User login
Framingham risk score may also predict cognitive decline
“In the absence of effective treatments for dementia, we need to monitor and control cardiovascular risk burden as a way to maintain patient’s cognitive health as they age,” said Weili Xu, PhD, Department of Epidemiology and Biostatistics, School of Public Health, Tianjin Medical University, Tianjin, China, in a press release.
“Given the progressive increase in the number of dementia cases worldwide, our findings have both clinical and public health relevance.”
Dr. Xu and first author Ruixue Song, MSc, also from Tianjin Medical University, published their findings online ahead of print May 18 in the Journal of the American College of Cardiology.
The World Health Organization projects that up to 82 million people will have dementia by 2050. Given the lack of effective treatments for dementia, identifying modifiable risk factors for cognitive decline and aggressively managing them is an increasingly appealing strategy.
Assessing cardiovascular risk and cognition
The researchers followed 1,588 dementia-free participants from the Rush Memory and Aging Project for 21 years (median, 5.8 years). FGCRS was assessed at baseline and categorized into tertiles (lowest, middle, and highest). Mean age of the studied population was 79.5 years, 75.8% of participants were female, and mean Framingham score was 15.6 (range, 4 to 28).
Annual evaluations included assessment of episodic memory (memory of everyday events), semantic memory (long-term memory), working memory (short-term memory), visuospatial ability (capacity to identify visual and spatial relationships among objects), and perceptual speed (ability to accurately and completely compare letters, numbers, objects, pictures, or patterns) using 19 tests to derive a composite score.
A subsample (n = 378) of participants underwent MRI, and structural total and regional brain volumes were estimated.
Linear regression was used to estimate beta-coefficients for the relationship between cardiovascular risk burden at baseline and longitudinally. If the beta-coefficient is negative, the interpretation is that for every 1-unit increase in the predictor variable (FGCRS), the outcome variable (cognitive function) will decrease by the beta-coefficient value.
At baseline, higher FGCRS was related to small but consistent (although not usually statistically significant) decreases in hippocampal volume, gray matter, and total brain volume.
Considered longitudinally, participants in the highest-risk tertile of FGCRS experienced faster decline in global cognition (beta = −0.019), episodic memory (beta = −0.023), working memory (beta = −0.021), and perceptual speed (beta = −0.027) during follow-up (P < .05 for all) than those in the lowest-risk tertile.
The declines in semantic memory (beta = –0.012) and visuospatial ability (beta = –0.010) did not reach statistical significance.
Bringing dementia prevention into the exam room early
Commenting on the research, Costantino Iadecola, MD, director of the Feil Family Brain and Mind Research Institute at Weill Cornell Medicine in New York City, said the study has immediate clinical usefulness.
“The link between the cardiovascular risk factors and dementia is well known, but in your doctor’s office, that link is not seen. If your GP or cardiologist sees you with high blood pressure, he’s not immediately going to think about the risk of dementia 20 years later,” said Dr. Iadecola.
“What this study does is it directly links a simple score that’s commonly used to assess cardiovascular risk to dementia risk, which can be used to counsel patients and, hopefully, reduce the risk of both cardiovascular disease and cognitive disorders.”
Dr. Iadecola wrote an editorial together with Neal S. Parikh, MD, MS, also from Weill Cornell Medicine, that accompanied the findings of the trial.
Even neurologists sometimes fail to make the connection between vascular risk and dementia, he said. “They think that by making a stroke patient move their hand better, they’re treating them, but 30% of stroke patients get dementia 6 or 8 months later and they’re missing this link between cerebrovascular pathology and dementia.
Dr. Iadecola is one of 26 experts who authored the recent Berlin Manifesto, an effort led by Vladimir Hachinski, MD, professor of neurology and epidemiology at Western University in Ontario, Canada, to raise awareness of the link between cardiovascular and brain health.
Dr. Hachinski coined the term “brain attack” and devised the Hachinski Ischemic Score that remains the standard for identifying a vascular component of cognitive impairment.
The current study has some strengths and limitations, noted Dr. Iadecola. The average age of participants was 80 years, which is appropriate given the high risk for cognitive decline at this age, but the generalizability of the study may be limited given that most participants were white women.
Going forward, he said, rigorous studies are needed to confirm these findings and to determine how to best prevent dementia through treatment of individual cardiovascular risk factors.
Dr. Xu has received grants from nonindustry entities, including the Swedish Research Council and the National Natural Science Foundation of China. The study was funded by the European Union’s Horizon 320230 research and innovation program. Dr. Iadecola is a member of the scientific advisory board for Broadview Ventures.
This article appeared on Medscape.com.
“In the absence of effective treatments for dementia, we need to monitor and control cardiovascular risk burden as a way to maintain patient’s cognitive health as they age,” said Weili Xu, PhD, Department of Epidemiology and Biostatistics, School of Public Health, Tianjin Medical University, Tianjin, China, in a press release.
“Given the progressive increase in the number of dementia cases worldwide, our findings have both clinical and public health relevance.”
Dr. Xu and first author Ruixue Song, MSc, also from Tianjin Medical University, published their findings online ahead of print May 18 in the Journal of the American College of Cardiology.
The World Health Organization projects that up to 82 million people will have dementia by 2050. Given the lack of effective treatments for dementia, identifying modifiable risk factors for cognitive decline and aggressively managing them is an increasingly appealing strategy.
Assessing cardiovascular risk and cognition
The researchers followed 1,588 dementia-free participants from the Rush Memory and Aging Project for 21 years (median, 5.8 years). FGCRS was assessed at baseline and categorized into tertiles (lowest, middle, and highest). Mean age of the studied population was 79.5 years, 75.8% of participants were female, and mean Framingham score was 15.6 (range, 4 to 28).
Annual evaluations included assessment of episodic memory (memory of everyday events), semantic memory (long-term memory), working memory (short-term memory), visuospatial ability (capacity to identify visual and spatial relationships among objects), and perceptual speed (ability to accurately and completely compare letters, numbers, objects, pictures, or patterns) using 19 tests to derive a composite score.
A subsample (n = 378) of participants underwent MRI, and structural total and regional brain volumes were estimated.
Linear regression was used to estimate beta-coefficients for the relationship between cardiovascular risk burden at baseline and longitudinally. If the beta-coefficient is negative, the interpretation is that for every 1-unit increase in the predictor variable (FGCRS), the outcome variable (cognitive function) will decrease by the beta-coefficient value.
At baseline, higher FGCRS was related to small but consistent (although not usually statistically significant) decreases in hippocampal volume, gray matter, and total brain volume.
Considered longitudinally, participants in the highest-risk tertile of FGCRS experienced faster decline in global cognition (beta = −0.019), episodic memory (beta = −0.023), working memory (beta = −0.021), and perceptual speed (beta = −0.027) during follow-up (P < .05 for all) than those in the lowest-risk tertile.
The declines in semantic memory (beta = –0.012) and visuospatial ability (beta = –0.010) did not reach statistical significance.
Bringing dementia prevention into the exam room early
Commenting on the research, Costantino Iadecola, MD, director of the Feil Family Brain and Mind Research Institute at Weill Cornell Medicine in New York City, said the study has immediate clinical usefulness.
“The link between the cardiovascular risk factors and dementia is well known, but in your doctor’s office, that link is not seen. If your GP or cardiologist sees you with high blood pressure, he’s not immediately going to think about the risk of dementia 20 years later,” said Dr. Iadecola.
“What this study does is it directly links a simple score that’s commonly used to assess cardiovascular risk to dementia risk, which can be used to counsel patients and, hopefully, reduce the risk of both cardiovascular disease and cognitive disorders.”
Dr. Iadecola wrote an editorial together with Neal S. Parikh, MD, MS, also from Weill Cornell Medicine, that accompanied the findings of the trial.
Even neurologists sometimes fail to make the connection between vascular risk and dementia, he said. “They think that by making a stroke patient move their hand better, they’re treating them, but 30% of stroke patients get dementia 6 or 8 months later and they’re missing this link between cerebrovascular pathology and dementia.
Dr. Iadecola is one of 26 experts who authored the recent Berlin Manifesto, an effort led by Vladimir Hachinski, MD, professor of neurology and epidemiology at Western University in Ontario, Canada, to raise awareness of the link between cardiovascular and brain health.
Dr. Hachinski coined the term “brain attack” and devised the Hachinski Ischemic Score that remains the standard for identifying a vascular component of cognitive impairment.
The current study has some strengths and limitations, noted Dr. Iadecola. The average age of participants was 80 years, which is appropriate given the high risk for cognitive decline at this age, but the generalizability of the study may be limited given that most participants were white women.
Going forward, he said, rigorous studies are needed to confirm these findings and to determine how to best prevent dementia through treatment of individual cardiovascular risk factors.
Dr. Xu has received grants from nonindustry entities, including the Swedish Research Council and the National Natural Science Foundation of China. The study was funded by the European Union’s Horizon 320230 research and innovation program. Dr. Iadecola is a member of the scientific advisory board for Broadview Ventures.
This article appeared on Medscape.com.
“In the absence of effective treatments for dementia, we need to monitor and control cardiovascular risk burden as a way to maintain patient’s cognitive health as they age,” said Weili Xu, PhD, Department of Epidemiology and Biostatistics, School of Public Health, Tianjin Medical University, Tianjin, China, in a press release.
“Given the progressive increase in the number of dementia cases worldwide, our findings have both clinical and public health relevance.”
Dr. Xu and first author Ruixue Song, MSc, also from Tianjin Medical University, published their findings online ahead of print May 18 in the Journal of the American College of Cardiology.
The World Health Organization projects that up to 82 million people will have dementia by 2050. Given the lack of effective treatments for dementia, identifying modifiable risk factors for cognitive decline and aggressively managing them is an increasingly appealing strategy.
Assessing cardiovascular risk and cognition
The researchers followed 1,588 dementia-free participants from the Rush Memory and Aging Project for 21 years (median, 5.8 years). FGCRS was assessed at baseline and categorized into tertiles (lowest, middle, and highest). Mean age of the studied population was 79.5 years, 75.8% of participants were female, and mean Framingham score was 15.6 (range, 4 to 28).
Annual evaluations included assessment of episodic memory (memory of everyday events), semantic memory (long-term memory), working memory (short-term memory), visuospatial ability (capacity to identify visual and spatial relationships among objects), and perceptual speed (ability to accurately and completely compare letters, numbers, objects, pictures, or patterns) using 19 tests to derive a composite score.
A subsample (n = 378) of participants underwent MRI, and structural total and regional brain volumes were estimated.
Linear regression was used to estimate beta-coefficients for the relationship between cardiovascular risk burden at baseline and longitudinally. If the beta-coefficient is negative, the interpretation is that for every 1-unit increase in the predictor variable (FGCRS), the outcome variable (cognitive function) will decrease by the beta-coefficient value.
At baseline, higher FGCRS was related to small but consistent (although not usually statistically significant) decreases in hippocampal volume, gray matter, and total brain volume.
Considered longitudinally, participants in the highest-risk tertile of FGCRS experienced faster decline in global cognition (beta = −0.019), episodic memory (beta = −0.023), working memory (beta = −0.021), and perceptual speed (beta = −0.027) during follow-up (P < .05 for all) than those in the lowest-risk tertile.
The declines in semantic memory (beta = –0.012) and visuospatial ability (beta = –0.010) did not reach statistical significance.
Bringing dementia prevention into the exam room early
Commenting on the research, Costantino Iadecola, MD, director of the Feil Family Brain and Mind Research Institute at Weill Cornell Medicine in New York City, said the study has immediate clinical usefulness.
“The link between the cardiovascular risk factors and dementia is well known, but in your doctor’s office, that link is not seen. If your GP or cardiologist sees you with high blood pressure, he’s not immediately going to think about the risk of dementia 20 years later,” said Dr. Iadecola.
“What this study does is it directly links a simple score that’s commonly used to assess cardiovascular risk to dementia risk, which can be used to counsel patients and, hopefully, reduce the risk of both cardiovascular disease and cognitive disorders.”
Dr. Iadecola wrote an editorial together with Neal S. Parikh, MD, MS, also from Weill Cornell Medicine, that accompanied the findings of the trial.
Even neurologists sometimes fail to make the connection between vascular risk and dementia, he said. “They think that by making a stroke patient move their hand better, they’re treating them, but 30% of stroke patients get dementia 6 or 8 months later and they’re missing this link between cerebrovascular pathology and dementia.
Dr. Iadecola is one of 26 experts who authored the recent Berlin Manifesto, an effort led by Vladimir Hachinski, MD, professor of neurology and epidemiology at Western University in Ontario, Canada, to raise awareness of the link between cardiovascular and brain health.
Dr. Hachinski coined the term “brain attack” and devised the Hachinski Ischemic Score that remains the standard for identifying a vascular component of cognitive impairment.
The current study has some strengths and limitations, noted Dr. Iadecola. The average age of participants was 80 years, which is appropriate given the high risk for cognitive decline at this age, but the generalizability of the study may be limited given that most participants were white women.
Going forward, he said, rigorous studies are needed to confirm these findings and to determine how to best prevent dementia through treatment of individual cardiovascular risk factors.
Dr. Xu has received grants from nonindustry entities, including the Swedish Research Council and the National Natural Science Foundation of China. The study was funded by the European Union’s Horizon 320230 research and innovation program. Dr. Iadecola is a member of the scientific advisory board for Broadview Ventures.
This article appeared on Medscape.com.
FROM THE JOURNAL OF THE AMERICAN COLLEGE OF CARDIOLOGY
AAN publishes ethical guidance on patient care during the pandemic
The document, which was published online May 15 in Neurology, reviews adaptations to the inpatient and outpatient settings and addresses the need to develop protocols for the allocation of scarce medical resources. The guidance is the product of a joint committee of the AAN, the American Neurological Association, the Child Neurology Society, and the Neurocritical Care Society Ethics Committee.
“Now is one of the most challenging times of our careers as neurologists,” said James C. Stevens, MD, president of the AAN, in a press release. “Clinics and hospitals are adapting to caring for the most ill, managing scarce resources, and trying to protect people without the disease. As neurologists, we must continue to adapt our daily practice, continue to care for our most ill neurology patients, and help contribute to the care of those afflicted with COVID-19.”
The role of telehealth
The authors recommended that ordinary appointments be held using telehealth, which, they say, already has become part of patient care. Telehealth enables neurologists to continue providing care while reducing the risk of exposure to and spread of SARS-CoV-2. The disadvantages of telehealth are that it limits physical examinations and behavioral health examinations, the authors acknowledged. “Each clinician should decide, in concert with his or her patient, if an in-person evaluation warrants the risk of an encounter,” according to the guidance.
Neurologists also should advise their patients that their neurologic condition could affect their relative risk of hospitalization and death resulting from COVID-19. Patients with multiple sclerosis or myasthenia gravis, for example, may be receiving corticosteroids or immunomodulatory therapies that make them more vulnerable to COVID-19 infection. “Even if desired services are available, neurologists and their patients ought to consider whether their care plans can safely be delayed in order to mitigate risk,” wrote the authors. Neurologists must try to maintain the customary standard of care, however, for patients with neurologic disease severe enough to warrant hospitalization, such as stroke or epilepsy.
The potential need for triage
Resources such as ventilators and ICU beds are limited, and health care facilities have had to triage them during the pandemic. Patients with a neurologic disease that decreases their likelihood of survival from a respiratory illness may not be offered these resources. Neurologists should discuss with patients and decision makers the ways in which reduced resources might affect patient care. Neurologists must “be aware of the burden of disease in their local community and how healthcare leaders plan on coping with a surge,” according to the guidance.
Advance directives, which should be a standard part of clinical care, take on increased importance during the pandemic. Patients who have not completed advance care planning documents should be encouraged to do so, according to the authors. These documents include patients’ preferences for “do not attempt resuscitation” status. Nevertheless, “we must assure patients with chronic illness that diminished resources in this healthcare crisis will not restrict their access to comfort and palliative care,” the document states.
Scarce resource allocation protocols
In the event that a surge in patients overwhelms a hospital’s contingencies and forces it to operate in crisis mode, it should have a scarce resource allocation protocol in place.
“This will surely be the most challenging aspect of patient care during this pandemic public health emergency,” wrote the authors. To ensure transparency and to mitigate the emotional effect of these decisions on patients and clinicians, scarce resource allocation protocols should be developed by teams that include intensivists, clinical ethicists, and nursing representatives who are not directly involved in the care of the critically ill patients. The goal of these protocols is to maximize the number of lives saved. They generally include an initial patient assessment followed by regular reevaluations to determine whether patients using scarce resources are benefiting less than other patients who need the same resources. The protocols should consider not only patients with COVID-19 infection, but also patients with stroke, traumatic injury, influenza, and heart failure who may need the same resources. Race, gender, ethnicity, socioeconomics, and perceived social worth should not influence care decisions, according to the guidance. Validated mortality prediction scales, such as the Glasgow Outcome Scale, can contribute to care decisions. Obtaining community input into these protocols will ensure trust in the health care system.
“If the situation necessitates hard decisions, we need to be fair, objective, transparent, and adamantly preserve our professional integrity,” wrote the authors. “Through it all, we owe it to our patients and families, as well as ourselves, to maintain our own health and wellness.”
The guidance was developed without funding, and the authors reported no relevant disclosures.
SOURCE: Rubin MA et al. Neurology. 2020 May 15. doi: 10.1212/WNL.0000000000009744.
The document, which was published online May 15 in Neurology, reviews adaptations to the inpatient and outpatient settings and addresses the need to develop protocols for the allocation of scarce medical resources. The guidance is the product of a joint committee of the AAN, the American Neurological Association, the Child Neurology Society, and the Neurocritical Care Society Ethics Committee.
“Now is one of the most challenging times of our careers as neurologists,” said James C. Stevens, MD, president of the AAN, in a press release. “Clinics and hospitals are adapting to caring for the most ill, managing scarce resources, and trying to protect people without the disease. As neurologists, we must continue to adapt our daily practice, continue to care for our most ill neurology patients, and help contribute to the care of those afflicted with COVID-19.”
The role of telehealth
The authors recommended that ordinary appointments be held using telehealth, which, they say, already has become part of patient care. Telehealth enables neurologists to continue providing care while reducing the risk of exposure to and spread of SARS-CoV-2. The disadvantages of telehealth are that it limits physical examinations and behavioral health examinations, the authors acknowledged. “Each clinician should decide, in concert with his or her patient, if an in-person evaluation warrants the risk of an encounter,” according to the guidance.
Neurologists also should advise their patients that their neurologic condition could affect their relative risk of hospitalization and death resulting from COVID-19. Patients with multiple sclerosis or myasthenia gravis, for example, may be receiving corticosteroids or immunomodulatory therapies that make them more vulnerable to COVID-19 infection. “Even if desired services are available, neurologists and their patients ought to consider whether their care plans can safely be delayed in order to mitigate risk,” wrote the authors. Neurologists must try to maintain the customary standard of care, however, for patients with neurologic disease severe enough to warrant hospitalization, such as stroke or epilepsy.
The potential need for triage
Resources such as ventilators and ICU beds are limited, and health care facilities have had to triage them during the pandemic. Patients with a neurologic disease that decreases their likelihood of survival from a respiratory illness may not be offered these resources. Neurologists should discuss with patients and decision makers the ways in which reduced resources might affect patient care. Neurologists must “be aware of the burden of disease in their local community and how healthcare leaders plan on coping with a surge,” according to the guidance.
Advance directives, which should be a standard part of clinical care, take on increased importance during the pandemic. Patients who have not completed advance care planning documents should be encouraged to do so, according to the authors. These documents include patients’ preferences for “do not attempt resuscitation” status. Nevertheless, “we must assure patients with chronic illness that diminished resources in this healthcare crisis will not restrict their access to comfort and palliative care,” the document states.
Scarce resource allocation protocols
In the event that a surge in patients overwhelms a hospital’s contingencies and forces it to operate in crisis mode, it should have a scarce resource allocation protocol in place.
“This will surely be the most challenging aspect of patient care during this pandemic public health emergency,” wrote the authors. To ensure transparency and to mitigate the emotional effect of these decisions on patients and clinicians, scarce resource allocation protocols should be developed by teams that include intensivists, clinical ethicists, and nursing representatives who are not directly involved in the care of the critically ill patients. The goal of these protocols is to maximize the number of lives saved. They generally include an initial patient assessment followed by regular reevaluations to determine whether patients using scarce resources are benefiting less than other patients who need the same resources. The protocols should consider not only patients with COVID-19 infection, but also patients with stroke, traumatic injury, influenza, and heart failure who may need the same resources. Race, gender, ethnicity, socioeconomics, and perceived social worth should not influence care decisions, according to the guidance. Validated mortality prediction scales, such as the Glasgow Outcome Scale, can contribute to care decisions. Obtaining community input into these protocols will ensure trust in the health care system.
“If the situation necessitates hard decisions, we need to be fair, objective, transparent, and adamantly preserve our professional integrity,” wrote the authors. “Through it all, we owe it to our patients and families, as well as ourselves, to maintain our own health and wellness.”
The guidance was developed without funding, and the authors reported no relevant disclosures.
SOURCE: Rubin MA et al. Neurology. 2020 May 15. doi: 10.1212/WNL.0000000000009744.
The document, which was published online May 15 in Neurology, reviews adaptations to the inpatient and outpatient settings and addresses the need to develop protocols for the allocation of scarce medical resources. The guidance is the product of a joint committee of the AAN, the American Neurological Association, the Child Neurology Society, and the Neurocritical Care Society Ethics Committee.
“Now is one of the most challenging times of our careers as neurologists,” said James C. Stevens, MD, president of the AAN, in a press release. “Clinics and hospitals are adapting to caring for the most ill, managing scarce resources, and trying to protect people without the disease. As neurologists, we must continue to adapt our daily practice, continue to care for our most ill neurology patients, and help contribute to the care of those afflicted with COVID-19.”
The role of telehealth
The authors recommended that ordinary appointments be held using telehealth, which, they say, already has become part of patient care. Telehealth enables neurologists to continue providing care while reducing the risk of exposure to and spread of SARS-CoV-2. The disadvantages of telehealth are that it limits physical examinations and behavioral health examinations, the authors acknowledged. “Each clinician should decide, in concert with his or her patient, if an in-person evaluation warrants the risk of an encounter,” according to the guidance.
Neurologists also should advise their patients that their neurologic condition could affect their relative risk of hospitalization and death resulting from COVID-19. Patients with multiple sclerosis or myasthenia gravis, for example, may be receiving corticosteroids or immunomodulatory therapies that make them more vulnerable to COVID-19 infection. “Even if desired services are available, neurologists and their patients ought to consider whether their care plans can safely be delayed in order to mitigate risk,” wrote the authors. Neurologists must try to maintain the customary standard of care, however, for patients with neurologic disease severe enough to warrant hospitalization, such as stroke or epilepsy.
The potential need for triage
Resources such as ventilators and ICU beds are limited, and health care facilities have had to triage them during the pandemic. Patients with a neurologic disease that decreases their likelihood of survival from a respiratory illness may not be offered these resources. Neurologists should discuss with patients and decision makers the ways in which reduced resources might affect patient care. Neurologists must “be aware of the burden of disease in their local community and how healthcare leaders plan on coping with a surge,” according to the guidance.
Advance directives, which should be a standard part of clinical care, take on increased importance during the pandemic. Patients who have not completed advance care planning documents should be encouraged to do so, according to the authors. These documents include patients’ preferences for “do not attempt resuscitation” status. Nevertheless, “we must assure patients with chronic illness that diminished resources in this healthcare crisis will not restrict their access to comfort and palliative care,” the document states.
Scarce resource allocation protocols
In the event that a surge in patients overwhelms a hospital’s contingencies and forces it to operate in crisis mode, it should have a scarce resource allocation protocol in place.
“This will surely be the most challenging aspect of patient care during this pandemic public health emergency,” wrote the authors. To ensure transparency and to mitigate the emotional effect of these decisions on patients and clinicians, scarce resource allocation protocols should be developed by teams that include intensivists, clinical ethicists, and nursing representatives who are not directly involved in the care of the critically ill patients. The goal of these protocols is to maximize the number of lives saved. They generally include an initial patient assessment followed by regular reevaluations to determine whether patients using scarce resources are benefiting less than other patients who need the same resources. The protocols should consider not only patients with COVID-19 infection, but also patients with stroke, traumatic injury, influenza, and heart failure who may need the same resources. Race, gender, ethnicity, socioeconomics, and perceived social worth should not influence care decisions, according to the guidance. Validated mortality prediction scales, such as the Glasgow Outcome Scale, can contribute to care decisions. Obtaining community input into these protocols will ensure trust in the health care system.
“If the situation necessitates hard decisions, we need to be fair, objective, transparent, and adamantly preserve our professional integrity,” wrote the authors. “Through it all, we owe it to our patients and families, as well as ourselves, to maintain our own health and wellness.”
The guidance was developed without funding, and the authors reported no relevant disclosures.
SOURCE: Rubin MA et al. Neurology. 2020 May 15. doi: 10.1212/WNL.0000000000009744.
FROM NEUROLOGY
Blood pressure lowering lessens risk of dementia, cognitive decline
“Although observational studies report hypertension to be an important risk factor for dementia, the benefit of blood pressure lowering on dementia or cognitive impairment in clinical trials is modest and lower than the risk reduction for stroke,” wrote Diarmaid Hughes, MB, of the NUI Galway and Saolta University Hospital Group in Galway, Ireland, and coauthors. They added, however, that “these findings have the potential to inform public health strategies to reduce the burden of dementia globally.” The study was published online ahead of print May 19 in JAMA.
A rich data set
To assess the relationship between lowering blood pressure and cognitive issues, the researchers performed a systemic search of randomized, clinical trials that compared blood pressure lowering via antihypertensive agents with a control, had at least 1 year of follow-up, included more than 1,000 participants, and reported on either dementia, cognitive impairment, cognitive decline, or a change in cognitive test scores as outcomes. Of the 14 studies deemed eligible, 12 reported either the incidence of dementia (n = 9) or a composite of dementia or cognitive impairment (n = 3) at follow-up and thus were included in the primary meta-analysis. The other two studies were used for secondary outcomes only.
The studies included 96,158 participants in total – 42.2% were women – and their mean age was 69 years. At baseline, participants’ mean systolic blood pressure was 154 mm Hg and their mean diastolic blood pressure was 83.3 mm Hg. The mean duration of follow-up was 49.24 months.
In the 12 trials that reported dementia or cognitive impairment, blood pressure lowering via antihypertensive agents, compared with control, was significantly associated with a reduction in those two outcomes (7.0% vs. 7.5% over a mean trial follow-up of 4.1 years; odds ratio, 0.93; 95% confidence interval, 0.88-0.98; absolute risk reduction, 0.39%; 95% CI, 0.09%-0.68%). Blood pressure lowering, compared with control, was also significantly associated with a reduction in cognitive decline (20.2% vs. 21.1% over a mean trial follow-up of 4.1 years; OR, 0.93; 95% CI, 0.88-0.99; ARR, 0.71%; 95% CI, 0.19%-1.2%) in the eight trials that reported it as an outcome. An analysis of the eight trials that reported a change in cognitive scores did not find a significant association between that outcome and blood pressure lowering.
Subpopulations should be examined
“This is a very broad brush stroke study, albeit a definitive one,” Richard J. Caselli, MD, of the Mayo Clinic in Phoenix said in an interview. “With all the thousands of people in this meta-analysis, there are going to be subpopulations of patients with certain characteristics or common conditions in which blood pressure lowering might have a bigger or a lesser impact on their risk factor. Is there a difference between certain racial groups? Does it matter what antihypertensive strategies are used? You can look at the interactions between blood pressure lowering and other conditions: diabetes, head injuries, air pollution, certain genetic risk factors. There are a number of additional findings that could come from a very rich data set like this.”
The authors acknowledged their study’s limitations, including the challenges of performing a meta-analysis of studies that drew from different populations and had potentially different definitions of dementia, cognitive impairment, and cognitive decline outcomes. In addition, the low incidence of dementia across clinical trials limited the researchers, and its underdetection in trials and the potential of survivor bias for healthier participants with blood pressure reductions were noted as “unmeasured sources of potential error.”
Three authors reported receiving grants or personal fees from the Wellcome Trust and the Health Research Board, the Chief Scientist Office, and Bayer AG, respectively.
SOURCE: Hughes D et al. JAMA. 2020 May 19. doi: 10.1001/jama.2020.4249.
“Although observational studies report hypertension to be an important risk factor for dementia, the benefit of blood pressure lowering on dementia or cognitive impairment in clinical trials is modest and lower than the risk reduction for stroke,” wrote Diarmaid Hughes, MB, of the NUI Galway and Saolta University Hospital Group in Galway, Ireland, and coauthors. They added, however, that “these findings have the potential to inform public health strategies to reduce the burden of dementia globally.” The study was published online ahead of print May 19 in JAMA.
A rich data set
To assess the relationship between lowering blood pressure and cognitive issues, the researchers performed a systemic search of randomized, clinical trials that compared blood pressure lowering via antihypertensive agents with a control, had at least 1 year of follow-up, included more than 1,000 participants, and reported on either dementia, cognitive impairment, cognitive decline, or a change in cognitive test scores as outcomes. Of the 14 studies deemed eligible, 12 reported either the incidence of dementia (n = 9) or a composite of dementia or cognitive impairment (n = 3) at follow-up and thus were included in the primary meta-analysis. The other two studies were used for secondary outcomes only.
The studies included 96,158 participants in total – 42.2% were women – and their mean age was 69 years. At baseline, participants’ mean systolic blood pressure was 154 mm Hg and their mean diastolic blood pressure was 83.3 mm Hg. The mean duration of follow-up was 49.24 months.
In the 12 trials that reported dementia or cognitive impairment, blood pressure lowering via antihypertensive agents, compared with control, was significantly associated with a reduction in those two outcomes (7.0% vs. 7.5% over a mean trial follow-up of 4.1 years; odds ratio, 0.93; 95% confidence interval, 0.88-0.98; absolute risk reduction, 0.39%; 95% CI, 0.09%-0.68%). Blood pressure lowering, compared with control, was also significantly associated with a reduction in cognitive decline (20.2% vs. 21.1% over a mean trial follow-up of 4.1 years; OR, 0.93; 95% CI, 0.88-0.99; ARR, 0.71%; 95% CI, 0.19%-1.2%) in the eight trials that reported it as an outcome. An analysis of the eight trials that reported a change in cognitive scores did not find a significant association between that outcome and blood pressure lowering.
Subpopulations should be examined
“This is a very broad brush stroke study, albeit a definitive one,” Richard J. Caselli, MD, of the Mayo Clinic in Phoenix said in an interview. “With all the thousands of people in this meta-analysis, there are going to be subpopulations of patients with certain characteristics or common conditions in which blood pressure lowering might have a bigger or a lesser impact on their risk factor. Is there a difference between certain racial groups? Does it matter what antihypertensive strategies are used? You can look at the interactions between blood pressure lowering and other conditions: diabetes, head injuries, air pollution, certain genetic risk factors. There are a number of additional findings that could come from a very rich data set like this.”
The authors acknowledged their study’s limitations, including the challenges of performing a meta-analysis of studies that drew from different populations and had potentially different definitions of dementia, cognitive impairment, and cognitive decline outcomes. In addition, the low incidence of dementia across clinical trials limited the researchers, and its underdetection in trials and the potential of survivor bias for healthier participants with blood pressure reductions were noted as “unmeasured sources of potential error.”
Three authors reported receiving grants or personal fees from the Wellcome Trust and the Health Research Board, the Chief Scientist Office, and Bayer AG, respectively.
SOURCE: Hughes D et al. JAMA. 2020 May 19. doi: 10.1001/jama.2020.4249.
“Although observational studies report hypertension to be an important risk factor for dementia, the benefit of blood pressure lowering on dementia or cognitive impairment in clinical trials is modest and lower than the risk reduction for stroke,” wrote Diarmaid Hughes, MB, of the NUI Galway and Saolta University Hospital Group in Galway, Ireland, and coauthors. They added, however, that “these findings have the potential to inform public health strategies to reduce the burden of dementia globally.” The study was published online ahead of print May 19 in JAMA.
A rich data set
To assess the relationship between lowering blood pressure and cognitive issues, the researchers performed a systemic search of randomized, clinical trials that compared blood pressure lowering via antihypertensive agents with a control, had at least 1 year of follow-up, included more than 1,000 participants, and reported on either dementia, cognitive impairment, cognitive decline, or a change in cognitive test scores as outcomes. Of the 14 studies deemed eligible, 12 reported either the incidence of dementia (n = 9) or a composite of dementia or cognitive impairment (n = 3) at follow-up and thus were included in the primary meta-analysis. The other two studies were used for secondary outcomes only.
The studies included 96,158 participants in total – 42.2% were women – and their mean age was 69 years. At baseline, participants’ mean systolic blood pressure was 154 mm Hg and their mean diastolic blood pressure was 83.3 mm Hg. The mean duration of follow-up was 49.24 months.
In the 12 trials that reported dementia or cognitive impairment, blood pressure lowering via antihypertensive agents, compared with control, was significantly associated with a reduction in those two outcomes (7.0% vs. 7.5% over a mean trial follow-up of 4.1 years; odds ratio, 0.93; 95% confidence interval, 0.88-0.98; absolute risk reduction, 0.39%; 95% CI, 0.09%-0.68%). Blood pressure lowering, compared with control, was also significantly associated with a reduction in cognitive decline (20.2% vs. 21.1% over a mean trial follow-up of 4.1 years; OR, 0.93; 95% CI, 0.88-0.99; ARR, 0.71%; 95% CI, 0.19%-1.2%) in the eight trials that reported it as an outcome. An analysis of the eight trials that reported a change in cognitive scores did not find a significant association between that outcome and blood pressure lowering.
Subpopulations should be examined
“This is a very broad brush stroke study, albeit a definitive one,” Richard J. Caselli, MD, of the Mayo Clinic in Phoenix said in an interview. “With all the thousands of people in this meta-analysis, there are going to be subpopulations of patients with certain characteristics or common conditions in which blood pressure lowering might have a bigger or a lesser impact on their risk factor. Is there a difference between certain racial groups? Does it matter what antihypertensive strategies are used? You can look at the interactions between blood pressure lowering and other conditions: diabetes, head injuries, air pollution, certain genetic risk factors. There are a number of additional findings that could come from a very rich data set like this.”
The authors acknowledged their study’s limitations, including the challenges of performing a meta-analysis of studies that drew from different populations and had potentially different definitions of dementia, cognitive impairment, and cognitive decline outcomes. In addition, the low incidence of dementia across clinical trials limited the researchers, and its underdetection in trials and the potential of survivor bias for healthier participants with blood pressure reductions were noted as “unmeasured sources of potential error.”
Three authors reported receiving grants or personal fees from the Wellcome Trust and the Health Research Board, the Chief Scientist Office, and Bayer AG, respectively.
SOURCE: Hughes D et al. JAMA. 2020 May 19. doi: 10.1001/jama.2020.4249.
FROM JAMA
Yoga is a good adjunct to migraine therapy
in Neurology.
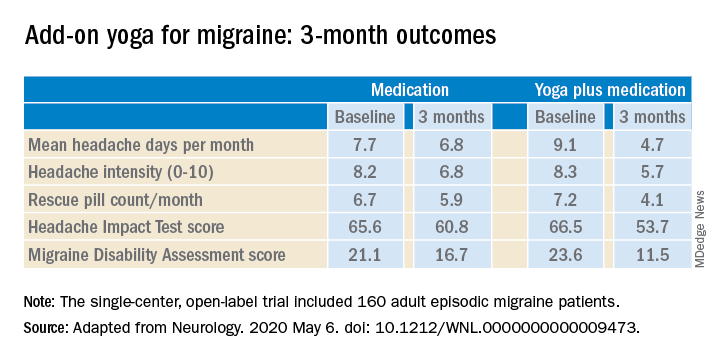
The structured yoga program resulted in “remarkably improved” outcomes at 3 months of follow-up in CONTAIN, with both headache frequency and use of medications cut in half, compared with baseline, according to the investigators.
Compared with the control group on standard antimigraine medications alone, the yoga group demonstrated significantly greater reductions in pain intensity, headache frequency, pill counts, and validated measures of disability and headache impact on daily life (see graphic).
“The good news is that practicing something as simple and accessible as yoga may help much more than medications alone. And all you need is a mat,” observed Dr. Bhatia, professor of neurology at the All India Institute of Medical Sciences in New Delhi.
The single-center, open-label, blinded-assessment CONTAIN trial included 160 adult episodic migraine patients ages 18-50 years experiencing 4-14 headaches per month. They were randomized to prophylactic and acute rescue medications alone or in combination with yoga instruction by a qualified yoga therapist in a class that met at the medical center 3 days per week for 1 month. This was followed by practice of the hour-long yoga program at home 5 days per week for the next 2 months, with twice-monthly telephone calls from the yoga center to encourage adherence and encouragement to call if questions arose. Both groups received counseling about the importance of lifestyle changes that may help with migraine, including diet, physical activity, adequate sleep, and stress reduction. Outcomes were assessed in an intent-to-treat analysis.
The yoga program included specific relaxation exercises, breathing techniques, meditation, and yoga postures, or asanas. The migraine-tailored program was vetted by yoga experts at five renowned Indian yoga centers.
No safety issues arose with the yoga program.
The investigators noted that the 47% reduction in migraine medication pill count and 49% decrease in headache frequency over the course of 3 months in the adjunctive yoga group have important implications, not only in a limited-resource country such as India, but also in the United States, where Americans spend an estimated $3.2 billion annually on prescription and over the counter headache medications, and the indirect cost associated with lost productivity due to migraine has been put at $13 billion per year.
Dr. Bhatia and colleagues speculated that the observed benefits of add-on yoga in migraineurs may involve previously described improved vagal tone and parasympathetic drive coupled with decreased sympathetic tone, increased nitric oxide levels, and loosening of stiff muscles, which can trigger headaches.
Real-life goals
Commenting on the research, neurologist Holly Yancy, DO, a headache specialist at the Banner Health - University Medicine Neuroscience Institute in Phoenix, said she was impressed by the high quality of this well-designed, adequately powered study of a complementary and alternative therapy.
“The primary and secondary endpoints were real-life goals of migraine treatment that we strive to achieve in clinical practice – and they were met in the study,” she observed. “To start with a month of in-house yoga classes to instill a baseline competence in yoga prior to transitioning to home practice and to provide resources for ongoing assistance for questions were nice touches.”
She noted that the control group also experienced reductions in migraine frequency, severity, and disability scores, albeit of significantly lesser magnitude than in the yoga group. This underscores how important it is in clinical practice to spend time counseling migraine patients on lifestyle choices.
“A trial such as this provides neurologists and other health care providers with an accessible, evidence-based treatment for migraines that can be used with other preventive treatments to decrease the frequency and the amount of medication their patients are taking. In addition, it is a behavioral therapy that can decrease triggers and potentially help patients cope with pain,” Dr. Yancy said.
“I suspect I’ll not hesitate to recommend yoga as an adjunctive treatment for patients in my clinic that are physically capable. I think it would be logical to try to extrapolate the concept to a chronic migraine population as well, though it would be ideal to base that recommendation on another study conducted with a chronic migraine population.”
Dr. Bhatia and his coinvestigators reported having no financial conflicts regarding their study, funded by the Government of India and the All India Institute of Medical Sciences.
SOURCE: Kumar A et al. Neurology. 2020 May 6. doi: 10.1212/WNL.0000000000009473.
in Neurology.

The structured yoga program resulted in “remarkably improved” outcomes at 3 months of follow-up in CONTAIN, with both headache frequency and use of medications cut in half, compared with baseline, according to the investigators.
Compared with the control group on standard antimigraine medications alone, the yoga group demonstrated significantly greater reductions in pain intensity, headache frequency, pill counts, and validated measures of disability and headache impact on daily life (see graphic).
“The good news is that practicing something as simple and accessible as yoga may help much more than medications alone. And all you need is a mat,” observed Dr. Bhatia, professor of neurology at the All India Institute of Medical Sciences in New Delhi.
The single-center, open-label, blinded-assessment CONTAIN trial included 160 adult episodic migraine patients ages 18-50 years experiencing 4-14 headaches per month. They were randomized to prophylactic and acute rescue medications alone or in combination with yoga instruction by a qualified yoga therapist in a class that met at the medical center 3 days per week for 1 month. This was followed by practice of the hour-long yoga program at home 5 days per week for the next 2 months, with twice-monthly telephone calls from the yoga center to encourage adherence and encouragement to call if questions arose. Both groups received counseling about the importance of lifestyle changes that may help with migraine, including diet, physical activity, adequate sleep, and stress reduction. Outcomes were assessed in an intent-to-treat analysis.
The yoga program included specific relaxation exercises, breathing techniques, meditation, and yoga postures, or asanas. The migraine-tailored program was vetted by yoga experts at five renowned Indian yoga centers.
No safety issues arose with the yoga program.
The investigators noted that the 47% reduction in migraine medication pill count and 49% decrease in headache frequency over the course of 3 months in the adjunctive yoga group have important implications, not only in a limited-resource country such as India, but also in the United States, where Americans spend an estimated $3.2 billion annually on prescription and over the counter headache medications, and the indirect cost associated with lost productivity due to migraine has been put at $13 billion per year.
Dr. Bhatia and colleagues speculated that the observed benefits of add-on yoga in migraineurs may involve previously described improved vagal tone and parasympathetic drive coupled with decreased sympathetic tone, increased nitric oxide levels, and loosening of stiff muscles, which can trigger headaches.
Real-life goals
Commenting on the research, neurologist Holly Yancy, DO, a headache specialist at the Banner Health - University Medicine Neuroscience Institute in Phoenix, said she was impressed by the high quality of this well-designed, adequately powered study of a complementary and alternative therapy.
“The primary and secondary endpoints were real-life goals of migraine treatment that we strive to achieve in clinical practice – and they were met in the study,” she observed. “To start with a month of in-house yoga classes to instill a baseline competence in yoga prior to transitioning to home practice and to provide resources for ongoing assistance for questions were nice touches.”
She noted that the control group also experienced reductions in migraine frequency, severity, and disability scores, albeit of significantly lesser magnitude than in the yoga group. This underscores how important it is in clinical practice to spend time counseling migraine patients on lifestyle choices.
“A trial such as this provides neurologists and other health care providers with an accessible, evidence-based treatment for migraines that can be used with other preventive treatments to decrease the frequency and the amount of medication their patients are taking. In addition, it is a behavioral therapy that can decrease triggers and potentially help patients cope with pain,” Dr. Yancy said.
“I suspect I’ll not hesitate to recommend yoga as an adjunctive treatment for patients in my clinic that are physically capable. I think it would be logical to try to extrapolate the concept to a chronic migraine population as well, though it would be ideal to base that recommendation on another study conducted with a chronic migraine population.”
Dr. Bhatia and his coinvestigators reported having no financial conflicts regarding their study, funded by the Government of India and the All India Institute of Medical Sciences.
SOURCE: Kumar A et al. Neurology. 2020 May 6. doi: 10.1212/WNL.0000000000009473.
in Neurology.

The structured yoga program resulted in “remarkably improved” outcomes at 3 months of follow-up in CONTAIN, with both headache frequency and use of medications cut in half, compared with baseline, according to the investigators.
Compared with the control group on standard antimigraine medications alone, the yoga group demonstrated significantly greater reductions in pain intensity, headache frequency, pill counts, and validated measures of disability and headache impact on daily life (see graphic).
“The good news is that practicing something as simple and accessible as yoga may help much more than medications alone. And all you need is a mat,” observed Dr. Bhatia, professor of neurology at the All India Institute of Medical Sciences in New Delhi.
The single-center, open-label, blinded-assessment CONTAIN trial included 160 adult episodic migraine patients ages 18-50 years experiencing 4-14 headaches per month. They were randomized to prophylactic and acute rescue medications alone or in combination with yoga instruction by a qualified yoga therapist in a class that met at the medical center 3 days per week for 1 month. This was followed by practice of the hour-long yoga program at home 5 days per week for the next 2 months, with twice-monthly telephone calls from the yoga center to encourage adherence and encouragement to call if questions arose. Both groups received counseling about the importance of lifestyle changes that may help with migraine, including diet, physical activity, adequate sleep, and stress reduction. Outcomes were assessed in an intent-to-treat analysis.
The yoga program included specific relaxation exercises, breathing techniques, meditation, and yoga postures, or asanas. The migraine-tailored program was vetted by yoga experts at five renowned Indian yoga centers.
No safety issues arose with the yoga program.
The investigators noted that the 47% reduction in migraine medication pill count and 49% decrease in headache frequency over the course of 3 months in the adjunctive yoga group have important implications, not only in a limited-resource country such as India, but also in the United States, where Americans spend an estimated $3.2 billion annually on prescription and over the counter headache medications, and the indirect cost associated with lost productivity due to migraine has been put at $13 billion per year.
Dr. Bhatia and colleagues speculated that the observed benefits of add-on yoga in migraineurs may involve previously described improved vagal tone and parasympathetic drive coupled with decreased sympathetic tone, increased nitric oxide levels, and loosening of stiff muscles, which can trigger headaches.
Real-life goals
Commenting on the research, neurologist Holly Yancy, DO, a headache specialist at the Banner Health - University Medicine Neuroscience Institute in Phoenix, said she was impressed by the high quality of this well-designed, adequately powered study of a complementary and alternative therapy.
“The primary and secondary endpoints were real-life goals of migraine treatment that we strive to achieve in clinical practice – and they were met in the study,” she observed. “To start with a month of in-house yoga classes to instill a baseline competence in yoga prior to transitioning to home practice and to provide resources for ongoing assistance for questions were nice touches.”
She noted that the control group also experienced reductions in migraine frequency, severity, and disability scores, albeit of significantly lesser magnitude than in the yoga group. This underscores how important it is in clinical practice to spend time counseling migraine patients on lifestyle choices.
“A trial such as this provides neurologists and other health care providers with an accessible, evidence-based treatment for migraines that can be used with other preventive treatments to decrease the frequency and the amount of medication their patients are taking. In addition, it is a behavioral therapy that can decrease triggers and potentially help patients cope with pain,” Dr. Yancy said.
“I suspect I’ll not hesitate to recommend yoga as an adjunctive treatment for patients in my clinic that are physically capable. I think it would be logical to try to extrapolate the concept to a chronic migraine population as well, though it would be ideal to base that recommendation on another study conducted with a chronic migraine population.”
Dr. Bhatia and his coinvestigators reported having no financial conflicts regarding their study, funded by the Government of India and the All India Institute of Medical Sciences.
SOURCE: Kumar A et al. Neurology. 2020 May 6. doi: 10.1212/WNL.0000000000009473.
FROM NEUROLOGY
Satralizumab monotherapy reduces NMOSD relapse rate
according to trial results published in the Lancet Neurology.
The findings help confirm the role of interleukin-6 in the pathobiology of aquaporin-4 autoantibody (AQP4-IgG)–seropositive disease. For patients who are AQP4-IgG seronegative, however, “there is insufficient evidence to indicate a risk reduction” with this drug, the investigators wrote. In addition, satralizumab did not significantly affect pain or fatigue.
“The limitations of the study include the relatively small group sizes and low number of relapses. Despite these limitations, a significant treatment benefit was observed with satralizumab, compared with placebo in the study population, with efficacy and safety comparable to satralizumab used in combination with immunosuppressants,” reported lead author Anthony Traboulsee, MD, professor of neurology at the University of British Columbia, Vancouver, and colleagues.
Satralizumab is a humanized monoclonal antibody targeting the IL-6 receptor. A prior phase 3 study, SakuraSky, found that the drug reduces the risk of NMOSD relapse when added to immunosuppressant therapy. To assess the safety and efficacy of satralizumab monotherapy, Dr. Traboulsee and colleagues conducted SakuraStar, a randomized, double-blind, placebo-controlled trial.
Evaluating drug as monotherapy
The phase 3 trial enrolled 95 patients aged 18-74 years at 44 sites in 13 countries. The investigators included patients with AQP4-IgG–seropositive or –seronegative neuromyelitis optica using the 2006 Wingerchuk criteria or with AQP4-IgG–seropositive NMOSD with at least one event of longitudinally extensive myelitis or optic neuritis. The researchers limited the number of AQP4-IgG–seronegative patients to about 30% of the study population. Eligible participants had at least one NMOSD attack or relapse in the past 12 months and a score of 6.5 or less on the Expanded Disability Status Scale (EDSS). The investigators excluded patients with a clinical relapse in the 30 days before study baseline. Participants were randomly assigned 2:1 to receive satralizumab 120 mg or placebo subcutaneously at weeks 0, 2, 4, and every 4 weeks thereafter. Concomitant immunosuppressant use was prohibited, although corticosteroids and intravenous immunoglobulin were permitted as rescue therapy for the treatment of relapse.
The primary endpoint was time to first relapse, and the trial was designed to continue until 44 relapses occurred or for 1.5 years after the last patient entered the trial, whichever occurred first.
“Because even one NMOSD attack can have serious neurological consequences, this design allowed patients who had an attack on placebo to enter the open-label phase and receive the active drug,” the researchers wrote. “The trial design used unequal randomization to minimize exposure to placebo; because patients were not permitted to receive concomitant immunosuppressant treatment in this trial, the design limited the number of patients not receiving any treatment for the disorder. Placebo was selected with the consideration that no drugs were approved for the treatment of NMOSD when the trial was designed.” Recent trials of eculizumab, inebilizumab, and satralizumab have found that the agents are effective treatments for NMOSD. In 2019, the Food and Drug Administration approved eculizumab, a complement inhibitor, for the treatment of AQP4-IgG–seropositive NMOSD.
For the primary endpoint of SakuraStar, the researchers defined relapses as new or worsening objective neurologic symptoms with at least one of the following:
- Increase of 1 or more EDSS points from a baseline EDSS score of more than 0, or increase of 2 or more EDSS points from a baseline EDSS score of 0
- Increase of 2 or more points on at least one appropriate symptom-specific functional system score for pyramidal, cerebellar, brain stem, sensory, bowel or bladder, or a single eye
- Increase of 1 or more points on more than one symptom-specific functional system score with a baseline of at least 1
- Increase of 1 or more points on a single-eye symptom-specific functional system score with a baseline score of at least 1
In addition, symptoms had to be attributable to NMOSD; persist for more than 24 hours; and not be attributable to confounding clinical factors such as fever, infection, injury, change in mood, or adverse reactions to medications. Researchers assessed EDSS and functional system scores within 7 days of a patient reporting symptoms.
The double-blind treatment period ended 1.5 years after the last enrolled patient was assigned to satralizumab or placebo. More than 80% of the participants were women, including 73% of the satralizumab group and 97% of the placebo group. In all, 95 participants were assigned to a treatment between 2014 and 2017 – 63 to satralizumab and 32 to placebo. Relapses occurred in 19 patients receiving satralizumab (30%) and 16 receiving placebo (50%). The hazard ratio was 0.45.
“Patients treated with placebo showed a shorter time to relapse and a higher withdrawal rate than did patients treated with satralizumab,” wrote Dr. Traboulsee and colleagues. The Kaplan-Meier method suggested that 76% of patients on satralizumab had not relapsed at 48 weeks, compared with 62% of patients on placebo. And at 96 weeks, 72% of patients on satralizumab had not relapsed, compared with 51% of patients on placebo.
Among patients who were AQP4-IgG seropositive, the proportion with protocol-defined relapse was 22% in the satralizumab group versus 57% in the placebo group. Among patients who were AQP4-IgG seronegative, the proportion with a protocol-defined relapse was 46% in the satralizumab group versus 33% in the placebo group.
The most common adverse events were urinary tract infection and upper respiratory tract infection, and most adverse events were mild to moderate. A higher rate of severe adverse events was reported in the satralizumab group than in the placebo group (32.1 vs. 9.9 events per 100 patient-years). The investigators considered most of the severe adverse events unrelated to the study treatment. “None of the severe adverse events led to discontinuation of the study drug except one severe event of pneumonia in the satralizumab group,” the researchers wrote.
Data confirm efficacy of IL-6 blockade
“Satralizumab was well tolerated and no meaningful adverse effects from the drug were reported and no deaths occurred,” said Michael Levy, MD, PhD, director of the NMO clinic and research laboratory at Massachusetts General Hospital, Boston, in an accompanying editorial. “This trial of satralizumab was done shortly after the completion of a parallel trial of satralizumab in patients with NMOSD in which the same dose of satralizumab reduced the risk of relapse by 62%, compared with placebo. The main difference between these two trials is that, in the first published study, participants were permitted to use background immunotherapy; otherwise, the trial designs were nearly identical, and the enrolled participants are comparable. Together, the findings from these studies suggest that background therapy seems to provide only a small additional benefit to satralizumab alone.”
Dr. Levy also discussed findings from a phase 2 study of tocilizumab for the prevention of relapse in patients with NMOSD published in the same issue of the Lancet Neurology. The satralizumab and tocilizumab data have “confirmed that IL-6 blockade reduces the risk of relapse in patients with NMOSD,” Dr. Levy said. “IL-6 is a crucial component of the immune system, but when IL-6 production is altered during autoimmune attacks and sepsis, there can be severe consequences.”
The phase 2 trial of tocilizumab, which was described at the 2019 annual congress of the European Committee for Treatment and Research in Multiple Sclerosis, included 118 patients, 87% of whom were AQP4-IgG seropositive. Patients received intravenous tocilizumab or oral azathioprine for up to 60 weeks. Overall, 14% of patients in the tocilizumab group relapsed, compared with 47% of patients in the azathioprine group.
“The main differences between this trial of tocilizumab and the two satralizumab trials are that the tocilizumab was administered intravenously, rather than subcutaneously, the study duration was approximately 1 year, and the investigators were not masked to the treatment allocation,” Dr. Levy said. “Similar to satralizumab, adverse effects with tocilizumab were mild, including asymptomatic elevations in liver enzymes and an increased incidence of respiratory and urinary infections, with no significant differences identified between the tocilizumab and azathioprine groups.”
Various immunopathologic mechanisms may influence outcomes in NMOSD. While satralizumab and tocilizumab target IL-6, eculizumab is a C5 complement inhibitor and inebilizumab is a CD19 B-cell depleting monoclonal antibody, Dr. Levy said. “The safety concerns regarding these approaches are all substantially outweighed by the benefit of preventing NMOSD relapses.”
A need to understand AQP4-IgG–seronegative disease
SakuraStar “provides convincing data for the efficacy of satralizumab monotherapy in NMOSD with subgroup analysis showing that the benefit was seen in AQP4-IgG seropositive patients,” commented Dean M. Wingerchuk, MD, director of the division of multiple sclerosis and autoimmune neurology at the Mayo Clinic in Phoenix. “The results help confirm the key role of IL-6 in the pathobiology of AQP4-IgG–seropositive disease.”
Questions about AQP4-IgG seronegative disease remain. “The results are quite similar to the SakuraSky trial, in which satralizumab was used in conjunction with other background immunosuppressive therapies, suggesting that the primary benefit may be derived primarily from satralizumab. Both trials also showed that satralizumab did not benefit the NMOSD without AQP4-IgG subgroup though the subject numbers are rather small. We need to know more about the clinical and laboratory characteristics of the seronegative patients as they likely comprise a heterogeneous group. For example, did any of them have other autoantibodies such as MOG-IgG? Depending on the results, those details may help us understand the relative role of IL-6 in AQP4-IgG–seronegative subgroups, an important area of further study.”
SakuraStar was funded by Chugai Pharmaceutical, a member of the Roche group. Dr. Traboulsee reported grants, personal fees, and nonfinancial support from Chugai Pharmaceutical during the study, and several coauthors were employees of Chugai Pharmaceutical. Additional coauthors reported personal fees from Chugai, Roche, and other companies. Dr. Levy has received consulting fees from Alexion, Viela Bio, Chugai Pharmaceutical, Quest Diagnostics, and UCB Pharmaceuticals.
SOURCE: Traboulsee A et al. Lancet Neurol. 2020;19(5):402-12.
according to trial results published in the Lancet Neurology.
The findings help confirm the role of interleukin-6 in the pathobiology of aquaporin-4 autoantibody (AQP4-IgG)–seropositive disease. For patients who are AQP4-IgG seronegative, however, “there is insufficient evidence to indicate a risk reduction” with this drug, the investigators wrote. In addition, satralizumab did not significantly affect pain or fatigue.
“The limitations of the study include the relatively small group sizes and low number of relapses. Despite these limitations, a significant treatment benefit was observed with satralizumab, compared with placebo in the study population, with efficacy and safety comparable to satralizumab used in combination with immunosuppressants,” reported lead author Anthony Traboulsee, MD, professor of neurology at the University of British Columbia, Vancouver, and colleagues.
Satralizumab is a humanized monoclonal antibody targeting the IL-6 receptor. A prior phase 3 study, SakuraSky, found that the drug reduces the risk of NMOSD relapse when added to immunosuppressant therapy. To assess the safety and efficacy of satralizumab monotherapy, Dr. Traboulsee and colleagues conducted SakuraStar, a randomized, double-blind, placebo-controlled trial.
Evaluating drug as monotherapy
The phase 3 trial enrolled 95 patients aged 18-74 years at 44 sites in 13 countries. The investigators included patients with AQP4-IgG–seropositive or –seronegative neuromyelitis optica using the 2006 Wingerchuk criteria or with AQP4-IgG–seropositive NMOSD with at least one event of longitudinally extensive myelitis or optic neuritis. The researchers limited the number of AQP4-IgG–seronegative patients to about 30% of the study population. Eligible participants had at least one NMOSD attack or relapse in the past 12 months and a score of 6.5 or less on the Expanded Disability Status Scale (EDSS). The investigators excluded patients with a clinical relapse in the 30 days before study baseline. Participants were randomly assigned 2:1 to receive satralizumab 120 mg or placebo subcutaneously at weeks 0, 2, 4, and every 4 weeks thereafter. Concomitant immunosuppressant use was prohibited, although corticosteroids and intravenous immunoglobulin were permitted as rescue therapy for the treatment of relapse.
The primary endpoint was time to first relapse, and the trial was designed to continue until 44 relapses occurred or for 1.5 years after the last patient entered the trial, whichever occurred first.
“Because even one NMOSD attack can have serious neurological consequences, this design allowed patients who had an attack on placebo to enter the open-label phase and receive the active drug,” the researchers wrote. “The trial design used unequal randomization to minimize exposure to placebo; because patients were not permitted to receive concomitant immunosuppressant treatment in this trial, the design limited the number of patients not receiving any treatment for the disorder. Placebo was selected with the consideration that no drugs were approved for the treatment of NMOSD when the trial was designed.” Recent trials of eculizumab, inebilizumab, and satralizumab have found that the agents are effective treatments for NMOSD. In 2019, the Food and Drug Administration approved eculizumab, a complement inhibitor, for the treatment of AQP4-IgG–seropositive NMOSD.
For the primary endpoint of SakuraStar, the researchers defined relapses as new or worsening objective neurologic symptoms with at least one of the following:
- Increase of 1 or more EDSS points from a baseline EDSS score of more than 0, or increase of 2 or more EDSS points from a baseline EDSS score of 0
- Increase of 2 or more points on at least one appropriate symptom-specific functional system score for pyramidal, cerebellar, brain stem, sensory, bowel or bladder, or a single eye
- Increase of 1 or more points on more than one symptom-specific functional system score with a baseline of at least 1
- Increase of 1 or more points on a single-eye symptom-specific functional system score with a baseline score of at least 1
In addition, symptoms had to be attributable to NMOSD; persist for more than 24 hours; and not be attributable to confounding clinical factors such as fever, infection, injury, change in mood, or adverse reactions to medications. Researchers assessed EDSS and functional system scores within 7 days of a patient reporting symptoms.
The double-blind treatment period ended 1.5 years after the last enrolled patient was assigned to satralizumab or placebo. More than 80% of the participants were women, including 73% of the satralizumab group and 97% of the placebo group. In all, 95 participants were assigned to a treatment between 2014 and 2017 – 63 to satralizumab and 32 to placebo. Relapses occurred in 19 patients receiving satralizumab (30%) and 16 receiving placebo (50%). The hazard ratio was 0.45.
“Patients treated with placebo showed a shorter time to relapse and a higher withdrawal rate than did patients treated with satralizumab,” wrote Dr. Traboulsee and colleagues. The Kaplan-Meier method suggested that 76% of patients on satralizumab had not relapsed at 48 weeks, compared with 62% of patients on placebo. And at 96 weeks, 72% of patients on satralizumab had not relapsed, compared with 51% of patients on placebo.
Among patients who were AQP4-IgG seropositive, the proportion with protocol-defined relapse was 22% in the satralizumab group versus 57% in the placebo group. Among patients who were AQP4-IgG seronegative, the proportion with a protocol-defined relapse was 46% in the satralizumab group versus 33% in the placebo group.
The most common adverse events were urinary tract infection and upper respiratory tract infection, and most adverse events were mild to moderate. A higher rate of severe adverse events was reported in the satralizumab group than in the placebo group (32.1 vs. 9.9 events per 100 patient-years). The investigators considered most of the severe adverse events unrelated to the study treatment. “None of the severe adverse events led to discontinuation of the study drug except one severe event of pneumonia in the satralizumab group,” the researchers wrote.
Data confirm efficacy of IL-6 blockade
“Satralizumab was well tolerated and no meaningful adverse effects from the drug were reported and no deaths occurred,” said Michael Levy, MD, PhD, director of the NMO clinic and research laboratory at Massachusetts General Hospital, Boston, in an accompanying editorial. “This trial of satralizumab was done shortly after the completion of a parallel trial of satralizumab in patients with NMOSD in which the same dose of satralizumab reduced the risk of relapse by 62%, compared with placebo. The main difference between these two trials is that, in the first published study, participants were permitted to use background immunotherapy; otherwise, the trial designs were nearly identical, and the enrolled participants are comparable. Together, the findings from these studies suggest that background therapy seems to provide only a small additional benefit to satralizumab alone.”
Dr. Levy also discussed findings from a phase 2 study of tocilizumab for the prevention of relapse in patients with NMOSD published in the same issue of the Lancet Neurology. The satralizumab and tocilizumab data have “confirmed that IL-6 blockade reduces the risk of relapse in patients with NMOSD,” Dr. Levy said. “IL-6 is a crucial component of the immune system, but when IL-6 production is altered during autoimmune attacks and sepsis, there can be severe consequences.”
The phase 2 trial of tocilizumab, which was described at the 2019 annual congress of the European Committee for Treatment and Research in Multiple Sclerosis, included 118 patients, 87% of whom were AQP4-IgG seropositive. Patients received intravenous tocilizumab or oral azathioprine for up to 60 weeks. Overall, 14% of patients in the tocilizumab group relapsed, compared with 47% of patients in the azathioprine group.
“The main differences between this trial of tocilizumab and the two satralizumab trials are that the tocilizumab was administered intravenously, rather than subcutaneously, the study duration was approximately 1 year, and the investigators were not masked to the treatment allocation,” Dr. Levy said. “Similar to satralizumab, adverse effects with tocilizumab were mild, including asymptomatic elevations in liver enzymes and an increased incidence of respiratory and urinary infections, with no significant differences identified between the tocilizumab and azathioprine groups.”
Various immunopathologic mechanisms may influence outcomes in NMOSD. While satralizumab and tocilizumab target IL-6, eculizumab is a C5 complement inhibitor and inebilizumab is a CD19 B-cell depleting monoclonal antibody, Dr. Levy said. “The safety concerns regarding these approaches are all substantially outweighed by the benefit of preventing NMOSD relapses.”
A need to understand AQP4-IgG–seronegative disease
SakuraStar “provides convincing data for the efficacy of satralizumab monotherapy in NMOSD with subgroup analysis showing that the benefit was seen in AQP4-IgG seropositive patients,” commented Dean M. Wingerchuk, MD, director of the division of multiple sclerosis and autoimmune neurology at the Mayo Clinic in Phoenix. “The results help confirm the key role of IL-6 in the pathobiology of AQP4-IgG–seropositive disease.”
Questions about AQP4-IgG seronegative disease remain. “The results are quite similar to the SakuraSky trial, in which satralizumab was used in conjunction with other background immunosuppressive therapies, suggesting that the primary benefit may be derived primarily from satralizumab. Both trials also showed that satralizumab did not benefit the NMOSD without AQP4-IgG subgroup though the subject numbers are rather small. We need to know more about the clinical and laboratory characteristics of the seronegative patients as they likely comprise a heterogeneous group. For example, did any of them have other autoantibodies such as MOG-IgG? Depending on the results, those details may help us understand the relative role of IL-6 in AQP4-IgG–seronegative subgroups, an important area of further study.”
SakuraStar was funded by Chugai Pharmaceutical, a member of the Roche group. Dr. Traboulsee reported grants, personal fees, and nonfinancial support from Chugai Pharmaceutical during the study, and several coauthors were employees of Chugai Pharmaceutical. Additional coauthors reported personal fees from Chugai, Roche, and other companies. Dr. Levy has received consulting fees from Alexion, Viela Bio, Chugai Pharmaceutical, Quest Diagnostics, and UCB Pharmaceuticals.
SOURCE: Traboulsee A et al. Lancet Neurol. 2020;19(5):402-12.
according to trial results published in the Lancet Neurology.
The findings help confirm the role of interleukin-6 in the pathobiology of aquaporin-4 autoantibody (AQP4-IgG)–seropositive disease. For patients who are AQP4-IgG seronegative, however, “there is insufficient evidence to indicate a risk reduction” with this drug, the investigators wrote. In addition, satralizumab did not significantly affect pain or fatigue.
“The limitations of the study include the relatively small group sizes and low number of relapses. Despite these limitations, a significant treatment benefit was observed with satralizumab, compared with placebo in the study population, with efficacy and safety comparable to satralizumab used in combination with immunosuppressants,” reported lead author Anthony Traboulsee, MD, professor of neurology at the University of British Columbia, Vancouver, and colleagues.
Satralizumab is a humanized monoclonal antibody targeting the IL-6 receptor. A prior phase 3 study, SakuraSky, found that the drug reduces the risk of NMOSD relapse when added to immunosuppressant therapy. To assess the safety and efficacy of satralizumab monotherapy, Dr. Traboulsee and colleagues conducted SakuraStar, a randomized, double-blind, placebo-controlled trial.
Evaluating drug as monotherapy
The phase 3 trial enrolled 95 patients aged 18-74 years at 44 sites in 13 countries. The investigators included patients with AQP4-IgG–seropositive or –seronegative neuromyelitis optica using the 2006 Wingerchuk criteria or with AQP4-IgG–seropositive NMOSD with at least one event of longitudinally extensive myelitis or optic neuritis. The researchers limited the number of AQP4-IgG–seronegative patients to about 30% of the study population. Eligible participants had at least one NMOSD attack or relapse in the past 12 months and a score of 6.5 or less on the Expanded Disability Status Scale (EDSS). The investigators excluded patients with a clinical relapse in the 30 days before study baseline. Participants were randomly assigned 2:1 to receive satralizumab 120 mg or placebo subcutaneously at weeks 0, 2, 4, and every 4 weeks thereafter. Concomitant immunosuppressant use was prohibited, although corticosteroids and intravenous immunoglobulin were permitted as rescue therapy for the treatment of relapse.
The primary endpoint was time to first relapse, and the trial was designed to continue until 44 relapses occurred or for 1.5 years after the last patient entered the trial, whichever occurred first.
“Because even one NMOSD attack can have serious neurological consequences, this design allowed patients who had an attack on placebo to enter the open-label phase and receive the active drug,” the researchers wrote. “The trial design used unequal randomization to minimize exposure to placebo; because patients were not permitted to receive concomitant immunosuppressant treatment in this trial, the design limited the number of patients not receiving any treatment for the disorder. Placebo was selected with the consideration that no drugs were approved for the treatment of NMOSD when the trial was designed.” Recent trials of eculizumab, inebilizumab, and satralizumab have found that the agents are effective treatments for NMOSD. In 2019, the Food and Drug Administration approved eculizumab, a complement inhibitor, for the treatment of AQP4-IgG–seropositive NMOSD.
For the primary endpoint of SakuraStar, the researchers defined relapses as new or worsening objective neurologic symptoms with at least one of the following:
- Increase of 1 or more EDSS points from a baseline EDSS score of more than 0, or increase of 2 or more EDSS points from a baseline EDSS score of 0
- Increase of 2 or more points on at least one appropriate symptom-specific functional system score for pyramidal, cerebellar, brain stem, sensory, bowel or bladder, or a single eye
- Increase of 1 or more points on more than one symptom-specific functional system score with a baseline of at least 1
- Increase of 1 or more points on a single-eye symptom-specific functional system score with a baseline score of at least 1
In addition, symptoms had to be attributable to NMOSD; persist for more than 24 hours; and not be attributable to confounding clinical factors such as fever, infection, injury, change in mood, or adverse reactions to medications. Researchers assessed EDSS and functional system scores within 7 days of a patient reporting symptoms.
The double-blind treatment period ended 1.5 years after the last enrolled patient was assigned to satralizumab or placebo. More than 80% of the participants were women, including 73% of the satralizumab group and 97% of the placebo group. In all, 95 participants were assigned to a treatment between 2014 and 2017 – 63 to satralizumab and 32 to placebo. Relapses occurred in 19 patients receiving satralizumab (30%) and 16 receiving placebo (50%). The hazard ratio was 0.45.
“Patients treated with placebo showed a shorter time to relapse and a higher withdrawal rate than did patients treated with satralizumab,” wrote Dr. Traboulsee and colleagues. The Kaplan-Meier method suggested that 76% of patients on satralizumab had not relapsed at 48 weeks, compared with 62% of patients on placebo. And at 96 weeks, 72% of patients on satralizumab had not relapsed, compared with 51% of patients on placebo.
Among patients who were AQP4-IgG seropositive, the proportion with protocol-defined relapse was 22% in the satralizumab group versus 57% in the placebo group. Among patients who were AQP4-IgG seronegative, the proportion with a protocol-defined relapse was 46% in the satralizumab group versus 33% in the placebo group.
The most common adverse events were urinary tract infection and upper respiratory tract infection, and most adverse events were mild to moderate. A higher rate of severe adverse events was reported in the satralizumab group than in the placebo group (32.1 vs. 9.9 events per 100 patient-years). The investigators considered most of the severe adverse events unrelated to the study treatment. “None of the severe adverse events led to discontinuation of the study drug except one severe event of pneumonia in the satralizumab group,” the researchers wrote.
Data confirm efficacy of IL-6 blockade
“Satralizumab was well tolerated and no meaningful adverse effects from the drug were reported and no deaths occurred,” said Michael Levy, MD, PhD, director of the NMO clinic and research laboratory at Massachusetts General Hospital, Boston, in an accompanying editorial. “This trial of satralizumab was done shortly after the completion of a parallel trial of satralizumab in patients with NMOSD in which the same dose of satralizumab reduced the risk of relapse by 62%, compared with placebo. The main difference between these two trials is that, in the first published study, participants were permitted to use background immunotherapy; otherwise, the trial designs were nearly identical, and the enrolled participants are comparable. Together, the findings from these studies suggest that background therapy seems to provide only a small additional benefit to satralizumab alone.”
Dr. Levy also discussed findings from a phase 2 study of tocilizumab for the prevention of relapse in patients with NMOSD published in the same issue of the Lancet Neurology. The satralizumab and tocilizumab data have “confirmed that IL-6 blockade reduces the risk of relapse in patients with NMOSD,” Dr. Levy said. “IL-6 is a crucial component of the immune system, but when IL-6 production is altered during autoimmune attacks and sepsis, there can be severe consequences.”
The phase 2 trial of tocilizumab, which was described at the 2019 annual congress of the European Committee for Treatment and Research in Multiple Sclerosis, included 118 patients, 87% of whom were AQP4-IgG seropositive. Patients received intravenous tocilizumab or oral azathioprine for up to 60 weeks. Overall, 14% of patients in the tocilizumab group relapsed, compared with 47% of patients in the azathioprine group.
“The main differences between this trial of tocilizumab and the two satralizumab trials are that the tocilizumab was administered intravenously, rather than subcutaneously, the study duration was approximately 1 year, and the investigators were not masked to the treatment allocation,” Dr. Levy said. “Similar to satralizumab, adverse effects with tocilizumab were mild, including asymptomatic elevations in liver enzymes and an increased incidence of respiratory and urinary infections, with no significant differences identified between the tocilizumab and azathioprine groups.”
Various immunopathologic mechanisms may influence outcomes in NMOSD. While satralizumab and tocilizumab target IL-6, eculizumab is a C5 complement inhibitor and inebilizumab is a CD19 B-cell depleting monoclonal antibody, Dr. Levy said. “The safety concerns regarding these approaches are all substantially outweighed by the benefit of preventing NMOSD relapses.”
A need to understand AQP4-IgG–seronegative disease
SakuraStar “provides convincing data for the efficacy of satralizumab monotherapy in NMOSD with subgroup analysis showing that the benefit was seen in AQP4-IgG seropositive patients,” commented Dean M. Wingerchuk, MD, director of the division of multiple sclerosis and autoimmune neurology at the Mayo Clinic in Phoenix. “The results help confirm the key role of IL-6 in the pathobiology of AQP4-IgG–seropositive disease.”
Questions about AQP4-IgG seronegative disease remain. “The results are quite similar to the SakuraSky trial, in which satralizumab was used in conjunction with other background immunosuppressive therapies, suggesting that the primary benefit may be derived primarily from satralizumab. Both trials also showed that satralizumab did not benefit the NMOSD without AQP4-IgG subgroup though the subject numbers are rather small. We need to know more about the clinical and laboratory characteristics of the seronegative patients as they likely comprise a heterogeneous group. For example, did any of them have other autoantibodies such as MOG-IgG? Depending on the results, those details may help us understand the relative role of IL-6 in AQP4-IgG–seronegative subgroups, an important area of further study.”
SakuraStar was funded by Chugai Pharmaceutical, a member of the Roche group. Dr. Traboulsee reported grants, personal fees, and nonfinancial support from Chugai Pharmaceutical during the study, and several coauthors were employees of Chugai Pharmaceutical. Additional coauthors reported personal fees from Chugai, Roche, and other companies. Dr. Levy has received consulting fees from Alexion, Viela Bio, Chugai Pharmaceutical, Quest Diagnostics, and UCB Pharmaceuticals.
SOURCE: Traboulsee A et al. Lancet Neurol. 2020;19(5):402-12.
FROM THE LANCET NEUROLOGY
Updated AAN advisory outlines when PFO closure may be option for patients with stroke
Patients with an embolic-appearing infarct who are younger than 60 years, have undergone a thorough evaluation to rule out other stroke mechanisms, and have discussed with doctors the potential risks and benefits may be candidates for the procedure.
“For patients with cryptogenic stroke and PFO, percutaneous PFO closure probably reduces the risk of stroke recurrence with [a hazard ratio] of 0.41 and an absolute risk reduction of 3.4% at 5 years; probably is associated with a periprocedural complication rate of 3.9%; and probably is associated with the development of serious nonperiprocedural atrial fibrillation, with a relative risk of 2.72,” according to the advisory authors’ meta-analysis.
Most procedural complications and instances of atrial fibrillation were “self-limited and are of uncertain long-term clinical consequence, given the lower rate of stroke in patients whose PFOs were closed,” the authors said. “Subgroup analysis suggests that the overall benefit seen across trials may not extend to those patients with small shunts and small, deep infarcts.” The authors estimated that the number of patients who need to be treated to prevent one stroke at 5 years is 29.
The advisory updates 2016 guidance that said clinicians should not routinely offer PFO closure outside of a research setting. Since then, three trials published in 2017 the New England Journal of Medicine (RESPECT, CLOSE, and REDUCE) and one trial published in 2018 in the Journal of the American College of Cardiology (DEFENSE-PFO) found that PFO closure reduces the risk of recurrent stroke in patients with a PFO who have had a cryptogenic stroke, compared with medical therapy alone. In addition, the Food and Drug Administration approved the Amplatzer PFO Occluder and Gore Cardioform Septal Occluder. These developments necessitated the practice advisory, the authors said. The advisory was published online April 29 in Neurology. It is endorsed by the American Heart Association/American Stroke Association, the Society for Cardiovascular Angiography and Interventions, and the European Academy of Neurology.
Systematic review
For the update, Steven R. Messé, MD, of the Hospital of the University of Pennsylvania in Philadelphia, and a panel of neurologists, internists, and cardiologists with expertise in stroke and PFO systematically reviewed relevant randomized studies published through August 2019 and conducted meta-analyses to make their recommendations. The literature search identified eight articles that met inclusion criteria, including one article that provided follow-up from a trial that had been included in the previous practice advisory.
“The risk of a second stroke in people with PFO and no other possible causes of stroke is very low, approximately 1% per year while being treated with just medication alone,” Dr. Messé said in a news release. “Also, it is difficult to determine with absolute certainty that the PFO is the cause of a person’s stroke. So it is important that people with PFO are educated about the benefits and risks of PFO closure.” For patients who opt to take medication only, doctors may consider prescribing antiplatelet or anticoagulant drugs, according to the advisory. “All patients with previous stroke should be treated with an antithrombotic medication indefinitely if there is no bleeding contraindication, regardless of whether a PFO is present or if it is closed,” Dr. Messé and colleagues wrote. “However, specific antithrombotic management for patients with stroke thought to be caused by PFO remains uncertain.”
Calls for thorough work-up
“If an alternative plausible higher-risk mechanism of stroke is identified, it is likely that the PFO was an innocent bystander,” the authors said. “Secondary stroke prevention is optimized by targeting the most likely etiology of the preceding event. ... The randomized PFO closure trials all mandated thorough evaluations for participants before enrollment ... to rule out other stroke mechanisms; moreover, all studies required TEE [transesophageal echocardiography] to characterize the PFO and ensure that it was the most likely etiology for the initial event.”
In patients being considered for PFO closure, clinicians should obtain brain imaging to confirm stroke size and distribution (level B); obtain vascular imaging of the cervical and intracranial vessels to look for dissection, vasculopathy, and atherosclerosis (level B); and perform hypercoagulable studies (level B), according to the advisory. Clinicians must perform a baseline ECG to look for atrial fibrillation (level A), and patients thought to be at risk of atrial fibrillation should receive prolonged cardiac monitoring for at least 28 days (level B).
Before PFO closure, a clinician with expertise in stroke should assess the patient to ensure that the PFO is the most plausible mechanism of stroke (level B). “If a higher-risk alternative mechanism of stroke is identified, clinicians should not routinely recommend PFO closure (level B),” the authors said. Patients also should be assessed by a clinician with expertise in assessing the anatomic features of a PFO and performing PFO closure (level B).
The randomized trials focused on patients whose PFOs were closed within 6 months of a stroke, and registry studies are needed to assess long-term outcomes, noted Dr. Messé and colleagues. “It remains unclear whether closure provides a similar benefit in these patients who otherwise still fit the studies’ inclusion criteria,” the authors said. “Long-term and large-scale safety registries for patients who have received PFO closure are needed to assess the risk of device erosion, fracture, embolization, and thrombotic and endocarditis risks, and the effect of residual shunts and incidence of atrial fibrillation.”
About 25% of the general adult population has a PFO. “It’s important to note that having a PFO is common, and that most people with PFO will never know they have it because it usually does not cause any problems,” Dr. Messé said. “However, while there is generally a very low risk of stroke in patients with PFO, in younger people who have had a stroke without any other possible causes identified, closing the PFO may reduce the risk of having another stroke better than medication alone.”
The practice advisory was developed with financial support from the AAN. Dr. Messé and most of the authors had no relevant conflicts of interest. Several authors disclosed ties to medical device and pharmaceutical companies.
SOURCE: Messé SR et al. Neurology. 2020 Apr 29. doi: 10.1212/WNL.0000000000009443.
Patients with an embolic-appearing infarct who are younger than 60 years, have undergone a thorough evaluation to rule out other stroke mechanisms, and have discussed with doctors the potential risks and benefits may be candidates for the procedure.
“For patients with cryptogenic stroke and PFO, percutaneous PFO closure probably reduces the risk of stroke recurrence with [a hazard ratio] of 0.41 and an absolute risk reduction of 3.4% at 5 years; probably is associated with a periprocedural complication rate of 3.9%; and probably is associated with the development of serious nonperiprocedural atrial fibrillation, with a relative risk of 2.72,” according to the advisory authors’ meta-analysis.
Most procedural complications and instances of atrial fibrillation were “self-limited and are of uncertain long-term clinical consequence, given the lower rate of stroke in patients whose PFOs were closed,” the authors said. “Subgroup analysis suggests that the overall benefit seen across trials may not extend to those patients with small shunts and small, deep infarcts.” The authors estimated that the number of patients who need to be treated to prevent one stroke at 5 years is 29.
The advisory updates 2016 guidance that said clinicians should not routinely offer PFO closure outside of a research setting. Since then, three trials published in 2017 the New England Journal of Medicine (RESPECT, CLOSE, and REDUCE) and one trial published in 2018 in the Journal of the American College of Cardiology (DEFENSE-PFO) found that PFO closure reduces the risk of recurrent stroke in patients with a PFO who have had a cryptogenic stroke, compared with medical therapy alone. In addition, the Food and Drug Administration approved the Amplatzer PFO Occluder and Gore Cardioform Septal Occluder. These developments necessitated the practice advisory, the authors said. The advisory was published online April 29 in Neurology. It is endorsed by the American Heart Association/American Stroke Association, the Society for Cardiovascular Angiography and Interventions, and the European Academy of Neurology.
Systematic review
For the update, Steven R. Messé, MD, of the Hospital of the University of Pennsylvania in Philadelphia, and a panel of neurologists, internists, and cardiologists with expertise in stroke and PFO systematically reviewed relevant randomized studies published through August 2019 and conducted meta-analyses to make their recommendations. The literature search identified eight articles that met inclusion criteria, including one article that provided follow-up from a trial that had been included in the previous practice advisory.
“The risk of a second stroke in people with PFO and no other possible causes of stroke is very low, approximately 1% per year while being treated with just medication alone,” Dr. Messé said in a news release. “Also, it is difficult to determine with absolute certainty that the PFO is the cause of a person’s stroke. So it is important that people with PFO are educated about the benefits and risks of PFO closure.” For patients who opt to take medication only, doctors may consider prescribing antiplatelet or anticoagulant drugs, according to the advisory. “All patients with previous stroke should be treated with an antithrombotic medication indefinitely if there is no bleeding contraindication, regardless of whether a PFO is present or if it is closed,” Dr. Messé and colleagues wrote. “However, specific antithrombotic management for patients with stroke thought to be caused by PFO remains uncertain.”
Calls for thorough work-up
“If an alternative plausible higher-risk mechanism of stroke is identified, it is likely that the PFO was an innocent bystander,” the authors said. “Secondary stroke prevention is optimized by targeting the most likely etiology of the preceding event. ... The randomized PFO closure trials all mandated thorough evaluations for participants before enrollment ... to rule out other stroke mechanisms; moreover, all studies required TEE [transesophageal echocardiography] to characterize the PFO and ensure that it was the most likely etiology for the initial event.”
In patients being considered for PFO closure, clinicians should obtain brain imaging to confirm stroke size and distribution (level B); obtain vascular imaging of the cervical and intracranial vessels to look for dissection, vasculopathy, and atherosclerosis (level B); and perform hypercoagulable studies (level B), according to the advisory. Clinicians must perform a baseline ECG to look for atrial fibrillation (level A), and patients thought to be at risk of atrial fibrillation should receive prolonged cardiac monitoring for at least 28 days (level B).
Before PFO closure, a clinician with expertise in stroke should assess the patient to ensure that the PFO is the most plausible mechanism of stroke (level B). “If a higher-risk alternative mechanism of stroke is identified, clinicians should not routinely recommend PFO closure (level B),” the authors said. Patients also should be assessed by a clinician with expertise in assessing the anatomic features of a PFO and performing PFO closure (level B).
The randomized trials focused on patients whose PFOs were closed within 6 months of a stroke, and registry studies are needed to assess long-term outcomes, noted Dr. Messé and colleagues. “It remains unclear whether closure provides a similar benefit in these patients who otherwise still fit the studies’ inclusion criteria,” the authors said. “Long-term and large-scale safety registries for patients who have received PFO closure are needed to assess the risk of device erosion, fracture, embolization, and thrombotic and endocarditis risks, and the effect of residual shunts and incidence of atrial fibrillation.”
About 25% of the general adult population has a PFO. “It’s important to note that having a PFO is common, and that most people with PFO will never know they have it because it usually does not cause any problems,” Dr. Messé said. “However, while there is generally a very low risk of stroke in patients with PFO, in younger people who have had a stroke without any other possible causes identified, closing the PFO may reduce the risk of having another stroke better than medication alone.”
The practice advisory was developed with financial support from the AAN. Dr. Messé and most of the authors had no relevant conflicts of interest. Several authors disclosed ties to medical device and pharmaceutical companies.
SOURCE: Messé SR et al. Neurology. 2020 Apr 29. doi: 10.1212/WNL.0000000000009443.
Patients with an embolic-appearing infarct who are younger than 60 years, have undergone a thorough evaluation to rule out other stroke mechanisms, and have discussed with doctors the potential risks and benefits may be candidates for the procedure.
“For patients with cryptogenic stroke and PFO, percutaneous PFO closure probably reduces the risk of stroke recurrence with [a hazard ratio] of 0.41 and an absolute risk reduction of 3.4% at 5 years; probably is associated with a periprocedural complication rate of 3.9%; and probably is associated with the development of serious nonperiprocedural atrial fibrillation, with a relative risk of 2.72,” according to the advisory authors’ meta-analysis.
Most procedural complications and instances of atrial fibrillation were “self-limited and are of uncertain long-term clinical consequence, given the lower rate of stroke in patients whose PFOs were closed,” the authors said. “Subgroup analysis suggests that the overall benefit seen across trials may not extend to those patients with small shunts and small, deep infarcts.” The authors estimated that the number of patients who need to be treated to prevent one stroke at 5 years is 29.
The advisory updates 2016 guidance that said clinicians should not routinely offer PFO closure outside of a research setting. Since then, three trials published in 2017 the New England Journal of Medicine (RESPECT, CLOSE, and REDUCE) and one trial published in 2018 in the Journal of the American College of Cardiology (DEFENSE-PFO) found that PFO closure reduces the risk of recurrent stroke in patients with a PFO who have had a cryptogenic stroke, compared with medical therapy alone. In addition, the Food and Drug Administration approved the Amplatzer PFO Occluder and Gore Cardioform Septal Occluder. These developments necessitated the practice advisory, the authors said. The advisory was published online April 29 in Neurology. It is endorsed by the American Heart Association/American Stroke Association, the Society for Cardiovascular Angiography and Interventions, and the European Academy of Neurology.
Systematic review
For the update, Steven R. Messé, MD, of the Hospital of the University of Pennsylvania in Philadelphia, and a panel of neurologists, internists, and cardiologists with expertise in stroke and PFO systematically reviewed relevant randomized studies published through August 2019 and conducted meta-analyses to make their recommendations. The literature search identified eight articles that met inclusion criteria, including one article that provided follow-up from a trial that had been included in the previous practice advisory.
“The risk of a second stroke in people with PFO and no other possible causes of stroke is very low, approximately 1% per year while being treated with just medication alone,” Dr. Messé said in a news release. “Also, it is difficult to determine with absolute certainty that the PFO is the cause of a person’s stroke. So it is important that people with PFO are educated about the benefits and risks of PFO closure.” For patients who opt to take medication only, doctors may consider prescribing antiplatelet or anticoagulant drugs, according to the advisory. “All patients with previous stroke should be treated with an antithrombotic medication indefinitely if there is no bleeding contraindication, regardless of whether a PFO is present or if it is closed,” Dr. Messé and colleagues wrote. “However, specific antithrombotic management for patients with stroke thought to be caused by PFO remains uncertain.”
Calls for thorough work-up
“If an alternative plausible higher-risk mechanism of stroke is identified, it is likely that the PFO was an innocent bystander,” the authors said. “Secondary stroke prevention is optimized by targeting the most likely etiology of the preceding event. ... The randomized PFO closure trials all mandated thorough evaluations for participants before enrollment ... to rule out other stroke mechanisms; moreover, all studies required TEE [transesophageal echocardiography] to characterize the PFO and ensure that it was the most likely etiology for the initial event.”
In patients being considered for PFO closure, clinicians should obtain brain imaging to confirm stroke size and distribution (level B); obtain vascular imaging of the cervical and intracranial vessels to look for dissection, vasculopathy, and atherosclerosis (level B); and perform hypercoagulable studies (level B), according to the advisory. Clinicians must perform a baseline ECG to look for atrial fibrillation (level A), and patients thought to be at risk of atrial fibrillation should receive prolonged cardiac monitoring for at least 28 days (level B).
Before PFO closure, a clinician with expertise in stroke should assess the patient to ensure that the PFO is the most plausible mechanism of stroke (level B). “If a higher-risk alternative mechanism of stroke is identified, clinicians should not routinely recommend PFO closure (level B),” the authors said. Patients also should be assessed by a clinician with expertise in assessing the anatomic features of a PFO and performing PFO closure (level B).
The randomized trials focused on patients whose PFOs were closed within 6 months of a stroke, and registry studies are needed to assess long-term outcomes, noted Dr. Messé and colleagues. “It remains unclear whether closure provides a similar benefit in these patients who otherwise still fit the studies’ inclusion criteria,” the authors said. “Long-term and large-scale safety registries for patients who have received PFO closure are needed to assess the risk of device erosion, fracture, embolization, and thrombotic and endocarditis risks, and the effect of residual shunts and incidence of atrial fibrillation.”
About 25% of the general adult population has a PFO. “It’s important to note that having a PFO is common, and that most people with PFO will never know they have it because it usually does not cause any problems,” Dr. Messé said. “However, while there is generally a very low risk of stroke in patients with PFO, in younger people who have had a stroke without any other possible causes identified, closing the PFO may reduce the risk of having another stroke better than medication alone.”
The practice advisory was developed with financial support from the AAN. Dr. Messé and most of the authors had no relevant conflicts of interest. Several authors disclosed ties to medical device and pharmaceutical companies.
SOURCE: Messé SR et al. Neurology. 2020 Apr 29. doi: 10.1212/WNL.0000000000009443.
FROM NEUROLOGY
Researchers identify a cause of L-DOPA–induced dyskinesia in Parkinson’s disease
The conclusion is based on animal studies that were published May 1 in Science Advances. “These studies show that, if we can downregulate RasGRP1 signaling before dopamine replacement, we have an opportunity to greatly improve [patients’] quality of life,” said Srinivasa Subramaniam, PhD, of the department of neuroscience at Scripps Research in Jupiter, Fla., in a press release. Dr. Subramaniam is one of the investigators.
Parkinson’s disease results from the loss of substantia nigral projections neurons, which causes decreased levels of dopamine in the dorsal striatum. Treatment with L-DOPA reduces the disease’s motor symptoms effectively, but ultimately leads to the onset of LID. Previous data suggest that LID results from the abnormal activation of dopamine-1 (D1)–dependent cyclic adenosine 3´,5´-monophosphate (cAMP)/protein kinase A (PKA), extracellular signal–regulated kinase (ERK), and mammalian target of rapamycin kinase complex 1 (mTORC1) signaling in the dorsal striatum.
Animal and biochemical data
Based on earlier animal studies, Dr. Subramaniam and colleagues hypothesized that RasGRP1 might regulate LID. To test this theory, the investigators created lesions in wild-type and RasGRP1 knockout mice to create models of Parkinson’s disease. The investigators saw similar Parkinsonian symptoms in both groups of mice on the drag, rotarod, turning, and open-field tests. After all mice received daily treatment with L-DOPA, RasGRP1 knockout mice had significantly fewer abnormal involuntary movements, compared with the wild-type mice. All aspects of dyskinesia appeared to be equally dampened in the knockout mice.
To analyze whether RasGRP1 deletion affected the efficacy of L-DOPA, the investigators subjected the treated mice to motor tests. Parkinsonian symptoms were decreased among wild-type and knockout mice on the drag and turning tests. “RasGRP1 promoted the adverse effects of L-DOPA but did not interfere with its therapeutic motor effects,” the investigators wrote. Compared with the wild-type mice, the knockout mice had no changes in basal motor behavior or coordination or amphetamine-induced motor activity.
In addition, Dr. Subramaniam and colleagues observed that RasGRP1 levels were increased in the striatum after L-DOPA injection, but not after injection of vehicle control. This and other biochemical findings indicated that striatal RasGRP1 is upregulated in an L-DOPA–dependent manner and is causally linked to the development of LID, according to the investigators.
Other observations indicated that RasGRP1 physiologically activates mTORC1 signaling, which contributes to LID. Using liquid chromatography and mass spectrometry, Dr. Subramaniam and colleagues saw that RasGRP1 acts upstream in response to L-DOPA and regulates a specific and diverse group of proteins to promote LID. When they examined a nonhuman primate model of Parkinson’s disease, they noted similar findings.
New therapeutic targets
“There is an immediate need for new therapeutic targets to stop LID ... in Parkinson’s disease,” said Dr. Subramaniam in a press release. “The treatments now available work poorly and have many additional unwanted side effects. We believe this [study] represents an important step toward better options for people with Parkinson’s disease.”
Future research should attempt to identify the best method of selectively reducing expression of RasGRP1 in the striatum without affecting its expression in other areas of the body, according to Dr. Subramaniam. “The good news is that in mice a total lack of RasGRP1 is not lethal, so we think that blocking RasGRP1 with drugs, or even with gene therapy, may have very few or no major side effects.”
The study was funded by grants from the National Institutes of Health. The investigators reported no conflicts of interest.
SOURCE: Eshraghi M et al. Sci Adv. 2020;6:eaaz7001.
The conclusion is based on animal studies that were published May 1 in Science Advances. “These studies show that, if we can downregulate RasGRP1 signaling before dopamine replacement, we have an opportunity to greatly improve [patients’] quality of life,” said Srinivasa Subramaniam, PhD, of the department of neuroscience at Scripps Research in Jupiter, Fla., in a press release. Dr. Subramaniam is one of the investigators.
Parkinson’s disease results from the loss of substantia nigral projections neurons, which causes decreased levels of dopamine in the dorsal striatum. Treatment with L-DOPA reduces the disease’s motor symptoms effectively, but ultimately leads to the onset of LID. Previous data suggest that LID results from the abnormal activation of dopamine-1 (D1)–dependent cyclic adenosine 3´,5´-monophosphate (cAMP)/protein kinase A (PKA), extracellular signal–regulated kinase (ERK), and mammalian target of rapamycin kinase complex 1 (mTORC1) signaling in the dorsal striatum.
Animal and biochemical data
Based on earlier animal studies, Dr. Subramaniam and colleagues hypothesized that RasGRP1 might regulate LID. To test this theory, the investigators created lesions in wild-type and RasGRP1 knockout mice to create models of Parkinson’s disease. The investigators saw similar Parkinsonian symptoms in both groups of mice on the drag, rotarod, turning, and open-field tests. After all mice received daily treatment with L-DOPA, RasGRP1 knockout mice had significantly fewer abnormal involuntary movements, compared with the wild-type mice. All aspects of dyskinesia appeared to be equally dampened in the knockout mice.
To analyze whether RasGRP1 deletion affected the efficacy of L-DOPA, the investigators subjected the treated mice to motor tests. Parkinsonian symptoms were decreased among wild-type and knockout mice on the drag and turning tests. “RasGRP1 promoted the adverse effects of L-DOPA but did not interfere with its therapeutic motor effects,” the investigators wrote. Compared with the wild-type mice, the knockout mice had no changes in basal motor behavior or coordination or amphetamine-induced motor activity.
In addition, Dr. Subramaniam and colleagues observed that RasGRP1 levels were increased in the striatum after L-DOPA injection, but not after injection of vehicle control. This and other biochemical findings indicated that striatal RasGRP1 is upregulated in an L-DOPA–dependent manner and is causally linked to the development of LID, according to the investigators.
Other observations indicated that RasGRP1 physiologically activates mTORC1 signaling, which contributes to LID. Using liquid chromatography and mass spectrometry, Dr. Subramaniam and colleagues saw that RasGRP1 acts upstream in response to L-DOPA and regulates a specific and diverse group of proteins to promote LID. When they examined a nonhuman primate model of Parkinson’s disease, they noted similar findings.
New therapeutic targets
“There is an immediate need for new therapeutic targets to stop LID ... in Parkinson’s disease,” said Dr. Subramaniam in a press release. “The treatments now available work poorly and have many additional unwanted side effects. We believe this [study] represents an important step toward better options for people with Parkinson’s disease.”
Future research should attempt to identify the best method of selectively reducing expression of RasGRP1 in the striatum without affecting its expression in other areas of the body, according to Dr. Subramaniam. “The good news is that in mice a total lack of RasGRP1 is not lethal, so we think that blocking RasGRP1 with drugs, or even with gene therapy, may have very few or no major side effects.”
The study was funded by grants from the National Institutes of Health. The investigators reported no conflicts of interest.
SOURCE: Eshraghi M et al. Sci Adv. 2020;6:eaaz7001.
The conclusion is based on animal studies that were published May 1 in Science Advances. “These studies show that, if we can downregulate RasGRP1 signaling before dopamine replacement, we have an opportunity to greatly improve [patients’] quality of life,” said Srinivasa Subramaniam, PhD, of the department of neuroscience at Scripps Research in Jupiter, Fla., in a press release. Dr. Subramaniam is one of the investigators.
Parkinson’s disease results from the loss of substantia nigral projections neurons, which causes decreased levels of dopamine in the dorsal striatum. Treatment with L-DOPA reduces the disease’s motor symptoms effectively, but ultimately leads to the onset of LID. Previous data suggest that LID results from the abnormal activation of dopamine-1 (D1)–dependent cyclic adenosine 3´,5´-monophosphate (cAMP)/protein kinase A (PKA), extracellular signal–regulated kinase (ERK), and mammalian target of rapamycin kinase complex 1 (mTORC1) signaling in the dorsal striatum.
Animal and biochemical data
Based on earlier animal studies, Dr. Subramaniam and colleagues hypothesized that RasGRP1 might regulate LID. To test this theory, the investigators created lesions in wild-type and RasGRP1 knockout mice to create models of Parkinson’s disease. The investigators saw similar Parkinsonian symptoms in both groups of mice on the drag, rotarod, turning, and open-field tests. After all mice received daily treatment with L-DOPA, RasGRP1 knockout mice had significantly fewer abnormal involuntary movements, compared with the wild-type mice. All aspects of dyskinesia appeared to be equally dampened in the knockout mice.
To analyze whether RasGRP1 deletion affected the efficacy of L-DOPA, the investigators subjected the treated mice to motor tests. Parkinsonian symptoms were decreased among wild-type and knockout mice on the drag and turning tests. “RasGRP1 promoted the adverse effects of L-DOPA but did not interfere with its therapeutic motor effects,” the investigators wrote. Compared with the wild-type mice, the knockout mice had no changes in basal motor behavior or coordination or amphetamine-induced motor activity.
In addition, Dr. Subramaniam and colleagues observed that RasGRP1 levels were increased in the striatum after L-DOPA injection, but not after injection of vehicle control. This and other biochemical findings indicated that striatal RasGRP1 is upregulated in an L-DOPA–dependent manner and is causally linked to the development of LID, according to the investigators.
Other observations indicated that RasGRP1 physiologically activates mTORC1 signaling, which contributes to LID. Using liquid chromatography and mass spectrometry, Dr. Subramaniam and colleagues saw that RasGRP1 acts upstream in response to L-DOPA and regulates a specific and diverse group of proteins to promote LID. When they examined a nonhuman primate model of Parkinson’s disease, they noted similar findings.
New therapeutic targets
“There is an immediate need for new therapeutic targets to stop LID ... in Parkinson’s disease,” said Dr. Subramaniam in a press release. “The treatments now available work poorly and have many additional unwanted side effects. We believe this [study] represents an important step toward better options for people with Parkinson’s disease.”
Future research should attempt to identify the best method of selectively reducing expression of RasGRP1 in the striatum without affecting its expression in other areas of the body, according to Dr. Subramaniam. “The good news is that in mice a total lack of RasGRP1 is not lethal, so we think that blocking RasGRP1 with drugs, or even with gene therapy, may have very few or no major side effects.”
The study was funded by grants from the National Institutes of Health. The investigators reported no conflicts of interest.
SOURCE: Eshraghi M et al. Sci Adv. 2020;6:eaaz7001.
FROM Science Advances
Pandemic necessitates new strategies to treat migraine
Patients with migraine who are unable to continue preventive procedures such as onabotulinumtoxinA injections during the COVID-19 pandemic may be at risk of worsening migraine, according to an article published March 30 in Headache. To address this scenario, including monoclonal antibodies against calcitonin gene-related peptide (CGRP) or the CGRP receptor, the authors said.
“This is a particularly vulnerable time for individuals with migraine and other disabling headache disorders, with many physical and mental stressors, increased anxiety, and changes in daily routine which may serve as triggering factors for worsening headache,” said lead author Christina L. Szperka, MD, director of the pediatric headache program at Children’s Hospital of Philadelphia, and colleagues.
Acute treatment
The authors described potential treatment regimens based on their experience as headache specialists and the experiences of their colleagues. For acute therapy options, NSAIDs, triptans, and neuroleptics may be used in combination when needed. Medications within the same drug category should not be combined, however, and triptans, dihydroergotamine, and lasmiditan should not be coadministered within 24 hours. Since the 2015 American Headache Society guideline for the acute treatment of migraine, the Food and Drug Administration has approved additional acute migraine medications, including ubrogepant, rimegepant, and lasmiditan, the authors said. The agency also cleared several neuromodulation devices for the acute treatment of migraine.
Although few drugs have been studied as treatments for unusually prolonged severe headaches, headache doctors often recommend NSAIDs before patients seek care at an emergency department or infusion center, the authors said. NSAID options include indomethacin, ketorolac, naproxen, nabumetone, diclofenac, and mefenamic acid. Neuroleptics also may be used. “Long-acting triptan medications can be used as bridge therapies, as is often done in the treatment of menstrually related migraine or in the treatment of medication overuse headache,” they said. “We propose a similar strategy can be trialed as a therapeutic option for refractory or persistent migraine.”
The authors also described the use of antiepileptics and corticosteroids, as well as drugs that may treat specific symptoms, such as difficulty sleeping (hydroxyzine or amitriptyline), neck or muscle pain (tizanidine), and aura with migraine (magnesium). Clinicians should avoid the use of opioids and butalbital, they said.
Preventive treatment
“While the injection of onabotulinumtoxinA is an effective treatment for chronic migraine, the procedure can put the patient and the provider at higher risk of COVID-19 given the close contact encounter,” wrote Dr. Szperka and colleagues. “We believe that other migraine preventive treatments should be utilized first when possible.” Since the publication of a guideline on preventive migraine therapies in 2012, the FDA has approved additional preventive therapies, including the anti-CGRP monoclonal antibodies erenumab‐aooe, galcanezumab‐gnlm, fremanezumab‐vfrm, and eptinezumab‐jjmr. “The first three are intended for self‐injection at home, with detailed instructions available for each product on its website,” they said.
Among angiotensin‐converting enzyme (ACE) inhibitors or angiotensin receptor blockers (ARBs), candesartan has evidence of efficacy and tolerability in migraine prevention. Lisinopril has been considered possibly effective. “There has been recent concern in the media about the possibility of these medications interfering with the body’s response to COVID‐19,” the authors said, although this theoretical concern was not based on experimental or clinical data. “For patients in need of a new preventive therapy, the potential for benefit with an ACE/ARB must be weighed against the theoretical increased risk of infection.”
In addition, studies indicate that melatonin may prevent migraine with few side effects and that zonisamide may be effective in patients who have an inadequate response to or experience side effects with topiramate.
Policy changes and telehealth options
Effectively treating patients with migraine during the pandemic requires policy changes, according to the authors. “Migraine preventive prior authorization restrictions need to be lifted for evidence‐based, FDA‐approved therapies; patients need to be able to access these medications quickly and easily. Patients should not be required to fail older medications,” they said. “Similarly, in order to permit the transition of patients from onabotulinumtoxinA to anti‐CGRP [monoclonal antibodies], insurers should remove the prohibition against simultaneous coverage of these drug classes.” Insurers also should loosen restrictions on the off-label use of acute and preventive medication for adolescents, Dr. Szperka and coauthors suggest.
“In the era of COVID‐19, telehealth has become an essential modality for most headache specialists, given the need for providers to take significant precautions for both their patients and themselves, limiting touch or close contact,” they said. Patients with headache may warrant additional screening for COVID-19 as well. “As headache has been reported as an early symptom of COVID‐19, patients with worsening or new onset severe headache should be reviewed for exposure risk and any other symptoms which may be consistent with COVID‐19 infection,” the authors said.
There was no direct funding for the report. Dr. Szperka and a coauthor receive salary support from the National Institutes of Health. Dr. Szperka also has received grant support from Pfizer, and her institution has received compensation for her consulting work for Allergan. Several coauthors disclosed consulting and serving on speakers’ bureaus for and receiving research support from various pharmaceutical companies.
SOURCE: Szperka CL et al. Headache. 2020 Mar 30. doi: 10.1111/head.13810.
Patients with migraine who are unable to continue preventive procedures such as onabotulinumtoxinA injections during the COVID-19 pandemic may be at risk of worsening migraine, according to an article published March 30 in Headache. To address this scenario, including monoclonal antibodies against calcitonin gene-related peptide (CGRP) or the CGRP receptor, the authors said.
“This is a particularly vulnerable time for individuals with migraine and other disabling headache disorders, with many physical and mental stressors, increased anxiety, and changes in daily routine which may serve as triggering factors for worsening headache,” said lead author Christina L. Szperka, MD, director of the pediatric headache program at Children’s Hospital of Philadelphia, and colleagues.
Acute treatment
The authors described potential treatment regimens based on their experience as headache specialists and the experiences of their colleagues. For acute therapy options, NSAIDs, triptans, and neuroleptics may be used in combination when needed. Medications within the same drug category should not be combined, however, and triptans, dihydroergotamine, and lasmiditan should not be coadministered within 24 hours. Since the 2015 American Headache Society guideline for the acute treatment of migraine, the Food and Drug Administration has approved additional acute migraine medications, including ubrogepant, rimegepant, and lasmiditan, the authors said. The agency also cleared several neuromodulation devices for the acute treatment of migraine.
Although few drugs have been studied as treatments for unusually prolonged severe headaches, headache doctors often recommend NSAIDs before patients seek care at an emergency department or infusion center, the authors said. NSAID options include indomethacin, ketorolac, naproxen, nabumetone, diclofenac, and mefenamic acid. Neuroleptics also may be used. “Long-acting triptan medications can be used as bridge therapies, as is often done in the treatment of menstrually related migraine or in the treatment of medication overuse headache,” they said. “We propose a similar strategy can be trialed as a therapeutic option for refractory or persistent migraine.”
The authors also described the use of antiepileptics and corticosteroids, as well as drugs that may treat specific symptoms, such as difficulty sleeping (hydroxyzine or amitriptyline), neck or muscle pain (tizanidine), and aura with migraine (magnesium). Clinicians should avoid the use of opioids and butalbital, they said.
Preventive treatment
“While the injection of onabotulinumtoxinA is an effective treatment for chronic migraine, the procedure can put the patient and the provider at higher risk of COVID-19 given the close contact encounter,” wrote Dr. Szperka and colleagues. “We believe that other migraine preventive treatments should be utilized first when possible.” Since the publication of a guideline on preventive migraine therapies in 2012, the FDA has approved additional preventive therapies, including the anti-CGRP monoclonal antibodies erenumab‐aooe, galcanezumab‐gnlm, fremanezumab‐vfrm, and eptinezumab‐jjmr. “The first three are intended for self‐injection at home, with detailed instructions available for each product on its website,” they said.
Among angiotensin‐converting enzyme (ACE) inhibitors or angiotensin receptor blockers (ARBs), candesartan has evidence of efficacy and tolerability in migraine prevention. Lisinopril has been considered possibly effective. “There has been recent concern in the media about the possibility of these medications interfering with the body’s response to COVID‐19,” the authors said, although this theoretical concern was not based on experimental or clinical data. “For patients in need of a new preventive therapy, the potential for benefit with an ACE/ARB must be weighed against the theoretical increased risk of infection.”
In addition, studies indicate that melatonin may prevent migraine with few side effects and that zonisamide may be effective in patients who have an inadequate response to or experience side effects with topiramate.
Policy changes and telehealth options
Effectively treating patients with migraine during the pandemic requires policy changes, according to the authors. “Migraine preventive prior authorization restrictions need to be lifted for evidence‐based, FDA‐approved therapies; patients need to be able to access these medications quickly and easily. Patients should not be required to fail older medications,” they said. “Similarly, in order to permit the transition of patients from onabotulinumtoxinA to anti‐CGRP [monoclonal antibodies], insurers should remove the prohibition against simultaneous coverage of these drug classes.” Insurers also should loosen restrictions on the off-label use of acute and preventive medication for adolescents, Dr. Szperka and coauthors suggest.
“In the era of COVID‐19, telehealth has become an essential modality for most headache specialists, given the need for providers to take significant precautions for both their patients and themselves, limiting touch or close contact,” they said. Patients with headache may warrant additional screening for COVID-19 as well. “As headache has been reported as an early symptom of COVID‐19, patients with worsening or new onset severe headache should be reviewed for exposure risk and any other symptoms which may be consistent with COVID‐19 infection,” the authors said.
There was no direct funding for the report. Dr. Szperka and a coauthor receive salary support from the National Institutes of Health. Dr. Szperka also has received grant support from Pfizer, and her institution has received compensation for her consulting work for Allergan. Several coauthors disclosed consulting and serving on speakers’ bureaus for and receiving research support from various pharmaceutical companies.
SOURCE: Szperka CL et al. Headache. 2020 Mar 30. doi: 10.1111/head.13810.
Patients with migraine who are unable to continue preventive procedures such as onabotulinumtoxinA injections during the COVID-19 pandemic may be at risk of worsening migraine, according to an article published March 30 in Headache. To address this scenario, including monoclonal antibodies against calcitonin gene-related peptide (CGRP) or the CGRP receptor, the authors said.
“This is a particularly vulnerable time for individuals with migraine and other disabling headache disorders, with many physical and mental stressors, increased anxiety, and changes in daily routine which may serve as triggering factors for worsening headache,” said lead author Christina L. Szperka, MD, director of the pediatric headache program at Children’s Hospital of Philadelphia, and colleagues.
Acute treatment
The authors described potential treatment regimens based on their experience as headache specialists and the experiences of their colleagues. For acute therapy options, NSAIDs, triptans, and neuroleptics may be used in combination when needed. Medications within the same drug category should not be combined, however, and triptans, dihydroergotamine, and lasmiditan should not be coadministered within 24 hours. Since the 2015 American Headache Society guideline for the acute treatment of migraine, the Food and Drug Administration has approved additional acute migraine medications, including ubrogepant, rimegepant, and lasmiditan, the authors said. The agency also cleared several neuromodulation devices for the acute treatment of migraine.
Although few drugs have been studied as treatments for unusually prolonged severe headaches, headache doctors often recommend NSAIDs before patients seek care at an emergency department or infusion center, the authors said. NSAID options include indomethacin, ketorolac, naproxen, nabumetone, diclofenac, and mefenamic acid. Neuroleptics also may be used. “Long-acting triptan medications can be used as bridge therapies, as is often done in the treatment of menstrually related migraine or in the treatment of medication overuse headache,” they said. “We propose a similar strategy can be trialed as a therapeutic option for refractory or persistent migraine.”
The authors also described the use of antiepileptics and corticosteroids, as well as drugs that may treat specific symptoms, such as difficulty sleeping (hydroxyzine or amitriptyline), neck or muscle pain (tizanidine), and aura with migraine (magnesium). Clinicians should avoid the use of opioids and butalbital, they said.
Preventive treatment
“While the injection of onabotulinumtoxinA is an effective treatment for chronic migraine, the procedure can put the patient and the provider at higher risk of COVID-19 given the close contact encounter,” wrote Dr. Szperka and colleagues. “We believe that other migraine preventive treatments should be utilized first when possible.” Since the publication of a guideline on preventive migraine therapies in 2012, the FDA has approved additional preventive therapies, including the anti-CGRP monoclonal antibodies erenumab‐aooe, galcanezumab‐gnlm, fremanezumab‐vfrm, and eptinezumab‐jjmr. “The first three are intended for self‐injection at home, with detailed instructions available for each product on its website,” they said.
Among angiotensin‐converting enzyme (ACE) inhibitors or angiotensin receptor blockers (ARBs), candesartan has evidence of efficacy and tolerability in migraine prevention. Lisinopril has been considered possibly effective. “There has been recent concern in the media about the possibility of these medications interfering with the body’s response to COVID‐19,” the authors said, although this theoretical concern was not based on experimental or clinical data. “For patients in need of a new preventive therapy, the potential for benefit with an ACE/ARB must be weighed against the theoretical increased risk of infection.”
In addition, studies indicate that melatonin may prevent migraine with few side effects and that zonisamide may be effective in patients who have an inadequate response to or experience side effects with topiramate.
Policy changes and telehealth options
Effectively treating patients with migraine during the pandemic requires policy changes, according to the authors. “Migraine preventive prior authorization restrictions need to be lifted for evidence‐based, FDA‐approved therapies; patients need to be able to access these medications quickly and easily. Patients should not be required to fail older medications,” they said. “Similarly, in order to permit the transition of patients from onabotulinumtoxinA to anti‐CGRP [monoclonal antibodies], insurers should remove the prohibition against simultaneous coverage of these drug classes.” Insurers also should loosen restrictions on the off-label use of acute and preventive medication for adolescents, Dr. Szperka and coauthors suggest.
“In the era of COVID‐19, telehealth has become an essential modality for most headache specialists, given the need for providers to take significant precautions for both their patients and themselves, limiting touch or close contact,” they said. Patients with headache may warrant additional screening for COVID-19 as well. “As headache has been reported as an early symptom of COVID‐19, patients with worsening or new onset severe headache should be reviewed for exposure risk and any other symptoms which may be consistent with COVID‐19 infection,” the authors said.
There was no direct funding for the report. Dr. Szperka and a coauthor receive salary support from the National Institutes of Health. Dr. Szperka also has received grant support from Pfizer, and her institution has received compensation for her consulting work for Allergan. Several coauthors disclosed consulting and serving on speakers’ bureaus for and receiving research support from various pharmaceutical companies.
SOURCE: Szperka CL et al. Headache. 2020 Mar 30. doi: 10.1111/head.13810.
FROM HEADACHE
Amid coronavirus concerns, researchers urge mental health interventions for patients with dementia
according to a letter published online ahead of print March 30 in Lancet. Consistent with recommendations from Alzheimer’s Disease International and other dementia experts, teams that include mental health professionals, social workers, nursing home administrators, and volunteers should collaborate to provide mental health care for people with dementia. Experts in dementia should lead each team and support team members from other disciplines, wrote Huali Wang, MD, chair of clinical research at Peking University Institute of Mental Health in Beijing, and colleagues.
Interventions could be administered through telehealth, said the authors. Teams led by mental health professionals could use electronic media to provide self-help guidance for reducing stress, such as relaxation or meditation exercise. These teams also could use telephone hotlines to support behavioral management, and psychological counselors could provide online consultations for caregivers in nursing homes or in the community. “We encourage people who have a parent with dementia to have more frequent contact or spend more time with their parent, or to take on some of the caregiving duties so as to give the carer some respite time,” wrote Dr. Wang and colleagues.
Many local authorities are banning visits to nursing home residents to reduce the latter’s risk of COVID-19 infection. As a consequence, these elderly people are becoming more isolated, and anxiety is increasing among nursing home staffs.
In China, five organizations, including the Chinese Society of Geriatric Psychiatry and Alzheimer’s Disease Chinese, responded to the COVID-19 outbreak by publishing recommendations for providing mental health and psychosocial support. Groups of providers from various disciplines offered free counseling services for people with dementia and their caregivers. “These approaches minimized the complex impact of both COVID-19 outbreak and dementia,” wrote the authors.
“China has contained the epidemic, and business is starting to return to normal,” they continued. “We believe that learning lessons from China would empower the world to tackle the COVID-19 pandemic, with little risk of compromising the quality of life of people living with dementia and their carers.”
Dr. Wang has received lecture fees from Eisai China and Lundbeck China. She owns the copyright for the neuropsychiatric symptoms individualized management system. Her coauthors reported serving as advisory board members and receiving fees from companies such as Biogen, Novartis, and Genentech.
SOURCE: Wang H et al. Lancet. 2020 Mar 30. doi: 10.1016/S0140-6736(20)30755-8.
according to a letter published online ahead of print March 30 in Lancet. Consistent with recommendations from Alzheimer’s Disease International and other dementia experts, teams that include mental health professionals, social workers, nursing home administrators, and volunteers should collaborate to provide mental health care for people with dementia. Experts in dementia should lead each team and support team members from other disciplines, wrote Huali Wang, MD, chair of clinical research at Peking University Institute of Mental Health in Beijing, and colleagues.
Interventions could be administered through telehealth, said the authors. Teams led by mental health professionals could use electronic media to provide self-help guidance for reducing stress, such as relaxation or meditation exercise. These teams also could use telephone hotlines to support behavioral management, and psychological counselors could provide online consultations for caregivers in nursing homes or in the community. “We encourage people who have a parent with dementia to have more frequent contact or spend more time with their parent, or to take on some of the caregiving duties so as to give the carer some respite time,” wrote Dr. Wang and colleagues.
Many local authorities are banning visits to nursing home residents to reduce the latter’s risk of COVID-19 infection. As a consequence, these elderly people are becoming more isolated, and anxiety is increasing among nursing home staffs.
In China, five organizations, including the Chinese Society of Geriatric Psychiatry and Alzheimer’s Disease Chinese, responded to the COVID-19 outbreak by publishing recommendations for providing mental health and psychosocial support. Groups of providers from various disciplines offered free counseling services for people with dementia and their caregivers. “These approaches minimized the complex impact of both COVID-19 outbreak and dementia,” wrote the authors.
“China has contained the epidemic, and business is starting to return to normal,” they continued. “We believe that learning lessons from China would empower the world to tackle the COVID-19 pandemic, with little risk of compromising the quality of life of people living with dementia and their carers.”
Dr. Wang has received lecture fees from Eisai China and Lundbeck China. She owns the copyright for the neuropsychiatric symptoms individualized management system. Her coauthors reported serving as advisory board members and receiving fees from companies such as Biogen, Novartis, and Genentech.
SOURCE: Wang H et al. Lancet. 2020 Mar 30. doi: 10.1016/S0140-6736(20)30755-8.
according to a letter published online ahead of print March 30 in Lancet. Consistent with recommendations from Alzheimer’s Disease International and other dementia experts, teams that include mental health professionals, social workers, nursing home administrators, and volunteers should collaborate to provide mental health care for people with dementia. Experts in dementia should lead each team and support team members from other disciplines, wrote Huali Wang, MD, chair of clinical research at Peking University Institute of Mental Health in Beijing, and colleagues.
Interventions could be administered through telehealth, said the authors. Teams led by mental health professionals could use electronic media to provide self-help guidance for reducing stress, such as relaxation or meditation exercise. These teams also could use telephone hotlines to support behavioral management, and psychological counselors could provide online consultations for caregivers in nursing homes or in the community. “We encourage people who have a parent with dementia to have more frequent contact or spend more time with their parent, or to take on some of the caregiving duties so as to give the carer some respite time,” wrote Dr. Wang and colleagues.
Many local authorities are banning visits to nursing home residents to reduce the latter’s risk of COVID-19 infection. As a consequence, these elderly people are becoming more isolated, and anxiety is increasing among nursing home staffs.
In China, five organizations, including the Chinese Society of Geriatric Psychiatry and Alzheimer’s Disease Chinese, responded to the COVID-19 outbreak by publishing recommendations for providing mental health and psychosocial support. Groups of providers from various disciplines offered free counseling services for people with dementia and their caregivers. “These approaches minimized the complex impact of both COVID-19 outbreak and dementia,” wrote the authors.
“China has contained the epidemic, and business is starting to return to normal,” they continued. “We believe that learning lessons from China would empower the world to tackle the COVID-19 pandemic, with little risk of compromising the quality of life of people living with dementia and their carers.”
Dr. Wang has received lecture fees from Eisai China and Lundbeck China. She owns the copyright for the neuropsychiatric symptoms individualized management system. Her coauthors reported serving as advisory board members and receiving fees from companies such as Biogen, Novartis, and Genentech.
SOURCE: Wang H et al. Lancet. 2020 Mar 30. doi: 10.1016/S0140-6736(20)30755-8.
REPORTING FROM THE LANCET
CBT by phone reduces depression in Parkinson’s disease
, according to trial results published in Neurology. The treatment’s effect on depression is “moderated by the reduction of negative thoughts,” the target of the intervention, the researchers said.
Telephone-based CBT may be a convenient option for patients, said lead study author Roseanne D. Dobkin, PhD, of the department of psychiatry at Rutgers Robert Wood Johnson Medical School in Piscataway, N.J., and the VA New Jersey Health Care System in Lyons. “A notable proportion of people with Parkinson’s [disease] do not receive the much needed mental health treatment to facilitate proactive coping with the daily challenges superimposed by their medical condition,” Dr. Dobkin said in a news release. “This study suggests that the effects of the [CBT] last long beyond when the treatment stopped and can be used alongside standard neurological care.”
An undertreated problem
Although depression affects about half of patients with Parkinson’s disease and is associated with physical and cognitive decline, it often goes overlooked and undertreated, the study authors said. Data about the efficacy and tolerability of antidepressants are mixed. CBT holds promise for reducing depression in Parkinson’s disease, prior research suggests, but patients may have limited access to in-person sessions because of physical and geographic barriers.
To assess the efficacy of telephone-based CBT for depression in Parkinson’s disease, compared with community-based treatment as usual, Dr. Dobkin and colleagues conducted a randomized controlled trial. Their study included 72 patients with Parkinson’s disease at an academic medical center. Participants had a depressive disorder, were between aged 35 and 85 years, had stable Parkinson’s disease and mental health treatment for at least 6 weeks, and had a family member or friend willing to participate in the study. The investigators excluded patients with possible dementia or marked cognitive impairment and active suicidal plans or intent.
Participants were randomly assigned to receive usual care plus telephone-based CBT or usual care only. Patients taking antidepressants were evenly divided between the groups.
Telephone-based CBT consisted of weekly 1-hour sessions for 10 weeks. During 6 months of follow-up, patients could receive one session per month if desired. The CBT “targeted negative thoughts (e.g., ‘I have no control’; ‘I am helpless’) and behaviors (e.g., avoidance, excessive worry, lack of exercise),” the investigators said. In addition, therapists trained patients’ care partners by telephone to help patients between sessions. Treatment as usual was defined by patients’ health care teams. For most participants in both groups, treatment as usual included taking antidepressant medication or receiving psychotherapy in the community.
Change in Hamilton Depression Rating Scale (HAM-D) score was the primary outcome. Secondary outcomes included whether patients considered their depression much improved and improvements in depression severity (as measured by the Beck Depression Inventory [BDI]), anxiety (as measured by the Hamilton Anxiety Rating Scale [HAM-A]), and quality of life. The researchers also assessed negative thinking using the Inference Questionnaire. Blinded raters assessed outcomes.
Sustained improvements
Thirty-seven patients were randomized to receive telephone-based CBT, and 35 were randomized to treatment as usual. Overall, 70% were taking antidepressants, and 14% continued receiving psychotherapy from community providers of their choice during the trial. Participants’ average age was 65 years, and 51% were female.
Post treatment, mean improvement in HAM-D score from baseline was 6.53 points in the telephone-based CBT group, compared with −0.27 points in the control group. “Effects at the end of treatment were maintained at 6-month follow-up,” the researchers reported.
About 40% of patients in the CBT group reported that their depression was much improved or very much improved, compared with none of the patients in the control group. Responders had mild to minimal symptomatology on the HAM-D, which indicates that the changes were clinically significant, the authors said.
Secondary outcomes also favored telephone-based CBT. “The intervention was feasible and highly acceptable, yielding an 88% retention rate over the 9-month trial,” Dr. Dobkin and colleagues said.
Compared with other control conditions, treatment-as-usual controls may enhance the effect size of an intervention, the authors noted. In addition, factors such as therapeutic relationship, time, and attention likely contribute to psychotherapy outcomes.
Success may hinge on cognitive ability
“The success of this trial highlights the need for further efficacy studies targeting neuropsychiatric manifestations of [Parkinson’s disease] and adds urgency to the discussion over policies regarding access to tele–mental health, especially for vulnerable populations with limited access to in-person mental health services,” Gregory M. Pontone, MD, and Kelly A. Mills, MD, wrote in an accompanying editorial. Dr. Pontone and Dr. Mills are affiliated with Johns Hopkins University in Baltimore.
“Only rudimentary evidence” exists to guide the treatment of depression in patients with Parkinson’s disease, the editorialists said. “Patient preference and tolerability suggest that nonpharmacologic therapies, such as CBT, are preferred as first-line treatment. Yet access to qualified CBT practitioners, especially those with a clinical knowledge of [Parkinson’s disease], is limited.”
Despite its advantages and the encouraging results, CBT may have important limitations as well, they said. Patients require a certain degree of cognitive ability to benefit from CBT, and the prevalence of dementia among patients with Parkinson’s disease is about 30%.
Nevertheless, the trial provided evidence of target engagement. “Though caveats include the single-blind design and potential confounding by time spent with patient and caregiver, the authors demonstrated that improvement was mediated by the mechanism of CBT – a reduction in negative thinking.”
The trial was funded by the Michael J. Fox Foundation for Parkinson’s Research and the Parkinson’s Alliance (Parkinson’s Unity Walk). Dr. Mills disclosed a patent pending for a system for phase-dependent cortical brain stimulation, National Institutes of Health funding, pending funding from the Michael J. Fox Foundation, and commercial research support from Global Kinetics Corporation. Dr. Pontone is a consultant for Acadia Pharmaceuticals.
SOURCE: Dobkin RD et al. Neurology. 2020 Apr 1. doi: 10.1212/WNL.0000000000009292.
, according to trial results published in Neurology. The treatment’s effect on depression is “moderated by the reduction of negative thoughts,” the target of the intervention, the researchers said.
Telephone-based CBT may be a convenient option for patients, said lead study author Roseanne D. Dobkin, PhD, of the department of psychiatry at Rutgers Robert Wood Johnson Medical School in Piscataway, N.J., and the VA New Jersey Health Care System in Lyons. “A notable proportion of people with Parkinson’s [disease] do not receive the much needed mental health treatment to facilitate proactive coping with the daily challenges superimposed by their medical condition,” Dr. Dobkin said in a news release. “This study suggests that the effects of the [CBT] last long beyond when the treatment stopped and can be used alongside standard neurological care.”
An undertreated problem
Although depression affects about half of patients with Parkinson’s disease and is associated with physical and cognitive decline, it often goes overlooked and undertreated, the study authors said. Data about the efficacy and tolerability of antidepressants are mixed. CBT holds promise for reducing depression in Parkinson’s disease, prior research suggests, but patients may have limited access to in-person sessions because of physical and geographic barriers.
To assess the efficacy of telephone-based CBT for depression in Parkinson’s disease, compared with community-based treatment as usual, Dr. Dobkin and colleagues conducted a randomized controlled trial. Their study included 72 patients with Parkinson’s disease at an academic medical center. Participants had a depressive disorder, were between aged 35 and 85 years, had stable Parkinson’s disease and mental health treatment for at least 6 weeks, and had a family member or friend willing to participate in the study. The investigators excluded patients with possible dementia or marked cognitive impairment and active suicidal plans or intent.
Participants were randomly assigned to receive usual care plus telephone-based CBT or usual care only. Patients taking antidepressants were evenly divided between the groups.
Telephone-based CBT consisted of weekly 1-hour sessions for 10 weeks. During 6 months of follow-up, patients could receive one session per month if desired. The CBT “targeted negative thoughts (e.g., ‘I have no control’; ‘I am helpless’) and behaviors (e.g., avoidance, excessive worry, lack of exercise),” the investigators said. In addition, therapists trained patients’ care partners by telephone to help patients between sessions. Treatment as usual was defined by patients’ health care teams. For most participants in both groups, treatment as usual included taking antidepressant medication or receiving psychotherapy in the community.
Change in Hamilton Depression Rating Scale (HAM-D) score was the primary outcome. Secondary outcomes included whether patients considered their depression much improved and improvements in depression severity (as measured by the Beck Depression Inventory [BDI]), anxiety (as measured by the Hamilton Anxiety Rating Scale [HAM-A]), and quality of life. The researchers also assessed negative thinking using the Inference Questionnaire. Blinded raters assessed outcomes.
Sustained improvements
Thirty-seven patients were randomized to receive telephone-based CBT, and 35 were randomized to treatment as usual. Overall, 70% were taking antidepressants, and 14% continued receiving psychotherapy from community providers of their choice during the trial. Participants’ average age was 65 years, and 51% were female.
Post treatment, mean improvement in HAM-D score from baseline was 6.53 points in the telephone-based CBT group, compared with −0.27 points in the control group. “Effects at the end of treatment were maintained at 6-month follow-up,” the researchers reported.
About 40% of patients in the CBT group reported that their depression was much improved or very much improved, compared with none of the patients in the control group. Responders had mild to minimal symptomatology on the HAM-D, which indicates that the changes were clinically significant, the authors said.
Secondary outcomes also favored telephone-based CBT. “The intervention was feasible and highly acceptable, yielding an 88% retention rate over the 9-month trial,” Dr. Dobkin and colleagues said.
Compared with other control conditions, treatment-as-usual controls may enhance the effect size of an intervention, the authors noted. In addition, factors such as therapeutic relationship, time, and attention likely contribute to psychotherapy outcomes.
Success may hinge on cognitive ability
“The success of this trial highlights the need for further efficacy studies targeting neuropsychiatric manifestations of [Parkinson’s disease] and adds urgency to the discussion over policies regarding access to tele–mental health, especially for vulnerable populations with limited access to in-person mental health services,” Gregory M. Pontone, MD, and Kelly A. Mills, MD, wrote in an accompanying editorial. Dr. Pontone and Dr. Mills are affiliated with Johns Hopkins University in Baltimore.
“Only rudimentary evidence” exists to guide the treatment of depression in patients with Parkinson’s disease, the editorialists said. “Patient preference and tolerability suggest that nonpharmacologic therapies, such as CBT, are preferred as first-line treatment. Yet access to qualified CBT practitioners, especially those with a clinical knowledge of [Parkinson’s disease], is limited.”
Despite its advantages and the encouraging results, CBT may have important limitations as well, they said. Patients require a certain degree of cognitive ability to benefit from CBT, and the prevalence of dementia among patients with Parkinson’s disease is about 30%.
Nevertheless, the trial provided evidence of target engagement. “Though caveats include the single-blind design and potential confounding by time spent with patient and caregiver, the authors demonstrated that improvement was mediated by the mechanism of CBT – a reduction in negative thinking.”
The trial was funded by the Michael J. Fox Foundation for Parkinson’s Research and the Parkinson’s Alliance (Parkinson’s Unity Walk). Dr. Mills disclosed a patent pending for a system for phase-dependent cortical brain stimulation, National Institutes of Health funding, pending funding from the Michael J. Fox Foundation, and commercial research support from Global Kinetics Corporation. Dr. Pontone is a consultant for Acadia Pharmaceuticals.
SOURCE: Dobkin RD et al. Neurology. 2020 Apr 1. doi: 10.1212/WNL.0000000000009292.
, according to trial results published in Neurology. The treatment’s effect on depression is “moderated by the reduction of negative thoughts,” the target of the intervention, the researchers said.
Telephone-based CBT may be a convenient option for patients, said lead study author Roseanne D. Dobkin, PhD, of the department of psychiatry at Rutgers Robert Wood Johnson Medical School in Piscataway, N.J., and the VA New Jersey Health Care System in Lyons. “A notable proportion of people with Parkinson’s [disease] do not receive the much needed mental health treatment to facilitate proactive coping with the daily challenges superimposed by their medical condition,” Dr. Dobkin said in a news release. “This study suggests that the effects of the [CBT] last long beyond when the treatment stopped and can be used alongside standard neurological care.”
An undertreated problem
Although depression affects about half of patients with Parkinson’s disease and is associated with physical and cognitive decline, it often goes overlooked and undertreated, the study authors said. Data about the efficacy and tolerability of antidepressants are mixed. CBT holds promise for reducing depression in Parkinson’s disease, prior research suggests, but patients may have limited access to in-person sessions because of physical and geographic barriers.
To assess the efficacy of telephone-based CBT for depression in Parkinson’s disease, compared with community-based treatment as usual, Dr. Dobkin and colleagues conducted a randomized controlled trial. Their study included 72 patients with Parkinson’s disease at an academic medical center. Participants had a depressive disorder, were between aged 35 and 85 years, had stable Parkinson’s disease and mental health treatment for at least 6 weeks, and had a family member or friend willing to participate in the study. The investigators excluded patients with possible dementia or marked cognitive impairment and active suicidal plans or intent.
Participants were randomly assigned to receive usual care plus telephone-based CBT or usual care only. Patients taking antidepressants were evenly divided between the groups.
Telephone-based CBT consisted of weekly 1-hour sessions for 10 weeks. During 6 months of follow-up, patients could receive one session per month if desired. The CBT “targeted negative thoughts (e.g., ‘I have no control’; ‘I am helpless’) and behaviors (e.g., avoidance, excessive worry, lack of exercise),” the investigators said. In addition, therapists trained patients’ care partners by telephone to help patients between sessions. Treatment as usual was defined by patients’ health care teams. For most participants in both groups, treatment as usual included taking antidepressant medication or receiving psychotherapy in the community.
Change in Hamilton Depression Rating Scale (HAM-D) score was the primary outcome. Secondary outcomes included whether patients considered their depression much improved and improvements in depression severity (as measured by the Beck Depression Inventory [BDI]), anxiety (as measured by the Hamilton Anxiety Rating Scale [HAM-A]), and quality of life. The researchers also assessed negative thinking using the Inference Questionnaire. Blinded raters assessed outcomes.
Sustained improvements
Thirty-seven patients were randomized to receive telephone-based CBT, and 35 were randomized to treatment as usual. Overall, 70% were taking antidepressants, and 14% continued receiving psychotherapy from community providers of their choice during the trial. Participants’ average age was 65 years, and 51% were female.
Post treatment, mean improvement in HAM-D score from baseline was 6.53 points in the telephone-based CBT group, compared with −0.27 points in the control group. “Effects at the end of treatment were maintained at 6-month follow-up,” the researchers reported.
About 40% of patients in the CBT group reported that their depression was much improved or very much improved, compared with none of the patients in the control group. Responders had mild to minimal symptomatology on the HAM-D, which indicates that the changes were clinically significant, the authors said.
Secondary outcomes also favored telephone-based CBT. “The intervention was feasible and highly acceptable, yielding an 88% retention rate over the 9-month trial,” Dr. Dobkin and colleagues said.
Compared with other control conditions, treatment-as-usual controls may enhance the effect size of an intervention, the authors noted. In addition, factors such as therapeutic relationship, time, and attention likely contribute to psychotherapy outcomes.
Success may hinge on cognitive ability
“The success of this trial highlights the need for further efficacy studies targeting neuropsychiatric manifestations of [Parkinson’s disease] and adds urgency to the discussion over policies regarding access to tele–mental health, especially for vulnerable populations with limited access to in-person mental health services,” Gregory M. Pontone, MD, and Kelly A. Mills, MD, wrote in an accompanying editorial. Dr. Pontone and Dr. Mills are affiliated with Johns Hopkins University in Baltimore.
“Only rudimentary evidence” exists to guide the treatment of depression in patients with Parkinson’s disease, the editorialists said. “Patient preference and tolerability suggest that nonpharmacologic therapies, such as CBT, are preferred as first-line treatment. Yet access to qualified CBT practitioners, especially those with a clinical knowledge of [Parkinson’s disease], is limited.”
Despite its advantages and the encouraging results, CBT may have important limitations as well, they said. Patients require a certain degree of cognitive ability to benefit from CBT, and the prevalence of dementia among patients with Parkinson’s disease is about 30%.
Nevertheless, the trial provided evidence of target engagement. “Though caveats include the single-blind design and potential confounding by time spent with patient and caregiver, the authors demonstrated that improvement was mediated by the mechanism of CBT – a reduction in negative thinking.”
The trial was funded by the Michael J. Fox Foundation for Parkinson’s Research and the Parkinson’s Alliance (Parkinson’s Unity Walk). Dr. Mills disclosed a patent pending for a system for phase-dependent cortical brain stimulation, National Institutes of Health funding, pending funding from the Michael J. Fox Foundation, and commercial research support from Global Kinetics Corporation. Dr. Pontone is a consultant for Acadia Pharmaceuticals.
SOURCE: Dobkin RD et al. Neurology. 2020 Apr 1. doi: 10.1212/WNL.0000000000009292.
FROM NEUROLOGY

