User login
FDA expands Xofluza indication to include postexposure flu prophylaxis
The US Food and Drug Administration (FDA) has expanded the indication for the antiviral baloxavir marboxil (Xofluza) to include postexposure prophylaxis of uncomplicated influenza in people aged 12 years and older.
“This expanded indication for Xofluza will provide an important option to help prevent influenza just in time for a flu season that is anticipated to be unlike any other because it will coincide with the coronavirus pandemic,” Debra Birnkrant, MD, director, Division of Antiviral Products, FDA Center for Drug Evaluation and Research, said in a press release.
In addition, Xofluza, which was previously available only in tablet form, is also now available as granules for mixing in water, the FDA said.
The agency first approved baloxavir marboxil in 2018 for the treatment of acute uncomplicated influenza in people aged 12 years or older who have been symptomatic for no more than 48 hours.
A year later, the FDA expanded the indication to include people at high risk of developing influenza-related complications, such as those with asthma, chronic lung disease, diabetes, heart disease, or morbid obesity, as well as adults aged 65 years or older.
The safety and efficacy of Xofluza for influenza postexposure prophylaxis is supported by a randomized, double-blind, controlled trial involving 607 people aged 12 years and older. After exposure to a person with influenza in their household, they received a single dose of Xofluza or placebo.
The primary endpoint was the proportion of individuals who became infected with influenza and presented with fever and at least one respiratory symptom from day 1 to day 10.
Of the 303 people who received Xofluza, 1% of individuals met these criteria, compared with 13% of those who received placebo.
The most common adverse effects of Xofluza include diarrhea, bronchitis, nausea, sinusitis, and headache.
Hypersensitivity, including anaphylaxis, can occur in patients taking Xofluza. The antiviral is contraindicated in people with a known hypersensitivity reaction to Xofluza.
Xofluza should not be coadministered with dairy products, calcium-fortified beverages, laxatives, antacids, or oral supplements containing calcium, iron, magnesium, selenium, aluminium, or zinc.
Full prescribing information is available online.
This article first appeared on Medscape.com.
The US Food and Drug Administration (FDA) has expanded the indication for the antiviral baloxavir marboxil (Xofluza) to include postexposure prophylaxis of uncomplicated influenza in people aged 12 years and older.
“This expanded indication for Xofluza will provide an important option to help prevent influenza just in time for a flu season that is anticipated to be unlike any other because it will coincide with the coronavirus pandemic,” Debra Birnkrant, MD, director, Division of Antiviral Products, FDA Center for Drug Evaluation and Research, said in a press release.
In addition, Xofluza, which was previously available only in tablet form, is also now available as granules for mixing in water, the FDA said.
The agency first approved baloxavir marboxil in 2018 for the treatment of acute uncomplicated influenza in people aged 12 years or older who have been symptomatic for no more than 48 hours.
A year later, the FDA expanded the indication to include people at high risk of developing influenza-related complications, such as those with asthma, chronic lung disease, diabetes, heart disease, or morbid obesity, as well as adults aged 65 years or older.
The safety and efficacy of Xofluza for influenza postexposure prophylaxis is supported by a randomized, double-blind, controlled trial involving 607 people aged 12 years and older. After exposure to a person with influenza in their household, they received a single dose of Xofluza or placebo.
The primary endpoint was the proportion of individuals who became infected with influenza and presented with fever and at least one respiratory symptom from day 1 to day 10.
Of the 303 people who received Xofluza, 1% of individuals met these criteria, compared with 13% of those who received placebo.
The most common adverse effects of Xofluza include diarrhea, bronchitis, nausea, sinusitis, and headache.
Hypersensitivity, including anaphylaxis, can occur in patients taking Xofluza. The antiviral is contraindicated in people with a known hypersensitivity reaction to Xofluza.
Xofluza should not be coadministered with dairy products, calcium-fortified beverages, laxatives, antacids, or oral supplements containing calcium, iron, magnesium, selenium, aluminium, or zinc.
Full prescribing information is available online.
This article first appeared on Medscape.com.
The US Food and Drug Administration (FDA) has expanded the indication for the antiviral baloxavir marboxil (Xofluza) to include postexposure prophylaxis of uncomplicated influenza in people aged 12 years and older.
“This expanded indication for Xofluza will provide an important option to help prevent influenza just in time for a flu season that is anticipated to be unlike any other because it will coincide with the coronavirus pandemic,” Debra Birnkrant, MD, director, Division of Antiviral Products, FDA Center for Drug Evaluation and Research, said in a press release.
In addition, Xofluza, which was previously available only in tablet form, is also now available as granules for mixing in water, the FDA said.
The agency first approved baloxavir marboxil in 2018 for the treatment of acute uncomplicated influenza in people aged 12 years or older who have been symptomatic for no more than 48 hours.
A year later, the FDA expanded the indication to include people at high risk of developing influenza-related complications, such as those with asthma, chronic lung disease, diabetes, heart disease, or morbid obesity, as well as adults aged 65 years or older.
The safety and efficacy of Xofluza for influenza postexposure prophylaxis is supported by a randomized, double-blind, controlled trial involving 607 people aged 12 years and older. After exposure to a person with influenza in their household, they received a single dose of Xofluza or placebo.
The primary endpoint was the proportion of individuals who became infected with influenza and presented with fever and at least one respiratory symptom from day 1 to day 10.
Of the 303 people who received Xofluza, 1% of individuals met these criteria, compared with 13% of those who received placebo.
The most common adverse effects of Xofluza include diarrhea, bronchitis, nausea, sinusitis, and headache.
Hypersensitivity, including anaphylaxis, can occur in patients taking Xofluza. The antiviral is contraindicated in people with a known hypersensitivity reaction to Xofluza.
Xofluza should not be coadministered with dairy products, calcium-fortified beverages, laxatives, antacids, or oral supplements containing calcium, iron, magnesium, selenium, aluminium, or zinc.
Full prescribing information is available online.
This article first appeared on Medscape.com.
Mortality rate of SARS-CoV-2 for similar patients is declining over time
Clinical question: Is the observed drop in COVID-19 mortality caused by changing demographics or improvements in patient care?
Background: At the start of the pandemic, COVID-19 had high mortality rates (6.9% in April according to the U.S. Centers for Disease Control and Prevention). More recently, the mortality rate had decreased to 1.9% of diagnosed cases at the end of September 2020. Concurrently, the median age of confirmed cases dropped as well, from 46 to 38 years, and availability of testing has expanded. It remains unclear whether the drop in mortality rate is because of affected patients with fewer comorbidities, less symptomatic patients, or improvements in clinical care.
Study design: Retrospective cohort study.
Setting: Large urban academic health system.
Synopsis: Researchers analyzed admissions from March 1 through Aug. 31, 2020, to NYU Langone Health System in New York of patients older than 18 years with laboratory-confirmed infection with SARS-CoV-2 during the hospitalization or in the preceding 2 weeks. In total, 5,118 patients qualified for analysis, of which 53% were hospitalized during March and April. Two separate multivariate logistic regression models for mortality were created based on patient demographics, comorbidities, and presenting vital signs and lab-result abnormalities. In the first model, the month of admission was not included, and the standardized mortality ratio (SMR) – the ratio of the sum of observed and expected deaths – for each month was obtained. In the second model, the month of admission was included as a covariate, and the average marginal effect (AME) – the difference in probability of death or discharge to hospice between March and a subsequent time period for equivalent patients – was calculated. The SMR declined progressively over time from 1.26 (95% confidence interval, 1.15-1.39) in March to 0.38 (95% CI, 0.12-0.88) in August. When accounting for demographic and clinical severity changes, the adjusted AME declined every subsequent month after March reaching a maximum of 18.2 (95% CI, 12.0-24.4) percentage point decrease in probability of death in August.
Bottom line: Mortality from SARS-CoV-2 was significantly lower at the end of the 6-month period when adjusted for demographic and clinical factors for patients admitted to a single health care system in the United States.
Citation: Horwitz LI et al. Trends in COVID-19 Risk-Adjusted Mortality Rates. J Hosp Med. 2020 Oct 23. doi: 10.12788/jhm.3552
Dr. Halpern is a med-peds hospitalist at Brigham and Women’s Hospital in Boston.
Clinical question: Is the observed drop in COVID-19 mortality caused by changing demographics or improvements in patient care?
Background: At the start of the pandemic, COVID-19 had high mortality rates (6.9% in April according to the U.S. Centers for Disease Control and Prevention). More recently, the mortality rate had decreased to 1.9% of diagnosed cases at the end of September 2020. Concurrently, the median age of confirmed cases dropped as well, from 46 to 38 years, and availability of testing has expanded. It remains unclear whether the drop in mortality rate is because of affected patients with fewer comorbidities, less symptomatic patients, or improvements in clinical care.
Study design: Retrospective cohort study.
Setting: Large urban academic health system.
Synopsis: Researchers analyzed admissions from March 1 through Aug. 31, 2020, to NYU Langone Health System in New York of patients older than 18 years with laboratory-confirmed infection with SARS-CoV-2 during the hospitalization or in the preceding 2 weeks. In total, 5,118 patients qualified for analysis, of which 53% were hospitalized during March and April. Two separate multivariate logistic regression models for mortality were created based on patient demographics, comorbidities, and presenting vital signs and lab-result abnormalities. In the first model, the month of admission was not included, and the standardized mortality ratio (SMR) – the ratio of the sum of observed and expected deaths – for each month was obtained. In the second model, the month of admission was included as a covariate, and the average marginal effect (AME) – the difference in probability of death or discharge to hospice between March and a subsequent time period for equivalent patients – was calculated. The SMR declined progressively over time from 1.26 (95% confidence interval, 1.15-1.39) in March to 0.38 (95% CI, 0.12-0.88) in August. When accounting for demographic and clinical severity changes, the adjusted AME declined every subsequent month after March reaching a maximum of 18.2 (95% CI, 12.0-24.4) percentage point decrease in probability of death in August.
Bottom line: Mortality from SARS-CoV-2 was significantly lower at the end of the 6-month period when adjusted for demographic and clinical factors for patients admitted to a single health care system in the United States.
Citation: Horwitz LI et al. Trends in COVID-19 Risk-Adjusted Mortality Rates. J Hosp Med. 2020 Oct 23. doi: 10.12788/jhm.3552
Dr. Halpern is a med-peds hospitalist at Brigham and Women’s Hospital in Boston.
Clinical question: Is the observed drop in COVID-19 mortality caused by changing demographics or improvements in patient care?
Background: At the start of the pandemic, COVID-19 had high mortality rates (6.9% in April according to the U.S. Centers for Disease Control and Prevention). More recently, the mortality rate had decreased to 1.9% of diagnosed cases at the end of September 2020. Concurrently, the median age of confirmed cases dropped as well, from 46 to 38 years, and availability of testing has expanded. It remains unclear whether the drop in mortality rate is because of affected patients with fewer comorbidities, less symptomatic patients, or improvements in clinical care.
Study design: Retrospective cohort study.
Setting: Large urban academic health system.
Synopsis: Researchers analyzed admissions from March 1 through Aug. 31, 2020, to NYU Langone Health System in New York of patients older than 18 years with laboratory-confirmed infection with SARS-CoV-2 during the hospitalization or in the preceding 2 weeks. In total, 5,118 patients qualified for analysis, of which 53% were hospitalized during March and April. Two separate multivariate logistic regression models for mortality were created based on patient demographics, comorbidities, and presenting vital signs and lab-result abnormalities. In the first model, the month of admission was not included, and the standardized mortality ratio (SMR) – the ratio of the sum of observed and expected deaths – for each month was obtained. In the second model, the month of admission was included as a covariate, and the average marginal effect (AME) – the difference in probability of death or discharge to hospice between March and a subsequent time period for equivalent patients – was calculated. The SMR declined progressively over time from 1.26 (95% confidence interval, 1.15-1.39) in March to 0.38 (95% CI, 0.12-0.88) in August. When accounting for demographic and clinical severity changes, the adjusted AME declined every subsequent month after March reaching a maximum of 18.2 (95% CI, 12.0-24.4) percentage point decrease in probability of death in August.
Bottom line: Mortality from SARS-CoV-2 was significantly lower at the end of the 6-month period when adjusted for demographic and clinical factors for patients admitted to a single health care system in the United States.
Citation: Horwitz LI et al. Trends in COVID-19 Risk-Adjusted Mortality Rates. J Hosp Med. 2020 Oct 23. doi: 10.12788/jhm.3552
Dr. Halpern is a med-peds hospitalist at Brigham and Women’s Hospital in Boston.
FROM THE JOURNAL OF HOSPITAL MEDICINE
Rationale for baricitinib’s use in COVID-19 patients demonstrated
It should not be surprising that the RA drug baricitinib (Olumiant), a Janus kinase (JAK) 1/2 inhibitor, might be beneficial in controlling the cytokine storm of hyperinflammation that can follow severe SARS-CoV-2 infections and lead to lung damage and acute respiratory distress syndrome – the leading cause of death from the virus.
But to demonstrate within a matter of months, at least preliminarily, that baricitinib reduces mortality and morbidity in hospitalized patients with COVID-19 pneumonia required a widely cross-disciplinary international team of researchers from 10 countries working at breakneck speed, said Justin Stebbing, PhD, the principal investigator of a new baricitinib study published Nov. 13 in Science Advances. “We went from modeling and mechanistic investigations to clinical tests in a number of settings and laboratory analysis in record time.”
The international team of 50 researchers included medical specialists in rheumatology, virology, geriatrics, oncology, and general medicine, along with experts in molecular and cellular biology, bioinformatics, statistics and trial design, computer modeling, pathology, genetics, and super-resolution microscopy, Dr. Stebbing, professor of cancer medicine and medical oncology at Imperial College, London, said in an interview.
Artificial intelligence, provided by the London-based firm BenevolentAI, was used to sift through a huge repository of structured medical information to identify drugs that might block the SARS-CoV-2 infection process. It predicted that baricitinib would be a promising candidate to inhibit inflammation and reduce viral load in COVID-19. Previous reports by Dr. Stebbing and colleagues (here and here) describe this AI-mediated testing, which was validated by the new study.
The researchers also used three-dimensional miniature human liver organoids in vitro and super-resolution microscopy to perform further lab investigations, which showed that baricitinib reversed expression of the SARS-CoV-2 receptor ACE2 triggered by type I interferons. Baricitinib inhibited the significant increase in ACE2 expression caused by interferon alpha-2, and thus cytokine-mediated inflammation, and also reduced infectivity, Dr. Stebbing said. “Our study of baricitinib shows that it has both antiviral and anticytokine effects and appears to be safe.”
71% mortality reduction
The team found a 71% reduction in mortality for a group of 83 hospitalized patients with COVID-19 pneumonia in Italy and Spain – early epicenters of the pandemic – who received baricitinib along with standard care, compared with propensity-matched groups that received only standard care. At that time, between mid-March and mid-April, standard COVID-19 care included antibiotics, glucocorticoids, hydroxychloroquine, low-molecular-weight heparin, and the antiretroviral combination lopinavir/ritonavir.
In the Spanish and Italian cohorts, baricitinib was generally well tolerated, although not without side effects, including bacterial infections and increases in liver enzyme levels, which may not have been related to baricitinib. Patients showed reductions in inflammation within days of starting treatment. “We did not observe thrombotic or vascular events in our cohorts, but most of the patients were receiving low molecular weight heparin,” he said.
The fact that baricitinib is approved by the Food and Drug Administration, is already well studied for safety, can be taken conveniently as a once-daily oral tablet, and is less expensive than many other antiviral treatments all make it an good target for further study, including randomized, controlled trials that are already underway, Dr. Stebbing noted. His study cohort also included elderly patients (median age, 81 years) who are the most likely to experience severe disease or death from COVID-19.
The National Library of Medicine’s clinicaltrials.gov registry of federally funded clinical studies lists 15 current research initiatives involving baricitinib and COVID-19. Dr. Stebbing suggested that data generated so far are helping to guide ongoing studies on dose and duration of treatment – in other words, who it works for, when to give it, and at what dose it should be taken and for how long.
Manufacturer Eli Lilly, which markets baricitinib in 2-mg or 4-mg tablets, announced in October that initial data are starting to emerge from 1,000-plus patients enrolled in ACTT-2 (the Adaptive COVID-19 Treatment Trial 2). ACTT-2 compared patients on the broad-spectrum intravenous antiviral drug remdesivir (Veklury) with those receiving remdesivir in combination with baricitinib. Based on ACTT-2 results that suggested a reduced time to recovery and improved clinical outcomes for the combination group, the FDA issued an emergency-use authorization on Nov. 19 for the combination of baricitinib and remdesivir for the treatment of suspected or laboratory confirmed COVID-19 in hospitalized adults and pediatric patients aged 2 years or older requiring supplemental oxygen, invasive mechanical ventilation, or extracorporeal membrane oxygenation.
Interrupting the cytokine outbreak
Baricitinib has the potential to reduce or interrupt the passage of SARS-CoV-2 into cells, and thus to inhibit the JAK1- and JAK2-mediated cytokine outbreak, researcher Heinz-Josef Lenz, MD, professor of medicine and preventive medicine at the University of Southern California’s Norris Comprehensive Cancer Center in Los Angeles, said in a comment. Baricitinib was also identified, using BenevolentAI’s proprietary, artificial intelligence-derived knowledge graph, as a numb-associated kinase inhibitor, with high affinity for AP2-associated protein kinase 1, an important endocytosis regulator.
Early clinical data suggest a potent biologic effect of baricitinib 2 mg or 4 mg daily on circulating interleukin-6 levels and other inflammatory markers, including C-reactive protein. Dr. Lenz said the evidence for advantageous action of baricitinib on viral endocytosis and excessive cytokine release constitutes the rationale for using it in combination with other antivirals such as remdesivir in patients with moderate to severe COVID-19 illness.
“Although baricitinib may display antiviral activity on its own, its anti-inflammatory effects could hypothetically delay viral clearance,” Dr. Lenz added. “The data from Stebbing et al. confirm the dual actions of baricitinib, demonstrating its ability to inhibit viral entry into primary human hepatocyte spheroids and the reduction in inflammatory markers in COVID-19 patients.”
Other JAK inhibitors were not advanced as promising candidates for the research team’s attention by its artificial intelligence search, Dr. Stebbing noted. “The history of the pandemic has taught us the importance of well-designed observational studies as well as randomized, controlled trials. When it comes to COVID, pyrite looks much like gold, as failed studies of four antivirals have shown.”
Although the current translational research study did not use a placebo group, it is an important next step toward future randomized, controlled trials. “What’s great about this study is its high degree of collaboration, done with real urgency,” he added. “It’s harder to produce a paper that crosses multiple boundaries, like this one does, than a single-focused piece of work. But we wanted to link all of these threads together.”
The study was supported by the Imperial Biomedical Research Centre and Experimental Cancer Medicine Centre, the National Institute for Health Research, and the U.K. National Health Service’s Accelerated Access Collaborative. Dr. Stebbing has served on scientific advisory boards for Eli Lilly and other companies. Dr. Lenz had no relevant disclosures to report.
SOURCE: Stebbing J et al. Sci Adv. 2020 Nov 13. doi: 10.1126/sciadv.abe4724.
It should not be surprising that the RA drug baricitinib (Olumiant), a Janus kinase (JAK) 1/2 inhibitor, might be beneficial in controlling the cytokine storm of hyperinflammation that can follow severe SARS-CoV-2 infections and lead to lung damage and acute respiratory distress syndrome – the leading cause of death from the virus.
But to demonstrate within a matter of months, at least preliminarily, that baricitinib reduces mortality and morbidity in hospitalized patients with COVID-19 pneumonia required a widely cross-disciplinary international team of researchers from 10 countries working at breakneck speed, said Justin Stebbing, PhD, the principal investigator of a new baricitinib study published Nov. 13 in Science Advances. “We went from modeling and mechanistic investigations to clinical tests in a number of settings and laboratory analysis in record time.”
The international team of 50 researchers included medical specialists in rheumatology, virology, geriatrics, oncology, and general medicine, along with experts in molecular and cellular biology, bioinformatics, statistics and trial design, computer modeling, pathology, genetics, and super-resolution microscopy, Dr. Stebbing, professor of cancer medicine and medical oncology at Imperial College, London, said in an interview.
Artificial intelligence, provided by the London-based firm BenevolentAI, was used to sift through a huge repository of structured medical information to identify drugs that might block the SARS-CoV-2 infection process. It predicted that baricitinib would be a promising candidate to inhibit inflammation and reduce viral load in COVID-19. Previous reports by Dr. Stebbing and colleagues (here and here) describe this AI-mediated testing, which was validated by the new study.
The researchers also used three-dimensional miniature human liver organoids in vitro and super-resolution microscopy to perform further lab investigations, which showed that baricitinib reversed expression of the SARS-CoV-2 receptor ACE2 triggered by type I interferons. Baricitinib inhibited the significant increase in ACE2 expression caused by interferon alpha-2, and thus cytokine-mediated inflammation, and also reduced infectivity, Dr. Stebbing said. “Our study of baricitinib shows that it has both antiviral and anticytokine effects and appears to be safe.”
71% mortality reduction
The team found a 71% reduction in mortality for a group of 83 hospitalized patients with COVID-19 pneumonia in Italy and Spain – early epicenters of the pandemic – who received baricitinib along with standard care, compared with propensity-matched groups that received only standard care. At that time, between mid-March and mid-April, standard COVID-19 care included antibiotics, glucocorticoids, hydroxychloroquine, low-molecular-weight heparin, and the antiretroviral combination lopinavir/ritonavir.
In the Spanish and Italian cohorts, baricitinib was generally well tolerated, although not without side effects, including bacterial infections and increases in liver enzyme levels, which may not have been related to baricitinib. Patients showed reductions in inflammation within days of starting treatment. “We did not observe thrombotic or vascular events in our cohorts, but most of the patients were receiving low molecular weight heparin,” he said.
The fact that baricitinib is approved by the Food and Drug Administration, is already well studied for safety, can be taken conveniently as a once-daily oral tablet, and is less expensive than many other antiviral treatments all make it an good target for further study, including randomized, controlled trials that are already underway, Dr. Stebbing noted. His study cohort also included elderly patients (median age, 81 years) who are the most likely to experience severe disease or death from COVID-19.
The National Library of Medicine’s clinicaltrials.gov registry of federally funded clinical studies lists 15 current research initiatives involving baricitinib and COVID-19. Dr. Stebbing suggested that data generated so far are helping to guide ongoing studies on dose and duration of treatment – in other words, who it works for, when to give it, and at what dose it should be taken and for how long.
Manufacturer Eli Lilly, which markets baricitinib in 2-mg or 4-mg tablets, announced in October that initial data are starting to emerge from 1,000-plus patients enrolled in ACTT-2 (the Adaptive COVID-19 Treatment Trial 2). ACTT-2 compared patients on the broad-spectrum intravenous antiviral drug remdesivir (Veklury) with those receiving remdesivir in combination with baricitinib. Based on ACTT-2 results that suggested a reduced time to recovery and improved clinical outcomes for the combination group, the FDA issued an emergency-use authorization on Nov. 19 for the combination of baricitinib and remdesivir for the treatment of suspected or laboratory confirmed COVID-19 in hospitalized adults and pediatric patients aged 2 years or older requiring supplemental oxygen, invasive mechanical ventilation, or extracorporeal membrane oxygenation.
Interrupting the cytokine outbreak
Baricitinib has the potential to reduce or interrupt the passage of SARS-CoV-2 into cells, and thus to inhibit the JAK1- and JAK2-mediated cytokine outbreak, researcher Heinz-Josef Lenz, MD, professor of medicine and preventive medicine at the University of Southern California’s Norris Comprehensive Cancer Center in Los Angeles, said in a comment. Baricitinib was also identified, using BenevolentAI’s proprietary, artificial intelligence-derived knowledge graph, as a numb-associated kinase inhibitor, with high affinity for AP2-associated protein kinase 1, an important endocytosis regulator.
Early clinical data suggest a potent biologic effect of baricitinib 2 mg or 4 mg daily on circulating interleukin-6 levels and other inflammatory markers, including C-reactive protein. Dr. Lenz said the evidence for advantageous action of baricitinib on viral endocytosis and excessive cytokine release constitutes the rationale for using it in combination with other antivirals such as remdesivir in patients with moderate to severe COVID-19 illness.
“Although baricitinib may display antiviral activity on its own, its anti-inflammatory effects could hypothetically delay viral clearance,” Dr. Lenz added. “The data from Stebbing et al. confirm the dual actions of baricitinib, demonstrating its ability to inhibit viral entry into primary human hepatocyte spheroids and the reduction in inflammatory markers in COVID-19 patients.”
Other JAK inhibitors were not advanced as promising candidates for the research team’s attention by its artificial intelligence search, Dr. Stebbing noted. “The history of the pandemic has taught us the importance of well-designed observational studies as well as randomized, controlled trials. When it comes to COVID, pyrite looks much like gold, as failed studies of four antivirals have shown.”
Although the current translational research study did not use a placebo group, it is an important next step toward future randomized, controlled trials. “What’s great about this study is its high degree of collaboration, done with real urgency,” he added. “It’s harder to produce a paper that crosses multiple boundaries, like this one does, than a single-focused piece of work. But we wanted to link all of these threads together.”
The study was supported by the Imperial Biomedical Research Centre and Experimental Cancer Medicine Centre, the National Institute for Health Research, and the U.K. National Health Service’s Accelerated Access Collaborative. Dr. Stebbing has served on scientific advisory boards for Eli Lilly and other companies. Dr. Lenz had no relevant disclosures to report.
SOURCE: Stebbing J et al. Sci Adv. 2020 Nov 13. doi: 10.1126/sciadv.abe4724.
It should not be surprising that the RA drug baricitinib (Olumiant), a Janus kinase (JAK) 1/2 inhibitor, might be beneficial in controlling the cytokine storm of hyperinflammation that can follow severe SARS-CoV-2 infections and lead to lung damage and acute respiratory distress syndrome – the leading cause of death from the virus.
But to demonstrate within a matter of months, at least preliminarily, that baricitinib reduces mortality and morbidity in hospitalized patients with COVID-19 pneumonia required a widely cross-disciplinary international team of researchers from 10 countries working at breakneck speed, said Justin Stebbing, PhD, the principal investigator of a new baricitinib study published Nov. 13 in Science Advances. “We went from modeling and mechanistic investigations to clinical tests in a number of settings and laboratory analysis in record time.”
The international team of 50 researchers included medical specialists in rheumatology, virology, geriatrics, oncology, and general medicine, along with experts in molecular and cellular biology, bioinformatics, statistics and trial design, computer modeling, pathology, genetics, and super-resolution microscopy, Dr. Stebbing, professor of cancer medicine and medical oncology at Imperial College, London, said in an interview.
Artificial intelligence, provided by the London-based firm BenevolentAI, was used to sift through a huge repository of structured medical information to identify drugs that might block the SARS-CoV-2 infection process. It predicted that baricitinib would be a promising candidate to inhibit inflammation and reduce viral load in COVID-19. Previous reports by Dr. Stebbing and colleagues (here and here) describe this AI-mediated testing, which was validated by the new study.
The researchers also used three-dimensional miniature human liver organoids in vitro and super-resolution microscopy to perform further lab investigations, which showed that baricitinib reversed expression of the SARS-CoV-2 receptor ACE2 triggered by type I interferons. Baricitinib inhibited the significant increase in ACE2 expression caused by interferon alpha-2, and thus cytokine-mediated inflammation, and also reduced infectivity, Dr. Stebbing said. “Our study of baricitinib shows that it has both antiviral and anticytokine effects and appears to be safe.”
71% mortality reduction
The team found a 71% reduction in mortality for a group of 83 hospitalized patients with COVID-19 pneumonia in Italy and Spain – early epicenters of the pandemic – who received baricitinib along with standard care, compared with propensity-matched groups that received only standard care. At that time, between mid-March and mid-April, standard COVID-19 care included antibiotics, glucocorticoids, hydroxychloroquine, low-molecular-weight heparin, and the antiretroviral combination lopinavir/ritonavir.
In the Spanish and Italian cohorts, baricitinib was generally well tolerated, although not without side effects, including bacterial infections and increases in liver enzyme levels, which may not have been related to baricitinib. Patients showed reductions in inflammation within days of starting treatment. “We did not observe thrombotic or vascular events in our cohorts, but most of the patients were receiving low molecular weight heparin,” he said.
The fact that baricitinib is approved by the Food and Drug Administration, is already well studied for safety, can be taken conveniently as a once-daily oral tablet, and is less expensive than many other antiviral treatments all make it an good target for further study, including randomized, controlled trials that are already underway, Dr. Stebbing noted. His study cohort also included elderly patients (median age, 81 years) who are the most likely to experience severe disease or death from COVID-19.
The National Library of Medicine’s clinicaltrials.gov registry of federally funded clinical studies lists 15 current research initiatives involving baricitinib and COVID-19. Dr. Stebbing suggested that data generated so far are helping to guide ongoing studies on dose and duration of treatment – in other words, who it works for, when to give it, and at what dose it should be taken and for how long.
Manufacturer Eli Lilly, which markets baricitinib in 2-mg or 4-mg tablets, announced in October that initial data are starting to emerge from 1,000-plus patients enrolled in ACTT-2 (the Adaptive COVID-19 Treatment Trial 2). ACTT-2 compared patients on the broad-spectrum intravenous antiviral drug remdesivir (Veklury) with those receiving remdesivir in combination with baricitinib. Based on ACTT-2 results that suggested a reduced time to recovery and improved clinical outcomes for the combination group, the FDA issued an emergency-use authorization on Nov. 19 for the combination of baricitinib and remdesivir for the treatment of suspected or laboratory confirmed COVID-19 in hospitalized adults and pediatric patients aged 2 years or older requiring supplemental oxygen, invasive mechanical ventilation, or extracorporeal membrane oxygenation.
Interrupting the cytokine outbreak
Baricitinib has the potential to reduce or interrupt the passage of SARS-CoV-2 into cells, and thus to inhibit the JAK1- and JAK2-mediated cytokine outbreak, researcher Heinz-Josef Lenz, MD, professor of medicine and preventive medicine at the University of Southern California’s Norris Comprehensive Cancer Center in Los Angeles, said in a comment. Baricitinib was also identified, using BenevolentAI’s proprietary, artificial intelligence-derived knowledge graph, as a numb-associated kinase inhibitor, with high affinity for AP2-associated protein kinase 1, an important endocytosis regulator.
Early clinical data suggest a potent biologic effect of baricitinib 2 mg or 4 mg daily on circulating interleukin-6 levels and other inflammatory markers, including C-reactive protein. Dr. Lenz said the evidence for advantageous action of baricitinib on viral endocytosis and excessive cytokine release constitutes the rationale for using it in combination with other antivirals such as remdesivir in patients with moderate to severe COVID-19 illness.
“Although baricitinib may display antiviral activity on its own, its anti-inflammatory effects could hypothetically delay viral clearance,” Dr. Lenz added. “The data from Stebbing et al. confirm the dual actions of baricitinib, demonstrating its ability to inhibit viral entry into primary human hepatocyte spheroids and the reduction in inflammatory markers in COVID-19 patients.”
Other JAK inhibitors were not advanced as promising candidates for the research team’s attention by its artificial intelligence search, Dr. Stebbing noted. “The history of the pandemic has taught us the importance of well-designed observational studies as well as randomized, controlled trials. When it comes to COVID, pyrite looks much like gold, as failed studies of four antivirals have shown.”
Although the current translational research study did not use a placebo group, it is an important next step toward future randomized, controlled trials. “What’s great about this study is its high degree of collaboration, done with real urgency,” he added. “It’s harder to produce a paper that crosses multiple boundaries, like this one does, than a single-focused piece of work. But we wanted to link all of these threads together.”
The study was supported by the Imperial Biomedical Research Centre and Experimental Cancer Medicine Centre, the National Institute for Health Research, and the U.K. National Health Service’s Accelerated Access Collaborative. Dr. Stebbing has served on scientific advisory boards for Eli Lilly and other companies. Dr. Lenz had no relevant disclosures to report.
SOURCE: Stebbing J et al. Sci Adv. 2020 Nov 13. doi: 10.1126/sciadv.abe4724.
FROM SCIENCE ADVANCES
Combo DAA treatments may benefit patients with resistant HCV genotype 3
Patients with hepatitis C virus (HCV) genotype 3 infection have shown resistance to direct-acting antiviral (DAA) treatments. However, a meta-analysis of 34 research reports found that DAA combo treatment can be effective in achieving sustained virologic response (SVR) in patients with HCV genotype 3, according to a study published online in Annals of Hepatology.
This study aimed to analyze the effectiveness of four regimens: sofosbuvir (SOF)/daclatasvir (DCV) with or without ribavirin (RBV); SOF/velpatasvir (VEL) with or without RBV; SOF/VEL/voxilaprevir (VOX);and glecaprevir (GLE)/pibrentasvir (PIB) in the treatment of HCV genotype 3–infected patients in real-world situations, according to Liwei Zhuang, of Beijing Ditan Hospital, Capital Medical University, and colleagues.
A total of 34 studies, comprising 7,328 patients from 22 countries, met the inclusion criteria and formed the basis of the analysis.
Promising results
The pooled SVR rate after 12 or 24 weeks of treatment for the four regimens was 92.1%.
For each regimen, the SVR rate was 91.2% in patients treated with SOF/DCV with or without RBV; 95.1% in patients treated with SOF/VEL with or without RBV; 85.0% in patients treated with SOF/VEL/VOX; and 98.5% in patients treated with GLE/PIB.
In addition, the pooled SVR rate of the four regimens was 95.2% in patients without cirrhosis and 89.4% in patients with cirrhosis, and the pooled SVR rate was 94.4% in treatment-naive patients and 88.0% in treatment-experienced patients. All results were within 95% confidence intervals.
The researchers pointed out that their meta-analysis had limitations. “We think that no strong conclusions can be drawn due to high heterogeneity in four DAA regimens administration in real-world setting from 22 countries, as well as small numbers of patients treated with SOF + VEL + VOX and GLE + PIB. More studies are needed in the future in order to better analyze the antiviral effectiveness of DAAs in GT3 HCV patients in real-world studies,” they authors stated.
However, they also concluded that “the antiviral effectiveness of treatment regimens for HCV-GT3 [genotype 3] infection, including SOF + DCV ± RBV, SOF + VEL ± RBV, GLE + PIB, and SOF + VEL + VOX, was good. The SVR rate of GLE + PIB was higher, and the treatment duration was shorter than other regimens.”
The study was funded by the Chinese government and public institutions. The authors reported that they had no conflicts of interest.
SOURCE: Zhuang L et al. Ann Hepatol. 2020 Oct 12. doi: 10.1016/j.aohep.2020.09.012.
Patients with hepatitis C virus (HCV) genotype 3 infection have shown resistance to direct-acting antiviral (DAA) treatments. However, a meta-analysis of 34 research reports found that DAA combo treatment can be effective in achieving sustained virologic response (SVR) in patients with HCV genotype 3, according to a study published online in Annals of Hepatology.
This study aimed to analyze the effectiveness of four regimens: sofosbuvir (SOF)/daclatasvir (DCV) with or without ribavirin (RBV); SOF/velpatasvir (VEL) with or without RBV; SOF/VEL/voxilaprevir (VOX);and glecaprevir (GLE)/pibrentasvir (PIB) in the treatment of HCV genotype 3–infected patients in real-world situations, according to Liwei Zhuang, of Beijing Ditan Hospital, Capital Medical University, and colleagues.
A total of 34 studies, comprising 7,328 patients from 22 countries, met the inclusion criteria and formed the basis of the analysis.
Promising results
The pooled SVR rate after 12 or 24 weeks of treatment for the four regimens was 92.1%.
For each regimen, the SVR rate was 91.2% in patients treated with SOF/DCV with or without RBV; 95.1% in patients treated with SOF/VEL with or without RBV; 85.0% in patients treated with SOF/VEL/VOX; and 98.5% in patients treated with GLE/PIB.
In addition, the pooled SVR rate of the four regimens was 95.2% in patients without cirrhosis and 89.4% in patients with cirrhosis, and the pooled SVR rate was 94.4% in treatment-naive patients and 88.0% in treatment-experienced patients. All results were within 95% confidence intervals.
The researchers pointed out that their meta-analysis had limitations. “We think that no strong conclusions can be drawn due to high heterogeneity in four DAA regimens administration in real-world setting from 22 countries, as well as small numbers of patients treated with SOF + VEL + VOX and GLE + PIB. More studies are needed in the future in order to better analyze the antiviral effectiveness of DAAs in GT3 HCV patients in real-world studies,” they authors stated.
However, they also concluded that “the antiviral effectiveness of treatment regimens for HCV-GT3 [genotype 3] infection, including SOF + DCV ± RBV, SOF + VEL ± RBV, GLE + PIB, and SOF + VEL + VOX, was good. The SVR rate of GLE + PIB was higher, and the treatment duration was shorter than other regimens.”
The study was funded by the Chinese government and public institutions. The authors reported that they had no conflicts of interest.
SOURCE: Zhuang L et al. Ann Hepatol. 2020 Oct 12. doi: 10.1016/j.aohep.2020.09.012.
Patients with hepatitis C virus (HCV) genotype 3 infection have shown resistance to direct-acting antiviral (DAA) treatments. However, a meta-analysis of 34 research reports found that DAA combo treatment can be effective in achieving sustained virologic response (SVR) in patients with HCV genotype 3, according to a study published online in Annals of Hepatology.
This study aimed to analyze the effectiveness of four regimens: sofosbuvir (SOF)/daclatasvir (DCV) with or without ribavirin (RBV); SOF/velpatasvir (VEL) with or without RBV; SOF/VEL/voxilaprevir (VOX);and glecaprevir (GLE)/pibrentasvir (PIB) in the treatment of HCV genotype 3–infected patients in real-world situations, according to Liwei Zhuang, of Beijing Ditan Hospital, Capital Medical University, and colleagues.
A total of 34 studies, comprising 7,328 patients from 22 countries, met the inclusion criteria and formed the basis of the analysis.
Promising results
The pooled SVR rate after 12 or 24 weeks of treatment for the four regimens was 92.1%.
For each regimen, the SVR rate was 91.2% in patients treated with SOF/DCV with or without RBV; 95.1% in patients treated with SOF/VEL with or without RBV; 85.0% in patients treated with SOF/VEL/VOX; and 98.5% in patients treated with GLE/PIB.
In addition, the pooled SVR rate of the four regimens was 95.2% in patients without cirrhosis and 89.4% in patients with cirrhosis, and the pooled SVR rate was 94.4% in treatment-naive patients and 88.0% in treatment-experienced patients. All results were within 95% confidence intervals.
The researchers pointed out that their meta-analysis had limitations. “We think that no strong conclusions can be drawn due to high heterogeneity in four DAA regimens administration in real-world setting from 22 countries, as well as small numbers of patients treated with SOF + VEL + VOX and GLE + PIB. More studies are needed in the future in order to better analyze the antiviral effectiveness of DAAs in GT3 HCV patients in real-world studies,” they authors stated.
However, they also concluded that “the antiviral effectiveness of treatment regimens for HCV-GT3 [genotype 3] infection, including SOF + DCV ± RBV, SOF + VEL ± RBV, GLE + PIB, and SOF + VEL + VOX, was good. The SVR rate of GLE + PIB was higher, and the treatment duration was shorter than other regimens.”
The study was funded by the Chinese government and public institutions. The authors reported that they had no conflicts of interest.
SOURCE: Zhuang L et al. Ann Hepatol. 2020 Oct 12. doi: 10.1016/j.aohep.2020.09.012.
FROM ANNALS OF HEPATOLOGY
Antidepressant shows early promise for mild COVID-19
Early treatment with the antidepressant fluvoxamine (Luvox) may help prevent respiratory deterioration in patients with mild symptomatic COVID-19, results of a preliminary randomized controlled trial suggest.
In the trial, none of the patients who took fluvoxamine within 7 days of first symptoms developed serious breathing difficulties or required hospitalization for respiratory deterioration.
“Most investigational treatments for COVID-19 have been aimed at the very sickest patients, but it’s also important to find therapies that prevent patients from getting sick enough to require supplemental oxygen or to have to go to the hospital,” study investigator Eric J. Lenze, MD, professor of psychiatry and director of the Healthy Mind Lab at Washington University, St. Louis, said in a statement.
“Our study suggests fluvoxamine may help fill that niche,” Lenze added.
The study was published online Nov. 12 in the JAMA.
Antiviral effects?
The study included 152 nonhospitalized adults (mean age, 46 years; 72% women) with confirmed SARS-CoV-2 infection and mild COVID-19 symptoms starting within 7 days and oxygen saturation of 92% or greater.
Eighty were randomly assigned to 100 mg of fluvoxamine three times daily for 15 days and 72 to matching placebo.
The primary outcome was clinical deterioration within 15 days of randomization defined by meeting two criteria. These included shortness of breath or hospitalization for shortness of breath or pneumonia and oxygen saturation <92% on room air or need for supplemental oxygen to achieve oxygen saturation of 92% or greater.
Clinical deterioration occurred in none of the 80 patients taking fluvoxamine compared with 6 of 72 (8.3%) patients taking placebo, an absolute difference of 8.7% (95% confidence interval, 1.8%-16.4%).
Clinical deterioration in the placebo group happened from 1 to 7 days after randomization and from 3 to 12 days after the onset of COVID-19 symptoms. Four of the 6 patients with clinical deterioration were admitted to the hospital for 4-21 days. One patient required mechanical ventilation for 10 days. No patients died.
Hypothesis generating
The authors cautioned that the study was small and with short follow-up and that the findings “need to be interpreted as hypothesis generating rather than as a demonstration of efficacy.”
However, they noted, if the drug turns out to be effective for COVID-19, the potential advantages of fluvoxamine for outpatient use include its safety, widespread availability, low cost, and oral administration.
Carolyn Machamer, PhD, member of the COVID-19 Early Treatment Fund (CETF) scientific advisory board, which funded the study, noted that there are several reasons fluvoxamine might be helpful in COVID-19.
“The preliminary data suggest the mechanism involves activation of the sigma-1 receptor, which has a number of documented activities. One strong possibility is that activation dampens cytokine release and thus the inflammatory response,” she said in an interview.
“Other possible mechanisms can include inhibition of platelet activation and modulation of autophagy. Coronaviruses usurp some autophagy machinery to remodel membranes for replicating their genomes, so this last mechanism might actually be antiviral,” said Dr. Machamer.
She added that a much larger trial is “crucial to see if the initial striking results can be reproduced, and the Healthy Mind Lab and CETF are currently coordinating these next steps.”
The editors of JAMA published an “Editor’s Note” with the study. In it, they wrote the pilot study addresses a “critically important question during the pandemic of how to prevent individuals who acquire COVID-19 from deteriorating to serious illness. If an effective treatment is found for this key gap in treatment, it will affect the health of millions of people worldwide.”
However, the study has “important limitations, and the findings should be interpreted as only hypothesis generating; they should not be used as the basis for current treatment decisions,” cautioned authors Christopher Seymour, MD, Howard Bauchner, MD, and Robert Golub, MD.
This study was supported by the Taylor Family Institute for Innovative Psychiatric Treatment at Washington University and the CETF. Additional support was provided by the Center for Brain Research in Mood Disorders at Washington University, the Bantly Foundation, and the National Institutes of Health.
Dr. Lenze has received grants from the Patient-Centered Outcomes Research Institute, Takeda, Alkermes, Janssen, Acadia, and the Barnes Jewish Hospital Foundation and has received consulting fees from Janssen and Jazz Pharmaceuticals. Dr. Machamer has disclosed no relevant financial relationships. Dr. Seymour has received grants from the National Institutes of Health and personal fees from Beckman Coulter and Edwards Lifesciences.
A version of this article originally appeared on Medscape.com.
Early treatment with the antidepressant fluvoxamine (Luvox) may help prevent respiratory deterioration in patients with mild symptomatic COVID-19, results of a preliminary randomized controlled trial suggest.
In the trial, none of the patients who took fluvoxamine within 7 days of first symptoms developed serious breathing difficulties or required hospitalization for respiratory deterioration.
“Most investigational treatments for COVID-19 have been aimed at the very sickest patients, but it’s also important to find therapies that prevent patients from getting sick enough to require supplemental oxygen or to have to go to the hospital,” study investigator Eric J. Lenze, MD, professor of psychiatry and director of the Healthy Mind Lab at Washington University, St. Louis, said in a statement.
“Our study suggests fluvoxamine may help fill that niche,” Lenze added.
The study was published online Nov. 12 in the JAMA.
Antiviral effects?
The study included 152 nonhospitalized adults (mean age, 46 years; 72% women) with confirmed SARS-CoV-2 infection and mild COVID-19 symptoms starting within 7 days and oxygen saturation of 92% or greater.
Eighty were randomly assigned to 100 mg of fluvoxamine three times daily for 15 days and 72 to matching placebo.
The primary outcome was clinical deterioration within 15 days of randomization defined by meeting two criteria. These included shortness of breath or hospitalization for shortness of breath or pneumonia and oxygen saturation <92% on room air or need for supplemental oxygen to achieve oxygen saturation of 92% or greater.
Clinical deterioration occurred in none of the 80 patients taking fluvoxamine compared with 6 of 72 (8.3%) patients taking placebo, an absolute difference of 8.7% (95% confidence interval, 1.8%-16.4%).
Clinical deterioration in the placebo group happened from 1 to 7 days after randomization and from 3 to 12 days after the onset of COVID-19 symptoms. Four of the 6 patients with clinical deterioration were admitted to the hospital for 4-21 days. One patient required mechanical ventilation for 10 days. No patients died.
Hypothesis generating
The authors cautioned that the study was small and with short follow-up and that the findings “need to be interpreted as hypothesis generating rather than as a demonstration of efficacy.”
However, they noted, if the drug turns out to be effective for COVID-19, the potential advantages of fluvoxamine for outpatient use include its safety, widespread availability, low cost, and oral administration.
Carolyn Machamer, PhD, member of the COVID-19 Early Treatment Fund (CETF) scientific advisory board, which funded the study, noted that there are several reasons fluvoxamine might be helpful in COVID-19.
“The preliminary data suggest the mechanism involves activation of the sigma-1 receptor, which has a number of documented activities. One strong possibility is that activation dampens cytokine release and thus the inflammatory response,” she said in an interview.
“Other possible mechanisms can include inhibition of platelet activation and modulation of autophagy. Coronaviruses usurp some autophagy machinery to remodel membranes for replicating their genomes, so this last mechanism might actually be antiviral,” said Dr. Machamer.
She added that a much larger trial is “crucial to see if the initial striking results can be reproduced, and the Healthy Mind Lab and CETF are currently coordinating these next steps.”
The editors of JAMA published an “Editor’s Note” with the study. In it, they wrote the pilot study addresses a “critically important question during the pandemic of how to prevent individuals who acquire COVID-19 from deteriorating to serious illness. If an effective treatment is found for this key gap in treatment, it will affect the health of millions of people worldwide.”
However, the study has “important limitations, and the findings should be interpreted as only hypothesis generating; they should not be used as the basis for current treatment decisions,” cautioned authors Christopher Seymour, MD, Howard Bauchner, MD, and Robert Golub, MD.
This study was supported by the Taylor Family Institute for Innovative Psychiatric Treatment at Washington University and the CETF. Additional support was provided by the Center for Brain Research in Mood Disorders at Washington University, the Bantly Foundation, and the National Institutes of Health.
Dr. Lenze has received grants from the Patient-Centered Outcomes Research Institute, Takeda, Alkermes, Janssen, Acadia, and the Barnes Jewish Hospital Foundation and has received consulting fees from Janssen and Jazz Pharmaceuticals. Dr. Machamer has disclosed no relevant financial relationships. Dr. Seymour has received grants from the National Institutes of Health and personal fees from Beckman Coulter and Edwards Lifesciences.
A version of this article originally appeared on Medscape.com.
Early treatment with the antidepressant fluvoxamine (Luvox) may help prevent respiratory deterioration in patients with mild symptomatic COVID-19, results of a preliminary randomized controlled trial suggest.
In the trial, none of the patients who took fluvoxamine within 7 days of first symptoms developed serious breathing difficulties or required hospitalization for respiratory deterioration.
“Most investigational treatments for COVID-19 have been aimed at the very sickest patients, but it’s also important to find therapies that prevent patients from getting sick enough to require supplemental oxygen or to have to go to the hospital,” study investigator Eric J. Lenze, MD, professor of psychiatry and director of the Healthy Mind Lab at Washington University, St. Louis, said in a statement.
“Our study suggests fluvoxamine may help fill that niche,” Lenze added.
The study was published online Nov. 12 in the JAMA.
Antiviral effects?
The study included 152 nonhospitalized adults (mean age, 46 years; 72% women) with confirmed SARS-CoV-2 infection and mild COVID-19 symptoms starting within 7 days and oxygen saturation of 92% or greater.
Eighty were randomly assigned to 100 mg of fluvoxamine three times daily for 15 days and 72 to matching placebo.
The primary outcome was clinical deterioration within 15 days of randomization defined by meeting two criteria. These included shortness of breath or hospitalization for shortness of breath or pneumonia and oxygen saturation <92% on room air or need for supplemental oxygen to achieve oxygen saturation of 92% or greater.
Clinical deterioration occurred in none of the 80 patients taking fluvoxamine compared with 6 of 72 (8.3%) patients taking placebo, an absolute difference of 8.7% (95% confidence interval, 1.8%-16.4%).
Clinical deterioration in the placebo group happened from 1 to 7 days after randomization and from 3 to 12 days after the onset of COVID-19 symptoms. Four of the 6 patients with clinical deterioration were admitted to the hospital for 4-21 days. One patient required mechanical ventilation for 10 days. No patients died.
Hypothesis generating
The authors cautioned that the study was small and with short follow-up and that the findings “need to be interpreted as hypothesis generating rather than as a demonstration of efficacy.”
However, they noted, if the drug turns out to be effective for COVID-19, the potential advantages of fluvoxamine for outpatient use include its safety, widespread availability, low cost, and oral administration.
Carolyn Machamer, PhD, member of the COVID-19 Early Treatment Fund (CETF) scientific advisory board, which funded the study, noted that there are several reasons fluvoxamine might be helpful in COVID-19.
“The preliminary data suggest the mechanism involves activation of the sigma-1 receptor, which has a number of documented activities. One strong possibility is that activation dampens cytokine release and thus the inflammatory response,” she said in an interview.
“Other possible mechanisms can include inhibition of platelet activation and modulation of autophagy. Coronaviruses usurp some autophagy machinery to remodel membranes for replicating their genomes, so this last mechanism might actually be antiviral,” said Dr. Machamer.
She added that a much larger trial is “crucial to see if the initial striking results can be reproduced, and the Healthy Mind Lab and CETF are currently coordinating these next steps.”
The editors of JAMA published an “Editor’s Note” with the study. In it, they wrote the pilot study addresses a “critically important question during the pandemic of how to prevent individuals who acquire COVID-19 from deteriorating to serious illness. If an effective treatment is found for this key gap in treatment, it will affect the health of millions of people worldwide.”
However, the study has “important limitations, and the findings should be interpreted as only hypothesis generating; they should not be used as the basis for current treatment decisions,” cautioned authors Christopher Seymour, MD, Howard Bauchner, MD, and Robert Golub, MD.
This study was supported by the Taylor Family Institute for Innovative Psychiatric Treatment at Washington University and the CETF. Additional support was provided by the Center for Brain Research in Mood Disorders at Washington University, the Bantly Foundation, and the National Institutes of Health.
Dr. Lenze has received grants from the Patient-Centered Outcomes Research Institute, Takeda, Alkermes, Janssen, Acadia, and the Barnes Jewish Hospital Foundation and has received consulting fees from Janssen and Jazz Pharmaceuticals. Dr. Machamer has disclosed no relevant financial relationships. Dr. Seymour has received grants from the National Institutes of Health and personal fees from Beckman Coulter and Edwards Lifesciences.
A version of this article originally appeared on Medscape.com.
Recurrent Cutaneous Exophiala Phaeohyphomycosis in an Immunosuppressed Patient
To the Editor:
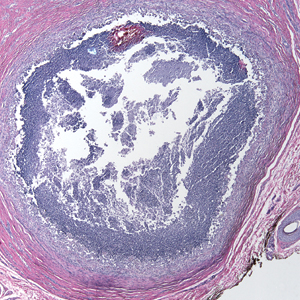
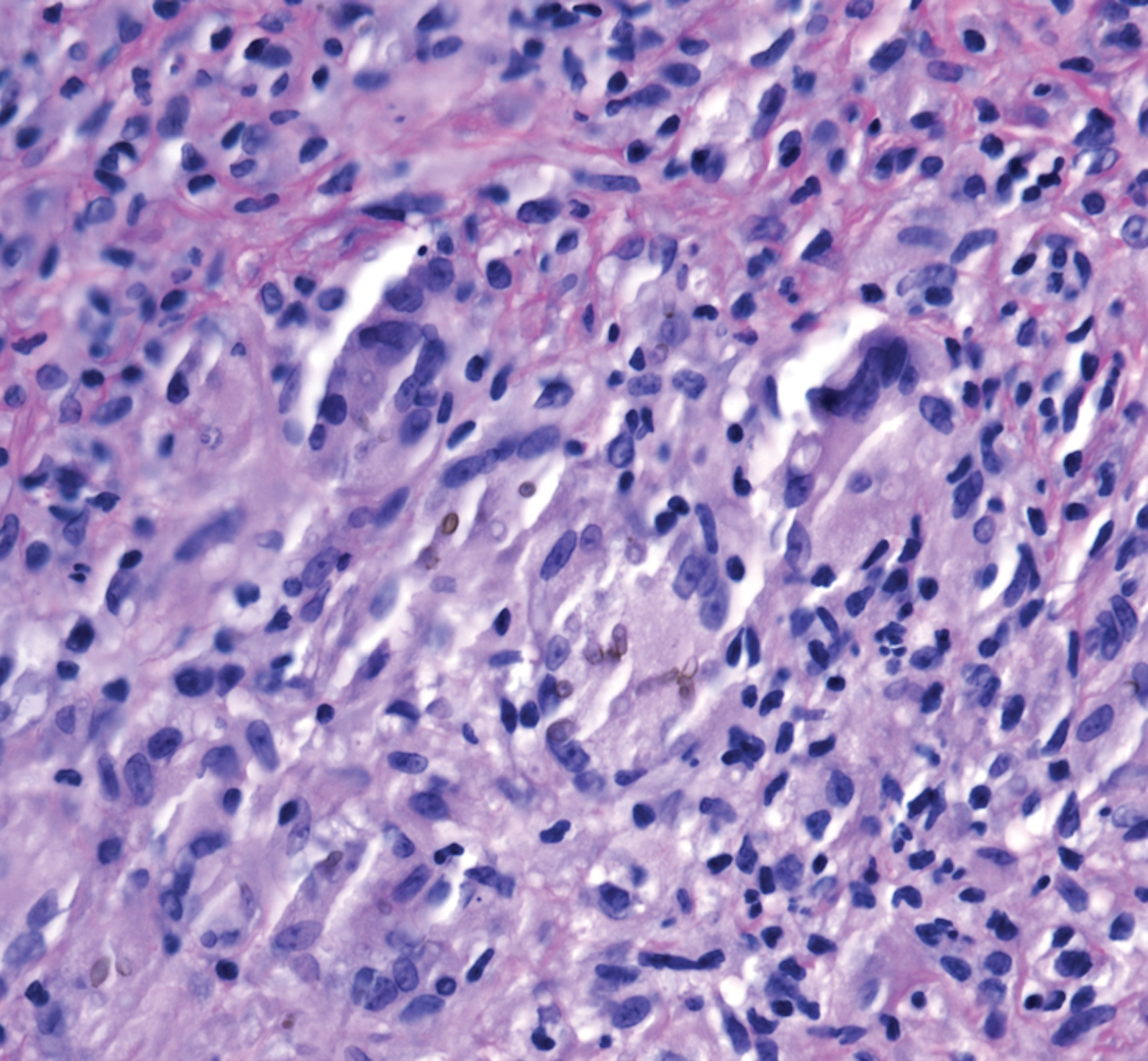
A 73-year-old man presented with a 2.5-cm, recurrent, fluctuant, multiloculated nodule on the left forearm. The lesion was nontender with occasional chalky, white to yellow discharge from multiple sinus tracts. He was otherwise well appearing without signs of systemic infection. He reported similar lesions in slightly different anatomic locations on the left forearm both 7 and 4 years prior to the current presentation. In both instances, the nodules were excised at an outside hospital without any additional treatment. Histopathology of the excised tissue from both prior occasions demonstrated brown septate hyphae surrounded by suppurative and granulomatous inflammation consistent with dematiaceous fungal infection of the dermis (Figures 1 and 2); the organisms were highlighted with periodic acid–Schiff stain.

The patient’s medical history was notable for advanced heart failure with an ejection fraction of 25% and autosomal-dominant polycystic kidney disease. He received an orthotopic kidney transplant 17 years prior to the current presentation. Medications included tacrolimus, mycophenolate mofetil, and prednisone. He denied any trauma or notable exposures to vegetation, and his travel history was unremarkable. A review of systems was negative.
At the current presentation, a sterile fungal culture was performed and found positive for Exophiala species, while bacterial and mycobacterial cultures were negative. A diagnosis of phaeohyphomycosis was made, and he was scheduled for re-excision. Out of concern for interactions with his immunosuppressive regimen, he chose to forgo any systemic antifungal therapy. He died from hospital-acquired pneumonia and volume overload unresponsive to diuretics or dialysis.
Phaeohyphomycosis is a rare fungal infection caused by several genera of dematiaceous fungi that are characterized by the presence of melaninlike cell wall pigments thought to locally hinder immune clearance by scavenging phagocyte-derived free radicals. These fungi are ubiquitous in soil and vegetation and usually penetrate the skin at sites of minor trauma.1 Phaeohyphomycosis typically affects immunosuppressed hosts, and its incidence among organ transplant recipients currently is 9%.2 The incidence in this population has been rising, however, as recent advances in immunosuppressive therapies have increased posttransplant survival.3
Subcutaneous phaeohyphomycosis can present with nodules, cysts, tumors, and/or verrucous plaques, and the diagnosis almost always requires clinicopathologic correlation.3 Rapid diagnosis can be made when septate brown hyphae and/or yeast forms are observed on hematoxylin and eosin stain. Rarely, patients present with disseminated infection, characterized by fungemia; central nervous system involvement; and/or infection of multiple deep structures including the eyes, lungs, bones, and sinuses.4 The risk for dissemination from the skin likely is related to the culprit organism’s genus; Lomentospora, Cladophialophora, and Verruconis often are associated with dissemination, while Alternaria, Exophiala, and Fonsecaea typically remain confined to the skin and subcutis.5 Due to this difference and its potential to impact management, obtaining a tissue fungal culture is advisable when phaeohyphomycosis is suspected.
There is no standard treatment of phaeohyphomycosis. Regimens typically consist of excision and prolonged courses of azole therapy, though excision alone with close follow-up may be a reasonable alternative.6 The latter is a particularly important consideration when managing phaeohyphomycosis in organ transplant recipients, as azoles are known cytochrome P450 3A4 inhibitors that can affect serum levels of common immunosuppressive medications including calcineurin inhibitors and mammalian target of rapamycin inhibitors.3 Local recurrence is common regardless of whether azole therapy is pursued,7 and dissemination of localized Exophiala infections is exceedingly rare.8 There is a strong argument to be made for our patient’s decision to forgo antifungal therapy.
This case underscores the difficulty inherent to eradicating local subcutaneous Exophiala phaeohyphomycosis while providing reassurance that with treatment, the risk of life-threatening complications is low. Obtaining tissue for both hematoxylin and eosin stain and sterile culture is crucial to ensuring prompt diagnosis and tailoring the optimal treatment and surveillance strategy to the culprit organism. To avoid delays in diagnosis and treatment, it is important for clinicians to consider phaeohyphomycosis in the differential diagnosis for recurrent nodulocystic lesions in immunosuppressed patients and to recognize that presentations may span many years.
- Bhardwaj S, Capoor MR, Kolte S, et al. Phaeohyphomycosis due to Exophiala jeanselmei: an emerging pathogen in India—case report and review. Mycopathologia. 2016;181:279-284.
- Isa-Isa R, Garcia C, Isa M, et al. Subcutaneous phaeohyphomycosis (mycotic cyst). Clin Dermatol. 2012;30:425-431.
- Tirico MCCP, Neto CF, Cruz LL, et al. Clinical spectrum of phaeohyphomycosis in solid organ transplant recipients. JAAD Case Rep. 2016;2:465-469.
- Revankar SG, Patterson JE, Sutton DA, et al. Disseminated phaeohyphomycosis: review of an emerging mycosis. Clin Infect Dis. 2002;34:467-476.
- Revankar SG, Baddley JW, Chen SC-A, et al. A mycoses study group international prospective study of phaeohyphomycosis: an analysis of 99 proven/probable cases. Open Forum Infect Dis. 2017;4:ofx200.
- Oberlin KE, Nichols AJ, Rosa R, et al. Phaeohyphomycosis due to Exophiala infections in solid organ transplant recipients: case report and literature review [published online June 26, 2017]. Transpl Infect Dis. 2017;19. doi:10.1111/tid.12723.
- Shirbur S, Telkar S, Goudar B, et al. Recurrent phaeohyphomycosis: a case report. J Clin Diagn Res. 2013;7:2015-2016.
- Li D-M, Li R-Y, de Hoog GS, et al. Fatal Exophiala infections in China, with a report of seven cases. Mycoses. 2011;54:E136-E142.
To the Editor:
A 73-year-old man presented with a 2.5-cm, recurrent, fluctuant, multiloculated nodule on the left forearm. The lesion was nontender with occasional chalky, white to yellow discharge from multiple sinus tracts. He was otherwise well appearing without signs of systemic infection. He reported similar lesions in slightly different anatomic locations on the left forearm both 7 and 4 years prior to the current presentation. In both instances, the nodules were excised at an outside hospital without any additional treatment. Histopathology of the excised tissue from both prior occasions demonstrated brown septate hyphae surrounded by suppurative and granulomatous inflammation consistent with dematiaceous fungal infection of the dermis (Figures 1 and 2); the organisms were highlighted with periodic acid–Schiff stain.


The patient’s medical history was notable for advanced heart failure with an ejection fraction of 25% and autosomal-dominant polycystic kidney disease. He received an orthotopic kidney transplant 17 years prior to the current presentation. Medications included tacrolimus, mycophenolate mofetil, and prednisone. He denied any trauma or notable exposures to vegetation, and his travel history was unremarkable. A review of systems was negative.
At the current presentation, a sterile fungal culture was performed and found positive for Exophiala species, while bacterial and mycobacterial cultures were negative. A diagnosis of phaeohyphomycosis was made, and he was scheduled for re-excision. Out of concern for interactions with his immunosuppressive regimen, he chose to forgo any systemic antifungal therapy. He died from hospital-acquired pneumonia and volume overload unresponsive to diuretics or dialysis.
Phaeohyphomycosis is a rare fungal infection caused by several genera of dematiaceous fungi that are characterized by the presence of melaninlike cell wall pigments thought to locally hinder immune clearance by scavenging phagocyte-derived free radicals. These fungi are ubiquitous in soil and vegetation and usually penetrate the skin at sites of minor trauma.1 Phaeohyphomycosis typically affects immunosuppressed hosts, and its incidence among organ transplant recipients currently is 9%.2 The incidence in this population has been rising, however, as recent advances in immunosuppressive therapies have increased posttransplant survival.3
Subcutaneous phaeohyphomycosis can present with nodules, cysts, tumors, and/or verrucous plaques, and the diagnosis almost always requires clinicopathologic correlation.3 Rapid diagnosis can be made when septate brown hyphae and/or yeast forms are observed on hematoxylin and eosin stain. Rarely, patients present with disseminated infection, characterized by fungemia; central nervous system involvement; and/or infection of multiple deep structures including the eyes, lungs, bones, and sinuses.4 The risk for dissemination from the skin likely is related to the culprit organism’s genus; Lomentospora, Cladophialophora, and Verruconis often are associated with dissemination, while Alternaria, Exophiala, and Fonsecaea typically remain confined to the skin and subcutis.5 Due to this difference and its potential to impact management, obtaining a tissue fungal culture is advisable when phaeohyphomycosis is suspected.
There is no standard treatment of phaeohyphomycosis. Regimens typically consist of excision and prolonged courses of azole therapy, though excision alone with close follow-up may be a reasonable alternative.6 The latter is a particularly important consideration when managing phaeohyphomycosis in organ transplant recipients, as azoles are known cytochrome P450 3A4 inhibitors that can affect serum levels of common immunosuppressive medications including calcineurin inhibitors and mammalian target of rapamycin inhibitors.3 Local recurrence is common regardless of whether azole therapy is pursued,7 and dissemination of localized Exophiala infections is exceedingly rare.8 There is a strong argument to be made for our patient’s decision to forgo antifungal therapy.
This case underscores the difficulty inherent to eradicating local subcutaneous Exophiala phaeohyphomycosis while providing reassurance that with treatment, the risk of life-threatening complications is low. Obtaining tissue for both hematoxylin and eosin stain and sterile culture is crucial to ensuring prompt diagnosis and tailoring the optimal treatment and surveillance strategy to the culprit organism. To avoid delays in diagnosis and treatment, it is important for clinicians to consider phaeohyphomycosis in the differential diagnosis for recurrent nodulocystic lesions in immunosuppressed patients and to recognize that presentations may span many years.
To the Editor:
A 73-year-old man presented with a 2.5-cm, recurrent, fluctuant, multiloculated nodule on the left forearm. The lesion was nontender with occasional chalky, white to yellow discharge from multiple sinus tracts. He was otherwise well appearing without signs of systemic infection. He reported similar lesions in slightly different anatomic locations on the left forearm both 7 and 4 years prior to the current presentation. In both instances, the nodules were excised at an outside hospital without any additional treatment. Histopathology of the excised tissue from both prior occasions demonstrated brown septate hyphae surrounded by suppurative and granulomatous inflammation consistent with dematiaceous fungal infection of the dermis (Figures 1 and 2); the organisms were highlighted with periodic acid–Schiff stain.


The patient’s medical history was notable for advanced heart failure with an ejection fraction of 25% and autosomal-dominant polycystic kidney disease. He received an orthotopic kidney transplant 17 years prior to the current presentation. Medications included tacrolimus, mycophenolate mofetil, and prednisone. He denied any trauma or notable exposures to vegetation, and his travel history was unremarkable. A review of systems was negative.
At the current presentation, a sterile fungal culture was performed and found positive for Exophiala species, while bacterial and mycobacterial cultures were negative. A diagnosis of phaeohyphomycosis was made, and he was scheduled for re-excision. Out of concern for interactions with his immunosuppressive regimen, he chose to forgo any systemic antifungal therapy. He died from hospital-acquired pneumonia and volume overload unresponsive to diuretics or dialysis.
Phaeohyphomycosis is a rare fungal infection caused by several genera of dematiaceous fungi that are characterized by the presence of melaninlike cell wall pigments thought to locally hinder immune clearance by scavenging phagocyte-derived free radicals. These fungi are ubiquitous in soil and vegetation and usually penetrate the skin at sites of minor trauma.1 Phaeohyphomycosis typically affects immunosuppressed hosts, and its incidence among organ transplant recipients currently is 9%.2 The incidence in this population has been rising, however, as recent advances in immunosuppressive therapies have increased posttransplant survival.3
Subcutaneous phaeohyphomycosis can present with nodules, cysts, tumors, and/or verrucous plaques, and the diagnosis almost always requires clinicopathologic correlation.3 Rapid diagnosis can be made when septate brown hyphae and/or yeast forms are observed on hematoxylin and eosin stain. Rarely, patients present with disseminated infection, characterized by fungemia; central nervous system involvement; and/or infection of multiple deep structures including the eyes, lungs, bones, and sinuses.4 The risk for dissemination from the skin likely is related to the culprit organism’s genus; Lomentospora, Cladophialophora, and Verruconis often are associated with dissemination, while Alternaria, Exophiala, and Fonsecaea typically remain confined to the skin and subcutis.5 Due to this difference and its potential to impact management, obtaining a tissue fungal culture is advisable when phaeohyphomycosis is suspected.
There is no standard treatment of phaeohyphomycosis. Regimens typically consist of excision and prolonged courses of azole therapy, though excision alone with close follow-up may be a reasonable alternative.6 The latter is a particularly important consideration when managing phaeohyphomycosis in organ transplant recipients, as azoles are known cytochrome P450 3A4 inhibitors that can affect serum levels of common immunosuppressive medications including calcineurin inhibitors and mammalian target of rapamycin inhibitors.3 Local recurrence is common regardless of whether azole therapy is pursued,7 and dissemination of localized Exophiala infections is exceedingly rare.8 There is a strong argument to be made for our patient’s decision to forgo antifungal therapy.
This case underscores the difficulty inherent to eradicating local subcutaneous Exophiala phaeohyphomycosis while providing reassurance that with treatment, the risk of life-threatening complications is low. Obtaining tissue for both hematoxylin and eosin stain and sterile culture is crucial to ensuring prompt diagnosis and tailoring the optimal treatment and surveillance strategy to the culprit organism. To avoid delays in diagnosis and treatment, it is important for clinicians to consider phaeohyphomycosis in the differential diagnosis for recurrent nodulocystic lesions in immunosuppressed patients and to recognize that presentations may span many years.
- Bhardwaj S, Capoor MR, Kolte S, et al. Phaeohyphomycosis due to Exophiala jeanselmei: an emerging pathogen in India—case report and review. Mycopathologia. 2016;181:279-284.
- Isa-Isa R, Garcia C, Isa M, et al. Subcutaneous phaeohyphomycosis (mycotic cyst). Clin Dermatol. 2012;30:425-431.
- Tirico MCCP, Neto CF, Cruz LL, et al. Clinical spectrum of phaeohyphomycosis in solid organ transplant recipients. JAAD Case Rep. 2016;2:465-469.
- Revankar SG, Patterson JE, Sutton DA, et al. Disseminated phaeohyphomycosis: review of an emerging mycosis. Clin Infect Dis. 2002;34:467-476.
- Revankar SG, Baddley JW, Chen SC-A, et al. A mycoses study group international prospective study of phaeohyphomycosis: an analysis of 99 proven/probable cases. Open Forum Infect Dis. 2017;4:ofx200.
- Oberlin KE, Nichols AJ, Rosa R, et al. Phaeohyphomycosis due to Exophiala infections in solid organ transplant recipients: case report and literature review [published online June 26, 2017]. Transpl Infect Dis. 2017;19. doi:10.1111/tid.12723.
- Shirbur S, Telkar S, Goudar B, et al. Recurrent phaeohyphomycosis: a case report. J Clin Diagn Res. 2013;7:2015-2016.
- Li D-M, Li R-Y, de Hoog GS, et al. Fatal Exophiala infections in China, with a report of seven cases. Mycoses. 2011;54:E136-E142.
- Bhardwaj S, Capoor MR, Kolte S, et al. Phaeohyphomycosis due to Exophiala jeanselmei: an emerging pathogen in India—case report and review. Mycopathologia. 2016;181:279-284.
- Isa-Isa R, Garcia C, Isa M, et al. Subcutaneous phaeohyphomycosis (mycotic cyst). Clin Dermatol. 2012;30:425-431.
- Tirico MCCP, Neto CF, Cruz LL, et al. Clinical spectrum of phaeohyphomycosis in solid organ transplant recipients. JAAD Case Rep. 2016;2:465-469.
- Revankar SG, Patterson JE, Sutton DA, et al. Disseminated phaeohyphomycosis: review of an emerging mycosis. Clin Infect Dis. 2002;34:467-476.
- Revankar SG, Baddley JW, Chen SC-A, et al. A mycoses study group international prospective study of phaeohyphomycosis: an analysis of 99 proven/probable cases. Open Forum Infect Dis. 2017;4:ofx200.
- Oberlin KE, Nichols AJ, Rosa R, et al. Phaeohyphomycosis due to Exophiala infections in solid organ transplant recipients: case report and literature review [published online June 26, 2017]. Transpl Infect Dis. 2017;19. doi:10.1111/tid.12723.
- Shirbur S, Telkar S, Goudar B, et al. Recurrent phaeohyphomycosis: a case report. J Clin Diagn Res. 2013;7:2015-2016.
- Li D-M, Li R-Y, de Hoog GS, et al. Fatal Exophiala infections in China, with a report of seven cases. Mycoses. 2011;54:E136-E142.
Practice Points
- Phaeohyphomycosis is an infection with dematiaceous fungi that most commonly affects immunosuppressed patients.
- Subcutaneous phaeohyphomycosis may present with nodulocystic lesions that recur over the course of years.
- Tissue fungal culture should be obtained when the diagnosis is suspected, as the risk for dissemination is related to the culprit organism.
- Surgical excision with close follow-up may be an appropriate management strategy for patients on immunosuppressive medications to avoid interactions with azole therapy.
Pronounced racial differences in HBsAg loss after stopping nucleos(t)ide
Loss of the hepatitis B surface antigen (HBsAg), a marker for functional cure of hepatitis B infection, is nearly six times more common among White patients than Asian patients following cessation of therapy with a nucleotide or nucleoside analogue, investigators in the RETRACT-B study group report.
Among 1,541 patients in a global retrospective cohort, the cumulative rate of HBsAg loss 4 years after cessation of therapy with entecavir (ETV), tenofovir disoproxil fumarate (TDF), or other nucleoside/nucleotide analogue (“nuc” or NA) was 11% in Asian patients, compared with 41% in Whites, which translated in multivariate analysis into a hazard ratio (HR) of 5.8 (P < .001), said Grishma Hirode, a clinical research associate and PhD candidate at the Toronto Centre for Liver Disease.
“On univariate Cox regression, the rate of S [antigen] loss was significantly higher among older patients, among [Whites], and among tenofovir-treated patients prior to stopping,” she said during the virtual annual meeting of the American Association for the Study of Liver Diseases.
Although NAs are effective at suppressing hepatitis B viral activity, functional cure as indicated by HBsAg loss is uncommon, Ms. Hirode noted.
“Finite use of antiviral therapy has been proposed as an alternative to long-term therapy, and the rationale for stopping nuc therapy is to induce a durable virologic remission in the form of an inactive carrier state, and ideally a functional cure,” she said.
The RETRACT-B (Response after End of Treatment with Antivirals in Chronic Hepatitis B) study group, comprising liver treatment centers in Canada, Europe, Hong Kong, and Taiwan, studies outcomes following cessation of nucleos(t)ide analogue therapy.
The investigators looked at data on 1,541 patients, including those with both hepatitis B e-antigen (HBeAg) positive and HBeAg-negative disease at the start of therapy, all of whom were HBeAg negative at the time of antiviral cessation and had undetectable serum HBV DNA. Patients with hepatitis C, hepatitis D and/or HIV co-infection were excluded, as were patients who had received interferon treatment less than 12 months before stopping.
The mean age at baseline was 53 years. Men comprised 73% of the sample. In all, 88% of patients were Asian, 10% White, and 2% other.
In patients for whom genotype data was known, 0.5% had type A, 43% type B, 11% type C, and 2% type D.
Nearly two-thirds of patients (60%) were on ETV at the time of drug cessation, 29% were on TDF, and 11% were on other agents.
In all, 5% of patients had cirrhosis at the time of nucleos(t)ide cessation, the mean HBsAg was 2.6 log10 IU/mL, and the mean alanine aminotransferase (ALT) level was 0.6 times the upper limit of normal.
The median duration of NA therapy was 3 years.
The cumulative rates of HBsAg loss over time among all patients was 3% at 1 year, 8% at 2 years. 12% at 3 years, and 14% at 4 years. Cumulative rates of antigen loss at year 4 were significantly greater for patients 50 and older vs. those younger than 50 (18% vs. 9%, respectively, P = .01), Whites vs. Asians (41% vs. 11%, P < .001), and in those who had been on TDF vs. ETV (17% vs. 12%, P = .001). There was no significant difference in cumulative HBsAg loss between patients who were HBeAg positive or negative at the start of NA therapy.
Cumulative rates of retreatment were 30% at 1 year, 43% at 2 years, 50% at 3 years, and 56% at 4 years. The only significant predictor for retreatment was age, with patients 50 and older being significantly more likely to be retreated by year 4 (63% vs. 45%, respectively, P < .001).
In a univariate model for HBsAg loss, the HR for age 50 and older was 1.7 (P = .01), the HR for White vs. Asian patients was 5.5 (P < .001), and the HR for TDF vs. ETV was 2.0 (P = .001).
A univariate model for retreatment showed an HR of 1.6 for patients 50 and older; all other parameters (sex, race, NA type, and HBeAg status at start of therapy) were not significantly different.
In multivariate models, only race/ethnicity remained significant as a predictor for HBsAg loss, with a HR of 5.8 for Whites vs. Asians (P < .001), and only age 50 and older remained significant as a predictor for retreatment, with a HR of 1.6 (P < .001).
The 4-year cumulative rate of virologic relapse, defined as an HBV DNA of 2000 IU/mL or higher) was 74%, the rate of combined DNA plus ALT relapse (ALT 2 or more times the upper limit of normal) was 56%, and the rate of ALT flares (5 or more times the upper limit of normal) was 33%.
In all, 15 patients (1%) experienced hepatic decompensation, and 12 (0.96%) died, with 9 of the deaths reported as liver-related.
Race/ethnicity differences previously seen
Liver specialist Anna Suk-Fong Lok, MD, professor of medicine at the University of Michigan in Ann Arbor, who was not involved in the study, said that the findings are not especially surprising.
“When the studies came out from Asian countries showing that patients who were taken off treatment had a higher rate of S antigen loss than patients who stayed on treatment, the rate of S antigen loss was not all that impressive, but when you look at the European studies the rate of S antigen loss was very high,” she said in an interview.
“The question of course is ‘Why?’ I don’t think we understand completely why. We can speculate, but none of these type studies give us a definitive answer,” she said.
Possible reasons for the racial differences in HBsAg loss include differences in hepatitis B genotype, she said.
“Another possibility is that Asian patients may have been infected either at the time of birth or as a young kid, so they may have been infected for a much longer period of time than [Whites], who usually acquire infections as adults,” Dr. Lok said.
There may also be differences between patient populations in immune responses following cessation of antiviral therapy, she added.
The study was supported by the RETRACT-B group. Ms. Hirode and Dr. Lok reported no relevant disclosures.
SOURCE: Hirode G et al. AASLD 2020. Abstract 23.
Loss of the hepatitis B surface antigen (HBsAg), a marker for functional cure of hepatitis B infection, is nearly six times more common among White patients than Asian patients following cessation of therapy with a nucleotide or nucleoside analogue, investigators in the RETRACT-B study group report.
Among 1,541 patients in a global retrospective cohort, the cumulative rate of HBsAg loss 4 years after cessation of therapy with entecavir (ETV), tenofovir disoproxil fumarate (TDF), or other nucleoside/nucleotide analogue (“nuc” or NA) was 11% in Asian patients, compared with 41% in Whites, which translated in multivariate analysis into a hazard ratio (HR) of 5.8 (P < .001), said Grishma Hirode, a clinical research associate and PhD candidate at the Toronto Centre for Liver Disease.
“On univariate Cox regression, the rate of S [antigen] loss was significantly higher among older patients, among [Whites], and among tenofovir-treated patients prior to stopping,” she said during the virtual annual meeting of the American Association for the Study of Liver Diseases.
Although NAs are effective at suppressing hepatitis B viral activity, functional cure as indicated by HBsAg loss is uncommon, Ms. Hirode noted.
“Finite use of antiviral therapy has been proposed as an alternative to long-term therapy, and the rationale for stopping nuc therapy is to induce a durable virologic remission in the form of an inactive carrier state, and ideally a functional cure,” she said.
The RETRACT-B (Response after End of Treatment with Antivirals in Chronic Hepatitis B) study group, comprising liver treatment centers in Canada, Europe, Hong Kong, and Taiwan, studies outcomes following cessation of nucleos(t)ide analogue therapy.
The investigators looked at data on 1,541 patients, including those with both hepatitis B e-antigen (HBeAg) positive and HBeAg-negative disease at the start of therapy, all of whom were HBeAg negative at the time of antiviral cessation and had undetectable serum HBV DNA. Patients with hepatitis C, hepatitis D and/or HIV co-infection were excluded, as were patients who had received interferon treatment less than 12 months before stopping.
The mean age at baseline was 53 years. Men comprised 73% of the sample. In all, 88% of patients were Asian, 10% White, and 2% other.
In patients for whom genotype data was known, 0.5% had type A, 43% type B, 11% type C, and 2% type D.
Nearly two-thirds of patients (60%) were on ETV at the time of drug cessation, 29% were on TDF, and 11% were on other agents.
In all, 5% of patients had cirrhosis at the time of nucleos(t)ide cessation, the mean HBsAg was 2.6 log10 IU/mL, and the mean alanine aminotransferase (ALT) level was 0.6 times the upper limit of normal.
The median duration of NA therapy was 3 years.
The cumulative rates of HBsAg loss over time among all patients was 3% at 1 year, 8% at 2 years. 12% at 3 years, and 14% at 4 years. Cumulative rates of antigen loss at year 4 were significantly greater for patients 50 and older vs. those younger than 50 (18% vs. 9%, respectively, P = .01), Whites vs. Asians (41% vs. 11%, P < .001), and in those who had been on TDF vs. ETV (17% vs. 12%, P = .001). There was no significant difference in cumulative HBsAg loss between patients who were HBeAg positive or negative at the start of NA therapy.
Cumulative rates of retreatment were 30% at 1 year, 43% at 2 years, 50% at 3 years, and 56% at 4 years. The only significant predictor for retreatment was age, with patients 50 and older being significantly more likely to be retreated by year 4 (63% vs. 45%, respectively, P < .001).
In a univariate model for HBsAg loss, the HR for age 50 and older was 1.7 (P = .01), the HR for White vs. Asian patients was 5.5 (P < .001), and the HR for TDF vs. ETV was 2.0 (P = .001).
A univariate model for retreatment showed an HR of 1.6 for patients 50 and older; all other parameters (sex, race, NA type, and HBeAg status at start of therapy) were not significantly different.
In multivariate models, only race/ethnicity remained significant as a predictor for HBsAg loss, with a HR of 5.8 for Whites vs. Asians (P < .001), and only age 50 and older remained significant as a predictor for retreatment, with a HR of 1.6 (P < .001).
The 4-year cumulative rate of virologic relapse, defined as an HBV DNA of 2000 IU/mL or higher) was 74%, the rate of combined DNA plus ALT relapse (ALT 2 or more times the upper limit of normal) was 56%, and the rate of ALT flares (5 or more times the upper limit of normal) was 33%.
In all, 15 patients (1%) experienced hepatic decompensation, and 12 (0.96%) died, with 9 of the deaths reported as liver-related.
Race/ethnicity differences previously seen
Liver specialist Anna Suk-Fong Lok, MD, professor of medicine at the University of Michigan in Ann Arbor, who was not involved in the study, said that the findings are not especially surprising.
“When the studies came out from Asian countries showing that patients who were taken off treatment had a higher rate of S antigen loss than patients who stayed on treatment, the rate of S antigen loss was not all that impressive, but when you look at the European studies the rate of S antigen loss was very high,” she said in an interview.
“The question of course is ‘Why?’ I don’t think we understand completely why. We can speculate, but none of these type studies give us a definitive answer,” she said.
Possible reasons for the racial differences in HBsAg loss include differences in hepatitis B genotype, she said.
“Another possibility is that Asian patients may have been infected either at the time of birth or as a young kid, so they may have been infected for a much longer period of time than [Whites], who usually acquire infections as adults,” Dr. Lok said.
There may also be differences between patient populations in immune responses following cessation of antiviral therapy, she added.
The study was supported by the RETRACT-B group. Ms. Hirode and Dr. Lok reported no relevant disclosures.
SOURCE: Hirode G et al. AASLD 2020. Abstract 23.
Loss of the hepatitis B surface antigen (HBsAg), a marker for functional cure of hepatitis B infection, is nearly six times more common among White patients than Asian patients following cessation of therapy with a nucleotide or nucleoside analogue, investigators in the RETRACT-B study group report.
Among 1,541 patients in a global retrospective cohort, the cumulative rate of HBsAg loss 4 years after cessation of therapy with entecavir (ETV), tenofovir disoproxil fumarate (TDF), or other nucleoside/nucleotide analogue (“nuc” or NA) was 11% in Asian patients, compared with 41% in Whites, which translated in multivariate analysis into a hazard ratio (HR) of 5.8 (P < .001), said Grishma Hirode, a clinical research associate and PhD candidate at the Toronto Centre for Liver Disease.
“On univariate Cox regression, the rate of S [antigen] loss was significantly higher among older patients, among [Whites], and among tenofovir-treated patients prior to stopping,” she said during the virtual annual meeting of the American Association for the Study of Liver Diseases.
Although NAs are effective at suppressing hepatitis B viral activity, functional cure as indicated by HBsAg loss is uncommon, Ms. Hirode noted.
“Finite use of antiviral therapy has been proposed as an alternative to long-term therapy, and the rationale for stopping nuc therapy is to induce a durable virologic remission in the form of an inactive carrier state, and ideally a functional cure,” she said.
The RETRACT-B (Response after End of Treatment with Antivirals in Chronic Hepatitis B) study group, comprising liver treatment centers in Canada, Europe, Hong Kong, and Taiwan, studies outcomes following cessation of nucleos(t)ide analogue therapy.
The investigators looked at data on 1,541 patients, including those with both hepatitis B e-antigen (HBeAg) positive and HBeAg-negative disease at the start of therapy, all of whom were HBeAg negative at the time of antiviral cessation and had undetectable serum HBV DNA. Patients with hepatitis C, hepatitis D and/or HIV co-infection were excluded, as were patients who had received interferon treatment less than 12 months before stopping.
The mean age at baseline was 53 years. Men comprised 73% of the sample. In all, 88% of patients were Asian, 10% White, and 2% other.
In patients for whom genotype data was known, 0.5% had type A, 43% type B, 11% type C, and 2% type D.
Nearly two-thirds of patients (60%) were on ETV at the time of drug cessation, 29% were on TDF, and 11% were on other agents.
In all, 5% of patients had cirrhosis at the time of nucleos(t)ide cessation, the mean HBsAg was 2.6 log10 IU/mL, and the mean alanine aminotransferase (ALT) level was 0.6 times the upper limit of normal.
The median duration of NA therapy was 3 years.
The cumulative rates of HBsAg loss over time among all patients was 3% at 1 year, 8% at 2 years. 12% at 3 years, and 14% at 4 years. Cumulative rates of antigen loss at year 4 were significantly greater for patients 50 and older vs. those younger than 50 (18% vs. 9%, respectively, P = .01), Whites vs. Asians (41% vs. 11%, P < .001), and in those who had been on TDF vs. ETV (17% vs. 12%, P = .001). There was no significant difference in cumulative HBsAg loss between patients who were HBeAg positive or negative at the start of NA therapy.
Cumulative rates of retreatment were 30% at 1 year, 43% at 2 years, 50% at 3 years, and 56% at 4 years. The only significant predictor for retreatment was age, with patients 50 and older being significantly more likely to be retreated by year 4 (63% vs. 45%, respectively, P < .001).
In a univariate model for HBsAg loss, the HR for age 50 and older was 1.7 (P = .01), the HR for White vs. Asian patients was 5.5 (P < .001), and the HR for TDF vs. ETV was 2.0 (P = .001).
A univariate model for retreatment showed an HR of 1.6 for patients 50 and older; all other parameters (sex, race, NA type, and HBeAg status at start of therapy) were not significantly different.
In multivariate models, only race/ethnicity remained significant as a predictor for HBsAg loss, with a HR of 5.8 for Whites vs. Asians (P < .001), and only age 50 and older remained significant as a predictor for retreatment, with a HR of 1.6 (P < .001).
The 4-year cumulative rate of virologic relapse, defined as an HBV DNA of 2000 IU/mL or higher) was 74%, the rate of combined DNA plus ALT relapse (ALT 2 or more times the upper limit of normal) was 56%, and the rate of ALT flares (5 or more times the upper limit of normal) was 33%.
In all, 15 patients (1%) experienced hepatic decompensation, and 12 (0.96%) died, with 9 of the deaths reported as liver-related.
Race/ethnicity differences previously seen
Liver specialist Anna Suk-Fong Lok, MD, professor of medicine at the University of Michigan in Ann Arbor, who was not involved in the study, said that the findings are not especially surprising.
“When the studies came out from Asian countries showing that patients who were taken off treatment had a higher rate of S antigen loss than patients who stayed on treatment, the rate of S antigen loss was not all that impressive, but when you look at the European studies the rate of S antigen loss was very high,” she said in an interview.
“The question of course is ‘Why?’ I don’t think we understand completely why. We can speculate, but none of these type studies give us a definitive answer,” she said.
Possible reasons for the racial differences in HBsAg loss include differences in hepatitis B genotype, she said.
“Another possibility is that Asian patients may have been infected either at the time of birth or as a young kid, so they may have been infected for a much longer period of time than [Whites], who usually acquire infections as adults,” Dr. Lok said.
There may also be differences between patient populations in immune responses following cessation of antiviral therapy, she added.
The study was supported by the RETRACT-B group. Ms. Hirode and Dr. Lok reported no relevant disclosures.
SOURCE: Hirode G et al. AASLD 2020. Abstract 23.
FROM THE LIVER MEETING DIGITAL EXPERIENCE
Vanquishing hepatitis C: A remarkable success story
One of the most remarkable stories in medicine must be the relatively brief 25 years between the discovery of the hepatitis C virus (HCV) in 1989 to its eventual cure in 2014.
HCV afflicted over 5 million Americans and was the cause of death in approximately 10,000 patients annually, the leading indication for liver transplantation, and the leading risk factor for hepatocellular carcinoma, clearly signaling it as one of the era’s major public health villains. Within that span of time, it is the work beginning in the mid-1990s until today that perhaps best defines the race for the HCV “cure.”
In the early to mid-1990s, polymerase chain reaction techniques were just becoming commonplace for HCV diagnosis, whereas HCV genotypes were emerging as major factors determining response to interferon therapy. The sustained viral response (SVR) rates were mired at around 6%-12% for a 24- to 48-week course of three-times-weekly injection therapy. Severe side effects were common and there was a relatively high relapse rate, even in patients who responded to treatment.
By 1996, the addition of ribavirin to the interferon treatment was associated with a modest but significant improvement in SVR rates to above 20%. And by 2000, the use of pegylated interferon – allowing once-weekly injection therapy – along with ribavirin, improved SVR rates to above 50% for the first time. The therapy was still poorly tolerated but was associated with better compliance.
The real breakthrough in therapy came in the early 2000s with the discovery and availability of HCV protease inhibitors: telaprevir and boceprevir. These agents could induce a more rapid decline in viral replication than interferon but could not be administered alone owing to the rapid emergence of resistant HCV variants. Therefore, these agents were administered with interferon and ribavirin as a three-drug cocktail to take advantage of interferon to prevent emergence of resistant variants. Although SVR rates improved substantially to around 75%, adverse events also increased and limited its usefulness in patients with more advanced liver disease, precisely those who were most in need of better therapies.
Nonetheless, the incredible advances in understanding the replication machinery of HCV that led to the discovery of the protease inhibitors in turn led to further elucidation and unlocking of three additional classes of HCV protein targets and inhibitors: NS5A complex inhibitors (e.g., ledipasvir), the NS5B nonnucleoside inhibitors (e.g., dasabuvir), and NS5B nucleoside inhibitors (e.g., sofosbuvir). It quickly became apparent that the use of combinations of these direct-acting antivirals (DAAs) could limit emergence of resistant variants while also providing rapid and profound viral suppression. Because HCV required viral replication to persist in the hepatocyte, it became possible to induce HCV eradication, and thus cure, with combinations of DAAs.
In addition, investigators soon learned that the duration of therapy no longer needed to be the generally accepted 24-48 weeks for SVR, but instead could be reduced eventually to 8-12 weeks. This shortened treatment duration allowed for more rapid testing of new agents and combinations, and the field took a rapid step forward between 2011 and 2017. HCV cure rates rose to 90%-95%.
The competition for Food and Drug Administration approval of new agents among several pharmaceutical companies also meant that the time-honored process of issuing treatment guidelines every 3-5 years by societies would not be adequate. Therefore, in 2013, the American Association for the Study of Liver Diseases and the Infectious Diseases Society of America joined forces to establish more nimble and responsive online HCV guidance. This important resource debuted in January 2014 just as the FDA approved the first DAA therapies.
The high cost initially associated with many of these new therapies (up to $1,000 per pill) significantly limited uptake owing to insurance and health plan cost factors. Early on, the cost was also analyzed by price per cure, seemingly to justify the high cost by the high cure rate. However, advocacy and negotiations ultimately led to marked reductions in the cost of a course of therapy (with some therapies at $225 per pill), thus making these treatments now widely available.
By 2020, the HCV field has shifted from therapeutic development to improving the care cascade by enhanced identification and testing of unsuspected but HCV infected individuals. This is our current challenge.
Moving toward noninvasive tests
While curative therapy has revolutionized HCV management, innovation in diagnostics eliminated a significant barrier in access to therapy: the liver biopsy.
Staging, or accurately identifying advanced fibrosis in persons infected with HCV, is essential for long-term follow-up. The presence of advanced disease affects drug choices, especially before the approval of all-oral therapy. Historically, a liver biopsy was obligatory before treatment. Invasive with a significant risk for complications, this requirement effectively prevented treatment in those who were unwilling to undergo the procedure and deterred those at risk from even being tested.
Over the past 25 years, numerous methods to noninvasively assess for liver fibrosis have been used. Serum biomarkers can be either indirect (based on routine tests) or direct (reflecting components of the extracellular matrix). Although highly available, they are only moderately useful for identifying advanced fibrosis and thus cannot replace liver biopsy in the care cascade. The technique of elastography dates back to the 1980s, though the role of vibration-controlled transient liver elastography in the assessment of hepatic fibrosis in patients with HCV was not recognized until around 2005 and it was not commonly used for nearly another decade.
Yet, a paradigm shift in the care cascade occurred with the release of the AASLD/IDSA guidance document in 2014. For the first time in the United States, noninvasive tests were recommended as first-line testing for the assessment of advanced fibrosis. Prior guidelines specifically stated that although noninvasive tests might be useful, they “should not replace the liver biopsy in routine clinical practice.” Current guidelines recommend combining elastography with serum biomarkers and considering biopsy only in patients with discordant results if the biopsy would affect clinical decision-making.
The last frontier
Curative therapy has also allowed the unthinkable: willingly exposing patients to the virus through donor-positive/recipient-negative solid organ transplant. Traditionally, an HCV-infected donor would be considered only for an HCV-positive recipient; however, with effective DAA therapy, the number of HCV actively infected patients in need of transplant has dwindled.
Unfortunately as a consequence of the opioid epidemic, the HCV-exposed donor population has blossomed. Given that HCV therapy is near universally curative, using organs from HCV-viremic donors can greatly expand the organ transplantation pool. Small studies[1-5] have demonstrated the safety and efficacy of this approach, both in HCV-positive liver donors as well as in other solid organs.
A disease pegged for elimination
In the past 25 years, HCV has evolved from non-A, non-B hepatitis into a disease pegged for elimination. This is a direct reflection of improved therapeutics with highly effective DAAs. Yet, without improved diagnostics, we would be unable to navigate patients through the clinical care cascade. These incredible strides in diagnostics and therapeutics allow us to push the cutting edge through iatrogenic infection of organ recipients, while recognizing that the largest hurdle to elimination remains in finding those who are chronically infected. Ultimately, the crux of elimination remains unchanged over the past 25 years and resides in screening and diagnosis with effective linkage to care.
Donald M. Jensen, MD, is a professor of medicine at Rush University Medical Center, Chicago. He was previously the director of the Center for Liver Disease at the University of Chicago until 2015. His research interest has been in newer HCV therapies. He recently received the Distinguished Service Award from the AASLD for his many contributions to the field.
Nancy S. Reau, MD, is chief of the hepatology section at Rush University Medical Center and a regular contributor to Medscape. She serves as editor of Clinical Liver Disease, a multimedia review journal, and recently as a member of HCVGuidelines.org, a web-based resource from the AASLD and the IDSA, as well as educational chair for the AASLD hepatitis C special interest group. She continues to have an active role in the hepatology interest group of the World Gastroenterology Organisation and the American Liver Foundation at the regional and national levels.
References
Woolley AE et al. Heart and lung transplants from HCV-infected donors to uninfected recipients. N Engl J Med. 2019;380:1606-17.
Franco A et al. Renal transplantation from seropositive hepatitis C virus donors to seronegative recipients in Spain: A prospective study. Transpl Int. 2019;32:710-6.
Goldberg DS et al. Transplanting HCV-infected kidneys into uninfected recipients. N Engl J Med. 2017;377:1105.
Kwong AJ et al. Liver transplantation for hepatitis C virus (HCV) nonviremic recipients with HCV viremic donors. Am J Transplant. 2019;19:1380-7.
Bethea E et al. Immediate administration of antiviral therapy after transplantation of hepatitis C–infected livers into uninfected recipients: Implications for therapeutic planning. Am J Transplant. 2020;20:1619-28.
This article first appeared on Medscape.com.
One of the most remarkable stories in medicine must be the relatively brief 25 years between the discovery of the hepatitis C virus (HCV) in 1989 to its eventual cure in 2014.
HCV afflicted over 5 million Americans and was the cause of death in approximately 10,000 patients annually, the leading indication for liver transplantation, and the leading risk factor for hepatocellular carcinoma, clearly signaling it as one of the era’s major public health villains. Within that span of time, it is the work beginning in the mid-1990s until today that perhaps best defines the race for the HCV “cure.”
In the early to mid-1990s, polymerase chain reaction techniques were just becoming commonplace for HCV diagnosis, whereas HCV genotypes were emerging as major factors determining response to interferon therapy. The sustained viral response (SVR) rates were mired at around 6%-12% for a 24- to 48-week course of three-times-weekly injection therapy. Severe side effects were common and there was a relatively high relapse rate, even in patients who responded to treatment.
By 1996, the addition of ribavirin to the interferon treatment was associated with a modest but significant improvement in SVR rates to above 20%. And by 2000, the use of pegylated interferon – allowing once-weekly injection therapy – along with ribavirin, improved SVR rates to above 50% for the first time. The therapy was still poorly tolerated but was associated with better compliance.
The real breakthrough in therapy came in the early 2000s with the discovery and availability of HCV protease inhibitors: telaprevir and boceprevir. These agents could induce a more rapid decline in viral replication than interferon but could not be administered alone owing to the rapid emergence of resistant HCV variants. Therefore, these agents were administered with interferon and ribavirin as a three-drug cocktail to take advantage of interferon to prevent emergence of resistant variants. Although SVR rates improved substantially to around 75%, adverse events also increased and limited its usefulness in patients with more advanced liver disease, precisely those who were most in need of better therapies.
Nonetheless, the incredible advances in understanding the replication machinery of HCV that led to the discovery of the protease inhibitors in turn led to further elucidation and unlocking of three additional classes of HCV protein targets and inhibitors: NS5A complex inhibitors (e.g., ledipasvir), the NS5B nonnucleoside inhibitors (e.g., dasabuvir), and NS5B nucleoside inhibitors (e.g., sofosbuvir). It quickly became apparent that the use of combinations of these direct-acting antivirals (DAAs) could limit emergence of resistant variants while also providing rapid and profound viral suppression. Because HCV required viral replication to persist in the hepatocyte, it became possible to induce HCV eradication, and thus cure, with combinations of DAAs.
In addition, investigators soon learned that the duration of therapy no longer needed to be the generally accepted 24-48 weeks for SVR, but instead could be reduced eventually to 8-12 weeks. This shortened treatment duration allowed for more rapid testing of new agents and combinations, and the field took a rapid step forward between 2011 and 2017. HCV cure rates rose to 90%-95%.
The competition for Food and Drug Administration approval of new agents among several pharmaceutical companies also meant that the time-honored process of issuing treatment guidelines every 3-5 years by societies would not be adequate. Therefore, in 2013, the American Association for the Study of Liver Diseases and the Infectious Diseases Society of America joined forces to establish more nimble and responsive online HCV guidance. This important resource debuted in January 2014 just as the FDA approved the first DAA therapies.
The high cost initially associated with many of these new therapies (up to $1,000 per pill) significantly limited uptake owing to insurance and health plan cost factors. Early on, the cost was also analyzed by price per cure, seemingly to justify the high cost by the high cure rate. However, advocacy and negotiations ultimately led to marked reductions in the cost of a course of therapy (with some therapies at $225 per pill), thus making these treatments now widely available.
By 2020, the HCV field has shifted from therapeutic development to improving the care cascade by enhanced identification and testing of unsuspected but HCV infected individuals. This is our current challenge.
Moving toward noninvasive tests
While curative therapy has revolutionized HCV management, innovation in diagnostics eliminated a significant barrier in access to therapy: the liver biopsy.
Staging, or accurately identifying advanced fibrosis in persons infected with HCV, is essential for long-term follow-up. The presence of advanced disease affects drug choices, especially before the approval of all-oral therapy. Historically, a liver biopsy was obligatory before treatment. Invasive with a significant risk for complications, this requirement effectively prevented treatment in those who were unwilling to undergo the procedure and deterred those at risk from even being tested.
Over the past 25 years, numerous methods to noninvasively assess for liver fibrosis have been used. Serum biomarkers can be either indirect (based on routine tests) or direct (reflecting components of the extracellular matrix). Although highly available, they are only moderately useful for identifying advanced fibrosis and thus cannot replace liver biopsy in the care cascade. The technique of elastography dates back to the 1980s, though the role of vibration-controlled transient liver elastography in the assessment of hepatic fibrosis in patients with HCV was not recognized until around 2005 and it was not commonly used for nearly another decade.
Yet, a paradigm shift in the care cascade occurred with the release of the AASLD/IDSA guidance document in 2014. For the first time in the United States, noninvasive tests were recommended as first-line testing for the assessment of advanced fibrosis. Prior guidelines specifically stated that although noninvasive tests might be useful, they “should not replace the liver biopsy in routine clinical practice.” Current guidelines recommend combining elastography with serum biomarkers and considering biopsy only in patients with discordant results if the biopsy would affect clinical decision-making.
The last frontier
Curative therapy has also allowed the unthinkable: willingly exposing patients to the virus through donor-positive/recipient-negative solid organ transplant. Traditionally, an HCV-infected donor would be considered only for an HCV-positive recipient; however, with effective DAA therapy, the number of HCV actively infected patients in need of transplant has dwindled.
Unfortunately as a consequence of the opioid epidemic, the HCV-exposed donor population has blossomed. Given that HCV therapy is near universally curative, using organs from HCV-viremic donors can greatly expand the organ transplantation pool. Small studies[1-5] have demonstrated the safety and efficacy of this approach, both in HCV-positive liver donors as well as in other solid organs.
A disease pegged for elimination
In the past 25 years, HCV has evolved from non-A, non-B hepatitis into a disease pegged for elimination. This is a direct reflection of improved therapeutics with highly effective DAAs. Yet, without improved diagnostics, we would be unable to navigate patients through the clinical care cascade. These incredible strides in diagnostics and therapeutics allow us to push the cutting edge through iatrogenic infection of organ recipients, while recognizing that the largest hurdle to elimination remains in finding those who are chronically infected. Ultimately, the crux of elimination remains unchanged over the past 25 years and resides in screening and diagnosis with effective linkage to care.
Donald M. Jensen, MD, is a professor of medicine at Rush University Medical Center, Chicago. He was previously the director of the Center for Liver Disease at the University of Chicago until 2015. His research interest has been in newer HCV therapies. He recently received the Distinguished Service Award from the AASLD for his many contributions to the field.
Nancy S. Reau, MD, is chief of the hepatology section at Rush University Medical Center and a regular contributor to Medscape. She serves as editor of Clinical Liver Disease, a multimedia review journal, and recently as a member of HCVGuidelines.org, a web-based resource from the AASLD and the IDSA, as well as educational chair for the AASLD hepatitis C special interest group. She continues to have an active role in the hepatology interest group of the World Gastroenterology Organisation and the American Liver Foundation at the regional and national levels.
References
Woolley AE et al. Heart and lung transplants from HCV-infected donors to uninfected recipients. N Engl J Med. 2019;380:1606-17.
Franco A et al. Renal transplantation from seropositive hepatitis C virus donors to seronegative recipients in Spain: A prospective study. Transpl Int. 2019;32:710-6.
Goldberg DS et al. Transplanting HCV-infected kidneys into uninfected recipients. N Engl J Med. 2017;377:1105.
Kwong AJ et al. Liver transplantation for hepatitis C virus (HCV) nonviremic recipients with HCV viremic donors. Am J Transplant. 2019;19:1380-7.
Bethea E et al. Immediate administration of antiviral therapy after transplantation of hepatitis C–infected livers into uninfected recipients: Implications for therapeutic planning. Am J Transplant. 2020;20:1619-28.
This article first appeared on Medscape.com.
One of the most remarkable stories in medicine must be the relatively brief 25 years between the discovery of the hepatitis C virus (HCV) in 1989 to its eventual cure in 2014.
HCV afflicted over 5 million Americans and was the cause of death in approximately 10,000 patients annually, the leading indication for liver transplantation, and the leading risk factor for hepatocellular carcinoma, clearly signaling it as one of the era’s major public health villains. Within that span of time, it is the work beginning in the mid-1990s until today that perhaps best defines the race for the HCV “cure.”
In the early to mid-1990s, polymerase chain reaction techniques were just becoming commonplace for HCV diagnosis, whereas HCV genotypes were emerging as major factors determining response to interferon therapy. The sustained viral response (SVR) rates were mired at around 6%-12% for a 24- to 48-week course of three-times-weekly injection therapy. Severe side effects were common and there was a relatively high relapse rate, even in patients who responded to treatment.
By 1996, the addition of ribavirin to the interferon treatment was associated with a modest but significant improvement in SVR rates to above 20%. And by 2000, the use of pegylated interferon – allowing once-weekly injection therapy – along with ribavirin, improved SVR rates to above 50% for the first time. The therapy was still poorly tolerated but was associated with better compliance.
The real breakthrough in therapy came in the early 2000s with the discovery and availability of HCV protease inhibitors: telaprevir and boceprevir. These agents could induce a more rapid decline in viral replication than interferon but could not be administered alone owing to the rapid emergence of resistant HCV variants. Therefore, these agents were administered with interferon and ribavirin as a three-drug cocktail to take advantage of interferon to prevent emergence of resistant variants. Although SVR rates improved substantially to around 75%, adverse events also increased and limited its usefulness in patients with more advanced liver disease, precisely those who were most in need of better therapies.
Nonetheless, the incredible advances in understanding the replication machinery of HCV that led to the discovery of the protease inhibitors in turn led to further elucidation and unlocking of three additional classes of HCV protein targets and inhibitors: NS5A complex inhibitors (e.g., ledipasvir), the NS5B nonnucleoside inhibitors (e.g., dasabuvir), and NS5B nucleoside inhibitors (e.g., sofosbuvir). It quickly became apparent that the use of combinations of these direct-acting antivirals (DAAs) could limit emergence of resistant variants while also providing rapid and profound viral suppression. Because HCV required viral replication to persist in the hepatocyte, it became possible to induce HCV eradication, and thus cure, with combinations of DAAs.
In addition, investigators soon learned that the duration of therapy no longer needed to be the generally accepted 24-48 weeks for SVR, but instead could be reduced eventually to 8-12 weeks. This shortened treatment duration allowed for more rapid testing of new agents and combinations, and the field took a rapid step forward between 2011 and 2017. HCV cure rates rose to 90%-95%.
The competition for Food and Drug Administration approval of new agents among several pharmaceutical companies also meant that the time-honored process of issuing treatment guidelines every 3-5 years by societies would not be adequate. Therefore, in 2013, the American Association for the Study of Liver Diseases and the Infectious Diseases Society of America joined forces to establish more nimble and responsive online HCV guidance. This important resource debuted in January 2014 just as the FDA approved the first DAA therapies.
The high cost initially associated with many of these new therapies (up to $1,000 per pill) significantly limited uptake owing to insurance and health plan cost factors. Early on, the cost was also analyzed by price per cure, seemingly to justify the high cost by the high cure rate. However, advocacy and negotiations ultimately led to marked reductions in the cost of a course of therapy (with some therapies at $225 per pill), thus making these treatments now widely available.
By 2020, the HCV field has shifted from therapeutic development to improving the care cascade by enhanced identification and testing of unsuspected but HCV infected individuals. This is our current challenge.
Moving toward noninvasive tests
While curative therapy has revolutionized HCV management, innovation in diagnostics eliminated a significant barrier in access to therapy: the liver biopsy.
Staging, or accurately identifying advanced fibrosis in persons infected with HCV, is essential for long-term follow-up. The presence of advanced disease affects drug choices, especially before the approval of all-oral therapy. Historically, a liver biopsy was obligatory before treatment. Invasive with a significant risk for complications, this requirement effectively prevented treatment in those who were unwilling to undergo the procedure and deterred those at risk from even being tested.
Over the past 25 years, numerous methods to noninvasively assess for liver fibrosis have been used. Serum biomarkers can be either indirect (based on routine tests) or direct (reflecting components of the extracellular matrix). Although highly available, they are only moderately useful for identifying advanced fibrosis and thus cannot replace liver biopsy in the care cascade. The technique of elastography dates back to the 1980s, though the role of vibration-controlled transient liver elastography in the assessment of hepatic fibrosis in patients with HCV was not recognized until around 2005 and it was not commonly used for nearly another decade.
Yet, a paradigm shift in the care cascade occurred with the release of the AASLD/IDSA guidance document in 2014. For the first time in the United States, noninvasive tests were recommended as first-line testing for the assessment of advanced fibrosis. Prior guidelines specifically stated that although noninvasive tests might be useful, they “should not replace the liver biopsy in routine clinical practice.” Current guidelines recommend combining elastography with serum biomarkers and considering biopsy only in patients with discordant results if the biopsy would affect clinical decision-making.
The last frontier
Curative therapy has also allowed the unthinkable: willingly exposing patients to the virus through donor-positive/recipient-negative solid organ transplant. Traditionally, an HCV-infected donor would be considered only for an HCV-positive recipient; however, with effective DAA therapy, the number of HCV actively infected patients in need of transplant has dwindled.
Unfortunately as a consequence of the opioid epidemic, the HCV-exposed donor population has blossomed. Given that HCV therapy is near universally curative, using organs from HCV-viremic donors can greatly expand the organ transplantation pool. Small studies[1-5] have demonstrated the safety and efficacy of this approach, both in HCV-positive liver donors as well as in other solid organs.
A disease pegged for elimination
In the past 25 years, HCV has evolved from non-A, non-B hepatitis into a disease pegged for elimination. This is a direct reflection of improved therapeutics with highly effective DAAs. Yet, without improved diagnostics, we would be unable to navigate patients through the clinical care cascade. These incredible strides in diagnostics and therapeutics allow us to push the cutting edge through iatrogenic infection of organ recipients, while recognizing that the largest hurdle to elimination remains in finding those who are chronically infected. Ultimately, the crux of elimination remains unchanged over the past 25 years and resides in screening and diagnosis with effective linkage to care.
Donald M. Jensen, MD, is a professor of medicine at Rush University Medical Center, Chicago. He was previously the director of the Center for Liver Disease at the University of Chicago until 2015. His research interest has been in newer HCV therapies. He recently received the Distinguished Service Award from the AASLD for his many contributions to the field.
Nancy S. Reau, MD, is chief of the hepatology section at Rush University Medical Center and a regular contributor to Medscape. She serves as editor of Clinical Liver Disease, a multimedia review journal, and recently as a member of HCVGuidelines.org, a web-based resource from the AASLD and the IDSA, as well as educational chair for the AASLD hepatitis C special interest group. She continues to have an active role in the hepatology interest group of the World Gastroenterology Organisation and the American Liver Foundation at the regional and national levels.
References
Woolley AE et al. Heart and lung transplants from HCV-infected donors to uninfected recipients. N Engl J Med. 2019;380:1606-17.
Franco A et al. Renal transplantation from seropositive hepatitis C virus donors to seronegative recipients in Spain: A prospective study. Transpl Int. 2019;32:710-6.
Goldberg DS et al. Transplanting HCV-infected kidneys into uninfected recipients. N Engl J Med. 2017;377:1105.
Kwong AJ et al. Liver transplantation for hepatitis C virus (HCV) nonviremic recipients with HCV viremic donors. Am J Transplant. 2019;19:1380-7.
Bethea E et al. Immediate administration of antiviral therapy after transplantation of hepatitis C–infected livers into uninfected recipients: Implications for therapeutic planning. Am J Transplant. 2020;20:1619-28.
This article first appeared on Medscape.com.
Harnessing the HIV care continuum model to improve HCV treatment success
Better linkage to care with providers who are familiar with both the HCV and HIV treatment cascade may not only improve access to HCV treatment, but it may also support patient retention, treatment adherence, and achievement of sustained virologic response (SVR) and viral suppression, said Stephanie LaMoy, CAN Community Health, North Point, Florida. She presented the results of a pilot study at the virtual Association of Nurses in AIDS Care 2020 Annual Meeting.
In an effort to identify strategies most important for improving care access among their patients with HCV, LaMoy and her colleagues assessed 12-month patient data collected from three of their clinics. These data were evaluated for HCV treatment access, engagement, and outcomes.
The pilot study included 126 patients who were reactive and another 24 HCV-positive patients who were referred from other sources. Active HCV infections requiring treatment were reported in 144 patients.
A total of 59 patients were linked to care but did not initiate treatment for their active infection. LaMoy said there were multiple causes, including homelessness, substance abuse, and inability to maintain contact.
In contrast, 85 patients with HCV infection started treatment, but 35 of these patients did not complete their regimen. Out of the 50 patients who reported completing treatment, 30 did not return to the clinic to confirm sustained viral suppression.
According to LaMoy, this raised a red flag, causing the investigators to consider a different approach to care.
HIV care continuum model and its role in HCV
To improve the rate at which patients with HCV infection complete treatment within their clinics, the researchers formed a panel to determine necessary interventions that could reduce barriers to care.
The HIV care continuum came into play. They chose this model based on knowledge that HCV and HIV share the same care continuum with similar goals in diagnosis, linkage to care, retention, and suppression.
Based on the consensus of the panel and consideration of the HIV care continuum model, they identified a number of interventions needed to mitigate HCV treatment barriers. These included the incorporation of peer navigators or linkage-to-care (LCC) coordinators, use of the mobile medical unit, greater implementation of onsite lab visits, and medication-assisted treatment.
The LCC coordinators proved to be particularly important, as these team members helped assist patients with social and financial support to address challenges with access to treatment. These coordinators can also help patients gain access to specialized providers, ultimately improving the chance of successful HCV management.
Additionally, LCC coordinators may help identify and reduce barriers associated with housing, transportation, and nutrition. Frequent patient contact by the LCC coordinators can encourage adherence and promote risk reduction education, such as providing referrals to needle exchange services.
“Linking individuals to care with providers who are familiar with the treatment cascade could help improve retention and should be a top priority for those involved in HCV screening and treatment,” said LaMoy. “An environment with knowledge, lack of judgment, and a tenacious need to heal the community that welcomes those with barriers to care is exactly what is needed for the patients in our program.”
National, community challenges fuel barriers to HCV treatment access
Substance use, trauma histories, and mental health problems can negatively affect care engagement and must be addressed before the benefits of HCV therapy can be realized.
Addressing these issues isn’t always easy, said Kathleen Bernock, FNP-BC, AACRN, AAHIVS, of the Bedford-Stuyvesant Family Health Center in New York City, in an email to Medscape Medical News. She pointed out that several states have harsh restrictions on who is able to access HCV treatment, and some states will not approve certain medications for people who actively use drugs.
“Even for states without these restrictions, many health systems are difficult to navigate and may not be welcoming to persons actively using,” said Bernock. Trauma-informed care can also be difficult to translate into clinics, she added.
“Decentralizing care to the communities most affected would greatly help mitigate these barriers,” suggested Bernock. Decentralization, she explained, might include co-locating services such as syringe exchanges, utilizing community health workers and patient navigators, and expanding capacity-to-treat to community-based providers.
“[And] with the expansion of telehealth services in the US,” said Bernock, “we now have even more avenues to reach people that we never had before.”
LaMoy and Bernock have disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
Better linkage to care with providers who are familiar with both the HCV and HIV treatment cascade may not only improve access to HCV treatment, but it may also support patient retention, treatment adherence, and achievement of sustained virologic response (SVR) and viral suppression, said Stephanie LaMoy, CAN Community Health, North Point, Florida. She presented the results of a pilot study at the virtual Association of Nurses in AIDS Care 2020 Annual Meeting.
In an effort to identify strategies most important for improving care access among their patients with HCV, LaMoy and her colleagues assessed 12-month patient data collected from three of their clinics. These data were evaluated for HCV treatment access, engagement, and outcomes.
The pilot study included 126 patients who were reactive and another 24 HCV-positive patients who were referred from other sources. Active HCV infections requiring treatment were reported in 144 patients.
A total of 59 patients were linked to care but did not initiate treatment for their active infection. LaMoy said there were multiple causes, including homelessness, substance abuse, and inability to maintain contact.
In contrast, 85 patients with HCV infection started treatment, but 35 of these patients did not complete their regimen. Out of the 50 patients who reported completing treatment, 30 did not return to the clinic to confirm sustained viral suppression.
According to LaMoy, this raised a red flag, causing the investigators to consider a different approach to care.
HIV care continuum model and its role in HCV
To improve the rate at which patients with HCV infection complete treatment within their clinics, the researchers formed a panel to determine necessary interventions that could reduce barriers to care.
The HIV care continuum came into play. They chose this model based on knowledge that HCV and HIV share the same care continuum with similar goals in diagnosis, linkage to care, retention, and suppression.
Based on the consensus of the panel and consideration of the HIV care continuum model, they identified a number of interventions needed to mitigate HCV treatment barriers. These included the incorporation of peer navigators or linkage-to-care (LCC) coordinators, use of the mobile medical unit, greater implementation of onsite lab visits, and medication-assisted treatment.
The LCC coordinators proved to be particularly important, as these team members helped assist patients with social and financial support to address challenges with access to treatment. These coordinators can also help patients gain access to specialized providers, ultimately improving the chance of successful HCV management.
Additionally, LCC coordinators may help identify and reduce barriers associated with housing, transportation, and nutrition. Frequent patient contact by the LCC coordinators can encourage adherence and promote risk reduction education, such as providing referrals to needle exchange services.
“Linking individuals to care with providers who are familiar with the treatment cascade could help improve retention and should be a top priority for those involved in HCV screening and treatment,” said LaMoy. “An environment with knowledge, lack of judgment, and a tenacious need to heal the community that welcomes those with barriers to care is exactly what is needed for the patients in our program.”
National, community challenges fuel barriers to HCV treatment access
Substance use, trauma histories, and mental health problems can negatively affect care engagement and must be addressed before the benefits of HCV therapy can be realized.
Addressing these issues isn’t always easy, said Kathleen Bernock, FNP-BC, AACRN, AAHIVS, of the Bedford-Stuyvesant Family Health Center in New York City, in an email to Medscape Medical News. She pointed out that several states have harsh restrictions on who is able to access HCV treatment, and some states will not approve certain medications for people who actively use drugs.
“Even for states without these restrictions, many health systems are difficult to navigate and may not be welcoming to persons actively using,” said Bernock. Trauma-informed care can also be difficult to translate into clinics, she added.
“Decentralizing care to the communities most affected would greatly help mitigate these barriers,” suggested Bernock. Decentralization, she explained, might include co-locating services such as syringe exchanges, utilizing community health workers and patient navigators, and expanding capacity-to-treat to community-based providers.
“[And] with the expansion of telehealth services in the US,” said Bernock, “we now have even more avenues to reach people that we never had before.”
LaMoy and Bernock have disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
Better linkage to care with providers who are familiar with both the HCV and HIV treatment cascade may not only improve access to HCV treatment, but it may also support patient retention, treatment adherence, and achievement of sustained virologic response (SVR) and viral suppression, said Stephanie LaMoy, CAN Community Health, North Point, Florida. She presented the results of a pilot study at the virtual Association of Nurses in AIDS Care 2020 Annual Meeting.
In an effort to identify strategies most important for improving care access among their patients with HCV, LaMoy and her colleagues assessed 12-month patient data collected from three of their clinics. These data were evaluated for HCV treatment access, engagement, and outcomes.
The pilot study included 126 patients who were reactive and another 24 HCV-positive patients who were referred from other sources. Active HCV infections requiring treatment were reported in 144 patients.
A total of 59 patients were linked to care but did not initiate treatment for their active infection. LaMoy said there were multiple causes, including homelessness, substance abuse, and inability to maintain contact.
In contrast, 85 patients with HCV infection started treatment, but 35 of these patients did not complete their regimen. Out of the 50 patients who reported completing treatment, 30 did not return to the clinic to confirm sustained viral suppression.
According to LaMoy, this raised a red flag, causing the investigators to consider a different approach to care.
HIV care continuum model and its role in HCV
To improve the rate at which patients with HCV infection complete treatment within their clinics, the researchers formed a panel to determine necessary interventions that could reduce barriers to care.
The HIV care continuum came into play. They chose this model based on knowledge that HCV and HIV share the same care continuum with similar goals in diagnosis, linkage to care, retention, and suppression.
Based on the consensus of the panel and consideration of the HIV care continuum model, they identified a number of interventions needed to mitigate HCV treatment barriers. These included the incorporation of peer navigators or linkage-to-care (LCC) coordinators, use of the mobile medical unit, greater implementation of onsite lab visits, and medication-assisted treatment.
The LCC coordinators proved to be particularly important, as these team members helped assist patients with social and financial support to address challenges with access to treatment. These coordinators can also help patients gain access to specialized providers, ultimately improving the chance of successful HCV management.
Additionally, LCC coordinators may help identify and reduce barriers associated with housing, transportation, and nutrition. Frequent patient contact by the LCC coordinators can encourage adherence and promote risk reduction education, such as providing referrals to needle exchange services.
“Linking individuals to care with providers who are familiar with the treatment cascade could help improve retention and should be a top priority for those involved in HCV screening and treatment,” said LaMoy. “An environment with knowledge, lack of judgment, and a tenacious need to heal the community that welcomes those with barriers to care is exactly what is needed for the patients in our program.”
National, community challenges fuel barriers to HCV treatment access
Substance use, trauma histories, and mental health problems can negatively affect care engagement and must be addressed before the benefits of HCV therapy can be realized.
Addressing these issues isn’t always easy, said Kathleen Bernock, FNP-BC, AACRN, AAHIVS, of the Bedford-Stuyvesant Family Health Center in New York City, in an email to Medscape Medical News. She pointed out that several states have harsh restrictions on who is able to access HCV treatment, and some states will not approve certain medications for people who actively use drugs.
“Even for states without these restrictions, many health systems are difficult to navigate and may not be welcoming to persons actively using,” said Bernock. Trauma-informed care can also be difficult to translate into clinics, she added.
“Decentralizing care to the communities most affected would greatly help mitigate these barriers,” suggested Bernock. Decentralization, she explained, might include co-locating services such as syringe exchanges, utilizing community health workers and patient navigators, and expanding capacity-to-treat to community-based providers.
“[And] with the expansion of telehealth services in the US,” said Bernock, “we now have even more avenues to reach people that we never had before.”
LaMoy and Bernock have disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
Abnormal anal paps in people with HIV can go more than a year without follow-up
That delay “revealed missed opportunities for a better experience on the patient, clinic, and provider level,” Jessica Wells, PhD, research assistant professor at the Nell Hodgson Woodruff School of Nursing at Emory University, Atlanta, said in an interview. After all, “a lot can happen in that 1 year,” including early development of human papillomavirus (HPV)–associated anal cancer.
Although it’s too soon to say how significant that delay is with respect to the natural history of anal cancer, Dr. Wells said the data are a potential signal of disparities.
“The findings from my study may foreshadow potential disparities if we don’t have the necessary resources in place to promote follow-up care after an abnormal Pap test, similar to the disparities that we see in cervical cancer,” she said during the virtual Association of Nurses in AIDS Care 2020 Annual Meeting.
Single-center study
In the United States, people living with HIV are 19 times more likely to develop anal cancer than the general population, according to a 2018 article in the Journal of Clinical Oncology. Another single-center study from Yale University found that, in minority communities, anal cancer rates were 75% higher than in White communities. Anal cancer rates were 72% higher in communities with greater poverty. As a result, many clinics are beginning to administer Pap tests to determine early signs of HPV infection and associated changes.
In Dr. Wells’ study, which was conducted from 2012 to 2015, 150 adults with HIV who were aged 21 and older were recruited from Grady Ponce De Leon Center in Atlanta. According to a 2018 study from that center, a large minority of participants had late-stage HIV and suppressed immune systems.
All participants had been referred for HRA after a recent abnormal anal Pap test. Participants filled out questionnaires on sociodemographics, internalized HIV-related stigma, depression, risk behaviors, social support, and knowledge about HPV and anal cancer.
Participants were disproportionately older (mean age, 50.9 years); cisgender (86.7%), Black (78%); and gay, lesbian, or bisexual (84.3%). Slightly more than 1 in 10 participants (11.3%) were transgender women.
Although for 6% of participants, Pap test results indicated high-grade squamous intraepithelial lesions (HSIL), an additional 8% had atypical Pap findings that couldn’t exclude HSIL – the kinds of results that are one step away from a cancer diagnosis. More than 80% of participants had low-grade or inconclusive results. Nearly half (44%) of participants’ Pap tests revealed low-grade squamous cell intraepithelial cell lesions (LSIL); 42% indicated atypical squamous cells of undetermined significance.
When Dr. Wells looked at how long participants had waited to undergo HRA, she found something that surprised her: although some participants underwent follow-up assessment in 17 days, for many, it took much longer. The longest wait was 2,350 days – more than 6 years.
“There were quite a few patients who had follow-up beyond 1,000-plus days,” Dr. Wells said in an interview. “I didn›t think the delays were that long — at most, I would say that patients will get scheduled and come back within a few weeks or months.”
What’s more, she discovered through the HPV knowledge questionnaire that many participants did not understand why they were having a follow-up appointment. Anecdotally, some confused HPV with HIV.
“There’s education to be done to inform this target population that those living with HIV are more prone or at increased risk of this virus causing cancer later,” she said. “There are a lot of campaigns around women living with HIV, that they need to do cervical cancer screening. I think we need to really expand this campaign to include that HPV can also cause anal cancer.”
Dr. Wells had planned to primarily investigate the impact of psychosocial factors on wait time to follow-up, but none of those factors were associated with longer wait times.
Systems-level factors
That led Ann Gakumo, PhD, chair of nursing at the College of Nursing and Health Sciences of the University of Massachusetts, Boston, to ask what other factors could account for the delay.
There were several, Dr. Wells said. Precarious housing, for example, could have influenced this lag in follow-up. About one in four participants were in transient housing, and one participant reported having been incarcerated. She gathered street addresses and plans to analyze that data to see whether the cases occurred in clusters in specific neighborhoods, as the Yale data indicated.
In addition, the anoscopy clinic was only available to receive patients one day a week and was staffed with only one clinician who was trained to perform HRA. Wait times could stretch for hours. Sometimes, participants had to leave the clinic to attend to other business, and their appointments needed to be rescheduled, Wells said.
In addition to the sometimes poor understanding of the importance of the follow-up test, Dr. Wells said, “we start to see a layering of these barriers. That’s where we start seeing breakdowns. So I’m hoping in a larger study I can address some of these barriers on a multilevel approach.”
This resonated with Dr. Gakumo.
“Oftentimes, we put so much of the responsibility for this on the part of the client and not enough on the part of the provider or on the systems level,” she said.
Guiding guidelines
Guidelines on follow-up for abnormal anal Pap test results are scarce, mostly because, unlike cervical cancer, the natural history of HPV-related anal cancers hasn’t been established. The HIV Medical Association does recommend anal Pap tests, but only in cases in which “access to appropriate referral for follow-up, including high-resolution anoscopy, is available.”
In an interview, Cecile Lahiri, MD, assistant professor of infectious disease at Emory University, said that, at Ponce De Leon Center, they recommend an anal Pap for women with HIV who have a history of cervical dysplasia.
There is a reliable association between high-grade abnormal Pap tests and cervical cancer, although low-grade changes can resolve on their own. In the case of anal cancer, especially in patients with HIV, low-grade cell changes are predictive; moreover, for such patients, anal cancer is more likely to recur and is harder to treat, Dr. Lahiri said.
“The cervical environment and the anal environment are very different,” said Dr. Lahiri, who works at the Grady Ponce De Leon Center but was not involved in Dr. Wells’ study. Dr. Lahiri is also a coinvestigator of the multisite, randomized, controlled Anal Cancer HSIL Outcomes Research (ANCHOR) study, which seeks to establish whether early treatment of high-grade anal Pap changes is better than a watch-and-wait approach.
Dr. Lahiri said that when the results of that trial become available, they are more likely to know how important early anoscopy and treatment are. The findings should inform guidelines and insurance coverage of anal Pap tests and anoscopy.
In the meantime, she said, she suspected that, with the ANCHOR trial in 2015, many sites’ capacity for anoscopy may have increased, and the wait times may have gone down.
“One of the most important pieces of the study is actually the time period in which it was conducted,” said Dr. Lahiri, who in 2015 became the clinic’s second physician trained in anoscopy. Currently, more than 200 people at the Ponce De Leon Center are enrolled in the ANCHOR trial. In addition, the general capacity for performing anoscopies has gone up nationwide as a result of the trial, which required that more providers learn how to properly perform an HRA. Many clinicians are not routinely trained in performing HRA, including gastroenterologists and surgeons, Dr. Lahiri said.
“It would be interesting to look at the differences, with the start of ANCHOR being the time point for before and after,” she said.
This article first appeared on Medscape.com.
That delay “revealed missed opportunities for a better experience on the patient, clinic, and provider level,” Jessica Wells, PhD, research assistant professor at the Nell Hodgson Woodruff School of Nursing at Emory University, Atlanta, said in an interview. After all, “a lot can happen in that 1 year,” including early development of human papillomavirus (HPV)–associated anal cancer.
Although it’s too soon to say how significant that delay is with respect to the natural history of anal cancer, Dr. Wells said the data are a potential signal of disparities.
“The findings from my study may foreshadow potential disparities if we don’t have the necessary resources in place to promote follow-up care after an abnormal Pap test, similar to the disparities that we see in cervical cancer,” she said during the virtual Association of Nurses in AIDS Care 2020 Annual Meeting.
Single-center study
In the United States, people living with HIV are 19 times more likely to develop anal cancer than the general population, according to a 2018 article in the Journal of Clinical Oncology. Another single-center study from Yale University found that, in minority communities, anal cancer rates were 75% higher than in White communities. Anal cancer rates were 72% higher in communities with greater poverty. As a result, many clinics are beginning to administer Pap tests to determine early signs of HPV infection and associated changes.
In Dr. Wells’ study, which was conducted from 2012 to 2015, 150 adults with HIV who were aged 21 and older were recruited from Grady Ponce De Leon Center in Atlanta. According to a 2018 study from that center, a large minority of participants had late-stage HIV and suppressed immune systems.
All participants had been referred for HRA after a recent abnormal anal Pap test. Participants filled out questionnaires on sociodemographics, internalized HIV-related stigma, depression, risk behaviors, social support, and knowledge about HPV and anal cancer.
Participants were disproportionately older (mean age, 50.9 years); cisgender (86.7%), Black (78%); and gay, lesbian, or bisexual (84.3%). Slightly more than 1 in 10 participants (11.3%) were transgender women.
Although for 6% of participants, Pap test results indicated high-grade squamous intraepithelial lesions (HSIL), an additional 8% had atypical Pap findings that couldn’t exclude HSIL – the kinds of results that are one step away from a cancer diagnosis. More than 80% of participants had low-grade or inconclusive results. Nearly half (44%) of participants’ Pap tests revealed low-grade squamous cell intraepithelial cell lesions (LSIL); 42% indicated atypical squamous cells of undetermined significance.
When Dr. Wells looked at how long participants had waited to undergo HRA, she found something that surprised her: although some participants underwent follow-up assessment in 17 days, for many, it took much longer. The longest wait was 2,350 days – more than 6 years.
“There were quite a few patients who had follow-up beyond 1,000-plus days,” Dr. Wells said in an interview. “I didn›t think the delays were that long — at most, I would say that patients will get scheduled and come back within a few weeks or months.”
What’s more, she discovered through the HPV knowledge questionnaire that many participants did not understand why they were having a follow-up appointment. Anecdotally, some confused HPV with HIV.
“There’s education to be done to inform this target population that those living with HIV are more prone or at increased risk of this virus causing cancer later,” she said. “There are a lot of campaigns around women living with HIV, that they need to do cervical cancer screening. I think we need to really expand this campaign to include that HPV can also cause anal cancer.”
Dr. Wells had planned to primarily investigate the impact of psychosocial factors on wait time to follow-up, but none of those factors were associated with longer wait times.
Systems-level factors
That led Ann Gakumo, PhD, chair of nursing at the College of Nursing and Health Sciences of the University of Massachusetts, Boston, to ask what other factors could account for the delay.
There were several, Dr. Wells said. Precarious housing, for example, could have influenced this lag in follow-up. About one in four participants were in transient housing, and one participant reported having been incarcerated. She gathered street addresses and plans to analyze that data to see whether the cases occurred in clusters in specific neighborhoods, as the Yale data indicated.
In addition, the anoscopy clinic was only available to receive patients one day a week and was staffed with only one clinician who was trained to perform HRA. Wait times could stretch for hours. Sometimes, participants had to leave the clinic to attend to other business, and their appointments needed to be rescheduled, Wells said.
In addition to the sometimes poor understanding of the importance of the follow-up test, Dr. Wells said, “we start to see a layering of these barriers. That’s where we start seeing breakdowns. So I’m hoping in a larger study I can address some of these barriers on a multilevel approach.”
This resonated with Dr. Gakumo.
“Oftentimes, we put so much of the responsibility for this on the part of the client and not enough on the part of the provider or on the systems level,” she said.
Guiding guidelines
Guidelines on follow-up for abnormal anal Pap test results are scarce, mostly because, unlike cervical cancer, the natural history of HPV-related anal cancers hasn’t been established. The HIV Medical Association does recommend anal Pap tests, but only in cases in which “access to appropriate referral for follow-up, including high-resolution anoscopy, is available.”
In an interview, Cecile Lahiri, MD, assistant professor of infectious disease at Emory University, said that, at Ponce De Leon Center, they recommend an anal Pap for women with HIV who have a history of cervical dysplasia.
There is a reliable association between high-grade abnormal Pap tests and cervical cancer, although low-grade changes can resolve on their own. In the case of anal cancer, especially in patients with HIV, low-grade cell changes are predictive; moreover, for such patients, anal cancer is more likely to recur and is harder to treat, Dr. Lahiri said.
“The cervical environment and the anal environment are very different,” said Dr. Lahiri, who works at the Grady Ponce De Leon Center but was not involved in Dr. Wells’ study. Dr. Lahiri is also a coinvestigator of the multisite, randomized, controlled Anal Cancer HSIL Outcomes Research (ANCHOR) study, which seeks to establish whether early treatment of high-grade anal Pap changes is better than a watch-and-wait approach.
Dr. Lahiri said that when the results of that trial become available, they are more likely to know how important early anoscopy and treatment are. The findings should inform guidelines and insurance coverage of anal Pap tests and anoscopy.
In the meantime, she said, she suspected that, with the ANCHOR trial in 2015, many sites’ capacity for anoscopy may have increased, and the wait times may have gone down.
“One of the most important pieces of the study is actually the time period in which it was conducted,” said Dr. Lahiri, who in 2015 became the clinic’s second physician trained in anoscopy. Currently, more than 200 people at the Ponce De Leon Center are enrolled in the ANCHOR trial. In addition, the general capacity for performing anoscopies has gone up nationwide as a result of the trial, which required that more providers learn how to properly perform an HRA. Many clinicians are not routinely trained in performing HRA, including gastroenterologists and surgeons, Dr. Lahiri said.
“It would be interesting to look at the differences, with the start of ANCHOR being the time point for before and after,” she said.
This article first appeared on Medscape.com.
That delay “revealed missed opportunities for a better experience on the patient, clinic, and provider level,” Jessica Wells, PhD, research assistant professor at the Nell Hodgson Woodruff School of Nursing at Emory University, Atlanta, said in an interview. After all, “a lot can happen in that 1 year,” including early development of human papillomavirus (HPV)–associated anal cancer.
Although it’s too soon to say how significant that delay is with respect to the natural history of anal cancer, Dr. Wells said the data are a potential signal of disparities.
“The findings from my study may foreshadow potential disparities if we don’t have the necessary resources in place to promote follow-up care after an abnormal Pap test, similar to the disparities that we see in cervical cancer,” she said during the virtual Association of Nurses in AIDS Care 2020 Annual Meeting.
Single-center study
In the United States, people living with HIV are 19 times more likely to develop anal cancer than the general population, according to a 2018 article in the Journal of Clinical Oncology. Another single-center study from Yale University found that, in minority communities, anal cancer rates were 75% higher than in White communities. Anal cancer rates were 72% higher in communities with greater poverty. As a result, many clinics are beginning to administer Pap tests to determine early signs of HPV infection and associated changes.
In Dr. Wells’ study, which was conducted from 2012 to 2015, 150 adults with HIV who were aged 21 and older were recruited from Grady Ponce De Leon Center in Atlanta. According to a 2018 study from that center, a large minority of participants had late-stage HIV and suppressed immune systems.
All participants had been referred for HRA after a recent abnormal anal Pap test. Participants filled out questionnaires on sociodemographics, internalized HIV-related stigma, depression, risk behaviors, social support, and knowledge about HPV and anal cancer.
Participants were disproportionately older (mean age, 50.9 years); cisgender (86.7%), Black (78%); and gay, lesbian, or bisexual (84.3%). Slightly more than 1 in 10 participants (11.3%) were transgender women.
Although for 6% of participants, Pap test results indicated high-grade squamous intraepithelial lesions (HSIL), an additional 8% had atypical Pap findings that couldn’t exclude HSIL – the kinds of results that are one step away from a cancer diagnosis. More than 80% of participants had low-grade or inconclusive results. Nearly half (44%) of participants’ Pap tests revealed low-grade squamous cell intraepithelial cell lesions (LSIL); 42% indicated atypical squamous cells of undetermined significance.
When Dr. Wells looked at how long participants had waited to undergo HRA, she found something that surprised her: although some participants underwent follow-up assessment in 17 days, for many, it took much longer. The longest wait was 2,350 days – more than 6 years.
“There were quite a few patients who had follow-up beyond 1,000-plus days,” Dr. Wells said in an interview. “I didn›t think the delays were that long — at most, I would say that patients will get scheduled and come back within a few weeks or months.”
What’s more, she discovered through the HPV knowledge questionnaire that many participants did not understand why they were having a follow-up appointment. Anecdotally, some confused HPV with HIV.
“There’s education to be done to inform this target population that those living with HIV are more prone or at increased risk of this virus causing cancer later,” she said. “There are a lot of campaigns around women living with HIV, that they need to do cervical cancer screening. I think we need to really expand this campaign to include that HPV can also cause anal cancer.”
Dr. Wells had planned to primarily investigate the impact of psychosocial factors on wait time to follow-up, but none of those factors were associated with longer wait times.
Systems-level factors
That led Ann Gakumo, PhD, chair of nursing at the College of Nursing and Health Sciences of the University of Massachusetts, Boston, to ask what other factors could account for the delay.
There were several, Dr. Wells said. Precarious housing, for example, could have influenced this lag in follow-up. About one in four participants were in transient housing, and one participant reported having been incarcerated. She gathered street addresses and plans to analyze that data to see whether the cases occurred in clusters in specific neighborhoods, as the Yale data indicated.
In addition, the anoscopy clinic was only available to receive patients one day a week and was staffed with only one clinician who was trained to perform HRA. Wait times could stretch for hours. Sometimes, participants had to leave the clinic to attend to other business, and their appointments needed to be rescheduled, Wells said.
In addition to the sometimes poor understanding of the importance of the follow-up test, Dr. Wells said, “we start to see a layering of these barriers. That’s where we start seeing breakdowns. So I’m hoping in a larger study I can address some of these barriers on a multilevel approach.”
This resonated with Dr. Gakumo.
“Oftentimes, we put so much of the responsibility for this on the part of the client and not enough on the part of the provider or on the systems level,” she said.
Guiding guidelines
Guidelines on follow-up for abnormal anal Pap test results are scarce, mostly because, unlike cervical cancer, the natural history of HPV-related anal cancers hasn’t been established. The HIV Medical Association does recommend anal Pap tests, but only in cases in which “access to appropriate referral for follow-up, including high-resolution anoscopy, is available.”
In an interview, Cecile Lahiri, MD, assistant professor of infectious disease at Emory University, said that, at Ponce De Leon Center, they recommend an anal Pap for women with HIV who have a history of cervical dysplasia.
There is a reliable association between high-grade abnormal Pap tests and cervical cancer, although low-grade changes can resolve on their own. In the case of anal cancer, especially in patients with HIV, low-grade cell changes are predictive; moreover, for such patients, anal cancer is more likely to recur and is harder to treat, Dr. Lahiri said.
“The cervical environment and the anal environment are very different,” said Dr. Lahiri, who works at the Grady Ponce De Leon Center but was not involved in Dr. Wells’ study. Dr. Lahiri is also a coinvestigator of the multisite, randomized, controlled Anal Cancer HSIL Outcomes Research (ANCHOR) study, which seeks to establish whether early treatment of high-grade anal Pap changes is better than a watch-and-wait approach.
Dr. Lahiri said that when the results of that trial become available, they are more likely to know how important early anoscopy and treatment are. The findings should inform guidelines and insurance coverage of anal Pap tests and anoscopy.
In the meantime, she said, she suspected that, with the ANCHOR trial in 2015, many sites’ capacity for anoscopy may have increased, and the wait times may have gone down.
“One of the most important pieces of the study is actually the time period in which it was conducted,” said Dr. Lahiri, who in 2015 became the clinic’s second physician trained in anoscopy. Currently, more than 200 people at the Ponce De Leon Center are enrolled in the ANCHOR trial. In addition, the general capacity for performing anoscopies has gone up nationwide as a result of the trial, which required that more providers learn how to properly perform an HRA. Many clinicians are not routinely trained in performing HRA, including gastroenterologists and surgeons, Dr. Lahiri said.
“It would be interesting to look at the differences, with the start of ANCHOR being the time point for before and after,” she said.
This article first appeared on Medscape.com.