User login
2019 Update on contraception
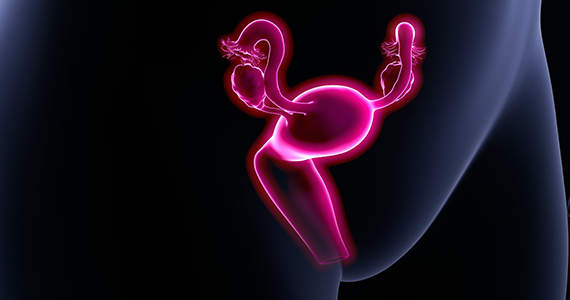
Long-acting reversible contraception (LARC) use continues to increase in the United States. According to the most recent estimates from 2014, 14% of women use either an intrauterine device (IUD) or the etonogestrel implant.1 Forms of LARC currently available in the United States include:
- 4 hormone-releasing IUDs
- 1 nonhormonal copper IUD, and
- 1 hormonal subdermal implant.
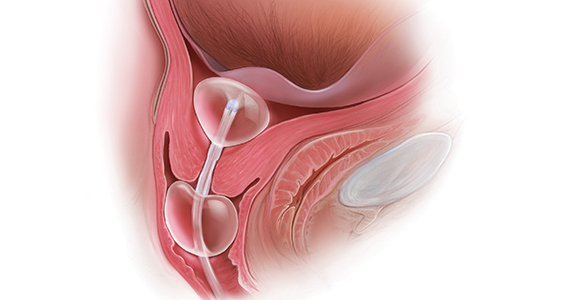
The hormone-releasing IUDs all contain levonorgestrel (LNG). These include two 52-mg LNG products and a 19.5-mg LNG IUD, which are currently approved by the US Food and Drug Administration (FDA) for contraception for 5 continuous years of use. In addition, a 13.5-mg LNG IUD is FDA-approved for 3 years of use. The hormonal subdermal implant, which contains etonogestrel, is FDA-approved for 3 years of use. Although major complications with IUDs (perforation, expulsion, intrauterine infection)and implants (subfascial implantation, distant migration) are rare, adverse effects that can affect continuation—such as irregular bleeding—are more common.2,3
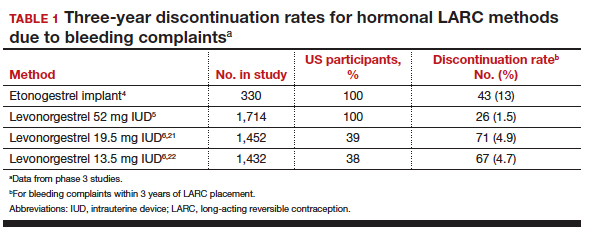
Contraceptive discontinuation due to bleeding concerns occurs more frequently with the etonogestrel implant than with LNG IUDs (TABLE 1). In a large prospective study in the United States, 13% of women discontinued the implant during 3 years of follow-up due to bleeding pattern changes.
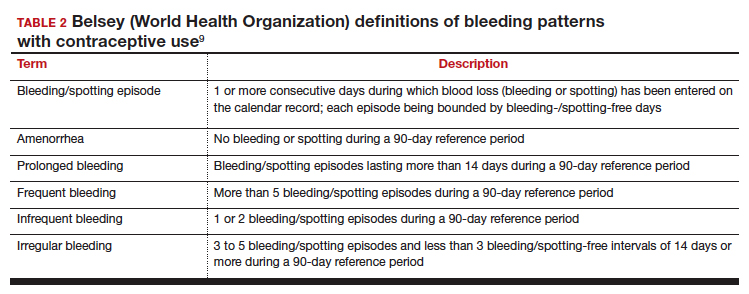
Notably, it is important to use standardized definitions to understand and compare bleeding concerns with LARC use. The Belsey criteria of the World Health Organization (WHO), a standard used for decades, describe bleeding patterns using 90-day reference periods or intervals (TABLE 2).9 Bleeding patterns that decrease flow (amenorrhea, infrequent bleeding) often are considered favorable, and those that increase bleeding or irregularity often are considered unfavorable. These criteria are commonly used in package labeling to describe bleeding patterns with extended use.
In this Update, we examine recent data evaluating differences in bleeding patterns with the 3 doses of the LNG IUD, predictors of abnormal bleeding with the etonogestrel implant, and the impact of timing on postpartum etonogestrel implant placement.
Continue to: Bleeding patterns with progestin-containing IUDs vary according to the LNG dose...
Bleeding patterns with progestin-containing IUDs vary according to the LNG dose
Goldthwaite LM, Creinin MD. Comparing bleeding patterns for the levonorgestrel 52 mg, 19.5 mg, and 13.5 mg intrauterine systems. Contraception. 2019;100:128-131.
Counseling on IUDs' different hormonal doses requires an understanding of patients' desires for contraceptive efficacy and bleeding expectations. A recent study provides guidance on what patients typically can expect for their bleeding patterns over the first few years with the 3 different doses of LNG IUDs.
Goldthwaite and Creinin used existing published or publicly available data to analyze differences in bleeding patterns associated with the 52-mg, 19.5-mg, and 13.5-mg LNG IUDs. Although two 52-mg LNG IUDs are available, published data using the WHO Belsey criteria are available only for one (Liletta; Allergan, Medicines360). The 2 products have been shown previously to have similar drug-release rates and LNG levels over 5 years.8
Comparing favorable bleeding patterns: Amenorrhea and infrequent bleeding
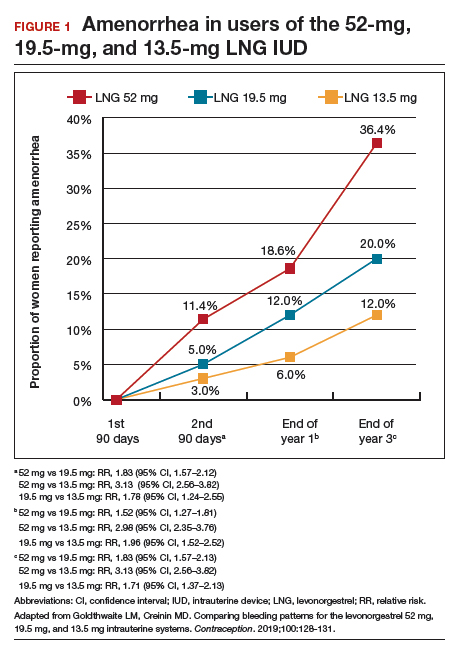
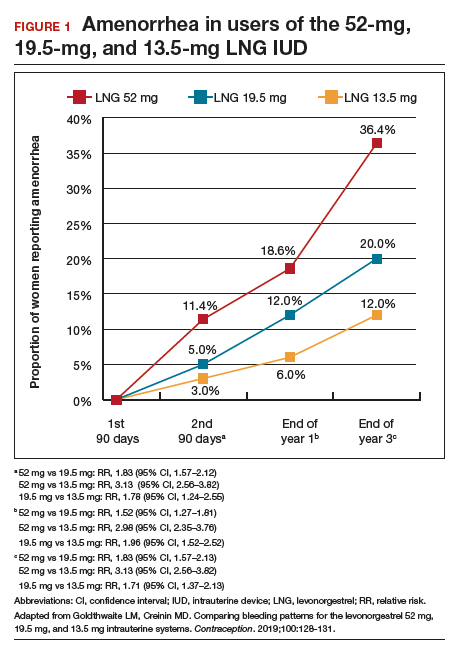
Among favorable bleeding patterns, amenorrhea was uncommon in the first 90 days and increased over time for all 3 IUDs. However, starting as soon as the second 90-day reference period, amenorrhea rates were significantly higher with the 52-mg LNG IUD compared with both of the lower-LNG dose IUDs, and this difference increased through 3 years of use (FIGURE 1).
Similarly, the 19.5-mg LNG IUD users had significantly higher rates of amenorrhea than the 13.5-mg LNG IUD users for all periods starting with the second 90-day reference period. At 3 years, 36% of women using the 52-mg LNG IUD had amenorrhea compared with 20% of those using the 19.5-mg LNG IUD (P<.0001) and 12% of those using the 13.5-mg LNG IUD (P<.0001).
Infrequent bleeding was similar for all 3 LNG IUDs in the first 90-day period, and it then increased most rapidly in the 52-mg LNG IUD users. At the end of year 1, 30% of the 52-mg LNG IUD users had infrequent bleeding compared with 26% of the 19.5-mg users (P = .01) and 20% of the 13.5-mg users (P<.0001). Although there was no difference in infrequent bleeding rates between the 52-mg and the 19.5-mg LNG IUD users at the end of year 1, those using a 52-mg LNG IUD had significantly higher rates of infrequent bleeding compared with the 13.5-mg LNG IUD at all time points.
Comparing unfavorable bleeding patterns: Frequent, prolonged, and irregular bleeding
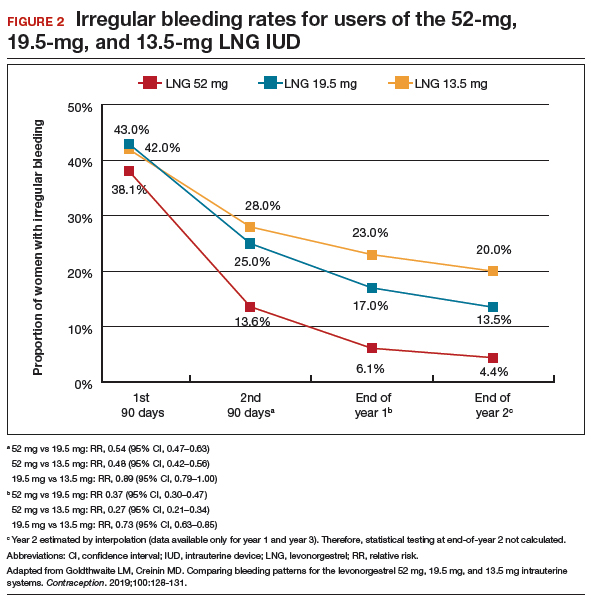
Frequent and prolonged bleeding were uncommon with all LNG doses. Irregular bleeding rates declined for users of the 3 IUDs over time. However, significantly fewer users of the 52-mg LNG IUD reported irregular bleeding at 1 year (6%) compared with users of the 19.5-mg (16.5%, P<.0001) and 13.5-mg (23%, P<.0001) LNG IUD (FIGURE 2).
Study limitations
Comparing the data from different studies has limitations. For example, the data were collected from different populations, with the lower-dose LNG products tested in women who had a lower body mass index (BMI) and higher parity. However, prior analysis of the data on the 52-mg LNG IUD demonstrated that bleeding pattern changes did not vary based on these factors.10
When considering the different progestin-based IUD options, it is important to counsel patients according to their preferences for potential adverse effects. A randomized trial during product development found no difference in systemic adverse effects with the 3 doses of LNG IUD, likely because the systemic hormone levels are incredibly low for all 3 products.11 The summary data in this report helps explain why women using the lower-dose LNG products have slightly higher discontinuation rates for bleeding complaints, a fact we can explain to our patients during counseling.
Overall, the 52-mg LNG IUD is associated with a higher likelihood of favorable bleeding patterns over the first few years of use, with higher rates of amenorrhea and infrequent bleeding and lower rates of irregular bleeding. For women who prefer to not have periods or to have infrequent periods, the 52-mg LNG IUD is most likely to provide that outcome. For a patient who prefers to have periods, there is no evidence that the lower-dose IUDs result in “regular” or “normal” menstrual bleeding, even though they do result in more bleeding/spotting days overall. To the contrary, the available data show that these women have a significantly higher likelihood of experiencing prolonged, frequent, and irregular bleeding. In fact, no studies have reported rates of “normal” bleeding with the progestin IUDs, likely because women uncommonly have “normal” bleeding with these contraception methods. If a patient does not desire amenorrhea or strongly prefers to have “regular bleeding,” alternative methods such as a copper IUD should be considered rather than counseling her toward a lower-dose progestin IUD.
Continue to: Predicting long-term bleeding patterns after etonogestrel implant insertion...
Predicting long-term bleeding patterns after etonogestrel implant insertion
Mansour D, Fraser IS, Edelman A, et al. Can initial vaginal bleeding patterns in etonogestrel implant users predict subsequent bleeding in the first two years of use? Contraception. 2019. doi: 10.1016/j.contraception.2019.05.017.
Data from 2014 indicate that the etonogestrel implant was used by nearly 1 million women in the United States and by 3% of women using contraception.1 The primary reason women discontinue implant use is because of changes in bleeding patterns. Given the high prevalence of bleeding concerns with the etonogestrel implant, we need more data to help counsel our patients on how they can expect their bleeding to change with implant use.
Etonogestral implant and bleeding pattern trends
Mansour and colleagues completed a secondary analysis of 12 phase 3 studies to evaluate the correlation between bleeding patterns early after placement of the etonogestrel implant (days 29-118) compared with bleeding patterns through 90-day intervals during the rest of the first year of use. To account for differences in timing of etonogestrel implant placement relative to the menstrual cycle and discontinuation of other methods like oral contraceptives, bleeding outcomes on days 0-28 were excluded. They also sought to investigate the correlation between bleeding patterns in year 1 compared with those in year 2.
Overall, these studies included 923 individuals across 11 countries; however, for the current analysis, the researchers excluded women from Asian countries who comprised more than 28% of the study population. These women report significantly fewer bleeding/spotting days with the etonogestrel implant and have a lower average body weight compared with European and American women.12
A prior analysis of the same data set looked at the number of bleeding/spotting days in groups of users rather than trends in individual patients, and, as mentioned, it also included Asian women, which diluted the overall number of bleeding days.12 In this new analysis, Mansour and colleagues used the Belsey criteria to analyze individual bleeding patterns as favorable (amenorrhea, infrequent bleeding, normal bleeding) or unfavorable (prolonged and/or frequent bleeding) from a patient perspective. In this way, we can understand trends in bleeding patterns for each patient over time, rather than seeing a static (cross-sectional) report of bleeding patterns at one point in time. Data were analyzed from 537 women in year 1 and 428 women in year 2. During the first 90-day reference period (days 29-118 after implant insertion), 61% of women reported favorable bleeding, and 39% reported unfavorable bleeding.
Favorable bleeding correlates with favorable patterns later
A favorable bleeding pattern in this first reference period correlated with favorable bleeding patterns through year 1, with 85%, 80%, and 80% of these women having a favorable pattern in reference periods 2, 3, and 4, respectively. Overall, 61% of women with a favorable pattern in reference period 1 had favorable bleeding throughout the entire first year of use. Only 3.7% of women with favorable bleeding in the first reference period discontinued the implant for bleeding in year 1. Further, women with favorable bleeding at year 1 commonly continued to have favorable bleeding in year 2, with a low discontinuation rate (2.5%) in year 2.
Individual patients who have a favorable bleeding pattern initially with etonogestrel implant placement are highly likely to continue having favorable bleeding at year 1 and year 2. Notably, of women with a favorable bleeding pattern in any 90-day reference period, about 80% will continue to have a favorable bleeding pattern in the next reference period. These women can be counseled that, even if they have a 90-day period with unfavorable bleeding, about two-thirds will have a favorable pattern in the next reference period. For those with initial unfavorable patterns, about one-third to one-half change to a favorable pattern in subsequent 90-day reference periods. For women who require intervention for unfavorable bleeding but wish to keep their etonogestrel implant, prior data support use of combined oral contraceptive pills, although bleeding resolution seems to be temporary, with 86% of women having bleeding recurrence within 10 days after treatment.13
Initial unfavorable bleeding portends less favorable patterns later
Women who had an unfavorable bleeding pattern initially, however, had a less predictable course over the first year. For those with an initial unfavorable pattern, only 37%, 47%, and 51% reported a favorable pattern in reference periods 2, 3, and 4. Despite these relatively low rates of favorable bleeding, only 13% of the women with an initial unfavorable bleeding pattern discontinued implant use for a bleeding complaint by the end of year 1; this rate was significantly higher than that for women with a favorable initial bleeding pattern (P<.0001). The discontinuation rate for bleeding complaints also remained higher in year 2, at 16.5%.
Limitations and strengths to consider
Although the etonogestrel implant is FDA-approved for 3 years of use, the bleeding data from the combined trials included information for only up to 2 years after placement. The studies included also did not uniformly assess BMI, which makes it difficult to find correlations between bleeding patterns and BMI. Importantly, the studies did not include women who were more than 30% above their ideal body weight, so these assessments do not apply to obese users.12 Exclusion of women from Southeast Asia in this analysis makes this study's findings more generalizable to populations in the United States and Europe.
Continue to: Early versus delayed postpartum etonogestrel implant insertion...
Early versus delayed postpartum etonogestrel implant insertion: Similar impacts on 12-month bleeding patterns
Vieira CS, de Nadai MN, de Melo Pereira do Carmo LS, et al. Timing of postpartum etonogestrel-releasing implant insertion and bleeding patterns, weight change, 12-month continuation and satisfaction rates: a randomized controlled trial. Contraception. 2019. doi:10.1016/j.contraception.2019.05.007.
Initiation of a desired LARC method shortly after delivery is associated with significant reductions in short interpregnancy intervals.14 With that goal in mind, Vieira and colleagues compared bleeding patterns in women who received an etonogestrel implant within 48 hours of delivery with those who received an implant at 6 weeks postdelivery.
The study was a secondary analysis of data from a randomized controlled trial of early versus delayed postpartum insertion of the etonogestrel implant conducted in Sao Paulo, Brazil. That primary trial's goal was to examine the impact of early versus delayed implant insertion on infant growth (100 women were randomly assigned to the 2 implant groups); no difference in infant growth at 12 months was seen in the 2 groups.15 In the secondary analysis, bleeding patterns and BMI were evaluated every 90 days for 12 months. The mean BMI at enrollment postpartum was 29.4 kg/m2 in the early-insertion group and 30.2 kg/m2 for the delayed-insertion group.
Bleeding patterns with early or delayed implant insertion were similar
Vieira and colleagues found similar bleeding patterns between the groups over 12 months of follow-up. Amenorrhea was reported by 56% of the early-insertion group in the first 90 days and by 62% in the delayed-insertion group. During the last 90 days of the year, 52% of the early-insertion and 46% of the delayed-insertion group reported amenorrhea. Amenorrhea rates did not differ between women who were exclusively breastfeeding and those nonexclusively breastfeeding.
Continuation rates were high at 1 year
Prolonged bleeding episodes were uncommon in both groups, with only 2% of women reporting prolonged bleeding in any given reference period. Twelve-month implant continuation rates were high in both groups: 98% in the early- and 100% in the delayed-insertion group. Additionally, the investigators found that both groups experienced a BMI decrease, with no difference between groups (10.3% and 11% in the early- and delayed-insertion groups, respectively).
Study limitations and strengths
This study included a larger number of participants than prior randomized, controlled trials that evaluated bleeding patterns with postpartum etonogestrel implant insertion, and it had very low rates of loss to follow-up. The study's low rate of 12-month implant discontinuation (2%) is lower than that of other studies that reported rates of 6% to 14%.16,17 Although the authors stated that this low rate may be due to thorough anticipatory counseling prior to placement, it is also possible that this study population does not reflect all populations. Regardless, the data clearly show that placing an etonogestrel implant prior to hospital discharge, compared with waiting for later placement, does not impact bleeding patterns over the ensuing year.
For patients who desire an etonogestrel implant for contraception postpartum, we now have additional information to counsel about the impact of implant placement on postpartum bleeding patterns. Overall, bleeding patterns are highly favorable and do not vary whether the implant is placed in the hospital or later. Additionally, the timing of placement does not impact implant continuation rates or BMI changes over 1 year. Further, the primary study assessed infant growth in the early- versus delayed-placement groups and found no differences in infant growth. Although the data are limited, immediate postpartum etonogestrel implant placement does not seem to affect the rate of breastfeeding or the volume of breast milk.18,19 Timing of implant placement, assuming adequate resources, should be based primarily on patient preference. And, given the correlation of immediate postpartum LARC placement to increased interpregnancy interval, particular efforts should be made to provide the implant in the immediate postpartum period, if the patient desires.20
- Kavanaugh ML, Jerman J. Contraceptive method use in the United States: trends and characteristics between 2008, 2012 and 2014. Contraception. 2018;97:14-21.
- Trussell J. Contraceptive failure in the United States. Contraception. 2011;83:397-404.
- Odom EB, Eisenberg DL, Fox IK. Difficult removal of subdermal contraceptive implants: a multidisciplinary approach involving a peripheral nerve expert. Contraception. 2017;96: 89-95.
- Funk S, Miller MM, Mishell DR Jr, et al; Implanon US Study Group. Safety and efficacy of Implanon, a single-rod implantable contraceptive containing etonogestrel. Contraception. 2005;71:319-326.
- Eisenberg DL, Schreiber CA, Turok DK, et al; ACCESS IUS Investigators. Three-year efficacy and safety of a new 52-mg levonorgestrel-releasing intrauterine system. Contraception. 2015;92:10-16.
- Nelson A, Apter D, Hauck B, et al. Two low-dose levonorgestrel intrauterine contraceptive systems: a randomized controlled trial. Obstet Gynecol. 2013;122:1205-1213.
- Beckert V, Ahlers C, Frenz AK, et al. Bleeding patterns with the 19.5mg LNG-IUS, with special focus on the first year of use: implications for counselling. Eur J Contracept Reprod Health Care. 2019;24:251-259.
- Teal SB, Turok DK, Chen BA, et al. Five-year contraceptive efficacy and safety of a levonorgestrel 52-mg intrauterine system. Obstet Gynecol. 2019;133:63-70.
- Belsey EM, Machines D, d’Arcangues C. The analysis of vaginal bleeding patterns induced by fertility regulating methods. Contraception. 1986;34:253-260.
- Schreiber CA, Teal SB, Blumenthal PD, et al. Bleeding patterns for the Liletta® levonorgestrel 52mg intrauterine system. Eur J Contracept Reprod Health Care. 2018;23:116–120.
- Gemzell-Danielsson K, Schellschmidt I, Apter D. A randomized, phase II study describing the efficacy, bleeding profile, and safety of two low-dose levonorgestrel-releasing intrauterine contraceptive systems and Mirena. Fertil Steril. 2012;97:616-22.e1-3.
- Mansour D, Korver T, Marintcheva-Petrova M, et al. The effects of Implanon on menstrual bleeding patterns. Eur J Contracept Reprod Health Care. 2008;13(suppl 1):13-28.
- Guiahi M, McBride M, Sheeder J, et al. Short-term treatment of bothersome bleeding for etonogestrel implant users using a 14-day oral contraceptive pill regimen: a randomized controlled trial. Obstet Gynecol. 2015;126:508-513.
- Brunson MR, Klein DA, Olsen CH, et al. Postpartum contraception: initiation and effectiveness in a large universal healthcare system. Am J Obstet Gynecol. 2017;217:55.e1-55.e9
- de Melo Pereira Carmo LS, Braga GC, Ferriani RA, et al. Timing of etonogestrel-releasing implants and growth of breastfed infants: a randomized controlled trial. Obstet Gynecol. 2017;130:100-107.
- Crockett AH, Pickell LB, Heberlein EC, et al. Six- and twelve-month documented removal rates among women electing postpartum inpatient compared to delayed or interval contraceptive implant insertions after Medicaid payment reform. Contraception. 2017;95:71-76.
- Wilson S, Tennant C, Sammel MD, et al. Immediate postpartum etonogestrel implant: a contraception option with long-term continuation. Contraception. 2014;90:259-264.
- Sothornwit J, Werawatakul Y, Kaewrudee S, et al. Immediate versus delayed postpartum insertion of contraceptive implant for contraception. Cochrane Database Syst Rev. 2017;4:CD011913.
- Braga GC, Ferriolli E, Quintana SM, et al. Immediate postpartum initiation of etonogestrel-releasing implant: a randomized controlled trial on breastfeeding impact. Contraception. 2015;92:536-542.
- Thiel de Bocanegra H, Chang R, Howell M, et al. Interpregnancy intervals: impact of postpartum contraceptive effectiveness and coverage. Am J Obstet Gynecol. 2014;210:311.e1-8.
- Kyleena [package insert]. Whippany, NJ: Bayer HealthCare Pharmaceuticals Inc;2016.
- Skyla [package insert]. Whippany, NJ: Bayer HealthCare Pharmaceuticals Inc; 2016.
Long-acting reversible contraception (LARC) use continues to increase in the United States. According to the most recent estimates from 2014, 14% of women use either an intrauterine device (IUD) or the etonogestrel implant.1 Forms of LARC currently available in the United States include:
- 4 hormone-releasing IUDs
- 1 nonhormonal copper IUD, and
- 1 hormonal subdermal implant.
The hormone-releasing IUDs all contain levonorgestrel (LNG). These include two 52-mg LNG products and a 19.5-mg LNG IUD, which are currently approved by the US Food and Drug Administration (FDA) for contraception for 5 continuous years of use. In addition, a 13.5-mg LNG IUD is FDA-approved for 3 years of use. The hormonal subdermal implant, which contains etonogestrel, is FDA-approved for 3 years of use. Although major complications with IUDs (perforation, expulsion, intrauterine infection)and implants (subfascial implantation, distant migration) are rare, adverse effects that can affect continuation—such as irregular bleeding—are more common.2,3
Contraceptive discontinuation due to bleeding concerns occurs more frequently with the etonogestrel implant than with LNG IUDs (TABLE 1). In a large prospective study in the United States, 13% of women discontinued the implant during 3 years of follow-up due to bleeding pattern changes.
Notably, it is important to use standardized definitions to understand and compare bleeding concerns with LARC use. The Belsey criteria of the World Health Organization (WHO), a standard used for decades, describe bleeding patterns using 90-day reference periods or intervals (TABLE 2).9 Bleeding patterns that decrease flow (amenorrhea, infrequent bleeding) often are considered favorable, and those that increase bleeding or irregularity often are considered unfavorable. These criteria are commonly used in package labeling to describe bleeding patterns with extended use.


In this Update, we examine recent data evaluating differences in bleeding patterns with the 3 doses of the LNG IUD, predictors of abnormal bleeding with the etonogestrel implant, and the impact of timing on postpartum etonogestrel implant placement.
Continue to: Bleeding patterns with progestin-containing IUDs vary according to the LNG dose...
Bleeding patterns with progestin-containing IUDs vary according to the LNG dose
Goldthwaite LM, Creinin MD. Comparing bleeding patterns for the levonorgestrel 52 mg, 19.5 mg, and 13.5 mg intrauterine systems. Contraception. 2019;100:128-131.
Counseling on IUDs' different hormonal doses requires an understanding of patients' desires for contraceptive efficacy and bleeding expectations. A recent study provides guidance on what patients typically can expect for their bleeding patterns over the first few years with the 3 different doses of LNG IUDs.
Goldthwaite and Creinin used existing published or publicly available data to analyze differences in bleeding patterns associated with the 52-mg, 19.5-mg, and 13.5-mg LNG IUDs. Although two 52-mg LNG IUDs are available, published data using the WHO Belsey criteria are available only for one (Liletta; Allergan, Medicines360). The 2 products have been shown previously to have similar drug-release rates and LNG levels over 5 years.8
Comparing favorable bleeding patterns: Amenorrhea and infrequent bleeding
Among favorable bleeding patterns, amenorrhea was uncommon in the first 90 days and increased over time for all 3 IUDs. However, starting as soon as the second 90-day reference period, amenorrhea rates were significantly higher with the 52-mg LNG IUD compared with both of the lower-LNG dose IUDs, and this difference increased through 3 years of use (FIGURE 1).
Similarly, the 19.5-mg LNG IUD users had significantly higher rates of amenorrhea than the 13.5-mg LNG IUD users for all periods starting with the second 90-day reference period. At 3 years, 36% of women using the 52-mg LNG IUD had amenorrhea compared with 20% of those using the 19.5-mg LNG IUD (P<.0001) and 12% of those using the 13.5-mg LNG IUD (P<.0001).
Infrequent bleeding was similar for all 3 LNG IUDs in the first 90-day period, and it then increased most rapidly in the 52-mg LNG IUD users. At the end of year 1, 30% of the 52-mg LNG IUD users had infrequent bleeding compared with 26% of the 19.5-mg users (P = .01) and 20% of the 13.5-mg users (P<.0001). Although there was no difference in infrequent bleeding rates between the 52-mg and the 19.5-mg LNG IUD users at the end of year 1, those using a 52-mg LNG IUD had significantly higher rates of infrequent bleeding compared with the 13.5-mg LNG IUD at all time points.

Comparing unfavorable bleeding patterns: Frequent, prolonged, and irregular bleeding
Frequent and prolonged bleeding were uncommon with all LNG doses. Irregular bleeding rates declined for users of the 3 IUDs over time. However, significantly fewer users of the 52-mg LNG IUD reported irregular bleeding at 1 year (6%) compared with users of the 19.5-mg (16.5%, P<.0001) and 13.5-mg (23%, P<.0001) LNG IUD (FIGURE 2).

Study limitations
Comparing the data from different studies has limitations. For example, the data were collected from different populations, with the lower-dose LNG products tested in women who had a lower body mass index (BMI) and higher parity. However, prior analysis of the data on the 52-mg LNG IUD demonstrated that bleeding pattern changes did not vary based on these factors.10
When considering the different progestin-based IUD options, it is important to counsel patients according to their preferences for potential adverse effects. A randomized trial during product development found no difference in systemic adverse effects with the 3 doses of LNG IUD, likely because the systemic hormone levels are incredibly low for all 3 products.11 The summary data in this report helps explain why women using the lower-dose LNG products have slightly higher discontinuation rates for bleeding complaints, a fact we can explain to our patients during counseling.
Overall, the 52-mg LNG IUD is associated with a higher likelihood of favorable bleeding patterns over the first few years of use, with higher rates of amenorrhea and infrequent bleeding and lower rates of irregular bleeding. For women who prefer to not have periods or to have infrequent periods, the 52-mg LNG IUD is most likely to provide that outcome. For a patient who prefers to have periods, there is no evidence that the lower-dose IUDs result in “regular” or “normal” menstrual bleeding, even though they do result in more bleeding/spotting days overall. To the contrary, the available data show that these women have a significantly higher likelihood of experiencing prolonged, frequent, and irregular bleeding. In fact, no studies have reported rates of “normal” bleeding with the progestin IUDs, likely because women uncommonly have “normal” bleeding with these contraception methods. If a patient does not desire amenorrhea or strongly prefers to have “regular bleeding,” alternative methods such as a copper IUD should be considered rather than counseling her toward a lower-dose progestin IUD.
Continue to: Predicting long-term bleeding patterns after etonogestrel implant insertion...
Predicting long-term bleeding patterns after etonogestrel implant insertion
Mansour D, Fraser IS, Edelman A, et al. Can initial vaginal bleeding patterns in etonogestrel implant users predict subsequent bleeding in the first two years of use? Contraception. 2019. doi: 10.1016/j.contraception.2019.05.017.
Data from 2014 indicate that the etonogestrel implant was used by nearly 1 million women in the United States and by 3% of women using contraception.1 The primary reason women discontinue implant use is because of changes in bleeding patterns. Given the high prevalence of bleeding concerns with the etonogestrel implant, we need more data to help counsel our patients on how they can expect their bleeding to change with implant use.
Etonogestral implant and bleeding pattern trends
Mansour and colleagues completed a secondary analysis of 12 phase 3 studies to evaluate the correlation between bleeding patterns early after placement of the etonogestrel implant (days 29-118) compared with bleeding patterns through 90-day intervals during the rest of the first year of use. To account for differences in timing of etonogestrel implant placement relative to the menstrual cycle and discontinuation of other methods like oral contraceptives, bleeding outcomes on days 0-28 were excluded. They also sought to investigate the correlation between bleeding patterns in year 1 compared with those in year 2.
Overall, these studies included 923 individuals across 11 countries; however, for the current analysis, the researchers excluded women from Asian countries who comprised more than 28% of the study population. These women report significantly fewer bleeding/spotting days with the etonogestrel implant and have a lower average body weight compared with European and American women.12
A prior analysis of the same data set looked at the number of bleeding/spotting days in groups of users rather than trends in individual patients, and, as mentioned, it also included Asian women, which diluted the overall number of bleeding days.12 In this new analysis, Mansour and colleagues used the Belsey criteria to analyze individual bleeding patterns as favorable (amenorrhea, infrequent bleeding, normal bleeding) or unfavorable (prolonged and/or frequent bleeding) from a patient perspective. In this way, we can understand trends in bleeding patterns for each patient over time, rather than seeing a static (cross-sectional) report of bleeding patterns at one point in time. Data were analyzed from 537 women in year 1 and 428 women in year 2. During the first 90-day reference period (days 29-118 after implant insertion), 61% of women reported favorable bleeding, and 39% reported unfavorable bleeding.
Favorable bleeding correlates with favorable patterns later
A favorable bleeding pattern in this first reference period correlated with favorable bleeding patterns through year 1, with 85%, 80%, and 80% of these women having a favorable pattern in reference periods 2, 3, and 4, respectively. Overall, 61% of women with a favorable pattern in reference period 1 had favorable bleeding throughout the entire first year of use. Only 3.7% of women with favorable bleeding in the first reference period discontinued the implant for bleeding in year 1. Further, women with favorable bleeding at year 1 commonly continued to have favorable bleeding in year 2, with a low discontinuation rate (2.5%) in year 2.
Individual patients who have a favorable bleeding pattern initially with etonogestrel implant placement are highly likely to continue having favorable bleeding at year 1 and year 2. Notably, of women with a favorable bleeding pattern in any 90-day reference period, about 80% will continue to have a favorable bleeding pattern in the next reference period. These women can be counseled that, even if they have a 90-day period with unfavorable bleeding, about two-thirds will have a favorable pattern in the next reference period. For those with initial unfavorable patterns, about one-third to one-half change to a favorable pattern in subsequent 90-day reference periods. For women who require intervention for unfavorable bleeding but wish to keep their etonogestrel implant, prior data support use of combined oral contraceptive pills, although bleeding resolution seems to be temporary, with 86% of women having bleeding recurrence within 10 days after treatment.13
Initial unfavorable bleeding portends less favorable patterns later
Women who had an unfavorable bleeding pattern initially, however, had a less predictable course over the first year. For those with an initial unfavorable pattern, only 37%, 47%, and 51% reported a favorable pattern in reference periods 2, 3, and 4. Despite these relatively low rates of favorable bleeding, only 13% of the women with an initial unfavorable bleeding pattern discontinued implant use for a bleeding complaint by the end of year 1; this rate was significantly higher than that for women with a favorable initial bleeding pattern (P<.0001). The discontinuation rate for bleeding complaints also remained higher in year 2, at 16.5%.
Limitations and strengths to consider
Although the etonogestrel implant is FDA-approved for 3 years of use, the bleeding data from the combined trials included information for only up to 2 years after placement. The studies included also did not uniformly assess BMI, which makes it difficult to find correlations between bleeding patterns and BMI. Importantly, the studies did not include women who were more than 30% above their ideal body weight, so these assessments do not apply to obese users.12 Exclusion of women from Southeast Asia in this analysis makes this study's findings more generalizable to populations in the United States and Europe.
Continue to: Early versus delayed postpartum etonogestrel implant insertion...
Early versus delayed postpartum etonogestrel implant insertion: Similar impacts on 12-month bleeding patterns
Vieira CS, de Nadai MN, de Melo Pereira do Carmo LS, et al. Timing of postpartum etonogestrel-releasing implant insertion and bleeding patterns, weight change, 12-month continuation and satisfaction rates: a randomized controlled trial. Contraception. 2019. doi:10.1016/j.contraception.2019.05.007.
Initiation of a desired LARC method shortly after delivery is associated with significant reductions in short interpregnancy intervals.14 With that goal in mind, Vieira and colleagues compared bleeding patterns in women who received an etonogestrel implant within 48 hours of delivery with those who received an implant at 6 weeks postdelivery.
The study was a secondary analysis of data from a randomized controlled trial of early versus delayed postpartum insertion of the etonogestrel implant conducted in Sao Paulo, Brazil. That primary trial's goal was to examine the impact of early versus delayed implant insertion on infant growth (100 women were randomly assigned to the 2 implant groups); no difference in infant growth at 12 months was seen in the 2 groups.15 In the secondary analysis, bleeding patterns and BMI were evaluated every 90 days for 12 months. The mean BMI at enrollment postpartum was 29.4 kg/m2 in the early-insertion group and 30.2 kg/m2 for the delayed-insertion group.
Bleeding patterns with early or delayed implant insertion were similar
Vieira and colleagues found similar bleeding patterns between the groups over 12 months of follow-up. Amenorrhea was reported by 56% of the early-insertion group in the first 90 days and by 62% in the delayed-insertion group. During the last 90 days of the year, 52% of the early-insertion and 46% of the delayed-insertion group reported amenorrhea. Amenorrhea rates did not differ between women who were exclusively breastfeeding and those nonexclusively breastfeeding.
Continuation rates were high at 1 year
Prolonged bleeding episodes were uncommon in both groups, with only 2% of women reporting prolonged bleeding in any given reference period. Twelve-month implant continuation rates were high in both groups: 98% in the early- and 100% in the delayed-insertion group. Additionally, the investigators found that both groups experienced a BMI decrease, with no difference between groups (10.3% and 11% in the early- and delayed-insertion groups, respectively).
Study limitations and strengths
This study included a larger number of participants than prior randomized, controlled trials that evaluated bleeding patterns with postpartum etonogestrel implant insertion, and it had very low rates of loss to follow-up. The study's low rate of 12-month implant discontinuation (2%) is lower than that of other studies that reported rates of 6% to 14%.16,17 Although the authors stated that this low rate may be due to thorough anticipatory counseling prior to placement, it is also possible that this study population does not reflect all populations. Regardless, the data clearly show that placing an etonogestrel implant prior to hospital discharge, compared with waiting for later placement, does not impact bleeding patterns over the ensuing year.
For patients who desire an etonogestrel implant for contraception postpartum, we now have additional information to counsel about the impact of implant placement on postpartum bleeding patterns. Overall, bleeding patterns are highly favorable and do not vary whether the implant is placed in the hospital or later. Additionally, the timing of placement does not impact implant continuation rates or BMI changes over 1 year. Further, the primary study assessed infant growth in the early- versus delayed-placement groups and found no differences in infant growth. Although the data are limited, immediate postpartum etonogestrel implant placement does not seem to affect the rate of breastfeeding or the volume of breast milk.18,19 Timing of implant placement, assuming adequate resources, should be based primarily on patient preference. And, given the correlation of immediate postpartum LARC placement to increased interpregnancy interval, particular efforts should be made to provide the implant in the immediate postpartum period, if the patient desires.20
Long-acting reversible contraception (LARC) use continues to increase in the United States. According to the most recent estimates from 2014, 14% of women use either an intrauterine device (IUD) or the etonogestrel implant.1 Forms of LARC currently available in the United States include:
- 4 hormone-releasing IUDs
- 1 nonhormonal copper IUD, and
- 1 hormonal subdermal implant.
The hormone-releasing IUDs all contain levonorgestrel (LNG). These include two 52-mg LNG products and a 19.5-mg LNG IUD, which are currently approved by the US Food and Drug Administration (FDA) for contraception for 5 continuous years of use. In addition, a 13.5-mg LNG IUD is FDA-approved for 3 years of use. The hormonal subdermal implant, which contains etonogestrel, is FDA-approved for 3 years of use. Although major complications with IUDs (perforation, expulsion, intrauterine infection)and implants (subfascial implantation, distant migration) are rare, adverse effects that can affect continuation—such as irregular bleeding—are more common.2,3
Contraceptive discontinuation due to bleeding concerns occurs more frequently with the etonogestrel implant than with LNG IUDs (TABLE 1). In a large prospective study in the United States, 13% of women discontinued the implant during 3 years of follow-up due to bleeding pattern changes.
Notably, it is important to use standardized definitions to understand and compare bleeding concerns with LARC use. The Belsey criteria of the World Health Organization (WHO), a standard used for decades, describe bleeding patterns using 90-day reference periods or intervals (TABLE 2).9 Bleeding patterns that decrease flow (amenorrhea, infrequent bleeding) often are considered favorable, and those that increase bleeding or irregularity often are considered unfavorable. These criteria are commonly used in package labeling to describe bleeding patterns with extended use.


In this Update, we examine recent data evaluating differences in bleeding patterns with the 3 doses of the LNG IUD, predictors of abnormal bleeding with the etonogestrel implant, and the impact of timing on postpartum etonogestrel implant placement.
Continue to: Bleeding patterns with progestin-containing IUDs vary according to the LNG dose...
Bleeding patterns with progestin-containing IUDs vary according to the LNG dose
Goldthwaite LM, Creinin MD. Comparing bleeding patterns for the levonorgestrel 52 mg, 19.5 mg, and 13.5 mg intrauterine systems. Contraception. 2019;100:128-131.
Counseling on IUDs' different hormonal doses requires an understanding of patients' desires for contraceptive efficacy and bleeding expectations. A recent study provides guidance on what patients typically can expect for their bleeding patterns over the first few years with the 3 different doses of LNG IUDs.
Goldthwaite and Creinin used existing published or publicly available data to analyze differences in bleeding patterns associated with the 52-mg, 19.5-mg, and 13.5-mg LNG IUDs. Although two 52-mg LNG IUDs are available, published data using the WHO Belsey criteria are available only for one (Liletta; Allergan, Medicines360). The 2 products have been shown previously to have similar drug-release rates and LNG levels over 5 years.8
Comparing favorable bleeding patterns: Amenorrhea and infrequent bleeding
Among favorable bleeding patterns, amenorrhea was uncommon in the first 90 days and increased over time for all 3 IUDs. However, starting as soon as the second 90-day reference period, amenorrhea rates were significantly higher with the 52-mg LNG IUD compared with both of the lower-LNG dose IUDs, and this difference increased through 3 years of use (FIGURE 1).
Similarly, the 19.5-mg LNG IUD users had significantly higher rates of amenorrhea than the 13.5-mg LNG IUD users for all periods starting with the second 90-day reference period. At 3 years, 36% of women using the 52-mg LNG IUD had amenorrhea compared with 20% of those using the 19.5-mg LNG IUD (P<.0001) and 12% of those using the 13.5-mg LNG IUD (P<.0001).
Infrequent bleeding was similar for all 3 LNG IUDs in the first 90-day period, and it then increased most rapidly in the 52-mg LNG IUD users. At the end of year 1, 30% of the 52-mg LNG IUD users had infrequent bleeding compared with 26% of the 19.5-mg users (P = .01) and 20% of the 13.5-mg users (P<.0001). Although there was no difference in infrequent bleeding rates between the 52-mg and the 19.5-mg LNG IUD users at the end of year 1, those using a 52-mg LNG IUD had significantly higher rates of infrequent bleeding compared with the 13.5-mg LNG IUD at all time points.

Comparing unfavorable bleeding patterns: Frequent, prolonged, and irregular bleeding
Frequent and prolonged bleeding were uncommon with all LNG doses. Irregular bleeding rates declined for users of the 3 IUDs over time. However, significantly fewer users of the 52-mg LNG IUD reported irregular bleeding at 1 year (6%) compared with users of the 19.5-mg (16.5%, P<.0001) and 13.5-mg (23%, P<.0001) LNG IUD (FIGURE 2).

Study limitations
Comparing the data from different studies has limitations. For example, the data were collected from different populations, with the lower-dose LNG products tested in women who had a lower body mass index (BMI) and higher parity. However, prior analysis of the data on the 52-mg LNG IUD demonstrated that bleeding pattern changes did not vary based on these factors.10
When considering the different progestin-based IUD options, it is important to counsel patients according to their preferences for potential adverse effects. A randomized trial during product development found no difference in systemic adverse effects with the 3 doses of LNG IUD, likely because the systemic hormone levels are incredibly low for all 3 products.11 The summary data in this report helps explain why women using the lower-dose LNG products have slightly higher discontinuation rates for bleeding complaints, a fact we can explain to our patients during counseling.
Overall, the 52-mg LNG IUD is associated with a higher likelihood of favorable bleeding patterns over the first few years of use, with higher rates of amenorrhea and infrequent bleeding and lower rates of irregular bleeding. For women who prefer to not have periods or to have infrequent periods, the 52-mg LNG IUD is most likely to provide that outcome. For a patient who prefers to have periods, there is no evidence that the lower-dose IUDs result in “regular” or “normal” menstrual bleeding, even though they do result in more bleeding/spotting days overall. To the contrary, the available data show that these women have a significantly higher likelihood of experiencing prolonged, frequent, and irregular bleeding. In fact, no studies have reported rates of “normal” bleeding with the progestin IUDs, likely because women uncommonly have “normal” bleeding with these contraception methods. If a patient does not desire amenorrhea or strongly prefers to have “regular bleeding,” alternative methods such as a copper IUD should be considered rather than counseling her toward a lower-dose progestin IUD.
Continue to: Predicting long-term bleeding patterns after etonogestrel implant insertion...
Predicting long-term bleeding patterns after etonogestrel implant insertion
Mansour D, Fraser IS, Edelman A, et al. Can initial vaginal bleeding patterns in etonogestrel implant users predict subsequent bleeding in the first two years of use? Contraception. 2019. doi: 10.1016/j.contraception.2019.05.017.
Data from 2014 indicate that the etonogestrel implant was used by nearly 1 million women in the United States and by 3% of women using contraception.1 The primary reason women discontinue implant use is because of changes in bleeding patterns. Given the high prevalence of bleeding concerns with the etonogestrel implant, we need more data to help counsel our patients on how they can expect their bleeding to change with implant use.
Etonogestral implant and bleeding pattern trends
Mansour and colleagues completed a secondary analysis of 12 phase 3 studies to evaluate the correlation between bleeding patterns early after placement of the etonogestrel implant (days 29-118) compared with bleeding patterns through 90-day intervals during the rest of the first year of use. To account for differences in timing of etonogestrel implant placement relative to the menstrual cycle and discontinuation of other methods like oral contraceptives, bleeding outcomes on days 0-28 were excluded. They also sought to investigate the correlation between bleeding patterns in year 1 compared with those in year 2.
Overall, these studies included 923 individuals across 11 countries; however, for the current analysis, the researchers excluded women from Asian countries who comprised more than 28% of the study population. These women report significantly fewer bleeding/spotting days with the etonogestrel implant and have a lower average body weight compared with European and American women.12
A prior analysis of the same data set looked at the number of bleeding/spotting days in groups of users rather than trends in individual patients, and, as mentioned, it also included Asian women, which diluted the overall number of bleeding days.12 In this new analysis, Mansour and colleagues used the Belsey criteria to analyze individual bleeding patterns as favorable (amenorrhea, infrequent bleeding, normal bleeding) or unfavorable (prolonged and/or frequent bleeding) from a patient perspective. In this way, we can understand trends in bleeding patterns for each patient over time, rather than seeing a static (cross-sectional) report of bleeding patterns at one point in time. Data were analyzed from 537 women in year 1 and 428 women in year 2. During the first 90-day reference period (days 29-118 after implant insertion), 61% of women reported favorable bleeding, and 39% reported unfavorable bleeding.
Favorable bleeding correlates with favorable patterns later
A favorable bleeding pattern in this first reference period correlated with favorable bleeding patterns through year 1, with 85%, 80%, and 80% of these women having a favorable pattern in reference periods 2, 3, and 4, respectively. Overall, 61% of women with a favorable pattern in reference period 1 had favorable bleeding throughout the entire first year of use. Only 3.7% of women with favorable bleeding in the first reference period discontinued the implant for bleeding in year 1. Further, women with favorable bleeding at year 1 commonly continued to have favorable bleeding in year 2, with a low discontinuation rate (2.5%) in year 2.
Individual patients who have a favorable bleeding pattern initially with etonogestrel implant placement are highly likely to continue having favorable bleeding at year 1 and year 2. Notably, of women with a favorable bleeding pattern in any 90-day reference period, about 80% will continue to have a favorable bleeding pattern in the next reference period. These women can be counseled that, even if they have a 90-day period with unfavorable bleeding, about two-thirds will have a favorable pattern in the next reference period. For those with initial unfavorable patterns, about one-third to one-half change to a favorable pattern in subsequent 90-day reference periods. For women who require intervention for unfavorable bleeding but wish to keep their etonogestrel implant, prior data support use of combined oral contraceptive pills, although bleeding resolution seems to be temporary, with 86% of women having bleeding recurrence within 10 days after treatment.13
Initial unfavorable bleeding portends less favorable patterns later
Women who had an unfavorable bleeding pattern initially, however, had a less predictable course over the first year. For those with an initial unfavorable pattern, only 37%, 47%, and 51% reported a favorable pattern in reference periods 2, 3, and 4. Despite these relatively low rates of favorable bleeding, only 13% of the women with an initial unfavorable bleeding pattern discontinued implant use for a bleeding complaint by the end of year 1; this rate was significantly higher than that for women with a favorable initial bleeding pattern (P<.0001). The discontinuation rate for bleeding complaints also remained higher in year 2, at 16.5%.
Limitations and strengths to consider
Although the etonogestrel implant is FDA-approved for 3 years of use, the bleeding data from the combined trials included information for only up to 2 years after placement. The studies included also did not uniformly assess BMI, which makes it difficult to find correlations between bleeding patterns and BMI. Importantly, the studies did not include women who were more than 30% above their ideal body weight, so these assessments do not apply to obese users.12 Exclusion of women from Southeast Asia in this analysis makes this study's findings more generalizable to populations in the United States and Europe.
Continue to: Early versus delayed postpartum etonogestrel implant insertion...
Early versus delayed postpartum etonogestrel implant insertion: Similar impacts on 12-month bleeding patterns
Vieira CS, de Nadai MN, de Melo Pereira do Carmo LS, et al. Timing of postpartum etonogestrel-releasing implant insertion and bleeding patterns, weight change, 12-month continuation and satisfaction rates: a randomized controlled trial. Contraception. 2019. doi:10.1016/j.contraception.2019.05.007.
Initiation of a desired LARC method shortly after delivery is associated with significant reductions in short interpregnancy intervals.14 With that goal in mind, Vieira and colleagues compared bleeding patterns in women who received an etonogestrel implant within 48 hours of delivery with those who received an implant at 6 weeks postdelivery.
The study was a secondary analysis of data from a randomized controlled trial of early versus delayed postpartum insertion of the etonogestrel implant conducted in Sao Paulo, Brazil. That primary trial's goal was to examine the impact of early versus delayed implant insertion on infant growth (100 women were randomly assigned to the 2 implant groups); no difference in infant growth at 12 months was seen in the 2 groups.15 In the secondary analysis, bleeding patterns and BMI were evaluated every 90 days for 12 months. The mean BMI at enrollment postpartum was 29.4 kg/m2 in the early-insertion group and 30.2 kg/m2 for the delayed-insertion group.
Bleeding patterns with early or delayed implant insertion were similar
Vieira and colleagues found similar bleeding patterns between the groups over 12 months of follow-up. Amenorrhea was reported by 56% of the early-insertion group in the first 90 days and by 62% in the delayed-insertion group. During the last 90 days of the year, 52% of the early-insertion and 46% of the delayed-insertion group reported amenorrhea. Amenorrhea rates did not differ between women who were exclusively breastfeeding and those nonexclusively breastfeeding.
Continuation rates were high at 1 year
Prolonged bleeding episodes were uncommon in both groups, with only 2% of women reporting prolonged bleeding in any given reference period. Twelve-month implant continuation rates were high in both groups: 98% in the early- and 100% in the delayed-insertion group. Additionally, the investigators found that both groups experienced a BMI decrease, with no difference between groups (10.3% and 11% in the early- and delayed-insertion groups, respectively).
Study limitations and strengths
This study included a larger number of participants than prior randomized, controlled trials that evaluated bleeding patterns with postpartum etonogestrel implant insertion, and it had very low rates of loss to follow-up. The study's low rate of 12-month implant discontinuation (2%) is lower than that of other studies that reported rates of 6% to 14%.16,17 Although the authors stated that this low rate may be due to thorough anticipatory counseling prior to placement, it is also possible that this study population does not reflect all populations. Regardless, the data clearly show that placing an etonogestrel implant prior to hospital discharge, compared with waiting for later placement, does not impact bleeding patterns over the ensuing year.
For patients who desire an etonogestrel implant for contraception postpartum, we now have additional information to counsel about the impact of implant placement on postpartum bleeding patterns. Overall, bleeding patterns are highly favorable and do not vary whether the implant is placed in the hospital or later. Additionally, the timing of placement does not impact implant continuation rates or BMI changes over 1 year. Further, the primary study assessed infant growth in the early- versus delayed-placement groups and found no differences in infant growth. Although the data are limited, immediate postpartum etonogestrel implant placement does not seem to affect the rate of breastfeeding or the volume of breast milk.18,19 Timing of implant placement, assuming adequate resources, should be based primarily on patient preference. And, given the correlation of immediate postpartum LARC placement to increased interpregnancy interval, particular efforts should be made to provide the implant in the immediate postpartum period, if the patient desires.20
- Kavanaugh ML, Jerman J. Contraceptive method use in the United States: trends and characteristics between 2008, 2012 and 2014. Contraception. 2018;97:14-21.
- Trussell J. Contraceptive failure in the United States. Contraception. 2011;83:397-404.
- Odom EB, Eisenberg DL, Fox IK. Difficult removal of subdermal contraceptive implants: a multidisciplinary approach involving a peripheral nerve expert. Contraception. 2017;96: 89-95.
- Funk S, Miller MM, Mishell DR Jr, et al; Implanon US Study Group. Safety and efficacy of Implanon, a single-rod implantable contraceptive containing etonogestrel. Contraception. 2005;71:319-326.
- Eisenberg DL, Schreiber CA, Turok DK, et al; ACCESS IUS Investigators. Three-year efficacy and safety of a new 52-mg levonorgestrel-releasing intrauterine system. Contraception. 2015;92:10-16.
- Nelson A, Apter D, Hauck B, et al. Two low-dose levonorgestrel intrauterine contraceptive systems: a randomized controlled trial. Obstet Gynecol. 2013;122:1205-1213.
- Beckert V, Ahlers C, Frenz AK, et al. Bleeding patterns with the 19.5mg LNG-IUS, with special focus on the first year of use: implications for counselling. Eur J Contracept Reprod Health Care. 2019;24:251-259.
- Teal SB, Turok DK, Chen BA, et al. Five-year contraceptive efficacy and safety of a levonorgestrel 52-mg intrauterine system. Obstet Gynecol. 2019;133:63-70.
- Belsey EM, Machines D, d’Arcangues C. The analysis of vaginal bleeding patterns induced by fertility regulating methods. Contraception. 1986;34:253-260.
- Schreiber CA, Teal SB, Blumenthal PD, et al. Bleeding patterns for the Liletta® levonorgestrel 52mg intrauterine system. Eur J Contracept Reprod Health Care. 2018;23:116–120.
- Gemzell-Danielsson K, Schellschmidt I, Apter D. A randomized, phase II study describing the efficacy, bleeding profile, and safety of two low-dose levonorgestrel-releasing intrauterine contraceptive systems and Mirena. Fertil Steril. 2012;97:616-22.e1-3.
- Mansour D, Korver T, Marintcheva-Petrova M, et al. The effects of Implanon on menstrual bleeding patterns. Eur J Contracept Reprod Health Care. 2008;13(suppl 1):13-28.
- Guiahi M, McBride M, Sheeder J, et al. Short-term treatment of bothersome bleeding for etonogestrel implant users using a 14-day oral contraceptive pill regimen: a randomized controlled trial. Obstet Gynecol. 2015;126:508-513.
- Brunson MR, Klein DA, Olsen CH, et al. Postpartum contraception: initiation and effectiveness in a large universal healthcare system. Am J Obstet Gynecol. 2017;217:55.e1-55.e9
- de Melo Pereira Carmo LS, Braga GC, Ferriani RA, et al. Timing of etonogestrel-releasing implants and growth of breastfed infants: a randomized controlled trial. Obstet Gynecol. 2017;130:100-107.
- Crockett AH, Pickell LB, Heberlein EC, et al. Six- and twelve-month documented removal rates among women electing postpartum inpatient compared to delayed or interval contraceptive implant insertions after Medicaid payment reform. Contraception. 2017;95:71-76.
- Wilson S, Tennant C, Sammel MD, et al. Immediate postpartum etonogestrel implant: a contraception option with long-term continuation. Contraception. 2014;90:259-264.
- Sothornwit J, Werawatakul Y, Kaewrudee S, et al. Immediate versus delayed postpartum insertion of contraceptive implant for contraception. Cochrane Database Syst Rev. 2017;4:CD011913.
- Braga GC, Ferriolli E, Quintana SM, et al. Immediate postpartum initiation of etonogestrel-releasing implant: a randomized controlled trial on breastfeeding impact. Contraception. 2015;92:536-542.
- Thiel de Bocanegra H, Chang R, Howell M, et al. Interpregnancy intervals: impact of postpartum contraceptive effectiveness and coverage. Am J Obstet Gynecol. 2014;210:311.e1-8.
- Kyleena [package insert]. Whippany, NJ: Bayer HealthCare Pharmaceuticals Inc;2016.
- Skyla [package insert]. Whippany, NJ: Bayer HealthCare Pharmaceuticals Inc; 2016.
- Kavanaugh ML, Jerman J. Contraceptive method use in the United States: trends and characteristics between 2008, 2012 and 2014. Contraception. 2018;97:14-21.
- Trussell J. Contraceptive failure in the United States. Contraception. 2011;83:397-404.
- Odom EB, Eisenberg DL, Fox IK. Difficult removal of subdermal contraceptive implants: a multidisciplinary approach involving a peripheral nerve expert. Contraception. 2017;96: 89-95.
- Funk S, Miller MM, Mishell DR Jr, et al; Implanon US Study Group. Safety and efficacy of Implanon, a single-rod implantable contraceptive containing etonogestrel. Contraception. 2005;71:319-326.
- Eisenberg DL, Schreiber CA, Turok DK, et al; ACCESS IUS Investigators. Three-year efficacy and safety of a new 52-mg levonorgestrel-releasing intrauterine system. Contraception. 2015;92:10-16.
- Nelson A, Apter D, Hauck B, et al. Two low-dose levonorgestrel intrauterine contraceptive systems: a randomized controlled trial. Obstet Gynecol. 2013;122:1205-1213.
- Beckert V, Ahlers C, Frenz AK, et al. Bleeding patterns with the 19.5mg LNG-IUS, with special focus on the first year of use: implications for counselling. Eur J Contracept Reprod Health Care. 2019;24:251-259.
- Teal SB, Turok DK, Chen BA, et al. Five-year contraceptive efficacy and safety of a levonorgestrel 52-mg intrauterine system. Obstet Gynecol. 2019;133:63-70.
- Belsey EM, Machines D, d’Arcangues C. The analysis of vaginal bleeding patterns induced by fertility regulating methods. Contraception. 1986;34:253-260.
- Schreiber CA, Teal SB, Blumenthal PD, et al. Bleeding patterns for the Liletta® levonorgestrel 52mg intrauterine system. Eur J Contracept Reprod Health Care. 2018;23:116–120.
- Gemzell-Danielsson K, Schellschmidt I, Apter D. A randomized, phase II study describing the efficacy, bleeding profile, and safety of two low-dose levonorgestrel-releasing intrauterine contraceptive systems and Mirena. Fertil Steril. 2012;97:616-22.e1-3.
- Mansour D, Korver T, Marintcheva-Petrova M, et al. The effects of Implanon on menstrual bleeding patterns. Eur J Contracept Reprod Health Care. 2008;13(suppl 1):13-28.
- Guiahi M, McBride M, Sheeder J, et al. Short-term treatment of bothersome bleeding for etonogestrel implant users using a 14-day oral contraceptive pill regimen: a randomized controlled trial. Obstet Gynecol. 2015;126:508-513.
- Brunson MR, Klein DA, Olsen CH, et al. Postpartum contraception: initiation and effectiveness in a large universal healthcare system. Am J Obstet Gynecol. 2017;217:55.e1-55.e9
- de Melo Pereira Carmo LS, Braga GC, Ferriani RA, et al. Timing of etonogestrel-releasing implants and growth of breastfed infants: a randomized controlled trial. Obstet Gynecol. 2017;130:100-107.
- Crockett AH, Pickell LB, Heberlein EC, et al. Six- and twelve-month documented removal rates among women electing postpartum inpatient compared to delayed or interval contraceptive implant insertions after Medicaid payment reform. Contraception. 2017;95:71-76.
- Wilson S, Tennant C, Sammel MD, et al. Immediate postpartum etonogestrel implant: a contraception option with long-term continuation. Contraception. 2014;90:259-264.
- Sothornwit J, Werawatakul Y, Kaewrudee S, et al. Immediate versus delayed postpartum insertion of contraceptive implant for contraception. Cochrane Database Syst Rev. 2017;4:CD011913.
- Braga GC, Ferriolli E, Quintana SM, et al. Immediate postpartum initiation of etonogestrel-releasing implant: a randomized controlled trial on breastfeeding impact. Contraception. 2015;92:536-542.
- Thiel de Bocanegra H, Chang R, Howell M, et al. Interpregnancy intervals: impact of postpartum contraceptive effectiveness and coverage. Am J Obstet Gynecol. 2014;210:311.e1-8.
- Kyleena [package insert]. Whippany, NJ: Bayer HealthCare Pharmaceuticals Inc;2016.
- Skyla [package insert]. Whippany, NJ: Bayer HealthCare Pharmaceuticals Inc; 2016.
Native tissue repair of POP: Apical suspension, anterior repair, and posterior repair



Videos courtesy of Mayo Clinic
Read the related article: Native tissue repair of POP: Surgical techniques to improve outcomes



Videos courtesy of Mayo Clinic
Read the related article: Native tissue repair of POP: Surgical techniques to improve outcomes



Videos courtesy of Mayo Clinic
Read the related article: Native tissue repair of POP: Surgical techniques to improve outcomes
Targeting US maternal mortality: ACOG’s recent strides and future action
Real progress was achieved in 2018 in the effort to reduce the US maternal mortality rate, the highest of any developed nation and where women of color are 3 to 4 times more likely than others to die of childbirth-related causes. Importantly, the United States is the only nation other than Afghanistan and Sudan where the rate is rising.1
In May 2019, the Centers for Disease Control and Prevention (CDC) published a Vital Signs document focused on preventable maternal deaths.2 It affirmed that about 60% of the 700 pregnancy-related deaths that occur annually in the United States are preventable, and it provided important information on when and why these deaths occur.
Among the CDC findings, about:
- one-third of deaths (31%) occurred during pregnancy (before delivery)
- one-third (36%) occurred at delivery or in the week after
- one-third (33%) occurred 1 week to 1 year postpartum.
In addition, the CDC highlighted that:
- Heart disease and stroke caused more than 1 in 3 deaths (34%). Infections and severe bleeding were other leading causes of death.
- Black and American Indian/Alaska Native women were about 3 times as likely to die from a pregnancy-related cause as white women.
The American College of Obstetricians and Gynecologists (ACOG), under the leadership of President Lisa Hollier, MD, MPH (2018–2019), fully embraced the challenge and responsibility of meaningfully improving health care for every mom. In this article, I review some of the critical steps taken in 2018 and preview ACOG’s continued commitment for 2019 and beyond.

Efforts succeed: Bills are now laws of the land
ACOG and our partner organizations, including the Society for Maternal-Fetal Medicine and the March of Dimes, have long recognized the value of state-based maternal mortality review committees (MMRCs) in slowing and reversing the rate of maternal mortality. An MMRC brings together local experts to examine the causes of maternal deaths—not to find fault, but to find ways to prevent future deaths. With the right framework and support, MMRCs already are providing us with data and driving policy recommendations.
Supporting MMRCs in all states. With this in mind, ACOG helped pass and push to enactment HR 1318, the Preventing Maternal Deaths Act of 2018 (Public Law No. 115-344), a bipartisan bill designed to help develop and provide support for MMRCs in every state. The bill was introduced in the US House of Representatives by Rep. Jaime Herrera Beutler (R-WA) and Rep. Diana DeGette (D-CO) and in the US Senate by Sen. Heidi Heitkamp (D-ND) and Sen. Shelley Moore Capito (R-WV). ACOG Fellow and US Rep. Michael Burgess, MD (R-TX), also was instrumental in the bill’s success. The CDC is actively working toward implementation of this law, and grantees are expected to be announced by the end of September.
Continue to: In addition, ACOG worked with Congress...
In addition, ACOG worked with Congress to secure $50 million in federal funding to reduce maternal mortality, allocated thusly:
- $12 million to support state MMRCs
- $3 million to support the Alliance for Innovation on Maternal Health
- $23 million for State Maternal Health Innovation Program grants
- $12 million to address maternal mortality in the Healthy Start program.
As these federal congressional initiatives worked their way into law, the states actively supported MMRCs as well. As of this writing, only 3 states—North Dakota, South Dakota, and Wyoming—have not yet developed an MMRC.3
Filling the gaps in ObGyn care. Another key ACOG-sponsored bill signed into law will help bring more ObGyns into shortage areas. Sponsored by Rep. Burgess, Rep. Anna Eshoo (D-CA), and Rep. Lucille Roybal-Allard (D-CA) and by Sen. Tammy Baldwin (D-WI) and Sen. Lisa Murkowski (R-AK), the Improving Access to Maternity Care Act (Public Law No. 115-320) requires the Department of Health and Human Services to identify maternity health professional target areas for use by the National Health Service Corps to bring ObGyns to where they are most needed.
Following up on that new law, ACOG currently is working closely with the American Academy of Family Physicians (AAFP) and the National Rural Health Association (NRHA) on the unique challenges women in rural areas face in accessing maternity and other women’s health care services. In June, Dr. Hollier represented ACOG at the Rural Maternal Health forum, which was convened by the Centers for Medicare and Medicaid and sponsored by ACOG, AAFP, and NRHA.4 We are pursuing policies designed to increase the number of ObGyns and other physicians who choose to train in rural areas and increase the clinical use of telehealth to help connect rural physicians and patients with subspecialists in urban areas.
Projects in the works
Congress is ready to do more. Already, 5 ACOG-supported bills have been introduced, including bills that extend women’s Medicaid coverage to 12 months postpartum (consistent with coverage for babies), support state perinatal quality collaboratives, and more. This interest is augmented by the work of the recently formed congressional Black Maternal Health Caucus, focused on reducing racial disparities in health care. In July, ACOG joined 12 members of Congress in a caucus summit to partner with these important congressional allies.
ACOG is expanding support for these legislative efforts through our work with another important ally, the American Medical Association (AMA). ACOG’s delegation to the 2019 Annual Meeting of the AMA House of Delegates in June scored important policy wins, including AMA support for Medicaid coverage for women 12 months postpartum and improving access to care in rural communities.
There is momentum on Capitol Hill to take action on these important issues, and ACOG’s priority is to ensure that any legislative package complements the important work many ObGyns are already doing to improve maternal health outcomes. ACOG has an important seat at the table and will continue to advocate each and every day for your practices and your patients as Congress deliberates legislative action.
Continue to: Your voice matters...
Your voice matters
Encourage your representatives in the House and the Senate to support ACOG-endorsed legislation and be sure they know the importance of ensuring access to women’s health care in your community. Get involved in advocacy; start by visiting the ACOG advocacy web page (www.acog.org/advocacy). Also note that members of Congress are back in their home states during seasonal breaks and many hold town halls and constituent meetings. The health of moms and babies is always an important issue, and you are the expert.
ACOG’s commitment to ensuring healthy moms and babies, and ensuring that our members can continue providing high-quality care, runs through everything we do.
Acknowledgments
The author thanks ACOG former Vice President for Health Policy Barbara Levy, MD, ACOG Senior Director Jeanne Mahoney, and ACOG Federal Affairs Director Rachel Tetlow for their helpful review and comments.
- Council on Patient Safety in Women's Health Care. Alliance for Innovation on Maternal Health Program. https://safehealthcareforeverywoman.org/aim-program/. Accessed August 19, 2019.
- Centers for Disease Control and Prevention. Vital signs: pregnancy-related deaths. https://www.cdc.gov/vitalsigns/maternal-deaths/index.html. Accessed August 19, 2019.
- American College of Obstetricians and Gynecologists. State Maternal Mortality Review Committees, PQCs, and AIM. https://www.acog.org/-/media/Departments/Government-Relations-and-Outreach/MMRC_AIM-State-Fact-Sheet_Mar-2019.pdf. Accessed August 19, 2019.
- Centers for Medicare and Medicaid Services. A conversation on maternal health care in rural communities: charting a path to improved access, quality and outcomes. June 12, 2019. https://www.cms.gov/About-CMS/Agency-Information/OMH/equity-initiatives/rural-health/rural-maternal-health.html. Accessed August 19, 2019.
Real progress was achieved in 2018 in the effort to reduce the US maternal mortality rate, the highest of any developed nation and where women of color are 3 to 4 times more likely than others to die of childbirth-related causes. Importantly, the United States is the only nation other than Afghanistan and Sudan where the rate is rising.1
In May 2019, the Centers for Disease Control and Prevention (CDC) published a Vital Signs document focused on preventable maternal deaths.2 It affirmed that about 60% of the 700 pregnancy-related deaths that occur annually in the United States are preventable, and it provided important information on when and why these deaths occur.
Among the CDC findings, about:
- one-third of deaths (31%) occurred during pregnancy (before delivery)
- one-third (36%) occurred at delivery or in the week after
- one-third (33%) occurred 1 week to 1 year postpartum.
In addition, the CDC highlighted that:
- Heart disease and stroke caused more than 1 in 3 deaths (34%). Infections and severe bleeding were other leading causes of death.
- Black and American Indian/Alaska Native women were about 3 times as likely to die from a pregnancy-related cause as white women.
The American College of Obstetricians and Gynecologists (ACOG), under the leadership of President Lisa Hollier, MD, MPH (2018–2019), fully embraced the challenge and responsibility of meaningfully improving health care for every mom. In this article, I review some of the critical steps taken in 2018 and preview ACOG’s continued commitment for 2019 and beyond.

Efforts succeed: Bills are now laws of the land
ACOG and our partner organizations, including the Society for Maternal-Fetal Medicine and the March of Dimes, have long recognized the value of state-based maternal mortality review committees (MMRCs) in slowing and reversing the rate of maternal mortality. An MMRC brings together local experts to examine the causes of maternal deaths—not to find fault, but to find ways to prevent future deaths. With the right framework and support, MMRCs already are providing us with data and driving policy recommendations.
Supporting MMRCs in all states. With this in mind, ACOG helped pass and push to enactment HR 1318, the Preventing Maternal Deaths Act of 2018 (Public Law No. 115-344), a bipartisan bill designed to help develop and provide support for MMRCs in every state. The bill was introduced in the US House of Representatives by Rep. Jaime Herrera Beutler (R-WA) and Rep. Diana DeGette (D-CO) and in the US Senate by Sen. Heidi Heitkamp (D-ND) and Sen. Shelley Moore Capito (R-WV). ACOG Fellow and US Rep. Michael Burgess, MD (R-TX), also was instrumental in the bill’s success. The CDC is actively working toward implementation of this law, and grantees are expected to be announced by the end of September.
Continue to: In addition, ACOG worked with Congress...
In addition, ACOG worked with Congress to secure $50 million in federal funding to reduce maternal mortality, allocated thusly:
- $12 million to support state MMRCs
- $3 million to support the Alliance for Innovation on Maternal Health
- $23 million for State Maternal Health Innovation Program grants
- $12 million to address maternal mortality in the Healthy Start program.
As these federal congressional initiatives worked their way into law, the states actively supported MMRCs as well. As of this writing, only 3 states—North Dakota, South Dakota, and Wyoming—have not yet developed an MMRC.3
Filling the gaps in ObGyn care. Another key ACOG-sponsored bill signed into law will help bring more ObGyns into shortage areas. Sponsored by Rep. Burgess, Rep. Anna Eshoo (D-CA), and Rep. Lucille Roybal-Allard (D-CA) and by Sen. Tammy Baldwin (D-WI) and Sen. Lisa Murkowski (R-AK), the Improving Access to Maternity Care Act (Public Law No. 115-320) requires the Department of Health and Human Services to identify maternity health professional target areas for use by the National Health Service Corps to bring ObGyns to where they are most needed.
Following up on that new law, ACOG currently is working closely with the American Academy of Family Physicians (AAFP) and the National Rural Health Association (NRHA) on the unique challenges women in rural areas face in accessing maternity and other women’s health care services. In June, Dr. Hollier represented ACOG at the Rural Maternal Health forum, which was convened by the Centers for Medicare and Medicaid and sponsored by ACOG, AAFP, and NRHA.4 We are pursuing policies designed to increase the number of ObGyns and other physicians who choose to train in rural areas and increase the clinical use of telehealth to help connect rural physicians and patients with subspecialists in urban areas.
Projects in the works
Congress is ready to do more. Already, 5 ACOG-supported bills have been introduced, including bills that extend women’s Medicaid coverage to 12 months postpartum (consistent with coverage for babies), support state perinatal quality collaboratives, and more. This interest is augmented by the work of the recently formed congressional Black Maternal Health Caucus, focused on reducing racial disparities in health care. In July, ACOG joined 12 members of Congress in a caucus summit to partner with these important congressional allies.
ACOG is expanding support for these legislative efforts through our work with another important ally, the American Medical Association (AMA). ACOG’s delegation to the 2019 Annual Meeting of the AMA House of Delegates in June scored important policy wins, including AMA support for Medicaid coverage for women 12 months postpartum and improving access to care in rural communities.
There is momentum on Capitol Hill to take action on these important issues, and ACOG’s priority is to ensure that any legislative package complements the important work many ObGyns are already doing to improve maternal health outcomes. ACOG has an important seat at the table and will continue to advocate each and every day for your practices and your patients as Congress deliberates legislative action.
Continue to: Your voice matters...
Your voice matters
Encourage your representatives in the House and the Senate to support ACOG-endorsed legislation and be sure they know the importance of ensuring access to women’s health care in your community. Get involved in advocacy; start by visiting the ACOG advocacy web page (www.acog.org/advocacy). Also note that members of Congress are back in their home states during seasonal breaks and many hold town halls and constituent meetings. The health of moms and babies is always an important issue, and you are the expert.
ACOG’s commitment to ensuring healthy moms and babies, and ensuring that our members can continue providing high-quality care, runs through everything we do.
Acknowledgments
The author thanks ACOG former Vice President for Health Policy Barbara Levy, MD, ACOG Senior Director Jeanne Mahoney, and ACOG Federal Affairs Director Rachel Tetlow for their helpful review and comments.
Real progress was achieved in 2018 in the effort to reduce the US maternal mortality rate, the highest of any developed nation and where women of color are 3 to 4 times more likely than others to die of childbirth-related causes. Importantly, the United States is the only nation other than Afghanistan and Sudan where the rate is rising.1
In May 2019, the Centers for Disease Control and Prevention (CDC) published a Vital Signs document focused on preventable maternal deaths.2 It affirmed that about 60% of the 700 pregnancy-related deaths that occur annually in the United States are preventable, and it provided important information on when and why these deaths occur.
Among the CDC findings, about:
- one-third of deaths (31%) occurred during pregnancy (before delivery)
- one-third (36%) occurred at delivery or in the week after
- one-third (33%) occurred 1 week to 1 year postpartum.
In addition, the CDC highlighted that:
- Heart disease and stroke caused more than 1 in 3 deaths (34%). Infections and severe bleeding were other leading causes of death.
- Black and American Indian/Alaska Native women were about 3 times as likely to die from a pregnancy-related cause as white women.
The American College of Obstetricians and Gynecologists (ACOG), under the leadership of President Lisa Hollier, MD, MPH (2018–2019), fully embraced the challenge and responsibility of meaningfully improving health care for every mom. In this article, I review some of the critical steps taken in 2018 and preview ACOG’s continued commitment for 2019 and beyond.

Efforts succeed: Bills are now laws of the land
ACOG and our partner organizations, including the Society for Maternal-Fetal Medicine and the March of Dimes, have long recognized the value of state-based maternal mortality review committees (MMRCs) in slowing and reversing the rate of maternal mortality. An MMRC brings together local experts to examine the causes of maternal deaths—not to find fault, but to find ways to prevent future deaths. With the right framework and support, MMRCs already are providing us with data and driving policy recommendations.
Supporting MMRCs in all states. With this in mind, ACOG helped pass and push to enactment HR 1318, the Preventing Maternal Deaths Act of 2018 (Public Law No. 115-344), a bipartisan bill designed to help develop and provide support for MMRCs in every state. The bill was introduced in the US House of Representatives by Rep. Jaime Herrera Beutler (R-WA) and Rep. Diana DeGette (D-CO) and in the US Senate by Sen. Heidi Heitkamp (D-ND) and Sen. Shelley Moore Capito (R-WV). ACOG Fellow and US Rep. Michael Burgess, MD (R-TX), also was instrumental in the bill’s success. The CDC is actively working toward implementation of this law, and grantees are expected to be announced by the end of September.
Continue to: In addition, ACOG worked with Congress...
In addition, ACOG worked with Congress to secure $50 million in federal funding to reduce maternal mortality, allocated thusly:
- $12 million to support state MMRCs
- $3 million to support the Alliance for Innovation on Maternal Health
- $23 million for State Maternal Health Innovation Program grants
- $12 million to address maternal mortality in the Healthy Start program.
As these federal congressional initiatives worked their way into law, the states actively supported MMRCs as well. As of this writing, only 3 states—North Dakota, South Dakota, and Wyoming—have not yet developed an MMRC.3
Filling the gaps in ObGyn care. Another key ACOG-sponsored bill signed into law will help bring more ObGyns into shortage areas. Sponsored by Rep. Burgess, Rep. Anna Eshoo (D-CA), and Rep. Lucille Roybal-Allard (D-CA) and by Sen. Tammy Baldwin (D-WI) and Sen. Lisa Murkowski (R-AK), the Improving Access to Maternity Care Act (Public Law No. 115-320) requires the Department of Health and Human Services to identify maternity health professional target areas for use by the National Health Service Corps to bring ObGyns to where they are most needed.
Following up on that new law, ACOG currently is working closely with the American Academy of Family Physicians (AAFP) and the National Rural Health Association (NRHA) on the unique challenges women in rural areas face in accessing maternity and other women’s health care services. In June, Dr. Hollier represented ACOG at the Rural Maternal Health forum, which was convened by the Centers for Medicare and Medicaid and sponsored by ACOG, AAFP, and NRHA.4 We are pursuing policies designed to increase the number of ObGyns and other physicians who choose to train in rural areas and increase the clinical use of telehealth to help connect rural physicians and patients with subspecialists in urban areas.
Projects in the works
Congress is ready to do more. Already, 5 ACOG-supported bills have been introduced, including bills that extend women’s Medicaid coverage to 12 months postpartum (consistent with coverage for babies), support state perinatal quality collaboratives, and more. This interest is augmented by the work of the recently formed congressional Black Maternal Health Caucus, focused on reducing racial disparities in health care. In July, ACOG joined 12 members of Congress in a caucus summit to partner with these important congressional allies.
ACOG is expanding support for these legislative efforts through our work with another important ally, the American Medical Association (AMA). ACOG’s delegation to the 2019 Annual Meeting of the AMA House of Delegates in June scored important policy wins, including AMA support for Medicaid coverage for women 12 months postpartum and improving access to care in rural communities.
There is momentum on Capitol Hill to take action on these important issues, and ACOG’s priority is to ensure that any legislative package complements the important work many ObGyns are already doing to improve maternal health outcomes. ACOG has an important seat at the table and will continue to advocate each and every day for your practices and your patients as Congress deliberates legislative action.
Continue to: Your voice matters...
Your voice matters
Encourage your representatives in the House and the Senate to support ACOG-endorsed legislation and be sure they know the importance of ensuring access to women’s health care in your community. Get involved in advocacy; start by visiting the ACOG advocacy web page (www.acog.org/advocacy). Also note that members of Congress are back in their home states during seasonal breaks and many hold town halls and constituent meetings. The health of moms and babies is always an important issue, and you are the expert.
ACOG’s commitment to ensuring healthy moms and babies, and ensuring that our members can continue providing high-quality care, runs through everything we do.
Acknowledgments
The author thanks ACOG former Vice President for Health Policy Barbara Levy, MD, ACOG Senior Director Jeanne Mahoney, and ACOG Federal Affairs Director Rachel Tetlow for their helpful review and comments.
- Council on Patient Safety in Women's Health Care. Alliance for Innovation on Maternal Health Program. https://safehealthcareforeverywoman.org/aim-program/. Accessed August 19, 2019.
- Centers for Disease Control and Prevention. Vital signs: pregnancy-related deaths. https://www.cdc.gov/vitalsigns/maternal-deaths/index.html. Accessed August 19, 2019.
- American College of Obstetricians and Gynecologists. State Maternal Mortality Review Committees, PQCs, and AIM. https://www.acog.org/-/media/Departments/Government-Relations-and-Outreach/MMRC_AIM-State-Fact-Sheet_Mar-2019.pdf. Accessed August 19, 2019.
- Centers for Medicare and Medicaid Services. A conversation on maternal health care in rural communities: charting a path to improved access, quality and outcomes. June 12, 2019. https://www.cms.gov/About-CMS/Agency-Information/OMH/equity-initiatives/rural-health/rural-maternal-health.html. Accessed August 19, 2019.
- Council on Patient Safety in Women's Health Care. Alliance for Innovation on Maternal Health Program. https://safehealthcareforeverywoman.org/aim-program/. Accessed August 19, 2019.
- Centers for Disease Control and Prevention. Vital signs: pregnancy-related deaths. https://www.cdc.gov/vitalsigns/maternal-deaths/index.html. Accessed August 19, 2019.
- American College of Obstetricians and Gynecologists. State Maternal Mortality Review Committees, PQCs, and AIM. https://www.acog.org/-/media/Departments/Government-Relations-and-Outreach/MMRC_AIM-State-Fact-Sheet_Mar-2019.pdf. Accessed August 19, 2019.
- Centers for Medicare and Medicaid Services. A conversation on maternal health care in rural communities: charting a path to improved access, quality and outcomes. June 12, 2019. https://www.cms.gov/About-CMS/Agency-Information/OMH/equity-initiatives/rural-health/rural-maternal-health.html. Accessed August 19, 2019.
Ocular Complications of Atopic Dermatitis
Atopic dermatitis (AD) is a chronic inflammatory skin condition with a lifetime prevalence of 15% to 20% in industrialized countries.1 It affects both children and adults and is predominantly characterized by a waxing and waning course of eczematous skin lesions and pruritus. In recent years, there is increasing recognition that AD can present with extracutaneous findings. Large-scale epidemiologic studies have reported a notably higher prevalence of ophthalmic complications in the AD population compared to the general population, in a severity-dependent manner.2,3 Potential complications include blepharitis, keratoconjunctivitis, keratoconus, glaucoma, cataracts, retinal detachment, ophthalmic herpes simplex virus infections, and dupilumab-associated ocular complications.
The etiology of each ocular complication in the context of AD is complex and likely multifactorial. Intrinsic immune dysregulation, physical trauma from eye rubbing, AD medication side effects, and genetics all have been speculated to play a role.2 Some of these ocular complications have a chronic course, while others present with sudden onset of symptoms; many of them can result in visual impairment if undiagnosed or left untreated. This article reviews several of the most common ocular comorbidities associated with AD. We discuss the clinical presentation, pathophysiology, and management strategies for each condition.
Blepharitis
Blepharitis, an inflammatory condition of the eyelids, is estimated to affect more than 6% of patients with AD compared to less than 1% of the general population.2 Blepharitis can be classified as anterior or posterior, based on the anatomic location of the affected region relative to the lash margin. Affected individuals may experience pruritus and irritation of the eyelids, tearing, a foreign body or burning sensation, crusting of the eyelids, and photophobia.4 Anterior blepharitis commonly is due to staphylococcal disease, and posterior blepharitis is secondary to structural changes and obstruction of meibomian gland orifices.
Although the pathophysiology is not well defined, xerosis in atopic patients is accompanied by barrier disruption and transepidermal water loss, which promote eyelid skin inflammation.
The mainstay of therapy for atopic blepharitis consists of conventional lid hygiene regimens, such as warm compresses and gentle scrubbing of the lid margins to remove crust and debris, which can be done with nonprescription cleansers, pads, and baby shampoos. Acute exacerbations may require topical antibiotics (ie, erythromycin or bacitracin applied to the lid margins once daily), topical calcineurin inhibitors (ie, cyclosporine ophthalmic emulsion 0.05%), or low-potency topical corticosteroids (ie, fluorometholone 0.1% or loteprednol etabonate 0.5% ophthalmic suspensions).5 Due to potential side effects of medications, especially topical corticosteroids, patients should be referred to ophthalmologists for definitive diagnosis and treatment.
Keratoconjunctivitis
Atopic keratoconjunctivitis (AKC) is a noninfectious inflammatory condition of the cornea and conjunctiva that occurs in an estimated 25% to 42% of patients with AD.6,7 It frequently presents in late adolescence and has a peak incidence between 30 and 50 years of age.8 The symptoms of AKC include ocular pruritus, redness, ropy mucoid discharge, burning discomfort, photophobia, and blurring of vision. Corneal involvement can progress to corneal neovascularization and punctate or macroepithelial erosions and ulcerations, which increase the risk for corneal scarring and visual impairment.7
Keratoconjunctivitis is a complex inflammatory disease characterized by infiltration of the conjunctival epithelium by eosinophils, mast cells, and lymphocytes. On examination, patients frequently are found to have concurrent AD of the periorbital skin as well as papillary hypertrophy of the tarsal conjunctiva with accompanying fibrosis, which can lead to entropion (turning inward of the lid margins and lashes) in severe cases.7 Ophthalmic evaluation is strongly recommended for patients with AKC to control symptoms, to limit exacerbations, and to prevent sight-threatening inflammation leading to vision loss. Treatment can be challenging given the chronicity of the condition and may require multiple treatment arms. Conservative measures include cool compresses and treatment with ophthalmic eye drops containing antihistamines (ie, ketotifen 0.025% [available over-the-counter]) and mast cell stabilizers (ie, olopatadine ophthalmic solution 0.1%).8 Atopic keratoconjunctivitis exacerbations may require short-term use of topical steroids or calcineurin inhibitors, or systemic equivalents for refractory cases.6 Long-term maintenance therapy typically consists of proper eye hygiene and steroid-sparing agents that reduce ocular inflammation, such as topical cyclosporine and tacrolimus, neither of which are associated with increased intraocular pressure (IOP)(Figure 1).8 Cornea disease resulting from chronic conjunctival/lid microtrauma can be managed with soft or scleral contact lenses.

Keratoconus
Keratoconus is a noninflammatory ocular disorder characterized by progressive thinning and conelike protrusion of the cornea. The corneal topographic changes result in high irregular astigmatism and reduced visual acuity, which can manifest as image blurring or distortion (Figure 2).2,9 Multiple case series and controlled studies have reported a positive association between keratoconus and a history of atopic disease.10,11
The precise etiology of keratoconus in the context of AD is unclear and likely is multifactorial. Habitual eye rubbing from periocular pruritus and discomfort has been reported to be a notable contributor to keratoconus.12 In addition, intrinsic inflammation and imbalance of cytokines and proteases also may contribute to development of keratoconus.13
Keratoconus is a progressive condition that can severely impact vision, making it critical to diagnose patients before irreversible vision loss occurs. Individuals with risk factors, such as AD of the eyelids, history of eye rubbing, or family history of keratoconus, should be advised to receive routine vision screening for worsening astigmatism, especially during the first few decades of life when keratoconus progresses rapidly.
The conservative management for early keratoconus includes glasses and gas-permeable contact lenses for correction of visual acuity and astigmatism. For advanced keratoconus, scleral lenses often are prescribed. These large-diameter, gas-permeable lenses are designed to rest on the sclera and arch over the entire cornea.9 Alternatively, corneal collagen cross-linking is a newer technique that utilizes riboflavin and UVA irradiation to strengthen the corneal tissue. It has proven to be safe and effective in slowing or stopping the progression of keratoconus, particularly when treated at the early stage, and received US Food and Drug Administration approval in 2016.9
Glaucoma
Glaucoma is a well-known complication of AD and can lead to irreversible ocular hypertension and optic nerve damage. Corticosteroid use is a major risk factor for glaucoma, and the rise in IOP is thought to be due to increased aqueous outflow resistance.14
Multiple case reports have linked glaucoma to long-term use of potent topical corticosteroids in the facial and palpebral regions, which has been attributed to direct steroid contact and absorption by ocular tissues, as glaucoma rarely occurs with topical steroid application elsewhere on the body.15-17 Systemic steroids (ie, prednisolone) taken for more than 8 weeks also have been associated with a marked rise in IOP.18
Certain risk factors may predispose a steroid user to increased IOP, including existing open-angle glaucoma, diabetes mellitus, collagen disease, and high myopia.15,19 Steroid responders and younger individuals also demonstrate increased sensitivity to steroids.20
Given that glaucoma often is asymptomatic until advanced stages, early detection is the key for proper intervention. Periodic glaucoma screening by an ophthalmologist would be appropriate for known steroid responders, as well as patients with a prolonged history of topical steroid application in the palpebral region and systemic steroid use, family history of glaucoma, or known ocular pathology.21 Furthermore, patients with concurrent glaucoma and AD should be jointly managed by dermatology and ophthalmology, and systemic and topical corticosteroid use should be minimized in favor of alterative agents such as calcineurin inhibitors.22
In addition to steroid-induced glaucoma, intrinsic atopic glaucoma recently has been proposed as a clinical entity and is characterized by increased inflammatory cytokines—IL-8 and CCL2—in the aqueous humor and abnormal accumulation of fibers in corneoscleral meshwork.23
Cataracts
Cataracts are estimated to affect 8% to 25% of patients with AD.21,24 Unlike age-related cataracts, cataracts associated with AD are observed in adolescents and young adults in addition to the older population. The progression of lenticular opacity can rapidly occur and has been reported to coincide with AD flares.25,26
Patients with AD typically present with anterior or posterior subcapsular cataracts instead of nuclear and cortical cataracts, which are more common in the general population.27,28 Anterior subcapsular cataracts are more specific to AD, whereas posterior subcapsular cataracts are associated with both prolonged corticosteroid use and AD.26 Children generally are more sensitive to steroids than adults and may develop cataracts more rapidly and at lower concentrations.29
The pathophysiology of cataract formation and progression in the context of AD is multifactorial. Cataract patients with AD have compromised blood-retinal barrier integrity as well as increased oxidative damage in the lens.30,31 Genetics and blunt trauma from eye rubbing are thought to play a role, and the latter has been associated with faster progression of cataracts.28 In contrast, corticosteroid-induced cataracts likely are caused by transcriptional changes and disrupted osmotic balance in the lens fibers, which can lead to fiber rupture and lens opacification.26,32 Systemic corticosteroids show the strongest association with cataract development, but inhaled and topical steroids also have been implicated.26
Although cataracts can be surgically corrected, prevention is critical. Patients with early-onset periorbital AD, prolonged use of topical or systemic corticosteroids, and family history of cataracts should be routinely screened. Anterior and posterior subcapsular cataracts are diagnosed with red reflex examinations that can be readily performed by the primary care physician or ophthalmologist.33 Atopic dermatitis patients with cataracts should be advised to use calcineurin inhibitors and alternative treatments in place of corticosteroids.
Retinal Detachment
Retinal detachment (RD) is a serious complication of AD that can present in individuals younger than 35 years. The incidence of RD in patients with AD has been estimated to be 4% to 8%.34 Retinal detachment manifests with visual disturbances such as flashing lights, shadows, visual field defect, and blurring of vision, but also may occur in the absence of vision changes.35,36
Across multiple case series, patients who developed RD were consistently found to have AD in the facial or periorbital region and a history of chronic eye rubbing. Multiple patients also presented with concurrent proliferative vitreoretinopathy, lens subluxation, and/or cataracts.35,37 The mechanism for RD has been attributed to ocular contusion from vigorous eye rubbing, as fundus findings between traumatic and AD-associated RD are similarly characterized by tractional breaks in the retina at vitreous base borders.37
Avoidance of eye rubbing and optimized treatment of facial AD may help prevent RD in patients with AD. Furthermore, all patients with symptoms of RD should be immediately referred to ophthalmology for surgical repair.
Herpetic Ocular Disease
Ocular herpes simplex virus infections cause ocular pain and are associated with notable visual morbidity, as recurrences can result in irreversible corneal scarring and neovascularization. Two retrospective case-control studies independently reported that individuals with a history of AD are at greater risk for herpetic ocular disease compared to age-matched controls.38,39 Furthermore, atopic disease is associated with higher recurrence rates and slower regeneration of the corneal epithelium.40
These findings suggest that AD patients with a history of recurrent herpetic ocular diseases should be closely monitored and treated with antiviral prophylaxis and/or topical corticosteroids, depending on the type of keratitis (epithelial or stromal).40 Furthermore, active ocular herpetic infections warrant urgent referral to an ophthalmologist.
Dupilumab-Associated Ocular Complications
Dupilumab, a monoclonal antibody that blocks IL-4 and IL-13 signaling, is the first biologic therapy to be approved for treatment of moderate to severe AD. Prior clinical trials have described a higher incidence of anterior conjunctivitis in dupilumab-treated AD patients (5%–28%) compared to placebo (2%–11%).41 Of note, the incidence may be as high as 70%, as reported in a recent case series.42 Interestingly, independent trials assessing dupilumab treatment in asthma, nasal polyposis, and eosinophilic esophagitis patients did not observe a higher incidence of conjunctivitis in dupilumab-treated patients compared to placebo, suggesting an AD-specific mechanism.43
Prominent features of dupilumab-associated conjunctivitis include hyperemia of the conjunctiva and limbus, in addition to ocular symptoms such as tearing, burning, and bilateral decrease in visual acuity. Marked reduction of conjunctival goblet cells has been reported.44 In addition to conjunctivitis, blepharitis also has been reported during dupilumab treatment.45
Standardized treatment guidelines for dupilumab-associated ocular complications have not yet been established. Surprisingly, antihistamine eye drops appear to be inefficacious in the treatment of dupilumab-associated conjunctivitis.41 However, the condition has been successfully managed with topical steroids (fluorometholone ophthalmic suspension 0.1%) and tacrolimus ointment 0.03%.41 Lifitegrast, an anti-inflammatory agent approved for chronic dry eye, also has been suggested as a treatment option for patients refractory to topical steroids.45 Alternatively, cessation of dupilumab could be considered in AD patients who experience severe ocular complications. Atopic dermatitis patients taking dupilumab who have any concerning signs for ocular complications should be referred to an ophthalmologist for further diagnosis and management.
Conclusion
Practicing dermatologists likely will encounter patients with concurrent AD and ocular complications. Although eye examinations are not routinely performed in the care of AD patients, dermatologists can proactively inquire about ocular symptoms and monitor patients longitudinally. Early diagnosis and treatment of these ocular conditions can prevent vision loss in these patients. Furthermore, symptomatic control of AD and careful consideration of the side-effect profiles of medications can potentially reduce the incidence of ocular complications in individuals with AD.
Patients with visual concerns or risk factors, such as a history of vigorous eye rubbing or chronic corticosteroid use, should be jointly managed with an ophthalmologist for optimized care. Moreover, acute exacerbations of ocular symptoms and visual deterioration warrant urgent referral to ophthalmology.
- Weidinger S, Beck LA, Bieber T, et al. Atopic dermatitis. Nat Rev Dis Primers. 2018;4:1.
- Govind K, Whang K, Khanna R, et al. Atopic dermatitis is associated with increased prevalence of multiple ocular comorbidities. J Allergy Clin Immunol Pract. 2019;7:298-299.
- Thyssen JP, Toft PB, Halling-Overgaard AS, et al. Incidence, prevalence, and risk of selected ocular disease in adults with atopic dermatitis. J Am Acad Dermatol. 2017;77:280-286.e281.
- Putnam CM. Diagnosis and management of blepharitis: an optometrist’s perspective. Clin Optom (Auckl). 2016;8:71-78.
- Amescua G, Akpek EK, Farid M, et al. Blepharitis Preferred Practice Pattern®. Ophthalmology. 2019;126:P56-P93.
- Bielory B, Bielory L. Atopic dermatitis and keratoconjunctivitis. Immunol Allergy Clin North Am. 2010;30:323-336.
- Guglielmetti S, Dart JK, Calder V. Atopic keratoconjunctivitis and atopic dermatitis. Curr Opin Allergy Clin Immunol. 2010;10:478-485.
- Chen JJ, Applebaum DS, Sun GS, et al. Atopic keratoconjunctivitis: a review. J Am Acad Dermatol. 2014;70:569-575.
- Andreanos KD, Hashemi K, Petrelli M, et al. Keratoconus treatment algorithm. Ophthalmol Ther. 2017;6:245-262.
- Rahi A, Davies P, Ruben M, et al. Keratoconus and coexisting atopic disease. Br J Ophthalmol. 1977;61:761-764.
- Gasset AR, Hinson WA, Frias JL. Keratoconus and atopic diseases. Ann Ophthalmol. 1978;10:991-994.
- Bawazeer AM, Hodge WG, Lorimer B. Atopy and keratoconus: a multivariate analysis. Br J Ophthalmol. 2000;84:834-836.
- Galvis V, Sherwin T, Tello A, et al. Keratoconus: an inflammatory disorder? Eye (Lond). 2015;29:843-859.
- Clark AF, Wordinger RJ. The role of steroids in outflow resistance. Exp Eye Res. 2009;88:752-759.
- Daniel BS, Orchard D. Ocular side-effects of topical corticosteroids: what a dermatologist needs to know. Australas J Dermatol. 2015;56:164-169.
- Garrott HM, Walland MJ. Glaucoma from topical corticosteroids to the eyelids. Clin Exp Ophthalmol. 2004;32:224-226.
- Aggarwal RK, Potamitis T, Chong NH, et al. Extensive visual loss with topical facial steroids. Eye (Lond). 1993;7(pt 5):664-666.
- Mandapati JS, Metta AK. Intraocular pressure variation in patients on long-term corticosteroids. Indian Dermatol Online J. 2011;2:67-69.
- Jones R 3rd, Rhee DJ. Corticosteroid-induced ocular hypertension and glaucoma: a brief review and update of the literature. Curr Opin Ophthalmol. 2006;17:163-167.
- Tamagawa-Mineoka R, Yasuoka N, Ueta M, et al. Influence of topical steroids on intraocular pressure in patients with atopic dermatitis. Allergol Int. 2018;67:388-391.
- Bercovitch L. Screening for ocular complications in atopic dermatitis. Arch Dermatol. 2011;147:588-589.
- Abramovits W, Hung P, Tong KB. Efficacy and economics of topical calcineurin inhibitors for the treatment of atopic dermatitis. Am J Clin Dermatol. 2006;7:213-222.
- Takakuwa K, Hamanaka T, Mori K, et al. Atopic glaucoma: clinical and pathophysiological analysis. J Glaucoma. 2015;24:662-668.
- Haeck IM, Rouwen TJ, Timmer-de Mik L, et al. Topical corticosteroids in atopic dermatitis and the risk of glaucoma and cataracts. J Am Acad Dermatol. 2011;64:275-281.
- Amemiya T, Matsuda H, Uehara M. Ocular findings in atopic dermatitis with special reference to the clinical features of atopic cataract. Ophthalmologica. 1980;180:129-132.
- Tatham A. Atopic dermatitis, cutaneous steroids and cataracts in children: two case reports. J Med Case Rep. 2008;2:124.
- Chew M, Chiang PP, Zheng Y, et al. The impact of cataract, cataract types, and cataract grades on vision-specific functioning using Rasch analysis. Am J Ophthalmol. 2012;154:29-38.
- Nagaki Y, Hayasaka S, Kadoi C. Cataract progression in patients with atopic dermatitis. J Cataract Refract Surg. 1999;25:96-99.
- Kaye LD, Kalenak JW, Price RL, et al. Ocular implications of long-term prednisone therapy in children. J Pediatr Ophthalmol Strabismus. 1993;30:142-144.
- Matsuo T, Saito H, Matsuo N. Cataract and aqueous flare levels in patients with atopic dermatitis. Am J Ophthalmol. 1997;124:36-39.
- Namazi MR, Handjani F, Amirahmadi M. Increased oxidative activity from hydrogen peroxide may be the cause of the predisposition to cataracts among patients with atopic dermatitis. Med Hypotheses. 2006;66:863-864.
- James ER. The etiology of steroid cataract. J Ocul Pharmacol Ther. 2007;23:403-420.
- Lambert SR, Teng JMC. Assessing whether the cataracts associated with atopic dermatitis are associated with steroids or inflammatory factors. JAMA Ophthalmol. 2018;136:918-919.
- Sasoh M, Mizutani H, Matsubara H, et al. Incidence of retinal detachment associated with atopic dermatitis in Japan: review of cases from 1992 to 2011. Clin Ophthalmol. 2015;9:1129-1134.
- Yoneda K, Okamoto H, Wada Y, et al. Atopic retinal detachment. report of four cases and a review of the literature. Br J Dermatol. 1995;133:586-591.
- Gnana Jothi V, McGimpsey S, Sharkey JA, et al. Retinal detachment repair and cataract surgery in patients with atopic dermatitis. Eye (Lond). 2017;31:1296-1301.
- Oka C, Ideta H, Nagasaki H, et al. Retinal detachment with atopic dermatitis similar to traumatic retinal detachment. Ophthalmology. 1994;101:1050-1054.
- Prabriputaloong T, Margolis TP, Lietman TM, et al. Atopic disease and herpes simplex eye disease: a population-based case-control study. Am J Ophthalmol. 2006;142:745-749.
- Borkar DS, Gonzales JA, Tham VM, et al. Association between atopy and herpetic eye disease: results from the pacific ocular inflammation study. JAMA Ophthalmol. 2014;132:326-331.
- Rezende RA, Hammersmith K, Bisol T, et al. Comparative study of ocular herpes simplex virus in patients with and without self-reported atopy. Am J Ophthalmol. 2006;141:1120-1125.
- Wollenberg A, Ariens L, Thurau S, et al. Conjunctivitis occurring in atopic dermatitis patients treated with dupilumab-clinical characteristics and treatment. J Allergy Clin Immunol Pract. 2018;6:1778-1780.e1.
- Ivert LU, Wahlgren CF, Ivert L, et al. Eye complications during dupilumab treatment for severe atopic dermatitis. Acta Derm Venereol. 2019;99:375-378.
- Akinlade B, Guttman-Yassky E, de Bruin-Weller M, et al. Conjunctivitis in dupilumab clinical trials [published online March 9, 2019]. Br J Dermatol. doi:10.1111/bjd.17869.
- Bakker DS, Ariens LFM, van Luijk C, et al. Goblet cell scarcity and conjunctival inflammation during treatment with dupilumab in patients with atopic dermatitis. Br J Dermatol. 2019;180:1248-1249.
- Zirwas MJ, Wulff K, Beckman K. Lifitegrast add-on treatment for dupilumab-induced ocular surface disease (DIOSD): a novel case report. JAAD Case Rep. 2019;5:34-36.
Atopic dermatitis (AD) is a chronic inflammatory skin condition with a lifetime prevalence of 15% to 20% in industrialized countries.1 It affects both children and adults and is predominantly characterized by a waxing and waning course of eczematous skin lesions and pruritus. In recent years, there is increasing recognition that AD can present with extracutaneous findings. Large-scale epidemiologic studies have reported a notably higher prevalence of ophthalmic complications in the AD population compared to the general population, in a severity-dependent manner.2,3 Potential complications include blepharitis, keratoconjunctivitis, keratoconus, glaucoma, cataracts, retinal detachment, ophthalmic herpes simplex virus infections, and dupilumab-associated ocular complications.
The etiology of each ocular complication in the context of AD is complex and likely multifactorial. Intrinsic immune dysregulation, physical trauma from eye rubbing, AD medication side effects, and genetics all have been speculated to play a role.2 Some of these ocular complications have a chronic course, while others present with sudden onset of symptoms; many of them can result in visual impairment if undiagnosed or left untreated. This article reviews several of the most common ocular comorbidities associated with AD. We discuss the clinical presentation, pathophysiology, and management strategies for each condition.
Blepharitis
Blepharitis, an inflammatory condition of the eyelids, is estimated to affect more than 6% of patients with AD compared to less than 1% of the general population.2 Blepharitis can be classified as anterior or posterior, based on the anatomic location of the affected region relative to the lash margin. Affected individuals may experience pruritus and irritation of the eyelids, tearing, a foreign body or burning sensation, crusting of the eyelids, and photophobia.4 Anterior blepharitis commonly is due to staphylococcal disease, and posterior blepharitis is secondary to structural changes and obstruction of meibomian gland orifices.
Although the pathophysiology is not well defined, xerosis in atopic patients is accompanied by barrier disruption and transepidermal water loss, which promote eyelid skin inflammation.
The mainstay of therapy for atopic blepharitis consists of conventional lid hygiene regimens, such as warm compresses and gentle scrubbing of the lid margins to remove crust and debris, which can be done with nonprescription cleansers, pads, and baby shampoos. Acute exacerbations may require topical antibiotics (ie, erythromycin or bacitracin applied to the lid margins once daily), topical calcineurin inhibitors (ie, cyclosporine ophthalmic emulsion 0.05%), or low-potency topical corticosteroids (ie, fluorometholone 0.1% or loteprednol etabonate 0.5% ophthalmic suspensions).5 Due to potential side effects of medications, especially topical corticosteroids, patients should be referred to ophthalmologists for definitive diagnosis and treatment.
Keratoconjunctivitis
Atopic keratoconjunctivitis (AKC) is a noninfectious inflammatory condition of the cornea and conjunctiva that occurs in an estimated 25% to 42% of patients with AD.6,7 It frequently presents in late adolescence and has a peak incidence between 30 and 50 years of age.8 The symptoms of AKC include ocular pruritus, redness, ropy mucoid discharge, burning discomfort, photophobia, and blurring of vision. Corneal involvement can progress to corneal neovascularization and punctate or macroepithelial erosions and ulcerations, which increase the risk for corneal scarring and visual impairment.7
Keratoconjunctivitis is a complex inflammatory disease characterized by infiltration of the conjunctival epithelium by eosinophils, mast cells, and lymphocytes. On examination, patients frequently are found to have concurrent AD of the periorbital skin as well as papillary hypertrophy of the tarsal conjunctiva with accompanying fibrosis, which can lead to entropion (turning inward of the lid margins and lashes) in severe cases.7 Ophthalmic evaluation is strongly recommended for patients with AKC to control symptoms, to limit exacerbations, and to prevent sight-threatening inflammation leading to vision loss. Treatment can be challenging given the chronicity of the condition and may require multiple treatment arms. Conservative measures include cool compresses and treatment with ophthalmic eye drops containing antihistamines (ie, ketotifen 0.025% [available over-the-counter]) and mast cell stabilizers (ie, olopatadine ophthalmic solution 0.1%).8 Atopic keratoconjunctivitis exacerbations may require short-term use of topical steroids or calcineurin inhibitors, or systemic equivalents for refractory cases.6 Long-term maintenance therapy typically consists of proper eye hygiene and steroid-sparing agents that reduce ocular inflammation, such as topical cyclosporine and tacrolimus, neither of which are associated with increased intraocular pressure (IOP)(Figure 1).8 Cornea disease resulting from chronic conjunctival/lid microtrauma can be managed with soft or scleral contact lenses.

Keratoconus
Keratoconus is a noninflammatory ocular disorder characterized by progressive thinning and conelike protrusion of the cornea. The corneal topographic changes result in high irregular astigmatism and reduced visual acuity, which can manifest as image blurring or distortion (Figure 2).2,9 Multiple case series and controlled studies have reported a positive association between keratoconus and a history of atopic disease.10,11

The precise etiology of keratoconus in the context of AD is unclear and likely is multifactorial. Habitual eye rubbing from periocular pruritus and discomfort has been reported to be a notable contributor to keratoconus.12 In addition, intrinsic inflammation and imbalance of cytokines and proteases also may contribute to development of keratoconus.13
Keratoconus is a progressive condition that can severely impact vision, making it critical to diagnose patients before irreversible vision loss occurs. Individuals with risk factors, such as AD of the eyelids, history of eye rubbing, or family history of keratoconus, should be advised to receive routine vision screening for worsening astigmatism, especially during the first few decades of life when keratoconus progresses rapidly.
The conservative management for early keratoconus includes glasses and gas-permeable contact lenses for correction of visual acuity and astigmatism. For advanced keratoconus, scleral lenses often are prescribed. These large-diameter, gas-permeable lenses are designed to rest on the sclera and arch over the entire cornea.9 Alternatively, corneal collagen cross-linking is a newer technique that utilizes riboflavin and UVA irradiation to strengthen the corneal tissue. It has proven to be safe and effective in slowing or stopping the progression of keratoconus, particularly when treated at the early stage, and received US Food and Drug Administration approval in 2016.9
Glaucoma
Glaucoma is a well-known complication of AD and can lead to irreversible ocular hypertension and optic nerve damage. Corticosteroid use is a major risk factor for glaucoma, and the rise in IOP is thought to be due to increased aqueous outflow resistance.14
Multiple case reports have linked glaucoma to long-term use of potent topical corticosteroids in the facial and palpebral regions, which has been attributed to direct steroid contact and absorption by ocular tissues, as glaucoma rarely occurs with topical steroid application elsewhere on the body.15-17 Systemic steroids (ie, prednisolone) taken for more than 8 weeks also have been associated with a marked rise in IOP.18
Certain risk factors may predispose a steroid user to increased IOP, including existing open-angle glaucoma, diabetes mellitus, collagen disease, and high myopia.15,19 Steroid responders and younger individuals also demonstrate increased sensitivity to steroids.20
Given that glaucoma often is asymptomatic until advanced stages, early detection is the key for proper intervention. Periodic glaucoma screening by an ophthalmologist would be appropriate for known steroid responders, as well as patients with a prolonged history of topical steroid application in the palpebral region and systemic steroid use, family history of glaucoma, or known ocular pathology.21 Furthermore, patients with concurrent glaucoma and AD should be jointly managed by dermatology and ophthalmology, and systemic and topical corticosteroid use should be minimized in favor of alterative agents such as calcineurin inhibitors.22
In addition to steroid-induced glaucoma, intrinsic atopic glaucoma recently has been proposed as a clinical entity and is characterized by increased inflammatory cytokines—IL-8 and CCL2—in the aqueous humor and abnormal accumulation of fibers in corneoscleral meshwork.23
Cataracts
Cataracts are estimated to affect 8% to 25% of patients with AD.21,24 Unlike age-related cataracts, cataracts associated with AD are observed in adolescents and young adults in addition to the older population. The progression of lenticular opacity can rapidly occur and has been reported to coincide with AD flares.25,26
Patients with AD typically present with anterior or posterior subcapsular cataracts instead of nuclear and cortical cataracts, which are more common in the general population.27,28 Anterior subcapsular cataracts are more specific to AD, whereas posterior subcapsular cataracts are associated with both prolonged corticosteroid use and AD.26 Children generally are more sensitive to steroids than adults and may develop cataracts more rapidly and at lower concentrations.29
The pathophysiology of cataract formation and progression in the context of AD is multifactorial. Cataract patients with AD have compromised blood-retinal barrier integrity as well as increased oxidative damage in the lens.30,31 Genetics and blunt trauma from eye rubbing are thought to play a role, and the latter has been associated with faster progression of cataracts.28 In contrast, corticosteroid-induced cataracts likely are caused by transcriptional changes and disrupted osmotic balance in the lens fibers, which can lead to fiber rupture and lens opacification.26,32 Systemic corticosteroids show the strongest association with cataract development, but inhaled and topical steroids also have been implicated.26
Although cataracts can be surgically corrected, prevention is critical. Patients with early-onset periorbital AD, prolonged use of topical or systemic corticosteroids, and family history of cataracts should be routinely screened. Anterior and posterior subcapsular cataracts are diagnosed with red reflex examinations that can be readily performed by the primary care physician or ophthalmologist.33 Atopic dermatitis patients with cataracts should be advised to use calcineurin inhibitors and alternative treatments in place of corticosteroids.
Retinal Detachment
Retinal detachment (RD) is a serious complication of AD that can present in individuals younger than 35 years. The incidence of RD in patients with AD has been estimated to be 4% to 8%.34 Retinal detachment manifests with visual disturbances such as flashing lights, shadows, visual field defect, and blurring of vision, but also may occur in the absence of vision changes.35,36
Across multiple case series, patients who developed RD were consistently found to have AD in the facial or periorbital region and a history of chronic eye rubbing. Multiple patients also presented with concurrent proliferative vitreoretinopathy, lens subluxation, and/or cataracts.35,37 The mechanism for RD has been attributed to ocular contusion from vigorous eye rubbing, as fundus findings between traumatic and AD-associated RD are similarly characterized by tractional breaks in the retina at vitreous base borders.37
Avoidance of eye rubbing and optimized treatment of facial AD may help prevent RD in patients with AD. Furthermore, all patients with symptoms of RD should be immediately referred to ophthalmology for surgical repair.
Herpetic Ocular Disease
Ocular herpes simplex virus infections cause ocular pain and are associated with notable visual morbidity, as recurrences can result in irreversible corneal scarring and neovascularization. Two retrospective case-control studies independently reported that individuals with a history of AD are at greater risk for herpetic ocular disease compared to age-matched controls.38,39 Furthermore, atopic disease is associated with higher recurrence rates and slower regeneration of the corneal epithelium.40
These findings suggest that AD patients with a history of recurrent herpetic ocular diseases should be closely monitored and treated with antiviral prophylaxis and/or topical corticosteroids, depending on the type of keratitis (epithelial or stromal).40 Furthermore, active ocular herpetic infections warrant urgent referral to an ophthalmologist.
Dupilumab-Associated Ocular Complications
Dupilumab, a monoclonal antibody that blocks IL-4 and IL-13 signaling, is the first biologic therapy to be approved for treatment of moderate to severe AD. Prior clinical trials have described a higher incidence of anterior conjunctivitis in dupilumab-treated AD patients (5%–28%) compared to placebo (2%–11%).41 Of note, the incidence may be as high as 70%, as reported in a recent case series.42 Interestingly, independent trials assessing dupilumab treatment in asthma, nasal polyposis, and eosinophilic esophagitis patients did not observe a higher incidence of conjunctivitis in dupilumab-treated patients compared to placebo, suggesting an AD-specific mechanism.43
Prominent features of dupilumab-associated conjunctivitis include hyperemia of the conjunctiva and limbus, in addition to ocular symptoms such as tearing, burning, and bilateral decrease in visual acuity. Marked reduction of conjunctival goblet cells has been reported.44 In addition to conjunctivitis, blepharitis also has been reported during dupilumab treatment.45
Standardized treatment guidelines for dupilumab-associated ocular complications have not yet been established. Surprisingly, antihistamine eye drops appear to be inefficacious in the treatment of dupilumab-associated conjunctivitis.41 However, the condition has been successfully managed with topical steroids (fluorometholone ophthalmic suspension 0.1%) and tacrolimus ointment 0.03%.41 Lifitegrast, an anti-inflammatory agent approved for chronic dry eye, also has been suggested as a treatment option for patients refractory to topical steroids.45 Alternatively, cessation of dupilumab could be considered in AD patients who experience severe ocular complications. Atopic dermatitis patients taking dupilumab who have any concerning signs for ocular complications should be referred to an ophthalmologist for further diagnosis and management.
Conclusion
Practicing dermatologists likely will encounter patients with concurrent AD and ocular complications. Although eye examinations are not routinely performed in the care of AD patients, dermatologists can proactively inquire about ocular symptoms and monitor patients longitudinally. Early diagnosis and treatment of these ocular conditions can prevent vision loss in these patients. Furthermore, symptomatic control of AD and careful consideration of the side-effect profiles of medications can potentially reduce the incidence of ocular complications in individuals with AD.
Patients with visual concerns or risk factors, such as a history of vigorous eye rubbing or chronic corticosteroid use, should be jointly managed with an ophthalmologist for optimized care. Moreover, acute exacerbations of ocular symptoms and visual deterioration warrant urgent referral to ophthalmology.
Atopic dermatitis (AD) is a chronic inflammatory skin condition with a lifetime prevalence of 15% to 20% in industrialized countries.1 It affects both children and adults and is predominantly characterized by a waxing and waning course of eczematous skin lesions and pruritus. In recent years, there is increasing recognition that AD can present with extracutaneous findings. Large-scale epidemiologic studies have reported a notably higher prevalence of ophthalmic complications in the AD population compared to the general population, in a severity-dependent manner.2,3 Potential complications include blepharitis, keratoconjunctivitis, keratoconus, glaucoma, cataracts, retinal detachment, ophthalmic herpes simplex virus infections, and dupilumab-associated ocular complications.
The etiology of each ocular complication in the context of AD is complex and likely multifactorial. Intrinsic immune dysregulation, physical trauma from eye rubbing, AD medication side effects, and genetics all have been speculated to play a role.2 Some of these ocular complications have a chronic course, while others present with sudden onset of symptoms; many of them can result in visual impairment if undiagnosed or left untreated. This article reviews several of the most common ocular comorbidities associated with AD. We discuss the clinical presentation, pathophysiology, and management strategies for each condition.
Blepharitis
Blepharitis, an inflammatory condition of the eyelids, is estimated to affect more than 6% of patients with AD compared to less than 1% of the general population.2 Blepharitis can be classified as anterior or posterior, based on the anatomic location of the affected region relative to the lash margin. Affected individuals may experience pruritus and irritation of the eyelids, tearing, a foreign body or burning sensation, crusting of the eyelids, and photophobia.4 Anterior blepharitis commonly is due to staphylococcal disease, and posterior blepharitis is secondary to structural changes and obstruction of meibomian gland orifices.
Although the pathophysiology is not well defined, xerosis in atopic patients is accompanied by barrier disruption and transepidermal water loss, which promote eyelid skin inflammation.
The mainstay of therapy for atopic blepharitis consists of conventional lid hygiene regimens, such as warm compresses and gentle scrubbing of the lid margins to remove crust and debris, which can be done with nonprescription cleansers, pads, and baby shampoos. Acute exacerbations may require topical antibiotics (ie, erythromycin or bacitracin applied to the lid margins once daily), topical calcineurin inhibitors (ie, cyclosporine ophthalmic emulsion 0.05%), or low-potency topical corticosteroids (ie, fluorometholone 0.1% or loteprednol etabonate 0.5% ophthalmic suspensions).5 Due to potential side effects of medications, especially topical corticosteroids, patients should be referred to ophthalmologists for definitive diagnosis and treatment.
Keratoconjunctivitis
Atopic keratoconjunctivitis (AKC) is a noninfectious inflammatory condition of the cornea and conjunctiva that occurs in an estimated 25% to 42% of patients with AD.6,7 It frequently presents in late adolescence and has a peak incidence between 30 and 50 years of age.8 The symptoms of AKC include ocular pruritus, redness, ropy mucoid discharge, burning discomfort, photophobia, and blurring of vision. Corneal involvement can progress to corneal neovascularization and punctate or macroepithelial erosions and ulcerations, which increase the risk for corneal scarring and visual impairment.7
Keratoconjunctivitis is a complex inflammatory disease characterized by infiltration of the conjunctival epithelium by eosinophils, mast cells, and lymphocytes. On examination, patients frequently are found to have concurrent AD of the periorbital skin as well as papillary hypertrophy of the tarsal conjunctiva with accompanying fibrosis, which can lead to entropion (turning inward of the lid margins and lashes) in severe cases.7 Ophthalmic evaluation is strongly recommended for patients with AKC to control symptoms, to limit exacerbations, and to prevent sight-threatening inflammation leading to vision loss. Treatment can be challenging given the chronicity of the condition and may require multiple treatment arms. Conservative measures include cool compresses and treatment with ophthalmic eye drops containing antihistamines (ie, ketotifen 0.025% [available over-the-counter]) and mast cell stabilizers (ie, olopatadine ophthalmic solution 0.1%).8 Atopic keratoconjunctivitis exacerbations may require short-term use of topical steroids or calcineurin inhibitors, or systemic equivalents for refractory cases.6 Long-term maintenance therapy typically consists of proper eye hygiene and steroid-sparing agents that reduce ocular inflammation, such as topical cyclosporine and tacrolimus, neither of which are associated with increased intraocular pressure (IOP)(Figure 1).8 Cornea disease resulting from chronic conjunctival/lid microtrauma can be managed with soft or scleral contact lenses.

Keratoconus
Keratoconus is a noninflammatory ocular disorder characterized by progressive thinning and conelike protrusion of the cornea. The corneal topographic changes result in high irregular astigmatism and reduced visual acuity, which can manifest as image blurring or distortion (Figure 2).2,9 Multiple case series and controlled studies have reported a positive association between keratoconus and a history of atopic disease.10,11

The precise etiology of keratoconus in the context of AD is unclear and likely is multifactorial. Habitual eye rubbing from periocular pruritus and discomfort has been reported to be a notable contributor to keratoconus.12 In addition, intrinsic inflammation and imbalance of cytokines and proteases also may contribute to development of keratoconus.13
Keratoconus is a progressive condition that can severely impact vision, making it critical to diagnose patients before irreversible vision loss occurs. Individuals with risk factors, such as AD of the eyelids, history of eye rubbing, or family history of keratoconus, should be advised to receive routine vision screening for worsening astigmatism, especially during the first few decades of life when keratoconus progresses rapidly.
The conservative management for early keratoconus includes glasses and gas-permeable contact lenses for correction of visual acuity and astigmatism. For advanced keratoconus, scleral lenses often are prescribed. These large-diameter, gas-permeable lenses are designed to rest on the sclera and arch over the entire cornea.9 Alternatively, corneal collagen cross-linking is a newer technique that utilizes riboflavin and UVA irradiation to strengthen the corneal tissue. It has proven to be safe and effective in slowing or stopping the progression of keratoconus, particularly when treated at the early stage, and received US Food and Drug Administration approval in 2016.9
Glaucoma
Glaucoma is a well-known complication of AD and can lead to irreversible ocular hypertension and optic nerve damage. Corticosteroid use is a major risk factor for glaucoma, and the rise in IOP is thought to be due to increased aqueous outflow resistance.14
Multiple case reports have linked glaucoma to long-term use of potent topical corticosteroids in the facial and palpebral regions, which has been attributed to direct steroid contact and absorption by ocular tissues, as glaucoma rarely occurs with topical steroid application elsewhere on the body.15-17 Systemic steroids (ie, prednisolone) taken for more than 8 weeks also have been associated with a marked rise in IOP.18
Certain risk factors may predispose a steroid user to increased IOP, including existing open-angle glaucoma, diabetes mellitus, collagen disease, and high myopia.15,19 Steroid responders and younger individuals also demonstrate increased sensitivity to steroids.20
Given that glaucoma often is asymptomatic until advanced stages, early detection is the key for proper intervention. Periodic glaucoma screening by an ophthalmologist would be appropriate for known steroid responders, as well as patients with a prolonged history of topical steroid application in the palpebral region and systemic steroid use, family history of glaucoma, or known ocular pathology.21 Furthermore, patients with concurrent glaucoma and AD should be jointly managed by dermatology and ophthalmology, and systemic and topical corticosteroid use should be minimized in favor of alterative agents such as calcineurin inhibitors.22
In addition to steroid-induced glaucoma, intrinsic atopic glaucoma recently has been proposed as a clinical entity and is characterized by increased inflammatory cytokines—IL-8 and CCL2—in the aqueous humor and abnormal accumulation of fibers in corneoscleral meshwork.23
Cataracts
Cataracts are estimated to affect 8% to 25% of patients with AD.21,24 Unlike age-related cataracts, cataracts associated with AD are observed in adolescents and young adults in addition to the older population. The progression of lenticular opacity can rapidly occur and has been reported to coincide with AD flares.25,26
Patients with AD typically present with anterior or posterior subcapsular cataracts instead of nuclear and cortical cataracts, which are more common in the general population.27,28 Anterior subcapsular cataracts are more specific to AD, whereas posterior subcapsular cataracts are associated with both prolonged corticosteroid use and AD.26 Children generally are more sensitive to steroids than adults and may develop cataracts more rapidly and at lower concentrations.29
The pathophysiology of cataract formation and progression in the context of AD is multifactorial. Cataract patients with AD have compromised blood-retinal barrier integrity as well as increased oxidative damage in the lens.30,31 Genetics and blunt trauma from eye rubbing are thought to play a role, and the latter has been associated with faster progression of cataracts.28 In contrast, corticosteroid-induced cataracts likely are caused by transcriptional changes and disrupted osmotic balance in the lens fibers, which can lead to fiber rupture and lens opacification.26,32 Systemic corticosteroids show the strongest association with cataract development, but inhaled and topical steroids also have been implicated.26
Although cataracts can be surgically corrected, prevention is critical. Patients with early-onset periorbital AD, prolonged use of topical or systemic corticosteroids, and family history of cataracts should be routinely screened. Anterior and posterior subcapsular cataracts are diagnosed with red reflex examinations that can be readily performed by the primary care physician or ophthalmologist.33 Atopic dermatitis patients with cataracts should be advised to use calcineurin inhibitors and alternative treatments in place of corticosteroids.
Retinal Detachment
Retinal detachment (RD) is a serious complication of AD that can present in individuals younger than 35 years. The incidence of RD in patients with AD has been estimated to be 4% to 8%.34 Retinal detachment manifests with visual disturbances such as flashing lights, shadows, visual field defect, and blurring of vision, but also may occur in the absence of vision changes.35,36
Across multiple case series, patients who developed RD were consistently found to have AD in the facial or periorbital region and a history of chronic eye rubbing. Multiple patients also presented with concurrent proliferative vitreoretinopathy, lens subluxation, and/or cataracts.35,37 The mechanism for RD has been attributed to ocular contusion from vigorous eye rubbing, as fundus findings between traumatic and AD-associated RD are similarly characterized by tractional breaks in the retina at vitreous base borders.37
Avoidance of eye rubbing and optimized treatment of facial AD may help prevent RD in patients with AD. Furthermore, all patients with symptoms of RD should be immediately referred to ophthalmology for surgical repair.
Herpetic Ocular Disease
Ocular herpes simplex virus infections cause ocular pain and are associated with notable visual morbidity, as recurrences can result in irreversible corneal scarring and neovascularization. Two retrospective case-control studies independently reported that individuals with a history of AD are at greater risk for herpetic ocular disease compared to age-matched controls.38,39 Furthermore, atopic disease is associated with higher recurrence rates and slower regeneration of the corneal epithelium.40
These findings suggest that AD patients with a history of recurrent herpetic ocular diseases should be closely monitored and treated with antiviral prophylaxis and/or topical corticosteroids, depending on the type of keratitis (epithelial or stromal).40 Furthermore, active ocular herpetic infections warrant urgent referral to an ophthalmologist.
Dupilumab-Associated Ocular Complications
Dupilumab, a monoclonal antibody that blocks IL-4 and IL-13 signaling, is the first biologic therapy to be approved for treatment of moderate to severe AD. Prior clinical trials have described a higher incidence of anterior conjunctivitis in dupilumab-treated AD patients (5%–28%) compared to placebo (2%–11%).41 Of note, the incidence may be as high as 70%, as reported in a recent case series.42 Interestingly, independent trials assessing dupilumab treatment in asthma, nasal polyposis, and eosinophilic esophagitis patients did not observe a higher incidence of conjunctivitis in dupilumab-treated patients compared to placebo, suggesting an AD-specific mechanism.43
Prominent features of dupilumab-associated conjunctivitis include hyperemia of the conjunctiva and limbus, in addition to ocular symptoms such as tearing, burning, and bilateral decrease in visual acuity. Marked reduction of conjunctival goblet cells has been reported.44 In addition to conjunctivitis, blepharitis also has been reported during dupilumab treatment.45
Standardized treatment guidelines for dupilumab-associated ocular complications have not yet been established. Surprisingly, antihistamine eye drops appear to be inefficacious in the treatment of dupilumab-associated conjunctivitis.41 However, the condition has been successfully managed with topical steroids (fluorometholone ophthalmic suspension 0.1%) and tacrolimus ointment 0.03%.41 Lifitegrast, an anti-inflammatory agent approved for chronic dry eye, also has been suggested as a treatment option for patients refractory to topical steroids.45 Alternatively, cessation of dupilumab could be considered in AD patients who experience severe ocular complications. Atopic dermatitis patients taking dupilumab who have any concerning signs for ocular complications should be referred to an ophthalmologist for further diagnosis and management.
Conclusion
Practicing dermatologists likely will encounter patients with concurrent AD and ocular complications. Although eye examinations are not routinely performed in the care of AD patients, dermatologists can proactively inquire about ocular symptoms and monitor patients longitudinally. Early diagnosis and treatment of these ocular conditions can prevent vision loss in these patients. Furthermore, symptomatic control of AD and careful consideration of the side-effect profiles of medications can potentially reduce the incidence of ocular complications in individuals with AD.
Patients with visual concerns or risk factors, such as a history of vigorous eye rubbing or chronic corticosteroid use, should be jointly managed with an ophthalmologist for optimized care. Moreover, acute exacerbations of ocular symptoms and visual deterioration warrant urgent referral to ophthalmology.
- Weidinger S, Beck LA, Bieber T, et al. Atopic dermatitis. Nat Rev Dis Primers. 2018;4:1.
- Govind K, Whang K, Khanna R, et al. Atopic dermatitis is associated with increased prevalence of multiple ocular comorbidities. J Allergy Clin Immunol Pract. 2019;7:298-299.
- Thyssen JP, Toft PB, Halling-Overgaard AS, et al. Incidence, prevalence, and risk of selected ocular disease in adults with atopic dermatitis. J Am Acad Dermatol. 2017;77:280-286.e281.
- Putnam CM. Diagnosis and management of blepharitis: an optometrist’s perspective. Clin Optom (Auckl). 2016;8:71-78.
- Amescua G, Akpek EK, Farid M, et al. Blepharitis Preferred Practice Pattern®. Ophthalmology. 2019;126:P56-P93.
- Bielory B, Bielory L. Atopic dermatitis and keratoconjunctivitis. Immunol Allergy Clin North Am. 2010;30:323-336.
- Guglielmetti S, Dart JK, Calder V. Atopic keratoconjunctivitis and atopic dermatitis. Curr Opin Allergy Clin Immunol. 2010;10:478-485.
- Chen JJ, Applebaum DS, Sun GS, et al. Atopic keratoconjunctivitis: a review. J Am Acad Dermatol. 2014;70:569-575.
- Andreanos KD, Hashemi K, Petrelli M, et al. Keratoconus treatment algorithm. Ophthalmol Ther. 2017;6:245-262.
- Rahi A, Davies P, Ruben M, et al. Keratoconus and coexisting atopic disease. Br J Ophthalmol. 1977;61:761-764.
- Gasset AR, Hinson WA, Frias JL. Keratoconus and atopic diseases. Ann Ophthalmol. 1978;10:991-994.
- Bawazeer AM, Hodge WG, Lorimer B. Atopy and keratoconus: a multivariate analysis. Br J Ophthalmol. 2000;84:834-836.
- Galvis V, Sherwin T, Tello A, et al. Keratoconus: an inflammatory disorder? Eye (Lond). 2015;29:843-859.
- Clark AF, Wordinger RJ. The role of steroids in outflow resistance. Exp Eye Res. 2009;88:752-759.
- Daniel BS, Orchard D. Ocular side-effects of topical corticosteroids: what a dermatologist needs to know. Australas J Dermatol. 2015;56:164-169.
- Garrott HM, Walland MJ. Glaucoma from topical corticosteroids to the eyelids. Clin Exp Ophthalmol. 2004;32:224-226.
- Aggarwal RK, Potamitis T, Chong NH, et al. Extensive visual loss with topical facial steroids. Eye (Lond). 1993;7(pt 5):664-666.
- Mandapati JS, Metta AK. Intraocular pressure variation in patients on long-term corticosteroids. Indian Dermatol Online J. 2011;2:67-69.
- Jones R 3rd, Rhee DJ. Corticosteroid-induced ocular hypertension and glaucoma: a brief review and update of the literature. Curr Opin Ophthalmol. 2006;17:163-167.
- Tamagawa-Mineoka R, Yasuoka N, Ueta M, et al. Influence of topical steroids on intraocular pressure in patients with atopic dermatitis. Allergol Int. 2018;67:388-391.
- Bercovitch L. Screening for ocular complications in atopic dermatitis. Arch Dermatol. 2011;147:588-589.
- Abramovits W, Hung P, Tong KB. Efficacy and economics of topical calcineurin inhibitors for the treatment of atopic dermatitis. Am J Clin Dermatol. 2006;7:213-222.
- Takakuwa K, Hamanaka T, Mori K, et al. Atopic glaucoma: clinical and pathophysiological analysis. J Glaucoma. 2015;24:662-668.
- Haeck IM, Rouwen TJ, Timmer-de Mik L, et al. Topical corticosteroids in atopic dermatitis and the risk of glaucoma and cataracts. J Am Acad Dermatol. 2011;64:275-281.
- Amemiya T, Matsuda H, Uehara M. Ocular findings in atopic dermatitis with special reference to the clinical features of atopic cataract. Ophthalmologica. 1980;180:129-132.
- Tatham A. Atopic dermatitis, cutaneous steroids and cataracts in children: two case reports. J Med Case Rep. 2008;2:124.
- Chew M, Chiang PP, Zheng Y, et al. The impact of cataract, cataract types, and cataract grades on vision-specific functioning using Rasch analysis. Am J Ophthalmol. 2012;154:29-38.
- Nagaki Y, Hayasaka S, Kadoi C. Cataract progression in patients with atopic dermatitis. J Cataract Refract Surg. 1999;25:96-99.
- Kaye LD, Kalenak JW, Price RL, et al. Ocular implications of long-term prednisone therapy in children. J Pediatr Ophthalmol Strabismus. 1993;30:142-144.
- Matsuo T, Saito H, Matsuo N. Cataract and aqueous flare levels in patients with atopic dermatitis. Am J Ophthalmol. 1997;124:36-39.
- Namazi MR, Handjani F, Amirahmadi M. Increased oxidative activity from hydrogen peroxide may be the cause of the predisposition to cataracts among patients with atopic dermatitis. Med Hypotheses. 2006;66:863-864.
- James ER. The etiology of steroid cataract. J Ocul Pharmacol Ther. 2007;23:403-420.
- Lambert SR, Teng JMC. Assessing whether the cataracts associated with atopic dermatitis are associated with steroids or inflammatory factors. JAMA Ophthalmol. 2018;136:918-919.
- Sasoh M, Mizutani H, Matsubara H, et al. Incidence of retinal detachment associated with atopic dermatitis in Japan: review of cases from 1992 to 2011. Clin Ophthalmol. 2015;9:1129-1134.
- Yoneda K, Okamoto H, Wada Y, et al. Atopic retinal detachment. report of four cases and a review of the literature. Br J Dermatol. 1995;133:586-591.
- Gnana Jothi V, McGimpsey S, Sharkey JA, et al. Retinal detachment repair and cataract surgery in patients with atopic dermatitis. Eye (Lond). 2017;31:1296-1301.
- Oka C, Ideta H, Nagasaki H, et al. Retinal detachment with atopic dermatitis similar to traumatic retinal detachment. Ophthalmology. 1994;101:1050-1054.
- Prabriputaloong T, Margolis TP, Lietman TM, et al. Atopic disease and herpes simplex eye disease: a population-based case-control study. Am J Ophthalmol. 2006;142:745-749.
- Borkar DS, Gonzales JA, Tham VM, et al. Association between atopy and herpetic eye disease: results from the pacific ocular inflammation study. JAMA Ophthalmol. 2014;132:326-331.
- Rezende RA, Hammersmith K, Bisol T, et al. Comparative study of ocular herpes simplex virus in patients with and without self-reported atopy. Am J Ophthalmol. 2006;141:1120-1125.
- Wollenberg A, Ariens L, Thurau S, et al. Conjunctivitis occurring in atopic dermatitis patients treated with dupilumab-clinical characteristics and treatment. J Allergy Clin Immunol Pract. 2018;6:1778-1780.e1.
- Ivert LU, Wahlgren CF, Ivert L, et al. Eye complications during dupilumab treatment for severe atopic dermatitis. Acta Derm Venereol. 2019;99:375-378.
- Akinlade B, Guttman-Yassky E, de Bruin-Weller M, et al. Conjunctivitis in dupilumab clinical trials [published online March 9, 2019]. Br J Dermatol. doi:10.1111/bjd.17869.
- Bakker DS, Ariens LFM, van Luijk C, et al. Goblet cell scarcity and conjunctival inflammation during treatment with dupilumab in patients with atopic dermatitis. Br J Dermatol. 2019;180:1248-1249.
- Zirwas MJ, Wulff K, Beckman K. Lifitegrast add-on treatment for dupilumab-induced ocular surface disease (DIOSD): a novel case report. JAAD Case Rep. 2019;5:34-36.
- Weidinger S, Beck LA, Bieber T, et al. Atopic dermatitis. Nat Rev Dis Primers. 2018;4:1.
- Govind K, Whang K, Khanna R, et al. Atopic dermatitis is associated with increased prevalence of multiple ocular comorbidities. J Allergy Clin Immunol Pract. 2019;7:298-299.
- Thyssen JP, Toft PB, Halling-Overgaard AS, et al. Incidence, prevalence, and risk of selected ocular disease in adults with atopic dermatitis. J Am Acad Dermatol. 2017;77:280-286.e281.
- Putnam CM. Diagnosis and management of blepharitis: an optometrist’s perspective. Clin Optom (Auckl). 2016;8:71-78.
- Amescua G, Akpek EK, Farid M, et al. Blepharitis Preferred Practice Pattern®. Ophthalmology. 2019;126:P56-P93.
- Bielory B, Bielory L. Atopic dermatitis and keratoconjunctivitis. Immunol Allergy Clin North Am. 2010;30:323-336.
- Guglielmetti S, Dart JK, Calder V. Atopic keratoconjunctivitis and atopic dermatitis. Curr Opin Allergy Clin Immunol. 2010;10:478-485.
- Chen JJ, Applebaum DS, Sun GS, et al. Atopic keratoconjunctivitis: a review. J Am Acad Dermatol. 2014;70:569-575.
- Andreanos KD, Hashemi K, Petrelli M, et al. Keratoconus treatment algorithm. Ophthalmol Ther. 2017;6:245-262.
- Rahi A, Davies P, Ruben M, et al. Keratoconus and coexisting atopic disease. Br J Ophthalmol. 1977;61:761-764.
- Gasset AR, Hinson WA, Frias JL. Keratoconus and atopic diseases. Ann Ophthalmol. 1978;10:991-994.
- Bawazeer AM, Hodge WG, Lorimer B. Atopy and keratoconus: a multivariate analysis. Br J Ophthalmol. 2000;84:834-836.
- Galvis V, Sherwin T, Tello A, et al. Keratoconus: an inflammatory disorder? Eye (Lond). 2015;29:843-859.
- Clark AF, Wordinger RJ. The role of steroids in outflow resistance. Exp Eye Res. 2009;88:752-759.
- Daniel BS, Orchard D. Ocular side-effects of topical corticosteroids: what a dermatologist needs to know. Australas J Dermatol. 2015;56:164-169.
- Garrott HM, Walland MJ. Glaucoma from topical corticosteroids to the eyelids. Clin Exp Ophthalmol. 2004;32:224-226.
- Aggarwal RK, Potamitis T, Chong NH, et al. Extensive visual loss with topical facial steroids. Eye (Lond). 1993;7(pt 5):664-666.
- Mandapati JS, Metta AK. Intraocular pressure variation in patients on long-term corticosteroids. Indian Dermatol Online J. 2011;2:67-69.
- Jones R 3rd, Rhee DJ. Corticosteroid-induced ocular hypertension and glaucoma: a brief review and update of the literature. Curr Opin Ophthalmol. 2006;17:163-167.
- Tamagawa-Mineoka R, Yasuoka N, Ueta M, et al. Influence of topical steroids on intraocular pressure in patients with atopic dermatitis. Allergol Int. 2018;67:388-391.
- Bercovitch L. Screening for ocular complications in atopic dermatitis. Arch Dermatol. 2011;147:588-589.
- Abramovits W, Hung P, Tong KB. Efficacy and economics of topical calcineurin inhibitors for the treatment of atopic dermatitis. Am J Clin Dermatol. 2006;7:213-222.
- Takakuwa K, Hamanaka T, Mori K, et al. Atopic glaucoma: clinical and pathophysiological analysis. J Glaucoma. 2015;24:662-668.
- Haeck IM, Rouwen TJ, Timmer-de Mik L, et al. Topical corticosteroids in atopic dermatitis and the risk of glaucoma and cataracts. J Am Acad Dermatol. 2011;64:275-281.
- Amemiya T, Matsuda H, Uehara M. Ocular findings in atopic dermatitis with special reference to the clinical features of atopic cataract. Ophthalmologica. 1980;180:129-132.
- Tatham A. Atopic dermatitis, cutaneous steroids and cataracts in children: two case reports. J Med Case Rep. 2008;2:124.
- Chew M, Chiang PP, Zheng Y, et al. The impact of cataract, cataract types, and cataract grades on vision-specific functioning using Rasch analysis. Am J Ophthalmol. 2012;154:29-38.
- Nagaki Y, Hayasaka S, Kadoi C. Cataract progression in patients with atopic dermatitis. J Cataract Refract Surg. 1999;25:96-99.
- Kaye LD, Kalenak JW, Price RL, et al. Ocular implications of long-term prednisone therapy in children. J Pediatr Ophthalmol Strabismus. 1993;30:142-144.
- Matsuo T, Saito H, Matsuo N. Cataract and aqueous flare levels in patients with atopic dermatitis. Am J Ophthalmol. 1997;124:36-39.
- Namazi MR, Handjani F, Amirahmadi M. Increased oxidative activity from hydrogen peroxide may be the cause of the predisposition to cataracts among patients with atopic dermatitis. Med Hypotheses. 2006;66:863-864.
- James ER. The etiology of steroid cataract. J Ocul Pharmacol Ther. 2007;23:403-420.
- Lambert SR, Teng JMC. Assessing whether the cataracts associated with atopic dermatitis are associated with steroids or inflammatory factors. JAMA Ophthalmol. 2018;136:918-919.
- Sasoh M, Mizutani H, Matsubara H, et al. Incidence of retinal detachment associated with atopic dermatitis in Japan: review of cases from 1992 to 2011. Clin Ophthalmol. 2015;9:1129-1134.
- Yoneda K, Okamoto H, Wada Y, et al. Atopic retinal detachment. report of four cases and a review of the literature. Br J Dermatol. 1995;133:586-591.
- Gnana Jothi V, McGimpsey S, Sharkey JA, et al. Retinal detachment repair and cataract surgery in patients with atopic dermatitis. Eye (Lond). 2017;31:1296-1301.
- Oka C, Ideta H, Nagasaki H, et al. Retinal detachment with atopic dermatitis similar to traumatic retinal detachment. Ophthalmology. 1994;101:1050-1054.
- Prabriputaloong T, Margolis TP, Lietman TM, et al. Atopic disease and herpes simplex eye disease: a population-based case-control study. Am J Ophthalmol. 2006;142:745-749.
- Borkar DS, Gonzales JA, Tham VM, et al. Association between atopy and herpetic eye disease: results from the pacific ocular inflammation study. JAMA Ophthalmol. 2014;132:326-331.
- Rezende RA, Hammersmith K, Bisol T, et al. Comparative study of ocular herpes simplex virus in patients with and without self-reported atopy. Am J Ophthalmol. 2006;141:1120-1125.
- Wollenberg A, Ariens L, Thurau S, et al. Conjunctivitis occurring in atopic dermatitis patients treated with dupilumab-clinical characteristics and treatment. J Allergy Clin Immunol Pract. 2018;6:1778-1780.e1.
- Ivert LU, Wahlgren CF, Ivert L, et al. Eye complications during dupilumab treatment for severe atopic dermatitis. Acta Derm Venereol. 2019;99:375-378.
- Akinlade B, Guttman-Yassky E, de Bruin-Weller M, et al. Conjunctivitis in dupilumab clinical trials [published online March 9, 2019]. Br J Dermatol. doi:10.1111/bjd.17869.
- Bakker DS, Ariens LFM, van Luijk C, et al. Goblet cell scarcity and conjunctival inflammation during treatment with dupilumab in patients with atopic dermatitis. Br J Dermatol. 2019;180:1248-1249.
- Zirwas MJ, Wulff K, Beckman K. Lifitegrast add-on treatment for dupilumab-induced ocular surface disease (DIOSD): a novel case report. JAAD Case Rep. 2019;5:34-36.
Practice Points
- Atopic dermatitis (AD) is associated with various ocular comorbidities that can result in permanent vision loss if untreated.
- Timely recognition of ocular complications in AD patients is critical, and dermatologists should proactively inquire about ocular symptoms in the review of systems.
- Patients with ocular symptoms should be jointly managed with ophthalmology.
Quality of Life in Patients With Atopic Dermatitis
Atopic dermatitis (AD) is a chronic, relapsing, inflammatory skin disease typically with childhood onset. In some cases, the condition persists, but AD usually resolves by the time a child reaches adulthood. Prevalence is difficult to estimate but, in developed countries, is approximately 15% to 30% among children and 2% to 10% among adults.1
Atopic dermatitis is characterized by chronically itchy dry skin, weeping erythematous papules and plaques, and lichenification. Furthermore, AD often is associated with other atopic diseases, such as food allergy, allergic rhinitis, and bronchial asthma.
In this article, we review the literature on the quality of life (QOL) of patients with AD. Our goals are to discuss the most common methods for measuring QOL in AD and how to use them; highlight specific alterations of QOL in AD; and review data about QOL of children with AD, which is underrepresented in the medical literature, as studies tend to focus on adults. In addition, we address the importance of assessing QOL in patients with AD due to the psychological burden of the disease.
Quality of Life
The harmful effects of AD can include a range of areas, including
Because QOL is an important instrument used in many AD studies, we call attention to the work of the Harmonising Outcome Measures for Eczema (HOME) initiative, which established a core outcome set for all AD clinical trials to enable comparison of results of individual studies.2 Quality of life was identified in HOME as one of 4 basic outcome measures that should be included in every AD trial (the others are clinician-reported signs, patient-reported symptoms, and long-term control).3 According to the recent agreement, the following QOL instruments should be used: Dermatology Life Quality Index (DLQI) for adults, Children’s Dermatology Life Quality Index (CDLQI) for children, and Infants’ Dermatitis Quality of Life Index (IDQOL) for infants.4
In dermatology, these instruments can be divided into 3 basic categories: generic, dermatology specific, and disease specific.5 Generic QOL questionnaires are beneficial when comparing the QOL of an AD patient to patients with other conditions or to healthy individuals. On the other hand, dermatology-specific and AD-specific methods are more effective instruments for detecting impairments linked directly to the disease and, therefore, are more sensitive to changes in QOL.5 Some of the most frequently used QOL measures5,6 for AD along with their key attributes are
Given that AD is a chronic disease that requires constant care, parents/guardians or the partner of the patient usually are affected as well. To detect this effect, the Family Dermatology Life Quality Index (FDLQI), a dermatology-specific instrument, measures the QOL in family members of dermatology patients.7 The Dermatitis Family Impact (DFI)8 is a disease-specific method for assessing how having a child with AD can impact the QOL of family members; it is a 10-item questionnaire completed by an adult family member. The FDLQI7 and DFI8 both help to understand the secondary impact of the disease.
In contrast, several other methods that also are administered by a parent/guardian assess how the parent perceives the QOL of their child with AD; these methods are essential for small children and infants who cannot answer questions themselves. The IDQOL9 was designed to assess the QOL of patients younger than 4 years using a parent-completed questionnaire. For older children and adolescents aged 4 to 16 years, the CDLQI10 is a widely used instrument; the questionnaire is completed by the child and is available in a cartoon format.10
For patients older than 16 years, 2 important instruments are the DLQI, a generic dermatology instrument, and the Quality of Life Index for Atopic Dermatitis (QoLIAD).11
Clearly it can be troublesome for researchers and clinicians to find the most suitable instrument to evaluate QOL in AD patients. To make this task easier, the European Academy of Dermatology and Venereology Task Force released a position paper with the following recommendations: (1) only validated instruments should be used, and (2) their use should be based on the age of the patients for which the instruments were designed. It is reommended that researchers use a combination of a generic and a dermatology-specific or AD-specific instrument, whereas clinicians should apply a dermatology-specific or AD-specific method, or both.5
Alterations of QOL in AD
Sleep Disturbance in AD
Sleep disorders observed in AD include difficulty falling asleep, frequent waking episodes, shorter sleep duration, and feelings of inadequate sleep, which often result in impairment of daily activity.12,13 Correlation has been found between sleep quality and QOL in both children and adults.14 Approximately 60% of children affected by AD experience a sleep disturbance,15 which seems to correlate well with disease severity.16 A US study found that adults with AD are more likely to experience a sleep disturbance, which often affects daytime functioning and work productivity.13
Financial Aspects and Impact on Work
The financial burden of AD is extensive.17 There are direct medical costs, including medication, visits to the physician, alternative therapies, and nonprescription products. Patients tend to spend relevant money on such items as moisturizers, bath products, antihistamines, topical steroids, and topical antibiotics.18,19 However, it seems that most of the cost of AD is due to indirect and nonmedical costs, including transportation to medical visits; loss of work days; extra childcare; and expenditures associated with lifestyle changes,19,20 such as modifying diet, wearing special clothes, using special bed linens, and purchasing special household items (eg, anti–dust mite vacuum cleaner, humidifier, new carpeting).17,19
Absenteeism from work often is a consequence of physician appointments; in addition, parents/guardians of a child with AD often miss work due to medical care. Even at work, patients (or parents/guardians) often experience decreased work productivity (so-called presenteeism) due to loss of sleep and anxiety.21 In addressing the effects of AD on work life, a systematic literature review found that AD strongly affects sick leave and might have an impact on job choice and change or loss of job.22
Furthermore, according to Su et al,23 the costs of AD are related to disease severity. Moreover, their data suggest that among chronic childhood diseases, the financial burden of AD is greater than the cost of asthma and similar to the cost of diabetes mellitus.23
Association Between QOL and Disease Severity
A large observational study found that improvement in AD severity was followed by an increase in QOL.24 A positive correlation between disease severity and QOL has been found in other studies,25,26 though no correlation or only moderate correlation also has been reported.27 Apparently, in addition to QOL, disease severity scores are substantial parameters in the evaluation of distress caused by AD; the HOME initiative has identified clinician-reported signs and patient-reported symptoms as 2 of 4 core outcomes domains to include in all future AD clinical trials.3 For measuring symptoms, the Patient-Oriented Eczema Measure (POEM) is the recommended instrument.28 Regarding clinical signs, the HOME group named the Eczema Area and Severity Index (EASI) as the preferred instrument.29
Psychological Burden
Stress is a triggering factor for AD, but the connection between skin and mind appears bidirectional. The biological reaction to stress probably lowers the itch threshold and disrupts the skin barrier.30 The Global Burden of Disease Study showed that skin diseases are the fourth leading cause of nonfatal disease burden.31 There are several factors—pruritus, scratch, and pain—that can all lead to sleep deprivation and daytime fatigue. Based on our experience, if lesions develop on visible areas, patients can feel stigmatized, which restricts their social life.
The most common psychological comorbidities of AD are anxiety and depression. In a cross-sectional, multicenter study, there was a significantly higher prevalence of depression (P<.001) and anxiety disorder (P=.02) among patients with common skin diseases compared to a control group.32 In a study that assessed AD patients, researchers found a higher risk of depression and anxiety.33 Suicidal ideation also is more common in the population with AD32,34; a study showed that the risk of suicidal ideation in adolescents was nearly 4-fold in patients with itching skin lesions compared to those without itch.34
According to Linnet and Jemec,35 mental and psychological comorbidities of AD are associated with lower QOL, not with clinical severity. As a result, to improve QOL in AD, one should take care of both dermatological and psychological problems. It has been demonstrated that psychological interventions, such as autogenic training, cognitive-behavioral therapy, relaxation techniques, habit reversal training,36 and hypnotherapy37 might be helpful in individual cases; educational interventions also are recommended.36 With these adjuvant therapies, psychological status, unpleasant clinical symptoms, and QOL could be improved, though further studies are needed to confirm these benefits.
Conclusion
Atopic dermatitis places a notable burden on patients and their families. The degree of burden is probably related to disease severity. For measuring QOL, researchers and clinicians should use validated methods suited to the age of the patients for which they were designed. More studies are needed to assess the effects of different treatments on QOL. Besides pharmacotherapy, psychotherapy and educational programs might be beneficial for improving QOL, another important area to be studied.
- Bieber T. Atopic dermatitis. N Engl J Med. 2008;358:1483-1494.
- Schmitt J, Williams H; HOME Development Group. Harmonising Outcome Measures for Eczema (HOME). report from the First International Consensus Meeting (HOME 1), 24 July 2010, Munich, Germany. Br J Dermatol. 2010;163:1166-1168.
- Schmitt J, Spuls P, Boers M, et al. Towards global consensus on outcome measures for atopic eczema research: results of the HOME II meeting. Allergy. 2012;67:1111-1117.
- Quality of Life (QoL). Harmonising Outcome Measures for Eczema (HOME) website. http://www.homeforeczema.org/research/quality-of-life.aspx. Accessed August 18, 2019.
Chernyshov PV, Tomas-Aragones L, Manolache L, et al; EADV Quality of Life Task Force. Quality of life measurement in atopic dermatitis. Position paper of the European Academy of Dermatology and Venereology (EADV) Task Force on quality of life. J Eur Acad Dermatol Venereol. 2017;31:576-593. - Hill MK, Kheirandish Pishkenari A, Braunberger TL, et al. Recent trends in disease severity and quality of life instruments for patients with atopic dermatitis: a systematic review. J Am Acad Dermatol. 2016;75:906-917.
- Basra MK, Sue-Ho R, Finlay AY. The Family Dermatology Life Quality Index: measuring the secondary impact of skin disease. Br J Dermatol. 2007;156:528-538.
- Dodington SR, Basra MK, Finlay AY, et al. The Dermatitis Family Impact questionnaire: a review of its measurement properties and clinical application. Br J Dermatol. 2013;169:31-46.
- Lewis-Jones MS, Finlay AY, Dykes PJ. The Infants’ Dermatitis Quality of Life Index. Br J Dermatol. 2001;144:104-110.
- Holme SA, Man I, Sharpe JL, et al. The Children’s Dermatology Life Quality Index: validation of the cartoon version. Br J Dermatol. 2003;148:285-290.
- Whalley D, McKenna SP, Dewar AL, et al. A new instrument for assessing quality of life in atopic dermatitis: international development of the Quality of Life Index for Atopic Dermatitis (QoLIAD). Br J Dermatol. 2004;150:274-283.
- Jeon C, Yan D, Nakamura M, et al. Frequency and management of sleep disturbance in adults with atopic dermatitis: a systematic review. Dermatol Ther (Heidelb). 2017;7:349-364.
- Yu SH, Attarian H, Zee P, et al. Burden of sleep and fatigue in US adults with atopic dermatitis. Dermatitis. 2016;27:50-58.
- Kong TS, Han TY, Lee JH, et al. Correlation between severity of atopic dermatitis and sleep quality in children and adults. Ann Dermatol. 2016;28:321-326.
- Fishbein AB, Mueller K, Kruse L, et al. Sleep disturbance in children with moderate/severe atopic dermatitis: a case-control study. J Am Acad Dermatol. 2018;78:336-341.
- Chamlin SL, Mattson CL, Frieden IJ, et al. The price of pruritus: sleep disturbance and cosleeping in atopic dermatitis. Arch Pediatr Adolesc Med. 2005;159:745-750.
- Emerson RM, Williams HC, Allen BR. What is the cost of atopic dermatitis in preschool children? Br J Dermatol. 2001;144:514-522.
- Filanovsky MG, Pootongkam S, Tamburro JE, et al. The financial and emotional impact of atopic dermatitis on children and their families. J Pediatr. 2016;169:284-290.
- Fivenson D, Arnold RJ, Kaniecki DJ, et al. The effect of atopic dermatitis on total burden of illness and quality of life on adults and children in a large managed care organization. J Manag Care Pharm. 2002;8:333-342.
- Carroll CL, Balkrishnan R, Feldman SR, et al. The burden of atopic dermatitis: impact on the patient, family, and society. Pediatr Dermatol. 2005;22:192-199.
- Drucker AM, Wang AR, Qureshi AA. Research gaps in quality of life and economic burden of atopic dermatitis: the National Eczema Association Burden of Disease Audit. JAMA Dermatol. 2016;152:873-874.
- Nørreslet LB, Ebbehøj NE, Ellekilde Bonde JP, et al. The impact of atopic dermatitis on work life—a systematic review. J Eur Acad Dermatol Venereol. 2018;32:23-38.
- Su JC, Kemp AS, Varigos GA, et al. Atopic eczema: its impact on the family and financial cost. Arch Dis Child. 1997;76:159-162.
- Coutanceau C, Stalder JF. Analysis of correlations between patient-oriented SCORAD (PO-SCORAD) and other assessment scores of atopic dermatitis severity and quality of life. Dermatology. 2014;229:248-255.
- Ben-Gashir MA, Seed PT, Hay RJ. Quality of life and disease severity are correlated in children with atopic dermatitis. Br J Dermatol. 2004;150:284-290.
- van Valburg RW, Willemsen MG, Dirven-Meijer PC, et al. Quality of life measurement and its relationship to disease severity in children with atopic dermatitis in general practice. Acta Derm Venereol. 2011;91:147-151.
- Haeck IM, ten Berge O, van Velsen SG, et al. Moderate correlation between quality of life and disease activity in adult patients with atopic dermatitis. J Eur Acad Dermatol Venereol. 2012;26:236-241.
- Spuls PI, Gerbens LAA, Simpson E, et al; HOME initiative collaborators. Patient-Oriented Eczema Measure (POEM), a core instrument to measure symptoms in clinical trials: a Harmonising Outcome Measures for Eczema (HOME) statement. Br J Dermatol. 2017;176:979-984.
- Schmitt J, Spuls PI, Thomas KS, et al; HOME initiative collaborators. The Harmonising Outcome Measures for Eczema (HOME) statement to assess clinical signs of atopic eczema in trials. J Allergy Clin Immunol. 2014;134:800-807.
- Oh SH, Bae BG, Park CO, et al. Association of stress with symptoms of atopic dermatitis. Acta Derm Venereol. 2010;90:582-588.
- Hay RJ, Johns NE, Williams HC, et al. The global burden of skin disease in 2010: an analysis of the prevalence and impact of skin conditions. J Invest Dermatol. 2014;134:1527-1534.
- Dalgard FJ, Gieler U, Tomas-Aragones L, et al. The psychological burden of skin diseases: a cross-sectional multicenter study among dermatological out-patients in 13 European countries. J Invest Dermatol. 2015;135:984-991.
- Cheng CM, Hsu JW, Huang KL, et al. Risk of developing major depressive disorder and anxiety disorders among adolescents and adults with atopic dermatitis: a nationwide longitudinal study. J Affect Disord. 2015;178:60-65.
Halvorsen JA, Lien L, Dalgard F, et al. Suicidal ideation, mental health problems, and social function in adolescents with eczema: a population-based study. J Invest Dermatol. 2014;134:1847-1854. - Linnet J, Jemec GB. An assessment of anxiety and dermatology life quality in patients with atopic dermatitis. Br J Dermatol. 1999;140:268-272.
- Ring J, Alomar A, Bieber T, et al; European Dermatology Forum; European Academy of Dermatology and Venereology; European Task Force on Atopic Dermatitis; European Federation of Allergy; European Society of Pediatric Dermatology; Global Allergy and Asthma European Network. Guidelines for treatment of atopic eczema (atopic dermatitis) Part II. J Eur Acad Dermatol Venereol. 2012;26:1176-1193.
- Perczel K, Gál J. Hypnotherapy of atopic dermatitis in an adult. case report. Orv Hetil. 2016;157:111-115.
Atopic dermatitis (AD) is a chronic, relapsing, inflammatory skin disease typically with childhood onset. In some cases, the condition persists, but AD usually resolves by the time a child reaches adulthood. Prevalence is difficult to estimate but, in developed countries, is approximately 15% to 30% among children and 2% to 10% among adults.1
Atopic dermatitis is characterized by chronically itchy dry skin, weeping erythematous papules and plaques, and lichenification. Furthermore, AD often is associated with other atopic diseases, such as food allergy, allergic rhinitis, and bronchial asthma.
In this article, we review the literature on the quality of life (QOL) of patients with AD. Our goals are to discuss the most common methods for measuring QOL in AD and how to use them; highlight specific alterations of QOL in AD; and review data about QOL of children with AD, which is underrepresented in the medical literature, as studies tend to focus on adults. In addition, we address the importance of assessing QOL in patients with AD due to the psychological burden of the disease.
Quality of Life
The harmful effects of AD can include a range of areas, including
Because QOL is an important instrument used in many AD studies, we call attention to the work of the Harmonising Outcome Measures for Eczema (HOME) initiative, which established a core outcome set for all AD clinical trials to enable comparison of results of individual studies.2 Quality of life was identified in HOME as one of 4 basic outcome measures that should be included in every AD trial (the others are clinician-reported signs, patient-reported symptoms, and long-term control).3 According to the recent agreement, the following QOL instruments should be used: Dermatology Life Quality Index (DLQI) for adults, Children’s Dermatology Life Quality Index (CDLQI) for children, and Infants’ Dermatitis Quality of Life Index (IDQOL) for infants.4
In dermatology, these instruments can be divided into 3 basic categories: generic, dermatology specific, and disease specific.5 Generic QOL questionnaires are beneficial when comparing the QOL of an AD patient to patients with other conditions or to healthy individuals. On the other hand, dermatology-specific and AD-specific methods are more effective instruments for detecting impairments linked directly to the disease and, therefore, are more sensitive to changes in QOL.5 Some of the most frequently used QOL measures5,6 for AD along with their key attributes are

Given that AD is a chronic disease that requires constant care, parents/guardians or the partner of the patient usually are affected as well. To detect this effect, the Family Dermatology Life Quality Index (FDLQI), a dermatology-specific instrument, measures the QOL in family members of dermatology patients.7 The Dermatitis Family Impact (DFI)8 is a disease-specific method for assessing how having a child with AD can impact the QOL of family members; it is a 10-item questionnaire completed by an adult family member. The FDLQI7 and DFI8 both help to understand the secondary impact of the disease.
In contrast, several other methods that also are administered by a parent/guardian assess how the parent perceives the QOL of their child with AD; these methods are essential for small children and infants who cannot answer questions themselves. The IDQOL9 was designed to assess the QOL of patients younger than 4 years using a parent-completed questionnaire. For older children and adolescents aged 4 to 16 years, the CDLQI10 is a widely used instrument; the questionnaire is completed by the child and is available in a cartoon format.10
For patients older than 16 years, 2 important instruments are the DLQI, a generic dermatology instrument, and the Quality of Life Index for Atopic Dermatitis (QoLIAD).11
Clearly it can be troublesome for researchers and clinicians to find the most suitable instrument to evaluate QOL in AD patients. To make this task easier, the European Academy of Dermatology and Venereology Task Force released a position paper with the following recommendations: (1) only validated instruments should be used, and (2) their use should be based on the age of the patients for which the instruments were designed. It is reommended that researchers use a combination of a generic and a dermatology-specific or AD-specific instrument, whereas clinicians should apply a dermatology-specific or AD-specific method, or both.5
Alterations of QOL in AD
Sleep Disturbance in AD
Sleep disorders observed in AD include difficulty falling asleep, frequent waking episodes, shorter sleep duration, and feelings of inadequate sleep, which often result in impairment of daily activity.12,13 Correlation has been found between sleep quality and QOL in both children and adults.14 Approximately 60% of children affected by AD experience a sleep disturbance,15 which seems to correlate well with disease severity.16 A US study found that adults with AD are more likely to experience a sleep disturbance, which often affects daytime functioning and work productivity.13
Financial Aspects and Impact on Work
The financial burden of AD is extensive.17 There are direct medical costs, including medication, visits to the physician, alternative therapies, and nonprescription products. Patients tend to spend relevant money on such items as moisturizers, bath products, antihistamines, topical steroids, and topical antibiotics.18,19 However, it seems that most of the cost of AD is due to indirect and nonmedical costs, including transportation to medical visits; loss of work days; extra childcare; and expenditures associated with lifestyle changes,19,20 such as modifying diet, wearing special clothes, using special bed linens, and purchasing special household items (eg, anti–dust mite vacuum cleaner, humidifier, new carpeting).17,19
Absenteeism from work often is a consequence of physician appointments; in addition, parents/guardians of a child with AD often miss work due to medical care. Even at work, patients (or parents/guardians) often experience decreased work productivity (so-called presenteeism) due to loss of sleep and anxiety.21 In addressing the effects of AD on work life, a systematic literature review found that AD strongly affects sick leave and might have an impact on job choice and change or loss of job.22
Furthermore, according to Su et al,23 the costs of AD are related to disease severity. Moreover, their data suggest that among chronic childhood diseases, the financial burden of AD is greater than the cost of asthma and similar to the cost of diabetes mellitus.23
Association Between QOL and Disease Severity
A large observational study found that improvement in AD severity was followed by an increase in QOL.24 A positive correlation between disease severity and QOL has been found in other studies,25,26 though no correlation or only moderate correlation also has been reported.27 Apparently, in addition to QOL, disease severity scores are substantial parameters in the evaluation of distress caused by AD; the HOME initiative has identified clinician-reported signs and patient-reported symptoms as 2 of 4 core outcomes domains to include in all future AD clinical trials.3 For measuring symptoms, the Patient-Oriented Eczema Measure (POEM) is the recommended instrument.28 Regarding clinical signs, the HOME group named the Eczema Area and Severity Index (EASI) as the preferred instrument.29
Psychological Burden
Stress is a triggering factor for AD, but the connection between skin and mind appears bidirectional. The biological reaction to stress probably lowers the itch threshold and disrupts the skin barrier.30 The Global Burden of Disease Study showed that skin diseases are the fourth leading cause of nonfatal disease burden.31 There are several factors—pruritus, scratch, and pain—that can all lead to sleep deprivation and daytime fatigue. Based on our experience, if lesions develop on visible areas, patients can feel stigmatized, which restricts their social life.
The most common psychological comorbidities of AD are anxiety and depression. In a cross-sectional, multicenter study, there was a significantly higher prevalence of depression (P<.001) and anxiety disorder (P=.02) among patients with common skin diseases compared to a control group.32 In a study that assessed AD patients, researchers found a higher risk of depression and anxiety.33 Suicidal ideation also is more common in the population with AD32,34; a study showed that the risk of suicidal ideation in adolescents was nearly 4-fold in patients with itching skin lesions compared to those without itch.34
According to Linnet and Jemec,35 mental and psychological comorbidities of AD are associated with lower QOL, not with clinical severity. As a result, to improve QOL in AD, one should take care of both dermatological and psychological problems. It has been demonstrated that psychological interventions, such as autogenic training, cognitive-behavioral therapy, relaxation techniques, habit reversal training,36 and hypnotherapy37 might be helpful in individual cases; educational interventions also are recommended.36 With these adjuvant therapies, psychological status, unpleasant clinical symptoms, and QOL could be improved, though further studies are needed to confirm these benefits.
Conclusion
Atopic dermatitis places a notable burden on patients and their families. The degree of burden is probably related to disease severity. For measuring QOL, researchers and clinicians should use validated methods suited to the age of the patients for which they were designed. More studies are needed to assess the effects of different treatments on QOL. Besides pharmacotherapy, psychotherapy and educational programs might be beneficial for improving QOL, another important area to be studied.
Atopic dermatitis (AD) is a chronic, relapsing, inflammatory skin disease typically with childhood onset. In some cases, the condition persists, but AD usually resolves by the time a child reaches adulthood. Prevalence is difficult to estimate but, in developed countries, is approximately 15% to 30% among children and 2% to 10% among adults.1
Atopic dermatitis is characterized by chronically itchy dry skin, weeping erythematous papules and plaques, and lichenification. Furthermore, AD often is associated with other atopic diseases, such as food allergy, allergic rhinitis, and bronchial asthma.
In this article, we review the literature on the quality of life (QOL) of patients with AD. Our goals are to discuss the most common methods for measuring QOL in AD and how to use them; highlight specific alterations of QOL in AD; and review data about QOL of children with AD, which is underrepresented in the medical literature, as studies tend to focus on adults. In addition, we address the importance of assessing QOL in patients with AD due to the psychological burden of the disease.
Quality of Life
The harmful effects of AD can include a range of areas, including
Because QOL is an important instrument used in many AD studies, we call attention to the work of the Harmonising Outcome Measures for Eczema (HOME) initiative, which established a core outcome set for all AD clinical trials to enable comparison of results of individual studies.2 Quality of life was identified in HOME as one of 4 basic outcome measures that should be included in every AD trial (the others are clinician-reported signs, patient-reported symptoms, and long-term control).3 According to the recent agreement, the following QOL instruments should be used: Dermatology Life Quality Index (DLQI) for adults, Children’s Dermatology Life Quality Index (CDLQI) for children, and Infants’ Dermatitis Quality of Life Index (IDQOL) for infants.4
In dermatology, these instruments can be divided into 3 basic categories: generic, dermatology specific, and disease specific.5 Generic QOL questionnaires are beneficial when comparing the QOL of an AD patient to patients with other conditions or to healthy individuals. On the other hand, dermatology-specific and AD-specific methods are more effective instruments for detecting impairments linked directly to the disease and, therefore, are more sensitive to changes in QOL.5 Some of the most frequently used QOL measures5,6 for AD along with their key attributes are

Given that AD is a chronic disease that requires constant care, parents/guardians or the partner of the patient usually are affected as well. To detect this effect, the Family Dermatology Life Quality Index (FDLQI), a dermatology-specific instrument, measures the QOL in family members of dermatology patients.7 The Dermatitis Family Impact (DFI)8 is a disease-specific method for assessing how having a child with AD can impact the QOL of family members; it is a 10-item questionnaire completed by an adult family member. The FDLQI7 and DFI8 both help to understand the secondary impact of the disease.
In contrast, several other methods that also are administered by a parent/guardian assess how the parent perceives the QOL of their child with AD; these methods are essential for small children and infants who cannot answer questions themselves. The IDQOL9 was designed to assess the QOL of patients younger than 4 years using a parent-completed questionnaire. For older children and adolescents aged 4 to 16 years, the CDLQI10 is a widely used instrument; the questionnaire is completed by the child and is available in a cartoon format.10
For patients older than 16 years, 2 important instruments are the DLQI, a generic dermatology instrument, and the Quality of Life Index for Atopic Dermatitis (QoLIAD).11
Clearly it can be troublesome for researchers and clinicians to find the most suitable instrument to evaluate QOL in AD patients. To make this task easier, the European Academy of Dermatology and Venereology Task Force released a position paper with the following recommendations: (1) only validated instruments should be used, and (2) their use should be based on the age of the patients for which the instruments were designed. It is reommended that researchers use a combination of a generic and a dermatology-specific or AD-specific instrument, whereas clinicians should apply a dermatology-specific or AD-specific method, or both.5
Alterations of QOL in AD
Sleep Disturbance in AD
Sleep disorders observed in AD include difficulty falling asleep, frequent waking episodes, shorter sleep duration, and feelings of inadequate sleep, which often result in impairment of daily activity.12,13 Correlation has been found between sleep quality and QOL in both children and adults.14 Approximately 60% of children affected by AD experience a sleep disturbance,15 which seems to correlate well with disease severity.16 A US study found that adults with AD are more likely to experience a sleep disturbance, which often affects daytime functioning and work productivity.13
Financial Aspects and Impact on Work
The financial burden of AD is extensive.17 There are direct medical costs, including medication, visits to the physician, alternative therapies, and nonprescription products. Patients tend to spend relevant money on such items as moisturizers, bath products, antihistamines, topical steroids, and topical antibiotics.18,19 However, it seems that most of the cost of AD is due to indirect and nonmedical costs, including transportation to medical visits; loss of work days; extra childcare; and expenditures associated with lifestyle changes,19,20 such as modifying diet, wearing special clothes, using special bed linens, and purchasing special household items (eg, anti–dust mite vacuum cleaner, humidifier, new carpeting).17,19
Absenteeism from work often is a consequence of physician appointments; in addition, parents/guardians of a child with AD often miss work due to medical care. Even at work, patients (or parents/guardians) often experience decreased work productivity (so-called presenteeism) due to loss of sleep and anxiety.21 In addressing the effects of AD on work life, a systematic literature review found that AD strongly affects sick leave and might have an impact on job choice and change or loss of job.22
Furthermore, according to Su et al,23 the costs of AD are related to disease severity. Moreover, their data suggest that among chronic childhood diseases, the financial burden of AD is greater than the cost of asthma and similar to the cost of diabetes mellitus.23
Association Between QOL and Disease Severity
A large observational study found that improvement in AD severity was followed by an increase in QOL.24 A positive correlation between disease severity and QOL has been found in other studies,25,26 though no correlation or only moderate correlation also has been reported.27 Apparently, in addition to QOL, disease severity scores are substantial parameters in the evaluation of distress caused by AD; the HOME initiative has identified clinician-reported signs and patient-reported symptoms as 2 of 4 core outcomes domains to include in all future AD clinical trials.3 For measuring symptoms, the Patient-Oriented Eczema Measure (POEM) is the recommended instrument.28 Regarding clinical signs, the HOME group named the Eczema Area and Severity Index (EASI) as the preferred instrument.29
Psychological Burden
Stress is a triggering factor for AD, but the connection between skin and mind appears bidirectional. The biological reaction to stress probably lowers the itch threshold and disrupts the skin barrier.30 The Global Burden of Disease Study showed that skin diseases are the fourth leading cause of nonfatal disease burden.31 There are several factors—pruritus, scratch, and pain—that can all lead to sleep deprivation and daytime fatigue. Based on our experience, if lesions develop on visible areas, patients can feel stigmatized, which restricts their social life.
The most common psychological comorbidities of AD are anxiety and depression. In a cross-sectional, multicenter study, there was a significantly higher prevalence of depression (P<.001) and anxiety disorder (P=.02) among patients with common skin diseases compared to a control group.32 In a study that assessed AD patients, researchers found a higher risk of depression and anxiety.33 Suicidal ideation also is more common in the population with AD32,34; a study showed that the risk of suicidal ideation in adolescents was nearly 4-fold in patients with itching skin lesions compared to those without itch.34
According to Linnet and Jemec,35 mental and psychological comorbidities of AD are associated with lower QOL, not with clinical severity. As a result, to improve QOL in AD, one should take care of both dermatological and psychological problems. It has been demonstrated that psychological interventions, such as autogenic training, cognitive-behavioral therapy, relaxation techniques, habit reversal training,36 and hypnotherapy37 might be helpful in individual cases; educational interventions also are recommended.36 With these adjuvant therapies, psychological status, unpleasant clinical symptoms, and QOL could be improved, though further studies are needed to confirm these benefits.
Conclusion
Atopic dermatitis places a notable burden on patients and their families. The degree of burden is probably related to disease severity. For measuring QOL, researchers and clinicians should use validated methods suited to the age of the patients for which they were designed. More studies are needed to assess the effects of different treatments on QOL. Besides pharmacotherapy, psychotherapy and educational programs might be beneficial for improving QOL, another important area to be studied.
- Bieber T. Atopic dermatitis. N Engl J Med. 2008;358:1483-1494.
- Schmitt J, Williams H; HOME Development Group. Harmonising Outcome Measures for Eczema (HOME). report from the First International Consensus Meeting (HOME 1), 24 July 2010, Munich, Germany. Br J Dermatol. 2010;163:1166-1168.
- Schmitt J, Spuls P, Boers M, et al. Towards global consensus on outcome measures for atopic eczema research: results of the HOME II meeting. Allergy. 2012;67:1111-1117.
- Quality of Life (QoL). Harmonising Outcome Measures for Eczema (HOME) website. http://www.homeforeczema.org/research/quality-of-life.aspx. Accessed August 18, 2019.
Chernyshov PV, Tomas-Aragones L, Manolache L, et al; EADV Quality of Life Task Force. Quality of life measurement in atopic dermatitis. Position paper of the European Academy of Dermatology and Venereology (EADV) Task Force on quality of life. J Eur Acad Dermatol Venereol. 2017;31:576-593. - Hill MK, Kheirandish Pishkenari A, Braunberger TL, et al. Recent trends in disease severity and quality of life instruments for patients with atopic dermatitis: a systematic review. J Am Acad Dermatol. 2016;75:906-917.
- Basra MK, Sue-Ho R, Finlay AY. The Family Dermatology Life Quality Index: measuring the secondary impact of skin disease. Br J Dermatol. 2007;156:528-538.
- Dodington SR, Basra MK, Finlay AY, et al. The Dermatitis Family Impact questionnaire: a review of its measurement properties and clinical application. Br J Dermatol. 2013;169:31-46.
- Lewis-Jones MS, Finlay AY, Dykes PJ. The Infants’ Dermatitis Quality of Life Index. Br J Dermatol. 2001;144:104-110.
- Holme SA, Man I, Sharpe JL, et al. The Children’s Dermatology Life Quality Index: validation of the cartoon version. Br J Dermatol. 2003;148:285-290.
- Whalley D, McKenna SP, Dewar AL, et al. A new instrument for assessing quality of life in atopic dermatitis: international development of the Quality of Life Index for Atopic Dermatitis (QoLIAD). Br J Dermatol. 2004;150:274-283.
- Jeon C, Yan D, Nakamura M, et al. Frequency and management of sleep disturbance in adults with atopic dermatitis: a systematic review. Dermatol Ther (Heidelb). 2017;7:349-364.
- Yu SH, Attarian H, Zee P, et al. Burden of sleep and fatigue in US adults with atopic dermatitis. Dermatitis. 2016;27:50-58.
- Kong TS, Han TY, Lee JH, et al. Correlation between severity of atopic dermatitis and sleep quality in children and adults. Ann Dermatol. 2016;28:321-326.
- Fishbein AB, Mueller K, Kruse L, et al. Sleep disturbance in children with moderate/severe atopic dermatitis: a case-control study. J Am Acad Dermatol. 2018;78:336-341.
- Chamlin SL, Mattson CL, Frieden IJ, et al. The price of pruritus: sleep disturbance and cosleeping in atopic dermatitis. Arch Pediatr Adolesc Med. 2005;159:745-750.
- Emerson RM, Williams HC, Allen BR. What is the cost of atopic dermatitis in preschool children? Br J Dermatol. 2001;144:514-522.
- Filanovsky MG, Pootongkam S, Tamburro JE, et al. The financial and emotional impact of atopic dermatitis on children and their families. J Pediatr. 2016;169:284-290.
- Fivenson D, Arnold RJ, Kaniecki DJ, et al. The effect of atopic dermatitis on total burden of illness and quality of life on adults and children in a large managed care organization. J Manag Care Pharm. 2002;8:333-342.
- Carroll CL, Balkrishnan R, Feldman SR, et al. The burden of atopic dermatitis: impact on the patient, family, and society. Pediatr Dermatol. 2005;22:192-199.
- Drucker AM, Wang AR, Qureshi AA. Research gaps in quality of life and economic burden of atopic dermatitis: the National Eczema Association Burden of Disease Audit. JAMA Dermatol. 2016;152:873-874.
- Nørreslet LB, Ebbehøj NE, Ellekilde Bonde JP, et al. The impact of atopic dermatitis on work life—a systematic review. J Eur Acad Dermatol Venereol. 2018;32:23-38.
- Su JC, Kemp AS, Varigos GA, et al. Atopic eczema: its impact on the family and financial cost. Arch Dis Child. 1997;76:159-162.
- Coutanceau C, Stalder JF. Analysis of correlations between patient-oriented SCORAD (PO-SCORAD) and other assessment scores of atopic dermatitis severity and quality of life. Dermatology. 2014;229:248-255.
- Ben-Gashir MA, Seed PT, Hay RJ. Quality of life and disease severity are correlated in children with atopic dermatitis. Br J Dermatol. 2004;150:284-290.
- van Valburg RW, Willemsen MG, Dirven-Meijer PC, et al. Quality of life measurement and its relationship to disease severity in children with atopic dermatitis in general practice. Acta Derm Venereol. 2011;91:147-151.
- Haeck IM, ten Berge O, van Velsen SG, et al. Moderate correlation between quality of life and disease activity in adult patients with atopic dermatitis. J Eur Acad Dermatol Venereol. 2012;26:236-241.
- Spuls PI, Gerbens LAA, Simpson E, et al; HOME initiative collaborators. Patient-Oriented Eczema Measure (POEM), a core instrument to measure symptoms in clinical trials: a Harmonising Outcome Measures for Eczema (HOME) statement. Br J Dermatol. 2017;176:979-984.
- Schmitt J, Spuls PI, Thomas KS, et al; HOME initiative collaborators. The Harmonising Outcome Measures for Eczema (HOME) statement to assess clinical signs of atopic eczema in trials. J Allergy Clin Immunol. 2014;134:800-807.
- Oh SH, Bae BG, Park CO, et al. Association of stress with symptoms of atopic dermatitis. Acta Derm Venereol. 2010;90:582-588.
- Hay RJ, Johns NE, Williams HC, et al. The global burden of skin disease in 2010: an analysis of the prevalence and impact of skin conditions. J Invest Dermatol. 2014;134:1527-1534.
- Dalgard FJ, Gieler U, Tomas-Aragones L, et al. The psychological burden of skin diseases: a cross-sectional multicenter study among dermatological out-patients in 13 European countries. J Invest Dermatol. 2015;135:984-991.
- Cheng CM, Hsu JW, Huang KL, et al. Risk of developing major depressive disorder and anxiety disorders among adolescents and adults with atopic dermatitis: a nationwide longitudinal study. J Affect Disord. 2015;178:60-65.
Halvorsen JA, Lien L, Dalgard F, et al. Suicidal ideation, mental health problems, and social function in adolescents with eczema: a population-based study. J Invest Dermatol. 2014;134:1847-1854. - Linnet J, Jemec GB. An assessment of anxiety and dermatology life quality in patients with atopic dermatitis. Br J Dermatol. 1999;140:268-272.
- Ring J, Alomar A, Bieber T, et al; European Dermatology Forum; European Academy of Dermatology and Venereology; European Task Force on Atopic Dermatitis; European Federation of Allergy; European Society of Pediatric Dermatology; Global Allergy and Asthma European Network. Guidelines for treatment of atopic eczema (atopic dermatitis) Part II. J Eur Acad Dermatol Venereol. 2012;26:1176-1193.
- Perczel K, Gál J. Hypnotherapy of atopic dermatitis in an adult. case report. Orv Hetil. 2016;157:111-115.
- Bieber T. Atopic dermatitis. N Engl J Med. 2008;358:1483-1494.
- Schmitt J, Williams H; HOME Development Group. Harmonising Outcome Measures for Eczema (HOME). report from the First International Consensus Meeting (HOME 1), 24 July 2010, Munich, Germany. Br J Dermatol. 2010;163:1166-1168.
- Schmitt J, Spuls P, Boers M, et al. Towards global consensus on outcome measures for atopic eczema research: results of the HOME II meeting. Allergy. 2012;67:1111-1117.
- Quality of Life (QoL). Harmonising Outcome Measures for Eczema (HOME) website. http://www.homeforeczema.org/research/quality-of-life.aspx. Accessed August 18, 2019.
Chernyshov PV, Tomas-Aragones L, Manolache L, et al; EADV Quality of Life Task Force. Quality of life measurement in atopic dermatitis. Position paper of the European Academy of Dermatology and Venereology (EADV) Task Force on quality of life. J Eur Acad Dermatol Venereol. 2017;31:576-593. - Hill MK, Kheirandish Pishkenari A, Braunberger TL, et al. Recent trends in disease severity and quality of life instruments for patients with atopic dermatitis: a systematic review. J Am Acad Dermatol. 2016;75:906-917.
- Basra MK, Sue-Ho R, Finlay AY. The Family Dermatology Life Quality Index: measuring the secondary impact of skin disease. Br J Dermatol. 2007;156:528-538.
- Dodington SR, Basra MK, Finlay AY, et al. The Dermatitis Family Impact questionnaire: a review of its measurement properties and clinical application. Br J Dermatol. 2013;169:31-46.
- Lewis-Jones MS, Finlay AY, Dykes PJ. The Infants’ Dermatitis Quality of Life Index. Br J Dermatol. 2001;144:104-110.
- Holme SA, Man I, Sharpe JL, et al. The Children’s Dermatology Life Quality Index: validation of the cartoon version. Br J Dermatol. 2003;148:285-290.
- Whalley D, McKenna SP, Dewar AL, et al. A new instrument for assessing quality of life in atopic dermatitis: international development of the Quality of Life Index for Atopic Dermatitis (QoLIAD). Br J Dermatol. 2004;150:274-283.
- Jeon C, Yan D, Nakamura M, et al. Frequency and management of sleep disturbance in adults with atopic dermatitis: a systematic review. Dermatol Ther (Heidelb). 2017;7:349-364.
- Yu SH, Attarian H, Zee P, et al. Burden of sleep and fatigue in US adults with atopic dermatitis. Dermatitis. 2016;27:50-58.
- Kong TS, Han TY, Lee JH, et al. Correlation between severity of atopic dermatitis and sleep quality in children and adults. Ann Dermatol. 2016;28:321-326.
- Fishbein AB, Mueller K, Kruse L, et al. Sleep disturbance in children with moderate/severe atopic dermatitis: a case-control study. J Am Acad Dermatol. 2018;78:336-341.
- Chamlin SL, Mattson CL, Frieden IJ, et al. The price of pruritus: sleep disturbance and cosleeping in atopic dermatitis. Arch Pediatr Adolesc Med. 2005;159:745-750.
- Emerson RM, Williams HC, Allen BR. What is the cost of atopic dermatitis in preschool children? Br J Dermatol. 2001;144:514-522.
- Filanovsky MG, Pootongkam S, Tamburro JE, et al. The financial and emotional impact of atopic dermatitis on children and their families. J Pediatr. 2016;169:284-290.
- Fivenson D, Arnold RJ, Kaniecki DJ, et al. The effect of atopic dermatitis on total burden of illness and quality of life on adults and children in a large managed care organization. J Manag Care Pharm. 2002;8:333-342.
- Carroll CL, Balkrishnan R, Feldman SR, et al. The burden of atopic dermatitis: impact on the patient, family, and society. Pediatr Dermatol. 2005;22:192-199.
- Drucker AM, Wang AR, Qureshi AA. Research gaps in quality of life and economic burden of atopic dermatitis: the National Eczema Association Burden of Disease Audit. JAMA Dermatol. 2016;152:873-874.
- Nørreslet LB, Ebbehøj NE, Ellekilde Bonde JP, et al. The impact of atopic dermatitis on work life—a systematic review. J Eur Acad Dermatol Venereol. 2018;32:23-38.
- Su JC, Kemp AS, Varigos GA, et al. Atopic eczema: its impact on the family and financial cost. Arch Dis Child. 1997;76:159-162.
- Coutanceau C, Stalder JF. Analysis of correlations between patient-oriented SCORAD (PO-SCORAD) and other assessment scores of atopic dermatitis severity and quality of life. Dermatology. 2014;229:248-255.
- Ben-Gashir MA, Seed PT, Hay RJ. Quality of life and disease severity are correlated in children with atopic dermatitis. Br J Dermatol. 2004;150:284-290.
- van Valburg RW, Willemsen MG, Dirven-Meijer PC, et al. Quality of life measurement and its relationship to disease severity in children with atopic dermatitis in general practice. Acta Derm Venereol. 2011;91:147-151.
- Haeck IM, ten Berge O, van Velsen SG, et al. Moderate correlation between quality of life and disease activity in adult patients with atopic dermatitis. J Eur Acad Dermatol Venereol. 2012;26:236-241.
- Spuls PI, Gerbens LAA, Simpson E, et al; HOME initiative collaborators. Patient-Oriented Eczema Measure (POEM), a core instrument to measure symptoms in clinical trials: a Harmonising Outcome Measures for Eczema (HOME) statement. Br J Dermatol. 2017;176:979-984.
- Schmitt J, Spuls PI, Thomas KS, et al; HOME initiative collaborators. The Harmonising Outcome Measures for Eczema (HOME) statement to assess clinical signs of atopic eczema in trials. J Allergy Clin Immunol. 2014;134:800-807.
- Oh SH, Bae BG, Park CO, et al. Association of stress with symptoms of atopic dermatitis. Acta Derm Venereol. 2010;90:582-588.
- Hay RJ, Johns NE, Williams HC, et al. The global burden of skin disease in 2010: an analysis of the prevalence and impact of skin conditions. J Invest Dermatol. 2014;134:1527-1534.
- Dalgard FJ, Gieler U, Tomas-Aragones L, et al. The psychological burden of skin diseases: a cross-sectional multicenter study among dermatological out-patients in 13 European countries. J Invest Dermatol. 2015;135:984-991.
- Cheng CM, Hsu JW, Huang KL, et al. Risk of developing major depressive disorder and anxiety disorders among adolescents and adults with atopic dermatitis: a nationwide longitudinal study. J Affect Disord. 2015;178:60-65.
Halvorsen JA, Lien L, Dalgard F, et al. Suicidal ideation, mental health problems, and social function in adolescents with eczema: a population-based study. J Invest Dermatol. 2014;134:1847-1854. - Linnet J, Jemec GB. An assessment of anxiety and dermatology life quality in patients with atopic dermatitis. Br J Dermatol. 1999;140:268-272.
- Ring J, Alomar A, Bieber T, et al; European Dermatology Forum; European Academy of Dermatology and Venereology; European Task Force on Atopic Dermatitis; European Federation of Allergy; European Society of Pediatric Dermatology; Global Allergy and Asthma European Network. Guidelines for treatment of atopic eczema (atopic dermatitis) Part II. J Eur Acad Dermatol Venereol. 2012;26:1176-1193.
- Perczel K, Gál J. Hypnotherapy of atopic dermatitis in an adult. case report. Orv Hetil. 2016;157:111-115.
Practice Points
- For assessing quality of life (QOL) in atopic dermatitis (AD), it is recommended that researchers use a combination of a generic and a dermatology-specific or AD-specific instrument, whereas clinicians should apply a dermatology-specific or an AD-specific method or both.
- Anxiety and depression are common comorbidities in AD; patients also may need psychological support.
- Patient education is key for improving QOL in AD.
- Financial aspects of the treatment of AD should be taken into consideration because AD requires constant care, which puts a financial burden on patients.
2019 Update on pelvic floor dysfunction
Fecal incontinence (FI), also known as accidental bowel leakage, is the involuntary loss of feces, which includes both liquid and solid stool as defined by the International Continence Society (ICS) and the International Urogynecological Association (IUGA).1,2 Fecal incontinence is common, occurring in 7% to 25% of community-dwelling women, and it increases with age.2-6 The condition is rarely addressed, with only 30% of women seeking care.6-8 This is due to patient embarrassment and the lack of a reliable screening tool. However, FI affects quality of life and mental health, and the associated economic burden likely will rise given the increased prevalence of FI among older women.2,4,7,9
Fecal incontinence occurs due to poor stool consistency, anal and pelvic muscle weakness, reduced rectal compliance, reduced or increased rectal sensation, or bowel inflammation or dysfunction. Many conditions can cause FI (TABLE 1).5,10,11 It is therefore important to elicit a full medical history with a focus on specific bowel symptoms, such as stool consistency type (TABLE 2),12 FI frequency, and duration of symptoms, as well as to perform a complete examination to identify any readily reversible or malignant causes. A colonoscopy is recommended for individuals who meet screening criteria or present with a change in bowel symptoms, such as diarrhea, bleeding, or obstruction.13,14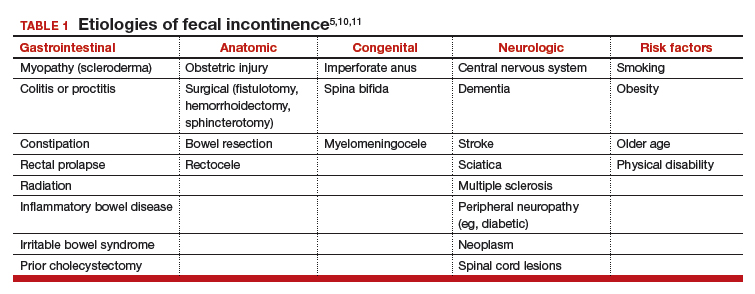

Fecal incontinence treatments include a range of approaches categorized from conservative, or first-line therapy, to fourth-line surgical managements (FIGURE 1).1,10,13,14 In this Update, we review the results of 3 well-designed trials that enrolled women with frequent nonneurogenic FI.

Common first- and second-line treatments produce equivalent improvements in FI symptoms at
6 months
Jelovsek JE, Markland AD, Whitehead WE, et al; National Institute of Child Health and Human Development Pelvic Floor Disorders Network. Controlling faecal incontinence in women by performing anal exercises with biofeedback or loperamide: a randomized clinical trial. Lancet Gastroenterol Hepatol. 2019;4:698-710.
In a multicenter, randomized trial of first- and second-line treatments for FI, Jelovsek and colleagues evaluated the efficacy of oral placebo, loperamide, pelvic floor physical therapy (PFPT) with biofeedback using anorectal manometry, or combination therapy over a 24-week period.
Continue to: Four treatments compared...
Four treatments compared
Three hundred women with FI occurring monthly for 3 months were included in the trial. Women were excluded if they had a stool classification of type 1 or type 7 on the Bristol Stool Scale, inflammatory bowel disease (IBD), history of rectovaginal fistula or cloacal defect, rectal prolapse, prior bowel diversion, fecal impaction, neurologic disorder leading to incontinence, use of loperamide or diphenoxylate within the last 30 days, childbirth within the last 3 months, need for antiretroviral drugs, hepatic impairment, or chronic abdominal pain without diarrhea.
Baseline characteristics and symptoms severity were similar among participants. The average age of the women was 63 years, with 79% white and 85% postmenopausal. Participants had a mean (SD) of 1.6 (1.8) leaks per day.
Participants were randomly assigned in a 0.5:1:1:1 fashion to receive oral placebo, loperamide, oral placebo with PFPT/biofeedback, or loperamide with PFPT/biofeedback. All participants received a standardized educational pamphlet that outlined dietary and behavioral recommendations.
Women assigned to PFPT/biofeedback received 6 sessions every other week. Loperamide was started at a dosage of 2 mg per day with the possibility of dose maintenance, escalation, reduction, or discontinuation.
Study outcomes. The primary outcome was a change from baseline to 24 weeks in the Vaizey FI symptom severity score, which assesses fecal frequency, urgency, and use of pads and medications. Secondary outcomes included assessment of a 7-day bowel diary and other quality-of-life measures. Data at 24 weeks were available for 89% of the women.
All treatment groups experienced improved FI symptoms
Based on changes in Vaizey scores after 24 weeks of treatment, women in all treatment groups had similar improvement in symptoms severity. However, those who received loperamide and PFPT/biofeedback had decreased pad changes per week and more accident-free days compared with women treated with placebo and biofeedback. Quality of life at 24 weeks was not statistically different between treatment groups as improvement was seen in all groups, including those who received oral placebo and patient education.
Adverse events. The proportion of gastrointestinal adverse effects was similar between treatment groups, ranging from 45% to 63%. Constipation was the most common adverse event overall and was more common in those taking loperamide, occurring in 51% of the loperamide plus PFPT/biofeedback group, 38% of those who received loperamide alone, 23% of the biofeedback with placebo group, and 12% of the placebo-alone group.
Strengths and limitations. Strengths of this study include its multisite, large sample size, low dropout rate, and sufficiently powered design to compare various combinations of first- and second-line therapies in women with a mean baseline FI of 1.6 leaks per day. Another strength is the robustness of the PFPT/biofeedback sessions that used anorectal manometry. This may, however, limit the study's external validity given that clinical use of this device is likely rare. Additionally, the population was comprised largely of postmenopausal and white women, which may make the findings less generalizable to other populations.
Women who suffer from frequent FI may require both loperamide and PFPT/biofeedback if they want to increase the likelihood of accident-free days and use of fewer pads. Should they note increased constipation or are not amenable to scheduled PFPT sessions, formalized education about dietary modifications, according to this study, will provide improvement in symptom severity.
Continue to: Novel vaginal bowel control system...
Novel vaginal bowel control system is effective, durable over 12 months for FI treatment
Richter HE, Dunivan G, Brown HW, et al. A 12-month clinical durability of effectiveness and safety evaluation of a vaginal bowel control system for the nonsurgical treatment of fecal incontinence. Female Pelvic Med Reconstr Surg. 2019;25:113-119.
Richter and colleagues characterized clinical success, effect on quality of life, and durability over 12 months of a novel vaginal bowel control device (Eclipse System; Pelvalon) for FI in a prospective cohort study. The device is a silicone-coated vaginal insert with a detachable pump and balloon that deflects the rectovaginal septum posteriorly, thus impeding the passage of stool in the rectum (FIGURE 2).
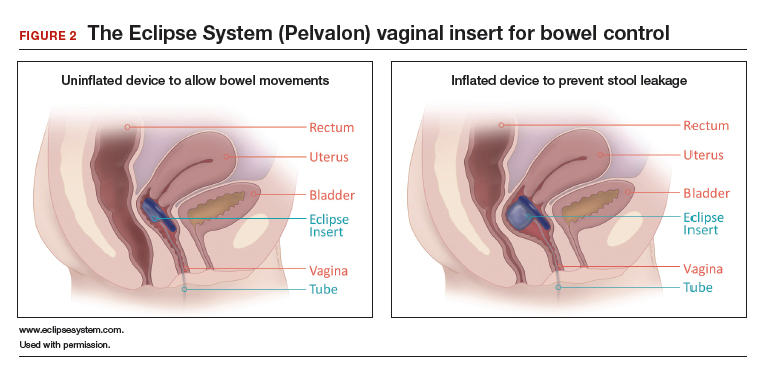
Study eligibility criteria and treatment protocol
Women were eligible for the study if they had 4 or more episodes of fecal soiling on a 2-week bowel diary and had FI for at least 6 months. Participants were excluded if they had prolapse outside the hymen, rectovaginal fistula, IBD, congenital anorectal malformation, urinary or colorectal infection, chronic pelvic or anorectal pain, pregnancy or planning pregnancy in the next 5 months, unmanaged chronic watery diarrhea, presence of an open wound or tear in the vagina, significant urogenital atrophy, or any psychiatric or neurologic disorder that would hinder the ability to participate.
Participants successfully fitted with the device (3 attempts were allowed) were entered into the study's run-in phase. Those who were successfully fitted and had a 50% or greater reduction in FI continued into the treatment phase with 12 months of follow-up.
Of the 137 women eligible for device fitting, 62% were successfully fitted. The 73 (86%) women who had a 50% or greater reduction in FI during the run-in period comprised the intent-to-treat study population. On average, these women were 61.3 years of age, with 70% white and 82% postmenopausal. At baseline, they had a mean of 14.1 episodes of FI over 2 weeks. (Prior to enrollment, 97.3% of women attempted self-management strategies, 17.8% to 23% failed conservative therapy, and 7.8% to 13.7% failed surgical therapy.) The follow-up rate at 12 months was 74%.
Study outcomes. The primary outcome was treatment success, defined as proportion of subjects with a 50% or greater reduction in FI episodes at 3 months; this outcome also was evaluated at 6 and 12 months. Secondary outcomes were the number of FI episodes and quality-of-life measures at 3, 6, and 12 months.
Treatment success, patient satisfaction high
In the treatment phase, women had sustained improvements in symptom severity and quality-of-life measures over 12 months. Treatment success was 73% at 3 months, 71% at 6 months, and 70% at 12 months. Complete continence was achieved in 46% of participants at 12 months, and major FI episodes (requiring immediate change of undergarments) decreased from 5.0 at baseline to 0.5 at 12 months. Quality-of-life measures were improved at 3 months, and improvement was sustained over 12 months. Satisfaction was 94% at 12 months.
Adverse events. No serious device-related adverse events occurred. Mild device-related adverse events were experienced by 45% of women during the fitting process and by 38% during treatment period. These included vaginal wall injury such as hyperemia and erosion; vaginal or pelvic discomfort; vaginal infection; constipation; and lower urinary tract issues such as urinary tract infection, urinary incontinence, and voiding dysfunction. No adverse events led to treatment discontinuation.
Strengths and limitations. Strengths of this study include that it was conducted at multiple clinical sites, had a large sample size, and had a 1-year follow-up period in a population with daily FI. A limitation was that only women who had a 50% or greater reduction in FI episodes during the run-in period were followed for 12 months; however, this was 86% of the original cohort. The use of a comparative group using other devices, such as anal plugs, would have strengthened this study.
The Eclipse intravaginal bowel control device (approved by the US Food and Drug Administration in 2015) provided a sustained 50% or greater reduction in FI episodes in more than 70% of women wearing the device for 1 year, with high patient satisfaction. Thus, for women who fail conservative treatment methods for FI, clinicians should consider referring them to a urogynecologist or specialist who is knowledgeable in fitting this vaginal bowel control device.
Continue to: Sacroneuromodulation for FI…
Sacral neuromodulation for FI is effective long-term
Hull T, Giese C, Wexner SD, et al; for the SNS Study Group. Long-term durability of sacral nerve stimulation therapy for chronic fecal incontinence. Dis Colon Rectum. 2013;56:234-245.
In this multicenter, prospective cohort study, Hull and colleagues evaluated the 5-year efficacy of sacral neuromodulation (SNM), also known as sacral nerve stimulation, for treatment of FI. This study followed an earlier investigation by Wexner and colleagues, which reported that 83% of 120 patients treated with SNM had a 50% or greater improvement in FI episodes at 12 months.16
Details of the study
The investigators enrolled 133 participants (92% female) who had more than 2 episodes of FI per week for longer than 6 months (12 months after vaginal delivery). Participants were excluded if they had congenital anorectal malformations, prior rectal surgery within the past 12 months (or 24 months if due to cancer), defects greater than 120° of the external anal sphincter (EAS), IBD, unmanaged chronic watery diarrhea, stool consistency type 6 or type 7 on the Bristol Stool Scale, sequela of pelvic radiation, active anal abscess or fistula, pregnancy, or planned pregnancy.
Eligible participants underwent a 2-stage procedure with the InterStim bowel control device (Medtronic). If participants experienced a 50% or greater reduction in incontinence episodes with a wearable external SNM device in the test stimulation (stage 1), they received the chronic SNM implant device (stage 2).
Participants who underwent device implantation were followed at 1, 3, and 6 months and annually for 5 years or until they exited the study. Bowel diaries and quality of life assessments were completed at baseline and at follow-up.
The primary outcome was therapeutic success, defined as 50% or greater improvement in FI episodes per week.
A total of 120 participants (90%) underwent implantation of the chronic lead and neuromodulator, and 76 (63%) were followed for 5 years. Baseline characteristics available in the initial study of 133 participants showed that the mean age was 60.5 years; 25% had undergone a prior anal sphincteroplasty; and 16.5% and 10.5% had EAS or internal anal sphincter (IAS) defects, respectively, on endoanal ultrasonography.16
Therapeutic success was high at 5 years
At the 5-year follow-up, 89% (64/72) of participants met therapeutic success, with a reduction in weekly FI episodes from 9.1 at baseline to 1.7 at 5 years. The number of incontinence pads required decreased, and more participants wore no pads at 5 years. In the intention-to-treat analysis, carrying forward the baseline FI rate in participants who lacked follow-up data, the therapeutic success rate was 69%. Quality-of-life measures improved at 5 years, both statistically and by minimal clinical difference.
Adverse events. Sixty-eight percent of participants experienced device-related adverse events, including implant site pain, change in sensation of stimulation, change in efficacy, implant site infection, or neurostimulator battery depletion (neurostimulator use commonly expires after 3 to 5 years). Of these events, 80% were successfully treated with medications, reprogramming, or no intervention. The 5-year probability of device revision or replacement was 24.4%, and the 5-year probability of device explant was 19.0%.
Strengths and limitations. Overall, this study was a well-designed, multicenter trial with long-term follow-up that showed significant improvement in FI with the use of SNM. Its strengths include the enrollment of postmenopausal women who had current defects in EAS and/or IAS on endoanal ultrasonography and 25% who had a prior sphincteroplasty. The findings therefore are relevant to the gynecologic population in whom anal sphincteroplasty would not be recommended. The study also accounted for dropouts and reported the adjusted success rate of 69% at 5 years in that group.
The lack of a control arm to rule out the placebo effect is a limitation of this study, although randomized trials comparing the effect of SNM "on" versus "off" showed greater improvement with the device "on."17
Sacral neuromodulation is an excellent therapy for women with daily FI who have failed noninvasive options and desire to proceed to a more durable, long-lasting device therapy. Although adverse events may occur, they are mild and most often resolve with device reprogramming.
- Sultan AH, Monga A, Lee J, et al. An International Urogynecological Association (IUGA)/International Continence Society (ICS) joint report on the terminology for female anorectal dysfunction. Neurourol Urodyn. 2017;36:10-34.
- Bharucha AE, Dunivan G, Goode PS, et al. Epidemiology, pathophysiology, and classification of fecal incontinence: state of the science summary for the National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK) workshop. Am J Gastroenterol. 2015;110:127-136.
- Bharucha AE, Zinsmeister AR, Locke GR, et al. Symptoms and quality of life in community women with fecal incontinence. Clin Gastroenterol Hepatol. 2006;4:1004-1008.
- Perry S, Shaw C, McGrother C, et al; Leicestershire MRC Incontinence Study Team. Prevalence of faecal incontinence in adults aged 40 years or more living in the community. Gut. 2002;50:480-484.
- Ditah I, Devaki P, Luma HN, et al. Prevalence, trends, and risk factors for fecal incontinence in United States adults, 2005-2010. Clin Gastroenterol Hepatol. 2014;12:636-643.e1-2.
- Brown HW, Wexner SD, Lukacz ES. Factors associated with care seeking among women with accidental bowel leakage. Female Pelvic Med Reconstr Surg. 2013;19:66-71.
- Norton NJ. The perspective of the patient. Gastroenterology. 2004;126(1 suppl 1):S175-S179.
- Guan W, Schmuhl NB, Brown HW. Response re: If we don't ask, they won't tell: screening for urinary and fecal incontinence by primary care providers. J Am Board Fam Med. 2019;32:119.3-120.
- Whitehead WE, Borrud L, Goode PS, et al; Pelvic Floor Disorders Network. Fecal incontinence in US adults: epidemiology and risk factors. Gastroenterology. 2009;137:512-517.
- Wald A, Bharucha AE, Cosman BC, et al. ACG clinical guideline: management of benign anorectal disorders. Am J Gastroenterol. 2014;109:1141-1157.
- Bharucha AE, Zinsmeister AR, Schleck CD, et al. Bowel disturbances are the most important risk factors for late onset fecal incontinence: a population-based case-control study in women. Gastroenterology. 2010;139:1559-1566.
- Lewis SJ, Heaton KW. Stool form scale as a useful guide to intestinal transit time. Scand J Gastroenterol. 1997;32:920-924.
- Paquette IM, Varma MG, Kaiser AM, et al. The American Society of Colon and Rectal Surgeons' clinical practice guideline for the treatment of fecal incontinence. Dis Colon Rectum. 2015;58:623-636.
- American College of Obstetricians and Gynecologists. ACOG practice bulletin no. 210: Fecal incontinence. Obstet Gynecol. 2019;133:e260-e273.
- Heymen S, Scarlett Y, Jones K, et al. Randomized controlled trial shows biofeedback to be superior to pelvic floor exercises for fecal incontinence. Dis Colon Rectum. 2009;52:1730-1737.
- Wexner SD, Coller JA, Devroede G, et al. Sacral nerve stimulation for fecal incontinence: results of a 120-patient prospective multicenter study. Ann Surg. 2010;251:441-449.
- Leroi AM, Parc Y, Lehur PA, et al. Efficacy of sacral nerve stimulation for fecal incontinence: results of a multicenter double-blind crossover study. Ann Surg. 2005;242:662-669.
Fecal incontinence (FI), also known as accidental bowel leakage, is the involuntary loss of feces, which includes both liquid and solid stool as defined by the International Continence Society (ICS) and the International Urogynecological Association (IUGA).1,2 Fecal incontinence is common, occurring in 7% to 25% of community-dwelling women, and it increases with age.2-6 The condition is rarely addressed, with only 30% of women seeking care.6-8 This is due to patient embarrassment and the lack of a reliable screening tool. However, FI affects quality of life and mental health, and the associated economic burden likely will rise given the increased prevalence of FI among older women.2,4,7,9
Fecal incontinence occurs due to poor stool consistency, anal and pelvic muscle weakness, reduced rectal compliance, reduced or increased rectal sensation, or bowel inflammation or dysfunction. Many conditions can cause FI (TABLE 1).5,10,11 It is therefore important to elicit a full medical history with a focus on specific bowel symptoms, such as stool consistency type (TABLE 2),12 FI frequency, and duration of symptoms, as well as to perform a complete examination to identify any readily reversible or malignant causes. A colonoscopy is recommended for individuals who meet screening criteria or present with a change in bowel symptoms, such as diarrhea, bleeding, or obstruction.13,14

Fecal incontinence treatments include a range of approaches categorized from conservative, or first-line therapy, to fourth-line surgical managements (FIGURE 1).1,10,13,14 In this Update, we review the results of 3 well-designed trials that enrolled women with frequent nonneurogenic FI.

Common first- and second-line treatments produce equivalent improvements in FI symptoms at
6 months
Jelovsek JE, Markland AD, Whitehead WE, et al; National Institute of Child Health and Human Development Pelvic Floor Disorders Network. Controlling faecal incontinence in women by performing anal exercises with biofeedback or loperamide: a randomized clinical trial. Lancet Gastroenterol Hepatol. 2019;4:698-710.
In a multicenter, randomized trial of first- and second-line treatments for FI, Jelovsek and colleagues evaluated the efficacy of oral placebo, loperamide, pelvic floor physical therapy (PFPT) with biofeedback using anorectal manometry, or combination therapy over a 24-week period.
Continue to: Four treatments compared...
Four treatments compared
Three hundred women with FI occurring monthly for 3 months were included in the trial. Women were excluded if they had a stool classification of type 1 or type 7 on the Bristol Stool Scale, inflammatory bowel disease (IBD), history of rectovaginal fistula or cloacal defect, rectal prolapse, prior bowel diversion, fecal impaction, neurologic disorder leading to incontinence, use of loperamide or diphenoxylate within the last 30 days, childbirth within the last 3 months, need for antiretroviral drugs, hepatic impairment, or chronic abdominal pain without diarrhea.
Baseline characteristics and symptoms severity were similar among participants. The average age of the women was 63 years, with 79% white and 85% postmenopausal. Participants had a mean (SD) of 1.6 (1.8) leaks per day.
Participants were randomly assigned in a 0.5:1:1:1 fashion to receive oral placebo, loperamide, oral placebo with PFPT/biofeedback, or loperamide with PFPT/biofeedback. All participants received a standardized educational pamphlet that outlined dietary and behavioral recommendations.
Women assigned to PFPT/biofeedback received 6 sessions every other week. Loperamide was started at a dosage of 2 mg per day with the possibility of dose maintenance, escalation, reduction, or discontinuation.
Study outcomes. The primary outcome was a change from baseline to 24 weeks in the Vaizey FI symptom severity score, which assesses fecal frequency, urgency, and use of pads and medications. Secondary outcomes included assessment of a 7-day bowel diary and other quality-of-life measures. Data at 24 weeks were available for 89% of the women.
All treatment groups experienced improved FI symptoms
Based on changes in Vaizey scores after 24 weeks of treatment, women in all treatment groups had similar improvement in symptoms severity. However, those who received loperamide and PFPT/biofeedback had decreased pad changes per week and more accident-free days compared with women treated with placebo and biofeedback. Quality of life at 24 weeks was not statistically different between treatment groups as improvement was seen in all groups, including those who received oral placebo and patient education.
Adverse events. The proportion of gastrointestinal adverse effects was similar between treatment groups, ranging from 45% to 63%. Constipation was the most common adverse event overall and was more common in those taking loperamide, occurring in 51% of the loperamide plus PFPT/biofeedback group, 38% of those who received loperamide alone, 23% of the biofeedback with placebo group, and 12% of the placebo-alone group.
Strengths and limitations. Strengths of this study include its multisite, large sample size, low dropout rate, and sufficiently powered design to compare various combinations of first- and second-line therapies in women with a mean baseline FI of 1.6 leaks per day. Another strength is the robustness of the PFPT/biofeedback sessions that used anorectal manometry. This may, however, limit the study's external validity given that clinical use of this device is likely rare. Additionally, the population was comprised largely of postmenopausal and white women, which may make the findings less generalizable to other populations.
Women who suffer from frequent FI may require both loperamide and PFPT/biofeedback if they want to increase the likelihood of accident-free days and use of fewer pads. Should they note increased constipation or are not amenable to scheduled PFPT sessions, formalized education about dietary modifications, according to this study, will provide improvement in symptom severity.
Continue to: Novel vaginal bowel control system...
Novel vaginal bowel control system is effective, durable over 12 months for FI treatment
Richter HE, Dunivan G, Brown HW, et al. A 12-month clinical durability of effectiveness and safety evaluation of a vaginal bowel control system for the nonsurgical treatment of fecal incontinence. Female Pelvic Med Reconstr Surg. 2019;25:113-119.
Richter and colleagues characterized clinical success, effect on quality of life, and durability over 12 months of a novel vaginal bowel control device (Eclipse System; Pelvalon) for FI in a prospective cohort study. The device is a silicone-coated vaginal insert with a detachable pump and balloon that deflects the rectovaginal septum posteriorly, thus impeding the passage of stool in the rectum (FIGURE 2).

Study eligibility criteria and treatment protocol
Women were eligible for the study if they had 4 or more episodes of fecal soiling on a 2-week bowel diary and had FI for at least 6 months. Participants were excluded if they had prolapse outside the hymen, rectovaginal fistula, IBD, congenital anorectal malformation, urinary or colorectal infection, chronic pelvic or anorectal pain, pregnancy or planning pregnancy in the next 5 months, unmanaged chronic watery diarrhea, presence of an open wound or tear in the vagina, significant urogenital atrophy, or any psychiatric or neurologic disorder that would hinder the ability to participate.
Participants successfully fitted with the device (3 attempts were allowed) were entered into the study's run-in phase. Those who were successfully fitted and had a 50% or greater reduction in FI continued into the treatment phase with 12 months of follow-up.
Of the 137 women eligible for device fitting, 62% were successfully fitted. The 73 (86%) women who had a 50% or greater reduction in FI during the run-in period comprised the intent-to-treat study population. On average, these women were 61.3 years of age, with 70% white and 82% postmenopausal. At baseline, they had a mean of 14.1 episodes of FI over 2 weeks. (Prior to enrollment, 97.3% of women attempted self-management strategies, 17.8% to 23% failed conservative therapy, and 7.8% to 13.7% failed surgical therapy.) The follow-up rate at 12 months was 74%.
Study outcomes. The primary outcome was treatment success, defined as proportion of subjects with a 50% or greater reduction in FI episodes at 3 months; this outcome also was evaluated at 6 and 12 months. Secondary outcomes were the number of FI episodes and quality-of-life measures at 3, 6, and 12 months.
Treatment success, patient satisfaction high
In the treatment phase, women had sustained improvements in symptom severity and quality-of-life measures over 12 months. Treatment success was 73% at 3 months, 71% at 6 months, and 70% at 12 months. Complete continence was achieved in 46% of participants at 12 months, and major FI episodes (requiring immediate change of undergarments) decreased from 5.0 at baseline to 0.5 at 12 months. Quality-of-life measures were improved at 3 months, and improvement was sustained over 12 months. Satisfaction was 94% at 12 months.
Adverse events. No serious device-related adverse events occurred. Mild device-related adverse events were experienced by 45% of women during the fitting process and by 38% during treatment period. These included vaginal wall injury such as hyperemia and erosion; vaginal or pelvic discomfort; vaginal infection; constipation; and lower urinary tract issues such as urinary tract infection, urinary incontinence, and voiding dysfunction. No adverse events led to treatment discontinuation.
Strengths and limitations. Strengths of this study include that it was conducted at multiple clinical sites, had a large sample size, and had a 1-year follow-up period in a population with daily FI. A limitation was that only women who had a 50% or greater reduction in FI episodes during the run-in period were followed for 12 months; however, this was 86% of the original cohort. The use of a comparative group using other devices, such as anal plugs, would have strengthened this study.
The Eclipse intravaginal bowel control device (approved by the US Food and Drug Administration in 2015) provided a sustained 50% or greater reduction in FI episodes in more than 70% of women wearing the device for 1 year, with high patient satisfaction. Thus, for women who fail conservative treatment methods for FI, clinicians should consider referring them to a urogynecologist or specialist who is knowledgeable in fitting this vaginal bowel control device.
Continue to: Sacroneuromodulation for FI…
Sacral neuromodulation for FI is effective long-term
Hull T, Giese C, Wexner SD, et al; for the SNS Study Group. Long-term durability of sacral nerve stimulation therapy for chronic fecal incontinence. Dis Colon Rectum. 2013;56:234-245.
In this multicenter, prospective cohort study, Hull and colleagues evaluated the 5-year efficacy of sacral neuromodulation (SNM), also known as sacral nerve stimulation, for treatment of FI. This study followed an earlier investigation by Wexner and colleagues, which reported that 83% of 120 patients treated with SNM had a 50% or greater improvement in FI episodes at 12 months.16
Details of the study
The investigators enrolled 133 participants (92% female) who had more than 2 episodes of FI per week for longer than 6 months (12 months after vaginal delivery). Participants were excluded if they had congenital anorectal malformations, prior rectal surgery within the past 12 months (or 24 months if due to cancer), defects greater than 120° of the external anal sphincter (EAS), IBD, unmanaged chronic watery diarrhea, stool consistency type 6 or type 7 on the Bristol Stool Scale, sequela of pelvic radiation, active anal abscess or fistula, pregnancy, or planned pregnancy.
Eligible participants underwent a 2-stage procedure with the InterStim bowel control device (Medtronic). If participants experienced a 50% or greater reduction in incontinence episodes with a wearable external SNM device in the test stimulation (stage 1), they received the chronic SNM implant device (stage 2).
Participants who underwent device implantation were followed at 1, 3, and 6 months and annually for 5 years or until they exited the study. Bowel diaries and quality of life assessments were completed at baseline and at follow-up.
The primary outcome was therapeutic success, defined as 50% or greater improvement in FI episodes per week.
A total of 120 participants (90%) underwent implantation of the chronic lead and neuromodulator, and 76 (63%) were followed for 5 years. Baseline characteristics available in the initial study of 133 participants showed that the mean age was 60.5 years; 25% had undergone a prior anal sphincteroplasty; and 16.5% and 10.5% had EAS or internal anal sphincter (IAS) defects, respectively, on endoanal ultrasonography.16
Therapeutic success was high at 5 years
At the 5-year follow-up, 89% (64/72) of participants met therapeutic success, with a reduction in weekly FI episodes from 9.1 at baseline to 1.7 at 5 years. The number of incontinence pads required decreased, and more participants wore no pads at 5 years. In the intention-to-treat analysis, carrying forward the baseline FI rate in participants who lacked follow-up data, the therapeutic success rate was 69%. Quality-of-life measures improved at 5 years, both statistically and by minimal clinical difference.
Adverse events. Sixty-eight percent of participants experienced device-related adverse events, including implant site pain, change in sensation of stimulation, change in efficacy, implant site infection, or neurostimulator battery depletion (neurostimulator use commonly expires after 3 to 5 years). Of these events, 80% were successfully treated with medications, reprogramming, or no intervention. The 5-year probability of device revision or replacement was 24.4%, and the 5-year probability of device explant was 19.0%.
Strengths and limitations. Overall, this study was a well-designed, multicenter trial with long-term follow-up that showed significant improvement in FI with the use of SNM. Its strengths include the enrollment of postmenopausal women who had current defects in EAS and/or IAS on endoanal ultrasonography and 25% who had a prior sphincteroplasty. The findings therefore are relevant to the gynecologic population in whom anal sphincteroplasty would not be recommended. The study also accounted for dropouts and reported the adjusted success rate of 69% at 5 years in that group.
The lack of a control arm to rule out the placebo effect is a limitation of this study, although randomized trials comparing the effect of SNM "on" versus "off" showed greater improvement with the device "on."17
Sacral neuromodulation is an excellent therapy for women with daily FI who have failed noninvasive options and desire to proceed to a more durable, long-lasting device therapy. Although adverse events may occur, they are mild and most often resolve with device reprogramming.
Fecal incontinence (FI), also known as accidental bowel leakage, is the involuntary loss of feces, which includes both liquid and solid stool as defined by the International Continence Society (ICS) and the International Urogynecological Association (IUGA).1,2 Fecal incontinence is common, occurring in 7% to 25% of community-dwelling women, and it increases with age.2-6 The condition is rarely addressed, with only 30% of women seeking care.6-8 This is due to patient embarrassment and the lack of a reliable screening tool. However, FI affects quality of life and mental health, and the associated economic burden likely will rise given the increased prevalence of FI among older women.2,4,7,9
Fecal incontinence occurs due to poor stool consistency, anal and pelvic muscle weakness, reduced rectal compliance, reduced or increased rectal sensation, or bowel inflammation or dysfunction. Many conditions can cause FI (TABLE 1).5,10,11 It is therefore important to elicit a full medical history with a focus on specific bowel symptoms, such as stool consistency type (TABLE 2),12 FI frequency, and duration of symptoms, as well as to perform a complete examination to identify any readily reversible or malignant causes. A colonoscopy is recommended for individuals who meet screening criteria or present with a change in bowel symptoms, such as diarrhea, bleeding, or obstruction.13,14

Fecal incontinence treatments include a range of approaches categorized from conservative, or first-line therapy, to fourth-line surgical managements (FIGURE 1).1,10,13,14 In this Update, we review the results of 3 well-designed trials that enrolled women with frequent nonneurogenic FI.

Common first- and second-line treatments produce equivalent improvements in FI symptoms at
6 months
Jelovsek JE, Markland AD, Whitehead WE, et al; National Institute of Child Health and Human Development Pelvic Floor Disorders Network. Controlling faecal incontinence in women by performing anal exercises with biofeedback or loperamide: a randomized clinical trial. Lancet Gastroenterol Hepatol. 2019;4:698-710.
In a multicenter, randomized trial of first- and second-line treatments for FI, Jelovsek and colleagues evaluated the efficacy of oral placebo, loperamide, pelvic floor physical therapy (PFPT) with biofeedback using anorectal manometry, or combination therapy over a 24-week period.
Continue to: Four treatments compared...
Four treatments compared
Three hundred women with FI occurring monthly for 3 months were included in the trial. Women were excluded if they had a stool classification of type 1 or type 7 on the Bristol Stool Scale, inflammatory bowel disease (IBD), history of rectovaginal fistula or cloacal defect, rectal prolapse, prior bowel diversion, fecal impaction, neurologic disorder leading to incontinence, use of loperamide or diphenoxylate within the last 30 days, childbirth within the last 3 months, need for antiretroviral drugs, hepatic impairment, or chronic abdominal pain without diarrhea.
Baseline characteristics and symptoms severity were similar among participants. The average age of the women was 63 years, with 79% white and 85% postmenopausal. Participants had a mean (SD) of 1.6 (1.8) leaks per day.
Participants were randomly assigned in a 0.5:1:1:1 fashion to receive oral placebo, loperamide, oral placebo with PFPT/biofeedback, or loperamide with PFPT/biofeedback. All participants received a standardized educational pamphlet that outlined dietary and behavioral recommendations.
Women assigned to PFPT/biofeedback received 6 sessions every other week. Loperamide was started at a dosage of 2 mg per day with the possibility of dose maintenance, escalation, reduction, or discontinuation.
Study outcomes. The primary outcome was a change from baseline to 24 weeks in the Vaizey FI symptom severity score, which assesses fecal frequency, urgency, and use of pads and medications. Secondary outcomes included assessment of a 7-day bowel diary and other quality-of-life measures. Data at 24 weeks were available for 89% of the women.
All treatment groups experienced improved FI symptoms
Based on changes in Vaizey scores after 24 weeks of treatment, women in all treatment groups had similar improvement in symptoms severity. However, those who received loperamide and PFPT/biofeedback had decreased pad changes per week and more accident-free days compared with women treated with placebo and biofeedback. Quality of life at 24 weeks was not statistically different between treatment groups as improvement was seen in all groups, including those who received oral placebo and patient education.
Adverse events. The proportion of gastrointestinal adverse effects was similar between treatment groups, ranging from 45% to 63%. Constipation was the most common adverse event overall and was more common in those taking loperamide, occurring in 51% of the loperamide plus PFPT/biofeedback group, 38% of those who received loperamide alone, 23% of the biofeedback with placebo group, and 12% of the placebo-alone group.
Strengths and limitations. Strengths of this study include its multisite, large sample size, low dropout rate, and sufficiently powered design to compare various combinations of first- and second-line therapies in women with a mean baseline FI of 1.6 leaks per day. Another strength is the robustness of the PFPT/biofeedback sessions that used anorectal manometry. This may, however, limit the study's external validity given that clinical use of this device is likely rare. Additionally, the population was comprised largely of postmenopausal and white women, which may make the findings less generalizable to other populations.
Women who suffer from frequent FI may require both loperamide and PFPT/biofeedback if they want to increase the likelihood of accident-free days and use of fewer pads. Should they note increased constipation or are not amenable to scheduled PFPT sessions, formalized education about dietary modifications, according to this study, will provide improvement in symptom severity.
Continue to: Novel vaginal bowel control system...
Novel vaginal bowel control system is effective, durable over 12 months for FI treatment
Richter HE, Dunivan G, Brown HW, et al. A 12-month clinical durability of effectiveness and safety evaluation of a vaginal bowel control system for the nonsurgical treatment of fecal incontinence. Female Pelvic Med Reconstr Surg. 2019;25:113-119.
Richter and colleagues characterized clinical success, effect on quality of life, and durability over 12 months of a novel vaginal bowel control device (Eclipse System; Pelvalon) for FI in a prospective cohort study. The device is a silicone-coated vaginal insert with a detachable pump and balloon that deflects the rectovaginal septum posteriorly, thus impeding the passage of stool in the rectum (FIGURE 2).

Study eligibility criteria and treatment protocol
Women were eligible for the study if they had 4 or more episodes of fecal soiling on a 2-week bowel diary and had FI for at least 6 months. Participants were excluded if they had prolapse outside the hymen, rectovaginal fistula, IBD, congenital anorectal malformation, urinary or colorectal infection, chronic pelvic or anorectal pain, pregnancy or planning pregnancy in the next 5 months, unmanaged chronic watery diarrhea, presence of an open wound or tear in the vagina, significant urogenital atrophy, or any psychiatric or neurologic disorder that would hinder the ability to participate.
Participants successfully fitted with the device (3 attempts were allowed) were entered into the study's run-in phase. Those who were successfully fitted and had a 50% or greater reduction in FI continued into the treatment phase with 12 months of follow-up.
Of the 137 women eligible for device fitting, 62% were successfully fitted. The 73 (86%) women who had a 50% or greater reduction in FI during the run-in period comprised the intent-to-treat study population. On average, these women were 61.3 years of age, with 70% white and 82% postmenopausal. At baseline, they had a mean of 14.1 episodes of FI over 2 weeks. (Prior to enrollment, 97.3% of women attempted self-management strategies, 17.8% to 23% failed conservative therapy, and 7.8% to 13.7% failed surgical therapy.) The follow-up rate at 12 months was 74%.
Study outcomes. The primary outcome was treatment success, defined as proportion of subjects with a 50% or greater reduction in FI episodes at 3 months; this outcome also was evaluated at 6 and 12 months. Secondary outcomes were the number of FI episodes and quality-of-life measures at 3, 6, and 12 months.
Treatment success, patient satisfaction high
In the treatment phase, women had sustained improvements in symptom severity and quality-of-life measures over 12 months. Treatment success was 73% at 3 months, 71% at 6 months, and 70% at 12 months. Complete continence was achieved in 46% of participants at 12 months, and major FI episodes (requiring immediate change of undergarments) decreased from 5.0 at baseline to 0.5 at 12 months. Quality-of-life measures were improved at 3 months, and improvement was sustained over 12 months. Satisfaction was 94% at 12 months.
Adverse events. No serious device-related adverse events occurred. Mild device-related adverse events were experienced by 45% of women during the fitting process and by 38% during treatment period. These included vaginal wall injury such as hyperemia and erosion; vaginal or pelvic discomfort; vaginal infection; constipation; and lower urinary tract issues such as urinary tract infection, urinary incontinence, and voiding dysfunction. No adverse events led to treatment discontinuation.
Strengths and limitations. Strengths of this study include that it was conducted at multiple clinical sites, had a large sample size, and had a 1-year follow-up period in a population with daily FI. A limitation was that only women who had a 50% or greater reduction in FI episodes during the run-in period were followed for 12 months; however, this was 86% of the original cohort. The use of a comparative group using other devices, such as anal plugs, would have strengthened this study.
The Eclipse intravaginal bowel control device (approved by the US Food and Drug Administration in 2015) provided a sustained 50% or greater reduction in FI episodes in more than 70% of women wearing the device for 1 year, with high patient satisfaction. Thus, for women who fail conservative treatment methods for FI, clinicians should consider referring them to a urogynecologist or specialist who is knowledgeable in fitting this vaginal bowel control device.
Continue to: Sacroneuromodulation for FI…
Sacral neuromodulation for FI is effective long-term
Hull T, Giese C, Wexner SD, et al; for the SNS Study Group. Long-term durability of sacral nerve stimulation therapy for chronic fecal incontinence. Dis Colon Rectum. 2013;56:234-245.
In this multicenter, prospective cohort study, Hull and colleagues evaluated the 5-year efficacy of sacral neuromodulation (SNM), also known as sacral nerve stimulation, for treatment of FI. This study followed an earlier investigation by Wexner and colleagues, which reported that 83% of 120 patients treated with SNM had a 50% or greater improvement in FI episodes at 12 months.16
Details of the study
The investigators enrolled 133 participants (92% female) who had more than 2 episodes of FI per week for longer than 6 months (12 months after vaginal delivery). Participants were excluded if they had congenital anorectal malformations, prior rectal surgery within the past 12 months (or 24 months if due to cancer), defects greater than 120° of the external anal sphincter (EAS), IBD, unmanaged chronic watery diarrhea, stool consistency type 6 or type 7 on the Bristol Stool Scale, sequela of pelvic radiation, active anal abscess or fistula, pregnancy, or planned pregnancy.
Eligible participants underwent a 2-stage procedure with the InterStim bowel control device (Medtronic). If participants experienced a 50% or greater reduction in incontinence episodes with a wearable external SNM device in the test stimulation (stage 1), they received the chronic SNM implant device (stage 2).
Participants who underwent device implantation were followed at 1, 3, and 6 months and annually for 5 years or until they exited the study. Bowel diaries and quality of life assessments were completed at baseline and at follow-up.
The primary outcome was therapeutic success, defined as 50% or greater improvement in FI episodes per week.
A total of 120 participants (90%) underwent implantation of the chronic lead and neuromodulator, and 76 (63%) were followed for 5 years. Baseline characteristics available in the initial study of 133 participants showed that the mean age was 60.5 years; 25% had undergone a prior anal sphincteroplasty; and 16.5% and 10.5% had EAS or internal anal sphincter (IAS) defects, respectively, on endoanal ultrasonography.16
Therapeutic success was high at 5 years
At the 5-year follow-up, 89% (64/72) of participants met therapeutic success, with a reduction in weekly FI episodes from 9.1 at baseline to 1.7 at 5 years. The number of incontinence pads required decreased, and more participants wore no pads at 5 years. In the intention-to-treat analysis, carrying forward the baseline FI rate in participants who lacked follow-up data, the therapeutic success rate was 69%. Quality-of-life measures improved at 5 years, both statistically and by minimal clinical difference.
Adverse events. Sixty-eight percent of participants experienced device-related adverse events, including implant site pain, change in sensation of stimulation, change in efficacy, implant site infection, or neurostimulator battery depletion (neurostimulator use commonly expires after 3 to 5 years). Of these events, 80% were successfully treated with medications, reprogramming, or no intervention. The 5-year probability of device revision or replacement was 24.4%, and the 5-year probability of device explant was 19.0%.
Strengths and limitations. Overall, this study was a well-designed, multicenter trial with long-term follow-up that showed significant improvement in FI with the use of SNM. Its strengths include the enrollment of postmenopausal women who had current defects in EAS and/or IAS on endoanal ultrasonography and 25% who had a prior sphincteroplasty. The findings therefore are relevant to the gynecologic population in whom anal sphincteroplasty would not be recommended. The study also accounted for dropouts and reported the adjusted success rate of 69% at 5 years in that group.
The lack of a control arm to rule out the placebo effect is a limitation of this study, although randomized trials comparing the effect of SNM "on" versus "off" showed greater improvement with the device "on."17
Sacral neuromodulation is an excellent therapy for women with daily FI who have failed noninvasive options and desire to proceed to a more durable, long-lasting device therapy. Although adverse events may occur, they are mild and most often resolve with device reprogramming.
- Sultan AH, Monga A, Lee J, et al. An International Urogynecological Association (IUGA)/International Continence Society (ICS) joint report on the terminology for female anorectal dysfunction. Neurourol Urodyn. 2017;36:10-34.
- Bharucha AE, Dunivan G, Goode PS, et al. Epidemiology, pathophysiology, and classification of fecal incontinence: state of the science summary for the National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK) workshop. Am J Gastroenterol. 2015;110:127-136.
- Bharucha AE, Zinsmeister AR, Locke GR, et al. Symptoms and quality of life in community women with fecal incontinence. Clin Gastroenterol Hepatol. 2006;4:1004-1008.
- Perry S, Shaw C, McGrother C, et al; Leicestershire MRC Incontinence Study Team. Prevalence of faecal incontinence in adults aged 40 years or more living in the community. Gut. 2002;50:480-484.
- Ditah I, Devaki P, Luma HN, et al. Prevalence, trends, and risk factors for fecal incontinence in United States adults, 2005-2010. Clin Gastroenterol Hepatol. 2014;12:636-643.e1-2.
- Brown HW, Wexner SD, Lukacz ES. Factors associated with care seeking among women with accidental bowel leakage. Female Pelvic Med Reconstr Surg. 2013;19:66-71.
- Norton NJ. The perspective of the patient. Gastroenterology. 2004;126(1 suppl 1):S175-S179.
- Guan W, Schmuhl NB, Brown HW. Response re: If we don't ask, they won't tell: screening for urinary and fecal incontinence by primary care providers. J Am Board Fam Med. 2019;32:119.3-120.
- Whitehead WE, Borrud L, Goode PS, et al; Pelvic Floor Disorders Network. Fecal incontinence in US adults: epidemiology and risk factors. Gastroenterology. 2009;137:512-517.
- Wald A, Bharucha AE, Cosman BC, et al. ACG clinical guideline: management of benign anorectal disorders. Am J Gastroenterol. 2014;109:1141-1157.
- Bharucha AE, Zinsmeister AR, Schleck CD, et al. Bowel disturbances are the most important risk factors for late onset fecal incontinence: a population-based case-control study in women. Gastroenterology. 2010;139:1559-1566.
- Lewis SJ, Heaton KW. Stool form scale as a useful guide to intestinal transit time. Scand J Gastroenterol. 1997;32:920-924.
- Paquette IM, Varma MG, Kaiser AM, et al. The American Society of Colon and Rectal Surgeons' clinical practice guideline for the treatment of fecal incontinence. Dis Colon Rectum. 2015;58:623-636.
- American College of Obstetricians and Gynecologists. ACOG practice bulletin no. 210: Fecal incontinence. Obstet Gynecol. 2019;133:e260-e273.
- Heymen S, Scarlett Y, Jones K, et al. Randomized controlled trial shows biofeedback to be superior to pelvic floor exercises for fecal incontinence. Dis Colon Rectum. 2009;52:1730-1737.
- Wexner SD, Coller JA, Devroede G, et al. Sacral nerve stimulation for fecal incontinence: results of a 120-patient prospective multicenter study. Ann Surg. 2010;251:441-449.
- Leroi AM, Parc Y, Lehur PA, et al. Efficacy of sacral nerve stimulation for fecal incontinence: results of a multicenter double-blind crossover study. Ann Surg. 2005;242:662-669.
- Sultan AH, Monga A, Lee J, et al. An International Urogynecological Association (IUGA)/International Continence Society (ICS) joint report on the terminology for female anorectal dysfunction. Neurourol Urodyn. 2017;36:10-34.
- Bharucha AE, Dunivan G, Goode PS, et al. Epidemiology, pathophysiology, and classification of fecal incontinence: state of the science summary for the National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK) workshop. Am J Gastroenterol. 2015;110:127-136.
- Bharucha AE, Zinsmeister AR, Locke GR, et al. Symptoms and quality of life in community women with fecal incontinence. Clin Gastroenterol Hepatol. 2006;4:1004-1008.
- Perry S, Shaw C, McGrother C, et al; Leicestershire MRC Incontinence Study Team. Prevalence of faecal incontinence in adults aged 40 years or more living in the community. Gut. 2002;50:480-484.
- Ditah I, Devaki P, Luma HN, et al. Prevalence, trends, and risk factors for fecal incontinence in United States adults, 2005-2010. Clin Gastroenterol Hepatol. 2014;12:636-643.e1-2.
- Brown HW, Wexner SD, Lukacz ES. Factors associated with care seeking among women with accidental bowel leakage. Female Pelvic Med Reconstr Surg. 2013;19:66-71.
- Norton NJ. The perspective of the patient. Gastroenterology. 2004;126(1 suppl 1):S175-S179.
- Guan W, Schmuhl NB, Brown HW. Response re: If we don't ask, they won't tell: screening for urinary and fecal incontinence by primary care providers. J Am Board Fam Med. 2019;32:119.3-120.
- Whitehead WE, Borrud L, Goode PS, et al; Pelvic Floor Disorders Network. Fecal incontinence in US adults: epidemiology and risk factors. Gastroenterology. 2009;137:512-517.
- Wald A, Bharucha AE, Cosman BC, et al. ACG clinical guideline: management of benign anorectal disorders. Am J Gastroenterol. 2014;109:1141-1157.
- Bharucha AE, Zinsmeister AR, Schleck CD, et al. Bowel disturbances are the most important risk factors for late onset fecal incontinence: a population-based case-control study in women. Gastroenterology. 2010;139:1559-1566.
- Lewis SJ, Heaton KW. Stool form scale as a useful guide to intestinal transit time. Scand J Gastroenterol. 1997;32:920-924.
- Paquette IM, Varma MG, Kaiser AM, et al. The American Society of Colon and Rectal Surgeons' clinical practice guideline for the treatment of fecal incontinence. Dis Colon Rectum. 2015;58:623-636.
- American College of Obstetricians and Gynecologists. ACOG practice bulletin no. 210: Fecal incontinence. Obstet Gynecol. 2019;133:e260-e273.
- Heymen S, Scarlett Y, Jones K, et al. Randomized controlled trial shows biofeedback to be superior to pelvic floor exercises for fecal incontinence. Dis Colon Rectum. 2009;52:1730-1737.
- Wexner SD, Coller JA, Devroede G, et al. Sacral nerve stimulation for fecal incontinence: results of a 120-patient prospective multicenter study. Ann Surg. 2010;251:441-449.
- Leroi AM, Parc Y, Lehur PA, et al. Efficacy of sacral nerve stimulation for fecal incontinence: results of a multicenter double-blind crossover study. Ann Surg. 2005;242:662-669.
Native tissue repair of POP: Surgical techniques to improve outcomes
“Take pride in your surgical work. Do it in such a way that you would be willing to sign your name to it…the operation was performed by me.”
—Raymond A. Lee, MD
The US Food and Drug Administration (FDA) recently ordered companies to cease selling transvaginal mesh intended for pelvic organ prolapse (POP) repair (but not for the treatment of stress urinary incontinence [SUI] or for abdominal sacrocolpopexy).1,2 The FDA is also requiring companies preparing premarket approval applications for mesh products for the treatment of transvaginal POP to continue safety and efficacy follow-up in existing section 522 postmarket surveillance studies.3
It is, therefore, incumbent upon gynecologic surgeons to understand the surgical options that remain and perfect their surgical approach to POP to optimize patient outcomes. POP may be performed transvaginally or transabdominally, with each approach offering its own set of risks and benefits. The ability to perform both effectively allows the surgeon to tailor the approach to the condition and circumstances encountered. It is also important to realize that “cures” are elusive in POP surgery. While we can frequently alleviate patient symptoms and improve quality of life, a lifelong “cure” is an unrealistic goal for most prolapse procedures.
This article focuses on transvaginal native tissue repair,4 specifically the Mayo approach.
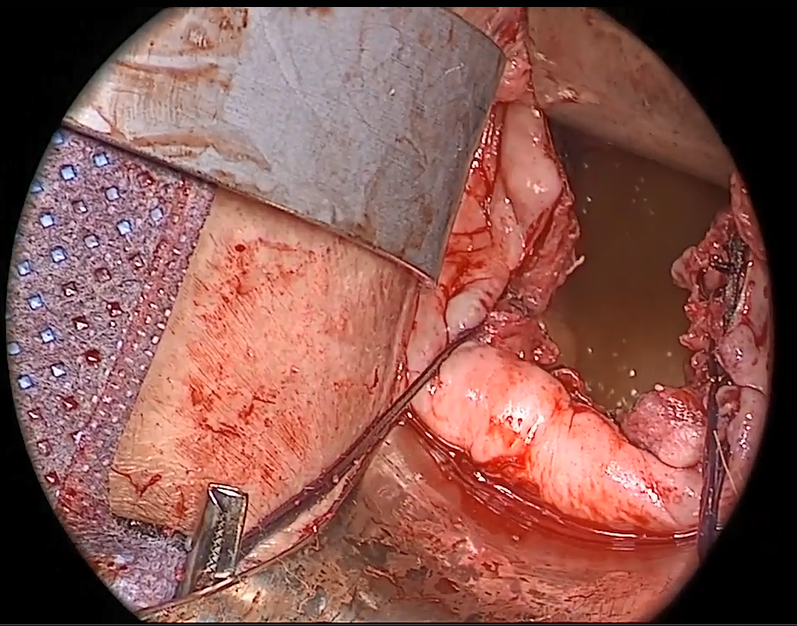
Watch video here
Vaginal surgery fundamentals
Before we explore the details of the Mayo technique, let’s review some basic principles of vaginal surgery. First, it is important to make a good clinical diagnosis so that you know which compartments (apex, anterior, or posterior) are involved. Although single compartment defects exist, multicompartment defects are far more common. Failing to recognize all compartment defects often results in incomplete repair, which can mean recurrent prolapse and additional interventions.
Second, exposure is critical when performing surgery by any route. You must be able to see your surgical field completely in order to properly execute your surgical approach. Table height, lighting, and retraction are all important to surgical success.
Lastly, it is important to know how to effectively execute your intended procedure. Native tissue repair is often criticized for having a high failure rate. It makes sense that mesh augmentation offers greater durability of a repair, but an effective native tissue repair will also effectively treat the majority of patients. An ineffective repair does not benefit the patient and contributes to high failure rates.
- Mesh slings for urinary incontinence and mesh use in sacrocolpopexy have not been banned by the FDA.
- Apical support is helpful to all other compartment support.
- Fixing the fascial defect between the base of the bladder and the apex will improve your anterior compartment outcomes.
- Monitor vaginal caliber throughout your posterior compartment repair.
Vaginal apex repairs
Data from the OPTIMAL trial suggest that uterosacral ligament suspension and sacrospinous ligament fixation are equally effective in treating apical prolapse.5 Our preference is a McCall culdoplasty (uterosacral ligament plication). It allows direct visualization (internally or externally) to place apical support stitches and plicates the ligaments in the midline of the vaginal cuff to help prevent enterocele protrusion. DeLancey has described the levels of support in the female pelvis and places importance on apical support.6 Keep in mind that anterior and posterior compartment prolapse is often accompanied by apical prolapse. Therefore, treating the apex is critical for overall success.
External vs internal McCall sutures: My technique. Envision the open vaginal cuff after completing a vaginal hysterectomy or after opening the vaginal cuff for a posthysterectomy vaginal vault prolapse (FIGURE 1). External (suture placed through the vaginal cuff epithelium into the peritoneal cavity, incorporating the uterosacral ligaments and intervening peritoneum, and ultimately brought back out through the posterior cuff and tied) or internal (suture placed in the intraperitoneal space, incorporating the uterosacral ligaments and intervening peritoneum, and tied internally) McCall sutures can be utilized (FIGURE 2). I prefer a combination of both. I use 0-polyglactin for external sutures, as the sutures will ultimately dissolve and not remain in the vaginal cavity. I usually place at least 2 external sutures with the lowest suture on the vaginal cuff being the deepest uterosacral stitch. Each subsequent suture is placed closer to the vaginal cuff and closer to the ends of the ligamentous stumps, starting deepest and working back toward the cuff with each stitch. I place 1 or 2 internal sutures (delayed absorbable or permanent) between my 2 external sutures. Because these sutures will be tied internally and located in the intraperitoneal space, permanent sutures may be used.
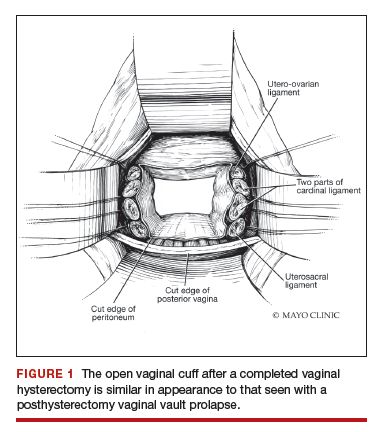
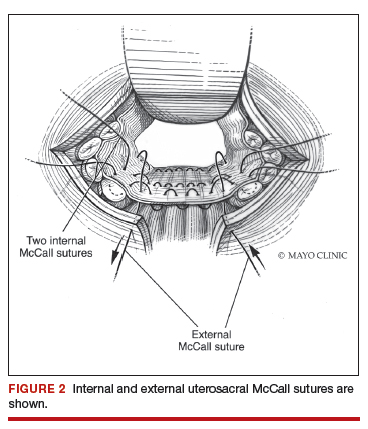
Avoiding ureteral injury: Tips for cystoscopy. A known risk of performing uterosacral ligament stitches is kinking or injury to the ureter. Therefore, cystoscopy is mandatory when performing this procedure. I tie one suture at a time starting with the internal sutures. I then perform cystoscopy after each suture tying. If I do not get ureteral spill after tying the suture, I remove and replace the suture and repeat cystoscopy until normal bilateral ureteral spill is achieved.
Key points for uterosacral ligament suspension. Achieving apical support at this point gives me the ability to build my anterior and posterior repair procedures off of this support. It is critical when performing uterosacral ligament suspension that you define the space between the ureter and rectum on each side. (Elevation of the cardinal pedicle and medial retraction of the rectum facilitate this.) The ligament runs down toward the sacrum when the patient is supine. You must follow that trajectory to be successful and avoid injury. One must also be careful not to be too deep on the ligament, as plication at that level may cause defecatory dysfunction.
Continue to: Anterior compartment repairs...
Anterior compartment repairs
The anterior compartment seems the most susceptible to forces within the pelvis and is a common site of prolapse. Many theories exist as to what causes a cystocele—distension, displacement, detachment, etc. While paravaginal defects exist, I believe that most cystoceles arise horizontally at the base of the bladder as the anterior endopelvic fascia detaches from the apex or cervix. The tissue then attenuates as the hernia progresses.
For surgical success: Make certain your repair addresses re-establishing continuity of the anterior endopelvic fascia with the fascia and ligaments at the vaginal apex; it will increase your success in treating anterior compartment prolapse.
We prefer to mobilize the epithelium in the midline from the vaginal apex to the mid‑urethra (if performing a midurethral sling, we stop short of the bladder neck and perform a separate suburethral incision). When incising the epithelium in the midline, the underlying fascia is also split in the midline, creating a midline defect. Once the epithelium is split and mobilized laterally off the underlying fascia, we can begin reconstruction.
The midline fascial defect that was just created is closed with a running 2-0 polyglactin from just beneath the bladder neck down to and including the fascia and uterosacral ligaments at the apex. This is accomplished in an upside down ‘T’ orientation (FIGURE 3). It is critical that the fascia is reunited at the base or you will leave the patient with a hernia.
For surgical success: To check intraoperatively that the fascia is reunited at the base, try to place an index finger between the base of the cystocele repair and the apex. If you can insert your finger, that is where the hernia still exists. If you meet resistance with your finger, you are palpating reunification of the anterior and apical fascia.
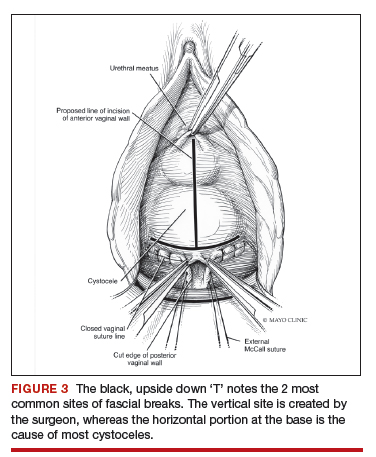
Technique for Kelly-Kennedy bladder neck plication. If the patient has mild incontinence that does not require a sling procedure, we now complete the second portion of the anterior repair starting with a Kelly-Kennedy bladder neck plication. Utilizing interrupted 1-0 polyglactin suture, vertical bites are taken periurethrally, starting at the midurethra and then the bladder neck. This nicely supports the urethra and proximal bladder neck and is very helpful for mild incontinence or for prophylactic benefit. Then starting beneath the bladder neck, the fascia is plicated again in the midline, reinforcing the suture line of the inverse ‘T’ with 2-0 polyglactin. The redundant epithelium is trimmed and reapproximated with interrupted 2-0 polyglactin (FIGURE 4). We tend to be more aggressive by adding the Kelly-Kennedy plication, which can lead to temporary voiding delay. We offer placement of a suprapubic catheter at the time of surgery or self-intermittent catherization.
Lastly, given that we have just dissected and then plicated the tissues beneath the bladder, I like to perform cystoscopy to be certain the bladder has not been violated. It is also important not to over-plicate the anterior fascia so that the sutures shear through the fascia and weaken the support or narrow the vaginal lumen.
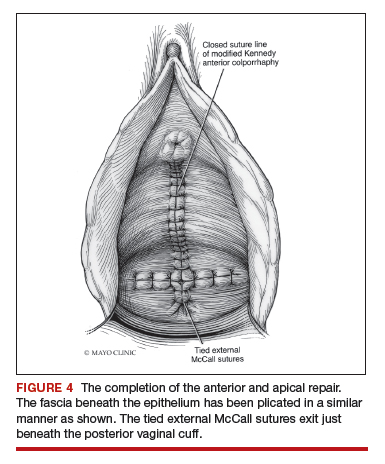
Continue to: Posterior compartment repairs...
Posterior compartment repairs
Like with the anterior compartment, opinions differ as to the site of posterior compartment prolapse. Midline, lateral, distal, and site-specific defects and surgical approaches have been described. Research suggests that there is no benefit to the use of mesh in the posterior compartment.7 It is very important to recognize that over-plication of the posterior compartment can lead to narrowing/stricture and dyspareunia. Therefore, monitor vaginal caliber throughout repair of the posterior compartment.
Although we believe that a midline defect in the endopelvic fascia is primarily responsible for rectoceles, we also appreciate that the fascia must be reconstructed all the way to the perineal body and that narrowing the genital hiatus is very important and often underappreciated (FIGURE 5). Thus, perineal reconstruction is universally performed. I will emphasize again that reconstruction must be performed while also monitoring vaginal caliber. If it is too tight with the patient under anesthesia, it will be too tight when the patient recovers. Avoidance is the best option. If the patient does not desire a functional vagina (eg, an elderly patient), then narrowing is a desired goal.

Perineal reconstruction technique and tips for success
A retractor at 12 o’clock to support the apex and anterior wall can be helpful for visualization in the posterior compartment. We start with a v-shaped incision on the perineum. The width is determined by how much you want to build up the perineum and narrow the vagina (the wider the incision, the more building up of the perineal body and vaginal narrowing). A strip of epithelium is then mobilized in the midline (be careful not to excise too much). This dissection is carried all the way up the midline to just short of the tied apical suspension sutures at the posterior vaginal apex. The posterior dissection tends to be the most vascular in my experience.
Utilize cautery to obtain hemostasis along your dissection margins while protecting the underlying rectum. We have not found it necessary to dissect the posterior epithelium off the underlying fascia (that is an option at this point, however, if you feel more comfortable doing this). With an index finger in the vagina, compressing the rectum posteriorly, interrupted 1-0 polyglactin suture is placed through the epithelium and underlying fascia (avoiding the rectum) on one side, then the other, and then tied. The next sutures are placed utilizing the same technique, and the caliber of the vagina is noted with the placement of each suture (if it is too tight, then remove and replace the suture and recheck). It is important to realize you want to plicate the fascia in the midline and not perform an aggressive levatorplasty that could lead to muscle pain. Additionally, each suture should get the same purchase of tissue on each side, and the spacing of each suture should be uniform, like rungs on a ladder. Ultimately, the repair is carried down to the hymenal ring. At this point, the perineal reconstruction is performed, plicating the perineal body in the midline with deeper horizontal sutures and then closing the perineal skin with interrupted or subcuticular sutures (FIGURE 6). Completion of these repairs should orient the vagina toward the hollow of the sacrum (FIGURE 7), allowing downward forces to compress the vaginal supports posteriorly onto the pelvic floor instead of forcing it out the vaginal lumen (FIGURE 8).
Our patients generally stay in the hospital overnight, and we place a vaginal pack to provide topical pressure throughout the vagina overnight. We tell patients no lifting more than 15 lb and no intercourse for 6 weeks. While we do not tend to use hydrodissection in our repairs, it is a perfectly acceptable option.
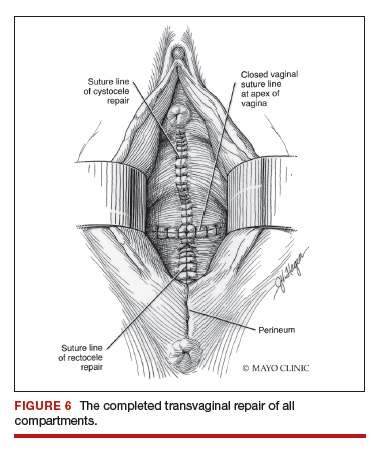
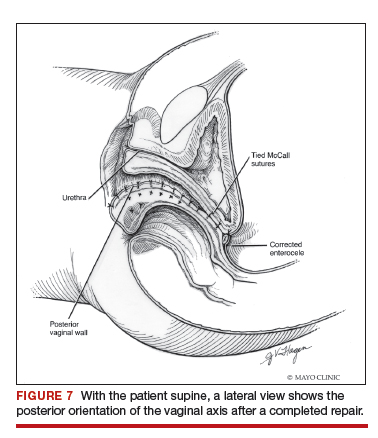
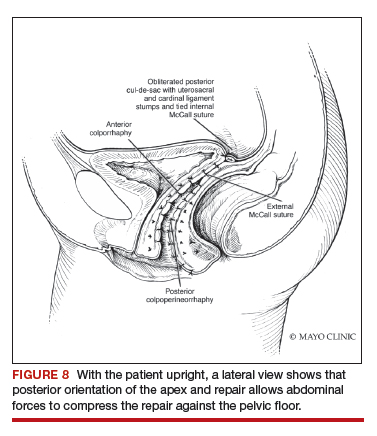
Continue to: Commit to knowledge of native tissue techniques...
Commit to knowledge of native tissue techniques
Given the recent FDA ban on the sale of transvaginal mesh for POP and the public’s negative perception of mesh (based often on misleading information in the media), it is incumbent upon gynecologic surgeons to invest in learning or relearning effective native tissue techniques for the transvaginal treatment of POP. While not perfect, they offer an effective nonmesh treatment option for many of our patients.
- US Food and Drug Administration. FDA takes action to protect women’s health, orders manufacturers of surgical mesh intended for transvaginal repair of pelvic organ prolapse to stop selling all devices. . Published April 16, 2019. Accessed August 6, 2019.
- US Food and Drug Administration. Urogynecological surgical mesh implants. . Published July 10, 2019. Accessed August 5, 2019.
- US Food and Drug Administration. Effective date of requirement for premarket approval for surgical mesh for transvaginal pelvic organ prolapse repair. https://www.federalregister.gov/documents/2016/01/05/2015-33163/effective-date-of-requirement-for-premarket-approval-for-surgical-mesh-for-transvaginal-pelvic-organ. Published January 5, 2016. Accessed August 5, 2019.
- Lee RA. Atlas of Gynecologic Surgery. W.B. Saunders: Philadelphia, PA; 1992.
- Jelovsek JE, Barber MD, Brubaker L, et al. Effect of uterosacral ligament suspension vs sacrospinous ligament fixation with or without perioperative behavioral therapy for pelvic organ vaginal prolapse on surgical outcomes and prolapse symptoms at 5 years in the OPTIMAL randomized clinical trial. JAMA. 2018;319:1554-1565.
- DeLancey JO. Anatomic aspects of vaginal eversion after hysterectomy. Am J Obstet Gynecol. 1992;166(6 part 1):1717-1728.
- Paraiso MF, Barber MD, Muir TW, et al. Rectocele repair: a randomized trial of three surgical techniques including graft augmentation. Am J Obstet Gynecol. 2006;195:1762- 1771.
“Take pride in your surgical work. Do it in such a way that you would be willing to sign your name to it…the operation was performed by me.”
—Raymond A. Lee, MD
The US Food and Drug Administration (FDA) recently ordered companies to cease selling transvaginal mesh intended for pelvic organ prolapse (POP) repair (but not for the treatment of stress urinary incontinence [SUI] or for abdominal sacrocolpopexy).1,2 The FDA is also requiring companies preparing premarket approval applications for mesh products for the treatment of transvaginal POP to continue safety and efficacy follow-up in existing section 522 postmarket surveillance studies.3
It is, therefore, incumbent upon gynecologic surgeons to understand the surgical options that remain and perfect their surgical approach to POP to optimize patient outcomes. POP may be performed transvaginally or transabdominally, with each approach offering its own set of risks and benefits. The ability to perform both effectively allows the surgeon to tailor the approach to the condition and circumstances encountered. It is also important to realize that “cures” are elusive in POP surgery. While we can frequently alleviate patient symptoms and improve quality of life, a lifelong “cure” is an unrealistic goal for most prolapse procedures.
This article focuses on transvaginal native tissue repair,4 specifically the Mayo approach.

Watch video here
Vaginal surgery fundamentals
Before we explore the details of the Mayo technique, let’s review some basic principles of vaginal surgery. First, it is important to make a good clinical diagnosis so that you know which compartments (apex, anterior, or posterior) are involved. Although single compartment defects exist, multicompartment defects are far more common. Failing to recognize all compartment defects often results in incomplete repair, which can mean recurrent prolapse and additional interventions.
Second, exposure is critical when performing surgery by any route. You must be able to see your surgical field completely in order to properly execute your surgical approach. Table height, lighting, and retraction are all important to surgical success.
Lastly, it is important to know how to effectively execute your intended procedure. Native tissue repair is often criticized for having a high failure rate. It makes sense that mesh augmentation offers greater durability of a repair, but an effective native tissue repair will also effectively treat the majority of patients. An ineffective repair does not benefit the patient and contributes to high failure rates.
- Mesh slings for urinary incontinence and mesh use in sacrocolpopexy have not been banned by the FDA.
- Apical support is helpful to all other compartment support.
- Fixing the fascial defect between the base of the bladder and the apex will improve your anterior compartment outcomes.
- Monitor vaginal caliber throughout your posterior compartment repair.
Vaginal apex repairs
Data from the OPTIMAL trial suggest that uterosacral ligament suspension and sacrospinous ligament fixation are equally effective in treating apical prolapse.5 Our preference is a McCall culdoplasty (uterosacral ligament plication). It allows direct visualization (internally or externally) to place apical support stitches and plicates the ligaments in the midline of the vaginal cuff to help prevent enterocele protrusion. DeLancey has described the levels of support in the female pelvis and places importance on apical support.6 Keep in mind that anterior and posterior compartment prolapse is often accompanied by apical prolapse. Therefore, treating the apex is critical for overall success.
External vs internal McCall sutures: My technique. Envision the open vaginal cuff after completing a vaginal hysterectomy or after opening the vaginal cuff for a posthysterectomy vaginal vault prolapse (FIGURE 1). External (suture placed through the vaginal cuff epithelium into the peritoneal cavity, incorporating the uterosacral ligaments and intervening peritoneum, and ultimately brought back out through the posterior cuff and tied) or internal (suture placed in the intraperitoneal space, incorporating the uterosacral ligaments and intervening peritoneum, and tied internally) McCall sutures can be utilized (FIGURE 2). I prefer a combination of both. I use 0-polyglactin for external sutures, as the sutures will ultimately dissolve and not remain in the vaginal cavity. I usually place at least 2 external sutures with the lowest suture on the vaginal cuff being the deepest uterosacral stitch. Each subsequent suture is placed closer to the vaginal cuff and closer to the ends of the ligamentous stumps, starting deepest and working back toward the cuff with each stitch. I place 1 or 2 internal sutures (delayed absorbable or permanent) between my 2 external sutures. Because these sutures will be tied internally and located in the intraperitoneal space, permanent sutures may be used.


Avoiding ureteral injury: Tips for cystoscopy. A known risk of performing uterosacral ligament stitches is kinking or injury to the ureter. Therefore, cystoscopy is mandatory when performing this procedure. I tie one suture at a time starting with the internal sutures. I then perform cystoscopy after each suture tying. If I do not get ureteral spill after tying the suture, I remove and replace the suture and repeat cystoscopy until normal bilateral ureteral spill is achieved.
Key points for uterosacral ligament suspension. Achieving apical support at this point gives me the ability to build my anterior and posterior repair procedures off of this support. It is critical when performing uterosacral ligament suspension that you define the space between the ureter and rectum on each side. (Elevation of the cardinal pedicle and medial retraction of the rectum facilitate this.) The ligament runs down toward the sacrum when the patient is supine. You must follow that trajectory to be successful and avoid injury. One must also be careful not to be too deep on the ligament, as plication at that level may cause defecatory dysfunction.
Continue to: Anterior compartment repairs...
Anterior compartment repairs
The anterior compartment seems the most susceptible to forces within the pelvis and is a common site of prolapse. Many theories exist as to what causes a cystocele—distension, displacement, detachment, etc. While paravaginal defects exist, I believe that most cystoceles arise horizontally at the base of the bladder as the anterior endopelvic fascia detaches from the apex or cervix. The tissue then attenuates as the hernia progresses.
For surgical success: Make certain your repair addresses re-establishing continuity of the anterior endopelvic fascia with the fascia and ligaments at the vaginal apex; it will increase your success in treating anterior compartment prolapse.
We prefer to mobilize the epithelium in the midline from the vaginal apex to the mid‑urethra (if performing a midurethral sling, we stop short of the bladder neck and perform a separate suburethral incision). When incising the epithelium in the midline, the underlying fascia is also split in the midline, creating a midline defect. Once the epithelium is split and mobilized laterally off the underlying fascia, we can begin reconstruction.
The midline fascial defect that was just created is closed with a running 2-0 polyglactin from just beneath the bladder neck down to and including the fascia and uterosacral ligaments at the apex. This is accomplished in an upside down ‘T’ orientation (FIGURE 3). It is critical that the fascia is reunited at the base or you will leave the patient with a hernia.
For surgical success: To check intraoperatively that the fascia is reunited at the base, try to place an index finger between the base of the cystocele repair and the apex. If you can insert your finger, that is where the hernia still exists. If you meet resistance with your finger, you are palpating reunification of the anterior and apical fascia.

Technique for Kelly-Kennedy bladder neck plication. If the patient has mild incontinence that does not require a sling procedure, we now complete the second portion of the anterior repair starting with a Kelly-Kennedy bladder neck plication. Utilizing interrupted 1-0 polyglactin suture, vertical bites are taken periurethrally, starting at the midurethra and then the bladder neck. This nicely supports the urethra and proximal bladder neck and is very helpful for mild incontinence or for prophylactic benefit. Then starting beneath the bladder neck, the fascia is plicated again in the midline, reinforcing the suture line of the inverse ‘T’ with 2-0 polyglactin. The redundant epithelium is trimmed and reapproximated with interrupted 2-0 polyglactin (FIGURE 4). We tend to be more aggressive by adding the Kelly-Kennedy plication, which can lead to temporary voiding delay. We offer placement of a suprapubic catheter at the time of surgery or self-intermittent catherization.
Lastly, given that we have just dissected and then plicated the tissues beneath the bladder, I like to perform cystoscopy to be certain the bladder has not been violated. It is also important not to over-plicate the anterior fascia so that the sutures shear through the fascia and weaken the support or narrow the vaginal lumen.

Continue to: Posterior compartment repairs...
Posterior compartment repairs
Like with the anterior compartment, opinions differ as to the site of posterior compartment prolapse. Midline, lateral, distal, and site-specific defects and surgical approaches have been described. Research suggests that there is no benefit to the use of mesh in the posterior compartment.7 It is very important to recognize that over-plication of the posterior compartment can lead to narrowing/stricture and dyspareunia. Therefore, monitor vaginal caliber throughout repair of the posterior compartment.
Although we believe that a midline defect in the endopelvic fascia is primarily responsible for rectoceles, we also appreciate that the fascia must be reconstructed all the way to the perineal body and that narrowing the genital hiatus is very important and often underappreciated (FIGURE 5). Thus, perineal reconstruction is universally performed. I will emphasize again that reconstruction must be performed while also monitoring vaginal caliber. If it is too tight with the patient under anesthesia, it will be too tight when the patient recovers. Avoidance is the best option. If the patient does not desire a functional vagina (eg, an elderly patient), then narrowing is a desired goal.

Perineal reconstruction technique and tips for success
A retractor at 12 o’clock to support the apex and anterior wall can be helpful for visualization in the posterior compartment. We start with a v-shaped incision on the perineum. The width is determined by how much you want to build up the perineum and narrow the vagina (the wider the incision, the more building up of the perineal body and vaginal narrowing). A strip of epithelium is then mobilized in the midline (be careful not to excise too much). This dissection is carried all the way up the midline to just short of the tied apical suspension sutures at the posterior vaginal apex. The posterior dissection tends to be the most vascular in my experience.
Utilize cautery to obtain hemostasis along your dissection margins while protecting the underlying rectum. We have not found it necessary to dissect the posterior epithelium off the underlying fascia (that is an option at this point, however, if you feel more comfortable doing this). With an index finger in the vagina, compressing the rectum posteriorly, interrupted 1-0 polyglactin suture is placed through the epithelium and underlying fascia (avoiding the rectum) on one side, then the other, and then tied. The next sutures are placed utilizing the same technique, and the caliber of the vagina is noted with the placement of each suture (if it is too tight, then remove and replace the suture and recheck). It is important to realize you want to plicate the fascia in the midline and not perform an aggressive levatorplasty that could lead to muscle pain. Additionally, each suture should get the same purchase of tissue on each side, and the spacing of each suture should be uniform, like rungs on a ladder. Ultimately, the repair is carried down to the hymenal ring. At this point, the perineal reconstruction is performed, plicating the perineal body in the midline with deeper horizontal sutures and then closing the perineal skin with interrupted or subcuticular sutures (FIGURE 6). Completion of these repairs should orient the vagina toward the hollow of the sacrum (FIGURE 7), allowing downward forces to compress the vaginal supports posteriorly onto the pelvic floor instead of forcing it out the vaginal lumen (FIGURE 8).
Our patients generally stay in the hospital overnight, and we place a vaginal pack to provide topical pressure throughout the vagina overnight. We tell patients no lifting more than 15 lb and no intercourse for 6 weeks. While we do not tend to use hydrodissection in our repairs, it is a perfectly acceptable option.



Continue to: Commit to knowledge of native tissue techniques...
Commit to knowledge of native tissue techniques
Given the recent FDA ban on the sale of transvaginal mesh for POP and the public’s negative perception of mesh (based often on misleading information in the media), it is incumbent upon gynecologic surgeons to invest in learning or relearning effective native tissue techniques for the transvaginal treatment of POP. While not perfect, they offer an effective nonmesh treatment option for many of our patients.
“Take pride in your surgical work. Do it in such a way that you would be willing to sign your name to it…the operation was performed by me.”
—Raymond A. Lee, MD
The US Food and Drug Administration (FDA) recently ordered companies to cease selling transvaginal mesh intended for pelvic organ prolapse (POP) repair (but not for the treatment of stress urinary incontinence [SUI] or for abdominal sacrocolpopexy).1,2 The FDA is also requiring companies preparing premarket approval applications for mesh products for the treatment of transvaginal POP to continue safety and efficacy follow-up in existing section 522 postmarket surveillance studies.3
It is, therefore, incumbent upon gynecologic surgeons to understand the surgical options that remain and perfect their surgical approach to POP to optimize patient outcomes. POP may be performed transvaginally or transabdominally, with each approach offering its own set of risks and benefits. The ability to perform both effectively allows the surgeon to tailor the approach to the condition and circumstances encountered. It is also important to realize that “cures” are elusive in POP surgery. While we can frequently alleviate patient symptoms and improve quality of life, a lifelong “cure” is an unrealistic goal for most prolapse procedures.
This article focuses on transvaginal native tissue repair,4 specifically the Mayo approach.

Watch video here
Vaginal surgery fundamentals
Before we explore the details of the Mayo technique, let’s review some basic principles of vaginal surgery. First, it is important to make a good clinical diagnosis so that you know which compartments (apex, anterior, or posterior) are involved. Although single compartment defects exist, multicompartment defects are far more common. Failing to recognize all compartment defects often results in incomplete repair, which can mean recurrent prolapse and additional interventions.
Second, exposure is critical when performing surgery by any route. You must be able to see your surgical field completely in order to properly execute your surgical approach. Table height, lighting, and retraction are all important to surgical success.
Lastly, it is important to know how to effectively execute your intended procedure. Native tissue repair is often criticized for having a high failure rate. It makes sense that mesh augmentation offers greater durability of a repair, but an effective native tissue repair will also effectively treat the majority of patients. An ineffective repair does not benefit the patient and contributes to high failure rates.
- Mesh slings for urinary incontinence and mesh use in sacrocolpopexy have not been banned by the FDA.
- Apical support is helpful to all other compartment support.
- Fixing the fascial defect between the base of the bladder and the apex will improve your anterior compartment outcomes.
- Monitor vaginal caliber throughout your posterior compartment repair.
Vaginal apex repairs
Data from the OPTIMAL trial suggest that uterosacral ligament suspension and sacrospinous ligament fixation are equally effective in treating apical prolapse.5 Our preference is a McCall culdoplasty (uterosacral ligament plication). It allows direct visualization (internally or externally) to place apical support stitches and plicates the ligaments in the midline of the vaginal cuff to help prevent enterocele protrusion. DeLancey has described the levels of support in the female pelvis and places importance on apical support.6 Keep in mind that anterior and posterior compartment prolapse is often accompanied by apical prolapse. Therefore, treating the apex is critical for overall success.
External vs internal McCall sutures: My technique. Envision the open vaginal cuff after completing a vaginal hysterectomy or after opening the vaginal cuff for a posthysterectomy vaginal vault prolapse (FIGURE 1). External (suture placed through the vaginal cuff epithelium into the peritoneal cavity, incorporating the uterosacral ligaments and intervening peritoneum, and ultimately brought back out through the posterior cuff and tied) or internal (suture placed in the intraperitoneal space, incorporating the uterosacral ligaments and intervening peritoneum, and tied internally) McCall sutures can be utilized (FIGURE 2). I prefer a combination of both. I use 0-polyglactin for external sutures, as the sutures will ultimately dissolve and not remain in the vaginal cavity. I usually place at least 2 external sutures with the lowest suture on the vaginal cuff being the deepest uterosacral stitch. Each subsequent suture is placed closer to the vaginal cuff and closer to the ends of the ligamentous stumps, starting deepest and working back toward the cuff with each stitch. I place 1 or 2 internal sutures (delayed absorbable or permanent) between my 2 external sutures. Because these sutures will be tied internally and located in the intraperitoneal space, permanent sutures may be used.


Avoiding ureteral injury: Tips for cystoscopy. A known risk of performing uterosacral ligament stitches is kinking or injury to the ureter. Therefore, cystoscopy is mandatory when performing this procedure. I tie one suture at a time starting with the internal sutures. I then perform cystoscopy after each suture tying. If I do not get ureteral spill after tying the suture, I remove and replace the suture and repeat cystoscopy until normal bilateral ureteral spill is achieved.
Key points for uterosacral ligament suspension. Achieving apical support at this point gives me the ability to build my anterior and posterior repair procedures off of this support. It is critical when performing uterosacral ligament suspension that you define the space between the ureter and rectum on each side. (Elevation of the cardinal pedicle and medial retraction of the rectum facilitate this.) The ligament runs down toward the sacrum when the patient is supine. You must follow that trajectory to be successful and avoid injury. One must also be careful not to be too deep on the ligament, as plication at that level may cause defecatory dysfunction.
Continue to: Anterior compartment repairs...
Anterior compartment repairs
The anterior compartment seems the most susceptible to forces within the pelvis and is a common site of prolapse. Many theories exist as to what causes a cystocele—distension, displacement, detachment, etc. While paravaginal defects exist, I believe that most cystoceles arise horizontally at the base of the bladder as the anterior endopelvic fascia detaches from the apex or cervix. The tissue then attenuates as the hernia progresses.
For surgical success: Make certain your repair addresses re-establishing continuity of the anterior endopelvic fascia with the fascia and ligaments at the vaginal apex; it will increase your success in treating anterior compartment prolapse.
We prefer to mobilize the epithelium in the midline from the vaginal apex to the mid‑urethra (if performing a midurethral sling, we stop short of the bladder neck and perform a separate suburethral incision). When incising the epithelium in the midline, the underlying fascia is also split in the midline, creating a midline defect. Once the epithelium is split and mobilized laterally off the underlying fascia, we can begin reconstruction.
The midline fascial defect that was just created is closed with a running 2-0 polyglactin from just beneath the bladder neck down to and including the fascia and uterosacral ligaments at the apex. This is accomplished in an upside down ‘T’ orientation (FIGURE 3). It is critical that the fascia is reunited at the base or you will leave the patient with a hernia.
For surgical success: To check intraoperatively that the fascia is reunited at the base, try to place an index finger between the base of the cystocele repair and the apex. If you can insert your finger, that is where the hernia still exists. If you meet resistance with your finger, you are palpating reunification of the anterior and apical fascia.

Technique for Kelly-Kennedy bladder neck plication. If the patient has mild incontinence that does not require a sling procedure, we now complete the second portion of the anterior repair starting with a Kelly-Kennedy bladder neck plication. Utilizing interrupted 1-0 polyglactin suture, vertical bites are taken periurethrally, starting at the midurethra and then the bladder neck. This nicely supports the urethra and proximal bladder neck and is very helpful for mild incontinence or for prophylactic benefit. Then starting beneath the bladder neck, the fascia is plicated again in the midline, reinforcing the suture line of the inverse ‘T’ with 2-0 polyglactin. The redundant epithelium is trimmed and reapproximated with interrupted 2-0 polyglactin (FIGURE 4). We tend to be more aggressive by adding the Kelly-Kennedy plication, which can lead to temporary voiding delay. We offer placement of a suprapubic catheter at the time of surgery or self-intermittent catherization.
Lastly, given that we have just dissected and then plicated the tissues beneath the bladder, I like to perform cystoscopy to be certain the bladder has not been violated. It is also important not to over-plicate the anterior fascia so that the sutures shear through the fascia and weaken the support or narrow the vaginal lumen.

Continue to: Posterior compartment repairs...
Posterior compartment repairs
Like with the anterior compartment, opinions differ as to the site of posterior compartment prolapse. Midline, lateral, distal, and site-specific defects and surgical approaches have been described. Research suggests that there is no benefit to the use of mesh in the posterior compartment.7 It is very important to recognize that over-plication of the posterior compartment can lead to narrowing/stricture and dyspareunia. Therefore, monitor vaginal caliber throughout repair of the posterior compartment.
Although we believe that a midline defect in the endopelvic fascia is primarily responsible for rectoceles, we also appreciate that the fascia must be reconstructed all the way to the perineal body and that narrowing the genital hiatus is very important and often underappreciated (FIGURE 5). Thus, perineal reconstruction is universally performed. I will emphasize again that reconstruction must be performed while also monitoring vaginal caliber. If it is too tight with the patient under anesthesia, it will be too tight when the patient recovers. Avoidance is the best option. If the patient does not desire a functional vagina (eg, an elderly patient), then narrowing is a desired goal.

Perineal reconstruction technique and tips for success
A retractor at 12 o’clock to support the apex and anterior wall can be helpful for visualization in the posterior compartment. We start with a v-shaped incision on the perineum. The width is determined by how much you want to build up the perineum and narrow the vagina (the wider the incision, the more building up of the perineal body and vaginal narrowing). A strip of epithelium is then mobilized in the midline (be careful not to excise too much). This dissection is carried all the way up the midline to just short of the tied apical suspension sutures at the posterior vaginal apex. The posterior dissection tends to be the most vascular in my experience.
Utilize cautery to obtain hemostasis along your dissection margins while protecting the underlying rectum. We have not found it necessary to dissect the posterior epithelium off the underlying fascia (that is an option at this point, however, if you feel more comfortable doing this). With an index finger in the vagina, compressing the rectum posteriorly, interrupted 1-0 polyglactin suture is placed through the epithelium and underlying fascia (avoiding the rectum) on one side, then the other, and then tied. The next sutures are placed utilizing the same technique, and the caliber of the vagina is noted with the placement of each suture (if it is too tight, then remove and replace the suture and recheck). It is important to realize you want to plicate the fascia in the midline and not perform an aggressive levatorplasty that could lead to muscle pain. Additionally, each suture should get the same purchase of tissue on each side, and the spacing of each suture should be uniform, like rungs on a ladder. Ultimately, the repair is carried down to the hymenal ring. At this point, the perineal reconstruction is performed, plicating the perineal body in the midline with deeper horizontal sutures and then closing the perineal skin with interrupted or subcuticular sutures (FIGURE 6). Completion of these repairs should orient the vagina toward the hollow of the sacrum (FIGURE 7), allowing downward forces to compress the vaginal supports posteriorly onto the pelvic floor instead of forcing it out the vaginal lumen (FIGURE 8).
Our patients generally stay in the hospital overnight, and we place a vaginal pack to provide topical pressure throughout the vagina overnight. We tell patients no lifting more than 15 lb and no intercourse for 6 weeks. While we do not tend to use hydrodissection in our repairs, it is a perfectly acceptable option.



Continue to: Commit to knowledge of native tissue techniques...
Commit to knowledge of native tissue techniques
Given the recent FDA ban on the sale of transvaginal mesh for POP and the public’s negative perception of mesh (based often on misleading information in the media), it is incumbent upon gynecologic surgeons to invest in learning or relearning effective native tissue techniques for the transvaginal treatment of POP. While not perfect, they offer an effective nonmesh treatment option for many of our patients.
- US Food and Drug Administration. FDA takes action to protect women’s health, orders manufacturers of surgical mesh intended for transvaginal repair of pelvic organ prolapse to stop selling all devices. . Published April 16, 2019. Accessed August 6, 2019.
- US Food and Drug Administration. Urogynecological surgical mesh implants. . Published July 10, 2019. Accessed August 5, 2019.
- US Food and Drug Administration. Effective date of requirement for premarket approval for surgical mesh for transvaginal pelvic organ prolapse repair. https://www.federalregister.gov/documents/2016/01/05/2015-33163/effective-date-of-requirement-for-premarket-approval-for-surgical-mesh-for-transvaginal-pelvic-organ. Published January 5, 2016. Accessed August 5, 2019.
- Lee RA. Atlas of Gynecologic Surgery. W.B. Saunders: Philadelphia, PA; 1992.
- Jelovsek JE, Barber MD, Brubaker L, et al. Effect of uterosacral ligament suspension vs sacrospinous ligament fixation with or without perioperative behavioral therapy for pelvic organ vaginal prolapse on surgical outcomes and prolapse symptoms at 5 years in the OPTIMAL randomized clinical trial. JAMA. 2018;319:1554-1565.
- DeLancey JO. Anatomic aspects of vaginal eversion after hysterectomy. Am J Obstet Gynecol. 1992;166(6 part 1):1717-1728.
- Paraiso MF, Barber MD, Muir TW, et al. Rectocele repair: a randomized trial of three surgical techniques including graft augmentation. Am J Obstet Gynecol. 2006;195:1762- 1771.
- US Food and Drug Administration. FDA takes action to protect women’s health, orders manufacturers of surgical mesh intended for transvaginal repair of pelvic organ prolapse to stop selling all devices. . Published April 16, 2019. Accessed August 6, 2019.
- US Food and Drug Administration. Urogynecological surgical mesh implants. . Published July 10, 2019. Accessed August 5, 2019.
- US Food and Drug Administration. Effective date of requirement for premarket approval for surgical mesh for transvaginal pelvic organ prolapse repair. https://www.federalregister.gov/documents/2016/01/05/2015-33163/effective-date-of-requirement-for-premarket-approval-for-surgical-mesh-for-transvaginal-pelvic-organ. Published January 5, 2016. Accessed August 5, 2019.
- Lee RA. Atlas of Gynecologic Surgery. W.B. Saunders: Philadelphia, PA; 1992.
- Jelovsek JE, Barber MD, Brubaker L, et al. Effect of uterosacral ligament suspension vs sacrospinous ligament fixation with or without perioperative behavioral therapy for pelvic organ vaginal prolapse on surgical outcomes and prolapse symptoms at 5 years in the OPTIMAL randomized clinical trial. JAMA. 2018;319:1554-1565.
- DeLancey JO. Anatomic aspects of vaginal eversion after hysterectomy. Am J Obstet Gynecol. 1992;166(6 part 1):1717-1728.
- Paraiso MF, Barber MD, Muir TW, et al. Rectocele repair: a randomized trial of three surgical techniques including graft augmentation. Am J Obstet Gynecol. 2006;195:1762- 1771.
Would routine use of tranexamic acid for PPH be cost-effective in the United States?
Sudhof LS, Shainker SA, Einerson BD. Tranexamic acid in the routine treatment of postpartum hemorrhage in the United States: a cost-effectiveness analysis. Am J Obstet Gynecol. Published online June 18, 2019. doi.org/10.1016/j.ajog.2019.06.030.
EXPERT COMMENTARY
Postpartum hemorrhage is a leading cause of morbidity and mortality in the United States. The World Maternal Antifibrinolytic (WOMAN) trial showed that the use of TXA, an antifibrinolytic agent, for PPH decreases hemorrhage-related mortality and laparotomy. Routine use of TXA for PPH has demonstrated cost-effectiveness in low-resource countries, where hemorrhage-related mortality rates are higher than in the United States. This study aimed to determine if routine use of TXA for PPH in the United States also is cost-effective.
Details of the study
Sudhof and colleagues conducted a decision-tree analysis to compare the cost-effectiveness of 3 strategies regarding routine use of TXA for PPH in the United States: no TXA, TXA given at any time, and TXA given within 3 hours of delivery.
Health care system perspective. In the primary analysis, the 3 strategies were evaluated from the perspective of the health care system. Outcomes included cost, number of laparotomies, and maternal deaths from delivery until 6 weeks postpartum. Rates of hemorrhage and related complications, as well as cost assumptions, were derived from multiple US-based studies. The relative risk reduction in death and laparotomy with TXA in the United States was assumed to be similar to that found in the WOMAN trial (19% and 36%, respectively).
Societal perspective. In the secondary analysis, the 3 TXA strategies were evaluated from the societal perspective, comparing quality-adjusted life-years (QALYs) and cost per QALY. For both the primary and secondary analyses, sensitivity analyses were performed across a range of values for each input.
Main findings. Tranexamic acid use would be cost saving if the relative risk reduction for maternal death with TXA was greater than approximately 5%, which is significantly lower than that seen in the WOMAN trial (19%). The primary analysis demonstrated that—assuming a 3% rate of PPH—giving TXA to women with PPH would save $11.3 million, prevent 334 laparotomies, and avert 9 maternal deaths annually in the United States. This cost saving nearly tripled if TXA was administered within 3 hours of delivery, with 5 additional maternal deaths prevented.
Secondary analysis incorporating QALYs also showed TXA use to be cost-effective. These findings held through various sensitivity analyses.
Continue to: Study strengths and limitations...
Study strengths and limitations
This study is novel in its critical objective to determine the cost-effectiveness of routine use of TXA for PPH in the United States. Robust modeling using Monte Carlo estimation and a variety of sensitivity analyses add reliability to the authors’ findings.
This work is limited, however, by the assumptions put into the authors’ models. For example, outcome data regarding effectiveness of TXA was taken from the WOMAN trial, which was not performed within the United States. In addition, it is difficult to quantify in dollars an event as profound as a maternal death. The authors recognize that they likely underestimate the “cost” of a maternal death, but that this underestimation would only increase the cost-effectiveness of TXA.
Finally, it is important to take into account that such economic analyses are helpful to inform institutional guidelines and hemorrhage protocols, but that patient-specific decision-making should be individualized based on the clinical scenario at hand.
Routine use of TXA for PPH, particularly within 3 hours of delivery, is likely cost-effective in the United States. Consideration should be given to including TXA in institutional hemorrhage protocols.
REBECCA F. HAMM, MD, and ADI HIRSHBERG, MD
Sudhof LS, Shainker SA, Einerson BD. Tranexamic acid in the routine treatment of postpartum hemorrhage in the United States: a cost-effectiveness analysis. Am J Obstet Gynecol. Published online June 18, 2019. doi.org/10.1016/j.ajog.2019.06.030.
EXPERT COMMENTARY
Postpartum hemorrhage is a leading cause of morbidity and mortality in the United States. The World Maternal Antifibrinolytic (WOMAN) trial showed that the use of TXA, an antifibrinolytic agent, for PPH decreases hemorrhage-related mortality and laparotomy. Routine use of TXA for PPH has demonstrated cost-effectiveness in low-resource countries, where hemorrhage-related mortality rates are higher than in the United States. This study aimed to determine if routine use of TXA for PPH in the United States also is cost-effective.
Details of the study
Sudhof and colleagues conducted a decision-tree analysis to compare the cost-effectiveness of 3 strategies regarding routine use of TXA for PPH in the United States: no TXA, TXA given at any time, and TXA given within 3 hours of delivery.
Health care system perspective. In the primary analysis, the 3 strategies were evaluated from the perspective of the health care system. Outcomes included cost, number of laparotomies, and maternal deaths from delivery until 6 weeks postpartum. Rates of hemorrhage and related complications, as well as cost assumptions, were derived from multiple US-based studies. The relative risk reduction in death and laparotomy with TXA in the United States was assumed to be similar to that found in the WOMAN trial (19% and 36%, respectively).
Societal perspective. In the secondary analysis, the 3 TXA strategies were evaluated from the societal perspective, comparing quality-adjusted life-years (QALYs) and cost per QALY. For both the primary and secondary analyses, sensitivity analyses were performed across a range of values for each input.
Main findings. Tranexamic acid use would be cost saving if the relative risk reduction for maternal death with TXA was greater than approximately 5%, which is significantly lower than that seen in the WOMAN trial (19%). The primary analysis demonstrated that—assuming a 3% rate of PPH—giving TXA to women with PPH would save $11.3 million, prevent 334 laparotomies, and avert 9 maternal deaths annually in the United States. This cost saving nearly tripled if TXA was administered within 3 hours of delivery, with 5 additional maternal deaths prevented.
Secondary analysis incorporating QALYs also showed TXA use to be cost-effective. These findings held through various sensitivity analyses.
Continue to: Study strengths and limitations...
Study strengths and limitations
This study is novel in its critical objective to determine the cost-effectiveness of routine use of TXA for PPH in the United States. Robust modeling using Monte Carlo estimation and a variety of sensitivity analyses add reliability to the authors’ findings.
This work is limited, however, by the assumptions put into the authors’ models. For example, outcome data regarding effectiveness of TXA was taken from the WOMAN trial, which was not performed within the United States. In addition, it is difficult to quantify in dollars an event as profound as a maternal death. The authors recognize that they likely underestimate the “cost” of a maternal death, but that this underestimation would only increase the cost-effectiveness of TXA.
Finally, it is important to take into account that such economic analyses are helpful to inform institutional guidelines and hemorrhage protocols, but that patient-specific decision-making should be individualized based on the clinical scenario at hand.
Routine use of TXA for PPH, particularly within 3 hours of delivery, is likely cost-effective in the United States. Consideration should be given to including TXA in institutional hemorrhage protocols.
REBECCA F. HAMM, MD, and ADI HIRSHBERG, MD
Sudhof LS, Shainker SA, Einerson BD. Tranexamic acid in the routine treatment of postpartum hemorrhage in the United States: a cost-effectiveness analysis. Am J Obstet Gynecol. Published online June 18, 2019. doi.org/10.1016/j.ajog.2019.06.030.
EXPERT COMMENTARY
Postpartum hemorrhage is a leading cause of morbidity and mortality in the United States. The World Maternal Antifibrinolytic (WOMAN) trial showed that the use of TXA, an antifibrinolytic agent, for PPH decreases hemorrhage-related mortality and laparotomy. Routine use of TXA for PPH has demonstrated cost-effectiveness in low-resource countries, where hemorrhage-related mortality rates are higher than in the United States. This study aimed to determine if routine use of TXA for PPH in the United States also is cost-effective.
Details of the study
Sudhof and colleagues conducted a decision-tree analysis to compare the cost-effectiveness of 3 strategies regarding routine use of TXA for PPH in the United States: no TXA, TXA given at any time, and TXA given within 3 hours of delivery.
Health care system perspective. In the primary analysis, the 3 strategies were evaluated from the perspective of the health care system. Outcomes included cost, number of laparotomies, and maternal deaths from delivery until 6 weeks postpartum. Rates of hemorrhage and related complications, as well as cost assumptions, were derived from multiple US-based studies. The relative risk reduction in death and laparotomy with TXA in the United States was assumed to be similar to that found in the WOMAN trial (19% and 36%, respectively).
Societal perspective. In the secondary analysis, the 3 TXA strategies were evaluated from the societal perspective, comparing quality-adjusted life-years (QALYs) and cost per QALY. For both the primary and secondary analyses, sensitivity analyses were performed across a range of values for each input.
Main findings. Tranexamic acid use would be cost saving if the relative risk reduction for maternal death with TXA was greater than approximately 5%, which is significantly lower than that seen in the WOMAN trial (19%). The primary analysis demonstrated that—assuming a 3% rate of PPH—giving TXA to women with PPH would save $11.3 million, prevent 334 laparotomies, and avert 9 maternal deaths annually in the United States. This cost saving nearly tripled if TXA was administered within 3 hours of delivery, with 5 additional maternal deaths prevented.
Secondary analysis incorporating QALYs also showed TXA use to be cost-effective. These findings held through various sensitivity analyses.
Continue to: Study strengths and limitations...
Study strengths and limitations
This study is novel in its critical objective to determine the cost-effectiveness of routine use of TXA for PPH in the United States. Robust modeling using Monte Carlo estimation and a variety of sensitivity analyses add reliability to the authors’ findings.
This work is limited, however, by the assumptions put into the authors’ models. For example, outcome data regarding effectiveness of TXA was taken from the WOMAN trial, which was not performed within the United States. In addition, it is difficult to quantify in dollars an event as profound as a maternal death. The authors recognize that they likely underestimate the “cost” of a maternal death, but that this underestimation would only increase the cost-effectiveness of TXA.
Finally, it is important to take into account that such economic analyses are helpful to inform institutional guidelines and hemorrhage protocols, but that patient-specific decision-making should be individualized based on the clinical scenario at hand.
Routine use of TXA for PPH, particularly within 3 hours of delivery, is likely cost-effective in the United States. Consideration should be given to including TXA in institutional hemorrhage protocols.
REBECCA F. HAMM, MD, and ADI HIRSHBERG, MD
The case for outpatient cervical ripening for IOL at term for low-risk pregnancies
Case 1 Induction at 39 weeks in a healthy nulliparous woman
A healthy 35-year-old woman (G1P0) at 39 weeks 0 days and with an uncomplicated pregnancy presents to your office for a routine prenatal visit. She inquires about scheduling an induction of labor, noting that she read a news story about induction at 39 weeks and that it might lower her chance of having a cesarean delivery (CD).
You perform a cervical exam—she is 1 cm dilated, 3 cm long, -2 station, posterior, and firm. You sweep her membranes after obtaining verbal consent. After describing the induction process, you explain that she might be hospitalized for several days before the birth given the need for cervical ripening. “You mean I need to stay in the hospital for the entire process?” she asks incredulously.
Over the past 20 years, the percentage of patients undergoing induction of labor (IOL) has increased from 10% to 25%.1 This percentage likely will rise over time, particularly in the wake of a recent randomized controlled trial suggesting potential maternal benefits, such as reduced CD rate, for nulliparas induced at 39 weeks compared with expectant management.2 Although there have not been any changes to guidelines for timing of IOL from such professional societies such as the American College of Obstetricians and Gynecologists (ACOG) or the Society for Maternal-Fetal Medicine, key considerations of rising IOL volume include patient experience, labor and delivery (L&D) units’ capacity and resources, and associated health care costs.
An essential part of successful induction involves patience. Induction can be a lengthy process, particularly for nulliparas with unripe cervices. Cervical ripening is a necessary component of successful labor induction, whether achieved mechanically or pharmacologically with synthetic prostaglandins, and it has been shown to lower the chance of CD.3,4 However, achieving a ripe cervix is often the lengthiest part of an induction, and not uncommonly consumes 12 to 24 hours or more of inpatient time. Investigators have sought ways to make this process more expeditious. For example, the FOR-MOMI trial demonstrated that the induction-to-delivery time was several hours shorter when cervical ripening combined mechanical and pharmacologic approaches (Foley balloon plus misoprostol), compared with either method alone, without any increase in maternal or fetal complication rates.5
Better yet, what if admission to the L&D unit for IOL at term could be deferred until the cervix is ripe? A number of hospitals in the United States have successfully introduced outpatient cervical ripening, and several small observational and randomized controlled trials have reported good results in terms of safety, efficacy and time saved, and patient experience. Here, we will make the case that outpatient cervical ripening should be the standard of care for low-risk pregnancies.
Mechanical cervical ripening
Safety
Although data are limited on the safety, the authors of an ACOG Practice Bulletin suggest that, based on the available evidence of mechanical ripening in an inpatient setting, it is also appropriate in the outpatient setting.6 Unlike cervical ripening using prostaglandins, mechanical ripening is not associated with tachysystole, fetal intolerance of labor, or meconium staining.3 A cohort study of nearly 2,000 low-risk patients who underwent Foley catheter placement for cervical ripening using an outpatient protocol but monitored overnight as inpatients and evaluated for adverse outcomes found no CD for fetal distress, vaginal bleeding, placental abruption, or intrapartum stillbirth.7 The authors posited that, given this safety profile in the inpatient setting, that mechanical cervical ripening with a Foley catheter would be appropriate for outpatient use in low-risk populations. Other systematic reviews have been reassuring as well, with exceedingly low complication rates during inpatient mechanical cervical ripening.8 These data advocate for the evaluation of cervical ripening in the outpatient setting.
The evidence for outpatient mechanical ripening, although again limited, also has demonstrated safety. There does not appear to be an increased rate of maternal or neonatal complications, including infectious morbidity, postpartum hemorrhage, CD, operative vaginal delivery, or fetal distress.9-12
Continue to: Efficacy and length-of-stay...
Efficacy and length-of-stay
Efficacy also generally has been shown to be similar when mechanical methods are used in the inpatient and outpatient settings. Small randomized trials of outpatient versus inpatient Foley catheter ripening have shown decreased length of stay (by 10 to 13 hours) and similar or less oxytocin use in the outpatient groups, as well as similar Bishop scores after cervical ripening and no difference in maternal or fetal outcomes.9,11,13,14
One major concern with increasing IOL prevalence is the availability of hospital resources and the associated health care costs, given the known increased length of inpatient stay due to cervical ripening time. Admission to an L&D unit is resource intensive; the costs are similar to admission to an intensive care unit in many hospitals given its level of acuity and high nurse/patient ratio. However, given the safety of outpatient mechanical cervical ripening described above, we argue that routinely admitting low-risk patients for mechanical ripening constitutes a suboptimal use of costly resources.
Indeed, data suggest significant inpatient time savings if cervical ripening can be accomplished prior to admission. A cost-effectiveness analysis in the Netherlands demonstrated a nearly 1,000-euro decrease in cost per induction when Foley catheter induction was done on an outpatient basis.15 Interestingly, a recent trial confined to multiparas found no differences in hospital time when comparing outpatient ripening with Foley balloon alone with inpatient ripening with Foley balloon plus simultaneous oxytocin.10 This certainly merits further study, but it may be that the largest time- and cost-savings are among nulliparas.
Patient preferences
Relatively few studies specifically have addressed patient experiences with outpatient versus inpatient mechanical cervical ripening. Outpatient cervical ripening may provide patients with the benefits of being in the comfort of their own homes with their preferred support persons, increased mobility, more bodily autonomy, and satisfaction with their birthing process.
In a pilot trial involving 48 women, inpatient was compared with outpatient cervical ripening using a Foley balloon. Those in the outpatient group reported getting more rest, feeling less isolated, and having enough privacy. However, participants in both groups were equally satisfied and equally likely to recommend their method of induction to others.11 Another study comparing outpatient versus inpatient Foley balloon cervical ripening found that 85% of patients who underwent outpatient ripening were satisfied with the induction method; however, no query or comparison was done with the inpatient group.12 A trial comparing outpatient mechanical cervical ripening with inpatient misoprostol found that outpatient participants reported several hours more sleep and less pain.16 And in a discrete choice experiment of British gravidas, participants favored the option of outpatient cervical ripening, even if it meant an extra 1.4 trips to the hospital and over an hour of extra travel time.17
While these preliminary findings provide some insight that patients may prefer an outpatient approach to cervical ripening, more studies are needed to fully evaluate patient desires.
Continue to: Our approach to mechanical cervical ripening...
Our approach to mechanical cervical ripening
Most patients undergoing scheduled IOL are reasonable candidates for outpatient cervical ripening based on safety and efficacy. By definition, scheduling in advance implies that the provider has determined that outpatient management is reasonable until that date, and the plan for outpatient ripening need not prolong this period.
FIGURES 1 and 2 show protocols for our 2 hospital centers, which regularly allow for outpatient mechanical cervical ripening. In the process of protocol development, we identified absolute and relative contraindications to determine appropriate candidates. We exclude women who require inpatient management of medical or obstetric conditions (for example, women with severe preeclampsia or any condition requiring continuous fetal monitoring). We also do not routinely recommend outpatient cervical ripening to patients who do not have the necessary social conditions to make this process as safe as possible (including stable housing, reliable transportation, and a support person), although this occurs with some exceptions depending on individual patient situations.
Some examples of ideal candidates for outpatient mechanical cervical ripening include those undergoing elective or routine prolonged gestation inductions, or inductions for well-controlled, stable conditions (chronic hypertension and gestational diabetes). At one center, after thorough counseling and assessment, outpatient cervical ripening is also offered to patients with mild risk factors, including twins, prior low transverse CD, stable preeclampsia without severe features, isolated oligohydramnios with otherwise reassuring fetal status, and other similar conditions.
After mechanical cervical ripening placement (either Foley catheter or mechanical dilators), the clinician completes a postprocedure safety checklist and detailed procedure documentation, including number and type of foreign bodies placed. If there are any concerns regarding maternal or fetal well-being, the patient is sent to L&D for evaluation. If the procedure was tolerated well, the patient is discharged home, after a reactive postprocedure nonstress test is done, with detailed instructions for self-care, as well as with a list of symptoms that warrant prompt evaluation prior to scheduled induction time. In a large California hospital group following a similar protocol, only about 5% of women presented in labor before their scheduled induction.18

Case 2 Cervical ripening for labor preparation in low-risk pregnancy
A 32-year-old woman (G1P0) with an uncomplicated pregnancy at 40 weeks and 3 days presents to your office for a routine prenatal visit. Her vital signs are normal, and her fetus is vertex with an estimated fetal weight of 7.5 lb by Leopald’s maneuvers. You perform a cervical exam and find that her cervix is closed, long, and posterior.
You discuss with her your recommendation for induction of labor by 41 weeks, and she agrees. You also discuss the need for cervical ripening and recommend misoprostol given her closed cervix. You explain that several doses may be needed to get her cervix ready for labor, and she asks, “Do I have to stay in the hospital that whole time?”
Pharmacologic cervical ripening
Efficacy
There are multiple pharmacologic agents that can be used for ripening an unfavorable cervix. The main agents used in the United States are prostaglandins, either PGE1 (oral or vaginal misoprostol) or PGE2 in a gel or sustained-release vaginal insert (dinoprostone).
Outpatient misoprostol to avoid labor induction. Many studies have looked at outpatient misoprostol use as a “prophylactic measure” (to prevent the need for labor induction). For example, Gaffaney and colleagues showed that administering outpatient oral misoprostol (100 µg every 24 hours for up to 3 doses) after 40 weeks’ gestation to women with an unfavorable cervix significantly decreased the time to delivery by a day and a half.19 Similarly, PonMalar and colleagues demonstrated that administering 25 µg of vaginal misoprostol in a single dose as an outpatient after stripping the membranes significantly reduced time to delivery by 2 days.20 And Stitely and colleagues found a significant reduction in the need for labor induction with the use of outpatient vaginal misoprostol. They administered up to 2 doses of misoprostol 25 µg vaginally every 24 hours for the 48 hours prior to a scheduled postdates induction and found a large reduction in the need for labor induction (11% vs 85%; P<.01).21
Continue to: Multiple protocols and regimens...
Multiple protocols and regimens have been studied but, overall, the findings suggest that administering outpatient misoprostol may shorten the time interval to spontaneous labor and decrease the need for a formal labor induction.19-23
Inpatient compared with outpatient prostaglandin use. These trials of “prophylactic” misoprostol generally have compared outpatient administration of misoprostol with placebo. Prostaglandins are one of the most common methods of inpatient cervical ripening, so what about comparisons of inpatient cervical ripening with outpatient prostaglandin administration? There are a handful of studies that make this comparison.
Chang and colleagues looked retrospectively at inpatient and outpatient misoprostol and found that outpatient administration saved 3 to 5 hours on labor and delivery.24 Biem and colleagues randomly assigned women to either inpatient cervical ripening with PGE2 intravaginal inserts or 1 hour of inpatient monitoring after PGE2 administration and then outpatient discharge until the onset of labor or for a nonstress test at 12 hours. They found that those who underwent outpatient ripening spent 8 hours less on labor and delivery and were more highly satisfied with the initial 12 hours of labor induction experience (56% vs 39%; P<.01).25
The largest randomized controlled trial conducted to study outpatient prostaglandin use was the OPRA study (involving 827 women). Investigators compared inpatient to outpatient PGE2 intravaginal gel.26 The primary outcome was total oxytocin administration, which was not different between groups. The study was underpowered, however, as 50% of women labored spontaneously postrandomization. But in the outpatient arm, less than half of the women required additional inpatient ripening, and nearly 40% returned in spontaneous labor, suggesting that outpatient prostaglandin administration may indeed save women a significant amount of time on labor and delivery.
Safety
The safety of outpatient administration of prostaglandins is the biggest concern, especially since, when prostaglandins are compared to outpatient Foley catheter use, Foleys are overall associated with less tachysystole, fetal intolerance, and meconium-stained fluid.3 Foley catheter use for cervical ripening may not be an appropriate choice for all patients, however. For instance, our case patient has a closed cervix, which could make Foley insertion uncomfortable or even impossible. Misoprostol use also offers the potential for flexibility in cervical ripening protocols as patients need not return for Foley balloon removal and indeed labor induction need not take place immediately after administration of misoprostol.
Patients also may prefer outpatient cervical ripening with misoprostol over a Foley. There are some data to suggest that women, overall, have a preference toward prostaglandins; in the PROBAAT-II trial, which compared inpatient oral misoprostol to Foley catheter for cervical ripening, 12% of women in the Foley arm would have preferred another method of induction (vs 6% in the misoprostol arm; P = .02).27 This preference may be magnified in an outpatient setting.
But, again, is outpatient administration of prostaglandins safe? The published trials thus far have not reported an increase in out-of-hospital deliveries or adverse fetal outcomes. However, studies have been of limited size to see more rare outcomes. Unfortunately, an adequately powered study to demonstrate safety is likely never to be accomplished, given that if used responsibly (in low-risk patients with adequate monitoring after administration) the incidence of adverse fetal outcomes during the at-home portion of cervical ripening is likely to be very low. With responsible use, outpatient administration of prostaglandins should be safe. Women are monitored after misoprostol administration and are not sent home if there are any concerns for fetal distress or if frequent contractions continue. Misoprostol reaches maximum blood concentration 30 minutes after oral administration and 70 to 80 minutes after vaginal administration.28 After this time, if contractions start to intensify it is likely that misoprostol has triggered spontaneous labor. In this setting, women are routinely allowed to spontaneously labor at home. One may even argue that outpatient misoprostol could lead to improved safety, as women essentially have a contraction stress test prior to spontaneous labor, and misoprostol administration as an outpatient, as opposed to as an inpatient, may allow for longer time intervals between doses, which could prevent dose stacking.
Continue to: Our approach to pharmacologic cervical ripening...
Our approach to pharmacologic cervical ripening
Our hospital has been conducting outpatient cervical ripening using vaginal misoprostol for more th
Conclusion
While the data continue to be limited, we strongly believe there is sufficient quality evidence from a safety and efficacy perspective to support implementation and evaluation of outpatient cervical ripening protocols for low-risk pregnancies. In the setting of renewed commitments to reducing suboptimal health care costs and utilization as well as increasing patient satisfaction and control in their birthing experiences, we posit it is the responsibility of obstetricians, L&D leadership, and health care institutions to explore the implementation of outpatient cervical ripening for appropriate candidates in their settings.
- Martin JA, Hamilton BE, Osterman MJ, et al. Births: final data for 2015. Natl Vital Stat Rep. 2017;66:1.
- Grobman WA, Rice MM, Reddy UM, et al. Labor induction versus expectant management in low-risk nulliparous women. N Engl J Med. 2018;379:513-523.
- Jozwiak M, Bloemenkamp KW, Kelly AJ, et al. Mechanical methods for induction of labor. Cochrane Database Syst Rev. 2012;(3):CD001233.
- Alfirevic Z, Kelly AJ, Dowswell T. Intravenous oxytocin alone for cervical ripening and induction of labour. Cochrane Database Syst Rev. 2009;(4):CD003246.
- Levine LD, Downes KL, Elovitz MA, et al. Mechanical and pharmacologic methods of labor induction: a randomized controlled trial. Obstet Gynecol. 2016;128:1357-1364.
- ACOG Committee on Practice Bulletins—Obstetrics. ACOG practice bulletin no. 107: induction of labor. Obstet Gynecol. 2009;114(2 pt 1):386-397. Reaffirmed 2019.
- Sciscione AC, Bedder CL, Hoffman MK, et al. The timing of adverse events with Foley catheter preinduction cervical ripening; implications for outpatient use. Am J Perinatol. 2014;31:781-786.
- Diederen M, Gommers J, Wilkinson C, et al. Safety of the balloon catheter for cervical ripening in outpatient care: complications during the period from insertion to expulsion of a balloon catheter in the process of labour induction: a systematic review. BJOG. 2018;125:1086-1095.
- McKenna DS, Duke JM. Effectiveness and infectious morbidity of outpatient cervical ripening with a Foley catheter. J Reprod Med. 2004;49:28-32.
- Kuper SG, Jauk VC, George DM, et al. Outpatient Foley catheter for induction of labor in parous women: a randomized controlled trial. Obstet Gynecol. 2018;132:94-101.
- Wilkinson C, Adelson P, Turnbull D. A comparison of inpatient with outpatient balloon catheter cervical ripening: a pilot randomized controlled trial. BMC Pregnancy Childbirth. 2015;15:126.
- Kruit H, Heikinheimo O, Ulander VM, et al. Foley catheter induction of labor as an outpatient procedure. J Perinatol. 2016;36:618-622.
- Sciscione AC, Muench M, Pollock M, et al. Transcervical Foley catheter for preinduction cervical ripening in an outpatient versus inpatient setting. Obstet Gynecol. 2001;98(5 pt 1):751-756.
- Policiano C, Pimenta M, Martins D, et al. Outpatient versus inpatient cervix priming with Foley catheter: a randomized trial. Eur J Obstet Gynecol Reprod Biol. 2017;210:1-6.
- Ten Eikelder M, van Baaren GJ, Oude Rengerink K, et al. Comparing induction of labour with oral misoprostol or Foley catheter at term: cost effectiveness analysis of a randomised controlled multi-centre non-inferiority trial. BJOG. 2018;125:375-383.
- Henry A, Madan A, Reid R, et al. Outpatient Foley catheter versus inpatient prostaglandin E2 gel for induction of labour: a randomised trial. BMC Pregnancy Childbirth. 2013;13:25.
- Howard K, Gerard K, Adelson P, et al. Women’s preferences for inpatient and outpatient priming for labour induction: a discrete choice experiment. BMC Health Serv Res. 2014;14:330.
- Main E, LaGrew D; California Maternal Quality Care Collaborative. Induction of labor risks, benefits, and techniques for increasing success. June 14, 2017. https://www .cmqcc.org/resource/induction-labor-risk-benefits-and-techniques-increasing -success. Accessed August 21, 2019.
- Gaffaney CA, Saul LL, Rumney PJ, et al. Outpatient oral misoprostol for prolonged pregnancies: a pilot investigation. Am J Perinatol. 2009;26:673-677.
- PonMalar J, Benjamin SJ, Abraham A, et al. Randomized double-blind placebo controlled study of preinduction cervical priming with 25 µg of misoprostol in the outpatient setting to prevent formal induction of labour. Arch Gynecol Obstet. 2017;295:33-38.
- Stitely ML, Browning J, Fowler M, et al. Outpatient cervical ripening with intravaginal misoprostol. Obstet Gynecol. 2000;96(5 pt 1):684-688.
- McKenna DS, Ester JB, Proffitt M, et al. Misoprostol outpatient cervical ripening without subsequent induction of labor: a randomized trial. Obstet Gynecol. 2004;104:579-584.
- Oboro VO, Tabowei TO. Outpatient misoprostol cervical ripening withoutsubsequent induction of labor to prevent post-term pregnancy. Acta Obstet Gynecol Scand. 2005;84:628-631.
- Chang DW, Velazquez MD, Colyer M, et al. Vaginal misoprostol for cervical ripening at term: comparison of outpatient vs. inpatient administration. J Reprod Med. 2005;50:735-739.
- Biem SR, Turnell RW, Olatunbosun O, et al. A randomized controlled trial of outpatient versus inpatient labour induction with vaginal controlled-release prostaglandin-E2: effectiveness and satisfaction. J Obstet Gynaecol Can. 2003;25:23-31.
- Wilkinson C, Bryce R, Adelson P, et al. A randomised controlled trial of outpatient compared with inpatient cervical ripening with prostaglandin E₂ (OPRA study). BJOG. 2015;122:94-104.
- Ten Eikelder ML, van de Meent MM, Mast K, et al. Women’s experiences with and preference for induction of labor with oral misoprostol or Foley catheter at term. Am J Perinatol. 2017;34:138-146.
- Tang OS, Gemzell-Danielsson K, Ho PC. Misoprostol: pharmacokinetic profiles, effects on the uterus and side-effects. Int J Gynaecol Obstet. 2007;99 (suppl 2):S160-S167.
Case 1 Induction at 39 weeks in a healthy nulliparous woman
A healthy 35-year-old woman (G1P0) at 39 weeks 0 days and with an uncomplicated pregnancy presents to your office for a routine prenatal visit. She inquires about scheduling an induction of labor, noting that she read a news story about induction at 39 weeks and that it might lower her chance of having a cesarean delivery (CD).
You perform a cervical exam—she is 1 cm dilated, 3 cm long, -2 station, posterior, and firm. You sweep her membranes after obtaining verbal consent. After describing the induction process, you explain that she might be hospitalized for several days before the birth given the need for cervical ripening. “You mean I need to stay in the hospital for the entire process?” she asks incredulously.
Over the past 20 years, the percentage of patients undergoing induction of labor (IOL) has increased from 10% to 25%.1 This percentage likely will rise over time, particularly in the wake of a recent randomized controlled trial suggesting potential maternal benefits, such as reduced CD rate, for nulliparas induced at 39 weeks compared with expectant management.2 Although there have not been any changes to guidelines for timing of IOL from such professional societies such as the American College of Obstetricians and Gynecologists (ACOG) or the Society for Maternal-Fetal Medicine, key considerations of rising IOL volume include patient experience, labor and delivery (L&D) units’ capacity and resources, and associated health care costs.
An essential part of successful induction involves patience. Induction can be a lengthy process, particularly for nulliparas with unripe cervices. Cervical ripening is a necessary component of successful labor induction, whether achieved mechanically or pharmacologically with synthetic prostaglandins, and it has been shown to lower the chance of CD.3,4 However, achieving a ripe cervix is often the lengthiest part of an induction, and not uncommonly consumes 12 to 24 hours or more of inpatient time. Investigators have sought ways to make this process more expeditious. For example, the FOR-MOMI trial demonstrated that the induction-to-delivery time was several hours shorter when cervical ripening combined mechanical and pharmacologic approaches (Foley balloon plus misoprostol), compared with either method alone, without any increase in maternal or fetal complication rates.5
Better yet, what if admission to the L&D unit for IOL at term could be deferred until the cervix is ripe? A number of hospitals in the United States have successfully introduced outpatient cervical ripening, and several small observational and randomized controlled trials have reported good results in terms of safety, efficacy and time saved, and patient experience. Here, we will make the case that outpatient cervical ripening should be the standard of care for low-risk pregnancies.

Mechanical cervical ripening
Safety
Although data are limited on the safety, the authors of an ACOG Practice Bulletin suggest that, based on the available evidence of mechanical ripening in an inpatient setting, it is also appropriate in the outpatient setting.6 Unlike cervical ripening using prostaglandins, mechanical ripening is not associated with tachysystole, fetal intolerance of labor, or meconium staining.3 A cohort study of nearly 2,000 low-risk patients who underwent Foley catheter placement for cervical ripening using an outpatient protocol but monitored overnight as inpatients and evaluated for adverse outcomes found no CD for fetal distress, vaginal bleeding, placental abruption, or intrapartum stillbirth.7 The authors posited that, given this safety profile in the inpatient setting, that mechanical cervical ripening with a Foley catheter would be appropriate for outpatient use in low-risk populations. Other systematic reviews have been reassuring as well, with exceedingly low complication rates during inpatient mechanical cervical ripening.8 These data advocate for the evaluation of cervical ripening in the outpatient setting.
The evidence for outpatient mechanical ripening, although again limited, also has demonstrated safety. There does not appear to be an increased rate of maternal or neonatal complications, including infectious morbidity, postpartum hemorrhage, CD, operative vaginal delivery, or fetal distress.9-12
Continue to: Efficacy and length-of-stay...
Efficacy and length-of-stay
Efficacy also generally has been shown to be similar when mechanical methods are used in the inpatient and outpatient settings. Small randomized trials of outpatient versus inpatient Foley catheter ripening have shown decreased length of stay (by 10 to 13 hours) and similar or less oxytocin use in the outpatient groups, as well as similar Bishop scores after cervical ripening and no difference in maternal or fetal outcomes.9,11,13,14
One major concern with increasing IOL prevalence is the availability of hospital resources and the associated health care costs, given the known increased length of inpatient stay due to cervical ripening time. Admission to an L&D unit is resource intensive; the costs are similar to admission to an intensive care unit in many hospitals given its level of acuity and high nurse/patient ratio. However, given the safety of outpatient mechanical cervical ripening described above, we argue that routinely admitting low-risk patients for mechanical ripening constitutes a suboptimal use of costly resources.
Indeed, data suggest significant inpatient time savings if cervical ripening can be accomplished prior to admission. A cost-effectiveness analysis in the Netherlands demonstrated a nearly 1,000-euro decrease in cost per induction when Foley catheter induction was done on an outpatient basis.15 Interestingly, a recent trial confined to multiparas found no differences in hospital time when comparing outpatient ripening with Foley balloon alone with inpatient ripening with Foley balloon plus simultaneous oxytocin.10 This certainly merits further study, but it may be that the largest time- and cost-savings are among nulliparas.
Patient preferences
Relatively few studies specifically have addressed patient experiences with outpatient versus inpatient mechanical cervical ripening. Outpatient cervical ripening may provide patients with the benefits of being in the comfort of their own homes with their preferred support persons, increased mobility, more bodily autonomy, and satisfaction with their birthing process.
In a pilot trial involving 48 women, inpatient was compared with outpatient cervical ripening using a Foley balloon. Those in the outpatient group reported getting more rest, feeling less isolated, and having enough privacy. However, participants in both groups were equally satisfied and equally likely to recommend their method of induction to others.11 Another study comparing outpatient versus inpatient Foley balloon cervical ripening found that 85% of patients who underwent outpatient ripening were satisfied with the induction method; however, no query or comparison was done with the inpatient group.12 A trial comparing outpatient mechanical cervical ripening with inpatient misoprostol found that outpatient participants reported several hours more sleep and less pain.16 And in a discrete choice experiment of British gravidas, participants favored the option of outpatient cervical ripening, even if it meant an extra 1.4 trips to the hospital and over an hour of extra travel time.17
While these preliminary findings provide some insight that patients may prefer an outpatient approach to cervical ripening, more studies are needed to fully evaluate patient desires.
Continue to: Our approach to mechanical cervical ripening...
Our approach to mechanical cervical ripening
Most patients undergoing scheduled IOL are reasonable candidates for outpatient cervical ripening based on safety and efficacy. By definition, scheduling in advance implies that the provider has determined that outpatient management is reasonable until that date, and the plan for outpatient ripening need not prolong this period.
FIGURES 1 and 2 show protocols for our 2 hospital centers, which regularly allow for outpatient mechanical cervical ripening. In the process of protocol development, we identified absolute and relative contraindications to determine appropriate candidates. We exclude women who require inpatient management of medical or obstetric conditions (for example, women with severe preeclampsia or any condition requiring continuous fetal monitoring). We also do not routinely recommend outpatient cervical ripening to patients who do not have the necessary social conditions to make this process as safe as possible (including stable housing, reliable transportation, and a support person), although this occurs with some exceptions depending on individual patient situations.
Some examples of ideal candidates for outpatient mechanical cervical ripening include those undergoing elective or routine prolonged gestation inductions, or inductions for well-controlled, stable conditions (chronic hypertension and gestational diabetes). At one center, after thorough counseling and assessment, outpatient cervical ripening is also offered to patients with mild risk factors, including twins, prior low transverse CD, stable preeclampsia without severe features, isolated oligohydramnios with otherwise reassuring fetal status, and other similar conditions.
After mechanical cervical ripening placement (either Foley catheter or mechanical dilators), the clinician completes a postprocedure safety checklist and detailed procedure documentation, including number and type of foreign bodies placed. If there are any concerns regarding maternal or fetal well-being, the patient is sent to L&D for evaluation. If the procedure was tolerated well, the patient is discharged home, after a reactive postprocedure nonstress test is done, with detailed instructions for self-care, as well as with a list of symptoms that warrant prompt evaluation prior to scheduled induction time. In a large California hospital group following a similar protocol, only about 5% of women presented in labor before their scheduled induction.18


Case 2 Cervical ripening for labor preparation in low-risk pregnancy
A 32-year-old woman (G1P0) with an uncomplicated pregnancy at 40 weeks and 3 days presents to your office for a routine prenatal visit. Her vital signs are normal, and her fetus is vertex with an estimated fetal weight of 7.5 lb by Leopald’s maneuvers. You perform a cervical exam and find that her cervix is closed, long, and posterior.
You discuss with her your recommendation for induction of labor by 41 weeks, and she agrees. You also discuss the need for cervical ripening and recommend misoprostol given her closed cervix. You explain that several doses may be needed to get her cervix ready for labor, and she asks, “Do I have to stay in the hospital that whole time?”
Pharmacologic cervical ripening
Efficacy
There are multiple pharmacologic agents that can be used for ripening an unfavorable cervix. The main agents used in the United States are prostaglandins, either PGE1 (oral or vaginal misoprostol) or PGE2 in a gel or sustained-release vaginal insert (dinoprostone).
Outpatient misoprostol to avoid labor induction. Many studies have looked at outpatient misoprostol use as a “prophylactic measure” (to prevent the need for labor induction). For example, Gaffaney and colleagues showed that administering outpatient oral misoprostol (100 µg every 24 hours for up to 3 doses) after 40 weeks’ gestation to women with an unfavorable cervix significantly decreased the time to delivery by a day and a half.19 Similarly, PonMalar and colleagues demonstrated that administering 25 µg of vaginal misoprostol in a single dose as an outpatient after stripping the membranes significantly reduced time to delivery by 2 days.20 And Stitely and colleagues found a significant reduction in the need for labor induction with the use of outpatient vaginal misoprostol. They administered up to 2 doses of misoprostol 25 µg vaginally every 24 hours for the 48 hours prior to a scheduled postdates induction and found a large reduction in the need for labor induction (11% vs 85%; P<.01).21
Continue to: Multiple protocols and regimens...
Multiple protocols and regimens have been studied but, overall, the findings suggest that administering outpatient misoprostol may shorten the time interval to spontaneous labor and decrease the need for a formal labor induction.19-23
Inpatient compared with outpatient prostaglandin use. These trials of “prophylactic” misoprostol generally have compared outpatient administration of misoprostol with placebo. Prostaglandins are one of the most common methods of inpatient cervical ripening, so what about comparisons of inpatient cervical ripening with outpatient prostaglandin administration? There are a handful of studies that make this comparison.
Chang and colleagues looked retrospectively at inpatient and outpatient misoprostol and found that outpatient administration saved 3 to 5 hours on labor and delivery.24 Biem and colleagues randomly assigned women to either inpatient cervical ripening with PGE2 intravaginal inserts or 1 hour of inpatient monitoring after PGE2 administration and then outpatient discharge until the onset of labor or for a nonstress test at 12 hours. They found that those who underwent outpatient ripening spent 8 hours less on labor and delivery and were more highly satisfied with the initial 12 hours of labor induction experience (56% vs 39%; P<.01).25
The largest randomized controlled trial conducted to study outpatient prostaglandin use was the OPRA study (involving 827 women). Investigators compared inpatient to outpatient PGE2 intravaginal gel.26 The primary outcome was total oxytocin administration, which was not different between groups. The study was underpowered, however, as 50% of women labored spontaneously postrandomization. But in the outpatient arm, less than half of the women required additional inpatient ripening, and nearly 40% returned in spontaneous labor, suggesting that outpatient prostaglandin administration may indeed save women a significant amount of time on labor and delivery.
Safety
The safety of outpatient administration of prostaglandins is the biggest concern, especially since, when prostaglandins are compared to outpatient Foley catheter use, Foleys are overall associated with less tachysystole, fetal intolerance, and meconium-stained fluid.3 Foley catheter use for cervical ripening may not be an appropriate choice for all patients, however. For instance, our case patient has a closed cervix, which could make Foley insertion uncomfortable or even impossible. Misoprostol use also offers the potential for flexibility in cervical ripening protocols as patients need not return for Foley balloon removal and indeed labor induction need not take place immediately after administration of misoprostol.
Patients also may prefer outpatient cervical ripening with misoprostol over a Foley. There are some data to suggest that women, overall, have a preference toward prostaglandins; in the PROBAAT-II trial, which compared inpatient oral misoprostol to Foley catheter for cervical ripening, 12% of women in the Foley arm would have preferred another method of induction (vs 6% in the misoprostol arm; P = .02).27 This preference may be magnified in an outpatient setting.
But, again, is outpatient administration of prostaglandins safe? The published trials thus far have not reported an increase in out-of-hospital deliveries or adverse fetal outcomes. However, studies have been of limited size to see more rare outcomes. Unfortunately, an adequately powered study to demonstrate safety is likely never to be accomplished, given that if used responsibly (in low-risk patients with adequate monitoring after administration) the incidence of adverse fetal outcomes during the at-home portion of cervical ripening is likely to be very low. With responsible use, outpatient administration of prostaglandins should be safe. Women are monitored after misoprostol administration and are not sent home if there are any concerns for fetal distress or if frequent contractions continue. Misoprostol reaches maximum blood concentration 30 minutes after oral administration and 70 to 80 minutes after vaginal administration.28 After this time, if contractions start to intensify it is likely that misoprostol has triggered spontaneous labor. In this setting, women are routinely allowed to spontaneously labor at home. One may even argue that outpatient misoprostol could lead to improved safety, as women essentially have a contraction stress test prior to spontaneous labor, and misoprostol administration as an outpatient, as opposed to as an inpatient, may allow for longer time intervals between doses, which could prevent dose stacking.
Continue to: Our approach to pharmacologic cervical ripening...
Our approach to pharmacologic cervical ripening
Our hospital has been conducting outpatient cervical ripening using vaginal misoprostol for more th

Conclusion
While the data continue to be limited, we strongly believe there is sufficient quality evidence from a safety and efficacy perspective to support implementation and evaluation of outpatient cervical ripening protocols for low-risk pregnancies. In the setting of renewed commitments to reducing suboptimal health care costs and utilization as well as increasing patient satisfaction and control in their birthing experiences, we posit it is the responsibility of obstetricians, L&D leadership, and health care institutions to explore the implementation of outpatient cervical ripening for appropriate candidates in their settings.
Case 1 Induction at 39 weeks in a healthy nulliparous woman
A healthy 35-year-old woman (G1P0) at 39 weeks 0 days and with an uncomplicated pregnancy presents to your office for a routine prenatal visit. She inquires about scheduling an induction of labor, noting that she read a news story about induction at 39 weeks and that it might lower her chance of having a cesarean delivery (CD).
You perform a cervical exam—she is 1 cm dilated, 3 cm long, -2 station, posterior, and firm. You sweep her membranes after obtaining verbal consent. After describing the induction process, you explain that she might be hospitalized for several days before the birth given the need for cervical ripening. “You mean I need to stay in the hospital for the entire process?” she asks incredulously.
Over the past 20 years, the percentage of patients undergoing induction of labor (IOL) has increased from 10% to 25%.1 This percentage likely will rise over time, particularly in the wake of a recent randomized controlled trial suggesting potential maternal benefits, such as reduced CD rate, for nulliparas induced at 39 weeks compared with expectant management.2 Although there have not been any changes to guidelines for timing of IOL from such professional societies such as the American College of Obstetricians and Gynecologists (ACOG) or the Society for Maternal-Fetal Medicine, key considerations of rising IOL volume include patient experience, labor and delivery (L&D) units’ capacity and resources, and associated health care costs.
An essential part of successful induction involves patience. Induction can be a lengthy process, particularly for nulliparas with unripe cervices. Cervical ripening is a necessary component of successful labor induction, whether achieved mechanically or pharmacologically with synthetic prostaglandins, and it has been shown to lower the chance of CD.3,4 However, achieving a ripe cervix is often the lengthiest part of an induction, and not uncommonly consumes 12 to 24 hours or more of inpatient time. Investigators have sought ways to make this process more expeditious. For example, the FOR-MOMI trial demonstrated that the induction-to-delivery time was several hours shorter when cervical ripening combined mechanical and pharmacologic approaches (Foley balloon plus misoprostol), compared with either method alone, without any increase in maternal or fetal complication rates.5
Better yet, what if admission to the L&D unit for IOL at term could be deferred until the cervix is ripe? A number of hospitals in the United States have successfully introduced outpatient cervical ripening, and several small observational and randomized controlled trials have reported good results in terms of safety, efficacy and time saved, and patient experience. Here, we will make the case that outpatient cervical ripening should be the standard of care for low-risk pregnancies.

Mechanical cervical ripening
Safety
Although data are limited on the safety, the authors of an ACOG Practice Bulletin suggest that, based on the available evidence of mechanical ripening in an inpatient setting, it is also appropriate in the outpatient setting.6 Unlike cervical ripening using prostaglandins, mechanical ripening is not associated with tachysystole, fetal intolerance of labor, or meconium staining.3 A cohort study of nearly 2,000 low-risk patients who underwent Foley catheter placement for cervical ripening using an outpatient protocol but monitored overnight as inpatients and evaluated for adverse outcomes found no CD for fetal distress, vaginal bleeding, placental abruption, or intrapartum stillbirth.7 The authors posited that, given this safety profile in the inpatient setting, that mechanical cervical ripening with a Foley catheter would be appropriate for outpatient use in low-risk populations. Other systematic reviews have been reassuring as well, with exceedingly low complication rates during inpatient mechanical cervical ripening.8 These data advocate for the evaluation of cervical ripening in the outpatient setting.
The evidence for outpatient mechanical ripening, although again limited, also has demonstrated safety. There does not appear to be an increased rate of maternal or neonatal complications, including infectious morbidity, postpartum hemorrhage, CD, operative vaginal delivery, or fetal distress.9-12
Continue to: Efficacy and length-of-stay...
Efficacy and length-of-stay
Efficacy also generally has been shown to be similar when mechanical methods are used in the inpatient and outpatient settings. Small randomized trials of outpatient versus inpatient Foley catheter ripening have shown decreased length of stay (by 10 to 13 hours) and similar or less oxytocin use in the outpatient groups, as well as similar Bishop scores after cervical ripening and no difference in maternal or fetal outcomes.9,11,13,14
One major concern with increasing IOL prevalence is the availability of hospital resources and the associated health care costs, given the known increased length of inpatient stay due to cervical ripening time. Admission to an L&D unit is resource intensive; the costs are similar to admission to an intensive care unit in many hospitals given its level of acuity and high nurse/patient ratio. However, given the safety of outpatient mechanical cervical ripening described above, we argue that routinely admitting low-risk patients for mechanical ripening constitutes a suboptimal use of costly resources.
Indeed, data suggest significant inpatient time savings if cervical ripening can be accomplished prior to admission. A cost-effectiveness analysis in the Netherlands demonstrated a nearly 1,000-euro decrease in cost per induction when Foley catheter induction was done on an outpatient basis.15 Interestingly, a recent trial confined to multiparas found no differences in hospital time when comparing outpatient ripening with Foley balloon alone with inpatient ripening with Foley balloon plus simultaneous oxytocin.10 This certainly merits further study, but it may be that the largest time- and cost-savings are among nulliparas.
Patient preferences
Relatively few studies specifically have addressed patient experiences with outpatient versus inpatient mechanical cervical ripening. Outpatient cervical ripening may provide patients with the benefits of being in the comfort of their own homes with their preferred support persons, increased mobility, more bodily autonomy, and satisfaction with their birthing process.
In a pilot trial involving 48 women, inpatient was compared with outpatient cervical ripening using a Foley balloon. Those in the outpatient group reported getting more rest, feeling less isolated, and having enough privacy. However, participants in both groups were equally satisfied and equally likely to recommend their method of induction to others.11 Another study comparing outpatient versus inpatient Foley balloon cervical ripening found that 85% of patients who underwent outpatient ripening were satisfied with the induction method; however, no query or comparison was done with the inpatient group.12 A trial comparing outpatient mechanical cervical ripening with inpatient misoprostol found that outpatient participants reported several hours more sleep and less pain.16 And in a discrete choice experiment of British gravidas, participants favored the option of outpatient cervical ripening, even if it meant an extra 1.4 trips to the hospital and over an hour of extra travel time.17
While these preliminary findings provide some insight that patients may prefer an outpatient approach to cervical ripening, more studies are needed to fully evaluate patient desires.
Continue to: Our approach to mechanical cervical ripening...
Our approach to mechanical cervical ripening
Most patients undergoing scheduled IOL are reasonable candidates for outpatient cervical ripening based on safety and efficacy. By definition, scheduling in advance implies that the provider has determined that outpatient management is reasonable until that date, and the plan for outpatient ripening need not prolong this period.
FIGURES 1 and 2 show protocols for our 2 hospital centers, which regularly allow for outpatient mechanical cervical ripening. In the process of protocol development, we identified absolute and relative contraindications to determine appropriate candidates. We exclude women who require inpatient management of medical or obstetric conditions (for example, women with severe preeclampsia or any condition requiring continuous fetal monitoring). We also do not routinely recommend outpatient cervical ripening to patients who do not have the necessary social conditions to make this process as safe as possible (including stable housing, reliable transportation, and a support person), although this occurs with some exceptions depending on individual patient situations.
Some examples of ideal candidates for outpatient mechanical cervical ripening include those undergoing elective or routine prolonged gestation inductions, or inductions for well-controlled, stable conditions (chronic hypertension and gestational diabetes). At one center, after thorough counseling and assessment, outpatient cervical ripening is also offered to patients with mild risk factors, including twins, prior low transverse CD, stable preeclampsia without severe features, isolated oligohydramnios with otherwise reassuring fetal status, and other similar conditions.
After mechanical cervical ripening placement (either Foley catheter or mechanical dilators), the clinician completes a postprocedure safety checklist and detailed procedure documentation, including number and type of foreign bodies placed. If there are any concerns regarding maternal or fetal well-being, the patient is sent to L&D for evaluation. If the procedure was tolerated well, the patient is discharged home, after a reactive postprocedure nonstress test is done, with detailed instructions for self-care, as well as with a list of symptoms that warrant prompt evaluation prior to scheduled induction time. In a large California hospital group following a similar protocol, only about 5% of women presented in labor before their scheduled induction.18


Case 2 Cervical ripening for labor preparation in low-risk pregnancy
A 32-year-old woman (G1P0) with an uncomplicated pregnancy at 40 weeks and 3 days presents to your office for a routine prenatal visit. Her vital signs are normal, and her fetus is vertex with an estimated fetal weight of 7.5 lb by Leopald’s maneuvers. You perform a cervical exam and find that her cervix is closed, long, and posterior.
You discuss with her your recommendation for induction of labor by 41 weeks, and she agrees. You also discuss the need for cervical ripening and recommend misoprostol given her closed cervix. You explain that several doses may be needed to get her cervix ready for labor, and she asks, “Do I have to stay in the hospital that whole time?”
Pharmacologic cervical ripening
Efficacy
There are multiple pharmacologic agents that can be used for ripening an unfavorable cervix. The main agents used in the United States are prostaglandins, either PGE1 (oral or vaginal misoprostol) or PGE2 in a gel or sustained-release vaginal insert (dinoprostone).
Outpatient misoprostol to avoid labor induction. Many studies have looked at outpatient misoprostol use as a “prophylactic measure” (to prevent the need for labor induction). For example, Gaffaney and colleagues showed that administering outpatient oral misoprostol (100 µg every 24 hours for up to 3 doses) after 40 weeks’ gestation to women with an unfavorable cervix significantly decreased the time to delivery by a day and a half.19 Similarly, PonMalar and colleagues demonstrated that administering 25 µg of vaginal misoprostol in a single dose as an outpatient after stripping the membranes significantly reduced time to delivery by 2 days.20 And Stitely and colleagues found a significant reduction in the need for labor induction with the use of outpatient vaginal misoprostol. They administered up to 2 doses of misoprostol 25 µg vaginally every 24 hours for the 48 hours prior to a scheduled postdates induction and found a large reduction in the need for labor induction (11% vs 85%; P<.01).21
Continue to: Multiple protocols and regimens...
Multiple protocols and regimens have been studied but, overall, the findings suggest that administering outpatient misoprostol may shorten the time interval to spontaneous labor and decrease the need for a formal labor induction.19-23
Inpatient compared with outpatient prostaglandin use. These trials of “prophylactic” misoprostol generally have compared outpatient administration of misoprostol with placebo. Prostaglandins are one of the most common methods of inpatient cervical ripening, so what about comparisons of inpatient cervical ripening with outpatient prostaglandin administration? There are a handful of studies that make this comparison.
Chang and colleagues looked retrospectively at inpatient and outpatient misoprostol and found that outpatient administration saved 3 to 5 hours on labor and delivery.24 Biem and colleagues randomly assigned women to either inpatient cervical ripening with PGE2 intravaginal inserts or 1 hour of inpatient monitoring after PGE2 administration and then outpatient discharge until the onset of labor or for a nonstress test at 12 hours. They found that those who underwent outpatient ripening spent 8 hours less on labor and delivery and were more highly satisfied with the initial 12 hours of labor induction experience (56% vs 39%; P<.01).25
The largest randomized controlled trial conducted to study outpatient prostaglandin use was the OPRA study (involving 827 women). Investigators compared inpatient to outpatient PGE2 intravaginal gel.26 The primary outcome was total oxytocin administration, which was not different between groups. The study was underpowered, however, as 50% of women labored spontaneously postrandomization. But in the outpatient arm, less than half of the women required additional inpatient ripening, and nearly 40% returned in spontaneous labor, suggesting that outpatient prostaglandin administration may indeed save women a significant amount of time on labor and delivery.
Safety
The safety of outpatient administration of prostaglandins is the biggest concern, especially since, when prostaglandins are compared to outpatient Foley catheter use, Foleys are overall associated with less tachysystole, fetal intolerance, and meconium-stained fluid.3 Foley catheter use for cervical ripening may not be an appropriate choice for all patients, however. For instance, our case patient has a closed cervix, which could make Foley insertion uncomfortable or even impossible. Misoprostol use also offers the potential for flexibility in cervical ripening protocols as patients need not return for Foley balloon removal and indeed labor induction need not take place immediately after administration of misoprostol.
Patients also may prefer outpatient cervical ripening with misoprostol over a Foley. There are some data to suggest that women, overall, have a preference toward prostaglandins; in the PROBAAT-II trial, which compared inpatient oral misoprostol to Foley catheter for cervical ripening, 12% of women in the Foley arm would have preferred another method of induction (vs 6% in the misoprostol arm; P = .02).27 This preference may be magnified in an outpatient setting.
But, again, is outpatient administration of prostaglandins safe? The published trials thus far have not reported an increase in out-of-hospital deliveries or adverse fetal outcomes. However, studies have been of limited size to see more rare outcomes. Unfortunately, an adequately powered study to demonstrate safety is likely never to be accomplished, given that if used responsibly (in low-risk patients with adequate monitoring after administration) the incidence of adverse fetal outcomes during the at-home portion of cervical ripening is likely to be very low. With responsible use, outpatient administration of prostaglandins should be safe. Women are monitored after misoprostol administration and are not sent home if there are any concerns for fetal distress or if frequent contractions continue. Misoprostol reaches maximum blood concentration 30 minutes after oral administration and 70 to 80 minutes after vaginal administration.28 After this time, if contractions start to intensify it is likely that misoprostol has triggered spontaneous labor. In this setting, women are routinely allowed to spontaneously labor at home. One may even argue that outpatient misoprostol could lead to improved safety, as women essentially have a contraction stress test prior to spontaneous labor, and misoprostol administration as an outpatient, as opposed to as an inpatient, may allow for longer time intervals between doses, which could prevent dose stacking.
Continue to: Our approach to pharmacologic cervical ripening...
Our approach to pharmacologic cervical ripening
Our hospital has been conducting outpatient cervical ripening using vaginal misoprostol for more th

Conclusion
While the data continue to be limited, we strongly believe there is sufficient quality evidence from a safety and efficacy perspective to support implementation and evaluation of outpatient cervical ripening protocols for low-risk pregnancies. In the setting of renewed commitments to reducing suboptimal health care costs and utilization as well as increasing patient satisfaction and control in their birthing experiences, we posit it is the responsibility of obstetricians, L&D leadership, and health care institutions to explore the implementation of outpatient cervical ripening for appropriate candidates in their settings.
- Martin JA, Hamilton BE, Osterman MJ, et al. Births: final data for 2015. Natl Vital Stat Rep. 2017;66:1.
- Grobman WA, Rice MM, Reddy UM, et al. Labor induction versus expectant management in low-risk nulliparous women. N Engl J Med. 2018;379:513-523.
- Jozwiak M, Bloemenkamp KW, Kelly AJ, et al. Mechanical methods for induction of labor. Cochrane Database Syst Rev. 2012;(3):CD001233.
- Alfirevic Z, Kelly AJ, Dowswell T. Intravenous oxytocin alone for cervical ripening and induction of labour. Cochrane Database Syst Rev. 2009;(4):CD003246.
- Levine LD, Downes KL, Elovitz MA, et al. Mechanical and pharmacologic methods of labor induction: a randomized controlled trial. Obstet Gynecol. 2016;128:1357-1364.
- ACOG Committee on Practice Bulletins—Obstetrics. ACOG practice bulletin no. 107: induction of labor. Obstet Gynecol. 2009;114(2 pt 1):386-397. Reaffirmed 2019.
- Sciscione AC, Bedder CL, Hoffman MK, et al. The timing of adverse events with Foley catheter preinduction cervical ripening; implications for outpatient use. Am J Perinatol. 2014;31:781-786.
- Diederen M, Gommers J, Wilkinson C, et al. Safety of the balloon catheter for cervical ripening in outpatient care: complications during the period from insertion to expulsion of a balloon catheter in the process of labour induction: a systematic review. BJOG. 2018;125:1086-1095.
- McKenna DS, Duke JM. Effectiveness and infectious morbidity of outpatient cervical ripening with a Foley catheter. J Reprod Med. 2004;49:28-32.
- Kuper SG, Jauk VC, George DM, et al. Outpatient Foley catheter for induction of labor in parous women: a randomized controlled trial. Obstet Gynecol. 2018;132:94-101.
- Wilkinson C, Adelson P, Turnbull D. A comparison of inpatient with outpatient balloon catheter cervical ripening: a pilot randomized controlled trial. BMC Pregnancy Childbirth. 2015;15:126.
- Kruit H, Heikinheimo O, Ulander VM, et al. Foley catheter induction of labor as an outpatient procedure. J Perinatol. 2016;36:618-622.
- Sciscione AC, Muench M, Pollock M, et al. Transcervical Foley catheter for preinduction cervical ripening in an outpatient versus inpatient setting. Obstet Gynecol. 2001;98(5 pt 1):751-756.
- Policiano C, Pimenta M, Martins D, et al. Outpatient versus inpatient cervix priming with Foley catheter: a randomized trial. Eur J Obstet Gynecol Reprod Biol. 2017;210:1-6.
- Ten Eikelder M, van Baaren GJ, Oude Rengerink K, et al. Comparing induction of labour with oral misoprostol or Foley catheter at term: cost effectiveness analysis of a randomised controlled multi-centre non-inferiority trial. BJOG. 2018;125:375-383.
- Henry A, Madan A, Reid R, et al. Outpatient Foley catheter versus inpatient prostaglandin E2 gel for induction of labour: a randomised trial. BMC Pregnancy Childbirth. 2013;13:25.
- Howard K, Gerard K, Adelson P, et al. Women’s preferences for inpatient and outpatient priming for labour induction: a discrete choice experiment. BMC Health Serv Res. 2014;14:330.
- Main E, LaGrew D; California Maternal Quality Care Collaborative. Induction of labor risks, benefits, and techniques for increasing success. June 14, 2017. https://www .cmqcc.org/resource/induction-labor-risk-benefits-and-techniques-increasing -success. Accessed August 21, 2019.
- Gaffaney CA, Saul LL, Rumney PJ, et al. Outpatient oral misoprostol for prolonged pregnancies: a pilot investigation. Am J Perinatol. 2009;26:673-677.
- PonMalar J, Benjamin SJ, Abraham A, et al. Randomized double-blind placebo controlled study of preinduction cervical priming with 25 µg of misoprostol in the outpatient setting to prevent formal induction of labour. Arch Gynecol Obstet. 2017;295:33-38.
- Stitely ML, Browning J, Fowler M, et al. Outpatient cervical ripening with intravaginal misoprostol. Obstet Gynecol. 2000;96(5 pt 1):684-688.
- McKenna DS, Ester JB, Proffitt M, et al. Misoprostol outpatient cervical ripening without subsequent induction of labor: a randomized trial. Obstet Gynecol. 2004;104:579-584.
- Oboro VO, Tabowei TO. Outpatient misoprostol cervical ripening withoutsubsequent induction of labor to prevent post-term pregnancy. Acta Obstet Gynecol Scand. 2005;84:628-631.
- Chang DW, Velazquez MD, Colyer M, et al. Vaginal misoprostol for cervical ripening at term: comparison of outpatient vs. inpatient administration. J Reprod Med. 2005;50:735-739.
- Biem SR, Turnell RW, Olatunbosun O, et al. A randomized controlled trial of outpatient versus inpatient labour induction with vaginal controlled-release prostaglandin-E2: effectiveness and satisfaction. J Obstet Gynaecol Can. 2003;25:23-31.
- Wilkinson C, Bryce R, Adelson P, et al. A randomised controlled trial of outpatient compared with inpatient cervical ripening with prostaglandin E₂ (OPRA study). BJOG. 2015;122:94-104.
- Ten Eikelder ML, van de Meent MM, Mast K, et al. Women’s experiences with and preference for induction of labor with oral misoprostol or Foley catheter at term. Am J Perinatol. 2017;34:138-146.
- Tang OS, Gemzell-Danielsson K, Ho PC. Misoprostol: pharmacokinetic profiles, effects on the uterus and side-effects. Int J Gynaecol Obstet. 2007;99 (suppl 2):S160-S167.
- Martin JA, Hamilton BE, Osterman MJ, et al. Births: final data for 2015. Natl Vital Stat Rep. 2017;66:1.
- Grobman WA, Rice MM, Reddy UM, et al. Labor induction versus expectant management in low-risk nulliparous women. N Engl J Med. 2018;379:513-523.
- Jozwiak M, Bloemenkamp KW, Kelly AJ, et al. Mechanical methods for induction of labor. Cochrane Database Syst Rev. 2012;(3):CD001233.
- Alfirevic Z, Kelly AJ, Dowswell T. Intravenous oxytocin alone for cervical ripening and induction of labour. Cochrane Database Syst Rev. 2009;(4):CD003246.
- Levine LD, Downes KL, Elovitz MA, et al. Mechanical and pharmacologic methods of labor induction: a randomized controlled trial. Obstet Gynecol. 2016;128:1357-1364.
- ACOG Committee on Practice Bulletins—Obstetrics. ACOG practice bulletin no. 107: induction of labor. Obstet Gynecol. 2009;114(2 pt 1):386-397. Reaffirmed 2019.
- Sciscione AC, Bedder CL, Hoffman MK, et al. The timing of adverse events with Foley catheter preinduction cervical ripening; implications for outpatient use. Am J Perinatol. 2014;31:781-786.
- Diederen M, Gommers J, Wilkinson C, et al. Safety of the balloon catheter for cervical ripening in outpatient care: complications during the period from insertion to expulsion of a balloon catheter in the process of labour induction: a systematic review. BJOG. 2018;125:1086-1095.
- McKenna DS, Duke JM. Effectiveness and infectious morbidity of outpatient cervical ripening with a Foley catheter. J Reprod Med. 2004;49:28-32.
- Kuper SG, Jauk VC, George DM, et al. Outpatient Foley catheter for induction of labor in parous women: a randomized controlled trial. Obstet Gynecol. 2018;132:94-101.
- Wilkinson C, Adelson P, Turnbull D. A comparison of inpatient with outpatient balloon catheter cervical ripening: a pilot randomized controlled trial. BMC Pregnancy Childbirth. 2015;15:126.
- Kruit H, Heikinheimo O, Ulander VM, et al. Foley catheter induction of labor as an outpatient procedure. J Perinatol. 2016;36:618-622.
- Sciscione AC, Muench M, Pollock M, et al. Transcervical Foley catheter for preinduction cervical ripening in an outpatient versus inpatient setting. Obstet Gynecol. 2001;98(5 pt 1):751-756.
- Policiano C, Pimenta M, Martins D, et al. Outpatient versus inpatient cervix priming with Foley catheter: a randomized trial. Eur J Obstet Gynecol Reprod Biol. 2017;210:1-6.
- Ten Eikelder M, van Baaren GJ, Oude Rengerink K, et al. Comparing induction of labour with oral misoprostol or Foley catheter at term: cost effectiveness analysis of a randomised controlled multi-centre non-inferiority trial. BJOG. 2018;125:375-383.
- Henry A, Madan A, Reid R, et al. Outpatient Foley catheter versus inpatient prostaglandin E2 gel for induction of labour: a randomised trial. BMC Pregnancy Childbirth. 2013;13:25.
- Howard K, Gerard K, Adelson P, et al. Women’s preferences for inpatient and outpatient priming for labour induction: a discrete choice experiment. BMC Health Serv Res. 2014;14:330.
- Main E, LaGrew D; California Maternal Quality Care Collaborative. Induction of labor risks, benefits, and techniques for increasing success. June 14, 2017. https://www .cmqcc.org/resource/induction-labor-risk-benefits-and-techniques-increasing -success. Accessed August 21, 2019.
- Gaffaney CA, Saul LL, Rumney PJ, et al. Outpatient oral misoprostol for prolonged pregnancies: a pilot investigation. Am J Perinatol. 2009;26:673-677.
- PonMalar J, Benjamin SJ, Abraham A, et al. Randomized double-blind placebo controlled study of preinduction cervical priming with 25 µg of misoprostol in the outpatient setting to prevent formal induction of labour. Arch Gynecol Obstet. 2017;295:33-38.
- Stitely ML, Browning J, Fowler M, et al. Outpatient cervical ripening with intravaginal misoprostol. Obstet Gynecol. 2000;96(5 pt 1):684-688.
- McKenna DS, Ester JB, Proffitt M, et al. Misoprostol outpatient cervical ripening without subsequent induction of labor: a randomized trial. Obstet Gynecol. 2004;104:579-584.
- Oboro VO, Tabowei TO. Outpatient misoprostol cervical ripening withoutsubsequent induction of labor to prevent post-term pregnancy. Acta Obstet Gynecol Scand. 2005;84:628-631.
- Chang DW, Velazquez MD, Colyer M, et al. Vaginal misoprostol for cervical ripening at term: comparison of outpatient vs. inpatient administration. J Reprod Med. 2005;50:735-739.
- Biem SR, Turnell RW, Olatunbosun O, et al. A randomized controlled trial of outpatient versus inpatient labour induction with vaginal controlled-release prostaglandin-E2: effectiveness and satisfaction. J Obstet Gynaecol Can. 2003;25:23-31.
- Wilkinson C, Bryce R, Adelson P, et al. A randomised controlled trial of outpatient compared with inpatient cervical ripening with prostaglandin E₂ (OPRA study). BJOG. 2015;122:94-104.
- Ten Eikelder ML, van de Meent MM, Mast K, et al. Women’s experiences with and preference for induction of labor with oral misoprostol or Foley catheter at term. Am J Perinatol. 2017;34:138-146.
- Tang OS, Gemzell-Danielsson K, Ho PC. Misoprostol: pharmacokinetic profiles, effects on the uterus and side-effects. Int J Gynaecol Obstet. 2007;99 (suppl 2):S160-S167.