User login
Diabetes drug’s new weight-loss indication fuels cost-benefit debate
The long list of side effects that follow ads for newer expensive drugs to treat type 2 diabetes sometimes include an unusual warning: They might cause weight loss. That side effect is one that many people – especially those with type 2 diabetes, which is associated with obesity – may desperately want.
So it’s no surprise that some of the same drugs are being reformulated and renamed by manufacturers as a new obesity treatment. No longer limited to the crowded field of treatments for type 2 diabetes, which affects about 10% of Americans, they join the far smaller number of drugs for obesity, which affects 42% of Americans and is ready to be mined for profit.
One that recently hit the market – winning Food and Drug Administration approval in June – is Novo Nordisk’s Wegovy (semaglutide), a higher-dose version of the company’s injectable diabetes drug, Ozempic.
Ozempic’s peppy ads suggest that people who use it might lose weight, but also include a disclaimer: that it “is not a weight-loss drug.” Now – with a new name – it is. And clinical trials showed using it leads to significant weight loss for many patients.
“People who go on this medication lose more weight than with any drug we’ve seen, ever,” said Fatima Cody Stanford, MD, MPH, an obesity medicine specialist at Massachusetts General Hospital and Harvard Medical School, both in Boston, who was not involved with any of the clinical trials.
But that leaves employers and insurers in the uncomfortable position of deciding if it’s worth it.
Wegovy’s monthly wholesale price tag – set at $1,349 – is about 58% more than Ozempic’s, although, the company pointed out, the drug’s injector pens contain more than twice as much of the active ingredient. Studies so far show that patients may need to take it indefinitely to maintain weight loss, translating to a tab that could top $323,000 over 20 years at the current price. Weight-loss treatments are not universally covered by insurance policies.
The arrival of this new class of weight-loss drugs – one from Lilly may soon follow – has created a thicket of issues for those who will pay for them. The decision is complicated by many unknowables concerning their long-term use and whether competition might eventually lower the price.
“The metric we try to use is value,” said James Gelfand, senior vice president for health policy at the ERISA Industry Committee, which represents large, self-insured employers. “If we pay for this drug, how much is this going to cost and how much value will it provide to the beneficiaries?”
Weight-loss treatments have had a lackluster past in this regard, with only modest results. Many employers and insurers likely remember Fen-Phen, a combination of fenfluramine and dexfenfluramine that was pulled from the market in the late 1990s for causing heart valve problems.
New drugs like Wegovy, more effective but also pricier than previous weight-loss treatments, will add more fuel to that debate.
Past treatments were shown to prompt weight loss in the range of 5%-10% of body weight. But many had relatively serious or unpleasant side effects.
Wegovy, however, helped patients lose an average of 15% of their body weight over 68 weeks in the main clinical trial that led to its approval. A comparison group that got a placebo injection lost an average of 2.5% over the same period. On the high end, nearly a third of patients in the treatment group lost 20% or more. Both groups had counseling on diet and exercise.
Side effects, generally considered mild, included nausea, diarrhea, vomiting, and constipation. A few patients developed pancreatitis, a serious inflammation of the pancreas. Like the diabetes medication, the drug carries a warning about a potential risk of a type of thyroid cancer.
Weight loss in those taking Wegovy puts it close to the 20%-25% losses seen with bariatric surgery, said Stanford, and well above the 3%-4% seen with diet and other lifestyle changes alone.
Participants also saw reductions in their waistlines and improvements in their blood pressure and blood sugar levels, which may mean they won’t develop diabetes, said Sean Wharton, MD, an internal medicine specialist and adjunct professor at York University in Toronto who was among the coauthors of the report outlining the results of the first clinical trial on Wegovy.
Since weight loss is known to reduce the risk of heart attack, high blood pressure and diabetes, might the new drug type be worth it?
Covering such treatment would be a sea change for Medicare, which specifically bars coverage for obesity medications or drugs for “anorexia, weight loss, or weight gain,” although it does pay for bariatric surgery. Pharmaceutical companies, patient advocates, and some medical professionals are backing proposed federal legislation to allow coverage. But the legislation, the Treat and Reduce Obesity Act, has not made progress despite being reintroduced every year since 2012, and sponsors are now asking federal officials instead to rewrite existing rules.
Private insurers will have to consider a cost-benefit analysis of adding Wegovy to their list of covered treatments, either broadly or with limits. Obesity was first recognized as a disease by the American Medical Association, easing the path for insurance coverage, in 2013.
“Employers are going to have a bit of a challenge” deciding whether to add the benefit to insurance offerings, said Steve Pearson, founder and president of the Institute for Clinical and Economic Review, which provides cost-benefit analyses of medical treatments but has not yet looked at Wegovy.
The trade-offs are embodied in patients like Phylander Pannell, a 49-year-old Largo, Md., woman who said she lost 65 pounds in a clinical trial of Wegovy. That study gave the drug to all participants for the first 20 weeks, then randomly assigned patients to get either the drug or a placebo for the next 48 weeks to determine what happens when the medication is stopped. Only after the trial ended did she find out she was in the treatment group the entire time.
Her weight fell slowly at first, then ramped up, eventually bringing her 190-pound frame down to about 125. Pains in her joints eased; she felt better all around.
“I definitely feel the drug was it for me,” said Ms. Pannell, who also followed the trial’s guidance on diet and exercise.
The study found that both groups lost weight in the initial 20 weeks, but those who continued to get the drug lost an additional average of 7.9% of their body weight. Those who got a placebo gained back nearly 7%.
After the trial ended, and the COVID-19 pandemic hit, Ms. Pannell regained some weight and is now at 155. She is eager to get back on the medication and hopes her job-based insurance will cover it.
Many employers do cover obesity drugs. For example, about 40% of private employer plans include Novo Nordisk’s once-daily injection called Saxenda on their health plans, said Michael Bachner, Novo Nordisk’s director of media relations.
He said the $1,349-a-month wholesale acquisition price of Wegovy was determined by making it equivalent to that of Saxenda, which is less effective.
Still, that is more than the $851 monthly wholesale price of Ozempic. But, he pointed out, the recommended dosage of Wegovy is more than twice that of Ozempic. Four milligrams come in the Ozempic injector pens for the month, while Wegovy has 9.6.
“There’s more drug in the pen,” Mr. Bachner said. “That drives the price up.”
He added: “This is not a 20-year-old drug that we now have a new indication for and are pricing it higher. It’s a whole different clinical program,” which required new trials.
Now scientists, employers, physicians, and patients will have to decide whether the new drugs are worth it.
Earlier estimates – some commissioned by Novo Nordisk – of the potential cost of adding an obesity drug benefit to Medicare showed an overall reduction in spending when better health from the resulting weight loss was factored in.
Still, those earlier estimates considered much less expensive drugs, including a range of generic and branded drugs costing as little as $7 a month to more than $300, a small fraction of Wegovy’s cost.
KHN (Kaiser Health News) is a national newsroom that produces in-depth journalism about health issues. Together with Policy Analysis and Polling, KHN is one of the three major operating programs at KFF (Kaiser Family Foundation). KFF is an endowed nonprofit organization providing information on health issues to the nation.
The long list of side effects that follow ads for newer expensive drugs to treat type 2 diabetes sometimes include an unusual warning: They might cause weight loss. That side effect is one that many people – especially those with type 2 diabetes, which is associated with obesity – may desperately want.
So it’s no surprise that some of the same drugs are being reformulated and renamed by manufacturers as a new obesity treatment. No longer limited to the crowded field of treatments for type 2 diabetes, which affects about 10% of Americans, they join the far smaller number of drugs for obesity, which affects 42% of Americans and is ready to be mined for profit.
One that recently hit the market – winning Food and Drug Administration approval in June – is Novo Nordisk’s Wegovy (semaglutide), a higher-dose version of the company’s injectable diabetes drug, Ozempic.
Ozempic’s peppy ads suggest that people who use it might lose weight, but also include a disclaimer: that it “is not a weight-loss drug.” Now – with a new name – it is. And clinical trials showed using it leads to significant weight loss for many patients.
“People who go on this medication lose more weight than with any drug we’ve seen, ever,” said Fatima Cody Stanford, MD, MPH, an obesity medicine specialist at Massachusetts General Hospital and Harvard Medical School, both in Boston, who was not involved with any of the clinical trials.
But that leaves employers and insurers in the uncomfortable position of deciding if it’s worth it.
Wegovy’s monthly wholesale price tag – set at $1,349 – is about 58% more than Ozempic’s, although, the company pointed out, the drug’s injector pens contain more than twice as much of the active ingredient. Studies so far show that patients may need to take it indefinitely to maintain weight loss, translating to a tab that could top $323,000 over 20 years at the current price. Weight-loss treatments are not universally covered by insurance policies.
The arrival of this new class of weight-loss drugs – one from Lilly may soon follow – has created a thicket of issues for those who will pay for them. The decision is complicated by many unknowables concerning their long-term use and whether competition might eventually lower the price.
“The metric we try to use is value,” said James Gelfand, senior vice president for health policy at the ERISA Industry Committee, which represents large, self-insured employers. “If we pay for this drug, how much is this going to cost and how much value will it provide to the beneficiaries?”
Weight-loss treatments have had a lackluster past in this regard, with only modest results. Many employers and insurers likely remember Fen-Phen, a combination of fenfluramine and dexfenfluramine that was pulled from the market in the late 1990s for causing heart valve problems.
New drugs like Wegovy, more effective but also pricier than previous weight-loss treatments, will add more fuel to that debate.
Past treatments were shown to prompt weight loss in the range of 5%-10% of body weight. But many had relatively serious or unpleasant side effects.
Wegovy, however, helped patients lose an average of 15% of their body weight over 68 weeks in the main clinical trial that led to its approval. A comparison group that got a placebo injection lost an average of 2.5% over the same period. On the high end, nearly a third of patients in the treatment group lost 20% or more. Both groups had counseling on diet and exercise.
Side effects, generally considered mild, included nausea, diarrhea, vomiting, and constipation. A few patients developed pancreatitis, a serious inflammation of the pancreas. Like the diabetes medication, the drug carries a warning about a potential risk of a type of thyroid cancer.
Weight loss in those taking Wegovy puts it close to the 20%-25% losses seen with bariatric surgery, said Stanford, and well above the 3%-4% seen with diet and other lifestyle changes alone.
Participants also saw reductions in their waistlines and improvements in their blood pressure and blood sugar levels, which may mean they won’t develop diabetes, said Sean Wharton, MD, an internal medicine specialist and adjunct professor at York University in Toronto who was among the coauthors of the report outlining the results of the first clinical trial on Wegovy.
Since weight loss is known to reduce the risk of heart attack, high blood pressure and diabetes, might the new drug type be worth it?
Covering such treatment would be a sea change for Medicare, which specifically bars coverage for obesity medications or drugs for “anorexia, weight loss, or weight gain,” although it does pay for bariatric surgery. Pharmaceutical companies, patient advocates, and some medical professionals are backing proposed federal legislation to allow coverage. But the legislation, the Treat and Reduce Obesity Act, has not made progress despite being reintroduced every year since 2012, and sponsors are now asking federal officials instead to rewrite existing rules.
Private insurers will have to consider a cost-benefit analysis of adding Wegovy to their list of covered treatments, either broadly or with limits. Obesity was first recognized as a disease by the American Medical Association, easing the path for insurance coverage, in 2013.
“Employers are going to have a bit of a challenge” deciding whether to add the benefit to insurance offerings, said Steve Pearson, founder and president of the Institute for Clinical and Economic Review, which provides cost-benefit analyses of medical treatments but has not yet looked at Wegovy.
The trade-offs are embodied in patients like Phylander Pannell, a 49-year-old Largo, Md., woman who said she lost 65 pounds in a clinical trial of Wegovy. That study gave the drug to all participants for the first 20 weeks, then randomly assigned patients to get either the drug or a placebo for the next 48 weeks to determine what happens when the medication is stopped. Only after the trial ended did she find out she was in the treatment group the entire time.
Her weight fell slowly at first, then ramped up, eventually bringing her 190-pound frame down to about 125. Pains in her joints eased; she felt better all around.
“I definitely feel the drug was it for me,” said Ms. Pannell, who also followed the trial’s guidance on diet and exercise.
The study found that both groups lost weight in the initial 20 weeks, but those who continued to get the drug lost an additional average of 7.9% of their body weight. Those who got a placebo gained back nearly 7%.
After the trial ended, and the COVID-19 pandemic hit, Ms. Pannell regained some weight and is now at 155. She is eager to get back on the medication and hopes her job-based insurance will cover it.
Many employers do cover obesity drugs. For example, about 40% of private employer plans include Novo Nordisk’s once-daily injection called Saxenda on their health plans, said Michael Bachner, Novo Nordisk’s director of media relations.
He said the $1,349-a-month wholesale acquisition price of Wegovy was determined by making it equivalent to that of Saxenda, which is less effective.
Still, that is more than the $851 monthly wholesale price of Ozempic. But, he pointed out, the recommended dosage of Wegovy is more than twice that of Ozempic. Four milligrams come in the Ozempic injector pens for the month, while Wegovy has 9.6.
“There’s more drug in the pen,” Mr. Bachner said. “That drives the price up.”
He added: “This is not a 20-year-old drug that we now have a new indication for and are pricing it higher. It’s a whole different clinical program,” which required new trials.
Now scientists, employers, physicians, and patients will have to decide whether the new drugs are worth it.
Earlier estimates – some commissioned by Novo Nordisk – of the potential cost of adding an obesity drug benefit to Medicare showed an overall reduction in spending when better health from the resulting weight loss was factored in.
Still, those earlier estimates considered much less expensive drugs, including a range of generic and branded drugs costing as little as $7 a month to more than $300, a small fraction of Wegovy’s cost.
KHN (Kaiser Health News) is a national newsroom that produces in-depth journalism about health issues. Together with Policy Analysis and Polling, KHN is one of the three major operating programs at KFF (Kaiser Family Foundation). KFF is an endowed nonprofit organization providing information on health issues to the nation.
The long list of side effects that follow ads for newer expensive drugs to treat type 2 diabetes sometimes include an unusual warning: They might cause weight loss. That side effect is one that many people – especially those with type 2 diabetes, which is associated with obesity – may desperately want.
So it’s no surprise that some of the same drugs are being reformulated and renamed by manufacturers as a new obesity treatment. No longer limited to the crowded field of treatments for type 2 diabetes, which affects about 10% of Americans, they join the far smaller number of drugs for obesity, which affects 42% of Americans and is ready to be mined for profit.
One that recently hit the market – winning Food and Drug Administration approval in June – is Novo Nordisk’s Wegovy (semaglutide), a higher-dose version of the company’s injectable diabetes drug, Ozempic.
Ozempic’s peppy ads suggest that people who use it might lose weight, but also include a disclaimer: that it “is not a weight-loss drug.” Now – with a new name – it is. And clinical trials showed using it leads to significant weight loss for many patients.
“People who go on this medication lose more weight than with any drug we’ve seen, ever,” said Fatima Cody Stanford, MD, MPH, an obesity medicine specialist at Massachusetts General Hospital and Harvard Medical School, both in Boston, who was not involved with any of the clinical trials.
But that leaves employers and insurers in the uncomfortable position of deciding if it’s worth it.
Wegovy’s monthly wholesale price tag – set at $1,349 – is about 58% more than Ozempic’s, although, the company pointed out, the drug’s injector pens contain more than twice as much of the active ingredient. Studies so far show that patients may need to take it indefinitely to maintain weight loss, translating to a tab that could top $323,000 over 20 years at the current price. Weight-loss treatments are not universally covered by insurance policies.
The arrival of this new class of weight-loss drugs – one from Lilly may soon follow – has created a thicket of issues for those who will pay for them. The decision is complicated by many unknowables concerning their long-term use and whether competition might eventually lower the price.
“The metric we try to use is value,” said James Gelfand, senior vice president for health policy at the ERISA Industry Committee, which represents large, self-insured employers. “If we pay for this drug, how much is this going to cost and how much value will it provide to the beneficiaries?”
Weight-loss treatments have had a lackluster past in this regard, with only modest results. Many employers and insurers likely remember Fen-Phen, a combination of fenfluramine and dexfenfluramine that was pulled from the market in the late 1990s for causing heart valve problems.
New drugs like Wegovy, more effective but also pricier than previous weight-loss treatments, will add more fuel to that debate.
Past treatments were shown to prompt weight loss in the range of 5%-10% of body weight. But many had relatively serious or unpleasant side effects.
Wegovy, however, helped patients lose an average of 15% of their body weight over 68 weeks in the main clinical trial that led to its approval. A comparison group that got a placebo injection lost an average of 2.5% over the same period. On the high end, nearly a third of patients in the treatment group lost 20% or more. Both groups had counseling on diet and exercise.
Side effects, generally considered mild, included nausea, diarrhea, vomiting, and constipation. A few patients developed pancreatitis, a serious inflammation of the pancreas. Like the diabetes medication, the drug carries a warning about a potential risk of a type of thyroid cancer.
Weight loss in those taking Wegovy puts it close to the 20%-25% losses seen with bariatric surgery, said Stanford, and well above the 3%-4% seen with diet and other lifestyle changes alone.
Participants also saw reductions in their waistlines and improvements in their blood pressure and blood sugar levels, which may mean they won’t develop diabetes, said Sean Wharton, MD, an internal medicine specialist and adjunct professor at York University in Toronto who was among the coauthors of the report outlining the results of the first clinical trial on Wegovy.
Since weight loss is known to reduce the risk of heart attack, high blood pressure and diabetes, might the new drug type be worth it?
Covering such treatment would be a sea change for Medicare, which specifically bars coverage for obesity medications or drugs for “anorexia, weight loss, or weight gain,” although it does pay for bariatric surgery. Pharmaceutical companies, patient advocates, and some medical professionals are backing proposed federal legislation to allow coverage. But the legislation, the Treat and Reduce Obesity Act, has not made progress despite being reintroduced every year since 2012, and sponsors are now asking federal officials instead to rewrite existing rules.
Private insurers will have to consider a cost-benefit analysis of adding Wegovy to their list of covered treatments, either broadly or with limits. Obesity was first recognized as a disease by the American Medical Association, easing the path for insurance coverage, in 2013.
“Employers are going to have a bit of a challenge” deciding whether to add the benefit to insurance offerings, said Steve Pearson, founder and president of the Institute for Clinical and Economic Review, which provides cost-benefit analyses of medical treatments but has not yet looked at Wegovy.
The trade-offs are embodied in patients like Phylander Pannell, a 49-year-old Largo, Md., woman who said she lost 65 pounds in a clinical trial of Wegovy. That study gave the drug to all participants for the first 20 weeks, then randomly assigned patients to get either the drug or a placebo for the next 48 weeks to determine what happens when the medication is stopped. Only after the trial ended did she find out she was in the treatment group the entire time.
Her weight fell slowly at first, then ramped up, eventually bringing her 190-pound frame down to about 125. Pains in her joints eased; she felt better all around.
“I definitely feel the drug was it for me,” said Ms. Pannell, who also followed the trial’s guidance on diet and exercise.
The study found that both groups lost weight in the initial 20 weeks, but those who continued to get the drug lost an additional average of 7.9% of their body weight. Those who got a placebo gained back nearly 7%.
After the trial ended, and the COVID-19 pandemic hit, Ms. Pannell regained some weight and is now at 155. She is eager to get back on the medication and hopes her job-based insurance will cover it.
Many employers do cover obesity drugs. For example, about 40% of private employer plans include Novo Nordisk’s once-daily injection called Saxenda on their health plans, said Michael Bachner, Novo Nordisk’s director of media relations.
He said the $1,349-a-month wholesale acquisition price of Wegovy was determined by making it equivalent to that of Saxenda, which is less effective.
Still, that is more than the $851 monthly wholesale price of Ozempic. But, he pointed out, the recommended dosage of Wegovy is more than twice that of Ozempic. Four milligrams come in the Ozempic injector pens for the month, while Wegovy has 9.6.
“There’s more drug in the pen,” Mr. Bachner said. “That drives the price up.”
He added: “This is not a 20-year-old drug that we now have a new indication for and are pricing it higher. It’s a whole different clinical program,” which required new trials.
Now scientists, employers, physicians, and patients will have to decide whether the new drugs are worth it.
Earlier estimates – some commissioned by Novo Nordisk – of the potential cost of adding an obesity drug benefit to Medicare showed an overall reduction in spending when better health from the resulting weight loss was factored in.
Still, those earlier estimates considered much less expensive drugs, including a range of generic and branded drugs costing as little as $7 a month to more than $300, a small fraction of Wegovy’s cost.
KHN (Kaiser Health News) is a national newsroom that produces in-depth journalism about health issues. Together with Policy Analysis and Polling, KHN is one of the three major operating programs at KFF (Kaiser Family Foundation). KFF is an endowed nonprofit organization providing information on health issues to the nation.
Real-World Experience With Automated Insulin Pump Technology in Veterans With Type 1 Diabetes
Insulin pump technology has been available since the 1970s. Innovation in insulin pumps has had significant impact on the management of diabetes mellitus (DM). In recent years, automated insulin pump technology (AIP) has proven to be a safe and effective way to treat DM. It has been studied mostly in highly organized randomized controlled trials (RCTs) in younger populations with type 1 DM (T1DM).1-3
One of the challenges in DM care has always been the wide variations in daily plasma glucose concentration that often cause major swings of hyperglycemia and hypoglycemia. Extreme variations in blood glucose have also been linked to adverse outcomes, including poor micro- and macrovascular outcomes.4,5 AIP technology is a hybrid closed-loop system that attempts to solve this problem by adjusting insulin delivery in response to real-time glucose information from a continuous glucose monitor (CGM). Glucose measurements are sent to the insulin pump in real time, which uses a specialized algorithm to determine whether insulin delivery should be up-titrated, down-titrated, or suspended.6
Several studies have shown that AIP technology reduces glucose variability and increases the percentage of time within the optimal glucose range.1-3,7 Its safety is especially indicated for patients with long-standing DM who often have hypoglycemia unawareness and recurrent episodes of hypoglycemia.7 Safety is the major advantage of the hybrid closed-loop system as long duration of DM makes patients particularly prone to emergency department (ED) visits and hospitalizations for severe hypoglycemia.8 Recurrent hypoglycemia also is associated with increased cardiovascular mortality in epidemiologic studies.9
Safety was the primary endpoint in the pivotal trial in a multicenter clinical study where 124 participants (mean age, 37.8 years; DM duration, 21.7 years; hemoglobin A1c [HbA1c], 7.4%) were monitored for 3 months while using a hybrid closed-loop pump, similar to the one used in our study.10 Remarkably, there were no device-related episodes of severe hypoglycemia or ketoacidosis. There was even a small but significant difference in HbA1c (7.4% at baseline, 6.9% at 3 months) and of the time in target range measured by CGM from 66.7% at baseline to 72.2% at 3 months). However, the mean age of the population studied was young (mean age, 37.8 years). It is unclear how these results would translate for a population of older patients with T1DM. Moreover, use of AIP systems have not been systematically tested outside of carefully controlled studies, as it would be in middle-aged veterans followed in outpatient US Department of Veterans Affairs (VA) clinics. Such an approach in the context of optimal glucose monitoring combined with use of structured DM education can significantly reduce impaired awareness of hypoglycemia in patients with T1DM of long duration.11
This is the first study to assess the feasibility of AIP technology in a real-world population of older veterans with T1DM in terms of safety and acceptability, because AIP has just recently become available for patient care in the Veterans Health Administration (VHA). This group of patients is of particular interest because they have been largely overlooked in earlier studies. They represent an older population with long-standing DM where hypoglycemia unawareness is often recurrent and incapacitating. In addition, long-standing DM makes optimal glycemic control mandatory to prevent microvascular complications.
Methods
In this retrospective review study,, we examined available data in patients with T1DM at the Malcom Randall VA Medical Center diabetes clinic in Gainesville, Florida, between March and December of 2018 who agreed to use AIP. In this clinic, the AIP system was offered to T1DM patients when the 4-year warranty of a previous insulin pump expired, they had frequent hypoglycemic events, or they were on multiple daily injections and were proficient with carbohydrate counting and adjusting insulin doses and willing to use an insulin pump. Veterans were trained on AIP use by a certified diabetes educator and pump trainer in sessions that lasted 2 to 4 hours depending on previous experience with AIP. Institutional review board approval was obtained at the University of Florida.
Demographic and clinical data before and after the initiation of AIP were collected, including standard insulin pump/CGM information for the Medtronic 670G and Guardian 3 Sensor AIPs. Several variables were evaluated, including age, gender, year of DM diagnosis, time of initiation of AIP, HbA1c, download data (percentage sensor wear, time in automated mode and manual mode, time in/above/below range, bolus information, insulin use, average sensor blood glucose, average meter blood glucose, pump settings), weight, body mass index (BMI), glucose meter information, history of hypoglycemia unawareness.
The primary outcome for this study was safety as assessed by percentage of time below target range on glucose sensor (time below target range is defined as < 70 mg/dL). We also addressed the secondary endpoint of efficacy as the percentage of time in-range defined as blood glucose per glucose sensor of 70 mg/dL to 180 mg/dL (efficacy), percentage of glucose sensor wear, and HbA1c.
Statistics
Comparisons of changes in continuous variables between groups were performed by an analysis of covariance (ANCOVA), adjusting for baseline levels. Fisher exact test (χ2) and unpaired t test were used to compare group differences at baseline for categorical and continuous variables, respectively, while Wilcoxon rank sum test was used for nonnormally distributed values. Changes in continuous measures within the same group were tested by paired t test or Wilcoxon matched-pairs signed rank test when applicable. Analyses were performed using Stata 11.0.
Results
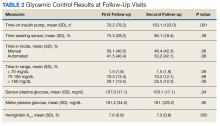
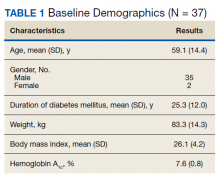
Thirty-seven veterans with T1DM using AIPs in 2018 were evaluated at baseline and at follow up visits (Tables 1 and 2). Time frame for follow-up was approximately 3 months, although there was some variation. Of note, the mean weight and BMI corresponded to mostly lean individuals, consistent with the diagnosis of T1DM.
Time below target range hypoglycemia (sensor glucose < 70 mg/dL) remained low at each follow-up visit (both 1.5%). Percentage of time in automated mode increased from first to second follow-up visit after initiation of AIP (41% vs 53%, P = .06). Percentage of sensor wear numerically increased from first to second follow-up visit (75% vs 85%, P = .39), same as time in range, defined as sensor glucose 70 to 180 mg/dL, from first to second follow-up visit (70% vs 73%, P = .09). Time above range, defined as sensor glucose > 180 mg/dL, demonstrated a strong trend toward decreasing between follow-up appointments (29% to 25%; P = .09). HbA1c decreased from 7.6% to 7.3% (P = .005).
About half of the patients (18 of 37) reported hypoglycemia unawareness before the initiation of the 670G AIP. On follow-up visit 61% (11 of 18) reported significant improvement in awareness. Of the remaining 18 patients who reported normal awareness before automated mode, 17% (3 of 18) described a new onset unawareness.
Discussion
This study evaluated the safety of adopting a new DM technology in the real world of an outpatient VA clinic. To the best of our knowledge, this is the first study evaluating the use of AIP specifically in a population of middle-aged veterans with longstanding T1DM. After a mean 7 months of follow-up, participants accepted AIP use as evidenced by increased sensor wear over time and experienced improvements in DM measures that indicate successful use (ie, time in automated mode, which represents reduced glycemic variability). These results show success of an AIP approach in a demographically older group of patients.
AIP has been shown to have positive effects on glycemic control such as time in target glucose range (goal ≥ 70%). In our relatively small pilot study, there was trend for an improvement in the time in range from the first to second clinical follow-up visit, suggesting true patient involvement with the use of the device. Studies involving overall younger cohorts have proved that AIP technology is safe and efficacious for outpatient management of T1DM.7,10,12,13 However, they were all conducted under the safety of a research setting, and trials enrolled a younger population believed to adapt with more ease to this new technology. Tauschmann and colleagues performed a multicenter, parallel randomized controlled trial that compared hybrid closed-loop AIP therapy with sensor-augmented pump therapy in patients with suboptimal T1DM control.12 Results showed that the hybrid closed-loop system increased the time that the glucose concentration was within the target range (70-180 mg/dL) from 54% in the sensor-augmented pump group to 65% on the closed-loop system (P < .001). A small but significant improvement in HBA1c (from 8.0 -7.4%) and low rates of hypoglycemia (2.6% of time below 70 mg/dL) were also noted.12
A similar benefit was observed in a 2019 landmark study by Brown and colleagues of 168 patients with T1DM at 7 university medical centers who were treated for 6 months with either a closed-loop system (closed-loop group) or a sensor-augmented pump (control group) in a parallel-group, unblinded, randomized trial study.13 Mean (SD) time in the target range increased in the closed-loop group from 61% (17) at baseline to 71% (12) during the 6 months. HbA1c decreased from 7.4 to 7.1% and time ≤ 70 mg/dL was just 1.6%. However, only 13% of patients were aged ≥ 40 years in the study by Tauschmann and colleagues, and mean age was 33 years in the Brown and colleagues study.12,13 In contrast, the mean (SD) age in our study was 59 (14) years. Our pilot study also showed comparable, or somewhat better results, as mean time in target range was 72%, HbA1c was 7.3%, and time ≤ 70 mg/dL was just 1.5%.
In the only other single-center study in adults with T1DM (mean age 45 years), Faulds and colleagues evaluated changes in glycemic control and adherence in patient using the same hybrid closed-loop system.14 Treatment resulted in a decrease in HbA1c compared with baseline similar to our study, most notably for patients who had higher baseline HbA1c. However, over its short duration (6 to 12 weeks), there was decreased time in automated mode in study patients, likely due to treatment burden. Our study in older patients showed a similar reduction in HbA1c from baseline up to the 7-month visit but with increased sensor wear and time in automated mode.
There are many possible reasons for improved time in target range in our older population. Contrary to common belief that older age may be a barrier to adopting complex technology, it is likely that older age and longer duration of DM motivates adherence to a therapy that reduces glucose swings, offers a greater sense of safety and control, and improves quality of life. This is underscored by improvements over time in sensor wear and time in automated mode, measures of adherence, and successful AIP management. In support of a motivation factor to adopt insulin pump therapy in patients with long-standing T1DM, Faulds and colleagues found that older age and higher baseline HbA1c were associated with less time spent in hypoglycemia.14
The close supervision of patients by a certified diabetes educator and pump trainer may have helped improve glycemic control. Veterans received initial training, weekly follow-ups for 4 to 5 visits, and then bimonthly visits. There was also good access to the DM care team through a secure VA messaging system. This allowed for prompt troubleshooting and gave veterans the support they needed for the successful technology adoption.
The use of real-time CGM led to improvements in hypoglycemia unawareness. The nature of automated insulin delivery not only allows the patient to use a immediate CGM, but automatically lowers the delivery of insulin, further minimizing the risk of hypoglycemia.15 This combined approach explains the improvement in self-reported hypoglycemia unawareness in our cohort which decreased by 61%. As in our study, very recently Pratley and colleagues reported in a 6-month follow-up study that the greatest benefit of CGM was not the -0.3% improvement of glycemic control (similar in magnitude to our study) but the 47% decrease in the primary outcome of CGM-measured time in hypoglycemia.16
Hybrid closed-loop insulin delivery improves glucose control while reducing the risk of hypoglycemia. There is consensus that this approach is cost-effective and saves resources in the management of these complex patients, so prone to severe microvascular complications and hypoglycemia.17,18 A recent analysis by Pease and colleagues concluded that the hybrid closed-loop system was safer and more cost-effective when compared with the current standard of care, comprising insulin injections and capillary glucose testing.19 This held true even after several sensitivity analyses were performed, including baseline glycemic control, treatment effects, technology costs, age, and time horizon. This is relevant to the VHA, which at all times must consider the most cost-effective approach. Therefore, while there is no such debate about the cost-effectiveness of AIP technology for younger adults with T1DM, this study closes the knowledge gap for middle-aged veterans.7,10,12,13 The current study demonstrates that even for older patients with long-standing T1DM, when proper access to supplies and support services are made available, treatment is associated with considerable success.
Finally, AIP is well suited for telehealth applications. Data can be uploaded remotely and sent to VA health care providers, which can facilitate care without the need to travel. Distance is often a barrier for access and optimal care of veterans. The current COVID-19 pandemic is another barrier to access that may persist in the near future and adds value to AIP management.
There were a few challenges with use of AIP. Although transition to AIP was smooth for most patients already on insulin pump therapy, several noted requests for calibration in the middle of the night in automated mode, which affected sleep. Also, AIP technology requires some computer literacy to navigate the menu and address sensor calibrations, which can be a challenge for some. Based on our results, we would recommend AIP in veterans who are appropriately trained in carbohydrate counting, understand the principles of insulin therapy, and are able to navigate a computer screen menu. Most T1DM patients already using insulin pump meet those recommendations, thus, they are good candidates.
Limitations
There are some limitations to our study. The small sample size and single-center nature prevent generalization. Also, the veteran population cannot be extrapolated to other populations. For instance, the majority of the patients in this study were male.
Conclusions
We report that an AIP approach for patients with long-standing T1DM is well accepted and engages patients into monitoring their blood sugars and achieving better glycemic control. This was achieved with minimal hypoglycemia in a population where often hypoglycemia unawareness makes DM care a challenge. Future studies within the VHA are needed to fully assess the long-term benefits and cost-effectiveness of this technology in veterans.
1. Saunders A, Messer LH, Forlenza GP. MiniMed 670G hybrid closed loop artificial pancreas system for the treatment of type 1 diabetes mellitus: overview of its safety and efficacy. Expert Rev Med Devices. 2019;16(10):845-853. doi:10.1080/17434440.2019.1670639
2. Beato-Víbora PI, Quirós-López C, Lázaro-Martín L, et al. Impact of sensor-augmented pump therapy with predictive low-glucose suspend function on glycemic control and patient satisfaction in adults and children with type 1 diabetes. Diabetes Technol Ther. 2018;20(11):738-743. doi:10.1089/dia.2018.0199
3. De Ridder F, den Brinker M, De Block C. The road from intermittently scanned continuous glucose monitoring to hybrid closed-loop systems. Part B: results from randomized controlled trials. Ther Adv Endocrinol Metab. 2019;10:2042018819871903. Published 2019 Aug 30. doi:10.1177/2042018819871903
4. Monnier L, Colette C, Wojtusciszyn A, et al. Toward defining the threshold between low and high glucose variability in dabetes. Diabetes Care. 2017;40(7):832-838. doi:10.2337/dc16-1769
5. Monnier L, Colette C, Owens DR. The application of simple metrics in the assessment of glycaemic variability. Diabetes Metab. 2018;44(4):313-319. doi:10.1016/j.diabet.2018.02.008
6. Thabit H, Hovorka R. Coming of age: the artificial pancreas for type 1 diabetes. Diabetologia. 2016;59(9):1795-1805. doi:10.1007/s00125-016-4022-4
7. Anderson SM, Buckingham BA, Breton MD, et al. Hybrid closed-loop control is safe and effective for people with type 1 diabetes who are at moderate to high risk for hypoglycemia. Diabetes Technol Ther. 2019;21(6):356-363. doi:10.1089/dia.2019.0018
8. Liu J, Wang R, Ganz ML, Paprocki Y, Schneider D, Weatherall J. The burden of severe hypoglycemia in type 1 diabetes. Curr Med Res Opin. 2018;34(1):171-177. doi:10.1080/03007995.2017.1391079
9. Rawshani A, Sattar N, Franzén S, et al. Excess mortality and cardiovascular disease in young adults with type 1 diabetes in relation to age at onset: a nationwide, register-based cohort study. Lancet. 2018;392(10146):477-486. doi:10.1016/S0140-6736(18)31506-X
10. Bergenstal RM, Garg S, Weinzimer SA, et al. Safety of a hybrid closed-loop insulin delivery system in patients with type 1 diabetes. JAMA. 2016;316(13):1407-1408. doi:10.1001/jama.2016.11708
11. Little SA, Speight J, Leelarathna L, et al. Sustained reduction in severe hypoglycemia in adults with type 1 diabetes complicated by impaired awareness of hypoglycemia: two-year follow-up in the HypoCOMPaSS randomized clinical trial. Diabetes Care. 2018;41(8):1600-1607. doi:10.2337/dc17-2682
12. Tauschmann M, Thabit H, Bally L, et al. Closed-loop insulin delivery in suboptimally controlled type 1 diabetes: a multicentre, 12-week randomised trial [published correction appears in Lancet. 2018 Oct 13;392(10155):1310]. Lancet. 2018;392(10155):1321-1329. doi:10.1016/S0140-6736(18)31947-0
13. Brown SA, Kovatchev BP, Raghinaru D, et al. Six-month randomized, multicenter trial of closed-loop control in type 1 diabetes. N Engl J Med. 2019;381(18):1707-1717. doi:10.1056/NEJMoa1907863
14. Faulds ER, Zappe J, Dungan KM. Real-world implications of hybrid close loop (HCL) insulin delivery system. Endocr Pract. 2019;25(5):477-484. doi:10.4158/EP-2018-0515
15. Rickels MR, Peleckis AJ, Dalton-Bakes C, et al. Continuous glucose monitoring for hypoglycemia avoidance and glucose counterregulation in long-standing type 1 diabetes. J Clin Endocrinol Metab. 2018;103(1):105-114. doi:10.1210/jc.2017-01516
16. Pratley RE, Kanapka LG, Rickels MR, et al. Effect of continuous glucose monitoring on hypoglycemia in older adults with type 1 diabetes: a randomized clinical trial. JAMA. 2020;323(23):2397-2406. doi:10.1001/jama.2020.6928
17. Bekiari E, Kitsios K, Thabit H, et al. Artificial pancreas treatment for outpatients with type 1 diabetes: systematic review and meta-analysis. BMJ. 2018;361:k1310. Published 2018 Apr 18. doi:10.1136/bmj.k1310
18. American Diabetes Association. Addendum. 7. Diabetes technology: standards of medical care in diabetes-2020. Diabetes Care. 2020;43(suppl 1):S77-S88. Diabetes Care. 2020;43(8):1981. doi:10.2337/dc20-ad08c
19. Pease A, Zomer E, Liew D, et al. Cost-effectiveness analysis of a hybrid closed-loop system versus multiple daily injections and capillary glucose testing for adults with type 1 dabetes. Diabetes Technol Ther. 2020;22(11):812-821. doi:10.1089/dia.2020.0064
Insulin pump technology has been available since the 1970s. Innovation in insulin pumps has had significant impact on the management of diabetes mellitus (DM). In recent years, automated insulin pump technology (AIP) has proven to be a safe and effective way to treat DM. It has been studied mostly in highly organized randomized controlled trials (RCTs) in younger populations with type 1 DM (T1DM).1-3
One of the challenges in DM care has always been the wide variations in daily plasma glucose concentration that often cause major swings of hyperglycemia and hypoglycemia. Extreme variations in blood glucose have also been linked to adverse outcomes, including poor micro- and macrovascular outcomes.4,5 AIP technology is a hybrid closed-loop system that attempts to solve this problem by adjusting insulin delivery in response to real-time glucose information from a continuous glucose monitor (CGM). Glucose measurements are sent to the insulin pump in real time, which uses a specialized algorithm to determine whether insulin delivery should be up-titrated, down-titrated, or suspended.6
Several studies have shown that AIP technology reduces glucose variability and increases the percentage of time within the optimal glucose range.1-3,7 Its safety is especially indicated for patients with long-standing DM who often have hypoglycemia unawareness and recurrent episodes of hypoglycemia.7 Safety is the major advantage of the hybrid closed-loop system as long duration of DM makes patients particularly prone to emergency department (ED) visits and hospitalizations for severe hypoglycemia.8 Recurrent hypoglycemia also is associated with increased cardiovascular mortality in epidemiologic studies.9
Safety was the primary endpoint in the pivotal trial in a multicenter clinical study where 124 participants (mean age, 37.8 years; DM duration, 21.7 years; hemoglobin A1c [HbA1c], 7.4%) were monitored for 3 months while using a hybrid closed-loop pump, similar to the one used in our study.10 Remarkably, there were no device-related episodes of severe hypoglycemia or ketoacidosis. There was even a small but significant difference in HbA1c (7.4% at baseline, 6.9% at 3 months) and of the time in target range measured by CGM from 66.7% at baseline to 72.2% at 3 months). However, the mean age of the population studied was young (mean age, 37.8 years). It is unclear how these results would translate for a population of older patients with T1DM. Moreover, use of AIP systems have not been systematically tested outside of carefully controlled studies, as it would be in middle-aged veterans followed in outpatient US Department of Veterans Affairs (VA) clinics. Such an approach in the context of optimal glucose monitoring combined with use of structured DM education can significantly reduce impaired awareness of hypoglycemia in patients with T1DM of long duration.11
This is the first study to assess the feasibility of AIP technology in a real-world population of older veterans with T1DM in terms of safety and acceptability, because AIP has just recently become available for patient care in the Veterans Health Administration (VHA). This group of patients is of particular interest because they have been largely overlooked in earlier studies. They represent an older population with long-standing DM where hypoglycemia unawareness is often recurrent and incapacitating. In addition, long-standing DM makes optimal glycemic control mandatory to prevent microvascular complications.
Methods
In this retrospective review study,, we examined available data in patients with T1DM at the Malcom Randall VA Medical Center diabetes clinic in Gainesville, Florida, between March and December of 2018 who agreed to use AIP. In this clinic, the AIP system was offered to T1DM patients when the 4-year warranty of a previous insulin pump expired, they had frequent hypoglycemic events, or they were on multiple daily injections and were proficient with carbohydrate counting and adjusting insulin doses and willing to use an insulin pump. Veterans were trained on AIP use by a certified diabetes educator and pump trainer in sessions that lasted 2 to 4 hours depending on previous experience with AIP. Institutional review board approval was obtained at the University of Florida.
Demographic and clinical data before and after the initiation of AIP were collected, including standard insulin pump/CGM information for the Medtronic 670G and Guardian 3 Sensor AIPs. Several variables were evaluated, including age, gender, year of DM diagnosis, time of initiation of AIP, HbA1c, download data (percentage sensor wear, time in automated mode and manual mode, time in/above/below range, bolus information, insulin use, average sensor blood glucose, average meter blood glucose, pump settings), weight, body mass index (BMI), glucose meter information, history of hypoglycemia unawareness.
The primary outcome for this study was safety as assessed by percentage of time below target range on glucose sensor (time below target range is defined as < 70 mg/dL). We also addressed the secondary endpoint of efficacy as the percentage of time in-range defined as blood glucose per glucose sensor of 70 mg/dL to 180 mg/dL (efficacy), percentage of glucose sensor wear, and HbA1c.
Statistics
Comparisons of changes in continuous variables between groups were performed by an analysis of covariance (ANCOVA), adjusting for baseline levels. Fisher exact test (χ2) and unpaired t test were used to compare group differences at baseline for categorical and continuous variables, respectively, while Wilcoxon rank sum test was used for nonnormally distributed values. Changes in continuous measures within the same group were tested by paired t test or Wilcoxon matched-pairs signed rank test when applicable. Analyses were performed using Stata 11.0.
Results
Thirty-seven veterans with T1DM using AIPs in 2018 were evaluated at baseline and at follow up visits (Tables 1 and 2). Time frame for follow-up was approximately 3 months, although there was some variation. Of note, the mean weight and BMI corresponded to mostly lean individuals, consistent with the diagnosis of T1DM.
Time below target range hypoglycemia (sensor glucose < 70 mg/dL) remained low at each follow-up visit (both 1.5%). Percentage of time in automated mode increased from first to second follow-up visit after initiation of AIP (41% vs 53%, P = .06). Percentage of sensor wear numerically increased from first to second follow-up visit (75% vs 85%, P = .39), same as time in range, defined as sensor glucose 70 to 180 mg/dL, from first to second follow-up visit (70% vs 73%, P = .09). Time above range, defined as sensor glucose > 180 mg/dL, demonstrated a strong trend toward decreasing between follow-up appointments (29% to 25%; P = .09). HbA1c decreased from 7.6% to 7.3% (P = .005).
About half of the patients (18 of 37) reported hypoglycemia unawareness before the initiation of the 670G AIP. On follow-up visit 61% (11 of 18) reported significant improvement in awareness. Of the remaining 18 patients who reported normal awareness before automated mode, 17% (3 of 18) described a new onset unawareness.
Discussion
This study evaluated the safety of adopting a new DM technology in the real world of an outpatient VA clinic. To the best of our knowledge, this is the first study evaluating the use of AIP specifically in a population of middle-aged veterans with longstanding T1DM. After a mean 7 months of follow-up, participants accepted AIP use as evidenced by increased sensor wear over time and experienced improvements in DM measures that indicate successful use (ie, time in automated mode, which represents reduced glycemic variability). These results show success of an AIP approach in a demographically older group of patients.
AIP has been shown to have positive effects on glycemic control such as time in target glucose range (goal ≥ 70%). In our relatively small pilot study, there was trend for an improvement in the time in range from the first to second clinical follow-up visit, suggesting true patient involvement with the use of the device. Studies involving overall younger cohorts have proved that AIP technology is safe and efficacious for outpatient management of T1DM.7,10,12,13 However, they were all conducted under the safety of a research setting, and trials enrolled a younger population believed to adapt with more ease to this new technology. Tauschmann and colleagues performed a multicenter, parallel randomized controlled trial that compared hybrid closed-loop AIP therapy with sensor-augmented pump therapy in patients with suboptimal T1DM control.12 Results showed that the hybrid closed-loop system increased the time that the glucose concentration was within the target range (70-180 mg/dL) from 54% in the sensor-augmented pump group to 65% on the closed-loop system (P < .001). A small but significant improvement in HBA1c (from 8.0 -7.4%) and low rates of hypoglycemia (2.6% of time below 70 mg/dL) were also noted.12
A similar benefit was observed in a 2019 landmark study by Brown and colleagues of 168 patients with T1DM at 7 university medical centers who were treated for 6 months with either a closed-loop system (closed-loop group) or a sensor-augmented pump (control group) in a parallel-group, unblinded, randomized trial study.13 Mean (SD) time in the target range increased in the closed-loop group from 61% (17) at baseline to 71% (12) during the 6 months. HbA1c decreased from 7.4 to 7.1% and time ≤ 70 mg/dL was just 1.6%. However, only 13% of patients were aged ≥ 40 years in the study by Tauschmann and colleagues, and mean age was 33 years in the Brown and colleagues study.12,13 In contrast, the mean (SD) age in our study was 59 (14) years. Our pilot study also showed comparable, or somewhat better results, as mean time in target range was 72%, HbA1c was 7.3%, and time ≤ 70 mg/dL was just 1.5%.
In the only other single-center study in adults with T1DM (mean age 45 years), Faulds and colleagues evaluated changes in glycemic control and adherence in patient using the same hybrid closed-loop system.14 Treatment resulted in a decrease in HbA1c compared with baseline similar to our study, most notably for patients who had higher baseline HbA1c. However, over its short duration (6 to 12 weeks), there was decreased time in automated mode in study patients, likely due to treatment burden. Our study in older patients showed a similar reduction in HbA1c from baseline up to the 7-month visit but with increased sensor wear and time in automated mode.
There are many possible reasons for improved time in target range in our older population. Contrary to common belief that older age may be a barrier to adopting complex technology, it is likely that older age and longer duration of DM motivates adherence to a therapy that reduces glucose swings, offers a greater sense of safety and control, and improves quality of life. This is underscored by improvements over time in sensor wear and time in automated mode, measures of adherence, and successful AIP management. In support of a motivation factor to adopt insulin pump therapy in patients with long-standing T1DM, Faulds and colleagues found that older age and higher baseline HbA1c were associated with less time spent in hypoglycemia.14
The close supervision of patients by a certified diabetes educator and pump trainer may have helped improve glycemic control. Veterans received initial training, weekly follow-ups for 4 to 5 visits, and then bimonthly visits. There was also good access to the DM care team through a secure VA messaging system. This allowed for prompt troubleshooting and gave veterans the support they needed for the successful technology adoption.
The use of real-time CGM led to improvements in hypoglycemia unawareness. The nature of automated insulin delivery not only allows the patient to use a immediate CGM, but automatically lowers the delivery of insulin, further minimizing the risk of hypoglycemia.15 This combined approach explains the improvement in self-reported hypoglycemia unawareness in our cohort which decreased by 61%. As in our study, very recently Pratley and colleagues reported in a 6-month follow-up study that the greatest benefit of CGM was not the -0.3% improvement of glycemic control (similar in magnitude to our study) but the 47% decrease in the primary outcome of CGM-measured time in hypoglycemia.16
Hybrid closed-loop insulin delivery improves glucose control while reducing the risk of hypoglycemia. There is consensus that this approach is cost-effective and saves resources in the management of these complex patients, so prone to severe microvascular complications and hypoglycemia.17,18 A recent analysis by Pease and colleagues concluded that the hybrid closed-loop system was safer and more cost-effective when compared with the current standard of care, comprising insulin injections and capillary glucose testing.19 This held true even after several sensitivity analyses were performed, including baseline glycemic control, treatment effects, technology costs, age, and time horizon. This is relevant to the VHA, which at all times must consider the most cost-effective approach. Therefore, while there is no such debate about the cost-effectiveness of AIP technology for younger adults with T1DM, this study closes the knowledge gap for middle-aged veterans.7,10,12,13 The current study demonstrates that even for older patients with long-standing T1DM, when proper access to supplies and support services are made available, treatment is associated with considerable success.
Finally, AIP is well suited for telehealth applications. Data can be uploaded remotely and sent to VA health care providers, which can facilitate care without the need to travel. Distance is often a barrier for access and optimal care of veterans. The current COVID-19 pandemic is another barrier to access that may persist in the near future and adds value to AIP management.
There were a few challenges with use of AIP. Although transition to AIP was smooth for most patients already on insulin pump therapy, several noted requests for calibration in the middle of the night in automated mode, which affected sleep. Also, AIP technology requires some computer literacy to navigate the menu and address sensor calibrations, which can be a challenge for some. Based on our results, we would recommend AIP in veterans who are appropriately trained in carbohydrate counting, understand the principles of insulin therapy, and are able to navigate a computer screen menu. Most T1DM patients already using insulin pump meet those recommendations, thus, they are good candidates.
Limitations
There are some limitations to our study. The small sample size and single-center nature prevent generalization. Also, the veteran population cannot be extrapolated to other populations. For instance, the majority of the patients in this study were male.
Conclusions
We report that an AIP approach for patients with long-standing T1DM is well accepted and engages patients into monitoring their blood sugars and achieving better glycemic control. This was achieved with minimal hypoglycemia in a population where often hypoglycemia unawareness makes DM care a challenge. Future studies within the VHA are needed to fully assess the long-term benefits and cost-effectiveness of this technology in veterans.
Insulin pump technology has been available since the 1970s. Innovation in insulin pumps has had significant impact on the management of diabetes mellitus (DM). In recent years, automated insulin pump technology (AIP) has proven to be a safe and effective way to treat DM. It has been studied mostly in highly organized randomized controlled trials (RCTs) in younger populations with type 1 DM (T1DM).1-3
One of the challenges in DM care has always been the wide variations in daily plasma glucose concentration that often cause major swings of hyperglycemia and hypoglycemia. Extreme variations in blood glucose have also been linked to adverse outcomes, including poor micro- and macrovascular outcomes.4,5 AIP technology is a hybrid closed-loop system that attempts to solve this problem by adjusting insulin delivery in response to real-time glucose information from a continuous glucose monitor (CGM). Glucose measurements are sent to the insulin pump in real time, which uses a specialized algorithm to determine whether insulin delivery should be up-titrated, down-titrated, or suspended.6
Several studies have shown that AIP technology reduces glucose variability and increases the percentage of time within the optimal glucose range.1-3,7 Its safety is especially indicated for patients with long-standing DM who often have hypoglycemia unawareness and recurrent episodes of hypoglycemia.7 Safety is the major advantage of the hybrid closed-loop system as long duration of DM makes patients particularly prone to emergency department (ED) visits and hospitalizations for severe hypoglycemia.8 Recurrent hypoglycemia also is associated with increased cardiovascular mortality in epidemiologic studies.9
Safety was the primary endpoint in the pivotal trial in a multicenter clinical study where 124 participants (mean age, 37.8 years; DM duration, 21.7 years; hemoglobin A1c [HbA1c], 7.4%) were monitored for 3 months while using a hybrid closed-loop pump, similar to the one used in our study.10 Remarkably, there were no device-related episodes of severe hypoglycemia or ketoacidosis. There was even a small but significant difference in HbA1c (7.4% at baseline, 6.9% at 3 months) and of the time in target range measured by CGM from 66.7% at baseline to 72.2% at 3 months). However, the mean age of the population studied was young (mean age, 37.8 years). It is unclear how these results would translate for a population of older patients with T1DM. Moreover, use of AIP systems have not been systematically tested outside of carefully controlled studies, as it would be in middle-aged veterans followed in outpatient US Department of Veterans Affairs (VA) clinics. Such an approach in the context of optimal glucose monitoring combined with use of structured DM education can significantly reduce impaired awareness of hypoglycemia in patients with T1DM of long duration.11
This is the first study to assess the feasibility of AIP technology in a real-world population of older veterans with T1DM in terms of safety and acceptability, because AIP has just recently become available for patient care in the Veterans Health Administration (VHA). This group of patients is of particular interest because they have been largely overlooked in earlier studies. They represent an older population with long-standing DM where hypoglycemia unawareness is often recurrent and incapacitating. In addition, long-standing DM makes optimal glycemic control mandatory to prevent microvascular complications.
Methods
In this retrospective review study,, we examined available data in patients with T1DM at the Malcom Randall VA Medical Center diabetes clinic in Gainesville, Florida, between March and December of 2018 who agreed to use AIP. In this clinic, the AIP system was offered to T1DM patients when the 4-year warranty of a previous insulin pump expired, they had frequent hypoglycemic events, or they were on multiple daily injections and were proficient with carbohydrate counting and adjusting insulin doses and willing to use an insulin pump. Veterans were trained on AIP use by a certified diabetes educator and pump trainer in sessions that lasted 2 to 4 hours depending on previous experience with AIP. Institutional review board approval was obtained at the University of Florida.
Demographic and clinical data before and after the initiation of AIP were collected, including standard insulin pump/CGM information for the Medtronic 670G and Guardian 3 Sensor AIPs. Several variables were evaluated, including age, gender, year of DM diagnosis, time of initiation of AIP, HbA1c, download data (percentage sensor wear, time in automated mode and manual mode, time in/above/below range, bolus information, insulin use, average sensor blood glucose, average meter blood glucose, pump settings), weight, body mass index (BMI), glucose meter information, history of hypoglycemia unawareness.
The primary outcome for this study was safety as assessed by percentage of time below target range on glucose sensor (time below target range is defined as < 70 mg/dL). We also addressed the secondary endpoint of efficacy as the percentage of time in-range defined as blood glucose per glucose sensor of 70 mg/dL to 180 mg/dL (efficacy), percentage of glucose sensor wear, and HbA1c.
Statistics
Comparisons of changes in continuous variables between groups were performed by an analysis of covariance (ANCOVA), adjusting for baseline levels. Fisher exact test (χ2) and unpaired t test were used to compare group differences at baseline for categorical and continuous variables, respectively, while Wilcoxon rank sum test was used for nonnormally distributed values. Changes in continuous measures within the same group were tested by paired t test or Wilcoxon matched-pairs signed rank test when applicable. Analyses were performed using Stata 11.0.
Results
Thirty-seven veterans with T1DM using AIPs in 2018 were evaluated at baseline and at follow up visits (Tables 1 and 2). Time frame for follow-up was approximately 3 months, although there was some variation. Of note, the mean weight and BMI corresponded to mostly lean individuals, consistent with the diagnosis of T1DM.
Time below target range hypoglycemia (sensor glucose < 70 mg/dL) remained low at each follow-up visit (both 1.5%). Percentage of time in automated mode increased from first to second follow-up visit after initiation of AIP (41% vs 53%, P = .06). Percentage of sensor wear numerically increased from first to second follow-up visit (75% vs 85%, P = .39), same as time in range, defined as sensor glucose 70 to 180 mg/dL, from first to second follow-up visit (70% vs 73%, P = .09). Time above range, defined as sensor glucose > 180 mg/dL, demonstrated a strong trend toward decreasing between follow-up appointments (29% to 25%; P = .09). HbA1c decreased from 7.6% to 7.3% (P = .005).
About half of the patients (18 of 37) reported hypoglycemia unawareness before the initiation of the 670G AIP. On follow-up visit 61% (11 of 18) reported significant improvement in awareness. Of the remaining 18 patients who reported normal awareness before automated mode, 17% (3 of 18) described a new onset unawareness.
Discussion
This study evaluated the safety of adopting a new DM technology in the real world of an outpatient VA clinic. To the best of our knowledge, this is the first study evaluating the use of AIP specifically in a population of middle-aged veterans with longstanding T1DM. After a mean 7 months of follow-up, participants accepted AIP use as evidenced by increased sensor wear over time and experienced improvements in DM measures that indicate successful use (ie, time in automated mode, which represents reduced glycemic variability). These results show success of an AIP approach in a demographically older group of patients.
AIP has been shown to have positive effects on glycemic control such as time in target glucose range (goal ≥ 70%). In our relatively small pilot study, there was trend for an improvement in the time in range from the first to second clinical follow-up visit, suggesting true patient involvement with the use of the device. Studies involving overall younger cohorts have proved that AIP technology is safe and efficacious for outpatient management of T1DM.7,10,12,13 However, they were all conducted under the safety of a research setting, and trials enrolled a younger population believed to adapt with more ease to this new technology. Tauschmann and colleagues performed a multicenter, parallel randomized controlled trial that compared hybrid closed-loop AIP therapy with sensor-augmented pump therapy in patients with suboptimal T1DM control.12 Results showed that the hybrid closed-loop system increased the time that the glucose concentration was within the target range (70-180 mg/dL) from 54% in the sensor-augmented pump group to 65% on the closed-loop system (P < .001). A small but significant improvement in HBA1c (from 8.0 -7.4%) and low rates of hypoglycemia (2.6% of time below 70 mg/dL) were also noted.12
A similar benefit was observed in a 2019 landmark study by Brown and colleagues of 168 patients with T1DM at 7 university medical centers who were treated for 6 months with either a closed-loop system (closed-loop group) or a sensor-augmented pump (control group) in a parallel-group, unblinded, randomized trial study.13 Mean (SD) time in the target range increased in the closed-loop group from 61% (17) at baseline to 71% (12) during the 6 months. HbA1c decreased from 7.4 to 7.1% and time ≤ 70 mg/dL was just 1.6%. However, only 13% of patients were aged ≥ 40 years in the study by Tauschmann and colleagues, and mean age was 33 years in the Brown and colleagues study.12,13 In contrast, the mean (SD) age in our study was 59 (14) years. Our pilot study also showed comparable, or somewhat better results, as mean time in target range was 72%, HbA1c was 7.3%, and time ≤ 70 mg/dL was just 1.5%.
In the only other single-center study in adults with T1DM (mean age 45 years), Faulds and colleagues evaluated changes in glycemic control and adherence in patient using the same hybrid closed-loop system.14 Treatment resulted in a decrease in HbA1c compared with baseline similar to our study, most notably for patients who had higher baseline HbA1c. However, over its short duration (6 to 12 weeks), there was decreased time in automated mode in study patients, likely due to treatment burden. Our study in older patients showed a similar reduction in HbA1c from baseline up to the 7-month visit but with increased sensor wear and time in automated mode.
There are many possible reasons for improved time in target range in our older population. Contrary to common belief that older age may be a barrier to adopting complex technology, it is likely that older age and longer duration of DM motivates adherence to a therapy that reduces glucose swings, offers a greater sense of safety and control, and improves quality of life. This is underscored by improvements over time in sensor wear and time in automated mode, measures of adherence, and successful AIP management. In support of a motivation factor to adopt insulin pump therapy in patients with long-standing T1DM, Faulds and colleagues found that older age and higher baseline HbA1c were associated with less time spent in hypoglycemia.14
The close supervision of patients by a certified diabetes educator and pump trainer may have helped improve glycemic control. Veterans received initial training, weekly follow-ups for 4 to 5 visits, and then bimonthly visits. There was also good access to the DM care team through a secure VA messaging system. This allowed for prompt troubleshooting and gave veterans the support they needed for the successful technology adoption.
The use of real-time CGM led to improvements in hypoglycemia unawareness. The nature of automated insulin delivery not only allows the patient to use a immediate CGM, but automatically lowers the delivery of insulin, further minimizing the risk of hypoglycemia.15 This combined approach explains the improvement in self-reported hypoglycemia unawareness in our cohort which decreased by 61%. As in our study, very recently Pratley and colleagues reported in a 6-month follow-up study that the greatest benefit of CGM was not the -0.3% improvement of glycemic control (similar in magnitude to our study) but the 47% decrease in the primary outcome of CGM-measured time in hypoglycemia.16
Hybrid closed-loop insulin delivery improves glucose control while reducing the risk of hypoglycemia. There is consensus that this approach is cost-effective and saves resources in the management of these complex patients, so prone to severe microvascular complications and hypoglycemia.17,18 A recent analysis by Pease and colleagues concluded that the hybrid closed-loop system was safer and more cost-effective when compared with the current standard of care, comprising insulin injections and capillary glucose testing.19 This held true even after several sensitivity analyses were performed, including baseline glycemic control, treatment effects, technology costs, age, and time horizon. This is relevant to the VHA, which at all times must consider the most cost-effective approach. Therefore, while there is no such debate about the cost-effectiveness of AIP technology for younger adults with T1DM, this study closes the knowledge gap for middle-aged veterans.7,10,12,13 The current study demonstrates that even for older patients with long-standing T1DM, when proper access to supplies and support services are made available, treatment is associated with considerable success.
Finally, AIP is well suited for telehealth applications. Data can be uploaded remotely and sent to VA health care providers, which can facilitate care without the need to travel. Distance is often a barrier for access and optimal care of veterans. The current COVID-19 pandemic is another barrier to access that may persist in the near future and adds value to AIP management.
There were a few challenges with use of AIP. Although transition to AIP was smooth for most patients already on insulin pump therapy, several noted requests for calibration in the middle of the night in automated mode, which affected sleep. Also, AIP technology requires some computer literacy to navigate the menu and address sensor calibrations, which can be a challenge for some. Based on our results, we would recommend AIP in veterans who are appropriately trained in carbohydrate counting, understand the principles of insulin therapy, and are able to navigate a computer screen menu. Most T1DM patients already using insulin pump meet those recommendations, thus, they are good candidates.
Limitations
There are some limitations to our study. The small sample size and single-center nature prevent generalization. Also, the veteran population cannot be extrapolated to other populations. For instance, the majority of the patients in this study were male.
Conclusions
We report that an AIP approach for patients with long-standing T1DM is well accepted and engages patients into monitoring their blood sugars and achieving better glycemic control. This was achieved with minimal hypoglycemia in a population where often hypoglycemia unawareness makes DM care a challenge. Future studies within the VHA are needed to fully assess the long-term benefits and cost-effectiveness of this technology in veterans.
1. Saunders A, Messer LH, Forlenza GP. MiniMed 670G hybrid closed loop artificial pancreas system for the treatment of type 1 diabetes mellitus: overview of its safety and efficacy. Expert Rev Med Devices. 2019;16(10):845-853. doi:10.1080/17434440.2019.1670639
2. Beato-Víbora PI, Quirós-López C, Lázaro-Martín L, et al. Impact of sensor-augmented pump therapy with predictive low-glucose suspend function on glycemic control and patient satisfaction in adults and children with type 1 diabetes. Diabetes Technol Ther. 2018;20(11):738-743. doi:10.1089/dia.2018.0199
3. De Ridder F, den Brinker M, De Block C. The road from intermittently scanned continuous glucose monitoring to hybrid closed-loop systems. Part B: results from randomized controlled trials. Ther Adv Endocrinol Metab. 2019;10:2042018819871903. Published 2019 Aug 30. doi:10.1177/2042018819871903
4. Monnier L, Colette C, Wojtusciszyn A, et al. Toward defining the threshold between low and high glucose variability in dabetes. Diabetes Care. 2017;40(7):832-838. doi:10.2337/dc16-1769
5. Monnier L, Colette C, Owens DR. The application of simple metrics in the assessment of glycaemic variability. Diabetes Metab. 2018;44(4):313-319. doi:10.1016/j.diabet.2018.02.008
6. Thabit H, Hovorka R. Coming of age: the artificial pancreas for type 1 diabetes. Diabetologia. 2016;59(9):1795-1805. doi:10.1007/s00125-016-4022-4
7. Anderson SM, Buckingham BA, Breton MD, et al. Hybrid closed-loop control is safe and effective for people with type 1 diabetes who are at moderate to high risk for hypoglycemia. Diabetes Technol Ther. 2019;21(6):356-363. doi:10.1089/dia.2019.0018
8. Liu J, Wang R, Ganz ML, Paprocki Y, Schneider D, Weatherall J. The burden of severe hypoglycemia in type 1 diabetes. Curr Med Res Opin. 2018;34(1):171-177. doi:10.1080/03007995.2017.1391079
9. Rawshani A, Sattar N, Franzén S, et al. Excess mortality and cardiovascular disease in young adults with type 1 diabetes in relation to age at onset: a nationwide, register-based cohort study. Lancet. 2018;392(10146):477-486. doi:10.1016/S0140-6736(18)31506-X
10. Bergenstal RM, Garg S, Weinzimer SA, et al. Safety of a hybrid closed-loop insulin delivery system in patients with type 1 diabetes. JAMA. 2016;316(13):1407-1408. doi:10.1001/jama.2016.11708
11. Little SA, Speight J, Leelarathna L, et al. Sustained reduction in severe hypoglycemia in adults with type 1 diabetes complicated by impaired awareness of hypoglycemia: two-year follow-up in the HypoCOMPaSS randomized clinical trial. Diabetes Care. 2018;41(8):1600-1607. doi:10.2337/dc17-2682
12. Tauschmann M, Thabit H, Bally L, et al. Closed-loop insulin delivery in suboptimally controlled type 1 diabetes: a multicentre, 12-week randomised trial [published correction appears in Lancet. 2018 Oct 13;392(10155):1310]. Lancet. 2018;392(10155):1321-1329. doi:10.1016/S0140-6736(18)31947-0
13. Brown SA, Kovatchev BP, Raghinaru D, et al. Six-month randomized, multicenter trial of closed-loop control in type 1 diabetes. N Engl J Med. 2019;381(18):1707-1717. doi:10.1056/NEJMoa1907863
14. Faulds ER, Zappe J, Dungan KM. Real-world implications of hybrid close loop (HCL) insulin delivery system. Endocr Pract. 2019;25(5):477-484. doi:10.4158/EP-2018-0515
15. Rickels MR, Peleckis AJ, Dalton-Bakes C, et al. Continuous glucose monitoring for hypoglycemia avoidance and glucose counterregulation in long-standing type 1 diabetes. J Clin Endocrinol Metab. 2018;103(1):105-114. doi:10.1210/jc.2017-01516
16. Pratley RE, Kanapka LG, Rickels MR, et al. Effect of continuous glucose monitoring on hypoglycemia in older adults with type 1 diabetes: a randomized clinical trial. JAMA. 2020;323(23):2397-2406. doi:10.1001/jama.2020.6928
17. Bekiari E, Kitsios K, Thabit H, et al. Artificial pancreas treatment for outpatients with type 1 diabetes: systematic review and meta-analysis. BMJ. 2018;361:k1310. Published 2018 Apr 18. doi:10.1136/bmj.k1310
18. American Diabetes Association. Addendum. 7. Diabetes technology: standards of medical care in diabetes-2020. Diabetes Care. 2020;43(suppl 1):S77-S88. Diabetes Care. 2020;43(8):1981. doi:10.2337/dc20-ad08c
19. Pease A, Zomer E, Liew D, et al. Cost-effectiveness analysis of a hybrid closed-loop system versus multiple daily injections and capillary glucose testing for adults with type 1 dabetes. Diabetes Technol Ther. 2020;22(11):812-821. doi:10.1089/dia.2020.0064
1. Saunders A, Messer LH, Forlenza GP. MiniMed 670G hybrid closed loop artificial pancreas system for the treatment of type 1 diabetes mellitus: overview of its safety and efficacy. Expert Rev Med Devices. 2019;16(10):845-853. doi:10.1080/17434440.2019.1670639
2. Beato-Víbora PI, Quirós-López C, Lázaro-Martín L, et al. Impact of sensor-augmented pump therapy with predictive low-glucose suspend function on glycemic control and patient satisfaction in adults and children with type 1 diabetes. Diabetes Technol Ther. 2018;20(11):738-743. doi:10.1089/dia.2018.0199
3. De Ridder F, den Brinker M, De Block C. The road from intermittently scanned continuous glucose monitoring to hybrid closed-loop systems. Part B: results from randomized controlled trials. Ther Adv Endocrinol Metab. 2019;10:2042018819871903. Published 2019 Aug 30. doi:10.1177/2042018819871903
4. Monnier L, Colette C, Wojtusciszyn A, et al. Toward defining the threshold between low and high glucose variability in dabetes. Diabetes Care. 2017;40(7):832-838. doi:10.2337/dc16-1769
5. Monnier L, Colette C, Owens DR. The application of simple metrics in the assessment of glycaemic variability. Diabetes Metab. 2018;44(4):313-319. doi:10.1016/j.diabet.2018.02.008
6. Thabit H, Hovorka R. Coming of age: the artificial pancreas for type 1 diabetes. Diabetologia. 2016;59(9):1795-1805. doi:10.1007/s00125-016-4022-4
7. Anderson SM, Buckingham BA, Breton MD, et al. Hybrid closed-loop control is safe and effective for people with type 1 diabetes who are at moderate to high risk for hypoglycemia. Diabetes Technol Ther. 2019;21(6):356-363. doi:10.1089/dia.2019.0018
8. Liu J, Wang R, Ganz ML, Paprocki Y, Schneider D, Weatherall J. The burden of severe hypoglycemia in type 1 diabetes. Curr Med Res Opin. 2018;34(1):171-177. doi:10.1080/03007995.2017.1391079
9. Rawshani A, Sattar N, Franzén S, et al. Excess mortality and cardiovascular disease in young adults with type 1 diabetes in relation to age at onset: a nationwide, register-based cohort study. Lancet. 2018;392(10146):477-486. doi:10.1016/S0140-6736(18)31506-X
10. Bergenstal RM, Garg S, Weinzimer SA, et al. Safety of a hybrid closed-loop insulin delivery system in patients with type 1 diabetes. JAMA. 2016;316(13):1407-1408. doi:10.1001/jama.2016.11708
11. Little SA, Speight J, Leelarathna L, et al. Sustained reduction in severe hypoglycemia in adults with type 1 diabetes complicated by impaired awareness of hypoglycemia: two-year follow-up in the HypoCOMPaSS randomized clinical trial. Diabetes Care. 2018;41(8):1600-1607. doi:10.2337/dc17-2682
12. Tauschmann M, Thabit H, Bally L, et al. Closed-loop insulin delivery in suboptimally controlled type 1 diabetes: a multicentre, 12-week randomised trial [published correction appears in Lancet. 2018 Oct 13;392(10155):1310]. Lancet. 2018;392(10155):1321-1329. doi:10.1016/S0140-6736(18)31947-0
13. Brown SA, Kovatchev BP, Raghinaru D, et al. Six-month randomized, multicenter trial of closed-loop control in type 1 diabetes. N Engl J Med. 2019;381(18):1707-1717. doi:10.1056/NEJMoa1907863
14. Faulds ER, Zappe J, Dungan KM. Real-world implications of hybrid close loop (HCL) insulin delivery system. Endocr Pract. 2019;25(5):477-484. doi:10.4158/EP-2018-0515
15. Rickels MR, Peleckis AJ, Dalton-Bakes C, et al. Continuous glucose monitoring for hypoglycemia avoidance and glucose counterregulation in long-standing type 1 diabetes. J Clin Endocrinol Metab. 2018;103(1):105-114. doi:10.1210/jc.2017-01516
16. Pratley RE, Kanapka LG, Rickels MR, et al. Effect of continuous glucose monitoring on hypoglycemia in older adults with type 1 diabetes: a randomized clinical trial. JAMA. 2020;323(23):2397-2406. doi:10.1001/jama.2020.6928
17. Bekiari E, Kitsios K, Thabit H, et al. Artificial pancreas treatment for outpatients with type 1 diabetes: systematic review and meta-analysis. BMJ. 2018;361:k1310. Published 2018 Apr 18. doi:10.1136/bmj.k1310
18. American Diabetes Association. Addendum. 7. Diabetes technology: standards of medical care in diabetes-2020. Diabetes Care. 2020;43(suppl 1):S77-S88. Diabetes Care. 2020;43(8):1981. doi:10.2337/dc20-ad08c
19. Pease A, Zomer E, Liew D, et al. Cost-effectiveness analysis of a hybrid closed-loop system versus multiple daily injections and capillary glucose testing for adults with type 1 dabetes. Diabetes Technol Ther. 2020;22(11):812-821. doi:10.1089/dia.2020.0064
‘Shocking’ early complications from teen-onset type 2 diabetes
Newly published data show alarmingly high rates and severity of early diabetes-specific complications in individuals who develop type 2 diabetes at a young age. This suggests intervention should be early and aggressive among these youngsters, said the researchers.
The results for the 500 young adult participants in the Treatment Options for Type 2 Diabetes in Adolescents and Youth 2 (TODAY 2) study were published online July 29 in the New England Journal of Medicine by the TODAY study group.
At follow-up – after originally participating in the TODAY trial when they were young teenagers – they had a mean age of 26.4 years.
At this time, more than two thirds had hypertension and half had dyslipidemia.
Overall, 60% had at least one diabetic microvascular complication (retinal disease, neuropathy, or diabetic kidney disease), and more than a quarter had two or more such complications.
“These data illustrate the serious personal and public health consequences of youth-onset type 2 diabetes in the transition to adulthood,” the researchers noted.
Don’t tread lightly just because they are young
“The fact that these youth are accumulating complications at a rapid rate and are broadly affected early in adulthood certainly suggests that aggressive therapy is needed, both for glycemic control and treatment of risk factors like hypertension and dyslipidemia,” study coauthor Philip S. Zeitler, MD, PhD, said in an interview.
“In the absence of studies specifically addressing this, we need to take a more aggressive approach than people might be inclined to, given that the age at diagnosis is young, around 14 years,” he added.
“Contrary to the inclination to be ‘gentle’ in treating them because they are kids, these data suggest that we can’t let these initial years go by without strong intervention, and we need to be prepared for polypharmacy.”
Unfortunately, as Dr. Zeitler and coauthors explained, youth-onset type 2 diabetes is characterized by a suboptimal response to currently approved diabetes medications.
New pediatric indications in the United States for drugs used to treat type 2 diabetes in adults, including the recent Food and Drug Administration approval of extended-release exenatide for children as young as 10 years of age, “helps, but only marginally,” said Dr. Zeitler, of the Clinical & Translational Research Center, Children’s Hospital Colorado, Aurora.
“In some cases, it will help get them covered by carriers, which is always good. But this is still a very limited set of medications. It doesn’t include more recently approved more potent glucagon-like peptide-1 (GLP-1) agonists, like semaglutide, and doesn’t include the sodium-glucose cotransporter 2 (SGLT2) inhibitors. Pediatricians are used to using medications off label and that is necessary here while we await further approvals,” he said.
And he noted that most individuals with youth-onset type 2 diabetes in the United States are covered by public insurance or are uninsured, depending on which state they live in. While the two major Medicaid programs in Colorado allow full access to adult formularies, that’s not the case everywhere. Moreover, patients often face further access barriers in states without expanded Medicaid.
Follow-up shows all metrics worsening over time
In TODAY 2, patients participated in an observational follow-up in their usual care settings in 2011-2020. At the start, they were receiving metformin with or without insulin for diabetes, but whether this continued and whether they were treated for other risk factors was down to individual circumstances.
Participants’ median A1c increased over time, and the percentage with A1c < 6% (< 48 mmol/mol) declined from 75% at the time of TODAY entry to just 19% at the 15-year end of follow-up.
The proportion with an A1c ≤ 10% (≤ 86 mmol/mol) rose from 0% at baseline to 34% at 15 years.
At that time, nearly 50% were taking both metformin and insulin, while more than a quarter were taking no medications.
The prevalence of hypertension increased from 19.2% at baseline to 67.5% at 15 years, while dyslipidemia rose from 20.8% to 51.6%.
Kidney disease prevalence increased from 8.0% at baseline to 54.8% at 15 years. Nerve disease rose from 1.0% to 32.4%. Retinal disease jumped from 13.7% with milder nonproliferative retinopathy in 2010-2011 to 51.0% with any eye disease in 2017-2018, including 8.8% with moderate to severe retinal changes and 3.5% with macular edema.
Overall, at the time of the last visit, 39.9% had no diabetes complications, 31.8% had one, 21.3% had two, and 7.1% had three complications.
Serious cardiovascular events in mid-20s
There were 17 adjudicated serious cardiovascular events, including four myocardial infarctions, six heart failure events, three diagnoses of coronary artery disease, and four strokes.
Six participants died, one each from myocardial infarction, kidney failure, and drug overdose, and three from sepsis.
Dr. Zeitler called the macrovascular events “shocking,” noting that although the numbers are small, for people in their mid-20s “they should be zero ... While we don’t yet know if the rates are the same or faster than in adults, even if they are the same, these kids are only in their late 20s, as opposed to adults experiencing these problems in their 50s, 60s, and 70s.
“The fact that these complications are occurring when these individuals should be in the prime of their life for both family and work has huge implications,” he stressed.
Findings have multiple causes
The reasons for the findings are both biologic and socioeconomic, Dr. Zeitler said.
“We know already that many kids with type 2 have rapid [deterioration of] beta-cell [function], which is probably very biologic. It stands to reason that an individual who can get diabetes as an adolescent probably has more fragile beta cells in some way,” he noted.
“But we also know that many other things contribute: stress, social determinants, access to quality care and medications, access to healthy foods and physical activity, availability of family supervision given the realities of families’ economic status and jobs, etc.”
It’s also known that youth with type 2 diabetes have much more severe insulin resistance than that of adults with the condition, and that “once the kids left ... the [TODAY] study, risk factor treatment in the community was less than ideal, and a lot of kids who met criteria for treatment of their blood pressure or lipids were not being treated. This is likely at least partly sociologic and partly the general pediatric hesitancy to use medications.”
He said the TODAY team will soon have some new data to show that “glycemia during the early years makes a difference, again supporting intensive intervention early on.”
The TODAY study was supported by grants from the National Institute of Diabetes and Digestive and Kidney Diseases. Dr. Zeitler had no further disclosures.
A version of this article first appeared on Medscape.com.
Newly published data show alarmingly high rates and severity of early diabetes-specific complications in individuals who develop type 2 diabetes at a young age. This suggests intervention should be early and aggressive among these youngsters, said the researchers.
The results for the 500 young adult participants in the Treatment Options for Type 2 Diabetes in Adolescents and Youth 2 (TODAY 2) study were published online July 29 in the New England Journal of Medicine by the TODAY study group.
At follow-up – after originally participating in the TODAY trial when they were young teenagers – they had a mean age of 26.4 years.
At this time, more than two thirds had hypertension and half had dyslipidemia.
Overall, 60% had at least one diabetic microvascular complication (retinal disease, neuropathy, or diabetic kidney disease), and more than a quarter had two or more such complications.
“These data illustrate the serious personal and public health consequences of youth-onset type 2 diabetes in the transition to adulthood,” the researchers noted.
Don’t tread lightly just because they are young
“The fact that these youth are accumulating complications at a rapid rate and are broadly affected early in adulthood certainly suggests that aggressive therapy is needed, both for glycemic control and treatment of risk factors like hypertension and dyslipidemia,” study coauthor Philip S. Zeitler, MD, PhD, said in an interview.
“In the absence of studies specifically addressing this, we need to take a more aggressive approach than people might be inclined to, given that the age at diagnosis is young, around 14 years,” he added.
“Contrary to the inclination to be ‘gentle’ in treating them because they are kids, these data suggest that we can’t let these initial years go by without strong intervention, and we need to be prepared for polypharmacy.”
Unfortunately, as Dr. Zeitler and coauthors explained, youth-onset type 2 diabetes is characterized by a suboptimal response to currently approved diabetes medications.
New pediatric indications in the United States for drugs used to treat type 2 diabetes in adults, including the recent Food and Drug Administration approval of extended-release exenatide for children as young as 10 years of age, “helps, but only marginally,” said Dr. Zeitler, of the Clinical & Translational Research Center, Children’s Hospital Colorado, Aurora.
“In some cases, it will help get them covered by carriers, which is always good. But this is still a very limited set of medications. It doesn’t include more recently approved more potent glucagon-like peptide-1 (GLP-1) agonists, like semaglutide, and doesn’t include the sodium-glucose cotransporter 2 (SGLT2) inhibitors. Pediatricians are used to using medications off label and that is necessary here while we await further approvals,” he said.
And he noted that most individuals with youth-onset type 2 diabetes in the United States are covered by public insurance or are uninsured, depending on which state they live in. While the two major Medicaid programs in Colorado allow full access to adult formularies, that’s not the case everywhere. Moreover, patients often face further access barriers in states without expanded Medicaid.
Follow-up shows all metrics worsening over time
In TODAY 2, patients participated in an observational follow-up in their usual care settings in 2011-2020. At the start, they were receiving metformin with or without insulin for diabetes, but whether this continued and whether they were treated for other risk factors was down to individual circumstances.
Participants’ median A1c increased over time, and the percentage with A1c < 6% (< 48 mmol/mol) declined from 75% at the time of TODAY entry to just 19% at the 15-year end of follow-up.
The proportion with an A1c ≤ 10% (≤ 86 mmol/mol) rose from 0% at baseline to 34% at 15 years.
At that time, nearly 50% were taking both metformin and insulin, while more than a quarter were taking no medications.
The prevalence of hypertension increased from 19.2% at baseline to 67.5% at 15 years, while dyslipidemia rose from 20.8% to 51.6%.
Kidney disease prevalence increased from 8.0% at baseline to 54.8% at 15 years. Nerve disease rose from 1.0% to 32.4%. Retinal disease jumped from 13.7% with milder nonproliferative retinopathy in 2010-2011 to 51.0% with any eye disease in 2017-2018, including 8.8% with moderate to severe retinal changes and 3.5% with macular edema.
Overall, at the time of the last visit, 39.9% had no diabetes complications, 31.8% had one, 21.3% had two, and 7.1% had three complications.
Serious cardiovascular events in mid-20s
There were 17 adjudicated serious cardiovascular events, including four myocardial infarctions, six heart failure events, three diagnoses of coronary artery disease, and four strokes.
Six participants died, one each from myocardial infarction, kidney failure, and drug overdose, and three from sepsis.
Dr. Zeitler called the macrovascular events “shocking,” noting that although the numbers are small, for people in their mid-20s “they should be zero ... While we don’t yet know if the rates are the same or faster than in adults, even if they are the same, these kids are only in their late 20s, as opposed to adults experiencing these problems in their 50s, 60s, and 70s.
“The fact that these complications are occurring when these individuals should be in the prime of their life for both family and work has huge implications,” he stressed.
Findings have multiple causes
The reasons for the findings are both biologic and socioeconomic, Dr. Zeitler said.
“We know already that many kids with type 2 have rapid [deterioration of] beta-cell [function], which is probably very biologic. It stands to reason that an individual who can get diabetes as an adolescent probably has more fragile beta cells in some way,” he noted.
“But we also know that many other things contribute: stress, social determinants, access to quality care and medications, access to healthy foods and physical activity, availability of family supervision given the realities of families’ economic status and jobs, etc.”
It’s also known that youth with type 2 diabetes have much more severe insulin resistance than that of adults with the condition, and that “once the kids left ... the [TODAY] study, risk factor treatment in the community was less than ideal, and a lot of kids who met criteria for treatment of their blood pressure or lipids were not being treated. This is likely at least partly sociologic and partly the general pediatric hesitancy to use medications.”
He said the TODAY team will soon have some new data to show that “glycemia during the early years makes a difference, again supporting intensive intervention early on.”
The TODAY study was supported by grants from the National Institute of Diabetes and Digestive and Kidney Diseases. Dr. Zeitler had no further disclosures.
A version of this article first appeared on Medscape.com.
Newly published data show alarmingly high rates and severity of early diabetes-specific complications in individuals who develop type 2 diabetes at a young age. This suggests intervention should be early and aggressive among these youngsters, said the researchers.
The results for the 500 young adult participants in the Treatment Options for Type 2 Diabetes in Adolescents and Youth 2 (TODAY 2) study were published online July 29 in the New England Journal of Medicine by the TODAY study group.
At follow-up – after originally participating in the TODAY trial when they were young teenagers – they had a mean age of 26.4 years.
At this time, more than two thirds had hypertension and half had dyslipidemia.
Overall, 60% had at least one diabetic microvascular complication (retinal disease, neuropathy, or diabetic kidney disease), and more than a quarter had two or more such complications.
“These data illustrate the serious personal and public health consequences of youth-onset type 2 diabetes in the transition to adulthood,” the researchers noted.
Don’t tread lightly just because they are young
“The fact that these youth are accumulating complications at a rapid rate and are broadly affected early in adulthood certainly suggests that aggressive therapy is needed, both for glycemic control and treatment of risk factors like hypertension and dyslipidemia,” study coauthor Philip S. Zeitler, MD, PhD, said in an interview.
“In the absence of studies specifically addressing this, we need to take a more aggressive approach than people might be inclined to, given that the age at diagnosis is young, around 14 years,” he added.
“Contrary to the inclination to be ‘gentle’ in treating them because they are kids, these data suggest that we can’t let these initial years go by without strong intervention, and we need to be prepared for polypharmacy.”
Unfortunately, as Dr. Zeitler and coauthors explained, youth-onset type 2 diabetes is characterized by a suboptimal response to currently approved diabetes medications.
New pediatric indications in the United States for drugs used to treat type 2 diabetes in adults, including the recent Food and Drug Administration approval of extended-release exenatide for children as young as 10 years of age, “helps, but only marginally,” said Dr. Zeitler, of the Clinical & Translational Research Center, Children’s Hospital Colorado, Aurora.
“In some cases, it will help get them covered by carriers, which is always good. But this is still a very limited set of medications. It doesn’t include more recently approved more potent glucagon-like peptide-1 (GLP-1) agonists, like semaglutide, and doesn’t include the sodium-glucose cotransporter 2 (SGLT2) inhibitors. Pediatricians are used to using medications off label and that is necessary here while we await further approvals,” he said.
And he noted that most individuals with youth-onset type 2 diabetes in the United States are covered by public insurance or are uninsured, depending on which state they live in. While the two major Medicaid programs in Colorado allow full access to adult formularies, that’s not the case everywhere. Moreover, patients often face further access barriers in states without expanded Medicaid.
Follow-up shows all metrics worsening over time
In TODAY 2, patients participated in an observational follow-up in their usual care settings in 2011-2020. At the start, they were receiving metformin with or without insulin for diabetes, but whether this continued and whether they were treated for other risk factors was down to individual circumstances.
Participants’ median A1c increased over time, and the percentage with A1c < 6% (< 48 mmol/mol) declined from 75% at the time of TODAY entry to just 19% at the 15-year end of follow-up.
The proportion with an A1c ≤ 10% (≤ 86 mmol/mol) rose from 0% at baseline to 34% at 15 years.
At that time, nearly 50% were taking both metformin and insulin, while more than a quarter were taking no medications.
The prevalence of hypertension increased from 19.2% at baseline to 67.5% at 15 years, while dyslipidemia rose from 20.8% to 51.6%.
Kidney disease prevalence increased from 8.0% at baseline to 54.8% at 15 years. Nerve disease rose from 1.0% to 32.4%. Retinal disease jumped from 13.7% with milder nonproliferative retinopathy in 2010-2011 to 51.0% with any eye disease in 2017-2018, including 8.8% with moderate to severe retinal changes and 3.5% with macular edema.
Overall, at the time of the last visit, 39.9% had no diabetes complications, 31.8% had one, 21.3% had two, and 7.1% had three complications.
Serious cardiovascular events in mid-20s
There were 17 adjudicated serious cardiovascular events, including four myocardial infarctions, six heart failure events, three diagnoses of coronary artery disease, and four strokes.
Six participants died, one each from myocardial infarction, kidney failure, and drug overdose, and three from sepsis.
Dr. Zeitler called the macrovascular events “shocking,” noting that although the numbers are small, for people in their mid-20s “they should be zero ... While we don’t yet know if the rates are the same or faster than in adults, even if they are the same, these kids are only in their late 20s, as opposed to adults experiencing these problems in their 50s, 60s, and 70s.
“The fact that these complications are occurring when these individuals should be in the prime of their life for both family and work has huge implications,” he stressed.
Findings have multiple causes
The reasons for the findings are both biologic and socioeconomic, Dr. Zeitler said.
“We know already that many kids with type 2 have rapid [deterioration of] beta-cell [function], which is probably very biologic. It stands to reason that an individual who can get diabetes as an adolescent probably has more fragile beta cells in some way,” he noted.
“But we also know that many other things contribute: stress, social determinants, access to quality care and medications, access to healthy foods and physical activity, availability of family supervision given the realities of families’ economic status and jobs, etc.”
It’s also known that youth with type 2 diabetes have much more severe insulin resistance than that of adults with the condition, and that “once the kids left ... the [TODAY] study, risk factor treatment in the community was less than ideal, and a lot of kids who met criteria for treatment of their blood pressure or lipids were not being treated. This is likely at least partly sociologic and partly the general pediatric hesitancy to use medications.”
He said the TODAY team will soon have some new data to show that “glycemia during the early years makes a difference, again supporting intensive intervention early on.”
The TODAY study was supported by grants from the National Institute of Diabetes and Digestive and Kidney Diseases. Dr. Zeitler had no further disclosures.
A version of this article first appeared on Medscape.com.
ESC heart failure guideline to integrate bounty of new meds
Today there are so many evidence-based drug therapies for heart failure with reduced ejection fraction (HFrEF) that physicians treating HF patients almost don’t know what to do them.
It’s an exciting new age that way, but to many vexingly unclear how best to merge the shiny new options with mainstay regimens based on time-honored renin-angiotensin system (RAS) inhibitors and beta-blockers.
To impart some clarity, the authors of a new HF guideline document recently took center stage at the Heart Failure Association of the European Society of Cardiology (ESC-HFA) annual meeting to preview their updated recommendations, with novel twists based on recent major trials, for the new age of HF pharmacotherapeutics.
The guideline committee considered the evidence base that existed “up until the end of March of this year,” Theresa A. McDonagh, MD, King’s College London, said during the presentation. The document “is now finalized, it’s with the publishers, and it will be presented in full with simultaneous publication at the ESC meeting” that starts August 27.
It describes a game plan, already followed by some clinicians in practice without official guidance, for initiating drugs from each of four classes in virtually all patients with HFrEF.
New indicated drugs, new perspective for HFrEF
Three of the drug categories are old acquaintances. Among them are the RAS inhibitors, which include angiotensin-receptor/neprilysin inhibitors, beta-blockers, and the mineralocorticoid receptor antagonists. The latter drugs are gaining new respect after having been underplayed in HF prescribing despite longstanding evidence of efficacy.
Completing the quartet of first-line HFrEF drug classes is a recent arrival to the HF arena, the sodium-glucose cotransporter 2 inhibitors.
“We now have new data and a simplified treatment algorithm for heart failure with reduced ejection fraction based on the early administration of the four major classes of drugs,” said Marco Metra, MD, University of Brescia (Italy), previewing the medical-therapy portions of the new guideline at the ESC-HFA sessions, which launched virtually and live in Florence, Italy, on July 29.
The new game plan offers a simple answer to a once-common but complex question: How and in what order are the different drug classes initiated in patients with HFrEF? In the new document, the stated goal is to get them all on board expeditiously and safely, by any means possible.
The guideline writers did not specify a sequence, preferring to leave that decision to physicians, said Dr. Metra, who stated only two guiding principles. The first is to consider the patient’s unique circumstances. The order in which the drugs are introduced might vary, depending on, for example, whether the patient has low or high blood pressure or renal dysfunction.
Second, “it is very important that we try to give all four classes of drugs to the patient in the shortest time possible, because this saves lives,” he said.
That there is no recommendation on sequencing the drugs has led some to the wrong interpretation that all should be started at once, observed coauthor Javed Butler, MD, MPH, University of Mississippi, Jackson, as a panelist during the presentation. Far from it, he said. “The doctor with the patient in front of you can make the best decision. The idea here is to get all the therapies on as soon as possible, as safely as possible.”
“The order in which they are introduced is not really important,” agreed Vijay Chopra, MD, Max Super Specialty Hospital Saket, New Delhi, another coauthor on the panel. “The important thing is that at least some dose of all the four drugs needs to be introduced in the first 4-6 weeks, and then up-titrated.”
Other medical therapy can be more tailored, Dr. Metra noted, such as loop diuretics for patients with congestion, iron for those with iron deficiency, and other drugs depending on whether there is, for example, atrial fibrillation or coronary disease.
Adoption of emerging definitions
The document adopts the emerging characterization of HFrEF by a left ventricular ejection fraction (LVEF) up to 40%.
And it will leverage an expanding evidence base for medication in a segment of patients once said to have HF with preserved ejection fraction (HFpEF), who had therefore lacked specific, guideline-directed medical therapies. Now, patients with an LVEF of 41%-49% will be said to have HF with mildly reduced ejection fraction (HFmrEF), a tweak to the recently introduced HF with “mid-range” LVEF that is designed to assert its nature as something to treat. The new document’s HFmrEF recommendations come with various class and level-of-evidence ratings.
That leaves HFpEF to be characterized by an LVEF of 50% in combination with structural or functional abnormalities associated with LV diastolic dysfunction or raised LV filling pressures, including raised natriuretic peptide levels.
The definitions are consistent with those proposed internationally by the ESC-HFA, the Heart Failure Society of America, and other groups in a statement published in March.
Expanded HFrEF med landscape
Since the 2016 ESC guideline on HF therapy, Dr. McDonagh said, “there’s been no substantial change in the evidence for many of the classical drugs that we use in heart failure. However, we had a lot of new and exciting evidence to consider,” especially in support of the SGLT2 inhibitors as one of the core medications in HFrEF.
The new data came from two controlled trials in particular. In DAPA-HF, patients with HFrEF who were initially without diabetes and who went on dapagliflozin (Farxiga, AstraZeneca) showed a 27% drop in cardiovascular (CV) death or worsening-HF events over a median of 18 months.
“That was followed up with very concordant results with empagliflozin [Jardiance, Boehringer Ingelheim/Eli Lilly] in HFrEF in the EMPEROR-Reduced trial,” Dr. McDonagh said. In that trial, comparable patients who took empagliflozin showed a 25% drop in a primary endpoint similar to that in DAPA-HF over the median 16-month follow-up.
Other HFrEF recommendations are for selected patients. They include ivabradine, already in the guidelines, for patients in sinus rhythm with an elevated resting heart rate who can’t take beta-blockers for whatever reason. But, Dr. McDonagh noted, “we had some new classes of drugs to consider as well.”
In particular, the oral soluble guanylate-cyclase receptor stimulator vericiguat (Verquvo) emerged about a year ago from the VICTORIA trial as a modest success for patients with HFrEF and a previous HF hospitalization. In the trial with more than 5,000 patients, treatment with vericiguat atop standard drug and device therapy was followed by a significant 10% drop in risk for CV death or HF hospitalization.
Available now or likely to be available in the United States, the European Union, Japan, and other countries, vericiguat is recommended in the new guideline for VICTORIA-like patients who don’t adequately respond to other indicated medications.
Little for HFpEF as newly defined
“Almost nothing is new” in the guidelines for HFpEF, Dr. Metra said. The document recommends screening for and treatment of any underlying disorder and comorbidities, plus diuretics for any congestion. “That’s what we have to date.”
But that evidence base might soon change. The new HFpEF recommendations could possibly be up-staged at the ESC sessions by the August 27 scheduled presentation of EMPEROR-Preserved, a randomized test of empagliflozin in HFpEF and – it could be said – HFmrEF. The trial entered patients with chronic HF and an LVEF greater than 40%.
Eli Lilly and Boehringer Ingelheim offered the world a peek at the results, which suggest the SGLT2 inhibitor had a positive impact on the primary endpoint of CV death or HF hospitalization. They announced the cursory top-line outcomes in early July as part of its regulatory obligations, noting that the trial had “met” its primary endpoint.
But many unknowns remain, including the degree of benefit and whether it varied among subgroups, and especially whether outcomes were different for HFmrEF than for HFpEF.
Upgrades for familiar agents
Still, HFmrEF gets noteworthy attention in the document. “For the first time, we have recommendations for these patients,” Dr. Metra said. “We already knew that diuretics are indicated for the treatment of congestion. But now, ACE inhibitors, ARBs, beta-blockers, mineralocorticoid antagonists, as well as sacubitril/valsartan, may be considered to improve outcomes in these patients.” Their upgrades in the new guidelines were based on review of trials in the CHARM program and of TOPCAT and PARAGON-HF, among others, he said.

The new document also includes “treatment algorithms based on phenotypes”; that is, comorbidities and less common HF precipitants. For example, “assessment of iron status is now mandated in all patients with heart failure,” Dr. Metra said.
AFFIRM-HF is the key trial in this arena, with its more than 1,100 iron-deficient patients with LVEF less than 50% who had been recently hospitalized for HF. A year of treatment with ferric carboxymaltose (Ferinject/Injectafer, Vifor) led to a 26% drop in risk for HF hospitalization, but without affecting mortality.
For those who are iron deficient, Dr. Metra said, “ferric carboxymaltose intravenously should be considered not only in patients with low ejection fraction and outpatients, but also in patients recently hospitalized for acute heart failure.”
The SGLT2 inhibitors are recommended in HFrEF patients with type 2 diabetes. And treatment with tafamidis (Vyndaqel, Pfizer) in patients with genetic or wild-type transthyretin cardiac amyloidosis gets a class I recommendation based on survival gains seen in the ATTR-ACT trial.
Also recommended is a full CV assessment for patients with cancer who are on cardiotoxic agents or otherwise might be at risk for chemotherapy cardiotoxicity. “Beta-blockers and ACE inhibitors should be considered in those who develop left ventricular systolic dysfunction after anticancer therapy,” Dr. Metra said.
The ongoing pandemic made its mark on the document’s genesis, as it has with most everything else. “For better or worse, we were a ‘COVID guideline,’ ” Dr. McDonagh said. The writing committee consisted of “a large task force of 31 individuals, including two patients,” and there were “only two face-to-face meetings prior to the first wave of COVID hitting Europe.”
The committee voted on each of the recommendations, “and we had to have agreement of more than 75% of the task force to assign a class of recommendation or level of evidence,” she said. “I think we did the best we could in the circumstances. We had the benefit of many discussions over Zoom, and I think at the end of the day we have achieved a consensus.”
With such a large body of participants and the 75% threshold for agreement, “you end up with perhaps a conservative guideline. But that’s not a bad thing for clinical practice, for guidelines to be conservative,” Dr. McDonagh said. “They’re mainly concerned with looking at evidence and safety.”
A version of this article first appeared on Medscape.com.
Today there are so many evidence-based drug therapies for heart failure with reduced ejection fraction (HFrEF) that physicians treating HF patients almost don’t know what to do them.
It’s an exciting new age that way, but to many vexingly unclear how best to merge the shiny new options with mainstay regimens based on time-honored renin-angiotensin system (RAS) inhibitors and beta-blockers.
To impart some clarity, the authors of a new HF guideline document recently took center stage at the Heart Failure Association of the European Society of Cardiology (ESC-HFA) annual meeting to preview their updated recommendations, with novel twists based on recent major trials, for the new age of HF pharmacotherapeutics.
The guideline committee considered the evidence base that existed “up until the end of March of this year,” Theresa A. McDonagh, MD, King’s College London, said during the presentation. The document “is now finalized, it’s with the publishers, and it will be presented in full with simultaneous publication at the ESC meeting” that starts August 27.
It describes a game plan, already followed by some clinicians in practice without official guidance, for initiating drugs from each of four classes in virtually all patients with HFrEF.
New indicated drugs, new perspective for HFrEF
Three of the drug categories are old acquaintances. Among them are the RAS inhibitors, which include angiotensin-receptor/neprilysin inhibitors, beta-blockers, and the mineralocorticoid receptor antagonists. The latter drugs are gaining new respect after having been underplayed in HF prescribing despite longstanding evidence of efficacy.
Completing the quartet of first-line HFrEF drug classes is a recent arrival to the HF arena, the sodium-glucose cotransporter 2 inhibitors.
“We now have new data and a simplified treatment algorithm for heart failure with reduced ejection fraction based on the early administration of the four major classes of drugs,” said Marco Metra, MD, University of Brescia (Italy), previewing the medical-therapy portions of the new guideline at the ESC-HFA sessions, which launched virtually and live in Florence, Italy, on July 29.
The new game plan offers a simple answer to a once-common but complex question: How and in what order are the different drug classes initiated in patients with HFrEF? In the new document, the stated goal is to get them all on board expeditiously and safely, by any means possible.
The guideline writers did not specify a sequence, preferring to leave that decision to physicians, said Dr. Metra, who stated only two guiding principles. The first is to consider the patient’s unique circumstances. The order in which the drugs are introduced might vary, depending on, for example, whether the patient has low or high blood pressure or renal dysfunction.
Second, “it is very important that we try to give all four classes of drugs to the patient in the shortest time possible, because this saves lives,” he said.
That there is no recommendation on sequencing the drugs has led some to the wrong interpretation that all should be started at once, observed coauthor Javed Butler, MD, MPH, University of Mississippi, Jackson, as a panelist during the presentation. Far from it, he said. “The doctor with the patient in front of you can make the best decision. The idea here is to get all the therapies on as soon as possible, as safely as possible.”
“The order in which they are introduced is not really important,” agreed Vijay Chopra, MD, Max Super Specialty Hospital Saket, New Delhi, another coauthor on the panel. “The important thing is that at least some dose of all the four drugs needs to be introduced in the first 4-6 weeks, and then up-titrated.”
Other medical therapy can be more tailored, Dr. Metra noted, such as loop diuretics for patients with congestion, iron for those with iron deficiency, and other drugs depending on whether there is, for example, atrial fibrillation or coronary disease.
Adoption of emerging definitions
The document adopts the emerging characterization of HFrEF by a left ventricular ejection fraction (LVEF) up to 40%.
And it will leverage an expanding evidence base for medication in a segment of patients once said to have HF with preserved ejection fraction (HFpEF), who had therefore lacked specific, guideline-directed medical therapies. Now, patients with an LVEF of 41%-49% will be said to have HF with mildly reduced ejection fraction (HFmrEF), a tweak to the recently introduced HF with “mid-range” LVEF that is designed to assert its nature as something to treat. The new document’s HFmrEF recommendations come with various class and level-of-evidence ratings.
That leaves HFpEF to be characterized by an LVEF of 50% in combination with structural or functional abnormalities associated with LV diastolic dysfunction or raised LV filling pressures, including raised natriuretic peptide levels.
The definitions are consistent with those proposed internationally by the ESC-HFA, the Heart Failure Society of America, and other groups in a statement published in March.
Expanded HFrEF med landscape
Since the 2016 ESC guideline on HF therapy, Dr. McDonagh said, “there’s been no substantial change in the evidence for many of the classical drugs that we use in heart failure. However, we had a lot of new and exciting evidence to consider,” especially in support of the SGLT2 inhibitors as one of the core medications in HFrEF.
The new data came from two controlled trials in particular. In DAPA-HF, patients with HFrEF who were initially without diabetes and who went on dapagliflozin (Farxiga, AstraZeneca) showed a 27% drop in cardiovascular (CV) death or worsening-HF events over a median of 18 months.
“That was followed up with very concordant results with empagliflozin [Jardiance, Boehringer Ingelheim/Eli Lilly] in HFrEF in the EMPEROR-Reduced trial,” Dr. McDonagh said. In that trial, comparable patients who took empagliflozin showed a 25% drop in a primary endpoint similar to that in DAPA-HF over the median 16-month follow-up.
Other HFrEF recommendations are for selected patients. They include ivabradine, already in the guidelines, for patients in sinus rhythm with an elevated resting heart rate who can’t take beta-blockers for whatever reason. But, Dr. McDonagh noted, “we had some new classes of drugs to consider as well.”
In particular, the oral soluble guanylate-cyclase receptor stimulator vericiguat (Verquvo) emerged about a year ago from the VICTORIA trial as a modest success for patients with HFrEF and a previous HF hospitalization. In the trial with more than 5,000 patients, treatment with vericiguat atop standard drug and device therapy was followed by a significant 10% drop in risk for CV death or HF hospitalization.
Available now or likely to be available in the United States, the European Union, Japan, and other countries, vericiguat is recommended in the new guideline for VICTORIA-like patients who don’t adequately respond to other indicated medications.
Little for HFpEF as newly defined
“Almost nothing is new” in the guidelines for HFpEF, Dr. Metra said. The document recommends screening for and treatment of any underlying disorder and comorbidities, plus diuretics for any congestion. “That’s what we have to date.”
But that evidence base might soon change. The new HFpEF recommendations could possibly be up-staged at the ESC sessions by the August 27 scheduled presentation of EMPEROR-Preserved, a randomized test of empagliflozin in HFpEF and – it could be said – HFmrEF. The trial entered patients with chronic HF and an LVEF greater than 40%.
Eli Lilly and Boehringer Ingelheim offered the world a peek at the results, which suggest the SGLT2 inhibitor had a positive impact on the primary endpoint of CV death or HF hospitalization. They announced the cursory top-line outcomes in early July as part of its regulatory obligations, noting that the trial had “met” its primary endpoint.
But many unknowns remain, including the degree of benefit and whether it varied among subgroups, and especially whether outcomes were different for HFmrEF than for HFpEF.
Upgrades for familiar agents
Still, HFmrEF gets noteworthy attention in the document. “For the first time, we have recommendations for these patients,” Dr. Metra said. “We already knew that diuretics are indicated for the treatment of congestion. But now, ACE inhibitors, ARBs, beta-blockers, mineralocorticoid antagonists, as well as sacubitril/valsartan, may be considered to improve outcomes in these patients.” Their upgrades in the new guidelines were based on review of trials in the CHARM program and of TOPCAT and PARAGON-HF, among others, he said.

The new document also includes “treatment algorithms based on phenotypes”; that is, comorbidities and less common HF precipitants. For example, “assessment of iron status is now mandated in all patients with heart failure,” Dr. Metra said.
AFFIRM-HF is the key trial in this arena, with its more than 1,100 iron-deficient patients with LVEF less than 50% who had been recently hospitalized for HF. A year of treatment with ferric carboxymaltose (Ferinject/Injectafer, Vifor) led to a 26% drop in risk for HF hospitalization, but without affecting mortality.
For those who are iron deficient, Dr. Metra said, “ferric carboxymaltose intravenously should be considered not only in patients with low ejection fraction and outpatients, but also in patients recently hospitalized for acute heart failure.”
The SGLT2 inhibitors are recommended in HFrEF patients with type 2 diabetes. And treatment with tafamidis (Vyndaqel, Pfizer) in patients with genetic or wild-type transthyretin cardiac amyloidosis gets a class I recommendation based on survival gains seen in the ATTR-ACT trial.
Also recommended is a full CV assessment for patients with cancer who are on cardiotoxic agents or otherwise might be at risk for chemotherapy cardiotoxicity. “Beta-blockers and ACE inhibitors should be considered in those who develop left ventricular systolic dysfunction after anticancer therapy,” Dr. Metra said.
The ongoing pandemic made its mark on the document’s genesis, as it has with most everything else. “For better or worse, we were a ‘COVID guideline,’ ” Dr. McDonagh said. The writing committee consisted of “a large task force of 31 individuals, including two patients,” and there were “only two face-to-face meetings prior to the first wave of COVID hitting Europe.”
The committee voted on each of the recommendations, “and we had to have agreement of more than 75% of the task force to assign a class of recommendation or level of evidence,” she said. “I think we did the best we could in the circumstances. We had the benefit of many discussions over Zoom, and I think at the end of the day we have achieved a consensus.”
With such a large body of participants and the 75% threshold for agreement, “you end up with perhaps a conservative guideline. But that’s not a bad thing for clinical practice, for guidelines to be conservative,” Dr. McDonagh said. “They’re mainly concerned with looking at evidence and safety.”
A version of this article first appeared on Medscape.com.
Today there are so many evidence-based drug therapies for heart failure with reduced ejection fraction (HFrEF) that physicians treating HF patients almost don’t know what to do them.
It’s an exciting new age that way, but to many vexingly unclear how best to merge the shiny new options with mainstay regimens based on time-honored renin-angiotensin system (RAS) inhibitors and beta-blockers.
To impart some clarity, the authors of a new HF guideline document recently took center stage at the Heart Failure Association of the European Society of Cardiology (ESC-HFA) annual meeting to preview their updated recommendations, with novel twists based on recent major trials, for the new age of HF pharmacotherapeutics.
The guideline committee considered the evidence base that existed “up until the end of March of this year,” Theresa A. McDonagh, MD, King’s College London, said during the presentation. The document “is now finalized, it’s with the publishers, and it will be presented in full with simultaneous publication at the ESC meeting” that starts August 27.
It describes a game plan, already followed by some clinicians in practice without official guidance, for initiating drugs from each of four classes in virtually all patients with HFrEF.
New indicated drugs, new perspective for HFrEF
Three of the drug categories are old acquaintances. Among them are the RAS inhibitors, which include angiotensin-receptor/neprilysin inhibitors, beta-blockers, and the mineralocorticoid receptor antagonists. The latter drugs are gaining new respect after having been underplayed in HF prescribing despite longstanding evidence of efficacy.
Completing the quartet of first-line HFrEF drug classes is a recent arrival to the HF arena, the sodium-glucose cotransporter 2 inhibitors.
“We now have new data and a simplified treatment algorithm for heart failure with reduced ejection fraction based on the early administration of the four major classes of drugs,” said Marco Metra, MD, University of Brescia (Italy), previewing the medical-therapy portions of the new guideline at the ESC-HFA sessions, which launched virtually and live in Florence, Italy, on July 29.
The new game plan offers a simple answer to a once-common but complex question: How and in what order are the different drug classes initiated in patients with HFrEF? In the new document, the stated goal is to get them all on board expeditiously and safely, by any means possible.
The guideline writers did not specify a sequence, preferring to leave that decision to physicians, said Dr. Metra, who stated only two guiding principles. The first is to consider the patient’s unique circumstances. The order in which the drugs are introduced might vary, depending on, for example, whether the patient has low or high blood pressure or renal dysfunction.
Second, “it is very important that we try to give all four classes of drugs to the patient in the shortest time possible, because this saves lives,” he said.
That there is no recommendation on sequencing the drugs has led some to the wrong interpretation that all should be started at once, observed coauthor Javed Butler, MD, MPH, University of Mississippi, Jackson, as a panelist during the presentation. Far from it, he said. “The doctor with the patient in front of you can make the best decision. The idea here is to get all the therapies on as soon as possible, as safely as possible.”
“The order in which they are introduced is not really important,” agreed Vijay Chopra, MD, Max Super Specialty Hospital Saket, New Delhi, another coauthor on the panel. “The important thing is that at least some dose of all the four drugs needs to be introduced in the first 4-6 weeks, and then up-titrated.”
Other medical therapy can be more tailored, Dr. Metra noted, such as loop diuretics for patients with congestion, iron for those with iron deficiency, and other drugs depending on whether there is, for example, atrial fibrillation or coronary disease.
Adoption of emerging definitions
The document adopts the emerging characterization of HFrEF by a left ventricular ejection fraction (LVEF) up to 40%.
And it will leverage an expanding evidence base for medication in a segment of patients once said to have HF with preserved ejection fraction (HFpEF), who had therefore lacked specific, guideline-directed medical therapies. Now, patients with an LVEF of 41%-49% will be said to have HF with mildly reduced ejection fraction (HFmrEF), a tweak to the recently introduced HF with “mid-range” LVEF that is designed to assert its nature as something to treat. The new document’s HFmrEF recommendations come with various class and level-of-evidence ratings.
That leaves HFpEF to be characterized by an LVEF of 50% in combination with structural or functional abnormalities associated with LV diastolic dysfunction or raised LV filling pressures, including raised natriuretic peptide levels.
The definitions are consistent with those proposed internationally by the ESC-HFA, the Heart Failure Society of America, and other groups in a statement published in March.
Expanded HFrEF med landscape
Since the 2016 ESC guideline on HF therapy, Dr. McDonagh said, “there’s been no substantial change in the evidence for many of the classical drugs that we use in heart failure. However, we had a lot of new and exciting evidence to consider,” especially in support of the SGLT2 inhibitors as one of the core medications in HFrEF.
The new data came from two controlled trials in particular. In DAPA-HF, patients with HFrEF who were initially without diabetes and who went on dapagliflozin (Farxiga, AstraZeneca) showed a 27% drop in cardiovascular (CV) death or worsening-HF events over a median of 18 months.
“That was followed up with very concordant results with empagliflozin [Jardiance, Boehringer Ingelheim/Eli Lilly] in HFrEF in the EMPEROR-Reduced trial,” Dr. McDonagh said. In that trial, comparable patients who took empagliflozin showed a 25% drop in a primary endpoint similar to that in DAPA-HF over the median 16-month follow-up.
Other HFrEF recommendations are for selected patients. They include ivabradine, already in the guidelines, for patients in sinus rhythm with an elevated resting heart rate who can’t take beta-blockers for whatever reason. But, Dr. McDonagh noted, “we had some new classes of drugs to consider as well.”
In particular, the oral soluble guanylate-cyclase receptor stimulator vericiguat (Verquvo) emerged about a year ago from the VICTORIA trial as a modest success for patients with HFrEF and a previous HF hospitalization. In the trial with more than 5,000 patients, treatment with vericiguat atop standard drug and device therapy was followed by a significant 10% drop in risk for CV death or HF hospitalization.
Available now or likely to be available in the United States, the European Union, Japan, and other countries, vericiguat is recommended in the new guideline for VICTORIA-like patients who don’t adequately respond to other indicated medications.
Little for HFpEF as newly defined
“Almost nothing is new” in the guidelines for HFpEF, Dr. Metra said. The document recommends screening for and treatment of any underlying disorder and comorbidities, plus diuretics for any congestion. “That’s what we have to date.”
But that evidence base might soon change. The new HFpEF recommendations could possibly be up-staged at the ESC sessions by the August 27 scheduled presentation of EMPEROR-Preserved, a randomized test of empagliflozin in HFpEF and – it could be said – HFmrEF. The trial entered patients with chronic HF and an LVEF greater than 40%.
Eli Lilly and Boehringer Ingelheim offered the world a peek at the results, which suggest the SGLT2 inhibitor had a positive impact on the primary endpoint of CV death or HF hospitalization. They announced the cursory top-line outcomes in early July as part of its regulatory obligations, noting that the trial had “met” its primary endpoint.
But many unknowns remain, including the degree of benefit and whether it varied among subgroups, and especially whether outcomes were different for HFmrEF than for HFpEF.
Upgrades for familiar agents
Still, HFmrEF gets noteworthy attention in the document. “For the first time, we have recommendations for these patients,” Dr. Metra said. “We already knew that diuretics are indicated for the treatment of congestion. But now, ACE inhibitors, ARBs, beta-blockers, mineralocorticoid antagonists, as well as sacubitril/valsartan, may be considered to improve outcomes in these patients.” Their upgrades in the new guidelines were based on review of trials in the CHARM program and of TOPCAT and PARAGON-HF, among others, he said.

The new document also includes “treatment algorithms based on phenotypes”; that is, comorbidities and less common HF precipitants. For example, “assessment of iron status is now mandated in all patients with heart failure,” Dr. Metra said.
AFFIRM-HF is the key trial in this arena, with its more than 1,100 iron-deficient patients with LVEF less than 50% who had been recently hospitalized for HF. A year of treatment with ferric carboxymaltose (Ferinject/Injectafer, Vifor) led to a 26% drop in risk for HF hospitalization, but without affecting mortality.
For those who are iron deficient, Dr. Metra said, “ferric carboxymaltose intravenously should be considered not only in patients with low ejection fraction and outpatients, but also in patients recently hospitalized for acute heart failure.”
The SGLT2 inhibitors are recommended in HFrEF patients with type 2 diabetes. And treatment with tafamidis (Vyndaqel, Pfizer) in patients with genetic or wild-type transthyretin cardiac amyloidosis gets a class I recommendation based on survival gains seen in the ATTR-ACT trial.
Also recommended is a full CV assessment for patients with cancer who are on cardiotoxic agents or otherwise might be at risk for chemotherapy cardiotoxicity. “Beta-blockers and ACE inhibitors should be considered in those who develop left ventricular systolic dysfunction after anticancer therapy,” Dr. Metra said.
The ongoing pandemic made its mark on the document’s genesis, as it has with most everything else. “For better or worse, we were a ‘COVID guideline,’ ” Dr. McDonagh said. The writing committee consisted of “a large task force of 31 individuals, including two patients,” and there were “only two face-to-face meetings prior to the first wave of COVID hitting Europe.”
The committee voted on each of the recommendations, “and we had to have agreement of more than 75% of the task force to assign a class of recommendation or level of evidence,” she said. “I think we did the best we could in the circumstances. We had the benefit of many discussions over Zoom, and I think at the end of the day we have achieved a consensus.”
With such a large body of participants and the 75% threshold for agreement, “you end up with perhaps a conservative guideline. But that’s not a bad thing for clinical practice, for guidelines to be conservative,” Dr. McDonagh said. “They’re mainly concerned with looking at evidence and safety.”
A version of this article first appeared on Medscape.com.
FDA approves first interchangeable biosimilar insulin
The
The approval will allow Semglee to function like a generic drug in the market and may reduce insulin costs.
It is indicated to improve glycemic control in adults and pediatric patients with type 1 diabetes and in adults with type 2 diabetes.
Originally approved in June 2020 as a biosimilar to glargine, Semglee is now an “interchangeable biosimilar,” meaning that it has no clinically meaningful difference from the reference product and also may be substituted for that product – in this case, glargine (Lantus) – without prescriber intervention, just as generic drugs typically are, subject to state pharmacy laws.
For approval as an interchangeable biosimilar, manufacturers are required to provide additional data reflecting how the interchangeable biosimilar may be used in the marketplace with patients.
“Biosimilar and interchangeable biosimilar products have the potential to reduce health care costs, similar to how generic drugs have reduced costs. Biosimilars marketed in the U.S. typically have launched with initial list prices 15% to 35% lower than comparative list prices of the reference products,” the FDA said in a statement.
Semglee comes in 10-mL and 3-mL prefilled pens, and is administered subcutaneously once daily, with individualized doses. The most common side effects are hypoglycemia, edema, lipodystrophy, weight gain, and allergic reactions.
The FDA released new materials for health care providers regarding biosimilar and interchangeable biosimilar products, including a fact sheet about interchangeable biosimilar products.
A version of this article first appeared on Medscape.com.
The
The approval will allow Semglee to function like a generic drug in the market and may reduce insulin costs.
It is indicated to improve glycemic control in adults and pediatric patients with type 1 diabetes and in adults with type 2 diabetes.
Originally approved in June 2020 as a biosimilar to glargine, Semglee is now an “interchangeable biosimilar,” meaning that it has no clinically meaningful difference from the reference product and also may be substituted for that product – in this case, glargine (Lantus) – without prescriber intervention, just as generic drugs typically are, subject to state pharmacy laws.
For approval as an interchangeable biosimilar, manufacturers are required to provide additional data reflecting how the interchangeable biosimilar may be used in the marketplace with patients.
“Biosimilar and interchangeable biosimilar products have the potential to reduce health care costs, similar to how generic drugs have reduced costs. Biosimilars marketed in the U.S. typically have launched with initial list prices 15% to 35% lower than comparative list prices of the reference products,” the FDA said in a statement.
Semglee comes in 10-mL and 3-mL prefilled pens, and is administered subcutaneously once daily, with individualized doses. The most common side effects are hypoglycemia, edema, lipodystrophy, weight gain, and allergic reactions.
The FDA released new materials for health care providers regarding biosimilar and interchangeable biosimilar products, including a fact sheet about interchangeable biosimilar products.
A version of this article first appeared on Medscape.com.
The
The approval will allow Semglee to function like a generic drug in the market and may reduce insulin costs.
It is indicated to improve glycemic control in adults and pediatric patients with type 1 diabetes and in adults with type 2 diabetes.
Originally approved in June 2020 as a biosimilar to glargine, Semglee is now an “interchangeable biosimilar,” meaning that it has no clinically meaningful difference from the reference product and also may be substituted for that product – in this case, glargine (Lantus) – without prescriber intervention, just as generic drugs typically are, subject to state pharmacy laws.
For approval as an interchangeable biosimilar, manufacturers are required to provide additional data reflecting how the interchangeable biosimilar may be used in the marketplace with patients.
“Biosimilar and interchangeable biosimilar products have the potential to reduce health care costs, similar to how generic drugs have reduced costs. Biosimilars marketed in the U.S. typically have launched with initial list prices 15% to 35% lower than comparative list prices of the reference products,” the FDA said in a statement.
Semglee comes in 10-mL and 3-mL prefilled pens, and is administered subcutaneously once daily, with individualized doses. The most common side effects are hypoglycemia, edema, lipodystrophy, weight gain, and allergic reactions.
The FDA released new materials for health care providers regarding biosimilar and interchangeable biosimilar products, including a fact sheet about interchangeable biosimilar products.
A version of this article first appeared on Medscape.com.
PCPs lag on albuminuria tests in patients with type 2 diabetes
U.S. primary care physicians are not properly checking patients with type 2 diabetes for chronic kidney disease (CKD) nearly as often as they should, meaning many of these patients miss getting a timely diagnosis.
Inadequate measurement of urinary albumin-to-creatinine ratio (uACR) is the issue.
Review of data from more than half a million U.S. primary care patients with type 2 diabetes seen at any of 1,164 practice sites run by any of 24 health care organizations during 2016-2019 showed that barely more than half, 52%, had both their uACR and estimated glomerular filtration rate (eGFR) checked annually as recommended by several U.S. medical societies, and just 73% had both values checked during a 3-year period, Nikita Stempniewicz, MSc, and associates reported in Diabetes Care.
More detailed data showed that measurement of eGFR was reasonably robust, measured at a 90% rate annually and in 97% of patients at least once every 3 years. But recording uACR values lagged, with a 53% annual rate and a 74% rate of measurement at least once every 3 years, reported Mr. Stempniewicz, director of research and analytics for the American Medical Group Association, a trade association based in Alexandria, Va. The 24 health care organizations that supplied the study’s data are all members of this association.
Prevailing recommendations from various medical societies call for annual monitoring of urinary albumin in patients with type 2 diabetes and specify the uACR, such as in the Standards of Medical Care in Diabetes from the American Diabetes Association, as well as in recommendations promoted by the National Kidney Foundation.
Missing half the CKD patients with eGFR only
“Half the patients with type 2 diabetes and chronic kidney disease have elevated albuminuria without decreased eGFR and would not be detected with eGFR testing alone,” Mr. Stempniewicz noted in an interview.
“Many patients who present for nephrology care are incompletely assessed with only low eGFR but no urine testing. Missing albuminuria testing and uACR values means patients with high levels of albuminuria but normal kidney function go undetected and thus are not able to benefit from evidenced-based interventions, including nephrology services,” said Joseph A. Vassalotti, MD, a nephrologist, chief medical officer for the National Kidney Foundation, and a coauthor of the report.
Not testing patients with type 2 diabetes regularly for their uACR “is a missed opportunity to identify the highest-risk patients and treat them,” added Josef Coresh, MD, PhD, a professor of clinical epidemiology at Johns Hopkins University, Baltimore, and senior author on the study. Measurement of albuminuria is especially important for these patients because medications from the sodium-glucose cotransporter 2 inhibitor class have been proven to slow progression of CKD in patients with type 2 diabetes, but these drugs are expensive, and in some cases have labeling that specifies the presence of albuminuria.
“I have no doubt that improving albuminuria testing is a critical step to identify patients with diabetes at highest risk who should get the best treatment possible, including SGLT2 inhibitors,” Dr. Coresh said in an interview.
The new report is not the first to document inadequate assessment of albuminuria and uACR among primary care physicians (PCPs), but it came from the largest reported U.S. study to date. “eGFR is commonly collected in a routine laboratory blood panel, but collecting urine requires additional work flow,” noted Cara B. Litvin, MD, a general internal medicine researcher at the Medical University of South Carolina, Charleston, who has tested interventions aimed at boosting CKD assessment by PCPs and was not involved in the new study.
“There have also been conflicting guidelines,” such as a “now-inactive guideline from the American College of Physicians that recommended against routine urine albumin screening in patients with diabetes and already on treatment with an angiotensin converting enzyme inhibitor or an angiotensin receptor blocker,” she said.
New renal drugs change the stakes
The availability of newer drugs for slowing CKD progression such as the SGLT2 inhibitors will help trigger greater support for routine albuminuria testing, Dr. Litvin predicted in an interview. “Now that we have more medications that can reduce albuminuria and improve outcomes, I see screening for albuminuria increasing.” Finerenone (Kerendia) is another new agent from a new class that recently received Food and Drug Administration approval for treating CKD in patients with type 2 diabetes.
Other drivers of increased uACR testing she expects include revised clinical practice guidelines, and new quality measures of clinical care.
“Undertesting of albuminuria means that [nephrologists] have incomplete data to detect and completely risk stratify the CKD population. That in turn results in a reduced ability to match population health interventions to the severity of the condition or the risk stratification based on eGFR and uACR,” Dr. Vassalotti said in an interview.
“We are missing opportunities to prevent or delay kidney failure and reduce the risk of cardiovascular events and cardiovascular death in these patients, particularly now that we have a number of medications that offer kidney and cardiovascular protection such as SGLT2 inhibitors,” he added. “Leaders in nephrology are beginning to understand the consequences of undertesting, and are working to innovate to improve risk stratification, CKD detection, and apply interventions to give Americans living with CKD better outcomes.”
Strategies proven to boost albuminuria testing
Mr. Stempniewicz and coauthors cited in their report potential strategies for improving albuminuria testing, including benchmarking to identify best-performing sites for albumin testing within a health system and encouraging replication of identified best practices at lower-performing sites, and implementation of clinical-decision support tools in the EHR such as pop-up test reminders.
These were among the tools tested in two studies led by Dr. Litvin. One study, with results reported in 2016, involved 12 small U.S. primary care practices with a total of more than 30,000 patients and compared performance in a series of clinical quality measures at baseline with performance after 2 years of receiving various interventions designed to boost awareness for albuminuria testing.
The second study, with findings reported in 2019, involved 21 U.S. primary care practices that collectively cared for more than 100,000 patients and randomized the practices to either undergo interventions aimed at boosting testing awareness or to serve as controls.
Results from both studies showed significant and substantial increases in serial testing for albuminuria in patients with diabetes or hypertension when practices received the interventions.
“We showed that [using a] clinical-decision support tool, along with standing orders to automatically collect urine specimens, dramatically increased screening for urinary albumin in primary care practices,” Dr. Litvin said. “However, perhaps because of conflicting guidelines and clinical inertia there hasn’t been a major impetus for primary care practices in general to improve screening.” She hopes that will quickly change.
“As we have shown, adoption of EHR-based reminders along with standing orders can very quickly improve screening for albuminuria in primary care.”
Variation in testing rates among sites ‘tremendous’
One finding of the new study gives Mr. Stempniewicz hope for greater future testing: The large variance that the researchers saw in albuminuria testing rates within individual health systems.
“The paper shows that higher rates of testing are completely achievable within each system. Some clinics do very well, and the other units can learn from these local successes,” he said. At least half the organizations in the study had individual sites that fell into the top 10% for testing rates across all the greater than 1,000 sites included, and those same organizations also had at least one site that fell into the bottom 10% for testing.
“The variation is tremendous, and highlights an opportunity for improvement,” declared Mr. Stempniewicz.
“For routine testing, you need systems that help people. Clinicians shouldn’t have to think about doing routine testing. It should just happen,” said Dr. Coresh.
The study was funded in part by Janssen. Mr. Stempniewicz and Dr. Litvin had no disclosures. Dr. Coresh is an adviser to Healthy.io, a company that markets a home albuminuria testing kit to patients. Dr. Vassalotti has received personal fees from Renalytix.
U.S. primary care physicians are not properly checking patients with type 2 diabetes for chronic kidney disease (CKD) nearly as often as they should, meaning many of these patients miss getting a timely diagnosis.
Inadequate measurement of urinary albumin-to-creatinine ratio (uACR) is the issue.
Review of data from more than half a million U.S. primary care patients with type 2 diabetes seen at any of 1,164 practice sites run by any of 24 health care organizations during 2016-2019 showed that barely more than half, 52%, had both their uACR and estimated glomerular filtration rate (eGFR) checked annually as recommended by several U.S. medical societies, and just 73% had both values checked during a 3-year period, Nikita Stempniewicz, MSc, and associates reported in Diabetes Care.
More detailed data showed that measurement of eGFR was reasonably robust, measured at a 90% rate annually and in 97% of patients at least once every 3 years. But recording uACR values lagged, with a 53% annual rate and a 74% rate of measurement at least once every 3 years, reported Mr. Stempniewicz, director of research and analytics for the American Medical Group Association, a trade association based in Alexandria, Va. The 24 health care organizations that supplied the study’s data are all members of this association.
Prevailing recommendations from various medical societies call for annual monitoring of urinary albumin in patients with type 2 diabetes and specify the uACR, such as in the Standards of Medical Care in Diabetes from the American Diabetes Association, as well as in recommendations promoted by the National Kidney Foundation.
Missing half the CKD patients with eGFR only
“Half the patients with type 2 diabetes and chronic kidney disease have elevated albuminuria without decreased eGFR and would not be detected with eGFR testing alone,” Mr. Stempniewicz noted in an interview.
“Many patients who present for nephrology care are incompletely assessed with only low eGFR but no urine testing. Missing albuminuria testing and uACR values means patients with high levels of albuminuria but normal kidney function go undetected and thus are not able to benefit from evidenced-based interventions, including nephrology services,” said Joseph A. Vassalotti, MD, a nephrologist, chief medical officer for the National Kidney Foundation, and a coauthor of the report.
Not testing patients with type 2 diabetes regularly for their uACR “is a missed opportunity to identify the highest-risk patients and treat them,” added Josef Coresh, MD, PhD, a professor of clinical epidemiology at Johns Hopkins University, Baltimore, and senior author on the study. Measurement of albuminuria is especially important for these patients because medications from the sodium-glucose cotransporter 2 inhibitor class have been proven to slow progression of CKD in patients with type 2 diabetes, but these drugs are expensive, and in some cases have labeling that specifies the presence of albuminuria.
“I have no doubt that improving albuminuria testing is a critical step to identify patients with diabetes at highest risk who should get the best treatment possible, including SGLT2 inhibitors,” Dr. Coresh said in an interview.
The new report is not the first to document inadequate assessment of albuminuria and uACR among primary care physicians (PCPs), but it came from the largest reported U.S. study to date. “eGFR is commonly collected in a routine laboratory blood panel, but collecting urine requires additional work flow,” noted Cara B. Litvin, MD, a general internal medicine researcher at the Medical University of South Carolina, Charleston, who has tested interventions aimed at boosting CKD assessment by PCPs and was not involved in the new study.
“There have also been conflicting guidelines,” such as a “now-inactive guideline from the American College of Physicians that recommended against routine urine albumin screening in patients with diabetes and already on treatment with an angiotensin converting enzyme inhibitor or an angiotensin receptor blocker,” she said.
New renal drugs change the stakes
The availability of newer drugs for slowing CKD progression such as the SGLT2 inhibitors will help trigger greater support for routine albuminuria testing, Dr. Litvin predicted in an interview. “Now that we have more medications that can reduce albuminuria and improve outcomes, I see screening for albuminuria increasing.” Finerenone (Kerendia) is another new agent from a new class that recently received Food and Drug Administration approval for treating CKD in patients with type 2 diabetes.
Other drivers of increased uACR testing she expects include revised clinical practice guidelines, and new quality measures of clinical care.
“Undertesting of albuminuria means that [nephrologists] have incomplete data to detect and completely risk stratify the CKD population. That in turn results in a reduced ability to match population health interventions to the severity of the condition or the risk stratification based on eGFR and uACR,” Dr. Vassalotti said in an interview.
“We are missing opportunities to prevent or delay kidney failure and reduce the risk of cardiovascular events and cardiovascular death in these patients, particularly now that we have a number of medications that offer kidney and cardiovascular protection such as SGLT2 inhibitors,” he added. “Leaders in nephrology are beginning to understand the consequences of undertesting, and are working to innovate to improve risk stratification, CKD detection, and apply interventions to give Americans living with CKD better outcomes.”
Strategies proven to boost albuminuria testing
Mr. Stempniewicz and coauthors cited in their report potential strategies for improving albuminuria testing, including benchmarking to identify best-performing sites for albumin testing within a health system and encouraging replication of identified best practices at lower-performing sites, and implementation of clinical-decision support tools in the EHR such as pop-up test reminders.
These were among the tools tested in two studies led by Dr. Litvin. One study, with results reported in 2016, involved 12 small U.S. primary care practices with a total of more than 30,000 patients and compared performance in a series of clinical quality measures at baseline with performance after 2 years of receiving various interventions designed to boost awareness for albuminuria testing.
The second study, with findings reported in 2019, involved 21 U.S. primary care practices that collectively cared for more than 100,000 patients and randomized the practices to either undergo interventions aimed at boosting testing awareness or to serve as controls.
Results from both studies showed significant and substantial increases in serial testing for albuminuria in patients with diabetes or hypertension when practices received the interventions.
“We showed that [using a] clinical-decision support tool, along with standing orders to automatically collect urine specimens, dramatically increased screening for urinary albumin in primary care practices,” Dr. Litvin said. “However, perhaps because of conflicting guidelines and clinical inertia there hasn’t been a major impetus for primary care practices in general to improve screening.” She hopes that will quickly change.
“As we have shown, adoption of EHR-based reminders along with standing orders can very quickly improve screening for albuminuria in primary care.”
Variation in testing rates among sites ‘tremendous’
One finding of the new study gives Mr. Stempniewicz hope for greater future testing: The large variance that the researchers saw in albuminuria testing rates within individual health systems.
“The paper shows that higher rates of testing are completely achievable within each system. Some clinics do very well, and the other units can learn from these local successes,” he said. At least half the organizations in the study had individual sites that fell into the top 10% for testing rates across all the greater than 1,000 sites included, and those same organizations also had at least one site that fell into the bottom 10% for testing.
“The variation is tremendous, and highlights an opportunity for improvement,” declared Mr. Stempniewicz.
“For routine testing, you need systems that help people. Clinicians shouldn’t have to think about doing routine testing. It should just happen,” said Dr. Coresh.
The study was funded in part by Janssen. Mr. Stempniewicz and Dr. Litvin had no disclosures. Dr. Coresh is an adviser to Healthy.io, a company that markets a home albuminuria testing kit to patients. Dr. Vassalotti has received personal fees from Renalytix.
U.S. primary care physicians are not properly checking patients with type 2 diabetes for chronic kidney disease (CKD) nearly as often as they should, meaning many of these patients miss getting a timely diagnosis.
Inadequate measurement of urinary albumin-to-creatinine ratio (uACR) is the issue.
Review of data from more than half a million U.S. primary care patients with type 2 diabetes seen at any of 1,164 practice sites run by any of 24 health care organizations during 2016-2019 showed that barely more than half, 52%, had both their uACR and estimated glomerular filtration rate (eGFR) checked annually as recommended by several U.S. medical societies, and just 73% had both values checked during a 3-year period, Nikita Stempniewicz, MSc, and associates reported in Diabetes Care.
More detailed data showed that measurement of eGFR was reasonably robust, measured at a 90% rate annually and in 97% of patients at least once every 3 years. But recording uACR values lagged, with a 53% annual rate and a 74% rate of measurement at least once every 3 years, reported Mr. Stempniewicz, director of research and analytics for the American Medical Group Association, a trade association based in Alexandria, Va. The 24 health care organizations that supplied the study’s data are all members of this association.
Prevailing recommendations from various medical societies call for annual monitoring of urinary albumin in patients with type 2 diabetes and specify the uACR, such as in the Standards of Medical Care in Diabetes from the American Diabetes Association, as well as in recommendations promoted by the National Kidney Foundation.
Missing half the CKD patients with eGFR only
“Half the patients with type 2 diabetes and chronic kidney disease have elevated albuminuria without decreased eGFR and would not be detected with eGFR testing alone,” Mr. Stempniewicz noted in an interview.
“Many patients who present for nephrology care are incompletely assessed with only low eGFR but no urine testing. Missing albuminuria testing and uACR values means patients with high levels of albuminuria but normal kidney function go undetected and thus are not able to benefit from evidenced-based interventions, including nephrology services,” said Joseph A. Vassalotti, MD, a nephrologist, chief medical officer for the National Kidney Foundation, and a coauthor of the report.
Not testing patients with type 2 diabetes regularly for their uACR “is a missed opportunity to identify the highest-risk patients and treat them,” added Josef Coresh, MD, PhD, a professor of clinical epidemiology at Johns Hopkins University, Baltimore, and senior author on the study. Measurement of albuminuria is especially important for these patients because medications from the sodium-glucose cotransporter 2 inhibitor class have been proven to slow progression of CKD in patients with type 2 diabetes, but these drugs are expensive, and in some cases have labeling that specifies the presence of albuminuria.
“I have no doubt that improving albuminuria testing is a critical step to identify patients with diabetes at highest risk who should get the best treatment possible, including SGLT2 inhibitors,” Dr. Coresh said in an interview.
The new report is not the first to document inadequate assessment of albuminuria and uACR among primary care physicians (PCPs), but it came from the largest reported U.S. study to date. “eGFR is commonly collected in a routine laboratory blood panel, but collecting urine requires additional work flow,” noted Cara B. Litvin, MD, a general internal medicine researcher at the Medical University of South Carolina, Charleston, who has tested interventions aimed at boosting CKD assessment by PCPs and was not involved in the new study.
“There have also been conflicting guidelines,” such as a “now-inactive guideline from the American College of Physicians that recommended against routine urine albumin screening in patients with diabetes and already on treatment with an angiotensin converting enzyme inhibitor or an angiotensin receptor blocker,” she said.
New renal drugs change the stakes
The availability of newer drugs for slowing CKD progression such as the SGLT2 inhibitors will help trigger greater support for routine albuminuria testing, Dr. Litvin predicted in an interview. “Now that we have more medications that can reduce albuminuria and improve outcomes, I see screening for albuminuria increasing.” Finerenone (Kerendia) is another new agent from a new class that recently received Food and Drug Administration approval for treating CKD in patients with type 2 diabetes.
Other drivers of increased uACR testing she expects include revised clinical practice guidelines, and new quality measures of clinical care.
“Undertesting of albuminuria means that [nephrologists] have incomplete data to detect and completely risk stratify the CKD population. That in turn results in a reduced ability to match population health interventions to the severity of the condition or the risk stratification based on eGFR and uACR,” Dr. Vassalotti said in an interview.
“We are missing opportunities to prevent or delay kidney failure and reduce the risk of cardiovascular events and cardiovascular death in these patients, particularly now that we have a number of medications that offer kidney and cardiovascular protection such as SGLT2 inhibitors,” he added. “Leaders in nephrology are beginning to understand the consequences of undertesting, and are working to innovate to improve risk stratification, CKD detection, and apply interventions to give Americans living with CKD better outcomes.”
Strategies proven to boost albuminuria testing
Mr. Stempniewicz and coauthors cited in their report potential strategies for improving albuminuria testing, including benchmarking to identify best-performing sites for albumin testing within a health system and encouraging replication of identified best practices at lower-performing sites, and implementation of clinical-decision support tools in the EHR such as pop-up test reminders.
These were among the tools tested in two studies led by Dr. Litvin. One study, with results reported in 2016, involved 12 small U.S. primary care practices with a total of more than 30,000 patients and compared performance in a series of clinical quality measures at baseline with performance after 2 years of receiving various interventions designed to boost awareness for albuminuria testing.
The second study, with findings reported in 2019, involved 21 U.S. primary care practices that collectively cared for more than 100,000 patients and randomized the practices to either undergo interventions aimed at boosting testing awareness or to serve as controls.
Results from both studies showed significant and substantial increases in serial testing for albuminuria in patients with diabetes or hypertension when practices received the interventions.
“We showed that [using a] clinical-decision support tool, along with standing orders to automatically collect urine specimens, dramatically increased screening for urinary albumin in primary care practices,” Dr. Litvin said. “However, perhaps because of conflicting guidelines and clinical inertia there hasn’t been a major impetus for primary care practices in general to improve screening.” She hopes that will quickly change.
“As we have shown, adoption of EHR-based reminders along with standing orders can very quickly improve screening for albuminuria in primary care.”
Variation in testing rates among sites ‘tremendous’
One finding of the new study gives Mr. Stempniewicz hope for greater future testing: The large variance that the researchers saw in albuminuria testing rates within individual health systems.
“The paper shows that higher rates of testing are completely achievable within each system. Some clinics do very well, and the other units can learn from these local successes,” he said. At least half the organizations in the study had individual sites that fell into the top 10% for testing rates across all the greater than 1,000 sites included, and those same organizations also had at least one site that fell into the bottom 10% for testing.
“The variation is tremendous, and highlights an opportunity for improvement,” declared Mr. Stempniewicz.
“For routine testing, you need systems that help people. Clinicians shouldn’t have to think about doing routine testing. It should just happen,” said Dr. Coresh.
The study was funded in part by Janssen. Mr. Stempniewicz and Dr. Litvin had no disclosures. Dr. Coresh is an adviser to Healthy.io, a company that markets a home albuminuria testing kit to patients. Dr. Vassalotti has received personal fees from Renalytix.
FROM DIABETES CARE
More on GRADE: Cognitive deficits linked to CV risk factors in T2D
In type 2 diabetes (T2D), a greater degree of hyperlipidemia and hypertension, although not hyperglycemia, was associated with measurable cognitive impairment even among patients with only a 4-year mean disease duration, according to a substudy of the GRADE trial.
The association of these cardiovascular (CV) risk factors with impairments in cognition has been reported before, but the findings are notable because the mean duration of T2D was short in a relatively healthy study population, reported a multicenter team of investigators.
The relative impairments in cognitive function “may not be clinically significant given the very small size of the differences,” conceded the authors of this study, led by José A. Luchsinger, MD, but they are consistent with previous reports of the same association in older patients with a longer duration of diabetes. In other words, the data suggest the risk of cognitive loss from CV risk factors in T2D patients begins early.
“A potential explanation for the small differences, compared with those previously reported, is that the GRADE cohort is relatively young with a healthier cardiovascular profile and shorter diabetes duration compared with other studies,” reported the investigators, whose results were published online July 20, 2021, in Diabetes Care.
99% complete cognitive assessments
In the GRADE (Glycemia Reduction Approaches in Diabetes: Comparative Effectiveness) trial, 5,018 (99.4%) of the 5,047 enrolled patients completed a battery of cognitive assessments at baseline. Patients were excluded from this study if they had any major CV event in the previous year, if they had T2D for more than 10 years, if they had significant renal impairment, and if they had any history of stage 3 or greater heart failure. Their mean age was 56.7 years.
By cross-sectional analysis, cognitive evaluations, including the Digit Symbol Substitution Test (DSST) and the Spanish English Verbal Learning Test, were evaluated in relation to baseline LDL cholesterol levels, systolic and diastolic blood pressure, hemoglobin A1c, and statin use.
Unlike previous studies in T2D patients, no relationship was observed between cognitive function and A1c level at baseline. However, LDL cholesterol greater than 100 mg/dL was associated with cognitive impairment as measured with the DSST after adjustment for age, sex, education, and general health. The mean difference relative to LDL cholesterol below 70 mg/dL was only 1.8 points, but this was highly significant (P < .001).
Similarly, significant but modest cognitive impairment on DSST score after adjustment for variables were seen for those with a systolic BP between 120 mg and <140 mg relative to either <120 mm Hg or at least 140 mm Hg (P = .014). The same was seen for diastolic BPs of 80 to <90 when compared with either <80 mm Hg or to 90 mm Hg or higher (P = .01).
For those taking statins versus no statins at baseline, there was a 1.4-point mean advantage in DSST score after adjusting for variables (P < .001).
Modest cognitive impairments recorded
Again, the absolute mean differences in the DSST cognitive scores, despite their statistical significance, were modest, according to the authors. In general, the mean difference was rarely greater than 2.0 points and often 1.0 point or less. The authors acknowledged that these changes are of an uncertain clinical significance, but they considered the findings consistent with the association of CV risk factors with cognitive deficits in older T2DM patients or T2DM patients with longer duration of disease.
One difference between this GRADE substudy and previous studies was the lack of an association between cognitive impairment and hyperglycemia. In the ACCORD trial for example, increased levels of blood glycemia were associated with lower performance on numerous tests of cognitive function.
In the Diabetes Control and Complications Trial (DCCT), poorer glycemic control was related to poorer performance on tests of executive function.
Both of those studies also linked hypertension and hyperlipidemia with cognitive deficits, but given that patients in ACCORD had T2DM of substantially longer duration and those in DCCT were older, “it seems reasonable to speculate that, in patients with diabetes duration of less than 10 years, the association between hyperglycemia and cognitive performance may not yet be evident,” the GRADE authors reported.
GRADE trial compares drugs in four classes
The GRADE trial was conducted to compare four classes of T2D therapies for long-term glycemic control as expressed by A1c control over time. The results of the trial, presented recently at the 2021 annual scientific sessions of the American Diabetes Association, found that insulin glargine and the glucagonlike peptide–1 receptor agonist liraglutide performed best on the primary endpoint of maintaining A1c below 7.0%. Both performed significantly better than the sulfonylurea glimepiride and the dipeptidyl peptidase–4 inhibitor sitagliptin.
This substudy of baseline cognitive function in the relatively large GRADE trial provided a unique opportunity to evaluate the impact of CV risk factors in patients with T2D of relatively short duration.
While the data support the adverse impact of inadequately controlled modifiable risk factors on cognitive function in T2D patients, David R. Matthews, DPhil, BM, BCh, emeritus professor of diabetes medicine at the University of Oxford (England), noted that the association was weak and advised a cautious interpretation.
“The effect size is very small indeed. The data are found as a subset of multiple testing,” he said in an interview. He suggested the associations might be the result of “data farming,” and he emphasized that the relationships between these risk factors and cognitive deficits are associations that do not imply causation.
Nevertheless, and despite their unclear clinical implications, Dr. Matthews said that these data might still have a message.
“It is another reminder that for many reasons we all need to be alert to the need for lowering hyperlipidemia and hypertension to normal levels – the benefits may not just be limited to cardiovascular outcome,” Dr. Matthews stated.
The lead author of the study, Dr. Luchsinger, also cautioned against overinterpreting the data.
While the data show that “lipid and blood pressure control within recommended guidelines are associated with marginally better cognitive function in patients with type 2 diabetes of less than 5 years duration on average,” he added that “the study is limited by its cross-sectional nature.”
He indicated that further analysis will be helpful in assessing the implications.
“Longitudinal analyses of the same group of individuals will be conducted next year,” noted Dr. Luchsinger, associate professor of medicine and epidemiology, Columbia University Medical Center, New York.
Dr. Luchsinger reported financial relationships with vTv therapeutics. Dr. Matthews reported no potential conflicts of interest.
In type 2 diabetes (T2D), a greater degree of hyperlipidemia and hypertension, although not hyperglycemia, was associated with measurable cognitive impairment even among patients with only a 4-year mean disease duration, according to a substudy of the GRADE trial.
The association of these cardiovascular (CV) risk factors with impairments in cognition has been reported before, but the findings are notable because the mean duration of T2D was short in a relatively healthy study population, reported a multicenter team of investigators.
The relative impairments in cognitive function “may not be clinically significant given the very small size of the differences,” conceded the authors of this study, led by José A. Luchsinger, MD, but they are consistent with previous reports of the same association in older patients with a longer duration of diabetes. In other words, the data suggest the risk of cognitive loss from CV risk factors in T2D patients begins early.
“A potential explanation for the small differences, compared with those previously reported, is that the GRADE cohort is relatively young with a healthier cardiovascular profile and shorter diabetes duration compared with other studies,” reported the investigators, whose results were published online July 20, 2021, in Diabetes Care.
99% complete cognitive assessments
In the GRADE (Glycemia Reduction Approaches in Diabetes: Comparative Effectiveness) trial, 5,018 (99.4%) of the 5,047 enrolled patients completed a battery of cognitive assessments at baseline. Patients were excluded from this study if they had any major CV event in the previous year, if they had T2D for more than 10 years, if they had significant renal impairment, and if they had any history of stage 3 or greater heart failure. Their mean age was 56.7 years.
By cross-sectional analysis, cognitive evaluations, including the Digit Symbol Substitution Test (DSST) and the Spanish English Verbal Learning Test, were evaluated in relation to baseline LDL cholesterol levels, systolic and diastolic blood pressure, hemoglobin A1c, and statin use.
Unlike previous studies in T2D patients, no relationship was observed between cognitive function and A1c level at baseline. However, LDL cholesterol greater than 100 mg/dL was associated with cognitive impairment as measured with the DSST after adjustment for age, sex, education, and general health. The mean difference relative to LDL cholesterol below 70 mg/dL was only 1.8 points, but this was highly significant (P < .001).
Similarly, significant but modest cognitive impairment on DSST score after adjustment for variables were seen for those with a systolic BP between 120 mg and <140 mg relative to either <120 mm Hg or at least 140 mm Hg (P = .014). The same was seen for diastolic BPs of 80 to <90 when compared with either <80 mm Hg or to 90 mm Hg or higher (P = .01).
For those taking statins versus no statins at baseline, there was a 1.4-point mean advantage in DSST score after adjusting for variables (P < .001).
Modest cognitive impairments recorded
Again, the absolute mean differences in the DSST cognitive scores, despite their statistical significance, were modest, according to the authors. In general, the mean difference was rarely greater than 2.0 points and often 1.0 point or less. The authors acknowledged that these changes are of an uncertain clinical significance, but they considered the findings consistent with the association of CV risk factors with cognitive deficits in older T2DM patients or T2DM patients with longer duration of disease.
One difference between this GRADE substudy and previous studies was the lack of an association between cognitive impairment and hyperglycemia. In the ACCORD trial for example, increased levels of blood glycemia were associated with lower performance on numerous tests of cognitive function.
In the Diabetes Control and Complications Trial (DCCT), poorer glycemic control was related to poorer performance on tests of executive function.
Both of those studies also linked hypertension and hyperlipidemia with cognitive deficits, but given that patients in ACCORD had T2DM of substantially longer duration and those in DCCT were older, “it seems reasonable to speculate that, in patients with diabetes duration of less than 10 years, the association between hyperglycemia and cognitive performance may not yet be evident,” the GRADE authors reported.
GRADE trial compares drugs in four classes
The GRADE trial was conducted to compare four classes of T2D therapies for long-term glycemic control as expressed by A1c control over time. The results of the trial, presented recently at the 2021 annual scientific sessions of the American Diabetes Association, found that insulin glargine and the glucagonlike peptide–1 receptor agonist liraglutide performed best on the primary endpoint of maintaining A1c below 7.0%. Both performed significantly better than the sulfonylurea glimepiride and the dipeptidyl peptidase–4 inhibitor sitagliptin.
This substudy of baseline cognitive function in the relatively large GRADE trial provided a unique opportunity to evaluate the impact of CV risk factors in patients with T2D of relatively short duration.
While the data support the adverse impact of inadequately controlled modifiable risk factors on cognitive function in T2D patients, David R. Matthews, DPhil, BM, BCh, emeritus professor of diabetes medicine at the University of Oxford (England), noted that the association was weak and advised a cautious interpretation.
“The effect size is very small indeed. The data are found as a subset of multiple testing,” he said in an interview. He suggested the associations might be the result of “data farming,” and he emphasized that the relationships between these risk factors and cognitive deficits are associations that do not imply causation.
Nevertheless, and despite their unclear clinical implications, Dr. Matthews said that these data might still have a message.
“It is another reminder that for many reasons we all need to be alert to the need for lowering hyperlipidemia and hypertension to normal levels – the benefits may not just be limited to cardiovascular outcome,” Dr. Matthews stated.
The lead author of the study, Dr. Luchsinger, also cautioned against overinterpreting the data.
While the data show that “lipid and blood pressure control within recommended guidelines are associated with marginally better cognitive function in patients with type 2 diabetes of less than 5 years duration on average,” he added that “the study is limited by its cross-sectional nature.”
He indicated that further analysis will be helpful in assessing the implications.
“Longitudinal analyses of the same group of individuals will be conducted next year,” noted Dr. Luchsinger, associate professor of medicine and epidemiology, Columbia University Medical Center, New York.
Dr. Luchsinger reported financial relationships with vTv therapeutics. Dr. Matthews reported no potential conflicts of interest.
In type 2 diabetes (T2D), a greater degree of hyperlipidemia and hypertension, although not hyperglycemia, was associated with measurable cognitive impairment even among patients with only a 4-year mean disease duration, according to a substudy of the GRADE trial.
The association of these cardiovascular (CV) risk factors with impairments in cognition has been reported before, but the findings are notable because the mean duration of T2D was short in a relatively healthy study population, reported a multicenter team of investigators.
The relative impairments in cognitive function “may not be clinically significant given the very small size of the differences,” conceded the authors of this study, led by José A. Luchsinger, MD, but they are consistent with previous reports of the same association in older patients with a longer duration of diabetes. In other words, the data suggest the risk of cognitive loss from CV risk factors in T2D patients begins early.
“A potential explanation for the small differences, compared with those previously reported, is that the GRADE cohort is relatively young with a healthier cardiovascular profile and shorter diabetes duration compared with other studies,” reported the investigators, whose results were published online July 20, 2021, in Diabetes Care.
99% complete cognitive assessments
In the GRADE (Glycemia Reduction Approaches in Diabetes: Comparative Effectiveness) trial, 5,018 (99.4%) of the 5,047 enrolled patients completed a battery of cognitive assessments at baseline. Patients were excluded from this study if they had any major CV event in the previous year, if they had T2D for more than 10 years, if they had significant renal impairment, and if they had any history of stage 3 or greater heart failure. Their mean age was 56.7 years.
By cross-sectional analysis, cognitive evaluations, including the Digit Symbol Substitution Test (DSST) and the Spanish English Verbal Learning Test, were evaluated in relation to baseline LDL cholesterol levels, systolic and diastolic blood pressure, hemoglobin A1c, and statin use.
Unlike previous studies in T2D patients, no relationship was observed between cognitive function and A1c level at baseline. However, LDL cholesterol greater than 100 mg/dL was associated with cognitive impairment as measured with the DSST after adjustment for age, sex, education, and general health. The mean difference relative to LDL cholesterol below 70 mg/dL was only 1.8 points, but this was highly significant (P < .001).
Similarly, significant but modest cognitive impairment on DSST score after adjustment for variables were seen for those with a systolic BP between 120 mg and <140 mg relative to either <120 mm Hg or at least 140 mm Hg (P = .014). The same was seen for diastolic BPs of 80 to <90 when compared with either <80 mm Hg or to 90 mm Hg or higher (P = .01).
For those taking statins versus no statins at baseline, there was a 1.4-point mean advantage in DSST score after adjusting for variables (P < .001).
Modest cognitive impairments recorded
Again, the absolute mean differences in the DSST cognitive scores, despite their statistical significance, were modest, according to the authors. In general, the mean difference was rarely greater than 2.0 points and often 1.0 point or less. The authors acknowledged that these changes are of an uncertain clinical significance, but they considered the findings consistent with the association of CV risk factors with cognitive deficits in older T2DM patients or T2DM patients with longer duration of disease.
One difference between this GRADE substudy and previous studies was the lack of an association between cognitive impairment and hyperglycemia. In the ACCORD trial for example, increased levels of blood glycemia were associated with lower performance on numerous tests of cognitive function.
In the Diabetes Control and Complications Trial (DCCT), poorer glycemic control was related to poorer performance on tests of executive function.
Both of those studies also linked hypertension and hyperlipidemia with cognitive deficits, but given that patients in ACCORD had T2DM of substantially longer duration and those in DCCT were older, “it seems reasonable to speculate that, in patients with diabetes duration of less than 10 years, the association between hyperglycemia and cognitive performance may not yet be evident,” the GRADE authors reported.
GRADE trial compares drugs in four classes
The GRADE trial was conducted to compare four classes of T2D therapies for long-term glycemic control as expressed by A1c control over time. The results of the trial, presented recently at the 2021 annual scientific sessions of the American Diabetes Association, found that insulin glargine and the glucagonlike peptide–1 receptor agonist liraglutide performed best on the primary endpoint of maintaining A1c below 7.0%. Both performed significantly better than the sulfonylurea glimepiride and the dipeptidyl peptidase–4 inhibitor sitagliptin.
This substudy of baseline cognitive function in the relatively large GRADE trial provided a unique opportunity to evaluate the impact of CV risk factors in patients with T2D of relatively short duration.
While the data support the adverse impact of inadequately controlled modifiable risk factors on cognitive function in T2D patients, David R. Matthews, DPhil, BM, BCh, emeritus professor of diabetes medicine at the University of Oxford (England), noted that the association was weak and advised a cautious interpretation.
“The effect size is very small indeed. The data are found as a subset of multiple testing,” he said in an interview. He suggested the associations might be the result of “data farming,” and he emphasized that the relationships between these risk factors and cognitive deficits are associations that do not imply causation.
Nevertheless, and despite their unclear clinical implications, Dr. Matthews said that these data might still have a message.
“It is another reminder that for many reasons we all need to be alert to the need for lowering hyperlipidemia and hypertension to normal levels – the benefits may not just be limited to cardiovascular outcome,” Dr. Matthews stated.
The lead author of the study, Dr. Luchsinger, also cautioned against overinterpreting the data.
While the data show that “lipid and blood pressure control within recommended guidelines are associated with marginally better cognitive function in patients with type 2 diabetes of less than 5 years duration on average,” he added that “the study is limited by its cross-sectional nature.”
He indicated that further analysis will be helpful in assessing the implications.
“Longitudinal analyses of the same group of individuals will be conducted next year,” noted Dr. Luchsinger, associate professor of medicine and epidemiology, Columbia University Medical Center, New York.
Dr. Luchsinger reported financial relationships with vTv therapeutics. Dr. Matthews reported no potential conflicts of interest.
FROM DIABETES CARE
Diabetes duration linked to increasing heart failure risk
In a multivariable analysis the rate of incident heart failure increased steadily and significantly as diabetes duration increased. Among the 168 study subjects (2% of the total study group) who had diabetes for at least 15 years, the subsequent incidence of heart failure was nearly threefold higher than among the 4,802 subjects (49%) who never had diabetes or prediabetes, reported Justin B. Echouffo-Tcheugui, MD, PhD, and coauthors in an article published in JACC Heart Failure.
People with prediabetes (32% of the study population) had a significant but modest increased rate of incident heart failure that was 16% higher than in control subjects who never developed diabetes. People with diabetes for durations of 0-4.9 years, 5.0-9.9 years, or 10-14.9 years, had steadily increasing relative incident heart failure rates of 29%, 97%, and 210%, respectively, compared with controls, reported Dr. Echouffo-Tcheugui, an endocrinologist at Johns Hopkins Medicine in Baltimore.
Similar rates of HFrEF and HFpEF
Among all 1,841 people in the dataset with diabetes for any length of time each additional 5 years of the disorder linked with a significant, relative 17% increase in the rate of incident heart failure. Incidence of heart failure rose even more sharply with added duration among those with a hemoglobin A1c of 7% or greater, compared with those with better glycemic control. And the rate of incident heart failure with reduced ejection fraction (HFrEF) roughly matched the rate of incident heart failure with preserved ejection fraction (HFpEF).
The study dataset included 9,734 adults enrolled into the Atherosclerosis Risk in Communities (ARIC) study, and during a median follow-up of 22.5 years they had nearly 2,000 episodes of either hospitalization or death secondary to incident heart failure. This included 617 (31%) events involving HFpEF, 495 events (25%) involving HFrEF, and 876 unclassified heart failure events.
The cohort averaged 63 years of age; 58% were women, 23% were Black, and 77% were White (the study design excluded people with other racial and ethnic backgrounds). The study design also excluded people with a history of heart failure or coronary artery disease, as well as those diagnosed with diabetes prior to age 18 resulting in a study group that presumably mostly had type 2 diabetes when diabetes was present. The report provided no data on the specific numbers of patients with type 1 or type 2 diabetes.
“It’s not surprising that a longer duration of diabetes is associated with heart failure, but the etiology remains problematic,” commented Robert H. Eckel, MD, an endocrinologist at the University of Colorado at Denver, Aurora. “The impact of diabetes on incident heart failure is not well know, particularly duration of diabetes,” although disorders often found in patients with diabetes, such as hypertension and diabetic cardiomyopathy, likely have roles in causing heart failure, he said.
Diabetes duration may signal need for an SGLT2 inhibitor
“With emerging novel treatments like the SGLT2 [sodium-glucose cotransporter 2] inhibitors for preventing heart failure hospitalizations and deaths in patients with type 2 diabetes, this is a timely analysis,” Dr. Eckel said in an interview.
“There is no question that with increased duration of type 2 diabetes” the need for an agent from the SGLT2-inhibitor class increases. Although, because of the proven protection these drugs give against heart failure events and progression of chronic kidney disease, treatment with this drug class should start early in patients with type 2 diabetes, he added.
Dr. Echouffo-Tcheugui and his coauthors agreed, citing two important clinical take-aways from their findings:
First, interventions that delay the onset of diabetes may potentially reduce incident heart failure; second, patients with diabetes might benefit from cardioprotective treatments such as SGLT2 inhibitors, the report said.
“Our observations suggest the potential prognostic relevance of diabetes duration in assessing heart failure,” the authors wrote. Integrating diabetes duration into heart failure risk estimation in people with diabetes “could help refine the selection of high-risk individuals who may derive the greatest absolute benefit from aggressive cardioprotective therapies such as SGLT2 inhibitors.”
The analysis also identified several other demographic and clinical factors that influenced the relative effect of diabetes duration. Longer duration was linked with higher rates of incident heart failure in women compared with men, in Blacks compared with Whites, in people younger than 65 compared with older people, in people with an A1c of 7% or higher, and in those with a body mass index of 30 kg/m2 or greater.
The ARIC study and the analyses run by Dr. Echouffo-Tcheugui and his coauthors received no commercial funding. Dr. Echouffo-Tcheugui and Dr. Eckel had no relevant disclosures.
In a multivariable analysis the rate of incident heart failure increased steadily and significantly as diabetes duration increased. Among the 168 study subjects (2% of the total study group) who had diabetes for at least 15 years, the subsequent incidence of heart failure was nearly threefold higher than among the 4,802 subjects (49%) who never had diabetes or prediabetes, reported Justin B. Echouffo-Tcheugui, MD, PhD, and coauthors in an article published in JACC Heart Failure.
People with prediabetes (32% of the study population) had a significant but modest increased rate of incident heart failure that was 16% higher than in control subjects who never developed diabetes. People with diabetes for durations of 0-4.9 years, 5.0-9.9 years, or 10-14.9 years, had steadily increasing relative incident heart failure rates of 29%, 97%, and 210%, respectively, compared with controls, reported Dr. Echouffo-Tcheugui, an endocrinologist at Johns Hopkins Medicine in Baltimore.
Similar rates of HFrEF and HFpEF
Among all 1,841 people in the dataset with diabetes for any length of time each additional 5 years of the disorder linked with a significant, relative 17% increase in the rate of incident heart failure. Incidence of heart failure rose even more sharply with added duration among those with a hemoglobin A1c of 7% or greater, compared with those with better glycemic control. And the rate of incident heart failure with reduced ejection fraction (HFrEF) roughly matched the rate of incident heart failure with preserved ejection fraction (HFpEF).
The study dataset included 9,734 adults enrolled into the Atherosclerosis Risk in Communities (ARIC) study, and during a median follow-up of 22.5 years they had nearly 2,000 episodes of either hospitalization or death secondary to incident heart failure. This included 617 (31%) events involving HFpEF, 495 events (25%) involving HFrEF, and 876 unclassified heart failure events.
The cohort averaged 63 years of age; 58% were women, 23% were Black, and 77% were White (the study design excluded people with other racial and ethnic backgrounds). The study design also excluded people with a history of heart failure or coronary artery disease, as well as those diagnosed with diabetes prior to age 18 resulting in a study group that presumably mostly had type 2 diabetes when diabetes was present. The report provided no data on the specific numbers of patients with type 1 or type 2 diabetes.
“It’s not surprising that a longer duration of diabetes is associated with heart failure, but the etiology remains problematic,” commented Robert H. Eckel, MD, an endocrinologist at the University of Colorado at Denver, Aurora. “The impact of diabetes on incident heart failure is not well know, particularly duration of diabetes,” although disorders often found in patients with diabetes, such as hypertension and diabetic cardiomyopathy, likely have roles in causing heart failure, he said.
Diabetes duration may signal need for an SGLT2 inhibitor
“With emerging novel treatments like the SGLT2 [sodium-glucose cotransporter 2] inhibitors for preventing heart failure hospitalizations and deaths in patients with type 2 diabetes, this is a timely analysis,” Dr. Eckel said in an interview.
“There is no question that with increased duration of type 2 diabetes” the need for an agent from the SGLT2-inhibitor class increases. Although, because of the proven protection these drugs give against heart failure events and progression of chronic kidney disease, treatment with this drug class should start early in patients with type 2 diabetes, he added.
Dr. Echouffo-Tcheugui and his coauthors agreed, citing two important clinical take-aways from their findings:
First, interventions that delay the onset of diabetes may potentially reduce incident heart failure; second, patients with diabetes might benefit from cardioprotective treatments such as SGLT2 inhibitors, the report said.
“Our observations suggest the potential prognostic relevance of diabetes duration in assessing heart failure,” the authors wrote. Integrating diabetes duration into heart failure risk estimation in people with diabetes “could help refine the selection of high-risk individuals who may derive the greatest absolute benefit from aggressive cardioprotective therapies such as SGLT2 inhibitors.”
The analysis also identified several other demographic and clinical factors that influenced the relative effect of diabetes duration. Longer duration was linked with higher rates of incident heart failure in women compared with men, in Blacks compared with Whites, in people younger than 65 compared with older people, in people with an A1c of 7% or higher, and in those with a body mass index of 30 kg/m2 or greater.
The ARIC study and the analyses run by Dr. Echouffo-Tcheugui and his coauthors received no commercial funding. Dr. Echouffo-Tcheugui and Dr. Eckel had no relevant disclosures.
In a multivariable analysis the rate of incident heart failure increased steadily and significantly as diabetes duration increased. Among the 168 study subjects (2% of the total study group) who had diabetes for at least 15 years, the subsequent incidence of heart failure was nearly threefold higher than among the 4,802 subjects (49%) who never had diabetes or prediabetes, reported Justin B. Echouffo-Tcheugui, MD, PhD, and coauthors in an article published in JACC Heart Failure.
People with prediabetes (32% of the study population) had a significant but modest increased rate of incident heart failure that was 16% higher than in control subjects who never developed diabetes. People with diabetes for durations of 0-4.9 years, 5.0-9.9 years, or 10-14.9 years, had steadily increasing relative incident heart failure rates of 29%, 97%, and 210%, respectively, compared with controls, reported Dr. Echouffo-Tcheugui, an endocrinologist at Johns Hopkins Medicine in Baltimore.
Similar rates of HFrEF and HFpEF
Among all 1,841 people in the dataset with diabetes for any length of time each additional 5 years of the disorder linked with a significant, relative 17% increase in the rate of incident heart failure. Incidence of heart failure rose even more sharply with added duration among those with a hemoglobin A1c of 7% or greater, compared with those with better glycemic control. And the rate of incident heart failure with reduced ejection fraction (HFrEF) roughly matched the rate of incident heart failure with preserved ejection fraction (HFpEF).
The study dataset included 9,734 adults enrolled into the Atherosclerosis Risk in Communities (ARIC) study, and during a median follow-up of 22.5 years they had nearly 2,000 episodes of either hospitalization or death secondary to incident heart failure. This included 617 (31%) events involving HFpEF, 495 events (25%) involving HFrEF, and 876 unclassified heart failure events.
The cohort averaged 63 years of age; 58% were women, 23% were Black, and 77% were White (the study design excluded people with other racial and ethnic backgrounds). The study design also excluded people with a history of heart failure or coronary artery disease, as well as those diagnosed with diabetes prior to age 18 resulting in a study group that presumably mostly had type 2 diabetes when diabetes was present. The report provided no data on the specific numbers of patients with type 1 or type 2 diabetes.
“It’s not surprising that a longer duration of diabetes is associated with heart failure, but the etiology remains problematic,” commented Robert H. Eckel, MD, an endocrinologist at the University of Colorado at Denver, Aurora. “The impact of diabetes on incident heart failure is not well know, particularly duration of diabetes,” although disorders often found in patients with diabetes, such as hypertension and diabetic cardiomyopathy, likely have roles in causing heart failure, he said.
Diabetes duration may signal need for an SGLT2 inhibitor
“With emerging novel treatments like the SGLT2 [sodium-glucose cotransporter 2] inhibitors for preventing heart failure hospitalizations and deaths in patients with type 2 diabetes, this is a timely analysis,” Dr. Eckel said in an interview.
“There is no question that with increased duration of type 2 diabetes” the need for an agent from the SGLT2-inhibitor class increases. Although, because of the proven protection these drugs give against heart failure events and progression of chronic kidney disease, treatment with this drug class should start early in patients with type 2 diabetes, he added.
Dr. Echouffo-Tcheugui and his coauthors agreed, citing two important clinical take-aways from their findings:
First, interventions that delay the onset of diabetes may potentially reduce incident heart failure; second, patients with diabetes might benefit from cardioprotective treatments such as SGLT2 inhibitors, the report said.
“Our observations suggest the potential prognostic relevance of diabetes duration in assessing heart failure,” the authors wrote. Integrating diabetes duration into heart failure risk estimation in people with diabetes “could help refine the selection of high-risk individuals who may derive the greatest absolute benefit from aggressive cardioprotective therapies such as SGLT2 inhibitors.”
The analysis also identified several other demographic and clinical factors that influenced the relative effect of diabetes duration. Longer duration was linked with higher rates of incident heart failure in women compared with men, in Blacks compared with Whites, in people younger than 65 compared with older people, in people with an A1c of 7% or higher, and in those with a body mass index of 30 kg/m2 or greater.
The ARIC study and the analyses run by Dr. Echouffo-Tcheugui and his coauthors received no commercial funding. Dr. Echouffo-Tcheugui and Dr. Eckel had no relevant disclosures.
FROM JACC HEART FAILURE
This is not the time to modify a HTN regimen
ILLUSTRATIVE CASE
A 67-year-old man with hypertension that is well controlled on hydrochlorothiazide 25 mg po daily was admitted to the family medicine inpatient service for community-acquired pneumonia requiring antibiotic therapy and oxygen support. Despite improvement in his overall condition, his blood pressure was consistently > 160/90 mm Hg during his hospitalization. He was treated with lisinopril 10 mg po daily in addition to his home medications, which helped achieve recommended blood pressure goals.
Prior to discharge, his blood pressure was noted to be 108/62 mm Hg. He asks if it is necessary to continue this new blood pressure medicine, as his home blood pressure readings had been within the goal set by his primary care physician. Should you continue this new antihypertensive agent at discharge?
Outpatient antihypertensive medication regimens are commonly intensified at hospital discharge in response to transient short-term elevations in blood pressure during inpatient encounters for noncardiac conditions.1,2 This is typically a reflexive response during a hospitalization, despite the unknown long-term, patient-oriented clinical outcomes. These short-term, in-hospital blood pressure elevations may be due to numerous temporary causes, such as stress/anxiety, a pain response, agitation, a medication adverse effect, or volume overload.3
The transition from inpatient to outpatient care is a high-risk period, especially for older adults, as functional status is generally worse at hospital discharge than prehospitalization baseline.4 To compound this problem, adverse drug reactions are a common cause of hospitalization for older adults. Changing blood pressure medications in response to acute physiologic changes during illness may contribute to patient harm. Although observational studies of adverse drug reactions related to blood pressure medications are numerous, researchers have only evaluated adverse drug reactions pertaining to hospital admissions.5-8 This study sought to evaluate the clinical outcomes associated with intensification of antihypertensive regimens at discharge among older adults.
STUDY SUMMARY
Increased risk of readmission, adverse events after intensification at discharge
This retrospective cohort study, which was conducted across multiple
Antihypertensive medication changes at discharge were evaluated using information pulled from VHA pharmacies, combined with clinical data merged from VHA and Medicare claims. Intensification was defined as either adding a new blood pressure medication or a dose increase of more than 20% on a previously prescribed antihypertensive medication. Patients were excluded if they were discharged with a secondary diagnosis that required modifications to a blood pressure medication (such as atrial fibrillation, acute coronary syndrome, or stroke), were hospitalized in the previous 30 days, were admitted from a skilled nursing facility, or received more than 20% of their care (including filling prescriptions) outside the VHA system.
Primary outcomes included hospital readmission or SAEs (falls, syncope, hypotension, serious electrolyte abnormalities, or acute kidney injury) within 30 days or having a cardiovascular event within 1 year of hospital discharge. Secondary outcomes included the change in systolic blood pressure (SBP) within 1 year after discharge. Propensity score matching was used as a balancing factor to create a matched-pairs cohort to compare those receiving blood pressure medication intensification at hospital discharge with those who did not.
Continue to: Intensification of the blood pressure...
Intensification of the blood pressure regimen at hospital discharge was associated with an increased risk in 30-day hospital readmission (hazard ratio [HR] = 1.23; 95% CI, 1.07–1.42; number needed to harm [NNH] = 27) and SAEs (HR = 1.41; 95% CI, 1.06–1.88; NNH = 63). There was no associated reduction in cardiovascular events (HR = 1.18; 95% CI, 0.99–1.40) or change in mean SBP within 1 year after hospital discharge in those who received intensification vs those who did not (mean BP, 134.7 vs 134.4 mm Hg; difference-in-differences estimate = 0.2 mm Hg; 95% CI, −2.0 to 2.4 mm Hg).
WHAT’S NEW
First study on outcomes related to HTN med changes at hospital discharge
This well-designed, retrospective cohort study provides important clinical data to help guide inpatient blood pressure management decisions for patients with noncardiac conditions. No clinical trials up to that time had assessed patient-oriented outcomes when antihypertensive medication regimens were intensified at hospital discharge.
CAVEATS
Study population: Primarily older men with noncardiac conditions
Selected populations benefit from intensive blood pressure control based on specific risk factors and medical conditions. In patients at high risk for cardiovascular disease, without a history of stroke or diabetes, intensive blood pressure control (SBP < 120 mm Hg) improves cardiovascular outcomes and overall survival compared with standard therapy (SBP < 140 mm Hg).9 This retrospective cohort study involved mainly elderly male patients with noncardiac conditions. The study also excluded patients with a secondary diagnosis requiring modifications to an antihypertensive regimen, such as atrial fibrillation, acute coronary syndrome, or cerebrovascular accident. Thus, the findings may not be applicable to these patient populations.
CHALLENGES TO IMPLEMENTATION
Clinicians will need to address individual needs
Physicians have to balance various antihypertensive management strategies, as competing medical specialty society guidelines recommend differing targets for optimal blood pressure control. Given the concern for medicolegal liability and potential harms of therapeutic inertia, inpatient physicians must consider whether hospitalization is the best time to alter medications for long-term outpatient blood pressure control. Finally, the decision to leave blood pressure management to outpatient physicians assumes the patient has a continuity relationship with a primary care medical home.
ACKNOWLEDGEMENT
The PURLs Surveillance System was supported in part by Grant Number UL1RR024999 from the National Center for Research Resources, a Clinical Translational Science Award to the University of Chicago. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center for Research Resources or the National Institutes of Health.
1. Anderson TS, Jing B, Auerbach A, et al. Clinical outcomes after intensifying antihypertensive medication regimens among older adults at hospital discharge. JAMA Intern Med. 2019;179:1528-1536.
2. Harris CM, Sridharan A, Landis R, et al. What happens to the medication regimens of older adults during and after an acute hospitalization? J Patient Saf. 2013;9:150-153.
3. Aung WM, Menon SV, Materson BJ. Management of hypertension in hospitalized patients. Hosp Pract (1995). 2015;43:101-106.
4. Covinsky KE, Palmer RM, Fortinsky RH, et al. Loss of independence in activities of daily living in older adults hospitalized with medical illnesses: increased vulnerability with age. J Am Geriatr Soc. 2003;51:451-458.
5. Omer HMRB, Hodson J, Pontefract SK, et al. Inpatient falls in older adults: a cohort study of antihypertensive prescribing pre- and post-fall. BMC Geriatr. 2018;18:58.
6. Alhawassi TM, Krass I, Pont LG. Antihypertensive-related adverse drug reactions among older hospitalized adults. Int J Clin Pharm. 2018;40:428-435.
7. Passarelli MCG, Jacob-Filho W, Figueras A. Adverse drug reactions in an elderly hospitalised population: inappropriate prescription is a leading cause. Drugs Aging. 2005;22:767-777.
8. Beckett NS, Peters R, Fletcher AE, et al; HYVET Study Group. Treatment of hypertension in patients 80 years of age or older. N Engl J Med. 2008;358:1887-1898.
9. SPRINT Research Group; Wright JT Jr, Williamson JD, Whelton PK, et al. A randomized trial of intensive versus standard blood-pressure control. N Engl J Med. 2015;373:2103-2116. Published correction appears in N Engl J Med. 2017;377:2506.
ILLUSTRATIVE CASE
A 67-year-old man with hypertension that is well controlled on hydrochlorothiazide 25 mg po daily was admitted to the family medicine inpatient service for community-acquired pneumonia requiring antibiotic therapy and oxygen support. Despite improvement in his overall condition, his blood pressure was consistently > 160/90 mm Hg during his hospitalization. He was treated with lisinopril 10 mg po daily in addition to his home medications, which helped achieve recommended blood pressure goals.
Prior to discharge, his blood pressure was noted to be 108/62 mm Hg. He asks if it is necessary to continue this new blood pressure medicine, as his home blood pressure readings had been within the goal set by his primary care physician. Should you continue this new antihypertensive agent at discharge?
Outpatient antihypertensive medication regimens are commonly intensified at hospital discharge in response to transient short-term elevations in blood pressure during inpatient encounters for noncardiac conditions.1,2 This is typically a reflexive response during a hospitalization, despite the unknown long-term, patient-oriented clinical outcomes. These short-term, in-hospital blood pressure elevations may be due to numerous temporary causes, such as stress/anxiety, a pain response, agitation, a medication adverse effect, or volume overload.3
The transition from inpatient to outpatient care is a high-risk period, especially for older adults, as functional status is generally worse at hospital discharge than prehospitalization baseline.4 To compound this problem, adverse drug reactions are a common cause of hospitalization for older adults. Changing blood pressure medications in response to acute physiologic changes during illness may contribute to patient harm. Although observational studies of adverse drug reactions related to blood pressure medications are numerous, researchers have only evaluated adverse drug reactions pertaining to hospital admissions.5-8 This study sought to evaluate the clinical outcomes associated with intensification of antihypertensive regimens at discharge among older adults.
STUDY SUMMARY
Increased risk of readmission, adverse events after intensification at discharge
This retrospective cohort study, which was conducted across multiple
Antihypertensive medication changes at discharge were evaluated using information pulled from VHA pharmacies, combined with clinical data merged from VHA and Medicare claims. Intensification was defined as either adding a new blood pressure medication or a dose increase of more than 20% on a previously prescribed antihypertensive medication. Patients were excluded if they were discharged with a secondary diagnosis that required modifications to a blood pressure medication (such as atrial fibrillation, acute coronary syndrome, or stroke), were hospitalized in the previous 30 days, were admitted from a skilled nursing facility, or received more than 20% of their care (including filling prescriptions) outside the VHA system.
Primary outcomes included hospital readmission or SAEs (falls, syncope, hypotension, serious electrolyte abnormalities, or acute kidney injury) within 30 days or having a cardiovascular event within 1 year of hospital discharge. Secondary outcomes included the change in systolic blood pressure (SBP) within 1 year after discharge. Propensity score matching was used as a balancing factor to create a matched-pairs cohort to compare those receiving blood pressure medication intensification at hospital discharge with those who did not.
Continue to: Intensification of the blood pressure...
Intensification of the blood pressure regimen at hospital discharge was associated with an increased risk in 30-day hospital readmission (hazard ratio [HR] = 1.23; 95% CI, 1.07–1.42; number needed to harm [NNH] = 27) and SAEs (HR = 1.41; 95% CI, 1.06–1.88; NNH = 63). There was no associated reduction in cardiovascular events (HR = 1.18; 95% CI, 0.99–1.40) or change in mean SBP within 1 year after hospital discharge in those who received intensification vs those who did not (mean BP, 134.7 vs 134.4 mm Hg; difference-in-differences estimate = 0.2 mm Hg; 95% CI, −2.0 to 2.4 mm Hg).
WHAT’S NEW
First study on outcomes related to HTN med changes at hospital discharge
This well-designed, retrospective cohort study provides important clinical data to help guide inpatient blood pressure management decisions for patients with noncardiac conditions. No clinical trials up to that time had assessed patient-oriented outcomes when antihypertensive medication regimens were intensified at hospital discharge.
CAVEATS
Study population: Primarily older men with noncardiac conditions
Selected populations benefit from intensive blood pressure control based on specific risk factors and medical conditions. In patients at high risk for cardiovascular disease, without a history of stroke or diabetes, intensive blood pressure control (SBP < 120 mm Hg) improves cardiovascular outcomes and overall survival compared with standard therapy (SBP < 140 mm Hg).9 This retrospective cohort study involved mainly elderly male patients with noncardiac conditions. The study also excluded patients with a secondary diagnosis requiring modifications to an antihypertensive regimen, such as atrial fibrillation, acute coronary syndrome, or cerebrovascular accident. Thus, the findings may not be applicable to these patient populations.
CHALLENGES TO IMPLEMENTATION
Clinicians will need to address individual needs
Physicians have to balance various antihypertensive management strategies, as competing medical specialty society guidelines recommend differing targets for optimal blood pressure control. Given the concern for medicolegal liability and potential harms of therapeutic inertia, inpatient physicians must consider whether hospitalization is the best time to alter medications for long-term outpatient blood pressure control. Finally, the decision to leave blood pressure management to outpatient physicians assumes the patient has a continuity relationship with a primary care medical home.
ACKNOWLEDGEMENT
The PURLs Surveillance System was supported in part by Grant Number UL1RR024999 from the National Center for Research Resources, a Clinical Translational Science Award to the University of Chicago. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center for Research Resources or the National Institutes of Health.
ILLUSTRATIVE CASE
A 67-year-old man with hypertension that is well controlled on hydrochlorothiazide 25 mg po daily was admitted to the family medicine inpatient service for community-acquired pneumonia requiring antibiotic therapy and oxygen support. Despite improvement in his overall condition, his blood pressure was consistently > 160/90 mm Hg during his hospitalization. He was treated with lisinopril 10 mg po daily in addition to his home medications, which helped achieve recommended blood pressure goals.
Prior to discharge, his blood pressure was noted to be 108/62 mm Hg. He asks if it is necessary to continue this new blood pressure medicine, as his home blood pressure readings had been within the goal set by his primary care physician. Should you continue this new antihypertensive agent at discharge?
Outpatient antihypertensive medication regimens are commonly intensified at hospital discharge in response to transient short-term elevations in blood pressure during inpatient encounters for noncardiac conditions.1,2 This is typically a reflexive response during a hospitalization, despite the unknown long-term, patient-oriented clinical outcomes. These short-term, in-hospital blood pressure elevations may be due to numerous temporary causes, such as stress/anxiety, a pain response, agitation, a medication adverse effect, or volume overload.3
The transition from inpatient to outpatient care is a high-risk period, especially for older adults, as functional status is generally worse at hospital discharge than prehospitalization baseline.4 To compound this problem, adverse drug reactions are a common cause of hospitalization for older adults. Changing blood pressure medications in response to acute physiologic changes during illness may contribute to patient harm. Although observational studies of adverse drug reactions related to blood pressure medications are numerous, researchers have only evaluated adverse drug reactions pertaining to hospital admissions.5-8 This study sought to evaluate the clinical outcomes associated with intensification of antihypertensive regimens at discharge among older adults.
STUDY SUMMARY
Increased risk of readmission, adverse events after intensification at discharge
This retrospective cohort study, which was conducted across multiple
Antihypertensive medication changes at discharge were evaluated using information pulled from VHA pharmacies, combined with clinical data merged from VHA and Medicare claims. Intensification was defined as either adding a new blood pressure medication or a dose increase of more than 20% on a previously prescribed antihypertensive medication. Patients were excluded if they were discharged with a secondary diagnosis that required modifications to a blood pressure medication (such as atrial fibrillation, acute coronary syndrome, or stroke), were hospitalized in the previous 30 days, were admitted from a skilled nursing facility, or received more than 20% of their care (including filling prescriptions) outside the VHA system.
Primary outcomes included hospital readmission or SAEs (falls, syncope, hypotension, serious electrolyte abnormalities, or acute kidney injury) within 30 days or having a cardiovascular event within 1 year of hospital discharge. Secondary outcomes included the change in systolic blood pressure (SBP) within 1 year after discharge. Propensity score matching was used as a balancing factor to create a matched-pairs cohort to compare those receiving blood pressure medication intensification at hospital discharge with those who did not.
Continue to: Intensification of the blood pressure...
Intensification of the blood pressure regimen at hospital discharge was associated with an increased risk in 30-day hospital readmission (hazard ratio [HR] = 1.23; 95% CI, 1.07–1.42; number needed to harm [NNH] = 27) and SAEs (HR = 1.41; 95% CI, 1.06–1.88; NNH = 63). There was no associated reduction in cardiovascular events (HR = 1.18; 95% CI, 0.99–1.40) or change in mean SBP within 1 year after hospital discharge in those who received intensification vs those who did not (mean BP, 134.7 vs 134.4 mm Hg; difference-in-differences estimate = 0.2 mm Hg; 95% CI, −2.0 to 2.4 mm Hg).
WHAT’S NEW
First study on outcomes related to HTN med changes at hospital discharge
This well-designed, retrospective cohort study provides important clinical data to help guide inpatient blood pressure management decisions for patients with noncardiac conditions. No clinical trials up to that time had assessed patient-oriented outcomes when antihypertensive medication regimens were intensified at hospital discharge.
CAVEATS
Study population: Primarily older men with noncardiac conditions
Selected populations benefit from intensive blood pressure control based on specific risk factors and medical conditions. In patients at high risk for cardiovascular disease, without a history of stroke or diabetes, intensive blood pressure control (SBP < 120 mm Hg) improves cardiovascular outcomes and overall survival compared with standard therapy (SBP < 140 mm Hg).9 This retrospective cohort study involved mainly elderly male patients with noncardiac conditions. The study also excluded patients with a secondary diagnosis requiring modifications to an antihypertensive regimen, such as atrial fibrillation, acute coronary syndrome, or cerebrovascular accident. Thus, the findings may not be applicable to these patient populations.
CHALLENGES TO IMPLEMENTATION
Clinicians will need to address individual needs
Physicians have to balance various antihypertensive management strategies, as competing medical specialty society guidelines recommend differing targets for optimal blood pressure control. Given the concern for medicolegal liability and potential harms of therapeutic inertia, inpatient physicians must consider whether hospitalization is the best time to alter medications for long-term outpatient blood pressure control. Finally, the decision to leave blood pressure management to outpatient physicians assumes the patient has a continuity relationship with a primary care medical home.
ACKNOWLEDGEMENT
The PURLs Surveillance System was supported in part by Grant Number UL1RR024999 from the National Center for Research Resources, a Clinical Translational Science Award to the University of Chicago. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center for Research Resources or the National Institutes of Health.
1. Anderson TS, Jing B, Auerbach A, et al. Clinical outcomes after intensifying antihypertensive medication regimens among older adults at hospital discharge. JAMA Intern Med. 2019;179:1528-1536.
2. Harris CM, Sridharan A, Landis R, et al. What happens to the medication regimens of older adults during and after an acute hospitalization? J Patient Saf. 2013;9:150-153.
3. Aung WM, Menon SV, Materson BJ. Management of hypertension in hospitalized patients. Hosp Pract (1995). 2015;43:101-106.
4. Covinsky KE, Palmer RM, Fortinsky RH, et al. Loss of independence in activities of daily living in older adults hospitalized with medical illnesses: increased vulnerability with age. J Am Geriatr Soc. 2003;51:451-458.
5. Omer HMRB, Hodson J, Pontefract SK, et al. Inpatient falls in older adults: a cohort study of antihypertensive prescribing pre- and post-fall. BMC Geriatr. 2018;18:58.
6. Alhawassi TM, Krass I, Pont LG. Antihypertensive-related adverse drug reactions among older hospitalized adults. Int J Clin Pharm. 2018;40:428-435.
7. Passarelli MCG, Jacob-Filho W, Figueras A. Adverse drug reactions in an elderly hospitalised population: inappropriate prescription is a leading cause. Drugs Aging. 2005;22:767-777.
8. Beckett NS, Peters R, Fletcher AE, et al; HYVET Study Group. Treatment of hypertension in patients 80 years of age or older. N Engl J Med. 2008;358:1887-1898.
9. SPRINT Research Group; Wright JT Jr, Williamson JD, Whelton PK, et al. A randomized trial of intensive versus standard blood-pressure control. N Engl J Med. 2015;373:2103-2116. Published correction appears in N Engl J Med. 2017;377:2506.
1. Anderson TS, Jing B, Auerbach A, et al. Clinical outcomes after intensifying antihypertensive medication regimens among older adults at hospital discharge. JAMA Intern Med. 2019;179:1528-1536.
2. Harris CM, Sridharan A, Landis R, et al. What happens to the medication regimens of older adults during and after an acute hospitalization? J Patient Saf. 2013;9:150-153.
3. Aung WM, Menon SV, Materson BJ. Management of hypertension in hospitalized patients. Hosp Pract (1995). 2015;43:101-106.
4. Covinsky KE, Palmer RM, Fortinsky RH, et al. Loss of independence in activities of daily living in older adults hospitalized with medical illnesses: increased vulnerability with age. J Am Geriatr Soc. 2003;51:451-458.
5. Omer HMRB, Hodson J, Pontefract SK, et al. Inpatient falls in older adults: a cohort study of antihypertensive prescribing pre- and post-fall. BMC Geriatr. 2018;18:58.
6. Alhawassi TM, Krass I, Pont LG. Antihypertensive-related adverse drug reactions among older hospitalized adults. Int J Clin Pharm. 2018;40:428-435.
7. Passarelli MCG, Jacob-Filho W, Figueras A. Adverse drug reactions in an elderly hospitalised population: inappropriate prescription is a leading cause. Drugs Aging. 2005;22:767-777.
8. Beckett NS, Peters R, Fletcher AE, et al; HYVET Study Group. Treatment of hypertension in patients 80 years of age or older. N Engl J Med. 2008;358:1887-1898.
9. SPRINT Research Group; Wright JT Jr, Williamson JD, Whelton PK, et al. A randomized trial of intensive versus standard blood-pressure control. N Engl J Med. 2015;373:2103-2116. Published correction appears in N Engl J Med. 2017;377:2506.
PRACTICE CHANGER
Avoid intensifying antihypertensive medication regimens at hospital discharge in older adults; making such changes increases the risk of serious adverse events (SAEs) and hospital readmission within 30 days without reducing the risk of serious cardiovascular events at 1 year post discharge.
STRENGTH OF RECOMMENDATION
B: Based on a large retrospective cohort study evaluating patient-oriented outcomes.1
Anderson TS, Jing B, Auerbach A, et al. Clinical outcomes after intensifying antihypertensive medication regimens among older adults at hospital discharge. JAMA Intern Med. 2019;179:1528-1536.
Call to Action: Multidisciplinary panel urges coordinated care for ‘NASH epidemic’
A multidisciplinary panel of U.S. experts released a “Call to Action” for improved screening, diagnosis, and treatment of patients with nonalcoholic steatohepatitis (NASH) and nonalcoholic fatty liver disease (NAFLD) on July 26, an effort organized by the American Gastroenterological Association in collaboration with seven other U.S. medical organizations including several endocrinology groups.
The published statement, “Preparing for the NASH Epidemic: A Call to Action,” proposes several urgent steps for the U.S. clinical community to provide better-focused and better-coordinated care for patients at risk for developing or having NAFLD or NASH, particularly among “emerging” at-risk cohorts such as patients with diabetes and obesity. It appears in the journals Gastroenterology, Diabetes Care, Metabolism: Clinical and Experimental, and Obesity.
The statement’s central pitch is that improvements in care won’t be possible unless the several medical specialties that deal with affected or at-risk patients stop working “in separate silos,” and instead create “a collective action plan,” and also organize multidisciplinary teams that “integrate primary care, hepatology, obesity medicine, endocrinology, and diabetology via well-defined care pathways.”
“The overarching goal” is a “unified, international public health response to NAFLD and NASH,” said the statement, which stemmed from a conference held in July 2020 that included representatives from not only the lead gastroenterology group but also the American Diabetes Association, the American Association for the Study of Liver Diseases, the American Association of Clinical Endocrinologists, The Endocrine Society, The American Academy of Family Physicians, The Obesity Society, and the American College of Osteopathic Family Physicians.
The statement cites sobering prevalence numbers, with estimates that NAFLD exists in more than half the patients with type 2 diabetes, while NASH affects about a third, rates that translate into many millions of affected Americans, given recent estimates that the U.S. prevalence of type 2 diabetes exceeds 30 million people. And the numbers continue to rise along with increases in the prevalence of obesity and type 2 diabetes.
“It’s an enormously common disease, and there are not enough gastroenterologists, to say nothing of hepatologists, to care for every patient with NAFLD,” said Anna Mae Diehl, MD, a gastroenterologist and professor at Duke University in Durham, N.C., who was not involved with the conference nor in writing the statement.
Clinical care pathways coming soon
Another key part of this initiative is development of clinical care pathways that will have “careful explication of each step in screening, diagnosis, and treatment,” and will be designed to inform the practice of primary care physicians (PCPs) as well as clinicians from the various specialties that deal with these patients.
The clinical care pathways are on track to come out later in 2021, said Fasiha Kanwal, MD, a professor and chief of gastroenterology at Baylor College of Medicine in Houston, and lead author on the Call to Action document.
“The Pathways will include practical recommendations about whom to screen and when to refer, and the criteria primary care physicians can use for diagnosis and risk stratification,” Dr. Kanwal said in an interview. “Patients can benefit from a standardized approach.”
The new document also includes results from a recent survey about NAFLD and NASH management completed by 751 U.S. physicians, including 401 (53%) primary care physicians, 175 gastroenterologists, (23%) and 175 endocrinologists (23%; percentages total 99% because of rounding).
The results showed “significant gaps in knowledge about whom to screen and how to diagnose and treat patients at high risk for NASH,” concluded the statement’s authors. Barely more than a third of the respondents knew that almost all patients with severe obesity likely have NAFLD, and fewer than half the endocrinologists and the primary care physicians appreciated that NAFLD is very common among patients with type 2 diabetes.
‘Understanding of NAFLD is not there’
“I applaud this effort that calls attention to an emerging public health problem. This paper and survey are great ideas. The findings are not surprising, but they’re important,” said Dr. Diehl said in an interview. “Much more needs to be done” including changes in social behavior and government policies.
“The public’s understanding of NAFLD is not there,” and many physicians also have an incomplete understanding of NAFLD and more serious stages of metabolic liver disease. “Physicians know that patients with obesity are at risk for heart disease, diabetes, and stroke, but they may not always be aware that these patients can also have cirrhosis,” noted Dr. Diehl, who published in 2019 a call to action for NAFLD of her own with some associates.
“My referrals are fueled by primary care physicians who recognize patients with significant liver disease. It would be great to outline recommended practice; I have no doubt that providers will embrace this,” as well as the broader concept of multidisciplinary teams, another focus of the statement. Dr. Diehl cited the “Cancer Center model,” where an oncologist takes primary responsibility for caring for a cancer patient while coordinating care with other specialists, an approach facilitated by EMRs that allow seamless data and chart sharing and something that many health systems have either already adopted or are moving toward.
She said the NASH Call to Action may help catalyze broader application of this model to many more patients with NAFLD or NASH, and noted that some U.S. centers already use this approach – including Dr. Diehl’s program at Duke – which brings together her gastroenterology colleagues with cardiologists, radiologists, endocrinologists, and bariatric surgeons. But she noted that for most patients with metabolic liver disease, the hub clinician needs to be a PCP, especially for patients with earlier-stage disease, because the number of affected patients is so huge.
“Key steps toward establishing such teams include establishing protocols for risk stratification and referral, definition of roles and responsibilities, and buy-in from institutions and payers. Clearly a lot of work needs to occur to get to these multidisciplinary teams,” said Dr. Kanwal.
Ralph A. DeFronzo, MD, professor and deputy director of the Texas Diabetes Institute at UT Health San Antonio, who was not involved with the conference or statement, had a different take on what the future of NASH and NAFLD care may look like.
“Endocrinologists, hepatologists, and obesity experts will work within their individual specialties to diagnose and manage NASH,” he said in an interview. But he acknowledged that “an integrated effort by specialists would be important” to help “primary care physicians who are less familiar with the disease.”
Controversy over pioglitazone?
Dr. DeFronzo endorsed development of clinical care pathways as “important,” but also as a potential source of “controversy, especially with respect to treatment.”
The Call to Action statement cites lifestyle-based therapies, such as an appropriate diet; regular, moderate exercise; and elimination when possible of obesogenic medications as cornerstone interventions for patients with NAFLD or early-stage NASH, interventions that can be prescribed by PCPs. For patients with NASH and stage 2 or worse fibrosis, the statement endorses liver-directed pharmacotherapy. While noting that no agents currently carry a Food and Drug Administration–approved indication for treating NASH, the statement cites evidence that 800 IU/day of vitamin E improves steatosis in patients with NASH but not type 2 diabetes.
For patients with type 2 diabetes, the statement notes that results from five randomized trials indicated that pioglitazone could reverse steatohepatitis, findings that led to its recommendation in guidelines from a modest circle of medical groups, including the American Association for the Study of Liver Diseases and the European Association for the Study of Diabetes. However, the 2021 Standards of Medical Care in Diabetes from the American Diabetes Association gives these agents limited endorsement, saying: “Pioglitazone, vitamin E treatment, and liraglutide treatment of biopsy-proven nonalcoholic steatohepatitis have each been shown to improve liver histology, but effects on longer-term clinical outcomes are not known.”
“The strongest evidence by far is for pioglitazone for treating NAFLD and NASH,” said Dr. DeFronzo, a vocal proponent of the drug for this indication. But he added that “hepatologists don’t feel comfortable with drugs from the thiazolidinedione class,” which includes pioglitazone.
“We don’t yet know how to optimally configure the health system for NAFLD and NASH to make it more efficient and helpful to patients, but models exist, and the approach is evolving,” said Dr. Diehl.
Dr. Diehl and Dr. Kanwal had no relevant disclosures. Dr. DeFronzo has been a speaker on behalf of AstraZeneca and Novo Nordisk, has been an adviser to AstraZeneca, Boehringer Ingelheim, Intarcia, and Janssen, and has received research funding from AstraZeneca, Janssen and Merck.
A multidisciplinary panel of U.S. experts released a “Call to Action” for improved screening, diagnosis, and treatment of patients with nonalcoholic steatohepatitis (NASH) and nonalcoholic fatty liver disease (NAFLD) on July 26, an effort organized by the American Gastroenterological Association in collaboration with seven other U.S. medical organizations including several endocrinology groups.
The published statement, “Preparing for the NASH Epidemic: A Call to Action,” proposes several urgent steps for the U.S. clinical community to provide better-focused and better-coordinated care for patients at risk for developing or having NAFLD or NASH, particularly among “emerging” at-risk cohorts such as patients with diabetes and obesity. It appears in the journals Gastroenterology, Diabetes Care, Metabolism: Clinical and Experimental, and Obesity.
The statement’s central pitch is that improvements in care won’t be possible unless the several medical specialties that deal with affected or at-risk patients stop working “in separate silos,” and instead create “a collective action plan,” and also organize multidisciplinary teams that “integrate primary care, hepatology, obesity medicine, endocrinology, and diabetology via well-defined care pathways.”
“The overarching goal” is a “unified, international public health response to NAFLD and NASH,” said the statement, which stemmed from a conference held in July 2020 that included representatives from not only the lead gastroenterology group but also the American Diabetes Association, the American Association for the Study of Liver Diseases, the American Association of Clinical Endocrinologists, The Endocrine Society, The American Academy of Family Physicians, The Obesity Society, and the American College of Osteopathic Family Physicians.
The statement cites sobering prevalence numbers, with estimates that NAFLD exists in more than half the patients with type 2 diabetes, while NASH affects about a third, rates that translate into many millions of affected Americans, given recent estimates that the U.S. prevalence of type 2 diabetes exceeds 30 million people. And the numbers continue to rise along with increases in the prevalence of obesity and type 2 diabetes.
“It’s an enormously common disease, and there are not enough gastroenterologists, to say nothing of hepatologists, to care for every patient with NAFLD,” said Anna Mae Diehl, MD, a gastroenterologist and professor at Duke University in Durham, N.C., who was not involved with the conference nor in writing the statement.
Clinical care pathways coming soon
Another key part of this initiative is development of clinical care pathways that will have “careful explication of each step in screening, diagnosis, and treatment,” and will be designed to inform the practice of primary care physicians (PCPs) as well as clinicians from the various specialties that deal with these patients.
The clinical care pathways are on track to come out later in 2021, said Fasiha Kanwal, MD, a professor and chief of gastroenterology at Baylor College of Medicine in Houston, and lead author on the Call to Action document.
“The Pathways will include practical recommendations about whom to screen and when to refer, and the criteria primary care physicians can use for diagnosis and risk stratification,” Dr. Kanwal said in an interview. “Patients can benefit from a standardized approach.”
The new document also includes results from a recent survey about NAFLD and NASH management completed by 751 U.S. physicians, including 401 (53%) primary care physicians, 175 gastroenterologists, (23%) and 175 endocrinologists (23%; percentages total 99% because of rounding).
The results showed “significant gaps in knowledge about whom to screen and how to diagnose and treat patients at high risk for NASH,” concluded the statement’s authors. Barely more than a third of the respondents knew that almost all patients with severe obesity likely have NAFLD, and fewer than half the endocrinologists and the primary care physicians appreciated that NAFLD is very common among patients with type 2 diabetes.
‘Understanding of NAFLD is not there’
“I applaud this effort that calls attention to an emerging public health problem. This paper and survey are great ideas. The findings are not surprising, but they’re important,” said Dr. Diehl said in an interview. “Much more needs to be done” including changes in social behavior and government policies.
“The public’s understanding of NAFLD is not there,” and many physicians also have an incomplete understanding of NAFLD and more serious stages of metabolic liver disease. “Physicians know that patients with obesity are at risk for heart disease, diabetes, and stroke, but they may not always be aware that these patients can also have cirrhosis,” noted Dr. Diehl, who published in 2019 a call to action for NAFLD of her own with some associates.
“My referrals are fueled by primary care physicians who recognize patients with significant liver disease. It would be great to outline recommended practice; I have no doubt that providers will embrace this,” as well as the broader concept of multidisciplinary teams, another focus of the statement. Dr. Diehl cited the “Cancer Center model,” where an oncologist takes primary responsibility for caring for a cancer patient while coordinating care with other specialists, an approach facilitated by EMRs that allow seamless data and chart sharing and something that many health systems have either already adopted or are moving toward.
She said the NASH Call to Action may help catalyze broader application of this model to many more patients with NAFLD or NASH, and noted that some U.S. centers already use this approach – including Dr. Diehl’s program at Duke – which brings together her gastroenterology colleagues with cardiologists, radiologists, endocrinologists, and bariatric surgeons. But she noted that for most patients with metabolic liver disease, the hub clinician needs to be a PCP, especially for patients with earlier-stage disease, because the number of affected patients is so huge.
“Key steps toward establishing such teams include establishing protocols for risk stratification and referral, definition of roles and responsibilities, and buy-in from institutions and payers. Clearly a lot of work needs to occur to get to these multidisciplinary teams,” said Dr. Kanwal.
Ralph A. DeFronzo, MD, professor and deputy director of the Texas Diabetes Institute at UT Health San Antonio, who was not involved with the conference or statement, had a different take on what the future of NASH and NAFLD care may look like.
“Endocrinologists, hepatologists, and obesity experts will work within their individual specialties to diagnose and manage NASH,” he said in an interview. But he acknowledged that “an integrated effort by specialists would be important” to help “primary care physicians who are less familiar with the disease.”
Controversy over pioglitazone?
Dr. DeFronzo endorsed development of clinical care pathways as “important,” but also as a potential source of “controversy, especially with respect to treatment.”
The Call to Action statement cites lifestyle-based therapies, such as an appropriate diet; regular, moderate exercise; and elimination when possible of obesogenic medications as cornerstone interventions for patients with NAFLD or early-stage NASH, interventions that can be prescribed by PCPs. For patients with NASH and stage 2 or worse fibrosis, the statement endorses liver-directed pharmacotherapy. While noting that no agents currently carry a Food and Drug Administration–approved indication for treating NASH, the statement cites evidence that 800 IU/day of vitamin E improves steatosis in patients with NASH but not type 2 diabetes.
For patients with type 2 diabetes, the statement notes that results from five randomized trials indicated that pioglitazone could reverse steatohepatitis, findings that led to its recommendation in guidelines from a modest circle of medical groups, including the American Association for the Study of Liver Diseases and the European Association for the Study of Diabetes. However, the 2021 Standards of Medical Care in Diabetes from the American Diabetes Association gives these agents limited endorsement, saying: “Pioglitazone, vitamin E treatment, and liraglutide treatment of biopsy-proven nonalcoholic steatohepatitis have each been shown to improve liver histology, but effects on longer-term clinical outcomes are not known.”
“The strongest evidence by far is for pioglitazone for treating NAFLD and NASH,” said Dr. DeFronzo, a vocal proponent of the drug for this indication. But he added that “hepatologists don’t feel comfortable with drugs from the thiazolidinedione class,” which includes pioglitazone.
“We don’t yet know how to optimally configure the health system for NAFLD and NASH to make it more efficient and helpful to patients, but models exist, and the approach is evolving,” said Dr. Diehl.
Dr. Diehl and Dr. Kanwal had no relevant disclosures. Dr. DeFronzo has been a speaker on behalf of AstraZeneca and Novo Nordisk, has been an adviser to AstraZeneca, Boehringer Ingelheim, Intarcia, and Janssen, and has received research funding from AstraZeneca, Janssen and Merck.
A multidisciplinary panel of U.S. experts released a “Call to Action” for improved screening, diagnosis, and treatment of patients with nonalcoholic steatohepatitis (NASH) and nonalcoholic fatty liver disease (NAFLD) on July 26, an effort organized by the American Gastroenterological Association in collaboration with seven other U.S. medical organizations including several endocrinology groups.
The published statement, “Preparing for the NASH Epidemic: A Call to Action,” proposes several urgent steps for the U.S. clinical community to provide better-focused and better-coordinated care for patients at risk for developing or having NAFLD or NASH, particularly among “emerging” at-risk cohorts such as patients with diabetes and obesity. It appears in the journals Gastroenterology, Diabetes Care, Metabolism: Clinical and Experimental, and Obesity.
The statement’s central pitch is that improvements in care won’t be possible unless the several medical specialties that deal with affected or at-risk patients stop working “in separate silos,” and instead create “a collective action plan,” and also organize multidisciplinary teams that “integrate primary care, hepatology, obesity medicine, endocrinology, and diabetology via well-defined care pathways.”
“The overarching goal” is a “unified, international public health response to NAFLD and NASH,” said the statement, which stemmed from a conference held in July 2020 that included representatives from not only the lead gastroenterology group but also the American Diabetes Association, the American Association for the Study of Liver Diseases, the American Association of Clinical Endocrinologists, The Endocrine Society, The American Academy of Family Physicians, The Obesity Society, and the American College of Osteopathic Family Physicians.
The statement cites sobering prevalence numbers, with estimates that NAFLD exists in more than half the patients with type 2 diabetes, while NASH affects about a third, rates that translate into many millions of affected Americans, given recent estimates that the U.S. prevalence of type 2 diabetes exceeds 30 million people. And the numbers continue to rise along with increases in the prevalence of obesity and type 2 diabetes.
“It’s an enormously common disease, and there are not enough gastroenterologists, to say nothing of hepatologists, to care for every patient with NAFLD,” said Anna Mae Diehl, MD, a gastroenterologist and professor at Duke University in Durham, N.C., who was not involved with the conference nor in writing the statement.
Clinical care pathways coming soon
Another key part of this initiative is development of clinical care pathways that will have “careful explication of each step in screening, diagnosis, and treatment,” and will be designed to inform the practice of primary care physicians (PCPs) as well as clinicians from the various specialties that deal with these patients.
The clinical care pathways are on track to come out later in 2021, said Fasiha Kanwal, MD, a professor and chief of gastroenterology at Baylor College of Medicine in Houston, and lead author on the Call to Action document.
“The Pathways will include practical recommendations about whom to screen and when to refer, and the criteria primary care physicians can use for diagnosis and risk stratification,” Dr. Kanwal said in an interview. “Patients can benefit from a standardized approach.”
The new document also includes results from a recent survey about NAFLD and NASH management completed by 751 U.S. physicians, including 401 (53%) primary care physicians, 175 gastroenterologists, (23%) and 175 endocrinologists (23%; percentages total 99% because of rounding).
The results showed “significant gaps in knowledge about whom to screen and how to diagnose and treat patients at high risk for NASH,” concluded the statement’s authors. Barely more than a third of the respondents knew that almost all patients with severe obesity likely have NAFLD, and fewer than half the endocrinologists and the primary care physicians appreciated that NAFLD is very common among patients with type 2 diabetes.
‘Understanding of NAFLD is not there’
“I applaud this effort that calls attention to an emerging public health problem. This paper and survey are great ideas. The findings are not surprising, but they’re important,” said Dr. Diehl said in an interview. “Much more needs to be done” including changes in social behavior and government policies.
“The public’s understanding of NAFLD is not there,” and many physicians also have an incomplete understanding of NAFLD and more serious stages of metabolic liver disease. “Physicians know that patients with obesity are at risk for heart disease, diabetes, and stroke, but they may not always be aware that these patients can also have cirrhosis,” noted Dr. Diehl, who published in 2019 a call to action for NAFLD of her own with some associates.
“My referrals are fueled by primary care physicians who recognize patients with significant liver disease. It would be great to outline recommended practice; I have no doubt that providers will embrace this,” as well as the broader concept of multidisciplinary teams, another focus of the statement. Dr. Diehl cited the “Cancer Center model,” where an oncologist takes primary responsibility for caring for a cancer patient while coordinating care with other specialists, an approach facilitated by EMRs that allow seamless data and chart sharing and something that many health systems have either already adopted or are moving toward.
She said the NASH Call to Action may help catalyze broader application of this model to many more patients with NAFLD or NASH, and noted that some U.S. centers already use this approach – including Dr. Diehl’s program at Duke – which brings together her gastroenterology colleagues with cardiologists, radiologists, endocrinologists, and bariatric surgeons. But she noted that for most patients with metabolic liver disease, the hub clinician needs to be a PCP, especially for patients with earlier-stage disease, because the number of affected patients is so huge.
“Key steps toward establishing such teams include establishing protocols for risk stratification and referral, definition of roles and responsibilities, and buy-in from institutions and payers. Clearly a lot of work needs to occur to get to these multidisciplinary teams,” said Dr. Kanwal.
Ralph A. DeFronzo, MD, professor and deputy director of the Texas Diabetes Institute at UT Health San Antonio, who was not involved with the conference or statement, had a different take on what the future of NASH and NAFLD care may look like.
“Endocrinologists, hepatologists, and obesity experts will work within their individual specialties to diagnose and manage NASH,” he said in an interview. But he acknowledged that “an integrated effort by specialists would be important” to help “primary care physicians who are less familiar with the disease.”
Controversy over pioglitazone?
Dr. DeFronzo endorsed development of clinical care pathways as “important,” but also as a potential source of “controversy, especially with respect to treatment.”
The Call to Action statement cites lifestyle-based therapies, such as an appropriate diet; regular, moderate exercise; and elimination when possible of obesogenic medications as cornerstone interventions for patients with NAFLD or early-stage NASH, interventions that can be prescribed by PCPs. For patients with NASH and stage 2 or worse fibrosis, the statement endorses liver-directed pharmacotherapy. While noting that no agents currently carry a Food and Drug Administration–approved indication for treating NASH, the statement cites evidence that 800 IU/day of vitamin E improves steatosis in patients with NASH but not type 2 diabetes.
For patients with type 2 diabetes, the statement notes that results from five randomized trials indicated that pioglitazone could reverse steatohepatitis, findings that led to its recommendation in guidelines from a modest circle of medical groups, including the American Association for the Study of Liver Diseases and the European Association for the Study of Diabetes. However, the 2021 Standards of Medical Care in Diabetes from the American Diabetes Association gives these agents limited endorsement, saying: “Pioglitazone, vitamin E treatment, and liraglutide treatment of biopsy-proven nonalcoholic steatohepatitis have each been shown to improve liver histology, but effects on longer-term clinical outcomes are not known.”
“The strongest evidence by far is for pioglitazone for treating NAFLD and NASH,” said Dr. DeFronzo, a vocal proponent of the drug for this indication. But he added that “hepatologists don’t feel comfortable with drugs from the thiazolidinedione class,” which includes pioglitazone.
“We don’t yet know how to optimally configure the health system for NAFLD and NASH to make it more efficient and helpful to patients, but models exist, and the approach is evolving,” said Dr. Diehl.
Dr. Diehl and Dr. Kanwal had no relevant disclosures. Dr. DeFronzo has been a speaker on behalf of AstraZeneca and Novo Nordisk, has been an adviser to AstraZeneca, Boehringer Ingelheim, Intarcia, and Janssen, and has received research funding from AstraZeneca, Janssen and Merck.