User login
More than a month after launch, iPLEDGE glitches persist
which mandates the program to prevent fetal exposure to the teratogenic effects of isotretinoin, and by the American Academy of Dermatology Association, whose members have repeatedly asked the FDA for meetings to discuss solutions. The AADA is the legislative and advocacy arm of AAD.
When the new program launched Dec. 13, 2021, the website crashed repeatedly, with physicians and patients complaining they got locked out or bounced off the platform when they tried to follow instructions to enter information. Hold times to talk to a live person stretched to hours.
The latest improvement attempt, announced Jan. 14 by the FDA, is a tool created by the Isotretinoin Products Manufacturers Group, the manufacturers responsible for the FDA-mandated REMS program. It is meant to allow prescribers and designees to send log-in links directly to patients’ email accounts through the iPLEDGE REMS portal, bypassing the troublesome call center.
And it’s not the answer, dermatologists said.
“The new tool does not solve issues such as prescribers or pharmacies not being able to access the site, unacceptably long call center wait times, inefficiencies caused by frequent attestation requirements for those who cannot become pregnant, patients becoming ‘locked out’ because they missed a window period through no fault of their own, among others,” said John Barbieri, MD, MBA, director of the Advanced Acne Therapeutics Clinic at Brigham and Women’s Hospital and instructor in dermatology at Harvard Medical School, both in Boston.
The day after the FDA update about the new tool, Klint Peebles, MD, a dermatologist at Kaiser Permanente in Washington, D.C., tweeted: “Lip service and empty words.” He noted that the situation has been “disastrous from the start” as the new platform launched.
Under the iPLEDGE program in place for the acne drug, physicians, patients, and pharmacies prescribing, using, or dispensing the drug must all be registered, with requirements that include the use of two forms of an effective contraceptive and regular pregnancy testing for patients who can become pregnant.
The aim of the new gender-neutral approach to the risk mitigation program is to make the experience more inclusive for transgender patients. The previous three risk categories (females of reproductive potential, females not of reproductive potential, and males) are now reduced to just two (those capable of getting pregnant and those not capable of getting pregnant).
The problem is the execution of the new platform. The transition from the old website to the new was done quickly. By most accounts, the Dec. 13 rollout was chaotic, a failure, and disastrous, triggering numerous expressions of frustration on Twitter and other social media, with some calling for the program to be halted until the bugs could be worked out.
“While the new gender-neutral categories are a welcome improvement to the system, the new categorization approach was not the underlying reason for the new platform and its failed rollout, which was instead due to a change in vendor,” Dr. Barbieri told this news organization.
AADA: More recent efforts to improve the system
“We have a letter to the FDA asking for a stakeholders meeting to include us, the IPMG, and pharmacists because there are ongoing problems, though there have been some improvement in terms of certain elements,” Ilona Frieden, MD, chair of the AADA’s iPLEDGE Workgroup and professor of dermatology at the University of California, San Francisco, said in an interview shortly after the FDA posted the update on the new tool. “That said, there are many patients who have not gotten isotretinoin during the 1 month since the roll-out of the new platform.”
What still needs to be fixed? “We have ongoing concerns about the lack of transparency of the IPMG, about call center wait times, actual number of prescriptions on the hands of patients compared to the previous month, and those patients who can get pregnant who – despite complying with all of the REMS requirements – are being locked out because of the lack of timely attestation to their negative pregnancy status due to the website, not the patients themselves,” Dr. Frieden told this news organization.
“We are continuing to advocate to have decreased attestation requirements for individuals who cannot become pregnant – because this will improve the efficiency of the system for those patients for whom the REMS program goals are truly intended – those who can become pregnant, since the primary aim of the REMS program is to minimize fetal exposure.”
An AADA spokesperson said that the IPMG has invited the AADA to a joint stakeholders meeting on Jan. 26, along with representatives from the FDA and pharmacy industry.
Spotty progress
“The iPLEDGE situation is as frustrating as ever,” said Neil S. Goldberg, MD, a dermatologist in Westchester County, New York, after the FDA’s Jan. 14 update was released. “It’s like they never tested the new website before deploying it.”
Among the issues he has experienced in his practice, he said, is an instance in which iPLEDGE swapped the first names of a mother and daughter, so it was impossible to fill the prescription. “It happened twice in the same day,” Dr. Goldberg said. The patient had to call iPLEDGE to fix this, but the call center wasn’t taking calls.
In today’s technology environment, he said, it’s hard to believe that “we have to put up with this.”
Some have seen success. ‘’The tool is working fine on our end,” said Mitesh Patel, PharmD, pharmacy manager at Sunshine Pharmacy in White Plains, N.Y. However, he added that some doctors and patients are still having issues. He encourages dermatologists still having issues with the system to reach out to independent pharmacies that have processed iPLEDGE prescriptions and ‘’lean on them to assist.”
This news organization contacted CVS and Walgreens about how the system is working at their locations, but has not yet received a response.
Dr. Goldberg, Dr. Frieden, Dr. Barbieri, and Dr. Peebles have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
This story was updated on 1/24/22.
which mandates the program to prevent fetal exposure to the teratogenic effects of isotretinoin, and by the American Academy of Dermatology Association, whose members have repeatedly asked the FDA for meetings to discuss solutions. The AADA is the legislative and advocacy arm of AAD.
When the new program launched Dec. 13, 2021, the website crashed repeatedly, with physicians and patients complaining they got locked out or bounced off the platform when they tried to follow instructions to enter information. Hold times to talk to a live person stretched to hours.
The latest improvement attempt, announced Jan. 14 by the FDA, is a tool created by the Isotretinoin Products Manufacturers Group, the manufacturers responsible for the FDA-mandated REMS program. It is meant to allow prescribers and designees to send log-in links directly to patients’ email accounts through the iPLEDGE REMS portal, bypassing the troublesome call center.
And it’s not the answer, dermatologists said.
“The new tool does not solve issues such as prescribers or pharmacies not being able to access the site, unacceptably long call center wait times, inefficiencies caused by frequent attestation requirements for those who cannot become pregnant, patients becoming ‘locked out’ because they missed a window period through no fault of their own, among others,” said John Barbieri, MD, MBA, director of the Advanced Acne Therapeutics Clinic at Brigham and Women’s Hospital and instructor in dermatology at Harvard Medical School, both in Boston.
The day after the FDA update about the new tool, Klint Peebles, MD, a dermatologist at Kaiser Permanente in Washington, D.C., tweeted: “Lip service and empty words.” He noted that the situation has been “disastrous from the start” as the new platform launched.
Under the iPLEDGE program in place for the acne drug, physicians, patients, and pharmacies prescribing, using, or dispensing the drug must all be registered, with requirements that include the use of two forms of an effective contraceptive and regular pregnancy testing for patients who can become pregnant.
The aim of the new gender-neutral approach to the risk mitigation program is to make the experience more inclusive for transgender patients. The previous three risk categories (females of reproductive potential, females not of reproductive potential, and males) are now reduced to just two (those capable of getting pregnant and those not capable of getting pregnant).
The problem is the execution of the new platform. The transition from the old website to the new was done quickly. By most accounts, the Dec. 13 rollout was chaotic, a failure, and disastrous, triggering numerous expressions of frustration on Twitter and other social media, with some calling for the program to be halted until the bugs could be worked out.
“While the new gender-neutral categories are a welcome improvement to the system, the new categorization approach was not the underlying reason for the new platform and its failed rollout, which was instead due to a change in vendor,” Dr. Barbieri told this news organization.
AADA: More recent efforts to improve the system
“We have a letter to the FDA asking for a stakeholders meeting to include us, the IPMG, and pharmacists because there are ongoing problems, though there have been some improvement in terms of certain elements,” Ilona Frieden, MD, chair of the AADA’s iPLEDGE Workgroup and professor of dermatology at the University of California, San Francisco, said in an interview shortly after the FDA posted the update on the new tool. “That said, there are many patients who have not gotten isotretinoin during the 1 month since the roll-out of the new platform.”
What still needs to be fixed? “We have ongoing concerns about the lack of transparency of the IPMG, about call center wait times, actual number of prescriptions on the hands of patients compared to the previous month, and those patients who can get pregnant who – despite complying with all of the REMS requirements – are being locked out because of the lack of timely attestation to their negative pregnancy status due to the website, not the patients themselves,” Dr. Frieden told this news organization.
“We are continuing to advocate to have decreased attestation requirements for individuals who cannot become pregnant – because this will improve the efficiency of the system for those patients for whom the REMS program goals are truly intended – those who can become pregnant, since the primary aim of the REMS program is to minimize fetal exposure.”
An AADA spokesperson said that the IPMG has invited the AADA to a joint stakeholders meeting on Jan. 26, along with representatives from the FDA and pharmacy industry.
Spotty progress
“The iPLEDGE situation is as frustrating as ever,” said Neil S. Goldberg, MD, a dermatologist in Westchester County, New York, after the FDA’s Jan. 14 update was released. “It’s like they never tested the new website before deploying it.”
Among the issues he has experienced in his practice, he said, is an instance in which iPLEDGE swapped the first names of a mother and daughter, so it was impossible to fill the prescription. “It happened twice in the same day,” Dr. Goldberg said. The patient had to call iPLEDGE to fix this, but the call center wasn’t taking calls.
In today’s technology environment, he said, it’s hard to believe that “we have to put up with this.”
Some have seen success. ‘’The tool is working fine on our end,” said Mitesh Patel, PharmD, pharmacy manager at Sunshine Pharmacy in White Plains, N.Y. However, he added that some doctors and patients are still having issues. He encourages dermatologists still having issues with the system to reach out to independent pharmacies that have processed iPLEDGE prescriptions and ‘’lean on them to assist.”
This news organization contacted CVS and Walgreens about how the system is working at their locations, but has not yet received a response.
Dr. Goldberg, Dr. Frieden, Dr. Barbieri, and Dr. Peebles have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
This story was updated on 1/24/22.
which mandates the program to prevent fetal exposure to the teratogenic effects of isotretinoin, and by the American Academy of Dermatology Association, whose members have repeatedly asked the FDA for meetings to discuss solutions. The AADA is the legislative and advocacy arm of AAD.
When the new program launched Dec. 13, 2021, the website crashed repeatedly, with physicians and patients complaining they got locked out or bounced off the platform when they tried to follow instructions to enter information. Hold times to talk to a live person stretched to hours.
The latest improvement attempt, announced Jan. 14 by the FDA, is a tool created by the Isotretinoin Products Manufacturers Group, the manufacturers responsible for the FDA-mandated REMS program. It is meant to allow prescribers and designees to send log-in links directly to patients’ email accounts through the iPLEDGE REMS portal, bypassing the troublesome call center.
And it’s not the answer, dermatologists said.
“The new tool does not solve issues such as prescribers or pharmacies not being able to access the site, unacceptably long call center wait times, inefficiencies caused by frequent attestation requirements for those who cannot become pregnant, patients becoming ‘locked out’ because they missed a window period through no fault of their own, among others,” said John Barbieri, MD, MBA, director of the Advanced Acne Therapeutics Clinic at Brigham and Women’s Hospital and instructor in dermatology at Harvard Medical School, both in Boston.
The day after the FDA update about the new tool, Klint Peebles, MD, a dermatologist at Kaiser Permanente in Washington, D.C., tweeted: “Lip service and empty words.” He noted that the situation has been “disastrous from the start” as the new platform launched.
Under the iPLEDGE program in place for the acne drug, physicians, patients, and pharmacies prescribing, using, or dispensing the drug must all be registered, with requirements that include the use of two forms of an effective contraceptive and regular pregnancy testing for patients who can become pregnant.
The aim of the new gender-neutral approach to the risk mitigation program is to make the experience more inclusive for transgender patients. The previous three risk categories (females of reproductive potential, females not of reproductive potential, and males) are now reduced to just two (those capable of getting pregnant and those not capable of getting pregnant).
The problem is the execution of the new platform. The transition from the old website to the new was done quickly. By most accounts, the Dec. 13 rollout was chaotic, a failure, and disastrous, triggering numerous expressions of frustration on Twitter and other social media, with some calling for the program to be halted until the bugs could be worked out.
“While the new gender-neutral categories are a welcome improvement to the system, the new categorization approach was not the underlying reason for the new platform and its failed rollout, which was instead due to a change in vendor,” Dr. Barbieri told this news organization.
AADA: More recent efforts to improve the system
“We have a letter to the FDA asking for a stakeholders meeting to include us, the IPMG, and pharmacists because there are ongoing problems, though there have been some improvement in terms of certain elements,” Ilona Frieden, MD, chair of the AADA’s iPLEDGE Workgroup and professor of dermatology at the University of California, San Francisco, said in an interview shortly after the FDA posted the update on the new tool. “That said, there are many patients who have not gotten isotretinoin during the 1 month since the roll-out of the new platform.”
What still needs to be fixed? “We have ongoing concerns about the lack of transparency of the IPMG, about call center wait times, actual number of prescriptions on the hands of patients compared to the previous month, and those patients who can get pregnant who – despite complying with all of the REMS requirements – are being locked out because of the lack of timely attestation to their negative pregnancy status due to the website, not the patients themselves,” Dr. Frieden told this news organization.
“We are continuing to advocate to have decreased attestation requirements for individuals who cannot become pregnant – because this will improve the efficiency of the system for those patients for whom the REMS program goals are truly intended – those who can become pregnant, since the primary aim of the REMS program is to minimize fetal exposure.”
An AADA spokesperson said that the IPMG has invited the AADA to a joint stakeholders meeting on Jan. 26, along with representatives from the FDA and pharmacy industry.
Spotty progress
“The iPLEDGE situation is as frustrating as ever,” said Neil S. Goldberg, MD, a dermatologist in Westchester County, New York, after the FDA’s Jan. 14 update was released. “It’s like they never tested the new website before deploying it.”
Among the issues he has experienced in his practice, he said, is an instance in which iPLEDGE swapped the first names of a mother and daughter, so it was impossible to fill the prescription. “It happened twice in the same day,” Dr. Goldberg said. The patient had to call iPLEDGE to fix this, but the call center wasn’t taking calls.
In today’s technology environment, he said, it’s hard to believe that “we have to put up with this.”
Some have seen success. ‘’The tool is working fine on our end,” said Mitesh Patel, PharmD, pharmacy manager at Sunshine Pharmacy in White Plains, N.Y. However, he added that some doctors and patients are still having issues. He encourages dermatologists still having issues with the system to reach out to independent pharmacies that have processed iPLEDGE prescriptions and ‘’lean on them to assist.”
This news organization contacted CVS and Walgreens about how the system is working at their locations, but has not yet received a response.
Dr. Goldberg, Dr. Frieden, Dr. Barbieri, and Dr. Peebles have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
This story was updated on 1/24/22.
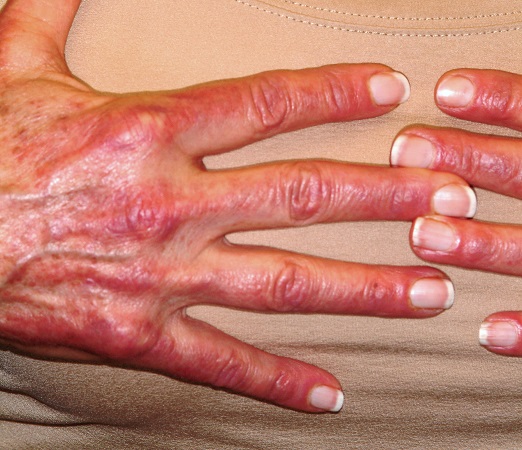
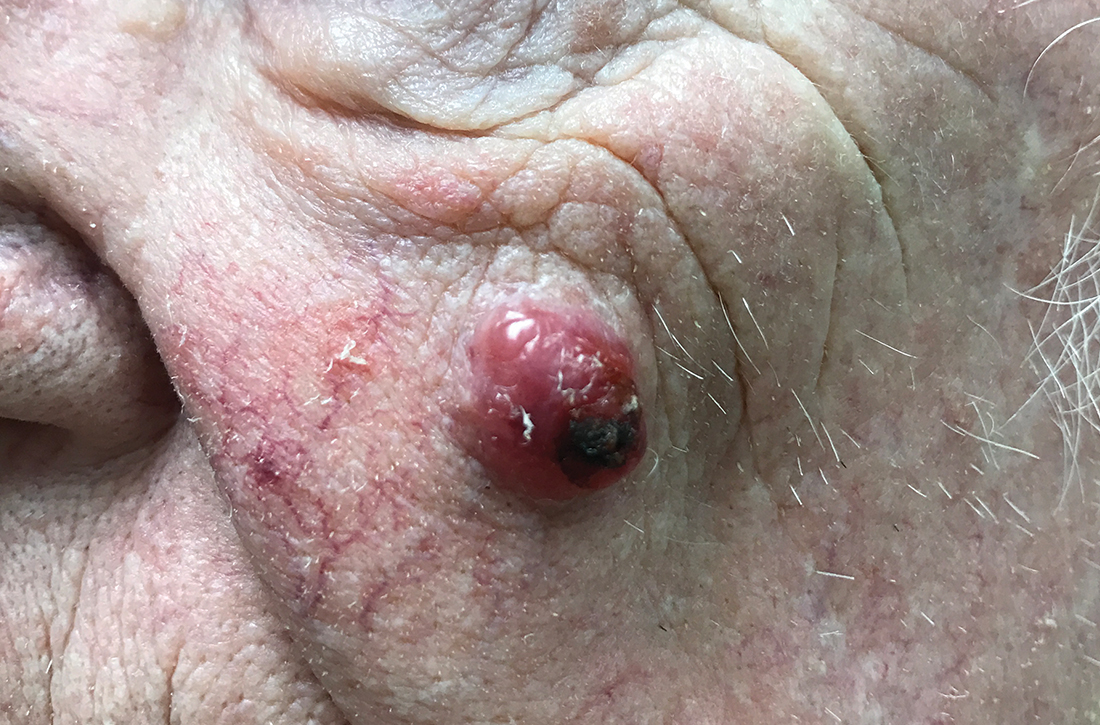
Nodule on the left cheek
An 85-year-old man with a history of skin cancer presented to my dermatology practice (NT) for evaluation of a “pimple” on his left cheek that failed to resolve after 2 months (FIGURE). The patient noted that the lesion had grown, but that he otherwise felt well.
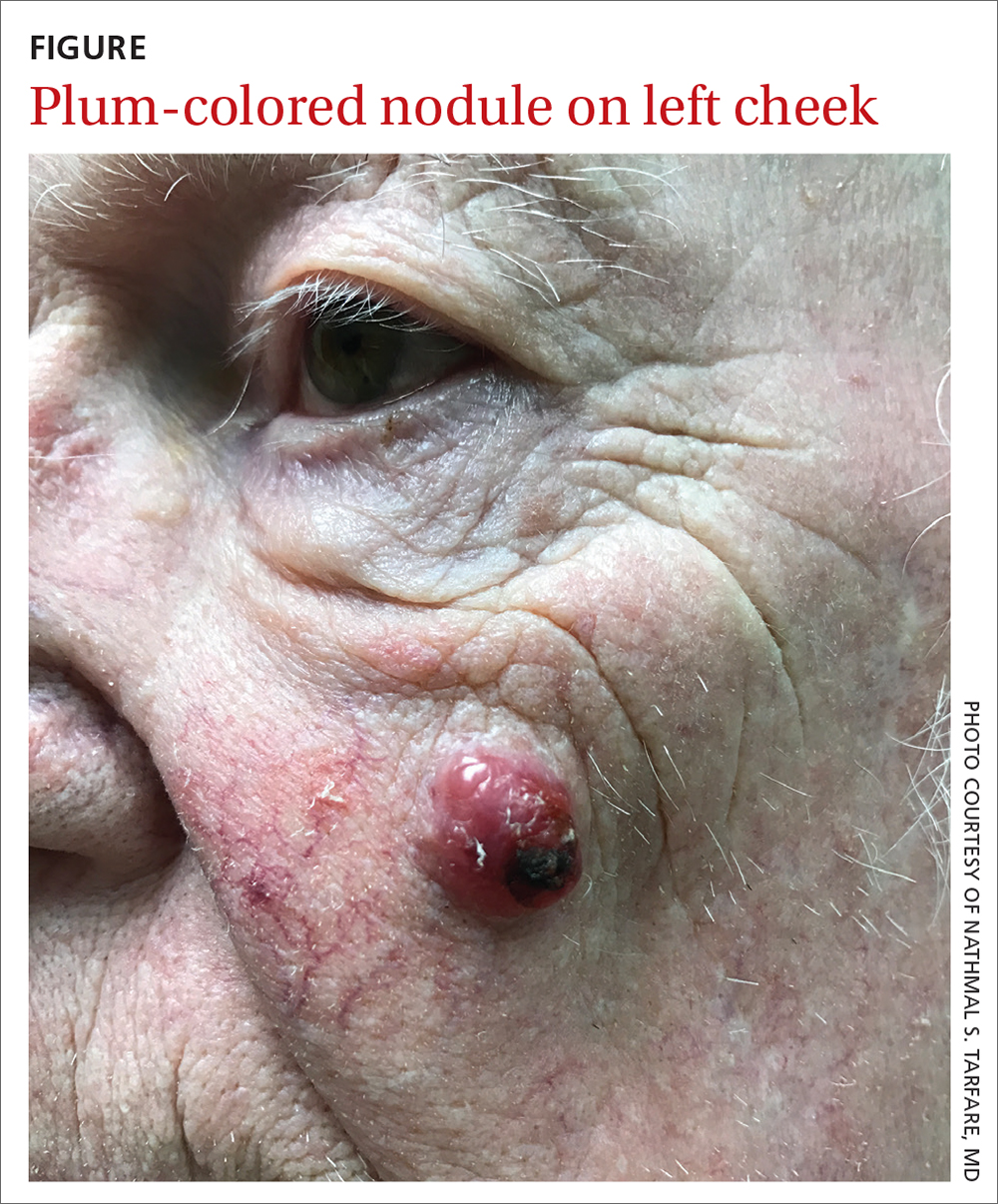
On examination, the lesion was plum colored, and the area was firm and nontender to palpation. The patient was referred to a plastic surgeon for an excisional biopsy to clarify the nature of the lesion.
WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Diagnosis: Merkel cell carcinoma
A biopsy performed 2 weeks after the initial visit confirmed the clinical suspicion for Merkel cell carcinoma (MCC).
MCC is a cutaneous neuroendocrine malignancy. Although its name acknowledges similarities between the tumor cells and Merkel cells, it is now considered unlikely that Merkel cells are the actual cells of origin.1
The majority of MCCs are asymptomatic despite rapid growth and are typically red or pink and occur on UV-exposed areas, as in our patient.2 A cyst or acneiform lesion is the single most common diagnosis given at the time of biopsy.2
The incidence of MCC is greatest in people of advanced age and in those who are immunosuppressed. In the United States, the estimated annual incidence rate rose from 0.5 cases per 100,000 people in 2000 to 0.7 cases per 100,000 people in 2013.3 MCC increases exponentially with advancing age, from 0.1 (per 100,000) in those ages 40 to 44 years to 9.8 in those older than 85 years.3 The growing cohort of ageing baby boomers and the increased number of immunosuppressed individuals in the community suggest that clinicians are now more likely to encounter MCC than in the past.
While UV radiation is highly associated with MCC, the major causative factor is considered to be Merkel cell polyomavirus (MCPyV).1 In fact, MCPyV has been linked to 80% of MCC cases.1,3 Most people have positive serology for MCPyV in early childhood, but the association between MCC and old age highlights the impact of immunosuppression on MCPyV activity and MCC development.1
Clinical suspicion is the first step in diagnosing MCC
The mnemonic AEIOU highlights the key clinical features of this aggressive tumor2,4:
- Asymptomatic
- Expanding rapidly (often grows in less than 3 months)
- Immune suppression (eg, chronic lymphocytic leukemia, solid organ transplant patient)
- Older than 50
- UV exposure on fair skin.
If a lesion is suspected to be MCC, the next step includes biopsy so that a definitive diagnosis can be made. A firm, nontender nodule that lacks fluctuance should raise suspicion for a neoplastic process.
Continue to: The differential is broad, ranging from cysts to melanoma
The differential is broad, ranging from cysts to melanoma
The differential diagnosis for an enlarging, plum-colored nodule on sun-exposed skin includes an abscess, a ruptured or inflamed epidermoid cyst, basal cell carcinoma, squamous cell carcinoma, and malignant melanoma.
An abscess is typically tender and expands within a matter of days rather than months.
A cyst can be ruled out by the clinical appearance and lack of an overlying pore.
Basal cell carcinoma can be characterized by a rolled border and central ulceration.
Squamous cell carcinomas often exhibit a verrucous surface with marked hyperkeratosis.
Continue to: Melanoma
Melanoma manifests with brown or irregular pigmentation and may be associated with a precursor lesion.
Tx includes excision and consistent follow-up
Complete excision is the critical first step to successful therapy. Sentinel lymph node studies are typically performed because of the high incidence of lymph node metastasis. Frequent follow-up is required because of the high risk of recurrent or persistent disease.
Local recurrence usually occurs within 1 year of diagnosis in more than 40% of patients.5 Distant metastasis can be treated with a programmed cell death ligand 1 blocking agent (avelumab) or a programmed cell death protein 1 inhibitor (nivolumab or pembrolizumab).6
Our patient was referred to a regional cancer center for sentinel lymph node evaluation, where he was found to have nodal disease. The patient was put on pembrolizumab and received radiation therapy but showed only limited response. Seven months after diagnosis, he passed away from metastatic MCC.
1. Pietropaolo V, Prezioso C, Moens U. Merkel cell polyomavirus and Merkel cell carcinoma. Cancers (Basel). 2020;12:1774. doi: 10.3390/cancers12071774
2. Heath M, Jaimes N, Lemos B, et al. Clinical characteristics of Merkel cell carcinoma at diagnosis in 195 patients: the AEIOU features. J Am Acad Dermatol. 2008;58:375-381. doi: 10.1016/j.jaad.2007.11.020
3. Paulson KG, Park SY, Vandeven NA, et al. Merkel cell carcinoma: current US incidence and projected increases based on changing demographics. J Am Acad Dermatol. 2018;78:457-463.e2. doi: 10.1016/j.jaad.2017.10.028
4. Voelker R. Why Merkel cell cancer is garnering more attention. JAMA. 2018;320:18-20. doi: 10.1001/jama.2018.7042
5. Allen PJ, Browne WB, Jacques DP, et al. Merkel cell carcinoma: prognosis and treatment of patients from a single institution. J Clin Oncol. 2005;23:2300-2309. doi: 10.1200/JCO.2005.02.329
6. D’Angelo SP, Russell J, Lebbé C, et al. Efficacy and safety of first-line avelumab treatment in patients with stage IV metastatic Merkel cell carcinoma: a preplanned interim analysis of a clinical trial. JAMA Oncol. 2018;4:e180077. doi: 10.1001/jamaoncol.2018.0077
An 85-year-old man with a history of skin cancer presented to my dermatology practice (NT) for evaluation of a “pimple” on his left cheek that failed to resolve after 2 months (FIGURE). The patient noted that the lesion had grown, but that he otherwise felt well.

On examination, the lesion was plum colored, and the area was firm and nontender to palpation. The patient was referred to a plastic surgeon for an excisional biopsy to clarify the nature of the lesion.
WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Diagnosis: Merkel cell carcinoma
A biopsy performed 2 weeks after the initial visit confirmed the clinical suspicion for Merkel cell carcinoma (MCC).
MCC is a cutaneous neuroendocrine malignancy. Although its name acknowledges similarities between the tumor cells and Merkel cells, it is now considered unlikely that Merkel cells are the actual cells of origin.1
The majority of MCCs are asymptomatic despite rapid growth and are typically red or pink and occur on UV-exposed areas, as in our patient.2 A cyst or acneiform lesion is the single most common diagnosis given at the time of biopsy.2
The incidence of MCC is greatest in people of advanced age and in those who are immunosuppressed. In the United States, the estimated annual incidence rate rose from 0.5 cases per 100,000 people in 2000 to 0.7 cases per 100,000 people in 2013.3 MCC increases exponentially with advancing age, from 0.1 (per 100,000) in those ages 40 to 44 years to 9.8 in those older than 85 years.3 The growing cohort of ageing baby boomers and the increased number of immunosuppressed individuals in the community suggest that clinicians are now more likely to encounter MCC than in the past.
While UV radiation is highly associated with MCC, the major causative factor is considered to be Merkel cell polyomavirus (MCPyV).1 In fact, MCPyV has been linked to 80% of MCC cases.1,3 Most people have positive serology for MCPyV in early childhood, but the association between MCC and old age highlights the impact of immunosuppression on MCPyV activity and MCC development.1
Clinical suspicion is the first step in diagnosing MCC
The mnemonic AEIOU highlights the key clinical features of this aggressive tumor2,4:
- Asymptomatic
- Expanding rapidly (often grows in less than 3 months)
- Immune suppression (eg, chronic lymphocytic leukemia, solid organ transplant patient)
- Older than 50
- UV exposure on fair skin.
If a lesion is suspected to be MCC, the next step includes biopsy so that a definitive diagnosis can be made. A firm, nontender nodule that lacks fluctuance should raise suspicion for a neoplastic process.
Continue to: The differential is broad, ranging from cysts to melanoma
The differential is broad, ranging from cysts to melanoma
The differential diagnosis for an enlarging, plum-colored nodule on sun-exposed skin includes an abscess, a ruptured or inflamed epidermoid cyst, basal cell carcinoma, squamous cell carcinoma, and malignant melanoma.
An abscess is typically tender and expands within a matter of days rather than months.
A cyst can be ruled out by the clinical appearance and lack of an overlying pore.
Basal cell carcinoma can be characterized by a rolled border and central ulceration.
Squamous cell carcinomas often exhibit a verrucous surface with marked hyperkeratosis.
Continue to: Melanoma
Melanoma manifests with brown or irregular pigmentation and may be associated with a precursor lesion.
Tx includes excision and consistent follow-up
Complete excision is the critical first step to successful therapy. Sentinel lymph node studies are typically performed because of the high incidence of lymph node metastasis. Frequent follow-up is required because of the high risk of recurrent or persistent disease.
Local recurrence usually occurs within 1 year of diagnosis in more than 40% of patients.5 Distant metastasis can be treated with a programmed cell death ligand 1 blocking agent (avelumab) or a programmed cell death protein 1 inhibitor (nivolumab or pembrolizumab).6
Our patient was referred to a regional cancer center for sentinel lymph node evaluation, where he was found to have nodal disease. The patient was put on pembrolizumab and received radiation therapy but showed only limited response. Seven months after diagnosis, he passed away from metastatic MCC.
An 85-year-old man with a history of skin cancer presented to my dermatology practice (NT) for evaluation of a “pimple” on his left cheek that failed to resolve after 2 months (FIGURE). The patient noted that the lesion had grown, but that he otherwise felt well.

On examination, the lesion was plum colored, and the area was firm and nontender to palpation. The patient was referred to a plastic surgeon for an excisional biopsy to clarify the nature of the lesion.
WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Diagnosis: Merkel cell carcinoma
A biopsy performed 2 weeks after the initial visit confirmed the clinical suspicion for Merkel cell carcinoma (MCC).
MCC is a cutaneous neuroendocrine malignancy. Although its name acknowledges similarities between the tumor cells and Merkel cells, it is now considered unlikely that Merkel cells are the actual cells of origin.1
The majority of MCCs are asymptomatic despite rapid growth and are typically red or pink and occur on UV-exposed areas, as in our patient.2 A cyst or acneiform lesion is the single most common diagnosis given at the time of biopsy.2
The incidence of MCC is greatest in people of advanced age and in those who are immunosuppressed. In the United States, the estimated annual incidence rate rose from 0.5 cases per 100,000 people in 2000 to 0.7 cases per 100,000 people in 2013.3 MCC increases exponentially with advancing age, from 0.1 (per 100,000) in those ages 40 to 44 years to 9.8 in those older than 85 years.3 The growing cohort of ageing baby boomers and the increased number of immunosuppressed individuals in the community suggest that clinicians are now more likely to encounter MCC than in the past.
While UV radiation is highly associated with MCC, the major causative factor is considered to be Merkel cell polyomavirus (MCPyV).1 In fact, MCPyV has been linked to 80% of MCC cases.1,3 Most people have positive serology for MCPyV in early childhood, but the association between MCC and old age highlights the impact of immunosuppression on MCPyV activity and MCC development.1
Clinical suspicion is the first step in diagnosing MCC
The mnemonic AEIOU highlights the key clinical features of this aggressive tumor2,4:
- Asymptomatic
- Expanding rapidly (often grows in less than 3 months)
- Immune suppression (eg, chronic lymphocytic leukemia, solid organ transplant patient)
- Older than 50
- UV exposure on fair skin.
If a lesion is suspected to be MCC, the next step includes biopsy so that a definitive diagnosis can be made. A firm, nontender nodule that lacks fluctuance should raise suspicion for a neoplastic process.
Continue to: The differential is broad, ranging from cysts to melanoma
The differential is broad, ranging from cysts to melanoma
The differential diagnosis for an enlarging, plum-colored nodule on sun-exposed skin includes an abscess, a ruptured or inflamed epidermoid cyst, basal cell carcinoma, squamous cell carcinoma, and malignant melanoma.
An abscess is typically tender and expands within a matter of days rather than months.
A cyst can be ruled out by the clinical appearance and lack of an overlying pore.
Basal cell carcinoma can be characterized by a rolled border and central ulceration.
Squamous cell carcinomas often exhibit a verrucous surface with marked hyperkeratosis.
Continue to: Melanoma
Melanoma manifests with brown or irregular pigmentation and may be associated with a precursor lesion.
Tx includes excision and consistent follow-up
Complete excision is the critical first step to successful therapy. Sentinel lymph node studies are typically performed because of the high incidence of lymph node metastasis. Frequent follow-up is required because of the high risk of recurrent or persistent disease.
Local recurrence usually occurs within 1 year of diagnosis in more than 40% of patients.5 Distant metastasis can be treated with a programmed cell death ligand 1 blocking agent (avelumab) or a programmed cell death protein 1 inhibitor (nivolumab or pembrolizumab).6
Our patient was referred to a regional cancer center for sentinel lymph node evaluation, where he was found to have nodal disease. The patient was put on pembrolizumab and received radiation therapy but showed only limited response. Seven months after diagnosis, he passed away from metastatic MCC.
1. Pietropaolo V, Prezioso C, Moens U. Merkel cell polyomavirus and Merkel cell carcinoma. Cancers (Basel). 2020;12:1774. doi: 10.3390/cancers12071774
2. Heath M, Jaimes N, Lemos B, et al. Clinical characteristics of Merkel cell carcinoma at diagnosis in 195 patients: the AEIOU features. J Am Acad Dermatol. 2008;58:375-381. doi: 10.1016/j.jaad.2007.11.020
3. Paulson KG, Park SY, Vandeven NA, et al. Merkel cell carcinoma: current US incidence and projected increases based on changing demographics. J Am Acad Dermatol. 2018;78:457-463.e2. doi: 10.1016/j.jaad.2017.10.028
4. Voelker R. Why Merkel cell cancer is garnering more attention. JAMA. 2018;320:18-20. doi: 10.1001/jama.2018.7042
5. Allen PJ, Browne WB, Jacques DP, et al. Merkel cell carcinoma: prognosis and treatment of patients from a single institution. J Clin Oncol. 2005;23:2300-2309. doi: 10.1200/JCO.2005.02.329
6. D’Angelo SP, Russell J, Lebbé C, et al. Efficacy and safety of first-line avelumab treatment in patients with stage IV metastatic Merkel cell carcinoma: a preplanned interim analysis of a clinical trial. JAMA Oncol. 2018;4:e180077. doi: 10.1001/jamaoncol.2018.0077
1. Pietropaolo V, Prezioso C, Moens U. Merkel cell polyomavirus and Merkel cell carcinoma. Cancers (Basel). 2020;12:1774. doi: 10.3390/cancers12071774
2. Heath M, Jaimes N, Lemos B, et al. Clinical characteristics of Merkel cell carcinoma at diagnosis in 195 patients: the AEIOU features. J Am Acad Dermatol. 2008;58:375-381. doi: 10.1016/j.jaad.2007.11.020
3. Paulson KG, Park SY, Vandeven NA, et al. Merkel cell carcinoma: current US incidence and projected increases based on changing demographics. J Am Acad Dermatol. 2018;78:457-463.e2. doi: 10.1016/j.jaad.2017.10.028
4. Voelker R. Why Merkel cell cancer is garnering more attention. JAMA. 2018;320:18-20. doi: 10.1001/jama.2018.7042
5. Allen PJ, Browne WB, Jacques DP, et al. Merkel cell carcinoma: prognosis and treatment of patients from a single institution. J Clin Oncol. 2005;23:2300-2309. doi: 10.1200/JCO.2005.02.329
6. D’Angelo SP, Russell J, Lebbé C, et al. Efficacy and safety of first-line avelumab treatment in patients with stage IV metastatic Merkel cell carcinoma: a preplanned interim analysis of a clinical trial. JAMA Oncol. 2018;4:e180077. doi: 10.1001/jamaoncol.2018.0077
Pioneering test predicts return of malignant melanoma
Their research, published in the British Journal of Dermatology, describes how early-stage melanomas at risk of spreading secrete transforming growth factor beta2 (TGF-beta2), which causes the reduction, or down-regulation, of the proteins AMBRA1 and loricrin, both of which are found in the skin overlaying the tumor. TGF-beta2 also causes the loss of claudin-1, which in turn leads to loss of skin integrity, facilitating ulceration.
Senior author Penny Lovat, PhD, professor of cellular dermatology and oncology at Newcastle University, and chief scientific officer at AMLo Biosciences, explained: “AMBRA1, loricrin, and claudin-1 are all proteins key to maintaining the integrity of the upper layer of the skin,” and that the loss of these proteins causes gaps to develop, allowing the tumor to spread and ulcerate – a process associated with high-risk tumors. Dr. Lovat likened the process to that of “mortar and bricks holding together a wall”, with the loss of these proteins being “like the mortar crumbling away in the wall.”
According to Cancer Research UK, there are over 16,000 new cases of melanoma skin cancer each year in the United Kingdom, with over 2,000 deaths annually. After being surgically removed, primary tumors are histologically staged, with even low-risk cases being followed up for a number of years, a process that can be time-consuming for patients and costly for the NHS.
Some reassurance for those with melanoma
The creators of the new test say that it is these low-risk patients that the test is able to identify, offering a degree of reassurance to those diagnosed with the disease, and potentially reducing the number of hospital clinic visits they require.
Dr. Lovat commented: “Our test offers a personalized prognosis as it more accurately predicts if your skin cancer is unlikely to spread.”
She added that the test will aid clinicians to identify genuinely low-risk patients diagnosed with an early-stage melanoma, reducing the number of follow-up appointments for those identified as low risk. It, therefore, offers the opportunity to save the NHS time and money.
Excellent news for those with skin cancer
Phil Brady, chief operating officer of the British Skin Foundation, echoed Dr. Lovat’s comments, saying: “The test can alleviate stress and anxiety for patients caused by this potentially deadly skin cancer, whilst increasing efficiency and reducing costs to the NHS.”
Nick Levell, MD, consultant dermatologist & British Skin Foundation spokesperson, who has not been involved in the research, commented how the arrival of the test was “excellent news,” adding that “people at low risk can be reassured and will not have to attend hospital so often for check-ups”.
The development of the new test AMBLor has been led by Dr. Lovat, in association with the university spin-out company AMLo Biosciences, and is accredited by the National Accreditation Body for the United Kingdom. The test involves tissue sections from the standard biopsy being sent in the post to the lab for analysis and costs £293 plus VAT. Currently available through a private referral service, the Newcastle team have applied for the test to be made available on the NHS.
A version of this article first appeared on Medscape UK.
Their research, published in the British Journal of Dermatology, describes how early-stage melanomas at risk of spreading secrete transforming growth factor beta2 (TGF-beta2), which causes the reduction, or down-regulation, of the proteins AMBRA1 and loricrin, both of which are found in the skin overlaying the tumor. TGF-beta2 also causes the loss of claudin-1, which in turn leads to loss of skin integrity, facilitating ulceration.
Senior author Penny Lovat, PhD, professor of cellular dermatology and oncology at Newcastle University, and chief scientific officer at AMLo Biosciences, explained: “AMBRA1, loricrin, and claudin-1 are all proteins key to maintaining the integrity of the upper layer of the skin,” and that the loss of these proteins causes gaps to develop, allowing the tumor to spread and ulcerate – a process associated with high-risk tumors. Dr. Lovat likened the process to that of “mortar and bricks holding together a wall”, with the loss of these proteins being “like the mortar crumbling away in the wall.”
According to Cancer Research UK, there are over 16,000 new cases of melanoma skin cancer each year in the United Kingdom, with over 2,000 deaths annually. After being surgically removed, primary tumors are histologically staged, with even low-risk cases being followed up for a number of years, a process that can be time-consuming for patients and costly for the NHS.
Some reassurance for those with melanoma
The creators of the new test say that it is these low-risk patients that the test is able to identify, offering a degree of reassurance to those diagnosed with the disease, and potentially reducing the number of hospital clinic visits they require.
Dr. Lovat commented: “Our test offers a personalized prognosis as it more accurately predicts if your skin cancer is unlikely to spread.”
She added that the test will aid clinicians to identify genuinely low-risk patients diagnosed with an early-stage melanoma, reducing the number of follow-up appointments for those identified as low risk. It, therefore, offers the opportunity to save the NHS time and money.
Excellent news for those with skin cancer
Phil Brady, chief operating officer of the British Skin Foundation, echoed Dr. Lovat’s comments, saying: “The test can alleviate stress and anxiety for patients caused by this potentially deadly skin cancer, whilst increasing efficiency and reducing costs to the NHS.”
Nick Levell, MD, consultant dermatologist & British Skin Foundation spokesperson, who has not been involved in the research, commented how the arrival of the test was “excellent news,” adding that “people at low risk can be reassured and will not have to attend hospital so often for check-ups”.
The development of the new test AMBLor has been led by Dr. Lovat, in association with the university spin-out company AMLo Biosciences, and is accredited by the National Accreditation Body for the United Kingdom. The test involves tissue sections from the standard biopsy being sent in the post to the lab for analysis and costs £293 plus VAT. Currently available through a private referral service, the Newcastle team have applied for the test to be made available on the NHS.
A version of this article first appeared on Medscape UK.
Their research, published in the British Journal of Dermatology, describes how early-stage melanomas at risk of spreading secrete transforming growth factor beta2 (TGF-beta2), which causes the reduction, or down-regulation, of the proteins AMBRA1 and loricrin, both of which are found in the skin overlaying the tumor. TGF-beta2 also causes the loss of claudin-1, which in turn leads to loss of skin integrity, facilitating ulceration.
Senior author Penny Lovat, PhD, professor of cellular dermatology and oncology at Newcastle University, and chief scientific officer at AMLo Biosciences, explained: “AMBRA1, loricrin, and claudin-1 are all proteins key to maintaining the integrity of the upper layer of the skin,” and that the loss of these proteins causes gaps to develop, allowing the tumor to spread and ulcerate – a process associated with high-risk tumors. Dr. Lovat likened the process to that of “mortar and bricks holding together a wall”, with the loss of these proteins being “like the mortar crumbling away in the wall.”
According to Cancer Research UK, there are over 16,000 new cases of melanoma skin cancer each year in the United Kingdom, with over 2,000 deaths annually. After being surgically removed, primary tumors are histologically staged, with even low-risk cases being followed up for a number of years, a process that can be time-consuming for patients and costly for the NHS.
Some reassurance for those with melanoma
The creators of the new test say that it is these low-risk patients that the test is able to identify, offering a degree of reassurance to those diagnosed with the disease, and potentially reducing the number of hospital clinic visits they require.
Dr. Lovat commented: “Our test offers a personalized prognosis as it more accurately predicts if your skin cancer is unlikely to spread.”
She added that the test will aid clinicians to identify genuinely low-risk patients diagnosed with an early-stage melanoma, reducing the number of follow-up appointments for those identified as low risk. It, therefore, offers the opportunity to save the NHS time and money.
Excellent news for those with skin cancer
Phil Brady, chief operating officer of the British Skin Foundation, echoed Dr. Lovat’s comments, saying: “The test can alleviate stress and anxiety for patients caused by this potentially deadly skin cancer, whilst increasing efficiency and reducing costs to the NHS.”
Nick Levell, MD, consultant dermatologist & British Skin Foundation spokesperson, who has not been involved in the research, commented how the arrival of the test was “excellent news,” adding that “people at low risk can be reassured and will not have to attend hospital so often for check-ups”.
The development of the new test AMBLor has been led by Dr. Lovat, in association with the university spin-out company AMLo Biosciences, and is accredited by the National Accreditation Body for the United Kingdom. The test involves tissue sections from the standard biopsy being sent in the post to the lab for analysis and costs £293 plus VAT. Currently available through a private referral service, the Newcastle team have applied for the test to be made available on the NHS.
A version of this article first appeared on Medscape UK.
FROM THE BRITISH JOURNAL OF DERMATOLOGY
Antibiotic choices for inpatients with SSTIs vary by race
– in a national cross-sectional study involving over 1,000 patients in 91 hospitals.
The potential racial disparity in management of SSTI was detected after data were adjusted for penicillin allergy history and for MRSA colonization/infection. The data were also adjusted for hospital day (since admission) in order to control for the administration of more empiric therapy early on.
Clindamycin, a beta-lactam alternative, is not recommended as an SSTI treatment given its frequent dosing requirements and high potential for adverse events including Clostridioides difficile infection (DCI). “Clindamycin is an option but it’s considered inferior. ... It covers MRSA but it shouldn’t be a go-to for skin and soft-tissue infections,” said senior author Kimberly Blumenthal, MD, MSc, assistant professor of medicine at Harvard University, and an allergist, immunologist, and drug allergy and epidemiology researcher at Massachusetts General Hospital, both in Boston.
Cefazolin, on the other hand, does not cover MRSA but is “a guideline-recommended first-line antibiotic for cellulitis SSTI in the hospital,” she said in an interview.
The findings, recently published in JAMA Network Open, offer a valuable portrait of the antibiotics being prescribed in the inpatient setting for SSTIs. Vancomycin, which typically is reserved for MRSA, was the most commonly prescribed antibiotic, regardless of race. Piperacillin-tazobactam, a beta-lactam, was the second most commonly prescribed antibiotic, again regardless of race.
Intravenously administered cefazolin was used in 13% of White inpatients versus 5% of Black inpatients. After controlling for kidney disease, diabetes, and ICU location (in addition to hospital day, penicillin allergy history, and MRSA), White inpatients had an increased likelihood of being prescribed cefazolin (adjusted odds ratio, 2.82; 95% confidence interval, 1.41-5.63) and a decreased likelihood of clindamycin use (aOR, 0.54; 95% CI, 0.30-0.96), compared with Black inpatients.
The investigators utilized the Acute Care Hospital Groups network within Vizient, a member-driven health care performance improvement company, to collect data for the study. Most of the hospitals (91%) that submitted data on adult inpatients with cellulitis or SSTIs (without other infections) were in urban settings and 9% were in rural settings; 60% were community hospitals and 40% were academic medical centers. The researchers accounted for “clustering by hospital” – such as the use of internal guidelines – in their methodology.
Differential management and prescribing practices associated with race and ethnicity have been demonstrated for cardiovascular disease and other chronic problems, but “to see such racial differences play out in acute care is striking,” Utibe R. Essien, MD, MPH, assistant professor of medicine at the University of Pittsburgh and a core investigator with the Center for Health Equity Research and Promotion at the Veterans Affairs Pittsburgh Healthcare System, said in an interview.
“In acute care, we tend to practice pretty similarly across the board ... so the findings give me pause,” said Dr. Essien, an internist and a coauthor of the study, who also works with the University of Pittsburgh’s Center for Pharmaceutical Policy and Prescribing.
Also notable was the prevalence of historical penicillin allergy documented in the dataset: 23% in Black inpatients and 18% in White inpatients with SSTI. It’s a surprisingly high prevalence overall, Dr. Blumenthal said, and the racial difference was surprising because penicillin allergy has been commonly described in the literature as being more common in the White population.
Even though penicillin allergy was controlled for in the study, “given that historical penicillin allergies are associated with increased clindamycin use and risk of CDI, but are often disproved with formal testing, racial disparities in penicillin allergy documentation and assessment require additional study,” she and her coauthors wrote.
Ideally, Dr. Blumenthal said, all inpatients would have access to allergy consultations or testing or some sort of infrastructure for assessing a history of penicillin allergy. At Mass General, allergy consults and challenge doses of beta-lactams (also called graded challenges) are frequently employed.
The study did not collect data on income, educational level, and other structural vulnerability factors. More research is needed to better understand “what’s going on in acute care settings and what the potential drivers of disparities may be,” said Dr. Essien, who co-authored a recent JAMA editorial on “achieving pharmacoequity” to reduce health disparities.
“If guidelines suggest that medication A is the ideal and optimal treatment, we really have to do our best to ensure that every patient, regardless of race or ethnicity, can get that treatment,” he said.
In the study, race was extracted from the medical record and may not have been correctly assigned, the authors noted. “Other race” was not specified in the dataset, and Hispanic ethnicity was not captured. The number of individuals identified as Asian and other races was small, prompting the researchers to focus on antibiotic use in Black and White patients (224 and 854 patients, respectively).
Dr. Blumenthal and Dr. Essien both reported that they had no relevant disclosures. The study was supported with National Institutes of Health grants and the Massachusetts General Hospital department of medicine transformative scholar program.
– in a national cross-sectional study involving over 1,000 patients in 91 hospitals.
The potential racial disparity in management of SSTI was detected after data were adjusted for penicillin allergy history and for MRSA colonization/infection. The data were also adjusted for hospital day (since admission) in order to control for the administration of more empiric therapy early on.
Clindamycin, a beta-lactam alternative, is not recommended as an SSTI treatment given its frequent dosing requirements and high potential for adverse events including Clostridioides difficile infection (DCI). “Clindamycin is an option but it’s considered inferior. ... It covers MRSA but it shouldn’t be a go-to for skin and soft-tissue infections,” said senior author Kimberly Blumenthal, MD, MSc, assistant professor of medicine at Harvard University, and an allergist, immunologist, and drug allergy and epidemiology researcher at Massachusetts General Hospital, both in Boston.
Cefazolin, on the other hand, does not cover MRSA but is “a guideline-recommended first-line antibiotic for cellulitis SSTI in the hospital,” she said in an interview.
The findings, recently published in JAMA Network Open, offer a valuable portrait of the antibiotics being prescribed in the inpatient setting for SSTIs. Vancomycin, which typically is reserved for MRSA, was the most commonly prescribed antibiotic, regardless of race. Piperacillin-tazobactam, a beta-lactam, was the second most commonly prescribed antibiotic, again regardless of race.
Intravenously administered cefazolin was used in 13% of White inpatients versus 5% of Black inpatients. After controlling for kidney disease, diabetes, and ICU location (in addition to hospital day, penicillin allergy history, and MRSA), White inpatients had an increased likelihood of being prescribed cefazolin (adjusted odds ratio, 2.82; 95% confidence interval, 1.41-5.63) and a decreased likelihood of clindamycin use (aOR, 0.54; 95% CI, 0.30-0.96), compared with Black inpatients.
The investigators utilized the Acute Care Hospital Groups network within Vizient, a member-driven health care performance improvement company, to collect data for the study. Most of the hospitals (91%) that submitted data on adult inpatients with cellulitis or SSTIs (without other infections) were in urban settings and 9% were in rural settings; 60% were community hospitals and 40% were academic medical centers. The researchers accounted for “clustering by hospital” – such as the use of internal guidelines – in their methodology.
Differential management and prescribing practices associated with race and ethnicity have been demonstrated for cardiovascular disease and other chronic problems, but “to see such racial differences play out in acute care is striking,” Utibe R. Essien, MD, MPH, assistant professor of medicine at the University of Pittsburgh and a core investigator with the Center for Health Equity Research and Promotion at the Veterans Affairs Pittsburgh Healthcare System, said in an interview.
“In acute care, we tend to practice pretty similarly across the board ... so the findings give me pause,” said Dr. Essien, an internist and a coauthor of the study, who also works with the University of Pittsburgh’s Center for Pharmaceutical Policy and Prescribing.
Also notable was the prevalence of historical penicillin allergy documented in the dataset: 23% in Black inpatients and 18% in White inpatients with SSTI. It’s a surprisingly high prevalence overall, Dr. Blumenthal said, and the racial difference was surprising because penicillin allergy has been commonly described in the literature as being more common in the White population.
Even though penicillin allergy was controlled for in the study, “given that historical penicillin allergies are associated with increased clindamycin use and risk of CDI, but are often disproved with formal testing, racial disparities in penicillin allergy documentation and assessment require additional study,” she and her coauthors wrote.
Ideally, Dr. Blumenthal said, all inpatients would have access to allergy consultations or testing or some sort of infrastructure for assessing a history of penicillin allergy. At Mass General, allergy consults and challenge doses of beta-lactams (also called graded challenges) are frequently employed.
The study did not collect data on income, educational level, and other structural vulnerability factors. More research is needed to better understand “what’s going on in acute care settings and what the potential drivers of disparities may be,” said Dr. Essien, who co-authored a recent JAMA editorial on “achieving pharmacoequity” to reduce health disparities.
“If guidelines suggest that medication A is the ideal and optimal treatment, we really have to do our best to ensure that every patient, regardless of race or ethnicity, can get that treatment,” he said.
In the study, race was extracted from the medical record and may not have been correctly assigned, the authors noted. “Other race” was not specified in the dataset, and Hispanic ethnicity was not captured. The number of individuals identified as Asian and other races was small, prompting the researchers to focus on antibiotic use in Black and White patients (224 and 854 patients, respectively).
Dr. Blumenthal and Dr. Essien both reported that they had no relevant disclosures. The study was supported with National Institutes of Health grants and the Massachusetts General Hospital department of medicine transformative scholar program.
– in a national cross-sectional study involving over 1,000 patients in 91 hospitals.
The potential racial disparity in management of SSTI was detected after data were adjusted for penicillin allergy history and for MRSA colonization/infection. The data were also adjusted for hospital day (since admission) in order to control for the administration of more empiric therapy early on.
Clindamycin, a beta-lactam alternative, is not recommended as an SSTI treatment given its frequent dosing requirements and high potential for adverse events including Clostridioides difficile infection (DCI). “Clindamycin is an option but it’s considered inferior. ... It covers MRSA but it shouldn’t be a go-to for skin and soft-tissue infections,” said senior author Kimberly Blumenthal, MD, MSc, assistant professor of medicine at Harvard University, and an allergist, immunologist, and drug allergy and epidemiology researcher at Massachusetts General Hospital, both in Boston.
Cefazolin, on the other hand, does not cover MRSA but is “a guideline-recommended first-line antibiotic for cellulitis SSTI in the hospital,” she said in an interview.
The findings, recently published in JAMA Network Open, offer a valuable portrait of the antibiotics being prescribed in the inpatient setting for SSTIs. Vancomycin, which typically is reserved for MRSA, was the most commonly prescribed antibiotic, regardless of race. Piperacillin-tazobactam, a beta-lactam, was the second most commonly prescribed antibiotic, again regardless of race.
Intravenously administered cefazolin was used in 13% of White inpatients versus 5% of Black inpatients. After controlling for kidney disease, diabetes, and ICU location (in addition to hospital day, penicillin allergy history, and MRSA), White inpatients had an increased likelihood of being prescribed cefazolin (adjusted odds ratio, 2.82; 95% confidence interval, 1.41-5.63) and a decreased likelihood of clindamycin use (aOR, 0.54; 95% CI, 0.30-0.96), compared with Black inpatients.
The investigators utilized the Acute Care Hospital Groups network within Vizient, a member-driven health care performance improvement company, to collect data for the study. Most of the hospitals (91%) that submitted data on adult inpatients with cellulitis or SSTIs (without other infections) were in urban settings and 9% were in rural settings; 60% were community hospitals and 40% were academic medical centers. The researchers accounted for “clustering by hospital” – such as the use of internal guidelines – in their methodology.
Differential management and prescribing practices associated with race and ethnicity have been demonstrated for cardiovascular disease and other chronic problems, but “to see such racial differences play out in acute care is striking,” Utibe R. Essien, MD, MPH, assistant professor of medicine at the University of Pittsburgh and a core investigator with the Center for Health Equity Research and Promotion at the Veterans Affairs Pittsburgh Healthcare System, said in an interview.
“In acute care, we tend to practice pretty similarly across the board ... so the findings give me pause,” said Dr. Essien, an internist and a coauthor of the study, who also works with the University of Pittsburgh’s Center for Pharmaceutical Policy and Prescribing.
Also notable was the prevalence of historical penicillin allergy documented in the dataset: 23% in Black inpatients and 18% in White inpatients with SSTI. It’s a surprisingly high prevalence overall, Dr. Blumenthal said, and the racial difference was surprising because penicillin allergy has been commonly described in the literature as being more common in the White population.
Even though penicillin allergy was controlled for in the study, “given that historical penicillin allergies are associated with increased clindamycin use and risk of CDI, but are often disproved with formal testing, racial disparities in penicillin allergy documentation and assessment require additional study,” she and her coauthors wrote.
Ideally, Dr. Blumenthal said, all inpatients would have access to allergy consultations or testing or some sort of infrastructure for assessing a history of penicillin allergy. At Mass General, allergy consults and challenge doses of beta-lactams (also called graded challenges) are frequently employed.
The study did not collect data on income, educational level, and other structural vulnerability factors. More research is needed to better understand “what’s going on in acute care settings and what the potential drivers of disparities may be,” said Dr. Essien, who co-authored a recent JAMA editorial on “achieving pharmacoequity” to reduce health disparities.
“If guidelines suggest that medication A is the ideal and optimal treatment, we really have to do our best to ensure that every patient, regardless of race or ethnicity, can get that treatment,” he said.
In the study, race was extracted from the medical record and may not have been correctly assigned, the authors noted. “Other race” was not specified in the dataset, and Hispanic ethnicity was not captured. The number of individuals identified as Asian and other races was small, prompting the researchers to focus on antibiotic use in Black and White patients (224 and 854 patients, respectively).
Dr. Blumenthal and Dr. Essien both reported that they had no relevant disclosures. The study was supported with National Institutes of Health grants and the Massachusetts General Hospital department of medicine transformative scholar program.
FROM JAMA NETWORK OPEN
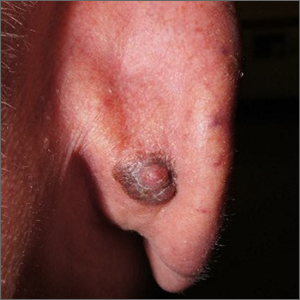
Dark plaque on back of ear
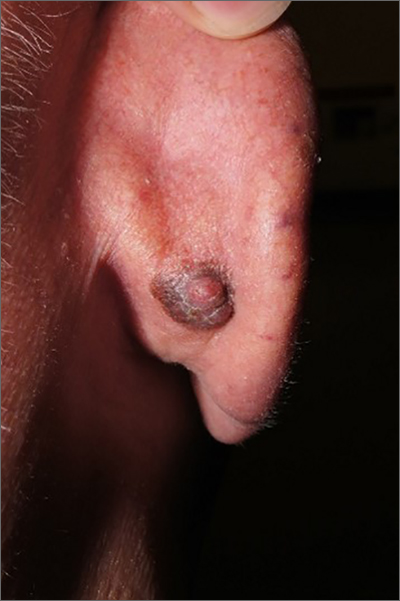
Dermoscopic findings were consistent with a melanocytic lesion and a scoop shave biopsy revealed a 2.7 mm thick nodular melanoma.
Melanoma is the most lethal skin cancer in the United States. The likelihood of metastatic spread to lymph nodes statistically increases beyond a probability of 5% when patients have primary lesions thicker than 0.8 mm.1 Thus, for patients with tumors thicker than 0.8 mm, or some other high-risk features such as high mitotic index, a sentinel lymph node biopsy (SLNB) is recommended. This patient underwent wide local excision and reconstruction of his ear. An SLNB was also performed and the results were negative.
The patient returned for a complete skin exam every 3 months. Ten months after the excision, he presented with episodes of headache and confusion. Magnetic resonance imaging revealed metastasis to the brain; a biopsy confirmed that it was melanoma. Two months later, after attempts at resection of the brain metastasis, the patient died.
This case demonstrates that patients with thick melanoma are at continued risk for recurrence and poor outcomes; they benefit from close surveillance and work-up of unusual symptoms that might suggest metastases. Phase 3 trials are currently underway to consider the use of adjuvant therapy in patients with advanced stage II melanoma who, on average, have worse outcomes than patients with early-stage III disease.2
Text courtesy of Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. Photos courtesy of Jonathan Karnes, MD (copyright retained).
1. NCCN Guidelines Version 1.2022 Melanoma: Cutaneous. National Comprehensive Cancer Network. December 3, 2021. Accessed January 4, 2022. https://www.nccn.org/professionals/physician_gls/pdf/cutaneous_melanoma.pdf
2. Poklepovic AS, Luke JJ. Considering adjuvant therapy for stage II melanoma. Cancer. 2020;126:1166-1174. doi: 10.1002/cncr.32585

Dermoscopic findings were consistent with a melanocytic lesion and a scoop shave biopsy revealed a 2.7 mm thick nodular melanoma.
Melanoma is the most lethal skin cancer in the United States. The likelihood of metastatic spread to lymph nodes statistically increases beyond a probability of 5% when patients have primary lesions thicker than 0.8 mm.1 Thus, for patients with tumors thicker than 0.8 mm, or some other high-risk features such as high mitotic index, a sentinel lymph node biopsy (SLNB) is recommended. This patient underwent wide local excision and reconstruction of his ear. An SLNB was also performed and the results were negative.
The patient returned for a complete skin exam every 3 months. Ten months after the excision, he presented with episodes of headache and confusion. Magnetic resonance imaging revealed metastasis to the brain; a biopsy confirmed that it was melanoma. Two months later, after attempts at resection of the brain metastasis, the patient died.
This case demonstrates that patients with thick melanoma are at continued risk for recurrence and poor outcomes; they benefit from close surveillance and work-up of unusual symptoms that might suggest metastases. Phase 3 trials are currently underway to consider the use of adjuvant therapy in patients with advanced stage II melanoma who, on average, have worse outcomes than patients with early-stage III disease.2
Text courtesy of Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. Photos courtesy of Jonathan Karnes, MD (copyright retained).

Dermoscopic findings were consistent with a melanocytic lesion and a scoop shave biopsy revealed a 2.7 mm thick nodular melanoma.
Melanoma is the most lethal skin cancer in the United States. The likelihood of metastatic spread to lymph nodes statistically increases beyond a probability of 5% when patients have primary lesions thicker than 0.8 mm.1 Thus, for patients with tumors thicker than 0.8 mm, or some other high-risk features such as high mitotic index, a sentinel lymph node biopsy (SLNB) is recommended. This patient underwent wide local excision and reconstruction of his ear. An SLNB was also performed and the results were negative.
The patient returned for a complete skin exam every 3 months. Ten months after the excision, he presented with episodes of headache and confusion. Magnetic resonance imaging revealed metastasis to the brain; a biopsy confirmed that it was melanoma. Two months later, after attempts at resection of the brain metastasis, the patient died.
This case demonstrates that patients with thick melanoma are at continued risk for recurrence and poor outcomes; they benefit from close surveillance and work-up of unusual symptoms that might suggest metastases. Phase 3 trials are currently underway to consider the use of adjuvant therapy in patients with advanced stage II melanoma who, on average, have worse outcomes than patients with early-stage III disease.2
Text courtesy of Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. Photos courtesy of Jonathan Karnes, MD (copyright retained).
1. NCCN Guidelines Version 1.2022 Melanoma: Cutaneous. National Comprehensive Cancer Network. December 3, 2021. Accessed January 4, 2022. https://www.nccn.org/professionals/physician_gls/pdf/cutaneous_melanoma.pdf
2. Poklepovic AS, Luke JJ. Considering adjuvant therapy for stage II melanoma. Cancer. 2020;126:1166-1174. doi: 10.1002/cncr.32585
1. NCCN Guidelines Version 1.2022 Melanoma: Cutaneous. National Comprehensive Cancer Network. December 3, 2021. Accessed January 4, 2022. https://www.nccn.org/professionals/physician_gls/pdf/cutaneous_melanoma.pdf
2. Poklepovic AS, Luke JJ. Considering adjuvant therapy for stage II melanoma. Cancer. 2020;126:1166-1174. doi: 10.1002/cncr.32585
Survey: Medical cannabis use for skin conditions lags behind interest, acceptance
A , according to the results of a recent survey.
Almost 89% of respondents were in favor of medical cannabis use for dermatologic diseases, and 73% said that they would be comfortable seeing a dermatologist who recommended such products to them, Samuel Yeroushalmi, a 4th-year medical student at George Washington University, Washington, and associates reported.
“Consumers and patients are already using MCPs [medical cannabis products] to treat inflammatory skin conditions, such as acne, rosacea, atopic dermatitis, and psoriasis, even without guidance from a dermatologist. While acceptance was high, there were clear barriers reported limiting use and uptake, such as patient skepticism and a lack of understanding,” Adam Friedman, MD, senior author and chair of the department of dermatology at the university, said in a separate statement.
Dermatologic use of OTC cannabis products without the recommendation of a dermatologist was reported by 18% of the 504 of 700 adults who responded in the SurveyMonkey online panel. Of the two-thirds who had seen a dermatologist, 20% received a recommendation for an OTC product and 11% were recommended a product that required a department of health medical card, the investigators said.
Uptake among the patients who did receive a recommendation, however, was high: 76% for OTC products and 72% for those that required a medical card. Among those who had received an OTC recommendation, 32% used the cannabis product for psoriasis and 30% each for acne and rosacea, Mr. Yeroushalmi and his coauthors said.
The most common indication among the respondents with dermatologist recommendations for products requiring a medical card was for acne (68%), followed by psoriasis and rosacea (28% each). Cost was the main deterrent (60%) for those who declined to use the recommended cannabis product, with skepticism, limited understanding, and product illegality in their state each at 50%, the researchers said.
“Though cost and legality concerns are nonmodifiable barriers, dermatologists have an opportunity to educate those who know little in the way of medical cannabis or are skeptic[s],” they wrote. The survey results show that many patients are interested, and “the future should be bright for MCPs; we just need to show and disseminate the science,” Dr. Friedman commented in the statement.
One of the authors was from the University of Maryland, College Park. The authors had no disclosures to report.
A , according to the results of a recent survey.
Almost 89% of respondents were in favor of medical cannabis use for dermatologic diseases, and 73% said that they would be comfortable seeing a dermatologist who recommended such products to them, Samuel Yeroushalmi, a 4th-year medical student at George Washington University, Washington, and associates reported.
“Consumers and patients are already using MCPs [medical cannabis products] to treat inflammatory skin conditions, such as acne, rosacea, atopic dermatitis, and psoriasis, even without guidance from a dermatologist. While acceptance was high, there were clear barriers reported limiting use and uptake, such as patient skepticism and a lack of understanding,” Adam Friedman, MD, senior author and chair of the department of dermatology at the university, said in a separate statement.
Dermatologic use of OTC cannabis products without the recommendation of a dermatologist was reported by 18% of the 504 of 700 adults who responded in the SurveyMonkey online panel. Of the two-thirds who had seen a dermatologist, 20% received a recommendation for an OTC product and 11% were recommended a product that required a department of health medical card, the investigators said.
Uptake among the patients who did receive a recommendation, however, was high: 76% for OTC products and 72% for those that required a medical card. Among those who had received an OTC recommendation, 32% used the cannabis product for psoriasis and 30% each for acne and rosacea, Mr. Yeroushalmi and his coauthors said.
The most common indication among the respondents with dermatologist recommendations for products requiring a medical card was for acne (68%), followed by psoriasis and rosacea (28% each). Cost was the main deterrent (60%) for those who declined to use the recommended cannabis product, with skepticism, limited understanding, and product illegality in their state each at 50%, the researchers said.
“Though cost and legality concerns are nonmodifiable barriers, dermatologists have an opportunity to educate those who know little in the way of medical cannabis or are skeptic[s],” they wrote. The survey results show that many patients are interested, and “the future should be bright for MCPs; we just need to show and disseminate the science,” Dr. Friedman commented in the statement.
One of the authors was from the University of Maryland, College Park. The authors had no disclosures to report.
A , according to the results of a recent survey.
Almost 89% of respondents were in favor of medical cannabis use for dermatologic diseases, and 73% said that they would be comfortable seeing a dermatologist who recommended such products to them, Samuel Yeroushalmi, a 4th-year medical student at George Washington University, Washington, and associates reported.
“Consumers and patients are already using MCPs [medical cannabis products] to treat inflammatory skin conditions, such as acne, rosacea, atopic dermatitis, and psoriasis, even without guidance from a dermatologist. While acceptance was high, there were clear barriers reported limiting use and uptake, such as patient skepticism and a lack of understanding,” Adam Friedman, MD, senior author and chair of the department of dermatology at the university, said in a separate statement.
Dermatologic use of OTC cannabis products without the recommendation of a dermatologist was reported by 18% of the 504 of 700 adults who responded in the SurveyMonkey online panel. Of the two-thirds who had seen a dermatologist, 20% received a recommendation for an OTC product and 11% were recommended a product that required a department of health medical card, the investigators said.
Uptake among the patients who did receive a recommendation, however, was high: 76% for OTC products and 72% for those that required a medical card. Among those who had received an OTC recommendation, 32% used the cannabis product for psoriasis and 30% each for acne and rosacea, Mr. Yeroushalmi and his coauthors said.
The most common indication among the respondents with dermatologist recommendations for products requiring a medical card was for acne (68%), followed by psoriasis and rosacea (28% each). Cost was the main deterrent (60%) for those who declined to use the recommended cannabis product, with skepticism, limited understanding, and product illegality in their state each at 50%, the researchers said.
“Though cost and legality concerns are nonmodifiable barriers, dermatologists have an opportunity to educate those who know little in the way of medical cannabis or are skeptic[s],” they wrote. The survey results show that many patients are interested, and “the future should be bright for MCPs; we just need to show and disseminate the science,” Dr. Friedman commented in the statement.
One of the authors was from the University of Maryland, College Park. The authors had no disclosures to report.
FROM JOURNAL OF DRUGS IN DERMATOLOGY
FDA approves two JAK-1 inhibitors for moderate to severe atopic dermatitis
The available for this indication in the United States.
“It’s big news because a few years ago we didn’t have any systemic treatments that are safer than the classical immunosuppressants like cyclosporine and methotrexate,” Emma Guttman-Yassky, MD, PhD, Waldman professor and system chair of dermatology at the Icahn School of Medicine at Mount Sinai in New York, told this news organization commenting on upadacitinib’s approval.
“The only oral approved drug for AD up to now was oral prednisone, which has terrible safety concerns. This is basically the first oral medication that we can provide our patients for long-term use.”
Upadacitinib
The approval of upadacitinib (Rinvoq), marketed by AbbVie, for moderate to severe AD in patients ages 12 and older, comes on the heels of findings from three pivotal phase 3 studies involving more than 2,500 adults and children 12 years of age and older with moderate to severe AD: Measure Up 1 and 2, led by Dr. Guttman-Yassky, which evaluated upadacitinib compared with placebo, and AD UP, which compared upadacitinib along with topical corticosteroids, compared with placebo.
Across the three studies, upadacitinib – both 15 mg and 30 mg once daily monotherapy – met all primary and secondary endpoints at week 16, with some patients achieving higher levels of skin clearance based on the Eczema Area and Severity Index 90 (EASI-90) and EASI-100.
“I always say that patients with AD need options,” Dr. Guttman-Yassky said. “We need biologics. We need oral medications. Not everybody likes an injectable. The plus of the class of JAK inhibitors in general is the quick onset of action.” Many patients in her clinic are maintained on upadacitinib more than two years later “and are super happy,” she said. “Many of them failed cyclosporine and other immunosuppressants such as methotrexate and prednisone.”
She predicted that health insurance companies will find coverage cost-effective “because it sets a new bar for efficacy, and because many patients have failed other treatments.”
Abrocitinib
Abrocitinib (Cibinqo), marketed by Pfizer, was approved for adults with moderate to severe AD. The approval was based on results of five clinical trials from a large-scale clinical trial program of more than 1,600 patients. The recommended doses are 100 mg and 200 mg, with the 200 mg dose recommended for patients who are not responding to the 100 mg dose.
The labeling of abrocitinib and upadacitinib include a boxed warning for JAK inhibitors, regarding the risk of serious infections, mortality, malignancy, major adverse cardiovascular events, and thrombosis.
Dr. Guttman-Yassky has served as a principal investigator for AbbVie and has received consulting fees from the company.
The available for this indication in the United States.
“It’s big news because a few years ago we didn’t have any systemic treatments that are safer than the classical immunosuppressants like cyclosporine and methotrexate,” Emma Guttman-Yassky, MD, PhD, Waldman professor and system chair of dermatology at the Icahn School of Medicine at Mount Sinai in New York, told this news organization commenting on upadacitinib’s approval.
“The only oral approved drug for AD up to now was oral prednisone, which has terrible safety concerns. This is basically the first oral medication that we can provide our patients for long-term use.”
Upadacitinib
The approval of upadacitinib (Rinvoq), marketed by AbbVie, for moderate to severe AD in patients ages 12 and older, comes on the heels of findings from three pivotal phase 3 studies involving more than 2,500 adults and children 12 years of age and older with moderate to severe AD: Measure Up 1 and 2, led by Dr. Guttman-Yassky, which evaluated upadacitinib compared with placebo, and AD UP, which compared upadacitinib along with topical corticosteroids, compared with placebo.
Across the three studies, upadacitinib – both 15 mg and 30 mg once daily monotherapy – met all primary and secondary endpoints at week 16, with some patients achieving higher levels of skin clearance based on the Eczema Area and Severity Index 90 (EASI-90) and EASI-100.
“I always say that patients with AD need options,” Dr. Guttman-Yassky said. “We need biologics. We need oral medications. Not everybody likes an injectable. The plus of the class of JAK inhibitors in general is the quick onset of action.” Many patients in her clinic are maintained on upadacitinib more than two years later “and are super happy,” she said. “Many of them failed cyclosporine and other immunosuppressants such as methotrexate and prednisone.”
She predicted that health insurance companies will find coverage cost-effective “because it sets a new bar for efficacy, and because many patients have failed other treatments.”
Abrocitinib
Abrocitinib (Cibinqo), marketed by Pfizer, was approved for adults with moderate to severe AD. The approval was based on results of five clinical trials from a large-scale clinical trial program of more than 1,600 patients. The recommended doses are 100 mg and 200 mg, with the 200 mg dose recommended for patients who are not responding to the 100 mg dose.
The labeling of abrocitinib and upadacitinib include a boxed warning for JAK inhibitors, regarding the risk of serious infections, mortality, malignancy, major adverse cardiovascular events, and thrombosis.
Dr. Guttman-Yassky has served as a principal investigator for AbbVie and has received consulting fees from the company.
The available for this indication in the United States.
“It’s big news because a few years ago we didn’t have any systemic treatments that are safer than the classical immunosuppressants like cyclosporine and methotrexate,” Emma Guttman-Yassky, MD, PhD, Waldman professor and system chair of dermatology at the Icahn School of Medicine at Mount Sinai in New York, told this news organization commenting on upadacitinib’s approval.
“The only oral approved drug for AD up to now was oral prednisone, which has terrible safety concerns. This is basically the first oral medication that we can provide our patients for long-term use.”
Upadacitinib
The approval of upadacitinib (Rinvoq), marketed by AbbVie, for moderate to severe AD in patients ages 12 and older, comes on the heels of findings from three pivotal phase 3 studies involving more than 2,500 adults and children 12 years of age and older with moderate to severe AD: Measure Up 1 and 2, led by Dr. Guttman-Yassky, which evaluated upadacitinib compared with placebo, and AD UP, which compared upadacitinib along with topical corticosteroids, compared with placebo.
Across the three studies, upadacitinib – both 15 mg and 30 mg once daily monotherapy – met all primary and secondary endpoints at week 16, with some patients achieving higher levels of skin clearance based on the Eczema Area and Severity Index 90 (EASI-90) and EASI-100.
“I always say that patients with AD need options,” Dr. Guttman-Yassky said. “We need biologics. We need oral medications. Not everybody likes an injectable. The plus of the class of JAK inhibitors in general is the quick onset of action.” Many patients in her clinic are maintained on upadacitinib more than two years later “and are super happy,” she said. “Many of them failed cyclosporine and other immunosuppressants such as methotrexate and prednisone.”
She predicted that health insurance companies will find coverage cost-effective “because it sets a new bar for efficacy, and because many patients have failed other treatments.”
Abrocitinib
Abrocitinib (Cibinqo), marketed by Pfizer, was approved for adults with moderate to severe AD. The approval was based on results of five clinical trials from a large-scale clinical trial program of more than 1,600 patients. The recommended doses are 100 mg and 200 mg, with the 200 mg dose recommended for patients who are not responding to the 100 mg dose.
The labeling of abrocitinib and upadacitinib include a boxed warning for JAK inhibitors, regarding the risk of serious infections, mortality, malignancy, major adverse cardiovascular events, and thrombosis.
Dr. Guttman-Yassky has served as a principal investigator for AbbVie and has received consulting fees from the company.
FDA updates status of iPLEDGE access problems
The, one month after a modified program was launched, the Food and Drug Administration announced on Jan. 14.
The IPMG has “created a new tool within the system to help resolve account access for some user groups without using the call center. This tool is intended to allow prescribers and designees to send login links directly to their patients’ desired email address through the Manage Patients page of the iPLEDGE REMS portal,” the FDA statement said.
“Prescribers can also send login links to their designees still having difficulty accessing their iPLEDGE account,” and users should check their emails for messages from iPLEDGE, including spam folders, the FDA advises. The iPLEDGE strategy is designed to prevent fetal exposure to isotretinoin, which is highly teratogenic.
Days after the new, gender-neutral approach to the isotretinoin risk mitigation program was launched on Dec. 13, the FDA convened an emergency meeting with representatives from the American Academy of Dermatology Association (AADA) to discuss the problematic rollout of the program, which was described as disastrous, chaotic, and a failure, with dermatologists on Twitter and elsewhere expressing anger and frustration over not being able to access the program or reach the call center.
A statement by the FDA on Dec. 23 followed, urging manufacturers to develop solutions for the website and to work with the AADA and pharmacy organizations to find solutions that would minimize treatment interruptions during the transition.
The modified REMS, launched on Dec. 13, is designed to make it more inclusive for transgender patients prescribed isotretinoin. Instead of three risk categories (females of reproductive potential, females not of reproductive potential, and males), patients who are prescribed isotretinoin for acne are assigned to one of two risk categories: those who can get pregnant and those who cannot get pregnant.
In the Jan. 14 statement, the FDA notes that the agency is continuing to work with the IPMG regarding the problems clinicians, pharmacists, and patients have had with accessing iPLEDGE over the last month.
“Although there has been progress, there is a significant amount of work still to be done,” the FDA acknowledged. “While we consider potential steps within the scope of FDA’s authorities, we will continue to meet with the IPMG for updates on the status of the problems with the iPLEDGE REMS and their progress towards having the system work as intended for all users.”
The, one month after a modified program was launched, the Food and Drug Administration announced on Jan. 14.
The IPMG has “created a new tool within the system to help resolve account access for some user groups without using the call center. This tool is intended to allow prescribers and designees to send login links directly to their patients’ desired email address through the Manage Patients page of the iPLEDGE REMS portal,” the FDA statement said.
“Prescribers can also send login links to their designees still having difficulty accessing their iPLEDGE account,” and users should check their emails for messages from iPLEDGE, including spam folders, the FDA advises. The iPLEDGE strategy is designed to prevent fetal exposure to isotretinoin, which is highly teratogenic.
Days after the new, gender-neutral approach to the isotretinoin risk mitigation program was launched on Dec. 13, the FDA convened an emergency meeting with representatives from the American Academy of Dermatology Association (AADA) to discuss the problematic rollout of the program, which was described as disastrous, chaotic, and a failure, with dermatologists on Twitter and elsewhere expressing anger and frustration over not being able to access the program or reach the call center.
A statement by the FDA on Dec. 23 followed, urging manufacturers to develop solutions for the website and to work with the AADA and pharmacy organizations to find solutions that would minimize treatment interruptions during the transition.
The modified REMS, launched on Dec. 13, is designed to make it more inclusive for transgender patients prescribed isotretinoin. Instead of three risk categories (females of reproductive potential, females not of reproductive potential, and males), patients who are prescribed isotretinoin for acne are assigned to one of two risk categories: those who can get pregnant and those who cannot get pregnant.
In the Jan. 14 statement, the FDA notes that the agency is continuing to work with the IPMG regarding the problems clinicians, pharmacists, and patients have had with accessing iPLEDGE over the last month.
“Although there has been progress, there is a significant amount of work still to be done,” the FDA acknowledged. “While we consider potential steps within the scope of FDA’s authorities, we will continue to meet with the IPMG for updates on the status of the problems with the iPLEDGE REMS and their progress towards having the system work as intended for all users.”
The, one month after a modified program was launched, the Food and Drug Administration announced on Jan. 14.
The IPMG has “created a new tool within the system to help resolve account access for some user groups without using the call center. This tool is intended to allow prescribers and designees to send login links directly to their patients’ desired email address through the Manage Patients page of the iPLEDGE REMS portal,” the FDA statement said.
“Prescribers can also send login links to their designees still having difficulty accessing their iPLEDGE account,” and users should check their emails for messages from iPLEDGE, including spam folders, the FDA advises. The iPLEDGE strategy is designed to prevent fetal exposure to isotretinoin, which is highly teratogenic.
Days after the new, gender-neutral approach to the isotretinoin risk mitigation program was launched on Dec. 13, the FDA convened an emergency meeting with representatives from the American Academy of Dermatology Association (AADA) to discuss the problematic rollout of the program, which was described as disastrous, chaotic, and a failure, with dermatologists on Twitter and elsewhere expressing anger and frustration over not being able to access the program or reach the call center.
A statement by the FDA on Dec. 23 followed, urging manufacturers to develop solutions for the website and to work with the AADA and pharmacy organizations to find solutions that would minimize treatment interruptions during the transition.
The modified REMS, launched on Dec. 13, is designed to make it more inclusive for transgender patients prescribed isotretinoin. Instead of three risk categories (females of reproductive potential, females not of reproductive potential, and males), patients who are prescribed isotretinoin for acne are assigned to one of two risk categories: those who can get pregnant and those who cannot get pregnant.
In the Jan. 14 statement, the FDA notes that the agency is continuing to work with the IPMG regarding the problems clinicians, pharmacists, and patients have had with accessing iPLEDGE over the last month.
“Although there has been progress, there is a significant amount of work still to be done,” the FDA acknowledged. “While we consider potential steps within the scope of FDA’s authorities, we will continue to meet with the IPMG for updates on the status of the problems with the iPLEDGE REMS and their progress towards having the system work as intended for all users.”
75-year-old White male presenting with progressive pruritus and a worsening rash
, although it can also be contracted through contaminated bedding and clothing. It can affect all races and ages.
Patients typically present with extremely pruritic, symmetric papules and excoriations. In nodular scabies, nodules and large papules are seen on exam. Thin lines in the skin called burrows may be present, especially in the webs between fingers. Female mites create burrows as they tunnel through the epidermis and lay eggs. The wrists, areola, waistline, and groin may all be involved, creating an imaginary circle between the areas described as the “circle of Hebra.” Penile and scrotal lesions are common in men.
Patients usually experience worse pruritus at night, which disturbs sleep. Crusted scabies is a severe form of scabies more often seen in those with immunocompromised immune systems. Clinically, thick crusted and scaly patches are present that are teeming with mites.
Diagnosis can be confirmed by performing a scabies prep, during which a burrow is scraped with a surgical blade. A drop of mineral oil is placed on the skin cells. The mite, ova, and feces can be visualized under the microscope. Wrists and hands usually have the highest yield for finding the parasites.
Topical treatments include permethrin 5% cream, lindane, benzyl benzoate, and crotamiton, and should be applied as two treatments a week apart. In the United States, permethrin is most commonly used. Ivermectin pills are used off label and are very effective and may be repeated for 1-2 weeks. All household contacts should be treated. Patients may still have pruritus for 2-4 weeks following treatment.
In this patient, a scabies prep was performed prior to performing repeat skin biopsies. Microscopic examination revealed ova, one mite, and feces. Treatment was initiated with ivermectin and permethrin.
Photos and case were submitted by Susannah Berke, MD, and Damon McClain, MD, Three Rivers Dermatology, Coraopolis, Pa.; and Dr. Bilu Martin.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to [email protected].
, although it can also be contracted through contaminated bedding and clothing. It can affect all races and ages.
Patients typically present with extremely pruritic, symmetric papules and excoriations. In nodular scabies, nodules and large papules are seen on exam. Thin lines in the skin called burrows may be present, especially in the webs between fingers. Female mites create burrows as they tunnel through the epidermis and lay eggs. The wrists, areola, waistline, and groin may all be involved, creating an imaginary circle between the areas described as the “circle of Hebra.” Penile and scrotal lesions are common in men.
Patients usually experience worse pruritus at night, which disturbs sleep. Crusted scabies is a severe form of scabies more often seen in those with immunocompromised immune systems. Clinically, thick crusted and scaly patches are present that are teeming with mites.
Diagnosis can be confirmed by performing a scabies prep, during which a burrow is scraped with a surgical blade. A drop of mineral oil is placed on the skin cells. The mite, ova, and feces can be visualized under the microscope. Wrists and hands usually have the highest yield for finding the parasites.
Topical treatments include permethrin 5% cream, lindane, benzyl benzoate, and crotamiton, and should be applied as two treatments a week apart. In the United States, permethrin is most commonly used. Ivermectin pills are used off label and are very effective and may be repeated for 1-2 weeks. All household contacts should be treated. Patients may still have pruritus for 2-4 weeks following treatment.
In this patient, a scabies prep was performed prior to performing repeat skin biopsies. Microscopic examination revealed ova, one mite, and feces. Treatment was initiated with ivermectin and permethrin.
Photos and case were submitted by Susannah Berke, MD, and Damon McClain, MD, Three Rivers Dermatology, Coraopolis, Pa.; and Dr. Bilu Martin.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to [email protected].
, although it can also be contracted through contaminated bedding and clothing. It can affect all races and ages.
Patients typically present with extremely pruritic, symmetric papules and excoriations. In nodular scabies, nodules and large papules are seen on exam. Thin lines in the skin called burrows may be present, especially in the webs between fingers. Female mites create burrows as they tunnel through the epidermis and lay eggs. The wrists, areola, waistline, and groin may all be involved, creating an imaginary circle between the areas described as the “circle of Hebra.” Penile and scrotal lesions are common in men.
Patients usually experience worse pruritus at night, which disturbs sleep. Crusted scabies is a severe form of scabies more often seen in those with immunocompromised immune systems. Clinically, thick crusted and scaly patches are present that are teeming with mites.
Diagnosis can be confirmed by performing a scabies prep, during which a burrow is scraped with a surgical blade. A drop of mineral oil is placed on the skin cells. The mite, ova, and feces can be visualized under the microscope. Wrists and hands usually have the highest yield for finding the parasites.
Topical treatments include permethrin 5% cream, lindane, benzyl benzoate, and crotamiton, and should be applied as two treatments a week apart. In the United States, permethrin is most commonly used. Ivermectin pills are used off label and are very effective and may be repeated for 1-2 weeks. All household contacts should be treated. Patients may still have pruritus for 2-4 weeks following treatment.
In this patient, a scabies prep was performed prior to performing repeat skin biopsies. Microscopic examination revealed ova, one mite, and feces. Treatment was initiated with ivermectin and permethrin.
Photos and case were submitted by Susannah Berke, MD, and Damon McClain, MD, Three Rivers Dermatology, Coraopolis, Pa.; and Dr. Bilu Martin.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to [email protected].
Sometimes You Can’t Blame the Sun
ANSWER
The correct answer is all of the above (choice “e”).
DISCUSSION
Most cases of dermatomyositis, which the patient’s presentation and lab results suggested, require nerve conduction studies, a check of serum aldolase levels, and skin and muscle biopsies to complete the work-up. However, the arrival at a diagnosis is only the first step.
Patients with dermatomyositis, particularly those older than 60, require evaluation for occult malignancy. There is evidence that the body’s immune response to the cancer is what drives the disease process. Hence the need for the studies listed, looking for breast, lung, and gastrointestinal cancers especially.
Dermatomyositis is thought to be an inflammatory myopathy, possibly driven by autoimmune factors. It is rare (about 1 to 22 per 100,000) and affects women more than men.
The “sunburn” rash is typical, especially on the face, chest, and dorsal hands, and usually clears completely when the cancer is found and cured. Other common findings include elevated creatine kinase, hand rashes (known as Gottron’s papules), and dystrophic calcification in skin and/or joints.
TREATMENT
Aside from addressing a possible malignancy, treatment of dermatomyositis usually starts with glucocorticoids, eventually tapered and replaced by steroid-sparing agents such as azathioprine or cyclosporine. These drugs have dramatically increased the chances of survival and eventual cure.
It’s common for the photosensitivity to persist long after the myositis has resolved, which is why sunscreen and other sun-protective measures are advised.
ANSWER
The correct answer is all of the above (choice “e”).
DISCUSSION
Most cases of dermatomyositis, which the patient’s presentation and lab results suggested, require nerve conduction studies, a check of serum aldolase levels, and skin and muscle biopsies to complete the work-up. However, the arrival at a diagnosis is only the first step.
Patients with dermatomyositis, particularly those older than 60, require evaluation for occult malignancy. There is evidence that the body’s immune response to the cancer is what drives the disease process. Hence the need for the studies listed, looking for breast, lung, and gastrointestinal cancers especially.
Dermatomyositis is thought to be an inflammatory myopathy, possibly driven by autoimmune factors. It is rare (about 1 to 22 per 100,000) and affects women more than men.
The “sunburn” rash is typical, especially on the face, chest, and dorsal hands, and usually clears completely when the cancer is found and cured. Other common findings include elevated creatine kinase, hand rashes (known as Gottron’s papules), and dystrophic calcification in skin and/or joints.
TREATMENT
Aside from addressing a possible malignancy, treatment of dermatomyositis usually starts with glucocorticoids, eventually tapered and replaced by steroid-sparing agents such as azathioprine or cyclosporine. These drugs have dramatically increased the chances of survival and eventual cure.
It’s common for the photosensitivity to persist long after the myositis has resolved, which is why sunscreen and other sun-protective measures are advised.
ANSWER
The correct answer is all of the above (choice “e”).
DISCUSSION
Most cases of dermatomyositis, which the patient’s presentation and lab results suggested, require nerve conduction studies, a check of serum aldolase levels, and skin and muscle biopsies to complete the work-up. However, the arrival at a diagnosis is only the first step.
Patients with dermatomyositis, particularly those older than 60, require evaluation for occult malignancy. There is evidence that the body’s immune response to the cancer is what drives the disease process. Hence the need for the studies listed, looking for breast, lung, and gastrointestinal cancers especially.
Dermatomyositis is thought to be an inflammatory myopathy, possibly driven by autoimmune factors. It is rare (about 1 to 22 per 100,000) and affects women more than men.
The “sunburn” rash is typical, especially on the face, chest, and dorsal hands, and usually clears completely when the cancer is found and cured. Other common findings include elevated creatine kinase, hand rashes (known as Gottron’s papules), and dystrophic calcification in skin and/or joints.
TREATMENT
Aside from addressing a possible malignancy, treatment of dermatomyositis usually starts with glucocorticoids, eventually tapered and replaced by steroid-sparing agents such as azathioprine or cyclosporine. These drugs have dramatically increased the chances of survival and eventual cure.
It’s common for the photosensitivity to persist long after the myositis has resolved, which is why sunscreen and other sun-protective measures are advised.
A 60-year-old woman was referred to dermatology for evaluation of “sunburn.” The rash was painful and unrelieved by topical medications, including class IV steroid creams. The redness was tender and warm to touch.
The rash had been present for months. During this period, the patient also had grown increasingly weak, leading her to quit her job. In the clinic, she was unable to stand from a seated position without difficulty. She reported no other health concerns and had quit smoking 5 years previously, after 30 years.
On examination, diffuse blanchable macular erythema on the patient’s face and chest was immediately observed. There was also an odd rash, composed of hundreds of tiny confluent papules, concentrated over the interphalangeal joints and dorsal hands. These too were warm and tender to touch. Most of her cuticles were peeling off; closer examination under magnification revealed tortuous capillaries on the distal cuticles of several fingers.
Bloodwork revealed a creatine kinase level slightly greater than 1000 U/L, and a positive antinuclear antibody, dilution unknown.