User login
Intensive glucose control after acute ischemic stroke does not improve functional outcomes
Background: Higher glucose immediately following acute ischemic stroke is known to be associated with poor outcomes. Patients with elevated glucoses in the aftermath of an acute ischemic stroke are more likely to have expansion of ischemic area and are more likely to have hemorrhagic conversion.
Study design: Randomized, controlled trial, with blinded outcome assessment.
Setting: 63 sites in the United States.
Synopsis: A total of 1,151 patients were randomized to either intensive (goal blood glucose, 80-130 mg/dL) or standard (goal blood glucose, 80-179 mg/dL) glucose control for up to the first 72 hours after presenting with acute ischemic stroke. Patients in the intensive control group were given continuous IV insulin and patients in the standard control group were given subcutaneous sliding. There was no difference between groups (intensive vs. standard) with regards to the primary outcome, which was the percentage of patients who achieved a modified Rankin Score at 90 days of 0-2 (20.5% vs 21.6%; adjusted relative risk, 0.97; 95% confidence interval, 0.87-1.08; P = .55). Severe hypoglycemia (blood glucose of less than 40 mg/dL) occurred in the intensive control group only. The American Heart Association/American Stroke Association guidelines support target blood glucose of 140-180 mg/dL, though limited evidence to support this guideline is noted.
Bottom line: Patients who underwent intensive glucose control regimens following acute ischemic stroke did not have significantly different functional outcomes at 90 days than those who had standard glucose control therapy.
Citation: Johnston KC et al. Intensive vs. standard treatment of hyperglycemia and functional outcome in patients with acute ischemic stroke: The SHINE randomized clinical trial. JAMA. 2019 Jul 23/30;322(4):326-35.
Dr. Fritz is assistant professor of medicine and the director of hospitalist operations at St. Louis University School of Medicine.
Background: Higher glucose immediately following acute ischemic stroke is known to be associated with poor outcomes. Patients with elevated glucoses in the aftermath of an acute ischemic stroke are more likely to have expansion of ischemic area and are more likely to have hemorrhagic conversion.
Study design: Randomized, controlled trial, with blinded outcome assessment.
Setting: 63 sites in the United States.
Synopsis: A total of 1,151 patients were randomized to either intensive (goal blood glucose, 80-130 mg/dL) or standard (goal blood glucose, 80-179 mg/dL) glucose control for up to the first 72 hours after presenting with acute ischemic stroke. Patients in the intensive control group were given continuous IV insulin and patients in the standard control group were given subcutaneous sliding. There was no difference between groups (intensive vs. standard) with regards to the primary outcome, which was the percentage of patients who achieved a modified Rankin Score at 90 days of 0-2 (20.5% vs 21.6%; adjusted relative risk, 0.97; 95% confidence interval, 0.87-1.08; P = .55). Severe hypoglycemia (blood glucose of less than 40 mg/dL) occurred in the intensive control group only. The American Heart Association/American Stroke Association guidelines support target blood glucose of 140-180 mg/dL, though limited evidence to support this guideline is noted.
Bottom line: Patients who underwent intensive glucose control regimens following acute ischemic stroke did not have significantly different functional outcomes at 90 days than those who had standard glucose control therapy.
Citation: Johnston KC et al. Intensive vs. standard treatment of hyperglycemia and functional outcome in patients with acute ischemic stroke: The SHINE randomized clinical trial. JAMA. 2019 Jul 23/30;322(4):326-35.
Dr. Fritz is assistant professor of medicine and the director of hospitalist operations at St. Louis University School of Medicine.
Background: Higher glucose immediately following acute ischemic stroke is known to be associated with poor outcomes. Patients with elevated glucoses in the aftermath of an acute ischemic stroke are more likely to have expansion of ischemic area and are more likely to have hemorrhagic conversion.
Study design: Randomized, controlled trial, with blinded outcome assessment.
Setting: 63 sites in the United States.
Synopsis: A total of 1,151 patients were randomized to either intensive (goal blood glucose, 80-130 mg/dL) or standard (goal blood glucose, 80-179 mg/dL) glucose control for up to the first 72 hours after presenting with acute ischemic stroke. Patients in the intensive control group were given continuous IV insulin and patients in the standard control group were given subcutaneous sliding. There was no difference between groups (intensive vs. standard) with regards to the primary outcome, which was the percentage of patients who achieved a modified Rankin Score at 90 days of 0-2 (20.5% vs 21.6%; adjusted relative risk, 0.97; 95% confidence interval, 0.87-1.08; P = .55). Severe hypoglycemia (blood glucose of less than 40 mg/dL) occurred in the intensive control group only. The American Heart Association/American Stroke Association guidelines support target blood glucose of 140-180 mg/dL, though limited evidence to support this guideline is noted.
Bottom line: Patients who underwent intensive glucose control regimens following acute ischemic stroke did not have significantly different functional outcomes at 90 days than those who had standard glucose control therapy.
Citation: Johnston KC et al. Intensive vs. standard treatment of hyperglycemia and functional outcome in patients with acute ischemic stroke: The SHINE randomized clinical trial. JAMA. 2019 Jul 23/30;322(4):326-35.
Dr. Fritz is assistant professor of medicine and the director of hospitalist operations at St. Louis University School of Medicine.
CDC shortens COVID-19 quarantine time to 10 or 7 days, with conditions
Citing new evidence and an “acceptable risk” of transmission, the agency hopes reducing the 14-day quarantine will increase overall compliance and improve public health and economic constraints.
The agency also suggested people postpone travel during the upcoming winter holidays and stay home because of the pandemic.
These shorter quarantine options do not replace initial CDC guidance. “CDC continues to recommend quarantining for 14 days as the best way to reduce risk for spreading COVID-19,” said Henry Walke, MD, MPH, the CDC’s COVID-19 incident manager, during a media briefing on Wednesday.
However, “after reviewing and analyzing new research and data, CDC has identified two acceptable alternative quarantine periods.”
People can now quarantine for 10 days without a COVID-19 test if they have no symptoms. Alternatively, a quarantine can end after 7 days for someone with a negative test and no symptoms. The agency recommends a polymerase chain reaction test or an antigen assay within 48 hours before the end of a quarantine.
The agency also suggests people still monitor for symptoms for a full 14 days.
Reducing the length of quarantine “may make it easier for people to take this critical public health action, by reducing the economic hardship associated with a longer period, especially if they cannot work during that time,” Dr. Walke said. “In addition, a shorter quarantine period can lessen stress on the public health system and communities, especially when new infections are rapidly rising.”
The federal guidance leaves flexibility for local jurisdictions to make their own quarantine recommendations, as warranted, he added.
An ‘acceptable risk’ calculation
Modeling by the CDC and academic and public health partners led to the new quarantine recommendations, said John Brooks, MD, chief medical officer for the CDC’s COVID-19 response. Multiple studies “point in the same direction, which is that we can safely reduce the length of quarantine but accept there is a small residual risk that a person who is leaving quarantine early could transmit to someone else.”
The residual risk is approximately 1%, with an upper limit of 10%, when people quarantine for 10 days. A 7-day quarantine carries a residual risk of about 5% and an upper limit of 12%.
“Ten days is where the risk got into a sweet spot we like, at about 1%,” Dr. Brooks said. “That is a very acceptable risk, I think, for many people.”
Although it remains unknown what proportion of people spending 14 days in quarantine leave early, “we are hearing anecdotally from our partners in public health that many people are discontinuing quarantine ahead of time because there is pressure to go back to work, to get people back into school – and it imposes a burden on the individual,” Dr. Brooks said.
“One of our hopes is that ... if we reduce the amount of time they have to spend in quarantine, people will be more compliant,” he added.
A reporter asked why the CDC is shortening quarantines when the pandemic numbers are increasing nationwide. The timing has to do with capacity, Dr. Brooks said. “We are in situation where the number of cases is rising, the number of contacts is rising and the number of people who require quarantine is rising. That is a lot of burden, not just on the people who have to quarantine, but on public health.”
Home for the holidays
Similar to its pre-Thanksgiving advisory, the CDC also recommends people avoid travel during the upcoming winter holidays. “The best way to protect yourself and others is to postpone travel and stay home,” Dr. Walke said.
If people do decide to travel, the agency recommends COVID-19 testing 1-3 days prior to travel and again 3-5 days afterward, as well as reducing nonessential activities for a full 7 days after returning home. Furthermore, if someone does not have follow-up testing, the CDC recommends reducing nonessential activities for 10 days.
Testing does not eliminate all risk, Dr. Walke said, “but when combined with reducing nonessential activities, symptom screening and continuing with precautions like wearing masks, social distancing and hand washing, it can make travel safer.”
“We are trying to reduce the number of infections by postponing travel over the winter holiday,” Cindy Friedman, MD, chief of the CDC Travelers’ Health Branch, said during the media briefing.
“Travel volume was high during Thanksgiving,” she said, “and even if only a small percentage of those travelers were asymptomatically infected, this can translate into hundreds of thousands of additional infections moving from one community to another.”
This article first appeared on Medscape.com.
Citing new evidence and an “acceptable risk” of transmission, the agency hopes reducing the 14-day quarantine will increase overall compliance and improve public health and economic constraints.
The agency also suggested people postpone travel during the upcoming winter holidays and stay home because of the pandemic.
These shorter quarantine options do not replace initial CDC guidance. “CDC continues to recommend quarantining for 14 days as the best way to reduce risk for spreading COVID-19,” said Henry Walke, MD, MPH, the CDC’s COVID-19 incident manager, during a media briefing on Wednesday.
However, “after reviewing and analyzing new research and data, CDC has identified two acceptable alternative quarantine periods.”
People can now quarantine for 10 days without a COVID-19 test if they have no symptoms. Alternatively, a quarantine can end after 7 days for someone with a negative test and no symptoms. The agency recommends a polymerase chain reaction test or an antigen assay within 48 hours before the end of a quarantine.
The agency also suggests people still monitor for symptoms for a full 14 days.
Reducing the length of quarantine “may make it easier for people to take this critical public health action, by reducing the economic hardship associated with a longer period, especially if they cannot work during that time,” Dr. Walke said. “In addition, a shorter quarantine period can lessen stress on the public health system and communities, especially when new infections are rapidly rising.”
The federal guidance leaves flexibility for local jurisdictions to make their own quarantine recommendations, as warranted, he added.
An ‘acceptable risk’ calculation
Modeling by the CDC and academic and public health partners led to the new quarantine recommendations, said John Brooks, MD, chief medical officer for the CDC’s COVID-19 response. Multiple studies “point in the same direction, which is that we can safely reduce the length of quarantine but accept there is a small residual risk that a person who is leaving quarantine early could transmit to someone else.”
The residual risk is approximately 1%, with an upper limit of 10%, when people quarantine for 10 days. A 7-day quarantine carries a residual risk of about 5% and an upper limit of 12%.
“Ten days is where the risk got into a sweet spot we like, at about 1%,” Dr. Brooks said. “That is a very acceptable risk, I think, for many people.”
Although it remains unknown what proportion of people spending 14 days in quarantine leave early, “we are hearing anecdotally from our partners in public health that many people are discontinuing quarantine ahead of time because there is pressure to go back to work, to get people back into school – and it imposes a burden on the individual,” Dr. Brooks said.
“One of our hopes is that ... if we reduce the amount of time they have to spend in quarantine, people will be more compliant,” he added.
A reporter asked why the CDC is shortening quarantines when the pandemic numbers are increasing nationwide. The timing has to do with capacity, Dr. Brooks said. “We are in situation where the number of cases is rising, the number of contacts is rising and the number of people who require quarantine is rising. That is a lot of burden, not just on the people who have to quarantine, but on public health.”
Home for the holidays
Similar to its pre-Thanksgiving advisory, the CDC also recommends people avoid travel during the upcoming winter holidays. “The best way to protect yourself and others is to postpone travel and stay home,” Dr. Walke said.
If people do decide to travel, the agency recommends COVID-19 testing 1-3 days prior to travel and again 3-5 days afterward, as well as reducing nonessential activities for a full 7 days after returning home. Furthermore, if someone does not have follow-up testing, the CDC recommends reducing nonessential activities for 10 days.
Testing does not eliminate all risk, Dr. Walke said, “but when combined with reducing nonessential activities, symptom screening and continuing with precautions like wearing masks, social distancing and hand washing, it can make travel safer.”
“We are trying to reduce the number of infections by postponing travel over the winter holiday,” Cindy Friedman, MD, chief of the CDC Travelers’ Health Branch, said during the media briefing.
“Travel volume was high during Thanksgiving,” she said, “and even if only a small percentage of those travelers were asymptomatically infected, this can translate into hundreds of thousands of additional infections moving from one community to another.”
This article first appeared on Medscape.com.
Citing new evidence and an “acceptable risk” of transmission, the agency hopes reducing the 14-day quarantine will increase overall compliance and improve public health and economic constraints.
The agency also suggested people postpone travel during the upcoming winter holidays and stay home because of the pandemic.
These shorter quarantine options do not replace initial CDC guidance. “CDC continues to recommend quarantining for 14 days as the best way to reduce risk for spreading COVID-19,” said Henry Walke, MD, MPH, the CDC’s COVID-19 incident manager, during a media briefing on Wednesday.
However, “after reviewing and analyzing new research and data, CDC has identified two acceptable alternative quarantine periods.”
People can now quarantine for 10 days without a COVID-19 test if they have no symptoms. Alternatively, a quarantine can end after 7 days for someone with a negative test and no symptoms. The agency recommends a polymerase chain reaction test or an antigen assay within 48 hours before the end of a quarantine.
The agency also suggests people still monitor for symptoms for a full 14 days.
Reducing the length of quarantine “may make it easier for people to take this critical public health action, by reducing the economic hardship associated with a longer period, especially if they cannot work during that time,” Dr. Walke said. “In addition, a shorter quarantine period can lessen stress on the public health system and communities, especially when new infections are rapidly rising.”
The federal guidance leaves flexibility for local jurisdictions to make their own quarantine recommendations, as warranted, he added.
An ‘acceptable risk’ calculation
Modeling by the CDC and academic and public health partners led to the new quarantine recommendations, said John Brooks, MD, chief medical officer for the CDC’s COVID-19 response. Multiple studies “point in the same direction, which is that we can safely reduce the length of quarantine but accept there is a small residual risk that a person who is leaving quarantine early could transmit to someone else.”
The residual risk is approximately 1%, with an upper limit of 10%, when people quarantine for 10 days. A 7-day quarantine carries a residual risk of about 5% and an upper limit of 12%.
“Ten days is where the risk got into a sweet spot we like, at about 1%,” Dr. Brooks said. “That is a very acceptable risk, I think, for many people.”
Although it remains unknown what proportion of people spending 14 days in quarantine leave early, “we are hearing anecdotally from our partners in public health that many people are discontinuing quarantine ahead of time because there is pressure to go back to work, to get people back into school – and it imposes a burden on the individual,” Dr. Brooks said.
“One of our hopes is that ... if we reduce the amount of time they have to spend in quarantine, people will be more compliant,” he added.
A reporter asked why the CDC is shortening quarantines when the pandemic numbers are increasing nationwide. The timing has to do with capacity, Dr. Brooks said. “We are in situation where the number of cases is rising, the number of contacts is rising and the number of people who require quarantine is rising. That is a lot of burden, not just on the people who have to quarantine, but on public health.”
Home for the holidays
Similar to its pre-Thanksgiving advisory, the CDC also recommends people avoid travel during the upcoming winter holidays. “The best way to protect yourself and others is to postpone travel and stay home,” Dr. Walke said.
If people do decide to travel, the agency recommends COVID-19 testing 1-3 days prior to travel and again 3-5 days afterward, as well as reducing nonessential activities for a full 7 days after returning home. Furthermore, if someone does not have follow-up testing, the CDC recommends reducing nonessential activities for 10 days.
Testing does not eliminate all risk, Dr. Walke said, “but when combined with reducing nonessential activities, symptom screening and continuing with precautions like wearing masks, social distancing and hand washing, it can make travel safer.”
“We are trying to reduce the number of infections by postponing travel over the winter holiday,” Cindy Friedman, MD, chief of the CDC Travelers’ Health Branch, said during the media briefing.
“Travel volume was high during Thanksgiving,” she said, “and even if only a small percentage of those travelers were asymptomatically infected, this can translate into hundreds of thousands of additional infections moving from one community to another.”
This article first appeared on Medscape.com.
CRP testing in acute COPD exacerbations cuts antibiotic use without compromising outcomes
Background: A previous study has shown that patients with acute COPD exacerbations had little difference in rate of clinical cure with placebo or antibiotics when CRP is less than 40 mg/L.
Study design: Multicenter, open-label, randomized controlled trial.
Setting: 86 general medical practices in the United Kingdom from January 2015 through September 2017.
Synopsis: More than 600 patients who presented to a primary care physician with an acute COPD exacerbation were randomized to point of care CRP testing vs. usual care. Clinicians in the CRP testing group were provided with a point-of-care testing unit along with an algorithm for results. If the CRP was greater than 40 mg/L, antibiotics were thought to be beneficial; but they were urged not to prescribe antibiotics if the level was less than 20 mg/L. For levels between 20 mg/L and 40 mg/L, it was suggested that antibiotics might be beneficial if the sputum is purulent.
The primary outcomes were patient-reported use of antibiotics for an acute COPD exacerbation within 4 weeks of randomization along with measurement of COPD-related health status on the Clinical COPD Questionnaire at 2 weeks of randomization. Fewer antibiotics were prescribed in the CRP testing group over the usual care group (57% vs. 77%). The adjusted mean difference in the Clinical COPD Questionnaire total score at 2 weeks was –0.19 points, in favor of the CRP-guided group.
Bottom line: The use of point-of-care testing CRP as an adjunctive guide to antibiotic use in acute COPD exacerbations may lower the amount of antibiotic prescribing without compromising clinical outcomes.
Citation: Butler CC et al. C-reactive protein testing to guide antibiotics prescribing for COPD exacerbations. N Engl J Med. 2019 Jul 11; 381:111-120.
Dr. Choksi is a hospitalist and associate professor of internal medicine at Saint Louis University, where she is assistant dean of admissions. She is president of the SHM St. Louis Chapter.
Background: A previous study has shown that patients with acute COPD exacerbations had little difference in rate of clinical cure with placebo or antibiotics when CRP is less than 40 mg/L.
Study design: Multicenter, open-label, randomized controlled trial.
Setting: 86 general medical practices in the United Kingdom from January 2015 through September 2017.
Synopsis: More than 600 patients who presented to a primary care physician with an acute COPD exacerbation were randomized to point of care CRP testing vs. usual care. Clinicians in the CRP testing group were provided with a point-of-care testing unit along with an algorithm for results. If the CRP was greater than 40 mg/L, antibiotics were thought to be beneficial; but they were urged not to prescribe antibiotics if the level was less than 20 mg/L. For levels between 20 mg/L and 40 mg/L, it was suggested that antibiotics might be beneficial if the sputum is purulent.
The primary outcomes were patient-reported use of antibiotics for an acute COPD exacerbation within 4 weeks of randomization along with measurement of COPD-related health status on the Clinical COPD Questionnaire at 2 weeks of randomization. Fewer antibiotics were prescribed in the CRP testing group over the usual care group (57% vs. 77%). The adjusted mean difference in the Clinical COPD Questionnaire total score at 2 weeks was –0.19 points, in favor of the CRP-guided group.
Bottom line: The use of point-of-care testing CRP as an adjunctive guide to antibiotic use in acute COPD exacerbations may lower the amount of antibiotic prescribing without compromising clinical outcomes.
Citation: Butler CC et al. C-reactive protein testing to guide antibiotics prescribing for COPD exacerbations. N Engl J Med. 2019 Jul 11; 381:111-120.
Dr. Choksi is a hospitalist and associate professor of internal medicine at Saint Louis University, where she is assistant dean of admissions. She is president of the SHM St. Louis Chapter.
Background: A previous study has shown that patients with acute COPD exacerbations had little difference in rate of clinical cure with placebo or antibiotics when CRP is less than 40 mg/L.
Study design: Multicenter, open-label, randomized controlled trial.
Setting: 86 general medical practices in the United Kingdom from January 2015 through September 2017.
Synopsis: More than 600 patients who presented to a primary care physician with an acute COPD exacerbation were randomized to point of care CRP testing vs. usual care. Clinicians in the CRP testing group were provided with a point-of-care testing unit along with an algorithm for results. If the CRP was greater than 40 mg/L, antibiotics were thought to be beneficial; but they were urged not to prescribe antibiotics if the level was less than 20 mg/L. For levels between 20 mg/L and 40 mg/L, it was suggested that antibiotics might be beneficial if the sputum is purulent.
The primary outcomes were patient-reported use of antibiotics for an acute COPD exacerbation within 4 weeks of randomization along with measurement of COPD-related health status on the Clinical COPD Questionnaire at 2 weeks of randomization. Fewer antibiotics were prescribed in the CRP testing group over the usual care group (57% vs. 77%). The adjusted mean difference in the Clinical COPD Questionnaire total score at 2 weeks was –0.19 points, in favor of the CRP-guided group.
Bottom line: The use of point-of-care testing CRP as an adjunctive guide to antibiotic use in acute COPD exacerbations may lower the amount of antibiotic prescribing without compromising clinical outcomes.
Citation: Butler CC et al. C-reactive protein testing to guide antibiotics prescribing for COPD exacerbations. N Engl J Med. 2019 Jul 11; 381:111-120.
Dr. Choksi is a hospitalist and associate professor of internal medicine at Saint Louis University, where she is assistant dean of admissions. She is president of the SHM St. Louis Chapter.
Three pillars of a successful coronavirus vaccine program in minorities
As COVID-19 cases soared to new daily highs across the United States, November 2020 brought some exciting and promising vaccine efficacy results. Currently, the United States has four COVID-19 vaccines in phase 3 trials: the Moderna vaccine (mRNA-1273), the Oxford/AstraZeneca vaccine (AZD1222), Pfizer/BioNTech’s (BNT162), and the Johnson & Johnson vaccine (JNJ-78436735).
While Pfizer/ BioNTech and Moderna received fast-track designation by the Food and Drug Administration, AZD1222 and JNJ-78436735 trials were resumed after a temporary hold. Pfizer/BioNTech and Moderna have also submitted an emergency-use authorization application to the FDA after favorable results from a completed phase 3 clinical trial. The results so far seem promising, with Oxford/AstraZeneca’s combined analysis from different dosing regimens resulting in an average efficacy of 70%. Pfizer/ BioNTech and Moderna have each reported vaccines that are 90% and 95% effective respectively in trials.
However, even with a safe and effective vaccine, there must be an equal emphasis on a successful coronavirus vaccine program’s three pillars in the communities that are the hardest hit: participation in the vaccine trials by minority populations, equitable allocation and distribution of vaccine for minority populations, and immunization uptake by minority populations.
1. Participation in the vaccine trials by minority populations
With a great emphasis on the inclusion of diverse populations, the Moderna vaccine clinical trials gained participation by racial and ethnic minorities. As of Oct. 21, 2020, the Moderna vaccine trial participants were 10% African American, 20% Hispanic, 4% Asian, 63% White, and 3% other.1 Pharmaceutical giant Pfizer also had approximately 42% of overall – and 45% of U.S. – participants from diverse backgrounds. The proportional registration of racially and ethnically diverse participants in other vaccine trials is also anticipated to be challenging.
Though there has been an improvement in minority participation in COVID-19 vaccine trials, it is still below the ideal representation when compared with U.S. census data.2 Ideally, participants in a clinical trial should represent the U.S. population to get a full picture of a medical product’s risks and benefits. However, recruitment rates in clinical trials have remained low among minorities for various reasons. Historically, African Americans make up only 5% of participants in U.S. clinical trials, while they represent 13% of the country’s general population; likewise, Hispanics are also underrepresented.3
The legacy of distrust in the medical system is deep-rooted and is one of the most substantial barriers to clinical trial participation. A plethora of unethical trials and experiments on the African American population have left a lasting impact. The most infamous and widely known was the “Tuskegee Study,” conducted by the United States Public Health Service to “observe the natural history of untreated syphilis” in Black populations. In the study, performed without informed consent, Black men with latent or late syphilis received no treatment, even after penicillin was discovered as a safe and reliable cure for syphilis. This human experimentation lasted for 40 years, resulting in 128 male patients who died from syphilis or its complications, 40 of their spouses infected, and 19 of their children with acquired congenital syphilis.
In another case, the father of modern gynecology, J. Marion Sims, allegedly performed experimental surgeries on enslaved Black women without consent. For more than 4 decades, North Carolina’s statewide eugenics program forcibly sterilized almost 7,600 people, many of whom were Black. Another story of exploitation involves Henrietta Lacks, whose cancer cells are the source of the HeLa cell line, responsible for some of the most important medical advances of all time. Though her cells were commercialized and generated millions for medical researchers, neither Ms. Lacks nor her family knew the cell cultures existed until more than 20 years after her death from cervical cancer. Many years later, victims and families of the Tuskegee experiment, individuals sterilized by the Eugenics Board of North Carolina, and the family of Henrietta Lacks received compensation, and Sims’s statue was taken down in 2018. Not too long ago, many criticized the FDA’s “Exception from Informed Consent policy” for compromising patients’ exercise of autonomy, and concern for overrepresenting African Americans in the U.S. EFIC trials.
Racial disparities in medical treatment and unconscious biases among providers are among the reasons for mistrust and lack of trial participation by minority populations today. Francis Collins, director of the National Institutes of Health, said that recent social upheaval sparked by the death of George Floyd has likely added to feelings of mistrust between minority groups and government or pharmaceutical companies. “Yet we need their participation if this is going to have a meaningful outcome,” he said.
While “Operation Warp Speed” is committed to developing and delivering a COVID-19 vaccine rapidly while adhering to safety and efficacy standards, the challenges to enrolling people from racial and ethnic minorities in trials have been a concern. The political partisanship and ever-shifting stances on widespread COVID-19 testing, use of facemasks, endorsement of unproven drugs for the disease, and accusations against the FDA for delaying human trials for the vaccine have contributed to the skepticism as well. Tremendous pressure for a rushed vaccine with unrealistic timelines, recent holds on AZD1222 and JNJ-78436735 as well as the AZD1222 dosage error during trials have also raised skepticism of the safety and efficacy of vaccine trials.
2. Equitable allocation and distribution of vaccine for minority populations
Enrollment in clinical trials is just a beginning; a more significant challenge would be the vaccine’s uptake when available to the general public. We still lack a consensus on whether it is lawful for race to be an explicit criterion for priority distribution of the COVID-19 vaccine. Recently the Centers for Disease Control and Prevention suggested that the vaccine amount allotted to jurisdictions might be based on critical populations recommended for vaccination by the Advisory Committee on Immunization Practices with input from the National Academies of Sciences, Engineering, and Medicine.
The NASEM framework lays out four-phased vaccine distribution approaches, emphasizing social equity by prioritizing vaccines for geographic areas identified through CDC’s social vulnerability index (SVI) or another more specific index. SVI has been a robust composite marker of minority status and language, household composition and transportation, and housing and disability, and predicted COVID-19 case counts in the United States in several studies. The National Academy of Medicine has also recommended racial minorities receive priority vaccination because they have been hard hit and are “worse off” socioeconomically.
3. Immunization uptake by minority populations
Though minority participation is crucial in developing the vaccine, more transparency, open discussions on ethical distribution, and awareness of side effects are required before vaccine approval or emergency-use authorization. Companies behind the four major COVID-19 vaccines in development have released their trials’ protocols, details on vaccine efficacy, and each product’s makeup to increase acceptance of the vaccine.
According to a recent Pew research study, about half of U.S. adults (51%) now say they would definitely or probably get a vaccine to prevent COVID-19 if it were available today. Nearly as many (49%) say they definitely or probably would not get vaccinated at this time. Intent to get a COVID-19 vaccine has fallen from 72% in May 2020, a 21–percentage point drop, and Black adults were much less likely to say they would get a vaccine than other Americans.3 This is concerning as previous studies have shown that race and ethnicity can influence immune responses to vaccination. There is evidence of racial and ethnic differences in immune response following rubella vaccination, Hib–tetanus toxoid conjugate vaccine, antibody responses to the influenza A virus components of IIV3 or 4, and immune responses after measles vaccination.4-9
On the other hand, significant differences in reporting rates of adverse events after human papillomavirus vaccinations were found in different race and ethnicity groups in the Vaccine Adverse Event Reporting System.10 Thus, there is ample evidence that race and ethnicity affect responsiveness to a vaccine. Inequity in participation in a clinical trial may lead to an ineffective or one with a suboptimal response or even an unsafe vaccine.
When we look at other immunization programs, according to various surveys in recent years, non-Hispanic Blacks have lower annual vaccination rates for flu, pneumonia, and human papillomavirus vaccinations nationally, compared with non-Hispanic White adults.11 It is a cause of concern as a proportion of the population must be vaccinated to reach “community immunity” or “herd immunity” from vaccination. Depending on varying biological, environmental, and sociobehavioral factors, the threshold for COVID-19 herd immunity may be between 55% and 82% of the population.12 Hence, neither a vaccine trial nor an immunization program can succeed without participation from all communities and age groups.
Role of hospitalists
Hospitalists, who give immunizations as part of the hospital inpatient quality reporting program, are uniquely placed in this pandemic. Working on the front lines, we may encounter questions, concerns, rejections, and discussions about the pros and cons of the COVID-19 vaccine from patients.
Investigators at Children’s National Hospital and George Washington University, both in Washington, recently recommended three steps physicians and others can take now to ensure more people get the COVID-19 vaccine when it is available. Engaging frontline health professionals was one of the suggested steps to encourage more people to get the vaccine.13 However, it is imperative to understand that vaccine hesitancy might be an issue for health care providers as well, if concerns for scientific standards and involvement of diverse populations are not addressed.
We are only starting to develop a safe and effective immunization program. We must bring more to unrepresented communities than just vaccine trials. Information, education, availability, and access to the vaccines will make for a successful COVID-19 immunization program.
Dr. Saigal is a hospitalist and clinical assistant professor of medicine in the division of hospital medicine at the Ohio State University Wexner Medical Center, Columbus.
References
1. Moderna. COVE study. 2020 Oct 21. https://www.modernatx.com/sites/default/files/content_documents/2020-COVE-Study-Enrollment-Completion-10.22.20.pdf
2. U.S. Census Bureau. Quick facts: Population estimates, July 1, 2019. https://www.census.gov/quickfacts/fact/table/US/PST045219
3. Pew Research Center. U.S. Public Now Divided Over Whether To Get COVID-19 Vaccine. 2020 Sep 17. https://www.pewresearch.org/science/2020/09/17/u-s-public-now-divided-over-whether-to-get-covid-19-vaccine/
4. Haralambieva IH et al. Associations between race sex and immune response variations to rubella vaccination in two independent cohorts. Vaccine. 2014;32:1946-53.
5. McQuillan GM et al. Seroprevalence of measles antibody in the U.S. population 1999-2004. J Infect Dis. 2007;196:1459–64. doi: 10.1086/522866.
6. Christy C et al. Effect of gender race and parental education on immunogenicity and reported reactogenicity of acellular and whole-cell pertussis vaccines. Pediatrics. 1995;96:584-7.
7. Poland GA et al. Measles antibody seroprevalence rates among immunized Inuit Innu and Caucasian subjects. Vaccine. 1999;17:1525-31.
8. Greenberg DP et al. Immunogenicity of Haemophilus influenzae type b tetanus toxoid conjugate vaccine in young infants. The Kaiser-UCLA Vaccine Study Group. J Infect Dis. 1994;170:76-81.
9. Kurupati R et al. Race-related differences in antibody responses to the inactivated influenza vaccine are linked to distinct prevaccination gene expression profiles in blood. Oncotarget. 2016;7(39):62898-911.
10. Huang J et al. Characterization of the differential adverse event rates by race/ethnicity groups for HPV vaccine by integrating data from different sources. Front Pharmacol. 2018;9:539.
11. https://minorityhealth.hhs.gov/omh/browse.aspx?lvl=4&lvlid=22
12. Sanche S et al. High contagiousness and rapid spread of severe acute respiratory syndrome coronavirus 2. Emerg Infect Dis. 2020;26(7).
13. American Medical Association. How to ready patients now so they’ll get a COVID-19 vaccine later. 2020 May 27. https://www.ama-assn.org/delivering-care/public-health/how-ready-patients-now-so-they-ll-get-covid-19-vaccine-later
As COVID-19 cases soared to new daily highs across the United States, November 2020 brought some exciting and promising vaccine efficacy results. Currently, the United States has four COVID-19 vaccines in phase 3 trials: the Moderna vaccine (mRNA-1273), the Oxford/AstraZeneca vaccine (AZD1222), Pfizer/BioNTech’s (BNT162), and the Johnson & Johnson vaccine (JNJ-78436735).
While Pfizer/ BioNTech and Moderna received fast-track designation by the Food and Drug Administration, AZD1222 and JNJ-78436735 trials were resumed after a temporary hold. Pfizer/BioNTech and Moderna have also submitted an emergency-use authorization application to the FDA after favorable results from a completed phase 3 clinical trial. The results so far seem promising, with Oxford/AstraZeneca’s combined analysis from different dosing regimens resulting in an average efficacy of 70%. Pfizer/ BioNTech and Moderna have each reported vaccines that are 90% and 95% effective respectively in trials.
However, even with a safe and effective vaccine, there must be an equal emphasis on a successful coronavirus vaccine program’s three pillars in the communities that are the hardest hit: participation in the vaccine trials by minority populations, equitable allocation and distribution of vaccine for minority populations, and immunization uptake by minority populations.
1. Participation in the vaccine trials by minority populations
With a great emphasis on the inclusion of diverse populations, the Moderna vaccine clinical trials gained participation by racial and ethnic minorities. As of Oct. 21, 2020, the Moderna vaccine trial participants were 10% African American, 20% Hispanic, 4% Asian, 63% White, and 3% other.1 Pharmaceutical giant Pfizer also had approximately 42% of overall – and 45% of U.S. – participants from diverse backgrounds. The proportional registration of racially and ethnically diverse participants in other vaccine trials is also anticipated to be challenging.
Though there has been an improvement in minority participation in COVID-19 vaccine trials, it is still below the ideal representation when compared with U.S. census data.2 Ideally, participants in a clinical trial should represent the U.S. population to get a full picture of a medical product’s risks and benefits. However, recruitment rates in clinical trials have remained low among minorities for various reasons. Historically, African Americans make up only 5% of participants in U.S. clinical trials, while they represent 13% of the country’s general population; likewise, Hispanics are also underrepresented.3
The legacy of distrust in the medical system is deep-rooted and is one of the most substantial barriers to clinical trial participation. A plethora of unethical trials and experiments on the African American population have left a lasting impact. The most infamous and widely known was the “Tuskegee Study,” conducted by the United States Public Health Service to “observe the natural history of untreated syphilis” in Black populations. In the study, performed without informed consent, Black men with latent or late syphilis received no treatment, even after penicillin was discovered as a safe and reliable cure for syphilis. This human experimentation lasted for 40 years, resulting in 128 male patients who died from syphilis or its complications, 40 of their spouses infected, and 19 of their children with acquired congenital syphilis.
In another case, the father of modern gynecology, J. Marion Sims, allegedly performed experimental surgeries on enslaved Black women without consent. For more than 4 decades, North Carolina’s statewide eugenics program forcibly sterilized almost 7,600 people, many of whom were Black. Another story of exploitation involves Henrietta Lacks, whose cancer cells are the source of the HeLa cell line, responsible for some of the most important medical advances of all time. Though her cells were commercialized and generated millions for medical researchers, neither Ms. Lacks nor her family knew the cell cultures existed until more than 20 years after her death from cervical cancer. Many years later, victims and families of the Tuskegee experiment, individuals sterilized by the Eugenics Board of North Carolina, and the family of Henrietta Lacks received compensation, and Sims’s statue was taken down in 2018. Not too long ago, many criticized the FDA’s “Exception from Informed Consent policy” for compromising patients’ exercise of autonomy, and concern for overrepresenting African Americans in the U.S. EFIC trials.
Racial disparities in medical treatment and unconscious biases among providers are among the reasons for mistrust and lack of trial participation by minority populations today. Francis Collins, director of the National Institutes of Health, said that recent social upheaval sparked by the death of George Floyd has likely added to feelings of mistrust between minority groups and government or pharmaceutical companies. “Yet we need their participation if this is going to have a meaningful outcome,” he said.
While “Operation Warp Speed” is committed to developing and delivering a COVID-19 vaccine rapidly while adhering to safety and efficacy standards, the challenges to enrolling people from racial and ethnic minorities in trials have been a concern. The political partisanship and ever-shifting stances on widespread COVID-19 testing, use of facemasks, endorsement of unproven drugs for the disease, and accusations against the FDA for delaying human trials for the vaccine have contributed to the skepticism as well. Tremendous pressure for a rushed vaccine with unrealistic timelines, recent holds on AZD1222 and JNJ-78436735 as well as the AZD1222 dosage error during trials have also raised skepticism of the safety and efficacy of vaccine trials.
2. Equitable allocation and distribution of vaccine for minority populations
Enrollment in clinical trials is just a beginning; a more significant challenge would be the vaccine’s uptake when available to the general public. We still lack a consensus on whether it is lawful for race to be an explicit criterion for priority distribution of the COVID-19 vaccine. Recently the Centers for Disease Control and Prevention suggested that the vaccine amount allotted to jurisdictions might be based on critical populations recommended for vaccination by the Advisory Committee on Immunization Practices with input from the National Academies of Sciences, Engineering, and Medicine.
The NASEM framework lays out four-phased vaccine distribution approaches, emphasizing social equity by prioritizing vaccines for geographic areas identified through CDC’s social vulnerability index (SVI) or another more specific index. SVI has been a robust composite marker of minority status and language, household composition and transportation, and housing and disability, and predicted COVID-19 case counts in the United States in several studies. The National Academy of Medicine has also recommended racial minorities receive priority vaccination because they have been hard hit and are “worse off” socioeconomically.
3. Immunization uptake by minority populations
Though minority participation is crucial in developing the vaccine, more transparency, open discussions on ethical distribution, and awareness of side effects are required before vaccine approval or emergency-use authorization. Companies behind the four major COVID-19 vaccines in development have released their trials’ protocols, details on vaccine efficacy, and each product’s makeup to increase acceptance of the vaccine.
According to a recent Pew research study, about half of U.S. adults (51%) now say they would definitely or probably get a vaccine to prevent COVID-19 if it were available today. Nearly as many (49%) say they definitely or probably would not get vaccinated at this time. Intent to get a COVID-19 vaccine has fallen from 72% in May 2020, a 21–percentage point drop, and Black adults were much less likely to say they would get a vaccine than other Americans.3 This is concerning as previous studies have shown that race and ethnicity can influence immune responses to vaccination. There is evidence of racial and ethnic differences in immune response following rubella vaccination, Hib–tetanus toxoid conjugate vaccine, antibody responses to the influenza A virus components of IIV3 or 4, and immune responses after measles vaccination.4-9
On the other hand, significant differences in reporting rates of adverse events after human papillomavirus vaccinations were found in different race and ethnicity groups in the Vaccine Adverse Event Reporting System.10 Thus, there is ample evidence that race and ethnicity affect responsiveness to a vaccine. Inequity in participation in a clinical trial may lead to an ineffective or one with a suboptimal response or even an unsafe vaccine.
When we look at other immunization programs, according to various surveys in recent years, non-Hispanic Blacks have lower annual vaccination rates for flu, pneumonia, and human papillomavirus vaccinations nationally, compared with non-Hispanic White adults.11 It is a cause of concern as a proportion of the population must be vaccinated to reach “community immunity” or “herd immunity” from vaccination. Depending on varying biological, environmental, and sociobehavioral factors, the threshold for COVID-19 herd immunity may be between 55% and 82% of the population.12 Hence, neither a vaccine trial nor an immunization program can succeed without participation from all communities and age groups.
Role of hospitalists
Hospitalists, who give immunizations as part of the hospital inpatient quality reporting program, are uniquely placed in this pandemic. Working on the front lines, we may encounter questions, concerns, rejections, and discussions about the pros and cons of the COVID-19 vaccine from patients.
Investigators at Children’s National Hospital and George Washington University, both in Washington, recently recommended three steps physicians and others can take now to ensure more people get the COVID-19 vaccine when it is available. Engaging frontline health professionals was one of the suggested steps to encourage more people to get the vaccine.13 However, it is imperative to understand that vaccine hesitancy might be an issue for health care providers as well, if concerns for scientific standards and involvement of diverse populations are not addressed.
We are only starting to develop a safe and effective immunization program. We must bring more to unrepresented communities than just vaccine trials. Information, education, availability, and access to the vaccines will make for a successful COVID-19 immunization program.
Dr. Saigal is a hospitalist and clinical assistant professor of medicine in the division of hospital medicine at the Ohio State University Wexner Medical Center, Columbus.
References
1. Moderna. COVE study. 2020 Oct 21. https://www.modernatx.com/sites/default/files/content_documents/2020-COVE-Study-Enrollment-Completion-10.22.20.pdf
2. U.S. Census Bureau. Quick facts: Population estimates, July 1, 2019. https://www.census.gov/quickfacts/fact/table/US/PST045219
3. Pew Research Center. U.S. Public Now Divided Over Whether To Get COVID-19 Vaccine. 2020 Sep 17. https://www.pewresearch.org/science/2020/09/17/u-s-public-now-divided-over-whether-to-get-covid-19-vaccine/
4. Haralambieva IH et al. Associations between race sex and immune response variations to rubella vaccination in two independent cohorts. Vaccine. 2014;32:1946-53.
5. McQuillan GM et al. Seroprevalence of measles antibody in the U.S. population 1999-2004. J Infect Dis. 2007;196:1459–64. doi: 10.1086/522866.
6. Christy C et al. Effect of gender race and parental education on immunogenicity and reported reactogenicity of acellular and whole-cell pertussis vaccines. Pediatrics. 1995;96:584-7.
7. Poland GA et al. Measles antibody seroprevalence rates among immunized Inuit Innu and Caucasian subjects. Vaccine. 1999;17:1525-31.
8. Greenberg DP et al. Immunogenicity of Haemophilus influenzae type b tetanus toxoid conjugate vaccine in young infants. The Kaiser-UCLA Vaccine Study Group. J Infect Dis. 1994;170:76-81.
9. Kurupati R et al. Race-related differences in antibody responses to the inactivated influenza vaccine are linked to distinct prevaccination gene expression profiles in blood. Oncotarget. 2016;7(39):62898-911.
10. Huang J et al. Characterization of the differential adverse event rates by race/ethnicity groups for HPV vaccine by integrating data from different sources. Front Pharmacol. 2018;9:539.
11. https://minorityhealth.hhs.gov/omh/browse.aspx?lvl=4&lvlid=22
12. Sanche S et al. High contagiousness and rapid spread of severe acute respiratory syndrome coronavirus 2. Emerg Infect Dis. 2020;26(7).
13. American Medical Association. How to ready patients now so they’ll get a COVID-19 vaccine later. 2020 May 27. https://www.ama-assn.org/delivering-care/public-health/how-ready-patients-now-so-they-ll-get-covid-19-vaccine-later
As COVID-19 cases soared to new daily highs across the United States, November 2020 brought some exciting and promising vaccine efficacy results. Currently, the United States has four COVID-19 vaccines in phase 3 trials: the Moderna vaccine (mRNA-1273), the Oxford/AstraZeneca vaccine (AZD1222), Pfizer/BioNTech’s (BNT162), and the Johnson & Johnson vaccine (JNJ-78436735).
While Pfizer/ BioNTech and Moderna received fast-track designation by the Food and Drug Administration, AZD1222 and JNJ-78436735 trials were resumed after a temporary hold. Pfizer/BioNTech and Moderna have also submitted an emergency-use authorization application to the FDA after favorable results from a completed phase 3 clinical trial. The results so far seem promising, with Oxford/AstraZeneca’s combined analysis from different dosing regimens resulting in an average efficacy of 70%. Pfizer/ BioNTech and Moderna have each reported vaccines that are 90% and 95% effective respectively in trials.
However, even with a safe and effective vaccine, there must be an equal emphasis on a successful coronavirus vaccine program’s three pillars in the communities that are the hardest hit: participation in the vaccine trials by minority populations, equitable allocation and distribution of vaccine for minority populations, and immunization uptake by minority populations.
1. Participation in the vaccine trials by minority populations
With a great emphasis on the inclusion of diverse populations, the Moderna vaccine clinical trials gained participation by racial and ethnic minorities. As of Oct. 21, 2020, the Moderna vaccine trial participants were 10% African American, 20% Hispanic, 4% Asian, 63% White, and 3% other.1 Pharmaceutical giant Pfizer also had approximately 42% of overall – and 45% of U.S. – participants from diverse backgrounds. The proportional registration of racially and ethnically diverse participants in other vaccine trials is also anticipated to be challenging.
Though there has been an improvement in minority participation in COVID-19 vaccine trials, it is still below the ideal representation when compared with U.S. census data.2 Ideally, participants in a clinical trial should represent the U.S. population to get a full picture of a medical product’s risks and benefits. However, recruitment rates in clinical trials have remained low among minorities for various reasons. Historically, African Americans make up only 5% of participants in U.S. clinical trials, while they represent 13% of the country’s general population; likewise, Hispanics are also underrepresented.3
The legacy of distrust in the medical system is deep-rooted and is one of the most substantial barriers to clinical trial participation. A plethora of unethical trials and experiments on the African American population have left a lasting impact. The most infamous and widely known was the “Tuskegee Study,” conducted by the United States Public Health Service to “observe the natural history of untreated syphilis” in Black populations. In the study, performed without informed consent, Black men with latent or late syphilis received no treatment, even after penicillin was discovered as a safe and reliable cure for syphilis. This human experimentation lasted for 40 years, resulting in 128 male patients who died from syphilis or its complications, 40 of their spouses infected, and 19 of their children with acquired congenital syphilis.
In another case, the father of modern gynecology, J. Marion Sims, allegedly performed experimental surgeries on enslaved Black women without consent. For more than 4 decades, North Carolina’s statewide eugenics program forcibly sterilized almost 7,600 people, many of whom were Black. Another story of exploitation involves Henrietta Lacks, whose cancer cells are the source of the HeLa cell line, responsible for some of the most important medical advances of all time. Though her cells were commercialized and generated millions for medical researchers, neither Ms. Lacks nor her family knew the cell cultures existed until more than 20 years after her death from cervical cancer. Many years later, victims and families of the Tuskegee experiment, individuals sterilized by the Eugenics Board of North Carolina, and the family of Henrietta Lacks received compensation, and Sims’s statue was taken down in 2018. Not too long ago, many criticized the FDA’s “Exception from Informed Consent policy” for compromising patients’ exercise of autonomy, and concern for overrepresenting African Americans in the U.S. EFIC trials.
Racial disparities in medical treatment and unconscious biases among providers are among the reasons for mistrust and lack of trial participation by minority populations today. Francis Collins, director of the National Institutes of Health, said that recent social upheaval sparked by the death of George Floyd has likely added to feelings of mistrust between minority groups and government or pharmaceutical companies. “Yet we need their participation if this is going to have a meaningful outcome,” he said.
While “Operation Warp Speed” is committed to developing and delivering a COVID-19 vaccine rapidly while adhering to safety and efficacy standards, the challenges to enrolling people from racial and ethnic minorities in trials have been a concern. The political partisanship and ever-shifting stances on widespread COVID-19 testing, use of facemasks, endorsement of unproven drugs for the disease, and accusations against the FDA for delaying human trials for the vaccine have contributed to the skepticism as well. Tremendous pressure for a rushed vaccine with unrealistic timelines, recent holds on AZD1222 and JNJ-78436735 as well as the AZD1222 dosage error during trials have also raised skepticism of the safety and efficacy of vaccine trials.
2. Equitable allocation and distribution of vaccine for minority populations
Enrollment in clinical trials is just a beginning; a more significant challenge would be the vaccine’s uptake when available to the general public. We still lack a consensus on whether it is lawful for race to be an explicit criterion for priority distribution of the COVID-19 vaccine. Recently the Centers for Disease Control and Prevention suggested that the vaccine amount allotted to jurisdictions might be based on critical populations recommended for vaccination by the Advisory Committee on Immunization Practices with input from the National Academies of Sciences, Engineering, and Medicine.
The NASEM framework lays out four-phased vaccine distribution approaches, emphasizing social equity by prioritizing vaccines for geographic areas identified through CDC’s social vulnerability index (SVI) or another more specific index. SVI has been a robust composite marker of minority status and language, household composition and transportation, and housing and disability, and predicted COVID-19 case counts in the United States in several studies. The National Academy of Medicine has also recommended racial minorities receive priority vaccination because they have been hard hit and are “worse off” socioeconomically.
3. Immunization uptake by minority populations
Though minority participation is crucial in developing the vaccine, more transparency, open discussions on ethical distribution, and awareness of side effects are required before vaccine approval or emergency-use authorization. Companies behind the four major COVID-19 vaccines in development have released their trials’ protocols, details on vaccine efficacy, and each product’s makeup to increase acceptance of the vaccine.
According to a recent Pew research study, about half of U.S. adults (51%) now say they would definitely or probably get a vaccine to prevent COVID-19 if it were available today. Nearly as many (49%) say they definitely or probably would not get vaccinated at this time. Intent to get a COVID-19 vaccine has fallen from 72% in May 2020, a 21–percentage point drop, and Black adults were much less likely to say they would get a vaccine than other Americans.3 This is concerning as previous studies have shown that race and ethnicity can influence immune responses to vaccination. There is evidence of racial and ethnic differences in immune response following rubella vaccination, Hib–tetanus toxoid conjugate vaccine, antibody responses to the influenza A virus components of IIV3 or 4, and immune responses after measles vaccination.4-9
On the other hand, significant differences in reporting rates of adverse events after human papillomavirus vaccinations were found in different race and ethnicity groups in the Vaccine Adverse Event Reporting System.10 Thus, there is ample evidence that race and ethnicity affect responsiveness to a vaccine. Inequity in participation in a clinical trial may lead to an ineffective or one with a suboptimal response or even an unsafe vaccine.
When we look at other immunization programs, according to various surveys in recent years, non-Hispanic Blacks have lower annual vaccination rates for flu, pneumonia, and human papillomavirus vaccinations nationally, compared with non-Hispanic White adults.11 It is a cause of concern as a proportion of the population must be vaccinated to reach “community immunity” or “herd immunity” from vaccination. Depending on varying biological, environmental, and sociobehavioral factors, the threshold for COVID-19 herd immunity may be between 55% and 82% of the population.12 Hence, neither a vaccine trial nor an immunization program can succeed without participation from all communities and age groups.
Role of hospitalists
Hospitalists, who give immunizations as part of the hospital inpatient quality reporting program, are uniquely placed in this pandemic. Working on the front lines, we may encounter questions, concerns, rejections, and discussions about the pros and cons of the COVID-19 vaccine from patients.
Investigators at Children’s National Hospital and George Washington University, both in Washington, recently recommended three steps physicians and others can take now to ensure more people get the COVID-19 vaccine when it is available. Engaging frontline health professionals was one of the suggested steps to encourage more people to get the vaccine.13 However, it is imperative to understand that vaccine hesitancy might be an issue for health care providers as well, if concerns for scientific standards and involvement of diverse populations are not addressed.
We are only starting to develop a safe and effective immunization program. We must bring more to unrepresented communities than just vaccine trials. Information, education, availability, and access to the vaccines will make for a successful COVID-19 immunization program.
Dr. Saigal is a hospitalist and clinical assistant professor of medicine in the division of hospital medicine at the Ohio State University Wexner Medical Center, Columbus.
References
1. Moderna. COVE study. 2020 Oct 21. https://www.modernatx.com/sites/default/files/content_documents/2020-COVE-Study-Enrollment-Completion-10.22.20.pdf
2. U.S. Census Bureau. Quick facts: Population estimates, July 1, 2019. https://www.census.gov/quickfacts/fact/table/US/PST045219
3. Pew Research Center. U.S. Public Now Divided Over Whether To Get COVID-19 Vaccine. 2020 Sep 17. https://www.pewresearch.org/science/2020/09/17/u-s-public-now-divided-over-whether-to-get-covid-19-vaccine/
4. Haralambieva IH et al. Associations between race sex and immune response variations to rubella vaccination in two independent cohorts. Vaccine. 2014;32:1946-53.
5. McQuillan GM et al. Seroprevalence of measles antibody in the U.S. population 1999-2004. J Infect Dis. 2007;196:1459–64. doi: 10.1086/522866.
6. Christy C et al. Effect of gender race and parental education on immunogenicity and reported reactogenicity of acellular and whole-cell pertussis vaccines. Pediatrics. 1995;96:584-7.
7. Poland GA et al. Measles antibody seroprevalence rates among immunized Inuit Innu and Caucasian subjects. Vaccine. 1999;17:1525-31.
8. Greenberg DP et al. Immunogenicity of Haemophilus influenzae type b tetanus toxoid conjugate vaccine in young infants. The Kaiser-UCLA Vaccine Study Group. J Infect Dis. 1994;170:76-81.
9. Kurupati R et al. Race-related differences in antibody responses to the inactivated influenza vaccine are linked to distinct prevaccination gene expression profiles in blood. Oncotarget. 2016;7(39):62898-911.
10. Huang J et al. Characterization of the differential adverse event rates by race/ethnicity groups for HPV vaccine by integrating data from different sources. Front Pharmacol. 2018;9:539.
11. https://minorityhealth.hhs.gov/omh/browse.aspx?lvl=4&lvlid=22
12. Sanche S et al. High contagiousness and rapid spread of severe acute respiratory syndrome coronavirus 2. Emerg Infect Dis. 2020;26(7).
13. American Medical Association. How to ready patients now so they’ll get a COVID-19 vaccine later. 2020 May 27. https://www.ama-assn.org/delivering-care/public-health/how-ready-patients-now-so-they-ll-get-covid-19-vaccine-later
U.S. passes 1.3 million COVID-19 cases in children
The news on children and COVID-19 for Thanksgiving week does not provide a lot of room for thankfulness.
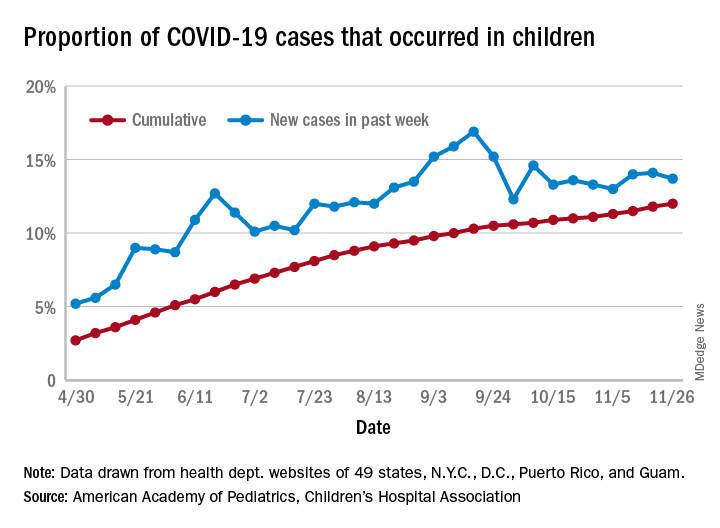
the American Academy of Pediatrics and the Children’s Hospital Association said in their latest weekly report.
For those not counting, the week ending Nov. 26 was the fifth in a row to show “the highest weekly increase since the pandemic began,” based on data the AAP and CHA have been collecting from 49 state health departments (New York does not report ages), as well as the District of Columbia, New York City, Puerto Rico, and Guam.
The 153,608 new cases bring the total number of COVID-19 cases in children to almost 1.34 million in those jurisdictions, which is 12% of the total number of cases (11.2 million) among all ages. For just the week ending Nov. 26, children represented 13.7% of all new cases in the United States, down from 14.1% the previous week, according to the AAP/CHA data.
Among the states reporting child cases, Florida has the lowest cumulative proportion of child cases, 6.4%, but the state is using an age range of 0-14 years (no other state goes lower than 17 years). New Jersey and Texas are next at 6.9%, although Texas “reported age for only 6% of total confirmed cases,” the AAP and CHA noted.
There are 35 states above the national number of 12.0%, the highest being Wyoming at 23.3%, followed by Tennessee at 18.3% and South Carolina at 18.2%. The two southern states are the only ones to use an age range of 0-20 years for child cases, the two groups said in this week’s report, which did not include the usual data on testing, hospitalization, and mortality because of the holiday.
The news on children and COVID-19 for Thanksgiving week does not provide a lot of room for thankfulness.

the American Academy of Pediatrics and the Children’s Hospital Association said in their latest weekly report.
For those not counting, the week ending Nov. 26 was the fifth in a row to show “the highest weekly increase since the pandemic began,” based on data the AAP and CHA have been collecting from 49 state health departments (New York does not report ages), as well as the District of Columbia, New York City, Puerto Rico, and Guam.
The 153,608 new cases bring the total number of COVID-19 cases in children to almost 1.34 million in those jurisdictions, which is 12% of the total number of cases (11.2 million) among all ages. For just the week ending Nov. 26, children represented 13.7% of all new cases in the United States, down from 14.1% the previous week, according to the AAP/CHA data.
Among the states reporting child cases, Florida has the lowest cumulative proportion of child cases, 6.4%, but the state is using an age range of 0-14 years (no other state goes lower than 17 years). New Jersey and Texas are next at 6.9%, although Texas “reported age for only 6% of total confirmed cases,” the AAP and CHA noted.
There are 35 states above the national number of 12.0%, the highest being Wyoming at 23.3%, followed by Tennessee at 18.3% and South Carolina at 18.2%. The two southern states are the only ones to use an age range of 0-20 years for child cases, the two groups said in this week’s report, which did not include the usual data on testing, hospitalization, and mortality because of the holiday.
The news on children and COVID-19 for Thanksgiving week does not provide a lot of room for thankfulness.

the American Academy of Pediatrics and the Children’s Hospital Association said in their latest weekly report.
For those not counting, the week ending Nov. 26 was the fifth in a row to show “the highest weekly increase since the pandemic began,” based on data the AAP and CHA have been collecting from 49 state health departments (New York does not report ages), as well as the District of Columbia, New York City, Puerto Rico, and Guam.
The 153,608 new cases bring the total number of COVID-19 cases in children to almost 1.34 million in those jurisdictions, which is 12% of the total number of cases (11.2 million) among all ages. For just the week ending Nov. 26, children represented 13.7% of all new cases in the United States, down from 14.1% the previous week, according to the AAP/CHA data.
Among the states reporting child cases, Florida has the lowest cumulative proportion of child cases, 6.4%, but the state is using an age range of 0-14 years (no other state goes lower than 17 years). New Jersey and Texas are next at 6.9%, although Texas “reported age for only 6% of total confirmed cases,” the AAP and CHA noted.
There are 35 states above the national number of 12.0%, the highest being Wyoming at 23.3%, followed by Tennessee at 18.3% and South Carolina at 18.2%. The two southern states are the only ones to use an age range of 0-20 years for child cases, the two groups said in this week’s report, which did not include the usual data on testing, hospitalization, and mortality because of the holiday.
ACIP: Health workers, long-term care residents first tier for COVID-19 vaccine
The Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices voted 13-1 that both groups be in the highest-priority group for vaccination. As such, ACIP recommends that both be included in phase 1a of the committee’s allocation plan.
The recommendation now goes to CDC director Robert Redfield, MD, for approval. State health departments are expected to rely on the recommendation, but ultimately can make their own decisions on how to allocate vaccine in their states.
“We hope that this vote gets us all one step closer to the day when we can all feel safe again and when this pandemic is over,” said Nancy Messonnier, MD, director of the CDC’s National Center for Immunization and Respiratory Diseases, at today’s meeting.
Health care workers are defined as paid and unpaid individuals serving in health care settings who have the potential for direct or indirect exposure to patients or infectious materials. Long-term care residents are defined as adults who reside in facilities that provide a variety of services, including medical and personal care. Phase 1a would not include children who live in such facilities.
“Our goal in phase 1a with regard to health care personnel is to preserve the workforce and health care capacity regardless of where exposure occurs,” said ACIP panelist Grace Lee, MD, MPH, professor of paediatrics at Stanford (Calif.) University. Thus vaccination would cover clinical support staff, such as nursing assistants, environmental services staff, and food support staff.
“It is crucial to maintain our health care capacity,” said ACIP member Sharon Frey, MD, clinical director at the Center for Vaccine Development at Saint Louis University. “But it’s also important to prevent severe disease and death in the group that is at highest risk of those complications and that includes those in long-term care facilities.”
CDC staff said that staff and residents in those facilities account for 6% of COVID-19 cases and 40% of deaths.
But Helen Keipp Talbot, MD, associate professor of medicine at Vanderbilt University, Nashville, Tenn., voted against putting long-term care residents into the 1a phase. “We have traditionally tried a vaccine in a young healthy population and then hope it works in our frail older adults. So we enter this realm of ‘we hope it works and that it’s safe,’ and that concerns me on many levels particularly for this vaccine,” she said, noting that the vaccines closest to FDA authorization have not been studied in elderly adults who live in nursing homes or assisted living facilities.
She added: “I have no reservations for health care workers taking this vaccine.”
Prioritization could change
The phase 1a allocation fits within the “four ethical principles” outlined by ACIP and CDC staff Nov. 23: to maximize benefits and minimize harms, promote justice, mitigate health inequities, and promote transparency.
“My vote reflects maximum benefit, minimum harm, promoting justice and mitigating the health inequalities that exist with regard to distribution of this vaccine,” said ACIP Chair Jose Romero, MD. Romero, chief medical officer of the Arkansas Department of Health, voted in favor of the phase 1a plan.
He and other panelists noted, however, that allocation priorities could change after the FDA reviews and authorizes a vaccine.
The FDA’s Vaccines and Related Biological Products Advisory Committee (VRBPAC) will meet December 10 to review the Pfizer/BioNTech’s messenger RNA-based vaccine (BNT162b2). The companies filed for emergency use on November 20.
A second vaccine, made by Moderna, is not far behind. The company reported on Nov. 30 that its messenger RNA vaccine was 94.1% effective and filed for emergency use the same day. The FDA’s VRBPAC will review the safety and efficacy data for the Moderna vaccine on Dec. 17.
“If individual vaccines receive emergency use authorization, we will have more data to consider, and that could lead to revision of our prioritization,” said ACIP member Robert Atmar, MD, John S. Dunn Research Foundation Clinical Professor in Infectious Diseases at Baylor College of Medicine, Houston.
ACIP will meet again after the Dec. 10 FDA advisory panel. But it won’t recommend a product until after the FDA has authorized it, said Amanda Cohn, MD, senior advisor for vaccines at the CDC’s National Center for Immunization and Respiratory Diseases.
Staggered immunization subprioritization urged
The CDC staff said that given the potential that not enough vaccine will be available immediately, it was recommending that health care organizations plan on creating a hierarchy of prioritization within institutions. And, they also urged staggering vaccination for personnel in similar units or positions, citing potential systemic or other reactions among health care workers.
“Consider planning for personnel to have time away from clinical care if health care personnel experience systemic symptoms post vaccination,” said Sarah Oliver, MD, MSPH, from the CDC.
The CDC will soon be issuing guidance on how to handle systemic symptoms with health care workers, Dr. Oliver noted.
Some 40 million doses of the Pfizer/BioNTech and Moderna vaccines are expected to be available by the end of December, with 5 million to 10 million a week coming online after that, Dr. Cohn said. That means not all health care workers will be vaccinated immediately. That may require “subprioritization, but for a limited period of time,” she said.
Dr. Messonnier said that, even with limited supplies, most of the states have told the CDC that they think they can vaccinate all of their health care workers within 3 weeks – some in less time.
The ACIP allocation plan is similar to but not exactly the same as that issued by the National Academy of Sciences, Engineering, and Medicine, which issued recommendations in October. That organization said that health care workers, first responders, older Americans living in congregate settings, and people with underlying health conditions should be the first to receive a vaccine.
ACIP has said that phase 1b would include essential workers, including police officers and firefighters, and those in education, transportation, and food and agriculture sectors. Phase 1c would include adults with high-risk medical conditions and those aged 65 years or older.
This article first appeared on Medscape.com.
The Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices voted 13-1 that both groups be in the highest-priority group for vaccination. As such, ACIP recommends that both be included in phase 1a of the committee’s allocation plan.
The recommendation now goes to CDC director Robert Redfield, MD, for approval. State health departments are expected to rely on the recommendation, but ultimately can make their own decisions on how to allocate vaccine in their states.
“We hope that this vote gets us all one step closer to the day when we can all feel safe again and when this pandemic is over,” said Nancy Messonnier, MD, director of the CDC’s National Center for Immunization and Respiratory Diseases, at today’s meeting.
Health care workers are defined as paid and unpaid individuals serving in health care settings who have the potential for direct or indirect exposure to patients or infectious materials. Long-term care residents are defined as adults who reside in facilities that provide a variety of services, including medical and personal care. Phase 1a would not include children who live in such facilities.
“Our goal in phase 1a with regard to health care personnel is to preserve the workforce and health care capacity regardless of where exposure occurs,” said ACIP panelist Grace Lee, MD, MPH, professor of paediatrics at Stanford (Calif.) University. Thus vaccination would cover clinical support staff, such as nursing assistants, environmental services staff, and food support staff.
“It is crucial to maintain our health care capacity,” said ACIP member Sharon Frey, MD, clinical director at the Center for Vaccine Development at Saint Louis University. “But it’s also important to prevent severe disease and death in the group that is at highest risk of those complications and that includes those in long-term care facilities.”
CDC staff said that staff and residents in those facilities account for 6% of COVID-19 cases and 40% of deaths.
But Helen Keipp Talbot, MD, associate professor of medicine at Vanderbilt University, Nashville, Tenn., voted against putting long-term care residents into the 1a phase. “We have traditionally tried a vaccine in a young healthy population and then hope it works in our frail older adults. So we enter this realm of ‘we hope it works and that it’s safe,’ and that concerns me on many levels particularly for this vaccine,” she said, noting that the vaccines closest to FDA authorization have not been studied in elderly adults who live in nursing homes or assisted living facilities.
She added: “I have no reservations for health care workers taking this vaccine.”
Prioritization could change
The phase 1a allocation fits within the “four ethical principles” outlined by ACIP and CDC staff Nov. 23: to maximize benefits and minimize harms, promote justice, mitigate health inequities, and promote transparency.
“My vote reflects maximum benefit, minimum harm, promoting justice and mitigating the health inequalities that exist with regard to distribution of this vaccine,” said ACIP Chair Jose Romero, MD. Romero, chief medical officer of the Arkansas Department of Health, voted in favor of the phase 1a plan.
He and other panelists noted, however, that allocation priorities could change after the FDA reviews and authorizes a vaccine.
The FDA’s Vaccines and Related Biological Products Advisory Committee (VRBPAC) will meet December 10 to review the Pfizer/BioNTech’s messenger RNA-based vaccine (BNT162b2). The companies filed for emergency use on November 20.
A second vaccine, made by Moderna, is not far behind. The company reported on Nov. 30 that its messenger RNA vaccine was 94.1% effective and filed for emergency use the same day. The FDA’s VRBPAC will review the safety and efficacy data for the Moderna vaccine on Dec. 17.
“If individual vaccines receive emergency use authorization, we will have more data to consider, and that could lead to revision of our prioritization,” said ACIP member Robert Atmar, MD, John S. Dunn Research Foundation Clinical Professor in Infectious Diseases at Baylor College of Medicine, Houston.
ACIP will meet again after the Dec. 10 FDA advisory panel. But it won’t recommend a product until after the FDA has authorized it, said Amanda Cohn, MD, senior advisor for vaccines at the CDC’s National Center for Immunization and Respiratory Diseases.
Staggered immunization subprioritization urged
The CDC staff said that given the potential that not enough vaccine will be available immediately, it was recommending that health care organizations plan on creating a hierarchy of prioritization within institutions. And, they also urged staggering vaccination for personnel in similar units or positions, citing potential systemic or other reactions among health care workers.
“Consider planning for personnel to have time away from clinical care if health care personnel experience systemic symptoms post vaccination,” said Sarah Oliver, MD, MSPH, from the CDC.
The CDC will soon be issuing guidance on how to handle systemic symptoms with health care workers, Dr. Oliver noted.
Some 40 million doses of the Pfizer/BioNTech and Moderna vaccines are expected to be available by the end of December, with 5 million to 10 million a week coming online after that, Dr. Cohn said. That means not all health care workers will be vaccinated immediately. That may require “subprioritization, but for a limited period of time,” she said.
Dr. Messonnier said that, even with limited supplies, most of the states have told the CDC that they think they can vaccinate all of their health care workers within 3 weeks – some in less time.
The ACIP allocation plan is similar to but not exactly the same as that issued by the National Academy of Sciences, Engineering, and Medicine, which issued recommendations in October. That organization said that health care workers, first responders, older Americans living in congregate settings, and people with underlying health conditions should be the first to receive a vaccine.
ACIP has said that phase 1b would include essential workers, including police officers and firefighters, and those in education, transportation, and food and agriculture sectors. Phase 1c would include adults with high-risk medical conditions and those aged 65 years or older.
This article first appeared on Medscape.com.
The Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices voted 13-1 that both groups be in the highest-priority group for vaccination. As such, ACIP recommends that both be included in phase 1a of the committee’s allocation plan.
The recommendation now goes to CDC director Robert Redfield, MD, for approval. State health departments are expected to rely on the recommendation, but ultimately can make their own decisions on how to allocate vaccine in their states.
“We hope that this vote gets us all one step closer to the day when we can all feel safe again and when this pandemic is over,” said Nancy Messonnier, MD, director of the CDC’s National Center for Immunization and Respiratory Diseases, at today’s meeting.
Health care workers are defined as paid and unpaid individuals serving in health care settings who have the potential for direct or indirect exposure to patients or infectious materials. Long-term care residents are defined as adults who reside in facilities that provide a variety of services, including medical and personal care. Phase 1a would not include children who live in such facilities.
“Our goal in phase 1a with regard to health care personnel is to preserve the workforce and health care capacity regardless of where exposure occurs,” said ACIP panelist Grace Lee, MD, MPH, professor of paediatrics at Stanford (Calif.) University. Thus vaccination would cover clinical support staff, such as nursing assistants, environmental services staff, and food support staff.
“It is crucial to maintain our health care capacity,” said ACIP member Sharon Frey, MD, clinical director at the Center for Vaccine Development at Saint Louis University. “But it’s also important to prevent severe disease and death in the group that is at highest risk of those complications and that includes those in long-term care facilities.”
CDC staff said that staff and residents in those facilities account for 6% of COVID-19 cases and 40% of deaths.
But Helen Keipp Talbot, MD, associate professor of medicine at Vanderbilt University, Nashville, Tenn., voted against putting long-term care residents into the 1a phase. “We have traditionally tried a vaccine in a young healthy population and then hope it works in our frail older adults. So we enter this realm of ‘we hope it works and that it’s safe,’ and that concerns me on many levels particularly for this vaccine,” she said, noting that the vaccines closest to FDA authorization have not been studied in elderly adults who live in nursing homes or assisted living facilities.
She added: “I have no reservations for health care workers taking this vaccine.”
Prioritization could change
The phase 1a allocation fits within the “four ethical principles” outlined by ACIP and CDC staff Nov. 23: to maximize benefits and minimize harms, promote justice, mitigate health inequities, and promote transparency.
“My vote reflects maximum benefit, minimum harm, promoting justice and mitigating the health inequalities that exist with regard to distribution of this vaccine,” said ACIP Chair Jose Romero, MD. Romero, chief medical officer of the Arkansas Department of Health, voted in favor of the phase 1a plan.
He and other panelists noted, however, that allocation priorities could change after the FDA reviews and authorizes a vaccine.
The FDA’s Vaccines and Related Biological Products Advisory Committee (VRBPAC) will meet December 10 to review the Pfizer/BioNTech’s messenger RNA-based vaccine (BNT162b2). The companies filed for emergency use on November 20.
A second vaccine, made by Moderna, is not far behind. The company reported on Nov. 30 that its messenger RNA vaccine was 94.1% effective and filed for emergency use the same day. The FDA’s VRBPAC will review the safety and efficacy data for the Moderna vaccine on Dec. 17.
“If individual vaccines receive emergency use authorization, we will have more data to consider, and that could lead to revision of our prioritization,” said ACIP member Robert Atmar, MD, John S. Dunn Research Foundation Clinical Professor in Infectious Diseases at Baylor College of Medicine, Houston.
ACIP will meet again after the Dec. 10 FDA advisory panel. But it won’t recommend a product until after the FDA has authorized it, said Amanda Cohn, MD, senior advisor for vaccines at the CDC’s National Center for Immunization and Respiratory Diseases.
Staggered immunization subprioritization urged
The CDC staff said that given the potential that not enough vaccine will be available immediately, it was recommending that health care organizations plan on creating a hierarchy of prioritization within institutions. And, they also urged staggering vaccination for personnel in similar units or positions, citing potential systemic or other reactions among health care workers.
“Consider planning for personnel to have time away from clinical care if health care personnel experience systemic symptoms post vaccination,” said Sarah Oliver, MD, MSPH, from the CDC.
The CDC will soon be issuing guidance on how to handle systemic symptoms with health care workers, Dr. Oliver noted.
Some 40 million doses of the Pfizer/BioNTech and Moderna vaccines are expected to be available by the end of December, with 5 million to 10 million a week coming online after that, Dr. Cohn said. That means not all health care workers will be vaccinated immediately. That may require “subprioritization, but for a limited period of time,” she said.
Dr. Messonnier said that, even with limited supplies, most of the states have told the CDC that they think they can vaccinate all of their health care workers within 3 weeks – some in less time.
The ACIP allocation plan is similar to but not exactly the same as that issued by the National Academy of Sciences, Engineering, and Medicine, which issued recommendations in October. That organization said that health care workers, first responders, older Americans living in congregate settings, and people with underlying health conditions should be the first to receive a vaccine.
ACIP has said that phase 1b would include essential workers, including police officers and firefighters, and those in education, transportation, and food and agriculture sectors. Phase 1c would include adults with high-risk medical conditions and those aged 65 years or older.
This article first appeared on Medscape.com.
CMS launches hospital-at-home program to free up hospital capacity
As an increasing number of health systems implement “hospital-at-home” (HaH) programs to increase their traditional hospital capacity, the Centers for Medicare & Medicaid Services has given the movement a boost by changing its regulations to allow acute care to be provided in a patient’s home under certain conditions.
The CMS announced Nov. 25 that it was launching its Acute Hospital Care at Home program “to increase the capacity of the American health care system” during the COVID-19 pandemic.
At the same time, the agency announced it was giving more flexibility to ambulatory surgery centers (ASCs) to provide hospital-level care.
The CMS said its new HaH program is an expansion of the Hospitals Without Walls initiative that was unveiled last March. Hospitals Without Walls is a set of “temporary new rules” that provide flexibility for hospitals to provide acute care outside of inpatient settings. Under those rules, hospitals are able to transfer patients to outside facilities, such as ASCs, inpatient rehabilitation hospitals, hotels, and dormitories, while still receiving Medicare hospital payments.
Under CMS’ new Acute Hospital Care at Home, which is not described as temporary, patients can be transferred from emergency departments or inpatient wards to hospital-level care at home. The CMS said the HaH program is designed for people with conditions such as the acute phases of asthma, heart failure, pneumonia, and chronic obstructive pulmonary disease. Altogether, the agency said, more than 60 acute conditions can be treated safely at home.
However, the agency didn’t say that facilities can’t admit COVID-19 patients to the hospital at home. Rami Karjian, MBA, cofounder and CEO of Medically Home, a firm that supplies health systems with technical services and software for HaH programs, said in an interview that several Medically Home clients plan to treat both COVID-19 and non-COVID-19 patients at home when they begin to participate in the CMS program in the near future.
The CMS said it consulted extensively with academic and private industry leaders in building its HaH program. Before rolling out the initiative, the agency noted, it conducted successful pilot programs in leading hospitals and health systems. The results of some of these pilots have been reported in academic journals.
Participating hospitals will be required to have specified screening protocols in place before beginning acute care at home, the CMS announced. An in-person physician evaluation will be required before starting care at home. A nurse will evaluate each patient once daily in person or remotely, and either nurses or paramedics will visit the patient in person twice a day.
In contrast, Medicare regulations require nursing staff to be available around the clock in traditional hospitals. So the CMS has to grant waivers to hospitals for HaH programs.
While not going into detail on the telemonitoring capabilities that will be required in the acute hospital care at home, the release said, “Today’s announcement builds upon the critical work by CMS to expand telehealth coverage to keep beneficiaries safe and prevent the spread of COVID-19.”
More flexibility for ASCs
The agency is also giving ASCs the flexibility to provide 24-hour nursing services only when one or more patients are receiving care on site. This flexibility will be available to any of the 5,700 ASCs that wish to participate, and will be immediately effective for the 85 ASCs currently participating in the Hospital Without Walls initiative, the CMS said.
The new ASC regulations, the CMS said, are aimed at allowing communities “to maintain surgical capacity and other life-saving non-COVID-19 [care], like cancer surgeries.” Patients who need such procedures will be able to receive them in ASCs without being exposed to known COVID-19 cases.
Similarly, the CMS said patients and families not diagnosed with COVID-19 may prefer to receive acute care at home if local hospitals are full of COVID-19 patients. In addition, the CMS said it anticipates patients may value the ability to be treated at home without the visitation restrictions of hospitals.
Early HaH participants
Six health systems with extensive experience in providing acute hospital care at home have been approved for the new HaH waivers from Medicare rules. They include Brigham and Women’s Hospital (Massachusetts); Huntsman Cancer Institute (Utah); Massachusetts General Hospital (Massachusetts); Mount Sinai Health System (New York City); Presbyterian Healthcare Services (New Mexico); and UnityPoint Health (Iowa).
The CMS said that it’s in discussions with other health care systems and expects new applications to be submitted soon.
To support these efforts, the CMS has launched an online portal to streamline the waiver request process. The agency said it will closely monitor the program to safeguard beneficiaries and will require participating hospitals to report quality and safety data on a regular basis.
Support from hospitals
The first health systems participating in the CMS HaH appear to be supportive of the program, with some hospital leaders submitting comments to the CMS about their view of the initiative.
“The CMS has taken an extraordinary step today, facilitating the rapid expansion of Hospitalization at Home, an innovative care model with proven results,” said Kenneth L. Davis, MD, president and CEO of the Mount Sinai Health System in New York City. “This important and timely move will enable hospitals across the country to use effective tools to safely care for patients during this pandemic.”
David Levine, MD, assistant professor of medicine and medical director of strategy and innovation for Brigham Health Home Hospital in Boston, was similarly laudatory: “Our research at Brigham Health Home has shown that we can deliver hospital-level care in our patients’ homes with lower readmission rates, more physical mobility, and a positive patient experience,” he said. “During these challenging times, a focus on the home is critical. We are so encouraged that CMS is taking this important step, which will allow hospitals across the country to increase their capacity while delivering the care all patients deserve.”
Scaling up quickly
If other hospitals and health systems recognize the value of HaH, how long might it take them to develop and implement these programs in the midst of a pandemic?
Atrium Health, a large health system in the Southeast, ramped up a hospital-at-home initiative last spring for its 10 hospitals in the Charlotte, N.C., area, in just 2 weeks. However, it had been working on the project for some time before the pandemic struck. Focusing mostly on COVID-19 patients, the initiative reduced the COVID-19 patient load by 20%-25% in Atrium’s hospitals.
Medically Home, the HaH infrastructure company, said in a news release that it “enables health systems to establish new hospital-at-home services in as little as 30 days.” Medically Home has partnered in this venture with Huron Consulting Group, which has about 200 HaH-trained consultants, and Cardinal Health, a large global medical supplies distributor.
Mr. Karjian said in an interview that he expects private insurers to follow CMS’ example, as they often do. “We think this decision will cause not only CMS but private insurers to cover hospital at home after the pandemic, if it becomes the standard of care, because patients have better outcomes when treated at home,” he said.
Asked for his view on why the CMS specified that patients could be admitted to an HaH only from emergency departments or inpatient settings, Mr. Karjian said that the CMS wants to make sure that patients have access to brick-and-mortar hospital care if that’s what they need. Also, he noted, this model is new to most hospitals, so the CMS wants to make sure it starts “with all the safety guardrails” in place.
Overall, Mr. Karjian said, “This is an exciting development for patients across the country. What CMS has done is terrific in terms of letting patients get the care they want, where they want it, and get the benefit of better outcomes while the nation is going through this capacity crunch for hospital beds.”
A version of this article originally appeared on Medscape.com.
As an increasing number of health systems implement “hospital-at-home” (HaH) programs to increase their traditional hospital capacity, the Centers for Medicare & Medicaid Services has given the movement a boost by changing its regulations to allow acute care to be provided in a patient’s home under certain conditions.
The CMS announced Nov. 25 that it was launching its Acute Hospital Care at Home program “to increase the capacity of the American health care system” during the COVID-19 pandemic.
At the same time, the agency announced it was giving more flexibility to ambulatory surgery centers (ASCs) to provide hospital-level care.
The CMS said its new HaH program is an expansion of the Hospitals Without Walls initiative that was unveiled last March. Hospitals Without Walls is a set of “temporary new rules” that provide flexibility for hospitals to provide acute care outside of inpatient settings. Under those rules, hospitals are able to transfer patients to outside facilities, such as ASCs, inpatient rehabilitation hospitals, hotels, and dormitories, while still receiving Medicare hospital payments.
Under CMS’ new Acute Hospital Care at Home, which is not described as temporary, patients can be transferred from emergency departments or inpatient wards to hospital-level care at home. The CMS said the HaH program is designed for people with conditions such as the acute phases of asthma, heart failure, pneumonia, and chronic obstructive pulmonary disease. Altogether, the agency said, more than 60 acute conditions can be treated safely at home.
However, the agency didn’t say that facilities can’t admit COVID-19 patients to the hospital at home. Rami Karjian, MBA, cofounder and CEO of Medically Home, a firm that supplies health systems with technical services and software for HaH programs, said in an interview that several Medically Home clients plan to treat both COVID-19 and non-COVID-19 patients at home when they begin to participate in the CMS program in the near future.
The CMS said it consulted extensively with academic and private industry leaders in building its HaH program. Before rolling out the initiative, the agency noted, it conducted successful pilot programs in leading hospitals and health systems. The results of some of these pilots have been reported in academic journals.
Participating hospitals will be required to have specified screening protocols in place before beginning acute care at home, the CMS announced. An in-person physician evaluation will be required before starting care at home. A nurse will evaluate each patient once daily in person or remotely, and either nurses or paramedics will visit the patient in person twice a day.
In contrast, Medicare regulations require nursing staff to be available around the clock in traditional hospitals. So the CMS has to grant waivers to hospitals for HaH programs.
While not going into detail on the telemonitoring capabilities that will be required in the acute hospital care at home, the release said, “Today’s announcement builds upon the critical work by CMS to expand telehealth coverage to keep beneficiaries safe and prevent the spread of COVID-19.”
More flexibility for ASCs
The agency is also giving ASCs the flexibility to provide 24-hour nursing services only when one or more patients are receiving care on site. This flexibility will be available to any of the 5,700 ASCs that wish to participate, and will be immediately effective for the 85 ASCs currently participating in the Hospital Without Walls initiative, the CMS said.
The new ASC regulations, the CMS said, are aimed at allowing communities “to maintain surgical capacity and other life-saving non-COVID-19 [care], like cancer surgeries.” Patients who need such procedures will be able to receive them in ASCs without being exposed to known COVID-19 cases.
Similarly, the CMS said patients and families not diagnosed with COVID-19 may prefer to receive acute care at home if local hospitals are full of COVID-19 patients. In addition, the CMS said it anticipates patients may value the ability to be treated at home without the visitation restrictions of hospitals.
Early HaH participants
Six health systems with extensive experience in providing acute hospital care at home have been approved for the new HaH waivers from Medicare rules. They include Brigham and Women’s Hospital (Massachusetts); Huntsman Cancer Institute (Utah); Massachusetts General Hospital (Massachusetts); Mount Sinai Health System (New York City); Presbyterian Healthcare Services (New Mexico); and UnityPoint Health (Iowa).
The CMS said that it’s in discussions with other health care systems and expects new applications to be submitted soon.
To support these efforts, the CMS has launched an online portal to streamline the waiver request process. The agency said it will closely monitor the program to safeguard beneficiaries and will require participating hospitals to report quality and safety data on a regular basis.
Support from hospitals
The first health systems participating in the CMS HaH appear to be supportive of the program, with some hospital leaders submitting comments to the CMS about their view of the initiative.
“The CMS has taken an extraordinary step today, facilitating the rapid expansion of Hospitalization at Home, an innovative care model with proven results,” said Kenneth L. Davis, MD, president and CEO of the Mount Sinai Health System in New York City. “This important and timely move will enable hospitals across the country to use effective tools to safely care for patients during this pandemic.”
David Levine, MD, assistant professor of medicine and medical director of strategy and innovation for Brigham Health Home Hospital in Boston, was similarly laudatory: “Our research at Brigham Health Home has shown that we can deliver hospital-level care in our patients’ homes with lower readmission rates, more physical mobility, and a positive patient experience,” he said. “During these challenging times, a focus on the home is critical. We are so encouraged that CMS is taking this important step, which will allow hospitals across the country to increase their capacity while delivering the care all patients deserve.”
Scaling up quickly
If other hospitals and health systems recognize the value of HaH, how long might it take them to develop and implement these programs in the midst of a pandemic?
Atrium Health, a large health system in the Southeast, ramped up a hospital-at-home initiative last spring for its 10 hospitals in the Charlotte, N.C., area, in just 2 weeks. However, it had been working on the project for some time before the pandemic struck. Focusing mostly on COVID-19 patients, the initiative reduced the COVID-19 patient load by 20%-25% in Atrium’s hospitals.
Medically Home, the HaH infrastructure company, said in a news release that it “enables health systems to establish new hospital-at-home services in as little as 30 days.” Medically Home has partnered in this venture with Huron Consulting Group, which has about 200 HaH-trained consultants, and Cardinal Health, a large global medical supplies distributor.
Mr. Karjian said in an interview that he expects private insurers to follow CMS’ example, as they often do. “We think this decision will cause not only CMS but private insurers to cover hospital at home after the pandemic, if it becomes the standard of care, because patients have better outcomes when treated at home,” he said.
Asked for his view on why the CMS specified that patients could be admitted to an HaH only from emergency departments or inpatient settings, Mr. Karjian said that the CMS wants to make sure that patients have access to brick-and-mortar hospital care if that’s what they need. Also, he noted, this model is new to most hospitals, so the CMS wants to make sure it starts “with all the safety guardrails” in place.
Overall, Mr. Karjian said, “This is an exciting development for patients across the country. What CMS has done is terrific in terms of letting patients get the care they want, where they want it, and get the benefit of better outcomes while the nation is going through this capacity crunch for hospital beds.”
A version of this article originally appeared on Medscape.com.
As an increasing number of health systems implement “hospital-at-home” (HaH) programs to increase their traditional hospital capacity, the Centers for Medicare & Medicaid Services has given the movement a boost by changing its regulations to allow acute care to be provided in a patient’s home under certain conditions.
The CMS announced Nov. 25 that it was launching its Acute Hospital Care at Home program “to increase the capacity of the American health care system” during the COVID-19 pandemic.
At the same time, the agency announced it was giving more flexibility to ambulatory surgery centers (ASCs) to provide hospital-level care.
The CMS said its new HaH program is an expansion of the Hospitals Without Walls initiative that was unveiled last March. Hospitals Without Walls is a set of “temporary new rules” that provide flexibility for hospitals to provide acute care outside of inpatient settings. Under those rules, hospitals are able to transfer patients to outside facilities, such as ASCs, inpatient rehabilitation hospitals, hotels, and dormitories, while still receiving Medicare hospital payments.
Under CMS’ new Acute Hospital Care at Home, which is not described as temporary, patients can be transferred from emergency departments or inpatient wards to hospital-level care at home. The CMS said the HaH program is designed for people with conditions such as the acute phases of asthma, heart failure, pneumonia, and chronic obstructive pulmonary disease. Altogether, the agency said, more than 60 acute conditions can be treated safely at home.
However, the agency didn’t say that facilities can’t admit COVID-19 patients to the hospital at home. Rami Karjian, MBA, cofounder and CEO of Medically Home, a firm that supplies health systems with technical services and software for HaH programs, said in an interview that several Medically Home clients plan to treat both COVID-19 and non-COVID-19 patients at home when they begin to participate in the CMS program in the near future.
The CMS said it consulted extensively with academic and private industry leaders in building its HaH program. Before rolling out the initiative, the agency noted, it conducted successful pilot programs in leading hospitals and health systems. The results of some of these pilots have been reported in academic journals.
Participating hospitals will be required to have specified screening protocols in place before beginning acute care at home, the CMS announced. An in-person physician evaluation will be required before starting care at home. A nurse will evaluate each patient once daily in person or remotely, and either nurses or paramedics will visit the patient in person twice a day.
In contrast, Medicare regulations require nursing staff to be available around the clock in traditional hospitals. So the CMS has to grant waivers to hospitals for HaH programs.
While not going into detail on the telemonitoring capabilities that will be required in the acute hospital care at home, the release said, “Today’s announcement builds upon the critical work by CMS to expand telehealth coverage to keep beneficiaries safe and prevent the spread of COVID-19.”
More flexibility for ASCs
The agency is also giving ASCs the flexibility to provide 24-hour nursing services only when one or more patients are receiving care on site. This flexibility will be available to any of the 5,700 ASCs that wish to participate, and will be immediately effective for the 85 ASCs currently participating in the Hospital Without Walls initiative, the CMS said.
The new ASC regulations, the CMS said, are aimed at allowing communities “to maintain surgical capacity and other life-saving non-COVID-19 [care], like cancer surgeries.” Patients who need such procedures will be able to receive them in ASCs without being exposed to known COVID-19 cases.
Similarly, the CMS said patients and families not diagnosed with COVID-19 may prefer to receive acute care at home if local hospitals are full of COVID-19 patients. In addition, the CMS said it anticipates patients may value the ability to be treated at home without the visitation restrictions of hospitals.
Early HaH participants
Six health systems with extensive experience in providing acute hospital care at home have been approved for the new HaH waivers from Medicare rules. They include Brigham and Women’s Hospital (Massachusetts); Huntsman Cancer Institute (Utah); Massachusetts General Hospital (Massachusetts); Mount Sinai Health System (New York City); Presbyterian Healthcare Services (New Mexico); and UnityPoint Health (Iowa).
The CMS said that it’s in discussions with other health care systems and expects new applications to be submitted soon.
To support these efforts, the CMS has launched an online portal to streamline the waiver request process. The agency said it will closely monitor the program to safeguard beneficiaries and will require participating hospitals to report quality and safety data on a regular basis.
Support from hospitals
The first health systems participating in the CMS HaH appear to be supportive of the program, with some hospital leaders submitting comments to the CMS about their view of the initiative.
“The CMS has taken an extraordinary step today, facilitating the rapid expansion of Hospitalization at Home, an innovative care model with proven results,” said Kenneth L. Davis, MD, president and CEO of the Mount Sinai Health System in New York City. “This important and timely move will enable hospitals across the country to use effective tools to safely care for patients during this pandemic.”
David Levine, MD, assistant professor of medicine and medical director of strategy and innovation for Brigham Health Home Hospital in Boston, was similarly laudatory: “Our research at Brigham Health Home has shown that we can deliver hospital-level care in our patients’ homes with lower readmission rates, more physical mobility, and a positive patient experience,” he said. “During these challenging times, a focus on the home is critical. We are so encouraged that CMS is taking this important step, which will allow hospitals across the country to increase their capacity while delivering the care all patients deserve.”
Scaling up quickly
If other hospitals and health systems recognize the value of HaH, how long might it take them to develop and implement these programs in the midst of a pandemic?
Atrium Health, a large health system in the Southeast, ramped up a hospital-at-home initiative last spring for its 10 hospitals in the Charlotte, N.C., area, in just 2 weeks. However, it had been working on the project for some time before the pandemic struck. Focusing mostly on COVID-19 patients, the initiative reduced the COVID-19 patient load by 20%-25% in Atrium’s hospitals.
Medically Home, the HaH infrastructure company, said in a news release that it “enables health systems to establish new hospital-at-home services in as little as 30 days.” Medically Home has partnered in this venture with Huron Consulting Group, which has about 200 HaH-trained consultants, and Cardinal Health, a large global medical supplies distributor.
Mr. Karjian said in an interview that he expects private insurers to follow CMS’ example, as they often do. “We think this decision will cause not only CMS but private insurers to cover hospital at home after the pandemic, if it becomes the standard of care, because patients have better outcomes when treated at home,” he said.
Asked for his view on why the CMS specified that patients could be admitted to an HaH only from emergency departments or inpatient settings, Mr. Karjian said that the CMS wants to make sure that patients have access to brick-and-mortar hospital care if that’s what they need. Also, he noted, this model is new to most hospitals, so the CMS wants to make sure it starts “with all the safety guardrails” in place.
Overall, Mr. Karjian said, “This is an exciting development for patients across the country. What CMS has done is terrific in terms of letting patients get the care they want, where they want it, and get the benefit of better outcomes while the nation is going through this capacity crunch for hospital beds.”
A version of this article originally appeared on Medscape.com.
Are more female physicians leaving medicine as pandemic surges?
For mid-career oncologist Tanya Wildes, MD, the pandemic was the last straw. In late September, she tweeted: “I have done the academically unfathomable: I am resigning my faculty position without another job lined up.”
She wasn’t burned out, she insisted. She loved her patients and her research. But she was also “100% confident” in her decision and “also 100% sad. This did not have to happen,” she lamented, asking not to disclose her workplace for fear of retribution.
Being a woman in medicine “is a hard life to start with,” Dr. Wildes said in an interview. “We all have that tenuous balance going on and the pandemic made everything just a little bit harder.”
She describes her prepandemic work-life balance as a “Jenga tower, with everything only just in place.” But she realized that the balance had tipped, when after a difficult clinic she felt emotionally wrung out. Her 11-year-old son had asked her to help him fly his model airplane. “I told him, ‘Honey, I can’t do it because if it crashes or gets stuck in a tree ... you’re going to be devastated and I have nothing left for you.’ “
This was a eureka moment, as “I realized, this is not who I want to be,” she said, holding back tears. “Seventy years from now my son is going to tell his grandchildren about the pandemic and I don’t want his memory of his mom to be that she couldn’t be there for him because she was too spent.”
When Dr. Wildes shared her story on Twitter, other female oncologists and physicians responded that they too have felt they’re under increased pressure this year, with the extra stress of the pandemic leading others to quit as well.
The trend of doctors leaving medicine has been noticeable. A July survey from the Physicians Foundation found that roughly 16,000 medical practices had already closed during the pandemic, with another 8,000 predicted to close within the next year.
“Similar patterns” were evident in another analysis by the Larry A. Green Center and the Primary Care Collaborative, as reported in The New York Times. In that survey, nearly one-fifth of primary care clinicians said “someone in their practice plans to retire early or has already retired because of COVID-19,” and 15% say “someone has left or plans to leave the practice.” About half said their mental exhaustion was at an all-time high, the survey found.
“COVID-19 is a burden, and that added burden has tipped people over the edge of many things,” said Monica Bertagnolli, MD, chief of the division of surgical oncology at Brigham and Women’s Hospital, Boston, and former president of the American Society of Clinical Oncology.
“It has illustrated that we do have a lot of people who are working kind of on the edge of not being able to handle everything,” she said.
While many in medicine are struggling, the pandemic seems to be pushing more women to leave, highlighting longtime gender disparities and increased caregiving burdens. And their absence may be felt for years to come.
Firm numbers are hard to come by, said Julie Silver, MD, associate professor, associate chair, and director of cancer rehabilitation in the department of physical medicine and rehabilitation at Harvard Medical School, Boston, and an expert in gender equity in medicine. But she sees some troubling trends.
“There are many indications that women are leaving medicine in disproportionately high numbers,” Dr. Silver said in an interview. “A lack of fair pay and promotion opportunities that were present before COVID-19 are now combined with a host of pandemic-related challenges.”
A survey of 1,809 women conducted in mid-April with the Physician Moms Facebook Group and accepted for online publication by the American Journal of Psychiatry found that 41% scored over the cutoff points for moderate or severe anxiety, with 46% meeting these criteria among front-line workers.
“It’s really important for society to recognize the extraordinary impact this pandemic is having on physician mothers, as there will be profound ripple effects on the ability of this key segment of the health care workforce to serve others if we do not address this problem urgently,” co-senior author Reshma Jagsi, MD, DPhil, a radiation oncologist at the University of Michigan, Ann Arbor, said in an interview.
Women weighed in on Twitter, in response to Dr. Silver’s tweet to #WomenInMedicine: “If you are thinking of leaving #medicine & need a reason to stay: we value you & need you.”
In reply, Emmy Betz, MD, MPH, associate professor of emergency medicine at the University of Colorado at Denver, Aurora, said via Twitter, “I’ve had lots of conversations with women considering leaving medicine.”
“I have thought about leaving many times. I love what I do, but medicine can be an unkind world at times,” responded Valerie Fitzhugh MD, associate professor and pathologist at New Jersey Medical School, Newark.
“Too late. Left at the end of July and it was the best decision ever,” wrote Michelle Gordon, DO, who was previously a board-certified general surgeon at Northern Westchester Surgical Associates in Putnam Valley, N.Y.
Prepandemic disparities accentuated
The pandemic “has merely accentuated – or made more apparent – some of the longstanding issues and struggles of women in oncology, women in medicine, women in academia,” said Sarah Holstein, MD, PhD, another mid-career oncologist and associate professor at the University of Nebraska Medical Center, Omaha.
“There are disparities in first-author/last-author publications, disparities in being asked to give speaking engagements, disparities in leadership,” Dr. Holstein said in an interview. “And then ... put on top of that the various surges with the pandemic where you are being asked to do clinical responsibilities you don’t normally do, perhaps some things you haven’t done since your training 10 or 20 years ago.”
This is backed up with data: There is already a “robust” body of prepandemic literature demonstrating pay gaps for female physicians and scientists, noted Dr. Silver, who founded the Her Time Is Now campaign for gender equity in medicine and runs a women’s leadership course at Harvard.
In addition, female physicians are more likely to be involved in “nonpromotable” work, group projects and educator roles that are often underappreciated and undercompensated, she said.
Writing recently in a blog post for the BMJ, Dr. Silver and colleagues predict that as a result of the pandemic, female physicians will “face disadvantages from unconscious bias in decisions about whose pay should be cut, whose operating schedules should take priority when resources are limited, and whose contributions merit retention ... The ground that women lose now will likely have a profound effect for many years to come, perhaps putting them at a disadvantage for the rest of their careers.”
There is already evidence of reduced publishing by female scientists during the pandemic, something that “could undermine the careers of an entire generation of women scholars,” noted Caitlyn Collins, PhD, assistant professor of sociology at Washington University in St. Louis.
“Science needs to address the culture of overwork,” Dr. Collins said in an interview. “Parents and other caregivers deserve support. The stress and ‘overwhelm’ they feel is not inevitable. A more fair, just, and humane approach to combining work and family is possible – what we need is the political will to pass better policies and a massive shift in our cultural understandings about how work should fit into family life, not the other way around.”
Lack of support for “vulnerable scientists,” particularly “junior scientists who are parents, women, or minorities” could lead to “severe attrition in cancer research in the coming years,” Cullen Taniguchi, MD, PhD, a radiation oncologist and associate professor at the University of Texas MD Anderson Cancer Center, Houston, and colleagues, warned in a recent letter to the journal Cancer Cell.
“The biggest worries of attrition will come from young faculty who started just before or after the pandemic,” Dr. Taniguchi said in an interview. “The first year in an academic setting is incredibly challenging but also important for establishing research efforts and building networks of colleagues to collaborate with. While completely necessary, the restrictions put in place during the pandemic made doing these things even more difficult.”
Another stressor: Caregiving at home
Another reason female physicians may be marginalized during the pandemic is that they are more often the primary caregivers at home.
“Anyone who is a caregiver, be it to kids, parents, or spouses, can relate to the challenges brought [on] by the pandemic,” said Ishwaria Subbiah, MD, a palliative care physician and medical oncologist at MD Anderson.
“Most of us work toward meeting our responsibilities by engaging a network of support, whether it’s home care workers, center-based or at-home child care, schools, or activities outside of school. The pandemic led to a level of disruption that brought most (if not all) of those responsibilities onto the caregivers themselves,” she said in an interview.
As the mother of an adult son with severe epilepsy, Dr. Bertagnolli has certainly experienced the challenges of parenting during the pandemic. “Our son is now 24 but he is handicapped, and lives with us. The care issues we have to deal with as professionals have been enormously magnified by COVID,” she said.
But she cautions against making gender distinctions when it comes to caregiving. “Has it fallen on the women? Well, this kind of stuff generally falls on the women, but I am certain it has fallen on an awful lot of men as well, because I think the world is changing that way, so it’s fallen on all of us.”
There is no question that female oncologists are bearing the brunt, both at work and at home, contended Dr. Taniguchi. “Absolutely. I have seen this first-hand,” he said.
“If it was difficult for women, underrepresented minorities, and junior faculty to find a voice in the room prepandemic, I think it can be harder in the times of virtual meetings when it is difficult to engage audiences,” he said.
Dr. Holstein said she is lucky to be well-supported at her institution, with both a female chief of hematology/oncology and a female chair of internal medicine, but still, she worries about the long-term consequences of the pandemic on the gender landscape of medicine.
“If you’re having to put aside research projects because you have extra responsibilities – again because women just tend to have a lot of other things going on – that might not be a big deal for 3 months, 6 months, but this is going to be a year or 2 years before ‘normal’ comes back,” she says. “One to two years of underpublishing or not getting the grants could be career killers for women in academic oncology.”
Cancer COVID-19 combo
As Dr. Wildes completed her final weeks of seeing cancer patients, she received an outpouring of support, which she says convinced her of the shared experience of all doctors, and especially female doctors, during the pandemic. But even more specifically, she feels that she has tapped into the unique burden shouldered by oncologists during this time.
“It’s intimidating being an oncologist; we are literally giving people poison for a living. Then throw into it a pandemic where early in March we had so little data. I was helping my patients make decisions about their cancer care based on a case series of four patients in China. The burden of those conversations is something I never want to have to live through again,” she said.
“Oncology is a particularly intense subspecialty within medicine,” agreed Dr. Subbiah. “The people we care for have received a life-altering and potentially life-limiting diagnosis. Coupled with that, the COVID-19 pandemic has brought an unprecedented cloud of uncertainty ... Whether the patients can see it overtly or not, oncologists carry the weight of this worry with them for not just one but all of their patients.”
Dr. Wildes said she plans to return to academic medicine and clinical care “in time,” but for now, the gap that she and others like her leave is troubling to those who have stayed on.
“We need these women in medicine,” said Dr. Holstein. “We have data suggesting that women take more time with their patients than men, that patient outcomes are better if they have a female physician. But also for the generations coming up, we need the mid-career and senior women to be in place to mentor and guide and make sure we continue to increase women in leadership.”
A version of this article originally appeared on Medscape.com.
For mid-career oncologist Tanya Wildes, MD, the pandemic was the last straw. In late September, she tweeted: “I have done the academically unfathomable: I am resigning my faculty position without another job lined up.”
She wasn’t burned out, she insisted. She loved her patients and her research. But she was also “100% confident” in her decision and “also 100% sad. This did not have to happen,” she lamented, asking not to disclose her workplace for fear of retribution.
Being a woman in medicine “is a hard life to start with,” Dr. Wildes said in an interview. “We all have that tenuous balance going on and the pandemic made everything just a little bit harder.”
She describes her prepandemic work-life balance as a “Jenga tower, with everything only just in place.” But she realized that the balance had tipped, when after a difficult clinic she felt emotionally wrung out. Her 11-year-old son had asked her to help him fly his model airplane. “I told him, ‘Honey, I can’t do it because if it crashes or gets stuck in a tree ... you’re going to be devastated and I have nothing left for you.’ “
This was a eureka moment, as “I realized, this is not who I want to be,” she said, holding back tears. “Seventy years from now my son is going to tell his grandchildren about the pandemic and I don’t want his memory of his mom to be that she couldn’t be there for him because she was too spent.”
When Dr. Wildes shared her story on Twitter, other female oncologists and physicians responded that they too have felt they’re under increased pressure this year, with the extra stress of the pandemic leading others to quit as well.
The trend of doctors leaving medicine has been noticeable. A July survey from the Physicians Foundation found that roughly 16,000 medical practices had already closed during the pandemic, with another 8,000 predicted to close within the next year.
“Similar patterns” were evident in another analysis by the Larry A. Green Center and the Primary Care Collaborative, as reported in The New York Times. In that survey, nearly one-fifth of primary care clinicians said “someone in their practice plans to retire early or has already retired because of COVID-19,” and 15% say “someone has left or plans to leave the practice.” About half said their mental exhaustion was at an all-time high, the survey found.
“COVID-19 is a burden, and that added burden has tipped people over the edge of many things,” said Monica Bertagnolli, MD, chief of the division of surgical oncology at Brigham and Women’s Hospital, Boston, and former president of the American Society of Clinical Oncology.
“It has illustrated that we do have a lot of people who are working kind of on the edge of not being able to handle everything,” she said.
While many in medicine are struggling, the pandemic seems to be pushing more women to leave, highlighting longtime gender disparities and increased caregiving burdens. And their absence may be felt for years to come.
Firm numbers are hard to come by, said Julie Silver, MD, associate professor, associate chair, and director of cancer rehabilitation in the department of physical medicine and rehabilitation at Harvard Medical School, Boston, and an expert in gender equity in medicine. But she sees some troubling trends.
“There are many indications that women are leaving medicine in disproportionately high numbers,” Dr. Silver said in an interview. “A lack of fair pay and promotion opportunities that were present before COVID-19 are now combined with a host of pandemic-related challenges.”
A survey of 1,809 women conducted in mid-April with the Physician Moms Facebook Group and accepted for online publication by the American Journal of Psychiatry found that 41% scored over the cutoff points for moderate or severe anxiety, with 46% meeting these criteria among front-line workers.
“It’s really important for society to recognize the extraordinary impact this pandemic is having on physician mothers, as there will be profound ripple effects on the ability of this key segment of the health care workforce to serve others if we do not address this problem urgently,” co-senior author Reshma Jagsi, MD, DPhil, a radiation oncologist at the University of Michigan, Ann Arbor, said in an interview.
Women weighed in on Twitter, in response to Dr. Silver’s tweet to #WomenInMedicine: “If you are thinking of leaving #medicine & need a reason to stay: we value you & need you.”
In reply, Emmy Betz, MD, MPH, associate professor of emergency medicine at the University of Colorado at Denver, Aurora, said via Twitter, “I’ve had lots of conversations with women considering leaving medicine.”
“I have thought about leaving many times. I love what I do, but medicine can be an unkind world at times,” responded Valerie Fitzhugh MD, associate professor and pathologist at New Jersey Medical School, Newark.
“Too late. Left at the end of July and it was the best decision ever,” wrote Michelle Gordon, DO, who was previously a board-certified general surgeon at Northern Westchester Surgical Associates in Putnam Valley, N.Y.
Prepandemic disparities accentuated
The pandemic “has merely accentuated – or made more apparent – some of the longstanding issues and struggles of women in oncology, women in medicine, women in academia,” said Sarah Holstein, MD, PhD, another mid-career oncologist and associate professor at the University of Nebraska Medical Center, Omaha.
“There are disparities in first-author/last-author publications, disparities in being asked to give speaking engagements, disparities in leadership,” Dr. Holstein said in an interview. “And then ... put on top of that the various surges with the pandemic where you are being asked to do clinical responsibilities you don’t normally do, perhaps some things you haven’t done since your training 10 or 20 years ago.”
This is backed up with data: There is already a “robust” body of prepandemic literature demonstrating pay gaps for female physicians and scientists, noted Dr. Silver, who founded the Her Time Is Now campaign for gender equity in medicine and runs a women’s leadership course at Harvard.
In addition, female physicians are more likely to be involved in “nonpromotable” work, group projects and educator roles that are often underappreciated and undercompensated, she said.
Writing recently in a blog post for the BMJ, Dr. Silver and colleagues predict that as a result of the pandemic, female physicians will “face disadvantages from unconscious bias in decisions about whose pay should be cut, whose operating schedules should take priority when resources are limited, and whose contributions merit retention ... The ground that women lose now will likely have a profound effect for many years to come, perhaps putting them at a disadvantage for the rest of their careers.”
There is already evidence of reduced publishing by female scientists during the pandemic, something that “could undermine the careers of an entire generation of women scholars,” noted Caitlyn Collins, PhD, assistant professor of sociology at Washington University in St. Louis.
“Science needs to address the culture of overwork,” Dr. Collins said in an interview. “Parents and other caregivers deserve support. The stress and ‘overwhelm’ they feel is not inevitable. A more fair, just, and humane approach to combining work and family is possible – what we need is the political will to pass better policies and a massive shift in our cultural understandings about how work should fit into family life, not the other way around.”
Lack of support for “vulnerable scientists,” particularly “junior scientists who are parents, women, or minorities” could lead to “severe attrition in cancer research in the coming years,” Cullen Taniguchi, MD, PhD, a radiation oncologist and associate professor at the University of Texas MD Anderson Cancer Center, Houston, and colleagues, warned in a recent letter to the journal Cancer Cell.
“The biggest worries of attrition will come from young faculty who started just before or after the pandemic,” Dr. Taniguchi said in an interview. “The first year in an academic setting is incredibly challenging but also important for establishing research efforts and building networks of colleagues to collaborate with. While completely necessary, the restrictions put in place during the pandemic made doing these things even more difficult.”
Another stressor: Caregiving at home
Another reason female physicians may be marginalized during the pandemic is that they are more often the primary caregivers at home.
“Anyone who is a caregiver, be it to kids, parents, or spouses, can relate to the challenges brought [on] by the pandemic,” said Ishwaria Subbiah, MD, a palliative care physician and medical oncologist at MD Anderson.
“Most of us work toward meeting our responsibilities by engaging a network of support, whether it’s home care workers, center-based or at-home child care, schools, or activities outside of school. The pandemic led to a level of disruption that brought most (if not all) of those responsibilities onto the caregivers themselves,” she said in an interview.
As the mother of an adult son with severe epilepsy, Dr. Bertagnolli has certainly experienced the challenges of parenting during the pandemic. “Our son is now 24 but he is handicapped, and lives with us. The care issues we have to deal with as professionals have been enormously magnified by COVID,” she said.
But she cautions against making gender distinctions when it comes to caregiving. “Has it fallen on the women? Well, this kind of stuff generally falls on the women, but I am certain it has fallen on an awful lot of men as well, because I think the world is changing that way, so it’s fallen on all of us.”
There is no question that female oncologists are bearing the brunt, both at work and at home, contended Dr. Taniguchi. “Absolutely. I have seen this first-hand,” he said.
“If it was difficult for women, underrepresented minorities, and junior faculty to find a voice in the room prepandemic, I think it can be harder in the times of virtual meetings when it is difficult to engage audiences,” he said.
Dr. Holstein said she is lucky to be well-supported at her institution, with both a female chief of hematology/oncology and a female chair of internal medicine, but still, she worries about the long-term consequences of the pandemic on the gender landscape of medicine.
“If you’re having to put aside research projects because you have extra responsibilities – again because women just tend to have a lot of other things going on – that might not be a big deal for 3 months, 6 months, but this is going to be a year or 2 years before ‘normal’ comes back,” she says. “One to two years of underpublishing or not getting the grants could be career killers for women in academic oncology.”
Cancer COVID-19 combo
As Dr. Wildes completed her final weeks of seeing cancer patients, she received an outpouring of support, which she says convinced her of the shared experience of all doctors, and especially female doctors, during the pandemic. But even more specifically, she feels that she has tapped into the unique burden shouldered by oncologists during this time.
“It’s intimidating being an oncologist; we are literally giving people poison for a living. Then throw into it a pandemic where early in March we had so little data. I was helping my patients make decisions about their cancer care based on a case series of four patients in China. The burden of those conversations is something I never want to have to live through again,” she said.
“Oncology is a particularly intense subspecialty within medicine,” agreed Dr. Subbiah. “The people we care for have received a life-altering and potentially life-limiting diagnosis. Coupled with that, the COVID-19 pandemic has brought an unprecedented cloud of uncertainty ... Whether the patients can see it overtly or not, oncologists carry the weight of this worry with them for not just one but all of their patients.”
Dr. Wildes said she plans to return to academic medicine and clinical care “in time,” but for now, the gap that she and others like her leave is troubling to those who have stayed on.
“We need these women in medicine,” said Dr. Holstein. “We have data suggesting that women take more time with their patients than men, that patient outcomes are better if they have a female physician. But also for the generations coming up, we need the mid-career and senior women to be in place to mentor and guide and make sure we continue to increase women in leadership.”
A version of this article originally appeared on Medscape.com.
For mid-career oncologist Tanya Wildes, MD, the pandemic was the last straw. In late September, she tweeted: “I have done the academically unfathomable: I am resigning my faculty position without another job lined up.”
She wasn’t burned out, she insisted. She loved her patients and her research. But she was also “100% confident” in her decision and “also 100% sad. This did not have to happen,” she lamented, asking not to disclose her workplace for fear of retribution.
Being a woman in medicine “is a hard life to start with,” Dr. Wildes said in an interview. “We all have that tenuous balance going on and the pandemic made everything just a little bit harder.”
She describes her prepandemic work-life balance as a “Jenga tower, with everything only just in place.” But she realized that the balance had tipped, when after a difficult clinic she felt emotionally wrung out. Her 11-year-old son had asked her to help him fly his model airplane. “I told him, ‘Honey, I can’t do it because if it crashes or gets stuck in a tree ... you’re going to be devastated and I have nothing left for you.’ “
This was a eureka moment, as “I realized, this is not who I want to be,” she said, holding back tears. “Seventy years from now my son is going to tell his grandchildren about the pandemic and I don’t want his memory of his mom to be that she couldn’t be there for him because she was too spent.”
When Dr. Wildes shared her story on Twitter, other female oncologists and physicians responded that they too have felt they’re under increased pressure this year, with the extra stress of the pandemic leading others to quit as well.
The trend of doctors leaving medicine has been noticeable. A July survey from the Physicians Foundation found that roughly 16,000 medical practices had already closed during the pandemic, with another 8,000 predicted to close within the next year.
“Similar patterns” were evident in another analysis by the Larry A. Green Center and the Primary Care Collaborative, as reported in The New York Times. In that survey, nearly one-fifth of primary care clinicians said “someone in their practice plans to retire early or has already retired because of COVID-19,” and 15% say “someone has left or plans to leave the practice.” About half said their mental exhaustion was at an all-time high, the survey found.
“COVID-19 is a burden, and that added burden has tipped people over the edge of many things,” said Monica Bertagnolli, MD, chief of the division of surgical oncology at Brigham and Women’s Hospital, Boston, and former president of the American Society of Clinical Oncology.
“It has illustrated that we do have a lot of people who are working kind of on the edge of not being able to handle everything,” she said.
While many in medicine are struggling, the pandemic seems to be pushing more women to leave, highlighting longtime gender disparities and increased caregiving burdens. And their absence may be felt for years to come.
Firm numbers are hard to come by, said Julie Silver, MD, associate professor, associate chair, and director of cancer rehabilitation in the department of physical medicine and rehabilitation at Harvard Medical School, Boston, and an expert in gender equity in medicine. But she sees some troubling trends.
“There are many indications that women are leaving medicine in disproportionately high numbers,” Dr. Silver said in an interview. “A lack of fair pay and promotion opportunities that were present before COVID-19 are now combined with a host of pandemic-related challenges.”
A survey of 1,809 women conducted in mid-April with the Physician Moms Facebook Group and accepted for online publication by the American Journal of Psychiatry found that 41% scored over the cutoff points for moderate or severe anxiety, with 46% meeting these criteria among front-line workers.
“It’s really important for society to recognize the extraordinary impact this pandemic is having on physician mothers, as there will be profound ripple effects on the ability of this key segment of the health care workforce to serve others if we do not address this problem urgently,” co-senior author Reshma Jagsi, MD, DPhil, a radiation oncologist at the University of Michigan, Ann Arbor, said in an interview.
Women weighed in on Twitter, in response to Dr. Silver’s tweet to #WomenInMedicine: “If you are thinking of leaving #medicine & need a reason to stay: we value you & need you.”
In reply, Emmy Betz, MD, MPH, associate professor of emergency medicine at the University of Colorado at Denver, Aurora, said via Twitter, “I’ve had lots of conversations with women considering leaving medicine.”
“I have thought about leaving many times. I love what I do, but medicine can be an unkind world at times,” responded Valerie Fitzhugh MD, associate professor and pathologist at New Jersey Medical School, Newark.
“Too late. Left at the end of July and it was the best decision ever,” wrote Michelle Gordon, DO, who was previously a board-certified general surgeon at Northern Westchester Surgical Associates in Putnam Valley, N.Y.
Prepandemic disparities accentuated
The pandemic “has merely accentuated – or made more apparent – some of the longstanding issues and struggles of women in oncology, women in medicine, women in academia,” said Sarah Holstein, MD, PhD, another mid-career oncologist and associate professor at the University of Nebraska Medical Center, Omaha.
“There are disparities in first-author/last-author publications, disparities in being asked to give speaking engagements, disparities in leadership,” Dr. Holstein said in an interview. “And then ... put on top of that the various surges with the pandemic where you are being asked to do clinical responsibilities you don’t normally do, perhaps some things you haven’t done since your training 10 or 20 years ago.”
This is backed up with data: There is already a “robust” body of prepandemic literature demonstrating pay gaps for female physicians and scientists, noted Dr. Silver, who founded the Her Time Is Now campaign for gender equity in medicine and runs a women’s leadership course at Harvard.
In addition, female physicians are more likely to be involved in “nonpromotable” work, group projects and educator roles that are often underappreciated and undercompensated, she said.
Writing recently in a blog post for the BMJ, Dr. Silver and colleagues predict that as a result of the pandemic, female physicians will “face disadvantages from unconscious bias in decisions about whose pay should be cut, whose operating schedules should take priority when resources are limited, and whose contributions merit retention ... The ground that women lose now will likely have a profound effect for many years to come, perhaps putting them at a disadvantage for the rest of their careers.”
There is already evidence of reduced publishing by female scientists during the pandemic, something that “could undermine the careers of an entire generation of women scholars,” noted Caitlyn Collins, PhD, assistant professor of sociology at Washington University in St. Louis.
“Science needs to address the culture of overwork,” Dr. Collins said in an interview. “Parents and other caregivers deserve support. The stress and ‘overwhelm’ they feel is not inevitable. A more fair, just, and humane approach to combining work and family is possible – what we need is the political will to pass better policies and a massive shift in our cultural understandings about how work should fit into family life, not the other way around.”
Lack of support for “vulnerable scientists,” particularly “junior scientists who are parents, women, or minorities” could lead to “severe attrition in cancer research in the coming years,” Cullen Taniguchi, MD, PhD, a radiation oncologist and associate professor at the University of Texas MD Anderson Cancer Center, Houston, and colleagues, warned in a recent letter to the journal Cancer Cell.
“The biggest worries of attrition will come from young faculty who started just before or after the pandemic,” Dr. Taniguchi said in an interview. “The first year in an academic setting is incredibly challenging but also important for establishing research efforts and building networks of colleagues to collaborate with. While completely necessary, the restrictions put in place during the pandemic made doing these things even more difficult.”
Another stressor: Caregiving at home
Another reason female physicians may be marginalized during the pandemic is that they are more often the primary caregivers at home.
“Anyone who is a caregiver, be it to kids, parents, or spouses, can relate to the challenges brought [on] by the pandemic,” said Ishwaria Subbiah, MD, a palliative care physician and medical oncologist at MD Anderson.
“Most of us work toward meeting our responsibilities by engaging a network of support, whether it’s home care workers, center-based or at-home child care, schools, or activities outside of school. The pandemic led to a level of disruption that brought most (if not all) of those responsibilities onto the caregivers themselves,” she said in an interview.
As the mother of an adult son with severe epilepsy, Dr. Bertagnolli has certainly experienced the challenges of parenting during the pandemic. “Our son is now 24 but he is handicapped, and lives with us. The care issues we have to deal with as professionals have been enormously magnified by COVID,” she said.
But she cautions against making gender distinctions when it comes to caregiving. “Has it fallen on the women? Well, this kind of stuff generally falls on the women, but I am certain it has fallen on an awful lot of men as well, because I think the world is changing that way, so it’s fallen on all of us.”
There is no question that female oncologists are bearing the brunt, both at work and at home, contended Dr. Taniguchi. “Absolutely. I have seen this first-hand,” he said.
“If it was difficult for women, underrepresented minorities, and junior faculty to find a voice in the room prepandemic, I think it can be harder in the times of virtual meetings when it is difficult to engage audiences,” he said.
Dr. Holstein said she is lucky to be well-supported at her institution, with both a female chief of hematology/oncology and a female chair of internal medicine, but still, she worries about the long-term consequences of the pandemic on the gender landscape of medicine.
“If you’re having to put aside research projects because you have extra responsibilities – again because women just tend to have a lot of other things going on – that might not be a big deal for 3 months, 6 months, but this is going to be a year or 2 years before ‘normal’ comes back,” she says. “One to two years of underpublishing or not getting the grants could be career killers for women in academic oncology.”
Cancer COVID-19 combo
As Dr. Wildes completed her final weeks of seeing cancer patients, she received an outpouring of support, which she says convinced her of the shared experience of all doctors, and especially female doctors, during the pandemic. But even more specifically, she feels that she has tapped into the unique burden shouldered by oncologists during this time.
“It’s intimidating being an oncologist; we are literally giving people poison for a living. Then throw into it a pandemic where early in March we had so little data. I was helping my patients make decisions about their cancer care based on a case series of four patients in China. The burden of those conversations is something I never want to have to live through again,” she said.
“Oncology is a particularly intense subspecialty within medicine,” agreed Dr. Subbiah. “The people we care for have received a life-altering and potentially life-limiting diagnosis. Coupled with that, the COVID-19 pandemic has brought an unprecedented cloud of uncertainty ... Whether the patients can see it overtly or not, oncologists carry the weight of this worry with them for not just one but all of their patients.”
Dr. Wildes said she plans to return to academic medicine and clinical care “in time,” but for now, the gap that she and others like her leave is troubling to those who have stayed on.
“We need these women in medicine,” said Dr. Holstein. “We have data suggesting that women take more time with their patients than men, that patient outcomes are better if they have a female physician. But also for the generations coming up, we need the mid-career and senior women to be in place to mentor and guide and make sure we continue to increase women in leadership.”
A version of this article originally appeared on Medscape.com.
Patients with HF have higher risks of postop mortality, complications after ambulatory noncardiac surgery
Background: Heart failure is a known risk factor for postoperative mortality and complications. Many of the studies used to establish this association, however, have focused on major high-risk surgeries and not on outpatient surgeries. Improved medical care has increased the survival rate of patients with heart failure and an increasing number of these patients are undergoing elective surgical procedures. This has led to an increasing need to better understand the degree to which heart failure affects preoperative risk in the outpatient setting.
Study design: A retrospective cohort study.
Setting: Multiple Veteran’s Affairs Hospitals using data from the VA Surgical Quality Improvement Program (VASQIP) and the VA Corporate Data Warehouse.
Synopsis: A total of 355,121 patients who underwent outpatient surgeries were analyzed. 19,353 patients had heart failure and 334,768 did not. Patients with heart failure had a higher risk of 90-day mortality with an adjusted odds ratio of 1.95 (95% confidence interval, 1.69-2.44), and this risk progressively increased as the ejection fraction decreased. The risk of 30-day complication also increased in patients with heart failure with an adjusted OR of 1.10 (95% CI, 1.02-1.19).
Limitations of this study include the patient population, which were all veterans and mostly male. The nature of the inclusion criteria was limiting as well, in that all the patients in this study were deemed fit for surgery. There were no data available for patients who had been considered but ultimately did not undergo surgery or for patients who were considered for ambulatory surgery but ultimately underwent inpatient surgery. These limitations may have resulted in a selection bias, which limited the generalizability of the study’s findings when assessing patients for ambulatory surgery.
Bottom line: Patients with heart failure had a higher risk of 90-day postoperative mortality and 30-day postoperative complication in ambulatory noncardiac surgery. The risk of postoperative mortality increased as systolic function decreased.
Citation: Lerman BJ et al. Association between heart failure and postoperative mortality among patients undergoing ambulatory noncardiac surgery. JAMA Surg. 2019 Jul 10. doi: 10.1001/jamasurg.2019.2110.
Dr. Cheatham is a hospitalist and clinical educator at St. Louis University School of Medicine.
Background: Heart failure is a known risk factor for postoperative mortality and complications. Many of the studies used to establish this association, however, have focused on major high-risk surgeries and not on outpatient surgeries. Improved medical care has increased the survival rate of patients with heart failure and an increasing number of these patients are undergoing elective surgical procedures. This has led to an increasing need to better understand the degree to which heart failure affects preoperative risk in the outpatient setting.
Study design: A retrospective cohort study.
Setting: Multiple Veteran’s Affairs Hospitals using data from the VA Surgical Quality Improvement Program (VASQIP) and the VA Corporate Data Warehouse.
Synopsis: A total of 355,121 patients who underwent outpatient surgeries were analyzed. 19,353 patients had heart failure and 334,768 did not. Patients with heart failure had a higher risk of 90-day mortality with an adjusted odds ratio of 1.95 (95% confidence interval, 1.69-2.44), and this risk progressively increased as the ejection fraction decreased. The risk of 30-day complication also increased in patients with heart failure with an adjusted OR of 1.10 (95% CI, 1.02-1.19).
Limitations of this study include the patient population, which were all veterans and mostly male. The nature of the inclusion criteria was limiting as well, in that all the patients in this study were deemed fit for surgery. There were no data available for patients who had been considered but ultimately did not undergo surgery or for patients who were considered for ambulatory surgery but ultimately underwent inpatient surgery. These limitations may have resulted in a selection bias, which limited the generalizability of the study’s findings when assessing patients for ambulatory surgery.
Bottom line: Patients with heart failure had a higher risk of 90-day postoperative mortality and 30-day postoperative complication in ambulatory noncardiac surgery. The risk of postoperative mortality increased as systolic function decreased.
Citation: Lerman BJ et al. Association between heart failure and postoperative mortality among patients undergoing ambulatory noncardiac surgery. JAMA Surg. 2019 Jul 10. doi: 10.1001/jamasurg.2019.2110.
Dr. Cheatham is a hospitalist and clinical educator at St. Louis University School of Medicine.
Background: Heart failure is a known risk factor for postoperative mortality and complications. Many of the studies used to establish this association, however, have focused on major high-risk surgeries and not on outpatient surgeries. Improved medical care has increased the survival rate of patients with heart failure and an increasing number of these patients are undergoing elective surgical procedures. This has led to an increasing need to better understand the degree to which heart failure affects preoperative risk in the outpatient setting.
Study design: A retrospective cohort study.
Setting: Multiple Veteran’s Affairs Hospitals using data from the VA Surgical Quality Improvement Program (VASQIP) and the VA Corporate Data Warehouse.
Synopsis: A total of 355,121 patients who underwent outpatient surgeries were analyzed. 19,353 patients had heart failure and 334,768 did not. Patients with heart failure had a higher risk of 90-day mortality with an adjusted odds ratio of 1.95 (95% confidence interval, 1.69-2.44), and this risk progressively increased as the ejection fraction decreased. The risk of 30-day complication also increased in patients with heart failure with an adjusted OR of 1.10 (95% CI, 1.02-1.19).
Limitations of this study include the patient population, which were all veterans and mostly male. The nature of the inclusion criteria was limiting as well, in that all the patients in this study were deemed fit for surgery. There were no data available for patients who had been considered but ultimately did not undergo surgery or for patients who were considered for ambulatory surgery but ultimately underwent inpatient surgery. These limitations may have resulted in a selection bias, which limited the generalizability of the study’s findings when assessing patients for ambulatory surgery.
Bottom line: Patients with heart failure had a higher risk of 90-day postoperative mortality and 30-day postoperative complication in ambulatory noncardiac surgery. The risk of postoperative mortality increased as systolic function decreased.
Citation: Lerman BJ et al. Association between heart failure and postoperative mortality among patients undergoing ambulatory noncardiac surgery. JAMA Surg. 2019 Jul 10. doi: 10.1001/jamasurg.2019.2110.
Dr. Cheatham is a hospitalist and clinical educator at St. Louis University School of Medicine.
Patient health suffers amid pandemic health care shortages
More than half (56%) of responding clinicians reported seeing a decline in patient health because of delayed or inaccessible care amid the pandemic, according to the results of the latest survey by the Larry A. Green Center and the Primary Care Collaborative. The survey was conducted in mid-October and the results were published online Nov. 17.
In addition, 37% of respondents said their patients with chronic conditions showed “noticeably worse health resulting from the pandemic.” And a resounding 85% said patient mental health had worsened.
“I think it’s worse than we thought,” said Rebecca Etz, PhD, codirector of the Larry Green Center. “It’s the outcome of not sufficiently sending resources to primary care either before or during the pandemic.” According to Dr. Etz, survey respondents noted substantial increases in patient weight gain as well as weight loss, anxiety and depression, sleep issues, domestic abuse, and poor oral and eye health, among others.
One clinician from Pennsylvania wrote: “Patients are becoming sicker during the pandemic. I’m seeing more uncontrolled [diabetes]and new [patients with diabetes]. They prefer telehealth yet [have] no access to glucose monitoring or a blood pressure cuff. I am concerned about patients’ isolation and mental health. People are delaying care.”
Now, with COVID numbers peaking across much of the country, many clinicians are trying to close the gap in care with telehealth – something they’re more prepared to do now than they were in March. Over two-thirds of practices are using telehealth for visits to keep up with patients who have stable chronic conditions, according to the survey.
Over 60% of physicians report using telehealth for mental health visits. But a much smaller number – only 16% of respondents – said their practice had added staff to help manage the rising number of behavioral and mental health cases. About one-third (35%) of practices say they’re not financially able to take on new staff.
“We’ve been looking for more ways for patients to do self-support. A big part of chronic disease is health behaviors,” Alex Krist, MD, MPH, a family doctor in Fairfax, Va., and chairperson of the U.S. Preventive Services Task Force, said in an interview. And unfortunately, on top of limited access to basic care, healthy habits that are essential to managing many chronic conditions have become more difficult and less consistent during the pandemic.
The survey – the 22nd iteration in a series of surveys the Green Center and the Primary Care Collaborative have conducted – received 580 respondents from 47 states and Guam. Over two-thirds of respondents were primary care physicians (MDs and DOs). Over half were owners, partners, or employees of a private practice, 66% of which were family medicine practices. And one fifth of respondents provided care in a rural area.
Funding and support for primary care has been wildly insufficient, Dr. Etz said in an interview. If that doesn’t change, patient health, clinic staffing, and public health strategies amid the pandemic will continue to suffer.
“When you think of the COVID vaccine, who do you think is going to be sending that out?” Dr. Etz asked. “If we don’t bolster primary care now how are they going to handle that.”
A version of this article originally appeared on Medscape.com.
More than half (56%) of responding clinicians reported seeing a decline in patient health because of delayed or inaccessible care amid the pandemic, according to the results of the latest survey by the Larry A. Green Center and the Primary Care Collaborative. The survey was conducted in mid-October and the results were published online Nov. 17.
In addition, 37% of respondents said their patients with chronic conditions showed “noticeably worse health resulting from the pandemic.” And a resounding 85% said patient mental health had worsened.
“I think it’s worse than we thought,” said Rebecca Etz, PhD, codirector of the Larry Green Center. “It’s the outcome of not sufficiently sending resources to primary care either before or during the pandemic.” According to Dr. Etz, survey respondents noted substantial increases in patient weight gain as well as weight loss, anxiety and depression, sleep issues, domestic abuse, and poor oral and eye health, among others.
One clinician from Pennsylvania wrote: “Patients are becoming sicker during the pandemic. I’m seeing more uncontrolled [diabetes]and new [patients with diabetes]. They prefer telehealth yet [have] no access to glucose monitoring or a blood pressure cuff. I am concerned about patients’ isolation and mental health. People are delaying care.”
Now, with COVID numbers peaking across much of the country, many clinicians are trying to close the gap in care with telehealth – something they’re more prepared to do now than they were in March. Over two-thirds of practices are using telehealth for visits to keep up with patients who have stable chronic conditions, according to the survey.
Over 60% of physicians report using telehealth for mental health visits. But a much smaller number – only 16% of respondents – said their practice had added staff to help manage the rising number of behavioral and mental health cases. About one-third (35%) of practices say they’re not financially able to take on new staff.
“We’ve been looking for more ways for patients to do self-support. A big part of chronic disease is health behaviors,” Alex Krist, MD, MPH, a family doctor in Fairfax, Va., and chairperson of the U.S. Preventive Services Task Force, said in an interview. And unfortunately, on top of limited access to basic care, healthy habits that are essential to managing many chronic conditions have become more difficult and less consistent during the pandemic.
The survey – the 22nd iteration in a series of surveys the Green Center and the Primary Care Collaborative have conducted – received 580 respondents from 47 states and Guam. Over two-thirds of respondents were primary care physicians (MDs and DOs). Over half were owners, partners, or employees of a private practice, 66% of which were family medicine practices. And one fifth of respondents provided care in a rural area.
Funding and support for primary care has been wildly insufficient, Dr. Etz said in an interview. If that doesn’t change, patient health, clinic staffing, and public health strategies amid the pandemic will continue to suffer.
“When you think of the COVID vaccine, who do you think is going to be sending that out?” Dr. Etz asked. “If we don’t bolster primary care now how are they going to handle that.”
A version of this article originally appeared on Medscape.com.
More than half (56%) of responding clinicians reported seeing a decline in patient health because of delayed or inaccessible care amid the pandemic, according to the results of the latest survey by the Larry A. Green Center and the Primary Care Collaborative. The survey was conducted in mid-October and the results were published online Nov. 17.
In addition, 37% of respondents said their patients with chronic conditions showed “noticeably worse health resulting from the pandemic.” And a resounding 85% said patient mental health had worsened.
“I think it’s worse than we thought,” said Rebecca Etz, PhD, codirector of the Larry Green Center. “It’s the outcome of not sufficiently sending resources to primary care either before or during the pandemic.” According to Dr. Etz, survey respondents noted substantial increases in patient weight gain as well as weight loss, anxiety and depression, sleep issues, domestic abuse, and poor oral and eye health, among others.
One clinician from Pennsylvania wrote: “Patients are becoming sicker during the pandemic. I’m seeing more uncontrolled [diabetes]and new [patients with diabetes]. They prefer telehealth yet [have] no access to glucose monitoring or a blood pressure cuff. I am concerned about patients’ isolation and mental health. People are delaying care.”
Now, with COVID numbers peaking across much of the country, many clinicians are trying to close the gap in care with telehealth – something they’re more prepared to do now than they were in March. Over two-thirds of practices are using telehealth for visits to keep up with patients who have stable chronic conditions, according to the survey.
Over 60% of physicians report using telehealth for mental health visits. But a much smaller number – only 16% of respondents – said their practice had added staff to help manage the rising number of behavioral and mental health cases. About one-third (35%) of practices say they’re not financially able to take on new staff.
“We’ve been looking for more ways for patients to do self-support. A big part of chronic disease is health behaviors,” Alex Krist, MD, MPH, a family doctor in Fairfax, Va., and chairperson of the U.S. Preventive Services Task Force, said in an interview. And unfortunately, on top of limited access to basic care, healthy habits that are essential to managing many chronic conditions have become more difficult and less consistent during the pandemic.
The survey – the 22nd iteration in a series of surveys the Green Center and the Primary Care Collaborative have conducted – received 580 respondents from 47 states and Guam. Over two-thirds of respondents were primary care physicians (MDs and DOs). Over half were owners, partners, or employees of a private practice, 66% of which were family medicine practices. And one fifth of respondents provided care in a rural area.
Funding and support for primary care has been wildly insufficient, Dr. Etz said in an interview. If that doesn’t change, patient health, clinic staffing, and public health strategies amid the pandemic will continue to suffer.
“When you think of the COVID vaccine, who do you think is going to be sending that out?” Dr. Etz asked. “If we don’t bolster primary care now how are they going to handle that.”
A version of this article originally appeared on Medscape.com.






