User login
UPDATE ON BREAST HEALTH
Women with ER-positive breast cancer may soon extend tamoxifen therapy to 10 years
Janelle Yates (February 2013)
Is overdiagnosis of breast cancer common among women screened
by mammography?
Andrew M. Kaunitz, MD (Examining the Evidence; January 2013)
Breast cancer genome analysis highlights 4 subtypes, link to
ovarian cancer
Janelle Yates (News for Your Practice; November 2012)
The effects of breast cancer on obstetric and gynecologic practices are pervasive. In this article, we touch on three aspects of breast cancer that are particularly relevant to the practicing ObGyn:
- the need to identify women at high risk for breast cancer and select those who would benefit from a discussion of the advantages and risks of chemoprophylaxis, which can reduce the likelihood of breast cancer by 50% or more
- the need for strategies to manage menopausal symptoms in the general population without increasing the risk of breast cancer. The traditional approach to this problem changed dramatically with the Women’s Health Initiative (WHI), which demonstrated an increased risk of breast cancer in women taking conjugated equine estrogen and progestin. The widely publicized initial findings of the estrogen-progestin arm of the WHI sharply contrast the equally relevant, somewhat unexpected, and less publicized results of the estrogen-alone arm, which demonstrated a substantial and statistically significant decrease in the incidence of breast cancer, even after estrogen was discontinued.
- the potential effects of breast cancer treatment on ovarian function in young women. This year, of the approximately 250,000 women who will be diagnosed with invasive breast cancer, more than 50,000 women will be of reproductive age. Most of these young women will require adjuvant chemotherapy; as a result, many will experience the premature onset of menopause. Along with the attendant loss of fertility these women will face, many will also develop distressing and life-altering menopausal symptoms. Management of these women before and after initiation of chemotherapy requires an understanding of both the expected effects of the chemotherapy and knowledge of how to actively manage these women with strategies to either prevent these events or to manage menopausal symptoms.
In women at normal risk for breast cancer, unopposed estrogen lowers the rate of the malignancy and the likelihood of mortality if the cancer occurs—but is not recommended as a prophylactic agent. Tamoxifen and other chemoprophylactic drugs can halve the rate of breast cancer in high-risk women but are not without drawbacks.
A look at the lower rate of breast cancer in the estrogen-alone arm of the WHI
Anderson GL, Chlebowski RT, Aragaki AK, et al. Conjugated equine oestrogen and breast cancer incidence and mortality in postmenopausal women with hysterectomy: extended follow-up of the Women’s Health Initiative randomised placebo-controlled trial. Lancet Oncol. 2012;13(5):476–486.
From 1993 through 1998, the WHI enrolled 10,739 postmenopausal women in the largest prospective trial evaluating the effect of hormone therapy (HT) on various clinical outcomes. The women were randomly allocated to three groups:
- conjugated estrogen with medroxyprogesterone acetate
- conjugated estrogen alone (in women with a prior hysterectomy)
- placebo.
The negative effects of estrogen plus progestin on the risk of breast cancer were the most widely discussed oucomes.1 Shortly after the findings from this arm of the study were published, the use of HT in the United States declined dramatically and unequivocally.2
In 2012, WHI published the results of the estrogen-alone arm in the British cancer specialty journal Lancet Oncology. As shown in the TABLE below, the incidence of breast cancer was statistically significantly lower (23%) in the estrogen group than in the placebo group. Women who were treated with estrogen alone were also 63% less likely to die of breast cancer, and all-cause mortality was 38% lower; both of these findings were statistically significant. Not only was there a significant reduction in the incidence of invasive breast cancer while the subjects were taking estrogen, but that reduction continued for a median of 4.7 years of follow-up after discontinuation of estrogen.
Breast cancer incidence and mortality in the estrogen-only arm of the WHI, compared with placebo*
| Event | Estrogen only (n = 5,310) | Placebo (n = 5,429) | Hazard ratio (95% confidence interval) |
|---|---|---|---|
| Invasive breast cancer | 151 (0.27%) | 199 (0.35%) | 0.77 (0.62–0.95) |
| Node-negative breast cancer | 88 (0.16%) | 134 (0.24%) | 0.67 (0.51–0.88) |
| Breast cancer mortality | 6 (0.009%) | 16 (0.024%) | 0.37 (0.13–0.91) |
| All-cause mortality | 30 (0.046%) | 50 (0.076%) | 0.62 (0.39–0.97) |
| * Median follow-up of 11.8 years | |||
The incidence figure is somewhat remarkable (199 in the placebo group versus 151 in the estrogen-alone group) in that it was nearly the exact reverse of the estrogen-progestin arm of the WHI trial (199 in the estrogen/progestin group vs 150 in the placebo group).3
Estrogen alone reduced both breast cancer incidence and breast cancer mortality while women were on therapy and for 5 years after discontinuing therapy. This finding should reassure women who have undergone hysterectomy, as well as their clinicians, that estrogen alone reduces the future likelihood of breast cancer. It should be noted that the effect of estrogen alone in women in higher-risk categories did not show a reduction in breast cancer, and for this reason, the authors cautioned against considering the use of estrogen alone in menopausal women as a breast cancer chemoprophylaxis agent.
All breast cancer chemoprophylactic agents carry risks as well as benefits
Goss P, Ingle J, Ales-Martinez J, et al. Exemestane for breast-cancer prevention in postmenopausal women. N Engl J Med. 2011;364(25):2381–2391.
Cheung A, Tile L, Cardew S, et al. Bone density and structure in healthy postmenopausal women treated with exemestane for the primary prevention of breast cancer: a nested substudy of the MAP.3 randomized controlled trial. Lancet Oncol. 2012;13(3):275–284.
Vogel V, Costantino J, Wickerham L, et al. Update of the National Surgical Adjuvant Breast and Bowel Project Study of Tamoxifen and Raloxifene (STAR) P-2 Trial: preventing breast cancer. Cancer Prev Res. 2010;3(6):696–706.
The number of new cases of breast cancer in the United States last year reached nearly a quarter-million. Clearly, reducing this number remains an important goal.4 Chemoprevention—the use of medication to reduce cancer risk—may be offered to women who are at high risk of developing breast cancer.
In the National Surgical Adjuvant Breast and Bowel Project (NSABP) P-1 trial, tamoxifen (a selective estrogen-receptor modulator) was shown to reduce the risk of invasive breast cancer by 49% in a high-risk population, resulting in the FDA approving tamoxifen as the first drug for breast cancer prevention.5 The P-1 trial was followed by the NSABP Study of Tamoxifen and Raloxifene (STAR) P-2 trial, which demonstrated relative equivalence between the two medications as cancer prevention agents in menopausal women.6 Serious side effects of these medications limit their use among eligible women, although raloxifene seems to be associated with fewer adverse events. In the update of the STAR trial with an average of 81 months of follow-up, the risk ratio for adverse events (raloxifene:tamoxifen) was 0.75 for thromboembolic events, 0.55 for endometrial cancer, and 0.19 for uterine hyperplasia.
Another drug used for cancer treatment has now entered the prevention scene. In 2011, the NCIC Clinical Trials Group Mammary Prevention.3 trial (NCIC CTG MAP.3) compared exemestane (an aromatase inhibitor) with placebo for menopausal women at high risk for breast cancer, demonstrating a 65% relative reduction in the incidence of invasive breast cancer. This study validated another option for cancer prevention in high-risk women, although its adoption is likely also to be limited by side effects, including vasomotor symptoms, a high rate of arthralgias, and vaginal dryness/dyspareunia. The greatest concern may be the potential effect on bone density. Though the rates of serious adverse events including fracture did not differ in the MAP.3 trial at 35 months of follow-up, women on exemestane had significantly larger losses of bone mineral density, compared with controls.
Chemoprophylaxis reduces the risk of breast cancer in high-risk women by about 50%. Who are good candidates for these medications? Based on these trials, menopausal women considered at high risk might include those with a Gail risk score of at least 1.66% (ie, risk of developing breast cancer in 5 years), age 60 years or older, and women with biopsy results demonstrating atypical hyperplasia or lobular carcinoma in situ (LCIS). (The Gail model is available at www.cancer.gov/bcrisktool.) Tamoxifen is the only option for premenopausal women age 35 and older. Those who have histologic markers of risk (atypical hyperplasia, LCIS) likely stand to derive the greatest benefit.4
Managing the reproductive health concerns of young women with breast cancer
Azim H, Kroman N, Paesmans M, et al. Prognostic impact of pregnancy after breast cancer according to estrogen receptor status: a multicenter retrospective study. J Clin Oncol. 2013;31(1):73–79.
Howard-Anderson J, Ganz P, Bower J, Stanton A. Quality of life, fertility concerns, and behavioral health outcomes in younger breast cancer survivors: a systematic review. J Natl Cancer Inst. 2012;104(5):1386–1405.
Of the approximately 230,000 new cases of invasive breast cancer identified in 2011, 50,430 cases involved women less than 50 years of age.4 For these women, the diagnosis of cancer raises multifaceted concerns, including the physical changes that accompany breast cancer treatment, concerns about recurrence and mortality, and significant sexual and reproductive consequences of treatment that alters ovarian function. Pregnancy-associated breast cancers (breast cancers diagnosed during pregnancy, lactation, and for 12 months postpartum) represent a small subset of these cancers and occur in about 1 in 3,000 pregnancies. One might anticipate that this rate will increase as women continue to delay childbearing, because pregnancy-associated breast cancers are more common in older women.
In the review article by Howard-Anderson and colleagues, the importance of these reproductive health consequences in young women diagnosed with breast cancer is highlighted. The women who transition to menopause as a result of chemotherapy (reported to range from 33%–73%) experience more symptoms, including hot flashes, night sweats, breast pain, vaginal dryness, and lack of sexual desire. Sixty-one percent of women younger than 40 years at diagnosis reported that they were concerned about menopause, and 30% reported that this concern influenced their treatment decisions. Thirty-nine percent of women in this group had major concerns about treatment-associated infertility, and only half of the women studied felt that their fertility concerns were adequately addressed.
On a positive note, for women who successfully achieve pregnancy after breast cancer, pregnancy outcomes appear to be similar to those of their nonpregnant peers. In the study by Azim and colleagues, women who became pregnant after a breast cancer diagnosis had disease-free survival that was statistically similar to that of matched women who did not have subsequent pregnancies. In addition, this outcome did not differ based on estrogen/progesterone receptor status (ER/PR positive or negative).
Both alkylating chemotherapeutic agents (eg, cyclophosphamide) and selective estrogen receptor modulating agents (for women with estrogen-receptor–positive tumors) are routine parts of adjuvant treatment for premenopausal women with invasive breast cancers.
These agents can have profound effects on both ovarian hormonal function and fertility. ObGyns and reproductive endocrinology/infertility specialists have a great opportunity to partner with our oncology colleagues to enhance the counseling that young women receive before, during, and after breast cancer treatment.
Women who are considering future childbearing should receive information about the impact of breast cancer treatment on fertility and options for fertility preservation prior to initiating treatment. For women who have completed childbearing, information on what to expect if menopause occurs and available options for symptom relief can be empowering as they make treatment decisions.
We want to hear from you! Tell us what you think.
1. Grady D. Study finds new risks in hormone therapy. New York Times. http://www.nytimes.com/2003/06/25/us/study-finds-new-risks-in-hormone-therapy.html?pagewanted=all&src=pm. Published June 25 2003. Accessed February 11, 2013.
2. Hersh AL, Stefanick ML, Stafford RS. National use of menopausal hormone therapy: annual trends and response to recent evidence. JAMA. 2004;291(1):47-53.
3. Chlebowski RT, Kuller LH, Prentice RL, et al. Women’s Health Initiative Investigators. Breast cancer after use of estrogen plus progestin in postmenopausal women. N Engl J Med. 2009;360(6):573-587.
4. American Cancer Society. Breast Cancer Facts and Figures 2011-2012. Atlanta, GA: American Cancer Society. http://www.cancer.org/research/cancerfactsfigures/breastcancerfactsfigures/breast-cancer-facts-and-figures-2011-2012. Accessed February 11, 2013.
5. Fisher B, Constantino J, Wickerham L, et al. Tamoxifen for prevention of breast cancer: report of the National Surgical Adjuvant Breast and Bowel Project P-1 Study. J Natl Cancer Inst. 1998;90(18):1371-1388.
6. Vogel V, Costantino J, Wickerham DL, et al. Effects of tamoxifen vs raloxifene on the risk of developing invasive breast cancer and other disease outcomes. The NSABP Study of Tamoxifen and Raloxifene (STAR) P-2 Trial. JAMA. 2006;295(23):2727-2741.
Women with ER-positive breast cancer may soon extend tamoxifen therapy to 10 years
Janelle Yates (February 2013)
Is overdiagnosis of breast cancer common among women screened
by mammography?
Andrew M. Kaunitz, MD (Examining the Evidence; January 2013)
Breast cancer genome analysis highlights 4 subtypes, link to
ovarian cancer
Janelle Yates (News for Your Practice; November 2012)
The effects of breast cancer on obstetric and gynecologic practices are pervasive. In this article, we touch on three aspects of breast cancer that are particularly relevant to the practicing ObGyn:
- the need to identify women at high risk for breast cancer and select those who would benefit from a discussion of the advantages and risks of chemoprophylaxis, which can reduce the likelihood of breast cancer by 50% or more
- the need for strategies to manage menopausal symptoms in the general population without increasing the risk of breast cancer. The traditional approach to this problem changed dramatically with the Women’s Health Initiative (WHI), which demonstrated an increased risk of breast cancer in women taking conjugated equine estrogen and progestin. The widely publicized initial findings of the estrogen-progestin arm of the WHI sharply contrast the equally relevant, somewhat unexpected, and less publicized results of the estrogen-alone arm, which demonstrated a substantial and statistically significant decrease in the incidence of breast cancer, even after estrogen was discontinued.
- the potential effects of breast cancer treatment on ovarian function in young women. This year, of the approximately 250,000 women who will be diagnosed with invasive breast cancer, more than 50,000 women will be of reproductive age. Most of these young women will require adjuvant chemotherapy; as a result, many will experience the premature onset of menopause. Along with the attendant loss of fertility these women will face, many will also develop distressing and life-altering menopausal symptoms. Management of these women before and after initiation of chemotherapy requires an understanding of both the expected effects of the chemotherapy and knowledge of how to actively manage these women with strategies to either prevent these events or to manage menopausal symptoms.

In women at normal risk for breast cancer, unopposed estrogen lowers the rate of the malignancy and the likelihood of mortality if the cancer occurs—but is not recommended as a prophylactic agent. Tamoxifen and other chemoprophylactic drugs can halve the rate of breast cancer in high-risk women but are not without drawbacks.
A look at the lower rate of breast cancer in the estrogen-alone arm of the WHI
Anderson GL, Chlebowski RT, Aragaki AK, et al. Conjugated equine oestrogen and breast cancer incidence and mortality in postmenopausal women with hysterectomy: extended follow-up of the Women’s Health Initiative randomised placebo-controlled trial. Lancet Oncol. 2012;13(5):476–486.
From 1993 through 1998, the WHI enrolled 10,739 postmenopausal women in the largest prospective trial evaluating the effect of hormone therapy (HT) on various clinical outcomes. The women were randomly allocated to three groups:
- conjugated estrogen with medroxyprogesterone acetate
- conjugated estrogen alone (in women with a prior hysterectomy)
- placebo.
The negative effects of estrogen plus progestin on the risk of breast cancer were the most widely discussed oucomes.1 Shortly after the findings from this arm of the study were published, the use of HT in the United States declined dramatically and unequivocally.2
In 2012, WHI published the results of the estrogen-alone arm in the British cancer specialty journal Lancet Oncology. As shown in the TABLE below, the incidence of breast cancer was statistically significantly lower (23%) in the estrogen group than in the placebo group. Women who were treated with estrogen alone were also 63% less likely to die of breast cancer, and all-cause mortality was 38% lower; both of these findings were statistically significant. Not only was there a significant reduction in the incidence of invasive breast cancer while the subjects were taking estrogen, but that reduction continued for a median of 4.7 years of follow-up after discontinuation of estrogen.
Breast cancer incidence and mortality in the estrogen-only arm of the WHI, compared with placebo*
| Event | Estrogen only (n = 5,310) | Placebo (n = 5,429) | Hazard ratio (95% confidence interval) |
|---|---|---|---|
| Invasive breast cancer | 151 (0.27%) | 199 (0.35%) | 0.77 (0.62–0.95) |
| Node-negative breast cancer | 88 (0.16%) | 134 (0.24%) | 0.67 (0.51–0.88) |
| Breast cancer mortality | 6 (0.009%) | 16 (0.024%) | 0.37 (0.13–0.91) |
| All-cause mortality | 30 (0.046%) | 50 (0.076%) | 0.62 (0.39–0.97) |
| * Median follow-up of 11.8 years | |||
The incidence figure is somewhat remarkable (199 in the placebo group versus 151 in the estrogen-alone group) in that it was nearly the exact reverse of the estrogen-progestin arm of the WHI trial (199 in the estrogen/progestin group vs 150 in the placebo group).3
Estrogen alone reduced both breast cancer incidence and breast cancer mortality while women were on therapy and for 5 years after discontinuing therapy. This finding should reassure women who have undergone hysterectomy, as well as their clinicians, that estrogen alone reduces the future likelihood of breast cancer. It should be noted that the effect of estrogen alone in women in higher-risk categories did not show a reduction in breast cancer, and for this reason, the authors cautioned against considering the use of estrogen alone in menopausal women as a breast cancer chemoprophylaxis agent.
All breast cancer chemoprophylactic agents carry risks as well as benefits
Goss P, Ingle J, Ales-Martinez J, et al. Exemestane for breast-cancer prevention in postmenopausal women. N Engl J Med. 2011;364(25):2381–2391.
Cheung A, Tile L, Cardew S, et al. Bone density and structure in healthy postmenopausal women treated with exemestane for the primary prevention of breast cancer: a nested substudy of the MAP.3 randomized controlled trial. Lancet Oncol. 2012;13(3):275–284.
Vogel V, Costantino J, Wickerham L, et al. Update of the National Surgical Adjuvant Breast and Bowel Project Study of Tamoxifen and Raloxifene (STAR) P-2 Trial: preventing breast cancer. Cancer Prev Res. 2010;3(6):696–706.
The number of new cases of breast cancer in the United States last year reached nearly a quarter-million. Clearly, reducing this number remains an important goal.4 Chemoprevention—the use of medication to reduce cancer risk—may be offered to women who are at high risk of developing breast cancer.
In the National Surgical Adjuvant Breast and Bowel Project (NSABP) P-1 trial, tamoxifen (a selective estrogen-receptor modulator) was shown to reduce the risk of invasive breast cancer by 49% in a high-risk population, resulting in the FDA approving tamoxifen as the first drug for breast cancer prevention.5 The P-1 trial was followed by the NSABP Study of Tamoxifen and Raloxifene (STAR) P-2 trial, which demonstrated relative equivalence between the two medications as cancer prevention agents in menopausal women.6 Serious side effects of these medications limit their use among eligible women, although raloxifene seems to be associated with fewer adverse events. In the update of the STAR trial with an average of 81 months of follow-up, the risk ratio for adverse events (raloxifene:tamoxifen) was 0.75 for thromboembolic events, 0.55 for endometrial cancer, and 0.19 for uterine hyperplasia.
Another drug used for cancer treatment has now entered the prevention scene. In 2011, the NCIC Clinical Trials Group Mammary Prevention.3 trial (NCIC CTG MAP.3) compared exemestane (an aromatase inhibitor) with placebo for menopausal women at high risk for breast cancer, demonstrating a 65% relative reduction in the incidence of invasive breast cancer. This study validated another option for cancer prevention in high-risk women, although its adoption is likely also to be limited by side effects, including vasomotor symptoms, a high rate of arthralgias, and vaginal dryness/dyspareunia. The greatest concern may be the potential effect on bone density. Though the rates of serious adverse events including fracture did not differ in the MAP.3 trial at 35 months of follow-up, women on exemestane had significantly larger losses of bone mineral density, compared with controls.
Chemoprophylaxis reduces the risk of breast cancer in high-risk women by about 50%. Who are good candidates for these medications? Based on these trials, menopausal women considered at high risk might include those with a Gail risk score of at least 1.66% (ie, risk of developing breast cancer in 5 years), age 60 years or older, and women with biopsy results demonstrating atypical hyperplasia or lobular carcinoma in situ (LCIS). (The Gail model is available at www.cancer.gov/bcrisktool.) Tamoxifen is the only option for premenopausal women age 35 and older. Those who have histologic markers of risk (atypical hyperplasia, LCIS) likely stand to derive the greatest benefit.4
Managing the reproductive health concerns of young women with breast cancer
Azim H, Kroman N, Paesmans M, et al. Prognostic impact of pregnancy after breast cancer according to estrogen receptor status: a multicenter retrospective study. J Clin Oncol. 2013;31(1):73–79.
Howard-Anderson J, Ganz P, Bower J, Stanton A. Quality of life, fertility concerns, and behavioral health outcomes in younger breast cancer survivors: a systematic review. J Natl Cancer Inst. 2012;104(5):1386–1405.
Of the approximately 230,000 new cases of invasive breast cancer identified in 2011, 50,430 cases involved women less than 50 years of age.4 For these women, the diagnosis of cancer raises multifaceted concerns, including the physical changes that accompany breast cancer treatment, concerns about recurrence and mortality, and significant sexual and reproductive consequences of treatment that alters ovarian function. Pregnancy-associated breast cancers (breast cancers diagnosed during pregnancy, lactation, and for 12 months postpartum) represent a small subset of these cancers and occur in about 1 in 3,000 pregnancies. One might anticipate that this rate will increase as women continue to delay childbearing, because pregnancy-associated breast cancers are more common in older women.
In the review article by Howard-Anderson and colleagues, the importance of these reproductive health consequences in young women diagnosed with breast cancer is highlighted. The women who transition to menopause as a result of chemotherapy (reported to range from 33%–73%) experience more symptoms, including hot flashes, night sweats, breast pain, vaginal dryness, and lack of sexual desire. Sixty-one percent of women younger than 40 years at diagnosis reported that they were concerned about menopause, and 30% reported that this concern influenced their treatment decisions. Thirty-nine percent of women in this group had major concerns about treatment-associated infertility, and only half of the women studied felt that their fertility concerns were adequately addressed.
On a positive note, for women who successfully achieve pregnancy after breast cancer, pregnancy outcomes appear to be similar to those of their nonpregnant peers. In the study by Azim and colleagues, women who became pregnant after a breast cancer diagnosis had disease-free survival that was statistically similar to that of matched women who did not have subsequent pregnancies. In addition, this outcome did not differ based on estrogen/progesterone receptor status (ER/PR positive or negative).
Both alkylating chemotherapeutic agents (eg, cyclophosphamide) and selective estrogen receptor modulating agents (for women with estrogen-receptor–positive tumors) are routine parts of adjuvant treatment for premenopausal women with invasive breast cancers.
These agents can have profound effects on both ovarian hormonal function and fertility. ObGyns and reproductive endocrinology/infertility specialists have a great opportunity to partner with our oncology colleagues to enhance the counseling that young women receive before, during, and after breast cancer treatment.
Women who are considering future childbearing should receive information about the impact of breast cancer treatment on fertility and options for fertility preservation prior to initiating treatment. For women who have completed childbearing, information on what to expect if menopause occurs and available options for symptom relief can be empowering as they make treatment decisions.
We want to hear from you! Tell us what you think.
Women with ER-positive breast cancer may soon extend tamoxifen therapy to 10 years
Janelle Yates (February 2013)
Is overdiagnosis of breast cancer common among women screened
by mammography?
Andrew M. Kaunitz, MD (Examining the Evidence; January 2013)
Breast cancer genome analysis highlights 4 subtypes, link to
ovarian cancer
Janelle Yates (News for Your Practice; November 2012)
The effects of breast cancer on obstetric and gynecologic practices are pervasive. In this article, we touch on three aspects of breast cancer that are particularly relevant to the practicing ObGyn:
- the need to identify women at high risk for breast cancer and select those who would benefit from a discussion of the advantages and risks of chemoprophylaxis, which can reduce the likelihood of breast cancer by 50% or more
- the need for strategies to manage menopausal symptoms in the general population without increasing the risk of breast cancer. The traditional approach to this problem changed dramatically with the Women’s Health Initiative (WHI), which demonstrated an increased risk of breast cancer in women taking conjugated equine estrogen and progestin. The widely publicized initial findings of the estrogen-progestin arm of the WHI sharply contrast the equally relevant, somewhat unexpected, and less publicized results of the estrogen-alone arm, which demonstrated a substantial and statistically significant decrease in the incidence of breast cancer, even after estrogen was discontinued.
- the potential effects of breast cancer treatment on ovarian function in young women. This year, of the approximately 250,000 women who will be diagnosed with invasive breast cancer, more than 50,000 women will be of reproductive age. Most of these young women will require adjuvant chemotherapy; as a result, many will experience the premature onset of menopause. Along with the attendant loss of fertility these women will face, many will also develop distressing and life-altering menopausal symptoms. Management of these women before and after initiation of chemotherapy requires an understanding of both the expected effects of the chemotherapy and knowledge of how to actively manage these women with strategies to either prevent these events or to manage menopausal symptoms.

In women at normal risk for breast cancer, unopposed estrogen lowers the rate of the malignancy and the likelihood of mortality if the cancer occurs—but is not recommended as a prophylactic agent. Tamoxifen and other chemoprophylactic drugs can halve the rate of breast cancer in high-risk women but are not without drawbacks.
A look at the lower rate of breast cancer in the estrogen-alone arm of the WHI
Anderson GL, Chlebowski RT, Aragaki AK, et al. Conjugated equine oestrogen and breast cancer incidence and mortality in postmenopausal women with hysterectomy: extended follow-up of the Women’s Health Initiative randomised placebo-controlled trial. Lancet Oncol. 2012;13(5):476–486.
From 1993 through 1998, the WHI enrolled 10,739 postmenopausal women in the largest prospective trial evaluating the effect of hormone therapy (HT) on various clinical outcomes. The women were randomly allocated to three groups:
- conjugated estrogen with medroxyprogesterone acetate
- conjugated estrogen alone (in women with a prior hysterectomy)
- placebo.
The negative effects of estrogen plus progestin on the risk of breast cancer were the most widely discussed oucomes.1 Shortly after the findings from this arm of the study were published, the use of HT in the United States declined dramatically and unequivocally.2
In 2012, WHI published the results of the estrogen-alone arm in the British cancer specialty journal Lancet Oncology. As shown in the TABLE below, the incidence of breast cancer was statistically significantly lower (23%) in the estrogen group than in the placebo group. Women who were treated with estrogen alone were also 63% less likely to die of breast cancer, and all-cause mortality was 38% lower; both of these findings were statistically significant. Not only was there a significant reduction in the incidence of invasive breast cancer while the subjects were taking estrogen, but that reduction continued for a median of 4.7 years of follow-up after discontinuation of estrogen.
Breast cancer incidence and mortality in the estrogen-only arm of the WHI, compared with placebo*
| Event | Estrogen only (n = 5,310) | Placebo (n = 5,429) | Hazard ratio (95% confidence interval) |
|---|---|---|---|
| Invasive breast cancer | 151 (0.27%) | 199 (0.35%) | 0.77 (0.62–0.95) |
| Node-negative breast cancer | 88 (0.16%) | 134 (0.24%) | 0.67 (0.51–0.88) |
| Breast cancer mortality | 6 (0.009%) | 16 (0.024%) | 0.37 (0.13–0.91) |
| All-cause mortality | 30 (0.046%) | 50 (0.076%) | 0.62 (0.39–0.97) |
| * Median follow-up of 11.8 years | |||
The incidence figure is somewhat remarkable (199 in the placebo group versus 151 in the estrogen-alone group) in that it was nearly the exact reverse of the estrogen-progestin arm of the WHI trial (199 in the estrogen/progestin group vs 150 in the placebo group).3
Estrogen alone reduced both breast cancer incidence and breast cancer mortality while women were on therapy and for 5 years after discontinuing therapy. This finding should reassure women who have undergone hysterectomy, as well as their clinicians, that estrogen alone reduces the future likelihood of breast cancer. It should be noted that the effect of estrogen alone in women in higher-risk categories did not show a reduction in breast cancer, and for this reason, the authors cautioned against considering the use of estrogen alone in menopausal women as a breast cancer chemoprophylaxis agent.
All breast cancer chemoprophylactic agents carry risks as well as benefits
Goss P, Ingle J, Ales-Martinez J, et al. Exemestane for breast-cancer prevention in postmenopausal women. N Engl J Med. 2011;364(25):2381–2391.
Cheung A, Tile L, Cardew S, et al. Bone density and structure in healthy postmenopausal women treated with exemestane for the primary prevention of breast cancer: a nested substudy of the MAP.3 randomized controlled trial. Lancet Oncol. 2012;13(3):275–284.
Vogel V, Costantino J, Wickerham L, et al. Update of the National Surgical Adjuvant Breast and Bowel Project Study of Tamoxifen and Raloxifene (STAR) P-2 Trial: preventing breast cancer. Cancer Prev Res. 2010;3(6):696–706.
The number of new cases of breast cancer in the United States last year reached nearly a quarter-million. Clearly, reducing this number remains an important goal.4 Chemoprevention—the use of medication to reduce cancer risk—may be offered to women who are at high risk of developing breast cancer.
In the National Surgical Adjuvant Breast and Bowel Project (NSABP) P-1 trial, tamoxifen (a selective estrogen-receptor modulator) was shown to reduce the risk of invasive breast cancer by 49% in a high-risk population, resulting in the FDA approving tamoxifen as the first drug for breast cancer prevention.5 The P-1 trial was followed by the NSABP Study of Tamoxifen and Raloxifene (STAR) P-2 trial, which demonstrated relative equivalence between the two medications as cancer prevention agents in menopausal women.6 Serious side effects of these medications limit their use among eligible women, although raloxifene seems to be associated with fewer adverse events. In the update of the STAR trial with an average of 81 months of follow-up, the risk ratio for adverse events (raloxifene:tamoxifen) was 0.75 for thromboembolic events, 0.55 for endometrial cancer, and 0.19 for uterine hyperplasia.
Another drug used for cancer treatment has now entered the prevention scene. In 2011, the NCIC Clinical Trials Group Mammary Prevention.3 trial (NCIC CTG MAP.3) compared exemestane (an aromatase inhibitor) with placebo for menopausal women at high risk for breast cancer, demonstrating a 65% relative reduction in the incidence of invasive breast cancer. This study validated another option for cancer prevention in high-risk women, although its adoption is likely also to be limited by side effects, including vasomotor symptoms, a high rate of arthralgias, and vaginal dryness/dyspareunia. The greatest concern may be the potential effect on bone density. Though the rates of serious adverse events including fracture did not differ in the MAP.3 trial at 35 months of follow-up, women on exemestane had significantly larger losses of bone mineral density, compared with controls.
Chemoprophylaxis reduces the risk of breast cancer in high-risk women by about 50%. Who are good candidates for these medications? Based on these trials, menopausal women considered at high risk might include those with a Gail risk score of at least 1.66% (ie, risk of developing breast cancer in 5 years), age 60 years or older, and women with biopsy results demonstrating atypical hyperplasia or lobular carcinoma in situ (LCIS). (The Gail model is available at www.cancer.gov/bcrisktool.) Tamoxifen is the only option for premenopausal women age 35 and older. Those who have histologic markers of risk (atypical hyperplasia, LCIS) likely stand to derive the greatest benefit.4
Managing the reproductive health concerns of young women with breast cancer
Azim H, Kroman N, Paesmans M, et al. Prognostic impact of pregnancy after breast cancer according to estrogen receptor status: a multicenter retrospective study. J Clin Oncol. 2013;31(1):73–79.
Howard-Anderson J, Ganz P, Bower J, Stanton A. Quality of life, fertility concerns, and behavioral health outcomes in younger breast cancer survivors: a systematic review. J Natl Cancer Inst. 2012;104(5):1386–1405.
Of the approximately 230,000 new cases of invasive breast cancer identified in 2011, 50,430 cases involved women less than 50 years of age.4 For these women, the diagnosis of cancer raises multifaceted concerns, including the physical changes that accompany breast cancer treatment, concerns about recurrence and mortality, and significant sexual and reproductive consequences of treatment that alters ovarian function. Pregnancy-associated breast cancers (breast cancers diagnosed during pregnancy, lactation, and for 12 months postpartum) represent a small subset of these cancers and occur in about 1 in 3,000 pregnancies. One might anticipate that this rate will increase as women continue to delay childbearing, because pregnancy-associated breast cancers are more common in older women.
In the review article by Howard-Anderson and colleagues, the importance of these reproductive health consequences in young women diagnosed with breast cancer is highlighted. The women who transition to menopause as a result of chemotherapy (reported to range from 33%–73%) experience more symptoms, including hot flashes, night sweats, breast pain, vaginal dryness, and lack of sexual desire. Sixty-one percent of women younger than 40 years at diagnosis reported that they were concerned about menopause, and 30% reported that this concern influenced their treatment decisions. Thirty-nine percent of women in this group had major concerns about treatment-associated infertility, and only half of the women studied felt that their fertility concerns were adequately addressed.
On a positive note, for women who successfully achieve pregnancy after breast cancer, pregnancy outcomes appear to be similar to those of their nonpregnant peers. In the study by Azim and colleagues, women who became pregnant after a breast cancer diagnosis had disease-free survival that was statistically similar to that of matched women who did not have subsequent pregnancies. In addition, this outcome did not differ based on estrogen/progesterone receptor status (ER/PR positive or negative).
Both alkylating chemotherapeutic agents (eg, cyclophosphamide) and selective estrogen receptor modulating agents (for women with estrogen-receptor–positive tumors) are routine parts of adjuvant treatment for premenopausal women with invasive breast cancers.
These agents can have profound effects on both ovarian hormonal function and fertility. ObGyns and reproductive endocrinology/infertility specialists have a great opportunity to partner with our oncology colleagues to enhance the counseling that young women receive before, during, and after breast cancer treatment.
Women who are considering future childbearing should receive information about the impact of breast cancer treatment on fertility and options for fertility preservation prior to initiating treatment. For women who have completed childbearing, information on what to expect if menopause occurs and available options for symptom relief can be empowering as they make treatment decisions.
We want to hear from you! Tell us what you think.
1. Grady D. Study finds new risks in hormone therapy. New York Times. http://www.nytimes.com/2003/06/25/us/study-finds-new-risks-in-hormone-therapy.html?pagewanted=all&src=pm. Published June 25 2003. Accessed February 11, 2013.
2. Hersh AL, Stefanick ML, Stafford RS. National use of menopausal hormone therapy: annual trends and response to recent evidence. JAMA. 2004;291(1):47-53.
3. Chlebowski RT, Kuller LH, Prentice RL, et al. Women’s Health Initiative Investigators. Breast cancer after use of estrogen plus progestin in postmenopausal women. N Engl J Med. 2009;360(6):573-587.
4. American Cancer Society. Breast Cancer Facts and Figures 2011-2012. Atlanta, GA: American Cancer Society. http://www.cancer.org/research/cancerfactsfigures/breastcancerfactsfigures/breast-cancer-facts-and-figures-2011-2012. Accessed February 11, 2013.
5. Fisher B, Constantino J, Wickerham L, et al. Tamoxifen for prevention of breast cancer: report of the National Surgical Adjuvant Breast and Bowel Project P-1 Study. J Natl Cancer Inst. 1998;90(18):1371-1388.
6. Vogel V, Costantino J, Wickerham DL, et al. Effects of tamoxifen vs raloxifene on the risk of developing invasive breast cancer and other disease outcomes. The NSABP Study of Tamoxifen and Raloxifene (STAR) P-2 Trial. JAMA. 2006;295(23):2727-2741.
1. Grady D. Study finds new risks in hormone therapy. New York Times. http://www.nytimes.com/2003/06/25/us/study-finds-new-risks-in-hormone-therapy.html?pagewanted=all&src=pm. Published June 25 2003. Accessed February 11, 2013.
2. Hersh AL, Stefanick ML, Stafford RS. National use of menopausal hormone therapy: annual trends and response to recent evidence. JAMA. 2004;291(1):47-53.
3. Chlebowski RT, Kuller LH, Prentice RL, et al. Women’s Health Initiative Investigators. Breast cancer after use of estrogen plus progestin in postmenopausal women. N Engl J Med. 2009;360(6):573-587.
4. American Cancer Society. Breast Cancer Facts and Figures 2011-2012. Atlanta, GA: American Cancer Society. http://www.cancer.org/research/cancerfactsfigures/breastcancerfactsfigures/breast-cancer-facts-and-figures-2011-2012. Accessed February 11, 2013.
5. Fisher B, Constantino J, Wickerham L, et al. Tamoxifen for prevention of breast cancer: report of the National Surgical Adjuvant Breast and Bowel Project P-1 Study. J Natl Cancer Inst. 1998;90(18):1371-1388.
6. Vogel V, Costantino J, Wickerham DL, et al. Effects of tamoxifen vs raloxifene on the risk of developing invasive breast cancer and other disease outcomes. The NSABP Study of Tamoxifen and Raloxifene (STAR) P-2 Trial. JAMA. 2006;295(23):2727-2741.
When is her pelvic pressure and bulge due to Pouch of Douglas hernia?
CASE: Pelvic organ prolapse or Pouch of Douglas hernia?
A 42-year-old G3P2 woman is referred to you by her primary care provider for pelvic organ prolapse. Her medical history reveals that she has been bothered by a sense of pelvic pressure and bulge progressing over several years, and she has noticed that her symptoms are particularly worse during and after bowel movements. She reports some improved bowel evacuation with external splinting of her perineum. Upon closer questioning, the patient reports a history of chronic constipation since childhood associated with straining and a sense of incomplete emptying. She reports spending up to 30 minutes three to four times per day on the commode to completely empty her bowels.
Physical examination reveals an overweight woman with a soft, nontender abdomen remarkable for laparoscopic incision scars from a previous tubal ligation. Inspection of the external genitalia at rest is normal. Cough stress test is negative. At maximum Valsalva, however, there is significant perineal ballooning present.
Speculum examination demonstrates grade 1 uterine prolapse, grade 1 cystocele, and grade 2 rectocele. There is no evidence of pelvic floor tension myalgia. She has weak pelvic muscle strength. Visualization of the anus at maximum Valsalva reveals there is some asymmetric rectal prolapse of the anterior rectal wall. Digital rectal exam is unremarkable.
Are these patient’s symptoms due to pelvic organ prolapse or Pouch of Douglas hernia?
Pelvic organ prolapse: A common problem
Pelvic organ prolapse has an estimated prevalence of 55% in women aged 50 to 59 years.1 More than 200,000 pelvic organ prolapse surgeries are performed annually in the United States.2 Typically, patients report:
- vaginal bulge causing discomfort
- pelvic pressure or heaviness, or
- rubbing of the vaginal bulge on undergarments.
In more advanced pelvic organ prolapse, patients may report voiding dysfunction or stool trapping that requires manual splinting of the prolapse to assist in bladder and bowel evacuation.
Pouch of Douglas hernia: A lesser-known
(recognized) phenomenon
Similar to pelvic organ prolapse, Pouch of Douglas hernia also can present with symptoms of:
- pelvic pressure
- vague perineal aching
- defecatory dysfunction.
The phenomenon has been variably referred to in the literature as enterocele, descending perineum syndrome, peritoneocele, or Pouch of Douglas hernia. The concept was first introduced in 19663 and describes descent of the entire pelvic floor and small bowel through a hernia in the Pouch of Douglas (FIGURE 1).
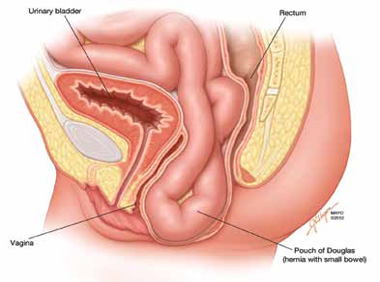
FIGURE 1: Pouch of Douglas hernia. The pelvic floor and small bowel descend into the Pouch of Douglas.
How does it occur? The pathophysiology is thought to be related to excessive abdominal straining in individuals with chronic constipation. This results in diminished pelvic floor muscle tone. Eventually, the whole pelvic floor descends, becoming funnel shaped due to stretching of the puborectalis muscle. Thus, stool is expelled by force, mostly through forces on the anterior rectal wall (which tends to prolapse after stool evacuation, with accompanied mucus secretion, soreness, and irritation).
Clinical pearl: Given the rectal wall prolapse that occurs after stool evacuation in Pouch of Douglas hernia, some patients will describe a rectal lump that bleeds after a bowel movement. The sensation of the rectal lump from the anterior rectal wall prolapse causes further straining.
Your patient reports pelvic pressure and bulge.
How do you proceed?
Physical examination
Look for perineal ballooning. Physical examination should start with inspection of the external genitalia. This inspection will identify any pelvic organ prolapse at or beyond the introitus. However, a Pouch of Douglas hernia will be missed if the patient is not examined during Valsalva or maximal strain. This maneuver will demonstrate the classic finding of perineal ballooning and is crucial to a final diagnosis of Pouch of Douglas hernia. Normally, the perineum will descend 1 cm to 2 cm during maximal strain; in Pouch of Douglas hernias, the perineum can descend up to 4 cm to 8 cm.4
Clinical pearl: It should be noted that, often, patients will not have a great deal of vaginal prolapse accompanying the perineal ballooning. In our opinion, this finding distinguishes Pouch of Douglas hernia from a vaginal vault prolapse caused by an enterocele.
Is rectal prolapse present? Beyond perineal ballooning, the presence of rectal prolapse should be evaluated. A rectocele of some degree is usually present. Asymmetric rectal prolapse affecting the anterior aspect of the rectal wall is consistent with a Pouch of Douglas hernia. This anatomic finding should be distinguished from true circumferential rectal prolapse, which remains in the differential diagnosis.
Basing the diagnosis of Pouch of Douglas hernia on physical examination alone can be difficult. Therefore, imaging studies are essential for accurate diagnosis.
Imaging investigations
Several imaging modalities can be used to diagnose such disorders of the pelvic floor as Pouch of Douglas hernia. These include:
- dynamic colpocystoproctography5
- defecography with oral barium6
- dynamic pelvic magnetic resonance imaging (MRI).7
In our experience, dynamic pelvic MRI has a high accuracy rate for diagnosing Pouch of Douglas hernia. FIGURE 2 illustrates the large Pouch of Douglas hernia filled with loops of small bowel. Perineal descent of the anorectal junction more than 3 cm below the pubococcygeal line during maximal straining is a diagnostic finding on imaging.7
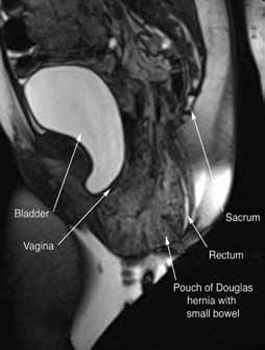
FIGURE 2: MRI
Sagittal MRI during maximal Valsalva straining, demonstrating Pouch of Douglas hernia filled with small bowel.
What are your patient’s treatment options?
Reduce straining during bowel movements. The primary goal of treatment for Pouch of Douglas hernia should be relief of bothersome symptoms. Therefore, further damage can be prevented by eliminating straining during defecation. This can be accomplished with a bowel regimen that combines an irritant suppository (glycerin or bisacodyl) with a fiber supplement (the latter to increase bulk of the stool). Oral laxatives have limited use as many patients have lax anal sphincters and liquid stool could cause fecal incontinence.
Pelvic floor strengthening. The importance of pelvic floor physical therapy should be stressed. Patients can benefit from the use of modalities such as biofeedback to learn appropriate pelvic floor muscle relaxation techniques during defecation.8 While there is limited published evidence supporting the use of pelvic floor physical therapy, our anecdotal experience suggests that patients can gain considerable benefit with such conservative therapy.
Surgical therapy
Surgical repair of Pouch of Douglas hernia requires obliteration of the deep cul-de-sac (to prevent the small bowel from filling this space) and simultaneous pelvic floor reconstruction of the vaginal apex and any other compartments that are prolapsing (if pelvic organ prolapse is present). In our experience, these patients typically have derived greatest benefit from an abdominal approach. This usually can be accomplished with a sacrocolpopexy (if vaginal vault prolapse exists) with a Moschowitz or Halban procedure,9 uterosacral ligament plication, or a modified sacrocolpopexy with mesh augmentation to the sidewalls of the pelvis.10 There are currently no studies supporting one particular approach over another, but the most important feature of a surgical intervention is obliteration of the cul-de-sac (FIGURES 3, 4, and 5).
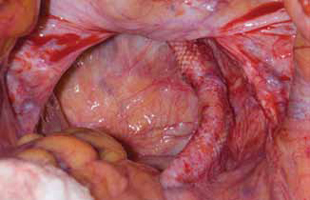
FIGURE 3: Open cul-de-sac. Open cul-de-sac after a prior abdominal sacrocolpopexy in a patient with a Pouch of Douglas hernia.
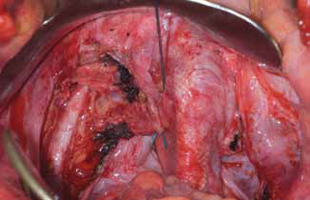
FIGURE 4: Obliterated cul-de-sac. Obliteration of the cul-de-sac with uterosacral ligament plication. Care is taken to prevent obstruction of the rectum at this level.
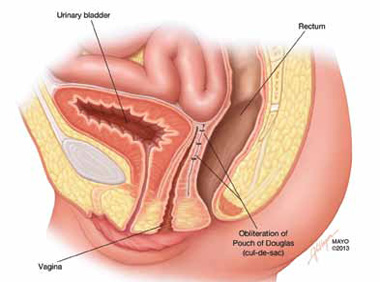
FIGURE 5: Cul-de-sac obliteration. Schematic diagram of obliteration of the cul-de-sac with uterosacral ligament plication sutures.
Final takeaways
Pouch of Douglas hernia is an important but often unrecognized cause of pelvic pressure and defecatory dysfunction. Perineal ballooning during maximal straining is highly suggestive of the diagnosis, with final diagnosis confirmed with various functional imaging studies of the pelvic floor. Management should include both conservative and surgical interventions to alleviate and prevent recurrence of symptoms.
ACKNOWLEDGMENT. The authors would like to thank Mr. John Hagen, Medical Illustrator, Mayo Clinic, for producing the illustrations in Figures 1 and 5.
We want to hear from you! Tell us what you think.
Urinary incontinence
Karen L. Noblett, MD, MAS, and Stephanie A. Jacobs, MD (Update, December 2012)
When and how to place an autologous rectus fascia
pubovaginal sling
Mickey Karram, MD, and Dani Zoorob, MD (Surgical Techniques, November 2012)
Pelvic floor dysfunction
Autumn L. Edenfield, MD, and Cindy L. Amundsen, MD (Update, October 2012)
Step by step: Obliterating the vaginal canal to correct pelvic organ prolapse
Mickey Karram, MD, and Janelle Evans, MD (Surgical Techniques, February 2012)
1. Samuelsson EC, Victor FT, Tibblin G, Svärdsudd KF. Signs of genital prolapse in a Swedish population of women 20 to 59 years of age and possible related factors. Am J Obstet Gynecol. 1999;180(2 Pt 1):299-305.
2. Boyles SH, Weber AM, Meyn L. Procedures for pelvic organ prolapse in the United States 1979-1997. Am J Obstet Gynecol. 2003;188(1):108-115.
3. Parks AG, Porter NH, Hardcastle J. The syndrome of the descending perineum. Proc R Soc Med. 1966;59(6):477-482.
4. Hardcastle JD. The descending perineum syndrome. Practitioner. 1969;203(217):612-619.
5. Maglinte DD, Bartram CI, Hale DA, et al. Functional imaging of the pelvic floor. Radiology. 2011;258(1):23-39.
6. Roos JE, Weishaupt D, Wildermuth S, Willmann JK, Marincek B, Hilfiker PR. Experience of 4 years with open MR defecography: pictorial review of anorectal anatomy and disease. Radiographics. 2002;22(4):817-832.
7. Fletcher JG, Busse RF, Riederer SJ, et al. Magnetic resonance imaging of anatomic and dynamic defects of the pelvic floor in defecatory disorders. Am J Gastroenterol. 2003;98(2):399-411.
8. Harewood GC, Coulie B, Camilleri M, Rath-Harvey D, Pemberton JH. Descending perineum syndrome: audit of clinical and laboratory features and outcome of pelvic floor retraining. Am J Gastroenterol. 1999;94(1):126-130.
9. Moschcowitz AV. The pathogenesis anatomy and cure of prolapse of the rectum. Surg Gyncol Obstetrics. 1912;15:7-21.
10. Gosselink MJ, van Dam JH, Huisman WM, Ginai AZ, Schouten WR. Treatment of enterocele by obliteration of the pelvic inlet. Dis Colon Rectum. 1999;42(7):940-944.
CASE: Pelvic organ prolapse or Pouch of Douglas hernia?
A 42-year-old G3P2 woman is referred to you by her primary care provider for pelvic organ prolapse. Her medical history reveals that she has been bothered by a sense of pelvic pressure and bulge progressing over several years, and she has noticed that her symptoms are particularly worse during and after bowel movements. She reports some improved bowel evacuation with external splinting of her perineum. Upon closer questioning, the patient reports a history of chronic constipation since childhood associated with straining and a sense of incomplete emptying. She reports spending up to 30 minutes three to four times per day on the commode to completely empty her bowels.
Physical examination reveals an overweight woman with a soft, nontender abdomen remarkable for laparoscopic incision scars from a previous tubal ligation. Inspection of the external genitalia at rest is normal. Cough stress test is negative. At maximum Valsalva, however, there is significant perineal ballooning present.
Speculum examination demonstrates grade 1 uterine prolapse, grade 1 cystocele, and grade 2 rectocele. There is no evidence of pelvic floor tension myalgia. She has weak pelvic muscle strength. Visualization of the anus at maximum Valsalva reveals there is some asymmetric rectal prolapse of the anterior rectal wall. Digital rectal exam is unremarkable.
Are these patient’s symptoms due to pelvic organ prolapse or Pouch of Douglas hernia?
Pelvic organ prolapse: A common problem
Pelvic organ prolapse has an estimated prevalence of 55% in women aged 50 to 59 years.1 More than 200,000 pelvic organ prolapse surgeries are performed annually in the United States.2 Typically, patients report:
- vaginal bulge causing discomfort
- pelvic pressure or heaviness, or
- rubbing of the vaginal bulge on undergarments.
In more advanced pelvic organ prolapse, patients may report voiding dysfunction or stool trapping that requires manual splinting of the prolapse to assist in bladder and bowel evacuation.
Pouch of Douglas hernia: A lesser-known
(recognized) phenomenon
Similar to pelvic organ prolapse, Pouch of Douglas hernia also can present with symptoms of:
- pelvic pressure
- vague perineal aching
- defecatory dysfunction.
The phenomenon has been variably referred to in the literature as enterocele, descending perineum syndrome, peritoneocele, or Pouch of Douglas hernia. The concept was first introduced in 19663 and describes descent of the entire pelvic floor and small bowel through a hernia in the Pouch of Douglas (FIGURE 1).

FIGURE 1: Pouch of Douglas hernia. The pelvic floor and small bowel descend into the Pouch of Douglas.
How does it occur? The pathophysiology is thought to be related to excessive abdominal straining in individuals with chronic constipation. This results in diminished pelvic floor muscle tone. Eventually, the whole pelvic floor descends, becoming funnel shaped due to stretching of the puborectalis muscle. Thus, stool is expelled by force, mostly through forces on the anterior rectal wall (which tends to prolapse after stool evacuation, with accompanied mucus secretion, soreness, and irritation).
Clinical pearl: Given the rectal wall prolapse that occurs after stool evacuation in Pouch of Douglas hernia, some patients will describe a rectal lump that bleeds after a bowel movement. The sensation of the rectal lump from the anterior rectal wall prolapse causes further straining.
Your patient reports pelvic pressure and bulge.
How do you proceed?
Physical examination
Look for perineal ballooning. Physical examination should start with inspection of the external genitalia. This inspection will identify any pelvic organ prolapse at or beyond the introitus. However, a Pouch of Douglas hernia will be missed if the patient is not examined during Valsalva or maximal strain. This maneuver will demonstrate the classic finding of perineal ballooning and is crucial to a final diagnosis of Pouch of Douglas hernia. Normally, the perineum will descend 1 cm to 2 cm during maximal strain; in Pouch of Douglas hernias, the perineum can descend up to 4 cm to 8 cm.4
Clinical pearl: It should be noted that, often, patients will not have a great deal of vaginal prolapse accompanying the perineal ballooning. In our opinion, this finding distinguishes Pouch of Douglas hernia from a vaginal vault prolapse caused by an enterocele.
Is rectal prolapse present? Beyond perineal ballooning, the presence of rectal prolapse should be evaluated. A rectocele of some degree is usually present. Asymmetric rectal prolapse affecting the anterior aspect of the rectal wall is consistent with a Pouch of Douglas hernia. This anatomic finding should be distinguished from true circumferential rectal prolapse, which remains in the differential diagnosis.
Basing the diagnosis of Pouch of Douglas hernia on physical examination alone can be difficult. Therefore, imaging studies are essential for accurate diagnosis.
Imaging investigations
Several imaging modalities can be used to diagnose such disorders of the pelvic floor as Pouch of Douglas hernia. These include:
- dynamic colpocystoproctography5
- defecography with oral barium6
- dynamic pelvic magnetic resonance imaging (MRI).7
In our experience, dynamic pelvic MRI has a high accuracy rate for diagnosing Pouch of Douglas hernia. FIGURE 2 illustrates the large Pouch of Douglas hernia filled with loops of small bowel. Perineal descent of the anorectal junction more than 3 cm below the pubococcygeal line during maximal straining is a diagnostic finding on imaging.7

FIGURE 2: MRI
Sagittal MRI during maximal Valsalva straining, demonstrating Pouch of Douglas hernia filled with small bowel.
What are your patient’s treatment options?
Reduce straining during bowel movements. The primary goal of treatment for Pouch of Douglas hernia should be relief of bothersome symptoms. Therefore, further damage can be prevented by eliminating straining during defecation. This can be accomplished with a bowel regimen that combines an irritant suppository (glycerin or bisacodyl) with a fiber supplement (the latter to increase bulk of the stool). Oral laxatives have limited use as many patients have lax anal sphincters and liquid stool could cause fecal incontinence.
Pelvic floor strengthening. The importance of pelvic floor physical therapy should be stressed. Patients can benefit from the use of modalities such as biofeedback to learn appropriate pelvic floor muscle relaxation techniques during defecation.8 While there is limited published evidence supporting the use of pelvic floor physical therapy, our anecdotal experience suggests that patients can gain considerable benefit with such conservative therapy.
Surgical therapy
Surgical repair of Pouch of Douglas hernia requires obliteration of the deep cul-de-sac (to prevent the small bowel from filling this space) and simultaneous pelvic floor reconstruction of the vaginal apex and any other compartments that are prolapsing (if pelvic organ prolapse is present). In our experience, these patients typically have derived greatest benefit from an abdominal approach. This usually can be accomplished with a sacrocolpopexy (if vaginal vault prolapse exists) with a Moschowitz or Halban procedure,9 uterosacral ligament plication, or a modified sacrocolpopexy with mesh augmentation to the sidewalls of the pelvis.10 There are currently no studies supporting one particular approach over another, but the most important feature of a surgical intervention is obliteration of the cul-de-sac (FIGURES 3, 4, and 5).

FIGURE 3: Open cul-de-sac. Open cul-de-sac after a prior abdominal sacrocolpopexy in a patient with a Pouch of Douglas hernia.

FIGURE 4: Obliterated cul-de-sac. Obliteration of the cul-de-sac with uterosacral ligament plication. Care is taken to prevent obstruction of the rectum at this level.

FIGURE 5: Cul-de-sac obliteration. Schematic diagram of obliteration of the cul-de-sac with uterosacral ligament plication sutures.
Final takeaways
Pouch of Douglas hernia is an important but often unrecognized cause of pelvic pressure and defecatory dysfunction. Perineal ballooning during maximal straining is highly suggestive of the diagnosis, with final diagnosis confirmed with various functional imaging studies of the pelvic floor. Management should include both conservative and surgical interventions to alleviate and prevent recurrence of symptoms.
ACKNOWLEDGMENT. The authors would like to thank Mr. John Hagen, Medical Illustrator, Mayo Clinic, for producing the illustrations in Figures 1 and 5.
We want to hear from you! Tell us what you think.
Urinary incontinence
Karen L. Noblett, MD, MAS, and Stephanie A. Jacobs, MD (Update, December 2012)
When and how to place an autologous rectus fascia
pubovaginal sling
Mickey Karram, MD, and Dani Zoorob, MD (Surgical Techniques, November 2012)
Pelvic floor dysfunction
Autumn L. Edenfield, MD, and Cindy L. Amundsen, MD (Update, October 2012)
Step by step: Obliterating the vaginal canal to correct pelvic organ prolapse
Mickey Karram, MD, and Janelle Evans, MD (Surgical Techniques, February 2012)
CASE: Pelvic organ prolapse or Pouch of Douglas hernia?
A 42-year-old G3P2 woman is referred to you by her primary care provider for pelvic organ prolapse. Her medical history reveals that she has been bothered by a sense of pelvic pressure and bulge progressing over several years, and she has noticed that her symptoms are particularly worse during and after bowel movements. She reports some improved bowel evacuation with external splinting of her perineum. Upon closer questioning, the patient reports a history of chronic constipation since childhood associated with straining and a sense of incomplete emptying. She reports spending up to 30 minutes three to four times per day on the commode to completely empty her bowels.
Physical examination reveals an overweight woman with a soft, nontender abdomen remarkable for laparoscopic incision scars from a previous tubal ligation. Inspection of the external genitalia at rest is normal. Cough stress test is negative. At maximum Valsalva, however, there is significant perineal ballooning present.
Speculum examination demonstrates grade 1 uterine prolapse, grade 1 cystocele, and grade 2 rectocele. There is no evidence of pelvic floor tension myalgia. She has weak pelvic muscle strength. Visualization of the anus at maximum Valsalva reveals there is some asymmetric rectal prolapse of the anterior rectal wall. Digital rectal exam is unremarkable.
Are these patient’s symptoms due to pelvic organ prolapse or Pouch of Douglas hernia?
Pelvic organ prolapse: A common problem
Pelvic organ prolapse has an estimated prevalence of 55% in women aged 50 to 59 years.1 More than 200,000 pelvic organ prolapse surgeries are performed annually in the United States.2 Typically, patients report:
- vaginal bulge causing discomfort
- pelvic pressure or heaviness, or
- rubbing of the vaginal bulge on undergarments.
In more advanced pelvic organ prolapse, patients may report voiding dysfunction or stool trapping that requires manual splinting of the prolapse to assist in bladder and bowel evacuation.
Pouch of Douglas hernia: A lesser-known
(recognized) phenomenon
Similar to pelvic organ prolapse, Pouch of Douglas hernia also can present with symptoms of:
- pelvic pressure
- vague perineal aching
- defecatory dysfunction.
The phenomenon has been variably referred to in the literature as enterocele, descending perineum syndrome, peritoneocele, or Pouch of Douglas hernia. The concept was first introduced in 19663 and describes descent of the entire pelvic floor and small bowel through a hernia in the Pouch of Douglas (FIGURE 1).

FIGURE 1: Pouch of Douglas hernia. The pelvic floor and small bowel descend into the Pouch of Douglas.
How does it occur? The pathophysiology is thought to be related to excessive abdominal straining in individuals with chronic constipation. This results in diminished pelvic floor muscle tone. Eventually, the whole pelvic floor descends, becoming funnel shaped due to stretching of the puborectalis muscle. Thus, stool is expelled by force, mostly through forces on the anterior rectal wall (which tends to prolapse after stool evacuation, with accompanied mucus secretion, soreness, and irritation).
Clinical pearl: Given the rectal wall prolapse that occurs after stool evacuation in Pouch of Douglas hernia, some patients will describe a rectal lump that bleeds after a bowel movement. The sensation of the rectal lump from the anterior rectal wall prolapse causes further straining.
Your patient reports pelvic pressure and bulge.
How do you proceed?
Physical examination
Look for perineal ballooning. Physical examination should start with inspection of the external genitalia. This inspection will identify any pelvic organ prolapse at or beyond the introitus. However, a Pouch of Douglas hernia will be missed if the patient is not examined during Valsalva or maximal strain. This maneuver will demonstrate the classic finding of perineal ballooning and is crucial to a final diagnosis of Pouch of Douglas hernia. Normally, the perineum will descend 1 cm to 2 cm during maximal strain; in Pouch of Douglas hernias, the perineum can descend up to 4 cm to 8 cm.4
Clinical pearl: It should be noted that, often, patients will not have a great deal of vaginal prolapse accompanying the perineal ballooning. In our opinion, this finding distinguishes Pouch of Douglas hernia from a vaginal vault prolapse caused by an enterocele.
Is rectal prolapse present? Beyond perineal ballooning, the presence of rectal prolapse should be evaluated. A rectocele of some degree is usually present. Asymmetric rectal prolapse affecting the anterior aspect of the rectal wall is consistent with a Pouch of Douglas hernia. This anatomic finding should be distinguished from true circumferential rectal prolapse, which remains in the differential diagnosis.
Basing the diagnosis of Pouch of Douglas hernia on physical examination alone can be difficult. Therefore, imaging studies are essential for accurate diagnosis.
Imaging investigations
Several imaging modalities can be used to diagnose such disorders of the pelvic floor as Pouch of Douglas hernia. These include:
- dynamic colpocystoproctography5
- defecography with oral barium6
- dynamic pelvic magnetic resonance imaging (MRI).7
In our experience, dynamic pelvic MRI has a high accuracy rate for diagnosing Pouch of Douglas hernia. FIGURE 2 illustrates the large Pouch of Douglas hernia filled with loops of small bowel. Perineal descent of the anorectal junction more than 3 cm below the pubococcygeal line during maximal straining is a diagnostic finding on imaging.7

FIGURE 2: MRI
Sagittal MRI during maximal Valsalva straining, demonstrating Pouch of Douglas hernia filled with small bowel.
What are your patient’s treatment options?
Reduce straining during bowel movements. The primary goal of treatment for Pouch of Douglas hernia should be relief of bothersome symptoms. Therefore, further damage can be prevented by eliminating straining during defecation. This can be accomplished with a bowel regimen that combines an irritant suppository (glycerin or bisacodyl) with a fiber supplement (the latter to increase bulk of the stool). Oral laxatives have limited use as many patients have lax anal sphincters and liquid stool could cause fecal incontinence.
Pelvic floor strengthening. The importance of pelvic floor physical therapy should be stressed. Patients can benefit from the use of modalities such as biofeedback to learn appropriate pelvic floor muscle relaxation techniques during defecation.8 While there is limited published evidence supporting the use of pelvic floor physical therapy, our anecdotal experience suggests that patients can gain considerable benefit with such conservative therapy.
Surgical therapy
Surgical repair of Pouch of Douglas hernia requires obliteration of the deep cul-de-sac (to prevent the small bowel from filling this space) and simultaneous pelvic floor reconstruction of the vaginal apex and any other compartments that are prolapsing (if pelvic organ prolapse is present). In our experience, these patients typically have derived greatest benefit from an abdominal approach. This usually can be accomplished with a sacrocolpopexy (if vaginal vault prolapse exists) with a Moschowitz or Halban procedure,9 uterosacral ligament plication, or a modified sacrocolpopexy with mesh augmentation to the sidewalls of the pelvis.10 There are currently no studies supporting one particular approach over another, but the most important feature of a surgical intervention is obliteration of the cul-de-sac (FIGURES 3, 4, and 5).

FIGURE 3: Open cul-de-sac. Open cul-de-sac after a prior abdominal sacrocolpopexy in a patient with a Pouch of Douglas hernia.

FIGURE 4: Obliterated cul-de-sac. Obliteration of the cul-de-sac with uterosacral ligament plication. Care is taken to prevent obstruction of the rectum at this level.

FIGURE 5: Cul-de-sac obliteration. Schematic diagram of obliteration of the cul-de-sac with uterosacral ligament plication sutures.
Final takeaways
Pouch of Douglas hernia is an important but often unrecognized cause of pelvic pressure and defecatory dysfunction. Perineal ballooning during maximal straining is highly suggestive of the diagnosis, with final diagnosis confirmed with various functional imaging studies of the pelvic floor. Management should include both conservative and surgical interventions to alleviate and prevent recurrence of symptoms.
ACKNOWLEDGMENT. The authors would like to thank Mr. John Hagen, Medical Illustrator, Mayo Clinic, for producing the illustrations in Figures 1 and 5.
We want to hear from you! Tell us what you think.
Urinary incontinence
Karen L. Noblett, MD, MAS, and Stephanie A. Jacobs, MD (Update, December 2012)
When and how to place an autologous rectus fascia
pubovaginal sling
Mickey Karram, MD, and Dani Zoorob, MD (Surgical Techniques, November 2012)
Pelvic floor dysfunction
Autumn L. Edenfield, MD, and Cindy L. Amundsen, MD (Update, October 2012)
Step by step: Obliterating the vaginal canal to correct pelvic organ prolapse
Mickey Karram, MD, and Janelle Evans, MD (Surgical Techniques, February 2012)
1. Samuelsson EC, Victor FT, Tibblin G, Svärdsudd KF. Signs of genital prolapse in a Swedish population of women 20 to 59 years of age and possible related factors. Am J Obstet Gynecol. 1999;180(2 Pt 1):299-305.
2. Boyles SH, Weber AM, Meyn L. Procedures for pelvic organ prolapse in the United States 1979-1997. Am J Obstet Gynecol. 2003;188(1):108-115.
3. Parks AG, Porter NH, Hardcastle J. The syndrome of the descending perineum. Proc R Soc Med. 1966;59(6):477-482.
4. Hardcastle JD. The descending perineum syndrome. Practitioner. 1969;203(217):612-619.
5. Maglinte DD, Bartram CI, Hale DA, et al. Functional imaging of the pelvic floor. Radiology. 2011;258(1):23-39.
6. Roos JE, Weishaupt D, Wildermuth S, Willmann JK, Marincek B, Hilfiker PR. Experience of 4 years with open MR defecography: pictorial review of anorectal anatomy and disease. Radiographics. 2002;22(4):817-832.
7. Fletcher JG, Busse RF, Riederer SJ, et al. Magnetic resonance imaging of anatomic and dynamic defects of the pelvic floor in defecatory disorders. Am J Gastroenterol. 2003;98(2):399-411.
8. Harewood GC, Coulie B, Camilleri M, Rath-Harvey D, Pemberton JH. Descending perineum syndrome: audit of clinical and laboratory features and outcome of pelvic floor retraining. Am J Gastroenterol. 1999;94(1):126-130.
9. Moschcowitz AV. The pathogenesis anatomy and cure of prolapse of the rectum. Surg Gyncol Obstetrics. 1912;15:7-21.
10. Gosselink MJ, van Dam JH, Huisman WM, Ginai AZ, Schouten WR. Treatment of enterocele by obliteration of the pelvic inlet. Dis Colon Rectum. 1999;42(7):940-944.
1. Samuelsson EC, Victor FT, Tibblin G, Svärdsudd KF. Signs of genital prolapse in a Swedish population of women 20 to 59 years of age and possible related factors. Am J Obstet Gynecol. 1999;180(2 Pt 1):299-305.
2. Boyles SH, Weber AM, Meyn L. Procedures for pelvic organ prolapse in the United States 1979-1997. Am J Obstet Gynecol. 2003;188(1):108-115.
3. Parks AG, Porter NH, Hardcastle J. The syndrome of the descending perineum. Proc R Soc Med. 1966;59(6):477-482.
4. Hardcastle JD. The descending perineum syndrome. Practitioner. 1969;203(217):612-619.
5. Maglinte DD, Bartram CI, Hale DA, et al. Functional imaging of the pelvic floor. Radiology. 2011;258(1):23-39.
6. Roos JE, Weishaupt D, Wildermuth S, Willmann JK, Marincek B, Hilfiker PR. Experience of 4 years with open MR defecography: pictorial review of anorectal anatomy and disease. Radiographics. 2002;22(4):817-832.
7. Fletcher JG, Busse RF, Riederer SJ, et al. Magnetic resonance imaging of anatomic and dynamic defects of the pelvic floor in defecatory disorders. Am J Gastroenterol. 2003;98(2):399-411.
8. Harewood GC, Coulie B, Camilleri M, Rath-Harvey D, Pemberton JH. Descending perineum syndrome: audit of clinical and laboratory features and outcome of pelvic floor retraining. Am J Gastroenterol. 1999;94(1):126-130.
9. Moschcowitz AV. The pathogenesis anatomy and cure of prolapse of the rectum. Surg Gyncol Obstetrics. 1912;15:7-21.
10. Gosselink MJ, van Dam JH, Huisman WM, Ginai AZ, Schouten WR. Treatment of enterocele by obliteration of the pelvic inlet. Dis Colon Rectum. 1999;42(7):940-944.
IN THIS ARTICLE
Clinical pearls at physical exam
Treatment options
Food and milk allergies increase growth impairment risk
Earn 0.25 hours AMA PRA Category 1 credit: Read this article, and click the link at the end to take the post-test.
SAN ANTONIO – Dietary restrictions prescribed for children with food allergies may lead to growth impairment, according to findings from a review of medical records for 245 food-allergic pediatric patients.
The risk of growth impairment was greatest for children whose dietary restrictions required elimination of more than two foods and/or elimination of cow’s milk, Dr. Brian P. Vickery reported at the annual meeting of the American Academy of Allergy, Asthma, and Immunology.
After age 2 years, the food-allergic children had lower mean percentiles for weight (67.5 vs. 72.5) and a lower body mass index (57.6 vs.68.0), than did 4,584 healthy age-matched controls.
Furthermore, the 52 patients with more than two food allergies (and thus more than two food restrictions), compared with 193 patients with one or two food allergies, had significantly lower mean percentiles for height (62.2 vs. 74.8) and weight (55.3 vs. 69.2). The 66 patients with milk allergy, compared with those with other food allergies, had lower mean percentiles for weight (54.5 vs. 70.6) and BMI (48.9 vs. 58.8), according to Dr. Vickery of the University of North Carolina at Chapel Hill.
Milk-allergic children younger than 2 years of age were particularly vulnerable to growth restriction, he said during a press briefing at the meeting.
The food-allergic children in this study, who were aged 1 month to 11 years and who presented to a University of North Carolina outpatient clinic between 2007 and 2011, also were compared with 205 "disease controls," consisting of children with either cystic fibrosis or celiac disease, two conditions that are associated with impaired growth. When children passed their second birthday, the effect of food allergy on growth was very similar to the effect of celiac disease on growth, Dr. Vickery said.
The findings of this study confirm those from a smaller study, conducted more than a decade ago, that also showed that milk allergy and multiple food allergies were associated with growth impairment.
That study is "the most commonly cited previous study to address the growth of food-allergic children in the United States," Dr. Vickery noted.
"The prevalence [of food allergy] has increased over the past 10 years, so we wanted to take another look in a bigger population to kind of reassess the impact of elimination diets on growth," he said.
The current findings demonstrate that a food allergy–associated elimination diet can place children at risk of impaired growth, compared with their healthy peers, regardless of whether they are under age 2 years, or are 2-11 years old, and that after age 2, the effect of food allergy on growth is very similar to that of chronic diseases known to affect growth, he said.
"While awareness of food allergy is increasing along with the prevalence of the disease, it is important to draw attention to the important consequences of elimination diets. We feel that providers should counsel patients and caregivers about the growth-related risks of the elimination diets that are used to treat food allergy, and ensure that families are excluding only the foods that are medically required or otherwise culturally indicated, that nutritional assessment and/or supplementation is provided as needed, and that subspecialty consultation is arranged, especially for children at highest risk," he said.
Dr. Vickery reported having no relevant financial disclosures.
To earn 0.25 hours AMA PRA Category 1 credit after reading this article, take the post-test here.
Earn 0.25 hours AMA PRA Category 1 credit: Read this article, and click the link at the end to take the post-test.
SAN ANTONIO – Dietary restrictions prescribed for children with food allergies may lead to growth impairment, according to findings from a review of medical records for 245 food-allergic pediatric patients.
The risk of growth impairment was greatest for children whose dietary restrictions required elimination of more than two foods and/or elimination of cow’s milk, Dr. Brian P. Vickery reported at the annual meeting of the American Academy of Allergy, Asthma, and Immunology.
After age 2 years, the food-allergic children had lower mean percentiles for weight (67.5 vs. 72.5) and a lower body mass index (57.6 vs.68.0), than did 4,584 healthy age-matched controls.
Furthermore, the 52 patients with more than two food allergies (and thus more than two food restrictions), compared with 193 patients with one or two food allergies, had significantly lower mean percentiles for height (62.2 vs. 74.8) and weight (55.3 vs. 69.2). The 66 patients with milk allergy, compared with those with other food allergies, had lower mean percentiles for weight (54.5 vs. 70.6) and BMI (48.9 vs. 58.8), according to Dr. Vickery of the University of North Carolina at Chapel Hill.
Milk-allergic children younger than 2 years of age were particularly vulnerable to growth restriction, he said during a press briefing at the meeting.
The food-allergic children in this study, who were aged 1 month to 11 years and who presented to a University of North Carolina outpatient clinic between 2007 and 2011, also were compared with 205 "disease controls," consisting of children with either cystic fibrosis or celiac disease, two conditions that are associated with impaired growth. When children passed their second birthday, the effect of food allergy on growth was very similar to the effect of celiac disease on growth, Dr. Vickery said.
The findings of this study confirm those from a smaller study, conducted more than a decade ago, that also showed that milk allergy and multiple food allergies were associated with growth impairment.
That study is "the most commonly cited previous study to address the growth of food-allergic children in the United States," Dr. Vickery noted.
"The prevalence [of food allergy] has increased over the past 10 years, so we wanted to take another look in a bigger population to kind of reassess the impact of elimination diets on growth," he said.
The current findings demonstrate that a food allergy–associated elimination diet can place children at risk of impaired growth, compared with their healthy peers, regardless of whether they are under age 2 years, or are 2-11 years old, and that after age 2, the effect of food allergy on growth is very similar to that of chronic diseases known to affect growth, he said.
"While awareness of food allergy is increasing along with the prevalence of the disease, it is important to draw attention to the important consequences of elimination diets. We feel that providers should counsel patients and caregivers about the growth-related risks of the elimination diets that are used to treat food allergy, and ensure that families are excluding only the foods that are medically required or otherwise culturally indicated, that nutritional assessment and/or supplementation is provided as needed, and that subspecialty consultation is arranged, especially for children at highest risk," he said.
Dr. Vickery reported having no relevant financial disclosures.
To earn 0.25 hours AMA PRA Category 1 credit after reading this article, take the post-test here.
Earn 0.25 hours AMA PRA Category 1 credit: Read this article, and click the link at the end to take the post-test.
SAN ANTONIO – Dietary restrictions prescribed for children with food allergies may lead to growth impairment, according to findings from a review of medical records for 245 food-allergic pediatric patients.
The risk of growth impairment was greatest for children whose dietary restrictions required elimination of more than two foods and/or elimination of cow’s milk, Dr. Brian P. Vickery reported at the annual meeting of the American Academy of Allergy, Asthma, and Immunology.
After age 2 years, the food-allergic children had lower mean percentiles for weight (67.5 vs. 72.5) and a lower body mass index (57.6 vs.68.0), than did 4,584 healthy age-matched controls.
Furthermore, the 52 patients with more than two food allergies (and thus more than two food restrictions), compared with 193 patients with one or two food allergies, had significantly lower mean percentiles for height (62.2 vs. 74.8) and weight (55.3 vs. 69.2). The 66 patients with milk allergy, compared with those with other food allergies, had lower mean percentiles for weight (54.5 vs. 70.6) and BMI (48.9 vs. 58.8), according to Dr. Vickery of the University of North Carolina at Chapel Hill.
Milk-allergic children younger than 2 years of age were particularly vulnerable to growth restriction, he said during a press briefing at the meeting.
The food-allergic children in this study, who were aged 1 month to 11 years and who presented to a University of North Carolina outpatient clinic between 2007 and 2011, also were compared with 205 "disease controls," consisting of children with either cystic fibrosis or celiac disease, two conditions that are associated with impaired growth. When children passed their second birthday, the effect of food allergy on growth was very similar to the effect of celiac disease on growth, Dr. Vickery said.
The findings of this study confirm those from a smaller study, conducted more than a decade ago, that also showed that milk allergy and multiple food allergies were associated with growth impairment.
That study is "the most commonly cited previous study to address the growth of food-allergic children in the United States," Dr. Vickery noted.
"The prevalence [of food allergy] has increased over the past 10 years, so we wanted to take another look in a bigger population to kind of reassess the impact of elimination diets on growth," he said.
The current findings demonstrate that a food allergy–associated elimination diet can place children at risk of impaired growth, compared with their healthy peers, regardless of whether they are under age 2 years, or are 2-11 years old, and that after age 2, the effect of food allergy on growth is very similar to that of chronic diseases known to affect growth, he said.
"While awareness of food allergy is increasing along with the prevalence of the disease, it is important to draw attention to the important consequences of elimination diets. We feel that providers should counsel patients and caregivers about the growth-related risks of the elimination diets that are used to treat food allergy, and ensure that families are excluding only the foods that are medically required or otherwise culturally indicated, that nutritional assessment and/or supplementation is provided as needed, and that subspecialty consultation is arranged, especially for children at highest risk," he said.
Dr. Vickery reported having no relevant financial disclosures.
To earn 0.25 hours AMA PRA Category 1 credit after reading this article, take the post-test here.
AT THE AAAAI ANNUAL MEETING
Kawasaki Disease
This illness is the most common cause of acquired heart disease in children in developed nations (with rheumatic fever remaining a more common cause in developing countries).1 The clinical features of KD are self-limited and will resolve even without appropriate therapy, but the patient can be left with significant coronary artery abnormalities.
A high index of clinical suspicion is needed to make a diagnosis of KD. Early diagnosis is critical because early treatment with IV gamma globulin can prevent coronary artery inflammation.2,3 Additionally, KD is potentially fatal; deaths continue to occur, even in the era of gamma globulin therapy.4 KD-associated mortality rates are fortunately very low—especially in Japan, where awareness of the illness is high, and diagnosis and treatment are usually prompt.5,6 Fatalities more commonly occur in children in whom the diagnosis is “missed,” not considered, or delayed.4Additionally, morbidity can be substantial in children who survive but are left with significant coronary artery abnormalities.7
KD has the highest incidence in children of Asian, particularly Japanese, descent: about 1 in 100 Japanese children develop KD by age 5 years.5Boys are more commonly affected than girls, at a ratio of 3 to 2.6 Also of note is that there is a 10-fold increased risk for KD in children with an affected sibling and a twofold increased risk for those with a previously affected parent.8,9 High incidence persists in Japanese children who have adopted a Western lifestyle10; thus, genetics are clearly a factor in KD incidence. However, KD occurs in all ethnic and racial groups worldwide. In areas with a low population of persons of Asian descent, most cases of KD occur in non-Asian children.11 In general, incidence in white children is one-tenth of that in Asian children, with an intermediate incidence in black and Hispanic children.
Clinical and epidemiologic features of KD strongly suggest an infectious cause, but no specific etiologic agent has yet been identified.12 The responsible agent could be a “new,” currently undiscovered virus.13
Patient Presentation and History
KD usually occurs in previously healthy children. Generally, the parent of a child with KD notices that the child seems more ill than is compatible with a typical viral illness. Though not included in the classic diagnostic criteria, marked irritability is very common in KD, particularly in infants. The history generally includes abrupt onset of fever that persists daily, with emergence of other KD signs and symptoms. On occasion, the parent will observe red eyes or a rash prior to fever onset.
In KD, the known signs and symptoms of illness may not all be present simultaneously (see figure). If a child presents to the clinician on day 7 of fever and has red cracked lips, red, swollen hands and feet, and conjunctival injection, and the parent states that the child had a red rash from day 2 through day 5 of illness that is no longer present on examination, the history of rash can be useful in making a diagnosis of KD.14 Such a patient should be considered to have prolonged fever with physical examination revealing four of the five other known clinical features—fulfilling the classic diagnostic criteria for KD.
Criteria for Diagnosis of KD
KD is recognized to occur in two forms: classic and incomplete (“atypical”) cases. Children with classic cases have prolonged fever and at least four of the five clinical findings as described by the American Heart Association (AHA) Committee on Endocarditis, Rheumatic Fever, and Kawasaki Disease14 (see Table 1,14). Patients with incomplete cases have prolonged fever with fewer than four of the five known clinical findings.
Classic and incomplete cases share the same laboratory profile and pathologic features. At present, the development of coronary artery abnormalities is the only means by which a patient with an incomplete case can be confirmed to have had KD. However, because early treatment can reduce the prevalence of coronary artery abnormalities,2 timely diagnosis of either a classic or an incomplete case of KD is essential. Use of the AHA Committee algorithm14 can help the clinician make this diagnosis.
Classic KD
Fever
Fever in children with KD is generally high-spiking, usually reaching 39°C to 40°C daily, and intermittent, with fever usually occurring several times per day, and normal temperatures between fevers. Classic diagnostic criteria require that fever be present for at least five days, but KD can be diagnosed prior to the fifth day of illness by an experienced clinician. Duration of fever is the single best predictor of the development of coronary artery abnormalities,15 and temperatures should be carefully monitored, rectally or orally, in any child in whom a diagnosis of KD is being considered.
Oral Mucosal Changes
Erythema of the lips, mouth, and pharynx are common findings. The lips can be so dry and chapped that they bleed. Oral ulcerations are not a feature of KD, and pharyngeal exudate is not present. A “strawberry” tongue, with prominent papillae on an erythematous base, may be observed.14
Conjunctival Injection
Redness of the bulbar conjunctiva (involving the globe of the eye rather than the palpebral conjunctiva of the eyelid) is a notable feature that can persist for weeks in some patients. There is generally minimal to no exudate. Children with KD may experience light sensitivity, likely related to anterior uveitis, which can be a feature of KD.14
Changes in the Hands and Feet
Redness and swelling of the hands and feet can be clinically striking and quite painful for the child. As a result, children with KD who experience this development will often refuse to walk and may refuse to hold objects. These are unusual complaints in minor viral illnesses of childhood and should heighten the clinician’s suspicion for KD. Some children with KD have more noticeable involvement of the hands or the feet alone, but these extremity changes are bilateral. Swelling of only one hand or foot, for example, should raise suspicion for a bacterial infection, such as cellulitis, arthritis, or osteomyelitis, and reduce suspicion for KD.
Desquamation of the fingers and toes, beginning just beneath the nailbeds, is commonly observed in the subacute phase of KD, at about two to three weeks after onset of fever.14 Therefore, this finding is not useful in making a diagnosis of KD in the first week of illness—the optimal time for therapy to be initiated.
Rash
Rash in KD generally takes one of three forms. Most common is an erythematous, maculopapular rash on the trunk and extremities. Some children have a scarlatiniform rash on the trunk and extremities, prompting an initial concern for group A streptococcal infection. Other children with KD have erythema multiforme, with typical target lesions. Bullae and pustules are not observed in KD, although on rare occasions, a very fine micropustular rash can be observed.14
Of note, some children with KD have a significant rash in the groin area, which can be confused with candidal diaper dermatitis. This rash can occur in toilet-trained children as well as those still in diapers; it is often associated with desquamation of the affected skin, which is not typical of candidal dermatitis. In any child with prolonged fever and a desquamating erythematous groin rash, it is essential to make a careful examination for other features of KD.
Cervical Lymphadenopathy
This feature of KD is the least commonly observed, particularly in infants.16 In some older children, however, it is the most striking feature and can lead to a misdiagnosis of bacterial cervical adenitis.14 Children with this misdiagnosis are often treated with antibiotics with no response and delay of the correct diagnosis. Careful examination will usually reveal other clinical features of KD. Small lymph nodes are commonly palpated in the neck of young children; the lymph node should be > 1.5 cm in diameter to fulfill this criterion, according to the AHA Committee criteria for diagnosing KD.14 Lymphadenopathy, when present, tends to involve primarily the anterior cervical nodes overlying the sternocleidomastoid muscles.17
Incomplete KD
Patients with incomplete or “atypical” KD have fever with fewer than four of the other clinical findings. The term atypical does not indicate the presence of clinical findings that are not characteristic of KD. The AHA Committee14 recommends that a diagnosis of incomplete KD be considered in a child with fever for five days or longer with at least two other clinical features of KD, and with compatible laboratory and/or echocardiographic findings, as shown in Table 1.14
Laboratory Workup
In the absence of a known pathologic cause of KD, no definitive diagnostic test exists. However, certain laboratory findings, shown in Table 214, are considered characteristic of the diagnosis. For example, a low peripheral white blood cell count with lymphocyte predominance is not considered a compatible laboratory profile for KD.
Generally, a complete blood count with differential, C-reactive protein (CRP), erythrocyte sedimentation rate (ESR), liver function tests, and urinalysis are ordered. On diagnosis of KD, an echocardiogram should also be obtained to determine whether early coronary artery dilation is present, and to serve as a comparison for future studies.14,18
Diagnosis
In the presence of the previously described diagnostic criteria, a diagnosis of KD can be made. Patients with a presentation suggestive of incomplete KD should be treated for the illness if they have elevated CRP or ESR and other compatible laboratory or echocardiographic features shown in Table 1.14 In all children with a history of prolonged fever, rash and nonpurulent conjunctivitis—especially those younger than 1 year but including adolescents—KD should be included in the differential diagnosis.
Several other illnesses should be considered in the differential diagnosis of KD.
Measles, in an unimmunized or immigrant child, is a diagnosis that should be considered. In measles, rash usually begins on the face and behind the ears, then progresses to involve the entire body. In patients with measles in whom the rash has just appeared, Koplik spots (small white spots on the reddened oral mucosa near the molars) may be visible. Patients with measles have the classic triad of cough, coryza (rhinorrhea), and conjunctivitis; conjunctivitis is generally exudative.19
Scarlet fever should be considered, along with possible KD, in patients with a scarlatiniform rash. Because in some geographic areas 20% to 30% of children can be carriers of group A streptococcus, a positive throat culture could indicate acute infection or the carrier state.20 Treatment with penicillin for 24 hours generally clarifies the diagnosis; if the patient fails to improve on this therapy, KD should once again be considered.
Toxic shock syndrome can share some clinical features with severe KD, and patients with KD can experience shock.21 The presence of a staphylococcal or streptococcal focus of infection indicates toxic shock syndrome as the diagnosis, whereas the presence of coronary dilation on echocardiography points to KD.
Drug reactions can sometimes be confused with KD, and some patients with KD have a rash that may itch. The presence of facial and eyelid swelling is more likely to suggest a drug reaction than KD, and laboratory testing generally shows markedly elevated acute-phase reactants in KD,22 compared with those who are experiencing a drug reaction.
Viral illnesses, such as enterovirus infection, can share clinical features of KD, but usually will yield the elevated neutrophil count and acute-phase reactant elevations found in patients with KD.14,22 Children with adenovirus infection usually present with exudative pharyngitis and conjunctivitis.23
Once Diagnosis Is Confirmed
Patients in whom a diagnosis of KD is confirmed should be treated in a hospital where pediatric echocardiography is available, including measurement of coronary artery diameters in small children. In these patients, a Z score is calculated, representing standard deviations exceeding mean coronary artery dimensions, based on the patient’s body surface area. A child with a right proximal or left anterior descending coronary artery Z score greater than 2.5 (ie, 2.5 standard deviations above the mean) is confirmed to have an enlarged coronary artery.14,18
Echocardiography technicians must be well trained and experienced to be able to perform the necessary measurements, particularly in febrile, ill, uncooperative children. Only a pediatric cardiologist is qualified to interpret the studies.14
Treatment and Management
As soon as possible after a diagnosis of KD is made, patients should be treated with IV gamma globulin, 2 g/kg over 8 to 12 hours, and oral aspirin 80 to 100 mg/kg/d, divided for administration every 6 hours.3,14,24 High-dose aspirin is needed for its anti-inflammatory effect; its benefits for patients with KD appear to greatly outweigh the extremely low risk for Reye syndrome associated with aspirin use. In the rare patient with KD who has concurrent, documented influenza or varicella (and thus an increased risk for Reye syndrome with aspirin use), aspirin should be withheld, and an expert in KD should be consulted.
Most patients with KD respond rapidly to this therapeutic approach, with resolution of fever and improvement in clinical signs within 24 to 48 hours. However, 15% to 20% of patients do not respond to this initial treatment.14,25 The optimal therapy for such patients is unknown, but a second 2 g/kg dose of IV gamma globulin, IV methylprednisolone 30 mg/kg for 1 to 3 days, or IV infliximab 5 mg/kg are additional options.25-28
Patients who still do not respond may require additional therapies (eg, steroid therapy, infliximab, perhaps other immunosuppressive therapies), as these children are at high risk for coronary artery abnormalities. Referral of such patients to a center with extensive experience in caring for patients with KD should be considered.
Classic treatment studies have demonstrated a reduction in the prevalence of coronary artery abnormalities two months after illness onset from 18% in patients who were treated with aspirin alone to 4% in those who received IV gamma globulin and aspirin.24 However, more recent research, using newer algorithms to calculate normal coronary artery size based on body surface area, reveals that coronary artery dilation is more common in KD than previously recognized.2 Specifically, about 18% of patients with KD who present within the first 10 days of fever onset have already experienced some degree of coronary artery dilation before therapy is initiated. This highlights the importance of earlier diagnosis and treatment.
On the 14th day after fever onset (or 2 to 3 days after fever has resolved), aspirin is reduced to a single daily dose of 3 to 5 mg/kg.14 Aspirin use is continued for its antithrombotic effect until the ESR has normalized, provided echocardiography results have remained normal throughout. Aspirin therapy may be required indefinitely in patients who develop coronary artery aneurysms; in severe cases, additional antiplatelet or antithrombotic therapies may be required to prevent aneurysmal thrombosis.
Patient Education
A diagnosis of KD can be frightening. In the United States, most parents of a child diagnosed with KD have never heard of the condition and will need education and support. Once the child has been discharged and the parents discuss the illness with friends and acquaintances, they generally become aware that other children they know have had KD. The illness is not rare, but the public is generally unfamiliar with it.
Parents can be referred to the Web sites of the American Heart Association (www.heart.org/HEARTORG/Conditions/More/CardiovascularConditionsof Childhood/Kawasaki-Disease_UCM_308777_Article.jsp) and the American Academy of Pediatrics (see www.healthychildren.org), where educational materials about KD are posted. In the US, the Kawasaki Disease Foundation (http://www.kdfoundation.org/) also provides information and support to families with children affected by KD.
Follow-Up
Children with KD should undergo echocardiography at diagnosis, at 2 to 3 weeks, and again at 6 to 8 weeks after fever onset.14 At that point, a child with KD who has developed no evidence of coronary artery abnormalities will require no additional echocardiograms, as no evidence of late-onset, KD-associated coronary artery dilation has been reported.14,29 Children who have developed coronary artery dilation, however, will require further studies, as recommended by their pediatric cardiologist.
Laboratory testing should be performed at diagnosis, and acute-phase reactants (eg, CRP) should be monitored after discharge until normal levels are reached. Follow-up visits (often scheduled at 2 to 3 weeks and 6 to 8 weeks after illness onset) should consist of physical examination and laboratory testing, generally including a complete blood count and CRP level. This value often normalizes within 2 to 3 weeks after illness onset and can be helpful in monitoring clinical response, particularly in patients with persistent fever who require continuing therapy. Because the ESR rises transiently after IV gamma globulin therapy,30 it is not considered useful in monitoring response to therapy; the ESR usually normalizes by 6 to 8 weeks after fever onset.
Conclusion
KD is a potentially life-threatening illness of early childhood that should be considered in the differential diagnosis of any child with prolonged, unexplained fever. In KD, fever, rash, conjunctival injection, oral mucosal changes, extremity changes, and cervical adenopathy are self-limited, but any resulting coronary artery dilation and aneurysms can persist lifelong.
A high index of clinical suspicion is required to make a diagnosis of KD. Children with incomplete KD may present with fever and fewer than four other known clinical features; these children must undergo laboratory and echocardiographic studies for a diagnosis to be confirmed. Early diagnosis is essential for effective therapy with IV gamma globulin and aspirin to be administered before KD-associated coronary artery abnormalities can develop.
Additional research is needed to identify the cause of the disease, making it possible to develop a specific diagnostic test and etiology-targeted therapy.
References
1. Taubert KA, Rowley AH, Shulman ST. Seven-year national survey of Kawasaki disease and acute rheumatic fever. Pediatr Infect Dis J.1994;13(8):704-708.
2. Dominguez SR, Anderson MS, Eladawy M, Glodé MP. Preventing coronary artery abnormalities: a need for earlier diagnosis and treatment of Kawasaki disease. Pediatr Infect Dis J. 2012 Jul 3. [Epub ahead of print]
3. Newburger JW, Takahashi M, Beiser AS, et al. A single intravenous infusion of gamma globulin as compared with four infusions in the treatment of acute Kawasaki syndrome. N Engl J Med. 1991;324(23):1633-1639.
4. Orenstein JM, Shulman ST, Fox LM, et al. Three linked vasculopathic processes characterize Kawasaki disease: a light and transmission electron microscopic study. PLoS One. 2012;7(6):e38998. Epub 2012 Jun 18.
5. Nakamura Y, Yashiro M, Uehara R, et al. Epidemiologic features of Kawasaki disease in Japan: results of the 2009-2010 nationwide survey. J Epidemiol. 2012;22(3):216-221.
6. Nakamura Y, Yanagawa H, Harada K, et al. Mortality among persons with a history of Kawasaki disease in Japan: the fifth look. Arch Pediatr Adolesc Med. 2002;156(2):162-165.
7. Suda K, Iemura M, Nishiono H, et al. Long-term prognosis of patients with Kawasaki disease complicated by giant coronary aneurysms: a single-institution experience. Circulation. 2011;123(17):1836-1842.
8. Fujita Y, Nakamura Y, Sakata K, et al. Kawasaki disease in families. Pediatrics. 1989;84(4):666-669.
9. Uehara R, Yashiro M, Nakamura Y, Yanagawa H. Kawasaki disease in parents and children. Acta Paediatr. 2003;92(6):694-697.
10. Dean AG, Melish ME, Hicks R, Palumbo NE. An epidemic of Kawasaki syndrome in Hawaii. J Pediatr. 1982;100(4):552-557.
11. Shulman ST, McAuley JB, Pachman LM, et al. Risk of coronary abnormalities due to Kawasaki disease in urban area with small Asian population. Am J Dis Child. 1987;141(4):420-425.
12. Rowley AH, Baker SC, Orenstein JM, Shulman ST. Searching for the cause of Kawasaki disease: cytoplasmic inclusion bodies provide new insight. Nat Rev Microbiol. 2008;6(5):394-401.
13. Rowley AH, Baker SC, Shulman ST, et al. Ultrastructural, immunofluorescence, and RNA evidence support the hypothesis of a “new” virus associated with Kawasaki disease. J Infect Dis. 2011;203(7):1021-1030.
14. Newburger JW, Takahashi M, Gerber MA, et al. Diagnosis, treatment, and long-term management of Kawasaki disease: a statement for health professionals from the Committee on Rheumatic Fever, Endocarditis, and Kawasaki Disease, Council on Cardiovascular Disease in the Young, American Heart Association. Pediatrics. 2004;114(6):1708-1733.
15. Koren G, Lavi S, Rose V, Rowe R. Kawasaki disease: review of risk factors for coronary aneurysms. J Pediatr. 1986;108(3):388-392.
16. Sung RY, Ng YM, Choi KC, et al; Hong Kong Kawasaki Disease Study Group. Lack of association of cervical lymphadenopathy and coronary artery complications in Kawasaki disease. Pediatr Infect Dis J. 2006;25(6):521-525.
17. April MM, Burns JC, Newburger JW, Healy GB. Kawasaki disease and cervical adenopathy. Arch Otolaryngol Head Neck Surg. 1989;115:512.
18. Bratincsak A, Reddy VD, Purohit PJ, et al. Coronary artery dilation in acute Kawasaki disease and acute illnesses associated with fever.Pediatr Infect Dis J. 2012;31(9):924-926.
19. CDC. Hospital-associated measles outbreak: Pennsylvania, March – April 2009. MMWR Morb Mortal Wkly Rep. 2012;61(2):30-32.
20. Roberts AL, Connolly KL, Kirse DJ, et al. Detection of group A Streptococcus in tonsils from pediatric patients reveals high rate of asymptomatic streptococcal carriage. BMC Pediatr. 2012 Jan 9;12:3.
21. Dominguez SR, Friedman K, Seewalk R, et al. Kawasaki disease in a pediatric intensive care unit: a case-control study. Pediatrics.2008;122(4):e786-e790.
22. Huang MY, Gupta-Malhotra M, Huang JJ, et al. Acute-phase reactants and a supplemental diagnostic aid for Kawasaki disease. Pediatr Cardiol. 2010;31(8):1209-1213.
23. Singh-Naz N, Rodriguez W. Adenoviral infections in children. Adv Pediatr Infect Dis. 1996;11:365-388.
24. Newburger JW, Takahashi M, Burns JC, et al. The treatment of Kawasaki syndrome with intravenous gamma globulin. N Engl J Med.1986;315(6):341-347.
25. Son MB, Gauvreau K, Ma L, et al. Treatment of Kawasaki disease: analysis of 27 US pediatric hospitals from 2001 to 2007. Pediatrics.2009;124(1):1-8.
26. Chiyonobu T, Yoshihara T, Mori K, et al. Early intravenous gamma globulin retreatment for refractory Kawasaki disease. Clin Pediatr (Phila).2003;42(3):269-272.
27. Shah I, Prabhu SS. Response of refractory Kawasaki disease to intravenous methylprednisolone. Ann Trop Paediatr. 2009;29(1):51-53.
28. Blaisdell LL, Hayman JA, Moran. Infliximab treatment for pediatric refractory Kawasaki disease. Pediatr Cardiol. 2011;32(7):1023-1027.
29. Senzaki H. Long-term outcome of Kawasaki disease. Circulation. 2008;118(25);2763-2772.
30. Lee KY, Lee HS, Hong JH, et al. High-dose intravenous immunoglobulin downregulates the activated levels of inflammatory indices except erythrocyte sedimentation rate in acute stage of Kawasaki disease. J Trop Pediatr. 2005;51(2):98-101.
This illness is the most common cause of acquired heart disease in children in developed nations (with rheumatic fever remaining a more common cause in developing countries).1 The clinical features of KD are self-limited and will resolve even without appropriate therapy, but the patient can be left with significant coronary artery abnormalities.
A high index of clinical suspicion is needed to make a diagnosis of KD. Early diagnosis is critical because early treatment with IV gamma globulin can prevent coronary artery inflammation.2,3 Additionally, KD is potentially fatal; deaths continue to occur, even in the era of gamma globulin therapy.4 KD-associated mortality rates are fortunately very low—especially in Japan, where awareness of the illness is high, and diagnosis and treatment are usually prompt.5,6 Fatalities more commonly occur in children in whom the diagnosis is “missed,” not considered, or delayed.4Additionally, morbidity can be substantial in children who survive but are left with significant coronary artery abnormalities.7
KD has the highest incidence in children of Asian, particularly Japanese, descent: about 1 in 100 Japanese children develop KD by age 5 years.5Boys are more commonly affected than girls, at a ratio of 3 to 2.6 Also of note is that there is a 10-fold increased risk for KD in children with an affected sibling and a twofold increased risk for those with a previously affected parent.8,9 High incidence persists in Japanese children who have adopted a Western lifestyle10; thus, genetics are clearly a factor in KD incidence. However, KD occurs in all ethnic and racial groups worldwide. In areas with a low population of persons of Asian descent, most cases of KD occur in non-Asian children.11 In general, incidence in white children is one-tenth of that in Asian children, with an intermediate incidence in black and Hispanic children.
Clinical and epidemiologic features of KD strongly suggest an infectious cause, but no specific etiologic agent has yet been identified.12 The responsible agent could be a “new,” currently undiscovered virus.13
Patient Presentation and History
KD usually occurs in previously healthy children. Generally, the parent of a child with KD notices that the child seems more ill than is compatible with a typical viral illness. Though not included in the classic diagnostic criteria, marked irritability is very common in KD, particularly in infants. The history generally includes abrupt onset of fever that persists daily, with emergence of other KD signs and symptoms. On occasion, the parent will observe red eyes or a rash prior to fever onset.
In KD, the known signs and symptoms of illness may not all be present simultaneously (see figure). If a child presents to the clinician on day 7 of fever and has red cracked lips, red, swollen hands and feet, and conjunctival injection, and the parent states that the child had a red rash from day 2 through day 5 of illness that is no longer present on examination, the history of rash can be useful in making a diagnosis of KD.14 Such a patient should be considered to have prolonged fever with physical examination revealing four of the five other known clinical features—fulfilling the classic diagnostic criteria for KD.
Criteria for Diagnosis of KD
KD is recognized to occur in two forms: classic and incomplete (“atypical”) cases. Children with classic cases have prolonged fever and at least four of the five clinical findings as described by the American Heart Association (AHA) Committee on Endocarditis, Rheumatic Fever, and Kawasaki Disease14 (see Table 1,14). Patients with incomplete cases have prolonged fever with fewer than four of the five known clinical findings.
Classic and incomplete cases share the same laboratory profile and pathologic features. At present, the development of coronary artery abnormalities is the only means by which a patient with an incomplete case can be confirmed to have had KD. However, because early treatment can reduce the prevalence of coronary artery abnormalities,2 timely diagnosis of either a classic or an incomplete case of KD is essential. Use of the AHA Committee algorithm14 can help the clinician make this diagnosis.
Classic KD
Fever
Fever in children with KD is generally high-spiking, usually reaching 39°C to 40°C daily, and intermittent, with fever usually occurring several times per day, and normal temperatures between fevers. Classic diagnostic criteria require that fever be present for at least five days, but KD can be diagnosed prior to the fifth day of illness by an experienced clinician. Duration of fever is the single best predictor of the development of coronary artery abnormalities,15 and temperatures should be carefully monitored, rectally or orally, in any child in whom a diagnosis of KD is being considered.
Oral Mucosal Changes
Erythema of the lips, mouth, and pharynx are common findings. The lips can be so dry and chapped that they bleed. Oral ulcerations are not a feature of KD, and pharyngeal exudate is not present. A “strawberry” tongue, with prominent papillae on an erythematous base, may be observed.14
Conjunctival Injection
Redness of the bulbar conjunctiva (involving the globe of the eye rather than the palpebral conjunctiva of the eyelid) is a notable feature that can persist for weeks in some patients. There is generally minimal to no exudate. Children with KD may experience light sensitivity, likely related to anterior uveitis, which can be a feature of KD.14
Changes in the Hands and Feet
Redness and swelling of the hands and feet can be clinically striking and quite painful for the child. As a result, children with KD who experience this development will often refuse to walk and may refuse to hold objects. These are unusual complaints in minor viral illnesses of childhood and should heighten the clinician’s suspicion for KD. Some children with KD have more noticeable involvement of the hands or the feet alone, but these extremity changes are bilateral. Swelling of only one hand or foot, for example, should raise suspicion for a bacterial infection, such as cellulitis, arthritis, or osteomyelitis, and reduce suspicion for KD.
Desquamation of the fingers and toes, beginning just beneath the nailbeds, is commonly observed in the subacute phase of KD, at about two to three weeks after onset of fever.14 Therefore, this finding is not useful in making a diagnosis of KD in the first week of illness—the optimal time for therapy to be initiated.
Rash
Rash in KD generally takes one of three forms. Most common is an erythematous, maculopapular rash on the trunk and extremities. Some children have a scarlatiniform rash on the trunk and extremities, prompting an initial concern for group A streptococcal infection. Other children with KD have erythema multiforme, with typical target lesions. Bullae and pustules are not observed in KD, although on rare occasions, a very fine micropustular rash can be observed.14
Of note, some children with KD have a significant rash in the groin area, which can be confused with candidal diaper dermatitis. This rash can occur in toilet-trained children as well as those still in diapers; it is often associated with desquamation of the affected skin, which is not typical of candidal dermatitis. In any child with prolonged fever and a desquamating erythematous groin rash, it is essential to make a careful examination for other features of KD.
Cervical Lymphadenopathy
This feature of KD is the least commonly observed, particularly in infants.16 In some older children, however, it is the most striking feature and can lead to a misdiagnosis of bacterial cervical adenitis.14 Children with this misdiagnosis are often treated with antibiotics with no response and delay of the correct diagnosis. Careful examination will usually reveal other clinical features of KD. Small lymph nodes are commonly palpated in the neck of young children; the lymph node should be > 1.5 cm in diameter to fulfill this criterion, according to the AHA Committee criteria for diagnosing KD.14 Lymphadenopathy, when present, tends to involve primarily the anterior cervical nodes overlying the sternocleidomastoid muscles.17
Incomplete KD
Patients with incomplete or “atypical” KD have fever with fewer than four of the other clinical findings. The term atypical does not indicate the presence of clinical findings that are not characteristic of KD. The AHA Committee14 recommends that a diagnosis of incomplete KD be considered in a child with fever for five days or longer with at least two other clinical features of KD, and with compatible laboratory and/or echocardiographic findings, as shown in Table 1.14
Laboratory Workup
In the absence of a known pathologic cause of KD, no definitive diagnostic test exists. However, certain laboratory findings, shown in Table 214, are considered characteristic of the diagnosis. For example, a low peripheral white blood cell count with lymphocyte predominance is not considered a compatible laboratory profile for KD.
Generally, a complete blood count with differential, C-reactive protein (CRP), erythrocyte sedimentation rate (ESR), liver function tests, and urinalysis are ordered. On diagnosis of KD, an echocardiogram should also be obtained to determine whether early coronary artery dilation is present, and to serve as a comparison for future studies.14,18
Diagnosis
In the presence of the previously described diagnostic criteria, a diagnosis of KD can be made. Patients with a presentation suggestive of incomplete KD should be treated for the illness if they have elevated CRP or ESR and other compatible laboratory or echocardiographic features shown in Table 1.14 In all children with a history of prolonged fever, rash and nonpurulent conjunctivitis—especially those younger than 1 year but including adolescents—KD should be included in the differential diagnosis.
Several other illnesses should be considered in the differential diagnosis of KD.
Measles, in an unimmunized or immigrant child, is a diagnosis that should be considered. In measles, rash usually begins on the face and behind the ears, then progresses to involve the entire body. In patients with measles in whom the rash has just appeared, Koplik spots (small white spots on the reddened oral mucosa near the molars) may be visible. Patients with measles have the classic triad of cough, coryza (rhinorrhea), and conjunctivitis; conjunctivitis is generally exudative.19
Scarlet fever should be considered, along with possible KD, in patients with a scarlatiniform rash. Because in some geographic areas 20% to 30% of children can be carriers of group A streptococcus, a positive throat culture could indicate acute infection or the carrier state.20 Treatment with penicillin for 24 hours generally clarifies the diagnosis; if the patient fails to improve on this therapy, KD should once again be considered.
Toxic shock syndrome can share some clinical features with severe KD, and patients with KD can experience shock.21 The presence of a staphylococcal or streptococcal focus of infection indicates toxic shock syndrome as the diagnosis, whereas the presence of coronary dilation on echocardiography points to KD.
Drug reactions can sometimes be confused with KD, and some patients with KD have a rash that may itch. The presence of facial and eyelid swelling is more likely to suggest a drug reaction than KD, and laboratory testing generally shows markedly elevated acute-phase reactants in KD,22 compared with those who are experiencing a drug reaction.
Viral illnesses, such as enterovirus infection, can share clinical features of KD, but usually will yield the elevated neutrophil count and acute-phase reactant elevations found in patients with KD.14,22 Children with adenovirus infection usually present with exudative pharyngitis and conjunctivitis.23
Once Diagnosis Is Confirmed
Patients in whom a diagnosis of KD is confirmed should be treated in a hospital where pediatric echocardiography is available, including measurement of coronary artery diameters in small children. In these patients, a Z score is calculated, representing standard deviations exceeding mean coronary artery dimensions, based on the patient’s body surface area. A child with a right proximal or left anterior descending coronary artery Z score greater than 2.5 (ie, 2.5 standard deviations above the mean) is confirmed to have an enlarged coronary artery.14,18
Echocardiography technicians must be well trained and experienced to be able to perform the necessary measurements, particularly in febrile, ill, uncooperative children. Only a pediatric cardiologist is qualified to interpret the studies.14
Treatment and Management
As soon as possible after a diagnosis of KD is made, patients should be treated with IV gamma globulin, 2 g/kg over 8 to 12 hours, and oral aspirin 80 to 100 mg/kg/d, divided for administration every 6 hours.3,14,24 High-dose aspirin is needed for its anti-inflammatory effect; its benefits for patients with KD appear to greatly outweigh the extremely low risk for Reye syndrome associated with aspirin use. In the rare patient with KD who has concurrent, documented influenza or varicella (and thus an increased risk for Reye syndrome with aspirin use), aspirin should be withheld, and an expert in KD should be consulted.
Most patients with KD respond rapidly to this therapeutic approach, with resolution of fever and improvement in clinical signs within 24 to 48 hours. However, 15% to 20% of patients do not respond to this initial treatment.14,25 The optimal therapy for such patients is unknown, but a second 2 g/kg dose of IV gamma globulin, IV methylprednisolone 30 mg/kg for 1 to 3 days, or IV infliximab 5 mg/kg are additional options.25-28
Patients who still do not respond may require additional therapies (eg, steroid therapy, infliximab, perhaps other immunosuppressive therapies), as these children are at high risk for coronary artery abnormalities. Referral of such patients to a center with extensive experience in caring for patients with KD should be considered.
Classic treatment studies have demonstrated a reduction in the prevalence of coronary artery abnormalities two months after illness onset from 18% in patients who were treated with aspirin alone to 4% in those who received IV gamma globulin and aspirin.24 However, more recent research, using newer algorithms to calculate normal coronary artery size based on body surface area, reveals that coronary artery dilation is more common in KD than previously recognized.2 Specifically, about 18% of patients with KD who present within the first 10 days of fever onset have already experienced some degree of coronary artery dilation before therapy is initiated. This highlights the importance of earlier diagnosis and treatment.
On the 14th day after fever onset (or 2 to 3 days after fever has resolved), aspirin is reduced to a single daily dose of 3 to 5 mg/kg.14 Aspirin use is continued for its antithrombotic effect until the ESR has normalized, provided echocardiography results have remained normal throughout. Aspirin therapy may be required indefinitely in patients who develop coronary artery aneurysms; in severe cases, additional antiplatelet or antithrombotic therapies may be required to prevent aneurysmal thrombosis.
Patient Education
A diagnosis of KD can be frightening. In the United States, most parents of a child diagnosed with KD have never heard of the condition and will need education and support. Once the child has been discharged and the parents discuss the illness with friends and acquaintances, they generally become aware that other children they know have had KD. The illness is not rare, but the public is generally unfamiliar with it.
Parents can be referred to the Web sites of the American Heart Association (www.heart.org/HEARTORG/Conditions/More/CardiovascularConditionsof Childhood/Kawasaki-Disease_UCM_308777_Article.jsp) and the American Academy of Pediatrics (see www.healthychildren.org), where educational materials about KD are posted. In the US, the Kawasaki Disease Foundation (http://www.kdfoundation.org/) also provides information and support to families with children affected by KD.
Follow-Up
Children with KD should undergo echocardiography at diagnosis, at 2 to 3 weeks, and again at 6 to 8 weeks after fever onset.14 At that point, a child with KD who has developed no evidence of coronary artery abnormalities will require no additional echocardiograms, as no evidence of late-onset, KD-associated coronary artery dilation has been reported.14,29 Children who have developed coronary artery dilation, however, will require further studies, as recommended by their pediatric cardiologist.
Laboratory testing should be performed at diagnosis, and acute-phase reactants (eg, CRP) should be monitored after discharge until normal levels are reached. Follow-up visits (often scheduled at 2 to 3 weeks and 6 to 8 weeks after illness onset) should consist of physical examination and laboratory testing, generally including a complete blood count and CRP level. This value often normalizes within 2 to 3 weeks after illness onset and can be helpful in monitoring clinical response, particularly in patients with persistent fever who require continuing therapy. Because the ESR rises transiently after IV gamma globulin therapy,30 it is not considered useful in monitoring response to therapy; the ESR usually normalizes by 6 to 8 weeks after fever onset.
Conclusion
KD is a potentially life-threatening illness of early childhood that should be considered in the differential diagnosis of any child with prolonged, unexplained fever. In KD, fever, rash, conjunctival injection, oral mucosal changes, extremity changes, and cervical adenopathy are self-limited, but any resulting coronary artery dilation and aneurysms can persist lifelong.
A high index of clinical suspicion is required to make a diagnosis of KD. Children with incomplete KD may present with fever and fewer than four other known clinical features; these children must undergo laboratory and echocardiographic studies for a diagnosis to be confirmed. Early diagnosis is essential for effective therapy with IV gamma globulin and aspirin to be administered before KD-associated coronary artery abnormalities can develop.
Additional research is needed to identify the cause of the disease, making it possible to develop a specific diagnostic test and etiology-targeted therapy.
References
1. Taubert KA, Rowley AH, Shulman ST. Seven-year national survey of Kawasaki disease and acute rheumatic fever. Pediatr Infect Dis J.1994;13(8):704-708.
2. Dominguez SR, Anderson MS, Eladawy M, Glodé MP. Preventing coronary artery abnormalities: a need for earlier diagnosis and treatment of Kawasaki disease. Pediatr Infect Dis J. 2012 Jul 3. [Epub ahead of print]
3. Newburger JW, Takahashi M, Beiser AS, et al. A single intravenous infusion of gamma globulin as compared with four infusions in the treatment of acute Kawasaki syndrome. N Engl J Med. 1991;324(23):1633-1639.
4. Orenstein JM, Shulman ST, Fox LM, et al. Three linked vasculopathic processes characterize Kawasaki disease: a light and transmission electron microscopic study. PLoS One. 2012;7(6):e38998. Epub 2012 Jun 18.
5. Nakamura Y, Yashiro M, Uehara R, et al. Epidemiologic features of Kawasaki disease in Japan: results of the 2009-2010 nationwide survey. J Epidemiol. 2012;22(3):216-221.
6. Nakamura Y, Yanagawa H, Harada K, et al. Mortality among persons with a history of Kawasaki disease in Japan: the fifth look. Arch Pediatr Adolesc Med. 2002;156(2):162-165.
7. Suda K, Iemura M, Nishiono H, et al. Long-term prognosis of patients with Kawasaki disease complicated by giant coronary aneurysms: a single-institution experience. Circulation. 2011;123(17):1836-1842.
8. Fujita Y, Nakamura Y, Sakata K, et al. Kawasaki disease in families. Pediatrics. 1989;84(4):666-669.
9. Uehara R, Yashiro M, Nakamura Y, Yanagawa H. Kawasaki disease in parents and children. Acta Paediatr. 2003;92(6):694-697.
10. Dean AG, Melish ME, Hicks R, Palumbo NE. An epidemic of Kawasaki syndrome in Hawaii. J Pediatr. 1982;100(4):552-557.
11. Shulman ST, McAuley JB, Pachman LM, et al. Risk of coronary abnormalities due to Kawasaki disease in urban area with small Asian population. Am J Dis Child. 1987;141(4):420-425.
12. Rowley AH, Baker SC, Orenstein JM, Shulman ST. Searching for the cause of Kawasaki disease: cytoplasmic inclusion bodies provide new insight. Nat Rev Microbiol. 2008;6(5):394-401.
13. Rowley AH, Baker SC, Shulman ST, et al. Ultrastructural, immunofluorescence, and RNA evidence support the hypothesis of a “new” virus associated with Kawasaki disease. J Infect Dis. 2011;203(7):1021-1030.
14. Newburger JW, Takahashi M, Gerber MA, et al. Diagnosis, treatment, and long-term management of Kawasaki disease: a statement for health professionals from the Committee on Rheumatic Fever, Endocarditis, and Kawasaki Disease, Council on Cardiovascular Disease in the Young, American Heart Association. Pediatrics. 2004;114(6):1708-1733.
15. Koren G, Lavi S, Rose V, Rowe R. Kawasaki disease: review of risk factors for coronary aneurysms. J Pediatr. 1986;108(3):388-392.
16. Sung RY, Ng YM, Choi KC, et al; Hong Kong Kawasaki Disease Study Group. Lack of association of cervical lymphadenopathy and coronary artery complications in Kawasaki disease. Pediatr Infect Dis J. 2006;25(6):521-525.
17. April MM, Burns JC, Newburger JW, Healy GB. Kawasaki disease and cervical adenopathy. Arch Otolaryngol Head Neck Surg. 1989;115:512.
18. Bratincsak A, Reddy VD, Purohit PJ, et al. Coronary artery dilation in acute Kawasaki disease and acute illnesses associated with fever.Pediatr Infect Dis J. 2012;31(9):924-926.
19. CDC. Hospital-associated measles outbreak: Pennsylvania, March – April 2009. MMWR Morb Mortal Wkly Rep. 2012;61(2):30-32.
20. Roberts AL, Connolly KL, Kirse DJ, et al. Detection of group A Streptococcus in tonsils from pediatric patients reveals high rate of asymptomatic streptococcal carriage. BMC Pediatr. 2012 Jan 9;12:3.
21. Dominguez SR, Friedman K, Seewalk R, et al. Kawasaki disease in a pediatric intensive care unit: a case-control study. Pediatrics.2008;122(4):e786-e790.
22. Huang MY, Gupta-Malhotra M, Huang JJ, et al. Acute-phase reactants and a supplemental diagnostic aid for Kawasaki disease. Pediatr Cardiol. 2010;31(8):1209-1213.
23. Singh-Naz N, Rodriguez W. Adenoviral infections in children. Adv Pediatr Infect Dis. 1996;11:365-388.
24. Newburger JW, Takahashi M, Burns JC, et al. The treatment of Kawasaki syndrome with intravenous gamma globulin. N Engl J Med.1986;315(6):341-347.
25. Son MB, Gauvreau K, Ma L, et al. Treatment of Kawasaki disease: analysis of 27 US pediatric hospitals from 2001 to 2007. Pediatrics.2009;124(1):1-8.
26. Chiyonobu T, Yoshihara T, Mori K, et al. Early intravenous gamma globulin retreatment for refractory Kawasaki disease. Clin Pediatr (Phila).2003;42(3):269-272.
27. Shah I, Prabhu SS. Response of refractory Kawasaki disease to intravenous methylprednisolone. Ann Trop Paediatr. 2009;29(1):51-53.
28. Blaisdell LL, Hayman JA, Moran. Infliximab treatment for pediatric refractory Kawasaki disease. Pediatr Cardiol. 2011;32(7):1023-1027.
29. Senzaki H. Long-term outcome of Kawasaki disease. Circulation. 2008;118(25);2763-2772.
30. Lee KY, Lee HS, Hong JH, et al. High-dose intravenous immunoglobulin downregulates the activated levels of inflammatory indices except erythrocyte sedimentation rate in acute stage of Kawasaki disease. J Trop Pediatr. 2005;51(2):98-101.
This illness is the most common cause of acquired heart disease in children in developed nations (with rheumatic fever remaining a more common cause in developing countries).1 The clinical features of KD are self-limited and will resolve even without appropriate therapy, but the patient can be left with significant coronary artery abnormalities.
A high index of clinical suspicion is needed to make a diagnosis of KD. Early diagnosis is critical because early treatment with IV gamma globulin can prevent coronary artery inflammation.2,3 Additionally, KD is potentially fatal; deaths continue to occur, even in the era of gamma globulin therapy.4 KD-associated mortality rates are fortunately very low—especially in Japan, where awareness of the illness is high, and diagnosis and treatment are usually prompt.5,6 Fatalities more commonly occur in children in whom the diagnosis is “missed,” not considered, or delayed.4Additionally, morbidity can be substantial in children who survive but are left with significant coronary artery abnormalities.7
KD has the highest incidence in children of Asian, particularly Japanese, descent: about 1 in 100 Japanese children develop KD by age 5 years.5Boys are more commonly affected than girls, at a ratio of 3 to 2.6 Also of note is that there is a 10-fold increased risk for KD in children with an affected sibling and a twofold increased risk for those with a previously affected parent.8,9 High incidence persists in Japanese children who have adopted a Western lifestyle10; thus, genetics are clearly a factor in KD incidence. However, KD occurs in all ethnic and racial groups worldwide. In areas with a low population of persons of Asian descent, most cases of KD occur in non-Asian children.11 In general, incidence in white children is one-tenth of that in Asian children, with an intermediate incidence in black and Hispanic children.
Clinical and epidemiologic features of KD strongly suggest an infectious cause, but no specific etiologic agent has yet been identified.12 The responsible agent could be a “new,” currently undiscovered virus.13
Patient Presentation and History
KD usually occurs in previously healthy children. Generally, the parent of a child with KD notices that the child seems more ill than is compatible with a typical viral illness. Though not included in the classic diagnostic criteria, marked irritability is very common in KD, particularly in infants. The history generally includes abrupt onset of fever that persists daily, with emergence of other KD signs and symptoms. On occasion, the parent will observe red eyes or a rash prior to fever onset.
In KD, the known signs and symptoms of illness may not all be present simultaneously (see figure). If a child presents to the clinician on day 7 of fever and has red cracked lips, red, swollen hands and feet, and conjunctival injection, and the parent states that the child had a red rash from day 2 through day 5 of illness that is no longer present on examination, the history of rash can be useful in making a diagnosis of KD.14 Such a patient should be considered to have prolonged fever with physical examination revealing four of the five other known clinical features—fulfilling the classic diagnostic criteria for KD.
Criteria for Diagnosis of KD
KD is recognized to occur in two forms: classic and incomplete (“atypical”) cases. Children with classic cases have prolonged fever and at least four of the five clinical findings as described by the American Heart Association (AHA) Committee on Endocarditis, Rheumatic Fever, and Kawasaki Disease14 (see Table 1,14). Patients with incomplete cases have prolonged fever with fewer than four of the five known clinical findings.
Classic and incomplete cases share the same laboratory profile and pathologic features. At present, the development of coronary artery abnormalities is the only means by which a patient with an incomplete case can be confirmed to have had KD. However, because early treatment can reduce the prevalence of coronary artery abnormalities,2 timely diagnosis of either a classic or an incomplete case of KD is essential. Use of the AHA Committee algorithm14 can help the clinician make this diagnosis.
Classic KD
Fever
Fever in children with KD is generally high-spiking, usually reaching 39°C to 40°C daily, and intermittent, with fever usually occurring several times per day, and normal temperatures between fevers. Classic diagnostic criteria require that fever be present for at least five days, but KD can be diagnosed prior to the fifth day of illness by an experienced clinician. Duration of fever is the single best predictor of the development of coronary artery abnormalities,15 and temperatures should be carefully monitored, rectally or orally, in any child in whom a diagnosis of KD is being considered.
Oral Mucosal Changes
Erythema of the lips, mouth, and pharynx are common findings. The lips can be so dry and chapped that they bleed. Oral ulcerations are not a feature of KD, and pharyngeal exudate is not present. A “strawberry” tongue, with prominent papillae on an erythematous base, may be observed.14
Conjunctival Injection
Redness of the bulbar conjunctiva (involving the globe of the eye rather than the palpebral conjunctiva of the eyelid) is a notable feature that can persist for weeks in some patients. There is generally minimal to no exudate. Children with KD may experience light sensitivity, likely related to anterior uveitis, which can be a feature of KD.14
Changes in the Hands and Feet
Redness and swelling of the hands and feet can be clinically striking and quite painful for the child. As a result, children with KD who experience this development will often refuse to walk and may refuse to hold objects. These are unusual complaints in minor viral illnesses of childhood and should heighten the clinician’s suspicion for KD. Some children with KD have more noticeable involvement of the hands or the feet alone, but these extremity changes are bilateral. Swelling of only one hand or foot, for example, should raise suspicion for a bacterial infection, such as cellulitis, arthritis, or osteomyelitis, and reduce suspicion for KD.
Desquamation of the fingers and toes, beginning just beneath the nailbeds, is commonly observed in the subacute phase of KD, at about two to three weeks after onset of fever.14 Therefore, this finding is not useful in making a diagnosis of KD in the first week of illness—the optimal time for therapy to be initiated.
Rash
Rash in KD generally takes one of three forms. Most common is an erythematous, maculopapular rash on the trunk and extremities. Some children have a scarlatiniform rash on the trunk and extremities, prompting an initial concern for group A streptococcal infection. Other children with KD have erythema multiforme, with typical target lesions. Bullae and pustules are not observed in KD, although on rare occasions, a very fine micropustular rash can be observed.14
Of note, some children with KD have a significant rash in the groin area, which can be confused with candidal diaper dermatitis. This rash can occur in toilet-trained children as well as those still in diapers; it is often associated with desquamation of the affected skin, which is not typical of candidal dermatitis. In any child with prolonged fever and a desquamating erythematous groin rash, it is essential to make a careful examination for other features of KD.
Cervical Lymphadenopathy
This feature of KD is the least commonly observed, particularly in infants.16 In some older children, however, it is the most striking feature and can lead to a misdiagnosis of bacterial cervical adenitis.14 Children with this misdiagnosis are often treated with antibiotics with no response and delay of the correct diagnosis. Careful examination will usually reveal other clinical features of KD. Small lymph nodes are commonly palpated in the neck of young children; the lymph node should be > 1.5 cm in diameter to fulfill this criterion, according to the AHA Committee criteria for diagnosing KD.14 Lymphadenopathy, when present, tends to involve primarily the anterior cervical nodes overlying the sternocleidomastoid muscles.17
Incomplete KD
Patients with incomplete or “atypical” KD have fever with fewer than four of the other clinical findings. The term atypical does not indicate the presence of clinical findings that are not characteristic of KD. The AHA Committee14 recommends that a diagnosis of incomplete KD be considered in a child with fever for five days or longer with at least two other clinical features of KD, and with compatible laboratory and/or echocardiographic findings, as shown in Table 1.14
Laboratory Workup
In the absence of a known pathologic cause of KD, no definitive diagnostic test exists. However, certain laboratory findings, shown in Table 214, are considered characteristic of the diagnosis. For example, a low peripheral white blood cell count with lymphocyte predominance is not considered a compatible laboratory profile for KD.
Generally, a complete blood count with differential, C-reactive protein (CRP), erythrocyte sedimentation rate (ESR), liver function tests, and urinalysis are ordered. On diagnosis of KD, an echocardiogram should also be obtained to determine whether early coronary artery dilation is present, and to serve as a comparison for future studies.14,18
Diagnosis
In the presence of the previously described diagnostic criteria, a diagnosis of KD can be made. Patients with a presentation suggestive of incomplete KD should be treated for the illness if they have elevated CRP or ESR and other compatible laboratory or echocardiographic features shown in Table 1.14 In all children with a history of prolonged fever, rash and nonpurulent conjunctivitis—especially those younger than 1 year but including adolescents—KD should be included in the differential diagnosis.
Several other illnesses should be considered in the differential diagnosis of KD.
Measles, in an unimmunized or immigrant child, is a diagnosis that should be considered. In measles, rash usually begins on the face and behind the ears, then progresses to involve the entire body. In patients with measles in whom the rash has just appeared, Koplik spots (small white spots on the reddened oral mucosa near the molars) may be visible. Patients with measles have the classic triad of cough, coryza (rhinorrhea), and conjunctivitis; conjunctivitis is generally exudative.19
Scarlet fever should be considered, along with possible KD, in patients with a scarlatiniform rash. Because in some geographic areas 20% to 30% of children can be carriers of group A streptococcus, a positive throat culture could indicate acute infection or the carrier state.20 Treatment with penicillin for 24 hours generally clarifies the diagnosis; if the patient fails to improve on this therapy, KD should once again be considered.
Toxic shock syndrome can share some clinical features with severe KD, and patients with KD can experience shock.21 The presence of a staphylococcal or streptococcal focus of infection indicates toxic shock syndrome as the diagnosis, whereas the presence of coronary dilation on echocardiography points to KD.
Drug reactions can sometimes be confused with KD, and some patients with KD have a rash that may itch. The presence of facial and eyelid swelling is more likely to suggest a drug reaction than KD, and laboratory testing generally shows markedly elevated acute-phase reactants in KD,22 compared with those who are experiencing a drug reaction.
Viral illnesses, such as enterovirus infection, can share clinical features of KD, but usually will yield the elevated neutrophil count and acute-phase reactant elevations found in patients with KD.14,22 Children with adenovirus infection usually present with exudative pharyngitis and conjunctivitis.23
Once Diagnosis Is Confirmed
Patients in whom a diagnosis of KD is confirmed should be treated in a hospital where pediatric echocardiography is available, including measurement of coronary artery diameters in small children. In these patients, a Z score is calculated, representing standard deviations exceeding mean coronary artery dimensions, based on the patient’s body surface area. A child with a right proximal or left anterior descending coronary artery Z score greater than 2.5 (ie, 2.5 standard deviations above the mean) is confirmed to have an enlarged coronary artery.14,18
Echocardiography technicians must be well trained and experienced to be able to perform the necessary measurements, particularly in febrile, ill, uncooperative children. Only a pediatric cardiologist is qualified to interpret the studies.14
Treatment and Management
As soon as possible after a diagnosis of KD is made, patients should be treated with IV gamma globulin, 2 g/kg over 8 to 12 hours, and oral aspirin 80 to 100 mg/kg/d, divided for administration every 6 hours.3,14,24 High-dose aspirin is needed for its anti-inflammatory effect; its benefits for patients with KD appear to greatly outweigh the extremely low risk for Reye syndrome associated with aspirin use. In the rare patient with KD who has concurrent, documented influenza or varicella (and thus an increased risk for Reye syndrome with aspirin use), aspirin should be withheld, and an expert in KD should be consulted.
Most patients with KD respond rapidly to this therapeutic approach, with resolution of fever and improvement in clinical signs within 24 to 48 hours. However, 15% to 20% of patients do not respond to this initial treatment.14,25 The optimal therapy for such patients is unknown, but a second 2 g/kg dose of IV gamma globulin, IV methylprednisolone 30 mg/kg for 1 to 3 days, or IV infliximab 5 mg/kg are additional options.25-28
Patients who still do not respond may require additional therapies (eg, steroid therapy, infliximab, perhaps other immunosuppressive therapies), as these children are at high risk for coronary artery abnormalities. Referral of such patients to a center with extensive experience in caring for patients with KD should be considered.
Classic treatment studies have demonstrated a reduction in the prevalence of coronary artery abnormalities two months after illness onset from 18% in patients who were treated with aspirin alone to 4% in those who received IV gamma globulin and aspirin.24 However, more recent research, using newer algorithms to calculate normal coronary artery size based on body surface area, reveals that coronary artery dilation is more common in KD than previously recognized.2 Specifically, about 18% of patients with KD who present within the first 10 days of fever onset have already experienced some degree of coronary artery dilation before therapy is initiated. This highlights the importance of earlier diagnosis and treatment.
On the 14th day after fever onset (or 2 to 3 days after fever has resolved), aspirin is reduced to a single daily dose of 3 to 5 mg/kg.14 Aspirin use is continued for its antithrombotic effect until the ESR has normalized, provided echocardiography results have remained normal throughout. Aspirin therapy may be required indefinitely in patients who develop coronary artery aneurysms; in severe cases, additional antiplatelet or antithrombotic therapies may be required to prevent aneurysmal thrombosis.
Patient Education
A diagnosis of KD can be frightening. In the United States, most parents of a child diagnosed with KD have never heard of the condition and will need education and support. Once the child has been discharged and the parents discuss the illness with friends and acquaintances, they generally become aware that other children they know have had KD. The illness is not rare, but the public is generally unfamiliar with it.
Parents can be referred to the Web sites of the American Heart Association (www.heart.org/HEARTORG/Conditions/More/CardiovascularConditionsof Childhood/Kawasaki-Disease_UCM_308777_Article.jsp) and the American Academy of Pediatrics (see www.healthychildren.org), where educational materials about KD are posted. In the US, the Kawasaki Disease Foundation (http://www.kdfoundation.org/) also provides information and support to families with children affected by KD.
Follow-Up
Children with KD should undergo echocardiography at diagnosis, at 2 to 3 weeks, and again at 6 to 8 weeks after fever onset.14 At that point, a child with KD who has developed no evidence of coronary artery abnormalities will require no additional echocardiograms, as no evidence of late-onset, KD-associated coronary artery dilation has been reported.14,29 Children who have developed coronary artery dilation, however, will require further studies, as recommended by their pediatric cardiologist.
Laboratory testing should be performed at diagnosis, and acute-phase reactants (eg, CRP) should be monitored after discharge until normal levels are reached. Follow-up visits (often scheduled at 2 to 3 weeks and 6 to 8 weeks after illness onset) should consist of physical examination and laboratory testing, generally including a complete blood count and CRP level. This value often normalizes within 2 to 3 weeks after illness onset and can be helpful in monitoring clinical response, particularly in patients with persistent fever who require continuing therapy. Because the ESR rises transiently after IV gamma globulin therapy,30 it is not considered useful in monitoring response to therapy; the ESR usually normalizes by 6 to 8 weeks after fever onset.
Conclusion
KD is a potentially life-threatening illness of early childhood that should be considered in the differential diagnosis of any child with prolonged, unexplained fever. In KD, fever, rash, conjunctival injection, oral mucosal changes, extremity changes, and cervical adenopathy are self-limited, but any resulting coronary artery dilation and aneurysms can persist lifelong.
A high index of clinical suspicion is required to make a diagnosis of KD. Children with incomplete KD may present with fever and fewer than four other known clinical features; these children must undergo laboratory and echocardiographic studies for a diagnosis to be confirmed. Early diagnosis is essential for effective therapy with IV gamma globulin and aspirin to be administered before KD-associated coronary artery abnormalities can develop.
Additional research is needed to identify the cause of the disease, making it possible to develop a specific diagnostic test and etiology-targeted therapy.
References
1. Taubert KA, Rowley AH, Shulman ST. Seven-year national survey of Kawasaki disease and acute rheumatic fever. Pediatr Infect Dis J.1994;13(8):704-708.
2. Dominguez SR, Anderson MS, Eladawy M, Glodé MP. Preventing coronary artery abnormalities: a need for earlier diagnosis and treatment of Kawasaki disease. Pediatr Infect Dis J. 2012 Jul 3. [Epub ahead of print]
3. Newburger JW, Takahashi M, Beiser AS, et al. A single intravenous infusion of gamma globulin as compared with four infusions in the treatment of acute Kawasaki syndrome. N Engl J Med. 1991;324(23):1633-1639.
4. Orenstein JM, Shulman ST, Fox LM, et al. Three linked vasculopathic processes characterize Kawasaki disease: a light and transmission electron microscopic study. PLoS One. 2012;7(6):e38998. Epub 2012 Jun 18.
5. Nakamura Y, Yashiro M, Uehara R, et al. Epidemiologic features of Kawasaki disease in Japan: results of the 2009-2010 nationwide survey. J Epidemiol. 2012;22(3):216-221.
6. Nakamura Y, Yanagawa H, Harada K, et al. Mortality among persons with a history of Kawasaki disease in Japan: the fifth look. Arch Pediatr Adolesc Med. 2002;156(2):162-165.
7. Suda K, Iemura M, Nishiono H, et al. Long-term prognosis of patients with Kawasaki disease complicated by giant coronary aneurysms: a single-institution experience. Circulation. 2011;123(17):1836-1842.
8. Fujita Y, Nakamura Y, Sakata K, et al. Kawasaki disease in families. Pediatrics. 1989;84(4):666-669.
9. Uehara R, Yashiro M, Nakamura Y, Yanagawa H. Kawasaki disease in parents and children. Acta Paediatr. 2003;92(6):694-697.
10. Dean AG, Melish ME, Hicks R, Palumbo NE. An epidemic of Kawasaki syndrome in Hawaii. J Pediatr. 1982;100(4):552-557.
11. Shulman ST, McAuley JB, Pachman LM, et al. Risk of coronary abnormalities due to Kawasaki disease in urban area with small Asian population. Am J Dis Child. 1987;141(4):420-425.
12. Rowley AH, Baker SC, Orenstein JM, Shulman ST. Searching for the cause of Kawasaki disease: cytoplasmic inclusion bodies provide new insight. Nat Rev Microbiol. 2008;6(5):394-401.
13. Rowley AH, Baker SC, Shulman ST, et al. Ultrastructural, immunofluorescence, and RNA evidence support the hypothesis of a “new” virus associated with Kawasaki disease. J Infect Dis. 2011;203(7):1021-1030.
14. Newburger JW, Takahashi M, Gerber MA, et al. Diagnosis, treatment, and long-term management of Kawasaki disease: a statement for health professionals from the Committee on Rheumatic Fever, Endocarditis, and Kawasaki Disease, Council on Cardiovascular Disease in the Young, American Heart Association. Pediatrics. 2004;114(6):1708-1733.
15. Koren G, Lavi S, Rose V, Rowe R. Kawasaki disease: review of risk factors for coronary aneurysms. J Pediatr. 1986;108(3):388-392.
16. Sung RY, Ng YM, Choi KC, et al; Hong Kong Kawasaki Disease Study Group. Lack of association of cervical lymphadenopathy and coronary artery complications in Kawasaki disease. Pediatr Infect Dis J. 2006;25(6):521-525.
17. April MM, Burns JC, Newburger JW, Healy GB. Kawasaki disease and cervical adenopathy. Arch Otolaryngol Head Neck Surg. 1989;115:512.
18. Bratincsak A, Reddy VD, Purohit PJ, et al. Coronary artery dilation in acute Kawasaki disease and acute illnesses associated with fever.Pediatr Infect Dis J. 2012;31(9):924-926.
19. CDC. Hospital-associated measles outbreak: Pennsylvania, March – April 2009. MMWR Morb Mortal Wkly Rep. 2012;61(2):30-32.
20. Roberts AL, Connolly KL, Kirse DJ, et al. Detection of group A Streptococcus in tonsils from pediatric patients reveals high rate of asymptomatic streptococcal carriage. BMC Pediatr. 2012 Jan 9;12:3.
21. Dominguez SR, Friedman K, Seewalk R, et al. Kawasaki disease in a pediatric intensive care unit: a case-control study. Pediatrics.2008;122(4):e786-e790.
22. Huang MY, Gupta-Malhotra M, Huang JJ, et al. Acute-phase reactants and a supplemental diagnostic aid for Kawasaki disease. Pediatr Cardiol. 2010;31(8):1209-1213.
23. Singh-Naz N, Rodriguez W. Adenoviral infections in children. Adv Pediatr Infect Dis. 1996;11:365-388.
24. Newburger JW, Takahashi M, Burns JC, et al. The treatment of Kawasaki syndrome with intravenous gamma globulin. N Engl J Med.1986;315(6):341-347.
25. Son MB, Gauvreau K, Ma L, et al. Treatment of Kawasaki disease: analysis of 27 US pediatric hospitals from 2001 to 2007. Pediatrics.2009;124(1):1-8.
26. Chiyonobu T, Yoshihara T, Mori K, et al. Early intravenous gamma globulin retreatment for refractory Kawasaki disease. Clin Pediatr (Phila).2003;42(3):269-272.
27. Shah I, Prabhu SS. Response of refractory Kawasaki disease to intravenous methylprednisolone. Ann Trop Paediatr. 2009;29(1):51-53.
28. Blaisdell LL, Hayman JA, Moran. Infliximab treatment for pediatric refractory Kawasaki disease. Pediatr Cardiol. 2011;32(7):1023-1027.
29. Senzaki H. Long-term outcome of Kawasaki disease. Circulation. 2008;118(25);2763-2772.
30. Lee KY, Lee HS, Hong JH, et al. High-dose intravenous immunoglobulin downregulates the activated levels of inflammatory indices except erythrocyte sedimentation rate in acute stage of Kawasaki disease. J Trop Pediatr. 2005;51(2):98-101.
Autoimmune Hemolytic Anemia
The autoimmune hemolytic anemias (AIHA) are rare but important hematologic diseases. They can range in severity from mildly symptomatic illness to a rapidly fatal syndrome. The incidence of AIHA is estimated to be between 0.6 and 3 cases per 100,000 persons. AIHA is mediated by antibodies, and in the majority of cases immunglobulin (Ig) G is the mediating antibody. This type of AIHA is referred to as "warm" AIHA because IgG antibodies bind best at body temperature. "Cold" AIHA is mediated by IgM antibodies, which bind maximally at temperatures below 37°C. This manual reviews the most common types of AIHA, with emphasis on diagnosis and treatment.
To read the full article in PDF:
The autoimmune hemolytic anemias (AIHA) are rare but important hematologic diseases. They can range in severity from mildly symptomatic illness to a rapidly fatal syndrome. The incidence of AIHA is estimated to be between 0.6 and 3 cases per 100,000 persons. AIHA is mediated by antibodies, and in the majority of cases immunglobulin (Ig) G is the mediating antibody. This type of AIHA is referred to as "warm" AIHA because IgG antibodies bind best at body temperature. "Cold" AIHA is mediated by IgM antibodies, which bind maximally at temperatures below 37°C. This manual reviews the most common types of AIHA, with emphasis on diagnosis and treatment.
To read the full article in PDF:
The autoimmune hemolytic anemias (AIHA) are rare but important hematologic diseases. They can range in severity from mildly symptomatic illness to a rapidly fatal syndrome. The incidence of AIHA is estimated to be between 0.6 and 3 cases per 100,000 persons. AIHA is mediated by antibodies, and in the majority of cases immunglobulin (Ig) G is the mediating antibody. This type of AIHA is referred to as "warm" AIHA because IgG antibodies bind best at body temperature. "Cold" AIHA is mediated by IgM antibodies, which bind maximally at temperatures below 37°C. This manual reviews the most common types of AIHA, with emphasis on diagnosis and treatment.
To read the full article in PDF:
Patient abusing alcohol or drugs? Help starts with a single question
• Screen patients for substance use disorders, with a single (validated) question for alcohol and another for drugs. A
• Follow a positive screen for alcohol with an assessment to distinguish between hazardous drinking and drinking that is indicative of alcohol dependence. C
• Approach a substance use disorder as you would any chronic medical condition, seeking to engage the patient to encourage behavior change. Motivational interviewing is a useful tool. C
• Consider pharmacotherapeutic options for patients with alcohol or drug dependence. A
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
Episodic heavy drinking, like alcoholism and drug addiction, is increasingly recognized as a medical problem that primary care physicians can, and should, address.1 But it is rarely the chief reason for an office visit. Nor is it a subject patients are likely to bring up.
However, patients are generally willing to talk to a trusted doctor who asks about their use (or misuse) of alcohol or other substances. And primary care physicians can do much to help—with brief interventions, a growing armamentarium of pharmacotherapy, and referrals as needed. In the pages that follow, you’ll find easy-to-use screening tools and effective intervention strategies.
SCREENING NEEDN'T BE TIME-CONSUMING
Screening for substance use isn’t difficult. In fact, it can usually be accomplished with 2 targeted questions—one for alcohol use and one for drugs.
Alcohol. Two single-question screens to detect hazardous drinking have been validated, despite having different parameters. Ask either:
Q: When was the last time you had more than ____ drinks (4 for women and 5 for men) in one day?
Q: How many times in the past year have you had ___ or more drinks (4 for women and 5 for men) in one day?
For the first question, any answer within the past 3 months is a positive screen for hazardous drinking.2 For the second, anything other than zero is positive.3,4
Initial screening can also be done with the AUDIT-C (TABLE), a validated short (3-question) version of the Alcohol Use Disorders Identification Test that can be self-administered.5,6
Drugs. Only one single-question screen for drug use has been validated:
Q: How many times in the past year have you used an illegal drug or taken a prescription medication for nonmedical reasons?
Any answer other than never is a positive screen for hazardous drug use.7
CASE Jason F, a healthy and fit 28-year-old, has been your patient, along with his family, for years. He’s in your office because of a knee injury he incurred while running, and you take a moment to ask him, for the first time, how much he drinks and whether he takes drugs. His answer—that he drinks 3 or 4 times a week and often has multiple drinks at parties or nights out with the guys—takes you a bit by surprise.
Now what?
TABLE
3-question AUDIT-C screen for alcohol dependence5,6
| 1. How often do you have a drink containing alcohol? | ||
| 0 Never | 3 2 to 3 times a week | |
| 1 Monthly or less | 4 4 or more times a week | |
| 2 2 to 4 times a month | ||
| 2. How many drinks containing alcohol do you have on a typical day when you are drinking? | ||
| 0 I don’t drink. | 2 5 or 6 | |
| 0 1 or 2 | 3 7 to 9 | |
| 1 3 or 4 | 4 10 or more | |
| 3. How often do you have 6 or more drinks on one occasion? | ||
| 0 Never | 3 Weekly | |
| 1 Less than monthly | 4 Daily or almost daily | |
| 2 Monthly | ||
| Scoring the AUDIT-C | ||
| Alcohol dependence | ||
| Men | Women | |
| Threshold score | 5 | 4 |
| Sensitivity (%) | 80 | 76 |
| Specificity (%) | 74 | 78 |
| AROC | 0.769 | 0.767 |
| AROC, area under the receiver operating characteristic curve; AUDIT, Alcohol Use Disorders Identification Test. | ||
TELL ME MORE ABOUT IT
It is important to respond to a positive screen by requesting more information. In the conversation that ensues, the patient may provide the details you need to determine whether the drinking or drug use is indicative of a diagnosable substance use disorder, an umbrella term for alcohol or drug abuse and alcohol or drug dependence.
Alcohol or drug abuse vs dependence. Criteria for alcohol or drug abuse in the Diagnostic and Statistical Manual of Mental Disorders, 4th ed, text revision (DSM-IV-TR) include risky behavior, such as drinking and driving; problems with work and/or close relationships; or run-ins with the law, such as an arrest for driving while intoxicated. Criteria for alcohol or drug dependence include the inability to cut down or stop using the substance; evidence of tolerance and withdrawal; spending more time ingesting the substance; increasing attention to substance use while interest in other activities diminishes; and continued use despite recurrent problems.8
Tools zero in on extent of problem
To learn more about your patient’s situation, consider using the following criteria and tools:
DSM criteria. Ask the following 2 questions, which are among DSM-IV-TR criteria for alcohol dependence:
- How many times in the last year have you had a lot more to drink than you intended?
- How many times in the last year have you been drinking in situations where it could have been hazardous, where you could have caused an accident or gotten hurt?
Any answer other than zero to either question is suggestive of a substance use disorder. In exploratory analyses, this approach had positive likelihood ratios of 4.7 to 16 and negative likelihood ratios of 0.05 to 0.30.9,10
Although the above questions refer to alcohol use, they could be revised to learn more about a patient’s use of marijuana or other drugs, as well. (There are few tools for the assessment of drug use, because any illegal or nonmedical use of controlled substances has clear risks of major harm.)
CAGE. Another tool that is effective in assessing alcohol use is the 4-question CAGE—an acronym for Cut down, Annoyed, Guilty, and Eye opener:
- Have you ever felt that you should cut down on your drinking?
- Have people annoyed you by criticizing your drinking?
- Have you ever felt bad or guilty about your drinking?
- Have you ever had a drink in the morning to get rid of a hangover?
One meta-analysis found that a positive CAGE test—ie, a positive response to one or more of the questions—had a sensitivity of 0.85 and a specificity of 0.78 in identifying alcohol dependence in a primary care setting (using DSM criteria as the gold standard).11
AUDIT. This 10-item tool, a longer version of the AUDIT-C (available at http://www.medstudentlearning.com/node/6556), can also be used to determine the extent of alcohol use. This test provides detailed information about the quantity and frequency of alcohol use; however, it does not clearly distinguish between hazardous drinking and alcohol use disorders.12
If the patient is a teen
Assessment methods can be adjusted without difficulty to fit the age of the patient. The National Institute on Alcohol Abuse and Alcoholism has published the Alcohol Screening and Brief Intervention for Youth: A Practitioner’s Guide, available at http://www.niaaa.nih.gov/Publications/EducationTrainingMaterials/Pages/YouthGuide.aspx. The 6-question CRAFFT (for Car, Relax, Alone, Forget, Friends, Trouble) is a validated tool designed to assess adolescents’ use of both alcohol and drugs (http://www.ceasar-boston.org/clinicians/crafft.php).13,14
CASE You give Mr. F the CAGE test, and he answers No to all 4 questions. You conclude that while his drinking may be hazardous, he does not appear to have alcohol abuse or dependence.
FOLLOW UP WITH A BRIEF INTERVENTION
For decades, evidence has shown that brief interventions are often effective in helping hazardous drinkers like Mr. F cut back to safer levels.15-18 In some cases, the impact has been great enough to reduce health care and societal costs for up to 4 years19 and to cut the risk of alcohol-related death by about half.20 As a result, the US Preventive Services Task Force has given a B rating to counseling to reduce alcohol misuse by primary care providers.21 (There is less evidence that brief interventions are effective for drug problems,22 or in settings other than primary care.23)
Treat drug/alcohol problems
If you determine that your patient is engaging in hazardous alcohol or drug use or has a diagnosable substance use disorder, you do not have to drop everything else or treat it as an acute event. What matters is long-term success, which is best achieved by partnering with the patient.
Start by approaching drug and alcohol problems as you would a case of newly elevated blood pressure. Bring up the problem, seeking to engage the patient in addres- sing it.
If he or she does not agree to quit or cut back on drinking the first time you broach the subject, don’t be surprised or discouraged. Keep in mind that patients do not always respond positively to advice about handling chronic medical conditions either, particularly at first, and that you’ll be working together over time. What’s important, in the jargon of the Stages of Change model,24 is to help the patient move from precontemplation to contemplation, and perhaps beyond that to planning or action.
Use motivational interviewing to partner with patients
Motivational interviewing is useful in helping patients change health-related behaviors. The technique, which is not hard to learn or apply, is based on the recognition that a simple shift in style toward a guiding (rather than directive) approach can often reap benefits that are immediately apparent. 25-28
Motivational Interviewing: Helping People Change (3rd ed, by William R. Miller and Stephen Rollnick; Guilford Press, 2013) is an excellent resource for clinicians who wish to master this technique. An online tutorial in screening and brief intervention for alcohol or drug misuse is available free at https://adept.missouri.edu. Video demonstrations of motivational interviewing to address these issues are also available here ; to access them, click on “Training”, then on “Go to SBIRT videos”).
CASE Before Mr. F’s visit is concluded, you initiate a conversation about alcohol use, stating: “As your doctor, I’m concerned that the amount of alcohol you’re drinking could be hazardous to your health. I recommend that you cut down to no more than 4 drinks in any one day and to no more than 14 drinks a week.” You make it clear that change is up to him, and ask what he thinks about what you’ve said.
You also schedule a return visit in one month, at which time you will continue the conversation.
PHARMACOTHERAPY IS A USEFUL TOOL
Increasingly, alcohol and drug dependence—like other chronic conditions—can be effectively addressed with medication.
Drugs to treat alcohol dependence
Naltrexone. A daily dose of naltrexone, starting at 25 mg daily for a few days and going as high as 100 mg/d, can help patients with alcohol dependence limit their drinking to safe levels (number needed to treat [NNT]=9).29 This will reduce the risk of alcohol-related harm while the patient considers quitting.
The most common adverse effect is nausea, but a low starting dose may alleviate it. Naltrexone, also available as a 380-mg intramuscular (IM) depot injection once every 4 weeks, is an opioid antagonist and should not be given to any patient who’s taking opioids.
A 2010 Cochrane review found only 4 trials of naltrexone IM, and failed to show significant reductions in drinking.29 But post hoc analyses of trials of both oral and IM naltrexone found that those in which compliance was assured (either by direct observation or IM administration) had better outcomes than those in which it was not.30 Another post hoc analysis found that patients whose alcohol dependence was more severe derived greater benefits from the drug than those who were less severely affected.31
Acamprosate. Two 333-mg pills tid can help newly abstinent drinkers remain alcohol-free (NNT=9).32,33 The most common adverse effect is diarrhea, which may subside with continued use.
Combining acamprosate and naltrexone does not appear to be more effective than either drug alone. In a recently published meta-analysis comparing the 2 drugs, those taking acamprosate had slightly better rates of abstinence from alcohol, while naltrexone was slightly better in reducing heavy drinking.34
Disulfiram Unlike naltrexone and acamprosate, which work by altering the brain’s reward circuits, disulfiram blocks metabolism of ethanol, leading to the accumulation of a toxic metabolite and its punishing syndrome. The major problem with the drug is noncompliance, which can be addressed by enlisting the help of a caregiver or partner to ensure that it is taken daily. 35-37
Other medications that have been tested (though not approved by the US Food and Drug Administration [FDA]) as a treatment for alcohol dependence include:
- topiramate, which has been found to have modest efficacy in increasing the number of abstinent days and decreasing heavy-drinking days;38
- baclofen, which has shown efficacy in small clinical trials;39 and
- ondansetron, which has been shown to effectively treat early-onset alcohol dependence.40,41
For patients with depression and alcohol dependence, the combination of naltrexone and sertraline has been found to be superior to either drug by itself—and to have fewer adverse effects. 42 Gabapentin and lorazepam have been compared in treating alcohol withdrawal, with gabapentin resulting in greater efficacy and fewer adverse effects than lorazepam.43,44
Pharmacotherapy for drug abuse, dependence
For methamphetamine abuse and dependence. Two randomized clinical trials have studied medications for methamphetamine abuse and dependence. In one small study, topiramate did not increase the proportion of patients who achieved abstinence, but in a post hoc subgroup analysis, it did appear to help newly abstinent patients avoid relapse.45
In another study, mirtazapine significantly decreased the proportion of patients whose weekly urine tests were positive for methamphetamine (from 73% to 44%); no significant change was found among those on placebo.46
Both drugs were well tolerated, but compliance was low in both trials despite weekly counseling. Each has only one clinical trial to support its use, and neither has FDA approval for addiction treatment.
For marijuana dependence. In a study of 50 people seeking treatment for marijuana dependence, gabapentin 400 mg 3 times a day significantly improved the proportion reporting no cannabis use and whose urine tested negative for the drug.47
Another recent trial randomized adolescents dependent on cannabis to placebo or N-acetylcysteine 1200 mg twice a day. Those on the active drug were 2.4 times more likely than those on placebo to have negative urine tests with a number needed to treat of 7.48 Both trials ran for about 3 months. Neither drug is FDA approved to treat marijuana dependence.
For opioid dependence. As maintenance medication for patients dependent on opioids, both methadone and buprenorphine have been shown to reduce the use of illicit opioids, lower mortality, and improve retention compared with treatment without medication.49 Methadone would be a better choice than buprenorphine, which is a partial agonist with a ceiling on both its good (eg, stopping craving) and bad (eg, overdose risk) effects. In an open-label observational study of patients’ preferences, those who chose methadone maintenance over buprenorphine were twice as likely to remain in treatment.50 Both drugs are FDA-approved for treating opioid dependence.
Methadone is a full agonist that can be given for opioid dependence only in a federally licensed methadone maintenance clinic. It has been shown to reduce the use of other opioids, reduce criminal behaviors,51 improve function in many areas,52 and reduce mortality.53 In a cohort study of Massachusetts Medicaid data, methadone reduced mortality, which was 75% higher among those receiving abstinence-based treatment.52,54
Buprenorphine (alone and in combination with naloxone), effectively reduces the use of illicit opioids and improves functional status.55-57
Naltrexone, an opioid antagonist, may be effective in the treatment of opioid dependence. As with disulfiram for alcohol dependence, a major limitation of naltrexone for opioid dependence is noncompliance. But once a patient has been on oral naltrexone, he or she can be switched to naltrexone IM, which can be administered every 4 weeks. A Cochrane review published in 2011 found no evidence that naltrexone was superior to placebo, 58 but since then another randomized clinical trial has been published that found naltrexone 380 mg IM every 4 weeks was superior to IM placebo. Patients on naltrexone were more likely to remain in treatment and have opioid-free urine tests, and reported less craving for opioids.59 Both oral and depot injection naltrexone are FDA approved for treatment of opioid dependence. No comparisons of naltrexone vs either methadone or buprenorphine have been published.
To learn more about pharmacotherapy for opioid dependence, see “Diagnosing and treating opioid dependence” (J Fam Pract. 2012;61:588-596).
DEALING WITH THE CHALLENGES
As noted earlier, some patients with substance use disorders, like some patients with depression or hypertension, respond well to care and counseling, and some do not. Just as with other conditions, consultation with a specialist often helps.
A major difference in arranging consultations for patients with substance use disorders, however, is that clinicians who specialize in substance abuse and dependence often work in health care systems that are largely, or entirely, separate from those in which primary care physicians typically work. This, plus the stigma that surrounds problems with substance use, presents barriers to patients, who may shy away from going across town or to another city to see a provider they don’t know for a problem they’re either resistant to “owning” or ashamed of.
Yet it is possible to reach across this divide and make it easier for patients. One way to do that might be to partner with a local alcohol- and drug-treatment program so that your patients are referred, not to a faceless agency, but rather to a specific clinician; you might even call the provider while the patient is in your office so they can “meet.” Another approach, taken by some multispecialty practices, is to add psychotherapists to the staff so that patients can simply walk down the hall to obtain the mental health care they need.
Reaching across this divide is also a useful strategy for primary care physicians, who may welcome opportunities to meet with someone from a local treatment agency, not just for referrals but to learn more about treating patients with substance use problems. The Patient Protection and Affordable Care Act, which cites substance use disorders as one of 6 chronic health conditions that primary care medical homes are expected to address, may lead to better integration of health care systems that address physical health, as well as mental health and substance use disorders.
- Friedman PD. Alcohol use in adults. N Engl J Med. 2013;368:365-373.
- Williams RH, Vinson DC. Validation of a single question screen for problem drinking. J Fam Pract. 2001;50:307-312.
- Smith PC, Schmidt SM, Allensworth-Davies D, et al. Primary care validation of a single-question alcohol screening test. J Gen Intern Med. 2009;74:783-788.
- Dawson DA, Pulay AJ, Grant BF. A comparison of two single-item screeners for hazardous drinking and alcohol use disorder. Alcohol Clin Exp Res. 2010;34:364-374.
- Bush K, Kivlahan DR, Mcdonell MB, et al. The AUDIT alcohol consumption questions (AUDIT-C): an effective brief screening test for problem drinking. Arch Intern Med. 1998;158:1789-1795.
- Dawson DA, Grant BF, Stinson FS, et al. Effectiveness of the derived Alcohol Use Disorders Identification Test (AUDIT-C) in screening for alcohol use disorders and risk drinking in the US general population. Alcohol Clin Exp Res. 2005;29:844-854.
- Smith PC, Schmidt SM, Allensworth-Davies D, et al. A single-question screening test for drug use in primary care. Arch Intern Med. 2010;170:1155-1160.
- American Psychiatric Asociation. Diagnostic and Statistical Manual of Mental Disorders. 4th ed text rev. (DSM-IV-TR). Arlington, Va: American Psychiatric Association; 2000.
- Saha TD, Stinson FS, Grant BF. The role of alcohol consumption in future classifications of alcohol use disorders. Drug Alcohol Depend. 2007;89:82-92.
- Vinson DC, Kruse RL, Seale JP. Simplifying alcohol assessment:two questions to identify alcohol use disorders. Alcohol Clin Exp Res. 2007;31:1392-139.
- Aertgeerts B, Buntinx F, Kester A. The value of the CAGE in screening for alcohol abuse and alcohol dependence in general clinical populations: a diagnostic meta-analysis. J Clin Epidemiol. 2004;57:30-39.
- Johnson JA, Lee A, Vinson DC, et al. Use of AUDIT-based measures to identify unhealthy alcohol use and alcohol dependence in primary care: a validation study. Alcohol Clin Exp Res. 2012;July 26 [Epub ahead of print].
- Center for Adolescent Substance Abuse Research. The CRAFFT Screening Tool. Boston MA:2009 [updated 2012). Available at: http://www.ceasar-boston.org/clinicians/crafft.php. Accessed January 15, 2013.
- Knight JR, Sherritt L, Harris SK, et al. Validity of brief alcohol screening tests among adolescents: a comparison of the AUDIT, POSIT, CAGE, and CRAFFT. Alcohol Clin Exp Res. 2003;27:67-734.
- Wallace P, Cutler S, Haines A. Randomised controlled trial of general practitioner intervention in patients with excessive alcohol consumption. BMJ. 1988;297:663-668.
- Fleming MF, Barry KL, Manwell LB, et al. Brief physician advice for problem alcohol drinkers: a randomized controlled trial in community-based primary care practices. JAMA. 1997;277:1039-1044.
- Bertholet N, Daeppen JB, Wietlisbach V, et al. Brief alcohol intervention in primary care reduces alcohol consumption: Systematic review and meta-analysis. Arch Intern Med. 2005;165:986-995.
- Kaner EFS, Dickinson HO, Beyer F, et al. Effectiveness of brief alcohol interventions in primary care populations. Cochrane Database Syst Rev. 2007(2);CD004148.-
- Fleming MF, Mundt MP, French MT, et al. Brief physician advice for problem drinkers: long-term efficacy and benefit-cost analysis. Alcohol Clin Exp Res. 2002;26:36-43.
- Cuijpers P, Riper H, Lemmers L. The effects on mortality of brief interventions for problem drinking: a meta-analysis. Addiction. 2004;99:839-845.
- U.S.Preventive Services Task Force. Screening and behavioral counseling interventions in primary care to reduce alcohol misuse: recommendation statement. Ann Intern Med. 2004;140:554-556.
- Saitz R, Alford DP, Bernstein J, et al. Screening and brief intervention ffor unhealthy drug use in primary care settings: randomized clinical trials are needed. J Addict Med. 2010;4:131-136.
- Field CA, Baird J, Saitz R, et al. The mixed evidence for brief intervention in emergency departments, trauma care centers, and inpatient hospital settings: what should we do? Alcohol Clin Exp Res. 2010;34:2004-2010.
- DiClemente CC, Prochaska JO. Toward a comprehensive transtheoretical model of change: stages of change and addictive behaviors. In: Miller WR, Heather N, eds. Treating Addictive Behaviors. New York, NY:Plenum Press;1998:3–24.
- Mason P, Butler CC. Health Behavior Change: A Guide for Practitioners. 2nd ed. London UK:Elsevier; 2010.
- Miller WR, Rollnick S. Motivational Interviewing: Preparing People for Change. 2nd ed. New York NY: Guilford Press;2002.
- Hettema J, Steele J, Miller WR. Motivational interviewing. Ann Rev Clin Psych. 2005;1:91-111.
- Rollnick S, Butler CC, Kinnersley P, et al. Motivational interviewing. BMJ. 2010;340:c1900.-
- Rosner S, Hackl-Herrwerth A, Leucht S, et al. Opioid antagonists for alcohol dependence. Cochrane Database Syst Rev. 2010;(12):CD001867.-
- Swift R, Oslin DW, Alexander M, et al. Adherence monitoring in naltrexone pharmacotherapy trials: a systematic review. J Stud Alcohol Drugs. 2011;72:1012-1018.
- Pettinati HM, Silverman BL, Battisti JJ, et al. Efficacy of extended-release naltrexone in patients with relatively higher severity of alcohol dependence. Alcohol Clin Exp Res. 2011;35:1804-1811.
- Mann K, Lehert P, Morgan MY. The efficacy of acamprosate in the maintenance of abstinence in alcohol-dependent individuals: results of a meta-analysis. Alcohol Clin Exp Res. 2004;28:51-63.
- Rosner S, Hackl-Herrwerth A, Leucht S, et al. Acamprosate for alcohol dependence. Cochrane Database Syst Rev. 2010(9);CD004332.-
- Maisel NC, Blodgett JC, Wilbourne PL, et al. Meta-analysis of naltrexone and acamprosate for treating alcohol use disorders: when are these medications most helpful? Addiction. 2012;October 17 [Epub ahead of print].
- Azrin NH. Improvements in the community-reinforcement approach to alcoholism. Behav Res Ther. 1976;14:339-348.
- Azrin NH, Sisson RW, Meyers R, Godley M. Alcoholism treatment by disulfiram and community reinforcement therapy. J Behav Ther Exp Psychiatr. 1982;13:105-112.
- Keane TM, Foy DW, Nunn B, et al. Spouse contracting to increase antabuse compliance in alcoholic veterans. J Clin Psychol. 1984;40:340-344.
- Arbaizar B, Diersen-Sotos T, Gomez-Acebo I, et al. Topiramate in the treatment of alcohol dependence: a meta-analysis. Actas Espanolas de Psiquiatria. 2010;38:8-12.
- Johnson BA. Update on neuropharmacological treatments for alcoholism: scientific basis and clinical findings. Biochem Pharmacol. 2008;75:34-56.
- Johnson BA, Roache JD, Javors MA, et al. Ondansetron for reduction of drinking among biologically predisposed alcoholic patients: a randomized controlled trial. JAMA. 2000;284:963-971.
- Johnson BA, Ait-Daoud N, Seneviratne C, et al. Pharmacogenetic approach at the serotonin transporter gene as a method of reducing the severity of alcohol drinking. Am J Psychiatr. 2011;168:265-275.
- Pettatini HM, Oslin D, Lampman KM, et al. A double-blind, placebo-controlled trial combining sertraline and naltrexone for treating co-occurring depression and alcohol dependence. Am J Psychiatr. 2010;167:668-675.
- Anton RF, Myrick H, Wright TM, et al. Gabapentin combined with naltrexone for the treatment of alcohol dependence. Am J Psychiatr. 2011;168:709-717.
- Myrick H, Malcolm R, Randall PK, et al. A double-blind trial of gabapentin versus lorazepam in the treatment of alcohol withdrawal. Alcohol Clin Exp Res. 2009;33:1582-1588.
- Elkashef A, Kahn R, Yu E, et al. Topiramate for the treatment of methamphetamine addiction: a multi-center placebo-controlled trial. Addiction. 2012;107:1297-1306.
- Colfax GN, Santos GM, Das M, et al. Mirtazapine to reduce methamphetamine use: a randomized controlled trial. Arch Gen Psychiatr. 2011;68:1168-1175.
- Mason BJ, Crean R, Goodell V, et al. A proof-of-concept randomized controlled study of gabapentin: effects on cannabis use, withdrawal and executive function deficits in cannabis-dependent adults. Neuropsychopharmacology. 2012;37:1689-1698.
- Gray KM, Carpenter MJ, Baker NL, et al. A double-blind randomized controlled trial of N-acetylcysteine in cannabis-dependent adolescents. Am J Psychiatr. 2012;169:805-812.
- Farrell M, Wodak A, Gowing L. Maintenance drugs to treat opioid dependence. BMJ. 2012;344.-
- Pinto H, Maskrey V, Swift L, et al. The SUMMIT trial: a field comparison of buprenorphine versus methadone maintenance treatment. J Subst Abuse Treat. 2010;39:340-352.
- Marsch LA. The efficacy of methadone maintenance interventions in reducing illicit opiate use HIV risk behavior and criminality: a meta-analysis. Addiction. 1998;93:515-532.
- Sees KL, Delucchi KL, Masson C, et al. Methadone maintenance vs 180-day psychosocially enriched detoxification for treatment of opioid dependence: a randomized controlled trial. JAMA. 2000;283:1303-1310.
- Clausen T, Anchersen K, Waal H. Mortality prior to during and after opioid maintenance treatment (OMT): a national prospective cross-registry study. Drug Alcohol Depend. 2008;94:151-157.
- Clark RE, Samnaliev M, Baxter JD, et al. The evidence doesn’t justify steps by state Medicaid programs to restrict opioid addiction treatment with buprenorphine. Health Affairs. 2011;30:1425-1433.
- Center for Substance Abuse Treatment. Clinical Guidelines for the Use of Buprenorphine in the Treatment of Opioid Addiction: Treatment Improvement Protocol (TIP) Series 40. Rockville Md: Substance Abuse and Mental Health Services Administration; 2004.
- Fudala PJ, Bridge TP, Herbert S, et al. Office-based treatment of opiate addiction with a sublingual-tablet formulation of buprenorphine and naloxone. N Engl J Med. 2003;349:949-958.
- Fiellin DA. Buprenorphine: effective treatment of opioid addiction starts in the office. Am Fam Physician. 2006;73:1513.-
- Minozzi S, Amato L, Vecchi S, et al. Oral naltrexone maintainance treatment for opioid dependence. Cochrane Database Syst Rev. 2011;(4):CD001333.-
- Krupitsky E, Nunes EV, Ling W, et al. Injectable extended-release naltrexone for opioid dependence: a double-blind, placebo-controlled, multicentre randomised trial. Lancet. 2011;377:1506-1513.
CORRESPONDENCE Daniel C. Vinson, MD, MSPH, MA306E Health Sciences, Family and Community Medicine, University of Missouri, Columbia, MO 652312; [email protected]
• Screen patients for substance use disorders, with a single (validated) question for alcohol and another for drugs. A
• Follow a positive screen for alcohol with an assessment to distinguish between hazardous drinking and drinking that is indicative of alcohol dependence. C
• Approach a substance use disorder as you would any chronic medical condition, seeking to engage the patient to encourage behavior change. Motivational interviewing is a useful tool. C
• Consider pharmacotherapeutic options for patients with alcohol or drug dependence. A
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
Episodic heavy drinking, like alcoholism and drug addiction, is increasingly recognized as a medical problem that primary care physicians can, and should, address.1 But it is rarely the chief reason for an office visit. Nor is it a subject patients are likely to bring up.
However, patients are generally willing to talk to a trusted doctor who asks about their use (or misuse) of alcohol or other substances. And primary care physicians can do much to help—with brief interventions, a growing armamentarium of pharmacotherapy, and referrals as needed. In the pages that follow, you’ll find easy-to-use screening tools and effective intervention strategies.
SCREENING NEEDN'T BE TIME-CONSUMING
Screening for substance use isn’t difficult. In fact, it can usually be accomplished with 2 targeted questions—one for alcohol use and one for drugs.
Alcohol. Two single-question screens to detect hazardous drinking have been validated, despite having different parameters. Ask either:
Q: When was the last time you had more than ____ drinks (4 for women and 5 for men) in one day?
Q: How many times in the past year have you had ___ or more drinks (4 for women and 5 for men) in one day?
For the first question, any answer within the past 3 months is a positive screen for hazardous drinking.2 For the second, anything other than zero is positive.3,4
Initial screening can also be done with the AUDIT-C (TABLE), a validated short (3-question) version of the Alcohol Use Disorders Identification Test that can be self-administered.5,6
Drugs. Only one single-question screen for drug use has been validated:
Q: How many times in the past year have you used an illegal drug or taken a prescription medication for nonmedical reasons?
Any answer other than never is a positive screen for hazardous drug use.7
CASE Jason F, a healthy and fit 28-year-old, has been your patient, along with his family, for years. He’s in your office because of a knee injury he incurred while running, and you take a moment to ask him, for the first time, how much he drinks and whether he takes drugs. His answer—that he drinks 3 or 4 times a week and often has multiple drinks at parties or nights out with the guys—takes you a bit by surprise.
Now what?
TABLE
3-question AUDIT-C screen for alcohol dependence5,6
| 1. How often do you have a drink containing alcohol? | ||
| 0 Never | 3 2 to 3 times a week | |
| 1 Monthly or less | 4 4 or more times a week | |
| 2 2 to 4 times a month | ||
| 2. How many drinks containing alcohol do you have on a typical day when you are drinking? | ||
| 0 I don’t drink. | 2 5 or 6 | |
| 0 1 or 2 | 3 7 to 9 | |
| 1 3 or 4 | 4 10 or more | |
| 3. How often do you have 6 or more drinks on one occasion? | ||
| 0 Never | 3 Weekly | |
| 1 Less than monthly | 4 Daily or almost daily | |
| 2 Monthly | ||
| Scoring the AUDIT-C | ||
| Alcohol dependence | ||
| Men | Women | |
| Threshold score | 5 | 4 |
| Sensitivity (%) | 80 | 76 |
| Specificity (%) | 74 | 78 |
| AROC | 0.769 | 0.767 |
| AROC, area under the receiver operating characteristic curve; AUDIT, Alcohol Use Disorders Identification Test. | ||
TELL ME MORE ABOUT IT
It is important to respond to a positive screen by requesting more information. In the conversation that ensues, the patient may provide the details you need to determine whether the drinking or drug use is indicative of a diagnosable substance use disorder, an umbrella term for alcohol or drug abuse and alcohol or drug dependence.
Alcohol or drug abuse vs dependence. Criteria for alcohol or drug abuse in the Diagnostic and Statistical Manual of Mental Disorders, 4th ed, text revision (DSM-IV-TR) include risky behavior, such as drinking and driving; problems with work and/or close relationships; or run-ins with the law, such as an arrest for driving while intoxicated. Criteria for alcohol or drug dependence include the inability to cut down or stop using the substance; evidence of tolerance and withdrawal; spending more time ingesting the substance; increasing attention to substance use while interest in other activities diminishes; and continued use despite recurrent problems.8
Tools zero in on extent of problem
To learn more about your patient’s situation, consider using the following criteria and tools:
DSM criteria. Ask the following 2 questions, which are among DSM-IV-TR criteria for alcohol dependence:
- How many times in the last year have you had a lot more to drink than you intended?
- How many times in the last year have you been drinking in situations where it could have been hazardous, where you could have caused an accident or gotten hurt?
Any answer other than zero to either question is suggestive of a substance use disorder. In exploratory analyses, this approach had positive likelihood ratios of 4.7 to 16 and negative likelihood ratios of 0.05 to 0.30.9,10
Although the above questions refer to alcohol use, they could be revised to learn more about a patient’s use of marijuana or other drugs, as well. (There are few tools for the assessment of drug use, because any illegal or nonmedical use of controlled substances has clear risks of major harm.)
CAGE. Another tool that is effective in assessing alcohol use is the 4-question CAGE—an acronym for Cut down, Annoyed, Guilty, and Eye opener:
- Have you ever felt that you should cut down on your drinking?
- Have people annoyed you by criticizing your drinking?
- Have you ever felt bad or guilty about your drinking?
- Have you ever had a drink in the morning to get rid of a hangover?
One meta-analysis found that a positive CAGE test—ie, a positive response to one or more of the questions—had a sensitivity of 0.85 and a specificity of 0.78 in identifying alcohol dependence in a primary care setting (using DSM criteria as the gold standard).11
AUDIT. This 10-item tool, a longer version of the AUDIT-C (available at http://www.medstudentlearning.com/node/6556), can also be used to determine the extent of alcohol use. This test provides detailed information about the quantity and frequency of alcohol use; however, it does not clearly distinguish between hazardous drinking and alcohol use disorders.12
If the patient is a teen
Assessment methods can be adjusted without difficulty to fit the age of the patient. The National Institute on Alcohol Abuse and Alcoholism has published the Alcohol Screening and Brief Intervention for Youth: A Practitioner’s Guide, available at http://www.niaaa.nih.gov/Publications/EducationTrainingMaterials/Pages/YouthGuide.aspx. The 6-question CRAFFT (for Car, Relax, Alone, Forget, Friends, Trouble) is a validated tool designed to assess adolescents’ use of both alcohol and drugs (http://www.ceasar-boston.org/clinicians/crafft.php).13,14
CASE You give Mr. F the CAGE test, and he answers No to all 4 questions. You conclude that while his drinking may be hazardous, he does not appear to have alcohol abuse or dependence.
FOLLOW UP WITH A BRIEF INTERVENTION
For decades, evidence has shown that brief interventions are often effective in helping hazardous drinkers like Mr. F cut back to safer levels.15-18 In some cases, the impact has been great enough to reduce health care and societal costs for up to 4 years19 and to cut the risk of alcohol-related death by about half.20 As a result, the US Preventive Services Task Force has given a B rating to counseling to reduce alcohol misuse by primary care providers.21 (There is less evidence that brief interventions are effective for drug problems,22 or in settings other than primary care.23)
Treat drug/alcohol problems
If you determine that your patient is engaging in hazardous alcohol or drug use or has a diagnosable substance use disorder, you do not have to drop everything else or treat it as an acute event. What matters is long-term success, which is best achieved by partnering with the patient.
Start by approaching drug and alcohol problems as you would a case of newly elevated blood pressure. Bring up the problem, seeking to engage the patient in addres- sing it.
If he or she does not agree to quit or cut back on drinking the first time you broach the subject, don’t be surprised or discouraged. Keep in mind that patients do not always respond positively to advice about handling chronic medical conditions either, particularly at first, and that you’ll be working together over time. What’s important, in the jargon of the Stages of Change model,24 is to help the patient move from precontemplation to contemplation, and perhaps beyond that to planning or action.
Use motivational interviewing to partner with patients
Motivational interviewing is useful in helping patients change health-related behaviors. The technique, which is not hard to learn or apply, is based on the recognition that a simple shift in style toward a guiding (rather than directive) approach can often reap benefits that are immediately apparent. 25-28
Motivational Interviewing: Helping People Change (3rd ed, by William R. Miller and Stephen Rollnick; Guilford Press, 2013) is an excellent resource for clinicians who wish to master this technique. An online tutorial in screening and brief intervention for alcohol or drug misuse is available free at https://adept.missouri.edu. Video demonstrations of motivational interviewing to address these issues are also available here ; to access them, click on “Training”, then on “Go to SBIRT videos”).
CASE Before Mr. F’s visit is concluded, you initiate a conversation about alcohol use, stating: “As your doctor, I’m concerned that the amount of alcohol you’re drinking could be hazardous to your health. I recommend that you cut down to no more than 4 drinks in any one day and to no more than 14 drinks a week.” You make it clear that change is up to him, and ask what he thinks about what you’ve said.
You also schedule a return visit in one month, at which time you will continue the conversation.
PHARMACOTHERAPY IS A USEFUL TOOL
Increasingly, alcohol and drug dependence—like other chronic conditions—can be effectively addressed with medication.
Drugs to treat alcohol dependence
Naltrexone. A daily dose of naltrexone, starting at 25 mg daily for a few days and going as high as 100 mg/d, can help patients with alcohol dependence limit their drinking to safe levels (number needed to treat [NNT]=9).29 This will reduce the risk of alcohol-related harm while the patient considers quitting.
The most common adverse effect is nausea, but a low starting dose may alleviate it. Naltrexone, also available as a 380-mg intramuscular (IM) depot injection once every 4 weeks, is an opioid antagonist and should not be given to any patient who’s taking opioids.
A 2010 Cochrane review found only 4 trials of naltrexone IM, and failed to show significant reductions in drinking.29 But post hoc analyses of trials of both oral and IM naltrexone found that those in which compliance was assured (either by direct observation or IM administration) had better outcomes than those in which it was not.30 Another post hoc analysis found that patients whose alcohol dependence was more severe derived greater benefits from the drug than those who were less severely affected.31
Acamprosate. Two 333-mg pills tid can help newly abstinent drinkers remain alcohol-free (NNT=9).32,33 The most common adverse effect is diarrhea, which may subside with continued use.
Combining acamprosate and naltrexone does not appear to be more effective than either drug alone. In a recently published meta-analysis comparing the 2 drugs, those taking acamprosate had slightly better rates of abstinence from alcohol, while naltrexone was slightly better in reducing heavy drinking.34
Disulfiram Unlike naltrexone and acamprosate, which work by altering the brain’s reward circuits, disulfiram blocks metabolism of ethanol, leading to the accumulation of a toxic metabolite and its punishing syndrome. The major problem with the drug is noncompliance, which can be addressed by enlisting the help of a caregiver or partner to ensure that it is taken daily. 35-37
Other medications that have been tested (though not approved by the US Food and Drug Administration [FDA]) as a treatment for alcohol dependence include:
- topiramate, which has been found to have modest efficacy in increasing the number of abstinent days and decreasing heavy-drinking days;38
- baclofen, which has shown efficacy in small clinical trials;39 and
- ondansetron, which has been shown to effectively treat early-onset alcohol dependence.40,41
For patients with depression and alcohol dependence, the combination of naltrexone and sertraline has been found to be superior to either drug by itself—and to have fewer adverse effects. 42 Gabapentin and lorazepam have been compared in treating alcohol withdrawal, with gabapentin resulting in greater efficacy and fewer adverse effects than lorazepam.43,44
Pharmacotherapy for drug abuse, dependence
For methamphetamine abuse and dependence. Two randomized clinical trials have studied medications for methamphetamine abuse and dependence. In one small study, topiramate did not increase the proportion of patients who achieved abstinence, but in a post hoc subgroup analysis, it did appear to help newly abstinent patients avoid relapse.45
In another study, mirtazapine significantly decreased the proportion of patients whose weekly urine tests were positive for methamphetamine (from 73% to 44%); no significant change was found among those on placebo.46
Both drugs were well tolerated, but compliance was low in both trials despite weekly counseling. Each has only one clinical trial to support its use, and neither has FDA approval for addiction treatment.
For marijuana dependence. In a study of 50 people seeking treatment for marijuana dependence, gabapentin 400 mg 3 times a day significantly improved the proportion reporting no cannabis use and whose urine tested negative for the drug.47
Another recent trial randomized adolescents dependent on cannabis to placebo or N-acetylcysteine 1200 mg twice a day. Those on the active drug were 2.4 times more likely than those on placebo to have negative urine tests with a number needed to treat of 7.48 Both trials ran for about 3 months. Neither drug is FDA approved to treat marijuana dependence.
For opioid dependence. As maintenance medication for patients dependent on opioids, both methadone and buprenorphine have been shown to reduce the use of illicit opioids, lower mortality, and improve retention compared with treatment without medication.49 Methadone would be a better choice than buprenorphine, which is a partial agonist with a ceiling on both its good (eg, stopping craving) and bad (eg, overdose risk) effects. In an open-label observational study of patients’ preferences, those who chose methadone maintenance over buprenorphine were twice as likely to remain in treatment.50 Both drugs are FDA-approved for treating opioid dependence.
Methadone is a full agonist that can be given for opioid dependence only in a federally licensed methadone maintenance clinic. It has been shown to reduce the use of other opioids, reduce criminal behaviors,51 improve function in many areas,52 and reduce mortality.53 In a cohort study of Massachusetts Medicaid data, methadone reduced mortality, which was 75% higher among those receiving abstinence-based treatment.52,54
Buprenorphine (alone and in combination with naloxone), effectively reduces the use of illicit opioids and improves functional status.55-57
Naltrexone, an opioid antagonist, may be effective in the treatment of opioid dependence. As with disulfiram for alcohol dependence, a major limitation of naltrexone for opioid dependence is noncompliance. But once a patient has been on oral naltrexone, he or she can be switched to naltrexone IM, which can be administered every 4 weeks. A Cochrane review published in 2011 found no evidence that naltrexone was superior to placebo, 58 but since then another randomized clinical trial has been published that found naltrexone 380 mg IM every 4 weeks was superior to IM placebo. Patients on naltrexone were more likely to remain in treatment and have opioid-free urine tests, and reported less craving for opioids.59 Both oral and depot injection naltrexone are FDA approved for treatment of opioid dependence. No comparisons of naltrexone vs either methadone or buprenorphine have been published.
To learn more about pharmacotherapy for opioid dependence, see “Diagnosing and treating opioid dependence” (J Fam Pract. 2012;61:588-596).
DEALING WITH THE CHALLENGES
As noted earlier, some patients with substance use disorders, like some patients with depression or hypertension, respond well to care and counseling, and some do not. Just as with other conditions, consultation with a specialist often helps.
A major difference in arranging consultations for patients with substance use disorders, however, is that clinicians who specialize in substance abuse and dependence often work in health care systems that are largely, or entirely, separate from those in which primary care physicians typically work. This, plus the stigma that surrounds problems with substance use, presents barriers to patients, who may shy away from going across town or to another city to see a provider they don’t know for a problem they’re either resistant to “owning” or ashamed of.
Yet it is possible to reach across this divide and make it easier for patients. One way to do that might be to partner with a local alcohol- and drug-treatment program so that your patients are referred, not to a faceless agency, but rather to a specific clinician; you might even call the provider while the patient is in your office so they can “meet.” Another approach, taken by some multispecialty practices, is to add psychotherapists to the staff so that patients can simply walk down the hall to obtain the mental health care they need.
Reaching across this divide is also a useful strategy for primary care physicians, who may welcome opportunities to meet with someone from a local treatment agency, not just for referrals but to learn more about treating patients with substance use problems. The Patient Protection and Affordable Care Act, which cites substance use disorders as one of 6 chronic health conditions that primary care medical homes are expected to address, may lead to better integration of health care systems that address physical health, as well as mental health and substance use disorders.
• Screen patients for substance use disorders, with a single (validated) question for alcohol and another for drugs. A
• Follow a positive screen for alcohol with an assessment to distinguish between hazardous drinking and drinking that is indicative of alcohol dependence. C
• Approach a substance use disorder as you would any chronic medical condition, seeking to engage the patient to encourage behavior change. Motivational interviewing is a useful tool. C
• Consider pharmacotherapeutic options for patients with alcohol or drug dependence. A
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
Episodic heavy drinking, like alcoholism and drug addiction, is increasingly recognized as a medical problem that primary care physicians can, and should, address.1 But it is rarely the chief reason for an office visit. Nor is it a subject patients are likely to bring up.
However, patients are generally willing to talk to a trusted doctor who asks about their use (or misuse) of alcohol or other substances. And primary care physicians can do much to help—with brief interventions, a growing armamentarium of pharmacotherapy, and referrals as needed. In the pages that follow, you’ll find easy-to-use screening tools and effective intervention strategies.
SCREENING NEEDN'T BE TIME-CONSUMING
Screening for substance use isn’t difficult. In fact, it can usually be accomplished with 2 targeted questions—one for alcohol use and one for drugs.
Alcohol. Two single-question screens to detect hazardous drinking have been validated, despite having different parameters. Ask either:
Q: When was the last time you had more than ____ drinks (4 for women and 5 for men) in one day?
Q: How many times in the past year have you had ___ or more drinks (4 for women and 5 for men) in one day?
For the first question, any answer within the past 3 months is a positive screen for hazardous drinking.2 For the second, anything other than zero is positive.3,4
Initial screening can also be done with the AUDIT-C (TABLE), a validated short (3-question) version of the Alcohol Use Disorders Identification Test that can be self-administered.5,6
Drugs. Only one single-question screen for drug use has been validated:
Q: How many times in the past year have you used an illegal drug or taken a prescription medication for nonmedical reasons?
Any answer other than never is a positive screen for hazardous drug use.7
CASE Jason F, a healthy and fit 28-year-old, has been your patient, along with his family, for years. He’s in your office because of a knee injury he incurred while running, and you take a moment to ask him, for the first time, how much he drinks and whether he takes drugs. His answer—that he drinks 3 or 4 times a week and often has multiple drinks at parties or nights out with the guys—takes you a bit by surprise.
Now what?
TABLE
3-question AUDIT-C screen for alcohol dependence5,6
| 1. How often do you have a drink containing alcohol? | ||
| 0 Never | 3 2 to 3 times a week | |
| 1 Monthly or less | 4 4 or more times a week | |
| 2 2 to 4 times a month | ||
| 2. How many drinks containing alcohol do you have on a typical day when you are drinking? | ||
| 0 I don’t drink. | 2 5 or 6 | |
| 0 1 or 2 | 3 7 to 9 | |
| 1 3 or 4 | 4 10 or more | |
| 3. How often do you have 6 or more drinks on one occasion? | ||
| 0 Never | 3 Weekly | |
| 1 Less than monthly | 4 Daily or almost daily | |
| 2 Monthly | ||
| Scoring the AUDIT-C | ||
| Alcohol dependence | ||
| Men | Women | |
| Threshold score | 5 | 4 |
| Sensitivity (%) | 80 | 76 |
| Specificity (%) | 74 | 78 |
| AROC | 0.769 | 0.767 |
| AROC, area under the receiver operating characteristic curve; AUDIT, Alcohol Use Disorders Identification Test. | ||
TELL ME MORE ABOUT IT
It is important to respond to a positive screen by requesting more information. In the conversation that ensues, the patient may provide the details you need to determine whether the drinking or drug use is indicative of a diagnosable substance use disorder, an umbrella term for alcohol or drug abuse and alcohol or drug dependence.
Alcohol or drug abuse vs dependence. Criteria for alcohol or drug abuse in the Diagnostic and Statistical Manual of Mental Disorders, 4th ed, text revision (DSM-IV-TR) include risky behavior, such as drinking and driving; problems with work and/or close relationships; or run-ins with the law, such as an arrest for driving while intoxicated. Criteria for alcohol or drug dependence include the inability to cut down or stop using the substance; evidence of tolerance and withdrawal; spending more time ingesting the substance; increasing attention to substance use while interest in other activities diminishes; and continued use despite recurrent problems.8
Tools zero in on extent of problem
To learn more about your patient’s situation, consider using the following criteria and tools:
DSM criteria. Ask the following 2 questions, which are among DSM-IV-TR criteria for alcohol dependence:
- How many times in the last year have you had a lot more to drink than you intended?
- How many times in the last year have you been drinking in situations where it could have been hazardous, where you could have caused an accident or gotten hurt?
Any answer other than zero to either question is suggestive of a substance use disorder. In exploratory analyses, this approach had positive likelihood ratios of 4.7 to 16 and negative likelihood ratios of 0.05 to 0.30.9,10
Although the above questions refer to alcohol use, they could be revised to learn more about a patient’s use of marijuana or other drugs, as well. (There are few tools for the assessment of drug use, because any illegal or nonmedical use of controlled substances has clear risks of major harm.)
CAGE. Another tool that is effective in assessing alcohol use is the 4-question CAGE—an acronym for Cut down, Annoyed, Guilty, and Eye opener:
- Have you ever felt that you should cut down on your drinking?
- Have people annoyed you by criticizing your drinking?
- Have you ever felt bad or guilty about your drinking?
- Have you ever had a drink in the morning to get rid of a hangover?
One meta-analysis found that a positive CAGE test—ie, a positive response to one or more of the questions—had a sensitivity of 0.85 and a specificity of 0.78 in identifying alcohol dependence in a primary care setting (using DSM criteria as the gold standard).11
AUDIT. This 10-item tool, a longer version of the AUDIT-C (available at http://www.medstudentlearning.com/node/6556), can also be used to determine the extent of alcohol use. This test provides detailed information about the quantity and frequency of alcohol use; however, it does not clearly distinguish between hazardous drinking and alcohol use disorders.12
If the patient is a teen
Assessment methods can be adjusted without difficulty to fit the age of the patient. The National Institute on Alcohol Abuse and Alcoholism has published the Alcohol Screening and Brief Intervention for Youth: A Practitioner’s Guide, available at http://www.niaaa.nih.gov/Publications/EducationTrainingMaterials/Pages/YouthGuide.aspx. The 6-question CRAFFT (for Car, Relax, Alone, Forget, Friends, Trouble) is a validated tool designed to assess adolescents’ use of both alcohol and drugs (http://www.ceasar-boston.org/clinicians/crafft.php).13,14
CASE You give Mr. F the CAGE test, and he answers No to all 4 questions. You conclude that while his drinking may be hazardous, he does not appear to have alcohol abuse or dependence.
FOLLOW UP WITH A BRIEF INTERVENTION
For decades, evidence has shown that brief interventions are often effective in helping hazardous drinkers like Mr. F cut back to safer levels.15-18 In some cases, the impact has been great enough to reduce health care and societal costs for up to 4 years19 and to cut the risk of alcohol-related death by about half.20 As a result, the US Preventive Services Task Force has given a B rating to counseling to reduce alcohol misuse by primary care providers.21 (There is less evidence that brief interventions are effective for drug problems,22 or in settings other than primary care.23)
Treat drug/alcohol problems
If you determine that your patient is engaging in hazardous alcohol or drug use or has a diagnosable substance use disorder, you do not have to drop everything else or treat it as an acute event. What matters is long-term success, which is best achieved by partnering with the patient.
Start by approaching drug and alcohol problems as you would a case of newly elevated blood pressure. Bring up the problem, seeking to engage the patient in addres- sing it.
If he or she does not agree to quit or cut back on drinking the first time you broach the subject, don’t be surprised or discouraged. Keep in mind that patients do not always respond positively to advice about handling chronic medical conditions either, particularly at first, and that you’ll be working together over time. What’s important, in the jargon of the Stages of Change model,24 is to help the patient move from precontemplation to contemplation, and perhaps beyond that to planning or action.
Use motivational interviewing to partner with patients
Motivational interviewing is useful in helping patients change health-related behaviors. The technique, which is not hard to learn or apply, is based on the recognition that a simple shift in style toward a guiding (rather than directive) approach can often reap benefits that are immediately apparent. 25-28
Motivational Interviewing: Helping People Change (3rd ed, by William R. Miller and Stephen Rollnick; Guilford Press, 2013) is an excellent resource for clinicians who wish to master this technique. An online tutorial in screening and brief intervention for alcohol or drug misuse is available free at https://adept.missouri.edu. Video demonstrations of motivational interviewing to address these issues are also available here ; to access them, click on “Training”, then on “Go to SBIRT videos”).
CASE Before Mr. F’s visit is concluded, you initiate a conversation about alcohol use, stating: “As your doctor, I’m concerned that the amount of alcohol you’re drinking could be hazardous to your health. I recommend that you cut down to no more than 4 drinks in any one day and to no more than 14 drinks a week.” You make it clear that change is up to him, and ask what he thinks about what you’ve said.
You also schedule a return visit in one month, at which time you will continue the conversation.
PHARMACOTHERAPY IS A USEFUL TOOL
Increasingly, alcohol and drug dependence—like other chronic conditions—can be effectively addressed with medication.
Drugs to treat alcohol dependence
Naltrexone. A daily dose of naltrexone, starting at 25 mg daily for a few days and going as high as 100 mg/d, can help patients with alcohol dependence limit their drinking to safe levels (number needed to treat [NNT]=9).29 This will reduce the risk of alcohol-related harm while the patient considers quitting.
The most common adverse effect is nausea, but a low starting dose may alleviate it. Naltrexone, also available as a 380-mg intramuscular (IM) depot injection once every 4 weeks, is an opioid antagonist and should not be given to any patient who’s taking opioids.
A 2010 Cochrane review found only 4 trials of naltrexone IM, and failed to show significant reductions in drinking.29 But post hoc analyses of trials of both oral and IM naltrexone found that those in which compliance was assured (either by direct observation or IM administration) had better outcomes than those in which it was not.30 Another post hoc analysis found that patients whose alcohol dependence was more severe derived greater benefits from the drug than those who were less severely affected.31
Acamprosate. Two 333-mg pills tid can help newly abstinent drinkers remain alcohol-free (NNT=9).32,33 The most common adverse effect is diarrhea, which may subside with continued use.
Combining acamprosate and naltrexone does not appear to be more effective than either drug alone. In a recently published meta-analysis comparing the 2 drugs, those taking acamprosate had slightly better rates of abstinence from alcohol, while naltrexone was slightly better in reducing heavy drinking.34
Disulfiram Unlike naltrexone and acamprosate, which work by altering the brain’s reward circuits, disulfiram blocks metabolism of ethanol, leading to the accumulation of a toxic metabolite and its punishing syndrome. The major problem with the drug is noncompliance, which can be addressed by enlisting the help of a caregiver or partner to ensure that it is taken daily. 35-37
Other medications that have been tested (though not approved by the US Food and Drug Administration [FDA]) as a treatment for alcohol dependence include:
- topiramate, which has been found to have modest efficacy in increasing the number of abstinent days and decreasing heavy-drinking days;38
- baclofen, which has shown efficacy in small clinical trials;39 and
- ondansetron, which has been shown to effectively treat early-onset alcohol dependence.40,41
For patients with depression and alcohol dependence, the combination of naltrexone and sertraline has been found to be superior to either drug by itself—and to have fewer adverse effects. 42 Gabapentin and lorazepam have been compared in treating alcohol withdrawal, with gabapentin resulting in greater efficacy and fewer adverse effects than lorazepam.43,44
Pharmacotherapy for drug abuse, dependence
For methamphetamine abuse and dependence. Two randomized clinical trials have studied medications for methamphetamine abuse and dependence. In one small study, topiramate did not increase the proportion of patients who achieved abstinence, but in a post hoc subgroup analysis, it did appear to help newly abstinent patients avoid relapse.45
In another study, mirtazapine significantly decreased the proportion of patients whose weekly urine tests were positive for methamphetamine (from 73% to 44%); no significant change was found among those on placebo.46
Both drugs were well tolerated, but compliance was low in both trials despite weekly counseling. Each has only one clinical trial to support its use, and neither has FDA approval for addiction treatment.
For marijuana dependence. In a study of 50 people seeking treatment for marijuana dependence, gabapentin 400 mg 3 times a day significantly improved the proportion reporting no cannabis use and whose urine tested negative for the drug.47
Another recent trial randomized adolescents dependent on cannabis to placebo or N-acetylcysteine 1200 mg twice a day. Those on the active drug were 2.4 times more likely than those on placebo to have negative urine tests with a number needed to treat of 7.48 Both trials ran for about 3 months. Neither drug is FDA approved to treat marijuana dependence.
For opioid dependence. As maintenance medication for patients dependent on opioids, both methadone and buprenorphine have been shown to reduce the use of illicit opioids, lower mortality, and improve retention compared with treatment without medication.49 Methadone would be a better choice than buprenorphine, which is a partial agonist with a ceiling on both its good (eg, stopping craving) and bad (eg, overdose risk) effects. In an open-label observational study of patients’ preferences, those who chose methadone maintenance over buprenorphine were twice as likely to remain in treatment.50 Both drugs are FDA-approved for treating opioid dependence.
Methadone is a full agonist that can be given for opioid dependence only in a federally licensed methadone maintenance clinic. It has been shown to reduce the use of other opioids, reduce criminal behaviors,51 improve function in many areas,52 and reduce mortality.53 In a cohort study of Massachusetts Medicaid data, methadone reduced mortality, which was 75% higher among those receiving abstinence-based treatment.52,54
Buprenorphine (alone and in combination with naloxone), effectively reduces the use of illicit opioids and improves functional status.55-57
Naltrexone, an opioid antagonist, may be effective in the treatment of opioid dependence. As with disulfiram for alcohol dependence, a major limitation of naltrexone for opioid dependence is noncompliance. But once a patient has been on oral naltrexone, he or she can be switched to naltrexone IM, which can be administered every 4 weeks. A Cochrane review published in 2011 found no evidence that naltrexone was superior to placebo, 58 but since then another randomized clinical trial has been published that found naltrexone 380 mg IM every 4 weeks was superior to IM placebo. Patients on naltrexone were more likely to remain in treatment and have opioid-free urine tests, and reported less craving for opioids.59 Both oral and depot injection naltrexone are FDA approved for treatment of opioid dependence. No comparisons of naltrexone vs either methadone or buprenorphine have been published.
To learn more about pharmacotherapy for opioid dependence, see “Diagnosing and treating opioid dependence” (J Fam Pract. 2012;61:588-596).
DEALING WITH THE CHALLENGES
As noted earlier, some patients with substance use disorders, like some patients with depression or hypertension, respond well to care and counseling, and some do not. Just as with other conditions, consultation with a specialist often helps.
A major difference in arranging consultations for patients with substance use disorders, however, is that clinicians who specialize in substance abuse and dependence often work in health care systems that are largely, or entirely, separate from those in which primary care physicians typically work. This, plus the stigma that surrounds problems with substance use, presents barriers to patients, who may shy away from going across town or to another city to see a provider they don’t know for a problem they’re either resistant to “owning” or ashamed of.
Yet it is possible to reach across this divide and make it easier for patients. One way to do that might be to partner with a local alcohol- and drug-treatment program so that your patients are referred, not to a faceless agency, but rather to a specific clinician; you might even call the provider while the patient is in your office so they can “meet.” Another approach, taken by some multispecialty practices, is to add psychotherapists to the staff so that patients can simply walk down the hall to obtain the mental health care they need.
Reaching across this divide is also a useful strategy for primary care physicians, who may welcome opportunities to meet with someone from a local treatment agency, not just for referrals but to learn more about treating patients with substance use problems. The Patient Protection and Affordable Care Act, which cites substance use disorders as one of 6 chronic health conditions that primary care medical homes are expected to address, may lead to better integration of health care systems that address physical health, as well as mental health and substance use disorders.
- Friedman PD. Alcohol use in adults. N Engl J Med. 2013;368:365-373.
- Williams RH, Vinson DC. Validation of a single question screen for problem drinking. J Fam Pract. 2001;50:307-312.
- Smith PC, Schmidt SM, Allensworth-Davies D, et al. Primary care validation of a single-question alcohol screening test. J Gen Intern Med. 2009;74:783-788.
- Dawson DA, Pulay AJ, Grant BF. A comparison of two single-item screeners for hazardous drinking and alcohol use disorder. Alcohol Clin Exp Res. 2010;34:364-374.
- Bush K, Kivlahan DR, Mcdonell MB, et al. The AUDIT alcohol consumption questions (AUDIT-C): an effective brief screening test for problem drinking. Arch Intern Med. 1998;158:1789-1795.
- Dawson DA, Grant BF, Stinson FS, et al. Effectiveness of the derived Alcohol Use Disorders Identification Test (AUDIT-C) in screening for alcohol use disorders and risk drinking in the US general population. Alcohol Clin Exp Res. 2005;29:844-854.
- Smith PC, Schmidt SM, Allensworth-Davies D, et al. A single-question screening test for drug use in primary care. Arch Intern Med. 2010;170:1155-1160.
- American Psychiatric Asociation. Diagnostic and Statistical Manual of Mental Disorders. 4th ed text rev. (DSM-IV-TR). Arlington, Va: American Psychiatric Association; 2000.
- Saha TD, Stinson FS, Grant BF. The role of alcohol consumption in future classifications of alcohol use disorders. Drug Alcohol Depend. 2007;89:82-92.
- Vinson DC, Kruse RL, Seale JP. Simplifying alcohol assessment:two questions to identify alcohol use disorders. Alcohol Clin Exp Res. 2007;31:1392-139.
- Aertgeerts B, Buntinx F, Kester A. The value of the CAGE in screening for alcohol abuse and alcohol dependence in general clinical populations: a diagnostic meta-analysis. J Clin Epidemiol. 2004;57:30-39.
- Johnson JA, Lee A, Vinson DC, et al. Use of AUDIT-based measures to identify unhealthy alcohol use and alcohol dependence in primary care: a validation study. Alcohol Clin Exp Res. 2012;July 26 [Epub ahead of print].
- Center for Adolescent Substance Abuse Research. The CRAFFT Screening Tool. Boston MA:2009 [updated 2012). Available at: http://www.ceasar-boston.org/clinicians/crafft.php. Accessed January 15, 2013.
- Knight JR, Sherritt L, Harris SK, et al. Validity of brief alcohol screening tests among adolescents: a comparison of the AUDIT, POSIT, CAGE, and CRAFFT. Alcohol Clin Exp Res. 2003;27:67-734.
- Wallace P, Cutler S, Haines A. Randomised controlled trial of general practitioner intervention in patients with excessive alcohol consumption. BMJ. 1988;297:663-668.
- Fleming MF, Barry KL, Manwell LB, et al. Brief physician advice for problem alcohol drinkers: a randomized controlled trial in community-based primary care practices. JAMA. 1997;277:1039-1044.
- Bertholet N, Daeppen JB, Wietlisbach V, et al. Brief alcohol intervention in primary care reduces alcohol consumption: Systematic review and meta-analysis. Arch Intern Med. 2005;165:986-995.
- Kaner EFS, Dickinson HO, Beyer F, et al. Effectiveness of brief alcohol interventions in primary care populations. Cochrane Database Syst Rev. 2007(2);CD004148.-
- Fleming MF, Mundt MP, French MT, et al. Brief physician advice for problem drinkers: long-term efficacy and benefit-cost analysis. Alcohol Clin Exp Res. 2002;26:36-43.
- Cuijpers P, Riper H, Lemmers L. The effects on mortality of brief interventions for problem drinking: a meta-analysis. Addiction. 2004;99:839-845.
- U.S.Preventive Services Task Force. Screening and behavioral counseling interventions in primary care to reduce alcohol misuse: recommendation statement. Ann Intern Med. 2004;140:554-556.
- Saitz R, Alford DP, Bernstein J, et al. Screening and brief intervention ffor unhealthy drug use in primary care settings: randomized clinical trials are needed. J Addict Med. 2010;4:131-136.
- Field CA, Baird J, Saitz R, et al. The mixed evidence for brief intervention in emergency departments, trauma care centers, and inpatient hospital settings: what should we do? Alcohol Clin Exp Res. 2010;34:2004-2010.
- DiClemente CC, Prochaska JO. Toward a comprehensive transtheoretical model of change: stages of change and addictive behaviors. In: Miller WR, Heather N, eds. Treating Addictive Behaviors. New York, NY:Plenum Press;1998:3–24.
- Mason P, Butler CC. Health Behavior Change: A Guide for Practitioners. 2nd ed. London UK:Elsevier; 2010.
- Miller WR, Rollnick S. Motivational Interviewing: Preparing People for Change. 2nd ed. New York NY: Guilford Press;2002.
- Hettema J, Steele J, Miller WR. Motivational interviewing. Ann Rev Clin Psych. 2005;1:91-111.
- Rollnick S, Butler CC, Kinnersley P, et al. Motivational interviewing. BMJ. 2010;340:c1900.-
- Rosner S, Hackl-Herrwerth A, Leucht S, et al. Opioid antagonists for alcohol dependence. Cochrane Database Syst Rev. 2010;(12):CD001867.-
- Swift R, Oslin DW, Alexander M, et al. Adherence monitoring in naltrexone pharmacotherapy trials: a systematic review. J Stud Alcohol Drugs. 2011;72:1012-1018.
- Pettinati HM, Silverman BL, Battisti JJ, et al. Efficacy of extended-release naltrexone in patients with relatively higher severity of alcohol dependence. Alcohol Clin Exp Res. 2011;35:1804-1811.
- Mann K, Lehert P, Morgan MY. The efficacy of acamprosate in the maintenance of abstinence in alcohol-dependent individuals: results of a meta-analysis. Alcohol Clin Exp Res. 2004;28:51-63.
- Rosner S, Hackl-Herrwerth A, Leucht S, et al. Acamprosate for alcohol dependence. Cochrane Database Syst Rev. 2010(9);CD004332.-
- Maisel NC, Blodgett JC, Wilbourne PL, et al. Meta-analysis of naltrexone and acamprosate for treating alcohol use disorders: when are these medications most helpful? Addiction. 2012;October 17 [Epub ahead of print].
- Azrin NH. Improvements in the community-reinforcement approach to alcoholism. Behav Res Ther. 1976;14:339-348.
- Azrin NH, Sisson RW, Meyers R, Godley M. Alcoholism treatment by disulfiram and community reinforcement therapy. J Behav Ther Exp Psychiatr. 1982;13:105-112.
- Keane TM, Foy DW, Nunn B, et al. Spouse contracting to increase antabuse compliance in alcoholic veterans. J Clin Psychol. 1984;40:340-344.
- Arbaizar B, Diersen-Sotos T, Gomez-Acebo I, et al. Topiramate in the treatment of alcohol dependence: a meta-analysis. Actas Espanolas de Psiquiatria. 2010;38:8-12.
- Johnson BA. Update on neuropharmacological treatments for alcoholism: scientific basis and clinical findings. Biochem Pharmacol. 2008;75:34-56.
- Johnson BA, Roache JD, Javors MA, et al. Ondansetron for reduction of drinking among biologically predisposed alcoholic patients: a randomized controlled trial. JAMA. 2000;284:963-971.
- Johnson BA, Ait-Daoud N, Seneviratne C, et al. Pharmacogenetic approach at the serotonin transporter gene as a method of reducing the severity of alcohol drinking. Am J Psychiatr. 2011;168:265-275.
- Pettatini HM, Oslin D, Lampman KM, et al. A double-blind, placebo-controlled trial combining sertraline and naltrexone for treating co-occurring depression and alcohol dependence. Am J Psychiatr. 2010;167:668-675.
- Anton RF, Myrick H, Wright TM, et al. Gabapentin combined with naltrexone for the treatment of alcohol dependence. Am J Psychiatr. 2011;168:709-717.
- Myrick H, Malcolm R, Randall PK, et al. A double-blind trial of gabapentin versus lorazepam in the treatment of alcohol withdrawal. Alcohol Clin Exp Res. 2009;33:1582-1588.
- Elkashef A, Kahn R, Yu E, et al. Topiramate for the treatment of methamphetamine addiction: a multi-center placebo-controlled trial. Addiction. 2012;107:1297-1306.
- Colfax GN, Santos GM, Das M, et al. Mirtazapine to reduce methamphetamine use: a randomized controlled trial. Arch Gen Psychiatr. 2011;68:1168-1175.
- Mason BJ, Crean R, Goodell V, et al. A proof-of-concept randomized controlled study of gabapentin: effects on cannabis use, withdrawal and executive function deficits in cannabis-dependent adults. Neuropsychopharmacology. 2012;37:1689-1698.
- Gray KM, Carpenter MJ, Baker NL, et al. A double-blind randomized controlled trial of N-acetylcysteine in cannabis-dependent adolescents. Am J Psychiatr. 2012;169:805-812.
- Farrell M, Wodak A, Gowing L. Maintenance drugs to treat opioid dependence. BMJ. 2012;344.-
- Pinto H, Maskrey V, Swift L, et al. The SUMMIT trial: a field comparison of buprenorphine versus methadone maintenance treatment. J Subst Abuse Treat. 2010;39:340-352.
- Marsch LA. The efficacy of methadone maintenance interventions in reducing illicit opiate use HIV risk behavior and criminality: a meta-analysis. Addiction. 1998;93:515-532.
- Sees KL, Delucchi KL, Masson C, et al. Methadone maintenance vs 180-day psychosocially enriched detoxification for treatment of opioid dependence: a randomized controlled trial. JAMA. 2000;283:1303-1310.
- Clausen T, Anchersen K, Waal H. Mortality prior to during and after opioid maintenance treatment (OMT): a national prospective cross-registry study. Drug Alcohol Depend. 2008;94:151-157.
- Clark RE, Samnaliev M, Baxter JD, et al. The evidence doesn’t justify steps by state Medicaid programs to restrict opioid addiction treatment with buprenorphine. Health Affairs. 2011;30:1425-1433.
- Center for Substance Abuse Treatment. Clinical Guidelines for the Use of Buprenorphine in the Treatment of Opioid Addiction: Treatment Improvement Protocol (TIP) Series 40. Rockville Md: Substance Abuse and Mental Health Services Administration; 2004.
- Fudala PJ, Bridge TP, Herbert S, et al. Office-based treatment of opiate addiction with a sublingual-tablet formulation of buprenorphine and naloxone. N Engl J Med. 2003;349:949-958.
- Fiellin DA. Buprenorphine: effective treatment of opioid addiction starts in the office. Am Fam Physician. 2006;73:1513.-
- Minozzi S, Amato L, Vecchi S, et al. Oral naltrexone maintainance treatment for opioid dependence. Cochrane Database Syst Rev. 2011;(4):CD001333.-
- Krupitsky E, Nunes EV, Ling W, et al. Injectable extended-release naltrexone for opioid dependence: a double-blind, placebo-controlled, multicentre randomised trial. Lancet. 2011;377:1506-1513.
CORRESPONDENCE Daniel C. Vinson, MD, MSPH, MA306E Health Sciences, Family and Community Medicine, University of Missouri, Columbia, MO 652312; [email protected]
- Friedman PD. Alcohol use in adults. N Engl J Med. 2013;368:365-373.
- Williams RH, Vinson DC. Validation of a single question screen for problem drinking. J Fam Pract. 2001;50:307-312.
- Smith PC, Schmidt SM, Allensworth-Davies D, et al. Primary care validation of a single-question alcohol screening test. J Gen Intern Med. 2009;74:783-788.
- Dawson DA, Pulay AJ, Grant BF. A comparison of two single-item screeners for hazardous drinking and alcohol use disorder. Alcohol Clin Exp Res. 2010;34:364-374.
- Bush K, Kivlahan DR, Mcdonell MB, et al. The AUDIT alcohol consumption questions (AUDIT-C): an effective brief screening test for problem drinking. Arch Intern Med. 1998;158:1789-1795.
- Dawson DA, Grant BF, Stinson FS, et al. Effectiveness of the derived Alcohol Use Disorders Identification Test (AUDIT-C) in screening for alcohol use disorders and risk drinking in the US general population. Alcohol Clin Exp Res. 2005;29:844-854.
- Smith PC, Schmidt SM, Allensworth-Davies D, et al. A single-question screening test for drug use in primary care. Arch Intern Med. 2010;170:1155-1160.
- American Psychiatric Asociation. Diagnostic and Statistical Manual of Mental Disorders. 4th ed text rev. (DSM-IV-TR). Arlington, Va: American Psychiatric Association; 2000.
- Saha TD, Stinson FS, Grant BF. The role of alcohol consumption in future classifications of alcohol use disorders. Drug Alcohol Depend. 2007;89:82-92.
- Vinson DC, Kruse RL, Seale JP. Simplifying alcohol assessment:two questions to identify alcohol use disorders. Alcohol Clin Exp Res. 2007;31:1392-139.
- Aertgeerts B, Buntinx F, Kester A. The value of the CAGE in screening for alcohol abuse and alcohol dependence in general clinical populations: a diagnostic meta-analysis. J Clin Epidemiol. 2004;57:30-39.
- Johnson JA, Lee A, Vinson DC, et al. Use of AUDIT-based measures to identify unhealthy alcohol use and alcohol dependence in primary care: a validation study. Alcohol Clin Exp Res. 2012;July 26 [Epub ahead of print].
- Center for Adolescent Substance Abuse Research. The CRAFFT Screening Tool. Boston MA:2009 [updated 2012). Available at: http://www.ceasar-boston.org/clinicians/crafft.php. Accessed January 15, 2013.
- Knight JR, Sherritt L, Harris SK, et al. Validity of brief alcohol screening tests among adolescents: a comparison of the AUDIT, POSIT, CAGE, and CRAFFT. Alcohol Clin Exp Res. 2003;27:67-734.
- Wallace P, Cutler S, Haines A. Randomised controlled trial of general practitioner intervention in patients with excessive alcohol consumption. BMJ. 1988;297:663-668.
- Fleming MF, Barry KL, Manwell LB, et al. Brief physician advice for problem alcohol drinkers: a randomized controlled trial in community-based primary care practices. JAMA. 1997;277:1039-1044.
- Bertholet N, Daeppen JB, Wietlisbach V, et al. Brief alcohol intervention in primary care reduces alcohol consumption: Systematic review and meta-analysis. Arch Intern Med. 2005;165:986-995.
- Kaner EFS, Dickinson HO, Beyer F, et al. Effectiveness of brief alcohol interventions in primary care populations. Cochrane Database Syst Rev. 2007(2);CD004148.-
- Fleming MF, Mundt MP, French MT, et al. Brief physician advice for problem drinkers: long-term efficacy and benefit-cost analysis. Alcohol Clin Exp Res. 2002;26:36-43.
- Cuijpers P, Riper H, Lemmers L. The effects on mortality of brief interventions for problem drinking: a meta-analysis. Addiction. 2004;99:839-845.
- U.S.Preventive Services Task Force. Screening and behavioral counseling interventions in primary care to reduce alcohol misuse: recommendation statement. Ann Intern Med. 2004;140:554-556.
- Saitz R, Alford DP, Bernstein J, et al. Screening and brief intervention ffor unhealthy drug use in primary care settings: randomized clinical trials are needed. J Addict Med. 2010;4:131-136.
- Field CA, Baird J, Saitz R, et al. The mixed evidence for brief intervention in emergency departments, trauma care centers, and inpatient hospital settings: what should we do? Alcohol Clin Exp Res. 2010;34:2004-2010.
- DiClemente CC, Prochaska JO. Toward a comprehensive transtheoretical model of change: stages of change and addictive behaviors. In: Miller WR, Heather N, eds. Treating Addictive Behaviors. New York, NY:Plenum Press;1998:3–24.
- Mason P, Butler CC. Health Behavior Change: A Guide for Practitioners. 2nd ed. London UK:Elsevier; 2010.
- Miller WR, Rollnick S. Motivational Interviewing: Preparing People for Change. 2nd ed. New York NY: Guilford Press;2002.
- Hettema J, Steele J, Miller WR. Motivational interviewing. Ann Rev Clin Psych. 2005;1:91-111.
- Rollnick S, Butler CC, Kinnersley P, et al. Motivational interviewing. BMJ. 2010;340:c1900.-
- Rosner S, Hackl-Herrwerth A, Leucht S, et al. Opioid antagonists for alcohol dependence. Cochrane Database Syst Rev. 2010;(12):CD001867.-
- Swift R, Oslin DW, Alexander M, et al. Adherence monitoring in naltrexone pharmacotherapy trials: a systematic review. J Stud Alcohol Drugs. 2011;72:1012-1018.
- Pettinati HM, Silverman BL, Battisti JJ, et al. Efficacy of extended-release naltrexone in patients with relatively higher severity of alcohol dependence. Alcohol Clin Exp Res. 2011;35:1804-1811.
- Mann K, Lehert P, Morgan MY. The efficacy of acamprosate in the maintenance of abstinence in alcohol-dependent individuals: results of a meta-analysis. Alcohol Clin Exp Res. 2004;28:51-63.
- Rosner S, Hackl-Herrwerth A, Leucht S, et al. Acamprosate for alcohol dependence. Cochrane Database Syst Rev. 2010(9);CD004332.-
- Maisel NC, Blodgett JC, Wilbourne PL, et al. Meta-analysis of naltrexone and acamprosate for treating alcohol use disorders: when are these medications most helpful? Addiction. 2012;October 17 [Epub ahead of print].
- Azrin NH. Improvements in the community-reinforcement approach to alcoholism. Behav Res Ther. 1976;14:339-348.
- Azrin NH, Sisson RW, Meyers R, Godley M. Alcoholism treatment by disulfiram and community reinforcement therapy. J Behav Ther Exp Psychiatr. 1982;13:105-112.
- Keane TM, Foy DW, Nunn B, et al. Spouse contracting to increase antabuse compliance in alcoholic veterans. J Clin Psychol. 1984;40:340-344.
- Arbaizar B, Diersen-Sotos T, Gomez-Acebo I, et al. Topiramate in the treatment of alcohol dependence: a meta-analysis. Actas Espanolas de Psiquiatria. 2010;38:8-12.
- Johnson BA. Update on neuropharmacological treatments for alcoholism: scientific basis and clinical findings. Biochem Pharmacol. 2008;75:34-56.
- Johnson BA, Roache JD, Javors MA, et al. Ondansetron for reduction of drinking among biologically predisposed alcoholic patients: a randomized controlled trial. JAMA. 2000;284:963-971.
- Johnson BA, Ait-Daoud N, Seneviratne C, et al. Pharmacogenetic approach at the serotonin transporter gene as a method of reducing the severity of alcohol drinking. Am J Psychiatr. 2011;168:265-275.
- Pettatini HM, Oslin D, Lampman KM, et al. A double-blind, placebo-controlled trial combining sertraline and naltrexone for treating co-occurring depression and alcohol dependence. Am J Psychiatr. 2010;167:668-675.
- Anton RF, Myrick H, Wright TM, et al. Gabapentin combined with naltrexone for the treatment of alcohol dependence. Am J Psychiatr. 2011;168:709-717.
- Myrick H, Malcolm R, Randall PK, et al. A double-blind trial of gabapentin versus lorazepam in the treatment of alcohol withdrawal. Alcohol Clin Exp Res. 2009;33:1582-1588.
- Elkashef A, Kahn R, Yu E, et al. Topiramate for the treatment of methamphetamine addiction: a multi-center placebo-controlled trial. Addiction. 2012;107:1297-1306.
- Colfax GN, Santos GM, Das M, et al. Mirtazapine to reduce methamphetamine use: a randomized controlled trial. Arch Gen Psychiatr. 2011;68:1168-1175.
- Mason BJ, Crean R, Goodell V, et al. A proof-of-concept randomized controlled study of gabapentin: effects on cannabis use, withdrawal and executive function deficits in cannabis-dependent adults. Neuropsychopharmacology. 2012;37:1689-1698.
- Gray KM, Carpenter MJ, Baker NL, et al. A double-blind randomized controlled trial of N-acetylcysteine in cannabis-dependent adolescents. Am J Psychiatr. 2012;169:805-812.
- Farrell M, Wodak A, Gowing L. Maintenance drugs to treat opioid dependence. BMJ. 2012;344.-
- Pinto H, Maskrey V, Swift L, et al. The SUMMIT trial: a field comparison of buprenorphine versus methadone maintenance treatment. J Subst Abuse Treat. 2010;39:340-352.
- Marsch LA. The efficacy of methadone maintenance interventions in reducing illicit opiate use HIV risk behavior and criminality: a meta-analysis. Addiction. 1998;93:515-532.
- Sees KL, Delucchi KL, Masson C, et al. Methadone maintenance vs 180-day psychosocially enriched detoxification for treatment of opioid dependence: a randomized controlled trial. JAMA. 2000;283:1303-1310.
- Clausen T, Anchersen K, Waal H. Mortality prior to during and after opioid maintenance treatment (OMT): a national prospective cross-registry study. Drug Alcohol Depend. 2008;94:151-157.
- Clark RE, Samnaliev M, Baxter JD, et al. The evidence doesn’t justify steps by state Medicaid programs to restrict opioid addiction treatment with buprenorphine. Health Affairs. 2011;30:1425-1433.
- Center for Substance Abuse Treatment. Clinical Guidelines for the Use of Buprenorphine in the Treatment of Opioid Addiction: Treatment Improvement Protocol (TIP) Series 40. Rockville Md: Substance Abuse and Mental Health Services Administration; 2004.
- Fudala PJ, Bridge TP, Herbert S, et al. Office-based treatment of opiate addiction with a sublingual-tablet formulation of buprenorphine and naloxone. N Engl J Med. 2003;349:949-958.
- Fiellin DA. Buprenorphine: effective treatment of opioid addiction starts in the office. Am Fam Physician. 2006;73:1513.-
- Minozzi S, Amato L, Vecchi S, et al. Oral naltrexone maintainance treatment for opioid dependence. Cochrane Database Syst Rev. 2011;(4):CD001333.-
- Krupitsky E, Nunes EV, Ling W, et al. Injectable extended-release naltrexone for opioid dependence: a double-blind, placebo-controlled, multicentre randomised trial. Lancet. 2011;377:1506-1513.
CORRESPONDENCE Daniel C. Vinson, MD, MSPH, MA306E Health Sciences, Family and Community Medicine, University of Missouri, Columbia, MO 652312; [email protected]
Decoding Pelvic Pain
A Profile of Patients in a VA Amputee System of Care Clinic
Outreach to Women Veterans of Iraq and Afghanistan: A VA and National Guard Collaboration
Tale of a Compliant Patient
Nearly 26 million US adults are estimated to have type 2 diabetes, including at least eight million who are unaware they have the disease.1 An additional 80 million adults are affected by prediabetes, with above-average glucose levels that do not yet meet American Diabetes Association (ADA) criteria for type 2 diabetes.1,2
In this context, the primary care provider is likely to encounter patients with a diagnosis of type 2 diabetes in its early stages. Which patients can be considered candidates for conservative management—that is, lifestyle modification—before antidiabetic medications are initiated?
Case Patient
A black woman, age 44, presented for a routine physical examination. She said she had been in good health until three months earlier, when she began to experience weakness and fatigue. She complained of increased thirst and frequent urination, requiring her to get up three or four times each night. She had increased her average fluid intake to nine glasses a day from the usual four. Despite a good appetite, she had lost about 7 lb over the previous three months.
The patient’s previous medical history was significant for four cesarean deliveries, gestational diabetes during all four pregnancies, hemorrhoids, constipation, keloid formation, and hyperlipidemia. Her family history was significant for diabetes (paternal side) and hyperlipidemia.
Findings from the patient’s physical exam included weight, 217 lb; height, 5’11”; BMI, 30.3; pulse, 80 beats/min and regular; and blood pressure, 138/78 mm Hg. With the exception of obesity and elevated systolic blood pressure, her physical exam findings were within normal limits.
Laboratory testing yielded the following results: urinalysis, 3+ glucose, negative for ketones and protein; fasting blood glucose, 145 mg/dL; hemoglobin A1C, 6.9%; total cholesterol, 230 mg/dL; HDL cholesterol (HDL-C), 21 mg/dL; LDL cholesterol (LDL-C), 144 mg/dL; and triglycerides, 400 mg/dL.
The patient’s history of gestational diabetes, her acknowledged sedentary lifestyle, body weight currently exceeding her ideal weight by 50 lb, and family history positive for diabetes put her at increased risk for diabetes. Also of concern was her elevated systolic blood pressure.
The patient’s A1C of 6.9% indicated that during the previous two months, her average blood glucose had been in excess of 125 mg/dL. A diagnosis of type 2 diabetes was made, based on the patient’s elevated fasting blood glucose and A1C, and the aforementioned symptoms of weight loss, fatigue, polydypsia, and polyuria.
Because the patient was perceived as self-reliant and responsible, it was recommended that she be treated conservatively with a broad lifestyle modification regimen. Goals were to reduce all of her diabetes indicators, including weight,3,4 blood pressure,5 and A1C, with an improved lipids profile—all, if possible, without the use of medication. The regimen comprised a low–glycemic index (GI) diet6,7 to reduce her glucose level, and aerobic exercise and strength training8 to improve her cardiovascular health. With resulting weight loss and reduced salt intake,3-5 it was expected that the patient would see reductions in her total cholesterol, LDL-C, and systolic blood pressure.
The patient’s management team recommended a daily plan of four small meals and one or two small snacks. She was advised to control portions by visually dividing her plate: Non-starchy (low-GI) vegetables should fill half of the plate, with the remaining half divided into equal sections for a small serving of protein and a small serving of carbohydrates with a GI of 55 or less (see figure7,9).
Her exercise program was based on recommendations from the American Association of Clinical Endocrinologists (AACE)10 and the US Department of Health and Human Services11: cardiovascular/aerobic exercise (five 30-minute sessions per week), ranging in intensity from moderate (eg, brisk walking, bicycling, doubles tennis) to vigorous (jogging or running, swimming laps, singles tennis); and strength training (two 30-minute sessions per week), such as weight-lifting, push-ups, and sit-ups.
Discussion
Type 2 diabetes mellitus affects an estimated 25.8 million residents of the United States, including an estimated 8.3 million who have not yet been diagnosed.1 According to the CDC,12 prevalence of diabetes is greatest in black and Hispanic women and men.
The sixth leading cause of death in the US, diabetes is also the leading cause of blindness (ie, diabetic retinopathy), kidney disease requiring dialysis, and lower-limb amputation. Diabetes is closely associated with cardiovascular disease (CVD), the leading cause of death in the US.12 Other diabetes-related health concerns include hypertension, peripheral diabetic neuropathy, dental disease, depression, delayed wound healing, and increased risk for infection.12
The standard criteria for a diagnosis of diabetes, according to the 2012 ADA Standards of Medical Care in Diabetes,13 are:
• A1C exceeding 6.5%, or
• A fasting blood glucose level at or exceeding 126 mg/dL, or
• A 2-hour plasma glucose at or exceeding 200 mg/dL during an oral glucose tolerance test, or
• A random blood glucose of 200 mg/dL or higher in a patient with classic symptoms of hyperglycemia or hyperglycemic crisis.13
Treatment Goals
Goals for diabetic patients advocated by the National Diabetes Education Program (NDEP)1 include maintaining blood pressure below 130/80 mm Hg, lowering LDL-C to below 100 mg/dL (and below 70 mg/dL in patients at risk for CVD), reducing triglyceride levels to below 150 mg/dL, and raising HDL-C above 40 mg/dL in women and above 50 mg/dL in men. Achieving these levels can reduce the diabetic patient’s risk for CVD and microvascular complications by 50%.1
Medical Nutrition Therapy
The NDEP and other entities1,14 refer to medical nutrition therapy as an integral component of diabetic management. This individualized meal plan, developed by a registered dietitian, is based on the patient’s nutritional status and preferences. To optimize the case patient’s glycemic control,10 a low-GI diet was selected.
As the ADA9 and Solomon et al15 have explained, this diet is based on foods that produce a reduced glucose response during the two hours after consumption. According to one literature review,16 use of a low-GI diet led a reduction in A1C of 0.43 percentage points. When combined with appropriate exercise, the low-GI diet can alleviate hypertension and postprandial hyperinsulinemia,15,17 as well as levels of C-reactive protein, an inflammatory marker that in high concentrations is associated with increased risk for CVD.18
The Role of Exercise
The ADA 2012 standards13 recommend a minimum of 150 minutes per week of moderate physical activity. The benefits of exercise in diabetic patients include improved control of blood glucose and reduction of risk for heart disease and other illnesses.3,11 Patients should be under the care of a provider who can help develop an appropriate, individualized plan.3,11
According to ADA recommendations,13 the patient’s lipid levels, blood pressure, and smoking status should be evaluated, with assessment for the presence of CVD in at-risk patients. For asymptomatic diabetic patients, the American College of Cardiology/American Heart Association recommend an ECG stress test before an exercise program is undertaken.19,20 Exercise is contraindicated in those with decompensated congestive heart failure, complex ventricular arrhythmias, unstable angina, significant aortic stenosis, or aortic aneurysm.19
Because initiating an exercise program incurs a slight risk for injury, patients should start gradually, warming up before each session and cooling down afterward.11,19 Diabetic patients should also wear proper shoes for maximum foot protection. They must also be instructed to watch for signs of hypoglycemia during physical activity and be prepared to treat it.11,13
Patient Outcome
After one year on her prescribed treatment regimen, the patient’s A1C was measured at 6.3%, and her LDL-C was 124 mg/dL. Another year later, her A1C was further reduced to 5.9%, and her LDL-C to 77.2 mg/dL. By then, she had lost 15 lb (BMI, 28.2) and perceived her current maintenance plan as enjoyable and manageable, long-term: She was exercising every day possible, with seven sessions of at least 30 minutes, and maintaining a diet of low-GI foods. The patient was committed to following up with her clinician for glucose monitoring every three months.
Conclusion
As illustrated in this patient’s case, the best plan for management of diabetes in its early stages is one that is realistic for the patient and that will prevent diabetic complications for as long as possible.
References
1. US Department of Health and Human Services, National Diabetes Education Program. Guiding Principles for Diabetes Care for Healthcare Professionals (2009). www.ndep.nih.gov/media/guid prin_hc_eng.pdf. Accessed January 24, 2013.
2. National Diabetes Information Clearinghouse, National Institute of Diabetes and Digestive and Kidney Diseases, NIH. Insulin resistance and prediabetes (2011). http://diabetes.niddk.nih.gov/dm/pubs/insulinresistance. Accessed January 25, 2013.
3. Hayes C, Kriska A. Role of physical activity in diabetes management and prevention. J Am Diet Assoc. 2008;108(4 suppl 1):S19-S23.
4. American Diabetes Association. Food and fitness: weight loss (2011). www.diabetes.org/food-and-fitness/fitness/weight-loss. Accessed January 25, 2013.
5. US Department of Health and Human Services, NIH. Seventh Report of the Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure (JNC 7 reference card, 2003). www.nhlbi.nih.gov/guidelines/hyper tension/phycard.pdf. Accessed January 24, 2013.
6. Ma Y, Olendzki BC, Merriam PA, et al. A randomized clinical trial comparing low-glycemic index versus ADA dietary education among individuals with type 2 diabetes. Nutrition. 2008;24(1):45-56.
7. American Diabetes Association. Glycemic index of foods (2013). www.diabetes.org/food-and-fit ness/food/planning-meals/the-glycemic-index-of-foods.html. Accessed January 25, 2013.
8. Pariser G, Ann Demeuro M, Gillette P, Stephen W. Outcomes of an education and exercise program for adults with type 2 diabetes, and comorbidities that limit their mobility: a preliminary project report. Cardiopulm Phys Ther J. 2010;21(2):5-12.
9. American Diabetes Association. Food and fitness: create your plate (2013). www.diabetes.org/food-and-fitness/food/planning-meals/create-your-plate. Accessed January 24, 2013.
10. Handelsman Y, Mechanick JI, Blonde L, et al. American Association of Clinical Endocrinologists Medical Guidelines for Clinical Practice for developing a diabetes mellitus comprehensive care plan. Endocr Pract. 2011;17 suppl 2:1-53.
11. CDC. 2008 Physical Activity Guidelines. www.health.gov/PAGuidelines/pdf/paguide.pdf. Accessed January 24, 2013.
12. CDC. 2011 National Diabetes Fact Sheet. www.cdc.gov/diabetes/pubs/factsheet11.htm. Accessed January 25, 2013.
13. American Diabetes Association. Standards of medical care in diabetes—2012. Diabetes Care. 2012;35 suppl 1:S11-S63.
14. Franz MJ, Powers MA, Leontos C, et al. The evidence for medical nutrition therapy for type 1 and type 2 diabetes in adults. J Am Diet Assoc. 2010;110(12):1852-1889.
15. Solomon TP, Haus JM, Kelly KR, et al. A low-glycemic index diet combined with exercise reduces insulin resistance, postprandial hyperinsulinemia, and glucose-dependent insulinotropic polypeptide responses in obese, prediabetic humans. Am J Clin Nutr. 2010;92(6):1359-1368.
16. Brand-Miller J, Hayne S, Petocz P, Colagiuri S. Low-glycemic index diets in the management of diabetes: a meta-analysis of randomized controlled trials. Diabetes Care. 2003;26(8):2261-2267.
17. Solomon TP, Haus JM, Kelly KR, et al. Randomized trial on the effects of a 7-d low–glycemic diet and exercise intervention on insulin resistance in older obese humans. Am J Clin Nutr. 2009;90(5):1222-1229.
18. Wolever TM, Gibbs AL, Mehling C, et al. The Canadian Trial of Carbohydrates in Diabetes (CCD), a 1-y controlled trial of low–glycemic-index dietary carbohydrate in type 2 diabetes: no effect on glycated hemoglobin but reduction in C-reactive protein. Am J Clin Nutr. 2008;87(1):114-125.
19. Metkus TS, Baughman KL, Thompson PD. Exercise prescription and primary prevention of cardiovascular disease. Circulation. 2010;121(23):2601-2604.
20. Gibbons RJ, Balady GJ, Bricker JT, et al. ACC/AHA 2002 guideline update for exercise testing: summary article: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Committee to Update the 1997 Exercise Testing Guidelines). Circulation. 2002;106(14);1883-1892.
Nearly 26 million US adults are estimated to have type 2 diabetes, including at least eight million who are unaware they have the disease.1 An additional 80 million adults are affected by prediabetes, with above-average glucose levels that do not yet meet American Diabetes Association (ADA) criteria for type 2 diabetes.1,2
In this context, the primary care provider is likely to encounter patients with a diagnosis of type 2 diabetes in its early stages. Which patients can be considered candidates for conservative management—that is, lifestyle modification—before antidiabetic medications are initiated?
Case Patient
A black woman, age 44, presented for a routine physical examination. She said she had been in good health until three months earlier, when she began to experience weakness and fatigue. She complained of increased thirst and frequent urination, requiring her to get up three or four times each night. She had increased her average fluid intake to nine glasses a day from the usual four. Despite a good appetite, she had lost about 7 lb over the previous three months.
The patient’s previous medical history was significant for four cesarean deliveries, gestational diabetes during all four pregnancies, hemorrhoids, constipation, keloid formation, and hyperlipidemia. Her family history was significant for diabetes (paternal side) and hyperlipidemia.
Findings from the patient’s physical exam included weight, 217 lb; height, 5’11”; BMI, 30.3; pulse, 80 beats/min and regular; and blood pressure, 138/78 mm Hg. With the exception of obesity and elevated systolic blood pressure, her physical exam findings were within normal limits.
Laboratory testing yielded the following results: urinalysis, 3+ glucose, negative for ketones and protein; fasting blood glucose, 145 mg/dL; hemoglobin A1C, 6.9%; total cholesterol, 230 mg/dL; HDL cholesterol (HDL-C), 21 mg/dL; LDL cholesterol (LDL-C), 144 mg/dL; and triglycerides, 400 mg/dL.
The patient’s history of gestational diabetes, her acknowledged sedentary lifestyle, body weight currently exceeding her ideal weight by 50 lb, and family history positive for diabetes put her at increased risk for diabetes. Also of concern was her elevated systolic blood pressure.
The patient’s A1C of 6.9% indicated that during the previous two months, her average blood glucose had been in excess of 125 mg/dL. A diagnosis of type 2 diabetes was made, based on the patient’s elevated fasting blood glucose and A1C, and the aforementioned symptoms of weight loss, fatigue, polydypsia, and polyuria.
Because the patient was perceived as self-reliant and responsible, it was recommended that she be treated conservatively with a broad lifestyle modification regimen. Goals were to reduce all of her diabetes indicators, including weight,3,4 blood pressure,5 and A1C, with an improved lipids profile—all, if possible, without the use of medication. The regimen comprised a low–glycemic index (GI) diet6,7 to reduce her glucose level, and aerobic exercise and strength training8 to improve her cardiovascular health. With resulting weight loss and reduced salt intake,3-5 it was expected that the patient would see reductions in her total cholesterol, LDL-C, and systolic blood pressure.
The patient’s management team recommended a daily plan of four small meals and one or two small snacks. She was advised to control portions by visually dividing her plate: Non-starchy (low-GI) vegetables should fill half of the plate, with the remaining half divided into equal sections for a small serving of protein and a small serving of carbohydrates with a GI of 55 or less (see figure7,9).
Her exercise program was based on recommendations from the American Association of Clinical Endocrinologists (AACE)10 and the US Department of Health and Human Services11: cardiovascular/aerobic exercise (five 30-minute sessions per week), ranging in intensity from moderate (eg, brisk walking, bicycling, doubles tennis) to vigorous (jogging or running, swimming laps, singles tennis); and strength training (two 30-minute sessions per week), such as weight-lifting, push-ups, and sit-ups.
Discussion
Type 2 diabetes mellitus affects an estimated 25.8 million residents of the United States, including an estimated 8.3 million who have not yet been diagnosed.1 According to the CDC,12 prevalence of diabetes is greatest in black and Hispanic women and men.
The sixth leading cause of death in the US, diabetes is also the leading cause of blindness (ie, diabetic retinopathy), kidney disease requiring dialysis, and lower-limb amputation. Diabetes is closely associated with cardiovascular disease (CVD), the leading cause of death in the US.12 Other diabetes-related health concerns include hypertension, peripheral diabetic neuropathy, dental disease, depression, delayed wound healing, and increased risk for infection.12
The standard criteria for a diagnosis of diabetes, according to the 2012 ADA Standards of Medical Care in Diabetes,13 are:
• A1C exceeding 6.5%, or
• A fasting blood glucose level at or exceeding 126 mg/dL, or
• A 2-hour plasma glucose at or exceeding 200 mg/dL during an oral glucose tolerance test, or
• A random blood glucose of 200 mg/dL or higher in a patient with classic symptoms of hyperglycemia or hyperglycemic crisis.13
Treatment Goals
Goals for diabetic patients advocated by the National Diabetes Education Program (NDEP)1 include maintaining blood pressure below 130/80 mm Hg, lowering LDL-C to below 100 mg/dL (and below 70 mg/dL in patients at risk for CVD), reducing triglyceride levels to below 150 mg/dL, and raising HDL-C above 40 mg/dL in women and above 50 mg/dL in men. Achieving these levels can reduce the diabetic patient’s risk for CVD and microvascular complications by 50%.1
Medical Nutrition Therapy
The NDEP and other entities1,14 refer to medical nutrition therapy as an integral component of diabetic management. This individualized meal plan, developed by a registered dietitian, is based on the patient’s nutritional status and preferences. To optimize the case patient’s glycemic control,10 a low-GI diet was selected.
As the ADA9 and Solomon et al15 have explained, this diet is based on foods that produce a reduced glucose response during the two hours after consumption. According to one literature review,16 use of a low-GI diet led a reduction in A1C of 0.43 percentage points. When combined with appropriate exercise, the low-GI diet can alleviate hypertension and postprandial hyperinsulinemia,15,17 as well as levels of C-reactive protein, an inflammatory marker that in high concentrations is associated with increased risk for CVD.18
The Role of Exercise
The ADA 2012 standards13 recommend a minimum of 150 minutes per week of moderate physical activity. The benefits of exercise in diabetic patients include improved control of blood glucose and reduction of risk for heart disease and other illnesses.3,11 Patients should be under the care of a provider who can help develop an appropriate, individualized plan.3,11
According to ADA recommendations,13 the patient’s lipid levels, blood pressure, and smoking status should be evaluated, with assessment for the presence of CVD in at-risk patients. For asymptomatic diabetic patients, the American College of Cardiology/American Heart Association recommend an ECG stress test before an exercise program is undertaken.19,20 Exercise is contraindicated in those with decompensated congestive heart failure, complex ventricular arrhythmias, unstable angina, significant aortic stenosis, or aortic aneurysm.19
Because initiating an exercise program incurs a slight risk for injury, patients should start gradually, warming up before each session and cooling down afterward.11,19 Diabetic patients should also wear proper shoes for maximum foot protection. They must also be instructed to watch for signs of hypoglycemia during physical activity and be prepared to treat it.11,13
Patient Outcome
After one year on her prescribed treatment regimen, the patient’s A1C was measured at 6.3%, and her LDL-C was 124 mg/dL. Another year later, her A1C was further reduced to 5.9%, and her LDL-C to 77.2 mg/dL. By then, she had lost 15 lb (BMI, 28.2) and perceived her current maintenance plan as enjoyable and manageable, long-term: She was exercising every day possible, with seven sessions of at least 30 minutes, and maintaining a diet of low-GI foods. The patient was committed to following up with her clinician for glucose monitoring every three months.
Conclusion
As illustrated in this patient’s case, the best plan for management of diabetes in its early stages is one that is realistic for the patient and that will prevent diabetic complications for as long as possible.
References
1. US Department of Health and Human Services, National Diabetes Education Program. Guiding Principles for Diabetes Care for Healthcare Professionals (2009). www.ndep.nih.gov/media/guid prin_hc_eng.pdf. Accessed January 24, 2013.
2. National Diabetes Information Clearinghouse, National Institute of Diabetes and Digestive and Kidney Diseases, NIH. Insulin resistance and prediabetes (2011). http://diabetes.niddk.nih.gov/dm/pubs/insulinresistance. Accessed January 25, 2013.
3. Hayes C, Kriska A. Role of physical activity in diabetes management and prevention. J Am Diet Assoc. 2008;108(4 suppl 1):S19-S23.
4. American Diabetes Association. Food and fitness: weight loss (2011). www.diabetes.org/food-and-fitness/fitness/weight-loss. Accessed January 25, 2013.
5. US Department of Health and Human Services, NIH. Seventh Report of the Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure (JNC 7 reference card, 2003). www.nhlbi.nih.gov/guidelines/hyper tension/phycard.pdf. Accessed January 24, 2013.
6. Ma Y, Olendzki BC, Merriam PA, et al. A randomized clinical trial comparing low-glycemic index versus ADA dietary education among individuals with type 2 diabetes. Nutrition. 2008;24(1):45-56.
7. American Diabetes Association. Glycemic index of foods (2013). www.diabetes.org/food-and-fit ness/food/planning-meals/the-glycemic-index-of-foods.html. Accessed January 25, 2013.
8. Pariser G, Ann Demeuro M, Gillette P, Stephen W. Outcomes of an education and exercise program for adults with type 2 diabetes, and comorbidities that limit their mobility: a preliminary project report. Cardiopulm Phys Ther J. 2010;21(2):5-12.
9. American Diabetes Association. Food and fitness: create your plate (2013). www.diabetes.org/food-and-fitness/food/planning-meals/create-your-plate. Accessed January 24, 2013.
10. Handelsman Y, Mechanick JI, Blonde L, et al. American Association of Clinical Endocrinologists Medical Guidelines for Clinical Practice for developing a diabetes mellitus comprehensive care plan. Endocr Pract. 2011;17 suppl 2:1-53.
11. CDC. 2008 Physical Activity Guidelines. www.health.gov/PAGuidelines/pdf/paguide.pdf. Accessed January 24, 2013.
12. CDC. 2011 National Diabetes Fact Sheet. www.cdc.gov/diabetes/pubs/factsheet11.htm. Accessed January 25, 2013.
13. American Diabetes Association. Standards of medical care in diabetes—2012. Diabetes Care. 2012;35 suppl 1:S11-S63.
14. Franz MJ, Powers MA, Leontos C, et al. The evidence for medical nutrition therapy for type 1 and type 2 diabetes in adults. J Am Diet Assoc. 2010;110(12):1852-1889.
15. Solomon TP, Haus JM, Kelly KR, et al. A low-glycemic index diet combined with exercise reduces insulin resistance, postprandial hyperinsulinemia, and glucose-dependent insulinotropic polypeptide responses in obese, prediabetic humans. Am J Clin Nutr. 2010;92(6):1359-1368.
16. Brand-Miller J, Hayne S, Petocz P, Colagiuri S. Low-glycemic index diets in the management of diabetes: a meta-analysis of randomized controlled trials. Diabetes Care. 2003;26(8):2261-2267.
17. Solomon TP, Haus JM, Kelly KR, et al. Randomized trial on the effects of a 7-d low–glycemic diet and exercise intervention on insulin resistance in older obese humans. Am J Clin Nutr. 2009;90(5):1222-1229.
18. Wolever TM, Gibbs AL, Mehling C, et al. The Canadian Trial of Carbohydrates in Diabetes (CCD), a 1-y controlled trial of low–glycemic-index dietary carbohydrate in type 2 diabetes: no effect on glycated hemoglobin but reduction in C-reactive protein. Am J Clin Nutr. 2008;87(1):114-125.
19. Metkus TS, Baughman KL, Thompson PD. Exercise prescription and primary prevention of cardiovascular disease. Circulation. 2010;121(23):2601-2604.
20. Gibbons RJ, Balady GJ, Bricker JT, et al. ACC/AHA 2002 guideline update for exercise testing: summary article: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Committee to Update the 1997 Exercise Testing Guidelines). Circulation. 2002;106(14);1883-1892.
Nearly 26 million US adults are estimated to have type 2 diabetes, including at least eight million who are unaware they have the disease.1 An additional 80 million adults are affected by prediabetes, with above-average glucose levels that do not yet meet American Diabetes Association (ADA) criteria for type 2 diabetes.1,2
In this context, the primary care provider is likely to encounter patients with a diagnosis of type 2 diabetes in its early stages. Which patients can be considered candidates for conservative management—that is, lifestyle modification—before antidiabetic medications are initiated?
Case Patient
A black woman, age 44, presented for a routine physical examination. She said she had been in good health until three months earlier, when she began to experience weakness and fatigue. She complained of increased thirst and frequent urination, requiring her to get up three or four times each night. She had increased her average fluid intake to nine glasses a day from the usual four. Despite a good appetite, she had lost about 7 lb over the previous three months.
The patient’s previous medical history was significant for four cesarean deliveries, gestational diabetes during all four pregnancies, hemorrhoids, constipation, keloid formation, and hyperlipidemia. Her family history was significant for diabetes (paternal side) and hyperlipidemia.
Findings from the patient’s physical exam included weight, 217 lb; height, 5’11”; BMI, 30.3; pulse, 80 beats/min and regular; and blood pressure, 138/78 mm Hg. With the exception of obesity and elevated systolic blood pressure, her physical exam findings were within normal limits.
Laboratory testing yielded the following results: urinalysis, 3+ glucose, negative for ketones and protein; fasting blood glucose, 145 mg/dL; hemoglobin A1C, 6.9%; total cholesterol, 230 mg/dL; HDL cholesterol (HDL-C), 21 mg/dL; LDL cholesterol (LDL-C), 144 mg/dL; and triglycerides, 400 mg/dL.
The patient’s history of gestational diabetes, her acknowledged sedentary lifestyle, body weight currently exceeding her ideal weight by 50 lb, and family history positive for diabetes put her at increased risk for diabetes. Also of concern was her elevated systolic blood pressure.
The patient’s A1C of 6.9% indicated that during the previous two months, her average blood glucose had been in excess of 125 mg/dL. A diagnosis of type 2 diabetes was made, based on the patient’s elevated fasting blood glucose and A1C, and the aforementioned symptoms of weight loss, fatigue, polydypsia, and polyuria.
Because the patient was perceived as self-reliant and responsible, it was recommended that she be treated conservatively with a broad lifestyle modification regimen. Goals were to reduce all of her diabetes indicators, including weight,3,4 blood pressure,5 and A1C, with an improved lipids profile—all, if possible, without the use of medication. The regimen comprised a low–glycemic index (GI) diet6,7 to reduce her glucose level, and aerobic exercise and strength training8 to improve her cardiovascular health. With resulting weight loss and reduced salt intake,3-5 it was expected that the patient would see reductions in her total cholesterol, LDL-C, and systolic blood pressure.
The patient’s management team recommended a daily plan of four small meals and one or two small snacks. She was advised to control portions by visually dividing her plate: Non-starchy (low-GI) vegetables should fill half of the plate, with the remaining half divided into equal sections for a small serving of protein and a small serving of carbohydrates with a GI of 55 or less (see figure7,9).
Her exercise program was based on recommendations from the American Association of Clinical Endocrinologists (AACE)10 and the US Department of Health and Human Services11: cardiovascular/aerobic exercise (five 30-minute sessions per week), ranging in intensity from moderate (eg, brisk walking, bicycling, doubles tennis) to vigorous (jogging or running, swimming laps, singles tennis); and strength training (two 30-minute sessions per week), such as weight-lifting, push-ups, and sit-ups.
Discussion
Type 2 diabetes mellitus affects an estimated 25.8 million residents of the United States, including an estimated 8.3 million who have not yet been diagnosed.1 According to the CDC,12 prevalence of diabetes is greatest in black and Hispanic women and men.
The sixth leading cause of death in the US, diabetes is also the leading cause of blindness (ie, diabetic retinopathy), kidney disease requiring dialysis, and lower-limb amputation. Diabetes is closely associated with cardiovascular disease (CVD), the leading cause of death in the US.12 Other diabetes-related health concerns include hypertension, peripheral diabetic neuropathy, dental disease, depression, delayed wound healing, and increased risk for infection.12
The standard criteria for a diagnosis of diabetes, according to the 2012 ADA Standards of Medical Care in Diabetes,13 are:
• A1C exceeding 6.5%, or
• A fasting blood glucose level at or exceeding 126 mg/dL, or
• A 2-hour plasma glucose at or exceeding 200 mg/dL during an oral glucose tolerance test, or
• A random blood glucose of 200 mg/dL or higher in a patient with classic symptoms of hyperglycemia or hyperglycemic crisis.13
Treatment Goals
Goals for diabetic patients advocated by the National Diabetes Education Program (NDEP)1 include maintaining blood pressure below 130/80 mm Hg, lowering LDL-C to below 100 mg/dL (and below 70 mg/dL in patients at risk for CVD), reducing triglyceride levels to below 150 mg/dL, and raising HDL-C above 40 mg/dL in women and above 50 mg/dL in men. Achieving these levels can reduce the diabetic patient’s risk for CVD and microvascular complications by 50%.1
Medical Nutrition Therapy
The NDEP and other entities1,14 refer to medical nutrition therapy as an integral component of diabetic management. This individualized meal plan, developed by a registered dietitian, is based on the patient’s nutritional status and preferences. To optimize the case patient’s glycemic control,10 a low-GI diet was selected.
As the ADA9 and Solomon et al15 have explained, this diet is based on foods that produce a reduced glucose response during the two hours after consumption. According to one literature review,16 use of a low-GI diet led a reduction in A1C of 0.43 percentage points. When combined with appropriate exercise, the low-GI diet can alleviate hypertension and postprandial hyperinsulinemia,15,17 as well as levels of C-reactive protein, an inflammatory marker that in high concentrations is associated with increased risk for CVD.18
The Role of Exercise
The ADA 2012 standards13 recommend a minimum of 150 minutes per week of moderate physical activity. The benefits of exercise in diabetic patients include improved control of blood glucose and reduction of risk for heart disease and other illnesses.3,11 Patients should be under the care of a provider who can help develop an appropriate, individualized plan.3,11
According to ADA recommendations,13 the patient’s lipid levels, blood pressure, and smoking status should be evaluated, with assessment for the presence of CVD in at-risk patients. For asymptomatic diabetic patients, the American College of Cardiology/American Heart Association recommend an ECG stress test before an exercise program is undertaken.19,20 Exercise is contraindicated in those with decompensated congestive heart failure, complex ventricular arrhythmias, unstable angina, significant aortic stenosis, or aortic aneurysm.19
Because initiating an exercise program incurs a slight risk for injury, patients should start gradually, warming up before each session and cooling down afterward.11,19 Diabetic patients should also wear proper shoes for maximum foot protection. They must also be instructed to watch for signs of hypoglycemia during physical activity and be prepared to treat it.11,13
Patient Outcome
After one year on her prescribed treatment regimen, the patient’s A1C was measured at 6.3%, and her LDL-C was 124 mg/dL. Another year later, her A1C was further reduced to 5.9%, and her LDL-C to 77.2 mg/dL. By then, she had lost 15 lb (BMI, 28.2) and perceived her current maintenance plan as enjoyable and manageable, long-term: She was exercising every day possible, with seven sessions of at least 30 minutes, and maintaining a diet of low-GI foods. The patient was committed to following up with her clinician for glucose monitoring every three months.
Conclusion
As illustrated in this patient’s case, the best plan for management of diabetes in its early stages is one that is realistic for the patient and that will prevent diabetic complications for as long as possible.
References
1. US Department of Health and Human Services, National Diabetes Education Program. Guiding Principles for Diabetes Care for Healthcare Professionals (2009). www.ndep.nih.gov/media/guid prin_hc_eng.pdf. Accessed January 24, 2013.
2. National Diabetes Information Clearinghouse, National Institute of Diabetes and Digestive and Kidney Diseases, NIH. Insulin resistance and prediabetes (2011). http://diabetes.niddk.nih.gov/dm/pubs/insulinresistance. Accessed January 25, 2013.
3. Hayes C, Kriska A. Role of physical activity in diabetes management and prevention. J Am Diet Assoc. 2008;108(4 suppl 1):S19-S23.
4. American Diabetes Association. Food and fitness: weight loss (2011). www.diabetes.org/food-and-fitness/fitness/weight-loss. Accessed January 25, 2013.
5. US Department of Health and Human Services, NIH. Seventh Report of the Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure (JNC 7 reference card, 2003). www.nhlbi.nih.gov/guidelines/hyper tension/phycard.pdf. Accessed January 24, 2013.
6. Ma Y, Olendzki BC, Merriam PA, et al. A randomized clinical trial comparing low-glycemic index versus ADA dietary education among individuals with type 2 diabetes. Nutrition. 2008;24(1):45-56.
7. American Diabetes Association. Glycemic index of foods (2013). www.diabetes.org/food-and-fit ness/food/planning-meals/the-glycemic-index-of-foods.html. Accessed January 25, 2013.
8. Pariser G, Ann Demeuro M, Gillette P, Stephen W. Outcomes of an education and exercise program for adults with type 2 diabetes, and comorbidities that limit their mobility: a preliminary project report. Cardiopulm Phys Ther J. 2010;21(2):5-12.
9. American Diabetes Association. Food and fitness: create your plate (2013). www.diabetes.org/food-and-fitness/food/planning-meals/create-your-plate. Accessed January 24, 2013.
10. Handelsman Y, Mechanick JI, Blonde L, et al. American Association of Clinical Endocrinologists Medical Guidelines for Clinical Practice for developing a diabetes mellitus comprehensive care plan. Endocr Pract. 2011;17 suppl 2:1-53.
11. CDC. 2008 Physical Activity Guidelines. www.health.gov/PAGuidelines/pdf/paguide.pdf. Accessed January 24, 2013.
12. CDC. 2011 National Diabetes Fact Sheet. www.cdc.gov/diabetes/pubs/factsheet11.htm. Accessed January 25, 2013.
13. American Diabetes Association. Standards of medical care in diabetes—2012. Diabetes Care. 2012;35 suppl 1:S11-S63.
14. Franz MJ, Powers MA, Leontos C, et al. The evidence for medical nutrition therapy for type 1 and type 2 diabetes in adults. J Am Diet Assoc. 2010;110(12):1852-1889.
15. Solomon TP, Haus JM, Kelly KR, et al. A low-glycemic index diet combined with exercise reduces insulin resistance, postprandial hyperinsulinemia, and glucose-dependent insulinotropic polypeptide responses in obese, prediabetic humans. Am J Clin Nutr. 2010;92(6):1359-1368.
16. Brand-Miller J, Hayne S, Petocz P, Colagiuri S. Low-glycemic index diets in the management of diabetes: a meta-analysis of randomized controlled trials. Diabetes Care. 2003;26(8):2261-2267.
17. Solomon TP, Haus JM, Kelly KR, et al. Randomized trial on the effects of a 7-d low–glycemic diet and exercise intervention on insulin resistance in older obese humans. Am J Clin Nutr. 2009;90(5):1222-1229.
18. Wolever TM, Gibbs AL, Mehling C, et al. The Canadian Trial of Carbohydrates in Diabetes (CCD), a 1-y controlled trial of low–glycemic-index dietary carbohydrate in type 2 diabetes: no effect on glycated hemoglobin but reduction in C-reactive protein. Am J Clin Nutr. 2008;87(1):114-125.
19. Metkus TS, Baughman KL, Thompson PD. Exercise prescription and primary prevention of cardiovascular disease. Circulation. 2010;121(23):2601-2604.
20. Gibbons RJ, Balady GJ, Bricker JT, et al. ACC/AHA 2002 guideline update for exercise testing: summary article: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Committee to Update the 1997 Exercise Testing Guidelines). Circulation. 2002;106(14);1883-1892.