User login
Vibrators, your practice, and your patients’ sexual health
Click here to access articles on sexual health published in OBG Management in 2012.
As health-care professionals, we are well trained in the importance of taking a detailed, targeted history and incorporating appropriate assessments to determine a treatment plan, which often includes prescriptions, a medical procedure, or a referral. Our extensive training allows us to make and implement expert decisions.
At the same time, I’ve found that the most valued aspect of our job is the relationships we develop with our patients. We know details that probably no one else knows about them. This is likely most true for us clinicians whose practices address women’s sexual health. A deep level of trust develops over time.
As part of that relationship of trust, we have unique opportunities to influence not only a woman’s health but also her overall quality of life. What I found over the years is that many women don’t necessarily need any prescription or referral or procedure. What they do often need is guidance, direction, and encouragement.
Some of my greatest achievements in delivering health care have not happened in a delivery room or because of a medication I prescribed. Instead, they have occurred in the office where, for example, I see a woman who took my recommendations 1 year ago to change her diet and begin exercising. Now, 30 lb lighter, she feels like a new person. This is one example of a knowledgeable physician applying “extensive training and expert decisions” to incorporate low-tech treatments with great success.
So what does this have to do with vibrators in a doctor’s office? Well, without many FDA-approved medications to treat sexual dysfunction, we are left with over-the-counter possibilities and such “low-tech” options as counseling and guidance.
Sexual dysfunction is common among our patients
According to the National Health and Social Life Survey, 43% of women are affected by some aspect of sexual dysfunction, and 22% associate that dysfunction with personal distress. The distress was highest for women aged 45 to 64. Additional correlates included poor self-assessed health, low education level, depression, anxiety, thyroid conditions, and urinary incontinence.1 Sexual complaints among women diagnosed with cancer can be as high as 90%.2
Does this describe anyone in your practice? We can agree that women’s sexual issues are common, whether we acknowledge that in our practice by querying women on their sexual health or not (and many of us are not).
We’re not talking to our patients about their sexual health enough. In a 2012 survey of more than 1,000 ObGyns about whether they talk with their patients about sex,3 investigators found that:
- 63% routinely ask whether their patient is sexually active
- 40% routinely ask if the patient is having any problems regarding sex
- 29% ask about sexual satisfaction
- 28% ask about sexual orientation or identity
- 14% ask about sexual pleasure.
Whatever the reason, talking about sexual health is easier for some people than for others, and that goes for both clinicians and patients.
What are our treatment options if sexual dysfunction is identified?
Unfortunately, the therapeutic armamentarium for women’s sexual health is limited—perhaps a contributor to many clinicians’ reluctance to broach the topic. Pharmacologically, we have no FDA-approved treatments for female sexual dysfunction except localized estrogen treatment for vulvovaginal atrophy and the Eros device for arousal and orgasmic disorders.
There are off-label options of testosterone, buproprion, or sildenafil citrate, but many practitioners and patients are reluctant to consider these options specifically because they are off label.
Over-the-counter. These options are available for women, and can be helpful. For patients with discomfort due to vaginal dryness, it is important to educate them on the benefits of vaginal moisturizers and lubricants. Many women don’t know about these options, and far fewer understand the difference between these products (ie, water-based vs hybrid vs silicone lubricants).
Patients are more likely to comply with treatment when products are available in the office
Here in my small Midwestern city, there are 15 moisturizers or lubricants on the shelf at Rite-Aid; 38 at Walgreens. My patients are unlikely to sort through the options, and many tell me they use “whatever my husband bought for me.” I’ve had a menopausal patient report using K-Y Intense. For an atrophic postmenopausal vagina, that product is very uncomfortable. She was now downright scared to try additional options.
This is the patient to whom I explain lubricant options by placing on her hand dabs of water-based, hybrid, and silicone products. I let her note the differences in feel and consistency, allowing her to determine which is most acceptable to her. My office offers a small selection of each of those products so she can leave the appointment with the selection of her choice—no label reading or comparison shopping in the aisle of Walgreens as her neighbor walks by; no accidental purchase of products that increase rather than relieve her discomfort.
Among US women aged 45 to 64, 30% have urinary incontinence, one of the conditions that correlate with an increased incidence of sexual distress.4 Incontinence treatments may include behavioral modification, medication, a referral to physical therapy, perhaps surgery, and Kegel exercises.
Physical therapists are masterful in their ability to successfully treat urinary incontinence, but the limitations of time, cost, and insurance coverage are a deterrent to successful follow-through. When performed correctly, Kegel exercises may result in a cure for stress urinary incontinence in 50% to 70% of women.5 Exercise tools can help women isolate the pelvic floor muscles and perform Kegel exercises correctly, but I don’t find them at Rite-Aid or Walgreens. In my office, I can describe how these tools can assist women in doing their exercises, can show them what the tools look like and how they are used, and have them available for purchase.
When vaginal dilators are indicated for vaginismus or postradiation changes to the vagina, I show the patient the dilators and how to use them, and I have them available for purchase.
Now, what about those vibrators?
An Internet-based survey of more than 2,000 women aged 18 to 60 indicated that 52% have used a vibrator.6 In my own (unpublished) survey of more than 100 women, 43% indicated they had used a sex toy or device (about equally in partnered and solo sex). About half of women have experience with a vibrator—which means half do not. That’s in spite of the fact that vibrator use correlates with more sexual satisfaction. It may help with a decrease in latency and increase the intensity of the orgasmic response. I commonly hear women say, “Orgasm takes so much longer and comes and goes so quickly it’s hardly worth it.” Those are the women who may benefit from introducing a vibrator.
A 52-year-old postmenopausal woman presented to me with inability to orgasm, estimating 5 years had passed since she’d last experienced one. She had a diagnosis of multiple sclerosis and was taking a selective serotonin reuptake inhibitor. She had tried a vibrator in the past, without success. As a physician, I knew that she needed a more powerful motor for more intense stimulation. I was able to let her feel the difference and obtain the appropriate vibrator. Imagine her appreciation when she returned after achieving success.
The suggestion that a woman introduce a vibrator into her sexual life is nuanced. It is important that we as providers explore and understand our patients’ concerns, beliefs, and attitudes toward this option. Women may lack information or have misinformation that can be addressed in a sensitive, professional way in the privacy and safety of an office visit.
Once a woman understands the benefits a vibrator may offer, how does she proceed? I live in a community that does not have a “sex shop,” and I don’t feel comfortable referring patients to one that is 30 miles away.
Google offers 23,000,000 results for “vibrators.” Amazon provides 400 pages of products. Adam and Eve, a leading online “sex toys” shop, offers 524 vibrators. To get to the options, a patient would need to navigate some distasteful images, guess at quality and safety, and wouldn’t find much relevant health information.
Three years ago I attended an Adult Novelty Expo. I can attest to the variety of products on the market, most of which I would never consider advising a patient to use. Some can give off an awful “toxic smell,” which I would prefer my patients not have in contact with their genitals. Some are extremely complex—one vibrator introduced at the expo boasted 42 different functions with 8 control buttons. A small, curated selection of safe, well-designed, and effective vibrators is welcomed in my office.
Providing vibrators for purchase in the office can bypass confusion
Part of what patients value is that they can evaluate and purchase these products with privacy, confidentiality, and convenience in a clinician’s office. These purchases result from a safe, nonjudgmental, informed discussion with their provider, in the context of a relationship that has been developed over time.
Feedback from happy customers/ patients. Knowing there is controversy about health-care providers selling products, I’m upfront in asking for feedback from patients and online customers. These are some of their comments:
- “I trust you completely. You have a history of taking a personal interest in my health.”
- “I am personally grateful for the store and have passed it on to my friends, because where else are we going to go for these things? … It seems like another way you are keeping your patients’ health and well-being in the forefront of your practice. You have created a safe place for us to reconnect with an important part of ourselves.”
- “Anyone who comes back to a professional after the first ‘I’ll give him a shot’ visit does so because she trusts that person. If she didn’t, she wouldn’t be there. One of the things she trusts is your judgment.”
- “If you, in your professional opinion and personal experience, honestly believe a lotion, device, or book to be beneficial, there is nothing wrong with saying so (and one can make the case that you are shirking your duties to your readers and patients if you do NOT mention these things).”
Their comments reinforce my earlier point that the trust we develop with our patients is very valuable and must not be compromised.
Opportunity for revenue
I’ve focused on issues of comprehensive, practical service to patients because that is my primary motivation for offering products through my practice. That said, there are, of course, opportunities for revenue through retail in or in conjunction with your practice. (See “How to expand your services,” above.) That revenue can offset other cost pressures or contribute to overall profitability. Or, you could decide to provide products at cost as a service. Either way, you need to be thorough in investigating the financial implications and overhead requirements.
In addition to locating and testing products, there will be time invested in setting up accounts, managing cash flow and inventory and conducting the transactions. You may find, as I have, that depending on scale and resources available, it’s most effective for a practice to work with a distribution or retail partner.
There are any number of ways to incorporate products into a practice, which makes a comprehensive guide to getting started difficult. These actions will help you start the process, though, and avoid some missteps along the way.
- Verify that policies are friendly. Check to be sure that what you’re planning will comply with the policies that govern your practice. My practice is part of a nonprofit hospital network, and it was important to be upfront and assure appropriate disclosures.
- Determine the range of products that it would be helpful to offer. The community and resources where you practice, as well as the focus of your practice, can provide direction on what variety of products you offer. I live in a small Midwestern city without a lot of options, so a full spectrum is important to my patients. If other sources were readily available, I might offer fewer types of products.
- Assure office staff capacity. While you’re deciding how ambitious to be, think through who in your office will take on what roles. Who will research products? How much inventory will you maintain? Who will restock the inventory? You can structure your plans appropriately. An online store, where your patients or staff use a Web site to purchase products that are shipped directly to patients, requires the least effort. At the other end of the spectrum is a collection of products in-office, ready for patients to purchase and take home.
- Set your criteria for products. There are many, many products available; they are not all created equal. For vibrators, for example, I look for safe materials and quality of design, such as “no pinch” points and ease of cleaning. Beyond that, I know my patients need a stronger motor for sensation and are flummoxed by complex controls.
- Research products that meet your criteria. At this point, resources diverge depending on your intent. A Web site like MiddlesexMD.com curates products across categories and manufacturers for you, with a special focus on sexual health. Other options include retailers like Amazon.com, goodvibes.com, or evesgarden.com.
- Contact distributors or manufacturers. Every company will offer different pricing, discounts, minimum order levels, and delivery. You’ll need to match the capabilities offered with the intent and capacity you’ve outlined. For practices who don’t have those resources, we at MiddlesexMD provide an affiliate program.
- Train your staff on both products and process. Because my staff has conversations with patients, I want to be sure that they’re all comfortable talking about and answering questions about products I may recommend. And, of course, they need to know how to charge for products, who to contact with questions, and how to reorder.
For me, selling products makes sense
I practiced medicine for years before bringing products into my office. My relationships with patients were well-established. But I’ve seen repeatedly and first-hand that women are more likely to follow through when I can show them what lubricants feel like or how a vibrator functions. I’m not sure I would still have an electric toothbrush if my dentist didn’t offer them for sale.
When my patients can walk out with products they’re ready to use—rather than with one more research project for their to-do lists—well, I think that’s useful and convenient. My patients, especially those who’ve revived their sexuality when they’d nearly given up, tell me it’s an invaluable part of my practice.
We want to hear from you! Tell us what you think.
1. Shifren JL, Monz BU, Russo PA, Segreti A, Johannes CB. Sexual problems and distress in United States women: prevalence and correlates. Obstet Gynecol. 2008;112(5):970-978.
2. Ratner ES, Richter CE, Minkin MJ, Foran-Tuller KA. How to talk about sexual issues with cancer patients. Beginning the dialogue. Contemp OB/GYN. 2012;57(5):40-51.
3. Sobecki JN, Curlin FA, Rasinski KA, Lindau ST. What we don’t talk about when we don’t talk about sex: Results of a national survey os U.S. Obstetrician/Gynecologists. J Sex Med. 2012;9(5):1285-1294.
4. Hunskaar S, Arnold EP, Burgio K, Diokno AC, Herzog AR, Mallett VT. Epidemiology and natural history of urinary incontinence. Int Urogynecol J Pelvic Floor Dysfunct. 2000;11(5):301-319.
5. Dumoulin C, Hay-Smith J. Pelvic floor muscle training versus no treatment for urinary incontinence in women: A Cochrane systematic review. Eur J Phys Rehabil Med. 2008;44(1):47-63.
6. Herbenick D, Reece M, Sanders S, Dodge B, Ghassemi A, Fortenberry JD. Prevalence and characteristics of vibrator use by women in the United States: Results from a nationally representative study. J Sex Med. 2009;6(7):1857-1866.
Click here to access articles on sexual health published in OBG Management in 2012.
As health-care professionals, we are well trained in the importance of taking a detailed, targeted history and incorporating appropriate assessments to determine a treatment plan, which often includes prescriptions, a medical procedure, or a referral. Our extensive training allows us to make and implement expert decisions.
At the same time, I’ve found that the most valued aspect of our job is the relationships we develop with our patients. We know details that probably no one else knows about them. This is likely most true for us clinicians whose practices address women’s sexual health. A deep level of trust develops over time.
As part of that relationship of trust, we have unique opportunities to influence not only a woman’s health but also her overall quality of life. What I found over the years is that many women don’t necessarily need any prescription or referral or procedure. What they do often need is guidance, direction, and encouragement.
Some of my greatest achievements in delivering health care have not happened in a delivery room or because of a medication I prescribed. Instead, they have occurred in the office where, for example, I see a woman who took my recommendations 1 year ago to change her diet and begin exercising. Now, 30 lb lighter, she feels like a new person. This is one example of a knowledgeable physician applying “extensive training and expert decisions” to incorporate low-tech treatments with great success.
So what does this have to do with vibrators in a doctor’s office? Well, without many FDA-approved medications to treat sexual dysfunction, we are left with over-the-counter possibilities and such “low-tech” options as counseling and guidance.
Sexual dysfunction is common among our patients
According to the National Health and Social Life Survey, 43% of women are affected by some aspect of sexual dysfunction, and 22% associate that dysfunction with personal distress. The distress was highest for women aged 45 to 64. Additional correlates included poor self-assessed health, low education level, depression, anxiety, thyroid conditions, and urinary incontinence.1 Sexual complaints among women diagnosed with cancer can be as high as 90%.2
Does this describe anyone in your practice? We can agree that women’s sexual issues are common, whether we acknowledge that in our practice by querying women on their sexual health or not (and many of us are not).
We’re not talking to our patients about their sexual health enough. In a 2012 survey of more than 1,000 ObGyns about whether they talk with their patients about sex,3 investigators found that:
- 63% routinely ask whether their patient is sexually active
- 40% routinely ask if the patient is having any problems regarding sex
- 29% ask about sexual satisfaction
- 28% ask about sexual orientation or identity
- 14% ask about sexual pleasure.
Whatever the reason, talking about sexual health is easier for some people than for others, and that goes for both clinicians and patients.
What are our treatment options if sexual dysfunction is identified?
Unfortunately, the therapeutic armamentarium for women’s sexual health is limited—perhaps a contributor to many clinicians’ reluctance to broach the topic. Pharmacologically, we have no FDA-approved treatments for female sexual dysfunction except localized estrogen treatment for vulvovaginal atrophy and the Eros device for arousal and orgasmic disorders.
There are off-label options of testosterone, buproprion, or sildenafil citrate, but many practitioners and patients are reluctant to consider these options specifically because they are off label.
Over-the-counter. These options are available for women, and can be helpful. For patients with discomfort due to vaginal dryness, it is important to educate them on the benefits of vaginal moisturizers and lubricants. Many women don’t know about these options, and far fewer understand the difference between these products (ie, water-based vs hybrid vs silicone lubricants).
Patients are more likely to comply with treatment when products are available in the office
Here in my small Midwestern city, there are 15 moisturizers or lubricants on the shelf at Rite-Aid; 38 at Walgreens. My patients are unlikely to sort through the options, and many tell me they use “whatever my husband bought for me.” I’ve had a menopausal patient report using K-Y Intense. For an atrophic postmenopausal vagina, that product is very uncomfortable. She was now downright scared to try additional options.
This is the patient to whom I explain lubricant options by placing on her hand dabs of water-based, hybrid, and silicone products. I let her note the differences in feel and consistency, allowing her to determine which is most acceptable to her. My office offers a small selection of each of those products so she can leave the appointment with the selection of her choice—no label reading or comparison shopping in the aisle of Walgreens as her neighbor walks by; no accidental purchase of products that increase rather than relieve her discomfort.
Among US women aged 45 to 64, 30% have urinary incontinence, one of the conditions that correlate with an increased incidence of sexual distress.4 Incontinence treatments may include behavioral modification, medication, a referral to physical therapy, perhaps surgery, and Kegel exercises.
Physical therapists are masterful in their ability to successfully treat urinary incontinence, but the limitations of time, cost, and insurance coverage are a deterrent to successful follow-through. When performed correctly, Kegel exercises may result in a cure for stress urinary incontinence in 50% to 70% of women.5 Exercise tools can help women isolate the pelvic floor muscles and perform Kegel exercises correctly, but I don’t find them at Rite-Aid or Walgreens. In my office, I can describe how these tools can assist women in doing their exercises, can show them what the tools look like and how they are used, and have them available for purchase.
When vaginal dilators are indicated for vaginismus or postradiation changes to the vagina, I show the patient the dilators and how to use them, and I have them available for purchase.
Now, what about those vibrators?
An Internet-based survey of more than 2,000 women aged 18 to 60 indicated that 52% have used a vibrator.6 In my own (unpublished) survey of more than 100 women, 43% indicated they had used a sex toy or device (about equally in partnered and solo sex). About half of women have experience with a vibrator—which means half do not. That’s in spite of the fact that vibrator use correlates with more sexual satisfaction. It may help with a decrease in latency and increase the intensity of the orgasmic response. I commonly hear women say, “Orgasm takes so much longer and comes and goes so quickly it’s hardly worth it.” Those are the women who may benefit from introducing a vibrator.
A 52-year-old postmenopausal woman presented to me with inability to orgasm, estimating 5 years had passed since she’d last experienced one. She had a diagnosis of multiple sclerosis and was taking a selective serotonin reuptake inhibitor. She had tried a vibrator in the past, without success. As a physician, I knew that she needed a more powerful motor for more intense stimulation. I was able to let her feel the difference and obtain the appropriate vibrator. Imagine her appreciation when she returned after achieving success.
The suggestion that a woman introduce a vibrator into her sexual life is nuanced. It is important that we as providers explore and understand our patients’ concerns, beliefs, and attitudes toward this option. Women may lack information or have misinformation that can be addressed in a sensitive, professional way in the privacy and safety of an office visit.
Once a woman understands the benefits a vibrator may offer, how does she proceed? I live in a community that does not have a “sex shop,” and I don’t feel comfortable referring patients to one that is 30 miles away.
Google offers 23,000,000 results for “vibrators.” Amazon provides 400 pages of products. Adam and Eve, a leading online “sex toys” shop, offers 524 vibrators. To get to the options, a patient would need to navigate some distasteful images, guess at quality and safety, and wouldn’t find much relevant health information.
Three years ago I attended an Adult Novelty Expo. I can attest to the variety of products on the market, most of which I would never consider advising a patient to use. Some can give off an awful “toxic smell,” which I would prefer my patients not have in contact with their genitals. Some are extremely complex—one vibrator introduced at the expo boasted 42 different functions with 8 control buttons. A small, curated selection of safe, well-designed, and effective vibrators is welcomed in my office.
Providing vibrators for purchase in the office can bypass confusion
Part of what patients value is that they can evaluate and purchase these products with privacy, confidentiality, and convenience in a clinician’s office. These purchases result from a safe, nonjudgmental, informed discussion with their provider, in the context of a relationship that has been developed over time.
Feedback from happy customers/ patients. Knowing there is controversy about health-care providers selling products, I’m upfront in asking for feedback from patients and online customers. These are some of their comments:
- “I trust you completely. You have a history of taking a personal interest in my health.”
- “I am personally grateful for the store and have passed it on to my friends, because where else are we going to go for these things? … It seems like another way you are keeping your patients’ health and well-being in the forefront of your practice. You have created a safe place for us to reconnect with an important part of ourselves.”
- “Anyone who comes back to a professional after the first ‘I’ll give him a shot’ visit does so because she trusts that person. If she didn’t, she wouldn’t be there. One of the things she trusts is your judgment.”
- “If you, in your professional opinion and personal experience, honestly believe a lotion, device, or book to be beneficial, there is nothing wrong with saying so (and one can make the case that you are shirking your duties to your readers and patients if you do NOT mention these things).”
Their comments reinforce my earlier point that the trust we develop with our patients is very valuable and must not be compromised.
Opportunity for revenue
I’ve focused on issues of comprehensive, practical service to patients because that is my primary motivation for offering products through my practice. That said, there are, of course, opportunities for revenue through retail in or in conjunction with your practice. (See “How to expand your services,” above.) That revenue can offset other cost pressures or contribute to overall profitability. Or, you could decide to provide products at cost as a service. Either way, you need to be thorough in investigating the financial implications and overhead requirements.
In addition to locating and testing products, there will be time invested in setting up accounts, managing cash flow and inventory and conducting the transactions. You may find, as I have, that depending on scale and resources available, it’s most effective for a practice to work with a distribution or retail partner.
There are any number of ways to incorporate products into a practice, which makes a comprehensive guide to getting started difficult. These actions will help you start the process, though, and avoid some missteps along the way.
- Verify that policies are friendly. Check to be sure that what you’re planning will comply with the policies that govern your practice. My practice is part of a nonprofit hospital network, and it was important to be upfront and assure appropriate disclosures.
- Determine the range of products that it would be helpful to offer. The community and resources where you practice, as well as the focus of your practice, can provide direction on what variety of products you offer. I live in a small Midwestern city without a lot of options, so a full spectrum is important to my patients. If other sources were readily available, I might offer fewer types of products.
- Assure office staff capacity. While you’re deciding how ambitious to be, think through who in your office will take on what roles. Who will research products? How much inventory will you maintain? Who will restock the inventory? You can structure your plans appropriately. An online store, where your patients or staff use a Web site to purchase products that are shipped directly to patients, requires the least effort. At the other end of the spectrum is a collection of products in-office, ready for patients to purchase and take home.
- Set your criteria for products. There are many, many products available; they are not all created equal. For vibrators, for example, I look for safe materials and quality of design, such as “no pinch” points and ease of cleaning. Beyond that, I know my patients need a stronger motor for sensation and are flummoxed by complex controls.
- Research products that meet your criteria. At this point, resources diverge depending on your intent. A Web site like MiddlesexMD.com curates products across categories and manufacturers for you, with a special focus on sexual health. Other options include retailers like Amazon.com, goodvibes.com, or evesgarden.com.
- Contact distributors or manufacturers. Every company will offer different pricing, discounts, minimum order levels, and delivery. You’ll need to match the capabilities offered with the intent and capacity you’ve outlined. For practices who don’t have those resources, we at MiddlesexMD provide an affiliate program.
- Train your staff on both products and process. Because my staff has conversations with patients, I want to be sure that they’re all comfortable talking about and answering questions about products I may recommend. And, of course, they need to know how to charge for products, who to contact with questions, and how to reorder.
For me, selling products makes sense
I practiced medicine for years before bringing products into my office. My relationships with patients were well-established. But I’ve seen repeatedly and first-hand that women are more likely to follow through when I can show them what lubricants feel like or how a vibrator functions. I’m not sure I would still have an electric toothbrush if my dentist didn’t offer them for sale.
When my patients can walk out with products they’re ready to use—rather than with one more research project for their to-do lists—well, I think that’s useful and convenient. My patients, especially those who’ve revived their sexuality when they’d nearly given up, tell me it’s an invaluable part of my practice.
We want to hear from you! Tell us what you think.
Click here to access articles on sexual health published in OBG Management in 2012.
As health-care professionals, we are well trained in the importance of taking a detailed, targeted history and incorporating appropriate assessments to determine a treatment plan, which often includes prescriptions, a medical procedure, or a referral. Our extensive training allows us to make and implement expert decisions.
At the same time, I’ve found that the most valued aspect of our job is the relationships we develop with our patients. We know details that probably no one else knows about them. This is likely most true for us clinicians whose practices address women’s sexual health. A deep level of trust develops over time.
As part of that relationship of trust, we have unique opportunities to influence not only a woman’s health but also her overall quality of life. What I found over the years is that many women don’t necessarily need any prescription or referral or procedure. What they do often need is guidance, direction, and encouragement.
Some of my greatest achievements in delivering health care have not happened in a delivery room or because of a medication I prescribed. Instead, they have occurred in the office where, for example, I see a woman who took my recommendations 1 year ago to change her diet and begin exercising. Now, 30 lb lighter, she feels like a new person. This is one example of a knowledgeable physician applying “extensive training and expert decisions” to incorporate low-tech treatments with great success.
So what does this have to do with vibrators in a doctor’s office? Well, without many FDA-approved medications to treat sexual dysfunction, we are left with over-the-counter possibilities and such “low-tech” options as counseling and guidance.
Sexual dysfunction is common among our patients
According to the National Health and Social Life Survey, 43% of women are affected by some aspect of sexual dysfunction, and 22% associate that dysfunction with personal distress. The distress was highest for women aged 45 to 64. Additional correlates included poor self-assessed health, low education level, depression, anxiety, thyroid conditions, and urinary incontinence.1 Sexual complaints among women diagnosed with cancer can be as high as 90%.2
Does this describe anyone in your practice? We can agree that women’s sexual issues are common, whether we acknowledge that in our practice by querying women on their sexual health or not (and many of us are not).
We’re not talking to our patients about their sexual health enough. In a 2012 survey of more than 1,000 ObGyns about whether they talk with their patients about sex,3 investigators found that:
- 63% routinely ask whether their patient is sexually active
- 40% routinely ask if the patient is having any problems regarding sex
- 29% ask about sexual satisfaction
- 28% ask about sexual orientation or identity
- 14% ask about sexual pleasure.
Whatever the reason, talking about sexual health is easier for some people than for others, and that goes for both clinicians and patients.
What are our treatment options if sexual dysfunction is identified?
Unfortunately, the therapeutic armamentarium for women’s sexual health is limited—perhaps a contributor to many clinicians’ reluctance to broach the topic. Pharmacologically, we have no FDA-approved treatments for female sexual dysfunction except localized estrogen treatment for vulvovaginal atrophy and the Eros device for arousal and orgasmic disorders.
There are off-label options of testosterone, buproprion, or sildenafil citrate, but many practitioners and patients are reluctant to consider these options specifically because they are off label.
Over-the-counter. These options are available for women, and can be helpful. For patients with discomfort due to vaginal dryness, it is important to educate them on the benefits of vaginal moisturizers and lubricants. Many women don’t know about these options, and far fewer understand the difference between these products (ie, water-based vs hybrid vs silicone lubricants).
Patients are more likely to comply with treatment when products are available in the office
Here in my small Midwestern city, there are 15 moisturizers or lubricants on the shelf at Rite-Aid; 38 at Walgreens. My patients are unlikely to sort through the options, and many tell me they use “whatever my husband bought for me.” I’ve had a menopausal patient report using K-Y Intense. For an atrophic postmenopausal vagina, that product is very uncomfortable. She was now downright scared to try additional options.
This is the patient to whom I explain lubricant options by placing on her hand dabs of water-based, hybrid, and silicone products. I let her note the differences in feel and consistency, allowing her to determine which is most acceptable to her. My office offers a small selection of each of those products so she can leave the appointment with the selection of her choice—no label reading or comparison shopping in the aisle of Walgreens as her neighbor walks by; no accidental purchase of products that increase rather than relieve her discomfort.
Among US women aged 45 to 64, 30% have urinary incontinence, one of the conditions that correlate with an increased incidence of sexual distress.4 Incontinence treatments may include behavioral modification, medication, a referral to physical therapy, perhaps surgery, and Kegel exercises.
Physical therapists are masterful in their ability to successfully treat urinary incontinence, but the limitations of time, cost, and insurance coverage are a deterrent to successful follow-through. When performed correctly, Kegel exercises may result in a cure for stress urinary incontinence in 50% to 70% of women.5 Exercise tools can help women isolate the pelvic floor muscles and perform Kegel exercises correctly, but I don’t find them at Rite-Aid or Walgreens. In my office, I can describe how these tools can assist women in doing their exercises, can show them what the tools look like and how they are used, and have them available for purchase.
When vaginal dilators are indicated for vaginismus or postradiation changes to the vagina, I show the patient the dilators and how to use them, and I have them available for purchase.
Now, what about those vibrators?
An Internet-based survey of more than 2,000 women aged 18 to 60 indicated that 52% have used a vibrator.6 In my own (unpublished) survey of more than 100 women, 43% indicated they had used a sex toy or device (about equally in partnered and solo sex). About half of women have experience with a vibrator—which means half do not. That’s in spite of the fact that vibrator use correlates with more sexual satisfaction. It may help with a decrease in latency and increase the intensity of the orgasmic response. I commonly hear women say, “Orgasm takes so much longer and comes and goes so quickly it’s hardly worth it.” Those are the women who may benefit from introducing a vibrator.
A 52-year-old postmenopausal woman presented to me with inability to orgasm, estimating 5 years had passed since she’d last experienced one. She had a diagnosis of multiple sclerosis and was taking a selective serotonin reuptake inhibitor. She had tried a vibrator in the past, without success. As a physician, I knew that she needed a more powerful motor for more intense stimulation. I was able to let her feel the difference and obtain the appropriate vibrator. Imagine her appreciation when she returned after achieving success.
The suggestion that a woman introduce a vibrator into her sexual life is nuanced. It is important that we as providers explore and understand our patients’ concerns, beliefs, and attitudes toward this option. Women may lack information or have misinformation that can be addressed in a sensitive, professional way in the privacy and safety of an office visit.
Once a woman understands the benefits a vibrator may offer, how does she proceed? I live in a community that does not have a “sex shop,” and I don’t feel comfortable referring patients to one that is 30 miles away.
Google offers 23,000,000 results for “vibrators.” Amazon provides 400 pages of products. Adam and Eve, a leading online “sex toys” shop, offers 524 vibrators. To get to the options, a patient would need to navigate some distasteful images, guess at quality and safety, and wouldn’t find much relevant health information.
Three years ago I attended an Adult Novelty Expo. I can attest to the variety of products on the market, most of which I would never consider advising a patient to use. Some can give off an awful “toxic smell,” which I would prefer my patients not have in contact with their genitals. Some are extremely complex—one vibrator introduced at the expo boasted 42 different functions with 8 control buttons. A small, curated selection of safe, well-designed, and effective vibrators is welcomed in my office.
Providing vibrators for purchase in the office can bypass confusion
Part of what patients value is that they can evaluate and purchase these products with privacy, confidentiality, and convenience in a clinician’s office. These purchases result from a safe, nonjudgmental, informed discussion with their provider, in the context of a relationship that has been developed over time.
Feedback from happy customers/ patients. Knowing there is controversy about health-care providers selling products, I’m upfront in asking for feedback from patients and online customers. These are some of their comments:
- “I trust you completely. You have a history of taking a personal interest in my health.”
- “I am personally grateful for the store and have passed it on to my friends, because where else are we going to go for these things? … It seems like another way you are keeping your patients’ health and well-being in the forefront of your practice. You have created a safe place for us to reconnect with an important part of ourselves.”
- “Anyone who comes back to a professional after the first ‘I’ll give him a shot’ visit does so because she trusts that person. If she didn’t, she wouldn’t be there. One of the things she trusts is your judgment.”
- “If you, in your professional opinion and personal experience, honestly believe a lotion, device, or book to be beneficial, there is nothing wrong with saying so (and one can make the case that you are shirking your duties to your readers and patients if you do NOT mention these things).”
Their comments reinforce my earlier point that the trust we develop with our patients is very valuable and must not be compromised.
Opportunity for revenue
I’ve focused on issues of comprehensive, practical service to patients because that is my primary motivation for offering products through my practice. That said, there are, of course, opportunities for revenue through retail in or in conjunction with your practice. (See “How to expand your services,” above.) That revenue can offset other cost pressures or contribute to overall profitability. Or, you could decide to provide products at cost as a service. Either way, you need to be thorough in investigating the financial implications and overhead requirements.
In addition to locating and testing products, there will be time invested in setting up accounts, managing cash flow and inventory and conducting the transactions. You may find, as I have, that depending on scale and resources available, it’s most effective for a practice to work with a distribution or retail partner.
There are any number of ways to incorporate products into a practice, which makes a comprehensive guide to getting started difficult. These actions will help you start the process, though, and avoid some missteps along the way.
- Verify that policies are friendly. Check to be sure that what you’re planning will comply with the policies that govern your practice. My practice is part of a nonprofit hospital network, and it was important to be upfront and assure appropriate disclosures.
- Determine the range of products that it would be helpful to offer. The community and resources where you practice, as well as the focus of your practice, can provide direction on what variety of products you offer. I live in a small Midwestern city without a lot of options, so a full spectrum is important to my patients. If other sources were readily available, I might offer fewer types of products.
- Assure office staff capacity. While you’re deciding how ambitious to be, think through who in your office will take on what roles. Who will research products? How much inventory will you maintain? Who will restock the inventory? You can structure your plans appropriately. An online store, where your patients or staff use a Web site to purchase products that are shipped directly to patients, requires the least effort. At the other end of the spectrum is a collection of products in-office, ready for patients to purchase and take home.
- Set your criteria for products. There are many, many products available; they are not all created equal. For vibrators, for example, I look for safe materials and quality of design, such as “no pinch” points and ease of cleaning. Beyond that, I know my patients need a stronger motor for sensation and are flummoxed by complex controls.
- Research products that meet your criteria. At this point, resources diverge depending on your intent. A Web site like MiddlesexMD.com curates products across categories and manufacturers for you, with a special focus on sexual health. Other options include retailers like Amazon.com, goodvibes.com, or evesgarden.com.
- Contact distributors or manufacturers. Every company will offer different pricing, discounts, minimum order levels, and delivery. You’ll need to match the capabilities offered with the intent and capacity you’ve outlined. For practices who don’t have those resources, we at MiddlesexMD provide an affiliate program.
- Train your staff on both products and process. Because my staff has conversations with patients, I want to be sure that they’re all comfortable talking about and answering questions about products I may recommend. And, of course, they need to know how to charge for products, who to contact with questions, and how to reorder.
For me, selling products makes sense
I practiced medicine for years before bringing products into my office. My relationships with patients were well-established. But I’ve seen repeatedly and first-hand that women are more likely to follow through when I can show them what lubricants feel like or how a vibrator functions. I’m not sure I would still have an electric toothbrush if my dentist didn’t offer them for sale.
When my patients can walk out with products they’re ready to use—rather than with one more research project for their to-do lists—well, I think that’s useful and convenient. My patients, especially those who’ve revived their sexuality when they’d nearly given up, tell me it’s an invaluable part of my practice.
We want to hear from you! Tell us what you think.
1. Shifren JL, Monz BU, Russo PA, Segreti A, Johannes CB. Sexual problems and distress in United States women: prevalence and correlates. Obstet Gynecol. 2008;112(5):970-978.
2. Ratner ES, Richter CE, Minkin MJ, Foran-Tuller KA. How to talk about sexual issues with cancer patients. Beginning the dialogue. Contemp OB/GYN. 2012;57(5):40-51.
3. Sobecki JN, Curlin FA, Rasinski KA, Lindau ST. What we don’t talk about when we don’t talk about sex: Results of a national survey os U.S. Obstetrician/Gynecologists. J Sex Med. 2012;9(5):1285-1294.
4. Hunskaar S, Arnold EP, Burgio K, Diokno AC, Herzog AR, Mallett VT. Epidemiology and natural history of urinary incontinence. Int Urogynecol J Pelvic Floor Dysfunct. 2000;11(5):301-319.
5. Dumoulin C, Hay-Smith J. Pelvic floor muscle training versus no treatment for urinary incontinence in women: A Cochrane systematic review. Eur J Phys Rehabil Med. 2008;44(1):47-63.
6. Herbenick D, Reece M, Sanders S, Dodge B, Ghassemi A, Fortenberry JD. Prevalence and characteristics of vibrator use by women in the United States: Results from a nationally representative study. J Sex Med. 2009;6(7):1857-1866.
1. Shifren JL, Monz BU, Russo PA, Segreti A, Johannes CB. Sexual problems and distress in United States women: prevalence and correlates. Obstet Gynecol. 2008;112(5):970-978.
2. Ratner ES, Richter CE, Minkin MJ, Foran-Tuller KA. How to talk about sexual issues with cancer patients. Beginning the dialogue. Contemp OB/GYN. 2012;57(5):40-51.
3. Sobecki JN, Curlin FA, Rasinski KA, Lindau ST. What we don’t talk about when we don’t talk about sex: Results of a national survey os U.S. Obstetrician/Gynecologists. J Sex Med. 2012;9(5):1285-1294.
4. Hunskaar S, Arnold EP, Burgio K, Diokno AC, Herzog AR, Mallett VT. Epidemiology and natural history of urinary incontinence. Int Urogynecol J Pelvic Floor Dysfunct. 2000;11(5):301-319.
5. Dumoulin C, Hay-Smith J. Pelvic floor muscle training versus no treatment for urinary incontinence in women: A Cochrane systematic review. Eur J Phys Rehabil Med. 2008;44(1):47-63.
6. Herbenick D, Reece M, Sanders S, Dodge B, Ghassemi A, Fortenberry JD. Prevalence and characteristics of vibrator use by women in the United States: Results from a nationally representative study. J Sex Med. 2009;6(7):1857-1866.
Preventive Dental Services in Primary Care
Millions of Americans currently lack access to dental care and oral health preventive services because of financial and/or geographic obstacles.1,2 Although community water fluoridation has benefited many Americans during the past 50 years, difficulties in obtaining dental care is a persistent public health challenge.2 Primary care providers (PCPs) can help improve health population-wide by educating patients in good dental hygiene and offering preventive oral health services to patients who would otherwise go without them.
The chief components of preventive dental care include daily brushing with fluoride toothpaste, flossing, routine dental check-ups and professional dental cleanings provided by a licensed dentist and staff, and use of dental sealants and topical fluoride. During the primary care appointment, a routine intraoral exam, including inspection of both hard and soft oral tissues for leukoplakia, gross decay, stained dental structures, and other pathologic changes, is essential. Primary care patients who use any form of tobacco should undergo inspection of the intraoral surfaces of the lips and assessment for adenopathy of the surrounding lymph nodes.
Counseling, including recommending routine preventive dental services (and, for some patients, allaying their fears of seeing a dentist2,3), and positive reinforcement of good oral hygiene practices should be addressed during every primary care encounter. Identifying patients without access to professional dental care is important, as the PCP may be their only source of oral health education, including referrals to local dental clinics.
Application of topical fluoride is an important means of improving dental health, and a cost-effective procedure for both patients and providers4 (see “Fluoride Use,”2,3,5-16). By following published guidelines for use of topical fluoride varnish, PCPs can reduce decay and mineralize enamel, thereby protecting the patient against enamel erosion.6,11,17 This service can and should be offered in primary care offices to patients who lack access to routine dental care and are likely to benefit from this effective weapon against tooth decay.
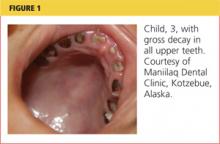
BEA, AGE 3
Bea lives with her mother and four older brothers and sisters. She is enrolled in Medicaid, but her mother has been unable to locate dental services for any of her children. With every call to a dental office within a 30-minute commute from their home, Bea’s mother has been told, “We don’t accept Medicaid.”
Under her state’s Medicaid plan, Bea has been seen for her primary health care needs since she was a newborn. She was bottle-fed from birth; her mother reports she has stopped taking the bottle to bed within the past few months. While conducting her three-year well-child exam, you note gross decay (see Figure 1).
Dental Services for Children
Nationwide, Medicaid is required to provide coverage for dental services for enrolled children, as well as those who qualify for the CHIP (Children’s Health Insurance Program).20 However, there is no mandate for privately owned dental practices to accept Medicaid payments.
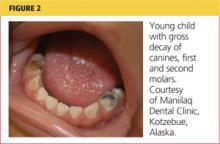
Lack of dental care is the greatest unmet health care need in American children.21 In children without health insurance, access to dental care is most challenging, and those enrolled in Medicaid are the second least likely to receive services. As a result of these and other factors, one-third of US children ages 3 to 5 have dental caries in their primary teeth, and one in every four US children is estimated to have untreated decay2,21-23 (see Figure 2). African-American and Hispanic children experience greater disparities in dental care than do those of other ethnicities.21
Dental decay is the most common chronic illness of childhood (five times as common as asthma), with higher incidence in low-income and minority preschool children.1,24,25 Tooth decay affects growth patterns and nutrition, with associated pain and infection interfering with school attendance.
The small number of dentists who treat children and are willing to accept Medicaid reimbursement is a significant concern. In most dental programs, dedicated pedodontic training is optional. The mismatch of number of qualified dentists to the growing population of such patients is projected to worsen in the coming years.23
Potential solutions to correct this mismatch of dental services accessible to the underserved have included26-31:
- Addition of paraprofessional dental therapists (persons who have received two years’ training in a specific academic program) to the health care team, in public health clinics, schools, and other settings
- Interprofessional dental education (ie, cross-training physicians, NPs, PAs, and RNs in preventive dental services)
- An increasingly diverse array of competent providers who are trained, authorized, and compensated to provide evidence-based care
- Removing licensure restrictions for non–US-trained dentists
- Financial incentives for dentists to practice in dental Health Professional Shortage Areas
- Improved education of parents regarding the benefits of routine dental services.
The Institute of Medicine (IOM)26 has challenged state legislatures to mandate a one-year dental residency to be completed among underserved populations before dental licensure is granted. This challenge reflects the IOM’s acknowledgement that dental services, essential to patients’ well-being, are grossly lacking in the US.
Dental Care Education for Parents
Healthy baby teeth help children eat well and speak clearly. Decayed and abscessed teeth cause the same degree of pain and suffering in young children as they do in adults. Thus, regardless of ethnicity or socioeconomic status, all parents should be routinely encouraged to oversee daily dental hygiene practices for their children; it is important for PCPs to explain to parents their expectations in this regard.
Dental care for children should begin as soon as the first tooth erupts. New teeth should be cleaned daily with a soft cloth and inspected by parents for any discolorations. From age 2 years until children are able to clean all dental surfaces adequately (age 7 or 8), parents should brush their children’s teeth at least once daily with a toothbrush, after applying a thin film of fluoride toothpaste. Children should undergo their first professional dental check-up at about age 1 year.19,26
Parents must be made aware of other factors that can impair their children’s dental health. For example, the foods most likely to promote dental decay contain highly fermentable carbohydrates, are highly processed and sticky, and cause oral pH levels to fall below 5.5 (at which point demineralization may occur).32 Examples include presweetened cereals, dried fruits, cookies, and potato chips. Cheese, peanuts, meat, and eggs are less cariogenic.
Other important teaching points are “don’ts”: putting a baby to bed with a bottle that contains anything but a sugar-free liquid; using spill-proof spouted cups filled with juice or other sugary drinks all day; serving a large proportion of cariogenic foods.
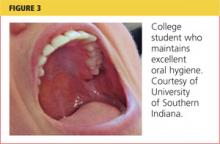
BREANNE, AGE 22
Breanne attends a community college, where she is studying business. She reports to the college health clinic, complaining of an intermittent sharp pain in one of her lower molars over the past four days, occurring when she eats or drinks anything cold.
Before she turned 21, Breanne received routine dental services under her parents’ insurance. Now a full-time student no longer covered by her parents’ plan, she has no income with which to purchase dental coverage. Years ago, Breanne received dental sealants on all of her molars in a school-based program, and she routinely brushes daily (see Figure 3). She says she “usually remembers to floss.”
Dental Concerns in Young Adults
Up to one-third of Americans, including college-age students, lack access to oral health services.33 Use of annual dental services, including school-based care, significantly reduces young adults’ risk for tooth loss.34 From early adulthood on, poor dental health is associated with cardiovascular and respiratory disease, cerebral ischemia, diabetes, difficulty with chewing and oral pain, and poor self-image.24
PCPs should emphasize the importance of preventive services as they examine the oral structure during routine health care encounters. Adding the question, “When did you last have a dental exam?” can launch an effective discussion about the importance of good dental health.
Tooth surfaces with pits and fissures are the most vulnerable to decay. Dental sealants can protect these vulnerable areas, reducing decay by 60% to 82%.2,32,35 Sealants are the most effective preventive measure for reducing cavity formation; they are not harmful to fillings and can actually reduce the progression of decay when applied over small existing carious lesions.35 Currently, however, only 30% and 38% of children and adolescents, respectively, have undergone application of sealants.36 Thus, it is vital for PCPs to recommend dental sealants to all their patients.35,37,38
Young adults should also be questioned about their exercise patterns. While daily exercise is of benefit to good health, young adults who engage in too-frequent, overly vigorous exercise have been found vulnerable to dental erosion.39 This irreversible condition can result from the rapid breathing, sweat-induced dehydration, and decreased salivary flow associated with strenuous exercise. Reduced salivary flow disrupts the protection against erosive acids afforded by bathing of teeth with saliva, leading to a fivefold greater risk for developing erosions. Patients should be encouraged to hydrate before exercise to reduce these risks.39
Dental Health Notes for Young Women
For female patients of childbearing age like Breanne, dental health is important not only for the patient, but for her future children as well. Poor maternal dental health has been associated with preterm births and low birth weights.40-43 Women who have health insurance, who are in good general health, and who regularly visit a primary care medical provider are also the most likely to see a dental care provider during pregnancy.40
Pregnancy is associated with a risk for poor dental care in any population.42,44 During pregnancy, Medicaid-enrolled women are 30% less likely to see a dentist than women with private health insurance, and women with no health insurance are 70% less likely to see a dentist.44 Among pregnant women who experience a dental problem, less than half will schedule a dental appointment. Postponing dental care till after delivery incurs additional risk, as the new mother will be busy with her child and may have lost her health care coverage in the interim.
Dental concerns commonly encountered during pregnancy are listed in Table 3.41,42 There is also evidence that maternal transmission of mutans streptococci can lead to early childhood caries,42,45 further highlighting the importance of dental care during pregnancy (eg, use of chlorhexidine rinses in pregnant women with dental caries42). The association between periodontal disease and preterm delivery and lower birth weights40-43 makes it critical to reduce maternal periodontal inflammation through timely dental referrals.
SAUL, AGE 42
Saul, a lifelong resident of Mississippi, has worked in construction since age 18. He has never had health or dental insurance and thus he rarely seeks preventive services. As he recalls, he has had “two fillings in my jaw teeth since I was a kid,” but otherwise has had no dental care. He brushes daily to avoid having to see a dentist. He is presenting today as a new patient only because he has a rash.
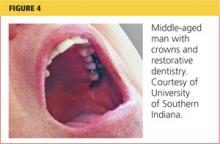
Benefits of Fluoride Use During Middle Age
Dental care needs peak between ages 55 and 64, with the majority of care prompted by dental decay46 (see Figure 4). A discussion about dental health should be raised when the PCP completes the review of systems or while discussing other preventive services. The option of fluoride varnish should not be overlooked in a patient of any age, particularly one with a recognizable deficiency of preventive dental services. Explaining the benefits of topical fluoride varnish (see “Fluoride Use”) and offering an opportunity to receive it in the office may persuade a patient like Saul to take advantage of this proven anticarious procedure.
Patients who undergo this safe procedure should be reminded to continue brushing daily with fluoridated toothpaste and drinking fluoridated water (if it is available).50
GILL, AGE 77
Gill grew up in Grant County, New Mexico, in an area not far from Silver City; there, fluoride supplementation in the local water did not begin until Gill was about 40. His family never stressed oral hygiene, and his remaining teeth show gross plaque and decay.
He sees you routinely for his chronic illnesses, and today he has presented for his annual physical. Gill has Medicare without supplemental insurance.
Dental Care in the Medicare Years
Medicare does not cover preventive dental services unless hospitalization is required; in that case, it is covered under Medicare Part A.51 Publicly supported dental services are primarily offered for children, leaving Medicare recipients responsible for the costs of dental care. As a last resort, some patients will seek dental care at a hospital emergency department (ED). Most often prompted to visit the ED for relief of pain triggered by dental fractures or abscesses, patients usually leave with pain medications and antibiotics only; their general dental health needs remain unmet.52
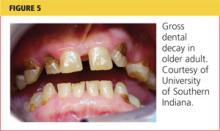
The most common dental concerns among elderly patients include edentulism (toothlessness, which is reported in one in four Medicare beneficiaries1), xerostomia (dry mouth), periodontal disease, ill-fitting or worn prosthetics, and progressive decay affecting fractured teeth1 (see Figure 5). As many elderly patients endure a combination of these uncomfortable, possibly painful, conditions, it is important for the PCP to inquire about them during the physical exam and the review of symptoms. At each step, a window of opportunity opens to make appropriate recommendations.
Significant Comorbidities
It is important for PCPs to recognize the association between certain diseases and poor oral health. With the progression of age, the risk increases for type 2 diabetes. Among older diabetic patients with periodontitis (and the systemic inflammation associated with it), the difficulty of maintaining glycemic control increases as well.53 Conversely, diabetic patients who are immunocompromised are at elevated risk for periodontitis. In more than 20 years of formal data collection, periodontists have identified and confirmed an anecdotal relationship between diabetes and severe periodontal disease.26
Elderly patients with poor dental health are also at increased risk for chronic kidney disease. Periodontal disease alone increases the risk for nephropathy 1.5-fold to twofold.54Finally, patients’ risk for oral cancer increases with age. Known risk factors for oral cancers include smoking, use of chewing tobacco, alcohol consumption, chronic friction, and exposure to ultraviolet radiation.55 Survival rates for oral cancers are poor, reemphasizing the need for a thorough oral exam during primary care visits.
Other Challenges for Older Patients
Members of ethnic minority groups and lower-income populations, men, patients with cognitive impairment, and persons who never completed high school are the most likely to avoid, discontinue, or lack access to dental services.56 Loss of dental coverage on retirement is associated with a significant decline in elderly patients’ use of dental services. Though now living on a fraction of their preretirement income, millions of retirees are required to pay out-of-pocket for dental services.57
Topical fluoride varnish alone cannot alleviate this multimillion dollar concern, but using it to reinforce retirees’ remaining enamel and prevent caries can help reduce their dental care expenditures. However, until federal and state policymakers act to cover preventive services,2,58 PCPs have little to offer elderly patients beyond oral hygiene education and in-office application of topical fluoride.
Strategies to Reinforce Dental Health
In practices where electronic medical records are in use, a recommendation for dental services should be printed as a patient reminder. Printed recommendations and referrals from PCPs do improve patient compliance.59 In addition to the printed office summary, older patients can be handed a summary of oral health suggestions from the National Institute on Aging, NIH59 (www.nia.nih.gov/health/publication/taking-care-your-teeth-and-mouth).
CONCLUSION
For most Americans with dental health insurance, access to services, and a willingness to practice good oral hygiene, dental care quality is above average. However, health care disparities and a lack of dental coverage prevent millions of Americans from routinely seeking dental services—making it essential for PCPs to promote oral hygiene and offer professional dental referrals with patients of all ages. Topical fluoride varnish, which can reduce the risk for decay as well as progression of existing decay, is an important preventive service that is within the scope of primary care practice.
1. Kaiser Commission. Medicaid and the uninsured: oral health in the US: key facts (2012). www.kff.org/uninsured/upload/8324.pdf. Accessed February 11, 2013.
2. Healthy People 2020. Oral health. www.healthypeople.gov/2020/topicsobjectives2020/overview.aspx?topicid=32. Accessed February 11, 2013.
3. CDC. Community water fluoridation: other fluoride products (2011). www.cdc.gov/fluoridation/other.htm. Accessed February 11, 2013.
4. American Academy of Pediatrics. Advocacy: Medicaid, fluoride varnish (2011). www.paaap.org/adv_medicaid_pg2.php. Accessed February 11, 2013.
5. National Institute of Dental and Craniofacial Research, NIH. The story of fluoridation (2011). www.nidcr.nih.gov/oralhealth/topics/fluoride/thestoryoffluoridation.htm. Accessed February 11, 2013.
6. Yu H, Attin T, Wiegand A, Buchalla W. Effects of various fluoride solutions on enamel erosion in vitro. Caries Res. 2010;44(4):390-401.
7. Bertness J, Holt K, eds; National Maternal & Child Oral Health Resource Center. Fluoride varnish: a resource guide (2010). www.mchoralhealth.org/PDFs/ResGuideFlVarnish.pdf. Accessed February 11, 2013.
8. Lee YE, Baek HJ, Choi YH, et al. Comparison of remineralization effect of three topical fluoride regimens on enamel initial carious lesions. J Dent. 2010;38(2):166-171.
9. New Zealand Guidelines Group. Guidelines for the use of fluorides (2009). Wellington, New Zealand: New Zealand Ministry of Health. www.guideline.gov/content.aspx?id=25685&search=fluoride+varnish. Accessed February 11, 2013.
10. O’Keefe E. Fluoride varnish may be effective in preschoolers. Evid Based Dent. 2011;12(2):41-42.
11. American Dental Association Council on Scientific Affairs. Professionally applied topical fluoride: evidence-based clinical recommendations. J Am Dent Assoc. 2006;137(8):1151-1159.
12. MassHealth. Health and Human Services. Fluoride varnish training for health-care professionals (2012). www.mass.gov/eohhs/gov/newsroom/masshealth/providers/fluoride-varnish-training-for-health-care.html. Accessed February 11, 2013.
13. Collins FM; Academy of Dental Therapeutics and Stomatology. The development and utilization of fluoride varnish (2011). www.ineedce.com/courses/2093/PDF/1106cei_varnish_web4.pdf. Accessed February 11, 2013.
14. New York State Department of Health. Improving the oral health of young children: fluoride varnish training materials and oral health information for child health care providers (2010). www.health.ny.gov/prevention/dental/child_oral_health_fluoride_varnish_for_hcp.htm. Accessed February 11, 2013.
15. Missouri Department of Health and Senior Services. Fluoride varnish application training for Missouri Oral Health Preventive Services Program. http://mohealthysmiles.typepad.com/Fluoride_Varnish_App_Training.pdf. Accessed February 15, 2013.
16. American Academy of Pediatrics. Oral health coding fact sheet for primary care physicians (2010). www2.aap.org/oralhealth/docs/OHCoding.pdf. Accessed February 11, 2013.
17. Chersoni S, Bertacci A, Pashley D, et al. In vivo effects of fluoride on enamel permeability. Clin Oral Investig. 2011;15(4):443-449.
18. National Interprofessional Initiative on Oral Health. Smiles for life curriculum (2011). www.niioh.org/smiles-life-curriculum. Accessed February 11, 2013.
19. Douglass AB, Gonsalves W, Maier R, et al. Smiles for Life: a national oral health curriculum for family medicine. Fam Med. 2007;39(2):88-90.
20. Medicaid.gov. Children’s Health Insurance Program (CHIP). www.medicaid .gov/Medicaid-CHIP-Program-Information/By-Topics/Childrens-Health-Insurance-Program-CHIP/Childrens-Health-Insurance-Program-CHIP.html. Accessed February 11, 2013.
21. Pourat N, Finocchio L. Racial and ethnic disparities in dental care for publicly insured children. Health Aff (Millwood). 2010;29(7):1356-1363.
22. Edelstein BL, Chinn CH. Update on disparities in oral health and access to dental care for America’s children. Acad Pediatr. 2009;9(6):415-419.
23. Seale NS, McWhorter AG, Moulradian WE. Dental education’s role in improving children’s oral health and access to care. Acad Pediatr. 2009;9(6):440-445.
24. National Institute of Dental and Craniofacial Research, NIH. Oral Health in America: A Report of the Surgeon General (Executive Summary; 2011). www.nidcr.nih.gov/datastatistics/surgeongeneral/report/executivesummary.htm. Accessed February 5, 2013.
25. Grembowski D, Spiekerman C, Milgrom P. Social gradients in dental health among low-income mothers and their young children. J Health Care Poor Underserved. 2012;23(2):570-588.
26. Institute of Medicine of the National Academies. Improving access to oral health care for vulnerable and underserved populations (2011). www.iom.edu/~/media/Files/Report Files/2011/Improving-Access-to-Oral-Health-Care-for-Vulnerable-and-Underserved-Populations/oralhealthaccess2011reportbrief.pdf. Accessed February 11, 2013.
27. Bertolami CN. Access to dental care: is there a problem? Am J Public Health. 2011;101(10):1817.
28. Bazargan N, Chi DL, Milgrom P. Exploring the potential for foreign-trained dentists to address workforce shortages and improve access to dental care for vulnerable populations in the United States: a case study from Washington State. BMC Health Serv Res. 2010;10:336-343.
29. Nash DA. Adding dental therapists to the health care team to improve access to oral health care for children. Acad Pediatr. 2009;9(6):446-451.
30. Liao CC, Ganz ML, Jiang H, Chelmow T. The impact of the public insurance expansions on children’s use of preventive dental care. Matern Child Health J. 2010;14(1):58-66.
31. Duley SI, Fitzpatrick PG, Zornosa X, Barnes WG. A center for oral health promotion: establishing an inter-professional paradigm for dental hygiene, health care management and nursing education. J Dent Hyg. 2012;86(2):63-70.
32. Ritter AV, Eidson RS, Donovan TE. Dental caries: etiology, clinical characteristics, risk assessment, and management. In: Heymann HO, Swift EJ Jr, Ritter AV, eds. Sturdevant’s Art and Science of Operative Dentistry. 6th ed. St. Louis: Elsevier-Mosby; 2012:41-89.
33. Shaefer HL, Miller M. Improving access to oral health care services among underserved populations in the US: is there a role for mid-level dental providers? J Health Care Poor Underserved. 2011;22(3):740-744.
34. Li KY, Wong MC, Lam KF, Schwarz E. Age, period, and cohort analysis of regular dental care behavior and edentulism: a marginal approach. BMC Oral Health. 2011;11:9.
35. Beauchamp J, Caufield PW, Crall JJ, et al. Evidence-based clinical recommendations for the use of pit-and-fissure sealants: a report of the American Dental Association Council on Scientific Affairs. J Am Dent Assoc. 2008;139(3):257-268.
36. National Institute of Dental and Craniofacial Research, NIH. Dental sealants in children (age 8 to 11)(2011). www.nidcr.nih.gov/DataStatistics/FindDataByTopic/DentalSealants/Children. Accessed February 11, 2013.
37. Gore DR. The use of dental sealants in adults: a long-neglected preventive measure. Int J Dent Hyg. 2010;8(3):198-203.
38. Devlin D, Henshaw M. Improving access to preventive dental services through a school-based dental sealant program. J Dent Hyg. 2011;85(3):211-219.
39. Mulic A, Tveit AB, Songe D, et al. Dental erosive wear and salivary flow rate in physically active young adults. BMC Oral Health. 2012;12:8.
40. Kaylor MB, Polivka BJ, Chaudry R, et al. Dental services utilization by women of child-bearing age by socioeconomic status. J Community Health. 2010;35(2):190-197.
41. Michalowicz BS, Hodges JS, DiAngelis AJ, et al; OPT Study. Treatment of periodontal disease and the risk of preterm birth. N Engl J Med. 2006;355(18):1885-1894.
42. Silk H, Douglass AB, Douglass JM, Silk L. Oral health during pregnancy. Am Fam Physician. 2008;77(8):1139-1144.
43. Clothier B, Stringer M, Jeffcoat MK. Periodontal disease and pregnancy outcomes: exposure, risk and intervention. Best Pract Res Clin Obstet Gynaecol. 2007;21(3):451-466.
44. Ressler-Maerlender J, Krishna R, Robison V. Oral health during pregnancy: current research. J Womens Health (Larchmt). 2005;14(10):880-882.
45. Berkowitz RJ. Acquisition and transmission of mutans streptococci. J Calif Dent Assoc. 2003;31(2):135-138.
46. Vollmer WM, Papas AS, Bader JD, et al; PACS Collaborative Research Group. Design of the Prevention of Adult Caries Study (PACS): a randomized clinical trial assessing the effect of chlorhexidine dental coating for the protection of adult caries. BMC Oral Health. 2010;10:23.
47. Dhull KS, Nandlal B. Effect of low-concentration daily fluoride application on fluoride release of giomer and compomer: an in vitro study. J Indian Soc Pedod Prev Dent. 2011;29(1):39-45.
48. Naumova EA, Kuehnl P, Hertenstein P, et al. Fluoride bioavailability in saliva and plaque. BMC Oral Health. 2012;12:3.
49. Duckworth RM, Maguire A, Omid N, et al. Effect of rinsing with mouthwash after brushing with fluoridated toothpaste on salivary fluoride concentration. Caries Res. 2009;43(5):391-396.
50. Opydo-Szymaczek J, Opydo J. Salivary fluoride concentrations and fluoride ingestion following application of preparations containing high concentration of fluoride. Biol Trace Elem Res. 2010;137(2):159-167.
51. Medicare.gov. Your Medicare coverage: dental services. www.medicare.gov/coverage/dental-services.html. Accessed February 11, 2013.
52. Quiñonez C, Ieraci L, Guttmann A. Potentially preventable hospital use for dental conditions: implications for expanding dental coverage for low income populations. J Health Care Poor Underserved. 2011;22(3):1048-1058.
53. Deshpande K, Jain A, Sharma RK, et al. Diabetes and periodontitis. J Indian Soc Periodontol. 2010;14(4):207-212.
54. Grubbs V, Plantinga LC, Tuot DS, Powe NR. Chronic kidney disease and use of dental services in a United States public healthcare system: a retrospective cohort study. BMC Nephrol. 2012;13:16.
55. Nair DR, Pruthy R, Pawar U, Chaturvedi P. Oral cancer: premalignant conditions and screening—an update. J Cancer Res Ther. 2012;8 suppl 1:S57-S66.
56. Wu B, Plassman BL, Lian J, Wei L. Cognitive function and dental care utilization among community-dwelling older adults. Am J Public Health. 2007;97(12):2216-2221.
57. Manski RJ, Moeller JF, St Clair PA, et al. The influence of changes in dental care coverage on dental care utilization among retirees and near-retirees in the United States, 2004-2006. Am J Public Health. 2011;101(10):1882-1891.
58. Moeller JF, Chen H, Manski RJ. Investing in preventive dental care for the Medicare population: a preliminary analysis. Am J Public Health. 2010;100(11):2262-2269.
59. National Institute on Aging, NIH. AgePage: Taking care of your teeth and mouth (2011). www.nia.nih.gov/health/publication/taking-care-your-teeth-and-mouth. Accessed February 6, 2013.
Millions of Americans currently lack access to dental care and oral health preventive services because of financial and/or geographic obstacles.1,2 Although community water fluoridation has benefited many Americans during the past 50 years, difficulties in obtaining dental care is a persistent public health challenge.2 Primary care providers (PCPs) can help improve health population-wide by educating patients in good dental hygiene and offering preventive oral health services to patients who would otherwise go without them.
The chief components of preventive dental care include daily brushing with fluoride toothpaste, flossing, routine dental check-ups and professional dental cleanings provided by a licensed dentist and staff, and use of dental sealants and topical fluoride. During the primary care appointment, a routine intraoral exam, including inspection of both hard and soft oral tissues for leukoplakia, gross decay, stained dental structures, and other pathologic changes, is essential. Primary care patients who use any form of tobacco should undergo inspection of the intraoral surfaces of the lips and assessment for adenopathy of the surrounding lymph nodes.
Counseling, including recommending routine preventive dental services (and, for some patients, allaying their fears of seeing a dentist2,3), and positive reinforcement of good oral hygiene practices should be addressed during every primary care encounter. Identifying patients without access to professional dental care is important, as the PCP may be their only source of oral health education, including referrals to local dental clinics.
Application of topical fluoride is an important means of improving dental health, and a cost-effective procedure for both patients and providers4 (see “Fluoride Use,”2,3,5-16). By following published guidelines for use of topical fluoride varnish, PCPs can reduce decay and mineralize enamel, thereby protecting the patient against enamel erosion.6,11,17 This service can and should be offered in primary care offices to patients who lack access to routine dental care and are likely to benefit from this effective weapon against tooth decay.
BEA, AGE 3
Bea lives with her mother and four older brothers and sisters. She is enrolled in Medicaid, but her mother has been unable to locate dental services for any of her children. With every call to a dental office within a 30-minute commute from their home, Bea’s mother has been told, “We don’t accept Medicaid.”
Under her state’s Medicaid plan, Bea has been seen for her primary health care needs since she was a newborn. She was bottle-fed from birth; her mother reports she has stopped taking the bottle to bed within the past few months. While conducting her three-year well-child exam, you note gross decay (see Figure 1).
Dental Services for Children
Nationwide, Medicaid is required to provide coverage for dental services for enrolled children, as well as those who qualify for the CHIP (Children’s Health Insurance Program).20 However, there is no mandate for privately owned dental practices to accept Medicaid payments.
Lack of dental care is the greatest unmet health care need in American children.21 In children without health insurance, access to dental care is most challenging, and those enrolled in Medicaid are the second least likely to receive services. As a result of these and other factors, one-third of US children ages 3 to 5 have dental caries in their primary teeth, and one in every four US children is estimated to have untreated decay2,21-23 (see Figure 2). African-American and Hispanic children experience greater disparities in dental care than do those of other ethnicities.21
Dental decay is the most common chronic illness of childhood (five times as common as asthma), with higher incidence in low-income and minority preschool children.1,24,25 Tooth decay affects growth patterns and nutrition, with associated pain and infection interfering with school attendance.
The small number of dentists who treat children and are willing to accept Medicaid reimbursement is a significant concern. In most dental programs, dedicated pedodontic training is optional. The mismatch of number of qualified dentists to the growing population of such patients is projected to worsen in the coming years.23
Potential solutions to correct this mismatch of dental services accessible to the underserved have included26-31:
- Addition of paraprofessional dental therapists (persons who have received two years’ training in a specific academic program) to the health care team, in public health clinics, schools, and other settings
- Interprofessional dental education (ie, cross-training physicians, NPs, PAs, and RNs in preventive dental services)
- An increasingly diverse array of competent providers who are trained, authorized, and compensated to provide evidence-based care
- Removing licensure restrictions for non–US-trained dentists
- Financial incentives for dentists to practice in dental Health Professional Shortage Areas
- Improved education of parents regarding the benefits of routine dental services.
The Institute of Medicine (IOM)26 has challenged state legislatures to mandate a one-year dental residency to be completed among underserved populations before dental licensure is granted. This challenge reflects the IOM’s acknowledgement that dental services, essential to patients’ well-being, are grossly lacking in the US.
Dental Care Education for Parents
Healthy baby teeth help children eat well and speak clearly. Decayed and abscessed teeth cause the same degree of pain and suffering in young children as they do in adults. Thus, regardless of ethnicity or socioeconomic status, all parents should be routinely encouraged to oversee daily dental hygiene practices for their children; it is important for PCPs to explain to parents their expectations in this regard.
Dental care for children should begin as soon as the first tooth erupts. New teeth should be cleaned daily with a soft cloth and inspected by parents for any discolorations. From age 2 years until children are able to clean all dental surfaces adequately (age 7 or 8), parents should brush their children’s teeth at least once daily with a toothbrush, after applying a thin film of fluoride toothpaste. Children should undergo their first professional dental check-up at about age 1 year.19,26
Parents must be made aware of other factors that can impair their children’s dental health. For example, the foods most likely to promote dental decay contain highly fermentable carbohydrates, are highly processed and sticky, and cause oral pH levels to fall below 5.5 (at which point demineralization may occur).32 Examples include presweetened cereals, dried fruits, cookies, and potato chips. Cheese, peanuts, meat, and eggs are less cariogenic.
Other important teaching points are “don’ts”: putting a baby to bed with a bottle that contains anything but a sugar-free liquid; using spill-proof spouted cups filled with juice or other sugary drinks all day; serving a large proportion of cariogenic foods.
BREANNE, AGE 22
Breanne attends a community college, where she is studying business. She reports to the college health clinic, complaining of an intermittent sharp pain in one of her lower molars over the past four days, occurring when she eats or drinks anything cold.
Before she turned 21, Breanne received routine dental services under her parents’ insurance. Now a full-time student no longer covered by her parents’ plan, she has no income with which to purchase dental coverage. Years ago, Breanne received dental sealants on all of her molars in a school-based program, and she routinely brushes daily (see Figure 3). She says she “usually remembers to floss.”
Dental Concerns in Young Adults
Up to one-third of Americans, including college-age students, lack access to oral health services.33 Use of annual dental services, including school-based care, significantly reduces young adults’ risk for tooth loss.34 From early adulthood on, poor dental health is associated with cardiovascular and respiratory disease, cerebral ischemia, diabetes, difficulty with chewing and oral pain, and poor self-image.24
PCPs should emphasize the importance of preventive services as they examine the oral structure during routine health care encounters. Adding the question, “When did you last have a dental exam?” can launch an effective discussion about the importance of good dental health.
Tooth surfaces with pits and fissures are the most vulnerable to decay. Dental sealants can protect these vulnerable areas, reducing decay by 60% to 82%.2,32,35 Sealants are the most effective preventive measure for reducing cavity formation; they are not harmful to fillings and can actually reduce the progression of decay when applied over small existing carious lesions.35 Currently, however, only 30% and 38% of children and adolescents, respectively, have undergone application of sealants.36 Thus, it is vital for PCPs to recommend dental sealants to all their patients.35,37,38
Young adults should also be questioned about their exercise patterns. While daily exercise is of benefit to good health, young adults who engage in too-frequent, overly vigorous exercise have been found vulnerable to dental erosion.39 This irreversible condition can result from the rapid breathing, sweat-induced dehydration, and decreased salivary flow associated with strenuous exercise. Reduced salivary flow disrupts the protection against erosive acids afforded by bathing of teeth with saliva, leading to a fivefold greater risk for developing erosions. Patients should be encouraged to hydrate before exercise to reduce these risks.39
Dental Health Notes for Young Women
For female patients of childbearing age like Breanne, dental health is important not only for the patient, but for her future children as well. Poor maternal dental health has been associated with preterm births and low birth weights.40-43 Women who have health insurance, who are in good general health, and who regularly visit a primary care medical provider are also the most likely to see a dental care provider during pregnancy.40
Pregnancy is associated with a risk for poor dental care in any population.42,44 During pregnancy, Medicaid-enrolled women are 30% less likely to see a dentist than women with private health insurance, and women with no health insurance are 70% less likely to see a dentist.44 Among pregnant women who experience a dental problem, less than half will schedule a dental appointment. Postponing dental care till after delivery incurs additional risk, as the new mother will be busy with her child and may have lost her health care coverage in the interim.
Dental concerns commonly encountered during pregnancy are listed in Table 3.41,42 There is also evidence that maternal transmission of mutans streptococci can lead to early childhood caries,42,45 further highlighting the importance of dental care during pregnancy (eg, use of chlorhexidine rinses in pregnant women with dental caries42). The association between periodontal disease and preterm delivery and lower birth weights40-43 makes it critical to reduce maternal periodontal inflammation through timely dental referrals.
SAUL, AGE 42
Saul, a lifelong resident of Mississippi, has worked in construction since age 18. He has never had health or dental insurance and thus he rarely seeks preventive services. As he recalls, he has had “two fillings in my jaw teeth since I was a kid,” but otherwise has had no dental care. He brushes daily to avoid having to see a dentist. He is presenting today as a new patient only because he has a rash.
Benefits of Fluoride Use During Middle Age
Dental care needs peak between ages 55 and 64, with the majority of care prompted by dental decay46 (see Figure 4). A discussion about dental health should be raised when the PCP completes the review of systems or while discussing other preventive services. The option of fluoride varnish should not be overlooked in a patient of any age, particularly one with a recognizable deficiency of preventive dental services. Explaining the benefits of topical fluoride varnish (see “Fluoride Use”) and offering an opportunity to receive it in the office may persuade a patient like Saul to take advantage of this proven anticarious procedure.
Patients who undergo this safe procedure should be reminded to continue brushing daily with fluoridated toothpaste and drinking fluoridated water (if it is available).50
GILL, AGE 77
Gill grew up in Grant County, New Mexico, in an area not far from Silver City; there, fluoride supplementation in the local water did not begin until Gill was about 40. His family never stressed oral hygiene, and his remaining teeth show gross plaque and decay.
He sees you routinely for his chronic illnesses, and today he has presented for his annual physical. Gill has Medicare without supplemental insurance.
Dental Care in the Medicare Years
Medicare does not cover preventive dental services unless hospitalization is required; in that case, it is covered under Medicare Part A.51 Publicly supported dental services are primarily offered for children, leaving Medicare recipients responsible for the costs of dental care. As a last resort, some patients will seek dental care at a hospital emergency department (ED). Most often prompted to visit the ED for relief of pain triggered by dental fractures or abscesses, patients usually leave with pain medications and antibiotics only; their general dental health needs remain unmet.52
The most common dental concerns among elderly patients include edentulism (toothlessness, which is reported in one in four Medicare beneficiaries1), xerostomia (dry mouth), periodontal disease, ill-fitting or worn prosthetics, and progressive decay affecting fractured teeth1 (see Figure 5). As many elderly patients endure a combination of these uncomfortable, possibly painful, conditions, it is important for the PCP to inquire about them during the physical exam and the review of symptoms. At each step, a window of opportunity opens to make appropriate recommendations.
Significant Comorbidities
It is important for PCPs to recognize the association between certain diseases and poor oral health. With the progression of age, the risk increases for type 2 diabetes. Among older diabetic patients with periodontitis (and the systemic inflammation associated with it), the difficulty of maintaining glycemic control increases as well.53 Conversely, diabetic patients who are immunocompromised are at elevated risk for periodontitis. In more than 20 years of formal data collection, periodontists have identified and confirmed an anecdotal relationship between diabetes and severe periodontal disease.26
Elderly patients with poor dental health are also at increased risk for chronic kidney disease. Periodontal disease alone increases the risk for nephropathy 1.5-fold to twofold.54Finally, patients’ risk for oral cancer increases with age. Known risk factors for oral cancers include smoking, use of chewing tobacco, alcohol consumption, chronic friction, and exposure to ultraviolet radiation.55 Survival rates for oral cancers are poor, reemphasizing the need for a thorough oral exam during primary care visits.
Other Challenges for Older Patients
Members of ethnic minority groups and lower-income populations, men, patients with cognitive impairment, and persons who never completed high school are the most likely to avoid, discontinue, or lack access to dental services.56 Loss of dental coverage on retirement is associated with a significant decline in elderly patients’ use of dental services. Though now living on a fraction of their preretirement income, millions of retirees are required to pay out-of-pocket for dental services.57
Topical fluoride varnish alone cannot alleviate this multimillion dollar concern, but using it to reinforce retirees’ remaining enamel and prevent caries can help reduce their dental care expenditures. However, until federal and state policymakers act to cover preventive services,2,58 PCPs have little to offer elderly patients beyond oral hygiene education and in-office application of topical fluoride.
Strategies to Reinforce Dental Health
In practices where electronic medical records are in use, a recommendation for dental services should be printed as a patient reminder. Printed recommendations and referrals from PCPs do improve patient compliance.59 In addition to the printed office summary, older patients can be handed a summary of oral health suggestions from the National Institute on Aging, NIH59 (www.nia.nih.gov/health/publication/taking-care-your-teeth-and-mouth).
CONCLUSION
For most Americans with dental health insurance, access to services, and a willingness to practice good oral hygiene, dental care quality is above average. However, health care disparities and a lack of dental coverage prevent millions of Americans from routinely seeking dental services—making it essential for PCPs to promote oral hygiene and offer professional dental referrals with patients of all ages. Topical fluoride varnish, which can reduce the risk for decay as well as progression of existing decay, is an important preventive service that is within the scope of primary care practice.
Millions of Americans currently lack access to dental care and oral health preventive services because of financial and/or geographic obstacles.1,2 Although community water fluoridation has benefited many Americans during the past 50 years, difficulties in obtaining dental care is a persistent public health challenge.2 Primary care providers (PCPs) can help improve health population-wide by educating patients in good dental hygiene and offering preventive oral health services to patients who would otherwise go without them.
The chief components of preventive dental care include daily brushing with fluoride toothpaste, flossing, routine dental check-ups and professional dental cleanings provided by a licensed dentist and staff, and use of dental sealants and topical fluoride. During the primary care appointment, a routine intraoral exam, including inspection of both hard and soft oral tissues for leukoplakia, gross decay, stained dental structures, and other pathologic changes, is essential. Primary care patients who use any form of tobacco should undergo inspection of the intraoral surfaces of the lips and assessment for adenopathy of the surrounding lymph nodes.
Counseling, including recommending routine preventive dental services (and, for some patients, allaying their fears of seeing a dentist2,3), and positive reinforcement of good oral hygiene practices should be addressed during every primary care encounter. Identifying patients without access to professional dental care is important, as the PCP may be their only source of oral health education, including referrals to local dental clinics.
Application of topical fluoride is an important means of improving dental health, and a cost-effective procedure for both patients and providers4 (see “Fluoride Use,”2,3,5-16). By following published guidelines for use of topical fluoride varnish, PCPs can reduce decay and mineralize enamel, thereby protecting the patient against enamel erosion.6,11,17 This service can and should be offered in primary care offices to patients who lack access to routine dental care and are likely to benefit from this effective weapon against tooth decay.
BEA, AGE 3
Bea lives with her mother and four older brothers and sisters. She is enrolled in Medicaid, but her mother has been unable to locate dental services for any of her children. With every call to a dental office within a 30-minute commute from their home, Bea’s mother has been told, “We don’t accept Medicaid.”
Under her state’s Medicaid plan, Bea has been seen for her primary health care needs since she was a newborn. She was bottle-fed from birth; her mother reports she has stopped taking the bottle to bed within the past few months. While conducting her three-year well-child exam, you note gross decay (see Figure 1).
Dental Services for Children
Nationwide, Medicaid is required to provide coverage for dental services for enrolled children, as well as those who qualify for the CHIP (Children’s Health Insurance Program).20 However, there is no mandate for privately owned dental practices to accept Medicaid payments.
Lack of dental care is the greatest unmet health care need in American children.21 In children without health insurance, access to dental care is most challenging, and those enrolled in Medicaid are the second least likely to receive services. As a result of these and other factors, one-third of US children ages 3 to 5 have dental caries in their primary teeth, and one in every four US children is estimated to have untreated decay2,21-23 (see Figure 2). African-American and Hispanic children experience greater disparities in dental care than do those of other ethnicities.21
Dental decay is the most common chronic illness of childhood (five times as common as asthma), with higher incidence in low-income and minority preschool children.1,24,25 Tooth decay affects growth patterns and nutrition, with associated pain and infection interfering with school attendance.
The small number of dentists who treat children and are willing to accept Medicaid reimbursement is a significant concern. In most dental programs, dedicated pedodontic training is optional. The mismatch of number of qualified dentists to the growing population of such patients is projected to worsen in the coming years.23
Potential solutions to correct this mismatch of dental services accessible to the underserved have included26-31:
- Addition of paraprofessional dental therapists (persons who have received two years’ training in a specific academic program) to the health care team, in public health clinics, schools, and other settings
- Interprofessional dental education (ie, cross-training physicians, NPs, PAs, and RNs in preventive dental services)
- An increasingly diverse array of competent providers who are trained, authorized, and compensated to provide evidence-based care
- Removing licensure restrictions for non–US-trained dentists
- Financial incentives for dentists to practice in dental Health Professional Shortage Areas
- Improved education of parents regarding the benefits of routine dental services.
The Institute of Medicine (IOM)26 has challenged state legislatures to mandate a one-year dental residency to be completed among underserved populations before dental licensure is granted. This challenge reflects the IOM’s acknowledgement that dental services, essential to patients’ well-being, are grossly lacking in the US.
Dental Care Education for Parents
Healthy baby teeth help children eat well and speak clearly. Decayed and abscessed teeth cause the same degree of pain and suffering in young children as they do in adults. Thus, regardless of ethnicity or socioeconomic status, all parents should be routinely encouraged to oversee daily dental hygiene practices for their children; it is important for PCPs to explain to parents their expectations in this regard.
Dental care for children should begin as soon as the first tooth erupts. New teeth should be cleaned daily with a soft cloth and inspected by parents for any discolorations. From age 2 years until children are able to clean all dental surfaces adequately (age 7 or 8), parents should brush their children’s teeth at least once daily with a toothbrush, after applying a thin film of fluoride toothpaste. Children should undergo their first professional dental check-up at about age 1 year.19,26
Parents must be made aware of other factors that can impair their children’s dental health. For example, the foods most likely to promote dental decay contain highly fermentable carbohydrates, are highly processed and sticky, and cause oral pH levels to fall below 5.5 (at which point demineralization may occur).32 Examples include presweetened cereals, dried fruits, cookies, and potato chips. Cheese, peanuts, meat, and eggs are less cariogenic.
Other important teaching points are “don’ts”: putting a baby to bed with a bottle that contains anything but a sugar-free liquid; using spill-proof spouted cups filled with juice or other sugary drinks all day; serving a large proportion of cariogenic foods.
BREANNE, AGE 22
Breanne attends a community college, where she is studying business. She reports to the college health clinic, complaining of an intermittent sharp pain in one of her lower molars over the past four days, occurring when she eats or drinks anything cold.
Before she turned 21, Breanne received routine dental services under her parents’ insurance. Now a full-time student no longer covered by her parents’ plan, she has no income with which to purchase dental coverage. Years ago, Breanne received dental sealants on all of her molars in a school-based program, and she routinely brushes daily (see Figure 3). She says she “usually remembers to floss.”
Dental Concerns in Young Adults
Up to one-third of Americans, including college-age students, lack access to oral health services.33 Use of annual dental services, including school-based care, significantly reduces young adults’ risk for tooth loss.34 From early adulthood on, poor dental health is associated with cardiovascular and respiratory disease, cerebral ischemia, diabetes, difficulty with chewing and oral pain, and poor self-image.24
PCPs should emphasize the importance of preventive services as they examine the oral structure during routine health care encounters. Adding the question, “When did you last have a dental exam?” can launch an effective discussion about the importance of good dental health.
Tooth surfaces with pits and fissures are the most vulnerable to decay. Dental sealants can protect these vulnerable areas, reducing decay by 60% to 82%.2,32,35 Sealants are the most effective preventive measure for reducing cavity formation; they are not harmful to fillings and can actually reduce the progression of decay when applied over small existing carious lesions.35 Currently, however, only 30% and 38% of children and adolescents, respectively, have undergone application of sealants.36 Thus, it is vital for PCPs to recommend dental sealants to all their patients.35,37,38
Young adults should also be questioned about their exercise patterns. While daily exercise is of benefit to good health, young adults who engage in too-frequent, overly vigorous exercise have been found vulnerable to dental erosion.39 This irreversible condition can result from the rapid breathing, sweat-induced dehydration, and decreased salivary flow associated with strenuous exercise. Reduced salivary flow disrupts the protection against erosive acids afforded by bathing of teeth with saliva, leading to a fivefold greater risk for developing erosions. Patients should be encouraged to hydrate before exercise to reduce these risks.39
Dental Health Notes for Young Women
For female patients of childbearing age like Breanne, dental health is important not only for the patient, but for her future children as well. Poor maternal dental health has been associated with preterm births and low birth weights.40-43 Women who have health insurance, who are in good general health, and who regularly visit a primary care medical provider are also the most likely to see a dental care provider during pregnancy.40
Pregnancy is associated with a risk for poor dental care in any population.42,44 During pregnancy, Medicaid-enrolled women are 30% less likely to see a dentist than women with private health insurance, and women with no health insurance are 70% less likely to see a dentist.44 Among pregnant women who experience a dental problem, less than half will schedule a dental appointment. Postponing dental care till after delivery incurs additional risk, as the new mother will be busy with her child and may have lost her health care coverage in the interim.
Dental concerns commonly encountered during pregnancy are listed in Table 3.41,42 There is also evidence that maternal transmission of mutans streptococci can lead to early childhood caries,42,45 further highlighting the importance of dental care during pregnancy (eg, use of chlorhexidine rinses in pregnant women with dental caries42). The association between periodontal disease and preterm delivery and lower birth weights40-43 makes it critical to reduce maternal periodontal inflammation through timely dental referrals.
SAUL, AGE 42
Saul, a lifelong resident of Mississippi, has worked in construction since age 18. He has never had health or dental insurance and thus he rarely seeks preventive services. As he recalls, he has had “two fillings in my jaw teeth since I was a kid,” but otherwise has had no dental care. He brushes daily to avoid having to see a dentist. He is presenting today as a new patient only because he has a rash.
Benefits of Fluoride Use During Middle Age
Dental care needs peak between ages 55 and 64, with the majority of care prompted by dental decay46 (see Figure 4). A discussion about dental health should be raised when the PCP completes the review of systems or while discussing other preventive services. The option of fluoride varnish should not be overlooked in a patient of any age, particularly one with a recognizable deficiency of preventive dental services. Explaining the benefits of topical fluoride varnish (see “Fluoride Use”) and offering an opportunity to receive it in the office may persuade a patient like Saul to take advantage of this proven anticarious procedure.
Patients who undergo this safe procedure should be reminded to continue brushing daily with fluoridated toothpaste and drinking fluoridated water (if it is available).50
GILL, AGE 77
Gill grew up in Grant County, New Mexico, in an area not far from Silver City; there, fluoride supplementation in the local water did not begin until Gill was about 40. His family never stressed oral hygiene, and his remaining teeth show gross plaque and decay.
He sees you routinely for his chronic illnesses, and today he has presented for his annual physical. Gill has Medicare without supplemental insurance.
Dental Care in the Medicare Years
Medicare does not cover preventive dental services unless hospitalization is required; in that case, it is covered under Medicare Part A.51 Publicly supported dental services are primarily offered for children, leaving Medicare recipients responsible for the costs of dental care. As a last resort, some patients will seek dental care at a hospital emergency department (ED). Most often prompted to visit the ED for relief of pain triggered by dental fractures or abscesses, patients usually leave with pain medications and antibiotics only; their general dental health needs remain unmet.52
The most common dental concerns among elderly patients include edentulism (toothlessness, which is reported in one in four Medicare beneficiaries1), xerostomia (dry mouth), periodontal disease, ill-fitting or worn prosthetics, and progressive decay affecting fractured teeth1 (see Figure 5). As many elderly patients endure a combination of these uncomfortable, possibly painful, conditions, it is important for the PCP to inquire about them during the physical exam and the review of symptoms. At each step, a window of opportunity opens to make appropriate recommendations.
Significant Comorbidities
It is important for PCPs to recognize the association between certain diseases and poor oral health. With the progression of age, the risk increases for type 2 diabetes. Among older diabetic patients with periodontitis (and the systemic inflammation associated with it), the difficulty of maintaining glycemic control increases as well.53 Conversely, diabetic patients who are immunocompromised are at elevated risk for periodontitis. In more than 20 years of formal data collection, periodontists have identified and confirmed an anecdotal relationship between diabetes and severe periodontal disease.26
Elderly patients with poor dental health are also at increased risk for chronic kidney disease. Periodontal disease alone increases the risk for nephropathy 1.5-fold to twofold.54Finally, patients’ risk for oral cancer increases with age. Known risk factors for oral cancers include smoking, use of chewing tobacco, alcohol consumption, chronic friction, and exposure to ultraviolet radiation.55 Survival rates for oral cancers are poor, reemphasizing the need for a thorough oral exam during primary care visits.
Other Challenges for Older Patients
Members of ethnic minority groups and lower-income populations, men, patients with cognitive impairment, and persons who never completed high school are the most likely to avoid, discontinue, or lack access to dental services.56 Loss of dental coverage on retirement is associated with a significant decline in elderly patients’ use of dental services. Though now living on a fraction of their preretirement income, millions of retirees are required to pay out-of-pocket for dental services.57
Topical fluoride varnish alone cannot alleviate this multimillion dollar concern, but using it to reinforce retirees’ remaining enamel and prevent caries can help reduce their dental care expenditures. However, until federal and state policymakers act to cover preventive services,2,58 PCPs have little to offer elderly patients beyond oral hygiene education and in-office application of topical fluoride.
Strategies to Reinforce Dental Health
In practices where electronic medical records are in use, a recommendation for dental services should be printed as a patient reminder. Printed recommendations and referrals from PCPs do improve patient compliance.59 In addition to the printed office summary, older patients can be handed a summary of oral health suggestions from the National Institute on Aging, NIH59 (www.nia.nih.gov/health/publication/taking-care-your-teeth-and-mouth).
CONCLUSION
For most Americans with dental health insurance, access to services, and a willingness to practice good oral hygiene, dental care quality is above average. However, health care disparities and a lack of dental coverage prevent millions of Americans from routinely seeking dental services—making it essential for PCPs to promote oral hygiene and offer professional dental referrals with patients of all ages. Topical fluoride varnish, which can reduce the risk for decay as well as progression of existing decay, is an important preventive service that is within the scope of primary care practice.
1. Kaiser Commission. Medicaid and the uninsured: oral health in the US: key facts (2012). www.kff.org/uninsured/upload/8324.pdf. Accessed February 11, 2013.
2. Healthy People 2020. Oral health. www.healthypeople.gov/2020/topicsobjectives2020/overview.aspx?topicid=32. Accessed February 11, 2013.
3. CDC. Community water fluoridation: other fluoride products (2011). www.cdc.gov/fluoridation/other.htm. Accessed February 11, 2013.
4. American Academy of Pediatrics. Advocacy: Medicaid, fluoride varnish (2011). www.paaap.org/adv_medicaid_pg2.php. Accessed February 11, 2013.
5. National Institute of Dental and Craniofacial Research, NIH. The story of fluoridation (2011). www.nidcr.nih.gov/oralhealth/topics/fluoride/thestoryoffluoridation.htm. Accessed February 11, 2013.
6. Yu H, Attin T, Wiegand A, Buchalla W. Effects of various fluoride solutions on enamel erosion in vitro. Caries Res. 2010;44(4):390-401.
7. Bertness J, Holt K, eds; National Maternal & Child Oral Health Resource Center. Fluoride varnish: a resource guide (2010). www.mchoralhealth.org/PDFs/ResGuideFlVarnish.pdf. Accessed February 11, 2013.
8. Lee YE, Baek HJ, Choi YH, et al. Comparison of remineralization effect of three topical fluoride regimens on enamel initial carious lesions. J Dent. 2010;38(2):166-171.
9. New Zealand Guidelines Group. Guidelines for the use of fluorides (2009). Wellington, New Zealand: New Zealand Ministry of Health. www.guideline.gov/content.aspx?id=25685&search=fluoride+varnish. Accessed February 11, 2013.
10. O’Keefe E. Fluoride varnish may be effective in preschoolers. Evid Based Dent. 2011;12(2):41-42.
11. American Dental Association Council on Scientific Affairs. Professionally applied topical fluoride: evidence-based clinical recommendations. J Am Dent Assoc. 2006;137(8):1151-1159.
12. MassHealth. Health and Human Services. Fluoride varnish training for health-care professionals (2012). www.mass.gov/eohhs/gov/newsroom/masshealth/providers/fluoride-varnish-training-for-health-care.html. Accessed February 11, 2013.
13. Collins FM; Academy of Dental Therapeutics and Stomatology. The development and utilization of fluoride varnish (2011). www.ineedce.com/courses/2093/PDF/1106cei_varnish_web4.pdf. Accessed February 11, 2013.
14. New York State Department of Health. Improving the oral health of young children: fluoride varnish training materials and oral health information for child health care providers (2010). www.health.ny.gov/prevention/dental/child_oral_health_fluoride_varnish_for_hcp.htm. Accessed February 11, 2013.
15. Missouri Department of Health and Senior Services. Fluoride varnish application training for Missouri Oral Health Preventive Services Program. http://mohealthysmiles.typepad.com/Fluoride_Varnish_App_Training.pdf. Accessed February 15, 2013.
16. American Academy of Pediatrics. Oral health coding fact sheet for primary care physicians (2010). www2.aap.org/oralhealth/docs/OHCoding.pdf. Accessed February 11, 2013.
17. Chersoni S, Bertacci A, Pashley D, et al. In vivo effects of fluoride on enamel permeability. Clin Oral Investig. 2011;15(4):443-449.
18. National Interprofessional Initiative on Oral Health. Smiles for life curriculum (2011). www.niioh.org/smiles-life-curriculum. Accessed February 11, 2013.
19. Douglass AB, Gonsalves W, Maier R, et al. Smiles for Life: a national oral health curriculum for family medicine. Fam Med. 2007;39(2):88-90.
20. Medicaid.gov. Children’s Health Insurance Program (CHIP). www.medicaid .gov/Medicaid-CHIP-Program-Information/By-Topics/Childrens-Health-Insurance-Program-CHIP/Childrens-Health-Insurance-Program-CHIP.html. Accessed February 11, 2013.
21. Pourat N, Finocchio L. Racial and ethnic disparities in dental care for publicly insured children. Health Aff (Millwood). 2010;29(7):1356-1363.
22. Edelstein BL, Chinn CH. Update on disparities in oral health and access to dental care for America’s children. Acad Pediatr. 2009;9(6):415-419.
23. Seale NS, McWhorter AG, Moulradian WE. Dental education’s role in improving children’s oral health and access to care. Acad Pediatr. 2009;9(6):440-445.
24. National Institute of Dental and Craniofacial Research, NIH. Oral Health in America: A Report of the Surgeon General (Executive Summary; 2011). www.nidcr.nih.gov/datastatistics/surgeongeneral/report/executivesummary.htm. Accessed February 5, 2013.
25. Grembowski D, Spiekerman C, Milgrom P. Social gradients in dental health among low-income mothers and their young children. J Health Care Poor Underserved. 2012;23(2):570-588.
26. Institute of Medicine of the National Academies. Improving access to oral health care for vulnerable and underserved populations (2011). www.iom.edu/~/media/Files/Report Files/2011/Improving-Access-to-Oral-Health-Care-for-Vulnerable-and-Underserved-Populations/oralhealthaccess2011reportbrief.pdf. Accessed February 11, 2013.
27. Bertolami CN. Access to dental care: is there a problem? Am J Public Health. 2011;101(10):1817.
28. Bazargan N, Chi DL, Milgrom P. Exploring the potential for foreign-trained dentists to address workforce shortages and improve access to dental care for vulnerable populations in the United States: a case study from Washington State. BMC Health Serv Res. 2010;10:336-343.
29. Nash DA. Adding dental therapists to the health care team to improve access to oral health care for children. Acad Pediatr. 2009;9(6):446-451.
30. Liao CC, Ganz ML, Jiang H, Chelmow T. The impact of the public insurance expansions on children’s use of preventive dental care. Matern Child Health J. 2010;14(1):58-66.
31. Duley SI, Fitzpatrick PG, Zornosa X, Barnes WG. A center for oral health promotion: establishing an inter-professional paradigm for dental hygiene, health care management and nursing education. J Dent Hyg. 2012;86(2):63-70.
32. Ritter AV, Eidson RS, Donovan TE. Dental caries: etiology, clinical characteristics, risk assessment, and management. In: Heymann HO, Swift EJ Jr, Ritter AV, eds. Sturdevant’s Art and Science of Operative Dentistry. 6th ed. St. Louis: Elsevier-Mosby; 2012:41-89.
33. Shaefer HL, Miller M. Improving access to oral health care services among underserved populations in the US: is there a role for mid-level dental providers? J Health Care Poor Underserved. 2011;22(3):740-744.
34. Li KY, Wong MC, Lam KF, Schwarz E. Age, period, and cohort analysis of regular dental care behavior and edentulism: a marginal approach. BMC Oral Health. 2011;11:9.
35. Beauchamp J, Caufield PW, Crall JJ, et al. Evidence-based clinical recommendations for the use of pit-and-fissure sealants: a report of the American Dental Association Council on Scientific Affairs. J Am Dent Assoc. 2008;139(3):257-268.
36. National Institute of Dental and Craniofacial Research, NIH. Dental sealants in children (age 8 to 11)(2011). www.nidcr.nih.gov/DataStatistics/FindDataByTopic/DentalSealants/Children. Accessed February 11, 2013.
37. Gore DR. The use of dental sealants in adults: a long-neglected preventive measure. Int J Dent Hyg. 2010;8(3):198-203.
38. Devlin D, Henshaw M. Improving access to preventive dental services through a school-based dental sealant program. J Dent Hyg. 2011;85(3):211-219.
39. Mulic A, Tveit AB, Songe D, et al. Dental erosive wear and salivary flow rate in physically active young adults. BMC Oral Health. 2012;12:8.
40. Kaylor MB, Polivka BJ, Chaudry R, et al. Dental services utilization by women of child-bearing age by socioeconomic status. J Community Health. 2010;35(2):190-197.
41. Michalowicz BS, Hodges JS, DiAngelis AJ, et al; OPT Study. Treatment of periodontal disease and the risk of preterm birth. N Engl J Med. 2006;355(18):1885-1894.
42. Silk H, Douglass AB, Douglass JM, Silk L. Oral health during pregnancy. Am Fam Physician. 2008;77(8):1139-1144.
43. Clothier B, Stringer M, Jeffcoat MK. Periodontal disease and pregnancy outcomes: exposure, risk and intervention. Best Pract Res Clin Obstet Gynaecol. 2007;21(3):451-466.
44. Ressler-Maerlender J, Krishna R, Robison V. Oral health during pregnancy: current research. J Womens Health (Larchmt). 2005;14(10):880-882.
45. Berkowitz RJ. Acquisition and transmission of mutans streptococci. J Calif Dent Assoc. 2003;31(2):135-138.
46. Vollmer WM, Papas AS, Bader JD, et al; PACS Collaborative Research Group. Design of the Prevention of Adult Caries Study (PACS): a randomized clinical trial assessing the effect of chlorhexidine dental coating for the protection of adult caries. BMC Oral Health. 2010;10:23.
47. Dhull KS, Nandlal B. Effect of low-concentration daily fluoride application on fluoride release of giomer and compomer: an in vitro study. J Indian Soc Pedod Prev Dent. 2011;29(1):39-45.
48. Naumova EA, Kuehnl P, Hertenstein P, et al. Fluoride bioavailability in saliva and plaque. BMC Oral Health. 2012;12:3.
49. Duckworth RM, Maguire A, Omid N, et al. Effect of rinsing with mouthwash after brushing with fluoridated toothpaste on salivary fluoride concentration. Caries Res. 2009;43(5):391-396.
50. Opydo-Szymaczek J, Opydo J. Salivary fluoride concentrations and fluoride ingestion following application of preparations containing high concentration of fluoride. Biol Trace Elem Res. 2010;137(2):159-167.
51. Medicare.gov. Your Medicare coverage: dental services. www.medicare.gov/coverage/dental-services.html. Accessed February 11, 2013.
52. Quiñonez C, Ieraci L, Guttmann A. Potentially preventable hospital use for dental conditions: implications for expanding dental coverage for low income populations. J Health Care Poor Underserved. 2011;22(3):1048-1058.
53. Deshpande K, Jain A, Sharma RK, et al. Diabetes and periodontitis. J Indian Soc Periodontol. 2010;14(4):207-212.
54. Grubbs V, Plantinga LC, Tuot DS, Powe NR. Chronic kidney disease and use of dental services in a United States public healthcare system: a retrospective cohort study. BMC Nephrol. 2012;13:16.
55. Nair DR, Pruthy R, Pawar U, Chaturvedi P. Oral cancer: premalignant conditions and screening—an update. J Cancer Res Ther. 2012;8 suppl 1:S57-S66.
56. Wu B, Plassman BL, Lian J, Wei L. Cognitive function and dental care utilization among community-dwelling older adults. Am J Public Health. 2007;97(12):2216-2221.
57. Manski RJ, Moeller JF, St Clair PA, et al. The influence of changes in dental care coverage on dental care utilization among retirees and near-retirees in the United States, 2004-2006. Am J Public Health. 2011;101(10):1882-1891.
58. Moeller JF, Chen H, Manski RJ. Investing in preventive dental care for the Medicare population: a preliminary analysis. Am J Public Health. 2010;100(11):2262-2269.
59. National Institute on Aging, NIH. AgePage: Taking care of your teeth and mouth (2011). www.nia.nih.gov/health/publication/taking-care-your-teeth-and-mouth. Accessed February 6, 2013.
1. Kaiser Commission. Medicaid and the uninsured: oral health in the US: key facts (2012). www.kff.org/uninsured/upload/8324.pdf. Accessed February 11, 2013.
2. Healthy People 2020. Oral health. www.healthypeople.gov/2020/topicsobjectives2020/overview.aspx?topicid=32. Accessed February 11, 2013.
3. CDC. Community water fluoridation: other fluoride products (2011). www.cdc.gov/fluoridation/other.htm. Accessed February 11, 2013.
4. American Academy of Pediatrics. Advocacy: Medicaid, fluoride varnish (2011). www.paaap.org/adv_medicaid_pg2.php. Accessed February 11, 2013.
5. National Institute of Dental and Craniofacial Research, NIH. The story of fluoridation (2011). www.nidcr.nih.gov/oralhealth/topics/fluoride/thestoryoffluoridation.htm. Accessed February 11, 2013.
6. Yu H, Attin T, Wiegand A, Buchalla W. Effects of various fluoride solutions on enamel erosion in vitro. Caries Res. 2010;44(4):390-401.
7. Bertness J, Holt K, eds; National Maternal & Child Oral Health Resource Center. Fluoride varnish: a resource guide (2010). www.mchoralhealth.org/PDFs/ResGuideFlVarnish.pdf. Accessed February 11, 2013.
8. Lee YE, Baek HJ, Choi YH, et al. Comparison of remineralization effect of three topical fluoride regimens on enamel initial carious lesions. J Dent. 2010;38(2):166-171.
9. New Zealand Guidelines Group. Guidelines for the use of fluorides (2009). Wellington, New Zealand: New Zealand Ministry of Health. www.guideline.gov/content.aspx?id=25685&search=fluoride+varnish. Accessed February 11, 2013.
10. O’Keefe E. Fluoride varnish may be effective in preschoolers. Evid Based Dent. 2011;12(2):41-42.
11. American Dental Association Council on Scientific Affairs. Professionally applied topical fluoride: evidence-based clinical recommendations. J Am Dent Assoc. 2006;137(8):1151-1159.
12. MassHealth. Health and Human Services. Fluoride varnish training for health-care professionals (2012). www.mass.gov/eohhs/gov/newsroom/masshealth/providers/fluoride-varnish-training-for-health-care.html. Accessed February 11, 2013.
13. Collins FM; Academy of Dental Therapeutics and Stomatology. The development and utilization of fluoride varnish (2011). www.ineedce.com/courses/2093/PDF/1106cei_varnish_web4.pdf. Accessed February 11, 2013.
14. New York State Department of Health. Improving the oral health of young children: fluoride varnish training materials and oral health information for child health care providers (2010). www.health.ny.gov/prevention/dental/child_oral_health_fluoride_varnish_for_hcp.htm. Accessed February 11, 2013.
15. Missouri Department of Health and Senior Services. Fluoride varnish application training for Missouri Oral Health Preventive Services Program. http://mohealthysmiles.typepad.com/Fluoride_Varnish_App_Training.pdf. Accessed February 15, 2013.
16. American Academy of Pediatrics. Oral health coding fact sheet for primary care physicians (2010). www2.aap.org/oralhealth/docs/OHCoding.pdf. Accessed February 11, 2013.
17. Chersoni S, Bertacci A, Pashley D, et al. In vivo effects of fluoride on enamel permeability. Clin Oral Investig. 2011;15(4):443-449.
18. National Interprofessional Initiative on Oral Health. Smiles for life curriculum (2011). www.niioh.org/smiles-life-curriculum. Accessed February 11, 2013.
19. Douglass AB, Gonsalves W, Maier R, et al. Smiles for Life: a national oral health curriculum for family medicine. Fam Med. 2007;39(2):88-90.
20. Medicaid.gov. Children’s Health Insurance Program (CHIP). www.medicaid .gov/Medicaid-CHIP-Program-Information/By-Topics/Childrens-Health-Insurance-Program-CHIP/Childrens-Health-Insurance-Program-CHIP.html. Accessed February 11, 2013.
21. Pourat N, Finocchio L. Racial and ethnic disparities in dental care for publicly insured children. Health Aff (Millwood). 2010;29(7):1356-1363.
22. Edelstein BL, Chinn CH. Update on disparities in oral health and access to dental care for America’s children. Acad Pediatr. 2009;9(6):415-419.
23. Seale NS, McWhorter AG, Moulradian WE. Dental education’s role in improving children’s oral health and access to care. Acad Pediatr. 2009;9(6):440-445.
24. National Institute of Dental and Craniofacial Research, NIH. Oral Health in America: A Report of the Surgeon General (Executive Summary; 2011). www.nidcr.nih.gov/datastatistics/surgeongeneral/report/executivesummary.htm. Accessed February 5, 2013.
25. Grembowski D, Spiekerman C, Milgrom P. Social gradients in dental health among low-income mothers and their young children. J Health Care Poor Underserved. 2012;23(2):570-588.
26. Institute of Medicine of the National Academies. Improving access to oral health care for vulnerable and underserved populations (2011). www.iom.edu/~/media/Files/Report Files/2011/Improving-Access-to-Oral-Health-Care-for-Vulnerable-and-Underserved-Populations/oralhealthaccess2011reportbrief.pdf. Accessed February 11, 2013.
27. Bertolami CN. Access to dental care: is there a problem? Am J Public Health. 2011;101(10):1817.
28. Bazargan N, Chi DL, Milgrom P. Exploring the potential for foreign-trained dentists to address workforce shortages and improve access to dental care for vulnerable populations in the United States: a case study from Washington State. BMC Health Serv Res. 2010;10:336-343.
29. Nash DA. Adding dental therapists to the health care team to improve access to oral health care for children. Acad Pediatr. 2009;9(6):446-451.
30. Liao CC, Ganz ML, Jiang H, Chelmow T. The impact of the public insurance expansions on children’s use of preventive dental care. Matern Child Health J. 2010;14(1):58-66.
31. Duley SI, Fitzpatrick PG, Zornosa X, Barnes WG. A center for oral health promotion: establishing an inter-professional paradigm for dental hygiene, health care management and nursing education. J Dent Hyg. 2012;86(2):63-70.
32. Ritter AV, Eidson RS, Donovan TE. Dental caries: etiology, clinical characteristics, risk assessment, and management. In: Heymann HO, Swift EJ Jr, Ritter AV, eds. Sturdevant’s Art and Science of Operative Dentistry. 6th ed. St. Louis: Elsevier-Mosby; 2012:41-89.
33. Shaefer HL, Miller M. Improving access to oral health care services among underserved populations in the US: is there a role for mid-level dental providers? J Health Care Poor Underserved. 2011;22(3):740-744.
34. Li KY, Wong MC, Lam KF, Schwarz E. Age, period, and cohort analysis of regular dental care behavior and edentulism: a marginal approach. BMC Oral Health. 2011;11:9.
35. Beauchamp J, Caufield PW, Crall JJ, et al. Evidence-based clinical recommendations for the use of pit-and-fissure sealants: a report of the American Dental Association Council on Scientific Affairs. J Am Dent Assoc. 2008;139(3):257-268.
36. National Institute of Dental and Craniofacial Research, NIH. Dental sealants in children (age 8 to 11)(2011). www.nidcr.nih.gov/DataStatistics/FindDataByTopic/DentalSealants/Children. Accessed February 11, 2013.
37. Gore DR. The use of dental sealants in adults: a long-neglected preventive measure. Int J Dent Hyg. 2010;8(3):198-203.
38. Devlin D, Henshaw M. Improving access to preventive dental services through a school-based dental sealant program. J Dent Hyg. 2011;85(3):211-219.
39. Mulic A, Tveit AB, Songe D, et al. Dental erosive wear and salivary flow rate in physically active young adults. BMC Oral Health. 2012;12:8.
40. Kaylor MB, Polivka BJ, Chaudry R, et al. Dental services utilization by women of child-bearing age by socioeconomic status. J Community Health. 2010;35(2):190-197.
41. Michalowicz BS, Hodges JS, DiAngelis AJ, et al; OPT Study. Treatment of periodontal disease and the risk of preterm birth. N Engl J Med. 2006;355(18):1885-1894.
42. Silk H, Douglass AB, Douglass JM, Silk L. Oral health during pregnancy. Am Fam Physician. 2008;77(8):1139-1144.
43. Clothier B, Stringer M, Jeffcoat MK. Periodontal disease and pregnancy outcomes: exposure, risk and intervention. Best Pract Res Clin Obstet Gynaecol. 2007;21(3):451-466.
44. Ressler-Maerlender J, Krishna R, Robison V. Oral health during pregnancy: current research. J Womens Health (Larchmt). 2005;14(10):880-882.
45. Berkowitz RJ. Acquisition and transmission of mutans streptococci. J Calif Dent Assoc. 2003;31(2):135-138.
46. Vollmer WM, Papas AS, Bader JD, et al; PACS Collaborative Research Group. Design of the Prevention of Adult Caries Study (PACS): a randomized clinical trial assessing the effect of chlorhexidine dental coating for the protection of adult caries. BMC Oral Health. 2010;10:23.
47. Dhull KS, Nandlal B. Effect of low-concentration daily fluoride application on fluoride release of giomer and compomer: an in vitro study. J Indian Soc Pedod Prev Dent. 2011;29(1):39-45.
48. Naumova EA, Kuehnl P, Hertenstein P, et al. Fluoride bioavailability in saliva and plaque. BMC Oral Health. 2012;12:3.
49. Duckworth RM, Maguire A, Omid N, et al. Effect of rinsing with mouthwash after brushing with fluoridated toothpaste on salivary fluoride concentration. Caries Res. 2009;43(5):391-396.
50. Opydo-Szymaczek J, Opydo J. Salivary fluoride concentrations and fluoride ingestion following application of preparations containing high concentration of fluoride. Biol Trace Elem Res. 2010;137(2):159-167.
51. Medicare.gov. Your Medicare coverage: dental services. www.medicare.gov/coverage/dental-services.html. Accessed February 11, 2013.
52. Quiñonez C, Ieraci L, Guttmann A. Potentially preventable hospital use for dental conditions: implications for expanding dental coverage for low income populations. J Health Care Poor Underserved. 2011;22(3):1048-1058.
53. Deshpande K, Jain A, Sharma RK, et al. Diabetes and periodontitis. J Indian Soc Periodontol. 2010;14(4):207-212.
54. Grubbs V, Plantinga LC, Tuot DS, Powe NR. Chronic kidney disease and use of dental services in a United States public healthcare system: a retrospective cohort study. BMC Nephrol. 2012;13:16.
55. Nair DR, Pruthy R, Pawar U, Chaturvedi P. Oral cancer: premalignant conditions and screening—an update. J Cancer Res Ther. 2012;8 suppl 1:S57-S66.
56. Wu B, Plassman BL, Lian J, Wei L. Cognitive function and dental care utilization among community-dwelling older adults. Am J Public Health. 2007;97(12):2216-2221.
57. Manski RJ, Moeller JF, St Clair PA, et al. The influence of changes in dental care coverage on dental care utilization among retirees and near-retirees in the United States, 2004-2006. Am J Public Health. 2011;101(10):1882-1891.
58. Moeller JF, Chen H, Manski RJ. Investing in preventive dental care for the Medicare population: a preliminary analysis. Am J Public Health. 2010;100(11):2262-2269.
59. National Institute on Aging, NIH. AgePage: Taking care of your teeth and mouth (2011). www.nia.nih.gov/health/publication/taking-care-your-teeth-and-mouth. Accessed February 6, 2013.
Chronic headache: Stop the pain before it starts
• Treat medication overuse headache by withdrawing abortive therapy and initiating prophylactic therapy. C
• Select prophylactic medications that are first-line therapy for chronic migraine or tension-type headache and are appropriate for the patient’s comorbidities. C
• Advise patients to limit intake of abortive headache medications to ≤9 per month. B
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
CASE Eric K, age 25, is in your office, seeking help for chronic headache—which he’s had nearly every day for the past 9 months. He says that the headaches vary in quality and intensity. Sometimes the pain is in the right temporal area; has a throbbing, pulsating quality; and is accompanied by nausea and photophobia. These headaches are incapacitating, with an intensity of 10 on a scale of one to 10. When they occur, the patient reports, all he can do is take migraine medication and lie down in a darkened room for several hours, until the pain goes away. He cannot identify any triggers.
He also gets headaches that are not incapacitating, but occur almost daily, the patient says, describing a dull bilateral pressure that usually begins in the afternoon and worsens until he takes headache medication. He denies any fever, chills, weight loss, visual changes, or tinnitus. His medical history is significant only for obesity, but a system review is positive for depression and insomnia. Physical examination reveals normal vital signs; normal head, eyes, ears, nose, and throat (HEENT); and normal fundoscopic and neurological exams.
Patients like Mr. K can be challenging for primary care physicians, but referral to a neurologist is indicated only in the most intractable cases. For the vast majority of patients with frequent headaches, family physicians can perform the diagnostic work-up and oversee treatment. This review will help with both.
What kind of headache?
While most headaches are sporadic in nature, the prevalence of “chronic daily headache” ranges from 3% to 5% worldwide.1-3
Chronic daily headache is not a diagnosis, however, nor is it an indication that a patient has a headache every day. According to the International Classification of Headache Disorders (ICHD-II), chronic daily headache encompasses several distinct primary headache disorders that have a frequency of ≥15 times per month for at least 3 months. These disorders are also classified by duration, as long (>4 hours) or short.4
The text and tables that follow focus on the diagnosis and treatment of the 4 primary headache disorders of long duration—chronic migraine (CM), chronic tension-type headache (CTTH), hemicrania continua (HC), and new daily persistent headache (NDPH) (TABLE 1).4,5 Although medication overuse headache (MOH) is not a primary headache, it is included in this review because it often contributes to and complicates treatment of primary headache disorders. What’s more, most chronic daily headache syndromes are inextricably linked to medication overuse.6,7
Which individuals are at risk?
Risk factors for chronic headache include female sex, older age, obesity, heavy caffeine consumption, tobacco use, low educational level, overuse of abortive headache medications, and a history of head and neck trauma.8 Episodic migraine (EM)—that is, migraines that occur ≤14 times a month—is also a risk factor for chronic headache.
ICHD-II classifies EM as a progressive disease that transforms to CM at a rate of 3% per year.9 Transformation of EM to CM has been found to occur after as few as 5 days of barbiturates or 8 days of opiates per month.10
Patients with EM should be warned about the potential for migraines to become chronic and have their acute headache medications replaced with a prophylactic drug if the frequency approaches 2 per week.
TABLE 1
Diagnosing and treating chronic headache4,5
| Type of headache | ICHD-II diagnostic criteria | First-line treatment |
|---|---|---|
| Chronic migraine |
| Prophylactic therapy:
|
| Chronic tension-type headache |
| Prophylactic therapy:
|
| Medication overuse headache |
| Discontinue overused acute meds; provide headache education; bridge with NSAIDs, prednisone, or botulinum toxin A; begin prophylactic medication |
| Hemicrania continua |
| Indomethacin |
| New daily persistent headache |
| Rule out secondary causes; treat according to migrainous or tension features of headache |
| ICHD-II, International Classification of Headache Disorders; NSAIDs, nonsteroidal anti-inflammatory drugs; SNRIs, serotonin-norepinephrine reuptake inhibitors; TCAs, tricyclic antidepressants. | ||
Pinpointing the type of headache
An accurate diagnosis requires a thorough headache history and a HEENT and neurological examination. The history should include questions about the characteristics of the headache, including the location, intensity, frequency, timing, associated symptoms, previous headache diagnoses, and triggers, and address comorbidities, medication use, caffeine intake, and family history.8 In the absence of red flags—age >50 years, history of headache or systemic illness, sudden onset, or papilledema, among other findings that may indicate more serious conditions (TABLE 2)7—advanced imaging and further work-up are not needed.
TABLE 2
Beyond headache: Red flags warrant additional testing7
| Red flag | Condition(s) to rule out |
|---|---|
| Age of onset >50 y | Giant cell arteritis, mass lesion, stroke |
| No prior history of headache OR change in characteristic from prior headaches | Cancer, aneurysm, stroke, cerebral sinus thrombosis, infection |
| “Thunderclap” headache | Ruptured aneurysm |
| Signs or symptoms of systemic illness (eg, fever, chills, weight loss) | Meningitis, encephalitis, cancer |
| History of systemic illness, such as cancer, autoimmune disease, or HIV | Brain metastasis, mass lesion, autoimmune meningitis, thrombosis |
| Headache brought on by change in head position or Valsalva maneuver | Spontaneous CSF leak or Chiari malformation |
| Occipital location of headache (in children) | Brain tumor |
| Neurological symptoms | Mass lesion, encephalitis |
| Papilledema | Idiopathic intracranial hypertension, cerebral sinus thrombosis |
| CSF, cerebrospinal fluid; HIV, human immunodeficiency virus. | |
Migraine or tension headache?
Chronic migraine. To be classified as CM, the headache must have occurred ≥15 days a month for 3 months or more and have features of migraine, such as unilateral location, pulsating quality, and moderate to severe intensity. Migraines are aggravated by physical activity and associated with nausea and/or vomiting, photophobia, and phonophobia, and may or may not be preceded by aura. Common triggers include stress, menstruation, alcohol, skipped meals, dehydration, and chocolate. Migraines typically respond to ergots and triptans.4,5
Partial treatment. Patients with CM often take medication early in the course of a headache. This sometimes results in a partially treated migraine that develops into a headache with tension-type features, such as a bilateral location, a pressing quality, and mild-to-moderate intensity, as well as a possible transformation to MOH. This is most likely to occur in patients who have migraines without an aura.
To avoid partial treatment, medications for acute migraine should be taken within 30 minutes of an attack, in a dose that’s sufficient to relieve the pain within 2 hours, with no need for a second dose—a protocol known as “one and done.” Efficacy of a triptan can be improved by adding a nonsteroidal anti-inflammatory drug (NSAID).10
A definitive diagnosis of CM is only possible in the absence of medication overuse.4,5 A patient who is overusing abortive headache medication and whose headache meets the criteria for CM should be given a diagnosis of probable CM instead.
Chronic tension-type headache. In addition to traits common to tension headaches, CTTH may be associated with mild nausea, photophobia, or phonophobia (but typically only one such feature at a time). There may also be tenderness to palpation of the pericranial muscles. Unlike migraine, CTTH is not affected by physical activity.
Here, too, overuse of headache medication is often a factor and should be stopped, if possible, before a definitive diagnosis of CTTH can be made.
Headache with overlapping features. It is possible for a patient to have chronic headache with features of both migraine and tension headache. Advise patients whose headaches have varying characteristics to keep a headache journal to determine which features are more prominent. Patients with smart phones can download a free app, such as iHeadache or My Headache Log Pro, to be used for this purpose.11,12
When to suspect medication overuse headache
MOH is sometimes referred to as a rebound headache or drug-induced headache. Headaches associated with medication overuse have variable intensity. Patients with MOH often awaken from sleep with a headache, and neck pain is highly prevalent.10
Quantifying overuse. According to ICHD-II, overuse is defined as using a single abortive headache medication ≥10 times a month or using 2 or more such drugs ≥15 times a month.5
Triptans have the potential to cause MOH more quickly and in lower doses compared with other acute headache medications. However, analgesics—especially combination products such as butalbital/acetaminophen/caffeine (Fioricet)—are most frequently associated with the development of MOH.13,14 NSAIDs have less potential to cause MOH and are sometimes given as bridge therapy for patients who are discontinuing their acute headache medication.
Less common primary headache disorders
Hemicrania continua, a rare cause of chronic daily headache, is unilateral, without shifting sides, and the intensity is moderate to severe—and unrelenting. HC is associated with autonomic features such as lacrimation, ptosis, and nasal congestion.
New daily persistent headache is characterized by an out-of-the-blue onset of a headache that becomes unremitting soon after it develops. To receive a diagnosis of NDPH, the patient must have a headache that started suddenly and has continued for 3 months or more.
Most patients diagnosed with NDPH are able to recall, to the day, when the headache started. More than 50% report a precipitating event, such as a viral illness, a stressful experience, or surgery.15 ICHD-II defines NDPH as having the characteristics of a tension headache. Notably, however, migrainous features are also common, and neurologists often diagnose NDPH with either migrainous or tension-type features.16
The sudden onset of NDPH is a red flag and, like other red flags, always warrants further work-up. Magnetic resonance imaging with gadolinium is preferred to computed tomography. Magnetic resonance venography or lumbar puncture may also be considered.15,16
Review comorbidities, rule out secondary causes
Patients who suffer from frequent headaches have a high prevalence of depression, anxiety,17,18 sleep disorders,19 obesity,20 irritable bowel disease, fibromyalgia,21 temporomandibular joint disorder,22 and chronic fatigue syndrome. Treatment of these disorders may increase the efficacy of headache treatment. Conversely, overuse of headache medications can make comorbidities harder to treat.
Treating chronic headache: Which drugs are best?
A multimodal approach combining pharmacologic and nonpharmacologic therapies is usually required for patients with chronic headache. The particular therapy and prognosis depend on the type of headache a patient has and the presence of comorbidities (TABLE 3).6,7,23,24
TABLE 3
Consider comorbidities in prophylaxis selection6,7,23,24
| Comorbidity | What to choose | What to avoid |
|---|---|---|
| Depression | Venlafaxine | — |
| Bipolar disorder | Valproic acid | Venlafaxine, amitriptyline, mirtazapine |
| Insomnia (CM) | Amitriptyline | — |
| Insomnia (CTTH) | Mirtazapine | — |
| Obesity | Topiramate | Amitriptyline |
| Hypertension | Metoprolol, propranolol | — |
| Cardiac conduction abnormalities | — | Amitriptyline |
| Fibromyalgia | Amitriptyline, tizanidine | — |
| CM, chronic migraine; CTTH, chronic tension-type headache. | ||
Choice for migraine prophylaxis? Here’s what the evidence tells us
Although most studies of the benefits of prophylaxis have involved patients with episodic or frequent migraine rather than CM, extrapolation of the findings to patients with CM is not unreasonable. And, although dozens of pharmacologic and complementary therapies have been studied for migraine prophylaxis and certain classes of drugs have been identified as effective, there are very few head-to-head trials comparing agents.
The American Academy of Neurology and the American Headache Society published a summary of the evidence in 2012.23 Key findings: The types of medication with the most evidence to support their use as first-line agents for CM are antidepressants, anticonvulsants, and beta-blockers.
Antidepressants, especially tricyclics (TCAs) and serotonin-norepinephrine reuptake inhibitors (SNRIs), have been found to be effective. Chief among them are amitriptyline, a TCA, which is inexpensive and may be beneficial for patients with coexisting insomnia due to its sedating effect, and venlafaxine, an SNRI, which may help treat comorbid depression.23 Amitriptyline is associated with weight gain and can prolong the QT interval at higher doses. There is insufficient or conflicting evidence of the value of selective serotonin reuptake inhibitors for migraine prophylaxis.
Anticonvulsants that have been studied most extensively for migraine are topiramate and sodium valproate. Both have level A ratings for established efficacy.23 Topiramate has also been shown to be noninferior to amitriptyline in reducing migraine frequency and is associated with weight loss, rather than weight gain.25 (Topiramate and valproic acid should be avoided in women who are hoping to become pregnant.) Gabapentin has conflicting evidence and is not recommended for migraine.23
Beta-blockers that appear to be most effective as prophylaxis for CM are propranolol, metoprolol, and timolol.23,26 Any of these would be the obvious choice for a patient with comorbid hypertension. Beta-blockers can take several months to have an effect on migraines, however. Their use as CM prophylaxis may be limited by their adverse effect profile, which includes erectile dysfunction, bradycardia, and hypotension, although the lower dosage needed for migraine prophylaxis may be a mitigating factor. Calcium channel blockers are commonly prescribed for migraine, but there is little evidence to support their use for CM.23
Other medications that are likely effective for migraine prophylaxis include naproxen24 and tizanidine27 (a muscle relaxant). Complementary and alternative treatments that appear to be effective include magnesium, feverfew, butterbur, and riboflavin, although the benefits may not be noticeable for several months.24
Botulinum toxin A is the only medication approved by the US Food and Drug Administration for prevention of CM. It is generally considered to be a second-line agent because of its high cost and the need for training and expertise to administer it. Botulinum toxin A is not effective as prophylaxis for EM.28
Treating other headache syndromes
Chronic tension-type headache. Treatment of CTTH applies similar principles to those of CM, and amitriptyline and venlafaxine—as well as mirtazapine, a sedating SNRI—have evidence to support their use for this type of headache.29 Overall, however, CTTH therapies have not been studied as extensively as those for migraine. There is conflicting evidence of the value of anticonvulsants for prophylaxis of CTTH, and botulinum toxin A has been shown to be no better than placebo.30
Medication overuse headache. Prophylactic medications are not effective in patients who are overusing acute headache medications, and patients with MOH should be instructed to stop the offending drugs. Withdrawal of triptans, simple analgesics, and ergots—either cold turkey or with a slow wean over 4 to 6 weeks—is fairly safe and can be done in an outpatient setting. Concomitant use of prednisone, long-acting NSAIDs, or botulinum toxin A can be used as “bridge therapy” to relieve acute pain. Start the patient on a prophylactic medication based on the best estimate of his or her baseline headache and comorbidities.31,32 For patients who have been overusing opiates or barbiturates, most experts recommend inpatient treatment to manage withdrawal symptoms and prevent complications.10
Most patients with MOH will improve with drug withdrawal, but some will be left with the same disabling headaches that caused them to overuse medication in the first place. For such patients, weekly office visits during the withdrawal period may be helpful. After completion of the bridge therapy, they will likely require abortive headache treatment, but its use must be limited to no more than twice a week. Referral to a specialty headache clinic may be appropriate for such patients.
Hemicrania continua. The treatment for HC is indomethacin. A 2- to 5-day course typically results in complete recovery.
New daily persistent headache. For patients with NDPH, the first step is ruling out secondary causes. Once that has been done, most experts recommend trying to characterize the headache as having features of either migraine or tension and treating accordingly with preventive therapy. If acute headache medication is still needed, limit the quantity you prescribe and stress the importance of taking it no more than twice a week.
CASE Mr. K receives a diagnosis of MOH and probable CM. You explain the way MOH develops and how his medication use has contributed to the escalation of his headaches, and ask him to stop all the headache medications he has been using and to keep a headache journal. You prescribe meloxicam as a short-term bridge therapy and low-dose venlafaxine, which is increased to 150 mg/d over the next 4 weeks; recommend riboflavin 400 mg/d; and refer Mr. K to a neurologist for botulinum toxin A.
You ask him to return in 4 weeks and explain that because he has successfully stopped the overuse of acute headache medications, he can begin taking them again—provided he limits their use to no more than twice a week.
Nonpharmacologic measures can help, too
Lifestyle modification can play an important role in the treatment of chronic daily headache. Advise patients of the importance of proper sleep hygiene, regular exercise, stress reduction, and a healthy diet, as well as avoiding known triggers and minimizing intake of caffeine. Tell patients that biofeedback, cognitive behavioral therapy, and physical therapy may play a role in conjunction with pharmacotherapy, especially for CTTH,26,29,33 but that hypnosis, acupuncture, chiropractic manipulation, transcutaneous electrical nerve stimulation, and hyperbaric oxygen have too little evidence to recommend for or against their use.26,34
In discussing treatment for chronic headache and the goals of therapy with a patient with chronic headache, it is important to be frank. Explain that while a complete cure is not always possible, a decrease in both the frequency and severity of headaches and an improvement in the quality of life and the patient’s ability to function are realistic goals.
CASE At the 3-month follow-up, Mr. K reports that his headaches are down to less than twice a week, and that he is undergoing cognitive behavioral therapy for depression. For acute headache pain, he takes sumatriptan 100 mg with ibuprofen 800 mg, and is careful not to do so more than twice a week.
1. Wiendels NJ, Neven AK, Rosendaal FR, et al. Chronic frequent headache in the general population: prevalence and associated factors. Cephalalgia. 2006;26:1434-1442.
2. Scher AI, Stewart WF, Liberman J, et al. Prevalence of frequent headache in a population sample. Headache. 1998;38:497-506.
3. Castillo J, Munoz P, Guitera V, et al. Kaplan Award 1998. Epidemiology of chronic daily headache in the general population. Headache. 1999;39:190-196.
4. Headache Classification Subcommittee of the International Headache Society. The International Classification of Headache Disorders: 2nd ed. Cephalalgia. 2004;24(suppl):S1-S9.
5. Headache Classification Committee, Olesen J, Bousser MG, Diener HC, et al. New appendix criteria open for a broader concept of chronic migraine. Cephalalgia. 2006;26:742-746.
6. Dodick DW. Clinical practice. Chronic daily headache. N Engl J Med. 2006;354:158-165.
7. Maizels M. The patient with daily headaches. Am Fam Physician. 2004;70:2299-2306.
8. Scher AI, Lipton RB, Stewart WF. Risk factors for chronic daily headache. Curr Pain Headache Rep. 2002;6:486-491.
9. Lipton RB. Tracing transformation: chronic migraine classification progression, and epidemiology. Neurology. 2009;72 (5 suppl):S3-S7.
10. Tepper SJ. Medication-overuse headache. Continuum. 2012;18:807-822.
11. iHeadache. Available at: https://itunes.apple.com/us/app/iheadache-free-headache-migraine/id374213833?mt=8. Accessed February 10, 2013.
12. My Headache Log Pro. Available at: https://play.google.com/store/apps/details?id=com.dontek.myheadachelog&hl=en. Accessed February 10 2013.
13. Bigal ME, Serrano D, Buse D, et al. Acute migraine medications and evolution from episodic to chronic migraine: a longitudinal population-based study. Headache. 2008;48:1157-1168.
14. Colas R, Munoz P, Temprano R, et al. Chronic daily headache with analgesic overuse: epidemiology and impact on quality of life. Neurology. 2004;62:1338-1342.
15. Li D, Rozen TD. The clinical characteristics of new daily persistent headache. Cephalalgia. 2002;22:66-69.
16. Young WB, Swanson JW. New daily-persistent headache: the switched-on headache. Neurology. 2010;74:1338-1339.
17. Verri AP, Proietti Cecchini A, Galli C, et al. Psychiatric comorbidity in chronic daily headache. Cephalalgia. 1998;18 (suppl 21):S45-S49.
18. Tietjen GE, Brandes JL, Digre KB, et al. High prevalence of somatic symptoms and depression in women with disabling chronic headache. Neurology. 2007;68:134.-
19. Kelman L, Rains JC. Headache and sleep: examination of sleep patterns and complaints in a large clinical sample of migraineurs. Headache. 2005;45:904-910.
20. Bigal ME, Lipton RB. Obesity is a risk factor for transformed migraine but not chronic tension-type headache. Neurology. 2006;67:252-257.
21. Peres MF, Young WB, Kaup AO, et al. Fibromyalgia is common in patients with transformed migraine. Neurology. 2001;57:1326-1328.
22. Ciancaglini R, Radaelli G. The relationship between headache and symptoms of temporomandibular disorders in the general population. J Dent. 2001;29:93-98.
23. Silberstein SD, Holland S, Freitag F, et al. Evidence-based guideline update: pharmacologic treatment for episodic migraine prevention in adults: report of the Quality Standards Subcommittee of the American Academy of Neurology and the American Headache Society. Neurology. 2012;78:1337-1345.
24. Holland S, Silberstein SD, Freitag F, et al. Evidence-based guideline update: NSAIDs and other complementary treatments for episodic migraine prevention in adults: report of the Quality Standards Subcommittee of the American Academy of Neurology and the American Headache Society. Neurology. 2012;78:1346-1353.
25. Dodick DW, Freitag F, Banks J, et al. Topiramate versus amitriptyline in migraine prevention: a 26-week, multicenter, randomized, double-blind, double-dummy, parallel-group noninferiority trial in adult migrainers. Clin Ther. 2009;31:542-559.
26. Linde K, Rossnagel K. Propranolol for migraine prophylaxis. Cochrane Database Syst Rev. 2004;(2):CD003225.-
27. Saper JR, Lake AE, Cantrell DT, et al. Chronic daily headache prophylaxis with tizanidine: a double-blind, placebo-controlled, multicenter outcome study. Headache. 2002;42:470-482.
28. Dodick DW, Turkel CC, DeGryse RE, et al. Onabotulinumtoxin A for treatment of chronic migraine: pooled results from the double-blind, randomized, placebo-controlled phases of the PREEMPT clinical program. Headache. 2010;50:921-936.
29. Bendsten L, Evers S, Linde M, et al. EFNS guideline on the treatment of tension-type headache – report of an EFNS task force. Eur J Neurol. 2010;17:1318-1325.
30. Silberstein SD, Gobel H, Jensen R, et al. Botulinum toxin type A in the prophylactic treatment of chronic tension-type headache: a multicenter, double-blind, randomized, placebo-controlled, parallel-group study. Cephalalgia. 2006;26:790-800.
31. Katsavara Z, Jensen R. Medication-overuse headache: where are we now? Curr Opin Neurol. 2007;20:326-330.
32. Zeeberg P, Olesen J, Jensen R. Discontinuation of medication overuse in headache patients: recovery of therapeutic responsiveness. Cephalalgia. 2006;26:1192-1198.
33. Garza I, Schwedt TJ. Diagnosis and management of chronic daily headache. Semin Neurol. 2010;30:154-166.
34. Li Y, Zheng H, Witt CM, et al. Acupuncture for migraine prophylaxis: a randomized controlled trial. CMAJ. 2012;184:401-410.
CORRESPONDENCE Kelly M. Latimer, MD, MPH, FAAFP, Naval Hospital, Camp Lejeune, NC 28542; [email protected]
• Treat medication overuse headache by withdrawing abortive therapy and initiating prophylactic therapy. C
• Select prophylactic medications that are first-line therapy for chronic migraine or tension-type headache and are appropriate for the patient’s comorbidities. C
• Advise patients to limit intake of abortive headache medications to ≤9 per month. B
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
CASE Eric K, age 25, is in your office, seeking help for chronic headache—which he’s had nearly every day for the past 9 months. He says that the headaches vary in quality and intensity. Sometimes the pain is in the right temporal area; has a throbbing, pulsating quality; and is accompanied by nausea and photophobia. These headaches are incapacitating, with an intensity of 10 on a scale of one to 10. When they occur, the patient reports, all he can do is take migraine medication and lie down in a darkened room for several hours, until the pain goes away. He cannot identify any triggers.
He also gets headaches that are not incapacitating, but occur almost daily, the patient says, describing a dull bilateral pressure that usually begins in the afternoon and worsens until he takes headache medication. He denies any fever, chills, weight loss, visual changes, or tinnitus. His medical history is significant only for obesity, but a system review is positive for depression and insomnia. Physical examination reveals normal vital signs; normal head, eyes, ears, nose, and throat (HEENT); and normal fundoscopic and neurological exams.
Patients like Mr. K can be challenging for primary care physicians, but referral to a neurologist is indicated only in the most intractable cases. For the vast majority of patients with frequent headaches, family physicians can perform the diagnostic work-up and oversee treatment. This review will help with both.
What kind of headache?
While most headaches are sporadic in nature, the prevalence of “chronic daily headache” ranges from 3% to 5% worldwide.1-3
Chronic daily headache is not a diagnosis, however, nor is it an indication that a patient has a headache every day. According to the International Classification of Headache Disorders (ICHD-II), chronic daily headache encompasses several distinct primary headache disorders that have a frequency of ≥15 times per month for at least 3 months. These disorders are also classified by duration, as long (>4 hours) or short.4
The text and tables that follow focus on the diagnosis and treatment of the 4 primary headache disorders of long duration—chronic migraine (CM), chronic tension-type headache (CTTH), hemicrania continua (HC), and new daily persistent headache (NDPH) (TABLE 1).4,5 Although medication overuse headache (MOH) is not a primary headache, it is included in this review because it often contributes to and complicates treatment of primary headache disorders. What’s more, most chronic daily headache syndromes are inextricably linked to medication overuse.6,7
Which individuals are at risk?
Risk factors for chronic headache include female sex, older age, obesity, heavy caffeine consumption, tobacco use, low educational level, overuse of abortive headache medications, and a history of head and neck trauma.8 Episodic migraine (EM)—that is, migraines that occur ≤14 times a month—is also a risk factor for chronic headache.
ICHD-II classifies EM as a progressive disease that transforms to CM at a rate of 3% per year.9 Transformation of EM to CM has been found to occur after as few as 5 days of barbiturates or 8 days of opiates per month.10
Patients with EM should be warned about the potential for migraines to become chronic and have their acute headache medications replaced with a prophylactic drug if the frequency approaches 2 per week.
TABLE 1
Diagnosing and treating chronic headache4,5
| Type of headache | ICHD-II diagnostic criteria | First-line treatment |
|---|---|---|
| Chronic migraine |
| Prophylactic therapy:
|
| Chronic tension-type headache |
| Prophylactic therapy:
|
| Medication overuse headache |
| Discontinue overused acute meds; provide headache education; bridge with NSAIDs, prednisone, or botulinum toxin A; begin prophylactic medication |
| Hemicrania continua |
| Indomethacin |
| New daily persistent headache |
| Rule out secondary causes; treat according to migrainous or tension features of headache |
| ICHD-II, International Classification of Headache Disorders; NSAIDs, nonsteroidal anti-inflammatory drugs; SNRIs, serotonin-norepinephrine reuptake inhibitors; TCAs, tricyclic antidepressants. | ||
Pinpointing the type of headache
An accurate diagnosis requires a thorough headache history and a HEENT and neurological examination. The history should include questions about the characteristics of the headache, including the location, intensity, frequency, timing, associated symptoms, previous headache diagnoses, and triggers, and address comorbidities, medication use, caffeine intake, and family history.8 In the absence of red flags—age >50 years, history of headache or systemic illness, sudden onset, or papilledema, among other findings that may indicate more serious conditions (TABLE 2)7—advanced imaging and further work-up are not needed.
TABLE 2
Beyond headache: Red flags warrant additional testing7
| Red flag | Condition(s) to rule out |
|---|---|
| Age of onset >50 y | Giant cell arteritis, mass lesion, stroke |
| No prior history of headache OR change in characteristic from prior headaches | Cancer, aneurysm, stroke, cerebral sinus thrombosis, infection |
| “Thunderclap” headache | Ruptured aneurysm |
| Signs or symptoms of systemic illness (eg, fever, chills, weight loss) | Meningitis, encephalitis, cancer |
| History of systemic illness, such as cancer, autoimmune disease, or HIV | Brain metastasis, mass lesion, autoimmune meningitis, thrombosis |
| Headache brought on by change in head position or Valsalva maneuver | Spontaneous CSF leak or Chiari malformation |
| Occipital location of headache (in children) | Brain tumor |
| Neurological symptoms | Mass lesion, encephalitis |
| Papilledema | Idiopathic intracranial hypertension, cerebral sinus thrombosis |
| CSF, cerebrospinal fluid; HIV, human immunodeficiency virus. | |
Migraine or tension headache?
Chronic migraine. To be classified as CM, the headache must have occurred ≥15 days a month for 3 months or more and have features of migraine, such as unilateral location, pulsating quality, and moderate to severe intensity. Migraines are aggravated by physical activity and associated with nausea and/or vomiting, photophobia, and phonophobia, and may or may not be preceded by aura. Common triggers include stress, menstruation, alcohol, skipped meals, dehydration, and chocolate. Migraines typically respond to ergots and triptans.4,5
Partial treatment. Patients with CM often take medication early in the course of a headache. This sometimes results in a partially treated migraine that develops into a headache with tension-type features, such as a bilateral location, a pressing quality, and mild-to-moderate intensity, as well as a possible transformation to MOH. This is most likely to occur in patients who have migraines without an aura.
To avoid partial treatment, medications for acute migraine should be taken within 30 minutes of an attack, in a dose that’s sufficient to relieve the pain within 2 hours, with no need for a second dose—a protocol known as “one and done.” Efficacy of a triptan can be improved by adding a nonsteroidal anti-inflammatory drug (NSAID).10
A definitive diagnosis of CM is only possible in the absence of medication overuse.4,5 A patient who is overusing abortive headache medication and whose headache meets the criteria for CM should be given a diagnosis of probable CM instead.
Chronic tension-type headache. In addition to traits common to tension headaches, CTTH may be associated with mild nausea, photophobia, or phonophobia (but typically only one such feature at a time). There may also be tenderness to palpation of the pericranial muscles. Unlike migraine, CTTH is not affected by physical activity.
Here, too, overuse of headache medication is often a factor and should be stopped, if possible, before a definitive diagnosis of CTTH can be made.
Headache with overlapping features. It is possible for a patient to have chronic headache with features of both migraine and tension headache. Advise patients whose headaches have varying characteristics to keep a headache journal to determine which features are more prominent. Patients with smart phones can download a free app, such as iHeadache or My Headache Log Pro, to be used for this purpose.11,12
When to suspect medication overuse headache
MOH is sometimes referred to as a rebound headache or drug-induced headache. Headaches associated with medication overuse have variable intensity. Patients with MOH often awaken from sleep with a headache, and neck pain is highly prevalent.10
Quantifying overuse. According to ICHD-II, overuse is defined as using a single abortive headache medication ≥10 times a month or using 2 or more such drugs ≥15 times a month.5
Triptans have the potential to cause MOH more quickly and in lower doses compared with other acute headache medications. However, analgesics—especially combination products such as butalbital/acetaminophen/caffeine (Fioricet)—are most frequently associated with the development of MOH.13,14 NSAIDs have less potential to cause MOH and are sometimes given as bridge therapy for patients who are discontinuing their acute headache medication.
Less common primary headache disorders
Hemicrania continua, a rare cause of chronic daily headache, is unilateral, without shifting sides, and the intensity is moderate to severe—and unrelenting. HC is associated with autonomic features such as lacrimation, ptosis, and nasal congestion.
New daily persistent headache is characterized by an out-of-the-blue onset of a headache that becomes unremitting soon after it develops. To receive a diagnosis of NDPH, the patient must have a headache that started suddenly and has continued for 3 months or more.
Most patients diagnosed with NDPH are able to recall, to the day, when the headache started. More than 50% report a precipitating event, such as a viral illness, a stressful experience, or surgery.15 ICHD-II defines NDPH as having the characteristics of a tension headache. Notably, however, migrainous features are also common, and neurologists often diagnose NDPH with either migrainous or tension-type features.16
The sudden onset of NDPH is a red flag and, like other red flags, always warrants further work-up. Magnetic resonance imaging with gadolinium is preferred to computed tomography. Magnetic resonance venography or lumbar puncture may also be considered.15,16
Review comorbidities, rule out secondary causes
Patients who suffer from frequent headaches have a high prevalence of depression, anxiety,17,18 sleep disorders,19 obesity,20 irritable bowel disease, fibromyalgia,21 temporomandibular joint disorder,22 and chronic fatigue syndrome. Treatment of these disorders may increase the efficacy of headache treatment. Conversely, overuse of headache medications can make comorbidities harder to treat.
Treating chronic headache: Which drugs are best?
A multimodal approach combining pharmacologic and nonpharmacologic therapies is usually required for patients with chronic headache. The particular therapy and prognosis depend on the type of headache a patient has and the presence of comorbidities (TABLE 3).6,7,23,24
TABLE 3
Consider comorbidities in prophylaxis selection6,7,23,24
| Comorbidity | What to choose | What to avoid |
|---|---|---|
| Depression | Venlafaxine | — |
| Bipolar disorder | Valproic acid | Venlafaxine, amitriptyline, mirtazapine |
| Insomnia (CM) | Amitriptyline | — |
| Insomnia (CTTH) | Mirtazapine | — |
| Obesity | Topiramate | Amitriptyline |
| Hypertension | Metoprolol, propranolol | — |
| Cardiac conduction abnormalities | — | Amitriptyline |
| Fibromyalgia | Amitriptyline, tizanidine | — |
| CM, chronic migraine; CTTH, chronic tension-type headache. | ||
Choice for migraine prophylaxis? Here’s what the evidence tells us
Although most studies of the benefits of prophylaxis have involved patients with episodic or frequent migraine rather than CM, extrapolation of the findings to patients with CM is not unreasonable. And, although dozens of pharmacologic and complementary therapies have been studied for migraine prophylaxis and certain classes of drugs have been identified as effective, there are very few head-to-head trials comparing agents.
The American Academy of Neurology and the American Headache Society published a summary of the evidence in 2012.23 Key findings: The types of medication with the most evidence to support their use as first-line agents for CM are antidepressants, anticonvulsants, and beta-blockers.
Antidepressants, especially tricyclics (TCAs) and serotonin-norepinephrine reuptake inhibitors (SNRIs), have been found to be effective. Chief among them are amitriptyline, a TCA, which is inexpensive and may be beneficial for patients with coexisting insomnia due to its sedating effect, and venlafaxine, an SNRI, which may help treat comorbid depression.23 Amitriptyline is associated with weight gain and can prolong the QT interval at higher doses. There is insufficient or conflicting evidence of the value of selective serotonin reuptake inhibitors for migraine prophylaxis.
Anticonvulsants that have been studied most extensively for migraine are topiramate and sodium valproate. Both have level A ratings for established efficacy.23 Topiramate has also been shown to be noninferior to amitriptyline in reducing migraine frequency and is associated with weight loss, rather than weight gain.25 (Topiramate and valproic acid should be avoided in women who are hoping to become pregnant.) Gabapentin has conflicting evidence and is not recommended for migraine.23
Beta-blockers that appear to be most effective as prophylaxis for CM are propranolol, metoprolol, and timolol.23,26 Any of these would be the obvious choice for a patient with comorbid hypertension. Beta-blockers can take several months to have an effect on migraines, however. Their use as CM prophylaxis may be limited by their adverse effect profile, which includes erectile dysfunction, bradycardia, and hypotension, although the lower dosage needed for migraine prophylaxis may be a mitigating factor. Calcium channel blockers are commonly prescribed for migraine, but there is little evidence to support their use for CM.23
Other medications that are likely effective for migraine prophylaxis include naproxen24 and tizanidine27 (a muscle relaxant). Complementary and alternative treatments that appear to be effective include magnesium, feverfew, butterbur, and riboflavin, although the benefits may not be noticeable for several months.24
Botulinum toxin A is the only medication approved by the US Food and Drug Administration for prevention of CM. It is generally considered to be a second-line agent because of its high cost and the need for training and expertise to administer it. Botulinum toxin A is not effective as prophylaxis for EM.28
Treating other headache syndromes
Chronic tension-type headache. Treatment of CTTH applies similar principles to those of CM, and amitriptyline and venlafaxine—as well as mirtazapine, a sedating SNRI—have evidence to support their use for this type of headache.29 Overall, however, CTTH therapies have not been studied as extensively as those for migraine. There is conflicting evidence of the value of anticonvulsants for prophylaxis of CTTH, and botulinum toxin A has been shown to be no better than placebo.30
Medication overuse headache. Prophylactic medications are not effective in patients who are overusing acute headache medications, and patients with MOH should be instructed to stop the offending drugs. Withdrawal of triptans, simple analgesics, and ergots—either cold turkey or with a slow wean over 4 to 6 weeks—is fairly safe and can be done in an outpatient setting. Concomitant use of prednisone, long-acting NSAIDs, or botulinum toxin A can be used as “bridge therapy” to relieve acute pain. Start the patient on a prophylactic medication based on the best estimate of his or her baseline headache and comorbidities.31,32 For patients who have been overusing opiates or barbiturates, most experts recommend inpatient treatment to manage withdrawal symptoms and prevent complications.10
Most patients with MOH will improve with drug withdrawal, but some will be left with the same disabling headaches that caused them to overuse medication in the first place. For such patients, weekly office visits during the withdrawal period may be helpful. After completion of the bridge therapy, they will likely require abortive headache treatment, but its use must be limited to no more than twice a week. Referral to a specialty headache clinic may be appropriate for such patients.
Hemicrania continua. The treatment for HC is indomethacin. A 2- to 5-day course typically results in complete recovery.
New daily persistent headache. For patients with NDPH, the first step is ruling out secondary causes. Once that has been done, most experts recommend trying to characterize the headache as having features of either migraine or tension and treating accordingly with preventive therapy. If acute headache medication is still needed, limit the quantity you prescribe and stress the importance of taking it no more than twice a week.
CASE Mr. K receives a diagnosis of MOH and probable CM. You explain the way MOH develops and how his medication use has contributed to the escalation of his headaches, and ask him to stop all the headache medications he has been using and to keep a headache journal. You prescribe meloxicam as a short-term bridge therapy and low-dose venlafaxine, which is increased to 150 mg/d over the next 4 weeks; recommend riboflavin 400 mg/d; and refer Mr. K to a neurologist for botulinum toxin A.
You ask him to return in 4 weeks and explain that because he has successfully stopped the overuse of acute headache medications, he can begin taking them again—provided he limits their use to no more than twice a week.
Nonpharmacologic measures can help, too
Lifestyle modification can play an important role in the treatment of chronic daily headache. Advise patients of the importance of proper sleep hygiene, regular exercise, stress reduction, and a healthy diet, as well as avoiding known triggers and minimizing intake of caffeine. Tell patients that biofeedback, cognitive behavioral therapy, and physical therapy may play a role in conjunction with pharmacotherapy, especially for CTTH,26,29,33 but that hypnosis, acupuncture, chiropractic manipulation, transcutaneous electrical nerve stimulation, and hyperbaric oxygen have too little evidence to recommend for or against their use.26,34
In discussing treatment for chronic headache and the goals of therapy with a patient with chronic headache, it is important to be frank. Explain that while a complete cure is not always possible, a decrease in both the frequency and severity of headaches and an improvement in the quality of life and the patient’s ability to function are realistic goals.
CASE At the 3-month follow-up, Mr. K reports that his headaches are down to less than twice a week, and that he is undergoing cognitive behavioral therapy for depression. For acute headache pain, he takes sumatriptan 100 mg with ibuprofen 800 mg, and is careful not to do so more than twice a week.
• Treat medication overuse headache by withdrawing abortive therapy and initiating prophylactic therapy. C
• Select prophylactic medications that are first-line therapy for chronic migraine or tension-type headache and are appropriate for the patient’s comorbidities. C
• Advise patients to limit intake of abortive headache medications to ≤9 per month. B
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
CASE Eric K, age 25, is in your office, seeking help for chronic headache—which he’s had nearly every day for the past 9 months. He says that the headaches vary in quality and intensity. Sometimes the pain is in the right temporal area; has a throbbing, pulsating quality; and is accompanied by nausea and photophobia. These headaches are incapacitating, with an intensity of 10 on a scale of one to 10. When they occur, the patient reports, all he can do is take migraine medication and lie down in a darkened room for several hours, until the pain goes away. He cannot identify any triggers.
He also gets headaches that are not incapacitating, but occur almost daily, the patient says, describing a dull bilateral pressure that usually begins in the afternoon and worsens until he takes headache medication. He denies any fever, chills, weight loss, visual changes, or tinnitus. His medical history is significant only for obesity, but a system review is positive for depression and insomnia. Physical examination reveals normal vital signs; normal head, eyes, ears, nose, and throat (HEENT); and normal fundoscopic and neurological exams.
Patients like Mr. K can be challenging for primary care physicians, but referral to a neurologist is indicated only in the most intractable cases. For the vast majority of patients with frequent headaches, family physicians can perform the diagnostic work-up and oversee treatment. This review will help with both.
What kind of headache?
While most headaches are sporadic in nature, the prevalence of “chronic daily headache” ranges from 3% to 5% worldwide.1-3
Chronic daily headache is not a diagnosis, however, nor is it an indication that a patient has a headache every day. According to the International Classification of Headache Disorders (ICHD-II), chronic daily headache encompasses several distinct primary headache disorders that have a frequency of ≥15 times per month for at least 3 months. These disorders are also classified by duration, as long (>4 hours) or short.4
The text and tables that follow focus on the diagnosis and treatment of the 4 primary headache disorders of long duration—chronic migraine (CM), chronic tension-type headache (CTTH), hemicrania continua (HC), and new daily persistent headache (NDPH) (TABLE 1).4,5 Although medication overuse headache (MOH) is not a primary headache, it is included in this review because it often contributes to and complicates treatment of primary headache disorders. What’s more, most chronic daily headache syndromes are inextricably linked to medication overuse.6,7
Which individuals are at risk?
Risk factors for chronic headache include female sex, older age, obesity, heavy caffeine consumption, tobacco use, low educational level, overuse of abortive headache medications, and a history of head and neck trauma.8 Episodic migraine (EM)—that is, migraines that occur ≤14 times a month—is also a risk factor for chronic headache.
ICHD-II classifies EM as a progressive disease that transforms to CM at a rate of 3% per year.9 Transformation of EM to CM has been found to occur after as few as 5 days of barbiturates or 8 days of opiates per month.10
Patients with EM should be warned about the potential for migraines to become chronic and have their acute headache medications replaced with a prophylactic drug if the frequency approaches 2 per week.
TABLE 1
Diagnosing and treating chronic headache4,5
| Type of headache | ICHD-II diagnostic criteria | First-line treatment |
|---|---|---|
| Chronic migraine |
| Prophylactic therapy:
|
| Chronic tension-type headache |
| Prophylactic therapy:
|
| Medication overuse headache |
| Discontinue overused acute meds; provide headache education; bridge with NSAIDs, prednisone, or botulinum toxin A; begin prophylactic medication |
| Hemicrania continua |
| Indomethacin |
| New daily persistent headache |
| Rule out secondary causes; treat according to migrainous or tension features of headache |
| ICHD-II, International Classification of Headache Disorders; NSAIDs, nonsteroidal anti-inflammatory drugs; SNRIs, serotonin-norepinephrine reuptake inhibitors; TCAs, tricyclic antidepressants. | ||
Pinpointing the type of headache
An accurate diagnosis requires a thorough headache history and a HEENT and neurological examination. The history should include questions about the characteristics of the headache, including the location, intensity, frequency, timing, associated symptoms, previous headache diagnoses, and triggers, and address comorbidities, medication use, caffeine intake, and family history.8 In the absence of red flags—age >50 years, history of headache or systemic illness, sudden onset, or papilledema, among other findings that may indicate more serious conditions (TABLE 2)7—advanced imaging and further work-up are not needed.
TABLE 2
Beyond headache: Red flags warrant additional testing7
| Red flag | Condition(s) to rule out |
|---|---|
| Age of onset >50 y | Giant cell arteritis, mass lesion, stroke |
| No prior history of headache OR change in characteristic from prior headaches | Cancer, aneurysm, stroke, cerebral sinus thrombosis, infection |
| “Thunderclap” headache | Ruptured aneurysm |
| Signs or symptoms of systemic illness (eg, fever, chills, weight loss) | Meningitis, encephalitis, cancer |
| History of systemic illness, such as cancer, autoimmune disease, or HIV | Brain metastasis, mass lesion, autoimmune meningitis, thrombosis |
| Headache brought on by change in head position or Valsalva maneuver | Spontaneous CSF leak or Chiari malformation |
| Occipital location of headache (in children) | Brain tumor |
| Neurological symptoms | Mass lesion, encephalitis |
| Papilledema | Idiopathic intracranial hypertension, cerebral sinus thrombosis |
| CSF, cerebrospinal fluid; HIV, human immunodeficiency virus. | |
Migraine or tension headache?
Chronic migraine. To be classified as CM, the headache must have occurred ≥15 days a month for 3 months or more and have features of migraine, such as unilateral location, pulsating quality, and moderate to severe intensity. Migraines are aggravated by physical activity and associated with nausea and/or vomiting, photophobia, and phonophobia, and may or may not be preceded by aura. Common triggers include stress, menstruation, alcohol, skipped meals, dehydration, and chocolate. Migraines typically respond to ergots and triptans.4,5
Partial treatment. Patients with CM often take medication early in the course of a headache. This sometimes results in a partially treated migraine that develops into a headache with tension-type features, such as a bilateral location, a pressing quality, and mild-to-moderate intensity, as well as a possible transformation to MOH. This is most likely to occur in patients who have migraines without an aura.
To avoid partial treatment, medications for acute migraine should be taken within 30 minutes of an attack, in a dose that’s sufficient to relieve the pain within 2 hours, with no need for a second dose—a protocol known as “one and done.” Efficacy of a triptan can be improved by adding a nonsteroidal anti-inflammatory drug (NSAID).10
A definitive diagnosis of CM is only possible in the absence of medication overuse.4,5 A patient who is overusing abortive headache medication and whose headache meets the criteria for CM should be given a diagnosis of probable CM instead.
Chronic tension-type headache. In addition to traits common to tension headaches, CTTH may be associated with mild nausea, photophobia, or phonophobia (but typically only one such feature at a time). There may also be tenderness to palpation of the pericranial muscles. Unlike migraine, CTTH is not affected by physical activity.
Here, too, overuse of headache medication is often a factor and should be stopped, if possible, before a definitive diagnosis of CTTH can be made.
Headache with overlapping features. It is possible for a patient to have chronic headache with features of both migraine and tension headache. Advise patients whose headaches have varying characteristics to keep a headache journal to determine which features are more prominent. Patients with smart phones can download a free app, such as iHeadache or My Headache Log Pro, to be used for this purpose.11,12
When to suspect medication overuse headache
MOH is sometimes referred to as a rebound headache or drug-induced headache. Headaches associated with medication overuse have variable intensity. Patients with MOH often awaken from sleep with a headache, and neck pain is highly prevalent.10
Quantifying overuse. According to ICHD-II, overuse is defined as using a single abortive headache medication ≥10 times a month or using 2 or more such drugs ≥15 times a month.5
Triptans have the potential to cause MOH more quickly and in lower doses compared with other acute headache medications. However, analgesics—especially combination products such as butalbital/acetaminophen/caffeine (Fioricet)—are most frequently associated with the development of MOH.13,14 NSAIDs have less potential to cause MOH and are sometimes given as bridge therapy for patients who are discontinuing their acute headache medication.
Less common primary headache disorders
Hemicrania continua, a rare cause of chronic daily headache, is unilateral, without shifting sides, and the intensity is moderate to severe—and unrelenting. HC is associated with autonomic features such as lacrimation, ptosis, and nasal congestion.
New daily persistent headache is characterized by an out-of-the-blue onset of a headache that becomes unremitting soon after it develops. To receive a diagnosis of NDPH, the patient must have a headache that started suddenly and has continued for 3 months or more.
Most patients diagnosed with NDPH are able to recall, to the day, when the headache started. More than 50% report a precipitating event, such as a viral illness, a stressful experience, or surgery.15 ICHD-II defines NDPH as having the characteristics of a tension headache. Notably, however, migrainous features are also common, and neurologists often diagnose NDPH with either migrainous or tension-type features.16
The sudden onset of NDPH is a red flag and, like other red flags, always warrants further work-up. Magnetic resonance imaging with gadolinium is preferred to computed tomography. Magnetic resonance venography or lumbar puncture may also be considered.15,16
Review comorbidities, rule out secondary causes
Patients who suffer from frequent headaches have a high prevalence of depression, anxiety,17,18 sleep disorders,19 obesity,20 irritable bowel disease, fibromyalgia,21 temporomandibular joint disorder,22 and chronic fatigue syndrome. Treatment of these disorders may increase the efficacy of headache treatment. Conversely, overuse of headache medications can make comorbidities harder to treat.
Treating chronic headache: Which drugs are best?
A multimodal approach combining pharmacologic and nonpharmacologic therapies is usually required for patients with chronic headache. The particular therapy and prognosis depend on the type of headache a patient has and the presence of comorbidities (TABLE 3).6,7,23,24
TABLE 3
Consider comorbidities in prophylaxis selection6,7,23,24
| Comorbidity | What to choose | What to avoid |
|---|---|---|
| Depression | Venlafaxine | — |
| Bipolar disorder | Valproic acid | Venlafaxine, amitriptyline, mirtazapine |
| Insomnia (CM) | Amitriptyline | — |
| Insomnia (CTTH) | Mirtazapine | — |
| Obesity | Topiramate | Amitriptyline |
| Hypertension | Metoprolol, propranolol | — |
| Cardiac conduction abnormalities | — | Amitriptyline |
| Fibromyalgia | Amitriptyline, tizanidine | — |
| CM, chronic migraine; CTTH, chronic tension-type headache. | ||
Choice for migraine prophylaxis? Here’s what the evidence tells us
Although most studies of the benefits of prophylaxis have involved patients with episodic or frequent migraine rather than CM, extrapolation of the findings to patients with CM is not unreasonable. And, although dozens of pharmacologic and complementary therapies have been studied for migraine prophylaxis and certain classes of drugs have been identified as effective, there are very few head-to-head trials comparing agents.
The American Academy of Neurology and the American Headache Society published a summary of the evidence in 2012.23 Key findings: The types of medication with the most evidence to support their use as first-line agents for CM are antidepressants, anticonvulsants, and beta-blockers.
Antidepressants, especially tricyclics (TCAs) and serotonin-norepinephrine reuptake inhibitors (SNRIs), have been found to be effective. Chief among them are amitriptyline, a TCA, which is inexpensive and may be beneficial for patients with coexisting insomnia due to its sedating effect, and venlafaxine, an SNRI, which may help treat comorbid depression.23 Amitriptyline is associated with weight gain and can prolong the QT interval at higher doses. There is insufficient or conflicting evidence of the value of selective serotonin reuptake inhibitors for migraine prophylaxis.
Anticonvulsants that have been studied most extensively for migraine are topiramate and sodium valproate. Both have level A ratings for established efficacy.23 Topiramate has also been shown to be noninferior to amitriptyline in reducing migraine frequency and is associated with weight loss, rather than weight gain.25 (Topiramate and valproic acid should be avoided in women who are hoping to become pregnant.) Gabapentin has conflicting evidence and is not recommended for migraine.23
Beta-blockers that appear to be most effective as prophylaxis for CM are propranolol, metoprolol, and timolol.23,26 Any of these would be the obvious choice for a patient with comorbid hypertension. Beta-blockers can take several months to have an effect on migraines, however. Their use as CM prophylaxis may be limited by their adverse effect profile, which includes erectile dysfunction, bradycardia, and hypotension, although the lower dosage needed for migraine prophylaxis may be a mitigating factor. Calcium channel blockers are commonly prescribed for migraine, but there is little evidence to support their use for CM.23
Other medications that are likely effective for migraine prophylaxis include naproxen24 and tizanidine27 (a muscle relaxant). Complementary and alternative treatments that appear to be effective include magnesium, feverfew, butterbur, and riboflavin, although the benefits may not be noticeable for several months.24
Botulinum toxin A is the only medication approved by the US Food and Drug Administration for prevention of CM. It is generally considered to be a second-line agent because of its high cost and the need for training and expertise to administer it. Botulinum toxin A is not effective as prophylaxis for EM.28
Treating other headache syndromes
Chronic tension-type headache. Treatment of CTTH applies similar principles to those of CM, and amitriptyline and venlafaxine—as well as mirtazapine, a sedating SNRI—have evidence to support their use for this type of headache.29 Overall, however, CTTH therapies have not been studied as extensively as those for migraine. There is conflicting evidence of the value of anticonvulsants for prophylaxis of CTTH, and botulinum toxin A has been shown to be no better than placebo.30
Medication overuse headache. Prophylactic medications are not effective in patients who are overusing acute headache medications, and patients with MOH should be instructed to stop the offending drugs. Withdrawal of triptans, simple analgesics, and ergots—either cold turkey or with a slow wean over 4 to 6 weeks—is fairly safe and can be done in an outpatient setting. Concomitant use of prednisone, long-acting NSAIDs, or botulinum toxin A can be used as “bridge therapy” to relieve acute pain. Start the patient on a prophylactic medication based on the best estimate of his or her baseline headache and comorbidities.31,32 For patients who have been overusing opiates or barbiturates, most experts recommend inpatient treatment to manage withdrawal symptoms and prevent complications.10
Most patients with MOH will improve with drug withdrawal, but some will be left with the same disabling headaches that caused them to overuse medication in the first place. For such patients, weekly office visits during the withdrawal period may be helpful. After completion of the bridge therapy, they will likely require abortive headache treatment, but its use must be limited to no more than twice a week. Referral to a specialty headache clinic may be appropriate for such patients.
Hemicrania continua. The treatment for HC is indomethacin. A 2- to 5-day course typically results in complete recovery.
New daily persistent headache. For patients with NDPH, the first step is ruling out secondary causes. Once that has been done, most experts recommend trying to characterize the headache as having features of either migraine or tension and treating accordingly with preventive therapy. If acute headache medication is still needed, limit the quantity you prescribe and stress the importance of taking it no more than twice a week.
CASE Mr. K receives a diagnosis of MOH and probable CM. You explain the way MOH develops and how his medication use has contributed to the escalation of his headaches, and ask him to stop all the headache medications he has been using and to keep a headache journal. You prescribe meloxicam as a short-term bridge therapy and low-dose venlafaxine, which is increased to 150 mg/d over the next 4 weeks; recommend riboflavin 400 mg/d; and refer Mr. K to a neurologist for botulinum toxin A.
You ask him to return in 4 weeks and explain that because he has successfully stopped the overuse of acute headache medications, he can begin taking them again—provided he limits their use to no more than twice a week.
Nonpharmacologic measures can help, too
Lifestyle modification can play an important role in the treatment of chronic daily headache. Advise patients of the importance of proper sleep hygiene, regular exercise, stress reduction, and a healthy diet, as well as avoiding known triggers and minimizing intake of caffeine. Tell patients that biofeedback, cognitive behavioral therapy, and physical therapy may play a role in conjunction with pharmacotherapy, especially for CTTH,26,29,33 but that hypnosis, acupuncture, chiropractic manipulation, transcutaneous electrical nerve stimulation, and hyperbaric oxygen have too little evidence to recommend for or against their use.26,34
In discussing treatment for chronic headache and the goals of therapy with a patient with chronic headache, it is important to be frank. Explain that while a complete cure is not always possible, a decrease in both the frequency and severity of headaches and an improvement in the quality of life and the patient’s ability to function are realistic goals.
CASE At the 3-month follow-up, Mr. K reports that his headaches are down to less than twice a week, and that he is undergoing cognitive behavioral therapy for depression. For acute headache pain, he takes sumatriptan 100 mg with ibuprofen 800 mg, and is careful not to do so more than twice a week.
1. Wiendels NJ, Neven AK, Rosendaal FR, et al. Chronic frequent headache in the general population: prevalence and associated factors. Cephalalgia. 2006;26:1434-1442.
2. Scher AI, Stewart WF, Liberman J, et al. Prevalence of frequent headache in a population sample. Headache. 1998;38:497-506.
3. Castillo J, Munoz P, Guitera V, et al. Kaplan Award 1998. Epidemiology of chronic daily headache in the general population. Headache. 1999;39:190-196.
4. Headache Classification Subcommittee of the International Headache Society. The International Classification of Headache Disorders: 2nd ed. Cephalalgia. 2004;24(suppl):S1-S9.
5. Headache Classification Committee, Olesen J, Bousser MG, Diener HC, et al. New appendix criteria open for a broader concept of chronic migraine. Cephalalgia. 2006;26:742-746.
6. Dodick DW. Clinical practice. Chronic daily headache. N Engl J Med. 2006;354:158-165.
7. Maizels M. The patient with daily headaches. Am Fam Physician. 2004;70:2299-2306.
8. Scher AI, Lipton RB, Stewart WF. Risk factors for chronic daily headache. Curr Pain Headache Rep. 2002;6:486-491.
9. Lipton RB. Tracing transformation: chronic migraine classification progression, and epidemiology. Neurology. 2009;72 (5 suppl):S3-S7.
10. Tepper SJ. Medication-overuse headache. Continuum. 2012;18:807-822.
11. iHeadache. Available at: https://itunes.apple.com/us/app/iheadache-free-headache-migraine/id374213833?mt=8. Accessed February 10, 2013.
12. My Headache Log Pro. Available at: https://play.google.com/store/apps/details?id=com.dontek.myheadachelog&hl=en. Accessed February 10 2013.
13. Bigal ME, Serrano D, Buse D, et al. Acute migraine medications and evolution from episodic to chronic migraine: a longitudinal population-based study. Headache. 2008;48:1157-1168.
14. Colas R, Munoz P, Temprano R, et al. Chronic daily headache with analgesic overuse: epidemiology and impact on quality of life. Neurology. 2004;62:1338-1342.
15. Li D, Rozen TD. The clinical characteristics of new daily persistent headache. Cephalalgia. 2002;22:66-69.
16. Young WB, Swanson JW. New daily-persistent headache: the switched-on headache. Neurology. 2010;74:1338-1339.
17. Verri AP, Proietti Cecchini A, Galli C, et al. Psychiatric comorbidity in chronic daily headache. Cephalalgia. 1998;18 (suppl 21):S45-S49.
18. Tietjen GE, Brandes JL, Digre KB, et al. High prevalence of somatic symptoms and depression in women with disabling chronic headache. Neurology. 2007;68:134.-
19. Kelman L, Rains JC. Headache and sleep: examination of sleep patterns and complaints in a large clinical sample of migraineurs. Headache. 2005;45:904-910.
20. Bigal ME, Lipton RB. Obesity is a risk factor for transformed migraine but not chronic tension-type headache. Neurology. 2006;67:252-257.
21. Peres MF, Young WB, Kaup AO, et al. Fibromyalgia is common in patients with transformed migraine. Neurology. 2001;57:1326-1328.
22. Ciancaglini R, Radaelli G. The relationship between headache and symptoms of temporomandibular disorders in the general population. J Dent. 2001;29:93-98.
23. Silberstein SD, Holland S, Freitag F, et al. Evidence-based guideline update: pharmacologic treatment for episodic migraine prevention in adults: report of the Quality Standards Subcommittee of the American Academy of Neurology and the American Headache Society. Neurology. 2012;78:1337-1345.
24. Holland S, Silberstein SD, Freitag F, et al. Evidence-based guideline update: NSAIDs and other complementary treatments for episodic migraine prevention in adults: report of the Quality Standards Subcommittee of the American Academy of Neurology and the American Headache Society. Neurology. 2012;78:1346-1353.
25. Dodick DW, Freitag F, Banks J, et al. Topiramate versus amitriptyline in migraine prevention: a 26-week, multicenter, randomized, double-blind, double-dummy, parallel-group noninferiority trial in adult migrainers. Clin Ther. 2009;31:542-559.
26. Linde K, Rossnagel K. Propranolol for migraine prophylaxis. Cochrane Database Syst Rev. 2004;(2):CD003225.-
27. Saper JR, Lake AE, Cantrell DT, et al. Chronic daily headache prophylaxis with tizanidine: a double-blind, placebo-controlled, multicenter outcome study. Headache. 2002;42:470-482.
28. Dodick DW, Turkel CC, DeGryse RE, et al. Onabotulinumtoxin A for treatment of chronic migraine: pooled results from the double-blind, randomized, placebo-controlled phases of the PREEMPT clinical program. Headache. 2010;50:921-936.
29. Bendsten L, Evers S, Linde M, et al. EFNS guideline on the treatment of tension-type headache – report of an EFNS task force. Eur J Neurol. 2010;17:1318-1325.
30. Silberstein SD, Gobel H, Jensen R, et al. Botulinum toxin type A in the prophylactic treatment of chronic tension-type headache: a multicenter, double-blind, randomized, placebo-controlled, parallel-group study. Cephalalgia. 2006;26:790-800.
31. Katsavara Z, Jensen R. Medication-overuse headache: where are we now? Curr Opin Neurol. 2007;20:326-330.
32. Zeeberg P, Olesen J, Jensen R. Discontinuation of medication overuse in headache patients: recovery of therapeutic responsiveness. Cephalalgia. 2006;26:1192-1198.
33. Garza I, Schwedt TJ. Diagnosis and management of chronic daily headache. Semin Neurol. 2010;30:154-166.
34. Li Y, Zheng H, Witt CM, et al. Acupuncture for migraine prophylaxis: a randomized controlled trial. CMAJ. 2012;184:401-410.
CORRESPONDENCE Kelly M. Latimer, MD, MPH, FAAFP, Naval Hospital, Camp Lejeune, NC 28542; [email protected]
1. Wiendels NJ, Neven AK, Rosendaal FR, et al. Chronic frequent headache in the general population: prevalence and associated factors. Cephalalgia. 2006;26:1434-1442.
2. Scher AI, Stewart WF, Liberman J, et al. Prevalence of frequent headache in a population sample. Headache. 1998;38:497-506.
3. Castillo J, Munoz P, Guitera V, et al. Kaplan Award 1998. Epidemiology of chronic daily headache in the general population. Headache. 1999;39:190-196.
4. Headache Classification Subcommittee of the International Headache Society. The International Classification of Headache Disorders: 2nd ed. Cephalalgia. 2004;24(suppl):S1-S9.
5. Headache Classification Committee, Olesen J, Bousser MG, Diener HC, et al. New appendix criteria open for a broader concept of chronic migraine. Cephalalgia. 2006;26:742-746.
6. Dodick DW. Clinical practice. Chronic daily headache. N Engl J Med. 2006;354:158-165.
7. Maizels M. The patient with daily headaches. Am Fam Physician. 2004;70:2299-2306.
8. Scher AI, Lipton RB, Stewart WF. Risk factors for chronic daily headache. Curr Pain Headache Rep. 2002;6:486-491.
9. Lipton RB. Tracing transformation: chronic migraine classification progression, and epidemiology. Neurology. 2009;72 (5 suppl):S3-S7.
10. Tepper SJ. Medication-overuse headache. Continuum. 2012;18:807-822.
11. iHeadache. Available at: https://itunes.apple.com/us/app/iheadache-free-headache-migraine/id374213833?mt=8. Accessed February 10, 2013.
12. My Headache Log Pro. Available at: https://play.google.com/store/apps/details?id=com.dontek.myheadachelog&hl=en. Accessed February 10 2013.
13. Bigal ME, Serrano D, Buse D, et al. Acute migraine medications and evolution from episodic to chronic migraine: a longitudinal population-based study. Headache. 2008;48:1157-1168.
14. Colas R, Munoz P, Temprano R, et al. Chronic daily headache with analgesic overuse: epidemiology and impact on quality of life. Neurology. 2004;62:1338-1342.
15. Li D, Rozen TD. The clinical characteristics of new daily persistent headache. Cephalalgia. 2002;22:66-69.
16. Young WB, Swanson JW. New daily-persistent headache: the switched-on headache. Neurology. 2010;74:1338-1339.
17. Verri AP, Proietti Cecchini A, Galli C, et al. Psychiatric comorbidity in chronic daily headache. Cephalalgia. 1998;18 (suppl 21):S45-S49.
18. Tietjen GE, Brandes JL, Digre KB, et al. High prevalence of somatic symptoms and depression in women with disabling chronic headache. Neurology. 2007;68:134.-
19. Kelman L, Rains JC. Headache and sleep: examination of sleep patterns and complaints in a large clinical sample of migraineurs. Headache. 2005;45:904-910.
20. Bigal ME, Lipton RB. Obesity is a risk factor for transformed migraine but not chronic tension-type headache. Neurology. 2006;67:252-257.
21. Peres MF, Young WB, Kaup AO, et al. Fibromyalgia is common in patients with transformed migraine. Neurology. 2001;57:1326-1328.
22. Ciancaglini R, Radaelli G. The relationship between headache and symptoms of temporomandibular disorders in the general population. J Dent. 2001;29:93-98.
23. Silberstein SD, Holland S, Freitag F, et al. Evidence-based guideline update: pharmacologic treatment for episodic migraine prevention in adults: report of the Quality Standards Subcommittee of the American Academy of Neurology and the American Headache Society. Neurology. 2012;78:1337-1345.
24. Holland S, Silberstein SD, Freitag F, et al. Evidence-based guideline update: NSAIDs and other complementary treatments for episodic migraine prevention in adults: report of the Quality Standards Subcommittee of the American Academy of Neurology and the American Headache Society. Neurology. 2012;78:1346-1353.
25. Dodick DW, Freitag F, Banks J, et al. Topiramate versus amitriptyline in migraine prevention: a 26-week, multicenter, randomized, double-blind, double-dummy, parallel-group noninferiority trial in adult migrainers. Clin Ther. 2009;31:542-559.
26. Linde K, Rossnagel K. Propranolol for migraine prophylaxis. Cochrane Database Syst Rev. 2004;(2):CD003225.-
27. Saper JR, Lake AE, Cantrell DT, et al. Chronic daily headache prophylaxis with tizanidine: a double-blind, placebo-controlled, multicenter outcome study. Headache. 2002;42:470-482.
28. Dodick DW, Turkel CC, DeGryse RE, et al. Onabotulinumtoxin A for treatment of chronic migraine: pooled results from the double-blind, randomized, placebo-controlled phases of the PREEMPT clinical program. Headache. 2010;50:921-936.
29. Bendsten L, Evers S, Linde M, et al. EFNS guideline on the treatment of tension-type headache – report of an EFNS task force. Eur J Neurol. 2010;17:1318-1325.
30. Silberstein SD, Gobel H, Jensen R, et al. Botulinum toxin type A in the prophylactic treatment of chronic tension-type headache: a multicenter, double-blind, randomized, placebo-controlled, parallel-group study. Cephalalgia. 2006;26:790-800.
31. Katsavara Z, Jensen R. Medication-overuse headache: where are we now? Curr Opin Neurol. 2007;20:326-330.
32. Zeeberg P, Olesen J, Jensen R. Discontinuation of medication overuse in headache patients: recovery of therapeutic responsiveness. Cephalalgia. 2006;26:1192-1198.
33. Garza I, Schwedt TJ. Diagnosis and management of chronic daily headache. Semin Neurol. 2010;30:154-166.
34. Li Y, Zheng H, Witt CM, et al. Acupuncture for migraine prophylaxis: a randomized controlled trial. CMAJ. 2012;184:401-410.
CORRESPONDENCE Kelly M. Latimer, MD, MPH, FAAFP, Naval Hospital, Camp Lejeune, NC 28542; [email protected]
The Realities of Panic Disorder
Steroid-Induced Myopathy and Associated Postoperative Complications
Delta NT-proBNP: Better Marker of Neuroendocrine Pathways Blockade in Decompensated Congestive Heart Failure?
Brain Metastasis
Diagnosing and Treating Opioid Dependence
CASE Sam M., age 48, is in your office for the first time in more than two years. He has gained a considerable amount of weight and appears a bit sluggish, and you wonder whether he's depressed. As you take a history, Sam reminds you that he was laid off 16 months ago and had been caring for his wife, who sustained a debilitating back injury. When you saw her recently, she told you she's back to work and pain-free. So you're taken aback when Sam asks you to refill his wife's oxycodone prescription for lingering pain that often keeps her up at night.
If Sam were your patient, would you suspect opioid dependence?
Dependence on opioid analgesics and the adverse consequences associated with it have steadily increased during the past decade. Consider the following:
• Between 2004 and 2008, the number of emergency department visits related to nonmedical prescription opioid use more than doubled, rising by 111%.1
• The increasing prevalence of opioid abuse has led to a recent spike in unintentional deaths,2 with the number of lives lost to opioid analgesic overdose now exceeding that of heroin or cocaine.3
• More than 75% of opioids used for nonmedical purposes were prescribed for someone else.4
The course of opioid use is highly variable. Some people start with a legitimate medical prescription for an opioid analgesic, then continue taking it after the pain subsides. Others experiment briefly with nonmedical prescription opioids or use them intermittently without adverse effect. Some progress from prescription opioids to heroin, despite its dangers.5 Still others experience a catastrophic outcome, such as an overdose or severe accident, the first time they use opioids.6 Rapid progression from misuse of opioids to dependence is most likely to occur in vulnerable populations, such as those with concurrent mental illness, other substance use disorders, or increased sensitivity to pain.7
Understanding the terms. Before we continue, a word about terminology is in order. Misuse generally refers to the use of a medication in a manner (ie, purpose, dose, or frequency) other than its intended use, whereas drug addiction is the repeated use of a drug despite resulting harm. Here we will use opioid dependence to mean a pattern of increasing use characterized by significant impairment and distress and an inability to stop, and opioid withdrawal to reflect a constellation of symptoms, such as insomnia, nausea, diarrhea, and muscle aches, that can follow physiological dependence (though not necessarily opioid dependence). Our definitions of these terms are consistent with those of the American Psychiatric Association (APA).8 Worth noting, however, is the fact that as the APA prepares for the publication of the 5th edition of its Diagnostic and Statistical Manual of Mental Disorders, its Substance Disorder Work Group has proposed replacing the term opioid dependence with opioid use disorder to reduce the confusion associated with these definitions.9
Assessing Illicit Opioid Use: Start With a Targeted Question
Most patients who are opioid dependent do not seek treatment for it10 and are typically free of medical sequelae associated with drug addiction when they see family practitioners. The absence of self-reporting and obvious physical signs and symptoms, coupled with the increase in illicit use of prescription opioids, underscores the need for clinicians to identify patients who are abusing opioids and ensure that they get the help they need.
Screening tools. There are a number of screening tools you can use for this purpose—eg, CAGE-Adapted to Include Drugs (CAGE-AID) and Drug Abuse Screening Test (DAST)11,12—but they have not been found to be significantly better than a careful substance abuse history.13
Straightforward questions. You can start by asking, "Do you take any medications for pain?" If the answer is Yes, get the name of the drug and inquire about the frequency of use and the route, the amount typically taken, and the duration of the current use pattern. Ask specifically about opioids when taking a substance abuse history. After a question about alcohol use, you can say, "Do you use any other drugs in a serious way? Marijuana? Opioids, like Percocet, Vicodin, or Oxycontin?" Although it can be very difficult to detect opioid dependence if the patient is not forthcoming, other likely indicators of drug-seeking behavior should trigger additional questions. (See "Opioid dependence: Red Flags to Keep in Mind."14-16)
"Brief" protocols. Recent studies of Screening, Brief Intervention, and Referral to Treatment (SBIRT) programs have found that the simple, time-limited interventions they offer (visit www.samhsa.gov/prevention/sbirt/SBIRTwhitepaper.pdf to learn more) lead to a reduction in self-reported illicit opioid use.17,18 Clinicians can readily incorporate SBIRT protocols into routine practice, as an evidence-based and often reimbursable approach to substance abuse.17
Additional Steps Before Initiating Treatment
After screening and diagnostic evaluation provide evidence that a patient is opioid dependent, you can take several steps to guide him or her to the appropriate treatment.
A thorough biopsychosocial assessment covering co-occurring psychiatric illnesses, pain, psychosocial stressors contributing to opioid use, and infectious disease screening is required to gain a clear picture of the patient's situation. In every case, acute emergencies such as suicidal ideation require immediate intervention, which may include hospitalization.19
Assess the patient's desire for help. After the initial assessment, it is often helpful to categorize the patient's "stage of change" (precontemplation, contemplation, preparation, action, or maintenance)20 and to tailor your next step accordingly. A patient who denies that opioid use is a problem or who is clearly ambivalent about seeking treatment may require a conversation that uses principles of motivational interviewing—a collaborative approach that aims to evoke and strengthen personal motivation for change.21 Consider a question that encourages him or her to express reasons for change, such as: "How would you like your current situation to be different?" As almost everyone abusing opioids has thoughts about stopping, such a question may help the patient focus on specific changes.
CASE When you question Sam about his interest in oxycodone, he breaks down. He's been unable to find work or to lose the excess weight he gained during the many months he cared for his wife. He tells you that soon after his wife stopped taking the pain pills, he started taking them. At first, he took one occasionally. Then he started taking the opioids every day, and finally, whenever he awakened at night. Now, Sam says, he has no more pills, and he's nauseous, depressed, and unable to sleep—and looking to you for help.
Sam fits the criteria for opioid withdrawal as a result of physiological dependence; further questioning reveals that he also suffers from opioid dependence, and that he is receptive to treatment.
Recommending Treatment and Following Up
Several options are available for patients who, like Sam, have signs and symptoms of opioid withdrawal as a result of physiological dependence. You can provide a referral to a clinician who specializes in addiction, recommend detoxification and/or treatment in an inpatient facility, or initiate pharmacologic treatment and provide a referral to a behavioral therapist. Whatever the initial approach, most patients will ultimately be treated as outpatients, with a combination of pharmacotherapy and behavioral therapy—often, with monitoring and oversight by a primary care provider. Which approach to pursue should be guided by evidence-based recommendations (see table17,22-27) and jointly decided by provider and patient.
Medication Plays a Key Role in Recovery
Recommend medication-assisted treatment, either with an agonist (buprenorphine or methadone) or an antagonist (naltrexone), for every patient with physiological opioid dependence. The goals of pharmacotherapy are to prevent or reduce withdrawal symptoms and craving, avoid relapse, and restore to a normal state any physiological functions (eg, sleep, bowel habits) that have been disrupted by opioid use.28 When continued for at least three months, medication has been shown to improve outcomes.23,24,29 In one recent study, 49% of opioid-dependent participants who were still taking buprenorphine-naloxone at 12 weeks had successful outcomes (minimal or no opioid use), compared with 7% of those undergoing a brief buprenorphine-naloxone taper.24
There are risks associated with medication-assisted therapy, however. Those of greatest concern are a potential increase in drug interactions, the risk of diversion (a concern with both buprenorphine and methadone), and the potential for accidental overdose.2,30
Buprenorphine, a partial m-opioid receptor agonist, is a Schedule III controlled substance and can be dispensed by a pharmacist, making inpatient opioid detoxification unnecessary for many opioid-dependent patients. Clinicians who wish to prescribe buprenorphine for the treatment of opioid dependence must complete an 8-hour course, offered by the American Medical Association and the APA, among other medical groups, and obtain a Drug Enforcement Administration (code "X") license.31
Buprenorphine has a high affinity for, and a slow dissociation from, m-opioid receptors, resulting in the displacement of other opioids from the m receptor and less severe withdrawal.32 As a partial agonist, buprenorphine attenuates opioid withdrawal symptoms with a ceiling, or near-maximal, effect at 16 mg, thereby lowering the risk for overdose.33 A sublingual formulation that combines buprenorphine with naloxone, an opioid antagonist that exerts its full effect when injected but is minimally absorbed sublingually, reduces the potential for abuse of buprenorphine without interfering with its effectiveness.34
Compared with methadone, buprenorphine is less likely to interact with antiretroviral medications or to cause QTc prolongation, erectile dysfunction, or cognitive or psychomotor impairment.31,35-37 Limitations include the ceiling effect, which can be a problem for cases in which more agonist is needed; cost (approximately $12/d); and the lack of approval by the FDA for use during pregnancy.
Buprenorphine maintenance involves three phases: induction, stabilization, and maintenance.38 Induction takes place in a clinician's office at the time the patient experiences opioid withdrawal symptoms, typically 6 to 48 hours after taking the last opioid. Extended treatment improves clinical outcomes,23,24 and longer-term maintenance (of indefinite duration) is frequently required.
Naltrexone is a m-receptor antagonist, and therefore does not cause physical dependence or have agonist effects such as euphoria and sedation. As a result, it has no diversion value and may appeal to those who view opioid-agonist pharmacotherapy as simply trading one drug for another.39 Naltrexone is not a controlled substance and is not subject to the regulatory requirements that buprenorphine and methadone face.
Although agonists can be started in the first day or two after a patient decides to stop using opioids, patients must be opioid-free for at least seven days before starting naltrexone. That's because its antagonist properties will precipitate withdrawal if another opioid is present on the opioid receptors. During the seven-day "washout" period, opioid withdrawal symptoms can be treated with medications such as clonidine and dicyclomine, but such symptoms make patients especially vulnerable to relapse while waiting to start naltrexone.
Oral naltrexone's effectiveness as a treatment for opioid dependence has been limited by poor patient adherence. But a long-acting intramuscular form of the drug, approved by the FDA in 2010 and requiring once-a-month injection, mitigates this concern.40,41
Methadone is a full m-opioid agonist, administered daily at specialized clinics, as a maintenance therapy for opioid dependence. Although office-based physicians can prescribe methadone for pain, the drug can only be used for opioid dependence under the auspices of state- and federally regulated opioid treatment programs (http://findtreatment.samhsa.gov/TreatmentLocator/faces/quickSearch.jspx; a mobile phone application is also available at www.samhsa.gov/mobile/treat mentlocator.aspx).
Methadone, a Schedule III controlled substance with a half-life averaging 24 to 36 hours, requires daily dosing.42 Its slow metabolism and long half-life increase the risk for overdose.
Methadone is best for patients who are highly dependent on opioids and likely to benefit from a structured treatment environment with daily supervision (although patients who are doing well may earn take-home privileges so they don't have to come to the clinic every day).43 New patients should receive an initial dose of 30 mg or less, and a maximum first-day dose of 40 mg.44
Methadone use remains the standard of care for pregnant women being treated for opioid dependence, while studies of the effects of buprenorphine and naltrexone on a developing fetus continue. Although methadone's efficacy, particularly in lower doses, is similar to that of buprenorphine,45 its adverse effect profile is worse. Adverse effects include drug interactions, the potential for respiratory depression (especially in combination with alcohol or sedatives), QTc prolongation (which requires monitoring by ECG), sedation, and weight gain, and should be considered before methadone is selected as a maintenance pharmacotherapy.30,37,46 And, because relapse rates within 12 months of tapering off methadone have been reported to exceed 80%,47 both the clinician and the patient need to consider the likelihood of long-term, even lifelong, maintenance before initiating treatment.
Behavioral Interventions Are a Vital Part of the Picture
Studies evaluating the extent to which various types and amounts of counseling improve outcomes compared with pharmacotherapy alone have had conflicting results.24,48 Nonetheless, most clinicians consider counseling a critical component of treatment for opioid dependence and recommend, at a minimum, either individual or group counseling (various modalities have been shown to be effective) and regular attendance at a self-help group like Narcotics Anonymous. Contingency management, a type of therapy that uses prizes as incentives for desired behaviors; and family therapy, individual counseling, and community-based programs have all been found to improve outcomes.6,49
CASE You refer Sam to an addiction psychiatrist, who stabilizes him on 16 mg/d buprenorphine/naloxone as part of an outpatient treatment program. Sam is enrolled in a weekly buprenorphine stabilization group, where he gives a urine sample each week. He also begins seeing a social worker weekly for counseling and attends Narcotics Anonymous meetings two to three times a week. At a follow-up appointment with you six months later, he reports that he has been abstinent from oxycodone for six months, his sleep is improved, and he feels better about his chances of finding another job.
Your Role in Safeguarding the Patient
With the rising prevalence of opioid overdose, patient education aimed at crisis prevention is crucial as well. Warn patients of the risk for accidental overdose, often associated with relapse, stressing the importance of continuing treatment and taking their maintenance medication exactly as prescribed.
There are other steps that can be taken to safeguard patients—for example, providing naloxone rescue kits to patients and their families when appropriate. The clinician can also institute diversion and overdose prevention measures for patients taking buprenorphine or methadone—providing a lock box for take-home medication, implementing treatment contracts, and using a designated pharmacy to dispense buprenorphine, for example.26,27,50
Regular monitoring, urine drug screens, and random pill counts, in which patients are typically given 24 hours to bring in their prescribed medication so it can be counted, can also help keep patients on track. Treatment for concurrent psychiatric disorders—depression, anxiety, and personality disorders are common among patients with opioid dependence—is also likely to improve treatment outcomes.
References
1. CDC. Emergency department visits involving nonmedical use of selected prescription drugs - United States, 2004-2008. MMWR Morb Mortal Wkly Rep. 2010;59:705-709.
2. Bohnert AS, Valenstein M, Bair MJ, et al. Association between opioid prescribing patterns and opioid overdose-related deaths. JAMA. 2011;305:1315-1321.
3. Warner M, Chen LH, Makuc DM. Increase in fatal poisonings involving opioid analgesics in the United States, 1999-2006. NCHS Data Brief. 2009;22:1-8.
4. Substance Abuse and Mental Health Services Administration. Results From the 2009 National Survey on Drug Use and Health: Volume I. Summary of National Findings. Rockville, Md: SAMHSA, Office of Applied Studies; 2010. NSDUH Series H-38A, HHS publication SMA 10-4856. www.samhsa.gov/dataNSDUH/2k9NSDUH/2k9Results.htm. Accessed August 22, 2012.
5. Hser YI, Huang D, Brecht ML, et al. Contrasting trajectories of heroin, cocaine, and methamphetamine use. J Addict Dis. 2008;27:13-21.
6. Veilleux JC, Colvin PJ, Anderson J, et al. A review of opioid dependence treatment: pharmacological and psychosocial interventions to treat opioid addiction. Clin Psychol Rev. 2011;30:155-166.
7. George O, Koob GF. Individual differences in prefrontal cortex function and the transition from drug use to drug dependence. Neurosci Biobehav Rev. 2011;35:232-247.
8. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 4th ed, text rev (DSM-IV-TR). Arlington, VA: American Psychiatric Association; 2000.
9. American Psychiatric Association. R 19 opioid use disorder (2012). www.dsm5.org/ProposedRevisions/Pages/proposedrevision.aspx?rid=460. Accessed June 20, 2012.
10. Substance Abuse and Mental Health Services Administration. Results From the 2008 National Survey on Drug Use and Health: National Findings. Rockville, MD: SAMHSA, Office of Applied Studies; 2009. NSDUH Series H-36, HHS publication SMA 09-4434. www.samhsa.gov/data/nsduh/2k8nsduh/2k8Results.htm. Accessed August 22, 2012.
11. Brown RL, Rounds LA. Conjoint screening questionnaires for alcohol and other drug abuse: criterion validity in a primary care practice. Wis Med J. 1995;94:135-140.
12. Skinner HA. The drug abuse screening test. Addict Behav. 1982;7:363-371.
13. US Preventive Services Task Force. Screening for Illicit Drug Use: US Preventive Services Task Force Recommendation Statement. January 2008. www.uspreventiveservicestaskforce.org/uspstf08/druguse/drugrs.htm. Accessed May 7, 2012.
14. Gourlay D, Caplan Y, Heit H. Urine Drug Testing in Clinical Practice: Dispelling the Myths and Designing Strategies. San Francisco, Calif: California Academy of Family Physicians; 2006.
15. Jackman R, Purvis J, Mallett B. Chronic nonmalignant pain in primary care. Am Fam Physician. 2008;78:1155-1162.
16. McBane S, Weigle N. Is it time to drug test your chronic pain patient? J Fam Pract. 2010;59:628-633.
17. Madras BK, Compton WM, Avula D, et al. Screening, brief interventions, referral to treatment (SBIRT) for illicit drug and alcohol use at multiple healthcare sites: comparison at intake and 6 months later. Drug Alcohol Depend. 2009;99:280-295.
18. The InSight Project Research Group. SBIRT outcomes in Houston: final report on InSight, a hospital district-based program for patients at risk for alcohol or drug use problems. Alcohol Clin Exp Res. 2009;33:1374-1381.
19. Borges G, Walters EE, Kessler RC. Associations of substance use, abuse, and dependence with subsequent suicidal behavior. Am J Epidemiol. 2000;151:781-789.
20. Prochaska JO, DiClemente CC. Stages and processes of self-change of smoking: toward an integrative model of change. J Consult Clin Psychol. 1983;51:390-395.
21. Smedslund G, Berg RC, Hammerstrom KT, et al. Motivational interviewing for substance abuse. Cochrane Database Syst Rev. 2011;(5):CD008063.
22. Gryczynski J, Mitchell SG, Peterson TR, et al. The relationship between services delivered and substance use outcomes in New Mexico's Screening, Brief Intervention, Referral and Treatment (SBIRT) Initiative. Drug Alcohol Depend. 2011;118:152-157.
23. Woody GE, Poole SA, Subramaniam G, et al. Extended vs short-term buprenorphine-naloxone for treatment of opioid-addicted youth: a randomized trial. JAMA. 2008;300:2003-2011.
24. Weiss RD, Potter JS, Fiellin DA, et al. Adjunctive counseling during brief and extended buprenorphine-naloxone treatment for prescription opioid dependence: a 2-phase randomized controlled trial. Arch Gen Psychiatry. 2011;68:1238-1246.
25. Johansson BA, Berglund M, Lindgren A. Efficacy of maintenance treatment with naltrexone for opioid dependence: a meta-analytical review. Addiction. 2006;101:491-503.
26. Hall AJ, Logan JE, Toblin RL, et al. Patterns of abuse among unintentional pharmaceutical overdose fatalities. JAMA. 2008;300:2613-2620.
27. Zacny J, Bigelow G, Compton P, et al. College on Problems of Drug Dependence taskforce on prescription opioid non-medical use and abuse: position statement. Drug Alcohol Depend. 2003;69:215-232.
28. Kreek MJ. Rationale for maintenance pharmacotherapy of opiate dependence. Res Publ Assoc Res Nerv Ment Dis. 1992;70:205-230.
29. Mattick RP, Breen C, Kimber J, et al. Methadone maintenance therapy versus no opioid replacement therapy for opioid dependence. Cochrane Database Syst Rev. 2009;(3):CD002209.
30. McCance-Katz EF, Sullivan LE, Nallani S. Drug interactions of clinical importance among the opioids, methadone and buprenorphine, and other frequently prescribed medications: a review. Am J Addict. 2010;19:4-16.
31. Office of National Drug Control Policy Reauthorization Act of 2006 (ONDCPRA), HR 6344, 109th Cong, 2nd Sess (2006).
32. Lewis JW, Walter D. Buprenorphine—background to its development as a treatment for opiate dependence. In: Blaine JD, ed. Buprenorphine: An Alternative Treatment for Opioid Dependence. Rockville, MD: National Institute on Drug Abuse; 1992:5-11. NIDA Research Monograph, No. 121. http://archives.drugabuse.gov/pdf/monographs/121.pdf. Accessed August 22, 2012.
33. Walsh SL, Preston KL, Stitzer ML, et al. Clinical pharmacology of buprenorphine: ceiling effects at high doses. Clin Pharmacol Ther. 1994;55:569-580.
34. Alho H, Sinclair D, Vuori E, et al. Abuse liability of buprenorphine-naloxone tablets in untreated IV drug users. Drug Alcohol Depend. 2007;88:75-78.
35. Hallinan R, Byrne A, Agho K, et al. Erectile dysfunction in men receiving methadone and buprenorphine maintenance treatment. J Sex Med. 2008;5:684-692.
36. Rapeli P, Fabritius C, Alho H, et al. Methadone vs. buprenorphine/naloxone during early opioid substitution treatment: a naturalistic comparison of cognitive performance relative to healthy controls. BMC Clin Pharmacol. 2007;7:5.
37. Wedam EF, Bigelow GE, Johnson RE, et al. QT-interval effects of methadone, levomethadyl, and buprenorphine in a randomized trial. Arch Intern Med. 2007;167:2469-2475.
38. Center for Substance Abuse Treatment. Clinical Guidelines for the Use of Buprenorphine in the Treatment of Opioid Addiction. Rockville, MD: Substance Abuse and Mental Health Services Administration; 2004. Treatment Improvement Protocol (TIP) Series 40. DHHS publication SMA 04‐3939.
39. Kleber HD. Methadone maintenance 4 decades later: thousands of lives saved but still controversial. JAMA. 2008;300:2303-2305.
40. Hulse GK, Morris N, Arnold-Reed D, et al. Improving clinical outcomes in treating heroin dependence: randomized, controlled trial of oral or implant naltrexone. Arch Gen Psychiatry. 2009;66:1108-1115.
41. FDA. FDA approves injectable drug to treat opioid-dependent patients. October 12, 2010. www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/2010/ucm229109.htm. Accessed September 11, 2012.
42. Inturrisi CE, Verebely K. The levels of methadone in the plasma in methadone maintenance. Clin Pharmacol Ther. 1972;13(5 pt 1):633-637.
43. Stitzer M, Bigelow G, Lawrence C, et al. Medication take-home as a reinforcer in a methadone maintenance program. Addict Behav. 1977;2:9-14.
44. Code of Federal Regulations. Title 42.8.12. Federal Opioid Treatment Standards. October 2010.
45. Johnson RE. Chutuape MA, Strain EC, et al. A comparison of levomethadyl acetate, buprenorphine, and methadone for opioid dependence. N Engl J Med. 2000;343:1290-1297.
46. Krantz MJ, Martin J, Stimmel B, et al. QTc interval screening in methadone treatment. Ann Intern Med. 2009;150:387-395.
47. Ball JC, Lange WR, Myers CP, et al. Reducing the risk of AIDS through methadone maintenance treatment. J Health Soc Behav. 1988;29:214-226.
48. Fiellin DA, Pantalon MV, Chawarski MC, et al. Counseling plus buprenorphine-naloxone maintenance therapy for opioid dependence. N Engl J Med. 2006;355:365-374.
49. Defulio A, Everly JJ, Leoutsakos JM, et al. Employment-based reinforcement of adherence to an FDA approved extended release formulation of naltrexone in opioid-dependent adults: a randomized controlled trial. Drug Alcohol Depend. 2012;120:48-54.
50. Savage SR. Management of opioid medications in patients with chronic pain and risk of substance misuse. Curr Psychiatry Rep. 2009;11:377-384.
This article was originally published in the Journal of Family Practice. 2012;61:588-597.
CASE Sam M., age 48, is in your office for the first time in more than two years. He has gained a considerable amount of weight and appears a bit sluggish, and you wonder whether he's depressed. As you take a history, Sam reminds you that he was laid off 16 months ago and had been caring for his wife, who sustained a debilitating back injury. When you saw her recently, she told you she's back to work and pain-free. So you're taken aback when Sam asks you to refill his wife's oxycodone prescription for lingering pain that often keeps her up at night.
If Sam were your patient, would you suspect opioid dependence?
Dependence on opioid analgesics and the adverse consequences associated with it have steadily increased during the past decade. Consider the following:
• Between 2004 and 2008, the number of emergency department visits related to nonmedical prescription opioid use more than doubled, rising by 111%.1
• The increasing prevalence of opioid abuse has led to a recent spike in unintentional deaths,2 with the number of lives lost to opioid analgesic overdose now exceeding that of heroin or cocaine.3
• More than 75% of opioids used for nonmedical purposes were prescribed for someone else.4
The course of opioid use is highly variable. Some people start with a legitimate medical prescription for an opioid analgesic, then continue taking it after the pain subsides. Others experiment briefly with nonmedical prescription opioids or use them intermittently without adverse effect. Some progress from prescription opioids to heroin, despite its dangers.5 Still others experience a catastrophic outcome, such as an overdose or severe accident, the first time they use opioids.6 Rapid progression from misuse of opioids to dependence is most likely to occur in vulnerable populations, such as those with concurrent mental illness, other substance use disorders, or increased sensitivity to pain.7
Understanding the terms. Before we continue, a word about terminology is in order. Misuse generally refers to the use of a medication in a manner (ie, purpose, dose, or frequency) other than its intended use, whereas drug addiction is the repeated use of a drug despite resulting harm. Here we will use opioid dependence to mean a pattern of increasing use characterized by significant impairment and distress and an inability to stop, and opioid withdrawal to reflect a constellation of symptoms, such as insomnia, nausea, diarrhea, and muscle aches, that can follow physiological dependence (though not necessarily opioid dependence). Our definitions of these terms are consistent with those of the American Psychiatric Association (APA).8 Worth noting, however, is the fact that as the APA prepares for the publication of the 5th edition of its Diagnostic and Statistical Manual of Mental Disorders, its Substance Disorder Work Group has proposed replacing the term opioid dependence with opioid use disorder to reduce the confusion associated with these definitions.9
Assessing Illicit Opioid Use: Start With a Targeted Question
Most patients who are opioid dependent do not seek treatment for it10 and are typically free of medical sequelae associated with drug addiction when they see family practitioners. The absence of self-reporting and obvious physical signs and symptoms, coupled with the increase in illicit use of prescription opioids, underscores the need for clinicians to identify patients who are abusing opioids and ensure that they get the help they need.
Screening tools. There are a number of screening tools you can use for this purpose—eg, CAGE-Adapted to Include Drugs (CAGE-AID) and Drug Abuse Screening Test (DAST)11,12—but they have not been found to be significantly better than a careful substance abuse history.13
Straightforward questions. You can start by asking, "Do you take any medications for pain?" If the answer is Yes, get the name of the drug and inquire about the frequency of use and the route, the amount typically taken, and the duration of the current use pattern. Ask specifically about opioids when taking a substance abuse history. After a question about alcohol use, you can say, "Do you use any other drugs in a serious way? Marijuana? Opioids, like Percocet, Vicodin, or Oxycontin?" Although it can be very difficult to detect opioid dependence if the patient is not forthcoming, other likely indicators of drug-seeking behavior should trigger additional questions. (See "Opioid dependence: Red Flags to Keep in Mind."14-16)
"Brief" protocols. Recent studies of Screening, Brief Intervention, and Referral to Treatment (SBIRT) programs have found that the simple, time-limited interventions they offer (visit www.samhsa.gov/prevention/sbirt/SBIRTwhitepaper.pdf to learn more) lead to a reduction in self-reported illicit opioid use.17,18 Clinicians can readily incorporate SBIRT protocols into routine practice, as an evidence-based and often reimbursable approach to substance abuse.17
Additional Steps Before Initiating Treatment
After screening and diagnostic evaluation provide evidence that a patient is opioid dependent, you can take several steps to guide him or her to the appropriate treatment.
A thorough biopsychosocial assessment covering co-occurring psychiatric illnesses, pain, psychosocial stressors contributing to opioid use, and infectious disease screening is required to gain a clear picture of the patient's situation. In every case, acute emergencies such as suicidal ideation require immediate intervention, which may include hospitalization.19
Assess the patient's desire for help. After the initial assessment, it is often helpful to categorize the patient's "stage of change" (precontemplation, contemplation, preparation, action, or maintenance)20 and to tailor your next step accordingly. A patient who denies that opioid use is a problem or who is clearly ambivalent about seeking treatment may require a conversation that uses principles of motivational interviewing—a collaborative approach that aims to evoke and strengthen personal motivation for change.21 Consider a question that encourages him or her to express reasons for change, such as: "How would you like your current situation to be different?" As almost everyone abusing opioids has thoughts about stopping, such a question may help the patient focus on specific changes.
CASE When you question Sam about his interest in oxycodone, he breaks down. He's been unable to find work or to lose the excess weight he gained during the many months he cared for his wife. He tells you that soon after his wife stopped taking the pain pills, he started taking them. At first, he took one occasionally. Then he started taking the opioids every day, and finally, whenever he awakened at night. Now, Sam says, he has no more pills, and he's nauseous, depressed, and unable to sleep—and looking to you for help.
Sam fits the criteria for opioid withdrawal as a result of physiological dependence; further questioning reveals that he also suffers from opioid dependence, and that he is receptive to treatment.
Recommending Treatment and Following Up
Several options are available for patients who, like Sam, have signs and symptoms of opioid withdrawal as a result of physiological dependence. You can provide a referral to a clinician who specializes in addiction, recommend detoxification and/or treatment in an inpatient facility, or initiate pharmacologic treatment and provide a referral to a behavioral therapist. Whatever the initial approach, most patients will ultimately be treated as outpatients, with a combination of pharmacotherapy and behavioral therapy—often, with monitoring and oversight by a primary care provider. Which approach to pursue should be guided by evidence-based recommendations (see table17,22-27) and jointly decided by provider and patient.
Medication Plays a Key Role in Recovery
Recommend medication-assisted treatment, either with an agonist (buprenorphine or methadone) or an antagonist (naltrexone), for every patient with physiological opioid dependence. The goals of pharmacotherapy are to prevent or reduce withdrawal symptoms and craving, avoid relapse, and restore to a normal state any physiological functions (eg, sleep, bowel habits) that have been disrupted by opioid use.28 When continued for at least three months, medication has been shown to improve outcomes.23,24,29 In one recent study, 49% of opioid-dependent participants who were still taking buprenorphine-naloxone at 12 weeks had successful outcomes (minimal or no opioid use), compared with 7% of those undergoing a brief buprenorphine-naloxone taper.24
There are risks associated with medication-assisted therapy, however. Those of greatest concern are a potential increase in drug interactions, the risk of diversion (a concern with both buprenorphine and methadone), and the potential for accidental overdose.2,30
Buprenorphine, a partial m-opioid receptor agonist, is a Schedule III controlled substance and can be dispensed by a pharmacist, making inpatient opioid detoxification unnecessary for many opioid-dependent patients. Clinicians who wish to prescribe buprenorphine for the treatment of opioid dependence must complete an 8-hour course, offered by the American Medical Association and the APA, among other medical groups, and obtain a Drug Enforcement Administration (code "X") license.31
Buprenorphine has a high affinity for, and a slow dissociation from, m-opioid receptors, resulting in the displacement of other opioids from the m receptor and less severe withdrawal.32 As a partial agonist, buprenorphine attenuates opioid withdrawal symptoms with a ceiling, or near-maximal, effect at 16 mg, thereby lowering the risk for overdose.33 A sublingual formulation that combines buprenorphine with naloxone, an opioid antagonist that exerts its full effect when injected but is minimally absorbed sublingually, reduces the potential for abuse of buprenorphine without interfering with its effectiveness.34
Compared with methadone, buprenorphine is less likely to interact with antiretroviral medications or to cause QTc prolongation, erectile dysfunction, or cognitive or psychomotor impairment.31,35-37 Limitations include the ceiling effect, which can be a problem for cases in which more agonist is needed; cost (approximately $12/d); and the lack of approval by the FDA for use during pregnancy.
Buprenorphine maintenance involves three phases: induction, stabilization, and maintenance.38 Induction takes place in a clinician's office at the time the patient experiences opioid withdrawal symptoms, typically 6 to 48 hours after taking the last opioid. Extended treatment improves clinical outcomes,23,24 and longer-term maintenance (of indefinite duration) is frequently required.
Naltrexone is a m-receptor antagonist, and therefore does not cause physical dependence or have agonist effects such as euphoria and sedation. As a result, it has no diversion value and may appeal to those who view opioid-agonist pharmacotherapy as simply trading one drug for another.39 Naltrexone is not a controlled substance and is not subject to the regulatory requirements that buprenorphine and methadone face.
Although agonists can be started in the first day or two after a patient decides to stop using opioids, patients must be opioid-free for at least seven days before starting naltrexone. That's because its antagonist properties will precipitate withdrawal if another opioid is present on the opioid receptors. During the seven-day "washout" period, opioid withdrawal symptoms can be treated with medications such as clonidine and dicyclomine, but such symptoms make patients especially vulnerable to relapse while waiting to start naltrexone.
Oral naltrexone's effectiveness as a treatment for opioid dependence has been limited by poor patient adherence. But a long-acting intramuscular form of the drug, approved by the FDA in 2010 and requiring once-a-month injection, mitigates this concern.40,41
Methadone is a full m-opioid agonist, administered daily at specialized clinics, as a maintenance therapy for opioid dependence. Although office-based physicians can prescribe methadone for pain, the drug can only be used for opioid dependence under the auspices of state- and federally regulated opioid treatment programs (http://findtreatment.samhsa.gov/TreatmentLocator/faces/quickSearch.jspx; a mobile phone application is also available at www.samhsa.gov/mobile/treat mentlocator.aspx).
Methadone, a Schedule III controlled substance with a half-life averaging 24 to 36 hours, requires daily dosing.42 Its slow metabolism and long half-life increase the risk for overdose.
Methadone is best for patients who are highly dependent on opioids and likely to benefit from a structured treatment environment with daily supervision (although patients who are doing well may earn take-home privileges so they don't have to come to the clinic every day).43 New patients should receive an initial dose of 30 mg or less, and a maximum first-day dose of 40 mg.44
Methadone use remains the standard of care for pregnant women being treated for opioid dependence, while studies of the effects of buprenorphine and naltrexone on a developing fetus continue. Although methadone's efficacy, particularly in lower doses, is similar to that of buprenorphine,45 its adverse effect profile is worse. Adverse effects include drug interactions, the potential for respiratory depression (especially in combination with alcohol or sedatives), QTc prolongation (which requires monitoring by ECG), sedation, and weight gain, and should be considered before methadone is selected as a maintenance pharmacotherapy.30,37,46 And, because relapse rates within 12 months of tapering off methadone have been reported to exceed 80%,47 both the clinician and the patient need to consider the likelihood of long-term, even lifelong, maintenance before initiating treatment.
Behavioral Interventions Are a Vital Part of the Picture
Studies evaluating the extent to which various types and amounts of counseling improve outcomes compared with pharmacotherapy alone have had conflicting results.24,48 Nonetheless, most clinicians consider counseling a critical component of treatment for opioid dependence and recommend, at a minimum, either individual or group counseling (various modalities have been shown to be effective) and regular attendance at a self-help group like Narcotics Anonymous. Contingency management, a type of therapy that uses prizes as incentives for desired behaviors; and family therapy, individual counseling, and community-based programs have all been found to improve outcomes.6,49
CASE You refer Sam to an addiction psychiatrist, who stabilizes him on 16 mg/d buprenorphine/naloxone as part of an outpatient treatment program. Sam is enrolled in a weekly buprenorphine stabilization group, where he gives a urine sample each week. He also begins seeing a social worker weekly for counseling and attends Narcotics Anonymous meetings two to three times a week. At a follow-up appointment with you six months later, he reports that he has been abstinent from oxycodone for six months, his sleep is improved, and he feels better about his chances of finding another job.
Your Role in Safeguarding the Patient
With the rising prevalence of opioid overdose, patient education aimed at crisis prevention is crucial as well. Warn patients of the risk for accidental overdose, often associated with relapse, stressing the importance of continuing treatment and taking their maintenance medication exactly as prescribed.
There are other steps that can be taken to safeguard patients—for example, providing naloxone rescue kits to patients and their families when appropriate. The clinician can also institute diversion and overdose prevention measures for patients taking buprenorphine or methadone—providing a lock box for take-home medication, implementing treatment contracts, and using a designated pharmacy to dispense buprenorphine, for example.26,27,50
Regular monitoring, urine drug screens, and random pill counts, in which patients are typically given 24 hours to bring in their prescribed medication so it can be counted, can also help keep patients on track. Treatment for concurrent psychiatric disorders—depression, anxiety, and personality disorders are common among patients with opioid dependence—is also likely to improve treatment outcomes.
References
1. CDC. Emergency department visits involving nonmedical use of selected prescription drugs - United States, 2004-2008. MMWR Morb Mortal Wkly Rep. 2010;59:705-709.
2. Bohnert AS, Valenstein M, Bair MJ, et al. Association between opioid prescribing patterns and opioid overdose-related deaths. JAMA. 2011;305:1315-1321.
3. Warner M, Chen LH, Makuc DM. Increase in fatal poisonings involving opioid analgesics in the United States, 1999-2006. NCHS Data Brief. 2009;22:1-8.
4. Substance Abuse and Mental Health Services Administration. Results From the 2009 National Survey on Drug Use and Health: Volume I. Summary of National Findings. Rockville, Md: SAMHSA, Office of Applied Studies; 2010. NSDUH Series H-38A, HHS publication SMA 10-4856. www.samhsa.gov/dataNSDUH/2k9NSDUH/2k9Results.htm. Accessed August 22, 2012.
5. Hser YI, Huang D, Brecht ML, et al. Contrasting trajectories of heroin, cocaine, and methamphetamine use. J Addict Dis. 2008;27:13-21.
6. Veilleux JC, Colvin PJ, Anderson J, et al. A review of opioid dependence treatment: pharmacological and psychosocial interventions to treat opioid addiction. Clin Psychol Rev. 2011;30:155-166.
7. George O, Koob GF. Individual differences in prefrontal cortex function and the transition from drug use to drug dependence. Neurosci Biobehav Rev. 2011;35:232-247.
8. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 4th ed, text rev (DSM-IV-TR). Arlington, VA: American Psychiatric Association; 2000.
9. American Psychiatric Association. R 19 opioid use disorder (2012). www.dsm5.org/ProposedRevisions/Pages/proposedrevision.aspx?rid=460. Accessed June 20, 2012.
10. Substance Abuse and Mental Health Services Administration. Results From the 2008 National Survey on Drug Use and Health: National Findings. Rockville, MD: SAMHSA, Office of Applied Studies; 2009. NSDUH Series H-36, HHS publication SMA 09-4434. www.samhsa.gov/data/nsduh/2k8nsduh/2k8Results.htm. Accessed August 22, 2012.
11. Brown RL, Rounds LA. Conjoint screening questionnaires for alcohol and other drug abuse: criterion validity in a primary care practice. Wis Med J. 1995;94:135-140.
12. Skinner HA. The drug abuse screening test. Addict Behav. 1982;7:363-371.
13. US Preventive Services Task Force. Screening for Illicit Drug Use: US Preventive Services Task Force Recommendation Statement. January 2008. www.uspreventiveservicestaskforce.org/uspstf08/druguse/drugrs.htm. Accessed May 7, 2012.
14. Gourlay D, Caplan Y, Heit H. Urine Drug Testing in Clinical Practice: Dispelling the Myths and Designing Strategies. San Francisco, Calif: California Academy of Family Physicians; 2006.
15. Jackman R, Purvis J, Mallett B. Chronic nonmalignant pain in primary care. Am Fam Physician. 2008;78:1155-1162.
16. McBane S, Weigle N. Is it time to drug test your chronic pain patient? J Fam Pract. 2010;59:628-633.
17. Madras BK, Compton WM, Avula D, et al. Screening, brief interventions, referral to treatment (SBIRT) for illicit drug and alcohol use at multiple healthcare sites: comparison at intake and 6 months later. Drug Alcohol Depend. 2009;99:280-295.
18. The InSight Project Research Group. SBIRT outcomes in Houston: final report on InSight, a hospital district-based program for patients at risk for alcohol or drug use problems. Alcohol Clin Exp Res. 2009;33:1374-1381.
19. Borges G, Walters EE, Kessler RC. Associations of substance use, abuse, and dependence with subsequent suicidal behavior. Am J Epidemiol. 2000;151:781-789.
20. Prochaska JO, DiClemente CC. Stages and processes of self-change of smoking: toward an integrative model of change. J Consult Clin Psychol. 1983;51:390-395.
21. Smedslund G, Berg RC, Hammerstrom KT, et al. Motivational interviewing for substance abuse. Cochrane Database Syst Rev. 2011;(5):CD008063.
22. Gryczynski J, Mitchell SG, Peterson TR, et al. The relationship between services delivered and substance use outcomes in New Mexico's Screening, Brief Intervention, Referral and Treatment (SBIRT) Initiative. Drug Alcohol Depend. 2011;118:152-157.
23. Woody GE, Poole SA, Subramaniam G, et al. Extended vs short-term buprenorphine-naloxone for treatment of opioid-addicted youth: a randomized trial. JAMA. 2008;300:2003-2011.
24. Weiss RD, Potter JS, Fiellin DA, et al. Adjunctive counseling during brief and extended buprenorphine-naloxone treatment for prescription opioid dependence: a 2-phase randomized controlled trial. Arch Gen Psychiatry. 2011;68:1238-1246.
25. Johansson BA, Berglund M, Lindgren A. Efficacy of maintenance treatment with naltrexone for opioid dependence: a meta-analytical review. Addiction. 2006;101:491-503.
26. Hall AJ, Logan JE, Toblin RL, et al. Patterns of abuse among unintentional pharmaceutical overdose fatalities. JAMA. 2008;300:2613-2620.
27. Zacny J, Bigelow G, Compton P, et al. College on Problems of Drug Dependence taskforce on prescription opioid non-medical use and abuse: position statement. Drug Alcohol Depend. 2003;69:215-232.
28. Kreek MJ. Rationale for maintenance pharmacotherapy of opiate dependence. Res Publ Assoc Res Nerv Ment Dis. 1992;70:205-230.
29. Mattick RP, Breen C, Kimber J, et al. Methadone maintenance therapy versus no opioid replacement therapy for opioid dependence. Cochrane Database Syst Rev. 2009;(3):CD002209.
30. McCance-Katz EF, Sullivan LE, Nallani S. Drug interactions of clinical importance among the opioids, methadone and buprenorphine, and other frequently prescribed medications: a review. Am J Addict. 2010;19:4-16.
31. Office of National Drug Control Policy Reauthorization Act of 2006 (ONDCPRA), HR 6344, 109th Cong, 2nd Sess (2006).
32. Lewis JW, Walter D. Buprenorphine—background to its development as a treatment for opiate dependence. In: Blaine JD, ed. Buprenorphine: An Alternative Treatment for Opioid Dependence. Rockville, MD: National Institute on Drug Abuse; 1992:5-11. NIDA Research Monograph, No. 121. http://archives.drugabuse.gov/pdf/monographs/121.pdf. Accessed August 22, 2012.
33. Walsh SL, Preston KL, Stitzer ML, et al. Clinical pharmacology of buprenorphine: ceiling effects at high doses. Clin Pharmacol Ther. 1994;55:569-580.
34. Alho H, Sinclair D, Vuori E, et al. Abuse liability of buprenorphine-naloxone tablets in untreated IV drug users. Drug Alcohol Depend. 2007;88:75-78.
35. Hallinan R, Byrne A, Agho K, et al. Erectile dysfunction in men receiving methadone and buprenorphine maintenance treatment. J Sex Med. 2008;5:684-692.
36. Rapeli P, Fabritius C, Alho H, et al. Methadone vs. buprenorphine/naloxone during early opioid substitution treatment: a naturalistic comparison of cognitive performance relative to healthy controls. BMC Clin Pharmacol. 2007;7:5.
37. Wedam EF, Bigelow GE, Johnson RE, et al. QT-interval effects of methadone, levomethadyl, and buprenorphine in a randomized trial. Arch Intern Med. 2007;167:2469-2475.
38. Center for Substance Abuse Treatment. Clinical Guidelines for the Use of Buprenorphine in the Treatment of Opioid Addiction. Rockville, MD: Substance Abuse and Mental Health Services Administration; 2004. Treatment Improvement Protocol (TIP) Series 40. DHHS publication SMA 04‐3939.
39. Kleber HD. Methadone maintenance 4 decades later: thousands of lives saved but still controversial. JAMA. 2008;300:2303-2305.
40. Hulse GK, Morris N, Arnold-Reed D, et al. Improving clinical outcomes in treating heroin dependence: randomized, controlled trial of oral or implant naltrexone. Arch Gen Psychiatry. 2009;66:1108-1115.
41. FDA. FDA approves injectable drug to treat opioid-dependent patients. October 12, 2010. www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/2010/ucm229109.htm. Accessed September 11, 2012.
42. Inturrisi CE, Verebely K. The levels of methadone in the plasma in methadone maintenance. Clin Pharmacol Ther. 1972;13(5 pt 1):633-637.
43. Stitzer M, Bigelow G, Lawrence C, et al. Medication take-home as a reinforcer in a methadone maintenance program. Addict Behav. 1977;2:9-14.
44. Code of Federal Regulations. Title 42.8.12. Federal Opioid Treatment Standards. October 2010.
45. Johnson RE. Chutuape MA, Strain EC, et al. A comparison of levomethadyl acetate, buprenorphine, and methadone for opioid dependence. N Engl J Med. 2000;343:1290-1297.
46. Krantz MJ, Martin J, Stimmel B, et al. QTc interval screening in methadone treatment. Ann Intern Med. 2009;150:387-395.
47. Ball JC, Lange WR, Myers CP, et al. Reducing the risk of AIDS through methadone maintenance treatment. J Health Soc Behav. 1988;29:214-226.
48. Fiellin DA, Pantalon MV, Chawarski MC, et al. Counseling plus buprenorphine-naloxone maintenance therapy for opioid dependence. N Engl J Med. 2006;355:365-374.
49. Defulio A, Everly JJ, Leoutsakos JM, et al. Employment-based reinforcement of adherence to an FDA approved extended release formulation of naltrexone in opioid-dependent adults: a randomized controlled trial. Drug Alcohol Depend. 2012;120:48-54.
50. Savage SR. Management of opioid medications in patients with chronic pain and risk of substance misuse. Curr Psychiatry Rep. 2009;11:377-384.
This article was originally published in the Journal of Family Practice. 2012;61:588-597.
CASE Sam M., age 48, is in your office for the first time in more than two years. He has gained a considerable amount of weight and appears a bit sluggish, and you wonder whether he's depressed. As you take a history, Sam reminds you that he was laid off 16 months ago and had been caring for his wife, who sustained a debilitating back injury. When you saw her recently, she told you she's back to work and pain-free. So you're taken aback when Sam asks you to refill his wife's oxycodone prescription for lingering pain that often keeps her up at night.
If Sam were your patient, would you suspect opioid dependence?
Dependence on opioid analgesics and the adverse consequences associated with it have steadily increased during the past decade. Consider the following:
• Between 2004 and 2008, the number of emergency department visits related to nonmedical prescription opioid use more than doubled, rising by 111%.1
• The increasing prevalence of opioid abuse has led to a recent spike in unintentional deaths,2 with the number of lives lost to opioid analgesic overdose now exceeding that of heroin or cocaine.3
• More than 75% of opioids used for nonmedical purposes were prescribed for someone else.4
The course of opioid use is highly variable. Some people start with a legitimate medical prescription for an opioid analgesic, then continue taking it after the pain subsides. Others experiment briefly with nonmedical prescription opioids or use them intermittently without adverse effect. Some progress from prescription opioids to heroin, despite its dangers.5 Still others experience a catastrophic outcome, such as an overdose or severe accident, the first time they use opioids.6 Rapid progression from misuse of opioids to dependence is most likely to occur in vulnerable populations, such as those with concurrent mental illness, other substance use disorders, or increased sensitivity to pain.7
Understanding the terms. Before we continue, a word about terminology is in order. Misuse generally refers to the use of a medication in a manner (ie, purpose, dose, or frequency) other than its intended use, whereas drug addiction is the repeated use of a drug despite resulting harm. Here we will use opioid dependence to mean a pattern of increasing use characterized by significant impairment and distress and an inability to stop, and opioid withdrawal to reflect a constellation of symptoms, such as insomnia, nausea, diarrhea, and muscle aches, that can follow physiological dependence (though not necessarily opioid dependence). Our definitions of these terms are consistent with those of the American Psychiatric Association (APA).8 Worth noting, however, is the fact that as the APA prepares for the publication of the 5th edition of its Diagnostic and Statistical Manual of Mental Disorders, its Substance Disorder Work Group has proposed replacing the term opioid dependence with opioid use disorder to reduce the confusion associated with these definitions.9
Assessing Illicit Opioid Use: Start With a Targeted Question
Most patients who are opioid dependent do not seek treatment for it10 and are typically free of medical sequelae associated with drug addiction when they see family practitioners. The absence of self-reporting and obvious physical signs and symptoms, coupled with the increase in illicit use of prescription opioids, underscores the need for clinicians to identify patients who are abusing opioids and ensure that they get the help they need.
Screening tools. There are a number of screening tools you can use for this purpose—eg, CAGE-Adapted to Include Drugs (CAGE-AID) and Drug Abuse Screening Test (DAST)11,12—but they have not been found to be significantly better than a careful substance abuse history.13
Straightforward questions. You can start by asking, "Do you take any medications for pain?" If the answer is Yes, get the name of the drug and inquire about the frequency of use and the route, the amount typically taken, and the duration of the current use pattern. Ask specifically about opioids when taking a substance abuse history. After a question about alcohol use, you can say, "Do you use any other drugs in a serious way? Marijuana? Opioids, like Percocet, Vicodin, or Oxycontin?" Although it can be very difficult to detect opioid dependence if the patient is not forthcoming, other likely indicators of drug-seeking behavior should trigger additional questions. (See "Opioid dependence: Red Flags to Keep in Mind."14-16)
"Brief" protocols. Recent studies of Screening, Brief Intervention, and Referral to Treatment (SBIRT) programs have found that the simple, time-limited interventions they offer (visit www.samhsa.gov/prevention/sbirt/SBIRTwhitepaper.pdf to learn more) lead to a reduction in self-reported illicit opioid use.17,18 Clinicians can readily incorporate SBIRT protocols into routine practice, as an evidence-based and often reimbursable approach to substance abuse.17
Additional Steps Before Initiating Treatment
After screening and diagnostic evaluation provide evidence that a patient is opioid dependent, you can take several steps to guide him or her to the appropriate treatment.
A thorough biopsychosocial assessment covering co-occurring psychiatric illnesses, pain, psychosocial stressors contributing to opioid use, and infectious disease screening is required to gain a clear picture of the patient's situation. In every case, acute emergencies such as suicidal ideation require immediate intervention, which may include hospitalization.19
Assess the patient's desire for help. After the initial assessment, it is often helpful to categorize the patient's "stage of change" (precontemplation, contemplation, preparation, action, or maintenance)20 and to tailor your next step accordingly. A patient who denies that opioid use is a problem or who is clearly ambivalent about seeking treatment may require a conversation that uses principles of motivational interviewing—a collaborative approach that aims to evoke and strengthen personal motivation for change.21 Consider a question that encourages him or her to express reasons for change, such as: "How would you like your current situation to be different?" As almost everyone abusing opioids has thoughts about stopping, such a question may help the patient focus on specific changes.
CASE When you question Sam about his interest in oxycodone, he breaks down. He's been unable to find work or to lose the excess weight he gained during the many months he cared for his wife. He tells you that soon after his wife stopped taking the pain pills, he started taking them. At first, he took one occasionally. Then he started taking the opioids every day, and finally, whenever he awakened at night. Now, Sam says, he has no more pills, and he's nauseous, depressed, and unable to sleep—and looking to you for help.
Sam fits the criteria for opioid withdrawal as a result of physiological dependence; further questioning reveals that he also suffers from opioid dependence, and that he is receptive to treatment.
Recommending Treatment and Following Up
Several options are available for patients who, like Sam, have signs and symptoms of opioid withdrawal as a result of physiological dependence. You can provide a referral to a clinician who specializes in addiction, recommend detoxification and/or treatment in an inpatient facility, or initiate pharmacologic treatment and provide a referral to a behavioral therapist. Whatever the initial approach, most patients will ultimately be treated as outpatients, with a combination of pharmacotherapy and behavioral therapy—often, with monitoring and oversight by a primary care provider. Which approach to pursue should be guided by evidence-based recommendations (see table17,22-27) and jointly decided by provider and patient.
Medication Plays a Key Role in Recovery
Recommend medication-assisted treatment, either with an agonist (buprenorphine or methadone) or an antagonist (naltrexone), for every patient with physiological opioid dependence. The goals of pharmacotherapy are to prevent or reduce withdrawal symptoms and craving, avoid relapse, and restore to a normal state any physiological functions (eg, sleep, bowel habits) that have been disrupted by opioid use.28 When continued for at least three months, medication has been shown to improve outcomes.23,24,29 In one recent study, 49% of opioid-dependent participants who were still taking buprenorphine-naloxone at 12 weeks had successful outcomes (minimal or no opioid use), compared with 7% of those undergoing a brief buprenorphine-naloxone taper.24
There are risks associated with medication-assisted therapy, however. Those of greatest concern are a potential increase in drug interactions, the risk of diversion (a concern with both buprenorphine and methadone), and the potential for accidental overdose.2,30
Buprenorphine, a partial m-opioid receptor agonist, is a Schedule III controlled substance and can be dispensed by a pharmacist, making inpatient opioid detoxification unnecessary for many opioid-dependent patients. Clinicians who wish to prescribe buprenorphine for the treatment of opioid dependence must complete an 8-hour course, offered by the American Medical Association and the APA, among other medical groups, and obtain a Drug Enforcement Administration (code "X") license.31
Buprenorphine has a high affinity for, and a slow dissociation from, m-opioid receptors, resulting in the displacement of other opioids from the m receptor and less severe withdrawal.32 As a partial agonist, buprenorphine attenuates opioid withdrawal symptoms with a ceiling, or near-maximal, effect at 16 mg, thereby lowering the risk for overdose.33 A sublingual formulation that combines buprenorphine with naloxone, an opioid antagonist that exerts its full effect when injected but is minimally absorbed sublingually, reduces the potential for abuse of buprenorphine without interfering with its effectiveness.34
Compared with methadone, buprenorphine is less likely to interact with antiretroviral medications or to cause QTc prolongation, erectile dysfunction, or cognitive or psychomotor impairment.31,35-37 Limitations include the ceiling effect, which can be a problem for cases in which more agonist is needed; cost (approximately $12/d); and the lack of approval by the FDA for use during pregnancy.
Buprenorphine maintenance involves three phases: induction, stabilization, and maintenance.38 Induction takes place in a clinician's office at the time the patient experiences opioid withdrawal symptoms, typically 6 to 48 hours after taking the last opioid. Extended treatment improves clinical outcomes,23,24 and longer-term maintenance (of indefinite duration) is frequently required.
Naltrexone is a m-receptor antagonist, and therefore does not cause physical dependence or have agonist effects such as euphoria and sedation. As a result, it has no diversion value and may appeal to those who view opioid-agonist pharmacotherapy as simply trading one drug for another.39 Naltrexone is not a controlled substance and is not subject to the regulatory requirements that buprenorphine and methadone face.
Although agonists can be started in the first day or two after a patient decides to stop using opioids, patients must be opioid-free for at least seven days before starting naltrexone. That's because its antagonist properties will precipitate withdrawal if another opioid is present on the opioid receptors. During the seven-day "washout" period, opioid withdrawal symptoms can be treated with medications such as clonidine and dicyclomine, but such symptoms make patients especially vulnerable to relapse while waiting to start naltrexone.
Oral naltrexone's effectiveness as a treatment for opioid dependence has been limited by poor patient adherence. But a long-acting intramuscular form of the drug, approved by the FDA in 2010 and requiring once-a-month injection, mitigates this concern.40,41
Methadone is a full m-opioid agonist, administered daily at specialized clinics, as a maintenance therapy for opioid dependence. Although office-based physicians can prescribe methadone for pain, the drug can only be used for opioid dependence under the auspices of state- and federally regulated opioid treatment programs (http://findtreatment.samhsa.gov/TreatmentLocator/faces/quickSearch.jspx; a mobile phone application is also available at www.samhsa.gov/mobile/treat mentlocator.aspx).
Methadone, a Schedule III controlled substance with a half-life averaging 24 to 36 hours, requires daily dosing.42 Its slow metabolism and long half-life increase the risk for overdose.
Methadone is best for patients who are highly dependent on opioids and likely to benefit from a structured treatment environment with daily supervision (although patients who are doing well may earn take-home privileges so they don't have to come to the clinic every day).43 New patients should receive an initial dose of 30 mg or less, and a maximum first-day dose of 40 mg.44
Methadone use remains the standard of care for pregnant women being treated for opioid dependence, while studies of the effects of buprenorphine and naltrexone on a developing fetus continue. Although methadone's efficacy, particularly in lower doses, is similar to that of buprenorphine,45 its adverse effect profile is worse. Adverse effects include drug interactions, the potential for respiratory depression (especially in combination with alcohol or sedatives), QTc prolongation (which requires monitoring by ECG), sedation, and weight gain, and should be considered before methadone is selected as a maintenance pharmacotherapy.30,37,46 And, because relapse rates within 12 months of tapering off methadone have been reported to exceed 80%,47 both the clinician and the patient need to consider the likelihood of long-term, even lifelong, maintenance before initiating treatment.
Behavioral Interventions Are a Vital Part of the Picture
Studies evaluating the extent to which various types and amounts of counseling improve outcomes compared with pharmacotherapy alone have had conflicting results.24,48 Nonetheless, most clinicians consider counseling a critical component of treatment for opioid dependence and recommend, at a minimum, either individual or group counseling (various modalities have been shown to be effective) and regular attendance at a self-help group like Narcotics Anonymous. Contingency management, a type of therapy that uses prizes as incentives for desired behaviors; and family therapy, individual counseling, and community-based programs have all been found to improve outcomes.6,49
CASE You refer Sam to an addiction psychiatrist, who stabilizes him on 16 mg/d buprenorphine/naloxone as part of an outpatient treatment program. Sam is enrolled in a weekly buprenorphine stabilization group, where he gives a urine sample each week. He also begins seeing a social worker weekly for counseling and attends Narcotics Anonymous meetings two to three times a week. At a follow-up appointment with you six months later, he reports that he has been abstinent from oxycodone for six months, his sleep is improved, and he feels better about his chances of finding another job.
Your Role in Safeguarding the Patient
With the rising prevalence of opioid overdose, patient education aimed at crisis prevention is crucial as well. Warn patients of the risk for accidental overdose, often associated with relapse, stressing the importance of continuing treatment and taking their maintenance medication exactly as prescribed.
There are other steps that can be taken to safeguard patients—for example, providing naloxone rescue kits to patients and their families when appropriate. The clinician can also institute diversion and overdose prevention measures for patients taking buprenorphine or methadone—providing a lock box for take-home medication, implementing treatment contracts, and using a designated pharmacy to dispense buprenorphine, for example.26,27,50
Regular monitoring, urine drug screens, and random pill counts, in which patients are typically given 24 hours to bring in their prescribed medication so it can be counted, can also help keep patients on track. Treatment for concurrent psychiatric disorders—depression, anxiety, and personality disorders are common among patients with opioid dependence—is also likely to improve treatment outcomes.
References
1. CDC. Emergency department visits involving nonmedical use of selected prescription drugs - United States, 2004-2008. MMWR Morb Mortal Wkly Rep. 2010;59:705-709.
2. Bohnert AS, Valenstein M, Bair MJ, et al. Association between opioid prescribing patterns and opioid overdose-related deaths. JAMA. 2011;305:1315-1321.
3. Warner M, Chen LH, Makuc DM. Increase in fatal poisonings involving opioid analgesics in the United States, 1999-2006. NCHS Data Brief. 2009;22:1-8.
4. Substance Abuse and Mental Health Services Administration. Results From the 2009 National Survey on Drug Use and Health: Volume I. Summary of National Findings. Rockville, Md: SAMHSA, Office of Applied Studies; 2010. NSDUH Series H-38A, HHS publication SMA 10-4856. www.samhsa.gov/dataNSDUH/2k9NSDUH/2k9Results.htm. Accessed August 22, 2012.
5. Hser YI, Huang D, Brecht ML, et al. Contrasting trajectories of heroin, cocaine, and methamphetamine use. J Addict Dis. 2008;27:13-21.
6. Veilleux JC, Colvin PJ, Anderson J, et al. A review of opioid dependence treatment: pharmacological and psychosocial interventions to treat opioid addiction. Clin Psychol Rev. 2011;30:155-166.
7. George O, Koob GF. Individual differences in prefrontal cortex function and the transition from drug use to drug dependence. Neurosci Biobehav Rev. 2011;35:232-247.
8. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 4th ed, text rev (DSM-IV-TR). Arlington, VA: American Psychiatric Association; 2000.
9. American Psychiatric Association. R 19 opioid use disorder (2012). www.dsm5.org/ProposedRevisions/Pages/proposedrevision.aspx?rid=460. Accessed June 20, 2012.
10. Substance Abuse and Mental Health Services Administration. Results From the 2008 National Survey on Drug Use and Health: National Findings. Rockville, MD: SAMHSA, Office of Applied Studies; 2009. NSDUH Series H-36, HHS publication SMA 09-4434. www.samhsa.gov/data/nsduh/2k8nsduh/2k8Results.htm. Accessed August 22, 2012.
11. Brown RL, Rounds LA. Conjoint screening questionnaires for alcohol and other drug abuse: criterion validity in a primary care practice. Wis Med J. 1995;94:135-140.
12. Skinner HA. The drug abuse screening test. Addict Behav. 1982;7:363-371.
13. US Preventive Services Task Force. Screening for Illicit Drug Use: US Preventive Services Task Force Recommendation Statement. January 2008. www.uspreventiveservicestaskforce.org/uspstf08/druguse/drugrs.htm. Accessed May 7, 2012.
14. Gourlay D, Caplan Y, Heit H. Urine Drug Testing in Clinical Practice: Dispelling the Myths and Designing Strategies. San Francisco, Calif: California Academy of Family Physicians; 2006.
15. Jackman R, Purvis J, Mallett B. Chronic nonmalignant pain in primary care. Am Fam Physician. 2008;78:1155-1162.
16. McBane S, Weigle N. Is it time to drug test your chronic pain patient? J Fam Pract. 2010;59:628-633.
17. Madras BK, Compton WM, Avula D, et al. Screening, brief interventions, referral to treatment (SBIRT) for illicit drug and alcohol use at multiple healthcare sites: comparison at intake and 6 months later. Drug Alcohol Depend. 2009;99:280-295.
18. The InSight Project Research Group. SBIRT outcomes in Houston: final report on InSight, a hospital district-based program for patients at risk for alcohol or drug use problems. Alcohol Clin Exp Res. 2009;33:1374-1381.
19. Borges G, Walters EE, Kessler RC. Associations of substance use, abuse, and dependence with subsequent suicidal behavior. Am J Epidemiol. 2000;151:781-789.
20. Prochaska JO, DiClemente CC. Stages and processes of self-change of smoking: toward an integrative model of change. J Consult Clin Psychol. 1983;51:390-395.
21. Smedslund G, Berg RC, Hammerstrom KT, et al. Motivational interviewing for substance abuse. Cochrane Database Syst Rev. 2011;(5):CD008063.
22. Gryczynski J, Mitchell SG, Peterson TR, et al. The relationship between services delivered and substance use outcomes in New Mexico's Screening, Brief Intervention, Referral and Treatment (SBIRT) Initiative. Drug Alcohol Depend. 2011;118:152-157.
23. Woody GE, Poole SA, Subramaniam G, et al. Extended vs short-term buprenorphine-naloxone for treatment of opioid-addicted youth: a randomized trial. JAMA. 2008;300:2003-2011.
24. Weiss RD, Potter JS, Fiellin DA, et al. Adjunctive counseling during brief and extended buprenorphine-naloxone treatment for prescription opioid dependence: a 2-phase randomized controlled trial. Arch Gen Psychiatry. 2011;68:1238-1246.
25. Johansson BA, Berglund M, Lindgren A. Efficacy of maintenance treatment with naltrexone for opioid dependence: a meta-analytical review. Addiction. 2006;101:491-503.
26. Hall AJ, Logan JE, Toblin RL, et al. Patterns of abuse among unintentional pharmaceutical overdose fatalities. JAMA. 2008;300:2613-2620.
27. Zacny J, Bigelow G, Compton P, et al. College on Problems of Drug Dependence taskforce on prescription opioid non-medical use and abuse: position statement. Drug Alcohol Depend. 2003;69:215-232.
28. Kreek MJ. Rationale for maintenance pharmacotherapy of opiate dependence. Res Publ Assoc Res Nerv Ment Dis. 1992;70:205-230.
29. Mattick RP, Breen C, Kimber J, et al. Methadone maintenance therapy versus no opioid replacement therapy for opioid dependence. Cochrane Database Syst Rev. 2009;(3):CD002209.
30. McCance-Katz EF, Sullivan LE, Nallani S. Drug interactions of clinical importance among the opioids, methadone and buprenorphine, and other frequently prescribed medications: a review. Am J Addict. 2010;19:4-16.
31. Office of National Drug Control Policy Reauthorization Act of 2006 (ONDCPRA), HR 6344, 109th Cong, 2nd Sess (2006).
32. Lewis JW, Walter D. Buprenorphine—background to its development as a treatment for opiate dependence. In: Blaine JD, ed. Buprenorphine: An Alternative Treatment for Opioid Dependence. Rockville, MD: National Institute on Drug Abuse; 1992:5-11. NIDA Research Monograph, No. 121. http://archives.drugabuse.gov/pdf/monographs/121.pdf. Accessed August 22, 2012.
33. Walsh SL, Preston KL, Stitzer ML, et al. Clinical pharmacology of buprenorphine: ceiling effects at high doses. Clin Pharmacol Ther. 1994;55:569-580.
34. Alho H, Sinclair D, Vuori E, et al. Abuse liability of buprenorphine-naloxone tablets in untreated IV drug users. Drug Alcohol Depend. 2007;88:75-78.
35. Hallinan R, Byrne A, Agho K, et al. Erectile dysfunction in men receiving methadone and buprenorphine maintenance treatment. J Sex Med. 2008;5:684-692.
36. Rapeli P, Fabritius C, Alho H, et al. Methadone vs. buprenorphine/naloxone during early opioid substitution treatment: a naturalistic comparison of cognitive performance relative to healthy controls. BMC Clin Pharmacol. 2007;7:5.
37. Wedam EF, Bigelow GE, Johnson RE, et al. QT-interval effects of methadone, levomethadyl, and buprenorphine in a randomized trial. Arch Intern Med. 2007;167:2469-2475.
38. Center for Substance Abuse Treatment. Clinical Guidelines for the Use of Buprenorphine in the Treatment of Opioid Addiction. Rockville, MD: Substance Abuse and Mental Health Services Administration; 2004. Treatment Improvement Protocol (TIP) Series 40. DHHS publication SMA 04‐3939.
39. Kleber HD. Methadone maintenance 4 decades later: thousands of lives saved but still controversial. JAMA. 2008;300:2303-2305.
40. Hulse GK, Morris N, Arnold-Reed D, et al. Improving clinical outcomes in treating heroin dependence: randomized, controlled trial of oral or implant naltrexone. Arch Gen Psychiatry. 2009;66:1108-1115.
41. FDA. FDA approves injectable drug to treat opioid-dependent patients. October 12, 2010. www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/2010/ucm229109.htm. Accessed September 11, 2012.
42. Inturrisi CE, Verebely K. The levels of methadone in the plasma in methadone maintenance. Clin Pharmacol Ther. 1972;13(5 pt 1):633-637.
43. Stitzer M, Bigelow G, Lawrence C, et al. Medication take-home as a reinforcer in a methadone maintenance program. Addict Behav. 1977;2:9-14.
44. Code of Federal Regulations. Title 42.8.12. Federal Opioid Treatment Standards. October 2010.
45. Johnson RE. Chutuape MA, Strain EC, et al. A comparison of levomethadyl acetate, buprenorphine, and methadone for opioid dependence. N Engl J Med. 2000;343:1290-1297.
46. Krantz MJ, Martin J, Stimmel B, et al. QTc interval screening in methadone treatment. Ann Intern Med. 2009;150:387-395.
47. Ball JC, Lange WR, Myers CP, et al. Reducing the risk of AIDS through methadone maintenance treatment. J Health Soc Behav. 1988;29:214-226.
48. Fiellin DA, Pantalon MV, Chawarski MC, et al. Counseling plus buprenorphine-naloxone maintenance therapy for opioid dependence. N Engl J Med. 2006;355:365-374.
49. Defulio A, Everly JJ, Leoutsakos JM, et al. Employment-based reinforcement of adherence to an FDA approved extended release formulation of naltrexone in opioid-dependent adults: a randomized controlled trial. Drug Alcohol Depend. 2012;120:48-54.
50. Savage SR. Management of opioid medications in patients with chronic pain and risk of substance misuse. Curr Psychiatry Rep. 2009;11:377-384.
This article was originally published in the Journal of Family Practice. 2012;61:588-597.