User login
Improved Transitional Care Through an Innovative Hospitalist Model: Expanding Clinician Practice From Acute to Subacute Care
Hospitalist physician rotations between acute inpatient hospitals and subacute care facilities with dedicated time in each environment may foster quality improvement and educational opportunities.
Care transitions between hospitals and skilled nursing facilities (SNFs) are a vulnerable time for patients. The current health care climate of decreasing hospital length of stay, readmission penalties, and increasing patient complexity has made hospital care transitions an important safety concern. Suboptimal transitions across clinical settings can result in adverse events, inadequately controlled comorbidities, deficient patient and caregiver preparation for discharge, medication errors, relocation stress, and overall increased morbidity and mortality.1,2 Such care transitions also may generate unnecessary spending, including avoidable readmissions, emergency department utilization, and duplicative laboratory and imaging studies. Approximately 23% of patients admitted to SNFs are readmitted to acute care hospitals within 30 days, and these patients have increased mortality rates in risk-adjusted analyses. 3,4
Compounding the magnitude of this risk and vulnerability is the significant growth in the number of patients discharged to SNFs over the past 30 years. In 2013, more than 20% of Medicare patients discharged from acute care hospitals were destined for SNFs.5,6 Paradoxically, despite the increasing need for SNF providers, there is a shortage of clinicians with training in geriatrics or nursing home care.7 The result is a growing need to identify organizational systems to optimize physician practice in these settings, enhance quality of care, especially around transitions, and increase educational training opportunities in SNFs for future practitioners.
Many SNFs today are staffed by physicians and other licensed clinicians whose exclusive practice location is the nursing facility or possibly several such facilities. This prevailing model of care can isolate the physicians, depriving them of interaction with clinicians in other specialties, and can contribute to burnout.8 This model does not lend itself to academic scholarship, quality improvement (QI), and student or resident training, as each of these endeavors depends on interprofessional collaboration as well as access to an academic medical center with additional resources.9
Few studies have described innovative hospitalist rotation models from acute to subacute care. The Cleveland Clinic implemented the Connected Care model where hospital-employed physicians and advanced practice professionals integrated into postacute care and reduced the 30-day hospital readmission rate from SNFs from 28% to 22%.10 Goth and colleagues performed a comparative effectiveness trial between a postacute care hospitalist (PACH) model and a community-based physician model of nursing home care. They found that the institution of a PACH model in a nursing home was associated with a significant increase in laboratory costs, nonsignificant reduction in medication errors and pharmacy costs, and no improvement in fall rates.11 The conclusion was that the PACH model may lead to greater clinician involvement and that the potential decrease in pharmacy costs and medications errors may offset the costs associated with additional laboratory testing. Overall, there has been a lack of studies on the impact of these hospitalist rotation models from acute to subacute care on educational programs, QI activities, and the interprofessional environment.
To achieve a system in which physicians in a SNF can excel in these areas, Veterans Affairs Boston Healthcare System (VABHS) adopted a staffing model in which academic hospitalist physicians rotate between the inpatient hospital and subacute settings. This report describes the model structure, the varying roles of the physicians, and early indicators of its positive effects on educational programs, QI activities, and the interprofessional environment.
Methods
The VABHS consists of a 159-bed acute care hospital in West Roxbury, Massachusetts; and a 110-bed SNF in Brockton, Massachusetts, with 3 units: a 65-bed transitional care unit (TCU), a 30-bed long-term care unit, and a 15-bed palliative care/hospice unit. The majority of patients admitted to the SNF are transferred from the acute care hospital in West Roxbury and other regional hospitals. Prior to 2015, the TCU was staffed with full-time clinicians who exclusively practiced in the SNF.
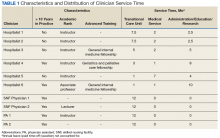
In the new staffing model, 6 hospitalist physicians divide their clinical time between the acute care hospital’s inpatient medical service and the TCU. The hospitalists come from varied backgrounds in terms of years in practice and advanced training (Table 1).
The amount of nonclinical (protected) time and clinical time on the acute inpatient service and the TCU varies for each physician. For example, a physician serves as principal investigator for several major research grants and has a hospital-wide administrative leadership role; as a result, the principal investigator has fewer months of clinical responsibility. Physicians are expected to use the protected time for scholarship, educational program development and teaching, QI, and administrative responsibilities. The VABHS leadership determines the amount of protected time based on individualized benchmarks for research, education, and administrative responsibilities that follow VA national and local institutional guidelines. These metrics and time allocations are negotiated at the time of recruitment and then are reviewed annually.
The TCU also is staffed with 4 full-time clinicians (2 physicians and 2 physician assistants) who provide additional continuity of care. The new hospitalist staffing model only required an approximate 10% increase in TCU clinical staffing full-time equivalents. Patients and admissions are divided equally among clinicians on service (census per clinician 12-15 patients), with redistribution of patients at times of transition from clinical to nonclinical time. Blocks of clinical time are scheduled for greater than 2 weeks at a time to preserve continuity. In addition, the new staffing model allocates assignment of clinical responsibilities that allows for clinicians to take leave without resultant shortages in clinical coverage.
To facilitate communication among physicians serving in the acute inpatient facility and the TCU, leaders of both of these programs meet monthly and ad hoc to review the transitions of care between the 2 settings. The description of this model and its assessment have been reviewed and deemed exempt from oversight by the VA Boston Healthcare System Research and Development Committee.
Results
Since the implementation of this staffing model in 2015, the system has grown considerably in the breadth and depth of educational programming, QI, and systems redesign in the TCU and, more broadly, in the SNF. The TCU, which previously had limited training opportunities, has experienced marked expansion of educational offerings. It is now a site for core general medicine rotations for first-year psychiatry residents and physician assistant students. The TCU also has expanded as a clinical site for transitions-in-care internal medicine resident curricula and electives, as well as a clinical site for a geriatrics fellowship.
A hospitalist developed and implemented a 4-week interprofessional curriculum for all clinical trainees and students, which occurs continuously. The curriculum includes a monthly academic conference and 12 didactic lectures and is taught by 16 interprofessional faculty from the TCU and the Palliative Care/Hospice Unit, including medicine, geriatric and palliative care physicians, physician assistants, social workers, physical and occupational therapists, pharmacists, and a geriatric psychologist. The goal of the curriculum is to provide learners the knowledge, attitudes, and skills necessary to perform effective, efficient, and safe transfers between clinical settings as well as education in transitional care. In addition, using a team of interprofessional faculty, the curriculum develops the interprofessional competencies of teamwork and communication. The curriculum also has provided a significant opportunity for interprofessional collaboration among faculty who have volunteered their teaching time in the development and teaching of the curriculum, with potential for improved clinical staff knowledge of other disciplines.
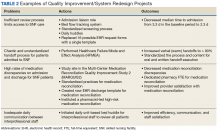
Quality improvement and system redesign projects in care transitions also have expanded (Table 2).
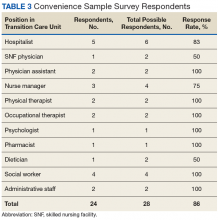
Early assessment indicates that the new staffing model is having positive effects on the clinical environment of the TCU. A survey was conducted of a convenience sample of all physicians, nurse managers, social workers, and other members of the clinical team in the TCU (N=24)(Table 3), with response categories ranging on a Likert scale from 1 (very negative) to 5 (very positive).
Although not rigorously analyzed using qualitative research methods, comments from respondents have consistently indicated that this staffing model increases the transfer of clinical and logistical knowledge among staff members working in the acute inpatient facility and the TCU.
Discussion
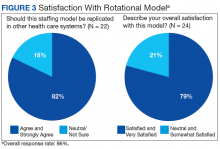
With greater numbers of increasingly complex patients transitioning from the hospital to SNF, health care systems need to expand the capacity of their skilled nursing systems, not only to provide clinical care, but also to support QI and medical education. The VABHS developed a physician staffing model with the goal of enriching physician practice and enhancing QI and educational opportunities in its SNF. The model offers an opportunity to improve transitions in care as physicians gain a greater knowledge of both the hospital and subacute clinical settings. This hospitalist rotation model may improve the knowledge necessary for caring for patients moving across care settings, as well as improve communication between settings. It also has served as a foundation for systematic innovation in QI and education at this institution. Clinical staff in the transitional care setting have reported positive effects of this model on clinical skills and patient care, educational opportunities, as well as a desire for replication in other health care systems.
The potential generalizability of this model requires careful consideration. The VABHS is a tertiary care integrated health care system, enabling physicians to work in multiple clinical settings. Other settings may not have the staffing or clinical volume to sustain such a model. In addition, this model may increase discontinuity in patient care as hospitalists move between acute and subacute settings and nonclinical roles. This loss of continuity may be a greater concern in the SNF setting, as the inpatient hospitalist model generally involves high provider turnover as shift work. Our survey included nurse managers, and not floor nurses due to survey administration limitations, and feedback may not have captured a comprehensive view from CLC staff. Moreover, some of the perceived positive impacts also may be related to professional and personal attributes of the physicians rather than the actual model of care. In addition, the survey response rate was 86%. However, the nature of the improvement work (focused on care transitions) and educational opportunities (interprofessional care) would likely not occur had the physicians been based in one clinical setting.
Other new physician staffing models have been designed to improve the continuity between the hospital, subacute, and outpatient settings. For example, the University of Chicago Comprehensive Care model pairs patients with trained hospitalists who provide both inpatient and outpatient care, thereby optimizing continuity between these settings.14 At CareMore Health System, high-risk patients also are paired with hospitalists, referred to as “extensivists,” who lead care teams that follow patients between settings and provide acute, postacute, and outpatient care.15 In these models, a single physician takes responsibility for the patient throughout transitions of care and through various care settings. Both models have shown reduction in hospital readmissions. One concern with such models is that the treatment teams need to coexist in the various settings of care, and the ability to impact and create systematic change within each environment is limited. This may limit QI, educational opportunities, and system level impact within each environment of care.
In comparison, the “transitionalist” model proposed here features hospitalist physicians rotating between the acute inpatient hospital and subacute care with dedicated time in each environment. This innovative organizational structure may enhance physician practice and enrich QI and educational opportunities in SNFs. Further evaluation will include the impact on quality metrics of patient care and patient satisfaction, as this model has the potential to influence quality, cost, and overall health outcomes.
Acknowledgments
We would like to thank Shivani Jindal, Matthew Russell, Matthew Ronan, Juman Hijab, Wei Shen, Sandra Vilbrun-Bruno, and Jack Earnshaw for their significant contributions to this staffing model. We would also like to thank Paul Conlin, Jay Orlander, and the leadership team of Veterans Affairs Boston Healthcare System for supporting this staffing model.
1. Forster AJ, Murff HJ, Peterson JF, Gandhi TK, Bates DW. Adverse drug events occurring following hospital discharge. J Gen Intern Med. 2005;20(4):317-323.
2. Murtaugh CM, Litke A. Transitions through postacute and long-term care settings: patterns of use and outcomes for a national cohort of elders. Med Care. 2002;40(3):227-236.
3. Burke RE, Whitfield EA, Hittle D, et al. Hospital readmission from post-acute care facilities: risk factors, timing, and outcomes. J Am Med Dir Assoc. 2016;17(3):249-255.
4. Mor V, Intrator O, Feng Z, Grabowski DC. The revolving door of rehospitalization from skilled nursing facilities. Health Aff (Millwood). 2010;29(1):57-64.
5. Tian W. An all-payer view of hospital discharge to postacute care, 2013: Statistical Brief #205. https://www.hcup-us.ahrq.gov/reports/statbriefs/sb205-Hospital-Discharge-Postacute-Care.jsp. Published May 2016. Accessed August 13, 2018.
6. Barnett ML, Grabowski DC, Mehrotra A. Home-to-home time–measuring what matters to patients and payers. N Engl J Med. 2017;377(1):4-6.
7. Golden AG, Silverman MA, Mintzer MJ. Is geriatric medicine terminally ill? Ann Intern Med. 2012;156(9):654-656.
8. Nazir A, Smalbrugge M, Moser A, et al. The prevalence of burnout among nursing home physicians: an international perspective. J Am Med Dir Assoc. 2018;19(1):86-88.
9. Coleman EA, Berenson RA. Lost in transition: challenges and opportunities for improving the quality of transitional care. Ann Intern Med. 2004;141(7):533-536.
10. Kim LD, Kou L, Hu B, Gorodeski EZ, Rothberg MB. Impact of a connected care model on 30-day readmission rates from skilled nursing facilities. J Hosp Med. 2017;12(4):238-244.
11. Gloth MF, Gloth MJ. A comparative effectiveness trial between a post-acute care hospitalist model and a community-based physician model of nursing home care. J Am Med Dir Assoc. 2011;12(5):384-386.
12. Baughman AW, Cain G, Ruopp MD, et al. Improving access to care by admission process redesign in a veterans affairs skilled nursing facility. Jt Comm J Qual Patient Saf. 2018;44(8):454-462.
13. Mixon A, Smith GR, Dalal A et al. The Multi-Center Medication Reconciliation Quality Improvement Study 2 (MARQUIS2): methods and implementation. Abstract 248. Present at: Society of Hospital Medicine Annual Meeting; 2018 Apr 8 – 11, 2018; Orlando, FL. https://www.shmabstracts.com/abstract/the-multi-center-medication-reconciliation-quality-improvement-study-2-marquis2-methods-and-implementation. Accessed August 13, 2018.
14. Meltzer DO, Ruhnke GW. Redesigning care for patients at increased hospitalization risk: the comprehensive care physician model. Health Aff (Millwood). 2014;33(5):770-777.
15. Powers BW, Milstein A, Jain SH. Delivery models for high-risk older patients: back to the future? JAMA. 2016;315(1):23-24.
Hospitalist physician rotations between acute inpatient hospitals and subacute care facilities with dedicated time in each environment may foster quality improvement and educational opportunities.
Hospitalist physician rotations between acute inpatient hospitals and subacute care facilities with dedicated time in each environment may foster quality improvement and educational opportunities.
Care transitions between hospitals and skilled nursing facilities (SNFs) are a vulnerable time for patients. The current health care climate of decreasing hospital length of stay, readmission penalties, and increasing patient complexity has made hospital care transitions an important safety concern. Suboptimal transitions across clinical settings can result in adverse events, inadequately controlled comorbidities, deficient patient and caregiver preparation for discharge, medication errors, relocation stress, and overall increased morbidity and mortality.1,2 Such care transitions also may generate unnecessary spending, including avoidable readmissions, emergency department utilization, and duplicative laboratory and imaging studies. Approximately 23% of patients admitted to SNFs are readmitted to acute care hospitals within 30 days, and these patients have increased mortality rates in risk-adjusted analyses. 3,4
Compounding the magnitude of this risk and vulnerability is the significant growth in the number of patients discharged to SNFs over the past 30 years. In 2013, more than 20% of Medicare patients discharged from acute care hospitals were destined for SNFs.5,6 Paradoxically, despite the increasing need for SNF providers, there is a shortage of clinicians with training in geriatrics or nursing home care.7 The result is a growing need to identify organizational systems to optimize physician practice in these settings, enhance quality of care, especially around transitions, and increase educational training opportunities in SNFs for future practitioners.
Many SNFs today are staffed by physicians and other licensed clinicians whose exclusive practice location is the nursing facility or possibly several such facilities. This prevailing model of care can isolate the physicians, depriving them of interaction with clinicians in other specialties, and can contribute to burnout.8 This model does not lend itself to academic scholarship, quality improvement (QI), and student or resident training, as each of these endeavors depends on interprofessional collaboration as well as access to an academic medical center with additional resources.9
Few studies have described innovative hospitalist rotation models from acute to subacute care. The Cleveland Clinic implemented the Connected Care model where hospital-employed physicians and advanced practice professionals integrated into postacute care and reduced the 30-day hospital readmission rate from SNFs from 28% to 22%.10 Goth and colleagues performed a comparative effectiveness trial between a postacute care hospitalist (PACH) model and a community-based physician model of nursing home care. They found that the institution of a PACH model in a nursing home was associated with a significant increase in laboratory costs, nonsignificant reduction in medication errors and pharmacy costs, and no improvement in fall rates.11 The conclusion was that the PACH model may lead to greater clinician involvement and that the potential decrease in pharmacy costs and medications errors may offset the costs associated with additional laboratory testing. Overall, there has been a lack of studies on the impact of these hospitalist rotation models from acute to subacute care on educational programs, QI activities, and the interprofessional environment.
To achieve a system in which physicians in a SNF can excel in these areas, Veterans Affairs Boston Healthcare System (VABHS) adopted a staffing model in which academic hospitalist physicians rotate between the inpatient hospital and subacute settings. This report describes the model structure, the varying roles of the physicians, and early indicators of its positive effects on educational programs, QI activities, and the interprofessional environment.
Methods
The VABHS consists of a 159-bed acute care hospital in West Roxbury, Massachusetts; and a 110-bed SNF in Brockton, Massachusetts, with 3 units: a 65-bed transitional care unit (TCU), a 30-bed long-term care unit, and a 15-bed palliative care/hospice unit. The majority of patients admitted to the SNF are transferred from the acute care hospital in West Roxbury and other regional hospitals. Prior to 2015, the TCU was staffed with full-time clinicians who exclusively practiced in the SNF.
In the new staffing model, 6 hospitalist physicians divide their clinical time between the acute care hospital’s inpatient medical service and the TCU. The hospitalists come from varied backgrounds in terms of years in practice and advanced training (Table 1).
The amount of nonclinical (protected) time and clinical time on the acute inpatient service and the TCU varies for each physician. For example, a physician serves as principal investigator for several major research grants and has a hospital-wide administrative leadership role; as a result, the principal investigator has fewer months of clinical responsibility. Physicians are expected to use the protected time for scholarship, educational program development and teaching, QI, and administrative responsibilities. The VABHS leadership determines the amount of protected time based on individualized benchmarks for research, education, and administrative responsibilities that follow VA national and local institutional guidelines. These metrics and time allocations are negotiated at the time of recruitment and then are reviewed annually.
The TCU also is staffed with 4 full-time clinicians (2 physicians and 2 physician assistants) who provide additional continuity of care. The new hospitalist staffing model only required an approximate 10% increase in TCU clinical staffing full-time equivalents. Patients and admissions are divided equally among clinicians on service (census per clinician 12-15 patients), with redistribution of patients at times of transition from clinical to nonclinical time. Blocks of clinical time are scheduled for greater than 2 weeks at a time to preserve continuity. In addition, the new staffing model allocates assignment of clinical responsibilities that allows for clinicians to take leave without resultant shortages in clinical coverage.
To facilitate communication among physicians serving in the acute inpatient facility and the TCU, leaders of both of these programs meet monthly and ad hoc to review the transitions of care between the 2 settings. The description of this model and its assessment have been reviewed and deemed exempt from oversight by the VA Boston Healthcare System Research and Development Committee.
Results
Since the implementation of this staffing model in 2015, the system has grown considerably in the breadth and depth of educational programming, QI, and systems redesign in the TCU and, more broadly, in the SNF. The TCU, which previously had limited training opportunities, has experienced marked expansion of educational offerings. It is now a site for core general medicine rotations for first-year psychiatry residents and physician assistant students. The TCU also has expanded as a clinical site for transitions-in-care internal medicine resident curricula and electives, as well as a clinical site for a geriatrics fellowship.
A hospitalist developed and implemented a 4-week interprofessional curriculum for all clinical trainees and students, which occurs continuously. The curriculum includes a monthly academic conference and 12 didactic lectures and is taught by 16 interprofessional faculty from the TCU and the Palliative Care/Hospice Unit, including medicine, geriatric and palliative care physicians, physician assistants, social workers, physical and occupational therapists, pharmacists, and a geriatric psychologist. The goal of the curriculum is to provide learners the knowledge, attitudes, and skills necessary to perform effective, efficient, and safe transfers between clinical settings as well as education in transitional care. In addition, using a team of interprofessional faculty, the curriculum develops the interprofessional competencies of teamwork and communication. The curriculum also has provided a significant opportunity for interprofessional collaboration among faculty who have volunteered their teaching time in the development and teaching of the curriculum, with potential for improved clinical staff knowledge of other disciplines.
Quality improvement and system redesign projects in care transitions also have expanded (Table 2).
Early assessment indicates that the new staffing model is having positive effects on the clinical environment of the TCU. A survey was conducted of a convenience sample of all physicians, nurse managers, social workers, and other members of the clinical team in the TCU (N=24)(Table 3), with response categories ranging on a Likert scale from 1 (very negative) to 5 (very positive).
Although not rigorously analyzed using qualitative research methods, comments from respondents have consistently indicated that this staffing model increases the transfer of clinical and logistical knowledge among staff members working in the acute inpatient facility and the TCU.
Discussion
With greater numbers of increasingly complex patients transitioning from the hospital to SNF, health care systems need to expand the capacity of their skilled nursing systems, not only to provide clinical care, but also to support QI and medical education. The VABHS developed a physician staffing model with the goal of enriching physician practice and enhancing QI and educational opportunities in its SNF. The model offers an opportunity to improve transitions in care as physicians gain a greater knowledge of both the hospital and subacute clinical settings. This hospitalist rotation model may improve the knowledge necessary for caring for patients moving across care settings, as well as improve communication between settings. It also has served as a foundation for systematic innovation in QI and education at this institution. Clinical staff in the transitional care setting have reported positive effects of this model on clinical skills and patient care, educational opportunities, as well as a desire for replication in other health care systems.
The potential generalizability of this model requires careful consideration. The VABHS is a tertiary care integrated health care system, enabling physicians to work in multiple clinical settings. Other settings may not have the staffing or clinical volume to sustain such a model. In addition, this model may increase discontinuity in patient care as hospitalists move between acute and subacute settings and nonclinical roles. This loss of continuity may be a greater concern in the SNF setting, as the inpatient hospitalist model generally involves high provider turnover as shift work. Our survey included nurse managers, and not floor nurses due to survey administration limitations, and feedback may not have captured a comprehensive view from CLC staff. Moreover, some of the perceived positive impacts also may be related to professional and personal attributes of the physicians rather than the actual model of care. In addition, the survey response rate was 86%. However, the nature of the improvement work (focused on care transitions) and educational opportunities (interprofessional care) would likely not occur had the physicians been based in one clinical setting.
Other new physician staffing models have been designed to improve the continuity between the hospital, subacute, and outpatient settings. For example, the University of Chicago Comprehensive Care model pairs patients with trained hospitalists who provide both inpatient and outpatient care, thereby optimizing continuity between these settings.14 At CareMore Health System, high-risk patients also are paired with hospitalists, referred to as “extensivists,” who lead care teams that follow patients between settings and provide acute, postacute, and outpatient care.15 In these models, a single physician takes responsibility for the patient throughout transitions of care and through various care settings. Both models have shown reduction in hospital readmissions. One concern with such models is that the treatment teams need to coexist in the various settings of care, and the ability to impact and create systematic change within each environment is limited. This may limit QI, educational opportunities, and system level impact within each environment of care.
In comparison, the “transitionalist” model proposed here features hospitalist physicians rotating between the acute inpatient hospital and subacute care with dedicated time in each environment. This innovative organizational structure may enhance physician practice and enrich QI and educational opportunities in SNFs. Further evaluation will include the impact on quality metrics of patient care and patient satisfaction, as this model has the potential to influence quality, cost, and overall health outcomes.
Acknowledgments
We would like to thank Shivani Jindal, Matthew Russell, Matthew Ronan, Juman Hijab, Wei Shen, Sandra Vilbrun-Bruno, and Jack Earnshaw for their significant contributions to this staffing model. We would also like to thank Paul Conlin, Jay Orlander, and the leadership team of Veterans Affairs Boston Healthcare System for supporting this staffing model.
Care transitions between hospitals and skilled nursing facilities (SNFs) are a vulnerable time for patients. The current health care climate of decreasing hospital length of stay, readmission penalties, and increasing patient complexity has made hospital care transitions an important safety concern. Suboptimal transitions across clinical settings can result in adverse events, inadequately controlled comorbidities, deficient patient and caregiver preparation for discharge, medication errors, relocation stress, and overall increased morbidity and mortality.1,2 Such care transitions also may generate unnecessary spending, including avoidable readmissions, emergency department utilization, and duplicative laboratory and imaging studies. Approximately 23% of patients admitted to SNFs are readmitted to acute care hospitals within 30 days, and these patients have increased mortality rates in risk-adjusted analyses. 3,4
Compounding the magnitude of this risk and vulnerability is the significant growth in the number of patients discharged to SNFs over the past 30 years. In 2013, more than 20% of Medicare patients discharged from acute care hospitals were destined for SNFs.5,6 Paradoxically, despite the increasing need for SNF providers, there is a shortage of clinicians with training in geriatrics or nursing home care.7 The result is a growing need to identify organizational systems to optimize physician practice in these settings, enhance quality of care, especially around transitions, and increase educational training opportunities in SNFs for future practitioners.
Many SNFs today are staffed by physicians and other licensed clinicians whose exclusive practice location is the nursing facility or possibly several such facilities. This prevailing model of care can isolate the physicians, depriving them of interaction with clinicians in other specialties, and can contribute to burnout.8 This model does not lend itself to academic scholarship, quality improvement (QI), and student or resident training, as each of these endeavors depends on interprofessional collaboration as well as access to an academic medical center with additional resources.9
Few studies have described innovative hospitalist rotation models from acute to subacute care. The Cleveland Clinic implemented the Connected Care model where hospital-employed physicians and advanced practice professionals integrated into postacute care and reduced the 30-day hospital readmission rate from SNFs from 28% to 22%.10 Goth and colleagues performed a comparative effectiveness trial between a postacute care hospitalist (PACH) model and a community-based physician model of nursing home care. They found that the institution of a PACH model in a nursing home was associated with a significant increase in laboratory costs, nonsignificant reduction in medication errors and pharmacy costs, and no improvement in fall rates.11 The conclusion was that the PACH model may lead to greater clinician involvement and that the potential decrease in pharmacy costs and medications errors may offset the costs associated with additional laboratory testing. Overall, there has been a lack of studies on the impact of these hospitalist rotation models from acute to subacute care on educational programs, QI activities, and the interprofessional environment.
To achieve a system in which physicians in a SNF can excel in these areas, Veterans Affairs Boston Healthcare System (VABHS) adopted a staffing model in which academic hospitalist physicians rotate between the inpatient hospital and subacute settings. This report describes the model structure, the varying roles of the physicians, and early indicators of its positive effects on educational programs, QI activities, and the interprofessional environment.
Methods
The VABHS consists of a 159-bed acute care hospital in West Roxbury, Massachusetts; and a 110-bed SNF in Brockton, Massachusetts, with 3 units: a 65-bed transitional care unit (TCU), a 30-bed long-term care unit, and a 15-bed palliative care/hospice unit. The majority of patients admitted to the SNF are transferred from the acute care hospital in West Roxbury and other regional hospitals. Prior to 2015, the TCU was staffed with full-time clinicians who exclusively practiced in the SNF.
In the new staffing model, 6 hospitalist physicians divide their clinical time between the acute care hospital’s inpatient medical service and the TCU. The hospitalists come from varied backgrounds in terms of years in practice and advanced training (Table 1).
The amount of nonclinical (protected) time and clinical time on the acute inpatient service and the TCU varies for each physician. For example, a physician serves as principal investigator for several major research grants and has a hospital-wide administrative leadership role; as a result, the principal investigator has fewer months of clinical responsibility. Physicians are expected to use the protected time for scholarship, educational program development and teaching, QI, and administrative responsibilities. The VABHS leadership determines the amount of protected time based on individualized benchmarks for research, education, and administrative responsibilities that follow VA national and local institutional guidelines. These metrics and time allocations are negotiated at the time of recruitment and then are reviewed annually.
The TCU also is staffed with 4 full-time clinicians (2 physicians and 2 physician assistants) who provide additional continuity of care. The new hospitalist staffing model only required an approximate 10% increase in TCU clinical staffing full-time equivalents. Patients and admissions are divided equally among clinicians on service (census per clinician 12-15 patients), with redistribution of patients at times of transition from clinical to nonclinical time. Blocks of clinical time are scheduled for greater than 2 weeks at a time to preserve continuity. In addition, the new staffing model allocates assignment of clinical responsibilities that allows for clinicians to take leave without resultant shortages in clinical coverage.
To facilitate communication among physicians serving in the acute inpatient facility and the TCU, leaders of both of these programs meet monthly and ad hoc to review the transitions of care between the 2 settings. The description of this model and its assessment have been reviewed and deemed exempt from oversight by the VA Boston Healthcare System Research and Development Committee.
Results
Since the implementation of this staffing model in 2015, the system has grown considerably in the breadth and depth of educational programming, QI, and systems redesign in the TCU and, more broadly, in the SNF. The TCU, which previously had limited training opportunities, has experienced marked expansion of educational offerings. It is now a site for core general medicine rotations for first-year psychiatry residents and physician assistant students. The TCU also has expanded as a clinical site for transitions-in-care internal medicine resident curricula and electives, as well as a clinical site for a geriatrics fellowship.
A hospitalist developed and implemented a 4-week interprofessional curriculum for all clinical trainees and students, which occurs continuously. The curriculum includes a monthly academic conference and 12 didactic lectures and is taught by 16 interprofessional faculty from the TCU and the Palliative Care/Hospice Unit, including medicine, geriatric and palliative care physicians, physician assistants, social workers, physical and occupational therapists, pharmacists, and a geriatric psychologist. The goal of the curriculum is to provide learners the knowledge, attitudes, and skills necessary to perform effective, efficient, and safe transfers between clinical settings as well as education in transitional care. In addition, using a team of interprofessional faculty, the curriculum develops the interprofessional competencies of teamwork and communication. The curriculum also has provided a significant opportunity for interprofessional collaboration among faculty who have volunteered their teaching time in the development and teaching of the curriculum, with potential for improved clinical staff knowledge of other disciplines.
Quality improvement and system redesign projects in care transitions also have expanded (Table 2).
Early assessment indicates that the new staffing model is having positive effects on the clinical environment of the TCU. A survey was conducted of a convenience sample of all physicians, nurse managers, social workers, and other members of the clinical team in the TCU (N=24)(Table 3), with response categories ranging on a Likert scale from 1 (very negative) to 5 (very positive).
Although not rigorously analyzed using qualitative research methods, comments from respondents have consistently indicated that this staffing model increases the transfer of clinical and logistical knowledge among staff members working in the acute inpatient facility and the TCU.
Discussion
With greater numbers of increasingly complex patients transitioning from the hospital to SNF, health care systems need to expand the capacity of their skilled nursing systems, not only to provide clinical care, but also to support QI and medical education. The VABHS developed a physician staffing model with the goal of enriching physician practice and enhancing QI and educational opportunities in its SNF. The model offers an opportunity to improve transitions in care as physicians gain a greater knowledge of both the hospital and subacute clinical settings. This hospitalist rotation model may improve the knowledge necessary for caring for patients moving across care settings, as well as improve communication between settings. It also has served as a foundation for systematic innovation in QI and education at this institution. Clinical staff in the transitional care setting have reported positive effects of this model on clinical skills and patient care, educational opportunities, as well as a desire for replication in other health care systems.
The potential generalizability of this model requires careful consideration. The VABHS is a tertiary care integrated health care system, enabling physicians to work in multiple clinical settings. Other settings may not have the staffing or clinical volume to sustain such a model. In addition, this model may increase discontinuity in patient care as hospitalists move between acute and subacute settings and nonclinical roles. This loss of continuity may be a greater concern in the SNF setting, as the inpatient hospitalist model generally involves high provider turnover as shift work. Our survey included nurse managers, and not floor nurses due to survey administration limitations, and feedback may not have captured a comprehensive view from CLC staff. Moreover, some of the perceived positive impacts also may be related to professional and personal attributes of the physicians rather than the actual model of care. In addition, the survey response rate was 86%. However, the nature of the improvement work (focused on care transitions) and educational opportunities (interprofessional care) would likely not occur had the physicians been based in one clinical setting.
Other new physician staffing models have been designed to improve the continuity between the hospital, subacute, and outpatient settings. For example, the University of Chicago Comprehensive Care model pairs patients with trained hospitalists who provide both inpatient and outpatient care, thereby optimizing continuity between these settings.14 At CareMore Health System, high-risk patients also are paired with hospitalists, referred to as “extensivists,” who lead care teams that follow patients between settings and provide acute, postacute, and outpatient care.15 In these models, a single physician takes responsibility for the patient throughout transitions of care and through various care settings. Both models have shown reduction in hospital readmissions. One concern with such models is that the treatment teams need to coexist in the various settings of care, and the ability to impact and create systematic change within each environment is limited. This may limit QI, educational opportunities, and system level impact within each environment of care.
In comparison, the “transitionalist” model proposed here features hospitalist physicians rotating between the acute inpatient hospital and subacute care with dedicated time in each environment. This innovative organizational structure may enhance physician practice and enrich QI and educational opportunities in SNFs. Further evaluation will include the impact on quality metrics of patient care and patient satisfaction, as this model has the potential to influence quality, cost, and overall health outcomes.
Acknowledgments
We would like to thank Shivani Jindal, Matthew Russell, Matthew Ronan, Juman Hijab, Wei Shen, Sandra Vilbrun-Bruno, and Jack Earnshaw for their significant contributions to this staffing model. We would also like to thank Paul Conlin, Jay Orlander, and the leadership team of Veterans Affairs Boston Healthcare System for supporting this staffing model.
1. Forster AJ, Murff HJ, Peterson JF, Gandhi TK, Bates DW. Adverse drug events occurring following hospital discharge. J Gen Intern Med. 2005;20(4):317-323.
2. Murtaugh CM, Litke A. Transitions through postacute and long-term care settings: patterns of use and outcomes for a national cohort of elders. Med Care. 2002;40(3):227-236.
3. Burke RE, Whitfield EA, Hittle D, et al. Hospital readmission from post-acute care facilities: risk factors, timing, and outcomes. J Am Med Dir Assoc. 2016;17(3):249-255.
4. Mor V, Intrator O, Feng Z, Grabowski DC. The revolving door of rehospitalization from skilled nursing facilities. Health Aff (Millwood). 2010;29(1):57-64.
5. Tian W. An all-payer view of hospital discharge to postacute care, 2013: Statistical Brief #205. https://www.hcup-us.ahrq.gov/reports/statbriefs/sb205-Hospital-Discharge-Postacute-Care.jsp. Published May 2016. Accessed August 13, 2018.
6. Barnett ML, Grabowski DC, Mehrotra A. Home-to-home time–measuring what matters to patients and payers. N Engl J Med. 2017;377(1):4-6.
7. Golden AG, Silverman MA, Mintzer MJ. Is geriatric medicine terminally ill? Ann Intern Med. 2012;156(9):654-656.
8. Nazir A, Smalbrugge M, Moser A, et al. The prevalence of burnout among nursing home physicians: an international perspective. J Am Med Dir Assoc. 2018;19(1):86-88.
9. Coleman EA, Berenson RA. Lost in transition: challenges and opportunities for improving the quality of transitional care. Ann Intern Med. 2004;141(7):533-536.
10. Kim LD, Kou L, Hu B, Gorodeski EZ, Rothberg MB. Impact of a connected care model on 30-day readmission rates from skilled nursing facilities. J Hosp Med. 2017;12(4):238-244.
11. Gloth MF, Gloth MJ. A comparative effectiveness trial between a post-acute care hospitalist model and a community-based physician model of nursing home care. J Am Med Dir Assoc. 2011;12(5):384-386.
12. Baughman AW, Cain G, Ruopp MD, et al. Improving access to care by admission process redesign in a veterans affairs skilled nursing facility. Jt Comm J Qual Patient Saf. 2018;44(8):454-462.
13. Mixon A, Smith GR, Dalal A et al. The Multi-Center Medication Reconciliation Quality Improvement Study 2 (MARQUIS2): methods and implementation. Abstract 248. Present at: Society of Hospital Medicine Annual Meeting; 2018 Apr 8 – 11, 2018; Orlando, FL. https://www.shmabstracts.com/abstract/the-multi-center-medication-reconciliation-quality-improvement-study-2-marquis2-methods-and-implementation. Accessed August 13, 2018.
14. Meltzer DO, Ruhnke GW. Redesigning care for patients at increased hospitalization risk: the comprehensive care physician model. Health Aff (Millwood). 2014;33(5):770-777.
15. Powers BW, Milstein A, Jain SH. Delivery models for high-risk older patients: back to the future? JAMA. 2016;315(1):23-24.
1. Forster AJ, Murff HJ, Peterson JF, Gandhi TK, Bates DW. Adverse drug events occurring following hospital discharge. J Gen Intern Med. 2005;20(4):317-323.
2. Murtaugh CM, Litke A. Transitions through postacute and long-term care settings: patterns of use and outcomes for a national cohort of elders. Med Care. 2002;40(3):227-236.
3. Burke RE, Whitfield EA, Hittle D, et al. Hospital readmission from post-acute care facilities: risk factors, timing, and outcomes. J Am Med Dir Assoc. 2016;17(3):249-255.
4. Mor V, Intrator O, Feng Z, Grabowski DC. The revolving door of rehospitalization from skilled nursing facilities. Health Aff (Millwood). 2010;29(1):57-64.
5. Tian W. An all-payer view of hospital discharge to postacute care, 2013: Statistical Brief #205. https://www.hcup-us.ahrq.gov/reports/statbriefs/sb205-Hospital-Discharge-Postacute-Care.jsp. Published May 2016. Accessed August 13, 2018.
6. Barnett ML, Grabowski DC, Mehrotra A. Home-to-home time–measuring what matters to patients and payers. N Engl J Med. 2017;377(1):4-6.
7. Golden AG, Silverman MA, Mintzer MJ. Is geriatric medicine terminally ill? Ann Intern Med. 2012;156(9):654-656.
8. Nazir A, Smalbrugge M, Moser A, et al. The prevalence of burnout among nursing home physicians: an international perspective. J Am Med Dir Assoc. 2018;19(1):86-88.
9. Coleman EA, Berenson RA. Lost in transition: challenges and opportunities for improving the quality of transitional care. Ann Intern Med. 2004;141(7):533-536.
10. Kim LD, Kou L, Hu B, Gorodeski EZ, Rothberg MB. Impact of a connected care model on 30-day readmission rates from skilled nursing facilities. J Hosp Med. 2017;12(4):238-244.
11. Gloth MF, Gloth MJ. A comparative effectiveness trial between a post-acute care hospitalist model and a community-based physician model of nursing home care. J Am Med Dir Assoc. 2011;12(5):384-386.
12. Baughman AW, Cain G, Ruopp MD, et al. Improving access to care by admission process redesign in a veterans affairs skilled nursing facility. Jt Comm J Qual Patient Saf. 2018;44(8):454-462.
13. Mixon A, Smith GR, Dalal A et al. The Multi-Center Medication Reconciliation Quality Improvement Study 2 (MARQUIS2): methods and implementation. Abstract 248. Present at: Society of Hospital Medicine Annual Meeting; 2018 Apr 8 – 11, 2018; Orlando, FL. https://www.shmabstracts.com/abstract/the-multi-center-medication-reconciliation-quality-improvement-study-2-marquis2-methods-and-implementation. Accessed August 13, 2018.
14. Meltzer DO, Ruhnke GW. Redesigning care for patients at increased hospitalization risk: the comprehensive care physician model. Health Aff (Millwood). 2014;33(5):770-777.
15. Powers BW, Milstein A, Jain SH. Delivery models for high-risk older patients: back to the future? JAMA. 2016;315(1):23-24.
Huddling for High-Performing Teams
In short team huddles, trainees and PACT teamlets meet to coordinate care and identify ways to improve team processes under the guidance of faculty members who reinforce collaborative practice and continuous improvement.
In 2011, 5 US Department of Veteran Affairs (VA) medical centers were selected by the VA Office of Academic Affiliations (OAA) to establish Centers of Excellence in Primary Care Education (CoEPCE). Part of the VA New Models of Care initiative, the 5 CoEPCEs (Boise, Cleveland, San Francisco, Seattle and West Haven) are utilizing VA primary care settings to develop and test innovative approaches to prepare physician residents, nurse practitioner (NP) students and residents (postgraduate), and other health professions trainees, such as pharmacy, social work, psychology, physician assistants (PAs), dieticians, etc for primary care practice.
The CoEPCEs are interprofessional academic patient aligned care teams (PACTs) defined by VA as a PACT that has at least 2 professions of trainees on the team engaged in learning.
The San Francisco VA Health Care System (SFVAHCS) Education in PACT (EdPACT)/CoEPCE developed and implemented a workplace learning model that embeds trainees into PACT teamlets and clinic workflow.1 Trainees are organized in practice partner triads with 2 second- or third-year internal medicine residents (R2s and R3s) and 1 NP student or resident. Physician residents rotate every 2 months between inpatient and outpatient settings and NP trainees are present continuously for 12 months. In this model, each trainee in the triad has his/her own patient panel and serves as a partner who delivers care to his/her partners’ patients when they are unavailable. Didactic sessions on clinical content and on topics related to the core domains occur 3-times weekly during pre- and postclinic conferences.2
Methods
In 2015, evaluators from the OAA reviewed background documents and conducted open-ended interviews with 9 CoEPCE staff, participating trainees, VA faculty, VA facility leadership, and affiliate faculty. Informants described their involvement, challenges encountered, and benefits of the huddle to participants, veterans, and the VA.
The Huddle
With the emphasis on patient-centered medical homes and team-based care in the Affordable Care Act, there is an urgent need to develop new training models that provide future health professionals with skills that support interprofessional communication and collaborative practice.2,3 A key aim of the CoEPCE is to expand workplace learning strategies and clinical opportunities for interprofessional trainees to work together as a team to anticipate and address the health care needs of veterans. Research suggests that patient care improves when team members develop a shared understanding of each other’s skill sets, care procedures, and values.4 In 2010, the SFVAHCS began phasing in VA-mandated PACTs. Each patient-aligned care teamlet serves about 1,200 patients and is composed of physician or NP primary care provider(s) (PCPs) and a registered nurse (RN) care manager, a licensed vocational nurse (LVN), and a medical support assistant (MSA). About every 3 teamlets also work with a profession-specific team member from the Social Work and Pharmacy departments. The implementation of PACT created an opportunity for the CoEPCE to add trainees of various professions to 13 preexisting PACTs in 3 SFVAHCS primary care clinics. This arrangement benefits both trainees and teamlets: trainees learn how to collaborate with clinic staff while the clinic PACT teamlets benefit from coaching by faculty skilled in team-based care.
As part of routine clinical activities, huddles provide opportunities for workplace learning related to coordination of care, building relationships, and developing a sense of camaraderie that is essential for team-based, patient-centered care. In their ideal state, huddles are “…the hub of interprofessional, team-based care”; they provide a venue where trainees can learn communication skills, team member roles, systems issues and resources, and clinical knowledge expected of full-time providers and staff.5 Embedding faculty in huddles as huddle coaches help ensure trainees are learning and applying these skills.
Planning and Implementation
After OAA funded the CoEPCE in 2011, faculty had 6 months to develop the EdPACT curriculum, which included a team building retreat, interactive didactic sessions, and workplace learning activities (ie, huddles). In July 2011, 10 trainee triads (each consisting of 2 physician residents and either a student NP or resident NP) were added to preexisting PACTs at the San Francisco VA Medical Center primary care clinic and 2 community-based outpatient clinics.
These trainee triads partnered with their PACT teamlets and huddled for 15 minutes at the beginning of each clinic day to plan for the day’s patients and future scheduled patients and to coordinate care needs for their panel of patients. CoEPCE staff built on this basic huddle model and made the following lasting modifications:
- Developed and implemented a huddle coach model and a huddle checklist to provide structure and feedback to the huddle (Online Resources);
- Scheduled huddles in NP student/resident’s exam room to reduce the hierarchy in the trainee triad;
- Incorporated trainees from other professions and levels into the huddle (psychology fellows, pharmacy residents, social work); and
- Linked the PACT teamlet (staff) to quality improvement projects that are discussed periodically in huddles and didactics.
Curriculum. The huddle allows for practical application of the 4 core domains: interprofessional collaboration (IPC), performance improvement (PI), sustained relationships (SR), and shared decision making (SDM) that shape the CoE curriculum.
Interprofessional collaboration (IPC) is the primary domain reinforced in the huddle. Trainees learn key content in half-day team retreats held at the beginning of the academic year and in interactive didactic sessions. These sessions, which draw on concepts from the Agency for Healthcare Research and Quality’s TeamSTEPPS, an evidence-based teamwork training program for health care workers, teach skills like closed-loop communication, check-backs, negotiation, and conflict resolution.
The CoE trainee triads also lead quality improvement (QI) projects, and the huddle is a venue for getting input, which reinforces the CoE’s performance improvement (PI) curriculum. For example, PACT teamlet staff members provide trainees with feedback on proposed QI interventions, such as increasing the use of telephone visits. The huddle supports SR among team members that enhance patient care while improving the quality of the clinic experience for team members. Strengthened communications and increased understanding of team member roles and system resources supports a patient-centered approach to care and lays the foundation for SDM between patients and team members.
Faculty Roles and Development. The CoEPCE physician and NP faculty members who precept and function as huddle coaches participate in monthly 2-hour faculty development sessions to address topics related to IPE. At least 1 session each year covers review of the items on the huddle checklist, tips on how to coach a huddle, discussions of the role of huddle coaches, and feedback and mentoring skills. Many huddle coach activities are inherent to clinical precepting, such as identifying appropriate clinical resources and answering clinical questions, but the core function of the huddle coach is to facilitate effective communication among team members.
Initially, a coach may guide the huddle by rounding up team members or directing the agenda of the huddle (ie, prompting the LVN to present the day’s patients and suggesting the group identify and discuss high-risk patients). As the year progresses, coaches often take a backseat, and the huddle may be facilitated by the trainees, the RN, LVN, or a combination of all members. During the huddle, coaches also may reinforce specific communication skills, such as a “check back” or ISBAR ( Identify who you are, describe the Situation, provide Background information, offer an Assessment of the situation/needs, make a Recommendation or Request)—skills that are taught during CoE didactic sessions.
The coach may call attention to particular feedback points, such as clarification of the order as an excellent example of a check-back. Each preceptor coaches 1 huddle per precepting session. After the teams huddle, preceptors do a smaller, shorter huddle in the precepting room to share successes, such as interprofessional trainees demonstrating backup behavior (eg, “in today’s huddle, I saw a great example of backup behavior when the medicine resident offered to show the NP student how to consent someone”) and discuss challenges (eg, getting all team members to the huddle).
Resources. The CoE staff schedule at least 20 huddles per week and coordinate preceptor and room schedules. The other required resources are clinic staff (RNs, LVNs, and MSAs) and exam rooms large enough to accommodate 8 or more people. Sufficient staffing coverage and staggered huddles also are important to allow cross-coverage for other clinical duties while team members and faculty are huddling.
Monitoring and Assessment. The CoE staff administer the Team Development Measure (TDM) twice yearly and a modified version of the TEAM 360 feedback survey once per year.6-9 The TDM member gages perceptions of team functioning (cohesiveness, communication, role clarity, and clarity of goals and means). Teams meet with a facilitator to debrief their TDM results and discuss ways to improve their team processes. Three-quarters of the way through the academic year, team members also complete the modified TEAM 360 survey on trainees. Each trainee receives a report describing his/her self-ratings and aggregate team member ratings on leadership, communication, interpersonal skills, and feedback.
Partnerships
In addition to CoEPCE staff and faculty support and engagement, huddles at SFVAHCS have benefited from partnerships with VA primary care leadership and with academic affiliates. In particular, support from the VA clinic directors and nurse managers was key to instituting changes to the clinics’ structure to include interprofessional trainees in huddles.
The affiliates—the University of California, San Francisco (UCSF) School of Medicine and School of Nursing—are integral partners and assist with NP student and medicine resident recruitment. These affiliates also participate in planning and refinement of CoEPCE curricular activities. The UCSF School of Nursing, School of Medicine, and Center for Faculty Educators were involved in the planning stages of the huddle model.
Challenges and Solutions
Having a staffing ratio that supports trainee participation in and learning through huddles is critical. Preceptor coverage must be in sufficient numbers to allow preceptors to coach the huddles, and clinical staff must be adequate to create cohesive and consistent teams for trainee providers. Clinic staff turnover and changes in teamlet staff can be very disruptive. Over time, teamlet staff often know key details and helpful contextual information about particular patients and clinic processes. This knowledge may be lost with turnover among teamlet staff. If team members miss huddles due to staffing shortages and clinical duties, there may be delays and errors in patient care. For example, if information discussed in the huddle is not relayed to the absent team member in a timely or accurate manner, care may be impacted. However, potential disruptions can be mitigated by a high-functioning team with strong communication skills and situational awareness who readily assist and distribute the workload.
Consistent huddling, huddle coaches, and checklists all help stabilize the group. Integration of trainees in the PACT team initially requires extra work because trainees are part-time and have panels significantly smaller than 1,200 (which means the teamlet staff are assigned to multiple trainee and provider huddles). However, teamlet staff find working with trainee teams personally rewarding, and developing highly functioning teams helps prevent burnout. Integration of pharmacy, psychology, and social work trainees takes time and thoughtful planning of activities and contributions that enhance team functioning while not overburdening trainees with additional responsibilities. If these other professions of health trainees are joining several teams’ huddles, their role may be to weigh in as needed vs preparing for and reviewing several PCPs’ schedules and patients in advance.
Factors for Success
The VA facility and primary care clinic leadership’s commitment to supporting staff participation in huddles was critical for integrating trainees into PACTs. Additionally, VA facility commitment to implementation of PACT was a key facilitating factor. Implementation of PACT, including huddles, has not been consistent at all VA facilities.10 The CoE’s approach to integrating trainees into the huddle was an opportunity to strengthen the huddle and to teach new staff members how to participate with team members in huddles. CoEPCE leadership, which has embraced change, meets regularly with facility leadership as well as an advisory board of UCSF leaders to update them on CoE activities. A critical factor for success was CoE expertise in interprofessional education and its ability to integrate concepts from the 4 core domains into an effective workplace learning experience, including attention to the physical space, scheduling, and the development and implementation of the huddle coach role and checklist.
Accomplishments and Benefits
There is evidence that SFVAHCS team huddles are achieving their goals and CoE trainees are being trained to provide team-based, patient-centered care to veterans. Key outcomes of the CoE’s approach to huddles include components in the next sections.
Interprofessional Educational Capacity. The CoEPCE faculty and staff consider the huddle to be one of the best ways to teach interprofessional communication and collaboration, team functioning, and clinical performance. Unlike a traditional didactic, classroom-based session on interprofessional collaboration, the huddle is an opportunity for health care professionals to work together to provide care in a clinic setting. It also is an activity in which the CoE has continued innovative activities, such as adding a preceptor huddle, incorporating additional professions, and encouraging panel management activities during huddles. The CoE has received significant interest and visibility and has been invited to share the model in numerous presentations.
Participants’ Knowledge, Attitudes, Skills, and Competencies. An aim of the CoE approach to huddles was to provide trainees with general skills in the core domain interprofessional collaboration, including teamwork and communication that transfer to other settings, such as inpatient teams and specialty clinics. Learning about other professions and their scopes of practice and areas of expertise can be helpful beyond huddles and primary care. Trainees also learn concepts and practices from the other core domains:
- Performance Improvement: The huddle is a venue for utilizing clinic metrics as well as focusing on QI projects that benefit from a team approach to solving problems;
- Sustained Relationships: The huddles support and teach the importance of relationships among the team. Trainees learn about the roles of clinic staff members, and clinic staff have more opportunities to interact with trainees and become comfortable with them, supporting coordinated care; and
- Shared Decision Making: The huddle is a venue for discussing options for providing patient decision-making support, such as discussing the pros and cons of colon cancer screening with a patient, improving patient-centered care.
Additionally, huddles can address differences in trainee clinical expertise. For example, new physician interns with less experience in the clinic receive more coaching on system resources and patient histories than they might otherwise. Nurse practitioner residents often participate in more than 1 huddle team and transition to a coaching role.
Sustained Relationships, Role Clarity, and Collaboration. The huddles are structured to facilitate SRs among trainees from different professions and among the PACT teamlet in detail as a team. The huddle increases team efficiency by educating trainees and staff about team member roles. For example, trainees learn how the LVNs and MSAs prepare for patient visits. Moreover, an opportunity exists to learn how provider and clinic staff expertise may overlap. Registered nurse care managers, who have their own hypertension clinics, can help manage a patient’s medication titration. Similarly, pharmacy trainees can suggest a referral for a complicated patient with diabetes to pharmacy clinic, where clinical pharmacists can adjust medications and provide patient education for hypertension, hyperlipidemia, and hyperglycemia. In this way, role clarity is improved and trainees learn how team members work within their scope of practice and are better able to “share the care.”
There is evidence that huddles have resulted in expanded participant interprofessional collaboration. The CoE administers the TDM twice a year and the huddle teams rate themselves on several dimensions—cohesiveness, communications, role clarity, and goals.6,7 The 2011/2012 findings showed that nearly all teams showed improvement, with the mean scores for all teams combined increasing from 59.4 in the fall to 64.6 in the spring (max score is 100).5 These scores increased again from 62.2 to 70.3 in 2012/2013, from 66.6 to 70.2 in 2013/2014, and from 64.6 to 69.9 in 2014/2015.
Expanding Clinical Knowledge. At the individual level, the huddle is an opportunity for a trainees to expand their clinical expertise in real time. The huddle provides exposure to a variety of patients and corresponding patient care needs. Trainees are encouraged to complete patient prerounds before the huddle in order to focus the huddle discussion on patients with chronic conditions, complex needs, recent hospitalizations, and upcoming appointments. The CoEPCE trainees tap into the expertise and experience of their team members and coach.
The clinic staff can get information from trainees about their plan of care while trainees get a more complete picture of a patient’s situation—for example, medical or social history or communication preferences. Additionally, trainees learn team skills, such as communication techniques and warm handoffs, which can be used in other clinical settings outside primary care and beyond the VA. As trainees advance, the huddle helps them learn to delegate appropriately, practice conflict negotiation, and develop leadership skills.
Participants’ Satisfaction With Interventions. There is qualitative evidence that clinic RNs and LVNs like huddles and appreciate having the opportunity to communicate in person with providers as well as to teach trainees how to work interprofessionally. Faculty members who are huddle coaches report that they develop a richer understanding of the skill set of trainees, information that can inform CoE curriculum design. Trainees appreciate the opportunity to develop relationships with team members. In end-of-year interviews, they describe their teams as their families, making them feel more connected to the clinic. They also enjoyed starting their day with familiar faces.
Primary Care Delivery System. The huddle is an important component of a system-wide transformation to provide team-based, patient-centered care to veterans. The efforts to strengthen and standardize the huddle have the potential to hasten this transformation while improving relationships and quality of care. Additionally, the CoE approach to integrating trainees into huddles has broader applicability and is being considered for adoption by other VA centers of excellence in primary care education.Primary Care Services. The huddle may contribute to efficiencies in a busy clinic setting. For example, the RN care manager can have upward of 1,200 patients on his/her panel and, between staff and trainees, as many as 12 health care providers with whom to communicate. The huddle strengthens the communications with providers and is an opportunity to touch base on the patients, coordinate care, and keep track of high-risk patients who might fall off the radar otherwise. The huddle is flexible and can occur with various clinic staff and providers. A 2-person huddle can occur between an RN and the primary provider. The QI projects that have been developed as a result of a huddle have improved clinic primary care services, such as completing opiate consents and urine toxicology or improving continuity through increased telephone clinic usage.Patient Outcomes. The huddle results in a more robust plan of care than might be developed by an individual provider who might not have time to consider options outside the individual’s scope of practice or expertise. While there are few clinical outcomes that are directly influenced by huddles alone, huddles may help indirectly improve patient outcomes on many fronts, including:
- Increased continuity of care because the patient now has a team focusing on care. At times throughout the day when team members cannot talk face to face with one another or with the patient, they know about the patient’s situation and are better able to establish a rapport when the patient calls or comes in for the visit. Trainees also become familiar with their practice partners’ patients, which allows them to ensure continuity when the patient’s primary trainee provider is out of clinic;
- Panel management and identifying and tracking sicker patients;
- Increased access, such as identifying patients who could receive care by a telephone visit, decreasing the number of no shows by making extra efforts to remind patients about appointments and improving follow up; and
- Improved population health outcomes from process improvements, such as the development of a process for having patients on opioids sign new contracts or identifying diabetics who might benefit from a group approach to care.
The Future
The huddle coach concept and checklist have been shared broadly and have applicability in other teaching settings where providers and clinic staff are learning how to implement huddles. A video and resources on “How to Huddle” are available at suzannecgordon.com/how-to-huddle/.
Under stage 2 of the CoEPCE program, the CoE will develop a huddle coaching program implementation kit composed of a huddle how-to guide and a coach training manual. The CoE team huddle is one of many VA huddles and an example of how the huddle continues to evolve. It is a versatile tool that can be used to focus on different topics and include different professions. Currently, it is being adapted to specialty care where there is large patient volume, such as cardiology and orthopedics.
1. Rugen KW, Watts SA, Janson SL, et al. Veteran Affairs Centers of Excellence in Primary Care Education: transforming nurse practitioner education. Nurs Outlook. 2014;62(2):78-88.
2. Chang A, Bowen JL, Buranosky RA, et al. Transforming primary care training--patient-centered medical home entrustable professional activities for internal medicine residents. J Gen Intern Med. 2013;28(6):801-809.
3. Zabar S, Adams J, Kurland S, et al. Charting a key competency domain: understanding resident physician interprofessional collaboration (IPC) skills. J Gen Intern Med. 2016;31(8):846-853.
4. Institute of Medicine. Measuring the Impact of Interprofessional Education (IPE) on Collaborative Practice and Patient Outcomes. Washington, DC: The National Academies Press; 2015.
5. Shunk R, Dulay M, Chou C, Janson S, O’Brien BC. Huddle-coaching: a dynamic intervention for trainees and staff to support team-based care. Acad Med. 2014;89(2):244-250.
6. Stock R, Mahoney E, Carney PA. Measuring team development in clinical care settings. Fam Med. 2013;45(10):691-700.
7. PeaceHealth. Team development measure. https://www.peacehealth.org/about-peacehealth/medical-professionals/eugene-springfield-cottage-grove/team-measure/Pages/measure. Accessed August 16, 2018.
8. American Board of Internal Medicine. Teamwork effectiveness assessment module. https://team.abim.org. Accessed August 16, 2018.
9. Chesluk BJ, Bernabeo E, Hess B, Lynn LA, Reddy S, Holmboe ES. A new tool to give hospitalists feedback to improve interprofessional teamwork and advance patient care. Health Aff (Millwood). 2012;31(11):2485-2492.
10. Rodriguez HP, Meredith LS, Hamilton AB, Yano EM, Rubenstein LV. Huddle up!: the adoption and use of structured team communication for VA medical home implementation. Health Care Manage Rev. 2015;40(4):286-299.
In short team huddles, trainees and PACT teamlets meet to coordinate care and identify ways to improve team processes under the guidance of faculty members who reinforce collaborative practice and continuous improvement.
In short team huddles, trainees and PACT teamlets meet to coordinate care and identify ways to improve team processes under the guidance of faculty members who reinforce collaborative practice and continuous improvement.
In 2011, 5 US Department of Veteran Affairs (VA) medical centers were selected by the VA Office of Academic Affiliations (OAA) to establish Centers of Excellence in Primary Care Education (CoEPCE). Part of the VA New Models of Care initiative, the 5 CoEPCEs (Boise, Cleveland, San Francisco, Seattle and West Haven) are utilizing VA primary care settings to develop and test innovative approaches to prepare physician residents, nurse practitioner (NP) students and residents (postgraduate), and other health professions trainees, such as pharmacy, social work, psychology, physician assistants (PAs), dieticians, etc for primary care practice.
The CoEPCEs are interprofessional academic patient aligned care teams (PACTs) defined by VA as a PACT that has at least 2 professions of trainees on the team engaged in learning.
The San Francisco VA Health Care System (SFVAHCS) Education in PACT (EdPACT)/CoEPCE developed and implemented a workplace learning model that embeds trainees into PACT teamlets and clinic workflow.1 Trainees are organized in practice partner triads with 2 second- or third-year internal medicine residents (R2s and R3s) and 1 NP student or resident. Physician residents rotate every 2 months between inpatient and outpatient settings and NP trainees are present continuously for 12 months. In this model, each trainee in the triad has his/her own patient panel and serves as a partner who delivers care to his/her partners’ patients when they are unavailable. Didactic sessions on clinical content and on topics related to the core domains occur 3-times weekly during pre- and postclinic conferences.2
Methods
In 2015, evaluators from the OAA reviewed background documents and conducted open-ended interviews with 9 CoEPCE staff, participating trainees, VA faculty, VA facility leadership, and affiliate faculty. Informants described their involvement, challenges encountered, and benefits of the huddle to participants, veterans, and the VA.
The Huddle
With the emphasis on patient-centered medical homes and team-based care in the Affordable Care Act, there is an urgent need to develop new training models that provide future health professionals with skills that support interprofessional communication and collaborative practice.2,3 A key aim of the CoEPCE is to expand workplace learning strategies and clinical opportunities for interprofessional trainees to work together as a team to anticipate and address the health care needs of veterans. Research suggests that patient care improves when team members develop a shared understanding of each other’s skill sets, care procedures, and values.4 In 2010, the SFVAHCS began phasing in VA-mandated PACTs. Each patient-aligned care teamlet serves about 1,200 patients and is composed of physician or NP primary care provider(s) (PCPs) and a registered nurse (RN) care manager, a licensed vocational nurse (LVN), and a medical support assistant (MSA). About every 3 teamlets also work with a profession-specific team member from the Social Work and Pharmacy departments. The implementation of PACT created an opportunity for the CoEPCE to add trainees of various professions to 13 preexisting PACTs in 3 SFVAHCS primary care clinics. This arrangement benefits both trainees and teamlets: trainees learn how to collaborate with clinic staff while the clinic PACT teamlets benefit from coaching by faculty skilled in team-based care.
As part of routine clinical activities, huddles provide opportunities for workplace learning related to coordination of care, building relationships, and developing a sense of camaraderie that is essential for team-based, patient-centered care. In their ideal state, huddles are “…the hub of interprofessional, team-based care”; they provide a venue where trainees can learn communication skills, team member roles, systems issues and resources, and clinical knowledge expected of full-time providers and staff.5 Embedding faculty in huddles as huddle coaches help ensure trainees are learning and applying these skills.
Planning and Implementation
After OAA funded the CoEPCE in 2011, faculty had 6 months to develop the EdPACT curriculum, which included a team building retreat, interactive didactic sessions, and workplace learning activities (ie, huddles). In July 2011, 10 trainee triads (each consisting of 2 physician residents and either a student NP or resident NP) were added to preexisting PACTs at the San Francisco VA Medical Center primary care clinic and 2 community-based outpatient clinics.
These trainee triads partnered with their PACT teamlets and huddled for 15 minutes at the beginning of each clinic day to plan for the day’s patients and future scheduled patients and to coordinate care needs for their panel of patients. CoEPCE staff built on this basic huddle model and made the following lasting modifications:
- Developed and implemented a huddle coach model and a huddle checklist to provide structure and feedback to the huddle (Online Resources);
- Scheduled huddles in NP student/resident’s exam room to reduce the hierarchy in the trainee triad;
- Incorporated trainees from other professions and levels into the huddle (psychology fellows, pharmacy residents, social work); and
- Linked the PACT teamlet (staff) to quality improvement projects that are discussed periodically in huddles and didactics.
Curriculum. The huddle allows for practical application of the 4 core domains: interprofessional collaboration (IPC), performance improvement (PI), sustained relationships (SR), and shared decision making (SDM) that shape the CoE curriculum.
Interprofessional collaboration (IPC) is the primary domain reinforced in the huddle. Trainees learn key content in half-day team retreats held at the beginning of the academic year and in interactive didactic sessions. These sessions, which draw on concepts from the Agency for Healthcare Research and Quality’s TeamSTEPPS, an evidence-based teamwork training program for health care workers, teach skills like closed-loop communication, check-backs, negotiation, and conflict resolution.
The CoE trainee triads also lead quality improvement (QI) projects, and the huddle is a venue for getting input, which reinforces the CoE’s performance improvement (PI) curriculum. For example, PACT teamlet staff members provide trainees with feedback on proposed QI interventions, such as increasing the use of telephone visits. The huddle supports SR among team members that enhance patient care while improving the quality of the clinic experience for team members. Strengthened communications and increased understanding of team member roles and system resources supports a patient-centered approach to care and lays the foundation for SDM between patients and team members.
Faculty Roles and Development. The CoEPCE physician and NP faculty members who precept and function as huddle coaches participate in monthly 2-hour faculty development sessions to address topics related to IPE. At least 1 session each year covers review of the items on the huddle checklist, tips on how to coach a huddle, discussions of the role of huddle coaches, and feedback and mentoring skills. Many huddle coach activities are inherent to clinical precepting, such as identifying appropriate clinical resources and answering clinical questions, but the core function of the huddle coach is to facilitate effective communication among team members.
Initially, a coach may guide the huddle by rounding up team members or directing the agenda of the huddle (ie, prompting the LVN to present the day’s patients and suggesting the group identify and discuss high-risk patients). As the year progresses, coaches often take a backseat, and the huddle may be facilitated by the trainees, the RN, LVN, or a combination of all members. During the huddle, coaches also may reinforce specific communication skills, such as a “check back” or ISBAR ( Identify who you are, describe the Situation, provide Background information, offer an Assessment of the situation/needs, make a Recommendation or Request)—skills that are taught during CoE didactic sessions.
The coach may call attention to particular feedback points, such as clarification of the order as an excellent example of a check-back. Each preceptor coaches 1 huddle per precepting session. After the teams huddle, preceptors do a smaller, shorter huddle in the precepting room to share successes, such as interprofessional trainees demonstrating backup behavior (eg, “in today’s huddle, I saw a great example of backup behavior when the medicine resident offered to show the NP student how to consent someone”) and discuss challenges (eg, getting all team members to the huddle).
Resources. The CoE staff schedule at least 20 huddles per week and coordinate preceptor and room schedules. The other required resources are clinic staff (RNs, LVNs, and MSAs) and exam rooms large enough to accommodate 8 or more people. Sufficient staffing coverage and staggered huddles also are important to allow cross-coverage for other clinical duties while team members and faculty are huddling.
Monitoring and Assessment. The CoE staff administer the Team Development Measure (TDM) twice yearly and a modified version of the TEAM 360 feedback survey once per year.6-9 The TDM member gages perceptions of team functioning (cohesiveness, communication, role clarity, and clarity of goals and means). Teams meet with a facilitator to debrief their TDM results and discuss ways to improve their team processes. Three-quarters of the way through the academic year, team members also complete the modified TEAM 360 survey on trainees. Each trainee receives a report describing his/her self-ratings and aggregate team member ratings on leadership, communication, interpersonal skills, and feedback.
Partnerships
In addition to CoEPCE staff and faculty support and engagement, huddles at SFVAHCS have benefited from partnerships with VA primary care leadership and with academic affiliates. In particular, support from the VA clinic directors and nurse managers was key to instituting changes to the clinics’ structure to include interprofessional trainees in huddles.
The affiliates—the University of California, San Francisco (UCSF) School of Medicine and School of Nursing—are integral partners and assist with NP student and medicine resident recruitment. These affiliates also participate in planning and refinement of CoEPCE curricular activities. The UCSF School of Nursing, School of Medicine, and Center for Faculty Educators were involved in the planning stages of the huddle model.
Challenges and Solutions
Having a staffing ratio that supports trainee participation in and learning through huddles is critical. Preceptor coverage must be in sufficient numbers to allow preceptors to coach the huddles, and clinical staff must be adequate to create cohesive and consistent teams for trainee providers. Clinic staff turnover and changes in teamlet staff can be very disruptive. Over time, teamlet staff often know key details and helpful contextual information about particular patients and clinic processes. This knowledge may be lost with turnover among teamlet staff. If team members miss huddles due to staffing shortages and clinical duties, there may be delays and errors in patient care. For example, if information discussed in the huddle is not relayed to the absent team member in a timely or accurate manner, care may be impacted. However, potential disruptions can be mitigated by a high-functioning team with strong communication skills and situational awareness who readily assist and distribute the workload.
Consistent huddling, huddle coaches, and checklists all help stabilize the group. Integration of trainees in the PACT team initially requires extra work because trainees are part-time and have panels significantly smaller than 1,200 (which means the teamlet staff are assigned to multiple trainee and provider huddles). However, teamlet staff find working with trainee teams personally rewarding, and developing highly functioning teams helps prevent burnout. Integration of pharmacy, psychology, and social work trainees takes time and thoughtful planning of activities and contributions that enhance team functioning while not overburdening trainees with additional responsibilities. If these other professions of health trainees are joining several teams’ huddles, their role may be to weigh in as needed vs preparing for and reviewing several PCPs’ schedules and patients in advance.
Factors for Success
The VA facility and primary care clinic leadership’s commitment to supporting staff participation in huddles was critical for integrating trainees into PACTs. Additionally, VA facility commitment to implementation of PACT was a key facilitating factor. Implementation of PACT, including huddles, has not been consistent at all VA facilities.10 The CoE’s approach to integrating trainees into the huddle was an opportunity to strengthen the huddle and to teach new staff members how to participate with team members in huddles. CoEPCE leadership, which has embraced change, meets regularly with facility leadership as well as an advisory board of UCSF leaders to update them on CoE activities. A critical factor for success was CoE expertise in interprofessional education and its ability to integrate concepts from the 4 core domains into an effective workplace learning experience, including attention to the physical space, scheduling, and the development and implementation of the huddle coach role and checklist.
Accomplishments and Benefits
There is evidence that SFVAHCS team huddles are achieving their goals and CoE trainees are being trained to provide team-based, patient-centered care to veterans. Key outcomes of the CoE’s approach to huddles include components in the next sections.
Interprofessional Educational Capacity. The CoEPCE faculty and staff consider the huddle to be one of the best ways to teach interprofessional communication and collaboration, team functioning, and clinical performance. Unlike a traditional didactic, classroom-based session on interprofessional collaboration, the huddle is an opportunity for health care professionals to work together to provide care in a clinic setting. It also is an activity in which the CoE has continued innovative activities, such as adding a preceptor huddle, incorporating additional professions, and encouraging panel management activities during huddles. The CoE has received significant interest and visibility and has been invited to share the model in numerous presentations.
Participants’ Knowledge, Attitudes, Skills, and Competencies. An aim of the CoE approach to huddles was to provide trainees with general skills in the core domain interprofessional collaboration, including teamwork and communication that transfer to other settings, such as inpatient teams and specialty clinics. Learning about other professions and their scopes of practice and areas of expertise can be helpful beyond huddles and primary care. Trainees also learn concepts and practices from the other core domains:
- Performance Improvement: The huddle is a venue for utilizing clinic metrics as well as focusing on QI projects that benefit from a team approach to solving problems;
- Sustained Relationships: The huddles support and teach the importance of relationships among the team. Trainees learn about the roles of clinic staff members, and clinic staff have more opportunities to interact with trainees and become comfortable with them, supporting coordinated care; and
- Shared Decision Making: The huddle is a venue for discussing options for providing patient decision-making support, such as discussing the pros and cons of colon cancer screening with a patient, improving patient-centered care.
Additionally, huddles can address differences in trainee clinical expertise. For example, new physician interns with less experience in the clinic receive more coaching on system resources and patient histories than they might otherwise. Nurse practitioner residents often participate in more than 1 huddle team and transition to a coaching role.
Sustained Relationships, Role Clarity, and Collaboration. The huddles are structured to facilitate SRs among trainees from different professions and among the PACT teamlet in detail as a team. The huddle increases team efficiency by educating trainees and staff about team member roles. For example, trainees learn how the LVNs and MSAs prepare for patient visits. Moreover, an opportunity exists to learn how provider and clinic staff expertise may overlap. Registered nurse care managers, who have their own hypertension clinics, can help manage a patient’s medication titration. Similarly, pharmacy trainees can suggest a referral for a complicated patient with diabetes to pharmacy clinic, where clinical pharmacists can adjust medications and provide patient education for hypertension, hyperlipidemia, and hyperglycemia. In this way, role clarity is improved and trainees learn how team members work within their scope of practice and are better able to “share the care.”
There is evidence that huddles have resulted in expanded participant interprofessional collaboration. The CoE administers the TDM twice a year and the huddle teams rate themselves on several dimensions—cohesiveness, communications, role clarity, and goals.6,7 The 2011/2012 findings showed that nearly all teams showed improvement, with the mean scores for all teams combined increasing from 59.4 in the fall to 64.6 in the spring (max score is 100).5 These scores increased again from 62.2 to 70.3 in 2012/2013, from 66.6 to 70.2 in 2013/2014, and from 64.6 to 69.9 in 2014/2015.
Expanding Clinical Knowledge. At the individual level, the huddle is an opportunity for a trainees to expand their clinical expertise in real time. The huddle provides exposure to a variety of patients and corresponding patient care needs. Trainees are encouraged to complete patient prerounds before the huddle in order to focus the huddle discussion on patients with chronic conditions, complex needs, recent hospitalizations, and upcoming appointments. The CoEPCE trainees tap into the expertise and experience of their team members and coach.
The clinic staff can get information from trainees about their plan of care while trainees get a more complete picture of a patient’s situation—for example, medical or social history or communication preferences. Additionally, trainees learn team skills, such as communication techniques and warm handoffs, which can be used in other clinical settings outside primary care and beyond the VA. As trainees advance, the huddle helps them learn to delegate appropriately, practice conflict negotiation, and develop leadership skills.
Participants’ Satisfaction With Interventions. There is qualitative evidence that clinic RNs and LVNs like huddles and appreciate having the opportunity to communicate in person with providers as well as to teach trainees how to work interprofessionally. Faculty members who are huddle coaches report that they develop a richer understanding of the skill set of trainees, information that can inform CoE curriculum design. Trainees appreciate the opportunity to develop relationships with team members. In end-of-year interviews, they describe their teams as their families, making them feel more connected to the clinic. They also enjoyed starting their day with familiar faces.
Primary Care Delivery System. The huddle is an important component of a system-wide transformation to provide team-based, patient-centered care to veterans. The efforts to strengthen and standardize the huddle have the potential to hasten this transformation while improving relationships and quality of care. Additionally, the CoE approach to integrating trainees into huddles has broader applicability and is being considered for adoption by other VA centers of excellence in primary care education.Primary Care Services. The huddle may contribute to efficiencies in a busy clinic setting. For example, the RN care manager can have upward of 1,200 patients on his/her panel and, between staff and trainees, as many as 12 health care providers with whom to communicate. The huddle strengthens the communications with providers and is an opportunity to touch base on the patients, coordinate care, and keep track of high-risk patients who might fall off the radar otherwise. The huddle is flexible and can occur with various clinic staff and providers. A 2-person huddle can occur between an RN and the primary provider. The QI projects that have been developed as a result of a huddle have improved clinic primary care services, such as completing opiate consents and urine toxicology or improving continuity through increased telephone clinic usage.Patient Outcomes. The huddle results in a more robust plan of care than might be developed by an individual provider who might not have time to consider options outside the individual’s scope of practice or expertise. While there are few clinical outcomes that are directly influenced by huddles alone, huddles may help indirectly improve patient outcomes on many fronts, including:
- Increased continuity of care because the patient now has a team focusing on care. At times throughout the day when team members cannot talk face to face with one another or with the patient, they know about the patient’s situation and are better able to establish a rapport when the patient calls or comes in for the visit. Trainees also become familiar with their practice partners’ patients, which allows them to ensure continuity when the patient’s primary trainee provider is out of clinic;
- Panel management and identifying and tracking sicker patients;
- Increased access, such as identifying patients who could receive care by a telephone visit, decreasing the number of no shows by making extra efforts to remind patients about appointments and improving follow up; and
- Improved population health outcomes from process improvements, such as the development of a process for having patients on opioids sign new contracts or identifying diabetics who might benefit from a group approach to care.
The Future
The huddle coach concept and checklist have been shared broadly and have applicability in other teaching settings where providers and clinic staff are learning how to implement huddles. A video and resources on “How to Huddle” are available at suzannecgordon.com/how-to-huddle/.
Under stage 2 of the CoEPCE program, the CoE will develop a huddle coaching program implementation kit composed of a huddle how-to guide and a coach training manual. The CoE team huddle is one of many VA huddles and an example of how the huddle continues to evolve. It is a versatile tool that can be used to focus on different topics and include different professions. Currently, it is being adapted to specialty care where there is large patient volume, such as cardiology and orthopedics.
In 2011, 5 US Department of Veteran Affairs (VA) medical centers were selected by the VA Office of Academic Affiliations (OAA) to establish Centers of Excellence in Primary Care Education (CoEPCE). Part of the VA New Models of Care initiative, the 5 CoEPCEs (Boise, Cleveland, San Francisco, Seattle and West Haven) are utilizing VA primary care settings to develop and test innovative approaches to prepare physician residents, nurse practitioner (NP) students and residents (postgraduate), and other health professions trainees, such as pharmacy, social work, psychology, physician assistants (PAs), dieticians, etc for primary care practice.
The CoEPCEs are interprofessional academic patient aligned care teams (PACTs) defined by VA as a PACT that has at least 2 professions of trainees on the team engaged in learning.
The San Francisco VA Health Care System (SFVAHCS) Education in PACT (EdPACT)/CoEPCE developed and implemented a workplace learning model that embeds trainees into PACT teamlets and clinic workflow.1 Trainees are organized in practice partner triads with 2 second- or third-year internal medicine residents (R2s and R3s) and 1 NP student or resident. Physician residents rotate every 2 months between inpatient and outpatient settings and NP trainees are present continuously for 12 months. In this model, each trainee in the triad has his/her own patient panel and serves as a partner who delivers care to his/her partners’ patients when they are unavailable. Didactic sessions on clinical content and on topics related to the core domains occur 3-times weekly during pre- and postclinic conferences.2
Methods
In 2015, evaluators from the OAA reviewed background documents and conducted open-ended interviews with 9 CoEPCE staff, participating trainees, VA faculty, VA facility leadership, and affiliate faculty. Informants described their involvement, challenges encountered, and benefits of the huddle to participants, veterans, and the VA.
The Huddle
With the emphasis on patient-centered medical homes and team-based care in the Affordable Care Act, there is an urgent need to develop new training models that provide future health professionals with skills that support interprofessional communication and collaborative practice.2,3 A key aim of the CoEPCE is to expand workplace learning strategies and clinical opportunities for interprofessional trainees to work together as a team to anticipate and address the health care needs of veterans. Research suggests that patient care improves when team members develop a shared understanding of each other’s skill sets, care procedures, and values.4 In 2010, the SFVAHCS began phasing in VA-mandated PACTs. Each patient-aligned care teamlet serves about 1,200 patients and is composed of physician or NP primary care provider(s) (PCPs) and a registered nurse (RN) care manager, a licensed vocational nurse (LVN), and a medical support assistant (MSA). About every 3 teamlets also work with a profession-specific team member from the Social Work and Pharmacy departments. The implementation of PACT created an opportunity for the CoEPCE to add trainees of various professions to 13 preexisting PACTs in 3 SFVAHCS primary care clinics. This arrangement benefits both trainees and teamlets: trainees learn how to collaborate with clinic staff while the clinic PACT teamlets benefit from coaching by faculty skilled in team-based care.
As part of routine clinical activities, huddles provide opportunities for workplace learning related to coordination of care, building relationships, and developing a sense of camaraderie that is essential for team-based, patient-centered care. In their ideal state, huddles are “…the hub of interprofessional, team-based care”; they provide a venue where trainees can learn communication skills, team member roles, systems issues and resources, and clinical knowledge expected of full-time providers and staff.5 Embedding faculty in huddles as huddle coaches help ensure trainees are learning and applying these skills.
Planning and Implementation
After OAA funded the CoEPCE in 2011, faculty had 6 months to develop the EdPACT curriculum, which included a team building retreat, interactive didactic sessions, and workplace learning activities (ie, huddles). In July 2011, 10 trainee triads (each consisting of 2 physician residents and either a student NP or resident NP) were added to preexisting PACTs at the San Francisco VA Medical Center primary care clinic and 2 community-based outpatient clinics.
These trainee triads partnered with their PACT teamlets and huddled for 15 minutes at the beginning of each clinic day to plan for the day’s patients and future scheduled patients and to coordinate care needs for their panel of patients. CoEPCE staff built on this basic huddle model and made the following lasting modifications:
- Developed and implemented a huddle coach model and a huddle checklist to provide structure and feedback to the huddle (Online Resources);
- Scheduled huddles in NP student/resident’s exam room to reduce the hierarchy in the trainee triad;
- Incorporated trainees from other professions and levels into the huddle (psychology fellows, pharmacy residents, social work); and
- Linked the PACT teamlet (staff) to quality improvement projects that are discussed periodically in huddles and didactics.
Curriculum. The huddle allows for practical application of the 4 core domains: interprofessional collaboration (IPC), performance improvement (PI), sustained relationships (SR), and shared decision making (SDM) that shape the CoE curriculum.
Interprofessional collaboration (IPC) is the primary domain reinforced in the huddle. Trainees learn key content in half-day team retreats held at the beginning of the academic year and in interactive didactic sessions. These sessions, which draw on concepts from the Agency for Healthcare Research and Quality’s TeamSTEPPS, an evidence-based teamwork training program for health care workers, teach skills like closed-loop communication, check-backs, negotiation, and conflict resolution.
The CoE trainee triads also lead quality improvement (QI) projects, and the huddle is a venue for getting input, which reinforces the CoE’s performance improvement (PI) curriculum. For example, PACT teamlet staff members provide trainees with feedback on proposed QI interventions, such as increasing the use of telephone visits. The huddle supports SR among team members that enhance patient care while improving the quality of the clinic experience for team members. Strengthened communications and increased understanding of team member roles and system resources supports a patient-centered approach to care and lays the foundation for SDM between patients and team members.
Faculty Roles and Development. The CoEPCE physician and NP faculty members who precept and function as huddle coaches participate in monthly 2-hour faculty development sessions to address topics related to IPE. At least 1 session each year covers review of the items on the huddle checklist, tips on how to coach a huddle, discussions of the role of huddle coaches, and feedback and mentoring skills. Many huddle coach activities are inherent to clinical precepting, such as identifying appropriate clinical resources and answering clinical questions, but the core function of the huddle coach is to facilitate effective communication among team members.
Initially, a coach may guide the huddle by rounding up team members or directing the agenda of the huddle (ie, prompting the LVN to present the day’s patients and suggesting the group identify and discuss high-risk patients). As the year progresses, coaches often take a backseat, and the huddle may be facilitated by the trainees, the RN, LVN, or a combination of all members. During the huddle, coaches also may reinforce specific communication skills, such as a “check back” or ISBAR ( Identify who you are, describe the Situation, provide Background information, offer an Assessment of the situation/needs, make a Recommendation or Request)—skills that are taught during CoE didactic sessions.
The coach may call attention to particular feedback points, such as clarification of the order as an excellent example of a check-back. Each preceptor coaches 1 huddle per precepting session. After the teams huddle, preceptors do a smaller, shorter huddle in the precepting room to share successes, such as interprofessional trainees demonstrating backup behavior (eg, “in today’s huddle, I saw a great example of backup behavior when the medicine resident offered to show the NP student how to consent someone”) and discuss challenges (eg, getting all team members to the huddle).
Resources. The CoE staff schedule at least 20 huddles per week and coordinate preceptor and room schedules. The other required resources are clinic staff (RNs, LVNs, and MSAs) and exam rooms large enough to accommodate 8 or more people. Sufficient staffing coverage and staggered huddles also are important to allow cross-coverage for other clinical duties while team members and faculty are huddling.
Monitoring and Assessment. The CoE staff administer the Team Development Measure (TDM) twice yearly and a modified version of the TEAM 360 feedback survey once per year.6-9 The TDM member gages perceptions of team functioning (cohesiveness, communication, role clarity, and clarity of goals and means). Teams meet with a facilitator to debrief their TDM results and discuss ways to improve their team processes. Three-quarters of the way through the academic year, team members also complete the modified TEAM 360 survey on trainees. Each trainee receives a report describing his/her self-ratings and aggregate team member ratings on leadership, communication, interpersonal skills, and feedback.
Partnerships
In addition to CoEPCE staff and faculty support and engagement, huddles at SFVAHCS have benefited from partnerships with VA primary care leadership and with academic affiliates. In particular, support from the VA clinic directors and nurse managers was key to instituting changes to the clinics’ structure to include interprofessional trainees in huddles.
The affiliates—the University of California, San Francisco (UCSF) School of Medicine and School of Nursing—are integral partners and assist with NP student and medicine resident recruitment. These affiliates also participate in planning and refinement of CoEPCE curricular activities. The UCSF School of Nursing, School of Medicine, and Center for Faculty Educators were involved in the planning stages of the huddle model.
Challenges and Solutions
Having a staffing ratio that supports trainee participation in and learning through huddles is critical. Preceptor coverage must be in sufficient numbers to allow preceptors to coach the huddles, and clinical staff must be adequate to create cohesive and consistent teams for trainee providers. Clinic staff turnover and changes in teamlet staff can be very disruptive. Over time, teamlet staff often know key details and helpful contextual information about particular patients and clinic processes. This knowledge may be lost with turnover among teamlet staff. If team members miss huddles due to staffing shortages and clinical duties, there may be delays and errors in patient care. For example, if information discussed in the huddle is not relayed to the absent team member in a timely or accurate manner, care may be impacted. However, potential disruptions can be mitigated by a high-functioning team with strong communication skills and situational awareness who readily assist and distribute the workload.
Consistent huddling, huddle coaches, and checklists all help stabilize the group. Integration of trainees in the PACT team initially requires extra work because trainees are part-time and have panels significantly smaller than 1,200 (which means the teamlet staff are assigned to multiple trainee and provider huddles). However, teamlet staff find working with trainee teams personally rewarding, and developing highly functioning teams helps prevent burnout. Integration of pharmacy, psychology, and social work trainees takes time and thoughtful planning of activities and contributions that enhance team functioning while not overburdening trainees with additional responsibilities. If these other professions of health trainees are joining several teams’ huddles, their role may be to weigh in as needed vs preparing for and reviewing several PCPs’ schedules and patients in advance.
Factors for Success
The VA facility and primary care clinic leadership’s commitment to supporting staff participation in huddles was critical for integrating trainees into PACTs. Additionally, VA facility commitment to implementation of PACT was a key facilitating factor. Implementation of PACT, including huddles, has not been consistent at all VA facilities.10 The CoE’s approach to integrating trainees into the huddle was an opportunity to strengthen the huddle and to teach new staff members how to participate with team members in huddles. CoEPCE leadership, which has embraced change, meets regularly with facility leadership as well as an advisory board of UCSF leaders to update them on CoE activities. A critical factor for success was CoE expertise in interprofessional education and its ability to integrate concepts from the 4 core domains into an effective workplace learning experience, including attention to the physical space, scheduling, and the development and implementation of the huddle coach role and checklist.
Accomplishments and Benefits
There is evidence that SFVAHCS team huddles are achieving their goals and CoE trainees are being trained to provide team-based, patient-centered care to veterans. Key outcomes of the CoE’s approach to huddles include components in the next sections.
Interprofessional Educational Capacity. The CoEPCE faculty and staff consider the huddle to be one of the best ways to teach interprofessional communication and collaboration, team functioning, and clinical performance. Unlike a traditional didactic, classroom-based session on interprofessional collaboration, the huddle is an opportunity for health care professionals to work together to provide care in a clinic setting. It also is an activity in which the CoE has continued innovative activities, such as adding a preceptor huddle, incorporating additional professions, and encouraging panel management activities during huddles. The CoE has received significant interest and visibility and has been invited to share the model in numerous presentations.
Participants’ Knowledge, Attitudes, Skills, and Competencies. An aim of the CoE approach to huddles was to provide trainees with general skills in the core domain interprofessional collaboration, including teamwork and communication that transfer to other settings, such as inpatient teams and specialty clinics. Learning about other professions and their scopes of practice and areas of expertise can be helpful beyond huddles and primary care. Trainees also learn concepts and practices from the other core domains:
- Performance Improvement: The huddle is a venue for utilizing clinic metrics as well as focusing on QI projects that benefit from a team approach to solving problems;
- Sustained Relationships: The huddles support and teach the importance of relationships among the team. Trainees learn about the roles of clinic staff members, and clinic staff have more opportunities to interact with trainees and become comfortable with them, supporting coordinated care; and
- Shared Decision Making: The huddle is a venue for discussing options for providing patient decision-making support, such as discussing the pros and cons of colon cancer screening with a patient, improving patient-centered care.
Additionally, huddles can address differences in trainee clinical expertise. For example, new physician interns with less experience in the clinic receive more coaching on system resources and patient histories than they might otherwise. Nurse practitioner residents often participate in more than 1 huddle team and transition to a coaching role.
Sustained Relationships, Role Clarity, and Collaboration. The huddles are structured to facilitate SRs among trainees from different professions and among the PACT teamlet in detail as a team. The huddle increases team efficiency by educating trainees and staff about team member roles. For example, trainees learn how the LVNs and MSAs prepare for patient visits. Moreover, an opportunity exists to learn how provider and clinic staff expertise may overlap. Registered nurse care managers, who have their own hypertension clinics, can help manage a patient’s medication titration. Similarly, pharmacy trainees can suggest a referral for a complicated patient with diabetes to pharmacy clinic, where clinical pharmacists can adjust medications and provide patient education for hypertension, hyperlipidemia, and hyperglycemia. In this way, role clarity is improved and trainees learn how team members work within their scope of practice and are better able to “share the care.”
There is evidence that huddles have resulted in expanded participant interprofessional collaboration. The CoE administers the TDM twice a year and the huddle teams rate themselves on several dimensions—cohesiveness, communications, role clarity, and goals.6,7 The 2011/2012 findings showed that nearly all teams showed improvement, with the mean scores for all teams combined increasing from 59.4 in the fall to 64.6 in the spring (max score is 100).5 These scores increased again from 62.2 to 70.3 in 2012/2013, from 66.6 to 70.2 in 2013/2014, and from 64.6 to 69.9 in 2014/2015.
Expanding Clinical Knowledge. At the individual level, the huddle is an opportunity for a trainees to expand their clinical expertise in real time. The huddle provides exposure to a variety of patients and corresponding patient care needs. Trainees are encouraged to complete patient prerounds before the huddle in order to focus the huddle discussion on patients with chronic conditions, complex needs, recent hospitalizations, and upcoming appointments. The CoEPCE trainees tap into the expertise and experience of their team members and coach.
The clinic staff can get information from trainees about their plan of care while trainees get a more complete picture of a patient’s situation—for example, medical or social history or communication preferences. Additionally, trainees learn team skills, such as communication techniques and warm handoffs, which can be used in other clinical settings outside primary care and beyond the VA. As trainees advance, the huddle helps them learn to delegate appropriately, practice conflict negotiation, and develop leadership skills.
Participants’ Satisfaction With Interventions. There is qualitative evidence that clinic RNs and LVNs like huddles and appreciate having the opportunity to communicate in person with providers as well as to teach trainees how to work interprofessionally. Faculty members who are huddle coaches report that they develop a richer understanding of the skill set of trainees, information that can inform CoE curriculum design. Trainees appreciate the opportunity to develop relationships with team members. In end-of-year interviews, they describe their teams as their families, making them feel more connected to the clinic. They also enjoyed starting their day with familiar faces.
Primary Care Delivery System. The huddle is an important component of a system-wide transformation to provide team-based, patient-centered care to veterans. The efforts to strengthen and standardize the huddle have the potential to hasten this transformation while improving relationships and quality of care. Additionally, the CoE approach to integrating trainees into huddles has broader applicability and is being considered for adoption by other VA centers of excellence in primary care education.Primary Care Services. The huddle may contribute to efficiencies in a busy clinic setting. For example, the RN care manager can have upward of 1,200 patients on his/her panel and, between staff and trainees, as many as 12 health care providers with whom to communicate. The huddle strengthens the communications with providers and is an opportunity to touch base on the patients, coordinate care, and keep track of high-risk patients who might fall off the radar otherwise. The huddle is flexible and can occur with various clinic staff and providers. A 2-person huddle can occur between an RN and the primary provider. The QI projects that have been developed as a result of a huddle have improved clinic primary care services, such as completing opiate consents and urine toxicology or improving continuity through increased telephone clinic usage.Patient Outcomes. The huddle results in a more robust plan of care than might be developed by an individual provider who might not have time to consider options outside the individual’s scope of practice or expertise. While there are few clinical outcomes that are directly influenced by huddles alone, huddles may help indirectly improve patient outcomes on many fronts, including:
- Increased continuity of care because the patient now has a team focusing on care. At times throughout the day when team members cannot talk face to face with one another or with the patient, they know about the patient’s situation and are better able to establish a rapport when the patient calls or comes in for the visit. Trainees also become familiar with their practice partners’ patients, which allows them to ensure continuity when the patient’s primary trainee provider is out of clinic;
- Panel management and identifying and tracking sicker patients;
- Increased access, such as identifying patients who could receive care by a telephone visit, decreasing the number of no shows by making extra efforts to remind patients about appointments and improving follow up; and
- Improved population health outcomes from process improvements, such as the development of a process for having patients on opioids sign new contracts or identifying diabetics who might benefit from a group approach to care.
The Future
The huddle coach concept and checklist have been shared broadly and have applicability in other teaching settings where providers and clinic staff are learning how to implement huddles. A video and resources on “How to Huddle” are available at suzannecgordon.com/how-to-huddle/.
Under stage 2 of the CoEPCE program, the CoE will develop a huddle coaching program implementation kit composed of a huddle how-to guide and a coach training manual. The CoE team huddle is one of many VA huddles and an example of how the huddle continues to evolve. It is a versatile tool that can be used to focus on different topics and include different professions. Currently, it is being adapted to specialty care where there is large patient volume, such as cardiology and orthopedics.
1. Rugen KW, Watts SA, Janson SL, et al. Veteran Affairs Centers of Excellence in Primary Care Education: transforming nurse practitioner education. Nurs Outlook. 2014;62(2):78-88.
2. Chang A, Bowen JL, Buranosky RA, et al. Transforming primary care training--patient-centered medical home entrustable professional activities for internal medicine residents. J Gen Intern Med. 2013;28(6):801-809.
3. Zabar S, Adams J, Kurland S, et al. Charting a key competency domain: understanding resident physician interprofessional collaboration (IPC) skills. J Gen Intern Med. 2016;31(8):846-853.
4. Institute of Medicine. Measuring the Impact of Interprofessional Education (IPE) on Collaborative Practice and Patient Outcomes. Washington, DC: The National Academies Press; 2015.
5. Shunk R, Dulay M, Chou C, Janson S, O’Brien BC. Huddle-coaching: a dynamic intervention for trainees and staff to support team-based care. Acad Med. 2014;89(2):244-250.
6. Stock R, Mahoney E, Carney PA. Measuring team development in clinical care settings. Fam Med. 2013;45(10):691-700.
7. PeaceHealth. Team development measure. https://www.peacehealth.org/about-peacehealth/medical-professionals/eugene-springfield-cottage-grove/team-measure/Pages/measure. Accessed August 16, 2018.
8. American Board of Internal Medicine. Teamwork effectiveness assessment module. https://team.abim.org. Accessed August 16, 2018.
9. Chesluk BJ, Bernabeo E, Hess B, Lynn LA, Reddy S, Holmboe ES. A new tool to give hospitalists feedback to improve interprofessional teamwork and advance patient care. Health Aff (Millwood). 2012;31(11):2485-2492.
10. Rodriguez HP, Meredith LS, Hamilton AB, Yano EM, Rubenstein LV. Huddle up!: the adoption and use of structured team communication for VA medical home implementation. Health Care Manage Rev. 2015;40(4):286-299.
1. Rugen KW, Watts SA, Janson SL, et al. Veteran Affairs Centers of Excellence in Primary Care Education: transforming nurse practitioner education. Nurs Outlook. 2014;62(2):78-88.
2. Chang A, Bowen JL, Buranosky RA, et al. Transforming primary care training--patient-centered medical home entrustable professional activities for internal medicine residents. J Gen Intern Med. 2013;28(6):801-809.
3. Zabar S, Adams J, Kurland S, et al. Charting a key competency domain: understanding resident physician interprofessional collaboration (IPC) skills. J Gen Intern Med. 2016;31(8):846-853.
4. Institute of Medicine. Measuring the Impact of Interprofessional Education (IPE) on Collaborative Practice and Patient Outcomes. Washington, DC: The National Academies Press; 2015.
5. Shunk R, Dulay M, Chou C, Janson S, O’Brien BC. Huddle-coaching: a dynamic intervention for trainees and staff to support team-based care. Acad Med. 2014;89(2):244-250.
6. Stock R, Mahoney E, Carney PA. Measuring team development in clinical care settings. Fam Med. 2013;45(10):691-700.
7. PeaceHealth. Team development measure. https://www.peacehealth.org/about-peacehealth/medical-professionals/eugene-springfield-cottage-grove/team-measure/Pages/measure. Accessed August 16, 2018.
8. American Board of Internal Medicine. Teamwork effectiveness assessment module. https://team.abim.org. Accessed August 16, 2018.
9. Chesluk BJ, Bernabeo E, Hess B, Lynn LA, Reddy S, Holmboe ES. A new tool to give hospitalists feedback to improve interprofessional teamwork and advance patient care. Health Aff (Millwood). 2012;31(11):2485-2492.
10. Rodriguez HP, Meredith LS, Hamilton AB, Yano EM, Rubenstein LV. Huddle up!: the adoption and use of structured team communication for VA medical home implementation. Health Care Manage Rev. 2015;40(4):286-299.
How Does Sleep Affect Migraine Risk?
Sleep efficiency, sleep duration, and wake after sleep onset may modify the risk of headache.
BALTIMORE—Multiple dimensions of sleep may temporally precede acute risk of migraine attack among patients with episodic migraine, according to a study presented at the 32nd Annual Meeting of the Associated Professional Sleep Societies. While some aspects of sleep appear to protect against next-day headache, “these novel pilot data support the hypothesis that self-reported fragmented sleep is associated with higher risk of migraine two days later,” said Suzanne M. Bertisch, MD, MPH, and colleagues. Dr. Bertisch is an Assistant Professor of Medicine at Harvard Medical School and a Director of Behavioral Sleep Medicine at Brigham and Women’s Hospital in Boston.
Retrospective studies have indicated that nearly half of patients with migraine identify too little or too much sleep as a migraine trigger. One prospective study reported a higher incidence of headache, both migraine and tension type, after two consecutive nights of four or fewer hours of sleep, as measured by self-report. This study accounted for daily stress but not other potential triggers of migraine. “To date, there have been no prospective studies on the association between objectively assessed sleep parameters and migraine incidence while accounting for other potential triggers of migraine,” Dr. Bertisch said.
A Cohort Study of Migraine Triggers
She and her colleagues assessed the independent contribution of sleep characteristics as temporal precedents of migraine. “We were particularly interested … in sleep duration, fragmentation, and self-reported quality.” To examine these factors, they developed a cohort study of migraine triggers that they conducted from March 2016 to August 2017.
The researchers enrolled 101 adults with episodic migraine from the greater Boston area. Inclusion criteria included at least two migraines per month but fewer than 15 headache days per month, a history of migraine for at least three years, and fulfillment of ICHD-3 criteria for episodic migraine. Exclusion criteria included untreated obstructive sleep apnea, pregnancy, and current opioid use.
Data were collected for six weeks. Study participants were prompted to complete morning and evening diaries that recorded information on sleep, including the pattern, fragmentation, and sleep quality; physical activity and daily mood; medications; and headache characteristics. Patients also wore wrist actigraphs for the duration of the six-week study. Each patient had about 40 days of diary and actigraphy data.
Patients reported the onset and duration of headaches, associated symptoms, whether their pain was a headache or a migraine, maximum pain intensity, and any abortive medications used. Data on daily covariates such as alcohol and caffeine consumption, self-reported physical activity, menstrual cycle, stress, and mood prior to bedtime also were collected.
Dr. Bertisch and colleagues used self-matched case–crossover analyses. “Each person served as [his or her] own control. This approach accounts for time-invariant confounders, including sex, genetics, and usual migraine frequency. We used a conditional logistic regression model that was self-matched by day of the week, because there might be an influence of weekend versus weekday sleep patterns, as well as migraine, and we adjusted for time-dependent covariates, including daily alcohol and caffeine use.”
High WASO Protected Against Next-Day Headache
The 98 participants included in the analyses reflected the known migraine prevalence. “They were generally younger women with an average age of 35, and generally a healthy population,” Dr. Bertisch said. “About one-third reported a history of migraine with aura, and about one-quarter used daily medications to prevent migraines. About 60% reported that sleeping too little triggered their migraines.”
The researchers collected data by actigraphy and diary during approximately 4,500 nights and found that their cohort’s sleep was relatively healthy. “They slept over seven hours per night, they had few sleep problems, they had high sleep efficiency, and they had modest alcohol or caffeine consumption during the study,” Dr. Bertisch said.
In an analysis of diary data, there was no association between odds of a next-day headache and sleep duration, wake after sleep onset (WASO), sleep efficiency, or sleep quality. “However, when we looked at the actigraphy measures, we did find associations with lower sleep efficiency and higher WASO, each associated with lower risk of next-day headache,” Dr. Bertisch said.
“When we looked at odds of headache two days later, we did find that a low sleep efficiency based on self-report was associated with a higher risk of migraine. We also noted a similar trend with self-reported short sleep duration. For actigraphy data, we found a somewhat similar pattern. We also found that sleep duration greater than 8.5 hours was associated with lower
In summary, sleep efficiency and high WASO may be associated with lower odds of next-day headache. For sleep two nights before the headache, low sleep efficiency was associated with higher odds of headache. Long sleep duration, as assessed by actigraphy, was associated with lower odds of headache two days later.
—Glenn S. Williams
Sleep efficiency, sleep duration, and wake after sleep onset may modify the risk of headache.
Sleep efficiency, sleep duration, and wake after sleep onset may modify the risk of headache.
BALTIMORE—Multiple dimensions of sleep may temporally precede acute risk of migraine attack among patients with episodic migraine, according to a study presented at the 32nd Annual Meeting of the Associated Professional Sleep Societies. While some aspects of sleep appear to protect against next-day headache, “these novel pilot data support the hypothesis that self-reported fragmented sleep is associated with higher risk of migraine two days later,” said Suzanne M. Bertisch, MD, MPH, and colleagues. Dr. Bertisch is an Assistant Professor of Medicine at Harvard Medical School and a Director of Behavioral Sleep Medicine at Brigham and Women’s Hospital in Boston.
Retrospective studies have indicated that nearly half of patients with migraine identify too little or too much sleep as a migraine trigger. One prospective study reported a higher incidence of headache, both migraine and tension type, after two consecutive nights of four or fewer hours of sleep, as measured by self-report. This study accounted for daily stress but not other potential triggers of migraine. “To date, there have been no prospective studies on the association between objectively assessed sleep parameters and migraine incidence while accounting for other potential triggers of migraine,” Dr. Bertisch said.
A Cohort Study of Migraine Triggers
She and her colleagues assessed the independent contribution of sleep characteristics as temporal precedents of migraine. “We were particularly interested … in sleep duration, fragmentation, and self-reported quality.” To examine these factors, they developed a cohort study of migraine triggers that they conducted from March 2016 to August 2017.
The researchers enrolled 101 adults with episodic migraine from the greater Boston area. Inclusion criteria included at least two migraines per month but fewer than 15 headache days per month, a history of migraine for at least three years, and fulfillment of ICHD-3 criteria for episodic migraine. Exclusion criteria included untreated obstructive sleep apnea, pregnancy, and current opioid use.
Data were collected for six weeks. Study participants were prompted to complete morning and evening diaries that recorded information on sleep, including the pattern, fragmentation, and sleep quality; physical activity and daily mood; medications; and headache characteristics. Patients also wore wrist actigraphs for the duration of the six-week study. Each patient had about 40 days of diary and actigraphy data.
Patients reported the onset and duration of headaches, associated symptoms, whether their pain was a headache or a migraine, maximum pain intensity, and any abortive medications used. Data on daily covariates such as alcohol and caffeine consumption, self-reported physical activity, menstrual cycle, stress, and mood prior to bedtime also were collected.
Dr. Bertisch and colleagues used self-matched case–crossover analyses. “Each person served as [his or her] own control. This approach accounts for time-invariant confounders, including sex, genetics, and usual migraine frequency. We used a conditional logistic regression model that was self-matched by day of the week, because there might be an influence of weekend versus weekday sleep patterns, as well as migraine, and we adjusted for time-dependent covariates, including daily alcohol and caffeine use.”
High WASO Protected Against Next-Day Headache
The 98 participants included in the analyses reflected the known migraine prevalence. “They were generally younger women with an average age of 35, and generally a healthy population,” Dr. Bertisch said. “About one-third reported a history of migraine with aura, and about one-quarter used daily medications to prevent migraines. About 60% reported that sleeping too little triggered their migraines.”
The researchers collected data by actigraphy and diary during approximately 4,500 nights and found that their cohort’s sleep was relatively healthy. “They slept over seven hours per night, they had few sleep problems, they had high sleep efficiency, and they had modest alcohol or caffeine consumption during the study,” Dr. Bertisch said.
In an analysis of diary data, there was no association between odds of a next-day headache and sleep duration, wake after sleep onset (WASO), sleep efficiency, or sleep quality. “However, when we looked at the actigraphy measures, we did find associations with lower sleep efficiency and higher WASO, each associated with lower risk of next-day headache,” Dr. Bertisch said.
“When we looked at odds of headache two days later, we did find that a low sleep efficiency based on self-report was associated with a higher risk of migraine. We also noted a similar trend with self-reported short sleep duration. For actigraphy data, we found a somewhat similar pattern. We also found that sleep duration greater than 8.5 hours was associated with lower
In summary, sleep efficiency and high WASO may be associated with lower odds of next-day headache. For sleep two nights before the headache, low sleep efficiency was associated with higher odds of headache. Long sleep duration, as assessed by actigraphy, was associated with lower odds of headache two days later.
—Glenn S. Williams
BALTIMORE—Multiple dimensions of sleep may temporally precede acute risk of migraine attack among patients with episodic migraine, according to a study presented at the 32nd Annual Meeting of the Associated Professional Sleep Societies. While some aspects of sleep appear to protect against next-day headache, “these novel pilot data support the hypothesis that self-reported fragmented sleep is associated with higher risk of migraine two days later,” said Suzanne M. Bertisch, MD, MPH, and colleagues. Dr. Bertisch is an Assistant Professor of Medicine at Harvard Medical School and a Director of Behavioral Sleep Medicine at Brigham and Women’s Hospital in Boston.
Retrospective studies have indicated that nearly half of patients with migraine identify too little or too much sleep as a migraine trigger. One prospective study reported a higher incidence of headache, both migraine and tension type, after two consecutive nights of four or fewer hours of sleep, as measured by self-report. This study accounted for daily stress but not other potential triggers of migraine. “To date, there have been no prospective studies on the association between objectively assessed sleep parameters and migraine incidence while accounting for other potential triggers of migraine,” Dr. Bertisch said.
A Cohort Study of Migraine Triggers
She and her colleagues assessed the independent contribution of sleep characteristics as temporal precedents of migraine. “We were particularly interested … in sleep duration, fragmentation, and self-reported quality.” To examine these factors, they developed a cohort study of migraine triggers that they conducted from March 2016 to August 2017.
The researchers enrolled 101 adults with episodic migraine from the greater Boston area. Inclusion criteria included at least two migraines per month but fewer than 15 headache days per month, a history of migraine for at least three years, and fulfillment of ICHD-3 criteria for episodic migraine. Exclusion criteria included untreated obstructive sleep apnea, pregnancy, and current opioid use.
Data were collected for six weeks. Study participants were prompted to complete morning and evening diaries that recorded information on sleep, including the pattern, fragmentation, and sleep quality; physical activity and daily mood; medications; and headache characteristics. Patients also wore wrist actigraphs for the duration of the six-week study. Each patient had about 40 days of diary and actigraphy data.
Patients reported the onset and duration of headaches, associated symptoms, whether their pain was a headache or a migraine, maximum pain intensity, and any abortive medications used. Data on daily covariates such as alcohol and caffeine consumption, self-reported physical activity, menstrual cycle, stress, and mood prior to bedtime also were collected.
Dr. Bertisch and colleagues used self-matched case–crossover analyses. “Each person served as [his or her] own control. This approach accounts for time-invariant confounders, including sex, genetics, and usual migraine frequency. We used a conditional logistic regression model that was self-matched by day of the week, because there might be an influence of weekend versus weekday sleep patterns, as well as migraine, and we adjusted for time-dependent covariates, including daily alcohol and caffeine use.”
High WASO Protected Against Next-Day Headache
The 98 participants included in the analyses reflected the known migraine prevalence. “They were generally younger women with an average age of 35, and generally a healthy population,” Dr. Bertisch said. “About one-third reported a history of migraine with aura, and about one-quarter used daily medications to prevent migraines. About 60% reported that sleeping too little triggered their migraines.”
The researchers collected data by actigraphy and diary during approximately 4,500 nights and found that their cohort’s sleep was relatively healthy. “They slept over seven hours per night, they had few sleep problems, they had high sleep efficiency, and they had modest alcohol or caffeine consumption during the study,” Dr. Bertisch said.
In an analysis of diary data, there was no association between odds of a next-day headache and sleep duration, wake after sleep onset (WASO), sleep efficiency, or sleep quality. “However, when we looked at the actigraphy measures, we did find associations with lower sleep efficiency and higher WASO, each associated with lower risk of next-day headache,” Dr. Bertisch said.
“When we looked at odds of headache two days later, we did find that a low sleep efficiency based on self-report was associated with a higher risk of migraine. We also noted a similar trend with self-reported short sleep duration. For actigraphy data, we found a somewhat similar pattern. We also found that sleep duration greater than 8.5 hours was associated with lower
In summary, sleep efficiency and high WASO may be associated with lower odds of next-day headache. For sleep two nights before the headache, low sleep efficiency was associated with higher odds of headache. Long sleep duration, as assessed by actigraphy, was associated with lower odds of headache two days later.
—Glenn S. Williams
Bedside Microscopy for the Beginner
Dermatologists are uniquely equipped amongst clinicians to make bedside diagnoses because of the focus on histopathology and microscopy inherent in our training. This skill is highly valuable in both an inpatient and outpatient setting because it may lead to a rapid diagnosis or be a useful adjunct in the initial clinical decision-making process. Although expert microscopists may be able to garner relevant information from scraping almost any type of lesion, bedside microscopy primarily is used by dermatologists in the United States for consideration of infectious etiologies of a variety of cutaneous manifestations.1,2
Basic Principles
Lesions that should be considered for bedside microscopic analysis in outpatient settings are scaly lesions, vesiculobullous lesions, inflammatory papules, and pustules1; microscopic evaluation also can be useful for myriad trichoscopic considerations.3,4 In some instances, direct visualization of the pathogen is possible (eg, cutaneous fungal infections, demodicidosis, scabetic infections), and in other circumstances reactive changes of keratinocytes or the presence of specific cell types can aid in diagnosis (eg, ballooning degeneration and multinucleation of keratinocytes in herpetic lesions, an abundance of eosinophils in erythema toxicum neonatorum). Different types of media are used to best prepare tissue based on the suspected etiology of the condition.
One major stumbling block for residents when beginning to perform bedside testing is the lack of dimensional understanding of the structures they are searching for; for example, medical students and residents often may mistake fibers for dermatophytes, which typically are much larger than fungal hyphae. Familiarizing oneself with the basic dimensions of different cell types or pathogens in relation to each other (Table) will help further refine the beginner’s ability to effectively search for and identify pathogenic features. This concept is further schematized in Figure 1 to help visualize scale differences.
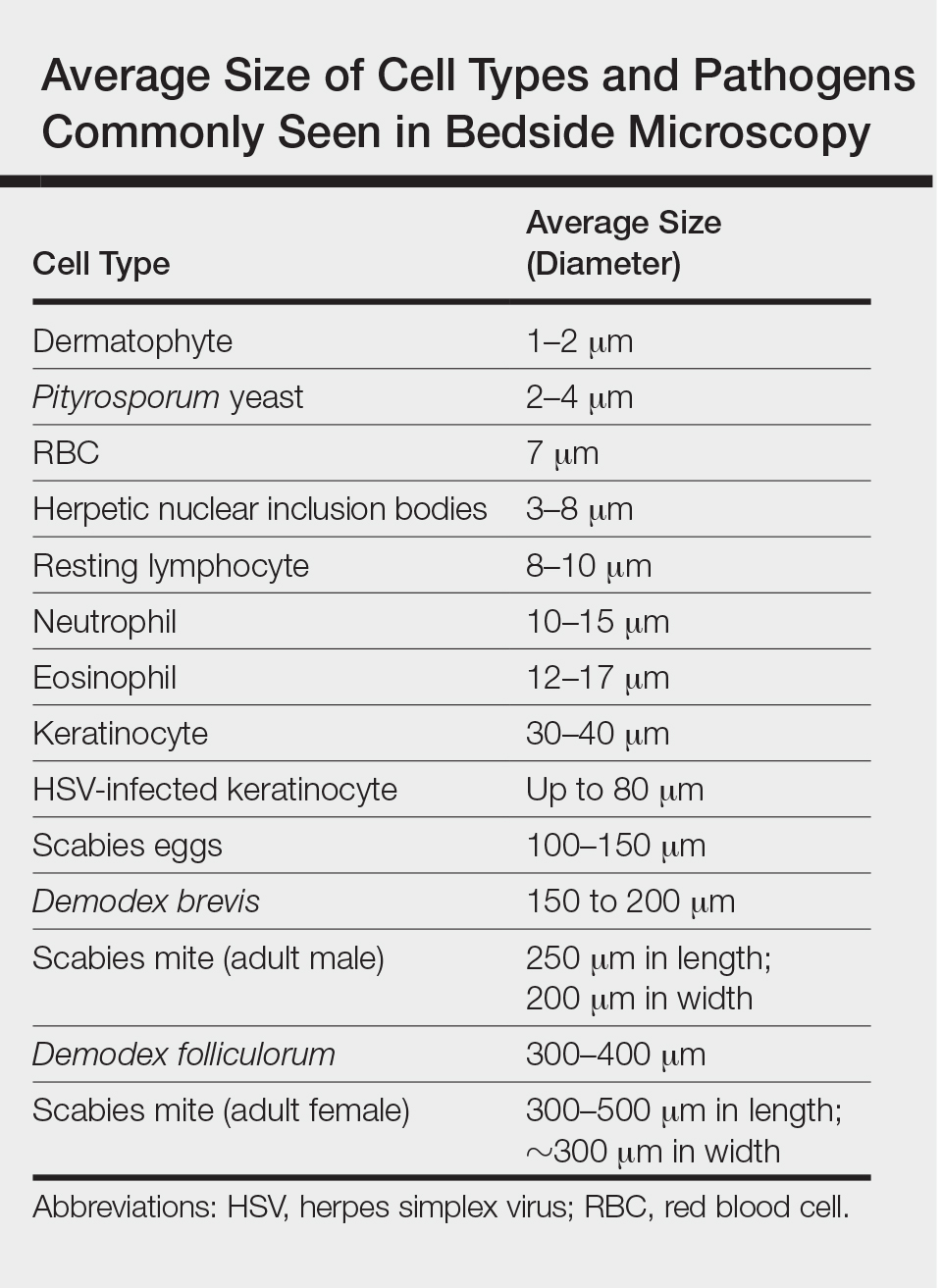
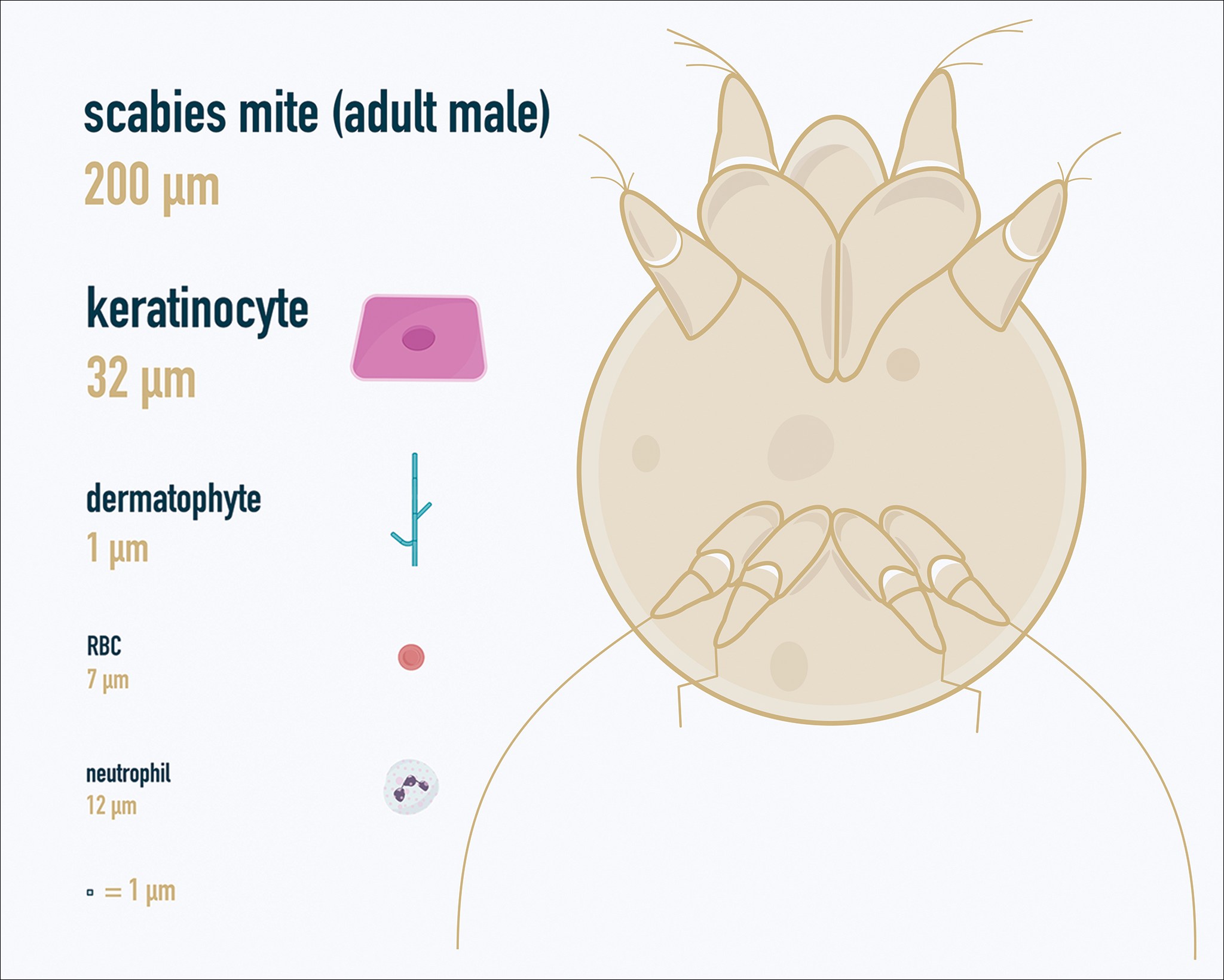
Examination of the Specimen
Slide preparation depends on the primary lesion in consideration and will be discussed in greater detail in the following sections. Once the slide is prepared, place it on the microscope stage and adjust the condenser and light source for optimal visualization. Scan the specimen in a gridlike fashion on low power (usually ×10) and then inspect suspicious findings on higher power (×40 or higher).
Dermatomycoses
Fungal infections of the skin can present as annular papulosquamous lesions, follicular pustules or papules, bullous lesions, hypopigmented patches, and mucosal exudate or erosions, among other manifestations.5 Potassium hydroxide (KOH) is the classic medium used in preparation of lesions being assessed for evidence of fungus because it leads to lysis of keratinocytes for better visualization of fungal hyphae and spores. Other media that contain KOH and additional substrates such as dimethyl sulfoxide or chlorazol black E can be used to better highlight fungal elements.6
Dermatophytosis
Dermatophytes lead to superficial infection of the epidermis and epidermal appendages and present in a variety of ways, including site-specific infections manifesting typically as erythematous, annular or arcuate scaling (eg, tinea faciei, tinea corporis, tinea cruris, tinea manus, tinea pedis), alopecia with broken hair shafts, black dots, boggy nodules and/or scaling of the scalp (eg, tinea capitis, favus, kerion), and dystrophic nails (eg, onychomycosis).5,7 For examination of lesional skin scrapings, one can either use clear cellophane tape against the skin to remove scale, which is especially useful in the case of pediatric patients, and then press the tape against a slide prepared with several drops of a KOH-based medium to directly visualize without a coverslip, or scrape the lesion with a No. 15 blade and place the scales onto the glass slide, with further preparation as described below.8 For assessment of alopecia or dystrophic nails, scrape lesional skin with a No. 15 blade to obtain affected hair follicles and proximal subungual debris, respectively.6,9
Once the cellular debris has been obtained and placed on the slide, a coverslip can be overlaid and KOH applied laterally to be taken up across the slide by capillary action. Allow the slide to sit for at least 5 minutes before analyzing to better visualize fungal elements. Both tinea and onychomycosis will show branching septate hyphae extending across keratinocytes; a common false-positive is identifying overlapping keratinocyte edges, which are a similar size, but they can be distinguished from fungi because they do not cross multiple keratinocytes.1,8 Tinea capitis may demonstrate similar findings or may reveal hair shafts with spores contained within or surrounding it, corresponding to endothrix or ectothrix infection, respectively.5
Pityriasis Versicolor and Malassezia Folliculitis
Pityriasis versicolor presents with hypopigmented to pink, finely scaling ovoid papules, usually on the upper back, shoulders, and neck, and is caused by Malassezia furfur and other Malassezia species.5 Malassezia folliculitis also is caused by this fungus and presents with monomorphic follicular papules and pustules. Scrapings from the scaly papules will demonstrate keratinocytes with the classic “spaghetti and meatballs” fungal elements, whereas Malassezia folliculitis demonstrates only spores.5,7
Candidiasis
One possible outpatient presentation of candidiasis is oral thrush, which can exhibit white mucosal exudate or erythematous patches. A tongue blade can be used to scrape the tongue or cheek wall, with subsequent preparatory steps with application of KOH as described for dermatophytes. Cutaneous candidiasis most often develops in intertriginous regions and will exhibit erosive painful lesions with satellite pustules. In both cases, analysis of the specimen will show shorter fatter hyphal elements than seen in dermatophytosis, with pseudohyphae, blunted ends, and potentially yeast forms.5
Vesiculobullous Lesions
The Tzanck smear has been used since the 1940s to differentiate between etiologies of blistering disorders and is now most commonly used for the quick identification of herpetic lesions.1 The test is performed by scraping the base of a deroofed vesicle, pustule, or bulla, and smearing the cellular materials onto a glass slide. The most commonly utilized media for staining in the outpatient setting at my institution (University of Texas Dell Medical School, Austin) is Giemsa, which is composed of azure II–eosin, glycerin, and methanol. It stains nuclei a reddish blue to pink and the cytoplasm blue.10 After being applied to the slide, the cells are allowed to air-dry for 5 to 10 minutes, and Giemsa stain is subsequently applied and allowed to incubate for 15 minutes, then rinsed carefully with water and directly examined.
Other stains that can be used to perform the Tzanck smear include commercial preparations that may be more accessible in the inpatient settings such as the Wright-Giemsa, Quik-Dip, and Diff-Quick.1,10
Examination of a Tzanck smear from a herpetic lesion will yield acantholytic, enlarged keratinocytes up to twice their usual size (referred to as ballooning degeneration), and multinucleation. In addition, molding of the nuclei to each other within the multinucleated cells and margination of the nuclear chromatin may be appreciated (Figure 2). Intranuclear inclusion bodies, also known as Cowdry type A bodies, can be seen that are nearly the size of red blood cells but are rare to find, with only 10% of specimens exhibiting this finding in a prospective review of 299 patients with herpetic vesiculobullous lesions.11 Evaluation of the contents of blisters caused by bullous pemphigoid and erythema toxicum neonatorum may yield high densities of eosinophils with normal keratinocyte morphology (Figure 3). Other blistering eruptions such as pemphigus vulgaris and bullous drug eruptions also have characteristic findings.1,2
Gout Preparation
Gout is a systemic disease caused by uric acid accumulation that can present with joint pain and white to red nodules on digits, joints, and ears (known as tophi). Material may be expressed from tophi and examined immediately by polarized light microscopy to confirm the diagnosis.5 Specimens will demonstrate needle-shaped, negatively birefringent monosodium urate crystals on polarized light microscopy (Figure 4). An ordinary light microscope can be converted for such use with the lenses of inexpensive polarized sunglasses, placing one lens between the light source and specimen and the other lens between the examiner’s eye and the specimen.12
Parasitic Infections
Two common parasitic infections identified in outpatient dermatology clinics are scabies mites and Demodex mites. Human scabies is extremely pruritic and caused by infestation with Sarcoptes scabiei var hominis; the typical presentation in an adult is erythematous and crusted papules, linear burrows, and vesiculopustules, especially of the interdigital spaces, wrists, axillae, umbilicus, and genital region.1,13 Demodicidosis presents with papules and pustules on the face, usually in a patient with background rosacea and diffuse erythema.1,5,14
If either of these conditions are suspected, mineral oil should be used to prepare the slide because it will maintain viability of the organisms, which are visualized better in motion. Adult scabies mites are roughly 10 times larger than keratinocytes, measuring approximately 250 to 450 µm in length with 8 legs.13 Eggs also may be visualized within the cellular debris and typically are 100 to 150 µm in size and ovoid in shape. Of note, polariscopic examination may be a useful adjunct for evaluation of scabies because scabetic spines and scybala (or fecal material) are polarizable.15
Two types of Demodex mites typically are found in the skin: Demodex folliculorum, which are similarly sized to scabies mites with a more oblong body and occur most commonly in mature hair follicles (eg, eyelashes), and Demodex brevis, which are about half the size (150–200 µm) and live in the sebaceous glands of vellus hairs (Figure 5).14 Both of these mites have 8 legs, similar to the scabies mite.
Hair Preparations
Hair preparations for bulbar examination (eg, trichogram) may prove useful in the evaluation of many types of alopecia, and elaboration on this topic is beyond the scope of this article. Microscopic evaluation of the hair shaft may be an underutilized technique in the outpatient setting and is capable of yielding a variety of diagnoses, including monilethrix, pili torti, and pili trianguli et canaliculi, among others.3 One particularly useful scenario for hair shaft examination (usually of the eyebrow) is in the setting of a patient with severe atopic dermatitis or a baby with ichthyosiform erythroderma, as discovery of trichorrhexis invaginata is pathognomonic for the diagnosis of Netherton syndrome.16 Lastly, evaluation of the hair shaft in patients with patchy and diffuse hair loss whose clinical impression is reminiscent of alopecia areata, or those with concerns of inability to grow hair beyond a short length, may lead to diagnosis of loose anagen syndrome, especially if more than 70% of hair fibers examined exhibit the classic findings of a ruffled proximal cuticle and lack of root sheath.4
Final Thoughts
Bedside microscopy is a rapid and cost-sensitive way to confirm diagnoses that are clinically suspected and remains a valuable tool to acquire during residency training.
- Wanat KA, Dominguez AR, Carter Z, et al. Bedside diagnostics in dermatology: viral, bacterial, and fungal infections. J Am Acad Dermatol. 2017;77:197-218.
- Micheletti RG, Dominguez AR, Wanat KA. Bedside diagnostics in dermatology: parasitic and noninfectious diseases. J Am Acad Dermatol. 2017;77:221-230.
- Whiting DA, Dy LC. Office diagnosis of hair shaft defects. Semin Cutan Med Surg. 2006;25:24-34.
- Tosti A. Loose anagen hair syndrome and loose anagen hair. Arch Dermatol. 2002;138:521-522.
- Bolognia JL, Schaffer JV, Cerroni L, eds. Dermatology. 4th ed. Philadelphia PA: Elsevier; 2017.
- Lilly KK, Koshnick RL, Grill JP, et al. Cost-effectiveness of diagnostic tests for toenail onychomycosis: a repeated-measure, single-blinded, cross-sectional evaluation of 7 diagnostic tests. J Am Acad Dermatol. 2006;55:620-626.
- Elder DE, ed. Lever’s Histopathology of the Skin. 10th ed. Philadelphia, PA: Wolters Kluwer Health/Lippincott Williams & Wilkins; 2009.
- Raghukumar S, Ravikumar BC. Potassium hydroxide mount with cellophane adhesive: a method for direct diagnosis of dermatophyte skin infections [published online May 29, 2018]. Clin Exp Dermatol. doi:10.1111/ced.13573.
- Bhat YJ, Zeerak S, Kanth F, et al. Clinicoepidemiological and mycological study of tinea capitis in the pediatric population of Kashmir Valley: a study from a tertiary care centre. Indian Dermatol Online J. 2017;8:100-103.
- Gupta LK, Singhi MK. Tzanck smear: a useful diagnostic tool. Indian J Dermatol Venereol Leprol. 2005;71:295-299.
- Durdu M, Baba M, Seçkin D. The value of Tzanck smear test in diagnosis of erosive, vesicular, bullous, and pustular skin lesions. J Am Acad Dermatol. 2008;59:958-964.
- Fagan TJ, Lidsky MD. Compensated polarized light microscopy using cellophane adhesive tape. Arthritis Rheum. 1974;17:256-262.
- Walton SF, Currie BJ. Problems in diagnosing scabies, a global disease in human and animal populations. Clin Microbiol Rev. 2007;20:268-279.
- Desch C, Nutting WB. Demodex folliculorum (Simon) and D. brevis akbulatova of man: redescription and reevaluation. J Parasitol. 1972;58:169-177.
- Foo CW, Florell SR, Bowen AR. Polarizable elements in scabies infestation: a clue to diagnosis. J Cutan Pathol. 2013;40:6-10.
- Akkurt ZM, Tuncel T, Ayhan E, et al. Rapid and easy diagnosis of Netherton syndrome with dermoscopy. J Cutan Med Surg. 2014;18:280-282.
Dermatologists are uniquely equipped amongst clinicians to make bedside diagnoses because of the focus on histopathology and microscopy inherent in our training. This skill is highly valuable in both an inpatient and outpatient setting because it may lead to a rapid diagnosis or be a useful adjunct in the initial clinical decision-making process. Although expert microscopists may be able to garner relevant information from scraping almost any type of lesion, bedside microscopy primarily is used by dermatologists in the United States for consideration of infectious etiologies of a variety of cutaneous manifestations.1,2
Basic Principles
Lesions that should be considered for bedside microscopic analysis in outpatient settings are scaly lesions, vesiculobullous lesions, inflammatory papules, and pustules1; microscopic evaluation also can be useful for myriad trichoscopic considerations.3,4 In some instances, direct visualization of the pathogen is possible (eg, cutaneous fungal infections, demodicidosis, scabetic infections), and in other circumstances reactive changes of keratinocytes or the presence of specific cell types can aid in diagnosis (eg, ballooning degeneration and multinucleation of keratinocytes in herpetic lesions, an abundance of eosinophils in erythema toxicum neonatorum). Different types of media are used to best prepare tissue based on the suspected etiology of the condition.
One major stumbling block for residents when beginning to perform bedside testing is the lack of dimensional understanding of the structures they are searching for; for example, medical students and residents often may mistake fibers for dermatophytes, which typically are much larger than fungal hyphae. Familiarizing oneself with the basic dimensions of different cell types or pathogens in relation to each other (Table) will help further refine the beginner’s ability to effectively search for and identify pathogenic features. This concept is further schematized in Figure 1 to help visualize scale differences.


Examination of the Specimen
Slide preparation depends on the primary lesion in consideration and will be discussed in greater detail in the following sections. Once the slide is prepared, place it on the microscope stage and adjust the condenser and light source for optimal visualization. Scan the specimen in a gridlike fashion on low power (usually ×10) and then inspect suspicious findings on higher power (×40 or higher).
Dermatomycoses
Fungal infections of the skin can present as annular papulosquamous lesions, follicular pustules or papules, bullous lesions, hypopigmented patches, and mucosal exudate or erosions, among other manifestations.5 Potassium hydroxide (KOH) is the classic medium used in preparation of lesions being assessed for evidence of fungus because it leads to lysis of keratinocytes for better visualization of fungal hyphae and spores. Other media that contain KOH and additional substrates such as dimethyl sulfoxide or chlorazol black E can be used to better highlight fungal elements.6
Dermatophytosis
Dermatophytes lead to superficial infection of the epidermis and epidermal appendages and present in a variety of ways, including site-specific infections manifesting typically as erythematous, annular or arcuate scaling (eg, tinea faciei, tinea corporis, tinea cruris, tinea manus, tinea pedis), alopecia with broken hair shafts, black dots, boggy nodules and/or scaling of the scalp (eg, tinea capitis, favus, kerion), and dystrophic nails (eg, onychomycosis).5,7 For examination of lesional skin scrapings, one can either use clear cellophane tape against the skin to remove scale, which is especially useful in the case of pediatric patients, and then press the tape against a slide prepared with several drops of a KOH-based medium to directly visualize without a coverslip, or scrape the lesion with a No. 15 blade and place the scales onto the glass slide, with further preparation as described below.8 For assessment of alopecia or dystrophic nails, scrape lesional skin with a No. 15 blade to obtain affected hair follicles and proximal subungual debris, respectively.6,9
Once the cellular debris has been obtained and placed on the slide, a coverslip can be overlaid and KOH applied laterally to be taken up across the slide by capillary action. Allow the slide to sit for at least 5 minutes before analyzing to better visualize fungal elements. Both tinea and onychomycosis will show branching septate hyphae extending across keratinocytes; a common false-positive is identifying overlapping keratinocyte edges, which are a similar size, but they can be distinguished from fungi because they do not cross multiple keratinocytes.1,8 Tinea capitis may demonstrate similar findings or may reveal hair shafts with spores contained within or surrounding it, corresponding to endothrix or ectothrix infection, respectively.5
Pityriasis Versicolor and Malassezia Folliculitis
Pityriasis versicolor presents with hypopigmented to pink, finely scaling ovoid papules, usually on the upper back, shoulders, and neck, and is caused by Malassezia furfur and other Malassezia species.5 Malassezia folliculitis also is caused by this fungus and presents with monomorphic follicular papules and pustules. Scrapings from the scaly papules will demonstrate keratinocytes with the classic “spaghetti and meatballs” fungal elements, whereas Malassezia folliculitis demonstrates only spores.5,7
Candidiasis
One possible outpatient presentation of candidiasis is oral thrush, which can exhibit white mucosal exudate or erythematous patches. A tongue blade can be used to scrape the tongue or cheek wall, with subsequent preparatory steps with application of KOH as described for dermatophytes. Cutaneous candidiasis most often develops in intertriginous regions and will exhibit erosive painful lesions with satellite pustules. In both cases, analysis of the specimen will show shorter fatter hyphal elements than seen in dermatophytosis, with pseudohyphae, blunted ends, and potentially yeast forms.5
Vesiculobullous Lesions
The Tzanck smear has been used since the 1940s to differentiate between etiologies of blistering disorders and is now most commonly used for the quick identification of herpetic lesions.1 The test is performed by scraping the base of a deroofed vesicle, pustule, or bulla, and smearing the cellular materials onto a glass slide. The most commonly utilized media for staining in the outpatient setting at my institution (University of Texas Dell Medical School, Austin) is Giemsa, which is composed of azure II–eosin, glycerin, and methanol. It stains nuclei a reddish blue to pink and the cytoplasm blue.10 After being applied to the slide, the cells are allowed to air-dry for 5 to 10 minutes, and Giemsa stain is subsequently applied and allowed to incubate for 15 minutes, then rinsed carefully with water and directly examined.
Other stains that can be used to perform the Tzanck smear include commercial preparations that may be more accessible in the inpatient settings such as the Wright-Giemsa, Quik-Dip, and Diff-Quick.1,10
Examination of a Tzanck smear from a herpetic lesion will yield acantholytic, enlarged keratinocytes up to twice their usual size (referred to as ballooning degeneration), and multinucleation. In addition, molding of the nuclei to each other within the multinucleated cells and margination of the nuclear chromatin may be appreciated (Figure 2). Intranuclear inclusion bodies, also known as Cowdry type A bodies, can be seen that are nearly the size of red blood cells but are rare to find, with only 10% of specimens exhibiting this finding in a prospective review of 299 patients with herpetic vesiculobullous lesions.11 Evaluation of the contents of blisters caused by bullous pemphigoid and erythema toxicum neonatorum may yield high densities of eosinophils with normal keratinocyte morphology (Figure 3). Other blistering eruptions such as pemphigus vulgaris and bullous drug eruptions also have characteristic findings.1,2


Gout Preparation
Gout is a systemic disease caused by uric acid accumulation that can present with joint pain and white to red nodules on digits, joints, and ears (known as tophi). Material may be expressed from tophi and examined immediately by polarized light microscopy to confirm the diagnosis.5 Specimens will demonstrate needle-shaped, negatively birefringent monosodium urate crystals on polarized light microscopy (Figure 4). An ordinary light microscope can be converted for such use with the lenses of inexpensive polarized sunglasses, placing one lens between the light source and specimen and the other lens between the examiner’s eye and the specimen.12

Parasitic Infections
Two common parasitic infections identified in outpatient dermatology clinics are scabies mites and Demodex mites. Human scabies is extremely pruritic and caused by infestation with Sarcoptes scabiei var hominis; the typical presentation in an adult is erythematous and crusted papules, linear burrows, and vesiculopustules, especially of the interdigital spaces, wrists, axillae, umbilicus, and genital region.1,13 Demodicidosis presents with papules and pustules on the face, usually in a patient with background rosacea and diffuse erythema.1,5,14
If either of these conditions are suspected, mineral oil should be used to prepare the slide because it will maintain viability of the organisms, which are visualized better in motion. Adult scabies mites are roughly 10 times larger than keratinocytes, measuring approximately 250 to 450 µm in length with 8 legs.13 Eggs also may be visualized within the cellular debris and typically are 100 to 150 µm in size and ovoid in shape. Of note, polariscopic examination may be a useful adjunct for evaluation of scabies because scabetic spines and scybala (or fecal material) are polarizable.15
Two types of Demodex mites typically are found in the skin: Demodex folliculorum, which are similarly sized to scabies mites with a more oblong body and occur most commonly in mature hair follicles (eg, eyelashes), and Demodex brevis, which are about half the size (150–200 µm) and live in the sebaceous glands of vellus hairs (Figure 5).14 Both of these mites have 8 legs, similar to the scabies mite.

Hair Preparations
Hair preparations for bulbar examination (eg, trichogram) may prove useful in the evaluation of many types of alopecia, and elaboration on this topic is beyond the scope of this article. Microscopic evaluation of the hair shaft may be an underutilized technique in the outpatient setting and is capable of yielding a variety of diagnoses, including monilethrix, pili torti, and pili trianguli et canaliculi, among others.3 One particularly useful scenario for hair shaft examination (usually of the eyebrow) is in the setting of a patient with severe atopic dermatitis or a baby with ichthyosiform erythroderma, as discovery of trichorrhexis invaginata is pathognomonic for the diagnosis of Netherton syndrome.16 Lastly, evaluation of the hair shaft in patients with patchy and diffuse hair loss whose clinical impression is reminiscent of alopecia areata, or those with concerns of inability to grow hair beyond a short length, may lead to diagnosis of loose anagen syndrome, especially if more than 70% of hair fibers examined exhibit the classic findings of a ruffled proximal cuticle and lack of root sheath.4
Final Thoughts
Bedside microscopy is a rapid and cost-sensitive way to confirm diagnoses that are clinically suspected and remains a valuable tool to acquire during residency training.
Dermatologists are uniquely equipped amongst clinicians to make bedside diagnoses because of the focus on histopathology and microscopy inherent in our training. This skill is highly valuable in both an inpatient and outpatient setting because it may lead to a rapid diagnosis or be a useful adjunct in the initial clinical decision-making process. Although expert microscopists may be able to garner relevant information from scraping almost any type of lesion, bedside microscopy primarily is used by dermatologists in the United States for consideration of infectious etiologies of a variety of cutaneous manifestations.1,2
Basic Principles
Lesions that should be considered for bedside microscopic analysis in outpatient settings are scaly lesions, vesiculobullous lesions, inflammatory papules, and pustules1; microscopic evaluation also can be useful for myriad trichoscopic considerations.3,4 In some instances, direct visualization of the pathogen is possible (eg, cutaneous fungal infections, demodicidosis, scabetic infections), and in other circumstances reactive changes of keratinocytes or the presence of specific cell types can aid in diagnosis (eg, ballooning degeneration and multinucleation of keratinocytes in herpetic lesions, an abundance of eosinophils in erythema toxicum neonatorum). Different types of media are used to best prepare tissue based on the suspected etiology of the condition.
One major stumbling block for residents when beginning to perform bedside testing is the lack of dimensional understanding of the structures they are searching for; for example, medical students and residents often may mistake fibers for dermatophytes, which typically are much larger than fungal hyphae. Familiarizing oneself with the basic dimensions of different cell types or pathogens in relation to each other (Table) will help further refine the beginner’s ability to effectively search for and identify pathogenic features. This concept is further schematized in Figure 1 to help visualize scale differences.


Examination of the Specimen
Slide preparation depends on the primary lesion in consideration and will be discussed in greater detail in the following sections. Once the slide is prepared, place it on the microscope stage and adjust the condenser and light source for optimal visualization. Scan the specimen in a gridlike fashion on low power (usually ×10) and then inspect suspicious findings on higher power (×40 or higher).
Dermatomycoses
Fungal infections of the skin can present as annular papulosquamous lesions, follicular pustules or papules, bullous lesions, hypopigmented patches, and mucosal exudate or erosions, among other manifestations.5 Potassium hydroxide (KOH) is the classic medium used in preparation of lesions being assessed for evidence of fungus because it leads to lysis of keratinocytes for better visualization of fungal hyphae and spores. Other media that contain KOH and additional substrates such as dimethyl sulfoxide or chlorazol black E can be used to better highlight fungal elements.6
Dermatophytosis
Dermatophytes lead to superficial infection of the epidermis and epidermal appendages and present in a variety of ways, including site-specific infections manifesting typically as erythematous, annular or arcuate scaling (eg, tinea faciei, tinea corporis, tinea cruris, tinea manus, tinea pedis), alopecia with broken hair shafts, black dots, boggy nodules and/or scaling of the scalp (eg, tinea capitis, favus, kerion), and dystrophic nails (eg, onychomycosis).5,7 For examination of lesional skin scrapings, one can either use clear cellophane tape against the skin to remove scale, which is especially useful in the case of pediatric patients, and then press the tape against a slide prepared with several drops of a KOH-based medium to directly visualize without a coverslip, or scrape the lesion with a No. 15 blade and place the scales onto the glass slide, with further preparation as described below.8 For assessment of alopecia or dystrophic nails, scrape lesional skin with a No. 15 blade to obtain affected hair follicles and proximal subungual debris, respectively.6,9
Once the cellular debris has been obtained and placed on the slide, a coverslip can be overlaid and KOH applied laterally to be taken up across the slide by capillary action. Allow the slide to sit for at least 5 minutes before analyzing to better visualize fungal elements. Both tinea and onychomycosis will show branching septate hyphae extending across keratinocytes; a common false-positive is identifying overlapping keratinocyte edges, which are a similar size, but they can be distinguished from fungi because they do not cross multiple keratinocytes.1,8 Tinea capitis may demonstrate similar findings or may reveal hair shafts with spores contained within or surrounding it, corresponding to endothrix or ectothrix infection, respectively.5
Pityriasis Versicolor and Malassezia Folliculitis
Pityriasis versicolor presents with hypopigmented to pink, finely scaling ovoid papules, usually on the upper back, shoulders, and neck, and is caused by Malassezia furfur and other Malassezia species.5 Malassezia folliculitis also is caused by this fungus and presents with monomorphic follicular papules and pustules. Scrapings from the scaly papules will demonstrate keratinocytes with the classic “spaghetti and meatballs” fungal elements, whereas Malassezia folliculitis demonstrates only spores.5,7
Candidiasis
One possible outpatient presentation of candidiasis is oral thrush, which can exhibit white mucosal exudate or erythematous patches. A tongue blade can be used to scrape the tongue or cheek wall, with subsequent preparatory steps with application of KOH as described for dermatophytes. Cutaneous candidiasis most often develops in intertriginous regions and will exhibit erosive painful lesions with satellite pustules. In both cases, analysis of the specimen will show shorter fatter hyphal elements than seen in dermatophytosis, with pseudohyphae, blunted ends, and potentially yeast forms.5
Vesiculobullous Lesions
The Tzanck smear has been used since the 1940s to differentiate between etiologies of blistering disorders and is now most commonly used for the quick identification of herpetic lesions.1 The test is performed by scraping the base of a deroofed vesicle, pustule, or bulla, and smearing the cellular materials onto a glass slide. The most commonly utilized media for staining in the outpatient setting at my institution (University of Texas Dell Medical School, Austin) is Giemsa, which is composed of azure II–eosin, glycerin, and methanol. It stains nuclei a reddish blue to pink and the cytoplasm blue.10 After being applied to the slide, the cells are allowed to air-dry for 5 to 10 minutes, and Giemsa stain is subsequently applied and allowed to incubate for 15 minutes, then rinsed carefully with water and directly examined.
Other stains that can be used to perform the Tzanck smear include commercial preparations that may be more accessible in the inpatient settings such as the Wright-Giemsa, Quik-Dip, and Diff-Quick.1,10
Examination of a Tzanck smear from a herpetic lesion will yield acantholytic, enlarged keratinocytes up to twice their usual size (referred to as ballooning degeneration), and multinucleation. In addition, molding of the nuclei to each other within the multinucleated cells and margination of the nuclear chromatin may be appreciated (Figure 2). Intranuclear inclusion bodies, also known as Cowdry type A bodies, can be seen that are nearly the size of red blood cells but are rare to find, with only 10% of specimens exhibiting this finding in a prospective review of 299 patients with herpetic vesiculobullous lesions.11 Evaluation of the contents of blisters caused by bullous pemphigoid and erythema toxicum neonatorum may yield high densities of eosinophils with normal keratinocyte morphology (Figure 3). Other blistering eruptions such as pemphigus vulgaris and bullous drug eruptions also have characteristic findings.1,2


Gout Preparation
Gout is a systemic disease caused by uric acid accumulation that can present with joint pain and white to red nodules on digits, joints, and ears (known as tophi). Material may be expressed from tophi and examined immediately by polarized light microscopy to confirm the diagnosis.5 Specimens will demonstrate needle-shaped, negatively birefringent monosodium urate crystals on polarized light microscopy (Figure 4). An ordinary light microscope can be converted for such use with the lenses of inexpensive polarized sunglasses, placing one lens between the light source and specimen and the other lens between the examiner’s eye and the specimen.12

Parasitic Infections
Two common parasitic infections identified in outpatient dermatology clinics are scabies mites and Demodex mites. Human scabies is extremely pruritic and caused by infestation with Sarcoptes scabiei var hominis; the typical presentation in an adult is erythematous and crusted papules, linear burrows, and vesiculopustules, especially of the interdigital spaces, wrists, axillae, umbilicus, and genital region.1,13 Demodicidosis presents with papules and pustules on the face, usually in a patient with background rosacea and diffuse erythema.1,5,14
If either of these conditions are suspected, mineral oil should be used to prepare the slide because it will maintain viability of the organisms, which are visualized better in motion. Adult scabies mites are roughly 10 times larger than keratinocytes, measuring approximately 250 to 450 µm in length with 8 legs.13 Eggs also may be visualized within the cellular debris and typically are 100 to 150 µm in size and ovoid in shape. Of note, polariscopic examination may be a useful adjunct for evaluation of scabies because scabetic spines and scybala (or fecal material) are polarizable.15
Two types of Demodex mites typically are found in the skin: Demodex folliculorum, which are similarly sized to scabies mites with a more oblong body and occur most commonly in mature hair follicles (eg, eyelashes), and Demodex brevis, which are about half the size (150–200 µm) and live in the sebaceous glands of vellus hairs (Figure 5).14 Both of these mites have 8 legs, similar to the scabies mite.

Hair Preparations
Hair preparations for bulbar examination (eg, trichogram) may prove useful in the evaluation of many types of alopecia, and elaboration on this topic is beyond the scope of this article. Microscopic evaluation of the hair shaft may be an underutilized technique in the outpatient setting and is capable of yielding a variety of diagnoses, including monilethrix, pili torti, and pili trianguli et canaliculi, among others.3 One particularly useful scenario for hair shaft examination (usually of the eyebrow) is in the setting of a patient with severe atopic dermatitis or a baby with ichthyosiform erythroderma, as discovery of trichorrhexis invaginata is pathognomonic for the diagnosis of Netherton syndrome.16 Lastly, evaluation of the hair shaft in patients with patchy and diffuse hair loss whose clinical impression is reminiscent of alopecia areata, or those with concerns of inability to grow hair beyond a short length, may lead to diagnosis of loose anagen syndrome, especially if more than 70% of hair fibers examined exhibit the classic findings of a ruffled proximal cuticle and lack of root sheath.4
Final Thoughts
Bedside microscopy is a rapid and cost-sensitive way to confirm diagnoses that are clinically suspected and remains a valuable tool to acquire during residency training.
- Wanat KA, Dominguez AR, Carter Z, et al. Bedside diagnostics in dermatology: viral, bacterial, and fungal infections. J Am Acad Dermatol. 2017;77:197-218.
- Micheletti RG, Dominguez AR, Wanat KA. Bedside diagnostics in dermatology: parasitic and noninfectious diseases. J Am Acad Dermatol. 2017;77:221-230.
- Whiting DA, Dy LC. Office diagnosis of hair shaft defects. Semin Cutan Med Surg. 2006;25:24-34.
- Tosti A. Loose anagen hair syndrome and loose anagen hair. Arch Dermatol. 2002;138:521-522.
- Bolognia JL, Schaffer JV, Cerroni L, eds. Dermatology. 4th ed. Philadelphia PA: Elsevier; 2017.
- Lilly KK, Koshnick RL, Grill JP, et al. Cost-effectiveness of diagnostic tests for toenail onychomycosis: a repeated-measure, single-blinded, cross-sectional evaluation of 7 diagnostic tests. J Am Acad Dermatol. 2006;55:620-626.
- Elder DE, ed. Lever’s Histopathology of the Skin. 10th ed. Philadelphia, PA: Wolters Kluwer Health/Lippincott Williams & Wilkins; 2009.
- Raghukumar S, Ravikumar BC. Potassium hydroxide mount with cellophane adhesive: a method for direct diagnosis of dermatophyte skin infections [published online May 29, 2018]. Clin Exp Dermatol. doi:10.1111/ced.13573.
- Bhat YJ, Zeerak S, Kanth F, et al. Clinicoepidemiological and mycological study of tinea capitis in the pediatric population of Kashmir Valley: a study from a tertiary care centre. Indian Dermatol Online J. 2017;8:100-103.
- Gupta LK, Singhi MK. Tzanck smear: a useful diagnostic tool. Indian J Dermatol Venereol Leprol. 2005;71:295-299.
- Durdu M, Baba M, Seçkin D. The value of Tzanck smear test in diagnosis of erosive, vesicular, bullous, and pustular skin lesions. J Am Acad Dermatol. 2008;59:958-964.
- Fagan TJ, Lidsky MD. Compensated polarized light microscopy using cellophane adhesive tape. Arthritis Rheum. 1974;17:256-262.
- Walton SF, Currie BJ. Problems in diagnosing scabies, a global disease in human and animal populations. Clin Microbiol Rev. 2007;20:268-279.
- Desch C, Nutting WB. Demodex folliculorum (Simon) and D. brevis akbulatova of man: redescription and reevaluation. J Parasitol. 1972;58:169-177.
- Foo CW, Florell SR, Bowen AR. Polarizable elements in scabies infestation: a clue to diagnosis. J Cutan Pathol. 2013;40:6-10.
- Akkurt ZM, Tuncel T, Ayhan E, et al. Rapid and easy diagnosis of Netherton syndrome with dermoscopy. J Cutan Med Surg. 2014;18:280-282.
- Wanat KA, Dominguez AR, Carter Z, et al. Bedside diagnostics in dermatology: viral, bacterial, and fungal infections. J Am Acad Dermatol. 2017;77:197-218.
- Micheletti RG, Dominguez AR, Wanat KA. Bedside diagnostics in dermatology: parasitic and noninfectious diseases. J Am Acad Dermatol. 2017;77:221-230.
- Whiting DA, Dy LC. Office diagnosis of hair shaft defects. Semin Cutan Med Surg. 2006;25:24-34.
- Tosti A. Loose anagen hair syndrome and loose anagen hair. Arch Dermatol. 2002;138:521-522.
- Bolognia JL, Schaffer JV, Cerroni L, eds. Dermatology. 4th ed. Philadelphia PA: Elsevier; 2017.
- Lilly KK, Koshnick RL, Grill JP, et al. Cost-effectiveness of diagnostic tests for toenail onychomycosis: a repeated-measure, single-blinded, cross-sectional evaluation of 7 diagnostic tests. J Am Acad Dermatol. 2006;55:620-626.
- Elder DE, ed. Lever’s Histopathology of the Skin. 10th ed. Philadelphia, PA: Wolters Kluwer Health/Lippincott Williams & Wilkins; 2009.
- Raghukumar S, Ravikumar BC. Potassium hydroxide mount with cellophane adhesive: a method for direct diagnosis of dermatophyte skin infections [published online May 29, 2018]. Clin Exp Dermatol. doi:10.1111/ced.13573.
- Bhat YJ, Zeerak S, Kanth F, et al. Clinicoepidemiological and mycological study of tinea capitis in the pediatric population of Kashmir Valley: a study from a tertiary care centre. Indian Dermatol Online J. 2017;8:100-103.
- Gupta LK, Singhi MK. Tzanck smear: a useful diagnostic tool. Indian J Dermatol Venereol Leprol. 2005;71:295-299.
- Durdu M, Baba M, Seçkin D. The value of Tzanck smear test in diagnosis of erosive, vesicular, bullous, and pustular skin lesions. J Am Acad Dermatol. 2008;59:958-964.
- Fagan TJ, Lidsky MD. Compensated polarized light microscopy using cellophane adhesive tape. Arthritis Rheum. 1974;17:256-262.
- Walton SF, Currie BJ. Problems in diagnosing scabies, a global disease in human and animal populations. Clin Microbiol Rev. 2007;20:268-279.
- Desch C, Nutting WB. Demodex folliculorum (Simon) and D. brevis akbulatova of man: redescription and reevaluation. J Parasitol. 1972;58:169-177.
- Foo CW, Florell SR, Bowen AR. Polarizable elements in scabies infestation: a clue to diagnosis. J Cutan Pathol. 2013;40:6-10.
- Akkurt ZM, Tuncel T, Ayhan E, et al. Rapid and easy diagnosis of Netherton syndrome with dermoscopy. J Cutan Med Surg. 2014;18:280-282.
How Can Neurologists Diagnose Spontaneous Intracranial Hypotension?
Imaging results may be negative, but risk factors and symptoms can make the diagnosis more certain.
SAN FRANCISCO—Spontaneous intracranial hypotension (SIH) sometimes may go undiagnosed, partly because the disorder is uncommon. But patients will receive the care that they need if neurologists, particularly headache specialists, understand how to identify the disorder.
Misconceptions About SIH
The name of the disorder can be misleading, said Deborah I. Friedman, MD, at the 60th Annual Scientific Meeting of the American Headache Society. SIH is not always spontaneous; it often has an antecedent cause. Furthermore, the main problem is not intracranial, it is a leak in the spinal column, most often in the low cervical or thoracic zone. Finally, CSF pressure is usually normal in these patients, said Dr. Friedman, Chief of the Division of Headache Medicine, Professor of Neurology, and Professor of Ophthalmology at the University of Texas Southwestern Medical Center in Dallas.
SIH is considered a rare disorder, and its published annual incidence is five cases per 100,000 people. But this prevalence may be a gross underestimate that results from the absence of an ICD-9 or ICD-10 code for the condition, according to Dr. Friedman.
SIH is “much more common than we think,” she asserted. “These people are out there. They are in your offices. I can guarantee you, there are patients you have been seeing for years in your practice that have [SIH]. I have missed it. I bet you have, too.”
Guidelines for Identifying SIH
Dr. Friedman offered advice from the perspective of a headache specialist to guide the diagnosis of SIH. “Most of the literature that is out there, and it is good literature, was not written by headache medicine specialists, it was written by famous and prominent neurosurgeons and neuroradiologists. But the people we see are not necessarily the people they see,” Dr. Friedman explained.
SIH can be challenging to diagnose because of its myriad presentations. “You need to be a detective,” said Dr. Friedman. The questions to ask center around whether the headache has postural, end-of-the-day, and Valsalva components. Joint hypermobility may provide another clue.
Headache is the most common symptom of SIH and the reason that patients with the disorder seek a headache specialist. A neurologist should consider the diagnosis in a patient with a new daily persistent headache or in a patient with a diagnosis of chronic migraine for whom no medication has worked. “The people who come in with a huge list of medications they have tried, and nothing works? That is unusual for migraine. Usually something works for migraine,” said Dr. Friedman.
SIH can result in a headache with an onset as sudden as that of thunderclap headache, but this characteristic is not necessary. The most common location of pain is posterior, but the pain can be centered anywhere in the head or face. Bilateral pain is more common than unilateral pain.
The most typical headache is orthostatic or worsens at the end of the day. The longer a patient has SIH, the less likely that it will have a postural component. Most patients are awakened by their headache in the middle of the night. The headache is often exertional and usually worsens with Valsalva maneuvers, including coughing, sneezing, lifting, bending forward, straining, singing, or sexual activity. Caffeine often works well for people with SIH. A neurologist should ask the patient about these issues, said Dr. Friedman.
Besides headache, other common symptoms of SIH include tinnitus, abnormal hearing (eg, hearing things as though one is underwater), neck pain, imbalance, pain between the shoulder blades, and blurred or double vision.
Typical risk factors include joint hypermobility; previous lumbar puncture, epidural, or spinal anesthesia; known disc disease or a personal or family history of retinal detachment at a young age; aneurysm; dissection; and valvular heart disease. Joint hypermobility is widespread among patients with SIH. These patients often enjoy yoga and were exceptionally flexible as children. Many participated in gymnastics, ballet, or cheerleading as children.
Examining and Treating the Patient
On physical examination, a neurologist can look for joint hypermobility. He or she should examine the eyes for spontaneous retinal venous pulsations indicative of normal CSF pressure. A neurologist also can put the patient in 5° of the Trendelenburg position for five to 10 minutes to see whether it improves the headache and other symptoms.
One of the first things that Dr. Friedman does when she suspects SIH is to refer the patient to the website of the Spinal CSF Leak Foundation (spinalcsfleak.org). She asks him or her to review the site and tell her whether the descriptions sound familiar.
The medical consensus is that the first-line diagnostic test is brain MRI with gadolinium enhancement. The diagnostic challenge, however, is that 30% of patients with SIH have normal results.
There is no consensus about the next step when the brain MRI is negative. CT with or without MR myelography is one possibility, and a T2-weighted spine MRI is another. Despite a thorough search, however, neurologists find no leak in about half of individuals with SIH.
Conservative treatment measures do not work well, according to Dr. Friedman. A reasonable strategy, even if a leak site has not been identified, is to treat with a high-volume epidural CT-guided targeted blood patch with fibrin sealant. “It gives relief about a third of the time,” according to Dr. Friedman.
—Bruce Jancin
Imaging results may be negative, but risk factors and symptoms can make the diagnosis more certain.
Imaging results may be negative, but risk factors and symptoms can make the diagnosis more certain.
SAN FRANCISCO—Spontaneous intracranial hypotension (SIH) sometimes may go undiagnosed, partly because the disorder is uncommon. But patients will receive the care that they need if neurologists, particularly headache specialists, understand how to identify the disorder.
Misconceptions About SIH
The name of the disorder can be misleading, said Deborah I. Friedman, MD, at the 60th Annual Scientific Meeting of the American Headache Society. SIH is not always spontaneous; it often has an antecedent cause. Furthermore, the main problem is not intracranial, it is a leak in the spinal column, most often in the low cervical or thoracic zone. Finally, CSF pressure is usually normal in these patients, said Dr. Friedman, Chief of the Division of Headache Medicine, Professor of Neurology, and Professor of Ophthalmology at the University of Texas Southwestern Medical Center in Dallas.
SIH is considered a rare disorder, and its published annual incidence is five cases per 100,000 people. But this prevalence may be a gross underestimate that results from the absence of an ICD-9 or ICD-10 code for the condition, according to Dr. Friedman.
SIH is “much more common than we think,” she asserted. “These people are out there. They are in your offices. I can guarantee you, there are patients you have been seeing for years in your practice that have [SIH]. I have missed it. I bet you have, too.”
Guidelines for Identifying SIH
Dr. Friedman offered advice from the perspective of a headache specialist to guide the diagnosis of SIH. “Most of the literature that is out there, and it is good literature, was not written by headache medicine specialists, it was written by famous and prominent neurosurgeons and neuroradiologists. But the people we see are not necessarily the people they see,” Dr. Friedman explained.
SIH can be challenging to diagnose because of its myriad presentations. “You need to be a detective,” said Dr. Friedman. The questions to ask center around whether the headache has postural, end-of-the-day, and Valsalva components. Joint hypermobility may provide another clue.
Headache is the most common symptom of SIH and the reason that patients with the disorder seek a headache specialist. A neurologist should consider the diagnosis in a patient with a new daily persistent headache or in a patient with a diagnosis of chronic migraine for whom no medication has worked. “The people who come in with a huge list of medications they have tried, and nothing works? That is unusual for migraine. Usually something works for migraine,” said Dr. Friedman.
SIH can result in a headache with an onset as sudden as that of thunderclap headache, but this characteristic is not necessary. The most common location of pain is posterior, but the pain can be centered anywhere in the head or face. Bilateral pain is more common than unilateral pain.
The most typical headache is orthostatic or worsens at the end of the day. The longer a patient has SIH, the less likely that it will have a postural component. Most patients are awakened by their headache in the middle of the night. The headache is often exertional and usually worsens with Valsalva maneuvers, including coughing, sneezing, lifting, bending forward, straining, singing, or sexual activity. Caffeine often works well for people with SIH. A neurologist should ask the patient about these issues, said Dr. Friedman.
Besides headache, other common symptoms of SIH include tinnitus, abnormal hearing (eg, hearing things as though one is underwater), neck pain, imbalance, pain between the shoulder blades, and blurred or double vision.
Typical risk factors include joint hypermobility; previous lumbar puncture, epidural, or spinal anesthesia; known disc disease or a personal or family history of retinal detachment at a young age; aneurysm; dissection; and valvular heart disease. Joint hypermobility is widespread among patients with SIH. These patients often enjoy yoga and were exceptionally flexible as children. Many participated in gymnastics, ballet, or cheerleading as children.
Examining and Treating the Patient
On physical examination, a neurologist can look for joint hypermobility. He or she should examine the eyes for spontaneous retinal venous pulsations indicative of normal CSF pressure. A neurologist also can put the patient in 5° of the Trendelenburg position for five to 10 minutes to see whether it improves the headache and other symptoms.
One of the first things that Dr. Friedman does when she suspects SIH is to refer the patient to the website of the Spinal CSF Leak Foundation (spinalcsfleak.org). She asks him or her to review the site and tell her whether the descriptions sound familiar.
The medical consensus is that the first-line diagnostic test is brain MRI with gadolinium enhancement. The diagnostic challenge, however, is that 30% of patients with SIH have normal results.
There is no consensus about the next step when the brain MRI is negative. CT with or without MR myelography is one possibility, and a T2-weighted spine MRI is another. Despite a thorough search, however, neurologists find no leak in about half of individuals with SIH.
Conservative treatment measures do not work well, according to Dr. Friedman. A reasonable strategy, even if a leak site has not been identified, is to treat with a high-volume epidural CT-guided targeted blood patch with fibrin sealant. “It gives relief about a third of the time,” according to Dr. Friedman.
—Bruce Jancin
SAN FRANCISCO—Spontaneous intracranial hypotension (SIH) sometimes may go undiagnosed, partly because the disorder is uncommon. But patients will receive the care that they need if neurologists, particularly headache specialists, understand how to identify the disorder.
Misconceptions About SIH
The name of the disorder can be misleading, said Deborah I. Friedman, MD, at the 60th Annual Scientific Meeting of the American Headache Society. SIH is not always spontaneous; it often has an antecedent cause. Furthermore, the main problem is not intracranial, it is a leak in the spinal column, most often in the low cervical or thoracic zone. Finally, CSF pressure is usually normal in these patients, said Dr. Friedman, Chief of the Division of Headache Medicine, Professor of Neurology, and Professor of Ophthalmology at the University of Texas Southwestern Medical Center in Dallas.
SIH is considered a rare disorder, and its published annual incidence is five cases per 100,000 people. But this prevalence may be a gross underestimate that results from the absence of an ICD-9 or ICD-10 code for the condition, according to Dr. Friedman.
SIH is “much more common than we think,” she asserted. “These people are out there. They are in your offices. I can guarantee you, there are patients you have been seeing for years in your practice that have [SIH]. I have missed it. I bet you have, too.”
Guidelines for Identifying SIH
Dr. Friedman offered advice from the perspective of a headache specialist to guide the diagnosis of SIH. “Most of the literature that is out there, and it is good literature, was not written by headache medicine specialists, it was written by famous and prominent neurosurgeons and neuroradiologists. But the people we see are not necessarily the people they see,” Dr. Friedman explained.
SIH can be challenging to diagnose because of its myriad presentations. “You need to be a detective,” said Dr. Friedman. The questions to ask center around whether the headache has postural, end-of-the-day, and Valsalva components. Joint hypermobility may provide another clue.
Headache is the most common symptom of SIH and the reason that patients with the disorder seek a headache specialist. A neurologist should consider the diagnosis in a patient with a new daily persistent headache or in a patient with a diagnosis of chronic migraine for whom no medication has worked. “The people who come in with a huge list of medications they have tried, and nothing works? That is unusual for migraine. Usually something works for migraine,” said Dr. Friedman.
SIH can result in a headache with an onset as sudden as that of thunderclap headache, but this characteristic is not necessary. The most common location of pain is posterior, but the pain can be centered anywhere in the head or face. Bilateral pain is more common than unilateral pain.
The most typical headache is orthostatic or worsens at the end of the day. The longer a patient has SIH, the less likely that it will have a postural component. Most patients are awakened by their headache in the middle of the night. The headache is often exertional and usually worsens with Valsalva maneuvers, including coughing, sneezing, lifting, bending forward, straining, singing, or sexual activity. Caffeine often works well for people with SIH. A neurologist should ask the patient about these issues, said Dr. Friedman.
Besides headache, other common symptoms of SIH include tinnitus, abnormal hearing (eg, hearing things as though one is underwater), neck pain, imbalance, pain between the shoulder blades, and blurred or double vision.
Typical risk factors include joint hypermobility; previous lumbar puncture, epidural, or spinal anesthesia; known disc disease or a personal or family history of retinal detachment at a young age; aneurysm; dissection; and valvular heart disease. Joint hypermobility is widespread among patients with SIH. These patients often enjoy yoga and were exceptionally flexible as children. Many participated in gymnastics, ballet, or cheerleading as children.
Examining and Treating the Patient
On physical examination, a neurologist can look for joint hypermobility. He or she should examine the eyes for spontaneous retinal venous pulsations indicative of normal CSF pressure. A neurologist also can put the patient in 5° of the Trendelenburg position for five to 10 minutes to see whether it improves the headache and other symptoms.
One of the first things that Dr. Friedman does when she suspects SIH is to refer the patient to the website of the Spinal CSF Leak Foundation (spinalcsfleak.org). She asks him or her to review the site and tell her whether the descriptions sound familiar.
The medical consensus is that the first-line diagnostic test is brain MRI with gadolinium enhancement. The diagnostic challenge, however, is that 30% of patients with SIH have normal results.
There is no consensus about the next step when the brain MRI is negative. CT with or without MR myelography is one possibility, and a T2-weighted spine MRI is another. Despite a thorough search, however, neurologists find no leak in about half of individuals with SIH.
Conservative treatment measures do not work well, according to Dr. Friedman. A reasonable strategy, even if a leak site has not been identified, is to treat with a high-volume epidural CT-guided targeted blood patch with fibrin sealant. “It gives relief about a third of the time,” according to Dr. Friedman.
—Bruce Jancin
Learning Curves: Historical Trends of FDA-Reported Adverse Events for Dermal Fillers
Dermal fillers are considered Class III medical devices by the US Food and Drug Administration (FDA).1 Reports of adverse events (AEs) for medical devices are made public by the FDA to allow for transparent postmarketing surveillance.2The AE trends extracted from these historical data may help distinguish between expected learning curves of new dermal fillers versus unsafe products that may require FDA intervention. Considering that aesthetic treatments are not medically necessary, a low risk profile is paramount and determining what constitutes normal learning curves is important for impartial assessment of AEs as new fillers come on the market. The concept of a 3-year learning curve can be an important tool for safety monitoring going forward, creating a bar for quality that could trigger increased surveillance if a product fails to meet an expected arc of diminished AEs over time. This study serves to evaluate historical AE data and to establish learning curves for FDA-approved dermal fillers.
Methods
We searched the OpenFDA Device Adverse Event Report Browser (http://openfda.shinyapps.io/devicereports/) for reported AEs within the FDA product code LMH (Implant, Dermal, For Aesthetic Use) that were received from January 1, 1983, to December 31, 2017. For each reported AE, information related to the date of the reported event and the device brand name were recorded. Devices implicated in each AE were classified based on primary composition according to the following 5 categories: collagen, hyaluronic acid (HA), hydroxylapatite, poly-L-lactic acid (PLLA), and polymethyl methacrylate (PMMA). Inaccurate entries of reported AEs or those intended for nonaesthetic use were excluded from the study. A total of 8530 AEs were included in the study. To normalize the data, we obtained annual reports for the number of procedures performed by filler type from the American Society of Plastic Surgeons (ASPS) cosmetic procedure trends. 3 We calculated the annual AE rates for each approved filler by dividing the number of AEs by the number of procedures performed that year.
Results
The trends of different filler types depicting the number of procedures performed over time are shown in Figure 1. Data from the ASPS dated back to 2005; therefore, the number of procedures performed prior to that were extrapolated with knowledge of products’ approval dates and market share, indicated by a dotted line. To determine AE rates for each year, we divided the number of AEs by the number of reported procedures for each filler type. The AE rates are displayed graphically in Figures 2 and 3 with superimposed FDA approval dates for each filler.4
Two major peaks in reported AE rates for all fillers were noted in the late 1990s and late 2000s, mostly associated with collagen and PLLA fillers, respectively (Figure 2). Overall, there has been a low rate of AEs associated with HA fillers since their initial approval in the early 2000s.
Individual filler AE rates also were analyzed. Hyaluronic acid fillers were associated with an extremely low rate of AEs, ranging from 1 to 4 AEs per 10,000 procedures (Figure 3A). This low AE rate profile underscores the safety of HA fillers, which has spurred their popularity. Adverseevent rates for collagen fillers spiked in the mid- to late 1990s and resolved over the course of the next 3 years (Figure 3B). Hydroxylapatite fillers had a rather uniform AE rate with an early indication of a drop-off after 2015 (Figure 3C). Poly-L-lactic acid fillers showed the steepest learning curve, with a peak of 1 AE per 100 procedures after they were approved in 2008 (Figure 3D); however, there is a comparable 3-year resolution of AE rates. Adverse events for PMMA fillers did not show specific resolution, meaning that they did not follow the 3-year arc that was seen for the other dermal fillers reported in the data set (Figure 3E).
Comment
Our study is unique in that it analyzes reported AE data over a 34-year period for injectable dermal fillers. To our knowledge, this novel method of calculating AE rates across dermal fillers and for individual products is the first of its kind that facilitates usage-normalized comparison of different filler types.
All OpenFDA data are self-reported and therefore have inherent limitations. Anyone can enter information on AEs in this system, including both patients and health care providers, so the quality of the input may be variable. However, this output is the only representation we have for nearly 35 years of AE history for this burgeoning category of popular aesthetic treatments. Another study limitation is that not everyone may know that reporting an AE in the OpenFDA is an option; therefore, we may be missing a portion of AEs due to underreporting. Underreporting may be especially at play in the years before the Internet was prevalent for residential use since access to the Internet would be required to report an AR on the website. However, examining the available data provides an important window into valuable information on complications that have occurred and have been reported for FDA-approved dermal fillers.
An additional challenge in constructing this study was assessing the total number of injectable dermal filler treatments being performed annually across filler types for normalization of the data. Although the absolute numbers of filler use as captured by the ASPS are smaller than the true total filler use across all injectors, the relative use of different filler products will be similar across all specialties because it reflects product popularity. Annual surveys on aesthetic procedures also are conducted by the American Society for Dermatologic Surgery and the American Association for Facial Plastic and Reconstructive Surgery, but neither one captures the relative usage of different filler types. Because individual filler companies do not publish their annual sales numbers by product, the ASPS data give us the best gauge of relative use of fillers by product type given the available information. We conclude that the comparison of AE rates would remain the same even if we had data for total annual filler use across specialties.
Our graphical depiction of the data clearly demonstrates the low AE profile of HA fillers, which is in line with the general consensus of their safety that has contributed to their vast popularity; however, this study represents the first time usage-normalized AE rates are compared to other filler compositions. Hyaluronic acid fillers have the unique feature of being able to be dissolved with the hyaluronidase enzyme, which can limit adverse event potential as compared to other ingredient classes of filler types and may be reflected in their low overall AE profile. The AE rate spike and resolution for collagen fillers represent what we refer to as a “normal learning curve” based on our analysis of the data set as a whole, suggesting an appropriate time course of increased familiarity with the product without inherent issues with the product itself. Multiple sequential anatomic site indications were approved for hydroxylapatite fillers from 2006 through 2015, which may have yielded overlapping learning curves for each approval, resulting in a rather uniform AE rate. The early drop-off in AE rates after the 2015 anatomic site approval may represent the beginning of a normal learning curve, and continued surveillance of AE rates would be of value to confirm this trend. We saw a similar 3-year learning curve for PLLA fillers as the curve for collagen fillers, suggesting a normal learning curve and no out-of-line safety issues. Polymethylmethacrylate fillers were approved in 2006 and were taken off the market for a period in the late 2000s, explaining the drop-off. Once they were back on the market, we do not see a typical learning curve for PMMA, which may warrant surveillance for safety by both clinicians and the FDA.
Conclusion
Our study represents a novel method of evaluating the safety of medical devices, specifically aesthetic fillers. We showed that every AE rate curve for different filler types tells a story. Reactions to AEs for new fillers should be placed in the context of whether they seem to be following the established learning curve.
- Dermal fillers (soft tissue fillers). US Food and Drug Administration website. https://www.fda.gov/MedicalDevices/ucm2007470.htm. Updated May 31, 2018. Accessed June 29, 2018.
- Kass-Hout TA, Xu Z, Mohebbi M, et al. OpenFDA: an innovative platform providing access to a wealth of FDA’s publicly available data. J Am Med Inform Assoc. 2016;23:596-600.
- Plastic surgery statistics. American Society of Plastic Surgeons website. https://www.plasticsurgery.org/documents/News/Statistics/2017/plastic-surgery-statistics-full-report-2017.pdf. Accessed June 28, 2018.
- Dermal fillers approved by the Center for Devices and Radiological Health. US Food and Drug Administration website. https://www.fda.gov/MedicalDevices/ProductsandMedicalProcedures/CosmeticDevices/WrinkleFillers/ucm227749.htm. Accessed June 28, 2018. Updated March 19, 2018.
Dermal fillers are considered Class III medical devices by the US Food and Drug Administration (FDA).1 Reports of adverse events (AEs) for medical devices are made public by the FDA to allow for transparent postmarketing surveillance.2The AE trends extracted from these historical data may help distinguish between expected learning curves of new dermal fillers versus unsafe products that may require FDA intervention. Considering that aesthetic treatments are not medically necessary, a low risk profile is paramount and determining what constitutes normal learning curves is important for impartial assessment of AEs as new fillers come on the market. The concept of a 3-year learning curve can be an important tool for safety monitoring going forward, creating a bar for quality that could trigger increased surveillance if a product fails to meet an expected arc of diminished AEs over time. This study serves to evaluate historical AE data and to establish learning curves for FDA-approved dermal fillers.
Methods
We searched the OpenFDA Device Adverse Event Report Browser (http://openfda.shinyapps.io/devicereports/) for reported AEs within the FDA product code LMH (Implant, Dermal, For Aesthetic Use) that were received from January 1, 1983, to December 31, 2017. For each reported AE, information related to the date of the reported event and the device brand name were recorded. Devices implicated in each AE were classified based on primary composition according to the following 5 categories: collagen, hyaluronic acid (HA), hydroxylapatite, poly-L-lactic acid (PLLA), and polymethyl methacrylate (PMMA). Inaccurate entries of reported AEs or those intended for nonaesthetic use were excluded from the study. A total of 8530 AEs were included in the study. To normalize the data, we obtained annual reports for the number of procedures performed by filler type from the American Society of Plastic Surgeons (ASPS) cosmetic procedure trends. 3 We calculated the annual AE rates for each approved filler by dividing the number of AEs by the number of procedures performed that year.
Results
The trends of different filler types depicting the number of procedures performed over time are shown in Figure 1. Data from the ASPS dated back to 2005; therefore, the number of procedures performed prior to that were extrapolated with knowledge of products’ approval dates and market share, indicated by a dotted line. To determine AE rates for each year, we divided the number of AEs by the number of reported procedures for each filler type. The AE rates are displayed graphically in Figures 2 and 3 with superimposed FDA approval dates for each filler.4

Two major peaks in reported AE rates for all fillers were noted in the late 1990s and late 2000s, mostly associated with collagen and PLLA fillers, respectively (Figure 2). Overall, there has been a low rate of AEs associated with HA fillers since their initial approval in the early 2000s.

Individual filler AE rates also were analyzed. Hyaluronic acid fillers were associated with an extremely low rate of AEs, ranging from 1 to 4 AEs per 10,000 procedures (Figure 3A). This low AE rate profile underscores the safety of HA fillers, which has spurred their popularity. Adverseevent rates for collagen fillers spiked in the mid- to late 1990s and resolved over the course of the next 3 years (Figure 3B). Hydroxylapatite fillers had a rather uniform AE rate with an early indication of a drop-off after 2015 (Figure 3C). Poly-L-lactic acid fillers showed the steepest learning curve, with a peak of 1 AE per 100 procedures after they were approved in 2008 (Figure 3D); however, there is a comparable 3-year resolution of AE rates. Adverse events for PMMA fillers did not show specific resolution, meaning that they did not follow the 3-year arc that was seen for the other dermal fillers reported in the data set (Figure 3E).

Comment
Our study is unique in that it analyzes reported AE data over a 34-year period for injectable dermal fillers. To our knowledge, this novel method of calculating AE rates across dermal fillers and for individual products is the first of its kind that facilitates usage-normalized comparison of different filler types.
All OpenFDA data are self-reported and therefore have inherent limitations. Anyone can enter information on AEs in this system, including both patients and health care providers, so the quality of the input may be variable. However, this output is the only representation we have for nearly 35 years of AE history for this burgeoning category of popular aesthetic treatments. Another study limitation is that not everyone may know that reporting an AE in the OpenFDA is an option; therefore, we may be missing a portion of AEs due to underreporting. Underreporting may be especially at play in the years before the Internet was prevalent for residential use since access to the Internet would be required to report an AR on the website. However, examining the available data provides an important window into valuable information on complications that have occurred and have been reported for FDA-approved dermal fillers.
An additional challenge in constructing this study was assessing the total number of injectable dermal filler treatments being performed annually across filler types for normalization of the data. Although the absolute numbers of filler use as captured by the ASPS are smaller than the true total filler use across all injectors, the relative use of different filler products will be similar across all specialties because it reflects product popularity. Annual surveys on aesthetic procedures also are conducted by the American Society for Dermatologic Surgery and the American Association for Facial Plastic and Reconstructive Surgery, but neither one captures the relative usage of different filler types. Because individual filler companies do not publish their annual sales numbers by product, the ASPS data give us the best gauge of relative use of fillers by product type given the available information. We conclude that the comparison of AE rates would remain the same even if we had data for total annual filler use across specialties.
Our graphical depiction of the data clearly demonstrates the low AE profile of HA fillers, which is in line with the general consensus of their safety that has contributed to their vast popularity; however, this study represents the first time usage-normalized AE rates are compared to other filler compositions. Hyaluronic acid fillers have the unique feature of being able to be dissolved with the hyaluronidase enzyme, which can limit adverse event potential as compared to other ingredient classes of filler types and may be reflected in their low overall AE profile. The AE rate spike and resolution for collagen fillers represent what we refer to as a “normal learning curve” based on our analysis of the data set as a whole, suggesting an appropriate time course of increased familiarity with the product without inherent issues with the product itself. Multiple sequential anatomic site indications were approved for hydroxylapatite fillers from 2006 through 2015, which may have yielded overlapping learning curves for each approval, resulting in a rather uniform AE rate. The early drop-off in AE rates after the 2015 anatomic site approval may represent the beginning of a normal learning curve, and continued surveillance of AE rates would be of value to confirm this trend. We saw a similar 3-year learning curve for PLLA fillers as the curve for collagen fillers, suggesting a normal learning curve and no out-of-line safety issues. Polymethylmethacrylate fillers were approved in 2006 and were taken off the market for a period in the late 2000s, explaining the drop-off. Once they were back on the market, we do not see a typical learning curve for PMMA, which may warrant surveillance for safety by both clinicians and the FDA.
Conclusion
Our study represents a novel method of evaluating the safety of medical devices, specifically aesthetic fillers. We showed that every AE rate curve for different filler types tells a story. Reactions to AEs for new fillers should be placed in the context of whether they seem to be following the established learning curve.
Dermal fillers are considered Class III medical devices by the US Food and Drug Administration (FDA).1 Reports of adverse events (AEs) for medical devices are made public by the FDA to allow for transparent postmarketing surveillance.2The AE trends extracted from these historical data may help distinguish between expected learning curves of new dermal fillers versus unsafe products that may require FDA intervention. Considering that aesthetic treatments are not medically necessary, a low risk profile is paramount and determining what constitutes normal learning curves is important for impartial assessment of AEs as new fillers come on the market. The concept of a 3-year learning curve can be an important tool for safety monitoring going forward, creating a bar for quality that could trigger increased surveillance if a product fails to meet an expected arc of diminished AEs over time. This study serves to evaluate historical AE data and to establish learning curves for FDA-approved dermal fillers.
Methods
We searched the OpenFDA Device Adverse Event Report Browser (http://openfda.shinyapps.io/devicereports/) for reported AEs within the FDA product code LMH (Implant, Dermal, For Aesthetic Use) that were received from January 1, 1983, to December 31, 2017. For each reported AE, information related to the date of the reported event and the device brand name were recorded. Devices implicated in each AE were classified based on primary composition according to the following 5 categories: collagen, hyaluronic acid (HA), hydroxylapatite, poly-L-lactic acid (PLLA), and polymethyl methacrylate (PMMA). Inaccurate entries of reported AEs or those intended for nonaesthetic use were excluded from the study. A total of 8530 AEs were included in the study. To normalize the data, we obtained annual reports for the number of procedures performed by filler type from the American Society of Plastic Surgeons (ASPS) cosmetic procedure trends. 3 We calculated the annual AE rates for each approved filler by dividing the number of AEs by the number of procedures performed that year.
Results
The trends of different filler types depicting the number of procedures performed over time are shown in Figure 1. Data from the ASPS dated back to 2005; therefore, the number of procedures performed prior to that were extrapolated with knowledge of products’ approval dates and market share, indicated by a dotted line. To determine AE rates for each year, we divided the number of AEs by the number of reported procedures for each filler type. The AE rates are displayed graphically in Figures 2 and 3 with superimposed FDA approval dates for each filler.4

Two major peaks in reported AE rates for all fillers were noted in the late 1990s and late 2000s, mostly associated with collagen and PLLA fillers, respectively (Figure 2). Overall, there has been a low rate of AEs associated with HA fillers since their initial approval in the early 2000s.

Individual filler AE rates also were analyzed. Hyaluronic acid fillers were associated with an extremely low rate of AEs, ranging from 1 to 4 AEs per 10,000 procedures (Figure 3A). This low AE rate profile underscores the safety of HA fillers, which has spurred their popularity. Adverseevent rates for collagen fillers spiked in the mid- to late 1990s and resolved over the course of the next 3 years (Figure 3B). Hydroxylapatite fillers had a rather uniform AE rate with an early indication of a drop-off after 2015 (Figure 3C). Poly-L-lactic acid fillers showed the steepest learning curve, with a peak of 1 AE per 100 procedures after they were approved in 2008 (Figure 3D); however, there is a comparable 3-year resolution of AE rates. Adverse events for PMMA fillers did not show specific resolution, meaning that they did not follow the 3-year arc that was seen for the other dermal fillers reported in the data set (Figure 3E).

Comment
Our study is unique in that it analyzes reported AE data over a 34-year period for injectable dermal fillers. To our knowledge, this novel method of calculating AE rates across dermal fillers and for individual products is the first of its kind that facilitates usage-normalized comparison of different filler types.
All OpenFDA data are self-reported and therefore have inherent limitations. Anyone can enter information on AEs in this system, including both patients and health care providers, so the quality of the input may be variable. However, this output is the only representation we have for nearly 35 years of AE history for this burgeoning category of popular aesthetic treatments. Another study limitation is that not everyone may know that reporting an AE in the OpenFDA is an option; therefore, we may be missing a portion of AEs due to underreporting. Underreporting may be especially at play in the years before the Internet was prevalent for residential use since access to the Internet would be required to report an AR on the website. However, examining the available data provides an important window into valuable information on complications that have occurred and have been reported for FDA-approved dermal fillers.
An additional challenge in constructing this study was assessing the total number of injectable dermal filler treatments being performed annually across filler types for normalization of the data. Although the absolute numbers of filler use as captured by the ASPS are smaller than the true total filler use across all injectors, the relative use of different filler products will be similar across all specialties because it reflects product popularity. Annual surveys on aesthetic procedures also are conducted by the American Society for Dermatologic Surgery and the American Association for Facial Plastic and Reconstructive Surgery, but neither one captures the relative usage of different filler types. Because individual filler companies do not publish their annual sales numbers by product, the ASPS data give us the best gauge of relative use of fillers by product type given the available information. We conclude that the comparison of AE rates would remain the same even if we had data for total annual filler use across specialties.
Our graphical depiction of the data clearly demonstrates the low AE profile of HA fillers, which is in line with the general consensus of their safety that has contributed to their vast popularity; however, this study represents the first time usage-normalized AE rates are compared to other filler compositions. Hyaluronic acid fillers have the unique feature of being able to be dissolved with the hyaluronidase enzyme, which can limit adverse event potential as compared to other ingredient classes of filler types and may be reflected in their low overall AE profile. The AE rate spike and resolution for collagen fillers represent what we refer to as a “normal learning curve” based on our analysis of the data set as a whole, suggesting an appropriate time course of increased familiarity with the product without inherent issues with the product itself. Multiple sequential anatomic site indications were approved for hydroxylapatite fillers from 2006 through 2015, which may have yielded overlapping learning curves for each approval, resulting in a rather uniform AE rate. The early drop-off in AE rates after the 2015 anatomic site approval may represent the beginning of a normal learning curve, and continued surveillance of AE rates would be of value to confirm this trend. We saw a similar 3-year learning curve for PLLA fillers as the curve for collagen fillers, suggesting a normal learning curve and no out-of-line safety issues. Polymethylmethacrylate fillers were approved in 2006 and were taken off the market for a period in the late 2000s, explaining the drop-off. Once they were back on the market, we do not see a typical learning curve for PMMA, which may warrant surveillance for safety by both clinicians and the FDA.
Conclusion
Our study represents a novel method of evaluating the safety of medical devices, specifically aesthetic fillers. We showed that every AE rate curve for different filler types tells a story. Reactions to AEs for new fillers should be placed in the context of whether they seem to be following the established learning curve.
- Dermal fillers (soft tissue fillers). US Food and Drug Administration website. https://www.fda.gov/MedicalDevices/ucm2007470.htm. Updated May 31, 2018. Accessed June 29, 2018.
- Kass-Hout TA, Xu Z, Mohebbi M, et al. OpenFDA: an innovative platform providing access to a wealth of FDA’s publicly available data. J Am Med Inform Assoc. 2016;23:596-600.
- Plastic surgery statistics. American Society of Plastic Surgeons website. https://www.plasticsurgery.org/documents/News/Statistics/2017/plastic-surgery-statistics-full-report-2017.pdf. Accessed June 28, 2018.
- Dermal fillers approved by the Center for Devices and Radiological Health. US Food and Drug Administration website. https://www.fda.gov/MedicalDevices/ProductsandMedicalProcedures/CosmeticDevices/WrinkleFillers/ucm227749.htm. Accessed June 28, 2018. Updated March 19, 2018.
- Dermal fillers (soft tissue fillers). US Food and Drug Administration website. https://www.fda.gov/MedicalDevices/ucm2007470.htm. Updated May 31, 2018. Accessed June 29, 2018.
- Kass-Hout TA, Xu Z, Mohebbi M, et al. OpenFDA: an innovative platform providing access to a wealth of FDA’s publicly available data. J Am Med Inform Assoc. 2016;23:596-600.
- Plastic surgery statistics. American Society of Plastic Surgeons website. https://www.plasticsurgery.org/documents/News/Statistics/2017/plastic-surgery-statistics-full-report-2017.pdf. Accessed June 28, 2018.
- Dermal fillers approved by the Center for Devices and Radiological Health. US Food and Drug Administration website. https://www.fda.gov/MedicalDevices/ProductsandMedicalProcedures/CosmeticDevices/WrinkleFillers/ucm227749.htm. Accessed June 28, 2018. Updated March 19, 2018.
Resident Pearl
- The US Food and Drug Administration’s (FDA) adverse event database, OpenFDA, provides extensive information regarding safety for a variety of cosmetic devices. Injectable dermal fillers are classified as a medical device by the FDA; therefore, safety studies can be performed using this publicly available database.
Applications for the 2019 Resident Corner Column Now Being Accepted
The Cutis Editorial Board is now accepting applications for the 2019 Resident Corner column. The Editorial Board will select 2 residents to serve as the Resident Corner columnists for 1 year (3 articles each). Articles are posted online only but will be referenced in Index Medicus. All applicants must be current residents and will still be in residency throughout 2019.
For consideration, send your curriculum vitae along with a brief (not to exceed 500 words) statement of why you enjoy Cutis and what you can offer your fellow residents in contributing a monthly column.
All materials should be submitted via email as 1 Word document to Melissa Sears by November 1. The residents who are selected to write the column for the upcoming year will be notified by November 15.
We look forward to continuing to educate dermatology residents on topics that are most important to them!
The Cutis Editorial Board is now accepting applications for the 2019 Resident Corner column. The Editorial Board will select 2 residents to serve as the Resident Corner columnists for 1 year (3 articles each). Articles are posted online only but will be referenced in Index Medicus. All applicants must be current residents and will still be in residency throughout 2019.
For consideration, send your curriculum vitae along with a brief (not to exceed 500 words) statement of why you enjoy Cutis and what you can offer your fellow residents in contributing a monthly column.
All materials should be submitted via email as 1 Word document to Melissa Sears by November 1. The residents who are selected to write the column for the upcoming year will be notified by November 15.
We look forward to continuing to educate dermatology residents on topics that are most important to them!
The Cutis Editorial Board is now accepting applications for the 2019 Resident Corner column. The Editorial Board will select 2 residents to serve as the Resident Corner columnists for 1 year (3 articles each). Articles are posted online only but will be referenced in Index Medicus. All applicants must be current residents and will still be in residency throughout 2019.
For consideration, send your curriculum vitae along with a brief (not to exceed 500 words) statement of why you enjoy Cutis and what you can offer your fellow residents in contributing a monthly column.
All materials should be submitted via email as 1 Word document to Melissa Sears by November 1. The residents who are selected to write the column for the upcoming year will be notified by November 15.
We look forward to continuing to educate dermatology residents on topics that are most important to them!
The impact of tuition-free medical education
On Aug.16, the New York University School of Medicine announced it was offering full-tuition scholarships to all current and future students in its MD Program, regardless of need or merit – and that this policy also would apply to all matriculating students for the remainder of their medical school education at NYU.
This bold initiative, they stated, was being instituted to simultaneously address the rising costs of medical education while still attracting the best and brightest to careers in medicine. In doing so, NYU School of Medicine (at which I hold a faculty appointment) became the first Top 10–ranked medical school in the nation to do so.
The symbolism of this announcement was noticeable: It was made at the medical school’s annual white coat ceremony, when each new student is presented with a white lab coat to mark the beginning of their medical education and training.
I count myself among many medical professionals and, indeed, others outside of medicine who have long advocated for free tuition for medical education, at the very least for those who have little or no means to pay. This particularly painful burden of debt often serves as a deterrent to many individuals who are considering a career in medicine or medical research.
According to the Association of American Medical Colleges,
What might develop as a result of NYU’s decision and that of several other schools who have adopted debt-reduction policies?
First, that these programs might have a ripple effect at other medical schools, and create a movement for more students to earn a medical degree without incurring a crushing financial burden. Some other schools, like Columbia University, already have taken steps, such as replacing all student loans with scholarships creating a “debt-free” medical school. It would, indeed, be a powerful message if other schools developed similar creative solutions to this problem.
Second, there is hope that debt relief will encourage more medical school graduates to pursue careers in such specialties as family medicine, psychiatry, pediatrics, and geriatrics – because they will not have the additional financial pressure to pursue careers in more lucrative specialties in order to pay off debt. While many medical school graduates point to other issues like complex reimbursement as a greater deterrent to a specific specialty choice, I certainly hope that debt relief will have a positive effect in shifting the subspecialty paradigm.
Third, these actions might incentivize the federal government to establish an AmeriCorps-type program, in which the cost of a medical education is covered in return for a commitment to practice medicine for a period of time in underserved areas of the country. Such an approach also might motivate more medical school graduates to pursue careers in primary care specialties and help address some of the ongoing concerns related to the uneven distribution of physicians in the United States.
Another issue that often comes up is the impact of debt burden on “burnout” among medical students. This is a complex subject – and one that actually affects physicians beyond medical school and into residency training and medical practice.
There is no doubt that debt weighs heavily on the minds of medical students – and many enter medical school having sustained significant debt already from previous education in colleges and universities. However, the causes and influences on burnout in training are multifactorial. Earlier in medical school, the impact of debt obligation may be less apparent because of the other challenges students face when beginning medical school.
However, once trainees begin residency and fellowship training – and especially during early career years when many are beginning to have families – concerns about increasing financial strain become even more prominent. For many young physicians, already stressed by other extenuating factors, it would be a tremendous relief not to have that debt pursuing them.
As someone who teaches and mentors medical students and residents, I firmly believe that most students pursue a career in medicine for altruistic reasons: to help cure illness and take care of patients, to make new scientific discoveries, and to train the next generation of physicians who will follow them into the medical profession. Unfortunately, outside economic influences – such as increased competition among health care systems, shrinking reimbursements, loss of joy and meaning in medicine, increasing isolation of the caregiver from the patient and the significant cost of a medical education – lead many physicians to burn out prematurely and, for some, to leave the profession altogether.
By eliminating medical school debt, we can remove one of these constraints and make the practice of medicine as rewarding and gratifying as it has been in the past, and more accessible to those who truly wish to care for others.
Dr. Bernstein is a professor in the departments of psychiatry and neurology at New York University and a past president of the American Psychiatric Association.
On Aug.16, the New York University School of Medicine announced it was offering full-tuition scholarships to all current and future students in its MD Program, regardless of need or merit – and that this policy also would apply to all matriculating students for the remainder of their medical school education at NYU.
This bold initiative, they stated, was being instituted to simultaneously address the rising costs of medical education while still attracting the best and brightest to careers in medicine. In doing so, NYU School of Medicine (at which I hold a faculty appointment) became the first Top 10–ranked medical school in the nation to do so.
The symbolism of this announcement was noticeable: It was made at the medical school’s annual white coat ceremony, when each new student is presented with a white lab coat to mark the beginning of their medical education and training.
I count myself among many medical professionals and, indeed, others outside of medicine who have long advocated for free tuition for medical education, at the very least for those who have little or no means to pay. This particularly painful burden of debt often serves as a deterrent to many individuals who are considering a career in medicine or medical research.
According to the Association of American Medical Colleges,
What might develop as a result of NYU’s decision and that of several other schools who have adopted debt-reduction policies?
First, that these programs might have a ripple effect at other medical schools, and create a movement for more students to earn a medical degree without incurring a crushing financial burden. Some other schools, like Columbia University, already have taken steps, such as replacing all student loans with scholarships creating a “debt-free” medical school. It would, indeed, be a powerful message if other schools developed similar creative solutions to this problem.
Second, there is hope that debt relief will encourage more medical school graduates to pursue careers in such specialties as family medicine, psychiatry, pediatrics, and geriatrics – because they will not have the additional financial pressure to pursue careers in more lucrative specialties in order to pay off debt. While many medical school graduates point to other issues like complex reimbursement as a greater deterrent to a specific specialty choice, I certainly hope that debt relief will have a positive effect in shifting the subspecialty paradigm.
Third, these actions might incentivize the federal government to establish an AmeriCorps-type program, in which the cost of a medical education is covered in return for a commitment to practice medicine for a period of time in underserved areas of the country. Such an approach also might motivate more medical school graduates to pursue careers in primary care specialties and help address some of the ongoing concerns related to the uneven distribution of physicians in the United States.
Another issue that often comes up is the impact of debt burden on “burnout” among medical students. This is a complex subject – and one that actually affects physicians beyond medical school and into residency training and medical practice.
There is no doubt that debt weighs heavily on the minds of medical students – and many enter medical school having sustained significant debt already from previous education in colleges and universities. However, the causes and influences on burnout in training are multifactorial. Earlier in medical school, the impact of debt obligation may be less apparent because of the other challenges students face when beginning medical school.
However, once trainees begin residency and fellowship training – and especially during early career years when many are beginning to have families – concerns about increasing financial strain become even more prominent. For many young physicians, already stressed by other extenuating factors, it would be a tremendous relief not to have that debt pursuing them.
As someone who teaches and mentors medical students and residents, I firmly believe that most students pursue a career in medicine for altruistic reasons: to help cure illness and take care of patients, to make new scientific discoveries, and to train the next generation of physicians who will follow them into the medical profession. Unfortunately, outside economic influences – such as increased competition among health care systems, shrinking reimbursements, loss of joy and meaning in medicine, increasing isolation of the caregiver from the patient and the significant cost of a medical education – lead many physicians to burn out prematurely and, for some, to leave the profession altogether.
By eliminating medical school debt, we can remove one of these constraints and make the practice of medicine as rewarding and gratifying as it has been in the past, and more accessible to those who truly wish to care for others.
Dr. Bernstein is a professor in the departments of psychiatry and neurology at New York University and a past president of the American Psychiatric Association.
On Aug.16, the New York University School of Medicine announced it was offering full-tuition scholarships to all current and future students in its MD Program, regardless of need or merit – and that this policy also would apply to all matriculating students for the remainder of their medical school education at NYU.
This bold initiative, they stated, was being instituted to simultaneously address the rising costs of medical education while still attracting the best and brightest to careers in medicine. In doing so, NYU School of Medicine (at which I hold a faculty appointment) became the first Top 10–ranked medical school in the nation to do so.
The symbolism of this announcement was noticeable: It was made at the medical school’s annual white coat ceremony, when each new student is presented with a white lab coat to mark the beginning of their medical education and training.
I count myself among many medical professionals and, indeed, others outside of medicine who have long advocated for free tuition for medical education, at the very least for those who have little or no means to pay. This particularly painful burden of debt often serves as a deterrent to many individuals who are considering a career in medicine or medical research.
According to the Association of American Medical Colleges,
What might develop as a result of NYU’s decision and that of several other schools who have adopted debt-reduction policies?
First, that these programs might have a ripple effect at other medical schools, and create a movement for more students to earn a medical degree without incurring a crushing financial burden. Some other schools, like Columbia University, already have taken steps, such as replacing all student loans with scholarships creating a “debt-free” medical school. It would, indeed, be a powerful message if other schools developed similar creative solutions to this problem.
Second, there is hope that debt relief will encourage more medical school graduates to pursue careers in such specialties as family medicine, psychiatry, pediatrics, and geriatrics – because they will not have the additional financial pressure to pursue careers in more lucrative specialties in order to pay off debt. While many medical school graduates point to other issues like complex reimbursement as a greater deterrent to a specific specialty choice, I certainly hope that debt relief will have a positive effect in shifting the subspecialty paradigm.
Third, these actions might incentivize the federal government to establish an AmeriCorps-type program, in which the cost of a medical education is covered in return for a commitment to practice medicine for a period of time in underserved areas of the country. Such an approach also might motivate more medical school graduates to pursue careers in primary care specialties and help address some of the ongoing concerns related to the uneven distribution of physicians in the United States.
Another issue that often comes up is the impact of debt burden on “burnout” among medical students. This is a complex subject – and one that actually affects physicians beyond medical school and into residency training and medical practice.
There is no doubt that debt weighs heavily on the minds of medical students – and many enter medical school having sustained significant debt already from previous education in colleges and universities. However, the causes and influences on burnout in training are multifactorial. Earlier in medical school, the impact of debt obligation may be less apparent because of the other challenges students face when beginning medical school.
However, once trainees begin residency and fellowship training – and especially during early career years when many are beginning to have families – concerns about increasing financial strain become even more prominent. For many young physicians, already stressed by other extenuating factors, it would be a tremendous relief not to have that debt pursuing them.
As someone who teaches and mentors medical students and residents, I firmly believe that most students pursue a career in medicine for altruistic reasons: to help cure illness and take care of patients, to make new scientific discoveries, and to train the next generation of physicians who will follow them into the medical profession. Unfortunately, outside economic influences – such as increased competition among health care systems, shrinking reimbursements, loss of joy and meaning in medicine, increasing isolation of the caregiver from the patient and the significant cost of a medical education – lead many physicians to burn out prematurely and, for some, to leave the profession altogether.
By eliminating medical school debt, we can remove one of these constraints and make the practice of medicine as rewarding and gratifying as it has been in the past, and more accessible to those who truly wish to care for others.
Dr. Bernstein is a professor in the departments of psychiatry and neurology at New York University and a past president of the American Psychiatric Association.
Longitudinal melanonychia: the good, the bad, and the confusing
CHICAGO – A discolored nail can give even seasoned dermatologists pause: Is the cause exogenous? Fungal or bacterial, perhaps? Could it be a subungual melanoma? Should it be followed, clipped, or biopsied? of the American Academy of Dermatology summer meeting.
The session came after a recent nationwide survey performed by Dr. Lipner and her collaborators who asked dermatologists at different practice stages how confident they were in the diagnosis and management of melanonychia. “On the whole, they were not very confident at all,” said Dr. Lipner, director of the nail division at Cornell University, New York.
Of 142 dermatology residents, as well as 58 junior and 199 senior attending dermatologists, just 18.2% performed nail exams at each visit, and most (58%) only looked at nails during the total body skin exam. Over half (62%) of resident physicians reported feeling not confident about melanonychia diagnosis and management, while that figure dropped to 8.6% for senior attending physicians. Still, most senior physicians (64.3%) were just “fairly confident” in their melanonychia skills (J Am Acad Dermatol. 2017 May;76[5]:994-6).
Tools of the trade
Dermoscopy can be an invaluable tool for determining the cause of longitudinal melanonychia (LM). “Contact dermoscopy is helpful, so I always have ultrasound gel available,” Dr. Lipner said. “The gel makes the nail more of a flat surface,” which makes accurate viewing easier. Other useful tools include a double-action nail clipper, which, she said, is a worthwhile investment.
Because patients who are concerned about one of their nails will often come to their appointment with nail polish still on the other nails, Dr. Lipner always has polish remover pads available in the office. It’s important to be able to see all nails, she said, but she and her collaborators, including first author Pierre Halteh, MD, who was then a medical student at Cornell, discovered from their survey that “few physicians (32/402; 8%) asked their patients to remove nail polish at every visit.”
Nonmelanocytic causes of LM
Longitudinal melanonychias can have a nonmelanocytic etiology, which can range from subungual hematomas to pseudomonas and fungal infections to exogenous pigment.
Overall, subungual hematomas are the most common cause of melanonychia, although longitudinal hematomas are not commonly seen. The more remote the causative trauma, the darker the subungual discoloration, Dr. Lipner said. “Dermoscopy is very helpful” for subungual hematomas, which will usually show a homogeneous pattern, although “you can also see peripheral fadings, streaks, and periungual hemorrhages,” she added.
It is important to monitor these patients “because melanomas can bleed,” she said. In-office photography, or even pictures taken by patients, can be used to track the hematoma to resolution.
When thinking about exogenous sources of pigment, in addition to clues from the history, a tip-off can be that the proximal nail fold is also discolored, Dr. Lipner pointed out. A wide variety of common and less-common culprits may crop up, including from tar, tobacco, henna and other hair dyes, potassium permanganate, and even newspaper print, she said. With an exogenous source, careful clinical and dermoscopic examination may show that the pigment does not extend all the way proximally to the lunula, although it may follow the outline of the proximal nail fold.
When fungus is the cause of LM, the band is often wider proximally and tapers distally, Dr. Lipner noted. While Trichophyton rubrum var. nigricans is a known culprit, nondermatophytes, such as Neoscytalidium dimidiatum, can also cause an LM that often runs along the proximal and lateral nail folds. “To make the diagnosis, sending a clipping to the dermatopathologist is helpful,” she said. Hyphae can often be seen on staining and culture, she said. Polymerase chain reaction “is also possible and very helpful for these nondermatophytes.”
Bacterial colonization of the nail bed can be a cause of LM. Pathogens can include Pseudomonas aeruginosa, which will often show the characteristic greenish tint. Klebsiella and Proteus species may result in more of a grayish-black discoloration. A history of wet work, such as farming and other agricultural and dairy occupations, as well as housekeeping work, increases the risk for bacterial colonization.
Commonly, a bacterial etiology will result in discoloration beginning at the lateral nail fold or at the juncture of the proximal and lateral nail folds. Dermoscopy will show irregular fading of the discoloration toward the medial aspect of the nail, and gram staining of affected clippings will show gram-negative rods.
Melanocytic causes of longitudinal melanonychia
The melanotic macule, sometimes called melanocytic activation, is the most common subtype of melanin-derived LM in adults, Dr. Lipner said. This benign condition results from increased melanin synthesis without an increase in the number of melanocytes, which will be evident on histopathologic examination of the nail bed. Any of a variety of triggers can provoke the increased pigment, which can range from endocrine disruptions to inflammatory conditions, such as psoriasis, to trauma (including nail biting or habit tics).
Pregnancy, normal ethnic variation, and chemotherapy administration are all also associated with melanotic macules. In any case, dermoscopy will show an LM characterized by a grayish background that contains darker grayish lines.
Melanocyte hyperplasia can also cause melanonychia, in which case the trick is sorting out which cases are benign and which are malignant, Dr. Lipner noted. And getting the diagnosis right in a timely fashion matters: “Ideally, we want to catch these melanomas in in situ stages where we can preserve the digit,” she said. “It’s been shown that there is no survival benefit for amputation versus en bloc excision for nail melanomas in situ.”
Nail matrix nevi are the most common cause of LM in children, Dr. Lipner said. Here, dermoscopy shows a brown background with brown lines, with regular color, thickness, and spacing.
On examination of a nail with a melanoma, “typically, we see features suggestive of melanoma but really no pathognomonic features,” she commented. Some signs that should prompt concern and a more thorough investigation, she said, include a dark brown or black band of LM; lack of homogeneity, such as the presence of lines of different colors; blurring of the borders of the pigmentation; and a triangular or wavering outline. Changes in the nail, such as fissuring or splitting, also are worrying, as is any associated discoloration of the periungual skin.
Dermoscopy may confirm the irregularity of the pigmentation pattern and show irregularly colored and spaced lines of varying thicknesses within the pigmented band. An LM caused by melanoma may also be marked by loss of parallelism within the pigmented band.
She pointed out that these concerning characteristics have been encapsulated in a mnemonic, first created in 2000, that’s meant to mirror the ABCDs of nonnail melanoma detection (J Am Acad Dermatol. Feb 2000;42[2 Pt 1]:269-74). Her survey found that overall, just one in four (24.8%) of respondents knew of the mnemonic for subungual melanomas.
Dr. Lipner reported that she has received research support from MOE Medical Devices and has served as a consultant to BAKO Therapeutics.
SOURCE: Lipner S. Summer AAD 2018, Presentation F004.
CHICAGO – A discolored nail can give even seasoned dermatologists pause: Is the cause exogenous? Fungal or bacterial, perhaps? Could it be a subungual melanoma? Should it be followed, clipped, or biopsied? of the American Academy of Dermatology summer meeting.
The session came after a recent nationwide survey performed by Dr. Lipner and her collaborators who asked dermatologists at different practice stages how confident they were in the diagnosis and management of melanonychia. “On the whole, they were not very confident at all,” said Dr. Lipner, director of the nail division at Cornell University, New York.
Of 142 dermatology residents, as well as 58 junior and 199 senior attending dermatologists, just 18.2% performed nail exams at each visit, and most (58%) only looked at nails during the total body skin exam. Over half (62%) of resident physicians reported feeling not confident about melanonychia diagnosis and management, while that figure dropped to 8.6% for senior attending physicians. Still, most senior physicians (64.3%) were just “fairly confident” in their melanonychia skills (J Am Acad Dermatol. 2017 May;76[5]:994-6).
Tools of the trade

Dermoscopy can be an invaluable tool for determining the cause of longitudinal melanonychia (LM). “Contact dermoscopy is helpful, so I always have ultrasound gel available,” Dr. Lipner said. “The gel makes the nail more of a flat surface,” which makes accurate viewing easier. Other useful tools include a double-action nail clipper, which, she said, is a worthwhile investment.
Because patients who are concerned about one of their nails will often come to their appointment with nail polish still on the other nails, Dr. Lipner always has polish remover pads available in the office. It’s important to be able to see all nails, she said, but she and her collaborators, including first author Pierre Halteh, MD, who was then a medical student at Cornell, discovered from their survey that “few physicians (32/402; 8%) asked their patients to remove nail polish at every visit.”
Nonmelanocytic causes of LM
Longitudinal melanonychias can have a nonmelanocytic etiology, which can range from subungual hematomas to pseudomonas and fungal infections to exogenous pigment.
Overall, subungual hematomas are the most common cause of melanonychia, although longitudinal hematomas are not commonly seen. The more remote the causative trauma, the darker the subungual discoloration, Dr. Lipner said. “Dermoscopy is very helpful” for subungual hematomas, which will usually show a homogeneous pattern, although “you can also see peripheral fadings, streaks, and periungual hemorrhages,” she added.
It is important to monitor these patients “because melanomas can bleed,” she said. In-office photography, or even pictures taken by patients, can be used to track the hematoma to resolution.
When thinking about exogenous sources of pigment, in addition to clues from the history, a tip-off can be that the proximal nail fold is also discolored, Dr. Lipner pointed out. A wide variety of common and less-common culprits may crop up, including from tar, tobacco, henna and other hair dyes, potassium permanganate, and even newspaper print, she said. With an exogenous source, careful clinical and dermoscopic examination may show that the pigment does not extend all the way proximally to the lunula, although it may follow the outline of the proximal nail fold.
When fungus is the cause of LM, the band is often wider proximally and tapers distally, Dr. Lipner noted. While Trichophyton rubrum var. nigricans is a known culprit, nondermatophytes, such as Neoscytalidium dimidiatum, can also cause an LM that often runs along the proximal and lateral nail folds. “To make the diagnosis, sending a clipping to the dermatopathologist is helpful,” she said. Hyphae can often be seen on staining and culture, she said. Polymerase chain reaction “is also possible and very helpful for these nondermatophytes.”
Bacterial colonization of the nail bed can be a cause of LM. Pathogens can include Pseudomonas aeruginosa, which will often show the characteristic greenish tint. Klebsiella and Proteus species may result in more of a grayish-black discoloration. A history of wet work, such as farming and other agricultural and dairy occupations, as well as housekeeping work, increases the risk for bacterial colonization.
Commonly, a bacterial etiology will result in discoloration beginning at the lateral nail fold or at the juncture of the proximal and lateral nail folds. Dermoscopy will show irregular fading of the discoloration toward the medial aspect of the nail, and gram staining of affected clippings will show gram-negative rods.
Melanocytic causes of longitudinal melanonychia
The melanotic macule, sometimes called melanocytic activation, is the most common subtype of melanin-derived LM in adults, Dr. Lipner said. This benign condition results from increased melanin synthesis without an increase in the number of melanocytes, which will be evident on histopathologic examination of the nail bed. Any of a variety of triggers can provoke the increased pigment, which can range from endocrine disruptions to inflammatory conditions, such as psoriasis, to trauma (including nail biting or habit tics).
Pregnancy, normal ethnic variation, and chemotherapy administration are all also associated with melanotic macules. In any case, dermoscopy will show an LM characterized by a grayish background that contains darker grayish lines.
Melanocyte hyperplasia can also cause melanonychia, in which case the trick is sorting out which cases are benign and which are malignant, Dr. Lipner noted. And getting the diagnosis right in a timely fashion matters: “Ideally, we want to catch these melanomas in in situ stages where we can preserve the digit,” she said. “It’s been shown that there is no survival benefit for amputation versus en bloc excision for nail melanomas in situ.”
Nail matrix nevi are the most common cause of LM in children, Dr. Lipner said. Here, dermoscopy shows a brown background with brown lines, with regular color, thickness, and spacing.
On examination of a nail with a melanoma, “typically, we see features suggestive of melanoma but really no pathognomonic features,” she commented. Some signs that should prompt concern and a more thorough investigation, she said, include a dark brown or black band of LM; lack of homogeneity, such as the presence of lines of different colors; blurring of the borders of the pigmentation; and a triangular or wavering outline. Changes in the nail, such as fissuring or splitting, also are worrying, as is any associated discoloration of the periungual skin.
Dermoscopy may confirm the irregularity of the pigmentation pattern and show irregularly colored and spaced lines of varying thicknesses within the pigmented band. An LM caused by melanoma may also be marked by loss of parallelism within the pigmented band.
She pointed out that these concerning characteristics have been encapsulated in a mnemonic, first created in 2000, that’s meant to mirror the ABCDs of nonnail melanoma detection (J Am Acad Dermatol. Feb 2000;42[2 Pt 1]:269-74). Her survey found that overall, just one in four (24.8%) of respondents knew of the mnemonic for subungual melanomas.
Dr. Lipner reported that she has received research support from MOE Medical Devices and has served as a consultant to BAKO Therapeutics.
SOURCE: Lipner S. Summer AAD 2018, Presentation F004.
CHICAGO – A discolored nail can give even seasoned dermatologists pause: Is the cause exogenous? Fungal or bacterial, perhaps? Could it be a subungual melanoma? Should it be followed, clipped, or biopsied? of the American Academy of Dermatology summer meeting.
The session came after a recent nationwide survey performed by Dr. Lipner and her collaborators who asked dermatologists at different practice stages how confident they were in the diagnosis and management of melanonychia. “On the whole, they were not very confident at all,” said Dr. Lipner, director of the nail division at Cornell University, New York.
Of 142 dermatology residents, as well as 58 junior and 199 senior attending dermatologists, just 18.2% performed nail exams at each visit, and most (58%) only looked at nails during the total body skin exam. Over half (62%) of resident physicians reported feeling not confident about melanonychia diagnosis and management, while that figure dropped to 8.6% for senior attending physicians. Still, most senior physicians (64.3%) were just “fairly confident” in their melanonychia skills (J Am Acad Dermatol. 2017 May;76[5]:994-6).
Tools of the trade

Dermoscopy can be an invaluable tool for determining the cause of longitudinal melanonychia (LM). “Contact dermoscopy is helpful, so I always have ultrasound gel available,” Dr. Lipner said. “The gel makes the nail more of a flat surface,” which makes accurate viewing easier. Other useful tools include a double-action nail clipper, which, she said, is a worthwhile investment.
Because patients who are concerned about one of their nails will often come to their appointment with nail polish still on the other nails, Dr. Lipner always has polish remover pads available in the office. It’s important to be able to see all nails, she said, but she and her collaborators, including first author Pierre Halteh, MD, who was then a medical student at Cornell, discovered from their survey that “few physicians (32/402; 8%) asked their patients to remove nail polish at every visit.”
Nonmelanocytic causes of LM
Longitudinal melanonychias can have a nonmelanocytic etiology, which can range from subungual hematomas to pseudomonas and fungal infections to exogenous pigment.
Overall, subungual hematomas are the most common cause of melanonychia, although longitudinal hematomas are not commonly seen. The more remote the causative trauma, the darker the subungual discoloration, Dr. Lipner said. “Dermoscopy is very helpful” for subungual hematomas, which will usually show a homogeneous pattern, although “you can also see peripheral fadings, streaks, and periungual hemorrhages,” she added.
It is important to monitor these patients “because melanomas can bleed,” she said. In-office photography, or even pictures taken by patients, can be used to track the hematoma to resolution.
When thinking about exogenous sources of pigment, in addition to clues from the history, a tip-off can be that the proximal nail fold is also discolored, Dr. Lipner pointed out. A wide variety of common and less-common culprits may crop up, including from tar, tobacco, henna and other hair dyes, potassium permanganate, and even newspaper print, she said. With an exogenous source, careful clinical and dermoscopic examination may show that the pigment does not extend all the way proximally to the lunula, although it may follow the outline of the proximal nail fold.
When fungus is the cause of LM, the band is often wider proximally and tapers distally, Dr. Lipner noted. While Trichophyton rubrum var. nigricans is a known culprit, nondermatophytes, such as Neoscytalidium dimidiatum, can also cause an LM that often runs along the proximal and lateral nail folds. “To make the diagnosis, sending a clipping to the dermatopathologist is helpful,” she said. Hyphae can often be seen on staining and culture, she said. Polymerase chain reaction “is also possible and very helpful for these nondermatophytes.”
Bacterial colonization of the nail bed can be a cause of LM. Pathogens can include Pseudomonas aeruginosa, which will often show the characteristic greenish tint. Klebsiella and Proteus species may result in more of a grayish-black discoloration. A history of wet work, such as farming and other agricultural and dairy occupations, as well as housekeeping work, increases the risk for bacterial colonization.
Commonly, a bacterial etiology will result in discoloration beginning at the lateral nail fold or at the juncture of the proximal and lateral nail folds. Dermoscopy will show irregular fading of the discoloration toward the medial aspect of the nail, and gram staining of affected clippings will show gram-negative rods.
Melanocytic causes of longitudinal melanonychia
The melanotic macule, sometimes called melanocytic activation, is the most common subtype of melanin-derived LM in adults, Dr. Lipner said. This benign condition results from increased melanin synthesis without an increase in the number of melanocytes, which will be evident on histopathologic examination of the nail bed. Any of a variety of triggers can provoke the increased pigment, which can range from endocrine disruptions to inflammatory conditions, such as psoriasis, to trauma (including nail biting or habit tics).
Pregnancy, normal ethnic variation, and chemotherapy administration are all also associated with melanotic macules. In any case, dermoscopy will show an LM characterized by a grayish background that contains darker grayish lines.
Melanocyte hyperplasia can also cause melanonychia, in which case the trick is sorting out which cases are benign and which are malignant, Dr. Lipner noted. And getting the diagnosis right in a timely fashion matters: “Ideally, we want to catch these melanomas in in situ stages where we can preserve the digit,” she said. “It’s been shown that there is no survival benefit for amputation versus en bloc excision for nail melanomas in situ.”
Nail matrix nevi are the most common cause of LM in children, Dr. Lipner said. Here, dermoscopy shows a brown background with brown lines, with regular color, thickness, and spacing.
On examination of a nail with a melanoma, “typically, we see features suggestive of melanoma but really no pathognomonic features,” she commented. Some signs that should prompt concern and a more thorough investigation, she said, include a dark brown or black band of LM; lack of homogeneity, such as the presence of lines of different colors; blurring of the borders of the pigmentation; and a triangular or wavering outline. Changes in the nail, such as fissuring or splitting, also are worrying, as is any associated discoloration of the periungual skin.
Dermoscopy may confirm the irregularity of the pigmentation pattern and show irregularly colored and spaced lines of varying thicknesses within the pigmented band. An LM caused by melanoma may also be marked by loss of parallelism within the pigmented band.
She pointed out that these concerning characteristics have been encapsulated in a mnemonic, first created in 2000, that’s meant to mirror the ABCDs of nonnail melanoma detection (J Am Acad Dermatol. Feb 2000;42[2 Pt 1]:269-74). Her survey found that overall, just one in four (24.8%) of respondents knew of the mnemonic for subungual melanomas.
Dr. Lipner reported that she has received research support from MOE Medical Devices and has served as a consultant to BAKO Therapeutics.
SOURCE: Lipner S. Summer AAD 2018, Presentation F004.
EXPERT ANALYSIS FROM SUMMER AAD 2018