User login
Anxiety sensitivity fuels depression in dissociative identity disorder
Anxiety sensitivity refers to fear of the signs and symptoms of anxiety based on the individual’s belief that the signs of anxiety will have harmful consequences, wrote Xi Pan, LICSW, MPA, of McLean Hospital, Belmont, Mass., and colleagues.
Anxiety sensitivity can include cognitive, physical, and social elements: for example, fear that the inability to focus signals mental illness, fear that a racing heart might cause a heart attack, or fear that exhibiting anxiety signs in public (e.g., sweaty palms) will cause embarrassment, the researchers said.
Previous studies have found associations between anxiety sensitivity and panic attacks, and anxiety sensitivity has been shown to contribute to worsening symptoms in patients with anxiety disorders, depressive disorders, and trauma-related disorders such as posttraumatic stress disorder. However, “anxiety sensitivity has not been studied in individuals with complex dissociative disorders such as dissociative identity disorder (DID)” – who often have co-occurring PTSD and depression, the researchers said.
In a study published in the Journal of Psychiatric Research, the authors analyzed data from 21 treatment-seeking adult women with histories of childhood trauma, current PTSD, and dissociative identity disorder. Participants completed the Anxiety Sensitivity Index (ASI), Beck Depression Inventory-II, Childhood Trauma Questionnaire, Multidimensional Inventory of Dissociation, and PTSD Checklist for DSM-5.
Anxiety sensitivity in cognitive, physical, and social domains was assessed using ASI subscales.
Pearson correlations showed that symptoms of depression were significantly associated with anxiety sensitivity total scores and across all anxiety subscales. However, no direct associations appeared between anxiety sensitivity and PTSD or severe dissociative symptoms.
In a multiple regression analysis, the ASI cognitive subscale was a positive predictor of depressive symptoms, although physical and social subscale scores were not.
The researchers also tested for an indirect relationship between anxiety sensitivity and dissociative symptoms through depression. “Specifically, more severe ASI cognitive concerns were associated with more depressive symptoms, and more depressive symptoms predicted more severe pathological dissociation symptoms,” they wrote.
The findings were limited by the inability to show a direct causal relationship between anxiety sensitivity and depression, the researchers noted. Other limitations included the small sample size, use of self-reports, and the population of mainly White women, which may not generalize to other populations, they said.
However, the results represent the first empirical investigation of the relationship between anxiety sensitivity and DID symptoms, and support the value of assessment for anxiety sensitivity in DID patients in clinical practice, they said.
“If high levels of anxiety sensitivity are identified, the individual may benefit from targeted interventions, which in turn may alleviate some symptoms of depression and dissociation in DID,” the researchers concluded.
The study was supported by the National Institute of Mental Health and the Julia Kasparian Fund for Neuroscience Research. The researchers had no financial conflicts to disclose.
Anxiety sensitivity refers to fear of the signs and symptoms of anxiety based on the individual’s belief that the signs of anxiety will have harmful consequences, wrote Xi Pan, LICSW, MPA, of McLean Hospital, Belmont, Mass., and colleagues.
Anxiety sensitivity can include cognitive, physical, and social elements: for example, fear that the inability to focus signals mental illness, fear that a racing heart might cause a heart attack, or fear that exhibiting anxiety signs in public (e.g., sweaty palms) will cause embarrassment, the researchers said.
Previous studies have found associations between anxiety sensitivity and panic attacks, and anxiety sensitivity has been shown to contribute to worsening symptoms in patients with anxiety disorders, depressive disorders, and trauma-related disorders such as posttraumatic stress disorder. However, “anxiety sensitivity has not been studied in individuals with complex dissociative disorders such as dissociative identity disorder (DID)” – who often have co-occurring PTSD and depression, the researchers said.
In a study published in the Journal of Psychiatric Research, the authors analyzed data from 21 treatment-seeking adult women with histories of childhood trauma, current PTSD, and dissociative identity disorder. Participants completed the Anxiety Sensitivity Index (ASI), Beck Depression Inventory-II, Childhood Trauma Questionnaire, Multidimensional Inventory of Dissociation, and PTSD Checklist for DSM-5.
Anxiety sensitivity in cognitive, physical, and social domains was assessed using ASI subscales.
Pearson correlations showed that symptoms of depression were significantly associated with anxiety sensitivity total scores and across all anxiety subscales. However, no direct associations appeared between anxiety sensitivity and PTSD or severe dissociative symptoms.
In a multiple regression analysis, the ASI cognitive subscale was a positive predictor of depressive symptoms, although physical and social subscale scores were not.
The researchers also tested for an indirect relationship between anxiety sensitivity and dissociative symptoms through depression. “Specifically, more severe ASI cognitive concerns were associated with more depressive symptoms, and more depressive symptoms predicted more severe pathological dissociation symptoms,” they wrote.
The findings were limited by the inability to show a direct causal relationship between anxiety sensitivity and depression, the researchers noted. Other limitations included the small sample size, use of self-reports, and the population of mainly White women, which may not generalize to other populations, they said.
However, the results represent the first empirical investigation of the relationship between anxiety sensitivity and DID symptoms, and support the value of assessment for anxiety sensitivity in DID patients in clinical practice, they said.
“If high levels of anxiety sensitivity are identified, the individual may benefit from targeted interventions, which in turn may alleviate some symptoms of depression and dissociation in DID,” the researchers concluded.
The study was supported by the National Institute of Mental Health and the Julia Kasparian Fund for Neuroscience Research. The researchers had no financial conflicts to disclose.
Anxiety sensitivity refers to fear of the signs and symptoms of anxiety based on the individual’s belief that the signs of anxiety will have harmful consequences, wrote Xi Pan, LICSW, MPA, of McLean Hospital, Belmont, Mass., and colleagues.
Anxiety sensitivity can include cognitive, physical, and social elements: for example, fear that the inability to focus signals mental illness, fear that a racing heart might cause a heart attack, or fear that exhibiting anxiety signs in public (e.g., sweaty palms) will cause embarrassment, the researchers said.
Previous studies have found associations between anxiety sensitivity and panic attacks, and anxiety sensitivity has been shown to contribute to worsening symptoms in patients with anxiety disorders, depressive disorders, and trauma-related disorders such as posttraumatic stress disorder. However, “anxiety sensitivity has not been studied in individuals with complex dissociative disorders such as dissociative identity disorder (DID)” – who often have co-occurring PTSD and depression, the researchers said.
In a study published in the Journal of Psychiatric Research, the authors analyzed data from 21 treatment-seeking adult women with histories of childhood trauma, current PTSD, and dissociative identity disorder. Participants completed the Anxiety Sensitivity Index (ASI), Beck Depression Inventory-II, Childhood Trauma Questionnaire, Multidimensional Inventory of Dissociation, and PTSD Checklist for DSM-5.
Anxiety sensitivity in cognitive, physical, and social domains was assessed using ASI subscales.
Pearson correlations showed that symptoms of depression were significantly associated with anxiety sensitivity total scores and across all anxiety subscales. However, no direct associations appeared between anxiety sensitivity and PTSD or severe dissociative symptoms.
In a multiple regression analysis, the ASI cognitive subscale was a positive predictor of depressive symptoms, although physical and social subscale scores were not.
The researchers also tested for an indirect relationship between anxiety sensitivity and dissociative symptoms through depression. “Specifically, more severe ASI cognitive concerns were associated with more depressive symptoms, and more depressive symptoms predicted more severe pathological dissociation symptoms,” they wrote.
The findings were limited by the inability to show a direct causal relationship between anxiety sensitivity and depression, the researchers noted. Other limitations included the small sample size, use of self-reports, and the population of mainly White women, which may not generalize to other populations, they said.
However, the results represent the first empirical investigation of the relationship between anxiety sensitivity and DID symptoms, and support the value of assessment for anxiety sensitivity in DID patients in clinical practice, they said.
“If high levels of anxiety sensitivity are identified, the individual may benefit from targeted interventions, which in turn may alleviate some symptoms of depression and dissociation in DID,” the researchers concluded.
The study was supported by the National Institute of Mental Health and the Julia Kasparian Fund for Neuroscience Research. The researchers had no financial conflicts to disclose.
FROM THE JOURNAL OF PSYCHIATRIC RESEARCH
Borderline patients have longer time to depression remission
Major depressive episodes (MDEs) occur in major depressive disorder (MDD) and bipolar disorder (BD), John J. Söderholm, MD, of the University of Helsinki and colleagues wrote. Borderline personality disorder (BPD) includes an increased risk for depression, but data on the relationship between BPD symptoms and depressive illness are limited. In particular, they noted “a lack of studies prospectively comparing the presence of (hypo)manic symptoms over time during the recovery process from MDE between MDD, MDE/BD, and MDE/BPD patients.”
In a cohort study published in the Journal of Affective Disorders, the researchers collected data from 39 adult MDE patients with MDD, 33 with BD, and 23 with BPD. The patients were diagnosed with MDE using the SCID-I/P and SCID-II interviews, mixed symptoms were identified using the Mix-MDE scale, and borderline symptoms were identified using the Borderline Personality Disorder Severity Index.
Over a 6-month follow-up period, the participants completed biweekly online assessments. The primary outcomes were time to first full remission of symptoms and duration and nature of mood episodes.
Overall, the mean number of distinct mood states was 5.75, and the median duration was 60.9 days. When identified by subcohorts, the median number of mood state periods for MDD, BD, and BPD was 4.49, 8.05, and 4.67, respectively. The median durations were 69.2 days, 40.30 days, and 75.6 days, respectively.
The rates of remission for depressive symptoms were similar for MDD, MDE/BD, and MDE/BPD patients. However, MDE/BD patients had a significantly shorter time to first remission (hazard ratio, 2.44). Patients in the BPD group had a significantly longer time to first remission (HR, 0.95).
“When the cohort was divided into quintiles according to BPD feature severity, there was an approximately 1-month difference in time to first period of remission between the first and third and between the third and fifth quintiles, with longer times seen in patients with more severe BPD symptoms,” the researchers wrote.
The study findings were limited by several factors including the small sample size and short follow-up period that prevented investigation of depressive recurrence, the researchers noted. Other limitations included the lack of diagnostic blinding and variation in patients’ treatment schedules.
However, the results were strengthened by the representative samples of subjects with various disorders, the prospective and multimodal assessment of affective states, and the comparison of three patient groups in a single study.
As BPD was associated with a longer time to remission from depressive symptoms, the results suggest that BPD severity may be an indicator of more severe disease in patients with MDD in the context of depression, the researchers concluded.
The study was supported by the Finska Lakaresallskapet, the City of Helsinki, the Hospital District of Helsinki and Uusimaa, and the Finnish Psychiatric Association. The researchers had no financial conflicts to disclose.
Major depressive episodes (MDEs) occur in major depressive disorder (MDD) and bipolar disorder (BD), John J. Söderholm, MD, of the University of Helsinki and colleagues wrote. Borderline personality disorder (BPD) includes an increased risk for depression, but data on the relationship between BPD symptoms and depressive illness are limited. In particular, they noted “a lack of studies prospectively comparing the presence of (hypo)manic symptoms over time during the recovery process from MDE between MDD, MDE/BD, and MDE/BPD patients.”
In a cohort study published in the Journal of Affective Disorders, the researchers collected data from 39 adult MDE patients with MDD, 33 with BD, and 23 with BPD. The patients were diagnosed with MDE using the SCID-I/P and SCID-II interviews, mixed symptoms were identified using the Mix-MDE scale, and borderline symptoms were identified using the Borderline Personality Disorder Severity Index.
Over a 6-month follow-up period, the participants completed biweekly online assessments. The primary outcomes were time to first full remission of symptoms and duration and nature of mood episodes.
Overall, the mean number of distinct mood states was 5.75, and the median duration was 60.9 days. When identified by subcohorts, the median number of mood state periods for MDD, BD, and BPD was 4.49, 8.05, and 4.67, respectively. The median durations were 69.2 days, 40.30 days, and 75.6 days, respectively.
The rates of remission for depressive symptoms were similar for MDD, MDE/BD, and MDE/BPD patients. However, MDE/BD patients had a significantly shorter time to first remission (hazard ratio, 2.44). Patients in the BPD group had a significantly longer time to first remission (HR, 0.95).
“When the cohort was divided into quintiles according to BPD feature severity, there was an approximately 1-month difference in time to first period of remission between the first and third and between the third and fifth quintiles, with longer times seen in patients with more severe BPD symptoms,” the researchers wrote.
The study findings were limited by several factors including the small sample size and short follow-up period that prevented investigation of depressive recurrence, the researchers noted. Other limitations included the lack of diagnostic blinding and variation in patients’ treatment schedules.
However, the results were strengthened by the representative samples of subjects with various disorders, the prospective and multimodal assessment of affective states, and the comparison of three patient groups in a single study.
As BPD was associated with a longer time to remission from depressive symptoms, the results suggest that BPD severity may be an indicator of more severe disease in patients with MDD in the context of depression, the researchers concluded.
The study was supported by the Finska Lakaresallskapet, the City of Helsinki, the Hospital District of Helsinki and Uusimaa, and the Finnish Psychiatric Association. The researchers had no financial conflicts to disclose.
Major depressive episodes (MDEs) occur in major depressive disorder (MDD) and bipolar disorder (BD), John J. Söderholm, MD, of the University of Helsinki and colleagues wrote. Borderline personality disorder (BPD) includes an increased risk for depression, but data on the relationship between BPD symptoms and depressive illness are limited. In particular, they noted “a lack of studies prospectively comparing the presence of (hypo)manic symptoms over time during the recovery process from MDE between MDD, MDE/BD, and MDE/BPD patients.”
In a cohort study published in the Journal of Affective Disorders, the researchers collected data from 39 adult MDE patients with MDD, 33 with BD, and 23 with BPD. The patients were diagnosed with MDE using the SCID-I/P and SCID-II interviews, mixed symptoms were identified using the Mix-MDE scale, and borderline symptoms were identified using the Borderline Personality Disorder Severity Index.
Over a 6-month follow-up period, the participants completed biweekly online assessments. The primary outcomes were time to first full remission of symptoms and duration and nature of mood episodes.
Overall, the mean number of distinct mood states was 5.75, and the median duration was 60.9 days. When identified by subcohorts, the median number of mood state periods for MDD, BD, and BPD was 4.49, 8.05, and 4.67, respectively. The median durations were 69.2 days, 40.30 days, and 75.6 days, respectively.
The rates of remission for depressive symptoms were similar for MDD, MDE/BD, and MDE/BPD patients. However, MDE/BD patients had a significantly shorter time to first remission (hazard ratio, 2.44). Patients in the BPD group had a significantly longer time to first remission (HR, 0.95).
“When the cohort was divided into quintiles according to BPD feature severity, there was an approximately 1-month difference in time to first period of remission between the first and third and between the third and fifth quintiles, with longer times seen in patients with more severe BPD symptoms,” the researchers wrote.
The study findings were limited by several factors including the small sample size and short follow-up period that prevented investigation of depressive recurrence, the researchers noted. Other limitations included the lack of diagnostic blinding and variation in patients’ treatment schedules.
However, the results were strengthened by the representative samples of subjects with various disorders, the prospective and multimodal assessment of affective states, and the comparison of three patient groups in a single study.
As BPD was associated with a longer time to remission from depressive symptoms, the results suggest that BPD severity may be an indicator of more severe disease in patients with MDD in the context of depression, the researchers concluded.
The study was supported by the Finska Lakaresallskapet, the City of Helsinki, the Hospital District of Helsinki and Uusimaa, and the Finnish Psychiatric Association. The researchers had no financial conflicts to disclose.
FROM THE JOURNAL OF AFFECTIVE DISORDERS
Low serum LDH shows potential as depression biomarker
The pathogenesis of depression is complex, and recent research has focused on the potential relationship between energy metabolism and depression, wrote Qian Yao, MD, of Wuhan University, Hubei, China, and colleagues.
Previous studies have suggested that serum lactate dehydrogenase (LDH) may be a biomarker for Parkinson’s disease, Huntington’s disease, and post-stroke depression, but the link between lactate metabolism and depression remains unclear, they said.
“We hypothesize that LDH may act as a potential biomarker for MDD, considering it represents a reduced energy metabolic status in depressive patients,” they explained.
In a study published in General Hospital Psychiatry, the researchers examined differences in serum LDH in 232 patients with major depressive disorder (MDD) and 110 healthy controls. They also examined whether LDH was predictive of suicide attempts in the MDD patients. Depression was assessed via the 24-item Hamilton Depression Scale (HAMD-24).
The mean age across both groups was 33 years; other clinical characteristics were similar between the groups.
The serum LDH level of the MDD group was significantly lower than the control group was (177.94 U/L vs. 196.50 U/L; P < .001). Analysis of blood lipid levels showed significantly lower levels of total cholesterol in the MDD group compared with controls, but no significant differences were noted in LDL cholesterol, HDL cholesterol, or triglycerides.
In a further analysis of subgroups of depression, the serum LDH in MDD patients who had attempted suicide was significantly lower compared to those without suicide attempts (169.96 vs. 181.25; P = .002), although the LDH level for the non-suicide MDD patients also was significantly lower than controls (181.25 vs. 196.50; P < .001). No significant correlation was noted between HAMD-24 score and suicide attempts.
Some gender differences also appeared. Both male and female MDD patients had significantly lower LDH levels compared with controls. However, in a regression analysis, a correlation between total cholesterol and LDL cholesterol as potential suicide markers was noted in female MDD patients, but not male MDD patients, which suggests an impact of gender on suicide risk in MDD, the researchers wrote in their discussion.
The findings were limited by several factors including the retrospective design, lack of investigation of changes in LDH isozymes in MDD patients, and lack of assessment of changes in LDH in cerebrospinal fluid, the researchers noted. However, the results “provide clear evidence that the concentration of LDH in serum is associated with early onset and clinical prognosis of depressive symptoms,” in MDD, which may inform diagnosis and guide clinical intervention, including early identification of suicide risk, they concluded.
The study was supported by the National Natural Science Foundation of China. The researchers had no financial conflicts to disclose.
The pathogenesis of depression is complex, and recent research has focused on the potential relationship between energy metabolism and depression, wrote Qian Yao, MD, of Wuhan University, Hubei, China, and colleagues.
Previous studies have suggested that serum lactate dehydrogenase (LDH) may be a biomarker for Parkinson’s disease, Huntington’s disease, and post-stroke depression, but the link between lactate metabolism and depression remains unclear, they said.
“We hypothesize that LDH may act as a potential biomarker for MDD, considering it represents a reduced energy metabolic status in depressive patients,” they explained.
In a study published in General Hospital Psychiatry, the researchers examined differences in serum LDH in 232 patients with major depressive disorder (MDD) and 110 healthy controls. They also examined whether LDH was predictive of suicide attempts in the MDD patients. Depression was assessed via the 24-item Hamilton Depression Scale (HAMD-24).
The mean age across both groups was 33 years; other clinical characteristics were similar between the groups.
The serum LDH level of the MDD group was significantly lower than the control group was (177.94 U/L vs. 196.50 U/L; P < .001). Analysis of blood lipid levels showed significantly lower levels of total cholesterol in the MDD group compared with controls, but no significant differences were noted in LDL cholesterol, HDL cholesterol, or triglycerides.
In a further analysis of subgroups of depression, the serum LDH in MDD patients who had attempted suicide was significantly lower compared to those without suicide attempts (169.96 vs. 181.25; P = .002), although the LDH level for the non-suicide MDD patients also was significantly lower than controls (181.25 vs. 196.50; P < .001). No significant correlation was noted between HAMD-24 score and suicide attempts.
Some gender differences also appeared. Both male and female MDD patients had significantly lower LDH levels compared with controls. However, in a regression analysis, a correlation between total cholesterol and LDL cholesterol as potential suicide markers was noted in female MDD patients, but not male MDD patients, which suggests an impact of gender on suicide risk in MDD, the researchers wrote in their discussion.
The findings were limited by several factors including the retrospective design, lack of investigation of changes in LDH isozymes in MDD patients, and lack of assessment of changes in LDH in cerebrospinal fluid, the researchers noted. However, the results “provide clear evidence that the concentration of LDH in serum is associated with early onset and clinical prognosis of depressive symptoms,” in MDD, which may inform diagnosis and guide clinical intervention, including early identification of suicide risk, they concluded.
The study was supported by the National Natural Science Foundation of China. The researchers had no financial conflicts to disclose.
The pathogenesis of depression is complex, and recent research has focused on the potential relationship between energy metabolism and depression, wrote Qian Yao, MD, of Wuhan University, Hubei, China, and colleagues.
Previous studies have suggested that serum lactate dehydrogenase (LDH) may be a biomarker for Parkinson’s disease, Huntington’s disease, and post-stroke depression, but the link between lactate metabolism and depression remains unclear, they said.
“We hypothesize that LDH may act as a potential biomarker for MDD, considering it represents a reduced energy metabolic status in depressive patients,” they explained.
In a study published in General Hospital Psychiatry, the researchers examined differences in serum LDH in 232 patients with major depressive disorder (MDD) and 110 healthy controls. They also examined whether LDH was predictive of suicide attempts in the MDD patients. Depression was assessed via the 24-item Hamilton Depression Scale (HAMD-24).
The mean age across both groups was 33 years; other clinical characteristics were similar between the groups.
The serum LDH level of the MDD group was significantly lower than the control group was (177.94 U/L vs. 196.50 U/L; P < .001). Analysis of blood lipid levels showed significantly lower levels of total cholesterol in the MDD group compared with controls, but no significant differences were noted in LDL cholesterol, HDL cholesterol, or triglycerides.
In a further analysis of subgroups of depression, the serum LDH in MDD patients who had attempted suicide was significantly lower compared to those without suicide attempts (169.96 vs. 181.25; P = .002), although the LDH level for the non-suicide MDD patients also was significantly lower than controls (181.25 vs. 196.50; P < .001). No significant correlation was noted between HAMD-24 score and suicide attempts.
Some gender differences also appeared. Both male and female MDD patients had significantly lower LDH levels compared with controls. However, in a regression analysis, a correlation between total cholesterol and LDL cholesterol as potential suicide markers was noted in female MDD patients, but not male MDD patients, which suggests an impact of gender on suicide risk in MDD, the researchers wrote in their discussion.
The findings were limited by several factors including the retrospective design, lack of investigation of changes in LDH isozymes in MDD patients, and lack of assessment of changes in LDH in cerebrospinal fluid, the researchers noted. However, the results “provide clear evidence that the concentration of LDH in serum is associated with early onset and clinical prognosis of depressive symptoms,” in MDD, which may inform diagnosis and guide clinical intervention, including early identification of suicide risk, they concluded.
The study was supported by the National Natural Science Foundation of China. The researchers had no financial conflicts to disclose.
FROM GENERAL HOSPITAL PSYCHIATRY
Self-management app may boost quality of life
In a randomized clinical trial of usual care plus the experimental smartphone-based intervention known as LiveWell vs. usual care alone, participants in the smartphone group who were categorized as low-risk or in asymptomatic recovery at baseline also showed reduced manic symptom severity.
The results suggest that “apps for individuals with bipolar disorder will likely be useful for some people in managing medication use, sleep duration, routine, and monitoring for and managing signs and symptoms” of the disorder, coinvestigator Evan H. Goulding, MD, PhD, assistant professor of psychiatry and behavioral sciences, Northwestern University, Chicago, told this news organization.
Use of the app may also “lead to decreased recurrence of mood episodes, impact overall depressive and manic symptom levels, and improve some aspects of quality of life,” Dr. Goulding added.
The findings were published online in JAMA Psychiatry.
Daily check-ins
The researchers randomly assigned 205 patients with BD to receive either usual care (n = 81; 56% women; mean age, 39 years) or usual care plus the smartphone-based self-management intervention LiveWell (n = 124; 65% women; mean age, 43 years) between March 2017 and April 2020. To be included, participants could not be experiencing a current mood episode or suicidal ideation.
The smartphone intervention included a daily check-in to monitor medication adherence, sleep, and wellness levels; coach visits to support adherence to the app; six phone calls over 16 weeks; and support from mental health professionals whenever needed. Participants in this group were asked to engage their mental health providers in the intervention as well.
Each participant in the control group had a visit with a coach who facilitated self-management support.
Investigators assessed all participants every 8 weeks until week 48 to gather information on mood symptoms and severity over the past 2 weeks and on quality of life.
The patients were also stratified into high- and low-risk relapse groups. The low-risk group was in asymptomatic recovery, meaning that they experienced two or fewer moderate symptoms of mania or depression in the previous 8 weeks. In addition, they had no moderate symptoms of mania or depression at study enrollment.
Patients in the high-risk group were recovering from an episode of mania or depression. They also had two or fewer moderate symptoms, but for 8 weeks or less.
Low-risk group fares better
Results showed that the smartphone intervention was significantly associated with a reduction in depressive symptoms vs. usual care (P = .02), as well as improvement in one aspect of the World Health Organization Quality of Life Assessment that measures social relationships (P = .02).
When the investigators stratified participants into risk groups, they found that for those in the low-risk group the smartphone-based intervention was associated with lower episode-relapse rates, lower mean percentage time symptomatic, and decreased manic symptom severity.
Mean estimated relapse rates by 48 weeks for the low-risk group were 12% for those in the intervention group and 37.2% for those in the control group. No differences were noted for the high-risk group.
Low-risk patients in the intervention group also had lower mean percentage-time symptomatic (17.9%) than those in the control group (26.1%) (Cohen d = .31).
“Our results are consistent with literature emphasizing the identification and facilitation of management plans for early warning signs of mood episodes and using these plans as an important self-management technique for avoiding relapse,” Dr. Goulding said.
Study limitations included low engagement by mental health professionals and low data generalizability to other populations, as the sample was mostly White (84% of the app group and 81% of the control group).
“There is a fairly large literature on risk factors, longitudinal trajectories, and stages of diseases that suggest we should already be able to predict relapse risk for individuals,” Dr. Goulding said.
“However, moving from overall risk to individual risk is trickier and will require larger datasets with longer follow-up to better understand what types of help should be delivered when and to whom,” he added.
‘Requires commitment’
John Torous, MD, director of the division of digital psychiatry at Beth Israel Deaconess Medical Center, Boston, noted that mental health apps such as LiveWell require “time and energy devoted by both the patient and their clinician for maximal efficacy, which requires commitment from and training for both parties as well.
“But with such an investment in people, there is good evidence apps can help people with bipolar disorder even during the more severe periods of the illness,” added Dr. Torous, who was not involved with the research.
The study was funded by the National Institute of Mental Health.
Dr. Goulding reports having received honoraria from Otsuka. Dr. Torous has reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
In a randomized clinical trial of usual care plus the experimental smartphone-based intervention known as LiveWell vs. usual care alone, participants in the smartphone group who were categorized as low-risk or in asymptomatic recovery at baseline also showed reduced manic symptom severity.
The results suggest that “apps for individuals with bipolar disorder will likely be useful for some people in managing medication use, sleep duration, routine, and monitoring for and managing signs and symptoms” of the disorder, coinvestigator Evan H. Goulding, MD, PhD, assistant professor of psychiatry and behavioral sciences, Northwestern University, Chicago, told this news organization.
Use of the app may also “lead to decreased recurrence of mood episodes, impact overall depressive and manic symptom levels, and improve some aspects of quality of life,” Dr. Goulding added.
The findings were published online in JAMA Psychiatry.
Daily check-ins
The researchers randomly assigned 205 patients with BD to receive either usual care (n = 81; 56% women; mean age, 39 years) or usual care plus the smartphone-based self-management intervention LiveWell (n = 124; 65% women; mean age, 43 years) between March 2017 and April 2020. To be included, participants could not be experiencing a current mood episode or suicidal ideation.
The smartphone intervention included a daily check-in to monitor medication adherence, sleep, and wellness levels; coach visits to support adherence to the app; six phone calls over 16 weeks; and support from mental health professionals whenever needed. Participants in this group were asked to engage their mental health providers in the intervention as well.
Each participant in the control group had a visit with a coach who facilitated self-management support.
Investigators assessed all participants every 8 weeks until week 48 to gather information on mood symptoms and severity over the past 2 weeks and on quality of life.
The patients were also stratified into high- and low-risk relapse groups. The low-risk group was in asymptomatic recovery, meaning that they experienced two or fewer moderate symptoms of mania or depression in the previous 8 weeks. In addition, they had no moderate symptoms of mania or depression at study enrollment.
Patients in the high-risk group were recovering from an episode of mania or depression. They also had two or fewer moderate symptoms, but for 8 weeks or less.
Low-risk group fares better
Results showed that the smartphone intervention was significantly associated with a reduction in depressive symptoms vs. usual care (P = .02), as well as improvement in one aspect of the World Health Organization Quality of Life Assessment that measures social relationships (P = .02).
When the investigators stratified participants into risk groups, they found that for those in the low-risk group the smartphone-based intervention was associated with lower episode-relapse rates, lower mean percentage time symptomatic, and decreased manic symptom severity.
Mean estimated relapse rates by 48 weeks for the low-risk group were 12% for those in the intervention group and 37.2% for those in the control group. No differences were noted for the high-risk group.
Low-risk patients in the intervention group also had lower mean percentage-time symptomatic (17.9%) than those in the control group (26.1%) (Cohen d = .31).
“Our results are consistent with literature emphasizing the identification and facilitation of management plans for early warning signs of mood episodes and using these plans as an important self-management technique for avoiding relapse,” Dr. Goulding said.
Study limitations included low engagement by mental health professionals and low data generalizability to other populations, as the sample was mostly White (84% of the app group and 81% of the control group).
“There is a fairly large literature on risk factors, longitudinal trajectories, and stages of diseases that suggest we should already be able to predict relapse risk for individuals,” Dr. Goulding said.
“However, moving from overall risk to individual risk is trickier and will require larger datasets with longer follow-up to better understand what types of help should be delivered when and to whom,” he added.
‘Requires commitment’
John Torous, MD, director of the division of digital psychiatry at Beth Israel Deaconess Medical Center, Boston, noted that mental health apps such as LiveWell require “time and energy devoted by both the patient and their clinician for maximal efficacy, which requires commitment from and training for both parties as well.
“But with such an investment in people, there is good evidence apps can help people with bipolar disorder even during the more severe periods of the illness,” added Dr. Torous, who was not involved with the research.
The study was funded by the National Institute of Mental Health.
Dr. Goulding reports having received honoraria from Otsuka. Dr. Torous has reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
In a randomized clinical trial of usual care plus the experimental smartphone-based intervention known as LiveWell vs. usual care alone, participants in the smartphone group who were categorized as low-risk or in asymptomatic recovery at baseline also showed reduced manic symptom severity.
The results suggest that “apps for individuals with bipolar disorder will likely be useful for some people in managing medication use, sleep duration, routine, and monitoring for and managing signs and symptoms” of the disorder, coinvestigator Evan H. Goulding, MD, PhD, assistant professor of psychiatry and behavioral sciences, Northwestern University, Chicago, told this news organization.
Use of the app may also “lead to decreased recurrence of mood episodes, impact overall depressive and manic symptom levels, and improve some aspects of quality of life,” Dr. Goulding added.
The findings were published online in JAMA Psychiatry.
Daily check-ins
The researchers randomly assigned 205 patients with BD to receive either usual care (n = 81; 56% women; mean age, 39 years) or usual care plus the smartphone-based self-management intervention LiveWell (n = 124; 65% women; mean age, 43 years) between March 2017 and April 2020. To be included, participants could not be experiencing a current mood episode or suicidal ideation.
The smartphone intervention included a daily check-in to monitor medication adherence, sleep, and wellness levels; coach visits to support adherence to the app; six phone calls over 16 weeks; and support from mental health professionals whenever needed. Participants in this group were asked to engage their mental health providers in the intervention as well.
Each participant in the control group had a visit with a coach who facilitated self-management support.
Investigators assessed all participants every 8 weeks until week 48 to gather information on mood symptoms and severity over the past 2 weeks and on quality of life.
The patients were also stratified into high- and low-risk relapse groups. The low-risk group was in asymptomatic recovery, meaning that they experienced two or fewer moderate symptoms of mania or depression in the previous 8 weeks. In addition, they had no moderate symptoms of mania or depression at study enrollment.
Patients in the high-risk group were recovering from an episode of mania or depression. They also had two or fewer moderate symptoms, but for 8 weeks or less.
Low-risk group fares better
Results showed that the smartphone intervention was significantly associated with a reduction in depressive symptoms vs. usual care (P = .02), as well as improvement in one aspect of the World Health Organization Quality of Life Assessment that measures social relationships (P = .02).
When the investigators stratified participants into risk groups, they found that for those in the low-risk group the smartphone-based intervention was associated with lower episode-relapse rates, lower mean percentage time symptomatic, and decreased manic symptom severity.
Mean estimated relapse rates by 48 weeks for the low-risk group were 12% for those in the intervention group and 37.2% for those in the control group. No differences were noted for the high-risk group.
Low-risk patients in the intervention group also had lower mean percentage-time symptomatic (17.9%) than those in the control group (26.1%) (Cohen d = .31).
“Our results are consistent with literature emphasizing the identification and facilitation of management plans for early warning signs of mood episodes and using these plans as an important self-management technique for avoiding relapse,” Dr. Goulding said.
Study limitations included low engagement by mental health professionals and low data generalizability to other populations, as the sample was mostly White (84% of the app group and 81% of the control group).
“There is a fairly large literature on risk factors, longitudinal trajectories, and stages of diseases that suggest we should already be able to predict relapse risk for individuals,” Dr. Goulding said.
“However, moving from overall risk to individual risk is trickier and will require larger datasets with longer follow-up to better understand what types of help should be delivered when and to whom,” he added.
‘Requires commitment’
John Torous, MD, director of the division of digital psychiatry at Beth Israel Deaconess Medical Center, Boston, noted that mental health apps such as LiveWell require “time and energy devoted by both the patient and their clinician for maximal efficacy, which requires commitment from and training for both parties as well.
“But with such an investment in people, there is good evidence apps can help people with bipolar disorder even during the more severe periods of the illness,” added Dr. Torous, who was not involved with the research.
The study was funded by the National Institute of Mental Health.
Dr. Goulding reports having received honoraria from Otsuka. Dr. Torous has reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM JAMA PSYCHIATRY
Postconcussion symptoms tied to high risk of depression
Results of a large meta-analysis that included 18 studies and more than 9,000 patients showed a fourfold higher risk of developing depressive symptoms in those with PPCS versus those without PPCS.
“In this meta-analysis, experiencing PPCS was associated with a higher risk of experiencing depressive symptoms,” write the investigators, led by Maude Lambert, PhD, of the School of Psychology, University of Ottawa, and Bloorview Research Institute, Toronto.
“There are several important clinical and health policy implications of the findings. Most notably, the development of strategies for effective prevention and earlier intervention to optimize mental health recovery following a concussion should be supported,” they add.
The study was published online in JAMA Network Open.
‘Important minority’
An “important minority” of 15%-30% of those with concussions continue to experience symptoms for months, or even years, following the injury, the investigators note.
Symptoms vary but can include headaches, fatigue, dizziness, cognitive difficulties, and emotional changes, which can “significantly impact an individual’s everyday functioning.”
The association between PPCS and mental health outcomes “has emerged as an area of interest” over the past decade, with multiple studies pointing to bidirectional associations between depressive symptoms and PPCS, the researchers note. Individuals with PPCS are at significantly higher risk of experiencing depressive symptoms, and depressive symptoms, in turn, predict more prolonged postconcussion recovery, they add.
The authors conducted a previous scoping review that showed individuals with PPCS had “greater mental health difficulties than individuals who recovered from concussion or healthy controls.”
But “quantitative summaries evaluating the magnitude and nature of the association between PPCS and mental health outcomes were not conducted,” so they decided to conduct a follow-up meta-analysis to corroborate the hypothesis that PPCS may be associated with depressive symptoms.
The researchers also wanted to “investigate potential moderators of that association and determine whether the association between depressive symptoms and PPCS differed based on age, sex, mental illness, history of concussion, and time since the injury.”
This could have “significant public health implications” as it represents an “important step” toward understanding the association between PPCS and mental health, paving the way for the “development of optimal postconcussion intervention strategies, targeting effective prevention and earlier intervention to enhance recovery trajectories, improve mental health, and promote well-being following concussion.”
To be included in the meta-analysis, a study had to focus on participants who had experienced a concussion, diagnosed by a health care professional, or as classified by diagnostic measures, and who experienced greater than or equal to 1 concussion symptom lasting greater than 4 weeks.
There was no explicit upper limit on duration, and individuals of all ages were eligible.
Depressive symptoms were defined as “an outcome that must be measured by a validated and standardized measure of depression.”
Biopsychosocial model
Of 580 reports assessed for eligibility, 18 were included in the meta-analysis, incorporating a total of 9,101 participants, with a median (range) sample size of 154 (48-4,462) participants and a mean (SD) participant age of 33.7 (14.4) years.
The mean length of time since the concussion was 21.3 (18.7) weeks. Of the participants, a mean of 36.1% (11.1%) had a history of greater than or equal to 2 concussions.
Close to three-quarters of the studies (72%) used a cross-sectional design, with most studies conducted in North America, and the remaining conducted in Europe, China, and New Zealand.
The researchers found a “significant positive association” between PPCS and postinjury depressive symptoms (odds ratio, 4.87; 95% confidence interval, 3.01-7.90; P < .001), “representing a large effect size.”
Funnel plot and Egger test analyses “suggested the presence of a publication bias.” However, even after accounting for publication bias, the effect size “of large magnitude” remained, the authors report (OR, 4.56; 95% CI, 2.82-7.37; P < .001).
No significant moderators were identified, “likely due to the small number of studies included,” they speculate.
They note that the current study “does not allow inference about the causal directionality of the association” between PPCS and postinjury depressive symptoms, so the question remains: Do PPCS induce depressive symptoms, or do depressive symptoms induce PPCS?”
Despite this unanswered question, the findings still have important clinical and public health implications, highlighting “the need for a greater understanding of the mechanisms of development and etiology of depressive symptoms postconcussion” and emphasizing “the necessary emergence for timely and effective treatment interventions for depressive symptoms to optimize the long-term prognosis of concussion,” the authors note.
They add that several research teams “have aimed to gain more insight into the etiology and underlying mechanisms of development and course of mental health difficulties in individuals who experience a concussion” and have arrived at a biopsychosocial framework, in light of “the myriad of contributing physiological, biological, and psychosocial factors.”
They recommend the establishment of “specialized multidisciplinary or interdisciplinary concussion care programs should include health care professionals with strong clinical foundations and training in mental health conditions.”
Speedy multidisciplinary care
Commenting on the research, Charles Tator, MD, PhD, professor of neurosurgery, University of Toronto, Division of Neurosurgery, Toronto Western Hospital, said the researchers “performed a thorough systematic review” showing “emphatically that depression occurs in this population.”
Dr. Tator, the director of the Canadian Concussion Centre, who was not involved with the current study, continued: “Nowadays clinical discoveries are validated through a progression of case reports, single-center retrospective cohort studies like ours, referenced by [Dr.] Lambert et al., and then confirmatory systematic reviews, each adding important layers of evidence.”
“This evaluative process has now endorsed the importance of early treatment of mental health symptoms in patients with persisting symptoms, which can include depression, anxiety, and PTSD,” he said.
He recommended that treatment should start with family physicians and nurse practitioners “but may require escalation to psychologists and social workers and then to psychiatrists who are often more skilled in medication selection.”
He encouraged “speedy multidisciplinary care,” noting that the possibility of suicide is worrisome.
No source of study funding was listed. A study coauthor, Shannon Scratch, PhD, has reported receiving funds from the Holland Bloorview Kids Rehabilitation Hospital Foundation (via the Holland Family Professorship in Acquired Brain Injury) during the conduct of this study. No other disclosures were reported. Dr. Tator has reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Results of a large meta-analysis that included 18 studies and more than 9,000 patients showed a fourfold higher risk of developing depressive symptoms in those with PPCS versus those without PPCS.
“In this meta-analysis, experiencing PPCS was associated with a higher risk of experiencing depressive symptoms,” write the investigators, led by Maude Lambert, PhD, of the School of Psychology, University of Ottawa, and Bloorview Research Institute, Toronto.
“There are several important clinical and health policy implications of the findings. Most notably, the development of strategies for effective prevention and earlier intervention to optimize mental health recovery following a concussion should be supported,” they add.
The study was published online in JAMA Network Open.
‘Important minority’
An “important minority” of 15%-30% of those with concussions continue to experience symptoms for months, or even years, following the injury, the investigators note.
Symptoms vary but can include headaches, fatigue, dizziness, cognitive difficulties, and emotional changes, which can “significantly impact an individual’s everyday functioning.”
The association between PPCS and mental health outcomes “has emerged as an area of interest” over the past decade, with multiple studies pointing to bidirectional associations between depressive symptoms and PPCS, the researchers note. Individuals with PPCS are at significantly higher risk of experiencing depressive symptoms, and depressive symptoms, in turn, predict more prolonged postconcussion recovery, they add.
The authors conducted a previous scoping review that showed individuals with PPCS had “greater mental health difficulties than individuals who recovered from concussion or healthy controls.”
But “quantitative summaries evaluating the magnitude and nature of the association between PPCS and mental health outcomes were not conducted,” so they decided to conduct a follow-up meta-analysis to corroborate the hypothesis that PPCS may be associated with depressive symptoms.
The researchers also wanted to “investigate potential moderators of that association and determine whether the association between depressive symptoms and PPCS differed based on age, sex, mental illness, history of concussion, and time since the injury.”
This could have “significant public health implications” as it represents an “important step” toward understanding the association between PPCS and mental health, paving the way for the “development of optimal postconcussion intervention strategies, targeting effective prevention and earlier intervention to enhance recovery trajectories, improve mental health, and promote well-being following concussion.”
To be included in the meta-analysis, a study had to focus on participants who had experienced a concussion, diagnosed by a health care professional, or as classified by diagnostic measures, and who experienced greater than or equal to 1 concussion symptom lasting greater than 4 weeks.
There was no explicit upper limit on duration, and individuals of all ages were eligible.
Depressive symptoms were defined as “an outcome that must be measured by a validated and standardized measure of depression.”
Biopsychosocial model
Of 580 reports assessed for eligibility, 18 were included in the meta-analysis, incorporating a total of 9,101 participants, with a median (range) sample size of 154 (48-4,462) participants and a mean (SD) participant age of 33.7 (14.4) years.
The mean length of time since the concussion was 21.3 (18.7) weeks. Of the participants, a mean of 36.1% (11.1%) had a history of greater than or equal to 2 concussions.
Close to three-quarters of the studies (72%) used a cross-sectional design, with most studies conducted in North America, and the remaining conducted in Europe, China, and New Zealand.
The researchers found a “significant positive association” between PPCS and postinjury depressive symptoms (odds ratio, 4.87; 95% confidence interval, 3.01-7.90; P < .001), “representing a large effect size.”
Funnel plot and Egger test analyses “suggested the presence of a publication bias.” However, even after accounting for publication bias, the effect size “of large magnitude” remained, the authors report (OR, 4.56; 95% CI, 2.82-7.37; P < .001).
No significant moderators were identified, “likely due to the small number of studies included,” they speculate.
They note that the current study “does not allow inference about the causal directionality of the association” between PPCS and postinjury depressive symptoms, so the question remains: Do PPCS induce depressive symptoms, or do depressive symptoms induce PPCS?”
Despite this unanswered question, the findings still have important clinical and public health implications, highlighting “the need for a greater understanding of the mechanisms of development and etiology of depressive symptoms postconcussion” and emphasizing “the necessary emergence for timely and effective treatment interventions for depressive symptoms to optimize the long-term prognosis of concussion,” the authors note.
They add that several research teams “have aimed to gain more insight into the etiology and underlying mechanisms of development and course of mental health difficulties in individuals who experience a concussion” and have arrived at a biopsychosocial framework, in light of “the myriad of contributing physiological, biological, and psychosocial factors.”
They recommend the establishment of “specialized multidisciplinary or interdisciplinary concussion care programs should include health care professionals with strong clinical foundations and training in mental health conditions.”
Speedy multidisciplinary care
Commenting on the research, Charles Tator, MD, PhD, professor of neurosurgery, University of Toronto, Division of Neurosurgery, Toronto Western Hospital, said the researchers “performed a thorough systematic review” showing “emphatically that depression occurs in this population.”
Dr. Tator, the director of the Canadian Concussion Centre, who was not involved with the current study, continued: “Nowadays clinical discoveries are validated through a progression of case reports, single-center retrospective cohort studies like ours, referenced by [Dr.] Lambert et al., and then confirmatory systematic reviews, each adding important layers of evidence.”
“This evaluative process has now endorsed the importance of early treatment of mental health symptoms in patients with persisting symptoms, which can include depression, anxiety, and PTSD,” he said.
He recommended that treatment should start with family physicians and nurse practitioners “but may require escalation to psychologists and social workers and then to psychiatrists who are often more skilled in medication selection.”
He encouraged “speedy multidisciplinary care,” noting that the possibility of suicide is worrisome.
No source of study funding was listed. A study coauthor, Shannon Scratch, PhD, has reported receiving funds from the Holland Bloorview Kids Rehabilitation Hospital Foundation (via the Holland Family Professorship in Acquired Brain Injury) during the conduct of this study. No other disclosures were reported. Dr. Tator has reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Results of a large meta-analysis that included 18 studies and more than 9,000 patients showed a fourfold higher risk of developing depressive symptoms in those with PPCS versus those without PPCS.
“In this meta-analysis, experiencing PPCS was associated with a higher risk of experiencing depressive symptoms,” write the investigators, led by Maude Lambert, PhD, of the School of Psychology, University of Ottawa, and Bloorview Research Institute, Toronto.
“There are several important clinical and health policy implications of the findings. Most notably, the development of strategies for effective prevention and earlier intervention to optimize mental health recovery following a concussion should be supported,” they add.
The study was published online in JAMA Network Open.
‘Important minority’
An “important minority” of 15%-30% of those with concussions continue to experience symptoms for months, or even years, following the injury, the investigators note.
Symptoms vary but can include headaches, fatigue, dizziness, cognitive difficulties, and emotional changes, which can “significantly impact an individual’s everyday functioning.”
The association between PPCS and mental health outcomes “has emerged as an area of interest” over the past decade, with multiple studies pointing to bidirectional associations between depressive symptoms and PPCS, the researchers note. Individuals with PPCS are at significantly higher risk of experiencing depressive symptoms, and depressive symptoms, in turn, predict more prolonged postconcussion recovery, they add.
The authors conducted a previous scoping review that showed individuals with PPCS had “greater mental health difficulties than individuals who recovered from concussion or healthy controls.”
But “quantitative summaries evaluating the magnitude and nature of the association between PPCS and mental health outcomes were not conducted,” so they decided to conduct a follow-up meta-analysis to corroborate the hypothesis that PPCS may be associated with depressive symptoms.
The researchers also wanted to “investigate potential moderators of that association and determine whether the association between depressive symptoms and PPCS differed based on age, sex, mental illness, history of concussion, and time since the injury.”
This could have “significant public health implications” as it represents an “important step” toward understanding the association between PPCS and mental health, paving the way for the “development of optimal postconcussion intervention strategies, targeting effective prevention and earlier intervention to enhance recovery trajectories, improve mental health, and promote well-being following concussion.”
To be included in the meta-analysis, a study had to focus on participants who had experienced a concussion, diagnosed by a health care professional, or as classified by diagnostic measures, and who experienced greater than or equal to 1 concussion symptom lasting greater than 4 weeks.
There was no explicit upper limit on duration, and individuals of all ages were eligible.
Depressive symptoms were defined as “an outcome that must be measured by a validated and standardized measure of depression.”
Biopsychosocial model
Of 580 reports assessed for eligibility, 18 were included in the meta-analysis, incorporating a total of 9,101 participants, with a median (range) sample size of 154 (48-4,462) participants and a mean (SD) participant age of 33.7 (14.4) years.
The mean length of time since the concussion was 21.3 (18.7) weeks. Of the participants, a mean of 36.1% (11.1%) had a history of greater than or equal to 2 concussions.
Close to three-quarters of the studies (72%) used a cross-sectional design, with most studies conducted in North America, and the remaining conducted in Europe, China, and New Zealand.
The researchers found a “significant positive association” between PPCS and postinjury depressive symptoms (odds ratio, 4.87; 95% confidence interval, 3.01-7.90; P < .001), “representing a large effect size.”
Funnel plot and Egger test analyses “suggested the presence of a publication bias.” However, even after accounting for publication bias, the effect size “of large magnitude” remained, the authors report (OR, 4.56; 95% CI, 2.82-7.37; P < .001).
No significant moderators were identified, “likely due to the small number of studies included,” they speculate.
They note that the current study “does not allow inference about the causal directionality of the association” between PPCS and postinjury depressive symptoms, so the question remains: Do PPCS induce depressive symptoms, or do depressive symptoms induce PPCS?”
Despite this unanswered question, the findings still have important clinical and public health implications, highlighting “the need for a greater understanding of the mechanisms of development and etiology of depressive symptoms postconcussion” and emphasizing “the necessary emergence for timely and effective treatment interventions for depressive symptoms to optimize the long-term prognosis of concussion,” the authors note.
They add that several research teams “have aimed to gain more insight into the etiology and underlying mechanisms of development and course of mental health difficulties in individuals who experience a concussion” and have arrived at a biopsychosocial framework, in light of “the myriad of contributing physiological, biological, and psychosocial factors.”
They recommend the establishment of “specialized multidisciplinary or interdisciplinary concussion care programs should include health care professionals with strong clinical foundations and training in mental health conditions.”
Speedy multidisciplinary care
Commenting on the research, Charles Tator, MD, PhD, professor of neurosurgery, University of Toronto, Division of Neurosurgery, Toronto Western Hospital, said the researchers “performed a thorough systematic review” showing “emphatically that depression occurs in this population.”
Dr. Tator, the director of the Canadian Concussion Centre, who was not involved with the current study, continued: “Nowadays clinical discoveries are validated through a progression of case reports, single-center retrospective cohort studies like ours, referenced by [Dr.] Lambert et al., and then confirmatory systematic reviews, each adding important layers of evidence.”
“This evaluative process has now endorsed the importance of early treatment of mental health symptoms in patients with persisting symptoms, which can include depression, anxiety, and PTSD,” he said.
He recommended that treatment should start with family physicians and nurse practitioners “but may require escalation to psychologists and social workers and then to psychiatrists who are often more skilled in medication selection.”
He encouraged “speedy multidisciplinary care,” noting that the possibility of suicide is worrisome.
No source of study funding was listed. A study coauthor, Shannon Scratch, PhD, has reported receiving funds from the Holland Bloorview Kids Rehabilitation Hospital Foundation (via the Holland Family Professorship in Acquired Brain Injury) during the conduct of this study. No other disclosures were reported. Dr. Tator has reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM JAMA NETWORK OPEN
Sleep complaints in major depression flag risk for other psychiatric disorders
Investigators studied 3-year incidence rates of psychiatric disorders in almost 3,000 patients experiencing an MDE. Results showed that having a history of difficulty falling asleep, early morning awakening, and hypersomnia increased risk for incident psychiatric disorders.
“The findings of this study suggest the potential value of including insomnia and hypersomnia in clinical assessments of all psychiatric disorders,” write the investigators, led by Bénédicte Barbotin, MD, Département de Psychiatrie et d’Addictologie, Assistance Publique-Hôpitaux de Paris, Hôpital Bichat-Claude Bernard, France.
“Insomnia and hypersomnia symptoms may be prodromal transdiagnostic biomarkers and easily modifiable therapeutic targets for the prevention of psychiatric disorders,” they add.
The findings were published online recently in the Journal of Clinical Psychiatry.
Bidirectional association
The researchers note that sleep disturbance is “one of the most common symptoms” associated with major depressive disorder (MDD) and may be “both a consequence and a cause.”
Moreover, improving sleep disturbances for patients with an MDE “tends to improve depressive symptom and outcomes,” they add.
Although the possibility of a bidirectional association between MDEs and sleep disturbances “offers a new perspective that sleep complaints might be a predictive prodromal symptom,” the association of sleep complaints with the subsequent development of other psychiatric disorders in MDEs “remains poorly documented,” the investigators write.
The observation that sleep complaints are associated with psychiatric complications and adverse outcomes, such as suicidality and substance overdose, suggests that longitudinal studies “may help to better understand these relationships.”
To investigate these issues, the researchers examined three sleep complaints among patients with MDE: trouble falling asleep, early morning awakening, and hypersomnia. They adjusted for an array of variables, including antisocial personality disorders, use of sedatives or tranquilizers, sociodemographic characteristics, MDE severity, poverty, obesity, educational level, and stressful life events.
They also used a “bifactor latent variable approach” to “disentangle” a number of effects, including those shared by all psychiatric disorders; those specific to dimensions of psychopathology, such as internalizing dimension; and those specific to individual psychiatric disorders, such as dysthymia.
“To our knowledge, this is the most extensive prospective assessment [ever conducted] of associations between sleep complaints and incident psychiatric disorders,” the investigators write.
They drew on data from Waves 1 and 2 of the National Epidemiological Survey on Alcohol and Related Conditions, a large nationally representative survey conducted in 2001-2002 (Wave 1) and 2004-2005 (Wave 2) by the National Institute on Alcoholism and Alcohol Abuse.
The analysis included 2,864 participants who experienced MDE in the year prior to Wave 1 and who completed interviews at both waves.
Researchers assessed past-year DSM-IV Axis I disorders and baseline sleep complaints at Wave 1, as well as incident DSM-IV Axis I disorders between the two waves – including substance use, mood, and anxiety disorders.
Screening needed?
Results showed a wide range of incidence rates for psychiatric disorders between Wave 1 and Wave 2, ranging from 2.7% for cannabis use to 8.2% for generalized anxiety disorder.
The lifetime prevalence of sleep complaints was higher among participants who developed a psychiatric disorder between the two waves than among those who did not have sleep complaints. The range (from lowest to highest percentage) is shown in the accompanying table.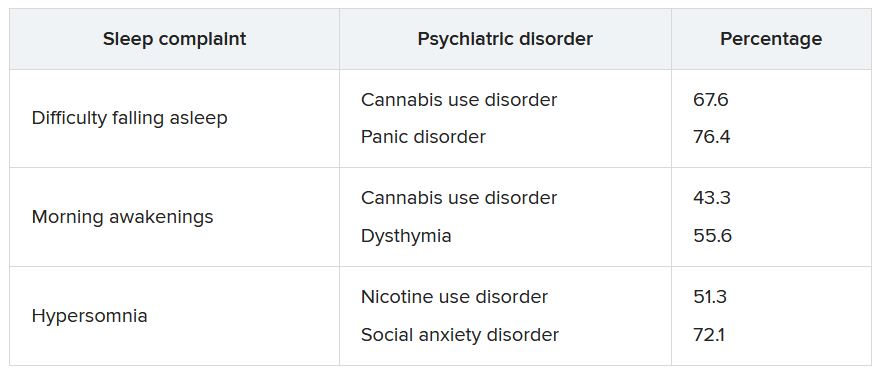
A higher number of sleep complaints was also associated with higher percentages of psychiatric disorders.
Hypersomnia, in particular, significantly increased the odds of having another psychiatric disorder. For patients with MDD who reported hypersomnia, the mean number of sleep disorders was significantly higher than for patients without hypersomnia (2.08 vs. 1.32; P < .001).
“This explains why hypersomnia appears more strongly associated with the incidence of psychiatric disorders,” the investigators write.
After adjusting for sociodemographic and clinical characteristics and antisocial personality disorder, the effects shared across all sleep complaints were “significantly associated with the incident general psychopathology factor, representing mechanisms that may lead to incidence of all psychiatric disorder in the model,” they add.
The researchers note that insomnia and hypersomnia can impair cognitive function, decision-making, problem-solving, and emotion processing networks, thereby increasing the onset of psychiatric disorders in vulnerable individuals.
Shared biological determinants, such as monoamine neurotransmitters that play a major role in depression, anxiety, substance use disorders, and the regulation of sleep stages, may also underlie both sleep disturbances and psychiatric disorders, they speculate.
“These results suggest the importance of systematically assessing insomnia and hypersomnia when evaluating psychiatric disorders and considering these symptoms as nonspecific prodromal or at-risk symptoms, also shared with suicidal behaviors,” the investigators write.
“In addition, since most individuals who developed a psychiatric disorder had at least one sleep complaint, all psychiatric disorders should be carefully screened among individuals with sleep complaints,” they add.
Transdiagnostic phenomenon
In a comment, Roger McIntyre, MD, professor of psychiatry and pharmacology at the University of Toronto, and head of the Mood Disorders Psychopharmacology Unit, noted that the study replicates previous observations that a bidirectional relationship exists between sleep disturbances and mental disorders and that there “seems to be a relationship between sleep disturbance and suicidality that is bidirectional.”
He added that he appreciated the fact that the investigators “took this knowledge one step further; and what they are saying is that within the syndrome of depression, it is the sleep disturbance that is predicting future problems.”
Dr. McIntyre, who is also chairman and executive director of the Brain and Cognitive Discover Foundation in Toronto, was not involved with the study.
The data suggest that, “conceptually, sleep disturbance is a transdiagnostic phenomenon that may also be the nexus when multiple comorbid mental disorders occur,” he said.
“If this is the case, clinically, there is an opportunity here to prevent incident mental disorders in persons with depression and sleep disturbance, prioritizing sleep management in any patient with a mood disorder,” Dr. McIntyre added.
He noted that “the testable hypothesis” is how this is occurring mechanistically.
“I would conjecture that it could be inflammation and/or insulin resistance that is part of sleep disturbance that could predispose and portend other mental illnesses – and likely other medical conditions too, such as obesity and diabetes,” he said.
The study received no specific funding from any funding agency, commercial, or not-for-profit sectors. The investigators’ relevant financial relationships are listed in the original article. Dr. McIntyre has received research grant support from CIHR/GACD/National Natural Science Foundation of China and the Milken Institute; has received speaker/consultation fees from Lundbeck, Janssen, Alkermes,Neumora Therapeutics, Boehringer Ingelheim, Sage, Biogen, Mitsubishi Tanabe, Purdue, Pfizer, Otsuka, Takeda, Neurocrine, Sunovion, Bausch Health, Axsome, Novo Nordisk, Kris, Sanofi, Eisai, Intra-Cellular, NewBridge Pharmaceuticals, Viatris, AbbVie, and Atai Life Sciences; and is a CEO of Braxia Scientific Corp.
A version of this article first appeared on Medscape.com.
Investigators studied 3-year incidence rates of psychiatric disorders in almost 3,000 patients experiencing an MDE. Results showed that having a history of difficulty falling asleep, early morning awakening, and hypersomnia increased risk for incident psychiatric disorders.
“The findings of this study suggest the potential value of including insomnia and hypersomnia in clinical assessments of all psychiatric disorders,” write the investigators, led by Bénédicte Barbotin, MD, Département de Psychiatrie et d’Addictologie, Assistance Publique-Hôpitaux de Paris, Hôpital Bichat-Claude Bernard, France.
“Insomnia and hypersomnia symptoms may be prodromal transdiagnostic biomarkers and easily modifiable therapeutic targets for the prevention of psychiatric disorders,” they add.
The findings were published online recently in the Journal of Clinical Psychiatry.
Bidirectional association
The researchers note that sleep disturbance is “one of the most common symptoms” associated with major depressive disorder (MDD) and may be “both a consequence and a cause.”
Moreover, improving sleep disturbances for patients with an MDE “tends to improve depressive symptom and outcomes,” they add.
Although the possibility of a bidirectional association between MDEs and sleep disturbances “offers a new perspective that sleep complaints might be a predictive prodromal symptom,” the association of sleep complaints with the subsequent development of other psychiatric disorders in MDEs “remains poorly documented,” the investigators write.
The observation that sleep complaints are associated with psychiatric complications and adverse outcomes, such as suicidality and substance overdose, suggests that longitudinal studies “may help to better understand these relationships.”
To investigate these issues, the researchers examined three sleep complaints among patients with MDE: trouble falling asleep, early morning awakening, and hypersomnia. They adjusted for an array of variables, including antisocial personality disorders, use of sedatives or tranquilizers, sociodemographic characteristics, MDE severity, poverty, obesity, educational level, and stressful life events.
They also used a “bifactor latent variable approach” to “disentangle” a number of effects, including those shared by all psychiatric disorders; those specific to dimensions of psychopathology, such as internalizing dimension; and those specific to individual psychiatric disorders, such as dysthymia.
“To our knowledge, this is the most extensive prospective assessment [ever conducted] of associations between sleep complaints and incident psychiatric disorders,” the investigators write.
They drew on data from Waves 1 and 2 of the National Epidemiological Survey on Alcohol and Related Conditions, a large nationally representative survey conducted in 2001-2002 (Wave 1) and 2004-2005 (Wave 2) by the National Institute on Alcoholism and Alcohol Abuse.
The analysis included 2,864 participants who experienced MDE in the year prior to Wave 1 and who completed interviews at both waves.
Researchers assessed past-year DSM-IV Axis I disorders and baseline sleep complaints at Wave 1, as well as incident DSM-IV Axis I disorders between the two waves – including substance use, mood, and anxiety disorders.
Screening needed?
Results showed a wide range of incidence rates for psychiatric disorders between Wave 1 and Wave 2, ranging from 2.7% for cannabis use to 8.2% for generalized anxiety disorder.
The lifetime prevalence of sleep complaints was higher among participants who developed a psychiatric disorder between the two waves than among those who did not have sleep complaints. The range (from lowest to highest percentage) is shown in the accompanying table.
A higher number of sleep complaints was also associated with higher percentages of psychiatric disorders.
Hypersomnia, in particular, significantly increased the odds of having another psychiatric disorder. For patients with MDD who reported hypersomnia, the mean number of sleep disorders was significantly higher than for patients without hypersomnia (2.08 vs. 1.32; P < .001).
“This explains why hypersomnia appears more strongly associated with the incidence of psychiatric disorders,” the investigators write.
After adjusting for sociodemographic and clinical characteristics and antisocial personality disorder, the effects shared across all sleep complaints were “significantly associated with the incident general psychopathology factor, representing mechanisms that may lead to incidence of all psychiatric disorder in the model,” they add.
The researchers note that insomnia and hypersomnia can impair cognitive function, decision-making, problem-solving, and emotion processing networks, thereby increasing the onset of psychiatric disorders in vulnerable individuals.
Shared biological determinants, such as monoamine neurotransmitters that play a major role in depression, anxiety, substance use disorders, and the regulation of sleep stages, may also underlie both sleep disturbances and psychiatric disorders, they speculate.
“These results suggest the importance of systematically assessing insomnia and hypersomnia when evaluating psychiatric disorders and considering these symptoms as nonspecific prodromal or at-risk symptoms, also shared with suicidal behaviors,” the investigators write.
“In addition, since most individuals who developed a psychiatric disorder had at least one sleep complaint, all psychiatric disorders should be carefully screened among individuals with sleep complaints,” they add.
Transdiagnostic phenomenon
In a comment, Roger McIntyre, MD, professor of psychiatry and pharmacology at the University of Toronto, and head of the Mood Disorders Psychopharmacology Unit, noted that the study replicates previous observations that a bidirectional relationship exists between sleep disturbances and mental disorders and that there “seems to be a relationship between sleep disturbance and suicidality that is bidirectional.”
He added that he appreciated the fact that the investigators “took this knowledge one step further; and what they are saying is that within the syndrome of depression, it is the sleep disturbance that is predicting future problems.”
Dr. McIntyre, who is also chairman and executive director of the Brain and Cognitive Discover Foundation in Toronto, was not involved with the study.
The data suggest that, “conceptually, sleep disturbance is a transdiagnostic phenomenon that may also be the nexus when multiple comorbid mental disorders occur,” he said.
“If this is the case, clinically, there is an opportunity here to prevent incident mental disorders in persons with depression and sleep disturbance, prioritizing sleep management in any patient with a mood disorder,” Dr. McIntyre added.
He noted that “the testable hypothesis” is how this is occurring mechanistically.
“I would conjecture that it could be inflammation and/or insulin resistance that is part of sleep disturbance that could predispose and portend other mental illnesses – and likely other medical conditions too, such as obesity and diabetes,” he said.
The study received no specific funding from any funding agency, commercial, or not-for-profit sectors. The investigators’ relevant financial relationships are listed in the original article. Dr. McIntyre has received research grant support from CIHR/GACD/National Natural Science Foundation of China and the Milken Institute; has received speaker/consultation fees from Lundbeck, Janssen, Alkermes,Neumora Therapeutics, Boehringer Ingelheim, Sage, Biogen, Mitsubishi Tanabe, Purdue, Pfizer, Otsuka, Takeda, Neurocrine, Sunovion, Bausch Health, Axsome, Novo Nordisk, Kris, Sanofi, Eisai, Intra-Cellular, NewBridge Pharmaceuticals, Viatris, AbbVie, and Atai Life Sciences; and is a CEO of Braxia Scientific Corp.
A version of this article first appeared on Medscape.com.
Investigators studied 3-year incidence rates of psychiatric disorders in almost 3,000 patients experiencing an MDE. Results showed that having a history of difficulty falling asleep, early morning awakening, and hypersomnia increased risk for incident psychiatric disorders.
“The findings of this study suggest the potential value of including insomnia and hypersomnia in clinical assessments of all psychiatric disorders,” write the investigators, led by Bénédicte Barbotin, MD, Département de Psychiatrie et d’Addictologie, Assistance Publique-Hôpitaux de Paris, Hôpital Bichat-Claude Bernard, France.
“Insomnia and hypersomnia symptoms may be prodromal transdiagnostic biomarkers and easily modifiable therapeutic targets for the prevention of psychiatric disorders,” they add.
The findings were published online recently in the Journal of Clinical Psychiatry.
Bidirectional association
The researchers note that sleep disturbance is “one of the most common symptoms” associated with major depressive disorder (MDD) and may be “both a consequence and a cause.”
Moreover, improving sleep disturbances for patients with an MDE “tends to improve depressive symptom and outcomes,” they add.
Although the possibility of a bidirectional association between MDEs and sleep disturbances “offers a new perspective that sleep complaints might be a predictive prodromal symptom,” the association of sleep complaints with the subsequent development of other psychiatric disorders in MDEs “remains poorly documented,” the investigators write.
The observation that sleep complaints are associated with psychiatric complications and adverse outcomes, such as suicidality and substance overdose, suggests that longitudinal studies “may help to better understand these relationships.”
To investigate these issues, the researchers examined three sleep complaints among patients with MDE: trouble falling asleep, early morning awakening, and hypersomnia. They adjusted for an array of variables, including antisocial personality disorders, use of sedatives or tranquilizers, sociodemographic characteristics, MDE severity, poverty, obesity, educational level, and stressful life events.
They also used a “bifactor latent variable approach” to “disentangle” a number of effects, including those shared by all psychiatric disorders; those specific to dimensions of psychopathology, such as internalizing dimension; and those specific to individual psychiatric disorders, such as dysthymia.
“To our knowledge, this is the most extensive prospective assessment [ever conducted] of associations between sleep complaints and incident psychiatric disorders,” the investigators write.
They drew on data from Waves 1 and 2 of the National Epidemiological Survey on Alcohol and Related Conditions, a large nationally representative survey conducted in 2001-2002 (Wave 1) and 2004-2005 (Wave 2) by the National Institute on Alcoholism and Alcohol Abuse.
The analysis included 2,864 participants who experienced MDE in the year prior to Wave 1 and who completed interviews at both waves.
Researchers assessed past-year DSM-IV Axis I disorders and baseline sleep complaints at Wave 1, as well as incident DSM-IV Axis I disorders between the two waves – including substance use, mood, and anxiety disorders.
Screening needed?
Results showed a wide range of incidence rates for psychiatric disorders between Wave 1 and Wave 2, ranging from 2.7% for cannabis use to 8.2% for generalized anxiety disorder.
The lifetime prevalence of sleep complaints was higher among participants who developed a psychiatric disorder between the two waves than among those who did not have sleep complaints. The range (from lowest to highest percentage) is shown in the accompanying table.
A higher number of sleep complaints was also associated with higher percentages of psychiatric disorders.
Hypersomnia, in particular, significantly increased the odds of having another psychiatric disorder. For patients with MDD who reported hypersomnia, the mean number of sleep disorders was significantly higher than for patients without hypersomnia (2.08 vs. 1.32; P < .001).
“This explains why hypersomnia appears more strongly associated with the incidence of psychiatric disorders,” the investigators write.
After adjusting for sociodemographic and clinical characteristics and antisocial personality disorder, the effects shared across all sleep complaints were “significantly associated with the incident general psychopathology factor, representing mechanisms that may lead to incidence of all psychiatric disorder in the model,” they add.
The researchers note that insomnia and hypersomnia can impair cognitive function, decision-making, problem-solving, and emotion processing networks, thereby increasing the onset of psychiatric disorders in vulnerable individuals.
Shared biological determinants, such as monoamine neurotransmitters that play a major role in depression, anxiety, substance use disorders, and the regulation of sleep stages, may also underlie both sleep disturbances and psychiatric disorders, they speculate.
“These results suggest the importance of systematically assessing insomnia and hypersomnia when evaluating psychiatric disorders and considering these symptoms as nonspecific prodromal or at-risk symptoms, also shared with suicidal behaviors,” the investigators write.
“In addition, since most individuals who developed a psychiatric disorder had at least one sleep complaint, all psychiatric disorders should be carefully screened among individuals with sleep complaints,” they add.
Transdiagnostic phenomenon
In a comment, Roger McIntyre, MD, professor of psychiatry and pharmacology at the University of Toronto, and head of the Mood Disorders Psychopharmacology Unit, noted that the study replicates previous observations that a bidirectional relationship exists between sleep disturbances and mental disorders and that there “seems to be a relationship between sleep disturbance and suicidality that is bidirectional.”
He added that he appreciated the fact that the investigators “took this knowledge one step further; and what they are saying is that within the syndrome of depression, it is the sleep disturbance that is predicting future problems.”
Dr. McIntyre, who is also chairman and executive director of the Brain and Cognitive Discover Foundation in Toronto, was not involved with the study.
The data suggest that, “conceptually, sleep disturbance is a transdiagnostic phenomenon that may also be the nexus when multiple comorbid mental disorders occur,” he said.
“If this is the case, clinically, there is an opportunity here to prevent incident mental disorders in persons with depression and sleep disturbance, prioritizing sleep management in any patient with a mood disorder,” Dr. McIntyre added.
He noted that “the testable hypothesis” is how this is occurring mechanistically.
“I would conjecture that it could be inflammation and/or insulin resistance that is part of sleep disturbance that could predispose and portend other mental illnesses – and likely other medical conditions too, such as obesity and diabetes,” he said.
The study received no specific funding from any funding agency, commercial, or not-for-profit sectors. The investigators’ relevant financial relationships are listed in the original article. Dr. McIntyre has received research grant support from CIHR/GACD/National Natural Science Foundation of China and the Milken Institute; has received speaker/consultation fees from Lundbeck, Janssen, Alkermes,Neumora Therapeutics, Boehringer Ingelheim, Sage, Biogen, Mitsubishi Tanabe, Purdue, Pfizer, Otsuka, Takeda, Neurocrine, Sunovion, Bausch Health, Axsome, Novo Nordisk, Kris, Sanofi, Eisai, Intra-Cellular, NewBridge Pharmaceuticals, Viatris, AbbVie, and Atai Life Sciences; and is a CEO of Braxia Scientific Corp.
A version of this article first appeared on Medscape.com.
FROM THE JOURNAL OF CLINICAL PSYCHIATRY
Medical student well-being during the COVID-19 pandemic
During the initial stage of the COVID-19 pandemic U.S. medical students were suspended from in-person clinical interaction. This decision was based on specific guidance from the Association of American Medical Colleges and subsequently implemented in medical schools across the United States.1 Our research project addressed students’ stress level before and after clinical in-person suspension and assessed medical students perceived COVID-19–related risk level. We were particularly curious to learn about students’ emotional struggles as they navigated the initial pedagogical uncertainty associated with the pandemic.
It is likely that heightened stress was greater than before and the rationale was likely multifactorial in nature.2
One key stressor U.S. medical students faced was the negative impacts of COVID-19 on medical education. U.S. Medical Licensing Examination exam-taking was severely impacted, and some students needed to reschedule their test dates because of increased restrictions at testing centers. Third-year medical students in particular were worried about how COVID-19 would influence their residency application; for example, in-person residency interviews and away rotations as fourth-year medical students. Another concern was not being able to be involved in clinical work during the direst stage of this public health emergency because of personal protective equipment shortages and attempts to reduce community spread of COVID-19.
Our study also showed that students had a relatively lower perceived risk level when it comes to COVID-19 than health care workers in the 2003 SARS epidemic, which we suspect is mostly attributable to the suspension of clinical in-person interaction. We also found that female gender and self-reported mental illness diagnosis were two risk factors for perceived stress level, consistent with our current literature.
The reality of clinical in-person inaction caused by PPE shortage and limited telehealth options, together with social isolation and uncertainty regarding future education opportunities, appear to have had a detrimental effect on medical students’ psychological wellbeing. This did not have to be the case. Some medical students found innovative ways to stay involved.
For example, in 2020 some of Dr. Zhang’s classmates helped proctor virtual group therapy sessions held by the local National Alliance on Mental Illness chapter. Medical students at the Icahn School of Medicine at Mount Sinai, New York were not only able to engage in telehealth but also join other task forces, such as PPE supply, distribution, and coordination, morale promotion, and administrative services.3 Finally, many medical students in New York volunteered in providing child care for frontline doctors to help relieve their burden.4 These actions, if implemented more widely, may have had a protective effect on the stress and well-being of medical students at that time.
While our study focused on the academic side of things, the personal impacts from COVID-19 need to be acknowledged – sickness from COVID-19 and its sequelae, family loss fromCOVID-19, financial struggle, and racial targeting of Asians to name a few. COVID-19 has influenced many families’ livelihood and changed our understanding of ourselves, others, and the world in unprecedented ways.
Fast forward to today – medical students are used to learning and living in a world with an ongoing pandemic, and medical education and residency application process have adapted to this new normal. The once-crippling uncertainty surrounding COVID-19 and disastrous PPE shortages have passed. Yet, COVID-19 continues to be a stressor. In fact, burnout related to “COVID-19 fatigue” has been on the rise and one recent national survey shows one in five physicians intends to leave practice within 2 years.5
Meanwhile, uncertainty continued to persist, as in August 2022 monkeypox was declared a public health emergency in the United States.6 What Dr. Zhang learned as a medical student during the initial months of COVID-19 continues to be relevant: connect with loved ones, understand the changing reality, process the emotions, recognize what is under one’s control, have a solution-oriented mindset, and be forgiving and patient with oneself and others.
Dr. Zhang is a second-year psychiatry resident physician at Saint Elizabeth’s Hospital/DC DBH, Washington. Dr. Himelhoch serves as professor and chair of the department of psychiatry at the University of Kentucky, Lexington. His research focuses on developing and studying the efficacy of innovative strategies aimed at improving the health and welfare among people with co-occurring psychiatric and substance use disorders.
References
1. Association of American Medical Colleges. Important Guidance for Medical Students on Clinical Rotations During the Coronavirus (COVID-19) Outbreak. 2020 Mar 17.
2. Zhang Y et al. Psychiatry Res. 2022;313:114595. doi: 10.1016/j.psychres.2022.114595.
3. Bahethi RR et al. Acad Med. 2021 Jun 1;96(6):859-63. doi: 10.1097/ACM.0000000000003863.
4. Krieger P and Goodnough A. Medical Students, Sidelined for Now, Find New Ways to Fight Coronavirus. The New York Times. 2020 Mar 23.
5. Abbasi J. JAMA. 2022 Apr 19;327(15):1435-7. doi: 10.1001/jama.2022.5074.
6. Department of Health & Human Services. Biden-Harris Administration Bolsters Monkeypox Response; HHS Secretary Becerra Declares Public Health Emergency. 2022 Aug 4.
During the initial stage of the COVID-19 pandemic U.S. medical students were suspended from in-person clinical interaction. This decision was based on specific guidance from the Association of American Medical Colleges and subsequently implemented in medical schools across the United States.1 Our research project addressed students’ stress level before and after clinical in-person suspension and assessed medical students perceived COVID-19–related risk level. We were particularly curious to learn about students’ emotional struggles as they navigated the initial pedagogical uncertainty associated with the pandemic.
It is likely that heightened stress was greater than before and the rationale was likely multifactorial in nature.2
One key stressor U.S. medical students faced was the negative impacts of COVID-19 on medical education. U.S. Medical Licensing Examination exam-taking was severely impacted, and some students needed to reschedule their test dates because of increased restrictions at testing centers. Third-year medical students in particular were worried about how COVID-19 would influence their residency application; for example, in-person residency interviews and away rotations as fourth-year medical students. Another concern was not being able to be involved in clinical work during the direst stage of this public health emergency because of personal protective equipment shortages and attempts to reduce community spread of COVID-19.
Our study also showed that students had a relatively lower perceived risk level when it comes to COVID-19 than health care workers in the 2003 SARS epidemic, which we suspect is mostly attributable to the suspension of clinical in-person interaction. We also found that female gender and self-reported mental illness diagnosis were two risk factors for perceived stress level, consistent with our current literature.
The reality of clinical in-person inaction caused by PPE shortage and limited telehealth options, together with social isolation and uncertainty regarding future education opportunities, appear to have had a detrimental effect on medical students’ psychological wellbeing. This did not have to be the case. Some medical students found innovative ways to stay involved.
For example, in 2020 some of Dr. Zhang’s classmates helped proctor virtual group therapy sessions held by the local National Alliance on Mental Illness chapter. Medical students at the Icahn School of Medicine at Mount Sinai, New York were not only able to engage in telehealth but also join other task forces, such as PPE supply, distribution, and coordination, morale promotion, and administrative services.3 Finally, many medical students in New York volunteered in providing child care for frontline doctors to help relieve their burden.4 These actions, if implemented more widely, may have had a protective effect on the stress and well-being of medical students at that time.
While our study focused on the academic side of things, the personal impacts from COVID-19 need to be acknowledged – sickness from COVID-19 and its sequelae, family loss fromCOVID-19, financial struggle, and racial targeting of Asians to name a few. COVID-19 has influenced many families’ livelihood and changed our understanding of ourselves, others, and the world in unprecedented ways.
Fast forward to today – medical students are used to learning and living in a world with an ongoing pandemic, and medical education and residency application process have adapted to this new normal. The once-crippling uncertainty surrounding COVID-19 and disastrous PPE shortages have passed. Yet, COVID-19 continues to be a stressor. In fact, burnout related to “COVID-19 fatigue” has been on the rise and one recent national survey shows one in five physicians intends to leave practice within 2 years.5
Meanwhile, uncertainty continued to persist, as in August 2022 monkeypox was declared a public health emergency in the United States.6 What Dr. Zhang learned as a medical student during the initial months of COVID-19 continues to be relevant: connect with loved ones, understand the changing reality, process the emotions, recognize what is under one’s control, have a solution-oriented mindset, and be forgiving and patient with oneself and others.
Dr. Zhang is a second-year psychiatry resident physician at Saint Elizabeth’s Hospital/DC DBH, Washington. Dr. Himelhoch serves as professor and chair of the department of psychiatry at the University of Kentucky, Lexington. His research focuses on developing and studying the efficacy of innovative strategies aimed at improving the health and welfare among people with co-occurring psychiatric and substance use disorders.
References
1. Association of American Medical Colleges. Important Guidance for Medical Students on Clinical Rotations During the Coronavirus (COVID-19) Outbreak. 2020 Mar 17.
2. Zhang Y et al. Psychiatry Res. 2022;313:114595. doi: 10.1016/j.psychres.2022.114595.
3. Bahethi RR et al. Acad Med. 2021 Jun 1;96(6):859-63. doi: 10.1097/ACM.0000000000003863.
4. Krieger P and Goodnough A. Medical Students, Sidelined for Now, Find New Ways to Fight Coronavirus. The New York Times. 2020 Mar 23.
5. Abbasi J. JAMA. 2022 Apr 19;327(15):1435-7. doi: 10.1001/jama.2022.5074.
6. Department of Health & Human Services. Biden-Harris Administration Bolsters Monkeypox Response; HHS Secretary Becerra Declares Public Health Emergency. 2022 Aug 4.
During the initial stage of the COVID-19 pandemic U.S. medical students were suspended from in-person clinical interaction. This decision was based on specific guidance from the Association of American Medical Colleges and subsequently implemented in medical schools across the United States.1 Our research project addressed students’ stress level before and after clinical in-person suspension and assessed medical students perceived COVID-19–related risk level. We were particularly curious to learn about students’ emotional struggles as they navigated the initial pedagogical uncertainty associated with the pandemic.
It is likely that heightened stress was greater than before and the rationale was likely multifactorial in nature.2
One key stressor U.S. medical students faced was the negative impacts of COVID-19 on medical education. U.S. Medical Licensing Examination exam-taking was severely impacted, and some students needed to reschedule their test dates because of increased restrictions at testing centers. Third-year medical students in particular were worried about how COVID-19 would influence their residency application; for example, in-person residency interviews and away rotations as fourth-year medical students. Another concern was not being able to be involved in clinical work during the direst stage of this public health emergency because of personal protective equipment shortages and attempts to reduce community spread of COVID-19.
Our study also showed that students had a relatively lower perceived risk level when it comes to COVID-19 than health care workers in the 2003 SARS epidemic, which we suspect is mostly attributable to the suspension of clinical in-person interaction. We also found that female gender and self-reported mental illness diagnosis were two risk factors for perceived stress level, consistent with our current literature.
The reality of clinical in-person inaction caused by PPE shortage and limited telehealth options, together with social isolation and uncertainty regarding future education opportunities, appear to have had a detrimental effect on medical students’ psychological wellbeing. This did not have to be the case. Some medical students found innovative ways to stay involved.
For example, in 2020 some of Dr. Zhang’s classmates helped proctor virtual group therapy sessions held by the local National Alliance on Mental Illness chapter. Medical students at the Icahn School of Medicine at Mount Sinai, New York were not only able to engage in telehealth but also join other task forces, such as PPE supply, distribution, and coordination, morale promotion, and administrative services.3 Finally, many medical students in New York volunteered in providing child care for frontline doctors to help relieve their burden.4 These actions, if implemented more widely, may have had a protective effect on the stress and well-being of medical students at that time.
While our study focused on the academic side of things, the personal impacts from COVID-19 need to be acknowledged – sickness from COVID-19 and its sequelae, family loss fromCOVID-19, financial struggle, and racial targeting of Asians to name a few. COVID-19 has influenced many families’ livelihood and changed our understanding of ourselves, others, and the world in unprecedented ways.
Fast forward to today – medical students are used to learning and living in a world with an ongoing pandemic, and medical education and residency application process have adapted to this new normal. The once-crippling uncertainty surrounding COVID-19 and disastrous PPE shortages have passed. Yet, COVID-19 continues to be a stressor. In fact, burnout related to “COVID-19 fatigue” has been on the rise and one recent national survey shows one in five physicians intends to leave practice within 2 years.5
Meanwhile, uncertainty continued to persist, as in August 2022 monkeypox was declared a public health emergency in the United States.6 What Dr. Zhang learned as a medical student during the initial months of COVID-19 continues to be relevant: connect with loved ones, understand the changing reality, process the emotions, recognize what is under one’s control, have a solution-oriented mindset, and be forgiving and patient with oneself and others.
Dr. Zhang is a second-year psychiatry resident physician at Saint Elizabeth’s Hospital/DC DBH, Washington. Dr. Himelhoch serves as professor and chair of the department of psychiatry at the University of Kentucky, Lexington. His research focuses on developing and studying the efficacy of innovative strategies aimed at improving the health and welfare among people with co-occurring psychiatric and substance use disorders.
References
1. Association of American Medical Colleges. Important Guidance for Medical Students on Clinical Rotations During the Coronavirus (COVID-19) Outbreak. 2020 Mar 17.
2. Zhang Y et al. Psychiatry Res. 2022;313:114595. doi: 10.1016/j.psychres.2022.114595.
3. Bahethi RR et al. Acad Med. 2021 Jun 1;96(6):859-63. doi: 10.1097/ACM.0000000000003863.
4. Krieger P and Goodnough A. Medical Students, Sidelined for Now, Find New Ways to Fight Coronavirus. The New York Times. 2020 Mar 23.
5. Abbasi J. JAMA. 2022 Apr 19;327(15):1435-7. doi: 10.1001/jama.2022.5074.
6. Department of Health & Human Services. Biden-Harris Administration Bolsters Monkeypox Response; HHS Secretary Becerra Declares Public Health Emergency. 2022 Aug 4.
More evidence suicidal thoughts, behaviors are genetically based
“It’s really important for us to continue to study the genetic risk factors for suicidal behaviors so we can really understand the biology and develop better treatments,” study investigator Allison E. Ashley-Koch, PhD, professor in the department of medicine at Duke University Medical Center, Durham, N.C., told this news organization.
The findings were published online in JAMA Psychiatry).
SITB heritability
Suicide is a leading cause of death, particularly among individuals aged 15-29 years. Whereas the global rate of suicide has decreased by 36% in the past 20 years, the rate in the United States has increased by 35%, with the greatest rise in military veterans.
Twin studies suggest heritability for SITB is between 30% and 55%, but the molecular genetic basis of SITB remains elusive.
To address this research gap, investigators conducted a study of 633,778 U.S. military veterans from the Million Veteran Program (MVP) cohort. Of these, 71% had European ancestry, 19% had African ancestry, 8% were Hispanic, and 1% were Asian. Just under 10% of the sample was female.
Study participants donated a blood sample and agreed to have their genetic information linked with their electronic health record data.
From diagnostic codes and other sources, researchers identified 121,211 individuals with SITB. They classified participants with no documented lifetime history of suicidal ideation, suicide attempt, or suicide death as controls.
Rates of SITB differed significantly by ancestry – 25% in those with African or Hispanic ancestry, 21% in those with Asian ancestry, and 16.8% in those with European ancestry. Rates also differed by age and sex; those with SITB were younger and more likely to be female.
In addition to age and sex, covariates included “genetic principal components,” which Dr. Ashley-Koch said accounts for combining data of individuals with different ethnic/racial backgrounds.
Through meta-analysis, the investigators identified seven genome-wide, significant cross-ancestry risk loci.
To evaluate whether the findings could be replicated, researchers used the International Suicide Genetics Consortium (ISGC), a primarily civilian international consortium of roughly 550,000 individuals of mostly European ancestry.
The analysis showed the top replicated cross-ancestry risk locus was rs6557168, an intronic single-nucleotide variant (SNV) in the ESR1 gene that encodes an estrogen receptor. Previous work identified ESR1 as a causal genetic driver gene for development of posttraumatic stress disorder and depression, both of which are risk factors for SITB among veterans.
The second-strongest replicated cross-ancestry locus was rs12808482, an intronic variant in the DRD2 gene, which encodes the D2 dopamine-receptor subtype. The authors noted DRD2 is highly expressed in brain tissue and has been associated with numerous psychiatric phenotypes.
Research suggests DRD2 is associated with other risk factors for SITB, such as schizophrenia, mood disorders, and attention-deficit/hyperactivity disorder, but DRD2 could also contribute to suicide risk directly. The authors noted it is highly expressed in the prefrontal cortex, nucleus accumbens, substantia nigra, and hippocampus.
Outstanding candidate gene
The study revealed a cross-ancestry GWS association for rs10671545, a variant in DCC, which is “also an outstanding candidate gene,” the investigators write.
They note it is expressed in brain tissue, is involved in synaptic plasticity, axon guidance, and circadian entrainment, and has been associated with multiple psychiatric phenotypes.
Researchers also found what they called “intriguing” cross-ancestry GWS associations for the TRAF3 gene, which regulates type-1 interferon production. Many patients receiving interferon therapy develop major depressive disorder and suicidal ideation.
TRAF3 is also associated with antisocial behavior, substance use, and ADHD. Lithium – a standard treatment for bipolar disorder that reduces suicide risk – modulates the expression of TRAF3.
Dr. Ashley-Koch noted the replication of these loci (ESR1, DRD2, TRAF3, and DCC) was in a population of mostly White civilians. “This suggests to us that at least some of the risk for suicidal thoughts and behaviors does cross ancestry and also crosses military and civilian populations.”
It was “exciting” that all four genes the study focused on had previously been implicated in other psychiatric disorders, said Dr. Ashley-Koch. “What gave us a little more confidence, above and beyond the replication, was that these genes are somehow important for psychiatric disorders, and not any psychiatric disorders, but the ones that are also associated with a high risk of suicide behavior, such as depression, PTSD, schizophrenia, and ADHD.”
The findings will not have an immediate impact on clinical practice, said Dr. Ashley-Koch.
“We need to take the next step, which is to try to understand how these genetic factors may impact risk and what else is happening biologically to increase that risk. Then once we do that, hopefully we can develop some new treatments,” she added.
‘Valuable and noble’ research
Commenting on the study, Elspeth Cameron Ritchie, MD, chief of psychiatry at Medstar Washington Hospital Center, Washington, said this kind of genetic research is “valuable and noble.”
Researchers have long been interested in risk factors for suicide among military personnel and veterans, said Dr. Ritchie. Evidence to date suggests being a young male is a risk factor as is feeling excluded or not fitting into the unit, and drug or alcohol addiction.
Dr. Ritchie noted other psychiatric disorders, including schizophrenia, depression, and bipolar disorder, are at least partially inherited.
She noted the study’s findings should not be used to discriminate against those who might have the identified genetic loci without clearer evidence of their impact.
“If we were able to identify these genes, would we start screening everybody who joins the military to see if they have these genes, and how would that impact the ability to recruit or retain personnel?”
She agreed additional work is needed to determine if and how carrying these genes might impact clinical care.
In addition, she pointed out that behavior is influenced not only by genetic load but also by environment. “This study may show the impact of the genetic load a little bit more clearly; right now, we tend to look at environmental factors.”
The study was supported by an award from the Clinical Science Research and Development (CSR&D) service of the Veterans Health Administration’s Office of Research and Development. The work was also supported in part by the joint U.S. Department of Veterans Affairs and U.S. Department of Energy MVP CHAMPION program.
Dr. Ashley-Koch reported grants from Veterans Administration during the conduct of the study. Several other coauthors report relationships with industry, nonprofit organizations, and government agencies. The full list can be found with the original article. Dr. Ritchie reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
“It’s really important for us to continue to study the genetic risk factors for suicidal behaviors so we can really understand the biology and develop better treatments,” study investigator Allison E. Ashley-Koch, PhD, professor in the department of medicine at Duke University Medical Center, Durham, N.C., told this news organization.
The findings were published online in JAMA Psychiatry).
SITB heritability
Suicide is a leading cause of death, particularly among individuals aged 15-29 years. Whereas the global rate of suicide has decreased by 36% in the past 20 years, the rate in the United States has increased by 35%, with the greatest rise in military veterans.
Twin studies suggest heritability for SITB is between 30% and 55%, but the molecular genetic basis of SITB remains elusive.
To address this research gap, investigators conducted a study of 633,778 U.S. military veterans from the Million Veteran Program (MVP) cohort. Of these, 71% had European ancestry, 19% had African ancestry, 8% were Hispanic, and 1% were Asian. Just under 10% of the sample was female.
Study participants donated a blood sample and agreed to have their genetic information linked with their electronic health record data.
From diagnostic codes and other sources, researchers identified 121,211 individuals with SITB. They classified participants with no documented lifetime history of suicidal ideation, suicide attempt, or suicide death as controls.
Rates of SITB differed significantly by ancestry – 25% in those with African or Hispanic ancestry, 21% in those with Asian ancestry, and 16.8% in those with European ancestry. Rates also differed by age and sex; those with SITB were younger and more likely to be female.
In addition to age and sex, covariates included “genetic principal components,” which Dr. Ashley-Koch said accounts for combining data of individuals with different ethnic/racial backgrounds.
Through meta-analysis, the investigators identified seven genome-wide, significant cross-ancestry risk loci.
To evaluate whether the findings could be replicated, researchers used the International Suicide Genetics Consortium (ISGC), a primarily civilian international consortium of roughly 550,000 individuals of mostly European ancestry.
The analysis showed the top replicated cross-ancestry risk locus was rs6557168, an intronic single-nucleotide variant (SNV) in the ESR1 gene that encodes an estrogen receptor. Previous work identified ESR1 as a causal genetic driver gene for development of posttraumatic stress disorder and depression, both of which are risk factors for SITB among veterans.
The second-strongest replicated cross-ancestry locus was rs12808482, an intronic variant in the DRD2 gene, which encodes the D2 dopamine-receptor subtype. The authors noted DRD2 is highly expressed in brain tissue and has been associated with numerous psychiatric phenotypes.
Research suggests DRD2 is associated with other risk factors for SITB, such as schizophrenia, mood disorders, and attention-deficit/hyperactivity disorder, but DRD2 could also contribute to suicide risk directly. The authors noted it is highly expressed in the prefrontal cortex, nucleus accumbens, substantia nigra, and hippocampus.
Outstanding candidate gene
The study revealed a cross-ancestry GWS association for rs10671545, a variant in DCC, which is “also an outstanding candidate gene,” the investigators write.
They note it is expressed in brain tissue, is involved in synaptic plasticity, axon guidance, and circadian entrainment, and has been associated with multiple psychiatric phenotypes.
Researchers also found what they called “intriguing” cross-ancestry GWS associations for the TRAF3 gene, which regulates type-1 interferon production. Many patients receiving interferon therapy develop major depressive disorder and suicidal ideation.
TRAF3 is also associated with antisocial behavior, substance use, and ADHD. Lithium – a standard treatment for bipolar disorder that reduces suicide risk – modulates the expression of TRAF3.
Dr. Ashley-Koch noted the replication of these loci (ESR1, DRD2, TRAF3, and DCC) was in a population of mostly White civilians. “This suggests to us that at least some of the risk for suicidal thoughts and behaviors does cross ancestry and also crosses military and civilian populations.”
It was “exciting” that all four genes the study focused on had previously been implicated in other psychiatric disorders, said Dr. Ashley-Koch. “What gave us a little more confidence, above and beyond the replication, was that these genes are somehow important for psychiatric disorders, and not any psychiatric disorders, but the ones that are also associated with a high risk of suicide behavior, such as depression, PTSD, schizophrenia, and ADHD.”
The findings will not have an immediate impact on clinical practice, said Dr. Ashley-Koch.
“We need to take the next step, which is to try to understand how these genetic factors may impact risk and what else is happening biologically to increase that risk. Then once we do that, hopefully we can develop some new treatments,” she added.
‘Valuable and noble’ research
Commenting on the study, Elspeth Cameron Ritchie, MD, chief of psychiatry at Medstar Washington Hospital Center, Washington, said this kind of genetic research is “valuable and noble.”
Researchers have long been interested in risk factors for suicide among military personnel and veterans, said Dr. Ritchie. Evidence to date suggests being a young male is a risk factor as is feeling excluded or not fitting into the unit, and drug or alcohol addiction.
Dr. Ritchie noted other psychiatric disorders, including schizophrenia, depression, and bipolar disorder, are at least partially inherited.
She noted the study’s findings should not be used to discriminate against those who might have the identified genetic loci without clearer evidence of their impact.
“If we were able to identify these genes, would we start screening everybody who joins the military to see if they have these genes, and how would that impact the ability to recruit or retain personnel?”
She agreed additional work is needed to determine if and how carrying these genes might impact clinical care.
In addition, she pointed out that behavior is influenced not only by genetic load but also by environment. “This study may show the impact of the genetic load a little bit more clearly; right now, we tend to look at environmental factors.”
The study was supported by an award from the Clinical Science Research and Development (CSR&D) service of the Veterans Health Administration’s Office of Research and Development. The work was also supported in part by the joint U.S. Department of Veterans Affairs and U.S. Department of Energy MVP CHAMPION program.
Dr. Ashley-Koch reported grants from Veterans Administration during the conduct of the study. Several other coauthors report relationships with industry, nonprofit organizations, and government agencies. The full list can be found with the original article. Dr. Ritchie reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
“It’s really important for us to continue to study the genetic risk factors for suicidal behaviors so we can really understand the biology and develop better treatments,” study investigator Allison E. Ashley-Koch, PhD, professor in the department of medicine at Duke University Medical Center, Durham, N.C., told this news organization.
The findings were published online in JAMA Psychiatry).
SITB heritability
Suicide is a leading cause of death, particularly among individuals aged 15-29 years. Whereas the global rate of suicide has decreased by 36% in the past 20 years, the rate in the United States has increased by 35%, with the greatest rise in military veterans.
Twin studies suggest heritability for SITB is between 30% and 55%, but the molecular genetic basis of SITB remains elusive.
To address this research gap, investigators conducted a study of 633,778 U.S. military veterans from the Million Veteran Program (MVP) cohort. Of these, 71% had European ancestry, 19% had African ancestry, 8% were Hispanic, and 1% were Asian. Just under 10% of the sample was female.
Study participants donated a blood sample and agreed to have their genetic information linked with their electronic health record data.
From diagnostic codes and other sources, researchers identified 121,211 individuals with SITB. They classified participants with no documented lifetime history of suicidal ideation, suicide attempt, or suicide death as controls.
Rates of SITB differed significantly by ancestry – 25% in those with African or Hispanic ancestry, 21% in those with Asian ancestry, and 16.8% in those with European ancestry. Rates also differed by age and sex; those with SITB were younger and more likely to be female.
In addition to age and sex, covariates included “genetic principal components,” which Dr. Ashley-Koch said accounts for combining data of individuals with different ethnic/racial backgrounds.
Through meta-analysis, the investigators identified seven genome-wide, significant cross-ancestry risk loci.
To evaluate whether the findings could be replicated, researchers used the International Suicide Genetics Consortium (ISGC), a primarily civilian international consortium of roughly 550,000 individuals of mostly European ancestry.
The analysis showed the top replicated cross-ancestry risk locus was rs6557168, an intronic single-nucleotide variant (SNV) in the ESR1 gene that encodes an estrogen receptor. Previous work identified ESR1 as a causal genetic driver gene for development of posttraumatic stress disorder and depression, both of which are risk factors for SITB among veterans.
The second-strongest replicated cross-ancestry locus was rs12808482, an intronic variant in the DRD2 gene, which encodes the D2 dopamine-receptor subtype. The authors noted DRD2 is highly expressed in brain tissue and has been associated with numerous psychiatric phenotypes.
Research suggests DRD2 is associated with other risk factors for SITB, such as schizophrenia, mood disorders, and attention-deficit/hyperactivity disorder, but DRD2 could also contribute to suicide risk directly. The authors noted it is highly expressed in the prefrontal cortex, nucleus accumbens, substantia nigra, and hippocampus.
Outstanding candidate gene
The study revealed a cross-ancestry GWS association for rs10671545, a variant in DCC, which is “also an outstanding candidate gene,” the investigators write.
They note it is expressed in brain tissue, is involved in synaptic plasticity, axon guidance, and circadian entrainment, and has been associated with multiple psychiatric phenotypes.
Researchers also found what they called “intriguing” cross-ancestry GWS associations for the TRAF3 gene, which regulates type-1 interferon production. Many patients receiving interferon therapy develop major depressive disorder and suicidal ideation.
TRAF3 is also associated with antisocial behavior, substance use, and ADHD. Lithium – a standard treatment for bipolar disorder that reduces suicide risk – modulates the expression of TRAF3.
Dr. Ashley-Koch noted the replication of these loci (ESR1, DRD2, TRAF3, and DCC) was in a population of mostly White civilians. “This suggests to us that at least some of the risk for suicidal thoughts and behaviors does cross ancestry and also crosses military and civilian populations.”
It was “exciting” that all four genes the study focused on had previously been implicated in other psychiatric disorders, said Dr. Ashley-Koch. “What gave us a little more confidence, above and beyond the replication, was that these genes are somehow important for psychiatric disorders, and not any psychiatric disorders, but the ones that are also associated with a high risk of suicide behavior, such as depression, PTSD, schizophrenia, and ADHD.”
The findings will not have an immediate impact on clinical practice, said Dr. Ashley-Koch.
“We need to take the next step, which is to try to understand how these genetic factors may impact risk and what else is happening biologically to increase that risk. Then once we do that, hopefully we can develop some new treatments,” she added.
‘Valuable and noble’ research
Commenting on the study, Elspeth Cameron Ritchie, MD, chief of psychiatry at Medstar Washington Hospital Center, Washington, said this kind of genetic research is “valuable and noble.”
Researchers have long been interested in risk factors for suicide among military personnel and veterans, said Dr. Ritchie. Evidence to date suggests being a young male is a risk factor as is feeling excluded or not fitting into the unit, and drug or alcohol addiction.
Dr. Ritchie noted other psychiatric disorders, including schizophrenia, depression, and bipolar disorder, are at least partially inherited.
She noted the study’s findings should not be used to discriminate against those who might have the identified genetic loci without clearer evidence of their impact.
“If we were able to identify these genes, would we start screening everybody who joins the military to see if they have these genes, and how would that impact the ability to recruit or retain personnel?”
She agreed additional work is needed to determine if and how carrying these genes might impact clinical care.
In addition, she pointed out that behavior is influenced not only by genetic load but also by environment. “This study may show the impact of the genetic load a little bit more clearly; right now, we tend to look at environmental factors.”
The study was supported by an award from the Clinical Science Research and Development (CSR&D) service of the Veterans Health Administration’s Office of Research and Development. The work was also supported in part by the joint U.S. Department of Veterans Affairs and U.S. Department of Energy MVP CHAMPION program.
Dr. Ashley-Koch reported grants from Veterans Administration during the conduct of the study. Several other coauthors report relationships with industry, nonprofit organizations, and government agencies. The full list can be found with the original article. Dr. Ritchie reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM JAMA PSYCHIATRY
Is the FDA serotonin syndrome warning unnecessary?
Results from a study that included more than 1,100 patients who were prescribed linezolid, about 20% of whom were also taking antidepressants, showed that serotonin syndrome occurred in fewer than 0.5% of participants – and that the percentage was actually lower among those who took antidepressants, compared with those who did not.
A comparison of participants who took antidepressants to propensity-matched patients who did not take antidepressants showed similar rates of altered mental status, hospitalization, and death between the two groups.
“In this cohort study of older patients who were prescribed linezolid, serotonin syndrome occurred rarely [and] concurrent antidepressants did not significantly increase the risk of serotonin syndrome,” Anthony Bai, MD, division of infectious diseases, department of medicine, Queen’s University, Kingston, Ont., and colleagues write.
“These findings suggested that linezolid is likely safe for patients receiving antidepressants. Nevertheless, prescribers should remain vigilant for this potential drug interaction,” they warn.
The findings were published online in JAMA Network Open.
Scarce data
Linezolid, a synthetic oxazolidinone antibiotic active against resistant gram-positive bacteria, has bioavailability of 100%, “making it ideal as first-line or step-down oral antibiotic therapy for bacteremia and pneumonia as well as skin and soft tissue infections,” the researchers write.
However, they note its use has been “limited because of concerns of drug interactions,” since it can reversibly inhibit monoamine oxidase (MAO).
Thus, “coadministration with antidepressants, such as nonselective MAO inhibitors, selective serotonin reuptake inhibitors (SSRIs), serotonin-norepinephrine reuptake inhibitors (SNRIs), and bupropion, may precipitate serotonin syndrome,” they write.
The investigators note that many patients who were taking antidepressants and who also needed linezolid for an infection “could not receive it because of this relative contraindication.” They add that data on the risk of serotonin syndrome associated with linezolid are “scarce” and are based largely on case reports or case series from passive surveillance.
Although a previous review of linezolid trials found “no conclusive evidence” that it increased risk for serotonin syndrome in patients taking serotonergic medication, data on patients outside of trials “are lacking.” In addition, an observational study suggested that an increased risk had a small sample size that “likely led to imprecise estimates with a wide CI and inconclusive results,” the researchers write.
Therefore, they sought to fill the knowledge gap by retrospectively analyzing data drawn from the ICES database, an independent nonprofit research institute funded by the Ontario Ministry of Health. This was done in order to “estimate the incidence of serotonin syndrome and how this risk changes because of concomitant antidepressant use in patients receiving linezolid treatment,” they write.
The study included a convenience sample of Ontario-based adults (n = 1,134, 52.5% men) who were dispensed oral linezolid 600 mg twice daily between Oct. 1, 2014, and Jan. 1, 2021. All patients were followed for 30 days.
Of these participants, 19% were also taking antidepressants. Close to half (47.9%) were taking an SSRI, 16.7% were taking an SNRI, 7% were taking a tricyclic antidepressant, and 3.3% were taking a norepinephrine and dopamine reuptake inhibitor.
Patients were divided into groups on the basis of age: 66-69 years (19.8%), 70-79 years (41.7%), and 80 years or older (38.4%).
Reassuring findings
Serotonin syndrome occurred in fewer than six patients (< .5%), although the exact numbers were not reported, owing to patient privacy concerns. However, on the basis of fewer than six events, the investigators calculated the risk difference for serotonin syndrome as ranging from −0.5% to 2.3%.
Fewer patients who were taking antidepressants experienced serotonin syndrome, compared with those who were not taking antidepressants.
The investigators estimated a propensity score for antidepressant use that incorporated several patient baseline characteristics, including age, sex, rural home address, Charlson Comorbidity Index, estimated glomerular filtration rate, history of substance use disorder, and days of use of linezolid and other serotonergic medications. They then matched patients who were not taking antidepressants with those who were taking antidepressants (n = 166 each).
The adjusted risk difference for serotonin syndrome was lower in the antidepressant group than in the no-antidepressant group (−1.2%; 95% confidence interval, −2.9% to 0.5%).
“Within this 95% CI, the worst-case scenario would be a 0.5% increase in the risk of serotonin syndrome due to antidepressants, which is equivalent to a number needed to harm of 200,” the researchers write.
For secondary outcomes, they found “similar rates” of altered mental status or confusion, hospitalization, and death within 30 days between the two propensity score–matched groups.
The investigators note that their findings have “limitations, due to the nature of retrospective observational studies.” Moreover, these types of studies are “not efficient because they often focus on a particular adverse event.”
Future research should move beyond observational studies to phase 4 studies, which would “prospectively monitor for all types of adverse events,” they write.
Still, “while waiting for higher-quality evidence, our study adds to the existing evidence for the safety of linezolid even in the context of concomitant antidepressants,” the researchers note.
“Based on the existing evidence, clinicians should be reassured that it appears safe to prescribe oral linezolid to patients taking antidepressants, especially if there are limited antibiotic options or alternative antibiotic options would be inferior,” they add.
‘Consequential relevance’
Commenting on the study, Ipsit Vahia, MD, associate chief of geriatric psychiatry and director of digital psychiatry translation at McLean Hospital, Boston, noted that although studies of drug interactions across age groups “may not accurately reflect the rates of risk for older adults,” the current study focused on linezolid use among older patients.
“One may expect higher rates of serotonin syndrome in older adults, who generally tend to be more sensitive to adverse reactions,” said Dr. Vahia, who is also director of the Technology and Aging Lab at McLean and was not involved with the current research.
“However, the study finds the risk to be low with a number needed to harm of 200,” Dr. Vahia said.
“This retrospective epidemiologic study does not shed light on why this number may be lower than expected, but it has consequential relevance in clinical practice for the management of severe infections among older adults using antidepressants,” he added.
The study was funded by a Queen’s University Research Initiation Grant. Dr. Bai and three of the four other investigators report no relevant financial relationships. Coinvestigator Mark Loeb, MD, reports having received personal fees from the Paladin Labs Advisory Committee, the International Centre for Professional Development in Health and Medicine Advisory Committee, and the Sunovion Advisory Committee outside the submitted work. Dr. Vahia serves as a consultant for Otsuka, has a research collaboration with Emerald Innovations, and receives honorarium as editor for The American Journal of Geriatric Psychiatry.
A version of this article first appeared on Medscape.com.
Results from a study that included more than 1,100 patients who were prescribed linezolid, about 20% of whom were also taking antidepressants, showed that serotonin syndrome occurred in fewer than 0.5% of participants – and that the percentage was actually lower among those who took antidepressants, compared with those who did not.
A comparison of participants who took antidepressants to propensity-matched patients who did not take antidepressants showed similar rates of altered mental status, hospitalization, and death between the two groups.
“In this cohort study of older patients who were prescribed linezolid, serotonin syndrome occurred rarely [and] concurrent antidepressants did not significantly increase the risk of serotonin syndrome,” Anthony Bai, MD, division of infectious diseases, department of medicine, Queen’s University, Kingston, Ont., and colleagues write.
“These findings suggested that linezolid is likely safe for patients receiving antidepressants. Nevertheless, prescribers should remain vigilant for this potential drug interaction,” they warn.
The findings were published online in JAMA Network Open.
Scarce data
Linezolid, a synthetic oxazolidinone antibiotic active against resistant gram-positive bacteria, has bioavailability of 100%, “making it ideal as first-line or step-down oral antibiotic therapy for bacteremia and pneumonia as well as skin and soft tissue infections,” the researchers write.
However, they note its use has been “limited because of concerns of drug interactions,” since it can reversibly inhibit monoamine oxidase (MAO).
Thus, “coadministration with antidepressants, such as nonselective MAO inhibitors, selective serotonin reuptake inhibitors (SSRIs), serotonin-norepinephrine reuptake inhibitors (SNRIs), and bupropion, may precipitate serotonin syndrome,” they write.
The investigators note that many patients who were taking antidepressants and who also needed linezolid for an infection “could not receive it because of this relative contraindication.” They add that data on the risk of serotonin syndrome associated with linezolid are “scarce” and are based largely on case reports or case series from passive surveillance.
Although a previous review of linezolid trials found “no conclusive evidence” that it increased risk for serotonin syndrome in patients taking serotonergic medication, data on patients outside of trials “are lacking.” In addition, an observational study suggested that an increased risk had a small sample size that “likely led to imprecise estimates with a wide CI and inconclusive results,” the researchers write.
Therefore, they sought to fill the knowledge gap by retrospectively analyzing data drawn from the ICES database, an independent nonprofit research institute funded by the Ontario Ministry of Health. This was done in order to “estimate the incidence of serotonin syndrome and how this risk changes because of concomitant antidepressant use in patients receiving linezolid treatment,” they write.
The study included a convenience sample of Ontario-based adults (n = 1,134, 52.5% men) who were dispensed oral linezolid 600 mg twice daily between Oct. 1, 2014, and Jan. 1, 2021. All patients were followed for 30 days.
Of these participants, 19% were also taking antidepressants. Close to half (47.9%) were taking an SSRI, 16.7% were taking an SNRI, 7% were taking a tricyclic antidepressant, and 3.3% were taking a norepinephrine and dopamine reuptake inhibitor.
Patients were divided into groups on the basis of age: 66-69 years (19.8%), 70-79 years (41.7%), and 80 years or older (38.4%).
Reassuring findings
Serotonin syndrome occurred in fewer than six patients (< .5%), although the exact numbers were not reported, owing to patient privacy concerns. However, on the basis of fewer than six events, the investigators calculated the risk difference for serotonin syndrome as ranging from −0.5% to 2.3%.
Fewer patients who were taking antidepressants experienced serotonin syndrome, compared with those who were not taking antidepressants.
The investigators estimated a propensity score for antidepressant use that incorporated several patient baseline characteristics, including age, sex, rural home address, Charlson Comorbidity Index, estimated glomerular filtration rate, history of substance use disorder, and days of use of linezolid and other serotonergic medications. They then matched patients who were not taking antidepressants with those who were taking antidepressants (n = 166 each).
The adjusted risk difference for serotonin syndrome was lower in the antidepressant group than in the no-antidepressant group (−1.2%; 95% confidence interval, −2.9% to 0.5%).
“Within this 95% CI, the worst-case scenario would be a 0.5% increase in the risk of serotonin syndrome due to antidepressants, which is equivalent to a number needed to harm of 200,” the researchers write.
For secondary outcomes, they found “similar rates” of altered mental status or confusion, hospitalization, and death within 30 days between the two propensity score–matched groups.
The investigators note that their findings have “limitations, due to the nature of retrospective observational studies.” Moreover, these types of studies are “not efficient because they often focus on a particular adverse event.”
Future research should move beyond observational studies to phase 4 studies, which would “prospectively monitor for all types of adverse events,” they write.
Still, “while waiting for higher-quality evidence, our study adds to the existing evidence for the safety of linezolid even in the context of concomitant antidepressants,” the researchers note.
“Based on the existing evidence, clinicians should be reassured that it appears safe to prescribe oral linezolid to patients taking antidepressants, especially if there are limited antibiotic options or alternative antibiotic options would be inferior,” they add.
‘Consequential relevance’
Commenting on the study, Ipsit Vahia, MD, associate chief of geriatric psychiatry and director of digital psychiatry translation at McLean Hospital, Boston, noted that although studies of drug interactions across age groups “may not accurately reflect the rates of risk for older adults,” the current study focused on linezolid use among older patients.
“One may expect higher rates of serotonin syndrome in older adults, who generally tend to be more sensitive to adverse reactions,” said Dr. Vahia, who is also director of the Technology and Aging Lab at McLean and was not involved with the current research.
“However, the study finds the risk to be low with a number needed to harm of 200,” Dr. Vahia said.
“This retrospective epidemiologic study does not shed light on why this number may be lower than expected, but it has consequential relevance in clinical practice for the management of severe infections among older adults using antidepressants,” he added.
The study was funded by a Queen’s University Research Initiation Grant. Dr. Bai and three of the four other investigators report no relevant financial relationships. Coinvestigator Mark Loeb, MD, reports having received personal fees from the Paladin Labs Advisory Committee, the International Centre for Professional Development in Health and Medicine Advisory Committee, and the Sunovion Advisory Committee outside the submitted work. Dr. Vahia serves as a consultant for Otsuka, has a research collaboration with Emerald Innovations, and receives honorarium as editor for The American Journal of Geriatric Psychiatry.
A version of this article first appeared on Medscape.com.
Results from a study that included more than 1,100 patients who were prescribed linezolid, about 20% of whom were also taking antidepressants, showed that serotonin syndrome occurred in fewer than 0.5% of participants – and that the percentage was actually lower among those who took antidepressants, compared with those who did not.
A comparison of participants who took antidepressants to propensity-matched patients who did not take antidepressants showed similar rates of altered mental status, hospitalization, and death between the two groups.
“In this cohort study of older patients who were prescribed linezolid, serotonin syndrome occurred rarely [and] concurrent antidepressants did not significantly increase the risk of serotonin syndrome,” Anthony Bai, MD, division of infectious diseases, department of medicine, Queen’s University, Kingston, Ont., and colleagues write.
“These findings suggested that linezolid is likely safe for patients receiving antidepressants. Nevertheless, prescribers should remain vigilant for this potential drug interaction,” they warn.
The findings were published online in JAMA Network Open.
Scarce data
Linezolid, a synthetic oxazolidinone antibiotic active against resistant gram-positive bacteria, has bioavailability of 100%, “making it ideal as first-line or step-down oral antibiotic therapy for bacteremia and pneumonia as well as skin and soft tissue infections,” the researchers write.
However, they note its use has been “limited because of concerns of drug interactions,” since it can reversibly inhibit monoamine oxidase (MAO).
Thus, “coadministration with antidepressants, such as nonselective MAO inhibitors, selective serotonin reuptake inhibitors (SSRIs), serotonin-norepinephrine reuptake inhibitors (SNRIs), and bupropion, may precipitate serotonin syndrome,” they write.
The investigators note that many patients who were taking antidepressants and who also needed linezolid for an infection “could not receive it because of this relative contraindication.” They add that data on the risk of serotonin syndrome associated with linezolid are “scarce” and are based largely on case reports or case series from passive surveillance.
Although a previous review of linezolid trials found “no conclusive evidence” that it increased risk for serotonin syndrome in patients taking serotonergic medication, data on patients outside of trials “are lacking.” In addition, an observational study suggested that an increased risk had a small sample size that “likely led to imprecise estimates with a wide CI and inconclusive results,” the researchers write.
Therefore, they sought to fill the knowledge gap by retrospectively analyzing data drawn from the ICES database, an independent nonprofit research institute funded by the Ontario Ministry of Health. This was done in order to “estimate the incidence of serotonin syndrome and how this risk changes because of concomitant antidepressant use in patients receiving linezolid treatment,” they write.
The study included a convenience sample of Ontario-based adults (n = 1,134, 52.5% men) who were dispensed oral linezolid 600 mg twice daily between Oct. 1, 2014, and Jan. 1, 2021. All patients were followed for 30 days.
Of these participants, 19% were also taking antidepressants. Close to half (47.9%) were taking an SSRI, 16.7% were taking an SNRI, 7% were taking a tricyclic antidepressant, and 3.3% were taking a norepinephrine and dopamine reuptake inhibitor.
Patients were divided into groups on the basis of age: 66-69 years (19.8%), 70-79 years (41.7%), and 80 years or older (38.4%).
Reassuring findings
Serotonin syndrome occurred in fewer than six patients (< .5%), although the exact numbers were not reported, owing to patient privacy concerns. However, on the basis of fewer than six events, the investigators calculated the risk difference for serotonin syndrome as ranging from −0.5% to 2.3%.
Fewer patients who were taking antidepressants experienced serotonin syndrome, compared with those who were not taking antidepressants.
The investigators estimated a propensity score for antidepressant use that incorporated several patient baseline characteristics, including age, sex, rural home address, Charlson Comorbidity Index, estimated glomerular filtration rate, history of substance use disorder, and days of use of linezolid and other serotonergic medications. They then matched patients who were not taking antidepressants with those who were taking antidepressants (n = 166 each).
The adjusted risk difference for serotonin syndrome was lower in the antidepressant group than in the no-antidepressant group (−1.2%; 95% confidence interval, −2.9% to 0.5%).
“Within this 95% CI, the worst-case scenario would be a 0.5% increase in the risk of serotonin syndrome due to antidepressants, which is equivalent to a number needed to harm of 200,” the researchers write.
For secondary outcomes, they found “similar rates” of altered mental status or confusion, hospitalization, and death within 30 days between the two propensity score–matched groups.
The investigators note that their findings have “limitations, due to the nature of retrospective observational studies.” Moreover, these types of studies are “not efficient because they often focus on a particular adverse event.”
Future research should move beyond observational studies to phase 4 studies, which would “prospectively monitor for all types of adverse events,” they write.
Still, “while waiting for higher-quality evidence, our study adds to the existing evidence for the safety of linezolid even in the context of concomitant antidepressants,” the researchers note.
“Based on the existing evidence, clinicians should be reassured that it appears safe to prescribe oral linezolid to patients taking antidepressants, especially if there are limited antibiotic options or alternative antibiotic options would be inferior,” they add.
‘Consequential relevance’
Commenting on the study, Ipsit Vahia, MD, associate chief of geriatric psychiatry and director of digital psychiatry translation at McLean Hospital, Boston, noted that although studies of drug interactions across age groups “may not accurately reflect the rates of risk for older adults,” the current study focused on linezolid use among older patients.
“One may expect higher rates of serotonin syndrome in older adults, who generally tend to be more sensitive to adverse reactions,” said Dr. Vahia, who is also director of the Technology and Aging Lab at McLean and was not involved with the current research.
“However, the study finds the risk to be low with a number needed to harm of 200,” Dr. Vahia said.
“This retrospective epidemiologic study does not shed light on why this number may be lower than expected, but it has consequential relevance in clinical practice for the management of severe infections among older adults using antidepressants,” he added.
The study was funded by a Queen’s University Research Initiation Grant. Dr. Bai and three of the four other investigators report no relevant financial relationships. Coinvestigator Mark Loeb, MD, reports having received personal fees from the Paladin Labs Advisory Committee, the International Centre for Professional Development in Health and Medicine Advisory Committee, and the Sunovion Advisory Committee outside the submitted work. Dr. Vahia serves as a consultant for Otsuka, has a research collaboration with Emerald Innovations, and receives honorarium as editor for The American Journal of Geriatric Psychiatry.
A version of this article first appeared on Medscape.com.
FROM JAMA NETWORK OPEN
IV ketamine a promising option for resistant depression in older adults
Results showed nearly 50% of participants responded to ketamine and 25% achieved complete remission from TRD, as measured by scores on the Montgomery-Asberg Depression Rating Scale (MADRS).
“Our pilot study suggests that IV ketamine is well-tolerated, safe, and associated with improvement in late-life TRD,” co-investigator Marie Anne Gebara, MD, assistant professor of psychiatry at the University of Pittsburgh, told this news organization.
Dr. Gebara pointed out the treatment “may not be appropriate for all patients with TRD,” such as those with a history of psychotic symptoms or uncontrolled hypertension; but “it appears to be a promising option.”
The findings were published online in the American Journal of Geriatric Psychiatry.
Lack of data in seniors
Although ketamine has been shown in prior research to rapidly reduce suicidal ideation in adults, there has been a lack of data on its efficacy and safety in older adults, the current investigators note.
“Almost 50% of older adults suffering from depression have TRD, which is a leading cause of disability, excess mortality from suicide, and dementia,” Dr. Gebara said.
She added that after two failed trials of antidepressants, “older adults have few evidence-based choices: aripiprazole or bupropion augmentation, transcranial magnetic stimulation, or electroconvulsive therapy. Novel treatments with rapid benefit are needed as long-term outcomes are poor and recurrence rates are high.”
Dr. Gebara and colleagues at five sites (Columbia University, New York State Psychiatric Institute, University of Toronto, University of Pittsburgh, and Washington University in St. Louis) each enrolled five participants aged 60 and older into the pilot study between October 2020 and November 2021, for a total of 25 participants (mean age, 71 years).
Each participant was recruited from patient registries or referred by behavioral health or primary care providers and diagnosed with TRD, which was defined as an episode of major depressive disorder without psychotic features that persisted despite two or more trials of antidepressants including at least one evidence-based second-line treatment.
Participants had to take an oral antidepressant dosage for at least 1 month prior to the start of the IV ketamine infusions, and continue their antidepressant for the length of the trial.
They received IV ketamine twice weekly for 4 weeks. The dosage was weight-dependent.
At the end of the 4 weeks, participants who achieved a MADRS total score of less than 10 or had a 30% or greater reduction from their baseline MADRS score entered another 4-week phase of the trial. This phase consisted of once-weekly administration of IV ketamine.
Larger plans
Results showed 15 of the 25 participants (60%) experienced a 30% or higher reduction in MADRS scores in the first phase of the study. The mean change in MADRS total score from the beginning to the end of the first phase was a decrease of 9.4 points (P < .01).
At the end of the continuation phase, half (48%) met criteria for response and 27% met criteria for remission.
After ketamine administration, the researchers also found an improvement in Fluid Cognition Composite Score (Cohen’s d value = .61), indicating a medium to large effect size, and in three measures of executive function.
Overall, adverse events were rare and did not keep patients from participating in the study, the investigators note. Five of the 25 participants reported infusion-induced hypertension that was transient.
Study limitations cited include the small sample size and the absence of randomization and placebo control or comparison treatment.
“We were very pleased with these findings because they establish the safety of this novel intervention in older adults,” Dr. Gebara said.
“After establishing safety and tolerability, we can plan for larger, randomized controlled trials that will allow us to determine the effectiveness of IV ketamine for older adults with TRD,” she added.
Multiple mechanisms
In a comment, Gerard Sanacora, MD, PhD, professor of psychiatry at Yale University and director of the Yale Depression Research Program, New Haven, Conn., noted multiple mechanisms likely contribute to the antidepressant effects of ketamine.
Dr. Sanacora has independently researched the effects of ketamine but was not involved with the current study.
“Much of the work to date has focused on the drug’s proximal effects on the glutamatergic neurotransmitter system and the resulting enhancement of adaptive neuroplasticity in several brain regions,” he said.
“However, there is also evidence to suggest other neurotransmitter systems and possibly even neuroinflammatory regulators are also contributing to the effect,” Dr. Sanacora added.
He noted that these mechanisms are also likely amplified by the “hope, optimism, expectations, and improved medical management overall that are known to be associated with treatments that require close monitoring and follow-up with health care providers.”
Dr. Gebara noted that “internal/department funds at each site” were used to support the study. She also reported receiving support from Otsuka US. Disclosures for the other investigators are listed in the original article. Dr. Sanacora has reported having “no major direct conflicts” with the study.
A version of this article first appeared on Medscape.com.
Results showed nearly 50% of participants responded to ketamine and 25% achieved complete remission from TRD, as measured by scores on the Montgomery-Asberg Depression Rating Scale (MADRS).
“Our pilot study suggests that IV ketamine is well-tolerated, safe, and associated with improvement in late-life TRD,” co-investigator Marie Anne Gebara, MD, assistant professor of psychiatry at the University of Pittsburgh, told this news organization.
Dr. Gebara pointed out the treatment “may not be appropriate for all patients with TRD,” such as those with a history of psychotic symptoms or uncontrolled hypertension; but “it appears to be a promising option.”
The findings were published online in the American Journal of Geriatric Psychiatry.
Lack of data in seniors
Although ketamine has been shown in prior research to rapidly reduce suicidal ideation in adults, there has been a lack of data on its efficacy and safety in older adults, the current investigators note.
“Almost 50% of older adults suffering from depression have TRD, which is a leading cause of disability, excess mortality from suicide, and dementia,” Dr. Gebara said.
She added that after two failed trials of antidepressants, “older adults have few evidence-based choices: aripiprazole or bupropion augmentation, transcranial magnetic stimulation, or electroconvulsive therapy. Novel treatments with rapid benefit are needed as long-term outcomes are poor and recurrence rates are high.”
Dr. Gebara and colleagues at five sites (Columbia University, New York State Psychiatric Institute, University of Toronto, University of Pittsburgh, and Washington University in St. Louis) each enrolled five participants aged 60 and older into the pilot study between October 2020 and November 2021, for a total of 25 participants (mean age, 71 years).
Each participant was recruited from patient registries or referred by behavioral health or primary care providers and diagnosed with TRD, which was defined as an episode of major depressive disorder without psychotic features that persisted despite two or more trials of antidepressants including at least one evidence-based second-line treatment.
Participants had to take an oral antidepressant dosage for at least 1 month prior to the start of the IV ketamine infusions, and continue their antidepressant for the length of the trial.
They received IV ketamine twice weekly for 4 weeks. The dosage was weight-dependent.
At the end of the 4 weeks, participants who achieved a MADRS total score of less than 10 or had a 30% or greater reduction from their baseline MADRS score entered another 4-week phase of the trial. This phase consisted of once-weekly administration of IV ketamine.
Larger plans
Results showed 15 of the 25 participants (60%) experienced a 30% or higher reduction in MADRS scores in the first phase of the study. The mean change in MADRS total score from the beginning to the end of the first phase was a decrease of 9.4 points (P < .01).
At the end of the continuation phase, half (48%) met criteria for response and 27% met criteria for remission.
After ketamine administration, the researchers also found an improvement in Fluid Cognition Composite Score (Cohen’s d value = .61), indicating a medium to large effect size, and in three measures of executive function.
Overall, adverse events were rare and did not keep patients from participating in the study, the investigators note. Five of the 25 participants reported infusion-induced hypertension that was transient.
Study limitations cited include the small sample size and the absence of randomization and placebo control or comparison treatment.
“We were very pleased with these findings because they establish the safety of this novel intervention in older adults,” Dr. Gebara said.
“After establishing safety and tolerability, we can plan for larger, randomized controlled trials that will allow us to determine the effectiveness of IV ketamine for older adults with TRD,” she added.
Multiple mechanisms
In a comment, Gerard Sanacora, MD, PhD, professor of psychiatry at Yale University and director of the Yale Depression Research Program, New Haven, Conn., noted multiple mechanisms likely contribute to the antidepressant effects of ketamine.
Dr. Sanacora has independently researched the effects of ketamine but was not involved with the current study.
“Much of the work to date has focused on the drug’s proximal effects on the glutamatergic neurotransmitter system and the resulting enhancement of adaptive neuroplasticity in several brain regions,” he said.
“However, there is also evidence to suggest other neurotransmitter systems and possibly even neuroinflammatory regulators are also contributing to the effect,” Dr. Sanacora added.
He noted that these mechanisms are also likely amplified by the “hope, optimism, expectations, and improved medical management overall that are known to be associated with treatments that require close monitoring and follow-up with health care providers.”
Dr. Gebara noted that “internal/department funds at each site” were used to support the study. She also reported receiving support from Otsuka US. Disclosures for the other investigators are listed in the original article. Dr. Sanacora has reported having “no major direct conflicts” with the study.
A version of this article first appeared on Medscape.com.
Results showed nearly 50% of participants responded to ketamine and 25% achieved complete remission from TRD, as measured by scores on the Montgomery-Asberg Depression Rating Scale (MADRS).
“Our pilot study suggests that IV ketamine is well-tolerated, safe, and associated with improvement in late-life TRD,” co-investigator Marie Anne Gebara, MD, assistant professor of psychiatry at the University of Pittsburgh, told this news organization.
Dr. Gebara pointed out the treatment “may not be appropriate for all patients with TRD,” such as those with a history of psychotic symptoms or uncontrolled hypertension; but “it appears to be a promising option.”
The findings were published online in the American Journal of Geriatric Psychiatry.
Lack of data in seniors
Although ketamine has been shown in prior research to rapidly reduce suicidal ideation in adults, there has been a lack of data on its efficacy and safety in older adults, the current investigators note.
“Almost 50% of older adults suffering from depression have TRD, which is a leading cause of disability, excess mortality from suicide, and dementia,” Dr. Gebara said.
She added that after two failed trials of antidepressants, “older adults have few evidence-based choices: aripiprazole or bupropion augmentation, transcranial magnetic stimulation, or electroconvulsive therapy. Novel treatments with rapid benefit are needed as long-term outcomes are poor and recurrence rates are high.”
Dr. Gebara and colleagues at five sites (Columbia University, New York State Psychiatric Institute, University of Toronto, University of Pittsburgh, and Washington University in St. Louis) each enrolled five participants aged 60 and older into the pilot study between October 2020 and November 2021, for a total of 25 participants (mean age, 71 years).
Each participant was recruited from patient registries or referred by behavioral health or primary care providers and diagnosed with TRD, which was defined as an episode of major depressive disorder without psychotic features that persisted despite two or more trials of antidepressants including at least one evidence-based second-line treatment.
Participants had to take an oral antidepressant dosage for at least 1 month prior to the start of the IV ketamine infusions, and continue their antidepressant for the length of the trial.
They received IV ketamine twice weekly for 4 weeks. The dosage was weight-dependent.
At the end of the 4 weeks, participants who achieved a MADRS total score of less than 10 or had a 30% or greater reduction from their baseline MADRS score entered another 4-week phase of the trial. This phase consisted of once-weekly administration of IV ketamine.
Larger plans
Results showed 15 of the 25 participants (60%) experienced a 30% or higher reduction in MADRS scores in the first phase of the study. The mean change in MADRS total score from the beginning to the end of the first phase was a decrease of 9.4 points (P < .01).
At the end of the continuation phase, half (48%) met criteria for response and 27% met criteria for remission.
After ketamine administration, the researchers also found an improvement in Fluid Cognition Composite Score (Cohen’s d value = .61), indicating a medium to large effect size, and in three measures of executive function.
Overall, adverse events were rare and did not keep patients from participating in the study, the investigators note. Five of the 25 participants reported infusion-induced hypertension that was transient.
Study limitations cited include the small sample size and the absence of randomization and placebo control or comparison treatment.
“We were very pleased with these findings because they establish the safety of this novel intervention in older adults,” Dr. Gebara said.
“After establishing safety and tolerability, we can plan for larger, randomized controlled trials that will allow us to determine the effectiveness of IV ketamine for older adults with TRD,” she added.
Multiple mechanisms
In a comment, Gerard Sanacora, MD, PhD, professor of psychiatry at Yale University and director of the Yale Depression Research Program, New Haven, Conn., noted multiple mechanisms likely contribute to the antidepressant effects of ketamine.
Dr. Sanacora has independently researched the effects of ketamine but was not involved with the current study.
“Much of the work to date has focused on the drug’s proximal effects on the glutamatergic neurotransmitter system and the resulting enhancement of adaptive neuroplasticity in several brain regions,” he said.
“However, there is also evidence to suggest other neurotransmitter systems and possibly even neuroinflammatory regulators are also contributing to the effect,” Dr. Sanacora added.
He noted that these mechanisms are also likely amplified by the “hope, optimism, expectations, and improved medical management overall that are known to be associated with treatments that require close monitoring and follow-up with health care providers.”
Dr. Gebara noted that “internal/department funds at each site” were used to support the study. She also reported receiving support from Otsuka US. Disclosures for the other investigators are listed in the original article. Dr. Sanacora has reported having “no major direct conflicts” with the study.
A version of this article first appeared on Medscape.com.
FROM THE AMERICAN JOURNAL OF GERIATRIC PSYCHIATRY
















