User login
Lung Cancer Screening After Pneumonia: Is It Necessary?
Grand Rounds: Five-Day-Old Infant With Hip "Clunk"
A 5-day-old infant was referred to the pediatric orthopedic clinic for evaluation of a left hip “clunk.” She is a firstborn child, born at full term (39 weeks) via cesarean delivery secondary to breech presentation. Her weight at birth was 7 lb 6 oz. The infant was noted to have a left hip clunk during a routine physical examination by her pediatrician, who made a referral to the pediatric orthopedic clinic for possible hip dysplasia. This is the patient’s first visit to the clinic.
There is no family history of hip dysplasia or other orthopedic abnormalities. The infant is a well-appearing, alert female measuring 20.5” in length and weighing 7 lb 4 oz. Vital signs are stable with no abnormality detected. The heart is regular in rate and rhythm, and the chest is clear bilaterally.
No cutaneous abnormalities are noted. The patient is able to move all her extremities spontaneously, and her spine is straight and normal with no evidence of spinal dysraphism. Her feet are normal bilaterally, with full range of motion and no equinovarus or metatarsus adductus deformity.
The neurologic examination is also unremarkable, with normal neonatal reflexes and excellent muscle tone throughout.
Examination of the infant’s hips reveals a positive result on the Barlow test on the left side (the hip can be dislocated). There is also a positive Ortolani sign (the hip can be reduced), with asymmetric thigh skin folds noted (see Figures 1A and 1B, respectively).
Based on these positive physical examination findings, the patient was diagnosed with developmental dysplasia of the hip (DDH). Initial ultrasonography to confirm the diagnosis was not considered necessary, as the physical examination demonstrated obvious instability.1 The infant was placed in a Pavlik harness, which her parents were instructed should be worn full-time (see Figures 2A and 2B). She was scheduled for weekly follow-up visits for adjustments to the harness and serial hip examinations.
At the second follow-up visit, ultrasonography was performed, confirming the presence of dysplasia with decreased femoral head coverage and a steep socket (acetabulum). Use of the Pavlik harness was continued full-time for six weeks.
At age 6 weeks, the infant underwent a follow-up ultrasound to assess for improvement in the degree of dysplasia. The test revealed normal hips bilaterally with no evidence of DDH. Therefore, use of the Pavlik harness was discontinued. The parents were instructed to bring the child back in six months for a repeat clinical examination and an anteroposterior x-ray of the pelvis.1
Discussion
The term developmental dysplasia of the hip (DDH) has replaced the more traditional term congenital hip dislocation because DDH more accurately reflects the variable characteristics that can be seen with this condition. As DDH may not be present at birth, the term congenital is misleading. We now know that DDH may occur in utero, perinatally, or during infancy and childhood.2,3
Generally, DDH is used to describe an abnormal relationship between the femoral head and the acetabulum (see Figure 34). The term represents a wide spectrum of abnormality, as shown in the Graf classification of hips in infants: type I refers to a normal hip; type II, immature development to mild dysplasia; type III, subluxation of the femoral head; and type 4, frank dislocation with severe instability.5
Diagnosing and managing DDH correctly requires the clinician to have a thorough understanding of the normal growth and development that occurs in the hip joint. Embryologically, the joint (including the femoral head and acetabulum) develops from the same primitive mesenchymal cells.6 By 11 to 12 weeks’ gestation, the initial structures of the hip joint are fully formed; theoretically, this is the earliest time at which a dislocation can occur.2,7 DDH that develops at this stage would be called teratologic; this condition is seen most frequently in patients who have underlying neuromuscular conditions, such as myelodysplasia (spina bifida) or arthrogryposis. A typical dislocation takes place during the perinatal period in an infant who is otherwise healthy.2
Etiology
DDH occurs in about 11 of every 1,000 infants, with frank dislocations occurring in one to two infants per 10,000.8 The left hip is involved in approximately 60% of cases, the right in 20%, and both hips in about 20%. In the most common intrauterine fetal position, the left hip is lower than the right (usually abutting the mother’s sacrum) and is often in adduction. This is likely the reason that the left hip is more commonly affected by DDH.
DDH is believed to be multifactorial, with physiologic, genetic, and mechanical factors implicated in the etiology.3 The incidence of DDH varies with factors such as the patient’s age, race, and gender, the experience and training of the examiner, and the diagnostic criteria that are used.
Known risk factors for a positive newborn screening are shown in the table.9,10 It is often helpful for clinicians to remember the “4F” mnemonic associated with DDH: female, firstborn, foot first, and family history.9
There is also an increased risk for DDH in patients with other conditions that are associated with intrauterine crowding. These include congenital muscular torticollis, metatarsus adductus, and congenital dislocation of the knee.2
Physical Examination
All newborn infants should be screened for DDH as part of the initial physical examination, with ultrasonography recommended for infants deemed at high risk for DDH and for those with inconclusive results on examination.1,10,11 Providers should be aware that the newborn hip examination requires a considerable amount of practice and expertise.
A thorough medical history should always be obtained first, including gestational age, presentation (breech vs vertex), type of delivery (cesarean vs vaginal), gender, birth order, family history of DDH, ligamentous laxity, or myopathy.8
The examining clinician begins by placing the infant on a firm, flat surface. The infant should be as relaxed as possible. Next, the clinician observes both lower extremities for asymmetric thigh or buttock skin folds. Bilateral DDH can be very difficult to diagnose on the basis of this examination due to the lack of asymmetry (hips will have symmetric abnormality).
The Galeazzi sign is elicited by placing the infant supine with the hips and knees flexed to 90°.12 With the hips in neutral abduction, the provider should determine whether the knees are at the same height. Unequal knee heights—a positive result for the Galeazzi sign—suggest femoral shortening (apparent leg length discrepancy), which may be explained by a hip dislocation. If both hips are dislocated, a false-negative result will often occur, since both will appear short and there will be no discrepancy.2,12
Among physical examination techniques, the Ortolani and Barlow maneuvers are considered most reliable to detect hip instability in newborns and infants younger than 6 months2,13,14 (review Figures 1A and 1B). The Ortolani test is used to detect the sensation of the dislocated hip reducing into the acetabulum, and the Barlow test elicits the unstable hip dislocating.2 A palpable and occasionally audible clunk is considered a positive result on the Barlow test and usually indicates a diagnosis of DDH.14 High-pitched clicks or snaps frequently occur with hip range-of-motion maneuvers and during Ortolani and Barlow testing. These sounds are often attributed to snapping of the iliotibial band over the greater trochanter and do not usually signify dysplasia.15
Because DDH is a dynamic and evolving process, the physical findings on clinical examination change significantly, depending on the age of the infant or child. As an infant approaches age 3 months, limited hip abduction (especially when asymmetric) is often the most reliable physical examination finding in patients with DDH.12 After age 3 to 4 months, Ortolani and Barlow testing will often produce negative results as progressive soft tissue contractures evolve.
Once a child begins to walk, gait abnormalities (eg, a short-limbed or waddling gait pattern) may raise suspicion for a diagnosis of DDH.7 It has been recommended that evaluation for DDH be performed at each routine office examination until the child is 12 months of age.1
Treatment
The Pavlik harness is considered first-line treatment for DDH in infants younger than 6 months. The harness is a dynamic splint that allows the infant to engage in a sphere of active motion that encourages stabilization and deepening of the socket. The harness is applied with the knees flexed to about 90° and the hips in about 70° of abduction and 100° to 110° of flexion (as shown in Figures 2A and 2B).9
The duration of treatment depends on the infant’s age at presentation and the severity of DDH. Progress is judged by serial examinations and dynamic ultrasounds. The harness is worn full-time until clinical and radiographic examinations both yield normal results. After six weeks of treatment, the hips are examined out of the harness, and a repeat ultrasound is usually obtained. If findings are normal, use of the harness is ordinarily discontinued. Some patients will require harness use for a longer period in cases of delayed development of the acetabulum and/or severe laxity of the ligaments.9
The Pavlik harness is successful more than 90% of the time in newborns with DDH.8 Success rates have been reported as greatest in infants younger than 8 weeks at the time of treatment initiation, those with only one affected hip, and those with less severe disease (Graf types II or III).16
If ultrasonography shows no improvement after two to three weeks, it is usually recommended that the harness be discontinued; most orthopedic surgeons will then proceed with a closed or open reduction and spica body casting. Similarly, when the diagnosis of DDH is delayed until after ages 6 to 8 months, a closed reduction under anesthesia and placement of a spica body cast is usually the recommended treatment to maintain the hip in the reduced position.17,18 Some older children (ages 1 to 5 years) may require bracing, traction, open reduction, and/or femoral or pelvic osteotomy.17,18 It is believed that undiagnosed, untreated DDH can lead to early-onset degenerative hip disease (arthritis).1
Patient/Family Education
The Pavlik harness is most effective when a consistent support system exists to educate parents about the importance of the harness, its care and maintenance, and the consequences of failure. Close monitoring of the infant’s progress is also essential to promoting adherence. Application and removal of the harness should be demonstrated to the parent or caregiver, as well as diapering, dressing, and undressing the infant; they should then be encouraged to practice immediately in the clinic or office.
During visits for harness adjustment, the strap position should be marked with indelible ink, allowing parents to reapply the device correctly, should removal be required (eg, for bathing).9 Ten percent of parents reportedly find reapplying the harness difficult during the first weeks of use. Difficulty in dressing and carrying an infant in a harness, feet slipping out of the harness, and skin irritation have been reported by about one-third of parents.19
Treatment adherence and subsequent success with the Pavlik harness is reported greatest (95%) in patients whose parents engage in demonstrations of harness use and follow instructions precisely.19 By providing a contact name and office number and following up with a phone call a few days after the harness is first applied, clinicians can significantly decrease parents’ anxiety and increase overall compliance.9
Conclusion
Despite recent increased awareness of DDH and the importance of thorough screening programs, hip dysplasia continues to be a frequently missed diagnosis in pediatrics. It is often up to the primary care clinician to screen for, assess, and potentially diagnose DDH. Therefore, a thorough understanding of this condition can promote early detection and diagnosis, with less invasive treatment and a more favorable outcome.
A proper hip examination should be a standard component of all newborn and infant well-child examinations. If DDH is suspected, appropriate referral to a pediatric orthopedic surgeon must be made so that timely treatment can be initiated. Early use of the Pavlik harness is significantly easier than the invasive surgery and prolonged immobilization necessitated by a delayed diagnosis. Whatever the course of treatment required, it is important for clinicians to support the patient and family: training and anticipatory guidance are essential components of DDH management.
1. Karmazyn BK, Gunderman RB, Coley BD, et al; American College of Radiology. ACR appropriateness criteria on developmental dysplasia of the hip—child. J Am Coll Radiol. 2009;6(8):551-557.
2. American Academy of Pediatrics, Committee on Quality Improvement, Subcommittee on Developmental Dysplasia of the Hip. Clinical practice guideline: early detection of developmental dysplasia of the hip. Pediatrics. 2000;105(4 pt 1):896-905.
3. Mencio GA. Developmental dysplasia of the hip. In: Sponseller PD, ed. Orthopaedic Knowledge Update: Pediatrics–2. Rosemont, IL: American Academy of Orthopaedic Surgeons; 2002:161-171.
4. Children’s Hospital at Westmead. Developmental dysplasia of the hip (DDH). www.chw.edu.au/parents/factsheets/developj.htm. Accessed March 26, 2010.
5. Graf R. Classification of hip joint dysplasia by means of sonography. Arch Orthop Trauma Surg. 1984; 102:248-255.
6. Weinstein SL. Developmental hip dysplasia and dislocation. In: Morrissy RT, Weinstein SL, eds. Lovell and Winter’s Pediatric Orthopaedics. 6th ed. Philadelphia, PA: Lippincott Williams & Wilkins; 2005:905-956.
7. Aronsson DD, Goldberg MJ, Kling TF Jr, Roy DR. Developmental dysplasia of the hip. Pediatrics. 1994; 94(2 pt 1):201-208.
8. Guille JT, Pizzutillo PD, MacEwan GD. Developmental dysplasia of the hip from birth to six months. J Am Acad Orthop Surg. 2000;8(4):232-242.
9. Hart ES, Albright MB, Rebello GN, Grottkau BE. Developmental dysplasia of the hip: nursing implications and anticipatory guidelines for parents. Orthop Nurs. 2006;25(2):100-109.
10. Dogruel H, Atalar H, Yavus OY, Sayli U. Clinical examination versus ultrasonography in detecting developmental dysplasia of the hip. Int Orthop. 2008; 32(3):415-419.
11. Mahan ST, Katz JN, Kim YJ. To screen or not to screen? A decision analysis of the utility of screening for developmental dysplasia of the hip. J Bone Joint Surg Am. 2009;91(7);1705-1719.
12. Storer SK, Skaggs DL. Developmental dysplasia of the hip. Am Fam Physician. 2006;74(8):1310-1316.
13. Ortolani M. Congenital hip dysplasia in the light of early and very early diagnosis. Clin Orthop Relat Res. 1976;119(1):6-10.
14. Barlow TG. Congenital dislocation of the hip in the newborn. Proc R Soc Med. 1966;59(11 part 1):1103-1106.
15. Bond CD, Hennrikus WL, DellaMaggiore ED. Prospective evaluation of newborn soft-tissue “clicks” with ultrasound. J Pediatr Orthop. 1997;17(2):199-201.
16. Atalar H, Sayli U, Yavuz OY, et al. Indicators of successful use of the Pavlik harness in infants with developmental dysplasia of the hip. Int Orthop. 2007; 31(2):145-150.
17. Rampal V, Sabourin M, Erdeneshoo E, et al. Closed reduction with traction for developmental dysplasia of the hip in children aged between one and five years. J Bone Joint Surg Br. 2008;90-B(7):858-863.
18. Clarke NMP, Sakthivel K. The diagnosis and management of congenital dislocation of the hip. Paediatr Child Health. 2008;18(6):268-271.
19. Hassan FA. Compliance of parents with regard to Pavlik harness treatment in developmental dysplasia of the hip. J Pediatr Orthop. 2009;18(3):111-115.
A 5-day-old infant was referred to the pediatric orthopedic clinic for evaluation of a left hip “clunk.” She is a firstborn child, born at full term (39 weeks) via cesarean delivery secondary to breech presentation. Her weight at birth was 7 lb 6 oz. The infant was noted to have a left hip clunk during a routine physical examination by her pediatrician, who made a referral to the pediatric orthopedic clinic for possible hip dysplasia. This is the patient’s first visit to the clinic.
There is no family history of hip dysplasia or other orthopedic abnormalities. The infant is a well-appearing, alert female measuring 20.5” in length and weighing 7 lb 4 oz. Vital signs are stable with no abnormality detected. The heart is regular in rate and rhythm, and the chest is clear bilaterally.
No cutaneous abnormalities are noted. The patient is able to move all her extremities spontaneously, and her spine is straight and normal with no evidence of spinal dysraphism. Her feet are normal bilaterally, with full range of motion and no equinovarus or metatarsus adductus deformity.
The neurologic examination is also unremarkable, with normal neonatal reflexes and excellent muscle tone throughout.
Examination of the infant’s hips reveals a positive result on the Barlow test on the left side (the hip can be dislocated). There is also a positive Ortolani sign (the hip can be reduced), with asymmetric thigh skin folds noted (see Figures 1A and 1B, respectively).
Based on these positive physical examination findings, the patient was diagnosed with developmental dysplasia of the hip (DDH). Initial ultrasonography to confirm the diagnosis was not considered necessary, as the physical examination demonstrated obvious instability.1 The infant was placed in a Pavlik harness, which her parents were instructed should be worn full-time (see Figures 2A and 2B). She was scheduled for weekly follow-up visits for adjustments to the harness and serial hip examinations.
At the second follow-up visit, ultrasonography was performed, confirming the presence of dysplasia with decreased femoral head coverage and a steep socket (acetabulum). Use of the Pavlik harness was continued full-time for six weeks.
At age 6 weeks, the infant underwent a follow-up ultrasound to assess for improvement in the degree of dysplasia. The test revealed normal hips bilaterally with no evidence of DDH. Therefore, use of the Pavlik harness was discontinued. The parents were instructed to bring the child back in six months for a repeat clinical examination and an anteroposterior x-ray of the pelvis.1
Discussion
The term developmental dysplasia of the hip (DDH) has replaced the more traditional term congenital hip dislocation because DDH more accurately reflects the variable characteristics that can be seen with this condition. As DDH may not be present at birth, the term congenital is misleading. We now know that DDH may occur in utero, perinatally, or during infancy and childhood.2,3
Generally, DDH is used to describe an abnormal relationship between the femoral head and the acetabulum (see Figure 34). The term represents a wide spectrum of abnormality, as shown in the Graf classification of hips in infants: type I refers to a normal hip; type II, immature development to mild dysplasia; type III, subluxation of the femoral head; and type 4, frank dislocation with severe instability.5
Diagnosing and managing DDH correctly requires the clinician to have a thorough understanding of the normal growth and development that occurs in the hip joint. Embryologically, the joint (including the femoral head and acetabulum) develops from the same primitive mesenchymal cells.6 By 11 to 12 weeks’ gestation, the initial structures of the hip joint are fully formed; theoretically, this is the earliest time at which a dislocation can occur.2,7 DDH that develops at this stage would be called teratologic; this condition is seen most frequently in patients who have underlying neuromuscular conditions, such as myelodysplasia (spina bifida) or arthrogryposis. A typical dislocation takes place during the perinatal period in an infant who is otherwise healthy.2
Etiology
DDH occurs in about 11 of every 1,000 infants, with frank dislocations occurring in one to two infants per 10,000.8 The left hip is involved in approximately 60% of cases, the right in 20%, and both hips in about 20%. In the most common intrauterine fetal position, the left hip is lower than the right (usually abutting the mother’s sacrum) and is often in adduction. This is likely the reason that the left hip is more commonly affected by DDH.
DDH is believed to be multifactorial, with physiologic, genetic, and mechanical factors implicated in the etiology.3 The incidence of DDH varies with factors such as the patient’s age, race, and gender, the experience and training of the examiner, and the diagnostic criteria that are used.
Known risk factors for a positive newborn screening are shown in the table.9,10 It is often helpful for clinicians to remember the “4F” mnemonic associated with DDH: female, firstborn, foot first, and family history.9
There is also an increased risk for DDH in patients with other conditions that are associated with intrauterine crowding. These include congenital muscular torticollis, metatarsus adductus, and congenital dislocation of the knee.2
Physical Examination
All newborn infants should be screened for DDH as part of the initial physical examination, with ultrasonography recommended for infants deemed at high risk for DDH and for those with inconclusive results on examination.1,10,11 Providers should be aware that the newborn hip examination requires a considerable amount of practice and expertise.
A thorough medical history should always be obtained first, including gestational age, presentation (breech vs vertex), type of delivery (cesarean vs vaginal), gender, birth order, family history of DDH, ligamentous laxity, or myopathy.8
The examining clinician begins by placing the infant on a firm, flat surface. The infant should be as relaxed as possible. Next, the clinician observes both lower extremities for asymmetric thigh or buttock skin folds. Bilateral DDH can be very difficult to diagnose on the basis of this examination due to the lack of asymmetry (hips will have symmetric abnormality).
The Galeazzi sign is elicited by placing the infant supine with the hips and knees flexed to 90°.12 With the hips in neutral abduction, the provider should determine whether the knees are at the same height. Unequal knee heights—a positive result for the Galeazzi sign—suggest femoral shortening (apparent leg length discrepancy), which may be explained by a hip dislocation. If both hips are dislocated, a false-negative result will often occur, since both will appear short and there will be no discrepancy.2,12
Among physical examination techniques, the Ortolani and Barlow maneuvers are considered most reliable to detect hip instability in newborns and infants younger than 6 months2,13,14 (review Figures 1A and 1B). The Ortolani test is used to detect the sensation of the dislocated hip reducing into the acetabulum, and the Barlow test elicits the unstable hip dislocating.2 A palpable and occasionally audible clunk is considered a positive result on the Barlow test and usually indicates a diagnosis of DDH.14 High-pitched clicks or snaps frequently occur with hip range-of-motion maneuvers and during Ortolani and Barlow testing. These sounds are often attributed to snapping of the iliotibial band over the greater trochanter and do not usually signify dysplasia.15
Because DDH is a dynamic and evolving process, the physical findings on clinical examination change significantly, depending on the age of the infant or child. As an infant approaches age 3 months, limited hip abduction (especially when asymmetric) is often the most reliable physical examination finding in patients with DDH.12 After age 3 to 4 months, Ortolani and Barlow testing will often produce negative results as progressive soft tissue contractures evolve.
Once a child begins to walk, gait abnormalities (eg, a short-limbed or waddling gait pattern) may raise suspicion for a diagnosis of DDH.7 It has been recommended that evaluation for DDH be performed at each routine office examination until the child is 12 months of age.1
Treatment
The Pavlik harness is considered first-line treatment for DDH in infants younger than 6 months. The harness is a dynamic splint that allows the infant to engage in a sphere of active motion that encourages stabilization and deepening of the socket. The harness is applied with the knees flexed to about 90° and the hips in about 70° of abduction and 100° to 110° of flexion (as shown in Figures 2A and 2B).9
The duration of treatment depends on the infant’s age at presentation and the severity of DDH. Progress is judged by serial examinations and dynamic ultrasounds. The harness is worn full-time until clinical and radiographic examinations both yield normal results. After six weeks of treatment, the hips are examined out of the harness, and a repeat ultrasound is usually obtained. If findings are normal, use of the harness is ordinarily discontinued. Some patients will require harness use for a longer period in cases of delayed development of the acetabulum and/or severe laxity of the ligaments.9
The Pavlik harness is successful more than 90% of the time in newborns with DDH.8 Success rates have been reported as greatest in infants younger than 8 weeks at the time of treatment initiation, those with only one affected hip, and those with less severe disease (Graf types II or III).16
If ultrasonography shows no improvement after two to three weeks, it is usually recommended that the harness be discontinued; most orthopedic surgeons will then proceed with a closed or open reduction and spica body casting. Similarly, when the diagnosis of DDH is delayed until after ages 6 to 8 months, a closed reduction under anesthesia and placement of a spica body cast is usually the recommended treatment to maintain the hip in the reduced position.17,18 Some older children (ages 1 to 5 years) may require bracing, traction, open reduction, and/or femoral or pelvic osteotomy.17,18 It is believed that undiagnosed, untreated DDH can lead to early-onset degenerative hip disease (arthritis).1
Patient/Family Education
The Pavlik harness is most effective when a consistent support system exists to educate parents about the importance of the harness, its care and maintenance, and the consequences of failure. Close monitoring of the infant’s progress is also essential to promoting adherence. Application and removal of the harness should be demonstrated to the parent or caregiver, as well as diapering, dressing, and undressing the infant; they should then be encouraged to practice immediately in the clinic or office.
During visits for harness adjustment, the strap position should be marked with indelible ink, allowing parents to reapply the device correctly, should removal be required (eg, for bathing).9 Ten percent of parents reportedly find reapplying the harness difficult during the first weeks of use. Difficulty in dressing and carrying an infant in a harness, feet slipping out of the harness, and skin irritation have been reported by about one-third of parents.19
Treatment adherence and subsequent success with the Pavlik harness is reported greatest (95%) in patients whose parents engage in demonstrations of harness use and follow instructions precisely.19 By providing a contact name and office number and following up with a phone call a few days after the harness is first applied, clinicians can significantly decrease parents’ anxiety and increase overall compliance.9
Conclusion
Despite recent increased awareness of DDH and the importance of thorough screening programs, hip dysplasia continues to be a frequently missed diagnosis in pediatrics. It is often up to the primary care clinician to screen for, assess, and potentially diagnose DDH. Therefore, a thorough understanding of this condition can promote early detection and diagnosis, with less invasive treatment and a more favorable outcome.
A proper hip examination should be a standard component of all newborn and infant well-child examinations. If DDH is suspected, appropriate referral to a pediatric orthopedic surgeon must be made so that timely treatment can be initiated. Early use of the Pavlik harness is significantly easier than the invasive surgery and prolonged immobilization necessitated by a delayed diagnosis. Whatever the course of treatment required, it is important for clinicians to support the patient and family: training and anticipatory guidance are essential components of DDH management.
A 5-day-old infant was referred to the pediatric orthopedic clinic for evaluation of a left hip “clunk.” She is a firstborn child, born at full term (39 weeks) via cesarean delivery secondary to breech presentation. Her weight at birth was 7 lb 6 oz. The infant was noted to have a left hip clunk during a routine physical examination by her pediatrician, who made a referral to the pediatric orthopedic clinic for possible hip dysplasia. This is the patient’s first visit to the clinic.
There is no family history of hip dysplasia or other orthopedic abnormalities. The infant is a well-appearing, alert female measuring 20.5” in length and weighing 7 lb 4 oz. Vital signs are stable with no abnormality detected. The heart is regular in rate and rhythm, and the chest is clear bilaterally.
No cutaneous abnormalities are noted. The patient is able to move all her extremities spontaneously, and her spine is straight and normal with no evidence of spinal dysraphism. Her feet are normal bilaterally, with full range of motion and no equinovarus or metatarsus adductus deformity.
The neurologic examination is also unremarkable, with normal neonatal reflexes and excellent muscle tone throughout.
Examination of the infant’s hips reveals a positive result on the Barlow test on the left side (the hip can be dislocated). There is also a positive Ortolani sign (the hip can be reduced), with asymmetric thigh skin folds noted (see Figures 1A and 1B, respectively).
Based on these positive physical examination findings, the patient was diagnosed with developmental dysplasia of the hip (DDH). Initial ultrasonography to confirm the diagnosis was not considered necessary, as the physical examination demonstrated obvious instability.1 The infant was placed in a Pavlik harness, which her parents were instructed should be worn full-time (see Figures 2A and 2B). She was scheduled for weekly follow-up visits for adjustments to the harness and serial hip examinations.
At the second follow-up visit, ultrasonography was performed, confirming the presence of dysplasia with decreased femoral head coverage and a steep socket (acetabulum). Use of the Pavlik harness was continued full-time for six weeks.
At age 6 weeks, the infant underwent a follow-up ultrasound to assess for improvement in the degree of dysplasia. The test revealed normal hips bilaterally with no evidence of DDH. Therefore, use of the Pavlik harness was discontinued. The parents were instructed to bring the child back in six months for a repeat clinical examination and an anteroposterior x-ray of the pelvis.1
Discussion
The term developmental dysplasia of the hip (DDH) has replaced the more traditional term congenital hip dislocation because DDH more accurately reflects the variable characteristics that can be seen with this condition. As DDH may not be present at birth, the term congenital is misleading. We now know that DDH may occur in utero, perinatally, or during infancy and childhood.2,3
Generally, DDH is used to describe an abnormal relationship between the femoral head and the acetabulum (see Figure 34). The term represents a wide spectrum of abnormality, as shown in the Graf classification of hips in infants: type I refers to a normal hip; type II, immature development to mild dysplasia; type III, subluxation of the femoral head; and type 4, frank dislocation with severe instability.5
Diagnosing and managing DDH correctly requires the clinician to have a thorough understanding of the normal growth and development that occurs in the hip joint. Embryologically, the joint (including the femoral head and acetabulum) develops from the same primitive mesenchymal cells.6 By 11 to 12 weeks’ gestation, the initial structures of the hip joint are fully formed; theoretically, this is the earliest time at which a dislocation can occur.2,7 DDH that develops at this stage would be called teratologic; this condition is seen most frequently in patients who have underlying neuromuscular conditions, such as myelodysplasia (spina bifida) or arthrogryposis. A typical dislocation takes place during the perinatal period in an infant who is otherwise healthy.2
Etiology
DDH occurs in about 11 of every 1,000 infants, with frank dislocations occurring in one to two infants per 10,000.8 The left hip is involved in approximately 60% of cases, the right in 20%, and both hips in about 20%. In the most common intrauterine fetal position, the left hip is lower than the right (usually abutting the mother’s sacrum) and is often in adduction. This is likely the reason that the left hip is more commonly affected by DDH.
DDH is believed to be multifactorial, with physiologic, genetic, and mechanical factors implicated in the etiology.3 The incidence of DDH varies with factors such as the patient’s age, race, and gender, the experience and training of the examiner, and the diagnostic criteria that are used.
Known risk factors for a positive newborn screening are shown in the table.9,10 It is often helpful for clinicians to remember the “4F” mnemonic associated with DDH: female, firstborn, foot first, and family history.9
There is also an increased risk for DDH in patients with other conditions that are associated with intrauterine crowding. These include congenital muscular torticollis, metatarsus adductus, and congenital dislocation of the knee.2
Physical Examination
All newborn infants should be screened for DDH as part of the initial physical examination, with ultrasonography recommended for infants deemed at high risk for DDH and for those with inconclusive results on examination.1,10,11 Providers should be aware that the newborn hip examination requires a considerable amount of practice and expertise.
A thorough medical history should always be obtained first, including gestational age, presentation (breech vs vertex), type of delivery (cesarean vs vaginal), gender, birth order, family history of DDH, ligamentous laxity, or myopathy.8
The examining clinician begins by placing the infant on a firm, flat surface. The infant should be as relaxed as possible. Next, the clinician observes both lower extremities for asymmetric thigh or buttock skin folds. Bilateral DDH can be very difficult to diagnose on the basis of this examination due to the lack of asymmetry (hips will have symmetric abnormality).
The Galeazzi sign is elicited by placing the infant supine with the hips and knees flexed to 90°.12 With the hips in neutral abduction, the provider should determine whether the knees are at the same height. Unequal knee heights—a positive result for the Galeazzi sign—suggest femoral shortening (apparent leg length discrepancy), which may be explained by a hip dislocation. If both hips are dislocated, a false-negative result will often occur, since both will appear short and there will be no discrepancy.2,12
Among physical examination techniques, the Ortolani and Barlow maneuvers are considered most reliable to detect hip instability in newborns and infants younger than 6 months2,13,14 (review Figures 1A and 1B). The Ortolani test is used to detect the sensation of the dislocated hip reducing into the acetabulum, and the Barlow test elicits the unstable hip dislocating.2 A palpable and occasionally audible clunk is considered a positive result on the Barlow test and usually indicates a diagnosis of DDH.14 High-pitched clicks or snaps frequently occur with hip range-of-motion maneuvers and during Ortolani and Barlow testing. These sounds are often attributed to snapping of the iliotibial band over the greater trochanter and do not usually signify dysplasia.15
Because DDH is a dynamic and evolving process, the physical findings on clinical examination change significantly, depending on the age of the infant or child. As an infant approaches age 3 months, limited hip abduction (especially when asymmetric) is often the most reliable physical examination finding in patients with DDH.12 After age 3 to 4 months, Ortolani and Barlow testing will often produce negative results as progressive soft tissue contractures evolve.
Once a child begins to walk, gait abnormalities (eg, a short-limbed or waddling gait pattern) may raise suspicion for a diagnosis of DDH.7 It has been recommended that evaluation for DDH be performed at each routine office examination until the child is 12 months of age.1
Treatment
The Pavlik harness is considered first-line treatment for DDH in infants younger than 6 months. The harness is a dynamic splint that allows the infant to engage in a sphere of active motion that encourages stabilization and deepening of the socket. The harness is applied with the knees flexed to about 90° and the hips in about 70° of abduction and 100° to 110° of flexion (as shown in Figures 2A and 2B).9
The duration of treatment depends on the infant’s age at presentation and the severity of DDH. Progress is judged by serial examinations and dynamic ultrasounds. The harness is worn full-time until clinical and radiographic examinations both yield normal results. After six weeks of treatment, the hips are examined out of the harness, and a repeat ultrasound is usually obtained. If findings are normal, use of the harness is ordinarily discontinued. Some patients will require harness use for a longer period in cases of delayed development of the acetabulum and/or severe laxity of the ligaments.9
The Pavlik harness is successful more than 90% of the time in newborns with DDH.8 Success rates have been reported as greatest in infants younger than 8 weeks at the time of treatment initiation, those with only one affected hip, and those with less severe disease (Graf types II or III).16
If ultrasonography shows no improvement after two to three weeks, it is usually recommended that the harness be discontinued; most orthopedic surgeons will then proceed with a closed or open reduction and spica body casting. Similarly, when the diagnosis of DDH is delayed until after ages 6 to 8 months, a closed reduction under anesthesia and placement of a spica body cast is usually the recommended treatment to maintain the hip in the reduced position.17,18 Some older children (ages 1 to 5 years) may require bracing, traction, open reduction, and/or femoral or pelvic osteotomy.17,18 It is believed that undiagnosed, untreated DDH can lead to early-onset degenerative hip disease (arthritis).1
Patient/Family Education
The Pavlik harness is most effective when a consistent support system exists to educate parents about the importance of the harness, its care and maintenance, and the consequences of failure. Close monitoring of the infant’s progress is also essential to promoting adherence. Application and removal of the harness should be demonstrated to the parent or caregiver, as well as diapering, dressing, and undressing the infant; they should then be encouraged to practice immediately in the clinic or office.
During visits for harness adjustment, the strap position should be marked with indelible ink, allowing parents to reapply the device correctly, should removal be required (eg, for bathing).9 Ten percent of parents reportedly find reapplying the harness difficult during the first weeks of use. Difficulty in dressing and carrying an infant in a harness, feet slipping out of the harness, and skin irritation have been reported by about one-third of parents.19
Treatment adherence and subsequent success with the Pavlik harness is reported greatest (95%) in patients whose parents engage in demonstrations of harness use and follow instructions precisely.19 By providing a contact name and office number and following up with a phone call a few days after the harness is first applied, clinicians can significantly decrease parents’ anxiety and increase overall compliance.9
Conclusion
Despite recent increased awareness of DDH and the importance of thorough screening programs, hip dysplasia continues to be a frequently missed diagnosis in pediatrics. It is often up to the primary care clinician to screen for, assess, and potentially diagnose DDH. Therefore, a thorough understanding of this condition can promote early detection and diagnosis, with less invasive treatment and a more favorable outcome.
A proper hip examination should be a standard component of all newborn and infant well-child examinations. If DDH is suspected, appropriate referral to a pediatric orthopedic surgeon must be made so that timely treatment can be initiated. Early use of the Pavlik harness is significantly easier than the invasive surgery and prolonged immobilization necessitated by a delayed diagnosis. Whatever the course of treatment required, it is important for clinicians to support the patient and family: training and anticipatory guidance are essential components of DDH management.
1. Karmazyn BK, Gunderman RB, Coley BD, et al; American College of Radiology. ACR appropriateness criteria on developmental dysplasia of the hip—child. J Am Coll Radiol. 2009;6(8):551-557.
2. American Academy of Pediatrics, Committee on Quality Improvement, Subcommittee on Developmental Dysplasia of the Hip. Clinical practice guideline: early detection of developmental dysplasia of the hip. Pediatrics. 2000;105(4 pt 1):896-905.
3. Mencio GA. Developmental dysplasia of the hip. In: Sponseller PD, ed. Orthopaedic Knowledge Update: Pediatrics–2. Rosemont, IL: American Academy of Orthopaedic Surgeons; 2002:161-171.
4. Children’s Hospital at Westmead. Developmental dysplasia of the hip (DDH). www.chw.edu.au/parents/factsheets/developj.htm. Accessed March 26, 2010.
5. Graf R. Classification of hip joint dysplasia by means of sonography. Arch Orthop Trauma Surg. 1984; 102:248-255.
6. Weinstein SL. Developmental hip dysplasia and dislocation. In: Morrissy RT, Weinstein SL, eds. Lovell and Winter’s Pediatric Orthopaedics. 6th ed. Philadelphia, PA: Lippincott Williams & Wilkins; 2005:905-956.
7. Aronsson DD, Goldberg MJ, Kling TF Jr, Roy DR. Developmental dysplasia of the hip. Pediatrics. 1994; 94(2 pt 1):201-208.
8. Guille JT, Pizzutillo PD, MacEwan GD. Developmental dysplasia of the hip from birth to six months. J Am Acad Orthop Surg. 2000;8(4):232-242.
9. Hart ES, Albright MB, Rebello GN, Grottkau BE. Developmental dysplasia of the hip: nursing implications and anticipatory guidelines for parents. Orthop Nurs. 2006;25(2):100-109.
10. Dogruel H, Atalar H, Yavus OY, Sayli U. Clinical examination versus ultrasonography in detecting developmental dysplasia of the hip. Int Orthop. 2008; 32(3):415-419.
11. Mahan ST, Katz JN, Kim YJ. To screen or not to screen? A decision analysis of the utility of screening for developmental dysplasia of the hip. J Bone Joint Surg Am. 2009;91(7);1705-1719.
12. Storer SK, Skaggs DL. Developmental dysplasia of the hip. Am Fam Physician. 2006;74(8):1310-1316.
13. Ortolani M. Congenital hip dysplasia in the light of early and very early diagnosis. Clin Orthop Relat Res. 1976;119(1):6-10.
14. Barlow TG. Congenital dislocation of the hip in the newborn. Proc R Soc Med. 1966;59(11 part 1):1103-1106.
15. Bond CD, Hennrikus WL, DellaMaggiore ED. Prospective evaluation of newborn soft-tissue “clicks” with ultrasound. J Pediatr Orthop. 1997;17(2):199-201.
16. Atalar H, Sayli U, Yavuz OY, et al. Indicators of successful use of the Pavlik harness in infants with developmental dysplasia of the hip. Int Orthop. 2007; 31(2):145-150.
17. Rampal V, Sabourin M, Erdeneshoo E, et al. Closed reduction with traction for developmental dysplasia of the hip in children aged between one and five years. J Bone Joint Surg Br. 2008;90-B(7):858-863.
18. Clarke NMP, Sakthivel K. The diagnosis and management of congenital dislocation of the hip. Paediatr Child Health. 2008;18(6):268-271.
19. Hassan FA. Compliance of parents with regard to Pavlik harness treatment in developmental dysplasia of the hip. J Pediatr Orthop. 2009;18(3):111-115.
1. Karmazyn BK, Gunderman RB, Coley BD, et al; American College of Radiology. ACR appropriateness criteria on developmental dysplasia of the hip—child. J Am Coll Radiol. 2009;6(8):551-557.
2. American Academy of Pediatrics, Committee on Quality Improvement, Subcommittee on Developmental Dysplasia of the Hip. Clinical practice guideline: early detection of developmental dysplasia of the hip. Pediatrics. 2000;105(4 pt 1):896-905.
3. Mencio GA. Developmental dysplasia of the hip. In: Sponseller PD, ed. Orthopaedic Knowledge Update: Pediatrics–2. Rosemont, IL: American Academy of Orthopaedic Surgeons; 2002:161-171.
4. Children’s Hospital at Westmead. Developmental dysplasia of the hip (DDH). www.chw.edu.au/parents/factsheets/developj.htm. Accessed March 26, 2010.
5. Graf R. Classification of hip joint dysplasia by means of sonography. Arch Orthop Trauma Surg. 1984; 102:248-255.
6. Weinstein SL. Developmental hip dysplasia and dislocation. In: Morrissy RT, Weinstein SL, eds. Lovell and Winter’s Pediatric Orthopaedics. 6th ed. Philadelphia, PA: Lippincott Williams & Wilkins; 2005:905-956.
7. Aronsson DD, Goldberg MJ, Kling TF Jr, Roy DR. Developmental dysplasia of the hip. Pediatrics. 1994; 94(2 pt 1):201-208.
8. Guille JT, Pizzutillo PD, MacEwan GD. Developmental dysplasia of the hip from birth to six months. J Am Acad Orthop Surg. 2000;8(4):232-242.
9. Hart ES, Albright MB, Rebello GN, Grottkau BE. Developmental dysplasia of the hip: nursing implications and anticipatory guidelines for parents. Orthop Nurs. 2006;25(2):100-109.
10. Dogruel H, Atalar H, Yavus OY, Sayli U. Clinical examination versus ultrasonography in detecting developmental dysplasia of the hip. Int Orthop. 2008; 32(3):415-419.
11. Mahan ST, Katz JN, Kim YJ. To screen or not to screen? A decision analysis of the utility of screening for developmental dysplasia of the hip. J Bone Joint Surg Am. 2009;91(7);1705-1719.
12. Storer SK, Skaggs DL. Developmental dysplasia of the hip. Am Fam Physician. 2006;74(8):1310-1316.
13. Ortolani M. Congenital hip dysplasia in the light of early and very early diagnosis. Clin Orthop Relat Res. 1976;119(1):6-10.
14. Barlow TG. Congenital dislocation of the hip in the newborn. Proc R Soc Med. 1966;59(11 part 1):1103-1106.
15. Bond CD, Hennrikus WL, DellaMaggiore ED. Prospective evaluation of newborn soft-tissue “clicks” with ultrasound. J Pediatr Orthop. 1997;17(2):199-201.
16. Atalar H, Sayli U, Yavuz OY, et al. Indicators of successful use of the Pavlik harness in infants with developmental dysplasia of the hip. Int Orthop. 2007; 31(2):145-150.
17. Rampal V, Sabourin M, Erdeneshoo E, et al. Closed reduction with traction for developmental dysplasia of the hip in children aged between one and five years. J Bone Joint Surg Br. 2008;90-B(7):858-863.
18. Clarke NMP, Sakthivel K. The diagnosis and management of congenital dislocation of the hip. Paediatr Child Health. 2008;18(6):268-271.
19. Hassan FA. Compliance of parents with regard to Pavlik harness treatment in developmental dysplasia of the hip. J Pediatr Orthop. 2009;18(3):111-115.
INH-Associated Hepatotoxicity
Efficacy and Emergent Sequelae in the Use of Biologics for Psoriasis
Obesity and Its Relationship With Pelvic and Lower-Extremity Orthopedic Trauma
UPDATE: MINIMALLY INVASIVE SURGERY
Endometrial polyps are a relatively common pathology, occurring in 24% to 41% of women who have abnormal bleeding, and in about 10% of asymptomatic women.1,2 Endometrial polyps may be associated with leiomyomas in women who have abnormal bleeding.1-3
Polyps originate as focal hyperplasia of basal endometrium and contain variable amounts of glands, stroma, and blood vessels. Glandular epithelium has higher estrogen- and progesterone-receptor expression than surrounding endometrium, whereas the stromal component of a polyp has hormone receptors similar to endometrium. This suggests that a polyp represents focal hyperplasia that is more glandular than stromal.4
In this Update, I outline the basics of diagnosis and treatment and report on several recent investigations:
- a retrospective analysis from Italy that found that endometrial polyps are associated with advancing age and that any apparent association between polyps and diabetes, hypertension, or obesity is likely age-related
- a cross-sectional study from Norway that found that some asymptomatic polyps regress spontaneously, usually when their length is 10.7 mm or less
- three studies that explore the variables associated with premalignant and malignant polyps
- an investigation of the relationship between endometrial polyps and the background endometrium that found atypical hyperplasia in endometrium remote from the polyp in a significant percentage of women.
Age is the most important variable when assessing a patient for endometrial pathology
Nappi L, Indraccolo U, Di Spiezio Sardo A, et al. Are diabetes, hypertension, and obesity independent risk factors for endometrial polyps? J Minim Invasive Gynecol. 2009;16(2):157–162.
In this retrospective analysis of 353 women who underwent office hysteroscopy, Nappi and co-workers set out to ascertain whether endometrial polyps are associated with diabetes, hypertension, or obesity, independent of age and menopausal status. They did find an association between age, menopause, hypertension, obesity, and the presence of endometrial polyps. However, after multivariable logistic regression, all variables except age lost statistical significance. The median age at which polyps were present was 53 years (range: 29–86 years).
Details of the trial
A total of 394 consecutive Caucasian women underwent hysteroscopy to assess abnormal uterine bleeding, infertility, cervical polyps, or abnormal sonographic patterns (e.g., postmenopausal endometrial thickness >5 mm, endometrial hyperechogenic spots). Of these women, 353 were included in the study, and demographic characteristics and data on diabetes, hypertension, and menopausal status were collected. Anthropometric parameters were also analyzed. When a polyp was detected, it was removed via office hysteroscopy, and histologic analysis was performed.
The prevalence of endometrial polyps is associated significantly with age. Other associations, such as hypertension, obesity, and diabetes, exist simply because the prevalence of these pathologies increases with age. Therefore, age is the most significant variable to consider when assessing a patient for endometrial polyps.
Small, asymptomatic uterine polyps may regress without treatment
Lieng M, Istre O, Sandvik L, Qvigstad E. Prevalence, 1-year regression rate, and clinical significance of asymptomatic endometrial polyps: cross-sectional study. J Minim Invasive Gynecol. 2009;16(4):465–471.
The treatment of asymptomatic polyps is controversial because their clinical consequences, malignant potential, and spontaneous regression rate are unknown. In this study, Lieng and colleagues prospectively estimated the prevalence and 1-year regression rate of incidentally diagnosed endometrial polyps in women 40 to 45 years old, as well as bleeding patterns and intensity.
They found polyps in 31 (12.1%) of 257 randomly selected women. At 1 year, the regression rate was 27%.
Details of the trial
At study inception, a standard 10-point visual analog scale was used to quantify each participant’s periodic bleeding, and a physical examination was performed. Transvaginal ultrasonography (US) and saline infusion sonography (SIS) were also performed. When a polyp was detected, researchers measured its length and used Doppler US to visualize the vessel feeding the polyp. An endometrial biopsy was also obtained.
The mean length of polyps was 14 mm (standard deviation [SD], 5.2 mm; 95% confidence interval [CI], 12.1–15.9; median, 13.4 mm; range, 6.7–28.7 mm), and the feeding vessel was identified for 22 of 31 polyps (71%). (For comparison, consider the findings of Clevenger and associates, who reported mean polyp diameters of 13.9 mm and 8.5 mm (P = .064), respectively, among women who had abnormal bleeding and women who did not.1)
When researchers compared women who had polyps with those who had none, they found no significant differences in age, body mass index, blood pressure, gynecologic symptoms, menopausal status, use of hormone therapy, or use of the levonorgestrel-releasing intrauterine device (Mirena). Women who had endometrial polyps scored significantly higher, however, than women who did not on the visual analog scale for periodic bleeding and on the Pictorial Blood Assessment Chart—even when women who had myomas were excluded from the analysis. Although mean hemoglobin levels were similar between groups, women who had polyps had a significantly lower mean ferritin level (25 μg/L vs 41 μg/L; P = .05).
Polyps that persisted were larger from the start
Polyps regressed spontaneously in eight women, six of whom had the feeding vessel visualized at the initial consultation. Polyps that persisted after 12 months were significantly larger (mean polyp length, 15.1 mm; SD, 5.3 mm; 95% CI, 12.7–17.5) at study inception than were those that regressed (mean polyp length, 10.7 mm; SD, 3.9 mm; 95% CI, 7.5–14.0). Polyps that persisted beyond 1 year became significantly longer during follow-up, increasing from a mean length of 15.1 mm to 18.1 mm (SD, 7.9 mm; 95% CI, 0.7–5.3; P = .01).
Twenty of the 22 women who had persistent polyps underwent transcervical resection, one underwent laparoscopic supracervical hysterectomy, and one refused treatment. There were no complications.
Histology revealed that the polyps were benign in 16 women (80%), polypoid in two women (10%), and myomas in two women (10%). No atypical or malignant changes were observed in the polypectomy patients or among all participants.
A small, separate series (three patients) found all polyps to be 5 mm to 8 mm in length at detection, with a regression rate of 100% over several months.5
When an endometrial polyp 10.7 mm in length or shorter is detected incidentally in an asymptomatic, premenopausal woman, it is appropriate to follow it for regression, growth, or the development of symptoms rather than remove it immediately.
What variables signal a greater risk of malignancy?
Baiocchi G, Manci N, Pazzaglia M, et al. Malignancy in endometrial polyps: a 12-year experience. Am J Obstet Gynecol. 2009;201(5):462.e1–e4.
Gregoriou O, Konidaris S, Vrachnis N, et al. Clinical parameters linked with malignancy in endometrial polyps. Climacteric. 2009;12(5):454–458.
Wang JH, Zhao J, Lin J. Opportunities and risk factors for premalignant and malignant transformation of endometrial polyps: management strategies. J Minim Invasive Gynecol. 2010;17(1):53–58.
These three studies explore various aspects of a fundamental challenge: how to discriminate between polyps likely to undergo malignant transformation and those that will not.
The answer: Look for menopausal status, abnormal uterine bleeding, diabetes, obesity, and hypertension. Polyps larger than 1 cm also appear more likely to become malignant.
Details of the trials
In a retrospective study involving 1,242 women who had endometrial polyps, Baiocchi and colleagues identified 95.2% of the polyps as benign, 1.3% as premalignant, and 3.5% as malignant. Four clinical variables were significantly associated with premalignant and malignant features:
- age
- menopausal status
- abnormal uterine bleeding
- hypertension.
In their series of 516 cases, Gregoriou and associates found 96.9% of polyps to be benign, 1.2% to be premalignant, and 1.9% to be malignant. Four variables were associated with premalignant and malignant features:
- age above 60 years
- menopausal status
- obesity
- diabetes.
And in a study involving 766 patients, Wang and colleagues found 96.2% of polyps to be benign, 3.26% to involve hyperplasia with atypia, and 0.52% to be malignant. Among the variables associated with premalignant and malignant polyps were:
- polyp diameter larger than 1 cm
- menopausal status
- abnormal uterine bleeding.
When endometrial polyps are identified, the following characteristics indicate an increased risk of malignancy: age above 60 years, menopausal status, abnormal uterine bleeding, obesity, hypertension, and diabetes. Polyps larger than 1 cm are also more likely to be premalignant or malignant in nature. When any of these conditions is present, polypectomy and histology are recommended.
When a patient complains of abnormal uterine bleeding, evaluation often begins with transvaginal ultrasonography (US). Among the challenges of assessing the endometrium using US is the unreliability of endometrial thickness as a predictor of pathology. For example, Breitkopf and colleagues found that transvaginal US missed intracavitary lesions in one of six premenopausal women who had abnormal bleeding and an endometrial stripe thinner than 5 mm, for a sensitivity of 74%.6
In a separate study, Marello and colleagues used the combination of hysteroscopy and directed biopsy—the gold standard of diagnosis—to evaluate 212 postmenopausal women who had an endometrial thickness of 4 mm or less.7 (This parameter has been suggested as a cutoff for symptomatic postmenopausal women.8) Of these 212 women, 10% were found to have histologically confirmed intracavitary pathology (16 polyps and 4 submucous myomas).7 Among 13 symptomatic women in this study, three (23%) were found to have an endometrial polyp.7
These studies suggest that endometrial thickness alone should not be used to exclude benign endometrial pathology in symptomatic women, be they premenopausal or postmenopausal. No data back routine US to measure endometrial thickness in asymptomatic postmenopausal women.
Hysteroscopy and SIS are preferred
Both hysteroscopy and saline infusion sonography (SIS) have significantly better sensitivity and specificity in the diagnosis of intracavitary pathology than transvaginal US alone in women who have abnormal bleeding (FIGURE 1; VIDEOS 1, 2, AND 3 related to this article in the OBG Management Video Library at obgmanagement.com).1,9 Hysteroscopy and SIS detect polyps with equal accuracy.10 However, hysteroscopy allows for removal of endometrial polyps and directed biopsy at the time of diagnosis.9
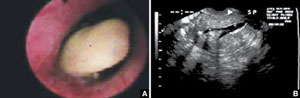
FIGURE 1 Imaging of polyps: go beyond transvaginal ultrasonography for optimal visualization
A. Hysteroscopic view of an endometrial polyp. B. The view with saline-infusion sonography.
In symptomatic women, resect the polyp
Polypectomy improves abnormal bleeding, according to a systematic review by Nathani and associates.11 All studies included in the review, which involved follow-up intervals between 2 and 52 months, reported such an improvement.11
When it is performed in the office, polypectomy offers several advantages over its inpatient counterpart:
- higher cost-effectiveness
- greater convenience
- avoidance of general anesthesia.
In both settings, it can be performed using mechanical or bipolar electrosurgical instrumentation (VIDEO 4).12
Segmental resection of the polyp while it is partially attached to the uterine wall is the optimal removal technique for large polyps (FIGURE 2). A grasping forceps can then be used to remove the polyp completely (VIDEO 5). Instruments such as a basket and snare are helpful in removing the polyp effectively.13
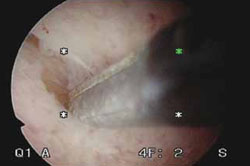
FIGURE 2 segmental resection of a polyp
During hysteroscopic polypectomy, the polyp is resected in segments while it is still partially attached to the endometrial wall.
Is histologic analysis of a polyp sufficient risk assessment?
Rahimi S, Marani C, Renzi C, Natale ME, Giovannini P, Zeloni R. Endometrial polyps and the risk of atypical hyperplasia on biopsies of unremarkable endometrium: a study on 694 patients with benign endometrial polyps. Int J Gynecol Pathol. 2009;28(6):522–528.
This study involved 694 consecutive patients who had benign endometrial polyps. Investigators sought to clarify the relationship between polyps and the underlying endometrium—specifically, to determine whether a polyp is a “circumscribed pathology of the endometrium or a polypoid expression of endometrial hyperplasia.” In describing the rationale for the study, the authors observe that the association between polyps and premalignant and malignant changes remains unclear.
Participants underwent hysteroscopy for removal of the polyps, at which time two biopsies of “unremarkable” endometrium, far from the base of the polyp, were also obtained.
Overall, endometrial hyperplasia without atypia was identified on hysteroscopically unremarkable endometrium in 18% of women, and atypia was identified in 7.3%. Among postmenopausal women, hyperplasia without atypia was identified in 21.6% of cases, atypia in 12%, and adenocarcinoma in 1.2%.
Multivariable analysis revealed that postmenopausal women who had polyps heavier than 1 g were 3.6 times more likely to have atypia (95% CI, 1.3–10.3). Among premenopausal women, the likelihood of atypia increased when the polyp weighed more than 0.4 g (odds ratio [OR], 3.5; 95% CI, 1.1–10.9) or the patient was older than 40 years (OR, 3.82; 95% CI, 1.1–13.2).
Endometrial lesions are not always evident at the time of hysteroscopy. Therefore, when evaluating an endometrial lesion such as a polyp, combine hysteroscopy with histopathologic assessment of the background endometrium (by means of a pipelle or curette), especially in women who have high-risk characteristics such as menopausal status or large polyps.
They’re accessible in the OBG Management Video Library at obgmanagement.com
VIDEO 1: Saline-infusion sonographic imaging of a polyp
VIDEO 2: Hysteroscopic imaging of a polyp in a menopausal patient. A flexible 3-mm scope is used to assess an asymptomatic menopausal woman in whom an enlarged endometrial stripe was identified during earlier imaging.
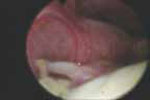
VIDEO 3: Hysteroscopic imaging of a polyp and associated hyperplasia. A flexible 3-mm scope is used to evaluate a menopausal women who has uterine bleeding, revealing a 2-cm polyp.
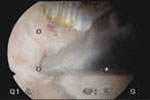
VIDEO 4: Office polypectomy. An 8-mm, apparently benign polyp is removed from a premenopausal woman using continuous-flow operative hysteroscopy in an office setting.
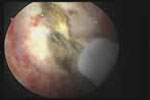
VIDEO 5: Removal of a large polyp. A large (2 cm x 2.5 cm) polyp is removed in pieces, with the polyp partially attached to the endometrial wall, from a premenopausal woman who has abnormal bleeding.
More: Watch Dr. Brent Seibel perform OR-based hysteroscopic polypectomy
1. Clevenger-Hoeft M, Syrop C, Stovall D, Van Voorhis B. Sonohysterography in premenopausal women with and without abnormal bleeding. Obstet Gynecol. 1999;94(4):516-520.
2. DeWaay DJ, Syrop CH, Nygaard IE, Davis WA, Van Voorhis BJ. Natural history of uterine polyps and leiomyomata. Obstet Gynecol. 2002;100(1):3-7.
3. Lieng M, Istre O, Sandvik L, Qvigstad E. Prevalence, 1-year regression rate, and clinical significance of asymptomatic endometrial polyps: cross-sectional study. J Minim Invasive Gynecol. 2009;16(4):465-471.
4. Lopes RG, Baracat EC, de Albuquerque Neto LC, et al. Analysis of estrogen- and progersterone-receptor expression in endometrial polyps. J Minim Invasive Gynecol. 2007;14(3):300-303.
5. Haimov-Kochman R, Deri-Hasid R, Hamani Y, Voss E. The natural course of endometrial polyps: could they vanish when left untreated? Fertil Steril. 2009;92(2):828.e11-e12.
6. Breitkopf DM, Frederickson RA, Snyder RR. Detection of benign endometrial masses by endometrial stripe measurement in premenopausal women. Obstet Gynecol. 2004;104(1):120-125.
7. Marello F, Bettocchi S, Greco P, et al. Hysteroscopic evaluation of menopausal patients with sonographically atrophic endometrium. J Am Gynecol Laparosc. 2000;7(2):197-200.
8. Karlsson B, Granberg S, Wikland M, Ylostalo P, Torvid K, Marsal K, Valentin L.
Transvaginal ultrasonography of the endometrium in women with postmenopausal bleeding—a Nordic multicenter study. Am J Obstet Gynecol. 1995;172(5):1488-1494.
9. Paschopoulos M, Lolis E, Alamanos Y, Koliopoulos G, Paraskevaidis E. Vaginoscopic hysteroscopy and transvaginal sonography in the evaluation of patients with abnormal uterine bleeding. J Am Assoc Gynecol Laparosc. 2001;8(4):506-510.
10. Jansen FW, de Kroon CD, van Dongen H, Grooters C, Louwé L, Trimbos-Kemper T. Diagnostic hysteroscopy and saline infusion sonography: prediction of intrauterine polyps and myomas. J Minim Invasive Gynecol. 2006;13(4):320-324.
11. Nathani F, Clark TJ. Uterine polypectomy in the management of abnormal uterine bleeding: a systematic review. J Minim Invasive Gynecol. 2006;13(4):260-268.
12. Garuti G, Centinaio G, Luerti M. Outpatient hysteroscopic polypectomy in postmenopausal women: a comparison between mechanical and electrosurgical resection. J Minim Invasive Gynecol. 2008;15(5):595-600.
13. Timmermans A, Veersema S. Ambulatory transcervical resection of polyps with the Duckbill polyp snare: a modality for treatment of endometrial polyps. J Minim Invasive Gynecol. 2005;12(1):37-39.
Endometrial polyps are a relatively common pathology, occurring in 24% to 41% of women who have abnormal bleeding, and in about 10% of asymptomatic women.1,2 Endometrial polyps may be associated with leiomyomas in women who have abnormal bleeding.1-3
Polyps originate as focal hyperplasia of basal endometrium and contain variable amounts of glands, stroma, and blood vessels. Glandular epithelium has higher estrogen- and progesterone-receptor expression than surrounding endometrium, whereas the stromal component of a polyp has hormone receptors similar to endometrium. This suggests that a polyp represents focal hyperplasia that is more glandular than stromal.4
In this Update, I outline the basics of diagnosis and treatment and report on several recent investigations:
- a retrospective analysis from Italy that found that endometrial polyps are associated with advancing age and that any apparent association between polyps and diabetes, hypertension, or obesity is likely age-related
- a cross-sectional study from Norway that found that some asymptomatic polyps regress spontaneously, usually when their length is 10.7 mm or less
- three studies that explore the variables associated with premalignant and malignant polyps
- an investigation of the relationship between endometrial polyps and the background endometrium that found atypical hyperplasia in endometrium remote from the polyp in a significant percentage of women.
Age is the most important variable when assessing a patient for endometrial pathology
Nappi L, Indraccolo U, Di Spiezio Sardo A, et al. Are diabetes, hypertension, and obesity independent risk factors for endometrial polyps? J Minim Invasive Gynecol. 2009;16(2):157–162.
In this retrospective analysis of 353 women who underwent office hysteroscopy, Nappi and co-workers set out to ascertain whether endometrial polyps are associated with diabetes, hypertension, or obesity, independent of age and menopausal status. They did find an association between age, menopause, hypertension, obesity, and the presence of endometrial polyps. However, after multivariable logistic regression, all variables except age lost statistical significance. The median age at which polyps were present was 53 years (range: 29–86 years).
Details of the trial
A total of 394 consecutive Caucasian women underwent hysteroscopy to assess abnormal uterine bleeding, infertility, cervical polyps, or abnormal sonographic patterns (e.g., postmenopausal endometrial thickness >5 mm, endometrial hyperechogenic spots). Of these women, 353 were included in the study, and demographic characteristics and data on diabetes, hypertension, and menopausal status were collected. Anthropometric parameters were also analyzed. When a polyp was detected, it was removed via office hysteroscopy, and histologic analysis was performed.
The prevalence of endometrial polyps is associated significantly with age. Other associations, such as hypertension, obesity, and diabetes, exist simply because the prevalence of these pathologies increases with age. Therefore, age is the most significant variable to consider when assessing a patient for endometrial polyps.
Small, asymptomatic uterine polyps may regress without treatment
Lieng M, Istre O, Sandvik L, Qvigstad E. Prevalence, 1-year regression rate, and clinical significance of asymptomatic endometrial polyps: cross-sectional study. J Minim Invasive Gynecol. 2009;16(4):465–471.
The treatment of asymptomatic polyps is controversial because their clinical consequences, malignant potential, and spontaneous regression rate are unknown. In this study, Lieng and colleagues prospectively estimated the prevalence and 1-year regression rate of incidentally diagnosed endometrial polyps in women 40 to 45 years old, as well as bleeding patterns and intensity.
They found polyps in 31 (12.1%) of 257 randomly selected women. At 1 year, the regression rate was 27%.
Details of the trial
At study inception, a standard 10-point visual analog scale was used to quantify each participant’s periodic bleeding, and a physical examination was performed. Transvaginal ultrasonography (US) and saline infusion sonography (SIS) were also performed. When a polyp was detected, researchers measured its length and used Doppler US to visualize the vessel feeding the polyp. An endometrial biopsy was also obtained.
The mean length of polyps was 14 mm (standard deviation [SD], 5.2 mm; 95% confidence interval [CI], 12.1–15.9; median, 13.4 mm; range, 6.7–28.7 mm), and the feeding vessel was identified for 22 of 31 polyps (71%). (For comparison, consider the findings of Clevenger and associates, who reported mean polyp diameters of 13.9 mm and 8.5 mm (P = .064), respectively, among women who had abnormal bleeding and women who did not.1)
When researchers compared women who had polyps with those who had none, they found no significant differences in age, body mass index, blood pressure, gynecologic symptoms, menopausal status, use of hormone therapy, or use of the levonorgestrel-releasing intrauterine device (Mirena). Women who had endometrial polyps scored significantly higher, however, than women who did not on the visual analog scale for periodic bleeding and on the Pictorial Blood Assessment Chart—even when women who had myomas were excluded from the analysis. Although mean hemoglobin levels were similar between groups, women who had polyps had a significantly lower mean ferritin level (25 μg/L vs 41 μg/L; P = .05).
Polyps that persisted were larger from the start
Polyps regressed spontaneously in eight women, six of whom had the feeding vessel visualized at the initial consultation. Polyps that persisted after 12 months were significantly larger (mean polyp length, 15.1 mm; SD, 5.3 mm; 95% CI, 12.7–17.5) at study inception than were those that regressed (mean polyp length, 10.7 mm; SD, 3.9 mm; 95% CI, 7.5–14.0). Polyps that persisted beyond 1 year became significantly longer during follow-up, increasing from a mean length of 15.1 mm to 18.1 mm (SD, 7.9 mm; 95% CI, 0.7–5.3; P = .01).
Twenty of the 22 women who had persistent polyps underwent transcervical resection, one underwent laparoscopic supracervical hysterectomy, and one refused treatment. There were no complications.
Histology revealed that the polyps were benign in 16 women (80%), polypoid in two women (10%), and myomas in two women (10%). No atypical or malignant changes were observed in the polypectomy patients or among all participants.
A small, separate series (three patients) found all polyps to be 5 mm to 8 mm in length at detection, with a regression rate of 100% over several months.5
When an endometrial polyp 10.7 mm in length or shorter is detected incidentally in an asymptomatic, premenopausal woman, it is appropriate to follow it for regression, growth, or the development of symptoms rather than remove it immediately.
What variables signal a greater risk of malignancy?
Baiocchi G, Manci N, Pazzaglia M, et al. Malignancy in endometrial polyps: a 12-year experience. Am J Obstet Gynecol. 2009;201(5):462.e1–e4.
Gregoriou O, Konidaris S, Vrachnis N, et al. Clinical parameters linked with malignancy in endometrial polyps. Climacteric. 2009;12(5):454–458.
Wang JH, Zhao J, Lin J. Opportunities and risk factors for premalignant and malignant transformation of endometrial polyps: management strategies. J Minim Invasive Gynecol. 2010;17(1):53–58.
These three studies explore various aspects of a fundamental challenge: how to discriminate between polyps likely to undergo malignant transformation and those that will not.
The answer: Look for menopausal status, abnormal uterine bleeding, diabetes, obesity, and hypertension. Polyps larger than 1 cm also appear more likely to become malignant.
Details of the trials
In a retrospective study involving 1,242 women who had endometrial polyps, Baiocchi and colleagues identified 95.2% of the polyps as benign, 1.3% as premalignant, and 3.5% as malignant. Four clinical variables were significantly associated with premalignant and malignant features:
- age
- menopausal status
- abnormal uterine bleeding
- hypertension.
In their series of 516 cases, Gregoriou and associates found 96.9% of polyps to be benign, 1.2% to be premalignant, and 1.9% to be malignant. Four variables were associated with premalignant and malignant features:
- age above 60 years
- menopausal status
- obesity
- diabetes.
And in a study involving 766 patients, Wang and colleagues found 96.2% of polyps to be benign, 3.26% to involve hyperplasia with atypia, and 0.52% to be malignant. Among the variables associated with premalignant and malignant polyps were:
- polyp diameter larger than 1 cm
- menopausal status
- abnormal uterine bleeding.
When endometrial polyps are identified, the following characteristics indicate an increased risk of malignancy: age above 60 years, menopausal status, abnormal uterine bleeding, obesity, hypertension, and diabetes. Polyps larger than 1 cm are also more likely to be premalignant or malignant in nature. When any of these conditions is present, polypectomy and histology are recommended.
When a patient complains of abnormal uterine bleeding, evaluation often begins with transvaginal ultrasonography (US). Among the challenges of assessing the endometrium using US is the unreliability of endometrial thickness as a predictor of pathology. For example, Breitkopf and colleagues found that transvaginal US missed intracavitary lesions in one of six premenopausal women who had abnormal bleeding and an endometrial stripe thinner than 5 mm, for a sensitivity of 74%.6
In a separate study, Marello and colleagues used the combination of hysteroscopy and directed biopsy—the gold standard of diagnosis—to evaluate 212 postmenopausal women who had an endometrial thickness of 4 mm or less.7 (This parameter has been suggested as a cutoff for symptomatic postmenopausal women.8) Of these 212 women, 10% were found to have histologically confirmed intracavitary pathology (16 polyps and 4 submucous myomas).7 Among 13 symptomatic women in this study, three (23%) were found to have an endometrial polyp.7
These studies suggest that endometrial thickness alone should not be used to exclude benign endometrial pathology in symptomatic women, be they premenopausal or postmenopausal. No data back routine US to measure endometrial thickness in asymptomatic postmenopausal women.
Hysteroscopy and SIS are preferred
Both hysteroscopy and saline infusion sonography (SIS) have significantly better sensitivity and specificity in the diagnosis of intracavitary pathology than transvaginal US alone in women who have abnormal bleeding (FIGURE 1; VIDEOS 1, 2, AND 3 related to this article in the OBG Management Video Library at obgmanagement.com).1,9 Hysteroscopy and SIS detect polyps with equal accuracy.10 However, hysteroscopy allows for removal of endometrial polyps and directed biopsy at the time of diagnosis.9

FIGURE 1 Imaging of polyps: go beyond transvaginal ultrasonography for optimal visualization
A. Hysteroscopic view of an endometrial polyp. B. The view with saline-infusion sonography.
In symptomatic women, resect the polyp
Polypectomy improves abnormal bleeding, according to a systematic review by Nathani and associates.11 All studies included in the review, which involved follow-up intervals between 2 and 52 months, reported such an improvement.11
When it is performed in the office, polypectomy offers several advantages over its inpatient counterpart:
- higher cost-effectiveness
- greater convenience
- avoidance of general anesthesia.
In both settings, it can be performed using mechanical or bipolar electrosurgical instrumentation (VIDEO 4).12
Segmental resection of the polyp while it is partially attached to the uterine wall is the optimal removal technique for large polyps (FIGURE 2). A grasping forceps can then be used to remove the polyp completely (VIDEO 5). Instruments such as a basket and snare are helpful in removing the polyp effectively.13

FIGURE 2 segmental resection of a polyp
During hysteroscopic polypectomy, the polyp is resected in segments while it is still partially attached to the endometrial wall.
Is histologic analysis of a polyp sufficient risk assessment?
Rahimi S, Marani C, Renzi C, Natale ME, Giovannini P, Zeloni R. Endometrial polyps and the risk of atypical hyperplasia on biopsies of unremarkable endometrium: a study on 694 patients with benign endometrial polyps. Int J Gynecol Pathol. 2009;28(6):522–528.
This study involved 694 consecutive patients who had benign endometrial polyps. Investigators sought to clarify the relationship between polyps and the underlying endometrium—specifically, to determine whether a polyp is a “circumscribed pathology of the endometrium or a polypoid expression of endometrial hyperplasia.” In describing the rationale for the study, the authors observe that the association between polyps and premalignant and malignant changes remains unclear.
Participants underwent hysteroscopy for removal of the polyps, at which time two biopsies of “unremarkable” endometrium, far from the base of the polyp, were also obtained.
Overall, endometrial hyperplasia without atypia was identified on hysteroscopically unremarkable endometrium in 18% of women, and atypia was identified in 7.3%. Among postmenopausal women, hyperplasia without atypia was identified in 21.6% of cases, atypia in 12%, and adenocarcinoma in 1.2%.
Multivariable analysis revealed that postmenopausal women who had polyps heavier than 1 g were 3.6 times more likely to have atypia (95% CI, 1.3–10.3). Among premenopausal women, the likelihood of atypia increased when the polyp weighed more than 0.4 g (odds ratio [OR], 3.5; 95% CI, 1.1–10.9) or the patient was older than 40 years (OR, 3.82; 95% CI, 1.1–13.2).
Endometrial lesions are not always evident at the time of hysteroscopy. Therefore, when evaluating an endometrial lesion such as a polyp, combine hysteroscopy with histopathologic assessment of the background endometrium (by means of a pipelle or curette), especially in women who have high-risk characteristics such as menopausal status or large polyps.
They’re accessible in the OBG Management Video Library at obgmanagement.com

VIDEO 1: Saline-infusion sonographic imaging of a polyp

VIDEO 2: Hysteroscopic imaging of a polyp in a menopausal patient. A flexible 3-mm scope is used to assess an asymptomatic menopausal woman in whom an enlarged endometrial stripe was identified during earlier imaging.

VIDEO 3: Hysteroscopic imaging of a polyp and associated hyperplasia. A flexible 3-mm scope is used to evaluate a menopausal women who has uterine bleeding, revealing a 2-cm polyp.

VIDEO 4: Office polypectomy. An 8-mm, apparently benign polyp is removed from a premenopausal woman using continuous-flow operative hysteroscopy in an office setting.

VIDEO 5: Removal of a large polyp. A large (2 cm x 2.5 cm) polyp is removed in pieces, with the polyp partially attached to the endometrial wall, from a premenopausal woman who has abnormal bleeding.
More: Watch Dr. Brent Seibel perform OR-based hysteroscopic polypectomy
Endometrial polyps are a relatively common pathology, occurring in 24% to 41% of women who have abnormal bleeding, and in about 10% of asymptomatic women.1,2 Endometrial polyps may be associated with leiomyomas in women who have abnormal bleeding.1-3
Polyps originate as focal hyperplasia of basal endometrium and contain variable amounts of glands, stroma, and blood vessels. Glandular epithelium has higher estrogen- and progesterone-receptor expression than surrounding endometrium, whereas the stromal component of a polyp has hormone receptors similar to endometrium. This suggests that a polyp represents focal hyperplasia that is more glandular than stromal.4
In this Update, I outline the basics of diagnosis and treatment and report on several recent investigations:
- a retrospective analysis from Italy that found that endometrial polyps are associated with advancing age and that any apparent association between polyps and diabetes, hypertension, or obesity is likely age-related
- a cross-sectional study from Norway that found that some asymptomatic polyps regress spontaneously, usually when their length is 10.7 mm or less
- three studies that explore the variables associated with premalignant and malignant polyps
- an investigation of the relationship between endometrial polyps and the background endometrium that found atypical hyperplasia in endometrium remote from the polyp in a significant percentage of women.
Age is the most important variable when assessing a patient for endometrial pathology
Nappi L, Indraccolo U, Di Spiezio Sardo A, et al. Are diabetes, hypertension, and obesity independent risk factors for endometrial polyps? J Minim Invasive Gynecol. 2009;16(2):157–162.
In this retrospective analysis of 353 women who underwent office hysteroscopy, Nappi and co-workers set out to ascertain whether endometrial polyps are associated with diabetes, hypertension, or obesity, independent of age and menopausal status. They did find an association between age, menopause, hypertension, obesity, and the presence of endometrial polyps. However, after multivariable logistic regression, all variables except age lost statistical significance. The median age at which polyps were present was 53 years (range: 29–86 years).
Details of the trial
A total of 394 consecutive Caucasian women underwent hysteroscopy to assess abnormal uterine bleeding, infertility, cervical polyps, or abnormal sonographic patterns (e.g., postmenopausal endometrial thickness >5 mm, endometrial hyperechogenic spots). Of these women, 353 were included in the study, and demographic characteristics and data on diabetes, hypertension, and menopausal status were collected. Anthropometric parameters were also analyzed. When a polyp was detected, it was removed via office hysteroscopy, and histologic analysis was performed.
The prevalence of endometrial polyps is associated significantly with age. Other associations, such as hypertension, obesity, and diabetes, exist simply because the prevalence of these pathologies increases with age. Therefore, age is the most significant variable to consider when assessing a patient for endometrial polyps.
Small, asymptomatic uterine polyps may regress without treatment
Lieng M, Istre O, Sandvik L, Qvigstad E. Prevalence, 1-year regression rate, and clinical significance of asymptomatic endometrial polyps: cross-sectional study. J Minim Invasive Gynecol. 2009;16(4):465–471.
The treatment of asymptomatic polyps is controversial because their clinical consequences, malignant potential, and spontaneous regression rate are unknown. In this study, Lieng and colleagues prospectively estimated the prevalence and 1-year regression rate of incidentally diagnosed endometrial polyps in women 40 to 45 years old, as well as bleeding patterns and intensity.
They found polyps in 31 (12.1%) of 257 randomly selected women. At 1 year, the regression rate was 27%.
Details of the trial
At study inception, a standard 10-point visual analog scale was used to quantify each participant’s periodic bleeding, and a physical examination was performed. Transvaginal ultrasonography (US) and saline infusion sonography (SIS) were also performed. When a polyp was detected, researchers measured its length and used Doppler US to visualize the vessel feeding the polyp. An endometrial biopsy was also obtained.
The mean length of polyps was 14 mm (standard deviation [SD], 5.2 mm; 95% confidence interval [CI], 12.1–15.9; median, 13.4 mm; range, 6.7–28.7 mm), and the feeding vessel was identified for 22 of 31 polyps (71%). (For comparison, consider the findings of Clevenger and associates, who reported mean polyp diameters of 13.9 mm and 8.5 mm (P = .064), respectively, among women who had abnormal bleeding and women who did not.1)
When researchers compared women who had polyps with those who had none, they found no significant differences in age, body mass index, blood pressure, gynecologic symptoms, menopausal status, use of hormone therapy, or use of the levonorgestrel-releasing intrauterine device (Mirena). Women who had endometrial polyps scored significantly higher, however, than women who did not on the visual analog scale for periodic bleeding and on the Pictorial Blood Assessment Chart—even when women who had myomas were excluded from the analysis. Although mean hemoglobin levels were similar between groups, women who had polyps had a significantly lower mean ferritin level (25 μg/L vs 41 μg/L; P = .05).
Polyps that persisted were larger from the start
Polyps regressed spontaneously in eight women, six of whom had the feeding vessel visualized at the initial consultation. Polyps that persisted after 12 months were significantly larger (mean polyp length, 15.1 mm; SD, 5.3 mm; 95% CI, 12.7–17.5) at study inception than were those that regressed (mean polyp length, 10.7 mm; SD, 3.9 mm; 95% CI, 7.5–14.0). Polyps that persisted beyond 1 year became significantly longer during follow-up, increasing from a mean length of 15.1 mm to 18.1 mm (SD, 7.9 mm; 95% CI, 0.7–5.3; P = .01).
Twenty of the 22 women who had persistent polyps underwent transcervical resection, one underwent laparoscopic supracervical hysterectomy, and one refused treatment. There were no complications.
Histology revealed that the polyps were benign in 16 women (80%), polypoid in two women (10%), and myomas in two women (10%). No atypical or malignant changes were observed in the polypectomy patients or among all participants.
A small, separate series (three patients) found all polyps to be 5 mm to 8 mm in length at detection, with a regression rate of 100% over several months.5
When an endometrial polyp 10.7 mm in length or shorter is detected incidentally in an asymptomatic, premenopausal woman, it is appropriate to follow it for regression, growth, or the development of symptoms rather than remove it immediately.
What variables signal a greater risk of malignancy?
Baiocchi G, Manci N, Pazzaglia M, et al. Malignancy in endometrial polyps: a 12-year experience. Am J Obstet Gynecol. 2009;201(5):462.e1–e4.
Gregoriou O, Konidaris S, Vrachnis N, et al. Clinical parameters linked with malignancy in endometrial polyps. Climacteric. 2009;12(5):454–458.
Wang JH, Zhao J, Lin J. Opportunities and risk factors for premalignant and malignant transformation of endometrial polyps: management strategies. J Minim Invasive Gynecol. 2010;17(1):53–58.
These three studies explore various aspects of a fundamental challenge: how to discriminate between polyps likely to undergo malignant transformation and those that will not.
The answer: Look for menopausal status, abnormal uterine bleeding, diabetes, obesity, and hypertension. Polyps larger than 1 cm also appear more likely to become malignant.
Details of the trials
In a retrospective study involving 1,242 women who had endometrial polyps, Baiocchi and colleagues identified 95.2% of the polyps as benign, 1.3% as premalignant, and 3.5% as malignant. Four clinical variables were significantly associated with premalignant and malignant features:
- age
- menopausal status
- abnormal uterine bleeding
- hypertension.
In their series of 516 cases, Gregoriou and associates found 96.9% of polyps to be benign, 1.2% to be premalignant, and 1.9% to be malignant. Four variables were associated with premalignant and malignant features:
- age above 60 years
- menopausal status
- obesity
- diabetes.
And in a study involving 766 patients, Wang and colleagues found 96.2% of polyps to be benign, 3.26% to involve hyperplasia with atypia, and 0.52% to be malignant. Among the variables associated with premalignant and malignant polyps were:
- polyp diameter larger than 1 cm
- menopausal status
- abnormal uterine bleeding.
When endometrial polyps are identified, the following characteristics indicate an increased risk of malignancy: age above 60 years, menopausal status, abnormal uterine bleeding, obesity, hypertension, and diabetes. Polyps larger than 1 cm are also more likely to be premalignant or malignant in nature. When any of these conditions is present, polypectomy and histology are recommended.
When a patient complains of abnormal uterine bleeding, evaluation often begins with transvaginal ultrasonography (US). Among the challenges of assessing the endometrium using US is the unreliability of endometrial thickness as a predictor of pathology. For example, Breitkopf and colleagues found that transvaginal US missed intracavitary lesions in one of six premenopausal women who had abnormal bleeding and an endometrial stripe thinner than 5 mm, for a sensitivity of 74%.6
In a separate study, Marello and colleagues used the combination of hysteroscopy and directed biopsy—the gold standard of diagnosis—to evaluate 212 postmenopausal women who had an endometrial thickness of 4 mm or less.7 (This parameter has been suggested as a cutoff for symptomatic postmenopausal women.8) Of these 212 women, 10% were found to have histologically confirmed intracavitary pathology (16 polyps and 4 submucous myomas).7 Among 13 symptomatic women in this study, three (23%) were found to have an endometrial polyp.7
These studies suggest that endometrial thickness alone should not be used to exclude benign endometrial pathology in symptomatic women, be they premenopausal or postmenopausal. No data back routine US to measure endometrial thickness in asymptomatic postmenopausal women.
Hysteroscopy and SIS are preferred
Both hysteroscopy and saline infusion sonography (SIS) have significantly better sensitivity and specificity in the diagnosis of intracavitary pathology than transvaginal US alone in women who have abnormal bleeding (FIGURE 1; VIDEOS 1, 2, AND 3 related to this article in the OBG Management Video Library at obgmanagement.com).1,9 Hysteroscopy and SIS detect polyps with equal accuracy.10 However, hysteroscopy allows for removal of endometrial polyps and directed biopsy at the time of diagnosis.9

FIGURE 1 Imaging of polyps: go beyond transvaginal ultrasonography for optimal visualization
A. Hysteroscopic view of an endometrial polyp. B. The view with saline-infusion sonography.
In symptomatic women, resect the polyp
Polypectomy improves abnormal bleeding, according to a systematic review by Nathani and associates.11 All studies included in the review, which involved follow-up intervals between 2 and 52 months, reported such an improvement.11
When it is performed in the office, polypectomy offers several advantages over its inpatient counterpart:
- higher cost-effectiveness
- greater convenience
- avoidance of general anesthesia.
In both settings, it can be performed using mechanical or bipolar electrosurgical instrumentation (VIDEO 4).12
Segmental resection of the polyp while it is partially attached to the uterine wall is the optimal removal technique for large polyps (FIGURE 2). A grasping forceps can then be used to remove the polyp completely (VIDEO 5). Instruments such as a basket and snare are helpful in removing the polyp effectively.13

FIGURE 2 segmental resection of a polyp
During hysteroscopic polypectomy, the polyp is resected in segments while it is still partially attached to the endometrial wall.
Is histologic analysis of a polyp sufficient risk assessment?
Rahimi S, Marani C, Renzi C, Natale ME, Giovannini P, Zeloni R. Endometrial polyps and the risk of atypical hyperplasia on biopsies of unremarkable endometrium: a study on 694 patients with benign endometrial polyps. Int J Gynecol Pathol. 2009;28(6):522–528.
This study involved 694 consecutive patients who had benign endometrial polyps. Investigators sought to clarify the relationship between polyps and the underlying endometrium—specifically, to determine whether a polyp is a “circumscribed pathology of the endometrium or a polypoid expression of endometrial hyperplasia.” In describing the rationale for the study, the authors observe that the association between polyps and premalignant and malignant changes remains unclear.
Participants underwent hysteroscopy for removal of the polyps, at which time two biopsies of “unremarkable” endometrium, far from the base of the polyp, were also obtained.
Overall, endometrial hyperplasia without atypia was identified on hysteroscopically unremarkable endometrium in 18% of women, and atypia was identified in 7.3%. Among postmenopausal women, hyperplasia without atypia was identified in 21.6% of cases, atypia in 12%, and adenocarcinoma in 1.2%.
Multivariable analysis revealed that postmenopausal women who had polyps heavier than 1 g were 3.6 times more likely to have atypia (95% CI, 1.3–10.3). Among premenopausal women, the likelihood of atypia increased when the polyp weighed more than 0.4 g (odds ratio [OR], 3.5; 95% CI, 1.1–10.9) or the patient was older than 40 years (OR, 3.82; 95% CI, 1.1–13.2).
Endometrial lesions are not always evident at the time of hysteroscopy. Therefore, when evaluating an endometrial lesion such as a polyp, combine hysteroscopy with histopathologic assessment of the background endometrium (by means of a pipelle or curette), especially in women who have high-risk characteristics such as menopausal status or large polyps.
They’re accessible in the OBG Management Video Library at obgmanagement.com

VIDEO 1: Saline-infusion sonographic imaging of a polyp

VIDEO 2: Hysteroscopic imaging of a polyp in a menopausal patient. A flexible 3-mm scope is used to assess an asymptomatic menopausal woman in whom an enlarged endometrial stripe was identified during earlier imaging.

VIDEO 3: Hysteroscopic imaging of a polyp and associated hyperplasia. A flexible 3-mm scope is used to evaluate a menopausal women who has uterine bleeding, revealing a 2-cm polyp.

VIDEO 4: Office polypectomy. An 8-mm, apparently benign polyp is removed from a premenopausal woman using continuous-flow operative hysteroscopy in an office setting.

VIDEO 5: Removal of a large polyp. A large (2 cm x 2.5 cm) polyp is removed in pieces, with the polyp partially attached to the endometrial wall, from a premenopausal woman who has abnormal bleeding.
More: Watch Dr. Brent Seibel perform OR-based hysteroscopic polypectomy
1. Clevenger-Hoeft M, Syrop C, Stovall D, Van Voorhis B. Sonohysterography in premenopausal women with and without abnormal bleeding. Obstet Gynecol. 1999;94(4):516-520.
2. DeWaay DJ, Syrop CH, Nygaard IE, Davis WA, Van Voorhis BJ. Natural history of uterine polyps and leiomyomata. Obstet Gynecol. 2002;100(1):3-7.
3. Lieng M, Istre O, Sandvik L, Qvigstad E. Prevalence, 1-year regression rate, and clinical significance of asymptomatic endometrial polyps: cross-sectional study. J Minim Invasive Gynecol. 2009;16(4):465-471.
4. Lopes RG, Baracat EC, de Albuquerque Neto LC, et al. Analysis of estrogen- and progersterone-receptor expression in endometrial polyps. J Minim Invasive Gynecol. 2007;14(3):300-303.
5. Haimov-Kochman R, Deri-Hasid R, Hamani Y, Voss E. The natural course of endometrial polyps: could they vanish when left untreated? Fertil Steril. 2009;92(2):828.e11-e12.
6. Breitkopf DM, Frederickson RA, Snyder RR. Detection of benign endometrial masses by endometrial stripe measurement in premenopausal women. Obstet Gynecol. 2004;104(1):120-125.
7. Marello F, Bettocchi S, Greco P, et al. Hysteroscopic evaluation of menopausal patients with sonographically atrophic endometrium. J Am Gynecol Laparosc. 2000;7(2):197-200.
8. Karlsson B, Granberg S, Wikland M, Ylostalo P, Torvid K, Marsal K, Valentin L.
Transvaginal ultrasonography of the endometrium in women with postmenopausal bleeding—a Nordic multicenter study. Am J Obstet Gynecol. 1995;172(5):1488-1494.
9. Paschopoulos M, Lolis E, Alamanos Y, Koliopoulos G, Paraskevaidis E. Vaginoscopic hysteroscopy and transvaginal sonography in the evaluation of patients with abnormal uterine bleeding. J Am Assoc Gynecol Laparosc. 2001;8(4):506-510.
10. Jansen FW, de Kroon CD, van Dongen H, Grooters C, Louwé L, Trimbos-Kemper T. Diagnostic hysteroscopy and saline infusion sonography: prediction of intrauterine polyps and myomas. J Minim Invasive Gynecol. 2006;13(4):320-324.
11. Nathani F, Clark TJ. Uterine polypectomy in the management of abnormal uterine bleeding: a systematic review. J Minim Invasive Gynecol. 2006;13(4):260-268.
12. Garuti G, Centinaio G, Luerti M. Outpatient hysteroscopic polypectomy in postmenopausal women: a comparison between mechanical and electrosurgical resection. J Minim Invasive Gynecol. 2008;15(5):595-600.
13. Timmermans A, Veersema S. Ambulatory transcervical resection of polyps with the Duckbill polyp snare: a modality for treatment of endometrial polyps. J Minim Invasive Gynecol. 2005;12(1):37-39.
1. Clevenger-Hoeft M, Syrop C, Stovall D, Van Voorhis B. Sonohysterography in premenopausal women with and without abnormal bleeding. Obstet Gynecol. 1999;94(4):516-520.
2. DeWaay DJ, Syrop CH, Nygaard IE, Davis WA, Van Voorhis BJ. Natural history of uterine polyps and leiomyomata. Obstet Gynecol. 2002;100(1):3-7.
3. Lieng M, Istre O, Sandvik L, Qvigstad E. Prevalence, 1-year regression rate, and clinical significance of asymptomatic endometrial polyps: cross-sectional study. J Minim Invasive Gynecol. 2009;16(4):465-471.
4. Lopes RG, Baracat EC, de Albuquerque Neto LC, et al. Analysis of estrogen- and progersterone-receptor expression in endometrial polyps. J Minim Invasive Gynecol. 2007;14(3):300-303.
5. Haimov-Kochman R, Deri-Hasid R, Hamani Y, Voss E. The natural course of endometrial polyps: could they vanish when left untreated? Fertil Steril. 2009;92(2):828.e11-e12.
6. Breitkopf DM, Frederickson RA, Snyder RR. Detection of benign endometrial masses by endometrial stripe measurement in premenopausal women. Obstet Gynecol. 2004;104(1):120-125.
7. Marello F, Bettocchi S, Greco P, et al. Hysteroscopic evaluation of menopausal patients with sonographically atrophic endometrium. J Am Gynecol Laparosc. 2000;7(2):197-200.
8. Karlsson B, Granberg S, Wikland M, Ylostalo P, Torvid K, Marsal K, Valentin L.
Transvaginal ultrasonography of the endometrium in women with postmenopausal bleeding—a Nordic multicenter study. Am J Obstet Gynecol. 1995;172(5):1488-1494.
9. Paschopoulos M, Lolis E, Alamanos Y, Koliopoulos G, Paraskevaidis E. Vaginoscopic hysteroscopy and transvaginal sonography in the evaluation of patients with abnormal uterine bleeding. J Am Assoc Gynecol Laparosc. 2001;8(4):506-510.
10. Jansen FW, de Kroon CD, van Dongen H, Grooters C, Louwé L, Trimbos-Kemper T. Diagnostic hysteroscopy and saline infusion sonography: prediction of intrauterine polyps and myomas. J Minim Invasive Gynecol. 2006;13(4):320-324.
11. Nathani F, Clark TJ. Uterine polypectomy in the management of abnormal uterine bleeding: a systematic review. J Minim Invasive Gynecol. 2006;13(4):260-268.
12. Garuti G, Centinaio G, Luerti M. Outpatient hysteroscopic polypectomy in postmenopausal women: a comparison between mechanical and electrosurgical resection. J Minim Invasive Gynecol. 2008;15(5):595-600.
13. Timmermans A, Veersema S. Ambulatory transcervical resection of polyps with the Duckbill polyp snare: a modality for treatment of endometrial polyps. J Minim Invasive Gynecol. 2005;12(1):37-39.
OBG Management ©2010 Dowden Health Media
The gynecologist’s role in managing menstrual migraine
A.G. is a student who misses 2 or 3 days of school each month because of debilitating menstrual migraine (MM) with vomiting. Her referring physician prescribed topiramate, but the drug caused bothersome cognitive effects without relieving her headaches. She is taking a 30-μg oral contraceptive (OC) that contains ethinyl estradiol and drospirenone. Despite starting and stopping the OC several times, she has had no relief. She also takes 100 mg topiramate at bedtime, and 7 to 10 tablets of sumatriptan (100 mg) a month—usually during the menstrual week.
The ObGyn discontinues topiramate and changes dosing of the OC to active pills only for 12 consecutive weeks. During the 13th week, the patient is instructed to take 0.9 mg conjugated equine estrogens twice daily for 7 days before resuming extended-cycle oral contraception. This regimen completely eliminates menstrual migraine. The infrequent migraines the patient does experience—which she attributes to weather fronts—are successfully managed with no more than two doses of a triptan in a month.
A busy gynecology practice sees more migraineurs in a day than a neurology practice sees in a month. But most of the migraineurs who visit gynecology offices have a chief complaint other than headache, and most leave without having mentioned the migraine—returning home to continue treating themselves with the same remedies their mothers used. Many of these women fail to seek a specific diagnosis, or treatment, until they develop chronic daily headaches or become unable to work.
Current theories of migraine implicate spreading cortical depression; trigeminal nerve activation with attendant vasodilation; and neurogenic inflammation.
Menstrual migraine (MM) is, arguably, the most common disabling condition encountered in women’s health. These migraines are more severe, last longer, and are more resistant to treatment than those that occur at other times in the cycle.1,2 And, although headache specialists are adept at diagnosing migraine and prescribing a host of antiepileptic drugs and other migraine preventives, many of these specialists are uncomfortable manipulating the hormonal underpinnings of these “super migraines.”
This article outlines diagnostic criteria, treatment options, and preventive strategies, including hormonal therapy. In the process, it demonstrates how, in many cases, a gynecologist’s expertise in managing hormonal triggers may be the determinant of successful treatment.
Migraine prefers women
Migraine is the most common of all disabling headaches and afflicts 13% of the US population, but with a markedly skewed distribution: It preferentially attacks women in a 3:1 preponderance. The lifetime prevalence of migraine in women is 33%. Migraine strikes women primarily during reproductive years, when many women seek routine health care from a gynecologist rather than an internist or general practitioner.3
Menstrual migraine is defined as migraine without aura that occurs in predictable association with menses. Its onset falls within a 5-day window, spanning 2 days before the onset of menses through the third day of bleeding.5 Although the complete exclusion of migraine with aura from diagnostic criteria is controversial, headache specialists generally agree that aura is uncommonly associated with MM, probably owing to the low-estrogen environment. (Higher concentrations of estrogen are associated with an increased likelihood of aura, similar to the association between estrogen and seizure activity.)
The migraine trigger appears to be estrogen withdrawal in susceptible persons—either during the natural menstrual cycle or as a result of cycling onto inert pills in an oral contraceptive (OC) regimen. A population study found that 39% of menstruating women experience headaches with menses, and almost one third of these headaches meet established criteria for MM or menstrual-related migraine.4
The distinction between these two entities, MM and menstrual-related migraine, is largely semantic. With MM, the menstrual attack is the patient’s only migraine. The broader term implies that she may have other attacks in addition to the menstrual ones. In this article, I’ve gathered both categories under the term MM, which also includes headaches that arise at the time of estrogen withdrawal associated with OC use.
Regardless of what we call them, these headaches have proved to be particularly vexing to headache medicine specialists who hesitate to address hormonal factors.
Diagnostic criteria are clear
Formal diagnosis of migraine requires that at least two of four signature characteristics plus at least one of two associated symptoms be present.5 The four characteristics are:
- moderate or severe pain
- throbbing
- unilateral location
- intensification of headache upon activity.
Any combination of two suffices for diagnosis—much to the astonishment of many patients who mistakenly believe that migraine must be severe or one-sided.
Associated symptoms include either nausea or both photophobia and phonophobia, the latter often signified by the simple preference to be in a dark, quiet room during an attack.
Untreated, migraine usually lasts between 4 and 72 hours.
A practical, clinical approach to diagnosis is to look for the episodic disabling headache. By disabling, I mean the presence of associated nausea or the need to stop one’s activities and lie down. A stable history of attacks with predictable menstrual association offers further confirmation.
Neck tension may be present
Symptoms of neck tension do not rule out migraine. In fact, neck pain is far more common at the time of treatment than is nausea, and migraineurs frequently describe neck pain that radiates forward during an attack.6
Treatment options begin with NSAIDs
Initial treatment of MM is no different than that of other migraines. Mild and moderate attacks can often be managed with nonsteroidal anti-inflammatory drugs (NSAIDs), which concomitantly treat comorbid dysmenorrhea.
A small randomized trial found that mefenamic acid (500 mg every 8 hours, as needed) was superior to placebo for acute MM, and a small crossover trial compared two formulations of diclofenac—50-mg sachets and tablets—with placebo in the treatment of acute migraine (TABLE). Almost one quarter of the patients taking the sachet form were pain-free at 2 hours, compared with 18.5% of patients taking the tablets and 11.7% of those who received placebo.7,8
Injectable ketorolac is more potent and is proving to be as effective as or more effective than triptans or opioids for severe, persistent migraine.
When migraine is more severe, or when NSAIDs no longer suffice, treatment advances to migraine-specific medications—specifically, ergotamines and triptans.
Brand names of drugs mentioned in this article
| Drug | Brand name |
|---|---|
| NSAIDs | |
| Diclofenac sodium | Voltaren (and others) |
| Ketorolac tromethamine | Toradol (and others) |
| Mefenamic acid | Ponstel (and others) |
| Naproxen sodium | Naprelan (and others) |
| Triptans | |
| Eletriptan hydrobromide | Relpax |
| Frovatriptan succinate | Frova |
| Naratriptan hydrochloride | Amerge |
| Sumatriptan succinate | Imitrex |
| Ergotamines | |
| Dihydroergotamine mesylate | D.H.E. 45, Migranal |
| Other | |
| Butalbital, acetaminophen, and caffeine | Fioricet |
| Leuprolide acetate | Lupron, Eligard |
Ergots and ergotamines have varied safety profiles in pregnancy
Dihydroergotamine belongs to the oldest family of migraine-specific drugs, although it is not widely used today. One reason may be that most migraineurs are women of reproductive age, and ergots are oxytocic and potential teratogens. Furthermore, they are not recommended in lactation. Ergotamines, however, are quite diverse, and some have more acceptable FDA use-in-pregnancy ratings. Injectable dihydroergotamine appears to be as effective as or less effective than triptans for migraine pain, but more effective than other drugs, such as NSAIDs or analgesics, for acute attacks.9 The nasal spray is more effective than placebo, but less effective than triptans.9
Triptans are relatively safe for women of reproductive age
Most information on use of triptans in pregnancy concerns sumatriptan. Like all triptans, it falls into Category C; however, its published profile is reassuring.10-12 Data from Sweden on 2,027 first-trimester exposures show a 3.6% risk of birth defects (major and minor), compared with an identical 3.6% risk in the general Swedish population.12 Sumatriptan is considerably less lipophilic than most of the drugs in its class and has been rated as compatible with breastfeeding.13
All triptans except eletriptan have been studied in the treatment of acute MM and found to be superior to placebo.14 These serotonin 1B/1D agonists are tailored therapy for acute migraine because, unlike analgesics, they address the underlying pathology of the attack, inhibiting the release of vasoactive peptides, promoting selective meningeal vasoconstriction, and blocking pain pathways in the brainstem. They also resolve the associated symptoms of nausea and photophobia. Triptans should not be used in the presence of untreated hypertension or vascular disease (cardiovascular, cerebrovascular, or peripheral vascular disease).
Watch for “rebound” headache. Like all acute treatment—be it a simple analgesic, NSAID, caffeine-containing compound, butalbital, ergotamine, or opioid—too-frequent use of triptans can produce medication-overuse headache, also referred to as rebound headache. In general, try to limit the use of agents for acute migraine, whether prescription or over-the-counter, to no more than 2 days a week to avoid this consequence. Also, be aware that some agents—notably, butalbital-containing compounds—may cause rebound headache, even when given as infrequently as 5 days of the month.15
Some preventive strategies are “nonspecific”
When treatment of acute MM is inadequate, management shifts toward prevention. Options include nonspecific (those that do not address the hormonal trigger) and specific (hormonal) strategies. Nonspecific strategies rely on the predictable onset of MM for proper timing of the therapeutic intervention; therefore, women who have irregular cycles are not good candidates for this approach. The presence of comorbid conditions such as dysmenorrhea, menometrorrhagia, and endometriosis may argue for early adoption of specific strategies that improve both conditions at once.
The fact that menstrual migraine (MM) is associated with estrogen withdrawal prompts a question: What happens immediately postpartum, when estrogen levels decline with loss of the placenta?
Although hormonal fluctuations generally stabilize during pregnancy, and most menstrual migraineurs experience fewer headaches during gestation, that protective effect erodes at delivery, when the incidence of migraine can be as high as 40% in the first week.41
Investigators who prospectively studied 49 migraineurs—two of whom were affected by migraine with aura and 47 by migraine without aura—found improvement in 46.8% of these women during the first trimester of pregnancy, 83% during the second trimester, and 87.2% during the third trimester, with complete remission rates of 10.6%, 53.2%, and 78.7%, respectively.42 During the first postpartum week, migraine recurred in 34% of women.42 Interestingly, women who had a history of MM were less likely to improve during the first and third trimesters of pregnancy.42
In a separate prospective investigation of 985 women who delivered over a 3-month period in one tertiary-care facility, 381 experienced postpartum headache.43 The median time to onset of the headache was 2 days, and the median duration was 4 hours. More than 75% of these headaches were primary headaches.43 Only a small percentage (4%) were incapacitating.43
When migraine may signal a more serious condition
Another question arises when a woman experiences a severe headache shortly after delivery: Could the headache be related to preeclampsia or another serious complication of pregnancy and delivery? According to Contag and colleagues, immediate assessment may be warranted.44
“Characteristics to consider are the association of the headache with elevated blood pressure (which could signal postpartum preeclampsia), the sudden onset of an atypical headache, and variations to the usual nature of the migraine, such as the onset of new neurological symptoms. Postpartum women with any of these characteristics should be evaluated in the emergency department, and neuroimaging should be strongly considered,” they write.44
Treatment reverts to prepregnancy options
Assuming preeclampsia or another serious condition is not the culprit, the treatment options for postpartum migraine revert to prepregnancy choices, as effect on the developing fetus is no longer a concern. Nonsteroidal anti-inflammatory drugs (NSAIDs) are the first line of therapy. If they are ineffective, proceed to an ergotamine or triptan.—Janelle Yates, Senior Editor
Nonhormonal (nonspecific) therapy NSAIDs may provide some relief. In one small study, naproxen sodium (550 mg twice daily) was administered for 2 weeks, beginning 1 week before the anticipated onset of menses. It modestly reduced the overall duration and severity of menstrual migraine16—a benefit that must be weighed against the risk of adverse events and the more recent FDA black box warning about its potentially serious heart and gastrointestinal risks.
Triptan regimens may prevent MM. Several triptans have been investigated for prevention of MM. A small, open-label study evaluated the use of oral sumatriptan (25 mg three times daily), beginning 2 to 3 days before the anticipated onset of MM and continuing for 5 days.17 Menstrual attacks were prevented in just over 50% of cases.
Naratriptan (1 mg twice daily), beginning 2 days before anticipated menses and continuing for 5 days, reduced the number of MM attacks by 50%, with side effects comparable to placebo.18 A higher dosage (2.5 mg twice daily) did not prove to be superior to placebo, for unexplained reasons.
In a similar trial, frovatriptan (2.5 mg daily or 2.5 mg twice daily), beginning 2 days before the anticipated onset of MM and continuing for 6 days, prevented at least 50% of MM.19 Twice-daily dosing was superior to daily administration.
Magnesium may shorten MM. In one small study, oral magnesium (360 mg daily), beginning on the 15th day of the cycle and continuing through the menses, shortened the duration of MM and improved menstrual complaints better than placebo.20
The goal of hormonal therapy is to eliminate or sufficiently minimize the premenstrual decline in estrogen that is believed to precipitate MM.21 An observational study of 229 women found that hormonal strategies prevented MM in 73% of cases (81% when taken as directed).22
It is fortunate that MM, by definition, is migraine without aura, because the use of combined OCs has been controversial in the setting of migraine with aura. International studies have reported a small but increased risk of stroke associated with their use, although a subset of women with migraine had no increased risk.23-25 In contrast, no U.S. study since 1975 has found an increased risk of stroke associated with the use of OCs.26 One large domestic study reviewed 3.6 million woman-years of use and found no increased risk of ischemic stroke with the low-dose OCs currently available, nor did a pooled analysis of U.S. studies.27,28
The discrepancy is likely explained by the strong relative contraindication, in the United States, to the use of OCs in smokers older than 35 years (the smoking prevalence in most of the European case series was more than 50%), as well as the more prevalent use of high-dose OCs in the international studies. High-dose pills were implicated in the majority of stroke cases in the World Health Organization (WHO) study, but were used by only 0.7% of cases and controls in the pooled U.S. studies.23 Nevertheless, both ACOG and WHO concluded that the risk of OC use usually outweighs the benefit in women older than 35 years whose migraines are complicated by focal neurologic deficits.
Extended-cycle OCs may offer a lengthy reprieve. Regimens that forego monthly withdrawal bleeds and provide extended administration of active pills can afford migraineurs a lengthy reprieve from MM.29 Breakthrough bleeding is the most common side effect but tends to decrease over time. It is preferable for the patient to take the pill at bedtime to avoid a drug nadir during susceptible stages of sleep, which may be associated with migraine generation. It also is prudent to avoid concomitant administration of drugs that might increase the rate of hepatic metabolism of estrogen (e.g., a high dosage of topiramate) and lead to a more rapid decline in concentration.
When using an extended-cycle regimen that allows for periodic withdrawal bleeds, give the patient supplemental estrogen during the withdrawal week if the decline in ethinyl estradiol (EE) exceeds 10 μg, a threshold that appears to elicit MM in susceptible women.21,22 For example, with an extended-cycle regimen that contains 30 μg of EE, it may be necessary to add 20 μg of estrogen during the placebo week to prevent MM, whereas an extended-cycle regimen that contains 20 μg EE, declining to 10 μg during the 13th week, would be adequate as packaged.
21/7 hormonal OCs may require supplemental estrogen to prevent MM. The late luteal-phase decline in estradiol concentration during the natural menstrual cycle is equivalent to the decline experienced with the transition to inert pills in 20- to 25-μg EE formulations. Therefore, these regimens confer the same risk of MM as the woman’s natural cycle. Products that contain incrementally higher dosages of EE (30, 35, and 50 μg) confer an increasingly greater risk of MM. Supplementation of estrogen during the placebo week may prevent MM by limiting the decline in estrogen.
In a small, open-label study, women took an OC containing 20 μg EE at bedtime on days 1 to 21 of the cycle, followed by 0.9 mg of conjugated equine estrogens (CEE) on days 22 to 28. All patients reported a reduction in migraine frequency of at least 50%, with a mean reduction of 78%.30
Parenteral options can be created utilizing a transdermal 20-μg EE/norelgestromin patch or a 15-μg EE/etonogestrel vaginal ring. With the first approach, I recommend adding a 0.1-mg EE patch during the withdrawal week to prevent MM. With the latter, a 0.075-mg EE patch may be used during the week following ring removal.31
Consider a menstrually targeted estrogen supplement. Some women who have contraindications to use of an OC may still be candidates for targeted strategies using lower dosages of supplemental estrogen rather than a combination OC. Among the options is perimenstrual administration of an estradiol patch or gel.32 One study found a 0.1-mg estrogen patch (worn 7 days and applied just before the expected onset of menses) to be effective, but lower dosages (0.025 to 0.05 mg EE) were not.33 Daily application of 1.5 mg estradiol gel for 7 days, beginning before the onset of MM, was also effective in preventing MM.34-36
With targeted strategies, timing is critical; if estrogen is begun too early, the incidence of migraine may rise after cessation.36
GnRH agonists may benefit women who have isolated MM. Administration of gonadotropin-releasing hormone (GnRH) agonists has been shown to ease MM, but the baseline frequency of headaches appears to be an important variable in the success of these agents.37-39 Leuprolide acetate markedly diminished MM in women who seldom experienced headaches other than their menstrual attacks.37 It did not benefit women who experienced migraine in the setting of chronic headache (≥15 days/month), probably because chronic migraine is influenced by other variables, such as nonrestorative sleep and overuse of medication.15,38,40
No evidence that progestin-only contraceptives are effective. Although these agents have been proposed for treatment of migraine, evidence of their benefit is lacking. It is my opinion that, more often than not, they exacerbate migraine.
1. MacGregor EA, Hackshaw A. Prevalence of migraine on each day of the natural menstrual cycle. Neurology. 2004;63(2):351-353.
2. Granella F, Sances G, Allais G, et al. Characteristics of menstrual and nonmenstrual attacks in women with menstrually related migraine referred to headache centres. Cephalalgia. 2004;24(9):707-716.
3. Launer LJ, Terwindt GM, Ferrari MD. The prevalence and characteristics of migraine in a population-based cohort: the GEM study. Neurology. 1999;53(3):537-542.
4. Couturier EG, Bomhof MA, Neven AK, van Duijn NP. Menstrual migraine in a representative Dutch population sample: prevalence, disability and treatment. Cephalalgia. 2003;23(4):302-308.
5. Headache Classification Subcommittee of the International Headache Society. The International Classification of Headache Disorders. 2nd ed. Cephalalgia. 2004;24(suppl 1):9-160.
6. Calhoun AH, Ford S, Millen C, Finkel AG, Truong Y, Nie Y. The prevalence of neck pain in migraine [abstract]. Headache. 2010 Jan 20.-[Epub ahead of print].
7. Al-Waili NS. Treatment of menstrual migraine with prostaglandin synthesis inhibitor mefenamic acid: double-blind study with placebo. Eur J Med Res. 2000;5(4):176-182.
8. Diener HC, Montagna P, Gács G, et al. Efficacy and tolerability of diclofenac potassium sachets in migraine: a randomized, double-blind, cross-over study in comparison with diclofenac potassium tablets and placebo. Cephalalgia. 2006;26(5):537-547.
9. Schürks M. Dihydroergotamine: role in the treatment of migraine. Expert Opin Drug Metab Toxicol. 2009;5(9):1141-1148.
10. Loder E. Safety of sumatriptan in pregnancy: a review of the data so far. CNS Drugs. 2003;17(1):1-7.
11. Shuhaiber S, Pastuszak A, Schick B, et al. Pregnancy outcome following first trimester exposure to sumatriptan. Neurology. 1998;51(2):581-583.
12. Cunnington M, Ephross S, Churchill P. The safety of sumatriptan and naratriptan in pregnancy: what have we learned? Headache. 2009;49(10):1414-1422.
13. Ressel G. AAP updates statement for transfer of drugs and other chemicals into breast milk. American Academy of Pediatrics. Am Fam Physician. 2002;65(5):979-980.
14. Ashkenazi A, Silberstein S. Menstrual migraine: a review of hormonal causes, prophylaxis and treatment. Expert Opin Pharmacother. 2007;8(11):1605-1613.
15. Bigal ME, Serrano D, Buse D, Scher A, Stewart WF, Lipton RB. Acute migraine medications and evolution from episodic to chronic migraine: a longitudinal population-based study. Headache. 2008;48(8):1157-1168.
16. Sances G, Martignoni E, Fioroni L, Blandini F, Facchinetti F, Nappi G. Naproxen sodium in menstrual migraine prophylaxis: a double-blind placebo controlled study. Headache. 1990;30(11):705-709.
17. Newman LC, Lipton RB, Lay CL, Solomon S. A pilot study of oral sumatriptan as intermittent prophylaxis of menstruation-related migraine. Neurology. 1998;51(1):307-309.
18. Newman L, Mannix LK, Landy S, et al. Naratriptan as short-term prophylaxis of menstrually associated migraine: a randomized, double-blind, placebo-controlled study. Headache. 2001;41(3):248-256.
19. Silberstein SD, Elkind AH, Schreiber C, Keywood C. A randomized trial of frovatriptan for the intermittent prevention of menstrual migraine. Neurology. 2004;63(2):261-269.
20. Facchinetti F, Sances G, Borella P, Genazzani AR, Nappi G. Magnesium prophylaxis of menstrual migraine: effects on intracellular magnesium. Headache. 1991;31(5):298-301.
21. Calhoun A. Women’s Issues in Headache. In: Loder E, ed. Headache. Philadelphia: American College of Physicians; 2004:157-177.
22. Calhoun A, Ford S. Elimination of menstrual-related migraine beneficially impacts chronification and medication overuse. Headache. 2008;48(8):1186-1193.
23. Ischaemic stroke and combined oral contraceptives: results of an international, multicentre, case-control study. WHO Collaborative Study of Cardiovascular Disease and Steroid Hormone Contraception. Lancet. 1996;348:498-505.
24. Chan WS, Ray J, Wai EK, et al. Risk of stroke in women exposed to low-dose oral contraceptives: a critical evaluation of the evidence. Arch Intern Med. 2004;164(7):741-747.
25. Etminan M, Takkouche B, Isorna FC, Samii A. Risk of ischaemic stroke in people with migraine: systematic review and meta-analysis of observational studies. BMJ. 2005;330(7482):63.-
26. Oral contraceptives and stroke in young women. Associated risk factors. JAMA. 1975;231:718-722.
27. Petitti DB, Sidney S, Bernstein A, Wolf S, Quesenberry C, Ziel HK. Stroke in users of low-dose oral contraceptives. N Engl J Med. 1996;335(1):8-15.
28. Schwartz SM, Petitti DB, Siscovick DS, et al. Stroke and use of low-dose oral contraceptives in young women: a pooled analysis of two US studies. Stroke. 1998;29(11):2277-2284.
29. Sulak PJ, Scow RD, Preece C, Riggs MW, Kuehl TJ. Hormone withdrawal symptoms in oral contraceptive users. Obstet Gynecol. 2000;95(2):261-266.
30. Calhoun AH. A novel specific prophylaxis for menstrual-associated migraine. South Med J. 2004;97(9):819-822.
31. Calhoun AH, Hutchinson S. Hormonal therapies for menstrual migraine. Curr Pain Headache Rep. 2009;13(5):381-385.
32. Pringsheim T, Davenport WJ, Dodick D. Acute treatment and prevention of menstrually related migraine headache: evidence-based review. Neurology. 2008;70(17):1555-1563.
33. Pradalier A VD, Beaulieu PH, et al. Correlation between oestradiol plasma level and therapeutic effect on menstrual migraine. In: FC Rose, ed. New advances in headache research. London: Smith-Gordon; 1994:129-132.
34. de Lignières B, Vincens M, Mauvais-Jarvis P, Mas JL, Touboul PJ, Bousser MG. Prevention of menstrual migraine by percutaneous oestradiol. Br Med J (Clin Res Ed). 1986;293(6561):1540.-
35. Dennerstein L, Morse C, Burrows G, Oats J, Brown J, Smith M. Menstrual migraine: a double-blind trial of percutaneous estradiol. Gynecol Endocrinol. 1988;2(2):113-120.
36. MacGregor EA, Frith A, Ellis J, Aspinall L, Hackshaw A. Prevention of menstrual attacks of migraine: a double-blind placebo-controlled crossover study. Neurology. 2006;67(12):2159-2163.
37. Murray SC, Muse KN. Effective treatment of severe menstrual migraine headaches with gonadotropin-releasing hormone agonist and “add-back” therapy. Fertil Steril. 1997;67(2):390-393.
38. Martin V, Wernke S, Mandell K, et al. Medical oophorectomy with and without estrogen add-back therapy in the prevention of migraine headache. Headache. 2003;43(4):309-321.
39. Lichten EM, Lichten JB, Whitty A, Pieper D. The confirmation of a biochemical marker for women’s hormonal migraine: the depo-estradiol challenge test. Headache. 1996;36(6):367-371.
40. Calhoun AH, Ford S. Behavioral sleep modification may revert transformed migraine to episodic migraine. Headache. 2007;47(8):1178-1183.
41. Stein GS. Headaches in the first postpartum week and their relationship to migraine. Headache. 1981;21(5):201-205.
42. Sances G, Granella F, Nappi RE, et al. Course of migraine during pregnancy and postpartum: a prospective study. Cephalalgia. 2003;23(3):197-205.
43. Goldszmidt E, Kern R, Chaput A, Macarthur A. The incidence and etiology of postpartum headaches: a prospective cohort study. Can J Anaesth. 2005;52(9):971-977.
44. Contag SA, Mertz HL, Bushnell CD. Migraine during pregnancy: Is it more than a headache? Nat Rev Neurol. 2009;5(8):449-456.
A.G. is a student who misses 2 or 3 days of school each month because of debilitating menstrual migraine (MM) with vomiting. Her referring physician prescribed topiramate, but the drug caused bothersome cognitive effects without relieving her headaches. She is taking a 30-μg oral contraceptive (OC) that contains ethinyl estradiol and drospirenone. Despite starting and stopping the OC several times, she has had no relief. She also takes 100 mg topiramate at bedtime, and 7 to 10 tablets of sumatriptan (100 mg) a month—usually during the menstrual week.
The ObGyn discontinues topiramate and changes dosing of the OC to active pills only for 12 consecutive weeks. During the 13th week, the patient is instructed to take 0.9 mg conjugated equine estrogens twice daily for 7 days before resuming extended-cycle oral contraception. This regimen completely eliminates menstrual migraine. The infrequent migraines the patient does experience—which she attributes to weather fronts—are successfully managed with no more than two doses of a triptan in a month.
A busy gynecology practice sees more migraineurs in a day than a neurology practice sees in a month. But most of the migraineurs who visit gynecology offices have a chief complaint other than headache, and most leave without having mentioned the migraine—returning home to continue treating themselves with the same remedies their mothers used. Many of these women fail to seek a specific diagnosis, or treatment, until they develop chronic daily headaches or become unable to work.

Current theories of migraine implicate spreading cortical depression; trigeminal nerve activation with attendant vasodilation; and neurogenic inflammation.
Menstrual migraine (MM) is, arguably, the most common disabling condition encountered in women’s health. These migraines are more severe, last longer, and are more resistant to treatment than those that occur at other times in the cycle.1,2 And, although headache specialists are adept at diagnosing migraine and prescribing a host of antiepileptic drugs and other migraine preventives, many of these specialists are uncomfortable manipulating the hormonal underpinnings of these “super migraines.”
This article outlines diagnostic criteria, treatment options, and preventive strategies, including hormonal therapy. In the process, it demonstrates how, in many cases, a gynecologist’s expertise in managing hormonal triggers may be the determinant of successful treatment.
Migraine prefers women
Migraine is the most common of all disabling headaches and afflicts 13% of the US population, but with a markedly skewed distribution: It preferentially attacks women in a 3:1 preponderance. The lifetime prevalence of migraine in women is 33%. Migraine strikes women primarily during reproductive years, when many women seek routine health care from a gynecologist rather than an internist or general practitioner.3
Menstrual migraine is defined as migraine without aura that occurs in predictable association with menses. Its onset falls within a 5-day window, spanning 2 days before the onset of menses through the third day of bleeding.5 Although the complete exclusion of migraine with aura from diagnostic criteria is controversial, headache specialists generally agree that aura is uncommonly associated with MM, probably owing to the low-estrogen environment. (Higher concentrations of estrogen are associated with an increased likelihood of aura, similar to the association between estrogen and seizure activity.)
The migraine trigger appears to be estrogen withdrawal in susceptible persons—either during the natural menstrual cycle or as a result of cycling onto inert pills in an oral contraceptive (OC) regimen. A population study found that 39% of menstruating women experience headaches with menses, and almost one third of these headaches meet established criteria for MM or menstrual-related migraine.4
The distinction between these two entities, MM and menstrual-related migraine, is largely semantic. With MM, the menstrual attack is the patient’s only migraine. The broader term implies that she may have other attacks in addition to the menstrual ones. In this article, I’ve gathered both categories under the term MM, which also includes headaches that arise at the time of estrogen withdrawal associated with OC use.
Regardless of what we call them, these headaches have proved to be particularly vexing to headache medicine specialists who hesitate to address hormonal factors.
Diagnostic criteria are clear
Formal diagnosis of migraine requires that at least two of four signature characteristics plus at least one of two associated symptoms be present.5 The four characteristics are:
- moderate or severe pain
- throbbing
- unilateral location
- intensification of headache upon activity.
Any combination of two suffices for diagnosis—much to the astonishment of many patients who mistakenly believe that migraine must be severe or one-sided.
Associated symptoms include either nausea or both photophobia and phonophobia, the latter often signified by the simple preference to be in a dark, quiet room during an attack.
Untreated, migraine usually lasts between 4 and 72 hours.
A practical, clinical approach to diagnosis is to look for the episodic disabling headache. By disabling, I mean the presence of associated nausea or the need to stop one’s activities and lie down. A stable history of attacks with predictable menstrual association offers further confirmation.
Neck tension may be present
Symptoms of neck tension do not rule out migraine. In fact, neck pain is far more common at the time of treatment than is nausea, and migraineurs frequently describe neck pain that radiates forward during an attack.6
Treatment options begin with NSAIDs
Initial treatment of MM is no different than that of other migraines. Mild and moderate attacks can often be managed with nonsteroidal anti-inflammatory drugs (NSAIDs), which concomitantly treat comorbid dysmenorrhea.
A small randomized trial found that mefenamic acid (500 mg every 8 hours, as needed) was superior to placebo for acute MM, and a small crossover trial compared two formulations of diclofenac—50-mg sachets and tablets—with placebo in the treatment of acute migraine (TABLE). Almost one quarter of the patients taking the sachet form were pain-free at 2 hours, compared with 18.5% of patients taking the tablets and 11.7% of those who received placebo.7,8
Injectable ketorolac is more potent and is proving to be as effective as or more effective than triptans or opioids for severe, persistent migraine.
When migraine is more severe, or when NSAIDs no longer suffice, treatment advances to migraine-specific medications—specifically, ergotamines and triptans.
Brand names of drugs mentioned in this article
| Drug | Brand name |
|---|---|
| NSAIDs | |
| Diclofenac sodium | Voltaren (and others) |
| Ketorolac tromethamine | Toradol (and others) |
| Mefenamic acid | Ponstel (and others) |
| Naproxen sodium | Naprelan (and others) |
| Triptans | |
| Eletriptan hydrobromide | Relpax |
| Frovatriptan succinate | Frova |
| Naratriptan hydrochloride | Amerge |
| Sumatriptan succinate | Imitrex |
| Ergotamines | |
| Dihydroergotamine mesylate | D.H.E. 45, Migranal |
| Other | |
| Butalbital, acetaminophen, and caffeine | Fioricet |
| Leuprolide acetate | Lupron, Eligard |
Ergots and ergotamines have varied safety profiles in pregnancy
Dihydroergotamine belongs to the oldest family of migraine-specific drugs, although it is not widely used today. One reason may be that most migraineurs are women of reproductive age, and ergots are oxytocic and potential teratogens. Furthermore, they are not recommended in lactation. Ergotamines, however, are quite diverse, and some have more acceptable FDA use-in-pregnancy ratings. Injectable dihydroergotamine appears to be as effective as or less effective than triptans for migraine pain, but more effective than other drugs, such as NSAIDs or analgesics, for acute attacks.9 The nasal spray is more effective than placebo, but less effective than triptans.9
Triptans are relatively safe for women of reproductive age
Most information on use of triptans in pregnancy concerns sumatriptan. Like all triptans, it falls into Category C; however, its published profile is reassuring.10-12 Data from Sweden on 2,027 first-trimester exposures show a 3.6% risk of birth defects (major and minor), compared with an identical 3.6% risk in the general Swedish population.12 Sumatriptan is considerably less lipophilic than most of the drugs in its class and has been rated as compatible with breastfeeding.13
All triptans except eletriptan have been studied in the treatment of acute MM and found to be superior to placebo.14 These serotonin 1B/1D agonists are tailored therapy for acute migraine because, unlike analgesics, they address the underlying pathology of the attack, inhibiting the release of vasoactive peptides, promoting selective meningeal vasoconstriction, and blocking pain pathways in the brainstem. They also resolve the associated symptoms of nausea and photophobia. Triptans should not be used in the presence of untreated hypertension or vascular disease (cardiovascular, cerebrovascular, or peripheral vascular disease).
Watch for “rebound” headache. Like all acute treatment—be it a simple analgesic, NSAID, caffeine-containing compound, butalbital, ergotamine, or opioid—too-frequent use of triptans can produce medication-overuse headache, also referred to as rebound headache. In general, try to limit the use of agents for acute migraine, whether prescription or over-the-counter, to no more than 2 days a week to avoid this consequence. Also, be aware that some agents—notably, butalbital-containing compounds—may cause rebound headache, even when given as infrequently as 5 days of the month.15
Some preventive strategies are “nonspecific”
When treatment of acute MM is inadequate, management shifts toward prevention. Options include nonspecific (those that do not address the hormonal trigger) and specific (hormonal) strategies. Nonspecific strategies rely on the predictable onset of MM for proper timing of the therapeutic intervention; therefore, women who have irregular cycles are not good candidates for this approach. The presence of comorbid conditions such as dysmenorrhea, menometrorrhagia, and endometriosis may argue for early adoption of specific strategies that improve both conditions at once.
The fact that menstrual migraine (MM) is associated with estrogen withdrawal prompts a question: What happens immediately postpartum, when estrogen levels decline with loss of the placenta?
Although hormonal fluctuations generally stabilize during pregnancy, and most menstrual migraineurs experience fewer headaches during gestation, that protective effect erodes at delivery, when the incidence of migraine can be as high as 40% in the first week.41
Investigators who prospectively studied 49 migraineurs—two of whom were affected by migraine with aura and 47 by migraine without aura—found improvement in 46.8% of these women during the first trimester of pregnancy, 83% during the second trimester, and 87.2% during the third trimester, with complete remission rates of 10.6%, 53.2%, and 78.7%, respectively.42 During the first postpartum week, migraine recurred in 34% of women.42 Interestingly, women who had a history of MM were less likely to improve during the first and third trimesters of pregnancy.42
In a separate prospective investigation of 985 women who delivered over a 3-month period in one tertiary-care facility, 381 experienced postpartum headache.43 The median time to onset of the headache was 2 days, and the median duration was 4 hours. More than 75% of these headaches were primary headaches.43 Only a small percentage (4%) were incapacitating.43
When migraine may signal a more serious condition
Another question arises when a woman experiences a severe headache shortly after delivery: Could the headache be related to preeclampsia or another serious complication of pregnancy and delivery? According to Contag and colleagues, immediate assessment may be warranted.44
“Characteristics to consider are the association of the headache with elevated blood pressure (which could signal postpartum preeclampsia), the sudden onset of an atypical headache, and variations to the usual nature of the migraine, such as the onset of new neurological symptoms. Postpartum women with any of these characteristics should be evaluated in the emergency department, and neuroimaging should be strongly considered,” they write.44
Treatment reverts to prepregnancy options
Assuming preeclampsia or another serious condition is not the culprit, the treatment options for postpartum migraine revert to prepregnancy choices, as effect on the developing fetus is no longer a concern. Nonsteroidal anti-inflammatory drugs (NSAIDs) are the first line of therapy. If they are ineffective, proceed to an ergotamine or triptan.—Janelle Yates, Senior Editor
Nonhormonal (nonspecific) therapy NSAIDs may provide some relief. In one small study, naproxen sodium (550 mg twice daily) was administered for 2 weeks, beginning 1 week before the anticipated onset of menses. It modestly reduced the overall duration and severity of menstrual migraine16—a benefit that must be weighed against the risk of adverse events and the more recent FDA black box warning about its potentially serious heart and gastrointestinal risks.
Triptan regimens may prevent MM. Several triptans have been investigated for prevention of MM. A small, open-label study evaluated the use of oral sumatriptan (25 mg three times daily), beginning 2 to 3 days before the anticipated onset of MM and continuing for 5 days.17 Menstrual attacks were prevented in just over 50% of cases.
Naratriptan (1 mg twice daily), beginning 2 days before anticipated menses and continuing for 5 days, reduced the number of MM attacks by 50%, with side effects comparable to placebo.18 A higher dosage (2.5 mg twice daily) did not prove to be superior to placebo, for unexplained reasons.
In a similar trial, frovatriptan (2.5 mg daily or 2.5 mg twice daily), beginning 2 days before the anticipated onset of MM and continuing for 6 days, prevented at least 50% of MM.19 Twice-daily dosing was superior to daily administration.
Magnesium may shorten MM. In one small study, oral magnesium (360 mg daily), beginning on the 15th day of the cycle and continuing through the menses, shortened the duration of MM and improved menstrual complaints better than placebo.20
The goal of hormonal therapy is to eliminate or sufficiently minimize the premenstrual decline in estrogen that is believed to precipitate MM.21 An observational study of 229 women found that hormonal strategies prevented MM in 73% of cases (81% when taken as directed).22
It is fortunate that MM, by definition, is migraine without aura, because the use of combined OCs has been controversial in the setting of migraine with aura. International studies have reported a small but increased risk of stroke associated with their use, although a subset of women with migraine had no increased risk.23-25 In contrast, no U.S. study since 1975 has found an increased risk of stroke associated with the use of OCs.26 One large domestic study reviewed 3.6 million woman-years of use and found no increased risk of ischemic stroke with the low-dose OCs currently available, nor did a pooled analysis of U.S. studies.27,28
The discrepancy is likely explained by the strong relative contraindication, in the United States, to the use of OCs in smokers older than 35 years (the smoking prevalence in most of the European case series was more than 50%), as well as the more prevalent use of high-dose OCs in the international studies. High-dose pills were implicated in the majority of stroke cases in the World Health Organization (WHO) study, but were used by only 0.7% of cases and controls in the pooled U.S. studies.23 Nevertheless, both ACOG and WHO concluded that the risk of OC use usually outweighs the benefit in women older than 35 years whose migraines are complicated by focal neurologic deficits.
Extended-cycle OCs may offer a lengthy reprieve. Regimens that forego monthly withdrawal bleeds and provide extended administration of active pills can afford migraineurs a lengthy reprieve from MM.29 Breakthrough bleeding is the most common side effect but tends to decrease over time. It is preferable for the patient to take the pill at bedtime to avoid a drug nadir during susceptible stages of sleep, which may be associated with migraine generation. It also is prudent to avoid concomitant administration of drugs that might increase the rate of hepatic metabolism of estrogen (e.g., a high dosage of topiramate) and lead to a more rapid decline in concentration.
When using an extended-cycle regimen that allows for periodic withdrawal bleeds, give the patient supplemental estrogen during the withdrawal week if the decline in ethinyl estradiol (EE) exceeds 10 μg, a threshold that appears to elicit MM in susceptible women.21,22 For example, with an extended-cycle regimen that contains 30 μg of EE, it may be necessary to add 20 μg of estrogen during the placebo week to prevent MM, whereas an extended-cycle regimen that contains 20 μg EE, declining to 10 μg during the 13th week, would be adequate as packaged.
21/7 hormonal OCs may require supplemental estrogen to prevent MM. The late luteal-phase decline in estradiol concentration during the natural menstrual cycle is equivalent to the decline experienced with the transition to inert pills in 20- to 25-μg EE formulations. Therefore, these regimens confer the same risk of MM as the woman’s natural cycle. Products that contain incrementally higher dosages of EE (30, 35, and 50 μg) confer an increasingly greater risk of MM. Supplementation of estrogen during the placebo week may prevent MM by limiting the decline in estrogen.
In a small, open-label study, women took an OC containing 20 μg EE at bedtime on days 1 to 21 of the cycle, followed by 0.9 mg of conjugated equine estrogens (CEE) on days 22 to 28. All patients reported a reduction in migraine frequency of at least 50%, with a mean reduction of 78%.30
Parenteral options can be created utilizing a transdermal 20-μg EE/norelgestromin patch or a 15-μg EE/etonogestrel vaginal ring. With the first approach, I recommend adding a 0.1-mg EE patch during the withdrawal week to prevent MM. With the latter, a 0.075-mg EE patch may be used during the week following ring removal.31
Consider a menstrually targeted estrogen supplement. Some women who have contraindications to use of an OC may still be candidates for targeted strategies using lower dosages of supplemental estrogen rather than a combination OC. Among the options is perimenstrual administration of an estradiol patch or gel.32 One study found a 0.1-mg estrogen patch (worn 7 days and applied just before the expected onset of menses) to be effective, but lower dosages (0.025 to 0.05 mg EE) were not.33 Daily application of 1.5 mg estradiol gel for 7 days, beginning before the onset of MM, was also effective in preventing MM.34-36
With targeted strategies, timing is critical; if estrogen is begun too early, the incidence of migraine may rise after cessation.36
GnRH agonists may benefit women who have isolated MM. Administration of gonadotropin-releasing hormone (GnRH) agonists has been shown to ease MM, but the baseline frequency of headaches appears to be an important variable in the success of these agents.37-39 Leuprolide acetate markedly diminished MM in women who seldom experienced headaches other than their menstrual attacks.37 It did not benefit women who experienced migraine in the setting of chronic headache (≥15 days/month), probably because chronic migraine is influenced by other variables, such as nonrestorative sleep and overuse of medication.15,38,40
No evidence that progestin-only contraceptives are effective. Although these agents have been proposed for treatment of migraine, evidence of their benefit is lacking. It is my opinion that, more often than not, they exacerbate migraine.
A.G. is a student who misses 2 or 3 days of school each month because of debilitating menstrual migraine (MM) with vomiting. Her referring physician prescribed topiramate, but the drug caused bothersome cognitive effects without relieving her headaches. She is taking a 30-μg oral contraceptive (OC) that contains ethinyl estradiol and drospirenone. Despite starting and stopping the OC several times, she has had no relief. She also takes 100 mg topiramate at bedtime, and 7 to 10 tablets of sumatriptan (100 mg) a month—usually during the menstrual week.
The ObGyn discontinues topiramate and changes dosing of the OC to active pills only for 12 consecutive weeks. During the 13th week, the patient is instructed to take 0.9 mg conjugated equine estrogens twice daily for 7 days before resuming extended-cycle oral contraception. This regimen completely eliminates menstrual migraine. The infrequent migraines the patient does experience—which she attributes to weather fronts—are successfully managed with no more than two doses of a triptan in a month.
A busy gynecology practice sees more migraineurs in a day than a neurology practice sees in a month. But most of the migraineurs who visit gynecology offices have a chief complaint other than headache, and most leave without having mentioned the migraine—returning home to continue treating themselves with the same remedies their mothers used. Many of these women fail to seek a specific diagnosis, or treatment, until they develop chronic daily headaches or become unable to work.

Current theories of migraine implicate spreading cortical depression; trigeminal nerve activation with attendant vasodilation; and neurogenic inflammation.
Menstrual migraine (MM) is, arguably, the most common disabling condition encountered in women’s health. These migraines are more severe, last longer, and are more resistant to treatment than those that occur at other times in the cycle.1,2 And, although headache specialists are adept at diagnosing migraine and prescribing a host of antiepileptic drugs and other migraine preventives, many of these specialists are uncomfortable manipulating the hormonal underpinnings of these “super migraines.”
This article outlines diagnostic criteria, treatment options, and preventive strategies, including hormonal therapy. In the process, it demonstrates how, in many cases, a gynecologist’s expertise in managing hormonal triggers may be the determinant of successful treatment.
Migraine prefers women
Migraine is the most common of all disabling headaches and afflicts 13% of the US population, but with a markedly skewed distribution: It preferentially attacks women in a 3:1 preponderance. The lifetime prevalence of migraine in women is 33%. Migraine strikes women primarily during reproductive years, when many women seek routine health care from a gynecologist rather than an internist or general practitioner.3
Menstrual migraine is defined as migraine without aura that occurs in predictable association with menses. Its onset falls within a 5-day window, spanning 2 days before the onset of menses through the third day of bleeding.5 Although the complete exclusion of migraine with aura from diagnostic criteria is controversial, headache specialists generally agree that aura is uncommonly associated with MM, probably owing to the low-estrogen environment. (Higher concentrations of estrogen are associated with an increased likelihood of aura, similar to the association between estrogen and seizure activity.)
The migraine trigger appears to be estrogen withdrawal in susceptible persons—either during the natural menstrual cycle or as a result of cycling onto inert pills in an oral contraceptive (OC) regimen. A population study found that 39% of menstruating women experience headaches with menses, and almost one third of these headaches meet established criteria for MM or menstrual-related migraine.4
The distinction between these two entities, MM and menstrual-related migraine, is largely semantic. With MM, the menstrual attack is the patient’s only migraine. The broader term implies that she may have other attacks in addition to the menstrual ones. In this article, I’ve gathered both categories under the term MM, which also includes headaches that arise at the time of estrogen withdrawal associated with OC use.
Regardless of what we call them, these headaches have proved to be particularly vexing to headache medicine specialists who hesitate to address hormonal factors.
Diagnostic criteria are clear
Formal diagnosis of migraine requires that at least two of four signature characteristics plus at least one of two associated symptoms be present.5 The four characteristics are:
- moderate or severe pain
- throbbing
- unilateral location
- intensification of headache upon activity.
Any combination of two suffices for diagnosis—much to the astonishment of many patients who mistakenly believe that migraine must be severe or one-sided.
Associated symptoms include either nausea or both photophobia and phonophobia, the latter often signified by the simple preference to be in a dark, quiet room during an attack.
Untreated, migraine usually lasts between 4 and 72 hours.
A practical, clinical approach to diagnosis is to look for the episodic disabling headache. By disabling, I mean the presence of associated nausea or the need to stop one’s activities and lie down. A stable history of attacks with predictable menstrual association offers further confirmation.
Neck tension may be present
Symptoms of neck tension do not rule out migraine. In fact, neck pain is far more common at the time of treatment than is nausea, and migraineurs frequently describe neck pain that radiates forward during an attack.6
Treatment options begin with NSAIDs
Initial treatment of MM is no different than that of other migraines. Mild and moderate attacks can often be managed with nonsteroidal anti-inflammatory drugs (NSAIDs), which concomitantly treat comorbid dysmenorrhea.
A small randomized trial found that mefenamic acid (500 mg every 8 hours, as needed) was superior to placebo for acute MM, and a small crossover trial compared two formulations of diclofenac—50-mg sachets and tablets—with placebo in the treatment of acute migraine (TABLE). Almost one quarter of the patients taking the sachet form were pain-free at 2 hours, compared with 18.5% of patients taking the tablets and 11.7% of those who received placebo.7,8
Injectable ketorolac is more potent and is proving to be as effective as or more effective than triptans or opioids for severe, persistent migraine.
When migraine is more severe, or when NSAIDs no longer suffice, treatment advances to migraine-specific medications—specifically, ergotamines and triptans.
Brand names of drugs mentioned in this article
| Drug | Brand name |
|---|---|
| NSAIDs | |
| Diclofenac sodium | Voltaren (and others) |
| Ketorolac tromethamine | Toradol (and others) |
| Mefenamic acid | Ponstel (and others) |
| Naproxen sodium | Naprelan (and others) |
| Triptans | |
| Eletriptan hydrobromide | Relpax |
| Frovatriptan succinate | Frova |
| Naratriptan hydrochloride | Amerge |
| Sumatriptan succinate | Imitrex |
| Ergotamines | |
| Dihydroergotamine mesylate | D.H.E. 45, Migranal |
| Other | |
| Butalbital, acetaminophen, and caffeine | Fioricet |
| Leuprolide acetate | Lupron, Eligard |
Ergots and ergotamines have varied safety profiles in pregnancy
Dihydroergotamine belongs to the oldest family of migraine-specific drugs, although it is not widely used today. One reason may be that most migraineurs are women of reproductive age, and ergots are oxytocic and potential teratogens. Furthermore, they are not recommended in lactation. Ergotamines, however, are quite diverse, and some have more acceptable FDA use-in-pregnancy ratings. Injectable dihydroergotamine appears to be as effective as or less effective than triptans for migraine pain, but more effective than other drugs, such as NSAIDs or analgesics, for acute attacks.9 The nasal spray is more effective than placebo, but less effective than triptans.9
Triptans are relatively safe for women of reproductive age
Most information on use of triptans in pregnancy concerns sumatriptan. Like all triptans, it falls into Category C; however, its published profile is reassuring.10-12 Data from Sweden on 2,027 first-trimester exposures show a 3.6% risk of birth defects (major and minor), compared with an identical 3.6% risk in the general Swedish population.12 Sumatriptan is considerably less lipophilic than most of the drugs in its class and has been rated as compatible with breastfeeding.13
All triptans except eletriptan have been studied in the treatment of acute MM and found to be superior to placebo.14 These serotonin 1B/1D agonists are tailored therapy for acute migraine because, unlike analgesics, they address the underlying pathology of the attack, inhibiting the release of vasoactive peptides, promoting selective meningeal vasoconstriction, and blocking pain pathways in the brainstem. They also resolve the associated symptoms of nausea and photophobia. Triptans should not be used in the presence of untreated hypertension or vascular disease (cardiovascular, cerebrovascular, or peripheral vascular disease).
Watch for “rebound” headache. Like all acute treatment—be it a simple analgesic, NSAID, caffeine-containing compound, butalbital, ergotamine, or opioid—too-frequent use of triptans can produce medication-overuse headache, also referred to as rebound headache. In general, try to limit the use of agents for acute migraine, whether prescription or over-the-counter, to no more than 2 days a week to avoid this consequence. Also, be aware that some agents—notably, butalbital-containing compounds—may cause rebound headache, even when given as infrequently as 5 days of the month.15
Some preventive strategies are “nonspecific”
When treatment of acute MM is inadequate, management shifts toward prevention. Options include nonspecific (those that do not address the hormonal trigger) and specific (hormonal) strategies. Nonspecific strategies rely on the predictable onset of MM for proper timing of the therapeutic intervention; therefore, women who have irregular cycles are not good candidates for this approach. The presence of comorbid conditions such as dysmenorrhea, menometrorrhagia, and endometriosis may argue for early adoption of specific strategies that improve both conditions at once.
The fact that menstrual migraine (MM) is associated with estrogen withdrawal prompts a question: What happens immediately postpartum, when estrogen levels decline with loss of the placenta?
Although hormonal fluctuations generally stabilize during pregnancy, and most menstrual migraineurs experience fewer headaches during gestation, that protective effect erodes at delivery, when the incidence of migraine can be as high as 40% in the first week.41
Investigators who prospectively studied 49 migraineurs—two of whom were affected by migraine with aura and 47 by migraine without aura—found improvement in 46.8% of these women during the first trimester of pregnancy, 83% during the second trimester, and 87.2% during the third trimester, with complete remission rates of 10.6%, 53.2%, and 78.7%, respectively.42 During the first postpartum week, migraine recurred in 34% of women.42 Interestingly, women who had a history of MM were less likely to improve during the first and third trimesters of pregnancy.42
In a separate prospective investigation of 985 women who delivered over a 3-month period in one tertiary-care facility, 381 experienced postpartum headache.43 The median time to onset of the headache was 2 days, and the median duration was 4 hours. More than 75% of these headaches were primary headaches.43 Only a small percentage (4%) were incapacitating.43
When migraine may signal a more serious condition
Another question arises when a woman experiences a severe headache shortly after delivery: Could the headache be related to preeclampsia or another serious complication of pregnancy and delivery? According to Contag and colleagues, immediate assessment may be warranted.44
“Characteristics to consider are the association of the headache with elevated blood pressure (which could signal postpartum preeclampsia), the sudden onset of an atypical headache, and variations to the usual nature of the migraine, such as the onset of new neurological symptoms. Postpartum women with any of these characteristics should be evaluated in the emergency department, and neuroimaging should be strongly considered,” they write.44
Treatment reverts to prepregnancy options
Assuming preeclampsia or another serious condition is not the culprit, the treatment options for postpartum migraine revert to prepregnancy choices, as effect on the developing fetus is no longer a concern. Nonsteroidal anti-inflammatory drugs (NSAIDs) are the first line of therapy. If they are ineffective, proceed to an ergotamine or triptan.—Janelle Yates, Senior Editor
Nonhormonal (nonspecific) therapy NSAIDs may provide some relief. In one small study, naproxen sodium (550 mg twice daily) was administered for 2 weeks, beginning 1 week before the anticipated onset of menses. It modestly reduced the overall duration and severity of menstrual migraine16—a benefit that must be weighed against the risk of adverse events and the more recent FDA black box warning about its potentially serious heart and gastrointestinal risks.
Triptan regimens may prevent MM. Several triptans have been investigated for prevention of MM. A small, open-label study evaluated the use of oral sumatriptan (25 mg three times daily), beginning 2 to 3 days before the anticipated onset of MM and continuing for 5 days.17 Menstrual attacks were prevented in just over 50% of cases.
Naratriptan (1 mg twice daily), beginning 2 days before anticipated menses and continuing for 5 days, reduced the number of MM attacks by 50%, with side effects comparable to placebo.18 A higher dosage (2.5 mg twice daily) did not prove to be superior to placebo, for unexplained reasons.
In a similar trial, frovatriptan (2.5 mg daily or 2.5 mg twice daily), beginning 2 days before the anticipated onset of MM and continuing for 6 days, prevented at least 50% of MM.19 Twice-daily dosing was superior to daily administration.
Magnesium may shorten MM. In one small study, oral magnesium (360 mg daily), beginning on the 15th day of the cycle and continuing through the menses, shortened the duration of MM and improved menstrual complaints better than placebo.20
The goal of hormonal therapy is to eliminate or sufficiently minimize the premenstrual decline in estrogen that is believed to precipitate MM.21 An observational study of 229 women found that hormonal strategies prevented MM in 73% of cases (81% when taken as directed).22
It is fortunate that MM, by definition, is migraine without aura, because the use of combined OCs has been controversial in the setting of migraine with aura. International studies have reported a small but increased risk of stroke associated with their use, although a subset of women with migraine had no increased risk.23-25 In contrast, no U.S. study since 1975 has found an increased risk of stroke associated with the use of OCs.26 One large domestic study reviewed 3.6 million woman-years of use and found no increased risk of ischemic stroke with the low-dose OCs currently available, nor did a pooled analysis of U.S. studies.27,28
The discrepancy is likely explained by the strong relative contraindication, in the United States, to the use of OCs in smokers older than 35 years (the smoking prevalence in most of the European case series was more than 50%), as well as the more prevalent use of high-dose OCs in the international studies. High-dose pills were implicated in the majority of stroke cases in the World Health Organization (WHO) study, but were used by only 0.7% of cases and controls in the pooled U.S. studies.23 Nevertheless, both ACOG and WHO concluded that the risk of OC use usually outweighs the benefit in women older than 35 years whose migraines are complicated by focal neurologic deficits.
Extended-cycle OCs may offer a lengthy reprieve. Regimens that forego monthly withdrawal bleeds and provide extended administration of active pills can afford migraineurs a lengthy reprieve from MM.29 Breakthrough bleeding is the most common side effect but tends to decrease over time. It is preferable for the patient to take the pill at bedtime to avoid a drug nadir during susceptible stages of sleep, which may be associated with migraine generation. It also is prudent to avoid concomitant administration of drugs that might increase the rate of hepatic metabolism of estrogen (e.g., a high dosage of topiramate) and lead to a more rapid decline in concentration.
When using an extended-cycle regimen that allows for periodic withdrawal bleeds, give the patient supplemental estrogen during the withdrawal week if the decline in ethinyl estradiol (EE) exceeds 10 μg, a threshold that appears to elicit MM in susceptible women.21,22 For example, with an extended-cycle regimen that contains 30 μg of EE, it may be necessary to add 20 μg of estrogen during the placebo week to prevent MM, whereas an extended-cycle regimen that contains 20 μg EE, declining to 10 μg during the 13th week, would be adequate as packaged.
21/7 hormonal OCs may require supplemental estrogen to prevent MM. The late luteal-phase decline in estradiol concentration during the natural menstrual cycle is equivalent to the decline experienced with the transition to inert pills in 20- to 25-μg EE formulations. Therefore, these regimens confer the same risk of MM as the woman’s natural cycle. Products that contain incrementally higher dosages of EE (30, 35, and 50 μg) confer an increasingly greater risk of MM. Supplementation of estrogen during the placebo week may prevent MM by limiting the decline in estrogen.
In a small, open-label study, women took an OC containing 20 μg EE at bedtime on days 1 to 21 of the cycle, followed by 0.9 mg of conjugated equine estrogens (CEE) on days 22 to 28. All patients reported a reduction in migraine frequency of at least 50%, with a mean reduction of 78%.30
Parenteral options can be created utilizing a transdermal 20-μg EE/norelgestromin patch or a 15-μg EE/etonogestrel vaginal ring. With the first approach, I recommend adding a 0.1-mg EE patch during the withdrawal week to prevent MM. With the latter, a 0.075-mg EE patch may be used during the week following ring removal.31
Consider a menstrually targeted estrogen supplement. Some women who have contraindications to use of an OC may still be candidates for targeted strategies using lower dosages of supplemental estrogen rather than a combination OC. Among the options is perimenstrual administration of an estradiol patch or gel.32 One study found a 0.1-mg estrogen patch (worn 7 days and applied just before the expected onset of menses) to be effective, but lower dosages (0.025 to 0.05 mg EE) were not.33 Daily application of 1.5 mg estradiol gel for 7 days, beginning before the onset of MM, was also effective in preventing MM.34-36
With targeted strategies, timing is critical; if estrogen is begun too early, the incidence of migraine may rise after cessation.36
GnRH agonists may benefit women who have isolated MM. Administration of gonadotropin-releasing hormone (GnRH) agonists has been shown to ease MM, but the baseline frequency of headaches appears to be an important variable in the success of these agents.37-39 Leuprolide acetate markedly diminished MM in women who seldom experienced headaches other than their menstrual attacks.37 It did not benefit women who experienced migraine in the setting of chronic headache (≥15 days/month), probably because chronic migraine is influenced by other variables, such as nonrestorative sleep and overuse of medication.15,38,40
No evidence that progestin-only contraceptives are effective. Although these agents have been proposed for treatment of migraine, evidence of their benefit is lacking. It is my opinion that, more often than not, they exacerbate migraine.
1. MacGregor EA, Hackshaw A. Prevalence of migraine on each day of the natural menstrual cycle. Neurology. 2004;63(2):351-353.
2. Granella F, Sances G, Allais G, et al. Characteristics of menstrual and nonmenstrual attacks in women with menstrually related migraine referred to headache centres. Cephalalgia. 2004;24(9):707-716.
3. Launer LJ, Terwindt GM, Ferrari MD. The prevalence and characteristics of migraine in a population-based cohort: the GEM study. Neurology. 1999;53(3):537-542.
4. Couturier EG, Bomhof MA, Neven AK, van Duijn NP. Menstrual migraine in a representative Dutch population sample: prevalence, disability and treatment. Cephalalgia. 2003;23(4):302-308.
5. Headache Classification Subcommittee of the International Headache Society. The International Classification of Headache Disorders. 2nd ed. Cephalalgia. 2004;24(suppl 1):9-160.
6. Calhoun AH, Ford S, Millen C, Finkel AG, Truong Y, Nie Y. The prevalence of neck pain in migraine [abstract]. Headache. 2010 Jan 20.-[Epub ahead of print].
7. Al-Waili NS. Treatment of menstrual migraine with prostaglandin synthesis inhibitor mefenamic acid: double-blind study with placebo. Eur J Med Res. 2000;5(4):176-182.
8. Diener HC, Montagna P, Gács G, et al. Efficacy and tolerability of diclofenac potassium sachets in migraine: a randomized, double-blind, cross-over study in comparison with diclofenac potassium tablets and placebo. Cephalalgia. 2006;26(5):537-547.
9. Schürks M. Dihydroergotamine: role in the treatment of migraine. Expert Opin Drug Metab Toxicol. 2009;5(9):1141-1148.
10. Loder E. Safety of sumatriptan in pregnancy: a review of the data so far. CNS Drugs. 2003;17(1):1-7.
11. Shuhaiber S, Pastuszak A, Schick B, et al. Pregnancy outcome following first trimester exposure to sumatriptan. Neurology. 1998;51(2):581-583.
12. Cunnington M, Ephross S, Churchill P. The safety of sumatriptan and naratriptan in pregnancy: what have we learned? Headache. 2009;49(10):1414-1422.
13. Ressel G. AAP updates statement for transfer of drugs and other chemicals into breast milk. American Academy of Pediatrics. Am Fam Physician. 2002;65(5):979-980.
14. Ashkenazi A, Silberstein S. Menstrual migraine: a review of hormonal causes, prophylaxis and treatment. Expert Opin Pharmacother. 2007;8(11):1605-1613.
15. Bigal ME, Serrano D, Buse D, Scher A, Stewart WF, Lipton RB. Acute migraine medications and evolution from episodic to chronic migraine: a longitudinal population-based study. Headache. 2008;48(8):1157-1168.
16. Sances G, Martignoni E, Fioroni L, Blandini F, Facchinetti F, Nappi G. Naproxen sodium in menstrual migraine prophylaxis: a double-blind placebo controlled study. Headache. 1990;30(11):705-709.
17. Newman LC, Lipton RB, Lay CL, Solomon S. A pilot study of oral sumatriptan as intermittent prophylaxis of menstruation-related migraine. Neurology. 1998;51(1):307-309.
18. Newman L, Mannix LK, Landy S, et al. Naratriptan as short-term prophylaxis of menstrually associated migraine: a randomized, double-blind, placebo-controlled study. Headache. 2001;41(3):248-256.
19. Silberstein SD, Elkind AH, Schreiber C, Keywood C. A randomized trial of frovatriptan for the intermittent prevention of menstrual migraine. Neurology. 2004;63(2):261-269.
20. Facchinetti F, Sances G, Borella P, Genazzani AR, Nappi G. Magnesium prophylaxis of menstrual migraine: effects on intracellular magnesium. Headache. 1991;31(5):298-301.
21. Calhoun A. Women’s Issues in Headache. In: Loder E, ed. Headache. Philadelphia: American College of Physicians; 2004:157-177.
22. Calhoun A, Ford S. Elimination of menstrual-related migraine beneficially impacts chronification and medication overuse. Headache. 2008;48(8):1186-1193.
23. Ischaemic stroke and combined oral contraceptives: results of an international, multicentre, case-control study. WHO Collaborative Study of Cardiovascular Disease and Steroid Hormone Contraception. Lancet. 1996;348:498-505.
24. Chan WS, Ray J, Wai EK, et al. Risk of stroke in women exposed to low-dose oral contraceptives: a critical evaluation of the evidence. Arch Intern Med. 2004;164(7):741-747.
25. Etminan M, Takkouche B, Isorna FC, Samii A. Risk of ischaemic stroke in people with migraine: systematic review and meta-analysis of observational studies. BMJ. 2005;330(7482):63.-
26. Oral contraceptives and stroke in young women. Associated risk factors. JAMA. 1975;231:718-722.
27. Petitti DB, Sidney S, Bernstein A, Wolf S, Quesenberry C, Ziel HK. Stroke in users of low-dose oral contraceptives. N Engl J Med. 1996;335(1):8-15.
28. Schwartz SM, Petitti DB, Siscovick DS, et al. Stroke and use of low-dose oral contraceptives in young women: a pooled analysis of two US studies. Stroke. 1998;29(11):2277-2284.
29. Sulak PJ, Scow RD, Preece C, Riggs MW, Kuehl TJ. Hormone withdrawal symptoms in oral contraceptive users. Obstet Gynecol. 2000;95(2):261-266.
30. Calhoun AH. A novel specific prophylaxis for menstrual-associated migraine. South Med J. 2004;97(9):819-822.
31. Calhoun AH, Hutchinson S. Hormonal therapies for menstrual migraine. Curr Pain Headache Rep. 2009;13(5):381-385.
32. Pringsheim T, Davenport WJ, Dodick D. Acute treatment and prevention of menstrually related migraine headache: evidence-based review. Neurology. 2008;70(17):1555-1563.
33. Pradalier A VD, Beaulieu PH, et al. Correlation between oestradiol plasma level and therapeutic effect on menstrual migraine. In: FC Rose, ed. New advances in headache research. London: Smith-Gordon; 1994:129-132.
34. de Lignières B, Vincens M, Mauvais-Jarvis P, Mas JL, Touboul PJ, Bousser MG. Prevention of menstrual migraine by percutaneous oestradiol. Br Med J (Clin Res Ed). 1986;293(6561):1540.-
35. Dennerstein L, Morse C, Burrows G, Oats J, Brown J, Smith M. Menstrual migraine: a double-blind trial of percutaneous estradiol. Gynecol Endocrinol. 1988;2(2):113-120.
36. MacGregor EA, Frith A, Ellis J, Aspinall L, Hackshaw A. Prevention of menstrual attacks of migraine: a double-blind placebo-controlled crossover study. Neurology. 2006;67(12):2159-2163.
37. Murray SC, Muse KN. Effective treatment of severe menstrual migraine headaches with gonadotropin-releasing hormone agonist and “add-back” therapy. Fertil Steril. 1997;67(2):390-393.
38. Martin V, Wernke S, Mandell K, et al. Medical oophorectomy with and without estrogen add-back therapy in the prevention of migraine headache. Headache. 2003;43(4):309-321.
39. Lichten EM, Lichten JB, Whitty A, Pieper D. The confirmation of a biochemical marker for women’s hormonal migraine: the depo-estradiol challenge test. Headache. 1996;36(6):367-371.
40. Calhoun AH, Ford S. Behavioral sleep modification may revert transformed migraine to episodic migraine. Headache. 2007;47(8):1178-1183.
41. Stein GS. Headaches in the first postpartum week and their relationship to migraine. Headache. 1981;21(5):201-205.
42. Sances G, Granella F, Nappi RE, et al. Course of migraine during pregnancy and postpartum: a prospective study. Cephalalgia. 2003;23(3):197-205.
43. Goldszmidt E, Kern R, Chaput A, Macarthur A. The incidence and etiology of postpartum headaches: a prospective cohort study. Can J Anaesth. 2005;52(9):971-977.
44. Contag SA, Mertz HL, Bushnell CD. Migraine during pregnancy: Is it more than a headache? Nat Rev Neurol. 2009;5(8):449-456.
1. MacGregor EA, Hackshaw A. Prevalence of migraine on each day of the natural menstrual cycle. Neurology. 2004;63(2):351-353.
2. Granella F, Sances G, Allais G, et al. Characteristics of menstrual and nonmenstrual attacks in women with menstrually related migraine referred to headache centres. Cephalalgia. 2004;24(9):707-716.
3. Launer LJ, Terwindt GM, Ferrari MD. The prevalence and characteristics of migraine in a population-based cohort: the GEM study. Neurology. 1999;53(3):537-542.
4. Couturier EG, Bomhof MA, Neven AK, van Duijn NP. Menstrual migraine in a representative Dutch population sample: prevalence, disability and treatment. Cephalalgia. 2003;23(4):302-308.
5. Headache Classification Subcommittee of the International Headache Society. The International Classification of Headache Disorders. 2nd ed. Cephalalgia. 2004;24(suppl 1):9-160.
6. Calhoun AH, Ford S, Millen C, Finkel AG, Truong Y, Nie Y. The prevalence of neck pain in migraine [abstract]. Headache. 2010 Jan 20.-[Epub ahead of print].
7. Al-Waili NS. Treatment of menstrual migraine with prostaglandin synthesis inhibitor mefenamic acid: double-blind study with placebo. Eur J Med Res. 2000;5(4):176-182.
8. Diener HC, Montagna P, Gács G, et al. Efficacy and tolerability of diclofenac potassium sachets in migraine: a randomized, double-blind, cross-over study in comparison with diclofenac potassium tablets and placebo. Cephalalgia. 2006;26(5):537-547.
9. Schürks M. Dihydroergotamine: role in the treatment of migraine. Expert Opin Drug Metab Toxicol. 2009;5(9):1141-1148.
10. Loder E. Safety of sumatriptan in pregnancy: a review of the data so far. CNS Drugs. 2003;17(1):1-7.
11. Shuhaiber S, Pastuszak A, Schick B, et al. Pregnancy outcome following first trimester exposure to sumatriptan. Neurology. 1998;51(2):581-583.
12. Cunnington M, Ephross S, Churchill P. The safety of sumatriptan and naratriptan in pregnancy: what have we learned? Headache. 2009;49(10):1414-1422.
13. Ressel G. AAP updates statement for transfer of drugs and other chemicals into breast milk. American Academy of Pediatrics. Am Fam Physician. 2002;65(5):979-980.
14. Ashkenazi A, Silberstein S. Menstrual migraine: a review of hormonal causes, prophylaxis and treatment. Expert Opin Pharmacother. 2007;8(11):1605-1613.
15. Bigal ME, Serrano D, Buse D, Scher A, Stewart WF, Lipton RB. Acute migraine medications and evolution from episodic to chronic migraine: a longitudinal population-based study. Headache. 2008;48(8):1157-1168.
16. Sances G, Martignoni E, Fioroni L, Blandini F, Facchinetti F, Nappi G. Naproxen sodium in menstrual migraine prophylaxis: a double-blind placebo controlled study. Headache. 1990;30(11):705-709.
17. Newman LC, Lipton RB, Lay CL, Solomon S. A pilot study of oral sumatriptan as intermittent prophylaxis of menstruation-related migraine. Neurology. 1998;51(1):307-309.
18. Newman L, Mannix LK, Landy S, et al. Naratriptan as short-term prophylaxis of menstrually associated migraine: a randomized, double-blind, placebo-controlled study. Headache. 2001;41(3):248-256.
19. Silberstein SD, Elkind AH, Schreiber C, Keywood C. A randomized trial of frovatriptan for the intermittent prevention of menstrual migraine. Neurology. 2004;63(2):261-269.
20. Facchinetti F, Sances G, Borella P, Genazzani AR, Nappi G. Magnesium prophylaxis of menstrual migraine: effects on intracellular magnesium. Headache. 1991;31(5):298-301.
21. Calhoun A. Women’s Issues in Headache. In: Loder E, ed. Headache. Philadelphia: American College of Physicians; 2004:157-177.
22. Calhoun A, Ford S. Elimination of menstrual-related migraine beneficially impacts chronification and medication overuse. Headache. 2008;48(8):1186-1193.
23. Ischaemic stroke and combined oral contraceptives: results of an international, multicentre, case-control study. WHO Collaborative Study of Cardiovascular Disease and Steroid Hormone Contraception. Lancet. 1996;348:498-505.
24. Chan WS, Ray J, Wai EK, et al. Risk of stroke in women exposed to low-dose oral contraceptives: a critical evaluation of the evidence. Arch Intern Med. 2004;164(7):741-747.
25. Etminan M, Takkouche B, Isorna FC, Samii A. Risk of ischaemic stroke in people with migraine: systematic review and meta-analysis of observational studies. BMJ. 2005;330(7482):63.-
26. Oral contraceptives and stroke in young women. Associated risk factors. JAMA. 1975;231:718-722.
27. Petitti DB, Sidney S, Bernstein A, Wolf S, Quesenberry C, Ziel HK. Stroke in users of low-dose oral contraceptives. N Engl J Med. 1996;335(1):8-15.
28. Schwartz SM, Petitti DB, Siscovick DS, et al. Stroke and use of low-dose oral contraceptives in young women: a pooled analysis of two US studies. Stroke. 1998;29(11):2277-2284.
29. Sulak PJ, Scow RD, Preece C, Riggs MW, Kuehl TJ. Hormone withdrawal symptoms in oral contraceptive users. Obstet Gynecol. 2000;95(2):261-266.
30. Calhoun AH. A novel specific prophylaxis for menstrual-associated migraine. South Med J. 2004;97(9):819-822.
31. Calhoun AH, Hutchinson S. Hormonal therapies for menstrual migraine. Curr Pain Headache Rep. 2009;13(5):381-385.
32. Pringsheim T, Davenport WJ, Dodick D. Acute treatment and prevention of menstrually related migraine headache: evidence-based review. Neurology. 2008;70(17):1555-1563.
33. Pradalier A VD, Beaulieu PH, et al. Correlation between oestradiol plasma level and therapeutic effect on menstrual migraine. In: FC Rose, ed. New advances in headache research. London: Smith-Gordon; 1994:129-132.
34. de Lignières B, Vincens M, Mauvais-Jarvis P, Mas JL, Touboul PJ, Bousser MG. Prevention of menstrual migraine by percutaneous oestradiol. Br Med J (Clin Res Ed). 1986;293(6561):1540.-
35. Dennerstein L, Morse C, Burrows G, Oats J, Brown J, Smith M. Menstrual migraine: a double-blind trial of percutaneous estradiol. Gynecol Endocrinol. 1988;2(2):113-120.
36. MacGregor EA, Frith A, Ellis J, Aspinall L, Hackshaw A. Prevention of menstrual attacks of migraine: a double-blind placebo-controlled crossover study. Neurology. 2006;67(12):2159-2163.
37. Murray SC, Muse KN. Effective treatment of severe menstrual migraine headaches with gonadotropin-releasing hormone agonist and “add-back” therapy. Fertil Steril. 1997;67(2):390-393.
38. Martin V, Wernke S, Mandell K, et al. Medical oophorectomy with and without estrogen add-back therapy in the prevention of migraine headache. Headache. 2003;43(4):309-321.
39. Lichten EM, Lichten JB, Whitty A, Pieper D. The confirmation of a biochemical marker for women’s hormonal migraine: the depo-estradiol challenge test. Headache. 1996;36(6):367-371.
40. Calhoun AH, Ford S. Behavioral sleep modification may revert transformed migraine to episodic migraine. Headache. 2007;47(8):1178-1183.
41. Stein GS. Headaches in the first postpartum week and their relationship to migraine. Headache. 1981;21(5):201-205.
42. Sances G, Granella F, Nappi RE, et al. Course of migraine during pregnancy and postpartum: a prospective study. Cephalalgia. 2003;23(3):197-205.
43. Goldszmidt E, Kern R, Chaput A, Macarthur A. The incidence and etiology of postpartum headaches: a prospective cohort study. Can J Anaesth. 2005;52(9):971-977.
44. Contag SA, Mertz HL, Bushnell CD. Migraine during pregnancy: Is it more than a headache? Nat Rev Neurol. 2009;5(8):449-456.
