User login
Don’t miss cardiovascular risk factors in transgender patients
Cardiovascular disease risk is elevated among transgender individuals seeking gender-affirming hormone therapy, according to a retrospective study in 427 patients.
The transgender population often experiences socioeconomic and health disparities, including reduced access to care, Kara J. Denby, MD, said in an interview.
Previous research suggests that the use of gender-affirming hormone therapy (GAHT) may place transgender persons at increased cardiovascular risk, she said.
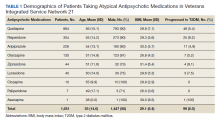
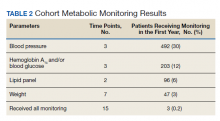
To identify the potential risk for transgender individuals, the researchers identified baseline cardiovascular risk in patients who had not yet undergone GAHT. Study participants were enrolled in a multidisciplinary transgender program, and the researchers collected data on demographics, medical history, vitals, medications, and laboratory results. The average age of the participants was 26 years, 172 identified as men, 236 as women, and 20 as nonbinary.
Overall, 55% of the participants had a chronic medical condition at baseline. Of these, 74 patients had hypertension, 41 had hyperlipidemia, 2 had a history of stroke, 7 had coronary artery disease, and 4 had chronic obstructive pulmonary disease.
For all patients who did not have documented atherosclerotic cardiovascular disease, their American College of Cardiology/American Heart Association ASCVD and QRISK3 risk scores were calculated. “The incidence of undiagnosed hypertension and hyperlipidemia was 6.8% and 11.3% respectively, and of these cases, only 64% and 24% were on appropriate therapies,” noted Dr. Denby of the Cleveland (Ohio) Clinic.
She reported the results Nov. 13 in a presentation at the at the virtual American Heart Association scientific sessions.
The findings were limited by the observational nature of the study.
However, the results suggest that transgender patients “appear to be at higher risk than their age-matched historical cohorts regardless of gender,” said Dr. Denby. More research is needed, but cardiovascular disease–prevention efforts may be inadequate in the transgender population given the elevated risk observed in this study, she concluded.
Growing transgender population is medically underserved
The transgender population is growing in the United States and internationally, said Dr. Denby. “This group has a history of being marginalized as a result of their transgender status with socioeconomic and health repercussions,” she said. “It is well known that transgender patients are less likely to have access to health care or utilize health care for a variety of reasons, including stigma and fear of mistreatment. This often leads transgender individuals to present to care late in disease processes which makes their disease harder to treat and often leads to emergent medical conditions,” she added.
“Transgender men and women are at high risk for cardiovascular disease and often aren’t screened at recommended intervals because of decreased health care use compared to their cisgender counterparts,” she said. “This may lead to untreated diseases that make them even more likely to suffer poor health outcomes.”
The current study is important because there are “almost no prior data regarding the cardiovascular health status of this population prior to gender-affirming care,” Dr. Denby emphasized. “There are data that gay, lesbian, and bisexual individuals are at higher risk for poor cardiovascular outcomes, but the same data are lacking in the transgender group,” she said.
“As transgender individuals have frequent physician visits while on hormonal therapy, this seems like the opportune time to screen for cardiovascular risk factors and treat previously undiagnosed diseases that can lead to poor health outcomes in the future,” Dr. Denby explained. “If we are able to intervene at an earlier age, perhaps we can help prevent poor health outcomes down the road,” she said.
Additional research can inform practice
Dr. Denby said she was not surprised by the findings. “This is a very high-risk population that often doesn’t follow closely in the health care system,” she said. “These data are very important in thinking holistically about transgender patients.” Clinicians can “use the opportunities we have when they present for gender-affirming care to optimize their overall health status, promote long-term health, and reduce the risks associated with hormonal therapy and gender-affirming surgeries,” she noted. “We hope to use this information to change our practice at the Cleveland Clinic and nationally as well. Transgender patients should be screened and aggressively treated for cardiovascular disease and risk factors,” she said.
Key barriers to overcome include determining the best way to reach out to transgender individuals and then making them feel comfortable in the clinical setting, Dr. Denby said. “This means that we must set up clinics that are approachable and safe for all comers. The lack of laws in many states that protect this vulnerable population also contributes to lack of access to care,” she added.
“We hope to continue research in this arena about how to effectively screen and treat transgender patients as they present to care, not only in the transgender clinic, but also to primary care providers (ob.gyn., internal medicine, family medicine, pediatrics) who also care for this population” since no specific guidelines currently exist to direct the screening for cardiovascular patients in particular, she said.
Findings offer foundation for LGBTQ cardiovascular studies
“This [study] provides us with a good rationale for why we should be considering cardiovascular health in transgender adults,” Billy A. Caceres, PhD, RN, of Columbia University School of Nursing, New York, said in an interview. “It is largely descriptive, but I think that that’s a good step in terms of at least understanding the magnitude of this problem. In addition, I think that what this abstract might do is help lead to future research that examines potentially the associations between not only gender-affirming hormone therapies but other potential social determinants like discrimination or poverty on the cardiovascular health of transgender people,” he noted.
Dr. Caceres served as chair of the writing group for the recent American Heart Association Scientific Statement: LGBTQ Heart Health published in Circulation. He had no financial conflicts to disclose.
The study received no outside funding. Dr. Denby had no financial conflicts to disclose.
SOURCE: Denby KJ et al. AHA 2020, Presentation P2274.
Cardiovascular disease risk is elevated among transgender individuals seeking gender-affirming hormone therapy, according to a retrospective study in 427 patients.
The transgender population often experiences socioeconomic and health disparities, including reduced access to care, Kara J. Denby, MD, said in an interview.
Previous research suggests that the use of gender-affirming hormone therapy (GAHT) may place transgender persons at increased cardiovascular risk, she said.
To identify the potential risk for transgender individuals, the researchers identified baseline cardiovascular risk in patients who had not yet undergone GAHT. Study participants were enrolled in a multidisciplinary transgender program, and the researchers collected data on demographics, medical history, vitals, medications, and laboratory results. The average age of the participants was 26 years, 172 identified as men, 236 as women, and 20 as nonbinary.
Overall, 55% of the participants had a chronic medical condition at baseline. Of these, 74 patients had hypertension, 41 had hyperlipidemia, 2 had a history of stroke, 7 had coronary artery disease, and 4 had chronic obstructive pulmonary disease.
For all patients who did not have documented atherosclerotic cardiovascular disease, their American College of Cardiology/American Heart Association ASCVD and QRISK3 risk scores were calculated. “The incidence of undiagnosed hypertension and hyperlipidemia was 6.8% and 11.3% respectively, and of these cases, only 64% and 24% were on appropriate therapies,” noted Dr. Denby of the Cleveland (Ohio) Clinic.
She reported the results Nov. 13 in a presentation at the at the virtual American Heart Association scientific sessions.
The findings were limited by the observational nature of the study.
However, the results suggest that transgender patients “appear to be at higher risk than their age-matched historical cohorts regardless of gender,” said Dr. Denby. More research is needed, but cardiovascular disease–prevention efforts may be inadequate in the transgender population given the elevated risk observed in this study, she concluded.
Growing transgender population is medically underserved
The transgender population is growing in the United States and internationally, said Dr. Denby. “This group has a history of being marginalized as a result of their transgender status with socioeconomic and health repercussions,” she said. “It is well known that transgender patients are less likely to have access to health care or utilize health care for a variety of reasons, including stigma and fear of mistreatment. This often leads transgender individuals to present to care late in disease processes which makes their disease harder to treat and often leads to emergent medical conditions,” she added.
“Transgender men and women are at high risk for cardiovascular disease and often aren’t screened at recommended intervals because of decreased health care use compared to their cisgender counterparts,” she said. “This may lead to untreated diseases that make them even more likely to suffer poor health outcomes.”
The current study is important because there are “almost no prior data regarding the cardiovascular health status of this population prior to gender-affirming care,” Dr. Denby emphasized. “There are data that gay, lesbian, and bisexual individuals are at higher risk for poor cardiovascular outcomes, but the same data are lacking in the transgender group,” she said.
“As transgender individuals have frequent physician visits while on hormonal therapy, this seems like the opportune time to screen for cardiovascular risk factors and treat previously undiagnosed diseases that can lead to poor health outcomes in the future,” Dr. Denby explained. “If we are able to intervene at an earlier age, perhaps we can help prevent poor health outcomes down the road,” she said.
Additional research can inform practice
Dr. Denby said she was not surprised by the findings. “This is a very high-risk population that often doesn’t follow closely in the health care system,” she said. “These data are very important in thinking holistically about transgender patients.” Clinicians can “use the opportunities we have when they present for gender-affirming care to optimize their overall health status, promote long-term health, and reduce the risks associated with hormonal therapy and gender-affirming surgeries,” she noted. “We hope to use this information to change our practice at the Cleveland Clinic and nationally as well. Transgender patients should be screened and aggressively treated for cardiovascular disease and risk factors,” she said.
Key barriers to overcome include determining the best way to reach out to transgender individuals and then making them feel comfortable in the clinical setting, Dr. Denby said. “This means that we must set up clinics that are approachable and safe for all comers. The lack of laws in many states that protect this vulnerable population also contributes to lack of access to care,” she added.
“We hope to continue research in this arena about how to effectively screen and treat transgender patients as they present to care, not only in the transgender clinic, but also to primary care providers (ob.gyn., internal medicine, family medicine, pediatrics) who also care for this population” since no specific guidelines currently exist to direct the screening for cardiovascular patients in particular, she said.
Findings offer foundation for LGBTQ cardiovascular studies
“This [study] provides us with a good rationale for why we should be considering cardiovascular health in transgender adults,” Billy A. Caceres, PhD, RN, of Columbia University School of Nursing, New York, said in an interview. “It is largely descriptive, but I think that that’s a good step in terms of at least understanding the magnitude of this problem. In addition, I think that what this abstract might do is help lead to future research that examines potentially the associations between not only gender-affirming hormone therapies but other potential social determinants like discrimination or poverty on the cardiovascular health of transgender people,” he noted.
Dr. Caceres served as chair of the writing group for the recent American Heart Association Scientific Statement: LGBTQ Heart Health published in Circulation. He had no financial conflicts to disclose.
The study received no outside funding. Dr. Denby had no financial conflicts to disclose.
SOURCE: Denby KJ et al. AHA 2020, Presentation P2274.
Cardiovascular disease risk is elevated among transgender individuals seeking gender-affirming hormone therapy, according to a retrospective study in 427 patients.
The transgender population often experiences socioeconomic and health disparities, including reduced access to care, Kara J. Denby, MD, said in an interview.
Previous research suggests that the use of gender-affirming hormone therapy (GAHT) may place transgender persons at increased cardiovascular risk, she said.
To identify the potential risk for transgender individuals, the researchers identified baseline cardiovascular risk in patients who had not yet undergone GAHT. Study participants were enrolled in a multidisciplinary transgender program, and the researchers collected data on demographics, medical history, vitals, medications, and laboratory results. The average age of the participants was 26 years, 172 identified as men, 236 as women, and 20 as nonbinary.
Overall, 55% of the participants had a chronic medical condition at baseline. Of these, 74 patients had hypertension, 41 had hyperlipidemia, 2 had a history of stroke, 7 had coronary artery disease, and 4 had chronic obstructive pulmonary disease.
For all patients who did not have documented atherosclerotic cardiovascular disease, their American College of Cardiology/American Heart Association ASCVD and QRISK3 risk scores were calculated. “The incidence of undiagnosed hypertension and hyperlipidemia was 6.8% and 11.3% respectively, and of these cases, only 64% and 24% were on appropriate therapies,” noted Dr. Denby of the Cleveland (Ohio) Clinic.
She reported the results Nov. 13 in a presentation at the at the virtual American Heart Association scientific sessions.
The findings were limited by the observational nature of the study.
However, the results suggest that transgender patients “appear to be at higher risk than their age-matched historical cohorts regardless of gender,” said Dr. Denby. More research is needed, but cardiovascular disease–prevention efforts may be inadequate in the transgender population given the elevated risk observed in this study, she concluded.
Growing transgender population is medically underserved
The transgender population is growing in the United States and internationally, said Dr. Denby. “This group has a history of being marginalized as a result of their transgender status with socioeconomic and health repercussions,” she said. “It is well known that transgender patients are less likely to have access to health care or utilize health care for a variety of reasons, including stigma and fear of mistreatment. This often leads transgender individuals to present to care late in disease processes which makes their disease harder to treat and often leads to emergent medical conditions,” she added.
“Transgender men and women are at high risk for cardiovascular disease and often aren’t screened at recommended intervals because of decreased health care use compared to their cisgender counterparts,” she said. “This may lead to untreated diseases that make them even more likely to suffer poor health outcomes.”
The current study is important because there are “almost no prior data regarding the cardiovascular health status of this population prior to gender-affirming care,” Dr. Denby emphasized. “There are data that gay, lesbian, and bisexual individuals are at higher risk for poor cardiovascular outcomes, but the same data are lacking in the transgender group,” she said.
“As transgender individuals have frequent physician visits while on hormonal therapy, this seems like the opportune time to screen for cardiovascular risk factors and treat previously undiagnosed diseases that can lead to poor health outcomes in the future,” Dr. Denby explained. “If we are able to intervene at an earlier age, perhaps we can help prevent poor health outcomes down the road,” she said.
Additional research can inform practice
Dr. Denby said she was not surprised by the findings. “This is a very high-risk population that often doesn’t follow closely in the health care system,” she said. “These data are very important in thinking holistically about transgender patients.” Clinicians can “use the opportunities we have when they present for gender-affirming care to optimize their overall health status, promote long-term health, and reduce the risks associated with hormonal therapy and gender-affirming surgeries,” she noted. “We hope to use this information to change our practice at the Cleveland Clinic and nationally as well. Transgender patients should be screened and aggressively treated for cardiovascular disease and risk factors,” she said.
Key barriers to overcome include determining the best way to reach out to transgender individuals and then making them feel comfortable in the clinical setting, Dr. Denby said. “This means that we must set up clinics that are approachable and safe for all comers. The lack of laws in many states that protect this vulnerable population also contributes to lack of access to care,” she added.
“We hope to continue research in this arena about how to effectively screen and treat transgender patients as they present to care, not only in the transgender clinic, but also to primary care providers (ob.gyn., internal medicine, family medicine, pediatrics) who also care for this population” since no specific guidelines currently exist to direct the screening for cardiovascular patients in particular, she said.
Findings offer foundation for LGBTQ cardiovascular studies
“This [study] provides us with a good rationale for why we should be considering cardiovascular health in transgender adults,” Billy A. Caceres, PhD, RN, of Columbia University School of Nursing, New York, said in an interview. “It is largely descriptive, but I think that that’s a good step in terms of at least understanding the magnitude of this problem. In addition, I think that what this abstract might do is help lead to future research that examines potentially the associations between not only gender-affirming hormone therapies but other potential social determinants like discrimination or poverty on the cardiovascular health of transgender people,” he noted.
Dr. Caceres served as chair of the writing group for the recent American Heart Association Scientific Statement: LGBTQ Heart Health published in Circulation. He had no financial conflicts to disclose.
The study received no outside funding. Dr. Denby had no financial conflicts to disclose.
SOURCE: Denby KJ et al. AHA 2020, Presentation P2274.
FROM AHA 2020
Semaglutide shows promise in NASH phase 2 study
according to a phase 2, double-blind, randomized, placebo-controlled trial published in the New England Journal of Medicine and presented at the 2020 American Association for the Study of Liver Diseases (AASLD) meeting.
“This bodes well for further study of semaglutide and is supported further by marked improvements in weight, glycemic control and lipid profile,” commented the study’s senior author Philip N. Newsome, PhD, FRCPE, of the University of Birmingham (England), in an interview.
The highest daily dose (0.4 mg) of the glucagonlike peptide-1 (GLP-1) receptor agonist, semaglutide, which is approved for the treatment of type 2 diabetes, led to levels of NASH resolution “which are higher than any previously demonstrated,” noted Dr. Newsome. “This was also accompanied by improvement in noninvasive markers of liver fibrosis and also less fibrosis progression, compared to placebo.”
“I think this represents an exciting advance and will, if confirmed in further studies, mark a step-change in our management of patients with NASH,” he added.
The multicenter study, conducted at 143 sites in 16 countries, included 320 patients, aged 18-75 years, with or without type 2 diabetes, who had histologic evidence of NASH and stage 1-3 liver fibrosis.
They were randomized in a 3:3:3:1:1:1 ratio to receive once-daily subcutaneous semaglutide at a dose of 0.1, 0.2, or 0.4 mg, or placebo for 72 weeks.
The primary endpoint was resolution of NASH and no worsening of fibrosis, with a secondary endpoint being improvement of fibrosis by at least one stage without worsening of NASH.
The study found 40% of patients in the 0.1-mg semaglutide group, 36% in the 0.2-mg group, and 59% in the 0.4-mg group achieved NASH resolution with no worsening of fibrosis, compared with 17% of the placebo group (odds ratio, 6.87; P < .001 for the highest semaglutide dose). However, the treatment did not lead to significant between-group differences in the secondary endpoint, which occurred in 43% of patients on the highest semaglutide dose compared to 33% in the placebo group (OR, 1.42; P = .48).
Treatment with semaglutide also resulted in dose-dependent reductions in body weight, as well as in glycated hemoglobin levels. Bodyweight was reduced by a mean of 5% in the 0.1-mg semaglutide group, followed by mean reductions of 9% and 13% in the 0.2-mg and 0.4-mg groups respectively. This compared to a mean reduction of 1% in the placebo group.
Similarly, glycated hemoglobin levels among patients with type 2 diabetes dropped by 0.63, 1.07, and 1.15 percentage points in the 0.1-mg, 0.2-mg, and 0.4-mg semaglutide groups respectively, compared with a drop of 0.01 percentage point in the placebo group.
“The fact that the percentage of patients who had an improvement in fibrosis stage was not significantly higher with semaglutide than with placebo – despite a greater benefit with respect to NASH resolution and dose-dependent weight loss – was unexpected, given that previous studies have suggested that resolution of NASH and improvements in activity scores for the components of nonalcoholic fatty liver disease are associated with regression of fibrosis,” wrote the authors. “However, the temporal association among NASH resolution, weight loss, and improvement in fibrosis stage is not fully understood. It is possible that the current trial was not of sufficient duration for improvements in fibrosis stage to become apparent.”
The authors also noted that the safety profile of semaglutide was “consistent with that observed in patients with type 2 diabetes in other trials and with the known effects of GLP-1 receptor agonists,” with gastrointestinal disorders being the most commonly reported.
Nausea, constipation, and vomiting were reported more often in the 0.4-mg semaglutide group than in the placebo group (nausea, 42% vs. 11%; constipation, 22% vs. 12%; and vomiting, 15% vs. 2%).
The overall incidence of benign, malignant, or unspecified neoplasms was 15% in the treatment groups versus 8% in the placebo group.
Rowen K. Zetterman, MD, who was not involved with the study, noted that “treatment of NASH is currently limited, and no therapies have yet been approved by the Food and Drug Administration.”
The findings are “important but not yet exciting,” added Dr. Zetterman, who is professor emeritus of internal medicine and associate vice chancellor for strategic planning for the University of Nebraska Medical Center, Omaha.
“Though reversal of liver fibrosis was not noted, the resolution of hepatic inflammation and liver cell injury by semaglutide suggests it may be slowing disease progression,” said Dr. Zetterman, who also serves on the editorial advisory board of Internal Medicine News. This “warrants additional studies where longer treatment with semaglutide may prove reversal of fibrosis and/or prevention of progression to cirrhosis.”
The study was sponsored by Novo Nordisk. Dr. Newsome reported disclosures related to Novo Nordisk during the conduct of the study, and to Boehringer Ingelheim, Bristol-Myers Squibb, Echosens, Gilead, Pfizer, Pharmaxis, and Poxel. Several of the other study authors reported receiving fees and grants from various pharmaceutical companies, including Novo Nordisk One author reported pending patents for the use of semaglutide. Dr. Zetterman had no relevant disclosures.
SOURCE: Newsome PN et al. N Engl J Med. 2020 Nov 13. doi: 10.1056/NEJMoa2028395.
according to a phase 2, double-blind, randomized, placebo-controlled trial published in the New England Journal of Medicine and presented at the 2020 American Association for the Study of Liver Diseases (AASLD) meeting.
“This bodes well for further study of semaglutide and is supported further by marked improvements in weight, glycemic control and lipid profile,” commented the study’s senior author Philip N. Newsome, PhD, FRCPE, of the University of Birmingham (England), in an interview.
The highest daily dose (0.4 mg) of the glucagonlike peptide-1 (GLP-1) receptor agonist, semaglutide, which is approved for the treatment of type 2 diabetes, led to levels of NASH resolution “which are higher than any previously demonstrated,” noted Dr. Newsome. “This was also accompanied by improvement in noninvasive markers of liver fibrosis and also less fibrosis progression, compared to placebo.”
“I think this represents an exciting advance and will, if confirmed in further studies, mark a step-change in our management of patients with NASH,” he added.
The multicenter study, conducted at 143 sites in 16 countries, included 320 patients, aged 18-75 years, with or without type 2 diabetes, who had histologic evidence of NASH and stage 1-3 liver fibrosis.
They were randomized in a 3:3:3:1:1:1 ratio to receive once-daily subcutaneous semaglutide at a dose of 0.1, 0.2, or 0.4 mg, or placebo for 72 weeks.
The primary endpoint was resolution of NASH and no worsening of fibrosis, with a secondary endpoint being improvement of fibrosis by at least one stage without worsening of NASH.
The study found 40% of patients in the 0.1-mg semaglutide group, 36% in the 0.2-mg group, and 59% in the 0.4-mg group achieved NASH resolution with no worsening of fibrosis, compared with 17% of the placebo group (odds ratio, 6.87; P < .001 for the highest semaglutide dose). However, the treatment did not lead to significant between-group differences in the secondary endpoint, which occurred in 43% of patients on the highest semaglutide dose compared to 33% in the placebo group (OR, 1.42; P = .48).
Treatment with semaglutide also resulted in dose-dependent reductions in body weight, as well as in glycated hemoglobin levels. Bodyweight was reduced by a mean of 5% in the 0.1-mg semaglutide group, followed by mean reductions of 9% and 13% in the 0.2-mg and 0.4-mg groups respectively. This compared to a mean reduction of 1% in the placebo group.
Similarly, glycated hemoglobin levels among patients with type 2 diabetes dropped by 0.63, 1.07, and 1.15 percentage points in the 0.1-mg, 0.2-mg, and 0.4-mg semaglutide groups respectively, compared with a drop of 0.01 percentage point in the placebo group.
“The fact that the percentage of patients who had an improvement in fibrosis stage was not significantly higher with semaglutide than with placebo – despite a greater benefit with respect to NASH resolution and dose-dependent weight loss – was unexpected, given that previous studies have suggested that resolution of NASH and improvements in activity scores for the components of nonalcoholic fatty liver disease are associated with regression of fibrosis,” wrote the authors. “However, the temporal association among NASH resolution, weight loss, and improvement in fibrosis stage is not fully understood. It is possible that the current trial was not of sufficient duration for improvements in fibrosis stage to become apparent.”
The authors also noted that the safety profile of semaglutide was “consistent with that observed in patients with type 2 diabetes in other trials and with the known effects of GLP-1 receptor agonists,” with gastrointestinal disorders being the most commonly reported.
Nausea, constipation, and vomiting were reported more often in the 0.4-mg semaglutide group than in the placebo group (nausea, 42% vs. 11%; constipation, 22% vs. 12%; and vomiting, 15% vs. 2%).
The overall incidence of benign, malignant, or unspecified neoplasms was 15% in the treatment groups versus 8% in the placebo group.
Rowen K. Zetterman, MD, who was not involved with the study, noted that “treatment of NASH is currently limited, and no therapies have yet been approved by the Food and Drug Administration.”
The findings are “important but not yet exciting,” added Dr. Zetterman, who is professor emeritus of internal medicine and associate vice chancellor for strategic planning for the University of Nebraska Medical Center, Omaha.
“Though reversal of liver fibrosis was not noted, the resolution of hepatic inflammation and liver cell injury by semaglutide suggests it may be slowing disease progression,” said Dr. Zetterman, who also serves on the editorial advisory board of Internal Medicine News. This “warrants additional studies where longer treatment with semaglutide may prove reversal of fibrosis and/or prevention of progression to cirrhosis.”
The study was sponsored by Novo Nordisk. Dr. Newsome reported disclosures related to Novo Nordisk during the conduct of the study, and to Boehringer Ingelheim, Bristol-Myers Squibb, Echosens, Gilead, Pfizer, Pharmaxis, and Poxel. Several of the other study authors reported receiving fees and grants from various pharmaceutical companies, including Novo Nordisk One author reported pending patents for the use of semaglutide. Dr. Zetterman had no relevant disclosures.
SOURCE: Newsome PN et al. N Engl J Med. 2020 Nov 13. doi: 10.1056/NEJMoa2028395.
according to a phase 2, double-blind, randomized, placebo-controlled trial published in the New England Journal of Medicine and presented at the 2020 American Association for the Study of Liver Diseases (AASLD) meeting.
“This bodes well for further study of semaglutide and is supported further by marked improvements in weight, glycemic control and lipid profile,” commented the study’s senior author Philip N. Newsome, PhD, FRCPE, of the University of Birmingham (England), in an interview.
The highest daily dose (0.4 mg) of the glucagonlike peptide-1 (GLP-1) receptor agonist, semaglutide, which is approved for the treatment of type 2 diabetes, led to levels of NASH resolution “which are higher than any previously demonstrated,” noted Dr. Newsome. “This was also accompanied by improvement in noninvasive markers of liver fibrosis and also less fibrosis progression, compared to placebo.”
“I think this represents an exciting advance and will, if confirmed in further studies, mark a step-change in our management of patients with NASH,” he added.
The multicenter study, conducted at 143 sites in 16 countries, included 320 patients, aged 18-75 years, with or without type 2 diabetes, who had histologic evidence of NASH and stage 1-3 liver fibrosis.
They were randomized in a 3:3:3:1:1:1 ratio to receive once-daily subcutaneous semaglutide at a dose of 0.1, 0.2, or 0.4 mg, or placebo for 72 weeks.
The primary endpoint was resolution of NASH and no worsening of fibrosis, with a secondary endpoint being improvement of fibrosis by at least one stage without worsening of NASH.
The study found 40% of patients in the 0.1-mg semaglutide group, 36% in the 0.2-mg group, and 59% in the 0.4-mg group achieved NASH resolution with no worsening of fibrosis, compared with 17% of the placebo group (odds ratio, 6.87; P < .001 for the highest semaglutide dose). However, the treatment did not lead to significant between-group differences in the secondary endpoint, which occurred in 43% of patients on the highest semaglutide dose compared to 33% in the placebo group (OR, 1.42; P = .48).
Treatment with semaglutide also resulted in dose-dependent reductions in body weight, as well as in glycated hemoglobin levels. Bodyweight was reduced by a mean of 5% in the 0.1-mg semaglutide group, followed by mean reductions of 9% and 13% in the 0.2-mg and 0.4-mg groups respectively. This compared to a mean reduction of 1% in the placebo group.
Similarly, glycated hemoglobin levels among patients with type 2 diabetes dropped by 0.63, 1.07, and 1.15 percentage points in the 0.1-mg, 0.2-mg, and 0.4-mg semaglutide groups respectively, compared with a drop of 0.01 percentage point in the placebo group.
“The fact that the percentage of patients who had an improvement in fibrosis stage was not significantly higher with semaglutide than with placebo – despite a greater benefit with respect to NASH resolution and dose-dependent weight loss – was unexpected, given that previous studies have suggested that resolution of NASH and improvements in activity scores for the components of nonalcoholic fatty liver disease are associated with regression of fibrosis,” wrote the authors. “However, the temporal association among NASH resolution, weight loss, and improvement in fibrosis stage is not fully understood. It is possible that the current trial was not of sufficient duration for improvements in fibrosis stage to become apparent.”
The authors also noted that the safety profile of semaglutide was “consistent with that observed in patients with type 2 diabetes in other trials and with the known effects of GLP-1 receptor agonists,” with gastrointestinal disorders being the most commonly reported.
Nausea, constipation, and vomiting were reported more often in the 0.4-mg semaglutide group than in the placebo group (nausea, 42% vs. 11%; constipation, 22% vs. 12%; and vomiting, 15% vs. 2%).
The overall incidence of benign, malignant, or unspecified neoplasms was 15% in the treatment groups versus 8% in the placebo group.
Rowen K. Zetterman, MD, who was not involved with the study, noted that “treatment of NASH is currently limited, and no therapies have yet been approved by the Food and Drug Administration.”
The findings are “important but not yet exciting,” added Dr. Zetterman, who is professor emeritus of internal medicine and associate vice chancellor for strategic planning for the University of Nebraska Medical Center, Omaha.
“Though reversal of liver fibrosis was not noted, the resolution of hepatic inflammation and liver cell injury by semaglutide suggests it may be slowing disease progression,” said Dr. Zetterman, who also serves on the editorial advisory board of Internal Medicine News. This “warrants additional studies where longer treatment with semaglutide may prove reversal of fibrosis and/or prevention of progression to cirrhosis.”
The study was sponsored by Novo Nordisk. Dr. Newsome reported disclosures related to Novo Nordisk during the conduct of the study, and to Boehringer Ingelheim, Bristol-Myers Squibb, Echosens, Gilead, Pfizer, Pharmaxis, and Poxel. Several of the other study authors reported receiving fees and grants from various pharmaceutical companies, including Novo Nordisk One author reported pending patents for the use of semaglutide. Dr. Zetterman had no relevant disclosures.
SOURCE: Newsome PN et al. N Engl J Med. 2020 Nov 13. doi: 10.1056/NEJMoa2028395.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Do electronic reminder systems help patients with T2DM to lose weight?
EVIDENCE SUMMARY
A meta-analysis of 6 RCTs studied the effect of smartphone self-care applications on A1C, weight, blood pressure, and lipids in adult patients with T2DM. All the interventions comprised 4 components: electronic self-management prompts and reminders, personal measuring devices, patient-driven data upload, and remote analysis of the data with feedback. The review excluded studies that used phone calls or lasted fewer than 3 months.
Some improvement in A1C found, but no effect on weight
Telehealth interventions improved A1C more than usual care (6 trials, 884 patients; mean difference = –0.40%; 95% CI, –0.69% to –0.11%).1 A subset of 4 studies with 560 patients evaluated changes in weight. Patients had a mean age of 61 years and average weight of 84 kg (in 3 of 4 studies reporting baseline weight). Aggregate weight loss was insignificant after 3 to 12 months (mean difference = –0.84 kg; 95% CI, –2.04 kg to 0.36 kg, P = .17). Investigators reported no harms. Limitations of the analysis included high heterogeneity in the main outcome of A1C (I2 = 70%) but low heterogeneity within the 4 studies assessing weight (I2 = 30%).
Other, small studies found no change in A1C
Two subsequent small RCTs came to different conclusions than the meta-analysis. One compared the impact of individualized physical activity–based text messages in response to pedometer readings with pedometer use alone.2 It included 126 adult patients (mean age, 50.5 years) with T2DM who had an A1C > 7% and access to an Internet-connected computer. Researchers excluded patients who were unable to perform moderate physical activity or who had cognitive deficits.
At enrollment, researchers supplied all patients with a pedometer and an appointment with a counselor to set goals for physical activity. They sent 2 text messages daily to the intervention group (and none to the control group) based on uploaded pedometer data. One message detailed physical activity progress and the second encouraged increased physical activity. The primary outcome was mean step counts per month; secondary outcomes included A1C and weight measured at 6 months.
The groups showed no significant difference in A1C (mean difference = 0.07%; 95% CI, –0.47% to 0.34%, P = .75) or weight loss (mean difference = 3.1 lb; 95% CI, –24.5 lb to 18.3 lb, P = .77). Many patients (43%) reported difficulty uploading step counts, receiving texts, and responding to texts. The dropout rate was 24%.
A second RCT with 150 patients, using a less elaborate protocol, assessed the effectiveness of tailored text-message reminders compared with nontailored text messages to improve A1C and body mass index (BMI).3 Patients were adult Iranians (mean age, 52.5 years) with T2DM who owned a cell phone and could receive and read text messages.
Patients filled out a diabetic self-care assessment to identify barriers to improving care and were randomized into 3 groups. The first group received tailored text messages (75% addressing the patient’s top 2 barriers to self-care and 25% general messages). The second group received nontailored text messages of encouragement. The control group received no text messages.
Continue to: After 3 months...
After 3 months, BMI was reduced in both messaging groups but not the control group (tailored text = –0.6 kg/m2, nontailored text = –0.5 kg/m2, controls = 0.7 kg/m2; P < .05). A1C levels didn’t change significantly. One limitation of the study was that 30% to 35% of the patients in the intervention group had a university-level education, compared with 12% in the control group.
Recommendations
The Department of Veterans Affairs issued guidelines in 2017 regarding management of patients with T2DM in primary care.4 The guidelines state that all patients should receive individualized self-management education using “modalities tailored to their preferences” (strong recommendation). They further recommend “offering one or more bidirectional telehealth interventions” in coordination with patients’ health care providers (weak recommendation).
The 2017 diabetes self-management recommendations endorsed by the American Diabetes Association state that “strong evidence” shows that incorporating text messaging into diabetes care improves outcomes, enhances feedback loops, and empowers patients.5
Editor’s takeaway
Telehealth offers mechanisms for patients and physicians to enhance communication about health behaviors and health status. But does it alter outcomes? The cited literature suggests that benefits aren’t a forgone conclusion and that acceptability, ease of use, cost, and individualization are critical issues in telehealth design.
1. Cui M, Wu X, Mao J, et al. T2DM self-management via smartphone applications: a systematic review and meta-analysis. PLoS ONE. 2016;11:e0166718.
2. Agboola S, Jethwani K, Lopez L, et al. Text to Move: A randomized controlled trial of a text-messaging program to improve physical activity behaviors in patients with type 2 diabetes mellitus. J Med Internet Res. 2016;18:e307.
3. Peimani M, Rambod C, Omidvar M, et al. Effectiveness of short message service-based intervention (SMS) on self-care in type 2 diabetes: a feasibility study. Prim Care Diabetes. 2016;10:251-258.
4. Guideline summary: VA/DoD clinical practice guideline for the management of type 2 diabetes mellitus in primary care. Rockville, MD: Agency for Healthcare Research and Quality; 2017. www.innovations.ahrq.gov/qualitytools/department-veterans-affairsdepartment-defense-vadod-clinical-practice-guideline-4. Accessed October 26, 2020.
5. Beck J, Greenwood DA, Blanton L, et al. 2017 National Standards for Diabetes Self-Management, Education and Support. Diabetes Care. 2017;40:1409-1419.
EVIDENCE SUMMARY
A meta-analysis of 6 RCTs studied the effect of smartphone self-care applications on A1C, weight, blood pressure, and lipids in adult patients with T2DM. All the interventions comprised 4 components: electronic self-management prompts and reminders, personal measuring devices, patient-driven data upload, and remote analysis of the data with feedback. The review excluded studies that used phone calls or lasted fewer than 3 months.
Some improvement in A1C found, but no effect on weight
Telehealth interventions improved A1C more than usual care (6 trials, 884 patients; mean difference = –0.40%; 95% CI, –0.69% to –0.11%).1 A subset of 4 studies with 560 patients evaluated changes in weight. Patients had a mean age of 61 years and average weight of 84 kg (in 3 of 4 studies reporting baseline weight). Aggregate weight loss was insignificant after 3 to 12 months (mean difference = –0.84 kg; 95% CI, –2.04 kg to 0.36 kg, P = .17). Investigators reported no harms. Limitations of the analysis included high heterogeneity in the main outcome of A1C (I2 = 70%) but low heterogeneity within the 4 studies assessing weight (I2 = 30%).
Other, small studies found no change in A1C
Two subsequent small RCTs came to different conclusions than the meta-analysis. One compared the impact of individualized physical activity–based text messages in response to pedometer readings with pedometer use alone.2 It included 126 adult patients (mean age, 50.5 years) with T2DM who had an A1C > 7% and access to an Internet-connected computer. Researchers excluded patients who were unable to perform moderate physical activity or who had cognitive deficits.
At enrollment, researchers supplied all patients with a pedometer and an appointment with a counselor to set goals for physical activity. They sent 2 text messages daily to the intervention group (and none to the control group) based on uploaded pedometer data. One message detailed physical activity progress and the second encouraged increased physical activity. The primary outcome was mean step counts per month; secondary outcomes included A1C and weight measured at 6 months.
The groups showed no significant difference in A1C (mean difference = 0.07%; 95% CI, –0.47% to 0.34%, P = .75) or weight loss (mean difference = 3.1 lb; 95% CI, –24.5 lb to 18.3 lb, P = .77). Many patients (43%) reported difficulty uploading step counts, receiving texts, and responding to texts. The dropout rate was 24%.
A second RCT with 150 patients, using a less elaborate protocol, assessed the effectiveness of tailored text-message reminders compared with nontailored text messages to improve A1C and body mass index (BMI).3 Patients were adult Iranians (mean age, 52.5 years) with T2DM who owned a cell phone and could receive and read text messages.
Patients filled out a diabetic self-care assessment to identify barriers to improving care and were randomized into 3 groups. The first group received tailored text messages (75% addressing the patient’s top 2 barriers to self-care and 25% general messages). The second group received nontailored text messages of encouragement. The control group received no text messages.
Continue to: After 3 months...
After 3 months, BMI was reduced in both messaging groups but not the control group (tailored text = –0.6 kg/m2, nontailored text = –0.5 kg/m2, controls = 0.7 kg/m2; P < .05). A1C levels didn’t change significantly. One limitation of the study was that 30% to 35% of the patients in the intervention group had a university-level education, compared with 12% in the control group.
Recommendations
The Department of Veterans Affairs issued guidelines in 2017 regarding management of patients with T2DM in primary care.4 The guidelines state that all patients should receive individualized self-management education using “modalities tailored to their preferences” (strong recommendation). They further recommend “offering one or more bidirectional telehealth interventions” in coordination with patients’ health care providers (weak recommendation).
The 2017 diabetes self-management recommendations endorsed by the American Diabetes Association state that “strong evidence” shows that incorporating text messaging into diabetes care improves outcomes, enhances feedback loops, and empowers patients.5
Editor’s takeaway
Telehealth offers mechanisms for patients and physicians to enhance communication about health behaviors and health status. But does it alter outcomes? The cited literature suggests that benefits aren’t a forgone conclusion and that acceptability, ease of use, cost, and individualization are critical issues in telehealth design.
EVIDENCE SUMMARY
A meta-analysis of 6 RCTs studied the effect of smartphone self-care applications on A1C, weight, blood pressure, and lipids in adult patients with T2DM. All the interventions comprised 4 components: electronic self-management prompts and reminders, personal measuring devices, patient-driven data upload, and remote analysis of the data with feedback. The review excluded studies that used phone calls or lasted fewer than 3 months.
Some improvement in A1C found, but no effect on weight
Telehealth interventions improved A1C more than usual care (6 trials, 884 patients; mean difference = –0.40%; 95% CI, –0.69% to –0.11%).1 A subset of 4 studies with 560 patients evaluated changes in weight. Patients had a mean age of 61 years and average weight of 84 kg (in 3 of 4 studies reporting baseline weight). Aggregate weight loss was insignificant after 3 to 12 months (mean difference = –0.84 kg; 95% CI, –2.04 kg to 0.36 kg, P = .17). Investigators reported no harms. Limitations of the analysis included high heterogeneity in the main outcome of A1C (I2 = 70%) but low heterogeneity within the 4 studies assessing weight (I2 = 30%).
Other, small studies found no change in A1C
Two subsequent small RCTs came to different conclusions than the meta-analysis. One compared the impact of individualized physical activity–based text messages in response to pedometer readings with pedometer use alone.2 It included 126 adult patients (mean age, 50.5 years) with T2DM who had an A1C > 7% and access to an Internet-connected computer. Researchers excluded patients who were unable to perform moderate physical activity or who had cognitive deficits.
At enrollment, researchers supplied all patients with a pedometer and an appointment with a counselor to set goals for physical activity. They sent 2 text messages daily to the intervention group (and none to the control group) based on uploaded pedometer data. One message detailed physical activity progress and the second encouraged increased physical activity. The primary outcome was mean step counts per month; secondary outcomes included A1C and weight measured at 6 months.
The groups showed no significant difference in A1C (mean difference = 0.07%; 95% CI, –0.47% to 0.34%, P = .75) or weight loss (mean difference = 3.1 lb; 95% CI, –24.5 lb to 18.3 lb, P = .77). Many patients (43%) reported difficulty uploading step counts, receiving texts, and responding to texts. The dropout rate was 24%.
A second RCT with 150 patients, using a less elaborate protocol, assessed the effectiveness of tailored text-message reminders compared with nontailored text messages to improve A1C and body mass index (BMI).3 Patients were adult Iranians (mean age, 52.5 years) with T2DM who owned a cell phone and could receive and read text messages.
Patients filled out a diabetic self-care assessment to identify barriers to improving care and were randomized into 3 groups. The first group received tailored text messages (75% addressing the patient’s top 2 barriers to self-care and 25% general messages). The second group received nontailored text messages of encouragement. The control group received no text messages.
Continue to: After 3 months...
After 3 months, BMI was reduced in both messaging groups but not the control group (tailored text = –0.6 kg/m2, nontailored text = –0.5 kg/m2, controls = 0.7 kg/m2; P < .05). A1C levels didn’t change significantly. One limitation of the study was that 30% to 35% of the patients in the intervention group had a university-level education, compared with 12% in the control group.
Recommendations
The Department of Veterans Affairs issued guidelines in 2017 regarding management of patients with T2DM in primary care.4 The guidelines state that all patients should receive individualized self-management education using “modalities tailored to their preferences” (strong recommendation). They further recommend “offering one or more bidirectional telehealth interventions” in coordination with patients’ health care providers (weak recommendation).
The 2017 diabetes self-management recommendations endorsed by the American Diabetes Association state that “strong evidence” shows that incorporating text messaging into diabetes care improves outcomes, enhances feedback loops, and empowers patients.5
Editor’s takeaway
Telehealth offers mechanisms for patients and physicians to enhance communication about health behaviors and health status. But does it alter outcomes? The cited literature suggests that benefits aren’t a forgone conclusion and that acceptability, ease of use, cost, and individualization are critical issues in telehealth design.
1. Cui M, Wu X, Mao J, et al. T2DM self-management via smartphone applications: a systematic review and meta-analysis. PLoS ONE. 2016;11:e0166718.
2. Agboola S, Jethwani K, Lopez L, et al. Text to Move: A randomized controlled trial of a text-messaging program to improve physical activity behaviors in patients with type 2 diabetes mellitus. J Med Internet Res. 2016;18:e307.
3. Peimani M, Rambod C, Omidvar M, et al. Effectiveness of short message service-based intervention (SMS) on self-care in type 2 diabetes: a feasibility study. Prim Care Diabetes. 2016;10:251-258.
4. Guideline summary: VA/DoD clinical practice guideline for the management of type 2 diabetes mellitus in primary care. Rockville, MD: Agency for Healthcare Research and Quality; 2017. www.innovations.ahrq.gov/qualitytools/department-veterans-affairsdepartment-defense-vadod-clinical-practice-guideline-4. Accessed October 26, 2020.
5. Beck J, Greenwood DA, Blanton L, et al. 2017 National Standards for Diabetes Self-Management, Education and Support. Diabetes Care. 2017;40:1409-1419.
1. Cui M, Wu X, Mao J, et al. T2DM self-management via smartphone applications: a systematic review and meta-analysis. PLoS ONE. 2016;11:e0166718.
2. Agboola S, Jethwani K, Lopez L, et al. Text to Move: A randomized controlled trial of a text-messaging program to improve physical activity behaviors in patients with type 2 diabetes mellitus. J Med Internet Res. 2016;18:e307.
3. Peimani M, Rambod C, Omidvar M, et al. Effectiveness of short message service-based intervention (SMS) on self-care in type 2 diabetes: a feasibility study. Prim Care Diabetes. 2016;10:251-258.
4. Guideline summary: VA/DoD clinical practice guideline for the management of type 2 diabetes mellitus in primary care. Rockville, MD: Agency for Healthcare Research and Quality; 2017. www.innovations.ahrq.gov/qualitytools/department-veterans-affairsdepartment-defense-vadod-clinical-practice-guideline-4. Accessed October 26, 2020.
5. Beck J, Greenwood DA, Blanton L, et al. 2017 National Standards for Diabetes Self-Management, Education and Support. Diabetes Care. 2017;40:1409-1419.
EVIDENCE-BASED ANSWER:
PROBABLY NOT—but they may augment self-management. Four-component telehealth systems—including electronic reminders, measuring devices, patient-driven data upload, and remote data analysis—likely don’t result in significant weight reductions in adults with type 2 diabetes (T2DM). However, their use may be associated with a decrease in hemoglobin A1C of about 0.4% (strength of recommendation [SOR]: B, meta-analysis of randomized controlled trials [RCTs] and conflicting smaller subsequent RCTs).
Telehealth is considered a reasonable option for augmenting diabetes self-management in patients who are facile with the technology (SOR: C, expert opinion).
PCI success vs. meds only in diabetes may depend on LDL-C control
In order for percutaneous coronary intervention (PCI) to shine, compared with meds alone in patients with type-2 diabetes and stable coronary disease (CAD), it needs help from aggressive control of LDL cholesterol (LDL-C) levels, suggests a patient-level meta-analysis of three major randomized trials.
Performing PCI in such patients with diabetes conferred further benefit over optimal medical therapy (OMT) for major adverse cardiac or cerebrovascular events (MACCE) only among those whose LDL-C levels had been pushed below the guidelines-specified threshold of 70 mg/dL within 1 year.
At that level of LDL-C control, PCI, compared with the meds-alone strategy, was followed by a nearly 40% drop in 4-year risk for the composite endpoint, which consisted of death from any cause or nonfatal myocardial infarction (MI) or stroke.
Also for patients reaching a 1-year LDL-C of <70 mg/dL, the risk of MACCE was similar for those who had been assigned to coronary bypass surgery (CABG), compared with PCI. But that risk was significantly lower for the CABG group among those reaching LDL-C levels above that threshold.
“The strategy of revascularization with the LDL lowering, that’s the combination that seems to be a winner” in such patients with diabetes and stable CAD, lead author Michael E. Farkouh, MD, MSc, said in an interview.
If their LDL-C “stays above 70 mg/dL, they don’t really enjoy any benefit of PCI. It’s a message to our interventional community to really drive that LDL down,” said Dr. Farkouh, of the University of Toronto. “Not only with statins, but perhaps with PCSK9 inhibitors, ezetimibe, and other therapies to lower that LDL-C.”
The analysis, published Nov. 2 in the Journal of the American College of Cardiology, pooled more than 4,000 patients with diabetes and stable CAD randomized in the BARI 2D, FREEDOM, and COURAGE trials.
The new study adds a twist to an ongoing theme throughout some meta-analyses and clinical trials like ISCHEMIA since the results of COURAGE were unveiled 13 years ago. The latter trial famously saw no significant difference in death, MI, or stroke in patients with stable CAD assigned to OMT with or without PCI. That set off years of controversy about the relative merits of the revascularization and meds-only strategies in stable CAD that persists today.
But, Dr. Farkouh proposed, whether PCI improves clinical outcomes, compared with meds alone, at least in patients with diabetes, may be tied to the success of LDL-C-lowering therapies in reaching that goal, which in the current study was below 70 mg/dL.
“In this analysis of pooled data from the three major trials, we demonstrate that attaining that level of LDL-C at 1 year portends a better outcome for PCI” in patients with diabetes and stable CAD, he said.
The findings “probably need to be studied further, but it is compelling to think that if we can drive the LDL-C down by one year after the procedure, we have better outcomes with PCI,” compared with a meds-only strategy in patients with diabetes and stable CAD. “That really vindicates a lot of those who believe in PCI,” Dr. Farkouh said.
“What’s surprising to me is, if the patient has an LDL less than 70, why is it that there is a benefit of PCI, compared to medical therapy alone? Because they’re already so aggressively managed, you would think there shouldn’t be a benefit,” Sripal Bangalore, MD, MHA, New York University, said in an interview. “For me, that part is difficult to understand.”
The finding somewhat contradicts the results of ISCHEMIA, in which OMT – including LDL-C-lowering therapy – was considered more aggressive than usually managed in practice, Bangalore said. Yet the trial saw no outcomes difference between PCI and the more conservative approach, leading some to speculate that PCI may be a better choice when, for whatever reason, medical therapy isn’t optimal.
The observed superiority of PCI over meds-only at the lowest LDL-C levels is, according to Dr. Banagalore, “more likely because of residual confounding, given the fact that they’re combining three different trials, which are aimed to address different sets of questions.” He was an investigator with the FREEDOM and ISCHEMIA trials but isn’t associated with the current report.
The main message from this observational analysis is that “of course, we want to get the LDL as low as possible in these patients with demonstrated cardiovascular disease and diabetes,” Donald M. Lloyd-Jones, MD, ScM, Northwestern University, Chicago, said in an interview. “Every one of these patients should be shooting for as low an LDL as possible.”
Regardless of revascularization strategy, he said, “we have to get people on a high-intensity statin, or at least their maximally targeted dose, and have a careful and thoughtful conversation about whether they need additional lowering with, perhaps, ezetimibe, if they’re not below the thresholds we’d like to see them at, in this case, 70 mg/dL.”
Still, the current findings that the relative effects of PCI and CABG in these patients may vary by degree of LDL-C reduction “are interesting, but would have to be tested a little bit more directly,” said Dr. Lloyd-Jones, who is not affiliated with the analysis.
An accompanying editorial, which also acknowledges the study’s limitations, says its results “are relevant for clinical practice and may pave the way toward the generation of novel personalized medicine models that can optimize care of patients with type-2 diabetes.”
They “support the concept of an individualized treatment strategy that accounts for a patient’s LDL-C level to estimate clinical outcomes and expected treatment effects after therapeutic interventions,” say the authors, led by Eliano P. Navarese, MD, PhD, Nicolaus Copernicus University, Bydgoszcz, Poland.
“For daily practice, these results also underscore the importance of follow-up LDL-C measurements, both as a risk stratifier and as an indicator for therapy adjustments,” they write, noting that “current guidelines provide no formal recommendation on when to check LDL-C after PCI.”
The meta-analysis followed a total of 4050 patients with diabetes and stable CAD from the three randomized trials, those with evaluable baseline and follow-up LDL-C measurements, for a median of 4 years after the 1-year LDL-C assessment. At that time, at least 90% of patients in each of the trials had statin prescriptions, the group reported.
At one year, 34.5% of the total cohort had an LDL-C <70 mg/dL; their mean was 55.8 mg/dL.
And 42.2% had an LDL-C from 70 mg/dL to <100 mg/dL; their mean was 83.4 mg/dL. Compared with patients with an LDL-C <70 mg/dL, their adjusted hazard ratio for the composite endpoint was not elevated at 1.07 (95% CI, 0.86-1.32, P = .54).
Finally, 23.2% had an LDL-C ≥100 mg/dL; the mean was 123.0 mg/dL. Compared with the group with the lowest 1-year LDL-C, their adjusted HR for MACCE was increased at 1.46 (95% CI, 1.15 - 1.85, P = .002).
That HR among the 42.3% of patients in the PCI cohort, compared with the 33.3% assigned to meds only, climbed significantly only among those in the lowest 1-year LDL-C stratum: HR, 0.61 (95% CI, 0.40-0.91, P = .016). Corresponding HRs in the mid-range and highest 1-year LDL strata were close to unity and nonsignificant at P = .71 and P = .98, respectively.
On the other hand, the 24.4% of patients assigned to CABG showed better MACCE outcomes than those in the meds-only group across all three 1-year LDL-C strata.
The risk of MACCE wasn’t significantly altered by CABG, compared with PCI among patients achieving a 1-year LDL-C less than 70 mg/dL. However, it fell by about one-half for CABG vs. PCI in both the mid-range and highest 1-year LDL-C strata, P = .003 and P = .022, respectively.
Dr. Bangalore said he’s entirely behind the results of the study’s comparison of PCI and CABG. “It’s exactly the hypothesis that I’ve been putting forward, that if you want to achieve results as good as CABG, do PCI with aggressive medical therapy.” That means second-generation drug-eluting stents for the target lesions, “and aggressive medical therapy to address all of the nontarget lesions, specifically in diabetics.”
It’s possible, Dr. Lloyd-Jones said, that there is “no longer a dichotomy between revascularization strategies,” with respect to clinical outcomes, in such patients who maintain an LDL less than 70 mg/dL, as the study suggests.
“But I wonder, if it had continued for another 4 years of follow-up, whether we would see the CABG patients start to have more events,” such that the CABG advantage goes away at higher LDL-C levels, he proposed.
Or, Dr. Lloyd-Jones speculated, if all patients had achieved LDL-C below 70 mg/dL, “would there be such a difference between the PCI and CABG groups? My bet would be that it would be small or abolished.”
Dr. Farkouh discloses receiving research grants from Amgen, Novo Nordisk, and Novartis. Disclosures for the other study authors can be found with the original article. Editorialist Dr. Navarese discloses receiving consulting fees or honoraria from Abbott, AstraZeneca, Amgen, Bayer, Sanofi, and Pfizer; and grants from Abbott and Amgen. Dr. Lloyd-Jones has disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
In order for percutaneous coronary intervention (PCI) to shine, compared with meds alone in patients with type-2 diabetes and stable coronary disease (CAD), it needs help from aggressive control of LDL cholesterol (LDL-C) levels, suggests a patient-level meta-analysis of three major randomized trials.
Performing PCI in such patients with diabetes conferred further benefit over optimal medical therapy (OMT) for major adverse cardiac or cerebrovascular events (MACCE) only among those whose LDL-C levels had been pushed below the guidelines-specified threshold of 70 mg/dL within 1 year.
At that level of LDL-C control, PCI, compared with the meds-alone strategy, was followed by a nearly 40% drop in 4-year risk for the composite endpoint, which consisted of death from any cause or nonfatal myocardial infarction (MI) or stroke.
Also for patients reaching a 1-year LDL-C of <70 mg/dL, the risk of MACCE was similar for those who had been assigned to coronary bypass surgery (CABG), compared with PCI. But that risk was significantly lower for the CABG group among those reaching LDL-C levels above that threshold.
“The strategy of revascularization with the LDL lowering, that’s the combination that seems to be a winner” in such patients with diabetes and stable CAD, lead author Michael E. Farkouh, MD, MSc, said in an interview.
If their LDL-C “stays above 70 mg/dL, they don’t really enjoy any benefit of PCI. It’s a message to our interventional community to really drive that LDL down,” said Dr. Farkouh, of the University of Toronto. “Not only with statins, but perhaps with PCSK9 inhibitors, ezetimibe, and other therapies to lower that LDL-C.”
The analysis, published Nov. 2 in the Journal of the American College of Cardiology, pooled more than 4,000 patients with diabetes and stable CAD randomized in the BARI 2D, FREEDOM, and COURAGE trials.
The new study adds a twist to an ongoing theme throughout some meta-analyses and clinical trials like ISCHEMIA since the results of COURAGE were unveiled 13 years ago. The latter trial famously saw no significant difference in death, MI, or stroke in patients with stable CAD assigned to OMT with or without PCI. That set off years of controversy about the relative merits of the revascularization and meds-only strategies in stable CAD that persists today.
But, Dr. Farkouh proposed, whether PCI improves clinical outcomes, compared with meds alone, at least in patients with diabetes, may be tied to the success of LDL-C-lowering therapies in reaching that goal, which in the current study was below 70 mg/dL.
“In this analysis of pooled data from the three major trials, we demonstrate that attaining that level of LDL-C at 1 year portends a better outcome for PCI” in patients with diabetes and stable CAD, he said.
The findings “probably need to be studied further, but it is compelling to think that if we can drive the LDL-C down by one year after the procedure, we have better outcomes with PCI,” compared with a meds-only strategy in patients with diabetes and stable CAD. “That really vindicates a lot of those who believe in PCI,” Dr. Farkouh said.
“What’s surprising to me is, if the patient has an LDL less than 70, why is it that there is a benefit of PCI, compared to medical therapy alone? Because they’re already so aggressively managed, you would think there shouldn’t be a benefit,” Sripal Bangalore, MD, MHA, New York University, said in an interview. “For me, that part is difficult to understand.”
The finding somewhat contradicts the results of ISCHEMIA, in which OMT – including LDL-C-lowering therapy – was considered more aggressive than usually managed in practice, Bangalore said. Yet the trial saw no outcomes difference between PCI and the more conservative approach, leading some to speculate that PCI may be a better choice when, for whatever reason, medical therapy isn’t optimal.
The observed superiority of PCI over meds-only at the lowest LDL-C levels is, according to Dr. Banagalore, “more likely because of residual confounding, given the fact that they’re combining three different trials, which are aimed to address different sets of questions.” He was an investigator with the FREEDOM and ISCHEMIA trials but isn’t associated with the current report.
The main message from this observational analysis is that “of course, we want to get the LDL as low as possible in these patients with demonstrated cardiovascular disease and diabetes,” Donald M. Lloyd-Jones, MD, ScM, Northwestern University, Chicago, said in an interview. “Every one of these patients should be shooting for as low an LDL as possible.”
Regardless of revascularization strategy, he said, “we have to get people on a high-intensity statin, or at least their maximally targeted dose, and have a careful and thoughtful conversation about whether they need additional lowering with, perhaps, ezetimibe, if they’re not below the thresholds we’d like to see them at, in this case, 70 mg/dL.”
Still, the current findings that the relative effects of PCI and CABG in these patients may vary by degree of LDL-C reduction “are interesting, but would have to be tested a little bit more directly,” said Dr. Lloyd-Jones, who is not affiliated with the analysis.
An accompanying editorial, which also acknowledges the study’s limitations, says its results “are relevant for clinical practice and may pave the way toward the generation of novel personalized medicine models that can optimize care of patients with type-2 diabetes.”
They “support the concept of an individualized treatment strategy that accounts for a patient’s LDL-C level to estimate clinical outcomes and expected treatment effects after therapeutic interventions,” say the authors, led by Eliano P. Navarese, MD, PhD, Nicolaus Copernicus University, Bydgoszcz, Poland.
“For daily practice, these results also underscore the importance of follow-up LDL-C measurements, both as a risk stratifier and as an indicator for therapy adjustments,” they write, noting that “current guidelines provide no formal recommendation on when to check LDL-C after PCI.”
The meta-analysis followed a total of 4050 patients with diabetes and stable CAD from the three randomized trials, those with evaluable baseline and follow-up LDL-C measurements, for a median of 4 years after the 1-year LDL-C assessment. At that time, at least 90% of patients in each of the trials had statin prescriptions, the group reported.
At one year, 34.5% of the total cohort had an LDL-C <70 mg/dL; their mean was 55.8 mg/dL.
And 42.2% had an LDL-C from 70 mg/dL to <100 mg/dL; their mean was 83.4 mg/dL. Compared with patients with an LDL-C <70 mg/dL, their adjusted hazard ratio for the composite endpoint was not elevated at 1.07 (95% CI, 0.86-1.32, P = .54).
Finally, 23.2% had an LDL-C ≥100 mg/dL; the mean was 123.0 mg/dL. Compared with the group with the lowest 1-year LDL-C, their adjusted HR for MACCE was increased at 1.46 (95% CI, 1.15 - 1.85, P = .002).
That HR among the 42.3% of patients in the PCI cohort, compared with the 33.3% assigned to meds only, climbed significantly only among those in the lowest 1-year LDL-C stratum: HR, 0.61 (95% CI, 0.40-0.91, P = .016). Corresponding HRs in the mid-range and highest 1-year LDL strata were close to unity and nonsignificant at P = .71 and P = .98, respectively.
On the other hand, the 24.4% of patients assigned to CABG showed better MACCE outcomes than those in the meds-only group across all three 1-year LDL-C strata.
The risk of MACCE wasn’t significantly altered by CABG, compared with PCI among patients achieving a 1-year LDL-C less than 70 mg/dL. However, it fell by about one-half for CABG vs. PCI in both the mid-range and highest 1-year LDL-C strata, P = .003 and P = .022, respectively.
Dr. Bangalore said he’s entirely behind the results of the study’s comparison of PCI and CABG. “It’s exactly the hypothesis that I’ve been putting forward, that if you want to achieve results as good as CABG, do PCI with aggressive medical therapy.” That means second-generation drug-eluting stents for the target lesions, “and aggressive medical therapy to address all of the nontarget lesions, specifically in diabetics.”
It’s possible, Dr. Lloyd-Jones said, that there is “no longer a dichotomy between revascularization strategies,” with respect to clinical outcomes, in such patients who maintain an LDL less than 70 mg/dL, as the study suggests.
“But I wonder, if it had continued for another 4 years of follow-up, whether we would see the CABG patients start to have more events,” such that the CABG advantage goes away at higher LDL-C levels, he proposed.
Or, Dr. Lloyd-Jones speculated, if all patients had achieved LDL-C below 70 mg/dL, “would there be such a difference between the PCI and CABG groups? My bet would be that it would be small or abolished.”
Dr. Farkouh discloses receiving research grants from Amgen, Novo Nordisk, and Novartis. Disclosures for the other study authors can be found with the original article. Editorialist Dr. Navarese discloses receiving consulting fees or honoraria from Abbott, AstraZeneca, Amgen, Bayer, Sanofi, and Pfizer; and grants from Abbott and Amgen. Dr. Lloyd-Jones has disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
In order for percutaneous coronary intervention (PCI) to shine, compared with meds alone in patients with type-2 diabetes and stable coronary disease (CAD), it needs help from aggressive control of LDL cholesterol (LDL-C) levels, suggests a patient-level meta-analysis of three major randomized trials.
Performing PCI in such patients with diabetes conferred further benefit over optimal medical therapy (OMT) for major adverse cardiac or cerebrovascular events (MACCE) only among those whose LDL-C levels had been pushed below the guidelines-specified threshold of 70 mg/dL within 1 year.
At that level of LDL-C control, PCI, compared with the meds-alone strategy, was followed by a nearly 40% drop in 4-year risk for the composite endpoint, which consisted of death from any cause or nonfatal myocardial infarction (MI) or stroke.
Also for patients reaching a 1-year LDL-C of <70 mg/dL, the risk of MACCE was similar for those who had been assigned to coronary bypass surgery (CABG), compared with PCI. But that risk was significantly lower for the CABG group among those reaching LDL-C levels above that threshold.
“The strategy of revascularization with the LDL lowering, that’s the combination that seems to be a winner” in such patients with diabetes and stable CAD, lead author Michael E. Farkouh, MD, MSc, said in an interview.
If their LDL-C “stays above 70 mg/dL, they don’t really enjoy any benefit of PCI. It’s a message to our interventional community to really drive that LDL down,” said Dr. Farkouh, of the University of Toronto. “Not only with statins, but perhaps with PCSK9 inhibitors, ezetimibe, and other therapies to lower that LDL-C.”
The analysis, published Nov. 2 in the Journal of the American College of Cardiology, pooled more than 4,000 patients with diabetes and stable CAD randomized in the BARI 2D, FREEDOM, and COURAGE trials.
The new study adds a twist to an ongoing theme throughout some meta-analyses and clinical trials like ISCHEMIA since the results of COURAGE were unveiled 13 years ago. The latter trial famously saw no significant difference in death, MI, or stroke in patients with stable CAD assigned to OMT with or without PCI. That set off years of controversy about the relative merits of the revascularization and meds-only strategies in stable CAD that persists today.
But, Dr. Farkouh proposed, whether PCI improves clinical outcomes, compared with meds alone, at least in patients with diabetes, may be tied to the success of LDL-C-lowering therapies in reaching that goal, which in the current study was below 70 mg/dL.
“In this analysis of pooled data from the three major trials, we demonstrate that attaining that level of LDL-C at 1 year portends a better outcome for PCI” in patients with diabetes and stable CAD, he said.
The findings “probably need to be studied further, but it is compelling to think that if we can drive the LDL-C down by one year after the procedure, we have better outcomes with PCI,” compared with a meds-only strategy in patients with diabetes and stable CAD. “That really vindicates a lot of those who believe in PCI,” Dr. Farkouh said.
“What’s surprising to me is, if the patient has an LDL less than 70, why is it that there is a benefit of PCI, compared to medical therapy alone? Because they’re already so aggressively managed, you would think there shouldn’t be a benefit,” Sripal Bangalore, MD, MHA, New York University, said in an interview. “For me, that part is difficult to understand.”
The finding somewhat contradicts the results of ISCHEMIA, in which OMT – including LDL-C-lowering therapy – was considered more aggressive than usually managed in practice, Bangalore said. Yet the trial saw no outcomes difference between PCI and the more conservative approach, leading some to speculate that PCI may be a better choice when, for whatever reason, medical therapy isn’t optimal.
The observed superiority of PCI over meds-only at the lowest LDL-C levels is, according to Dr. Banagalore, “more likely because of residual confounding, given the fact that they’re combining three different trials, which are aimed to address different sets of questions.” He was an investigator with the FREEDOM and ISCHEMIA trials but isn’t associated with the current report.
The main message from this observational analysis is that “of course, we want to get the LDL as low as possible in these patients with demonstrated cardiovascular disease and diabetes,” Donald M. Lloyd-Jones, MD, ScM, Northwestern University, Chicago, said in an interview. “Every one of these patients should be shooting for as low an LDL as possible.”
Regardless of revascularization strategy, he said, “we have to get people on a high-intensity statin, or at least their maximally targeted dose, and have a careful and thoughtful conversation about whether they need additional lowering with, perhaps, ezetimibe, if they’re not below the thresholds we’d like to see them at, in this case, 70 mg/dL.”
Still, the current findings that the relative effects of PCI and CABG in these patients may vary by degree of LDL-C reduction “are interesting, but would have to be tested a little bit more directly,” said Dr. Lloyd-Jones, who is not affiliated with the analysis.
An accompanying editorial, which also acknowledges the study’s limitations, says its results “are relevant for clinical practice and may pave the way toward the generation of novel personalized medicine models that can optimize care of patients with type-2 diabetes.”
They “support the concept of an individualized treatment strategy that accounts for a patient’s LDL-C level to estimate clinical outcomes and expected treatment effects after therapeutic interventions,” say the authors, led by Eliano P. Navarese, MD, PhD, Nicolaus Copernicus University, Bydgoszcz, Poland.
“For daily practice, these results also underscore the importance of follow-up LDL-C measurements, both as a risk stratifier and as an indicator for therapy adjustments,” they write, noting that “current guidelines provide no formal recommendation on when to check LDL-C after PCI.”
The meta-analysis followed a total of 4050 patients with diabetes and stable CAD from the three randomized trials, those with evaluable baseline and follow-up LDL-C measurements, for a median of 4 years after the 1-year LDL-C assessment. At that time, at least 90% of patients in each of the trials had statin prescriptions, the group reported.
At one year, 34.5% of the total cohort had an LDL-C <70 mg/dL; their mean was 55.8 mg/dL.
And 42.2% had an LDL-C from 70 mg/dL to <100 mg/dL; their mean was 83.4 mg/dL. Compared with patients with an LDL-C <70 mg/dL, their adjusted hazard ratio for the composite endpoint was not elevated at 1.07 (95% CI, 0.86-1.32, P = .54).
Finally, 23.2% had an LDL-C ≥100 mg/dL; the mean was 123.0 mg/dL. Compared with the group with the lowest 1-year LDL-C, their adjusted HR for MACCE was increased at 1.46 (95% CI, 1.15 - 1.85, P = .002).
That HR among the 42.3% of patients in the PCI cohort, compared with the 33.3% assigned to meds only, climbed significantly only among those in the lowest 1-year LDL-C stratum: HR, 0.61 (95% CI, 0.40-0.91, P = .016). Corresponding HRs in the mid-range and highest 1-year LDL strata were close to unity and nonsignificant at P = .71 and P = .98, respectively.
On the other hand, the 24.4% of patients assigned to CABG showed better MACCE outcomes than those in the meds-only group across all three 1-year LDL-C strata.
The risk of MACCE wasn’t significantly altered by CABG, compared with PCI among patients achieving a 1-year LDL-C less than 70 mg/dL. However, it fell by about one-half for CABG vs. PCI in both the mid-range and highest 1-year LDL-C strata, P = .003 and P = .022, respectively.
Dr. Bangalore said he’s entirely behind the results of the study’s comparison of PCI and CABG. “It’s exactly the hypothesis that I’ve been putting forward, that if you want to achieve results as good as CABG, do PCI with aggressive medical therapy.” That means second-generation drug-eluting stents for the target lesions, “and aggressive medical therapy to address all of the nontarget lesions, specifically in diabetics.”
It’s possible, Dr. Lloyd-Jones said, that there is “no longer a dichotomy between revascularization strategies,” with respect to clinical outcomes, in such patients who maintain an LDL less than 70 mg/dL, as the study suggests.
“But I wonder, if it had continued for another 4 years of follow-up, whether we would see the CABG patients start to have more events,” such that the CABG advantage goes away at higher LDL-C levels, he proposed.
Or, Dr. Lloyd-Jones speculated, if all patients had achieved LDL-C below 70 mg/dL, “would there be such a difference between the PCI and CABG groups? My bet would be that it would be small or abolished.”
Dr. Farkouh discloses receiving research grants from Amgen, Novo Nordisk, and Novartis. Disclosures for the other study authors can be found with the original article. Editorialist Dr. Navarese discloses receiving consulting fees or honoraria from Abbott, AstraZeneca, Amgen, Bayer, Sanofi, and Pfizer; and grants from Abbott and Amgen. Dr. Lloyd-Jones has disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
Proposed Medicare rule would expand CGM coverage
A new proposed rule from the Centers for Medicare & Medicaid Services (CMS) would expand coverage for continuous glucose monitors (CGMs) under Medicare to include devices that aren’t approved for making treatment decisions.
If accepted, the proposed rule would classify all approved CGMs as durable medical equipment under Medicare Part B and establish payment amounts for all related supplies. The move primarily affects Medtronic’s Guardian Connect System, which has not been approved by the U.S. Food and Drug Administration to replace the need for fingersticks in determining insulin or other glucose-lowering medication dosing.
Two other CGM systems, the Dexcom G6 and Abbott Libre, have “therapeutic” indications and are, therefore, already covered under Medicare, as is the combined insulin pump–CGM Tandem Diabetes Care Control-IQ Technology system.
According to a CMS statement, “CGMs that are not approved for use in making diabetes treatment decisions can be used to alert beneficiaries about potentially dangerous glucose levels while they sleep and that they should further test their glucose levels using a blood glucose monitor. ... This proposal would give Medicare beneficiaries and their physicians a wider range of technology and devices to choose from in managing diabetes.”
Sean Salmon, executive vice president and president of the Diabetes Group at Medtronic said in an interview that the company is “very encouraged” by the proposal. “Importantly, the proposed rule would enable continuity of therapy for people on Medtronic insulin pumps aging into Medicare – including Medtronic hybrid closed loop systems, which automatically adjust insulin delivery based on readings from the integrated CGM.”
The type 1 diabetes research and advocacy organization JDRF also applauded the proposed rule, noting in a statement, “CGM technology can be an integral component of artificial pancreas systems and important on its own to significantly improve diabetes management and enable users to avoid potential crises and risks for long-term complications. JDRF is heartened by this proposed change as it has long advocated for coverage, affordability and choice of all therapies to help ensure people with T1D have what they need to survive.”
The proposal is part of a broader set of proposed changes to Medicare Durable Medical Equipment, Prosthetics, Orthotic Devices and Supplies (DMEPOS) coverage and payment policies. Comments on the entire document can be submitted through Jan. 4, 2021 to the Federal Register.
A new proposed rule from the Centers for Medicare & Medicaid Services (CMS) would expand coverage for continuous glucose monitors (CGMs) under Medicare to include devices that aren’t approved for making treatment decisions.
If accepted, the proposed rule would classify all approved CGMs as durable medical equipment under Medicare Part B and establish payment amounts for all related supplies. The move primarily affects Medtronic’s Guardian Connect System, which has not been approved by the U.S. Food and Drug Administration to replace the need for fingersticks in determining insulin or other glucose-lowering medication dosing.
Two other CGM systems, the Dexcom G6 and Abbott Libre, have “therapeutic” indications and are, therefore, already covered under Medicare, as is the combined insulin pump–CGM Tandem Diabetes Care Control-IQ Technology system.
According to a CMS statement, “CGMs that are not approved for use in making diabetes treatment decisions can be used to alert beneficiaries about potentially dangerous glucose levels while they sleep and that they should further test their glucose levels using a blood glucose monitor. ... This proposal would give Medicare beneficiaries and their physicians a wider range of technology and devices to choose from in managing diabetes.”
Sean Salmon, executive vice president and president of the Diabetes Group at Medtronic said in an interview that the company is “very encouraged” by the proposal. “Importantly, the proposed rule would enable continuity of therapy for people on Medtronic insulin pumps aging into Medicare – including Medtronic hybrid closed loop systems, which automatically adjust insulin delivery based on readings from the integrated CGM.”
The type 1 diabetes research and advocacy organization JDRF also applauded the proposed rule, noting in a statement, “CGM technology can be an integral component of artificial pancreas systems and important on its own to significantly improve diabetes management and enable users to avoid potential crises and risks for long-term complications. JDRF is heartened by this proposed change as it has long advocated for coverage, affordability and choice of all therapies to help ensure people with T1D have what they need to survive.”
The proposal is part of a broader set of proposed changes to Medicare Durable Medical Equipment, Prosthetics, Orthotic Devices and Supplies (DMEPOS) coverage and payment policies. Comments on the entire document can be submitted through Jan. 4, 2021 to the Federal Register.
A new proposed rule from the Centers for Medicare & Medicaid Services (CMS) would expand coverage for continuous glucose monitors (CGMs) under Medicare to include devices that aren’t approved for making treatment decisions.
If accepted, the proposed rule would classify all approved CGMs as durable medical equipment under Medicare Part B and establish payment amounts for all related supplies. The move primarily affects Medtronic’s Guardian Connect System, which has not been approved by the U.S. Food and Drug Administration to replace the need for fingersticks in determining insulin or other glucose-lowering medication dosing.
Two other CGM systems, the Dexcom G6 and Abbott Libre, have “therapeutic” indications and are, therefore, already covered under Medicare, as is the combined insulin pump–CGM Tandem Diabetes Care Control-IQ Technology system.
According to a CMS statement, “CGMs that are not approved for use in making diabetes treatment decisions can be used to alert beneficiaries about potentially dangerous glucose levels while they sleep and that they should further test their glucose levels using a blood glucose monitor. ... This proposal would give Medicare beneficiaries and their physicians a wider range of technology and devices to choose from in managing diabetes.”
Sean Salmon, executive vice president and president of the Diabetes Group at Medtronic said in an interview that the company is “very encouraged” by the proposal. “Importantly, the proposed rule would enable continuity of therapy for people on Medtronic insulin pumps aging into Medicare – including Medtronic hybrid closed loop systems, which automatically adjust insulin delivery based on readings from the integrated CGM.”
The type 1 diabetes research and advocacy organization JDRF also applauded the proposed rule, noting in a statement, “CGM technology can be an integral component of artificial pancreas systems and important on its own to significantly improve diabetes management and enable users to avoid potential crises and risks for long-term complications. JDRF is heartened by this proposed change as it has long advocated for coverage, affordability and choice of all therapies to help ensure people with T1D have what they need to survive.”
The proposal is part of a broader set of proposed changes to Medicare Durable Medical Equipment, Prosthetics, Orthotic Devices and Supplies (DMEPOS) coverage and payment policies. Comments on the entire document can be submitted through Jan. 4, 2021 to the Federal Register.
Medication adherence challenges and helpers
For most chronic diseases, up to 20%-30% of the pills that are prescribed are not taken. In the case of inhalers for asthma and COPD, patients miss over half of the prescribed doses.
There are many things that contribute to the problem of poor adherence, but people often just simply forget. Thankfully, there are tools designed to help remind patients of what they need to take and when. A survey of apps developed to help patients remember to take their medicines found more than 700 available in Apple and Android app stores.1 Most apps focus on medication alerts, reminders, and medication logs.2 A recent review showed that apps have some – yet limited – effectiveness in increasing adherence, with patient self-reported improvements of 7%-40%.3
Another perhaps more promising area of improving adherence involves high-tech advances in the way medications can be taken. Inhalers are a primary target as they are complicated devices. A patient has to breathe in at the correct time after the inhaler is actuated, and the inhaler works optimally only if the rate of inhalation is sufficient to carry the medication into the lungs.
A number of companies have developed attachments for inhalers (and even inhalers themselves) that can record when the medication is taken through a Bluetooth connection to a patient’s smartphone. These can also assess inspiratory flow. Reminders to take the medication are built into the app, and those reminders disappear if the medication is taken. Patients can receive feedback about the quality of their timing and inspiratory rate to maximize medication delivery to the lungs.4
We learned long ago that it is difficult to take medications three to four times a day, so extended-release tablets were developed to reduce the frequency to once or twice a day. A great deal of work is now being done behind the scenes to develop medications that decrease the need for patients to remember to take their medications. The best examples of this are the long-acting reversible contraception (LARC) devices, specifically IUDs and Nexplanon. Compared with traditional oral contraceptives that need to be taken daily, LARCs reduce the rate of pregnancy by five- to tenfold.
We also now have medications for osteoporosis that can be taken monthly, or even annually. When bisphosphonates were first developed for osteoporosis prevention, they needed to be taken daily. Then a weekly bisphosphonate was developed. Now there is a once-monthly oral bisphosphonate, Ibandronate, and even a once yearly IV bisphosphonate.
Exciting developments have also occurred in the management of diabetes. We may be tempted to take for granted how once-daily long-acting insulin, which releases insulin slowly over the course of a day, has revolutionized the diabetic treatment since its Food and Drug Administration approval in 2000. Yet progress did not end there. The first GLP-1 receptor agonist for diabetes was approved in 2005 and was a twice-a-day medicine. Shortly afterward, a daily GLP-1 was approved, and now there are three once-weekly GLP-1 receptor agonists.
Several pharmaceutical manufacturers are now working on a once-weekly insulin,5 as well as an implantable GLP-1 receptor agonist that will need to be replaced every 6-12 months.6 Imagine your patient coming in once a year to replace his or her potent glucose lowering medication – one that offers a low incidence of hypoglycemia, maintains glucose control all year long, and requires no adherence to a complicated medication regimen.
Similar technology is being used to develop a once-yearly anti-HIV prophylactic medication delivery system.7 This could help prevent the spread of HIV in areas of the world where it may be difficult for people to take daily medications.7
The many technological advances we have described may help us reduce our likelihood of missing a dose of a medication. We are hopeful that progress in this area will continue, and that one day medication adherence will require even less effort from patients than it does today.
Dr. Notte is a family physician and chief medical officer of Abington (Pa.) Hospital–Jefferson Health. Follow him on Twitter (@doctornotte). Dr. Skolnik is professor of family and community medicine at Sidney Kimmel Medical College, Philadelphia, and associate director of the family medicine residency program at Abington Hospital–Jefferson Health. They have no conflicts related to the content of this piece.
References
1. Tabi K et al. Mobile apps for medication management: Review and analysis. JMIR Mhealth Uhealth. 2019 Sep 7(9):13608.
2. Park JYE et al. Mobile phone apps targeting medication adherence: Quality assessment and content analysis of user reviews. JMIR Mhealth Uhealth. 2019 Jan 31;7(1):e11919.
3. Pérez-Jover V et al. Mobile apps for increasing treatment adherence: Systematic review. J Med Internet Res. 2019;21(6):e12505. doi: 10.2196/12505.
4. 4 Smart inhalers that could be lifesaving for people living with asthma & COPD. MyTherapy, July 11, 2019.
5. Rosenstock J et al. Once-weekly insulin for type 2 diabetes without previous insulin treatment. N Engl J Med. 2020 Sep 22. doi: 10.1056/NEJMoa2022474.
6. GLP-1 agonists: From 2 daily injections to 1 per week and beyond. DiaTribe, Jan. 10, 2018.
7. Long-acting HIV prevention tools. Hiv.gov, July 20, 2019.
For most chronic diseases, up to 20%-30% of the pills that are prescribed are not taken. In the case of inhalers for asthma and COPD, patients miss over half of the prescribed doses.
There are many things that contribute to the problem of poor adherence, but people often just simply forget. Thankfully, there are tools designed to help remind patients of what they need to take and when. A survey of apps developed to help patients remember to take their medicines found more than 700 available in Apple and Android app stores.1 Most apps focus on medication alerts, reminders, and medication logs.2 A recent review showed that apps have some – yet limited – effectiveness in increasing adherence, with patient self-reported improvements of 7%-40%.3
Another perhaps more promising area of improving adherence involves high-tech advances in the way medications can be taken. Inhalers are a primary target as they are complicated devices. A patient has to breathe in at the correct time after the inhaler is actuated, and the inhaler works optimally only if the rate of inhalation is sufficient to carry the medication into the lungs.
A number of companies have developed attachments for inhalers (and even inhalers themselves) that can record when the medication is taken through a Bluetooth connection to a patient’s smartphone. These can also assess inspiratory flow. Reminders to take the medication are built into the app, and those reminders disappear if the medication is taken. Patients can receive feedback about the quality of their timing and inspiratory rate to maximize medication delivery to the lungs.4
We learned long ago that it is difficult to take medications three to four times a day, so extended-release tablets were developed to reduce the frequency to once or twice a day. A great deal of work is now being done behind the scenes to develop medications that decrease the need for patients to remember to take their medications. The best examples of this are the long-acting reversible contraception (LARC) devices, specifically IUDs and Nexplanon. Compared with traditional oral contraceptives that need to be taken daily, LARCs reduce the rate of pregnancy by five- to tenfold.
We also now have medications for osteoporosis that can be taken monthly, or even annually. When bisphosphonates were first developed for osteoporosis prevention, they needed to be taken daily. Then a weekly bisphosphonate was developed. Now there is a once-monthly oral bisphosphonate, Ibandronate, and even a once yearly IV bisphosphonate.
Exciting developments have also occurred in the management of diabetes. We may be tempted to take for granted how once-daily long-acting insulin, which releases insulin slowly over the course of a day, has revolutionized the diabetic treatment since its Food and Drug Administration approval in 2000. Yet progress did not end there. The first GLP-1 receptor agonist for diabetes was approved in 2005 and was a twice-a-day medicine. Shortly afterward, a daily GLP-1 was approved, and now there are three once-weekly GLP-1 receptor agonists.
Several pharmaceutical manufacturers are now working on a once-weekly insulin,5 as well as an implantable GLP-1 receptor agonist that will need to be replaced every 6-12 months.6 Imagine your patient coming in once a year to replace his or her potent glucose lowering medication – one that offers a low incidence of hypoglycemia, maintains glucose control all year long, and requires no adherence to a complicated medication regimen.
Similar technology is being used to develop a once-yearly anti-HIV prophylactic medication delivery system.7 This could help prevent the spread of HIV in areas of the world where it may be difficult for people to take daily medications.7
The many technological advances we have described may help us reduce our likelihood of missing a dose of a medication. We are hopeful that progress in this area will continue, and that one day medication adherence will require even less effort from patients than it does today.
Dr. Notte is a family physician and chief medical officer of Abington (Pa.) Hospital–Jefferson Health. Follow him on Twitter (@doctornotte). Dr. Skolnik is professor of family and community medicine at Sidney Kimmel Medical College, Philadelphia, and associate director of the family medicine residency program at Abington Hospital–Jefferson Health. They have no conflicts related to the content of this piece.
References
1. Tabi K et al. Mobile apps for medication management: Review and analysis. JMIR Mhealth Uhealth. 2019 Sep 7(9):13608.
2. Park JYE et al. Mobile phone apps targeting medication adherence: Quality assessment and content analysis of user reviews. JMIR Mhealth Uhealth. 2019 Jan 31;7(1):e11919.
3. Pérez-Jover V et al. Mobile apps for increasing treatment adherence: Systematic review. J Med Internet Res. 2019;21(6):e12505. doi: 10.2196/12505.
4. 4 Smart inhalers that could be lifesaving for people living with asthma & COPD. MyTherapy, July 11, 2019.
5. Rosenstock J et al. Once-weekly insulin for type 2 diabetes without previous insulin treatment. N Engl J Med. 2020 Sep 22. doi: 10.1056/NEJMoa2022474.
6. GLP-1 agonists: From 2 daily injections to 1 per week and beyond. DiaTribe, Jan. 10, 2018.
7. Long-acting HIV prevention tools. Hiv.gov, July 20, 2019.
For most chronic diseases, up to 20%-30% of the pills that are prescribed are not taken. In the case of inhalers for asthma and COPD, patients miss over half of the prescribed doses.
There are many things that contribute to the problem of poor adherence, but people often just simply forget. Thankfully, there are tools designed to help remind patients of what they need to take and when. A survey of apps developed to help patients remember to take their medicines found more than 700 available in Apple and Android app stores.1 Most apps focus on medication alerts, reminders, and medication logs.2 A recent review showed that apps have some – yet limited – effectiveness in increasing adherence, with patient self-reported improvements of 7%-40%.3
Another perhaps more promising area of improving adherence involves high-tech advances in the way medications can be taken. Inhalers are a primary target as they are complicated devices. A patient has to breathe in at the correct time after the inhaler is actuated, and the inhaler works optimally only if the rate of inhalation is sufficient to carry the medication into the lungs.
A number of companies have developed attachments for inhalers (and even inhalers themselves) that can record when the medication is taken through a Bluetooth connection to a patient’s smartphone. These can also assess inspiratory flow. Reminders to take the medication are built into the app, and those reminders disappear if the medication is taken. Patients can receive feedback about the quality of their timing and inspiratory rate to maximize medication delivery to the lungs.4
We learned long ago that it is difficult to take medications three to four times a day, so extended-release tablets were developed to reduce the frequency to once or twice a day. A great deal of work is now being done behind the scenes to develop medications that decrease the need for patients to remember to take their medications. The best examples of this are the long-acting reversible contraception (LARC) devices, specifically IUDs and Nexplanon. Compared with traditional oral contraceptives that need to be taken daily, LARCs reduce the rate of pregnancy by five- to tenfold.
We also now have medications for osteoporosis that can be taken monthly, or even annually. When bisphosphonates were first developed for osteoporosis prevention, they needed to be taken daily. Then a weekly bisphosphonate was developed. Now there is a once-monthly oral bisphosphonate, Ibandronate, and even a once yearly IV bisphosphonate.
Exciting developments have also occurred in the management of diabetes. We may be tempted to take for granted how once-daily long-acting insulin, which releases insulin slowly over the course of a day, has revolutionized the diabetic treatment since its Food and Drug Administration approval in 2000. Yet progress did not end there. The first GLP-1 receptor agonist for diabetes was approved in 2005 and was a twice-a-day medicine. Shortly afterward, a daily GLP-1 was approved, and now there are three once-weekly GLP-1 receptor agonists.
Several pharmaceutical manufacturers are now working on a once-weekly insulin,5 as well as an implantable GLP-1 receptor agonist that will need to be replaced every 6-12 months.6 Imagine your patient coming in once a year to replace his or her potent glucose lowering medication – one that offers a low incidence of hypoglycemia, maintains glucose control all year long, and requires no adherence to a complicated medication regimen.
Similar technology is being used to develop a once-yearly anti-HIV prophylactic medication delivery system.7 This could help prevent the spread of HIV in areas of the world where it may be difficult for people to take daily medications.7
The many technological advances we have described may help us reduce our likelihood of missing a dose of a medication. We are hopeful that progress in this area will continue, and that one day medication adherence will require even less effort from patients than it does today.
Dr. Notte is a family physician and chief medical officer of Abington (Pa.) Hospital–Jefferson Health. Follow him on Twitter (@doctornotte). Dr. Skolnik is professor of family and community medicine at Sidney Kimmel Medical College, Philadelphia, and associate director of the family medicine residency program at Abington Hospital–Jefferson Health. They have no conflicts related to the content of this piece.
References
1. Tabi K et al. Mobile apps for medication management: Review and analysis. JMIR Mhealth Uhealth. 2019 Sep 7(9):13608.
2. Park JYE et al. Mobile phone apps targeting medication adherence: Quality assessment and content analysis of user reviews. JMIR Mhealth Uhealth. 2019 Jan 31;7(1):e11919.
3. Pérez-Jover V et al. Mobile apps for increasing treatment adherence: Systematic review. J Med Internet Res. 2019;21(6):e12505. doi: 10.2196/12505.
4. 4 Smart inhalers that could be lifesaving for people living with asthma & COPD. MyTherapy, July 11, 2019.
5. Rosenstock J et al. Once-weekly insulin for type 2 diabetes without previous insulin treatment. N Engl J Med. 2020 Sep 22. doi: 10.1056/NEJMoa2022474.
6. GLP-1 agonists: From 2 daily injections to 1 per week and beyond. DiaTribe, Jan. 10, 2018.
7. Long-acting HIV prevention tools. Hiv.gov, July 20, 2019.
First-of-its kind guideline on lipid monitoring in endocrine diseases
Endocrine diseases of any type – not just diabetes – can represent a cardiovascular risk and patients with those disorders should be screened for high cholesterol, according to a new clinical practice guideline from the Endocrine Society.
“The simple recommendation to check a lipid panel in patients with endocrine diseases and calculate cardiovascular risk may be practice changing because that is not done routinely,” Connie Newman, MD, chair of the Endocrine Society committee that developed the guideline, said in an interview.
“Usually the focus is on assessment and treatment of the endocrine disease, rather than on assessment and treatment of atherosclerotic cardiovascular disease risk,” said Newman, an adjunct professor of medicine in the department of medicine, division of endocrinology, diabetes & metabolism, at New York University.
Whereas diabetes, well-known for its increased cardiovascular risk profile, is commonly addressed in other cardiovascular and cholesterol practice management guidelines, the array of other endocrine diseases are not typically included.
“This guideline is the first of its kind,” Dr. Newman said. “The Endocrine Society has not previously issued a guideline on lipid management in endocrine disorders [and] other organizations have not written guidelines on this topic.
“Rather, guidelines have been written on cholesterol management, but these do not describe cholesterol management in patients with endocrine diseases such as thyroid disease [hypothyroidism and hyperthyroidism], Cushing’s syndrome, acromegaly, growth hormone deficiency, menopause, male hypogonadism, and obesity,” she noted.
But these conditions carry a host of cardiovascular risk factors that may require careful monitoring and management.
“Although endocrine hormones, such as thyroid hormone, cortisol, estrogen, testosterone, growth hormone, and insulin, affect pathways for lipid metabolism, physicians lack guidance on lipid abnormalities, cardiovascular risk, and treatment to reduce lipids and cardiovascular risk in patients with endocrine diseases,” she explained.
Vinaya Simha, MD, an internal medicine specialist at the Mayo Clinic in Rochester, Minn., agrees that the guideline is notable in addressing an unmet need.
Recommendations that stand out to Dr. Simha include the suggestion of adding eicosapentaenoic acid (EPA) ethyl ester to reduce the risk of cardiovascular disease in adults with diabetes or atherosclerotic cardiovascular disease who have elevated triglyceride levels despite statin treatment.
James L. Rosenzweig, MD, an endocrinologist at Hebrew SeniorLife in Boston, agreed that this is an important addition to an area that needs more guidance.
“Many of these clinical situations can exacerbate dyslipidemia and some also increase the cardiovascular risk to a greater extent in combination with elevated cholesterol and/or triglycerides,” he said in an interview.
“In many cases, treatment of the underlying disorder appropriately can have an important impact in resolving the lipid disorder. In others, more aggressive pharmacological treatment is indicated,” he said.
“I think that this will be a valuable resource, especially for endocrinologists, but it can be used as well by providers in other disciplines.”
Key recommendations for different endocrine conditions
The guideline, published in the Journal of Clinical Endocrinology & Metabolism, details those risks and provides evidence-based recommendations on their management and treatment.
Key recommendations include:
- Obtain a lipid panel and evaluate cardiovascular risk factors in all adults with endocrine disorders.
- In patients with and risk factors for cardiovascular disease, start statin therapy in addition to lifestyle modification to reduce cardiovascular risk. “This could mean earlier treatment because other guidelines recommend consideration of therapy at age 40,” Dr. Newman said.
- Statin therapy is also recommended for adults over 40 with with a duration of diabetes of more than 20 years and/or microvascular complications, regardless of their cardiovascular risk score. “This means earlier treatment of patients with type 1 diabetes with statins in order to reduce cardiovascular disease risk,” Dr. Newman noted.
- In patients with hyperlipidemia, rule out as the cause before treating with lipid-lowering medications. And among patients who are found to have hypothyroidism, reevaluate the lipid profile when the patient has thyroid hormone levels in the normal range.
- Adults with persistent endogenous Cushing’s syndrome should have their lipid profile monitored. Statin therapy should be considered in addition to lifestyle modifications, irrespective of the cardiovascular risk score.
- In postmenopausal women, high cholesterol or triglycerides should be treated with statins rather than hormone therapy.
- Evaluate and treat lipids and other cardiovascular risk factors in women who enter menopause early (before the age of 40-45 years).
Nice summary of ‘risk-enhancing’ endocrine disorders
Dr. Simha said in an interview that the new guideline is “probably the first comprehensive statement addressing lipid treatment in patients with a broad range of endocrine disorders besides diabetes.”
“Most of the treatment recommendations are congruent with other current guidelines such as the American College of Cardiology/American Heart Association [guidelines], but there is specific mention of which endocrine disorders represent enhanced cardiovascular risk,” she explained.
The new recommendations are notable for including “a nice summary of how different endocrine disorders affect lipid values, and also which endocrine disorders need to be considered as ‘risk-enhancing factors,’ ” Dr. Simha noted.
“The use of EPA in patients with hypertriglyceridemia is novel, compared to the ACC/AHA recommendation. This reflects new data which is now available,” she added.
The American Association of Clinical Endocrinologists also just issued a new algorithm on lipid management and prevention of cardiovascular disease in which treatment of hypertriglyceridemia is emphasized.
In addition, the new Endocrine Society guideline “also mentions an LDL [cholesterol] treatment threshold of 70 mg/dL, and 55 mg/dL in some patient categories, which previous guidelines have not,” Dr. Simha noted.
Overall, Dr. Newman added that the goal of the guideline is to increase awareness of key issues with endocrine diseases that may not necessarily be on clinicians’ radars.
“We hope that it will make a lipid panel and cardiovascular risk evaluation routine in adults with endocrine diseases and cause a greater focus on therapies to reduce heart disease and stroke,” she said.
Dr. Newman, Dr. Simha, and Dr. Rosenzweig reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Endocrine diseases of any type – not just diabetes – can represent a cardiovascular risk and patients with those disorders should be screened for high cholesterol, according to a new clinical practice guideline from the Endocrine Society.
“The simple recommendation to check a lipid panel in patients with endocrine diseases and calculate cardiovascular risk may be practice changing because that is not done routinely,” Connie Newman, MD, chair of the Endocrine Society committee that developed the guideline, said in an interview.
“Usually the focus is on assessment and treatment of the endocrine disease, rather than on assessment and treatment of atherosclerotic cardiovascular disease risk,” said Newman, an adjunct professor of medicine in the department of medicine, division of endocrinology, diabetes & metabolism, at New York University.
Whereas diabetes, well-known for its increased cardiovascular risk profile, is commonly addressed in other cardiovascular and cholesterol practice management guidelines, the array of other endocrine diseases are not typically included.
“This guideline is the first of its kind,” Dr. Newman said. “The Endocrine Society has not previously issued a guideline on lipid management in endocrine disorders [and] other organizations have not written guidelines on this topic.
“Rather, guidelines have been written on cholesterol management, but these do not describe cholesterol management in patients with endocrine diseases such as thyroid disease [hypothyroidism and hyperthyroidism], Cushing’s syndrome, acromegaly, growth hormone deficiency, menopause, male hypogonadism, and obesity,” she noted.
But these conditions carry a host of cardiovascular risk factors that may require careful monitoring and management.
“Although endocrine hormones, such as thyroid hormone, cortisol, estrogen, testosterone, growth hormone, and insulin, affect pathways for lipid metabolism, physicians lack guidance on lipid abnormalities, cardiovascular risk, and treatment to reduce lipids and cardiovascular risk in patients with endocrine diseases,” she explained.
Vinaya Simha, MD, an internal medicine specialist at the Mayo Clinic in Rochester, Minn., agrees that the guideline is notable in addressing an unmet need.
Recommendations that stand out to Dr. Simha include the suggestion of adding eicosapentaenoic acid (EPA) ethyl ester to reduce the risk of cardiovascular disease in adults with diabetes or atherosclerotic cardiovascular disease who have elevated triglyceride levels despite statin treatment.
James L. Rosenzweig, MD, an endocrinologist at Hebrew SeniorLife in Boston, agreed that this is an important addition to an area that needs more guidance.
“Many of these clinical situations can exacerbate dyslipidemia and some also increase the cardiovascular risk to a greater extent in combination with elevated cholesterol and/or triglycerides,” he said in an interview.
“In many cases, treatment of the underlying disorder appropriately can have an important impact in resolving the lipid disorder. In others, more aggressive pharmacological treatment is indicated,” he said.
“I think that this will be a valuable resource, especially for endocrinologists, but it can be used as well by providers in other disciplines.”
Key recommendations for different endocrine conditions
The guideline, published in the Journal of Clinical Endocrinology & Metabolism, details those risks and provides evidence-based recommendations on their management and treatment.
Key recommendations include:
- Obtain a lipid panel and evaluate cardiovascular risk factors in all adults with endocrine disorders.
- In patients with and risk factors for cardiovascular disease, start statin therapy in addition to lifestyle modification to reduce cardiovascular risk. “This could mean earlier treatment because other guidelines recommend consideration of therapy at age 40,” Dr. Newman said.
- Statin therapy is also recommended for adults over 40 with with a duration of diabetes of more than 20 years and/or microvascular complications, regardless of their cardiovascular risk score. “This means earlier treatment of patients with type 1 diabetes with statins in order to reduce cardiovascular disease risk,” Dr. Newman noted.
- In patients with hyperlipidemia, rule out as the cause before treating with lipid-lowering medications. And among patients who are found to have hypothyroidism, reevaluate the lipid profile when the patient has thyroid hormone levels in the normal range.
- Adults with persistent endogenous Cushing’s syndrome should have their lipid profile monitored. Statin therapy should be considered in addition to lifestyle modifications, irrespective of the cardiovascular risk score.
- In postmenopausal women, high cholesterol or triglycerides should be treated with statins rather than hormone therapy.
- Evaluate and treat lipids and other cardiovascular risk factors in women who enter menopause early (before the age of 40-45 years).
Nice summary of ‘risk-enhancing’ endocrine disorders
Dr. Simha said in an interview that the new guideline is “probably the first comprehensive statement addressing lipid treatment in patients with a broad range of endocrine disorders besides diabetes.”
“Most of the treatment recommendations are congruent with other current guidelines such as the American College of Cardiology/American Heart Association [guidelines], but there is specific mention of which endocrine disorders represent enhanced cardiovascular risk,” she explained.
The new recommendations are notable for including “a nice summary of how different endocrine disorders affect lipid values, and also which endocrine disorders need to be considered as ‘risk-enhancing factors,’ ” Dr. Simha noted.
“The use of EPA in patients with hypertriglyceridemia is novel, compared to the ACC/AHA recommendation. This reflects new data which is now available,” she added.
The American Association of Clinical Endocrinologists also just issued a new algorithm on lipid management and prevention of cardiovascular disease in which treatment of hypertriglyceridemia is emphasized.
In addition, the new Endocrine Society guideline “also mentions an LDL [cholesterol] treatment threshold of 70 mg/dL, and 55 mg/dL in some patient categories, which previous guidelines have not,” Dr. Simha noted.
Overall, Dr. Newman added that the goal of the guideline is to increase awareness of key issues with endocrine diseases that may not necessarily be on clinicians’ radars.
“We hope that it will make a lipid panel and cardiovascular risk evaluation routine in adults with endocrine diseases and cause a greater focus on therapies to reduce heart disease and stroke,” she said.
Dr. Newman, Dr. Simha, and Dr. Rosenzweig reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Endocrine diseases of any type – not just diabetes – can represent a cardiovascular risk and patients with those disorders should be screened for high cholesterol, according to a new clinical practice guideline from the Endocrine Society.
“The simple recommendation to check a lipid panel in patients with endocrine diseases and calculate cardiovascular risk may be practice changing because that is not done routinely,” Connie Newman, MD, chair of the Endocrine Society committee that developed the guideline, said in an interview.
“Usually the focus is on assessment and treatment of the endocrine disease, rather than on assessment and treatment of atherosclerotic cardiovascular disease risk,” said Newman, an adjunct professor of medicine in the department of medicine, division of endocrinology, diabetes & metabolism, at New York University.
Whereas diabetes, well-known for its increased cardiovascular risk profile, is commonly addressed in other cardiovascular and cholesterol practice management guidelines, the array of other endocrine diseases are not typically included.
“This guideline is the first of its kind,” Dr. Newman said. “The Endocrine Society has not previously issued a guideline on lipid management in endocrine disorders [and] other organizations have not written guidelines on this topic.
“Rather, guidelines have been written on cholesterol management, but these do not describe cholesterol management in patients with endocrine diseases such as thyroid disease [hypothyroidism and hyperthyroidism], Cushing’s syndrome, acromegaly, growth hormone deficiency, menopause, male hypogonadism, and obesity,” she noted.
But these conditions carry a host of cardiovascular risk factors that may require careful monitoring and management.
“Although endocrine hormones, such as thyroid hormone, cortisol, estrogen, testosterone, growth hormone, and insulin, affect pathways for lipid metabolism, physicians lack guidance on lipid abnormalities, cardiovascular risk, and treatment to reduce lipids and cardiovascular risk in patients with endocrine diseases,” she explained.
Vinaya Simha, MD, an internal medicine specialist at the Mayo Clinic in Rochester, Minn., agrees that the guideline is notable in addressing an unmet need.
Recommendations that stand out to Dr. Simha include the suggestion of adding eicosapentaenoic acid (EPA) ethyl ester to reduce the risk of cardiovascular disease in adults with diabetes or atherosclerotic cardiovascular disease who have elevated triglyceride levels despite statin treatment.
James L. Rosenzweig, MD, an endocrinologist at Hebrew SeniorLife in Boston, agreed that this is an important addition to an area that needs more guidance.
“Many of these clinical situations can exacerbate dyslipidemia and some also increase the cardiovascular risk to a greater extent in combination with elevated cholesterol and/or triglycerides,” he said in an interview.
“In many cases, treatment of the underlying disorder appropriately can have an important impact in resolving the lipid disorder. In others, more aggressive pharmacological treatment is indicated,” he said.
“I think that this will be a valuable resource, especially for endocrinologists, but it can be used as well by providers in other disciplines.”
Key recommendations for different endocrine conditions
The guideline, published in the Journal of Clinical Endocrinology & Metabolism, details those risks and provides evidence-based recommendations on their management and treatment.
Key recommendations include:
- Obtain a lipid panel and evaluate cardiovascular risk factors in all adults with endocrine disorders.
- In patients with and risk factors for cardiovascular disease, start statin therapy in addition to lifestyle modification to reduce cardiovascular risk. “This could mean earlier treatment because other guidelines recommend consideration of therapy at age 40,” Dr. Newman said.
- Statin therapy is also recommended for adults over 40 with with a duration of diabetes of more than 20 years and/or microvascular complications, regardless of their cardiovascular risk score. “This means earlier treatment of patients with type 1 diabetes with statins in order to reduce cardiovascular disease risk,” Dr. Newman noted.
- In patients with hyperlipidemia, rule out as the cause before treating with lipid-lowering medications. And among patients who are found to have hypothyroidism, reevaluate the lipid profile when the patient has thyroid hormone levels in the normal range.
- Adults with persistent endogenous Cushing’s syndrome should have their lipid profile monitored. Statin therapy should be considered in addition to lifestyle modifications, irrespective of the cardiovascular risk score.
- In postmenopausal women, high cholesterol or triglycerides should be treated with statins rather than hormone therapy.
- Evaluate and treat lipids and other cardiovascular risk factors in women who enter menopause early (before the age of 40-45 years).
Nice summary of ‘risk-enhancing’ endocrine disorders
Dr. Simha said in an interview that the new guideline is “probably the first comprehensive statement addressing lipid treatment in patients with a broad range of endocrine disorders besides diabetes.”
“Most of the treatment recommendations are congruent with other current guidelines such as the American College of Cardiology/American Heart Association [guidelines], but there is specific mention of which endocrine disorders represent enhanced cardiovascular risk,” she explained.
The new recommendations are notable for including “a nice summary of how different endocrine disorders affect lipid values, and also which endocrine disorders need to be considered as ‘risk-enhancing factors,’ ” Dr. Simha noted.
“The use of EPA in patients with hypertriglyceridemia is novel, compared to the ACC/AHA recommendation. This reflects new data which is now available,” she added.
The American Association of Clinical Endocrinologists also just issued a new algorithm on lipid management and prevention of cardiovascular disease in which treatment of hypertriglyceridemia is emphasized.
In addition, the new Endocrine Society guideline “also mentions an LDL [cholesterol] treatment threshold of 70 mg/dL, and 55 mg/dL in some patient categories, which previous guidelines have not,” Dr. Simha noted.
Overall, Dr. Newman added that the goal of the guideline is to increase awareness of key issues with endocrine diseases that may not necessarily be on clinicians’ radars.
“We hope that it will make a lipid panel and cardiovascular risk evaluation routine in adults with endocrine diseases and cause a greater focus on therapies to reduce heart disease and stroke,” she said.
Dr. Newman, Dr. Simha, and Dr. Rosenzweig reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Evaluation of Metabolic Monitoring and Diabetes Incidence in the First Year for Veterans Newly Started on an Atypical Antipsychotic
Atypical antipsychotic use may result in metabolic abnormalities, such as hyperglycemia, dyslipidemia, weight gain, and metabolic syndrome. These adverse effects (AEs) can cause progression to type 2 diabetes mellitus (T2DM) as well as increased risk of cardiovascular disease and cardiac mortality. Individuals diagnosed with T2DM have medical expenses that are about 2.3 times higher than individuals without diabetes.1,2 The risk of experiencing metabolic abnormalities is likely elevated for patients who were antipsychotic-naïve prior to initiation.3
In response to an increased awareness of atypical antipsychotic-related AEs, the American Diabetes Association (ADA) and American Psychiatric Association (APA) released a consensus statement in 2004 with a metabolic monitoring protocol for patients initiating or changing to a new antipsychotic medication.4 Within the first year after initiation, the ADA/APA consensus statements recommends that clinicians acquire a personal and family history, weight, body mass index (BMI), waist circumference, blood pressure (BP), fasting plasma glucose, and fasting lipid profile at the initial patient visit. Patient weight is recommended to be collected at 4 weeks and again 8 weeks later. Twelve weeks after the initial visit, weight, BMI, BP, fasting plasma glucose and a fasting lipid profile are recommended to be collected and assessed for abnormalities. Weight is then recommended to be assessed every 3 months thereafter. Review of personal and family history, waist circumference, BP, and a fasting plasma glucose is recommended to occur annually. Finally, a fasting lipid profile is to be collected every 5 years.
Since the initial consensus statement release, metabolic monitoring of patients prescribed antipsychotic medications has been found to be inadequate within several large health care organizations.5,6 Mittal and colleagues reviewed metabolic monitoring practices occurring in 32 facilities within the Veterans Health Administration (VHA) and found that monitoring practices in the first 90 days after antipsychotic initiation were largely nonadherent to the ADA/APA consensus statement recommendations.6 Medical staff in Veterans Integrated Service Network 21 (VISN 21) currently serve about 268,000 veterans actively receiving care across California, Nevada, and the Pacific Islands.To support veteran care in the fields of mental health and medication safety, the VISN 21 pharmacy benefits manager office created a clinical dashboard that identifies veterans who are currently prescribed an antipsychotic and have not completed at least 1 annual blood glucose test. While this dashboard is a valuable tool for tracking patient care for those who have been prescribed an antipsychotic > 1 year, it does not consider the ADA/APA recommendations for more frequent monitoring in the first year after initiation. A literature review found no citations of a systematic evaluation of adherence to ADA/APA monitoring recommendations or patient progression to T2DM in the first year after antipsychotic initiation for an antipsychotic-naïve veteran population. The goal of this quality improvement project is to assess VHA health care provider and patient adherence to the 2004 consensus statement recommendations within the first year after initiation for previously antipsychotic-naïve patients receiving an atypical antipsychotic and determine rate of progression to T2DM.
Methods
The project was reviewed by the University of Nevada-Reno Institutional Review Board and determined to be a nonresearch quality improvement project. This was a retrospective chart analysis that included patients receiving their first-ever atypical antipsychotic across 8 US Department of Veterans Affairs (VA) medical centers within VISN 21. Clinical patient data, including prescription, vital sign, and laboratory information, were extracted from the VA Corporate Data Warehouse using transact sequential query language.
Veterans were included in the final cohort if they met the following criteria: aged ≥ 18 years at antipsychotic initiation, initiated their first-ever atypical antipsychotic within the VHA between February 2014 and February 2019, continued the antipsychotic for ≥ 1 year, had a medication possession ratio (MPR) > 80%, and had previously established care within VHA as evidenced by having ≥ 1 primary care or outpatient mental health visit in the 6 months prior to initiation. The MPR is defined as the sum of the day’s supply of all dispensed medications in the project time frame divided by the total number of days in the project time frame.
Veterans were excluded if they initiated any other antipsychotic during the first course, had a prior diagnosis of T2DM, had any prior use of antidiabetic medications, or had a hemoglobin A1c (HbA1c) > 6.4 in the year prior to initiation.
The primary outcome was completion of all recommended metabolic monitoring time points in the first year after atypical antipsychotic initiation. The secondary outcome was incidence of T2DM as evidenced by either a HbA1c > 6.4 or diagnosis of T2DM entered into the electronic health record. Baseline monitoring for BP, blood glucose, and lipids were considered complete if a data point was collected between 3 months prior and 1 month after atypical antipsychotic initiation. Baseline monitoring for weight was considered complete if a data point was collected between 3 months prior and 2 weeks after initiation. Follow-up monitoring for BP, blood glucose, and lipids were considered completed if a data point was collected at 3 and 12 months (mean, 1 month). Follow-up monitoring for weight was considered completed if collected at 1, 2, and 3 months (mean, 2 weeks) and at 6, 9, and 12 months (mean, 1 month). Waist circumference data and patient and family history are not collected as capturable data points. Therefore, the authors were unable to include these in the final data extraction.
Results
The final cohort consisted of 1,651 veterans who met the inclusion criteria. Overall, at antipsychotic initiation the cohort had a mean (SD) age of 55 (14.6) years, was largely male (88%), and was considered overweight with a mean (SD) BMI of 29.1 (6.4) (Table 1).
Appropriate BP monitoring was completed most often with 492 patients (30%) meeting ADA/APA recommendations followed by HbA1c and/or blood glucose monitoring with 203 patients (12%) completing all time points. Recommended lipid monitoring was completed by 96 patients (6%). Weight monitoring was completed least often with 47 patients (3%) completing all recommended time points. Regarding completion of all metabolic monitoring time points, 3 (0.2%) patients in the final cohort were found to have completed all recommended monitoring. Ninety-nine patients (6%) were found to have progressed to T2DM as indicated by an HbA1c > 6.4 and/or entry of a T2DM ninth or tenth edition International Statistical Classification of Diseases code into the chart (Table 2).
Discussion
No previous literature exists that reviews adherence to recommended metabolic monitoring guidance up to 1 year after antipsychotic initiation in a previously antipsychotic-naïve cohort within the VHA. Metabolic monitoring was overall incomplete with 0.2% of the cohort completing all recommended monitoring time points. Weight was the parameter that was least completed. Based on these findings, the authors concluded that efforts are needed to improve completion rates of atypical antipsychotic metabolic monitoring. In the final cohort, 6% of patients were noted to have progressed to T2DM in the first year after atypical antipsychotic initiation. The actual number of patients progressing to T2DM may be larger because not all received adequate blood glucose monitoring. For comparison, the Centers for Disease Control and Prevention released information in 2015 that stated that the US population has an annual T2DM incidence of about 1% for adults aged 45 to 64 years.7
We understand that individuals with mental health disorders are at increased risk of T2DM compared with that of the general population and hope that this comparison only serves to drive home the point that appropriate metabolic monitoring is vital for this subgroup. The strengths of this project include identification of an area for improvement and encouraging evidence-based monitoring. Utilization of clinical data is a cost-effective and efficient method to improve patient care.
Limitations
Limitations of this study include the data’s dependence on accuracy of entry by the end-user and a lack of available data regarding prescriptions dispensed outside of the VHA. Vital signs data may have been entered into patient notes and not documented in the vitals section of the current medical record causing the appearance of missing data. Access to VHA health services and patient adherence to follow-up appointments were not assessed in this project and could affect patient ability to complete follow-up. The final analysis included only patients who remained on 1 atypical antipsychotic for a year and were considered adherent with an MPR > 80% and did not consider less adherent patients. It is also possible that health care providers who closely monitor metabolic parameters after atypical antipsychotic initiation more frequently switch patients to an alternative atypical antipsychotic while others who monitor less also switch medications less frequently. This could lead to selection of patients with health care providers who are less adherent to metabolic monitoring recommendations.
Conclusions
As a result of this study, in VISN 21 several strategies will be implemented to improve monitoring. First, the results of this project will be shared with the subject matter experts of the VISN 21 Mental Health Task Force. This task force serves as a venue for clinicians to meet virtually, discuss clinical topics, as well as to create and distribute strategies to improve patient care. Clinicians at this forum will be encouraged to implement monitoring protocols into routine practice, share best practices with colleagues, and increase patient awareness about the importance of metabolic monitoring. Second, modifications may be applied to the electronic health record to guide metabolic monitoring order entry at the time of prescription entry, which includes development of clinical reminders and laboratory order sets. Third, the clinical data manager team may be leveraged to create an electronic report identifying patients currently receiving suboptimal monitoring in the first year after antipsychotic initiation. The patients identified in this report will be discussed at the recurring VISN 21 Mental Health Task Force meeting, and strong practices will be shared with the medical centers across VISN 21. Other strategies under consideration include requiring proof of metabolic monitoring completion prior to allowing further atypical antipsychotic refills and providing direct provider education regarding the ADA/APA metabolic monitoring recommendations via the academic detailing service in effort to standardize clinical care.
1. Goff DC, Sullivan LM, McEvoy JP, et al. A comparison of ten-year cardiac risk estimates in schizophrenia patients from the CATIE study and matched controls. Schizophr Res. 2005;80(1):45-53. doi:10.1016/j.schres.2005.08.010
2. American Diabetes Association. Economic costs of diabetes in the U.S. in 2012. Diabetes Care. 2013;36(4):1033-1046. doi:10.2337/dc12-2625
3. Rajkumar AP, Horsdal HT, Wimberley T, et al. Endogenous and antipsychotic-related risks for diabetes mellitus in young people with schizophrenia: a Danish population-based cohort study. Am J Psychiatry. 2017;174(7):686-694. doi:10.1176/appi.ajp.2016.16040442
4. American Diabetes Association; American Psychiatric Association; American Association of Clinical Endocrinologists; North American Association for the Study of Obesity. Consensus development conference on antipsychotic drugs and obesity and diabetes. Diabetes Care. 2004;27(2):596-601. doi:10.2337/diacare.27.2.596
5. Morrato EH, Druss B, Hartung DM, et al. Metabolic testing rates in 3 state Medicaid programs after FDA warnings and ADA/APA recommendations for second-generation antipsychotic drugs. Arch Gen Psychiatry. 2010;67(1):17-24. doi:10.1001/archgenpsychiatry.2009.179
6. Mittal D, Li C, Williams JS, Viverito K, Landes RD, Owen RR. Monitoring veterans for metabolic side effects when prescribing antipsychotics. Psychiatr Serv. 2013;64(1):28-35. doi:10.1176/appi.ps.201100445
7. Centers for Disease Control and Prevention. National diabetes statistics report, 2020. https://www.cdc.gov/diabetes/data/statistics-report/index.html. Updated August 28, 2020. Accessed October 9, 2020.
Atypical antipsychotic use may result in metabolic abnormalities, such as hyperglycemia, dyslipidemia, weight gain, and metabolic syndrome. These adverse effects (AEs) can cause progression to type 2 diabetes mellitus (T2DM) as well as increased risk of cardiovascular disease and cardiac mortality. Individuals diagnosed with T2DM have medical expenses that are about 2.3 times higher than individuals without diabetes.1,2 The risk of experiencing metabolic abnormalities is likely elevated for patients who were antipsychotic-naïve prior to initiation.3
In response to an increased awareness of atypical antipsychotic-related AEs, the American Diabetes Association (ADA) and American Psychiatric Association (APA) released a consensus statement in 2004 with a metabolic monitoring protocol for patients initiating or changing to a new antipsychotic medication.4 Within the first year after initiation, the ADA/APA consensus statements recommends that clinicians acquire a personal and family history, weight, body mass index (BMI), waist circumference, blood pressure (BP), fasting plasma glucose, and fasting lipid profile at the initial patient visit. Patient weight is recommended to be collected at 4 weeks and again 8 weeks later. Twelve weeks after the initial visit, weight, BMI, BP, fasting plasma glucose and a fasting lipid profile are recommended to be collected and assessed for abnormalities. Weight is then recommended to be assessed every 3 months thereafter. Review of personal and family history, waist circumference, BP, and a fasting plasma glucose is recommended to occur annually. Finally, a fasting lipid profile is to be collected every 5 years.
Since the initial consensus statement release, metabolic monitoring of patients prescribed antipsychotic medications has been found to be inadequate within several large health care organizations.5,6 Mittal and colleagues reviewed metabolic monitoring practices occurring in 32 facilities within the Veterans Health Administration (VHA) and found that monitoring practices in the first 90 days after antipsychotic initiation were largely nonadherent to the ADA/APA consensus statement recommendations.6 Medical staff in Veterans Integrated Service Network 21 (VISN 21) currently serve about 268,000 veterans actively receiving care across California, Nevada, and the Pacific Islands.To support veteran care in the fields of mental health and medication safety, the VISN 21 pharmacy benefits manager office created a clinical dashboard that identifies veterans who are currently prescribed an antipsychotic and have not completed at least 1 annual blood glucose test. While this dashboard is a valuable tool for tracking patient care for those who have been prescribed an antipsychotic > 1 year, it does not consider the ADA/APA recommendations for more frequent monitoring in the first year after initiation. A literature review found no citations of a systematic evaluation of adherence to ADA/APA monitoring recommendations or patient progression to T2DM in the first year after antipsychotic initiation for an antipsychotic-naïve veteran population. The goal of this quality improvement project is to assess VHA health care provider and patient adherence to the 2004 consensus statement recommendations within the first year after initiation for previously antipsychotic-naïve patients receiving an atypical antipsychotic and determine rate of progression to T2DM.
Methods
The project was reviewed by the University of Nevada-Reno Institutional Review Board and determined to be a nonresearch quality improvement project. This was a retrospective chart analysis that included patients receiving their first-ever atypical antipsychotic across 8 US Department of Veterans Affairs (VA) medical centers within VISN 21. Clinical patient data, including prescription, vital sign, and laboratory information, were extracted from the VA Corporate Data Warehouse using transact sequential query language.
Veterans were included in the final cohort if they met the following criteria: aged ≥ 18 years at antipsychotic initiation, initiated their first-ever atypical antipsychotic within the VHA between February 2014 and February 2019, continued the antipsychotic for ≥ 1 year, had a medication possession ratio (MPR) > 80%, and had previously established care within VHA as evidenced by having ≥ 1 primary care or outpatient mental health visit in the 6 months prior to initiation. The MPR is defined as the sum of the day’s supply of all dispensed medications in the project time frame divided by the total number of days in the project time frame.
Veterans were excluded if they initiated any other antipsychotic during the first course, had a prior diagnosis of T2DM, had any prior use of antidiabetic medications, or had a hemoglobin A1c (HbA1c) > 6.4 in the year prior to initiation.
The primary outcome was completion of all recommended metabolic monitoring time points in the first year after atypical antipsychotic initiation. The secondary outcome was incidence of T2DM as evidenced by either a HbA1c > 6.4 or diagnosis of T2DM entered into the electronic health record. Baseline monitoring for BP, blood glucose, and lipids were considered complete if a data point was collected between 3 months prior and 1 month after atypical antipsychotic initiation. Baseline monitoring for weight was considered complete if a data point was collected between 3 months prior and 2 weeks after initiation. Follow-up monitoring for BP, blood glucose, and lipids were considered completed if a data point was collected at 3 and 12 months (mean, 1 month). Follow-up monitoring for weight was considered completed if collected at 1, 2, and 3 months (mean, 2 weeks) and at 6, 9, and 12 months (mean, 1 month). Waist circumference data and patient and family history are not collected as capturable data points. Therefore, the authors were unable to include these in the final data extraction.
Results
The final cohort consisted of 1,651 veterans who met the inclusion criteria. Overall, at antipsychotic initiation the cohort had a mean (SD) age of 55 (14.6) years, was largely male (88%), and was considered overweight with a mean (SD) BMI of 29.1 (6.4) (Table 1).
Appropriate BP monitoring was completed most often with 492 patients (30%) meeting ADA/APA recommendations followed by HbA1c and/or blood glucose monitoring with 203 patients (12%) completing all time points. Recommended lipid monitoring was completed by 96 patients (6%). Weight monitoring was completed least often with 47 patients (3%) completing all recommended time points. Regarding completion of all metabolic monitoring time points, 3 (0.2%) patients in the final cohort were found to have completed all recommended monitoring. Ninety-nine patients (6%) were found to have progressed to T2DM as indicated by an HbA1c > 6.4 and/or entry of a T2DM ninth or tenth edition International Statistical Classification of Diseases code into the chart (Table 2).
Discussion
No previous literature exists that reviews adherence to recommended metabolic monitoring guidance up to 1 year after antipsychotic initiation in a previously antipsychotic-naïve cohort within the VHA. Metabolic monitoring was overall incomplete with 0.2% of the cohort completing all recommended monitoring time points. Weight was the parameter that was least completed. Based on these findings, the authors concluded that efforts are needed to improve completion rates of atypical antipsychotic metabolic monitoring. In the final cohort, 6% of patients were noted to have progressed to T2DM in the first year after atypical antipsychotic initiation. The actual number of patients progressing to T2DM may be larger because not all received adequate blood glucose monitoring. For comparison, the Centers for Disease Control and Prevention released information in 2015 that stated that the US population has an annual T2DM incidence of about 1% for adults aged 45 to 64 years.7
We understand that individuals with mental health disorders are at increased risk of T2DM compared with that of the general population and hope that this comparison only serves to drive home the point that appropriate metabolic monitoring is vital for this subgroup. The strengths of this project include identification of an area for improvement and encouraging evidence-based monitoring. Utilization of clinical data is a cost-effective and efficient method to improve patient care.
Limitations
Limitations of this study include the data’s dependence on accuracy of entry by the end-user and a lack of available data regarding prescriptions dispensed outside of the VHA. Vital signs data may have been entered into patient notes and not documented in the vitals section of the current medical record causing the appearance of missing data. Access to VHA health services and patient adherence to follow-up appointments were not assessed in this project and could affect patient ability to complete follow-up. The final analysis included only patients who remained on 1 atypical antipsychotic for a year and were considered adherent with an MPR > 80% and did not consider less adherent patients. It is also possible that health care providers who closely monitor metabolic parameters after atypical antipsychotic initiation more frequently switch patients to an alternative atypical antipsychotic while others who monitor less also switch medications less frequently. This could lead to selection of patients with health care providers who are less adherent to metabolic monitoring recommendations.
Conclusions
As a result of this study, in VISN 21 several strategies will be implemented to improve monitoring. First, the results of this project will be shared with the subject matter experts of the VISN 21 Mental Health Task Force. This task force serves as a venue for clinicians to meet virtually, discuss clinical topics, as well as to create and distribute strategies to improve patient care. Clinicians at this forum will be encouraged to implement monitoring protocols into routine practice, share best practices with colleagues, and increase patient awareness about the importance of metabolic monitoring. Second, modifications may be applied to the electronic health record to guide metabolic monitoring order entry at the time of prescription entry, which includes development of clinical reminders and laboratory order sets. Third, the clinical data manager team may be leveraged to create an electronic report identifying patients currently receiving suboptimal monitoring in the first year after antipsychotic initiation. The patients identified in this report will be discussed at the recurring VISN 21 Mental Health Task Force meeting, and strong practices will be shared with the medical centers across VISN 21. Other strategies under consideration include requiring proof of metabolic monitoring completion prior to allowing further atypical antipsychotic refills and providing direct provider education regarding the ADA/APA metabolic monitoring recommendations via the academic detailing service in effort to standardize clinical care.
Atypical antipsychotic use may result in metabolic abnormalities, such as hyperglycemia, dyslipidemia, weight gain, and metabolic syndrome. These adverse effects (AEs) can cause progression to type 2 diabetes mellitus (T2DM) as well as increased risk of cardiovascular disease and cardiac mortality. Individuals diagnosed with T2DM have medical expenses that are about 2.3 times higher than individuals without diabetes.1,2 The risk of experiencing metabolic abnormalities is likely elevated for patients who were antipsychotic-naïve prior to initiation.3
In response to an increased awareness of atypical antipsychotic-related AEs, the American Diabetes Association (ADA) and American Psychiatric Association (APA) released a consensus statement in 2004 with a metabolic monitoring protocol for patients initiating or changing to a new antipsychotic medication.4 Within the first year after initiation, the ADA/APA consensus statements recommends that clinicians acquire a personal and family history, weight, body mass index (BMI), waist circumference, blood pressure (BP), fasting plasma glucose, and fasting lipid profile at the initial patient visit. Patient weight is recommended to be collected at 4 weeks and again 8 weeks later. Twelve weeks after the initial visit, weight, BMI, BP, fasting plasma glucose and a fasting lipid profile are recommended to be collected and assessed for abnormalities. Weight is then recommended to be assessed every 3 months thereafter. Review of personal and family history, waist circumference, BP, and a fasting plasma glucose is recommended to occur annually. Finally, a fasting lipid profile is to be collected every 5 years.
Since the initial consensus statement release, metabolic monitoring of patients prescribed antipsychotic medications has been found to be inadequate within several large health care organizations.5,6 Mittal and colleagues reviewed metabolic monitoring practices occurring in 32 facilities within the Veterans Health Administration (VHA) and found that monitoring practices in the first 90 days after antipsychotic initiation were largely nonadherent to the ADA/APA consensus statement recommendations.6 Medical staff in Veterans Integrated Service Network 21 (VISN 21) currently serve about 268,000 veterans actively receiving care across California, Nevada, and the Pacific Islands.To support veteran care in the fields of mental health and medication safety, the VISN 21 pharmacy benefits manager office created a clinical dashboard that identifies veterans who are currently prescribed an antipsychotic and have not completed at least 1 annual blood glucose test. While this dashboard is a valuable tool for tracking patient care for those who have been prescribed an antipsychotic > 1 year, it does not consider the ADA/APA recommendations for more frequent monitoring in the first year after initiation. A literature review found no citations of a systematic evaluation of adherence to ADA/APA monitoring recommendations or patient progression to T2DM in the first year after antipsychotic initiation for an antipsychotic-naïve veteran population. The goal of this quality improvement project is to assess VHA health care provider and patient adherence to the 2004 consensus statement recommendations within the first year after initiation for previously antipsychotic-naïve patients receiving an atypical antipsychotic and determine rate of progression to T2DM.
Methods
The project was reviewed by the University of Nevada-Reno Institutional Review Board and determined to be a nonresearch quality improvement project. This was a retrospective chart analysis that included patients receiving their first-ever atypical antipsychotic across 8 US Department of Veterans Affairs (VA) medical centers within VISN 21. Clinical patient data, including prescription, vital sign, and laboratory information, were extracted from the VA Corporate Data Warehouse using transact sequential query language.
Veterans were included in the final cohort if they met the following criteria: aged ≥ 18 years at antipsychotic initiation, initiated their first-ever atypical antipsychotic within the VHA between February 2014 and February 2019, continued the antipsychotic for ≥ 1 year, had a medication possession ratio (MPR) > 80%, and had previously established care within VHA as evidenced by having ≥ 1 primary care or outpatient mental health visit in the 6 months prior to initiation. The MPR is defined as the sum of the day’s supply of all dispensed medications in the project time frame divided by the total number of days in the project time frame.
Veterans were excluded if they initiated any other antipsychotic during the first course, had a prior diagnosis of T2DM, had any prior use of antidiabetic medications, or had a hemoglobin A1c (HbA1c) > 6.4 in the year prior to initiation.
The primary outcome was completion of all recommended metabolic monitoring time points in the first year after atypical antipsychotic initiation. The secondary outcome was incidence of T2DM as evidenced by either a HbA1c > 6.4 or diagnosis of T2DM entered into the electronic health record. Baseline monitoring for BP, blood glucose, and lipids were considered complete if a data point was collected between 3 months prior and 1 month after atypical antipsychotic initiation. Baseline monitoring for weight was considered complete if a data point was collected between 3 months prior and 2 weeks after initiation. Follow-up monitoring for BP, blood glucose, and lipids were considered completed if a data point was collected at 3 and 12 months (mean, 1 month). Follow-up monitoring for weight was considered completed if collected at 1, 2, and 3 months (mean, 2 weeks) and at 6, 9, and 12 months (mean, 1 month). Waist circumference data and patient and family history are not collected as capturable data points. Therefore, the authors were unable to include these in the final data extraction.
Results
The final cohort consisted of 1,651 veterans who met the inclusion criteria. Overall, at antipsychotic initiation the cohort had a mean (SD) age of 55 (14.6) years, was largely male (88%), and was considered overweight with a mean (SD) BMI of 29.1 (6.4) (Table 1).
Appropriate BP monitoring was completed most often with 492 patients (30%) meeting ADA/APA recommendations followed by HbA1c and/or blood glucose monitoring with 203 patients (12%) completing all time points. Recommended lipid monitoring was completed by 96 patients (6%). Weight monitoring was completed least often with 47 patients (3%) completing all recommended time points. Regarding completion of all metabolic monitoring time points, 3 (0.2%) patients in the final cohort were found to have completed all recommended monitoring. Ninety-nine patients (6%) were found to have progressed to T2DM as indicated by an HbA1c > 6.4 and/or entry of a T2DM ninth or tenth edition International Statistical Classification of Diseases code into the chart (Table 2).
Discussion
No previous literature exists that reviews adherence to recommended metabolic monitoring guidance up to 1 year after antipsychotic initiation in a previously antipsychotic-naïve cohort within the VHA. Metabolic monitoring was overall incomplete with 0.2% of the cohort completing all recommended monitoring time points. Weight was the parameter that was least completed. Based on these findings, the authors concluded that efforts are needed to improve completion rates of atypical antipsychotic metabolic monitoring. In the final cohort, 6% of patients were noted to have progressed to T2DM in the first year after atypical antipsychotic initiation. The actual number of patients progressing to T2DM may be larger because not all received adequate blood glucose monitoring. For comparison, the Centers for Disease Control and Prevention released information in 2015 that stated that the US population has an annual T2DM incidence of about 1% for adults aged 45 to 64 years.7
We understand that individuals with mental health disorders are at increased risk of T2DM compared with that of the general population and hope that this comparison only serves to drive home the point that appropriate metabolic monitoring is vital for this subgroup. The strengths of this project include identification of an area for improvement and encouraging evidence-based monitoring. Utilization of clinical data is a cost-effective and efficient method to improve patient care.
Limitations
Limitations of this study include the data’s dependence on accuracy of entry by the end-user and a lack of available data regarding prescriptions dispensed outside of the VHA. Vital signs data may have been entered into patient notes and not documented in the vitals section of the current medical record causing the appearance of missing data. Access to VHA health services and patient adherence to follow-up appointments were not assessed in this project and could affect patient ability to complete follow-up. The final analysis included only patients who remained on 1 atypical antipsychotic for a year and were considered adherent with an MPR > 80% and did not consider less adherent patients. It is also possible that health care providers who closely monitor metabolic parameters after atypical antipsychotic initiation more frequently switch patients to an alternative atypical antipsychotic while others who monitor less also switch medications less frequently. This could lead to selection of patients with health care providers who are less adherent to metabolic monitoring recommendations.
Conclusions
As a result of this study, in VISN 21 several strategies will be implemented to improve monitoring. First, the results of this project will be shared with the subject matter experts of the VISN 21 Mental Health Task Force. This task force serves as a venue for clinicians to meet virtually, discuss clinical topics, as well as to create and distribute strategies to improve patient care. Clinicians at this forum will be encouraged to implement monitoring protocols into routine practice, share best practices with colleagues, and increase patient awareness about the importance of metabolic monitoring. Second, modifications may be applied to the electronic health record to guide metabolic monitoring order entry at the time of prescription entry, which includes development of clinical reminders and laboratory order sets. Third, the clinical data manager team may be leveraged to create an electronic report identifying patients currently receiving suboptimal monitoring in the first year after antipsychotic initiation. The patients identified in this report will be discussed at the recurring VISN 21 Mental Health Task Force meeting, and strong practices will be shared with the medical centers across VISN 21. Other strategies under consideration include requiring proof of metabolic monitoring completion prior to allowing further atypical antipsychotic refills and providing direct provider education regarding the ADA/APA metabolic monitoring recommendations via the academic detailing service in effort to standardize clinical care.
1. Goff DC, Sullivan LM, McEvoy JP, et al. A comparison of ten-year cardiac risk estimates in schizophrenia patients from the CATIE study and matched controls. Schizophr Res. 2005;80(1):45-53. doi:10.1016/j.schres.2005.08.010
2. American Diabetes Association. Economic costs of diabetes in the U.S. in 2012. Diabetes Care. 2013;36(4):1033-1046. doi:10.2337/dc12-2625
3. Rajkumar AP, Horsdal HT, Wimberley T, et al. Endogenous and antipsychotic-related risks for diabetes mellitus in young people with schizophrenia: a Danish population-based cohort study. Am J Psychiatry. 2017;174(7):686-694. doi:10.1176/appi.ajp.2016.16040442
4. American Diabetes Association; American Psychiatric Association; American Association of Clinical Endocrinologists; North American Association for the Study of Obesity. Consensus development conference on antipsychotic drugs and obesity and diabetes. Diabetes Care. 2004;27(2):596-601. doi:10.2337/diacare.27.2.596
5. Morrato EH, Druss B, Hartung DM, et al. Metabolic testing rates in 3 state Medicaid programs after FDA warnings and ADA/APA recommendations for second-generation antipsychotic drugs. Arch Gen Psychiatry. 2010;67(1):17-24. doi:10.1001/archgenpsychiatry.2009.179
6. Mittal D, Li C, Williams JS, Viverito K, Landes RD, Owen RR. Monitoring veterans for metabolic side effects when prescribing antipsychotics. Psychiatr Serv. 2013;64(1):28-35. doi:10.1176/appi.ps.201100445
7. Centers for Disease Control and Prevention. National diabetes statistics report, 2020. https://www.cdc.gov/diabetes/data/statistics-report/index.html. Updated August 28, 2020. Accessed October 9, 2020.
1. Goff DC, Sullivan LM, McEvoy JP, et al. A comparison of ten-year cardiac risk estimates in schizophrenia patients from the CATIE study and matched controls. Schizophr Res. 2005;80(1):45-53. doi:10.1016/j.schres.2005.08.010
2. American Diabetes Association. Economic costs of diabetes in the U.S. in 2012. Diabetes Care. 2013;36(4):1033-1046. doi:10.2337/dc12-2625
3. Rajkumar AP, Horsdal HT, Wimberley T, et al. Endogenous and antipsychotic-related risks for diabetes mellitus in young people with schizophrenia: a Danish population-based cohort study. Am J Psychiatry. 2017;174(7):686-694. doi:10.1176/appi.ajp.2016.16040442
4. American Diabetes Association; American Psychiatric Association; American Association of Clinical Endocrinologists; North American Association for the Study of Obesity. Consensus development conference on antipsychotic drugs and obesity and diabetes. Diabetes Care. 2004;27(2):596-601. doi:10.2337/diacare.27.2.596
5. Morrato EH, Druss B, Hartung DM, et al. Metabolic testing rates in 3 state Medicaid programs after FDA warnings and ADA/APA recommendations for second-generation antipsychotic drugs. Arch Gen Psychiatry. 2010;67(1):17-24. doi:10.1001/archgenpsychiatry.2009.179
6. Mittal D, Li C, Williams JS, Viverito K, Landes RD, Owen RR. Monitoring veterans for metabolic side effects when prescribing antipsychotics. Psychiatr Serv. 2013;64(1):28-35. doi:10.1176/appi.ps.201100445
7. Centers for Disease Control and Prevention. National diabetes statistics report, 2020. https://www.cdc.gov/diabetes/data/statistics-report/index.html. Updated August 28, 2020. Accessed October 9, 2020.
Diabetes screening program in optometry offices to expand
The program is sponsored by VSP Vision Care, a vision benefits company with over 40,000 network optometrists and nearly 90 million consumer members worldwide. “Optometrists are often the first to detect signs of diabetes by looking at the blood vessels in the eye during a comprehensive eye exam,” the company said in a statement.
In the pilot program, conducted from May 2019 to February 2020* in 12 VSP practices in five states, 818 patients who had come in for their annual vision exam were given the American Diabetes Association Risk Test for type 2 diabetes, and 287 identified at risk were offered an in-office fingerstick hemoglobin A1c test.
Materials were provided free to the optometrists, who were paid a professional fee to perform the HbA1c screenings.
Of the 287 eligible for the HbA1c test, 85% took it. Of those 244, 31% and 5% had levels in the prediabetes and diabetes range, respectively. None had been aware of their status previously, and 92% rated the screening as an extremely or very positive experience.
Now, VSP is expanding the pilot program for another year with two large clients in Ohio covering about 90,000 members.
“Coupled with the fact that VSP members are more likely to get their annual eye exam over their annual physical exam with their primary care physician, HbA1c screenings provided by eye doctors offer another critical way to detect the chronic condition earlier and help prevent eye disease and even vision loss caused by diabetes,” according to the statement.
In an interview, a VSP spokesperson explained that if the patient provides their primary care provider information to the optometrist, the optometrist will send a referral with exam information to that provider and also instruct the patient to make an appointment with the provider for follow-up testing and care.
The optometrist also educates the patient about the connection between eye health and overall health and provides them with a flier that gives tips on lifestyle changes they can make to help slow or prevent the progression to type 2 diabetes, the spokesperson said.
Thirty states, including Ohio, allow optometrists to perform in-office blood testing, including HbA1c screening, provided they obtain a Clinical Laboratory Improvement Amendments Certificate of Waiver. VSP is providing online training for participating optometrists on administering the HbA1c screening.
The pilot program is part of an alliance between VSP and the American Diabetes Association formed in November 2019 to raise awareness of eye health in people with diabetes and those at risk for it.
*Correction: The original article included the wrong end date for the pilot program.
The program is sponsored by VSP Vision Care, a vision benefits company with over 40,000 network optometrists and nearly 90 million consumer members worldwide. “Optometrists are often the first to detect signs of diabetes by looking at the blood vessels in the eye during a comprehensive eye exam,” the company said in a statement.
In the pilot program, conducted from May 2019 to February 2020* in 12 VSP practices in five states, 818 patients who had come in for their annual vision exam were given the American Diabetes Association Risk Test for type 2 diabetes, and 287 identified at risk were offered an in-office fingerstick hemoglobin A1c test.
Materials were provided free to the optometrists, who were paid a professional fee to perform the HbA1c screenings.
Of the 287 eligible for the HbA1c test, 85% took it. Of those 244, 31% and 5% had levels in the prediabetes and diabetes range, respectively. None had been aware of their status previously, and 92% rated the screening as an extremely or very positive experience.
Now, VSP is expanding the pilot program for another year with two large clients in Ohio covering about 90,000 members.
“Coupled with the fact that VSP members are more likely to get their annual eye exam over their annual physical exam with their primary care physician, HbA1c screenings provided by eye doctors offer another critical way to detect the chronic condition earlier and help prevent eye disease and even vision loss caused by diabetes,” according to the statement.
In an interview, a VSP spokesperson explained that if the patient provides their primary care provider information to the optometrist, the optometrist will send a referral with exam information to that provider and also instruct the patient to make an appointment with the provider for follow-up testing and care.
The optometrist also educates the patient about the connection between eye health and overall health and provides them with a flier that gives tips on lifestyle changes they can make to help slow or prevent the progression to type 2 diabetes, the spokesperson said.
Thirty states, including Ohio, allow optometrists to perform in-office blood testing, including HbA1c screening, provided they obtain a Clinical Laboratory Improvement Amendments Certificate of Waiver. VSP is providing online training for participating optometrists on administering the HbA1c screening.
The pilot program is part of an alliance between VSP and the American Diabetes Association formed in November 2019 to raise awareness of eye health in people with diabetes and those at risk for it.
*Correction: The original article included the wrong end date for the pilot program.
The program is sponsored by VSP Vision Care, a vision benefits company with over 40,000 network optometrists and nearly 90 million consumer members worldwide. “Optometrists are often the first to detect signs of diabetes by looking at the blood vessels in the eye during a comprehensive eye exam,” the company said in a statement.
In the pilot program, conducted from May 2019 to February 2020* in 12 VSP practices in five states, 818 patients who had come in for their annual vision exam were given the American Diabetes Association Risk Test for type 2 diabetes, and 287 identified at risk were offered an in-office fingerstick hemoglobin A1c test.
Materials were provided free to the optometrists, who were paid a professional fee to perform the HbA1c screenings.
Of the 287 eligible for the HbA1c test, 85% took it. Of those 244, 31% and 5% had levels in the prediabetes and diabetes range, respectively. None had been aware of their status previously, and 92% rated the screening as an extremely or very positive experience.
Now, VSP is expanding the pilot program for another year with two large clients in Ohio covering about 90,000 members.
“Coupled with the fact that VSP members are more likely to get their annual eye exam over their annual physical exam with their primary care physician, HbA1c screenings provided by eye doctors offer another critical way to detect the chronic condition earlier and help prevent eye disease and even vision loss caused by diabetes,” according to the statement.
In an interview, a VSP spokesperson explained that if the patient provides their primary care provider information to the optometrist, the optometrist will send a referral with exam information to that provider and also instruct the patient to make an appointment with the provider for follow-up testing and care.
The optometrist also educates the patient about the connection between eye health and overall health and provides them with a flier that gives tips on lifestyle changes they can make to help slow or prevent the progression to type 2 diabetes, the spokesperson said.
Thirty states, including Ohio, allow optometrists to perform in-office blood testing, including HbA1c screening, provided they obtain a Clinical Laboratory Improvement Amendments Certificate of Waiver. VSP is providing online training for participating optometrists on administering the HbA1c screening.
The pilot program is part of an alliance between VSP and the American Diabetes Association formed in November 2019 to raise awareness of eye health in people with diabetes and those at risk for it.
*Correction: The original article included the wrong end date for the pilot program.
HIV drugs prevent type 2 diabetes, may be path to new therapy
A class of drugs long used to treat HIV and hepatitis B viral infections appears to prevent the development of diabetes in a substantial proportion of patients who take these agents, an analysis of multiple databases has shown.
“Nucleoside reverse transcriptase inhibitors [NRTIs], drugs approved to treat HIV-1 and hepatitis B infections, also block inflammasome activation,” Jayakrishna Ambati, MD, University of Virginia, Charlottesville, and colleagues wrote in Nature Communications.
“[We showed that] the adjusted risk of incident diabetes is 33% lower in patients with NRTI exposure. ... These data suggest the possibility of repurposing an approved class of drugs for prevention of diabetes,” they wrote.
The researchers made a small chemical modification to NRTIs that led to their developing a new class of drugs, which they have termed “kamuvudines.” Kamuvudines are nontoxic derivatives of NRTIs, Dr. Ambati said in an interview.
“People take NRTIs because they need to live with HIV, but giving them to the general population is not a great idea because of the toxicities associated with long-term NRTI use. So our focus is not to go forward specifically with NRTIs but rather with these new molecules that are far less toxic, and that is how we envision a clinical trial going forward,” Dr. Ambati noted.
Researchers screened five databases of >100,000 patients
Dr. Ambati and colleagues analyzed information from five databases in which patients who had been exposed to an NRTI but who had not previously been diagnosed with type 2 diabetes were assessed for the subsequent development of diabetes over varying time intervals. In one, the Veterans Health Administration database – from the largest integrated health care system in the United States – the analysis spanned a period of 17 years.
Of 79,744 patients with a confirmed diagnosis of HIV or hepatitis B in the Veterans Health Administration database, the risk for type 2 diabetes was reduced by 34% among NRTI users, compared with nonusers after adjusting for potential confounders (P < .0001).
The reduction in diabetes risk was similar among HIV-positive and hepatitis B–positive patients.
These results were reaffirmed by further analyses of four other databases, the investigators reported. One of these, the employer-based health insurance Truven database, had data on 23,634 patients who had been diagnosed with HIV or hepatitis B. After adjusting for potential confounders, NRTI users had a 39% lower risk of developing type 2 diabetes, compared with nonusers (P < .0001).
The risk of developing type 2 diabetes was somewhat lower among NRTI users in the Pearl Diver database, which includes predominantly private health insurance claims. Of 16,045 patients diagnosed with HIV or hepatitis B included in this database, the risk for type 2 diabetes was 26% lower among NRTI users, compared with nonusers (P = .004).
A similar magnitude of risk reduction was seen in the analysis of the Clinformatics dataset. Among 6,341 users of NRTIs, the risk for type 2 diabetes was 27% lower than it was for nonusers (P = .009).
The least reduction in diabetes risk was in the Medicare database, in which only 3,097 patients had been diagnosed with either HIV or hepatitis B. Among these patients, the risk for diabetes was 17% lower among NRTI users than it was for nonusers (P = .137).
One-third reduction across multiple databases enhances confidence
“Collectively, among 128,861 patients with HIV-1 or hepatitis B, users of NRTIs had a 33% reduced hazard of developing type 2 diabetes,” Dr. Ambati and colleagues emphasize.
“The fact that the protective effect against the development of diabetes was replicated in multiple databases in studies from multiple institutions enhances confidence in the results,” Dr. Ambati noted in a statement from the University of Virginia.
Dr. Ambati and colleagues also showed that the NRTI lamivudine restores insulin sensitivity in human cells from type 2 diabetes patients.
That drug prevented induction of insulin resistance in human cells from people who did not have diabetes. It also prevented inflammasome activation in mice fed a high-fat diet.
“These investigations of human cell, mouse and population database systems collectively suggest a potential beneficial effect of NRTIs in forestalling diabetes onset,” they stressed.
Trial assessing kamuvudines slated to begin next year
In the interview, Dr. Ambati explained that inflammasomes are protein complexes that form a large superstructure within the cell. “When activated, they lead to the production of some very powerful inflammatory cytokines, including interleukin-1 beta and IL-18.”
Although there are many different types of inflammasomes, the one implicated in type 2 diabetes, as well as many other chronic diseases, including macular degeneration, is the NLRP3 inflammasome.
Activation of this molecule promotes insulin resistance, a key driver of type 2 diabetes, he explained.
Importantly, previous research showed that the way the NRTIs block this inflammasome has nothing to do with their anti-HIV activity.
After making a small chemical modification in the NRTIs, Dr. Ambati and colleagues were able to show that the resulting agents, which they have dubbed “kamuvudines,” are able to block inflammasome activation independently of their antiviral effects.
They hope that this modification will reduce the toxicities associated with the agents. This would be necessary if kamuvudines were to be more widely used in a noninfected, healthier population, Ambati stressed.
Dr. Ambati and his colleague, Paul Ashton, PhD, cofounder of Inflammasone Therapeutics, plan a clinical trial with one of these kamuvudines in macular degeneration, which they hope will begin early next year.
“We are trying to pick a disease where we can show efficacy fairly quickly in a small number of people,” Dr. Ashton explained in an interview. “We’re very enthusiastic about this as it looks really, really promising.”
Dr. Ambati and Dr. Ashton cofounded Inflammasone Therapeutics, located in Boston. Dr. Ashton is the CEO of the company.
A version of this article originally appeared on Medscape.com.
A class of drugs long used to treat HIV and hepatitis B viral infections appears to prevent the development of diabetes in a substantial proportion of patients who take these agents, an analysis of multiple databases has shown.
“Nucleoside reverse transcriptase inhibitors [NRTIs], drugs approved to treat HIV-1 and hepatitis B infections, also block inflammasome activation,” Jayakrishna Ambati, MD, University of Virginia, Charlottesville, and colleagues wrote in Nature Communications.
“[We showed that] the adjusted risk of incident diabetes is 33% lower in patients with NRTI exposure. ... These data suggest the possibility of repurposing an approved class of drugs for prevention of diabetes,” they wrote.
The researchers made a small chemical modification to NRTIs that led to their developing a new class of drugs, which they have termed “kamuvudines.” Kamuvudines are nontoxic derivatives of NRTIs, Dr. Ambati said in an interview.
“People take NRTIs because they need to live with HIV, but giving them to the general population is not a great idea because of the toxicities associated with long-term NRTI use. So our focus is not to go forward specifically with NRTIs but rather with these new molecules that are far less toxic, and that is how we envision a clinical trial going forward,” Dr. Ambati noted.
Researchers screened five databases of >100,000 patients
Dr. Ambati and colleagues analyzed information from five databases in which patients who had been exposed to an NRTI but who had not previously been diagnosed with type 2 diabetes were assessed for the subsequent development of diabetes over varying time intervals. In one, the Veterans Health Administration database – from the largest integrated health care system in the United States – the analysis spanned a period of 17 years.
Of 79,744 patients with a confirmed diagnosis of HIV or hepatitis B in the Veterans Health Administration database, the risk for type 2 diabetes was reduced by 34% among NRTI users, compared with nonusers after adjusting for potential confounders (P < .0001).
The reduction in diabetes risk was similar among HIV-positive and hepatitis B–positive patients.
These results were reaffirmed by further analyses of four other databases, the investigators reported. One of these, the employer-based health insurance Truven database, had data on 23,634 patients who had been diagnosed with HIV or hepatitis B. After adjusting for potential confounders, NRTI users had a 39% lower risk of developing type 2 diabetes, compared with nonusers (P < .0001).
The risk of developing type 2 diabetes was somewhat lower among NRTI users in the Pearl Diver database, which includes predominantly private health insurance claims. Of 16,045 patients diagnosed with HIV or hepatitis B included in this database, the risk for type 2 diabetes was 26% lower among NRTI users, compared with nonusers (P = .004).
A similar magnitude of risk reduction was seen in the analysis of the Clinformatics dataset. Among 6,341 users of NRTIs, the risk for type 2 diabetes was 27% lower than it was for nonusers (P = .009).
The least reduction in diabetes risk was in the Medicare database, in which only 3,097 patients had been diagnosed with either HIV or hepatitis B. Among these patients, the risk for diabetes was 17% lower among NRTI users than it was for nonusers (P = .137).
One-third reduction across multiple databases enhances confidence
“Collectively, among 128,861 patients with HIV-1 or hepatitis B, users of NRTIs had a 33% reduced hazard of developing type 2 diabetes,” Dr. Ambati and colleagues emphasize.
“The fact that the protective effect against the development of diabetes was replicated in multiple databases in studies from multiple institutions enhances confidence in the results,” Dr. Ambati noted in a statement from the University of Virginia.
Dr. Ambati and colleagues also showed that the NRTI lamivudine restores insulin sensitivity in human cells from type 2 diabetes patients.
That drug prevented induction of insulin resistance in human cells from people who did not have diabetes. It also prevented inflammasome activation in mice fed a high-fat diet.
“These investigations of human cell, mouse and population database systems collectively suggest a potential beneficial effect of NRTIs in forestalling diabetes onset,” they stressed.
Trial assessing kamuvudines slated to begin next year
In the interview, Dr. Ambati explained that inflammasomes are protein complexes that form a large superstructure within the cell. “When activated, they lead to the production of some very powerful inflammatory cytokines, including interleukin-1 beta and IL-18.”
Although there are many different types of inflammasomes, the one implicated in type 2 diabetes, as well as many other chronic diseases, including macular degeneration, is the NLRP3 inflammasome.
Activation of this molecule promotes insulin resistance, a key driver of type 2 diabetes, he explained.
Importantly, previous research showed that the way the NRTIs block this inflammasome has nothing to do with their anti-HIV activity.
After making a small chemical modification in the NRTIs, Dr. Ambati and colleagues were able to show that the resulting agents, which they have dubbed “kamuvudines,” are able to block inflammasome activation independently of their antiviral effects.
They hope that this modification will reduce the toxicities associated with the agents. This would be necessary if kamuvudines were to be more widely used in a noninfected, healthier population, Ambati stressed.
Dr. Ambati and his colleague, Paul Ashton, PhD, cofounder of Inflammasone Therapeutics, plan a clinical trial with one of these kamuvudines in macular degeneration, which they hope will begin early next year.
“We are trying to pick a disease where we can show efficacy fairly quickly in a small number of people,” Dr. Ashton explained in an interview. “We’re very enthusiastic about this as it looks really, really promising.”
Dr. Ambati and Dr. Ashton cofounded Inflammasone Therapeutics, located in Boston. Dr. Ashton is the CEO of the company.
A version of this article originally appeared on Medscape.com.
A class of drugs long used to treat HIV and hepatitis B viral infections appears to prevent the development of diabetes in a substantial proportion of patients who take these agents, an analysis of multiple databases has shown.
“Nucleoside reverse transcriptase inhibitors [NRTIs], drugs approved to treat HIV-1 and hepatitis B infections, also block inflammasome activation,” Jayakrishna Ambati, MD, University of Virginia, Charlottesville, and colleagues wrote in Nature Communications.
“[We showed that] the adjusted risk of incident diabetes is 33% lower in patients with NRTI exposure. ... These data suggest the possibility of repurposing an approved class of drugs for prevention of diabetes,” they wrote.
The researchers made a small chemical modification to NRTIs that led to their developing a new class of drugs, which they have termed “kamuvudines.” Kamuvudines are nontoxic derivatives of NRTIs, Dr. Ambati said in an interview.
“People take NRTIs because they need to live with HIV, but giving them to the general population is not a great idea because of the toxicities associated with long-term NRTI use. So our focus is not to go forward specifically with NRTIs but rather with these new molecules that are far less toxic, and that is how we envision a clinical trial going forward,” Dr. Ambati noted.
Researchers screened five databases of >100,000 patients
Dr. Ambati and colleagues analyzed information from five databases in which patients who had been exposed to an NRTI but who had not previously been diagnosed with type 2 diabetes were assessed for the subsequent development of diabetes over varying time intervals. In one, the Veterans Health Administration database – from the largest integrated health care system in the United States – the analysis spanned a period of 17 years.
Of 79,744 patients with a confirmed diagnosis of HIV or hepatitis B in the Veterans Health Administration database, the risk for type 2 diabetes was reduced by 34% among NRTI users, compared with nonusers after adjusting for potential confounders (P < .0001).
The reduction in diabetes risk was similar among HIV-positive and hepatitis B–positive patients.
These results were reaffirmed by further analyses of four other databases, the investigators reported. One of these, the employer-based health insurance Truven database, had data on 23,634 patients who had been diagnosed with HIV or hepatitis B. After adjusting for potential confounders, NRTI users had a 39% lower risk of developing type 2 diabetes, compared with nonusers (P < .0001).
The risk of developing type 2 diabetes was somewhat lower among NRTI users in the Pearl Diver database, which includes predominantly private health insurance claims. Of 16,045 patients diagnosed with HIV or hepatitis B included in this database, the risk for type 2 diabetes was 26% lower among NRTI users, compared with nonusers (P = .004).
A similar magnitude of risk reduction was seen in the analysis of the Clinformatics dataset. Among 6,341 users of NRTIs, the risk for type 2 diabetes was 27% lower than it was for nonusers (P = .009).
The least reduction in diabetes risk was in the Medicare database, in which only 3,097 patients had been diagnosed with either HIV or hepatitis B. Among these patients, the risk for diabetes was 17% lower among NRTI users than it was for nonusers (P = .137).
One-third reduction across multiple databases enhances confidence
“Collectively, among 128,861 patients with HIV-1 or hepatitis B, users of NRTIs had a 33% reduced hazard of developing type 2 diabetes,” Dr. Ambati and colleagues emphasize.
“The fact that the protective effect against the development of diabetes was replicated in multiple databases in studies from multiple institutions enhances confidence in the results,” Dr. Ambati noted in a statement from the University of Virginia.
Dr. Ambati and colleagues also showed that the NRTI lamivudine restores insulin sensitivity in human cells from type 2 diabetes patients.
That drug prevented induction of insulin resistance in human cells from people who did not have diabetes. It also prevented inflammasome activation in mice fed a high-fat diet.
“These investigations of human cell, mouse and population database systems collectively suggest a potential beneficial effect of NRTIs in forestalling diabetes onset,” they stressed.
Trial assessing kamuvudines slated to begin next year
In the interview, Dr. Ambati explained that inflammasomes are protein complexes that form a large superstructure within the cell. “When activated, they lead to the production of some very powerful inflammatory cytokines, including interleukin-1 beta and IL-18.”
Although there are many different types of inflammasomes, the one implicated in type 2 diabetes, as well as many other chronic diseases, including macular degeneration, is the NLRP3 inflammasome.
Activation of this molecule promotes insulin resistance, a key driver of type 2 diabetes, he explained.
Importantly, previous research showed that the way the NRTIs block this inflammasome has nothing to do with their anti-HIV activity.
After making a small chemical modification in the NRTIs, Dr. Ambati and colleagues were able to show that the resulting agents, which they have dubbed “kamuvudines,” are able to block inflammasome activation independently of their antiviral effects.
They hope that this modification will reduce the toxicities associated with the agents. This would be necessary if kamuvudines were to be more widely used in a noninfected, healthier population, Ambati stressed.
Dr. Ambati and his colleague, Paul Ashton, PhD, cofounder of Inflammasone Therapeutics, plan a clinical trial with one of these kamuvudines in macular degeneration, which they hope will begin early next year.
“We are trying to pick a disease where we can show efficacy fairly quickly in a small number of people,” Dr. Ashton explained in an interview. “We’re very enthusiastic about this as it looks really, really promising.”
Dr. Ambati and Dr. Ashton cofounded Inflammasone Therapeutics, located in Boston. Dr. Ashton is the CEO of the company.
A version of this article originally appeared on Medscape.com.