User login
Can Popular Weight-Loss Drugs Protect Against Obesity-Related Cancers?
Can Popular Weight-Loss Drugs Protect Against Obesity-Related Cancers?
New data suggest that glucagon-like peptide 1 (GLP-1) receptor agonists, used to treat diabetes and obesity, may also help guard against obesity-related cancers.
In a large observational study, new GLP-1 agonist users with obesity and diabetes had a significantly lower risk for 14 obesity-related cancers than similar individuals who received dipeptidyl peptidase-4 (DPP-4) inhibitors, which are weight-neutral.
This study provides a “reassuring safety signal” showing that GLP-1 drugs are linked to a modest drop in obesity-related cancer risk, and not a higher risk for these cancers, said lead investigator Lucas Mavromatis, medical student at NYU Grossman School of Medicine in New York City, during a press conference at American Society of Clinical Oncology (ASCO) 2025 annual meeting.
However, there were some nuances to the findings. The protective effect of GLP-1 agonists was only significant for colon and rectal cancers and for women, Mavromatis reported. And although GLP-1 users had an 8% lower risk of dying from any cause, the survival benefit was also only significant for women.
Still, the overall “message to patients is GLP-1 receptor treatments remain a strong option for patients with diabetes and obesity and may have an additional, small favorable benefit in cancer,” Mavromatis explained at the press briefing.
'Intriguing Hypothesis'
Obesity is linked to an increased risk of developing more than a dozen cancer types, including esophageal, colon, rectal, stomach, liver, gallbladder, pancreatic, kidney, postmenopausal breast, ovarian, endometrial and thyroid, as well as multiple myeloma and meningiomas.
About 12% of Americans have been prescribed a GLP-1 medication to treat diabetes and/or obesity. However, little is known about how these drugs affect cancer risk.
To investigate, Mavromatis and colleagues used the Optum healthcare database to identify 170,030 adults with obesity and type 2 diabetes from 43 health systems in the United States.
Between 2013 and 2023, half started a GLP-1 agonist and half started a DPP-4 inhibitor, with propensity score matching used to balance characteristics of the two cohorts.
Participants were a mean age of 56.8 years, with an average body mass index of 38.5; more than 70% were White individuals and more than 14% were Black individuals.
During a mean follow-up of 3.9 years, 2501 new obesity-related cancers were identified in the GLP-1 group and 2671 in the DPP-4 group — representing a 7% overall reduced risk for any obesity-related cancer in the GLP-1 group (hazard ratio [HR], 0.93).
When analyzing each of the 14 obesity-related cancers separately, the protective link between GLP-1 use and cancer was primarily driven by colon and rectal cancers. GLP-1 users had a 16% lower risk for colon cancer (HR, 0.84) and a 28% lower risk for rectal cancer (HR, 0.72).
“No other cancers had statistically significant associations with GLP-1 use,” Mavromatis told briefing attendees. But “importantly, no cancers had statistically significant adverse associations with GLP-1 use,” he added.
Experts have expressed some concern about a possible link between GLP-1 use and pancreatic cancer given that pancreatitis is a known side effect of GLP-1 use. However, “this is not borne out by epidemiological data,” Mavromatis said.
“Additionally, we were not able to specifically assess medullary thyroid cancer, which is on the warning label for several GLP-1 medications, but we did see a reassuring lack of association between GLP-1 use and thyroid cancer as a whole,” he added.
During follow-up, there were 2783 deaths in the GLP-1 group and 2961 deaths in the DPP-4 group — translating to an 8% lower risk for death due to any cause among GLP-1 users (HR, 0.92; P = .001).
Mavromatis and colleagues observed sex differences as well. Women taking a GLP-1 had an 8% lower risk for obesity-related cancers (HR, 0.92; P = .01) and a 20% lower risk for death from any cause (HR, 0.80; P < .001) compared with women taking a DPP-4 inhibitor.
Among men, researchers found no statistically significant difference between GLP-1 and DPP-4 use for obesity-related cancer risk (HR, 0.95; P = .29) or all-cause mortality (HR, 1.04; P = .34).
Overall, Mavromatis said, it’s important to note that the absolute risk reduction seen in the study is “small and the number of patients that would need to be given one of these medications to prevent an obesity-related cancer, based on our data, would be very large.”
Mavromatis also noted that the length of follow-up was short, and the study assessed primarily older and weaker GLP-1 agonists compared with newer agents on the market. Therefore, longer-term studies with newer GLP-1s are needed to confirm the effects seen as well as safety.
In a statement, ASCO President Robin Zon, MD, said this trial raises the “intriguing hypothesis” that the increasingly popular GLP-1 medications might offer some benefit in reducing the risk of developing cancer.
Zon said she sees many patients with obesity, and given the clear link between cancer and obesity, defining the clinical role of GLP-1 medications in cancer prevention is “important.”
This study “leads us in the direction” of a potential protective effect of GLP-1s on cancer, but “there are a lot of questions that are generated by this particular study, especially as we move forward and we think about prevention of cancers,” Zon told the briefing.
This study was funded by the National Institute of Diabetes and Digestive and Kidney Diseases of the National Institutes of Health. Mavromatis reported no relevant disclosures. Zon reported stock or ownership interests in Oncolytics Biotech, TG Therapeutics, Select Sector SPDR Health Care, AstraZeneca, CRISPR, McKesson, and Berkshire Hathaway.
A version of this article first appeared on Medscape.com.
New data suggest that glucagon-like peptide 1 (GLP-1) receptor agonists, used to treat diabetes and obesity, may also help guard against obesity-related cancers.
In a large observational study, new GLP-1 agonist users with obesity and diabetes had a significantly lower risk for 14 obesity-related cancers than similar individuals who received dipeptidyl peptidase-4 (DPP-4) inhibitors, which are weight-neutral.
This study provides a “reassuring safety signal” showing that GLP-1 drugs are linked to a modest drop in obesity-related cancer risk, and not a higher risk for these cancers, said lead investigator Lucas Mavromatis, medical student at NYU Grossman School of Medicine in New York City, during a press conference at American Society of Clinical Oncology (ASCO) 2025 annual meeting.
However, there were some nuances to the findings. The protective effect of GLP-1 agonists was only significant for colon and rectal cancers and for women, Mavromatis reported. And although GLP-1 users had an 8% lower risk of dying from any cause, the survival benefit was also only significant for women.
Still, the overall “message to patients is GLP-1 receptor treatments remain a strong option for patients with diabetes and obesity and may have an additional, small favorable benefit in cancer,” Mavromatis explained at the press briefing.
'Intriguing Hypothesis'
Obesity is linked to an increased risk of developing more than a dozen cancer types, including esophageal, colon, rectal, stomach, liver, gallbladder, pancreatic, kidney, postmenopausal breast, ovarian, endometrial and thyroid, as well as multiple myeloma and meningiomas.
About 12% of Americans have been prescribed a GLP-1 medication to treat diabetes and/or obesity. However, little is known about how these drugs affect cancer risk.
To investigate, Mavromatis and colleagues used the Optum healthcare database to identify 170,030 adults with obesity and type 2 diabetes from 43 health systems in the United States.
Between 2013 and 2023, half started a GLP-1 agonist and half started a DPP-4 inhibitor, with propensity score matching used to balance characteristics of the two cohorts.
Participants were a mean age of 56.8 years, with an average body mass index of 38.5; more than 70% were White individuals and more than 14% were Black individuals.
During a mean follow-up of 3.9 years, 2501 new obesity-related cancers were identified in the GLP-1 group and 2671 in the DPP-4 group — representing a 7% overall reduced risk for any obesity-related cancer in the GLP-1 group (hazard ratio [HR], 0.93).
When analyzing each of the 14 obesity-related cancers separately, the protective link between GLP-1 use and cancer was primarily driven by colon and rectal cancers. GLP-1 users had a 16% lower risk for colon cancer (HR, 0.84) and a 28% lower risk for rectal cancer (HR, 0.72).
“No other cancers had statistically significant associations with GLP-1 use,” Mavromatis told briefing attendees. But “importantly, no cancers had statistically significant adverse associations with GLP-1 use,” he added.
Experts have expressed some concern about a possible link between GLP-1 use and pancreatic cancer given that pancreatitis is a known side effect of GLP-1 use. However, “this is not borne out by epidemiological data,” Mavromatis said.
“Additionally, we were not able to specifically assess medullary thyroid cancer, which is on the warning label for several GLP-1 medications, but we did see a reassuring lack of association between GLP-1 use and thyroid cancer as a whole,” he added.
During follow-up, there were 2783 deaths in the GLP-1 group and 2961 deaths in the DPP-4 group — translating to an 8% lower risk for death due to any cause among GLP-1 users (HR, 0.92; P = .001).
Mavromatis and colleagues observed sex differences as well. Women taking a GLP-1 had an 8% lower risk for obesity-related cancers (HR, 0.92; P = .01) and a 20% lower risk for death from any cause (HR, 0.80; P < .001) compared with women taking a DPP-4 inhibitor.
Among men, researchers found no statistically significant difference between GLP-1 and DPP-4 use for obesity-related cancer risk (HR, 0.95; P = .29) or all-cause mortality (HR, 1.04; P = .34).
Overall, Mavromatis said, it’s important to note that the absolute risk reduction seen in the study is “small and the number of patients that would need to be given one of these medications to prevent an obesity-related cancer, based on our data, would be very large.”
Mavromatis also noted that the length of follow-up was short, and the study assessed primarily older and weaker GLP-1 agonists compared with newer agents on the market. Therefore, longer-term studies with newer GLP-1s are needed to confirm the effects seen as well as safety.
In a statement, ASCO President Robin Zon, MD, said this trial raises the “intriguing hypothesis” that the increasingly popular GLP-1 medications might offer some benefit in reducing the risk of developing cancer.
Zon said she sees many patients with obesity, and given the clear link between cancer and obesity, defining the clinical role of GLP-1 medications in cancer prevention is “important.”
This study “leads us in the direction” of a potential protective effect of GLP-1s on cancer, but “there are a lot of questions that are generated by this particular study, especially as we move forward and we think about prevention of cancers,” Zon told the briefing.
This study was funded by the National Institute of Diabetes and Digestive and Kidney Diseases of the National Institutes of Health. Mavromatis reported no relevant disclosures. Zon reported stock or ownership interests in Oncolytics Biotech, TG Therapeutics, Select Sector SPDR Health Care, AstraZeneca, CRISPR, McKesson, and Berkshire Hathaway.
A version of this article first appeared on Medscape.com.
New data suggest that glucagon-like peptide 1 (GLP-1) receptor agonists, used to treat diabetes and obesity, may also help guard against obesity-related cancers.
In a large observational study, new GLP-1 agonist users with obesity and diabetes had a significantly lower risk for 14 obesity-related cancers than similar individuals who received dipeptidyl peptidase-4 (DPP-4) inhibitors, which are weight-neutral.
This study provides a “reassuring safety signal” showing that GLP-1 drugs are linked to a modest drop in obesity-related cancer risk, and not a higher risk for these cancers, said lead investigator Lucas Mavromatis, medical student at NYU Grossman School of Medicine in New York City, during a press conference at American Society of Clinical Oncology (ASCO) 2025 annual meeting.
However, there were some nuances to the findings. The protective effect of GLP-1 agonists was only significant for colon and rectal cancers and for women, Mavromatis reported. And although GLP-1 users had an 8% lower risk of dying from any cause, the survival benefit was also only significant for women.
Still, the overall “message to patients is GLP-1 receptor treatments remain a strong option for patients with diabetes and obesity and may have an additional, small favorable benefit in cancer,” Mavromatis explained at the press briefing.
'Intriguing Hypothesis'
Obesity is linked to an increased risk of developing more than a dozen cancer types, including esophageal, colon, rectal, stomach, liver, gallbladder, pancreatic, kidney, postmenopausal breast, ovarian, endometrial and thyroid, as well as multiple myeloma and meningiomas.
About 12% of Americans have been prescribed a GLP-1 medication to treat diabetes and/or obesity. However, little is known about how these drugs affect cancer risk.
To investigate, Mavromatis and colleagues used the Optum healthcare database to identify 170,030 adults with obesity and type 2 diabetes from 43 health systems in the United States.
Between 2013 and 2023, half started a GLP-1 agonist and half started a DPP-4 inhibitor, with propensity score matching used to balance characteristics of the two cohorts.
Participants were a mean age of 56.8 years, with an average body mass index of 38.5; more than 70% were White individuals and more than 14% were Black individuals.
During a mean follow-up of 3.9 years, 2501 new obesity-related cancers were identified in the GLP-1 group and 2671 in the DPP-4 group — representing a 7% overall reduced risk for any obesity-related cancer in the GLP-1 group (hazard ratio [HR], 0.93).
When analyzing each of the 14 obesity-related cancers separately, the protective link between GLP-1 use and cancer was primarily driven by colon and rectal cancers. GLP-1 users had a 16% lower risk for colon cancer (HR, 0.84) and a 28% lower risk for rectal cancer (HR, 0.72).
“No other cancers had statistically significant associations with GLP-1 use,” Mavromatis told briefing attendees. But “importantly, no cancers had statistically significant adverse associations with GLP-1 use,” he added.
Experts have expressed some concern about a possible link between GLP-1 use and pancreatic cancer given that pancreatitis is a known side effect of GLP-1 use. However, “this is not borne out by epidemiological data,” Mavromatis said.
“Additionally, we were not able to specifically assess medullary thyroid cancer, which is on the warning label for several GLP-1 medications, but we did see a reassuring lack of association between GLP-1 use and thyroid cancer as a whole,” he added.
During follow-up, there were 2783 deaths in the GLP-1 group and 2961 deaths in the DPP-4 group — translating to an 8% lower risk for death due to any cause among GLP-1 users (HR, 0.92; P = .001).
Mavromatis and colleagues observed sex differences as well. Women taking a GLP-1 had an 8% lower risk for obesity-related cancers (HR, 0.92; P = .01) and a 20% lower risk for death from any cause (HR, 0.80; P < .001) compared with women taking a DPP-4 inhibitor.
Among men, researchers found no statistically significant difference between GLP-1 and DPP-4 use for obesity-related cancer risk (HR, 0.95; P = .29) or all-cause mortality (HR, 1.04; P = .34).
Overall, Mavromatis said, it’s important to note that the absolute risk reduction seen in the study is “small and the number of patients that would need to be given one of these medications to prevent an obesity-related cancer, based on our data, would be very large.”
Mavromatis also noted that the length of follow-up was short, and the study assessed primarily older and weaker GLP-1 agonists compared with newer agents on the market. Therefore, longer-term studies with newer GLP-1s are needed to confirm the effects seen as well as safety.
In a statement, ASCO President Robin Zon, MD, said this trial raises the “intriguing hypothesis” that the increasingly popular GLP-1 medications might offer some benefit in reducing the risk of developing cancer.
Zon said she sees many patients with obesity, and given the clear link between cancer and obesity, defining the clinical role of GLP-1 medications in cancer prevention is “important.”
This study “leads us in the direction” of a potential protective effect of GLP-1s on cancer, but “there are a lot of questions that are generated by this particular study, especially as we move forward and we think about prevention of cancers,” Zon told the briefing.
This study was funded by the National Institute of Diabetes and Digestive and Kidney Diseases of the National Institutes of Health. Mavromatis reported no relevant disclosures. Zon reported stock or ownership interests in Oncolytics Biotech, TG Therapeutics, Select Sector SPDR Health Care, AstraZeneca, CRISPR, McKesson, and Berkshire Hathaway.
A version of this article first appeared on Medscape.com.
Can Popular Weight-Loss Drugs Protect Against Obesity-Related Cancers?
Can Popular Weight-Loss Drugs Protect Against Obesity-Related Cancers?
Statin-Induced Necrotizing Autoimmune Myopathy in a Patient With Complex Diabetes Management
Statin-Induced Necrotizing Autoimmune Myopathy in a Patient With Complex Diabetes Management
Muscle-related complaints occur in 7% to 25% of patients taking statin medications.1 In most instances, these adverse effects are quickly resolved when the medication is discontinued, but in rare occurrences, the statin can trigger an autoimmune response that progresses even after stopping use. This uncommon condition is typically accompanied by symmetrical proximal muscle weakness and an elevated CPK leading to a necrotizing myopathy requiring treatment with immunosuppressive therapy. Although less common, some patients may also present with dysphagia, myalgia, weight loss, and/or skin rash.1
Statin medications have been the cornerstone of lipid-lowering therapy due to their mechanism of inhibiting 3-hydroxy-3-methylglutaryl coenzyme A reductase (HMGCR), which is the rate-limiting step within the cholesterol synthesis pathway to produce mevalonic acid. There is a proven genetic association with human leukocyte antigen (HLA)-DRB1*11:01 in adults and anti-HMGCR–associated myopathy.1 The incidence of statin-induced necrotizing autoimmune myopathy (SINAM) in relation to each specific statin agent remains unknown; however, a systematic review of case reports found higher correlations for atorvastatin and simvastatin.2
There are 2 ways to confirm a SINAM diagnosis. The first and simplest includes checking for the presence of antibodies against HMGCR. The anti-HMGCR antibody test is typically used as a definitive diagnosis because it has a high specificity for SINAM.3 The second and more invasive diagnosis method involves a muscle biopsy, which is identified as positive if the biopsy shows the presence of necrotic muscle fibers.1,3
The anti-HMGCR antibody test can serve as a marker for disease activity because the antibodies are strongly correlated with CPK levels.1 CPK levels indicate the severity of muscle injury and is often used in addition to either of the confirmatory tests because it is faster and less expensive. Anti-HMGCR titers may remain positive while CPK returns to baseline when SINAM is dormant. In addition, clinicians may use an electromyography (EMG) test to measure the muscle response in association to nerve stimulation. 1 This test can show potential features of myopathic lesions such as positive sharp waves, spontaneous fibrillations, or myotonic repetitive potentials.
Typical treatment includes glucocorticoids as first-line agents, but SINAM can be difficult to treat due to its complicated pathophysiology processes.3 Escalation of therapy is sometimes required beyond a single agent; in these complex scenarios, methotrexate and/or intravenous (IV) immunoglobulin (IVIG) therapy are frequently added to the steroid therapy. There have been concerns with steroid use in specific patient populations due to the undesired adverse effect (AE) profile, and as a result IVIG has been used as monotherapy at a dose of 2 g/kg per month.3 Studies looking at IVIG monotherapy showed a reduction in CPK levels and improvement in strength after just 2 to 3 rounds of monthly treatment.3 Some patients receiving IVIG monotherapy even achieved baseline strength and no longer reported muscle-related symptoms, although the total treatment duration varied. A systematic review of 39 articles where glucocorticoids, IVIG, methotrexate and/or a combination were used to treat SINAM found an average time to remission of 8.6 months. Additionally, this systematic review observed more patients returned to baseline or experienced improvement in symptoms when being treated with a combination of glucocorticoid plus IVIG plus methotrexate.2 Suggested dosing recommendations are available in Table 1.

Patients diagnosed with HMGCR antibody myopathy are contraindicated for future statin therapy.1 Rechallenge of statins in this patient population has led to worsening of disease and therefore these patients should have a severe statin allergy listed in their medical documentation record.
CASE PRESENTATION
A 59-year-old male patient with a medical history including atrial fibrillation, peripheral vascular disease, type 2 diabetes mellitus (T2DM), hypertension, and peripheral neuropathy was referred by his primary care clinical pharmacist practitioner for an outpatient neurology consult. The patient reported a 4-month history of fatigue, lower extremity paresthesia, and progressive proximal muscle weakness which began in his legs, mostly noticeable when walking upstairs but quickly developed into bilateral arm weakness. The patient reported significant impact on his quality of life: he could no longer lift his arms above his head and had difficulty with daily activities such as brushing his hair or getting up from a chair. He reported multiple falls at home, and began to use a cane for assistance with ambulation. He confirmed adherence to atorvastatin over the past year. Laboratory testing on the day of the visit revealed an elevated CPK level at 9729 mcg/L (reference range for men, 30-300 mcg/L).
The patient was urged to go to the emergency department where his CPK level had increased to 12,990 mcg/L (Figure 1). The workup began to find the source of rhabdomyolysis and elevated liver enzymes differentiating autoimmune vs medication-induced myopathy. Upon admission atorvastatin was discontinued, anti-HMGCR antibody level was ordered, and IV fluids were started.
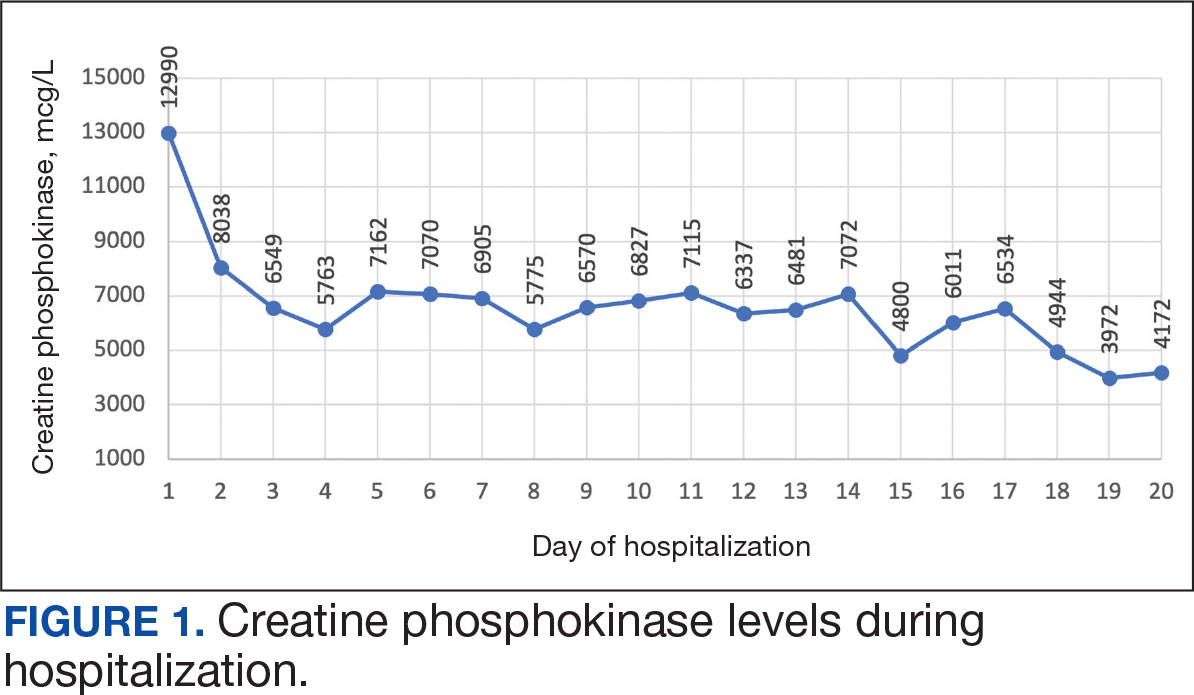
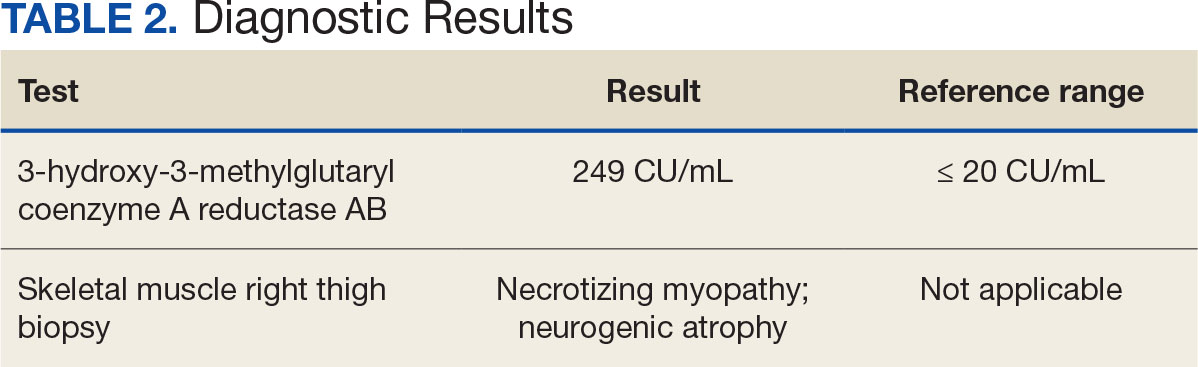
After 8 days of hospital admission with minimal improvement, Rheumatology and Neurology services were consulted in the setting of persistent CPK elevation and the potential neuropathic component of muscle weakness. Both consulting services agreed to consider muscle biopsy and EMG if the patient did not begin to show signs of improvement. The patient’s CPK levels remained elevated with minimal change in muscle weakness. The next step was a right quadricep muscle biopsy performed on Day 14 of admission. Sixteen days after admission, the anti-HMGCR antibody test (originally obtained upon admission) was positive and elevated at 249 CU/mL (reference range, < 20 CU/mL negative; reference range, ≥ 60 CU/mL strong positive), which confirmed the SINAM diagnosis (Table 2).
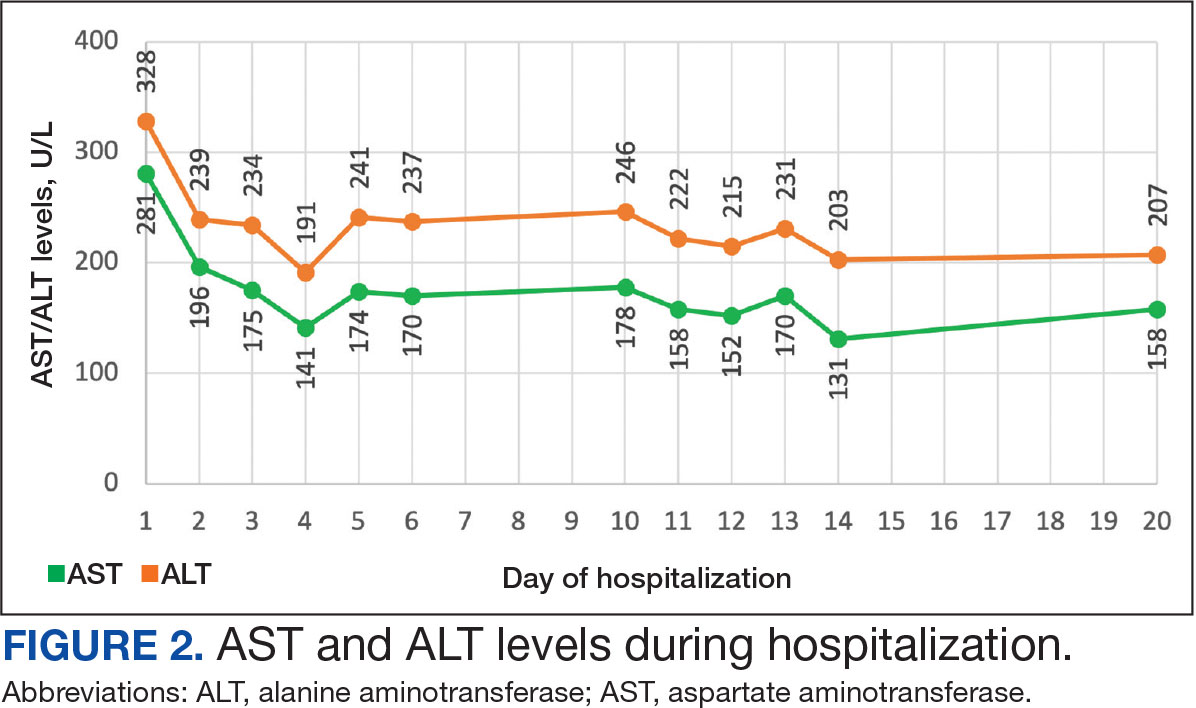
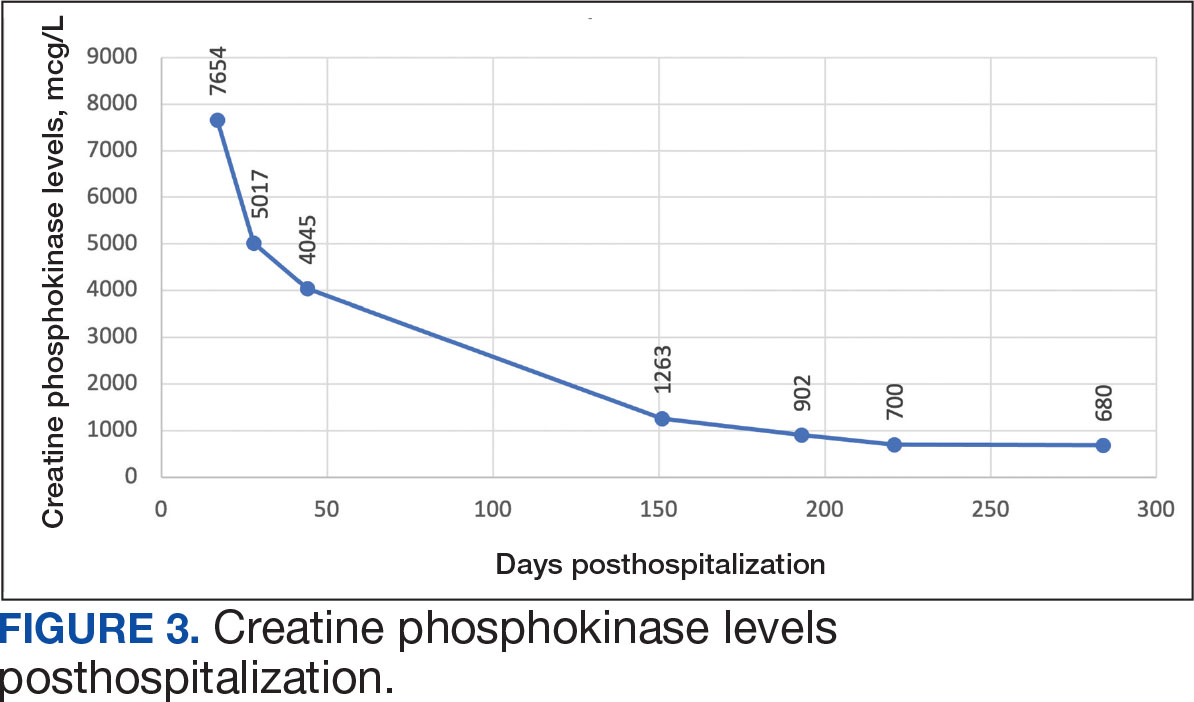
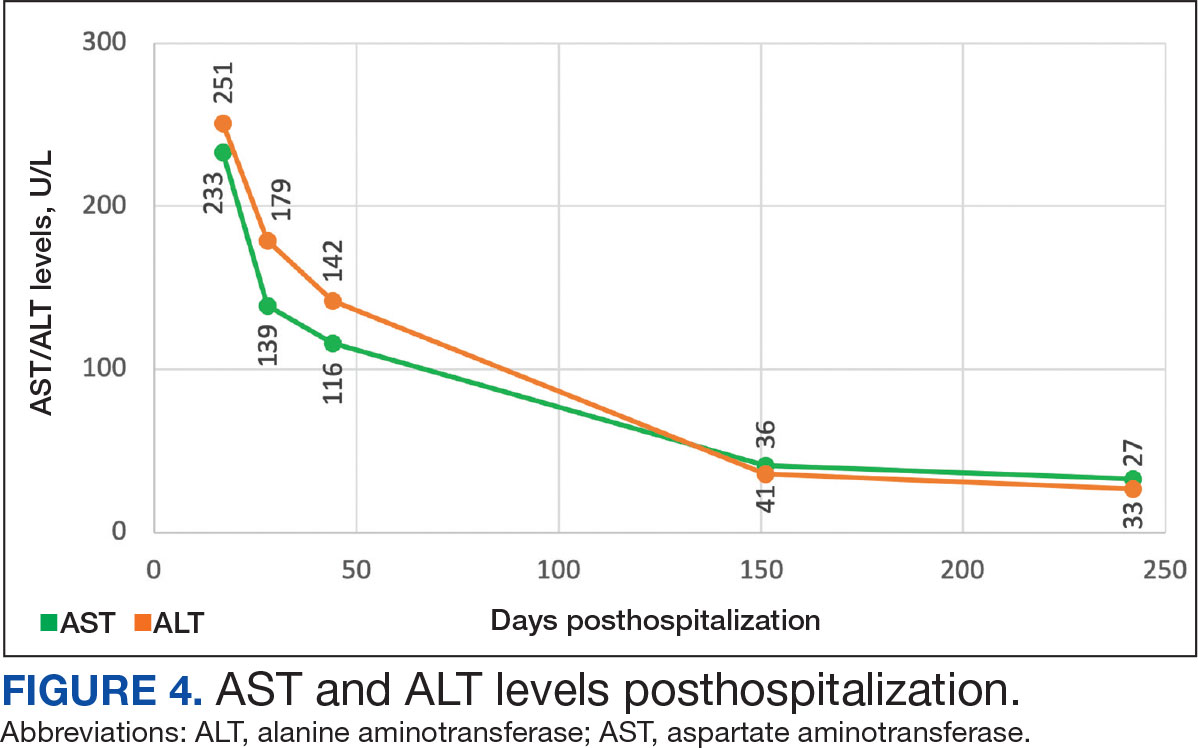
On Day 17 of hospitalization, the Neurology service initiated IVIG monotherapy to avoid the undesired glycemic AEs associated with glucocorticoids. The patient had a history of T2DM that was difficult to manage and his hemoglobin A1c level was the best it had ever been (6.2%) relative to a peak A1c of 11.0% 9 months prior. The patient was treated with a total IVIG dose of 2 g/kg divided into 3 daily doses while still obtaining CPK levels with daily laboratory tests to assist with trending the extent of disease severity improvement (Figures 2-4). After a 20-day hospital stay, the patient was discharged home with rehabilitation services and a scheduled outpatient EMG the following week.
The patient continued to report generalized body weakness, pain, and deconditioning upon discharge and was unable to attend the EMG neurology appointment. The patient did eventually attend a follow-up appointment about 6 weeks after hospital discharge and reported continued weakness. The Neurology service prescribed a 2-day IVIG regimen (total dose = 2 g/kg) monthly for the next 2 months. The patient returned to the neurology clinic 8 weeks later following 2 rounds of IVIG posthospitalization and reported that his muscle strength was returning, and he was able to slowly reintroduce exercise into his daily routine. During a follow-up appointment about 11 months after the initial hospitalization, the patient’s primary care clinical pharmacist provided education of effective management of cholesterol without statins, including use of proprotein convertase subtilisin/kexin type 9 (PCSK9) inhibitors as recommended by the Neurology service. At this time, the patient’s calculated low-density lipoprotein (LDL) was 110 mg/dL (reference range, 0-99 mg/dL). The patient preferred to work on a healthy diet and positive lifestyle choices before trialing any lipid lowering therapies.
The patient appeared to tolerate this treatment regimen following 7 rounds of IVIG. He noted fatigue for about 24 hours after his infusion sessions but otherwise reported no additional AEs. He has continued to attend weekly physical therapy sessions and is able to walk without the assistance of a cane. He can now walk a mile before he begins to feel fatigued or experience bilateral lower leg pain. The pain appears neuropathic in nature, as the patient reports ongoing “pins and needles” sensation in his legs and feet. The patient has noticed a major improvement in his overall function, strength, and exercise tolerance since starting IVIG treatments and although he is not yet back to his baseline, he is motivated to continue his recovery. Neurology is considering ongoing treatment with IVIG monthly infusions given his continued clinical improvement.
DISCUSSION
There is limited evidence on the use of IVIG monotherapy for SINAM, although it may be a viable option for patients deemed poor candidates for glucocorticoid or methotrexate therapy. This particularly applies to patients with DM for which there may be concerns for managing blood glucose levels with steroid use. The Johns Hopkins Myositis Center evaluated 3 patients with SINAM who declined glucocorticoid therapy and had documented DM and weakness in the proximal arms and legs. Following 2 to 3 monthly rounds of IVIG 2 g/kg monotherapy, these patients had reduced CPK levels and had improvement in both arm and hip-flexion strength. Two patients reported no muscle-related symptoms after completing IVIG monotherapy treatment for 9 and 19 months.3
The optimal treatment duration for IVIG monotherapy for SINAM is still uncertain given the limited available data. The patient in this case report showed clinically significant muscle-related improvement following 7 monthly rounds of 2 g/kg IVIG treatments. The mechanism of action for IVIG in this setting is still unknown, although the medication may allow muscle regeneration to surpass muscle destruction, thus leading to resolution of the muscle-related symptoms.3
There are numerous concerns with IVIG use to consider prior to initiating treatment, including expense, AEs, patient response, and comorbidities. IVIG is considerably more expensive than glucocorticoid and methotrexate alternatives. Systemic reactions have been shown to occur in 5% to 15% of patients receiving IVIG infusion.4 The majority of these infusion reactions occur early during infusion or within a few hours after administration is complete.5 Early AEs to monitor for include injection site reactions, flu-like symptoms, dermatologic reactions, anaphylaxis, transfusion-related acute lung injury, and transfusion-associated circulatory overload. Additional AEs may be delayed, including thromboembolic events, acute kidney injury, aseptic meningitis, hemolysis, neutropenia, and blood-borne infection.6 IVIG has a boxed warning for thrombosis, renal dysfunction, and acute renal failure risk.7 There are multiple strategies documented to reduce the risk of IVIG reactions including slowing the infusion rate, ensuring adequate hydration, and/or giving analgesics, antihistamines, or steroids prior to infusion.6 The patient in this case had monthly IVIG infusions without the need of any pretreatment medications and only reported fatigue for about 24 hours following the infusion.
An essential question is how to provide safe cholesterol management for patients with SINAM. Some evidence has suggested that other lipid-lowering medications that avoid the mevalonate pathway, such as fenofibrate or ezetimibe, may be used cautiously initially at lower doses.1 Due to the severity of SINAM, it is crucial to closely monitor and ensure tolerability as new lipid-lowering agents are introduced. More evidence suggests that PCSK9 inhibitors are a safer option.8 PCSK9 inhibitors avoid the mevalonate pathway and block PCSK9 from binding to LDL receptors, allowing LDL to be removed from circulation.
Tiniakou et al followed 8 individuals for a mean 1.5 years who had anti-HMGCR immune-mediated myopathy at high cardiovascular risk. Muscle strength, CPK levels, and serum anti-HMGCR antibody titers were assessed at baseline and again after initiation of PCSK9 inhibitor. None of the patients experienced a decline in their muscle strength. CPK, anti-HMGCR antibody levels, and LDL trended down in all participants and 2 patients were able to reduce their immunosuppression treatment while still achieving clinical improvement. Tiniakou et al suggest that PCSK9 inhibitors are a safe and effective option to lower cholesterol in patients with SINAM.8
Alirocumab is the preferred PCSK9 inhibitor for patients at the US Department of Veterans Affairs (VA). The VA Pharmacy Benefits Management (PBM) Service guidance recommends alirocumab for patients with a history of atherosclerotic cardiovascular disease (ASCVD) or severe hypercholesterolemia.9 PBM guidance suggests alirocumab use for patients with a contraindication, intolerance, or insufficient LDL reduction with a maximally tolerated dose of statin and ezetimibe with a desire to reduce ASCVD risk by lowering LDL. Per the PBM Criteria for Use guidance, patients should follow the stepwise approach and trial ezetimibe prior to being considered for PCSK9 inhibitor therapy. Given the patient’s contraindication to future statin use and severity of myopathy, in this case the Neurology Service felt that the safest option to reach goal LDL reduction would be a PCSK9 inhibitor. Consideration can be made for alirocumab use when considering an alternative lipid lowering therapy.
CONCLUSIONS
This report demonstrates a case of SINAM caused by atorvastatin therapy. Patients presenting with proximal muscle weakness and elevated CPK even after statin discontinuation should be considered for a full workup to determine whether SINAM may be involved. This uncommon form of myopathy can be diagnosed based on the detection of anti-HMGCR antibodies and/or presence of necrosis on muscle biopsy. A combination of glucocorticoid, methotrexate, and IVIG is recommended for a patient’s best chance of muscle symptom improvement. IVIG monotherapy should be considered for patients with glycemic control concerns.
- Tiniakou E. Statin-associated autoimmune myopathy: current perspectives. Ther Clin Risk Manag. 2020;16:483-492. doi:10.2147/TCRM.S197941
- Somagutta MKR, Shama N, Pormento MKL, et al. Statin-induced necrotizing autoimmune myopathy: a systematic review. Reumatologia. 2022;60(1):63-69. doi:10.5114/reum.2022.114108
- Mammen AL, Tiniakou E. Intravenous immune globulin for statin-triggered autoimmune myopathy. N Engl J Med. 2015;373(17):1680-1682. doi:10.1056/NEJMc1506163
- Stiehm ER. Adverse effects of human immunoglobulin therapy. Transfus Med Rev. 2013;27(3):171-178. doi:10.1016/j.tmrv.2013.05.004
- Ameratunga R, Sinclair J, Kolbe J. Increased risk of adverse events when changing intravenous immunoglobulin preparations. Clin Exp Immunol. 2004;136(1):111-113. doi:10.1111/j.1365-2249.2004.02412.x
- Abbas A, Rajabally YA. Complications of immunoglobulin therapy and implications for treatment of inflammatory neuropathy: a review. Curr Drug Saf. 2019;14(1):3-13. doi:10.2174/1574886313666181017121139
- Privigen. Prescribing information. CSL Behring LLC; 2022. Accessed March 17, 2025. https://labeling.cslbehring.com/PI/US/Privigen/EN/Privigen-Prescribing-Information.pdf
- Tiniakou E, Rivera E, Mammen AL, Christopher-Stine L. Use of proprotein convertase subtilisin/Kexin Type 9 inhibitors in statin-associated immune-mediated necrotizing myopathy: a case series. Arthritis Rheumatol. 2019;71(10):1723-1726. doi:10.1002/art.40919
- US Department of Veterans Affairs, Pharmacy Benefits Management (PBM) Services. Proprotein Convertase Subtilisin/Kexin Type 9 (PCSK9 Inhibitor) (Alirocumabpreferred, Evolocumab-non-preferred) Criteria for Use. June 2024. Accessed March 25, 2025. https://www.va.gov/formularyadvisor/DOC/128
- Jayatilaka S, Desai K, Rijal S, Zimmerman D. Statin-induced autoimmune necrotizing myopathy. J Prim Care Community Health. 2021;12:21501327211028714. doi:10.1177/21501327211028714
Muscle-related complaints occur in 7% to 25% of patients taking statin medications.1 In most instances, these adverse effects are quickly resolved when the medication is discontinued, but in rare occurrences, the statin can trigger an autoimmune response that progresses even after stopping use. This uncommon condition is typically accompanied by symmetrical proximal muscle weakness and an elevated CPK leading to a necrotizing myopathy requiring treatment with immunosuppressive therapy. Although less common, some patients may also present with dysphagia, myalgia, weight loss, and/or skin rash.1
Statin medications have been the cornerstone of lipid-lowering therapy due to their mechanism of inhibiting 3-hydroxy-3-methylglutaryl coenzyme A reductase (HMGCR), which is the rate-limiting step within the cholesterol synthesis pathway to produce mevalonic acid. There is a proven genetic association with human leukocyte antigen (HLA)-DRB1*11:01 in adults and anti-HMGCR–associated myopathy.1 The incidence of statin-induced necrotizing autoimmune myopathy (SINAM) in relation to each specific statin agent remains unknown; however, a systematic review of case reports found higher correlations for atorvastatin and simvastatin.2
There are 2 ways to confirm a SINAM diagnosis. The first and simplest includes checking for the presence of antibodies against HMGCR. The anti-HMGCR antibody test is typically used as a definitive diagnosis because it has a high specificity for SINAM.3 The second and more invasive diagnosis method involves a muscle biopsy, which is identified as positive if the biopsy shows the presence of necrotic muscle fibers.1,3
The anti-HMGCR antibody test can serve as a marker for disease activity because the antibodies are strongly correlated with CPK levels.1 CPK levels indicate the severity of muscle injury and is often used in addition to either of the confirmatory tests because it is faster and less expensive. Anti-HMGCR titers may remain positive while CPK returns to baseline when SINAM is dormant. In addition, clinicians may use an electromyography (EMG) test to measure the muscle response in association to nerve stimulation. 1 This test can show potential features of myopathic lesions such as positive sharp waves, spontaneous fibrillations, or myotonic repetitive potentials.
Typical treatment includes glucocorticoids as first-line agents, but SINAM can be difficult to treat due to its complicated pathophysiology processes.3 Escalation of therapy is sometimes required beyond a single agent; in these complex scenarios, methotrexate and/or intravenous (IV) immunoglobulin (IVIG) therapy are frequently added to the steroid therapy. There have been concerns with steroid use in specific patient populations due to the undesired adverse effect (AE) profile, and as a result IVIG has been used as monotherapy at a dose of 2 g/kg per month.3 Studies looking at IVIG monotherapy showed a reduction in CPK levels and improvement in strength after just 2 to 3 rounds of monthly treatment.3 Some patients receiving IVIG monotherapy even achieved baseline strength and no longer reported muscle-related symptoms, although the total treatment duration varied. A systematic review of 39 articles where glucocorticoids, IVIG, methotrexate and/or a combination were used to treat SINAM found an average time to remission of 8.6 months. Additionally, this systematic review observed more patients returned to baseline or experienced improvement in symptoms when being treated with a combination of glucocorticoid plus IVIG plus methotrexate.2 Suggested dosing recommendations are available in Table 1.

Patients diagnosed with HMGCR antibody myopathy are contraindicated for future statin therapy.1 Rechallenge of statins in this patient population has led to worsening of disease and therefore these patients should have a severe statin allergy listed in their medical documentation record.
CASE PRESENTATION
A 59-year-old male patient with a medical history including atrial fibrillation, peripheral vascular disease, type 2 diabetes mellitus (T2DM), hypertension, and peripheral neuropathy was referred by his primary care clinical pharmacist practitioner for an outpatient neurology consult. The patient reported a 4-month history of fatigue, lower extremity paresthesia, and progressive proximal muscle weakness which began in his legs, mostly noticeable when walking upstairs but quickly developed into bilateral arm weakness. The patient reported significant impact on his quality of life: he could no longer lift his arms above his head and had difficulty with daily activities such as brushing his hair or getting up from a chair. He reported multiple falls at home, and began to use a cane for assistance with ambulation. He confirmed adherence to atorvastatin over the past year. Laboratory testing on the day of the visit revealed an elevated CPK level at 9729 mcg/L (reference range for men, 30-300 mcg/L).
The patient was urged to go to the emergency department where his CPK level had increased to 12,990 mcg/L (Figure 1). The workup began to find the source of rhabdomyolysis and elevated liver enzymes differentiating autoimmune vs medication-induced myopathy. Upon admission atorvastatin was discontinued, anti-HMGCR antibody level was ordered, and IV fluids were started.

After 8 days of hospital admission with minimal improvement, Rheumatology and Neurology services were consulted in the setting of persistent CPK elevation and the potential neuropathic component of muscle weakness. Both consulting services agreed to consider muscle biopsy and EMG if the patient did not begin to show signs of improvement. The patient’s CPK levels remained elevated with minimal change in muscle weakness. The next step was a right quadricep muscle biopsy performed on Day 14 of admission. Sixteen days after admission, the anti-HMGCR antibody test (originally obtained upon admission) was positive and elevated at 249 CU/mL (reference range, < 20 CU/mL negative; reference range, ≥ 60 CU/mL strong positive), which confirmed the SINAM diagnosis (Table 2).

On Day 17 of hospitalization, the Neurology service initiated IVIG monotherapy to avoid the undesired glycemic AEs associated with glucocorticoids. The patient had a history of T2DM that was difficult to manage and his hemoglobin A1c level was the best it had ever been (6.2%) relative to a peak A1c of 11.0% 9 months prior. The patient was treated with a total IVIG dose of 2 g/kg divided into 3 daily doses while still obtaining CPK levels with daily laboratory tests to assist with trending the extent of disease severity improvement (Figures 2-4). After a 20-day hospital stay, the patient was discharged home with rehabilitation services and a scheduled outpatient EMG the following week.



The patient continued to report generalized body weakness, pain, and deconditioning upon discharge and was unable to attend the EMG neurology appointment. The patient did eventually attend a follow-up appointment about 6 weeks after hospital discharge and reported continued weakness. The Neurology service prescribed a 2-day IVIG regimen (total dose = 2 g/kg) monthly for the next 2 months. The patient returned to the neurology clinic 8 weeks later following 2 rounds of IVIG posthospitalization and reported that his muscle strength was returning, and he was able to slowly reintroduce exercise into his daily routine. During a follow-up appointment about 11 months after the initial hospitalization, the patient’s primary care clinical pharmacist provided education of effective management of cholesterol without statins, including use of proprotein convertase subtilisin/kexin type 9 (PCSK9) inhibitors as recommended by the Neurology service. At this time, the patient’s calculated low-density lipoprotein (LDL) was 110 mg/dL (reference range, 0-99 mg/dL). The patient preferred to work on a healthy diet and positive lifestyle choices before trialing any lipid lowering therapies.
The patient appeared to tolerate this treatment regimen following 7 rounds of IVIG. He noted fatigue for about 24 hours after his infusion sessions but otherwise reported no additional AEs. He has continued to attend weekly physical therapy sessions and is able to walk without the assistance of a cane. He can now walk a mile before he begins to feel fatigued or experience bilateral lower leg pain. The pain appears neuropathic in nature, as the patient reports ongoing “pins and needles” sensation in his legs and feet. The patient has noticed a major improvement in his overall function, strength, and exercise tolerance since starting IVIG treatments and although he is not yet back to his baseline, he is motivated to continue his recovery. Neurology is considering ongoing treatment with IVIG monthly infusions given his continued clinical improvement.
DISCUSSION
There is limited evidence on the use of IVIG monotherapy for SINAM, although it may be a viable option for patients deemed poor candidates for glucocorticoid or methotrexate therapy. This particularly applies to patients with DM for which there may be concerns for managing blood glucose levels with steroid use. The Johns Hopkins Myositis Center evaluated 3 patients with SINAM who declined glucocorticoid therapy and had documented DM and weakness in the proximal arms and legs. Following 2 to 3 monthly rounds of IVIG 2 g/kg monotherapy, these patients had reduced CPK levels and had improvement in both arm and hip-flexion strength. Two patients reported no muscle-related symptoms after completing IVIG monotherapy treatment for 9 and 19 months.3
The optimal treatment duration for IVIG monotherapy for SINAM is still uncertain given the limited available data. The patient in this case report showed clinically significant muscle-related improvement following 7 monthly rounds of 2 g/kg IVIG treatments. The mechanism of action for IVIG in this setting is still unknown, although the medication may allow muscle regeneration to surpass muscle destruction, thus leading to resolution of the muscle-related symptoms.3
There are numerous concerns with IVIG use to consider prior to initiating treatment, including expense, AEs, patient response, and comorbidities. IVIG is considerably more expensive than glucocorticoid and methotrexate alternatives. Systemic reactions have been shown to occur in 5% to 15% of patients receiving IVIG infusion.4 The majority of these infusion reactions occur early during infusion or within a few hours after administration is complete.5 Early AEs to monitor for include injection site reactions, flu-like symptoms, dermatologic reactions, anaphylaxis, transfusion-related acute lung injury, and transfusion-associated circulatory overload. Additional AEs may be delayed, including thromboembolic events, acute kidney injury, aseptic meningitis, hemolysis, neutropenia, and blood-borne infection.6 IVIG has a boxed warning for thrombosis, renal dysfunction, and acute renal failure risk.7 There are multiple strategies documented to reduce the risk of IVIG reactions including slowing the infusion rate, ensuring adequate hydration, and/or giving analgesics, antihistamines, or steroids prior to infusion.6 The patient in this case had monthly IVIG infusions without the need of any pretreatment medications and only reported fatigue for about 24 hours following the infusion.
An essential question is how to provide safe cholesterol management for patients with SINAM. Some evidence has suggested that other lipid-lowering medications that avoid the mevalonate pathway, such as fenofibrate or ezetimibe, may be used cautiously initially at lower doses.1 Due to the severity of SINAM, it is crucial to closely monitor and ensure tolerability as new lipid-lowering agents are introduced. More evidence suggests that PCSK9 inhibitors are a safer option.8 PCSK9 inhibitors avoid the mevalonate pathway and block PCSK9 from binding to LDL receptors, allowing LDL to be removed from circulation.
Tiniakou et al followed 8 individuals for a mean 1.5 years who had anti-HMGCR immune-mediated myopathy at high cardiovascular risk. Muscle strength, CPK levels, and serum anti-HMGCR antibody titers were assessed at baseline and again after initiation of PCSK9 inhibitor. None of the patients experienced a decline in their muscle strength. CPK, anti-HMGCR antibody levels, and LDL trended down in all participants and 2 patients were able to reduce their immunosuppression treatment while still achieving clinical improvement. Tiniakou et al suggest that PCSK9 inhibitors are a safe and effective option to lower cholesterol in patients with SINAM.8
Alirocumab is the preferred PCSK9 inhibitor for patients at the US Department of Veterans Affairs (VA). The VA Pharmacy Benefits Management (PBM) Service guidance recommends alirocumab for patients with a history of atherosclerotic cardiovascular disease (ASCVD) or severe hypercholesterolemia.9 PBM guidance suggests alirocumab use for patients with a contraindication, intolerance, or insufficient LDL reduction with a maximally tolerated dose of statin and ezetimibe with a desire to reduce ASCVD risk by lowering LDL. Per the PBM Criteria for Use guidance, patients should follow the stepwise approach and trial ezetimibe prior to being considered for PCSK9 inhibitor therapy. Given the patient’s contraindication to future statin use and severity of myopathy, in this case the Neurology Service felt that the safest option to reach goal LDL reduction would be a PCSK9 inhibitor. Consideration can be made for alirocumab use when considering an alternative lipid lowering therapy.
CONCLUSIONS
This report demonstrates a case of SINAM caused by atorvastatin therapy. Patients presenting with proximal muscle weakness and elevated CPK even after statin discontinuation should be considered for a full workup to determine whether SINAM may be involved. This uncommon form of myopathy can be diagnosed based on the detection of anti-HMGCR antibodies and/or presence of necrosis on muscle biopsy. A combination of glucocorticoid, methotrexate, and IVIG is recommended for a patient’s best chance of muscle symptom improvement. IVIG monotherapy should be considered for patients with glycemic control concerns.
Muscle-related complaints occur in 7% to 25% of patients taking statin medications.1 In most instances, these adverse effects are quickly resolved when the medication is discontinued, but in rare occurrences, the statin can trigger an autoimmune response that progresses even after stopping use. This uncommon condition is typically accompanied by symmetrical proximal muscle weakness and an elevated CPK leading to a necrotizing myopathy requiring treatment with immunosuppressive therapy. Although less common, some patients may also present with dysphagia, myalgia, weight loss, and/or skin rash.1
Statin medications have been the cornerstone of lipid-lowering therapy due to their mechanism of inhibiting 3-hydroxy-3-methylglutaryl coenzyme A reductase (HMGCR), which is the rate-limiting step within the cholesterol synthesis pathway to produce mevalonic acid. There is a proven genetic association with human leukocyte antigen (HLA)-DRB1*11:01 in adults and anti-HMGCR–associated myopathy.1 The incidence of statin-induced necrotizing autoimmune myopathy (SINAM) in relation to each specific statin agent remains unknown; however, a systematic review of case reports found higher correlations for atorvastatin and simvastatin.2
There are 2 ways to confirm a SINAM diagnosis. The first and simplest includes checking for the presence of antibodies against HMGCR. The anti-HMGCR antibody test is typically used as a definitive diagnosis because it has a high specificity for SINAM.3 The second and more invasive diagnosis method involves a muscle biopsy, which is identified as positive if the biopsy shows the presence of necrotic muscle fibers.1,3
The anti-HMGCR antibody test can serve as a marker for disease activity because the antibodies are strongly correlated with CPK levels.1 CPK levels indicate the severity of muscle injury and is often used in addition to either of the confirmatory tests because it is faster and less expensive. Anti-HMGCR titers may remain positive while CPK returns to baseline when SINAM is dormant. In addition, clinicians may use an electromyography (EMG) test to measure the muscle response in association to nerve stimulation. 1 This test can show potential features of myopathic lesions such as positive sharp waves, spontaneous fibrillations, or myotonic repetitive potentials.
Typical treatment includes glucocorticoids as first-line agents, but SINAM can be difficult to treat due to its complicated pathophysiology processes.3 Escalation of therapy is sometimes required beyond a single agent; in these complex scenarios, methotrexate and/or intravenous (IV) immunoglobulin (IVIG) therapy are frequently added to the steroid therapy. There have been concerns with steroid use in specific patient populations due to the undesired adverse effect (AE) profile, and as a result IVIG has been used as monotherapy at a dose of 2 g/kg per month.3 Studies looking at IVIG monotherapy showed a reduction in CPK levels and improvement in strength after just 2 to 3 rounds of monthly treatment.3 Some patients receiving IVIG monotherapy even achieved baseline strength and no longer reported muscle-related symptoms, although the total treatment duration varied. A systematic review of 39 articles where glucocorticoids, IVIG, methotrexate and/or a combination were used to treat SINAM found an average time to remission of 8.6 months. Additionally, this systematic review observed more patients returned to baseline or experienced improvement in symptoms when being treated with a combination of glucocorticoid plus IVIG plus methotrexate.2 Suggested dosing recommendations are available in Table 1.

Patients diagnosed with HMGCR antibody myopathy are contraindicated for future statin therapy.1 Rechallenge of statins in this patient population has led to worsening of disease and therefore these patients should have a severe statin allergy listed in their medical documentation record.
CASE PRESENTATION
A 59-year-old male patient with a medical history including atrial fibrillation, peripheral vascular disease, type 2 diabetes mellitus (T2DM), hypertension, and peripheral neuropathy was referred by his primary care clinical pharmacist practitioner for an outpatient neurology consult. The patient reported a 4-month history of fatigue, lower extremity paresthesia, and progressive proximal muscle weakness which began in his legs, mostly noticeable when walking upstairs but quickly developed into bilateral arm weakness. The patient reported significant impact on his quality of life: he could no longer lift his arms above his head and had difficulty with daily activities such as brushing his hair or getting up from a chair. He reported multiple falls at home, and began to use a cane for assistance with ambulation. He confirmed adherence to atorvastatin over the past year. Laboratory testing on the day of the visit revealed an elevated CPK level at 9729 mcg/L (reference range for men, 30-300 mcg/L).
The patient was urged to go to the emergency department where his CPK level had increased to 12,990 mcg/L (Figure 1). The workup began to find the source of rhabdomyolysis and elevated liver enzymes differentiating autoimmune vs medication-induced myopathy. Upon admission atorvastatin was discontinued, anti-HMGCR antibody level was ordered, and IV fluids were started.

After 8 days of hospital admission with minimal improvement, Rheumatology and Neurology services were consulted in the setting of persistent CPK elevation and the potential neuropathic component of muscle weakness. Both consulting services agreed to consider muscle biopsy and EMG if the patient did not begin to show signs of improvement. The patient’s CPK levels remained elevated with minimal change in muscle weakness. The next step was a right quadricep muscle biopsy performed on Day 14 of admission. Sixteen days after admission, the anti-HMGCR antibody test (originally obtained upon admission) was positive and elevated at 249 CU/mL (reference range, < 20 CU/mL negative; reference range, ≥ 60 CU/mL strong positive), which confirmed the SINAM diagnosis (Table 2).

On Day 17 of hospitalization, the Neurology service initiated IVIG monotherapy to avoid the undesired glycemic AEs associated with glucocorticoids. The patient had a history of T2DM that was difficult to manage and his hemoglobin A1c level was the best it had ever been (6.2%) relative to a peak A1c of 11.0% 9 months prior. The patient was treated with a total IVIG dose of 2 g/kg divided into 3 daily doses while still obtaining CPK levels with daily laboratory tests to assist with trending the extent of disease severity improvement (Figures 2-4). After a 20-day hospital stay, the patient was discharged home with rehabilitation services and a scheduled outpatient EMG the following week.



The patient continued to report generalized body weakness, pain, and deconditioning upon discharge and was unable to attend the EMG neurology appointment. The patient did eventually attend a follow-up appointment about 6 weeks after hospital discharge and reported continued weakness. The Neurology service prescribed a 2-day IVIG regimen (total dose = 2 g/kg) monthly for the next 2 months. The patient returned to the neurology clinic 8 weeks later following 2 rounds of IVIG posthospitalization and reported that his muscle strength was returning, and he was able to slowly reintroduce exercise into his daily routine. During a follow-up appointment about 11 months after the initial hospitalization, the patient’s primary care clinical pharmacist provided education of effective management of cholesterol without statins, including use of proprotein convertase subtilisin/kexin type 9 (PCSK9) inhibitors as recommended by the Neurology service. At this time, the patient’s calculated low-density lipoprotein (LDL) was 110 mg/dL (reference range, 0-99 mg/dL). The patient preferred to work on a healthy diet and positive lifestyle choices before trialing any lipid lowering therapies.
The patient appeared to tolerate this treatment regimen following 7 rounds of IVIG. He noted fatigue for about 24 hours after his infusion sessions but otherwise reported no additional AEs. He has continued to attend weekly physical therapy sessions and is able to walk without the assistance of a cane. He can now walk a mile before he begins to feel fatigued or experience bilateral lower leg pain. The pain appears neuropathic in nature, as the patient reports ongoing “pins and needles” sensation in his legs and feet. The patient has noticed a major improvement in his overall function, strength, and exercise tolerance since starting IVIG treatments and although he is not yet back to his baseline, he is motivated to continue his recovery. Neurology is considering ongoing treatment with IVIG monthly infusions given his continued clinical improvement.
DISCUSSION
There is limited evidence on the use of IVIG monotherapy for SINAM, although it may be a viable option for patients deemed poor candidates for glucocorticoid or methotrexate therapy. This particularly applies to patients with DM for which there may be concerns for managing blood glucose levels with steroid use. The Johns Hopkins Myositis Center evaluated 3 patients with SINAM who declined glucocorticoid therapy and had documented DM and weakness in the proximal arms and legs. Following 2 to 3 monthly rounds of IVIG 2 g/kg monotherapy, these patients had reduced CPK levels and had improvement in both arm and hip-flexion strength. Two patients reported no muscle-related symptoms after completing IVIG monotherapy treatment for 9 and 19 months.3
The optimal treatment duration for IVIG monotherapy for SINAM is still uncertain given the limited available data. The patient in this case report showed clinically significant muscle-related improvement following 7 monthly rounds of 2 g/kg IVIG treatments. The mechanism of action for IVIG in this setting is still unknown, although the medication may allow muscle regeneration to surpass muscle destruction, thus leading to resolution of the muscle-related symptoms.3
There are numerous concerns with IVIG use to consider prior to initiating treatment, including expense, AEs, patient response, and comorbidities. IVIG is considerably more expensive than glucocorticoid and methotrexate alternatives. Systemic reactions have been shown to occur in 5% to 15% of patients receiving IVIG infusion.4 The majority of these infusion reactions occur early during infusion or within a few hours after administration is complete.5 Early AEs to monitor for include injection site reactions, flu-like symptoms, dermatologic reactions, anaphylaxis, transfusion-related acute lung injury, and transfusion-associated circulatory overload. Additional AEs may be delayed, including thromboembolic events, acute kidney injury, aseptic meningitis, hemolysis, neutropenia, and blood-borne infection.6 IVIG has a boxed warning for thrombosis, renal dysfunction, and acute renal failure risk.7 There are multiple strategies documented to reduce the risk of IVIG reactions including slowing the infusion rate, ensuring adequate hydration, and/or giving analgesics, antihistamines, or steroids prior to infusion.6 The patient in this case had monthly IVIG infusions without the need of any pretreatment medications and only reported fatigue for about 24 hours following the infusion.
An essential question is how to provide safe cholesterol management for patients with SINAM. Some evidence has suggested that other lipid-lowering medications that avoid the mevalonate pathway, such as fenofibrate or ezetimibe, may be used cautiously initially at lower doses.1 Due to the severity of SINAM, it is crucial to closely monitor and ensure tolerability as new lipid-lowering agents are introduced. More evidence suggests that PCSK9 inhibitors are a safer option.8 PCSK9 inhibitors avoid the mevalonate pathway and block PCSK9 from binding to LDL receptors, allowing LDL to be removed from circulation.
Tiniakou et al followed 8 individuals for a mean 1.5 years who had anti-HMGCR immune-mediated myopathy at high cardiovascular risk. Muscle strength, CPK levels, and serum anti-HMGCR antibody titers were assessed at baseline and again after initiation of PCSK9 inhibitor. None of the patients experienced a decline in their muscle strength. CPK, anti-HMGCR antibody levels, and LDL trended down in all participants and 2 patients were able to reduce their immunosuppression treatment while still achieving clinical improvement. Tiniakou et al suggest that PCSK9 inhibitors are a safe and effective option to lower cholesterol in patients with SINAM.8
Alirocumab is the preferred PCSK9 inhibitor for patients at the US Department of Veterans Affairs (VA). The VA Pharmacy Benefits Management (PBM) Service guidance recommends alirocumab for patients with a history of atherosclerotic cardiovascular disease (ASCVD) or severe hypercholesterolemia.9 PBM guidance suggests alirocumab use for patients with a contraindication, intolerance, or insufficient LDL reduction with a maximally tolerated dose of statin and ezetimibe with a desire to reduce ASCVD risk by lowering LDL. Per the PBM Criteria for Use guidance, patients should follow the stepwise approach and trial ezetimibe prior to being considered for PCSK9 inhibitor therapy. Given the patient’s contraindication to future statin use and severity of myopathy, in this case the Neurology Service felt that the safest option to reach goal LDL reduction would be a PCSK9 inhibitor. Consideration can be made for alirocumab use when considering an alternative lipid lowering therapy.
CONCLUSIONS
This report demonstrates a case of SINAM caused by atorvastatin therapy. Patients presenting with proximal muscle weakness and elevated CPK even after statin discontinuation should be considered for a full workup to determine whether SINAM may be involved. This uncommon form of myopathy can be diagnosed based on the detection of anti-HMGCR antibodies and/or presence of necrosis on muscle biopsy. A combination of glucocorticoid, methotrexate, and IVIG is recommended for a patient’s best chance of muscle symptom improvement. IVIG monotherapy should be considered for patients with glycemic control concerns.
- Tiniakou E. Statin-associated autoimmune myopathy: current perspectives. Ther Clin Risk Manag. 2020;16:483-492. doi:10.2147/TCRM.S197941
- Somagutta MKR, Shama N, Pormento MKL, et al. Statin-induced necrotizing autoimmune myopathy: a systematic review. Reumatologia. 2022;60(1):63-69. doi:10.5114/reum.2022.114108
- Mammen AL, Tiniakou E. Intravenous immune globulin for statin-triggered autoimmune myopathy. N Engl J Med. 2015;373(17):1680-1682. doi:10.1056/NEJMc1506163
- Stiehm ER. Adverse effects of human immunoglobulin therapy. Transfus Med Rev. 2013;27(3):171-178. doi:10.1016/j.tmrv.2013.05.004
- Ameratunga R, Sinclair J, Kolbe J. Increased risk of adverse events when changing intravenous immunoglobulin preparations. Clin Exp Immunol. 2004;136(1):111-113. doi:10.1111/j.1365-2249.2004.02412.x
- Abbas A, Rajabally YA. Complications of immunoglobulin therapy and implications for treatment of inflammatory neuropathy: a review. Curr Drug Saf. 2019;14(1):3-13. doi:10.2174/1574886313666181017121139
- Privigen. Prescribing information. CSL Behring LLC; 2022. Accessed March 17, 2025. https://labeling.cslbehring.com/PI/US/Privigen/EN/Privigen-Prescribing-Information.pdf
- Tiniakou E, Rivera E, Mammen AL, Christopher-Stine L. Use of proprotein convertase subtilisin/Kexin Type 9 inhibitors in statin-associated immune-mediated necrotizing myopathy: a case series. Arthritis Rheumatol. 2019;71(10):1723-1726. doi:10.1002/art.40919
- US Department of Veterans Affairs, Pharmacy Benefits Management (PBM) Services. Proprotein Convertase Subtilisin/Kexin Type 9 (PCSK9 Inhibitor) (Alirocumabpreferred, Evolocumab-non-preferred) Criteria for Use. June 2024. Accessed March 25, 2025. https://www.va.gov/formularyadvisor/DOC/128
- Jayatilaka S, Desai K, Rijal S, Zimmerman D. Statin-induced autoimmune necrotizing myopathy. J Prim Care Community Health. 2021;12:21501327211028714. doi:10.1177/21501327211028714
- Tiniakou E. Statin-associated autoimmune myopathy: current perspectives. Ther Clin Risk Manag. 2020;16:483-492. doi:10.2147/TCRM.S197941
- Somagutta MKR, Shama N, Pormento MKL, et al. Statin-induced necrotizing autoimmune myopathy: a systematic review. Reumatologia. 2022;60(1):63-69. doi:10.5114/reum.2022.114108
- Mammen AL, Tiniakou E. Intravenous immune globulin for statin-triggered autoimmune myopathy. N Engl J Med. 2015;373(17):1680-1682. doi:10.1056/NEJMc1506163
- Stiehm ER. Adverse effects of human immunoglobulin therapy. Transfus Med Rev. 2013;27(3):171-178. doi:10.1016/j.tmrv.2013.05.004
- Ameratunga R, Sinclair J, Kolbe J. Increased risk of adverse events when changing intravenous immunoglobulin preparations. Clin Exp Immunol. 2004;136(1):111-113. doi:10.1111/j.1365-2249.2004.02412.x
- Abbas A, Rajabally YA. Complications of immunoglobulin therapy and implications for treatment of inflammatory neuropathy: a review. Curr Drug Saf. 2019;14(1):3-13. doi:10.2174/1574886313666181017121139
- Privigen. Prescribing information. CSL Behring LLC; 2022. Accessed March 17, 2025. https://labeling.cslbehring.com/PI/US/Privigen/EN/Privigen-Prescribing-Information.pdf
- Tiniakou E, Rivera E, Mammen AL, Christopher-Stine L. Use of proprotein convertase subtilisin/Kexin Type 9 inhibitors in statin-associated immune-mediated necrotizing myopathy: a case series. Arthritis Rheumatol. 2019;71(10):1723-1726. doi:10.1002/art.40919
- US Department of Veterans Affairs, Pharmacy Benefits Management (PBM) Services. Proprotein Convertase Subtilisin/Kexin Type 9 (PCSK9 Inhibitor) (Alirocumabpreferred, Evolocumab-non-preferred) Criteria for Use. June 2024. Accessed March 25, 2025. https://www.va.gov/formularyadvisor/DOC/128
- Jayatilaka S, Desai K, Rijal S, Zimmerman D. Statin-induced autoimmune necrotizing myopathy. J Prim Care Community Health. 2021;12:21501327211028714. doi:10.1177/21501327211028714
Statin-Induced Necrotizing Autoimmune Myopathy in a Patient With Complex Diabetes Management
Statin-Induced Necrotizing Autoimmune Myopathy in a Patient With Complex Diabetes Management
GLP-1s Have Real-World Benefits and Risks In Large Scale VA Study
A study of more than 2 million veterans with diabetes builds on evidence of broad-ranging benefits and risks of glucagon-like peptide 1 receptor agonists (GLP-1 RAs) in the clinical setting, providing an “atlas” mapping extensive outcomes and some new insights to potentially explore in more rigorous clinical trials.
“This is the largest study on GLP-1 receptor agonists,” first author Ziyad Al-Aly, MD, chief of research and development at the US Department of Veterans Affairs (VA) St. Louis Healthcare System, in St. Louis, told this news organization regarding the research, published this week, in Nature Medicine.
“The [study] reflects the real experiences of people using GLP-1 RAs [in the VA] clinical setting,” he said.
“Altogether, our discovery approach confirms previous studies and clinical trials and also uncovers previously unreported benefits and risks of GLP-1 RAs,” the authors wrote.
For the comprehensive study, Al-Aly and his colleagues evaluated data from the US Department of Veterans Affairs on more than 2 million veterans treated for diabetes between October 2017 and December 2023, assessing GLP-1 RA treatment in comparison with other diabetes therapies regarding a striking 175 clinical outcomes.
Of the patients, 215,970 initiated treatment with GLP-1 RAs; 159,465 started sulfonylureas, 117,989 dipeptidyl peptidase 4 inhibitors, and 258,614 were initiated on sodium-glucose cotransporter-2 inhibitors.
The study also included a composite group of the latter three drug groups (n = 536,068), and a control group of 1,203,097 of patients receiving usual care, who were compared with usual care with the addition of GLP-1 RAs.
After inverse probability weighting, the groups were well-balanced in terms of their baseline characteristics. While the majority in the VA cohort overall were White men, the study adjusted for gender, age, race, comorbidities, and an extensive array of covariates.
With an average follow-up of 3.68 years, after the multivariate adjustment, GLP-1 RAs showed “effectiveness and risks that extended beyond those currently recognized,” in comparison with each of the treatment groups and with the main control group of usual care, the authors reported.
For the largest comparison with the main control group of usual care alone, the addition of GLP-1 RAs was associated with a decreased risk in 24% of the outcomes evaluated, and an increased risk in 10.86% of outcomes, with no significant difference for the remaining 65.14% of outcomes.
Of the various benefits, key improvements included a reduced risk for several substance use disorders including alcohol (hazard ratio [HR], 0.89) and opioid (HR, 0.87) use, suicidal ideation, attempt or intentional self-harm (HR, 0.90), seizures (HR, 0.90), neurocognitive disorders including Alzheimer disease (HR, 0.88) and dementia (HR, 0.92), coagulation and clotting disorders (HR, 0.92), and cardiac arrest (HR, 0.78).
Further benefits vs usual care alone included a reduced risk for infectious illnesses (HR, 0.88), acute kidney injury (HR, 0.88), and chronic kidney disease (CKD) (HR, 0.97; P <.05 for all the outcomes).
In terms of risks associated with GLP-1 RAs, in addition to the well-known risks for nausea and vomiting, additional increased risks vs usual care included gastrointestinal disorders such as noninfectious gastroenteritis (HR, 1.12), hypotension (HR, 1.06), arthritis (HR, 1.11), tendinitis and synovitis (HR, 1.10), interstitial nephritis (HR, 1.06), nephrolithiasis (HR, 1.15), and the known risk for drug-induced acute pancreatitis (HR, 2.46).
Neuropsychiatric Effects
Among the various benefits in the study, Al-Aly said some of the most intriguing are those involving the brain.
“I am struck by the consistent effects on many neuropsychiatric disorders — this aligns with data showing the presence of GLP-1 receptors in the brain and evidence showing that GLP-1s permeates through the blood brain barrier and acts on the brain to reduce inflammation and oxidative stress, improve neuroplasticity, etc.,” he said.
“Clearly, there is a neurotropic effect. There is also the possibility of an effect on the immune system/fighting infection — with reduced risks of infections, sepsis, etc.”
The reductions in suicidal ideation are encouraging after earlier reports of suicidal thoughts and self-injury among young users of GLP-1 RAs prompted concerns, including a 2023 review of the drug use by the European Medicines Agency that ultimately found no causal association, the authors added. The US Food and Drug Administration also found no association with GLP-1s and suicide risk.
The reductions in addictive behaviors are also encouraging and are consistent with the role of GLP-1 receptors in the brain in terms of impulse control and reward signaling that can relate to addictive behaviors, Al-Aly explained.
The reduced risks for dementia and Alzheimer disease are likewise consistent with preclinical studies in animal models of Alzheimer disease, as well as clinical studies showing a reduced risk for dementia in patients with type 2 diabetes, the authors noted.
The observed reduced risk for seizures further “adds to an emerging body of knowledge, both mechanistic and early clinical data, indicative of the anticonvulsant properties of GLP-1 RA use,” they added.
“GLP-1 RAs should be further evaluated in future studies as potential adjuvant therapeutics for epilepsy and its associated comorbidities,” the authors suggested.
Kidneys
While the findings support evidence of protective effects of GLP-1 RAs on the kidneys and a reduction in CKD risk, notable risks observed, also involving the kidneys, include nephrolithiasis or kidney stones.
Al-Aly noted the mechanisms with kidney stone formation are very different from CKD, and he speculated that the risk for the former could in fact stem from potentially low hydration with GLP-1 RA use.
“When patients are on GLP-1 RAs, they definitely eat a lot less to lose weight, but they also hydrate themselves less,” he explained in a press briefing. “They drink less water because they feel full very quickly after eating, and I’m just theorizing, but perhaps chronic dehydration [is behind] the increased risk of kidney stones.”
Modest Effects?
While, overall, the benefits of GLP-1 RA drugs showed modest benefits ranging between a 10% and 20% reduction for most outcomes, Al-Aly said those effects are still important.
“The modest effect does not negate the potential value of these drugs, especially for conditions where few effective treatment options exist, for example, dementia,” he said in the press statement.
“This may also imply that these drugs are most beneficial when used in conjunction with other interventions, such as lifestyle changes or other medications.”
Potential Confounders A Concern
Commenting on the study, David M. Nathan, MD, founder of the MGH Diabetes Center and a professor of medicine at Harvard Medical School, in Boston, Massachusetts, noted that, while the study is hypothesis-generating, the key limitation is its observational nature.
“The authors did a perfectly respectable job of doing all you can do to adjust for [confounders], but with these kinds of studies, as much as you try to statistically account for differences in the populations before they were put on the drug, you can never truly adjust for all the potential confounders that may influence the results,” he told this news organization.
In addition, the 3.8-year follow-up time of the study, as the authors acknowledge, is especially short considering that GLP-1 RAs are generally recommended to be taken indefinitely.
“You have to take these drugs presumably for a lifetime and we have no idea what the longer-term benefits and risks are,” Nathan said.
Nathan, who was among the first investigators to evaluate GLP-1 RAs about 30 years ago, underscored that “I do think that these drugs are generally really spectacular; they’ve taken over the world and they are probably the single greatest pharmaceutical story of the 21st century.”
“But much more rigorous randomized trials would be needed to prove study results that haven’t already been established in previous clinical trials,” he said.
“The types of [randomized] trials that are necessary are very expensive and require a huge amount of work, but at the end of the day, they provide proof as to what does and doesn’t work, and what the true risks are,” he added. “Whether the GLP-RAs will cure all ills and bring about world peace needs to be proved.”
In further comments provided through the Science Media Center, Stephen O’Rahilly, FRS, a professor of clinical biochemistry and medicine and director of the Wellcome-MRC Institute of Metabolic Science-Metabolic Research Laboratories, University of Cambridge, Cambridge, England, echoed Nathan’s concern that “studies such as these have to be interpreted very cautiously as the people studied have not been randomly allocated to GLP-1 RA treatment, so any difference between those taking and not taking the class of drug could potentially be attributable to factors other than the drug.”
He noted, however, that “the study provides useful reassurance about the safety of this class of drugs. The expected benefits on heart disease, stroke and other cardiovascular and most kidney diseases are clearly seen.”
Al-Aly reported being an uncompensated consultant for Pfizer. Nathan, who has previously conducted clinical trials on GLP-1 RAs, currently has no relationships to report. O’Rahilly reported receiving remuneration from several pharmaceutical companies for scientific advice relating to the development of drugs for metabolic diseases, but none involving GLP-1 RAs in the past 3 years.
A version of this article first appeared on Medscape.com.
A study of more than 2 million veterans with diabetes builds on evidence of broad-ranging benefits and risks of glucagon-like peptide 1 receptor agonists (GLP-1 RAs) in the clinical setting, providing an “atlas” mapping extensive outcomes and some new insights to potentially explore in more rigorous clinical trials.
“This is the largest study on GLP-1 receptor agonists,” first author Ziyad Al-Aly, MD, chief of research and development at the US Department of Veterans Affairs (VA) St. Louis Healthcare System, in St. Louis, told this news organization regarding the research, published this week, in Nature Medicine.
“The [study] reflects the real experiences of people using GLP-1 RAs [in the VA] clinical setting,” he said.
“Altogether, our discovery approach confirms previous studies and clinical trials and also uncovers previously unreported benefits and risks of GLP-1 RAs,” the authors wrote.
For the comprehensive study, Al-Aly and his colleagues evaluated data from the US Department of Veterans Affairs on more than 2 million veterans treated for diabetes between October 2017 and December 2023, assessing GLP-1 RA treatment in comparison with other diabetes therapies regarding a striking 175 clinical outcomes.
Of the patients, 215,970 initiated treatment with GLP-1 RAs; 159,465 started sulfonylureas, 117,989 dipeptidyl peptidase 4 inhibitors, and 258,614 were initiated on sodium-glucose cotransporter-2 inhibitors.
The study also included a composite group of the latter three drug groups (n = 536,068), and a control group of 1,203,097 of patients receiving usual care, who were compared with usual care with the addition of GLP-1 RAs.
After inverse probability weighting, the groups were well-balanced in terms of their baseline characteristics. While the majority in the VA cohort overall were White men, the study adjusted for gender, age, race, comorbidities, and an extensive array of covariates.
With an average follow-up of 3.68 years, after the multivariate adjustment, GLP-1 RAs showed “effectiveness and risks that extended beyond those currently recognized,” in comparison with each of the treatment groups and with the main control group of usual care, the authors reported.
For the largest comparison with the main control group of usual care alone, the addition of GLP-1 RAs was associated with a decreased risk in 24% of the outcomes evaluated, and an increased risk in 10.86% of outcomes, with no significant difference for the remaining 65.14% of outcomes.
Of the various benefits, key improvements included a reduced risk for several substance use disorders including alcohol (hazard ratio [HR], 0.89) and opioid (HR, 0.87) use, suicidal ideation, attempt or intentional self-harm (HR, 0.90), seizures (HR, 0.90), neurocognitive disorders including Alzheimer disease (HR, 0.88) and dementia (HR, 0.92), coagulation and clotting disorders (HR, 0.92), and cardiac arrest (HR, 0.78).
Further benefits vs usual care alone included a reduced risk for infectious illnesses (HR, 0.88), acute kidney injury (HR, 0.88), and chronic kidney disease (CKD) (HR, 0.97; P <.05 for all the outcomes).
In terms of risks associated with GLP-1 RAs, in addition to the well-known risks for nausea and vomiting, additional increased risks vs usual care included gastrointestinal disorders such as noninfectious gastroenteritis (HR, 1.12), hypotension (HR, 1.06), arthritis (HR, 1.11), tendinitis and synovitis (HR, 1.10), interstitial nephritis (HR, 1.06), nephrolithiasis (HR, 1.15), and the known risk for drug-induced acute pancreatitis (HR, 2.46).
Neuropsychiatric Effects
Among the various benefits in the study, Al-Aly said some of the most intriguing are those involving the brain.
“I am struck by the consistent effects on many neuropsychiatric disorders — this aligns with data showing the presence of GLP-1 receptors in the brain and evidence showing that GLP-1s permeates through the blood brain barrier and acts on the brain to reduce inflammation and oxidative stress, improve neuroplasticity, etc.,” he said.
“Clearly, there is a neurotropic effect. There is also the possibility of an effect on the immune system/fighting infection — with reduced risks of infections, sepsis, etc.”
The reductions in suicidal ideation are encouraging after earlier reports of suicidal thoughts and self-injury among young users of GLP-1 RAs prompted concerns, including a 2023 review of the drug use by the European Medicines Agency that ultimately found no causal association, the authors added. The US Food and Drug Administration also found no association with GLP-1s and suicide risk.
The reductions in addictive behaviors are also encouraging and are consistent with the role of GLP-1 receptors in the brain in terms of impulse control and reward signaling that can relate to addictive behaviors, Al-Aly explained.
The reduced risks for dementia and Alzheimer disease are likewise consistent with preclinical studies in animal models of Alzheimer disease, as well as clinical studies showing a reduced risk for dementia in patients with type 2 diabetes, the authors noted.
The observed reduced risk for seizures further “adds to an emerging body of knowledge, both mechanistic and early clinical data, indicative of the anticonvulsant properties of GLP-1 RA use,” they added.
“GLP-1 RAs should be further evaluated in future studies as potential adjuvant therapeutics for epilepsy and its associated comorbidities,” the authors suggested.
Kidneys
While the findings support evidence of protective effects of GLP-1 RAs on the kidneys and a reduction in CKD risk, notable risks observed, also involving the kidneys, include nephrolithiasis or kidney stones.
Al-Aly noted the mechanisms with kidney stone formation are very different from CKD, and he speculated that the risk for the former could in fact stem from potentially low hydration with GLP-1 RA use.
“When patients are on GLP-1 RAs, they definitely eat a lot less to lose weight, but they also hydrate themselves less,” he explained in a press briefing. “They drink less water because they feel full very quickly after eating, and I’m just theorizing, but perhaps chronic dehydration [is behind] the increased risk of kidney stones.”
Modest Effects?
While, overall, the benefits of GLP-1 RA drugs showed modest benefits ranging between a 10% and 20% reduction for most outcomes, Al-Aly said those effects are still important.
“The modest effect does not negate the potential value of these drugs, especially for conditions where few effective treatment options exist, for example, dementia,” he said in the press statement.
“This may also imply that these drugs are most beneficial when used in conjunction with other interventions, such as lifestyle changes or other medications.”
Potential Confounders A Concern
Commenting on the study, David M. Nathan, MD, founder of the MGH Diabetes Center and a professor of medicine at Harvard Medical School, in Boston, Massachusetts, noted that, while the study is hypothesis-generating, the key limitation is its observational nature.
“The authors did a perfectly respectable job of doing all you can do to adjust for [confounders], but with these kinds of studies, as much as you try to statistically account for differences in the populations before they were put on the drug, you can never truly adjust for all the potential confounders that may influence the results,” he told this news organization.
In addition, the 3.8-year follow-up time of the study, as the authors acknowledge, is especially short considering that GLP-1 RAs are generally recommended to be taken indefinitely.
“You have to take these drugs presumably for a lifetime and we have no idea what the longer-term benefits and risks are,” Nathan said.
Nathan, who was among the first investigators to evaluate GLP-1 RAs about 30 years ago, underscored that “I do think that these drugs are generally really spectacular; they’ve taken over the world and they are probably the single greatest pharmaceutical story of the 21st century.”
“But much more rigorous randomized trials would be needed to prove study results that haven’t already been established in previous clinical trials,” he said.
“The types of [randomized] trials that are necessary are very expensive and require a huge amount of work, but at the end of the day, they provide proof as to what does and doesn’t work, and what the true risks are,” he added. “Whether the GLP-RAs will cure all ills and bring about world peace needs to be proved.”
In further comments provided through the Science Media Center, Stephen O’Rahilly, FRS, a professor of clinical biochemistry and medicine and director of the Wellcome-MRC Institute of Metabolic Science-Metabolic Research Laboratories, University of Cambridge, Cambridge, England, echoed Nathan’s concern that “studies such as these have to be interpreted very cautiously as the people studied have not been randomly allocated to GLP-1 RA treatment, so any difference between those taking and not taking the class of drug could potentially be attributable to factors other than the drug.”
He noted, however, that “the study provides useful reassurance about the safety of this class of drugs. The expected benefits on heart disease, stroke and other cardiovascular and most kidney diseases are clearly seen.”
Al-Aly reported being an uncompensated consultant for Pfizer. Nathan, who has previously conducted clinical trials on GLP-1 RAs, currently has no relationships to report. O’Rahilly reported receiving remuneration from several pharmaceutical companies for scientific advice relating to the development of drugs for metabolic diseases, but none involving GLP-1 RAs in the past 3 years.
A version of this article first appeared on Medscape.com.
A study of more than 2 million veterans with diabetes builds on evidence of broad-ranging benefits and risks of glucagon-like peptide 1 receptor agonists (GLP-1 RAs) in the clinical setting, providing an “atlas” mapping extensive outcomes and some new insights to potentially explore in more rigorous clinical trials.
“This is the largest study on GLP-1 receptor agonists,” first author Ziyad Al-Aly, MD, chief of research and development at the US Department of Veterans Affairs (VA) St. Louis Healthcare System, in St. Louis, told this news organization regarding the research, published this week, in Nature Medicine.
“The [study] reflects the real experiences of people using GLP-1 RAs [in the VA] clinical setting,” he said.
“Altogether, our discovery approach confirms previous studies and clinical trials and also uncovers previously unreported benefits and risks of GLP-1 RAs,” the authors wrote.
For the comprehensive study, Al-Aly and his colleagues evaluated data from the US Department of Veterans Affairs on more than 2 million veterans treated for diabetes between October 2017 and December 2023, assessing GLP-1 RA treatment in comparison with other diabetes therapies regarding a striking 175 clinical outcomes.
Of the patients, 215,970 initiated treatment with GLP-1 RAs; 159,465 started sulfonylureas, 117,989 dipeptidyl peptidase 4 inhibitors, and 258,614 were initiated on sodium-glucose cotransporter-2 inhibitors.
The study also included a composite group of the latter three drug groups (n = 536,068), and a control group of 1,203,097 of patients receiving usual care, who were compared with usual care with the addition of GLP-1 RAs.
After inverse probability weighting, the groups were well-balanced in terms of their baseline characteristics. While the majority in the VA cohort overall were White men, the study adjusted for gender, age, race, comorbidities, and an extensive array of covariates.
With an average follow-up of 3.68 years, after the multivariate adjustment, GLP-1 RAs showed “effectiveness and risks that extended beyond those currently recognized,” in comparison with each of the treatment groups and with the main control group of usual care, the authors reported.
For the largest comparison with the main control group of usual care alone, the addition of GLP-1 RAs was associated with a decreased risk in 24% of the outcomes evaluated, and an increased risk in 10.86% of outcomes, with no significant difference for the remaining 65.14% of outcomes.
Of the various benefits, key improvements included a reduced risk for several substance use disorders including alcohol (hazard ratio [HR], 0.89) and opioid (HR, 0.87) use, suicidal ideation, attempt or intentional self-harm (HR, 0.90), seizures (HR, 0.90), neurocognitive disorders including Alzheimer disease (HR, 0.88) and dementia (HR, 0.92), coagulation and clotting disorders (HR, 0.92), and cardiac arrest (HR, 0.78).
Further benefits vs usual care alone included a reduced risk for infectious illnesses (HR, 0.88), acute kidney injury (HR, 0.88), and chronic kidney disease (CKD) (HR, 0.97; P <.05 for all the outcomes).
In terms of risks associated with GLP-1 RAs, in addition to the well-known risks for nausea and vomiting, additional increased risks vs usual care included gastrointestinal disorders such as noninfectious gastroenteritis (HR, 1.12), hypotension (HR, 1.06), arthritis (HR, 1.11), tendinitis and synovitis (HR, 1.10), interstitial nephritis (HR, 1.06), nephrolithiasis (HR, 1.15), and the known risk for drug-induced acute pancreatitis (HR, 2.46).
Neuropsychiatric Effects
Among the various benefits in the study, Al-Aly said some of the most intriguing are those involving the brain.
“I am struck by the consistent effects on many neuropsychiatric disorders — this aligns with data showing the presence of GLP-1 receptors in the brain and evidence showing that GLP-1s permeates through the blood brain barrier and acts on the brain to reduce inflammation and oxidative stress, improve neuroplasticity, etc.,” he said.
“Clearly, there is a neurotropic effect. There is also the possibility of an effect on the immune system/fighting infection — with reduced risks of infections, sepsis, etc.”
The reductions in suicidal ideation are encouraging after earlier reports of suicidal thoughts and self-injury among young users of GLP-1 RAs prompted concerns, including a 2023 review of the drug use by the European Medicines Agency that ultimately found no causal association, the authors added. The US Food and Drug Administration also found no association with GLP-1s and suicide risk.
The reductions in addictive behaviors are also encouraging and are consistent with the role of GLP-1 receptors in the brain in terms of impulse control and reward signaling that can relate to addictive behaviors, Al-Aly explained.
The reduced risks for dementia and Alzheimer disease are likewise consistent with preclinical studies in animal models of Alzheimer disease, as well as clinical studies showing a reduced risk for dementia in patients with type 2 diabetes, the authors noted.
The observed reduced risk for seizures further “adds to an emerging body of knowledge, both mechanistic and early clinical data, indicative of the anticonvulsant properties of GLP-1 RA use,” they added.
“GLP-1 RAs should be further evaluated in future studies as potential adjuvant therapeutics for epilepsy and its associated comorbidities,” the authors suggested.
Kidneys
While the findings support evidence of protective effects of GLP-1 RAs on the kidneys and a reduction in CKD risk, notable risks observed, also involving the kidneys, include nephrolithiasis or kidney stones.
Al-Aly noted the mechanisms with kidney stone formation are very different from CKD, and he speculated that the risk for the former could in fact stem from potentially low hydration with GLP-1 RA use.
“When patients are on GLP-1 RAs, they definitely eat a lot less to lose weight, but they also hydrate themselves less,” he explained in a press briefing. “They drink less water because they feel full very quickly after eating, and I’m just theorizing, but perhaps chronic dehydration [is behind] the increased risk of kidney stones.”
Modest Effects?
While, overall, the benefits of GLP-1 RA drugs showed modest benefits ranging between a 10% and 20% reduction for most outcomes, Al-Aly said those effects are still important.
“The modest effect does not negate the potential value of these drugs, especially for conditions where few effective treatment options exist, for example, dementia,” he said in the press statement.
“This may also imply that these drugs are most beneficial when used in conjunction with other interventions, such as lifestyle changes or other medications.”
Potential Confounders A Concern
Commenting on the study, David M. Nathan, MD, founder of the MGH Diabetes Center and a professor of medicine at Harvard Medical School, in Boston, Massachusetts, noted that, while the study is hypothesis-generating, the key limitation is its observational nature.
“The authors did a perfectly respectable job of doing all you can do to adjust for [confounders], but with these kinds of studies, as much as you try to statistically account for differences in the populations before they were put on the drug, you can never truly adjust for all the potential confounders that may influence the results,” he told this news organization.
In addition, the 3.8-year follow-up time of the study, as the authors acknowledge, is especially short considering that GLP-1 RAs are generally recommended to be taken indefinitely.
“You have to take these drugs presumably for a lifetime and we have no idea what the longer-term benefits and risks are,” Nathan said.
Nathan, who was among the first investigators to evaluate GLP-1 RAs about 30 years ago, underscored that “I do think that these drugs are generally really spectacular; they’ve taken over the world and they are probably the single greatest pharmaceutical story of the 21st century.”
“But much more rigorous randomized trials would be needed to prove study results that haven’t already been established in previous clinical trials,” he said.
“The types of [randomized] trials that are necessary are very expensive and require a huge amount of work, but at the end of the day, they provide proof as to what does and doesn’t work, and what the true risks are,” he added. “Whether the GLP-RAs will cure all ills and bring about world peace needs to be proved.”
In further comments provided through the Science Media Center, Stephen O’Rahilly, FRS, a professor of clinical biochemistry and medicine and director of the Wellcome-MRC Institute of Metabolic Science-Metabolic Research Laboratories, University of Cambridge, Cambridge, England, echoed Nathan’s concern that “studies such as these have to be interpreted very cautiously as the people studied have not been randomly allocated to GLP-1 RA treatment, so any difference between those taking and not taking the class of drug could potentially be attributable to factors other than the drug.”
He noted, however, that “the study provides useful reassurance about the safety of this class of drugs. The expected benefits on heart disease, stroke and other cardiovascular and most kidney diseases are clearly seen.”
Al-Aly reported being an uncompensated consultant for Pfizer. Nathan, who has previously conducted clinical trials on GLP-1 RAs, currently has no relationships to report. O’Rahilly reported receiving remuneration from several pharmaceutical companies for scientific advice relating to the development of drugs for metabolic diseases, but none involving GLP-1 RAs in the past 3 years.
A version of this article first appeared on Medscape.com.
FROM NATURE MEDICINE
New Weight Loss Drugs May Fight Obesity-Related Cancer, Too
The latest glucagon-like peptide 1 (GLP-1) receptor agonists have been heralded for their potential to not only boost weight loss and glucose control but also improve cardiovascular, gastric, hepatic, and renal values.
Throughout 2024, research has also indicated GLP-1 drugs may reduce risks for obesity-related cancer.
In a US study of more than 1.6 million patients with type 2 diabetes, cancer researchers found that patients who took a GLP-1 drug had significant risk reductions for 10 of 13 obesity-associated cancers, as compared with patients who only took insulin.
They also saw a declining risk for stomach cancer, though it wasn’t considered statistically significant, but not a reduced risk for postmenopausal breast cancer or thyroid cancer.
The associations make sense, particularly because GLP-1 drugs have unexpected effects on modulating immune functions linked to obesity-associated cancers.
“The protective effects of GLP-1s against obesity-associated cancers likely stem from multiple mechanisms,” said lead author Lindsey Wang, a medical student and research scholar at Case Western Reserve University in Cleveland.
“These drugs promote substantial weight loss, reducing obesity-related cancer risks,” she said. “They also enhance insulin sensitivity and lower insulin levels, decreasing cancer cell growth signals.”
Additional GLP-1 Studies
The Case Western team also published a study in December 2023 that found people with type 2 diabetes who took GLP-1s had a 44% lower risk for colorectal cancer than those who took insulin and a 25% lower risk than those who took metformin. The research suggested even greater risk reductions among those with overweight or obesity, with GLP-1 users having a 50% lower risk than those who took insulin and a 42% lower risk than those who took metformin.
In another recent Case Western study, both bariatric surgery and GLP-1 drugs reduced the risk for obesity-related cancers. While those who had bariatric surgery had a 22% risk reduction over 10 years, as compared with those who received no treatment, those taking GLP-1 had a 39% risk reduction.
Other studies worldwide have looked at GLP-1 drugs and tumor effects among various cancer cell lines. In a study using pancreatic cancer cell lines, GLP-1 liraglutide suppressed cancer cell growth and led to cell death. Similarly, a study using breast cancer cells found liraglutide reduced cancer cell viability and the ability for cells to migrate.
As researchers identify additional links between GLP-1s and improvements across organ systems, the knock-on effects could lead to lower cancer risks as well. For example, studies presented at The Liver Meeting in San Diego in November pointed to GLP-1s reducing fatty liver disease, which can slow the progression to liver cancer.
“Separate from obesity, having higher levels of body fat is associated with an increased risk of several forms of cancer,” said Neil Iyengar, MD, an oncologist at Memorial Sloan Kettering Cancer Center in New York City. Iyengar researches the relationship between obesity and cancer.
“I foresee that this class of drugs will revolutionize obesity and the cancer burden that comes with it, if people can get access,” he said. “This really is an exciting development.”
Ongoing GLP-1 Research
On the other hand, cancer researchers have also expressed concerns about potential associations between GLP-1s and increased cancer risks. In the obesity-associated cancer study by Case Western researchers, patients with type 2 diabetes taking a GLP-1 drug appeared to have a slightly higher risk for kidney cancer than those taking metformin.
In addition, GLP-1 studies in animals have indicated that the drugs may increase the risks for medullary thyroid cancer and pancreatic cancer. However, the data on increased risks in humans remain inconclusive, and more recent studies refute these findings.
For instance, cancer researchers in India conducted a systematic review and meta-analysis of semaglutide and cancer risks, finding that 37 randomized controlled trials and 19 real-world studies didn’t find increased risks for any cancer, including pancreatic and thyroid cancers.
In another systematic review by Brazilian researchers, 50 trials found GLP-1s didn’t increase the risk for breast cancer or benign breast neoplasms.
In 2025, new retrospective studies will show more nuanced data, especially as more patients — both with and without type 2 diabetes — take semaglutide, tirzepatide, and new GLP-1 drugs in the research pipeline.
“The holy grail has always been getting a medication to treat obesity,” said Anne McTiernan, MD, PhD, an epidemiologist and obesity researcher at the Fred Hutchinson Cancer Center in Seattle.
“There have been trials focused on these medications’ effects on diabetes and cardiovascular disease treatment, but no trials have tested their effects on cancer risk,” she said. “Usually, many years of follow-up of large numbers of patients are needed to see cancer effects of a carcinogen or cancer-preventing intervention.”
Those clinical trials are likely coming soon, she said. Researchers will need to conduct prospective clinical trials to examine the direct relationship between GLP-1 drugs and cancer risks, as well as the underlying mechanisms linked to cancer cell growth, activation of immune cells, and anti-inflammatory properties.
Because GLP-1 medications aren’t intended to be taken forever, researchers will also need to consider the associations with long-term cancer risks. Even so, weight loss and other obesity-related improvements could contribute to overall lower cancer risks in the end.
“If taking these drugs for a limited amount of time can help people lose weight and get on an exercise plan, then that’s helping lower cancer risk long-term,” said Sonali Thosani, MD, associate professor of endocrine neoplasia and hormonal disorders at the University of Texas MD Anderson Cancer Center in Houston.
“But it all comes back to someone making lifestyle changes and sticking to them, even after they stop taking the drugs,” she said. “If they can do that, then you’ll probably see a net positive for long-term cancer risks and other long-term health risks.”
A version of this article appeared on Medscape.com.
The latest glucagon-like peptide 1 (GLP-1) receptor agonists have been heralded for their potential to not only boost weight loss and glucose control but also improve cardiovascular, gastric, hepatic, and renal values.
Throughout 2024, research has also indicated GLP-1 drugs may reduce risks for obesity-related cancer.
In a US study of more than 1.6 million patients with type 2 diabetes, cancer researchers found that patients who took a GLP-1 drug had significant risk reductions for 10 of 13 obesity-associated cancers, as compared with patients who only took insulin.
They also saw a declining risk for stomach cancer, though it wasn’t considered statistically significant, but not a reduced risk for postmenopausal breast cancer or thyroid cancer.
The associations make sense, particularly because GLP-1 drugs have unexpected effects on modulating immune functions linked to obesity-associated cancers.
“The protective effects of GLP-1s against obesity-associated cancers likely stem from multiple mechanisms,” said lead author Lindsey Wang, a medical student and research scholar at Case Western Reserve University in Cleveland.
“These drugs promote substantial weight loss, reducing obesity-related cancer risks,” she said. “They also enhance insulin sensitivity and lower insulin levels, decreasing cancer cell growth signals.”
Additional GLP-1 Studies
The Case Western team also published a study in December 2023 that found people with type 2 diabetes who took GLP-1s had a 44% lower risk for colorectal cancer than those who took insulin and a 25% lower risk than those who took metformin. The research suggested even greater risk reductions among those with overweight or obesity, with GLP-1 users having a 50% lower risk than those who took insulin and a 42% lower risk than those who took metformin.
In another recent Case Western study, both bariatric surgery and GLP-1 drugs reduced the risk for obesity-related cancers. While those who had bariatric surgery had a 22% risk reduction over 10 years, as compared with those who received no treatment, those taking GLP-1 had a 39% risk reduction.
Other studies worldwide have looked at GLP-1 drugs and tumor effects among various cancer cell lines. In a study using pancreatic cancer cell lines, GLP-1 liraglutide suppressed cancer cell growth and led to cell death. Similarly, a study using breast cancer cells found liraglutide reduced cancer cell viability and the ability for cells to migrate.
As researchers identify additional links between GLP-1s and improvements across organ systems, the knock-on effects could lead to lower cancer risks as well. For example, studies presented at The Liver Meeting in San Diego in November pointed to GLP-1s reducing fatty liver disease, which can slow the progression to liver cancer.
“Separate from obesity, having higher levels of body fat is associated with an increased risk of several forms of cancer,” said Neil Iyengar, MD, an oncologist at Memorial Sloan Kettering Cancer Center in New York City. Iyengar researches the relationship between obesity and cancer.
“I foresee that this class of drugs will revolutionize obesity and the cancer burden that comes with it, if people can get access,” he said. “This really is an exciting development.”
Ongoing GLP-1 Research
On the other hand, cancer researchers have also expressed concerns about potential associations between GLP-1s and increased cancer risks. In the obesity-associated cancer study by Case Western researchers, patients with type 2 diabetes taking a GLP-1 drug appeared to have a slightly higher risk for kidney cancer than those taking metformin.
In addition, GLP-1 studies in animals have indicated that the drugs may increase the risks for medullary thyroid cancer and pancreatic cancer. However, the data on increased risks in humans remain inconclusive, and more recent studies refute these findings.
For instance, cancer researchers in India conducted a systematic review and meta-analysis of semaglutide and cancer risks, finding that 37 randomized controlled trials and 19 real-world studies didn’t find increased risks for any cancer, including pancreatic and thyroid cancers.
In another systematic review by Brazilian researchers, 50 trials found GLP-1s didn’t increase the risk for breast cancer or benign breast neoplasms.
In 2025, new retrospective studies will show more nuanced data, especially as more patients — both with and without type 2 diabetes — take semaglutide, tirzepatide, and new GLP-1 drugs in the research pipeline.
“The holy grail has always been getting a medication to treat obesity,” said Anne McTiernan, MD, PhD, an epidemiologist and obesity researcher at the Fred Hutchinson Cancer Center in Seattle.
“There have been trials focused on these medications’ effects on diabetes and cardiovascular disease treatment, but no trials have tested their effects on cancer risk,” she said. “Usually, many years of follow-up of large numbers of patients are needed to see cancer effects of a carcinogen or cancer-preventing intervention.”
Those clinical trials are likely coming soon, she said. Researchers will need to conduct prospective clinical trials to examine the direct relationship between GLP-1 drugs and cancer risks, as well as the underlying mechanisms linked to cancer cell growth, activation of immune cells, and anti-inflammatory properties.
Because GLP-1 medications aren’t intended to be taken forever, researchers will also need to consider the associations with long-term cancer risks. Even so, weight loss and other obesity-related improvements could contribute to overall lower cancer risks in the end.
“If taking these drugs for a limited amount of time can help people lose weight and get on an exercise plan, then that’s helping lower cancer risk long-term,” said Sonali Thosani, MD, associate professor of endocrine neoplasia and hormonal disorders at the University of Texas MD Anderson Cancer Center in Houston.
“But it all comes back to someone making lifestyle changes and sticking to them, even after they stop taking the drugs,” she said. “If they can do that, then you’ll probably see a net positive for long-term cancer risks and other long-term health risks.”
A version of this article appeared on Medscape.com.
The latest glucagon-like peptide 1 (GLP-1) receptor agonists have been heralded for their potential to not only boost weight loss and glucose control but also improve cardiovascular, gastric, hepatic, and renal values.
Throughout 2024, research has also indicated GLP-1 drugs may reduce risks for obesity-related cancer.
In a US study of more than 1.6 million patients with type 2 diabetes, cancer researchers found that patients who took a GLP-1 drug had significant risk reductions for 10 of 13 obesity-associated cancers, as compared with patients who only took insulin.
They also saw a declining risk for stomach cancer, though it wasn’t considered statistically significant, but not a reduced risk for postmenopausal breast cancer or thyroid cancer.
The associations make sense, particularly because GLP-1 drugs have unexpected effects on modulating immune functions linked to obesity-associated cancers.
“The protective effects of GLP-1s against obesity-associated cancers likely stem from multiple mechanisms,” said lead author Lindsey Wang, a medical student and research scholar at Case Western Reserve University in Cleveland.
“These drugs promote substantial weight loss, reducing obesity-related cancer risks,” she said. “They also enhance insulin sensitivity and lower insulin levels, decreasing cancer cell growth signals.”
Additional GLP-1 Studies
The Case Western team also published a study in December 2023 that found people with type 2 diabetes who took GLP-1s had a 44% lower risk for colorectal cancer than those who took insulin and a 25% lower risk than those who took metformin. The research suggested even greater risk reductions among those with overweight or obesity, with GLP-1 users having a 50% lower risk than those who took insulin and a 42% lower risk than those who took metformin.
In another recent Case Western study, both bariatric surgery and GLP-1 drugs reduced the risk for obesity-related cancers. While those who had bariatric surgery had a 22% risk reduction over 10 years, as compared with those who received no treatment, those taking GLP-1 had a 39% risk reduction.
Other studies worldwide have looked at GLP-1 drugs and tumor effects among various cancer cell lines. In a study using pancreatic cancer cell lines, GLP-1 liraglutide suppressed cancer cell growth and led to cell death. Similarly, a study using breast cancer cells found liraglutide reduced cancer cell viability and the ability for cells to migrate.
As researchers identify additional links between GLP-1s and improvements across organ systems, the knock-on effects could lead to lower cancer risks as well. For example, studies presented at The Liver Meeting in San Diego in November pointed to GLP-1s reducing fatty liver disease, which can slow the progression to liver cancer.
“Separate from obesity, having higher levels of body fat is associated with an increased risk of several forms of cancer,” said Neil Iyengar, MD, an oncologist at Memorial Sloan Kettering Cancer Center in New York City. Iyengar researches the relationship between obesity and cancer.
“I foresee that this class of drugs will revolutionize obesity and the cancer burden that comes with it, if people can get access,” he said. “This really is an exciting development.”
Ongoing GLP-1 Research
On the other hand, cancer researchers have also expressed concerns about potential associations between GLP-1s and increased cancer risks. In the obesity-associated cancer study by Case Western researchers, patients with type 2 diabetes taking a GLP-1 drug appeared to have a slightly higher risk for kidney cancer than those taking metformin.
In addition, GLP-1 studies in animals have indicated that the drugs may increase the risks for medullary thyroid cancer and pancreatic cancer. However, the data on increased risks in humans remain inconclusive, and more recent studies refute these findings.
For instance, cancer researchers in India conducted a systematic review and meta-analysis of semaglutide and cancer risks, finding that 37 randomized controlled trials and 19 real-world studies didn’t find increased risks for any cancer, including pancreatic and thyroid cancers.
In another systematic review by Brazilian researchers, 50 trials found GLP-1s didn’t increase the risk for breast cancer or benign breast neoplasms.
In 2025, new retrospective studies will show more nuanced data, especially as more patients — both with and without type 2 diabetes — take semaglutide, tirzepatide, and new GLP-1 drugs in the research pipeline.
“The holy grail has always been getting a medication to treat obesity,” said Anne McTiernan, MD, PhD, an epidemiologist and obesity researcher at the Fred Hutchinson Cancer Center in Seattle.
“There have been trials focused on these medications’ effects on diabetes and cardiovascular disease treatment, but no trials have tested their effects on cancer risk,” she said. “Usually, many years of follow-up of large numbers of patients are needed to see cancer effects of a carcinogen or cancer-preventing intervention.”
Those clinical trials are likely coming soon, she said. Researchers will need to conduct prospective clinical trials to examine the direct relationship between GLP-1 drugs and cancer risks, as well as the underlying mechanisms linked to cancer cell growth, activation of immune cells, and anti-inflammatory properties.
Because GLP-1 medications aren’t intended to be taken forever, researchers will also need to consider the associations with long-term cancer risks. Even so, weight loss and other obesity-related improvements could contribute to overall lower cancer risks in the end.
“If taking these drugs for a limited amount of time can help people lose weight and get on an exercise plan, then that’s helping lower cancer risk long-term,” said Sonali Thosani, MD, associate professor of endocrine neoplasia and hormonal disorders at the University of Texas MD Anderson Cancer Center in Houston.
“But it all comes back to someone making lifestyle changes and sticking to them, even after they stop taking the drugs,” she said. “If they can do that, then you’ll probably see a net positive for long-term cancer risks and other long-term health risks.”
A version of this article appeared on Medscape.com.
High Radon Levels Linked to Gestational Diabetes
New data link higher county-level radon exposure to gestational diabetes (GD) in women who haven’t previously given birth, emphasizing the need to consider environmental risks in maternal and fetal healthcare.
Yijia Zhang, PhD, with the Department of Obstetrics and Gynecology, Vagelos College of Physicians and Surgeons at Columbia University Irving Medical Center in New York, and colleagues found in a study of 9107 nulliparous pregnant women that those living in US counties with higher radon levels (2 picocuries [pCi]/L) had higher odds of developing GD than those in counties with lower (< 1 pCi/L) radon levels (odds ratio [OR], 1.37; 95% CI, 1.02-1.84.) The researchers used three radon categories, and the middle level was 1 to < 2 pCi/L.
Findings were published online on January 10 in JAMA Network Open. The researchers used data from The Nulliparous Pregnancy Outcomes Study: Monitoring Mothers-to-Be (nuMoM2b), a multicenter, prospective cohort study that examines factors associated with pregnancy-related outcomes.
“To our knowledge, this is the first study to examine the association between radon exposure and the risk of GD,” the authors wrote.
GD Affects 10% of Pregnancies
GD affects about 10% of pregnancies every year in the United States, according to the Centers for Disease Control and Prevention, and can affect women and offspring long term as it raises mothers’ risk of type 2 diabetes and cardiovascular disease and raises the risk for childhood obesity. Radon exposure’s link with lung cancer risk has been well established, but its link to other health risks is uncertain, the authors note.
The authors said their findings are hypothesis-generating and said, “It is vital to conduct studies that incorporate individual-level indoor radon exposure data,” to get closer to understanding the underlying mechanisms.
Individual-Level Exposure Measures Needed
They note that the average radon level in a county might not reflect an individual’s exposure and individual-level residential factors involved with radon exposure, such as household mitigation, and whether a dwelling has a basement, for instance, “are crucial for enhancing the precision of exposure assessment.”
In an invited commentary, Alberto Ruano-Ravina, PhD, and Lucía Martín-Gisbert, MSc, both with the Department of Preventive Medicine and Public Health at the University of Santiago de Compostela in Galicia, Spain, also urged that individual-level studies be conducted to further investigate radon’s link to health risks, noting that “[r]adon is possibly the most prevalent indoor carcinogen to which human beings are exposed.”
“There is no reason for not having these studies once we have some evidence of an association from ecological studies,” they wrote. They point out that reliable radon assessments are easy and inexpensive.
“The potential association of radon exposure with gestational diabetes or any other disease should be better analyzed using exclusively radon-prone areas. An observance of a dose-response effect may be indicative of a causal relationship, and it could be easily evidenced in radon-prone areas should such a relationship exist,” the commenters wrote.
Such areas have low, medium, high, and extremely high concentration levels, the commenters wrote. Zhang’s team, they point out, had to use only three exposure levels because the number of residents in high-exposure areas (exceeding 3 pCi/L) was too small.
“It is time now to move forward and really understand the full implications of radon exposure for health,” they concluded.
One coauthor reported serving on the board of directors for Merck for Mothers and as a board member for March for Moms outside the submitted work. One coauthor reported grants from the National Heart, Lung, and Blood Institute and the National Institutes of Health (NIH) during the conduct of the study. Four coauthors reported grants from the NIH during the conduct of the study. One coauthor reported grants from the NIH during the conduct of the study and being a cofounder of Naima Health and receiving personal fees from Organon outside the submitted work. Both commenters reported no relevant financial disclosures.
A version of this article appeared on Medscape.com.
New data link higher county-level radon exposure to gestational diabetes (GD) in women who haven’t previously given birth, emphasizing the need to consider environmental risks in maternal and fetal healthcare.
Yijia Zhang, PhD, with the Department of Obstetrics and Gynecology, Vagelos College of Physicians and Surgeons at Columbia University Irving Medical Center in New York, and colleagues found in a study of 9107 nulliparous pregnant women that those living in US counties with higher radon levels (2 picocuries [pCi]/L) had higher odds of developing GD than those in counties with lower (< 1 pCi/L) radon levels (odds ratio [OR], 1.37; 95% CI, 1.02-1.84.) The researchers used three radon categories, and the middle level was 1 to < 2 pCi/L.
Findings were published online on January 10 in JAMA Network Open. The researchers used data from The Nulliparous Pregnancy Outcomes Study: Monitoring Mothers-to-Be (nuMoM2b), a multicenter, prospective cohort study that examines factors associated with pregnancy-related outcomes.
“To our knowledge, this is the first study to examine the association between radon exposure and the risk of GD,” the authors wrote.
GD Affects 10% of Pregnancies
GD affects about 10% of pregnancies every year in the United States, according to the Centers for Disease Control and Prevention, and can affect women and offspring long term as it raises mothers’ risk of type 2 diabetes and cardiovascular disease and raises the risk for childhood obesity. Radon exposure’s link with lung cancer risk has been well established, but its link to other health risks is uncertain, the authors note.
The authors said their findings are hypothesis-generating and said, “It is vital to conduct studies that incorporate individual-level indoor radon exposure data,” to get closer to understanding the underlying mechanisms.
Individual-Level Exposure Measures Needed
They note that the average radon level in a county might not reflect an individual’s exposure and individual-level residential factors involved with radon exposure, such as household mitigation, and whether a dwelling has a basement, for instance, “are crucial for enhancing the precision of exposure assessment.”
In an invited commentary, Alberto Ruano-Ravina, PhD, and Lucía Martín-Gisbert, MSc, both with the Department of Preventive Medicine and Public Health at the University of Santiago de Compostela in Galicia, Spain, also urged that individual-level studies be conducted to further investigate radon’s link to health risks, noting that “[r]adon is possibly the most prevalent indoor carcinogen to which human beings are exposed.”
“There is no reason for not having these studies once we have some evidence of an association from ecological studies,” they wrote. They point out that reliable radon assessments are easy and inexpensive.
“The potential association of radon exposure with gestational diabetes or any other disease should be better analyzed using exclusively radon-prone areas. An observance of a dose-response effect may be indicative of a causal relationship, and it could be easily evidenced in radon-prone areas should such a relationship exist,” the commenters wrote.
Such areas have low, medium, high, and extremely high concentration levels, the commenters wrote. Zhang’s team, they point out, had to use only three exposure levels because the number of residents in high-exposure areas (exceeding 3 pCi/L) was too small.
“It is time now to move forward and really understand the full implications of radon exposure for health,” they concluded.
One coauthor reported serving on the board of directors for Merck for Mothers and as a board member for March for Moms outside the submitted work. One coauthor reported grants from the National Heart, Lung, and Blood Institute and the National Institutes of Health (NIH) during the conduct of the study. Four coauthors reported grants from the NIH during the conduct of the study. One coauthor reported grants from the NIH during the conduct of the study and being a cofounder of Naima Health and receiving personal fees from Organon outside the submitted work. Both commenters reported no relevant financial disclosures.
A version of this article appeared on Medscape.com.
New data link higher county-level radon exposure to gestational diabetes (GD) in women who haven’t previously given birth, emphasizing the need to consider environmental risks in maternal and fetal healthcare.
Yijia Zhang, PhD, with the Department of Obstetrics and Gynecology, Vagelos College of Physicians and Surgeons at Columbia University Irving Medical Center in New York, and colleagues found in a study of 9107 nulliparous pregnant women that those living in US counties with higher radon levels (2 picocuries [pCi]/L) had higher odds of developing GD than those in counties with lower (< 1 pCi/L) radon levels (odds ratio [OR], 1.37; 95% CI, 1.02-1.84.) The researchers used three radon categories, and the middle level was 1 to < 2 pCi/L.
Findings were published online on January 10 in JAMA Network Open. The researchers used data from The Nulliparous Pregnancy Outcomes Study: Monitoring Mothers-to-Be (nuMoM2b), a multicenter, prospective cohort study that examines factors associated with pregnancy-related outcomes.
“To our knowledge, this is the first study to examine the association between radon exposure and the risk of GD,” the authors wrote.
GD Affects 10% of Pregnancies
GD affects about 10% of pregnancies every year in the United States, according to the Centers for Disease Control and Prevention, and can affect women and offspring long term as it raises mothers’ risk of type 2 diabetes and cardiovascular disease and raises the risk for childhood obesity. Radon exposure’s link with lung cancer risk has been well established, but its link to other health risks is uncertain, the authors note.
The authors said their findings are hypothesis-generating and said, “It is vital to conduct studies that incorporate individual-level indoor radon exposure data,” to get closer to understanding the underlying mechanisms.
Individual-Level Exposure Measures Needed
They note that the average radon level in a county might not reflect an individual’s exposure and individual-level residential factors involved with radon exposure, such as household mitigation, and whether a dwelling has a basement, for instance, “are crucial for enhancing the precision of exposure assessment.”
In an invited commentary, Alberto Ruano-Ravina, PhD, and Lucía Martín-Gisbert, MSc, both with the Department of Preventive Medicine and Public Health at the University of Santiago de Compostela in Galicia, Spain, also urged that individual-level studies be conducted to further investigate radon’s link to health risks, noting that “[r]adon is possibly the most prevalent indoor carcinogen to which human beings are exposed.”
“There is no reason for not having these studies once we have some evidence of an association from ecological studies,” they wrote. They point out that reliable radon assessments are easy and inexpensive.
“The potential association of radon exposure with gestational diabetes or any other disease should be better analyzed using exclusively radon-prone areas. An observance of a dose-response effect may be indicative of a causal relationship, and it could be easily evidenced in radon-prone areas should such a relationship exist,” the commenters wrote.
Such areas have low, medium, high, and extremely high concentration levels, the commenters wrote. Zhang’s team, they point out, had to use only three exposure levels because the number of residents in high-exposure areas (exceeding 3 pCi/L) was too small.
“It is time now to move forward and really understand the full implications of radon exposure for health,” they concluded.
One coauthor reported serving on the board of directors for Merck for Mothers and as a board member for March for Moms outside the submitted work. One coauthor reported grants from the National Heart, Lung, and Blood Institute and the National Institutes of Health (NIH) during the conduct of the study. Four coauthors reported grants from the NIH during the conduct of the study. One coauthor reported grants from the NIH during the conduct of the study and being a cofounder of Naima Health and receiving personal fees from Organon outside the submitted work. Both commenters reported no relevant financial disclosures.
A version of this article appeared on Medscape.com.
Nutrition, Drugs, or Bariatric Surgery: What’s the Best Approach for Sustained Weight Loss?
Given that more than 100 million US adults have obesity, including 22 million with severe obesity, physicians regularly see patients with the condition in their practices.
Fortunately, doctors have more tools than ever to help their patients. But the question remains: Which method is the safest and most effective? Is it diet and lifestyle changes, one of the recently approved anti-obesity medications (AOMs), bariatric surgery, or a combination approach?
There are no head-to-head trials comparing these three approaches, said Vanita Rahman, MD, clinic director of the Barnard Medical Center, Washington, DC, at the International Conference on Nutrition in Medicine, sponsored by the Physicians Committee for Responsible Medicine.
Instead, doctors must evaluate the merits and drawbacks of each intervention and decide with their patients which treatment is best for them, she told Medscape Medical News. When she sees patients, Rahman shares the pertinent research with them, so they are able to make an informed choice.
Looking at the Options
In her presentation at the conference, Rahman summarized the guidelines issued by the American Heart Association/American College of Cardiology/The Obesity Society for Management of Overweight and Obesity in Adults and the American Association of Clinical Endocrinologists and American College of Endocrinology Comprehensive Clinical Practice Guidelines For Medical Care of Patients with Obesity, including lifestyle changes, AOMs, and bariatric surgery (Table 1).
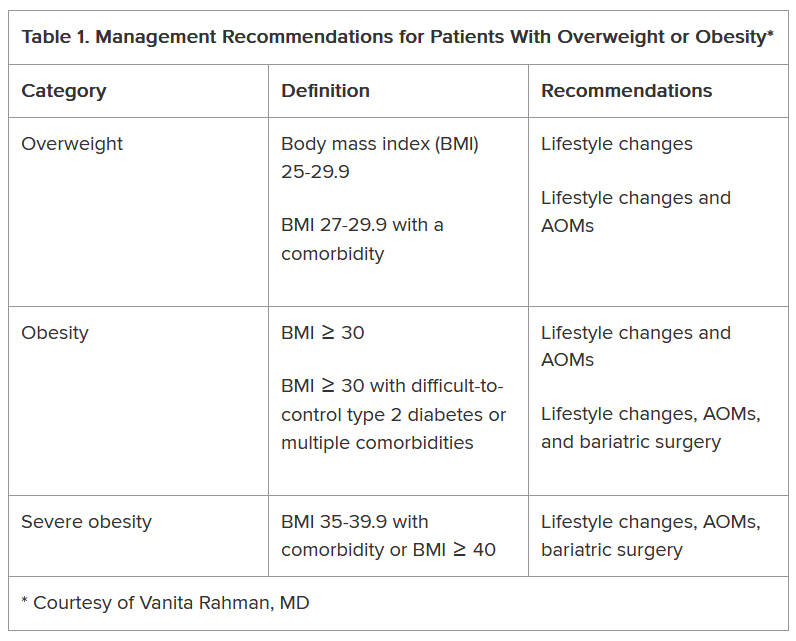
As shown, the current clinical guidelines offer recommendations that consider such factors as the patient’s BMI and presence of one or more comorbidities. Generally, they begin with lifestyle changes for people with overweight, the possibility of an AOM for those with obesity, and bariatric surgery as an option for those with severe obesity-related complications.
“In obesity, we traditionally thought the process was ‘either-or’ — either lifestyle or surgery or medication — and somehow lifestyle is better,” Sheethal Reddy, PhD, a psychologist at the Bariatric Center at Emory University Hospital Midtown, Atlanta, told Medscape Medical News.
Now physicians often use a combination of methods, but lifestyle is foundational to all of them, she said.
“If you don’t make lifestyle changes, none of the approaches will ultimately be effective,” said Reddy, who also is an assistant professor in the Division of General and GI Surgery at Emory School of Medicine, Atlanta.
Lifestyle changes don’t just involve diet and nutrition but include physical exercise.
“Being sedentary affects everything — sleep quality, appetite regulation, and metabolism. Without sufficient exercise, the body isn’t functioning well enough to have a healthy metabolism,” Reddy said.
How Durable Are the Interventions?
Although bariatric surgery has demonstrated effectiveness in helping patients lose weight, many of them regain some or most of it, Rahman said.
A systematic review and meta-analysis found weight regain in 49% of patients who underwent bariatric surgery patients, with the highest prevalence after Roux-en-Y gastric bypass.
Another study of approximately 45,000 patients who underwent bariatric surgery found differences not only in the percentage of total weight loss among Roux-en-Y gastric bypass, sleeve gastrectomy, and adjustable gastric band procedures but also in how much of that weight stayed off between 1 and 5 years following the procedure (Table 2).
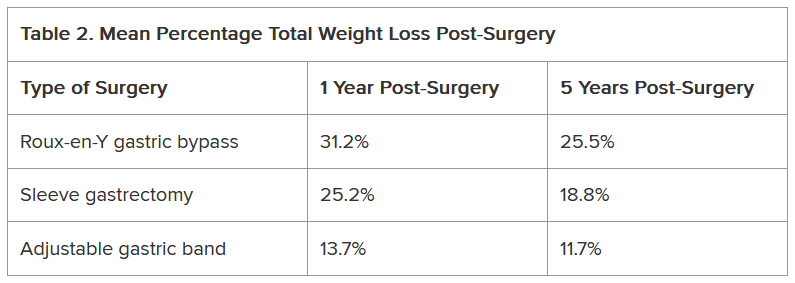
Weight regain also is a risk with AOMs, if they’re discontinued.
The STEP 1 trial tested the effectiveness of semaglutide — a glucagon-like peptide 1 (GLP-1) receptor agonist — as an adjunct to lifestyle intervention for weight loss in patients with obesity or with overweight and at least one comorbidity but not diabetes. Mean weight loss with semaglutide was 17.3% but that figure dropped 11.6 percentage points after treatment was discontinued.
Other studies also have found that patients regain weight after GLP-1 discontinuation.
Tirzepatide, a GLP-1 and glucose-dependent insulinotropic polypeptide (GIP) combination, has shown efficacy with weight reduction, but patients experienced some weight regain upon discontinuation. In one study, patients experienced a mean weight loss of 20.9% after 36 weeks of tirzepatide. In the study’s subsequent 52-week double-blind, placebo-controlled period, patients who stopped taking the medication experienced a weight regain of 14%, whereas those who remained on the medication lost an additional 5.5% of weight.
GLP-1 and GLP-1/GIP medications do not address the factors that contribute to overweight and obesity, Rahman said. “They simply suppress the appetite; therefore, weight gain occurs after stopping them.”
Patients may stop taking anti-obesity drugs for a variety of reasons, including side effects. Rahman noted that the common side effects include nausea, vomiting, and constipation, whereas rare side effects include gastroparesis, gallbladder and biliary disease, thyroid cancer, and suicidal thoughts. GLP-1 and GLP-1/GIP medications also carry a risk for non-arteritic anterior ischemic optic neuropathy, she said.
Moreover, health insurance does not always cover these medications, which likely affects patient access to the drugs and compliance rates.
“Given the side effects and frequent lack of insurance coverage, significant questions remain about long-term safety and feasibility of these agents,” Rahman said.
What About Nutritional Approaches?
The lifestyle interventions in the semaglutide and tirzepatide studies included 500 kcal/d deficit diets, which is difficult for people to maintain, noted Rahman, who is the author of the book Simply Plant Based: Fabulous Food for a Healthy Life.
Additionally, bariatric surgery has been associated with long-term micronutrient deficiencies, including deficiencies in vitamins A, D, E, K, B1, and B12, as well as folate, iron, zinc, copper, selenium, and calcium, she said.
The best approach to food from a patient compliance standpoint and to avoid nutrient deficiencies is a whole-food, plant-based diet, Rahman said. She advocates this nutritional approach, along with physical activity, for patients regardless of whether they’ve selected lifestyle intervention alone or combined with an AOM or bariatric surgery to address obesity.
Rahman cited a 5-year heart disease study comparing an intensive lifestyle program involving a vegetarian diet, aerobic exercise, stress management training, smoking cessation, and group psychosocial support to treatment as usual. Patients in the lifestyle group lost 10.9 kg at 1 year and sustained weight loss of 5.8 kg at 5 years, whereas weight in the control group remained relatively unchanged from baseline.
She also pointed to the findings of a study of patients with obesity or with overweight and at least one comorbidity that compared standard care with a low-fat, whole-food, plant-based diet with vitamin B12 supplementation. At 6 months, mean BMI reduction was greater in the intervention group than the standard care group (−4.4 vs −0.4).
In her practice, Rahman has seen the benefits of a whole-food, plant-based diet for patients with obesity.
If people are committed to this type of dietary approach and are given the tools and resources to do it effectively, “their thinking changes, their taste buds change, and they grow to enjoy this new way of eating,” she said. “They see results, and it’s a lifestyle that can be sustained long-term.”
Addressing Drivers of Weight Gain
Patients also need help addressing the various factors that may contribute to overweight and obesity, including overconsumption of ultra-processed foods, substandard nutritional quality of restaurant foods, increasing portion sizes, distraction during eating, emotional eating, late-night eating, and cultural/traditional values surrounding food, Rahman noted.
Supatra Tovar, PsyD, RD, a clinical psychologist with a practice in Pasadena, California, agreed that identifying the reasons for weight gain is critical for treatment.
“If you’re not addressing underlying issues, such as a person’s relationship with food, behaviors around food, the tendency to mindlessly eat or emotionally eat or eat to seek comfort, the person’s weight problems won’t ultimately be fully solved by any of the three approaches — dieting, medications, or bariatric surgery,” she said.
Some of her patients “engage in extreme dieting and deprivation, and many who use medications or have had bariatric surgery hardly eat and often develop nutritional deficiencies,” said Tovar, author of the book Deprogram Diet Culture: Rethink Your Relationship with Food, Heal Your Mind, and Live a Diet-Free Life.
The key to healthy and sustained weight loss is to “become attuned to the body’s signals, learn how to honor hunger, stop eating when satisfied, and eat more healthful foods, such as fruits and vegetables, whole grains, lean proteins — especially plant-based proteins — and the body gives signals that this is what it wants,” she said.
Tovar doesn’t give her clients a specific diet or set of portions.
“I teach them to listen to their bodies,” she said. “They’ve lost significant amounts of weight and continued to keep it off because they’ve done this kind of work.”
When Lifestyle Changes Aren’t Enough
For many patients, lifestyle interventions are insufficient to address the degree of overweight and obesity and common comorbidities, said W. Timothy Garvey, MD, associate director and professor, Department of Nutrition Sciences, School of Health Professions, University of Alabama at Birmingham.
“Of course, nutritional approaches are very important, not only for weight but also for general health-related reasons,” said Garvey, lead author of the 2016 American Association of Clinical Endocrinologists obesity guidelines. “We’ve seen that the Mediterranean and some plant-based diets can prevent progression from prediabetes to diabetes and improve other parameters that reflect metabolic health.”
However, it’s “not common that patients can follow these diets, lose weight, and keep it off,” Garvey cautioned. Up to 50% of weight that’s lost through lifestyle changes is typically regained by 1-year follow-up, with almost all remaining lost weight subsequently regained in the majority of individuals because the person “has to fight against pathophysiological process that drive weight regain,” he noted.
Weight-loss medications can address these pathophysiologic processes by “addressing interactions of satiety hormones with feeding centers in the brain, suppressing the appetite, and making it easier for patients to adhere to a reduced-calorie diet.”
Garvey views the weight-loss medications in the same light as drugs for diabetes and hypertension, in that people need to keep taking them to sustain the benefit.
There’s still a role for bariatric surgery because not everyone can tolerate the AOMs or achieve sufficient weight loss.
“Patients with very high BMI who have trouble ambulating might benefit from a combination of bariatric surgery and medication,” Garvey said.
While some side effects are associated with AOMs, being an “alarmist” about them can be detrimental to patients, he warned.
Rahman and Tovar are authors of books about weight loss. Reddy reported no relevant financial relationships. Garvey is a consultant on advisory boards for Boehringer Ingelheim, Eli Lilly, Novo Nordisk, Pfizer, Fractyl Health, Alnylam Pharmaceuticals, Inogen, Zealand, Allurion, Carmot/Roche, Terns Pharmaceuticals, Neurocrine, Keros Therapeutics, and Regeneron. He is the site principal investigator for multi-centered clinical trials sponsored by his university and funded by Novo Nordisk, Eli Lilly, Epitomee, Neurovalens, and Pfizer. He serves as a consultant on the advisory board for the nonprofit Milken Foundation and is a member of the Data Monitoring Committee for phase 3 clinical trials conducted by Boehringer-Ingelheim and Eli Lilly.
A version of this article first appeared on Medscape.com.
Given that more than 100 million US adults have obesity, including 22 million with severe obesity, physicians regularly see patients with the condition in their practices.
Fortunately, doctors have more tools than ever to help their patients. But the question remains: Which method is the safest and most effective? Is it diet and lifestyle changes, one of the recently approved anti-obesity medications (AOMs), bariatric surgery, or a combination approach?
There are no head-to-head trials comparing these three approaches, said Vanita Rahman, MD, clinic director of the Barnard Medical Center, Washington, DC, at the International Conference on Nutrition in Medicine, sponsored by the Physicians Committee for Responsible Medicine.
Instead, doctors must evaluate the merits and drawbacks of each intervention and decide with their patients which treatment is best for them, she told Medscape Medical News. When she sees patients, Rahman shares the pertinent research with them, so they are able to make an informed choice.
Looking at the Options
In her presentation at the conference, Rahman summarized the guidelines issued by the American Heart Association/American College of Cardiology/The Obesity Society for Management of Overweight and Obesity in Adults and the American Association of Clinical Endocrinologists and American College of Endocrinology Comprehensive Clinical Practice Guidelines For Medical Care of Patients with Obesity, including lifestyle changes, AOMs, and bariatric surgery (Table 1).

As shown, the current clinical guidelines offer recommendations that consider such factors as the patient’s BMI and presence of one or more comorbidities. Generally, they begin with lifestyle changes for people with overweight, the possibility of an AOM for those with obesity, and bariatric surgery as an option for those with severe obesity-related complications.
“In obesity, we traditionally thought the process was ‘either-or’ — either lifestyle or surgery or medication — and somehow lifestyle is better,” Sheethal Reddy, PhD, a psychologist at the Bariatric Center at Emory University Hospital Midtown, Atlanta, told Medscape Medical News.
Now physicians often use a combination of methods, but lifestyle is foundational to all of them, she said.
“If you don’t make lifestyle changes, none of the approaches will ultimately be effective,” said Reddy, who also is an assistant professor in the Division of General and GI Surgery at Emory School of Medicine, Atlanta.
Lifestyle changes don’t just involve diet and nutrition but include physical exercise.
“Being sedentary affects everything — sleep quality, appetite regulation, and metabolism. Without sufficient exercise, the body isn’t functioning well enough to have a healthy metabolism,” Reddy said.
How Durable Are the Interventions?
Although bariatric surgery has demonstrated effectiveness in helping patients lose weight, many of them regain some or most of it, Rahman said.
A systematic review and meta-analysis found weight regain in 49% of patients who underwent bariatric surgery patients, with the highest prevalence after Roux-en-Y gastric bypass.
Another study of approximately 45,000 patients who underwent bariatric surgery found differences not only in the percentage of total weight loss among Roux-en-Y gastric bypass, sleeve gastrectomy, and adjustable gastric band procedures but also in how much of that weight stayed off between 1 and 5 years following the procedure (Table 2).

Weight regain also is a risk with AOMs, if they’re discontinued.
The STEP 1 trial tested the effectiveness of semaglutide — a glucagon-like peptide 1 (GLP-1) receptor agonist — as an adjunct to lifestyle intervention for weight loss in patients with obesity or with overweight and at least one comorbidity but not diabetes. Mean weight loss with semaglutide was 17.3% but that figure dropped 11.6 percentage points after treatment was discontinued.
Other studies also have found that patients regain weight after GLP-1 discontinuation.
Tirzepatide, a GLP-1 and glucose-dependent insulinotropic polypeptide (GIP) combination, has shown efficacy with weight reduction, but patients experienced some weight regain upon discontinuation. In one study, patients experienced a mean weight loss of 20.9% after 36 weeks of tirzepatide. In the study’s subsequent 52-week double-blind, placebo-controlled period, patients who stopped taking the medication experienced a weight regain of 14%, whereas those who remained on the medication lost an additional 5.5% of weight.
GLP-1 and GLP-1/GIP medications do not address the factors that contribute to overweight and obesity, Rahman said. “They simply suppress the appetite; therefore, weight gain occurs after stopping them.”
Patients may stop taking anti-obesity drugs for a variety of reasons, including side effects. Rahman noted that the common side effects include nausea, vomiting, and constipation, whereas rare side effects include gastroparesis, gallbladder and biliary disease, thyroid cancer, and suicidal thoughts. GLP-1 and GLP-1/GIP medications also carry a risk for non-arteritic anterior ischemic optic neuropathy, she said.
Moreover, health insurance does not always cover these medications, which likely affects patient access to the drugs and compliance rates.
“Given the side effects and frequent lack of insurance coverage, significant questions remain about long-term safety and feasibility of these agents,” Rahman said.
What About Nutritional Approaches?
The lifestyle interventions in the semaglutide and tirzepatide studies included 500 kcal/d deficit diets, which is difficult for people to maintain, noted Rahman, who is the author of the book Simply Plant Based: Fabulous Food for a Healthy Life.
Additionally, bariatric surgery has been associated with long-term micronutrient deficiencies, including deficiencies in vitamins A, D, E, K, B1, and B12, as well as folate, iron, zinc, copper, selenium, and calcium, she said.
The best approach to food from a patient compliance standpoint and to avoid nutrient deficiencies is a whole-food, plant-based diet, Rahman said. She advocates this nutritional approach, along with physical activity, for patients regardless of whether they’ve selected lifestyle intervention alone or combined with an AOM or bariatric surgery to address obesity.
Rahman cited a 5-year heart disease study comparing an intensive lifestyle program involving a vegetarian diet, aerobic exercise, stress management training, smoking cessation, and group psychosocial support to treatment as usual. Patients in the lifestyle group lost 10.9 kg at 1 year and sustained weight loss of 5.8 kg at 5 years, whereas weight in the control group remained relatively unchanged from baseline.
She also pointed to the findings of a study of patients with obesity or with overweight and at least one comorbidity that compared standard care with a low-fat, whole-food, plant-based diet with vitamin B12 supplementation. At 6 months, mean BMI reduction was greater in the intervention group than the standard care group (−4.4 vs −0.4).
In her practice, Rahman has seen the benefits of a whole-food, plant-based diet for patients with obesity.
If people are committed to this type of dietary approach and are given the tools and resources to do it effectively, “their thinking changes, their taste buds change, and they grow to enjoy this new way of eating,” she said. “They see results, and it’s a lifestyle that can be sustained long-term.”
Addressing Drivers of Weight Gain
Patients also need help addressing the various factors that may contribute to overweight and obesity, including overconsumption of ultra-processed foods, substandard nutritional quality of restaurant foods, increasing portion sizes, distraction during eating, emotional eating, late-night eating, and cultural/traditional values surrounding food, Rahman noted.
Supatra Tovar, PsyD, RD, a clinical psychologist with a practice in Pasadena, California, agreed that identifying the reasons for weight gain is critical for treatment.
“If you’re not addressing underlying issues, such as a person’s relationship with food, behaviors around food, the tendency to mindlessly eat or emotionally eat or eat to seek comfort, the person’s weight problems won’t ultimately be fully solved by any of the three approaches — dieting, medications, or bariatric surgery,” she said.
Some of her patients “engage in extreme dieting and deprivation, and many who use medications or have had bariatric surgery hardly eat and often develop nutritional deficiencies,” said Tovar, author of the book Deprogram Diet Culture: Rethink Your Relationship with Food, Heal Your Mind, and Live a Diet-Free Life.
The key to healthy and sustained weight loss is to “become attuned to the body’s signals, learn how to honor hunger, stop eating when satisfied, and eat more healthful foods, such as fruits and vegetables, whole grains, lean proteins — especially plant-based proteins — and the body gives signals that this is what it wants,” she said.
Tovar doesn’t give her clients a specific diet or set of portions.
“I teach them to listen to their bodies,” she said. “They’ve lost significant amounts of weight and continued to keep it off because they’ve done this kind of work.”
When Lifestyle Changes Aren’t Enough
For many patients, lifestyle interventions are insufficient to address the degree of overweight and obesity and common comorbidities, said W. Timothy Garvey, MD, associate director and professor, Department of Nutrition Sciences, School of Health Professions, University of Alabama at Birmingham.
“Of course, nutritional approaches are very important, not only for weight but also for general health-related reasons,” said Garvey, lead author of the 2016 American Association of Clinical Endocrinologists obesity guidelines. “We’ve seen that the Mediterranean and some plant-based diets can prevent progression from prediabetes to diabetes and improve other parameters that reflect metabolic health.”
However, it’s “not common that patients can follow these diets, lose weight, and keep it off,” Garvey cautioned. Up to 50% of weight that’s lost through lifestyle changes is typically regained by 1-year follow-up, with almost all remaining lost weight subsequently regained in the majority of individuals because the person “has to fight against pathophysiological process that drive weight regain,” he noted.
Weight-loss medications can address these pathophysiologic processes by “addressing interactions of satiety hormones with feeding centers in the brain, suppressing the appetite, and making it easier for patients to adhere to a reduced-calorie diet.”
Garvey views the weight-loss medications in the same light as drugs for diabetes and hypertension, in that people need to keep taking them to sustain the benefit.
There’s still a role for bariatric surgery because not everyone can tolerate the AOMs or achieve sufficient weight loss.
“Patients with very high BMI who have trouble ambulating might benefit from a combination of bariatric surgery and medication,” Garvey said.
While some side effects are associated with AOMs, being an “alarmist” about them can be detrimental to patients, he warned.
Rahman and Tovar are authors of books about weight loss. Reddy reported no relevant financial relationships. Garvey is a consultant on advisory boards for Boehringer Ingelheim, Eli Lilly, Novo Nordisk, Pfizer, Fractyl Health, Alnylam Pharmaceuticals, Inogen, Zealand, Allurion, Carmot/Roche, Terns Pharmaceuticals, Neurocrine, Keros Therapeutics, and Regeneron. He is the site principal investigator for multi-centered clinical trials sponsored by his university and funded by Novo Nordisk, Eli Lilly, Epitomee, Neurovalens, and Pfizer. He serves as a consultant on the advisory board for the nonprofit Milken Foundation and is a member of the Data Monitoring Committee for phase 3 clinical trials conducted by Boehringer-Ingelheim and Eli Lilly.
A version of this article first appeared on Medscape.com.
Given that more than 100 million US adults have obesity, including 22 million with severe obesity, physicians regularly see patients with the condition in their practices.
Fortunately, doctors have more tools than ever to help their patients. But the question remains: Which method is the safest and most effective? Is it diet and lifestyle changes, one of the recently approved anti-obesity medications (AOMs), bariatric surgery, or a combination approach?
There are no head-to-head trials comparing these three approaches, said Vanita Rahman, MD, clinic director of the Barnard Medical Center, Washington, DC, at the International Conference on Nutrition in Medicine, sponsored by the Physicians Committee for Responsible Medicine.
Instead, doctors must evaluate the merits and drawbacks of each intervention and decide with their patients which treatment is best for them, she told Medscape Medical News. When she sees patients, Rahman shares the pertinent research with them, so they are able to make an informed choice.
Looking at the Options
In her presentation at the conference, Rahman summarized the guidelines issued by the American Heart Association/American College of Cardiology/The Obesity Society for Management of Overweight and Obesity in Adults and the American Association of Clinical Endocrinologists and American College of Endocrinology Comprehensive Clinical Practice Guidelines For Medical Care of Patients with Obesity, including lifestyle changes, AOMs, and bariatric surgery (Table 1).

As shown, the current clinical guidelines offer recommendations that consider such factors as the patient’s BMI and presence of one or more comorbidities. Generally, they begin with lifestyle changes for people with overweight, the possibility of an AOM for those with obesity, and bariatric surgery as an option for those with severe obesity-related complications.
“In obesity, we traditionally thought the process was ‘either-or’ — either lifestyle or surgery or medication — and somehow lifestyle is better,” Sheethal Reddy, PhD, a psychologist at the Bariatric Center at Emory University Hospital Midtown, Atlanta, told Medscape Medical News.
Now physicians often use a combination of methods, but lifestyle is foundational to all of them, she said.
“If you don’t make lifestyle changes, none of the approaches will ultimately be effective,” said Reddy, who also is an assistant professor in the Division of General and GI Surgery at Emory School of Medicine, Atlanta.
Lifestyle changes don’t just involve diet and nutrition but include physical exercise.
“Being sedentary affects everything — sleep quality, appetite regulation, and metabolism. Without sufficient exercise, the body isn’t functioning well enough to have a healthy metabolism,” Reddy said.
How Durable Are the Interventions?
Although bariatric surgery has demonstrated effectiveness in helping patients lose weight, many of them regain some or most of it, Rahman said.
A systematic review and meta-analysis found weight regain in 49% of patients who underwent bariatric surgery patients, with the highest prevalence after Roux-en-Y gastric bypass.
Another study of approximately 45,000 patients who underwent bariatric surgery found differences not only in the percentage of total weight loss among Roux-en-Y gastric bypass, sleeve gastrectomy, and adjustable gastric band procedures but also in how much of that weight stayed off between 1 and 5 years following the procedure (Table 2).

Weight regain also is a risk with AOMs, if they’re discontinued.
The STEP 1 trial tested the effectiveness of semaglutide — a glucagon-like peptide 1 (GLP-1) receptor agonist — as an adjunct to lifestyle intervention for weight loss in patients with obesity or with overweight and at least one comorbidity but not diabetes. Mean weight loss with semaglutide was 17.3% but that figure dropped 11.6 percentage points after treatment was discontinued.
Other studies also have found that patients regain weight after GLP-1 discontinuation.
Tirzepatide, a GLP-1 and glucose-dependent insulinotropic polypeptide (GIP) combination, has shown efficacy with weight reduction, but patients experienced some weight regain upon discontinuation. In one study, patients experienced a mean weight loss of 20.9% after 36 weeks of tirzepatide. In the study’s subsequent 52-week double-blind, placebo-controlled period, patients who stopped taking the medication experienced a weight regain of 14%, whereas those who remained on the medication lost an additional 5.5% of weight.
GLP-1 and GLP-1/GIP medications do not address the factors that contribute to overweight and obesity, Rahman said. “They simply suppress the appetite; therefore, weight gain occurs after stopping them.”
Patients may stop taking anti-obesity drugs for a variety of reasons, including side effects. Rahman noted that the common side effects include nausea, vomiting, and constipation, whereas rare side effects include gastroparesis, gallbladder and biliary disease, thyroid cancer, and suicidal thoughts. GLP-1 and GLP-1/GIP medications also carry a risk for non-arteritic anterior ischemic optic neuropathy, she said.
Moreover, health insurance does not always cover these medications, which likely affects patient access to the drugs and compliance rates.
“Given the side effects and frequent lack of insurance coverage, significant questions remain about long-term safety and feasibility of these agents,” Rahman said.
What About Nutritional Approaches?
The lifestyle interventions in the semaglutide and tirzepatide studies included 500 kcal/d deficit diets, which is difficult for people to maintain, noted Rahman, who is the author of the book Simply Plant Based: Fabulous Food for a Healthy Life.
Additionally, bariatric surgery has been associated with long-term micronutrient deficiencies, including deficiencies in vitamins A, D, E, K, B1, and B12, as well as folate, iron, zinc, copper, selenium, and calcium, she said.
The best approach to food from a patient compliance standpoint and to avoid nutrient deficiencies is a whole-food, plant-based diet, Rahman said. She advocates this nutritional approach, along with physical activity, for patients regardless of whether they’ve selected lifestyle intervention alone or combined with an AOM or bariatric surgery to address obesity.
Rahman cited a 5-year heart disease study comparing an intensive lifestyle program involving a vegetarian diet, aerobic exercise, stress management training, smoking cessation, and group psychosocial support to treatment as usual. Patients in the lifestyle group lost 10.9 kg at 1 year and sustained weight loss of 5.8 kg at 5 years, whereas weight in the control group remained relatively unchanged from baseline.
She also pointed to the findings of a study of patients with obesity or with overweight and at least one comorbidity that compared standard care with a low-fat, whole-food, plant-based diet with vitamin B12 supplementation. At 6 months, mean BMI reduction was greater in the intervention group than the standard care group (−4.4 vs −0.4).
In her practice, Rahman has seen the benefits of a whole-food, plant-based diet for patients with obesity.
If people are committed to this type of dietary approach and are given the tools and resources to do it effectively, “their thinking changes, their taste buds change, and they grow to enjoy this new way of eating,” she said. “They see results, and it’s a lifestyle that can be sustained long-term.”
Addressing Drivers of Weight Gain
Patients also need help addressing the various factors that may contribute to overweight and obesity, including overconsumption of ultra-processed foods, substandard nutritional quality of restaurant foods, increasing portion sizes, distraction during eating, emotional eating, late-night eating, and cultural/traditional values surrounding food, Rahman noted.
Supatra Tovar, PsyD, RD, a clinical psychologist with a practice in Pasadena, California, agreed that identifying the reasons for weight gain is critical for treatment.
“If you’re not addressing underlying issues, such as a person’s relationship with food, behaviors around food, the tendency to mindlessly eat or emotionally eat or eat to seek comfort, the person’s weight problems won’t ultimately be fully solved by any of the three approaches — dieting, medications, or bariatric surgery,” she said.
Some of her patients “engage in extreme dieting and deprivation, and many who use medications or have had bariatric surgery hardly eat and often develop nutritional deficiencies,” said Tovar, author of the book Deprogram Diet Culture: Rethink Your Relationship with Food, Heal Your Mind, and Live a Diet-Free Life.
The key to healthy and sustained weight loss is to “become attuned to the body’s signals, learn how to honor hunger, stop eating when satisfied, and eat more healthful foods, such as fruits and vegetables, whole grains, lean proteins — especially plant-based proteins — and the body gives signals that this is what it wants,” she said.
Tovar doesn’t give her clients a specific diet or set of portions.
“I teach them to listen to their bodies,” she said. “They’ve lost significant amounts of weight and continued to keep it off because they’ve done this kind of work.”
When Lifestyle Changes Aren’t Enough
For many patients, lifestyle interventions are insufficient to address the degree of overweight and obesity and common comorbidities, said W. Timothy Garvey, MD, associate director and professor, Department of Nutrition Sciences, School of Health Professions, University of Alabama at Birmingham.
“Of course, nutritional approaches are very important, not only for weight but also for general health-related reasons,” said Garvey, lead author of the 2016 American Association of Clinical Endocrinologists obesity guidelines. “We’ve seen that the Mediterranean and some plant-based diets can prevent progression from prediabetes to diabetes and improve other parameters that reflect metabolic health.”
However, it’s “not common that patients can follow these diets, lose weight, and keep it off,” Garvey cautioned. Up to 50% of weight that’s lost through lifestyle changes is typically regained by 1-year follow-up, with almost all remaining lost weight subsequently regained in the majority of individuals because the person “has to fight against pathophysiological process that drive weight regain,” he noted.
Weight-loss medications can address these pathophysiologic processes by “addressing interactions of satiety hormones with feeding centers in the brain, suppressing the appetite, and making it easier for patients to adhere to a reduced-calorie diet.”
Garvey views the weight-loss medications in the same light as drugs for diabetes and hypertension, in that people need to keep taking them to sustain the benefit.
There’s still a role for bariatric surgery because not everyone can tolerate the AOMs or achieve sufficient weight loss.
“Patients with very high BMI who have trouble ambulating might benefit from a combination of bariatric surgery and medication,” Garvey said.
While some side effects are associated with AOMs, being an “alarmist” about them can be detrimental to patients, he warned.
Rahman and Tovar are authors of books about weight loss. Reddy reported no relevant financial relationships. Garvey is a consultant on advisory boards for Boehringer Ingelheim, Eli Lilly, Novo Nordisk, Pfizer, Fractyl Health, Alnylam Pharmaceuticals, Inogen, Zealand, Allurion, Carmot/Roche, Terns Pharmaceuticals, Neurocrine, Keros Therapeutics, and Regeneron. He is the site principal investigator for multi-centered clinical trials sponsored by his university and funded by Novo Nordisk, Eli Lilly, Epitomee, Neurovalens, and Pfizer. He serves as a consultant on the advisory board for the nonprofit Milken Foundation and is a member of the Data Monitoring Committee for phase 3 clinical trials conducted by Boehringer-Ingelheim and Eli Lilly.
A version of this article first appeared on Medscape.com.
Are Patients On GLP-1s Getting the Right Nutrients?
As the use of glucagon-like peptide 1 receptor agonists (GLP-1 RAs) continues to exponentially expand obesity treatment, concerns have arisen regarding their impact on nutrition in people who take them.
While the medications’ dampening effects on appetite result in an average weight reduction ≥ 15%, they also pose a risk for malnutrition.
“It’s important to eat a balanced diet when taking these medications,” Deena Adimoolam, MD, an endocrinologist based in New York City and a member of the national advisory committees for the Endocrine Society and the American Diabetes Association, said in an interview.
The decreased caloric intake resulting from the use of GLP-1 RAs makes it essential for patients to consume nutrient-dense foods. Clinicians can help patients achieve a healthy diet by anticipating nutrition problems, advising them on recommended target ranges of nutrient intake, and referring them for appropriate counseling.
Where to Begin
The task begins with “setting the right expectations before the patient starts treatment,” said Scott Isaacs, MD, president-elect of the American Association of Clinical Endocrinology.
To that end, it’s important to explain to patients how the medications affect appetite and how to adapt. GLP-1 RAs don’t completely turn off the appetite, and the effect at the beginning will likely be very mild, Isaacs said in an interview.
Some patients don’t notice a change for 2-3 months, although others see an effect sooner.
“Typically, people will notice that the main impact is on satiation, meaning they’ll fill up more quickly,” said Isaacs, who is an adjunct associate professor at Emory University School of Medicine, Atlanta, Georgia. “It’s important to tell them to stop eating when they feel full because eating when full can increase the side effects, such as nausea, vomiting, diarrhea, and constipation.”
A review article, written by lead author Jaime Almandoz, MD, University of Texas Southwestern Medical Center, Dallas, in Obesity offers a “5 A’s model” as a guide on how to begin discussing overweight or obesity with patients. This involves asking for permission to discuss weight and asking about food and vitamin/supplement intake; assessing the patient’s medical history and root causes of obesity, and conducting a physical examination; advising the patient regarding treatment options and reasonable expectations; agreeing on treatment and lifestyle goals; and assisting the patient to address challenges, referring them as needed to for additional support (eg, a dietitian), as well as arranging for follow-up.
Impact of GLP-1 RAs on Food Preferences
Besides reducing hunger and increasing satiety, GLP-1 RAs may affect food preferences, according to a research review published in The International Journal of Obesity. It cites a 2014 study that found that people taking GLP-1 RAs displayed decreased neuronal responses to images of food measured by functional magnetic resonance imaging in the areas of brain associated with appetite and reward. This might affect taste preferences and food intake.
Additionally, a 2023 study suggested that during the weight-loss phase of treatment (as opposed to the maintenance phase), patients may experience reduced cravings for dairy and starchy food, less desire to eat salty or spicy foods, and less difficulty controlling eating and resisting cravings.
“Altered food preferences, decreased food cravings, and reduced food intake may contribute to long-term weight loss,” according to the research review. Tailored treatments focusing on the weight maintenance phase are needed, the authors wrote.
Are Patients Vulnerable to Malnutrition?
A recent review found that total caloric intake was reduced by 16%-39% in patients taking a GLP-1 RA or dual glucose-dependent insulinotropic polypeptide (GIP)/GLP-1 RA, but few studies evaluated the composition of these patients’ diets. Research that examines the qualitative changes in macronutrient and micronutrient intake of patients on these medications is needed, the authors wrote.
They outlined several nutritional concerns, including whether GLP-1 RA or GIP/ GLP-1 RA use could result in protein intake insufficient for maintaining muscle strength, mass, and function or in inadequate dietary quality (ie, poor intake of micronutrients, fiber, and fluid).
“Although we don’t necessarily see ‘malnutrition’ in our practice, we do see patients who lose too much weight after months and months of treatment, patients who aren’t hungry and don’t eat all day and have one big meal at the end of the day because they don’t feel like eating, and people who continue to eat unhealthy foods,” Isaacs said.
Some patients, however, have medical histories placing them at a greater risk for malnutrition. “Identification of these individuals may help prevent more serious nutritional and medical complications that might occur with decreased food intake associated with AOMs [anti-obesity medications],” Almandoz and colleagues noted in their review.
What Should Patients Eat?
Nutritional needs vary based on the patient’s age, sex, body weight, physical activity, and other factors, Almandoz and colleagues wrote. For this reason, energy intake during weight loss should be “personalized.”
The authors also recommended specific sources of the various dietary components and noted red flags signaling potential deficiencies
Nutritional needs vary based on the degree of appetite suppression in the patient, Adimoolam said. “I recommend at least two servings of fruits and vegetables daily, and drinking plenty of water throughout the day,” she added.
Protein in particular is a “key macronutrient,” and insufficient intake can lead to a variety of adverse effects, including sarcopenia — which is already a concern in individuals being treated with GLP-1 RAs. Meal replacement products (eg, shakes or bars) can supplement diets to help meet protein needs, especially if appetite is significantly reduced.
“There are definitely concerns for sarcopenia, so we have our patients taking these drugs try to eat healthy lean proteins – 100 g/d — and exercise,” Isaacs said. Exercise, including resistance training, not only improves muscle mass but also potentiates the effects of the GLP-1 RAs in patients with obesity and type 2 diabetes.
Adequate hydration is essential for patients taking GLP-1 RAs. “One of the commonly described side effects is fatigue, but there’s no biological reason why these medications should cause fatigue. My opinion is that these patients are dehydrated, and that may be causing the fatigue,” Isaacs said.
Some patients taking GLP-1 RAs lose interest in food. Isaacs regarded this as an “adverse reaction to the medication, which necessitates either stopping it altogether, changing the dose, or adjusting the diet.” There are “many different solutions, and one size doesn’t fit all,” he said.
Dietary and Behavioral Counseling
The drugs don’t necessarily motivate a person to eat healthier food, only to eat less food, Isaacs noted.
“The person might be eating low-volume but high-calorie food, such as bag of chips or a cookie instead of an apple,” Isaacs said. Patients who are losing weight “may not realize that weight loss isn’t the only important outcome. Because they’re losing weight, they think it’s okay to eat junk food.”
Patients need education and guidance about how to eat while on these medications. Most patients find counseling about meal planning helpful, he said.
Isaacs gives nutritional guidance to his patients when he prescribes a weight loss medication. “But most physicians don’t have time to offer that type of specific counseling on an ongoing basis,” he said. Isaacs refers patients requiring more detailed and long-term guidance to a dietitian.
Patients with monotonous diets of poor quality are at increased risk for nutrition deficiencies, and counseling by a registered dietitian could help improve their dietary quality.
Registered dietitians can develop a multifaceted approach not only focusing on medication management but also on customizing the patient’s diet, assisting with lifestyle adjustments, and addressing the mental health issues surrounding obesity and its management.
People seeking obesity treatment often have psychiatric conditions, psychological distress, or disordered eating patterns, and questions and concerns have emerged about how GLP-1 RA use might affect existing mental health problems. For example, if the medication suppresses the feeling of gratification a person once got from eating high-energy dense foods, that individual may “seek rewards or pleasure elsewhere, and possibly from unhealthy sources.”
Psychological issues also may emerge as a result of weight loss, so it’s helpful to take a multidisciplinary approach that includes mental health practitioners to support patients who are being treated with GLP-1 RAs. Patients taking these agents should be monitored for the emergence or worsening of psychiatric conditions, such as depression and suicidal ideation.
Achieving significant weight loss may lead to “unexpected changes” in the dynamics of patients’ relationship with others, “which can be distressing.” Clinicians should be “sensitive to patients’ social and emotional needs” and provide support or refer patients for help with coping strategies.
GLP-1 RAs have enormous potential to improve health outcomes in patients with obesity. Careful patient selection, close monitoring, and support for patients with nutrition and other lifestyle issues can increase the chances that these agents will fulfill their potential.
Isaacs declared no relevant financial relationships.
A version of this article appeared on Medscape.com.
As the use of glucagon-like peptide 1 receptor agonists (GLP-1 RAs) continues to exponentially expand obesity treatment, concerns have arisen regarding their impact on nutrition in people who take them.
While the medications’ dampening effects on appetite result in an average weight reduction ≥ 15%, they also pose a risk for malnutrition.
“It’s important to eat a balanced diet when taking these medications,” Deena Adimoolam, MD, an endocrinologist based in New York City and a member of the national advisory committees for the Endocrine Society and the American Diabetes Association, said in an interview.
The decreased caloric intake resulting from the use of GLP-1 RAs makes it essential for patients to consume nutrient-dense foods. Clinicians can help patients achieve a healthy diet by anticipating nutrition problems, advising them on recommended target ranges of nutrient intake, and referring them for appropriate counseling.
Where to Begin
The task begins with “setting the right expectations before the patient starts treatment,” said Scott Isaacs, MD, president-elect of the American Association of Clinical Endocrinology.
To that end, it’s important to explain to patients how the medications affect appetite and how to adapt. GLP-1 RAs don’t completely turn off the appetite, and the effect at the beginning will likely be very mild, Isaacs said in an interview.
Some patients don’t notice a change for 2-3 months, although others see an effect sooner.
“Typically, people will notice that the main impact is on satiation, meaning they’ll fill up more quickly,” said Isaacs, who is an adjunct associate professor at Emory University School of Medicine, Atlanta, Georgia. “It’s important to tell them to stop eating when they feel full because eating when full can increase the side effects, such as nausea, vomiting, diarrhea, and constipation.”
A review article, written by lead author Jaime Almandoz, MD, University of Texas Southwestern Medical Center, Dallas, in Obesity offers a “5 A’s model” as a guide on how to begin discussing overweight or obesity with patients. This involves asking for permission to discuss weight and asking about food and vitamin/supplement intake; assessing the patient’s medical history and root causes of obesity, and conducting a physical examination; advising the patient regarding treatment options and reasonable expectations; agreeing on treatment and lifestyle goals; and assisting the patient to address challenges, referring them as needed to for additional support (eg, a dietitian), as well as arranging for follow-up.
Impact of GLP-1 RAs on Food Preferences
Besides reducing hunger and increasing satiety, GLP-1 RAs may affect food preferences, according to a research review published in The International Journal of Obesity. It cites a 2014 study that found that people taking GLP-1 RAs displayed decreased neuronal responses to images of food measured by functional magnetic resonance imaging in the areas of brain associated with appetite and reward. This might affect taste preferences and food intake.
Additionally, a 2023 study suggested that during the weight-loss phase of treatment (as opposed to the maintenance phase), patients may experience reduced cravings for dairy and starchy food, less desire to eat salty or spicy foods, and less difficulty controlling eating and resisting cravings.
“Altered food preferences, decreased food cravings, and reduced food intake may contribute to long-term weight loss,” according to the research review. Tailored treatments focusing on the weight maintenance phase are needed, the authors wrote.
Are Patients Vulnerable to Malnutrition?
A recent review found that total caloric intake was reduced by 16%-39% in patients taking a GLP-1 RA or dual glucose-dependent insulinotropic polypeptide (GIP)/GLP-1 RA, but few studies evaluated the composition of these patients’ diets. Research that examines the qualitative changes in macronutrient and micronutrient intake of patients on these medications is needed, the authors wrote.
They outlined several nutritional concerns, including whether GLP-1 RA or GIP/ GLP-1 RA use could result in protein intake insufficient for maintaining muscle strength, mass, and function or in inadequate dietary quality (ie, poor intake of micronutrients, fiber, and fluid).
“Although we don’t necessarily see ‘malnutrition’ in our practice, we do see patients who lose too much weight after months and months of treatment, patients who aren’t hungry and don’t eat all day and have one big meal at the end of the day because they don’t feel like eating, and people who continue to eat unhealthy foods,” Isaacs said.
Some patients, however, have medical histories placing them at a greater risk for malnutrition. “Identification of these individuals may help prevent more serious nutritional and medical complications that might occur with decreased food intake associated with AOMs [anti-obesity medications],” Almandoz and colleagues noted in their review.
What Should Patients Eat?
Nutritional needs vary based on the patient’s age, sex, body weight, physical activity, and other factors, Almandoz and colleagues wrote. For this reason, energy intake during weight loss should be “personalized.”
The authors also recommended specific sources of the various dietary components and noted red flags signaling potential deficiencies
Nutritional needs vary based on the degree of appetite suppression in the patient, Adimoolam said. “I recommend at least two servings of fruits and vegetables daily, and drinking plenty of water throughout the day,” she added.
Protein in particular is a “key macronutrient,” and insufficient intake can lead to a variety of adverse effects, including sarcopenia — which is already a concern in individuals being treated with GLP-1 RAs. Meal replacement products (eg, shakes or bars) can supplement diets to help meet protein needs, especially if appetite is significantly reduced.
“There are definitely concerns for sarcopenia, so we have our patients taking these drugs try to eat healthy lean proteins – 100 g/d — and exercise,” Isaacs said. Exercise, including resistance training, not only improves muscle mass but also potentiates the effects of the GLP-1 RAs in patients with obesity and type 2 diabetes.
Adequate hydration is essential for patients taking GLP-1 RAs. “One of the commonly described side effects is fatigue, but there’s no biological reason why these medications should cause fatigue. My opinion is that these patients are dehydrated, and that may be causing the fatigue,” Isaacs said.
Some patients taking GLP-1 RAs lose interest in food. Isaacs regarded this as an “adverse reaction to the medication, which necessitates either stopping it altogether, changing the dose, or adjusting the diet.” There are “many different solutions, and one size doesn’t fit all,” he said.
Dietary and Behavioral Counseling
The drugs don’t necessarily motivate a person to eat healthier food, only to eat less food, Isaacs noted.
“The person might be eating low-volume but high-calorie food, such as bag of chips or a cookie instead of an apple,” Isaacs said. Patients who are losing weight “may not realize that weight loss isn’t the only important outcome. Because they’re losing weight, they think it’s okay to eat junk food.”
Patients need education and guidance about how to eat while on these medications. Most patients find counseling about meal planning helpful, he said.
Isaacs gives nutritional guidance to his patients when he prescribes a weight loss medication. “But most physicians don’t have time to offer that type of specific counseling on an ongoing basis,” he said. Isaacs refers patients requiring more detailed and long-term guidance to a dietitian.
Patients with monotonous diets of poor quality are at increased risk for nutrition deficiencies, and counseling by a registered dietitian could help improve their dietary quality.
Registered dietitians can develop a multifaceted approach not only focusing on medication management but also on customizing the patient’s diet, assisting with lifestyle adjustments, and addressing the mental health issues surrounding obesity and its management.
People seeking obesity treatment often have psychiatric conditions, psychological distress, or disordered eating patterns, and questions and concerns have emerged about how GLP-1 RA use might affect existing mental health problems. For example, if the medication suppresses the feeling of gratification a person once got from eating high-energy dense foods, that individual may “seek rewards or pleasure elsewhere, and possibly from unhealthy sources.”
Psychological issues also may emerge as a result of weight loss, so it’s helpful to take a multidisciplinary approach that includes mental health practitioners to support patients who are being treated with GLP-1 RAs. Patients taking these agents should be monitored for the emergence or worsening of psychiatric conditions, such as depression and suicidal ideation.
Achieving significant weight loss may lead to “unexpected changes” in the dynamics of patients’ relationship with others, “which can be distressing.” Clinicians should be “sensitive to patients’ social and emotional needs” and provide support or refer patients for help with coping strategies.
GLP-1 RAs have enormous potential to improve health outcomes in patients with obesity. Careful patient selection, close monitoring, and support for patients with nutrition and other lifestyle issues can increase the chances that these agents will fulfill their potential.
Isaacs declared no relevant financial relationships.
A version of this article appeared on Medscape.com.
As the use of glucagon-like peptide 1 receptor agonists (GLP-1 RAs) continues to exponentially expand obesity treatment, concerns have arisen regarding their impact on nutrition in people who take them.
While the medications’ dampening effects on appetite result in an average weight reduction ≥ 15%, they also pose a risk for malnutrition.
“It’s important to eat a balanced diet when taking these medications,” Deena Adimoolam, MD, an endocrinologist based in New York City and a member of the national advisory committees for the Endocrine Society and the American Diabetes Association, said in an interview.
The decreased caloric intake resulting from the use of GLP-1 RAs makes it essential for patients to consume nutrient-dense foods. Clinicians can help patients achieve a healthy diet by anticipating nutrition problems, advising them on recommended target ranges of nutrient intake, and referring them for appropriate counseling.
Where to Begin
The task begins with “setting the right expectations before the patient starts treatment,” said Scott Isaacs, MD, president-elect of the American Association of Clinical Endocrinology.
To that end, it’s important to explain to patients how the medications affect appetite and how to adapt. GLP-1 RAs don’t completely turn off the appetite, and the effect at the beginning will likely be very mild, Isaacs said in an interview.
Some patients don’t notice a change for 2-3 months, although others see an effect sooner.
“Typically, people will notice that the main impact is on satiation, meaning they’ll fill up more quickly,” said Isaacs, who is an adjunct associate professor at Emory University School of Medicine, Atlanta, Georgia. “It’s important to tell them to stop eating when they feel full because eating when full can increase the side effects, such as nausea, vomiting, diarrhea, and constipation.”
A review article, written by lead author Jaime Almandoz, MD, University of Texas Southwestern Medical Center, Dallas, in Obesity offers a “5 A’s model” as a guide on how to begin discussing overweight or obesity with patients. This involves asking for permission to discuss weight and asking about food and vitamin/supplement intake; assessing the patient’s medical history and root causes of obesity, and conducting a physical examination; advising the patient regarding treatment options and reasonable expectations; agreeing on treatment and lifestyle goals; and assisting the patient to address challenges, referring them as needed to for additional support (eg, a dietitian), as well as arranging for follow-up.
Impact of GLP-1 RAs on Food Preferences
Besides reducing hunger and increasing satiety, GLP-1 RAs may affect food preferences, according to a research review published in The International Journal of Obesity. It cites a 2014 study that found that people taking GLP-1 RAs displayed decreased neuronal responses to images of food measured by functional magnetic resonance imaging in the areas of brain associated with appetite and reward. This might affect taste preferences and food intake.
Additionally, a 2023 study suggested that during the weight-loss phase of treatment (as opposed to the maintenance phase), patients may experience reduced cravings for dairy and starchy food, less desire to eat salty or spicy foods, and less difficulty controlling eating and resisting cravings.
“Altered food preferences, decreased food cravings, and reduced food intake may contribute to long-term weight loss,” according to the research review. Tailored treatments focusing on the weight maintenance phase are needed, the authors wrote.
Are Patients Vulnerable to Malnutrition?
A recent review found that total caloric intake was reduced by 16%-39% in patients taking a GLP-1 RA or dual glucose-dependent insulinotropic polypeptide (GIP)/GLP-1 RA, but few studies evaluated the composition of these patients’ diets. Research that examines the qualitative changes in macronutrient and micronutrient intake of patients on these medications is needed, the authors wrote.
They outlined several nutritional concerns, including whether GLP-1 RA or GIP/ GLP-1 RA use could result in protein intake insufficient for maintaining muscle strength, mass, and function or in inadequate dietary quality (ie, poor intake of micronutrients, fiber, and fluid).
“Although we don’t necessarily see ‘malnutrition’ in our practice, we do see patients who lose too much weight after months and months of treatment, patients who aren’t hungry and don’t eat all day and have one big meal at the end of the day because they don’t feel like eating, and people who continue to eat unhealthy foods,” Isaacs said.
Some patients, however, have medical histories placing them at a greater risk for malnutrition. “Identification of these individuals may help prevent more serious nutritional and medical complications that might occur with decreased food intake associated with AOMs [anti-obesity medications],” Almandoz and colleagues noted in their review.
What Should Patients Eat?
Nutritional needs vary based on the patient’s age, sex, body weight, physical activity, and other factors, Almandoz and colleagues wrote. For this reason, energy intake during weight loss should be “personalized.”
The authors also recommended specific sources of the various dietary components and noted red flags signaling potential deficiencies
Nutritional needs vary based on the degree of appetite suppression in the patient, Adimoolam said. “I recommend at least two servings of fruits and vegetables daily, and drinking plenty of water throughout the day,” she added.
Protein in particular is a “key macronutrient,” and insufficient intake can lead to a variety of adverse effects, including sarcopenia — which is already a concern in individuals being treated with GLP-1 RAs. Meal replacement products (eg, shakes or bars) can supplement diets to help meet protein needs, especially if appetite is significantly reduced.
“There are definitely concerns for sarcopenia, so we have our patients taking these drugs try to eat healthy lean proteins – 100 g/d — and exercise,” Isaacs said. Exercise, including resistance training, not only improves muscle mass but also potentiates the effects of the GLP-1 RAs in patients with obesity and type 2 diabetes.
Adequate hydration is essential for patients taking GLP-1 RAs. “One of the commonly described side effects is fatigue, but there’s no biological reason why these medications should cause fatigue. My opinion is that these patients are dehydrated, and that may be causing the fatigue,” Isaacs said.
Some patients taking GLP-1 RAs lose interest in food. Isaacs regarded this as an “adverse reaction to the medication, which necessitates either stopping it altogether, changing the dose, or adjusting the diet.” There are “many different solutions, and one size doesn’t fit all,” he said.
Dietary and Behavioral Counseling
The drugs don’t necessarily motivate a person to eat healthier food, only to eat less food, Isaacs noted.
“The person might be eating low-volume but high-calorie food, such as bag of chips or a cookie instead of an apple,” Isaacs said. Patients who are losing weight “may not realize that weight loss isn’t the only important outcome. Because they’re losing weight, they think it’s okay to eat junk food.”
Patients need education and guidance about how to eat while on these medications. Most patients find counseling about meal planning helpful, he said.
Isaacs gives nutritional guidance to his patients when he prescribes a weight loss medication. “But most physicians don’t have time to offer that type of specific counseling on an ongoing basis,” he said. Isaacs refers patients requiring more detailed and long-term guidance to a dietitian.
Patients with monotonous diets of poor quality are at increased risk for nutrition deficiencies, and counseling by a registered dietitian could help improve their dietary quality.
Registered dietitians can develop a multifaceted approach not only focusing on medication management but also on customizing the patient’s diet, assisting with lifestyle adjustments, and addressing the mental health issues surrounding obesity and its management.
People seeking obesity treatment often have psychiatric conditions, psychological distress, or disordered eating patterns, and questions and concerns have emerged about how GLP-1 RA use might affect existing mental health problems. For example, if the medication suppresses the feeling of gratification a person once got from eating high-energy dense foods, that individual may “seek rewards or pleasure elsewhere, and possibly from unhealthy sources.”
Psychological issues also may emerge as a result of weight loss, so it’s helpful to take a multidisciplinary approach that includes mental health practitioners to support patients who are being treated with GLP-1 RAs. Patients taking these agents should be monitored for the emergence or worsening of psychiatric conditions, such as depression and suicidal ideation.
Achieving significant weight loss may lead to “unexpected changes” in the dynamics of patients’ relationship with others, “which can be distressing.” Clinicians should be “sensitive to patients’ social and emotional needs” and provide support or refer patients for help with coping strategies.
GLP-1 RAs have enormous potential to improve health outcomes in patients with obesity. Careful patient selection, close monitoring, and support for patients with nutrition and other lifestyle issues can increase the chances that these agents will fulfill their potential.
Isaacs declared no relevant financial relationships.
A version of this article appeared on Medscape.com.
Can GLP-1s Reduce Alzheimer’s Disease Risk?
Tina is a lovely 67-year-old woman who was recently found to be an APOE gene carrier (a gene associated with increased risk of developing Alzheimer’s disease as well as an earlier age of disease onset), with diffused amyloid protein deposition her brain.
Her neuropsychiatric testing was consistent with mild cognitive impairment. Although Tina is not a doctor herself, her entire family consists of doctors, and she came to me under their advisement to consider semaglutide (Ozempic) for early Alzheimer’s disease prevention.
This would usually be simple, but in Tina’s case, there was a complicating factor: At 5’ and 90 pounds, she was already considerably underweight and was at risk of becoming severely undernourished.
To understand the potential role for glucagon-like peptide-1 (GLP-1) receptor agonists such as Ozempic in prevention, a quick primer on Alzheimer’s Disease is necessary.
The exact cause of Alzheimer’s disease remains elusive, but it is probably due to a combination of factors, including:
- Buildup of abnormal amyloid and tau proteins around brain cells
- Brain shrinkage, with subsequent damage to blood vessels and mitochondria, and inflammation
- Genetic predisposition
- Lifestyle factors, including obesity, high blood pressure, high cholesterol, and diabetes.
Once in the brain, they can reduce inflammation and improve functioning of the neurons. In early rodent trials, GLP-1 receptor agonists led to reduced amyloid and tau aggregation, downregulation of inflammation, and improved memory.
In 2021, multiple studies showed that liraglutide, an early GLP-1 receptor agonist, improved cognitive function and MRI volume in patients with Alzheimer’s disease.
A study recently published in Alzheimer’s & Dementia analyzed data from 1 million people with type 2 diabetes and no prior Alzheimer’s disease diagnosis. The authors compared Alzheimer’s disease occurrence in patients taking various diabetes medications, including insulin, metformin, and GLP-1 receptor agonists. The study found that participants taking semaglutide had up to a 70% reduction in Alzheimer’s risk. The results were consistent across gender, age, and weight.
Given the reassuring safety profile of GLP-1 receptor agonists and lack of other effective treatment or prophylaxis for Alzheimer’s disease, I agreed to start her on dulaglutide (Trulicity). My rationale was twofold:
1. In studies, dulaglutide has the highest uptake in the brain tissue at 68%. By contrast, there is virtually zero uptake in brain tissue for semaglutide (Ozempic/Wegovy) and tirzepatide (Mounjaro/Zepbound). Because this class of drugs exert their effects in the brain tissue, I wanted to give her a GLP-1 receptor agonist with a high percent uptake.
2. Trulicity has a minimal effect on weight loss compared with the newer-generation GLP-1 receptor agonists. Even so, I connected Tina to my dietitian to ensure that she would receive a high-protein, high-calorie diet.
Tina has now been taking Trulicity for 6 months. Although it is certainly too early to draw firm conclusions about the efficacy of her treatment, she is not experiencing any weight loss and is cognitively stable, according to her neurologist.
The EVOKE and EVOKE+ phase 3 trials are currently underway to evaluate the efficacy of semaglutide to treat mild cognitive impairment and early Alzheimer’s in amyloid-positive patients. Results are expected in 2025, but in the meantime, I feel comforted knowing that Tina is receiving a potentially beneficial and definitively low-risk treatment.
Dr Messer, Clinical Assistant Professor, Mount Sinai School of Medicine; Associate Professor, Hofstra School of Medicine, New York, NY, has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
Tina is a lovely 67-year-old woman who was recently found to be an APOE gene carrier (a gene associated with increased risk of developing Alzheimer’s disease as well as an earlier age of disease onset), with diffused amyloid protein deposition her brain.
Her neuropsychiatric testing was consistent with mild cognitive impairment. Although Tina is not a doctor herself, her entire family consists of doctors, and she came to me under their advisement to consider semaglutide (Ozempic) for early Alzheimer’s disease prevention.
This would usually be simple, but in Tina’s case, there was a complicating factor: At 5’ and 90 pounds, she was already considerably underweight and was at risk of becoming severely undernourished.
To understand the potential role for glucagon-like peptide-1 (GLP-1) receptor agonists such as Ozempic in prevention, a quick primer on Alzheimer’s Disease is necessary.
The exact cause of Alzheimer’s disease remains elusive, but it is probably due to a combination of factors, including:
- Buildup of abnormal amyloid and tau proteins around brain cells
- Brain shrinkage, with subsequent damage to blood vessels and mitochondria, and inflammation
- Genetic predisposition
- Lifestyle factors, including obesity, high blood pressure, high cholesterol, and diabetes.
Once in the brain, they can reduce inflammation and improve functioning of the neurons. In early rodent trials, GLP-1 receptor agonists led to reduced amyloid and tau aggregation, downregulation of inflammation, and improved memory.
In 2021, multiple studies showed that liraglutide, an early GLP-1 receptor agonist, improved cognitive function and MRI volume in patients with Alzheimer’s disease.
A study recently published in Alzheimer’s & Dementia analyzed data from 1 million people with type 2 diabetes and no prior Alzheimer’s disease diagnosis. The authors compared Alzheimer’s disease occurrence in patients taking various diabetes medications, including insulin, metformin, and GLP-1 receptor agonists. The study found that participants taking semaglutide had up to a 70% reduction in Alzheimer’s risk. The results were consistent across gender, age, and weight.
Given the reassuring safety profile of GLP-1 receptor agonists and lack of other effective treatment or prophylaxis for Alzheimer’s disease, I agreed to start her on dulaglutide (Trulicity). My rationale was twofold:
1. In studies, dulaglutide has the highest uptake in the brain tissue at 68%. By contrast, there is virtually zero uptake in brain tissue for semaglutide (Ozempic/Wegovy) and tirzepatide (Mounjaro/Zepbound). Because this class of drugs exert their effects in the brain tissue, I wanted to give her a GLP-1 receptor agonist with a high percent uptake.
2. Trulicity has a minimal effect on weight loss compared with the newer-generation GLP-1 receptor agonists. Even so, I connected Tina to my dietitian to ensure that she would receive a high-protein, high-calorie diet.
Tina has now been taking Trulicity for 6 months. Although it is certainly too early to draw firm conclusions about the efficacy of her treatment, she is not experiencing any weight loss and is cognitively stable, according to her neurologist.
The EVOKE and EVOKE+ phase 3 trials are currently underway to evaluate the efficacy of semaglutide to treat mild cognitive impairment and early Alzheimer’s in amyloid-positive patients. Results are expected in 2025, but in the meantime, I feel comforted knowing that Tina is receiving a potentially beneficial and definitively low-risk treatment.
Dr Messer, Clinical Assistant Professor, Mount Sinai School of Medicine; Associate Professor, Hofstra School of Medicine, New York, NY, has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
Tina is a lovely 67-year-old woman who was recently found to be an APOE gene carrier (a gene associated with increased risk of developing Alzheimer’s disease as well as an earlier age of disease onset), with diffused amyloid protein deposition her brain.
Her neuropsychiatric testing was consistent with mild cognitive impairment. Although Tina is not a doctor herself, her entire family consists of doctors, and she came to me under their advisement to consider semaglutide (Ozempic) for early Alzheimer’s disease prevention.
This would usually be simple, but in Tina’s case, there was a complicating factor: At 5’ and 90 pounds, she was already considerably underweight and was at risk of becoming severely undernourished.
To understand the potential role for glucagon-like peptide-1 (GLP-1) receptor agonists such as Ozempic in prevention, a quick primer on Alzheimer’s Disease is necessary.
The exact cause of Alzheimer’s disease remains elusive, but it is probably due to a combination of factors, including:
- Buildup of abnormal amyloid and tau proteins around brain cells
- Brain shrinkage, with subsequent damage to blood vessels and mitochondria, and inflammation
- Genetic predisposition
- Lifestyle factors, including obesity, high blood pressure, high cholesterol, and diabetes.
Once in the brain, they can reduce inflammation and improve functioning of the neurons. In early rodent trials, GLP-1 receptor agonists led to reduced amyloid and tau aggregation, downregulation of inflammation, and improved memory.
In 2021, multiple studies showed that liraglutide, an early GLP-1 receptor agonist, improved cognitive function and MRI volume in patients with Alzheimer’s disease.
A study recently published in Alzheimer’s & Dementia analyzed data from 1 million people with type 2 diabetes and no prior Alzheimer’s disease diagnosis. The authors compared Alzheimer’s disease occurrence in patients taking various diabetes medications, including insulin, metformin, and GLP-1 receptor agonists. The study found that participants taking semaglutide had up to a 70% reduction in Alzheimer’s risk. The results were consistent across gender, age, and weight.
Given the reassuring safety profile of GLP-1 receptor agonists and lack of other effective treatment or prophylaxis for Alzheimer’s disease, I agreed to start her on dulaglutide (Trulicity). My rationale was twofold:
1. In studies, dulaglutide has the highest uptake in the brain tissue at 68%. By contrast, there is virtually zero uptake in brain tissue for semaglutide (Ozempic/Wegovy) and tirzepatide (Mounjaro/Zepbound). Because this class of drugs exert their effects in the brain tissue, I wanted to give her a GLP-1 receptor agonist with a high percent uptake.
2. Trulicity has a minimal effect on weight loss compared with the newer-generation GLP-1 receptor agonists. Even so, I connected Tina to my dietitian to ensure that she would receive a high-protein, high-calorie diet.
Tina has now been taking Trulicity for 6 months. Although it is certainly too early to draw firm conclusions about the efficacy of her treatment, she is not experiencing any weight loss and is cognitively stable, according to her neurologist.
The EVOKE and EVOKE+ phase 3 trials are currently underway to evaluate the efficacy of semaglutide to treat mild cognitive impairment and early Alzheimer’s in amyloid-positive patients. Results are expected in 2025, but in the meantime, I feel comforted knowing that Tina is receiving a potentially beneficial and definitively low-risk treatment.
Dr Messer, Clinical Assistant Professor, Mount Sinai School of Medicine; Associate Professor, Hofstra School of Medicine, New York, NY, has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
How Does End of Life Impact Diabetes Care?
TOPLINE:
Among older adults with type 2 diabetes (T2D), the use of antidiabetes medications declined in the last year before death, with notable shifts from metformin and sulfonylureas toward insulin therapy.
METHODOLOGY:
Current recommendations emphasize a more liberal approach to glycemic control in people with a high burden of comorbidities and shorter life expectancy, but little is known about the changes and discontinuation patterns of diabetes medications among older adults near the end of life.
.
All medication classes available during the study period were considered, including short-acting and long-acting insulins, metformin, sulfonylureas, dipeptidyl peptidase-4 inhibitors, glucagon-like peptide 1 (GLP-1) receptor agonists, sodium-glucose cotransporter-2 (SGLT2) inhibitors, and other medications.
Analysis included temporal trends in prescribing antidiabetes medications, stratified by frailty using a validated claims-based frailty index, with scores ≥ 0.30 indicating higher frailty.
Antidiabetes medication fills were assessed within 1 year before death, examining changes across three time periods: 12 to 8 months, 8 to 4 months, and 4 to 0 months before death.
TAKEAWAY:
The proportion of older patients receiving antidiabetes medications increased slightly from 71.4% in 2015 to 72.9% in 2019, with metformin showing the largest increase from 40.7% to 46.5% (standardized mean difference [SMD], −0.12) and sulfonylureas showing the largest decrease from 37.0% to 31.8% (SMD, 0.11).
The use of newer diabetes medications with cardiovascular benefits, such as GLP-1 receptor agonists and SGLT2 inhibitors, remained less common but showed increasing trends over time.
The use of any antidiabetes medication decreased from 66.1% in the 9 to 12 months before death to 60.8% in the last 4 months of life (P < .01), primarily due to the reduced use of metformin and sulfonylureas.
The use of both short-acting and long-acting insulin agents increased toward the end of life (from 28.0% to 32.9% and from 41.2% to 43.9%, respectively; both P < .001) , particularly among frailer individuals.
IN PRACTICE:
“[The study] findings underscore important implications for diabetes management in patients nearing the end of life. With ~70% of patients with T2D using at least one antidiabetes medication, there is a need to consider further de-escalation or deprescribing in this vulnerable population,” the authors wrote.
SOURCE:
The study was led by Alexander Kutz, MD, MPH, MSc, Division of Pharmacoepidemiology and Pharmacoeconomics, Department of Medicine, Brigham and Women’s Hospital and Harvard Medical School, Boston, and was published online in Diabetes Care.
LIMITATIONS:
The study lacked details on the reasons for changes in medication patterns, making it unclear whether these changes were due to clinical guidelines or to reduce adverse events. Moreover, the study could not capture transitions or substitutions between medications, information on the dosage data, and causes of death.
DISCLOSURES:
This study was supported by the Division of Pharmacoepidemiology and Pharmacoeconomics, Department of Medicine, Brigham and Women’s Hospital, Harvard Medical School. Some authors reported receiving personal fees or research grants from the National Institutes of Health and other institutions and a few pharmaceutical companies. One author reported acting as a principal investigator and receiving a research grant from Boehringer-Ingelheim, unrelated to the work.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
TOPLINE:
Among older adults with type 2 diabetes (T2D), the use of antidiabetes medications declined in the last year before death, with notable shifts from metformin and sulfonylureas toward insulin therapy.
METHODOLOGY:
Current recommendations emphasize a more liberal approach to glycemic control in people with a high burden of comorbidities and shorter life expectancy, but little is known about the changes and discontinuation patterns of diabetes medications among older adults near the end of life.
.
All medication classes available during the study period were considered, including short-acting and long-acting insulins, metformin, sulfonylureas, dipeptidyl peptidase-4 inhibitors, glucagon-like peptide 1 (GLP-1) receptor agonists, sodium-glucose cotransporter-2 (SGLT2) inhibitors, and other medications.
Analysis included temporal trends in prescribing antidiabetes medications, stratified by frailty using a validated claims-based frailty index, with scores ≥ 0.30 indicating higher frailty.
Antidiabetes medication fills were assessed within 1 year before death, examining changes across three time periods: 12 to 8 months, 8 to 4 months, and 4 to 0 months before death.
TAKEAWAY:
The proportion of older patients receiving antidiabetes medications increased slightly from 71.4% in 2015 to 72.9% in 2019, with metformin showing the largest increase from 40.7% to 46.5% (standardized mean difference [SMD], −0.12) and sulfonylureas showing the largest decrease from 37.0% to 31.8% (SMD, 0.11).
The use of newer diabetes medications with cardiovascular benefits, such as GLP-1 receptor agonists and SGLT2 inhibitors, remained less common but showed increasing trends over time.
The use of any antidiabetes medication decreased from 66.1% in the 9 to 12 months before death to 60.8% in the last 4 months of life (P < .01), primarily due to the reduced use of metformin and sulfonylureas.
The use of both short-acting and long-acting insulin agents increased toward the end of life (from 28.0% to 32.9% and from 41.2% to 43.9%, respectively; both P < .001) , particularly among frailer individuals.
IN PRACTICE:
“[The study] findings underscore important implications for diabetes management in patients nearing the end of life. With ~70% of patients with T2D using at least one antidiabetes medication, there is a need to consider further de-escalation or deprescribing in this vulnerable population,” the authors wrote.
SOURCE:
The study was led by Alexander Kutz, MD, MPH, MSc, Division of Pharmacoepidemiology and Pharmacoeconomics, Department of Medicine, Brigham and Women’s Hospital and Harvard Medical School, Boston, and was published online in Diabetes Care.
LIMITATIONS:
The study lacked details on the reasons for changes in medication patterns, making it unclear whether these changes were due to clinical guidelines or to reduce adverse events. Moreover, the study could not capture transitions or substitutions between medications, information on the dosage data, and causes of death.
DISCLOSURES:
This study was supported by the Division of Pharmacoepidemiology and Pharmacoeconomics, Department of Medicine, Brigham and Women’s Hospital, Harvard Medical School. Some authors reported receiving personal fees or research grants from the National Institutes of Health and other institutions and a few pharmaceutical companies. One author reported acting as a principal investigator and receiving a research grant from Boehringer-Ingelheim, unrelated to the work.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
TOPLINE:
Among older adults with type 2 diabetes (T2D), the use of antidiabetes medications declined in the last year before death, with notable shifts from metformin and sulfonylureas toward insulin therapy.
METHODOLOGY:
Current recommendations emphasize a more liberal approach to glycemic control in people with a high burden of comorbidities and shorter life expectancy, but little is known about the changes and discontinuation patterns of diabetes medications among older adults near the end of life.
.
All medication classes available during the study period were considered, including short-acting and long-acting insulins, metformin, sulfonylureas, dipeptidyl peptidase-4 inhibitors, glucagon-like peptide 1 (GLP-1) receptor agonists, sodium-glucose cotransporter-2 (SGLT2) inhibitors, and other medications.
Analysis included temporal trends in prescribing antidiabetes medications, stratified by frailty using a validated claims-based frailty index, with scores ≥ 0.30 indicating higher frailty.
Antidiabetes medication fills were assessed within 1 year before death, examining changes across three time periods: 12 to 8 months, 8 to 4 months, and 4 to 0 months before death.
TAKEAWAY:
The proportion of older patients receiving antidiabetes medications increased slightly from 71.4% in 2015 to 72.9% in 2019, with metformin showing the largest increase from 40.7% to 46.5% (standardized mean difference [SMD], −0.12) and sulfonylureas showing the largest decrease from 37.0% to 31.8% (SMD, 0.11).
The use of newer diabetes medications with cardiovascular benefits, such as GLP-1 receptor agonists and SGLT2 inhibitors, remained less common but showed increasing trends over time.
The use of any antidiabetes medication decreased from 66.1% in the 9 to 12 months before death to 60.8% in the last 4 months of life (P < .01), primarily due to the reduced use of metformin and sulfonylureas.
The use of both short-acting and long-acting insulin agents increased toward the end of life (from 28.0% to 32.9% and from 41.2% to 43.9%, respectively; both P < .001) , particularly among frailer individuals.
IN PRACTICE:
“[The study] findings underscore important implications for diabetes management in patients nearing the end of life. With ~70% of patients with T2D using at least one antidiabetes medication, there is a need to consider further de-escalation or deprescribing in this vulnerable population,” the authors wrote.
SOURCE:
The study was led by Alexander Kutz, MD, MPH, MSc, Division of Pharmacoepidemiology and Pharmacoeconomics, Department of Medicine, Brigham and Women’s Hospital and Harvard Medical School, Boston, and was published online in Diabetes Care.
LIMITATIONS:
The study lacked details on the reasons for changes in medication patterns, making it unclear whether these changes were due to clinical guidelines or to reduce adverse events. Moreover, the study could not capture transitions or substitutions between medications, information on the dosage data, and causes of death.
DISCLOSURES:
This study was supported by the Division of Pharmacoepidemiology and Pharmacoeconomics, Department of Medicine, Brigham and Women’s Hospital, Harvard Medical School. Some authors reported receiving personal fees or research grants from the National Institutes of Health and other institutions and a few pharmaceutical companies. One author reported acting as a principal investigator and receiving a research grant from Boehringer-Ingelheim, unrelated to the work.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
Choline Alfoscerate Has Modest Benefits on Cognition in Type 2 Diabetes
TOPLINE:
METHODOLOGY:
- Prior studies have demonstrated the efficacy of choline alfoscerate, a phospholipid metabolite naturally found in the brain, in improving cognitive function in patients with neurodegenerative conditions, but its use in patients with T2D remains unexplored.
- Researchers at a hospital in Korea enrolled patients aged over 60 years with T2D and mild cognitive impairment (assessed by Mini-Mental State Examination [MMSE] scores of 25-28), who were randomly assigned to receive either 1200 mg/d choline alfoscerate or placebo for 12 months.
- The primary efficacy endpoint was the change in the total MMSE score from baseline to month 6; secondary efficacy endpoints included changes in cognitive performance and quality of life, measured by the 36-Item Short Form Health Survey, at 6 and 12 months.
TAKEAWAY:
- Thirty-six patients (average age, 71.8 years; 25% men) with an average diabetes duration of 12.1 years were randomized to receive choline alfoscerate (n = 18) or placebo (n = 18).
- At 6 months, there was modest but nonsignificant improvement in MMSE score with choline alfoscerate vs placebo (P = .059).
- After 12 months, the choline alfoscerate group showed an increase in the MMSE score from 26.2 to 27.1, whereas the placebo group showed a slight decline from 26.6 to 25.8, which represented a significant improvement for the treatment arm (P < .001).
- Physical health scores were significantly superior in the choline alfoscerate group vs the placebo group at 6 months (P = .014), with similar observations at 12 months (P = .039).
- No serious adverse events were reported in either group.
IN PRACTICE:
“Choline alfoscerate could be considered an anticipated therapeutic option to preserve cognitive function and subsequently physical health in elderly patients with diabetes and mild cognitive impairment,” the authors wrote.
SOURCE:
The study was led by Minji Sohn, PhD, Department of Internal Medicine, Seoul National University Bundang Hospital, Seoul National University College of Medicine, Seongnam, Republic of Korea, and published online in Diabetes, Obesity and Metabolism.
LIMITATIONS:
The study population primarily comprised non–insulin-dependent patients with controlled glycemia and minimal comorbidities, which may have limited the applicability of the results to a broader population. The small sample size may have contributed to the lack of statistical significance in some outcomes. Moreover, the 12-month study duration may have not been sufficient to investigate the long-term effects of choline alfoscerate.
DISCLOSURES:
This study was funded by Daewoong Pharmaceutical through subcontracting with Seoul National University Bundang Hospital. The authors declared no competing interests.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
TOPLINE:
METHODOLOGY:
- Prior studies have demonstrated the efficacy of choline alfoscerate, a phospholipid metabolite naturally found in the brain, in improving cognitive function in patients with neurodegenerative conditions, but its use in patients with T2D remains unexplored.
- Researchers at a hospital in Korea enrolled patients aged over 60 years with T2D and mild cognitive impairment (assessed by Mini-Mental State Examination [MMSE] scores of 25-28), who were randomly assigned to receive either 1200 mg/d choline alfoscerate or placebo for 12 months.
- The primary efficacy endpoint was the change in the total MMSE score from baseline to month 6; secondary efficacy endpoints included changes in cognitive performance and quality of life, measured by the 36-Item Short Form Health Survey, at 6 and 12 months.
TAKEAWAY:
- Thirty-six patients (average age, 71.8 years; 25% men) with an average diabetes duration of 12.1 years were randomized to receive choline alfoscerate (n = 18) or placebo (n = 18).
- At 6 months, there was modest but nonsignificant improvement in MMSE score with choline alfoscerate vs placebo (P = .059).
- After 12 months, the choline alfoscerate group showed an increase in the MMSE score from 26.2 to 27.1, whereas the placebo group showed a slight decline from 26.6 to 25.8, which represented a significant improvement for the treatment arm (P < .001).
- Physical health scores were significantly superior in the choline alfoscerate group vs the placebo group at 6 months (P = .014), with similar observations at 12 months (P = .039).
- No serious adverse events were reported in either group.
IN PRACTICE:
“Choline alfoscerate could be considered an anticipated therapeutic option to preserve cognitive function and subsequently physical health in elderly patients with diabetes and mild cognitive impairment,” the authors wrote.
SOURCE:
The study was led by Minji Sohn, PhD, Department of Internal Medicine, Seoul National University Bundang Hospital, Seoul National University College of Medicine, Seongnam, Republic of Korea, and published online in Diabetes, Obesity and Metabolism.
LIMITATIONS:
The study population primarily comprised non–insulin-dependent patients with controlled glycemia and minimal comorbidities, which may have limited the applicability of the results to a broader population. The small sample size may have contributed to the lack of statistical significance in some outcomes. Moreover, the 12-month study duration may have not been sufficient to investigate the long-term effects of choline alfoscerate.
DISCLOSURES:
This study was funded by Daewoong Pharmaceutical through subcontracting with Seoul National University Bundang Hospital. The authors declared no competing interests.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
TOPLINE:
METHODOLOGY:
- Prior studies have demonstrated the efficacy of choline alfoscerate, a phospholipid metabolite naturally found in the brain, in improving cognitive function in patients with neurodegenerative conditions, but its use in patients with T2D remains unexplored.
- Researchers at a hospital in Korea enrolled patients aged over 60 years with T2D and mild cognitive impairment (assessed by Mini-Mental State Examination [MMSE] scores of 25-28), who were randomly assigned to receive either 1200 mg/d choline alfoscerate or placebo for 12 months.
- The primary efficacy endpoint was the change in the total MMSE score from baseline to month 6; secondary efficacy endpoints included changes in cognitive performance and quality of life, measured by the 36-Item Short Form Health Survey, at 6 and 12 months.
TAKEAWAY:
- Thirty-six patients (average age, 71.8 years; 25% men) with an average diabetes duration of 12.1 years were randomized to receive choline alfoscerate (n = 18) or placebo (n = 18).
- At 6 months, there was modest but nonsignificant improvement in MMSE score with choline alfoscerate vs placebo (P = .059).
- After 12 months, the choline alfoscerate group showed an increase in the MMSE score from 26.2 to 27.1, whereas the placebo group showed a slight decline from 26.6 to 25.8, which represented a significant improvement for the treatment arm (P < .001).
- Physical health scores were significantly superior in the choline alfoscerate group vs the placebo group at 6 months (P = .014), with similar observations at 12 months (P = .039).
- No serious adverse events were reported in either group.
IN PRACTICE:
“Choline alfoscerate could be considered an anticipated therapeutic option to preserve cognitive function and subsequently physical health in elderly patients with diabetes and mild cognitive impairment,” the authors wrote.
SOURCE:
The study was led by Minji Sohn, PhD, Department of Internal Medicine, Seoul National University Bundang Hospital, Seoul National University College of Medicine, Seongnam, Republic of Korea, and published online in Diabetes, Obesity and Metabolism.
LIMITATIONS:
The study population primarily comprised non–insulin-dependent patients with controlled glycemia and minimal comorbidities, which may have limited the applicability of the results to a broader population. The small sample size may have contributed to the lack of statistical significance in some outcomes. Moreover, the 12-month study duration may have not been sufficient to investigate the long-term effects of choline alfoscerate.
DISCLOSURES:
This study was funded by Daewoong Pharmaceutical through subcontracting with Seoul National University Bundang Hospital. The authors declared no competing interests.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.