User login
Intermittent fasting: What to tell patients
Intermittent fasting is the purposeful, restricted intake of food (and sometimes water), usually for health or religious reasons. Common forms are alternative-day fasting or time-restricted fasting, with variable ratios of days or hours for fasting and eating/drinking.1 For example, fasting during Ramadan, the ninth month of the Islamic calendar, occurs from dawn to sunset, for a variable duration due to latitude and seasonal shifts.2 Clinicians are likely to care for a patient who occasionally fasts. While there are potential benefits of fasting, clinicians need to consider the implications for patients who fast, particularly those receiving psychotropic medications.
Potential benefits for weight loss, mood
Some research suggests fasting is popular and may have benefits for an individual’s physical and mental health. In a 2020 online poll (N = 1,241), 24% of respondents said they had tried intermittent fasting, and 87% said the practice was very effective (50%) or somewhat effective (37%) in helping them lose weight.3 While more randomized control trials are needed to examine the practice’s effectiveness in promoting and maintaining weight loss, fasting has been linked to better glucose control in both humans and animals, and patients may have better adherence with fasting compared to caloric restriction alone.1 Improved mood, alertness, tranquility, and sometimes euphoria have been documented among individuals who fast, but these benefits may not be sustained.4 A prospective study of 462 participants who fasted during Ramadan found the practice reduced depression in patients with diabetes, possibly due to mindfulness, decreased inflammation from improved insulin sensitivity, and/or social cohesion.5
Be aware of the potential risks
Fasting may either improve or destabilize mood in people with bipolar disorder by disrupting circadian rhythm and sleep.2 Fasting might exacerbate underlying eating disorders.2 Increased dehydration escalates the risk for orthostatic hypotension, which might require discontinuing clozapine.6 Hypotension and toxicity might arise during lithium pharmacotherapy. The Table4 summarizes things to consider when caring for a patient who fasts while receiving pharmacotherapy.

Provide patients with guidance
Advise patients not to fast if you believe it might exacerbate their mental illness, and encourage them to discuss with their primary care physicians any potential worsening of physical illnesses.2 When caring for a patient who fasts for religious reasons, consider consulting with the patient’s religious leaders.2 If patients choose to fast, monitor them for mood destabilization and/or medication adverse effects. If possible, avoid altering drug treatment regimens during fasting, and carefully monitor whenever a pharmaceutical change is necessary. When appropriate, the use of long-acting injectable medications may minimize adverse effects while maintaining mood stability. Encourage patients who fast to ensure they remain hydrated and practice sleep hygiene while they fast.7
1. Dong TA, Sandesara PB, Dhindsa DS, et al. Intermittent fasting: a heart healthy dietary pattern? Am J Med. 2020;133(8):901-907.
2. Fond G, Macgregor A, Leboyer M, et al. Fasting in mood disorders: neurobiology and effectiveness. A review of the literature. Psychiatry Res. 2013;209(3):253-258.
3. Ballard J. Americans say this popular diet is effective and inexpensive. YouGov. February 24, 2020. Accessed January 6, 2022. https://today.yougov.com/topics/food/articles-reports/2020/02/24/most-effective-diet-intermittent-fasting-poll
4. Furqan Z, Awaad R, Kurdyak P, et al. Considerations for clinicians treating Muslim patients with psychiatric disorders during Ramadan. Lancet Psychiatry. 2019;6(7):556-557.
5. Al-Ozairi E, AlAwadhi MM, Al-Ozairi A, et al. A prospective study of the effect of fasting during the month of Ramadan on depression and diabetes distress in people with type 2 diabetes. Diabet Res Clin Pract. 2019;153:145-149.
6. Chehovich C, Demler TL, Leppien E. Impact of Ramadan fasting on medical and psychiatric health. Int Clin Psychopharmacol. 2019;34(6):317-322.
7. Farooq S, Nazar Z, Akhtar J, et al. Effect of fasting during Ramadan on serum lithium level and mental state in bipolar affective disorder. Int Clin Psychopharmacol. 2010;25(6):323-327.
Intermittent fasting is the purposeful, restricted intake of food (and sometimes water), usually for health or religious reasons. Common forms are alternative-day fasting or time-restricted fasting, with variable ratios of days or hours for fasting and eating/drinking.1 For example, fasting during Ramadan, the ninth month of the Islamic calendar, occurs from dawn to sunset, for a variable duration due to latitude and seasonal shifts.2 Clinicians are likely to care for a patient who occasionally fasts. While there are potential benefits of fasting, clinicians need to consider the implications for patients who fast, particularly those receiving psychotropic medications.
Potential benefits for weight loss, mood
Some research suggests fasting is popular and may have benefits for an individual’s physical and mental health. In a 2020 online poll (N = 1,241), 24% of respondents said they had tried intermittent fasting, and 87% said the practice was very effective (50%) or somewhat effective (37%) in helping them lose weight.3 While more randomized control trials are needed to examine the practice’s effectiveness in promoting and maintaining weight loss, fasting has been linked to better glucose control in both humans and animals, and patients may have better adherence with fasting compared to caloric restriction alone.1 Improved mood, alertness, tranquility, and sometimes euphoria have been documented among individuals who fast, but these benefits may not be sustained.4 A prospective study of 462 participants who fasted during Ramadan found the practice reduced depression in patients with diabetes, possibly due to mindfulness, decreased inflammation from improved insulin sensitivity, and/or social cohesion.5
Be aware of the potential risks
Fasting may either improve or destabilize mood in people with bipolar disorder by disrupting circadian rhythm and sleep.2 Fasting might exacerbate underlying eating disorders.2 Increased dehydration escalates the risk for orthostatic hypotension, which might require discontinuing clozapine.6 Hypotension and toxicity might arise during lithium pharmacotherapy. The Table4 summarizes things to consider when caring for a patient who fasts while receiving pharmacotherapy.

Provide patients with guidance
Advise patients not to fast if you believe it might exacerbate their mental illness, and encourage them to discuss with their primary care physicians any potential worsening of physical illnesses.2 When caring for a patient who fasts for religious reasons, consider consulting with the patient’s religious leaders.2 If patients choose to fast, monitor them for mood destabilization and/or medication adverse effects. If possible, avoid altering drug treatment regimens during fasting, and carefully monitor whenever a pharmaceutical change is necessary. When appropriate, the use of long-acting injectable medications may minimize adverse effects while maintaining mood stability. Encourage patients who fast to ensure they remain hydrated and practice sleep hygiene while they fast.7
Intermittent fasting is the purposeful, restricted intake of food (and sometimes water), usually for health or religious reasons. Common forms are alternative-day fasting or time-restricted fasting, with variable ratios of days or hours for fasting and eating/drinking.1 For example, fasting during Ramadan, the ninth month of the Islamic calendar, occurs from dawn to sunset, for a variable duration due to latitude and seasonal shifts.2 Clinicians are likely to care for a patient who occasionally fasts. While there are potential benefits of fasting, clinicians need to consider the implications for patients who fast, particularly those receiving psychotropic medications.
Potential benefits for weight loss, mood
Some research suggests fasting is popular and may have benefits for an individual’s physical and mental health. In a 2020 online poll (N = 1,241), 24% of respondents said they had tried intermittent fasting, and 87% said the practice was very effective (50%) or somewhat effective (37%) in helping them lose weight.3 While more randomized control trials are needed to examine the practice’s effectiveness in promoting and maintaining weight loss, fasting has been linked to better glucose control in both humans and animals, and patients may have better adherence with fasting compared to caloric restriction alone.1 Improved mood, alertness, tranquility, and sometimes euphoria have been documented among individuals who fast, but these benefits may not be sustained.4 A prospective study of 462 participants who fasted during Ramadan found the practice reduced depression in patients with diabetes, possibly due to mindfulness, decreased inflammation from improved insulin sensitivity, and/or social cohesion.5
Be aware of the potential risks
Fasting may either improve or destabilize mood in people with bipolar disorder by disrupting circadian rhythm and sleep.2 Fasting might exacerbate underlying eating disorders.2 Increased dehydration escalates the risk for orthostatic hypotension, which might require discontinuing clozapine.6 Hypotension and toxicity might arise during lithium pharmacotherapy. The Table4 summarizes things to consider when caring for a patient who fasts while receiving pharmacotherapy.

Provide patients with guidance
Advise patients not to fast if you believe it might exacerbate their mental illness, and encourage them to discuss with their primary care physicians any potential worsening of physical illnesses.2 When caring for a patient who fasts for religious reasons, consider consulting with the patient’s religious leaders.2 If patients choose to fast, monitor them for mood destabilization and/or medication adverse effects. If possible, avoid altering drug treatment regimens during fasting, and carefully monitor whenever a pharmaceutical change is necessary. When appropriate, the use of long-acting injectable medications may minimize adverse effects while maintaining mood stability. Encourage patients who fast to ensure they remain hydrated and practice sleep hygiene while they fast.7
1. Dong TA, Sandesara PB, Dhindsa DS, et al. Intermittent fasting: a heart healthy dietary pattern? Am J Med. 2020;133(8):901-907.
2. Fond G, Macgregor A, Leboyer M, et al. Fasting in mood disorders: neurobiology and effectiveness. A review of the literature. Psychiatry Res. 2013;209(3):253-258.
3. Ballard J. Americans say this popular diet is effective and inexpensive. YouGov. February 24, 2020. Accessed January 6, 2022. https://today.yougov.com/topics/food/articles-reports/2020/02/24/most-effective-diet-intermittent-fasting-poll
4. Furqan Z, Awaad R, Kurdyak P, et al. Considerations for clinicians treating Muslim patients with psychiatric disorders during Ramadan. Lancet Psychiatry. 2019;6(7):556-557.
5. Al-Ozairi E, AlAwadhi MM, Al-Ozairi A, et al. A prospective study of the effect of fasting during the month of Ramadan on depression and diabetes distress in people with type 2 diabetes. Diabet Res Clin Pract. 2019;153:145-149.
6. Chehovich C, Demler TL, Leppien E. Impact of Ramadan fasting on medical and psychiatric health. Int Clin Psychopharmacol. 2019;34(6):317-322.
7. Farooq S, Nazar Z, Akhtar J, et al. Effect of fasting during Ramadan on serum lithium level and mental state in bipolar affective disorder. Int Clin Psychopharmacol. 2010;25(6):323-327.
1. Dong TA, Sandesara PB, Dhindsa DS, et al. Intermittent fasting: a heart healthy dietary pattern? Am J Med. 2020;133(8):901-907.
2. Fond G, Macgregor A, Leboyer M, et al. Fasting in mood disorders: neurobiology and effectiveness. A review of the literature. Psychiatry Res. 2013;209(3):253-258.
3. Ballard J. Americans say this popular diet is effective and inexpensive. YouGov. February 24, 2020. Accessed January 6, 2022. https://today.yougov.com/topics/food/articles-reports/2020/02/24/most-effective-diet-intermittent-fasting-poll
4. Furqan Z, Awaad R, Kurdyak P, et al. Considerations for clinicians treating Muslim patients with psychiatric disorders during Ramadan. Lancet Psychiatry. 2019;6(7):556-557.
5. Al-Ozairi E, AlAwadhi MM, Al-Ozairi A, et al. A prospective study of the effect of fasting during the month of Ramadan on depression and diabetes distress in people with type 2 diabetes. Diabet Res Clin Pract. 2019;153:145-149.
6. Chehovich C, Demler TL, Leppien E. Impact of Ramadan fasting on medical and psychiatric health. Int Clin Psychopharmacol. 2019;34(6):317-322.
7. Farooq S, Nazar Z, Akhtar J, et al. Effect of fasting during Ramadan on serum lithium level and mental state in bipolar affective disorder. Int Clin Psychopharmacol. 2010;25(6):323-327.
One in five female oncologists considering leaving academia, survey finds
More than half of respondents in academic medicine said they believe their gender adversely affects their likelihood for promotion, and 1 in 5 said they were considering leaving academia in the next 5 years.
Given the percentage of female oncologists planning to exit academia, “gender inequality is at high risk of continuing if the culture is not addressed,” write the authors in their study, published online Dec. 30 in JAMA Network Open.
Although women currently outnumber men in U.S. medical schools – a shift that first occurred in 2019 – female representation in academic oncology dwindles at more senior levels. Women represent 45% of hematology and oncology residents, only about 36% of academic faculty, and an even smaller percentage of leadership positions in academic medicine. Women, for instance, occupy about 31% of the chair positions in medical oncology, 17.4% in radiation oncology, and 11% in surgical oncology.
A team of researchers led by Emily C. Merfeld, MD, of the University of Wisconsin Hospitals and Clinics, Madison, set out to understand the factors influencing female oncologists’ decisions to pursue academic versus nonacademic career paths.
Dr. Merfeld and colleagues analyzed survey responses from 667 female oncologists between August 1 and Oct. 31, 2020 – 422 (63.2%) in academic medicine and 245 (36.8%) in nonacademic practice.
Overall, 1 in 4 oncologists said their spouse or partner and family “extremely or moderately” affected their decision to pursue academic practice.
Almost 43% of academic oncologists perceived time spent with loved ones as the biggest sacrifice related to pursuing a career in academic medicine. Approximately the same percentage (41.6%) of nonacademic oncologists perceived the pressure to achieve academic promotion as the most significant sacrifice associated with academic oncology, whereas only 22.4% perceived less time with loved ones as the biggest sacrifice.
“Although work-life balance was a concern for academic oncologists and may be a factor in female oncologists leaving academia, survey data suggested that women in nonacademic practice faced similar challenges,” the authors write.
More specifically, women in academic oncology reported working 2 more hours on the weekends compared to women not in academic medicine; however, both groups worked a similar number of hours during the week.
On the hiring front, almost 24% of academic oncologists said their gender had a “negative or somewhat negative” impact on their ability to get a job, compared with 21% of nonacademic oncologists. Conversely, nearly 28% of academic oncologists said their gender had a “positive or somewhat positive” influence on whether they were hired compared with 41.2% of nonacademic oncologists.
Respondents, however, perceived that gender strongly influenced promotion opportunities. More than half of the respondents – 54.6% of academic oncologists and 50.6% of nonacademic oncologists – believed they were less likely to be promoted than their male colleagues.
This perception aligns with findings from prior studies, which “found women were less likely than men to be promoted to associate professor, full professor, or department chair positions,” the authors write.
Overall, most respondents in each group – 71.3% in academic medicine and 68.6% in nonacademic practice – said they would choose the same career path again. But almost 22% of those in academia said they were “likely or very likely” to leave academic oncology in the next 5 years. Of these women, 28.2% said they would switch to industry employment and 25% would move to community practice.
“Contrary to popular assumptions,” the researchers note, “a spouse or partner and/or family were not a major factor in female oncologists favoring nonacademic careers, because this factor was similarly important to both academic and nonacademic oncologists.”
However, they note, “the increased financial compensation in nonacademic oncology may play a large role in some women’s career decisions.”
Making headway on gender equality?
In 2013, oncologist Katherine Reeder-Hayes, MD, MBA, now an associate at the University of North Carolina, Chapel Hill, published a study on gender equality in oncology in which she concluded that despite “an increasingly significant presence in the oncology physician workforce” women remained “under-represented in leadership positions and at the senior levels of academic medicine.”
Since then, Dr. Reeder-Hayes says that she has seen progress but recognizes the need for more.
“To some extent, I think that representation is improving over time due to factors outside the workplace – women are entering medical school in large numbers and may have more supportive partners and more social support for pursuing a professional career in general, [compared with] a decade or two ago,” Dr. Reeder-Hayes told this news organization.
On a personal level, she noted, “I do see many midcareer women assuming key leadership roles in my own institution.” However, she added, “I think the translation of those good candidates into increased representation in leadership probably varies widely across different institutions.”
In a 2019 editorial, researchers highlighted this variation while calling attention to the “notable progress” made by the American Association for Cancer Research (AACR). Specifically, the editorialists reported that women represent 40% of AACR members, 45% of the AACR Board of Directors, and half of the last 10 association presidents.
Editorial coauthor Elizabeth Jaffee, MD, deputy director of the Sidney Kimmel Comprehensive Cancer Center at Johns Hopkins, Baltimore, and former AACR president, told this news organization that she attributes this progress to “concrete measures to ensure equality throughout the organization,” which include gender balance on nominating and program committees as well as research meetings and providing opportunities for mentoring, leadership training, and networking.
Despite this positive change, the COVID-19 pandemic threatens to widen the gender imbalance. In a recent article, Julie Silver, MD, an expert in gender equity in medicine, told this news organization that she anticipates trouble ahead.
“There are many indications that women are leaving medicine in disproportionately high numbers,” said Dr. Silver, associate chair and director of cancer rehabilitation in the department of physical medicine and rehabilitation at Harvard Medical School, Boston. “A lack of fair pay and promotion opportunities that were present before COVID-19 are now combined with a host of pandemic-related challenges.”
In addition to salary and promotion disparities, the U.S. continues to suffer from “a chronic shortage of available, affordable, and high-quality childcare and a lack of federal-level policy initiatives or employer initiatives to broaden paid family leave and develop childcare infrastructure and workforce,” Dr. Reeder-Hayes said. Providing extended leave for new parents and on-site childcare could go a long way to improving this problem, she said.
However, Dr. Reeder-Hayes noted that perhaps the “leaky pipeline” problem in oncology highlights the fact that women “are making good decisions that reflect balanced life priorities, [and that] if we don’t structure job responsibilities, childcare, and pacing of promotion and tenure in ways that allow people to nurture other parts of their lives, employees will feel they’re being asked to sacrifice key things.”
In other words, she said, “it’s the workplace that needs to change if we’re going to convince [women], and many men with similar values, to stay.”
A version of this article first appeared on Medscape.com.
More than half of respondents in academic medicine said they believe their gender adversely affects their likelihood for promotion, and 1 in 5 said they were considering leaving academia in the next 5 years.
Given the percentage of female oncologists planning to exit academia, “gender inequality is at high risk of continuing if the culture is not addressed,” write the authors in their study, published online Dec. 30 in JAMA Network Open.
Although women currently outnumber men in U.S. medical schools – a shift that first occurred in 2019 – female representation in academic oncology dwindles at more senior levels. Women represent 45% of hematology and oncology residents, only about 36% of academic faculty, and an even smaller percentage of leadership positions in academic medicine. Women, for instance, occupy about 31% of the chair positions in medical oncology, 17.4% in radiation oncology, and 11% in surgical oncology.
A team of researchers led by Emily C. Merfeld, MD, of the University of Wisconsin Hospitals and Clinics, Madison, set out to understand the factors influencing female oncologists’ decisions to pursue academic versus nonacademic career paths.
Dr. Merfeld and colleagues analyzed survey responses from 667 female oncologists between August 1 and Oct. 31, 2020 – 422 (63.2%) in academic medicine and 245 (36.8%) in nonacademic practice.
Overall, 1 in 4 oncologists said their spouse or partner and family “extremely or moderately” affected their decision to pursue academic practice.
Almost 43% of academic oncologists perceived time spent with loved ones as the biggest sacrifice related to pursuing a career in academic medicine. Approximately the same percentage (41.6%) of nonacademic oncologists perceived the pressure to achieve academic promotion as the most significant sacrifice associated with academic oncology, whereas only 22.4% perceived less time with loved ones as the biggest sacrifice.
“Although work-life balance was a concern for academic oncologists and may be a factor in female oncologists leaving academia, survey data suggested that women in nonacademic practice faced similar challenges,” the authors write.
More specifically, women in academic oncology reported working 2 more hours on the weekends compared to women not in academic medicine; however, both groups worked a similar number of hours during the week.
On the hiring front, almost 24% of academic oncologists said their gender had a “negative or somewhat negative” impact on their ability to get a job, compared with 21% of nonacademic oncologists. Conversely, nearly 28% of academic oncologists said their gender had a “positive or somewhat positive” influence on whether they were hired compared with 41.2% of nonacademic oncologists.
Respondents, however, perceived that gender strongly influenced promotion opportunities. More than half of the respondents – 54.6% of academic oncologists and 50.6% of nonacademic oncologists – believed they were less likely to be promoted than their male colleagues.
This perception aligns with findings from prior studies, which “found women were less likely than men to be promoted to associate professor, full professor, or department chair positions,” the authors write.
Overall, most respondents in each group – 71.3% in academic medicine and 68.6% in nonacademic practice – said they would choose the same career path again. But almost 22% of those in academia said they were “likely or very likely” to leave academic oncology in the next 5 years. Of these women, 28.2% said they would switch to industry employment and 25% would move to community practice.
“Contrary to popular assumptions,” the researchers note, “a spouse or partner and/or family were not a major factor in female oncologists favoring nonacademic careers, because this factor was similarly important to both academic and nonacademic oncologists.”
However, they note, “the increased financial compensation in nonacademic oncology may play a large role in some women’s career decisions.”
Making headway on gender equality?
In 2013, oncologist Katherine Reeder-Hayes, MD, MBA, now an associate at the University of North Carolina, Chapel Hill, published a study on gender equality in oncology in which she concluded that despite “an increasingly significant presence in the oncology physician workforce” women remained “under-represented in leadership positions and at the senior levels of academic medicine.”
Since then, Dr. Reeder-Hayes says that she has seen progress but recognizes the need for more.
“To some extent, I think that representation is improving over time due to factors outside the workplace – women are entering medical school in large numbers and may have more supportive partners and more social support for pursuing a professional career in general, [compared with] a decade or two ago,” Dr. Reeder-Hayes told this news organization.
On a personal level, she noted, “I do see many midcareer women assuming key leadership roles in my own institution.” However, she added, “I think the translation of those good candidates into increased representation in leadership probably varies widely across different institutions.”
In a 2019 editorial, researchers highlighted this variation while calling attention to the “notable progress” made by the American Association for Cancer Research (AACR). Specifically, the editorialists reported that women represent 40% of AACR members, 45% of the AACR Board of Directors, and half of the last 10 association presidents.
Editorial coauthor Elizabeth Jaffee, MD, deputy director of the Sidney Kimmel Comprehensive Cancer Center at Johns Hopkins, Baltimore, and former AACR president, told this news organization that she attributes this progress to “concrete measures to ensure equality throughout the organization,” which include gender balance on nominating and program committees as well as research meetings and providing opportunities for mentoring, leadership training, and networking.
Despite this positive change, the COVID-19 pandemic threatens to widen the gender imbalance. In a recent article, Julie Silver, MD, an expert in gender equity in medicine, told this news organization that she anticipates trouble ahead.
“There are many indications that women are leaving medicine in disproportionately high numbers,” said Dr. Silver, associate chair and director of cancer rehabilitation in the department of physical medicine and rehabilitation at Harvard Medical School, Boston. “A lack of fair pay and promotion opportunities that were present before COVID-19 are now combined with a host of pandemic-related challenges.”
In addition to salary and promotion disparities, the U.S. continues to suffer from “a chronic shortage of available, affordable, and high-quality childcare and a lack of federal-level policy initiatives or employer initiatives to broaden paid family leave and develop childcare infrastructure and workforce,” Dr. Reeder-Hayes said. Providing extended leave for new parents and on-site childcare could go a long way to improving this problem, she said.
However, Dr. Reeder-Hayes noted that perhaps the “leaky pipeline” problem in oncology highlights the fact that women “are making good decisions that reflect balanced life priorities, [and that] if we don’t structure job responsibilities, childcare, and pacing of promotion and tenure in ways that allow people to nurture other parts of their lives, employees will feel they’re being asked to sacrifice key things.”
In other words, she said, “it’s the workplace that needs to change if we’re going to convince [women], and many men with similar values, to stay.”
A version of this article first appeared on Medscape.com.
More than half of respondents in academic medicine said they believe their gender adversely affects their likelihood for promotion, and 1 in 5 said they were considering leaving academia in the next 5 years.
Given the percentage of female oncologists planning to exit academia, “gender inequality is at high risk of continuing if the culture is not addressed,” write the authors in their study, published online Dec. 30 in JAMA Network Open.
Although women currently outnumber men in U.S. medical schools – a shift that first occurred in 2019 – female representation in academic oncology dwindles at more senior levels. Women represent 45% of hematology and oncology residents, only about 36% of academic faculty, and an even smaller percentage of leadership positions in academic medicine. Women, for instance, occupy about 31% of the chair positions in medical oncology, 17.4% in radiation oncology, and 11% in surgical oncology.
A team of researchers led by Emily C. Merfeld, MD, of the University of Wisconsin Hospitals and Clinics, Madison, set out to understand the factors influencing female oncologists’ decisions to pursue academic versus nonacademic career paths.
Dr. Merfeld and colleagues analyzed survey responses from 667 female oncologists between August 1 and Oct. 31, 2020 – 422 (63.2%) in academic medicine and 245 (36.8%) in nonacademic practice.
Overall, 1 in 4 oncologists said their spouse or partner and family “extremely or moderately” affected their decision to pursue academic practice.
Almost 43% of academic oncologists perceived time spent with loved ones as the biggest sacrifice related to pursuing a career in academic medicine. Approximately the same percentage (41.6%) of nonacademic oncologists perceived the pressure to achieve academic promotion as the most significant sacrifice associated with academic oncology, whereas only 22.4% perceived less time with loved ones as the biggest sacrifice.
“Although work-life balance was a concern for academic oncologists and may be a factor in female oncologists leaving academia, survey data suggested that women in nonacademic practice faced similar challenges,” the authors write.
More specifically, women in academic oncology reported working 2 more hours on the weekends compared to women not in academic medicine; however, both groups worked a similar number of hours during the week.
On the hiring front, almost 24% of academic oncologists said their gender had a “negative or somewhat negative” impact on their ability to get a job, compared with 21% of nonacademic oncologists. Conversely, nearly 28% of academic oncologists said their gender had a “positive or somewhat positive” influence on whether they were hired compared with 41.2% of nonacademic oncologists.
Respondents, however, perceived that gender strongly influenced promotion opportunities. More than half of the respondents – 54.6% of academic oncologists and 50.6% of nonacademic oncologists – believed they were less likely to be promoted than their male colleagues.
This perception aligns with findings from prior studies, which “found women were less likely than men to be promoted to associate professor, full professor, or department chair positions,” the authors write.
Overall, most respondents in each group – 71.3% in academic medicine and 68.6% in nonacademic practice – said they would choose the same career path again. But almost 22% of those in academia said they were “likely or very likely” to leave academic oncology in the next 5 years. Of these women, 28.2% said they would switch to industry employment and 25% would move to community practice.
“Contrary to popular assumptions,” the researchers note, “a spouse or partner and/or family were not a major factor in female oncologists favoring nonacademic careers, because this factor was similarly important to both academic and nonacademic oncologists.”
However, they note, “the increased financial compensation in nonacademic oncology may play a large role in some women’s career decisions.”
Making headway on gender equality?
In 2013, oncologist Katherine Reeder-Hayes, MD, MBA, now an associate at the University of North Carolina, Chapel Hill, published a study on gender equality in oncology in which she concluded that despite “an increasingly significant presence in the oncology physician workforce” women remained “under-represented in leadership positions and at the senior levels of academic medicine.”
Since then, Dr. Reeder-Hayes says that she has seen progress but recognizes the need for more.
“To some extent, I think that representation is improving over time due to factors outside the workplace – women are entering medical school in large numbers and may have more supportive partners and more social support for pursuing a professional career in general, [compared with] a decade or two ago,” Dr. Reeder-Hayes told this news organization.
On a personal level, she noted, “I do see many midcareer women assuming key leadership roles in my own institution.” However, she added, “I think the translation of those good candidates into increased representation in leadership probably varies widely across different institutions.”
In a 2019 editorial, researchers highlighted this variation while calling attention to the “notable progress” made by the American Association for Cancer Research (AACR). Specifically, the editorialists reported that women represent 40% of AACR members, 45% of the AACR Board of Directors, and half of the last 10 association presidents.
Editorial coauthor Elizabeth Jaffee, MD, deputy director of the Sidney Kimmel Comprehensive Cancer Center at Johns Hopkins, Baltimore, and former AACR president, told this news organization that she attributes this progress to “concrete measures to ensure equality throughout the organization,” which include gender balance on nominating and program committees as well as research meetings and providing opportunities for mentoring, leadership training, and networking.
Despite this positive change, the COVID-19 pandemic threatens to widen the gender imbalance. In a recent article, Julie Silver, MD, an expert in gender equity in medicine, told this news organization that she anticipates trouble ahead.
“There are many indications that women are leaving medicine in disproportionately high numbers,” said Dr. Silver, associate chair and director of cancer rehabilitation in the department of physical medicine and rehabilitation at Harvard Medical School, Boston. “A lack of fair pay and promotion opportunities that were present before COVID-19 are now combined with a host of pandemic-related challenges.”
In addition to salary and promotion disparities, the U.S. continues to suffer from “a chronic shortage of available, affordable, and high-quality childcare and a lack of federal-level policy initiatives or employer initiatives to broaden paid family leave and develop childcare infrastructure and workforce,” Dr. Reeder-Hayes said. Providing extended leave for new parents and on-site childcare could go a long way to improving this problem, she said.
However, Dr. Reeder-Hayes noted that perhaps the “leaky pipeline” problem in oncology highlights the fact that women “are making good decisions that reflect balanced life priorities, [and that] if we don’t structure job responsibilities, childcare, and pacing of promotion and tenure in ways that allow people to nurture other parts of their lives, employees will feel they’re being asked to sacrifice key things.”
In other words, she said, “it’s the workplace that needs to change if we’re going to convince [women], and many men with similar values, to stay.”
A version of this article first appeared on Medscape.com.
FROM JAMA NETWORK OPEN
Sarcoidosis
THE COMPARISON
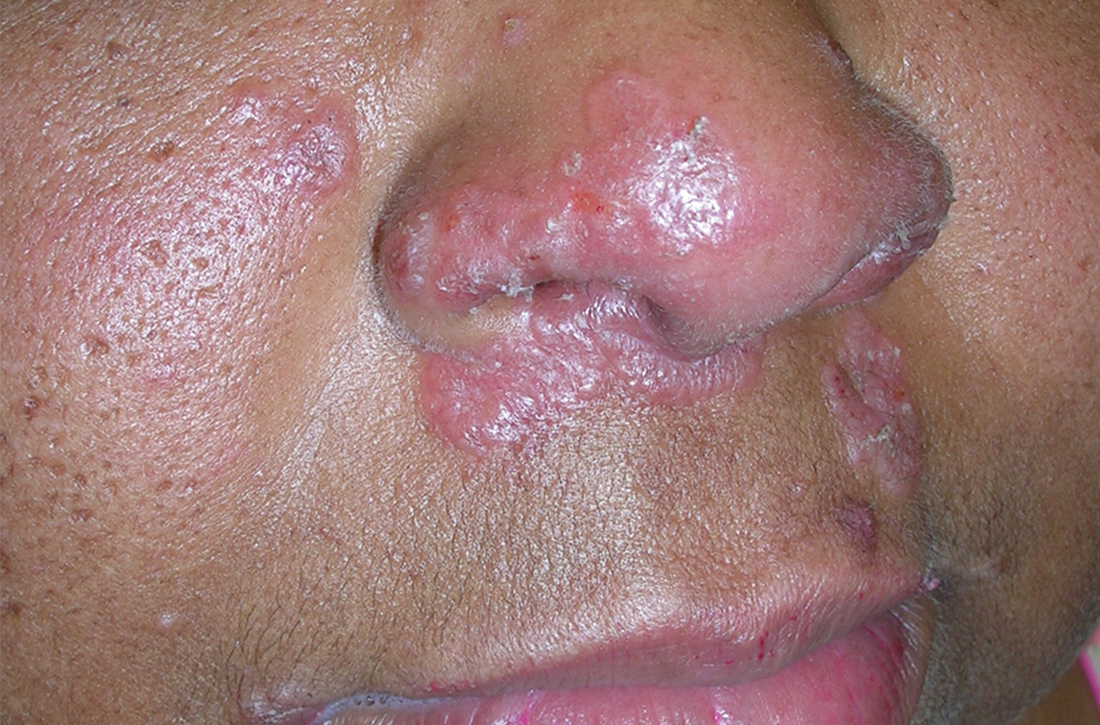
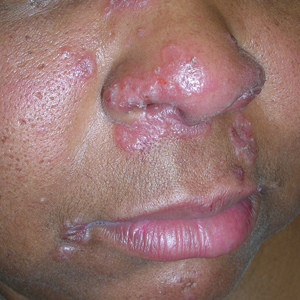
A Pink, elevated, granulomatous, indurated plaques on the face, including the nasal alae, of a 52-year-old woman with a darker skin tone.
B Orange and pink, elevated, granulomatous, indurated plaques on the face of a 55-year-old woman with a lighter skin tone.
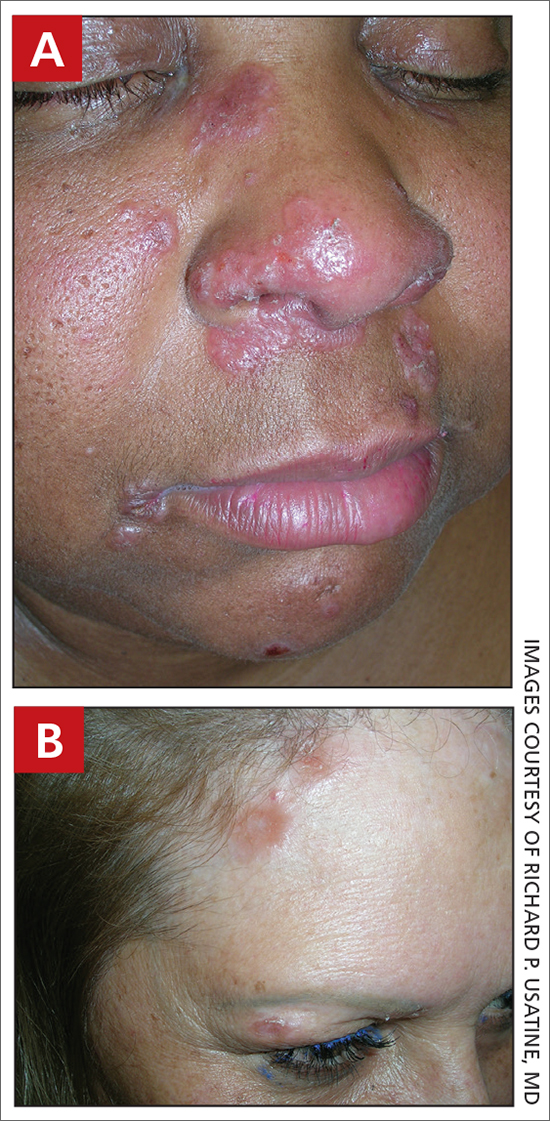
Sarcoidosis is a granulomatous disease that may affect the skin in addition to multiple body organ systems, including the lungs. Bilateral hilar adenopathy on a chest radiograph is the most common finding. Sarcoidosis also has a variety of cutaneous manifestations. Early diagnosis is vital, as patients with sarcoidosis and pulmonary fibrosis have a shortened life span compared to the overall population.1 With a growing skin of color population, it is important to recognize sarcoidosis as soon as possible.2
Epidemiology
People of African descent have the highest sarcoidosis prevalence in the United States.3 In the United States, the incidence of sarcoidosis in Black individuals peaks in the fourth decade of life. A 5-year study in a US health maintenance organization found that the age-adjusted annual incidence was 10.9 per 100,000 cases among Whites and 35.5 per 100,000 cases among Blacks.4
Key clinical features in people with darker skin tones:
• Papules are seen in sarcoidosis, primarily on the face, and may start as orange hued or yellow-brown and then become brown-red or pink to violaceous before involuting into faint macules.5-7
• When round or oval sarcoid plaques appear, they often are more erythematous.
• In skin of color, plaques may become hypopigmented.8
• Erythema nodosum, the most common nonspecific cutaneous lesion seen in sarcoidosis, is less commonly seen in those of African and Asian descent.9-11 This is in contrast to distinctive forms of specific sarcoid skin lesions such as lupus pernio and scar sarcoidosis, as well as papules and plaques and minor forms of specific sarcoid skin lesions including subcutaneous nodules; hypopigmented macules; psoriasiform lesions; and ulcerative, localized erythrodermic, ichthyosiform, scalp, and nail lesions.
• Lupus pernio is a cutaneous manifestation of sarcoidosis that appears on the face. It looks similar to lupus erythematosus and occurs most commonly in women of African descent.8,12
• Hypopigmented lesions are more common in those with darker skin tones.9
• Ulcerative lesions are more common in those of African descent and women.13
• Scalp sarcoidosis is more common in patients of African descent.14
• Sarcoidosis may develop at sites of trauma, such as scars and tattoos.15-17
Worth noting
The cutaneous lesions seen in sarcoidosis may be emotionally devastating and disfiguring. Due to the variety of clinical manifestations, sarcoidosis may be misdiagnosed, leading to delays in treatment.18
Health disparity highlight
Patients older than 40 years presenting with sarcoidosis and those of African descent have a worse prognosis.19 Despite adjustment for race, ethnic group, age, and sex, patients with low income and financial barriers present with more severe sarcoidosis.20
1. Nardi A, Brillet P-Y, Letoumelin P, et al. Stage IV sarcoidosis: comparison of survival with the general population and causes of death. Eur Respir J. 2011;38:1368-1373.
2. Heath CR, David J, Taylor SC. Sarcoidosis: are there differences in your skin of color patients? J Am Acad Dermatol. 2012;66: 121.e1-121.e14.
3. Sève P, Pacheco Y, Durupt F, et al. Sarcoidosis: a clinical overview from symptoms to diagnosis. Cells. 2021;10:766. doi:10.3390/ cells10040766
4. Rybicki BA, Major M, Popovich J Jr, et al. Racial differences in sarcoidosis incidence: a 5-year study in a health maintenance organization. Am J Epidemiol. 1997;145:234-241. doi:10.1093/ oxfordjournals.aje.a009096
5. Mahajan VK, Sharma NL, Sharma RC, et al. Cutaneous sarcoidosis: clinical profile of 23 Indian patients. Indian J Dermatol Venerol Leprol. 2007;73:16-21.
6. Yanardag H, Pamuk ON, Karayel T. Cutaneous involvement in sarcoidosis: analysis of features in 170 patients. Respir Med. 2003;97:978-982.
7. Olive KE, Kartaria YP. Cutaneous manifestations of sarcoidosis to other organ system involvement, abnormal laboratory measurements, and disease course. Arch Intern Med. 1985;145:1811-1814.
8. Mañá J, Marcoval J, Graells J, et al. Cutaneous involvement in sarcoidosis. relationship to systemic disease. Arch Dermatol. 1997;133:882-888. doi:10.1001/archderm.1997.03890430098013
9. Minus HR, Grimes PE. Cutaneous manifestations of sarcoidosis in blacks. Cutis. 1983;32:361-364.
10. Edmondstone WM, Wilson AG. Sarcoidosis in Caucasians, blacks and Asians in London. Br J Dis Chest. 1985;79:27-36.
11. James DG, Neville E, Siltzbach LE. Worldwide review of sarcoidosis. Ann N Y Acad Sci. 1976;278:321-334.
12. Hunninghake GW, Costabel U, Ando M, et al. ATS/ERS/WASOG statement on sarcoidosis. American Thoracic Society/European Respiratory Society/World Association of Sarcoidosis and other Granulomatous Disorders. Sarcoidosis Vasc Diffuse Lung Dis. 1999;16:149-173.
13. Albertini JG, Tyler W, Miller OF III. Ulcerative sarcoidosis: case report and review of literature. Arch Dermatol. 1997;133:215-219.
14. Marchell RM, Judson MA. Chronic cutaneous lesions of sarcoidosis. Clin Dermatol. 2007;25:295-302.
15. Nayar M. Sarcoidosis on ritual scarification. Int J Dermatol. 1993;32:116-118.
16. Chudomirova K, Velichkva L, Anavi B. Recurrent sarcoidosis in skin scars accompanying systemic sarcoidosis. J Eur Acad Dermatol Venerol. 2003;17:360-361.
17. Kim YC, Triffet MK, Gibson LE. Foreign bodies in sarcoidosis. Am J Dermatopathol. 2000;22:408-412.
18. Iannuzzi MC, Rybicki BA, Teirstein AS. Sarcoidosis. N Engl J Med. 2007; 357:2153-2165.
19. Nunes H, Bouvry D, Soler P, et al. Sarcoidosis. Orphanet J Rare Dis. 2007;2:46. doi:10.1186/1750-1172-2-46
20. Baughman RP, Teirstein AS, Judson MA, et al. Clinical characteristics of patients in a case control study of sarcoidosis. Am J Respir Crit Care Med. 2001;164:1885-1889.
THE COMPARISON
A Pink, elevated, granulomatous, indurated plaques on the face, including the nasal alae, of a 52-year-old woman with a darker skin tone.
B Orange and pink, elevated, granulomatous, indurated plaques on the face of a 55-year-old woman with a lighter skin tone.

Sarcoidosis is a granulomatous disease that may affect the skin in addition to multiple body organ systems, including the lungs. Bilateral hilar adenopathy on a chest radiograph is the most common finding. Sarcoidosis also has a variety of cutaneous manifestations. Early diagnosis is vital, as patients with sarcoidosis and pulmonary fibrosis have a shortened life span compared to the overall population.1 With a growing skin of color population, it is important to recognize sarcoidosis as soon as possible.2
Epidemiology
People of African descent have the highest sarcoidosis prevalence in the United States.3 In the United States, the incidence of sarcoidosis in Black individuals peaks in the fourth decade of life. A 5-year study in a US health maintenance organization found that the age-adjusted annual incidence was 10.9 per 100,000 cases among Whites and 35.5 per 100,000 cases among Blacks.4
Key clinical features in people with darker skin tones:
• Papules are seen in sarcoidosis, primarily on the face, and may start as orange hued or yellow-brown and then become brown-red or pink to violaceous before involuting into faint macules.5-7
• When round or oval sarcoid plaques appear, they often are more erythematous.
• In skin of color, plaques may become hypopigmented.8
• Erythema nodosum, the most common nonspecific cutaneous lesion seen in sarcoidosis, is less commonly seen in those of African and Asian descent.9-11 This is in contrast to distinctive forms of specific sarcoid skin lesions such as lupus pernio and scar sarcoidosis, as well as papules and plaques and minor forms of specific sarcoid skin lesions including subcutaneous nodules; hypopigmented macules; psoriasiform lesions; and ulcerative, localized erythrodermic, ichthyosiform, scalp, and nail lesions.
• Lupus pernio is a cutaneous manifestation of sarcoidosis that appears on the face. It looks similar to lupus erythematosus and occurs most commonly in women of African descent.8,12
• Hypopigmented lesions are more common in those with darker skin tones.9
• Ulcerative lesions are more common in those of African descent and women.13
• Scalp sarcoidosis is more common in patients of African descent.14
• Sarcoidosis may develop at sites of trauma, such as scars and tattoos.15-17
Worth noting
The cutaneous lesions seen in sarcoidosis may be emotionally devastating and disfiguring. Due to the variety of clinical manifestations, sarcoidosis may be misdiagnosed, leading to delays in treatment.18
Health disparity highlight
Patients older than 40 years presenting with sarcoidosis and those of African descent have a worse prognosis.19 Despite adjustment for race, ethnic group, age, and sex, patients with low income and financial barriers present with more severe sarcoidosis.20
THE COMPARISON
A Pink, elevated, granulomatous, indurated plaques on the face, including the nasal alae, of a 52-year-old woman with a darker skin tone.
B Orange and pink, elevated, granulomatous, indurated plaques on the face of a 55-year-old woman with a lighter skin tone.

Sarcoidosis is a granulomatous disease that may affect the skin in addition to multiple body organ systems, including the lungs. Bilateral hilar adenopathy on a chest radiograph is the most common finding. Sarcoidosis also has a variety of cutaneous manifestations. Early diagnosis is vital, as patients with sarcoidosis and pulmonary fibrosis have a shortened life span compared to the overall population.1 With a growing skin of color population, it is important to recognize sarcoidosis as soon as possible.2
Epidemiology
People of African descent have the highest sarcoidosis prevalence in the United States.3 In the United States, the incidence of sarcoidosis in Black individuals peaks in the fourth decade of life. A 5-year study in a US health maintenance organization found that the age-adjusted annual incidence was 10.9 per 100,000 cases among Whites and 35.5 per 100,000 cases among Blacks.4
Key clinical features in people with darker skin tones:
• Papules are seen in sarcoidosis, primarily on the face, and may start as orange hued or yellow-brown and then become brown-red or pink to violaceous before involuting into faint macules.5-7
• When round or oval sarcoid plaques appear, they often are more erythematous.
• In skin of color, plaques may become hypopigmented.8
• Erythema nodosum, the most common nonspecific cutaneous lesion seen in sarcoidosis, is less commonly seen in those of African and Asian descent.9-11 This is in contrast to distinctive forms of specific sarcoid skin lesions such as lupus pernio and scar sarcoidosis, as well as papules and plaques and minor forms of specific sarcoid skin lesions including subcutaneous nodules; hypopigmented macules; psoriasiform lesions; and ulcerative, localized erythrodermic, ichthyosiform, scalp, and nail lesions.
• Lupus pernio is a cutaneous manifestation of sarcoidosis that appears on the face. It looks similar to lupus erythematosus and occurs most commonly in women of African descent.8,12
• Hypopigmented lesions are more common in those with darker skin tones.9
• Ulcerative lesions are more common in those of African descent and women.13
• Scalp sarcoidosis is more common in patients of African descent.14
• Sarcoidosis may develop at sites of trauma, such as scars and tattoos.15-17
Worth noting
The cutaneous lesions seen in sarcoidosis may be emotionally devastating and disfiguring. Due to the variety of clinical manifestations, sarcoidosis may be misdiagnosed, leading to delays in treatment.18
Health disparity highlight
Patients older than 40 years presenting with sarcoidosis and those of African descent have a worse prognosis.19 Despite adjustment for race, ethnic group, age, and sex, patients with low income and financial barriers present with more severe sarcoidosis.20
1. Nardi A, Brillet P-Y, Letoumelin P, et al. Stage IV sarcoidosis: comparison of survival with the general population and causes of death. Eur Respir J. 2011;38:1368-1373.
2. Heath CR, David J, Taylor SC. Sarcoidosis: are there differences in your skin of color patients? J Am Acad Dermatol. 2012;66: 121.e1-121.e14.
3. Sève P, Pacheco Y, Durupt F, et al. Sarcoidosis: a clinical overview from symptoms to diagnosis. Cells. 2021;10:766. doi:10.3390/ cells10040766
4. Rybicki BA, Major M, Popovich J Jr, et al. Racial differences in sarcoidosis incidence: a 5-year study in a health maintenance organization. Am J Epidemiol. 1997;145:234-241. doi:10.1093/ oxfordjournals.aje.a009096
5. Mahajan VK, Sharma NL, Sharma RC, et al. Cutaneous sarcoidosis: clinical profile of 23 Indian patients. Indian J Dermatol Venerol Leprol. 2007;73:16-21.
6. Yanardag H, Pamuk ON, Karayel T. Cutaneous involvement in sarcoidosis: analysis of features in 170 patients. Respir Med. 2003;97:978-982.
7. Olive KE, Kartaria YP. Cutaneous manifestations of sarcoidosis to other organ system involvement, abnormal laboratory measurements, and disease course. Arch Intern Med. 1985;145:1811-1814.
8. Mañá J, Marcoval J, Graells J, et al. Cutaneous involvement in sarcoidosis. relationship to systemic disease. Arch Dermatol. 1997;133:882-888. doi:10.1001/archderm.1997.03890430098013
9. Minus HR, Grimes PE. Cutaneous manifestations of sarcoidosis in blacks. Cutis. 1983;32:361-364.
10. Edmondstone WM, Wilson AG. Sarcoidosis in Caucasians, blacks and Asians in London. Br J Dis Chest. 1985;79:27-36.
11. James DG, Neville E, Siltzbach LE. Worldwide review of sarcoidosis. Ann N Y Acad Sci. 1976;278:321-334.
12. Hunninghake GW, Costabel U, Ando M, et al. ATS/ERS/WASOG statement on sarcoidosis. American Thoracic Society/European Respiratory Society/World Association of Sarcoidosis and other Granulomatous Disorders. Sarcoidosis Vasc Diffuse Lung Dis. 1999;16:149-173.
13. Albertini JG, Tyler W, Miller OF III. Ulcerative sarcoidosis: case report and review of literature. Arch Dermatol. 1997;133:215-219.
14. Marchell RM, Judson MA. Chronic cutaneous lesions of sarcoidosis. Clin Dermatol. 2007;25:295-302.
15. Nayar M. Sarcoidosis on ritual scarification. Int J Dermatol. 1993;32:116-118.
16. Chudomirova K, Velichkva L, Anavi B. Recurrent sarcoidosis in skin scars accompanying systemic sarcoidosis. J Eur Acad Dermatol Venerol. 2003;17:360-361.
17. Kim YC, Triffet MK, Gibson LE. Foreign bodies in sarcoidosis. Am J Dermatopathol. 2000;22:408-412.
18. Iannuzzi MC, Rybicki BA, Teirstein AS. Sarcoidosis. N Engl J Med. 2007; 357:2153-2165.
19. Nunes H, Bouvry D, Soler P, et al. Sarcoidosis. Orphanet J Rare Dis. 2007;2:46. doi:10.1186/1750-1172-2-46
20. Baughman RP, Teirstein AS, Judson MA, et al. Clinical characteristics of patients in a case control study of sarcoidosis. Am J Respir Crit Care Med. 2001;164:1885-1889.
1. Nardi A, Brillet P-Y, Letoumelin P, et al. Stage IV sarcoidosis: comparison of survival with the general population and causes of death. Eur Respir J. 2011;38:1368-1373.
2. Heath CR, David J, Taylor SC. Sarcoidosis: are there differences in your skin of color patients? J Am Acad Dermatol. 2012;66: 121.e1-121.e14.
3. Sève P, Pacheco Y, Durupt F, et al. Sarcoidosis: a clinical overview from symptoms to diagnosis. Cells. 2021;10:766. doi:10.3390/ cells10040766
4. Rybicki BA, Major M, Popovich J Jr, et al. Racial differences in sarcoidosis incidence: a 5-year study in a health maintenance organization. Am J Epidemiol. 1997;145:234-241. doi:10.1093/ oxfordjournals.aje.a009096
5. Mahajan VK, Sharma NL, Sharma RC, et al. Cutaneous sarcoidosis: clinical profile of 23 Indian patients. Indian J Dermatol Venerol Leprol. 2007;73:16-21.
6. Yanardag H, Pamuk ON, Karayel T. Cutaneous involvement in sarcoidosis: analysis of features in 170 patients. Respir Med. 2003;97:978-982.
7. Olive KE, Kartaria YP. Cutaneous manifestations of sarcoidosis to other organ system involvement, abnormal laboratory measurements, and disease course. Arch Intern Med. 1985;145:1811-1814.
8. Mañá J, Marcoval J, Graells J, et al. Cutaneous involvement in sarcoidosis. relationship to systemic disease. Arch Dermatol. 1997;133:882-888. doi:10.1001/archderm.1997.03890430098013
9. Minus HR, Grimes PE. Cutaneous manifestations of sarcoidosis in blacks. Cutis. 1983;32:361-364.
10. Edmondstone WM, Wilson AG. Sarcoidosis in Caucasians, blacks and Asians in London. Br J Dis Chest. 1985;79:27-36.
11. James DG, Neville E, Siltzbach LE. Worldwide review of sarcoidosis. Ann N Y Acad Sci. 1976;278:321-334.
12. Hunninghake GW, Costabel U, Ando M, et al. ATS/ERS/WASOG statement on sarcoidosis. American Thoracic Society/European Respiratory Society/World Association of Sarcoidosis and other Granulomatous Disorders. Sarcoidosis Vasc Diffuse Lung Dis. 1999;16:149-173.
13. Albertini JG, Tyler W, Miller OF III. Ulcerative sarcoidosis: case report and review of literature. Arch Dermatol. 1997;133:215-219.
14. Marchell RM, Judson MA. Chronic cutaneous lesions of sarcoidosis. Clin Dermatol. 2007;25:295-302.
15. Nayar M. Sarcoidosis on ritual scarification. Int J Dermatol. 1993;32:116-118.
16. Chudomirova K, Velichkva L, Anavi B. Recurrent sarcoidosis in skin scars accompanying systemic sarcoidosis. J Eur Acad Dermatol Venerol. 2003;17:360-361.
17. Kim YC, Triffet MK, Gibson LE. Foreign bodies in sarcoidosis. Am J Dermatopathol. 2000;22:408-412.
18. Iannuzzi MC, Rybicki BA, Teirstein AS. Sarcoidosis. N Engl J Med. 2007; 357:2153-2165.
19. Nunes H, Bouvry D, Soler P, et al. Sarcoidosis. Orphanet J Rare Dis. 2007;2:46. doi:10.1186/1750-1172-2-46
20. Baughman RP, Teirstein AS, Judson MA, et al. Clinical characteristics of patients in a case control study of sarcoidosis. Am J Respir Crit Care Med. 2001;164:1885-1889.
100 coauthored papers, 10 years: Cancer transplant pioneers model 'team science'
On July 29, 2021, Sergio Giralt, MD, deputy division head of the division of hematologic malignancies and Miguel-Angel Perales, MD, chief of the adult bone marrow transplant service at MSKCC, published their 100th peer-reviewed paper as coauthors. Listing hundreds of such articles on a CV is standard for top-tier physicians, but the pair had gone one better: 100 publications written together in 10 years.
Their centenary article hit scientific newsstands almost exactly a decade after their first joint paper, which appeared in September 2011, not long after they met.
Born in Cuba, Dr. Giralt grew up in Venezuela. From the age of 14, he knew that medicine was his path, and in 1984 he earned a medical degree from the Universidad Central de Venezuela, Caracas. Next came a research position at Harvard Medical School, a residency at the Good Samaritan Hospital, Cincinnati, and a fellowship at the University of Texas MD Anderson Cancer Center, Houston. Dr. Giralt arrived at MSKCC in 2010 as the new chief of the adult bone marrow transplant service. There he was introduced to a new colleague, Dr. Perales. They soon learned that in addition to expertise in hematology, they had second language in common: Spanish.
Dr. Giralt said: “We both have a Spanish background and in a certain sense, there was an affinity there. ... We both have shared experiences.”
Dr. Perales was brought up in Belgium, a European nation with three official languages: French, Dutch, and German. He speaks five tongues in all and learned Spanish from his father, who came from Spain.
Fluency in Spanish enables both physicians to take care of the many New Yorkers who are more comfortable in that language – especially when navigating cancer treatment. However, both Dr. Giralt and Dr. Perales said that a second language is more than a professional tool. They described the enjoyable change of persona that happens when they switch to Spanish.
“People who are multilingual have different roles [as much as] different languages,” said Dr. Perales. “When I’m in Spanish, part of my brain is [thinking back to] summer vacations and hanging out with my cousins.”
When it comes to clinical science, however, English is the language of choice.
Global leaders in HSCT
Dr. Giralt and Dr. Perales are known worldwide in the field of allogeneic HSCT, a potentially curative treatment for an elongating list of both malignant and nonmalignant diseases.
In 1973, MSKCC conducted the first bone-marrow transplant from an unrelated donor. Fifty years on, medical oncologists in the United States conduct approximately 8,500 allogeneic transplants each year, 72% to treat acute leukemias or myelodysplastic syndrome (MDS).
However, stripping the immune system with intensive chemotherapy ‘conditioning,’ then rebuilding it with non-diseased donor hematopoietic cells is a hazardous undertaking. Older patients are less likely to survive the intensive conditioning, so historically have missed out. Also, even with a good human leukocyte antigen (HLA) match, the recipient needs often brutal immunosuppression.
Since Dr. Giralt and Dr. Perales began their partnership in 2010, the goals of their work have not changed: to develop safer, lower-intensity transplantation suitable for older, more vulnerable patients and reduce fearsome posttransplant sequelae such as graft-versus-host disease (GVHD).
Dr. Giralt’s publication list spans more than 600 peer-reviewed papers, articles and book chapters, almost exclusively on HSCT. Dr. Perales has more than 300 publication credits on the topic.
The two paired up on their first paper just months after Dr. Giralt arrived at MSKCC. That article, published in Biology of Blood and Marrow Transplantation, compared umbilical cord blood for HSCT with donor blood in 367 people with a variety of hematologic malignancies, including acute and chronic leukemias, MDS, and lymphoma.
The MSKCC team found that transplant-related mortality in the first 180 days was higher for the cord blood (21%), but thereafter mortality and relapse were much lower than for donated blood, with the result that 2-year progression-free survival of 55% was similar. Dr. Perales, Dr. Giralt and their coauthors concluded that the data provided “strong support” for further work on cord blood as an alternative stem-cell source.
During their first decade of collaboration, Dr. Giralt and Dr. Perales worked on any promising avenue that could improve outcomes and the experience of HSCT recipients, including reduced-intensity conditioning regimens to allow older adults to benefit from curative HSCT and donor T-cell depletion by CD34 selection, to reduce graft-versus-host disease (GVHD).
The CD34 protein is typically found on the surface of early stage and highly active stem cell types. Selecting these cell types using a range of techniques can eliminate many other potentially interfering or inactive cells. This enriches the transplant population with the most effective cells and can lower the risk of GVHD.
The 100th paper on which Dr. Giralt and Dr. Perales were coauthors was published in Blood Advances on July 27, 2021. The retrospective study examined the fate of 58 MSKCC patients with a rare form of chronic lymphocytic leukemia, CLL with Richter’s transformation (CLL-RT). It was the largest such study to date of this rare disease.
M.D. Anderson Cancer Center had shown in 2006 that, despite chemotherapy, overall survival in patients with CLL-RT was approximately 8 months. HSCT improved survival dramatically (75% at 3 years; n = 7). However, with the advent of novel targeted drugs for CLL such as ibrutinib (Imbruvica), venetoclax (Venclexta), or idelalisib (Zydelig), the MSKCC team asked themselves: What was the role of reduced-intensive conditioning HSCT? Was it even safe? Among other findings, Dr. Giralt and Dr. Perales’ 100th paper showed that reduced-intensity HSCT remained a viable alternative after a CLL-RT patient progressed on a novel agent.
Impact of the pandemic
When COVID-19 hit, the team lost many research staff and developed a huge backlog, said Dr. Giralt. He and Dr. Perales realized that they needed to be “thoughtful and careful” about which studies to continue. “For example, the CD-34 selection trials we did not close because these are our workhorse trials,” Dr. Giralt said. “We have people we need to treat, and some of the patients that we need to treat can only be treated on trial.”
The team was also able to pivot some of their work into COVID 19 itself, and they collected crucial information on HSCT in recovered COVID-19 patients, as an example.
“We were living through a critical time, but that doesn’t mean we [aren’t] obligated to continue our mission, our research mission,” said Dr. Giralt. “It really is team science. The way we look at it ... there’s a common thread: We both like to do allogeneic transplant, and we both believe in trying to make CD-34 selection better. So we’re both very much [working on] how can we improve what we call ‘the Memorial way’ of doing transplants. Where we separate is, Miguel does primarily lymphoma. He doesn’t do myeloma [like me]. So in those two areas, we’re helping develop the junior faculty in a different way.”
Something more in common
Right from the start, Dr. Perales and Dr. Giralt also shared a commitment to mentoring. Since 2010, Dr. Perales has mentored 22 up-and-coming junior faculty, including 10 from Europe (8 from Spain) and 2 from Latin America.
“[It makes] the research enterprise much more productive but [these young scientists] really increase the visibility of the program,” said Dr. Giralt.
He cited Dr. Perales’ track record of mentoring as one of the reasons for his promotion to chief of the adult bone marrow transplant service. In March 2020, Dr. Perales seamlessly stepped into Dr. Giralt’s shoes, while Dr. Giralt moved on to his present role as deputy division head of the division of hematologic malignancies.
Dr. Perales said: “The key aspect [of these promotions] is the fantastic working relationship that we’ve had over the years. ... I consider Sergio my mentor, but also a good friend and colleague. And so I think it’s this ability that we’ve had to work together and that relationship of trust, which has been key.”
“Sergio is somebody who lifts people up,” Dr. Perales added. “Many people will tell you that Sergio has helped them in their career. ... And I think that’s a lesson I’ve learned from him: training the next generation. And [that’s] not just in the U.S., but outside. I think that’s a key role that we have. And our responsibility.”
Asked to comment on their 100th-paper milestone, Dr. Perales firmly turned the spotlight from himself and Dr. Giralt to the junior investigators who have passed through the doors of the bone-marrow transplant program: “This body of work represents not just our collaboration but also the many contributions of our team at MSK ... and beyond MSK.”
This article was updated 1/26/22.
On July 29, 2021, Sergio Giralt, MD, deputy division head of the division of hematologic malignancies and Miguel-Angel Perales, MD, chief of the adult bone marrow transplant service at MSKCC, published their 100th peer-reviewed paper as coauthors. Listing hundreds of such articles on a CV is standard for top-tier physicians, but the pair had gone one better: 100 publications written together in 10 years.
Their centenary article hit scientific newsstands almost exactly a decade after their first joint paper, which appeared in September 2011, not long after they met.
Born in Cuba, Dr. Giralt grew up in Venezuela. From the age of 14, he knew that medicine was his path, and in 1984 he earned a medical degree from the Universidad Central de Venezuela, Caracas. Next came a research position at Harvard Medical School, a residency at the Good Samaritan Hospital, Cincinnati, and a fellowship at the University of Texas MD Anderson Cancer Center, Houston. Dr. Giralt arrived at MSKCC in 2010 as the new chief of the adult bone marrow transplant service. There he was introduced to a new colleague, Dr. Perales. They soon learned that in addition to expertise in hematology, they had second language in common: Spanish.
Dr. Giralt said: “We both have a Spanish background and in a certain sense, there was an affinity there. ... We both have shared experiences.”
Dr. Perales was brought up in Belgium, a European nation with three official languages: French, Dutch, and German. He speaks five tongues in all and learned Spanish from his father, who came from Spain.
Fluency in Spanish enables both physicians to take care of the many New Yorkers who are more comfortable in that language – especially when navigating cancer treatment. However, both Dr. Giralt and Dr. Perales said that a second language is more than a professional tool. They described the enjoyable change of persona that happens when they switch to Spanish.
“People who are multilingual have different roles [as much as] different languages,” said Dr. Perales. “When I’m in Spanish, part of my brain is [thinking back to] summer vacations and hanging out with my cousins.”
When it comes to clinical science, however, English is the language of choice.
Global leaders in HSCT
Dr. Giralt and Dr. Perales are known worldwide in the field of allogeneic HSCT, a potentially curative treatment for an elongating list of both malignant and nonmalignant diseases.
In 1973, MSKCC conducted the first bone-marrow transplant from an unrelated donor. Fifty years on, medical oncologists in the United States conduct approximately 8,500 allogeneic transplants each year, 72% to treat acute leukemias or myelodysplastic syndrome (MDS).
However, stripping the immune system with intensive chemotherapy ‘conditioning,’ then rebuilding it with non-diseased donor hematopoietic cells is a hazardous undertaking. Older patients are less likely to survive the intensive conditioning, so historically have missed out. Also, even with a good human leukocyte antigen (HLA) match, the recipient needs often brutal immunosuppression.
Since Dr. Giralt and Dr. Perales began their partnership in 2010, the goals of their work have not changed: to develop safer, lower-intensity transplantation suitable for older, more vulnerable patients and reduce fearsome posttransplant sequelae such as graft-versus-host disease (GVHD).
Dr. Giralt’s publication list spans more than 600 peer-reviewed papers, articles and book chapters, almost exclusively on HSCT. Dr. Perales has more than 300 publication credits on the topic.
The two paired up on their first paper just months after Dr. Giralt arrived at MSKCC. That article, published in Biology of Blood and Marrow Transplantation, compared umbilical cord blood for HSCT with donor blood in 367 people with a variety of hematologic malignancies, including acute and chronic leukemias, MDS, and lymphoma.
The MSKCC team found that transplant-related mortality in the first 180 days was higher for the cord blood (21%), but thereafter mortality and relapse were much lower than for donated blood, with the result that 2-year progression-free survival of 55% was similar. Dr. Perales, Dr. Giralt and their coauthors concluded that the data provided “strong support” for further work on cord blood as an alternative stem-cell source.
During their first decade of collaboration, Dr. Giralt and Dr. Perales worked on any promising avenue that could improve outcomes and the experience of HSCT recipients, including reduced-intensity conditioning regimens to allow older adults to benefit from curative HSCT and donor T-cell depletion by CD34 selection, to reduce graft-versus-host disease (GVHD).
The CD34 protein is typically found on the surface of early stage and highly active stem cell types. Selecting these cell types using a range of techniques can eliminate many other potentially interfering or inactive cells. This enriches the transplant population with the most effective cells and can lower the risk of GVHD.
The 100th paper on which Dr. Giralt and Dr. Perales were coauthors was published in Blood Advances on July 27, 2021. The retrospective study examined the fate of 58 MSKCC patients with a rare form of chronic lymphocytic leukemia, CLL with Richter’s transformation (CLL-RT). It was the largest such study to date of this rare disease.
M.D. Anderson Cancer Center had shown in 2006 that, despite chemotherapy, overall survival in patients with CLL-RT was approximately 8 months. HSCT improved survival dramatically (75% at 3 years; n = 7). However, with the advent of novel targeted drugs for CLL such as ibrutinib (Imbruvica), venetoclax (Venclexta), or idelalisib (Zydelig), the MSKCC team asked themselves: What was the role of reduced-intensive conditioning HSCT? Was it even safe? Among other findings, Dr. Giralt and Dr. Perales’ 100th paper showed that reduced-intensity HSCT remained a viable alternative after a CLL-RT patient progressed on a novel agent.
Impact of the pandemic
When COVID-19 hit, the team lost many research staff and developed a huge backlog, said Dr. Giralt. He and Dr. Perales realized that they needed to be “thoughtful and careful” about which studies to continue. “For example, the CD-34 selection trials we did not close because these are our workhorse trials,” Dr. Giralt said. “We have people we need to treat, and some of the patients that we need to treat can only be treated on trial.”
The team was also able to pivot some of their work into COVID 19 itself, and they collected crucial information on HSCT in recovered COVID-19 patients, as an example.
“We were living through a critical time, but that doesn’t mean we [aren’t] obligated to continue our mission, our research mission,” said Dr. Giralt. “It really is team science. The way we look at it ... there’s a common thread: We both like to do allogeneic transplant, and we both believe in trying to make CD-34 selection better. So we’re both very much [working on] how can we improve what we call ‘the Memorial way’ of doing transplants. Where we separate is, Miguel does primarily lymphoma. He doesn’t do myeloma [like me]. So in those two areas, we’re helping develop the junior faculty in a different way.”
Something more in common
Right from the start, Dr. Perales and Dr. Giralt also shared a commitment to mentoring. Since 2010, Dr. Perales has mentored 22 up-and-coming junior faculty, including 10 from Europe (8 from Spain) and 2 from Latin America.
“[It makes] the research enterprise much more productive but [these young scientists] really increase the visibility of the program,” said Dr. Giralt.
He cited Dr. Perales’ track record of mentoring as one of the reasons for his promotion to chief of the adult bone marrow transplant service. In March 2020, Dr. Perales seamlessly stepped into Dr. Giralt’s shoes, while Dr. Giralt moved on to his present role as deputy division head of the division of hematologic malignancies.
Dr. Perales said: “The key aspect [of these promotions] is the fantastic working relationship that we’ve had over the years. ... I consider Sergio my mentor, but also a good friend and colleague. And so I think it’s this ability that we’ve had to work together and that relationship of trust, which has been key.”
“Sergio is somebody who lifts people up,” Dr. Perales added. “Many people will tell you that Sergio has helped them in their career. ... And I think that’s a lesson I’ve learned from him: training the next generation. And [that’s] not just in the U.S., but outside. I think that’s a key role that we have. And our responsibility.”
Asked to comment on their 100th-paper milestone, Dr. Perales firmly turned the spotlight from himself and Dr. Giralt to the junior investigators who have passed through the doors of the bone-marrow transplant program: “This body of work represents not just our collaboration but also the many contributions of our team at MSK ... and beyond MSK.”
This article was updated 1/26/22.
On July 29, 2021, Sergio Giralt, MD, deputy division head of the division of hematologic malignancies and Miguel-Angel Perales, MD, chief of the adult bone marrow transplant service at MSKCC, published their 100th peer-reviewed paper as coauthors. Listing hundreds of such articles on a CV is standard for top-tier physicians, but the pair had gone one better: 100 publications written together in 10 years.
Their centenary article hit scientific newsstands almost exactly a decade after their first joint paper, which appeared in September 2011, not long after they met.
Born in Cuba, Dr. Giralt grew up in Venezuela. From the age of 14, he knew that medicine was his path, and in 1984 he earned a medical degree from the Universidad Central de Venezuela, Caracas. Next came a research position at Harvard Medical School, a residency at the Good Samaritan Hospital, Cincinnati, and a fellowship at the University of Texas MD Anderson Cancer Center, Houston. Dr. Giralt arrived at MSKCC in 2010 as the new chief of the adult bone marrow transplant service. There he was introduced to a new colleague, Dr. Perales. They soon learned that in addition to expertise in hematology, they had second language in common: Spanish.
Dr. Giralt said: “We both have a Spanish background and in a certain sense, there was an affinity there. ... We both have shared experiences.”
Dr. Perales was brought up in Belgium, a European nation with three official languages: French, Dutch, and German. He speaks five tongues in all and learned Spanish from his father, who came from Spain.
Fluency in Spanish enables both physicians to take care of the many New Yorkers who are more comfortable in that language – especially when navigating cancer treatment. However, both Dr. Giralt and Dr. Perales said that a second language is more than a professional tool. They described the enjoyable change of persona that happens when they switch to Spanish.
“People who are multilingual have different roles [as much as] different languages,” said Dr. Perales. “When I’m in Spanish, part of my brain is [thinking back to] summer vacations and hanging out with my cousins.”
When it comes to clinical science, however, English is the language of choice.
Global leaders in HSCT
Dr. Giralt and Dr. Perales are known worldwide in the field of allogeneic HSCT, a potentially curative treatment for an elongating list of both malignant and nonmalignant diseases.
In 1973, MSKCC conducted the first bone-marrow transplant from an unrelated donor. Fifty years on, medical oncologists in the United States conduct approximately 8,500 allogeneic transplants each year, 72% to treat acute leukemias or myelodysplastic syndrome (MDS).
However, stripping the immune system with intensive chemotherapy ‘conditioning,’ then rebuilding it with non-diseased donor hematopoietic cells is a hazardous undertaking. Older patients are less likely to survive the intensive conditioning, so historically have missed out. Also, even with a good human leukocyte antigen (HLA) match, the recipient needs often brutal immunosuppression.
Since Dr. Giralt and Dr. Perales began their partnership in 2010, the goals of their work have not changed: to develop safer, lower-intensity transplantation suitable for older, more vulnerable patients and reduce fearsome posttransplant sequelae such as graft-versus-host disease (GVHD).
Dr. Giralt’s publication list spans more than 600 peer-reviewed papers, articles and book chapters, almost exclusively on HSCT. Dr. Perales has more than 300 publication credits on the topic.
The two paired up on their first paper just months after Dr. Giralt arrived at MSKCC. That article, published in Biology of Blood and Marrow Transplantation, compared umbilical cord blood for HSCT with donor blood in 367 people with a variety of hematologic malignancies, including acute and chronic leukemias, MDS, and lymphoma.
The MSKCC team found that transplant-related mortality in the first 180 days was higher for the cord blood (21%), but thereafter mortality and relapse were much lower than for donated blood, with the result that 2-year progression-free survival of 55% was similar. Dr. Perales, Dr. Giralt and their coauthors concluded that the data provided “strong support” for further work on cord blood as an alternative stem-cell source.
During their first decade of collaboration, Dr. Giralt and Dr. Perales worked on any promising avenue that could improve outcomes and the experience of HSCT recipients, including reduced-intensity conditioning regimens to allow older adults to benefit from curative HSCT and donor T-cell depletion by CD34 selection, to reduce graft-versus-host disease (GVHD).
The CD34 protein is typically found on the surface of early stage and highly active stem cell types. Selecting these cell types using a range of techniques can eliminate many other potentially interfering or inactive cells. This enriches the transplant population with the most effective cells and can lower the risk of GVHD.
The 100th paper on which Dr. Giralt and Dr. Perales were coauthors was published in Blood Advances on July 27, 2021. The retrospective study examined the fate of 58 MSKCC patients with a rare form of chronic lymphocytic leukemia, CLL with Richter’s transformation (CLL-RT). It was the largest such study to date of this rare disease.
M.D. Anderson Cancer Center had shown in 2006 that, despite chemotherapy, overall survival in patients with CLL-RT was approximately 8 months. HSCT improved survival dramatically (75% at 3 years; n = 7). However, with the advent of novel targeted drugs for CLL such as ibrutinib (Imbruvica), venetoclax (Venclexta), or idelalisib (Zydelig), the MSKCC team asked themselves: What was the role of reduced-intensive conditioning HSCT? Was it even safe? Among other findings, Dr. Giralt and Dr. Perales’ 100th paper showed that reduced-intensity HSCT remained a viable alternative after a CLL-RT patient progressed on a novel agent.
Impact of the pandemic
When COVID-19 hit, the team lost many research staff and developed a huge backlog, said Dr. Giralt. He and Dr. Perales realized that they needed to be “thoughtful and careful” about which studies to continue. “For example, the CD-34 selection trials we did not close because these are our workhorse trials,” Dr. Giralt said. “We have people we need to treat, and some of the patients that we need to treat can only be treated on trial.”
The team was also able to pivot some of their work into COVID 19 itself, and they collected crucial information on HSCT in recovered COVID-19 patients, as an example.
“We were living through a critical time, but that doesn’t mean we [aren’t] obligated to continue our mission, our research mission,” said Dr. Giralt. “It really is team science. The way we look at it ... there’s a common thread: We both like to do allogeneic transplant, and we both believe in trying to make CD-34 selection better. So we’re both very much [working on] how can we improve what we call ‘the Memorial way’ of doing transplants. Where we separate is, Miguel does primarily lymphoma. He doesn’t do myeloma [like me]. So in those two areas, we’re helping develop the junior faculty in a different way.”
Something more in common
Right from the start, Dr. Perales and Dr. Giralt also shared a commitment to mentoring. Since 2010, Dr. Perales has mentored 22 up-and-coming junior faculty, including 10 from Europe (8 from Spain) and 2 from Latin America.
“[It makes] the research enterprise much more productive but [these young scientists] really increase the visibility of the program,” said Dr. Giralt.
He cited Dr. Perales’ track record of mentoring as one of the reasons for his promotion to chief of the adult bone marrow transplant service. In March 2020, Dr. Perales seamlessly stepped into Dr. Giralt’s shoes, while Dr. Giralt moved on to his present role as deputy division head of the division of hematologic malignancies.
Dr. Perales said: “The key aspect [of these promotions] is the fantastic working relationship that we’ve had over the years. ... I consider Sergio my mentor, but also a good friend and colleague. And so I think it’s this ability that we’ve had to work together and that relationship of trust, which has been key.”
“Sergio is somebody who lifts people up,” Dr. Perales added. “Many people will tell you that Sergio has helped them in their career. ... And I think that’s a lesson I’ve learned from him: training the next generation. And [that’s] not just in the U.S., but outside. I think that’s a key role that we have. And our responsibility.”
Asked to comment on their 100th-paper milestone, Dr. Perales firmly turned the spotlight from himself and Dr. Giralt to the junior investigators who have passed through the doors of the bone-marrow transplant program: “This body of work represents not just our collaboration but also the many contributions of our team at MSK ... and beyond MSK.”
This article was updated 1/26/22.
Antibiotic choices for inpatients with SSTIs vary by race
– in a national cross-sectional study involving over 1,000 patients in 91 hospitals.
The potential racial disparity in management of SSTI was detected after data were adjusted for penicillin allergy history and for MRSA colonization/infection. The data were also adjusted for hospital day (since admission) in order to control for the administration of more empiric therapy early on.
Clindamycin, a beta-lactam alternative, is not recommended as an SSTI treatment given its frequent dosing requirements and high potential for adverse events including Clostridioides difficile infection (DCI). “Clindamycin is an option but it’s considered inferior. ... It covers MRSA but it shouldn’t be a go-to for skin and soft-tissue infections,” said senior author Kimberly Blumenthal, MD, MSc, assistant professor of medicine at Harvard University, and an allergist, immunologist, and drug allergy and epidemiology researcher at Massachusetts General Hospital, both in Boston.
Cefazolin, on the other hand, does not cover MRSA but is “a guideline-recommended first-line antibiotic for cellulitis SSTI in the hospital,” she said in an interview.
The findings, recently published in JAMA Network Open, offer a valuable portrait of the antibiotics being prescribed in the inpatient setting for SSTIs. Vancomycin, which typically is reserved for MRSA, was the most commonly prescribed antibiotic, regardless of race. Piperacillin-tazobactam, a beta-lactam, was the second most commonly prescribed antibiotic, again regardless of race.
Intravenously administered cefazolin was used in 13% of White inpatients versus 5% of Black inpatients. After controlling for kidney disease, diabetes, and ICU location (in addition to hospital day, penicillin allergy history, and MRSA), White inpatients had an increased likelihood of being prescribed cefazolin (adjusted odds ratio, 2.82; 95% confidence interval, 1.41-5.63) and a decreased likelihood of clindamycin use (aOR, 0.54; 95% CI, 0.30-0.96), compared with Black inpatients.
The investigators utilized the Acute Care Hospital Groups network within Vizient, a member-driven health care performance improvement company, to collect data for the study. Most of the hospitals (91%) that submitted data on adult inpatients with cellulitis or SSTIs (without other infections) were in urban settings and 9% were in rural settings; 60% were community hospitals and 40% were academic medical centers. The researchers accounted for “clustering by hospital” – such as the use of internal guidelines – in their methodology.
Differential management and prescribing practices associated with race and ethnicity have been demonstrated for cardiovascular disease and other chronic problems, but “to see such racial differences play out in acute care is striking,” Utibe R. Essien, MD, MPH, assistant professor of medicine at the University of Pittsburgh and a core investigator with the Center for Health Equity Research and Promotion at the Veterans Affairs Pittsburgh Healthcare System, said in an interview.
“In acute care, we tend to practice pretty similarly across the board ... so the findings give me pause,” said Dr. Essien, an internist and a coauthor of the study, who also works with the University of Pittsburgh’s Center for Pharmaceutical Policy and Prescribing.
Also notable was the prevalence of historical penicillin allergy documented in the dataset: 23% in Black inpatients and 18% in White inpatients with SSTI. It’s a surprisingly high prevalence overall, Dr. Blumenthal said, and the racial difference was surprising because penicillin allergy has been commonly described in the literature as being more common in the White population.
Even though penicillin allergy was controlled for in the study, “given that historical penicillin allergies are associated with increased clindamycin use and risk of CDI, but are often disproved with formal testing, racial disparities in penicillin allergy documentation and assessment require additional study,” she and her coauthors wrote.
Ideally, Dr. Blumenthal said, all inpatients would have access to allergy consultations or testing or some sort of infrastructure for assessing a history of penicillin allergy. At Mass General, allergy consults and challenge doses of beta-lactams (also called graded challenges) are frequently employed.
The study did not collect data on income, educational level, and other structural vulnerability factors. More research is needed to better understand “what’s going on in acute care settings and what the potential drivers of disparities may be,” said Dr. Essien, who co-authored a recent JAMA editorial on “achieving pharmacoequity” to reduce health disparities.
“If guidelines suggest that medication A is the ideal and optimal treatment, we really have to do our best to ensure that every patient, regardless of race or ethnicity, can get that treatment,” he said.
In the study, race was extracted from the medical record and may not have been correctly assigned, the authors noted. “Other race” was not specified in the dataset, and Hispanic ethnicity was not captured. The number of individuals identified as Asian and other races was small, prompting the researchers to focus on antibiotic use in Black and White patients (224 and 854 patients, respectively).
Dr. Blumenthal and Dr. Essien both reported that they had no relevant disclosures. The study was supported with National Institutes of Health grants and the Massachusetts General Hospital department of medicine transformative scholar program.
– in a national cross-sectional study involving over 1,000 patients in 91 hospitals.
The potential racial disparity in management of SSTI was detected after data were adjusted for penicillin allergy history and for MRSA colonization/infection. The data were also adjusted for hospital day (since admission) in order to control for the administration of more empiric therapy early on.
Clindamycin, a beta-lactam alternative, is not recommended as an SSTI treatment given its frequent dosing requirements and high potential for adverse events including Clostridioides difficile infection (DCI). “Clindamycin is an option but it’s considered inferior. ... It covers MRSA but it shouldn’t be a go-to for skin and soft-tissue infections,” said senior author Kimberly Blumenthal, MD, MSc, assistant professor of medicine at Harvard University, and an allergist, immunologist, and drug allergy and epidemiology researcher at Massachusetts General Hospital, both in Boston.
Cefazolin, on the other hand, does not cover MRSA but is “a guideline-recommended first-line antibiotic for cellulitis SSTI in the hospital,” she said in an interview.
The findings, recently published in JAMA Network Open, offer a valuable portrait of the antibiotics being prescribed in the inpatient setting for SSTIs. Vancomycin, which typically is reserved for MRSA, was the most commonly prescribed antibiotic, regardless of race. Piperacillin-tazobactam, a beta-lactam, was the second most commonly prescribed antibiotic, again regardless of race.
Intravenously administered cefazolin was used in 13% of White inpatients versus 5% of Black inpatients. After controlling for kidney disease, diabetes, and ICU location (in addition to hospital day, penicillin allergy history, and MRSA), White inpatients had an increased likelihood of being prescribed cefazolin (adjusted odds ratio, 2.82; 95% confidence interval, 1.41-5.63) and a decreased likelihood of clindamycin use (aOR, 0.54; 95% CI, 0.30-0.96), compared with Black inpatients.
The investigators utilized the Acute Care Hospital Groups network within Vizient, a member-driven health care performance improvement company, to collect data for the study. Most of the hospitals (91%) that submitted data on adult inpatients with cellulitis or SSTIs (without other infections) were in urban settings and 9% were in rural settings; 60% were community hospitals and 40% were academic medical centers. The researchers accounted for “clustering by hospital” – such as the use of internal guidelines – in their methodology.
Differential management and prescribing practices associated with race and ethnicity have been demonstrated for cardiovascular disease and other chronic problems, but “to see such racial differences play out in acute care is striking,” Utibe R. Essien, MD, MPH, assistant professor of medicine at the University of Pittsburgh and a core investigator with the Center for Health Equity Research and Promotion at the Veterans Affairs Pittsburgh Healthcare System, said in an interview.
“In acute care, we tend to practice pretty similarly across the board ... so the findings give me pause,” said Dr. Essien, an internist and a coauthor of the study, who also works with the University of Pittsburgh’s Center for Pharmaceutical Policy and Prescribing.
Also notable was the prevalence of historical penicillin allergy documented in the dataset: 23% in Black inpatients and 18% in White inpatients with SSTI. It’s a surprisingly high prevalence overall, Dr. Blumenthal said, and the racial difference was surprising because penicillin allergy has been commonly described in the literature as being more common in the White population.
Even though penicillin allergy was controlled for in the study, “given that historical penicillin allergies are associated with increased clindamycin use and risk of CDI, but are often disproved with formal testing, racial disparities in penicillin allergy documentation and assessment require additional study,” she and her coauthors wrote.
Ideally, Dr. Blumenthal said, all inpatients would have access to allergy consultations or testing or some sort of infrastructure for assessing a history of penicillin allergy. At Mass General, allergy consults and challenge doses of beta-lactams (also called graded challenges) are frequently employed.
The study did not collect data on income, educational level, and other structural vulnerability factors. More research is needed to better understand “what’s going on in acute care settings and what the potential drivers of disparities may be,” said Dr. Essien, who co-authored a recent JAMA editorial on “achieving pharmacoequity” to reduce health disparities.
“If guidelines suggest that medication A is the ideal and optimal treatment, we really have to do our best to ensure that every patient, regardless of race or ethnicity, can get that treatment,” he said.
In the study, race was extracted from the medical record and may not have been correctly assigned, the authors noted. “Other race” was not specified in the dataset, and Hispanic ethnicity was not captured. The number of individuals identified as Asian and other races was small, prompting the researchers to focus on antibiotic use in Black and White patients (224 and 854 patients, respectively).
Dr. Blumenthal and Dr. Essien both reported that they had no relevant disclosures. The study was supported with National Institutes of Health grants and the Massachusetts General Hospital department of medicine transformative scholar program.
– in a national cross-sectional study involving over 1,000 patients in 91 hospitals.
The potential racial disparity in management of SSTI was detected after data were adjusted for penicillin allergy history and for MRSA colonization/infection. The data were also adjusted for hospital day (since admission) in order to control for the administration of more empiric therapy early on.
Clindamycin, a beta-lactam alternative, is not recommended as an SSTI treatment given its frequent dosing requirements and high potential for adverse events including Clostridioides difficile infection (DCI). “Clindamycin is an option but it’s considered inferior. ... It covers MRSA but it shouldn’t be a go-to for skin and soft-tissue infections,” said senior author Kimberly Blumenthal, MD, MSc, assistant professor of medicine at Harvard University, and an allergist, immunologist, and drug allergy and epidemiology researcher at Massachusetts General Hospital, both in Boston.
Cefazolin, on the other hand, does not cover MRSA but is “a guideline-recommended first-line antibiotic for cellulitis SSTI in the hospital,” she said in an interview.
The findings, recently published in JAMA Network Open, offer a valuable portrait of the antibiotics being prescribed in the inpatient setting for SSTIs. Vancomycin, which typically is reserved for MRSA, was the most commonly prescribed antibiotic, regardless of race. Piperacillin-tazobactam, a beta-lactam, was the second most commonly prescribed antibiotic, again regardless of race.
Intravenously administered cefazolin was used in 13% of White inpatients versus 5% of Black inpatients. After controlling for kidney disease, diabetes, and ICU location (in addition to hospital day, penicillin allergy history, and MRSA), White inpatients had an increased likelihood of being prescribed cefazolin (adjusted odds ratio, 2.82; 95% confidence interval, 1.41-5.63) and a decreased likelihood of clindamycin use (aOR, 0.54; 95% CI, 0.30-0.96), compared with Black inpatients.
The investigators utilized the Acute Care Hospital Groups network within Vizient, a member-driven health care performance improvement company, to collect data for the study. Most of the hospitals (91%) that submitted data on adult inpatients with cellulitis or SSTIs (without other infections) were in urban settings and 9% were in rural settings; 60% were community hospitals and 40% were academic medical centers. The researchers accounted for “clustering by hospital” – such as the use of internal guidelines – in their methodology.
Differential management and prescribing practices associated with race and ethnicity have been demonstrated for cardiovascular disease and other chronic problems, but “to see such racial differences play out in acute care is striking,” Utibe R. Essien, MD, MPH, assistant professor of medicine at the University of Pittsburgh and a core investigator with the Center for Health Equity Research and Promotion at the Veterans Affairs Pittsburgh Healthcare System, said in an interview.
“In acute care, we tend to practice pretty similarly across the board ... so the findings give me pause,” said Dr. Essien, an internist and a coauthor of the study, who also works with the University of Pittsburgh’s Center for Pharmaceutical Policy and Prescribing.
Also notable was the prevalence of historical penicillin allergy documented in the dataset: 23% in Black inpatients and 18% in White inpatients with SSTI. It’s a surprisingly high prevalence overall, Dr. Blumenthal said, and the racial difference was surprising because penicillin allergy has been commonly described in the literature as being more common in the White population.
Even though penicillin allergy was controlled for in the study, “given that historical penicillin allergies are associated with increased clindamycin use and risk of CDI, but are often disproved with formal testing, racial disparities in penicillin allergy documentation and assessment require additional study,” she and her coauthors wrote.
Ideally, Dr. Blumenthal said, all inpatients would have access to allergy consultations or testing or some sort of infrastructure for assessing a history of penicillin allergy. At Mass General, allergy consults and challenge doses of beta-lactams (also called graded challenges) are frequently employed.
The study did not collect data on income, educational level, and other structural vulnerability factors. More research is needed to better understand “what’s going on in acute care settings and what the potential drivers of disparities may be,” said Dr. Essien, who co-authored a recent JAMA editorial on “achieving pharmacoequity” to reduce health disparities.
“If guidelines suggest that medication A is the ideal and optimal treatment, we really have to do our best to ensure that every patient, regardless of race or ethnicity, can get that treatment,” he said.
In the study, race was extracted from the medical record and may not have been correctly assigned, the authors noted. “Other race” was not specified in the dataset, and Hispanic ethnicity was not captured. The number of individuals identified as Asian and other races was small, prompting the researchers to focus on antibiotic use in Black and White patients (224 and 854 patients, respectively).
Dr. Blumenthal and Dr. Essien both reported that they had no relevant disclosures. The study was supported with National Institutes of Health grants and the Massachusetts General Hospital department of medicine transformative scholar program.
FROM JAMA NETWORK OPEN
What’s in a White Coat? The Changing Trends in Physician Attire and What it Means for Dermatology
The White Coat Ceremony is an enduring memory from my medical school years. Amidst the tumult of memories of seemingly endless sleepless nights spent in libraries and cramming for clerkship examinations between surgical cases, I recall a sunny spring day in 2016 where I gathered with my classmates, family, and friends in the medical school campus courtyard. There were several short, mostly forgotten speeches after which proud fathers and mothers, partners, or siblings slipped the all-important white coat onto the shoulders of the physicians-to-be. At that moment, I felt the weight of tradition centuries in the making resting on my shoulders. Of course, the pomp of the ceremony might have felt a tad overblown had I known that the whole thing had fewer years under its belt than the movie Die Hard.
That’s right, the first White Coat Ceremony was held 5 years after the release of that Bruce Willis classic. Dr. Arnold Gold, a pediatric neurologist on faculty at Columbia University, conceived the ceremony in 1993, and it spread rapidly to medical schools—and later nursing schools—across the United States.1 Although the values highlighted by the White Coat Ceremony—humanism and compassion in medicine—are timeless, the ceremony itself is a more modern undertaking. What, then, of the white coat itself? Is it the timeless symbol of doctoring—of medicine—that we all presume it to be? Or is it a symbol of modern marketing, just a trend that caught on? And is it encountering its twilight—as trends often do—in the face of changing fashion and, more fundamentally, in changes to who our physicians are and to their roles in our society?
The Cleanliness of the White Coat
Until the end of the 19th century, physicians in the Western world most frequently dressed in black formal wear. The rationale behind this attire seems to have been twofold. First, society as a whole perceived the physician’s work as a serious and formal matter, and any medical encounter had to reflect the gravity of the occasion. Additionally, physicians’ visits often were a portent of impending demise, as physicians in the era prior to antibiotics and antisepsis frequently had little to offer their patients outside of—at best—anecdotal treatments and—at worst—sheer quackery.2 Black may have seemed a respectful choice for patients who likely faced dire outcomes regardless of the treatment afforded.3
With the turn of the century came a new understanding of the concepts of antisepsis and disease transmission. While Joseph Lister first published on the use of antisepsis in 1867, his practices did not become commonplace until the early 1900s.4 Around the same time came the Flexner report,5 the publication of William Osler’s Principles and Practice of Medicine,6 and the establishment of the modern medical residency, all of which contributed to the shift from the patient’s own bedside and to the hospital as the house of medicine, with cleanliness and antisepsis as part of its core principles.7 The white coat arose as a symbol of purity and freedom from disease. Throughout the 20th century and into the 21st, it has remained the predominant symbol of cleanliness and professionalism for the medical practitioner.
Patient Preference of Physician Attire
Although the white coat may serve as a professional symbol and is well respected medicine, it also plays an important role in the layperson’s perception of their health care providers.8 There is little denying that patients prefer their physicians, almost uniformly, to wear a white coat. A systematic review of physician attire that included 30 studies mainly from North America, Europe, and the United Kingdom found that patient preference for formal attire and white coats is near universal.9 Patients routinely rate physicians wearing a white coat as more intelligent and trustworthy and feel more confident in the care they will receive.10-13 They also freely admit that a physician’s appearance influences their satisfaction with their care.14 The recent adoption of the fleece, or softshell, jacket has not yet pervaded patients’ perceptions of what is considered appropriate physician attire. A 500-respondent survey found that patients were more likely to rate a model wearing a white coat as more professional and experienced compared to the same model wearing a fleece or softshell jacket or other formal attire sans white coat.15
Closer examination of the same data, however, reveals results reproduced with startling consistency across several studies, which suggest those of us adopting other attire need not dig those white coats out of the closet just yet. First, while many studies point to patient preference for white coats, this preference is uniformly strongest in older patients, beginning around age 40 years and becoming an entrenched preference in those older than 65 years.9,14,16-18 On the other hand, younger patient populations display little to no such preference, and some studies indicate that younger patients actually prefer scrubs over formal attire in specific settings such as surgical offices, procedural spaces, or the emergency department.12,14,19 This suggests that bias in favor of traditional physician garb may be more linked to age demographics and may continue to shift as the overall population ages. Additionally, although patients might profess a strong preference for physician attire in theory, it often does not translate into any impact on the patient’s perception of the physician following a clinic visit. The large systematic review on the topic noted that only 25% of studies that surveyed patients about a clinical visit following the encounter reported that physician attire influenced their satisfaction with that visit, suggesting that attire may be less likely to influence patients in the real-world context of receiving care.9 In fact, a prospective study of patient perception of medical staff and interactions found that staff style of dress not only had no bearing on the perception of staff or visit satisfaction but that patients often failed to even accurately recall physician attire when surveyed.20 Another survey study echoed these conclusions, finding that physician attire had no effect on the perception of a proposed treatment plan.21
What do we know about patient perception of physician attire in the dermatology setting specifically, where visits can be unique in their tendency to transition from medical to procedural in the span of a 15-minute encounter depending on the patient’s chief concern? A survey study of dermatology patients at the general, surgical, and wound care dermatology clinics of an academic medical center (Miami, Florida) found that professional attire with a white coat was strongly preferred across a litany of scenarios assessing many aspects of dermatologic care.21 Similarly, a study of patients visiting a single institution’s dermatology and pediatric dermatology clinics surveyed patients and parents regarding attire prior to an appointment and specifically asked if a white coat should be worn.13 Fifty-four percent of the adult patients (n=176) surveyed professed a preference for physicians in white coats, with a stronger preference for white coats reported by those 50 years and older (55%; n=113). Parents or guardians presenting to the pediatric dermatology clinic, on the other hand, favored less formal attire.13 A recent, real-world study performed at an outpatient dermatology clinic examined the influence of changing physician attire on a patient’s perceptions of care received during clinic encounters. They found no substantial difference in patient satisfaction scores before and following the adoption of a new clinic uniform that transitioned from formal attire to fitted scrubs.22
Racial and Gender Bias Affecting Attire Preference
With any study of preference, there is the underlying concern over respondent bias. Many of the studies discussed here have found secondarily that a patient’s implicit bias does not end at the clothes their physician is wearing. The survey study of dermatology patients from the academic medical center in Miami, Florida, found that patients preferred that Black physicians of either sex be garbed in professional attire at all times but generally were more accepting of White physicians in less formal attire.21 Adamson et al23 published a response to the study’s findings urging dermatologists to recognize that a physician’s race and gender influence patients’ perceptions in much the same way that physician attire seems to and encouraged the development of a more diverse dermatologic workforce to help combat this prejudice. The issue of bias is not limited to the specialty of dermatology; the recent survey study by Xun et al15 found that respondents consistently rated female models garbed in physician attire as less professional than male model counterparts. Additionally, female models wearing white coats were mistakenly identified as medical technicians, physician assistants, or nurses with substantially more frequency than males, despite being clothed in the traditional physician garb. Several other publications on the subject have uncovered implicit bias, though it is rarely, if ever, the principle focus of the study.10,24,25 As is unfortunately true in many professions, female physicians and physicians from ethnic minorities face barriers to being perceived as fully competent physicians.
Impact of the COVID-19 Pandemic
Finally, of course, there is the ever-present question of the effect of the pandemic. Although the exact role of the white coat as a fomite for infection—and especially for the spread of viral illness—remains controversial, the perception nonetheless has helped catalyze the movement to alternatives such as short-sleeved white coats, technical jackets, and more recently, fitted scrubs.26-29 As with much in this realm, facts seem less important than perceptions; Zahrina et al30 found that when patients were presented with information regarding the risk for microbial contamination associated with white coats, preference for physicians in professional garb plummeted from 72% to only 22%. To date no articles have examined patient perceptions of the white coat in the context of microbial transmission in the age of COVID-19, but future articles on this topic are likely and may serve to further the demise of the white coat.
Final Thoughts
From my vantage point, it seems the white coat will be claimed by the outgoing tide. During this most recent residency interview season, I do not recall a single medical student wearing a short white coat. The closest I came was a quick glimpse of a crumpled white jacket slung over an arm or stuffed in a shoulder bag. Rotating interns and residents from other services on rotation in our department present in softshell or fleece jackets. Fitted scrubs in the newest trendy colors speckle a previously all-white canvas. I, for one, have not donned my own white coat in at least a year, and perhaps it is all for the best. Physician attire is one small aspect of the practice of medicine and likely bears little, if any, relation to the wearer’s qualifications. Our focus should be on building rapport with our patients, providing high-quality care, reducing the risk for nosocomial infection, and developing a health care system that is fair and equitable for patients and health care workers alike, not on who is wearing what. Perhaps the introduction of new physician attire is a small part of the disruption we need to help address persistent gender and racial biases in our field and help shepherd our patients and colleagues to a worldview that is more open and accepting of physicians of diverse backgrounds.
- White Coat Ceremony. Gold Foundation website. Accessed December 26, 2021. https://www.gold-foundation.org/programs/white-coat-ceremony/
- Shryock RH. The Development of Modern Medicine. University of Pennsylvania Press; 2017.
- Hochberg MS. The doctor’s white coat—an historical perspective. Virtual Mentor. 2007;9:310-314.
- Lister J. On the antiseptic principle in the practice of surgery. Lancet. 1867;90:353-356.
- Flexner A. Medical Education in the United States and Canada: A Report to the Carnegie Foundation for the Advancement of Teaching. Carnegie Foundation for the Advancement of Teaching; 1910.
- Osler W. Principles and Practice of Medicine: Designed for the Use of Practitioners and Students of Medicine. D. Appleton & Company; 1892.
- Blumhagen DW. The doctor’s white coat: the image of the physician in modern America. Ann Intern Med. 1979;91:111-116.
- Verghese BG, Kashinath SK, Jadhav N, et al. Physician attire: physicians’ perspectives on attire in a community hospital setting among non-surgical specialties. J Community Hosp Intern Med Perspect. 2020;10:1-5.
- Petrilli CM, Mack M, Petrilli JJ, et al. Understanding the role of physician attire on patient perceptions: a systematic review of the literature—targeting attire to improve likelihood of rapport (TAILOR) investigators. BMJ Open. 2015;5:E006678.
- Rehman SU, Nietert PJ, Cope DW, et al. What to wear today? effect of doctor’s attire on the trust and confidence of patients. Am J Med. 2005;118:1279-1286.
- Jennings JD, Ciaravino SG, Ramsey FV, et al. Physicians’ attire influences patients’ perceptions in the urban outpatient orthopaedic surgery setting. Clin Orthop Relat Res. 2016;474:1908-1918.
- Gherardi G, Cameron J, West A, et al. Are we dressed to impress? a descriptive survey assessing patients preference of doctors’ attire in the hospital setting. Clin Med (Lond). 2009;9:519-524.
- Thomas MW, Burkhart CN, Lugo-Somolinos A, et al. Patients’ perceptions of physician attire in dermatology clinics. Arch Dermatol. 2011;147:505-506.
- Petrilli CM, Saint S, Jennings JJ, et al. Understanding patient preference for physician attire: a cross-sectional observational study of 10 academic medical centres in the USA. BMJ Open. 2018;8:E021239.
- Xun H, Chen J, Sun AH, et al. Public perceptions of physician attire and professionalism in the US. JAMA Network Open. 2021;4:E2117779.
- Kamata K, Kuriyama A, Chopra V, et al. Patient preferences for physician attire: a multicenter study in Japan [published online February 11, 2020]. J Hosp Med. 2020;15:204-210.
- Budny AM, Rogers LC, Mandracchia VJ, et al. The physician’s attire and its influence on patient confidence. J Am Podiatr Assoc. 2006;96:132-138.
- Lill MM, Wilkinson TJ. Judging a book by its cover: descriptive survey of patients’ preferences for doctors’ appearance and mode of address. Br Med J. 2005;331:1524-1527.
- Hossler EW, Shipp D, Palmer M, et al. Impact of provider attire on patient satisfaction in an outpatient dermatology clinic. Cutis. 2018;102:127-129.
- Boon D, Wardrope J. What should doctors wear in the accident and emergency department? patients’ perception. J Accid Emerg Med. 1994;11:175-177.
- Fox JD, Prado G, Baquerizo Nole KL, et al. Patient preference in dermatologist attire in the medical, surgical, and wound care settings. JAMA Dermatol. 2016;152:913-919.
- Bray JK, Porter C, Feldman SR. The effect of physician appearance on patient perceptions of treatment plans. Dermatol Online J. 2021;27. doi:10.5070/D327553611
- Adamson AS, Wright SW, Pandya AG. A missed opportunity to discuss racial and gender bias in dermatology. JAMA Dermatol. 2017;153:110-111.
- Hartmans C, Heremans S, Lagrain M, et al. The doctor’s new clothes: professional or fashionable? Primary Health Care. 2013;3:135.
- Kurihara H, Maeno T, Maeno T. Importance of physicians’ attire: factors influencing the impression it makes on patients, a cross-sectional study. Asia Pac Fam Med. 2014;13:2.
- Treakle AM, Thom KA, Furuno JP, et al. Bacterial contamination of health care workers’ white coats. Am J Infect Control. 2009;37:101-105.
- Banu A, Anand M, Nagi N, et al. White coats as a vehicle for bacterial dissemination. J Clin Diagn Res. 2012;6:1381-1384.
- Haun N, Hooper-Lane C, Safdar N. Healthcare personnel attire and devices as fomites: a systematic review. Infect Control Hosp Epidemiol. 2016;37:1367-1373.
- Tse G, Withey S, Yeo JM, et al. Bare below the elbows: was the target the white coat? J Hosp Infect. 2015;91:299-301.
- Zahrina AZ, Haymond P, Rosanna P, et al. Does the attire of a primary care physician affect patients’ perceptions and their levels of trust in the doctor? Malays Fam Physician. 2018;13:3-11.
The White Coat Ceremony is an enduring memory from my medical school years. Amidst the tumult of memories of seemingly endless sleepless nights spent in libraries and cramming for clerkship examinations between surgical cases, I recall a sunny spring day in 2016 where I gathered with my classmates, family, and friends in the medical school campus courtyard. There were several short, mostly forgotten speeches after which proud fathers and mothers, partners, or siblings slipped the all-important white coat onto the shoulders of the physicians-to-be. At that moment, I felt the weight of tradition centuries in the making resting on my shoulders. Of course, the pomp of the ceremony might have felt a tad overblown had I known that the whole thing had fewer years under its belt than the movie Die Hard.
That’s right, the first White Coat Ceremony was held 5 years after the release of that Bruce Willis classic. Dr. Arnold Gold, a pediatric neurologist on faculty at Columbia University, conceived the ceremony in 1993, and it spread rapidly to medical schools—and later nursing schools—across the United States.1 Although the values highlighted by the White Coat Ceremony—humanism and compassion in medicine—are timeless, the ceremony itself is a more modern undertaking. What, then, of the white coat itself? Is it the timeless symbol of doctoring—of medicine—that we all presume it to be? Or is it a symbol of modern marketing, just a trend that caught on? And is it encountering its twilight—as trends often do—in the face of changing fashion and, more fundamentally, in changes to who our physicians are and to their roles in our society?
The Cleanliness of the White Coat
Until the end of the 19th century, physicians in the Western world most frequently dressed in black formal wear. The rationale behind this attire seems to have been twofold. First, society as a whole perceived the physician’s work as a serious and formal matter, and any medical encounter had to reflect the gravity of the occasion. Additionally, physicians’ visits often were a portent of impending demise, as physicians in the era prior to antibiotics and antisepsis frequently had little to offer their patients outside of—at best—anecdotal treatments and—at worst—sheer quackery.2 Black may have seemed a respectful choice for patients who likely faced dire outcomes regardless of the treatment afforded.3
With the turn of the century came a new understanding of the concepts of antisepsis and disease transmission. While Joseph Lister first published on the use of antisepsis in 1867, his practices did not become commonplace until the early 1900s.4 Around the same time came the Flexner report,5 the publication of William Osler’s Principles and Practice of Medicine,6 and the establishment of the modern medical residency, all of which contributed to the shift from the patient’s own bedside and to the hospital as the house of medicine, with cleanliness and antisepsis as part of its core principles.7 The white coat arose as a symbol of purity and freedom from disease. Throughout the 20th century and into the 21st, it has remained the predominant symbol of cleanliness and professionalism for the medical practitioner.
Patient Preference of Physician Attire
Although the white coat may serve as a professional symbol and is well respected medicine, it also plays an important role in the layperson’s perception of their health care providers.8 There is little denying that patients prefer their physicians, almost uniformly, to wear a white coat. A systematic review of physician attire that included 30 studies mainly from North America, Europe, and the United Kingdom found that patient preference for formal attire and white coats is near universal.9 Patients routinely rate physicians wearing a white coat as more intelligent and trustworthy and feel more confident in the care they will receive.10-13 They also freely admit that a physician’s appearance influences their satisfaction with their care.14 The recent adoption of the fleece, or softshell, jacket has not yet pervaded patients’ perceptions of what is considered appropriate physician attire. A 500-respondent survey found that patients were more likely to rate a model wearing a white coat as more professional and experienced compared to the same model wearing a fleece or softshell jacket or other formal attire sans white coat.15
Closer examination of the same data, however, reveals results reproduced with startling consistency across several studies, which suggest those of us adopting other attire need not dig those white coats out of the closet just yet. First, while many studies point to patient preference for white coats, this preference is uniformly strongest in older patients, beginning around age 40 years and becoming an entrenched preference in those older than 65 years.9,14,16-18 On the other hand, younger patient populations display little to no such preference, and some studies indicate that younger patients actually prefer scrubs over formal attire in specific settings such as surgical offices, procedural spaces, or the emergency department.12,14,19 This suggests that bias in favor of traditional physician garb may be more linked to age demographics and may continue to shift as the overall population ages. Additionally, although patients might profess a strong preference for physician attire in theory, it often does not translate into any impact on the patient’s perception of the physician following a clinic visit. The large systematic review on the topic noted that only 25% of studies that surveyed patients about a clinical visit following the encounter reported that physician attire influenced their satisfaction with that visit, suggesting that attire may be less likely to influence patients in the real-world context of receiving care.9 In fact, a prospective study of patient perception of medical staff and interactions found that staff style of dress not only had no bearing on the perception of staff or visit satisfaction but that patients often failed to even accurately recall physician attire when surveyed.20 Another survey study echoed these conclusions, finding that physician attire had no effect on the perception of a proposed treatment plan.21
What do we know about patient perception of physician attire in the dermatology setting specifically, where visits can be unique in their tendency to transition from medical to procedural in the span of a 15-minute encounter depending on the patient’s chief concern? A survey study of dermatology patients at the general, surgical, and wound care dermatology clinics of an academic medical center (Miami, Florida) found that professional attire with a white coat was strongly preferred across a litany of scenarios assessing many aspects of dermatologic care.21 Similarly, a study of patients visiting a single institution’s dermatology and pediatric dermatology clinics surveyed patients and parents regarding attire prior to an appointment and specifically asked if a white coat should be worn.13 Fifty-four percent of the adult patients (n=176) surveyed professed a preference for physicians in white coats, with a stronger preference for white coats reported by those 50 years and older (55%; n=113). Parents or guardians presenting to the pediatric dermatology clinic, on the other hand, favored less formal attire.13 A recent, real-world study performed at an outpatient dermatology clinic examined the influence of changing physician attire on a patient’s perceptions of care received during clinic encounters. They found no substantial difference in patient satisfaction scores before and following the adoption of a new clinic uniform that transitioned from formal attire to fitted scrubs.22
Racial and Gender Bias Affecting Attire Preference
With any study of preference, there is the underlying concern over respondent bias. Many of the studies discussed here have found secondarily that a patient’s implicit bias does not end at the clothes their physician is wearing. The survey study of dermatology patients from the academic medical center in Miami, Florida, found that patients preferred that Black physicians of either sex be garbed in professional attire at all times but generally were more accepting of White physicians in less formal attire.21 Adamson et al23 published a response to the study’s findings urging dermatologists to recognize that a physician’s race and gender influence patients’ perceptions in much the same way that physician attire seems to and encouraged the development of a more diverse dermatologic workforce to help combat this prejudice. The issue of bias is not limited to the specialty of dermatology; the recent survey study by Xun et al15 found that respondents consistently rated female models garbed in physician attire as less professional than male model counterparts. Additionally, female models wearing white coats were mistakenly identified as medical technicians, physician assistants, or nurses with substantially more frequency than males, despite being clothed in the traditional physician garb. Several other publications on the subject have uncovered implicit bias, though it is rarely, if ever, the principle focus of the study.10,24,25 As is unfortunately true in many professions, female physicians and physicians from ethnic minorities face barriers to being perceived as fully competent physicians.
Impact of the COVID-19 Pandemic
Finally, of course, there is the ever-present question of the effect of the pandemic. Although the exact role of the white coat as a fomite for infection—and especially for the spread of viral illness—remains controversial, the perception nonetheless has helped catalyze the movement to alternatives such as short-sleeved white coats, technical jackets, and more recently, fitted scrubs.26-29 As with much in this realm, facts seem less important than perceptions; Zahrina et al30 found that when patients were presented with information regarding the risk for microbial contamination associated with white coats, preference for physicians in professional garb plummeted from 72% to only 22%. To date no articles have examined patient perceptions of the white coat in the context of microbial transmission in the age of COVID-19, but future articles on this topic are likely and may serve to further the demise of the white coat.
Final Thoughts
From my vantage point, it seems the white coat will be claimed by the outgoing tide. During this most recent residency interview season, I do not recall a single medical student wearing a short white coat. The closest I came was a quick glimpse of a crumpled white jacket slung over an arm or stuffed in a shoulder bag. Rotating interns and residents from other services on rotation in our department present in softshell or fleece jackets. Fitted scrubs in the newest trendy colors speckle a previously all-white canvas. I, for one, have not donned my own white coat in at least a year, and perhaps it is all for the best. Physician attire is one small aspect of the practice of medicine and likely bears little, if any, relation to the wearer’s qualifications. Our focus should be on building rapport with our patients, providing high-quality care, reducing the risk for nosocomial infection, and developing a health care system that is fair and equitable for patients and health care workers alike, not on who is wearing what. Perhaps the introduction of new physician attire is a small part of the disruption we need to help address persistent gender and racial biases in our field and help shepherd our patients and colleagues to a worldview that is more open and accepting of physicians of diverse backgrounds.
The White Coat Ceremony is an enduring memory from my medical school years. Amidst the tumult of memories of seemingly endless sleepless nights spent in libraries and cramming for clerkship examinations between surgical cases, I recall a sunny spring day in 2016 where I gathered with my classmates, family, and friends in the medical school campus courtyard. There were several short, mostly forgotten speeches after which proud fathers and mothers, partners, or siblings slipped the all-important white coat onto the shoulders of the physicians-to-be. At that moment, I felt the weight of tradition centuries in the making resting on my shoulders. Of course, the pomp of the ceremony might have felt a tad overblown had I known that the whole thing had fewer years under its belt than the movie Die Hard.
That’s right, the first White Coat Ceremony was held 5 years after the release of that Bruce Willis classic. Dr. Arnold Gold, a pediatric neurologist on faculty at Columbia University, conceived the ceremony in 1993, and it spread rapidly to medical schools—and later nursing schools—across the United States.1 Although the values highlighted by the White Coat Ceremony—humanism and compassion in medicine—are timeless, the ceremony itself is a more modern undertaking. What, then, of the white coat itself? Is it the timeless symbol of doctoring—of medicine—that we all presume it to be? Or is it a symbol of modern marketing, just a trend that caught on? And is it encountering its twilight—as trends often do—in the face of changing fashion and, more fundamentally, in changes to who our physicians are and to their roles in our society?
The Cleanliness of the White Coat
Until the end of the 19th century, physicians in the Western world most frequently dressed in black formal wear. The rationale behind this attire seems to have been twofold. First, society as a whole perceived the physician’s work as a serious and formal matter, and any medical encounter had to reflect the gravity of the occasion. Additionally, physicians’ visits often were a portent of impending demise, as physicians in the era prior to antibiotics and antisepsis frequently had little to offer their patients outside of—at best—anecdotal treatments and—at worst—sheer quackery.2 Black may have seemed a respectful choice for patients who likely faced dire outcomes regardless of the treatment afforded.3
With the turn of the century came a new understanding of the concepts of antisepsis and disease transmission. While Joseph Lister first published on the use of antisepsis in 1867, his practices did not become commonplace until the early 1900s.4 Around the same time came the Flexner report,5 the publication of William Osler’s Principles and Practice of Medicine,6 and the establishment of the modern medical residency, all of which contributed to the shift from the patient’s own bedside and to the hospital as the house of medicine, with cleanliness and antisepsis as part of its core principles.7 The white coat arose as a symbol of purity and freedom from disease. Throughout the 20th century and into the 21st, it has remained the predominant symbol of cleanliness and professionalism for the medical practitioner.
Patient Preference of Physician Attire
Although the white coat may serve as a professional symbol and is well respected medicine, it also plays an important role in the layperson’s perception of their health care providers.8 There is little denying that patients prefer their physicians, almost uniformly, to wear a white coat. A systematic review of physician attire that included 30 studies mainly from North America, Europe, and the United Kingdom found that patient preference for formal attire and white coats is near universal.9 Patients routinely rate physicians wearing a white coat as more intelligent and trustworthy and feel more confident in the care they will receive.10-13 They also freely admit that a physician’s appearance influences their satisfaction with their care.14 The recent adoption of the fleece, or softshell, jacket has not yet pervaded patients’ perceptions of what is considered appropriate physician attire. A 500-respondent survey found that patients were more likely to rate a model wearing a white coat as more professional and experienced compared to the same model wearing a fleece or softshell jacket or other formal attire sans white coat.15
Closer examination of the same data, however, reveals results reproduced with startling consistency across several studies, which suggest those of us adopting other attire need not dig those white coats out of the closet just yet. First, while many studies point to patient preference for white coats, this preference is uniformly strongest in older patients, beginning around age 40 years and becoming an entrenched preference in those older than 65 years.9,14,16-18 On the other hand, younger patient populations display little to no such preference, and some studies indicate that younger patients actually prefer scrubs over formal attire in specific settings such as surgical offices, procedural spaces, or the emergency department.12,14,19 This suggests that bias in favor of traditional physician garb may be more linked to age demographics and may continue to shift as the overall population ages. Additionally, although patients might profess a strong preference for physician attire in theory, it often does not translate into any impact on the patient’s perception of the physician following a clinic visit. The large systematic review on the topic noted that only 25% of studies that surveyed patients about a clinical visit following the encounter reported that physician attire influenced their satisfaction with that visit, suggesting that attire may be less likely to influence patients in the real-world context of receiving care.9 In fact, a prospective study of patient perception of medical staff and interactions found that staff style of dress not only had no bearing on the perception of staff or visit satisfaction but that patients often failed to even accurately recall physician attire when surveyed.20 Another survey study echoed these conclusions, finding that physician attire had no effect on the perception of a proposed treatment plan.21
What do we know about patient perception of physician attire in the dermatology setting specifically, where visits can be unique in their tendency to transition from medical to procedural in the span of a 15-minute encounter depending on the patient’s chief concern? A survey study of dermatology patients at the general, surgical, and wound care dermatology clinics of an academic medical center (Miami, Florida) found that professional attire with a white coat was strongly preferred across a litany of scenarios assessing many aspects of dermatologic care.21 Similarly, a study of patients visiting a single institution’s dermatology and pediatric dermatology clinics surveyed patients and parents regarding attire prior to an appointment and specifically asked if a white coat should be worn.13 Fifty-four percent of the adult patients (n=176) surveyed professed a preference for physicians in white coats, with a stronger preference for white coats reported by those 50 years and older (55%; n=113). Parents or guardians presenting to the pediatric dermatology clinic, on the other hand, favored less formal attire.13 A recent, real-world study performed at an outpatient dermatology clinic examined the influence of changing physician attire on a patient’s perceptions of care received during clinic encounters. They found no substantial difference in patient satisfaction scores before and following the adoption of a new clinic uniform that transitioned from formal attire to fitted scrubs.22
Racial and Gender Bias Affecting Attire Preference
With any study of preference, there is the underlying concern over respondent bias. Many of the studies discussed here have found secondarily that a patient’s implicit bias does not end at the clothes their physician is wearing. The survey study of dermatology patients from the academic medical center in Miami, Florida, found that patients preferred that Black physicians of either sex be garbed in professional attire at all times but generally were more accepting of White physicians in less formal attire.21 Adamson et al23 published a response to the study’s findings urging dermatologists to recognize that a physician’s race and gender influence patients’ perceptions in much the same way that physician attire seems to and encouraged the development of a more diverse dermatologic workforce to help combat this prejudice. The issue of bias is not limited to the specialty of dermatology; the recent survey study by Xun et al15 found that respondents consistently rated female models garbed in physician attire as less professional than male model counterparts. Additionally, female models wearing white coats were mistakenly identified as medical technicians, physician assistants, or nurses with substantially more frequency than males, despite being clothed in the traditional physician garb. Several other publications on the subject have uncovered implicit bias, though it is rarely, if ever, the principle focus of the study.10,24,25 As is unfortunately true in many professions, female physicians and physicians from ethnic minorities face barriers to being perceived as fully competent physicians.
Impact of the COVID-19 Pandemic
Finally, of course, there is the ever-present question of the effect of the pandemic. Although the exact role of the white coat as a fomite for infection—and especially for the spread of viral illness—remains controversial, the perception nonetheless has helped catalyze the movement to alternatives such as short-sleeved white coats, technical jackets, and more recently, fitted scrubs.26-29 As with much in this realm, facts seem less important than perceptions; Zahrina et al30 found that when patients were presented with information regarding the risk for microbial contamination associated with white coats, preference for physicians in professional garb plummeted from 72% to only 22%. To date no articles have examined patient perceptions of the white coat in the context of microbial transmission in the age of COVID-19, but future articles on this topic are likely and may serve to further the demise of the white coat.
Final Thoughts
From my vantage point, it seems the white coat will be claimed by the outgoing tide. During this most recent residency interview season, I do not recall a single medical student wearing a short white coat. The closest I came was a quick glimpse of a crumpled white jacket slung over an arm or stuffed in a shoulder bag. Rotating interns and residents from other services on rotation in our department present in softshell or fleece jackets. Fitted scrubs in the newest trendy colors speckle a previously all-white canvas. I, for one, have not donned my own white coat in at least a year, and perhaps it is all for the best. Physician attire is one small aspect of the practice of medicine and likely bears little, if any, relation to the wearer’s qualifications. Our focus should be on building rapport with our patients, providing high-quality care, reducing the risk for nosocomial infection, and developing a health care system that is fair and equitable for patients and health care workers alike, not on who is wearing what. Perhaps the introduction of new physician attire is a small part of the disruption we need to help address persistent gender and racial biases in our field and help shepherd our patients and colleagues to a worldview that is more open and accepting of physicians of diverse backgrounds.
- White Coat Ceremony. Gold Foundation website. Accessed December 26, 2021. https://www.gold-foundation.org/programs/white-coat-ceremony/
- Shryock RH. The Development of Modern Medicine. University of Pennsylvania Press; 2017.
- Hochberg MS. The doctor’s white coat—an historical perspective. Virtual Mentor. 2007;9:310-314.
- Lister J. On the antiseptic principle in the practice of surgery. Lancet. 1867;90:353-356.
- Flexner A. Medical Education in the United States and Canada: A Report to the Carnegie Foundation for the Advancement of Teaching. Carnegie Foundation for the Advancement of Teaching; 1910.
- Osler W. Principles and Practice of Medicine: Designed for the Use of Practitioners and Students of Medicine. D. Appleton & Company; 1892.
- Blumhagen DW. The doctor’s white coat: the image of the physician in modern America. Ann Intern Med. 1979;91:111-116.
- Verghese BG, Kashinath SK, Jadhav N, et al. Physician attire: physicians’ perspectives on attire in a community hospital setting among non-surgical specialties. J Community Hosp Intern Med Perspect. 2020;10:1-5.
- Petrilli CM, Mack M, Petrilli JJ, et al. Understanding the role of physician attire on patient perceptions: a systematic review of the literature—targeting attire to improve likelihood of rapport (TAILOR) investigators. BMJ Open. 2015;5:E006678.
- Rehman SU, Nietert PJ, Cope DW, et al. What to wear today? effect of doctor’s attire on the trust and confidence of patients. Am J Med. 2005;118:1279-1286.
- Jennings JD, Ciaravino SG, Ramsey FV, et al. Physicians’ attire influences patients’ perceptions in the urban outpatient orthopaedic surgery setting. Clin Orthop Relat Res. 2016;474:1908-1918.
- Gherardi G, Cameron J, West A, et al. Are we dressed to impress? a descriptive survey assessing patients preference of doctors’ attire in the hospital setting. Clin Med (Lond). 2009;9:519-524.
- Thomas MW, Burkhart CN, Lugo-Somolinos A, et al. Patients’ perceptions of physician attire in dermatology clinics. Arch Dermatol. 2011;147:505-506.
- Petrilli CM, Saint S, Jennings JJ, et al. Understanding patient preference for physician attire: a cross-sectional observational study of 10 academic medical centres in the USA. BMJ Open. 2018;8:E021239.
- Xun H, Chen J, Sun AH, et al. Public perceptions of physician attire and professionalism in the US. JAMA Network Open. 2021;4:E2117779.
- Kamata K, Kuriyama A, Chopra V, et al. Patient preferences for physician attire: a multicenter study in Japan [published online February 11, 2020]. J Hosp Med. 2020;15:204-210.
- Budny AM, Rogers LC, Mandracchia VJ, et al. The physician’s attire and its influence on patient confidence. J Am Podiatr Assoc. 2006;96:132-138.
- Lill MM, Wilkinson TJ. Judging a book by its cover: descriptive survey of patients’ preferences for doctors’ appearance and mode of address. Br Med J. 2005;331:1524-1527.
- Hossler EW, Shipp D, Palmer M, et al. Impact of provider attire on patient satisfaction in an outpatient dermatology clinic. Cutis. 2018;102:127-129.
- Boon D, Wardrope J. What should doctors wear in the accident and emergency department? patients’ perception. J Accid Emerg Med. 1994;11:175-177.
- Fox JD, Prado G, Baquerizo Nole KL, et al. Patient preference in dermatologist attire in the medical, surgical, and wound care settings. JAMA Dermatol. 2016;152:913-919.
- Bray JK, Porter C, Feldman SR. The effect of physician appearance on patient perceptions of treatment plans. Dermatol Online J. 2021;27. doi:10.5070/D327553611
- Adamson AS, Wright SW, Pandya AG. A missed opportunity to discuss racial and gender bias in dermatology. JAMA Dermatol. 2017;153:110-111.
- Hartmans C, Heremans S, Lagrain M, et al. The doctor’s new clothes: professional or fashionable? Primary Health Care. 2013;3:135.
- Kurihara H, Maeno T, Maeno T. Importance of physicians’ attire: factors influencing the impression it makes on patients, a cross-sectional study. Asia Pac Fam Med. 2014;13:2.
- Treakle AM, Thom KA, Furuno JP, et al. Bacterial contamination of health care workers’ white coats. Am J Infect Control. 2009;37:101-105.
- Banu A, Anand M, Nagi N, et al. White coats as a vehicle for bacterial dissemination. J Clin Diagn Res. 2012;6:1381-1384.
- Haun N, Hooper-Lane C, Safdar N. Healthcare personnel attire and devices as fomites: a systematic review. Infect Control Hosp Epidemiol. 2016;37:1367-1373.
- Tse G, Withey S, Yeo JM, et al. Bare below the elbows: was the target the white coat? J Hosp Infect. 2015;91:299-301.
- Zahrina AZ, Haymond P, Rosanna P, et al. Does the attire of a primary care physician affect patients’ perceptions and their levels of trust in the doctor? Malays Fam Physician. 2018;13:3-11.
- White Coat Ceremony. Gold Foundation website. Accessed December 26, 2021. https://www.gold-foundation.org/programs/white-coat-ceremony/
- Shryock RH. The Development of Modern Medicine. University of Pennsylvania Press; 2017.
- Hochberg MS. The doctor’s white coat—an historical perspective. Virtual Mentor. 2007;9:310-314.
- Lister J. On the antiseptic principle in the practice of surgery. Lancet. 1867;90:353-356.
- Flexner A. Medical Education in the United States and Canada: A Report to the Carnegie Foundation for the Advancement of Teaching. Carnegie Foundation for the Advancement of Teaching; 1910.
- Osler W. Principles and Practice of Medicine: Designed for the Use of Practitioners and Students of Medicine. D. Appleton & Company; 1892.
- Blumhagen DW. The doctor’s white coat: the image of the physician in modern America. Ann Intern Med. 1979;91:111-116.
- Verghese BG, Kashinath SK, Jadhav N, et al. Physician attire: physicians’ perspectives on attire in a community hospital setting among non-surgical specialties. J Community Hosp Intern Med Perspect. 2020;10:1-5.
- Petrilli CM, Mack M, Petrilli JJ, et al. Understanding the role of physician attire on patient perceptions: a systematic review of the literature—targeting attire to improve likelihood of rapport (TAILOR) investigators. BMJ Open. 2015;5:E006678.
- Rehman SU, Nietert PJ, Cope DW, et al. What to wear today? effect of doctor’s attire on the trust and confidence of patients. Am J Med. 2005;118:1279-1286.
- Jennings JD, Ciaravino SG, Ramsey FV, et al. Physicians’ attire influences patients’ perceptions in the urban outpatient orthopaedic surgery setting. Clin Orthop Relat Res. 2016;474:1908-1918.
- Gherardi G, Cameron J, West A, et al. Are we dressed to impress? a descriptive survey assessing patients preference of doctors’ attire in the hospital setting. Clin Med (Lond). 2009;9:519-524.
- Thomas MW, Burkhart CN, Lugo-Somolinos A, et al. Patients’ perceptions of physician attire in dermatology clinics. Arch Dermatol. 2011;147:505-506.
- Petrilli CM, Saint S, Jennings JJ, et al. Understanding patient preference for physician attire: a cross-sectional observational study of 10 academic medical centres in the USA. BMJ Open. 2018;8:E021239.
- Xun H, Chen J, Sun AH, et al. Public perceptions of physician attire and professionalism in the US. JAMA Network Open. 2021;4:E2117779.
- Kamata K, Kuriyama A, Chopra V, et al. Patient preferences for physician attire: a multicenter study in Japan [published online February 11, 2020]. J Hosp Med. 2020;15:204-210.
- Budny AM, Rogers LC, Mandracchia VJ, et al. The physician’s attire and its influence on patient confidence. J Am Podiatr Assoc. 2006;96:132-138.
- Lill MM, Wilkinson TJ. Judging a book by its cover: descriptive survey of patients’ preferences for doctors’ appearance and mode of address. Br Med J. 2005;331:1524-1527.
- Hossler EW, Shipp D, Palmer M, et al. Impact of provider attire on patient satisfaction in an outpatient dermatology clinic. Cutis. 2018;102:127-129.
- Boon D, Wardrope J. What should doctors wear in the accident and emergency department? patients’ perception. J Accid Emerg Med. 1994;11:175-177.
- Fox JD, Prado G, Baquerizo Nole KL, et al. Patient preference in dermatologist attire in the medical, surgical, and wound care settings. JAMA Dermatol. 2016;152:913-919.
- Bray JK, Porter C, Feldman SR. The effect of physician appearance on patient perceptions of treatment plans. Dermatol Online J. 2021;27. doi:10.5070/D327553611
- Adamson AS, Wright SW, Pandya AG. A missed opportunity to discuss racial and gender bias in dermatology. JAMA Dermatol. 2017;153:110-111.
- Hartmans C, Heremans S, Lagrain M, et al. The doctor’s new clothes: professional or fashionable? Primary Health Care. 2013;3:135.
- Kurihara H, Maeno T, Maeno T. Importance of physicians’ attire: factors influencing the impression it makes on patients, a cross-sectional study. Asia Pac Fam Med. 2014;13:2.
- Treakle AM, Thom KA, Furuno JP, et al. Bacterial contamination of health care workers’ white coats. Am J Infect Control. 2009;37:101-105.
- Banu A, Anand M, Nagi N, et al. White coats as a vehicle for bacterial dissemination. J Clin Diagn Res. 2012;6:1381-1384.
- Haun N, Hooper-Lane C, Safdar N. Healthcare personnel attire and devices as fomites: a systematic review. Infect Control Hosp Epidemiol. 2016;37:1367-1373.
- Tse G, Withey S, Yeo JM, et al. Bare below the elbows: was the target the white coat? J Hosp Infect. 2015;91:299-301.
- Zahrina AZ, Haymond P, Rosanna P, et al. Does the attire of a primary care physician affect patients’ perceptions and their levels of trust in the doctor? Malays Fam Physician. 2018;13:3-11.
Resident Pearls
- Until the end of the 19th century, Western physicians most commonly wore black formal wear. The rise of the physician’s white coat occurred in conjunction with the shift to hospital medicine.
- Patient surveys repeatedly have demonstrated a preference for physicians to wear white coats; whether or not this has any bearing on patient satisfaction in real-world scenarios is less clear.
- The impact of the COVID-19 pandemic on trends in white coat wear has not yet been elucidated.
Hypertension protocols curb racial bias in therapeutic inertia
Therapeutic inertia regarding intensification of blood pressure treatment has been shown to be more of an issue in Black patients, but this was not the case in the SPRINT trial, which involved a strict standardized approach to blood pressure management, a new analysis shows.
“Overall, we found that therapeutic inertia was similar in different races in the SPRINT trial. We did not see disparities that have been reported in previous observational studies,” lead author, Alexander Zheutlin, MD, University of Utah School of Medicine, Salt Lake City, told this news organization.
“These results show that a well-resourced approach in which a standardized approach to blood pressure measurement and treatment intensification is followed can overcome the racial disparity that is seen in therapeutic inertia and the treatment of blood pressure,” he added.
The study was published online in JAMA Network Open on Jan. 10.
The authors explain that hypertension remains a leading modifiable cause of racial disparities in cardiovascular disease. Despite similar treatment rates and increased availability of safe, effective, and affordable antihypertensive medications, blood pressure control rates among Black and Hispanic adults remain significantly lower than among White adults in the United States, and one of the factors contributing to this is thought to be therapeutic inertia – the phenomenon of clinicians not initiating or up-titrating clinically indicated therapy in the setting of unmet treatment goals.
The current analysis of the SPRINT trial was conducted to investigate whether racial and ethnic differences in therapeutic inertia in hypertension were present when blood pressure care was standardized and protocolized.
The landmark SPRINT trial compared intensive (<120 mm Hg) with standard (<140 mm Hg) systolic blood pressure treatment goals in adults 50 years and older at high risk for cardiovascular disease. The present analysis was restricted to participant visits with measured blood pressure above the target goal and included 4,141 patients in the standard group and 4,415 patients in the intensive group.
Results showed that the overall prevalence of therapeutic inertia – defined as no antihypertensive medication intensification at each study visit where the blood pressure was above target goal – was either similar or lower for Black and Hispanic participants than for White participants. This pattern was observed whether participants were randomized to the standard or intensive treatment group.
“These findings support the idea that a standardized approach to blood pressure management, as implemented in SPRINT, may help ensure equitable care is provided to all patients and could reduce the contribution of therapeutic inertia to disparities in uncontrolled blood pressure,” the authors say.
They point out that therapeutic inertia has been identified as a key clinician-level barrier to blood pressure control and is estimated to be present in more than 80% of clinic visits in community practice, whereas in the current analysis of the SPRINT trial, therapeutic inertia was present in 50% to 60% of participant visits with uncontrolled blood pressure.
“In SPRINT, blood pressure had to be measured at defined intervals with a specific method, and there were clear instructions on intensifying treatment if blood pressure was above a certain goal,” Dr. Zheutlin noted. “Our results show that within such strict confines, therapeutic inertia does not seem to be different between different racial groups. This suggests that we could make better gains in blood pressure control and more equitable treatment if we adopted a standardized approach to hypertension management.”
He added: “Many guidelines have been published on when to start treatment and the targets for blood pressure, but there is a lot of variation in how we turn these guidelines into protocols. We need to bring in more consistent protocols on blood pressure measurement and intensification and ensure they are followed. In practice, if the BP is 5 or 10 mm Hg above target, a clinician may defer a decision to intensify treatment and intensification never gets done. But if there was a strict protocol to follow, there would be less chance of this happening.”
Therapeutic inertia still high
In an accompanying commentary, Matthew Rivara, MD, Nisha Bansal, MD, and Bessie Young, MD, University of Washington, Seattle, say the current SPRINT analysis has broad implications for reducing racial and ethnic disparities in achievement of evidence-based treatment targets in the general population.
“In hypertension management, standardized protocols for medication adjustments may limit clinician practice heterogeneity to ultimately reduce differences in blood pressure control among racial and ethnic minority populations,” they write. But they add that such protocols must be implemented thoughtfully to incorporate individualized clinical assessment and clinician-patient shared decision-making.
Dr. Rivara et al. point out that the rates of therapeutic inertia in SPRINT, while lower than community-based estimates, were still very high. They suggest reasons for this could include clinician concerns about medication efficacy, adverse effects, and patient mistrust of medical professionals. Outside the clinical trial environment, additional considerations may include prescription drug and laboratory test costs, pharmacy access, and competing demands during busy clinic visits.
To address these challenges, they say that clinicians need education on current clinical practice guidelines, managing complications of intensified antihypertensive therapies, and shared decisionmaking, including culturally sensitive collaborative care. Similarly, care systems must support patients on how to address concerns about treatments.
Finally, further research is needed to better define the specific factors associated with therapeutic inertia to allow tailored interventions to overcome this inertia.
“In designing and performing such research, it is vital that investigators engage with racial and ethnic minority groups to better explore the intersection of race, ethnicity, therapeutic decision-making, trust, and shared decisionmaking,” they add.
The SPRINT trial was funded with federal funds from the National Institutes of Health. Dr. Zheutlin reported receiving grants from the NIH during the conduct of the study.
A version of this article first appeared on Medscape.com.
Therapeutic inertia regarding intensification of blood pressure treatment has been shown to be more of an issue in Black patients, but this was not the case in the SPRINT trial, which involved a strict standardized approach to blood pressure management, a new analysis shows.
“Overall, we found that therapeutic inertia was similar in different races in the SPRINT trial. We did not see disparities that have been reported in previous observational studies,” lead author, Alexander Zheutlin, MD, University of Utah School of Medicine, Salt Lake City, told this news organization.
“These results show that a well-resourced approach in which a standardized approach to blood pressure measurement and treatment intensification is followed can overcome the racial disparity that is seen in therapeutic inertia and the treatment of blood pressure,” he added.
The study was published online in JAMA Network Open on Jan. 10.
The authors explain that hypertension remains a leading modifiable cause of racial disparities in cardiovascular disease. Despite similar treatment rates and increased availability of safe, effective, and affordable antihypertensive medications, blood pressure control rates among Black and Hispanic adults remain significantly lower than among White adults in the United States, and one of the factors contributing to this is thought to be therapeutic inertia – the phenomenon of clinicians not initiating or up-titrating clinically indicated therapy in the setting of unmet treatment goals.
The current analysis of the SPRINT trial was conducted to investigate whether racial and ethnic differences in therapeutic inertia in hypertension were present when blood pressure care was standardized and protocolized.
The landmark SPRINT trial compared intensive (<120 mm Hg) with standard (<140 mm Hg) systolic blood pressure treatment goals in adults 50 years and older at high risk for cardiovascular disease. The present analysis was restricted to participant visits with measured blood pressure above the target goal and included 4,141 patients in the standard group and 4,415 patients in the intensive group.
Results showed that the overall prevalence of therapeutic inertia – defined as no antihypertensive medication intensification at each study visit where the blood pressure was above target goal – was either similar or lower for Black and Hispanic participants than for White participants. This pattern was observed whether participants were randomized to the standard or intensive treatment group.
“These findings support the idea that a standardized approach to blood pressure management, as implemented in SPRINT, may help ensure equitable care is provided to all patients and could reduce the contribution of therapeutic inertia to disparities in uncontrolled blood pressure,” the authors say.
They point out that therapeutic inertia has been identified as a key clinician-level barrier to blood pressure control and is estimated to be present in more than 80% of clinic visits in community practice, whereas in the current analysis of the SPRINT trial, therapeutic inertia was present in 50% to 60% of participant visits with uncontrolled blood pressure.
“In SPRINT, blood pressure had to be measured at defined intervals with a specific method, and there were clear instructions on intensifying treatment if blood pressure was above a certain goal,” Dr. Zheutlin noted. “Our results show that within such strict confines, therapeutic inertia does not seem to be different between different racial groups. This suggests that we could make better gains in blood pressure control and more equitable treatment if we adopted a standardized approach to hypertension management.”
He added: “Many guidelines have been published on when to start treatment and the targets for blood pressure, but there is a lot of variation in how we turn these guidelines into protocols. We need to bring in more consistent protocols on blood pressure measurement and intensification and ensure they are followed. In practice, if the BP is 5 or 10 mm Hg above target, a clinician may defer a decision to intensify treatment and intensification never gets done. But if there was a strict protocol to follow, there would be less chance of this happening.”
Therapeutic inertia still high
In an accompanying commentary, Matthew Rivara, MD, Nisha Bansal, MD, and Bessie Young, MD, University of Washington, Seattle, say the current SPRINT analysis has broad implications for reducing racial and ethnic disparities in achievement of evidence-based treatment targets in the general population.
“In hypertension management, standardized protocols for medication adjustments may limit clinician practice heterogeneity to ultimately reduce differences in blood pressure control among racial and ethnic minority populations,” they write. But they add that such protocols must be implemented thoughtfully to incorporate individualized clinical assessment and clinician-patient shared decision-making.
Dr. Rivara et al. point out that the rates of therapeutic inertia in SPRINT, while lower than community-based estimates, were still very high. They suggest reasons for this could include clinician concerns about medication efficacy, adverse effects, and patient mistrust of medical professionals. Outside the clinical trial environment, additional considerations may include prescription drug and laboratory test costs, pharmacy access, and competing demands during busy clinic visits.
To address these challenges, they say that clinicians need education on current clinical practice guidelines, managing complications of intensified antihypertensive therapies, and shared decisionmaking, including culturally sensitive collaborative care. Similarly, care systems must support patients on how to address concerns about treatments.
Finally, further research is needed to better define the specific factors associated with therapeutic inertia to allow tailored interventions to overcome this inertia.
“In designing and performing such research, it is vital that investigators engage with racial and ethnic minority groups to better explore the intersection of race, ethnicity, therapeutic decision-making, trust, and shared decisionmaking,” they add.
The SPRINT trial was funded with federal funds from the National Institutes of Health. Dr. Zheutlin reported receiving grants from the NIH during the conduct of the study.
A version of this article first appeared on Medscape.com.
Therapeutic inertia regarding intensification of blood pressure treatment has been shown to be more of an issue in Black patients, but this was not the case in the SPRINT trial, which involved a strict standardized approach to blood pressure management, a new analysis shows.
“Overall, we found that therapeutic inertia was similar in different races in the SPRINT trial. We did not see disparities that have been reported in previous observational studies,” lead author, Alexander Zheutlin, MD, University of Utah School of Medicine, Salt Lake City, told this news organization.
“These results show that a well-resourced approach in which a standardized approach to blood pressure measurement and treatment intensification is followed can overcome the racial disparity that is seen in therapeutic inertia and the treatment of blood pressure,” he added.
The study was published online in JAMA Network Open on Jan. 10.
The authors explain that hypertension remains a leading modifiable cause of racial disparities in cardiovascular disease. Despite similar treatment rates and increased availability of safe, effective, and affordable antihypertensive medications, blood pressure control rates among Black and Hispanic adults remain significantly lower than among White adults in the United States, and one of the factors contributing to this is thought to be therapeutic inertia – the phenomenon of clinicians not initiating or up-titrating clinically indicated therapy in the setting of unmet treatment goals.
The current analysis of the SPRINT trial was conducted to investigate whether racial and ethnic differences in therapeutic inertia in hypertension were present when blood pressure care was standardized and protocolized.
The landmark SPRINT trial compared intensive (<120 mm Hg) with standard (<140 mm Hg) systolic blood pressure treatment goals in adults 50 years and older at high risk for cardiovascular disease. The present analysis was restricted to participant visits with measured blood pressure above the target goal and included 4,141 patients in the standard group and 4,415 patients in the intensive group.
Results showed that the overall prevalence of therapeutic inertia – defined as no antihypertensive medication intensification at each study visit where the blood pressure was above target goal – was either similar or lower for Black and Hispanic participants than for White participants. This pattern was observed whether participants were randomized to the standard or intensive treatment group.
“These findings support the idea that a standardized approach to blood pressure management, as implemented in SPRINT, may help ensure equitable care is provided to all patients and could reduce the contribution of therapeutic inertia to disparities in uncontrolled blood pressure,” the authors say.
They point out that therapeutic inertia has been identified as a key clinician-level barrier to blood pressure control and is estimated to be present in more than 80% of clinic visits in community practice, whereas in the current analysis of the SPRINT trial, therapeutic inertia was present in 50% to 60% of participant visits with uncontrolled blood pressure.
“In SPRINT, blood pressure had to be measured at defined intervals with a specific method, and there were clear instructions on intensifying treatment if blood pressure was above a certain goal,” Dr. Zheutlin noted. “Our results show that within such strict confines, therapeutic inertia does not seem to be different between different racial groups. This suggests that we could make better gains in blood pressure control and more equitable treatment if we adopted a standardized approach to hypertension management.”
He added: “Many guidelines have been published on when to start treatment and the targets for blood pressure, but there is a lot of variation in how we turn these guidelines into protocols. We need to bring in more consistent protocols on blood pressure measurement and intensification and ensure they are followed. In practice, if the BP is 5 or 10 mm Hg above target, a clinician may defer a decision to intensify treatment and intensification never gets done. But if there was a strict protocol to follow, there would be less chance of this happening.”
Therapeutic inertia still high
In an accompanying commentary, Matthew Rivara, MD, Nisha Bansal, MD, and Bessie Young, MD, University of Washington, Seattle, say the current SPRINT analysis has broad implications for reducing racial and ethnic disparities in achievement of evidence-based treatment targets in the general population.
“In hypertension management, standardized protocols for medication adjustments may limit clinician practice heterogeneity to ultimately reduce differences in blood pressure control among racial and ethnic minority populations,” they write. But they add that such protocols must be implemented thoughtfully to incorporate individualized clinical assessment and clinician-patient shared decision-making.
Dr. Rivara et al. point out that the rates of therapeutic inertia in SPRINT, while lower than community-based estimates, were still very high. They suggest reasons for this could include clinician concerns about medication efficacy, adverse effects, and patient mistrust of medical professionals. Outside the clinical trial environment, additional considerations may include prescription drug and laboratory test costs, pharmacy access, and competing demands during busy clinic visits.
To address these challenges, they say that clinicians need education on current clinical practice guidelines, managing complications of intensified antihypertensive therapies, and shared decisionmaking, including culturally sensitive collaborative care. Similarly, care systems must support patients on how to address concerns about treatments.
Finally, further research is needed to better define the specific factors associated with therapeutic inertia to allow tailored interventions to overcome this inertia.
“In designing and performing such research, it is vital that investigators engage with racial and ethnic minority groups to better explore the intersection of race, ethnicity, therapeutic decision-making, trust, and shared decisionmaking,” they add.
The SPRINT trial was funded with federal funds from the National Institutes of Health. Dr. Zheutlin reported receiving grants from the NIH during the conduct of the study.
A version of this article first appeared on Medscape.com.
FROM JAMA NETWORK OPEN
AHA advice for diabetes patients to stay heart healthy
A new document from the American Heart Association summarizes the latest research on cardiovascular risk factor management in type 2 diabetes, including medications, lifestyle, and social determinants of health.
Despite the availability of effective therapies for improving cardiovascular risk, in the United States fewer than one in five people with type 2 diabetes and without known cardiovascular disease meet control targets for a combination of A1c, blood pressure, LDL cholesterol, and nonsmoking status.
That proportion drops to less than 1 in 10 if body mass index less than 30 kg/m2 is included among the targets, and even less than that among individuals with established atherosclerotic cardiovascular disease, Joshua J. Joseph, MD, and colleagues point out in their paper, published online Jan. 10 in Circulation.
“This new scientific statement is an urgent call to action to follow the latest evidence-based approaches and to develop new best practices to advance type 2 diabetes treatment and care and reduce cardiovascular disease risk,” wrote Dr. Joseph, assistant professor of medicine in the division of endocrinology, diabetes, and metabolism at The Ohio State University, Columbus, Ohio, and coauthors.
The statement is not a guideline but an expert analysis that may inform future clinical practice guidelines, according to a press release from the AHA.
The new statement reviews evidence through June 2020 for lifestyle management of diabetes and weight, glycemic targets and control, blood pressure management, lipid management, antithrombotic therapy, and screening for cardiovascular and renal complications, including imaging. It also discusses the clinical implications of recent cardiovascular outcomes trials of newer glucose-lowering medications.
However, Dr. Joseph and colleagues point out, clinical care and treatment account for just 10%-20% of modifiable contributors to health outcomes. The other 80%-90% relate to social determinants of health, including health-related behaviors, socioeconomic factors, environmental factors, and racism.
“If we are to continue to advance the management of cardiovascular risk factors, we must also address the [social determinants of health] in the delivery of health care,” they noted.
Overall, they advise a patient-centered approach, meaning “reframing our clinical encounters to think about patients as people who live in families, communities, and societies that must be considered in their cardiovascular risk management.”
“People with [type 2 diabetes] face numerous barriers to health including access to care and equitable care, which must be considered when developing individualized care plans with our patients,” Dr. Joseph said in the AHA press release.
Lifestyle, medications for lowering A1c, BP, lipids
For lifestyle management, the authors say, “culturally appropriate recommendations through diabetes self-management education and support and medical nutrition therapy are key to meeting individualized goals for behavioral change and diabetes self-management.”
The document summarizes recommendations from other professional societies regarding glycemic targets and glucose lowering medications, i.e., target A1c levels of either < 7% or < 6.5% for the majority, with adjustments based on individual factors, such as life expectancy. It advises on use of metformin as first-line therapy followed by a sodium-glucose cotransporter-2 inhibitor or a glucagon-like peptide-1 agonist for those with established cardiovascular disease or risk factors.
“Cost may be a barrier to taking some [type 2 diabetes] medications as prescribed; however, many of these medications are now more commonly covered by more health insurance plans,” Dr. Joseph said.
“Another barrier is recognition by patients that these newer [type 2 diabetes] medications are also effective in reducing the risk of heart disease, stroke, heart failure, and kidney disease.”
Blood pressure treatment guidelines differ between those of the AHA/American College of Cardiology (ACC) and the American Diabetes Association (ADA), most notably that the AHA/ACC guidelines advise a general target of < 130/80 mm Hg, whereas ADA advises < 140/90 mm Hg or < 130/80 mm Hg for those with high risk if it can be safely achieved.
The decision should be “patient-centered with shared decision-making,” Dr. Joseph and colleagues advised.
For lipid-lowering, the document cites the 2018 ACC/AHA cholesterol guidelines, which include advising statins as first-line therapy for both primary and secondary prevention in diabetes, with highest intensity statins used in those at highest risk. But again, treatment should be individualized, and other agents should be used for patients in whom statins don’t work or aren’t tolerated.
And while use of antiplatelets – that is, aspirin – is well established as secondary prevention in type 2 diabetes, given new data suggesting that the risk for major bleeding could outweigh the benefits for primary prevention, “the relative benefits of antithrombotic approaches need to be weighed carefully against risks using a patient-centered approach,” the authors advised.
Among the many imaging tests available to facilitate cardiovascular risk stratification in type 2 diabetes, coronary artery calcification (CAC) CT screening is one of the few with sufficient data to support routine use in selected patients. The National Lipid Association, for example, recommends escalation to high-intensity statin for CAC > 100.
“One avenue to continue to address and advance diabetes management is through breaking down the four walls of the clinic or hospital through community engagement, clinic-to-community connections, and academic-community-government partnerships that may help address and support modifiable lifestyle behaviors such as physical activity, nutrition, smoking cessation and stress management,” Dr. Joseph concluded.
The AHA receives funding primarily from individuals. Foundations and corporations, including pharmaceutical, device manufacturers, and other companies, also make donations and fund AHA programs and events. The AHA’s strict policies prevent these relationships from influencing the science content. Revenues from pharmaceutical and biotech companies, device manufacturers, and health insurance providers and the AHA’s financial information are available on the association’s website. Dr. Joseph has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A new document from the American Heart Association summarizes the latest research on cardiovascular risk factor management in type 2 diabetes, including medications, lifestyle, and social determinants of health.
Despite the availability of effective therapies for improving cardiovascular risk, in the United States fewer than one in five people with type 2 diabetes and without known cardiovascular disease meet control targets for a combination of A1c, blood pressure, LDL cholesterol, and nonsmoking status.
That proportion drops to less than 1 in 10 if body mass index less than 30 kg/m2 is included among the targets, and even less than that among individuals with established atherosclerotic cardiovascular disease, Joshua J. Joseph, MD, and colleagues point out in their paper, published online Jan. 10 in Circulation.
“This new scientific statement is an urgent call to action to follow the latest evidence-based approaches and to develop new best practices to advance type 2 diabetes treatment and care and reduce cardiovascular disease risk,” wrote Dr. Joseph, assistant professor of medicine in the division of endocrinology, diabetes, and metabolism at The Ohio State University, Columbus, Ohio, and coauthors.
The statement is not a guideline but an expert analysis that may inform future clinical practice guidelines, according to a press release from the AHA.
The new statement reviews evidence through June 2020 for lifestyle management of diabetes and weight, glycemic targets and control, blood pressure management, lipid management, antithrombotic therapy, and screening for cardiovascular and renal complications, including imaging. It also discusses the clinical implications of recent cardiovascular outcomes trials of newer glucose-lowering medications.
However, Dr. Joseph and colleagues point out, clinical care and treatment account for just 10%-20% of modifiable contributors to health outcomes. The other 80%-90% relate to social determinants of health, including health-related behaviors, socioeconomic factors, environmental factors, and racism.
“If we are to continue to advance the management of cardiovascular risk factors, we must also address the [social determinants of health] in the delivery of health care,” they noted.
Overall, they advise a patient-centered approach, meaning “reframing our clinical encounters to think about patients as people who live in families, communities, and societies that must be considered in their cardiovascular risk management.”
“People with [type 2 diabetes] face numerous barriers to health including access to care and equitable care, which must be considered when developing individualized care plans with our patients,” Dr. Joseph said in the AHA press release.
Lifestyle, medications for lowering A1c, BP, lipids
For lifestyle management, the authors say, “culturally appropriate recommendations through diabetes self-management education and support and medical nutrition therapy are key to meeting individualized goals for behavioral change and diabetes self-management.”
The document summarizes recommendations from other professional societies regarding glycemic targets and glucose lowering medications, i.e., target A1c levels of either < 7% or < 6.5% for the majority, with adjustments based on individual factors, such as life expectancy. It advises on use of metformin as first-line therapy followed by a sodium-glucose cotransporter-2 inhibitor or a glucagon-like peptide-1 agonist for those with established cardiovascular disease or risk factors.
“Cost may be a barrier to taking some [type 2 diabetes] medications as prescribed; however, many of these medications are now more commonly covered by more health insurance plans,” Dr. Joseph said.
“Another barrier is recognition by patients that these newer [type 2 diabetes] medications are also effective in reducing the risk of heart disease, stroke, heart failure, and kidney disease.”
Blood pressure treatment guidelines differ between those of the AHA/American College of Cardiology (ACC) and the American Diabetes Association (ADA), most notably that the AHA/ACC guidelines advise a general target of < 130/80 mm Hg, whereas ADA advises < 140/90 mm Hg or < 130/80 mm Hg for those with high risk if it can be safely achieved.
The decision should be “patient-centered with shared decision-making,” Dr. Joseph and colleagues advised.
For lipid-lowering, the document cites the 2018 ACC/AHA cholesterol guidelines, which include advising statins as first-line therapy for both primary and secondary prevention in diabetes, with highest intensity statins used in those at highest risk. But again, treatment should be individualized, and other agents should be used for patients in whom statins don’t work or aren’t tolerated.
And while use of antiplatelets – that is, aspirin – is well established as secondary prevention in type 2 diabetes, given new data suggesting that the risk for major bleeding could outweigh the benefits for primary prevention, “the relative benefits of antithrombotic approaches need to be weighed carefully against risks using a patient-centered approach,” the authors advised.
Among the many imaging tests available to facilitate cardiovascular risk stratification in type 2 diabetes, coronary artery calcification (CAC) CT screening is one of the few with sufficient data to support routine use in selected patients. The National Lipid Association, for example, recommends escalation to high-intensity statin for CAC > 100.
“One avenue to continue to address and advance diabetes management is through breaking down the four walls of the clinic or hospital through community engagement, clinic-to-community connections, and academic-community-government partnerships that may help address and support modifiable lifestyle behaviors such as physical activity, nutrition, smoking cessation and stress management,” Dr. Joseph concluded.
The AHA receives funding primarily from individuals. Foundations and corporations, including pharmaceutical, device manufacturers, and other companies, also make donations and fund AHA programs and events. The AHA’s strict policies prevent these relationships from influencing the science content. Revenues from pharmaceutical and biotech companies, device manufacturers, and health insurance providers and the AHA’s financial information are available on the association’s website. Dr. Joseph has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A new document from the American Heart Association summarizes the latest research on cardiovascular risk factor management in type 2 diabetes, including medications, lifestyle, and social determinants of health.
Despite the availability of effective therapies for improving cardiovascular risk, in the United States fewer than one in five people with type 2 diabetes and without known cardiovascular disease meet control targets for a combination of A1c, blood pressure, LDL cholesterol, and nonsmoking status.
That proportion drops to less than 1 in 10 if body mass index less than 30 kg/m2 is included among the targets, and even less than that among individuals with established atherosclerotic cardiovascular disease, Joshua J. Joseph, MD, and colleagues point out in their paper, published online Jan. 10 in Circulation.
“This new scientific statement is an urgent call to action to follow the latest evidence-based approaches and to develop new best practices to advance type 2 diabetes treatment and care and reduce cardiovascular disease risk,” wrote Dr. Joseph, assistant professor of medicine in the division of endocrinology, diabetes, and metabolism at The Ohio State University, Columbus, Ohio, and coauthors.
The statement is not a guideline but an expert analysis that may inform future clinical practice guidelines, according to a press release from the AHA.
The new statement reviews evidence through June 2020 for lifestyle management of diabetes and weight, glycemic targets and control, blood pressure management, lipid management, antithrombotic therapy, and screening for cardiovascular and renal complications, including imaging. It also discusses the clinical implications of recent cardiovascular outcomes trials of newer glucose-lowering medications.
However, Dr. Joseph and colleagues point out, clinical care and treatment account for just 10%-20% of modifiable contributors to health outcomes. The other 80%-90% relate to social determinants of health, including health-related behaviors, socioeconomic factors, environmental factors, and racism.
“If we are to continue to advance the management of cardiovascular risk factors, we must also address the [social determinants of health] in the delivery of health care,” they noted.
Overall, they advise a patient-centered approach, meaning “reframing our clinical encounters to think about patients as people who live in families, communities, and societies that must be considered in their cardiovascular risk management.”
“People with [type 2 diabetes] face numerous barriers to health including access to care and equitable care, which must be considered when developing individualized care plans with our patients,” Dr. Joseph said in the AHA press release.
Lifestyle, medications for lowering A1c, BP, lipids
For lifestyle management, the authors say, “culturally appropriate recommendations through diabetes self-management education and support and medical nutrition therapy are key to meeting individualized goals for behavioral change and diabetes self-management.”
The document summarizes recommendations from other professional societies regarding glycemic targets and glucose lowering medications, i.e., target A1c levels of either < 7% or < 6.5% for the majority, with adjustments based on individual factors, such as life expectancy. It advises on use of metformin as first-line therapy followed by a sodium-glucose cotransporter-2 inhibitor or a glucagon-like peptide-1 agonist for those with established cardiovascular disease or risk factors.
“Cost may be a barrier to taking some [type 2 diabetes] medications as prescribed; however, many of these medications are now more commonly covered by more health insurance plans,” Dr. Joseph said.
“Another barrier is recognition by patients that these newer [type 2 diabetes] medications are also effective in reducing the risk of heart disease, stroke, heart failure, and kidney disease.”
Blood pressure treatment guidelines differ between those of the AHA/American College of Cardiology (ACC) and the American Diabetes Association (ADA), most notably that the AHA/ACC guidelines advise a general target of < 130/80 mm Hg, whereas ADA advises < 140/90 mm Hg or < 130/80 mm Hg for those with high risk if it can be safely achieved.
The decision should be “patient-centered with shared decision-making,” Dr. Joseph and colleagues advised.
For lipid-lowering, the document cites the 2018 ACC/AHA cholesterol guidelines, which include advising statins as first-line therapy for both primary and secondary prevention in diabetes, with highest intensity statins used in those at highest risk. But again, treatment should be individualized, and other agents should be used for patients in whom statins don’t work or aren’t tolerated.
And while use of antiplatelets – that is, aspirin – is well established as secondary prevention in type 2 diabetes, given new data suggesting that the risk for major bleeding could outweigh the benefits for primary prevention, “the relative benefits of antithrombotic approaches need to be weighed carefully against risks using a patient-centered approach,” the authors advised.
Among the many imaging tests available to facilitate cardiovascular risk stratification in type 2 diabetes, coronary artery calcification (CAC) CT screening is one of the few with sufficient data to support routine use in selected patients. The National Lipid Association, for example, recommends escalation to high-intensity statin for CAC > 100.
“One avenue to continue to address and advance diabetes management is through breaking down the four walls of the clinic or hospital through community engagement, clinic-to-community connections, and academic-community-government partnerships that may help address and support modifiable lifestyle behaviors such as physical activity, nutrition, smoking cessation and stress management,” Dr. Joseph concluded.
The AHA receives funding primarily from individuals. Foundations and corporations, including pharmaceutical, device manufacturers, and other companies, also make donations and fund AHA programs and events. The AHA’s strict policies prevent these relationships from influencing the science content. Revenues from pharmaceutical and biotech companies, device manufacturers, and health insurance providers and the AHA’s financial information are available on the association’s website. Dr. Joseph has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Sarcoidosis

THE COMPARISON
A Pink, elevated, granulomatous, indurated plaques on the face, including the nasal alae, of a 52-year-old woman with a darker skin tone.
B Orange and pink, elevated, granulomatous, indurated plaques on the face of a 55-year-old woman with a lighter skin tone.
Sarcoidosis is a granulomatous disease that may affect the skin in addition to multiple body organ systems, including the lungs. Bilateral hilar adenopathy on a chest radiograph is the most common finding. Sarcoidosis also has a variety of cutaneous manifestations. Early diagnosis is vital, as patients with with sarcoidosis and pulmonary fibrosis have a shortened life span compared to the overall population.1 With a growing skin of color population, it is important to recognize sarcoidosis as soon as possible.2
Epidemiology
People of African descent have the highest sarcoidosis prevalence in the United States.3 In the United States, the incidence of sarcoidosis in Black individuals peaks in the fourth decade of life. A 5-year study in a US health maintenance organization found that the age-adjusted annual incidence was 10.9 per 100,000 cases among Whites and 35.5 per 100,000 cases among Blacks.4
Key clinical features in people with darker skin tones:
• Papules are seen in sarcoidosis, primarily on the face, and may start as orange hued or yellow-brown and then become brown-red or pink to violaceous before involuting into faint macules.5-7
• When round or oval sarcoid plaques appear, they often are more erythematous. In skin of color, plaques may become hypopigmented.8
• Erythema nodosum, the most common nonspecific cutaneous lesion seen in sarcoidosis, is less commonly seen in those of African and Asian descent.9-11 This is in contrast to distinctive forms of specific sarcoid skin lesions such as lupus pernio and scar sarcoidosis, as well as papules and plaques and minor forms of specific sarcoid skin lesions including subcutaneous nodules; hypopigmented macules; psoriasiform lesions; and ulcerative, localized erythrodermic, ichthyosiform, scalp, and nail lesions.
• Lupus pernio is a cutaneous manifestation of sarcoidosis that appears on the face. It looks similar to lupus erythematosus and occurs most commonly in women of African descent.8,12
• Hypopigmented lesions are more common in those with darker skin tones.9
• Ulcerative lesions are more common in those of African descent and women.13
• Scalp sarcoidosis is more common in patients of African descent.14
• Sarcoidosis may develop at sites of trauma, such as scars and tattoos.15-17
Worth noting
The cutaneous lesions seen in sarcoidosis may be emotionally devastating and disfiguring. Due to the variety of clinical manifestations, sarcoidosis may be misdiagnosed, leading to delays in treatment.18
Health disparity highlight
Patients older than 40 years presenting with sarcoidosis and those of African descent have a worse prognosis.19 Despite adjusting for race, ethnic group, age, and sex, patients with low income and financial barriers present with more severe sarcoidosis.20
- Nardi A, Brillet P-Y, Letoumelin P, et al. Stage IV sarcoidosis: comparison of survival with the general population and causes of death. Eur Respir J. 2011;38:1368-1373.
- Heath CR, David J, Taylor SC. Sarcoidosis: are there differences in your skin of color patients? J Am Acad Dermatol. 2012;66:121.e1-121.e14.
- Sève P, Pacheco Y, Durupt F, et al. Sarcoidosis: a clinical overview from symptoms to diagnosis. Cells. 2021;10:766. doi:10.3390/cells10040766
- Rybicki BA, Major M, Popovich J Jr, et al. Racial differences in sarcoidosis incidence: a 5-year study in a health maintenance organization. Am J Epidemiol. 1997;145:234-241. doi:10.1093/oxfordjournals.aje.a009096
- Mahajan VK, Sharma NL, Sharma RC, et al. Cutaneous sarcoidosis: clinical profile of 23 Indian patients. Indian J Dermatol Venerol Leprol. 2007;73:16-21.
- Yanardag H, Pamuk ON, Karayel T. Cutaneous involvement in sarcoidosis: analysis if the features in 170 patients. Respir Med. 2003;97:978-982.
- Olive KE, Kartaria YP. Cutaneous manifestations of sarcoidosis to other organ system involvement, abnormal laboratory measurements, and disease course. Arch Intern Med. 1985;145:1811-1814.
- Mañá J, Marcoval J, Graells J, et al. Cutaneous involvement in sarcoidosis. relationship to systemic disease. Arch Dermatol. 1997;133:882-888. doi:10.1001/archderm.1997.03890430098013
- Minus HR, Grimes PE. Cutaneous manifestations of sarcoidosis in blacks. Cutis. 1983;32:361-364.
- Edmondstone WM, Wilson AG. Sarcoidosis in Caucasians, blacks and Asians in London. Br J Dis Chest. 1985;79:27-36.
- James DG, Neville E, Siltzbach LE. Worldwide review of sarcoidosis. Ann N Y Acad Sci. 1976;278:321-334.
- Hunninghake GW, Costabel U, Ando M, et al. ATS/ERS/WASOG statement on sarcoidosis. American Thoracic Society/European Respiratory Society/World Association of Sarcoidosis and other Granulomatous Disorders. Sarcoidosis Vasc Diffuse Lung Dis. 1999;16:149-173.
- Albertini JG, Tyler W, Miller OF III. Ulcerative sarcoidosis: case report and review of literature. Arch Dermatol. 1997;133:215-219.
- Marchell RM, Judson MA. Chronic cutaneous lesions of sarcoidosis. Clin Dermatol. 2007;25:295-302.
- Nayar M. Sarcoidosis on ritual scarification. Int J Dermatol. 1993;32:116-118.
- Chudomirova K, Velichkva L, Anavi B. Recurrent sarcoidosis in skin scars accompanying systemic sarcoidosis. J Eur Acad Dermatol Venerol. 2003;17:360-361.
- Kim YC, Triffet MK, Gibson LE. Foreign bodies in sarcoidosis. Am J Dermatopathol. 2000;22:408-412.
- Iannuzzi MC, Rybicki BA, Teirstein AS. Sarcoidosis. N Engl J Med. 2007; 357:2153-2165.
- Nunes H, Bouvry D, Soler P, et al. Sarcoidosis. Orphanet J Rare Dis. 2007;2:46. doi:10.1186/1750-1172-2-46
- Baughman RP, Teirstein AS, Judson MA, et al. Clinical characteristics of patients in a case control study of sarcoidosis. Am J Respir Crit Care Med. 2001;164:1885-1889.

THE COMPARISON
A Pink, elevated, granulomatous, indurated plaques on the face, including the nasal alae, of a 52-year-old woman with a darker skin tone.
B Orange and pink, elevated, granulomatous, indurated plaques on the face of a 55-year-old woman with a lighter skin tone.
Sarcoidosis is a granulomatous disease that may affect the skin in addition to multiple body organ systems, including the lungs. Bilateral hilar adenopathy on a chest radiograph is the most common finding. Sarcoidosis also has a variety of cutaneous manifestations. Early diagnosis is vital, as patients with with sarcoidosis and pulmonary fibrosis have a shortened life span compared to the overall population.1 With a growing skin of color population, it is important to recognize sarcoidosis as soon as possible.2
Epidemiology
People of African descent have the highest sarcoidosis prevalence in the United States.3 In the United States, the incidence of sarcoidosis in Black individuals peaks in the fourth decade of life. A 5-year study in a US health maintenance organization found that the age-adjusted annual incidence was 10.9 per 100,000 cases among Whites and 35.5 per 100,000 cases among Blacks.4
Key clinical features in people with darker skin tones:
• Papules are seen in sarcoidosis, primarily on the face, and may start as orange hued or yellow-brown and then become brown-red or pink to violaceous before involuting into faint macules.5-7
• When round or oval sarcoid plaques appear, they often are more erythematous. In skin of color, plaques may become hypopigmented.8
• Erythema nodosum, the most common nonspecific cutaneous lesion seen in sarcoidosis, is less commonly seen in those of African and Asian descent.9-11 This is in contrast to distinctive forms of specific sarcoid skin lesions such as lupus pernio and scar sarcoidosis, as well as papules and plaques and minor forms of specific sarcoid skin lesions including subcutaneous nodules; hypopigmented macules; psoriasiform lesions; and ulcerative, localized erythrodermic, ichthyosiform, scalp, and nail lesions.
• Lupus pernio is a cutaneous manifestation of sarcoidosis that appears on the face. It looks similar to lupus erythematosus and occurs most commonly in women of African descent.8,12
• Hypopigmented lesions are more common in those with darker skin tones.9
• Ulcerative lesions are more common in those of African descent and women.13
• Scalp sarcoidosis is more common in patients of African descent.14
• Sarcoidosis may develop at sites of trauma, such as scars and tattoos.15-17
Worth noting
The cutaneous lesions seen in sarcoidosis may be emotionally devastating and disfiguring. Due to the variety of clinical manifestations, sarcoidosis may be misdiagnosed, leading to delays in treatment.18
Health disparity highlight
Patients older than 40 years presenting with sarcoidosis and those of African descent have a worse prognosis.19 Despite adjusting for race, ethnic group, age, and sex, patients with low income and financial barriers present with more severe sarcoidosis.20

THE COMPARISON
A Pink, elevated, granulomatous, indurated plaques on the face, including the nasal alae, of a 52-year-old woman with a darker skin tone.
B Orange and pink, elevated, granulomatous, indurated plaques on the face of a 55-year-old woman with a lighter skin tone.
Sarcoidosis is a granulomatous disease that may affect the skin in addition to multiple body organ systems, including the lungs. Bilateral hilar adenopathy on a chest radiograph is the most common finding. Sarcoidosis also has a variety of cutaneous manifestations. Early diagnosis is vital, as patients with with sarcoidosis and pulmonary fibrosis have a shortened life span compared to the overall population.1 With a growing skin of color population, it is important to recognize sarcoidosis as soon as possible.2
Epidemiology
People of African descent have the highest sarcoidosis prevalence in the United States.3 In the United States, the incidence of sarcoidosis in Black individuals peaks in the fourth decade of life. A 5-year study in a US health maintenance organization found that the age-adjusted annual incidence was 10.9 per 100,000 cases among Whites and 35.5 per 100,000 cases among Blacks.4
Key clinical features in people with darker skin tones:
• Papules are seen in sarcoidosis, primarily on the face, and may start as orange hued or yellow-brown and then become brown-red or pink to violaceous before involuting into faint macules.5-7
• When round or oval sarcoid plaques appear, they often are more erythematous. In skin of color, plaques may become hypopigmented.8
• Erythema nodosum, the most common nonspecific cutaneous lesion seen in sarcoidosis, is less commonly seen in those of African and Asian descent.9-11 This is in contrast to distinctive forms of specific sarcoid skin lesions such as lupus pernio and scar sarcoidosis, as well as papules and plaques and minor forms of specific sarcoid skin lesions including subcutaneous nodules; hypopigmented macules; psoriasiform lesions; and ulcerative, localized erythrodermic, ichthyosiform, scalp, and nail lesions.
• Lupus pernio is a cutaneous manifestation of sarcoidosis that appears on the face. It looks similar to lupus erythematosus and occurs most commonly in women of African descent.8,12
• Hypopigmented lesions are more common in those with darker skin tones.9
• Ulcerative lesions are more common in those of African descent and women.13
• Scalp sarcoidosis is more common in patients of African descent.14
• Sarcoidosis may develop at sites of trauma, such as scars and tattoos.15-17
Worth noting
The cutaneous lesions seen in sarcoidosis may be emotionally devastating and disfiguring. Due to the variety of clinical manifestations, sarcoidosis may be misdiagnosed, leading to delays in treatment.18
Health disparity highlight
Patients older than 40 years presenting with sarcoidosis and those of African descent have a worse prognosis.19 Despite adjusting for race, ethnic group, age, and sex, patients with low income and financial barriers present with more severe sarcoidosis.20
- Nardi A, Brillet P-Y, Letoumelin P, et al. Stage IV sarcoidosis: comparison of survival with the general population and causes of death. Eur Respir J. 2011;38:1368-1373.
- Heath CR, David J, Taylor SC. Sarcoidosis: are there differences in your skin of color patients? J Am Acad Dermatol. 2012;66:121.e1-121.e14.
- Sève P, Pacheco Y, Durupt F, et al. Sarcoidosis: a clinical overview from symptoms to diagnosis. Cells. 2021;10:766. doi:10.3390/cells10040766
- Rybicki BA, Major M, Popovich J Jr, et al. Racial differences in sarcoidosis incidence: a 5-year study in a health maintenance organization. Am J Epidemiol. 1997;145:234-241. doi:10.1093/oxfordjournals.aje.a009096
- Mahajan VK, Sharma NL, Sharma RC, et al. Cutaneous sarcoidosis: clinical profile of 23 Indian patients. Indian J Dermatol Venerol Leprol. 2007;73:16-21.
- Yanardag H, Pamuk ON, Karayel T. Cutaneous involvement in sarcoidosis: analysis if the features in 170 patients. Respir Med. 2003;97:978-982.
- Olive KE, Kartaria YP. Cutaneous manifestations of sarcoidosis to other organ system involvement, abnormal laboratory measurements, and disease course. Arch Intern Med. 1985;145:1811-1814.
- Mañá J, Marcoval J, Graells J, et al. Cutaneous involvement in sarcoidosis. relationship to systemic disease. Arch Dermatol. 1997;133:882-888. doi:10.1001/archderm.1997.03890430098013
- Minus HR, Grimes PE. Cutaneous manifestations of sarcoidosis in blacks. Cutis. 1983;32:361-364.
- Edmondstone WM, Wilson AG. Sarcoidosis in Caucasians, blacks and Asians in London. Br J Dis Chest. 1985;79:27-36.
- James DG, Neville E, Siltzbach LE. Worldwide review of sarcoidosis. Ann N Y Acad Sci. 1976;278:321-334.
- Hunninghake GW, Costabel U, Ando M, et al. ATS/ERS/WASOG statement on sarcoidosis. American Thoracic Society/European Respiratory Society/World Association of Sarcoidosis and other Granulomatous Disorders. Sarcoidosis Vasc Diffuse Lung Dis. 1999;16:149-173.
- Albertini JG, Tyler W, Miller OF III. Ulcerative sarcoidosis: case report and review of literature. Arch Dermatol. 1997;133:215-219.
- Marchell RM, Judson MA. Chronic cutaneous lesions of sarcoidosis. Clin Dermatol. 2007;25:295-302.
- Nayar M. Sarcoidosis on ritual scarification. Int J Dermatol. 1993;32:116-118.
- Chudomirova K, Velichkva L, Anavi B. Recurrent sarcoidosis in skin scars accompanying systemic sarcoidosis. J Eur Acad Dermatol Venerol. 2003;17:360-361.
- Kim YC, Triffet MK, Gibson LE. Foreign bodies in sarcoidosis. Am J Dermatopathol. 2000;22:408-412.
- Iannuzzi MC, Rybicki BA, Teirstein AS. Sarcoidosis. N Engl J Med. 2007; 357:2153-2165.
- Nunes H, Bouvry D, Soler P, et al. Sarcoidosis. Orphanet J Rare Dis. 2007;2:46. doi:10.1186/1750-1172-2-46
- Baughman RP, Teirstein AS, Judson MA, et al. Clinical characteristics of patients in a case control study of sarcoidosis. Am J Respir Crit Care Med. 2001;164:1885-1889.
- Nardi A, Brillet P-Y, Letoumelin P, et al. Stage IV sarcoidosis: comparison of survival with the general population and causes of death. Eur Respir J. 2011;38:1368-1373.
- Heath CR, David J, Taylor SC. Sarcoidosis: are there differences in your skin of color patients? J Am Acad Dermatol. 2012;66:121.e1-121.e14.
- Sève P, Pacheco Y, Durupt F, et al. Sarcoidosis: a clinical overview from symptoms to diagnosis. Cells. 2021;10:766. doi:10.3390/cells10040766
- Rybicki BA, Major M, Popovich J Jr, et al. Racial differences in sarcoidosis incidence: a 5-year study in a health maintenance organization. Am J Epidemiol. 1997;145:234-241. doi:10.1093/oxfordjournals.aje.a009096
- Mahajan VK, Sharma NL, Sharma RC, et al. Cutaneous sarcoidosis: clinical profile of 23 Indian patients. Indian J Dermatol Venerol Leprol. 2007;73:16-21.
- Yanardag H, Pamuk ON, Karayel T. Cutaneous involvement in sarcoidosis: analysis if the features in 170 patients. Respir Med. 2003;97:978-982.
- Olive KE, Kartaria YP. Cutaneous manifestations of sarcoidosis to other organ system involvement, abnormal laboratory measurements, and disease course. Arch Intern Med. 1985;145:1811-1814.
- Mañá J, Marcoval J, Graells J, et al. Cutaneous involvement in sarcoidosis. relationship to systemic disease. Arch Dermatol. 1997;133:882-888. doi:10.1001/archderm.1997.03890430098013
- Minus HR, Grimes PE. Cutaneous manifestations of sarcoidosis in blacks. Cutis. 1983;32:361-364.
- Edmondstone WM, Wilson AG. Sarcoidosis in Caucasians, blacks and Asians in London. Br J Dis Chest. 1985;79:27-36.
- James DG, Neville E, Siltzbach LE. Worldwide review of sarcoidosis. Ann N Y Acad Sci. 1976;278:321-334.
- Hunninghake GW, Costabel U, Ando M, et al. ATS/ERS/WASOG statement on sarcoidosis. American Thoracic Society/European Respiratory Society/World Association of Sarcoidosis and other Granulomatous Disorders. Sarcoidosis Vasc Diffuse Lung Dis. 1999;16:149-173.
- Albertini JG, Tyler W, Miller OF III. Ulcerative sarcoidosis: case report and review of literature. Arch Dermatol. 1997;133:215-219.
- Marchell RM, Judson MA. Chronic cutaneous lesions of sarcoidosis. Clin Dermatol. 2007;25:295-302.
- Nayar M. Sarcoidosis on ritual scarification. Int J Dermatol. 1993;32:116-118.
- Chudomirova K, Velichkva L, Anavi B. Recurrent sarcoidosis in skin scars accompanying systemic sarcoidosis. J Eur Acad Dermatol Venerol. 2003;17:360-361.
- Kim YC, Triffet MK, Gibson LE. Foreign bodies in sarcoidosis. Am J Dermatopathol. 2000;22:408-412.
- Iannuzzi MC, Rybicki BA, Teirstein AS. Sarcoidosis. N Engl J Med. 2007; 357:2153-2165.
- Nunes H, Bouvry D, Soler P, et al. Sarcoidosis. Orphanet J Rare Dis. 2007;2:46. doi:10.1186/1750-1172-2-46
- Baughman RP, Teirstein AS, Judson MA, et al. Clinical characteristics of patients in a case control study of sarcoidosis. Am J Respir Crit Care Med. 2001;164:1885-1889.
The Leaky Pipeline: A Narrative Review of Diversity in Dermatology
With a majority-minority population expected in the United States by 2044, improving diversity and cultural competency in the dermatology workforce is now more important than ever. A more diverse workforce increases the cultural competence of all providers, provides greater opportunities for mentorship and sponsorship of underrepresented minority (URM) trainees, establishes a more inclusive environment for learners, and enhances the knowledge and productivity of the workforce.1-3 Additionally, it is imperative to address clinical care disparities seen in minority patients in dermatology, including treatment of skin cancer, psoriasis, acne, atopic dermatitis, and other diseases.4-7
Despite the attention that has been devoted to improving diversity in medicine,8-10 dermatology remains one of the least diverse specialties, prompting additional calls to action within the field.11 Why does the lack of diversity still exist in dermatology, and what is the path to correcting this problem? In this article, we review the evidence of diversity barriers at different stages of medical education training that may impede academic advancement for minority learners pursuing careers in dermatology.
Undergraduate Medical Education
The term leaky pipeline refers to the progressive decline in the number of URMs along a given career path, including in dermatology. The Association of American Medical Colleges defines URMs as racial/ethnic populations that are “underrepresented in the medical profession relative to their numbers in the general population.”9 The first leak in the pipeline is that URMs are not applying to medical school. From 2002 and 2017, rates of both application and matriculation to medical school were lower by 30% to 70% in URM groups compared to White students, including Hispanic, Black, and American Indian/Alaska Native students.12,13 The decision not to apply to medical school was greater in URM undergraduate students irrespective of scholastic ability as measured by SAT scores.14
A striking statistic is that the number of Black men matriculating into medical school in 2014 was less than it was in 1978 despite the increase in the number of US medical schools and efforts to recruit more diverse student populations. The Association of American Medical Colleges identified potential reasons for this decline, including poor early education, lack of mentorship, negative perceptions of Black men due to racial stereotypes, and lack of financial and academic resources to support the application process.8,13,15-17 Implicit racial bias by admission committees also may play a role.
Medical School Matriculation and Applying to Dermatology Residency
There is greater representation of URM students in medical school than in dermatology residency, which means URM students are either not applying to dermatology programs or they are not matching into the specialty. In the Electronic Residency Application Service’s 2016-2017 application cycle (N=776), there were 76 (9.8%) URM dermatology residency applicants.18 In 2018, there was a notable decline in representation of Black students among residency applicants (4.9%) to matched residents (3.7%), and there were only 133 (9.3%) URM dermatology residents in total (PGY2-PGY4 classes).19 The lack of exposure to medical subspecialties and the recommendation by medical schools for URM medical students to pursue careers in primary care have been cited as reasons that these students may not apply to residency programs in specialty care.20,21 The presence of an Accreditation Council for Graduate Medical Education dermatology residency program, fellowships, and dermatology interest groups at their medical schools correlated with higher proportions of URM students applying to dermatology programs.20
Underrepresented minority students face critical challenges during medical school, including receiving lower grades in both standardized and school-designated assessments and clerkship grades.21,22 A 2019 National Board of Medical Examiners study found that Hispanic and Black test takers scored 12.1 and 16.6 points lower than White men, respectively, on the
A recent cross-sectional study showed that lack of equitable resources, lack of support, financial constrictions, and lack of group identity were 4 barriers to URM students matching into dermatology.26 Dermatology is a competitive specialty with the highest median Electronic Residency Application Service applications submitted per US applicant (n=90)27 and an approximate total cost per US applicant of $10,781.28,29 Disadvantaged URM applicants noted relying on loans while non-URM applicants cited family financial support as being beneficial.26 In addition, an increasing number of applicants take gap years for research, which pose additional costs for finances and resources. In contrast, mentorship and participation in pipeline/enrichment programs were factors associated with URM students matching into dermatology.26
Dermatology Residency and the Transition to Advanced Dermatology Fellowships
Similar to the transition from medical school into dermatology residency, URM dermatology residents are either not applying to fellowships or are not getting in. In the 2018-2019 academic year, there were no Black, Hispanic, Native Hawaiian/Pacific Islander, or American Indian/Alaska Native Mohs micrographic surgery and dermatologic oncology fellows.19 Similarly, there were no Black, Native Hawaiian/Pacific Islander, or American Indian/Alaska Native dermatopathology fellows. There were 4 (6%) Hispanic dermatopathology fellows.19
There also is marked underrepresentation of minority groups—and minimal growth over time—in the dermatology procedural subspecialty. Whereas the percentage of female Mohs surgeons increased considerably from 1985 to 2005 (12.7% to 40.9%, respectively), the percentage of URM Mohs surgeons remained steady from 4.2% to 4.6%, respectively, and remained at 4.5% in 2014.30
There are no available data on the race/ethnicity of fellowship applicants, as these demographic data for the application process have not been consistently or traditionally collected. The reasons why there are so few URM dermatology fellows is not known; whether this is due to a lack of mentorship or whether other factors lead to residents not applying for advanced training needs further study. Financial factors related to prolonged training, which include lower salaries and delayed loan repayment, may present barriers to applying to fellowships.
Lack of URM Academic Faculty in Dermatology
At the academic faculty level, URM representation continues to worsen. Lett et al31 found that there is declining racial and ethnic representation in clinical academic medicine relative to US census data for 16 US medical specialties, including dermatology, with growing underrepresentation of Black and Hispanic faculty at the associate professor and full professor levels and underrepresentation in all faculty ranks. From 1970 to 2018, URM faculty in dermatology only increased from 4.8% to 7.4%, respectively. Non-URM female and male faculty members increased by 13.8 and 10.8 faculty members per year, respectively, while URM female and male faculty members increased by 1.2 and 0.8 faculty members per year, respectively.32
Underrepresentation of minorities seen in dermatology faculty may result from clinical demands, minority taxation (defined as the extensive service requirements uniquely experienced by URM faculty to disproportionately serve as representatives on academic committees and to mentor URM students), and barriers to academic promotion, which are challenges uniquely encountered by URMs in academic dermatology.33 Increased clinical demand may result from the fact that URM physicians are more likely to care for underserved populations, those of lower socioeconomic status, non-English–speaking patients, those on Medicaid, and those who are uninsured, which may impact renumeration. Minority tax experienced by URM faculty includes mentoring URM medical students, providing cultural expertise to departments and institutions, and participating in community service projects and outreach programs. Specifically, many institutional committees require the participation of a URM member, resulting in URM faculty members experiencing higher committee service burden. Many, if not all, of these responsibilities often are not compensated through salary or academic promotion.
A Call to Action
There are several steps that can be taken to create a pathway to dermatology that is inclusive, flexible, and supportive of URMs.
• Increase early exposure to dermatology in medical school. Early exposure and mentorship opportunities are associated with higher rates of students pursuing specialty field careers.34 Increased early opportunities allow for URM students to consider and explore a career in dermatology; receive mentorship; and ensure that dermatology, including topics related to skin of color (SOC), is incorporated into their learning. The American Academy of Dermatology has contributed to these efforts by its presence at every national meeting of the Student National Medical Association and Latino Medical Student Association, as well as its involvement with Nth Dimensions, which offers various educational opportunities for URM medical students.
• Implement equitable grading and holistic review processes in medical school. Racial/ethnic differences in clinical grading and standardized test scores in medical school demonstrate why holistic review of dermatology residency applicants is needed and why other metrics such as USMLE scores and AΩA status should be de-emphasized or eliminated when evaluating candidates. To support equity, many medical schools have eliminated honors grading, and some schools have eliminated AΩA distinction.
• Increase diversity of dermatology residents and residency programs. Implicit bias training for a medical school admissions committee has been shown to increase diversity in medical school enrollment.35 Whether implicit bias training and other diversity training may benefit dermatology residency selection must be examined, including study of unintended consequences, such as reduced diversity, increased microaggressions toward minority colleagues, and the illusion of fairness.36-39 Increasing representation is not sufficient—creating inclusive residency training environments is a critical parallel aim. Prioritizing diversity in dermatology residency recruitment is imperative. Creating dermatology residency positions specifically for URM residents may be an important option, as done at the University of Pennsylvania (Philadelphia, Pennsylvania) and Duke University (Durham, North Carolina).
• Create effective programs for URM mentorship. Due to the competitive nature of dermatology residency, the need for mentors in dermatology is critically vital for URM medical students, especially those without a home dermatology program at their medical school. Further development of formal mentorship and pipeline programs is essential at both the local and national levels. Some national examples of these initiatives include diversity mentorship programs offered by the American Academy of Dermatology, Skin of Color Society, Women’s Dermatologic Society, and Student National Medical Association. Many institutional programs also offer invaluable opportunities, such as the summer research fellowship at the University of California, San Francisco (UCSF); visiting clerkship grants for URMs at the University of Pennsylvania (Philadelphia, Pennsylvania) and Johns Hopkins University (Baltimore, Maryland); and integrated programs, such as the Visiting Elective Scholarship Program at UCSF, which provides funding and faculty mentorship for URM students completing an away rotation at UCSF.
• Establish longitudinal skin-of-color curricula and increased opportunities for research. More robust SOC training may lead to an increasingly diverse workforce. It is important that medical student and dermatology resident and fellow education include training on SOC to ensure high-quality care to diverse patient populations, which also may enhance the knowledge of trainees, encourage clinical and research interest in this field, and reduce health care disparities. Increasing research opportunities and offering formalized longitudinal training in SOC as well as incorporating more diverse images in medical school education may foster greater interest in this field at a time when trainees are establishing their career interests. At present, there is considerable room for improvement. Nijhawan et al40 surveyed 63 dermatology chief residents and 41 program directors and found only 14.3% and 14.6%, respectively, reported having an expert who conducts clinic specializing in SOC. Only 52.4% and 65.9% reported having didactic sessions or lectures focused on SOC diseases, and 30.2% and 12.2% reported having a dedicated rotation for residents to gain experience in SOC.40 A more recent study showed that when faculty were asked to incorporate more SOC content into lectures, the most commonly identified barrier to implementation was a lack of SOC images.41 Additionally, there remains a paucity of published research on this topic, with SOC articles representing only 2.7% of the literature.42 These numbers demonstrate the continued need for a more inclusive and comprehensive curriculum in dermatology residency programs and more robust funding for SOC research.
• Recruit and support URM faculty. Increasing diversity in dermatology residency programs likely will increase the number of potential URMs pursuing additional fellowship training and academic dermatology with active career mentorship and support. In addition, promoting faculty retention by combatting the progressive loss of URMs at all faculty levels is paramount. Mentorship for URM physicians has been shown to play a key role in the decision to pursue academic medicine as well as academic productivity and job satisfaction.43,44 The visibility, cultural competency, clinical work, academic productivity, and mentorship efforts that URM faculty provide are essential to enhancing patient care, teaching diverse groups of learners, and recruiting more diverse trainees. Protected time to participate in professional development opportunities has been shown to improve recruitment and retention of URM faculty and offer additional opportunities for junior faculty to find mentors.35,36 Incentivizing clinical care of underserved populations also may augment financial stability for URM physicians who choose to care for these patients. Finally, diversity work and community service should be legitimized and count toward faculty promotion.
Conclusion
There are numerous factors that contribute to the leaky pipeline in dermatology (eFigure). Many challenges that are unique to the URM population disadvantage these students from entering medical school, applying to dermatology residency, matching into dermatology fellowships, pursuing and staying in faculty positions, and achieving faculty advancement into leadership positions. With each progressive step along this trajectory, there is less minority representation. All dermatologists, regardless of race/ethnicity, need to play an active role and must prioritize diversity, equity, and inclusion efforts at all levels of education and training for the betterment of the specialty.
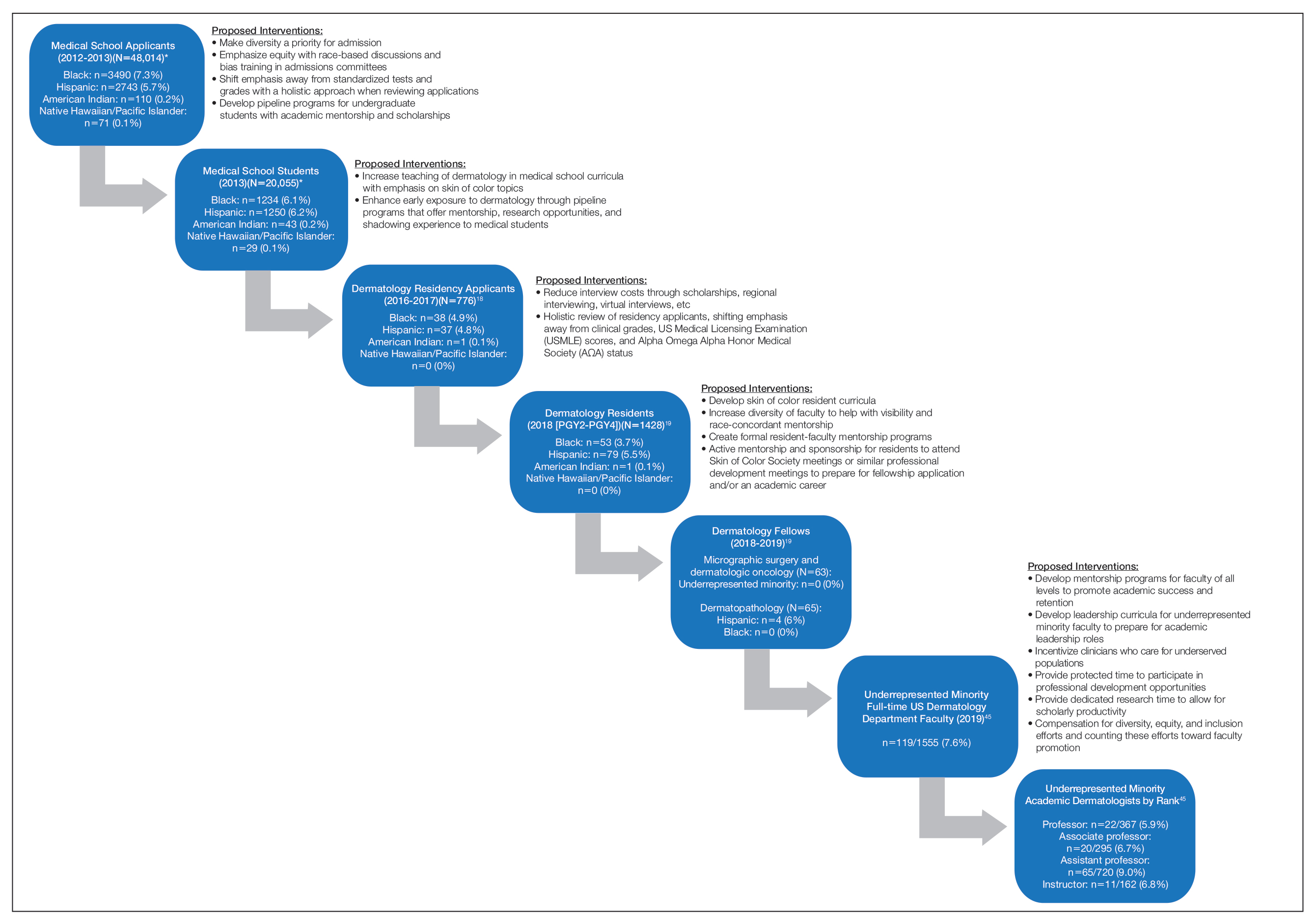
- Dixon G, Kind T, Wright J, et al. Factors that influence the choice of academic pediatrics by underrepresented minorities. Pediatrics. 2019;144:E20182759. doi:10.1542/peds.2018-2759
- Yehia BR, Cronholm PF, Wilson N, et al. Mentorship and pursuit of academic medicine careers: a mixed methods study of residents from diverse backgrounds. BMC Med Educ. 2014:14:2-26. doi:10.1186/1472-6920-14-26
- Saha S, Guiton G, Wimmers PF, et al. Student body racial and ethnic composition and diversity-related outcomes in US medical schools. JAMA. 2008;300:1135-1145. doi:10.1001/jama.300.10.1135
- Hsu DY, Gordon K, Silverberg JI. The patient burden of psoriasis in the United States. J Am Acad Dermatol. 2016;75:33-41. doi:10.1016/j.jaad.2016.03.048
- Silverberg JI. Racial and ethnic disparities in atopic dermatitis. Curr Dermatol Rep. 2015;4:44-48.
- Buster KJ, Sevens EI, Elmets CA. Dermatologic health disparities. Dermatol Clin. 2012;30:53-59. doi:10.1016/j.det.2011.08.002
- Barbieri JS, Shin DB, Wang S, et al. Association of race/ethnicity with differences in health care use and treatment for acne. JAMA Dermatol. 2020;156:312-319. doi:10.1001/jamadermatol.2019.4818
- Smedley BD, Stith AY, Colburn L, et al. The Right Thing To Do, The Smart Thing to Do: Enhancing Diversity in the Health Professions. National Academies Press; 2001.
- Association of American Medical Colleges. Minorities in medical education: fact and figures 2019. Accessed December 9, 2021. https://www.aamc.org/datareports/workforce/report/diversity-medicine-facts-and-figures-2019
- Liaison Committee on Medical Education (LCME) standards on diversity. University of South Florida Health website. Accessed December 9, 2021. https://health.usf.edu/~/media/Files/Medicine/MD%20Program/Diversity/LCMEStandardsonDiversity1.ashx?la=en
- Granstein RD, Cornelius L, Shinkai K. Diversity in dermatology—a call for action. JAMA Dermatol. 2017;153:499-500. doi:10.1001/jamadermatol.2017.0296
- Lett LA, Murdock HM, Orji W, et al. Trends in racial/ethnic representation among US medical students. JAMA Netw Open. 2019;2:e1910490. doi:10.1001/jamanetworkopen.2019.10490
- Association of American Medical Colleges. Altering the course: Black males in medicine. Published 2015. Accessed December 8, 2021. https://store.aamc.org/downloadable/download/sample/sample_id/84/
- Barr DA, Gonzalez ME, Wanat SF. The leaky pipeline: factors associated with early decline in interest in premedical studies among underrepresented minority undergraduate students. Acad Med. 2008;83:5:503-511. doi:10.1097/ACM.0b013e31816bda16
- Flores RL. The rising gap between rich and poor: a look at the persistence of educational disparities in the United States and why we should worry. Cogent Soc Sci. 2017;3:1323698.
- Jackson D. Why am I behind? an examination of low income and minority students’ preparedness for college. McNair Sch J. 2012;13:121-138.
- Rothstein R. The racial achievement gap, segregated schools, andsegregated neighborhoods: a constitutional insult. Race Soc Probl. 2015;7:21-30.
- Association of American Medical Colleges. Residency Applicants From US MD Granting Medical Schools to ACGME-Accredited Programs by Specialty and Race/Ethnicity. Association of American Medical Colleges; 2017.
- Brotherton SE, Etzel SL. Graduate medical education, 2018-2019. JAMA. 2019;322:996-1016. doi:10.1001/jama.2019.10155
- Barnes LA, Bae GH, Nambudiri V. Sex and racial/ethnic diversity of US medical students and their exposure to dermatology programs. JAMA Dermatol. 2019;155:490-491. doi:10.1001/jamadermatol.2018.5025
- Soliman YS, Rzepecki AK, Guzman AK. Understanding perceived barriers of minority medical students pursuing a career in dermatology. JAMA Dermatol. 2019;155:252-254. doi:10.1001/jamadermatol.2018.4813
- Low D, Pollack SW, Liao Z, et al. Racial/ethnic disparities in clinical grading in medical school. Teach Learn Med. 2019;31:487-496. doi:10.1080/10401334.2019.1597724
- Rubright JD, Jodoin M, Barone MA. Examining demographics, prior academic performance and United States medical licensing examination scores. Acad Med. 2019;94;364-370. doi:10.1097/ACM.0000000000002366
- Boatright D, Ross D, O’Connor P, et al. Racial disparities in medical student membership in the alpha omega honor society. JAMA Intern Med. 2017;177:659-665. doi:10.1001/jamainternmed.2016.9623
- Gorouhi F, Alikhan A, Rezaei A, et al. Dermatology residency selection criteria with an emphasis on program characteristics: a national program director survey [published online March 17, 2014]. Dermatol Res Pract. doi:10.1155/2014/692760
- Vasquez R, Jeong H, Florez-Pollack S, et al. What are the barriers faced by underrepresented minorities applying to dermatology? a qualitative cross-sectional study of applicants applying to a large dermatology residency program. J Am Acad Dermatol. 2020;83:1770-1773. doi:10.1016/j.jaad.2020.03.067
- Results of the 2019 NRMP applicant survey by preferred specialty and applicant type. National Resident Matching Program website. Published July 2019. Accessed December 8, 2021. https://www.nrmp.org/wp-content/uploads/2019/06/Applicant-Survey-Report-2019.pdf
- Mansouri B, Walker GD, Mitchell J, et al. The cost of applying to dermatology residency: 2014 data estimates. J Am Acad Dermatol. 2016;74:754-756. doi:10.1016/j.jaad.2015.10.049
- Polacco MA, Lally J, Walls A, et al. Digging into debt: the financial burden associated with the otolaryngology match. Otolaryngol Head Neck Surg. 2017;12:1091-1096. doi:10.1177/0194599816686538
- Feng H, Feng PW, Geronemus RG. Diversity in the US Mohs micrographic surgery workforce. Dermatol Surg. 2020:46:1451-1455. doi:10.1097/DSS.0000000000002080
- Lett LA, Orji WU, Sebro R. Declining racial and ethnic representation in clinical academic medicine: a longitudinal study of 16 US medical specialties. PLoS ONE. 2018;13:e0207274. doi:10.1371/journal.pone.020727432. Xierali IM, Nivet MA, Pandya AG. US Dermatology department faculty diversity trends by sex and underrepresented-in-medicine status, 1970-2018. JAMA Dermatol. 2020;156:280-287. doi:10.1001/jamadermatol.2019.4297
- Okoye GA. Supporting underrepresented minority women in academic dermatology. Intl J Womens Dermatol. 2020;6:57-60. doi:10.1016/j.ijwd.2019.09.009
- Bernstein J, Dicaprio MR, Mehta S. The relationship between required medical school instruction in musculoskeletal medicine and application rates to orthopaedic surgery residency programs. J Bone Joint Surg Am. 2004;86:2335-2338. doi:10.2106/00004623-200410000-00031
- Capers Q, Clinchot D, McDougle L, et al. Implicit racial bias in medical school admissions. Acad Med. 2017;92:365-369. doi:10.1097/ACM.0000000000001388
- Dobbin F, Kalev A. Why diversity programs fail. Harvard Business Rev. 2016;52-60. Accessed December 8, 2021. https://hbr.org/2016/07/why-diversity-programs-fail
- Kalev A, Dobbin F, Kelly E. Best practices or best guesses? assessing the efficacy of corporate affirmative action and diversity policies. Am Sociol Rev. 2006;71:589-617.
- Sanchez JI, Medkik N. The effects of diversity awareness training on differential treatment. Group Organ Manag. 2004;29:517-536.
- Kaiser CR, Major B, Jurcevic I, et al. Presumed fair: ironic effects of organizational diversity structures. J Pers Soc Psychol. 2013;104:504-519. doi:10.1037/a0030838
- Nijhawan RI, Jacob SE, Woolery-Lloyd H. Skin of color education in dermatology residency programs: does residency training reflect the changing demographics of the United States? J Am Acad Dermatol. 2008;59:615-617.
- Jia JL, Gordon JS, Lester JC, et al. Integrating skin of color and sexual and gender minority content into dermatology residency curricula: a prospective program initiative [published online April 16, 2021]. J Am Acad Dermatol. doi:10.1016/j.jaad.2021.04.018
- Amuzie AU, Lia JL, Taylor SC, et al. Skin of color article representation in dermatology literature 2009-2019: higher citation counts and opportunities for inclusion [published online March 24, 2021]. J Am Acad Dermatol. doi:10.1016/j.jaad.2021.03.063
- Beech BM, Calles-Escandon J, Hairston KC, et al. Mentoring programs for underrepresented minority faculty in academic medical center: a systematic review of the literature. Acad Med. 2013;88:541-549. doi:10.1097/ACM.0b013e31828589e3
- Daley S, Wingard DL, Reznik V. Improving the retention of underrepresented minority faculty in academic medicine. J Natl Med Assoc. 2006;98:1435-1440. doi:10.1016/s0027-9684(15)31449-8
- Association of American Medical Colleges. US medical school faculty by sex, race/ethnicity, rank, and department, 2019. Published December 31, 2019. Accessed December 20, 2021. https://www.aamc.org/media/8476/download?attachment
With a majority-minority population expected in the United States by 2044, improving diversity and cultural competency in the dermatology workforce is now more important than ever. A more diverse workforce increases the cultural competence of all providers, provides greater opportunities for mentorship and sponsorship of underrepresented minority (URM) trainees, establishes a more inclusive environment for learners, and enhances the knowledge and productivity of the workforce.1-3 Additionally, it is imperative to address clinical care disparities seen in minority patients in dermatology, including treatment of skin cancer, psoriasis, acne, atopic dermatitis, and other diseases.4-7
Despite the attention that has been devoted to improving diversity in medicine,8-10 dermatology remains one of the least diverse specialties, prompting additional calls to action within the field.11 Why does the lack of diversity still exist in dermatology, and what is the path to correcting this problem? In this article, we review the evidence of diversity barriers at different stages of medical education training that may impede academic advancement for minority learners pursuing careers in dermatology.
Undergraduate Medical Education
The term leaky pipeline refers to the progressive decline in the number of URMs along a given career path, including in dermatology. The Association of American Medical Colleges defines URMs as racial/ethnic populations that are “underrepresented in the medical profession relative to their numbers in the general population.”9 The first leak in the pipeline is that URMs are not applying to medical school. From 2002 and 2017, rates of both application and matriculation to medical school were lower by 30% to 70% in URM groups compared to White students, including Hispanic, Black, and American Indian/Alaska Native students.12,13 The decision not to apply to medical school was greater in URM undergraduate students irrespective of scholastic ability as measured by SAT scores.14
A striking statistic is that the number of Black men matriculating into medical school in 2014 was less than it was in 1978 despite the increase in the number of US medical schools and efforts to recruit more diverse student populations. The Association of American Medical Colleges identified potential reasons for this decline, including poor early education, lack of mentorship, negative perceptions of Black men due to racial stereotypes, and lack of financial and academic resources to support the application process.8,13,15-17 Implicit racial bias by admission committees also may play a role.
Medical School Matriculation and Applying to Dermatology Residency
There is greater representation of URM students in medical school than in dermatology residency, which means URM students are either not applying to dermatology programs or they are not matching into the specialty. In the Electronic Residency Application Service’s 2016-2017 application cycle (N=776), there were 76 (9.8%) URM dermatology residency applicants.18 In 2018, there was a notable decline in representation of Black students among residency applicants (4.9%) to matched residents (3.7%), and there were only 133 (9.3%) URM dermatology residents in total (PGY2-PGY4 classes).19 The lack of exposure to medical subspecialties and the recommendation by medical schools for URM medical students to pursue careers in primary care have been cited as reasons that these students may not apply to residency programs in specialty care.20,21 The presence of an Accreditation Council for Graduate Medical Education dermatology residency program, fellowships, and dermatology interest groups at their medical schools correlated with higher proportions of URM students applying to dermatology programs.20
Underrepresented minority students face critical challenges during medical school, including receiving lower grades in both standardized and school-designated assessments and clerkship grades.21,22 A 2019 National Board of Medical Examiners study found that Hispanic and Black test takers scored 12.1 and 16.6 points lower than White men, respectively, on the
A recent cross-sectional study showed that lack of equitable resources, lack of support, financial constrictions, and lack of group identity were 4 barriers to URM students matching into dermatology.26 Dermatology is a competitive specialty with the highest median Electronic Residency Application Service applications submitted per US applicant (n=90)27 and an approximate total cost per US applicant of $10,781.28,29 Disadvantaged URM applicants noted relying on loans while non-URM applicants cited family financial support as being beneficial.26 In addition, an increasing number of applicants take gap years for research, which pose additional costs for finances and resources. In contrast, mentorship and participation in pipeline/enrichment programs were factors associated with URM students matching into dermatology.26
Dermatology Residency and the Transition to Advanced Dermatology Fellowships
Similar to the transition from medical school into dermatology residency, URM dermatology residents are either not applying to fellowships or are not getting in. In the 2018-2019 academic year, there were no Black, Hispanic, Native Hawaiian/Pacific Islander, or American Indian/Alaska Native Mohs micrographic surgery and dermatologic oncology fellows.19 Similarly, there were no Black, Native Hawaiian/Pacific Islander, or American Indian/Alaska Native dermatopathology fellows. There were 4 (6%) Hispanic dermatopathology fellows.19
There also is marked underrepresentation of minority groups—and minimal growth over time—in the dermatology procedural subspecialty. Whereas the percentage of female Mohs surgeons increased considerably from 1985 to 2005 (12.7% to 40.9%, respectively), the percentage of URM Mohs surgeons remained steady from 4.2% to 4.6%, respectively, and remained at 4.5% in 2014.30
There are no available data on the race/ethnicity of fellowship applicants, as these demographic data for the application process have not been consistently or traditionally collected. The reasons why there are so few URM dermatology fellows is not known; whether this is due to a lack of mentorship or whether other factors lead to residents not applying for advanced training needs further study. Financial factors related to prolonged training, which include lower salaries and delayed loan repayment, may present barriers to applying to fellowships.
Lack of URM Academic Faculty in Dermatology
At the academic faculty level, URM representation continues to worsen. Lett et al31 found that there is declining racial and ethnic representation in clinical academic medicine relative to US census data for 16 US medical specialties, including dermatology, with growing underrepresentation of Black and Hispanic faculty at the associate professor and full professor levels and underrepresentation in all faculty ranks. From 1970 to 2018, URM faculty in dermatology only increased from 4.8% to 7.4%, respectively. Non-URM female and male faculty members increased by 13.8 and 10.8 faculty members per year, respectively, while URM female and male faculty members increased by 1.2 and 0.8 faculty members per year, respectively.32
Underrepresentation of minorities seen in dermatology faculty may result from clinical demands, minority taxation (defined as the extensive service requirements uniquely experienced by URM faculty to disproportionately serve as representatives on academic committees and to mentor URM students), and barriers to academic promotion, which are challenges uniquely encountered by URMs in academic dermatology.33 Increased clinical demand may result from the fact that URM physicians are more likely to care for underserved populations, those of lower socioeconomic status, non-English–speaking patients, those on Medicaid, and those who are uninsured, which may impact renumeration. Minority tax experienced by URM faculty includes mentoring URM medical students, providing cultural expertise to departments and institutions, and participating in community service projects and outreach programs. Specifically, many institutional committees require the participation of a URM member, resulting in URM faculty members experiencing higher committee service burden. Many, if not all, of these responsibilities often are not compensated through salary or academic promotion.
A Call to Action
There are several steps that can be taken to create a pathway to dermatology that is inclusive, flexible, and supportive of URMs.
• Increase early exposure to dermatology in medical school. Early exposure and mentorship opportunities are associated with higher rates of students pursuing specialty field careers.34 Increased early opportunities allow for URM students to consider and explore a career in dermatology; receive mentorship; and ensure that dermatology, including topics related to skin of color (SOC), is incorporated into their learning. The American Academy of Dermatology has contributed to these efforts by its presence at every national meeting of the Student National Medical Association and Latino Medical Student Association, as well as its involvement with Nth Dimensions, which offers various educational opportunities for URM medical students.
• Implement equitable grading and holistic review processes in medical school. Racial/ethnic differences in clinical grading and standardized test scores in medical school demonstrate why holistic review of dermatology residency applicants is needed and why other metrics such as USMLE scores and AΩA status should be de-emphasized or eliminated when evaluating candidates. To support equity, many medical schools have eliminated honors grading, and some schools have eliminated AΩA distinction.
• Increase diversity of dermatology residents and residency programs. Implicit bias training for a medical school admissions committee has been shown to increase diversity in medical school enrollment.35 Whether implicit bias training and other diversity training may benefit dermatology residency selection must be examined, including study of unintended consequences, such as reduced diversity, increased microaggressions toward minority colleagues, and the illusion of fairness.36-39 Increasing representation is not sufficient—creating inclusive residency training environments is a critical parallel aim. Prioritizing diversity in dermatology residency recruitment is imperative. Creating dermatology residency positions specifically for URM residents may be an important option, as done at the University of Pennsylvania (Philadelphia, Pennsylvania) and Duke University (Durham, North Carolina).
• Create effective programs for URM mentorship. Due to the competitive nature of dermatology residency, the need for mentors in dermatology is critically vital for URM medical students, especially those without a home dermatology program at their medical school. Further development of formal mentorship and pipeline programs is essential at both the local and national levels. Some national examples of these initiatives include diversity mentorship programs offered by the American Academy of Dermatology, Skin of Color Society, Women’s Dermatologic Society, and Student National Medical Association. Many institutional programs also offer invaluable opportunities, such as the summer research fellowship at the University of California, San Francisco (UCSF); visiting clerkship grants for URMs at the University of Pennsylvania (Philadelphia, Pennsylvania) and Johns Hopkins University (Baltimore, Maryland); and integrated programs, such as the Visiting Elective Scholarship Program at UCSF, which provides funding and faculty mentorship for URM students completing an away rotation at UCSF.
• Establish longitudinal skin-of-color curricula and increased opportunities for research. More robust SOC training may lead to an increasingly diverse workforce. It is important that medical student and dermatology resident and fellow education include training on SOC to ensure high-quality care to diverse patient populations, which also may enhance the knowledge of trainees, encourage clinical and research interest in this field, and reduce health care disparities. Increasing research opportunities and offering formalized longitudinal training in SOC as well as incorporating more diverse images in medical school education may foster greater interest in this field at a time when trainees are establishing their career interests. At present, there is considerable room for improvement. Nijhawan et al40 surveyed 63 dermatology chief residents and 41 program directors and found only 14.3% and 14.6%, respectively, reported having an expert who conducts clinic specializing in SOC. Only 52.4% and 65.9% reported having didactic sessions or lectures focused on SOC diseases, and 30.2% and 12.2% reported having a dedicated rotation for residents to gain experience in SOC.40 A more recent study showed that when faculty were asked to incorporate more SOC content into lectures, the most commonly identified barrier to implementation was a lack of SOC images.41 Additionally, there remains a paucity of published research on this topic, with SOC articles representing only 2.7% of the literature.42 These numbers demonstrate the continued need for a more inclusive and comprehensive curriculum in dermatology residency programs and more robust funding for SOC research.
• Recruit and support URM faculty. Increasing diversity in dermatology residency programs likely will increase the number of potential URMs pursuing additional fellowship training and academic dermatology with active career mentorship and support. In addition, promoting faculty retention by combatting the progressive loss of URMs at all faculty levels is paramount. Mentorship for URM physicians has been shown to play a key role in the decision to pursue academic medicine as well as academic productivity and job satisfaction.43,44 The visibility, cultural competency, clinical work, academic productivity, and mentorship efforts that URM faculty provide are essential to enhancing patient care, teaching diverse groups of learners, and recruiting more diverse trainees. Protected time to participate in professional development opportunities has been shown to improve recruitment and retention of URM faculty and offer additional opportunities for junior faculty to find mentors.35,36 Incentivizing clinical care of underserved populations also may augment financial stability for URM physicians who choose to care for these patients. Finally, diversity work and community service should be legitimized and count toward faculty promotion.
Conclusion
There are numerous factors that contribute to the leaky pipeline in dermatology (eFigure). Many challenges that are unique to the URM population disadvantage these students from entering medical school, applying to dermatology residency, matching into dermatology fellowships, pursuing and staying in faculty positions, and achieving faculty advancement into leadership positions. With each progressive step along this trajectory, there is less minority representation. All dermatologists, regardless of race/ethnicity, need to play an active role and must prioritize diversity, equity, and inclusion efforts at all levels of education and training for the betterment of the specialty.

With a majority-minority population expected in the United States by 2044, improving diversity and cultural competency in the dermatology workforce is now more important than ever. A more diverse workforce increases the cultural competence of all providers, provides greater opportunities for mentorship and sponsorship of underrepresented minority (URM) trainees, establishes a more inclusive environment for learners, and enhances the knowledge and productivity of the workforce.1-3 Additionally, it is imperative to address clinical care disparities seen in minority patients in dermatology, including treatment of skin cancer, psoriasis, acne, atopic dermatitis, and other diseases.4-7
Despite the attention that has been devoted to improving diversity in medicine,8-10 dermatology remains one of the least diverse specialties, prompting additional calls to action within the field.11 Why does the lack of diversity still exist in dermatology, and what is the path to correcting this problem? In this article, we review the evidence of diversity barriers at different stages of medical education training that may impede academic advancement for minority learners pursuing careers in dermatology.
Undergraduate Medical Education
The term leaky pipeline refers to the progressive decline in the number of URMs along a given career path, including in dermatology. The Association of American Medical Colleges defines URMs as racial/ethnic populations that are “underrepresented in the medical profession relative to their numbers in the general population.”9 The first leak in the pipeline is that URMs are not applying to medical school. From 2002 and 2017, rates of both application and matriculation to medical school were lower by 30% to 70% in URM groups compared to White students, including Hispanic, Black, and American Indian/Alaska Native students.12,13 The decision not to apply to medical school was greater in URM undergraduate students irrespective of scholastic ability as measured by SAT scores.14
A striking statistic is that the number of Black men matriculating into medical school in 2014 was less than it was in 1978 despite the increase in the number of US medical schools and efforts to recruit more diverse student populations. The Association of American Medical Colleges identified potential reasons for this decline, including poor early education, lack of mentorship, negative perceptions of Black men due to racial stereotypes, and lack of financial and academic resources to support the application process.8,13,15-17 Implicit racial bias by admission committees also may play a role.
Medical School Matriculation and Applying to Dermatology Residency
There is greater representation of URM students in medical school than in dermatology residency, which means URM students are either not applying to dermatology programs or they are not matching into the specialty. In the Electronic Residency Application Service’s 2016-2017 application cycle (N=776), there were 76 (9.8%) URM dermatology residency applicants.18 In 2018, there was a notable decline in representation of Black students among residency applicants (4.9%) to matched residents (3.7%), and there were only 133 (9.3%) URM dermatology residents in total (PGY2-PGY4 classes).19 The lack of exposure to medical subspecialties and the recommendation by medical schools for URM medical students to pursue careers in primary care have been cited as reasons that these students may not apply to residency programs in specialty care.20,21 The presence of an Accreditation Council for Graduate Medical Education dermatology residency program, fellowships, and dermatology interest groups at their medical schools correlated with higher proportions of URM students applying to dermatology programs.20
Underrepresented minority students face critical challenges during medical school, including receiving lower grades in both standardized and school-designated assessments and clerkship grades.21,22 A 2019 National Board of Medical Examiners study found that Hispanic and Black test takers scored 12.1 and 16.6 points lower than White men, respectively, on the
A recent cross-sectional study showed that lack of equitable resources, lack of support, financial constrictions, and lack of group identity were 4 barriers to URM students matching into dermatology.26 Dermatology is a competitive specialty with the highest median Electronic Residency Application Service applications submitted per US applicant (n=90)27 and an approximate total cost per US applicant of $10,781.28,29 Disadvantaged URM applicants noted relying on loans while non-URM applicants cited family financial support as being beneficial.26 In addition, an increasing number of applicants take gap years for research, which pose additional costs for finances and resources. In contrast, mentorship and participation in pipeline/enrichment programs were factors associated with URM students matching into dermatology.26
Dermatology Residency and the Transition to Advanced Dermatology Fellowships
Similar to the transition from medical school into dermatology residency, URM dermatology residents are either not applying to fellowships or are not getting in. In the 2018-2019 academic year, there were no Black, Hispanic, Native Hawaiian/Pacific Islander, or American Indian/Alaska Native Mohs micrographic surgery and dermatologic oncology fellows.19 Similarly, there were no Black, Native Hawaiian/Pacific Islander, or American Indian/Alaska Native dermatopathology fellows. There were 4 (6%) Hispanic dermatopathology fellows.19
There also is marked underrepresentation of minority groups—and minimal growth over time—in the dermatology procedural subspecialty. Whereas the percentage of female Mohs surgeons increased considerably from 1985 to 2005 (12.7% to 40.9%, respectively), the percentage of URM Mohs surgeons remained steady from 4.2% to 4.6%, respectively, and remained at 4.5% in 2014.30
There are no available data on the race/ethnicity of fellowship applicants, as these demographic data for the application process have not been consistently or traditionally collected. The reasons why there are so few URM dermatology fellows is not known; whether this is due to a lack of mentorship or whether other factors lead to residents not applying for advanced training needs further study. Financial factors related to prolonged training, which include lower salaries and delayed loan repayment, may present barriers to applying to fellowships.
Lack of URM Academic Faculty in Dermatology
At the academic faculty level, URM representation continues to worsen. Lett et al31 found that there is declining racial and ethnic representation in clinical academic medicine relative to US census data for 16 US medical specialties, including dermatology, with growing underrepresentation of Black and Hispanic faculty at the associate professor and full professor levels and underrepresentation in all faculty ranks. From 1970 to 2018, URM faculty in dermatology only increased from 4.8% to 7.4%, respectively. Non-URM female and male faculty members increased by 13.8 and 10.8 faculty members per year, respectively, while URM female and male faculty members increased by 1.2 and 0.8 faculty members per year, respectively.32
Underrepresentation of minorities seen in dermatology faculty may result from clinical demands, minority taxation (defined as the extensive service requirements uniquely experienced by URM faculty to disproportionately serve as representatives on academic committees and to mentor URM students), and barriers to academic promotion, which are challenges uniquely encountered by URMs in academic dermatology.33 Increased clinical demand may result from the fact that URM physicians are more likely to care for underserved populations, those of lower socioeconomic status, non-English–speaking patients, those on Medicaid, and those who are uninsured, which may impact renumeration. Minority tax experienced by URM faculty includes mentoring URM medical students, providing cultural expertise to departments and institutions, and participating in community service projects and outreach programs. Specifically, many institutional committees require the participation of a URM member, resulting in URM faculty members experiencing higher committee service burden. Many, if not all, of these responsibilities often are not compensated through salary or academic promotion.
A Call to Action
There are several steps that can be taken to create a pathway to dermatology that is inclusive, flexible, and supportive of URMs.
• Increase early exposure to dermatology in medical school. Early exposure and mentorship opportunities are associated with higher rates of students pursuing specialty field careers.34 Increased early opportunities allow for URM students to consider and explore a career in dermatology; receive mentorship; and ensure that dermatology, including topics related to skin of color (SOC), is incorporated into their learning. The American Academy of Dermatology has contributed to these efforts by its presence at every national meeting of the Student National Medical Association and Latino Medical Student Association, as well as its involvement with Nth Dimensions, which offers various educational opportunities for URM medical students.
• Implement equitable grading and holistic review processes in medical school. Racial/ethnic differences in clinical grading and standardized test scores in medical school demonstrate why holistic review of dermatology residency applicants is needed and why other metrics such as USMLE scores and AΩA status should be de-emphasized or eliminated when evaluating candidates. To support equity, many medical schools have eliminated honors grading, and some schools have eliminated AΩA distinction.
• Increase diversity of dermatology residents and residency programs. Implicit bias training for a medical school admissions committee has been shown to increase diversity in medical school enrollment.35 Whether implicit bias training and other diversity training may benefit dermatology residency selection must be examined, including study of unintended consequences, such as reduced diversity, increased microaggressions toward minority colleagues, and the illusion of fairness.36-39 Increasing representation is not sufficient—creating inclusive residency training environments is a critical parallel aim. Prioritizing diversity in dermatology residency recruitment is imperative. Creating dermatology residency positions specifically for URM residents may be an important option, as done at the University of Pennsylvania (Philadelphia, Pennsylvania) and Duke University (Durham, North Carolina).
• Create effective programs for URM mentorship. Due to the competitive nature of dermatology residency, the need for mentors in dermatology is critically vital for URM medical students, especially those without a home dermatology program at their medical school. Further development of formal mentorship and pipeline programs is essential at both the local and national levels. Some national examples of these initiatives include diversity mentorship programs offered by the American Academy of Dermatology, Skin of Color Society, Women’s Dermatologic Society, and Student National Medical Association. Many institutional programs also offer invaluable opportunities, such as the summer research fellowship at the University of California, San Francisco (UCSF); visiting clerkship grants for URMs at the University of Pennsylvania (Philadelphia, Pennsylvania) and Johns Hopkins University (Baltimore, Maryland); and integrated programs, such as the Visiting Elective Scholarship Program at UCSF, which provides funding and faculty mentorship for URM students completing an away rotation at UCSF.
• Establish longitudinal skin-of-color curricula and increased opportunities for research. More robust SOC training may lead to an increasingly diverse workforce. It is important that medical student and dermatology resident and fellow education include training on SOC to ensure high-quality care to diverse patient populations, which also may enhance the knowledge of trainees, encourage clinical and research interest in this field, and reduce health care disparities. Increasing research opportunities and offering formalized longitudinal training in SOC as well as incorporating more diverse images in medical school education may foster greater interest in this field at a time when trainees are establishing their career interests. At present, there is considerable room for improvement. Nijhawan et al40 surveyed 63 dermatology chief residents and 41 program directors and found only 14.3% and 14.6%, respectively, reported having an expert who conducts clinic specializing in SOC. Only 52.4% and 65.9% reported having didactic sessions or lectures focused on SOC diseases, and 30.2% and 12.2% reported having a dedicated rotation for residents to gain experience in SOC.40 A more recent study showed that when faculty were asked to incorporate more SOC content into lectures, the most commonly identified barrier to implementation was a lack of SOC images.41 Additionally, there remains a paucity of published research on this topic, with SOC articles representing only 2.7% of the literature.42 These numbers demonstrate the continued need for a more inclusive and comprehensive curriculum in dermatology residency programs and more robust funding for SOC research.
• Recruit and support URM faculty. Increasing diversity in dermatology residency programs likely will increase the number of potential URMs pursuing additional fellowship training and academic dermatology with active career mentorship and support. In addition, promoting faculty retention by combatting the progressive loss of URMs at all faculty levels is paramount. Mentorship for URM physicians has been shown to play a key role in the decision to pursue academic medicine as well as academic productivity and job satisfaction.43,44 The visibility, cultural competency, clinical work, academic productivity, and mentorship efforts that URM faculty provide are essential to enhancing patient care, teaching diverse groups of learners, and recruiting more diverse trainees. Protected time to participate in professional development opportunities has been shown to improve recruitment and retention of URM faculty and offer additional opportunities for junior faculty to find mentors.35,36 Incentivizing clinical care of underserved populations also may augment financial stability for URM physicians who choose to care for these patients. Finally, diversity work and community service should be legitimized and count toward faculty promotion.
Conclusion
There are numerous factors that contribute to the leaky pipeline in dermatology (eFigure). Many challenges that are unique to the URM population disadvantage these students from entering medical school, applying to dermatology residency, matching into dermatology fellowships, pursuing and staying in faculty positions, and achieving faculty advancement into leadership positions. With each progressive step along this trajectory, there is less minority representation. All dermatologists, regardless of race/ethnicity, need to play an active role and must prioritize diversity, equity, and inclusion efforts at all levels of education and training for the betterment of the specialty.

- Dixon G, Kind T, Wright J, et al. Factors that influence the choice of academic pediatrics by underrepresented minorities. Pediatrics. 2019;144:E20182759. doi:10.1542/peds.2018-2759
- Yehia BR, Cronholm PF, Wilson N, et al. Mentorship and pursuit of academic medicine careers: a mixed methods study of residents from diverse backgrounds. BMC Med Educ. 2014:14:2-26. doi:10.1186/1472-6920-14-26
- Saha S, Guiton G, Wimmers PF, et al. Student body racial and ethnic composition and diversity-related outcomes in US medical schools. JAMA. 2008;300:1135-1145. doi:10.1001/jama.300.10.1135
- Hsu DY, Gordon K, Silverberg JI. The patient burden of psoriasis in the United States. J Am Acad Dermatol. 2016;75:33-41. doi:10.1016/j.jaad.2016.03.048
- Silverberg JI. Racial and ethnic disparities in atopic dermatitis. Curr Dermatol Rep. 2015;4:44-48.
- Buster KJ, Sevens EI, Elmets CA. Dermatologic health disparities. Dermatol Clin. 2012;30:53-59. doi:10.1016/j.det.2011.08.002
- Barbieri JS, Shin DB, Wang S, et al. Association of race/ethnicity with differences in health care use and treatment for acne. JAMA Dermatol. 2020;156:312-319. doi:10.1001/jamadermatol.2019.4818
- Smedley BD, Stith AY, Colburn L, et al. The Right Thing To Do, The Smart Thing to Do: Enhancing Diversity in the Health Professions. National Academies Press; 2001.
- Association of American Medical Colleges. Minorities in medical education: fact and figures 2019. Accessed December 9, 2021. https://www.aamc.org/datareports/workforce/report/diversity-medicine-facts-and-figures-2019
- Liaison Committee on Medical Education (LCME) standards on diversity. University of South Florida Health website. Accessed December 9, 2021. https://health.usf.edu/~/media/Files/Medicine/MD%20Program/Diversity/LCMEStandardsonDiversity1.ashx?la=en
- Granstein RD, Cornelius L, Shinkai K. Diversity in dermatology—a call for action. JAMA Dermatol. 2017;153:499-500. doi:10.1001/jamadermatol.2017.0296
- Lett LA, Murdock HM, Orji W, et al. Trends in racial/ethnic representation among US medical students. JAMA Netw Open. 2019;2:e1910490. doi:10.1001/jamanetworkopen.2019.10490
- Association of American Medical Colleges. Altering the course: Black males in medicine. Published 2015. Accessed December 8, 2021. https://store.aamc.org/downloadable/download/sample/sample_id/84/
- Barr DA, Gonzalez ME, Wanat SF. The leaky pipeline: factors associated with early decline in interest in premedical studies among underrepresented minority undergraduate students. Acad Med. 2008;83:5:503-511. doi:10.1097/ACM.0b013e31816bda16
- Flores RL. The rising gap between rich and poor: a look at the persistence of educational disparities in the United States and why we should worry. Cogent Soc Sci. 2017;3:1323698.
- Jackson D. Why am I behind? an examination of low income and minority students’ preparedness for college. McNair Sch J. 2012;13:121-138.
- Rothstein R. The racial achievement gap, segregated schools, andsegregated neighborhoods: a constitutional insult. Race Soc Probl. 2015;7:21-30.
- Association of American Medical Colleges. Residency Applicants From US MD Granting Medical Schools to ACGME-Accredited Programs by Specialty and Race/Ethnicity. Association of American Medical Colleges; 2017.
- Brotherton SE, Etzel SL. Graduate medical education, 2018-2019. JAMA. 2019;322:996-1016. doi:10.1001/jama.2019.10155
- Barnes LA, Bae GH, Nambudiri V. Sex and racial/ethnic diversity of US medical students and their exposure to dermatology programs. JAMA Dermatol. 2019;155:490-491. doi:10.1001/jamadermatol.2018.5025
- Soliman YS, Rzepecki AK, Guzman AK. Understanding perceived barriers of minority medical students pursuing a career in dermatology. JAMA Dermatol. 2019;155:252-254. doi:10.1001/jamadermatol.2018.4813
- Low D, Pollack SW, Liao Z, et al. Racial/ethnic disparities in clinical grading in medical school. Teach Learn Med. 2019;31:487-496. doi:10.1080/10401334.2019.1597724
- Rubright JD, Jodoin M, Barone MA. Examining demographics, prior academic performance and United States medical licensing examination scores. Acad Med. 2019;94;364-370. doi:10.1097/ACM.0000000000002366
- Boatright D, Ross D, O’Connor P, et al. Racial disparities in medical student membership in the alpha omega honor society. JAMA Intern Med. 2017;177:659-665. doi:10.1001/jamainternmed.2016.9623
- Gorouhi F, Alikhan A, Rezaei A, et al. Dermatology residency selection criteria with an emphasis on program characteristics: a national program director survey [published online March 17, 2014]. Dermatol Res Pract. doi:10.1155/2014/692760
- Vasquez R, Jeong H, Florez-Pollack S, et al. What are the barriers faced by underrepresented minorities applying to dermatology? a qualitative cross-sectional study of applicants applying to a large dermatology residency program. J Am Acad Dermatol. 2020;83:1770-1773. doi:10.1016/j.jaad.2020.03.067
- Results of the 2019 NRMP applicant survey by preferred specialty and applicant type. National Resident Matching Program website. Published July 2019. Accessed December 8, 2021. https://www.nrmp.org/wp-content/uploads/2019/06/Applicant-Survey-Report-2019.pdf
- Mansouri B, Walker GD, Mitchell J, et al. The cost of applying to dermatology residency: 2014 data estimates. J Am Acad Dermatol. 2016;74:754-756. doi:10.1016/j.jaad.2015.10.049
- Polacco MA, Lally J, Walls A, et al. Digging into debt: the financial burden associated with the otolaryngology match. Otolaryngol Head Neck Surg. 2017;12:1091-1096. doi:10.1177/0194599816686538
- Feng H, Feng PW, Geronemus RG. Diversity in the US Mohs micrographic surgery workforce. Dermatol Surg. 2020:46:1451-1455. doi:10.1097/DSS.0000000000002080
- Lett LA, Orji WU, Sebro R. Declining racial and ethnic representation in clinical academic medicine: a longitudinal study of 16 US medical specialties. PLoS ONE. 2018;13:e0207274. doi:10.1371/journal.pone.020727432. Xierali IM, Nivet MA, Pandya AG. US Dermatology department faculty diversity trends by sex and underrepresented-in-medicine status, 1970-2018. JAMA Dermatol. 2020;156:280-287. doi:10.1001/jamadermatol.2019.4297
- Okoye GA. Supporting underrepresented minority women in academic dermatology. Intl J Womens Dermatol. 2020;6:57-60. doi:10.1016/j.ijwd.2019.09.009
- Bernstein J, Dicaprio MR, Mehta S. The relationship between required medical school instruction in musculoskeletal medicine and application rates to orthopaedic surgery residency programs. J Bone Joint Surg Am. 2004;86:2335-2338. doi:10.2106/00004623-200410000-00031
- Capers Q, Clinchot D, McDougle L, et al. Implicit racial bias in medical school admissions. Acad Med. 2017;92:365-369. doi:10.1097/ACM.0000000000001388
- Dobbin F, Kalev A. Why diversity programs fail. Harvard Business Rev. 2016;52-60. Accessed December 8, 2021. https://hbr.org/2016/07/why-diversity-programs-fail
- Kalev A, Dobbin F, Kelly E. Best practices or best guesses? assessing the efficacy of corporate affirmative action and diversity policies. Am Sociol Rev. 2006;71:589-617.
- Sanchez JI, Medkik N. The effects of diversity awareness training on differential treatment. Group Organ Manag. 2004;29:517-536.
- Kaiser CR, Major B, Jurcevic I, et al. Presumed fair: ironic effects of organizational diversity structures. J Pers Soc Psychol. 2013;104:504-519. doi:10.1037/a0030838
- Nijhawan RI, Jacob SE, Woolery-Lloyd H. Skin of color education in dermatology residency programs: does residency training reflect the changing demographics of the United States? J Am Acad Dermatol. 2008;59:615-617.
- Jia JL, Gordon JS, Lester JC, et al. Integrating skin of color and sexual and gender minority content into dermatology residency curricula: a prospective program initiative [published online April 16, 2021]. J Am Acad Dermatol. doi:10.1016/j.jaad.2021.04.018
- Amuzie AU, Lia JL, Taylor SC, et al. Skin of color article representation in dermatology literature 2009-2019: higher citation counts and opportunities for inclusion [published online March 24, 2021]. J Am Acad Dermatol. doi:10.1016/j.jaad.2021.03.063
- Beech BM, Calles-Escandon J, Hairston KC, et al. Mentoring programs for underrepresented minority faculty in academic medical center: a systematic review of the literature. Acad Med. 2013;88:541-549. doi:10.1097/ACM.0b013e31828589e3
- Daley S, Wingard DL, Reznik V. Improving the retention of underrepresented minority faculty in academic medicine. J Natl Med Assoc. 2006;98:1435-1440. doi:10.1016/s0027-9684(15)31449-8
- Association of American Medical Colleges. US medical school faculty by sex, race/ethnicity, rank, and department, 2019. Published December 31, 2019. Accessed December 20, 2021. https://www.aamc.org/media/8476/download?attachment
- Dixon G, Kind T, Wright J, et al. Factors that influence the choice of academic pediatrics by underrepresented minorities. Pediatrics. 2019;144:E20182759. doi:10.1542/peds.2018-2759
- Yehia BR, Cronholm PF, Wilson N, et al. Mentorship and pursuit of academic medicine careers: a mixed methods study of residents from diverse backgrounds. BMC Med Educ. 2014:14:2-26. doi:10.1186/1472-6920-14-26
- Saha S, Guiton G, Wimmers PF, et al. Student body racial and ethnic composition and diversity-related outcomes in US medical schools. JAMA. 2008;300:1135-1145. doi:10.1001/jama.300.10.1135
- Hsu DY, Gordon K, Silverberg JI. The patient burden of psoriasis in the United States. J Am Acad Dermatol. 2016;75:33-41. doi:10.1016/j.jaad.2016.03.048
- Silverberg JI. Racial and ethnic disparities in atopic dermatitis. Curr Dermatol Rep. 2015;4:44-48.
- Buster KJ, Sevens EI, Elmets CA. Dermatologic health disparities. Dermatol Clin. 2012;30:53-59. doi:10.1016/j.det.2011.08.002
- Barbieri JS, Shin DB, Wang S, et al. Association of race/ethnicity with differences in health care use and treatment for acne. JAMA Dermatol. 2020;156:312-319. doi:10.1001/jamadermatol.2019.4818
- Smedley BD, Stith AY, Colburn L, et al. The Right Thing To Do, The Smart Thing to Do: Enhancing Diversity in the Health Professions. National Academies Press; 2001.
- Association of American Medical Colleges. Minorities in medical education: fact and figures 2019. Accessed December 9, 2021. https://www.aamc.org/datareports/workforce/report/diversity-medicine-facts-and-figures-2019
- Liaison Committee on Medical Education (LCME) standards on diversity. University of South Florida Health website. Accessed December 9, 2021. https://health.usf.edu/~/media/Files/Medicine/MD%20Program/Diversity/LCMEStandardsonDiversity1.ashx?la=en
- Granstein RD, Cornelius L, Shinkai K. Diversity in dermatology—a call for action. JAMA Dermatol. 2017;153:499-500. doi:10.1001/jamadermatol.2017.0296
- Lett LA, Murdock HM, Orji W, et al. Trends in racial/ethnic representation among US medical students. JAMA Netw Open. 2019;2:e1910490. doi:10.1001/jamanetworkopen.2019.10490
- Association of American Medical Colleges. Altering the course: Black males in medicine. Published 2015. Accessed December 8, 2021. https://store.aamc.org/downloadable/download/sample/sample_id/84/
- Barr DA, Gonzalez ME, Wanat SF. The leaky pipeline: factors associated with early decline in interest in premedical studies among underrepresented minority undergraduate students. Acad Med. 2008;83:5:503-511. doi:10.1097/ACM.0b013e31816bda16
- Flores RL. The rising gap between rich and poor: a look at the persistence of educational disparities in the United States and why we should worry. Cogent Soc Sci. 2017;3:1323698.
- Jackson D. Why am I behind? an examination of low income and minority students’ preparedness for college. McNair Sch J. 2012;13:121-138.
- Rothstein R. The racial achievement gap, segregated schools, andsegregated neighborhoods: a constitutional insult. Race Soc Probl. 2015;7:21-30.
- Association of American Medical Colleges. Residency Applicants From US MD Granting Medical Schools to ACGME-Accredited Programs by Specialty and Race/Ethnicity. Association of American Medical Colleges; 2017.
- Brotherton SE, Etzel SL. Graduate medical education, 2018-2019. JAMA. 2019;322:996-1016. doi:10.1001/jama.2019.10155
- Barnes LA, Bae GH, Nambudiri V. Sex and racial/ethnic diversity of US medical students and their exposure to dermatology programs. JAMA Dermatol. 2019;155:490-491. doi:10.1001/jamadermatol.2018.5025
- Soliman YS, Rzepecki AK, Guzman AK. Understanding perceived barriers of minority medical students pursuing a career in dermatology. JAMA Dermatol. 2019;155:252-254. doi:10.1001/jamadermatol.2018.4813
- Low D, Pollack SW, Liao Z, et al. Racial/ethnic disparities in clinical grading in medical school. Teach Learn Med. 2019;31:487-496. doi:10.1080/10401334.2019.1597724
- Rubright JD, Jodoin M, Barone MA. Examining demographics, prior academic performance and United States medical licensing examination scores. Acad Med. 2019;94;364-370. doi:10.1097/ACM.0000000000002366
- Boatright D, Ross D, O’Connor P, et al. Racial disparities in medical student membership in the alpha omega honor society. JAMA Intern Med. 2017;177:659-665. doi:10.1001/jamainternmed.2016.9623
- Gorouhi F, Alikhan A, Rezaei A, et al. Dermatology residency selection criteria with an emphasis on program characteristics: a national program director survey [published online March 17, 2014]. Dermatol Res Pract. doi:10.1155/2014/692760
- Vasquez R, Jeong H, Florez-Pollack S, et al. What are the barriers faced by underrepresented minorities applying to dermatology? a qualitative cross-sectional study of applicants applying to a large dermatology residency program. J Am Acad Dermatol. 2020;83:1770-1773. doi:10.1016/j.jaad.2020.03.067
- Results of the 2019 NRMP applicant survey by preferred specialty and applicant type. National Resident Matching Program website. Published July 2019. Accessed December 8, 2021. https://www.nrmp.org/wp-content/uploads/2019/06/Applicant-Survey-Report-2019.pdf
- Mansouri B, Walker GD, Mitchell J, et al. The cost of applying to dermatology residency: 2014 data estimates. J Am Acad Dermatol. 2016;74:754-756. doi:10.1016/j.jaad.2015.10.049
- Polacco MA, Lally J, Walls A, et al. Digging into debt: the financial burden associated with the otolaryngology match. Otolaryngol Head Neck Surg. 2017;12:1091-1096. doi:10.1177/0194599816686538
- Feng H, Feng PW, Geronemus RG. Diversity in the US Mohs micrographic surgery workforce. Dermatol Surg. 2020:46:1451-1455. doi:10.1097/DSS.0000000000002080
- Lett LA, Orji WU, Sebro R. Declining racial and ethnic representation in clinical academic medicine: a longitudinal study of 16 US medical specialties. PLoS ONE. 2018;13:e0207274. doi:10.1371/journal.pone.020727432. Xierali IM, Nivet MA, Pandya AG. US Dermatology department faculty diversity trends by sex and underrepresented-in-medicine status, 1970-2018. JAMA Dermatol. 2020;156:280-287. doi:10.1001/jamadermatol.2019.4297
- Okoye GA. Supporting underrepresented minority women in academic dermatology. Intl J Womens Dermatol. 2020;6:57-60. doi:10.1016/j.ijwd.2019.09.009
- Bernstein J, Dicaprio MR, Mehta S. The relationship between required medical school instruction in musculoskeletal medicine and application rates to orthopaedic surgery residency programs. J Bone Joint Surg Am. 2004;86:2335-2338. doi:10.2106/00004623-200410000-00031
- Capers Q, Clinchot D, McDougle L, et al. Implicit racial bias in medical school admissions. Acad Med. 2017;92:365-369. doi:10.1097/ACM.0000000000001388
- Dobbin F, Kalev A. Why diversity programs fail. Harvard Business Rev. 2016;52-60. Accessed December 8, 2021. https://hbr.org/2016/07/why-diversity-programs-fail
- Kalev A, Dobbin F, Kelly E. Best practices or best guesses? assessing the efficacy of corporate affirmative action and diversity policies. Am Sociol Rev. 2006;71:589-617.
- Sanchez JI, Medkik N. The effects of diversity awareness training on differential treatment. Group Organ Manag. 2004;29:517-536.
- Kaiser CR, Major B, Jurcevic I, et al. Presumed fair: ironic effects of organizational diversity structures. J Pers Soc Psychol. 2013;104:504-519. doi:10.1037/a0030838
- Nijhawan RI, Jacob SE, Woolery-Lloyd H. Skin of color education in dermatology residency programs: does residency training reflect the changing demographics of the United States? J Am Acad Dermatol. 2008;59:615-617.
- Jia JL, Gordon JS, Lester JC, et al. Integrating skin of color and sexual and gender minority content into dermatology residency curricula: a prospective program initiative [published online April 16, 2021]. J Am Acad Dermatol. doi:10.1016/j.jaad.2021.04.018
- Amuzie AU, Lia JL, Taylor SC, et al. Skin of color article representation in dermatology literature 2009-2019: higher citation counts and opportunities for inclusion [published online March 24, 2021]. J Am Acad Dermatol. doi:10.1016/j.jaad.2021.03.063
- Beech BM, Calles-Escandon J, Hairston KC, et al. Mentoring programs for underrepresented minority faculty in academic medical center: a systematic review of the literature. Acad Med. 2013;88:541-549. doi:10.1097/ACM.0b013e31828589e3
- Daley S, Wingard DL, Reznik V. Improving the retention of underrepresented minority faculty in academic medicine. J Natl Med Assoc. 2006;98:1435-1440. doi:10.1016/s0027-9684(15)31449-8
- Association of American Medical Colleges. US medical school faculty by sex, race/ethnicity, rank, and department, 2019. Published December 31, 2019. Accessed December 20, 2021. https://www.aamc.org/media/8476/download?attachment
Practice Points
- Dermatology remains the second least diverse specialty in medicine, which has important implications for the workforce and clinical excellence of the specialty.
- Barriers presenting at different stages of medical education and training result in the loss of underrepresented minority (URM) learners pursuing or advancing careers in dermatology.
- Understanding these barriers is the first step to creating and implementing important structural changes to the way we mentor, teach, and support URM students in the specialty.