User login
Lonely elderly patients suffer worse health outcomes
WASHINGTON – More lonely elderly patients suffered from health symptoms and received very aggressive end of life care than nonlonely elderly patients, according to a study presented at the annual meeting of the Society of General Internal Medicine.
“Loneliness and social isolation are very common problems, especially in older Americans, and inflict about 30%-40% of older Americans. But while we know that this may have implications for their quality [of] life and may actually lead to premature death, we know very little about the end of life experience,” said Nauzley Abedini, MD, MSc, a hospitalist in internal medicine at the University of Michigan, Ann Arbor.
The study sought to determine the association between loneliness and end of life experience as measured by symptom burden, intensity of care, and advance care planning in adults. The pooled cohort study used data from the Health and Retirement Study (HRS) to analyze older Americans (aged 50 years or more) who died between 2004 and 2014. Investigators conducted postmortem “exit interviews” with the next of kin after each participant’s death. There were 2,896 participants included in the survey. Of these participants, 34% (942) were lonely; the remaining 1,954 of elderly adults were classified as nonlonely.
Loneliness was defined using the three-item Revised University of California, Los Angeles, Loneliness Scale score from a decedent’s last HRS interview prior to death. These items included feeling left out, feeling isolated, and lacking companionship. Investigators used this data to create a loneliness variable on previously established cutpoints for “lonely” and “nonlonely” participants. The data was used from the most recent survey prior to death.
Results showed more lonely older adults suffered from health symptoms in the last year of life, compared with nonlonely older adults (69.1% vs. 59.5%; odds ratio, 1.52; 95% confidence interval, 1.30-1.78). These symptoms included being troubled by pain, having difficulty breathing, experiencing severe fatigue, and having periodic confusion.
Patients with loneliness associated with intensity of health care at the end of life were more likely to die in a nursing home than at home, compared with nonlonely adults (OR, 1.68; 95% CI, 1.25-2.27). The lonely patients also were more likely to use life support during their last 2 years of life (OR, 1.41; 95% CI, 1.16-1.70).
“For clinicians, we need to identify end of life as an additional vulnerable time for people who are lonely. Currently, most of our interventions in terms of screening for loneliness are in the outpatient setting, but I would argue that working in hospitals, hospices, nursing homes, and community organizations, where these folks are living and dying, would be useful places to screen for this,” Dr. Abedini said.
The authors had no disclosures.
WASHINGTON – More lonely elderly patients suffered from health symptoms and received very aggressive end of life care than nonlonely elderly patients, according to a study presented at the annual meeting of the Society of General Internal Medicine.
“Loneliness and social isolation are very common problems, especially in older Americans, and inflict about 30%-40% of older Americans. But while we know that this may have implications for their quality [of] life and may actually lead to premature death, we know very little about the end of life experience,” said Nauzley Abedini, MD, MSc, a hospitalist in internal medicine at the University of Michigan, Ann Arbor.
The study sought to determine the association between loneliness and end of life experience as measured by symptom burden, intensity of care, and advance care planning in adults. The pooled cohort study used data from the Health and Retirement Study (HRS) to analyze older Americans (aged 50 years or more) who died between 2004 and 2014. Investigators conducted postmortem “exit interviews” with the next of kin after each participant’s death. There were 2,896 participants included in the survey. Of these participants, 34% (942) were lonely; the remaining 1,954 of elderly adults were classified as nonlonely.
Loneliness was defined using the three-item Revised University of California, Los Angeles, Loneliness Scale score from a decedent’s last HRS interview prior to death. These items included feeling left out, feeling isolated, and lacking companionship. Investigators used this data to create a loneliness variable on previously established cutpoints for “lonely” and “nonlonely” participants. The data was used from the most recent survey prior to death.
Results showed more lonely older adults suffered from health symptoms in the last year of life, compared with nonlonely older adults (69.1% vs. 59.5%; odds ratio, 1.52; 95% confidence interval, 1.30-1.78). These symptoms included being troubled by pain, having difficulty breathing, experiencing severe fatigue, and having periodic confusion.
Patients with loneliness associated with intensity of health care at the end of life were more likely to die in a nursing home than at home, compared with nonlonely adults (OR, 1.68; 95% CI, 1.25-2.27). The lonely patients also were more likely to use life support during their last 2 years of life (OR, 1.41; 95% CI, 1.16-1.70).
“For clinicians, we need to identify end of life as an additional vulnerable time for people who are lonely. Currently, most of our interventions in terms of screening for loneliness are in the outpatient setting, but I would argue that working in hospitals, hospices, nursing homes, and community organizations, where these folks are living and dying, would be useful places to screen for this,” Dr. Abedini said.
The authors had no disclosures.
WASHINGTON – More lonely elderly patients suffered from health symptoms and received very aggressive end of life care than nonlonely elderly patients, according to a study presented at the annual meeting of the Society of General Internal Medicine.
“Loneliness and social isolation are very common problems, especially in older Americans, and inflict about 30%-40% of older Americans. But while we know that this may have implications for their quality [of] life and may actually lead to premature death, we know very little about the end of life experience,” said Nauzley Abedini, MD, MSc, a hospitalist in internal medicine at the University of Michigan, Ann Arbor.
The study sought to determine the association between loneliness and end of life experience as measured by symptom burden, intensity of care, and advance care planning in adults. The pooled cohort study used data from the Health and Retirement Study (HRS) to analyze older Americans (aged 50 years or more) who died between 2004 and 2014. Investigators conducted postmortem “exit interviews” with the next of kin after each participant’s death. There were 2,896 participants included in the survey. Of these participants, 34% (942) were lonely; the remaining 1,954 of elderly adults were classified as nonlonely.
Loneliness was defined using the three-item Revised University of California, Los Angeles, Loneliness Scale score from a decedent’s last HRS interview prior to death. These items included feeling left out, feeling isolated, and lacking companionship. Investigators used this data to create a loneliness variable on previously established cutpoints for “lonely” and “nonlonely” participants. The data was used from the most recent survey prior to death.
Results showed more lonely older adults suffered from health symptoms in the last year of life, compared with nonlonely older adults (69.1% vs. 59.5%; odds ratio, 1.52; 95% confidence interval, 1.30-1.78). These symptoms included being troubled by pain, having difficulty breathing, experiencing severe fatigue, and having periodic confusion.
Patients with loneliness associated with intensity of health care at the end of life were more likely to die in a nursing home than at home, compared with nonlonely adults (OR, 1.68; 95% CI, 1.25-2.27). The lonely patients also were more likely to use life support during their last 2 years of life (OR, 1.41; 95% CI, 1.16-1.70).
“For clinicians, we need to identify end of life as an additional vulnerable time for people who are lonely. Currently, most of our interventions in terms of screening for loneliness are in the outpatient setting, but I would argue that working in hospitals, hospices, nursing homes, and community organizations, where these folks are living and dying, would be useful places to screen for this,” Dr. Abedini said.
The authors had no disclosures.
REPORTING FROM SGIM 2019
Insomnia meds get boxed warning from FDA
The Food and Drug Administration will now require that certain
Complex sleep behaviors have been seen with these medications in patients with and without a history of them, at low doses, and even after one dose of the medication. They’ve also been observed with and without concomitant use of alcohol or other CNS depressants.
Health care professionals should advise patients about these risks, even though they are rare. Patients should contact health care professionals if they either experience a complex sleep behavior while not fully awake on one of these medicines or have performed activities they don’t remember while taking the medicine.
More information about these risks and the safety warnings can be found in the FDA’s safety announcement. Other information is also available in a press announcement from the agency.
The Food and Drug Administration will now require that certain
Complex sleep behaviors have been seen with these medications in patients with and without a history of them, at low doses, and even after one dose of the medication. They’ve also been observed with and without concomitant use of alcohol or other CNS depressants.
Health care professionals should advise patients about these risks, even though they are rare. Patients should contact health care professionals if they either experience a complex sleep behavior while not fully awake on one of these medicines or have performed activities they don’t remember while taking the medicine.
More information about these risks and the safety warnings can be found in the FDA’s safety announcement. Other information is also available in a press announcement from the agency.
The Food and Drug Administration will now require that certain
Complex sleep behaviors have been seen with these medications in patients with and without a history of them, at low doses, and even after one dose of the medication. They’ve also been observed with and without concomitant use of alcohol or other CNS depressants.
Health care professionals should advise patients about these risks, even though they are rare. Patients should contact health care professionals if they either experience a complex sleep behavior while not fully awake on one of these medicines or have performed activities they don’t remember while taking the medicine.
More information about these risks and the safety warnings can be found in the FDA’s safety announcement. Other information is also available in a press announcement from the agency.
ERRATUM
A recent letter, “Hypoglycemia in the elderly: Watch for atypical symptoms” (J Fam Pract. 2019;68:116) provided an incomplete list of the letter’s authors. The list should have read: Jan Brož, MD, Jana Urbanová, MD, PhD, Prague, Czech Republic; Brian M. Frier, MD, BSc, Edinburgh, United Kingdom.
A recent letter, “Hypoglycemia in the elderly: Watch for atypical symptoms” (J Fam Pract. 2019;68:116) provided an incomplete list of the letter’s authors. The list should have read: Jan Brož, MD, Jana Urbanová, MD, PhD, Prague, Czech Republic; Brian M. Frier, MD, BSc, Edinburgh, United Kingdom.
A recent letter, “Hypoglycemia in the elderly: Watch for atypical symptoms” (J Fam Pract. 2019;68:116) provided an incomplete list of the letter’s authors. The list should have read: Jan Brož, MD, Jana Urbanová, MD, PhD, Prague, Czech Republic; Brian M. Frier, MD, BSc, Edinburgh, United Kingdom.
Experts propose new definition and recommendations for Alzheimer’s-like disorder
An international group of experts has proposed a new name, staging criteria, and recommendations for a recently recognized brain disorder that mimics Alzheimer’s disease and is marked by a proteinopathy caused by malformed transactive response DNA-binding protein of 43 kDa (TDP-43).
The term limbic-predominant age-related TDP-43 encephalopathy (LATE) was coined in an effort to raise awareness and kick-start research into this “pathway to dementia,” the experts wrote in a report appearing in Brain.
“As there is currently no universally agreed-upon terminology or staging system for common age-related TDP-43 proteinopathy, this condition is understudied and not well recognized, even among investigators in the field of dementia research,” wrote the authors of the report, led by Peter T. Nelson, MD, PhD, of the University of Kentucky, Lexington.
LATE neuropathologic changes, associated with a progressive amnesia syndrome that mimics Alzheimer’s, are seen in more than 20% of individuals past the age of 80 years, according to large, community-based autopsy series. It coexists with Alzheimer’s disease in many patients, lowering the threshold for developing dementia, authors said.
The term LATE is designed to encompass several other terms related to TDP-43 pathology, including hippocampal sclerosis and cerebral age-related TDP-43 with sclerosis, Dr. Nelson and coauthors noted in their report.
The TDP-43 protein is encoded by the TARDBP gene and provides several functions related to the regulation of gene expression, the authors wrote.
Misfolded TDP-43 was known to play a causative role in amyotrophic lateral sclerosis and frontotemporal lobar degeneration, the authors noted, and then was also identified in the brains of older individuals with hippocampal sclerosis or Alzheimer’s disease neuropathologic changes.
The authors proposed a three-stage classification system for LATE neuropathologic change based on TDP-43 immunohistochemistry performed during routine autopsy evaluation of the amygdala, hippocampus, and middle frontal gyrus.
The amygdala is an area affected early in the course of the disease (Stage 1), whereas involvement of the hippocampus represents a more intermediate stage (Stage 2), and the middle frontal gyrus is more affected in advanced stages of the disease (Stage 3), according to the schema.
Five genes have been identified with risk alleles for LATE neuropathologic changes, authors said. Of note, several groups have found that the apolipoprotein E epsilon 4 (APOE4) allele, known to be a risk factor for Alzheimer’s disease neuropathologic changes and Lewy body disease, is also linked to increased risk of TDP-43 proteinopathy.
There are no established biomarkers specific to TDP-43 proteinopathy yet, which hampers development of clinical trials designed to test interventions to treat or prevent LATE, Dr. Nelson and colleagues said in their report.
LATE could also obscure the effects of potentially disease-modifying agents being tested in Alzheimer’s disease clinical trials, which can complicate the interpretation of study results, they added.
“Until there are biomarkers for LATE, clinical trials should be powered to account for TDP-43 proteinopathy,” they wrote.
Dr. Nelson and coauthors of the report in Brain reported no competing interests.
SOURCE: Nelson PT, et al. Brain. 2019 Apr 30. doi: 10.1093/brain/awz099
Alois Alzheimer’s original patient was 51 years old, and for roughly 70 years Alzheimer’s disease was considered a rare disease that caused presenile dementia. In the 1970s, Robert Katzman, MD, and Robert D. Terry, MD, equated the neuropathologic features of Alzheimer’s disease with the more common senile dementia, and since then we have recognized Alzheimer’s disease as the most common form of dementia. Autopsy studies of patients dying in their 80s and 90s, however, has revealed that far more common than pure Alzheimer’s disease is a mixed neuropathologic picture. In addition, with the advent of biomarker studies a substantial number of individuals have “suspected non-Alzheimer pathology.”
Interestingly, the authors identify the apolipoprotein E epsilon 4 (APOE4) allele as a predisposing factor for LATE, although given the advanced age of the LATE patient population, one could argue that a certain degree of resilience extended their lives into the LATE age range.
In contrast, in the Alzheimer’s Disease Sequencing Project, among those with autopsy confirmation, the prevalence of APOE4 in Braak stage 5-6 declines with succeeding decades so that, by the 80s and 90s, the prevalence of APOE2 is actually higher at 7.3% vs. 4.1% with APOE4 for ages 80 to younger than 85 years, 9.3% with APOE2 vs. 8.6% with APOE4 for 85 to younger than 90 years, and 16.7% with APOE2 vs. 6.9% with APOE4 for ages 90 years and above.
Our understanding of age-related cognitive decline, from the normal to the pathological ends of the spectrum, continues to evolve, and LATE is simply the latest addition to our growing knowledge base that will further inform clinical diagnosis, research, and experimental therapeutics.
Richard J. Caselli, MD, is professor of neurology at the Mayo Clinic Arizona in Scottsdale and associate director and clinical core director of the Arizona Alzheimer’s Disease Center.
Alois Alzheimer’s original patient was 51 years old, and for roughly 70 years Alzheimer’s disease was considered a rare disease that caused presenile dementia. In the 1970s, Robert Katzman, MD, and Robert D. Terry, MD, equated the neuropathologic features of Alzheimer’s disease with the more common senile dementia, and since then we have recognized Alzheimer’s disease as the most common form of dementia. Autopsy studies of patients dying in their 80s and 90s, however, has revealed that far more common than pure Alzheimer’s disease is a mixed neuropathologic picture. In addition, with the advent of biomarker studies a substantial number of individuals have “suspected non-Alzheimer pathology.”
Interestingly, the authors identify the apolipoprotein E epsilon 4 (APOE4) allele as a predisposing factor for LATE, although given the advanced age of the LATE patient population, one could argue that a certain degree of resilience extended their lives into the LATE age range.
In contrast, in the Alzheimer’s Disease Sequencing Project, among those with autopsy confirmation, the prevalence of APOE4 in Braak stage 5-6 declines with succeeding decades so that, by the 80s and 90s, the prevalence of APOE2 is actually higher at 7.3% vs. 4.1% with APOE4 for ages 80 to younger than 85 years, 9.3% with APOE2 vs. 8.6% with APOE4 for 85 to younger than 90 years, and 16.7% with APOE2 vs. 6.9% with APOE4 for ages 90 years and above.
Our understanding of age-related cognitive decline, from the normal to the pathological ends of the spectrum, continues to evolve, and LATE is simply the latest addition to our growing knowledge base that will further inform clinical diagnosis, research, and experimental therapeutics.
Richard J. Caselli, MD, is professor of neurology at the Mayo Clinic Arizona in Scottsdale and associate director and clinical core director of the Arizona Alzheimer’s Disease Center.
Alois Alzheimer’s original patient was 51 years old, and for roughly 70 years Alzheimer’s disease was considered a rare disease that caused presenile dementia. In the 1970s, Robert Katzman, MD, and Robert D. Terry, MD, equated the neuropathologic features of Alzheimer’s disease with the more common senile dementia, and since then we have recognized Alzheimer’s disease as the most common form of dementia. Autopsy studies of patients dying in their 80s and 90s, however, has revealed that far more common than pure Alzheimer’s disease is a mixed neuropathologic picture. In addition, with the advent of biomarker studies a substantial number of individuals have “suspected non-Alzheimer pathology.”
Interestingly, the authors identify the apolipoprotein E epsilon 4 (APOE4) allele as a predisposing factor for LATE, although given the advanced age of the LATE patient population, one could argue that a certain degree of resilience extended their lives into the LATE age range.
In contrast, in the Alzheimer’s Disease Sequencing Project, among those with autopsy confirmation, the prevalence of APOE4 in Braak stage 5-6 declines with succeeding decades so that, by the 80s and 90s, the prevalence of APOE2 is actually higher at 7.3% vs. 4.1% with APOE4 for ages 80 to younger than 85 years, 9.3% with APOE2 vs. 8.6% with APOE4 for 85 to younger than 90 years, and 16.7% with APOE2 vs. 6.9% with APOE4 for ages 90 years and above.
Our understanding of age-related cognitive decline, from the normal to the pathological ends of the spectrum, continues to evolve, and LATE is simply the latest addition to our growing knowledge base that will further inform clinical diagnosis, research, and experimental therapeutics.
Richard J. Caselli, MD, is professor of neurology at the Mayo Clinic Arizona in Scottsdale and associate director and clinical core director of the Arizona Alzheimer’s Disease Center.
An international group of experts has proposed a new name, staging criteria, and recommendations for a recently recognized brain disorder that mimics Alzheimer’s disease and is marked by a proteinopathy caused by malformed transactive response DNA-binding protein of 43 kDa (TDP-43).
The term limbic-predominant age-related TDP-43 encephalopathy (LATE) was coined in an effort to raise awareness and kick-start research into this “pathway to dementia,” the experts wrote in a report appearing in Brain.
“As there is currently no universally agreed-upon terminology or staging system for common age-related TDP-43 proteinopathy, this condition is understudied and not well recognized, even among investigators in the field of dementia research,” wrote the authors of the report, led by Peter T. Nelson, MD, PhD, of the University of Kentucky, Lexington.
LATE neuropathologic changes, associated with a progressive amnesia syndrome that mimics Alzheimer’s, are seen in more than 20% of individuals past the age of 80 years, according to large, community-based autopsy series. It coexists with Alzheimer’s disease in many patients, lowering the threshold for developing dementia, authors said.
The term LATE is designed to encompass several other terms related to TDP-43 pathology, including hippocampal sclerosis and cerebral age-related TDP-43 with sclerosis, Dr. Nelson and coauthors noted in their report.
The TDP-43 protein is encoded by the TARDBP gene and provides several functions related to the regulation of gene expression, the authors wrote.
Misfolded TDP-43 was known to play a causative role in amyotrophic lateral sclerosis and frontotemporal lobar degeneration, the authors noted, and then was also identified in the brains of older individuals with hippocampal sclerosis or Alzheimer’s disease neuropathologic changes.
The authors proposed a three-stage classification system for LATE neuropathologic change based on TDP-43 immunohistochemistry performed during routine autopsy evaluation of the amygdala, hippocampus, and middle frontal gyrus.
The amygdala is an area affected early in the course of the disease (Stage 1), whereas involvement of the hippocampus represents a more intermediate stage (Stage 2), and the middle frontal gyrus is more affected in advanced stages of the disease (Stage 3), according to the schema.
Five genes have been identified with risk alleles for LATE neuropathologic changes, authors said. Of note, several groups have found that the apolipoprotein E epsilon 4 (APOE4) allele, known to be a risk factor for Alzheimer’s disease neuropathologic changes and Lewy body disease, is also linked to increased risk of TDP-43 proteinopathy.
There are no established biomarkers specific to TDP-43 proteinopathy yet, which hampers development of clinical trials designed to test interventions to treat or prevent LATE, Dr. Nelson and colleagues said in their report.
LATE could also obscure the effects of potentially disease-modifying agents being tested in Alzheimer’s disease clinical trials, which can complicate the interpretation of study results, they added.
“Until there are biomarkers for LATE, clinical trials should be powered to account for TDP-43 proteinopathy,” they wrote.
Dr. Nelson and coauthors of the report in Brain reported no competing interests.
SOURCE: Nelson PT, et al. Brain. 2019 Apr 30. doi: 10.1093/brain/awz099
An international group of experts has proposed a new name, staging criteria, and recommendations for a recently recognized brain disorder that mimics Alzheimer’s disease and is marked by a proteinopathy caused by malformed transactive response DNA-binding protein of 43 kDa (TDP-43).
The term limbic-predominant age-related TDP-43 encephalopathy (LATE) was coined in an effort to raise awareness and kick-start research into this “pathway to dementia,” the experts wrote in a report appearing in Brain.
“As there is currently no universally agreed-upon terminology or staging system for common age-related TDP-43 proteinopathy, this condition is understudied and not well recognized, even among investigators in the field of dementia research,” wrote the authors of the report, led by Peter T. Nelson, MD, PhD, of the University of Kentucky, Lexington.
LATE neuropathologic changes, associated with a progressive amnesia syndrome that mimics Alzheimer’s, are seen in more than 20% of individuals past the age of 80 years, according to large, community-based autopsy series. It coexists with Alzheimer’s disease in many patients, lowering the threshold for developing dementia, authors said.
The term LATE is designed to encompass several other terms related to TDP-43 pathology, including hippocampal sclerosis and cerebral age-related TDP-43 with sclerosis, Dr. Nelson and coauthors noted in their report.
The TDP-43 protein is encoded by the TARDBP gene and provides several functions related to the regulation of gene expression, the authors wrote.
Misfolded TDP-43 was known to play a causative role in amyotrophic lateral sclerosis and frontotemporal lobar degeneration, the authors noted, and then was also identified in the brains of older individuals with hippocampal sclerosis or Alzheimer’s disease neuropathologic changes.
The authors proposed a three-stage classification system for LATE neuropathologic change based on TDP-43 immunohistochemistry performed during routine autopsy evaluation of the amygdala, hippocampus, and middle frontal gyrus.
The amygdala is an area affected early in the course of the disease (Stage 1), whereas involvement of the hippocampus represents a more intermediate stage (Stage 2), and the middle frontal gyrus is more affected in advanced stages of the disease (Stage 3), according to the schema.
Five genes have been identified with risk alleles for LATE neuropathologic changes, authors said. Of note, several groups have found that the apolipoprotein E epsilon 4 (APOE4) allele, known to be a risk factor for Alzheimer’s disease neuropathologic changes and Lewy body disease, is also linked to increased risk of TDP-43 proteinopathy.
There are no established biomarkers specific to TDP-43 proteinopathy yet, which hampers development of clinical trials designed to test interventions to treat or prevent LATE, Dr. Nelson and colleagues said in their report.
LATE could also obscure the effects of potentially disease-modifying agents being tested in Alzheimer’s disease clinical trials, which can complicate the interpretation of study results, they added.
“Until there are biomarkers for LATE, clinical trials should be powered to account for TDP-43 proteinopathy,” they wrote.
Dr. Nelson and coauthors of the report in Brain reported no competing interests.
SOURCE: Nelson PT, et al. Brain. 2019 Apr 30. doi: 10.1093/brain/awz099
FROM BRAIN
AD biomarker not tied to increased interest in physician-assisted death
Being diagnosed with an elevated amyloid-beta biomarker that indicates greater risk of Alzheimer’s disease did not lead to increased consideration of physician-assisted death (PAD), according to an analysis of patients interviewed during clinical trials on cognitive decline.
“Our findings suggest that learning one’s amyloid imaging result does not change baseline attitudes regarding the acceptability of PAD,” wrote Emily A. Largent, PhD, of the department of medical ethics and health policy at the University of Pennsylvania, Philadelphia, and coauthors. The study was published as a research letter in JAMA Neurology.
Participants were recruited from two ongoing clinical trials, one of which included patients with elevated amyloid-beta (n = 50), whereas the other did not (n = 30). All participants completed an interview 4-12 weeks after receiving their biomarker results; 47 and 30 participants, respectively, also completed a follow-up interview at 12 months.
When asked whether they had considered PAD, nearly two-thirds of interviewees with the Alzheimer’s disease biomarker stated that they neither had nor would. Roughly one in five from that group said they would pursue PAD if they began to suffer from cognitive impairment or became a burden on others. Interviewees who did not have elevated amyloid beta, when asked whether a reversed result would have led to PAD or suicide, showed interest in roughly similar proportion to their at-risk counterparts.
The coauthors acknowledged the limitations of their study, including not asking about other end-of-life preferences or perceived quality of life for people with dementia. They also noted that, although their sample mirrors the populations of the two studies they drew from, “its homogeneity limits generalizability.” As such, they stressed that
The study was supported by grants from the Alzheimer’s Association and the National Institute on Aging. One author reported receiving grants from those two organizations during the study; another reported receiving grants from Lilly and Novartis. No other conflicts of interest were reported.
SOURCE: Largent EA et al. JAMA Neurol. 2019 Apr 29. doi: 10.1001/jamaneurol.2019.0797.
The fascinating thing about this study is that the idea for it arose when some of the individuals spontaneously mentioned assisted suicide during their initial interview, Annette L. Hanson, MD, said in an interview.
“Would these subjects have thought of suicide in the absence of the Brittany Maynard publicity campaign? I doubt it.”
Dr. Hanson, a forensic psychiatrist, is assistant professor of psychiatry at the University of Maryland and at Johns Hopkins University, both in Baltimore.
The fascinating thing about this study is that the idea for it arose when some of the individuals spontaneously mentioned assisted suicide during their initial interview, Annette L. Hanson, MD, said in an interview.
“Would these subjects have thought of suicide in the absence of the Brittany Maynard publicity campaign? I doubt it.”
Dr. Hanson, a forensic psychiatrist, is assistant professor of psychiatry at the University of Maryland and at Johns Hopkins University, both in Baltimore.
The fascinating thing about this study is that the idea for it arose when some of the individuals spontaneously mentioned assisted suicide during their initial interview, Annette L. Hanson, MD, said in an interview.
“Would these subjects have thought of suicide in the absence of the Brittany Maynard publicity campaign? I doubt it.”
Dr. Hanson, a forensic psychiatrist, is assistant professor of psychiatry at the University of Maryland and at Johns Hopkins University, both in Baltimore.
Being diagnosed with an elevated amyloid-beta biomarker that indicates greater risk of Alzheimer’s disease did not lead to increased consideration of physician-assisted death (PAD), according to an analysis of patients interviewed during clinical trials on cognitive decline.
“Our findings suggest that learning one’s amyloid imaging result does not change baseline attitudes regarding the acceptability of PAD,” wrote Emily A. Largent, PhD, of the department of medical ethics and health policy at the University of Pennsylvania, Philadelphia, and coauthors. The study was published as a research letter in JAMA Neurology.
Participants were recruited from two ongoing clinical trials, one of which included patients with elevated amyloid-beta (n = 50), whereas the other did not (n = 30). All participants completed an interview 4-12 weeks after receiving their biomarker results; 47 and 30 participants, respectively, also completed a follow-up interview at 12 months.
When asked whether they had considered PAD, nearly two-thirds of interviewees with the Alzheimer’s disease biomarker stated that they neither had nor would. Roughly one in five from that group said they would pursue PAD if they began to suffer from cognitive impairment or became a burden on others. Interviewees who did not have elevated amyloid beta, when asked whether a reversed result would have led to PAD or suicide, showed interest in roughly similar proportion to their at-risk counterparts.
The coauthors acknowledged the limitations of their study, including not asking about other end-of-life preferences or perceived quality of life for people with dementia. They also noted that, although their sample mirrors the populations of the two studies they drew from, “its homogeneity limits generalizability.” As such, they stressed that
The study was supported by grants from the Alzheimer’s Association and the National Institute on Aging. One author reported receiving grants from those two organizations during the study; another reported receiving grants from Lilly and Novartis. No other conflicts of interest were reported.
SOURCE: Largent EA et al. JAMA Neurol. 2019 Apr 29. doi: 10.1001/jamaneurol.2019.0797.
Being diagnosed with an elevated amyloid-beta biomarker that indicates greater risk of Alzheimer’s disease did not lead to increased consideration of physician-assisted death (PAD), according to an analysis of patients interviewed during clinical trials on cognitive decline.
“Our findings suggest that learning one’s amyloid imaging result does not change baseline attitudes regarding the acceptability of PAD,” wrote Emily A. Largent, PhD, of the department of medical ethics and health policy at the University of Pennsylvania, Philadelphia, and coauthors. The study was published as a research letter in JAMA Neurology.
Participants were recruited from two ongoing clinical trials, one of which included patients with elevated amyloid-beta (n = 50), whereas the other did not (n = 30). All participants completed an interview 4-12 weeks after receiving their biomarker results; 47 and 30 participants, respectively, also completed a follow-up interview at 12 months.
When asked whether they had considered PAD, nearly two-thirds of interviewees with the Alzheimer’s disease biomarker stated that they neither had nor would. Roughly one in five from that group said they would pursue PAD if they began to suffer from cognitive impairment or became a burden on others. Interviewees who did not have elevated amyloid beta, when asked whether a reversed result would have led to PAD or suicide, showed interest in roughly similar proportion to their at-risk counterparts.
The coauthors acknowledged the limitations of their study, including not asking about other end-of-life preferences or perceived quality of life for people with dementia. They also noted that, although their sample mirrors the populations of the two studies they drew from, “its homogeneity limits generalizability.” As such, they stressed that
The study was supported by grants from the Alzheimer’s Association and the National Institute on Aging. One author reported receiving grants from those two organizations during the study; another reported receiving grants from Lilly and Novartis. No other conflicts of interest were reported.
SOURCE: Largent EA et al. JAMA Neurol. 2019 Apr 29. doi: 10.1001/jamaneurol.2019.0797.
FROM JAMA NEUROLOGY
Medical cannabis relieved pain, decreased opioid use in elderly
results of a recent retrospective chart review suggest. Treatment with medical cannabis improved pain, sleep, anxiety, and neuropathy in patients aged 75 years of age and older, and was associated with reduced use of opioids in about one-third of cases, according to authors of the study, which will be presented at the annual meeting of the American Academy of Neurology.
“Our findings are promising and can help fuel further research into medical marijuana as an additional option for this group of people who often have chronic conditions,” said lead investigator Laszlo Mechtler, MD, of Dent Neurologic Institute in Buffalo, N.Y., in a news release. However, additional randomized, placebo-controlled studies are needed to confirm results of this study, Dr. Mechtler added.
The chart review focused on 204 elderly patients who participated in New York State’s medical marijuana program and were followed in a neurologic outpatient setting. The cohort included 129 female and 75 male patients, ranging in age from 75 to 102 years, with a mean age of 81 years. The medical marijuana was taken by mouth as a liquid extract tincture, capsule, or in an electronic vaporizer.
With an average exposure time of 16.8 weeks, 69% of patients experienced symptomatic benefit, according to patient self-report. The most commonly reported benefit was relief of chronic pain in 49%, while improvements in sleep, neuropathy, and anxiety were reported in 18%, 15%, and 10%, respectively. Reductions in opioid pain medication were noted in about one-third of cases, they found.
While 34% of patients had adverse effects on medical marijuana, only 21% reported adverse effects after cannabinoid doses were adjusted, investigators said. Adverse effects led to discontinuation of medical cannabis in seven patients, or 3.4% of the overall cohort. Somnolence, disequilibrium, and gastrointestinal disturbance were the most common adverse effects, occurring in 13%, 7%, and 7% of patients, respectively. Euphoria was reported in 3% of patients.
Among patients who had no reported adverse effects, the most commonly used formulation was a balanced 1:1 tincture of tetrahydrocannabinol to cannabidiol, investigators said.
Further trials could explore optimal dosing of medical cannabis in elderly patients and shed more light on adverse effects such as somnolence and disequilibrium, according to Dr. Mechtler and colleagues.
The study was supported by the Dent Family Foundation.
SOURCE: Bargnes V et al. AAN 2019, Abstract P4.1-014.
results of a recent retrospective chart review suggest. Treatment with medical cannabis improved pain, sleep, anxiety, and neuropathy in patients aged 75 years of age and older, and was associated with reduced use of opioids in about one-third of cases, according to authors of the study, which will be presented at the annual meeting of the American Academy of Neurology.
“Our findings are promising and can help fuel further research into medical marijuana as an additional option for this group of people who often have chronic conditions,” said lead investigator Laszlo Mechtler, MD, of Dent Neurologic Institute in Buffalo, N.Y., in a news release. However, additional randomized, placebo-controlled studies are needed to confirm results of this study, Dr. Mechtler added.
The chart review focused on 204 elderly patients who participated in New York State’s medical marijuana program and were followed in a neurologic outpatient setting. The cohort included 129 female and 75 male patients, ranging in age from 75 to 102 years, with a mean age of 81 years. The medical marijuana was taken by mouth as a liquid extract tincture, capsule, or in an electronic vaporizer.
With an average exposure time of 16.8 weeks, 69% of patients experienced symptomatic benefit, according to patient self-report. The most commonly reported benefit was relief of chronic pain in 49%, while improvements in sleep, neuropathy, and anxiety were reported in 18%, 15%, and 10%, respectively. Reductions in opioid pain medication were noted in about one-third of cases, they found.
While 34% of patients had adverse effects on medical marijuana, only 21% reported adverse effects after cannabinoid doses were adjusted, investigators said. Adverse effects led to discontinuation of medical cannabis in seven patients, or 3.4% of the overall cohort. Somnolence, disequilibrium, and gastrointestinal disturbance were the most common adverse effects, occurring in 13%, 7%, and 7% of patients, respectively. Euphoria was reported in 3% of patients.
Among patients who had no reported adverse effects, the most commonly used formulation was a balanced 1:1 tincture of tetrahydrocannabinol to cannabidiol, investigators said.
Further trials could explore optimal dosing of medical cannabis in elderly patients and shed more light on adverse effects such as somnolence and disequilibrium, according to Dr. Mechtler and colleagues.
The study was supported by the Dent Family Foundation.
SOURCE: Bargnes V et al. AAN 2019, Abstract P4.1-014.
results of a recent retrospective chart review suggest. Treatment with medical cannabis improved pain, sleep, anxiety, and neuropathy in patients aged 75 years of age and older, and was associated with reduced use of opioids in about one-third of cases, according to authors of the study, which will be presented at the annual meeting of the American Academy of Neurology.
“Our findings are promising and can help fuel further research into medical marijuana as an additional option for this group of people who often have chronic conditions,” said lead investigator Laszlo Mechtler, MD, of Dent Neurologic Institute in Buffalo, N.Y., in a news release. However, additional randomized, placebo-controlled studies are needed to confirm results of this study, Dr. Mechtler added.
The chart review focused on 204 elderly patients who participated in New York State’s medical marijuana program and were followed in a neurologic outpatient setting. The cohort included 129 female and 75 male patients, ranging in age from 75 to 102 years, with a mean age of 81 years. The medical marijuana was taken by mouth as a liquid extract tincture, capsule, or in an electronic vaporizer.
With an average exposure time of 16.8 weeks, 69% of patients experienced symptomatic benefit, according to patient self-report. The most commonly reported benefit was relief of chronic pain in 49%, while improvements in sleep, neuropathy, and anxiety were reported in 18%, 15%, and 10%, respectively. Reductions in opioid pain medication were noted in about one-third of cases, they found.
While 34% of patients had adverse effects on medical marijuana, only 21% reported adverse effects after cannabinoid doses were adjusted, investigators said. Adverse effects led to discontinuation of medical cannabis in seven patients, or 3.4% of the overall cohort. Somnolence, disequilibrium, and gastrointestinal disturbance were the most common adverse effects, occurring in 13%, 7%, and 7% of patients, respectively. Euphoria was reported in 3% of patients.
Among patients who had no reported adverse effects, the most commonly used formulation was a balanced 1:1 tincture of tetrahydrocannabinol to cannabidiol, investigators said.
Further trials could explore optimal dosing of medical cannabis in elderly patients and shed more light on adverse effects such as somnolence and disequilibrium, according to Dr. Mechtler and colleagues.
The study was supported by the Dent Family Foundation.
SOURCE: Bargnes V et al. AAN 2019, Abstract P4.1-014.
FROM AAN 2019
New sleep apnea guidelines offer evidence-based recommendations
New guidelines on treating obstructive sleep apnea with positive airway pressure include recommendations for using positive airway pressure (PAP) versus no therapy, using either continuous PAP (CPAP) or automatic PAP (APAP) for ongoing treatment, and providing educational interventions to patients starting PAP. The complete guidelines, issued by the American Academy of Sleep Medicine, were published in the Journal of Clinical Sleep Medicine.
The guidelines were driven by improvements in PAP adherence and device technology, wrote lead author Susheel P. Patil, MD, of Johns Hopkins University, Baltimore, and his colleagues.
The guidelines begin with a pair of Good Practice Statements to ensure effective and appropriate management of obstructive sleep apnea (OSA) in adults. First, “Treatment of OSA with PAP therapy should be based on a diagnosis of OSA established using objective sleep apnea testing.” Second, “Adequate follow-up, including troubleshooting and monitoring of objective efficacy and usage data to ensure adequate treatment and adherence, should occur following PAP therapy initiation and during treatment of OSA.”
The nine recommendations, approved by the AASM board of directors, include four strong recommendations that clinicians should follow under most circumstances, and five conditional recommendations that are suggested but lack strong clinical support for their appropriateness for all patients in all circumstances.
The first of the strong recommendations, for using PAP versus no therapy to treat adults with OSA and excessive sleepiness, was based on a high level of evidence from a meta-analysis of 38 randomized, controlled trials and the conclusion that the benefits of PAP outweighed the harms.
The second strong recommendation for using either CPAP or APAP for ongoing treatment was based on data from 26 trials that showed no clinically significant difference between the two. The third strong recommendation that PAP therapy be initiated using either APAP at home or in-laboratory PAP titration in adults with OSA and no significant comorbidities was supported by a meta-analysis of 10 trials that showed no clinically significant difference between at-home and laboratory initiation, and that each option has its benefits. The authors noted that “the majority of well-informed adult patients with OSA and without significant comorbidities would prefer initiation of PAP using the most rapid, convenient, and cost-effective strategy.” This comment supports the fourth strong recommendation for providing educational interventions to patients starting PAP.
The conditional recommendations include using PAP versus no therapy for adults with OSA and impaired quality of life related to poor sleep, such as insomnia, snoring, morning headaches, and daytime fatigue. Other conditional recommendations include using PAP versus no therapy for adults with OSA and comorbid hypertension, choosing CPAP or APAP over bilateral PAP for routine treatment of OSA in adults, providing behavioral interventions or troubleshooting during patients’ initial use of PAP, and using telemonitoring-guided interventions to monitor patients during their initial use of PAP.
“The ultimate judgment regarding any specific care must be made by the treating clinician and the patient, taking into consideration the individual circumstances of the patient, available treatment options, and resources,” the authors noted.
“When implementing the recommendations, providers should consider additional strategies that will maximize the individual patient’s comfort and adherence such as nasal/intranasal over oronasal mask interface and heated humidification,” they added.
The guidelines were developed by a task force commissioned by the AASM that included board-certified sleep specialists and experts in PAP use, and will be reviewed and updated as new information surfaces, the authors wrote.
Dr. Patil reported no financial conflicts; several coauthors reported conflicts that were managed by their not voting on guidelines related to those conflicts.
SOURCE: Patil SP et al. J Clin Sleep Med. 2018 Feb 15;15(2):335-43.
Octavian C. Ioachimescu, MD, FCCP, comments: The last guidelines and practice parameters for the use of positive airway pressure (PAP) as therapy for adult patients with obstructive sleep apnea, were published in 2006 and 2008, respectively. Since then, new technological advances, an ever-growing body of literature, and shifting practice patterns led to an acute need for a thorough reassessment, a comprehensive update of the previous recommendations, and the potential of issuing new ones for emerging areas. As such, the American Academy of Sleep Medicine commissioned a task force of content experts to review the existing evidence, to issue new guidelines and to publish an associated systematic review and a meta-analysis of the literature on this topic.
A welcome recommendation is the endorsement by the task force of the use of telemedicine capabilities in monitoring patients’ adherence to PAP therapy. Another interesting aspect is that, while our literature is represented by a mix of both randomized and nonrandomized controlled trials, occasionally there seems to be an interesting dichotomy in the results: Randomized trials tend to point in one direction, while nonrandomized studies pooled in the meta-analysis seem to point to the contrary or to give the impression of more definitive effects. While this is clearly not the place to make an extensive analysis of the strengths and the potential pitfalls of randomized versus nonrandomized studies, this clearly raises some issues. One is that our randomized studies are typically small, underpowered, and hence with nonconvincing risk or hazard reduction assessments. Second, the dichotomy in the results may be driven by publication bias, expense, and difficulty in performing adequately-powered, long-term trials that essentially may be studying small effects.
Guidelines are not intended to be used in an Occam’s razor approach, but in a fashion that would allow individualization of therapy while critically appraising the existing evidence for various interventions in specific conditions and maintaining a very stringent and critical view on generalizability, expected results, and adequate management of reasonable expectations. In addition, the areas that are unclear, with conflicting evidence or in which the guidelines allow “too much” latitude to the treating clinician, may be seen as either an invitation to remain “creative,” or one for abstaining from action in the name of equipoise. I would advise that both extremes are to be avoided.
Octavian C. Ioachimescu, MD, FCCP, comments: The last guidelines and practice parameters for the use of positive airway pressure (PAP) as therapy for adult patients with obstructive sleep apnea, were published in 2006 and 2008, respectively. Since then, new technological advances, an ever-growing body of literature, and shifting practice patterns led to an acute need for a thorough reassessment, a comprehensive update of the previous recommendations, and the potential of issuing new ones for emerging areas. As such, the American Academy of Sleep Medicine commissioned a task force of content experts to review the existing evidence, to issue new guidelines and to publish an associated systematic review and a meta-analysis of the literature on this topic.
A welcome recommendation is the endorsement by the task force of the use of telemedicine capabilities in monitoring patients’ adherence to PAP therapy. Another interesting aspect is that, while our literature is represented by a mix of both randomized and nonrandomized controlled trials, occasionally there seems to be an interesting dichotomy in the results: Randomized trials tend to point in one direction, while nonrandomized studies pooled in the meta-analysis seem to point to the contrary or to give the impression of more definitive effects. While this is clearly not the place to make an extensive analysis of the strengths and the potential pitfalls of randomized versus nonrandomized studies, this clearly raises some issues. One is that our randomized studies are typically small, underpowered, and hence with nonconvincing risk or hazard reduction assessments. Second, the dichotomy in the results may be driven by publication bias, expense, and difficulty in performing adequately-powered, long-term trials that essentially may be studying small effects.
Guidelines are not intended to be used in an Occam’s razor approach, but in a fashion that would allow individualization of therapy while critically appraising the existing evidence for various interventions in specific conditions and maintaining a very stringent and critical view on generalizability, expected results, and adequate management of reasonable expectations. In addition, the areas that are unclear, with conflicting evidence or in which the guidelines allow “too much” latitude to the treating clinician, may be seen as either an invitation to remain “creative,” or one for abstaining from action in the name of equipoise. I would advise that both extremes are to be avoided.
Octavian C. Ioachimescu, MD, FCCP, comments: The last guidelines and practice parameters for the use of positive airway pressure (PAP) as therapy for adult patients with obstructive sleep apnea, were published in 2006 and 2008, respectively. Since then, new technological advances, an ever-growing body of literature, and shifting practice patterns led to an acute need for a thorough reassessment, a comprehensive update of the previous recommendations, and the potential of issuing new ones for emerging areas. As such, the American Academy of Sleep Medicine commissioned a task force of content experts to review the existing evidence, to issue new guidelines and to publish an associated systematic review and a meta-analysis of the literature on this topic.
A welcome recommendation is the endorsement by the task force of the use of telemedicine capabilities in monitoring patients’ adherence to PAP therapy. Another interesting aspect is that, while our literature is represented by a mix of both randomized and nonrandomized controlled trials, occasionally there seems to be an interesting dichotomy in the results: Randomized trials tend to point in one direction, while nonrandomized studies pooled in the meta-analysis seem to point to the contrary or to give the impression of more definitive effects. While this is clearly not the place to make an extensive analysis of the strengths and the potential pitfalls of randomized versus nonrandomized studies, this clearly raises some issues. One is that our randomized studies are typically small, underpowered, and hence with nonconvincing risk or hazard reduction assessments. Second, the dichotomy in the results may be driven by publication bias, expense, and difficulty in performing adequately-powered, long-term trials that essentially may be studying small effects.
Guidelines are not intended to be used in an Occam’s razor approach, but in a fashion that would allow individualization of therapy while critically appraising the existing evidence for various interventions in specific conditions and maintaining a very stringent and critical view on generalizability, expected results, and adequate management of reasonable expectations. In addition, the areas that are unclear, with conflicting evidence or in which the guidelines allow “too much” latitude to the treating clinician, may be seen as either an invitation to remain “creative,” or one for abstaining from action in the name of equipoise. I would advise that both extremes are to be avoided.
New guidelines on treating obstructive sleep apnea with positive airway pressure include recommendations for using positive airway pressure (PAP) versus no therapy, using either continuous PAP (CPAP) or automatic PAP (APAP) for ongoing treatment, and providing educational interventions to patients starting PAP. The complete guidelines, issued by the American Academy of Sleep Medicine, were published in the Journal of Clinical Sleep Medicine.
The guidelines were driven by improvements in PAP adherence and device technology, wrote lead author Susheel P. Patil, MD, of Johns Hopkins University, Baltimore, and his colleagues.
The guidelines begin with a pair of Good Practice Statements to ensure effective and appropriate management of obstructive sleep apnea (OSA) in adults. First, “Treatment of OSA with PAP therapy should be based on a diagnosis of OSA established using objective sleep apnea testing.” Second, “Adequate follow-up, including troubleshooting and monitoring of objective efficacy and usage data to ensure adequate treatment and adherence, should occur following PAP therapy initiation and during treatment of OSA.”
The nine recommendations, approved by the AASM board of directors, include four strong recommendations that clinicians should follow under most circumstances, and five conditional recommendations that are suggested but lack strong clinical support for their appropriateness for all patients in all circumstances.
The first of the strong recommendations, for using PAP versus no therapy to treat adults with OSA and excessive sleepiness, was based on a high level of evidence from a meta-analysis of 38 randomized, controlled trials and the conclusion that the benefits of PAP outweighed the harms.
The second strong recommendation for using either CPAP or APAP for ongoing treatment was based on data from 26 trials that showed no clinically significant difference between the two. The third strong recommendation that PAP therapy be initiated using either APAP at home or in-laboratory PAP titration in adults with OSA and no significant comorbidities was supported by a meta-analysis of 10 trials that showed no clinically significant difference between at-home and laboratory initiation, and that each option has its benefits. The authors noted that “the majority of well-informed adult patients with OSA and without significant comorbidities would prefer initiation of PAP using the most rapid, convenient, and cost-effective strategy.” This comment supports the fourth strong recommendation for providing educational interventions to patients starting PAP.
The conditional recommendations include using PAP versus no therapy for adults with OSA and impaired quality of life related to poor sleep, such as insomnia, snoring, morning headaches, and daytime fatigue. Other conditional recommendations include using PAP versus no therapy for adults with OSA and comorbid hypertension, choosing CPAP or APAP over bilateral PAP for routine treatment of OSA in adults, providing behavioral interventions or troubleshooting during patients’ initial use of PAP, and using telemonitoring-guided interventions to monitor patients during their initial use of PAP.
“The ultimate judgment regarding any specific care must be made by the treating clinician and the patient, taking into consideration the individual circumstances of the patient, available treatment options, and resources,” the authors noted.
“When implementing the recommendations, providers should consider additional strategies that will maximize the individual patient’s comfort and adherence such as nasal/intranasal over oronasal mask interface and heated humidification,” they added.
The guidelines were developed by a task force commissioned by the AASM that included board-certified sleep specialists and experts in PAP use, and will be reviewed and updated as new information surfaces, the authors wrote.
Dr. Patil reported no financial conflicts; several coauthors reported conflicts that were managed by their not voting on guidelines related to those conflicts.
SOURCE: Patil SP et al. J Clin Sleep Med. 2018 Feb 15;15(2):335-43.
New guidelines on treating obstructive sleep apnea with positive airway pressure include recommendations for using positive airway pressure (PAP) versus no therapy, using either continuous PAP (CPAP) or automatic PAP (APAP) for ongoing treatment, and providing educational interventions to patients starting PAP. The complete guidelines, issued by the American Academy of Sleep Medicine, were published in the Journal of Clinical Sleep Medicine.
The guidelines were driven by improvements in PAP adherence and device technology, wrote lead author Susheel P. Patil, MD, of Johns Hopkins University, Baltimore, and his colleagues.
The guidelines begin with a pair of Good Practice Statements to ensure effective and appropriate management of obstructive sleep apnea (OSA) in adults. First, “Treatment of OSA with PAP therapy should be based on a diagnosis of OSA established using objective sleep apnea testing.” Second, “Adequate follow-up, including troubleshooting and monitoring of objective efficacy and usage data to ensure adequate treatment and adherence, should occur following PAP therapy initiation and during treatment of OSA.”
The nine recommendations, approved by the AASM board of directors, include four strong recommendations that clinicians should follow under most circumstances, and five conditional recommendations that are suggested but lack strong clinical support for their appropriateness for all patients in all circumstances.
The first of the strong recommendations, for using PAP versus no therapy to treat adults with OSA and excessive sleepiness, was based on a high level of evidence from a meta-analysis of 38 randomized, controlled trials and the conclusion that the benefits of PAP outweighed the harms.
The second strong recommendation for using either CPAP or APAP for ongoing treatment was based on data from 26 trials that showed no clinically significant difference between the two. The third strong recommendation that PAP therapy be initiated using either APAP at home or in-laboratory PAP titration in adults with OSA and no significant comorbidities was supported by a meta-analysis of 10 trials that showed no clinically significant difference between at-home and laboratory initiation, and that each option has its benefits. The authors noted that “the majority of well-informed adult patients with OSA and without significant comorbidities would prefer initiation of PAP using the most rapid, convenient, and cost-effective strategy.” This comment supports the fourth strong recommendation for providing educational interventions to patients starting PAP.
The conditional recommendations include using PAP versus no therapy for adults with OSA and impaired quality of life related to poor sleep, such as insomnia, snoring, morning headaches, and daytime fatigue. Other conditional recommendations include using PAP versus no therapy for adults with OSA and comorbid hypertension, choosing CPAP or APAP over bilateral PAP for routine treatment of OSA in adults, providing behavioral interventions or troubleshooting during patients’ initial use of PAP, and using telemonitoring-guided interventions to monitor patients during their initial use of PAP.
“The ultimate judgment regarding any specific care must be made by the treating clinician and the patient, taking into consideration the individual circumstances of the patient, available treatment options, and resources,” the authors noted.
“When implementing the recommendations, providers should consider additional strategies that will maximize the individual patient’s comfort and adherence such as nasal/intranasal over oronasal mask interface and heated humidification,” they added.
The guidelines were developed by a task force commissioned by the AASM that included board-certified sleep specialists and experts in PAP use, and will be reviewed and updated as new information surfaces, the authors wrote.
Dr. Patil reported no financial conflicts; several coauthors reported conflicts that were managed by their not voting on guidelines related to those conflicts.
SOURCE: Patil SP et al. J Clin Sleep Med. 2018 Feb 15;15(2):335-43.
FROM THE JOURNAL OF CLINICAL SLEEP MEDICINE
Geroscience brings bench science to the real-world problems of aging
NEW ORLEANS – Patients ask their doctors whether dietary manipulation can extend lifespan and promote healthy aging. Right now, basic scientists and clinicians from many disciplines are teaming up under the broad umbrella of the field of geroscience to try to answer these and other concerns relevant to an aging population.
“The idea here is that, instead of going after each disease one at a time, as we do ... [we] instead go after disease vulnerability – and this is something that is shared, as a function of age,” Rozalyn Anderson, PhD, said of this new discipline. The work touches on disparate diseases such as cancer, dementia, and diabetes, she pointed out during a video interview at the annual meeting of the Endocrine Society.
“I separate these things out into ‘front-end’ and ‘back-end,’ work,” said Dr. Anderson of the University of Wisconsin-Madison’s aging and caloric restriction program. She explained that the caloric restriction she researches is back-end work to support the rapidly evolving field of nutritional modulation of aging.
When the basic science builds the framework, physicians and scientists can turn to front-end research, looking at humans to see which dietary manipulations are effective – and which are achievable.
“Take a paradigm that works, and then try to understand how it works,” said Dr. Anderson. “So [for example], we have this paradigm, and it’s tremendously effective in rodents. It’s effective in flies, in worms, in yeast, in spiders, in dogs – and in nonhuman primates.” Then, she and her team try to pull out clues “about the biology of aging itself, and what creates disease vulnerability as a function of age,” she said.
“The most important thing of all is that we can modify aging. This is not a foregone conclusion – no one would have believed it. But even in a primate species, we can change how they age. And the way in which we change is through nutrition.”
Dr Anderson added that “the paradigm of caloric restriction is tremendously effective, but [in reality], people are not going to do it.” It’s simply not practical to ask individuals to restrict calories by 30% or more over a lifespan, so “.”
Dr. Anderson reported no relevant conflicts of interest or disclosures.
NEW ORLEANS – Patients ask their doctors whether dietary manipulation can extend lifespan and promote healthy aging. Right now, basic scientists and clinicians from many disciplines are teaming up under the broad umbrella of the field of geroscience to try to answer these and other concerns relevant to an aging population.
“The idea here is that, instead of going after each disease one at a time, as we do ... [we] instead go after disease vulnerability – and this is something that is shared, as a function of age,” Rozalyn Anderson, PhD, said of this new discipline. The work touches on disparate diseases such as cancer, dementia, and diabetes, she pointed out during a video interview at the annual meeting of the Endocrine Society.
“I separate these things out into ‘front-end’ and ‘back-end,’ work,” said Dr. Anderson of the University of Wisconsin-Madison’s aging and caloric restriction program. She explained that the caloric restriction she researches is back-end work to support the rapidly evolving field of nutritional modulation of aging.
When the basic science builds the framework, physicians and scientists can turn to front-end research, looking at humans to see which dietary manipulations are effective – and which are achievable.
“Take a paradigm that works, and then try to understand how it works,” said Dr. Anderson. “So [for example], we have this paradigm, and it’s tremendously effective in rodents. It’s effective in flies, in worms, in yeast, in spiders, in dogs – and in nonhuman primates.” Then, she and her team try to pull out clues “about the biology of aging itself, and what creates disease vulnerability as a function of age,” she said.
“The most important thing of all is that we can modify aging. This is not a foregone conclusion – no one would have believed it. But even in a primate species, we can change how they age. And the way in which we change is through nutrition.”
Dr Anderson added that “the paradigm of caloric restriction is tremendously effective, but [in reality], people are not going to do it.” It’s simply not practical to ask individuals to restrict calories by 30% or more over a lifespan, so “.”
Dr. Anderson reported no relevant conflicts of interest or disclosures.
NEW ORLEANS – Patients ask their doctors whether dietary manipulation can extend lifespan and promote healthy aging. Right now, basic scientists and clinicians from many disciplines are teaming up under the broad umbrella of the field of geroscience to try to answer these and other concerns relevant to an aging population.
“The idea here is that, instead of going after each disease one at a time, as we do ... [we] instead go after disease vulnerability – and this is something that is shared, as a function of age,” Rozalyn Anderson, PhD, said of this new discipline. The work touches on disparate diseases such as cancer, dementia, and diabetes, she pointed out during a video interview at the annual meeting of the Endocrine Society.
“I separate these things out into ‘front-end’ and ‘back-end,’ work,” said Dr. Anderson of the University of Wisconsin-Madison’s aging and caloric restriction program. She explained that the caloric restriction she researches is back-end work to support the rapidly evolving field of nutritional modulation of aging.
When the basic science builds the framework, physicians and scientists can turn to front-end research, looking at humans to see which dietary manipulations are effective – and which are achievable.
“Take a paradigm that works, and then try to understand how it works,” said Dr. Anderson. “So [for example], we have this paradigm, and it’s tremendously effective in rodents. It’s effective in flies, in worms, in yeast, in spiders, in dogs – and in nonhuman primates.” Then, she and her team try to pull out clues “about the biology of aging itself, and what creates disease vulnerability as a function of age,” she said.
“The most important thing of all is that we can modify aging. This is not a foregone conclusion – no one would have believed it. But even in a primate species, we can change how they age. And the way in which we change is through nutrition.”
Dr Anderson added that “the paradigm of caloric restriction is tremendously effective, but [in reality], people are not going to do it.” It’s simply not practical to ask individuals to restrict calories by 30% or more over a lifespan, so “.”
Dr. Anderson reported no relevant conflicts of interest or disclosures.
REPORTING FROM ENDO 2019
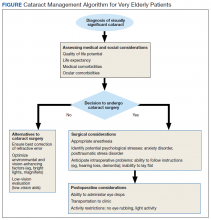
A Primary Care Provider’s Guide to Cataract Surgery in the Very Elderly
Cataract surgery is the most commonly performed surgical procedure in the US, including within the Veterans Health Administration (VHA).1,2 As the risk of surgical complications has decreased with improved techniques and instrumentation, the threshold for performing surgery has lowered.3 A substantial number of patients do not develop clinically significant cataracts until they are “very elderly,” defined as aged ≥ 85 years by the World Health Organization and National Institute of Aging.4
Should the general approach to cataract evaluation and surgery differ in this subset of patients? Advanced age is associated with a variety of systemic and ocular comorbidities that theoretically increase the risk of cataract surgery and reduce the potential visual benefit it might yield. However, the impact of age on the outcomes of cataract surgery differs even among the very elderly. There are no universally acknowledged guidelines that address the perioperative evaluation and management of cataracts in the very elderly, whose systemic and ocular health have greater variability than those of their younger counterparts. For very elderly patients who are found to have visually significant cataracts by their ophthalmologists, input from the primary care provider (PCP), who has insight into a patient’s health and well-being, is vital for formulating a management plan. Herein, we provide a framework for PCPs to assist very elderly patients and their ophthalmologists in making an informed decision regarding cataract surgery and in planning for perioperative care.
Cataract Surgery
Cataract surgeons recommend surgical extraction when there is a clinically significant lens opacity that imposes functional impairment, such as inability to read, perform near work, watch television, or drive.4 The standard of care for a clinically significant cataract is surgical removal of the crystalline lens and replacement with an artificial intraocular lens (IOL). At times, the onset of vision loss from a cataract is insidious such that patients may not be aware of their declining vision or the deterioration in quality of life (QOL) that it causes.
Despite the higher burden of ocular comorbidity (eg, age-related macular degeneration, glaucoma) relative to their younger counterparts, most very elderly patients obtain functionally important improvement in their vision, QOL, and cognitive function after surgery.5-16 Cataract surgery can also reduce the risk of dementia and the risk of falls and hip fractures.6,9,12-14,16-18 Ophthalmic complications of cataract surgery in the very elderly include posterior capsule tear (< 1%-9%), vitreous loss (< 1%-8%), zonular rupture (2%-5%), and retained lens fragments (≤ 1%).5,8-11,17,19-21 There is no evidence from well-controlled studies that suggests that very elderly cataract surgery patients are at higher risk of ocular complications relative to that of their younger counterparts.22
Surgery Alternatives
In some very elderly patients, cataract surgery may not be the best option, and PCPs can aid in establishing an alternative plan. Such patients include those with a limited life expectancy, incapacitating anxiety over surgery, or those in whom the potential for visual improvement is marginal because of ocular or systemic comorbidities—eg, vision-limiting glaucoma or age-related macular degeneration, history of stroke to the visual pathway, or restriction to bed. Alternatives to cataract surgery in these instances include changing environmental conditions to improve visual function, such as enhanced lighting and contrast, and/or use of low-vision aids (referring patients to low-vision professionals often improves QOL).23 Low-vision specialists also have a variety of nonvisual aids that can expand functional capabilities: large-print and talking versions of reading materials, telephones, remote controls, clocks, scales, calculators, and glucose monitors; glare-free lights for stairs, floors, and counters; and specialty glasses that use light-emitting diode screens and live video streams to magnify sight.23-25
Medical Evaluation
For patients who decide to proceed with surgery, it can be helpful to have a medical evaluation by their PCPs to minimize potential complications during surgery. The very elderly may be at increased risk of intraoperative transient hypertension, restlessness, and electrocardiogram abnormalities.5,7,17 Systemic comorbidities that become more prevalent with age, such as diabetes mellitus (DM), hypertension, heart disease, chronic obstructive pulmonary disease, and dementia, may adversely impact the risk of sedation and/or general anesthesia. In the VHA, providers also must be aware of combat-related disorders that can confound cataract surgery, such as posttraumatic stress disorder (PTSD), anxiety, and claustrophobia.26,27
Anesthesia in cataract surgery ranges from topical to general, and the selection largely rests on patient physical and psychological comfort and cooperation. Often, intracameral (inside the anterior eye) anesthetic is used with topical anesthesia to provide additional comfort.27 Patients who have high levels of anxiety about surgery may not tolerate topical anesthesia alone.28 In these cases, retrobulbar anesthesia may be performed to block all sensation and motility of the eye. IV sedation is performed prior to the retrobulbar injection to calm patients. Although cataract surgery is typically performed with topical or retrobulbar anesthesia (reducing the potential for systemic complications), there are cases in which general anesthesia may be considered.27 Very elderly patients may become confused or disoriented in the operating room (OR), leading to surgical complications and less than optimal outcomes.5 A higher rate of intraoperative “restlessness,” which occurred in patients who had comorbid dementia, and transient hypertension were found in a study on cataract surgery in the very elderly, but well-controlled studies are lacking.5 Dementia can impose problems with intraoperative cooperation, which is vital for successful surgery in patients who undergo topical or local anesthesia. If these potential problems are thought likely preoperatively, light sedation or general anesthesia—in conjunction with input from the patient’s PCP—are options to minimize disruptive behavior in the OR.
Additional features of the VHA population may influence the selection of anesthesia. The VHA has an important educational mission, and retrobulbar anesthesia may be preferred to minimize unpredictable intraoperative behavior in cases where resident surgeons are performing surgery under attending supervision.27,29,30 The prevalence of PTSD among veterans also may impact the selection of anesthesia. Patients with PTSD have displayed greater levels of anxiety and more discomfort, requiring more sedation and longer surgical times compared with that of a control group.28 Ophthalmic comorbidities prevalent among the predominantly older male population in the VHA include the use of α-1 antagonist prostate medications, such as tamsulosin and terazosin. These medications are associated with intraoperative floppy iris syndrome, which can increase case difficulty and prolong operative time.29
Surgery Preparation
Cataract surgery induces minimal physiologic stress since most surgeries are performed under local or topical anesthesia. Unless the preoperative medical history or physical examination detects an active or unattended medical condition that needs to be addressed, preoperative laboratory testing is generally not required.31-33 Current general guidelines for preoperative testing for cataract surgery exist but do not address specific issues facing very elderly patients. The American Academy of Ophthalmology advises against preoperative medical tests for eye surgery unless there are medical indications: an electrocardiogram for patients with a history of heart disease, a blood glucose test for those with DM, and a potassium test for patients who are on diuretics.31 The direct correlation of age with these comorbidities may translate into higher rates of preoperative testing among very elderly patients. In the VHA, 45% of ophthalmology services studied routinely performed preoperative electrocardiography, chemical analysis, and complete blood counts prior to performing cataract surgery.27 Patients who live with chronic bacterial colonization from indwelling catheters, ostomies, or bed sores need to be given instructions for proper hygienic practices to minimize risks of postoperative infection.34
Some patients undergoing cataract surgery may not be candidates for topical or local anesthesia alone. Sedation is often used to reduce anxiety and discomfort of surgery, but very elderly patients have narrower margins of therapeutic safety because of advanced aged or medical comorbidities. Since patients need to follow basic commands in the OR for ideal surgical execution, general anesthesia may need to be considered for those with dementia, deafness, anxiety attacks, or language barriers.
Postsurgical Care
Although cataract surgery is a less invasive procedure than it was in the past, full postoperative recovery regularly spans a month. During this time, proper healing relies on the regular administration of eye drops and a refrain from heavy lifting, straining, and eye rubbing. Very elderly patients may need varying degrees of assistance with postsurgical care. For example, adherence to the regimen of eye drops can be complicated by decreased dexterity from arthritis and difficulty remembering the administration schedule in some patients. Reliable transportation also is an important factor as patients are routinely scheduled for postoperative visits at the 1- day, 1-week, and 1-month mark. PCPs can assist in ensuring patients have prearranged assistance for eye care and transportation to and from appointments. Additionally, very elderly patients with a history of constipation may benefit from stool softeners and/or laxatives to help prevent straining.
Conclusion
The limited literature on clinical outcomes of cataract surgery in the very elderly indicates that most have successful surgery and improved postoperative QOL.22 Much of the benefits derived from cataract surgery in the very elderly can be ascribed to thoughtful preoperative evaluation and planning with the PCP.
1. US Census Bureau. An aging nation: the older population in the United States. https://www.census.gov/library/publications/2014/demo/p25-1140.html Published May 2014. Accessed March 18, 2019.
2. VA Office of Inspector General. Healthcare inspection: evaluation of cataract surgeries and outcomes in veterans health administration facilities. Report No. 11-02487-158. https://www.va.gov/oig/pubs/vaoig-11-02487-158.pdf. Published March 28, 2013. Accessed March 11, 2019.
3. Lee CM, Afshari NA. The global state of cataract blindness. Curr Opin Ophthalmol. 2017;28(1):98-103.
4. American Academy of Ophthalmology. Cataract in the adult eye preferred practice pattern—2016. https://www.aao.org/preferred-practice-pattern/cataract-in-adult-eye-ppp-2016. Published October 2016. Accessed March 19, 2019.
5. Mutoh T, Isome S, Matsumoto Y, Chikuda M. Cataract surgery in patients older than 90 years of age. Can J Ophthalmol. 2012;47(2):140-144.
6. Monestam E, Wachmeister L. Impact of cataract surgery on the visual ability of the very old. Am J Ophthalmol. 2004;137(1):145-155.
7. Lai FH, Lok JY, Chow PP, Young AL. Clinical outcomes of cataract surgery in very elderly adults. J Am Geriatr Soc. 2014;62(1):165-170.
8. Michalska-Malecka K, Nowak M, Gos´ciniewicz P, et al. Results of cataract surgery in the very elderly population. Clin Interv Aging. 2013;8:1041-1046.
9. Syam PP, Eleftheriadis H, Casswell AG, Brittain GP, McLeod BK, Liu CS. Clinical outcome following cataract surgery in very elderly patients. Eye (Lond). 2004;18(1):59-62.
10. Rosen E, Rubowitz A, Assia EI. Visual outcome following cataract extraction in patients aged 90 years and older. Eye (Lond). 2009;23(5):1120-1124.
11. Mehmet B, Abuzer G. Results of cataract surgery in the very elderly population. J Optom. 2009;2(3):138-141.
12. To KG, Meuleners L, Bulsara M, et al. A longitudinal cohort study of the impact of first- and both-eye cataract surgery on falls and other injuries in Vietnam. Clin Interv Aging. 2014;9:743-751.
13. Song E, Sun H, Xu Y, Ma Y, Zhu H, Pan CW. Age-related cataract, cataract surgery and subsequent mortality: a systematic review and meta-analysis. PLoS One. 2014;9(11):e112054.
14. Brannan S, Dewar C, Sen J, Clarke D, Marshall T, Murray PI. A prospective study of the rate of falls before and after cataract surgery. Br J Ophthalmol. 2003;87(5):560-562.
15. Jefferis JM, Mosimann UP, Clarke MP. Cataract and cognitive impairment: a review of the literature. Br J Ophthalmol. 2011;95(1):17-23.
16. Yu WK, Chen YT, Wang SJ, Kuo SC, Shia BC, Liu CJ. Cataract surgery is associated with a reduced risk of dementia: a nationwide population-based cohort study. Eur J Neurol. 2015;22(10):1370-1377, e1379-1380.
17. Tseng VL, Greenberg PB, Wu WC, et al. Cataract surgery complications in nonagenarians. Ophthalmology. 2011;118(7):1229-1235.
18. Jefferis JM, Clarke MP, Taylor JP. Effect of cataract surgery on cognition, mood, and visual hallucinations in older adults. J Cataract Refract Surg. 2015;41(6):1241-1247.
19. Celebi AR. The relationship between age and the intraoperative complication rate during phacoemulsification surgery. Aging Clin Exp Res. 2014;26(2):177-181.
20. Berler DK. Intraoperative complications during cataract surgery in the very old. Trans Am Ophthalmol Soc. 2000;98:127-130; discussion 130-132.
21. Lai FHP, Lok JYC, Chow PPC, Young AL. Clinical outcomes of cataract surgery in very elderly adults. J Am Geriatr Soc. 2014;62(1):165-170.
22. Li E, Margo CE, Greenberg PB. Cataract surgery outcomes in the very elderly. J Cataract Refract Surg. 2018;44(9):1144-1149.
23. Young JS. Age-related eye diseases and recommendations for low-vision AIDS. Home Healthc Now. 2015;33(1):10-17; quiz 18-19.
24. Virgili G, Acosta R, Grover LL, Bentley SA, Giacomelli G. Reading aids for adults with low vision. Cochrane Database Syst Rev. 2013;(10):CD003303.
25. Young JS. Age-related eye diseases: a review of current treatment and recommendations for low-vision aids. Home Healthc Nurse. 2008;26(8):464-471; quiz 472-473.
26. Thomas MM, Harpaz-Rotem I, Tsai J, Southwick SM, Pietrzak RH. Mental and physical health conditions in US combat veterans: results from the National Health and Resilience in Veterans study. Prim Care Companion CNS Disord. 2017;19(3):17m02118.
27. Havnaer AG, Greenberg PB, Cockerham GC, Clark MA, Chomsky A. Cataract surgery practices in the United States Veterans Health Administration. J Cataract Refract Surg. 2017;43(4):543-551.
28. Rapoport Y, Wayman LL, Chomsky AS. The effect of post-traumatic-stress-disorder on intra-operative analgesia in a veteran population during cataract procedures carried out using retrobulbar or topical anesthesia: a retrospective study. BMC Ophthalmol. 2017;17(1):85.
29. Payal AR, Gonzalez-Gonzalez LA, Chen X, et al. Outcomes of cataract surgery with residents as primary surgeons in the Veterans Affairs Healthcare System. J Cataract Refract Surg. 2016;42(3):370-384.
30. US Department of Veterans Affairs. Mission of the office of academic affiliations. https://www.va.gov/oaa/oaa_mission.asp. Updated November 30, 2018. Accessed March 18, 2019.
31. American Academy of Ophthalmology. Choosing wisely: five things ophthalmologists and patients should question. https://www.aao.org/choosing-wisely. Published February 2013. Accessed March 18, 2019.
32. Martin SK, Cifu AS. Routine preoperative laboratory tests for elective surgery. JAMA. 2017;318(6):567-568.
33. Schein OD, Katz J, Bass EB, et al; Study of Medical Testing for Cataract Surgery. The value of routine preoperative medical testing before cataract surgery. N Engl J Med. 2000;342(3):168-175.
34. Margo CE. Asymptomatic bacteriuria and acute-onset endophthalmitis after cataract surgery. Can J Ophthalmol. 2015;50(4):e51-52.
35. Fukui K, Fujioka M, Yamasaki K, Yamakawa S, Matsuo H, Noguchi M. Risk factors for postoperative complications among the elderly after plastic surgery procedures performed under general anesthesia. Plast Surg Int. 2018:7053839.
Cataract surgery is the most commonly performed surgical procedure in the US, including within the Veterans Health Administration (VHA).1,2 As the risk of surgical complications has decreased with improved techniques and instrumentation, the threshold for performing surgery has lowered.3 A substantial number of patients do not develop clinically significant cataracts until they are “very elderly,” defined as aged ≥ 85 years by the World Health Organization and National Institute of Aging.4
Should the general approach to cataract evaluation and surgery differ in this subset of patients? Advanced age is associated with a variety of systemic and ocular comorbidities that theoretically increase the risk of cataract surgery and reduce the potential visual benefit it might yield. However, the impact of age on the outcomes of cataract surgery differs even among the very elderly. There are no universally acknowledged guidelines that address the perioperative evaluation and management of cataracts in the very elderly, whose systemic and ocular health have greater variability than those of their younger counterparts. For very elderly patients who are found to have visually significant cataracts by their ophthalmologists, input from the primary care provider (PCP), who has insight into a patient’s health and well-being, is vital for formulating a management plan. Herein, we provide a framework for PCPs to assist very elderly patients and their ophthalmologists in making an informed decision regarding cataract surgery and in planning for perioperative care.
Cataract Surgery
Cataract surgeons recommend surgical extraction when there is a clinically significant lens opacity that imposes functional impairment, such as inability to read, perform near work, watch television, or drive.4 The standard of care for a clinically significant cataract is surgical removal of the crystalline lens and replacement with an artificial intraocular lens (IOL). At times, the onset of vision loss from a cataract is insidious such that patients may not be aware of their declining vision or the deterioration in quality of life (QOL) that it causes.
Despite the higher burden of ocular comorbidity (eg, age-related macular degeneration, glaucoma) relative to their younger counterparts, most very elderly patients obtain functionally important improvement in their vision, QOL, and cognitive function after surgery.5-16 Cataract surgery can also reduce the risk of dementia and the risk of falls and hip fractures.6,9,12-14,16-18 Ophthalmic complications of cataract surgery in the very elderly include posterior capsule tear (< 1%-9%), vitreous loss (< 1%-8%), zonular rupture (2%-5%), and retained lens fragments (≤ 1%).5,8-11,17,19-21 There is no evidence from well-controlled studies that suggests that very elderly cataract surgery patients are at higher risk of ocular complications relative to that of their younger counterparts.22
Surgery Alternatives
In some very elderly patients, cataract surgery may not be the best option, and PCPs can aid in establishing an alternative plan. Such patients include those with a limited life expectancy, incapacitating anxiety over surgery, or those in whom the potential for visual improvement is marginal because of ocular or systemic comorbidities—eg, vision-limiting glaucoma or age-related macular degeneration, history of stroke to the visual pathway, or restriction to bed. Alternatives to cataract surgery in these instances include changing environmental conditions to improve visual function, such as enhanced lighting and contrast, and/or use of low-vision aids (referring patients to low-vision professionals often improves QOL).23 Low-vision specialists also have a variety of nonvisual aids that can expand functional capabilities: large-print and talking versions of reading materials, telephones, remote controls, clocks, scales, calculators, and glucose monitors; glare-free lights for stairs, floors, and counters; and specialty glasses that use light-emitting diode screens and live video streams to magnify sight.23-25
Medical Evaluation
For patients who decide to proceed with surgery, it can be helpful to have a medical evaluation by their PCPs to minimize potential complications during surgery. The very elderly may be at increased risk of intraoperative transient hypertension, restlessness, and electrocardiogram abnormalities.5,7,17 Systemic comorbidities that become more prevalent with age, such as diabetes mellitus (DM), hypertension, heart disease, chronic obstructive pulmonary disease, and dementia, may adversely impact the risk of sedation and/or general anesthesia. In the VHA, providers also must be aware of combat-related disorders that can confound cataract surgery, such as posttraumatic stress disorder (PTSD), anxiety, and claustrophobia.26,27
Anesthesia in cataract surgery ranges from topical to general, and the selection largely rests on patient physical and psychological comfort and cooperation. Often, intracameral (inside the anterior eye) anesthetic is used with topical anesthesia to provide additional comfort.27 Patients who have high levels of anxiety about surgery may not tolerate topical anesthesia alone.28 In these cases, retrobulbar anesthesia may be performed to block all sensation and motility of the eye. IV sedation is performed prior to the retrobulbar injection to calm patients. Although cataract surgery is typically performed with topical or retrobulbar anesthesia (reducing the potential for systemic complications), there are cases in which general anesthesia may be considered.27 Very elderly patients may become confused or disoriented in the operating room (OR), leading to surgical complications and less than optimal outcomes.5 A higher rate of intraoperative “restlessness,” which occurred in patients who had comorbid dementia, and transient hypertension were found in a study on cataract surgery in the very elderly, but well-controlled studies are lacking.5 Dementia can impose problems with intraoperative cooperation, which is vital for successful surgery in patients who undergo topical or local anesthesia. If these potential problems are thought likely preoperatively, light sedation or general anesthesia—in conjunction with input from the patient’s PCP—are options to minimize disruptive behavior in the OR.
Additional features of the VHA population may influence the selection of anesthesia. The VHA has an important educational mission, and retrobulbar anesthesia may be preferred to minimize unpredictable intraoperative behavior in cases where resident surgeons are performing surgery under attending supervision.27,29,30 The prevalence of PTSD among veterans also may impact the selection of anesthesia. Patients with PTSD have displayed greater levels of anxiety and more discomfort, requiring more sedation and longer surgical times compared with that of a control group.28 Ophthalmic comorbidities prevalent among the predominantly older male population in the VHA include the use of α-1 antagonist prostate medications, such as tamsulosin and terazosin. These medications are associated with intraoperative floppy iris syndrome, which can increase case difficulty and prolong operative time.29
Surgery Preparation
Cataract surgery induces minimal physiologic stress since most surgeries are performed under local or topical anesthesia. Unless the preoperative medical history or physical examination detects an active or unattended medical condition that needs to be addressed, preoperative laboratory testing is generally not required.31-33 Current general guidelines for preoperative testing for cataract surgery exist but do not address specific issues facing very elderly patients. The American Academy of Ophthalmology advises against preoperative medical tests for eye surgery unless there are medical indications: an electrocardiogram for patients with a history of heart disease, a blood glucose test for those with DM, and a potassium test for patients who are on diuretics.31 The direct correlation of age with these comorbidities may translate into higher rates of preoperative testing among very elderly patients. In the VHA, 45% of ophthalmology services studied routinely performed preoperative electrocardiography, chemical analysis, and complete blood counts prior to performing cataract surgery.27 Patients who live with chronic bacterial colonization from indwelling catheters, ostomies, or bed sores need to be given instructions for proper hygienic practices to minimize risks of postoperative infection.34
Some patients undergoing cataract surgery may not be candidates for topical or local anesthesia alone. Sedation is often used to reduce anxiety and discomfort of surgery, but very elderly patients have narrower margins of therapeutic safety because of advanced aged or medical comorbidities. Since patients need to follow basic commands in the OR for ideal surgical execution, general anesthesia may need to be considered for those with dementia, deafness, anxiety attacks, or language barriers.
Postsurgical Care
Although cataract surgery is a less invasive procedure than it was in the past, full postoperative recovery regularly spans a month. During this time, proper healing relies on the regular administration of eye drops and a refrain from heavy lifting, straining, and eye rubbing. Very elderly patients may need varying degrees of assistance with postsurgical care. For example, adherence to the regimen of eye drops can be complicated by decreased dexterity from arthritis and difficulty remembering the administration schedule in some patients. Reliable transportation also is an important factor as patients are routinely scheduled for postoperative visits at the 1- day, 1-week, and 1-month mark. PCPs can assist in ensuring patients have prearranged assistance for eye care and transportation to and from appointments. Additionally, very elderly patients with a history of constipation may benefit from stool softeners and/or laxatives to help prevent straining.
Conclusion
The limited literature on clinical outcomes of cataract surgery in the very elderly indicates that most have successful surgery and improved postoperative QOL.22 Much of the benefits derived from cataract surgery in the very elderly can be ascribed to thoughtful preoperative evaluation and planning with the PCP.
Cataract surgery is the most commonly performed surgical procedure in the US, including within the Veterans Health Administration (VHA).1,2 As the risk of surgical complications has decreased with improved techniques and instrumentation, the threshold for performing surgery has lowered.3 A substantial number of patients do not develop clinically significant cataracts until they are “very elderly,” defined as aged ≥ 85 years by the World Health Organization and National Institute of Aging.4
Should the general approach to cataract evaluation and surgery differ in this subset of patients? Advanced age is associated with a variety of systemic and ocular comorbidities that theoretically increase the risk of cataract surgery and reduce the potential visual benefit it might yield. However, the impact of age on the outcomes of cataract surgery differs even among the very elderly. There are no universally acknowledged guidelines that address the perioperative evaluation and management of cataracts in the very elderly, whose systemic and ocular health have greater variability than those of their younger counterparts. For very elderly patients who are found to have visually significant cataracts by their ophthalmologists, input from the primary care provider (PCP), who has insight into a patient’s health and well-being, is vital for formulating a management plan. Herein, we provide a framework for PCPs to assist very elderly patients and their ophthalmologists in making an informed decision regarding cataract surgery and in planning for perioperative care.
Cataract Surgery
Cataract surgeons recommend surgical extraction when there is a clinically significant lens opacity that imposes functional impairment, such as inability to read, perform near work, watch television, or drive.4 The standard of care for a clinically significant cataract is surgical removal of the crystalline lens and replacement with an artificial intraocular lens (IOL). At times, the onset of vision loss from a cataract is insidious such that patients may not be aware of their declining vision or the deterioration in quality of life (QOL) that it causes.
Despite the higher burden of ocular comorbidity (eg, age-related macular degeneration, glaucoma) relative to their younger counterparts, most very elderly patients obtain functionally important improvement in their vision, QOL, and cognitive function after surgery.5-16 Cataract surgery can also reduce the risk of dementia and the risk of falls and hip fractures.6,9,12-14,16-18 Ophthalmic complications of cataract surgery in the very elderly include posterior capsule tear (< 1%-9%), vitreous loss (< 1%-8%), zonular rupture (2%-5%), and retained lens fragments (≤ 1%).5,8-11,17,19-21 There is no evidence from well-controlled studies that suggests that very elderly cataract surgery patients are at higher risk of ocular complications relative to that of their younger counterparts.22
Surgery Alternatives
In some very elderly patients, cataract surgery may not be the best option, and PCPs can aid in establishing an alternative plan. Such patients include those with a limited life expectancy, incapacitating anxiety over surgery, or those in whom the potential for visual improvement is marginal because of ocular or systemic comorbidities—eg, vision-limiting glaucoma or age-related macular degeneration, history of stroke to the visual pathway, or restriction to bed. Alternatives to cataract surgery in these instances include changing environmental conditions to improve visual function, such as enhanced lighting and contrast, and/or use of low-vision aids (referring patients to low-vision professionals often improves QOL).23 Low-vision specialists also have a variety of nonvisual aids that can expand functional capabilities: large-print and talking versions of reading materials, telephones, remote controls, clocks, scales, calculators, and glucose monitors; glare-free lights for stairs, floors, and counters; and specialty glasses that use light-emitting diode screens and live video streams to magnify sight.23-25
Medical Evaluation
For patients who decide to proceed with surgery, it can be helpful to have a medical evaluation by their PCPs to minimize potential complications during surgery. The very elderly may be at increased risk of intraoperative transient hypertension, restlessness, and electrocardiogram abnormalities.5,7,17 Systemic comorbidities that become more prevalent with age, such as diabetes mellitus (DM), hypertension, heart disease, chronic obstructive pulmonary disease, and dementia, may adversely impact the risk of sedation and/or general anesthesia. In the VHA, providers also must be aware of combat-related disorders that can confound cataract surgery, such as posttraumatic stress disorder (PTSD), anxiety, and claustrophobia.26,27
Anesthesia in cataract surgery ranges from topical to general, and the selection largely rests on patient physical and psychological comfort and cooperation. Often, intracameral (inside the anterior eye) anesthetic is used with topical anesthesia to provide additional comfort.27 Patients who have high levels of anxiety about surgery may not tolerate topical anesthesia alone.28 In these cases, retrobulbar anesthesia may be performed to block all sensation and motility of the eye. IV sedation is performed prior to the retrobulbar injection to calm patients. Although cataract surgery is typically performed with topical or retrobulbar anesthesia (reducing the potential for systemic complications), there are cases in which general anesthesia may be considered.27 Very elderly patients may become confused or disoriented in the operating room (OR), leading to surgical complications and less than optimal outcomes.5 A higher rate of intraoperative “restlessness,” which occurred in patients who had comorbid dementia, and transient hypertension were found in a study on cataract surgery in the very elderly, but well-controlled studies are lacking.5 Dementia can impose problems with intraoperative cooperation, which is vital for successful surgery in patients who undergo topical or local anesthesia. If these potential problems are thought likely preoperatively, light sedation or general anesthesia—in conjunction with input from the patient’s PCP—are options to minimize disruptive behavior in the OR.
Additional features of the VHA population may influence the selection of anesthesia. The VHA has an important educational mission, and retrobulbar anesthesia may be preferred to minimize unpredictable intraoperative behavior in cases where resident surgeons are performing surgery under attending supervision.27,29,30 The prevalence of PTSD among veterans also may impact the selection of anesthesia. Patients with PTSD have displayed greater levels of anxiety and more discomfort, requiring more sedation and longer surgical times compared with that of a control group.28 Ophthalmic comorbidities prevalent among the predominantly older male population in the VHA include the use of α-1 antagonist prostate medications, such as tamsulosin and terazosin. These medications are associated with intraoperative floppy iris syndrome, which can increase case difficulty and prolong operative time.29
Surgery Preparation
Cataract surgery induces minimal physiologic stress since most surgeries are performed under local or topical anesthesia. Unless the preoperative medical history or physical examination detects an active or unattended medical condition that needs to be addressed, preoperative laboratory testing is generally not required.31-33 Current general guidelines for preoperative testing for cataract surgery exist but do not address specific issues facing very elderly patients. The American Academy of Ophthalmology advises against preoperative medical tests for eye surgery unless there are medical indications: an electrocardiogram for patients with a history of heart disease, a blood glucose test for those with DM, and a potassium test for patients who are on diuretics.31 The direct correlation of age with these comorbidities may translate into higher rates of preoperative testing among very elderly patients. In the VHA, 45% of ophthalmology services studied routinely performed preoperative electrocardiography, chemical analysis, and complete blood counts prior to performing cataract surgery.27 Patients who live with chronic bacterial colonization from indwelling catheters, ostomies, or bed sores need to be given instructions for proper hygienic practices to minimize risks of postoperative infection.34
Some patients undergoing cataract surgery may not be candidates for topical or local anesthesia alone. Sedation is often used to reduce anxiety and discomfort of surgery, but very elderly patients have narrower margins of therapeutic safety because of advanced aged or medical comorbidities. Since patients need to follow basic commands in the OR for ideal surgical execution, general anesthesia may need to be considered for those with dementia, deafness, anxiety attacks, or language barriers.
Postsurgical Care
Although cataract surgery is a less invasive procedure than it was in the past, full postoperative recovery regularly spans a month. During this time, proper healing relies on the regular administration of eye drops and a refrain from heavy lifting, straining, and eye rubbing. Very elderly patients may need varying degrees of assistance with postsurgical care. For example, adherence to the regimen of eye drops can be complicated by decreased dexterity from arthritis and difficulty remembering the administration schedule in some patients. Reliable transportation also is an important factor as patients are routinely scheduled for postoperative visits at the 1- day, 1-week, and 1-month mark. PCPs can assist in ensuring patients have prearranged assistance for eye care and transportation to and from appointments. Additionally, very elderly patients with a history of constipation may benefit from stool softeners and/or laxatives to help prevent straining.
Conclusion
The limited literature on clinical outcomes of cataract surgery in the very elderly indicates that most have successful surgery and improved postoperative QOL.22 Much of the benefits derived from cataract surgery in the very elderly can be ascribed to thoughtful preoperative evaluation and planning with the PCP.
1. US Census Bureau. An aging nation: the older population in the United States. https://www.census.gov/library/publications/2014/demo/p25-1140.html Published May 2014. Accessed March 18, 2019.
2. VA Office of Inspector General. Healthcare inspection: evaluation of cataract surgeries and outcomes in veterans health administration facilities. Report No. 11-02487-158. https://www.va.gov/oig/pubs/vaoig-11-02487-158.pdf. Published March 28, 2013. Accessed March 11, 2019.
3. Lee CM, Afshari NA. The global state of cataract blindness. Curr Opin Ophthalmol. 2017;28(1):98-103.
4. American Academy of Ophthalmology. Cataract in the adult eye preferred practice pattern—2016. https://www.aao.org/preferred-practice-pattern/cataract-in-adult-eye-ppp-2016. Published October 2016. Accessed March 19, 2019.
5. Mutoh T, Isome S, Matsumoto Y, Chikuda M. Cataract surgery in patients older than 90 years of age. Can J Ophthalmol. 2012;47(2):140-144.
6. Monestam E, Wachmeister L. Impact of cataract surgery on the visual ability of the very old. Am J Ophthalmol. 2004;137(1):145-155.
7. Lai FH, Lok JY, Chow PP, Young AL. Clinical outcomes of cataract surgery in very elderly adults. J Am Geriatr Soc. 2014;62(1):165-170.
8. Michalska-Malecka K, Nowak M, Gos´ciniewicz P, et al. Results of cataract surgery in the very elderly population. Clin Interv Aging. 2013;8:1041-1046.
9. Syam PP, Eleftheriadis H, Casswell AG, Brittain GP, McLeod BK, Liu CS. Clinical outcome following cataract surgery in very elderly patients. Eye (Lond). 2004;18(1):59-62.
10. Rosen E, Rubowitz A, Assia EI. Visual outcome following cataract extraction in patients aged 90 years and older. Eye (Lond). 2009;23(5):1120-1124.
11. Mehmet B, Abuzer G. Results of cataract surgery in the very elderly population. J Optom. 2009;2(3):138-141.
12. To KG, Meuleners L, Bulsara M, et al. A longitudinal cohort study of the impact of first- and both-eye cataract surgery on falls and other injuries in Vietnam. Clin Interv Aging. 2014;9:743-751.
13. Song E, Sun H, Xu Y, Ma Y, Zhu H, Pan CW. Age-related cataract, cataract surgery and subsequent mortality: a systematic review and meta-analysis. PLoS One. 2014;9(11):e112054.
14. Brannan S, Dewar C, Sen J, Clarke D, Marshall T, Murray PI. A prospective study of the rate of falls before and after cataract surgery. Br J Ophthalmol. 2003;87(5):560-562.
15. Jefferis JM, Mosimann UP, Clarke MP. Cataract and cognitive impairment: a review of the literature. Br J Ophthalmol. 2011;95(1):17-23.
16. Yu WK, Chen YT, Wang SJ, Kuo SC, Shia BC, Liu CJ. Cataract surgery is associated with a reduced risk of dementia: a nationwide population-based cohort study. Eur J Neurol. 2015;22(10):1370-1377, e1379-1380.
17. Tseng VL, Greenberg PB, Wu WC, et al. Cataract surgery complications in nonagenarians. Ophthalmology. 2011;118(7):1229-1235.
18. Jefferis JM, Clarke MP, Taylor JP. Effect of cataract surgery on cognition, mood, and visual hallucinations in older adults. J Cataract Refract Surg. 2015;41(6):1241-1247.
19. Celebi AR. The relationship between age and the intraoperative complication rate during phacoemulsification surgery. Aging Clin Exp Res. 2014;26(2):177-181.
20. Berler DK. Intraoperative complications during cataract surgery in the very old. Trans Am Ophthalmol Soc. 2000;98:127-130; discussion 130-132.
21. Lai FHP, Lok JYC, Chow PPC, Young AL. Clinical outcomes of cataract surgery in very elderly adults. J Am Geriatr Soc. 2014;62(1):165-170.
22. Li E, Margo CE, Greenberg PB. Cataract surgery outcomes in the very elderly. J Cataract Refract Surg. 2018;44(9):1144-1149.
23. Young JS. Age-related eye diseases and recommendations for low-vision AIDS. Home Healthc Now. 2015;33(1):10-17; quiz 18-19.
24. Virgili G, Acosta R, Grover LL, Bentley SA, Giacomelli G. Reading aids for adults with low vision. Cochrane Database Syst Rev. 2013;(10):CD003303.
25. Young JS. Age-related eye diseases: a review of current treatment and recommendations for low-vision aids. Home Healthc Nurse. 2008;26(8):464-471; quiz 472-473.
26. Thomas MM, Harpaz-Rotem I, Tsai J, Southwick SM, Pietrzak RH. Mental and physical health conditions in US combat veterans: results from the National Health and Resilience in Veterans study. Prim Care Companion CNS Disord. 2017;19(3):17m02118.
27. Havnaer AG, Greenberg PB, Cockerham GC, Clark MA, Chomsky A. Cataract surgery practices in the United States Veterans Health Administration. J Cataract Refract Surg. 2017;43(4):543-551.
28. Rapoport Y, Wayman LL, Chomsky AS. The effect of post-traumatic-stress-disorder on intra-operative analgesia in a veteran population during cataract procedures carried out using retrobulbar or topical anesthesia: a retrospective study. BMC Ophthalmol. 2017;17(1):85.
29. Payal AR, Gonzalez-Gonzalez LA, Chen X, et al. Outcomes of cataract surgery with residents as primary surgeons in the Veterans Affairs Healthcare System. J Cataract Refract Surg. 2016;42(3):370-384.
30. US Department of Veterans Affairs. Mission of the office of academic affiliations. https://www.va.gov/oaa/oaa_mission.asp. Updated November 30, 2018. Accessed March 18, 2019.
31. American Academy of Ophthalmology. Choosing wisely: five things ophthalmologists and patients should question. https://www.aao.org/choosing-wisely. Published February 2013. Accessed March 18, 2019.
32. Martin SK, Cifu AS. Routine preoperative laboratory tests for elective surgery. JAMA. 2017;318(6):567-568.
33. Schein OD, Katz J, Bass EB, et al; Study of Medical Testing for Cataract Surgery. The value of routine preoperative medical testing before cataract surgery. N Engl J Med. 2000;342(3):168-175.
34. Margo CE. Asymptomatic bacteriuria and acute-onset endophthalmitis after cataract surgery. Can J Ophthalmol. 2015;50(4):e51-52.
35. Fukui K, Fujioka M, Yamasaki K, Yamakawa S, Matsuo H, Noguchi M. Risk factors for postoperative complications among the elderly after plastic surgery procedures performed under general anesthesia. Plast Surg Int. 2018:7053839.
1. US Census Bureau. An aging nation: the older population in the United States. https://www.census.gov/library/publications/2014/demo/p25-1140.html Published May 2014. Accessed March 18, 2019.
2. VA Office of Inspector General. Healthcare inspection: evaluation of cataract surgeries and outcomes in veterans health administration facilities. Report No. 11-02487-158. https://www.va.gov/oig/pubs/vaoig-11-02487-158.pdf. Published March 28, 2013. Accessed March 11, 2019.
3. Lee CM, Afshari NA. The global state of cataract blindness. Curr Opin Ophthalmol. 2017;28(1):98-103.
4. American Academy of Ophthalmology. Cataract in the adult eye preferred practice pattern—2016. https://www.aao.org/preferred-practice-pattern/cataract-in-adult-eye-ppp-2016. Published October 2016. Accessed March 19, 2019.
5. Mutoh T, Isome S, Matsumoto Y, Chikuda M. Cataract surgery in patients older than 90 years of age. Can J Ophthalmol. 2012;47(2):140-144.
6. Monestam E, Wachmeister L. Impact of cataract surgery on the visual ability of the very old. Am J Ophthalmol. 2004;137(1):145-155.
7. Lai FH, Lok JY, Chow PP, Young AL. Clinical outcomes of cataract surgery in very elderly adults. J Am Geriatr Soc. 2014;62(1):165-170.
8. Michalska-Malecka K, Nowak M, Gos´ciniewicz P, et al. Results of cataract surgery in the very elderly population. Clin Interv Aging. 2013;8:1041-1046.
9. Syam PP, Eleftheriadis H, Casswell AG, Brittain GP, McLeod BK, Liu CS. Clinical outcome following cataract surgery in very elderly patients. Eye (Lond). 2004;18(1):59-62.
10. Rosen E, Rubowitz A, Assia EI. Visual outcome following cataract extraction in patients aged 90 years and older. Eye (Lond). 2009;23(5):1120-1124.
11. Mehmet B, Abuzer G. Results of cataract surgery in the very elderly population. J Optom. 2009;2(3):138-141.
12. To KG, Meuleners L, Bulsara M, et al. A longitudinal cohort study of the impact of first- and both-eye cataract surgery on falls and other injuries in Vietnam. Clin Interv Aging. 2014;9:743-751.
13. Song E, Sun H, Xu Y, Ma Y, Zhu H, Pan CW. Age-related cataract, cataract surgery and subsequent mortality: a systematic review and meta-analysis. PLoS One. 2014;9(11):e112054.
14. Brannan S, Dewar C, Sen J, Clarke D, Marshall T, Murray PI. A prospective study of the rate of falls before and after cataract surgery. Br J Ophthalmol. 2003;87(5):560-562.
15. Jefferis JM, Mosimann UP, Clarke MP. Cataract and cognitive impairment: a review of the literature. Br J Ophthalmol. 2011;95(1):17-23.
16. Yu WK, Chen YT, Wang SJ, Kuo SC, Shia BC, Liu CJ. Cataract surgery is associated with a reduced risk of dementia: a nationwide population-based cohort study. Eur J Neurol. 2015;22(10):1370-1377, e1379-1380.
17. Tseng VL, Greenberg PB, Wu WC, et al. Cataract surgery complications in nonagenarians. Ophthalmology. 2011;118(7):1229-1235.
18. Jefferis JM, Clarke MP, Taylor JP. Effect of cataract surgery on cognition, mood, and visual hallucinations in older adults. J Cataract Refract Surg. 2015;41(6):1241-1247.
19. Celebi AR. The relationship between age and the intraoperative complication rate during phacoemulsification surgery. Aging Clin Exp Res. 2014;26(2):177-181.
20. Berler DK. Intraoperative complications during cataract surgery in the very old. Trans Am Ophthalmol Soc. 2000;98:127-130; discussion 130-132.
21. Lai FHP, Lok JYC, Chow PPC, Young AL. Clinical outcomes of cataract surgery in very elderly adults. J Am Geriatr Soc. 2014;62(1):165-170.
22. Li E, Margo CE, Greenberg PB. Cataract surgery outcomes in the very elderly. J Cataract Refract Surg. 2018;44(9):1144-1149.
23. Young JS. Age-related eye diseases and recommendations for low-vision AIDS. Home Healthc Now. 2015;33(1):10-17; quiz 18-19.
24. Virgili G, Acosta R, Grover LL, Bentley SA, Giacomelli G. Reading aids for adults with low vision. Cochrane Database Syst Rev. 2013;(10):CD003303.
25. Young JS. Age-related eye diseases: a review of current treatment and recommendations for low-vision aids. Home Healthc Nurse. 2008;26(8):464-471; quiz 472-473.
26. Thomas MM, Harpaz-Rotem I, Tsai J, Southwick SM, Pietrzak RH. Mental and physical health conditions in US combat veterans: results from the National Health and Resilience in Veterans study. Prim Care Companion CNS Disord. 2017;19(3):17m02118.
27. Havnaer AG, Greenberg PB, Cockerham GC, Clark MA, Chomsky A. Cataract surgery practices in the United States Veterans Health Administration. J Cataract Refract Surg. 2017;43(4):543-551.
28. Rapoport Y, Wayman LL, Chomsky AS. The effect of post-traumatic-stress-disorder on intra-operative analgesia in a veteran population during cataract procedures carried out using retrobulbar or topical anesthesia: a retrospective study. BMC Ophthalmol. 2017;17(1):85.
29. Payal AR, Gonzalez-Gonzalez LA, Chen X, et al. Outcomes of cataract surgery with residents as primary surgeons in the Veterans Affairs Healthcare System. J Cataract Refract Surg. 2016;42(3):370-384.
30. US Department of Veterans Affairs. Mission of the office of academic affiliations. https://www.va.gov/oaa/oaa_mission.asp. Updated November 30, 2018. Accessed March 18, 2019.
31. American Academy of Ophthalmology. Choosing wisely: five things ophthalmologists and patients should question. https://www.aao.org/choosing-wisely. Published February 2013. Accessed March 18, 2019.
32. Martin SK, Cifu AS. Routine preoperative laboratory tests for elective surgery. JAMA. 2017;318(6):567-568.
33. Schein OD, Katz J, Bass EB, et al; Study of Medical Testing for Cataract Surgery. The value of routine preoperative medical testing before cataract surgery. N Engl J Med. 2000;342(3):168-175.
34. Margo CE. Asymptomatic bacteriuria and acute-onset endophthalmitis after cataract surgery. Can J Ophthalmol. 2015;50(4):e51-52.
35. Fukui K, Fujioka M, Yamasaki K, Yamakawa S, Matsuo H, Noguchi M. Risk factors for postoperative complications among the elderly after plastic surgery procedures performed under general anesthesia. Plast Surg Int. 2018:7053839.
What are the risks of long-term PPI use for GERD symptoms in patients > 65 years?
EVIDENCE SUMMARY
A 2017 meta-analysis of 16 RCTs examined the risk of cardiovascular events in 7540 adult patients taking PPIs for GERD (mean ages 45-55 years).1 The primary outcome was cardiovascular events—including acute myocardial infarction, myocardial ischemia, angina pectoris, cardiac failure, and coronary artery stenosis—and cardiac disorders.
Analysis of pooled data found that PPI use was associated with a 70% increase in cardiovascular risk (relative risk [RR] = 1.7; 95% confidence interval [CI], 1.13-2.56; number needed to harm [NNH] = 241) when compared with controls (placebo, H2 blocker, or surgery). A subgroup analysis found that PPI use for longer than 8 weeks was associated with an even higher risk of adverse cardiovascular events (6 trials, 2296 patients; RR = 2.33; 95% CI, 1.33-4.08; NNH = 67) when compared with controls. The meta-analysis wasn’t limited by heterogeneity (I2 = 0).
C difficile infection risk is higherfor PPI users
A 2016 meta-analysis of 23 observational studies (19 case-control, 4 retrospective cohort; 186,033 patients) examined the risk of hospital-acquired C difficile infections in adults prescribed PPI for any indication.2 PPI exposure varied from use at time of diagnosis or hospitalization to any use within 90 days. Of the 23 studies, 16 reported sufficient data to calculate the mean age for the patients which was 69.9 years.
The risk of C difficile infection was found to be higher with PPI use than no use (pooled odds ratio [OR] = 1.81; 95% CI, 1.52-2.14). Although a significant association was found across a large group, the results were limited by considerable heterogeneity (I2 = 82%).
Risk of community-acquired pneumonia also increases with PPI use
A 2015 systematic review and meta-analysis of 33 trials (18 case-control, 10 cohort, 4 RCTs, and 1 case-crossover study) examined the risk of CAP in adult patients prescribed PPI for any indication for durations ranging from less than 1 month to > 6 months.3 The systematic review was distilled to 26 studies because of overlapping study populations. These 26 studies included 226,769 cases of CAP among 6,351,656 patients. The primary outcome was development of CAP, the secondary outcome was hospitalization for CAP.
PPI use, compared with no use, was associated with an increased risk of developing CAP (pooled OR = 1.49; 95% CI, 1.16-1.92) and an increased risk of hospitalization for CAP (pooled OR = 1.61; 95% CI, 1.12-2.31).
In a subgroup analysis for age, patients older than 65 years were also found to have an increased risk of developing CAP with PPI use (11 trials, total number of patients not provided; OR = 1.33; 95% CI, 1.13-1.58). Despite the significant associations of PPI use with risk revealed in the primary, secondary, and subgroup analyses, the results were limited by marked heterogeneity, with an I2 > 99%.
Continue to: Hip and vertebral fracture risks associated with PPIs
Hip and vertebral fracture riskis associated with PPIs
A 2011 systematic review and meta-analysis investigated the risk of fracture in adult patients taking PPIs for any indication.4 The analysis included 10 observational studies (4 cohort, 6 case-control) with a total of 223,210 fracture cases. The authors examined the incidence of hip, vertebral, and wrist or forearm fractures.
No significant association was found between PPI use and wrist or forearm fracture (3 studies; pooled OR = 1.09; 95% CI, 0.95-1.24). A modest association was noted between PPI use and both hip fractures (9 trials; OR = 1.25; 95% CI, 1.14-1.37) and vertebral fractures (4 trials; OR = 1.5; 95% CI, 1.32-1.72).
Subgroup analysis didn’t reveal evidence of an effect of duration of PPI use on fracture. Investigators didn’t conduct subgroup analysis of different patient ages. Final results were limited by significant heterogeneity with an I2 of 86%.
RECOMMENDATIONS
A 2015 American Geriatrics Society Beers Criteria update recommends limiting PPI use because of increased risk of C difficile infections and fractures. It also recommends against using PPIs for longer than 8 weeks except for high-risk patients (such as patients taking oral corticosteroids or chronic nonsteroidal anti-inflammatory drug users), patients with Barrett’s esophagitis, or patients who need maintenance after failure of a drug discontinuation trial or H2 blockers (quality of evidence, high; SOR, strong).5
Editor’s take
1. Sun S, Cui Z, Zhou M, et al. Proton pump inhibitor monotherapy and the risk of cardiovascular events in patients with gastro-esophageal reflux disease: a meta-analysis. Neurogastroenterol Motil. 2017;29:e12926.
2. Arriola V, Tischendorf J, Musuuza J, et al. Assessing the risk of hospital-acquired clostridium difficile infection with proton pump inhibitor use: a meta-analysis. Infect Control Hosp Epidemiol. 2016;37:1408-1417.
3. Lambert AA, Lam JO, Paik JJ, et al. Risk of community-acquired pneumonia with outpatient proton-pump inhibitor therapy: a systematic review and meta-analysis. PLoS One. 2015;10:e0128004.
4. Ngamruengphong S, Leontiadis GI, Radhi S, et al. Proton pump inhibitors and risk of fracture: a systematic review and meta-analysis of observational studies. Am J Gastroenterol. 2011;106:1209-1218.
5. American Geriatrics Society 2015 Beers Criteria Update Expert Panel. American Geriatrics Society 2015 Updated Beers Criteria for Potentially Inappropriate Medication Use in Older Adults. J Am Geriatr Soc. 2015;63:2227-2246.
EVIDENCE SUMMARY
A 2017 meta-analysis of 16 RCTs examined the risk of cardiovascular events in 7540 adult patients taking PPIs for GERD (mean ages 45-55 years).1 The primary outcome was cardiovascular events—including acute myocardial infarction, myocardial ischemia, angina pectoris, cardiac failure, and coronary artery stenosis—and cardiac disorders.
Analysis of pooled data found that PPI use was associated with a 70% increase in cardiovascular risk (relative risk [RR] = 1.7; 95% confidence interval [CI], 1.13-2.56; number needed to harm [NNH] = 241) when compared with controls (placebo, H2 blocker, or surgery). A subgroup analysis found that PPI use for longer than 8 weeks was associated with an even higher risk of adverse cardiovascular events (6 trials, 2296 patients; RR = 2.33; 95% CI, 1.33-4.08; NNH = 67) when compared with controls. The meta-analysis wasn’t limited by heterogeneity (I2 = 0).
C difficile infection risk is higherfor PPI users
A 2016 meta-analysis of 23 observational studies (19 case-control, 4 retrospective cohort; 186,033 patients) examined the risk of hospital-acquired C difficile infections in adults prescribed PPI for any indication.2 PPI exposure varied from use at time of diagnosis or hospitalization to any use within 90 days. Of the 23 studies, 16 reported sufficient data to calculate the mean age for the patients which was 69.9 years.
The risk of C difficile infection was found to be higher with PPI use than no use (pooled odds ratio [OR] = 1.81; 95% CI, 1.52-2.14). Although a significant association was found across a large group, the results were limited by considerable heterogeneity (I2 = 82%).
Risk of community-acquired pneumonia also increases with PPI use
A 2015 systematic review and meta-analysis of 33 trials (18 case-control, 10 cohort, 4 RCTs, and 1 case-crossover study) examined the risk of CAP in adult patients prescribed PPI for any indication for durations ranging from less than 1 month to > 6 months.3 The systematic review was distilled to 26 studies because of overlapping study populations. These 26 studies included 226,769 cases of CAP among 6,351,656 patients. The primary outcome was development of CAP, the secondary outcome was hospitalization for CAP.
PPI use, compared with no use, was associated with an increased risk of developing CAP (pooled OR = 1.49; 95% CI, 1.16-1.92) and an increased risk of hospitalization for CAP (pooled OR = 1.61; 95% CI, 1.12-2.31).
In a subgroup analysis for age, patients older than 65 years were also found to have an increased risk of developing CAP with PPI use (11 trials, total number of patients not provided; OR = 1.33; 95% CI, 1.13-1.58). Despite the significant associations of PPI use with risk revealed in the primary, secondary, and subgroup analyses, the results were limited by marked heterogeneity, with an I2 > 99%.
Continue to: Hip and vertebral fracture risks associated with PPIs
Hip and vertebral fracture riskis associated with PPIs
A 2011 systematic review and meta-analysis investigated the risk of fracture in adult patients taking PPIs for any indication.4 The analysis included 10 observational studies (4 cohort, 6 case-control) with a total of 223,210 fracture cases. The authors examined the incidence of hip, vertebral, and wrist or forearm fractures.
No significant association was found between PPI use and wrist or forearm fracture (3 studies; pooled OR = 1.09; 95% CI, 0.95-1.24). A modest association was noted between PPI use and both hip fractures (9 trials; OR = 1.25; 95% CI, 1.14-1.37) and vertebral fractures (4 trials; OR = 1.5; 95% CI, 1.32-1.72).
Subgroup analysis didn’t reveal evidence of an effect of duration of PPI use on fracture. Investigators didn’t conduct subgroup analysis of different patient ages. Final results were limited by significant heterogeneity with an I2 of 86%.
RECOMMENDATIONS
A 2015 American Geriatrics Society Beers Criteria update recommends limiting PPI use because of increased risk of C difficile infections and fractures. It also recommends against using PPIs for longer than 8 weeks except for high-risk patients (such as patients taking oral corticosteroids or chronic nonsteroidal anti-inflammatory drug users), patients with Barrett’s esophagitis, or patients who need maintenance after failure of a drug discontinuation trial or H2 blockers (quality of evidence, high; SOR, strong).5
Editor’s take
EVIDENCE SUMMARY
A 2017 meta-analysis of 16 RCTs examined the risk of cardiovascular events in 7540 adult patients taking PPIs for GERD (mean ages 45-55 years).1 The primary outcome was cardiovascular events—including acute myocardial infarction, myocardial ischemia, angina pectoris, cardiac failure, and coronary artery stenosis—and cardiac disorders.
Analysis of pooled data found that PPI use was associated with a 70% increase in cardiovascular risk (relative risk [RR] = 1.7; 95% confidence interval [CI], 1.13-2.56; number needed to harm [NNH] = 241) when compared with controls (placebo, H2 blocker, or surgery). A subgroup analysis found that PPI use for longer than 8 weeks was associated with an even higher risk of adverse cardiovascular events (6 trials, 2296 patients; RR = 2.33; 95% CI, 1.33-4.08; NNH = 67) when compared with controls. The meta-analysis wasn’t limited by heterogeneity (I2 = 0).
C difficile infection risk is higherfor PPI users
A 2016 meta-analysis of 23 observational studies (19 case-control, 4 retrospective cohort; 186,033 patients) examined the risk of hospital-acquired C difficile infections in adults prescribed PPI for any indication.2 PPI exposure varied from use at time of diagnosis or hospitalization to any use within 90 days. Of the 23 studies, 16 reported sufficient data to calculate the mean age for the patients which was 69.9 years.
The risk of C difficile infection was found to be higher with PPI use than no use (pooled odds ratio [OR] = 1.81; 95% CI, 1.52-2.14). Although a significant association was found across a large group, the results were limited by considerable heterogeneity (I2 = 82%).
Risk of community-acquired pneumonia also increases with PPI use
A 2015 systematic review and meta-analysis of 33 trials (18 case-control, 10 cohort, 4 RCTs, and 1 case-crossover study) examined the risk of CAP in adult patients prescribed PPI for any indication for durations ranging from less than 1 month to > 6 months.3 The systematic review was distilled to 26 studies because of overlapping study populations. These 26 studies included 226,769 cases of CAP among 6,351,656 patients. The primary outcome was development of CAP, the secondary outcome was hospitalization for CAP.
PPI use, compared with no use, was associated with an increased risk of developing CAP (pooled OR = 1.49; 95% CI, 1.16-1.92) and an increased risk of hospitalization for CAP (pooled OR = 1.61; 95% CI, 1.12-2.31).
In a subgroup analysis for age, patients older than 65 years were also found to have an increased risk of developing CAP with PPI use (11 trials, total number of patients not provided; OR = 1.33; 95% CI, 1.13-1.58). Despite the significant associations of PPI use with risk revealed in the primary, secondary, and subgroup analyses, the results were limited by marked heterogeneity, with an I2 > 99%.
Continue to: Hip and vertebral fracture risks associated with PPIs
Hip and vertebral fracture riskis associated with PPIs
A 2011 systematic review and meta-analysis investigated the risk of fracture in adult patients taking PPIs for any indication.4 The analysis included 10 observational studies (4 cohort, 6 case-control) with a total of 223,210 fracture cases. The authors examined the incidence of hip, vertebral, and wrist or forearm fractures.
No significant association was found between PPI use and wrist or forearm fracture (3 studies; pooled OR = 1.09; 95% CI, 0.95-1.24). A modest association was noted between PPI use and both hip fractures (9 trials; OR = 1.25; 95% CI, 1.14-1.37) and vertebral fractures (4 trials; OR = 1.5; 95% CI, 1.32-1.72).
Subgroup analysis didn’t reveal evidence of an effect of duration of PPI use on fracture. Investigators didn’t conduct subgroup analysis of different patient ages. Final results were limited by significant heterogeneity with an I2 of 86%.
RECOMMENDATIONS
A 2015 American Geriatrics Society Beers Criteria update recommends limiting PPI use because of increased risk of C difficile infections and fractures. It also recommends against using PPIs for longer than 8 weeks except for high-risk patients (such as patients taking oral corticosteroids or chronic nonsteroidal anti-inflammatory drug users), patients with Barrett’s esophagitis, or patients who need maintenance after failure of a drug discontinuation trial or H2 blockers (quality of evidence, high; SOR, strong).5
Editor’s take
1. Sun S, Cui Z, Zhou M, et al. Proton pump inhibitor monotherapy and the risk of cardiovascular events in patients with gastro-esophageal reflux disease: a meta-analysis. Neurogastroenterol Motil. 2017;29:e12926.
2. Arriola V, Tischendorf J, Musuuza J, et al. Assessing the risk of hospital-acquired clostridium difficile infection with proton pump inhibitor use: a meta-analysis. Infect Control Hosp Epidemiol. 2016;37:1408-1417.
3. Lambert AA, Lam JO, Paik JJ, et al. Risk of community-acquired pneumonia with outpatient proton-pump inhibitor therapy: a systematic review and meta-analysis. PLoS One. 2015;10:e0128004.
4. Ngamruengphong S, Leontiadis GI, Radhi S, et al. Proton pump inhibitors and risk of fracture: a systematic review and meta-analysis of observational studies. Am J Gastroenterol. 2011;106:1209-1218.
5. American Geriatrics Society 2015 Beers Criteria Update Expert Panel. American Geriatrics Society 2015 Updated Beers Criteria for Potentially Inappropriate Medication Use in Older Adults. J Am Geriatr Soc. 2015;63:2227-2246.
1. Sun S, Cui Z, Zhou M, et al. Proton pump inhibitor monotherapy and the risk of cardiovascular events in patients with gastro-esophageal reflux disease: a meta-analysis. Neurogastroenterol Motil. 2017;29:e12926.
2. Arriola V, Tischendorf J, Musuuza J, et al. Assessing the risk of hospital-acquired clostridium difficile infection with proton pump inhibitor use: a meta-analysis. Infect Control Hosp Epidemiol. 2016;37:1408-1417.
3. Lambert AA, Lam JO, Paik JJ, et al. Risk of community-acquired pneumonia with outpatient proton-pump inhibitor therapy: a systematic review and meta-analysis. PLoS One. 2015;10:e0128004.
4. Ngamruengphong S, Leontiadis GI, Radhi S, et al. Proton pump inhibitors and risk of fracture: a systematic review and meta-analysis of observational studies. Am J Gastroenterol. 2011;106:1209-1218.
5. American Geriatrics Society 2015 Beers Criteria Update Expert Panel. American Geriatrics Society 2015 Updated Beers Criteria for Potentially Inappropriate Medication Use in Older Adults. J Am Geriatr Soc. 2015;63:2227-2246.
EVIDENCE-BASED ANSWER:
The use of proton pump inhibitors (PPIs) to control gastroesophageal reflux disease (GERD) is significantly associated with an increased risk of cardiovascular events such as acute myocardial infarction and myocardial ischemia, especially with treatment longer than 8 weeks (strength of recommendation [SOR]: A, systematic review of randomized, controlled trials [RCTs]). This summary is based on data extrapolated from studies on all adults because there is limited evidence that specifically addresses patients older than 65 years.
Adults taking PPIs also appear to be at increased risk of Clostridium difficile infection, community-acquired pneumonia (CAP; with use for < 30 days), and fracture (SOR: B, systematic reviews of heterogeneous prospective and retrospective observational studies).