User login
ALA report: Federal and state actions to limit tobacco use fall short
Tobacco use is currently at an all-time low thanks to public and private efforts, but more aggressive action from federal, state, and local governments is needed to protect the public, according to a review of tobacco control trends in the United States.
The American Lung Association (ALA) released “State of Tobacco Control” 2019, its 17th annual state-by-state analysis and list of recommended policy priorities to limit tobacco use. Although the report notes some positive steps taken by the federal and state governments, shortfalls in policy and legislation also are highlighted. The report states, “We know how and are ready to save more lives, but we need our elected officials to do much more. To many, solving America’s tobacco crisis might seem like a complex puzzle with no solution. And yet we have known for years what pieces are needed to reduce the disease and death caused by tobacco use.”
In this report, the federal government and each state are graded on a scale, A through F, for policy actions and laws to limit tobacco use. The grading methodology is based on a detailed point system cataloging the implementation and strength of specific actions and policies to limit tobacco use.
Areas of Impact
The report focused on six areas of public policy that affect exposure to and use of tobacco:
- Smoke-free air: Protecting the public from secondhand smoke should be a priority for policymakers, according the report, but 22 states have no smoke-free workplace laws in place. Laws restricting e-cigarettes in workplaces and public buildings have lagged behind tobacco laws in many states.
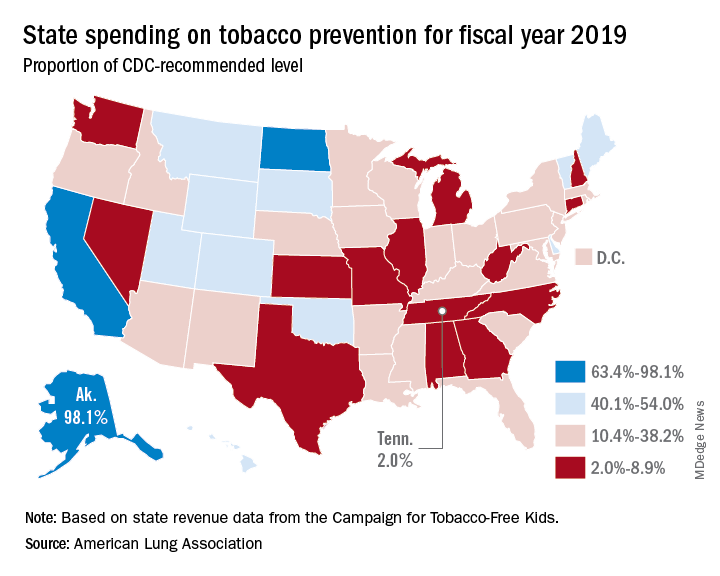
- Tobacco prevention funding: Dedicated funds to prevent tobacco addiction before it starts is a key element of a public health attack on tobacco use, but no U.S. state currently spends what the Centers for Disease Control and Prevention has recommended. Twenty years ago, the Master Settlement Agreement between the tobacco industry and 46 states and the District of Columbia guaranteed ongoing payments to the states to be used for tobacco prevention and control. Although those funds have been collected in the states to the tune of $27 billion since 1998, overall only 2.4% of those funds have been spent for this purpose, and the rest has been budgeted for other purposes.
- Tobacco taxes: Sales taxes on tobacco products have been highly effective in preventing young people from taking up tobacco use, but those taxation rates have remained unchanged in 2018 in all but the District of Columbia and Oklahoma. The tobacco industry spent $22 million in a successful effort to defeat ballot measures to increase sales taxes on tobacco in Montana and South Dakota.
- Tobacco 21: “Increasing the legal age of sale for tobacco products to 21 would decrease tobacco use by 12% and could prevent 223,000 deaths among those born between 2000 and 2019,” the report noted, citing a 2015 report by the Institute of Medicine. So far, the this restriction has been legislated in six states, the District of Columbia, and numerous local governments. The ALA considers increasing the age for tobacco sales to 21 to be a public health priority.
- Helping smokers quit: The addictive qualities of tobacco mean that many smokers struggle unsuccessfully to quit, and medical intervention is needed to help them. The report notes that current law requires that Medicaid expansion health plans and private insurance plans cover comprehensive smoking cessation treatment. However, not all states have the expanded Medicaid program, and many of those with Medicaid expansion don’t offer coverage of all Food and Drug–approved cessation treatments. Despite laws requiring smoking cessation coverage, many private insurance plans still do not include this coverage. The ALA recommends enforcement of the current law with regard to tobacco cessation insurance coverage.
- FDA regulation of tobacco products: The FDA has announced plans to make a major effort to reduce tobacco use in young people, decrease nicotine in cigarettes, and to restrict flavored tobacco products. But these plans fall short of the aggressive action needed to curb the tobacco “epidemic,” according to the report. Delayed action and timid policy have “resulted in tobacco companies becoming more emboldened to devise new and egregious ways to addict youth and sustain addiction among current users.” The ALA report points to the steep rise in e-cigarette use among youth with a 20.8% rise in high school students using these products in 2018, a rise from 11.7% in 2017. This trend is not likely to be reversed by the FDA proposals to date, which rely on voluntary action by the industry to curb youth use, sales restrictions to youth, and restrictions on some flavored tobacco products.
The report card
Federal government efforts in regulation of tobacco products, taxation, and health insurance coverage of cessation all received an F in this report, while mass media campaigns were given an A.
The states didn’t fare much better. They were graded on prevention and control funding, smoke-free air, taxation, access to cessation services, and minimum age for sales. A total of 19 states received a grade of F in four or five of these areas.
Funding for prevention and control was evaluated as the percentage of the amount recommended by the CDC, adjusted for a variety of state-specific factors such as prevalence of tobacco use, cost and complexity of conducting mass media campaigns, and proportion of the audience below 200% of the federal poverty level. A limitation of this methodology of grading funding is that it doesn’t evaluate effectiveness of the spending or the level of spending in different program categories. The higher spenders on prevention and control were Alaska at 98.1% and California at 74.5% of the CDC recommended level. The lowest spenders were Georgia at 2.8% and Missouri at 3.0%.
All but eight states received an F on minimum age for tobacco sales because most have an age limit 18 instead of the ALA and CDC recommendation of age 21.
Harold Wimmer, the CEO of the American Lung Association, wrote, “Aggressive action by our country’s federal and state policymakers is urgently required. However, ‘State of Tobacco Control’ 2019 has found a disturbing failure by federal and state governments to take action to put in place meaningful and proven-effective policies that would have prevented, and reduced tobacco use during 2018. This failure to act places the lung health and lives of Americans at risk. We have also found that this lack of action has emboldened tobacco companies to be even more brazen in producing and marketing products squarely aimed at kids, such as the JUUL e-cigarettes that look like an easily concealed USB drive, which now dominate the market driven by youth use.”
The full report is available for download at the ALA website.
SOURCE: American Lung Association, “State of Tobacco Control 2019”.
Tobacco use is currently at an all-time low thanks to public and private efforts, but more aggressive action from federal, state, and local governments is needed to protect the public, according to a review of tobacco control trends in the United States.

The American Lung Association (ALA) released “State of Tobacco Control” 2019, its 17th annual state-by-state analysis and list of recommended policy priorities to limit tobacco use. Although the report notes some positive steps taken by the federal and state governments, shortfalls in policy and legislation also are highlighted. The report states, “We know how and are ready to save more lives, but we need our elected officials to do much more. To many, solving America’s tobacco crisis might seem like a complex puzzle with no solution. And yet we have known for years what pieces are needed to reduce the disease and death caused by tobacco use.”
In this report, the federal government and each state are graded on a scale, A through F, for policy actions and laws to limit tobacco use. The grading methodology is based on a detailed point system cataloging the implementation and strength of specific actions and policies to limit tobacco use.
Areas of Impact
The report focused on six areas of public policy that affect exposure to and use of tobacco:
- Smoke-free air: Protecting the public from secondhand smoke should be a priority for policymakers, according the report, but 22 states have no smoke-free workplace laws in place. Laws restricting e-cigarettes in workplaces and public buildings have lagged behind tobacco laws in many states.
- Tobacco prevention funding: Dedicated funds to prevent tobacco addiction before it starts is a key element of a public health attack on tobacco use, but no U.S. state currently spends what the Centers for Disease Control and Prevention has recommended. Twenty years ago, the Master Settlement Agreement between the tobacco industry and 46 states and the District of Columbia guaranteed ongoing payments to the states to be used for tobacco prevention and control. Although those funds have been collected in the states to the tune of $27 billion since 1998, overall only 2.4% of those funds have been spent for this purpose, and the rest has been budgeted for other purposes.
- Tobacco taxes: Sales taxes on tobacco products have been highly effective in preventing young people from taking up tobacco use, but those taxation rates have remained unchanged in 2018 in all but the District of Columbia and Oklahoma. The tobacco industry spent $22 million in a successful effort to defeat ballot measures to increase sales taxes on tobacco in Montana and South Dakota.
- Tobacco 21: “Increasing the legal age of sale for tobacco products to 21 would decrease tobacco use by 12% and could prevent 223,000 deaths among those born between 2000 and 2019,” the report noted, citing a 2015 report by the Institute of Medicine. So far, the this restriction has been legislated in six states, the District of Columbia, and numerous local governments. The ALA considers increasing the age for tobacco sales to 21 to be a public health priority.
- Helping smokers quit: The addictive qualities of tobacco mean that many smokers struggle unsuccessfully to quit, and medical intervention is needed to help them. The report notes that current law requires that Medicaid expansion health plans and private insurance plans cover comprehensive smoking cessation treatment. However, not all states have the expanded Medicaid program, and many of those with Medicaid expansion don’t offer coverage of all Food and Drug–approved cessation treatments. Despite laws requiring smoking cessation coverage, many private insurance plans still do not include this coverage. The ALA recommends enforcement of the current law with regard to tobacco cessation insurance coverage.
- FDA regulation of tobacco products: The FDA has announced plans to make a major effort to reduce tobacco use in young people, decrease nicotine in cigarettes, and to restrict flavored tobacco products. But these plans fall short of the aggressive action needed to curb the tobacco “epidemic,” according to the report. Delayed action and timid policy have “resulted in tobacco companies becoming more emboldened to devise new and egregious ways to addict youth and sustain addiction among current users.” The ALA report points to the steep rise in e-cigarette use among youth with a 20.8% rise in high school students using these products in 2018, a rise from 11.7% in 2017. This trend is not likely to be reversed by the FDA proposals to date, which rely on voluntary action by the industry to curb youth use, sales restrictions to youth, and restrictions on some flavored tobacco products.
The report card
Federal government efforts in regulation of tobacco products, taxation, and health insurance coverage of cessation all received an F in this report, while mass media campaigns were given an A.
The states didn’t fare much better. They were graded on prevention and control funding, smoke-free air, taxation, access to cessation services, and minimum age for sales. A total of 19 states received a grade of F in four or five of these areas.
Funding for prevention and control was evaluated as the percentage of the amount recommended by the CDC, adjusted for a variety of state-specific factors such as prevalence of tobacco use, cost and complexity of conducting mass media campaigns, and proportion of the audience below 200% of the federal poverty level. A limitation of this methodology of grading funding is that it doesn’t evaluate effectiveness of the spending or the level of spending in different program categories. The higher spenders on prevention and control were Alaska at 98.1% and California at 74.5% of the CDC recommended level. The lowest spenders were Georgia at 2.8% and Missouri at 3.0%.
All but eight states received an F on minimum age for tobacco sales because most have an age limit 18 instead of the ALA and CDC recommendation of age 21.
Harold Wimmer, the CEO of the American Lung Association, wrote, “Aggressive action by our country’s federal and state policymakers is urgently required. However, ‘State of Tobacco Control’ 2019 has found a disturbing failure by federal and state governments to take action to put in place meaningful and proven-effective policies that would have prevented, and reduced tobacco use during 2018. This failure to act places the lung health and lives of Americans at risk. We have also found that this lack of action has emboldened tobacco companies to be even more brazen in producing and marketing products squarely aimed at kids, such as the JUUL e-cigarettes that look like an easily concealed USB drive, which now dominate the market driven by youth use.”
The full report is available for download at the ALA website.
SOURCE: American Lung Association, “State of Tobacco Control 2019”.
Tobacco use is currently at an all-time low thanks to public and private efforts, but more aggressive action from federal, state, and local governments is needed to protect the public, according to a review of tobacco control trends in the United States.

The American Lung Association (ALA) released “State of Tobacco Control” 2019, its 17th annual state-by-state analysis and list of recommended policy priorities to limit tobacco use. Although the report notes some positive steps taken by the federal and state governments, shortfalls in policy and legislation also are highlighted. The report states, “We know how and are ready to save more lives, but we need our elected officials to do much more. To many, solving America’s tobacco crisis might seem like a complex puzzle with no solution. And yet we have known for years what pieces are needed to reduce the disease and death caused by tobacco use.”
In this report, the federal government and each state are graded on a scale, A through F, for policy actions and laws to limit tobacco use. The grading methodology is based on a detailed point system cataloging the implementation and strength of specific actions and policies to limit tobacco use.
Areas of Impact
The report focused on six areas of public policy that affect exposure to and use of tobacco:
- Smoke-free air: Protecting the public from secondhand smoke should be a priority for policymakers, according the report, but 22 states have no smoke-free workplace laws in place. Laws restricting e-cigarettes in workplaces and public buildings have lagged behind tobacco laws in many states.
- Tobacco prevention funding: Dedicated funds to prevent tobacco addiction before it starts is a key element of a public health attack on tobacco use, but no U.S. state currently spends what the Centers for Disease Control and Prevention has recommended. Twenty years ago, the Master Settlement Agreement between the tobacco industry and 46 states and the District of Columbia guaranteed ongoing payments to the states to be used for tobacco prevention and control. Although those funds have been collected in the states to the tune of $27 billion since 1998, overall only 2.4% of those funds have been spent for this purpose, and the rest has been budgeted for other purposes.
- Tobacco taxes: Sales taxes on tobacco products have been highly effective in preventing young people from taking up tobacco use, but those taxation rates have remained unchanged in 2018 in all but the District of Columbia and Oklahoma. The tobacco industry spent $22 million in a successful effort to defeat ballot measures to increase sales taxes on tobacco in Montana and South Dakota.
- Tobacco 21: “Increasing the legal age of sale for tobacco products to 21 would decrease tobacco use by 12% and could prevent 223,000 deaths among those born between 2000 and 2019,” the report noted, citing a 2015 report by the Institute of Medicine. So far, the this restriction has been legislated in six states, the District of Columbia, and numerous local governments. The ALA considers increasing the age for tobacco sales to 21 to be a public health priority.
- Helping smokers quit: The addictive qualities of tobacco mean that many smokers struggle unsuccessfully to quit, and medical intervention is needed to help them. The report notes that current law requires that Medicaid expansion health plans and private insurance plans cover comprehensive smoking cessation treatment. However, not all states have the expanded Medicaid program, and many of those with Medicaid expansion don’t offer coverage of all Food and Drug–approved cessation treatments. Despite laws requiring smoking cessation coverage, many private insurance plans still do not include this coverage. The ALA recommends enforcement of the current law with regard to tobacco cessation insurance coverage.
- FDA regulation of tobacco products: The FDA has announced plans to make a major effort to reduce tobacco use in young people, decrease nicotine in cigarettes, and to restrict flavored tobacco products. But these plans fall short of the aggressive action needed to curb the tobacco “epidemic,” according to the report. Delayed action and timid policy have “resulted in tobacco companies becoming more emboldened to devise new and egregious ways to addict youth and sustain addiction among current users.” The ALA report points to the steep rise in e-cigarette use among youth with a 20.8% rise in high school students using these products in 2018, a rise from 11.7% in 2017. This trend is not likely to be reversed by the FDA proposals to date, which rely on voluntary action by the industry to curb youth use, sales restrictions to youth, and restrictions on some flavored tobacco products.
The report card
Federal government efforts in regulation of tobacco products, taxation, and health insurance coverage of cessation all received an F in this report, while mass media campaigns were given an A.
The states didn’t fare much better. They were graded on prevention and control funding, smoke-free air, taxation, access to cessation services, and minimum age for sales. A total of 19 states received a grade of F in four or five of these areas.
Funding for prevention and control was evaluated as the percentage of the amount recommended by the CDC, adjusted for a variety of state-specific factors such as prevalence of tobacco use, cost and complexity of conducting mass media campaigns, and proportion of the audience below 200% of the federal poverty level. A limitation of this methodology of grading funding is that it doesn’t evaluate effectiveness of the spending or the level of spending in different program categories. The higher spenders on prevention and control were Alaska at 98.1% and California at 74.5% of the CDC recommended level. The lowest spenders were Georgia at 2.8% and Missouri at 3.0%.
All but eight states received an F on minimum age for tobacco sales because most have an age limit 18 instead of the ALA and CDC recommendation of age 21.
Harold Wimmer, the CEO of the American Lung Association, wrote, “Aggressive action by our country’s federal and state policymakers is urgently required. However, ‘State of Tobacco Control’ 2019 has found a disturbing failure by federal and state governments to take action to put in place meaningful and proven-effective policies that would have prevented, and reduced tobacco use during 2018. This failure to act places the lung health and lives of Americans at risk. We have also found that this lack of action has emboldened tobacco companies to be even more brazen in producing and marketing products squarely aimed at kids, such as the JUUL e-cigarettes that look like an easily concealed USB drive, which now dominate the market driven by youth use.”
The full report is available for download at the ALA website.
SOURCE: American Lung Association, “State of Tobacco Control 2019”.
Loan Repayment Plan for Substance Use Clinicians
The opioid emergency claims > 130 lives every day, says Health Resources and Services Administration (HRSA) Administrator George Sigounas, MS, PhD. By strengthening the health workforce, HRSA hopes to ensure that there are enough clinicians to cope with the growing epidemic.
That is why, in December 2018, HRSA launched a program that Sigounas says is “critical to HHS’ response to the opioid crisis.” The new National Health Service Corps (NHSC) Substance Use Disorder (SUD) Workforce Loan Repayment Program (LRP) will provide eligible health care clinicians with student loan repayment assistance in exchange for service in underserved communities.
A clinician may be awarded up to $75,000 for 3 years of full-time service at an NHSC-approved SUD site and $37,500 for part-time. Eligible providers use evidence-based treatment models to treat SUDs and must be trained and licensed to provide SUD treatment at NHSC-approved facilities. Qualification criteria are available at https://nhsc.hrsa.gov/loan-repayment/nhsc-sud-workforce-loan-repayment-program.html.
Clinicians also can apply to the NHSC Loan Repayment Program for primary care, dental, and behavioral health professionals. If accepted, they may receive up to $50,000 for 2 years of full-time service, $25,000 for part-time.
Military reservists also are eligible to participate in either the NHSC LRP or the NHSC Students to Service Loan Repayment Program. (Military training or service will not satisfy the NHSC service commitment.) More information is available at https://nhsc.hrsa.gov/loan-repayment/military-reservists.html.
Clinicians can only apply for 1 program. Sigounas says, “I am grateful to the clinicians who will apply and are looking to make a positive impact on patients, caregivers, and hard-hit communities throughout the country.”
The opioid emergency claims > 130 lives every day, says Health Resources and Services Administration (HRSA) Administrator George Sigounas, MS, PhD. By strengthening the health workforce, HRSA hopes to ensure that there are enough clinicians to cope with the growing epidemic.
That is why, in December 2018, HRSA launched a program that Sigounas says is “critical to HHS’ response to the opioid crisis.” The new National Health Service Corps (NHSC) Substance Use Disorder (SUD) Workforce Loan Repayment Program (LRP) will provide eligible health care clinicians with student loan repayment assistance in exchange for service in underserved communities.
A clinician may be awarded up to $75,000 for 3 years of full-time service at an NHSC-approved SUD site and $37,500 for part-time. Eligible providers use evidence-based treatment models to treat SUDs and must be trained and licensed to provide SUD treatment at NHSC-approved facilities. Qualification criteria are available at https://nhsc.hrsa.gov/loan-repayment/nhsc-sud-workforce-loan-repayment-program.html.
Clinicians also can apply to the NHSC Loan Repayment Program for primary care, dental, and behavioral health professionals. If accepted, they may receive up to $50,000 for 2 years of full-time service, $25,000 for part-time.
Military reservists also are eligible to participate in either the NHSC LRP or the NHSC Students to Service Loan Repayment Program. (Military training or service will not satisfy the NHSC service commitment.) More information is available at https://nhsc.hrsa.gov/loan-repayment/military-reservists.html.
Clinicians can only apply for 1 program. Sigounas says, “I am grateful to the clinicians who will apply and are looking to make a positive impact on patients, caregivers, and hard-hit communities throughout the country.”
The opioid emergency claims > 130 lives every day, says Health Resources and Services Administration (HRSA) Administrator George Sigounas, MS, PhD. By strengthening the health workforce, HRSA hopes to ensure that there are enough clinicians to cope with the growing epidemic.
That is why, in December 2018, HRSA launched a program that Sigounas says is “critical to HHS’ response to the opioid crisis.” The new National Health Service Corps (NHSC) Substance Use Disorder (SUD) Workforce Loan Repayment Program (LRP) will provide eligible health care clinicians with student loan repayment assistance in exchange for service in underserved communities.
A clinician may be awarded up to $75,000 for 3 years of full-time service at an NHSC-approved SUD site and $37,500 for part-time. Eligible providers use evidence-based treatment models to treat SUDs and must be trained and licensed to provide SUD treatment at NHSC-approved facilities. Qualification criteria are available at https://nhsc.hrsa.gov/loan-repayment/nhsc-sud-workforce-loan-repayment-program.html.
Clinicians also can apply to the NHSC Loan Repayment Program for primary care, dental, and behavioral health professionals. If accepted, they may receive up to $50,000 for 2 years of full-time service, $25,000 for part-time.
Military reservists also are eligible to participate in either the NHSC LRP or the NHSC Students to Service Loan Repayment Program. (Military training or service will not satisfy the NHSC service commitment.) More information is available at https://nhsc.hrsa.gov/loan-repayment/military-reservists.html.
Clinicians can only apply for 1 program. Sigounas says, “I am grateful to the clinicians who will apply and are looking to make a positive impact on patients, caregivers, and hard-hit communities throughout the country.”
Cloud of inconsistency hangs over cannabis data
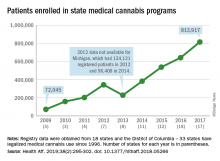
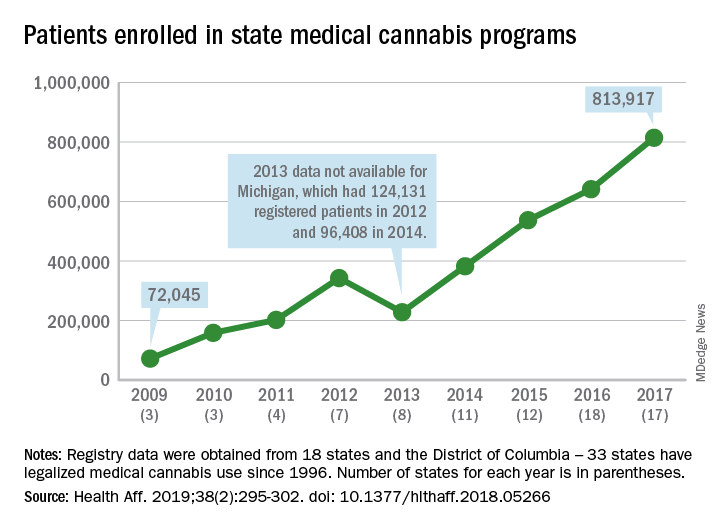
More people are using medical cannabis as it becomes legal in more states, but the lack of standardization in states’ data collection hindered investigators’ efforts to track that use.
Legalized medical cannabis is now available in 33 states and the District of Columbia, and the number of users has risen from just over 72,000 in 2009 to almost 814,000 in 2017. That 814,000, however, covers only 16 states and D.C., since 1 state (Connecticut) does not publish reports on medical cannabis use, 12 did not have statistics available, 2 (New York and Vermont) didn’t report data for 2017, and 2 (California and Maine) have voluntary registries that are unlikely to be accurate, according to Kevin F. Boehnke, PhD, of the University of Michigan, Ann Arbor, and his associates.
Michigan had the largest reported number of patients enrolled in its medical cannabis program in 2017, almost 270,000. California – the state with the oldest medical cannabis legislation (passed in 1996) and the largest overall population but a voluntary cannabis registry – reported its highest number of enrollees, 12,659, in 2009-2010, the investigators said. Colorado had more than 116,000 patients in its medical cannabis program in 2010 (Health Aff. 2019;38[2]:295-302).
The “many inconsistencies in data quality across states [suggest] the need for further standardization of data collection. Such standardization would add transparency to understanding how medical cannabis programs are used, which would help guide both research and policy needs,” Dr. Boehnke and his associates wrote.
More consistency was seen in the reasons for using medical cannabis. Chronic pain made up 62.2% of all qualifying conditions reported by patients during 1999-2016, with the annual average varying between 33.3% and 73%. Multiple sclerosis spasticity symptoms had the second-highest number of reports over the study period, followed by chemotherapy-induced nausea and vomiting, posttraumatic stress disorder, and cancer, they reported.
The investigators also looked at the appropriateness of cannabis and determined that its use in 85.5% of patient-reported conditions was “supported by conclusive or substantial evidence of therapeutic effectiveness, according to the 2017 National Academies report” on the health effects of cannabis.
“We believe not only that it is inappropriate for cannabis to remain a Schedule I substance, but also that state and federal policy makers should begin evaluating evidence-based ways for safely integrating cannabis research and products into the health care system,” they concluded.
SOURCE: Boehnke KF et al. Health Aff. 2019;38(2):295-302.
More people are using medical cannabis as it becomes legal in more states, but the lack of standardization in states’ data collection hindered investigators’ efforts to track that use.
Legalized medical cannabis is now available in 33 states and the District of Columbia, and the number of users has risen from just over 72,000 in 2009 to almost 814,000 in 2017. That 814,000, however, covers only 16 states and D.C., since 1 state (Connecticut) does not publish reports on medical cannabis use, 12 did not have statistics available, 2 (New York and Vermont) didn’t report data for 2017, and 2 (California and Maine) have voluntary registries that are unlikely to be accurate, according to Kevin F. Boehnke, PhD, of the University of Michigan, Ann Arbor, and his associates.
Michigan had the largest reported number of patients enrolled in its medical cannabis program in 2017, almost 270,000. California – the state with the oldest medical cannabis legislation (passed in 1996) and the largest overall population but a voluntary cannabis registry – reported its highest number of enrollees, 12,659, in 2009-2010, the investigators said. Colorado had more than 116,000 patients in its medical cannabis program in 2010 (Health Aff. 2019;38[2]:295-302).
The “many inconsistencies in data quality across states [suggest] the need for further standardization of data collection. Such standardization would add transparency to understanding how medical cannabis programs are used, which would help guide both research and policy needs,” Dr. Boehnke and his associates wrote.
More consistency was seen in the reasons for using medical cannabis. Chronic pain made up 62.2% of all qualifying conditions reported by patients during 1999-2016, with the annual average varying between 33.3% and 73%. Multiple sclerosis spasticity symptoms had the second-highest number of reports over the study period, followed by chemotherapy-induced nausea and vomiting, posttraumatic stress disorder, and cancer, they reported.
The investigators also looked at the appropriateness of cannabis and determined that its use in 85.5% of patient-reported conditions was “supported by conclusive or substantial evidence of therapeutic effectiveness, according to the 2017 National Academies report” on the health effects of cannabis.
“We believe not only that it is inappropriate for cannabis to remain a Schedule I substance, but also that state and federal policy makers should begin evaluating evidence-based ways for safely integrating cannabis research and products into the health care system,” they concluded.
SOURCE: Boehnke KF et al. Health Aff. 2019;38(2):295-302.
More people are using medical cannabis as it becomes legal in more states, but the lack of standardization in states’ data collection hindered investigators’ efforts to track that use.
Legalized medical cannabis is now available in 33 states and the District of Columbia, and the number of users has risen from just over 72,000 in 2009 to almost 814,000 in 2017. That 814,000, however, covers only 16 states and D.C., since 1 state (Connecticut) does not publish reports on medical cannabis use, 12 did not have statistics available, 2 (New York and Vermont) didn’t report data for 2017, and 2 (California and Maine) have voluntary registries that are unlikely to be accurate, according to Kevin F. Boehnke, PhD, of the University of Michigan, Ann Arbor, and his associates.
Michigan had the largest reported number of patients enrolled in its medical cannabis program in 2017, almost 270,000. California – the state with the oldest medical cannabis legislation (passed in 1996) and the largest overall population but a voluntary cannabis registry – reported its highest number of enrollees, 12,659, in 2009-2010, the investigators said. Colorado had more than 116,000 patients in its medical cannabis program in 2010 (Health Aff. 2019;38[2]:295-302).
The “many inconsistencies in data quality across states [suggest] the need for further standardization of data collection. Such standardization would add transparency to understanding how medical cannabis programs are used, which would help guide both research and policy needs,” Dr. Boehnke and his associates wrote.
More consistency was seen in the reasons for using medical cannabis. Chronic pain made up 62.2% of all qualifying conditions reported by patients during 1999-2016, with the annual average varying between 33.3% and 73%. Multiple sclerosis spasticity symptoms had the second-highest number of reports over the study period, followed by chemotherapy-induced nausea and vomiting, posttraumatic stress disorder, and cancer, they reported.
The investigators also looked at the appropriateness of cannabis and determined that its use in 85.5% of patient-reported conditions was “supported by conclusive or substantial evidence of therapeutic effectiveness, according to the 2017 National Academies report” on the health effects of cannabis.
“We believe not only that it is inappropriate for cannabis to remain a Schedule I substance, but also that state and federal policy makers should begin evaluating evidence-based ways for safely integrating cannabis research and products into the health care system,” they concluded.
SOURCE: Boehnke KF et al. Health Aff. 2019;38(2):295-302.
FROM HEALTH AFFAIRS
President Trump calls for end to HIV/AIDS, pediatric cancer
HIV/AIDS, pediatric cancer research, abortion, prescription drug prices, and preexisting conditions were among the health care highlights of President Donald Trump’s second State of the Union address Feb. 5.

Mr. Trump promised to push for funds to end HIV/AIDS and childhood cancer within in 10 years. “In recent years, we have made remarkable progress in the fight against HIV and AIDS. Scientific breakthroughs have brought a once-distant dream within reach,” he said to assembled members of Congress and leaders of the executive and judicial branches of government. “My budget will ask Democrats and Republicans to make the needed commitment to eliminate the HIV epidemic in the United States within 10 years.”
Following the speech, Alex Azar, secretary of the Department of Health and Human Services, offered more details in a blog post on the agency’s website.
Funding for the initiative, dubbed “Ending the HIV Epidemic: A Plan for America,” will have three components.
The first involves increasing investments in “geographic hotspots” though existing programs like the Ryan White HIV/AIDS Program and a new community health center–based program to provide antiretroviral therapy (ART) and preexposure prophylaxis (PrEP) to those at the highest risk of contracting the disease.
Second is the use of data to track where the disease is spreading most rapidly to help target prevention, care, and treatment at the local level. The third will provide funds for the creation of a local HIV HealthForce in these targeted areas to expand HIV prevention and treatment efforts.
A fact sheet on this initiative called for a 75% reduction in new cases of HIV infection in 5 years and at least a 90% reduction within 10 years.
President Trump called for similar efforts to address pediatric cancer.
“Tonight I am also asking you to join me in another fight that all American can get behind – the fight against childhood cancer,” he said, adding that his budget request will come with a line item of $500 million over 10 years to fund research. “Many childhood cancers have not seen new therapies in decades.”
President Trump also asked Congress to legislate a prohibition of late-term abortion.
“There could be no greater contrast to the beautiful image of a mother holding her infant child than the chilling displays our nation saw in recent days,” he said. “Lawmakers in New York cheered with delight upon the passage of legislation that would allow a baby to be ripped from the mother’s womb moments from birth. These are living, feeling beautiful babies who will never get the chance to share their love and their dreams with the world. ... Let us work together to build a culture that cherishes innocent life.”
He also touched on the recurring themes regarding lowering the cost of health care and prescription drugs, as well as protecting those with preexisting conditions, something he called a major priority.
“It’s unacceptable that Americans pay vastly more than people in other countries for the exact same drugs, often made in the exact same place. This is wrong. This is unfair and together we will stop it, and we will stop it fast,” he said.
He did not offer any specific policy recommendation on how to address prescription drug costs, other than a comment on the need for greater price transparency.
“I am asking Congress to pass legislation that finally takes on the problem of global freeloading and delivers fairness and price transparency for American patients,” he said.
“We should also require drug companies, insurance companies, and hospitals to disclose real prices to foster competition and bring costs way down.”
SOURCE: Trump D. State of the Union Address, Feb. 5, 2019.
HIV/AIDS, pediatric cancer research, abortion, prescription drug prices, and preexisting conditions were among the health care highlights of President Donald Trump’s second State of the Union address Feb. 5.

Mr. Trump promised to push for funds to end HIV/AIDS and childhood cancer within in 10 years. “In recent years, we have made remarkable progress in the fight against HIV and AIDS. Scientific breakthroughs have brought a once-distant dream within reach,” he said to assembled members of Congress and leaders of the executive and judicial branches of government. “My budget will ask Democrats and Republicans to make the needed commitment to eliminate the HIV epidemic in the United States within 10 years.”
Following the speech, Alex Azar, secretary of the Department of Health and Human Services, offered more details in a blog post on the agency’s website.
Funding for the initiative, dubbed “Ending the HIV Epidemic: A Plan for America,” will have three components.
The first involves increasing investments in “geographic hotspots” though existing programs like the Ryan White HIV/AIDS Program and a new community health center–based program to provide antiretroviral therapy (ART) and preexposure prophylaxis (PrEP) to those at the highest risk of contracting the disease.
Second is the use of data to track where the disease is spreading most rapidly to help target prevention, care, and treatment at the local level. The third will provide funds for the creation of a local HIV HealthForce in these targeted areas to expand HIV prevention and treatment efforts.
A fact sheet on this initiative called for a 75% reduction in new cases of HIV infection in 5 years and at least a 90% reduction within 10 years.
President Trump called for similar efforts to address pediatric cancer.
“Tonight I am also asking you to join me in another fight that all American can get behind – the fight against childhood cancer,” he said, adding that his budget request will come with a line item of $500 million over 10 years to fund research. “Many childhood cancers have not seen new therapies in decades.”
President Trump also asked Congress to legislate a prohibition of late-term abortion.
“There could be no greater contrast to the beautiful image of a mother holding her infant child than the chilling displays our nation saw in recent days,” he said. “Lawmakers in New York cheered with delight upon the passage of legislation that would allow a baby to be ripped from the mother’s womb moments from birth. These are living, feeling beautiful babies who will never get the chance to share their love and their dreams with the world. ... Let us work together to build a culture that cherishes innocent life.”
He also touched on the recurring themes regarding lowering the cost of health care and prescription drugs, as well as protecting those with preexisting conditions, something he called a major priority.
“It’s unacceptable that Americans pay vastly more than people in other countries for the exact same drugs, often made in the exact same place. This is wrong. This is unfair and together we will stop it, and we will stop it fast,” he said.
He did not offer any specific policy recommendation on how to address prescription drug costs, other than a comment on the need for greater price transparency.
“I am asking Congress to pass legislation that finally takes on the problem of global freeloading and delivers fairness and price transparency for American patients,” he said.
“We should also require drug companies, insurance companies, and hospitals to disclose real prices to foster competition and bring costs way down.”
SOURCE: Trump D. State of the Union Address, Feb. 5, 2019.
HIV/AIDS, pediatric cancer research, abortion, prescription drug prices, and preexisting conditions were among the health care highlights of President Donald Trump’s second State of the Union address Feb. 5.

Mr. Trump promised to push for funds to end HIV/AIDS and childhood cancer within in 10 years. “In recent years, we have made remarkable progress in the fight against HIV and AIDS. Scientific breakthroughs have brought a once-distant dream within reach,” he said to assembled members of Congress and leaders of the executive and judicial branches of government. “My budget will ask Democrats and Republicans to make the needed commitment to eliminate the HIV epidemic in the United States within 10 years.”
Following the speech, Alex Azar, secretary of the Department of Health and Human Services, offered more details in a blog post on the agency’s website.
Funding for the initiative, dubbed “Ending the HIV Epidemic: A Plan for America,” will have three components.
The first involves increasing investments in “geographic hotspots” though existing programs like the Ryan White HIV/AIDS Program and a new community health center–based program to provide antiretroviral therapy (ART) and preexposure prophylaxis (PrEP) to those at the highest risk of contracting the disease.
Second is the use of data to track where the disease is spreading most rapidly to help target prevention, care, and treatment at the local level. The third will provide funds for the creation of a local HIV HealthForce in these targeted areas to expand HIV prevention and treatment efforts.
A fact sheet on this initiative called for a 75% reduction in new cases of HIV infection in 5 years and at least a 90% reduction within 10 years.
President Trump called for similar efforts to address pediatric cancer.
“Tonight I am also asking you to join me in another fight that all American can get behind – the fight against childhood cancer,” he said, adding that his budget request will come with a line item of $500 million over 10 years to fund research. “Many childhood cancers have not seen new therapies in decades.”
President Trump also asked Congress to legislate a prohibition of late-term abortion.
“There could be no greater contrast to the beautiful image of a mother holding her infant child than the chilling displays our nation saw in recent days,” he said. “Lawmakers in New York cheered with delight upon the passage of legislation that would allow a baby to be ripped from the mother’s womb moments from birth. These are living, feeling beautiful babies who will never get the chance to share their love and their dreams with the world. ... Let us work together to build a culture that cherishes innocent life.”
He also touched on the recurring themes regarding lowering the cost of health care and prescription drugs, as well as protecting those with preexisting conditions, something he called a major priority.
“It’s unacceptable that Americans pay vastly more than people in other countries for the exact same drugs, often made in the exact same place. This is wrong. This is unfair and together we will stop it, and we will stop it fast,” he said.
He did not offer any specific policy recommendation on how to address prescription drug costs, other than a comment on the need for greater price transparency.
“I am asking Congress to pass legislation that finally takes on the problem of global freeloading and delivers fairness and price transparency for American patients,” he said.
“We should also require drug companies, insurance companies, and hospitals to disclose real prices to foster competition and bring costs way down.”
SOURCE: Trump D. State of the Union Address, Feb. 5, 2019.
Key clinical point: President Trump calls for an end to HIV/AIDS and pediatric cancer in 10 years.
Major finding: His budget will request $500 million for cancer research and as yet undisclosed amount for HIV/AIDS research.
Study details: More specific details on the proposals will likely come when the president makes his budget submission to Congress in the coming weeks.
Disclosures: There are no disclosures.
Source: Trump D. State of the Union Address, Feb. 5, 2019.
Winners and losers under bold Trump plan to slash drug rebate deals
Few consumers have heard of the secret, business-to-business payments that the Trump administration wants to ban in an attempt to control drug costs.
But the administration’s plan for drug rebates, announced Jan. 31, would end the pharmaceutical business as usual, shift billions in revenue and cause far-reaching, unforeseen change, say health policy authorities.
In pointed language sure to anger middlemen who benefit from the deals, administration officials proposed banning rebates paid by drug companies to ensure coverage for their products under Medicare and Medicaid plans.
“A shadowy system of kickbacks,” was how Health and Human Services Secretary Alex Azar described the current system in a speech on Feb. 1.
The proposal is a regulatory change applying only to Medicare plans for seniors and managed Medicaid plans for low-income people. But private insurers, who often take cues from government programs, might make a similar shift, administration officials said.
Drug rebates are essentially discounts off the list price. Outlawing them would divert $29 billion in rebates now paid to insurers and pharmacy benefit managers into “seniors’ pocketbooks at the pharmacy counter,” Azar said.
The measure already faces fierce opposition from some in the industry and is unlikely to be implemented as presented or by the proposed 2020 effective date, health policy analysts said.
In any event, it’s hardly a pure win for seniors or patients in general. Consumers are unlikely to collect the full benefit of eliminated rebates.
At the same time, the change would produce uncertain ricochets, including higher drug-plan premiums for consumers, that would produce new winners and losers across the economy.
“It is the most significant proposal that the administration has introduced so far” to try to control drug prices, said Rachel Sachs, a law professor at Washington University in St. Louis. “But I’m struck by the uncertainty that the administration has in what the effects would be.”
Chronically ill patients who take lots of expensive medicine
The list price for many brand-name medicines has doubled or tripled in recent years. But virtually the only ones affected by the full increases are the many patients who pay cash or whose out-of-pocket payments are based on the posted price.
By banning rebates, the administration says its intention is to ensure discounts are passed all the way to the patient instead of the middlemen, the so-called pharmacy benefit managers or PBMs. That means consumers using expensive drugs might see their out-of-pocket costs go down.
If rebates were eliminated for commercial insurance, where deductibles and out-of-pocket costs are generally much higher, chronically ill patients could benefit much more.
Drug companies
Ending rebates would give the administration a drug-policy “win” that doesn’t directly threaten pharmaceutical company profits.
“We applaud the administration for taking steps to reform the rebate system” Stephen Ubl, CEO of PhRMA, the main lobby for branded drugs, said after the proposal came out.
The change might also slow the soaring list-price increases that have become a publicity nightmare for the industry. When list prices pop by 5% or 10% each year, drugmakers pay part of the proceeds to insurers and PBMs in the form of rebates to guarantee health-plan coverage.
No one is claiming that eliminating rebates would stop escalating list prices, even if all insurers adopted the practice. But some believe it would remove an important factor.
Pharmacy benefit managers
PBMs reap billions of dollars in rebate revenue in return for putting particular products on lists of covered drugs. The administration is essentially proposing to make those payments illegal, at least for Medicare and Medicaid plans.
PBMs, which claim they control costs by negotiating with drugmakers, might have to go back to their roots – processing pharmacy claims for a fee. After recent industry consolidation into a few enormous companies, on the other hand, they might have the market power to charge very high fees, replacing much of the lost rebate revenue.
PBMs “are concerned” that the move “would increase drug costs and force Medicare beneficiaries to pay higher premiums and out-of-pocket expenses,” said JC Scott, CEO of the Pharmaceutical Care Management Association, the PBM lobby.
Insurance companies
Insurers, who often receive rebates directly, could also be hurt financially.
“From the start, the focus on rebates has been a distraction from the real issue – the problem is the price” of the drugs, said Matt Eyles, CEO of America’s Health Insurance Plans, a trade group. “We are not middlemen – we are your bargaining power, working hard to negotiate lower prices.”
Patients without chronic conditions and high drug costs
Lower out-of-pocket costs at the pharmacy counter would be financed, at least in part, by higher premiums for Medicare and Medicaid plans paid by consumers and the government. Premiums for Medicare Part D plans could rise from $3.20 to $5.64 per month, according to consultants hired by the Department of Health and Human Services.
“There is likely to be a wide variation in how much savings people see based on the drugs they take and the point-of-sale discounts that are negotiated,” said Elizabeth Carpenter, policy practice director at Avalere, a consultancy.
Consumers who don’t need expensive drugs every month could see insurance costs go up slightly without getting the benefits of lower out-of-pocket expense for purchased drugs.
Other policy changes giving health plans more negotiating power against drugmakers would keep a lid on premium increases, administration officials argue.
Kaiser Health News is a nonprofit national health policy news service. It is an editorially independent program of the Henry J. Kaiser Family Foundation that is not affiliated with Kaiser Permanente.
Few consumers have heard of the secret, business-to-business payments that the Trump administration wants to ban in an attempt to control drug costs.
But the administration’s plan for drug rebates, announced Jan. 31, would end the pharmaceutical business as usual, shift billions in revenue and cause far-reaching, unforeseen change, say health policy authorities.
In pointed language sure to anger middlemen who benefit from the deals, administration officials proposed banning rebates paid by drug companies to ensure coverage for their products under Medicare and Medicaid plans.
“A shadowy system of kickbacks,” was how Health and Human Services Secretary Alex Azar described the current system in a speech on Feb. 1.
The proposal is a regulatory change applying only to Medicare plans for seniors and managed Medicaid plans for low-income people. But private insurers, who often take cues from government programs, might make a similar shift, administration officials said.
Drug rebates are essentially discounts off the list price. Outlawing them would divert $29 billion in rebates now paid to insurers and pharmacy benefit managers into “seniors’ pocketbooks at the pharmacy counter,” Azar said.
The measure already faces fierce opposition from some in the industry and is unlikely to be implemented as presented or by the proposed 2020 effective date, health policy analysts said.
In any event, it’s hardly a pure win for seniors or patients in general. Consumers are unlikely to collect the full benefit of eliminated rebates.
At the same time, the change would produce uncertain ricochets, including higher drug-plan premiums for consumers, that would produce new winners and losers across the economy.
“It is the most significant proposal that the administration has introduced so far” to try to control drug prices, said Rachel Sachs, a law professor at Washington University in St. Louis. “But I’m struck by the uncertainty that the administration has in what the effects would be.”
Chronically ill patients who take lots of expensive medicine
The list price for many brand-name medicines has doubled or tripled in recent years. But virtually the only ones affected by the full increases are the many patients who pay cash or whose out-of-pocket payments are based on the posted price.
By banning rebates, the administration says its intention is to ensure discounts are passed all the way to the patient instead of the middlemen, the so-called pharmacy benefit managers or PBMs. That means consumers using expensive drugs might see their out-of-pocket costs go down.
If rebates were eliminated for commercial insurance, where deductibles and out-of-pocket costs are generally much higher, chronically ill patients could benefit much more.
Drug companies
Ending rebates would give the administration a drug-policy “win” that doesn’t directly threaten pharmaceutical company profits.
“We applaud the administration for taking steps to reform the rebate system” Stephen Ubl, CEO of PhRMA, the main lobby for branded drugs, said after the proposal came out.
The change might also slow the soaring list-price increases that have become a publicity nightmare for the industry. When list prices pop by 5% or 10% each year, drugmakers pay part of the proceeds to insurers and PBMs in the form of rebates to guarantee health-plan coverage.
No one is claiming that eliminating rebates would stop escalating list prices, even if all insurers adopted the practice. But some believe it would remove an important factor.
Pharmacy benefit managers
PBMs reap billions of dollars in rebate revenue in return for putting particular products on lists of covered drugs. The administration is essentially proposing to make those payments illegal, at least for Medicare and Medicaid plans.
PBMs, which claim they control costs by negotiating with drugmakers, might have to go back to their roots – processing pharmacy claims for a fee. After recent industry consolidation into a few enormous companies, on the other hand, they might have the market power to charge very high fees, replacing much of the lost rebate revenue.
PBMs “are concerned” that the move “would increase drug costs and force Medicare beneficiaries to pay higher premiums and out-of-pocket expenses,” said JC Scott, CEO of the Pharmaceutical Care Management Association, the PBM lobby.
Insurance companies
Insurers, who often receive rebates directly, could also be hurt financially.
“From the start, the focus on rebates has been a distraction from the real issue – the problem is the price” of the drugs, said Matt Eyles, CEO of America’s Health Insurance Plans, a trade group. “We are not middlemen – we are your bargaining power, working hard to negotiate lower prices.”
Patients without chronic conditions and high drug costs
Lower out-of-pocket costs at the pharmacy counter would be financed, at least in part, by higher premiums for Medicare and Medicaid plans paid by consumers and the government. Premiums for Medicare Part D plans could rise from $3.20 to $5.64 per month, according to consultants hired by the Department of Health and Human Services.
“There is likely to be a wide variation in how much savings people see based on the drugs they take and the point-of-sale discounts that are negotiated,” said Elizabeth Carpenter, policy practice director at Avalere, a consultancy.
Consumers who don’t need expensive drugs every month could see insurance costs go up slightly without getting the benefits of lower out-of-pocket expense for purchased drugs.
Other policy changes giving health plans more negotiating power against drugmakers would keep a lid on premium increases, administration officials argue.
Kaiser Health News is a nonprofit national health policy news service. It is an editorially independent program of the Henry J. Kaiser Family Foundation that is not affiliated with Kaiser Permanente.
Few consumers have heard of the secret, business-to-business payments that the Trump administration wants to ban in an attempt to control drug costs.
But the administration’s plan for drug rebates, announced Jan. 31, would end the pharmaceutical business as usual, shift billions in revenue and cause far-reaching, unforeseen change, say health policy authorities.
In pointed language sure to anger middlemen who benefit from the deals, administration officials proposed banning rebates paid by drug companies to ensure coverage for their products under Medicare and Medicaid plans.
“A shadowy system of kickbacks,” was how Health and Human Services Secretary Alex Azar described the current system in a speech on Feb. 1.
The proposal is a regulatory change applying only to Medicare plans for seniors and managed Medicaid plans for low-income people. But private insurers, who often take cues from government programs, might make a similar shift, administration officials said.
Drug rebates are essentially discounts off the list price. Outlawing them would divert $29 billion in rebates now paid to insurers and pharmacy benefit managers into “seniors’ pocketbooks at the pharmacy counter,” Azar said.
The measure already faces fierce opposition from some in the industry and is unlikely to be implemented as presented or by the proposed 2020 effective date, health policy analysts said.
In any event, it’s hardly a pure win for seniors or patients in general. Consumers are unlikely to collect the full benefit of eliminated rebates.
At the same time, the change would produce uncertain ricochets, including higher drug-plan premiums for consumers, that would produce new winners and losers across the economy.
“It is the most significant proposal that the administration has introduced so far” to try to control drug prices, said Rachel Sachs, a law professor at Washington University in St. Louis. “But I’m struck by the uncertainty that the administration has in what the effects would be.”
Chronically ill patients who take lots of expensive medicine
The list price for many brand-name medicines has doubled or tripled in recent years. But virtually the only ones affected by the full increases are the many patients who pay cash or whose out-of-pocket payments are based on the posted price.
By banning rebates, the administration says its intention is to ensure discounts are passed all the way to the patient instead of the middlemen, the so-called pharmacy benefit managers or PBMs. That means consumers using expensive drugs might see their out-of-pocket costs go down.
If rebates were eliminated for commercial insurance, where deductibles and out-of-pocket costs are generally much higher, chronically ill patients could benefit much more.
Drug companies
Ending rebates would give the administration a drug-policy “win” that doesn’t directly threaten pharmaceutical company profits.
“We applaud the administration for taking steps to reform the rebate system” Stephen Ubl, CEO of PhRMA, the main lobby for branded drugs, said after the proposal came out.
The change might also slow the soaring list-price increases that have become a publicity nightmare for the industry. When list prices pop by 5% or 10% each year, drugmakers pay part of the proceeds to insurers and PBMs in the form of rebates to guarantee health-plan coverage.
No one is claiming that eliminating rebates would stop escalating list prices, even if all insurers adopted the practice. But some believe it would remove an important factor.
Pharmacy benefit managers
PBMs reap billions of dollars in rebate revenue in return for putting particular products on lists of covered drugs. The administration is essentially proposing to make those payments illegal, at least for Medicare and Medicaid plans.
PBMs, which claim they control costs by negotiating with drugmakers, might have to go back to their roots – processing pharmacy claims for a fee. After recent industry consolidation into a few enormous companies, on the other hand, they might have the market power to charge very high fees, replacing much of the lost rebate revenue.
PBMs “are concerned” that the move “would increase drug costs and force Medicare beneficiaries to pay higher premiums and out-of-pocket expenses,” said JC Scott, CEO of the Pharmaceutical Care Management Association, the PBM lobby.
Insurance companies
Insurers, who often receive rebates directly, could also be hurt financially.
“From the start, the focus on rebates has been a distraction from the real issue – the problem is the price” of the drugs, said Matt Eyles, CEO of America’s Health Insurance Plans, a trade group. “We are not middlemen – we are your bargaining power, working hard to negotiate lower prices.”
Patients without chronic conditions and high drug costs
Lower out-of-pocket costs at the pharmacy counter would be financed, at least in part, by higher premiums for Medicare and Medicaid plans paid by consumers and the government. Premiums for Medicare Part D plans could rise from $3.20 to $5.64 per month, according to consultants hired by the Department of Health and Human Services.
“There is likely to be a wide variation in how much savings people see based on the drugs they take and the point-of-sale discounts that are negotiated,” said Elizabeth Carpenter, policy practice director at Avalere, a consultancy.
Consumers who don’t need expensive drugs every month could see insurance costs go up slightly without getting the benefits of lower out-of-pocket expense for purchased drugs.
Other policy changes giving health plans more negotiating power against drugmakers would keep a lid on premium increases, administration officials argue.
Kaiser Health News is a nonprofit national health policy news service. It is an editorially independent program of the Henry J. Kaiser Family Foundation that is not affiliated with Kaiser Permanente.
CMS proposing more flexibility in Medicare Advantage, Part D
More flexibility in benefits design could be coming to Medicare Advantage and the Part D prescription drug benefit if proposals offered by the Centers for Medicare & Medicaid Services are finalized.
The agency issued its proposed update for both programs for the 2020 plan year, which would allow Medicare Advantage plan sponsors to offer more specialized supplemental benefits for beneficiaries with chronic illnesses.
“For the 2020 plan year and beyond, Medicare Advantage plans will have greater flexibility to offer chronically ill patients any benefit that improves or maintains their health,” Demetrios Kouzoukas, CMS principal deputy administrator for Medicare and director of the Center for Medicare, said during a Jan. 30 press teleconference. “For example, plans could provide home-delivered or special meals in a far broader set of circumstances than what is allowed today.”
He noted that it would be up to the plans to determine what kinds of supplemental benefits would be offered and added that the offering of these benefits would not require a waiver, but would be evaluated as part of the plan’s overall bid submitted to the agency.
“We recognize that Medicare beneficiaries frequently have multiple chronic conditions,” Mr. Kouzoukas said. “We are excited that these changes will allow these beneficiaries to have new options for improving their health as a result of innovative health plan benefits.”
For Medicare Part D, the agency is specifically encouraging plan sponsors “to provide lower cost sharing for opioid reversal agents such as naloxone,” he added. The proposal also offers additional flexibility for plans to offer targeted benefits and cost sharing reductions to patients with chronic pain or those undergoing addiction treatment, according to a fact sheet highlighting key proposals.
Comments on the proposals are due by March 1. CMS expects to finalize the changes by the beginning of April.
More flexibility in benefits design could be coming to Medicare Advantage and the Part D prescription drug benefit if proposals offered by the Centers for Medicare & Medicaid Services are finalized.
The agency issued its proposed update for both programs for the 2020 plan year, which would allow Medicare Advantage plan sponsors to offer more specialized supplemental benefits for beneficiaries with chronic illnesses.
“For the 2020 plan year and beyond, Medicare Advantage plans will have greater flexibility to offer chronically ill patients any benefit that improves or maintains their health,” Demetrios Kouzoukas, CMS principal deputy administrator for Medicare and director of the Center for Medicare, said during a Jan. 30 press teleconference. “For example, plans could provide home-delivered or special meals in a far broader set of circumstances than what is allowed today.”
He noted that it would be up to the plans to determine what kinds of supplemental benefits would be offered and added that the offering of these benefits would not require a waiver, but would be evaluated as part of the plan’s overall bid submitted to the agency.
“We recognize that Medicare beneficiaries frequently have multiple chronic conditions,” Mr. Kouzoukas said. “We are excited that these changes will allow these beneficiaries to have new options for improving their health as a result of innovative health plan benefits.”
For Medicare Part D, the agency is specifically encouraging plan sponsors “to provide lower cost sharing for opioid reversal agents such as naloxone,” he added. The proposal also offers additional flexibility for plans to offer targeted benefits and cost sharing reductions to patients with chronic pain or those undergoing addiction treatment, according to a fact sheet highlighting key proposals.
Comments on the proposals are due by March 1. CMS expects to finalize the changes by the beginning of April.
More flexibility in benefits design could be coming to Medicare Advantage and the Part D prescription drug benefit if proposals offered by the Centers for Medicare & Medicaid Services are finalized.
The agency issued its proposed update for both programs for the 2020 plan year, which would allow Medicare Advantage plan sponsors to offer more specialized supplemental benefits for beneficiaries with chronic illnesses.
“For the 2020 plan year and beyond, Medicare Advantage plans will have greater flexibility to offer chronically ill patients any benefit that improves or maintains their health,” Demetrios Kouzoukas, CMS principal deputy administrator for Medicare and director of the Center for Medicare, said during a Jan. 30 press teleconference. “For example, plans could provide home-delivered or special meals in a far broader set of circumstances than what is allowed today.”
He noted that it would be up to the plans to determine what kinds of supplemental benefits would be offered and added that the offering of these benefits would not require a waiver, but would be evaluated as part of the plan’s overall bid submitted to the agency.
“We recognize that Medicare beneficiaries frequently have multiple chronic conditions,” Mr. Kouzoukas said. “We are excited that these changes will allow these beneficiaries to have new options for improving their health as a result of innovative health plan benefits.”
For Medicare Part D, the agency is specifically encouraging plan sponsors “to provide lower cost sharing for opioid reversal agents such as naloxone,” he added. The proposal also offers additional flexibility for plans to offer targeted benefits and cost sharing reductions to patients with chronic pain or those undergoing addiction treatment, according to a fact sheet highlighting key proposals.
Comments on the proposals are due by March 1. CMS expects to finalize the changes by the beginning of April.
Residential HCV program improves veterans’ diagnosis and care
Integrating comprehensive and collaborative hepatitis C virus (HCV) care within a Veterans Affairs residential treatment program can substantially increase diagnosis and treatment of HCV-infected veterans with substance use disorder (SUD), according to the results of an evaluation study for the period from December 2014 to April 2018.
A total of 97.5% (582/597) of patient admissions to the program were screened for HCV infection, and 12.7% (74/582) of the cases were confirmed to be HCV positive. All of the positive cases were sent to an infectious disease (ID) clinic for further evaluation and, if appropriate, to begin HCV pharmacotherapy, according to the report, published in the Journal of Substance Abuse Treatment.
Of the HCV-positive cases, 78.4% (58/74) received pharmacotherapy, with a sustained virologic response rate of 82.8% (48/58), wrote Mary Jane Burton, MD, of the G.V. (Sonny) Montgomery VA Medical Center, Jackson, Miss., and her colleagues.
As part of the program, all veterans admitted to the SUD residential program were offered screening for HCV. Veterans with negative screening results received education about how to remain HCV negative via handouts and veterans who screened positive received brief supportive counseling and were referred to the ID clinic via a consult. Veterans confirmed to have chronic HCV infection receive education and evaluation in the HCV clinic while they attend the residential SUD program. Treatment for HCV is instituted as early as feasible and prescribing is in accordance with VA guidelines (Department of Veterans Affairs, 2018), with the goal of initiating pharmacotherapy treatment for HCV while the veteran is still in the residential program, according to the researchers.
Following discharge from the program, veterans on HCV treatment are scheduled for follow-up every 2 weeks in the HCV treatment clinic for the remainder of their pharmacotherapy, the researchers added.
Patient-level barriers to HCV treatment among the SUD population include reduced health literacy, low health care utilization, comorbid mental health conditions, and poor social support, according to the literature. Because multidisciplinary approaches to HCV treatment that mitigate these barriers have been shown to increase treatment uptake among these patients, the VA program was initiated, the researchers stated. Dr. Burton and her colleagues reported that 18.9% (14/74) of the HCV-positive cases were newly diagnosed and would have likely gone undetected without this program (J Substance Abuse Treatment. 2019;98:9-14).
“We have demonstrated that integrating a comprehensive HCV screening, education, referral, and treatment program within residential SUD treatment is feasible and effective in diagnosing previously unrecognized HCV infections, transitioning veterans into HCV care, and promoting treatment initiation,” the researchers concluded.
The Department of Veterans Affairs and the VA Center for Innovation supported the study. Dr. Burton reported research support from Merck Sharpe & Dohme.
Integrating comprehensive and collaborative hepatitis C virus (HCV) care within a Veterans Affairs residential treatment program can substantially increase diagnosis and treatment of HCV-infected veterans with substance use disorder (SUD), according to the results of an evaluation study for the period from December 2014 to April 2018.
A total of 97.5% (582/597) of patient admissions to the program were screened for HCV infection, and 12.7% (74/582) of the cases were confirmed to be HCV positive. All of the positive cases were sent to an infectious disease (ID) clinic for further evaluation and, if appropriate, to begin HCV pharmacotherapy, according to the report, published in the Journal of Substance Abuse Treatment.
Of the HCV-positive cases, 78.4% (58/74) received pharmacotherapy, with a sustained virologic response rate of 82.8% (48/58), wrote Mary Jane Burton, MD, of the G.V. (Sonny) Montgomery VA Medical Center, Jackson, Miss., and her colleagues.
As part of the program, all veterans admitted to the SUD residential program were offered screening for HCV. Veterans with negative screening results received education about how to remain HCV negative via handouts and veterans who screened positive received brief supportive counseling and were referred to the ID clinic via a consult. Veterans confirmed to have chronic HCV infection receive education and evaluation in the HCV clinic while they attend the residential SUD program. Treatment for HCV is instituted as early as feasible and prescribing is in accordance with VA guidelines (Department of Veterans Affairs, 2018), with the goal of initiating pharmacotherapy treatment for HCV while the veteran is still in the residential program, according to the researchers.
Following discharge from the program, veterans on HCV treatment are scheduled for follow-up every 2 weeks in the HCV treatment clinic for the remainder of their pharmacotherapy, the researchers added.
Patient-level barriers to HCV treatment among the SUD population include reduced health literacy, low health care utilization, comorbid mental health conditions, and poor social support, according to the literature. Because multidisciplinary approaches to HCV treatment that mitigate these barriers have been shown to increase treatment uptake among these patients, the VA program was initiated, the researchers stated. Dr. Burton and her colleagues reported that 18.9% (14/74) of the HCV-positive cases were newly diagnosed and would have likely gone undetected without this program (J Substance Abuse Treatment. 2019;98:9-14).
“We have demonstrated that integrating a comprehensive HCV screening, education, referral, and treatment program within residential SUD treatment is feasible and effective in diagnosing previously unrecognized HCV infections, transitioning veterans into HCV care, and promoting treatment initiation,” the researchers concluded.
The Department of Veterans Affairs and the VA Center for Innovation supported the study. Dr. Burton reported research support from Merck Sharpe & Dohme.
Integrating comprehensive and collaborative hepatitis C virus (HCV) care within a Veterans Affairs residential treatment program can substantially increase diagnosis and treatment of HCV-infected veterans with substance use disorder (SUD), according to the results of an evaluation study for the period from December 2014 to April 2018.
A total of 97.5% (582/597) of patient admissions to the program were screened for HCV infection, and 12.7% (74/582) of the cases were confirmed to be HCV positive. All of the positive cases were sent to an infectious disease (ID) clinic for further evaluation and, if appropriate, to begin HCV pharmacotherapy, according to the report, published in the Journal of Substance Abuse Treatment.
Of the HCV-positive cases, 78.4% (58/74) received pharmacotherapy, with a sustained virologic response rate of 82.8% (48/58), wrote Mary Jane Burton, MD, of the G.V. (Sonny) Montgomery VA Medical Center, Jackson, Miss., and her colleagues.
As part of the program, all veterans admitted to the SUD residential program were offered screening for HCV. Veterans with negative screening results received education about how to remain HCV negative via handouts and veterans who screened positive received brief supportive counseling and were referred to the ID clinic via a consult. Veterans confirmed to have chronic HCV infection receive education and evaluation in the HCV clinic while they attend the residential SUD program. Treatment for HCV is instituted as early as feasible and prescribing is in accordance with VA guidelines (Department of Veterans Affairs, 2018), with the goal of initiating pharmacotherapy treatment for HCV while the veteran is still in the residential program, according to the researchers.
Following discharge from the program, veterans on HCV treatment are scheduled for follow-up every 2 weeks in the HCV treatment clinic for the remainder of their pharmacotherapy, the researchers added.
Patient-level barriers to HCV treatment among the SUD population include reduced health literacy, low health care utilization, comorbid mental health conditions, and poor social support, according to the literature. Because multidisciplinary approaches to HCV treatment that mitigate these barriers have been shown to increase treatment uptake among these patients, the VA program was initiated, the researchers stated. Dr. Burton and her colleagues reported that 18.9% (14/74) of the HCV-positive cases were newly diagnosed and would have likely gone undetected without this program (J Substance Abuse Treatment. 2019;98:9-14).
“We have demonstrated that integrating a comprehensive HCV screening, education, referral, and treatment program within residential SUD treatment is feasible and effective in diagnosing previously unrecognized HCV infections, transitioning veterans into HCV care, and promoting treatment initiation,” the researchers concluded.
The Department of Veterans Affairs and the VA Center for Innovation supported the study. Dr. Burton reported research support from Merck Sharpe & Dohme.
FROM THE JOURNAL OF SUBSTANCE ABUSE TREATMENT
Drug-pricing policies find new momentum as ‘a 2020 thing’
The next presidential primary contests are more than a year away.
“This is a 2020 thing,” said Peter B. Bach, MD, who directs the Center for Health Policy and Outcomes at Memorial Sloan Kettering Cancer Center in New York and tracks drug-pricing policy.
Spurred on by midterm election results that showed health care to be a deciding issue, lawmakers – some of whom have already launched presidential-run exploratory committees – are pushing a bevy of new proposals and approaches.
Few if any of those ideas will likely make it to the president’s desk. Nevertheless, Senate Democrats eyeing higher office and seeking street cred in the debate are devising more innovative and aggressive strategies to take on Big Pharma.
“Democrats feel as if they’re really able to experiment,” said Rachel Sachs, an associate law professor at Washington University, St. Louis, who tracks drug-pricing laws.
Some Republicans are also proposing drug-pricing reform, although experts say their approaches are generally less dramatic.
Here are some of the ideas either introduced in legislation or that senators’ offices confirmed they are considering:
- Make a public option for generic drugs. The government could manufacture generics (directly or through a private contractor) if there is a shortage or aren’t enough competitors to keep prices down. This comes from a bill put forth by Sen. Elizabeth Warren (D-Mass.) and Rep. Jan Schakowsky (D-Ill.).
- Let Medicare negotiate drug prices. This idea has many backers – what differs is the method of enforcement. Sen. Sherrod Brown (D-Ohio) has suggested that if the company and the government can’t reach an agreement, the government could take away the company’s patent rights. A proposal from Sen. Bernie Sanders (I-Vt.) and Rep. Elijah Cummings (D-Md.) would address stalled negotiations by letting Medicare pay the lowest amount among: Medicaid’s best price, the highest price a single federal purchaser pays or the median price paid for a specific drug in Canada, France, Germany, Japan, and the United Kingdom.
- Pay what they do abroad. Legislation from Mr. Sanders and Rep. Ro Khanna (D-Calif.) would require companies to price their drugs no higher than the median of what’s charged in Canada, France, Germany, Japan, and the United Kingdom. If manufacturers fail to comply, other companies could get the rights to make those drugs, too.
- Penalize price gouging. This would target manufacturers who raise drug prices more than 30% in 5 years. Punishments could include requiring the company to reimburse those who paid the elevated price, forcing the drug maker to lower its price, or charging a penalty up to three times what a company received from boosting the price. Backers include senators Richard Blumenthal (D-Conn.), Kamala Harris (D-Calif.), Jeff Merkley (D-Ore.), and Amy Klobuchar (D-Minn.).
- Import drugs. A Sanders-Cummings bill would let patients, wholesalers, and pharmacies import drugs from abroad – starting with Canada, and leaving the door open for some other countries. Sen. Chuck Grassley (R-Iowa) and Ms. Klobuchar have a separate bill that is specific to patients getting medicine from Canada alone.
- Abolish “pay for delay.” From Mr. Grassley and Ms. Klobuchar, this legislation would tackle deals in which a branded drugmaker pays off a generic one to keep a competing product from coming to market.
This flurry of proposed lawmaking could add momentum to one of the few policy areas in which conventional Washington wisdom suggests House Democrats, Senate Republicans, and the White House may be able to find common ground.
“Everything is up in the air and anything is possible,” said Walid Gellad, MD, codirector of the Center for Pharmaceutical Policy and Prescribing at the University of Pittsburgh. “There are things that can happen that maybe weren’t going to happen before.”
And there’s political pressure. Polls consistently suggest voters have a strong appetite for action. As a candidate, President Trump vowed to make drug prices a top priority. In recent months, the administration has taken steps in this direction, like testing changes to Medicare that might reduce out-of-pocket drug costs. But Congress has been relatively quiet, especially when it comes to challenging the pharmaceutical industry, which remains one of Capitol Hill’s most potent lobbying forces.
One aspect of prescription drug pricing that could see bipartisan action is insulin prices, which have skyrocketed, stoking widespread outcry, and could be a target for bipartisan work. Ms. Warren’s legislation singles out the drug as one the government could produce, and Mr. Cummings has already called in major insulin manufacturers for a drug-pricing hearing later this month. In addition, Rep. Diana DeGette (D-Colo.), the new chair of the House Energy and Commerce Oversight and Investigations Subcommittee, has listed prescription drug pricing as a high priority for her panel. As cochair of the Congressional Diabetes Caucus, Ms. DeGette worked with Rep. Tom Reed (R-N.Y.) to produce a report on the high cost of insulin.
To be sure, some of the concepts, such as drug importation and bolstering development of generic drugs, have been around a long time. But some of the legislation at hand suggests a new kind of thinking.
House Speaker Nancy Pelosi (D-Calif.) has labeled drug pricing a top priority, and the pharmaceutical industry has been bracing for a fight with the new Democratic majority.
Meanwhile, in the GOP-controlled Senate, two powerful lawmakers – Sen. Lamar Alexander (R-Tenn.) and Mr. Grassley – have indicated they want to use their influence to tackle the issue. Mr. Alexander, who chairs the Health, Education, Labor and Pensions Committee, has said cutting health care costs, including drug prices, will be high on his panel’s to-do list this Congress. Mr. Grassley runs the Finance Committee, which oversees pricing issues for Medicare and Medicaid.
“The solution to high drug prices is not just having the government spending more money. ... You need to look at prices,” Dr. Gellad said. “These proposals deal with price. They all directly affect price.”
Given the drug industry’s full-throated opposition to virtually any pricing legislation, Ms. Sachs said, “it is not at all surprising to me to see the Democrats start exploring some of these more radical proposals.”
Still, though, Senate staffers almost uniformly argued that the drug-pricing issue requires more than one single piece of legislation.
For instance, the price-gouging penalty spearheaded by Mr. Blumenthal doesn’t stop drugs from having high initial list prices. Letting Medicare negotiate doesn’t mean people covered by other plans will necessarily see the same savings. Empowering the government to produce competing drugs doesn’t promise to keep prices down long term and doesn’t guarantee that patients will see those savings.
“We need to use every tool available to bring down drug prices and improve competition,” said an aide in Ms. Warren’s office.
KHN’s coverage of prescription drug development, costs and pricing is supported in part by the Laura and John Arnold Foundation. Kaiser Health News is a nonprofit national health policy news service. It is an editorially independent program of the Henry J. Kaiser Family Foundation that is not affiliated with Kaiser Permanente.
The next presidential primary contests are more than a year away.
“This is a 2020 thing,” said Peter B. Bach, MD, who directs the Center for Health Policy and Outcomes at Memorial Sloan Kettering Cancer Center in New York and tracks drug-pricing policy.
Spurred on by midterm election results that showed health care to be a deciding issue, lawmakers – some of whom have already launched presidential-run exploratory committees – are pushing a bevy of new proposals and approaches.
Few if any of those ideas will likely make it to the president’s desk. Nevertheless, Senate Democrats eyeing higher office and seeking street cred in the debate are devising more innovative and aggressive strategies to take on Big Pharma.
“Democrats feel as if they’re really able to experiment,” said Rachel Sachs, an associate law professor at Washington University, St. Louis, who tracks drug-pricing laws.
Some Republicans are also proposing drug-pricing reform, although experts say their approaches are generally less dramatic.
Here are some of the ideas either introduced in legislation or that senators’ offices confirmed they are considering:
- Make a public option for generic drugs. The government could manufacture generics (directly or through a private contractor) if there is a shortage or aren’t enough competitors to keep prices down. This comes from a bill put forth by Sen. Elizabeth Warren (D-Mass.) and Rep. Jan Schakowsky (D-Ill.).
- Let Medicare negotiate drug prices. This idea has many backers – what differs is the method of enforcement. Sen. Sherrod Brown (D-Ohio) has suggested that if the company and the government can’t reach an agreement, the government could take away the company’s patent rights. A proposal from Sen. Bernie Sanders (I-Vt.) and Rep. Elijah Cummings (D-Md.) would address stalled negotiations by letting Medicare pay the lowest amount among: Medicaid’s best price, the highest price a single federal purchaser pays or the median price paid for a specific drug in Canada, France, Germany, Japan, and the United Kingdom.
- Pay what they do abroad. Legislation from Mr. Sanders and Rep. Ro Khanna (D-Calif.) would require companies to price their drugs no higher than the median of what’s charged in Canada, France, Germany, Japan, and the United Kingdom. If manufacturers fail to comply, other companies could get the rights to make those drugs, too.
- Penalize price gouging. This would target manufacturers who raise drug prices more than 30% in 5 years. Punishments could include requiring the company to reimburse those who paid the elevated price, forcing the drug maker to lower its price, or charging a penalty up to three times what a company received from boosting the price. Backers include senators Richard Blumenthal (D-Conn.), Kamala Harris (D-Calif.), Jeff Merkley (D-Ore.), and Amy Klobuchar (D-Minn.).
- Import drugs. A Sanders-Cummings bill would let patients, wholesalers, and pharmacies import drugs from abroad – starting with Canada, and leaving the door open for some other countries. Sen. Chuck Grassley (R-Iowa) and Ms. Klobuchar have a separate bill that is specific to patients getting medicine from Canada alone.
- Abolish “pay for delay.” From Mr. Grassley and Ms. Klobuchar, this legislation would tackle deals in which a branded drugmaker pays off a generic one to keep a competing product from coming to market.
This flurry of proposed lawmaking could add momentum to one of the few policy areas in which conventional Washington wisdom suggests House Democrats, Senate Republicans, and the White House may be able to find common ground.
“Everything is up in the air and anything is possible,” said Walid Gellad, MD, codirector of the Center for Pharmaceutical Policy and Prescribing at the University of Pittsburgh. “There are things that can happen that maybe weren’t going to happen before.”
And there’s political pressure. Polls consistently suggest voters have a strong appetite for action. As a candidate, President Trump vowed to make drug prices a top priority. In recent months, the administration has taken steps in this direction, like testing changes to Medicare that might reduce out-of-pocket drug costs. But Congress has been relatively quiet, especially when it comes to challenging the pharmaceutical industry, which remains one of Capitol Hill’s most potent lobbying forces.
One aspect of prescription drug pricing that could see bipartisan action is insulin prices, which have skyrocketed, stoking widespread outcry, and could be a target for bipartisan work. Ms. Warren’s legislation singles out the drug as one the government could produce, and Mr. Cummings has already called in major insulin manufacturers for a drug-pricing hearing later this month. In addition, Rep. Diana DeGette (D-Colo.), the new chair of the House Energy and Commerce Oversight and Investigations Subcommittee, has listed prescription drug pricing as a high priority for her panel. As cochair of the Congressional Diabetes Caucus, Ms. DeGette worked with Rep. Tom Reed (R-N.Y.) to produce a report on the high cost of insulin.
To be sure, some of the concepts, such as drug importation and bolstering development of generic drugs, have been around a long time. But some of the legislation at hand suggests a new kind of thinking.
House Speaker Nancy Pelosi (D-Calif.) has labeled drug pricing a top priority, and the pharmaceutical industry has been bracing for a fight with the new Democratic majority.
Meanwhile, in the GOP-controlled Senate, two powerful lawmakers – Sen. Lamar Alexander (R-Tenn.) and Mr. Grassley – have indicated they want to use their influence to tackle the issue. Mr. Alexander, who chairs the Health, Education, Labor and Pensions Committee, has said cutting health care costs, including drug prices, will be high on his panel’s to-do list this Congress. Mr. Grassley runs the Finance Committee, which oversees pricing issues for Medicare and Medicaid.
“The solution to high drug prices is not just having the government spending more money. ... You need to look at prices,” Dr. Gellad said. “These proposals deal with price. They all directly affect price.”
Given the drug industry’s full-throated opposition to virtually any pricing legislation, Ms. Sachs said, “it is not at all surprising to me to see the Democrats start exploring some of these more radical proposals.”
Still, though, Senate staffers almost uniformly argued that the drug-pricing issue requires more than one single piece of legislation.
For instance, the price-gouging penalty spearheaded by Mr. Blumenthal doesn’t stop drugs from having high initial list prices. Letting Medicare negotiate doesn’t mean people covered by other plans will necessarily see the same savings. Empowering the government to produce competing drugs doesn’t promise to keep prices down long term and doesn’t guarantee that patients will see those savings.
“We need to use every tool available to bring down drug prices and improve competition,” said an aide in Ms. Warren’s office.
KHN’s coverage of prescription drug development, costs and pricing is supported in part by the Laura and John Arnold Foundation. Kaiser Health News is a nonprofit national health policy news service. It is an editorially independent program of the Henry J. Kaiser Family Foundation that is not affiliated with Kaiser Permanente.
The next presidential primary contests are more than a year away.
“This is a 2020 thing,” said Peter B. Bach, MD, who directs the Center for Health Policy and Outcomes at Memorial Sloan Kettering Cancer Center in New York and tracks drug-pricing policy.
Spurred on by midterm election results that showed health care to be a deciding issue, lawmakers – some of whom have already launched presidential-run exploratory committees – are pushing a bevy of new proposals and approaches.
Few if any of those ideas will likely make it to the president’s desk. Nevertheless, Senate Democrats eyeing higher office and seeking street cred in the debate are devising more innovative and aggressive strategies to take on Big Pharma.
“Democrats feel as if they’re really able to experiment,” said Rachel Sachs, an associate law professor at Washington University, St. Louis, who tracks drug-pricing laws.
Some Republicans are also proposing drug-pricing reform, although experts say their approaches are generally less dramatic.
Here are some of the ideas either introduced in legislation or that senators’ offices confirmed they are considering:
- Make a public option for generic drugs. The government could manufacture generics (directly or through a private contractor) if there is a shortage or aren’t enough competitors to keep prices down. This comes from a bill put forth by Sen. Elizabeth Warren (D-Mass.) and Rep. Jan Schakowsky (D-Ill.).
- Let Medicare negotiate drug prices. This idea has many backers – what differs is the method of enforcement. Sen. Sherrod Brown (D-Ohio) has suggested that if the company and the government can’t reach an agreement, the government could take away the company’s patent rights. A proposal from Sen. Bernie Sanders (I-Vt.) and Rep. Elijah Cummings (D-Md.) would address stalled negotiations by letting Medicare pay the lowest amount among: Medicaid’s best price, the highest price a single federal purchaser pays or the median price paid for a specific drug in Canada, France, Germany, Japan, and the United Kingdom.
- Pay what they do abroad. Legislation from Mr. Sanders and Rep. Ro Khanna (D-Calif.) would require companies to price their drugs no higher than the median of what’s charged in Canada, France, Germany, Japan, and the United Kingdom. If manufacturers fail to comply, other companies could get the rights to make those drugs, too.
- Penalize price gouging. This would target manufacturers who raise drug prices more than 30% in 5 years. Punishments could include requiring the company to reimburse those who paid the elevated price, forcing the drug maker to lower its price, or charging a penalty up to three times what a company received from boosting the price. Backers include senators Richard Blumenthal (D-Conn.), Kamala Harris (D-Calif.), Jeff Merkley (D-Ore.), and Amy Klobuchar (D-Minn.).
- Import drugs. A Sanders-Cummings bill would let patients, wholesalers, and pharmacies import drugs from abroad – starting with Canada, and leaving the door open for some other countries. Sen. Chuck Grassley (R-Iowa) and Ms. Klobuchar have a separate bill that is specific to patients getting medicine from Canada alone.
- Abolish “pay for delay.” From Mr. Grassley and Ms. Klobuchar, this legislation would tackle deals in which a branded drugmaker pays off a generic one to keep a competing product from coming to market.
This flurry of proposed lawmaking could add momentum to one of the few policy areas in which conventional Washington wisdom suggests House Democrats, Senate Republicans, and the White House may be able to find common ground.
“Everything is up in the air and anything is possible,” said Walid Gellad, MD, codirector of the Center for Pharmaceutical Policy and Prescribing at the University of Pittsburgh. “There are things that can happen that maybe weren’t going to happen before.”
And there’s political pressure. Polls consistently suggest voters have a strong appetite for action. As a candidate, President Trump vowed to make drug prices a top priority. In recent months, the administration has taken steps in this direction, like testing changes to Medicare that might reduce out-of-pocket drug costs. But Congress has been relatively quiet, especially when it comes to challenging the pharmaceutical industry, which remains one of Capitol Hill’s most potent lobbying forces.
One aspect of prescription drug pricing that could see bipartisan action is insulin prices, which have skyrocketed, stoking widespread outcry, and could be a target for bipartisan work. Ms. Warren’s legislation singles out the drug as one the government could produce, and Mr. Cummings has already called in major insulin manufacturers for a drug-pricing hearing later this month. In addition, Rep. Diana DeGette (D-Colo.), the new chair of the House Energy and Commerce Oversight and Investigations Subcommittee, has listed prescription drug pricing as a high priority for her panel. As cochair of the Congressional Diabetes Caucus, Ms. DeGette worked with Rep. Tom Reed (R-N.Y.) to produce a report on the high cost of insulin.
To be sure, some of the concepts, such as drug importation and bolstering development of generic drugs, have been around a long time. But some of the legislation at hand suggests a new kind of thinking.
House Speaker Nancy Pelosi (D-Calif.) has labeled drug pricing a top priority, and the pharmaceutical industry has been bracing for a fight with the new Democratic majority.
Meanwhile, in the GOP-controlled Senate, two powerful lawmakers – Sen. Lamar Alexander (R-Tenn.) and Mr. Grassley – have indicated they want to use their influence to tackle the issue. Mr. Alexander, who chairs the Health, Education, Labor and Pensions Committee, has said cutting health care costs, including drug prices, will be high on his panel’s to-do list this Congress. Mr. Grassley runs the Finance Committee, which oversees pricing issues for Medicare and Medicaid.
“The solution to high drug prices is not just having the government spending more money. ... You need to look at prices,” Dr. Gellad said. “These proposals deal with price. They all directly affect price.”
Given the drug industry’s full-throated opposition to virtually any pricing legislation, Ms. Sachs said, “it is not at all surprising to me to see the Democrats start exploring some of these more radical proposals.”
Still, though, Senate staffers almost uniformly argued that the drug-pricing issue requires more than one single piece of legislation.
For instance, the price-gouging penalty spearheaded by Mr. Blumenthal doesn’t stop drugs from having high initial list prices. Letting Medicare negotiate doesn’t mean people covered by other plans will necessarily see the same savings. Empowering the government to produce competing drugs doesn’t promise to keep prices down long term and doesn’t guarantee that patients will see those savings.
“We need to use every tool available to bring down drug prices and improve competition,” said an aide in Ms. Warren’s office.
KHN’s coverage of prescription drug development, costs and pricing is supported in part by the Laura and John Arnold Foundation. Kaiser Health News is a nonprofit national health policy news service. It is an editorially independent program of the Henry J. Kaiser Family Foundation that is not affiliated with Kaiser Permanente.
Survey: Americans support Medicare for all
A majority of Americans support the concept of Medicare for all, but “larger majorities favor more incremental changes to the health care system,” according to a new survey by the Kaiser Family Foundation.
Support for a Medicare-for-all health care system came in at 56% (strongly favor, 34%; somewhat favor, 22%) among the 1,190 respondents to the latest KFF Health Tracking Poll, which was conducted Jan. 9-14, 2019. That support came largely from Democrats, 81% of whom favored the plan, compared with only 23% of Republicans, the Kaiser investigators said Jan. 23.
A Medicare buy-in plan for Americans aged 50-64 years also was highly popular, receiving support from 77% of all respondents – 85% of Democrats, 75% of Independents, and 69% of Republicans. Support by party identification was similar for a proposal to enable all those who don’t have employer-based insurance to get coverage through state Medicaid programs, which received 75% support overall, they reported.
Just behind those proposals at 74% support was a federally administered health plan that would be open to anyone but would allow people to keep the coverage they have. It was the most popular proposal among Democrats (91%) but did not garner a majority among Republicans (47%), the investigators said.
Support for the Medicare-for-all plan varied considerably, depending on number of arguments presented to respondents. When told that such a proposal would guarantee insurance as a right for all Americans, 71% favored it, and when they heard that it would eliminate health insurance premiums and reduce out-of-pockets costs, 67% of respondents expressed support. Favorable responses, however, were in the minority when people were told that Medicare-for-all would eliminate private health insurance companies (37%), threaten the current Medicare program (32%), and lead to some delayed medical tests and treatments (26%), according to the Kaiser report.
A majority of Americans support the concept of Medicare for all, but “larger majorities favor more incremental changes to the health care system,” according to a new survey by the Kaiser Family Foundation.

Support for a Medicare-for-all health care system came in at 56% (strongly favor, 34%; somewhat favor, 22%) among the 1,190 respondents to the latest KFF Health Tracking Poll, which was conducted Jan. 9-14, 2019. That support came largely from Democrats, 81% of whom favored the plan, compared with only 23% of Republicans, the Kaiser investigators said Jan. 23.
A Medicare buy-in plan for Americans aged 50-64 years also was highly popular, receiving support from 77% of all respondents – 85% of Democrats, 75% of Independents, and 69% of Republicans. Support by party identification was similar for a proposal to enable all those who don’t have employer-based insurance to get coverage through state Medicaid programs, which received 75% support overall, they reported.
Just behind those proposals at 74% support was a federally administered health plan that would be open to anyone but would allow people to keep the coverage they have. It was the most popular proposal among Democrats (91%) but did not garner a majority among Republicans (47%), the investigators said.
Support for the Medicare-for-all plan varied considerably, depending on number of arguments presented to respondents. When told that such a proposal would guarantee insurance as a right for all Americans, 71% favored it, and when they heard that it would eliminate health insurance premiums and reduce out-of-pockets costs, 67% of respondents expressed support. Favorable responses, however, were in the minority when people were told that Medicare-for-all would eliminate private health insurance companies (37%), threaten the current Medicare program (32%), and lead to some delayed medical tests and treatments (26%), according to the Kaiser report.
A majority of Americans support the concept of Medicare for all, but “larger majorities favor more incremental changes to the health care system,” according to a new survey by the Kaiser Family Foundation.

Support for a Medicare-for-all health care system came in at 56% (strongly favor, 34%; somewhat favor, 22%) among the 1,190 respondents to the latest KFF Health Tracking Poll, which was conducted Jan. 9-14, 2019. That support came largely from Democrats, 81% of whom favored the plan, compared with only 23% of Republicans, the Kaiser investigators said Jan. 23.
A Medicare buy-in plan for Americans aged 50-64 years also was highly popular, receiving support from 77% of all respondents – 85% of Democrats, 75% of Independents, and 69% of Republicans. Support by party identification was similar for a proposal to enable all those who don’t have employer-based insurance to get coverage through state Medicaid programs, which received 75% support overall, they reported.
Just behind those proposals at 74% support was a federally administered health plan that would be open to anyone but would allow people to keep the coverage they have. It was the most popular proposal among Democrats (91%) but did not garner a majority among Republicans (47%), the investigators said.
Support for the Medicare-for-all plan varied considerably, depending on number of arguments presented to respondents. When told that such a proposal would guarantee insurance as a right for all Americans, 71% favored it, and when they heard that it would eliminate health insurance premiums and reduce out-of-pockets costs, 67% of respondents expressed support. Favorable responses, however, were in the minority when people were told that Medicare-for-all would eliminate private health insurance companies (37%), threaten the current Medicare program (32%), and lead to some delayed medical tests and treatments (26%), according to the Kaiser report.
Trump zeroes in on surprise medical bills in White House chat with patients, experts
President Trump on Jan. 23 instructed administration officials to investigate how to prevent surprise medical bills, broadening his focus on drug prices to include other issues of price transparency in health care.

several attendees said.
“The pricing is hurting patients, and we’ve stopped a lot of it, but we’re going to stop all of it,” Mr. Trump said during a roundtable discussion when reporters were briefly allowed into the otherwise closed-door meeting.
David Silverstein, the founder of a Colorado-based nonprofit called Broken Healthcare who attended, said Mr. Trump struck an aggressive tone, calling for a solution with “the biggest teeth you can find.”
“Reading the tea leaves, I think there’s big change coming,” Mr. Silverstein said.
Surprise billing, or the practice of charging patients for care that is more expensive than anticipated or not covered by their insurance, has received a flood of attention in the past year, particularly as Kaiser Health News and other news organizations have undertaken investigations into patients’ most outrageous medical bills.
Attendees said each of 10 invited guests – among them patients as well as doctors with their own stories of unexpected bills – was given an opportunity to talk, though Mr. Trump did not stay to hear all of their stories during the roughly hour-long gathering.
The group included Paul Davis, a retired doctor from Findlay, Ohio, whose family’s experience with a $17,850 bill for a simple urine test was detailed in a KHN-NPR “Bill of the Month” feature last year.
Mr. Davis’ daughter, Elizabeth Moreno, was a college student in Texas when she had spinal surgery to remedy debilitating back pain. After the surgery, she was asked to provide a urine sample and later received a bill from an out-of-network lab in Houston that tested it. Experts said such tests rarely cost more than $200, not nearly what the lab charged Ms. Moreno and her insurance company. But fearing damage to his daughter’s credit, Mr. Davis paid the lab $5,000 and filed a complaint with the Texas attorney general’s office, alleging “price gouging of staggering proportions.”
Mr. Davis said White House officials made it clear that price transparency is a “high priority” for Trump, and while they did not see eye to eye on every subject, he said he was struck by their sincerity.
“These people seemed earnest in wanting to do something constructive to fix this,” Mr. Davis said.
Dr. Martin Makary, a surgeon and health policy expert at Johns Hopkins University who has written about transparency in health care and attended the meeting, said it was a good opportunity for the White House to hear firsthand about a serious and widespread issue.
“This is how most of America lives, and [Americans are] getting hammered,” he said.
Mr. Trump has often railed against high prescription drug prices but has said less about other problems with the nation’s health care system. In October, shortly before the midterm elections, he unveiled a proposal to tie the price Medicare pays for some drugs to the prices paid for the same drugs overseas, for example.
Mr. Trump, Mr. Azar, and Mr. Acosta said efforts to control costs in health care were yielding positive results, discussing in particular the expansion of association health plans and the new requirement that hospitals post their list prices online. The president also took credit for the recent increase in generic drug approvals, which he said would help lower drug prices.
Discussing the partial government shutdown, Mr. Trump said Americans “want to see what we’re doing, like today we lowered prescription drug prices, first time in 50 years,” according to a White House pool report.
Mr. Trump appeared to be referring to a recent claim by the White House Council of Economic Advisers that prescription drug prices fell last year.
However, as STAT pointed out in a recent fact check, the report from which that claim was gleaned said “growth in relative drug prices has slowed since January 2017,” not that there was an overall decrease in prices.
Annual increases in overall drug spending have leveled off as pharmaceutical companies have released fewer blockbuster drugs; patents have expired on brand-name drugs; and the waning effect of a spike driven by the release of astronomically expensive drugs to treat hepatitis C. Drugmakers are also wary of increasing their prices in the midst of growing political pressure.
Since Democrats seized control of the House of Representatives this month, party leaders have rushed to announce investigations and schedule hearings dealing with health care, focusing in particular on drug costs and protections for those with preexisting conditions.
Recently, the House Oversight Committee announced a “sweeping” investigation into drug prices, pointing to an AARP report saying the vast majority of brand-name drugs had more than doubled in price between 2005 and 2017.
KHN correspondents Shefali Luthra and Jay Hancock contributed to this report. Kaiser Health News is a nonprofit national health policy news service. It is an editorially independent program of the Henry J. Kaiser Family Foundation that is not affiliated with Kaiser Permanente.
President Trump on Jan. 23 instructed administration officials to investigate how to prevent surprise medical bills, broadening his focus on drug prices to include other issues of price transparency in health care.

several attendees said.
“The pricing is hurting patients, and we’ve stopped a lot of it, but we’re going to stop all of it,” Mr. Trump said during a roundtable discussion when reporters were briefly allowed into the otherwise closed-door meeting.
David Silverstein, the founder of a Colorado-based nonprofit called Broken Healthcare who attended, said Mr. Trump struck an aggressive tone, calling for a solution with “the biggest teeth you can find.”
“Reading the tea leaves, I think there’s big change coming,” Mr. Silverstein said.
Surprise billing, or the practice of charging patients for care that is more expensive than anticipated or not covered by their insurance, has received a flood of attention in the past year, particularly as Kaiser Health News and other news organizations have undertaken investigations into patients’ most outrageous medical bills.
Attendees said each of 10 invited guests – among them patients as well as doctors with their own stories of unexpected bills – was given an opportunity to talk, though Mr. Trump did not stay to hear all of their stories during the roughly hour-long gathering.
The group included Paul Davis, a retired doctor from Findlay, Ohio, whose family’s experience with a $17,850 bill for a simple urine test was detailed in a KHN-NPR “Bill of the Month” feature last year.
Mr. Davis’ daughter, Elizabeth Moreno, was a college student in Texas when she had spinal surgery to remedy debilitating back pain. After the surgery, she was asked to provide a urine sample and later received a bill from an out-of-network lab in Houston that tested it. Experts said such tests rarely cost more than $200, not nearly what the lab charged Ms. Moreno and her insurance company. But fearing damage to his daughter’s credit, Mr. Davis paid the lab $5,000 and filed a complaint with the Texas attorney general’s office, alleging “price gouging of staggering proportions.”
Mr. Davis said White House officials made it clear that price transparency is a “high priority” for Trump, and while they did not see eye to eye on every subject, he said he was struck by their sincerity.
“These people seemed earnest in wanting to do something constructive to fix this,” Mr. Davis said.
Dr. Martin Makary, a surgeon and health policy expert at Johns Hopkins University who has written about transparency in health care and attended the meeting, said it was a good opportunity for the White House to hear firsthand about a serious and widespread issue.
“This is how most of America lives, and [Americans are] getting hammered,” he said.
Mr. Trump has often railed against high prescription drug prices but has said less about other problems with the nation’s health care system. In October, shortly before the midterm elections, he unveiled a proposal to tie the price Medicare pays for some drugs to the prices paid for the same drugs overseas, for example.
Mr. Trump, Mr. Azar, and Mr. Acosta said efforts to control costs in health care were yielding positive results, discussing in particular the expansion of association health plans and the new requirement that hospitals post their list prices online. The president also took credit for the recent increase in generic drug approvals, which he said would help lower drug prices.
Discussing the partial government shutdown, Mr. Trump said Americans “want to see what we’re doing, like today we lowered prescription drug prices, first time in 50 years,” according to a White House pool report.
Mr. Trump appeared to be referring to a recent claim by the White House Council of Economic Advisers that prescription drug prices fell last year.
However, as STAT pointed out in a recent fact check, the report from which that claim was gleaned said “growth in relative drug prices has slowed since January 2017,” not that there was an overall decrease in prices.
Annual increases in overall drug spending have leveled off as pharmaceutical companies have released fewer blockbuster drugs; patents have expired on brand-name drugs; and the waning effect of a spike driven by the release of astronomically expensive drugs to treat hepatitis C. Drugmakers are also wary of increasing their prices in the midst of growing political pressure.
Since Democrats seized control of the House of Representatives this month, party leaders have rushed to announce investigations and schedule hearings dealing with health care, focusing in particular on drug costs and protections for those with preexisting conditions.
Recently, the House Oversight Committee announced a “sweeping” investigation into drug prices, pointing to an AARP report saying the vast majority of brand-name drugs had more than doubled in price between 2005 and 2017.
KHN correspondents Shefali Luthra and Jay Hancock contributed to this report. Kaiser Health News is a nonprofit national health policy news service. It is an editorially independent program of the Henry J. Kaiser Family Foundation that is not affiliated with Kaiser Permanente.
President Trump on Jan. 23 instructed administration officials to investigate how to prevent surprise medical bills, broadening his focus on drug prices to include other issues of price transparency in health care.

several attendees said.
“The pricing is hurting patients, and we’ve stopped a lot of it, but we’re going to stop all of it,” Mr. Trump said during a roundtable discussion when reporters were briefly allowed into the otherwise closed-door meeting.
David Silverstein, the founder of a Colorado-based nonprofit called Broken Healthcare who attended, said Mr. Trump struck an aggressive tone, calling for a solution with “the biggest teeth you can find.”
“Reading the tea leaves, I think there’s big change coming,” Mr. Silverstein said.
Surprise billing, or the practice of charging patients for care that is more expensive than anticipated or not covered by their insurance, has received a flood of attention in the past year, particularly as Kaiser Health News and other news organizations have undertaken investigations into patients’ most outrageous medical bills.
Attendees said each of 10 invited guests – among them patients as well as doctors with their own stories of unexpected bills – was given an opportunity to talk, though Mr. Trump did not stay to hear all of their stories during the roughly hour-long gathering.
The group included Paul Davis, a retired doctor from Findlay, Ohio, whose family’s experience with a $17,850 bill for a simple urine test was detailed in a KHN-NPR “Bill of the Month” feature last year.
Mr. Davis’ daughter, Elizabeth Moreno, was a college student in Texas when she had spinal surgery to remedy debilitating back pain. After the surgery, she was asked to provide a urine sample and later received a bill from an out-of-network lab in Houston that tested it. Experts said such tests rarely cost more than $200, not nearly what the lab charged Ms. Moreno and her insurance company. But fearing damage to his daughter’s credit, Mr. Davis paid the lab $5,000 and filed a complaint with the Texas attorney general’s office, alleging “price gouging of staggering proportions.”
Mr. Davis said White House officials made it clear that price transparency is a “high priority” for Trump, and while they did not see eye to eye on every subject, he said he was struck by their sincerity.
“These people seemed earnest in wanting to do something constructive to fix this,” Mr. Davis said.
Dr. Martin Makary, a surgeon and health policy expert at Johns Hopkins University who has written about transparency in health care and attended the meeting, said it was a good opportunity for the White House to hear firsthand about a serious and widespread issue.
“This is how most of America lives, and [Americans are] getting hammered,” he said.
Mr. Trump has often railed against high prescription drug prices but has said less about other problems with the nation’s health care system. In October, shortly before the midterm elections, he unveiled a proposal to tie the price Medicare pays for some drugs to the prices paid for the same drugs overseas, for example.
Mr. Trump, Mr. Azar, and Mr. Acosta said efforts to control costs in health care were yielding positive results, discussing in particular the expansion of association health plans and the new requirement that hospitals post their list prices online. The president also took credit for the recent increase in generic drug approvals, which he said would help lower drug prices.
Discussing the partial government shutdown, Mr. Trump said Americans “want to see what we’re doing, like today we lowered prescription drug prices, first time in 50 years,” according to a White House pool report.
Mr. Trump appeared to be referring to a recent claim by the White House Council of Economic Advisers that prescription drug prices fell last year.
However, as STAT pointed out in a recent fact check, the report from which that claim was gleaned said “growth in relative drug prices has slowed since January 2017,” not that there was an overall decrease in prices.
Annual increases in overall drug spending have leveled off as pharmaceutical companies have released fewer blockbuster drugs; patents have expired on brand-name drugs; and the waning effect of a spike driven by the release of astronomically expensive drugs to treat hepatitis C. Drugmakers are also wary of increasing their prices in the midst of growing political pressure.
Since Democrats seized control of the House of Representatives this month, party leaders have rushed to announce investigations and schedule hearings dealing with health care, focusing in particular on drug costs and protections for those with preexisting conditions.
Recently, the House Oversight Committee announced a “sweeping” investigation into drug prices, pointing to an AARP report saying the vast majority of brand-name drugs had more than doubled in price between 2005 and 2017.
KHN correspondents Shefali Luthra and Jay Hancock contributed to this report. Kaiser Health News is a nonprofit national health policy news service. It is an editorially independent program of the Henry J. Kaiser Family Foundation that is not affiliated with Kaiser Permanente.