User login
Grand Rounds: Man, 65, With Delayed Pain After Hand Injury
A 65-year-old man presented to the emergency department (ED) with a two-week history of progressively severe pain in his right hand and difficulty moving his fingers. He reported that approximately two weeks earlier, while shoveling snow, he slipped and fell, landing on his right hand. Initially, he had no problems with his hand. He finished his shoveling and continued his normal daily activities.
Within two to three days he started to experience pain in his right hand, which grew progressively worse.
Because he did not have a primary care provider, the patient had a limited medical history. He reported having a mildly elevated prostate-specific antigen test years earlier. He underwent an appendectomy at age 15. He denied any other medical problems.
The patient was taking no medications and reported no known allergies to medications. He denied the use of tobacco, said he had one or two beers on an average day, and denied IV drug use. He was an artist and was married with one adult child. His family history was unremarkable with the exception of an alcoholic sister who died of cirrhosis at age 70.
During triage, vital signs were essentially normal: blood pressure, 142/74 mm Hg; heart rate, 78 beats/min; and respiratory rate, 20 breaths/min. The patient was afebrile at 37.2°C (98.9°F). Physical examination was remarkable for some edema and warmth of the right hand without any notable erythema. There was no evidence of any wound. Fingers all had good sensation; however, flexion of the index and long fingers elicited a significant increase in pain.
The remainder of the exam was unremarkable. The patient’s head was normocephalic and atraumatic. Pupils were equal, round, and reactive to light. Eyes were anicteric, and no rhinorrhea was evident. The neck was supple without palpable lymphadenopathy. Lungs were clear to auscultation bilaterally. No wheezes, rales, or rhonchi were appreciated. The heart had a regular rate and rhythm; no murmurs, rubs, or gallups were noted. The abdomen was soft and non-tender. The extremities, except as previously stated, were normal, with good pulses, sensation, and strength.
Initially, only radiographs of the right hand were ordered (see Figures 1 and 2). These demonstrated soft tissue swelling on the dorsum of the hand, and an area of hypodensity between the first and second metacarpals. There were no fractures, dislocations, or other bone or joint abnormalities.
After a review of the radiographs, it was clear that the patient’s diagnosis was not a simple answer of hand contusion or fracture; thus, the evaluation was expanded. Vital signs were repeated three hours after triage: blood pressure, 128/74 mm Hg; heart rate, 76 beats/min; and respiration, 20 breaths/min. The patient was now febrile at 37.6°C (99.7°F). Because of his fever and the anomaly on the patient’s hand radiograph, expansion of the evaluation continued.
Laboratory studies included a complete blood count: white blood cells (WBCs), 30,700/mcL (reference range,1 4,500 to 11,000/mcL); hemoglobin, 13.3 g/dL (13.8 to 17.2 g/dL for men); hematocrit, 40.0% (41% to 50% for men); platelets, 217,000/mcL (130 to 400 x 103/mcL). Initial chemistry panel results were normal except for serum glucose, 143 mg/dL (70 to 125 mg/dL).
Liver function test results were normal except for aspartate aminotransferase, 33 U/L (reference range,1 10 to 30 U/L) and albumin, 2.5 g/dL (3.5 to 5.0 g/dL). Once WBCs were found to exceed 30,000/mcL, the search for a cause was widened once more.
The continued studies included a chest radiograph with normal results, unremarkable CT of the abdomen and pelvis with IV contrast, blood cultures, and urinalysis. The urinalysis showed: blood, moderate; protein, trace; nitrites, positive; leukocytes, large; WBCs > 50/high-power field (reference range,1 5/high-power field or less); and numerous bacteria.
The final study performed in the ED evaluation of the patient was a CT of the right hand with IV contrast (see Figure 3). It demonstrated diffuse edema and a 9.0-mm area of low attenuation with some rim enhancement. The differential for these findings includes an abscess or a foreign body; the latter was deemed unlikely in light of the patient’s physical exam. In consideration of his elevated WBC count, the high number of WBCs in his urine, the fever, and the CT results, the patient was diagnosed with an abscess in his right hand that had been seeded, it was surmised, by an occult urosepsis after his fall.
Before the patient’s admission, a hand surgeon was consulted. The surgeon agreed with the diagnosis, and the patient was taken to the operating room (OR). He had been given piperacillin/tazobactam in the ED.
In the OR, the surgeon made a 3.0-cm incision, conducted an exploration, and identified a cavity that contained a small amount of purulence. He determined the lesion to be a resolving abscess. The wound was washed out, and the area was closed with a Penrose drain.
The patient was continued on the piperacillin/tazobactam. His blood culture was positive for gram-positive rods, and a low-grade fever persisted. An infectious disease specialist was consulted, and levofloxacin was added to the patient’s regimen.
After 24 hours of treatment, findings on urinalysis improved: blood, small; protein, trace; nitrites, negative; leukocytes, small; WBCs, 15 to 20/high-power field; and no bacteria. Over the next three days, the patient’s condition continued to improve. His hand drain was removed, and the pain and swelling subsided. He became afebrile, and his WBC count fell to 24,700/mcL. He was discharged to home with prescriptions for cephalexin and levofloxacin. Follow-up for postoperative care was arranged with the hand surgeon.
Discussion
Pyomyositis is defined as abscess formation deep within large striated muscles.1 Although this condition is uncommon, it is believed that an occult bacteremia can seed an area of damaged muscle (compared with healthy muscle, which ordinarily resists infection), allowing an abscess to form.1,2
Epidemiology
In a 2002 review involving 676 patients with primary pyomyositis, Bickels et al3 reported the condition in ages ranging from two months to 82 years (mean, 28.1 years). In a majority of cases, only a single muscle was involved; 112 patients (16.6%) were identified with multiple-site involvement. Only seven cases (0.1%) involved the hand.
In 452 cases (66.9%), a bacterial agent was identified. Among these, 350 (77%) had a positive culture for Staphylococcus aureus. Other isolates included Streptococcus pyogenes, Escherichia coli, Salmonella enteritidus, and Mycobacterium tuberculosis.1,3 It should be noted that community-acquired methicillin-resistant S aureus (CA-MRSA) is being implicated with increasing frequency in cases of pyomyositis.4-6
Because pyomyositis is not a reportable disease and has not been studied in large clinical trials, its incidence is uncertain, and proposed risk factors have not all been confirmed2 (see Table2,7).
Pathophysiology
While the etiology of primary pyomyositis is unclear, it is believed to be caused by a combination of bacteremia (chronic or transient) and damaged muscle. In a 1960 study published in the Lancet, Smith and Vickers8 performed autopsies on 327 patients who had died of culture-positive septicemia. Only two patients were found to have a muscle abscess. At that time, the investigators concluded that both muscle injury and bacteremia would need to be present in order for an abscess to form. In animal studies, bacteremia (eg, S aureus) does not appear to lead to pyomyositis except in cases of muscle abnormality or trauma (eg, electric shock, pinching injury).9,10
When a history of trauma can be identified in patients with pyomyositis, the condition typically develops near the affected muscle, and the infection appears within days to weeks.3 In cases in which an antecedent infection is identified and hematogenous spread of the bacteria to the skeletal muscle occurs, this is termed secondary pyomyositis.11
Disease Progression
Pyomyositis generally progresses in three stages, beginning with inflammation and advancing to a focal abscess, then to a septic state.3 The first stage develops between seven and 21 days after the initial incident, is typically subacute, involves mild pain and swelling with a “woody” texture, and is occasionally associated with fevers.2
Diagnosis of pyomyositis is usually made during the second stage, 10 to 21 days after the initial incident; by that time, the pain has increased, and the fever is more pronounced. Third-stage infection usually involves fluctuance and sepsis.2
Although MRI is considered most useful in the diagnosis of pyomyositis, CT and ultrasound allow for percutaneous needle aspiration and drainage.3
Treatment
The correct treatment for pyomyositis depends upon the stage at which the disease is identified. During the first stage (before formation of an abscess), antibiotic treatment alone may be sufficient.1 Once an abscess has formed, an incision and drainage will be required, in conjunction with or followed by appropriate antibiotic therapy.
When pyomyositis is properly treated during the first or second stage, a full recovery is likely.2,3 By the third stage, surgical debridement is required. Additionally, osteomyelitis may develop in the adjacent bones, followed by muscle scarring, residual weakness, and functional impairment.2,3 Reported pyomyositis-associated mortality ranges between less than 1% and 4%.2,12
The Case Patient
The case presented here was of particular interest for two reasons. First, the patient had a traumatic injury that initially caused him no concern but worsened progressively over 14 days. Although this is not the typical presentation of a traumatic injury, the ED staff could very easily have performed a radiograph, made a diagnosis of traumatic hand injury, and discharged the patient.
Second, men in their 60s do not commonly have urinary tract infections.13 The patient was questioned frequently by several providers about sexual behaviors, medical problems, and urinary symptoms. Repeatedly, he denied all of these issues. While a urinalysis may be omitted in the evaluation of an otherwise healthy, asymptomatic patient, its results in this case were a key piece of data.
It should be noted that the patient thought it inappropriate to be asked for urine samples. He repeatedly said, “It’s my hand!”
Conclusion
Even in patients presenting with the most routine complaint, a careful evaluation can reveal unexpected, serious problems. This patient complained of pain in his hand some time after a fall and ultimately was treated for an occult urosepsis and hand abscess—pyomyositis, which rarely occurs in small muscles, such as those of the hand. Either condition, left untreated, could have led to serious morbidity or even mortality.
1. Beers MH, Berkow R, eds. Merck Manual of Diagnosis and Therapy. 18th ed. Whitehouse Station, NJ: Merck Research Laboratories, 2006:1142-1143.
2. Crum-Cianflone NF. Bacterial, fungal, parasitic and viral myositis. Clin Microbiol Rev. 2008;21(3):473-494.
3. Bickels J, Ben-Sira L, Kessler A, Wientroub S. Current concepts review: primary pyomyositis. J Bone Joint Surg Am. 2002;84-A(12):2277-2286.
4. Lo BM, Fickenscher BA. Primary pyomyositis caused by ca-MRSA. Int J Emerg Med. 2008;1(4):331-332.
5. Ruiz ME, Yohannes S, Wladyka CG. Pyomyositis caused by methicillin-resistant Staphylococcus aureus. N Engl J Med. 2005;352(14):1488–1489.
6. Pannaraj PS, Hulten KG, Gonzalez BE, et al. Infective pyomyositis and myositis in children in the era of community-acquired, methicillin-resistant Staphylococcus aureus infection. Clin Infect Dis. 2006;43(8):953–960.
7. Ükinç K, Bayraktar M, Uzun O. A case of type 2 diabetes complicated with primary pyomyositis. Endocrinologist. 2009;19(3):129-130.
8. Smith IM, Vickers AB. Natural history of 338 treated and untreated patients with staphylococcal septicaemia (1936-1955). Lancet. 1960;1(7138):1318-1322.
9. Phoon E-S, Sebastin SJ, Tay S-C. Primary pyomyositis (bacterial myositis) of the pronator quadratus. J Hand Surg Eur Vol. 2009;34(4):549-551.
10. Christin L, Sarosi GA. Pyomyositis in North America: case reports and review. Clin Infect Dis. 1992; 15(4):668-677.
11. Sokolowski MJ, Koh JL. Pyomyositis of the shoulder girdle. Orthopedics. 2006;29(11):1030-1032.
12. Crum NF. Bacterial pyomyositis in the United States. Am J Med. 2004;117(6):420–428.
13. Foxman B. Epidemiology of urinary tract infections: incidence, morbidity, and economic costs. Am J Med. 2002;113 suppl 1A:5S-13S.
A 65-year-old man presented to the emergency department (ED) with a two-week history of progressively severe pain in his right hand and difficulty moving his fingers. He reported that approximately two weeks earlier, while shoveling snow, he slipped and fell, landing on his right hand. Initially, he had no problems with his hand. He finished his shoveling and continued his normal daily activities.
Within two to three days he started to experience pain in his right hand, which grew progressively worse.
Because he did not have a primary care provider, the patient had a limited medical history. He reported having a mildly elevated prostate-specific antigen test years earlier. He underwent an appendectomy at age 15. He denied any other medical problems.
The patient was taking no medications and reported no known allergies to medications. He denied the use of tobacco, said he had one or two beers on an average day, and denied IV drug use. He was an artist and was married with one adult child. His family history was unremarkable with the exception of an alcoholic sister who died of cirrhosis at age 70.
During triage, vital signs were essentially normal: blood pressure, 142/74 mm Hg; heart rate, 78 beats/min; and respiratory rate, 20 breaths/min. The patient was afebrile at 37.2°C (98.9°F). Physical examination was remarkable for some edema and warmth of the right hand without any notable erythema. There was no evidence of any wound. Fingers all had good sensation; however, flexion of the index and long fingers elicited a significant increase in pain.
The remainder of the exam was unremarkable. The patient’s head was normocephalic and atraumatic. Pupils were equal, round, and reactive to light. Eyes were anicteric, and no rhinorrhea was evident. The neck was supple without palpable lymphadenopathy. Lungs were clear to auscultation bilaterally. No wheezes, rales, or rhonchi were appreciated. The heart had a regular rate and rhythm; no murmurs, rubs, or gallups were noted. The abdomen was soft and non-tender. The extremities, except as previously stated, were normal, with good pulses, sensation, and strength.
Initially, only radiographs of the right hand were ordered (see Figures 1 and 2). These demonstrated soft tissue swelling on the dorsum of the hand, and an area of hypodensity between the first and second metacarpals. There were no fractures, dislocations, or other bone or joint abnormalities.
After a review of the radiographs, it was clear that the patient’s diagnosis was not a simple answer of hand contusion or fracture; thus, the evaluation was expanded. Vital signs were repeated three hours after triage: blood pressure, 128/74 mm Hg; heart rate, 76 beats/min; and respiration, 20 breaths/min. The patient was now febrile at 37.6°C (99.7°F). Because of his fever and the anomaly on the patient’s hand radiograph, expansion of the evaluation continued.
Laboratory studies included a complete blood count: white blood cells (WBCs), 30,700/mcL (reference range,1 4,500 to 11,000/mcL); hemoglobin, 13.3 g/dL (13.8 to 17.2 g/dL for men); hematocrit, 40.0% (41% to 50% for men); platelets, 217,000/mcL (130 to 400 x 103/mcL). Initial chemistry panel results were normal except for serum glucose, 143 mg/dL (70 to 125 mg/dL).
Liver function test results were normal except for aspartate aminotransferase, 33 U/L (reference range,1 10 to 30 U/L) and albumin, 2.5 g/dL (3.5 to 5.0 g/dL). Once WBCs were found to exceed 30,000/mcL, the search for a cause was widened once more.
The continued studies included a chest radiograph with normal results, unremarkable CT of the abdomen and pelvis with IV contrast, blood cultures, and urinalysis. The urinalysis showed: blood, moderate; protein, trace; nitrites, positive; leukocytes, large; WBCs > 50/high-power field (reference range,1 5/high-power field or less); and numerous bacteria.
The final study performed in the ED evaluation of the patient was a CT of the right hand with IV contrast (see Figure 3). It demonstrated diffuse edema and a 9.0-mm area of low attenuation with some rim enhancement. The differential for these findings includes an abscess or a foreign body; the latter was deemed unlikely in light of the patient’s physical exam. In consideration of his elevated WBC count, the high number of WBCs in his urine, the fever, and the CT results, the patient was diagnosed with an abscess in his right hand that had been seeded, it was surmised, by an occult urosepsis after his fall.
Before the patient’s admission, a hand surgeon was consulted. The surgeon agreed with the diagnosis, and the patient was taken to the operating room (OR). He had been given piperacillin/tazobactam in the ED.
In the OR, the surgeon made a 3.0-cm incision, conducted an exploration, and identified a cavity that contained a small amount of purulence. He determined the lesion to be a resolving abscess. The wound was washed out, and the area was closed with a Penrose drain.
The patient was continued on the piperacillin/tazobactam. His blood culture was positive for gram-positive rods, and a low-grade fever persisted. An infectious disease specialist was consulted, and levofloxacin was added to the patient’s regimen.
After 24 hours of treatment, findings on urinalysis improved: blood, small; protein, trace; nitrites, negative; leukocytes, small; WBCs, 15 to 20/high-power field; and no bacteria. Over the next three days, the patient’s condition continued to improve. His hand drain was removed, and the pain and swelling subsided. He became afebrile, and his WBC count fell to 24,700/mcL. He was discharged to home with prescriptions for cephalexin and levofloxacin. Follow-up for postoperative care was arranged with the hand surgeon.
Discussion
Pyomyositis is defined as abscess formation deep within large striated muscles.1 Although this condition is uncommon, it is believed that an occult bacteremia can seed an area of damaged muscle (compared with healthy muscle, which ordinarily resists infection), allowing an abscess to form.1,2
Epidemiology
In a 2002 review involving 676 patients with primary pyomyositis, Bickels et al3 reported the condition in ages ranging from two months to 82 years (mean, 28.1 years). In a majority of cases, only a single muscle was involved; 112 patients (16.6%) were identified with multiple-site involvement. Only seven cases (0.1%) involved the hand.
In 452 cases (66.9%), a bacterial agent was identified. Among these, 350 (77%) had a positive culture for Staphylococcus aureus. Other isolates included Streptococcus pyogenes, Escherichia coli, Salmonella enteritidus, and Mycobacterium tuberculosis.1,3 It should be noted that community-acquired methicillin-resistant S aureus (CA-MRSA) is being implicated with increasing frequency in cases of pyomyositis.4-6
Because pyomyositis is not a reportable disease and has not been studied in large clinical trials, its incidence is uncertain, and proposed risk factors have not all been confirmed2 (see Table2,7).
Pathophysiology
While the etiology of primary pyomyositis is unclear, it is believed to be caused by a combination of bacteremia (chronic or transient) and damaged muscle. In a 1960 study published in the Lancet, Smith and Vickers8 performed autopsies on 327 patients who had died of culture-positive septicemia. Only two patients were found to have a muscle abscess. At that time, the investigators concluded that both muscle injury and bacteremia would need to be present in order for an abscess to form. In animal studies, bacteremia (eg, S aureus) does not appear to lead to pyomyositis except in cases of muscle abnormality or trauma (eg, electric shock, pinching injury).9,10
When a history of trauma can be identified in patients with pyomyositis, the condition typically develops near the affected muscle, and the infection appears within days to weeks.3 In cases in which an antecedent infection is identified and hematogenous spread of the bacteria to the skeletal muscle occurs, this is termed secondary pyomyositis.11
Disease Progression
Pyomyositis generally progresses in three stages, beginning with inflammation and advancing to a focal abscess, then to a septic state.3 The first stage develops between seven and 21 days after the initial incident, is typically subacute, involves mild pain and swelling with a “woody” texture, and is occasionally associated with fevers.2
Diagnosis of pyomyositis is usually made during the second stage, 10 to 21 days after the initial incident; by that time, the pain has increased, and the fever is more pronounced. Third-stage infection usually involves fluctuance and sepsis.2
Although MRI is considered most useful in the diagnosis of pyomyositis, CT and ultrasound allow for percutaneous needle aspiration and drainage.3
Treatment
The correct treatment for pyomyositis depends upon the stage at which the disease is identified. During the first stage (before formation of an abscess), antibiotic treatment alone may be sufficient.1 Once an abscess has formed, an incision and drainage will be required, in conjunction with or followed by appropriate antibiotic therapy.
When pyomyositis is properly treated during the first or second stage, a full recovery is likely.2,3 By the third stage, surgical debridement is required. Additionally, osteomyelitis may develop in the adjacent bones, followed by muscle scarring, residual weakness, and functional impairment.2,3 Reported pyomyositis-associated mortality ranges between less than 1% and 4%.2,12
The Case Patient
The case presented here was of particular interest for two reasons. First, the patient had a traumatic injury that initially caused him no concern but worsened progressively over 14 days. Although this is not the typical presentation of a traumatic injury, the ED staff could very easily have performed a radiograph, made a diagnosis of traumatic hand injury, and discharged the patient.
Second, men in their 60s do not commonly have urinary tract infections.13 The patient was questioned frequently by several providers about sexual behaviors, medical problems, and urinary symptoms. Repeatedly, he denied all of these issues. While a urinalysis may be omitted in the evaluation of an otherwise healthy, asymptomatic patient, its results in this case were a key piece of data.
It should be noted that the patient thought it inappropriate to be asked for urine samples. He repeatedly said, “It’s my hand!”
Conclusion
Even in patients presenting with the most routine complaint, a careful evaluation can reveal unexpected, serious problems. This patient complained of pain in his hand some time after a fall and ultimately was treated for an occult urosepsis and hand abscess—pyomyositis, which rarely occurs in small muscles, such as those of the hand. Either condition, left untreated, could have led to serious morbidity or even mortality.
A 65-year-old man presented to the emergency department (ED) with a two-week history of progressively severe pain in his right hand and difficulty moving his fingers. He reported that approximately two weeks earlier, while shoveling snow, he slipped and fell, landing on his right hand. Initially, he had no problems with his hand. He finished his shoveling and continued his normal daily activities.
Within two to three days he started to experience pain in his right hand, which grew progressively worse.
Because he did not have a primary care provider, the patient had a limited medical history. He reported having a mildly elevated prostate-specific antigen test years earlier. He underwent an appendectomy at age 15. He denied any other medical problems.
The patient was taking no medications and reported no known allergies to medications. He denied the use of tobacco, said he had one or two beers on an average day, and denied IV drug use. He was an artist and was married with one adult child. His family history was unremarkable with the exception of an alcoholic sister who died of cirrhosis at age 70.
During triage, vital signs were essentially normal: blood pressure, 142/74 mm Hg; heart rate, 78 beats/min; and respiratory rate, 20 breaths/min. The patient was afebrile at 37.2°C (98.9°F). Physical examination was remarkable for some edema and warmth of the right hand without any notable erythema. There was no evidence of any wound. Fingers all had good sensation; however, flexion of the index and long fingers elicited a significant increase in pain.
The remainder of the exam was unremarkable. The patient’s head was normocephalic and atraumatic. Pupils were equal, round, and reactive to light. Eyes were anicteric, and no rhinorrhea was evident. The neck was supple without palpable lymphadenopathy. Lungs were clear to auscultation bilaterally. No wheezes, rales, or rhonchi were appreciated. The heart had a regular rate and rhythm; no murmurs, rubs, or gallups were noted. The abdomen was soft and non-tender. The extremities, except as previously stated, were normal, with good pulses, sensation, and strength.
Initially, only radiographs of the right hand were ordered (see Figures 1 and 2). These demonstrated soft tissue swelling on the dorsum of the hand, and an area of hypodensity between the first and second metacarpals. There were no fractures, dislocations, or other bone or joint abnormalities.
After a review of the radiographs, it was clear that the patient’s diagnosis was not a simple answer of hand contusion or fracture; thus, the evaluation was expanded. Vital signs were repeated three hours after triage: blood pressure, 128/74 mm Hg; heart rate, 76 beats/min; and respiration, 20 breaths/min. The patient was now febrile at 37.6°C (99.7°F). Because of his fever and the anomaly on the patient’s hand radiograph, expansion of the evaluation continued.
Laboratory studies included a complete blood count: white blood cells (WBCs), 30,700/mcL (reference range,1 4,500 to 11,000/mcL); hemoglobin, 13.3 g/dL (13.8 to 17.2 g/dL for men); hematocrit, 40.0% (41% to 50% for men); platelets, 217,000/mcL (130 to 400 x 103/mcL). Initial chemistry panel results were normal except for serum glucose, 143 mg/dL (70 to 125 mg/dL).
Liver function test results were normal except for aspartate aminotransferase, 33 U/L (reference range,1 10 to 30 U/L) and albumin, 2.5 g/dL (3.5 to 5.0 g/dL). Once WBCs were found to exceed 30,000/mcL, the search for a cause was widened once more.
The continued studies included a chest radiograph with normal results, unremarkable CT of the abdomen and pelvis with IV contrast, blood cultures, and urinalysis. The urinalysis showed: blood, moderate; protein, trace; nitrites, positive; leukocytes, large; WBCs > 50/high-power field (reference range,1 5/high-power field or less); and numerous bacteria.
The final study performed in the ED evaluation of the patient was a CT of the right hand with IV contrast (see Figure 3). It demonstrated diffuse edema and a 9.0-mm area of low attenuation with some rim enhancement. The differential for these findings includes an abscess or a foreign body; the latter was deemed unlikely in light of the patient’s physical exam. In consideration of his elevated WBC count, the high number of WBCs in his urine, the fever, and the CT results, the patient was diagnosed with an abscess in his right hand that had been seeded, it was surmised, by an occult urosepsis after his fall.
Before the patient’s admission, a hand surgeon was consulted. The surgeon agreed with the diagnosis, and the patient was taken to the operating room (OR). He had been given piperacillin/tazobactam in the ED.
In the OR, the surgeon made a 3.0-cm incision, conducted an exploration, and identified a cavity that contained a small amount of purulence. He determined the lesion to be a resolving abscess. The wound was washed out, and the area was closed with a Penrose drain.
The patient was continued on the piperacillin/tazobactam. His blood culture was positive for gram-positive rods, and a low-grade fever persisted. An infectious disease specialist was consulted, and levofloxacin was added to the patient’s regimen.
After 24 hours of treatment, findings on urinalysis improved: blood, small; protein, trace; nitrites, negative; leukocytes, small; WBCs, 15 to 20/high-power field; and no bacteria. Over the next three days, the patient’s condition continued to improve. His hand drain was removed, and the pain and swelling subsided. He became afebrile, and his WBC count fell to 24,700/mcL. He was discharged to home with prescriptions for cephalexin and levofloxacin. Follow-up for postoperative care was arranged with the hand surgeon.
Discussion
Pyomyositis is defined as abscess formation deep within large striated muscles.1 Although this condition is uncommon, it is believed that an occult bacteremia can seed an area of damaged muscle (compared with healthy muscle, which ordinarily resists infection), allowing an abscess to form.1,2
Epidemiology
In a 2002 review involving 676 patients with primary pyomyositis, Bickels et al3 reported the condition in ages ranging from two months to 82 years (mean, 28.1 years). In a majority of cases, only a single muscle was involved; 112 patients (16.6%) were identified with multiple-site involvement. Only seven cases (0.1%) involved the hand.
In 452 cases (66.9%), a bacterial agent was identified. Among these, 350 (77%) had a positive culture for Staphylococcus aureus. Other isolates included Streptococcus pyogenes, Escherichia coli, Salmonella enteritidus, and Mycobacterium tuberculosis.1,3 It should be noted that community-acquired methicillin-resistant S aureus (CA-MRSA) is being implicated with increasing frequency in cases of pyomyositis.4-6
Because pyomyositis is not a reportable disease and has not been studied in large clinical trials, its incidence is uncertain, and proposed risk factors have not all been confirmed2 (see Table2,7).
Pathophysiology
While the etiology of primary pyomyositis is unclear, it is believed to be caused by a combination of bacteremia (chronic or transient) and damaged muscle. In a 1960 study published in the Lancet, Smith and Vickers8 performed autopsies on 327 patients who had died of culture-positive septicemia. Only two patients were found to have a muscle abscess. At that time, the investigators concluded that both muscle injury and bacteremia would need to be present in order for an abscess to form. In animal studies, bacteremia (eg, S aureus) does not appear to lead to pyomyositis except in cases of muscle abnormality or trauma (eg, electric shock, pinching injury).9,10
When a history of trauma can be identified in patients with pyomyositis, the condition typically develops near the affected muscle, and the infection appears within days to weeks.3 In cases in which an antecedent infection is identified and hematogenous spread of the bacteria to the skeletal muscle occurs, this is termed secondary pyomyositis.11
Disease Progression
Pyomyositis generally progresses in three stages, beginning with inflammation and advancing to a focal abscess, then to a septic state.3 The first stage develops between seven and 21 days after the initial incident, is typically subacute, involves mild pain and swelling with a “woody” texture, and is occasionally associated with fevers.2
Diagnosis of pyomyositis is usually made during the second stage, 10 to 21 days after the initial incident; by that time, the pain has increased, and the fever is more pronounced. Third-stage infection usually involves fluctuance and sepsis.2
Although MRI is considered most useful in the diagnosis of pyomyositis, CT and ultrasound allow for percutaneous needle aspiration and drainage.3
Treatment
The correct treatment for pyomyositis depends upon the stage at which the disease is identified. During the first stage (before formation of an abscess), antibiotic treatment alone may be sufficient.1 Once an abscess has formed, an incision and drainage will be required, in conjunction with or followed by appropriate antibiotic therapy.
When pyomyositis is properly treated during the first or second stage, a full recovery is likely.2,3 By the third stage, surgical debridement is required. Additionally, osteomyelitis may develop in the adjacent bones, followed by muscle scarring, residual weakness, and functional impairment.2,3 Reported pyomyositis-associated mortality ranges between less than 1% and 4%.2,12
The Case Patient
The case presented here was of particular interest for two reasons. First, the patient had a traumatic injury that initially caused him no concern but worsened progressively over 14 days. Although this is not the typical presentation of a traumatic injury, the ED staff could very easily have performed a radiograph, made a diagnosis of traumatic hand injury, and discharged the patient.
Second, men in their 60s do not commonly have urinary tract infections.13 The patient was questioned frequently by several providers about sexual behaviors, medical problems, and urinary symptoms. Repeatedly, he denied all of these issues. While a urinalysis may be omitted in the evaluation of an otherwise healthy, asymptomatic patient, its results in this case were a key piece of data.
It should be noted that the patient thought it inappropriate to be asked for urine samples. He repeatedly said, “It’s my hand!”
Conclusion
Even in patients presenting with the most routine complaint, a careful evaluation can reveal unexpected, serious problems. This patient complained of pain in his hand some time after a fall and ultimately was treated for an occult urosepsis and hand abscess—pyomyositis, which rarely occurs in small muscles, such as those of the hand. Either condition, left untreated, could have led to serious morbidity or even mortality.
1. Beers MH, Berkow R, eds. Merck Manual of Diagnosis and Therapy. 18th ed. Whitehouse Station, NJ: Merck Research Laboratories, 2006:1142-1143.
2. Crum-Cianflone NF. Bacterial, fungal, parasitic and viral myositis. Clin Microbiol Rev. 2008;21(3):473-494.
3. Bickels J, Ben-Sira L, Kessler A, Wientroub S. Current concepts review: primary pyomyositis. J Bone Joint Surg Am. 2002;84-A(12):2277-2286.
4. Lo BM, Fickenscher BA. Primary pyomyositis caused by ca-MRSA. Int J Emerg Med. 2008;1(4):331-332.
5. Ruiz ME, Yohannes S, Wladyka CG. Pyomyositis caused by methicillin-resistant Staphylococcus aureus. N Engl J Med. 2005;352(14):1488–1489.
6. Pannaraj PS, Hulten KG, Gonzalez BE, et al. Infective pyomyositis and myositis in children in the era of community-acquired, methicillin-resistant Staphylococcus aureus infection. Clin Infect Dis. 2006;43(8):953–960.
7. Ükinç K, Bayraktar M, Uzun O. A case of type 2 diabetes complicated with primary pyomyositis. Endocrinologist. 2009;19(3):129-130.
8. Smith IM, Vickers AB. Natural history of 338 treated and untreated patients with staphylococcal septicaemia (1936-1955). Lancet. 1960;1(7138):1318-1322.
9. Phoon E-S, Sebastin SJ, Tay S-C. Primary pyomyositis (bacterial myositis) of the pronator quadratus. J Hand Surg Eur Vol. 2009;34(4):549-551.
10. Christin L, Sarosi GA. Pyomyositis in North America: case reports and review. Clin Infect Dis. 1992; 15(4):668-677.
11. Sokolowski MJ, Koh JL. Pyomyositis of the shoulder girdle. Orthopedics. 2006;29(11):1030-1032.
12. Crum NF. Bacterial pyomyositis in the United States. Am J Med. 2004;117(6):420–428.
13. Foxman B. Epidemiology of urinary tract infections: incidence, morbidity, and economic costs. Am J Med. 2002;113 suppl 1A:5S-13S.
1. Beers MH, Berkow R, eds. Merck Manual of Diagnosis and Therapy. 18th ed. Whitehouse Station, NJ: Merck Research Laboratories, 2006:1142-1143.
2. Crum-Cianflone NF. Bacterial, fungal, parasitic and viral myositis. Clin Microbiol Rev. 2008;21(3):473-494.
3. Bickels J, Ben-Sira L, Kessler A, Wientroub S. Current concepts review: primary pyomyositis. J Bone Joint Surg Am. 2002;84-A(12):2277-2286.
4. Lo BM, Fickenscher BA. Primary pyomyositis caused by ca-MRSA. Int J Emerg Med. 2008;1(4):331-332.
5. Ruiz ME, Yohannes S, Wladyka CG. Pyomyositis caused by methicillin-resistant Staphylococcus aureus. N Engl J Med. 2005;352(14):1488–1489.
6. Pannaraj PS, Hulten KG, Gonzalez BE, et al. Infective pyomyositis and myositis in children in the era of community-acquired, methicillin-resistant Staphylococcus aureus infection. Clin Infect Dis. 2006;43(8):953–960.
7. Ükinç K, Bayraktar M, Uzun O. A case of type 2 diabetes complicated with primary pyomyositis. Endocrinologist. 2009;19(3):129-130.
8. Smith IM, Vickers AB. Natural history of 338 treated and untreated patients with staphylococcal septicaemia (1936-1955). Lancet. 1960;1(7138):1318-1322.
9. Phoon E-S, Sebastin SJ, Tay S-C. Primary pyomyositis (bacterial myositis) of the pronator quadratus. J Hand Surg Eur Vol. 2009;34(4):549-551.
10. Christin L, Sarosi GA. Pyomyositis in North America: case reports and review. Clin Infect Dis. 1992; 15(4):668-677.
11. Sokolowski MJ, Koh JL. Pyomyositis of the shoulder girdle. Orthopedics. 2006;29(11):1030-1032.
12. Crum NF. Bacterial pyomyositis in the United States. Am J Med. 2004;117(6):420–428.
13. Foxman B. Epidemiology of urinary tract infections: incidence, morbidity, and economic costs. Am J Med. 2002;113 suppl 1A:5S-13S.
Vitamin-Induced Hemolysis
Evaluation and Treatment of Body Fluid Exposure
Morel-Lavallée Lesion
UPDATE: MENOPAUSE
Much has changed in the management of menopausal women. The Women’s Health Initiative (WHI) and other trials shed light on the risk-benefit ratio of hormone therapy (HT) and significantly altered patterns of usage. A new fracture risk-assessment tool devised by the World Health Organization is now available for widespread use; it continues to be refined so that it can be applied to specific populations with greater accuracy. And the management of low bone mass and osteoporosis has evolved so that we can determine with greater precision exactly who merits our attention.
This year, the Update on Menopause describes:
- a reanalysis of WHI data, focusing on the relationship between hormone therapy and the risk of coronary artery disease (CAD)
- a study from Finland that explores the risk of endometrial cancer associated with various progestin regimens in women who are taking estrogen and who have an intact uterus
- guidance from the North American Menopause Society and the National Osteoporosis Foundation on who, how, and when to evaluate for a likelihood of fracture
- insight into the benefits of and need for vitamin D among menopausal women
- information on a new selective estrogen-receptor modulator under development.
Hormone therapy and CAD: Is the glass half full…or half empty?
Toh S, Hernández-Díaz S, Logan R, Rossouw JE, Hernán MA. Coronary heart disease in postmenopausal recipients of estrogen plus progestin therapy: Does the increased risk ever disappear? A randomized trial. Ann Intern Med. 2010;152(4):211–217.
North American Menopause Society. Position Statement: Estrogen and progestogens use in postmenopausal women: 2010 position statement of The North American Menopause Society. Menopause. 2010;17(2):242–255. DOI: 10.1097/gme.0b013e3181d0f6b9. http://www.menopause.org/PSht10.pdf. Accessed April 1, 2010.
When estrogen therapy is initiated within 10 years of menopause, it may reduce the risk of CAD, according to data from the WHI randomized trial and observational data.
The picture isn’t as clear in regard to estrogen-progestin HT. In a just-published study, Toh and colleagues reassessed data from the WHI trial of continuous oral conjugated equine estrogen plus medroxyprogesterone acetate versus placebo. They also compared the WHI findings with those of the large observational Nurses’ Health Study (NHS). Here are some of their findings:
- participants became less consistent in taking study medication over time—a finding of many long-term studies. This trend prompted Toh and colleagues to adjust their analysis for adherence
- among women who used HT within 10 years after the menopausal transition, the hazard ratio (HR) for CAD was 0.64 (95% confidence interval [CI], 0.21, 1.99) in the WHI and 0.68 (95% CI, 0.24, 1.91) in the NHS. Both hazard ratios suggest that the risk of CAD is lower in Ht users than in nonusers—although the difference is not significant
- when investigators pooled the WHI and NHS findings, the hazard ratio for CAD associated with combination HT was 0.66 (95% CI, 0.31, 1.42). Note that, as the number of participants increases, the confidence limits narrow.
Toh and colleagues concluded that their analysis demonstrated no diminished risk of CAD with HT use. My reading of these data is different: Combination HT does not increase the risk of CAD in women who have been postmenopausal for less than 10 years.
Focus on risk was unbalanced
The question of whether combination HT reduces the risk of CAD in younger women is somewhat moot. I am not aware of any ObGyn in the United States who uses HT to prevent CAD, and the great majority of symptomatic women who consider initiating HT have been menopausal for less than a decade. For these reasons, I find the conclusions drawn by Toh and colleagues a bit mystifying—and the title they chose for their study may be misleading:
Coronary heart disease in postmenopausal recipients of estrogen plus progestin therapy: Does the increased risk ever disappear?
Nevertheless, fear that HT might increase the risk of CAD is common among symptomatic menopausal women and their physicians. What this important analysis can offer is reassurance to symptomatic women who have been menopausal for less than 10 years: namely, that HT—estrogen alone or estrogen plus progestin—does not increase the risk of myocardial infarction or death from CAD.
You should counsel symptomatic women who have been menopausal less than 10 years that short-term use of estrogen therapy or estrogen-progestin therapy does not appear to increase their risk of CAD.
In women taking estrogen, sequential progestin therapy raises the risk of endometrial cancer
Jaakkola S, Lyytinen H, Pukkala E, Ylikorkala O. Endometrial cancer in postmenopausal women using estradiol-progestin therapy. Obstet Gynecol. 2009;114(6):1197–1204.
Because unopposed estrogen raises the risk of endometrial hyperplasia and adenocarcinoma, we prescribe progestational therapy when a menopausal woman who has an intact uterus decides to use estrogen.
Some ObGyns prescribe continuous progestin in this setting; others, sequential progestin (e.g., continuous estrogen along with 14 days of progestin each month). Still others prescribe “long-cycle” sequential regimens comprising continuous estrogen plus 2 weeks of progestin every 3 months. However, the long-term endometrial safety of these regimens has not been adequately explored.
In a national cohort study in Finland, Jaakkola and coworkers followed more than 200,000 menopausal women who used estrogen plus progestin (E+P) HT from 1994 to 2006. The incidence of endometrial cancer in these women was compared with that of all women in Finland. Overall, 1,400 newly diagnosed cases of endometrial cancer were identified.
Investigators found that, compared with the general population:
- E+P users overall had a 54% greater risk of endometrioid endometrial adenocarcinoma, the tumor most closely associated with use of unopposed estrogen
- monthly sequential E+P users had a 69% (5 years’ use) and 156% (10 years’ use) greater risk of this tumor
- at 5 and 10 years of use, long-cycle sequential HT was associated with a substantially higher risk of endometrial adenocarcinoma than was monthly sequential HT
- the endometrial safety profiles of transdermal and oral sequential HT were comparable; so were those of medroxyprogesterone acetate and norethindrone acetate
- in contrast to sequential HT regimens, continuous E+P was associated with a lower risk of endometrial cancer than that found in the overall population (76% risk reduction after 3 to 5 years of use).
These important findings clarify the safety profiles of long-term continuous E+P, which is protective, and sequential E+P, which substantially raises the risk of endometrial adenocarcinoma.
The authors estimate that, for every 1,000 women who use E+P for 10 years, eight additional cases of cancer will be diagnosed if that therapy is sequential and monthly; on the other hand, three or four fewer cases will be diagnosed if HT is continuous.
Women who use E+P hormone therapy should take the progestin continuously rather than sequentially.
Any woman who uses sequential progestin over the long term should undergo regular endometrial monitoring via transvaginal ultrasonography, endometrial biopsy, or both.
Guidance on who to assess for osteoporosis—and when to treat them
The North American Menopause Society. NAMS continuing medical education activity: Management of osteoporosis in postmenopausal women: 2010 position statement of The North American Menopause Society. Menopause. 2010;17(1):23–24.
National Osteoporosis Foundation. Clinician’s Guide to Prevention and Treatment of Osteoporosis. Washington, D.C.: NOF: 2008. http://www.nof.org/professionals/NOF_Clinicians_Guide.pdf. Accessed April 1, 2010.
Kaunitz AM, McClung MR, Feldman RG, Wysocki S. Postmenopausal osteoporosis: fracture risk and prevention. OBG Management. 2009;21(10)(suppl):S1–6.
The North American Menopause Society (NAMS) has updated its position statement on the management of osteoporosis in postmenopausal women, incorporating many recommendations from guidelines issued in 2008 by the National Osteoporosis Foundation. You may find the recommendations on bone mineral density (BMD) assessment and use of medications to prevent fractures of particular interest.
BMD assessment should focus on women at high risk of fracture
In the United States, many women at low risk of fracture begin BMD assessment in their 50s via dual-energy x-ray absorptiometry (DXA) imaging of the lumbar spine and hip (FIGURE). As a result, many of these menopausal women are given a diagnosis of low bone mass (often termed “osteopenia” when in fact their DXA findings do not meet the criteria for osteoporosis) and are given a prescription for a bisphosphonate, often of indefinite duration.
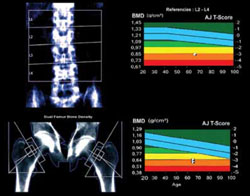
FIGURE When DXA imaging reveals low BMD
DXA scan of the lumbar spine and both hips showing osteoporosis in a 69-year-old woman. The colored graphs show the range of bone mineral density (BMD) across eight decades. The blue bars represent normal bone density; the white squares within the orange and red bars denote this patient’s low BMD. NAMS’s latest position statement clarifies exactly which women should be assessed, concluding that BMD measurement is appropriate in postmenopausal women who are not only at least 50 years old, but who also have one or more of the following risk factors for fracture:
- a history of fracture after menopause
- body mass index (BMI) <21 kg/m2 or weight <127 lb
- parental history of hip fracture
- current smoking, rheumatoid arthritis, or excessive alcohol intake (i.e., three or more drinks per day).
In the absence of these risk factors, BMD assessment should begin at 65 years of age.
Once initiated, treatment is usually long-term
The NAMS statement indicates that drug therapy—using bisphosphonates as first-line agents—is appropriate in postmenopausal women who have any of the following:
- a history of osteoporotic hip or vertebral fracture
- DXA-defined T-score lower than –2.5, indicating osteoporosis
- T-score from –1.0 to –2.5 (low bone mass) plus either a FRAX score that indicates a 10-year risk of hip fracture of at least 3% or a 10-year overall risk of osteoporotic fracture of at least 20%.
The World Health Organization (WHO) Fracture Risk Algorithm (FRAX) was developed to calculate the 10-year risk of hip fracture and the 10-year overall risk of osteoporotic fracture (online at http://www.shef.ac.uk/FRAX/). (For a description of two menopausal cases in which this Web site was used to determine appropriate clinical management, see the article by Kaunitz and colleagues cited above. Also, be aware that use of the FRAX Web site is inappropriate for making clinical decisions about women who are already using prescription antifracture therapy.)
NAMS points out that 1) treatment should usually span a period of years and 2) the risk of fracture after discontinuation of treatment has not been adequately studied.
Lasofoxifene seems unlikely to offer net benefits greater than what women obtain from existing therapies
Clinicians and menopausal women would welcome any agent that can prevent osteoporotic fracture with minimal adverse outcomes. Lasofoxifene appears to fulfill the first half of this equation, but its potential risks and questionable long-term effects cast doubt on its overall utility.
In a manufacturer-sponsored international trial, investigators randomized 8,556 women (mean age, 67 years) who met BMD criteria for osteoporosis to the selective estrogen-receptor modulator (SERM) lasofoxifene (0.25 mg daily or 0.5 mg daily) or placebo for 5 years. Women who received 0.5 mg daily of lasofoxifene had a substantially lower risk of vertebral (HR, 0.58) and nonvertebral (HR, 0.76) fracture than did women who received placebo.1
This dosage of lasofoxifene was also associated with a lower risk of estrogen-receptor–positive breast cancer (HR, 0.19), CAD events (HR, 0.68), and stroke (HR, 0.64), but a twofold higher risk of venous thromboembolic events overall and more than fourfold higher risk of pulmonary embolism. The incidence of endometrial cancer and endometrial hyperplasia was low (fewer than three women in each group), but endometrial polyps and hypertrophy were substantially more common among women who received either dosage of lasofoxifene.1
At 3 years, questionable benefit
Although these findings indicate that lasofoxifene lowers the risk of radiologically confirmed vertebral fracture, data submitted to the FDA reveal that the risk of clinical vertebral fracture was not reduced at 3 years.
Both raloxifene and lasofoxifene are associated with a heightened risk of venous thromboembolic events. Although lasofoxifene did not raise the risk of endometrial neoplasia or hyperplasia in the trial just described, the rates of other endometrial outcomes suggest that this agent has a proliferative effect on the endometrium.1
The reduced risk of estrogen-receptor–positive breast cancer and CAD events is intriguing. However, as an editorial writer points out, a clinician would need to treat 492 women for 1 year to prevent one major CAD event.2
Lasofoxifene does not seem to offer any clinically important benefit over existing SERMs. Moreover, alendronate, a generic bisphosphonate proven to prevent clinical vertebral and nonvertebral fracture, often fills the bill for the prevention of osteoporotic fracture in menopausal women.
References
1. Cummings SR, Ensrud K, Delmas PF, et al. Lasofoxifene in postmenopausal women with osteoporosis. N Engl J Med. 2010;362(8):686-696.
2. Becker C. Another selective estrogen-receptor modulator for osteoporosis. N Engl J Med. 2010;362(8):752-754.
BMD reassessment should be minimal
BMD assessment by means of DXA imaging to monitor the effects of therapy is appropriate after 1 or 2 years of treatment. Thereafter, repeat measurement is of little value in women whose BMD has stabilized or increased on therapy. A follow-up DXA scan is of limited use in predicting the effectiveness of antiresorptive therapy in lowering the risk of fracture. Moreover, changes in BMD can lag behind actual therapeutic benefits (i.e., fracture prevention).
In menopausal women who are not taking prescription antifracture therapy, the follow-up measurement of BMD is not useful until 2 to 5 years after initial testing. Although BMD may be lost rapidly in the initial years after menopause (or after discontinuation of HT), subsequently it plateaus or declines slowly.
If a woman is not using prescription antifracture therapy and is within 2 or 3 years of menopause (or if she has discontinued menopausal HT in the past 2 to 3 years), retesting in 2 years is prudent. However, if the same woman were 5 or more years post-menopausal and had not recently discontinued HT, follow-up BMD assessment can be deferred for 3 to 5 years.
This updated guidance from NAMS emphasizes that BMD need not be assessed in women younger than 65 years unless they have specific risk factors. FRAX evaluation also makes it possible to estimate the 10-year risk of fracture in women who have low bone mass but who do not meet criteria for osteoporosis. FRAX evaluation indicates that prescription therapy is rarely required for women in their 50s or 60s who have low bone mass (but not osteoporosis). For women in their 70s or 80s who have low bone mass (but not osteoporosis), however, FRAX evaluation often leads to a recommendation to initiate prescription antifracture treatment.
Many postmenopausal women who are in their 50s or 60s and who have T-scores of –1.0 to –2.5 are given bisphosphonates, despite being at low risk of fracture. Adherence to the NAMS guidelines will help prevent unnecessary assessment and treatment. In particular, the NAMS recommendations for follow-up BMD assessment—i.e., one-time evaluation 1 to 2 years after initiating therapy and no further assessment in women found to have stable BMD at the first follow-up DXA—should simplify clinical management in this setting.
Hold off on ordering DXA testing until women meet criteria for BMD assessment. In women who do not have osteoporosis, limit use of bisphosphonates to those who have an elevated 10-year risk of fracture, as assessed using the FRAX tool.
Our menopausal patients should be taking more vitamin D supplements
Bischoff-Ferrari H. Vitamin D: what is an adequate vitamin D level and how much supplementation is necessary? Best Pract Res Clin Rheumatol. 2009;23(6):789.
Bischoff-Ferrari HA, Dawson-Hughes B, Staehelin HB, et al. Fall prevention with supplemental and active forms of vitamin D: a meta-analysis of randomised controlled trials. BMJ. 2009;339:b3692.
Stewart JW, Alekel DL, Ritland LM, et al. Serum 25-hydroxyvitamin D is related to indicators of overall physical fitness in healthy postmenopausal women. Menopause. 2009;16(6):1093–1101.
Office of Dietary Supplements. Dietary Supplement Fact Sheet: Vitamin D. National Institutes of Health Web site. http://dietary-supplements.info.nih.gov/factsheets/vitamind.asp. Updated November 13, 2009. Accessed April 2, 2010.
We have long recognized the important role vitamin D plays in promoting calcium absorption from the gut and maintaining adequate serum calcium and phosphate concentrations to enable normal bone mineralization. Now, studies reveal that the vitamin also helps prevent falls and promotes overall fitness in menopausal women. It has also become clear that traditional targets for vitamin D supplementation are inadequate.
Two recent meta-analyses of double-blind, randomized trials concluded that vitamin D reduces the risk of falls in a dose-dependent manner. Dr. Heike Bischoff-Ferrari, a Swiss scientist and a leading vitamin D researcher, points out that 1) a minimum of 700 to 1,000 IU of vitamin D supplementation daily is appropriate in menopausal women and 2) a higher amount is indicated for those who are obese or deficient in vitamin D.
Compare the current recommended dietary allowance (RDA) for adults 51 to 70 years old: 400 IU daily. The federal Food and Nutrition Board is expected to update the vitamin D RDA this spring. The 2010 NAMS statement on osteoporosis recommends a daily vitamin D intake of 800 to 1,000 IU for menopausal women.
Ask your patient to add up the aggregate daily amount of vitamin D she ingests with her multivitamin and calcium and vitamin D supplements. If it is less than 800 IU, have her purchase over-the-counter vitamin D supplements (available in 400, 1,000, and 2,000 IU capsules). Obese patients and those known to be deficient may need to ingest higher daily amounts of vitamin D.
Much has changed in the management of menopausal women. The Women’s Health Initiative (WHI) and other trials shed light on the risk-benefit ratio of hormone therapy (HT) and significantly altered patterns of usage. A new fracture risk-assessment tool devised by the World Health Organization is now available for widespread use; it continues to be refined so that it can be applied to specific populations with greater accuracy. And the management of low bone mass and osteoporosis has evolved so that we can determine with greater precision exactly who merits our attention.
This year, the Update on Menopause describes:
- a reanalysis of WHI data, focusing on the relationship between hormone therapy and the risk of coronary artery disease (CAD)
- a study from Finland that explores the risk of endometrial cancer associated with various progestin regimens in women who are taking estrogen and who have an intact uterus
- guidance from the North American Menopause Society and the National Osteoporosis Foundation on who, how, and when to evaluate for a likelihood of fracture
- insight into the benefits of and need for vitamin D among menopausal women
- information on a new selective estrogen-receptor modulator under development.
Hormone therapy and CAD: Is the glass half full…or half empty?
Toh S, Hernández-Díaz S, Logan R, Rossouw JE, Hernán MA. Coronary heart disease in postmenopausal recipients of estrogen plus progestin therapy: Does the increased risk ever disappear? A randomized trial. Ann Intern Med. 2010;152(4):211–217.
North American Menopause Society. Position Statement: Estrogen and progestogens use in postmenopausal women: 2010 position statement of The North American Menopause Society. Menopause. 2010;17(2):242–255. DOI: 10.1097/gme.0b013e3181d0f6b9. http://www.menopause.org/PSht10.pdf. Accessed April 1, 2010.
When estrogen therapy is initiated within 10 years of menopause, it may reduce the risk of CAD, according to data from the WHI randomized trial and observational data.
The picture isn’t as clear in regard to estrogen-progestin HT. In a just-published study, Toh and colleagues reassessed data from the WHI trial of continuous oral conjugated equine estrogen plus medroxyprogesterone acetate versus placebo. They also compared the WHI findings with those of the large observational Nurses’ Health Study (NHS). Here are some of their findings:
- participants became less consistent in taking study medication over time—a finding of many long-term studies. This trend prompted Toh and colleagues to adjust their analysis for adherence
- among women who used HT within 10 years after the menopausal transition, the hazard ratio (HR) for CAD was 0.64 (95% confidence interval [CI], 0.21, 1.99) in the WHI and 0.68 (95% CI, 0.24, 1.91) in the NHS. Both hazard ratios suggest that the risk of CAD is lower in Ht users than in nonusers—although the difference is not significant
- when investigators pooled the WHI and NHS findings, the hazard ratio for CAD associated with combination HT was 0.66 (95% CI, 0.31, 1.42). Note that, as the number of participants increases, the confidence limits narrow.
Toh and colleagues concluded that their analysis demonstrated no diminished risk of CAD with HT use. My reading of these data is different: Combination HT does not increase the risk of CAD in women who have been postmenopausal for less than 10 years.
Focus on risk was unbalanced
The question of whether combination HT reduces the risk of CAD in younger women is somewhat moot. I am not aware of any ObGyn in the United States who uses HT to prevent CAD, and the great majority of symptomatic women who consider initiating HT have been menopausal for less than a decade. For these reasons, I find the conclusions drawn by Toh and colleagues a bit mystifying—and the title they chose for their study may be misleading:
Coronary heart disease in postmenopausal recipients of estrogen plus progestin therapy: Does the increased risk ever disappear?
Nevertheless, fear that HT might increase the risk of CAD is common among symptomatic menopausal women and their physicians. What this important analysis can offer is reassurance to symptomatic women who have been menopausal for less than 10 years: namely, that HT—estrogen alone or estrogen plus progestin—does not increase the risk of myocardial infarction or death from CAD.
You should counsel symptomatic women who have been menopausal less than 10 years that short-term use of estrogen therapy or estrogen-progestin therapy does not appear to increase their risk of CAD.
In women taking estrogen, sequential progestin therapy raises the risk of endometrial cancer
Jaakkola S, Lyytinen H, Pukkala E, Ylikorkala O. Endometrial cancer in postmenopausal women using estradiol-progestin therapy. Obstet Gynecol. 2009;114(6):1197–1204.
Because unopposed estrogen raises the risk of endometrial hyperplasia and adenocarcinoma, we prescribe progestational therapy when a menopausal woman who has an intact uterus decides to use estrogen.
Some ObGyns prescribe continuous progestin in this setting; others, sequential progestin (e.g., continuous estrogen along with 14 days of progestin each month). Still others prescribe “long-cycle” sequential regimens comprising continuous estrogen plus 2 weeks of progestin every 3 months. However, the long-term endometrial safety of these regimens has not been adequately explored.
In a national cohort study in Finland, Jaakkola and coworkers followed more than 200,000 menopausal women who used estrogen plus progestin (E+P) HT from 1994 to 2006. The incidence of endometrial cancer in these women was compared with that of all women in Finland. Overall, 1,400 newly diagnosed cases of endometrial cancer were identified.
Investigators found that, compared with the general population:
- E+P users overall had a 54% greater risk of endometrioid endometrial adenocarcinoma, the tumor most closely associated with use of unopposed estrogen
- monthly sequential E+P users had a 69% (5 years’ use) and 156% (10 years’ use) greater risk of this tumor
- at 5 and 10 years of use, long-cycle sequential HT was associated with a substantially higher risk of endometrial adenocarcinoma than was monthly sequential HT
- the endometrial safety profiles of transdermal and oral sequential HT were comparable; so were those of medroxyprogesterone acetate and norethindrone acetate
- in contrast to sequential HT regimens, continuous E+P was associated with a lower risk of endometrial cancer than that found in the overall population (76% risk reduction after 3 to 5 years of use).
These important findings clarify the safety profiles of long-term continuous E+P, which is protective, and sequential E+P, which substantially raises the risk of endometrial adenocarcinoma.
The authors estimate that, for every 1,000 women who use E+P for 10 years, eight additional cases of cancer will be diagnosed if that therapy is sequential and monthly; on the other hand, three or four fewer cases will be diagnosed if HT is continuous.
Women who use E+P hormone therapy should take the progestin continuously rather than sequentially.
Any woman who uses sequential progestin over the long term should undergo regular endometrial monitoring via transvaginal ultrasonography, endometrial biopsy, or both.
Guidance on who to assess for osteoporosis—and when to treat them
The North American Menopause Society. NAMS continuing medical education activity: Management of osteoporosis in postmenopausal women: 2010 position statement of The North American Menopause Society. Menopause. 2010;17(1):23–24.
National Osteoporosis Foundation. Clinician’s Guide to Prevention and Treatment of Osteoporosis. Washington, D.C.: NOF: 2008. http://www.nof.org/professionals/NOF_Clinicians_Guide.pdf. Accessed April 1, 2010.
Kaunitz AM, McClung MR, Feldman RG, Wysocki S. Postmenopausal osteoporosis: fracture risk and prevention. OBG Management. 2009;21(10)(suppl):S1–6.
The North American Menopause Society (NAMS) has updated its position statement on the management of osteoporosis in postmenopausal women, incorporating many recommendations from guidelines issued in 2008 by the National Osteoporosis Foundation. You may find the recommendations on bone mineral density (BMD) assessment and use of medications to prevent fractures of particular interest.
BMD assessment should focus on women at high risk of fracture
In the United States, many women at low risk of fracture begin BMD assessment in their 50s via dual-energy x-ray absorptiometry (DXA) imaging of the lumbar spine and hip (FIGURE). As a result, many of these menopausal women are given a diagnosis of low bone mass (often termed “osteopenia” when in fact their DXA findings do not meet the criteria for osteoporosis) and are given a prescription for a bisphosphonate, often of indefinite duration.

FIGURE When DXA imaging reveals low BMD
DXA scan of the lumbar spine and both hips showing osteoporosis in a 69-year-old woman. The colored graphs show the range of bone mineral density (BMD) across eight decades. The blue bars represent normal bone density; the white squares within the orange and red bars denote this patient’s low BMD. NAMS’s latest position statement clarifies exactly which women should be assessed, concluding that BMD measurement is appropriate in postmenopausal women who are not only at least 50 years old, but who also have one or more of the following risk factors for fracture:
- a history of fracture after menopause
- body mass index (BMI) <21 kg/m2 or weight <127 lb
- parental history of hip fracture
- current smoking, rheumatoid arthritis, or excessive alcohol intake (i.e., three or more drinks per day).
In the absence of these risk factors, BMD assessment should begin at 65 years of age.
Once initiated, treatment is usually long-term
The NAMS statement indicates that drug therapy—using bisphosphonates as first-line agents—is appropriate in postmenopausal women who have any of the following:
- a history of osteoporotic hip or vertebral fracture
- DXA-defined T-score lower than –2.5, indicating osteoporosis
- T-score from –1.0 to –2.5 (low bone mass) plus either a FRAX score that indicates a 10-year risk of hip fracture of at least 3% or a 10-year overall risk of osteoporotic fracture of at least 20%.
The World Health Organization (WHO) Fracture Risk Algorithm (FRAX) was developed to calculate the 10-year risk of hip fracture and the 10-year overall risk of osteoporotic fracture (online at http://www.shef.ac.uk/FRAX/). (For a description of two menopausal cases in which this Web site was used to determine appropriate clinical management, see the article by Kaunitz and colleagues cited above. Also, be aware that use of the FRAX Web site is inappropriate for making clinical decisions about women who are already using prescription antifracture therapy.)
NAMS points out that 1) treatment should usually span a period of years and 2) the risk of fracture after discontinuation of treatment has not been adequately studied.
Lasofoxifene seems unlikely to offer net benefits greater than what women obtain from existing therapies
Clinicians and menopausal women would welcome any agent that can prevent osteoporotic fracture with minimal adverse outcomes. Lasofoxifene appears to fulfill the first half of this equation, but its potential risks and questionable long-term effects cast doubt on its overall utility.
In a manufacturer-sponsored international trial, investigators randomized 8,556 women (mean age, 67 years) who met BMD criteria for osteoporosis to the selective estrogen-receptor modulator (SERM) lasofoxifene (0.25 mg daily or 0.5 mg daily) or placebo for 5 years. Women who received 0.5 mg daily of lasofoxifene had a substantially lower risk of vertebral (HR, 0.58) and nonvertebral (HR, 0.76) fracture than did women who received placebo.1
This dosage of lasofoxifene was also associated with a lower risk of estrogen-receptor–positive breast cancer (HR, 0.19), CAD events (HR, 0.68), and stroke (HR, 0.64), but a twofold higher risk of venous thromboembolic events overall and more than fourfold higher risk of pulmonary embolism. The incidence of endometrial cancer and endometrial hyperplasia was low (fewer than three women in each group), but endometrial polyps and hypertrophy were substantially more common among women who received either dosage of lasofoxifene.1
At 3 years, questionable benefit
Although these findings indicate that lasofoxifene lowers the risk of radiologically confirmed vertebral fracture, data submitted to the FDA reveal that the risk of clinical vertebral fracture was not reduced at 3 years.
Both raloxifene and lasofoxifene are associated with a heightened risk of venous thromboembolic events. Although lasofoxifene did not raise the risk of endometrial neoplasia or hyperplasia in the trial just described, the rates of other endometrial outcomes suggest that this agent has a proliferative effect on the endometrium.1
The reduced risk of estrogen-receptor–positive breast cancer and CAD events is intriguing. However, as an editorial writer points out, a clinician would need to treat 492 women for 1 year to prevent one major CAD event.2
Lasofoxifene does not seem to offer any clinically important benefit over existing SERMs. Moreover, alendronate, a generic bisphosphonate proven to prevent clinical vertebral and nonvertebral fracture, often fills the bill for the prevention of osteoporotic fracture in menopausal women.
References
1. Cummings SR, Ensrud K, Delmas PF, et al. Lasofoxifene in postmenopausal women with osteoporosis. N Engl J Med. 2010;362(8):686-696.
2. Becker C. Another selective estrogen-receptor modulator for osteoporosis. N Engl J Med. 2010;362(8):752-754.
BMD reassessment should be minimal
BMD assessment by means of DXA imaging to monitor the effects of therapy is appropriate after 1 or 2 years of treatment. Thereafter, repeat measurement is of little value in women whose BMD has stabilized or increased on therapy. A follow-up DXA scan is of limited use in predicting the effectiveness of antiresorptive therapy in lowering the risk of fracture. Moreover, changes in BMD can lag behind actual therapeutic benefits (i.e., fracture prevention).
In menopausal women who are not taking prescription antifracture therapy, the follow-up measurement of BMD is not useful until 2 to 5 years after initial testing. Although BMD may be lost rapidly in the initial years after menopause (or after discontinuation of HT), subsequently it plateaus or declines slowly.
If a woman is not using prescription antifracture therapy and is within 2 or 3 years of menopause (or if she has discontinued menopausal HT in the past 2 to 3 years), retesting in 2 years is prudent. However, if the same woman were 5 or more years post-menopausal and had not recently discontinued HT, follow-up BMD assessment can be deferred for 3 to 5 years.
This updated guidance from NAMS emphasizes that BMD need not be assessed in women younger than 65 years unless they have specific risk factors. FRAX evaluation also makes it possible to estimate the 10-year risk of fracture in women who have low bone mass but who do not meet criteria for osteoporosis. FRAX evaluation indicates that prescription therapy is rarely required for women in their 50s or 60s who have low bone mass (but not osteoporosis). For women in their 70s or 80s who have low bone mass (but not osteoporosis), however, FRAX evaluation often leads to a recommendation to initiate prescription antifracture treatment.
Many postmenopausal women who are in their 50s or 60s and who have T-scores of –1.0 to –2.5 are given bisphosphonates, despite being at low risk of fracture. Adherence to the NAMS guidelines will help prevent unnecessary assessment and treatment. In particular, the NAMS recommendations for follow-up BMD assessment—i.e., one-time evaluation 1 to 2 years after initiating therapy and no further assessment in women found to have stable BMD at the first follow-up DXA—should simplify clinical management in this setting.
Hold off on ordering DXA testing until women meet criteria for BMD assessment. In women who do not have osteoporosis, limit use of bisphosphonates to those who have an elevated 10-year risk of fracture, as assessed using the FRAX tool.
Our menopausal patients should be taking more vitamin D supplements
Bischoff-Ferrari H. Vitamin D: what is an adequate vitamin D level and how much supplementation is necessary? Best Pract Res Clin Rheumatol. 2009;23(6):789.
Bischoff-Ferrari HA, Dawson-Hughes B, Staehelin HB, et al. Fall prevention with supplemental and active forms of vitamin D: a meta-analysis of randomised controlled trials. BMJ. 2009;339:b3692.
Stewart JW, Alekel DL, Ritland LM, et al. Serum 25-hydroxyvitamin D is related to indicators of overall physical fitness in healthy postmenopausal women. Menopause. 2009;16(6):1093–1101.
Office of Dietary Supplements. Dietary Supplement Fact Sheet: Vitamin D. National Institutes of Health Web site. http://dietary-supplements.info.nih.gov/factsheets/vitamind.asp. Updated November 13, 2009. Accessed April 2, 2010.
We have long recognized the important role vitamin D plays in promoting calcium absorption from the gut and maintaining adequate serum calcium and phosphate concentrations to enable normal bone mineralization. Now, studies reveal that the vitamin also helps prevent falls and promotes overall fitness in menopausal women. It has also become clear that traditional targets for vitamin D supplementation are inadequate.
Two recent meta-analyses of double-blind, randomized trials concluded that vitamin D reduces the risk of falls in a dose-dependent manner. Dr. Heike Bischoff-Ferrari, a Swiss scientist and a leading vitamin D researcher, points out that 1) a minimum of 700 to 1,000 IU of vitamin D supplementation daily is appropriate in menopausal women and 2) a higher amount is indicated for those who are obese or deficient in vitamin D.
Compare the current recommended dietary allowance (RDA) for adults 51 to 70 years old: 400 IU daily. The federal Food and Nutrition Board is expected to update the vitamin D RDA this spring. The 2010 NAMS statement on osteoporosis recommends a daily vitamin D intake of 800 to 1,000 IU for menopausal women.
Ask your patient to add up the aggregate daily amount of vitamin D she ingests with her multivitamin and calcium and vitamin D supplements. If it is less than 800 IU, have her purchase over-the-counter vitamin D supplements (available in 400, 1,000, and 2,000 IU capsules). Obese patients and those known to be deficient may need to ingest higher daily amounts of vitamin D.
Much has changed in the management of menopausal women. The Women’s Health Initiative (WHI) and other trials shed light on the risk-benefit ratio of hormone therapy (HT) and significantly altered patterns of usage. A new fracture risk-assessment tool devised by the World Health Organization is now available for widespread use; it continues to be refined so that it can be applied to specific populations with greater accuracy. And the management of low bone mass and osteoporosis has evolved so that we can determine with greater precision exactly who merits our attention.
This year, the Update on Menopause describes:
- a reanalysis of WHI data, focusing on the relationship between hormone therapy and the risk of coronary artery disease (CAD)
- a study from Finland that explores the risk of endometrial cancer associated with various progestin regimens in women who are taking estrogen and who have an intact uterus
- guidance from the North American Menopause Society and the National Osteoporosis Foundation on who, how, and when to evaluate for a likelihood of fracture
- insight into the benefits of and need for vitamin D among menopausal women
- information on a new selective estrogen-receptor modulator under development.
Hormone therapy and CAD: Is the glass half full…or half empty?
Toh S, Hernández-Díaz S, Logan R, Rossouw JE, Hernán MA. Coronary heart disease in postmenopausal recipients of estrogen plus progestin therapy: Does the increased risk ever disappear? A randomized trial. Ann Intern Med. 2010;152(4):211–217.
North American Menopause Society. Position Statement: Estrogen and progestogens use in postmenopausal women: 2010 position statement of The North American Menopause Society. Menopause. 2010;17(2):242–255. DOI: 10.1097/gme.0b013e3181d0f6b9. http://www.menopause.org/PSht10.pdf. Accessed April 1, 2010.
When estrogen therapy is initiated within 10 years of menopause, it may reduce the risk of CAD, according to data from the WHI randomized trial and observational data.
The picture isn’t as clear in regard to estrogen-progestin HT. In a just-published study, Toh and colleagues reassessed data from the WHI trial of continuous oral conjugated equine estrogen plus medroxyprogesterone acetate versus placebo. They also compared the WHI findings with those of the large observational Nurses’ Health Study (NHS). Here are some of their findings:
- participants became less consistent in taking study medication over time—a finding of many long-term studies. This trend prompted Toh and colleagues to adjust their analysis for adherence
- among women who used HT within 10 years after the menopausal transition, the hazard ratio (HR) for CAD was 0.64 (95% confidence interval [CI], 0.21, 1.99) in the WHI and 0.68 (95% CI, 0.24, 1.91) in the NHS. Both hazard ratios suggest that the risk of CAD is lower in Ht users than in nonusers—although the difference is not significant
- when investigators pooled the WHI and NHS findings, the hazard ratio for CAD associated with combination HT was 0.66 (95% CI, 0.31, 1.42). Note that, as the number of participants increases, the confidence limits narrow.
Toh and colleagues concluded that their analysis demonstrated no diminished risk of CAD with HT use. My reading of these data is different: Combination HT does not increase the risk of CAD in women who have been postmenopausal for less than 10 years.
Focus on risk was unbalanced
The question of whether combination HT reduces the risk of CAD in younger women is somewhat moot. I am not aware of any ObGyn in the United States who uses HT to prevent CAD, and the great majority of symptomatic women who consider initiating HT have been menopausal for less than a decade. For these reasons, I find the conclusions drawn by Toh and colleagues a bit mystifying—and the title they chose for their study may be misleading:
Coronary heart disease in postmenopausal recipients of estrogen plus progestin therapy: Does the increased risk ever disappear?
Nevertheless, fear that HT might increase the risk of CAD is common among symptomatic menopausal women and their physicians. What this important analysis can offer is reassurance to symptomatic women who have been menopausal for less than 10 years: namely, that HT—estrogen alone or estrogen plus progestin—does not increase the risk of myocardial infarction or death from CAD.
You should counsel symptomatic women who have been menopausal less than 10 years that short-term use of estrogen therapy or estrogen-progestin therapy does not appear to increase their risk of CAD.
In women taking estrogen, sequential progestin therapy raises the risk of endometrial cancer
Jaakkola S, Lyytinen H, Pukkala E, Ylikorkala O. Endometrial cancer in postmenopausal women using estradiol-progestin therapy. Obstet Gynecol. 2009;114(6):1197–1204.
Because unopposed estrogen raises the risk of endometrial hyperplasia and adenocarcinoma, we prescribe progestational therapy when a menopausal woman who has an intact uterus decides to use estrogen.
Some ObGyns prescribe continuous progestin in this setting; others, sequential progestin (e.g., continuous estrogen along with 14 days of progestin each month). Still others prescribe “long-cycle” sequential regimens comprising continuous estrogen plus 2 weeks of progestin every 3 months. However, the long-term endometrial safety of these regimens has not been adequately explored.
In a national cohort study in Finland, Jaakkola and coworkers followed more than 200,000 menopausal women who used estrogen plus progestin (E+P) HT from 1994 to 2006. The incidence of endometrial cancer in these women was compared with that of all women in Finland. Overall, 1,400 newly diagnosed cases of endometrial cancer were identified.
Investigators found that, compared with the general population:
- E+P users overall had a 54% greater risk of endometrioid endometrial adenocarcinoma, the tumor most closely associated with use of unopposed estrogen
- monthly sequential E+P users had a 69% (5 years’ use) and 156% (10 years’ use) greater risk of this tumor
- at 5 and 10 years of use, long-cycle sequential HT was associated with a substantially higher risk of endometrial adenocarcinoma than was monthly sequential HT
- the endometrial safety profiles of transdermal and oral sequential HT were comparable; so were those of medroxyprogesterone acetate and norethindrone acetate
- in contrast to sequential HT regimens, continuous E+P was associated with a lower risk of endometrial cancer than that found in the overall population (76% risk reduction after 3 to 5 years of use).
These important findings clarify the safety profiles of long-term continuous E+P, which is protective, and sequential E+P, which substantially raises the risk of endometrial adenocarcinoma.
The authors estimate that, for every 1,000 women who use E+P for 10 years, eight additional cases of cancer will be diagnosed if that therapy is sequential and monthly; on the other hand, three or four fewer cases will be diagnosed if HT is continuous.
Women who use E+P hormone therapy should take the progestin continuously rather than sequentially.
Any woman who uses sequential progestin over the long term should undergo regular endometrial monitoring via transvaginal ultrasonography, endometrial biopsy, or both.
Guidance on who to assess for osteoporosis—and when to treat them
The North American Menopause Society. NAMS continuing medical education activity: Management of osteoporosis in postmenopausal women: 2010 position statement of The North American Menopause Society. Menopause. 2010;17(1):23–24.
National Osteoporosis Foundation. Clinician’s Guide to Prevention and Treatment of Osteoporosis. Washington, D.C.: NOF: 2008. http://www.nof.org/professionals/NOF_Clinicians_Guide.pdf. Accessed April 1, 2010.
Kaunitz AM, McClung MR, Feldman RG, Wysocki S. Postmenopausal osteoporosis: fracture risk and prevention. OBG Management. 2009;21(10)(suppl):S1–6.
The North American Menopause Society (NAMS) has updated its position statement on the management of osteoporosis in postmenopausal women, incorporating many recommendations from guidelines issued in 2008 by the National Osteoporosis Foundation. You may find the recommendations on bone mineral density (BMD) assessment and use of medications to prevent fractures of particular interest.
BMD assessment should focus on women at high risk of fracture
In the United States, many women at low risk of fracture begin BMD assessment in their 50s via dual-energy x-ray absorptiometry (DXA) imaging of the lumbar spine and hip (FIGURE). As a result, many of these menopausal women are given a diagnosis of low bone mass (often termed “osteopenia” when in fact their DXA findings do not meet the criteria for osteoporosis) and are given a prescription for a bisphosphonate, often of indefinite duration.

FIGURE When DXA imaging reveals low BMD
DXA scan of the lumbar spine and both hips showing osteoporosis in a 69-year-old woman. The colored graphs show the range of bone mineral density (BMD) across eight decades. The blue bars represent normal bone density; the white squares within the orange and red bars denote this patient’s low BMD. NAMS’s latest position statement clarifies exactly which women should be assessed, concluding that BMD measurement is appropriate in postmenopausal women who are not only at least 50 years old, but who also have one or more of the following risk factors for fracture:
- a history of fracture after menopause
- body mass index (BMI) <21 kg/m2 or weight <127 lb
- parental history of hip fracture
- current smoking, rheumatoid arthritis, or excessive alcohol intake (i.e., three or more drinks per day).
In the absence of these risk factors, BMD assessment should begin at 65 years of age.
Once initiated, treatment is usually long-term
The NAMS statement indicates that drug therapy—using bisphosphonates as first-line agents—is appropriate in postmenopausal women who have any of the following:
- a history of osteoporotic hip or vertebral fracture
- DXA-defined T-score lower than –2.5, indicating osteoporosis
- T-score from –1.0 to –2.5 (low bone mass) plus either a FRAX score that indicates a 10-year risk of hip fracture of at least 3% or a 10-year overall risk of osteoporotic fracture of at least 20%.
The World Health Organization (WHO) Fracture Risk Algorithm (FRAX) was developed to calculate the 10-year risk of hip fracture and the 10-year overall risk of osteoporotic fracture (online at http://www.shef.ac.uk/FRAX/). (For a description of two menopausal cases in which this Web site was used to determine appropriate clinical management, see the article by Kaunitz and colleagues cited above. Also, be aware that use of the FRAX Web site is inappropriate for making clinical decisions about women who are already using prescription antifracture therapy.)
NAMS points out that 1) treatment should usually span a period of years and 2) the risk of fracture after discontinuation of treatment has not been adequately studied.
Lasofoxifene seems unlikely to offer net benefits greater than what women obtain from existing therapies
Clinicians and menopausal women would welcome any agent that can prevent osteoporotic fracture with minimal adverse outcomes. Lasofoxifene appears to fulfill the first half of this equation, but its potential risks and questionable long-term effects cast doubt on its overall utility.
In a manufacturer-sponsored international trial, investigators randomized 8,556 women (mean age, 67 years) who met BMD criteria for osteoporosis to the selective estrogen-receptor modulator (SERM) lasofoxifene (0.25 mg daily or 0.5 mg daily) or placebo for 5 years. Women who received 0.5 mg daily of lasofoxifene had a substantially lower risk of vertebral (HR, 0.58) and nonvertebral (HR, 0.76) fracture than did women who received placebo.1
This dosage of lasofoxifene was also associated with a lower risk of estrogen-receptor–positive breast cancer (HR, 0.19), CAD events (HR, 0.68), and stroke (HR, 0.64), but a twofold higher risk of venous thromboembolic events overall and more than fourfold higher risk of pulmonary embolism. The incidence of endometrial cancer and endometrial hyperplasia was low (fewer than three women in each group), but endometrial polyps and hypertrophy were substantially more common among women who received either dosage of lasofoxifene.1
At 3 years, questionable benefit
Although these findings indicate that lasofoxifene lowers the risk of radiologically confirmed vertebral fracture, data submitted to the FDA reveal that the risk of clinical vertebral fracture was not reduced at 3 years.
Both raloxifene and lasofoxifene are associated with a heightened risk of venous thromboembolic events. Although lasofoxifene did not raise the risk of endometrial neoplasia or hyperplasia in the trial just described, the rates of other endometrial outcomes suggest that this agent has a proliferative effect on the endometrium.1
The reduced risk of estrogen-receptor–positive breast cancer and CAD events is intriguing. However, as an editorial writer points out, a clinician would need to treat 492 women for 1 year to prevent one major CAD event.2
Lasofoxifene does not seem to offer any clinically important benefit over existing SERMs. Moreover, alendronate, a generic bisphosphonate proven to prevent clinical vertebral and nonvertebral fracture, often fills the bill for the prevention of osteoporotic fracture in menopausal women.
References
1. Cummings SR, Ensrud K, Delmas PF, et al. Lasofoxifene in postmenopausal women with osteoporosis. N Engl J Med. 2010;362(8):686-696.
2. Becker C. Another selective estrogen-receptor modulator for osteoporosis. N Engl J Med. 2010;362(8):752-754.
BMD reassessment should be minimal
BMD assessment by means of DXA imaging to monitor the effects of therapy is appropriate after 1 or 2 years of treatment. Thereafter, repeat measurement is of little value in women whose BMD has stabilized or increased on therapy. A follow-up DXA scan is of limited use in predicting the effectiveness of antiresorptive therapy in lowering the risk of fracture. Moreover, changes in BMD can lag behind actual therapeutic benefits (i.e., fracture prevention).
In menopausal women who are not taking prescription antifracture therapy, the follow-up measurement of BMD is not useful until 2 to 5 years after initial testing. Although BMD may be lost rapidly in the initial years after menopause (or after discontinuation of HT), subsequently it plateaus or declines slowly.
If a woman is not using prescription antifracture therapy and is within 2 or 3 years of menopause (or if she has discontinued menopausal HT in the past 2 to 3 years), retesting in 2 years is prudent. However, if the same woman were 5 or more years post-menopausal and had not recently discontinued HT, follow-up BMD assessment can be deferred for 3 to 5 years.
This updated guidance from NAMS emphasizes that BMD need not be assessed in women younger than 65 years unless they have specific risk factors. FRAX evaluation also makes it possible to estimate the 10-year risk of fracture in women who have low bone mass but who do not meet criteria for osteoporosis. FRAX evaluation indicates that prescription therapy is rarely required for women in their 50s or 60s who have low bone mass (but not osteoporosis). For women in their 70s or 80s who have low bone mass (but not osteoporosis), however, FRAX evaluation often leads to a recommendation to initiate prescription antifracture treatment.
Many postmenopausal women who are in their 50s or 60s and who have T-scores of –1.0 to –2.5 are given bisphosphonates, despite being at low risk of fracture. Adherence to the NAMS guidelines will help prevent unnecessary assessment and treatment. In particular, the NAMS recommendations for follow-up BMD assessment—i.e., one-time evaluation 1 to 2 years after initiating therapy and no further assessment in women found to have stable BMD at the first follow-up DXA—should simplify clinical management in this setting.
Hold off on ordering DXA testing until women meet criteria for BMD assessment. In women who do not have osteoporosis, limit use of bisphosphonates to those who have an elevated 10-year risk of fracture, as assessed using the FRAX tool.
Our menopausal patients should be taking more vitamin D supplements
Bischoff-Ferrari H. Vitamin D: what is an adequate vitamin D level and how much supplementation is necessary? Best Pract Res Clin Rheumatol. 2009;23(6):789.
Bischoff-Ferrari HA, Dawson-Hughes B, Staehelin HB, et al. Fall prevention with supplemental and active forms of vitamin D: a meta-analysis of randomised controlled trials. BMJ. 2009;339:b3692.
Stewart JW, Alekel DL, Ritland LM, et al. Serum 25-hydroxyvitamin D is related to indicators of overall physical fitness in healthy postmenopausal women. Menopause. 2009;16(6):1093–1101.
Office of Dietary Supplements. Dietary Supplement Fact Sheet: Vitamin D. National Institutes of Health Web site. http://dietary-supplements.info.nih.gov/factsheets/vitamind.asp. Updated November 13, 2009. Accessed April 2, 2010.
We have long recognized the important role vitamin D plays in promoting calcium absorption from the gut and maintaining adequate serum calcium and phosphate concentrations to enable normal bone mineralization. Now, studies reveal that the vitamin also helps prevent falls and promotes overall fitness in menopausal women. It has also become clear that traditional targets for vitamin D supplementation are inadequate.
Two recent meta-analyses of double-blind, randomized trials concluded that vitamin D reduces the risk of falls in a dose-dependent manner. Dr. Heike Bischoff-Ferrari, a Swiss scientist and a leading vitamin D researcher, points out that 1) a minimum of 700 to 1,000 IU of vitamin D supplementation daily is appropriate in menopausal women and 2) a higher amount is indicated for those who are obese or deficient in vitamin D.
Compare the current recommended dietary allowance (RDA) for adults 51 to 70 years old: 400 IU daily. The federal Food and Nutrition Board is expected to update the vitamin D RDA this spring. The 2010 NAMS statement on osteoporosis recommends a daily vitamin D intake of 800 to 1,000 IU for menopausal women.
Ask your patient to add up the aggregate daily amount of vitamin D she ingests with her multivitamin and calcium and vitamin D supplements. If it is less than 800 IU, have her purchase over-the-counter vitamin D supplements (available in 400, 1,000, and 2,000 IU capsules). Obese patients and those known to be deficient may need to ingest higher daily amounts of vitamin D.
How to manage a short cervix to lower the risk of preterm delivery
CASE 1: Short cervix in the middle trimester
During routine second-trimester ultrasonography, a 34-year-old primigravida at 22 weeks’ gestation is found to have a cervix 15 mm in length. She has no other risk factors for spontaneous preterm birth.
What steps do you take to ensure that her pregnancy progresses uneventfully to term?
Cervical length is not routinely measured in low-risk women—i.e., those without a history of spontaneous preterm birth—but a short cervix is sometimes detected during ultrasonographic imaging for other indications, as it was in this case. When a short cervix is detected incidentally, I educate the patient to watch for early warning signs of preterm labor. I also recommend pelvic rest and a sedentary lifestyle.
The rate of preterm birth declined slightly in 2007—the first decrease in more than 20 years—but the phenomenon remains the leading cause of perinatal morbidity and mortality in developed nations.1 In the United States, more than 500,000 babies each year, or 12% to 13% of all births, are delivered before 37 weeks’ gestation. Most of these births are spontaneous and involve preterm labor or premature rupture of membranes. Medical costs for a preterm newborn exceed those of a term infant by a multiple of more than 10, and the average hospitalization exceeds that of a term infant by a multiple of more than six.
In this article, I discuss the rationale and technique for ultrasonographic cervical measurement to determine the likelihood of preterm birth. I also examine the data on the short cervix in various settings, and describe strategies for cervical assessment and preterm birth prevention, including cerclage and progesterone, framing the discussion in terms of gestational age.
Accurate and reproducible measurement of cervical length depends on correct technique. Use of transvaginal ultrasonography (TVUS) limits variations between measurements to 5% to 10%, a marked improvement over digital examination and transabdominal US.
Here are the five steps involved, in the order performed:
- Ensure that the patient’s bladder is empty. This precaution is necessary to prevent dynamic, or spontaneous, lengthening or shortening of the cervix.
- Counsel and position the patient. Explain the procedure to the patient and have her assume the dorsal lithotomy position.
- Introduce the probe into the anterior vaginal fornix using real-time visualization, and obtain a mid-sagittal view of the cervix. Withdraw the probe just enough to allow the image to blur, then advance the probe just enough for the image to regain clarity. This sequence prevents the practitioner from exerting excessive pressure on the cervix, which can falsely elongate it.
- Place an electronic caliper (on-screen) at the notch that represents the internal cervical os, and another at the external os (FIGURE 1).
- Measure the distance between the notches and report the shortest of three separately obtained measurements.
FIGURE 1 How to measure the cervix
Electronic calipers mark the internal and external os in this cervix measuring 42 mm via midsagittal transvaginal ultrasonography.
Watch for these pitfalls!
Before 20 weeks’ gestation, the lower uterine segment is not particularly well developed, making it difficult to reliably determine the location of the internal os (FIGURE 2). Moreover, focal myometrial contractions of the lower uterine segment, which are common, may give the false impression of increased cervical length or dilation of the internal os (FIGURE 2).24
FIGURE 2 Measurement may be difficult in early pregnancy
A. This transvaginal sonogram demonstrates the difficulty of determining the location of the internal os when the lower uterine segment is undeveloped (arrows). B. When lower uterine contractions occur with the anterior and posterior walls in opposition (arrowheads), transvaginal imaging may give the false appearance of dilation with funneling (arrow).
Why we assess cervical length
In the past, the cervix was viewed as either competent—i.e., capable of maintaining a pregnancy until term—or as “incompetent.” More recent evidence has broadened our understanding of cervical function, which is now viewed along a continuum.
In landmark research in the mid-1990s, investigators compared cervical lengths, measured via transvaginal ultrasonography (TVUS), between two groups of pregnant women—those who had a history of preterm birth and those who did not.
The result? Gestational age at delivery in the first pregnancy correlated significantly—and continuously—with cervical length between 20 and 30 weeks’ gestation in the subsequent pregnancy.2 Investigators also observed that the risk of spontaneous preterm birth increased with decreasing cervical length. A length of 25 mm (10th percentile) offered a clinically appropriate threshold for identification of the risk of preterm delivery.3
The value of cervical-length measurement lies in its high negative predictive value for recurrent spontaneous preterm birth.4 As a general rule of thumb, routine assessment of cervical length in asymptomatic women who do not have a history of preterm birth is not recommended because of the high rate of false-positive results and the low positive predictive value for preterm delivery. We also lack an evidence-based consensus on how to manage an abnormally short cervix in these women.
CASE RESOLVED
The patient is counseled to watch for signs of preterm labor and to remain sedentary. Her pregnancy progresses without incident until 40 weeks, when she undergoes induction of labor for oligohydramnios and delivers a healthy infant weighing 3,855 g.
When a short cervix is detected at less than 20 weeks
If a woman has a history of spontaneous preterm birth, a short cervix this early in gestation raises the question of cervical insufficiency. No objective criteria have been devised to identify this condition. Nor is there a widely accepted definition. A reasonable description does exist, however:
- …a clinical diagnosis characterized by recurrent painless dilation and spontaneous midtrimester birth, generally in the absence of predisposing conditions such as spontaneous membrane rupture, bleeding, and infection, characteristics that shift the presumed underlying cause away from cervical incompetence and support other components of the preterm birth syndrome.5
A patient who fits this description may be a candidate for cervical cerclage. Alternatively, it is reasonable to reassess the patient in 3 to 7 days, after restricting physical activity, keeping in mind the pitfalls of TVUS assessment of the lower uterine segment in early gestation (see the box on imaging). If cervical length remains short, consider cerclage.6,7
Watch for inflammation
Occasionally, echogenic material is observed in the amniotic fluid at the level of a short cervix. This debris is an inflammatory exudate of fibrin, white blood cells, and bacteria. The presence of this sludge (FIGURE 3) signifies a risk of preterm birth much greater than that associated with a short cervix alone.8 Because cerclage in the presence of inflammation may further heighten the risk of spontaneous preterm birth, I recommend caution.
FIGURE 3 Watch for signs of inflammation
When inflammatory exudate (asterisk) is identified at the level of a short cervix, the risk of spontaneous preterm birth is elevated beyond the risk associated with a short cervix alone.
When a short cervix is detected between 20 and 24 weeks
CASE 2: Is recurrent preterm birth likely?
A 30-year-old woman 21 weeks pregnant with her second child reports for TVUS. Because her first child was delivered preterm at 30 weeks, she has been undergoing periodic measurement of her cervix. Until today, it has been longer than 25 mm, but now it is 20 mm. What is the best strategy to avert another preterm birth?
If a short cervix (<25 mm) is noted in a high-risk patient at this gestational age, consider the possibility of preterm labor and ruptured membranes. If these conditions are present, they should be managed according to existing guidelines.4,9 If they are absent, consider cerclage.
A recent meta-analysis of randomized trials of cerclage for the prevention of preterm birth in a singleton, high-risk pregnancy with a short cervix suggests that cerclage is associated with a significantly lower risk of delivery before 35 weeks’ gestation (relative risk [RR], 0.61; 95% confidence interval [CI], 0.40–0.92). Among singleton pregnancies involving both a short cervix and a history of midtrimester loss, cerclage is again associated with a reduced likelihood of delivery before 35 weeks (39% vs 23.4%; number needed to treat [NNT], 8; RR, 0.57; 95% CI, 0.33–0.99).10 A recent randomized trial of cerclage versus no cerclage in women who had a history of spontaneous preterm birth produced similar findings.11 This trial is described in detail in the box.
Another randomized trial compared the relative merits of ultrasound-indicated cerclage for a cervix shorter than 20 mm to a history-indicated cerclage among women with a prior spontaneous preterm birth between 16 and 34 weeks. Thirty-nine of 123 women randomized to the former group received a cerclage, as did 25 of 125 subjects in the latter group. There were no significant differences in the primary outcome of delivery before 34 weeks or secondary measures of loss before 24 weeks, preterm premature membrane rupture, mean gestational age at delivery, or neonatal outcomes. This study was not designed or powered to evaluate the efficacy of cerclage in preventing recurrent, spontaneous preterm birth. It simply compares two ways of selecting high-risk women for cerclage.12
We lack data supporting placement of cerclage for an incidentally detected short cervix in women who lack a history of spontaneous preterm birth or midtrimester loss.7
Other groups known to be at increased risk of spontaneous preterm birth include women carrying twins and women who have undergone cervical cone biopsy or loop electrosurgical excision procedure (LEEP). Among twin gestations, cerclage for a short cervix is associated with an increased rate of preterm birth (<35 weeks).10 Cerclage for a short cervix has not been evaluated among women who have a history of LEEP.6
CASE RESOLVED
After preterm labor is ruled out, the patient is counseled about her options and chooses cervical cerclage. Her pregnancy proceeds uneventfully until 36 weeks’ gestation, when she delivers a healthy infant weighing 2,950 g.
When a short cervix is detected between 24 and 34 weeks
CASE 3: Is it preterm labor?
A primigravida at 31 weeks’ gestation presents to the labor floor reporting regular contractions. She denies bleeding or rupture of membranes. Contractions are noted at 3-minute intervals, and digital cervical examination reveals that she is dilated 1 cm, with 70% effacement. TVUS determines cervical length to be 17 mm.
How should she be managed?
From 24 to 34 weeks’ gestation, the prevention, diagnosis, and treatment of preterm labor become the main concerns. Because our ability to predict and prevent preterm birth is limited, clinical management focuses on a reliable diagnosis of preterm labor to allow for selective, timely interventions to optimize neonatal outcomes. These interventions include tocolysis to permit maternal transport; antibiotic prophylaxis for group B strep; and steroid administration to accelerate fetal lung maturity. Equally important is the ability to reliably rule out preterm labor among symptomatic (contracting) women to avoid the potential morbidity, cost, and inconvenience of these interventions.
Before 37 weeks, a diagnosis of preterm labor requires the following findings:
- six or more contractions per hour
- cervical dilation, as identified by digital examination, of at least 3 cm and 80% effacement.
This diagnosis is more reliable when ruptured membranes or vaginal bleeding are present.
The significance of contractions without these findings is less clear. Therefore, we follow an algorithm that incorporates TVUS measurement of cervical length and evaluation of fetal fibronectin (fFN) (FIGURE 4).13
fFN is a glycoprotein that is normally confined to the extracellular matrix of the fetal membranes between 24 and 34 weeks’ gestation. Detection of fFN in cervicovaginal secretions during this window is associated with an increased risk of spontaneous preterm birth, whereas its absence demonstrates a negative predictive value for delivery within 7 days of testing of more than 97%.
During initial assessment of a regularly contracting preterm patient, perform a vaginal speculum examination. If the membranes are intact, use a vaginal swab to assess the patient for the presence of fFN, and set the specimen aside. Also obtain a culture for group B strep.
If a digital cervical examination and the contraction pattern establish a diagnosis of preterm labor, administer a tocolytic, When fetal prophylactic antibiotics, and steroids. If the diagnosis remains unclear, evaluate the cervix via TVUS. A cervical length above 30 mm effectively rules out preterm labor and obviates the need to send the fFN swab for assessment. As a result, the patient can be managed expectantly.
In contrast, a cervical length below 20 mm effectively confirms the diagnosis of preterm labor, and treatment can proceed. Again, the fFN swab may be discarded. The swab is sent for processing only if cervical length is 20 mm to 30 mm (FIGURE 4). A positive fFN result leads to the presumptive diagnosis of preterm labor, whereas a negative result permits expectant management.
We also recommend that women who display symptoms of preterm labor be screened for asymptomatic bacteriuria. Identification and treatment of this condition significantly reduce the risk of preterm delivery (RR, 0.56; 95% CI, 0.43–0.73).14 In addition, diagnosis and treatment of bacterial vaginosis in symptomatic women who have a history of spontaneous preterm birth can also reduce the risk of recurrent preterm delivery (RR, 0.42; 95% CI, 0.27–0.67).15
Not all obstetric care providers have the resources necessary for TVUS assessment of cervical length. When that is the case, fFN offers high sensitivity and negative predictive value and can help guide initial clinical decision-making. Keep in mind, however, that not all facilities offer fFN testing. In addition, in some cases, cervical manipulation may have occurred before fFN testing was performed, precluding its validity. In such cases, the incorporation of TVUS assessment of cervical length into clinical evaluation may help guide decision-making.

FIGURE 4 How to identify preterm labor at 24 to 34 weeks
CASE RESOLVED
The determination of short cervical length (17 mm) by TVUS confirms the diagnosis of preterm labor. The patient is admitted to the hospital and treated with a tocolytic, prophylactic antibiotics, and steroids. Three days later, preterm labor recurs, and she delivers an otherwise healthy infant. Future pregnancies will be managed according to the algorithm presented in FIGURE 5.
Cerclage may benefit women who have a history of spontaneous preterm birth
A recent multicenter randomized trial evaluated the efficacy of cerclage in preventing preterm birth among 302 women who had a history of spontaneous preterm birth before 34 weeks’ gestation.11 Any woman who had a cervix shorter than 25 mm between 16-0/7 and 22-6/7 weeks’ gestation was randomized to cerclage or no cerclage.
Cerclage did not significantly reduce preterm delivery before 35 weeks’ gestation, the primary outcome. Thirty-two percent of women who received cerclage and 42% of women who did not receive cerclage delivered before 35 weeks (P = .09). However, among women who had a cervix shorter than 15 mm at randomization, cerclage reduced the rate of delivery before 35 weeks by more than 75% (P = .006). Cerclage also reduced the rate of spontaneous birth before 37 weeks, compared with no cerclage (45% vs 60%; P = .01), as well as the rate of previable preterm birth before 24 weeks (6.1% vs 14%; P = .03) and the rate of perinatal death (8.8% vs 16%; P = .046).
As this study demonstrates, ultrasonographically indicated cerclage produces a number of highly clinically significant benefits in women who have a history of spontaneous preterm birth. Another important question is whether supplemental progesterone offers additional benefit beyond that conferred by cerclage in this population.
The role of progesterone in preventing preterm birth
CASE 4: History of preterm and term delivery
A woman in her third pregnancy is referred for placement of cervical cerclage, based on her obstetric history. Her first pregnancy was marked by preterm labor at 26 weeks, resulting in spontaneous preterm birth at 28 weeks. In her second pregnancy, she had cerclage placed electively at 13 weeks and delivered spontaneously at 40 weeks with no complications of pregnancy.
Is another cerclage indicated—or would progesterone be more effective?
Supplemental 17-hydroxyprogesterone caproate, given weekly in an intramuscular dosage of 250 mg, significantly reduced the rate of recurrent spontaneous preterm birth when it was administered from 16 to 36 weeks’ gestation in women carrying a singleton fetus.16 The protective effect of progesterone is most apparent in women who have a history of very preterm delivery (<32 weeks). No such benefit has been observed in twin and triplet gestations, however.17-19
Among women who do benefit from progesterone, the effect may vary. For this reason, researchers have explored the measurement of midtrimester cervical length as a means of stratifying response to progesterone.
For example, investigators randomized women who had a history of spontaneous preterm birth to daily treatment with 90 mg of vaginal progesterone gel or placebo, starting between 16 and 22-6/7 weeks and continuing until 37 weeks or delivery, whichever came first. No difference in the rate of delivery at or before 32 weeks was observed. However, a secondary analysis among progesterone-treated women who had a cervical length below 28 mm found significant declines in the rate of delivery at or before 32 weeks, admission to a NICU, and length of stay.20,21
In another trial, investigators assessed cervical length via TVUS between 20 and 25 weeks’ gestation in a general obstetric population that included women carrying twins. Women who had a cervical length below 15 mm were offered randomization to daily oral progesterone (200 mg) or placebo from 24 weeks to 33-6/7 weeks’ gestation. The rate of spontaneous preterm birth was significantly lower in the progesterone group (19% vs 34%).22
Because the ideal formulation of progesterone for the prevention of preterm birth is unknown, ACOG recommends restricting its use to women who have a documented history of spontaneous preterm birth at less than 37 weeks’ gestation.23 My practice follows the protocol of Meis and colleagues within the framework of an overall systematic algorithm (FIGURE 5).

FIGURE 5 Cervical length in the second trimester
*For a singleton gestation only.
CASE RESOLVED
The ObGyn reviews the patient’s obstetric history and determines that the first pregnancy was more suggestive of preterm labor than cervical insufficiency. Therefore, the ObGyn opts for progesterone rather than cerclage to prolong the gestation. The patient begins weekly injections of 17-hydroxyprogesterone caproate, starting at 16 weeks’ gestation, with TVUS measurement of cervical length every 2 weeks. The cervix remains longer than 25 mm through 24 weeks’ gestation, at which time TVUS assessment is stopped. Progesterone injections continue through 36 weeks, and the patient spontaneously delivers a healthy 3,700-g infant at term.
1. Hamilton BE, Martin JA, Ventura SJ. Births: Preliminary data for 2007. National vital statistics reports; 2009;57(12). National Center for Health Statistics Web site. http://www.cdc.gov/nchs/data/nvsr/nvsr57/nvsr57_12.pdf. March 18, 2009. Accessed March 30, 2010.
2. Iams JD, Johnson FF, Sonek J, et al. Cervical competence as a continuum: a study of ultrasonographic cervical length and obstetric performance. Am J Obstet Gynecol. 1995;172(4 pt 1):1097-1106.
3. Iams JD, Goldenberg RL, Meis PJ, et al. The length of the cervix and the risk of spontaneous premature delivery. National Institute of Child Health and Human Development Maternal Fetal Medicine Unit Network. N Engl J Med. 1996;334(9):567-572.
4. Clinical Management Guidelines for Obstetrician-Gynecologists. No. 43, May 2003: Management of Preterm Labor. Obstet Gynecol. 2003;101(5 pt 1):1039-1047.
5. Owen J, Iams JD, Hauth JC. Vaginal sonography and cervical incompetence. Am J Obstet Gynecol. 2003;188(2):586-596.
6. Harger JH. Cerclage and cervical insufficiency: an evidence-based analysis. Obstet Gynecol. 2002;100(6):1313-1327.
7. Clinical Management Guidelines for Obstetrician-Gynecologists. No. 48, Nov 2003: Cervical insufficiency. Obstet Gynecol. 2003;102(5 pt 1):1091-1099.
8. Kusanovic JP, Espinoza J, Romero R, et al. Clinical significance of the presence of amniotic fluid ‘sludge’ in asymptomatic patients at high risk for spontaneous preterm delivery. Ultrasound Obstet Gynecol. 2007;30(5):706-714.
9. ACOG Practice Bulletin No. 80. Premature rupture of membranes. Obstet Gynecol. 2007;109(4):1007-1020.DOI:10.1097/01.AOG.0000263888.69178.1f.
10. Berghella V, Odibo AO, To MS, Rust OA, Althuisius SM. Cerclage for short cervix on ultrasonography: meta-analysis of trials using individual patient-level data. Obstet Gynecol. 2005;106(1):181-189.
11. Owen J, Hankins G, Iams JD, et al. Multicenter randomized trial of cerclage for preterm birth prevention in high-risk women with shortened mid-trimester cervical length. Am J Obstet Gynecol. 2009;201(4):375.e1-e8.
12. Simcox R, Seed PT, Bennett P, Teoh TG, Poston L, Shennan AH. A randomized control trial of cervical scanning vs history to determine cerclage in women at high risk of preterm birth (CIRCLE trial). Am J Obstet Gynecol. 2009;200(6):623.e1-e6.
13. Iams JD. Prediction and early detection of preterm labor. Obstet Gynecol. 2003;101(2):402-412.
14. Romero R, Oyarzun E, Mazor M, Sirtori M, Hobbins JC, Bracken M. Meta-analysis of the relationship between asymptomatic bacteriuria and preterm delivery/low birth weight. Obstet Gynecol. 1989;73(4):576-582.
15. Leitich H, Brunbauer M, Bodnar-Aderl B, Kaider A, Egarter C, Husslein P. Antibiotic treatment of bacterial vaginosis in pregnancy: a meta-analysis. Am J Obstet Gynecol. 2003;188(3):752-758.
16. Meis PJ, Klebanoff M, Thom E, et al. For National Institute of Child Health and Human Development Maternal-Fetal Medicine Units Network. Prevention of recurrent preterm birth by 17 alpha-hydroxyprogesterone caproate. N Engl J Med. 2003;348(24):2379-2385.
17. Norman JE, Mackenzie F, Owen P, et al. Progesterone for the prevention of preterm birth in twin pregnancy (STOPPIT): a randomised, double-blind, placebo-controlled study and meta-analysis. Lancet. 2009;373(9680):2034-2040.
18. Rouse DJ, Caritis SN, Peaceman AM, et al. For National Institute of Child Health and Human Development Maternal-Fetal Medicine Units Network. A trial of 17 alpha-hydroxyprogesterone caproate to prevent prematurity in twins. N Engl J Med. 2007;357(5):454-461.
19. Caritis SN, Rouse DJ, Peaceman AM, et al. for Eunice Kennedy Shriver National Institute of Child Health and Human Development (NICHD), Medicine Units Network (MFMU). Prevention of preterm birth in triplets using 17 alpha-hydroxyprogesterone caproate: a randomized controlled trial. Obstet Gynecol. 2009;113(2 pt 1):285-292.
20. O’Brien JM, Adair CD, Lewis DF, et al. Progesterone vaginal gel for the reduction of recurrent preterm birth: primary results from a randomized, double-blind, placebo-controlled trial. Ultrasound Obstet Gynecol. 2007;30(5):687-696.
21. DeFranco EA, O’Brien JM, Adair CD, et al. Vaginal progesterone is associated with a decrease in risk for early preterm birth and improved neonatal outcome in women with a short cervix: a secondary analysis from a randomized, double-blind, placebo-controlled trial. Ultrasound Obstet Gynecol. 2007;30(5):697-705.
22. Fonseca EB, Celik E, Parra M, et al. For Fetal Medicine Foundation Second Trimester Screening Group. Progesterone and the risk of preterm birth among women with a short cervix. N Engl J Med. 2007;357(5):462-469.
23. ACOG Committee Opinion No. 419: Use of progesterone to reduce preterm birth. Obstet Gynecol. 2008;112(4):963.-
24. Yost NP, Bloom SL, Twickler DM, Leveno KJ. Pitfalls in ultrasonic cervical length measurement for predicting preterm birth. Obstet Gynecol. 1999;93(4):510-516.
CASE 1: Short cervix in the middle trimester
During routine second-trimester ultrasonography, a 34-year-old primigravida at 22 weeks’ gestation is found to have a cervix 15 mm in length. She has no other risk factors for spontaneous preterm birth.
What steps do you take to ensure that her pregnancy progresses uneventfully to term?
Cervical length is not routinely measured in low-risk women—i.e., those without a history of spontaneous preterm birth—but a short cervix is sometimes detected during ultrasonographic imaging for other indications, as it was in this case. When a short cervix is detected incidentally, I educate the patient to watch for early warning signs of preterm labor. I also recommend pelvic rest and a sedentary lifestyle.
The rate of preterm birth declined slightly in 2007—the first decrease in more than 20 years—but the phenomenon remains the leading cause of perinatal morbidity and mortality in developed nations.1 In the United States, more than 500,000 babies each year, or 12% to 13% of all births, are delivered before 37 weeks’ gestation. Most of these births are spontaneous and involve preterm labor or premature rupture of membranes. Medical costs for a preterm newborn exceed those of a term infant by a multiple of more than 10, and the average hospitalization exceeds that of a term infant by a multiple of more than six.
In this article, I discuss the rationale and technique for ultrasonographic cervical measurement to determine the likelihood of preterm birth. I also examine the data on the short cervix in various settings, and describe strategies for cervical assessment and preterm birth prevention, including cerclage and progesterone, framing the discussion in terms of gestational age.
Accurate and reproducible measurement of cervical length depends on correct technique. Use of transvaginal ultrasonography (TVUS) limits variations between measurements to 5% to 10%, a marked improvement over digital examination and transabdominal US.
Here are the five steps involved, in the order performed:
- Ensure that the patient’s bladder is empty. This precaution is necessary to prevent dynamic, or spontaneous, lengthening or shortening of the cervix.
- Counsel and position the patient. Explain the procedure to the patient and have her assume the dorsal lithotomy position.
- Introduce the probe into the anterior vaginal fornix using real-time visualization, and obtain a mid-sagittal view of the cervix. Withdraw the probe just enough to allow the image to blur, then advance the probe just enough for the image to regain clarity. This sequence prevents the practitioner from exerting excessive pressure on the cervix, which can falsely elongate it.
- Place an electronic caliper (on-screen) at the notch that represents the internal cervical os, and another at the external os (FIGURE 1).
- Measure the distance between the notches and report the shortest of three separately obtained measurements.

FIGURE 1 How to measure the cervix
Electronic calipers mark the internal and external os in this cervix measuring 42 mm via midsagittal transvaginal ultrasonography.
Watch for these pitfalls!
Before 20 weeks’ gestation, the lower uterine segment is not particularly well developed, making it difficult to reliably determine the location of the internal os (FIGURE 2). Moreover, focal myometrial contractions of the lower uterine segment, which are common, may give the false impression of increased cervical length or dilation of the internal os (FIGURE 2).24

FIGURE 2 Measurement may be difficult in early pregnancy
A. This transvaginal sonogram demonstrates the difficulty of determining the location of the internal os when the lower uterine segment is undeveloped (arrows). B. When lower uterine contractions occur with the anterior and posterior walls in opposition (arrowheads), transvaginal imaging may give the false appearance of dilation with funneling (arrow).
Why we assess cervical length
In the past, the cervix was viewed as either competent—i.e., capable of maintaining a pregnancy until term—or as “incompetent.” More recent evidence has broadened our understanding of cervical function, which is now viewed along a continuum.
In landmark research in the mid-1990s, investigators compared cervical lengths, measured via transvaginal ultrasonography (TVUS), between two groups of pregnant women—those who had a history of preterm birth and those who did not.
The result? Gestational age at delivery in the first pregnancy correlated significantly—and continuously—with cervical length between 20 and 30 weeks’ gestation in the subsequent pregnancy.2 Investigators also observed that the risk of spontaneous preterm birth increased with decreasing cervical length. A length of 25 mm (10th percentile) offered a clinically appropriate threshold for identification of the risk of preterm delivery.3
The value of cervical-length measurement lies in its high negative predictive value for recurrent spontaneous preterm birth.4 As a general rule of thumb, routine assessment of cervical length in asymptomatic women who do not have a history of preterm birth is not recommended because of the high rate of false-positive results and the low positive predictive value for preterm delivery. We also lack an evidence-based consensus on how to manage an abnormally short cervix in these women.
CASE RESOLVED
The patient is counseled to watch for signs of preterm labor and to remain sedentary. Her pregnancy progresses without incident until 40 weeks, when she undergoes induction of labor for oligohydramnios and delivers a healthy infant weighing 3,855 g.
When a short cervix is detected at less than 20 weeks
If a woman has a history of spontaneous preterm birth, a short cervix this early in gestation raises the question of cervical insufficiency. No objective criteria have been devised to identify this condition. Nor is there a widely accepted definition. A reasonable description does exist, however:
- …a clinical diagnosis characterized by recurrent painless dilation and spontaneous midtrimester birth, generally in the absence of predisposing conditions such as spontaneous membrane rupture, bleeding, and infection, characteristics that shift the presumed underlying cause away from cervical incompetence and support other components of the preterm birth syndrome.5
A patient who fits this description may be a candidate for cervical cerclage. Alternatively, it is reasonable to reassess the patient in 3 to 7 days, after restricting physical activity, keeping in mind the pitfalls of TVUS assessment of the lower uterine segment in early gestation (see the box on imaging). If cervical length remains short, consider cerclage.6,7
Watch for inflammation
Occasionally, echogenic material is observed in the amniotic fluid at the level of a short cervix. This debris is an inflammatory exudate of fibrin, white blood cells, and bacteria. The presence of this sludge (FIGURE 3) signifies a risk of preterm birth much greater than that associated with a short cervix alone.8 Because cerclage in the presence of inflammation may further heighten the risk of spontaneous preterm birth, I recommend caution.

FIGURE 3 Watch for signs of inflammation
When inflammatory exudate (asterisk) is identified at the level of a short cervix, the risk of spontaneous preterm birth is elevated beyond the risk associated with a short cervix alone.
When a short cervix is detected between 20 and 24 weeks
CASE 2: Is recurrent preterm birth likely?
A 30-year-old woman 21 weeks pregnant with her second child reports for TVUS. Because her first child was delivered preterm at 30 weeks, she has been undergoing periodic measurement of her cervix. Until today, it has been longer than 25 mm, but now it is 20 mm. What is the best strategy to avert another preterm birth?
If a short cervix (<25 mm) is noted in a high-risk patient at this gestational age, consider the possibility of preterm labor and ruptured membranes. If these conditions are present, they should be managed according to existing guidelines.4,9 If they are absent, consider cerclage.
A recent meta-analysis of randomized trials of cerclage for the prevention of preterm birth in a singleton, high-risk pregnancy with a short cervix suggests that cerclage is associated with a significantly lower risk of delivery before 35 weeks’ gestation (relative risk [RR], 0.61; 95% confidence interval [CI], 0.40–0.92). Among singleton pregnancies involving both a short cervix and a history of midtrimester loss, cerclage is again associated with a reduced likelihood of delivery before 35 weeks (39% vs 23.4%; number needed to treat [NNT], 8; RR, 0.57; 95% CI, 0.33–0.99).10 A recent randomized trial of cerclage versus no cerclage in women who had a history of spontaneous preterm birth produced similar findings.11 This trial is described in detail in the box.
Another randomized trial compared the relative merits of ultrasound-indicated cerclage for a cervix shorter than 20 mm to a history-indicated cerclage among women with a prior spontaneous preterm birth between 16 and 34 weeks. Thirty-nine of 123 women randomized to the former group received a cerclage, as did 25 of 125 subjects in the latter group. There were no significant differences in the primary outcome of delivery before 34 weeks or secondary measures of loss before 24 weeks, preterm premature membrane rupture, mean gestational age at delivery, or neonatal outcomes. This study was not designed or powered to evaluate the efficacy of cerclage in preventing recurrent, spontaneous preterm birth. It simply compares two ways of selecting high-risk women for cerclage.12
We lack data supporting placement of cerclage for an incidentally detected short cervix in women who lack a history of spontaneous preterm birth or midtrimester loss.7
Other groups known to be at increased risk of spontaneous preterm birth include women carrying twins and women who have undergone cervical cone biopsy or loop electrosurgical excision procedure (LEEP). Among twin gestations, cerclage for a short cervix is associated with an increased rate of preterm birth (<35 weeks).10 Cerclage for a short cervix has not been evaluated among women who have a history of LEEP.6
CASE RESOLVED
After preterm labor is ruled out, the patient is counseled about her options and chooses cervical cerclage. Her pregnancy proceeds uneventfully until 36 weeks’ gestation, when she delivers a healthy infant weighing 2,950 g.
When a short cervix is detected between 24 and 34 weeks
CASE 3: Is it preterm labor?
A primigravida at 31 weeks’ gestation presents to the labor floor reporting regular contractions. She denies bleeding or rupture of membranes. Contractions are noted at 3-minute intervals, and digital cervical examination reveals that she is dilated 1 cm, with 70% effacement. TVUS determines cervical length to be 17 mm.
How should she be managed?
From 24 to 34 weeks’ gestation, the prevention, diagnosis, and treatment of preterm labor become the main concerns. Because our ability to predict and prevent preterm birth is limited, clinical management focuses on a reliable diagnosis of preterm labor to allow for selective, timely interventions to optimize neonatal outcomes. These interventions include tocolysis to permit maternal transport; antibiotic prophylaxis for group B strep; and steroid administration to accelerate fetal lung maturity. Equally important is the ability to reliably rule out preterm labor among symptomatic (contracting) women to avoid the potential morbidity, cost, and inconvenience of these interventions.
Before 37 weeks, a diagnosis of preterm labor requires the following findings:
- six or more contractions per hour
- cervical dilation, as identified by digital examination, of at least 3 cm and 80% effacement.
This diagnosis is more reliable when ruptured membranes or vaginal bleeding are present.
The significance of contractions without these findings is less clear. Therefore, we follow an algorithm that incorporates TVUS measurement of cervical length and evaluation of fetal fibronectin (fFN) (FIGURE 4).13
fFN is a glycoprotein that is normally confined to the extracellular matrix of the fetal membranes between 24 and 34 weeks’ gestation. Detection of fFN in cervicovaginal secretions during this window is associated with an increased risk of spontaneous preterm birth, whereas its absence demonstrates a negative predictive value for delivery within 7 days of testing of more than 97%.
During initial assessment of a regularly contracting preterm patient, perform a vaginal speculum examination. If the membranes are intact, use a vaginal swab to assess the patient for the presence of fFN, and set the specimen aside. Also obtain a culture for group B strep.
If a digital cervical examination and the contraction pattern establish a diagnosis of preterm labor, administer a tocolytic, When fetal prophylactic antibiotics, and steroids. If the diagnosis remains unclear, evaluate the cervix via TVUS. A cervical length above 30 mm effectively rules out preterm labor and obviates the need to send the fFN swab for assessment. As a result, the patient can be managed expectantly.
In contrast, a cervical length below 20 mm effectively confirms the diagnosis of preterm labor, and treatment can proceed. Again, the fFN swab may be discarded. The swab is sent for processing only if cervical length is 20 mm to 30 mm (FIGURE 4). A positive fFN result leads to the presumptive diagnosis of preterm labor, whereas a negative result permits expectant management.
We also recommend that women who display symptoms of preterm labor be screened for asymptomatic bacteriuria. Identification and treatment of this condition significantly reduce the risk of preterm delivery (RR, 0.56; 95% CI, 0.43–0.73).14 In addition, diagnosis and treatment of bacterial vaginosis in symptomatic women who have a history of spontaneous preterm birth can also reduce the risk of recurrent preterm delivery (RR, 0.42; 95% CI, 0.27–0.67).15
Not all obstetric care providers have the resources necessary for TVUS assessment of cervical length. When that is the case, fFN offers high sensitivity and negative predictive value and can help guide initial clinical decision-making. Keep in mind, however, that not all facilities offer fFN testing. In addition, in some cases, cervical manipulation may have occurred before fFN testing was performed, precluding its validity. In such cases, the incorporation of TVUS assessment of cervical length into clinical evaluation may help guide decision-making.

FIGURE 4 How to identify preterm labor at 24 to 34 weeks
CASE RESOLVED
The determination of short cervical length (17 mm) by TVUS confirms the diagnosis of preterm labor. The patient is admitted to the hospital and treated with a tocolytic, prophylactic antibiotics, and steroids. Three days later, preterm labor recurs, and she delivers an otherwise healthy infant. Future pregnancies will be managed according to the algorithm presented in FIGURE 5.
Cerclage may benefit women who have a history of spontaneous preterm birth
A recent multicenter randomized trial evaluated the efficacy of cerclage in preventing preterm birth among 302 women who had a history of spontaneous preterm birth before 34 weeks’ gestation.11 Any woman who had a cervix shorter than 25 mm between 16-0/7 and 22-6/7 weeks’ gestation was randomized to cerclage or no cerclage.
Cerclage did not significantly reduce preterm delivery before 35 weeks’ gestation, the primary outcome. Thirty-two percent of women who received cerclage and 42% of women who did not receive cerclage delivered before 35 weeks (P = .09). However, among women who had a cervix shorter than 15 mm at randomization, cerclage reduced the rate of delivery before 35 weeks by more than 75% (P = .006). Cerclage also reduced the rate of spontaneous birth before 37 weeks, compared with no cerclage (45% vs 60%; P = .01), as well as the rate of previable preterm birth before 24 weeks (6.1% vs 14%; P = .03) and the rate of perinatal death (8.8% vs 16%; P = .046).
As this study demonstrates, ultrasonographically indicated cerclage produces a number of highly clinically significant benefits in women who have a history of spontaneous preterm birth. Another important question is whether supplemental progesterone offers additional benefit beyond that conferred by cerclage in this population.
The role of progesterone in preventing preterm birth
CASE 4: History of preterm and term delivery
A woman in her third pregnancy is referred for placement of cervical cerclage, based on her obstetric history. Her first pregnancy was marked by preterm labor at 26 weeks, resulting in spontaneous preterm birth at 28 weeks. In her second pregnancy, she had cerclage placed electively at 13 weeks and delivered spontaneously at 40 weeks with no complications of pregnancy.
Is another cerclage indicated—or would progesterone be more effective?
Supplemental 17-hydroxyprogesterone caproate, given weekly in an intramuscular dosage of 250 mg, significantly reduced the rate of recurrent spontaneous preterm birth when it was administered from 16 to 36 weeks’ gestation in women carrying a singleton fetus.16 The protective effect of progesterone is most apparent in women who have a history of very preterm delivery (<32 weeks). No such benefit has been observed in twin and triplet gestations, however.17-19
Among women who do benefit from progesterone, the effect may vary. For this reason, researchers have explored the measurement of midtrimester cervical length as a means of stratifying response to progesterone.
For example, investigators randomized women who had a history of spontaneous preterm birth to daily treatment with 90 mg of vaginal progesterone gel or placebo, starting between 16 and 22-6/7 weeks and continuing until 37 weeks or delivery, whichever came first. No difference in the rate of delivery at or before 32 weeks was observed. However, a secondary analysis among progesterone-treated women who had a cervical length below 28 mm found significant declines in the rate of delivery at or before 32 weeks, admission to a NICU, and length of stay.20,21
In another trial, investigators assessed cervical length via TVUS between 20 and 25 weeks’ gestation in a general obstetric population that included women carrying twins. Women who had a cervical length below 15 mm were offered randomization to daily oral progesterone (200 mg) or placebo from 24 weeks to 33-6/7 weeks’ gestation. The rate of spontaneous preterm birth was significantly lower in the progesterone group (19% vs 34%).22
Because the ideal formulation of progesterone for the prevention of preterm birth is unknown, ACOG recommends restricting its use to women who have a documented history of spontaneous preterm birth at less than 37 weeks’ gestation.23 My practice follows the protocol of Meis and colleagues within the framework of an overall systematic algorithm (FIGURE 5).

FIGURE 5 Cervical length in the second trimester
*For a singleton gestation only.
CASE RESOLVED
The ObGyn reviews the patient’s obstetric history and determines that the first pregnancy was more suggestive of preterm labor than cervical insufficiency. Therefore, the ObGyn opts for progesterone rather than cerclage to prolong the gestation. The patient begins weekly injections of 17-hydroxyprogesterone caproate, starting at 16 weeks’ gestation, with TVUS measurement of cervical length every 2 weeks. The cervix remains longer than 25 mm through 24 weeks’ gestation, at which time TVUS assessment is stopped. Progesterone injections continue through 36 weeks, and the patient spontaneously delivers a healthy 3,700-g infant at term.
CASE 1: Short cervix in the middle trimester
During routine second-trimester ultrasonography, a 34-year-old primigravida at 22 weeks’ gestation is found to have a cervix 15 mm in length. She has no other risk factors for spontaneous preterm birth.
What steps do you take to ensure that her pregnancy progresses uneventfully to term?
Cervical length is not routinely measured in low-risk women—i.e., those without a history of spontaneous preterm birth—but a short cervix is sometimes detected during ultrasonographic imaging for other indications, as it was in this case. When a short cervix is detected incidentally, I educate the patient to watch for early warning signs of preterm labor. I also recommend pelvic rest and a sedentary lifestyle.
The rate of preterm birth declined slightly in 2007—the first decrease in more than 20 years—but the phenomenon remains the leading cause of perinatal morbidity and mortality in developed nations.1 In the United States, more than 500,000 babies each year, or 12% to 13% of all births, are delivered before 37 weeks’ gestation. Most of these births are spontaneous and involve preterm labor or premature rupture of membranes. Medical costs for a preterm newborn exceed those of a term infant by a multiple of more than 10, and the average hospitalization exceeds that of a term infant by a multiple of more than six.
In this article, I discuss the rationale and technique for ultrasonographic cervical measurement to determine the likelihood of preterm birth. I also examine the data on the short cervix in various settings, and describe strategies for cervical assessment and preterm birth prevention, including cerclage and progesterone, framing the discussion in terms of gestational age.
Accurate and reproducible measurement of cervical length depends on correct technique. Use of transvaginal ultrasonography (TVUS) limits variations between measurements to 5% to 10%, a marked improvement over digital examination and transabdominal US.
Here are the five steps involved, in the order performed:
- Ensure that the patient’s bladder is empty. This precaution is necessary to prevent dynamic, or spontaneous, lengthening or shortening of the cervix.
- Counsel and position the patient. Explain the procedure to the patient and have her assume the dorsal lithotomy position.
- Introduce the probe into the anterior vaginal fornix using real-time visualization, and obtain a mid-sagittal view of the cervix. Withdraw the probe just enough to allow the image to blur, then advance the probe just enough for the image to regain clarity. This sequence prevents the practitioner from exerting excessive pressure on the cervix, which can falsely elongate it.
- Place an electronic caliper (on-screen) at the notch that represents the internal cervical os, and another at the external os (FIGURE 1).
- Measure the distance between the notches and report the shortest of three separately obtained measurements.

FIGURE 1 How to measure the cervix
Electronic calipers mark the internal and external os in this cervix measuring 42 mm via midsagittal transvaginal ultrasonography.
Watch for these pitfalls!
Before 20 weeks’ gestation, the lower uterine segment is not particularly well developed, making it difficult to reliably determine the location of the internal os (FIGURE 2). Moreover, focal myometrial contractions of the lower uterine segment, which are common, may give the false impression of increased cervical length or dilation of the internal os (FIGURE 2).24

FIGURE 2 Measurement may be difficult in early pregnancy
A. This transvaginal sonogram demonstrates the difficulty of determining the location of the internal os when the lower uterine segment is undeveloped (arrows). B. When lower uterine contractions occur with the anterior and posterior walls in opposition (arrowheads), transvaginal imaging may give the false appearance of dilation with funneling (arrow).
Why we assess cervical length
In the past, the cervix was viewed as either competent—i.e., capable of maintaining a pregnancy until term—or as “incompetent.” More recent evidence has broadened our understanding of cervical function, which is now viewed along a continuum.
In landmark research in the mid-1990s, investigators compared cervical lengths, measured via transvaginal ultrasonography (TVUS), between two groups of pregnant women—those who had a history of preterm birth and those who did not.
The result? Gestational age at delivery in the first pregnancy correlated significantly—and continuously—with cervical length between 20 and 30 weeks’ gestation in the subsequent pregnancy.2 Investigators also observed that the risk of spontaneous preterm birth increased with decreasing cervical length. A length of 25 mm (10th percentile) offered a clinically appropriate threshold for identification of the risk of preterm delivery.3
The value of cervical-length measurement lies in its high negative predictive value for recurrent spontaneous preterm birth.4 As a general rule of thumb, routine assessment of cervical length in asymptomatic women who do not have a history of preterm birth is not recommended because of the high rate of false-positive results and the low positive predictive value for preterm delivery. We also lack an evidence-based consensus on how to manage an abnormally short cervix in these women.
CASE RESOLVED
The patient is counseled to watch for signs of preterm labor and to remain sedentary. Her pregnancy progresses without incident until 40 weeks, when she undergoes induction of labor for oligohydramnios and delivers a healthy infant weighing 3,855 g.
When a short cervix is detected at less than 20 weeks
If a woman has a history of spontaneous preterm birth, a short cervix this early in gestation raises the question of cervical insufficiency. No objective criteria have been devised to identify this condition. Nor is there a widely accepted definition. A reasonable description does exist, however:
- …a clinical diagnosis characterized by recurrent painless dilation and spontaneous midtrimester birth, generally in the absence of predisposing conditions such as spontaneous membrane rupture, bleeding, and infection, characteristics that shift the presumed underlying cause away from cervical incompetence and support other components of the preterm birth syndrome.5
A patient who fits this description may be a candidate for cervical cerclage. Alternatively, it is reasonable to reassess the patient in 3 to 7 days, after restricting physical activity, keeping in mind the pitfalls of TVUS assessment of the lower uterine segment in early gestation (see the box on imaging). If cervical length remains short, consider cerclage.6,7
Watch for inflammation
Occasionally, echogenic material is observed in the amniotic fluid at the level of a short cervix. This debris is an inflammatory exudate of fibrin, white blood cells, and bacteria. The presence of this sludge (FIGURE 3) signifies a risk of preterm birth much greater than that associated with a short cervix alone.8 Because cerclage in the presence of inflammation may further heighten the risk of spontaneous preterm birth, I recommend caution.

FIGURE 3 Watch for signs of inflammation
When inflammatory exudate (asterisk) is identified at the level of a short cervix, the risk of spontaneous preterm birth is elevated beyond the risk associated with a short cervix alone.
When a short cervix is detected between 20 and 24 weeks
CASE 2: Is recurrent preterm birth likely?
A 30-year-old woman 21 weeks pregnant with her second child reports for TVUS. Because her first child was delivered preterm at 30 weeks, she has been undergoing periodic measurement of her cervix. Until today, it has been longer than 25 mm, but now it is 20 mm. What is the best strategy to avert another preterm birth?
If a short cervix (<25 mm) is noted in a high-risk patient at this gestational age, consider the possibility of preterm labor and ruptured membranes. If these conditions are present, they should be managed according to existing guidelines.4,9 If they are absent, consider cerclage.
A recent meta-analysis of randomized trials of cerclage for the prevention of preterm birth in a singleton, high-risk pregnancy with a short cervix suggests that cerclage is associated with a significantly lower risk of delivery before 35 weeks’ gestation (relative risk [RR], 0.61; 95% confidence interval [CI], 0.40–0.92). Among singleton pregnancies involving both a short cervix and a history of midtrimester loss, cerclage is again associated with a reduced likelihood of delivery before 35 weeks (39% vs 23.4%; number needed to treat [NNT], 8; RR, 0.57; 95% CI, 0.33–0.99).10 A recent randomized trial of cerclage versus no cerclage in women who had a history of spontaneous preterm birth produced similar findings.11 This trial is described in detail in the box.
Another randomized trial compared the relative merits of ultrasound-indicated cerclage for a cervix shorter than 20 mm to a history-indicated cerclage among women with a prior spontaneous preterm birth between 16 and 34 weeks. Thirty-nine of 123 women randomized to the former group received a cerclage, as did 25 of 125 subjects in the latter group. There were no significant differences in the primary outcome of delivery before 34 weeks or secondary measures of loss before 24 weeks, preterm premature membrane rupture, mean gestational age at delivery, or neonatal outcomes. This study was not designed or powered to evaluate the efficacy of cerclage in preventing recurrent, spontaneous preterm birth. It simply compares two ways of selecting high-risk women for cerclage.12
We lack data supporting placement of cerclage for an incidentally detected short cervix in women who lack a history of spontaneous preterm birth or midtrimester loss.7
Other groups known to be at increased risk of spontaneous preterm birth include women carrying twins and women who have undergone cervical cone biopsy or loop electrosurgical excision procedure (LEEP). Among twin gestations, cerclage for a short cervix is associated with an increased rate of preterm birth (<35 weeks).10 Cerclage for a short cervix has not been evaluated among women who have a history of LEEP.6
CASE RESOLVED
After preterm labor is ruled out, the patient is counseled about her options and chooses cervical cerclage. Her pregnancy proceeds uneventfully until 36 weeks’ gestation, when she delivers a healthy infant weighing 2,950 g.
When a short cervix is detected between 24 and 34 weeks
CASE 3: Is it preterm labor?
A primigravida at 31 weeks’ gestation presents to the labor floor reporting regular contractions. She denies bleeding or rupture of membranes. Contractions are noted at 3-minute intervals, and digital cervical examination reveals that she is dilated 1 cm, with 70% effacement. TVUS determines cervical length to be 17 mm.
How should she be managed?
From 24 to 34 weeks’ gestation, the prevention, diagnosis, and treatment of preterm labor become the main concerns. Because our ability to predict and prevent preterm birth is limited, clinical management focuses on a reliable diagnosis of preterm labor to allow for selective, timely interventions to optimize neonatal outcomes. These interventions include tocolysis to permit maternal transport; antibiotic prophylaxis for group B strep; and steroid administration to accelerate fetal lung maturity. Equally important is the ability to reliably rule out preterm labor among symptomatic (contracting) women to avoid the potential morbidity, cost, and inconvenience of these interventions.
Before 37 weeks, a diagnosis of preterm labor requires the following findings:
- six or more contractions per hour
- cervical dilation, as identified by digital examination, of at least 3 cm and 80% effacement.
This diagnosis is more reliable when ruptured membranes or vaginal bleeding are present.
The significance of contractions without these findings is less clear. Therefore, we follow an algorithm that incorporates TVUS measurement of cervical length and evaluation of fetal fibronectin (fFN) (FIGURE 4).13
fFN is a glycoprotein that is normally confined to the extracellular matrix of the fetal membranes between 24 and 34 weeks’ gestation. Detection of fFN in cervicovaginal secretions during this window is associated with an increased risk of spontaneous preterm birth, whereas its absence demonstrates a negative predictive value for delivery within 7 days of testing of more than 97%.
During initial assessment of a regularly contracting preterm patient, perform a vaginal speculum examination. If the membranes are intact, use a vaginal swab to assess the patient for the presence of fFN, and set the specimen aside. Also obtain a culture for group B strep.
If a digital cervical examination and the contraction pattern establish a diagnosis of preterm labor, administer a tocolytic, When fetal prophylactic antibiotics, and steroids. If the diagnosis remains unclear, evaluate the cervix via TVUS. A cervical length above 30 mm effectively rules out preterm labor and obviates the need to send the fFN swab for assessment. As a result, the patient can be managed expectantly.
In contrast, a cervical length below 20 mm effectively confirms the diagnosis of preterm labor, and treatment can proceed. Again, the fFN swab may be discarded. The swab is sent for processing only if cervical length is 20 mm to 30 mm (FIGURE 4). A positive fFN result leads to the presumptive diagnosis of preterm labor, whereas a negative result permits expectant management.
We also recommend that women who display symptoms of preterm labor be screened for asymptomatic bacteriuria. Identification and treatment of this condition significantly reduce the risk of preterm delivery (RR, 0.56; 95% CI, 0.43–0.73).14 In addition, diagnosis and treatment of bacterial vaginosis in symptomatic women who have a history of spontaneous preterm birth can also reduce the risk of recurrent preterm delivery (RR, 0.42; 95% CI, 0.27–0.67).15
Not all obstetric care providers have the resources necessary for TVUS assessment of cervical length. When that is the case, fFN offers high sensitivity and negative predictive value and can help guide initial clinical decision-making. Keep in mind, however, that not all facilities offer fFN testing. In addition, in some cases, cervical manipulation may have occurred before fFN testing was performed, precluding its validity. In such cases, the incorporation of TVUS assessment of cervical length into clinical evaluation may help guide decision-making.

FIGURE 4 How to identify preterm labor at 24 to 34 weeks
CASE RESOLVED
The determination of short cervical length (17 mm) by TVUS confirms the diagnosis of preterm labor. The patient is admitted to the hospital and treated with a tocolytic, prophylactic antibiotics, and steroids. Three days later, preterm labor recurs, and she delivers an otherwise healthy infant. Future pregnancies will be managed according to the algorithm presented in FIGURE 5.
Cerclage may benefit women who have a history of spontaneous preterm birth
A recent multicenter randomized trial evaluated the efficacy of cerclage in preventing preterm birth among 302 women who had a history of spontaneous preterm birth before 34 weeks’ gestation.11 Any woman who had a cervix shorter than 25 mm between 16-0/7 and 22-6/7 weeks’ gestation was randomized to cerclage or no cerclage.
Cerclage did not significantly reduce preterm delivery before 35 weeks’ gestation, the primary outcome. Thirty-two percent of women who received cerclage and 42% of women who did not receive cerclage delivered before 35 weeks (P = .09). However, among women who had a cervix shorter than 15 mm at randomization, cerclage reduced the rate of delivery before 35 weeks by more than 75% (P = .006). Cerclage also reduced the rate of spontaneous birth before 37 weeks, compared with no cerclage (45% vs 60%; P = .01), as well as the rate of previable preterm birth before 24 weeks (6.1% vs 14%; P = .03) and the rate of perinatal death (8.8% vs 16%; P = .046).
As this study demonstrates, ultrasonographically indicated cerclage produces a number of highly clinically significant benefits in women who have a history of spontaneous preterm birth. Another important question is whether supplemental progesterone offers additional benefit beyond that conferred by cerclage in this population.
The role of progesterone in preventing preterm birth
CASE 4: History of preterm and term delivery
A woman in her third pregnancy is referred for placement of cervical cerclage, based on her obstetric history. Her first pregnancy was marked by preterm labor at 26 weeks, resulting in spontaneous preterm birth at 28 weeks. In her second pregnancy, she had cerclage placed electively at 13 weeks and delivered spontaneously at 40 weeks with no complications of pregnancy.
Is another cerclage indicated—or would progesterone be more effective?
Supplemental 17-hydroxyprogesterone caproate, given weekly in an intramuscular dosage of 250 mg, significantly reduced the rate of recurrent spontaneous preterm birth when it was administered from 16 to 36 weeks’ gestation in women carrying a singleton fetus.16 The protective effect of progesterone is most apparent in women who have a history of very preterm delivery (<32 weeks). No such benefit has been observed in twin and triplet gestations, however.17-19
Among women who do benefit from progesterone, the effect may vary. For this reason, researchers have explored the measurement of midtrimester cervical length as a means of stratifying response to progesterone.
For example, investigators randomized women who had a history of spontaneous preterm birth to daily treatment with 90 mg of vaginal progesterone gel or placebo, starting between 16 and 22-6/7 weeks and continuing until 37 weeks or delivery, whichever came first. No difference in the rate of delivery at or before 32 weeks was observed. However, a secondary analysis among progesterone-treated women who had a cervical length below 28 mm found significant declines in the rate of delivery at or before 32 weeks, admission to a NICU, and length of stay.20,21
In another trial, investigators assessed cervical length via TVUS between 20 and 25 weeks’ gestation in a general obstetric population that included women carrying twins. Women who had a cervical length below 15 mm were offered randomization to daily oral progesterone (200 mg) or placebo from 24 weeks to 33-6/7 weeks’ gestation. The rate of spontaneous preterm birth was significantly lower in the progesterone group (19% vs 34%).22
Because the ideal formulation of progesterone for the prevention of preterm birth is unknown, ACOG recommends restricting its use to women who have a documented history of spontaneous preterm birth at less than 37 weeks’ gestation.23 My practice follows the protocol of Meis and colleagues within the framework of an overall systematic algorithm (FIGURE 5).

FIGURE 5 Cervical length in the second trimester
*For a singleton gestation only.
CASE RESOLVED
The ObGyn reviews the patient’s obstetric history and determines that the first pregnancy was more suggestive of preterm labor than cervical insufficiency. Therefore, the ObGyn opts for progesterone rather than cerclage to prolong the gestation. The patient begins weekly injections of 17-hydroxyprogesterone caproate, starting at 16 weeks’ gestation, with TVUS measurement of cervical length every 2 weeks. The cervix remains longer than 25 mm through 24 weeks’ gestation, at which time TVUS assessment is stopped. Progesterone injections continue through 36 weeks, and the patient spontaneously delivers a healthy 3,700-g infant at term.
1. Hamilton BE, Martin JA, Ventura SJ. Births: Preliminary data for 2007. National vital statistics reports; 2009;57(12). National Center for Health Statistics Web site. http://www.cdc.gov/nchs/data/nvsr/nvsr57/nvsr57_12.pdf. March 18, 2009. Accessed March 30, 2010.
2. Iams JD, Johnson FF, Sonek J, et al. Cervical competence as a continuum: a study of ultrasonographic cervical length and obstetric performance. Am J Obstet Gynecol. 1995;172(4 pt 1):1097-1106.
3. Iams JD, Goldenberg RL, Meis PJ, et al. The length of the cervix and the risk of spontaneous premature delivery. National Institute of Child Health and Human Development Maternal Fetal Medicine Unit Network. N Engl J Med. 1996;334(9):567-572.
4. Clinical Management Guidelines for Obstetrician-Gynecologists. No. 43, May 2003: Management of Preterm Labor. Obstet Gynecol. 2003;101(5 pt 1):1039-1047.
5. Owen J, Iams JD, Hauth JC. Vaginal sonography and cervical incompetence. Am J Obstet Gynecol. 2003;188(2):586-596.
6. Harger JH. Cerclage and cervical insufficiency: an evidence-based analysis. Obstet Gynecol. 2002;100(6):1313-1327.
7. Clinical Management Guidelines for Obstetrician-Gynecologists. No. 48, Nov 2003: Cervical insufficiency. Obstet Gynecol. 2003;102(5 pt 1):1091-1099.
8. Kusanovic JP, Espinoza J, Romero R, et al. Clinical significance of the presence of amniotic fluid ‘sludge’ in asymptomatic patients at high risk for spontaneous preterm delivery. Ultrasound Obstet Gynecol. 2007;30(5):706-714.
9. ACOG Practice Bulletin No. 80. Premature rupture of membranes. Obstet Gynecol. 2007;109(4):1007-1020.DOI:10.1097/01.AOG.0000263888.69178.1f.
10. Berghella V, Odibo AO, To MS, Rust OA, Althuisius SM. Cerclage for short cervix on ultrasonography: meta-analysis of trials using individual patient-level data. Obstet Gynecol. 2005;106(1):181-189.
11. Owen J, Hankins G, Iams JD, et al. Multicenter randomized trial of cerclage for preterm birth prevention in high-risk women with shortened mid-trimester cervical length. Am J Obstet Gynecol. 2009;201(4):375.e1-e8.
12. Simcox R, Seed PT, Bennett P, Teoh TG, Poston L, Shennan AH. A randomized control trial of cervical scanning vs history to determine cerclage in women at high risk of preterm birth (CIRCLE trial). Am J Obstet Gynecol. 2009;200(6):623.e1-e6.
13. Iams JD. Prediction and early detection of preterm labor. Obstet Gynecol. 2003;101(2):402-412.
14. Romero R, Oyarzun E, Mazor M, Sirtori M, Hobbins JC, Bracken M. Meta-analysis of the relationship between asymptomatic bacteriuria and preterm delivery/low birth weight. Obstet Gynecol. 1989;73(4):576-582.
15. Leitich H, Brunbauer M, Bodnar-Aderl B, Kaider A, Egarter C, Husslein P. Antibiotic treatment of bacterial vaginosis in pregnancy: a meta-analysis. Am J Obstet Gynecol. 2003;188(3):752-758.
16. Meis PJ, Klebanoff M, Thom E, et al. For National Institute of Child Health and Human Development Maternal-Fetal Medicine Units Network. Prevention of recurrent preterm birth by 17 alpha-hydroxyprogesterone caproate. N Engl J Med. 2003;348(24):2379-2385.
17. Norman JE, Mackenzie F, Owen P, et al. Progesterone for the prevention of preterm birth in twin pregnancy (STOPPIT): a randomised, double-blind, placebo-controlled study and meta-analysis. Lancet. 2009;373(9680):2034-2040.
18. Rouse DJ, Caritis SN, Peaceman AM, et al. For National Institute of Child Health and Human Development Maternal-Fetal Medicine Units Network. A trial of 17 alpha-hydroxyprogesterone caproate to prevent prematurity in twins. N Engl J Med. 2007;357(5):454-461.
19. Caritis SN, Rouse DJ, Peaceman AM, et al. for Eunice Kennedy Shriver National Institute of Child Health and Human Development (NICHD), Medicine Units Network (MFMU). Prevention of preterm birth in triplets using 17 alpha-hydroxyprogesterone caproate: a randomized controlled trial. Obstet Gynecol. 2009;113(2 pt 1):285-292.
20. O’Brien JM, Adair CD, Lewis DF, et al. Progesterone vaginal gel for the reduction of recurrent preterm birth: primary results from a randomized, double-blind, placebo-controlled trial. Ultrasound Obstet Gynecol. 2007;30(5):687-696.
21. DeFranco EA, O’Brien JM, Adair CD, et al. Vaginal progesterone is associated with a decrease in risk for early preterm birth and improved neonatal outcome in women with a short cervix: a secondary analysis from a randomized, double-blind, placebo-controlled trial. Ultrasound Obstet Gynecol. 2007;30(5):697-705.
22. Fonseca EB, Celik E, Parra M, et al. For Fetal Medicine Foundation Second Trimester Screening Group. Progesterone and the risk of preterm birth among women with a short cervix. N Engl J Med. 2007;357(5):462-469.
23. ACOG Committee Opinion No. 419: Use of progesterone to reduce preterm birth. Obstet Gynecol. 2008;112(4):963.-
24. Yost NP, Bloom SL, Twickler DM, Leveno KJ. Pitfalls in ultrasonic cervical length measurement for predicting preterm birth. Obstet Gynecol. 1999;93(4):510-516.
1. Hamilton BE, Martin JA, Ventura SJ. Births: Preliminary data for 2007. National vital statistics reports; 2009;57(12). National Center for Health Statistics Web site. http://www.cdc.gov/nchs/data/nvsr/nvsr57/nvsr57_12.pdf. March 18, 2009. Accessed March 30, 2010.
2. Iams JD, Johnson FF, Sonek J, et al. Cervical competence as a continuum: a study of ultrasonographic cervical length and obstetric performance. Am J Obstet Gynecol. 1995;172(4 pt 1):1097-1106.
3. Iams JD, Goldenberg RL, Meis PJ, et al. The length of the cervix and the risk of spontaneous premature delivery. National Institute of Child Health and Human Development Maternal Fetal Medicine Unit Network. N Engl J Med. 1996;334(9):567-572.
4. Clinical Management Guidelines for Obstetrician-Gynecologists. No. 43, May 2003: Management of Preterm Labor. Obstet Gynecol. 2003;101(5 pt 1):1039-1047.
5. Owen J, Iams JD, Hauth JC. Vaginal sonography and cervical incompetence. Am J Obstet Gynecol. 2003;188(2):586-596.
6. Harger JH. Cerclage and cervical insufficiency: an evidence-based analysis. Obstet Gynecol. 2002;100(6):1313-1327.
7. Clinical Management Guidelines for Obstetrician-Gynecologists. No. 48, Nov 2003: Cervical insufficiency. Obstet Gynecol. 2003;102(5 pt 1):1091-1099.
8. Kusanovic JP, Espinoza J, Romero R, et al. Clinical significance of the presence of amniotic fluid ‘sludge’ in asymptomatic patients at high risk for spontaneous preterm delivery. Ultrasound Obstet Gynecol. 2007;30(5):706-714.
9. ACOG Practice Bulletin No. 80. Premature rupture of membranes. Obstet Gynecol. 2007;109(4):1007-1020.DOI:10.1097/01.AOG.0000263888.69178.1f.
10. Berghella V, Odibo AO, To MS, Rust OA, Althuisius SM. Cerclage for short cervix on ultrasonography: meta-analysis of trials using individual patient-level data. Obstet Gynecol. 2005;106(1):181-189.
11. Owen J, Hankins G, Iams JD, et al. Multicenter randomized trial of cerclage for preterm birth prevention in high-risk women with shortened mid-trimester cervical length. Am J Obstet Gynecol. 2009;201(4):375.e1-e8.
12. Simcox R, Seed PT, Bennett P, Teoh TG, Poston L, Shennan AH. A randomized control trial of cervical scanning vs history to determine cerclage in women at high risk of preterm birth (CIRCLE trial). Am J Obstet Gynecol. 2009;200(6):623.e1-e6.
13. Iams JD. Prediction and early detection of preterm labor. Obstet Gynecol. 2003;101(2):402-412.
14. Romero R, Oyarzun E, Mazor M, Sirtori M, Hobbins JC, Bracken M. Meta-analysis of the relationship between asymptomatic bacteriuria and preterm delivery/low birth weight. Obstet Gynecol. 1989;73(4):576-582.
15. Leitich H, Brunbauer M, Bodnar-Aderl B, Kaider A, Egarter C, Husslein P. Antibiotic treatment of bacterial vaginosis in pregnancy: a meta-analysis. Am J Obstet Gynecol. 2003;188(3):752-758.
16. Meis PJ, Klebanoff M, Thom E, et al. For National Institute of Child Health and Human Development Maternal-Fetal Medicine Units Network. Prevention of recurrent preterm birth by 17 alpha-hydroxyprogesterone caproate. N Engl J Med. 2003;348(24):2379-2385.
17. Norman JE, Mackenzie F, Owen P, et al. Progesterone for the prevention of preterm birth in twin pregnancy (STOPPIT): a randomised, double-blind, placebo-controlled study and meta-analysis. Lancet. 2009;373(9680):2034-2040.
18. Rouse DJ, Caritis SN, Peaceman AM, et al. For National Institute of Child Health and Human Development Maternal-Fetal Medicine Units Network. A trial of 17 alpha-hydroxyprogesterone caproate to prevent prematurity in twins. N Engl J Med. 2007;357(5):454-461.
19. Caritis SN, Rouse DJ, Peaceman AM, et al. for Eunice Kennedy Shriver National Institute of Child Health and Human Development (NICHD), Medicine Units Network (MFMU). Prevention of preterm birth in triplets using 17 alpha-hydroxyprogesterone caproate: a randomized controlled trial. Obstet Gynecol. 2009;113(2 pt 1):285-292.
20. O’Brien JM, Adair CD, Lewis DF, et al. Progesterone vaginal gel for the reduction of recurrent preterm birth: primary results from a randomized, double-blind, placebo-controlled trial. Ultrasound Obstet Gynecol. 2007;30(5):687-696.
21. DeFranco EA, O’Brien JM, Adair CD, et al. Vaginal progesterone is associated with a decrease in risk for early preterm birth and improved neonatal outcome in women with a short cervix: a secondary analysis from a randomized, double-blind, placebo-controlled trial. Ultrasound Obstet Gynecol. 2007;30(5):697-705.
22. Fonseca EB, Celik E, Parra M, et al. For Fetal Medicine Foundation Second Trimester Screening Group. Progesterone and the risk of preterm birth among women with a short cervix. N Engl J Med. 2007;357(5):462-469.
23. ACOG Committee Opinion No. 419: Use of progesterone to reduce preterm birth. Obstet Gynecol. 2008;112(4):963.-
24. Yost NP, Bloom SL, Twickler DM, Leveno KJ. Pitfalls in ultrasonic cervical length measurement for predicting preterm birth. Obstet Gynecol. 1999;93(4):510-516.
