User login
Noninvasive ventilation: Options and cautions for patients with COVID-19
Early on in the COVID-19 pandemic,
“We were concerned that, if we put them on high-flow nasal cannula or a noninvasive ventilation, that we would create aerosols that would then be a risk to clinicians,” Meghan Lane-Fall, MD, MSHP, FCCM, said at a Society for Critical Care Medicine virtual meeting called COVID-19: What’s Next. “However, we’ve gotten much more comfortable with infection control. We’ve gotten much more comfortable with controlling these aerosols, with making sure that our clinicians are protected with the appropriate protective equipment. We’ve also realized that patients who end up becoming intubated have really poor outcomes, so we’ve looked at our practice critically and tried to figure out how to support patients noninvasively when that’s possible.”
Respiratory support options
According to Dr. Lane-Fall, an associate professor of anesthesiology and critical care at the University of Pennsylvania, Philadelphia, there are two basic types of respiratory support in patients with moderate, severe, or critical COVID-19: noninvasive and invasive. Noninvasive options include CPAP or BiPAP which can be delivered through nasal pillows, masks, and helmets, as well as high-flow nasal oxygen. Invasive options include endotracheal intubation, tracheostomy, and extracorporeal membrane oxygenation (ECMO), usually the veno-venous (VV) form. “But it’s uncommon to need VV ECMO, even in patients who have critical COVID-19,” she said.
Factors that favor noninvasive ventilation include stably high oxygen requirements, normal mental status, ward location of care, and moderate to severe COVID-19. Factors that favor invasive ventilation include someone who’s deteriorating rapidly, “whose oxygen requirements aren’t stable or who is cardiopulmonary compromised,” said Dr. Lane-Fall, who is also co–medical director of the Trauma Surgery Intensive Care Unit at Penn Presbyterian Medical Center, also in Philadelphia. Other factors include the need for other invasive procedures such as surgery or if they have severe to critical COVID-19, “not just pneumonia, but [illness that’s] progressing into [acute respiratory distress syndrome],” she said.
Indications for urgent endotracheal intubation as opposed to giving a trial of noninvasive ventilation or high-flow nasal oxygen include altered mental status, inability to protect airway, copious amounts of secretions, a Glasgow Coma Scale score of less than 8, severe respiratory acidosis, hypopnea or apnea, shock, or an inability to tolerate noninvasive support. “This is a relative contraindication,” Dr. Lane-Fall said. “I’ve certainly talked people through the BiPAP mask or the helmet. If you tell a patient, ‘I don’t want to have to put in a breathing tube; I want to maintain you on this,’ often they’ll be able to work through it.”
Safety precautions
Aerosolizing procedures require attention to location, personnel, and equipment, including personal protective equipment (PPE), said Dr. Lane-Fall, who is an anesthesiologist by training. “When you are intubating someone, whether they have COVID-19 or not, you are sort of in the belly of the beast,” she said. “You are very exposed to secretions that occur at the time of endotracheal intubation. That’s why it’s important for us to have PPE and barriers to protect ourselves from potential exposure to aerosols during the care of patients with COVID-19.”
In February 2020, the non-for-profit Anesthesia Patient Safety Foundation published recommendations for airway management in patients with suspected COVID-19. A separate guidance was published the British Journal of Anaesthesiology based on emergency tracheal intubation in 202 patients with COVID-19 in Wuhan, China. “The idea here is that you want to intubate under controlled conditions,” said Dr. Lane-Fall, who is an author of the guidance. “You want to use the most experienced operator. You want to have full PPE, including an N95 mask, or something more protective like a powered air purifying respirator or an N95 mask with a face shield. You want the eyes, nose, and mouth of the operator covered completely.”
CPR, another aerosolizing procedure, requires vigilant safety precautions as well. “We struggled with this a little bit at our institution, because our inclination as intensivists when someone is pulseless is to run into the room and start chest compressions and to start resuscitation,” Dr. Lane-Fall said. “But the act of chest compression itself can create aerosols that can present risk to clinicians. We had to tell our clinicians that they have to put on PPE before they do CPR. The buzz phrase here is that there is no emergency in a pandemic. The idea here is that the good of that one patient is outweighed by the good of all the other patients that you could care for if you didn’t have COVID-19 as a clinician. So we have had to encourage our staff to put on PPE first before attending to patients first, even if it delays patient care. Once you have donned PPE, when you’re administering CPR, the number of staff should be minimized. You should have a compressor, and someone to relieve the compressor, and a code leader, someone tending to the airway. But in general, anyone who’s not actively involved should not be in the room.”
Risks during extubation
Extubation of COVID-19 patients is also an aerosolizing procedure not just because you’re pulling an endotracheal tube out of the airway but because coughing is a normal part of extubation. “We’ve had to be careful with how we approach extubation in COVID-19 patients,” Dr. Lane-Fall said. “Ideally you’re doing this in a negative pressure environment. We have also had to use full PPE, covering the eyes and face, and putting on a gown for precaution.”
Reintubation of COVID-19 patients is not uncommon. She and her colleagues at Penn Medicine created procedures for having intubators at the ready outside the room in case the patient were to decompensate clinically. “Another thing we learned is that it’s useful to do a leak test prior to extubation, because there may be airway edema related to prolonged intubation in these patients,” Dr. Lane-Fall said. “We found that, if a leak is absent on checking the cuff leak, the use of steroids for a day or 2 may help decrease airway edema. That improves the chances of extubation success.”
Strategies for aerosol containment
She concluded her remarks by reviewing airway control adjuncts and clinician safety. This includes physically isolating COVID-19 patients in negative pressure rooms and avoiding and minimizing aerosols, including the use of rapid intubation, “where we induce anesthesia for intubation but we don’t bag-mask the patient because that creates aerosols,” she said. The Anesthesia Patient Safety Foundation guidelines advocate for the use of video laryngoscopy so that you can visualize the glottis easily “and make sure that you successfully intubate the glottis and not the esophagus,” she said.
A smart strategy for aerosol containment is to use the most experienced laryngoscopist available. “If you are in a teaching program, ideally you’re using your most experienced resident, or you’re using fellows or attending physicians,” Dr. Lane-Fall said. “This is not the space for an inexperienced learner.”
Another way to make intubation faster and easier in COVID-19 patients is to use an intubation box, which features a plexiglass shield that enables the intubator to use their hands to get in the patient’s airway while being protected from viral droplets generated during intubation. The box can be cleaned after each use. Blueprints for an open source intubation box can be found at http://www.intubationbox.com.
Expert view on aerosol containment in COVID-19
“While there is a dearth of evidence from controlled trials, recommendations mentioned in this story are based on the best available evidence and are in agreement with guidelines from several expert groups,” said David L. Bowton, MD, FCCP, FCCM, of the department of anesthesiology at Wake Forest Baptist Health in Winston-Salem, NC. “The recommendation of Dr. Lane-Fall’s that is perhaps most controversial is the use of an intubation box. Multiple designs for these intubation/aerosol containment devices have been proposed, and the data supporting their ease of use and efficacy has been mixed [See Anaesthesia 2020;75(8):1014-21 and Anaesthesia. 2020. doi: 10.1111/anae.15188]. While bag valve mask ventilation should be avoided if possible, it may be a valuable rescue tool in the severely hypoxemic patient when used with two-person technique to achieve a tight seal and a PEEP valve and an HME over the exhalation port to minimize aerosol spread.
“It cannot be stressed enough that the most skilled individual should be tasked with intubating the patient and as few providers as possible [usually three] should be in the room and have donned full PPE. Negative pressure rooms should be used whenever feasible. Noninvasive ventilation appears safer from an infection control standpoint than initially feared and its use has become more widespread. However, noninvasive ventilation is not without its hazards, and Dr. Lane-Fall’s enumeration of the patient characteristics applicable to the selection of patients for noninvasive ventilation are extremely important. At our institution, the use of noninvasive ventilation and especially high-flow oxygen therapy has increased. Staff have become more comfortable with the donning and doffing of PPE.”
Dr. Lane-Fall reported having no financial disclosures.
Early on in the COVID-19 pandemic,
“We were concerned that, if we put them on high-flow nasal cannula or a noninvasive ventilation, that we would create aerosols that would then be a risk to clinicians,” Meghan Lane-Fall, MD, MSHP, FCCM, said at a Society for Critical Care Medicine virtual meeting called COVID-19: What’s Next. “However, we’ve gotten much more comfortable with infection control. We’ve gotten much more comfortable with controlling these aerosols, with making sure that our clinicians are protected with the appropriate protective equipment. We’ve also realized that patients who end up becoming intubated have really poor outcomes, so we’ve looked at our practice critically and tried to figure out how to support patients noninvasively when that’s possible.”
Respiratory support options
According to Dr. Lane-Fall, an associate professor of anesthesiology and critical care at the University of Pennsylvania, Philadelphia, there are two basic types of respiratory support in patients with moderate, severe, or critical COVID-19: noninvasive and invasive. Noninvasive options include CPAP or BiPAP which can be delivered through nasal pillows, masks, and helmets, as well as high-flow nasal oxygen. Invasive options include endotracheal intubation, tracheostomy, and extracorporeal membrane oxygenation (ECMO), usually the veno-venous (VV) form. “But it’s uncommon to need VV ECMO, even in patients who have critical COVID-19,” she said.
Factors that favor noninvasive ventilation include stably high oxygen requirements, normal mental status, ward location of care, and moderate to severe COVID-19. Factors that favor invasive ventilation include someone who’s deteriorating rapidly, “whose oxygen requirements aren’t stable or who is cardiopulmonary compromised,” said Dr. Lane-Fall, who is also co–medical director of the Trauma Surgery Intensive Care Unit at Penn Presbyterian Medical Center, also in Philadelphia. Other factors include the need for other invasive procedures such as surgery or if they have severe to critical COVID-19, “not just pneumonia, but [illness that’s] progressing into [acute respiratory distress syndrome],” she said.
Indications for urgent endotracheal intubation as opposed to giving a trial of noninvasive ventilation or high-flow nasal oxygen include altered mental status, inability to protect airway, copious amounts of secretions, a Glasgow Coma Scale score of less than 8, severe respiratory acidosis, hypopnea or apnea, shock, or an inability to tolerate noninvasive support. “This is a relative contraindication,” Dr. Lane-Fall said. “I’ve certainly talked people through the BiPAP mask or the helmet. If you tell a patient, ‘I don’t want to have to put in a breathing tube; I want to maintain you on this,’ often they’ll be able to work through it.”
Safety precautions
Aerosolizing procedures require attention to location, personnel, and equipment, including personal protective equipment (PPE), said Dr. Lane-Fall, who is an anesthesiologist by training. “When you are intubating someone, whether they have COVID-19 or not, you are sort of in the belly of the beast,” she said. “You are very exposed to secretions that occur at the time of endotracheal intubation. That’s why it’s important for us to have PPE and barriers to protect ourselves from potential exposure to aerosols during the care of patients with COVID-19.”
In February 2020, the non-for-profit Anesthesia Patient Safety Foundation published recommendations for airway management in patients with suspected COVID-19. A separate guidance was published the British Journal of Anaesthesiology based on emergency tracheal intubation in 202 patients with COVID-19 in Wuhan, China. “The idea here is that you want to intubate under controlled conditions,” said Dr. Lane-Fall, who is an author of the guidance. “You want to use the most experienced operator. You want to have full PPE, including an N95 mask, or something more protective like a powered air purifying respirator or an N95 mask with a face shield. You want the eyes, nose, and mouth of the operator covered completely.”
CPR, another aerosolizing procedure, requires vigilant safety precautions as well. “We struggled with this a little bit at our institution, because our inclination as intensivists when someone is pulseless is to run into the room and start chest compressions and to start resuscitation,” Dr. Lane-Fall said. “But the act of chest compression itself can create aerosols that can present risk to clinicians. We had to tell our clinicians that they have to put on PPE before they do CPR. The buzz phrase here is that there is no emergency in a pandemic. The idea here is that the good of that one patient is outweighed by the good of all the other patients that you could care for if you didn’t have COVID-19 as a clinician. So we have had to encourage our staff to put on PPE first before attending to patients first, even if it delays patient care. Once you have donned PPE, when you’re administering CPR, the number of staff should be minimized. You should have a compressor, and someone to relieve the compressor, and a code leader, someone tending to the airway. But in general, anyone who’s not actively involved should not be in the room.”
Risks during extubation
Extubation of COVID-19 patients is also an aerosolizing procedure not just because you’re pulling an endotracheal tube out of the airway but because coughing is a normal part of extubation. “We’ve had to be careful with how we approach extubation in COVID-19 patients,” Dr. Lane-Fall said. “Ideally you’re doing this in a negative pressure environment. We have also had to use full PPE, covering the eyes and face, and putting on a gown for precaution.”
Reintubation of COVID-19 patients is not uncommon. She and her colleagues at Penn Medicine created procedures for having intubators at the ready outside the room in case the patient were to decompensate clinically. “Another thing we learned is that it’s useful to do a leak test prior to extubation, because there may be airway edema related to prolonged intubation in these patients,” Dr. Lane-Fall said. “We found that, if a leak is absent on checking the cuff leak, the use of steroids for a day or 2 may help decrease airway edema. That improves the chances of extubation success.”
Strategies for aerosol containment
She concluded her remarks by reviewing airway control adjuncts and clinician safety. This includes physically isolating COVID-19 patients in negative pressure rooms and avoiding and minimizing aerosols, including the use of rapid intubation, “where we induce anesthesia for intubation but we don’t bag-mask the patient because that creates aerosols,” she said. The Anesthesia Patient Safety Foundation guidelines advocate for the use of video laryngoscopy so that you can visualize the glottis easily “and make sure that you successfully intubate the glottis and not the esophagus,” she said.
A smart strategy for aerosol containment is to use the most experienced laryngoscopist available. “If you are in a teaching program, ideally you’re using your most experienced resident, or you’re using fellows or attending physicians,” Dr. Lane-Fall said. “This is not the space for an inexperienced learner.”
Another way to make intubation faster and easier in COVID-19 patients is to use an intubation box, which features a plexiglass shield that enables the intubator to use their hands to get in the patient’s airway while being protected from viral droplets generated during intubation. The box can be cleaned after each use. Blueprints for an open source intubation box can be found at http://www.intubationbox.com.
Expert view on aerosol containment in COVID-19
“While there is a dearth of evidence from controlled trials, recommendations mentioned in this story are based on the best available evidence and are in agreement with guidelines from several expert groups,” said David L. Bowton, MD, FCCP, FCCM, of the department of anesthesiology at Wake Forest Baptist Health in Winston-Salem, NC. “The recommendation of Dr. Lane-Fall’s that is perhaps most controversial is the use of an intubation box. Multiple designs for these intubation/aerosol containment devices have been proposed, and the data supporting their ease of use and efficacy has been mixed [See Anaesthesia 2020;75(8):1014-21 and Anaesthesia. 2020. doi: 10.1111/anae.15188]. While bag valve mask ventilation should be avoided if possible, it may be a valuable rescue tool in the severely hypoxemic patient when used with two-person technique to achieve a tight seal and a PEEP valve and an HME over the exhalation port to minimize aerosol spread.
“It cannot be stressed enough that the most skilled individual should be tasked with intubating the patient and as few providers as possible [usually three] should be in the room and have donned full PPE. Negative pressure rooms should be used whenever feasible. Noninvasive ventilation appears safer from an infection control standpoint than initially feared and its use has become more widespread. However, noninvasive ventilation is not without its hazards, and Dr. Lane-Fall’s enumeration of the patient characteristics applicable to the selection of patients for noninvasive ventilation are extremely important. At our institution, the use of noninvasive ventilation and especially high-flow oxygen therapy has increased. Staff have become more comfortable with the donning and doffing of PPE.”
Dr. Lane-Fall reported having no financial disclosures.
Early on in the COVID-19 pandemic,
“We were concerned that, if we put them on high-flow nasal cannula or a noninvasive ventilation, that we would create aerosols that would then be a risk to clinicians,” Meghan Lane-Fall, MD, MSHP, FCCM, said at a Society for Critical Care Medicine virtual meeting called COVID-19: What’s Next. “However, we’ve gotten much more comfortable with infection control. We’ve gotten much more comfortable with controlling these aerosols, with making sure that our clinicians are protected with the appropriate protective equipment. We’ve also realized that patients who end up becoming intubated have really poor outcomes, so we’ve looked at our practice critically and tried to figure out how to support patients noninvasively when that’s possible.”
Respiratory support options
According to Dr. Lane-Fall, an associate professor of anesthesiology and critical care at the University of Pennsylvania, Philadelphia, there are two basic types of respiratory support in patients with moderate, severe, or critical COVID-19: noninvasive and invasive. Noninvasive options include CPAP or BiPAP which can be delivered through nasal pillows, masks, and helmets, as well as high-flow nasal oxygen. Invasive options include endotracheal intubation, tracheostomy, and extracorporeal membrane oxygenation (ECMO), usually the veno-venous (VV) form. “But it’s uncommon to need VV ECMO, even in patients who have critical COVID-19,” she said.
Factors that favor noninvasive ventilation include stably high oxygen requirements, normal mental status, ward location of care, and moderate to severe COVID-19. Factors that favor invasive ventilation include someone who’s deteriorating rapidly, “whose oxygen requirements aren’t stable or who is cardiopulmonary compromised,” said Dr. Lane-Fall, who is also co–medical director of the Trauma Surgery Intensive Care Unit at Penn Presbyterian Medical Center, also in Philadelphia. Other factors include the need for other invasive procedures such as surgery or if they have severe to critical COVID-19, “not just pneumonia, but [illness that’s] progressing into [acute respiratory distress syndrome],” she said.
Indications for urgent endotracheal intubation as opposed to giving a trial of noninvasive ventilation or high-flow nasal oxygen include altered mental status, inability to protect airway, copious amounts of secretions, a Glasgow Coma Scale score of less than 8, severe respiratory acidosis, hypopnea or apnea, shock, or an inability to tolerate noninvasive support. “This is a relative contraindication,” Dr. Lane-Fall said. “I’ve certainly talked people through the BiPAP mask or the helmet. If you tell a patient, ‘I don’t want to have to put in a breathing tube; I want to maintain you on this,’ often they’ll be able to work through it.”
Safety precautions
Aerosolizing procedures require attention to location, personnel, and equipment, including personal protective equipment (PPE), said Dr. Lane-Fall, who is an anesthesiologist by training. “When you are intubating someone, whether they have COVID-19 or not, you are sort of in the belly of the beast,” she said. “You are very exposed to secretions that occur at the time of endotracheal intubation. That’s why it’s important for us to have PPE and barriers to protect ourselves from potential exposure to aerosols during the care of patients with COVID-19.”
In February 2020, the non-for-profit Anesthesia Patient Safety Foundation published recommendations for airway management in patients with suspected COVID-19. A separate guidance was published the British Journal of Anaesthesiology based on emergency tracheal intubation in 202 patients with COVID-19 in Wuhan, China. “The idea here is that you want to intubate under controlled conditions,” said Dr. Lane-Fall, who is an author of the guidance. “You want to use the most experienced operator. You want to have full PPE, including an N95 mask, or something more protective like a powered air purifying respirator or an N95 mask with a face shield. You want the eyes, nose, and mouth of the operator covered completely.”
CPR, another aerosolizing procedure, requires vigilant safety precautions as well. “We struggled with this a little bit at our institution, because our inclination as intensivists when someone is pulseless is to run into the room and start chest compressions and to start resuscitation,” Dr. Lane-Fall said. “But the act of chest compression itself can create aerosols that can present risk to clinicians. We had to tell our clinicians that they have to put on PPE before they do CPR. The buzz phrase here is that there is no emergency in a pandemic. The idea here is that the good of that one patient is outweighed by the good of all the other patients that you could care for if you didn’t have COVID-19 as a clinician. So we have had to encourage our staff to put on PPE first before attending to patients first, even if it delays patient care. Once you have donned PPE, when you’re administering CPR, the number of staff should be minimized. You should have a compressor, and someone to relieve the compressor, and a code leader, someone tending to the airway. But in general, anyone who’s not actively involved should not be in the room.”
Risks during extubation
Extubation of COVID-19 patients is also an aerosolizing procedure not just because you’re pulling an endotracheal tube out of the airway but because coughing is a normal part of extubation. “We’ve had to be careful with how we approach extubation in COVID-19 patients,” Dr. Lane-Fall said. “Ideally you’re doing this in a negative pressure environment. We have also had to use full PPE, covering the eyes and face, and putting on a gown for precaution.”
Reintubation of COVID-19 patients is not uncommon. She and her colleagues at Penn Medicine created procedures for having intubators at the ready outside the room in case the patient were to decompensate clinically. “Another thing we learned is that it’s useful to do a leak test prior to extubation, because there may be airway edema related to prolonged intubation in these patients,” Dr. Lane-Fall said. “We found that, if a leak is absent on checking the cuff leak, the use of steroids for a day or 2 may help decrease airway edema. That improves the chances of extubation success.”
Strategies for aerosol containment
She concluded her remarks by reviewing airway control adjuncts and clinician safety. This includes physically isolating COVID-19 patients in negative pressure rooms and avoiding and minimizing aerosols, including the use of rapid intubation, “where we induce anesthesia for intubation but we don’t bag-mask the patient because that creates aerosols,” she said. The Anesthesia Patient Safety Foundation guidelines advocate for the use of video laryngoscopy so that you can visualize the glottis easily “and make sure that you successfully intubate the glottis and not the esophagus,” she said.
A smart strategy for aerosol containment is to use the most experienced laryngoscopist available. “If you are in a teaching program, ideally you’re using your most experienced resident, or you’re using fellows or attending physicians,” Dr. Lane-Fall said. “This is not the space for an inexperienced learner.”
Another way to make intubation faster and easier in COVID-19 patients is to use an intubation box, which features a plexiglass shield that enables the intubator to use their hands to get in the patient’s airway while being protected from viral droplets generated during intubation. The box can be cleaned after each use. Blueprints for an open source intubation box can be found at http://www.intubationbox.com.
Expert view on aerosol containment in COVID-19
“While there is a dearth of evidence from controlled trials, recommendations mentioned in this story are based on the best available evidence and are in agreement with guidelines from several expert groups,” said David L. Bowton, MD, FCCP, FCCM, of the department of anesthesiology at Wake Forest Baptist Health in Winston-Salem, NC. “The recommendation of Dr. Lane-Fall’s that is perhaps most controversial is the use of an intubation box. Multiple designs for these intubation/aerosol containment devices have been proposed, and the data supporting their ease of use and efficacy has been mixed [See Anaesthesia 2020;75(8):1014-21 and Anaesthesia. 2020. doi: 10.1111/anae.15188]. While bag valve mask ventilation should be avoided if possible, it may be a valuable rescue tool in the severely hypoxemic patient when used with two-person technique to achieve a tight seal and a PEEP valve and an HME over the exhalation port to minimize aerosol spread.
“It cannot be stressed enough that the most skilled individual should be tasked with intubating the patient and as few providers as possible [usually three] should be in the room and have donned full PPE. Negative pressure rooms should be used whenever feasible. Noninvasive ventilation appears safer from an infection control standpoint than initially feared and its use has become more widespread. However, noninvasive ventilation is not without its hazards, and Dr. Lane-Fall’s enumeration of the patient characteristics applicable to the selection of patients for noninvasive ventilation are extremely important. At our institution, the use of noninvasive ventilation and especially high-flow oxygen therapy has increased. Staff have become more comfortable with the donning and doffing of PPE.”
Dr. Lane-Fall reported having no financial disclosures.
FROM AN SCCM VIRTUAL MEETING
Nocturnal oxygen no help for isolated desaturation in COPD
Nocturnal oxygen therapy for patients with COPD and isolated nocturnal oxygen desaturation does not improve survival or delay disease progression, according to findings published Sept. 17 in The New England Journal of Medicine. The new report adds to evidence that the widely implemented and costly practice may be unnecessary.
Patients with COPD who do not qualify for long-term oxygen therapy (LTOT) are commonly prescribed nocturnal oxygen in the belief that it can delay disease progression, possibly by decreasing alveolar hypoventilation and ventilation-perfusion mismatch.
But investigations so far and the new study from the International Nocturnal Oxygen (INOX) Trial have not borne this out.
“There is no indication that nocturnal oxygen has a positive or negative effect on survival or progression to long-term oxygen therapy in patients with nocturnal hypoxemia in COPD. Consequently, there is no reason for physicians to screen for nocturnal hypoxemia in COPD,” study leader Yves Lacasse, MD, told Medscape Medical News.
Lacasse is from the Institut Universitaire de Cardiologie et de Pneumologie de Québec–Université Laval, Quebec, Canada.
The idea that the therapy helps is firmly entrenched.
In the early 1980s, two trials indicated that patients who had COPD and severe chronic daytime hypoxemia benefit from LTOT (15-18 hours a day or longer).
A decade later, two landmark trials (the Nocturnal Oxygen Therapy Trial and the British Medical Research Council Trial) added to evidence that LTOT may prolong life for patients with COPD and severe daytime hypoxemia.
“The good news from both trials was that oxygen saves lives. From this moment, oxygen therapy became a standard of care, and confirmatory trials would be considered unethical,” Lacasse explained.
“Oxygen therapy gained widespread acceptance by official organizations for treatment of most chronic cardiorespiratory conditions complicated by severe hypoxemia, even if proof of efficacy is lacking. New indications emerged, such as isolated nocturnal oxygen desaturation. Even in COPD, inappropriate prescriptions of home oxygen therapy are not unusual. Oxygen is everywhere,” Lacasse continued.
A meta-analysis from 2005 identified two trials that evaluated home oxygen therapy specifically for isolated nocturnal desaturation. Both found no survival benefit from nocturnal oxygen.
The study by Lacasse and colleagues assessed effects on mortality or worsening of disease (progression to LTOT) with 3-4 years of nocturnal oxygen supplementation.
Participants, whose oxygen saturation was less than 90% for at least 30% of the recording time on nocturnal oximetry, received oxygen or ambient air from a sham device as a placebo for at least 4 hours per session. The goal of treatment was nocturnal oxygen saturation exceeding 90% for at least 90% of the recorded time.
The trial protocol excluded patients with severe obesity, apnea, lung cancer, left heart failure, interstitial lung disease, or bronchiectasis.
The study was initially powered in 2010 to include 600 participants, with half to receive placebo. The study assumed mortality of 20% among control patients over 3 years; 20% of patients progressed to LTOT.
When recruiting lagged, the data safety monitoring board and steering committee extended follow-up to 4 years. In 2014, they requested an interim analysis, and recruitment ceased. Overall, 243 patients participated.
Lacasse cited several reasons for the difficulty with recruitment as well as retention: unwillingness to take the risk of receiving placebo instead of a readily available treatment, fading interest over time, and frailty that affects compliance.
Patients in the study came from 28 community or university-affiliated hospitals in Canada, Portugal, Spain, and France. At the 3-year mark, 39% of patients (48 of 123) who were assigned to nocturnal oxygen therapy and 42% (50 of 119) of those taking placebo had met criteria for LTOT or had died (difference, −3.0 percentage points; P = .64). The groups did not differ appreciably in rates of exacerbation and hospitalization.
The researchers could not analyze subgroups because the patients were very similar with regard to the severity of nocturnal oxygen desaturation, Lacasse said.
Economics enters into the picture – home oxygen therapy is second only to hospitalization as the most expensive healthcare expenditure associated with clinical care for COPD in developed countries. “The math is simple. There is enormous potential for saving money if the results of our clinical trial are applied appropriately,” said Lacasse.
William Bailey, MD, professor emeritus of pulmonary, allergy, and critical care medicine at the University of Alabama at Birmingham, agrees that the practice is overused.
“There is a built-in bias in the medical community. Most believe that anyone with lung disease benefits from oxygen. Even some of our investigators had a hard time believing the results. The study was well designed, carefully carried out, and I feel confident that the results are reliable,” he said.
Shawn P. E. Nishi, MD, director of bronchoscopy and advanced pulmonary procedures, division of pulmonary and critical care medicine, the University of Texas Medical Branch, Galveston, Texas, mentioned the study’s main limitation, which the authors readily acknowledge.
“Unfortunately, the trial had difficulty recruiting subjects, with less than half of expected enrollment achieved, and was underpowered to make any conclusions. Other studies have examined nocturnal oxygen use and have not shown a mortality benefit,” Nishi explained.
She added that the study did not evaluate use of LTOT for improving outcomes other than mortality, including quality of life, cardiovascular morbidity, depression, cognitive function, exercise capacity, and frequency of COPD exacerbations or hospitalization.
Other limitations of the study include suboptimal adherence to the therapy and interpretation of the clinical significance on the basis of a survey of Canadian pulmonologists.
This article first appeared on Medscape.com.
Nocturnal oxygen therapy for patients with COPD and isolated nocturnal oxygen desaturation does not improve survival or delay disease progression, according to findings published Sept. 17 in The New England Journal of Medicine. The new report adds to evidence that the widely implemented and costly practice may be unnecessary.
Patients with COPD who do not qualify for long-term oxygen therapy (LTOT) are commonly prescribed nocturnal oxygen in the belief that it can delay disease progression, possibly by decreasing alveolar hypoventilation and ventilation-perfusion mismatch.
But investigations so far and the new study from the International Nocturnal Oxygen (INOX) Trial have not borne this out.
“There is no indication that nocturnal oxygen has a positive or negative effect on survival or progression to long-term oxygen therapy in patients with nocturnal hypoxemia in COPD. Consequently, there is no reason for physicians to screen for nocturnal hypoxemia in COPD,” study leader Yves Lacasse, MD, told Medscape Medical News.
Lacasse is from the Institut Universitaire de Cardiologie et de Pneumologie de Québec–Université Laval, Quebec, Canada.
The idea that the therapy helps is firmly entrenched.
In the early 1980s, two trials indicated that patients who had COPD and severe chronic daytime hypoxemia benefit from LTOT (15-18 hours a day or longer).
A decade later, two landmark trials (the Nocturnal Oxygen Therapy Trial and the British Medical Research Council Trial) added to evidence that LTOT may prolong life for patients with COPD and severe daytime hypoxemia.
“The good news from both trials was that oxygen saves lives. From this moment, oxygen therapy became a standard of care, and confirmatory trials would be considered unethical,” Lacasse explained.
“Oxygen therapy gained widespread acceptance by official organizations for treatment of most chronic cardiorespiratory conditions complicated by severe hypoxemia, even if proof of efficacy is lacking. New indications emerged, such as isolated nocturnal oxygen desaturation. Even in COPD, inappropriate prescriptions of home oxygen therapy are not unusual. Oxygen is everywhere,” Lacasse continued.
A meta-analysis from 2005 identified two trials that evaluated home oxygen therapy specifically for isolated nocturnal desaturation. Both found no survival benefit from nocturnal oxygen.
The study by Lacasse and colleagues assessed effects on mortality or worsening of disease (progression to LTOT) with 3-4 years of nocturnal oxygen supplementation.
Participants, whose oxygen saturation was less than 90% for at least 30% of the recording time on nocturnal oximetry, received oxygen or ambient air from a sham device as a placebo for at least 4 hours per session. The goal of treatment was nocturnal oxygen saturation exceeding 90% for at least 90% of the recorded time.
The trial protocol excluded patients with severe obesity, apnea, lung cancer, left heart failure, interstitial lung disease, or bronchiectasis.
The study was initially powered in 2010 to include 600 participants, with half to receive placebo. The study assumed mortality of 20% among control patients over 3 years; 20% of patients progressed to LTOT.
When recruiting lagged, the data safety monitoring board and steering committee extended follow-up to 4 years. In 2014, they requested an interim analysis, and recruitment ceased. Overall, 243 patients participated.
Lacasse cited several reasons for the difficulty with recruitment as well as retention: unwillingness to take the risk of receiving placebo instead of a readily available treatment, fading interest over time, and frailty that affects compliance.
Patients in the study came from 28 community or university-affiliated hospitals in Canada, Portugal, Spain, and France. At the 3-year mark, 39% of patients (48 of 123) who were assigned to nocturnal oxygen therapy and 42% (50 of 119) of those taking placebo had met criteria for LTOT or had died (difference, −3.0 percentage points; P = .64). The groups did not differ appreciably in rates of exacerbation and hospitalization.
The researchers could not analyze subgroups because the patients were very similar with regard to the severity of nocturnal oxygen desaturation, Lacasse said.
Economics enters into the picture – home oxygen therapy is second only to hospitalization as the most expensive healthcare expenditure associated with clinical care for COPD in developed countries. “The math is simple. There is enormous potential for saving money if the results of our clinical trial are applied appropriately,” said Lacasse.
William Bailey, MD, professor emeritus of pulmonary, allergy, and critical care medicine at the University of Alabama at Birmingham, agrees that the practice is overused.
“There is a built-in bias in the medical community. Most believe that anyone with lung disease benefits from oxygen. Even some of our investigators had a hard time believing the results. The study was well designed, carefully carried out, and I feel confident that the results are reliable,” he said.
Shawn P. E. Nishi, MD, director of bronchoscopy and advanced pulmonary procedures, division of pulmonary and critical care medicine, the University of Texas Medical Branch, Galveston, Texas, mentioned the study’s main limitation, which the authors readily acknowledge.
“Unfortunately, the trial had difficulty recruiting subjects, with less than half of expected enrollment achieved, and was underpowered to make any conclusions. Other studies have examined nocturnal oxygen use and have not shown a mortality benefit,” Nishi explained.
She added that the study did not evaluate use of LTOT for improving outcomes other than mortality, including quality of life, cardiovascular morbidity, depression, cognitive function, exercise capacity, and frequency of COPD exacerbations or hospitalization.
Other limitations of the study include suboptimal adherence to the therapy and interpretation of the clinical significance on the basis of a survey of Canadian pulmonologists.
This article first appeared on Medscape.com.
Nocturnal oxygen therapy for patients with COPD and isolated nocturnal oxygen desaturation does not improve survival or delay disease progression, according to findings published Sept. 17 in The New England Journal of Medicine. The new report adds to evidence that the widely implemented and costly practice may be unnecessary.
Patients with COPD who do not qualify for long-term oxygen therapy (LTOT) are commonly prescribed nocturnal oxygen in the belief that it can delay disease progression, possibly by decreasing alveolar hypoventilation and ventilation-perfusion mismatch.
But investigations so far and the new study from the International Nocturnal Oxygen (INOX) Trial have not borne this out.
“There is no indication that nocturnal oxygen has a positive or negative effect on survival or progression to long-term oxygen therapy in patients with nocturnal hypoxemia in COPD. Consequently, there is no reason for physicians to screen for nocturnal hypoxemia in COPD,” study leader Yves Lacasse, MD, told Medscape Medical News.
Lacasse is from the Institut Universitaire de Cardiologie et de Pneumologie de Québec–Université Laval, Quebec, Canada.
The idea that the therapy helps is firmly entrenched.
In the early 1980s, two trials indicated that patients who had COPD and severe chronic daytime hypoxemia benefit from LTOT (15-18 hours a day or longer).
A decade later, two landmark trials (the Nocturnal Oxygen Therapy Trial and the British Medical Research Council Trial) added to evidence that LTOT may prolong life for patients with COPD and severe daytime hypoxemia.
“The good news from both trials was that oxygen saves lives. From this moment, oxygen therapy became a standard of care, and confirmatory trials would be considered unethical,” Lacasse explained.
“Oxygen therapy gained widespread acceptance by official organizations for treatment of most chronic cardiorespiratory conditions complicated by severe hypoxemia, even if proof of efficacy is lacking. New indications emerged, such as isolated nocturnal oxygen desaturation. Even in COPD, inappropriate prescriptions of home oxygen therapy are not unusual. Oxygen is everywhere,” Lacasse continued.
A meta-analysis from 2005 identified two trials that evaluated home oxygen therapy specifically for isolated nocturnal desaturation. Both found no survival benefit from nocturnal oxygen.
The study by Lacasse and colleagues assessed effects on mortality or worsening of disease (progression to LTOT) with 3-4 years of nocturnal oxygen supplementation.
Participants, whose oxygen saturation was less than 90% for at least 30% of the recording time on nocturnal oximetry, received oxygen or ambient air from a sham device as a placebo for at least 4 hours per session. The goal of treatment was nocturnal oxygen saturation exceeding 90% for at least 90% of the recorded time.
The trial protocol excluded patients with severe obesity, apnea, lung cancer, left heart failure, interstitial lung disease, or bronchiectasis.
The study was initially powered in 2010 to include 600 participants, with half to receive placebo. The study assumed mortality of 20% among control patients over 3 years; 20% of patients progressed to LTOT.
When recruiting lagged, the data safety monitoring board and steering committee extended follow-up to 4 years. In 2014, they requested an interim analysis, and recruitment ceased. Overall, 243 patients participated.
Lacasse cited several reasons for the difficulty with recruitment as well as retention: unwillingness to take the risk of receiving placebo instead of a readily available treatment, fading interest over time, and frailty that affects compliance.
Patients in the study came from 28 community or university-affiliated hospitals in Canada, Portugal, Spain, and France. At the 3-year mark, 39% of patients (48 of 123) who were assigned to nocturnal oxygen therapy and 42% (50 of 119) of those taking placebo had met criteria for LTOT or had died (difference, −3.0 percentage points; P = .64). The groups did not differ appreciably in rates of exacerbation and hospitalization.
The researchers could not analyze subgroups because the patients were very similar with regard to the severity of nocturnal oxygen desaturation, Lacasse said.
Economics enters into the picture – home oxygen therapy is second only to hospitalization as the most expensive healthcare expenditure associated with clinical care for COPD in developed countries. “The math is simple. There is enormous potential for saving money if the results of our clinical trial are applied appropriately,” said Lacasse.
William Bailey, MD, professor emeritus of pulmonary, allergy, and critical care medicine at the University of Alabama at Birmingham, agrees that the practice is overused.
“There is a built-in bias in the medical community. Most believe that anyone with lung disease benefits from oxygen. Even some of our investigators had a hard time believing the results. The study was well designed, carefully carried out, and I feel confident that the results are reliable,” he said.
Shawn P. E. Nishi, MD, director of bronchoscopy and advanced pulmonary procedures, division of pulmonary and critical care medicine, the University of Texas Medical Branch, Galveston, Texas, mentioned the study’s main limitation, which the authors readily acknowledge.
“Unfortunately, the trial had difficulty recruiting subjects, with less than half of expected enrollment achieved, and was underpowered to make any conclusions. Other studies have examined nocturnal oxygen use and have not shown a mortality benefit,” Nishi explained.
She added that the study did not evaluate use of LTOT for improving outcomes other than mortality, including quality of life, cardiovascular morbidity, depression, cognitive function, exercise capacity, and frequency of COPD exacerbations or hospitalization.
Other limitations of the study include suboptimal adherence to the therapy and interpretation of the clinical significance on the basis of a survey of Canadian pulmonologists.
This article first appeared on Medscape.com.
2020-2021 respiratory viral season: Onset, presentations, and testing likely to differ in pandemic
Respiratory virus seasons usually follow a fairly well-known pattern. Enterovirus 68 (EV-D68) is a summer-to-early fall virus with biennial peak years. Rhinovirus (HRv) and adenovirus (Adv) occur nearly year-round but may have small upticks in the first month or so that children return to school. Early in the school year, upper respiratory infections from both HRv and Adv and viral sore throats from Adv are common, with conjunctivitis from Adv outbreaks in some years. October to November is human parainfluenza (HPiV) 1 and 2 season, often presenting as croup. Human metapneumovirus infections span October through April. In late November to December, influenza begins, usually with an A type, later transitioning to a B type in February through April. Also in December, respiratory syncytial virus (RSV) starts, characteristically with bronchiolitis presentations, peaking in February to March and tapering off in May. In late March to April, HPiV 3 also appears for 4-6 weeks.

Will 2020-2021 be different?
Summer was remarkably free of expected enterovirus activity, suggesting that the seasonal parade may differ this year. Remember that the 2019-2020 respiratory season suddenly and nearly completely stopped in March because of social distancing and lockdowns needed to address the SARS-CoV-2 pandemic.
The mild influenza season in the southern hemisphere suggests that our influenza season also could be mild. But perhaps not – most southern hemisphere countries that are surveyed for influenza activities had the most intense SARS-CoV-2 mitigations, making the observed mildness potentially related more to social mitigation than less virulent influenza strains. If so, southern hemisphere influenza data may not apply to the United States, where social distancing and masks are ignored or used inconsistently by almost half the population.
Further, the stop-and-go pattern of in-person school/college attendance adds to uncertainties for the usual orderly virus-specific seasonality. The result may be multiple stop-and-go “pop-up” or “mini” outbreaks for any given virus potentially reflected as exaggerated local or regional differences in circulation of various viruses. The erratic seasonality also would increase coinfections, which could present with more severe or different symptoms.
SARS-CoV-2’s potential interaction
Will the relatively mild presentations for most children with SARS-CoV-2 hold up in the setting of coinfections or sequential respiratory viral infections? Could SARS-CoV-2 cause worse/more prolonged symptoms or more sequelae if paired simultaneously or in tandem with a traditional respiratory virus? To date, data on the frequency and severity of SARS-CoV-2 coinfections are conflicting and sparse, but it appears that non-SARS-CoV-2 viruses can be involved in 15%-50% pediatric acute respiratory infections.1,2
However, it may not be important to know about coinfecting viruses other than influenza (can be treated) or SARS-CoV-2 (needs quarantine and contact tracing), unless symptoms are atypical or more severe than usual. For example, a young child with bronchiolitis is most likely infected with RSV, but HPiV, influenza, metapneumovirus, HRv, and even SARS-CoV-2 can cause bronchiolitis. Even so, testing outpatients for RSV or non-influenza is not routine or even clinically helpful. Supportive treatment and restriction from daycare attendance are sufficient management for outpatient ARIs whether presenting as bronchiolitis or not.
Considerations for SARS-CoV-2 testing: Outpatient bronchiolitis
If a child presents with classic bronchiolitis but has above moderate to severe symptoms, is SARS-CoV-2 a consideration? Perhaps, if SARS-CoV-2 acts similarly to non-SARS-CoV-2s.
A recent report from the 30th Multicenter Airway Research Collaboration (MARC-30) surveillance study (2007-2014) of children hospitalized with clinical bronchiolitis evaluated respiratory viruses, including RSV and the four common non-SARS coronaviruses using molecular testing.3 Among 1,880 subjects, a CoV (alpha CoV: NL63 or 229E, or beta CoV: KKU1 or OC43) was detected in 12%. Yet most had only RSV (n = 1,661); 32 had only CoV (n = 32). But note that 219 had both.
Bronchiolitis subjects with CoV were older – median 3.7 (1.4-5.8) vs. 2.8 (1.9-7.2) years – and more likely male than were RSV subjects (68% vs. 58%). OC43 was most frequent followed by equal numbers of HKU1 and NL63, while 229E was the least frequent. Medical utilization and severity did not differ among the CoVs, or between RSV+CoV vs. RSV alone, unless one considered CoV viral load as a variable. ICU use increased when the polymerase chain reaction cycle threshold result indicated a high CoV viral load.
These data suggest CoVs are not infrequent coinfectors with RSV in bronchiolitis – and that SARS-CoV-2 is the same. Therefore, a bronchiolitis presentation doesn’t necessarily take us off the hook for the need to consider SARS-CoV-2 testing, particularly in the somewhat older bronchiolitis patient with more than mild symptoms.
Considerations for SARS-CoV-2 testing: Outpatient influenza-like illness
In 2020-2021, the Centers for Disease Control and Prevention recommends considering empiric antiviral treatment for ILIs (fever plus either cough or sore throat) based upon our clinical judgement, even in non-high-risk children.4
While pediatric COVID-19 illnesses are predominantly asymptomatic or mild, a febrile ARI is also a SARS-CoV-2 compatible presentation. So, if all we use is our clinical judgment, how do we know if the febrile ARI is due to influenza or SARS-CoV-2 or both? At least one study used a highly sensitive and specific molecular influenza test to show that the accuracy of clinically diagnosing influenza in children is not much better than flipping a coin and would lead to potential antiviral overuse.5
So, it seems ideal to test for influenza when possible. Point-of-care (POC) tests are frequently used for outpatients. Eight POC Clinical Laboratory Improvement Amendments (CLIA)–waived kits, some also detecting RSV, are available but most have modest sensitivity (60%-80%) compared with lab-based molecular tests.6 That said, if supplies and kits for one of the POC tests are available to us during these SARS-CoV-2 stressed times (back orders seem more common this year), a positive influenza test in the first 48 hours of symptoms confirms the option to prescribe an antiviral. Yet how will we have confidence that the febrile ARI is not also partly due to SARS-CoV-2? Currently febrile ARIs usually are considered SARS-CoV-2 and the children are sent for SARS-CoV-2 testing. During influenza season, it seems we will need to continue to send febrile outpatients for SARS-CoV-2 testing, even if POC influenza positive, via whatever mechanisms are available as time goes on.
We expect more rapid pediatric testing modalities for SARS-CoV-2 (maybe even saliva tests) to become available over the next months. Indeed, rapid antigen tests and rapid molecular tests are being evaluated in adults and seem destined for CLIA waivers as POC tests, and even home testing kits. Pediatric approvals hopefully also will occur. So, the pathways for SARS-CoV-2 testing available now will likely change over this winter. But be aware that supplies/kits will be prioritized to locations within high need areas and bulk purchase contracts. So POC kits may remain scarce for practices, meaning a reference laboratory still could be the way to go for SARS-CoV-2 for at least the rest of 2020. Reference labs are becoming creative as well; one combined detection of influenza A, influenza B, RSV, and SARS-CoV-2 into one test, and hopes to get approval for swab collection that can be done by families at home and mailed in.
Summary
Expect variations on the traditional parade of seasonal respiratory viruses, with increased numbers of coinfections. Choosing the outpatient who needs influenza testing is the same as in past years, although we have CDC permissive recommendations to prescribe antivirals for any outpatient ILI within the first 48 hours of symptoms. Still, POC testing for influenza remains potentially valuable in the ILI patient. The choice of whether and how to test for SARS-CoV-2 given its potential to be a primary or coinfecting agent in presentations linked more closely to a traditional virus (e.g. RSV bronchiolitis) will be a test of our clinical judgement until more data and easier testing are available. Further complicating coinfection recognition is the fact that many sick visits occur by telehealth and much testing is done at drive-through SARS-CoV-2 testing facilities with no clinician exam. Unless we are liberal in SARS-CoV-2 testing, detecting SARS-CoV-2 coinfections is easier said than done given its usually mild presentation being overshadowed by any coinfecting virus.
But understanding who has SARS-CoV-2, even as a coinfection, still is essential in controlling the pandemic. We will need to be vigilant for evolving approaches to SARS-CoV-2 testing in the context of symptomatic ARI presentations, knowing this will likely remain a moving target for the foreseeable future.
Dr. Harrison is professor of pediatrics and pediatric infectious diseases at Children’s Mercy Hospital-Kansas City, Mo. Children’s Mercy Hospital receives grant funding to study two candidate RSV vaccines. The hospital also receives CDC funding under the New Vaccine Surveillance Network for multicenter surveillance of acute respiratory infections, including influenza, RSV, and parainfluenza virus. Email Dr. Harrison at [email protected].
References
1. Pediatrics. 2020;146(1):e20200961.
2. JAMA. 2020 May 26;323(20):2085-6.
3. Pediatrics. 2020. doi: 10.1542/peds.2020-1267.
4. www.cdc.gov/flu/professionals/antivirals/summary-clinicians.htm.
5. J. Pediatr. 2020. doi: 10.1016/j.jpeds.2020.08.007.
6. www.cdc.gov/flu/professionals/diagnosis/table-nucleic-acid-detection.html.
Respiratory virus seasons usually follow a fairly well-known pattern. Enterovirus 68 (EV-D68) is a summer-to-early fall virus with biennial peak years. Rhinovirus (HRv) and adenovirus (Adv) occur nearly year-round but may have small upticks in the first month or so that children return to school. Early in the school year, upper respiratory infections from both HRv and Adv and viral sore throats from Adv are common, with conjunctivitis from Adv outbreaks in some years. October to November is human parainfluenza (HPiV) 1 and 2 season, often presenting as croup. Human metapneumovirus infections span October through April. In late November to December, influenza begins, usually with an A type, later transitioning to a B type in February through April. Also in December, respiratory syncytial virus (RSV) starts, characteristically with bronchiolitis presentations, peaking in February to March and tapering off in May. In late March to April, HPiV 3 also appears for 4-6 weeks.

Will 2020-2021 be different?
Summer was remarkably free of expected enterovirus activity, suggesting that the seasonal parade may differ this year. Remember that the 2019-2020 respiratory season suddenly and nearly completely stopped in March because of social distancing and lockdowns needed to address the SARS-CoV-2 pandemic.
The mild influenza season in the southern hemisphere suggests that our influenza season also could be mild. But perhaps not – most southern hemisphere countries that are surveyed for influenza activities had the most intense SARS-CoV-2 mitigations, making the observed mildness potentially related more to social mitigation than less virulent influenza strains. If so, southern hemisphere influenza data may not apply to the United States, where social distancing and masks are ignored or used inconsistently by almost half the population.
Further, the stop-and-go pattern of in-person school/college attendance adds to uncertainties for the usual orderly virus-specific seasonality. The result may be multiple stop-and-go “pop-up” or “mini” outbreaks for any given virus potentially reflected as exaggerated local or regional differences in circulation of various viruses. The erratic seasonality also would increase coinfections, which could present with more severe or different symptoms.
SARS-CoV-2’s potential interaction
Will the relatively mild presentations for most children with SARS-CoV-2 hold up in the setting of coinfections or sequential respiratory viral infections? Could SARS-CoV-2 cause worse/more prolonged symptoms or more sequelae if paired simultaneously or in tandem with a traditional respiratory virus? To date, data on the frequency and severity of SARS-CoV-2 coinfections are conflicting and sparse, but it appears that non-SARS-CoV-2 viruses can be involved in 15%-50% pediatric acute respiratory infections.1,2
However, it may not be important to know about coinfecting viruses other than influenza (can be treated) or SARS-CoV-2 (needs quarantine and contact tracing), unless symptoms are atypical or more severe than usual. For example, a young child with bronchiolitis is most likely infected with RSV, but HPiV, influenza, metapneumovirus, HRv, and even SARS-CoV-2 can cause bronchiolitis. Even so, testing outpatients for RSV or non-influenza is not routine or even clinically helpful. Supportive treatment and restriction from daycare attendance are sufficient management for outpatient ARIs whether presenting as bronchiolitis or not.
Considerations for SARS-CoV-2 testing: Outpatient bronchiolitis
If a child presents with classic bronchiolitis but has above moderate to severe symptoms, is SARS-CoV-2 a consideration? Perhaps, if SARS-CoV-2 acts similarly to non-SARS-CoV-2s.
A recent report from the 30th Multicenter Airway Research Collaboration (MARC-30) surveillance study (2007-2014) of children hospitalized with clinical bronchiolitis evaluated respiratory viruses, including RSV and the four common non-SARS coronaviruses using molecular testing.3 Among 1,880 subjects, a CoV (alpha CoV: NL63 or 229E, or beta CoV: KKU1 or OC43) was detected in 12%. Yet most had only RSV (n = 1,661); 32 had only CoV (n = 32). But note that 219 had both.
Bronchiolitis subjects with CoV were older – median 3.7 (1.4-5.8) vs. 2.8 (1.9-7.2) years – and more likely male than were RSV subjects (68% vs. 58%). OC43 was most frequent followed by equal numbers of HKU1 and NL63, while 229E was the least frequent. Medical utilization and severity did not differ among the CoVs, or between RSV+CoV vs. RSV alone, unless one considered CoV viral load as a variable. ICU use increased when the polymerase chain reaction cycle threshold result indicated a high CoV viral load.
These data suggest CoVs are not infrequent coinfectors with RSV in bronchiolitis – and that SARS-CoV-2 is the same. Therefore, a bronchiolitis presentation doesn’t necessarily take us off the hook for the need to consider SARS-CoV-2 testing, particularly in the somewhat older bronchiolitis patient with more than mild symptoms.
Considerations for SARS-CoV-2 testing: Outpatient influenza-like illness
In 2020-2021, the Centers for Disease Control and Prevention recommends considering empiric antiviral treatment for ILIs (fever plus either cough or sore throat) based upon our clinical judgement, even in non-high-risk children.4
While pediatric COVID-19 illnesses are predominantly asymptomatic or mild, a febrile ARI is also a SARS-CoV-2 compatible presentation. So, if all we use is our clinical judgment, how do we know if the febrile ARI is due to influenza or SARS-CoV-2 or both? At least one study used a highly sensitive and specific molecular influenza test to show that the accuracy of clinically diagnosing influenza in children is not much better than flipping a coin and would lead to potential antiviral overuse.5
So, it seems ideal to test for influenza when possible. Point-of-care (POC) tests are frequently used for outpatients. Eight POC Clinical Laboratory Improvement Amendments (CLIA)–waived kits, some also detecting RSV, are available but most have modest sensitivity (60%-80%) compared with lab-based molecular tests.6 That said, if supplies and kits for one of the POC tests are available to us during these SARS-CoV-2 stressed times (back orders seem more common this year), a positive influenza test in the first 48 hours of symptoms confirms the option to prescribe an antiviral. Yet how will we have confidence that the febrile ARI is not also partly due to SARS-CoV-2? Currently febrile ARIs usually are considered SARS-CoV-2 and the children are sent for SARS-CoV-2 testing. During influenza season, it seems we will need to continue to send febrile outpatients for SARS-CoV-2 testing, even if POC influenza positive, via whatever mechanisms are available as time goes on.
We expect more rapid pediatric testing modalities for SARS-CoV-2 (maybe even saliva tests) to become available over the next months. Indeed, rapid antigen tests and rapid molecular tests are being evaluated in adults and seem destined for CLIA waivers as POC tests, and even home testing kits. Pediatric approvals hopefully also will occur. So, the pathways for SARS-CoV-2 testing available now will likely change over this winter. But be aware that supplies/kits will be prioritized to locations within high need areas and bulk purchase contracts. So POC kits may remain scarce for practices, meaning a reference laboratory still could be the way to go for SARS-CoV-2 for at least the rest of 2020. Reference labs are becoming creative as well; one combined detection of influenza A, influenza B, RSV, and SARS-CoV-2 into one test, and hopes to get approval for swab collection that can be done by families at home and mailed in.
Summary
Expect variations on the traditional parade of seasonal respiratory viruses, with increased numbers of coinfections. Choosing the outpatient who needs influenza testing is the same as in past years, although we have CDC permissive recommendations to prescribe antivirals for any outpatient ILI within the first 48 hours of symptoms. Still, POC testing for influenza remains potentially valuable in the ILI patient. The choice of whether and how to test for SARS-CoV-2 given its potential to be a primary or coinfecting agent in presentations linked more closely to a traditional virus (e.g. RSV bronchiolitis) will be a test of our clinical judgement until more data and easier testing are available. Further complicating coinfection recognition is the fact that many sick visits occur by telehealth and much testing is done at drive-through SARS-CoV-2 testing facilities with no clinician exam. Unless we are liberal in SARS-CoV-2 testing, detecting SARS-CoV-2 coinfections is easier said than done given its usually mild presentation being overshadowed by any coinfecting virus.
But understanding who has SARS-CoV-2, even as a coinfection, still is essential in controlling the pandemic. We will need to be vigilant for evolving approaches to SARS-CoV-2 testing in the context of symptomatic ARI presentations, knowing this will likely remain a moving target for the foreseeable future.
Dr. Harrison is professor of pediatrics and pediatric infectious diseases at Children’s Mercy Hospital-Kansas City, Mo. Children’s Mercy Hospital receives grant funding to study two candidate RSV vaccines. The hospital also receives CDC funding under the New Vaccine Surveillance Network for multicenter surveillance of acute respiratory infections, including influenza, RSV, and parainfluenza virus. Email Dr. Harrison at [email protected].
References
1. Pediatrics. 2020;146(1):e20200961.
2. JAMA. 2020 May 26;323(20):2085-6.
3. Pediatrics. 2020. doi: 10.1542/peds.2020-1267.
4. www.cdc.gov/flu/professionals/antivirals/summary-clinicians.htm.
5. J. Pediatr. 2020. doi: 10.1016/j.jpeds.2020.08.007.
6. www.cdc.gov/flu/professionals/diagnosis/table-nucleic-acid-detection.html.
Respiratory virus seasons usually follow a fairly well-known pattern. Enterovirus 68 (EV-D68) is a summer-to-early fall virus with biennial peak years. Rhinovirus (HRv) and adenovirus (Adv) occur nearly year-round but may have small upticks in the first month or so that children return to school. Early in the school year, upper respiratory infections from both HRv and Adv and viral sore throats from Adv are common, with conjunctivitis from Adv outbreaks in some years. October to November is human parainfluenza (HPiV) 1 and 2 season, often presenting as croup. Human metapneumovirus infections span October through April. In late November to December, influenza begins, usually with an A type, later transitioning to a B type in February through April. Also in December, respiratory syncytial virus (RSV) starts, characteristically with bronchiolitis presentations, peaking in February to March and tapering off in May. In late March to April, HPiV 3 also appears for 4-6 weeks.

Will 2020-2021 be different?
Summer was remarkably free of expected enterovirus activity, suggesting that the seasonal parade may differ this year. Remember that the 2019-2020 respiratory season suddenly and nearly completely stopped in March because of social distancing and lockdowns needed to address the SARS-CoV-2 pandemic.
The mild influenza season in the southern hemisphere suggests that our influenza season also could be mild. But perhaps not – most southern hemisphere countries that are surveyed for influenza activities had the most intense SARS-CoV-2 mitigations, making the observed mildness potentially related more to social mitigation than less virulent influenza strains. If so, southern hemisphere influenza data may not apply to the United States, where social distancing and masks are ignored or used inconsistently by almost half the population.
Further, the stop-and-go pattern of in-person school/college attendance adds to uncertainties for the usual orderly virus-specific seasonality. The result may be multiple stop-and-go “pop-up” or “mini” outbreaks for any given virus potentially reflected as exaggerated local or regional differences in circulation of various viruses. The erratic seasonality also would increase coinfections, which could present with more severe or different symptoms.
SARS-CoV-2’s potential interaction
Will the relatively mild presentations for most children with SARS-CoV-2 hold up in the setting of coinfections or sequential respiratory viral infections? Could SARS-CoV-2 cause worse/more prolonged symptoms or more sequelae if paired simultaneously or in tandem with a traditional respiratory virus? To date, data on the frequency and severity of SARS-CoV-2 coinfections are conflicting and sparse, but it appears that non-SARS-CoV-2 viruses can be involved in 15%-50% pediatric acute respiratory infections.1,2
However, it may not be important to know about coinfecting viruses other than influenza (can be treated) or SARS-CoV-2 (needs quarantine and contact tracing), unless symptoms are atypical or more severe than usual. For example, a young child with bronchiolitis is most likely infected with RSV, but HPiV, influenza, metapneumovirus, HRv, and even SARS-CoV-2 can cause bronchiolitis. Even so, testing outpatients for RSV or non-influenza is not routine or even clinically helpful. Supportive treatment and restriction from daycare attendance are sufficient management for outpatient ARIs whether presenting as bronchiolitis or not.
Considerations for SARS-CoV-2 testing: Outpatient bronchiolitis
If a child presents with classic bronchiolitis but has above moderate to severe symptoms, is SARS-CoV-2 a consideration? Perhaps, if SARS-CoV-2 acts similarly to non-SARS-CoV-2s.
A recent report from the 30th Multicenter Airway Research Collaboration (MARC-30) surveillance study (2007-2014) of children hospitalized with clinical bronchiolitis evaluated respiratory viruses, including RSV and the four common non-SARS coronaviruses using molecular testing.3 Among 1,880 subjects, a CoV (alpha CoV: NL63 or 229E, or beta CoV: KKU1 or OC43) was detected in 12%. Yet most had only RSV (n = 1,661); 32 had only CoV (n = 32). But note that 219 had both.
Bronchiolitis subjects with CoV were older – median 3.7 (1.4-5.8) vs. 2.8 (1.9-7.2) years – and more likely male than were RSV subjects (68% vs. 58%). OC43 was most frequent followed by equal numbers of HKU1 and NL63, while 229E was the least frequent. Medical utilization and severity did not differ among the CoVs, or between RSV+CoV vs. RSV alone, unless one considered CoV viral load as a variable. ICU use increased when the polymerase chain reaction cycle threshold result indicated a high CoV viral load.
These data suggest CoVs are not infrequent coinfectors with RSV in bronchiolitis – and that SARS-CoV-2 is the same. Therefore, a bronchiolitis presentation doesn’t necessarily take us off the hook for the need to consider SARS-CoV-2 testing, particularly in the somewhat older bronchiolitis patient with more than mild symptoms.
Considerations for SARS-CoV-2 testing: Outpatient influenza-like illness
In 2020-2021, the Centers for Disease Control and Prevention recommends considering empiric antiviral treatment for ILIs (fever plus either cough or sore throat) based upon our clinical judgement, even in non-high-risk children.4
While pediatric COVID-19 illnesses are predominantly asymptomatic or mild, a febrile ARI is also a SARS-CoV-2 compatible presentation. So, if all we use is our clinical judgment, how do we know if the febrile ARI is due to influenza or SARS-CoV-2 or both? At least one study used a highly sensitive and specific molecular influenza test to show that the accuracy of clinically diagnosing influenza in children is not much better than flipping a coin and would lead to potential antiviral overuse.5
So, it seems ideal to test for influenza when possible. Point-of-care (POC) tests are frequently used for outpatients. Eight POC Clinical Laboratory Improvement Amendments (CLIA)–waived kits, some also detecting RSV, are available but most have modest sensitivity (60%-80%) compared with lab-based molecular tests.6 That said, if supplies and kits for one of the POC tests are available to us during these SARS-CoV-2 stressed times (back orders seem more common this year), a positive influenza test in the first 48 hours of symptoms confirms the option to prescribe an antiviral. Yet how will we have confidence that the febrile ARI is not also partly due to SARS-CoV-2? Currently febrile ARIs usually are considered SARS-CoV-2 and the children are sent for SARS-CoV-2 testing. During influenza season, it seems we will need to continue to send febrile outpatients for SARS-CoV-2 testing, even if POC influenza positive, via whatever mechanisms are available as time goes on.
We expect more rapid pediatric testing modalities for SARS-CoV-2 (maybe even saliva tests) to become available over the next months. Indeed, rapid antigen tests and rapid molecular tests are being evaluated in adults and seem destined for CLIA waivers as POC tests, and even home testing kits. Pediatric approvals hopefully also will occur. So, the pathways for SARS-CoV-2 testing available now will likely change over this winter. But be aware that supplies/kits will be prioritized to locations within high need areas and bulk purchase contracts. So POC kits may remain scarce for practices, meaning a reference laboratory still could be the way to go for SARS-CoV-2 for at least the rest of 2020. Reference labs are becoming creative as well; one combined detection of influenza A, influenza B, RSV, and SARS-CoV-2 into one test, and hopes to get approval for swab collection that can be done by families at home and mailed in.
Summary
Expect variations on the traditional parade of seasonal respiratory viruses, with increased numbers of coinfections. Choosing the outpatient who needs influenza testing is the same as in past years, although we have CDC permissive recommendations to prescribe antivirals for any outpatient ILI within the first 48 hours of symptoms. Still, POC testing for influenza remains potentially valuable in the ILI patient. The choice of whether and how to test for SARS-CoV-2 given its potential to be a primary or coinfecting agent in presentations linked more closely to a traditional virus (e.g. RSV bronchiolitis) will be a test of our clinical judgement until more data and easier testing are available. Further complicating coinfection recognition is the fact that many sick visits occur by telehealth and much testing is done at drive-through SARS-CoV-2 testing facilities with no clinician exam. Unless we are liberal in SARS-CoV-2 testing, detecting SARS-CoV-2 coinfections is easier said than done given its usually mild presentation being overshadowed by any coinfecting virus.
But understanding who has SARS-CoV-2, even as a coinfection, still is essential in controlling the pandemic. We will need to be vigilant for evolving approaches to SARS-CoV-2 testing in the context of symptomatic ARI presentations, knowing this will likely remain a moving target for the foreseeable future.
Dr. Harrison is professor of pediatrics and pediatric infectious diseases at Children’s Mercy Hospital-Kansas City, Mo. Children’s Mercy Hospital receives grant funding to study two candidate RSV vaccines. The hospital also receives CDC funding under the New Vaccine Surveillance Network for multicenter surveillance of acute respiratory infections, including influenza, RSV, and parainfluenza virus. Email Dr. Harrison at [email protected].
References
1. Pediatrics. 2020;146(1):e20200961.
2. JAMA. 2020 May 26;323(20):2085-6.
3. Pediatrics. 2020. doi: 10.1542/peds.2020-1267.
4. www.cdc.gov/flu/professionals/antivirals/summary-clinicians.htm.
5. J. Pediatr. 2020. doi: 10.1016/j.jpeds.2020.08.007.
6. www.cdc.gov/flu/professionals/diagnosis/table-nucleic-acid-detection.html.
Dr. Fauci: ‘About 40%-45% of infections are asymptomatic’
Anthony Fauci, MD, highlighting the latest COVID-19 developments on Friday, said, “It is now clear that about 40%-45% of infections are asymptomatic.”
Asymptomatic carriers can account for a large proportion — up to 50% — of virus transmissions, Fauci, director of the National Institute of Allergy and Infectious Diseases, told a virtual crowd of critical care clinicians gathered by the Society of Critical Care Medicine.
Such transmissions have made response strategies, such as contact tracing, extremely difficult, he said.
Lew Kaplan, MD, president of SCCM, told Medscape Medical News after the presentation: “That really supports the universal wearing of masks and the capstone message from that – you should protect one another.
“That kind of social responsibility that sits within the public health domain to me is as important as the vaccine candidates and the science behind the receptors. It underpins the necessary relationship and the interdependence of the medical community with the public,” Kaplan added.
Fauci’s plenary led the SCCM’s conference, “COVID-19: What’s Next/Preparing for the Second Wave,” running today and Saturday.
Why U.S. response lags behind Spain and Italy
“This virus has literally exploded upon the planet in a pandemic manner which is unparalleled to anything we’ve seen in the last 102 years since the pandemic of 1918,” Fauci said.
“Unfortunately, the United States has been hit harder than any other country in the world, with 6 million reported cases.”
He explained that in the European Union countries the disease spiked early on and returned to a low baseline. “Unfortunately for them,” Fauci said, “as they’re trying to open up their economy, it’s coming back up.”
The United States, he explained, plateaued at about 20,000 cases a day, then a surge of cases in Florida, California, Texas, and Arizona brought the cases to 70,000 a day. Now cases have returned to 35,000-40,000 a day.
The difference in the trajectory of the response, he said, is that, compared with Spain and Italy for example, the United States has not shut down mobility in parks, outdoor spaces, and grocery stores nearly as much as some European countries did.
He pointed to numerous clusters of cases, spread from social or work gatherings, including the well-known Skagit County Washington state choir practice in March, in which a symptomatic choir member infected 87% of the 61 people rehearsing.
Vaccine by end of the year
As for a vaccine timeline, Fauci told SCCM members, “We project that by the end of this year, namely November/December, we will know if we have a safe and effective vaccine and we are cautiously optimistic that we will be successful, based on promising data in the animal model as well as good immunological data that we see from the phase 1 and phase 2 trials.”
However, also on Friday, Fauci told MSNBC’s Andrea Mitchell that a sense of normalcy is not likely before the middle of next year.
“By the time you mobilize the distribution of the vaccinations, and you get the majority, or more, of the population vaccinated and protected, that’s likely not going to happen [until] the mid- or end of 2021,” he said.
According to the Centers for Disease Control and Prevention (CDC) case tracker, as of Thursday, COVID-19 had resulted in more than 190,000 deaths overall and more than 256,000 new cases in the United States in the past 7 days.
Fauci has warned that the next few months will be critical in the virus’ trajectory, with the double onslaught of COVID-19 and the flu season.
On Thursday, Fauci said, “We need to hunker down and get through this fall and winter because it’s not going to be easy.”
Fauci remains a top trusted source in COVID-19 information, poll numbers show.
A Kaiser Family Foundation poll released Thursday found that 68% of US adults had a fair amount or a great deal of trust that Fauci would provide reliable information on COVID-19, just slightly more that the 67% who said they trust the CDC information. About half (53%) say they trust Deborah Birx, MD, the coordinator for the White House Coronavirus Task Force, as a reliable source of information.
The poll also found that 54% of Americans said they would not get a COVID-19 vaccine if one was approved by the US Food and Drug Administration before the November election and was made available and free to all who wanted it.
Kaplan and Fauci report no relevant financial relationships.
This article first appeared on Medscape.com.
Anthony Fauci, MD, highlighting the latest COVID-19 developments on Friday, said, “It is now clear that about 40%-45% of infections are asymptomatic.”
Asymptomatic carriers can account for a large proportion — up to 50% — of virus transmissions, Fauci, director of the National Institute of Allergy and Infectious Diseases, told a virtual crowd of critical care clinicians gathered by the Society of Critical Care Medicine.
Such transmissions have made response strategies, such as contact tracing, extremely difficult, he said.
Lew Kaplan, MD, president of SCCM, told Medscape Medical News after the presentation: “That really supports the universal wearing of masks and the capstone message from that – you should protect one another.
“That kind of social responsibility that sits within the public health domain to me is as important as the vaccine candidates and the science behind the receptors. It underpins the necessary relationship and the interdependence of the medical community with the public,” Kaplan added.
Fauci’s plenary led the SCCM’s conference, “COVID-19: What’s Next/Preparing for the Second Wave,” running today and Saturday.
Why U.S. response lags behind Spain and Italy
“This virus has literally exploded upon the planet in a pandemic manner which is unparalleled to anything we’ve seen in the last 102 years since the pandemic of 1918,” Fauci said.
“Unfortunately, the United States has been hit harder than any other country in the world, with 6 million reported cases.”
He explained that in the European Union countries the disease spiked early on and returned to a low baseline. “Unfortunately for them,” Fauci said, “as they’re trying to open up their economy, it’s coming back up.”
The United States, he explained, plateaued at about 20,000 cases a day, then a surge of cases in Florida, California, Texas, and Arizona brought the cases to 70,000 a day. Now cases have returned to 35,000-40,000 a day.
The difference in the trajectory of the response, he said, is that, compared with Spain and Italy for example, the United States has not shut down mobility in parks, outdoor spaces, and grocery stores nearly as much as some European countries did.
He pointed to numerous clusters of cases, spread from social or work gatherings, including the well-known Skagit County Washington state choir practice in March, in which a symptomatic choir member infected 87% of the 61 people rehearsing.
Vaccine by end of the year
As for a vaccine timeline, Fauci told SCCM members, “We project that by the end of this year, namely November/December, we will know if we have a safe and effective vaccine and we are cautiously optimistic that we will be successful, based on promising data in the animal model as well as good immunological data that we see from the phase 1 and phase 2 trials.”
However, also on Friday, Fauci told MSNBC’s Andrea Mitchell that a sense of normalcy is not likely before the middle of next year.
“By the time you mobilize the distribution of the vaccinations, and you get the majority, or more, of the population vaccinated and protected, that’s likely not going to happen [until] the mid- or end of 2021,” he said.
According to the Centers for Disease Control and Prevention (CDC) case tracker, as of Thursday, COVID-19 had resulted in more than 190,000 deaths overall and more than 256,000 new cases in the United States in the past 7 days.
Fauci has warned that the next few months will be critical in the virus’ trajectory, with the double onslaught of COVID-19 and the flu season.
On Thursday, Fauci said, “We need to hunker down and get through this fall and winter because it’s not going to be easy.”
Fauci remains a top trusted source in COVID-19 information, poll numbers show.
A Kaiser Family Foundation poll released Thursday found that 68% of US adults had a fair amount or a great deal of trust that Fauci would provide reliable information on COVID-19, just slightly more that the 67% who said they trust the CDC information. About half (53%) say they trust Deborah Birx, MD, the coordinator for the White House Coronavirus Task Force, as a reliable source of information.
The poll also found that 54% of Americans said they would not get a COVID-19 vaccine if one was approved by the US Food and Drug Administration before the November election and was made available and free to all who wanted it.
Kaplan and Fauci report no relevant financial relationships.
This article first appeared on Medscape.com.
Anthony Fauci, MD, highlighting the latest COVID-19 developments on Friday, said, “It is now clear that about 40%-45% of infections are asymptomatic.”
Asymptomatic carriers can account for a large proportion — up to 50% — of virus transmissions, Fauci, director of the National Institute of Allergy and Infectious Diseases, told a virtual crowd of critical care clinicians gathered by the Society of Critical Care Medicine.
Such transmissions have made response strategies, such as contact tracing, extremely difficult, he said.
Lew Kaplan, MD, president of SCCM, told Medscape Medical News after the presentation: “That really supports the universal wearing of masks and the capstone message from that – you should protect one another.
“That kind of social responsibility that sits within the public health domain to me is as important as the vaccine candidates and the science behind the receptors. It underpins the necessary relationship and the interdependence of the medical community with the public,” Kaplan added.
Fauci’s plenary led the SCCM’s conference, “COVID-19: What’s Next/Preparing for the Second Wave,” running today and Saturday.
Why U.S. response lags behind Spain and Italy
“This virus has literally exploded upon the planet in a pandemic manner which is unparalleled to anything we’ve seen in the last 102 years since the pandemic of 1918,” Fauci said.
“Unfortunately, the United States has been hit harder than any other country in the world, with 6 million reported cases.”
He explained that in the European Union countries the disease spiked early on and returned to a low baseline. “Unfortunately for them,” Fauci said, “as they’re trying to open up their economy, it’s coming back up.”
The United States, he explained, plateaued at about 20,000 cases a day, then a surge of cases in Florida, California, Texas, and Arizona brought the cases to 70,000 a day. Now cases have returned to 35,000-40,000 a day.
The difference in the trajectory of the response, he said, is that, compared with Spain and Italy for example, the United States has not shut down mobility in parks, outdoor spaces, and grocery stores nearly as much as some European countries did.
He pointed to numerous clusters of cases, spread from social or work gatherings, including the well-known Skagit County Washington state choir practice in March, in which a symptomatic choir member infected 87% of the 61 people rehearsing.
Vaccine by end of the year
As for a vaccine timeline, Fauci told SCCM members, “We project that by the end of this year, namely November/December, we will know if we have a safe and effective vaccine and we are cautiously optimistic that we will be successful, based on promising data in the animal model as well as good immunological data that we see from the phase 1 and phase 2 trials.”
However, also on Friday, Fauci told MSNBC’s Andrea Mitchell that a sense of normalcy is not likely before the middle of next year.
“By the time you mobilize the distribution of the vaccinations, and you get the majority, or more, of the population vaccinated and protected, that’s likely not going to happen [until] the mid- or end of 2021,” he said.
According to the Centers for Disease Control and Prevention (CDC) case tracker, as of Thursday, COVID-19 had resulted in more than 190,000 deaths overall and more than 256,000 new cases in the United States in the past 7 days.
Fauci has warned that the next few months will be critical in the virus’ trajectory, with the double onslaught of COVID-19 and the flu season.
On Thursday, Fauci said, “We need to hunker down and get through this fall and winter because it’s not going to be easy.”
Fauci remains a top trusted source in COVID-19 information, poll numbers show.
A Kaiser Family Foundation poll released Thursday found that 68% of US adults had a fair amount or a great deal of trust that Fauci would provide reliable information on COVID-19, just slightly more that the 67% who said they trust the CDC information. About half (53%) say they trust Deborah Birx, MD, the coordinator for the White House Coronavirus Task Force, as a reliable source of information.
The poll also found that 54% of Americans said they would not get a COVID-19 vaccine if one was approved by the US Food and Drug Administration before the November election and was made available and free to all who wanted it.
Kaplan and Fauci report no relevant financial relationships.
This article first appeared on Medscape.com.
Many providers don’t follow hypertension guidelines
Many health care professionals are not following current, evidence-based guidelines to screen for and diagnose hypertension, and appear to have substantial gaps in knowledge, beliefs, and use of recommended practices, results from a large survey suggest.
“One surprising finding was that there was so much trust in the stethoscope, because the automated monitors are a better way to take blood pressure,” lead author Beverly Green, MD, of Kaiser Permanente Washington Health Research Institute, Seattle, said in an interview.
The results of the survey were presented Sept. 10 at the virtual joint scientific sessions of the American Heart Association Council on Hypertension, AHA Council on Kidney in Cardiovascular Disease, and American Society of Hypertension.
The U.S. Preventive Services Task Force (USPSTF) and the American Heart Association/American College of Cardiology recommend out-of-office blood pressure measurements – via ambulatory blood pressure monitoring (ABPM) or home BP monitoring – before making a new diagnosis of hypertension.
To gauge provider knowledge, beliefs, and practices related to BP diagnostic tests, the researchers surveyed 282 providers: 102 medical assistants (MA), 28 licensed practical nurses (LPNs), 33 registered nurses (RNs), 86 primary care physicians, and 33 advanced practitioners (APs).
More than three-quarters of providers (79%) felt that BP measured manually with a stethoscope and ABPM were “very or highly” accurate ways to measure BP when making a new diagnosis of hypertension.
Most did not think that automated clinic BPs, home BP, or kiosk BP measurements were very or highly accurate.
Nearly all providers surveyed (96%) reported that they “always or almost always” rely on clinic BP measurements when diagnosing hypertension, but the majority of physicians/APs would prefer using ABPM (61%) if available.
The problem with ABPM, said Dr. Green, is “it’s just not very available or convenient for patients, and a lot of providers think that patients won’t tolerate it.” Yet, without it, there is a risk for misclassification, she said.
Karen A. Griffin, MD, who chairs the AHA Council on Hypertension, said it became “customary to use clinic BP since ABPM was not previously reimbursed for the routine diagnosis of hypertension.
“Now that the payment for ABPM has been expanded, the number of machines at most institutions is not adequate for the need. Consequently, it will take some time to catch up with the current guidelines for diagnosing hypertension,” she said in an interview.
The provider survey by Dr. Green and colleagues also shows slow uptake of updated thresholds for high blood pressure.
Eighty-four percent of physicians/APs and 68% of MA/LPN/RNs said they used a clinic BP threshold of at least 140/90 mm Hg for making a new diagnosis of hypertension.
Only 3.5% and 9.0%, respectively, reported using the updated threshold of at least 130/80 mm Hg put forth in 2017.
Dr. Griffin said part of this stems from the fact that the survey began before the updated guidelines were released in 2017, “not to mention the fact that some societies have opposed the new threshold of 130/80 mm Hg.”
“I think, with time, the data on morbidity and mortality associated with the goal of 130/80 mm Hg will hopefully convince those who have not yet implemented these new guidelines that it is a safe and effective BP goal,” Dr. Griffin said.
This research had no specific funding. Dr. Green and Dr. Griffin have no relevant disclosures.
A version of this article originally appeared on Medscape.com.
Many health care professionals are not following current, evidence-based guidelines to screen for and diagnose hypertension, and appear to have substantial gaps in knowledge, beliefs, and use of recommended practices, results from a large survey suggest.
“One surprising finding was that there was so much trust in the stethoscope, because the automated monitors are a better way to take blood pressure,” lead author Beverly Green, MD, of Kaiser Permanente Washington Health Research Institute, Seattle, said in an interview.
The results of the survey were presented Sept. 10 at the virtual joint scientific sessions of the American Heart Association Council on Hypertension, AHA Council on Kidney in Cardiovascular Disease, and American Society of Hypertension.
The U.S. Preventive Services Task Force (USPSTF) and the American Heart Association/American College of Cardiology recommend out-of-office blood pressure measurements – via ambulatory blood pressure monitoring (ABPM) or home BP monitoring – before making a new diagnosis of hypertension.
To gauge provider knowledge, beliefs, and practices related to BP diagnostic tests, the researchers surveyed 282 providers: 102 medical assistants (MA), 28 licensed practical nurses (LPNs), 33 registered nurses (RNs), 86 primary care physicians, and 33 advanced practitioners (APs).
More than three-quarters of providers (79%) felt that BP measured manually with a stethoscope and ABPM were “very or highly” accurate ways to measure BP when making a new diagnosis of hypertension.
Most did not think that automated clinic BPs, home BP, or kiosk BP measurements were very or highly accurate.
Nearly all providers surveyed (96%) reported that they “always or almost always” rely on clinic BP measurements when diagnosing hypertension, but the majority of physicians/APs would prefer using ABPM (61%) if available.
The problem with ABPM, said Dr. Green, is “it’s just not very available or convenient for patients, and a lot of providers think that patients won’t tolerate it.” Yet, without it, there is a risk for misclassification, she said.
Karen A. Griffin, MD, who chairs the AHA Council on Hypertension, said it became “customary to use clinic BP since ABPM was not previously reimbursed for the routine diagnosis of hypertension.
“Now that the payment for ABPM has been expanded, the number of machines at most institutions is not adequate for the need. Consequently, it will take some time to catch up with the current guidelines for diagnosing hypertension,” she said in an interview.
The provider survey by Dr. Green and colleagues also shows slow uptake of updated thresholds for high blood pressure.
Eighty-four percent of physicians/APs and 68% of MA/LPN/RNs said they used a clinic BP threshold of at least 140/90 mm Hg for making a new diagnosis of hypertension.
Only 3.5% and 9.0%, respectively, reported using the updated threshold of at least 130/80 mm Hg put forth in 2017.
Dr. Griffin said part of this stems from the fact that the survey began before the updated guidelines were released in 2017, “not to mention the fact that some societies have opposed the new threshold of 130/80 mm Hg.”
“I think, with time, the data on morbidity and mortality associated with the goal of 130/80 mm Hg will hopefully convince those who have not yet implemented these new guidelines that it is a safe and effective BP goal,” Dr. Griffin said.
This research had no specific funding. Dr. Green and Dr. Griffin have no relevant disclosures.
A version of this article originally appeared on Medscape.com.
Many health care professionals are not following current, evidence-based guidelines to screen for and diagnose hypertension, and appear to have substantial gaps in knowledge, beliefs, and use of recommended practices, results from a large survey suggest.
“One surprising finding was that there was so much trust in the stethoscope, because the automated monitors are a better way to take blood pressure,” lead author Beverly Green, MD, of Kaiser Permanente Washington Health Research Institute, Seattle, said in an interview.
The results of the survey were presented Sept. 10 at the virtual joint scientific sessions of the American Heart Association Council on Hypertension, AHA Council on Kidney in Cardiovascular Disease, and American Society of Hypertension.
The U.S. Preventive Services Task Force (USPSTF) and the American Heart Association/American College of Cardiology recommend out-of-office blood pressure measurements – via ambulatory blood pressure monitoring (ABPM) or home BP monitoring – before making a new diagnosis of hypertension.
To gauge provider knowledge, beliefs, and practices related to BP diagnostic tests, the researchers surveyed 282 providers: 102 medical assistants (MA), 28 licensed practical nurses (LPNs), 33 registered nurses (RNs), 86 primary care physicians, and 33 advanced practitioners (APs).
More than three-quarters of providers (79%) felt that BP measured manually with a stethoscope and ABPM were “very or highly” accurate ways to measure BP when making a new diagnosis of hypertension.
Most did not think that automated clinic BPs, home BP, or kiosk BP measurements were very or highly accurate.
Nearly all providers surveyed (96%) reported that they “always or almost always” rely on clinic BP measurements when diagnosing hypertension, but the majority of physicians/APs would prefer using ABPM (61%) if available.
The problem with ABPM, said Dr. Green, is “it’s just not very available or convenient for patients, and a lot of providers think that patients won’t tolerate it.” Yet, without it, there is a risk for misclassification, she said.
Karen A. Griffin, MD, who chairs the AHA Council on Hypertension, said it became “customary to use clinic BP since ABPM was not previously reimbursed for the routine diagnosis of hypertension.
“Now that the payment for ABPM has been expanded, the number of machines at most institutions is not adequate for the need. Consequently, it will take some time to catch up with the current guidelines for diagnosing hypertension,” she said in an interview.
The provider survey by Dr. Green and colleagues also shows slow uptake of updated thresholds for high blood pressure.
Eighty-four percent of physicians/APs and 68% of MA/LPN/RNs said they used a clinic BP threshold of at least 140/90 mm Hg for making a new diagnosis of hypertension.
Only 3.5% and 9.0%, respectively, reported using the updated threshold of at least 130/80 mm Hg put forth in 2017.
Dr. Griffin said part of this stems from the fact that the survey began before the updated guidelines were released in 2017, “not to mention the fact that some societies have opposed the new threshold of 130/80 mm Hg.”
“I think, with time, the data on morbidity and mortality associated with the goal of 130/80 mm Hg will hopefully convince those who have not yet implemented these new guidelines that it is a safe and effective BP goal,” Dr. Griffin said.
This research had no specific funding. Dr. Green and Dr. Griffin have no relevant disclosures.
A version of this article originally appeared on Medscape.com.
Children and COVID-19: New cases may be leveling off
Growth in new pediatric COVID-19 cases has evened out in recent weeks, but children now represent 10% of all COVID-19 cases in the United States, and that measurement has been rising throughout the pandemic, according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.
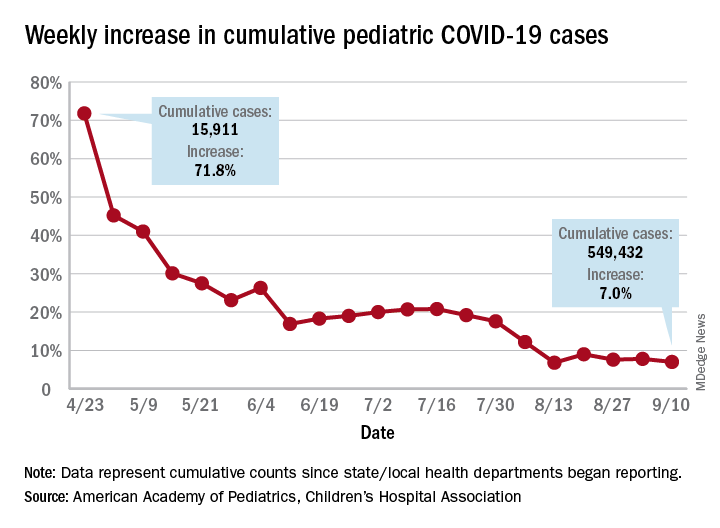
the AAP and the CHA said in the report, based on data from 49 states (New York City is included but not New York state), the District of Columbia, Puerto Rico, and Guam.
The weekly percentage of increase in the number of new cases has not reached double digits since early August and has been no higher than 7.8% over the last 3 weeks. The number of child COVID-19 cases, however, has finally reached 10% of the total for Americans of all ages, which stands at 5.49 million in the jurisdictions included in the report, the AHA and CHA reported.
Measures, however, continue to show low levels of severe illness in children, they noted, including the following:
- Child cases as a proportion of all COVID-19 hospitalizations: 1.7%.
- Hospitalization rate for children: 1.8%.
- Child deaths as a proportion of all deaths: 0.07%.
- Percent of child cases resulting in death: 0.01%.
The number of cumulative cases per 100,000 children is now up to 728.5 nationally, with a range by state that goes from 154.0 in Vermont to 1,670.3 in Tennessee, which is one of only two states reporting cases in those aged 0-20 years as children (the other is South Carolina). The age range for children is 0-17 or 0-19 for most other states, although Florida uses a range of 0-14, the report notes.
Other than Tennessee, there are 10 states with overall rates higher than 1,000 COVID-19 cases per 100,000 children, and there are nine states with cumulative totals over 15,000 cases (California is the highest with just over 75,000), according to the report.
Growth in new pediatric COVID-19 cases has evened out in recent weeks, but children now represent 10% of all COVID-19 cases in the United States, and that measurement has been rising throughout the pandemic, according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.

the AAP and the CHA said in the report, based on data from 49 states (New York City is included but not New York state), the District of Columbia, Puerto Rico, and Guam.
The weekly percentage of increase in the number of new cases has not reached double digits since early August and has been no higher than 7.8% over the last 3 weeks. The number of child COVID-19 cases, however, has finally reached 10% of the total for Americans of all ages, which stands at 5.49 million in the jurisdictions included in the report, the AHA and CHA reported.
Measures, however, continue to show low levels of severe illness in children, they noted, including the following:
- Child cases as a proportion of all COVID-19 hospitalizations: 1.7%.
- Hospitalization rate for children: 1.8%.
- Child deaths as a proportion of all deaths: 0.07%.
- Percent of child cases resulting in death: 0.01%.
The number of cumulative cases per 100,000 children is now up to 728.5 nationally, with a range by state that goes from 154.0 in Vermont to 1,670.3 in Tennessee, which is one of only two states reporting cases in those aged 0-20 years as children (the other is South Carolina). The age range for children is 0-17 or 0-19 for most other states, although Florida uses a range of 0-14, the report notes.
Other than Tennessee, there are 10 states with overall rates higher than 1,000 COVID-19 cases per 100,000 children, and there are nine states with cumulative totals over 15,000 cases (California is the highest with just over 75,000), according to the report.
Growth in new pediatric COVID-19 cases has evened out in recent weeks, but children now represent 10% of all COVID-19 cases in the United States, and that measurement has been rising throughout the pandemic, according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.

the AAP and the CHA said in the report, based on data from 49 states (New York City is included but not New York state), the District of Columbia, Puerto Rico, and Guam.
The weekly percentage of increase in the number of new cases has not reached double digits since early August and has been no higher than 7.8% over the last 3 weeks. The number of child COVID-19 cases, however, has finally reached 10% of the total for Americans of all ages, which stands at 5.49 million in the jurisdictions included in the report, the AHA and CHA reported.
Measures, however, continue to show low levels of severe illness in children, they noted, including the following:
- Child cases as a proportion of all COVID-19 hospitalizations: 1.7%.
- Hospitalization rate for children: 1.8%.
- Child deaths as a proportion of all deaths: 0.07%.
- Percent of child cases resulting in death: 0.01%.
The number of cumulative cases per 100,000 children is now up to 728.5 nationally, with a range by state that goes from 154.0 in Vermont to 1,670.3 in Tennessee, which is one of only two states reporting cases in those aged 0-20 years as children (the other is South Carolina). The age range for children is 0-17 or 0-19 for most other states, although Florida uses a range of 0-14, the report notes.
Other than Tennessee, there are 10 states with overall rates higher than 1,000 COVID-19 cases per 100,000 children, and there are nine states with cumulative totals over 15,000 cases (California is the highest with just over 75,000), according to the report.
Tough to tell COVID from smoke inhalation symptoms — And flu season’s coming
The patients walk into Dr. Melissa Marshall’s community clinics in Northern California with the telltale symptoms. They’re having trouble breathing. It may even hurt to inhale. They’ve got a cough, and the sore throat is definitely there.
A straight case of COVID-19? Not so fast. This is wildfire country.
Up and down the West Coast, hospitals and health facilities are reporting an influx of patients with problems most likely related to smoke inhalation. As fires rage largely uncontrolled amid dry heat and high winds, smoke and ash are billowing and settling on coastal areas like San Francisco and cities and towns hundreds of miles inland as well, turning the sky orange or gray and making even ordinary breathing difficult.
But that, Marshall said, is only part of the challenge.
“Obviously, there’s overlap in the symptoms,” said Marshall, the CEO of CommuniCare, a collection of six clinics in Yolo County, near Sacramento, that treats mostly underinsured and uninsured patients. “Any time someone comes in with even some of those symptoms, we ask ourselves, ‘Is it COVID?’ At the end of the day, clinically speaking, I still want to rule out the virus.”
The protocol is to treat the symptoms, whatever their cause, while recommending that the patient quarantine until test results for the virus come back, she said.
It is a scene playing out in numerous hospitals. Administrators and physicians, finely attuned to COVID-19’s ability to spread quickly and wreak havoc, simply won’t take a chance when they recognize symptoms that could emanate from the virus.
“We’ve seen an increase in patients presenting to the emergency department with respiratory distress,” said Dr. Nanette Mickiewicz, president and CEO of Dominican Hospital in Santa Cruz. “As this can also be a symptom of COVID-19, we’re treating these patients as we would any person under investigation for coronavirus until we can rule them out through our screening process.” During the workup, symptoms that are more specific to COVID-19, like fever, would become apparent.
For the workers at Dominican, the issue moved to the top of the list quickly. Santa Cruz and San Mateo counties have borne the brunt of the CZU Lightning Complex fires, which as of Sept. 10 had burned more than 86,000 acres, destroying 1,100 structures and threatening more than 7,600 others. Nearly a month after they began, the fires were approximately 84% contained, but thousands of people remained evacuated.
Dominican, a Dignity Health hospital, is “open, safe and providing care,” Mickiewicz said. Multiple tents erected outside the building serve as an extension of its ER waiting room. They also are used to perform what has come to be understood as an essential role: separating those with symptoms of COVID-19 from those without.
At the two Solano County hospitals operated by NorthBay Healthcare, the path of some of the wildfires prompted officials to review their evacuation procedures, said spokesperson Steve Huddleston. They ultimately avoided the need to evacuate patients, and new ones arrived with COVID-like symptoms that may actually have been from smoke inhalation.
Huddleston said NorthBay’s intake process “calls for anyone with COVID characteristics to be handled as [a] patient under investigation for COVID, which means they’re separated, screened and managed by staff in special PPE.” At the two hospitals, which have handled nearly 200 COVID cases so far, the protocol is well established.
Hospitals in California, though not under siege in most cases, are dealing with multiple issues they might typically face only sporadically. In Napa County, Adventist Health St. Helena Hospital evacuated 51 patients on a single August night as a fire approached, moving them to 10 other facilities according to their needs and bed space. After a 10-day closure, the hospital was allowed to reopen as evacuation orders were lifted, the fire having been contained some distance away.
The wildfires are also taking a personal toll on health care workers. CommuniCare’s Marshall lost her family’s home in rural Winters, along with 20 acres of olive trees and other plantings that surrounded it, in the Aug. 19 fires that swept through Solano County.
“They called it a ‘firenado,’ ” Marshall said. An apparent confluence of three fires raged out of control, demolishing thousands of acres. With her family safely accounted for and temporary housing arranged by a friend, she returned to work. “Our clinics interact with a very vulnerable population,” she said, “and this is a critical time for them.”
While she pondered how her family would rebuild, the CEO was faced with another immediate crisis: the clinic’s shortage of supplies. Last month, CommuniCare got down to 19 COVID test kits on hand, and ran so low on swabs “that we were literally turning to our veterinary friends for reinforcements,” the doctor said. The clinic’s COVID test results, meanwhile, were taking nearly two weeks to be returned from an overwhelmed outside lab, rendering contact tracing almost useless.
Those situations have been addressed, at least temporarily, Marshall said. But although the West Coast is in the most dangerous time of year for wildfires, generally September to December, another complication for health providers lies on the horizon: flu season.
The Southern Hemisphere, whose influenza trends during our summer months typically predict what’s to come for the U.S., has had very little of the disease this year, presumably because of restricted travel, social distancing and face masks. But it’s too early to be sure what the U.S. flu season will entail.
“You can start to see some cases of the flu in late October,” said Marshall, “and the reality is that it’s going to carry a number of characteristics that could also be symptomatic of COVID. And nothing changes: You have to rule it out, just to eliminate the risk.”
KHN (Kaiser Health News) is a nonprofit news service covering health issues. It is an editorially independent program of KFF (Kaiser Family Foundation), which is not affiliated with Kaiser Permanente. This KHN story first published on California Healthline, a service of the California Health Care Foundation.
The patients walk into Dr. Melissa Marshall’s community clinics in Northern California with the telltale symptoms. They’re having trouble breathing. It may even hurt to inhale. They’ve got a cough, and the sore throat is definitely there.
A straight case of COVID-19? Not so fast. This is wildfire country.
Up and down the West Coast, hospitals and health facilities are reporting an influx of patients with problems most likely related to smoke inhalation. As fires rage largely uncontrolled amid dry heat and high winds, smoke and ash are billowing and settling on coastal areas like San Francisco and cities and towns hundreds of miles inland as well, turning the sky orange or gray and making even ordinary breathing difficult.
But that, Marshall said, is only part of the challenge.
“Obviously, there’s overlap in the symptoms,” said Marshall, the CEO of CommuniCare, a collection of six clinics in Yolo County, near Sacramento, that treats mostly underinsured and uninsured patients. “Any time someone comes in with even some of those symptoms, we ask ourselves, ‘Is it COVID?’ At the end of the day, clinically speaking, I still want to rule out the virus.”
The protocol is to treat the symptoms, whatever their cause, while recommending that the patient quarantine until test results for the virus come back, she said.
It is a scene playing out in numerous hospitals. Administrators and physicians, finely attuned to COVID-19’s ability to spread quickly and wreak havoc, simply won’t take a chance when they recognize symptoms that could emanate from the virus.
“We’ve seen an increase in patients presenting to the emergency department with respiratory distress,” said Dr. Nanette Mickiewicz, president and CEO of Dominican Hospital in Santa Cruz. “As this can also be a symptom of COVID-19, we’re treating these patients as we would any person under investigation for coronavirus until we can rule them out through our screening process.” During the workup, symptoms that are more specific to COVID-19, like fever, would become apparent.
For the workers at Dominican, the issue moved to the top of the list quickly. Santa Cruz and San Mateo counties have borne the brunt of the CZU Lightning Complex fires, which as of Sept. 10 had burned more than 86,000 acres, destroying 1,100 structures and threatening more than 7,600 others. Nearly a month after they began, the fires were approximately 84% contained, but thousands of people remained evacuated.
Dominican, a Dignity Health hospital, is “open, safe and providing care,” Mickiewicz said. Multiple tents erected outside the building serve as an extension of its ER waiting room. They also are used to perform what has come to be understood as an essential role: separating those with symptoms of COVID-19 from those without.
At the two Solano County hospitals operated by NorthBay Healthcare, the path of some of the wildfires prompted officials to review their evacuation procedures, said spokesperson Steve Huddleston. They ultimately avoided the need to evacuate patients, and new ones arrived with COVID-like symptoms that may actually have been from smoke inhalation.
Huddleston said NorthBay’s intake process “calls for anyone with COVID characteristics to be handled as [a] patient under investigation for COVID, which means they’re separated, screened and managed by staff in special PPE.” At the two hospitals, which have handled nearly 200 COVID cases so far, the protocol is well established.
Hospitals in California, though not under siege in most cases, are dealing with multiple issues they might typically face only sporadically. In Napa County, Adventist Health St. Helena Hospital evacuated 51 patients on a single August night as a fire approached, moving them to 10 other facilities according to their needs and bed space. After a 10-day closure, the hospital was allowed to reopen as evacuation orders were lifted, the fire having been contained some distance away.
The wildfires are also taking a personal toll on health care workers. CommuniCare’s Marshall lost her family’s home in rural Winters, along with 20 acres of olive trees and other plantings that surrounded it, in the Aug. 19 fires that swept through Solano County.
“They called it a ‘firenado,’ ” Marshall said. An apparent confluence of three fires raged out of control, demolishing thousands of acres. With her family safely accounted for and temporary housing arranged by a friend, she returned to work. “Our clinics interact with a very vulnerable population,” she said, “and this is a critical time for them.”
While she pondered how her family would rebuild, the CEO was faced with another immediate crisis: the clinic’s shortage of supplies. Last month, CommuniCare got down to 19 COVID test kits on hand, and ran so low on swabs “that we were literally turning to our veterinary friends for reinforcements,” the doctor said. The clinic’s COVID test results, meanwhile, were taking nearly two weeks to be returned from an overwhelmed outside lab, rendering contact tracing almost useless.
Those situations have been addressed, at least temporarily, Marshall said. But although the West Coast is in the most dangerous time of year for wildfires, generally September to December, another complication for health providers lies on the horizon: flu season.
The Southern Hemisphere, whose influenza trends during our summer months typically predict what’s to come for the U.S., has had very little of the disease this year, presumably because of restricted travel, social distancing and face masks. But it’s too early to be sure what the U.S. flu season will entail.
“You can start to see some cases of the flu in late October,” said Marshall, “and the reality is that it’s going to carry a number of characteristics that could also be symptomatic of COVID. And nothing changes: You have to rule it out, just to eliminate the risk.”
KHN (Kaiser Health News) is a nonprofit news service covering health issues. It is an editorially independent program of KFF (Kaiser Family Foundation), which is not affiliated with Kaiser Permanente. This KHN story first published on California Healthline, a service of the California Health Care Foundation.
The patients walk into Dr. Melissa Marshall’s community clinics in Northern California with the telltale symptoms. They’re having trouble breathing. It may even hurt to inhale. They’ve got a cough, and the sore throat is definitely there.
A straight case of COVID-19? Not so fast. This is wildfire country.
Up and down the West Coast, hospitals and health facilities are reporting an influx of patients with problems most likely related to smoke inhalation. As fires rage largely uncontrolled amid dry heat and high winds, smoke and ash are billowing and settling on coastal areas like San Francisco and cities and towns hundreds of miles inland as well, turning the sky orange or gray and making even ordinary breathing difficult.
But that, Marshall said, is only part of the challenge.
“Obviously, there’s overlap in the symptoms,” said Marshall, the CEO of CommuniCare, a collection of six clinics in Yolo County, near Sacramento, that treats mostly underinsured and uninsured patients. “Any time someone comes in with even some of those symptoms, we ask ourselves, ‘Is it COVID?’ At the end of the day, clinically speaking, I still want to rule out the virus.”
The protocol is to treat the symptoms, whatever their cause, while recommending that the patient quarantine until test results for the virus come back, she said.
It is a scene playing out in numerous hospitals. Administrators and physicians, finely attuned to COVID-19’s ability to spread quickly and wreak havoc, simply won’t take a chance when they recognize symptoms that could emanate from the virus.
“We’ve seen an increase in patients presenting to the emergency department with respiratory distress,” said Dr. Nanette Mickiewicz, president and CEO of Dominican Hospital in Santa Cruz. “As this can also be a symptom of COVID-19, we’re treating these patients as we would any person under investigation for coronavirus until we can rule them out through our screening process.” During the workup, symptoms that are more specific to COVID-19, like fever, would become apparent.
For the workers at Dominican, the issue moved to the top of the list quickly. Santa Cruz and San Mateo counties have borne the brunt of the CZU Lightning Complex fires, which as of Sept. 10 had burned more than 86,000 acres, destroying 1,100 structures and threatening more than 7,600 others. Nearly a month after they began, the fires were approximately 84% contained, but thousands of people remained evacuated.
Dominican, a Dignity Health hospital, is “open, safe and providing care,” Mickiewicz said. Multiple tents erected outside the building serve as an extension of its ER waiting room. They also are used to perform what has come to be understood as an essential role: separating those with symptoms of COVID-19 from those without.
At the two Solano County hospitals operated by NorthBay Healthcare, the path of some of the wildfires prompted officials to review their evacuation procedures, said spokesperson Steve Huddleston. They ultimately avoided the need to evacuate patients, and new ones arrived with COVID-like symptoms that may actually have been from smoke inhalation.
Huddleston said NorthBay’s intake process “calls for anyone with COVID characteristics to be handled as [a] patient under investigation for COVID, which means they’re separated, screened and managed by staff in special PPE.” At the two hospitals, which have handled nearly 200 COVID cases so far, the protocol is well established.
Hospitals in California, though not under siege in most cases, are dealing with multiple issues they might typically face only sporadically. In Napa County, Adventist Health St. Helena Hospital evacuated 51 patients on a single August night as a fire approached, moving them to 10 other facilities according to their needs and bed space. After a 10-day closure, the hospital was allowed to reopen as evacuation orders were lifted, the fire having been contained some distance away.
The wildfires are also taking a personal toll on health care workers. CommuniCare’s Marshall lost her family’s home in rural Winters, along with 20 acres of olive trees and other plantings that surrounded it, in the Aug. 19 fires that swept through Solano County.
“They called it a ‘firenado,’ ” Marshall said. An apparent confluence of three fires raged out of control, demolishing thousands of acres. With her family safely accounted for and temporary housing arranged by a friend, she returned to work. “Our clinics interact with a very vulnerable population,” she said, “and this is a critical time for them.”
While she pondered how her family would rebuild, the CEO was faced with another immediate crisis: the clinic’s shortage of supplies. Last month, CommuniCare got down to 19 COVID test kits on hand, and ran so low on swabs “that we were literally turning to our veterinary friends for reinforcements,” the doctor said. The clinic’s COVID test results, meanwhile, were taking nearly two weeks to be returned from an overwhelmed outside lab, rendering contact tracing almost useless.
Those situations have been addressed, at least temporarily, Marshall said. But although the West Coast is in the most dangerous time of year for wildfires, generally September to December, another complication for health providers lies on the horizon: flu season.
The Southern Hemisphere, whose influenza trends during our summer months typically predict what’s to come for the U.S., has had very little of the disease this year, presumably because of restricted travel, social distancing and face masks. But it’s too early to be sure what the U.S. flu season will entail.
“You can start to see some cases of the flu in late October,” said Marshall, “and the reality is that it’s going to carry a number of characteristics that could also be symptomatic of COVID. And nothing changes: You have to rule it out, just to eliminate the risk.”
KHN (Kaiser Health News) is a nonprofit news service covering health issues. It is an editorially independent program of KFF (Kaiser Family Foundation), which is not affiliated with Kaiser Permanente. This KHN story first published on California Healthline, a service of the California Health Care Foundation.
COVID-19 outcomes no worse in patients on TNF inhibitors or methotrexate
Continued use of tumor necrosis factor inhibitors or methotrexate is acceptable in most patients who acquire COVID-19, results of a recent cohort study suggest.
Among patients on tumor necrosis factor inhibitors (TNFi) or methotrexate who developed COVID-19, death and hospitalization rates were similar to matched COVID-19 patients not on those medications, according to authors of the multicenter research network study.
Reassuringly, likelihood of hospitalization and mortality were not significantly different between 214 patients with COVID-19 taking TNFi or methotrexate and 31,862 matched COVID-19 patients not on those medications, according to the investigators, whose findings were published recently in the Journal of the American Academy of Dermatology.
Zachary Zinn, MD, corresponding author on the study, said in an interview that the findings suggest these medicines can be safely continued in the majority of patients taking them during the COVID-19 pandemic.
“If you’re a prescribing physician who’s giving patients TNF inhibitors or methotrexate or both, I think you can comfortably tell your patients there is good data that these do not lead to worse outcomes if you get COVID-19,” said Dr. Zinn, associate professor in the department of dermatology at West Virginia University, Morgantown.
The findings from these researchers corroborate a growing body of evidence suggesting that immunosuppressive treatments can be continued in patients with dermatologic and rheumatic conditions.
In recent guidance from the National Psoriasis Foundation, released Sept. 4, an expert consensus panel cited 15 studies that they said suggested that treatments for psoriasis or psoriatic arthritis “do not meaningfully alter the risk of acquiring SARS-CoV-2 infection or having worse COVID-19 outcomes.”
That said, the data to date are mainly from small case series and registry studies based on spontaneously reported COVID-19 cases, which suggests a continued need for shared decision making. In addition, chronic systemic corticosteroids should be avoided for management of psoriatic arthritis, the guidance states, based on rheumatology and gastroenterology literature suggesting this treatment is linked to worse COVID-19 outcomes.
In the interview, Dr. Zinn noted that some previous studies of immunosuppressive treatments in patients who acquire COVID-19 have aggregated data on numerous classes of biologic medications, lessening the strength of data for each specific medication.
“By focusing specifically on TNF inhibitors and methotrexate, this study gives better guidance to prescribers of these medications,” he said.
To see whether TNFi or methotrexate increased risk of worsened COVID-19 outcomes, Dr. Zinn and coinvestigators evaluated data from TriNetX, a research network that includes approximately 53 million unique patient records, predominantly in the United States.
They identified 32,076 adult patients with COVID-19, of whom 214 had recent exposure to TNFi or methotrexate. The patients in the TNFi/methotrexate group were similar in age to those without exposure to those drugs, at 55.1 versus 53.2 years, respectively. However, patients in the drug exposure group were more frequently White, female, and had substantially more comorbidities, including diabetes and obesity, according to the investigators.
Nevertheless, the likelihood of hospitalization was not statistically different in the TNFi/methotrexate group versus the non-TNFi/methotrexate group, with a risk ratio of 0.91 (95% confidence interval, 0.68-1.22; P = .5260).
Likewise, the likelihood of death was not different between groups, with a RR of 0.87 (95% CI, 0.42-1.78; P = .6958). Looking at subgroups of patients exposed to TNFi or methotrexate only didn’t change the results, the investigators added.
Taken together, the findings argue against interruption of these treatments because of the fear of the possibly worse COVID-19 outcomes, the investigators concluded, although they emphasized the need for more research.
“Because the COVID-19 pandemic is ongoing, there is a desperate need for evidence-based data on biologic and immunomodulator exposure in the setting of COVID-19 infection,” they wrote.
Dr. Zinn and coauthors reported no conflicts of interest and no funding sources related to the study.
SOURCE: Zinn Z et al. J Am Acad Dermatol. 2020 Sep 11. doi: 10.1016/j.jaad.2020.09.009.
Continued use of tumor necrosis factor inhibitors or methotrexate is acceptable in most patients who acquire COVID-19, results of a recent cohort study suggest.
Among patients on tumor necrosis factor inhibitors (TNFi) or methotrexate who developed COVID-19, death and hospitalization rates were similar to matched COVID-19 patients not on those medications, according to authors of the multicenter research network study.
Reassuringly, likelihood of hospitalization and mortality were not significantly different between 214 patients with COVID-19 taking TNFi or methotrexate and 31,862 matched COVID-19 patients not on those medications, according to the investigators, whose findings were published recently in the Journal of the American Academy of Dermatology.
Zachary Zinn, MD, corresponding author on the study, said in an interview that the findings suggest these medicines can be safely continued in the majority of patients taking them during the COVID-19 pandemic.
“If you’re a prescribing physician who’s giving patients TNF inhibitors or methotrexate or both, I think you can comfortably tell your patients there is good data that these do not lead to worse outcomes if you get COVID-19,” said Dr. Zinn, associate professor in the department of dermatology at West Virginia University, Morgantown.
The findings from these researchers corroborate a growing body of evidence suggesting that immunosuppressive treatments can be continued in patients with dermatologic and rheumatic conditions.
In recent guidance from the National Psoriasis Foundation, released Sept. 4, an expert consensus panel cited 15 studies that they said suggested that treatments for psoriasis or psoriatic arthritis “do not meaningfully alter the risk of acquiring SARS-CoV-2 infection or having worse COVID-19 outcomes.”
That said, the data to date are mainly from small case series and registry studies based on spontaneously reported COVID-19 cases, which suggests a continued need for shared decision making. In addition, chronic systemic corticosteroids should be avoided for management of psoriatic arthritis, the guidance states, based on rheumatology and gastroenterology literature suggesting this treatment is linked to worse COVID-19 outcomes.
In the interview, Dr. Zinn noted that some previous studies of immunosuppressive treatments in patients who acquire COVID-19 have aggregated data on numerous classes of biologic medications, lessening the strength of data for each specific medication.
“By focusing specifically on TNF inhibitors and methotrexate, this study gives better guidance to prescribers of these medications,” he said.
To see whether TNFi or methotrexate increased risk of worsened COVID-19 outcomes, Dr. Zinn and coinvestigators evaluated data from TriNetX, a research network that includes approximately 53 million unique patient records, predominantly in the United States.
They identified 32,076 adult patients with COVID-19, of whom 214 had recent exposure to TNFi or methotrexate. The patients in the TNFi/methotrexate group were similar in age to those without exposure to those drugs, at 55.1 versus 53.2 years, respectively. However, patients in the drug exposure group were more frequently White, female, and had substantially more comorbidities, including diabetes and obesity, according to the investigators.
Nevertheless, the likelihood of hospitalization was not statistically different in the TNFi/methotrexate group versus the non-TNFi/methotrexate group, with a risk ratio of 0.91 (95% confidence interval, 0.68-1.22; P = .5260).
Likewise, the likelihood of death was not different between groups, with a RR of 0.87 (95% CI, 0.42-1.78; P = .6958). Looking at subgroups of patients exposed to TNFi or methotrexate only didn’t change the results, the investigators added.
Taken together, the findings argue against interruption of these treatments because of the fear of the possibly worse COVID-19 outcomes, the investigators concluded, although they emphasized the need for more research.
“Because the COVID-19 pandemic is ongoing, there is a desperate need for evidence-based data on biologic and immunomodulator exposure in the setting of COVID-19 infection,” they wrote.
Dr. Zinn and coauthors reported no conflicts of interest and no funding sources related to the study.
SOURCE: Zinn Z et al. J Am Acad Dermatol. 2020 Sep 11. doi: 10.1016/j.jaad.2020.09.009.
Continued use of tumor necrosis factor inhibitors or methotrexate is acceptable in most patients who acquire COVID-19, results of a recent cohort study suggest.
Among patients on tumor necrosis factor inhibitors (TNFi) or methotrexate who developed COVID-19, death and hospitalization rates were similar to matched COVID-19 patients not on those medications, according to authors of the multicenter research network study.
Reassuringly, likelihood of hospitalization and mortality were not significantly different between 214 patients with COVID-19 taking TNFi or methotrexate and 31,862 matched COVID-19 patients not on those medications, according to the investigators, whose findings were published recently in the Journal of the American Academy of Dermatology.
Zachary Zinn, MD, corresponding author on the study, said in an interview that the findings suggest these medicines can be safely continued in the majority of patients taking them during the COVID-19 pandemic.
“If you’re a prescribing physician who’s giving patients TNF inhibitors or methotrexate or both, I think you can comfortably tell your patients there is good data that these do not lead to worse outcomes if you get COVID-19,” said Dr. Zinn, associate professor in the department of dermatology at West Virginia University, Morgantown.
The findings from these researchers corroborate a growing body of evidence suggesting that immunosuppressive treatments can be continued in patients with dermatologic and rheumatic conditions.
In recent guidance from the National Psoriasis Foundation, released Sept. 4, an expert consensus panel cited 15 studies that they said suggested that treatments for psoriasis or psoriatic arthritis “do not meaningfully alter the risk of acquiring SARS-CoV-2 infection or having worse COVID-19 outcomes.”
That said, the data to date are mainly from small case series and registry studies based on spontaneously reported COVID-19 cases, which suggests a continued need for shared decision making. In addition, chronic systemic corticosteroids should be avoided for management of psoriatic arthritis, the guidance states, based on rheumatology and gastroenterology literature suggesting this treatment is linked to worse COVID-19 outcomes.
In the interview, Dr. Zinn noted that some previous studies of immunosuppressive treatments in patients who acquire COVID-19 have aggregated data on numerous classes of biologic medications, lessening the strength of data for each specific medication.
“By focusing specifically on TNF inhibitors and methotrexate, this study gives better guidance to prescribers of these medications,” he said.
To see whether TNFi or methotrexate increased risk of worsened COVID-19 outcomes, Dr. Zinn and coinvestigators evaluated data from TriNetX, a research network that includes approximately 53 million unique patient records, predominantly in the United States.
They identified 32,076 adult patients with COVID-19, of whom 214 had recent exposure to TNFi or methotrexate. The patients in the TNFi/methotrexate group were similar in age to those without exposure to those drugs, at 55.1 versus 53.2 years, respectively. However, patients in the drug exposure group were more frequently White, female, and had substantially more comorbidities, including diabetes and obesity, according to the investigators.
Nevertheless, the likelihood of hospitalization was not statistically different in the TNFi/methotrexate group versus the non-TNFi/methotrexate group, with a risk ratio of 0.91 (95% confidence interval, 0.68-1.22; P = .5260).
Likewise, the likelihood of death was not different between groups, with a RR of 0.87 (95% CI, 0.42-1.78; P = .6958). Looking at subgroups of patients exposed to TNFi or methotrexate only didn’t change the results, the investigators added.
Taken together, the findings argue against interruption of these treatments because of the fear of the possibly worse COVID-19 outcomes, the investigators concluded, although they emphasized the need for more research.
“Because the COVID-19 pandemic is ongoing, there is a desperate need for evidence-based data on biologic and immunomodulator exposure in the setting of COVID-19 infection,” they wrote.
Dr. Zinn and coauthors reported no conflicts of interest and no funding sources related to the study.
SOURCE: Zinn Z et al. J Am Acad Dermatol. 2020 Sep 11. doi: 10.1016/j.jaad.2020.09.009.
FROM THE JOURNAL OF THE AMERICAN ACADEMY OF DERMATOLOGY
Physician income drops, burnout spikes globally in pandemic
according to the results of a Medscape survey.
More than 7,500 physicians – nearly 5,000 in the United States, and others in Brazil, France, Germany, Mexico, Portugal, Spain, and the United Kingdom – responded to questions about their struggles to save patients and how the pandemic has changed their income and their lives at home and at work.
The pain was evident in this response from an emergency medicine physician in Spain: “It has been the worst time in my life ever, in both my personal and professional life.”
Conversely, some reported positive effects.
An internist in Brazil wrote: “I feel more proud of my career than ever before.”
One quarter of U.S. physicians considering earlier retirement
Physicians in the United States were asked what career changes, if any, they were considering in light of their experience with COVID-19. Although a little more than half (51%) said they were not planning any changes, 25% answered, “retiring earlier than previously planned,” and 12% answered, “a career change away from medicine.”
The number of physicians reporting an income drop was highest in Brazil (63% reported a drop), followed by the United States (62%), Mexico (56%), Portugal (49%), Germany (42%), France (41%), and Spain (31%). The question was not asked in the United Kingdom survey.
In the United States, the size of the drop has been substantial: 9% lost 76%-100% of their income; 14% lost 51%-75%; 28% lost 26%-50%; 33% lost 11%-25%; and 15% lost 1%-10%.
The U.S. specialists with the largest drop in income were ophthalmologists, who lost 51%, followed by allergists (46%), plastic surgeons (46%), and otolaryngologists (45%).
“I’m looking for a new profession due to economic impact,” an otolaryngologist in the United States said. “We are at risk while essentially using our private savings to keep our practice solvent.”
More than half of U.S. physicians (54%) have personally treated patients with COVID-19. Percentages were higher in France, Spain, and the United Kingdom (percentages ranged from 60%-68%).
The United States led all eight countries in treating patients with COVID-19 via telemedicine, at 26%. Germany had the lowest telemedicine percentage, at 10%.
Burnout intensifies
About two thirds of US physicians (64%) said that burnout had intensified during the crisis (70% of female physicians and 61% of male physicians said it had).
Many factors are feeding the burnout.
A critical care physician in the United States responded, “It is terrible to see people arriving at their rooms and assuming they were going to die soon; to see people saying goodbye to their families before dying or before being intubated.”
In all eight countries, a substantial percentage of physicians reported they “sometimes, often or always” treated patients with COVID-19 without the proper personal protective equipment. Spain had by far the largest percentage who answered that way (67%), followed by France (45%), Mexico (40%), the United Kingdom (34%), Brazil and Germany (28% each); and the United States and Portugal (23% each).
A U.S. rheumatologist wrote: “The fact that we were sent to take care of infectious patients without proper protection equipment made me feel we were betrayed in this fight.”
Sense of duty to volunteer to treat COVID-19 patients varied substantially among countries, from 69% who felt that way in Spain to 40% in Brazil. Half (50%) in the United States felt that way.
“Altruism must take second place where a real and present threat exists to my own personal existence,” one U.S. internist wrote.
Numbers personally infected
One fifth of physicians in Spain and the United Kingdom had personally been infected with the virus. Brazil, France, and Mexico had the next highest numbers, with 13%-15% of physicians infected; 5%-6% in the United States, Germany, and Portugal said they had been infected.
The percentage of physicians who reported that immediate family members had been infected ranged from 25% in Spain to 6% in Portugal. Among US physicians, 9% reported that family members had been diagnosed with COVID-19.
In the United States, 44% of respondents who had family living with them at home during the pandemic reported that relationships at home were more stressed because of stay-at-home guidelines and social distancing. Almost half (47%) said there had been no change, and 9% said relationships were less stressed.
Eating is coping mechanism of choice
Physicians were asked what they were doing more of during the pandemic, and food seemed to be the top source of comfort in all eight countries.
Loneliness reports differ across globe
Portugal had the highest percentage (51%) of physicians reporting increased loneliness. Next were Brazil (48%), the United States (46%), the United Kingdom (42%), France (41%), Spain and Mexico (40% each), and Germany (32%).
All eight countries lacked workplace activities to help physicians with grief. More than half (55%) of U.K. physicians reported having such activities available at their workplace, whereas only 25% of physicians in Germany did; 12%-24% of respondents across the countries were unsure about the offerings.
This article first appeared on Medscape.com.
according to the results of a Medscape survey.
More than 7,500 physicians – nearly 5,000 in the United States, and others in Brazil, France, Germany, Mexico, Portugal, Spain, and the United Kingdom – responded to questions about their struggles to save patients and how the pandemic has changed their income and their lives at home and at work.
The pain was evident in this response from an emergency medicine physician in Spain: “It has been the worst time in my life ever, in both my personal and professional life.”
Conversely, some reported positive effects.
An internist in Brazil wrote: “I feel more proud of my career than ever before.”
One quarter of U.S. physicians considering earlier retirement
Physicians in the United States were asked what career changes, if any, they were considering in light of their experience with COVID-19. Although a little more than half (51%) said they were not planning any changes, 25% answered, “retiring earlier than previously planned,” and 12% answered, “a career change away from medicine.”
The number of physicians reporting an income drop was highest in Brazil (63% reported a drop), followed by the United States (62%), Mexico (56%), Portugal (49%), Germany (42%), France (41%), and Spain (31%). The question was not asked in the United Kingdom survey.
In the United States, the size of the drop has been substantial: 9% lost 76%-100% of their income; 14% lost 51%-75%; 28% lost 26%-50%; 33% lost 11%-25%; and 15% lost 1%-10%.
The U.S. specialists with the largest drop in income were ophthalmologists, who lost 51%, followed by allergists (46%), plastic surgeons (46%), and otolaryngologists (45%).
“I’m looking for a new profession due to economic impact,” an otolaryngologist in the United States said. “We are at risk while essentially using our private savings to keep our practice solvent.”
More than half of U.S. physicians (54%) have personally treated patients with COVID-19. Percentages were higher in France, Spain, and the United Kingdom (percentages ranged from 60%-68%).
The United States led all eight countries in treating patients with COVID-19 via telemedicine, at 26%. Germany had the lowest telemedicine percentage, at 10%.
Burnout intensifies
About two thirds of US physicians (64%) said that burnout had intensified during the crisis (70% of female physicians and 61% of male physicians said it had).
Many factors are feeding the burnout.
A critical care physician in the United States responded, “It is terrible to see people arriving at their rooms and assuming they were going to die soon; to see people saying goodbye to their families before dying or before being intubated.”
In all eight countries, a substantial percentage of physicians reported they “sometimes, often or always” treated patients with COVID-19 without the proper personal protective equipment. Spain had by far the largest percentage who answered that way (67%), followed by France (45%), Mexico (40%), the United Kingdom (34%), Brazil and Germany (28% each); and the United States and Portugal (23% each).
A U.S. rheumatologist wrote: “The fact that we were sent to take care of infectious patients without proper protection equipment made me feel we were betrayed in this fight.”
Sense of duty to volunteer to treat COVID-19 patients varied substantially among countries, from 69% who felt that way in Spain to 40% in Brazil. Half (50%) in the United States felt that way.
“Altruism must take second place where a real and present threat exists to my own personal existence,” one U.S. internist wrote.
Numbers personally infected
One fifth of physicians in Spain and the United Kingdom had personally been infected with the virus. Brazil, France, and Mexico had the next highest numbers, with 13%-15% of physicians infected; 5%-6% in the United States, Germany, and Portugal said they had been infected.
The percentage of physicians who reported that immediate family members had been infected ranged from 25% in Spain to 6% in Portugal. Among US physicians, 9% reported that family members had been diagnosed with COVID-19.
In the United States, 44% of respondents who had family living with them at home during the pandemic reported that relationships at home were more stressed because of stay-at-home guidelines and social distancing. Almost half (47%) said there had been no change, and 9% said relationships were less stressed.
Eating is coping mechanism of choice
Physicians were asked what they were doing more of during the pandemic, and food seemed to be the top source of comfort in all eight countries.
Loneliness reports differ across globe
Portugal had the highest percentage (51%) of physicians reporting increased loneliness. Next were Brazil (48%), the United States (46%), the United Kingdom (42%), France (41%), Spain and Mexico (40% each), and Germany (32%).
All eight countries lacked workplace activities to help physicians with grief. More than half (55%) of U.K. physicians reported having such activities available at their workplace, whereas only 25% of physicians in Germany did; 12%-24% of respondents across the countries were unsure about the offerings.
This article first appeared on Medscape.com.
according to the results of a Medscape survey.
More than 7,500 physicians – nearly 5,000 in the United States, and others in Brazil, France, Germany, Mexico, Portugal, Spain, and the United Kingdom – responded to questions about their struggles to save patients and how the pandemic has changed their income and their lives at home and at work.
The pain was evident in this response from an emergency medicine physician in Spain: “It has been the worst time in my life ever, in both my personal and professional life.”
Conversely, some reported positive effects.
An internist in Brazil wrote: “I feel more proud of my career than ever before.”
One quarter of U.S. physicians considering earlier retirement
Physicians in the United States were asked what career changes, if any, they were considering in light of their experience with COVID-19. Although a little more than half (51%) said they were not planning any changes, 25% answered, “retiring earlier than previously planned,” and 12% answered, “a career change away from medicine.”
The number of physicians reporting an income drop was highest in Brazil (63% reported a drop), followed by the United States (62%), Mexico (56%), Portugal (49%), Germany (42%), France (41%), and Spain (31%). The question was not asked in the United Kingdom survey.
In the United States, the size of the drop has been substantial: 9% lost 76%-100% of their income; 14% lost 51%-75%; 28% lost 26%-50%; 33% lost 11%-25%; and 15% lost 1%-10%.
The U.S. specialists with the largest drop in income were ophthalmologists, who lost 51%, followed by allergists (46%), plastic surgeons (46%), and otolaryngologists (45%).
“I’m looking for a new profession due to economic impact,” an otolaryngologist in the United States said. “We are at risk while essentially using our private savings to keep our practice solvent.”
More than half of U.S. physicians (54%) have personally treated patients with COVID-19. Percentages were higher in France, Spain, and the United Kingdom (percentages ranged from 60%-68%).
The United States led all eight countries in treating patients with COVID-19 via telemedicine, at 26%. Germany had the lowest telemedicine percentage, at 10%.
Burnout intensifies
About two thirds of US physicians (64%) said that burnout had intensified during the crisis (70% of female physicians and 61% of male physicians said it had).
Many factors are feeding the burnout.
A critical care physician in the United States responded, “It is terrible to see people arriving at their rooms and assuming they were going to die soon; to see people saying goodbye to their families before dying or before being intubated.”
In all eight countries, a substantial percentage of physicians reported they “sometimes, often or always” treated patients with COVID-19 without the proper personal protective equipment. Spain had by far the largest percentage who answered that way (67%), followed by France (45%), Mexico (40%), the United Kingdom (34%), Brazil and Germany (28% each); and the United States and Portugal (23% each).
A U.S. rheumatologist wrote: “The fact that we were sent to take care of infectious patients without proper protection equipment made me feel we were betrayed in this fight.”
Sense of duty to volunteer to treat COVID-19 patients varied substantially among countries, from 69% who felt that way in Spain to 40% in Brazil. Half (50%) in the United States felt that way.
“Altruism must take second place where a real and present threat exists to my own personal existence,” one U.S. internist wrote.
Numbers personally infected
One fifth of physicians in Spain and the United Kingdom had personally been infected with the virus. Brazil, France, and Mexico had the next highest numbers, with 13%-15% of physicians infected; 5%-6% in the United States, Germany, and Portugal said they had been infected.
The percentage of physicians who reported that immediate family members had been infected ranged from 25% in Spain to 6% in Portugal. Among US physicians, 9% reported that family members had been diagnosed with COVID-19.
In the United States, 44% of respondents who had family living with them at home during the pandemic reported that relationships at home were more stressed because of stay-at-home guidelines and social distancing. Almost half (47%) said there had been no change, and 9% said relationships were less stressed.
Eating is coping mechanism of choice
Physicians were asked what they were doing more of during the pandemic, and food seemed to be the top source of comfort in all eight countries.
Loneliness reports differ across globe
Portugal had the highest percentage (51%) of physicians reporting increased loneliness. Next were Brazil (48%), the United States (46%), the United Kingdom (42%), France (41%), Spain and Mexico (40% each), and Germany (32%).
All eight countries lacked workplace activities to help physicians with grief. More than half (55%) of U.K. physicians reported having such activities available at their workplace, whereas only 25% of physicians in Germany did; 12%-24% of respondents across the countries were unsure about the offerings.
This article first appeared on Medscape.com.
Infectious COVID-19 can persist in gut for weeks
Stool tests were positive among people with no GI symptoms, and in some cases up to 6 days after nasopharyngeal swabs yielded negative results.
The small pilot study suggests a quiescent but active infection in the gut. Stool testing revealed genomic evidence of active infection in 7 of the 15 participants tested in one of two hospitals in Hong Kong.
“We found active and prolonged SARS-CoV-2 infection in the stool of patients with COVID-19, even after recovery, suggesting that coronavirus could remain in the gut of asymptomatic carriers,” senior author Siew C. Ng, MBBS, PhD, told Medscape Medical News.
“Due to the potential threat of fecal-oral transmission, it is important to maintain long-term coronavirus and health surveillance,” said Ng, Associate Director of the Centre for Gut Microbiota Research at the Chinese University of Hong Kong (CUHK).
“Discharged patients and their caretakers should remain vigilant and observe strict personal and toileting hygiene,” she added.
The prospective, observational study was published online July 20 in Gut.
Ramping up COVID-19 testing
As a follow-up to these and other findings – including the testing of more than 2,000 stool samples in children and the needy arriving at Hong Kong airports starting March 29 – the same investigators are establishing a CUHK Coronavirus Testing Center.
As of Aug. 31, the detection rate in tested children was 0.28%. The Center plans to offer as many as 2,000 COVID-19 tests daily going forward to help identify asymptomatic carriers, the investigators announced in a Sept. 7 news release.
In contrast to nasopharyngeal sampling, stool specimens are “more convenient, safe and non-invasive to collect in the pediatric population,” professor Paul Chan, chairman of the Department of Microbiology, CU Medicine, said in the release. “This makes the stool test a better option for COVID-19 screening in babies, young children and those whose respiratory samples are difficult to collect.”
Even though previous researchers identified SARS-CoV-2 in the stool, the activity and infectivity of the virus in the gastrointestinal tract during and after COVID-19 respiratory positivity remained largely unknown.
Active infection detected in stool
This prospective study involved 15 people hospitalized with COVID-19 in March and April. Participants were a median 55 years old (range, 22-71 years) and all presented with respiratory symptoms. Only one patient had concurrent GI symptoms at admission. Median length of stay was 21 days.
Investigators collected fecal samples serially until discharge. They extracted viral DNA to test for transcriptional genetic evidence of active infection, which they detected in 7 of 15 patients. The patient with GI symptoms was not in this positive group.
The findings suggest a “quiescent but active GI infection,” the researchers note.
Three of the seven patients continued to test positive for active infection in their stool up to 6 days after respiratory clearance of SARS-CoV-2.
Microbiome matters
The investigators also extracted, amplified, and sequenced DNA from the stool samples. Their “metagenomic” profile revealed the type and amounts of bacterial strains in each patient’s gut microbiome.
Interestingly, bacterial strains differed between people with high SARS-CoV-2 infectivity versus participants with low to no evidence of active infection.
“Stool with high viral activity had higher abundance of pathogenic bacteria,” Ng said. In contrast, people with low or no infectivity had more beneficial bacterial strains, including bacteria that play critical roles in boosting host immunity.
Each patient’s microbiome composition changed during the course of the study. Whether the microbiome alters the course of COVID-19 or COVID-19 alters the composition of the microbiome requires further study, the authors note.
The U.S. Food and Drug Administration and officials in other countries have contacted the Hong Kong investigators for more details on their stool testing strategy, professor Francis K.L. Chan, dean of the faculty of medicine and director of the Centre for Gut Microbiota Research at CUHK, stated in the news release.
Further research into revealing the infectivity and pathogenesis of SARS-CoV-2 in the GI tract is warranted. The value of modulating the human gut microbiome in this patient population could be worthwhile to investigate as well, the researchers said.
Novel finding
“Some of it is not-so-new news and some is new,” David A. Johnson, MD, told Medscape Medical News when asked to comment on the study.
For example, previous researchers have detected SARS-CoV-2 virus in the stool. However, this study takes it a step further and shows that the virus present in stool can remain infectious on the basis of metagenomic signatures.
Furthermore, the virus can remain infectious in the gut even after a patient tests negative for COVID-19 through nasopharyngeal sampling – in this report up to 6 days later, said Johnson, professor of medicine, chief of gastroenterology, Eastern Virginia Medical School in Norfolk, Va.
The study carries important implications for people who currently test negative following active COVID-19 infection, he added. Centers for Disease Control and Prevention criteria clear a person as negative after two nasopharyngeal swabs at least 24 hours apart.
People in this category could believe they are no longer infectious and might return to a setting where they could infect others, Johnson said.
One potential means for spreading SARS-CoV-2 from the gut is from a toilet plume, as Johnson previously highlighted in a video report for Medscape Medical News.
The study authors disclosed no relevant financial relationships. Johnson serves as an adviser to WebMD/Medscape.
This article first appeared on Medscape.com.
Stool tests were positive among people with no GI symptoms, and in some cases up to 6 days after nasopharyngeal swabs yielded negative results.
The small pilot study suggests a quiescent but active infection in the gut. Stool testing revealed genomic evidence of active infection in 7 of the 15 participants tested in one of two hospitals in Hong Kong.
“We found active and prolonged SARS-CoV-2 infection in the stool of patients with COVID-19, even after recovery, suggesting that coronavirus could remain in the gut of asymptomatic carriers,” senior author Siew C. Ng, MBBS, PhD, told Medscape Medical News.
“Due to the potential threat of fecal-oral transmission, it is important to maintain long-term coronavirus and health surveillance,” said Ng, Associate Director of the Centre for Gut Microbiota Research at the Chinese University of Hong Kong (CUHK).
“Discharged patients and their caretakers should remain vigilant and observe strict personal and toileting hygiene,” she added.
The prospective, observational study was published online July 20 in Gut.
Ramping up COVID-19 testing
As a follow-up to these and other findings – including the testing of more than 2,000 stool samples in children and the needy arriving at Hong Kong airports starting March 29 – the same investigators are establishing a CUHK Coronavirus Testing Center.
As of Aug. 31, the detection rate in tested children was 0.28%. The Center plans to offer as many as 2,000 COVID-19 tests daily going forward to help identify asymptomatic carriers, the investigators announced in a Sept. 7 news release.
In contrast to nasopharyngeal sampling, stool specimens are “more convenient, safe and non-invasive to collect in the pediatric population,” professor Paul Chan, chairman of the Department of Microbiology, CU Medicine, said in the release. “This makes the stool test a better option for COVID-19 screening in babies, young children and those whose respiratory samples are difficult to collect.”
Even though previous researchers identified SARS-CoV-2 in the stool, the activity and infectivity of the virus in the gastrointestinal tract during and after COVID-19 respiratory positivity remained largely unknown.
Active infection detected in stool
This prospective study involved 15 people hospitalized with COVID-19 in March and April. Participants were a median 55 years old (range, 22-71 years) and all presented with respiratory symptoms. Only one patient had concurrent GI symptoms at admission. Median length of stay was 21 days.
Investigators collected fecal samples serially until discharge. They extracted viral DNA to test for transcriptional genetic evidence of active infection, which they detected in 7 of 15 patients. The patient with GI symptoms was not in this positive group.
The findings suggest a “quiescent but active GI infection,” the researchers note.
Three of the seven patients continued to test positive for active infection in their stool up to 6 days after respiratory clearance of SARS-CoV-2.
Microbiome matters
The investigators also extracted, amplified, and sequenced DNA from the stool samples. Their “metagenomic” profile revealed the type and amounts of bacterial strains in each patient’s gut microbiome.
Interestingly, bacterial strains differed between people with high SARS-CoV-2 infectivity versus participants with low to no evidence of active infection.
“Stool with high viral activity had higher abundance of pathogenic bacteria,” Ng said. In contrast, people with low or no infectivity had more beneficial bacterial strains, including bacteria that play critical roles in boosting host immunity.
Each patient’s microbiome composition changed during the course of the study. Whether the microbiome alters the course of COVID-19 or COVID-19 alters the composition of the microbiome requires further study, the authors note.
The U.S. Food and Drug Administration and officials in other countries have contacted the Hong Kong investigators for more details on their stool testing strategy, professor Francis K.L. Chan, dean of the faculty of medicine and director of the Centre for Gut Microbiota Research at CUHK, stated in the news release.
Further research into revealing the infectivity and pathogenesis of SARS-CoV-2 in the GI tract is warranted. The value of modulating the human gut microbiome in this patient population could be worthwhile to investigate as well, the researchers said.
Novel finding
“Some of it is not-so-new news and some is new,” David A. Johnson, MD, told Medscape Medical News when asked to comment on the study.
For example, previous researchers have detected SARS-CoV-2 virus in the stool. However, this study takes it a step further and shows that the virus present in stool can remain infectious on the basis of metagenomic signatures.
Furthermore, the virus can remain infectious in the gut even after a patient tests negative for COVID-19 through nasopharyngeal sampling – in this report up to 6 days later, said Johnson, professor of medicine, chief of gastroenterology, Eastern Virginia Medical School in Norfolk, Va.
The study carries important implications for people who currently test negative following active COVID-19 infection, he added. Centers for Disease Control and Prevention criteria clear a person as negative after two nasopharyngeal swabs at least 24 hours apart.
People in this category could believe they are no longer infectious and might return to a setting where they could infect others, Johnson said.
One potential means for spreading SARS-CoV-2 from the gut is from a toilet plume, as Johnson previously highlighted in a video report for Medscape Medical News.
The study authors disclosed no relevant financial relationships. Johnson serves as an adviser to WebMD/Medscape.
This article first appeared on Medscape.com.
Stool tests were positive among people with no GI symptoms, and in some cases up to 6 days after nasopharyngeal swabs yielded negative results.
The small pilot study suggests a quiescent but active infection in the gut. Stool testing revealed genomic evidence of active infection in 7 of the 15 participants tested in one of two hospitals in Hong Kong.
“We found active and prolonged SARS-CoV-2 infection in the stool of patients with COVID-19, even after recovery, suggesting that coronavirus could remain in the gut of asymptomatic carriers,” senior author Siew C. Ng, MBBS, PhD, told Medscape Medical News.
“Due to the potential threat of fecal-oral transmission, it is important to maintain long-term coronavirus and health surveillance,” said Ng, Associate Director of the Centre for Gut Microbiota Research at the Chinese University of Hong Kong (CUHK).
“Discharged patients and their caretakers should remain vigilant and observe strict personal and toileting hygiene,” she added.
The prospective, observational study was published online July 20 in Gut.
Ramping up COVID-19 testing
As a follow-up to these and other findings – including the testing of more than 2,000 stool samples in children and the needy arriving at Hong Kong airports starting March 29 – the same investigators are establishing a CUHK Coronavirus Testing Center.
As of Aug. 31, the detection rate in tested children was 0.28%. The Center plans to offer as many as 2,000 COVID-19 tests daily going forward to help identify asymptomatic carriers, the investigators announced in a Sept. 7 news release.
In contrast to nasopharyngeal sampling, stool specimens are “more convenient, safe and non-invasive to collect in the pediatric population,” professor Paul Chan, chairman of the Department of Microbiology, CU Medicine, said in the release. “This makes the stool test a better option for COVID-19 screening in babies, young children and those whose respiratory samples are difficult to collect.”
Even though previous researchers identified SARS-CoV-2 in the stool, the activity and infectivity of the virus in the gastrointestinal tract during and after COVID-19 respiratory positivity remained largely unknown.
Active infection detected in stool
This prospective study involved 15 people hospitalized with COVID-19 in March and April. Participants were a median 55 years old (range, 22-71 years) and all presented with respiratory symptoms. Only one patient had concurrent GI symptoms at admission. Median length of stay was 21 days.
Investigators collected fecal samples serially until discharge. They extracted viral DNA to test for transcriptional genetic evidence of active infection, which they detected in 7 of 15 patients. The patient with GI symptoms was not in this positive group.
The findings suggest a “quiescent but active GI infection,” the researchers note.
Three of the seven patients continued to test positive for active infection in their stool up to 6 days after respiratory clearance of SARS-CoV-2.
Microbiome matters
The investigators also extracted, amplified, and sequenced DNA from the stool samples. Their “metagenomic” profile revealed the type and amounts of bacterial strains in each patient’s gut microbiome.
Interestingly, bacterial strains differed between people with high SARS-CoV-2 infectivity versus participants with low to no evidence of active infection.
“Stool with high viral activity had higher abundance of pathogenic bacteria,” Ng said. In contrast, people with low or no infectivity had more beneficial bacterial strains, including bacteria that play critical roles in boosting host immunity.
Each patient’s microbiome composition changed during the course of the study. Whether the microbiome alters the course of COVID-19 or COVID-19 alters the composition of the microbiome requires further study, the authors note.
The U.S. Food and Drug Administration and officials in other countries have contacted the Hong Kong investigators for more details on their stool testing strategy, professor Francis K.L. Chan, dean of the faculty of medicine and director of the Centre for Gut Microbiota Research at CUHK, stated in the news release.
Further research into revealing the infectivity and pathogenesis of SARS-CoV-2 in the GI tract is warranted. The value of modulating the human gut microbiome in this patient population could be worthwhile to investigate as well, the researchers said.
Novel finding
“Some of it is not-so-new news and some is new,” David A. Johnson, MD, told Medscape Medical News when asked to comment on the study.
For example, previous researchers have detected SARS-CoV-2 virus in the stool. However, this study takes it a step further and shows that the virus present in stool can remain infectious on the basis of metagenomic signatures.
Furthermore, the virus can remain infectious in the gut even after a patient tests negative for COVID-19 through nasopharyngeal sampling – in this report up to 6 days later, said Johnson, professor of medicine, chief of gastroenterology, Eastern Virginia Medical School in Norfolk, Va.
The study carries important implications for people who currently test negative following active COVID-19 infection, he added. Centers for Disease Control and Prevention criteria clear a person as negative after two nasopharyngeal swabs at least 24 hours apart.
People in this category could believe they are no longer infectious and might return to a setting where they could infect others, Johnson said.
One potential means for spreading SARS-CoV-2 from the gut is from a toilet plume, as Johnson previously highlighted in a video report for Medscape Medical News.
The study authors disclosed no relevant financial relationships. Johnson serves as an adviser to WebMD/Medscape.
This article first appeared on Medscape.com.