User login
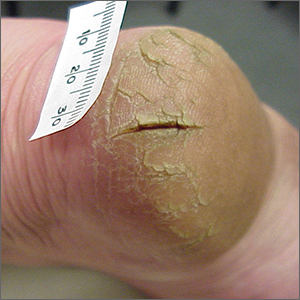
Painful heels
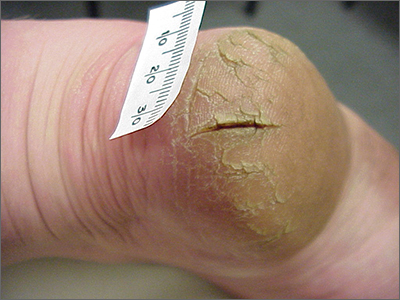
This patient was given a diagnosis of xerosis of the feet, commonly called fissured or cracked heels. Scaling and fissuring are also common in tinea pedis, but the location is often between the toes and there are finer splits and scale.
Xerosis is severely dry skin with hyperkeratosis due to abnormal keratinization;1 it leads to inflexibility and subsequent fissuring of the heel pads. The cracks can be painful and even bleed.
Although the condition is common, well-controlled trials and definitive evidence in the literature are sparse. The authors of one systematic review were unable to draw conclusions regarding the efficacy of various treatments due to wide variation in research methodologies and outcome measures; they did, however, note that urea-containing products (followed by ammonium lactate products) were studied the most.2
In clinical practice, frequently applied topical emollients are recommended. Exfoliating products, including prescription Lac-Hydrin (ammonium lactate 12% cream) and the over-the-counter version, Am-Lactin, may be helpful. Mechanical debridement with a file or pumice stone can be used (with caution) to reduce the hyperkeratotic plaques. If these measures fail, topical steroids may be added to the emollients. In addition, patients have used cyanoacrylate glues to hold the fissures together with a reported reduction in pain.3
This patient had already tried standard topical emollients. She was prescribed ammonium lactate cream to be used as an exfoliating moisturizer topically twice daily along with triamcinolone acetonide (TAC) 0.1% ointment to be applied twice daily. She was instructed to wean off the TAC once the xerosis was controlled with the ammonium lactate cream.
Photo and text courtesy of Daniel Stulberg, MD, FAAFP, Professor and Chair, Department of Family and Community Medicine, Western Michigan University Homer Stryker, MD School of Medicine, Kalamazoo.
1. Mazereeuw J, Bonafé JL. La xérose [Xerosis]. Ann Dermatol Venereol. 2002;129(1 Pt 2):137-142
2. Parker J, Scharfbillig R, Jones S. Moisturisers for the treatment of foot xerosis: a systematic review. J Foot Ankle Res. 2017;10:9. doi: 10.1186/s13047-017-0190-9
3. Hashimoto H. Superglue for the treatment of heel fissures. J Am Podiatr Med Assoc. 1999;89:434-435. doi: 10.7547/87507315-89-8-434

This patient was given a diagnosis of xerosis of the feet, commonly called fissured or cracked heels. Scaling and fissuring are also common in tinea pedis, but the location is often between the toes and there are finer splits and scale.
Xerosis is severely dry skin with hyperkeratosis due to abnormal keratinization;1 it leads to inflexibility and subsequent fissuring of the heel pads. The cracks can be painful and even bleed.
Although the condition is common, well-controlled trials and definitive evidence in the literature are sparse. The authors of one systematic review were unable to draw conclusions regarding the efficacy of various treatments due to wide variation in research methodologies and outcome measures; they did, however, note that urea-containing products (followed by ammonium lactate products) were studied the most.2
In clinical practice, frequently applied topical emollients are recommended. Exfoliating products, including prescription Lac-Hydrin (ammonium lactate 12% cream) and the over-the-counter version, Am-Lactin, may be helpful. Mechanical debridement with a file or pumice stone can be used (with caution) to reduce the hyperkeratotic plaques. If these measures fail, topical steroids may be added to the emollients. In addition, patients have used cyanoacrylate glues to hold the fissures together with a reported reduction in pain.3
This patient had already tried standard topical emollients. She was prescribed ammonium lactate cream to be used as an exfoliating moisturizer topically twice daily along with triamcinolone acetonide (TAC) 0.1% ointment to be applied twice daily. She was instructed to wean off the TAC once the xerosis was controlled with the ammonium lactate cream.
Photo and text courtesy of Daniel Stulberg, MD, FAAFP, Professor and Chair, Department of Family and Community Medicine, Western Michigan University Homer Stryker, MD School of Medicine, Kalamazoo.

This patient was given a diagnosis of xerosis of the feet, commonly called fissured or cracked heels. Scaling and fissuring are also common in tinea pedis, but the location is often between the toes and there are finer splits and scale.
Xerosis is severely dry skin with hyperkeratosis due to abnormal keratinization;1 it leads to inflexibility and subsequent fissuring of the heel pads. The cracks can be painful and even bleed.
Although the condition is common, well-controlled trials and definitive evidence in the literature are sparse. The authors of one systematic review were unable to draw conclusions regarding the efficacy of various treatments due to wide variation in research methodologies and outcome measures; they did, however, note that urea-containing products (followed by ammonium lactate products) were studied the most.2
In clinical practice, frequently applied topical emollients are recommended. Exfoliating products, including prescription Lac-Hydrin (ammonium lactate 12% cream) and the over-the-counter version, Am-Lactin, may be helpful. Mechanical debridement with a file or pumice stone can be used (with caution) to reduce the hyperkeratotic plaques. If these measures fail, topical steroids may be added to the emollients. In addition, patients have used cyanoacrylate glues to hold the fissures together with a reported reduction in pain.3
This patient had already tried standard topical emollients. She was prescribed ammonium lactate cream to be used as an exfoliating moisturizer topically twice daily along with triamcinolone acetonide (TAC) 0.1% ointment to be applied twice daily. She was instructed to wean off the TAC once the xerosis was controlled with the ammonium lactate cream.
Photo and text courtesy of Daniel Stulberg, MD, FAAFP, Professor and Chair, Department of Family and Community Medicine, Western Michigan University Homer Stryker, MD School of Medicine, Kalamazoo.
1. Mazereeuw J, Bonafé JL. La xérose [Xerosis]. Ann Dermatol Venereol. 2002;129(1 Pt 2):137-142
2. Parker J, Scharfbillig R, Jones S. Moisturisers for the treatment of foot xerosis: a systematic review. J Foot Ankle Res. 2017;10:9. doi: 10.1186/s13047-017-0190-9
3. Hashimoto H. Superglue for the treatment of heel fissures. J Am Podiatr Med Assoc. 1999;89:434-435. doi: 10.7547/87507315-89-8-434
1. Mazereeuw J, Bonafé JL. La xérose [Xerosis]. Ann Dermatol Venereol. 2002;129(1 Pt 2):137-142
2. Parker J, Scharfbillig R, Jones S. Moisturisers for the treatment of foot xerosis: a systematic review. J Foot Ankle Res. 2017;10:9. doi: 10.1186/s13047-017-0190-9
3. Hashimoto H. Superglue for the treatment of heel fissures. J Am Podiatr Med Assoc. 1999;89:434-435. doi: 10.7547/87507315-89-8-434
Atopic dermatitis: Five things to know
Atopic dermatitis (AD) is a chronic, pruritic inflammatory skin condition that typically affects the face (cheeks), neck, arms, and legs but usually spares the groin and axillary regions. AD usually starts in early infancy but also affects some adults. AD is often associated with elevated levels of immunoglobulin E (IgE). That it is the first disease to present in a series of allergic diseases – including food allergy, asthma, and allergic rhinitis, in order – and has given rise to the “atopic march” theory, which suggests that AD is part of a progression that may lead to subsequent allergic disease at other epithelial barrier surfaces.
.
1. Essential features of AD are pruritus and eczema
The diagnosis of AD is primarily observational. It is made on the basis of patient and family history, pattern of lesions, morphology, and clinical signs. No genetic features or biomarkers are specific enough to reliably aid in diagnosis or severity assessment. Many individual findings are used to diagnose AD, as summarized by the American Academy of Dermatology based on essential, important, associated, and exclusionary features:
- Essential features (must be present for diagnosis) are pruritus and eczema (acute, subacute, or chronic) with typical morphology and age-specific patterns and chronic or relapsing history.
- Important features (usually seen in AD and support the diagnosis) are early age of onset, atopy (personal/family history, IgE reactivity), and xerosis.
- Associated features (nonspecific but suggestive) are atypical vascular response (e.g., delayed blanch response); keratosis pilaris (and some others); ocular/periorbital changes; other regional findings (e.g., perioral changes); and perifollicular accentuation, lichenification, or prurigo lesions.
- Exclusionary conditions (must be excluded to make the AD diagnosis) are scabies, seborrheic dermatitis, contact dermatitis, ichthyoses, cutaneous T-cell lymphoma, psoriasis, photosensitivity dermatoses, immune deficiency diseases, and erythroderma due to other causes.
AD should be differentiated from other red, scaly skin conditions. It is often difficult to separate AD from seborrheic dermatitis in infancy, and the two conditions may overlap in this age group. Particularly if the condition is not responding to therapy, the diagnosis of AD should be re-reviewed and other disorders considered, including more serious nutritional, metabolic, and immunologic conditions in children and cutaneous T-cell lymphoma in adults. Allergic contact dermatitis may be both an alternative diagnosis to AD and an exacerbator of AD in some individuals.
2. Associated comorbidities of AD may exacerbate the condition and lead to other atopic disorders
Reported comorbidities of AD include other atopic or allergic conditions, autoimmune diseases, infections, metabolic conditions, mental health disorders, and cardiovascular disease. Certain aspects of AD, such as chronic pruritus, psychosocial distress, and inflammation, can lead to anxiety, depression, and suicidality. AD is associated with and may predispose to higher risk for other atopic disorders, including asthma, hay fever, food allergy, and eosinophilic esophagitis.
Persons with AD also appear to be at higher risk for infectious diseases. The prevalence of cutaneous and systemic infections in patients with AD is significantly higher than those without AD. Infectious complications can include skin and soft-tissue infections, bacteremia, eczema herpeticum, osteomyelitis, endocarditis, and septic arthritis.
3. Climate change has a profound impact on AD
The incidence of AD has increased over the past several decades, and environmental factors such as climate change have been implicated as a potential mechanism. Climate change–related factors affect the skin’s capacity to maintain homeostasis, leading to various cutaneous diseases. AD, psoriasis, pemphigus, acne vulgaris, melasma, and photoaging are all associated with rising levels of air pollution. Elevated temperatures due to global warming induce disruption of the skin microbiome, thereby affecting AD.
Extreme weather events due to climate change, including floods and wildfires, are implicated in cutaneous injuries, skin infections, and acute worsening of inflammatory skin disorders.
4. The impact and appearance of AD varies in different racial groups
It was once believed that AD was just one single disease affecting people of many different races. More recently, it has been proposed that AD is in fact a group of different diseases. Both epidemiologic and genetic factors may play a role in influencing the main features of AD.
Spongiotic processes such as AD that would be pink or erythematous on white skin are often hypopigmented in individuals with darkly pigmented skin. AD has a higher prevalence and severity in Black and mixed-race populations, probably owing to a combination of environmental and intrinsic factors. Black skin has been shown to have increased transepidermal water loss and lower levels of ceramides, which are important components of the lipid barrier in the stratum corneum.
The American College of Allergy, Asthma & Immunology, along with the Allergy & Asthma Network, are partnering to create Eczema in Skin of Color, a website to aid physicians and patients in recognizing eczema in people with all skin types.
5. New and emerging therapies are poised to improve outcomes with AD treatment
Ruxolitinib cream, a topical Janus kinase (JAK)-1/JAK2 inhibitor, was approved for AD by the U.S. Food and Drug Administration in September 2021. The approval was based on results from the Topical Ruxolitinib Evaluation in AD (TRuE-AD) clinical trial program, which consisted of phase 3 studies that investigated 1,249 patients aged greater than or equal to 12 years with mild to moderate AD (Investigator’s Global Assessment score of 2-3) with a body surface area of 3%-20% (excluding scalp). The 2023 AAD guidelines for topical treatment recommend ruxolitinib cream for adults with mild to moderate AD.
Tralokinumab is a monoclonal antibody that inhibits the interleukin-13 cytokines, which prevents the release of cytokines, chemokines, and IgE. It was approved by the FDA in 2021 for treatment of moderate to severe AD. It is administered by subcutaneous injection every 2 weeks. Approval was based on the phase 3 trials ECZTRA 1, 2, and 3, which assessed the efficacy of tralokinumab in 1,934 adults.
Abrocitinib is an oral, once-daily JAK1 inhibitor for treatment of adults living with refractory, moderate to severe AD. FDA approval was based on results of five clinical trials from a large-scale trial program of more than 1,600 patients. Across the trials, abrocitinib demonstrated a consistent safety profile and profound improvements in skin clearance, extent of disease, and severity, as well as rapid improvement in itch after 2 weeks, for some people living with AD vs placebo.
Upadacitinib, another oral JAK1 inhibitor, was approved by the FDA in January 2022 for refractory moderate to severe AD. Approval was based on three double-blind phase 3 trials (Measure Up 1, Measure Up 2, AD Up) in which 2,584 patients with moderate to severe AD were randomized to receive oral upadacitinib 15 mg/d and 30 mg/d. In Measure Up 1 and Measure Up 2, upadacitinib was evaluated as monotherapy; in AD Up, upadacitinib was evaluated in combination with topical corticosteroids.
On the horizon
Baricitinib, an oral JAK1/2 inhibitor, is not yet approved by the FDA for AD. It is, however, approved for moderate to severe AD treatment in the European Union and many other countries. A 2022 review of studies evaluating baricitinib for the treatment of moderate to severe AD in adults (BREEZE-AD1, -AD2, -AD3, -AD4, -AD5, -AD6) reported that current evidence supports baricitinib, used as monotherapy or in combination with topical corticosteroids, as a safe and effective agent that can be used as an alternative to subcutaneous biologics in adults with moderate to severe AD.
Topical JAK inhibitors
A 2023 systematic review (19 studies, 3,600 participants) reported on several topical JAK inhibitors that are effective for treating AD. It suggests a stronger safety profile and better results, compared with systemic JAK inhibitors. The review focused on topical delgocitinib, tofacitinib, ruxolitinib, cerdulatinib, and ifidancitinib. All agents were effective in treating AD. All of these topical JAK inhibitors had minimal risk for mild to moderate adverse effects.
Biologics
Lebrikizumab was evaluated in a phase 2b, double-blind, placebo-controlled randomized clinical trial. After 16 weeks (280 participants), patients with moderate to severe AD showed a dose-dependent significant improvement in the primary endpoint, compared with placebo. Two phase 3 trials (ADvocate1, ADvocate2) evaluated the safety and efficacy of monotherapy with lebrikizumab in adults and adolescents with moderate to severe AD.
Nemolizumab, assessed in long-term phase 3 trials of AD-associated pruritus, resulted in clinically meaningful improvements from the beginning of treatment to week 68. Nemolizumab is being evaluated in two identical phase 3 studies (Arcadia 1, Arcadia 2) and a long-term extension study.
Dr. Kim is Professor and Vice Chair of Research in the department of dermatology, as well as Director of the Mark Lebwohl Center for Neuroinflammation and Sensation at the Icahn School of Medicine at Mount Sinai, New York. He reported conflicts of interest with 23andMe, Abrax Japan, AbbVie, Almirall, Amgen, and KiiRNA Biotech.
A version of this article first appeared on Medscape.com.
Atopic dermatitis (AD) is a chronic, pruritic inflammatory skin condition that typically affects the face (cheeks), neck, arms, and legs but usually spares the groin and axillary regions. AD usually starts in early infancy but also affects some adults. AD is often associated with elevated levels of immunoglobulin E (IgE). That it is the first disease to present in a series of allergic diseases – including food allergy, asthma, and allergic rhinitis, in order – and has given rise to the “atopic march” theory, which suggests that AD is part of a progression that may lead to subsequent allergic disease at other epithelial barrier surfaces.
.
1. Essential features of AD are pruritus and eczema
The diagnosis of AD is primarily observational. It is made on the basis of patient and family history, pattern of lesions, morphology, and clinical signs. No genetic features or biomarkers are specific enough to reliably aid in diagnosis or severity assessment. Many individual findings are used to diagnose AD, as summarized by the American Academy of Dermatology based on essential, important, associated, and exclusionary features:
- Essential features (must be present for diagnosis) are pruritus and eczema (acute, subacute, or chronic) with typical morphology and age-specific patterns and chronic or relapsing history.
- Important features (usually seen in AD and support the diagnosis) are early age of onset, atopy (personal/family history, IgE reactivity), and xerosis.
- Associated features (nonspecific but suggestive) are atypical vascular response (e.g., delayed blanch response); keratosis pilaris (and some others); ocular/periorbital changes; other regional findings (e.g., perioral changes); and perifollicular accentuation, lichenification, or prurigo lesions.
- Exclusionary conditions (must be excluded to make the AD diagnosis) are scabies, seborrheic dermatitis, contact dermatitis, ichthyoses, cutaneous T-cell lymphoma, psoriasis, photosensitivity dermatoses, immune deficiency diseases, and erythroderma due to other causes.
AD should be differentiated from other red, scaly skin conditions. It is often difficult to separate AD from seborrheic dermatitis in infancy, and the two conditions may overlap in this age group. Particularly if the condition is not responding to therapy, the diagnosis of AD should be re-reviewed and other disorders considered, including more serious nutritional, metabolic, and immunologic conditions in children and cutaneous T-cell lymphoma in adults. Allergic contact dermatitis may be both an alternative diagnosis to AD and an exacerbator of AD in some individuals.
2. Associated comorbidities of AD may exacerbate the condition and lead to other atopic disorders
Reported comorbidities of AD include other atopic or allergic conditions, autoimmune diseases, infections, metabolic conditions, mental health disorders, and cardiovascular disease. Certain aspects of AD, such as chronic pruritus, psychosocial distress, and inflammation, can lead to anxiety, depression, and suicidality. AD is associated with and may predispose to higher risk for other atopic disorders, including asthma, hay fever, food allergy, and eosinophilic esophagitis.
Persons with AD also appear to be at higher risk for infectious diseases. The prevalence of cutaneous and systemic infections in patients with AD is significantly higher than those without AD. Infectious complications can include skin and soft-tissue infections, bacteremia, eczema herpeticum, osteomyelitis, endocarditis, and septic arthritis.
3. Climate change has a profound impact on AD
The incidence of AD has increased over the past several decades, and environmental factors such as climate change have been implicated as a potential mechanism. Climate change–related factors affect the skin’s capacity to maintain homeostasis, leading to various cutaneous diseases. AD, psoriasis, pemphigus, acne vulgaris, melasma, and photoaging are all associated with rising levels of air pollution. Elevated temperatures due to global warming induce disruption of the skin microbiome, thereby affecting AD.
Extreme weather events due to climate change, including floods and wildfires, are implicated in cutaneous injuries, skin infections, and acute worsening of inflammatory skin disorders.
4. The impact and appearance of AD varies in different racial groups
It was once believed that AD was just one single disease affecting people of many different races. More recently, it has been proposed that AD is in fact a group of different diseases. Both epidemiologic and genetic factors may play a role in influencing the main features of AD.
Spongiotic processes such as AD that would be pink or erythematous on white skin are often hypopigmented in individuals with darkly pigmented skin. AD has a higher prevalence and severity in Black and mixed-race populations, probably owing to a combination of environmental and intrinsic factors. Black skin has been shown to have increased transepidermal water loss and lower levels of ceramides, which are important components of the lipid barrier in the stratum corneum.
The American College of Allergy, Asthma & Immunology, along with the Allergy & Asthma Network, are partnering to create Eczema in Skin of Color, a website to aid physicians and patients in recognizing eczema in people with all skin types.
5. New and emerging therapies are poised to improve outcomes with AD treatment
Ruxolitinib cream, a topical Janus kinase (JAK)-1/JAK2 inhibitor, was approved for AD by the U.S. Food and Drug Administration in September 2021. The approval was based on results from the Topical Ruxolitinib Evaluation in AD (TRuE-AD) clinical trial program, which consisted of phase 3 studies that investigated 1,249 patients aged greater than or equal to 12 years with mild to moderate AD (Investigator’s Global Assessment score of 2-3) with a body surface area of 3%-20% (excluding scalp). The 2023 AAD guidelines for topical treatment recommend ruxolitinib cream for adults with mild to moderate AD.
Tralokinumab is a monoclonal antibody that inhibits the interleukin-13 cytokines, which prevents the release of cytokines, chemokines, and IgE. It was approved by the FDA in 2021 for treatment of moderate to severe AD. It is administered by subcutaneous injection every 2 weeks. Approval was based on the phase 3 trials ECZTRA 1, 2, and 3, which assessed the efficacy of tralokinumab in 1,934 adults.
Abrocitinib is an oral, once-daily JAK1 inhibitor for treatment of adults living with refractory, moderate to severe AD. FDA approval was based on results of five clinical trials from a large-scale trial program of more than 1,600 patients. Across the trials, abrocitinib demonstrated a consistent safety profile and profound improvements in skin clearance, extent of disease, and severity, as well as rapid improvement in itch after 2 weeks, for some people living with AD vs placebo.
Upadacitinib, another oral JAK1 inhibitor, was approved by the FDA in January 2022 for refractory moderate to severe AD. Approval was based on three double-blind phase 3 trials (Measure Up 1, Measure Up 2, AD Up) in which 2,584 patients with moderate to severe AD were randomized to receive oral upadacitinib 15 mg/d and 30 mg/d. In Measure Up 1 and Measure Up 2, upadacitinib was evaluated as monotherapy; in AD Up, upadacitinib was evaluated in combination with topical corticosteroids.
On the horizon
Baricitinib, an oral JAK1/2 inhibitor, is not yet approved by the FDA for AD. It is, however, approved for moderate to severe AD treatment in the European Union and many other countries. A 2022 review of studies evaluating baricitinib for the treatment of moderate to severe AD in adults (BREEZE-AD1, -AD2, -AD3, -AD4, -AD5, -AD6) reported that current evidence supports baricitinib, used as monotherapy or in combination with topical corticosteroids, as a safe and effective agent that can be used as an alternative to subcutaneous biologics in adults with moderate to severe AD.
Topical JAK inhibitors
A 2023 systematic review (19 studies, 3,600 participants) reported on several topical JAK inhibitors that are effective for treating AD. It suggests a stronger safety profile and better results, compared with systemic JAK inhibitors. The review focused on topical delgocitinib, tofacitinib, ruxolitinib, cerdulatinib, and ifidancitinib. All agents were effective in treating AD. All of these topical JAK inhibitors had minimal risk for mild to moderate adverse effects.
Biologics
Lebrikizumab was evaluated in a phase 2b, double-blind, placebo-controlled randomized clinical trial. After 16 weeks (280 participants), patients with moderate to severe AD showed a dose-dependent significant improvement in the primary endpoint, compared with placebo. Two phase 3 trials (ADvocate1, ADvocate2) evaluated the safety and efficacy of monotherapy with lebrikizumab in adults and adolescents with moderate to severe AD.
Nemolizumab, assessed in long-term phase 3 trials of AD-associated pruritus, resulted in clinically meaningful improvements from the beginning of treatment to week 68. Nemolizumab is being evaluated in two identical phase 3 studies (Arcadia 1, Arcadia 2) and a long-term extension study.
Dr. Kim is Professor and Vice Chair of Research in the department of dermatology, as well as Director of the Mark Lebwohl Center for Neuroinflammation and Sensation at the Icahn School of Medicine at Mount Sinai, New York. He reported conflicts of interest with 23andMe, Abrax Japan, AbbVie, Almirall, Amgen, and KiiRNA Biotech.
A version of this article first appeared on Medscape.com.
Atopic dermatitis (AD) is a chronic, pruritic inflammatory skin condition that typically affects the face (cheeks), neck, arms, and legs but usually spares the groin and axillary regions. AD usually starts in early infancy but also affects some adults. AD is often associated with elevated levels of immunoglobulin E (IgE). That it is the first disease to present in a series of allergic diseases – including food allergy, asthma, and allergic rhinitis, in order – and has given rise to the “atopic march” theory, which suggests that AD is part of a progression that may lead to subsequent allergic disease at other epithelial barrier surfaces.
.
1. Essential features of AD are pruritus and eczema
The diagnosis of AD is primarily observational. It is made on the basis of patient and family history, pattern of lesions, morphology, and clinical signs. No genetic features or biomarkers are specific enough to reliably aid in diagnosis or severity assessment. Many individual findings are used to diagnose AD, as summarized by the American Academy of Dermatology based on essential, important, associated, and exclusionary features:
- Essential features (must be present for diagnosis) are pruritus and eczema (acute, subacute, or chronic) with typical morphology and age-specific patterns and chronic or relapsing history.
- Important features (usually seen in AD and support the diagnosis) are early age of onset, atopy (personal/family history, IgE reactivity), and xerosis.
- Associated features (nonspecific but suggestive) are atypical vascular response (e.g., delayed blanch response); keratosis pilaris (and some others); ocular/periorbital changes; other regional findings (e.g., perioral changes); and perifollicular accentuation, lichenification, or prurigo lesions.
- Exclusionary conditions (must be excluded to make the AD diagnosis) are scabies, seborrheic dermatitis, contact dermatitis, ichthyoses, cutaneous T-cell lymphoma, psoriasis, photosensitivity dermatoses, immune deficiency diseases, and erythroderma due to other causes.
AD should be differentiated from other red, scaly skin conditions. It is often difficult to separate AD from seborrheic dermatitis in infancy, and the two conditions may overlap in this age group. Particularly if the condition is not responding to therapy, the diagnosis of AD should be re-reviewed and other disorders considered, including more serious nutritional, metabolic, and immunologic conditions in children and cutaneous T-cell lymphoma in adults. Allergic contact dermatitis may be both an alternative diagnosis to AD and an exacerbator of AD in some individuals.
2. Associated comorbidities of AD may exacerbate the condition and lead to other atopic disorders
Reported comorbidities of AD include other atopic or allergic conditions, autoimmune diseases, infections, metabolic conditions, mental health disorders, and cardiovascular disease. Certain aspects of AD, such as chronic pruritus, psychosocial distress, and inflammation, can lead to anxiety, depression, and suicidality. AD is associated with and may predispose to higher risk for other atopic disorders, including asthma, hay fever, food allergy, and eosinophilic esophagitis.
Persons with AD also appear to be at higher risk for infectious diseases. The prevalence of cutaneous and systemic infections in patients with AD is significantly higher than those without AD. Infectious complications can include skin and soft-tissue infections, bacteremia, eczema herpeticum, osteomyelitis, endocarditis, and septic arthritis.
3. Climate change has a profound impact on AD
The incidence of AD has increased over the past several decades, and environmental factors such as climate change have been implicated as a potential mechanism. Climate change–related factors affect the skin’s capacity to maintain homeostasis, leading to various cutaneous diseases. AD, psoriasis, pemphigus, acne vulgaris, melasma, and photoaging are all associated with rising levels of air pollution. Elevated temperatures due to global warming induce disruption of the skin microbiome, thereby affecting AD.
Extreme weather events due to climate change, including floods and wildfires, are implicated in cutaneous injuries, skin infections, and acute worsening of inflammatory skin disorders.
4. The impact and appearance of AD varies in different racial groups
It was once believed that AD was just one single disease affecting people of many different races. More recently, it has been proposed that AD is in fact a group of different diseases. Both epidemiologic and genetic factors may play a role in influencing the main features of AD.
Spongiotic processes such as AD that would be pink or erythematous on white skin are often hypopigmented in individuals with darkly pigmented skin. AD has a higher prevalence and severity in Black and mixed-race populations, probably owing to a combination of environmental and intrinsic factors. Black skin has been shown to have increased transepidermal water loss and lower levels of ceramides, which are important components of the lipid barrier in the stratum corneum.
The American College of Allergy, Asthma & Immunology, along with the Allergy & Asthma Network, are partnering to create Eczema in Skin of Color, a website to aid physicians and patients in recognizing eczema in people with all skin types.
5. New and emerging therapies are poised to improve outcomes with AD treatment
Ruxolitinib cream, a topical Janus kinase (JAK)-1/JAK2 inhibitor, was approved for AD by the U.S. Food and Drug Administration in September 2021. The approval was based on results from the Topical Ruxolitinib Evaluation in AD (TRuE-AD) clinical trial program, which consisted of phase 3 studies that investigated 1,249 patients aged greater than or equal to 12 years with mild to moderate AD (Investigator’s Global Assessment score of 2-3) with a body surface area of 3%-20% (excluding scalp). The 2023 AAD guidelines for topical treatment recommend ruxolitinib cream for adults with mild to moderate AD.
Tralokinumab is a monoclonal antibody that inhibits the interleukin-13 cytokines, which prevents the release of cytokines, chemokines, and IgE. It was approved by the FDA in 2021 for treatment of moderate to severe AD. It is administered by subcutaneous injection every 2 weeks. Approval was based on the phase 3 trials ECZTRA 1, 2, and 3, which assessed the efficacy of tralokinumab in 1,934 adults.
Abrocitinib is an oral, once-daily JAK1 inhibitor for treatment of adults living with refractory, moderate to severe AD. FDA approval was based on results of five clinical trials from a large-scale trial program of more than 1,600 patients. Across the trials, abrocitinib demonstrated a consistent safety profile and profound improvements in skin clearance, extent of disease, and severity, as well as rapid improvement in itch after 2 weeks, for some people living with AD vs placebo.
Upadacitinib, another oral JAK1 inhibitor, was approved by the FDA in January 2022 for refractory moderate to severe AD. Approval was based on three double-blind phase 3 trials (Measure Up 1, Measure Up 2, AD Up) in which 2,584 patients with moderate to severe AD were randomized to receive oral upadacitinib 15 mg/d and 30 mg/d. In Measure Up 1 and Measure Up 2, upadacitinib was evaluated as monotherapy; in AD Up, upadacitinib was evaluated in combination with topical corticosteroids.
On the horizon
Baricitinib, an oral JAK1/2 inhibitor, is not yet approved by the FDA for AD. It is, however, approved for moderate to severe AD treatment in the European Union and many other countries. A 2022 review of studies evaluating baricitinib for the treatment of moderate to severe AD in adults (BREEZE-AD1, -AD2, -AD3, -AD4, -AD5, -AD6) reported that current evidence supports baricitinib, used as monotherapy or in combination with topical corticosteroids, as a safe and effective agent that can be used as an alternative to subcutaneous biologics in adults with moderate to severe AD.
Topical JAK inhibitors
A 2023 systematic review (19 studies, 3,600 participants) reported on several topical JAK inhibitors that are effective for treating AD. It suggests a stronger safety profile and better results, compared with systemic JAK inhibitors. The review focused on topical delgocitinib, tofacitinib, ruxolitinib, cerdulatinib, and ifidancitinib. All agents were effective in treating AD. All of these topical JAK inhibitors had minimal risk for mild to moderate adverse effects.
Biologics
Lebrikizumab was evaluated in a phase 2b, double-blind, placebo-controlled randomized clinical trial. After 16 weeks (280 participants), patients with moderate to severe AD showed a dose-dependent significant improvement in the primary endpoint, compared with placebo. Two phase 3 trials (ADvocate1, ADvocate2) evaluated the safety and efficacy of monotherapy with lebrikizumab in adults and adolescents with moderate to severe AD.
Nemolizumab, assessed in long-term phase 3 trials of AD-associated pruritus, resulted in clinically meaningful improvements from the beginning of treatment to week 68. Nemolizumab is being evaluated in two identical phase 3 studies (Arcadia 1, Arcadia 2) and a long-term extension study.
Dr. Kim is Professor and Vice Chair of Research in the department of dermatology, as well as Director of the Mark Lebwohl Center for Neuroinflammation and Sensation at the Icahn School of Medicine at Mount Sinai, New York. He reported conflicts of interest with 23andMe, Abrax Japan, AbbVie, Almirall, Amgen, and KiiRNA Biotech.
A version of this article first appeared on Medscape.com.
3-D stereophotogrammetry helps detect progression of craniofacial morphea
TOPLINE:
over time.
METHODOLOGY:
- Existing tools that detect disease progression in patients with CM are limited.
- In a prospective cohort study, researchers evaluated the use of 3-D stereophotogrammetry, a noninvasive, radiation-free imaging modality, to detect disease progression in 27 consecutive patients with CM seen at Boston Children’s Hospital and Brigham and Women’s Hospital from April 1, 2019, to March 1, 2023.
- After clinical and 3-D stereophotogrammetry assessments were performed at 2- to 12-month intervals, the 3-D images were rated by an expert (a board-certified plastic craniofacial surgeon) and a nonexpert (a board-certified dermatologist) as demonstrating progression or no progression.
- Kappa coefficients were used to calculate inter-rater reliability.
TAKEAWAY:
- Most of the study participants (73%) were female, their median age was 14 years (range, 5-40 years), and each underwent 3-D stereophotogrammetry imaging at least two times spaced a median of 3 months apart.
- On the basis of clinical assessments during the 48-month study period, 10 patients (37%) experienced progression of their disease.
- 3-D stereophotogrammetry not only corroborated clinical impressions of disease progression with strong inter-rater reliability (kappa = 0.80; 95% confidence interval, 0.61-0.99), but it also detected occult progression of asymmetry not noted on clinical examination in three additional patients.
- In subgroup analyses, assessment of 3-D images demonstrated substantial to near-perfect inter-rater reliability in patients with Fitzpatrick skin types IV-VI.
IN PRACTICE:
“Further work is necessary to validate this measure in a larger cohort and to guide its incorporation into medical decision-making for patients with CM,” the researchers wrote.
SOURCE:
Katharina S. Shaw, MD, of the department of dermatology at the University of Pennsylvania, Philadelphia, led the research. The study was published online in JAMA Dermatology.
LIMITATIONS:
The sample was small, and a criterion standard for assessing CM was lacking.
DISCLOSURES:
The researchers reported having no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
TOPLINE:
over time.
METHODOLOGY:
- Existing tools that detect disease progression in patients with CM are limited.
- In a prospective cohort study, researchers evaluated the use of 3-D stereophotogrammetry, a noninvasive, radiation-free imaging modality, to detect disease progression in 27 consecutive patients with CM seen at Boston Children’s Hospital and Brigham and Women’s Hospital from April 1, 2019, to March 1, 2023.
- After clinical and 3-D stereophotogrammetry assessments were performed at 2- to 12-month intervals, the 3-D images were rated by an expert (a board-certified plastic craniofacial surgeon) and a nonexpert (a board-certified dermatologist) as demonstrating progression or no progression.
- Kappa coefficients were used to calculate inter-rater reliability.
TAKEAWAY:
- Most of the study participants (73%) were female, their median age was 14 years (range, 5-40 years), and each underwent 3-D stereophotogrammetry imaging at least two times spaced a median of 3 months apart.
- On the basis of clinical assessments during the 48-month study period, 10 patients (37%) experienced progression of their disease.
- 3-D stereophotogrammetry not only corroborated clinical impressions of disease progression with strong inter-rater reliability (kappa = 0.80; 95% confidence interval, 0.61-0.99), but it also detected occult progression of asymmetry not noted on clinical examination in three additional patients.
- In subgroup analyses, assessment of 3-D images demonstrated substantial to near-perfect inter-rater reliability in patients with Fitzpatrick skin types IV-VI.
IN PRACTICE:
“Further work is necessary to validate this measure in a larger cohort and to guide its incorporation into medical decision-making for patients with CM,” the researchers wrote.
SOURCE:
Katharina S. Shaw, MD, of the department of dermatology at the University of Pennsylvania, Philadelphia, led the research. The study was published online in JAMA Dermatology.
LIMITATIONS:
The sample was small, and a criterion standard for assessing CM was lacking.
DISCLOSURES:
The researchers reported having no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
TOPLINE:
over time.
METHODOLOGY:
- Existing tools that detect disease progression in patients with CM are limited.
- In a prospective cohort study, researchers evaluated the use of 3-D stereophotogrammetry, a noninvasive, radiation-free imaging modality, to detect disease progression in 27 consecutive patients with CM seen at Boston Children’s Hospital and Brigham and Women’s Hospital from April 1, 2019, to March 1, 2023.
- After clinical and 3-D stereophotogrammetry assessments were performed at 2- to 12-month intervals, the 3-D images were rated by an expert (a board-certified plastic craniofacial surgeon) and a nonexpert (a board-certified dermatologist) as demonstrating progression or no progression.
- Kappa coefficients were used to calculate inter-rater reliability.
TAKEAWAY:
- Most of the study participants (73%) were female, their median age was 14 years (range, 5-40 years), and each underwent 3-D stereophotogrammetry imaging at least two times spaced a median of 3 months apart.
- On the basis of clinical assessments during the 48-month study period, 10 patients (37%) experienced progression of their disease.
- 3-D stereophotogrammetry not only corroborated clinical impressions of disease progression with strong inter-rater reliability (kappa = 0.80; 95% confidence interval, 0.61-0.99), but it also detected occult progression of asymmetry not noted on clinical examination in three additional patients.
- In subgroup analyses, assessment of 3-D images demonstrated substantial to near-perfect inter-rater reliability in patients with Fitzpatrick skin types IV-VI.
IN PRACTICE:
“Further work is necessary to validate this measure in a larger cohort and to guide its incorporation into medical decision-making for patients with CM,” the researchers wrote.
SOURCE:
Katharina S. Shaw, MD, of the department of dermatology at the University of Pennsylvania, Philadelphia, led the research. The study was published online in JAMA Dermatology.
LIMITATIONS:
The sample was small, and a criterion standard for assessing CM was lacking.
DISCLOSURES:
The researchers reported having no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
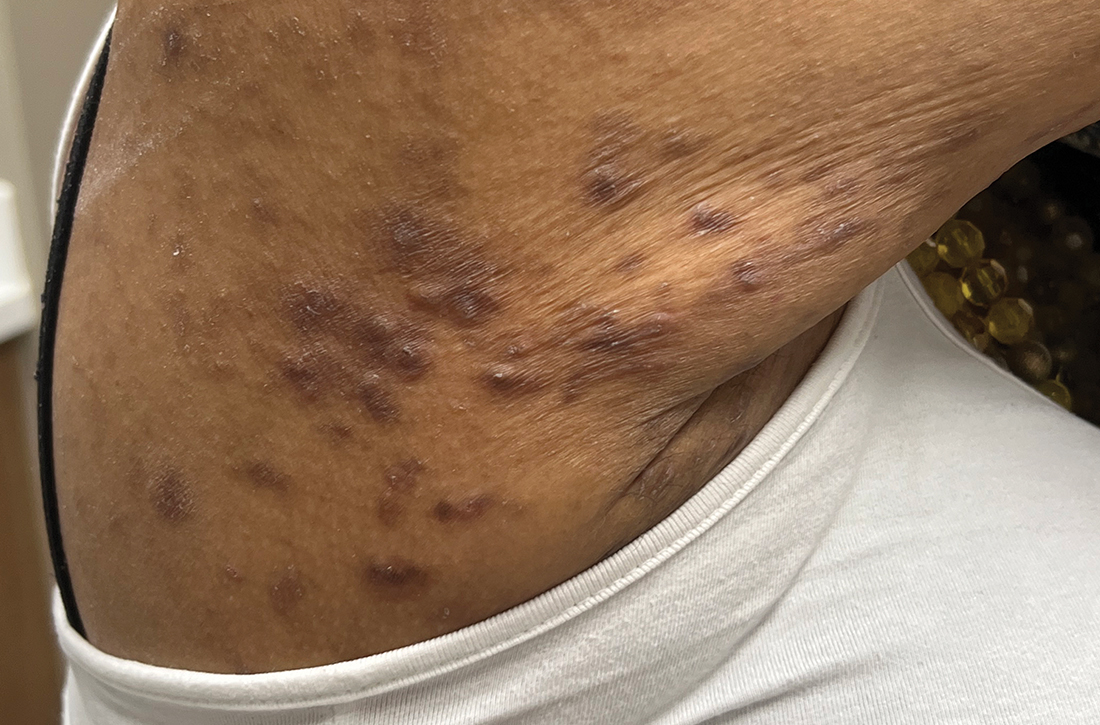
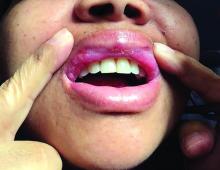
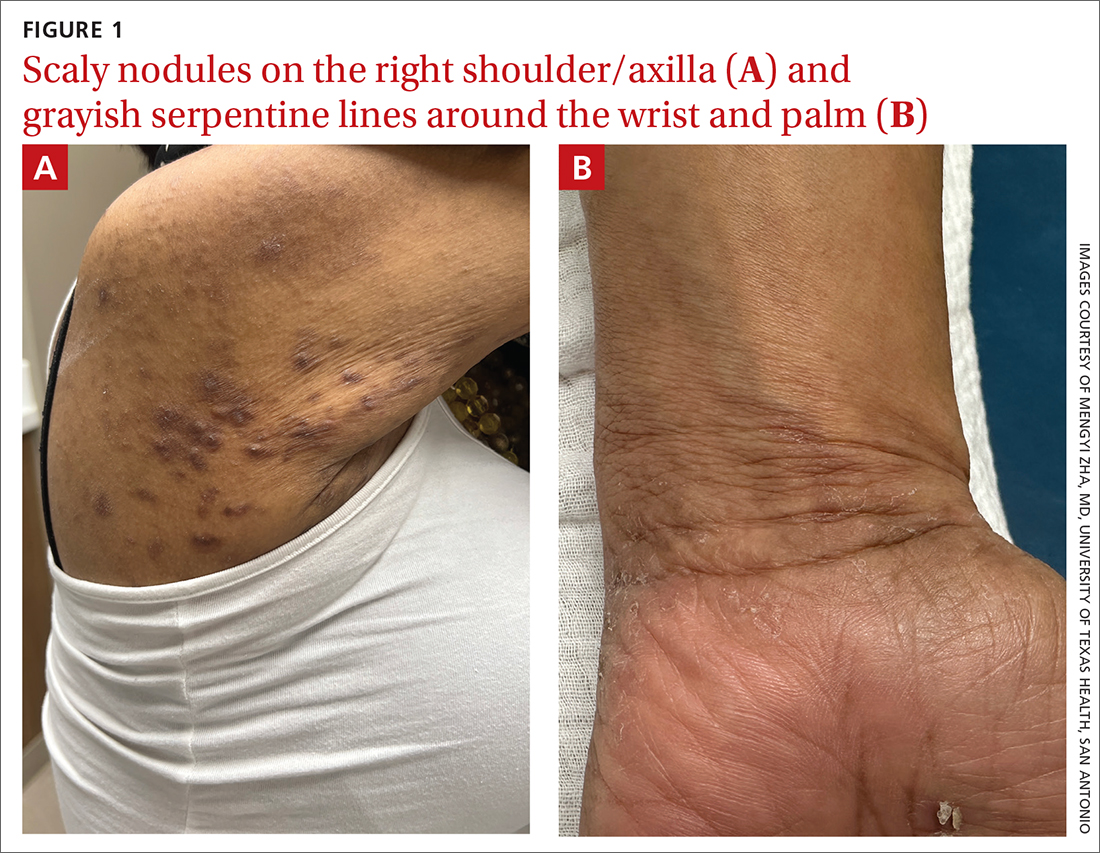
Pruritic rash and nocturnal itching
A 62-YEAR-OLD HISPANIC WOMAN with a history of well-controlled diabetes and hypertension presented with an intensely pruritic rash of 3 months’ duration. She reported poor sleep due to scratching throughout the night. She denied close contact with individuals with similar rashes or itching, new intimate partners, or recent travel. She worked in an office setting and had stable, noncrowded housing.
A physical exam revealed brown and purple scaly papules and many excoriation marks. The rash was concentrated along clothing lines, around intertriginous areas, and on her ankles, wrists, and the interdigital spaces (FIGURE 1A and 1B).
WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Diagnosis: Scabies
Scabies is a diagnosis that should be considered in any patient with new-onset, widespread, nocturnal-dominant pruritus1 and it was suspected, in this case, after the initial history taking and physical exam. (See “Consider these diagnoses in cases of pruritic skin conditions” for more on lichen planus and prurigo nodularis, which were also included in the differential diagnosis.)
SIDEBAR
Consider these diagnoses in cases of pruritic skin conditions
Lichen planus is a chronic inflammatory condition that mostly affects the skin and mucosa. Characteristic findings are groups of shiny, flat-topped, firm papules. This patient’s widespread nodular lesions with rough scales were not typical of lichen planus, which usually manifests with flat (hence the name “planus”) and shiny lesions.
Prurigo nodularis is a chronic condition that manifests as intensely itchy, firm papules. The lesions can appear anywhere on the body, but more commonly are found on the extremities, back, and torso. The recent manifestation of the patient’s lesions and her lack of a history of chronic dermatitis argued against this diagnosis.
The use of a handheld dermatoscope confirmed the diagnosis by revealing white to yellow scales following the serpiginous lines. These serpiginous lines resembled scabies burrows, and at the end of some burrows, small triangular and hyperpigmented structures resembling “delta-winged jets” were seen. These “delta-winged jets” were the mite’s pigmented mouth parts and anterior legs. The burrows, which contain eggs and feces, have been described as the “contrails” behind the jets (FIGURE 2).
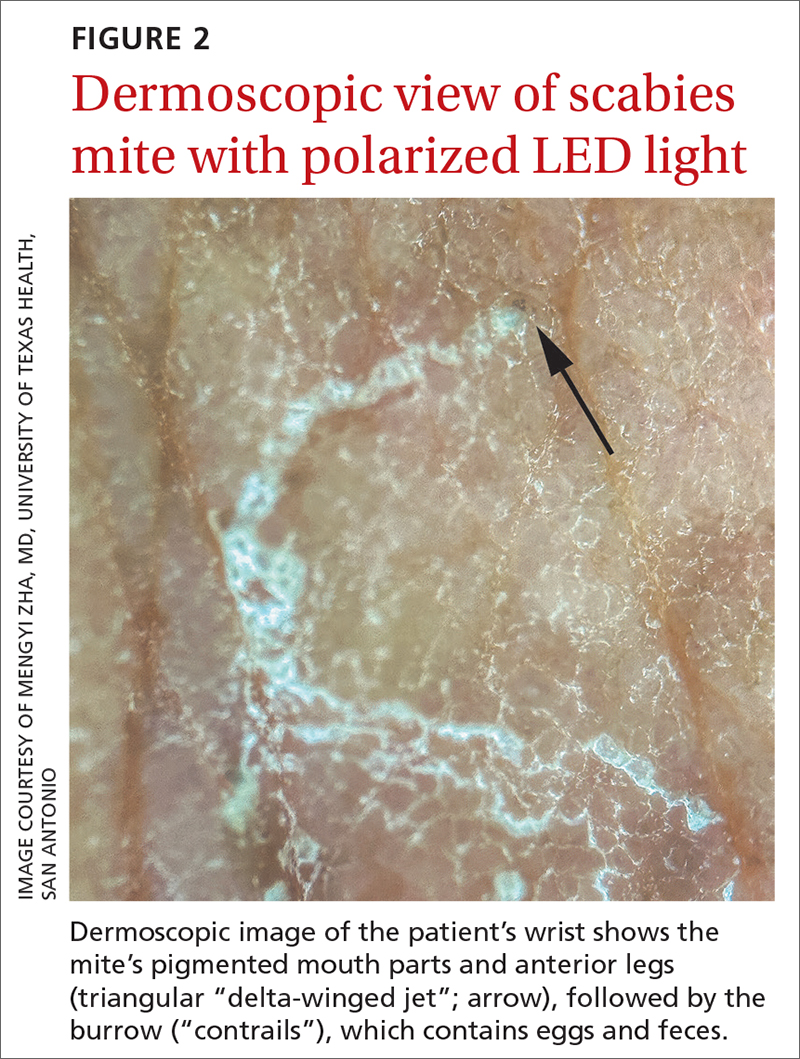
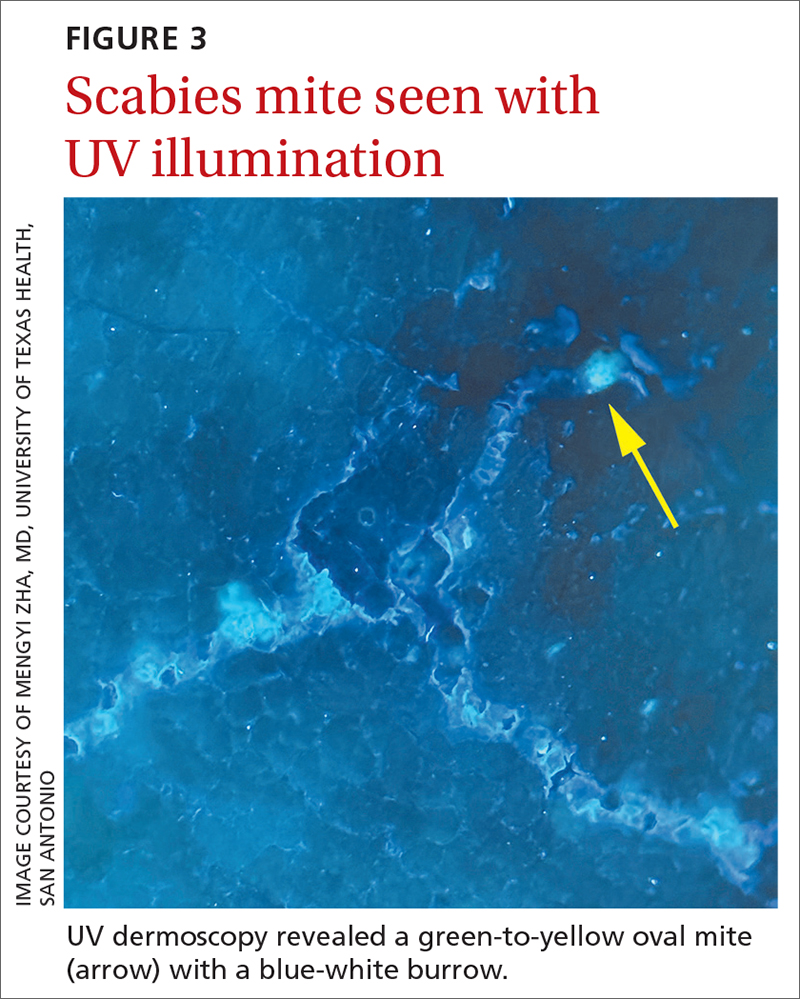
The use of a new UV illumination feature on our dermatoscope (which we’ll describe shortly) made for an even more dramatic diagnostic visual. With the click of a button, the mites fluoresced green to yellow and the burrows fluoresced white to blue (FIGURE 3).
Meeting the criteria. The clinical and dermoscopic findings met the 2020 International Alliance for the Control of Scabies (IACS) Consensus Criteria for the Diagnosis of Scabies,2 confirming the diagnosis in this patient. Scabies infestation poses a significant public health burden globally, with an estimated incidence of more than 454 million in 2016.3
Visualization is key to the diagnosis
Traditionally, the diagnosis of scabies infestation is made by direct visualization of mites via microscopy of skin scrapings.4 However, this approach is seldom feasible in a family medicine office. Fortunately, the 2020 IACS criteria included dermoscopy as a Level A diagnostic method for confirmed scabies.
Continue to: The pros and cons of dermoscopy
The pros and cons of dermoscopy. A handheld dermatoscope is an accessible, convenient tool for any clinician who treats the skin. It has been demonstrated that, in the hands of experts and novices alike, dermoscopy has a sensitivity of 91% and specificity of 86% for the diagnosis of scabies.5
However, accurate identification of the dermoscopic findings can depend on the operator and can be harder to achieve in patients who have skin of color.2 This is largely because the mite’s brown-to-black triangular head is small (sometimes hidden under skin scales) and easy to miss, especially against darker skin.
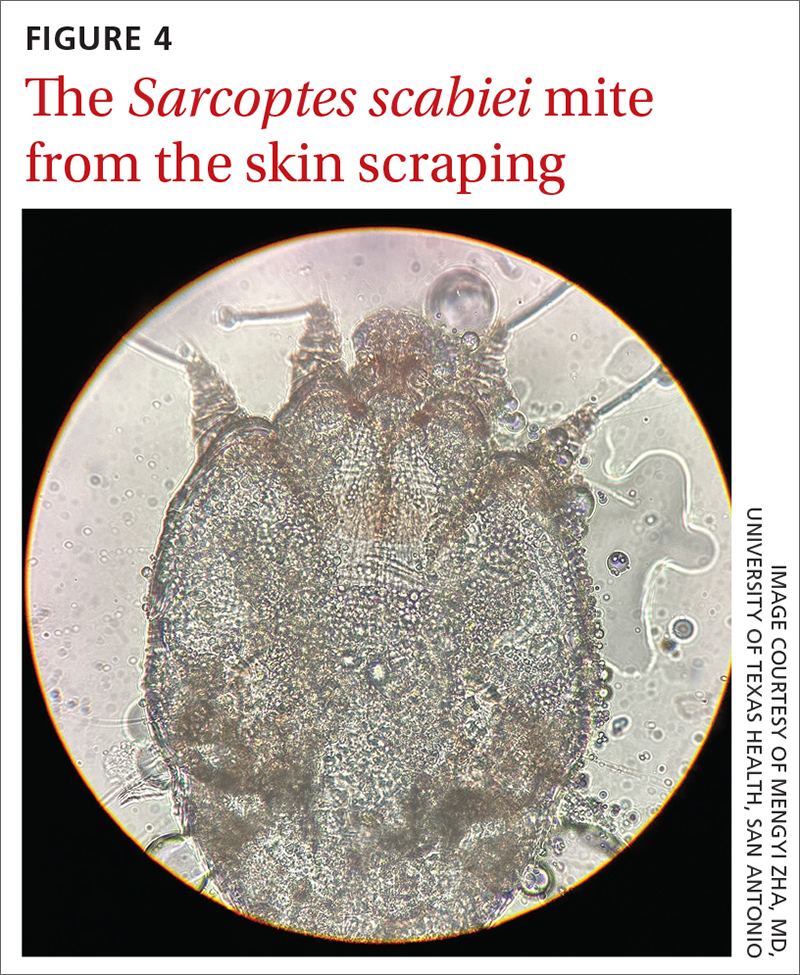
A new technologic feature helps. In this case, we used the built-in 365-nm UV illumination feature of our handheld dermatoscope (Dermlite-5) and both mites and burrows fluoresced intensely (FIGURE 3). A skin scraping at the location of the fluorescent body under microscopic examination confirmed that the organism was a Sarcoptes scabiei mite (FIGURE 4).
UV light dermoscopy can decrease operator error and ameliorate the challenge of diagnosing scabies in skin of color. Specifically, when using UV dermoscopy it’s easier to:
- locate mites, regardless of the patient’s skin color
- see the mite’s entire body, rather than just a small portion (thus increasing diagnostic certainty).
New diagnostic feature, classic treatment
Due to the severity of the patient’s scabies, she was prescribed both permethrin 5% cream and oral ivermectin 200 mcg/kg, both to be used immediately and repeated in 1 week. Notably, a systematic review indicated that topical permethrin is a superior treatment to oral ivermectin.6 However, in cases of widespread scabies and crusted scabies, it is standard of care to treat with both medications.
The patient’s pruritus was treated with cetirizine as needed. She was told that the itching might persist for a few weeks after treatment was completed.
Reinfestation was a concern with this patient because she was unable to identify a source for the mites. To minimize the likelihood of reinfestation, we advised her to decontaminate her bedding, clothing, and towels by washing them in hot water (≥ 122° F) or placing in a sealed plastic bag for at least 1 week.1 For crusted scabies cases, thorough vacuuming of a patient’s furniture and carpets is recommended.
1. Gunning K, Kiraly B, Pippitt K. Lice and scabies: treatment update. Am Fam Physician. 2019;99:635-642.
2. Engelman D, Yoshizumi J, Hay RJ, et al. The 2020 International Alliance for the Control of Scabies Consensus Criteria for the Diagnosis of Scabies. Br J Dermatol. 2020;183:808-820. doi: 10.1111/bjd.18943
A 62-YEAR-OLD HISPANIC WOMAN with a history of well-controlled diabetes and hypertension presented with an intensely pruritic rash of 3 months’ duration. She reported poor sleep due to scratching throughout the night. She denied close contact with individuals with similar rashes or itching, new intimate partners, or recent travel. She worked in an office setting and had stable, noncrowded housing.
A physical exam revealed brown and purple scaly papules and many excoriation marks. The rash was concentrated along clothing lines, around intertriginous areas, and on her ankles, wrists, and the interdigital spaces (FIGURE 1A and 1B).

WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Diagnosis: Scabies
Scabies is a diagnosis that should be considered in any patient with new-onset, widespread, nocturnal-dominant pruritus1 and it was suspected, in this case, after the initial history taking and physical exam. (See “Consider these diagnoses in cases of pruritic skin conditions” for more on lichen planus and prurigo nodularis, which were also included in the differential diagnosis.)
SIDEBAR
Consider these diagnoses in cases of pruritic skin conditions
Lichen planus is a chronic inflammatory condition that mostly affects the skin and mucosa. Characteristic findings are groups of shiny, flat-topped, firm papules. This patient’s widespread nodular lesions with rough scales were not typical of lichen planus, which usually manifests with flat (hence the name “planus”) and shiny lesions.
Prurigo nodularis is a chronic condition that manifests as intensely itchy, firm papules. The lesions can appear anywhere on the body, but more commonly are found on the extremities, back, and torso. The recent manifestation of the patient’s lesions and her lack of a history of chronic dermatitis argued against this diagnosis.
The use of a handheld dermatoscope confirmed the diagnosis by revealing white to yellow scales following the serpiginous lines. These serpiginous lines resembled scabies burrows, and at the end of some burrows, small triangular and hyperpigmented structures resembling “delta-winged jets” were seen. These “delta-winged jets” were the mite’s pigmented mouth parts and anterior legs. The burrows, which contain eggs and feces, have been described as the “contrails” behind the jets (FIGURE 2).

The use of a new UV illumination feature on our dermatoscope (which we’ll describe shortly) made for an even more dramatic diagnostic visual. With the click of a button, the mites fluoresced green to yellow and the burrows fluoresced white to blue (FIGURE 3).

Meeting the criteria. The clinical and dermoscopic findings met the 2020 International Alliance for the Control of Scabies (IACS) Consensus Criteria for the Diagnosis of Scabies,2 confirming the diagnosis in this patient. Scabies infestation poses a significant public health burden globally, with an estimated incidence of more than 454 million in 2016.3
Visualization is key to the diagnosis
Traditionally, the diagnosis of scabies infestation is made by direct visualization of mites via microscopy of skin scrapings.4 However, this approach is seldom feasible in a family medicine office. Fortunately, the 2020 IACS criteria included dermoscopy as a Level A diagnostic method for confirmed scabies.
Continue to: The pros and cons of dermoscopy
The pros and cons of dermoscopy. A handheld dermatoscope is an accessible, convenient tool for any clinician who treats the skin. It has been demonstrated that, in the hands of experts and novices alike, dermoscopy has a sensitivity of 91% and specificity of 86% for the diagnosis of scabies.5
However, accurate identification of the dermoscopic findings can depend on the operator and can be harder to achieve in patients who have skin of color.2 This is largely because the mite’s brown-to-black triangular head is small (sometimes hidden under skin scales) and easy to miss, especially against darker skin.
A new technologic feature helps. In this case, we used the built-in 365-nm UV illumination feature of our handheld dermatoscope (Dermlite-5) and both mites and burrows fluoresced intensely (FIGURE 3). A skin scraping at the location of the fluorescent body under microscopic examination confirmed that the organism was a Sarcoptes scabiei mite (FIGURE 4).

UV light dermoscopy can decrease operator error and ameliorate the challenge of diagnosing scabies in skin of color. Specifically, when using UV dermoscopy it’s easier to:
- locate mites, regardless of the patient’s skin color
- see the mite’s entire body, rather than just a small portion (thus increasing diagnostic certainty).
New diagnostic feature, classic treatment
Due to the severity of the patient’s scabies, she was prescribed both permethrin 5% cream and oral ivermectin 200 mcg/kg, both to be used immediately and repeated in 1 week. Notably, a systematic review indicated that topical permethrin is a superior treatment to oral ivermectin.6 However, in cases of widespread scabies and crusted scabies, it is standard of care to treat with both medications.
The patient’s pruritus was treated with cetirizine as needed. She was told that the itching might persist for a few weeks after treatment was completed.
Reinfestation was a concern with this patient because she was unable to identify a source for the mites. To minimize the likelihood of reinfestation, we advised her to decontaminate her bedding, clothing, and towels by washing them in hot water (≥ 122° F) or placing in a sealed plastic bag for at least 1 week.1 For crusted scabies cases, thorough vacuuming of a patient’s furniture and carpets is recommended.
A 62-YEAR-OLD HISPANIC WOMAN with a history of well-controlled diabetes and hypertension presented with an intensely pruritic rash of 3 months’ duration. She reported poor sleep due to scratching throughout the night. She denied close contact with individuals with similar rashes or itching, new intimate partners, or recent travel. She worked in an office setting and had stable, noncrowded housing.
A physical exam revealed brown and purple scaly papules and many excoriation marks. The rash was concentrated along clothing lines, around intertriginous areas, and on her ankles, wrists, and the interdigital spaces (FIGURE 1A and 1B).

WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Diagnosis: Scabies
Scabies is a diagnosis that should be considered in any patient with new-onset, widespread, nocturnal-dominant pruritus1 and it was suspected, in this case, after the initial history taking and physical exam. (See “Consider these diagnoses in cases of pruritic skin conditions” for more on lichen planus and prurigo nodularis, which were also included in the differential diagnosis.)
SIDEBAR
Consider these diagnoses in cases of pruritic skin conditions
Lichen planus is a chronic inflammatory condition that mostly affects the skin and mucosa. Characteristic findings are groups of shiny, flat-topped, firm papules. This patient’s widespread nodular lesions with rough scales were not typical of lichen planus, which usually manifests with flat (hence the name “planus”) and shiny lesions.
Prurigo nodularis is a chronic condition that manifests as intensely itchy, firm papules. The lesions can appear anywhere on the body, but more commonly are found on the extremities, back, and torso. The recent manifestation of the patient’s lesions and her lack of a history of chronic dermatitis argued against this diagnosis.
The use of a handheld dermatoscope confirmed the diagnosis by revealing white to yellow scales following the serpiginous lines. These serpiginous lines resembled scabies burrows, and at the end of some burrows, small triangular and hyperpigmented structures resembling “delta-winged jets” were seen. These “delta-winged jets” were the mite’s pigmented mouth parts and anterior legs. The burrows, which contain eggs and feces, have been described as the “contrails” behind the jets (FIGURE 2).

The use of a new UV illumination feature on our dermatoscope (which we’ll describe shortly) made for an even more dramatic diagnostic visual. With the click of a button, the mites fluoresced green to yellow and the burrows fluoresced white to blue (FIGURE 3).

Meeting the criteria. The clinical and dermoscopic findings met the 2020 International Alliance for the Control of Scabies (IACS) Consensus Criteria for the Diagnosis of Scabies,2 confirming the diagnosis in this patient. Scabies infestation poses a significant public health burden globally, with an estimated incidence of more than 454 million in 2016.3
Visualization is key to the diagnosis
Traditionally, the diagnosis of scabies infestation is made by direct visualization of mites via microscopy of skin scrapings.4 However, this approach is seldom feasible in a family medicine office. Fortunately, the 2020 IACS criteria included dermoscopy as a Level A diagnostic method for confirmed scabies.
Continue to: The pros and cons of dermoscopy
The pros and cons of dermoscopy. A handheld dermatoscope is an accessible, convenient tool for any clinician who treats the skin. It has been demonstrated that, in the hands of experts and novices alike, dermoscopy has a sensitivity of 91% and specificity of 86% for the diagnosis of scabies.5
However, accurate identification of the dermoscopic findings can depend on the operator and can be harder to achieve in patients who have skin of color.2 This is largely because the mite’s brown-to-black triangular head is small (sometimes hidden under skin scales) and easy to miss, especially against darker skin.
A new technologic feature helps. In this case, we used the built-in 365-nm UV illumination feature of our handheld dermatoscope (Dermlite-5) and both mites and burrows fluoresced intensely (FIGURE 3). A skin scraping at the location of the fluorescent body under microscopic examination confirmed that the organism was a Sarcoptes scabiei mite (FIGURE 4).

UV light dermoscopy can decrease operator error and ameliorate the challenge of diagnosing scabies in skin of color. Specifically, when using UV dermoscopy it’s easier to:
- locate mites, regardless of the patient’s skin color
- see the mite’s entire body, rather than just a small portion (thus increasing diagnostic certainty).
New diagnostic feature, classic treatment
Due to the severity of the patient’s scabies, she was prescribed both permethrin 5% cream and oral ivermectin 200 mcg/kg, both to be used immediately and repeated in 1 week. Notably, a systematic review indicated that topical permethrin is a superior treatment to oral ivermectin.6 However, in cases of widespread scabies and crusted scabies, it is standard of care to treat with both medications.
The patient’s pruritus was treated with cetirizine as needed. She was told that the itching might persist for a few weeks after treatment was completed.
Reinfestation was a concern with this patient because she was unable to identify a source for the mites. To minimize the likelihood of reinfestation, we advised her to decontaminate her bedding, clothing, and towels by washing them in hot water (≥ 122° F) or placing in a sealed plastic bag for at least 1 week.1 For crusted scabies cases, thorough vacuuming of a patient’s furniture and carpets is recommended.
1. Gunning K, Kiraly B, Pippitt K. Lice and scabies: treatment update. Am Fam Physician. 2019;99:635-642.
2. Engelman D, Yoshizumi J, Hay RJ, et al. The 2020 International Alliance for the Control of Scabies Consensus Criteria for the Diagnosis of Scabies. Br J Dermatol. 2020;183:808-820. doi: 10.1111/bjd.18943
1. Gunning K, Kiraly B, Pippitt K. Lice and scabies: treatment update. Am Fam Physician. 2019;99:635-642.
2. Engelman D, Yoshizumi J, Hay RJ, et al. The 2020 International Alliance for the Control of Scabies Consensus Criteria for the Diagnosis of Scabies. Br J Dermatol. 2020;183:808-820. doi: 10.1111/bjd.18943
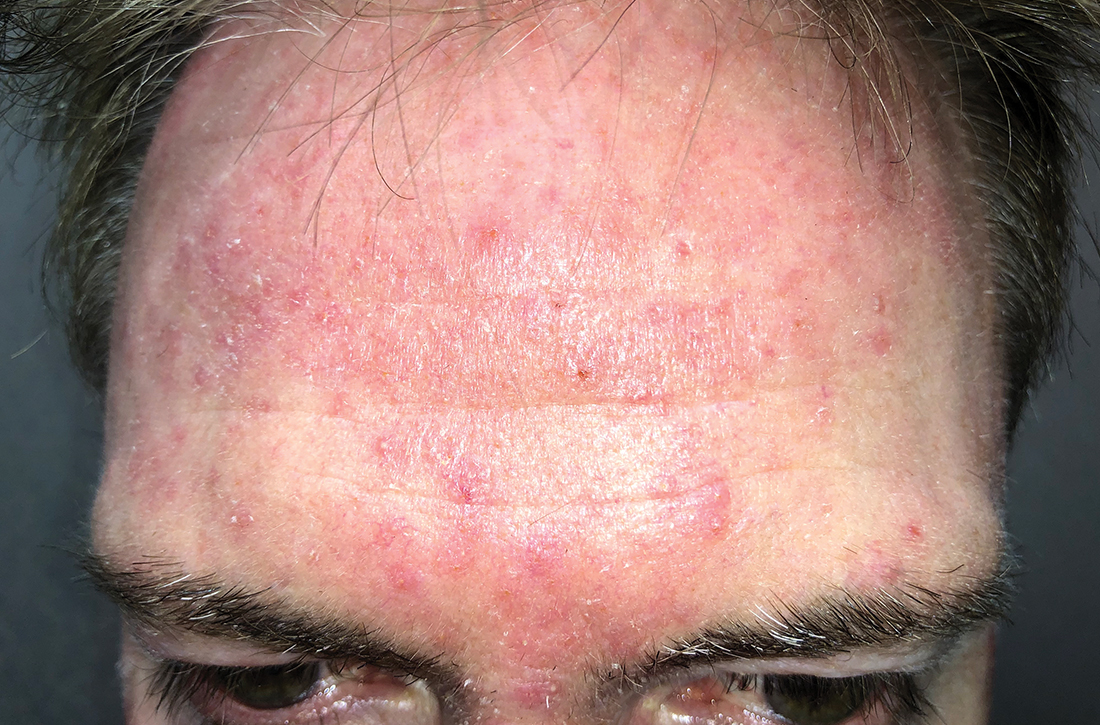
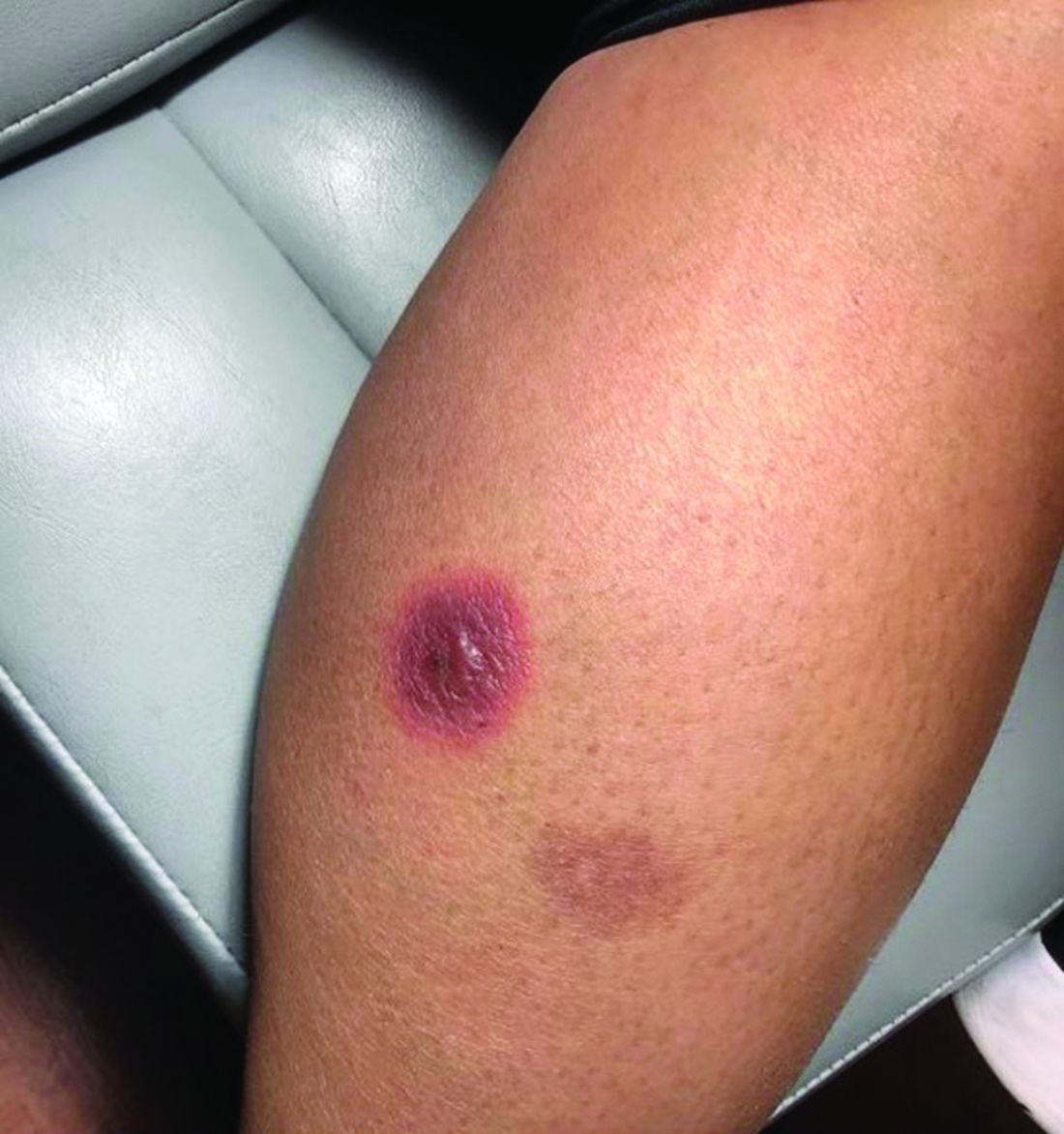
Not acne, but what?
AN OTHERWISE HEALTHY
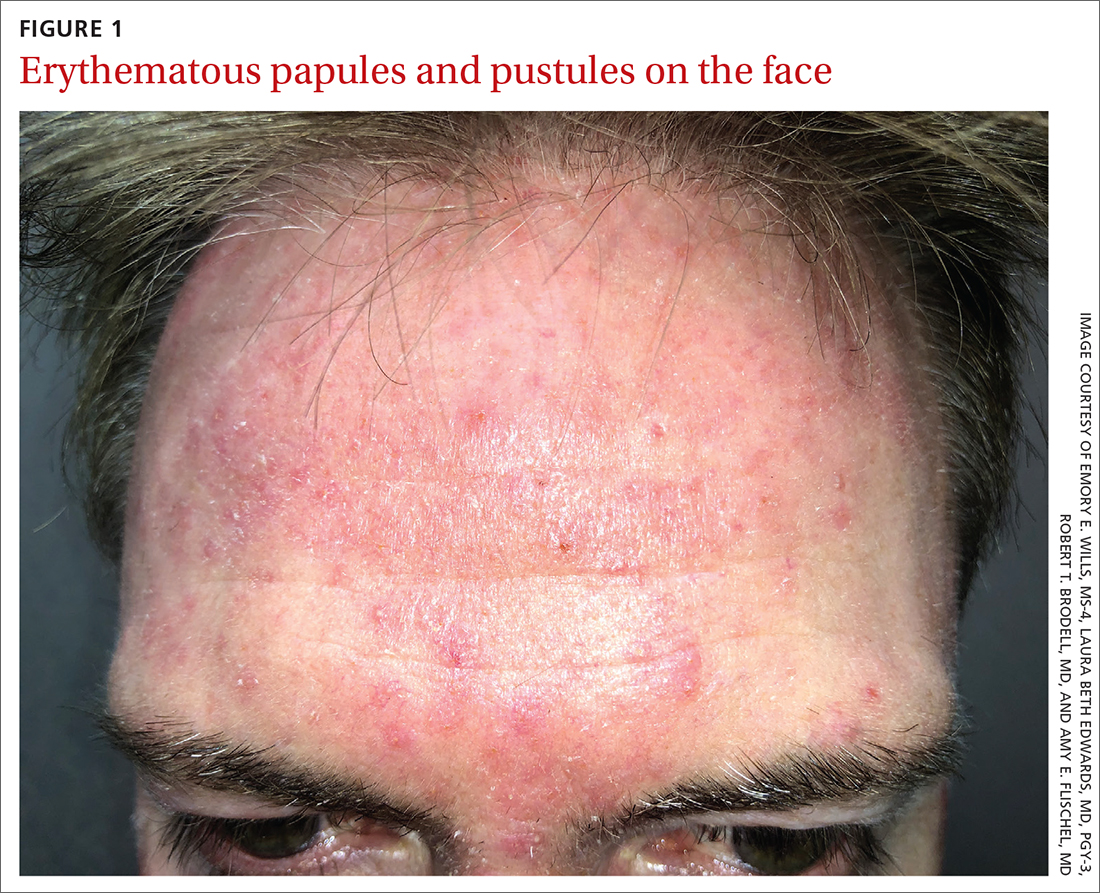
Scattered papules and pustules were present on the forehead, nose, and cheeks, with background erythema and telangiectasias (FIGURE 1). A few pinpoint crusted excoriations were noted. A sample was taken from the papules and pustules using a #15 blade and submitted for examination.
WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Diagnosis: Rosacea with Demodex mites
Under light microscopy, the scraping revealed Demodex mites (FIGURE 2). It has been proposed that these mites play a role in the inflammatory process seen in rosacea, although studies have yet to determine whether the inflammatory symptoms of rosacea cause the mites to proliferate or if the mites contribute to the initial inflammatory process.1,2
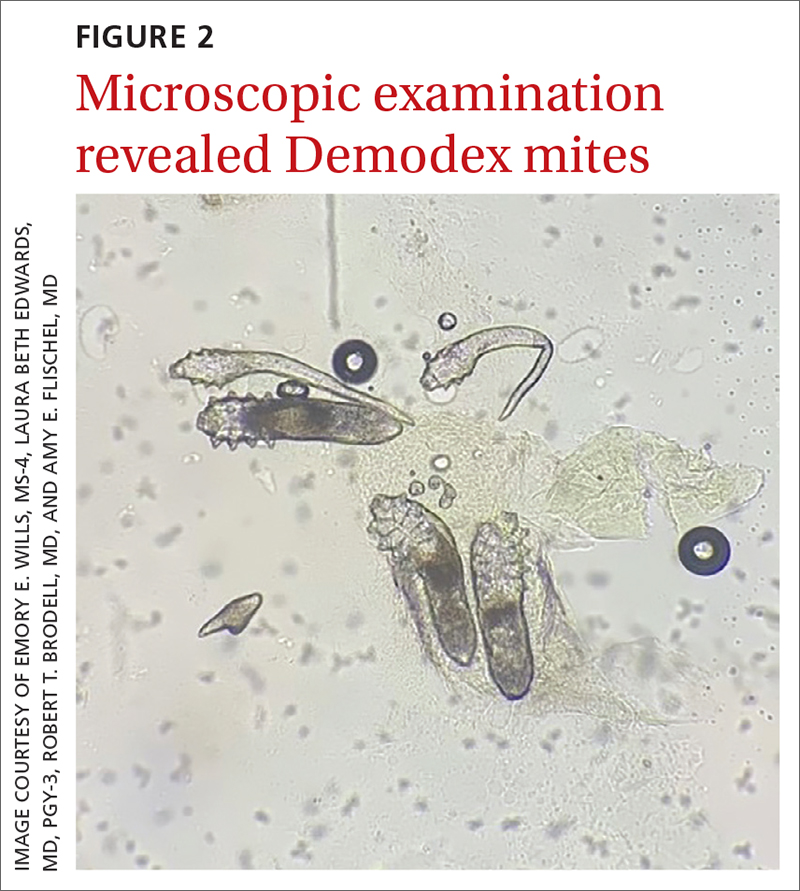
Demodex folliculorum and D brevis are part of normal skin flora; they are found in about 12% of all follicles and most commonly involve the face.3 They often become abundant in the presence of numerous sebaceous glands. Men have more sebaceous glands than women do, and thus run a greater risk for infestation with mites. An abnormal proliferation of Demodex mites can lead to demodicosis.
Demodex mites can be examined microscopically via the skin surface sampling technique known as scraping, which was done in this case. Samples taken from the papules and pustules utilizing a #15 blade are placed in immersion oil on a glass slide, cover-slipped, and examined by light microscopy.
Rosacea is thought to be an inflammatory disease in which the immune system is triggered by a variety of factors, including UV light, heat, stress, alcohol, hormonal influences, and microorganisms.1,4 The disease is found in up to 10% of the population worldwide.1
The diagnosis of rosacea requires at least 1 of the 2 “core features”—persistent central facial erythema or phymatous changes—or 2 of 4 “major features”: papules/pustules, ocular manifestation, flushing, and telangiectasias. There are 3 phenotypes: ocular, papulopustular, and erythematotelangiectatic.5,6
Continue to: The connection
The connection. Papulopustular and erythematotelangiectatic rosacea may be caused by a proliferation of Demodex mites and increased vascular endothelial growth factor production.2 In fact, a proliferation of Demodex is seen in almost all cases of papulopustular rosacea and more than 60% of cases of erythematotelangiectatic rosacea.2
Patient age and distribution of lesions narrowed the differential
Acne vulgaris is an inflammatory disease of the pilosebaceous units caused by increased sebum production, inflammation, and bacterial colonization (Propionibacterium acnes) of hair follicles on the face, neck, chest, and other areas. Both inflammatory and noninflammatory lesions can be present, and in serious cases, scarring can result.7 The case patient’s age and accompanying broad erythema were more consistent with rosacea than acne vulgaris.
Seborrheic dermatitis is a common skin condition usually stemming from an inflammatory reaction to a common yeast. Classic symptoms include scaling and erythema of the scalp and central face, as well as pruritus. Topical antifungals such as ketoconazole 2% cream and 2% shampoo are the mainstay of treatment.8 The broad distribution and papulopustules in this patient argue against the diagnosis of seborrheic dermatitis.
Systemic lupus erythematosus is a systemic inflammatory disease that often has cutaneous manifestations. Acute lupus manifests as an erythematous “butterfly rash” across the face and cheeks. Chronic discoid lupus involves depigmented plaques, erythematous macules, telangiectasias, and scarring with loss of normal hair follicles. These findings classically are photodistributed.9 The classic broad erythema extending from the cheeks over the bridge of the nose was not present in this patient.
Treatment is primarily topical
Mild cases of rosacea often can be managed with topical antibiotic creams. More severe cases may require systemic antibiotics such as tetracycline or doxycycline, although these are used with caution due to the potential for antibiotic resistance.
Ivermectin 1% cream is a US Food and Drug Administration–approved medication that is applied once daily for up to a year to treat the inflammatory pustules associated with Demodex mites. Although it is costly, studies have shown better results with topical ivermectin than with other topical medications (eg, metronidazole 0.75% gel or cream). However, metronidazole 0.75% gel applied twice daily and oral tetracycline 250 mg or doxycycline 100 mg daily or twice daily for at least 2 months often are utilized when the cost of topical ivermectin is prohibitive.10
Our patient was treated with a combination of doxycycline 100 mg daily for 30 days and
1. Forton FMN. Rosacea, an infectious disease: why rosacea with papulopustules should be considered a demodicosis. A narrative review. J Eur Acad Dermatol Venereol. 2022;36:987-1002. doi: 10.1111/jdv.18049
2. Forton FMN. The pathogenic role of demodex mites in rosacea: a potential therapeutic target already in erythematotelangiectatic rosacea? Dermatol Ther (Heidelb). 2020;10:1229-1253. doi: 10.1007/s13555-020-00458-9
3. Elston DM. Demodex mites: facts and controversies. Clin Dermatol. 2010;28:502-504. doi: 10.1016/j.clindermatol.2010.03.006
4. Erbağci Z, OzgöztaŞi O. The significance of demodex folliculorum density in rosacea. Int J Dermatol. 1998;37:421-425. doi: 10.1046/j.1365-4362.1998.00218.x
5. Tan J, Almeida LMC, Criber B, et al. Updating the diagnosis, classification and assessment of rosacea: recommendations from the global ROSacea COnsensus (ROSCO) panel. Br J Dermatol. 2017;176:431-438. doi: 10.1111/bjd.15122
6. Gallo RL, Granstein RD, Kang S, et al. Standard classification and pathophysiology of rosacea: the 2017 update by the National Rosacea Society Expert Committee. J Am Acad Dermatol. 2018;78:148-155. doi: 10.1016/j.jaad.2017.08.037
7. Williams HC, Dellavalle RP, Garner S. Acne vulgaris. Lancet. 2012;379:361-372. doi: 10.1016/S0140-6736(11)60321-8.
8. Clark GW, Pope SM, Jaboori KA. Diagnosis and treatment of seborrheic dermatitis. Am Fam Physician. 2015;91:185-190.
9. Yell JA, Mbuagbaw J, Burge SM. Cutaneous manifestations of systemic lupus erythematosus. Br J Dermatol. 1996;135:355-362.
10. Raedler LA. Soolantra (ivermectin) 1% cream: a novel, antibiotic-free agent approved for the treatment of patients with rosacea. Am Health Drug Benefits. 2015;8(Spec Feature):122-125.
AN OTHERWISE HEALTHY
Scattered papules and pustules were present on the forehead, nose, and cheeks, with background erythema and telangiectasias (FIGURE 1). A few pinpoint crusted excoriations were noted. A sample was taken from the papules and pustules using a #15 blade and submitted for examination.

WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Diagnosis: Rosacea with Demodex mites
Under light microscopy, the scraping revealed Demodex mites (FIGURE 2). It has been proposed that these mites play a role in the inflammatory process seen in rosacea, although studies have yet to determine whether the inflammatory symptoms of rosacea cause the mites to proliferate or if the mites contribute to the initial inflammatory process.1,2

Demodex folliculorum and D brevis are part of normal skin flora; they are found in about 12% of all follicles and most commonly involve the face.3 They often become abundant in the presence of numerous sebaceous glands. Men have more sebaceous glands than women do, and thus run a greater risk for infestation with mites. An abnormal proliferation of Demodex mites can lead to demodicosis.
Demodex mites can be examined microscopically via the skin surface sampling technique known as scraping, which was done in this case. Samples taken from the papules and pustules utilizing a #15 blade are placed in immersion oil on a glass slide, cover-slipped, and examined by light microscopy.
Rosacea is thought to be an inflammatory disease in which the immune system is triggered by a variety of factors, including UV light, heat, stress, alcohol, hormonal influences, and microorganisms.1,4 The disease is found in up to 10% of the population worldwide.1
The diagnosis of rosacea requires at least 1 of the 2 “core features”—persistent central facial erythema or phymatous changes—or 2 of 4 “major features”: papules/pustules, ocular manifestation, flushing, and telangiectasias. There are 3 phenotypes: ocular, papulopustular, and erythematotelangiectatic.5,6
Continue to: The connection
The connection. Papulopustular and erythematotelangiectatic rosacea may be caused by a proliferation of Demodex mites and increased vascular endothelial growth factor production.2 In fact, a proliferation of Demodex is seen in almost all cases of papulopustular rosacea and more than 60% of cases of erythematotelangiectatic rosacea.2
Patient age and distribution of lesions narrowed the differential
Acne vulgaris is an inflammatory disease of the pilosebaceous units caused by increased sebum production, inflammation, and bacterial colonization (Propionibacterium acnes) of hair follicles on the face, neck, chest, and other areas. Both inflammatory and noninflammatory lesions can be present, and in serious cases, scarring can result.7 The case patient’s age and accompanying broad erythema were more consistent with rosacea than acne vulgaris.
Seborrheic dermatitis is a common skin condition usually stemming from an inflammatory reaction to a common yeast. Classic symptoms include scaling and erythema of the scalp and central face, as well as pruritus. Topical antifungals such as ketoconazole 2% cream and 2% shampoo are the mainstay of treatment.8 The broad distribution and papulopustules in this patient argue against the diagnosis of seborrheic dermatitis.
Systemic lupus erythematosus is a systemic inflammatory disease that often has cutaneous manifestations. Acute lupus manifests as an erythematous “butterfly rash” across the face and cheeks. Chronic discoid lupus involves depigmented plaques, erythematous macules, telangiectasias, and scarring with loss of normal hair follicles. These findings classically are photodistributed.9 The classic broad erythema extending from the cheeks over the bridge of the nose was not present in this patient.
Treatment is primarily topical
Mild cases of rosacea often can be managed with topical antibiotic creams. More severe cases may require systemic antibiotics such as tetracycline or doxycycline, although these are used with caution due to the potential for antibiotic resistance.
Ivermectin 1% cream is a US Food and Drug Administration–approved medication that is applied once daily for up to a year to treat the inflammatory pustules associated with Demodex mites. Although it is costly, studies have shown better results with topical ivermectin than with other topical medications (eg, metronidazole 0.75% gel or cream). However, metronidazole 0.75% gel applied twice daily and oral tetracycline 250 mg or doxycycline 100 mg daily or twice daily for at least 2 months often are utilized when the cost of topical ivermectin is prohibitive.10
Our patient was treated with a combination of doxycycline 100 mg daily for 30 days and
AN OTHERWISE HEALTHY
Scattered papules and pustules were present on the forehead, nose, and cheeks, with background erythema and telangiectasias (FIGURE 1). A few pinpoint crusted excoriations were noted. A sample was taken from the papules and pustules using a #15 blade and submitted for examination.

WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Diagnosis: Rosacea with Demodex mites
Under light microscopy, the scraping revealed Demodex mites (FIGURE 2). It has been proposed that these mites play a role in the inflammatory process seen in rosacea, although studies have yet to determine whether the inflammatory symptoms of rosacea cause the mites to proliferate or if the mites contribute to the initial inflammatory process.1,2

Demodex folliculorum and D brevis are part of normal skin flora; they are found in about 12% of all follicles and most commonly involve the face.3 They often become abundant in the presence of numerous sebaceous glands. Men have more sebaceous glands than women do, and thus run a greater risk for infestation with mites. An abnormal proliferation of Demodex mites can lead to demodicosis.
Demodex mites can be examined microscopically via the skin surface sampling technique known as scraping, which was done in this case. Samples taken from the papules and pustules utilizing a #15 blade are placed in immersion oil on a glass slide, cover-slipped, and examined by light microscopy.
Rosacea is thought to be an inflammatory disease in which the immune system is triggered by a variety of factors, including UV light, heat, stress, alcohol, hormonal influences, and microorganisms.1,4 The disease is found in up to 10% of the population worldwide.1
The diagnosis of rosacea requires at least 1 of the 2 “core features”—persistent central facial erythema or phymatous changes—or 2 of 4 “major features”: papules/pustules, ocular manifestation, flushing, and telangiectasias. There are 3 phenotypes: ocular, papulopustular, and erythematotelangiectatic.5,6
Continue to: The connection
The connection. Papulopustular and erythematotelangiectatic rosacea may be caused by a proliferation of Demodex mites and increased vascular endothelial growth factor production.2 In fact, a proliferation of Demodex is seen in almost all cases of papulopustular rosacea and more than 60% of cases of erythematotelangiectatic rosacea.2
Patient age and distribution of lesions narrowed the differential
Acne vulgaris is an inflammatory disease of the pilosebaceous units caused by increased sebum production, inflammation, and bacterial colonization (Propionibacterium acnes) of hair follicles on the face, neck, chest, and other areas. Both inflammatory and noninflammatory lesions can be present, and in serious cases, scarring can result.7 The case patient’s age and accompanying broad erythema were more consistent with rosacea than acne vulgaris.
Seborrheic dermatitis is a common skin condition usually stemming from an inflammatory reaction to a common yeast. Classic symptoms include scaling and erythema of the scalp and central face, as well as pruritus. Topical antifungals such as ketoconazole 2% cream and 2% shampoo are the mainstay of treatment.8 The broad distribution and papulopustules in this patient argue against the diagnosis of seborrheic dermatitis.
Systemic lupus erythematosus is a systemic inflammatory disease that often has cutaneous manifestations. Acute lupus manifests as an erythematous “butterfly rash” across the face and cheeks. Chronic discoid lupus involves depigmented plaques, erythematous macules, telangiectasias, and scarring with loss of normal hair follicles. These findings classically are photodistributed.9 The classic broad erythema extending from the cheeks over the bridge of the nose was not present in this patient.
Treatment is primarily topical
Mild cases of rosacea often can be managed with topical antibiotic creams. More severe cases may require systemic antibiotics such as tetracycline or doxycycline, although these are used with caution due to the potential for antibiotic resistance.
Ivermectin 1% cream is a US Food and Drug Administration–approved medication that is applied once daily for up to a year to treat the inflammatory pustules associated with Demodex mites. Although it is costly, studies have shown better results with topical ivermectin than with other topical medications (eg, metronidazole 0.75% gel or cream). However, metronidazole 0.75% gel applied twice daily and oral tetracycline 250 mg or doxycycline 100 mg daily or twice daily for at least 2 months often are utilized when the cost of topical ivermectin is prohibitive.10
Our patient was treated with a combination of doxycycline 100 mg daily for 30 days and
1. Forton FMN. Rosacea, an infectious disease: why rosacea with papulopustules should be considered a demodicosis. A narrative review. J Eur Acad Dermatol Venereol. 2022;36:987-1002. doi: 10.1111/jdv.18049
2. Forton FMN. The pathogenic role of demodex mites in rosacea: a potential therapeutic target already in erythematotelangiectatic rosacea? Dermatol Ther (Heidelb). 2020;10:1229-1253. doi: 10.1007/s13555-020-00458-9
3. Elston DM. Demodex mites: facts and controversies. Clin Dermatol. 2010;28:502-504. doi: 10.1016/j.clindermatol.2010.03.006
4. Erbağci Z, OzgöztaŞi O. The significance of demodex folliculorum density in rosacea. Int J Dermatol. 1998;37:421-425. doi: 10.1046/j.1365-4362.1998.00218.x
5. Tan J, Almeida LMC, Criber B, et al. Updating the diagnosis, classification and assessment of rosacea: recommendations from the global ROSacea COnsensus (ROSCO) panel. Br J Dermatol. 2017;176:431-438. doi: 10.1111/bjd.15122
6. Gallo RL, Granstein RD, Kang S, et al. Standard classification and pathophysiology of rosacea: the 2017 update by the National Rosacea Society Expert Committee. J Am Acad Dermatol. 2018;78:148-155. doi: 10.1016/j.jaad.2017.08.037
7. Williams HC, Dellavalle RP, Garner S. Acne vulgaris. Lancet. 2012;379:361-372. doi: 10.1016/S0140-6736(11)60321-8.
8. Clark GW, Pope SM, Jaboori KA. Diagnosis and treatment of seborrheic dermatitis. Am Fam Physician. 2015;91:185-190.
9. Yell JA, Mbuagbaw J, Burge SM. Cutaneous manifestations of systemic lupus erythematosus. Br J Dermatol. 1996;135:355-362.
10. Raedler LA. Soolantra (ivermectin) 1% cream: a novel, antibiotic-free agent approved for the treatment of patients with rosacea. Am Health Drug Benefits. 2015;8(Spec Feature):122-125.
1. Forton FMN. Rosacea, an infectious disease: why rosacea with papulopustules should be considered a demodicosis. A narrative review. J Eur Acad Dermatol Venereol. 2022;36:987-1002. doi: 10.1111/jdv.18049
2. Forton FMN. The pathogenic role of demodex mites in rosacea: a potential therapeutic target already in erythematotelangiectatic rosacea? Dermatol Ther (Heidelb). 2020;10:1229-1253. doi: 10.1007/s13555-020-00458-9
3. Elston DM. Demodex mites: facts and controversies. Clin Dermatol. 2010;28:502-504. doi: 10.1016/j.clindermatol.2010.03.006
4. Erbağci Z, OzgöztaŞi O. The significance of demodex folliculorum density in rosacea. Int J Dermatol. 1998;37:421-425. doi: 10.1046/j.1365-4362.1998.00218.x
5. Tan J, Almeida LMC, Criber B, et al. Updating the diagnosis, classification and assessment of rosacea: recommendations from the global ROSacea COnsensus (ROSCO) panel. Br J Dermatol. 2017;176:431-438. doi: 10.1111/bjd.15122
6. Gallo RL, Granstein RD, Kang S, et al. Standard classification and pathophysiology of rosacea: the 2017 update by the National Rosacea Society Expert Committee. J Am Acad Dermatol. 2018;78:148-155. doi: 10.1016/j.jaad.2017.08.037
7. Williams HC, Dellavalle RP, Garner S. Acne vulgaris. Lancet. 2012;379:361-372. doi: 10.1016/S0140-6736(11)60321-8.
8. Clark GW, Pope SM, Jaboori KA. Diagnosis and treatment of seborrheic dermatitis. Am Fam Physician. 2015;91:185-190.
9. Yell JA, Mbuagbaw J, Burge SM. Cutaneous manifestations of systemic lupus erythematosus. Br J Dermatol. 1996;135:355-362.
10. Raedler LA. Soolantra (ivermectin) 1% cream: a novel, antibiotic-free agent approved for the treatment of patients with rosacea. Am Health Drug Benefits. 2015;8(Spec Feature):122-125.
Analysis spotlights economic burden of vitiligo in the U.S.
TOPLINE:
METHODOLOGY:
- No published studies have quantified the medical costs and health care resource utilization (HCRU) among patients with vitiligo in the United States, compared with the general population.
- Drawing from the Merative MarketScan Commercial Claims and Encounters database, researchers reviewed the records of 49,512 patients diagnosed with vitiligo between Jan. 1, 2008, and Dec. 31, 2020, and those of 99,024 matched control persons who did not have vitiligo.
- Costs were in 2021 dollars during a 1-year postindex period. The student t test and chi square analysis were used to determine P values.
TAKEAWAY:
- In both cohorts, the median age of patients was 43 years, 79.2% were female, and most (39%) were from the southern region of the United States.
- All-cause total health care costs for patients with vitiligo were significantly higher than those of matched controls ($15,551 vs. $7,735; P < .0001).
- Similarly, medical costs for patients with vitiligo were significantly higher than those of control persons ($11,953 vs. $5,722), as were pharmacy costs ($3,598 vs. $2,014; P < .001 for both associations).
- A significantly greater proportion of patients with vitiligo had higher all-cause HCRU, compared with matched control persons. That included at least one ED visit (17.5% vs 13.4%), at least one inpatient visit (12.9% vs 6.8%), and at least one outpatient visit (99.8% vs. 88.3%; P < .0001 for all associations).
IN PRACTICE:
“These findings reveal an unmet need for cost-effective treatments and highlight the importance of fully identifying the drivers of economic burden for patients with vitiligo,” the authors concluded.
SOURCE:
Khaled Ezzedine, MD, PhD, of the department of dermatology at the Henri Mondor University Hospital, Créteil, France, led the study, which was published in the Journal of Investigative Dermatology.
LIMITATIONS:
The investigators did not evaluate indirect medical costs of vitiligo, such as work productivity, early retirement, and lost opportunities. Also, the results may not be generalizable to populations outside of the United States.
DISCLOSURES:
Dr. Ezzedine has received honoraria as a consultant for AbbVie, Incyte, La Roche–Posay, Pfizer, Pierre Fabre, Sanofi, and Viela Bio. One author is an investigator for Incyte and is a consultant for several pharmaceutical companies. Three authors are AbbVie employees.
A version of this article first appeared on Medscape.com.
TOPLINE:
METHODOLOGY:
- No published studies have quantified the medical costs and health care resource utilization (HCRU) among patients with vitiligo in the United States, compared with the general population.
- Drawing from the Merative MarketScan Commercial Claims and Encounters database, researchers reviewed the records of 49,512 patients diagnosed with vitiligo between Jan. 1, 2008, and Dec. 31, 2020, and those of 99,024 matched control persons who did not have vitiligo.
- Costs were in 2021 dollars during a 1-year postindex period. The student t test and chi square analysis were used to determine P values.
TAKEAWAY:
- In both cohorts, the median age of patients was 43 years, 79.2% were female, and most (39%) were from the southern region of the United States.
- All-cause total health care costs for patients with vitiligo were significantly higher than those of matched controls ($15,551 vs. $7,735; P < .0001).
- Similarly, medical costs for patients with vitiligo were significantly higher than those of control persons ($11,953 vs. $5,722), as were pharmacy costs ($3,598 vs. $2,014; P < .001 for both associations).
- A significantly greater proportion of patients with vitiligo had higher all-cause HCRU, compared with matched control persons. That included at least one ED visit (17.5% vs 13.4%), at least one inpatient visit (12.9% vs 6.8%), and at least one outpatient visit (99.8% vs. 88.3%; P < .0001 for all associations).
IN PRACTICE:
“These findings reveal an unmet need for cost-effective treatments and highlight the importance of fully identifying the drivers of economic burden for patients with vitiligo,” the authors concluded.
SOURCE:
Khaled Ezzedine, MD, PhD, of the department of dermatology at the Henri Mondor University Hospital, Créteil, France, led the study, which was published in the Journal of Investigative Dermatology.
LIMITATIONS:
The investigators did not evaluate indirect medical costs of vitiligo, such as work productivity, early retirement, and lost opportunities. Also, the results may not be generalizable to populations outside of the United States.
DISCLOSURES:
Dr. Ezzedine has received honoraria as a consultant for AbbVie, Incyte, La Roche–Posay, Pfizer, Pierre Fabre, Sanofi, and Viela Bio. One author is an investigator for Incyte and is a consultant for several pharmaceutical companies. Three authors are AbbVie employees.
A version of this article first appeared on Medscape.com.
TOPLINE:
METHODOLOGY:
- No published studies have quantified the medical costs and health care resource utilization (HCRU) among patients with vitiligo in the United States, compared with the general population.
- Drawing from the Merative MarketScan Commercial Claims and Encounters database, researchers reviewed the records of 49,512 patients diagnosed with vitiligo between Jan. 1, 2008, and Dec. 31, 2020, and those of 99,024 matched control persons who did not have vitiligo.
- Costs were in 2021 dollars during a 1-year postindex period. The student t test and chi square analysis were used to determine P values.
TAKEAWAY:
- In both cohorts, the median age of patients was 43 years, 79.2% were female, and most (39%) were from the southern region of the United States.
- All-cause total health care costs for patients with vitiligo were significantly higher than those of matched controls ($15,551 vs. $7,735; P < .0001).
- Similarly, medical costs for patients with vitiligo were significantly higher than those of control persons ($11,953 vs. $5,722), as were pharmacy costs ($3,598 vs. $2,014; P < .001 for both associations).
- A significantly greater proportion of patients with vitiligo had higher all-cause HCRU, compared with matched control persons. That included at least one ED visit (17.5% vs 13.4%), at least one inpatient visit (12.9% vs 6.8%), and at least one outpatient visit (99.8% vs. 88.3%; P < .0001 for all associations).
IN PRACTICE:
“These findings reveal an unmet need for cost-effective treatments and highlight the importance of fully identifying the drivers of economic burden for patients with vitiligo,” the authors concluded.
SOURCE:
Khaled Ezzedine, MD, PhD, of the department of dermatology at the Henri Mondor University Hospital, Créteil, France, led the study, which was published in the Journal of Investigative Dermatology.
LIMITATIONS:
The investigators did not evaluate indirect medical costs of vitiligo, such as work productivity, early retirement, and lost opportunities. Also, the results may not be generalizable to populations outside of the United States.
DISCLOSURES:
Dr. Ezzedine has received honoraria as a consultant for AbbVie, Incyte, La Roche–Posay, Pfizer, Pierre Fabre, Sanofi, and Viela Bio. One author is an investigator for Incyte and is a consultant for several pharmaceutical companies. Three authors are AbbVie employees.
A version of this article first appeared on Medscape.com.
Pediatric psoriasis: Black children, males more likely to have palmoplantar subtype, study finds
TOPLINE:
.
METHODOLOGY:
- Researchers reviewed data on 330 children and youths aged 0-18 years who had received a primary psoriasis diagnosis and who were seen at an academic pediatric dermatology clinic from 2012 to 2022. Among these patients, 50 cases of palmoplantar psoriasis (PP) were identified by pediatric dermatologists.
- The study population was stratified by race/ethnicity on the basis of self-identification. The cohort included White, Black, and Hispanic/Latino patients, as well as patients who identified as other; 71.5% were White persons, 59.1% were female patients.
- The researchers used a regression analysis to investigate the association between race/ethnicity and PP after controlling for multiple confounding variables, including age and gender.
TAKEAWAY:
- Black children were significantly more likely to have PP than White children (adjusted odds ratio, 6.386; P < .0001). PP was diagnosed in 41.9%, 11.5%, and 8.9% of Black, Hispanic/Latino, and White children, respectively.
- Male gender was also identified as an independent risk factor for PP (aOR, 2.241).
- Nail involvement occurred in significantly more Black and Hispanic/Latino patients than in White patients (53.2%, 50.0%, and 33.9%, respectively).
- Black patients had significantly more palm and sole involvement, compared with the other groups (P < .0001 for both); however, White children had significantly more scalp involvement, compared with the other groups (P = .04).
IN PRACTICE:
“Further research is warranted to better understand the degree to which these associations are affected by racial disparities and environmental factors,” as well as potential genetic associations, the researchers noted.
SOURCE:
The corresponding author on the study was Amy Theos, MD, of the department of dermatology at the University of Alabama, Birmingham. The study was published online in Pediatric Dermatology.
LIMITATIONS:
The findings were limited by the small sample size and incomplete data for some patients.
DISCLOSURES:
The study received no outside funding. The researchers had no financial conflicts to disclose.
A version of this article first appeared on Medscape.com.
TOPLINE:
.
METHODOLOGY:
- Researchers reviewed data on 330 children and youths aged 0-18 years who had received a primary psoriasis diagnosis and who were seen at an academic pediatric dermatology clinic from 2012 to 2022. Among these patients, 50 cases of palmoplantar psoriasis (PP) were identified by pediatric dermatologists.
- The study population was stratified by race/ethnicity on the basis of self-identification. The cohort included White, Black, and Hispanic/Latino patients, as well as patients who identified as other; 71.5% were White persons, 59.1% were female patients.
- The researchers used a regression analysis to investigate the association between race/ethnicity and PP after controlling for multiple confounding variables, including age and gender.
TAKEAWAY:
- Black children were significantly more likely to have PP than White children (adjusted odds ratio, 6.386; P < .0001). PP was diagnosed in 41.9%, 11.5%, and 8.9% of Black, Hispanic/Latino, and White children, respectively.
- Male gender was also identified as an independent risk factor for PP (aOR, 2.241).
- Nail involvement occurred in significantly more Black and Hispanic/Latino patients than in White patients (53.2%, 50.0%, and 33.9%, respectively).
- Black patients had significantly more palm and sole involvement, compared with the other groups (P < .0001 for both); however, White children had significantly more scalp involvement, compared with the other groups (P = .04).
IN PRACTICE:
“Further research is warranted to better understand the degree to which these associations are affected by racial disparities and environmental factors,” as well as potential genetic associations, the researchers noted.
SOURCE:
The corresponding author on the study was Amy Theos, MD, of the department of dermatology at the University of Alabama, Birmingham. The study was published online in Pediatric Dermatology.
LIMITATIONS:
The findings were limited by the small sample size and incomplete data for some patients.
DISCLOSURES:
The study received no outside funding. The researchers had no financial conflicts to disclose.
A version of this article first appeared on Medscape.com.
TOPLINE:
.
METHODOLOGY:
- Researchers reviewed data on 330 children and youths aged 0-18 years who had received a primary psoriasis diagnosis and who were seen at an academic pediatric dermatology clinic from 2012 to 2022. Among these patients, 50 cases of palmoplantar psoriasis (PP) were identified by pediatric dermatologists.
- The study population was stratified by race/ethnicity on the basis of self-identification. The cohort included White, Black, and Hispanic/Latino patients, as well as patients who identified as other; 71.5% were White persons, 59.1% were female patients.
- The researchers used a regression analysis to investigate the association between race/ethnicity and PP after controlling for multiple confounding variables, including age and gender.
TAKEAWAY:
- Black children were significantly more likely to have PP than White children (adjusted odds ratio, 6.386; P < .0001). PP was diagnosed in 41.9%, 11.5%, and 8.9% of Black, Hispanic/Latino, and White children, respectively.
- Male gender was also identified as an independent risk factor for PP (aOR, 2.241).
- Nail involvement occurred in significantly more Black and Hispanic/Latino patients than in White patients (53.2%, 50.0%, and 33.9%, respectively).
- Black patients had significantly more palm and sole involvement, compared with the other groups (P < .0001 for both); however, White children had significantly more scalp involvement, compared with the other groups (P = .04).
IN PRACTICE:
“Further research is warranted to better understand the degree to which these associations are affected by racial disparities and environmental factors,” as well as potential genetic associations, the researchers noted.
SOURCE:
The corresponding author on the study was Amy Theos, MD, of the department of dermatology at the University of Alabama, Birmingham. The study was published online in Pediatric Dermatology.
LIMITATIONS:
The findings were limited by the small sample size and incomplete data for some patients.
DISCLOSURES:
The study received no outside funding. The researchers had no financial conflicts to disclose.
A version of this article first appeared on Medscape.com.
A 42-year-old woman presented with a few days of erosions on her buccal mucosa, tongue, and soft palate
in which lesions present in the same location upon repeated intake of the offending drug. The lesions typically present within 30 minutes to 8 hours of administration of the drug. These reactions can be considered allergic or pseudo-allergic, in which case, there is no notable adaptive immune response. CD8+ T cells appear to play a role in the epidermal injury via release of interferons and interactions with other inflammatory cells.
There are numerous drugs that can precipitate these findings. NSAIDs; antibiotics, such as tetracyclines, sulfonamides; and phenytoin are common offenders. In the case of our patient, naproxen was the offending medication.
The classic presentation of FDE features annular, erythematous to violaceous macules on the skin or mucosa that can be asymptomatic or can produce burning, pain, or pruritus. The most common locations include the trunk and extremities, but the palms, soles, face, scalp, and mucosa can also be impacted. The oral mucosa seems to be the most common mucosal location. Intravenous administration of a drug is associated with more severe symptoms. Systemic symptoms are typically absent, and the eruption may initially be in one location, but may appear elsewhere upon repeated exposure to the offending medication.
The differential diagnosis includes arthropod bite reactions, urticaria, and erythema multiforme. Although FDEs are typically a clinical diagnosis, the histopathology will commonly show a vacuolar interface dermatitis. Furthermore, a variety of immune cells can be found, including neutrophilic, eosinophilic, and lymphocytic infiltrate. A combination of two or more histological patterns often favors the diagnosis of FDE.
Steroid creams can be prescribed to decrease the inflammatory reaction and improve symptoms; however, the definitive treatment of this condition is cessation of the offending agent. Postinflammatory hyperpigmentation is a common symptom after resolution of the condition, and it may take months to fade away. Further darkening can be prevented by practicing sun safety measures such as wearing sunblock, covering the affected areas, and avoiding prolonged sun exposure.
This case and the photos were submitted by Lucas Shapiro, BS, of Nova Southeastern University College of Osteopathic Medicine, Fort Lauderdale, Fla., and Igor Chaplik, DO, Aesthetix Dermatology, Fort Lauderdale. The column was edited by Donna Bilu Martin, MD.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to [email protected].
References
Shaker G et al. Cureus. 2022 Aug 23;14(8):e28299.
Srivastava R et al. Indian J Dent. 2015 Apr-Jun;6(2):103-6.
Weyers W, Metze D. Dermatol Pract Concept. 2011 Jan 31;1(1):33-47.
in which lesions present in the same location upon repeated intake of the offending drug. The lesions typically present within 30 minutes to 8 hours of administration of the drug. These reactions can be considered allergic or pseudo-allergic, in which case, there is no notable adaptive immune response. CD8+ T cells appear to play a role in the epidermal injury via release of interferons and interactions with other inflammatory cells.
There are numerous drugs that can precipitate these findings. NSAIDs; antibiotics, such as tetracyclines, sulfonamides; and phenytoin are common offenders. In the case of our patient, naproxen was the offending medication.
The classic presentation of FDE features annular, erythematous to violaceous macules on the skin or mucosa that can be asymptomatic or can produce burning, pain, or pruritus. The most common locations include the trunk and extremities, but the palms, soles, face, scalp, and mucosa can also be impacted. The oral mucosa seems to be the most common mucosal location. Intravenous administration of a drug is associated with more severe symptoms. Systemic symptoms are typically absent, and the eruption may initially be in one location, but may appear elsewhere upon repeated exposure to the offending medication.
The differential diagnosis includes arthropod bite reactions, urticaria, and erythema multiforme. Although FDEs are typically a clinical diagnosis, the histopathology will commonly show a vacuolar interface dermatitis. Furthermore, a variety of immune cells can be found, including neutrophilic, eosinophilic, and lymphocytic infiltrate. A combination of two or more histological patterns often favors the diagnosis of FDE.
Steroid creams can be prescribed to decrease the inflammatory reaction and improve symptoms; however, the definitive treatment of this condition is cessation of the offending agent. Postinflammatory hyperpigmentation is a common symptom after resolution of the condition, and it may take months to fade away. Further darkening can be prevented by practicing sun safety measures such as wearing sunblock, covering the affected areas, and avoiding prolonged sun exposure.
This case and the photos were submitted by Lucas Shapiro, BS, of Nova Southeastern University College of Osteopathic Medicine, Fort Lauderdale, Fla., and Igor Chaplik, DO, Aesthetix Dermatology, Fort Lauderdale. The column was edited by Donna Bilu Martin, MD.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to [email protected].
References
Shaker G et al. Cureus. 2022 Aug 23;14(8):e28299.
Srivastava R et al. Indian J Dent. 2015 Apr-Jun;6(2):103-6.
Weyers W, Metze D. Dermatol Pract Concept. 2011 Jan 31;1(1):33-47.
in which lesions present in the same location upon repeated intake of the offending drug. The lesions typically present within 30 minutes to 8 hours of administration of the drug. These reactions can be considered allergic or pseudo-allergic, in which case, there is no notable adaptive immune response. CD8+ T cells appear to play a role in the epidermal injury via release of interferons and interactions with other inflammatory cells.
There are numerous drugs that can precipitate these findings. NSAIDs; antibiotics, such as tetracyclines, sulfonamides; and phenytoin are common offenders. In the case of our patient, naproxen was the offending medication.
The classic presentation of FDE features annular, erythematous to violaceous macules on the skin or mucosa that can be asymptomatic or can produce burning, pain, or pruritus. The most common locations include the trunk and extremities, but the palms, soles, face, scalp, and mucosa can also be impacted. The oral mucosa seems to be the most common mucosal location. Intravenous administration of a drug is associated with more severe symptoms. Systemic symptoms are typically absent, and the eruption may initially be in one location, but may appear elsewhere upon repeated exposure to the offending medication.
The differential diagnosis includes arthropod bite reactions, urticaria, and erythema multiforme. Although FDEs are typically a clinical diagnosis, the histopathology will commonly show a vacuolar interface dermatitis. Furthermore, a variety of immune cells can be found, including neutrophilic, eosinophilic, and lymphocytic infiltrate. A combination of two or more histological patterns often favors the diagnosis of FDE.
Steroid creams can be prescribed to decrease the inflammatory reaction and improve symptoms; however, the definitive treatment of this condition is cessation of the offending agent. Postinflammatory hyperpigmentation is a common symptom after resolution of the condition, and it may take months to fade away. Further darkening can be prevented by practicing sun safety measures such as wearing sunblock, covering the affected areas, and avoiding prolonged sun exposure.
This case and the photos were submitted by Lucas Shapiro, BS, of Nova Southeastern University College of Osteopathic Medicine, Fort Lauderdale, Fla., and Igor Chaplik, DO, Aesthetix Dermatology, Fort Lauderdale. The column was edited by Donna Bilu Martin, MD.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to [email protected].
References
Shaker G et al. Cureus. 2022 Aug 23;14(8):e28299.
Srivastava R et al. Indian J Dent. 2015 Apr-Jun;6(2):103-6.
Weyers W, Metze D. Dermatol Pract Concept. 2011 Jan 31;1(1):33-47.
Patch testing finds higher prevalence of ACD among children with AD
, a finding that investigators say underscores the value of considering ACD in patients with AD and referring more children for testing.
ACD is underdetected in children with AD. In some cases, it may be misconstrued to be AD, and patch testing, the gold standard for diagnosing ACD, is often not performed, said senior author JiaDe Yu, MD, MS, a pediatric dermatologist and director of contact and occupational dermatology at Massachusetts General Hospital, Boston, and his co-authors, in the study published in the Journal of the American Academy of Dermatology.
Dr. Yu and his colleagues utilized a database in which dermatologists and some allergists, all of whom had substantive experience in patch testing and in diagnosing and managing ACD in children, entered information about children who were referred to them for testing.
Of 912 children referred for patch testing between 2018 and 2022 from 14 geographically diverse centers in the United States (615 with AD and 297 without AD), those with AD were more likely to have more than one positive reaction (odds radio, 1.57; 95% confidence interval, 1.14-2.14; P = .005) and had a greater number of positive results overall (2.3 vs. 1.9; P = .012).
AD and ACD both present with red, itchy, eczema-like patches and plaques and can be “really hard to differentiate,” Dr. Yu said in an interview.
“Not everybody with AD needs patch testing,” he said, “but I do think some [patients] who have rashes in unusual locations or rashes that don’t seem to improve within an appropriate amount of time to topical medications ... are the children who probably should have patch testing.”
Candidates for patch testing include children with AD who present with isolated head or neck, hand or foot, or anal or genital dermatitis, Dr. Yu and his colleagues write in the study. In addition, Dr. Yu said in the interview, “if you have a child who has AD that involves the elbow and back of the knees but then they get new-onset facial dermatitis, say, or new-onset eyelid dermatitis ... there’s [significant] value in patch testing.”
Children with AD in the study had a more generalized distribution of dermatitis and were significantly less likely to have dermatitis affecting the anal or genital region, the authors note in the study.
Asked to comment on the results, Jennifer Perryman, MD, a dermatologist at UCHealth, Greeley, Colo., who performs patch testing in children and adults, said that ACD is indeed “often underdiagnosed” in children with AD, and the study “solidifies” the importance of considering ACD in this population.
“Clinicians should think about testing children when AD is [not well controlled or] is getting worse, is in an atypical distribution, or if they are considering systemic treatment,” she said in an e-mail.
“I tell my patients, ‘I know you have AD, but you could also have comorbid ACD, and if we can find and control that, we can make you better without adding more to your routine, medications, etc.’ ” said Dr. Perryman, who was not involved in the research.
Top allergens
The top 10 allergens between children with and without AD were largely similar, the authors of the study report. Nickel was the most common allergen identified in both groups, and cobalt was in the top five for both groups. Fragrances (including hydroperoxides of linalool), preservatives (including methylisothiazolinone [MI]), and neomycin ranked in the top 10 in both groups, though prevalence differed.
MI, a preservative frequently used in personal care products and in other products like school glue and paint, was the second most common allergen identified in children with AD. Allergy to MI has “recently become an epidemic in the United States, with rapidly increasing prevalence and importance as a source of ACD among both children and adults,” the authors note.
Children with AD were significantly more likely, however, to have ACD to bacitracin (OR, 3.23; P = .030) and to cocamidopropyl betaine (OR, 3.69; P = .0007), the latter of which is a popular surfactant used in “baby” and “gentle” skincare products. This is unsurprising, given that children with AD are “more often exposed to a myriad of topical treatments,” Dr. Yu and his colleagues write.
Although not a top 10 allergen for either group, ACD to “carba mix,” a combination of three chemicals used to make medical adhesives and other rubber products (such as pacifiers, toys, school supplies, and rubber gloves) was significantly more common in children with AD than in those without (OR, 3.36; P = .025).
Among other findings from the study: Children with AD were more likely to have a longer history of dermatitis (4.1 vs. 1.6 years, P < .0001) prior to patch testing. Testing occurred at a mean age of 11 and 12.3 years for children with and without AD, respectively.
The number of allergens tested and the patch testing series chosen per patient were “not statistically different” between the children with and without AD, the researchers report.
Patch testing availability
Clinicians may be hesitant to subject a child to patch testing, but the process is well tolerated in most children, Dr. Perryman said. She uses a modified panel for children that omits less relevant allergens and usually limits patch testing to age 2 years or older due to a young child’s smaller surface area.
Dr. Yu, who developed an interest in patch testing during his residency at the Medical College of Wisconsin, Milwaukee, where he worked with a patch-testing expert, will test children as young as 3-4 months with a “small selection of patches.”
The challenge with a call for more patch testing is a shortage of trained physicians. “In all of Boston, where we have hundreds of dermatologists, there are only about four of us who really do patch testing. My wait time is about 6 months,” said Dr. Yu, who is also an assistant professor at Harvard Medical School, Boston.
Allergists at Massachusetts General Hospital do “some patch testing ... but they refer a lot of the most complicated cases to me,” he said, noting that patch testing and management of ACD involves detailed counseling for patients about avoidance of allergens. “Overall dermatologists represent the largest group of doctors who have proficiency in patch testing, and there just aren’t many of us.”
Dr. Perryman also said that patch testing is often performed by dermatologists who specialize in treating ACD and AD, though there seems to be “regional variance” in the level of involvement of dermatologists and allergists in patch testing.
Not all residency programs have hands-on patch testing opportunities, Dr. Yu said. A study published in Dermatitis, which he co-authored, showed that in 2020, 47.5% of dermatology residency programs had formal patch testing rotations. This represented improvement but is still not enough, he said.
The American Contact Dermatitis Society offers patch-testing mentorship programs, and the American Academy of Dermatology has recently begun offered a patch testing workshop at its annual meetings, said Dr. Yu, who received 4 weeks of training in the Society’s mentorship program and is now involved in the American Academy of Dermatology’s workshops and as a trainer/lecturer at the Contact Dermatitis Institute.
The study was supported by the Dermatology Foundation. Dr. Yu and his co-investigators reported no conflicts of interest. Dr. Perryman had no disclosures.
A version of this article first appeared on Medscape.com.
, a finding that investigators say underscores the value of considering ACD in patients with AD and referring more children for testing.
ACD is underdetected in children with AD. In some cases, it may be misconstrued to be AD, and patch testing, the gold standard for diagnosing ACD, is often not performed, said senior author JiaDe Yu, MD, MS, a pediatric dermatologist and director of contact and occupational dermatology at Massachusetts General Hospital, Boston, and his co-authors, in the study published in the Journal of the American Academy of Dermatology.
Dr. Yu and his colleagues utilized a database in which dermatologists and some allergists, all of whom had substantive experience in patch testing and in diagnosing and managing ACD in children, entered information about children who were referred to them for testing.
Of 912 children referred for patch testing between 2018 and 2022 from 14 geographically diverse centers in the United States (615 with AD and 297 without AD), those with AD were more likely to have more than one positive reaction (odds radio, 1.57; 95% confidence interval, 1.14-2.14; P = .005) and had a greater number of positive results overall (2.3 vs. 1.9; P = .012).
AD and ACD both present with red, itchy, eczema-like patches and plaques and can be “really hard to differentiate,” Dr. Yu said in an interview.
“Not everybody with AD needs patch testing,” he said, “but I do think some [patients] who have rashes in unusual locations or rashes that don’t seem to improve within an appropriate amount of time to topical medications ... are the children who probably should have patch testing.”
Candidates for patch testing include children with AD who present with isolated head or neck, hand or foot, or anal or genital dermatitis, Dr. Yu and his colleagues write in the study. In addition, Dr. Yu said in the interview, “if you have a child who has AD that involves the elbow and back of the knees but then they get new-onset facial dermatitis, say, or new-onset eyelid dermatitis ... there’s [significant] value in patch testing.”
Children with AD in the study had a more generalized distribution of dermatitis and were significantly less likely to have dermatitis affecting the anal or genital region, the authors note in the study.
Asked to comment on the results, Jennifer Perryman, MD, a dermatologist at UCHealth, Greeley, Colo., who performs patch testing in children and adults, said that ACD is indeed “often underdiagnosed” in children with AD, and the study “solidifies” the importance of considering ACD in this population.
“Clinicians should think about testing children when AD is [not well controlled or] is getting worse, is in an atypical distribution, or if they are considering systemic treatment,” she said in an e-mail.
“I tell my patients, ‘I know you have AD, but you could also have comorbid ACD, and if we can find and control that, we can make you better without adding more to your routine, medications, etc.’ ” said Dr. Perryman, who was not involved in the research.
Top allergens
The top 10 allergens between children with and without AD were largely similar, the authors of the study report. Nickel was the most common allergen identified in both groups, and cobalt was in the top five for both groups. Fragrances (including hydroperoxides of linalool), preservatives (including methylisothiazolinone [MI]), and neomycin ranked in the top 10 in both groups, though prevalence differed.
MI, a preservative frequently used in personal care products and in other products like school glue and paint, was the second most common allergen identified in children with AD. Allergy to MI has “recently become an epidemic in the United States, with rapidly increasing prevalence and importance as a source of ACD among both children and adults,” the authors note.
Children with AD were significantly more likely, however, to have ACD to bacitracin (OR, 3.23; P = .030) and to cocamidopropyl betaine (OR, 3.69; P = .0007), the latter of which is a popular surfactant used in “baby” and “gentle” skincare products. This is unsurprising, given that children with AD are “more often exposed to a myriad of topical treatments,” Dr. Yu and his colleagues write.
Although not a top 10 allergen for either group, ACD to “carba mix,” a combination of three chemicals used to make medical adhesives and other rubber products (such as pacifiers, toys, school supplies, and rubber gloves) was significantly more common in children with AD than in those without (OR, 3.36; P = .025).
Among other findings from the study: Children with AD were more likely to have a longer history of dermatitis (4.1 vs. 1.6 years, P < .0001) prior to patch testing. Testing occurred at a mean age of 11 and 12.3 years for children with and without AD, respectively.
The number of allergens tested and the patch testing series chosen per patient were “not statistically different” between the children with and without AD, the researchers report.
Patch testing availability
Clinicians may be hesitant to subject a child to patch testing, but the process is well tolerated in most children, Dr. Perryman said. She uses a modified panel for children that omits less relevant allergens and usually limits patch testing to age 2 years or older due to a young child’s smaller surface area.
Dr. Yu, who developed an interest in patch testing during his residency at the Medical College of Wisconsin, Milwaukee, where he worked with a patch-testing expert, will test children as young as 3-4 months with a “small selection of patches.”
The challenge with a call for more patch testing is a shortage of trained physicians. “In all of Boston, where we have hundreds of dermatologists, there are only about four of us who really do patch testing. My wait time is about 6 months,” said Dr. Yu, who is also an assistant professor at Harvard Medical School, Boston.
Allergists at Massachusetts General Hospital do “some patch testing ... but they refer a lot of the most complicated cases to me,” he said, noting that patch testing and management of ACD involves detailed counseling for patients about avoidance of allergens. “Overall dermatologists represent the largest group of doctors who have proficiency in patch testing, and there just aren’t many of us.”
Dr. Perryman also said that patch testing is often performed by dermatologists who specialize in treating ACD and AD, though there seems to be “regional variance” in the level of involvement of dermatologists and allergists in patch testing.
Not all residency programs have hands-on patch testing opportunities, Dr. Yu said. A study published in Dermatitis, which he co-authored, showed that in 2020, 47.5% of dermatology residency programs had formal patch testing rotations. This represented improvement but is still not enough, he said.
The American Contact Dermatitis Society offers patch-testing mentorship programs, and the American Academy of Dermatology has recently begun offered a patch testing workshop at its annual meetings, said Dr. Yu, who received 4 weeks of training in the Society’s mentorship program and is now involved in the American Academy of Dermatology’s workshops and as a trainer/lecturer at the Contact Dermatitis Institute.
The study was supported by the Dermatology Foundation. Dr. Yu and his co-investigators reported no conflicts of interest. Dr. Perryman had no disclosures.
A version of this article first appeared on Medscape.com.
, a finding that investigators say underscores the value of considering ACD in patients with AD and referring more children for testing.
ACD is underdetected in children with AD. In some cases, it may be misconstrued to be AD, and patch testing, the gold standard for diagnosing ACD, is often not performed, said senior author JiaDe Yu, MD, MS, a pediatric dermatologist and director of contact and occupational dermatology at Massachusetts General Hospital, Boston, and his co-authors, in the study published in the Journal of the American Academy of Dermatology.
Dr. Yu and his colleagues utilized a database in which dermatologists and some allergists, all of whom had substantive experience in patch testing and in diagnosing and managing ACD in children, entered information about children who were referred to them for testing.
Of 912 children referred for patch testing between 2018 and 2022 from 14 geographically diverse centers in the United States (615 with AD and 297 without AD), those with AD were more likely to have more than one positive reaction (odds radio, 1.57; 95% confidence interval, 1.14-2.14; P = .005) and had a greater number of positive results overall (2.3 vs. 1.9; P = .012).
AD and ACD both present with red, itchy, eczema-like patches and plaques and can be “really hard to differentiate,” Dr. Yu said in an interview.
“Not everybody with AD needs patch testing,” he said, “but I do think some [patients] who have rashes in unusual locations or rashes that don’t seem to improve within an appropriate amount of time to topical medications ... are the children who probably should have patch testing.”
Candidates for patch testing include children with AD who present with isolated head or neck, hand or foot, or anal or genital dermatitis, Dr. Yu and his colleagues write in the study. In addition, Dr. Yu said in the interview, “if you have a child who has AD that involves the elbow and back of the knees but then they get new-onset facial dermatitis, say, or new-onset eyelid dermatitis ... there’s [significant] value in patch testing.”
Children with AD in the study had a more generalized distribution of dermatitis and were significantly less likely to have dermatitis affecting the anal or genital region, the authors note in the study.
Asked to comment on the results, Jennifer Perryman, MD, a dermatologist at UCHealth, Greeley, Colo., who performs patch testing in children and adults, said that ACD is indeed “often underdiagnosed” in children with AD, and the study “solidifies” the importance of considering ACD in this population.
“Clinicians should think about testing children when AD is [not well controlled or] is getting worse, is in an atypical distribution, or if they are considering systemic treatment,” she said in an e-mail.
“I tell my patients, ‘I know you have AD, but you could also have comorbid ACD, and if we can find and control that, we can make you better without adding more to your routine, medications, etc.’ ” said Dr. Perryman, who was not involved in the research.
Top allergens
The top 10 allergens between children with and without AD were largely similar, the authors of the study report. Nickel was the most common allergen identified in both groups, and cobalt was in the top five for both groups. Fragrances (including hydroperoxides of linalool), preservatives (including methylisothiazolinone [MI]), and neomycin ranked in the top 10 in both groups, though prevalence differed.
MI, a preservative frequently used in personal care products and in other products like school glue and paint, was the second most common allergen identified in children with AD. Allergy to MI has “recently become an epidemic in the United States, with rapidly increasing prevalence and importance as a source of ACD among both children and adults,” the authors note.
Children with AD were significantly more likely, however, to have ACD to bacitracin (OR, 3.23; P = .030) and to cocamidopropyl betaine (OR, 3.69; P = .0007), the latter of which is a popular surfactant used in “baby” and “gentle” skincare products. This is unsurprising, given that children with AD are “more often exposed to a myriad of topical treatments,” Dr. Yu and his colleagues write.
Although not a top 10 allergen for either group, ACD to “carba mix,” a combination of three chemicals used to make medical adhesives and other rubber products (such as pacifiers, toys, school supplies, and rubber gloves) was significantly more common in children with AD than in those without (OR, 3.36; P = .025).
Among other findings from the study: Children with AD were more likely to have a longer history of dermatitis (4.1 vs. 1.6 years, P < .0001) prior to patch testing. Testing occurred at a mean age of 11 and 12.3 years for children with and without AD, respectively.
The number of allergens tested and the patch testing series chosen per patient were “not statistically different” between the children with and without AD, the researchers report.
Patch testing availability
Clinicians may be hesitant to subject a child to patch testing, but the process is well tolerated in most children, Dr. Perryman said. She uses a modified panel for children that omits less relevant allergens and usually limits patch testing to age 2 years or older due to a young child’s smaller surface area.
Dr. Yu, who developed an interest in patch testing during his residency at the Medical College of Wisconsin, Milwaukee, where he worked with a patch-testing expert, will test children as young as 3-4 months with a “small selection of patches.”
The challenge with a call for more patch testing is a shortage of trained physicians. “In all of Boston, where we have hundreds of dermatologists, there are only about four of us who really do patch testing. My wait time is about 6 months,” said Dr. Yu, who is also an assistant professor at Harvard Medical School, Boston.
Allergists at Massachusetts General Hospital do “some patch testing ... but they refer a lot of the most complicated cases to me,” he said, noting that patch testing and management of ACD involves detailed counseling for patients about avoidance of allergens. “Overall dermatologists represent the largest group of doctors who have proficiency in patch testing, and there just aren’t many of us.”
Dr. Perryman also said that patch testing is often performed by dermatologists who specialize in treating ACD and AD, though there seems to be “regional variance” in the level of involvement of dermatologists and allergists in patch testing.
Not all residency programs have hands-on patch testing opportunities, Dr. Yu said. A study published in Dermatitis, which he co-authored, showed that in 2020, 47.5% of dermatology residency programs had formal patch testing rotations. This represented improvement but is still not enough, he said.
The American Contact Dermatitis Society offers patch-testing mentorship programs, and the American Academy of Dermatology has recently begun offered a patch testing workshop at its annual meetings, said Dr. Yu, who received 4 weeks of training in the Society’s mentorship program and is now involved in the American Academy of Dermatology’s workshops and as a trainer/lecturer at the Contact Dermatitis Institute.
The study was supported by the Dermatology Foundation. Dr. Yu and his co-investigators reported no conflicts of interest. Dr. Perryman had no disclosures.
A version of this article first appeared on Medscape.com.
FROM THE JOURNAL OF THE AMERICAN ACADEMY OF DERMATOLOGY
Intravenous formulation of secukinumab gets FDA approval
The Food and Drug Administration has approved an intravenous (IV) formulation of secukinumab (Cosentyx) for the treatment of adults with psoriatic arthritis (PsA), ankylosing spondylitis (AS), and nonradiographic axial spondyloarthritis (nr-axSpA).
Secukinumab is the only treatment approved in an IV formulation that specifically targets and blocks interleukin-17A and the only non–tumor necrosis factor alpha IV option available to treat the three indications of PsA, AS, and nr-axSpA, according to a press release from the drug’s manufacturer, Novartis.
The approval marks the first new IV treatment in 6 years for these three conditions. The drug was first approved in 2015 and up to now has been available only as a subcutaneous injection.
The new formulation is also approved for secukinumab’s other indications of plaque psoriasis in people aged 6 years or older, children aged 2 years or older with PsA, and enthesitis-related arthritis in patients aged 4 years or older.
“A significant portion of the millions of PsA, AS, and nr-axSpA patients in the United States require treatment through IV infusions for a variety of reasons, including not being comfortable with self-injections or simply preferring to have treatments administered in their health care provider’s office,” Philip J. Mease, MD, clinical professor at the University of Washington, Seattle, and director of rheumatology research at the Swedish Medical Center, Seattle, said in the press release. “The approval of Cosentyx as an IV formulation is an important milestone for patients because it expands the treatment options available to them with a different mechanism of action than existing biologic IV therapies, along with the comfort and familiarity of an established treatment.”
This IV formulation is administered monthly in a 30-minute, weight-based dosing regimen. This new option will become available before the end of the year, Novartis said.
“With this approval of Cosentyx as an IV formulation, along with the subcutaneous formulation, we can broaden the use of Cosentyx to help more patients manage their condition with a medicine backed by more than a decade of clinical research and 8 years of real-world experience,” said Christy Siegel, vice president and head of immunology, Novartis U.S.
A version of this article first appeared on Medscape.com.
The Food and Drug Administration has approved an intravenous (IV) formulation of secukinumab (Cosentyx) for the treatment of adults with psoriatic arthritis (PsA), ankylosing spondylitis (AS), and nonradiographic axial spondyloarthritis (nr-axSpA).
Secukinumab is the only treatment approved in an IV formulation that specifically targets and blocks interleukin-17A and the only non–tumor necrosis factor alpha IV option available to treat the three indications of PsA, AS, and nr-axSpA, according to a press release from the drug’s manufacturer, Novartis.
The approval marks the first new IV treatment in 6 years for these three conditions. The drug was first approved in 2015 and up to now has been available only as a subcutaneous injection.
The new formulation is also approved for secukinumab’s other indications of plaque psoriasis in people aged 6 years or older, children aged 2 years or older with PsA, and enthesitis-related arthritis in patients aged 4 years or older.
“A significant portion of the millions of PsA, AS, and nr-axSpA patients in the United States require treatment through IV infusions for a variety of reasons, including not being comfortable with self-injections or simply preferring to have treatments administered in their health care provider’s office,” Philip J. Mease, MD, clinical professor at the University of Washington, Seattle, and director of rheumatology research at the Swedish Medical Center, Seattle, said in the press release. “The approval of Cosentyx as an IV formulation is an important milestone for patients because it expands the treatment options available to them with a different mechanism of action than existing biologic IV therapies, along with the comfort and familiarity of an established treatment.”
This IV formulation is administered monthly in a 30-minute, weight-based dosing regimen. This new option will become available before the end of the year, Novartis said.
“With this approval of Cosentyx as an IV formulation, along with the subcutaneous formulation, we can broaden the use of Cosentyx to help more patients manage their condition with a medicine backed by more than a decade of clinical research and 8 years of real-world experience,” said Christy Siegel, vice president and head of immunology, Novartis U.S.
A version of this article first appeared on Medscape.com.
The Food and Drug Administration has approved an intravenous (IV) formulation of secukinumab (Cosentyx) for the treatment of adults with psoriatic arthritis (PsA), ankylosing spondylitis (AS), and nonradiographic axial spondyloarthritis (nr-axSpA).
Secukinumab is the only treatment approved in an IV formulation that specifically targets and blocks interleukin-17A and the only non–tumor necrosis factor alpha IV option available to treat the three indications of PsA, AS, and nr-axSpA, according to a press release from the drug’s manufacturer, Novartis.
The approval marks the first new IV treatment in 6 years for these three conditions. The drug was first approved in 2015 and up to now has been available only as a subcutaneous injection.
The new formulation is also approved for secukinumab’s other indications of plaque psoriasis in people aged 6 years or older, children aged 2 years or older with PsA, and enthesitis-related arthritis in patients aged 4 years or older.
“A significant portion of the millions of PsA, AS, and nr-axSpA patients in the United States require treatment through IV infusions for a variety of reasons, including not being comfortable with self-injections or simply preferring to have treatments administered in their health care provider’s office,” Philip J. Mease, MD, clinical professor at the University of Washington, Seattle, and director of rheumatology research at the Swedish Medical Center, Seattle, said in the press release. “The approval of Cosentyx as an IV formulation is an important milestone for patients because it expands the treatment options available to them with a different mechanism of action than existing biologic IV therapies, along with the comfort and familiarity of an established treatment.”
This IV formulation is administered monthly in a 30-minute, weight-based dosing regimen. This new option will become available before the end of the year, Novartis said.
“With this approval of Cosentyx as an IV formulation, along with the subcutaneous formulation, we can broaden the use of Cosentyx to help more patients manage their condition with a medicine backed by more than a decade of clinical research and 8 years of real-world experience,” said Christy Siegel, vice president and head of immunology, Novartis U.S.
A version of this article first appeared on Medscape.com.