User login
Pembrolizumab prolonged PFS vs. brentuximab vedotin in r/r Hodgkin lymphoma
Pembrolizumab treatment significantly improved progression-free survival versus brentuximab vedotin in a randomized, phase 3 trial including patients with relapsed or refractory classical Hodgkin lymphoma, an investigator has reported.
Median progression-free survival (PFS) was 13.2 versus 8.3 months in favor of pembrolizumab, according to the report on the KEYNOTE-204 trial, which included patients with classical Hodgkin lymphoma who either had relapsed after autologous stem cell transplant (SCT) or were ineligible for autologous SCT.
of Princess Margaret Cancer Centre in Toronto.
“This PFS benefit extended to key subgroups, including those ineligible for autologous transplant, patients with primary refractory disease, and patients who were brentuximab-vedotin naive,” Dr. Kuruvilla added in his presentation, which was part of the American Society of Clinical Oncology virtual scientific program.
Pneumonitis was more frequent in the pembrolizumab arm, but “appeared in general to be quite well managed” among patients who experienced this adverse event, according to Dr. Kuruvilla, who said that treatment with the programmed death–1 inhibitor should be considered “the preferred treatment option and the new standard of care” for patients with relapsed/refractory classic Hodgkin lymphoma who have relapsed after autologous SCT or are ineligible for it.
Although the pneumonitis findings are important to keep in mind, results of KEYNOTE-204 are indeed “practice defining” and immediately impactful, said Mark J. Roschewski, MD, clinical investigator in the lymphoid malignancies branch at the Center for Cancer Research, part of the National Cancer Institute, Bethesda, Md.
“I would select pembrolizumab over brentuximab for this patient population, particularly those that are refractory to chemotherapy,” he said in a commentary on the study also included in the virtual ASCO proceedings.
“There may be specific patient populations that I’d reconsider, such as those that might be at high risk for lung toxicity,” he added. “They may not be suitable for this, but it’s something to at least to be aware of.”
Although the antibody-drug conjugate brentuximab vedotin has been considered the standard of care for patients with relapse after autologous SCT, there has historically been no standard of care for patients who are ineligible for transplant because of chemorefractory disease, advanced age, or comorbidities, Dr. Kuruvilla said in his presentation.
In the KEYNOTE-204 study, 304 patients with relapsed/refractory classic Hodgkin lymphoma were randomized to receive either pembrolizumab 200 mg or brentuximab at 1.8 mg/kg intravenously every 3 weeks for up to 35 cycles.
The median age of patients was 36 years in the pembrolizumab arm and 35 years in the brentuximab vedotin arm, according to the report. Approximately 37% of the patients had previously undergone autologous SCT. About 40% had been refractory to frontline therapy, while 28% relapsed within 12 months of therapy and 32% relapsed later than 12 months.
Median PFS by blinded independent central review was 13.2 versus 8.3 months in the pembrolizumab and brentuximab arms, respectively (hazard ratio, 0.65; 95% confidence interval, 0.48-0.88; P = .00271), Dr. Kuruvilla reported.
The benefit extended to “key subgroups” in the trial, he added, including those who were ineligible for autologous SCT, those with primary refractory disease, and those who were naive to brentuximab vedotin, with HRs of 0.61, 0.52, and 0.67, respectively.
Pembrolizumab was also associated with more durable responses versus brentuximab vedotin, according to the investigator.
The overall response rate was 65.6% and 54.2%, respectively, for pembrolizumab and brentuximab, although this difference of approximately 11 percentage points did not meet criteria for statistical significance, he said. Duration of response was 20.7 months or pembrolizumab and 13.8 months for brentuximab.
The rate of serious treatment-related adverse events was similar between groups, according to Dr. Kuruvilla, who reported grade 3-5 events occurring in 19.6% and 25.0% of the pembrolizumab and brentuximab arms. Serious treatment-related adverse events were numerically more frequent in the pembrolizumab arm (16.2% vs. 10.5%) and there was one treatment-related death caused by pneumonia, seen in the pembrolizumab arm.
Pneumonitis occurred in 2.6% of the brentuximab-treated patients and in 10.8% of pembrolizumab-treated patients, of which half of cases were grade 3-4, according to the report.
In the pembrolizumab arm, pneumonitis was felt to be drug-related in 15 of 16 cases, according to Dr. Kuruvilla, who added that 15 of 16 patients required corticosteroid therapy. “This has led to the resolution of the pneumonitis in 12 of 16 patients, with ongoing resolution in one further patient.”
Research funding for KEYNOTE-204 came from Merck Sharp & Dohme. Dr. Kuruvilla provided disclosures related to Merck and a variety of other pharmaceutical companies. Dr. Roschewski said he had no relationships to disclose.
SOURCE: Kuruvilla J et al. ASCO 2020, Abstract 8005.
Pembrolizumab treatment significantly improved progression-free survival versus brentuximab vedotin in a randomized, phase 3 trial including patients with relapsed or refractory classical Hodgkin lymphoma, an investigator has reported.
Median progression-free survival (PFS) was 13.2 versus 8.3 months in favor of pembrolizumab, according to the report on the KEYNOTE-204 trial, which included patients with classical Hodgkin lymphoma who either had relapsed after autologous stem cell transplant (SCT) or were ineligible for autologous SCT.
of Princess Margaret Cancer Centre in Toronto.
“This PFS benefit extended to key subgroups, including those ineligible for autologous transplant, patients with primary refractory disease, and patients who were brentuximab-vedotin naive,” Dr. Kuruvilla added in his presentation, which was part of the American Society of Clinical Oncology virtual scientific program.
Pneumonitis was more frequent in the pembrolizumab arm, but “appeared in general to be quite well managed” among patients who experienced this adverse event, according to Dr. Kuruvilla, who said that treatment with the programmed death–1 inhibitor should be considered “the preferred treatment option and the new standard of care” for patients with relapsed/refractory classic Hodgkin lymphoma who have relapsed after autologous SCT or are ineligible for it.
Although the pneumonitis findings are important to keep in mind, results of KEYNOTE-204 are indeed “practice defining” and immediately impactful, said Mark J. Roschewski, MD, clinical investigator in the lymphoid malignancies branch at the Center for Cancer Research, part of the National Cancer Institute, Bethesda, Md.
“I would select pembrolizumab over brentuximab for this patient population, particularly those that are refractory to chemotherapy,” he said in a commentary on the study also included in the virtual ASCO proceedings.
“There may be specific patient populations that I’d reconsider, such as those that might be at high risk for lung toxicity,” he added. “They may not be suitable for this, but it’s something to at least to be aware of.”
Although the antibody-drug conjugate brentuximab vedotin has been considered the standard of care for patients with relapse after autologous SCT, there has historically been no standard of care for patients who are ineligible for transplant because of chemorefractory disease, advanced age, or comorbidities, Dr. Kuruvilla said in his presentation.
In the KEYNOTE-204 study, 304 patients with relapsed/refractory classic Hodgkin lymphoma were randomized to receive either pembrolizumab 200 mg or brentuximab at 1.8 mg/kg intravenously every 3 weeks for up to 35 cycles.
The median age of patients was 36 years in the pembrolizumab arm and 35 years in the brentuximab vedotin arm, according to the report. Approximately 37% of the patients had previously undergone autologous SCT. About 40% had been refractory to frontline therapy, while 28% relapsed within 12 months of therapy and 32% relapsed later than 12 months.
Median PFS by blinded independent central review was 13.2 versus 8.3 months in the pembrolizumab and brentuximab arms, respectively (hazard ratio, 0.65; 95% confidence interval, 0.48-0.88; P = .00271), Dr. Kuruvilla reported.
The benefit extended to “key subgroups” in the trial, he added, including those who were ineligible for autologous SCT, those with primary refractory disease, and those who were naive to brentuximab vedotin, with HRs of 0.61, 0.52, and 0.67, respectively.
Pembrolizumab was also associated with more durable responses versus brentuximab vedotin, according to the investigator.
The overall response rate was 65.6% and 54.2%, respectively, for pembrolizumab and brentuximab, although this difference of approximately 11 percentage points did not meet criteria for statistical significance, he said. Duration of response was 20.7 months or pembrolizumab and 13.8 months for brentuximab.
The rate of serious treatment-related adverse events was similar between groups, according to Dr. Kuruvilla, who reported grade 3-5 events occurring in 19.6% and 25.0% of the pembrolizumab and brentuximab arms. Serious treatment-related adverse events were numerically more frequent in the pembrolizumab arm (16.2% vs. 10.5%) and there was one treatment-related death caused by pneumonia, seen in the pembrolizumab arm.
Pneumonitis occurred in 2.6% of the brentuximab-treated patients and in 10.8% of pembrolizumab-treated patients, of which half of cases were grade 3-4, according to the report.
In the pembrolizumab arm, pneumonitis was felt to be drug-related in 15 of 16 cases, according to Dr. Kuruvilla, who added that 15 of 16 patients required corticosteroid therapy. “This has led to the resolution of the pneumonitis in 12 of 16 patients, with ongoing resolution in one further patient.”
Research funding for KEYNOTE-204 came from Merck Sharp & Dohme. Dr. Kuruvilla provided disclosures related to Merck and a variety of other pharmaceutical companies. Dr. Roschewski said he had no relationships to disclose.
SOURCE: Kuruvilla J et al. ASCO 2020, Abstract 8005.
Pembrolizumab treatment significantly improved progression-free survival versus brentuximab vedotin in a randomized, phase 3 trial including patients with relapsed or refractory classical Hodgkin lymphoma, an investigator has reported.
Median progression-free survival (PFS) was 13.2 versus 8.3 months in favor of pembrolizumab, according to the report on the KEYNOTE-204 trial, which included patients with classical Hodgkin lymphoma who either had relapsed after autologous stem cell transplant (SCT) or were ineligible for autologous SCT.
of Princess Margaret Cancer Centre in Toronto.
“This PFS benefit extended to key subgroups, including those ineligible for autologous transplant, patients with primary refractory disease, and patients who were brentuximab-vedotin naive,” Dr. Kuruvilla added in his presentation, which was part of the American Society of Clinical Oncology virtual scientific program.
Pneumonitis was more frequent in the pembrolizumab arm, but “appeared in general to be quite well managed” among patients who experienced this adverse event, according to Dr. Kuruvilla, who said that treatment with the programmed death–1 inhibitor should be considered “the preferred treatment option and the new standard of care” for patients with relapsed/refractory classic Hodgkin lymphoma who have relapsed after autologous SCT or are ineligible for it.
Although the pneumonitis findings are important to keep in mind, results of KEYNOTE-204 are indeed “practice defining” and immediately impactful, said Mark J. Roschewski, MD, clinical investigator in the lymphoid malignancies branch at the Center for Cancer Research, part of the National Cancer Institute, Bethesda, Md.
“I would select pembrolizumab over brentuximab for this patient population, particularly those that are refractory to chemotherapy,” he said in a commentary on the study also included in the virtual ASCO proceedings.
“There may be specific patient populations that I’d reconsider, such as those that might be at high risk for lung toxicity,” he added. “They may not be suitable for this, but it’s something to at least to be aware of.”
Although the antibody-drug conjugate brentuximab vedotin has been considered the standard of care for patients with relapse after autologous SCT, there has historically been no standard of care for patients who are ineligible for transplant because of chemorefractory disease, advanced age, or comorbidities, Dr. Kuruvilla said in his presentation.
In the KEYNOTE-204 study, 304 patients with relapsed/refractory classic Hodgkin lymphoma were randomized to receive either pembrolizumab 200 mg or brentuximab at 1.8 mg/kg intravenously every 3 weeks for up to 35 cycles.
The median age of patients was 36 years in the pembrolizumab arm and 35 years in the brentuximab vedotin arm, according to the report. Approximately 37% of the patients had previously undergone autologous SCT. About 40% had been refractory to frontline therapy, while 28% relapsed within 12 months of therapy and 32% relapsed later than 12 months.
Median PFS by blinded independent central review was 13.2 versus 8.3 months in the pembrolizumab and brentuximab arms, respectively (hazard ratio, 0.65; 95% confidence interval, 0.48-0.88; P = .00271), Dr. Kuruvilla reported.
The benefit extended to “key subgroups” in the trial, he added, including those who were ineligible for autologous SCT, those with primary refractory disease, and those who were naive to brentuximab vedotin, with HRs of 0.61, 0.52, and 0.67, respectively.
Pembrolizumab was also associated with more durable responses versus brentuximab vedotin, according to the investigator.
The overall response rate was 65.6% and 54.2%, respectively, for pembrolizumab and brentuximab, although this difference of approximately 11 percentage points did not meet criteria for statistical significance, he said. Duration of response was 20.7 months or pembrolizumab and 13.8 months for brentuximab.
The rate of serious treatment-related adverse events was similar between groups, according to Dr. Kuruvilla, who reported grade 3-5 events occurring in 19.6% and 25.0% of the pembrolizumab and brentuximab arms. Serious treatment-related adverse events were numerically more frequent in the pembrolizumab arm (16.2% vs. 10.5%) and there was one treatment-related death caused by pneumonia, seen in the pembrolizumab arm.
Pneumonitis occurred in 2.6% of the brentuximab-treated patients and in 10.8% of pembrolizumab-treated patients, of which half of cases were grade 3-4, according to the report.
In the pembrolizumab arm, pneumonitis was felt to be drug-related in 15 of 16 cases, according to Dr. Kuruvilla, who added that 15 of 16 patients required corticosteroid therapy. “This has led to the resolution of the pneumonitis in 12 of 16 patients, with ongoing resolution in one further patient.”
Research funding for KEYNOTE-204 came from Merck Sharp & Dohme. Dr. Kuruvilla provided disclosures related to Merck and a variety of other pharmaceutical companies. Dr. Roschewski said he had no relationships to disclose.
SOURCE: Kuruvilla J et al. ASCO 2020, Abstract 8005.
FROM ASCO 2020
Convalescent plasma: ‘Flavor of the month’ or valid COVID-19 treatment?
On March 31, soon after the Food and Drug Administration authorized emergency use of antibody-packed plasma from recovered patients with COVID-19, Marisa Leuzzi became the first donor at an American Red Cross center. She hoped it could help her aunt, Renee Bannister, who was failing after 3 weeks on a ventilator at Virtua Hospital in Voorhees, N.J.
It may have worked; 11 days after receiving the plasma, Ms. Bannister was weaned off the ventilator and she is now awake and speaking, said Red Cross spokesperson Stephanie Rendon.
This kind of anecdote is fueling demand for the therapy, which can be provided through an expanded access program led by the Mayo Clinic, backed by the FDA, and the plasma paid for by the U.S. Department of Health & Human Services. But while this program is collecting safety and outcomes data, it’s not a randomized, controlled trial.
Others, however, are pursuing that data.
“One of the things I don’t want this to be is the flavor of the month,” Shmuel Shoham, MD, associate professor of medicine at Johns Hopkins University, said in an interview.
Dr. Shoham, principal investigator for a study evaluating convalescent plasma to prevent the infection in high-risk individuals, said some clinicians, desperate for any treatment, have tried potential therapies such as hydroxychloroquine and remdesivir without evidence of safety or efficacy in COVID-19.
The National Institutes of Health recently said something similar for convalescent plasma, that “there are insufficient clinical data to recommend either for or against” its use for COVID-19.
But plasma has promise, according to a Johns Hopkins School of Medicine’s Bloomberg Distinguished Professor, Arturo Casadevall, MD, PhD, in Baltimore, and Liise-anne Pirofski, MD, a professor at Albert Einstein College of Medicine, New York. They lay out the case for convalescent plasma in an article published online March 13 in the Journal of Clinical Investigation. Passive antibody therapy, they wrote, has been used to stem polio, measles, mumps, and influenza, and more recently has shown some success against SARS-CoV-1 and Middle East respiratory syndrome (MERS).
“The special attraction of this modality of treatment is that, unlike vaccines or newly developed drugs, it could, in principle, be made available very rapidly,” said researchers with the National COVID-19 Convalescent Plasma Project, which includes physicians and scientists from 57 institutions in 46 states. But where principle veers from reality is in availability of the plasma itself, and donors are in short supply.
Aiming to prevent infection
So far, the FDA has approved 12 plasma trials – including Dr. Shoham’s – and the NIH’s clinicaltrials.gov lists more than two dozen convalescent plasma studies in the United States and elsewhere.
Most are single-arm trials to determine if one infusion can decrease the need for intubation or help those on a ventilator improve. Two others, one at Johns Hopkins and one at Stanford (Calif.) Hospital are investigating whether convalescent plasma might be used before severe disease sets in.
“A general principle of passive antibody therapy is that it is more effective when used for prophylaxis than for treatment of disease,” Dr. Casadevall and Dr. Pirofski wrote.
Stanford’s randomized, double-blind study will evaluate regular versus convalescent plasma in ED patients who are not sick enough to require hospitalization.
The Johns Hopkins trial, which aims to protect against infection in the first place, will begin at Johns Hopkins, Baltimore, and at Hopkins-affiliated hospitals throughout Maryland, Dr. Shoham said. He hopes it will expand nationwide eventually, and said that they expect to enroll the first patients soon.
To start, the prevention study will enroll only 150 patients, each of whom must have had close contact with someone who has COVID-19 within the previous 120 hours and be asymptomatic. The number of subjects is small, compared with the trial size of other potential therapies, and an issue, Shoham said, “that keeps me up at night.” But finding thousands of enrollees for plasma studies is hard, in part because it’s so difficult to recruit donors.
Participants will receive normal plasma (which will act as a placebo) or convalescent plasma.
The primary endpoint is cumulative incidence of COVID-19, defined as symptoms and a polymerase chain reaction–positive test; participants will be tracked for 90 days. Hospitals and health care workers could then decide if they want to use the therapy, he said.
The study will not answer whether participants will continue to have antibodies beyond the 90 days. Convalescent plasma is given as a rapid response to an emergent pathogen – a short-term boost of immunity rather than a long-term therapeutic.
What can we learn from expanded access?
Meanwhile, some 2,200 hospitals are participating in the expanded access program being led by the Mayo Clinic nationwide; more than 9,000 patients had received infusions at press time.
One participant is Northwell Health, a 23-hospital system that sprawls across the U.S. COVID epicenter: four of the five boroughs of New York City and Long Island.
Convalescent plasma is an in-demand therapy, said Christina Brennan, MD, vice president of clinical research at Northwell. “We get patients, family members, they say my family member is at X hospital – if it’s not being offered there, can you have them transferred?” she said in an interview.
When Northwell – through the New York Blood Bank – opened up donor registration, 800 people signed up in the first 24 hours, Dr. Brennan said. As of mid-May, 527 patients had received a transfusion.
Who’s the best donor and when should donation occur?
The Red Cross, hospitals, and independent blood banks are all soliciting donors, who can sign up at the Red Cross website. The FDA recommends that donors have a history of COVID-19 as confirmed by molecular or antibody testing, be symptom free for 14 days, have a negative follow-up molecular test, and be virus free at the time of collection. The FDA also suggests measuring a donor’s SARS-CoV-2 neutralizing antibody titers, if available, with a recommendation of at least 1:160.
But questions remain, such as whether there is a theoretical risk for antibody-dependent enhancement (ADE) of infection with SARS-CoV-2. “Antibodies to one type of coronavirus could enhance infection to another viral strain,” of coronavirus, Dr. Casadevall wrote. ADE has been observed in both severe acute respiratory syndrome (SARS) and MERS.
The other risk is that donors may still be shedding active virus. While the FDA suggests that donors are unlikely to still be infectious 14 days after infection, that is as of yet unproven. Both COVID-19 diagnostics and antibody tests have high rates of false negatives, which raises the specter that infection could be spread via the plasma donation.
Daniele Focosi, MD, PhD, from Pisa (Italy) University Hospital and colleagues raise that concern in a preprint review on convalescent plasma in COVID-19. “Although the recipient is already infected, theoretically transmission of more infectious particles could worsen clinical conditions,” they wrote, noting that “such a concern can be somewhat reduced by treatment with modern pathogen inactivation techniques.”
No evidence exists that SARS-CoV-2 can be transmitted through blood, but “we don’t know for sure,” Dr. Shoham said in an interview. A reassuring point: Even those with severe infection do not have viral RNA in their blood, he said, adding, “We don’t think there’s going to be viral transmission of this particular virus with transfusion.”
For another highly infectious pathogen, the Ebola virus, the World Health Organization recommended in 2014 that potential plasma donors wait at least 28 days after infection.
It’s also not known how long SARS-CoV-2 antibodies persist in the blood; longer viability could mean a longer donation window. Dr. Focosi noted that a previous Chinese study had shown that SARS-specific antibodies in people infected with the first SARS virus, SARS-CoV-1, persisted for 2 years.
Dr. Casadevall and Dr. Pirofski have disclosed no relevant financial relationships. Shoham has disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
On March 31, soon after the Food and Drug Administration authorized emergency use of antibody-packed plasma from recovered patients with COVID-19, Marisa Leuzzi became the first donor at an American Red Cross center. She hoped it could help her aunt, Renee Bannister, who was failing after 3 weeks on a ventilator at Virtua Hospital in Voorhees, N.J.
It may have worked; 11 days after receiving the plasma, Ms. Bannister was weaned off the ventilator and she is now awake and speaking, said Red Cross spokesperson Stephanie Rendon.
This kind of anecdote is fueling demand for the therapy, which can be provided through an expanded access program led by the Mayo Clinic, backed by the FDA, and the plasma paid for by the U.S. Department of Health & Human Services. But while this program is collecting safety and outcomes data, it’s not a randomized, controlled trial.
Others, however, are pursuing that data.
“One of the things I don’t want this to be is the flavor of the month,” Shmuel Shoham, MD, associate professor of medicine at Johns Hopkins University, said in an interview.
Dr. Shoham, principal investigator for a study evaluating convalescent plasma to prevent the infection in high-risk individuals, said some clinicians, desperate for any treatment, have tried potential therapies such as hydroxychloroquine and remdesivir without evidence of safety or efficacy in COVID-19.
The National Institutes of Health recently said something similar for convalescent plasma, that “there are insufficient clinical data to recommend either for or against” its use for COVID-19.
But plasma has promise, according to a Johns Hopkins School of Medicine’s Bloomberg Distinguished Professor, Arturo Casadevall, MD, PhD, in Baltimore, and Liise-anne Pirofski, MD, a professor at Albert Einstein College of Medicine, New York. They lay out the case for convalescent plasma in an article published online March 13 in the Journal of Clinical Investigation. Passive antibody therapy, they wrote, has been used to stem polio, measles, mumps, and influenza, and more recently has shown some success against SARS-CoV-1 and Middle East respiratory syndrome (MERS).
“The special attraction of this modality of treatment is that, unlike vaccines or newly developed drugs, it could, in principle, be made available very rapidly,” said researchers with the National COVID-19 Convalescent Plasma Project, which includes physicians and scientists from 57 institutions in 46 states. But where principle veers from reality is in availability of the plasma itself, and donors are in short supply.
Aiming to prevent infection
So far, the FDA has approved 12 plasma trials – including Dr. Shoham’s – and the NIH’s clinicaltrials.gov lists more than two dozen convalescent plasma studies in the United States and elsewhere.
Most are single-arm trials to determine if one infusion can decrease the need for intubation or help those on a ventilator improve. Two others, one at Johns Hopkins and one at Stanford (Calif.) Hospital are investigating whether convalescent plasma might be used before severe disease sets in.
“A general principle of passive antibody therapy is that it is more effective when used for prophylaxis than for treatment of disease,” Dr. Casadevall and Dr. Pirofski wrote.
Stanford’s randomized, double-blind study will evaluate regular versus convalescent plasma in ED patients who are not sick enough to require hospitalization.
The Johns Hopkins trial, which aims to protect against infection in the first place, will begin at Johns Hopkins, Baltimore, and at Hopkins-affiliated hospitals throughout Maryland, Dr. Shoham said. He hopes it will expand nationwide eventually, and said that they expect to enroll the first patients soon.
To start, the prevention study will enroll only 150 patients, each of whom must have had close contact with someone who has COVID-19 within the previous 120 hours and be asymptomatic. The number of subjects is small, compared with the trial size of other potential therapies, and an issue, Shoham said, “that keeps me up at night.” But finding thousands of enrollees for plasma studies is hard, in part because it’s so difficult to recruit donors.
Participants will receive normal plasma (which will act as a placebo) or convalescent plasma.
The primary endpoint is cumulative incidence of COVID-19, defined as symptoms and a polymerase chain reaction–positive test; participants will be tracked for 90 days. Hospitals and health care workers could then decide if they want to use the therapy, he said.
The study will not answer whether participants will continue to have antibodies beyond the 90 days. Convalescent plasma is given as a rapid response to an emergent pathogen – a short-term boost of immunity rather than a long-term therapeutic.
What can we learn from expanded access?
Meanwhile, some 2,200 hospitals are participating in the expanded access program being led by the Mayo Clinic nationwide; more than 9,000 patients had received infusions at press time.
One participant is Northwell Health, a 23-hospital system that sprawls across the U.S. COVID epicenter: four of the five boroughs of New York City and Long Island.
Convalescent plasma is an in-demand therapy, said Christina Brennan, MD, vice president of clinical research at Northwell. “We get patients, family members, they say my family member is at X hospital – if it’s not being offered there, can you have them transferred?” she said in an interview.
When Northwell – through the New York Blood Bank – opened up donor registration, 800 people signed up in the first 24 hours, Dr. Brennan said. As of mid-May, 527 patients had received a transfusion.
Who’s the best donor and when should donation occur?
The Red Cross, hospitals, and independent blood banks are all soliciting donors, who can sign up at the Red Cross website. The FDA recommends that donors have a history of COVID-19 as confirmed by molecular or antibody testing, be symptom free for 14 days, have a negative follow-up molecular test, and be virus free at the time of collection. The FDA also suggests measuring a donor’s SARS-CoV-2 neutralizing antibody titers, if available, with a recommendation of at least 1:160.
But questions remain, such as whether there is a theoretical risk for antibody-dependent enhancement (ADE) of infection with SARS-CoV-2. “Antibodies to one type of coronavirus could enhance infection to another viral strain,” of coronavirus, Dr. Casadevall wrote. ADE has been observed in both severe acute respiratory syndrome (SARS) and MERS.
The other risk is that donors may still be shedding active virus. While the FDA suggests that donors are unlikely to still be infectious 14 days after infection, that is as of yet unproven. Both COVID-19 diagnostics and antibody tests have high rates of false negatives, which raises the specter that infection could be spread via the plasma donation.
Daniele Focosi, MD, PhD, from Pisa (Italy) University Hospital and colleagues raise that concern in a preprint review on convalescent plasma in COVID-19. “Although the recipient is already infected, theoretically transmission of more infectious particles could worsen clinical conditions,” they wrote, noting that “such a concern can be somewhat reduced by treatment with modern pathogen inactivation techniques.”
No evidence exists that SARS-CoV-2 can be transmitted through blood, but “we don’t know for sure,” Dr. Shoham said in an interview. A reassuring point: Even those with severe infection do not have viral RNA in their blood, he said, adding, “We don’t think there’s going to be viral transmission of this particular virus with transfusion.”
For another highly infectious pathogen, the Ebola virus, the World Health Organization recommended in 2014 that potential plasma donors wait at least 28 days after infection.
It’s also not known how long SARS-CoV-2 antibodies persist in the blood; longer viability could mean a longer donation window. Dr. Focosi noted that a previous Chinese study had shown that SARS-specific antibodies in people infected with the first SARS virus, SARS-CoV-1, persisted for 2 years.
Dr. Casadevall and Dr. Pirofski have disclosed no relevant financial relationships. Shoham has disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
On March 31, soon after the Food and Drug Administration authorized emergency use of antibody-packed plasma from recovered patients with COVID-19, Marisa Leuzzi became the first donor at an American Red Cross center. She hoped it could help her aunt, Renee Bannister, who was failing after 3 weeks on a ventilator at Virtua Hospital in Voorhees, N.J.
It may have worked; 11 days after receiving the plasma, Ms. Bannister was weaned off the ventilator and she is now awake and speaking, said Red Cross spokesperson Stephanie Rendon.
This kind of anecdote is fueling demand for the therapy, which can be provided through an expanded access program led by the Mayo Clinic, backed by the FDA, and the plasma paid for by the U.S. Department of Health & Human Services. But while this program is collecting safety and outcomes data, it’s not a randomized, controlled trial.
Others, however, are pursuing that data.
“One of the things I don’t want this to be is the flavor of the month,” Shmuel Shoham, MD, associate professor of medicine at Johns Hopkins University, said in an interview.
Dr. Shoham, principal investigator for a study evaluating convalescent plasma to prevent the infection in high-risk individuals, said some clinicians, desperate for any treatment, have tried potential therapies such as hydroxychloroquine and remdesivir without evidence of safety or efficacy in COVID-19.
The National Institutes of Health recently said something similar for convalescent plasma, that “there are insufficient clinical data to recommend either for or against” its use for COVID-19.
But plasma has promise, according to a Johns Hopkins School of Medicine’s Bloomberg Distinguished Professor, Arturo Casadevall, MD, PhD, in Baltimore, and Liise-anne Pirofski, MD, a professor at Albert Einstein College of Medicine, New York. They lay out the case for convalescent plasma in an article published online March 13 in the Journal of Clinical Investigation. Passive antibody therapy, they wrote, has been used to stem polio, measles, mumps, and influenza, and more recently has shown some success against SARS-CoV-1 and Middle East respiratory syndrome (MERS).
“The special attraction of this modality of treatment is that, unlike vaccines or newly developed drugs, it could, in principle, be made available very rapidly,” said researchers with the National COVID-19 Convalescent Plasma Project, which includes physicians and scientists from 57 institutions in 46 states. But where principle veers from reality is in availability of the plasma itself, and donors are in short supply.
Aiming to prevent infection
So far, the FDA has approved 12 plasma trials – including Dr. Shoham’s – and the NIH’s clinicaltrials.gov lists more than two dozen convalescent plasma studies in the United States and elsewhere.
Most are single-arm trials to determine if one infusion can decrease the need for intubation or help those on a ventilator improve. Two others, one at Johns Hopkins and one at Stanford (Calif.) Hospital are investigating whether convalescent plasma might be used before severe disease sets in.
“A general principle of passive antibody therapy is that it is more effective when used for prophylaxis than for treatment of disease,” Dr. Casadevall and Dr. Pirofski wrote.
Stanford’s randomized, double-blind study will evaluate regular versus convalescent plasma in ED patients who are not sick enough to require hospitalization.
The Johns Hopkins trial, which aims to protect against infection in the first place, will begin at Johns Hopkins, Baltimore, and at Hopkins-affiliated hospitals throughout Maryland, Dr. Shoham said. He hopes it will expand nationwide eventually, and said that they expect to enroll the first patients soon.
To start, the prevention study will enroll only 150 patients, each of whom must have had close contact with someone who has COVID-19 within the previous 120 hours and be asymptomatic. The number of subjects is small, compared with the trial size of other potential therapies, and an issue, Shoham said, “that keeps me up at night.” But finding thousands of enrollees for plasma studies is hard, in part because it’s so difficult to recruit donors.
Participants will receive normal plasma (which will act as a placebo) or convalescent plasma.
The primary endpoint is cumulative incidence of COVID-19, defined as symptoms and a polymerase chain reaction–positive test; participants will be tracked for 90 days. Hospitals and health care workers could then decide if they want to use the therapy, he said.
The study will not answer whether participants will continue to have antibodies beyond the 90 days. Convalescent plasma is given as a rapid response to an emergent pathogen – a short-term boost of immunity rather than a long-term therapeutic.
What can we learn from expanded access?
Meanwhile, some 2,200 hospitals are participating in the expanded access program being led by the Mayo Clinic nationwide; more than 9,000 patients had received infusions at press time.
One participant is Northwell Health, a 23-hospital system that sprawls across the U.S. COVID epicenter: four of the five boroughs of New York City and Long Island.
Convalescent plasma is an in-demand therapy, said Christina Brennan, MD, vice president of clinical research at Northwell. “We get patients, family members, they say my family member is at X hospital – if it’s not being offered there, can you have them transferred?” she said in an interview.
When Northwell – through the New York Blood Bank – opened up donor registration, 800 people signed up in the first 24 hours, Dr. Brennan said. As of mid-May, 527 patients had received a transfusion.
Who’s the best donor and when should donation occur?
The Red Cross, hospitals, and independent blood banks are all soliciting donors, who can sign up at the Red Cross website. The FDA recommends that donors have a history of COVID-19 as confirmed by molecular or antibody testing, be symptom free for 14 days, have a negative follow-up molecular test, and be virus free at the time of collection. The FDA also suggests measuring a donor’s SARS-CoV-2 neutralizing antibody titers, if available, with a recommendation of at least 1:160.
But questions remain, such as whether there is a theoretical risk for antibody-dependent enhancement (ADE) of infection with SARS-CoV-2. “Antibodies to one type of coronavirus could enhance infection to another viral strain,” of coronavirus, Dr. Casadevall wrote. ADE has been observed in both severe acute respiratory syndrome (SARS) and MERS.
The other risk is that donors may still be shedding active virus. While the FDA suggests that donors are unlikely to still be infectious 14 days after infection, that is as of yet unproven. Both COVID-19 diagnostics and antibody tests have high rates of false negatives, which raises the specter that infection could be spread via the plasma donation.
Daniele Focosi, MD, PhD, from Pisa (Italy) University Hospital and colleagues raise that concern in a preprint review on convalescent plasma in COVID-19. “Although the recipient is already infected, theoretically transmission of more infectious particles could worsen clinical conditions,” they wrote, noting that “such a concern can be somewhat reduced by treatment with modern pathogen inactivation techniques.”
No evidence exists that SARS-CoV-2 can be transmitted through blood, but “we don’t know for sure,” Dr. Shoham said in an interview. A reassuring point: Even those with severe infection do not have viral RNA in their blood, he said, adding, “We don’t think there’s going to be viral transmission of this particular virus with transfusion.”
For another highly infectious pathogen, the Ebola virus, the World Health Organization recommended in 2014 that potential plasma donors wait at least 28 days after infection.
It’s also not known how long SARS-CoV-2 antibodies persist in the blood; longer viability could mean a longer donation window. Dr. Focosi noted that a previous Chinese study had shown that SARS-specific antibodies in people infected with the first SARS virus, SARS-CoV-1, persisted for 2 years.
Dr. Casadevall and Dr. Pirofski have disclosed no relevant financial relationships. Shoham has disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
Patient-focused precautions, testing help blunt pandemic effects on heme-onc unit
Keeping hematologic oncology patients on their treatment regimens and caring for inpatients with hematologic malignancies remained “manageable” during the first 2 months of the COVID-19 pandemic at Levine Cancer Institute in Charlotte, N.C.
That level of manageability has partly been because a surge in cases so far hasn’t arrived at Levine or in most of the surrounding North Carolina and South Carolina communities it serves. As of May 15, 2020, the total number of confirmed and reported COVID-19 cases had reached about 19,000 in North Carolina, and just under 9,000 in South Carolina, out of a total population in the two states of close to 16 million. What’s happened instead at Levine Cancer Institute (LCI) has been a steady but low drumbeat of cases that, by mid-May 2020, totaled fewer than 10 patients with hematologic malignancies diagnosed with COVID-19.
“For a large system with multiple sites throughout North and South Carolina that saw 17,200 new patients in 2019 – including solid tumor, benign hematology, and malignant hematology patients – with 198,000 total patient visits, it is safe to say that we are off to a good start. However, we remain in the early throes of the pandemic and we will need to remain vigilant going forward,” said Peter Voorhees, MD, professor of medicine and director of Medical Operations and Outreach Services in LCI’s Department of Hematologic Oncology and Blood Disorders.
The limited effects to date of COVID-19 at LCI has been thanks to a regimen of great caution for preventing infections that’s been consistently conveyed to LCI patients from before the pandemic’s onset, liberal testing that started early, a proactive plan to defer and temporarily replace infusion care when medically appropriate, a novel staffing approach designed to minimize and contain potential staff outbreaks, and an early pivot to virtual patient contact when feasible.
COVID-19 has had limited penetration into the LCI case load because patients have, in general, “been very careful,” said Dr. Voorhees.
“My impression is that the incidence has been low partly because our patients, especially those with hematologic malignancies including those on active chemotherapy, were already getting warned to be cautious even before the coronavirus using distancing, masking, and meticulous hand hygiene,” he said in an interview that reviewed the steps LCI took starting in March to confront and manage the effects of the then-nascent pandemic. “Since we started screening asymptomatic patients in the inpatient and outpatient settings we have identified only one patient with COVID-19 infection, which supports the low rate of infection in our patient population thus far.”
Another key step was the launch of “robust” testing for the COVID-19 virus starting on March 9, using an in-house assay from LCI’s parent health system, Atrium Health, that delivered results within 24 hours. Testing became available at LCI “earlier than at many other health systems.” At first, testing was limited to patients or staff presenting with symptoms, but in the following weeks, it expanded to more patients, including those without symptoms who were scheduled for treatment at the apheresis center, cell donors and cell recipients, patients arriving for inpatient chemotherapy or cellular therapy, patients arriving from a skilled nursing facility or similar environments, and more recently, outpatient chemotherapy patients. “We’re now doing a lot of screening,” Dr. Voorhees said. “In general, screening has been well received because patients recognize that it’s for their own safety.”
Another piece of COVID-19 preparedness was a move toward technology as an alternative to face-to-face encounters between patients and staff. “We adopted virtual technology early.” When medically appropriate, they provided either video consultations with more tech-savvy patients or telephone-based virtual visits for patients who preferred a more familiar interface. As LCI starts the process of reentry for patients whose face-to-face encounters were deferred, virtual visits will remain an important facet of maintaining care while limiting exposure for appropriate patients and facilitating adequate space for social distancing in the clinics and infusion centers.
Atrium Health also launched a “virtual hospital” geared to intensified remote management of COVID-19 patients who aren’t sick enough for hospitalization. “People who test positive automatically enter the virtual hospital and have regular interactions with their team of providers,” with LCI providing additional support for their patients who get infected. Patients receive an equipment kit that lets them monitor and transmit their vital signs. The virtual hospital program also helps expedite personal needs like delivery of prescriptions and food. “It helps patients manage at home, and has been incredibly useful,” said Dr. Voorhees.
Perhaps the most challenging step LCI clinicians took to preclude a potential COVID-19 case surge was to review all patients receiving infusional therapy or planned cellular therapy and triage those who could potentially tolerate a temporary change to either an oral, at-home regimen or to a brief hold on their treatment. Some patients on maintenance, outpatient infusion-therapy regimens “expressed concern about coming to the clinic. We looked at the patients scheduled to come for infusions and decided which visits were essential and which were deferrable without disrupting care by briefly using a noninfusional approach,” said Dr. Voorhees. The number of patients who had their regimens modified or held was “relatively small,” and with the recent recognition that a surge of infections has not occurred, “we’re now rolling out cautious reentry of those patients back to their originally prescribed chemotherapy.”
In addition to concerns of exposure at infusion clinics, there are concerns about the heightened susceptibility of immunosuppressed hematologic oncology patients to COVID-19 and their risk for more severe infection. “Our view is that, if patients tested positive, continuing immunosuppressive treatment would likely be detrimental,” so when possible treatment is temporarily suspended and then resumed when the infection has cleared. “When patients test positive for a prolonged period, a decision to resume treatment must be in the best interests of the patient and weigh the benefits of resuming therapy against the risks of incurring a more severe infection by restarting potentially immunosuppressive therapy,” Dr. Voorhees said.
The enhanced risk that cancer patients face if they develop COVID-19 was documented in a recent review of 218 cancer patients hospitalized for COVID-19 during parts of March and April in a large New York health system. The results showed an overall mortality rate of 28%, including a 37% rate among 54 patients with hematologic malignancies and a 25% rate among 164 patients with solid tumors. The mortality rate “may not be quite as high as they reported because that depends on how many patients you test, but there is no question that patients with more comorbidities are at higher risk. Patients with active cancer on chemotherapy are a particularly vulnerable population, and many have expressed concerns about their vulnerability,” he observed.
For the few LCI patients who developed COVID-19 infection, the medical staff has had several therapeutic options they could match to each patient’s needs, with help from the Atrium Health infectious disease team. LCI and Atrium Health are participating in several COVID-19 clinical treatment trials, including an investigational convalescent plasma protocol spearheaded by the Mayo Clinic. They have also opened a randomized, phase 2 trial evaluating the safety and efficacy of selinexor (Xpovio), an oral drug that’s Food and Drug Administration approved for patients with multiple myeloma, for treatment of moderate or severe COVID-19 infection. Additional studies evaluating blockade of granulocyte-macrophage colony-stimulating factor, as well as inhaled antiviral therapy, have recently launched, and several additional studies are poised to open in the coming weeks.
The LCI and Atrium Health team also has a supply of the antiviral agent remdesivir as part of the FDA’s expanded access protocol and emergency use authorization. They also have a supply of and experience administering the interleukin-6 receptor inhibitor tocilizumab (Actemra), which showed some suggestion of efficacy in limited experience treating patients with severe or critical COVID-19 infections. Clinicians at LCI have not used the investigational and unproven agents hydroxychloroquine, chloroquine, and azithromycin to either prevent or treat COVID-19.
LCI also instituted measures to try to minimize the risk that staff members could become infected and transmit the virus while asymptomatic. Following conversations held early on with COVID-19–experienced health authorities in China and Italy, the patient-facing LCI staff split into two teams starting on March 23 that alternated responsibility for direct patient interactions every 2 weeks. When one of these teams was off from direct patient contact they continued to care for patients remotely through virtual technologies. The concept was that, if a staffer became infected while remaining asymptomatic during their contact with patients, their status would either become diagnosable or resolve during their 2 weeks away from seeing any patients. Perhaps in part because of this approach infections among staff members “have not been a big issue. We’ve had an incredibly low infection rate among the LCI staff,” Dr. Voorhees noted.
By mid-May, with the imminent threat of a sudden CODIV-19 surge moderated, heme-onc operations at LCI began to cautiously revert to more normal operations. “We’re continuing patient screening for signs and symptoms of COVID-19 infection, testing for asymptomatic infections, and requiring masking and social distancing in the clinics and hospitals, but we’re starting to slowly restore the number of patients at our clinics [virtual and face to face[ and infusion centers,” and the staff’s division into two teams ended. “The idea was to get past a surge and make sure our system was not overwhelmed. We anticipated a local surge in late April, but then it kept getting pushed back. Current projections are for the infection rate among LCI patients to remain low provided that community spread remains stable or, ideally, decreases.” The LCI infectious disease staff is closely monitoring infection rates for early recognition of an outbreak, with plans to follow any new cases with contact tracing. So far, the COVID-19 pandemic at LCI “has been very manageable,” Dr. Voorhees concluded.
“We’re now better positioned to deal with a case surge if it were to happen. We could resume the two-team approach, hospital-wide plans are now in place for a future surge, and we are now up and running with robust testing and inpatient and outpatient virtual technology. The first time, we were all learning on the fly.”
The LCI biostatistics team has been prospectively collecting the Institutes’s COVID-19 patient data, with plans to report their findings.
Dr. Voorhees has had financial relationships with Bristol-Myers Squibb/Celgene, Janssen, Novartis, and Oncopeptides, none of which are relevant to this article.
Keeping hematologic oncology patients on their treatment regimens and caring for inpatients with hematologic malignancies remained “manageable” during the first 2 months of the COVID-19 pandemic at Levine Cancer Institute in Charlotte, N.C.
That level of manageability has partly been because a surge in cases so far hasn’t arrived at Levine or in most of the surrounding North Carolina and South Carolina communities it serves. As of May 15, 2020, the total number of confirmed and reported COVID-19 cases had reached about 19,000 in North Carolina, and just under 9,000 in South Carolina, out of a total population in the two states of close to 16 million. What’s happened instead at Levine Cancer Institute (LCI) has been a steady but low drumbeat of cases that, by mid-May 2020, totaled fewer than 10 patients with hematologic malignancies diagnosed with COVID-19.
“For a large system with multiple sites throughout North and South Carolina that saw 17,200 new patients in 2019 – including solid tumor, benign hematology, and malignant hematology patients – with 198,000 total patient visits, it is safe to say that we are off to a good start. However, we remain in the early throes of the pandemic and we will need to remain vigilant going forward,” said Peter Voorhees, MD, professor of medicine and director of Medical Operations and Outreach Services in LCI’s Department of Hematologic Oncology and Blood Disorders.
The limited effects to date of COVID-19 at LCI has been thanks to a regimen of great caution for preventing infections that’s been consistently conveyed to LCI patients from before the pandemic’s onset, liberal testing that started early, a proactive plan to defer and temporarily replace infusion care when medically appropriate, a novel staffing approach designed to minimize and contain potential staff outbreaks, and an early pivot to virtual patient contact when feasible.
COVID-19 has had limited penetration into the LCI case load because patients have, in general, “been very careful,” said Dr. Voorhees.
“My impression is that the incidence has been low partly because our patients, especially those with hematologic malignancies including those on active chemotherapy, were already getting warned to be cautious even before the coronavirus using distancing, masking, and meticulous hand hygiene,” he said in an interview that reviewed the steps LCI took starting in March to confront and manage the effects of the then-nascent pandemic. “Since we started screening asymptomatic patients in the inpatient and outpatient settings we have identified only one patient with COVID-19 infection, which supports the low rate of infection in our patient population thus far.”
Another key step was the launch of “robust” testing for the COVID-19 virus starting on March 9, using an in-house assay from LCI’s parent health system, Atrium Health, that delivered results within 24 hours. Testing became available at LCI “earlier than at many other health systems.” At first, testing was limited to patients or staff presenting with symptoms, but in the following weeks, it expanded to more patients, including those without symptoms who were scheduled for treatment at the apheresis center, cell donors and cell recipients, patients arriving for inpatient chemotherapy or cellular therapy, patients arriving from a skilled nursing facility or similar environments, and more recently, outpatient chemotherapy patients. “We’re now doing a lot of screening,” Dr. Voorhees said. “In general, screening has been well received because patients recognize that it’s for their own safety.”
Another piece of COVID-19 preparedness was a move toward technology as an alternative to face-to-face encounters between patients and staff. “We adopted virtual technology early.” When medically appropriate, they provided either video consultations with more tech-savvy patients or telephone-based virtual visits for patients who preferred a more familiar interface. As LCI starts the process of reentry for patients whose face-to-face encounters were deferred, virtual visits will remain an important facet of maintaining care while limiting exposure for appropriate patients and facilitating adequate space for social distancing in the clinics and infusion centers.
Atrium Health also launched a “virtual hospital” geared to intensified remote management of COVID-19 patients who aren’t sick enough for hospitalization. “People who test positive automatically enter the virtual hospital and have regular interactions with their team of providers,” with LCI providing additional support for their patients who get infected. Patients receive an equipment kit that lets them monitor and transmit their vital signs. The virtual hospital program also helps expedite personal needs like delivery of prescriptions and food. “It helps patients manage at home, and has been incredibly useful,” said Dr. Voorhees.
Perhaps the most challenging step LCI clinicians took to preclude a potential COVID-19 case surge was to review all patients receiving infusional therapy or planned cellular therapy and triage those who could potentially tolerate a temporary change to either an oral, at-home regimen or to a brief hold on their treatment. Some patients on maintenance, outpatient infusion-therapy regimens “expressed concern about coming to the clinic. We looked at the patients scheduled to come for infusions and decided which visits were essential and which were deferrable without disrupting care by briefly using a noninfusional approach,” said Dr. Voorhees. The number of patients who had their regimens modified or held was “relatively small,” and with the recent recognition that a surge of infections has not occurred, “we’re now rolling out cautious reentry of those patients back to their originally prescribed chemotherapy.”
In addition to concerns of exposure at infusion clinics, there are concerns about the heightened susceptibility of immunosuppressed hematologic oncology patients to COVID-19 and their risk for more severe infection. “Our view is that, if patients tested positive, continuing immunosuppressive treatment would likely be detrimental,” so when possible treatment is temporarily suspended and then resumed when the infection has cleared. “When patients test positive for a prolonged period, a decision to resume treatment must be in the best interests of the patient and weigh the benefits of resuming therapy against the risks of incurring a more severe infection by restarting potentially immunosuppressive therapy,” Dr. Voorhees said.
The enhanced risk that cancer patients face if they develop COVID-19 was documented in a recent review of 218 cancer patients hospitalized for COVID-19 during parts of March and April in a large New York health system. The results showed an overall mortality rate of 28%, including a 37% rate among 54 patients with hematologic malignancies and a 25% rate among 164 patients with solid tumors. The mortality rate “may not be quite as high as they reported because that depends on how many patients you test, but there is no question that patients with more comorbidities are at higher risk. Patients with active cancer on chemotherapy are a particularly vulnerable population, and many have expressed concerns about their vulnerability,” he observed.
For the few LCI patients who developed COVID-19 infection, the medical staff has had several therapeutic options they could match to each patient’s needs, with help from the Atrium Health infectious disease team. LCI and Atrium Health are participating in several COVID-19 clinical treatment trials, including an investigational convalescent plasma protocol spearheaded by the Mayo Clinic. They have also opened a randomized, phase 2 trial evaluating the safety and efficacy of selinexor (Xpovio), an oral drug that’s Food and Drug Administration approved for patients with multiple myeloma, for treatment of moderate or severe COVID-19 infection. Additional studies evaluating blockade of granulocyte-macrophage colony-stimulating factor, as well as inhaled antiviral therapy, have recently launched, and several additional studies are poised to open in the coming weeks.
The LCI and Atrium Health team also has a supply of the antiviral agent remdesivir as part of the FDA’s expanded access protocol and emergency use authorization. They also have a supply of and experience administering the interleukin-6 receptor inhibitor tocilizumab (Actemra), which showed some suggestion of efficacy in limited experience treating patients with severe or critical COVID-19 infections. Clinicians at LCI have not used the investigational and unproven agents hydroxychloroquine, chloroquine, and azithromycin to either prevent or treat COVID-19.
LCI also instituted measures to try to minimize the risk that staff members could become infected and transmit the virus while asymptomatic. Following conversations held early on with COVID-19–experienced health authorities in China and Italy, the patient-facing LCI staff split into two teams starting on March 23 that alternated responsibility for direct patient interactions every 2 weeks. When one of these teams was off from direct patient contact they continued to care for patients remotely through virtual technologies. The concept was that, if a staffer became infected while remaining asymptomatic during their contact with patients, their status would either become diagnosable or resolve during their 2 weeks away from seeing any patients. Perhaps in part because of this approach infections among staff members “have not been a big issue. We’ve had an incredibly low infection rate among the LCI staff,” Dr. Voorhees noted.
By mid-May, with the imminent threat of a sudden CODIV-19 surge moderated, heme-onc operations at LCI began to cautiously revert to more normal operations. “We’re continuing patient screening for signs and symptoms of COVID-19 infection, testing for asymptomatic infections, and requiring masking and social distancing in the clinics and hospitals, but we’re starting to slowly restore the number of patients at our clinics [virtual and face to face[ and infusion centers,” and the staff’s division into two teams ended. “The idea was to get past a surge and make sure our system was not overwhelmed. We anticipated a local surge in late April, but then it kept getting pushed back. Current projections are for the infection rate among LCI patients to remain low provided that community spread remains stable or, ideally, decreases.” The LCI infectious disease staff is closely monitoring infection rates for early recognition of an outbreak, with plans to follow any new cases with contact tracing. So far, the COVID-19 pandemic at LCI “has been very manageable,” Dr. Voorhees concluded.
“We’re now better positioned to deal with a case surge if it were to happen. We could resume the two-team approach, hospital-wide plans are now in place for a future surge, and we are now up and running with robust testing and inpatient and outpatient virtual technology. The first time, we were all learning on the fly.”
The LCI biostatistics team has been prospectively collecting the Institutes’s COVID-19 patient data, with plans to report their findings.
Dr. Voorhees has had financial relationships with Bristol-Myers Squibb/Celgene, Janssen, Novartis, and Oncopeptides, none of which are relevant to this article.
Keeping hematologic oncology patients on their treatment regimens and caring for inpatients with hematologic malignancies remained “manageable” during the first 2 months of the COVID-19 pandemic at Levine Cancer Institute in Charlotte, N.C.
That level of manageability has partly been because a surge in cases so far hasn’t arrived at Levine or in most of the surrounding North Carolina and South Carolina communities it serves. As of May 15, 2020, the total number of confirmed and reported COVID-19 cases had reached about 19,000 in North Carolina, and just under 9,000 in South Carolina, out of a total population in the two states of close to 16 million. What’s happened instead at Levine Cancer Institute (LCI) has been a steady but low drumbeat of cases that, by mid-May 2020, totaled fewer than 10 patients with hematologic malignancies diagnosed with COVID-19.
“For a large system with multiple sites throughout North and South Carolina that saw 17,200 new patients in 2019 – including solid tumor, benign hematology, and malignant hematology patients – with 198,000 total patient visits, it is safe to say that we are off to a good start. However, we remain in the early throes of the pandemic and we will need to remain vigilant going forward,” said Peter Voorhees, MD, professor of medicine and director of Medical Operations and Outreach Services in LCI’s Department of Hematologic Oncology and Blood Disorders.
The limited effects to date of COVID-19 at LCI has been thanks to a regimen of great caution for preventing infections that’s been consistently conveyed to LCI patients from before the pandemic’s onset, liberal testing that started early, a proactive plan to defer and temporarily replace infusion care when medically appropriate, a novel staffing approach designed to minimize and contain potential staff outbreaks, and an early pivot to virtual patient contact when feasible.
COVID-19 has had limited penetration into the LCI case load because patients have, in general, “been very careful,” said Dr. Voorhees.
“My impression is that the incidence has been low partly because our patients, especially those with hematologic malignancies including those on active chemotherapy, were already getting warned to be cautious even before the coronavirus using distancing, masking, and meticulous hand hygiene,” he said in an interview that reviewed the steps LCI took starting in March to confront and manage the effects of the then-nascent pandemic. “Since we started screening asymptomatic patients in the inpatient and outpatient settings we have identified only one patient with COVID-19 infection, which supports the low rate of infection in our patient population thus far.”
Another key step was the launch of “robust” testing for the COVID-19 virus starting on March 9, using an in-house assay from LCI’s parent health system, Atrium Health, that delivered results within 24 hours. Testing became available at LCI “earlier than at many other health systems.” At first, testing was limited to patients or staff presenting with symptoms, but in the following weeks, it expanded to more patients, including those without symptoms who were scheduled for treatment at the apheresis center, cell donors and cell recipients, patients arriving for inpatient chemotherapy or cellular therapy, patients arriving from a skilled nursing facility or similar environments, and more recently, outpatient chemotherapy patients. “We’re now doing a lot of screening,” Dr. Voorhees said. “In general, screening has been well received because patients recognize that it’s for their own safety.”
Another piece of COVID-19 preparedness was a move toward technology as an alternative to face-to-face encounters between patients and staff. “We adopted virtual technology early.” When medically appropriate, they provided either video consultations with more tech-savvy patients or telephone-based virtual visits for patients who preferred a more familiar interface. As LCI starts the process of reentry for patients whose face-to-face encounters were deferred, virtual visits will remain an important facet of maintaining care while limiting exposure for appropriate patients and facilitating adequate space for social distancing in the clinics and infusion centers.
Atrium Health also launched a “virtual hospital” geared to intensified remote management of COVID-19 patients who aren’t sick enough for hospitalization. “People who test positive automatically enter the virtual hospital and have regular interactions with their team of providers,” with LCI providing additional support for their patients who get infected. Patients receive an equipment kit that lets them monitor and transmit their vital signs. The virtual hospital program also helps expedite personal needs like delivery of prescriptions and food. “It helps patients manage at home, and has been incredibly useful,” said Dr. Voorhees.
Perhaps the most challenging step LCI clinicians took to preclude a potential COVID-19 case surge was to review all patients receiving infusional therapy or planned cellular therapy and triage those who could potentially tolerate a temporary change to either an oral, at-home regimen or to a brief hold on their treatment. Some patients on maintenance, outpatient infusion-therapy regimens “expressed concern about coming to the clinic. We looked at the patients scheduled to come for infusions and decided which visits were essential and which were deferrable without disrupting care by briefly using a noninfusional approach,” said Dr. Voorhees. The number of patients who had their regimens modified or held was “relatively small,” and with the recent recognition that a surge of infections has not occurred, “we’re now rolling out cautious reentry of those patients back to their originally prescribed chemotherapy.”
In addition to concerns of exposure at infusion clinics, there are concerns about the heightened susceptibility of immunosuppressed hematologic oncology patients to COVID-19 and their risk for more severe infection. “Our view is that, if patients tested positive, continuing immunosuppressive treatment would likely be detrimental,” so when possible treatment is temporarily suspended and then resumed when the infection has cleared. “When patients test positive for a prolonged period, a decision to resume treatment must be in the best interests of the patient and weigh the benefits of resuming therapy against the risks of incurring a more severe infection by restarting potentially immunosuppressive therapy,” Dr. Voorhees said.
The enhanced risk that cancer patients face if they develop COVID-19 was documented in a recent review of 218 cancer patients hospitalized for COVID-19 during parts of March and April in a large New York health system. The results showed an overall mortality rate of 28%, including a 37% rate among 54 patients with hematologic malignancies and a 25% rate among 164 patients with solid tumors. The mortality rate “may not be quite as high as they reported because that depends on how many patients you test, but there is no question that patients with more comorbidities are at higher risk. Patients with active cancer on chemotherapy are a particularly vulnerable population, and many have expressed concerns about their vulnerability,” he observed.
For the few LCI patients who developed COVID-19 infection, the medical staff has had several therapeutic options they could match to each patient’s needs, with help from the Atrium Health infectious disease team. LCI and Atrium Health are participating in several COVID-19 clinical treatment trials, including an investigational convalescent plasma protocol spearheaded by the Mayo Clinic. They have also opened a randomized, phase 2 trial evaluating the safety and efficacy of selinexor (Xpovio), an oral drug that’s Food and Drug Administration approved for patients with multiple myeloma, for treatment of moderate or severe COVID-19 infection. Additional studies evaluating blockade of granulocyte-macrophage colony-stimulating factor, as well as inhaled antiviral therapy, have recently launched, and several additional studies are poised to open in the coming weeks.
The LCI and Atrium Health team also has a supply of the antiviral agent remdesivir as part of the FDA’s expanded access protocol and emergency use authorization. They also have a supply of and experience administering the interleukin-6 receptor inhibitor tocilizumab (Actemra), which showed some suggestion of efficacy in limited experience treating patients with severe or critical COVID-19 infections. Clinicians at LCI have not used the investigational and unproven agents hydroxychloroquine, chloroquine, and azithromycin to either prevent or treat COVID-19.
LCI also instituted measures to try to minimize the risk that staff members could become infected and transmit the virus while asymptomatic. Following conversations held early on with COVID-19–experienced health authorities in China and Italy, the patient-facing LCI staff split into two teams starting on March 23 that alternated responsibility for direct patient interactions every 2 weeks. When one of these teams was off from direct patient contact they continued to care for patients remotely through virtual technologies. The concept was that, if a staffer became infected while remaining asymptomatic during their contact with patients, their status would either become diagnosable or resolve during their 2 weeks away from seeing any patients. Perhaps in part because of this approach infections among staff members “have not been a big issue. We’ve had an incredibly low infection rate among the LCI staff,” Dr. Voorhees noted.
By mid-May, with the imminent threat of a sudden CODIV-19 surge moderated, heme-onc operations at LCI began to cautiously revert to more normal operations. “We’re continuing patient screening for signs and symptoms of COVID-19 infection, testing for asymptomatic infections, and requiring masking and social distancing in the clinics and hospitals, but we’re starting to slowly restore the number of patients at our clinics [virtual and face to face[ and infusion centers,” and the staff’s division into two teams ended. “The idea was to get past a surge and make sure our system was not overwhelmed. We anticipated a local surge in late April, but then it kept getting pushed back. Current projections are for the infection rate among LCI patients to remain low provided that community spread remains stable or, ideally, decreases.” The LCI infectious disease staff is closely monitoring infection rates for early recognition of an outbreak, with plans to follow any new cases with contact tracing. So far, the COVID-19 pandemic at LCI “has been very manageable,” Dr. Voorhees concluded.
“We’re now better positioned to deal with a case surge if it were to happen. We could resume the two-team approach, hospital-wide plans are now in place for a future surge, and we are now up and running with robust testing and inpatient and outpatient virtual technology. The first time, we were all learning on the fly.”
The LCI biostatistics team has been prospectively collecting the Institutes’s COVID-19 patient data, with plans to report their findings.
Dr. Voorhees has had financial relationships with Bristol-Myers Squibb/Celgene, Janssen, Novartis, and Oncopeptides, none of which are relevant to this article.
Implementation of a Patient Blood Management Program in a Large, Diverse Multi-Hospital System
From BJC HealthCare, St. Louis, MO.
Abstract
Background: There is limited literature relating to patient blood management (PBM) programs in large multi-hospital systems or addressing challenges of implementation across diverse systems comprised of community and academic hospitals.
Objective: To establish a PBM program to improve utilization of blood transfusion units at a multi-hospital system in the Midwest (BJC HealthCare).
Methods: High-impact strategies in establishing the PBM program included formation of Clinical Expert Councils (CECs) of providers, establishment of consensus utilization guidelines, and development of a robust reporting tool. CECs enabled collaboration and facilitated standardization across a complex system of academic, private practice, and tertiary facilities with a diverse community of medical providers. Consensus guidelines and the PBM reporting tool were key to creating meaningful reports to drive provider practice change.
Results: Over the 5 years following implementation of the PBM program, there has been a steady decrease in red blood cell (RBC) utilization. Noticeable changes have taken place at individual hospitals in the system, including reductions in transfusions falling outside guideline parameters from 300 per quarter to less than 8 per quarter at 1 of our community hospitals. No negative impact on patient care has been identified.
Conclusion: In response to current transfusion guidelines and the need for optimizing stewardship of blood product resources, this hospital system successfully implemented a robust PBM program that engaged academic and non-academic community providers and decreased utilization of blood transfusion resources in line with consensus guidelines.
Keywords: quality improvement; RBC transfusion; transfusion practices; provider practice change; utilization trends.
Evidence from clinical trials and published clinical guidelines support the adoption of a restrictive blood transfusion approach in hospitalized, stable patients as best practice.1-5 As such, the development and implementation of patient blood management (PBM) programs has become an increasingly important process improvement for reducing variability in transfusion practices and clinical outcomes.
As recently as 2013, BJC HealthCare, a multi-hospital system in the Midwest, had no standardized, system-wide blood management program, and transfusion practices varied widely across providers and between individual hospitals based on size, patient population, and resources. The system consisted of 13 hospitals, ranging from large tertiary to smaller community and academic hospitals. Although adults constituted the vast majority of the patient population, the hospital system also included a pediatric specialty hospital, St. Louis Children’s Hospital. In addition, some sites were staffed by private practice providers and others by university-based providers, including blood bank medical directors. Due to the diversity of settings and populations, efforts to align transfusion and other practices often faced multiple challenges. However, improving the management of blood transfusions was identified as a key resource stewardship priority in 2013, and implementation of a system-wide program began after extensive discussions and consensus approval by senior hospital system and medical leadership. The primary aim of the program was to optimize overall blood product resource stewardship. Specifically, we sought to control or reduce costs per patient-care episode using strategies that would not negatively impact patient care and could potentially even improve patient outcomes (eg, by avoiding unnecessary transfusions and their attendant risks).
There is a plethora of literature related to the implemention of PBM programs in individual hospitals,6-18 but few reports specifically relate to large multi-hospital health systems,19-21 or directly address the unique challenges of implementation across a diverse system of community and academic hospitals and providers.19 Here, we discuss our experience with establishing a PBM program in a large, diverse, multi-hospital health system, provide examples of innovative strategies, and address challenges faced and lessons learned. Future endeavors of the PBM program at BJC HealthCare are also described.
Setting
BJC HealthCare is one of the largest nonprofit health care organizations in the United States, delivering services to the greater St. Louis, southern Illinois, and mid-Missouri regions, and addressing the health care needs of urban, suburban, and rural communities. As of 2018, the system included 15 hospitals and multiple community health locations comprising more than 3400 staffed beds, 31,500 employees, and 4300 physicians with privileges. The system annually has more than 151,000 hospital admissions, 81,000 outpatient surgery visits, and 537,000 emergency department visits. In addition to inpatient and outpatient care, services include primary care, community health and wellness, workplace health, home health, community mental health, rehabilitation, long-term care, and hospice. As a nonprofit system, BJC is the largest provider of charity care, unreimbursed care, and community benefit in Missouri, highlighting the fact that resource stewardship is a critical issue across the entire system and the communities served.22
PBM Project
Preparation for large-scale change across several hospitals began with creating a framework for the initiative, which consisted of a “burning platform,” a guiding vision, and a coalition. The burning platform identifies the importance and urgency of a change and helps to establish commitment. Between 2012 and 2014, the American Association of Blood Banks (AABB) released new evidence-based guidelines and recommendations calling for more restrictive transfusion practices pertaining to red blood cells (RBCs; ie, a hemoglobin threshold of 7 to 8 g/dL) in both inpatient and outpatient care.2 In addition, use of single-unit transfusions was recognized as best practice by the AABB in the Choosing Wisely campaign.23 Historically, adult patients requiring transfusions were given 2 units in succession. The new recommendations provided a strong basis for changing transfusion practices at BJC. It was believed that aligning transfusion practices with the new guidelines was consistent with the mission and vision of the work: that these changes could lead to optimization of resources, cost control, reductions in unnecessary blood transfusions, and potentially improved care (eg, fewer transfusion-related complications). We used the national guidelines to initiate discussions and to identify clinical conditions and associated laboratory parameters for transfusion therapy.
Once this burning platform was established, a team comprised of physicians, blood bank experts, quality consultants, data analysts, and supply managers, referred to as the Outcomes Team, was formed to lead the change efforts across the system. Initial projects for the team included developing system-wide consensus-based transfusion guidelines, providing education to providers on the new evidence in transfusion practice, and sharing BJC-specific historical utilization data. The guiding principle for the group was that “blood is a valuable resource, but not without risk, and less is more.” In order to disseminate the vision of the initiative across the system, campaign signs with the slogans “7 is the new 10” (referring to the g/dL transfusion threshold) and “1 is the new 2” (referring to the new practice of the preferential transfusion of single units rather than 2 at a time) were displayed in system hospitals.
Last, a guiding coalition of system leaders was needed to help push the initiative forward and sustain the program once fully implemented. Thus, a multidisciplinary PBM Clinical Expert Counciel (CEC) was formed to assist with implementation and maintenance of the program.
Role of PBM Clinical Expert Council
The PBM CEC was designed to improve overall physician and expert engagement and provide a forum where stakeholders from across the system could participate to voice their expert opinion. CECs (which BJC formed in other clinical areas as well) are multidisciplinary teams consisting of clinical, administrative, and technical staff. The open, multidisciplinary structure of the councils allows for collaboration that promotes change across a complex multi-hospital system. Each hospital is represented by key physicians and technical leaders, opening opportunity for both horizontal and vertical partnership.
As part of the overall physician engagement strategy, the PBM CEC was launched across BJC in November 2013 as a decision-making body for gaining system consensus on matters relating to blood management. The initial goals for the PBM CEC were to share information and educate providers and others on the latest evidence, to subsequently debate and develop consensus for guidelines to be applied across BJC, and to identify and adopt gold standard practices to drive and sustain compliance across the system. More specifically, we wanted to focus on how to avoid unnecessary blood transfusions known to be associated with increased risk for adverse reactions, other morbidity, mortality, and longer length of stay. Council members met quarterly to address 6 key drivers: patient safety, informatics and data, quality improvement, efficiencies and workflows, education and competency, and communication and engagement. Members then voted to approve guidelines, policies, and procedures. The group continues to assist in updating and standardizing guidelines and providing input on improving the functionality of the PBM reporting tool.
Development of the PBM Reporting Tool
Providing and sharing data on blood utilization and practices with the CEC and hospital leaders was imperative to driving change. The Outcomes Team deliberated on how best to generate and provide such information, conducting comparisons between selected vendor-based tools and potential internal BJC solutions. After investigation, BJC leadership approved the development of an in-house PBM dashboard tool using Tableau Desktop (Tableau Software, Inc.). The tool consists of an executive page with 5 additional tabs for navigating to the appropriate information (Figure 1 and Figure 2); data within the tool are organized by facility, service, provider, ICD diagnosis, transfusion indication, and the Clinical Classifications Software category, as defined by the Agency for Healthcare Research and Quality.
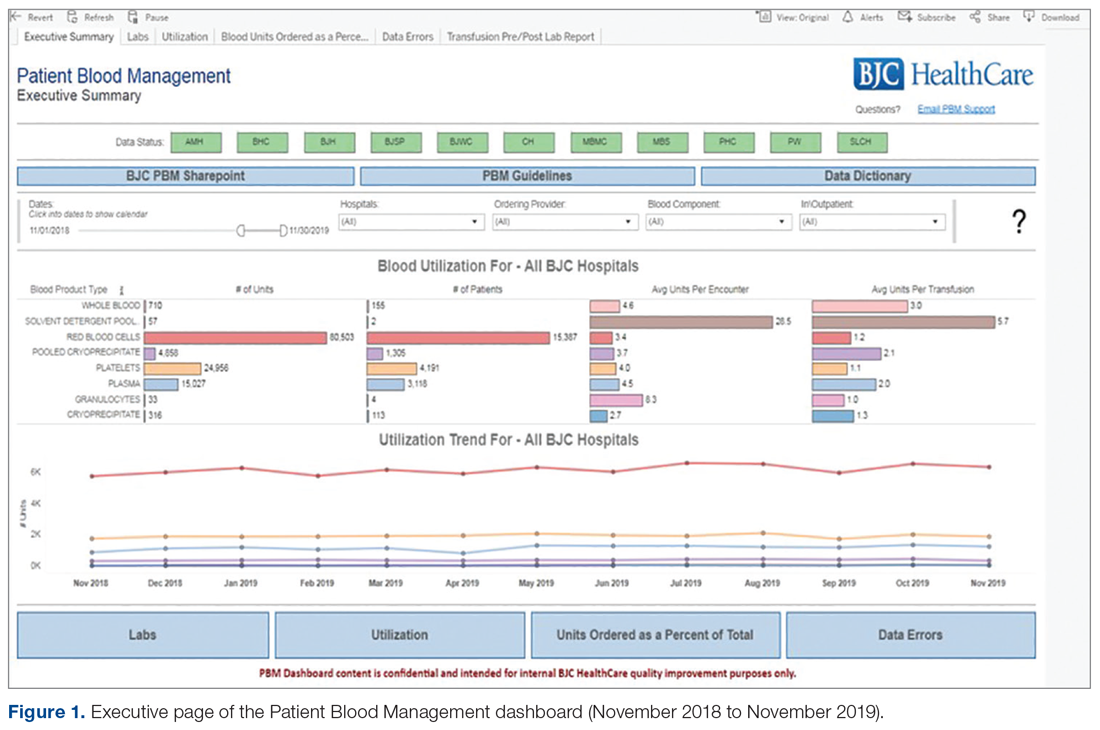
The PBM reporting tool was launched on December 31, 2014. The next priority after the launch was to validate the tool’s blood utilization data and implement enhancements to make the tool more effective for users. A super-user group consisting of blood bank supervisors and managers was established. The goals of the user group were to preview any enhancements before presenting the tool to the larger CEC, test and validate data once new information was added, and share and prioritize future enhancements. User group meetings were held monthly to share best practices and discuss individual facilities’ blood utilization data. In addition, each facility’s representative(s) shared how they were driving changes in provider practice and discussed challenges specific to their facility. Enhancements suggested through the user group included: incorporation of additional lab values into the tool to correspond with other blood products (eg, fibrinogen, hematocrit, international normalized ratio, and platelet count), addition of the specific location where the blood product was administered, and standard naming conventions of locations to allow comparisons across facilities (eg, Emergency Department instead of ED, ER, or EU).
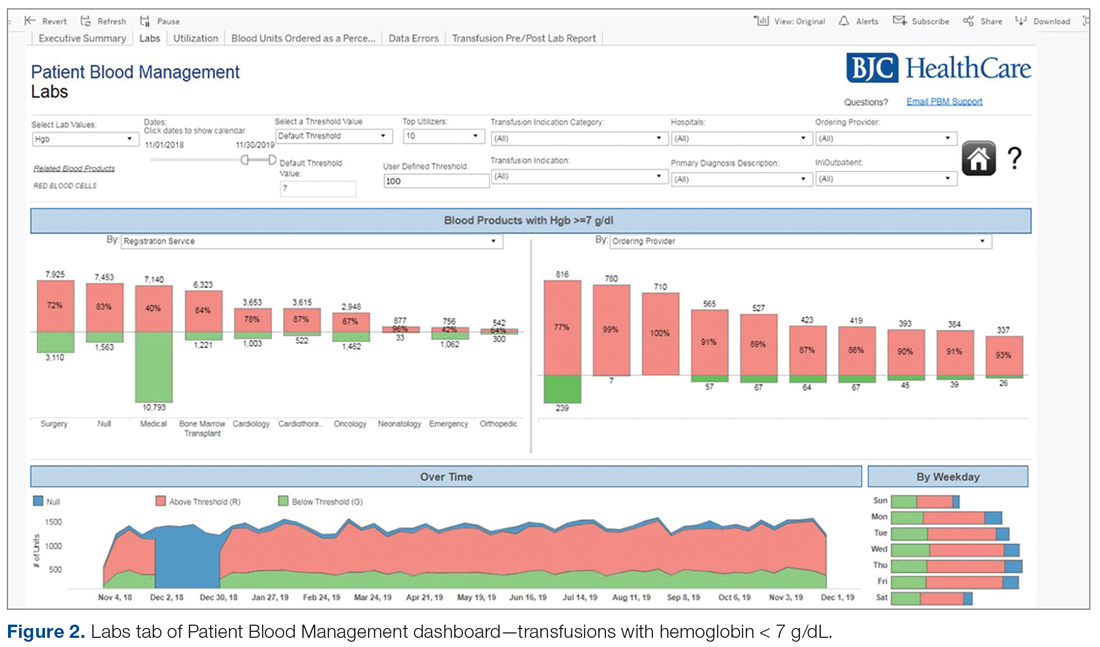
All hospital users were given access to a test version of the reporting tool where they could review enhancements, identify what worked well and what could be done better, and suggest corrections. As changes were made to the hospital lab systems, a sample of data was reviewed and validated with affected facilities to confirm the continued accuracy of the data. To ensure its practicality to users, the tool continues to be improved upon with input from council stakeholders and subject-matter experts.
Measurements
To monitor blood utilization across the health system, we tracked the total RBC units administered by hospital, service, and provider and also tracked pre- and post-transfusion hemoglobin values.
Results
Overall, the system has seen a steady decrease in RBC utilization over the 5 years since the PBM program was implemented (Table
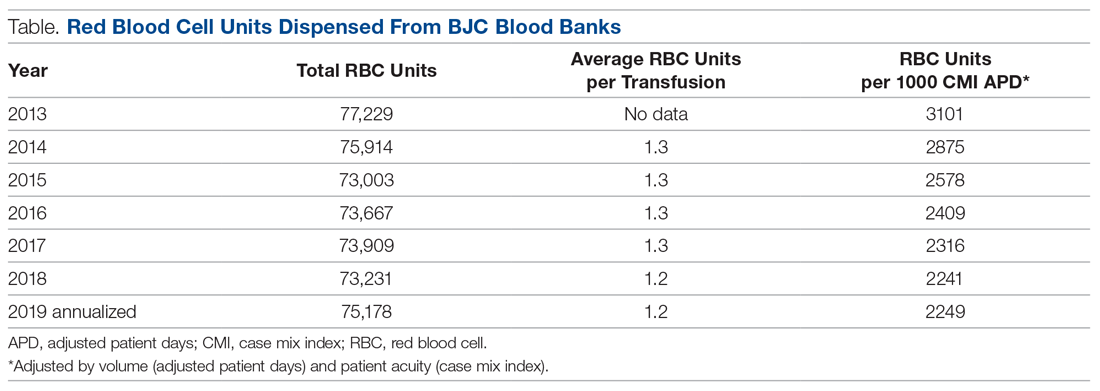
In addition to system-wide improvement, noticeable changes have taken place at individual hospitals in the BJC system. For example, Boone Hospital Center in Columbia, Missouri, began critically reviewing all RBC transfusions starting in 2015 and, to raise awareness, communicating with any provider who transfused a patient outside of transfusion guidelines. Since then, Boone Hospital has seen a dramatic reduction in transfusions considered noncompliant (ie, falling outside guideline parameters), from 300 transfusions per quarter, down to less than 8 per quarter. St. Louis Children’s Hospital also began reviewing blood products utilized by providers that fell outside of the standardized guidelines. At this hospital, physician champions discuss any outliers with the providers involved and use multiple methods for disseminating information to providers, including grand rounds, faculty meetings, and new resident orientations.
Another success has been the partnership between Barnes Jewish St. Peters and Progress West Hospitals in providing PBM education. Their joint effort resulted in implementation of education modules in BJC’s internal learning system, and has provided PBM-related education to more than 367 nurses, blood bank staff, and physicians.
Challenges and Lessons Learned
Implementation of the PBM program was generally successful, but it was not without challenges. One of the biggest challenges was addressing the variation in care and practices across the hospital enterprise. Due to the varying sizes and service goals of individual hospitals, lack of standardization was a significant barrier to change. Gaining trust and buy-in was imperative to increasing compliance with new transfusion policies. The primary concern was finding a balance between respecting physician autonomy and emphasizing and aligning practices with new evidence in the literature. Thus, understanding and applying principles of thoughtful change management was imperative to advancing the framework of the PBM program. The CEC venue enabled collaboration among hospitals and staff and was ultimately used to facilitate the necessary standardization process. To gain the trust of hospital and medical staff, the Outcomes Team conducted several site visits, enabling face-to-face interaction with frontline staff and operational leaders. Moreover, the team’s emphasis on the use of the latest evidence-based guidelines in discussions with hospital and medical staff underscored the need for change.
Frank et al19 describes using an approach similar to our Outcomes Team at the Johns Hopkins Health System. A designated multidisciplinary quality improvement team, referred to as the “clinical community,” worked on implementing best practices for blood management across a system of 5 hospitals. The authors reported similar results, with an overall decrease in number of units transfused, as well as substantial cost savings.19 Our project, along with the project implemented by Frank et al, shows how a “consensus-community” approach, involving stakeholders and various experts across the system, can be be used to align practices among multiple hospitals.
Development of a robust PBM reporting tool was key to creating meaningful monthly reports and driving provider practice change. However, this did require several training sessions, site visits, and computer-based training. Members of the Outcomes Team engaged in one-on-one sessions with tool users as a way of addressing specific areas of concern raised by staff at individual blood banks, and also took part in system-wide initiatives. The team also attended blood bank staff meetings and hospital transfusion committee meetings to educate staff on the evidence and initiative, provide demos of the reporting tool, and allow for a more robust discussion of how the data could be used and shared with other departments. These sessions provided opportunities to identify and prioritize future enhancements, as well as opportunities for continued education and discussion at hospitals, which were critical to ongoing improvement of the reporting tool.
Conclusion and Future Directions
Blood products remain extremely valuable and scarce resources, and all health care professionals must work to prevent unnecessary transfusions and improve clinical outcomes by adhering to the latest evidence-based guidelines. In response to current transfusion guidelines and the need to optimize blood product resources, our system successfully implemented a robust PBM program that engaged both academic and non-academic providers and communities. Several elements of the program helped us overcome the challenges relating to standardization of transfusion practices: consensus-based development of guidelines using the latest scientific evidence; formation and utilization of the CEC venue to gain system-wide consensus around both guidelines and approaches to change; development of a trustworthy and accessible PBM reporting tool (as well as continuing education sessions to improve adoption and utilization of the tool); and ongoing multidisciplinary discussions and support of thoughtful change and sustaining activities. We have seen a system-wide decrease in the number of RBC units transfused (absolute and per case mix-adjusted patient day) since implementing the PBM program, and in the following years have noted a trending decrease in transfusion-related safety events. Although there was a slight increase in reported safety events from 2018 to 2019, this was likely due to the systematic implementation of a new electronic medical record system and improved reporting infrastructure.
Upcoming phases of our system-wide PBM program will include looking at opportunities to improve blood utilization in other specific clinical areas. For example, we have begun discussions with hematology and oncology experts across the system to expand their patient population data within the PBM reporting tool, and to identify areas of opportunity for provider practice change within their specialty. We are also reviewing cardiothoracic surgery transfusion data to identify opportunities for reducing blood utilization in specific clinical scenarios. In addition, we are working to incorporate our 2 newest hospital system members (Memorial Hospital East and Memorial Hospital Belleville) into the PBM program. In collaboration with perioperative leaders across the system, the surgical blood ordering process is being reviewed. The goal of this effort is to reduce blood products ordered in preparation for surgical procedures. We are also currently investigating whether an impact on safety events (ie, reduction in transfusion reactions) can yet be detected. Last, our health care system recently launched a system-wide electronic medical record, and we are eager to see how this will provide us with new methods to monitor and analyze blood administration and utilization data. We look forward to reporting on the expansion of our program and on any clinical outcome improvements gained through avoidance of unnecessary transfusions.
Acknowledgment: The authors thank the leadership within the Center for Clinical Excellence and Supply Chain at BJC HealthCare for their support of this manuscript, as well as all system participants who have contributed to these efforts, especially Mohammad Agha, MD, MHA, current physician leader of the PBM CEC, for his thoughtful edits of this manuscript.
Corresponding author: Audrey A. Gronemeyer, MPH, Center for Clinical Excellence, BJC HealthCare, 8300 Eager Road, Suite 400A, St. Louis, MO 63144; [email protected].
Financial disclosures: None.
1. Carson JL, Grossman BJ, Kleinman S, et al. Red blood cell transfusion: A clinical practice guideline from the AABB*. Ann Intern Med. 2012;157:49-58.
2. Goodnough LT, Levy JH, Murphy MF. Concepts of blood transfusion in adults. Lancet. 2013;381:1845-1854.
3. Hébert PC, Carson JL. Transfusion threshold of 7 g per deciliter—The new normal. N Engl J Med. 2014;371:1459-1461.
4. Gani F, Cerullo M, Ejaz A, et al. Implementation of a blood management program at a tertiary care hospital: Effect on transfusion practices and clinical outcomes among patients undergoing surgery. Ann Surg. 2019;269:1073-1079.
5. Podlasek SJ, Thakkar RN, Rotello LC, et al. Implementing a “why give 2 when 1 will do?” Choosing Wisely campaign. Transfusion. 2016;56:2164.
6. Boral LI, Bernard A, Hjorth T, et al. How do I implement a more restrictive transfusion trigger of hemoglobin level of 7 g/dL at my hospital? Transfusion. 2015;55:937-945.
7. Geissler RG, Kosters C, Franz D, et al. Utilization of blood components in trauma surgery: A single-center, retrospective analysis before and after the implementation of an educative PBM initiative. Transfuse Med Hemother. 2015;42:83-89.
8. Goel R, Cushing MM, Tobian AA. Pediatric patient blood management programs: Not just transfusing little adults. Transfus Med Rev. 2016;30:235-241.
9. Gupta PB, DeMario VM, Amin RM, et al. Patient blood management program improves blood use and clinical outcomes in orthopedic surgery. Anesthesiology. 2018;129;1082-1091.
10. Leahy MF, Roberts H, Mukhtar SA, et al. A pragmatic approach to embedding patient blood management in a tertiary hospital. Transfusion. 2014;54:1133-1145.
11. Leahy MF, Hofmann A, Towler S, et al. Improved outcomes and reduced costs associated with a health-system-wide patient blood management program: A retrospective observational study in four major adult tertiary-care hospitals. Transfusion. 2017;57:1347-1358.
12. Meybohm P, Herrmann E, Steinbicker AU, et al. Patient blood management is associated with a substantial reduction of red blood cell utilization and safe for patient’s outcome: A prospective, multicenter cohort study with a noninferiority design. Ann Surg. 2016;264:203-211.
13. Morgan PN, Coleman PL, Martinez-Garduno CM, et al. Implementation of a patient blood management program in an Australian private hospital orthopedic unit. J Blood Med. 2018;9;83-90.
14. Norgaard A, Stensballe J, de Lichtenberg TH, et al. Three-year follow-up of implementation of evidence-based transfusion practice in a tertiary hospital. Vox Sang. 2017;112:229-239.
15. Meuller MM, Van Remoortel H, Meybohm P, et al. Patient blood management: Recommendations from the 2018 Frankfurt Consensus Conference. JAMA. 2019;321:983-997.
16. Oliver JC, Griffin RL, Hannon T, Marques MB. The success of our patient blood management program depended on an institution-wide change in transfusion practices. Transfusion. 2014;54:2617-2624.
17. Thakkar RN, Lee KH, Ness PM, et al. Relative impact of a patient blood management program on utilization of all three major blood components. Transfusion. 2016;56:2212-2220.
18. Yang WW, Thakkar RN, Gehrie EA, et al. Single-unit transfusions and hemoglobin trigger: relative impact on red cell utilization. Transfusion. 2017;57:1163-1170.
19. Frank SM, Thakkar RN, Podlasek SJ, et al. Implementing a health system-wide patient blood management program with a clinical community approach. Anesthesiology. 2017;127;754-764.
20. Verdecchia NM, Wisniewski MK, Waters JH, et al. Changes in blood product utilization in a seven-hospital system after the implementation of a patient blood management program: A 9-year follow-up. Hematology. 2016;21:490-499.
21. Yazer MH, Waters JH. How do I implement a hospital-based blood management program? Transfusion. 2012;52:1640-1645.
22. BJC HealthCare. Facts and Figures.. BJC HealthCare website. www.bjc.org/About-Us/Facts-Figures. Accessed November 18, 2019.
23. Callum JL, Waters JH, Shaz BH, et al. The AABB recommendations for the Choosing Wisely campaign of the American Board of Internal Medicine. Transfusion. 2014;54:2344-2352.
From BJC HealthCare, St. Louis, MO.
Abstract
Background: There is limited literature relating to patient blood management (PBM) programs in large multi-hospital systems or addressing challenges of implementation across diverse systems comprised of community and academic hospitals.
Objective: To establish a PBM program to improve utilization of blood transfusion units at a multi-hospital system in the Midwest (BJC HealthCare).
Methods: High-impact strategies in establishing the PBM program included formation of Clinical Expert Councils (CECs) of providers, establishment of consensus utilization guidelines, and development of a robust reporting tool. CECs enabled collaboration and facilitated standardization across a complex system of academic, private practice, and tertiary facilities with a diverse community of medical providers. Consensus guidelines and the PBM reporting tool were key to creating meaningful reports to drive provider practice change.
Results: Over the 5 years following implementation of the PBM program, there has been a steady decrease in red blood cell (RBC) utilization. Noticeable changes have taken place at individual hospitals in the system, including reductions in transfusions falling outside guideline parameters from 300 per quarter to less than 8 per quarter at 1 of our community hospitals. No negative impact on patient care has been identified.
Conclusion: In response to current transfusion guidelines and the need for optimizing stewardship of blood product resources, this hospital system successfully implemented a robust PBM program that engaged academic and non-academic community providers and decreased utilization of blood transfusion resources in line with consensus guidelines.
Keywords: quality improvement; RBC transfusion; transfusion practices; provider practice change; utilization trends.
Evidence from clinical trials and published clinical guidelines support the adoption of a restrictive blood transfusion approach in hospitalized, stable patients as best practice.1-5 As such, the development and implementation of patient blood management (PBM) programs has become an increasingly important process improvement for reducing variability in transfusion practices and clinical outcomes.
As recently as 2013, BJC HealthCare, a multi-hospital system in the Midwest, had no standardized, system-wide blood management program, and transfusion practices varied widely across providers and between individual hospitals based on size, patient population, and resources. The system consisted of 13 hospitals, ranging from large tertiary to smaller community and academic hospitals. Although adults constituted the vast majority of the patient population, the hospital system also included a pediatric specialty hospital, St. Louis Children’s Hospital. In addition, some sites were staffed by private practice providers and others by university-based providers, including blood bank medical directors. Due to the diversity of settings and populations, efforts to align transfusion and other practices often faced multiple challenges. However, improving the management of blood transfusions was identified as a key resource stewardship priority in 2013, and implementation of a system-wide program began after extensive discussions and consensus approval by senior hospital system and medical leadership. The primary aim of the program was to optimize overall blood product resource stewardship. Specifically, we sought to control or reduce costs per patient-care episode using strategies that would not negatively impact patient care and could potentially even improve patient outcomes (eg, by avoiding unnecessary transfusions and their attendant risks).
There is a plethora of literature related to the implemention of PBM programs in individual hospitals,6-18 but few reports specifically relate to large multi-hospital health systems,19-21 or directly address the unique challenges of implementation across a diverse system of community and academic hospitals and providers.19 Here, we discuss our experience with establishing a PBM program in a large, diverse, multi-hospital health system, provide examples of innovative strategies, and address challenges faced and lessons learned. Future endeavors of the PBM program at BJC HealthCare are also described.
Setting
BJC HealthCare is one of the largest nonprofit health care organizations in the United States, delivering services to the greater St. Louis, southern Illinois, and mid-Missouri regions, and addressing the health care needs of urban, suburban, and rural communities. As of 2018, the system included 15 hospitals and multiple community health locations comprising more than 3400 staffed beds, 31,500 employees, and 4300 physicians with privileges. The system annually has more than 151,000 hospital admissions, 81,000 outpatient surgery visits, and 537,000 emergency department visits. In addition to inpatient and outpatient care, services include primary care, community health and wellness, workplace health, home health, community mental health, rehabilitation, long-term care, and hospice. As a nonprofit system, BJC is the largest provider of charity care, unreimbursed care, and community benefit in Missouri, highlighting the fact that resource stewardship is a critical issue across the entire system and the communities served.22
PBM Project
Preparation for large-scale change across several hospitals began with creating a framework for the initiative, which consisted of a “burning platform,” a guiding vision, and a coalition. The burning platform identifies the importance and urgency of a change and helps to establish commitment. Between 2012 and 2014, the American Association of Blood Banks (AABB) released new evidence-based guidelines and recommendations calling for more restrictive transfusion practices pertaining to red blood cells (RBCs; ie, a hemoglobin threshold of 7 to 8 g/dL) in both inpatient and outpatient care.2 In addition, use of single-unit transfusions was recognized as best practice by the AABB in the Choosing Wisely campaign.23 Historically, adult patients requiring transfusions were given 2 units in succession. The new recommendations provided a strong basis for changing transfusion practices at BJC. It was believed that aligning transfusion practices with the new guidelines was consistent with the mission and vision of the work: that these changes could lead to optimization of resources, cost control, reductions in unnecessary blood transfusions, and potentially improved care (eg, fewer transfusion-related complications). We used the national guidelines to initiate discussions and to identify clinical conditions and associated laboratory parameters for transfusion therapy.
Once this burning platform was established, a team comprised of physicians, blood bank experts, quality consultants, data analysts, and supply managers, referred to as the Outcomes Team, was formed to lead the change efforts across the system. Initial projects for the team included developing system-wide consensus-based transfusion guidelines, providing education to providers on the new evidence in transfusion practice, and sharing BJC-specific historical utilization data. The guiding principle for the group was that “blood is a valuable resource, but not without risk, and less is more.” In order to disseminate the vision of the initiative across the system, campaign signs with the slogans “7 is the new 10” (referring to the g/dL transfusion threshold) and “1 is the new 2” (referring to the new practice of the preferential transfusion of single units rather than 2 at a time) were displayed in system hospitals.
Last, a guiding coalition of system leaders was needed to help push the initiative forward and sustain the program once fully implemented. Thus, a multidisciplinary PBM Clinical Expert Counciel (CEC) was formed to assist with implementation and maintenance of the program.
Role of PBM Clinical Expert Council
The PBM CEC was designed to improve overall physician and expert engagement and provide a forum where stakeholders from across the system could participate to voice their expert opinion. CECs (which BJC formed in other clinical areas as well) are multidisciplinary teams consisting of clinical, administrative, and technical staff. The open, multidisciplinary structure of the councils allows for collaboration that promotes change across a complex multi-hospital system. Each hospital is represented by key physicians and technical leaders, opening opportunity for both horizontal and vertical partnership.
As part of the overall physician engagement strategy, the PBM CEC was launched across BJC in November 2013 as a decision-making body for gaining system consensus on matters relating to blood management. The initial goals for the PBM CEC were to share information and educate providers and others on the latest evidence, to subsequently debate and develop consensus for guidelines to be applied across BJC, and to identify and adopt gold standard practices to drive and sustain compliance across the system. More specifically, we wanted to focus on how to avoid unnecessary blood transfusions known to be associated with increased risk for adverse reactions, other morbidity, mortality, and longer length of stay. Council members met quarterly to address 6 key drivers: patient safety, informatics and data, quality improvement, efficiencies and workflows, education and competency, and communication and engagement. Members then voted to approve guidelines, policies, and procedures. The group continues to assist in updating and standardizing guidelines and providing input on improving the functionality of the PBM reporting tool.
Development of the PBM Reporting Tool
Providing and sharing data on blood utilization and practices with the CEC and hospital leaders was imperative to driving change. The Outcomes Team deliberated on how best to generate and provide such information, conducting comparisons between selected vendor-based tools and potential internal BJC solutions. After investigation, BJC leadership approved the development of an in-house PBM dashboard tool using Tableau Desktop (Tableau Software, Inc.). The tool consists of an executive page with 5 additional tabs for navigating to the appropriate information (Figure 1 and Figure 2); data within the tool are organized by facility, service, provider, ICD diagnosis, transfusion indication, and the Clinical Classifications Software category, as defined by the Agency for Healthcare Research and Quality.

The PBM reporting tool was launched on December 31, 2014. The next priority after the launch was to validate the tool’s blood utilization data and implement enhancements to make the tool more effective for users. A super-user group consisting of blood bank supervisors and managers was established. The goals of the user group were to preview any enhancements before presenting the tool to the larger CEC, test and validate data once new information was added, and share and prioritize future enhancements. User group meetings were held monthly to share best practices and discuss individual facilities’ blood utilization data. In addition, each facility’s representative(s) shared how they were driving changes in provider practice and discussed challenges specific to their facility. Enhancements suggested through the user group included: incorporation of additional lab values into the tool to correspond with other blood products (eg, fibrinogen, hematocrit, international normalized ratio, and platelet count), addition of the specific location where the blood product was administered, and standard naming conventions of locations to allow comparisons across facilities (eg, Emergency Department instead of ED, ER, or EU).

All hospital users were given access to a test version of the reporting tool where they could review enhancements, identify what worked well and what could be done better, and suggest corrections. As changes were made to the hospital lab systems, a sample of data was reviewed and validated with affected facilities to confirm the continued accuracy of the data. To ensure its practicality to users, the tool continues to be improved upon with input from council stakeholders and subject-matter experts.
Measurements
To monitor blood utilization across the health system, we tracked the total RBC units administered by hospital, service, and provider and also tracked pre- and post-transfusion hemoglobin values.
Results
Overall, the system has seen a steady decrease in RBC utilization over the 5 years since the PBM program was implemented (Table

In addition to system-wide improvement, noticeable changes have taken place at individual hospitals in the BJC system. For example, Boone Hospital Center in Columbia, Missouri, began critically reviewing all RBC transfusions starting in 2015 and, to raise awareness, communicating with any provider who transfused a patient outside of transfusion guidelines. Since then, Boone Hospital has seen a dramatic reduction in transfusions considered noncompliant (ie, falling outside guideline parameters), from 300 transfusions per quarter, down to less than 8 per quarter. St. Louis Children’s Hospital also began reviewing blood products utilized by providers that fell outside of the standardized guidelines. At this hospital, physician champions discuss any outliers with the providers involved and use multiple methods for disseminating information to providers, including grand rounds, faculty meetings, and new resident orientations.
Another success has been the partnership between Barnes Jewish St. Peters and Progress West Hospitals in providing PBM education. Their joint effort resulted in implementation of education modules in BJC’s internal learning system, and has provided PBM-related education to more than 367 nurses, blood bank staff, and physicians.
Challenges and Lessons Learned
Implementation of the PBM program was generally successful, but it was not without challenges. One of the biggest challenges was addressing the variation in care and practices across the hospital enterprise. Due to the varying sizes and service goals of individual hospitals, lack of standardization was a significant barrier to change. Gaining trust and buy-in was imperative to increasing compliance with new transfusion policies. The primary concern was finding a balance between respecting physician autonomy and emphasizing and aligning practices with new evidence in the literature. Thus, understanding and applying principles of thoughtful change management was imperative to advancing the framework of the PBM program. The CEC venue enabled collaboration among hospitals and staff and was ultimately used to facilitate the necessary standardization process. To gain the trust of hospital and medical staff, the Outcomes Team conducted several site visits, enabling face-to-face interaction with frontline staff and operational leaders. Moreover, the team’s emphasis on the use of the latest evidence-based guidelines in discussions with hospital and medical staff underscored the need for change.
Frank et al19 describes using an approach similar to our Outcomes Team at the Johns Hopkins Health System. A designated multidisciplinary quality improvement team, referred to as the “clinical community,” worked on implementing best practices for blood management across a system of 5 hospitals. The authors reported similar results, with an overall decrease in number of units transfused, as well as substantial cost savings.19 Our project, along with the project implemented by Frank et al, shows how a “consensus-community” approach, involving stakeholders and various experts across the system, can be be used to align practices among multiple hospitals.
Development of a robust PBM reporting tool was key to creating meaningful monthly reports and driving provider practice change. However, this did require several training sessions, site visits, and computer-based training. Members of the Outcomes Team engaged in one-on-one sessions with tool users as a way of addressing specific areas of concern raised by staff at individual blood banks, and also took part in system-wide initiatives. The team also attended blood bank staff meetings and hospital transfusion committee meetings to educate staff on the evidence and initiative, provide demos of the reporting tool, and allow for a more robust discussion of how the data could be used and shared with other departments. These sessions provided opportunities to identify and prioritize future enhancements, as well as opportunities for continued education and discussion at hospitals, which were critical to ongoing improvement of the reporting tool.
Conclusion and Future Directions
Blood products remain extremely valuable and scarce resources, and all health care professionals must work to prevent unnecessary transfusions and improve clinical outcomes by adhering to the latest evidence-based guidelines. In response to current transfusion guidelines and the need to optimize blood product resources, our system successfully implemented a robust PBM program that engaged both academic and non-academic providers and communities. Several elements of the program helped us overcome the challenges relating to standardization of transfusion practices: consensus-based development of guidelines using the latest scientific evidence; formation and utilization of the CEC venue to gain system-wide consensus around both guidelines and approaches to change; development of a trustworthy and accessible PBM reporting tool (as well as continuing education sessions to improve adoption and utilization of the tool); and ongoing multidisciplinary discussions and support of thoughtful change and sustaining activities. We have seen a system-wide decrease in the number of RBC units transfused (absolute and per case mix-adjusted patient day) since implementing the PBM program, and in the following years have noted a trending decrease in transfusion-related safety events. Although there was a slight increase in reported safety events from 2018 to 2019, this was likely due to the systematic implementation of a new electronic medical record system and improved reporting infrastructure.
Upcoming phases of our system-wide PBM program will include looking at opportunities to improve blood utilization in other specific clinical areas. For example, we have begun discussions with hematology and oncology experts across the system to expand their patient population data within the PBM reporting tool, and to identify areas of opportunity for provider practice change within their specialty. We are also reviewing cardiothoracic surgery transfusion data to identify opportunities for reducing blood utilization in specific clinical scenarios. In addition, we are working to incorporate our 2 newest hospital system members (Memorial Hospital East and Memorial Hospital Belleville) into the PBM program. In collaboration with perioperative leaders across the system, the surgical blood ordering process is being reviewed. The goal of this effort is to reduce blood products ordered in preparation for surgical procedures. We are also currently investigating whether an impact on safety events (ie, reduction in transfusion reactions) can yet be detected. Last, our health care system recently launched a system-wide electronic medical record, and we are eager to see how this will provide us with new methods to monitor and analyze blood administration and utilization data. We look forward to reporting on the expansion of our program and on any clinical outcome improvements gained through avoidance of unnecessary transfusions.
Acknowledgment: The authors thank the leadership within the Center for Clinical Excellence and Supply Chain at BJC HealthCare for their support of this manuscript, as well as all system participants who have contributed to these efforts, especially Mohammad Agha, MD, MHA, current physician leader of the PBM CEC, for his thoughtful edits of this manuscript.
Corresponding author: Audrey A. Gronemeyer, MPH, Center for Clinical Excellence, BJC HealthCare, 8300 Eager Road, Suite 400A, St. Louis, MO 63144; [email protected].
Financial disclosures: None.
From BJC HealthCare, St. Louis, MO.
Abstract
Background: There is limited literature relating to patient blood management (PBM) programs in large multi-hospital systems or addressing challenges of implementation across diverse systems comprised of community and academic hospitals.
Objective: To establish a PBM program to improve utilization of blood transfusion units at a multi-hospital system in the Midwest (BJC HealthCare).
Methods: High-impact strategies in establishing the PBM program included formation of Clinical Expert Councils (CECs) of providers, establishment of consensus utilization guidelines, and development of a robust reporting tool. CECs enabled collaboration and facilitated standardization across a complex system of academic, private practice, and tertiary facilities with a diverse community of medical providers. Consensus guidelines and the PBM reporting tool were key to creating meaningful reports to drive provider practice change.
Results: Over the 5 years following implementation of the PBM program, there has been a steady decrease in red blood cell (RBC) utilization. Noticeable changes have taken place at individual hospitals in the system, including reductions in transfusions falling outside guideline parameters from 300 per quarter to less than 8 per quarter at 1 of our community hospitals. No negative impact on patient care has been identified.
Conclusion: In response to current transfusion guidelines and the need for optimizing stewardship of blood product resources, this hospital system successfully implemented a robust PBM program that engaged academic and non-academic community providers and decreased utilization of blood transfusion resources in line with consensus guidelines.
Keywords: quality improvement; RBC transfusion; transfusion practices; provider practice change; utilization trends.
Evidence from clinical trials and published clinical guidelines support the adoption of a restrictive blood transfusion approach in hospitalized, stable patients as best practice.1-5 As such, the development and implementation of patient blood management (PBM) programs has become an increasingly important process improvement for reducing variability in transfusion practices and clinical outcomes.
As recently as 2013, BJC HealthCare, a multi-hospital system in the Midwest, had no standardized, system-wide blood management program, and transfusion practices varied widely across providers and between individual hospitals based on size, patient population, and resources. The system consisted of 13 hospitals, ranging from large tertiary to smaller community and academic hospitals. Although adults constituted the vast majority of the patient population, the hospital system also included a pediatric specialty hospital, St. Louis Children’s Hospital. In addition, some sites were staffed by private practice providers and others by university-based providers, including blood bank medical directors. Due to the diversity of settings and populations, efforts to align transfusion and other practices often faced multiple challenges. However, improving the management of blood transfusions was identified as a key resource stewardship priority in 2013, and implementation of a system-wide program began after extensive discussions and consensus approval by senior hospital system and medical leadership. The primary aim of the program was to optimize overall blood product resource stewardship. Specifically, we sought to control or reduce costs per patient-care episode using strategies that would not negatively impact patient care and could potentially even improve patient outcomes (eg, by avoiding unnecessary transfusions and their attendant risks).
There is a plethora of literature related to the implemention of PBM programs in individual hospitals,6-18 but few reports specifically relate to large multi-hospital health systems,19-21 or directly address the unique challenges of implementation across a diverse system of community and academic hospitals and providers.19 Here, we discuss our experience with establishing a PBM program in a large, diverse, multi-hospital health system, provide examples of innovative strategies, and address challenges faced and lessons learned. Future endeavors of the PBM program at BJC HealthCare are also described.
Setting
BJC HealthCare is one of the largest nonprofit health care organizations in the United States, delivering services to the greater St. Louis, southern Illinois, and mid-Missouri regions, and addressing the health care needs of urban, suburban, and rural communities. As of 2018, the system included 15 hospitals and multiple community health locations comprising more than 3400 staffed beds, 31,500 employees, and 4300 physicians with privileges. The system annually has more than 151,000 hospital admissions, 81,000 outpatient surgery visits, and 537,000 emergency department visits. In addition to inpatient and outpatient care, services include primary care, community health and wellness, workplace health, home health, community mental health, rehabilitation, long-term care, and hospice. As a nonprofit system, BJC is the largest provider of charity care, unreimbursed care, and community benefit in Missouri, highlighting the fact that resource stewardship is a critical issue across the entire system and the communities served.22
PBM Project
Preparation for large-scale change across several hospitals began with creating a framework for the initiative, which consisted of a “burning platform,” a guiding vision, and a coalition. The burning platform identifies the importance and urgency of a change and helps to establish commitment. Between 2012 and 2014, the American Association of Blood Banks (AABB) released new evidence-based guidelines and recommendations calling for more restrictive transfusion practices pertaining to red blood cells (RBCs; ie, a hemoglobin threshold of 7 to 8 g/dL) in both inpatient and outpatient care.2 In addition, use of single-unit transfusions was recognized as best practice by the AABB in the Choosing Wisely campaign.23 Historically, adult patients requiring transfusions were given 2 units in succession. The new recommendations provided a strong basis for changing transfusion practices at BJC. It was believed that aligning transfusion practices with the new guidelines was consistent with the mission and vision of the work: that these changes could lead to optimization of resources, cost control, reductions in unnecessary blood transfusions, and potentially improved care (eg, fewer transfusion-related complications). We used the national guidelines to initiate discussions and to identify clinical conditions and associated laboratory parameters for transfusion therapy.
Once this burning platform was established, a team comprised of physicians, blood bank experts, quality consultants, data analysts, and supply managers, referred to as the Outcomes Team, was formed to lead the change efforts across the system. Initial projects for the team included developing system-wide consensus-based transfusion guidelines, providing education to providers on the new evidence in transfusion practice, and sharing BJC-specific historical utilization data. The guiding principle for the group was that “blood is a valuable resource, but not without risk, and less is more.” In order to disseminate the vision of the initiative across the system, campaign signs with the slogans “7 is the new 10” (referring to the g/dL transfusion threshold) and “1 is the new 2” (referring to the new practice of the preferential transfusion of single units rather than 2 at a time) were displayed in system hospitals.
Last, a guiding coalition of system leaders was needed to help push the initiative forward and sustain the program once fully implemented. Thus, a multidisciplinary PBM Clinical Expert Counciel (CEC) was formed to assist with implementation and maintenance of the program.
Role of PBM Clinical Expert Council
The PBM CEC was designed to improve overall physician and expert engagement and provide a forum where stakeholders from across the system could participate to voice their expert opinion. CECs (which BJC formed in other clinical areas as well) are multidisciplinary teams consisting of clinical, administrative, and technical staff. The open, multidisciplinary structure of the councils allows for collaboration that promotes change across a complex multi-hospital system. Each hospital is represented by key physicians and technical leaders, opening opportunity for both horizontal and vertical partnership.
As part of the overall physician engagement strategy, the PBM CEC was launched across BJC in November 2013 as a decision-making body for gaining system consensus on matters relating to blood management. The initial goals for the PBM CEC were to share information and educate providers and others on the latest evidence, to subsequently debate and develop consensus for guidelines to be applied across BJC, and to identify and adopt gold standard practices to drive and sustain compliance across the system. More specifically, we wanted to focus on how to avoid unnecessary blood transfusions known to be associated with increased risk for adverse reactions, other morbidity, mortality, and longer length of stay. Council members met quarterly to address 6 key drivers: patient safety, informatics and data, quality improvement, efficiencies and workflows, education and competency, and communication and engagement. Members then voted to approve guidelines, policies, and procedures. The group continues to assist in updating and standardizing guidelines and providing input on improving the functionality of the PBM reporting tool.
Development of the PBM Reporting Tool
Providing and sharing data on blood utilization and practices with the CEC and hospital leaders was imperative to driving change. The Outcomes Team deliberated on how best to generate and provide such information, conducting comparisons between selected vendor-based tools and potential internal BJC solutions. After investigation, BJC leadership approved the development of an in-house PBM dashboard tool using Tableau Desktop (Tableau Software, Inc.). The tool consists of an executive page with 5 additional tabs for navigating to the appropriate information (Figure 1 and Figure 2); data within the tool are organized by facility, service, provider, ICD diagnosis, transfusion indication, and the Clinical Classifications Software category, as defined by the Agency for Healthcare Research and Quality.

The PBM reporting tool was launched on December 31, 2014. The next priority after the launch was to validate the tool’s blood utilization data and implement enhancements to make the tool more effective for users. A super-user group consisting of blood bank supervisors and managers was established. The goals of the user group were to preview any enhancements before presenting the tool to the larger CEC, test and validate data once new information was added, and share and prioritize future enhancements. User group meetings were held monthly to share best practices and discuss individual facilities’ blood utilization data. In addition, each facility’s representative(s) shared how they were driving changes in provider practice and discussed challenges specific to their facility. Enhancements suggested through the user group included: incorporation of additional lab values into the tool to correspond with other blood products (eg, fibrinogen, hematocrit, international normalized ratio, and platelet count), addition of the specific location where the blood product was administered, and standard naming conventions of locations to allow comparisons across facilities (eg, Emergency Department instead of ED, ER, or EU).

All hospital users were given access to a test version of the reporting tool where they could review enhancements, identify what worked well and what could be done better, and suggest corrections. As changes were made to the hospital lab systems, a sample of data was reviewed and validated with affected facilities to confirm the continued accuracy of the data. To ensure its practicality to users, the tool continues to be improved upon with input from council stakeholders and subject-matter experts.
Measurements
To monitor blood utilization across the health system, we tracked the total RBC units administered by hospital, service, and provider and also tracked pre- and post-transfusion hemoglobin values.
Results
Overall, the system has seen a steady decrease in RBC utilization over the 5 years since the PBM program was implemented (Table

In addition to system-wide improvement, noticeable changes have taken place at individual hospitals in the BJC system. For example, Boone Hospital Center in Columbia, Missouri, began critically reviewing all RBC transfusions starting in 2015 and, to raise awareness, communicating with any provider who transfused a patient outside of transfusion guidelines. Since then, Boone Hospital has seen a dramatic reduction in transfusions considered noncompliant (ie, falling outside guideline parameters), from 300 transfusions per quarter, down to less than 8 per quarter. St. Louis Children’s Hospital also began reviewing blood products utilized by providers that fell outside of the standardized guidelines. At this hospital, physician champions discuss any outliers with the providers involved and use multiple methods for disseminating information to providers, including grand rounds, faculty meetings, and new resident orientations.
Another success has been the partnership between Barnes Jewish St. Peters and Progress West Hospitals in providing PBM education. Their joint effort resulted in implementation of education modules in BJC’s internal learning system, and has provided PBM-related education to more than 367 nurses, blood bank staff, and physicians.
Challenges and Lessons Learned
Implementation of the PBM program was generally successful, but it was not without challenges. One of the biggest challenges was addressing the variation in care and practices across the hospital enterprise. Due to the varying sizes and service goals of individual hospitals, lack of standardization was a significant barrier to change. Gaining trust and buy-in was imperative to increasing compliance with new transfusion policies. The primary concern was finding a balance between respecting physician autonomy and emphasizing and aligning practices with new evidence in the literature. Thus, understanding and applying principles of thoughtful change management was imperative to advancing the framework of the PBM program. The CEC venue enabled collaboration among hospitals and staff and was ultimately used to facilitate the necessary standardization process. To gain the trust of hospital and medical staff, the Outcomes Team conducted several site visits, enabling face-to-face interaction with frontline staff and operational leaders. Moreover, the team’s emphasis on the use of the latest evidence-based guidelines in discussions with hospital and medical staff underscored the need for change.
Frank et al19 describes using an approach similar to our Outcomes Team at the Johns Hopkins Health System. A designated multidisciplinary quality improvement team, referred to as the “clinical community,” worked on implementing best practices for blood management across a system of 5 hospitals. The authors reported similar results, with an overall decrease in number of units transfused, as well as substantial cost savings.19 Our project, along with the project implemented by Frank et al, shows how a “consensus-community” approach, involving stakeholders and various experts across the system, can be be used to align practices among multiple hospitals.
Development of a robust PBM reporting tool was key to creating meaningful monthly reports and driving provider practice change. However, this did require several training sessions, site visits, and computer-based training. Members of the Outcomes Team engaged in one-on-one sessions with tool users as a way of addressing specific areas of concern raised by staff at individual blood banks, and also took part in system-wide initiatives. The team also attended blood bank staff meetings and hospital transfusion committee meetings to educate staff on the evidence and initiative, provide demos of the reporting tool, and allow for a more robust discussion of how the data could be used and shared with other departments. These sessions provided opportunities to identify and prioritize future enhancements, as well as opportunities for continued education and discussion at hospitals, which were critical to ongoing improvement of the reporting tool.
Conclusion and Future Directions
Blood products remain extremely valuable and scarce resources, and all health care professionals must work to prevent unnecessary transfusions and improve clinical outcomes by adhering to the latest evidence-based guidelines. In response to current transfusion guidelines and the need to optimize blood product resources, our system successfully implemented a robust PBM program that engaged both academic and non-academic providers and communities. Several elements of the program helped us overcome the challenges relating to standardization of transfusion practices: consensus-based development of guidelines using the latest scientific evidence; formation and utilization of the CEC venue to gain system-wide consensus around both guidelines and approaches to change; development of a trustworthy and accessible PBM reporting tool (as well as continuing education sessions to improve adoption and utilization of the tool); and ongoing multidisciplinary discussions and support of thoughtful change and sustaining activities. We have seen a system-wide decrease in the number of RBC units transfused (absolute and per case mix-adjusted patient day) since implementing the PBM program, and in the following years have noted a trending decrease in transfusion-related safety events. Although there was a slight increase in reported safety events from 2018 to 2019, this was likely due to the systematic implementation of a new electronic medical record system and improved reporting infrastructure.
Upcoming phases of our system-wide PBM program will include looking at opportunities to improve blood utilization in other specific clinical areas. For example, we have begun discussions with hematology and oncology experts across the system to expand their patient population data within the PBM reporting tool, and to identify areas of opportunity for provider practice change within their specialty. We are also reviewing cardiothoracic surgery transfusion data to identify opportunities for reducing blood utilization in specific clinical scenarios. In addition, we are working to incorporate our 2 newest hospital system members (Memorial Hospital East and Memorial Hospital Belleville) into the PBM program. In collaboration with perioperative leaders across the system, the surgical blood ordering process is being reviewed. The goal of this effort is to reduce blood products ordered in preparation for surgical procedures. We are also currently investigating whether an impact on safety events (ie, reduction in transfusion reactions) can yet be detected. Last, our health care system recently launched a system-wide electronic medical record, and we are eager to see how this will provide us with new methods to monitor and analyze blood administration and utilization data. We look forward to reporting on the expansion of our program and on any clinical outcome improvements gained through avoidance of unnecessary transfusions.
Acknowledgment: The authors thank the leadership within the Center for Clinical Excellence and Supply Chain at BJC HealthCare for their support of this manuscript, as well as all system participants who have contributed to these efforts, especially Mohammad Agha, MD, MHA, current physician leader of the PBM CEC, for his thoughtful edits of this manuscript.
Corresponding author: Audrey A. Gronemeyer, MPH, Center for Clinical Excellence, BJC HealthCare, 8300 Eager Road, Suite 400A, St. Louis, MO 63144; [email protected].
Financial disclosures: None.
1. Carson JL, Grossman BJ, Kleinman S, et al. Red blood cell transfusion: A clinical practice guideline from the AABB*. Ann Intern Med. 2012;157:49-58.
2. Goodnough LT, Levy JH, Murphy MF. Concepts of blood transfusion in adults. Lancet. 2013;381:1845-1854.
3. Hébert PC, Carson JL. Transfusion threshold of 7 g per deciliter—The new normal. N Engl J Med. 2014;371:1459-1461.
4. Gani F, Cerullo M, Ejaz A, et al. Implementation of a blood management program at a tertiary care hospital: Effect on transfusion practices and clinical outcomes among patients undergoing surgery. Ann Surg. 2019;269:1073-1079.
5. Podlasek SJ, Thakkar RN, Rotello LC, et al. Implementing a “why give 2 when 1 will do?” Choosing Wisely campaign. Transfusion. 2016;56:2164.
6. Boral LI, Bernard A, Hjorth T, et al. How do I implement a more restrictive transfusion trigger of hemoglobin level of 7 g/dL at my hospital? Transfusion. 2015;55:937-945.
7. Geissler RG, Kosters C, Franz D, et al. Utilization of blood components in trauma surgery: A single-center, retrospective analysis before and after the implementation of an educative PBM initiative. Transfuse Med Hemother. 2015;42:83-89.
8. Goel R, Cushing MM, Tobian AA. Pediatric patient blood management programs: Not just transfusing little adults. Transfus Med Rev. 2016;30:235-241.
9. Gupta PB, DeMario VM, Amin RM, et al. Patient blood management program improves blood use and clinical outcomes in orthopedic surgery. Anesthesiology. 2018;129;1082-1091.
10. Leahy MF, Roberts H, Mukhtar SA, et al. A pragmatic approach to embedding patient blood management in a tertiary hospital. Transfusion. 2014;54:1133-1145.
11. Leahy MF, Hofmann A, Towler S, et al. Improved outcomes and reduced costs associated with a health-system-wide patient blood management program: A retrospective observational study in four major adult tertiary-care hospitals. Transfusion. 2017;57:1347-1358.
12. Meybohm P, Herrmann E, Steinbicker AU, et al. Patient blood management is associated with a substantial reduction of red blood cell utilization and safe for patient’s outcome: A prospective, multicenter cohort study with a noninferiority design. Ann Surg. 2016;264:203-211.
13. Morgan PN, Coleman PL, Martinez-Garduno CM, et al. Implementation of a patient blood management program in an Australian private hospital orthopedic unit. J Blood Med. 2018;9;83-90.
14. Norgaard A, Stensballe J, de Lichtenberg TH, et al. Three-year follow-up of implementation of evidence-based transfusion practice in a tertiary hospital. Vox Sang. 2017;112:229-239.
15. Meuller MM, Van Remoortel H, Meybohm P, et al. Patient blood management: Recommendations from the 2018 Frankfurt Consensus Conference. JAMA. 2019;321:983-997.
16. Oliver JC, Griffin RL, Hannon T, Marques MB. The success of our patient blood management program depended on an institution-wide change in transfusion practices. Transfusion. 2014;54:2617-2624.
17. Thakkar RN, Lee KH, Ness PM, et al. Relative impact of a patient blood management program on utilization of all three major blood components. Transfusion. 2016;56:2212-2220.
18. Yang WW, Thakkar RN, Gehrie EA, et al. Single-unit transfusions and hemoglobin trigger: relative impact on red cell utilization. Transfusion. 2017;57:1163-1170.
19. Frank SM, Thakkar RN, Podlasek SJ, et al. Implementing a health system-wide patient blood management program with a clinical community approach. Anesthesiology. 2017;127;754-764.
20. Verdecchia NM, Wisniewski MK, Waters JH, et al. Changes in blood product utilization in a seven-hospital system after the implementation of a patient blood management program: A 9-year follow-up. Hematology. 2016;21:490-499.
21. Yazer MH, Waters JH. How do I implement a hospital-based blood management program? Transfusion. 2012;52:1640-1645.
22. BJC HealthCare. Facts and Figures.. BJC HealthCare website. www.bjc.org/About-Us/Facts-Figures. Accessed November 18, 2019.
23. Callum JL, Waters JH, Shaz BH, et al. The AABB recommendations for the Choosing Wisely campaign of the American Board of Internal Medicine. Transfusion. 2014;54:2344-2352.
1. Carson JL, Grossman BJ, Kleinman S, et al. Red blood cell transfusion: A clinical practice guideline from the AABB*. Ann Intern Med. 2012;157:49-58.
2. Goodnough LT, Levy JH, Murphy MF. Concepts of blood transfusion in adults. Lancet. 2013;381:1845-1854.
3. Hébert PC, Carson JL. Transfusion threshold of 7 g per deciliter—The new normal. N Engl J Med. 2014;371:1459-1461.
4. Gani F, Cerullo M, Ejaz A, et al. Implementation of a blood management program at a tertiary care hospital: Effect on transfusion practices and clinical outcomes among patients undergoing surgery. Ann Surg. 2019;269:1073-1079.
5. Podlasek SJ, Thakkar RN, Rotello LC, et al. Implementing a “why give 2 when 1 will do?” Choosing Wisely campaign. Transfusion. 2016;56:2164.
6. Boral LI, Bernard A, Hjorth T, et al. How do I implement a more restrictive transfusion trigger of hemoglobin level of 7 g/dL at my hospital? Transfusion. 2015;55:937-945.
7. Geissler RG, Kosters C, Franz D, et al. Utilization of blood components in trauma surgery: A single-center, retrospective analysis before and after the implementation of an educative PBM initiative. Transfuse Med Hemother. 2015;42:83-89.
8. Goel R, Cushing MM, Tobian AA. Pediatric patient blood management programs: Not just transfusing little adults. Transfus Med Rev. 2016;30:235-241.
9. Gupta PB, DeMario VM, Amin RM, et al. Patient blood management program improves blood use and clinical outcomes in orthopedic surgery. Anesthesiology. 2018;129;1082-1091.
10. Leahy MF, Roberts H, Mukhtar SA, et al. A pragmatic approach to embedding patient blood management in a tertiary hospital. Transfusion. 2014;54:1133-1145.
11. Leahy MF, Hofmann A, Towler S, et al. Improved outcomes and reduced costs associated with a health-system-wide patient blood management program: A retrospective observational study in four major adult tertiary-care hospitals. Transfusion. 2017;57:1347-1358.
12. Meybohm P, Herrmann E, Steinbicker AU, et al. Patient blood management is associated with a substantial reduction of red blood cell utilization and safe for patient’s outcome: A prospective, multicenter cohort study with a noninferiority design. Ann Surg. 2016;264:203-211.
13. Morgan PN, Coleman PL, Martinez-Garduno CM, et al. Implementation of a patient blood management program in an Australian private hospital orthopedic unit. J Blood Med. 2018;9;83-90.
14. Norgaard A, Stensballe J, de Lichtenberg TH, et al. Three-year follow-up of implementation of evidence-based transfusion practice in a tertiary hospital. Vox Sang. 2017;112:229-239.
15. Meuller MM, Van Remoortel H, Meybohm P, et al. Patient blood management: Recommendations from the 2018 Frankfurt Consensus Conference. JAMA. 2019;321:983-997.
16. Oliver JC, Griffin RL, Hannon T, Marques MB. The success of our patient blood management program depended on an institution-wide change in transfusion practices. Transfusion. 2014;54:2617-2624.
17. Thakkar RN, Lee KH, Ness PM, et al. Relative impact of a patient blood management program on utilization of all three major blood components. Transfusion. 2016;56:2212-2220.
18. Yang WW, Thakkar RN, Gehrie EA, et al. Single-unit transfusions and hemoglobin trigger: relative impact on red cell utilization. Transfusion. 2017;57:1163-1170.
19. Frank SM, Thakkar RN, Podlasek SJ, et al. Implementing a health system-wide patient blood management program with a clinical community approach. Anesthesiology. 2017;127;754-764.
20. Verdecchia NM, Wisniewski MK, Waters JH, et al. Changes in blood product utilization in a seven-hospital system after the implementation of a patient blood management program: A 9-year follow-up. Hematology. 2016;21:490-499.
21. Yazer MH, Waters JH. How do I implement a hospital-based blood management program? Transfusion. 2012;52:1640-1645.
22. BJC HealthCare. Facts and Figures.. BJC HealthCare website. www.bjc.org/About-Us/Facts-Figures. Accessed November 18, 2019.
23. Callum JL, Waters JH, Shaz BH, et al. The AABB recommendations for the Choosing Wisely campaign of the American Board of Internal Medicine. Transfusion. 2014;54:2344-2352.
Is anemia due to folate deficiency a myth?
A 46-year-old man who lives in Tacoma, Wash., is seen for fatigue. He has a no significant past medical history. He is not taking any medications. His physical exam is unremarkable. His hemoglobin is 12 gm/dL, hematocrit is 37 gm/dL, mean corpuscular volume (MCV) is 103 fL, and thyroid-stimulating hormone level is 1.2 mU/L.
What workup do you recommend?
A) B12, folate testing
B) Alcohol history, B12, folate testing
C) Alcohol history, B12 testing
I would choose doing a careful alcohol history and vitamin B12 testing.
Dr. Seppä and colleagues looked at all outpatients who had a blood count done over an 8-month period.1 A total of 9,527 blood counts were ordered, and 287 (3%) had macrocytosis.1 Further workup was done for 113 of the patients. The most common cause found for macrocytosis was alcohol abuse, in 74 (65%) of the patients (80% of the men and 36% of the women). In several studies, vitamin B12 deficiency was the cause of macrocytosis in 5%-7% of patients.2,3
In 1978, a study by Davidson and Hamilton looked at 200 consecutive patients with MCVs over 100, and were able to find a cause in 80%.4 Sixteen of these patients had a low B12 level and 10 had a low folate level.
In 1998, the Food and Drug Administration required folic acid fortification of enriched grain products in the United States to help decrease the risk of neural tube defects. Similar fortification efforts were undertaken in Canada. Since 1998, anemia due to folate deficiency has essentially disappeared in individuals who have access to fortified grain products.
Joelson and colleagues looked at data on folate testing from the year prior to fortification of the grain supply (1997) and after (2004).5 They found that, in 1997, 4.8% of tests had a folate level less than 160 ng/mL compared with only 0.6% of tests in 2004.
When a more stringent cutoff for deficiency was used (94 ng/mL) 0.98% of tests were below that level in 1997, and 0.09% in 2004. The mean RBC folate level in 1997 was 420 ng/mL and rose to 697 ng/mL in 2004. Of the patients who did have low folate levels, only a minority had elevated MCVs.
Shojania et al. looked at folate testing in Canada after widespread fortification had started.6 They found that 0.5% of 2,154 serum folate levels were low and 0.7% of 560 red blood cell folate levels were low. Folate deficiency was not the cause of anemia in any of the patients with low folate levels.
Theisen-Toupal and colleagues did a retrospective study looking at folate testing over an 11-year period after fortification.7 The researchers examined the results of 84,187 assessments of folate levels. Forty-seven (0.056%) of the tests found patients with folate deficiency, 166 (0.197%), found patients with low-normal folate levels, 57,411 (68.195%) of tests yielded normal results, and 26,563 (31.552%) of tests found high folate levels. The opinion of the authors was that folate testing should be severely reduced or eliminated. Furthermore, the American Society for Clinical Pathology, as part of the Choosing Wisely campaign, states: “Do not order red blood cell folate levels at all.”8
So what does this all mean? We have been taught to have a reflex response to the evaluation of macrocytosis to test for B12 and folate. Neither of these are particularly common causes of macrocytosis, and in countries where there is grain fortification, folate deficiency is exceedingly uncommon, and should not be tested for early in any diagnostic process.
Dr. Paauw is professor of medicine in the division of general internal medicine at the University of Washington, Seattle, and he serves as third-year medical student clerkship director at the University of Washington. He is a member of the editorial advisory board of Internal Medicine News. Dr. Paauw has no conflicts to disclose. Contact him at [email protected].
References
1. Seppä K et al. Evaluation of macrocytosis by general practitioners. J Stud Alcohol. 1996 Jan;57(1):97-100.
2. Seppä K et al. Blood count and hematologic morphology in nonanemic macrocytosis: Differences between alcohol abuse and pernicious anemia. Alcohol. 1993 Sep-Oct;10(5):343-7.
3. Wymer A, Becker DM. Recognition and evaluation of red blood cell macrocytosis in the primary care setting. J Gen Intern Med. 1990 May-Jun;5(3):192-7.
4. Davidson RJ, Hamilton PJ. High mean red cell volume: Its incidence and significance in routine haematology. J Clin Pathol. 1978 May;31[5]:493-8.
5. Joelson DW, Fiebig EW. Diminished need for folate measurements among indigent populations in the post folic acid supplementation era. Arch Pathol Lab Med. 2007 Mar;131(3):477-80.
6. Shojania AM, von Kuster K. Ordering folate assays is no longer justified for investigation of anemias, in folic acid fortified countries. BMC Res Notes. 2010 Jan 25;3:22. doi: 10.1186/1756-0500-3-22.
7. Theisen-Toupal et al. Low yield of outpatient serum folate testing. JAMA Intern Med. 2014 Oct. doi: 10.1001/jamainternmed.2014.3593.
8. Choosing Wisely: American Society for Clinical Pathology, Oct. 19, 2017. Recommendation.
A 46-year-old man who lives in Tacoma, Wash., is seen for fatigue. He has a no significant past medical history. He is not taking any medications. His physical exam is unremarkable. His hemoglobin is 12 gm/dL, hematocrit is 37 gm/dL, mean corpuscular volume (MCV) is 103 fL, and thyroid-stimulating hormone level is 1.2 mU/L.
What workup do you recommend?
A) B12, folate testing
B) Alcohol history, B12, folate testing
C) Alcohol history, B12 testing
I would choose doing a careful alcohol history and vitamin B12 testing.
Dr. Seppä and colleagues looked at all outpatients who had a blood count done over an 8-month period.1 A total of 9,527 blood counts were ordered, and 287 (3%) had macrocytosis.1 Further workup was done for 113 of the patients. The most common cause found for macrocytosis was alcohol abuse, in 74 (65%) of the patients (80% of the men and 36% of the women). In several studies, vitamin B12 deficiency was the cause of macrocytosis in 5%-7% of patients.2,3
In 1978, a study by Davidson and Hamilton looked at 200 consecutive patients with MCVs over 100, and were able to find a cause in 80%.4 Sixteen of these patients had a low B12 level and 10 had a low folate level.
In 1998, the Food and Drug Administration required folic acid fortification of enriched grain products in the United States to help decrease the risk of neural tube defects. Similar fortification efforts were undertaken in Canada. Since 1998, anemia due to folate deficiency has essentially disappeared in individuals who have access to fortified grain products.
Joelson and colleagues looked at data on folate testing from the year prior to fortification of the grain supply (1997) and after (2004).5 They found that, in 1997, 4.8% of tests had a folate level less than 160 ng/mL compared with only 0.6% of tests in 2004.
When a more stringent cutoff for deficiency was used (94 ng/mL) 0.98% of tests were below that level in 1997, and 0.09% in 2004. The mean RBC folate level in 1997 was 420 ng/mL and rose to 697 ng/mL in 2004. Of the patients who did have low folate levels, only a minority had elevated MCVs.
Shojania et al. looked at folate testing in Canada after widespread fortification had started.6 They found that 0.5% of 2,154 serum folate levels were low and 0.7% of 560 red blood cell folate levels were low. Folate deficiency was not the cause of anemia in any of the patients with low folate levels.
Theisen-Toupal and colleagues did a retrospective study looking at folate testing over an 11-year period after fortification.7 The researchers examined the results of 84,187 assessments of folate levels. Forty-seven (0.056%) of the tests found patients with folate deficiency, 166 (0.197%), found patients with low-normal folate levels, 57,411 (68.195%) of tests yielded normal results, and 26,563 (31.552%) of tests found high folate levels. The opinion of the authors was that folate testing should be severely reduced or eliminated. Furthermore, the American Society for Clinical Pathology, as part of the Choosing Wisely campaign, states: “Do not order red blood cell folate levels at all.”8
So what does this all mean? We have been taught to have a reflex response to the evaluation of macrocytosis to test for B12 and folate. Neither of these are particularly common causes of macrocytosis, and in countries where there is grain fortification, folate deficiency is exceedingly uncommon, and should not be tested for early in any diagnostic process.
Dr. Paauw is professor of medicine in the division of general internal medicine at the University of Washington, Seattle, and he serves as third-year medical student clerkship director at the University of Washington. He is a member of the editorial advisory board of Internal Medicine News. Dr. Paauw has no conflicts to disclose. Contact him at [email protected].
References
1. Seppä K et al. Evaluation of macrocytosis by general practitioners. J Stud Alcohol. 1996 Jan;57(1):97-100.
2. Seppä K et al. Blood count and hematologic morphology in nonanemic macrocytosis: Differences between alcohol abuse and pernicious anemia. Alcohol. 1993 Sep-Oct;10(5):343-7.
3. Wymer A, Becker DM. Recognition and evaluation of red blood cell macrocytosis in the primary care setting. J Gen Intern Med. 1990 May-Jun;5(3):192-7.
4. Davidson RJ, Hamilton PJ. High mean red cell volume: Its incidence and significance in routine haematology. J Clin Pathol. 1978 May;31[5]:493-8.
5. Joelson DW, Fiebig EW. Diminished need for folate measurements among indigent populations in the post folic acid supplementation era. Arch Pathol Lab Med. 2007 Mar;131(3):477-80.
6. Shojania AM, von Kuster K. Ordering folate assays is no longer justified for investigation of anemias, in folic acid fortified countries. BMC Res Notes. 2010 Jan 25;3:22. doi: 10.1186/1756-0500-3-22.
7. Theisen-Toupal et al. Low yield of outpatient serum folate testing. JAMA Intern Med. 2014 Oct. doi: 10.1001/jamainternmed.2014.3593.
8. Choosing Wisely: American Society for Clinical Pathology, Oct. 19, 2017. Recommendation.
A 46-year-old man who lives in Tacoma, Wash., is seen for fatigue. He has a no significant past medical history. He is not taking any medications. His physical exam is unremarkable. His hemoglobin is 12 gm/dL, hematocrit is 37 gm/dL, mean corpuscular volume (MCV) is 103 fL, and thyroid-stimulating hormone level is 1.2 mU/L.
What workup do you recommend?
A) B12, folate testing
B) Alcohol history, B12, folate testing
C) Alcohol history, B12 testing
I would choose doing a careful alcohol history and vitamin B12 testing.
Dr. Seppä and colleagues looked at all outpatients who had a blood count done over an 8-month period.1 A total of 9,527 blood counts were ordered, and 287 (3%) had macrocytosis.1 Further workup was done for 113 of the patients. The most common cause found for macrocytosis was alcohol abuse, in 74 (65%) of the patients (80% of the men and 36% of the women). In several studies, vitamin B12 deficiency was the cause of macrocytosis in 5%-7% of patients.2,3
In 1978, a study by Davidson and Hamilton looked at 200 consecutive patients with MCVs over 100, and were able to find a cause in 80%.4 Sixteen of these patients had a low B12 level and 10 had a low folate level.
In 1998, the Food and Drug Administration required folic acid fortification of enriched grain products in the United States to help decrease the risk of neural tube defects. Similar fortification efforts were undertaken in Canada. Since 1998, anemia due to folate deficiency has essentially disappeared in individuals who have access to fortified grain products.
Joelson and colleagues looked at data on folate testing from the year prior to fortification of the grain supply (1997) and after (2004).5 They found that, in 1997, 4.8% of tests had a folate level less than 160 ng/mL compared with only 0.6% of tests in 2004.
When a more stringent cutoff for deficiency was used (94 ng/mL) 0.98% of tests were below that level in 1997, and 0.09% in 2004. The mean RBC folate level in 1997 was 420 ng/mL and rose to 697 ng/mL in 2004. Of the patients who did have low folate levels, only a minority had elevated MCVs.
Shojania et al. looked at folate testing in Canada after widespread fortification had started.6 They found that 0.5% of 2,154 serum folate levels were low and 0.7% of 560 red blood cell folate levels were low. Folate deficiency was not the cause of anemia in any of the patients with low folate levels.
Theisen-Toupal and colleagues did a retrospective study looking at folate testing over an 11-year period after fortification.7 The researchers examined the results of 84,187 assessments of folate levels. Forty-seven (0.056%) of the tests found patients with folate deficiency, 166 (0.197%), found patients with low-normal folate levels, 57,411 (68.195%) of tests yielded normal results, and 26,563 (31.552%) of tests found high folate levels. The opinion of the authors was that folate testing should be severely reduced or eliminated. Furthermore, the American Society for Clinical Pathology, as part of the Choosing Wisely campaign, states: “Do not order red blood cell folate levels at all.”8
So what does this all mean? We have been taught to have a reflex response to the evaluation of macrocytosis to test for B12 and folate. Neither of these are particularly common causes of macrocytosis, and in countries where there is grain fortification, folate deficiency is exceedingly uncommon, and should not be tested for early in any diagnostic process.
Dr. Paauw is professor of medicine in the division of general internal medicine at the University of Washington, Seattle, and he serves as third-year medical student clerkship director at the University of Washington. He is a member of the editorial advisory board of Internal Medicine News. Dr. Paauw has no conflicts to disclose. Contact him at [email protected].
References
1. Seppä K et al. Evaluation of macrocytosis by general practitioners. J Stud Alcohol. 1996 Jan;57(1):97-100.
2. Seppä K et al. Blood count and hematologic morphology in nonanemic macrocytosis: Differences between alcohol abuse and pernicious anemia. Alcohol. 1993 Sep-Oct;10(5):343-7.
3. Wymer A, Becker DM. Recognition and evaluation of red blood cell macrocytosis in the primary care setting. J Gen Intern Med. 1990 May-Jun;5(3):192-7.
4. Davidson RJ, Hamilton PJ. High mean red cell volume: Its incidence and significance in routine haematology. J Clin Pathol. 1978 May;31[5]:493-8.
5. Joelson DW, Fiebig EW. Diminished need for folate measurements among indigent populations in the post folic acid supplementation era. Arch Pathol Lab Med. 2007 Mar;131(3):477-80.
6. Shojania AM, von Kuster K. Ordering folate assays is no longer justified for investigation of anemias, in folic acid fortified countries. BMC Res Notes. 2010 Jan 25;3:22. doi: 10.1186/1756-0500-3-22.
7. Theisen-Toupal et al. Low yield of outpatient serum folate testing. JAMA Intern Med. 2014 Oct. doi: 10.1001/jamainternmed.2014.3593.
8. Choosing Wisely: American Society for Clinical Pathology, Oct. 19, 2017. Recommendation.
Inflammation, thrombosis biomarkers tied to COVID-19 deaths
Their prospective cohort study of 1150 patients hospitalized with the disease in New York City also revealed a high proportion of racial and ethnic minorities, and confirmed high rates of critical illness and mortality.
“Of particular interest is the finding that over three quarters of critically ill patients required a ventilator and almost one third required renal dialysis support,” Max O’Donnell, MD, MPH, assistant professor of medicine and epidemiology at Columbia University in New York City, said in a press release.
O’Donnell and colleagues published the results of their study online today in The Lancet. It is the largest prospective cohort study published in the United States, they said.
“Although the clinical spectrum of disease has been characterised in reports from China and Italy, until now, detailed understanding of how the virus is affecting critically ill patients in the US has been limited to reports from a small number of cases,” said Natalie Yip, MD, assistant professor of medicine at Columbia University.
In the cohort, drawn from two NewYork-Presbyterian hospitals, the researchers focused on the 257 (22%) patients who required intensive care. When they estimated inflammation through interleukin-6 (IL-6) concentrations and thrombosis through D-dimer concentrations, they found a 10% increased risk for death with every 10% increase of IL-6 (adjusted hazard ratio [aHR], 1.11; 95% confidence interval [CI], 1.02–1.20) or D-dimer concentration (aHR, 1.10; 95% CI, 1.01–1.19).
“The association of mortality with higher concentrations of IL-6 and d-dimer is particularly relevant for two reasons,” write Giacomo Grasselli, from the Fondazione IRCCS Ca’ Granda Ospediale Maggiore Policlinico, and Alberto Zanella, from the University of Milan, Italy, in an accompanying commentary.
“First, it confirms the key pathogenic role played by the activation of systemic inflammation and endothelial-vascular damage in the development of organ dysfunction,” they write. “Second, it provides the rationale for the design of clinical trials for measuring the efficacy of treatment with immunomodulating and anticoagulant drugs.”
Seventeen percent of patients received interleukin receptor antagonists and 26% received corticosteroids, but the authors did not report any data on the effects of these treatments, or any data about anticoagulant therapies administered.
Severe disease common
The study also highlighted a high proportion of ethnic and racial minorities. Sixty-two percent of the critically ill patients were Hispanic or Latinx, 19% Black, 32% White, and 3% Asian.
Their median age was 62 years and 67% were men. Eighty-two percent had at least one chronic illness, most commonly hypertension (63%), followed by diabetes (36%). Forty-six percent were obese.
As of April 28, 2020, 101 (39%) of the critically ill patients had died following a median of 9 days (interquartile range (IQR), 5–15) in the hospital and 94 (37%) remained hospitalized. Of the 203 patients who received invasive mechanical ventilation, 84 (41%) had died.
The poor prognosis of patients requiring ventilation is consistent with data from a report on patients treated in National Health Service intensive care units in England, Wales, and Northern Ireland through May 15. Overall, 11,292 patients with COVID-19 required critical care, and 4855 needed advanced respiratory support. Approximately half of the patients receiving mechanical ventilation had died 30 days after starting critical care.
In the New York study, patients spent an average of 18 days on a ventilator (IQR, 9–28 days). This is a longer period than reported in smaller studies of cases from Washington state, but corresponds with a recent report from Italy, the researchers said.
Remarkably, O’Donnell and colleagues report that almost a third (31%) of critically ill patients developed severe kidney damage and required dialysis.
Mortality was associated with several baseline factors, including older age (aHR, 1.31 [95% CI, 1.09–1.57] per 10-year increase), chronic cardiac disease (aHR, 1.76; 95% CI, 1.08–2·86), and chronic pulmonary disease (aHR, 2.94; 95% CI, 1.48–5.84).
Authors of the New York study reported financial relationships to ICE Neurosystems, ALung Technologies, Baxter, BREETHE, Xenios, Hemovent, Gilead Sciences, Amazon, and Karyopharm Therapeutics. Grasselli reports personal fees from Biotest, Draeger, Fisher & Paykel, Maquet, Merck Sharp & Dohme, and Pfizer, all outside the area of work commented on here. Zanella has disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
Their prospective cohort study of 1150 patients hospitalized with the disease in New York City also revealed a high proportion of racial and ethnic minorities, and confirmed high rates of critical illness and mortality.
“Of particular interest is the finding that over three quarters of critically ill patients required a ventilator and almost one third required renal dialysis support,” Max O’Donnell, MD, MPH, assistant professor of medicine and epidemiology at Columbia University in New York City, said in a press release.
O’Donnell and colleagues published the results of their study online today in The Lancet. It is the largest prospective cohort study published in the United States, they said.
“Although the clinical spectrum of disease has been characterised in reports from China and Italy, until now, detailed understanding of how the virus is affecting critically ill patients in the US has been limited to reports from a small number of cases,” said Natalie Yip, MD, assistant professor of medicine at Columbia University.
In the cohort, drawn from two NewYork-Presbyterian hospitals, the researchers focused on the 257 (22%) patients who required intensive care. When they estimated inflammation through interleukin-6 (IL-6) concentrations and thrombosis through D-dimer concentrations, they found a 10% increased risk for death with every 10% increase of IL-6 (adjusted hazard ratio [aHR], 1.11; 95% confidence interval [CI], 1.02–1.20) or D-dimer concentration (aHR, 1.10; 95% CI, 1.01–1.19).
“The association of mortality with higher concentrations of IL-6 and d-dimer is particularly relevant for two reasons,” write Giacomo Grasselli, from the Fondazione IRCCS Ca’ Granda Ospediale Maggiore Policlinico, and Alberto Zanella, from the University of Milan, Italy, in an accompanying commentary.
“First, it confirms the key pathogenic role played by the activation of systemic inflammation and endothelial-vascular damage in the development of organ dysfunction,” they write. “Second, it provides the rationale for the design of clinical trials for measuring the efficacy of treatment with immunomodulating and anticoagulant drugs.”
Seventeen percent of patients received interleukin receptor antagonists and 26% received corticosteroids, but the authors did not report any data on the effects of these treatments, or any data about anticoagulant therapies administered.
Severe disease common
The study also highlighted a high proportion of ethnic and racial minorities. Sixty-two percent of the critically ill patients were Hispanic or Latinx, 19% Black, 32% White, and 3% Asian.
Their median age was 62 years and 67% were men. Eighty-two percent had at least one chronic illness, most commonly hypertension (63%), followed by diabetes (36%). Forty-six percent were obese.
As of April 28, 2020, 101 (39%) of the critically ill patients had died following a median of 9 days (interquartile range (IQR), 5–15) in the hospital and 94 (37%) remained hospitalized. Of the 203 patients who received invasive mechanical ventilation, 84 (41%) had died.
The poor prognosis of patients requiring ventilation is consistent with data from a report on patients treated in National Health Service intensive care units in England, Wales, and Northern Ireland through May 15. Overall, 11,292 patients with COVID-19 required critical care, and 4855 needed advanced respiratory support. Approximately half of the patients receiving mechanical ventilation had died 30 days after starting critical care.
In the New York study, patients spent an average of 18 days on a ventilator (IQR, 9–28 days). This is a longer period than reported in smaller studies of cases from Washington state, but corresponds with a recent report from Italy, the researchers said.
Remarkably, O’Donnell and colleagues report that almost a third (31%) of critically ill patients developed severe kidney damage and required dialysis.
Mortality was associated with several baseline factors, including older age (aHR, 1.31 [95% CI, 1.09–1.57] per 10-year increase), chronic cardiac disease (aHR, 1.76; 95% CI, 1.08–2·86), and chronic pulmonary disease (aHR, 2.94; 95% CI, 1.48–5.84).
Authors of the New York study reported financial relationships to ICE Neurosystems, ALung Technologies, Baxter, BREETHE, Xenios, Hemovent, Gilead Sciences, Amazon, and Karyopharm Therapeutics. Grasselli reports personal fees from Biotest, Draeger, Fisher & Paykel, Maquet, Merck Sharp & Dohme, and Pfizer, all outside the area of work commented on here. Zanella has disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
Their prospective cohort study of 1150 patients hospitalized with the disease in New York City also revealed a high proportion of racial and ethnic minorities, and confirmed high rates of critical illness and mortality.
“Of particular interest is the finding that over three quarters of critically ill patients required a ventilator and almost one third required renal dialysis support,” Max O’Donnell, MD, MPH, assistant professor of medicine and epidemiology at Columbia University in New York City, said in a press release.
O’Donnell and colleagues published the results of their study online today in The Lancet. It is the largest prospective cohort study published in the United States, they said.
“Although the clinical spectrum of disease has been characterised in reports from China and Italy, until now, detailed understanding of how the virus is affecting critically ill patients in the US has been limited to reports from a small number of cases,” said Natalie Yip, MD, assistant professor of medicine at Columbia University.
In the cohort, drawn from two NewYork-Presbyterian hospitals, the researchers focused on the 257 (22%) patients who required intensive care. When they estimated inflammation through interleukin-6 (IL-6) concentrations and thrombosis through D-dimer concentrations, they found a 10% increased risk for death with every 10% increase of IL-6 (adjusted hazard ratio [aHR], 1.11; 95% confidence interval [CI], 1.02–1.20) or D-dimer concentration (aHR, 1.10; 95% CI, 1.01–1.19).
“The association of mortality with higher concentrations of IL-6 and d-dimer is particularly relevant for two reasons,” write Giacomo Grasselli, from the Fondazione IRCCS Ca’ Granda Ospediale Maggiore Policlinico, and Alberto Zanella, from the University of Milan, Italy, in an accompanying commentary.
“First, it confirms the key pathogenic role played by the activation of systemic inflammation and endothelial-vascular damage in the development of organ dysfunction,” they write. “Second, it provides the rationale for the design of clinical trials for measuring the efficacy of treatment with immunomodulating and anticoagulant drugs.”
Seventeen percent of patients received interleukin receptor antagonists and 26% received corticosteroids, but the authors did not report any data on the effects of these treatments, or any data about anticoagulant therapies administered.
Severe disease common
The study also highlighted a high proportion of ethnic and racial minorities. Sixty-two percent of the critically ill patients were Hispanic or Latinx, 19% Black, 32% White, and 3% Asian.
Their median age was 62 years and 67% were men. Eighty-two percent had at least one chronic illness, most commonly hypertension (63%), followed by diabetes (36%). Forty-six percent were obese.
As of April 28, 2020, 101 (39%) of the critically ill patients had died following a median of 9 days (interquartile range (IQR), 5–15) in the hospital and 94 (37%) remained hospitalized. Of the 203 patients who received invasive mechanical ventilation, 84 (41%) had died.
The poor prognosis of patients requiring ventilation is consistent with data from a report on patients treated in National Health Service intensive care units in England, Wales, and Northern Ireland through May 15. Overall, 11,292 patients with COVID-19 required critical care, and 4855 needed advanced respiratory support. Approximately half of the patients receiving mechanical ventilation had died 30 days after starting critical care.
In the New York study, patients spent an average of 18 days on a ventilator (IQR, 9–28 days). This is a longer period than reported in smaller studies of cases from Washington state, but corresponds with a recent report from Italy, the researchers said.
Remarkably, O’Donnell and colleagues report that almost a third (31%) of critically ill patients developed severe kidney damage and required dialysis.
Mortality was associated with several baseline factors, including older age (aHR, 1.31 [95% CI, 1.09–1.57] per 10-year increase), chronic cardiac disease (aHR, 1.76; 95% CI, 1.08–2·86), and chronic pulmonary disease (aHR, 2.94; 95% CI, 1.48–5.84).
Authors of the New York study reported financial relationships to ICE Neurosystems, ALung Technologies, Baxter, BREETHE, Xenios, Hemovent, Gilead Sciences, Amazon, and Karyopharm Therapeutics. Grasselli reports personal fees from Biotest, Draeger, Fisher & Paykel, Maquet, Merck Sharp & Dohme, and Pfizer, all outside the area of work commented on here. Zanella has disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
DOACs linked to lower fracture risk versus warfarin in AFib patients
results of a recent population-based cohort study show.
The choice of direct oral anticoagulant (DOAC) didn’t appear to have an impact, as each individual agent yielded a substantially lower risk of fracture versus the vitamin K antagonist, with risk reductions ranging from 38% to 48%, according to the study authors.
This is one of the latest reports to suggest DOACs could have an edge over warfarin for preventing fractures, providing new evidence that “may help inform the benefit risk assessment” when it comes to choosing an anticoagulant for a patient with atrial fibrillation (AFib) in the clinic, wrote the authors, led by Wallis C.Y. Lau, PhD, with the University College London.
“There exists a compelling case for evaluating whether the risk for osteoporotic fractures should be considered at the point of prescribing an oral anticoagulant to minimize fracture risk,” Dr. Lau and coauthors wrote in a report on the study that appears in Annals of Internal Medicine.
The case is especially compelling since fracture risk is “often neglected” when choosing an anticoagulant, the authors wrote. Surgeries to treat fracture are difficult because of the need for perioperative management of anticoagulation as “a balance between the risk for stroke and excessive bleeding must be achieved,” they added.
Based on these data, physicians should strongly consider DOACs as an alternative to vitamin K antagonists to reduce the risk of osteoporosis over the long term in patients with AFib, according to Victor Lawrence Roberts, MD, a Florida endocrinologist.
“Osteoporosis takes years, sometimes decades to develop, and if you then overlay warfarin on top of a readily evolving metabolic bone disease, you probably accelerate that process, said Dr. Roberts, professor of internal medicine at the University of Central Florida, Orlando, and editorial advisory board member of Internal Medicine News.
There’s a considerable amount of concerning preclinical data that warfarin could increase osteoporotic fracture risk. Of note, vitamin K antagonists modulate osteocalcin, a calcium-binding bone matrix protein, Dr. Roberts said.
“Osteocalcin is important for bone metabolism and health, and inhibiting osteocalcin will inhibit the ability to have a healthy bone matrix,” he explained.
The impact of anticoagulants on fracture risk is particularly relevant to patients with AFib, according to Dr. Lau and colleagues, who referenced one 2017 report showing a higher incidence of hip fracture among AFib patients versus those without AFib.
In their more recent study, Dr. Lau and colleagues reviewed electronic health records in a Hong Kong database for 23,515 older adults with a new diagnosis of AFib who received a new prescription of warfarin or DOACs including apixaban, dabigatran, or rivaroxaban.
DOAC use was consistently associated with a lower risk of osteoporotic fractures versus warfarin, regardless of the DOAC considered. The hazard ratios were 0.62 (95% confidence interval, 0.41-0.94) for apixaban, 0.65 (95% CI, 0.49-0.86) for dabigatran, and 0.52 (95% CI, 0.37-0.73) for rivaroxaban versus warfarin, the report showed.
Head-to-head comparisons between DOACS didn’t yield any statistically significant differences, though the analyses were underpowered in this respect, according to the investigators.
“This study can only rule out more than a twofold higher or a 50% lower relative risk for osteoporotic fractures between individual DOACs,” they wrote. “However, any absolute risk differences were small and would likely be of minor clinical significance.”
The reduced risk of fracture for DOACs versus warfarin was consistent in men and women with AFib, suggesting that women may particularly benefit from DOACs, given that they have a higher risk of fracture than men, the investigators added.
The results of this study suggest yet another benefit of DOACs over warfarin in patients with AFib, according to internist Noel Deep, MD, who is the chief medical officer of Aspirus Langlade Hospital in Antigo, Wisconsin.
“The lower risk of osteoporotic fractures with DOACS, in addition to other advantages such as lower risk of intracranial bleeding, once- or twice-daily consistent dosing, no dietary restrictions, and no blood tests to regulate the dose might be another reason that physicians may favor them over warfarin in older individuals requiring anticoagulation,” Dr. Deep said in an interview.
Results of this and several other recent studies may help in recommending DOACs to internal medicine patients who have a diagnosis of AFib requiring anticoagulation, according to Dr. Deep, who is also a physician at Aspirus Antigo Clinic and a member of Internal Medicine News’ editorial advisory board. These include a 2019 U.S.-based study of more than 167,000 patients with AFib (JAMA Intern Med. 2019;180[2]:245‐253) showing that use of DOACs, particularly apixaban, were linked to lower fracture risk versus warfarin use. Similarly, a Danish national registry study also published in 2019 showed that the absolute risk of osteoporotic fractures was low overall and significantly lower in patients who received DOACs (J Am Coll Cardiol. 2019;74[17]:2150-2158).
Funding for the study came from the University of Hong Kong and University College London Strategic Planning Fund. The study authors reported disclosures related to Bayer, Bristol-Myers Squibb, Pfizer, Janssen, Amgen, Takeda, IQVIA, and others.
SOURCE: Lau WCY et al. Ann Intern Med. 2020 May 18. doi: 10.7326/M19-3671.
results of a recent population-based cohort study show.
The choice of direct oral anticoagulant (DOAC) didn’t appear to have an impact, as each individual agent yielded a substantially lower risk of fracture versus the vitamin K antagonist, with risk reductions ranging from 38% to 48%, according to the study authors.
This is one of the latest reports to suggest DOACs could have an edge over warfarin for preventing fractures, providing new evidence that “may help inform the benefit risk assessment” when it comes to choosing an anticoagulant for a patient with atrial fibrillation (AFib) in the clinic, wrote the authors, led by Wallis C.Y. Lau, PhD, with the University College London.
“There exists a compelling case for evaluating whether the risk for osteoporotic fractures should be considered at the point of prescribing an oral anticoagulant to minimize fracture risk,” Dr. Lau and coauthors wrote in a report on the study that appears in Annals of Internal Medicine.
The case is especially compelling since fracture risk is “often neglected” when choosing an anticoagulant, the authors wrote. Surgeries to treat fracture are difficult because of the need for perioperative management of anticoagulation as “a balance between the risk for stroke and excessive bleeding must be achieved,” they added.
Based on these data, physicians should strongly consider DOACs as an alternative to vitamin K antagonists to reduce the risk of osteoporosis over the long term in patients with AFib, according to Victor Lawrence Roberts, MD, a Florida endocrinologist.
“Osteoporosis takes years, sometimes decades to develop, and if you then overlay warfarin on top of a readily evolving metabolic bone disease, you probably accelerate that process, said Dr. Roberts, professor of internal medicine at the University of Central Florida, Orlando, and editorial advisory board member of Internal Medicine News.
There’s a considerable amount of concerning preclinical data that warfarin could increase osteoporotic fracture risk. Of note, vitamin K antagonists modulate osteocalcin, a calcium-binding bone matrix protein, Dr. Roberts said.
“Osteocalcin is important for bone metabolism and health, and inhibiting osteocalcin will inhibit the ability to have a healthy bone matrix,” he explained.
The impact of anticoagulants on fracture risk is particularly relevant to patients with AFib, according to Dr. Lau and colleagues, who referenced one 2017 report showing a higher incidence of hip fracture among AFib patients versus those without AFib.
In their more recent study, Dr. Lau and colleagues reviewed electronic health records in a Hong Kong database for 23,515 older adults with a new diagnosis of AFib who received a new prescription of warfarin or DOACs including apixaban, dabigatran, or rivaroxaban.
DOAC use was consistently associated with a lower risk of osteoporotic fractures versus warfarin, regardless of the DOAC considered. The hazard ratios were 0.62 (95% confidence interval, 0.41-0.94) for apixaban, 0.65 (95% CI, 0.49-0.86) for dabigatran, and 0.52 (95% CI, 0.37-0.73) for rivaroxaban versus warfarin, the report showed.
Head-to-head comparisons between DOACS didn’t yield any statistically significant differences, though the analyses were underpowered in this respect, according to the investigators.
“This study can only rule out more than a twofold higher or a 50% lower relative risk for osteoporotic fractures between individual DOACs,” they wrote. “However, any absolute risk differences were small and would likely be of minor clinical significance.”
The reduced risk of fracture for DOACs versus warfarin was consistent in men and women with AFib, suggesting that women may particularly benefit from DOACs, given that they have a higher risk of fracture than men, the investigators added.
The results of this study suggest yet another benefit of DOACs over warfarin in patients with AFib, according to internist Noel Deep, MD, who is the chief medical officer of Aspirus Langlade Hospital in Antigo, Wisconsin.
“The lower risk of osteoporotic fractures with DOACS, in addition to other advantages such as lower risk of intracranial bleeding, once- or twice-daily consistent dosing, no dietary restrictions, and no blood tests to regulate the dose might be another reason that physicians may favor them over warfarin in older individuals requiring anticoagulation,” Dr. Deep said in an interview.
Results of this and several other recent studies may help in recommending DOACs to internal medicine patients who have a diagnosis of AFib requiring anticoagulation, according to Dr. Deep, who is also a physician at Aspirus Antigo Clinic and a member of Internal Medicine News’ editorial advisory board. These include a 2019 U.S.-based study of more than 167,000 patients with AFib (JAMA Intern Med. 2019;180[2]:245‐253) showing that use of DOACs, particularly apixaban, were linked to lower fracture risk versus warfarin use. Similarly, a Danish national registry study also published in 2019 showed that the absolute risk of osteoporotic fractures was low overall and significantly lower in patients who received DOACs (J Am Coll Cardiol. 2019;74[17]:2150-2158).
Funding for the study came from the University of Hong Kong and University College London Strategic Planning Fund. The study authors reported disclosures related to Bayer, Bristol-Myers Squibb, Pfizer, Janssen, Amgen, Takeda, IQVIA, and others.
SOURCE: Lau WCY et al. Ann Intern Med. 2020 May 18. doi: 10.7326/M19-3671.
results of a recent population-based cohort study show.
The choice of direct oral anticoagulant (DOAC) didn’t appear to have an impact, as each individual agent yielded a substantially lower risk of fracture versus the vitamin K antagonist, with risk reductions ranging from 38% to 48%, according to the study authors.
This is one of the latest reports to suggest DOACs could have an edge over warfarin for preventing fractures, providing new evidence that “may help inform the benefit risk assessment” when it comes to choosing an anticoagulant for a patient with atrial fibrillation (AFib) in the clinic, wrote the authors, led by Wallis C.Y. Lau, PhD, with the University College London.
“There exists a compelling case for evaluating whether the risk for osteoporotic fractures should be considered at the point of prescribing an oral anticoagulant to minimize fracture risk,” Dr. Lau and coauthors wrote in a report on the study that appears in Annals of Internal Medicine.
The case is especially compelling since fracture risk is “often neglected” when choosing an anticoagulant, the authors wrote. Surgeries to treat fracture are difficult because of the need for perioperative management of anticoagulation as “a balance between the risk for stroke and excessive bleeding must be achieved,” they added.
Based on these data, physicians should strongly consider DOACs as an alternative to vitamin K antagonists to reduce the risk of osteoporosis over the long term in patients with AFib, according to Victor Lawrence Roberts, MD, a Florida endocrinologist.
“Osteoporosis takes years, sometimes decades to develop, and if you then overlay warfarin on top of a readily evolving metabolic bone disease, you probably accelerate that process, said Dr. Roberts, professor of internal medicine at the University of Central Florida, Orlando, and editorial advisory board member of Internal Medicine News.
There’s a considerable amount of concerning preclinical data that warfarin could increase osteoporotic fracture risk. Of note, vitamin K antagonists modulate osteocalcin, a calcium-binding bone matrix protein, Dr. Roberts said.
“Osteocalcin is important for bone metabolism and health, and inhibiting osteocalcin will inhibit the ability to have a healthy bone matrix,” he explained.
The impact of anticoagulants on fracture risk is particularly relevant to patients with AFib, according to Dr. Lau and colleagues, who referenced one 2017 report showing a higher incidence of hip fracture among AFib patients versus those without AFib.
In their more recent study, Dr. Lau and colleagues reviewed electronic health records in a Hong Kong database for 23,515 older adults with a new diagnosis of AFib who received a new prescription of warfarin or DOACs including apixaban, dabigatran, or rivaroxaban.
DOAC use was consistently associated with a lower risk of osteoporotic fractures versus warfarin, regardless of the DOAC considered. The hazard ratios were 0.62 (95% confidence interval, 0.41-0.94) for apixaban, 0.65 (95% CI, 0.49-0.86) for dabigatran, and 0.52 (95% CI, 0.37-0.73) for rivaroxaban versus warfarin, the report showed.
Head-to-head comparisons between DOACS didn’t yield any statistically significant differences, though the analyses were underpowered in this respect, according to the investigators.
“This study can only rule out more than a twofold higher or a 50% lower relative risk for osteoporotic fractures between individual DOACs,” they wrote. “However, any absolute risk differences were small and would likely be of minor clinical significance.”
The reduced risk of fracture for DOACs versus warfarin was consistent in men and women with AFib, suggesting that women may particularly benefit from DOACs, given that they have a higher risk of fracture than men, the investigators added.
The results of this study suggest yet another benefit of DOACs over warfarin in patients with AFib, according to internist Noel Deep, MD, who is the chief medical officer of Aspirus Langlade Hospital in Antigo, Wisconsin.
“The lower risk of osteoporotic fractures with DOACS, in addition to other advantages such as lower risk of intracranial bleeding, once- or twice-daily consistent dosing, no dietary restrictions, and no blood tests to regulate the dose might be another reason that physicians may favor them over warfarin in older individuals requiring anticoagulation,” Dr. Deep said in an interview.
Results of this and several other recent studies may help in recommending DOACs to internal medicine patients who have a diagnosis of AFib requiring anticoagulation, according to Dr. Deep, who is also a physician at Aspirus Antigo Clinic and a member of Internal Medicine News’ editorial advisory board. These include a 2019 U.S.-based study of more than 167,000 patients with AFib (JAMA Intern Med. 2019;180[2]:245‐253) showing that use of DOACs, particularly apixaban, were linked to lower fracture risk versus warfarin use. Similarly, a Danish national registry study also published in 2019 showed that the absolute risk of osteoporotic fractures was low overall and significantly lower in patients who received DOACs (J Am Coll Cardiol. 2019;74[17]:2150-2158).
Funding for the study came from the University of Hong Kong and University College London Strategic Planning Fund. The study authors reported disclosures related to Bayer, Bristol-Myers Squibb, Pfizer, Janssen, Amgen, Takeda, IQVIA, and others.
SOURCE: Lau WCY et al. Ann Intern Med. 2020 May 18. doi: 10.7326/M19-3671.
FROM ANNALS OF INTERNAL MEDICINE
Universal CAR-T therapy produces CRs in relapsed/refractory T-ALL
according to initial findings from an ongoing study.
The first five patients enrolled in this first-in-human study received conditioning and an infusion of the premanufactured CD7-targeted CAR T-cell therapy, TruUCAR GC027.
All five patients achieved a complete remission (CR) or CR with incomplete count recovery (CRi), although one patient had a morphological relapse at 1 month.
Xinxin Wang, PhD, reported these results at the AACR Virtual Meeting I. Dr. Wang is employed by Gracell Biotechnologies in Shanghai, China, which is the company developing TruUCAR GC027.
The CAR T-cell therapy is manufactured using lentivirus and leukopaks from HLA-mismatched healthy donors, according to Dr. Wang. TruUCAR GC027 contains second-generation CAR T cells with genomic disruption of TCR-alpha and CD7 to help prevent graft-versus-host disease and fratricide.
TruUCAR GC027 was previously shown to expand and have antileukemic activity in a murine model, Dr. Wang noted.
Patients and treatment
The five patients in the phase 1 study had a median age of 24 years (range, 19 to 38 years). They had heavily pretreated T-ALL, with a median of 5 prior lines of therapy (range, 1-9). Baseline bone marrow tumor burden ranged from 4% to 80.2% (median, 38.2%).
None of the patients received prior allogeneic hematopoietic stem cell transplant.
All patients received a preconditioning chemotherapy regimen. One patient received TruUCAR GC027 at dose level 1 (6 x 106 cells/kg), three patients received dose level 2 (1 x 107 cells/kg), and one patient received dose level 3 (1.5 x 107 cells/kg) – each as a single infusion.
Expansion, response, and safety
“GC027 expansion, analyzed by flow [cytometry] was observed in most of the patients treated,” Dr. Wang said. “We started to see GC027 in the peripheral blood as early as day 5, with peaks around day 7-14.”
All five patients had a CR or CRi at the first postinfusion evaluation, which occurred at day 14 in four of the five patients. Four patients also achieved minimum residual disease (MRD) negativity by 1 month of follow-up and remained in MRD-negative CR at the February 6, 2020, data cutoff.
One patient achieved MRD-positive CR at day 14 but experienced morphological relapse at 1 month.
In the four patients with MRD-negative CR at 1 month, cellular expansion was observed as early as day 5 and continued for 2 weeks, but the patient who relapsed at day 29 showed no cellular expansion on flow cytometry, Dr. Wang said.
However, by a more sensitive quantitative polymerase chain reaction analysis, cellular expansion was observed in all five patients starting as early as day 1 after infusion, although the patient who relapsed had the shortest duration of expansion.
All patients developed cytokine release syndrome (CRS). Four patients experienced grade 3 CRS, and one experienced grade 4 CRS.
“The CRS was manageable and reversible,” Dr. Wang said, adding that none of the patients experienced neurotoxicity or graft-versus-host disease.
Prolonged cytopenia occurred in four patients, including one grade 1 case, two grade 3 cases, and one grade 4 case. Grade 3 pulmonary infections occurred in three patients, and grade 3 neutropenia occurred in all five patients.
‘Very impressive’ early results
Dr. Wang said the responses observed in this trial are notable because T-ALL constitutes 20%-25% of all adult ALL and 12%-15% of all pediatric ALL. T-ALL is highly aggressive, with event-free and overall survival of less than 25% in the relapsed setting. Dr. Wang noted that, despite the high unmet medical need and lack of treatment options for T-ALL, the development of novel immunotherapies has lagged.
One challenge is that T-ALL and normal T cells share common surface antigens, so targeted therapies for T-ALL will also target normal T cells. Another challenge is the potential contamination by malignant cells in autologous T-cell products, Dr. Wang said, noting that this can be avoided with universal CAR T cells.
Further, CD7 is a good target for T-ALL because it is expressed in more than 95% of T-ALL patients, she added.
“[TruUCAR GC027] demonstrated a very promising early response rate ... and showed a manageable toxicity profile at all three dose levels,” Dr. Wang said in closing, noting that further evaluation is warranted.
Indeed, the results of this next-generation CAR T-cell trial are “very impressive,” said invited discussant Yvonne Y. Chen, PhD, of the University of California, Los Angeles.
There have been concerns that “off-the-shelf” CAR T-cell products like TruUCAR GC027 might be limited by factors such as a reduced level of CAR T-cell persistence and therefore reduced efficacy leading to a need for repeat dosing, Dr. Chen noted. However, Dr. Wang and her colleagues showed a 100% CR/CRi rate with a single dose of CAR T cells and without graft-versus-host disease or neurotoxicity, Dr. Chen emphasized.
“I think it’s also important to note, however, that there’s quite a high incidence rate of grade 3 or higher toxicities, including CRS,” Dr. Chen said. “I suspect this may have something to do with the fairly high dosing levels used in this trial.”
The “big question,” however, is durability of the response, Dr. Chen said. “And this is something that the field will really watch as this trial progresses beyond the 7-month monitoring period ... reported today.”
Dr. Wang is an employee of Gracell Biotechnologies. Dr. Chen is cofounder of Kalthera Therapeutics and a scientific adviser for Gritstone Oncology and Notch Therapeutics.
SOURCE: Wang X et al. AACR 2020, Abstract CT052.
according to initial findings from an ongoing study.
The first five patients enrolled in this first-in-human study received conditioning and an infusion of the premanufactured CD7-targeted CAR T-cell therapy, TruUCAR GC027.
All five patients achieved a complete remission (CR) or CR with incomplete count recovery (CRi), although one patient had a morphological relapse at 1 month.
Xinxin Wang, PhD, reported these results at the AACR Virtual Meeting I. Dr. Wang is employed by Gracell Biotechnologies in Shanghai, China, which is the company developing TruUCAR GC027.
The CAR T-cell therapy is manufactured using lentivirus and leukopaks from HLA-mismatched healthy donors, according to Dr. Wang. TruUCAR GC027 contains second-generation CAR T cells with genomic disruption of TCR-alpha and CD7 to help prevent graft-versus-host disease and fratricide.
TruUCAR GC027 was previously shown to expand and have antileukemic activity in a murine model, Dr. Wang noted.
Patients and treatment
The five patients in the phase 1 study had a median age of 24 years (range, 19 to 38 years). They had heavily pretreated T-ALL, with a median of 5 prior lines of therapy (range, 1-9). Baseline bone marrow tumor burden ranged from 4% to 80.2% (median, 38.2%).
None of the patients received prior allogeneic hematopoietic stem cell transplant.
All patients received a preconditioning chemotherapy regimen. One patient received TruUCAR GC027 at dose level 1 (6 x 106 cells/kg), three patients received dose level 2 (1 x 107 cells/kg), and one patient received dose level 3 (1.5 x 107 cells/kg) – each as a single infusion.
Expansion, response, and safety
“GC027 expansion, analyzed by flow [cytometry] was observed in most of the patients treated,” Dr. Wang said. “We started to see GC027 in the peripheral blood as early as day 5, with peaks around day 7-14.”
All five patients had a CR or CRi at the first postinfusion evaluation, which occurred at day 14 in four of the five patients. Four patients also achieved minimum residual disease (MRD) negativity by 1 month of follow-up and remained in MRD-negative CR at the February 6, 2020, data cutoff.
One patient achieved MRD-positive CR at day 14 but experienced morphological relapse at 1 month.
In the four patients with MRD-negative CR at 1 month, cellular expansion was observed as early as day 5 and continued for 2 weeks, but the patient who relapsed at day 29 showed no cellular expansion on flow cytometry, Dr. Wang said.
However, by a more sensitive quantitative polymerase chain reaction analysis, cellular expansion was observed in all five patients starting as early as day 1 after infusion, although the patient who relapsed had the shortest duration of expansion.
All patients developed cytokine release syndrome (CRS). Four patients experienced grade 3 CRS, and one experienced grade 4 CRS.
“The CRS was manageable and reversible,” Dr. Wang said, adding that none of the patients experienced neurotoxicity or graft-versus-host disease.
Prolonged cytopenia occurred in four patients, including one grade 1 case, two grade 3 cases, and one grade 4 case. Grade 3 pulmonary infections occurred in three patients, and grade 3 neutropenia occurred in all five patients.
‘Very impressive’ early results
Dr. Wang said the responses observed in this trial are notable because T-ALL constitutes 20%-25% of all adult ALL and 12%-15% of all pediatric ALL. T-ALL is highly aggressive, with event-free and overall survival of less than 25% in the relapsed setting. Dr. Wang noted that, despite the high unmet medical need and lack of treatment options for T-ALL, the development of novel immunotherapies has lagged.
One challenge is that T-ALL and normal T cells share common surface antigens, so targeted therapies for T-ALL will also target normal T cells. Another challenge is the potential contamination by malignant cells in autologous T-cell products, Dr. Wang said, noting that this can be avoided with universal CAR T cells.
Further, CD7 is a good target for T-ALL because it is expressed in more than 95% of T-ALL patients, she added.
“[TruUCAR GC027] demonstrated a very promising early response rate ... and showed a manageable toxicity profile at all three dose levels,” Dr. Wang said in closing, noting that further evaluation is warranted.
Indeed, the results of this next-generation CAR T-cell trial are “very impressive,” said invited discussant Yvonne Y. Chen, PhD, of the University of California, Los Angeles.
There have been concerns that “off-the-shelf” CAR T-cell products like TruUCAR GC027 might be limited by factors such as a reduced level of CAR T-cell persistence and therefore reduced efficacy leading to a need for repeat dosing, Dr. Chen noted. However, Dr. Wang and her colleagues showed a 100% CR/CRi rate with a single dose of CAR T cells and without graft-versus-host disease or neurotoxicity, Dr. Chen emphasized.
“I think it’s also important to note, however, that there’s quite a high incidence rate of grade 3 or higher toxicities, including CRS,” Dr. Chen said. “I suspect this may have something to do with the fairly high dosing levels used in this trial.”
The “big question,” however, is durability of the response, Dr. Chen said. “And this is something that the field will really watch as this trial progresses beyond the 7-month monitoring period ... reported today.”
Dr. Wang is an employee of Gracell Biotechnologies. Dr. Chen is cofounder of Kalthera Therapeutics and a scientific adviser for Gritstone Oncology and Notch Therapeutics.
SOURCE: Wang X et al. AACR 2020, Abstract CT052.
according to initial findings from an ongoing study.
The first five patients enrolled in this first-in-human study received conditioning and an infusion of the premanufactured CD7-targeted CAR T-cell therapy, TruUCAR GC027.
All five patients achieved a complete remission (CR) or CR with incomplete count recovery (CRi), although one patient had a morphological relapse at 1 month.
Xinxin Wang, PhD, reported these results at the AACR Virtual Meeting I. Dr. Wang is employed by Gracell Biotechnologies in Shanghai, China, which is the company developing TruUCAR GC027.
The CAR T-cell therapy is manufactured using lentivirus and leukopaks from HLA-mismatched healthy donors, according to Dr. Wang. TruUCAR GC027 contains second-generation CAR T cells with genomic disruption of TCR-alpha and CD7 to help prevent graft-versus-host disease and fratricide.
TruUCAR GC027 was previously shown to expand and have antileukemic activity in a murine model, Dr. Wang noted.
Patients and treatment
The five patients in the phase 1 study had a median age of 24 years (range, 19 to 38 years). They had heavily pretreated T-ALL, with a median of 5 prior lines of therapy (range, 1-9). Baseline bone marrow tumor burden ranged from 4% to 80.2% (median, 38.2%).
None of the patients received prior allogeneic hematopoietic stem cell transplant.
All patients received a preconditioning chemotherapy regimen. One patient received TruUCAR GC027 at dose level 1 (6 x 106 cells/kg), three patients received dose level 2 (1 x 107 cells/kg), and one patient received dose level 3 (1.5 x 107 cells/kg) – each as a single infusion.
Expansion, response, and safety
“GC027 expansion, analyzed by flow [cytometry] was observed in most of the patients treated,” Dr. Wang said. “We started to see GC027 in the peripheral blood as early as day 5, with peaks around day 7-14.”
All five patients had a CR or CRi at the first postinfusion evaluation, which occurred at day 14 in four of the five patients. Four patients also achieved minimum residual disease (MRD) negativity by 1 month of follow-up and remained in MRD-negative CR at the February 6, 2020, data cutoff.
One patient achieved MRD-positive CR at day 14 but experienced morphological relapse at 1 month.
In the four patients with MRD-negative CR at 1 month, cellular expansion was observed as early as day 5 and continued for 2 weeks, but the patient who relapsed at day 29 showed no cellular expansion on flow cytometry, Dr. Wang said.
However, by a more sensitive quantitative polymerase chain reaction analysis, cellular expansion was observed in all five patients starting as early as day 1 after infusion, although the patient who relapsed had the shortest duration of expansion.
All patients developed cytokine release syndrome (CRS). Four patients experienced grade 3 CRS, and one experienced grade 4 CRS.
“The CRS was manageable and reversible,” Dr. Wang said, adding that none of the patients experienced neurotoxicity or graft-versus-host disease.
Prolonged cytopenia occurred in four patients, including one grade 1 case, two grade 3 cases, and one grade 4 case. Grade 3 pulmonary infections occurred in three patients, and grade 3 neutropenia occurred in all five patients.
‘Very impressive’ early results
Dr. Wang said the responses observed in this trial are notable because T-ALL constitutes 20%-25% of all adult ALL and 12%-15% of all pediatric ALL. T-ALL is highly aggressive, with event-free and overall survival of less than 25% in the relapsed setting. Dr. Wang noted that, despite the high unmet medical need and lack of treatment options for T-ALL, the development of novel immunotherapies has lagged.
One challenge is that T-ALL and normal T cells share common surface antigens, so targeted therapies for T-ALL will also target normal T cells. Another challenge is the potential contamination by malignant cells in autologous T-cell products, Dr. Wang said, noting that this can be avoided with universal CAR T cells.
Further, CD7 is a good target for T-ALL because it is expressed in more than 95% of T-ALL patients, she added.
“[TruUCAR GC027] demonstrated a very promising early response rate ... and showed a manageable toxicity profile at all three dose levels,” Dr. Wang said in closing, noting that further evaluation is warranted.
Indeed, the results of this next-generation CAR T-cell trial are “very impressive,” said invited discussant Yvonne Y. Chen, PhD, of the University of California, Los Angeles.
There have been concerns that “off-the-shelf” CAR T-cell products like TruUCAR GC027 might be limited by factors such as a reduced level of CAR T-cell persistence and therefore reduced efficacy leading to a need for repeat dosing, Dr. Chen noted. However, Dr. Wang and her colleagues showed a 100% CR/CRi rate with a single dose of CAR T cells and without graft-versus-host disease or neurotoxicity, Dr. Chen emphasized.
“I think it’s also important to note, however, that there’s quite a high incidence rate of grade 3 or higher toxicities, including CRS,” Dr. Chen said. “I suspect this may have something to do with the fairly high dosing levels used in this trial.”
The “big question,” however, is durability of the response, Dr. Chen said. “And this is something that the field will really watch as this trial progresses beyond the 7-month monitoring period ... reported today.”
Dr. Wang is an employee of Gracell Biotechnologies. Dr. Chen is cofounder of Kalthera Therapeutics and a scientific adviser for Gritstone Oncology and Notch Therapeutics.
SOURCE: Wang X et al. AACR 2020, Abstract CT052.
FROM AACR 2020
Secondary acute lymphoblastic leukemia more lethal than de novo
The application of improved chemotherapy regimens and novel chemotherapy for acute lymphoblastic leukemia (ALL) has increased the complete remission rate to 85%-90%, however, secondary ALL is common, and the prolonged long-term survival rate is only 30%-50% among ALL patients.
Favorable outcomes decrease with increasing age, and overall survival is greater for adult patients with de novo ALL, compared with patients with secondary ALL, according to the Jiansheng Zhong of the department of hematology, Guangzhou Red Cross Hospital, Jinan University, Guangzhou, Guangdong, China, and colleagues in a new study published online in Clinical Lymphoma, Myeloma & Leukemia.
The researchers retrospectively analyzed the results of 8,305 ALL patients undergoing chemotherapy from the Surveillance, Epidemiology, and End Results (SEER) database during 1975 to 2015, of which 7,454 (80.1%) cases were in the de novo ALL group, and 851 (19.9%) cases were in the secondary acute lymphoblastic leukemia (sALL) group. They used propensity matching before assessing overall survival between the two groups.
Demographically, the results showed that women ALL patients had a lower risk of death than men [hazard ratio (HR) = .93, P < .01], and that the mortality in blacks was higher than that of whites (HR = 1.29, P < .001).
For both ALL groups, patients aged 45-75 years and patients 75 years and older had a higher risk of death than younger patients (HR = 1.82, P < .001 and HR = 3.85, P < .001, respectively).
Although the mean age of de novo ALL group was significantly less than that of the sALL group (51.1 vs. 60.3 years, P < .001), after the propensity matching, the 1-, 2-, 3-, 4- and 5-year overall survival of the de novo ALL group was higher than that of the sALL group at all ages (18-75 years, P < .001).
The authors speculated that one reason for the across-the-board increased mortality in sALL, compared with de novo ALL, might be the fact that sALL patients have been reported to have more MLL gene rearrangements and chromosomal aberrations than are found in de novo ALL. This has previously been suggested as the reason for poor prognosis in secondary ALL patients.
One limitation of the study mentioned by the authors was the lack of individualized chemotherapy data available for analysis. “Considering that the features of sALL and chemotherapeutic modalities or therapy protocols may affect the mortality of sALL, more work is needed to be done in the future to demonstrate the association between chemotherapy and the prognosis of ALL patients, and the influence of cytogenetic lesions and molecular characteristics on sALL,” they concluded.
The authors declared they had no conflicts of interest.
SOURCE: Zhong J et al. Clin Lymphoma Myeloma Leuk. 2020 Apr 30; doi.org/10.1016/j.clml.2020.04.013.
The application of improved chemotherapy regimens and novel chemotherapy for acute lymphoblastic leukemia (ALL) has increased the complete remission rate to 85%-90%, however, secondary ALL is common, and the prolonged long-term survival rate is only 30%-50% among ALL patients.
Favorable outcomes decrease with increasing age, and overall survival is greater for adult patients with de novo ALL, compared with patients with secondary ALL, according to the Jiansheng Zhong of the department of hematology, Guangzhou Red Cross Hospital, Jinan University, Guangzhou, Guangdong, China, and colleagues in a new study published online in Clinical Lymphoma, Myeloma & Leukemia.
The researchers retrospectively analyzed the results of 8,305 ALL patients undergoing chemotherapy from the Surveillance, Epidemiology, and End Results (SEER) database during 1975 to 2015, of which 7,454 (80.1%) cases were in the de novo ALL group, and 851 (19.9%) cases were in the secondary acute lymphoblastic leukemia (sALL) group. They used propensity matching before assessing overall survival between the two groups.
Demographically, the results showed that women ALL patients had a lower risk of death than men [hazard ratio (HR) = .93, P < .01], and that the mortality in blacks was higher than that of whites (HR = 1.29, P < .001).
For both ALL groups, patients aged 45-75 years and patients 75 years and older had a higher risk of death than younger patients (HR = 1.82, P < .001 and HR = 3.85, P < .001, respectively).
Although the mean age of de novo ALL group was significantly less than that of the sALL group (51.1 vs. 60.3 years, P < .001), after the propensity matching, the 1-, 2-, 3-, 4- and 5-year overall survival of the de novo ALL group was higher than that of the sALL group at all ages (18-75 years, P < .001).
The authors speculated that one reason for the across-the-board increased mortality in sALL, compared with de novo ALL, might be the fact that sALL patients have been reported to have more MLL gene rearrangements and chromosomal aberrations than are found in de novo ALL. This has previously been suggested as the reason for poor prognosis in secondary ALL patients.
One limitation of the study mentioned by the authors was the lack of individualized chemotherapy data available for analysis. “Considering that the features of sALL and chemotherapeutic modalities or therapy protocols may affect the mortality of sALL, more work is needed to be done in the future to demonstrate the association between chemotherapy and the prognosis of ALL patients, and the influence of cytogenetic lesions and molecular characteristics on sALL,” they concluded.
The authors declared they had no conflicts of interest.
SOURCE: Zhong J et al. Clin Lymphoma Myeloma Leuk. 2020 Apr 30; doi.org/10.1016/j.clml.2020.04.013.
The application of improved chemotherapy regimens and novel chemotherapy for acute lymphoblastic leukemia (ALL) has increased the complete remission rate to 85%-90%, however, secondary ALL is common, and the prolonged long-term survival rate is only 30%-50% among ALL patients.
Favorable outcomes decrease with increasing age, and overall survival is greater for adult patients with de novo ALL, compared with patients with secondary ALL, according to the Jiansheng Zhong of the department of hematology, Guangzhou Red Cross Hospital, Jinan University, Guangzhou, Guangdong, China, and colleagues in a new study published online in Clinical Lymphoma, Myeloma & Leukemia.
The researchers retrospectively analyzed the results of 8,305 ALL patients undergoing chemotherapy from the Surveillance, Epidemiology, and End Results (SEER) database during 1975 to 2015, of which 7,454 (80.1%) cases were in the de novo ALL group, and 851 (19.9%) cases were in the secondary acute lymphoblastic leukemia (sALL) group. They used propensity matching before assessing overall survival between the two groups.
Demographically, the results showed that women ALL patients had a lower risk of death than men [hazard ratio (HR) = .93, P < .01], and that the mortality in blacks was higher than that of whites (HR = 1.29, P < .001).
For both ALL groups, patients aged 45-75 years and patients 75 years and older had a higher risk of death than younger patients (HR = 1.82, P < .001 and HR = 3.85, P < .001, respectively).
Although the mean age of de novo ALL group was significantly less than that of the sALL group (51.1 vs. 60.3 years, P < .001), after the propensity matching, the 1-, 2-, 3-, 4- and 5-year overall survival of the de novo ALL group was higher than that of the sALL group at all ages (18-75 years, P < .001).
The authors speculated that one reason for the across-the-board increased mortality in sALL, compared with de novo ALL, might be the fact that sALL patients have been reported to have more MLL gene rearrangements and chromosomal aberrations than are found in de novo ALL. This has previously been suggested as the reason for poor prognosis in secondary ALL patients.
One limitation of the study mentioned by the authors was the lack of individualized chemotherapy data available for analysis. “Considering that the features of sALL and chemotherapeutic modalities or therapy protocols may affect the mortality of sALL, more work is needed to be done in the future to demonstrate the association between chemotherapy and the prognosis of ALL patients, and the influence of cytogenetic lesions and molecular characteristics on sALL,” they concluded.
The authors declared they had no conflicts of interest.
SOURCE: Zhong J et al. Clin Lymphoma Myeloma Leuk. 2020 Apr 30; doi.org/10.1016/j.clml.2020.04.013.
FROM Clinical Lymphoma, Myeloma & Leukemia
Blood test detects colon cancer in single-center study
Blood assay studied for colorectal cancer screening.
A blood test detected 11 of 11 cases of colorectal cancer in a study involving 354 patients, and also spotted a majority of cases – 40 out of 53 – in which participants had advanced adenomas, an investigator said.
Results from a single-center study of CellMax Life’s FirstSight blood test were released as a poster as part of the annual Digestive Disease Week®, which was canceled because of COVID-19.
For a study conducted at one site, the Veterans Affairs Palo Alto (Calif.) Healthcare System, Shai Friedland, MD, and colleagues recruited 354 patients between ages 45 and 80 who were scheduled for elective colonoscopy. The researchers excluded people with a personal history of cancer or inflammatory bowel disease. They used CellMax’s FirstSight test on blood samples from the study participants.
The FirstSight test result was positive for colorectal cancer in all 11 patients in the study who were found by colonoscopy to have this condition, said Dr. Friedland, who is a professor of medicine at Stanford (Calif.) University and chief of gastroenterology at the VA Palo Alto Healthcare System. Thus, the test showed a sensitivity of 100% in this instance.
Among the 53 study participants found by colonoscopy to have advanced adenoma, 40 were positive on FirstSight; thus, so the test has a sensitivity of 75.5% for this result.
Among 79 patients who had negative colonoscopy results, meaning they were judged free of cancer or polyps, the test showed 8 as having signs of disease or growths.
“If you had a large adenoma that was removed years ago and now you have a negative colonoscopy, your score might still be high,” Dr. Friedland said in a recorded presentation for DDW. “In other words, the changes that are detectable in your blood might persist even after the polypectomy.”
He said there are plans to soon start a large-scale multicenter study of the CellMax assay.
“The blood test has the potential to fill an unmet need by giving patients a highly sensitive convenient option for colorectal cancer screening,” he said.
CellMax already is seeking to position its test as a more convenient alternative to either colonoscopy or the Cologuard screening test. Many patients put off cancer screening because of the need to take time off from work and the invasive nature of colonoscopy. Exact Sciences has used direct-to-consumer advertising to promote its Cologuard home-based test as a more convenient alternative to colonoscopy, but its product requires patients to collect their own stool samples and mail them to a lab, a process many people find off-putting.
Public health advocates, including the U.S. Preventive Services Task Force (USPSTF), have for years been pressing for wider screening of American adults for colon cancer. USPSTF is in the midst of updating its recommendations on colon cancer. In announcing its latest update of these recommendations in 2016, USPSTF said “the best screening test is the one that gets done” (JAMA. 2016;315[23]:2564-75).
USPSTF pressed for maximizing the total proportion of the eligible population, a point Dr. Friedland echoed in a CellMax press release.
“For colon cancer screening to be most effective, it is essential to detect precancerous polyps and then perform a colonoscopy to remove the polyps,” said Dr. Friedland in the CellMax press release. “Giving patients the option of getting a blood test for screening would undoubtedly increase compliance and thereby reduce mortality from colorectal cancer.”
In the DDW presentation, Dr. Friedland and colleagues also said the CellMax test showed greater sensitivity (100%) for colorectal cancer and advanced precancerous lesions (75.5%) than did Cologuard (92.3% for colorectal cancer and 42.4% for advanced precancerous lesions).
Cara Connelly, Director of Public Relations and Corporate Communications for Exact Sciences said that the company “is dedicated to getting more people screened for colorectal cancer and applaud the researchers for their efforts. We look forward to hearing more about the performance of this test in a prospective multisite study with nonsymptomatic patients.”
Naresh T. Gunaratnam, MD, a gastroenterologist and research director at Huron Gastro in Ypsilanti, Mich., said he is concerned that aggressive promotion of alternative tests may obscure the benefits of colonoscopy. Dr. Gunaratnam, a 2019 winner of the American Gastroenterological Association (AGA) Distinguished Clinician Award, has been a public critic of the marketing of colon cancer tests, which emphasize the convenience of these products. When asked by MDedge to comment on the CellMax-funded study, Dr. Gunaratnam said alternative tests do have a place for the care of patients who cannot or will not have a colonoscopy.
“But if you convince a patient who would be willing to have a colonoscopy not to, that’s a disservice,” he said.
“If you want the best test, the one that is best at finding cancers and finding polyps and the only one that can remove the polyp, that’s colonoscopy,” Dr. Gunaratnam added. “One day there may be a pill you can swallow that blows up the polyps, but we’re not there yet. We have to mechanically remove them.”
SOURCE: Friedland S et al. DDW 2020, eposter 575.
Blood assay studied for colorectal cancer screening.
Blood assay studied for colorectal cancer screening.
A blood test detected 11 of 11 cases of colorectal cancer in a study involving 354 patients, and also spotted a majority of cases – 40 out of 53 – in which participants had advanced adenomas, an investigator said.
Results from a single-center study of CellMax Life’s FirstSight blood test were released as a poster as part of the annual Digestive Disease Week®, which was canceled because of COVID-19.
For a study conducted at one site, the Veterans Affairs Palo Alto (Calif.) Healthcare System, Shai Friedland, MD, and colleagues recruited 354 patients between ages 45 and 80 who were scheduled for elective colonoscopy. The researchers excluded people with a personal history of cancer or inflammatory bowel disease. They used CellMax’s FirstSight test on blood samples from the study participants.
The FirstSight test result was positive for colorectal cancer in all 11 patients in the study who were found by colonoscopy to have this condition, said Dr. Friedland, who is a professor of medicine at Stanford (Calif.) University and chief of gastroenterology at the VA Palo Alto Healthcare System. Thus, the test showed a sensitivity of 100% in this instance.
Among the 53 study participants found by colonoscopy to have advanced adenoma, 40 were positive on FirstSight; thus, so the test has a sensitivity of 75.5% for this result.
Among 79 patients who had negative colonoscopy results, meaning they were judged free of cancer or polyps, the test showed 8 as having signs of disease or growths.
“If you had a large adenoma that was removed years ago and now you have a negative colonoscopy, your score might still be high,” Dr. Friedland said in a recorded presentation for DDW. “In other words, the changes that are detectable in your blood might persist even after the polypectomy.”
He said there are plans to soon start a large-scale multicenter study of the CellMax assay.
“The blood test has the potential to fill an unmet need by giving patients a highly sensitive convenient option for colorectal cancer screening,” he said.
CellMax already is seeking to position its test as a more convenient alternative to either colonoscopy or the Cologuard screening test. Many patients put off cancer screening because of the need to take time off from work and the invasive nature of colonoscopy. Exact Sciences has used direct-to-consumer advertising to promote its Cologuard home-based test as a more convenient alternative to colonoscopy, but its product requires patients to collect their own stool samples and mail them to a lab, a process many people find off-putting.
Public health advocates, including the U.S. Preventive Services Task Force (USPSTF), have for years been pressing for wider screening of American adults for colon cancer. USPSTF is in the midst of updating its recommendations on colon cancer. In announcing its latest update of these recommendations in 2016, USPSTF said “the best screening test is the one that gets done” (JAMA. 2016;315[23]:2564-75).
USPSTF pressed for maximizing the total proportion of the eligible population, a point Dr. Friedland echoed in a CellMax press release.
“For colon cancer screening to be most effective, it is essential to detect precancerous polyps and then perform a colonoscopy to remove the polyps,” said Dr. Friedland in the CellMax press release. “Giving patients the option of getting a blood test for screening would undoubtedly increase compliance and thereby reduce mortality from colorectal cancer.”
In the DDW presentation, Dr. Friedland and colleagues also said the CellMax test showed greater sensitivity (100%) for colorectal cancer and advanced precancerous lesions (75.5%) than did Cologuard (92.3% for colorectal cancer and 42.4% for advanced precancerous lesions).
Cara Connelly, Director of Public Relations and Corporate Communications for Exact Sciences said that the company “is dedicated to getting more people screened for colorectal cancer and applaud the researchers for their efforts. We look forward to hearing more about the performance of this test in a prospective multisite study with nonsymptomatic patients.”
Naresh T. Gunaratnam, MD, a gastroenterologist and research director at Huron Gastro in Ypsilanti, Mich., said he is concerned that aggressive promotion of alternative tests may obscure the benefits of colonoscopy. Dr. Gunaratnam, a 2019 winner of the American Gastroenterological Association (AGA) Distinguished Clinician Award, has been a public critic of the marketing of colon cancer tests, which emphasize the convenience of these products. When asked by MDedge to comment on the CellMax-funded study, Dr. Gunaratnam said alternative tests do have a place for the care of patients who cannot or will not have a colonoscopy.
“But if you convince a patient who would be willing to have a colonoscopy not to, that’s a disservice,” he said.
“If you want the best test, the one that is best at finding cancers and finding polyps and the only one that can remove the polyp, that’s colonoscopy,” Dr. Gunaratnam added. “One day there may be a pill you can swallow that blows up the polyps, but we’re not there yet. We have to mechanically remove them.”
SOURCE: Friedland S et al. DDW 2020, eposter 575.
A blood test detected 11 of 11 cases of colorectal cancer in a study involving 354 patients, and also spotted a majority of cases – 40 out of 53 – in which participants had advanced adenomas, an investigator said.
Results from a single-center study of CellMax Life’s FirstSight blood test were released as a poster as part of the annual Digestive Disease Week®, which was canceled because of COVID-19.
For a study conducted at one site, the Veterans Affairs Palo Alto (Calif.) Healthcare System, Shai Friedland, MD, and colleagues recruited 354 patients between ages 45 and 80 who were scheduled for elective colonoscopy. The researchers excluded people with a personal history of cancer or inflammatory bowel disease. They used CellMax’s FirstSight test on blood samples from the study participants.
The FirstSight test result was positive for colorectal cancer in all 11 patients in the study who were found by colonoscopy to have this condition, said Dr. Friedland, who is a professor of medicine at Stanford (Calif.) University and chief of gastroenterology at the VA Palo Alto Healthcare System. Thus, the test showed a sensitivity of 100% in this instance.
Among the 53 study participants found by colonoscopy to have advanced adenoma, 40 were positive on FirstSight; thus, so the test has a sensitivity of 75.5% for this result.
Among 79 patients who had negative colonoscopy results, meaning they were judged free of cancer or polyps, the test showed 8 as having signs of disease or growths.
“If you had a large adenoma that was removed years ago and now you have a negative colonoscopy, your score might still be high,” Dr. Friedland said in a recorded presentation for DDW. “In other words, the changes that are detectable in your blood might persist even after the polypectomy.”
He said there are plans to soon start a large-scale multicenter study of the CellMax assay.
“The blood test has the potential to fill an unmet need by giving patients a highly sensitive convenient option for colorectal cancer screening,” he said.
CellMax already is seeking to position its test as a more convenient alternative to either colonoscopy or the Cologuard screening test. Many patients put off cancer screening because of the need to take time off from work and the invasive nature of colonoscopy. Exact Sciences has used direct-to-consumer advertising to promote its Cologuard home-based test as a more convenient alternative to colonoscopy, but its product requires patients to collect their own stool samples and mail them to a lab, a process many people find off-putting.
Public health advocates, including the U.S. Preventive Services Task Force (USPSTF), have for years been pressing for wider screening of American adults for colon cancer. USPSTF is in the midst of updating its recommendations on colon cancer. In announcing its latest update of these recommendations in 2016, USPSTF said “the best screening test is the one that gets done” (JAMA. 2016;315[23]:2564-75).
USPSTF pressed for maximizing the total proportion of the eligible population, a point Dr. Friedland echoed in a CellMax press release.
“For colon cancer screening to be most effective, it is essential to detect precancerous polyps and then perform a colonoscopy to remove the polyps,” said Dr. Friedland in the CellMax press release. “Giving patients the option of getting a blood test for screening would undoubtedly increase compliance and thereby reduce mortality from colorectal cancer.”
In the DDW presentation, Dr. Friedland and colleagues also said the CellMax test showed greater sensitivity (100%) for colorectal cancer and advanced precancerous lesions (75.5%) than did Cologuard (92.3% for colorectal cancer and 42.4% for advanced precancerous lesions).
Cara Connelly, Director of Public Relations and Corporate Communications for Exact Sciences said that the company “is dedicated to getting more people screened for colorectal cancer and applaud the researchers for their efforts. We look forward to hearing more about the performance of this test in a prospective multisite study with nonsymptomatic patients.”
Naresh T. Gunaratnam, MD, a gastroenterologist and research director at Huron Gastro in Ypsilanti, Mich., said he is concerned that aggressive promotion of alternative tests may obscure the benefits of colonoscopy. Dr. Gunaratnam, a 2019 winner of the American Gastroenterological Association (AGA) Distinguished Clinician Award, has been a public critic of the marketing of colon cancer tests, which emphasize the convenience of these products. When asked by MDedge to comment on the CellMax-funded study, Dr. Gunaratnam said alternative tests do have a place for the care of patients who cannot or will not have a colonoscopy.
“But if you convince a patient who would be willing to have a colonoscopy not to, that’s a disservice,” he said.
“If you want the best test, the one that is best at finding cancers and finding polyps and the only one that can remove the polyp, that’s colonoscopy,” Dr. Gunaratnam added. “One day there may be a pill you can swallow that blows up the polyps, but we’re not there yet. We have to mechanically remove them.”
SOURCE: Friedland S et al. DDW 2020, eposter 575.
FROM DDW 2020