fact that the rate of discontinuation of the study drug was lowest in the VX-548 arm. Patients could stop taking the pill they were assigned for any reason, ranging from perceived lack of efficacy to side effects. A low discontinuation rate indicates to me a sort of “voting with your feet” that suggests this might be a well-tolerated and reasonably effective drug.
VX-548 isn’t on the market yet; phase 3 trials are ongoing. But whether it is this particular drug or another in this class, I’m happy to see researchers trying to find new ways to target that most primeval form of suffering: pain.
Dr. Wilson is an associate professor of medicine and public health and director of Yale’s Clinical and Translational Research Accelerator, New Haven, Conn. He disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.






