User login
Untargeted CT scans leads to overdiagnoses in lung cancer
Such overdiagnosis was illustrated in a recent study from JAMA Internal Medicine of women in Taiwan who were screened for lung cancer using low-dose CT (LDCT).
The study, and the accompanying editorial, pointed out the potential for large databases of routine clinical data to track long-term outcomes, and potentially identify patient subgroups that could benefit from early diagnosis using digital technologies.
The Taiwanese findings echo a similar trend identified in a 2018 post hoc analysis of the Danish Lung Cancer Screening Trial, which estimated that 67.2% of cancers found during that CT screening program for current or former smokers were overdiagnosed. The authors recommended that researchers report rates of overdiagnosis in future screening studies.
The authors of the Taiwan study noted that LDCT is low cost and is frequently offered to individuals who are not considered at high risk of lung cancer, and advertisements in Taiwan often target women, who rarely smoke. The researchers examined data from the Taiwan National Cancer Registry. They looked for evidence of an increased incidence of early-stage detection and reduced incidence of late-stage diagnosis. They found that, from 2004 to 2018, there was an increase of lung cancer incidence from 2.3 to 14.4 per 100,000 (difference, 12.1; 95% confidence interval, 11.3-12.8), but no significant difference in the incidence of late-stage disease (from 18.7 to 19.3 per 100,000; difference, 0.6; 95% CI, –0.5 to 1.7).
“This combination of findings, an additional 12.1 early-stage cancers per 100,000 population and no reduction in late-stage cancers, is strongly suggestive of overdiagnosis,” the authors wrote.
It can be difficult to convince people of the potential harms of overdiagnosis, especially when patients have a nodule removed and remain healthy years later. “It’s very counterintuitive, but it’s a reality, and I think this paper paints the reality very, very clearly,” said Daniel Capurro, MD, PhD, deputy director of the Centre for the Digital Transformation of Health at University of Melbourne, and an author of the editorial.
The issue is that some lung cancers progress so slowly that they may never cause a problem clinically, and their removal can lead to unnecessary cost and risk. And it’s not just cancer. “There are a bunch of other conditions that are defined by specific criteria, but we don’t add the prognosis to that definition. At the individual patient level, we don’t know the prognosis,” Dr. Capurro said.
Dr. Capurro discussed the increasing use of digital technologies like smartphone apps. Machine learning can potentially use such data to diagnose conditions like sleep or mood disorders before they become clinically significant, allowing earlier intervention, but they could also lead to overdiagnosis. Dr. Capurro proposed using longitudinal databases to track patient outcomes, which could be applied to digital screening technologies.
“You might be able to find unknown patterns that help discriminate between these pathological definitions. You should be able to train (digital screens) with the pathological definition plus the disease trajectory as a way to improve that label,” he said.
The study was funded by the Taiwan Ministry of Health and Welfare Clinical Trial Center.
Such overdiagnosis was illustrated in a recent study from JAMA Internal Medicine of women in Taiwan who were screened for lung cancer using low-dose CT (LDCT).
The study, and the accompanying editorial, pointed out the potential for large databases of routine clinical data to track long-term outcomes, and potentially identify patient subgroups that could benefit from early diagnosis using digital technologies.
The Taiwanese findings echo a similar trend identified in a 2018 post hoc analysis of the Danish Lung Cancer Screening Trial, which estimated that 67.2% of cancers found during that CT screening program for current or former smokers were overdiagnosed. The authors recommended that researchers report rates of overdiagnosis in future screening studies.
The authors of the Taiwan study noted that LDCT is low cost and is frequently offered to individuals who are not considered at high risk of lung cancer, and advertisements in Taiwan often target women, who rarely smoke. The researchers examined data from the Taiwan National Cancer Registry. They looked for evidence of an increased incidence of early-stage detection and reduced incidence of late-stage diagnosis. They found that, from 2004 to 2018, there was an increase of lung cancer incidence from 2.3 to 14.4 per 100,000 (difference, 12.1; 95% confidence interval, 11.3-12.8), but no significant difference in the incidence of late-stage disease (from 18.7 to 19.3 per 100,000; difference, 0.6; 95% CI, –0.5 to 1.7).
“This combination of findings, an additional 12.1 early-stage cancers per 100,000 population and no reduction in late-stage cancers, is strongly suggestive of overdiagnosis,” the authors wrote.
It can be difficult to convince people of the potential harms of overdiagnosis, especially when patients have a nodule removed and remain healthy years later. “It’s very counterintuitive, but it’s a reality, and I think this paper paints the reality very, very clearly,” said Daniel Capurro, MD, PhD, deputy director of the Centre for the Digital Transformation of Health at University of Melbourne, and an author of the editorial.
The issue is that some lung cancers progress so slowly that they may never cause a problem clinically, and their removal can lead to unnecessary cost and risk. And it’s not just cancer. “There are a bunch of other conditions that are defined by specific criteria, but we don’t add the prognosis to that definition. At the individual patient level, we don’t know the prognosis,” Dr. Capurro said.
Dr. Capurro discussed the increasing use of digital technologies like smartphone apps. Machine learning can potentially use such data to diagnose conditions like sleep or mood disorders before they become clinically significant, allowing earlier intervention, but they could also lead to overdiagnosis. Dr. Capurro proposed using longitudinal databases to track patient outcomes, which could be applied to digital screening technologies.
“You might be able to find unknown patterns that help discriminate between these pathological definitions. You should be able to train (digital screens) with the pathological definition plus the disease trajectory as a way to improve that label,” he said.
The study was funded by the Taiwan Ministry of Health and Welfare Clinical Trial Center.
Such overdiagnosis was illustrated in a recent study from JAMA Internal Medicine of women in Taiwan who were screened for lung cancer using low-dose CT (LDCT).
The study, and the accompanying editorial, pointed out the potential for large databases of routine clinical data to track long-term outcomes, and potentially identify patient subgroups that could benefit from early diagnosis using digital technologies.
The Taiwanese findings echo a similar trend identified in a 2018 post hoc analysis of the Danish Lung Cancer Screening Trial, which estimated that 67.2% of cancers found during that CT screening program for current or former smokers were overdiagnosed. The authors recommended that researchers report rates of overdiagnosis in future screening studies.
The authors of the Taiwan study noted that LDCT is low cost and is frequently offered to individuals who are not considered at high risk of lung cancer, and advertisements in Taiwan often target women, who rarely smoke. The researchers examined data from the Taiwan National Cancer Registry. They looked for evidence of an increased incidence of early-stage detection and reduced incidence of late-stage diagnosis. They found that, from 2004 to 2018, there was an increase of lung cancer incidence from 2.3 to 14.4 per 100,000 (difference, 12.1; 95% confidence interval, 11.3-12.8), but no significant difference in the incidence of late-stage disease (from 18.7 to 19.3 per 100,000; difference, 0.6; 95% CI, –0.5 to 1.7).
“This combination of findings, an additional 12.1 early-stage cancers per 100,000 population and no reduction in late-stage cancers, is strongly suggestive of overdiagnosis,” the authors wrote.
It can be difficult to convince people of the potential harms of overdiagnosis, especially when patients have a nodule removed and remain healthy years later. “It’s very counterintuitive, but it’s a reality, and I think this paper paints the reality very, very clearly,” said Daniel Capurro, MD, PhD, deputy director of the Centre for the Digital Transformation of Health at University of Melbourne, and an author of the editorial.
The issue is that some lung cancers progress so slowly that they may never cause a problem clinically, and their removal can lead to unnecessary cost and risk. And it’s not just cancer. “There are a bunch of other conditions that are defined by specific criteria, but we don’t add the prognosis to that definition. At the individual patient level, we don’t know the prognosis,” Dr. Capurro said.
Dr. Capurro discussed the increasing use of digital technologies like smartphone apps. Machine learning can potentially use such data to diagnose conditions like sleep or mood disorders before they become clinically significant, allowing earlier intervention, but they could also lead to overdiagnosis. Dr. Capurro proposed using longitudinal databases to track patient outcomes, which could be applied to digital screening technologies.
“You might be able to find unknown patterns that help discriminate between these pathological definitions. You should be able to train (digital screens) with the pathological definition plus the disease trajectory as a way to improve that label,” he said.
The study was funded by the Taiwan Ministry of Health and Welfare Clinical Trial Center.
FROM JAMA INTERNAL MEDICINE
Ways to lessen toxic effects of chemo in older adults
Age-related changes that potentiate adverse drug reactions include alterations in absorption, distribution, metabolism, and excretion. As such, older patients often require adjustments in medications to optimize safety and use. Medication adjustment is especially important for older patients on complex medication regimens for multiple conditions, such as those undergoing cancer treatment. Three recent high-quality randomized trials evaluated the use of geriatric assessment (GA) in older adults with cancer.1-3
Interdisciplinary GA can identify aging-related conditions associated with poor outcomes in older patients with cancer (e.g., toxic effects of chemotherapy) and provide recommendations aimed at improving health outcomes. The results of these trials suggest that interdisciplinary GA can improve care outcomes and oncologists’ communication for older adults with cancer, and should be considered an emerging standard of care.
Geriatric assessment and chemotherapy-related toxic effects
A cluster randomized trial1 at City of Hope National Medical Center conducted between August 2015 and February 2019 enrolled 613 participants and randomly assigned them to receive a GA-guided intervention or usual standard of care in a 2-to-1 ratio. Participants were eligible for the study if they were aged ≥65 years; had a diagnosis of solid malignant neoplasm of any stage; were starting a new chemotherapy regimen; and were fluent in English, Spanish, or Chinese.
The intervention included a GA at baseline followed by assessments focused on six common areas: sleep problems, problems with eating and feeding, incontinence, confusion, evidence of falls, and skin breakdown. An interdisciplinary team (oncologist, nurse practitioner, pharmacist, physical therapist, occupational therapist, social worker, and nutritionist) performed the assessment and developed a plan of care. Interventions were multifactorial and could include referral to specialists; recommendations for medication changes; symptom management; nutritional intervention with diet recommendations and supplementation; and interventions targeting social, spiritual, and functional well-being. Follow-up by a nurse practitioner continued until completion of chemotherapy or 6 months after starting chemotherapy, whichever was earlier.
The primary outcome was grade 3 or higher chemotherapy-related toxic effects using National Cancer Institute criteria, and secondary outcomes were advance directive completion, emergency room visits and unplanned hospitalizations, and survival up to 12 months. Results showed a 10% absolute reduction in the incidence of grade 3 or higher toxic effects (P = .02), with a number needed to treat of 10. Advance directive completion also increased by 15%, but no differences were observed for other outcomes. This study offers high-quality evidence that a GA-based intervention can reduce toxic effects of chemotherapy regimens for older adults with cancer.
Geriatric assessment in community oncology practices
A recent study by Supriya G. Mohile, MD, and colleagues2 is the first nationwide multicenter clinical trial to demonstrate the effects of GA and GA-guided management. This study was conducted in 40 oncology practices from the University of Rochester National Cancer Institute Community Oncology Research Program network. Centers were randomly assigned to intervention or usual care (362 patients treated by 68 oncologists in the intervention group and 371 patients treated by 91 oncologists in the usual-care group). Eligibility criteria were age ≥70 years; impairment in at least one GA domain other than polypharmacy; incurable advanced solid tumor or lymphoma with a plan to start new cancer treatment with a high risk for toxic effects within 4 weeks; and English language fluency. Both study groups underwent a baseline GA that assessed patients’ physical performance, functional status, comorbidity, cognition, nutrition, social support, polypharmacy, and psychological status. For the intervention group, a summary and management recommendations were provided to the treating oncologists.
The primary outcome was grade 3 or higher toxic effects within 3 months of starting a new regimen; secondary outcomes included treatment intensity and survival and GA outcomes within 3 months. A smaller proportion of patients in the intervention group experienced toxicity (51% vs. 71%), with an absolute risk reduction of 20%. Patients in the intervention group also had fewer falls and a greater reduction in medications used; there were no other differences in secondary outcomes. This study offers very strong and generalizable evidence that incorporating GA in the care of older adults with cancer at risk for toxicity can reduce toxicity as well as improve other outcomes, such as falls and polypharmacy.
Geriatric assessment and oncologist-patient communication
A secondary analysis3 of data from Dr. Mohile and colleagues2 evaluated the effect of GA-guided recommendations on oncologist-patient communication regarding comorbidities. Patients (n = 541) included in this analysis were 76.6 years of age on average and had 3.2 (standard deviation, 1.9) comorbid conditions. All patients underwent GA, but only oncologists in the intervention arm received GA-based recommendations. Clinical encounters between oncologist and patient immediately following the GA were audio recorded and analyzed to examine communication between oncologists and participants as it relates to chronic comorbid conditions.
In the intervention arm, more discussions regarding comorbidities took place, and more participants’ concerns about comorbidities were acknowledged. More importantly, participants in the intervention group were 2.4 times more likely to have their concerns about comorbidities addressed through referral or education, compared with the usual-care group (P = .004). Moreover, 41% of oncologists in the intervention arm modified dosage or cancer treatment schedule because of concern about tolerability or comorbidities. This study demonstrates beneficial effects of GA in increasing communication and perhaps consideration of comorbidities of older adults when planning cancer treatment.
Dr. Hung is professor of geriatrics and palliative care at Mount Sinai Hospital, New York. He disclosed no relevant conflicts of interest.
References
1. Li D et al. JAMA Oncol. 2021;7:e214158.
2. Mohile SG et al. Lancet. 2021;398:1894-1904.
3. Kleckner AS et al. JCO Oncol Pract. 2022;18:e9-19.
A version of this article first appeared on Medscape.com.
Age-related changes that potentiate adverse drug reactions include alterations in absorption, distribution, metabolism, and excretion. As such, older patients often require adjustments in medications to optimize safety and use. Medication adjustment is especially important for older patients on complex medication regimens for multiple conditions, such as those undergoing cancer treatment. Three recent high-quality randomized trials evaluated the use of geriatric assessment (GA) in older adults with cancer.1-3
Interdisciplinary GA can identify aging-related conditions associated with poor outcomes in older patients with cancer (e.g., toxic effects of chemotherapy) and provide recommendations aimed at improving health outcomes. The results of these trials suggest that interdisciplinary GA can improve care outcomes and oncologists’ communication for older adults with cancer, and should be considered an emerging standard of care.
Geriatric assessment and chemotherapy-related toxic effects
A cluster randomized trial1 at City of Hope National Medical Center conducted between August 2015 and February 2019 enrolled 613 participants and randomly assigned them to receive a GA-guided intervention or usual standard of care in a 2-to-1 ratio. Participants were eligible for the study if they were aged ≥65 years; had a diagnosis of solid malignant neoplasm of any stage; were starting a new chemotherapy regimen; and were fluent in English, Spanish, or Chinese.
The intervention included a GA at baseline followed by assessments focused on six common areas: sleep problems, problems with eating and feeding, incontinence, confusion, evidence of falls, and skin breakdown. An interdisciplinary team (oncologist, nurse practitioner, pharmacist, physical therapist, occupational therapist, social worker, and nutritionist) performed the assessment and developed a plan of care. Interventions were multifactorial and could include referral to specialists; recommendations for medication changes; symptom management; nutritional intervention with diet recommendations and supplementation; and interventions targeting social, spiritual, and functional well-being. Follow-up by a nurse practitioner continued until completion of chemotherapy or 6 months after starting chemotherapy, whichever was earlier.
The primary outcome was grade 3 or higher chemotherapy-related toxic effects using National Cancer Institute criteria, and secondary outcomes were advance directive completion, emergency room visits and unplanned hospitalizations, and survival up to 12 months. Results showed a 10% absolute reduction in the incidence of grade 3 or higher toxic effects (P = .02), with a number needed to treat of 10. Advance directive completion also increased by 15%, but no differences were observed for other outcomes. This study offers high-quality evidence that a GA-based intervention can reduce toxic effects of chemotherapy regimens for older adults with cancer.
Geriatric assessment in community oncology practices
A recent study by Supriya G. Mohile, MD, and colleagues2 is the first nationwide multicenter clinical trial to demonstrate the effects of GA and GA-guided management. This study was conducted in 40 oncology practices from the University of Rochester National Cancer Institute Community Oncology Research Program network. Centers were randomly assigned to intervention or usual care (362 patients treated by 68 oncologists in the intervention group and 371 patients treated by 91 oncologists in the usual-care group). Eligibility criteria were age ≥70 years; impairment in at least one GA domain other than polypharmacy; incurable advanced solid tumor or lymphoma with a plan to start new cancer treatment with a high risk for toxic effects within 4 weeks; and English language fluency. Both study groups underwent a baseline GA that assessed patients’ physical performance, functional status, comorbidity, cognition, nutrition, social support, polypharmacy, and psychological status. For the intervention group, a summary and management recommendations were provided to the treating oncologists.
The primary outcome was grade 3 or higher toxic effects within 3 months of starting a new regimen; secondary outcomes included treatment intensity and survival and GA outcomes within 3 months. A smaller proportion of patients in the intervention group experienced toxicity (51% vs. 71%), with an absolute risk reduction of 20%. Patients in the intervention group also had fewer falls and a greater reduction in medications used; there were no other differences in secondary outcomes. This study offers very strong and generalizable evidence that incorporating GA in the care of older adults with cancer at risk for toxicity can reduce toxicity as well as improve other outcomes, such as falls and polypharmacy.
Geriatric assessment and oncologist-patient communication
A secondary analysis3 of data from Dr. Mohile and colleagues2 evaluated the effect of GA-guided recommendations on oncologist-patient communication regarding comorbidities. Patients (n = 541) included in this analysis were 76.6 years of age on average and had 3.2 (standard deviation, 1.9) comorbid conditions. All patients underwent GA, but only oncologists in the intervention arm received GA-based recommendations. Clinical encounters between oncologist and patient immediately following the GA were audio recorded and analyzed to examine communication between oncologists and participants as it relates to chronic comorbid conditions.
In the intervention arm, more discussions regarding comorbidities took place, and more participants’ concerns about comorbidities were acknowledged. More importantly, participants in the intervention group were 2.4 times more likely to have their concerns about comorbidities addressed through referral or education, compared with the usual-care group (P = .004). Moreover, 41% of oncologists in the intervention arm modified dosage or cancer treatment schedule because of concern about tolerability or comorbidities. This study demonstrates beneficial effects of GA in increasing communication and perhaps consideration of comorbidities of older adults when planning cancer treatment.
Dr. Hung is professor of geriatrics and palliative care at Mount Sinai Hospital, New York. He disclosed no relevant conflicts of interest.
References
1. Li D et al. JAMA Oncol. 2021;7:e214158.
2. Mohile SG et al. Lancet. 2021;398:1894-1904.
3. Kleckner AS et al. JCO Oncol Pract. 2022;18:e9-19.
A version of this article first appeared on Medscape.com.
Age-related changes that potentiate adverse drug reactions include alterations in absorption, distribution, metabolism, and excretion. As such, older patients often require adjustments in medications to optimize safety and use. Medication adjustment is especially important for older patients on complex medication regimens for multiple conditions, such as those undergoing cancer treatment. Three recent high-quality randomized trials evaluated the use of geriatric assessment (GA) in older adults with cancer.1-3
Interdisciplinary GA can identify aging-related conditions associated with poor outcomes in older patients with cancer (e.g., toxic effects of chemotherapy) and provide recommendations aimed at improving health outcomes. The results of these trials suggest that interdisciplinary GA can improve care outcomes and oncologists’ communication for older adults with cancer, and should be considered an emerging standard of care.
Geriatric assessment and chemotherapy-related toxic effects
A cluster randomized trial1 at City of Hope National Medical Center conducted between August 2015 and February 2019 enrolled 613 participants and randomly assigned them to receive a GA-guided intervention or usual standard of care in a 2-to-1 ratio. Participants were eligible for the study if they were aged ≥65 years; had a diagnosis of solid malignant neoplasm of any stage; were starting a new chemotherapy regimen; and were fluent in English, Spanish, or Chinese.
The intervention included a GA at baseline followed by assessments focused on six common areas: sleep problems, problems with eating and feeding, incontinence, confusion, evidence of falls, and skin breakdown. An interdisciplinary team (oncologist, nurse practitioner, pharmacist, physical therapist, occupational therapist, social worker, and nutritionist) performed the assessment and developed a plan of care. Interventions were multifactorial and could include referral to specialists; recommendations for medication changes; symptom management; nutritional intervention with diet recommendations and supplementation; and interventions targeting social, spiritual, and functional well-being. Follow-up by a nurse practitioner continued until completion of chemotherapy or 6 months after starting chemotherapy, whichever was earlier.
The primary outcome was grade 3 or higher chemotherapy-related toxic effects using National Cancer Institute criteria, and secondary outcomes were advance directive completion, emergency room visits and unplanned hospitalizations, and survival up to 12 months. Results showed a 10% absolute reduction in the incidence of grade 3 or higher toxic effects (P = .02), with a number needed to treat of 10. Advance directive completion also increased by 15%, but no differences were observed for other outcomes. This study offers high-quality evidence that a GA-based intervention can reduce toxic effects of chemotherapy regimens for older adults with cancer.
Geriatric assessment in community oncology practices
A recent study by Supriya G. Mohile, MD, and colleagues2 is the first nationwide multicenter clinical trial to demonstrate the effects of GA and GA-guided management. This study was conducted in 40 oncology practices from the University of Rochester National Cancer Institute Community Oncology Research Program network. Centers were randomly assigned to intervention or usual care (362 patients treated by 68 oncologists in the intervention group and 371 patients treated by 91 oncologists in the usual-care group). Eligibility criteria were age ≥70 years; impairment in at least one GA domain other than polypharmacy; incurable advanced solid tumor or lymphoma with a plan to start new cancer treatment with a high risk for toxic effects within 4 weeks; and English language fluency. Both study groups underwent a baseline GA that assessed patients’ physical performance, functional status, comorbidity, cognition, nutrition, social support, polypharmacy, and psychological status. For the intervention group, a summary and management recommendations were provided to the treating oncologists.
The primary outcome was grade 3 or higher toxic effects within 3 months of starting a new regimen; secondary outcomes included treatment intensity and survival and GA outcomes within 3 months. A smaller proportion of patients in the intervention group experienced toxicity (51% vs. 71%), with an absolute risk reduction of 20%. Patients in the intervention group also had fewer falls and a greater reduction in medications used; there were no other differences in secondary outcomes. This study offers very strong and generalizable evidence that incorporating GA in the care of older adults with cancer at risk for toxicity can reduce toxicity as well as improve other outcomes, such as falls and polypharmacy.
Geriatric assessment and oncologist-patient communication
A secondary analysis3 of data from Dr. Mohile and colleagues2 evaluated the effect of GA-guided recommendations on oncologist-patient communication regarding comorbidities. Patients (n = 541) included in this analysis were 76.6 years of age on average and had 3.2 (standard deviation, 1.9) comorbid conditions. All patients underwent GA, but only oncologists in the intervention arm received GA-based recommendations. Clinical encounters between oncologist and patient immediately following the GA were audio recorded and analyzed to examine communication between oncologists and participants as it relates to chronic comorbid conditions.
In the intervention arm, more discussions regarding comorbidities took place, and more participants’ concerns about comorbidities were acknowledged. More importantly, participants in the intervention group were 2.4 times more likely to have their concerns about comorbidities addressed through referral or education, compared with the usual-care group (P = .004). Moreover, 41% of oncologists in the intervention arm modified dosage or cancer treatment schedule because of concern about tolerability or comorbidities. This study demonstrates beneficial effects of GA in increasing communication and perhaps consideration of comorbidities of older adults when planning cancer treatment.
Dr. Hung is professor of geriatrics and palliative care at Mount Sinai Hospital, New York. He disclosed no relevant conflicts of interest.
References
1. Li D et al. JAMA Oncol. 2021;7:e214158.
2. Mohile SG et al. Lancet. 2021;398:1894-1904.
3. Kleckner AS et al. JCO Oncol Pract. 2022;18:e9-19.
A version of this article first appeared on Medscape.com.
Intensity-modulated radiotherapy reduces esophagitis in palliative NSCLC
Reducing the dose of radiation to the esophagus during palliative radiotherapy for advanced lung cancer significantly reduced the incidence of esophagitis among a small group of patients, according to a new randomized study.
The study, called Palliative Radiation for Advanced Central Lung Tumors with Intentional Avoidance of the Esophagus (PROACTIVE), explored the use of a technique called intensity-modulated radiotherapy (IMRT) to sculpt the radiation dose around the esophagus, reducing its exposure. IMRT is a standard technique to avoid healthy tissue with higher radiation doses in the curative setting, but it hasn’t been explored much in the palliative setting, said investigators led by Alexander V. Louie, MD, PhD, a radiation oncologist at the University of Toronto’s Odette Cancer Centre.
The study included 90 patients (mean age 70 years, 56% female) with stage 3 and 4 non–small cell lung cancer. They were randomized evenly to standard radiotherapy with the esophagus getting the same dose as the tumor, or to esophagus-sparing IMRT (ES-IMRT) with the esophagus exposed to no more than 80% of the prescribed dose.
The overall survival was similar between both groups: 8.6 months for standard therapy and 8.7 months for IMRT. Forty percent of patients received 20 Gy in 5 fractions and the rest 30 Gy in 10 fractions. The reduction in esophagitis with IMRT was most evident in the 30 Gy group.
Only one patient in the esophagus-sparing group developed grade 2 esophagitis versus 11 patients (24%) in the standard radiotherapy group. There were no grade 3 or higher cases. There was also an almost 4-point improvement (54.3 points with ES-IMRT versus 50.5 points) on an esophagus-related quality of life (QOL) measure, which is a subscale of the Functional Assessment of Cancer Therapy: Esophagus questionnaire, but it wasn’t statistically significant (P = 0.06).
“The ES-IMRT technique we describe herein represents a paradigm shift in palliative radiotherapy planning,” the investigators wrote. “This technique holds merit for translation into clinical practice.”
However, in their editorial, Ashley A. Weiner, MD, PhD, and Joel E. Tepper, MD, of the Lineberger Comprehensive Cancer Center at the University of North Carolina at Chapel Hill, wrote that it is too preliminary to recommend esophagus-sparing IMRT to patients. “In the absence of meeting the primary quality of life end point and without demonstration of adequate symptom palliation, one cannot recommend ES-IMRT as a standard therapy for palliation of thoracic symptoms due to NSCLC,” they wrote.
They said the study raises an important issue: The balance between tumor coverage and sparing healthy tissue when symptom palliation instead of cure is the goal.
Striking the right balance “is challenging and part of the art of radiotherapy” particularly in the palliative setting, where there are many unanswered questions, they said.
Unlike in curative intent scenarios, “the ideal dose to the tumor to provide a maximal palliative benefit is unknown, and perhaps there is no ideal dose,” Dr. Weiner and Dr. Tepper said.
It’s also unclear whether the entire tumor needs to be irradiated to the full dose when the goal is simply to shrink the tumor and relieve symptoms. “Perhaps a lower dose to a portion of the tumor makes sense,” especially when radiation doses for palliation are “somewhat arbitrary,” they said.
Indeed, the portion of the tumor next to the esophagus in the IMRT subjects was necessarily undercovered to achieve the esophagus-sparing effect. “It seems very likely to us that underdosing a small portion of the tumor will have little adverse effect” on palliation, Dr. Weiner and Dr. Tepper wrote.
Also, “if undercovering” the tumor with lower doses “results in adequate tumor reduction for palliation,” they wondered if IMRT – a more expensive and complex technique than standard radiotherapy – is even needed.
The work was funded by the Canadian Cancer Society. Dr. Louie reported payments from AstraZeneca as an advisor and personal fees from Varian Medical Systems and Reflexion, the makers of IMRT technology. The authors of the editorial reported no conflicts of interest.
Reducing the dose of radiation to the esophagus during palliative radiotherapy for advanced lung cancer significantly reduced the incidence of esophagitis among a small group of patients, according to a new randomized study.
The study, called Palliative Radiation for Advanced Central Lung Tumors with Intentional Avoidance of the Esophagus (PROACTIVE), explored the use of a technique called intensity-modulated radiotherapy (IMRT) to sculpt the radiation dose around the esophagus, reducing its exposure. IMRT is a standard technique to avoid healthy tissue with higher radiation doses in the curative setting, but it hasn’t been explored much in the palliative setting, said investigators led by Alexander V. Louie, MD, PhD, a radiation oncologist at the University of Toronto’s Odette Cancer Centre.
The study included 90 patients (mean age 70 years, 56% female) with stage 3 and 4 non–small cell lung cancer. They were randomized evenly to standard radiotherapy with the esophagus getting the same dose as the tumor, or to esophagus-sparing IMRT (ES-IMRT) with the esophagus exposed to no more than 80% of the prescribed dose.
The overall survival was similar between both groups: 8.6 months for standard therapy and 8.7 months for IMRT. Forty percent of patients received 20 Gy in 5 fractions and the rest 30 Gy in 10 fractions. The reduction in esophagitis with IMRT was most evident in the 30 Gy group.
Only one patient in the esophagus-sparing group developed grade 2 esophagitis versus 11 patients (24%) in the standard radiotherapy group. There were no grade 3 or higher cases. There was also an almost 4-point improvement (54.3 points with ES-IMRT versus 50.5 points) on an esophagus-related quality of life (QOL) measure, which is a subscale of the Functional Assessment of Cancer Therapy: Esophagus questionnaire, but it wasn’t statistically significant (P = 0.06).
“The ES-IMRT technique we describe herein represents a paradigm shift in palliative radiotherapy planning,” the investigators wrote. “This technique holds merit for translation into clinical practice.”
However, in their editorial, Ashley A. Weiner, MD, PhD, and Joel E. Tepper, MD, of the Lineberger Comprehensive Cancer Center at the University of North Carolina at Chapel Hill, wrote that it is too preliminary to recommend esophagus-sparing IMRT to patients. “In the absence of meeting the primary quality of life end point and without demonstration of adequate symptom palliation, one cannot recommend ES-IMRT as a standard therapy for palliation of thoracic symptoms due to NSCLC,” they wrote.
They said the study raises an important issue: The balance between tumor coverage and sparing healthy tissue when symptom palliation instead of cure is the goal.
Striking the right balance “is challenging and part of the art of radiotherapy” particularly in the palliative setting, where there are many unanswered questions, they said.
Unlike in curative intent scenarios, “the ideal dose to the tumor to provide a maximal palliative benefit is unknown, and perhaps there is no ideal dose,” Dr. Weiner and Dr. Tepper said.
It’s also unclear whether the entire tumor needs to be irradiated to the full dose when the goal is simply to shrink the tumor and relieve symptoms. “Perhaps a lower dose to a portion of the tumor makes sense,” especially when radiation doses for palliation are “somewhat arbitrary,” they said.
Indeed, the portion of the tumor next to the esophagus in the IMRT subjects was necessarily undercovered to achieve the esophagus-sparing effect. “It seems very likely to us that underdosing a small portion of the tumor will have little adverse effect” on palliation, Dr. Weiner and Dr. Tepper wrote.
Also, “if undercovering” the tumor with lower doses “results in adequate tumor reduction for palliation,” they wondered if IMRT – a more expensive and complex technique than standard radiotherapy – is even needed.
The work was funded by the Canadian Cancer Society. Dr. Louie reported payments from AstraZeneca as an advisor and personal fees from Varian Medical Systems and Reflexion, the makers of IMRT technology. The authors of the editorial reported no conflicts of interest.
Reducing the dose of radiation to the esophagus during palliative radiotherapy for advanced lung cancer significantly reduced the incidence of esophagitis among a small group of patients, according to a new randomized study.
The study, called Palliative Radiation for Advanced Central Lung Tumors with Intentional Avoidance of the Esophagus (PROACTIVE), explored the use of a technique called intensity-modulated radiotherapy (IMRT) to sculpt the radiation dose around the esophagus, reducing its exposure. IMRT is a standard technique to avoid healthy tissue with higher radiation doses in the curative setting, but it hasn’t been explored much in the palliative setting, said investigators led by Alexander V. Louie, MD, PhD, a radiation oncologist at the University of Toronto’s Odette Cancer Centre.
The study included 90 patients (mean age 70 years, 56% female) with stage 3 and 4 non–small cell lung cancer. They were randomized evenly to standard radiotherapy with the esophagus getting the same dose as the tumor, or to esophagus-sparing IMRT (ES-IMRT) with the esophagus exposed to no more than 80% of the prescribed dose.
The overall survival was similar between both groups: 8.6 months for standard therapy and 8.7 months for IMRT. Forty percent of patients received 20 Gy in 5 fractions and the rest 30 Gy in 10 fractions. The reduction in esophagitis with IMRT was most evident in the 30 Gy group.
Only one patient in the esophagus-sparing group developed grade 2 esophagitis versus 11 patients (24%) in the standard radiotherapy group. There were no grade 3 or higher cases. There was also an almost 4-point improvement (54.3 points with ES-IMRT versus 50.5 points) on an esophagus-related quality of life (QOL) measure, which is a subscale of the Functional Assessment of Cancer Therapy: Esophagus questionnaire, but it wasn’t statistically significant (P = 0.06).
“The ES-IMRT technique we describe herein represents a paradigm shift in palliative radiotherapy planning,” the investigators wrote. “This technique holds merit for translation into clinical practice.”
However, in their editorial, Ashley A. Weiner, MD, PhD, and Joel E. Tepper, MD, of the Lineberger Comprehensive Cancer Center at the University of North Carolina at Chapel Hill, wrote that it is too preliminary to recommend esophagus-sparing IMRT to patients. “In the absence of meeting the primary quality of life end point and without demonstration of adequate symptom palliation, one cannot recommend ES-IMRT as a standard therapy for palliation of thoracic symptoms due to NSCLC,” they wrote.
They said the study raises an important issue: The balance between tumor coverage and sparing healthy tissue when symptom palliation instead of cure is the goal.
Striking the right balance “is challenging and part of the art of radiotherapy” particularly in the palliative setting, where there are many unanswered questions, they said.
Unlike in curative intent scenarios, “the ideal dose to the tumor to provide a maximal palliative benefit is unknown, and perhaps there is no ideal dose,” Dr. Weiner and Dr. Tepper said.
It’s also unclear whether the entire tumor needs to be irradiated to the full dose when the goal is simply to shrink the tumor and relieve symptoms. “Perhaps a lower dose to a portion of the tumor makes sense,” especially when radiation doses for palliation are “somewhat arbitrary,” they said.
Indeed, the portion of the tumor next to the esophagus in the IMRT subjects was necessarily undercovered to achieve the esophagus-sparing effect. “It seems very likely to us that underdosing a small portion of the tumor will have little adverse effect” on palliation, Dr. Weiner and Dr. Tepper wrote.
Also, “if undercovering” the tumor with lower doses “results in adequate tumor reduction for palliation,” they wondered if IMRT – a more expensive and complex technique than standard radiotherapy – is even needed.
The work was funded by the Canadian Cancer Society. Dr. Louie reported payments from AstraZeneca as an advisor and personal fees from Varian Medical Systems and Reflexion, the makers of IMRT technology. The authors of the editorial reported no conflicts of interest.
FROM JAMA ONCOLOGY
Lung cancer with ILD patients fare poorly after thoracic radiotherapy
Most lung cancer patients with interstitial lung disease will not benefit from thoracic radiotherapy, based on data from a systematic review of 24 studies.
Thoracic radiotherapy remains a key part of lung cancer treatment for early and metastatic disease. However, patients with both small cell lung cancer (SCLC) and non–small cell lung cancer (NSCLC) associated with interstitial lung disease (ILD) fare worse than do those without ILD, often because of acute exacerbation of ILD and severe or fatal pneumonitis, wrote Animesh Saha, MD, of Apollo Multi-Specialty Hospitals, Kolkata, India, and colleagues. Consequently, clinicians may hesitate to offer radiotherapy to these patients.
In a review published in Clinical Oncology, the researchers identified 24 studies, including phase II and phase III randomized or nonrandomized trials, as well as prospective, observational studies and retrospective real-world studies. The goal of the review was to report the incidence and predictors of radiation pneumonitis associated with different types of thoracic radiotherapy for lung cancer patients with ILD, the researchers said. Treatment types included curative-intent fractionated radiotherapy or chemoradiotherapy or moderately hypofractionated (nonstereotactic ablative radiotherapy [SABR]) and hyperfractionated radiotherapy as well as particle beam therapies.
The studies included patients with SCLC or NSCLC and any form of ILD, including subclinical, radiologically diagnosed, or symptomatic, the researchers said.
Overall, the median incidence of grade 3 or higher radiation pneumonitis was 19.7%; the median incidence in patients treated with conventional radical radiotherapy, SABR, and particle beam therapy was 31.8%, 11.9%, and 20.25%, respectively.
Eighteen studies reported grade 5 radiation pneumonitis; the overall median incidence was 6%, but as high as 60% in some studies. When separated by treatment type, the median incidence was 2.7%, 6.25%, and 6.25%, respectively, in patients treated with radical radiotherapy (non-SABR), SABR, and particle beam therapy.
Independent predictors of severe radiation pneumonitis (grade 2 or higher and grade 3 or higher) included subclinical or radiological ILD, the researchers said. Among ILD subtypes, studies have shown increased risk for severe radiation pneumonitis among those with non-IPF or non-UIP pattern fibrosis.
In addition, patient-related factors of low forced vital capacity (FVC) and low forced expiratory volume in 1 second (FEV1), have been associated with severe radiation pneumonitis, the researchers said. They also found increased risk for patients with lower lobe tumor location compared to other lobes.
As for treatment-related factors, a history of gemcitabine chemotherapy was associated with an increased risk of grade 3 or higher radiation pneumonitis.
“There is always concern about using thoracic radiotherapy in lung cancer patients with coexisting ILD in view of the risks involved,” the researchers wrote in their discussion of the findings. “Although thoracic radiotherapy is expected to produce similar local control, overall survival is worse in lung cancer patients with ILD than without, probably due to the poor prognosis associated with ILD and associated treatment-related mortality,” they said.
The findings were limited by several factors including the heterogeneity of the studies and study population and the retrospective design of most of the studies, the researchers noted.
However, the results highlight the increased risk of severe and fatal radiation pneumonitis in lung cancer patients with ILD and the need for careful patient selection and counseling if thoracic radiotherapy is to be considered, they concluded.
The study received no outside funding. The researchers had no financial conflicts to disclose.
Most lung cancer patients with interstitial lung disease will not benefit from thoracic radiotherapy, based on data from a systematic review of 24 studies.
Thoracic radiotherapy remains a key part of lung cancer treatment for early and metastatic disease. However, patients with both small cell lung cancer (SCLC) and non–small cell lung cancer (NSCLC) associated with interstitial lung disease (ILD) fare worse than do those without ILD, often because of acute exacerbation of ILD and severe or fatal pneumonitis, wrote Animesh Saha, MD, of Apollo Multi-Specialty Hospitals, Kolkata, India, and colleagues. Consequently, clinicians may hesitate to offer radiotherapy to these patients.
In a review published in Clinical Oncology, the researchers identified 24 studies, including phase II and phase III randomized or nonrandomized trials, as well as prospective, observational studies and retrospective real-world studies. The goal of the review was to report the incidence and predictors of radiation pneumonitis associated with different types of thoracic radiotherapy for lung cancer patients with ILD, the researchers said. Treatment types included curative-intent fractionated radiotherapy or chemoradiotherapy or moderately hypofractionated (nonstereotactic ablative radiotherapy [SABR]) and hyperfractionated radiotherapy as well as particle beam therapies.
The studies included patients with SCLC or NSCLC and any form of ILD, including subclinical, radiologically diagnosed, or symptomatic, the researchers said.
Overall, the median incidence of grade 3 or higher radiation pneumonitis was 19.7%; the median incidence in patients treated with conventional radical radiotherapy, SABR, and particle beam therapy was 31.8%, 11.9%, and 20.25%, respectively.
Eighteen studies reported grade 5 radiation pneumonitis; the overall median incidence was 6%, but as high as 60% in some studies. When separated by treatment type, the median incidence was 2.7%, 6.25%, and 6.25%, respectively, in patients treated with radical radiotherapy (non-SABR), SABR, and particle beam therapy.
Independent predictors of severe radiation pneumonitis (grade 2 or higher and grade 3 or higher) included subclinical or radiological ILD, the researchers said. Among ILD subtypes, studies have shown increased risk for severe radiation pneumonitis among those with non-IPF or non-UIP pattern fibrosis.
In addition, patient-related factors of low forced vital capacity (FVC) and low forced expiratory volume in 1 second (FEV1), have been associated with severe radiation pneumonitis, the researchers said. They also found increased risk for patients with lower lobe tumor location compared to other lobes.
As for treatment-related factors, a history of gemcitabine chemotherapy was associated with an increased risk of grade 3 or higher radiation pneumonitis.
“There is always concern about using thoracic radiotherapy in lung cancer patients with coexisting ILD in view of the risks involved,” the researchers wrote in their discussion of the findings. “Although thoracic radiotherapy is expected to produce similar local control, overall survival is worse in lung cancer patients with ILD than without, probably due to the poor prognosis associated with ILD and associated treatment-related mortality,” they said.
The findings were limited by several factors including the heterogeneity of the studies and study population and the retrospective design of most of the studies, the researchers noted.
However, the results highlight the increased risk of severe and fatal radiation pneumonitis in lung cancer patients with ILD and the need for careful patient selection and counseling if thoracic radiotherapy is to be considered, they concluded.
The study received no outside funding. The researchers had no financial conflicts to disclose.
Most lung cancer patients with interstitial lung disease will not benefit from thoracic radiotherapy, based on data from a systematic review of 24 studies.
Thoracic radiotherapy remains a key part of lung cancer treatment for early and metastatic disease. However, patients with both small cell lung cancer (SCLC) and non–small cell lung cancer (NSCLC) associated with interstitial lung disease (ILD) fare worse than do those without ILD, often because of acute exacerbation of ILD and severe or fatal pneumonitis, wrote Animesh Saha, MD, of Apollo Multi-Specialty Hospitals, Kolkata, India, and colleagues. Consequently, clinicians may hesitate to offer radiotherapy to these patients.
In a review published in Clinical Oncology, the researchers identified 24 studies, including phase II and phase III randomized or nonrandomized trials, as well as prospective, observational studies and retrospective real-world studies. The goal of the review was to report the incidence and predictors of radiation pneumonitis associated with different types of thoracic radiotherapy for lung cancer patients with ILD, the researchers said. Treatment types included curative-intent fractionated radiotherapy or chemoradiotherapy or moderately hypofractionated (nonstereotactic ablative radiotherapy [SABR]) and hyperfractionated radiotherapy as well as particle beam therapies.
The studies included patients with SCLC or NSCLC and any form of ILD, including subclinical, radiologically diagnosed, or symptomatic, the researchers said.
Overall, the median incidence of grade 3 or higher radiation pneumonitis was 19.7%; the median incidence in patients treated with conventional radical radiotherapy, SABR, and particle beam therapy was 31.8%, 11.9%, and 20.25%, respectively.
Eighteen studies reported grade 5 radiation pneumonitis; the overall median incidence was 6%, but as high as 60% in some studies. When separated by treatment type, the median incidence was 2.7%, 6.25%, and 6.25%, respectively, in patients treated with radical radiotherapy (non-SABR), SABR, and particle beam therapy.
Independent predictors of severe radiation pneumonitis (grade 2 or higher and grade 3 or higher) included subclinical or radiological ILD, the researchers said. Among ILD subtypes, studies have shown increased risk for severe radiation pneumonitis among those with non-IPF or non-UIP pattern fibrosis.
In addition, patient-related factors of low forced vital capacity (FVC) and low forced expiratory volume in 1 second (FEV1), have been associated with severe radiation pneumonitis, the researchers said. They also found increased risk for patients with lower lobe tumor location compared to other lobes.
As for treatment-related factors, a history of gemcitabine chemotherapy was associated with an increased risk of grade 3 or higher radiation pneumonitis.
“There is always concern about using thoracic radiotherapy in lung cancer patients with coexisting ILD in view of the risks involved,” the researchers wrote in their discussion of the findings. “Although thoracic radiotherapy is expected to produce similar local control, overall survival is worse in lung cancer patients with ILD than without, probably due to the poor prognosis associated with ILD and associated treatment-related mortality,” they said.
The findings were limited by several factors including the heterogeneity of the studies and study population and the retrospective design of most of the studies, the researchers noted.
However, the results highlight the increased risk of severe and fatal radiation pneumonitis in lung cancer patients with ILD and the need for careful patient selection and counseling if thoracic radiotherapy is to be considered, they concluded.
The study received no outside funding. The researchers had no financial conflicts to disclose.
FROM CLINICAL ONCOLOGY
Cough and moderate hoarseness
Based on the patient's presentation, history, and imaging results, the likely diagnosis is non–small cell lung cancer (NSCLC) of an adenocarcinoma subtype. NSCLC accounts for about 80% of all lung cancer cases. Adenocarcinoma, in particular, is the most common type of lung cancer in the United States, accounting for about 40% of cases. This subtype is also the most common histology among nonsmokers. Still, individuals aged 55 to 77 years with a smoking history of 30 pack-years or more are considered to be the highest-risk group for lung cancer; those who quit less than 15 years ago — like the patient in the present case — are still considered to be in this risk group. Most cases of lung cancer are diagnosed at a late stage when symptoms have already begun to manifest. However, it should be noted that women are more likely to develop adenocarcinoma, are generally younger when they present with symptoms, and are more likely to present with localized disease. It remains to be proven whether the use of HRT affects the risk for lung cancer in women. Deaths from lung cancer, and in particular NSCLC, were shown to be higher among patients undergoing HRT, though no increase in lung cancer death was reported in women receiving estrogen alone.
In addition to the imaging described in this case, workup for NSCLC should include immunohistochemical (IHC) analyses to identify tumor type and lineage (adenocarcinoma, squamous cell carcinoma, metastatic malignancy, or primary pleural mesothelioma). Separate IHC analyses are then used to guide treatment decisions, identifying whether anaplastic lymphoma kinase inhibitor therapy or programmed death-ligand 1 inhibitor therapy would be appropriate.
Tissue should also be conserved for molecular testing. Management of NSCLC is primarily informed by the presence of targetable mutations. Among adenocarcinoma cases, the most common mutations are in the EGFR and KRAS genes. KRAS mutations, unlike EGFR mutations, are associated with a history of smoking and are considered prognostic biomarkers. Because overlapping targetable alterations are uncommon, patients who are confirmed to be harboring KRAS mutations will likely not benefit from additional molecular testing. Presence of the KRAS mutation suggests a poor response to EGFR tyrosine kinase inhibitors, though it does not appear to impact chemotherapeutic efficacy. Although no targeted therapies are yet available for this population, immune checkpoint inhibitors appear to be beneficial. National Comprehensive Cancer Network guidelines advise that all patients with adenocarcinoma be tested for EGFR mutations and that DNA mutational analysis is the preferred method.
Karl J. D'Silva, MD, Clinical Assistant Professor, Department of Medicine, Tufts University School of Medicine, Boston; Medical Director, Department of Oncology and Hematology, Lahey Hospital and Medical Center, Peabody, Massachusetts.
Karl J. D'Silva, MD, has disclosed no relevant financial relationships.
Based on the patient's presentation, history, and imaging results, the likely diagnosis is non–small cell lung cancer (NSCLC) of an adenocarcinoma subtype. NSCLC accounts for about 80% of all lung cancer cases. Adenocarcinoma, in particular, is the most common type of lung cancer in the United States, accounting for about 40% of cases. This subtype is also the most common histology among nonsmokers. Still, individuals aged 55 to 77 years with a smoking history of 30 pack-years or more are considered to be the highest-risk group for lung cancer; those who quit less than 15 years ago — like the patient in the present case — are still considered to be in this risk group. Most cases of lung cancer are diagnosed at a late stage when symptoms have already begun to manifest. However, it should be noted that women are more likely to develop adenocarcinoma, are generally younger when they present with symptoms, and are more likely to present with localized disease. It remains to be proven whether the use of HRT affects the risk for lung cancer in women. Deaths from lung cancer, and in particular NSCLC, were shown to be higher among patients undergoing HRT, though no increase in lung cancer death was reported in women receiving estrogen alone.
In addition to the imaging described in this case, workup for NSCLC should include immunohistochemical (IHC) analyses to identify tumor type and lineage (adenocarcinoma, squamous cell carcinoma, metastatic malignancy, or primary pleural mesothelioma). Separate IHC analyses are then used to guide treatment decisions, identifying whether anaplastic lymphoma kinase inhibitor therapy or programmed death-ligand 1 inhibitor therapy would be appropriate.
Tissue should also be conserved for molecular testing. Management of NSCLC is primarily informed by the presence of targetable mutations. Among adenocarcinoma cases, the most common mutations are in the EGFR and KRAS genes. KRAS mutations, unlike EGFR mutations, are associated with a history of smoking and are considered prognostic biomarkers. Because overlapping targetable alterations are uncommon, patients who are confirmed to be harboring KRAS mutations will likely not benefit from additional molecular testing. Presence of the KRAS mutation suggests a poor response to EGFR tyrosine kinase inhibitors, though it does not appear to impact chemotherapeutic efficacy. Although no targeted therapies are yet available for this population, immune checkpoint inhibitors appear to be beneficial. National Comprehensive Cancer Network guidelines advise that all patients with adenocarcinoma be tested for EGFR mutations and that DNA mutational analysis is the preferred method.
Karl J. D'Silva, MD, Clinical Assistant Professor, Department of Medicine, Tufts University School of Medicine, Boston; Medical Director, Department of Oncology and Hematology, Lahey Hospital and Medical Center, Peabody, Massachusetts.
Karl J. D'Silva, MD, has disclosed no relevant financial relationships.
Based on the patient's presentation, history, and imaging results, the likely diagnosis is non–small cell lung cancer (NSCLC) of an adenocarcinoma subtype. NSCLC accounts for about 80% of all lung cancer cases. Adenocarcinoma, in particular, is the most common type of lung cancer in the United States, accounting for about 40% of cases. This subtype is also the most common histology among nonsmokers. Still, individuals aged 55 to 77 years with a smoking history of 30 pack-years or more are considered to be the highest-risk group for lung cancer; those who quit less than 15 years ago — like the patient in the present case — are still considered to be in this risk group. Most cases of lung cancer are diagnosed at a late stage when symptoms have already begun to manifest. However, it should be noted that women are more likely to develop adenocarcinoma, are generally younger when they present with symptoms, and are more likely to present with localized disease. It remains to be proven whether the use of HRT affects the risk for lung cancer in women. Deaths from lung cancer, and in particular NSCLC, were shown to be higher among patients undergoing HRT, though no increase in lung cancer death was reported in women receiving estrogen alone.
In addition to the imaging described in this case, workup for NSCLC should include immunohistochemical (IHC) analyses to identify tumor type and lineage (adenocarcinoma, squamous cell carcinoma, metastatic malignancy, or primary pleural mesothelioma). Separate IHC analyses are then used to guide treatment decisions, identifying whether anaplastic lymphoma kinase inhibitor therapy or programmed death-ligand 1 inhibitor therapy would be appropriate.
Tissue should also be conserved for molecular testing. Management of NSCLC is primarily informed by the presence of targetable mutations. Among adenocarcinoma cases, the most common mutations are in the EGFR and KRAS genes. KRAS mutations, unlike EGFR mutations, are associated with a history of smoking and are considered prognostic biomarkers. Because overlapping targetable alterations are uncommon, patients who are confirmed to be harboring KRAS mutations will likely not benefit from additional molecular testing. Presence of the KRAS mutation suggests a poor response to EGFR tyrosine kinase inhibitors, though it does not appear to impact chemotherapeutic efficacy. Although no targeted therapies are yet available for this population, immune checkpoint inhibitors appear to be beneficial. National Comprehensive Cancer Network guidelines advise that all patients with adenocarcinoma be tested for EGFR mutations and that DNA mutational analysis is the preferred method.
Karl J. D'Silva, MD, Clinical Assistant Professor, Department of Medicine, Tufts University School of Medicine, Boston; Medical Director, Department of Oncology and Hematology, Lahey Hospital and Medical Center, Peabody, Massachusetts.
Karl J. D'Silva, MD, has disclosed no relevant financial relationships.
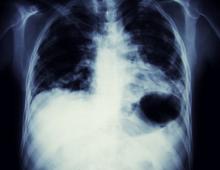
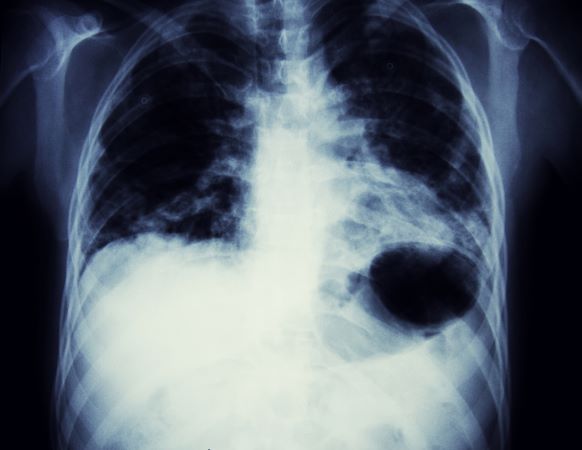
A 56-year-old woman presents with dyspnea, a persistent cough, and moderate hoarseness. She has no significant medical history other than thyroiditis. Her current medications include hormone replacement therapy (HRT). Although the patient reports a 20–pack-year history of smoking tobacco, she notes that she quit 11 years ago and has not been previously screened for lung cancer. A chest radiograph is ordered, which demonstrates a mass in the upper lobe of the right lung.
Filling opioid prescriptions akin to a Sisyphean task
Pain management is a huge part of how we in palliative care help patients – and most of the time, I think we do it well, but in the regulatory environment of the opioid epidemic,
A patient – let’s call her Joan – calls me in distress. She is a 62-year-old woman with widespread metastatic breast cancer. Her pain is mainly due to bone metastases, but she also has discomfort due to the cancer’s invasion of the thin membranes that line her lungs and abdomen.
She was started on a combination opioid and acetaminophen tablet about 2 months ago by her oncologist, but is now requiring it around the clock, nearing the ceiling dose for this particular medication.
Given that her pain is escalating, Joan and I discuss starting a long-acting opioid to better manage the peak and trough effect of short-acting opioids, which can make a patient feel that the pain is relieved only for a few hours at a time, with sharp spikes throughout the day that mandate the next dose of short-acting opioid. This tethers the patient to the clock, having to take as many as six or eight doses of medication per day, and can be very disruptive to daily life.
I send an e-prescription for the same opioid Joan’s currently taking, but in a long-acting format that will slow-release over 8-10 hours, relieving her of the need to take a medication every 3-4 hours. I have learned over the years that nearly every long-acting opioid automatically generates a prior authorization request from the patient’s insurance company and so I immediately email our prior authorization team to submit to Joan’s insurance right away to avoid this extra delay.
Our prior authorization team is exceptionally responsive and submits these requests with urgency every time – they understand that cancer pain is a serious problem and we can’t wait 5 business days for answers. They are typically able to obtain an approved prior authorization for nearly every long-acting opioid I write within 24-48 hours.
But here’s where things go sideways.
First, the insurance company denies the prior authorization request, demanding that I revise the prescription from the long-acting version of the opioid she is currently taking to a cheaper, older opioid that she’s never tried before. In other words, they won’t cover the drug I requested without Joan first trying a completely different drug and failing it. This only makes sense for the insurance company’s bottom line – it makes no clinical sense at all. Why would I try a novel compound that Joan’s never had and one to which I have no idea how she’ll respond when I could keep her on the same compound knowing that she tolerates it just fine?
Past experience tells me insurance companies rarely budge on this, and appealing the decision would just introduce even more delay of care, so I begrudgingly change the prescription and send it again to the pharmacy. I message Joan to let her know that her insurance won’t cover my drug of choice and that we have to try this older one first.
A few hours later, Joan sends me a message: “My pharmacy says it’s going to take A WEEK to get the long-acting medicine!”
In the meantime, Joan has been using her short-acting opioid faster than anticipated because of her escalating pain – so she’s now running low on that as well.
I write for more of her short-acting opioid and e-script it to her pharmacy.
Within a few hours, we get another automatic response from her insurance that we’re going to need a prior authorization for additional short-acting opioid because she’s exceeded “quantity limitations,” which as far as I can tell is a completely arbitrary number not based on clinical evidence.
The prior auth team jumps on it and submits to override the quantity limit – successfully – and sends the override code to her pharmacy to reprocess the prescription.
But now the pharmacist tells Joan that they won’t fill the Rx anyway because it’s “too early.” They tell her that “state laws” prevent them from filling the scrip.
Is this true? I have no idea. I’m not an expert on California pharmacy law. All I know is that my patient is in pain and something needs to happen quickly.
I write for a second short-acting opioid – again a completely different compound. Ironically, this Rx goes through instantly without need for prior authorization. But now Joan has to switch to another new drug for no good medical reason.
If you’re still with me this far into the weeds, I’m grateful. In all it took a combined 4 hours of work (between myself and the prior auth team) to get two opioid Rx’s filled – and these were completely different medications than the ones I originally wrote for. I also had to move her prescriptions to the hospital’s pharmacy (another inconvenience for Joan and her family) so that she could get the medications in a timely manner. All this work to ensure that a single patient had adequate and timely pain relief and to prevent her from having to make an unnecessary visit to the emergency department for pain crisis.
This is just a regular day in outpatient palliative care in the era of the opioid epidemic.
The epidemic has caused tremendous pain and suffering for millions of people over the past 2 decades – namely those lost to opioid overdoses and their loved ones. And for the most part, tightening access to opioids for routine aches and pains among a relatively healthy population is not wrong, in my opinion, as long as those restrictions are based in good faith on robust evidence.
But the hidden cost of the Centers for Disease Control and Prevention’s 2016 opioid prescribing guidelines for nonmalignant pain, as well as the flurry of restrictive state laws they generated, is felt every day by patients with serious illness even though the guidelines were never meant to affect them. Patients with active cancer, receiving palliative care services, or at the end of life, were supposed to be exempted from these guidelines since good evidence supports the use of opioids in these populations.
Instead of preserving access to desperately needed pain medicine for those suffering with serious illness, states and insurers have aggressively sought to gatekeep opioids from everyone, resulting in stigma, delays, and needless suffering.
Several recent studies have revealed the effects of this gatekeeping on patients with cancer.
A qualitative study with 26 advanced cancer patients described the demoralization and stigma many patients felt when taking opioids, which they directly tied to media messaging around the opioid epidemic. Even when they reluctantly agreed to take opioids to treat cancer-related pain, there were systemic impediments to achieving adequate pain relief – similar to my experience with Joan – that were directly caused by insurance and pharmacy constraints.
Those of us who care for oncology patients also appear to be undertreating cancer-related pain. Another recent study that found the amount of opioid medications prescribed to an advanced cancer patient near the end of life dropped by 38% between 2007 and 2017. The authors suggest that a direct consequence of this decline in appropriate opioid prescribing is an observed 50% rise in emergency department visits over the same time period by cancer patients for pain-related reasons.
This makes sense – if patients aren’t routinely prescribed the opioids they need to manage their cancer-related pain; or, if the stigma against using opioids is so harsh that it causes patients to shun opioids; or, if there are so many system barriers in place to prevent patients from obtaining opioids in a timely manner – then patients’ pain will crescendo, leaving them with little alternative but to head to the emergency department.
This undertreatment is corroborated by another study that examined data from the Centers for Medicare & Medicaid Services Part D prescriber database between 2013 and 2017, finding that both oncologists and nononcologists prescribed about 21% fewer opioids to Medicare beneficiaries during that time, compared with the period prior to 2013.
Interestingly, the researchers also found that opioid prescribing by palliative care providers increased by 15% over the same period. On a positive note, this suggests the presence of a growing outpatient palliative care workforce. But it may also reflect growing unease among oncologists with the perceived liability for prescribing opioids and a desire to ask other specialists to take on this liability. At the same time, it may reflect the very real and ever-increasing administrative burden associated with prescribing opioids and the fact that busy oncologists may not have time to spend on this aspect of cancer care. Thus, as palliative care clinicians become more visible and numerous in the outpatient arena, oncologists may increasingly ask palliative care clinicians like myself to take this on.
The problem with this is that merely handing off the administrative burden to another clinician doesn’t address the underlying problem. Anecdotal evidence suggests (and my own experiences corroborate) this administrative burden can cause real harm. A survey of 1,000 physicians conducted by the American Medical Association in 2021 found that 93% of respondents reported a delay in patient care due to prior authorization burden and 34% of respondents reported that their patients had suffered a “serious adverse event” due to prior authorization requirements.
The CDC recently announced it will take steps to revise the 2016 opioid prescribing guidelines for chronic pain after hearing from members of the medical community as well as patients living with chronic pain about the harsh, unintended consequences of the guidelines. I can only hope that insurance companies will follow suit, revising their opioid prior authorization requirements to finally come into alignment with the rational, safe use of opioids in patients with advanced cancer. It’s too bad that any improvement in the future will be too late for the millions of patients who have suffered irreversible iatrogenic harms due to delays in achieving adequate pain relief.
Sarah F. D’Ambruoso, NP, is a palliative care nurse practitioner in Santa Monica, Calif.
Pain management is a huge part of how we in palliative care help patients – and most of the time, I think we do it well, but in the regulatory environment of the opioid epidemic,
A patient – let’s call her Joan – calls me in distress. She is a 62-year-old woman with widespread metastatic breast cancer. Her pain is mainly due to bone metastases, but she also has discomfort due to the cancer’s invasion of the thin membranes that line her lungs and abdomen.
She was started on a combination opioid and acetaminophen tablet about 2 months ago by her oncologist, but is now requiring it around the clock, nearing the ceiling dose for this particular medication.
Given that her pain is escalating, Joan and I discuss starting a long-acting opioid to better manage the peak and trough effect of short-acting opioids, which can make a patient feel that the pain is relieved only for a few hours at a time, with sharp spikes throughout the day that mandate the next dose of short-acting opioid. This tethers the patient to the clock, having to take as many as six or eight doses of medication per day, and can be very disruptive to daily life.
I send an e-prescription for the same opioid Joan’s currently taking, but in a long-acting format that will slow-release over 8-10 hours, relieving her of the need to take a medication every 3-4 hours. I have learned over the years that nearly every long-acting opioid automatically generates a prior authorization request from the patient’s insurance company and so I immediately email our prior authorization team to submit to Joan’s insurance right away to avoid this extra delay.
Our prior authorization team is exceptionally responsive and submits these requests with urgency every time – they understand that cancer pain is a serious problem and we can’t wait 5 business days for answers. They are typically able to obtain an approved prior authorization for nearly every long-acting opioid I write within 24-48 hours.
But here’s where things go sideways.
First, the insurance company denies the prior authorization request, demanding that I revise the prescription from the long-acting version of the opioid she is currently taking to a cheaper, older opioid that she’s never tried before. In other words, they won’t cover the drug I requested without Joan first trying a completely different drug and failing it. This only makes sense for the insurance company’s bottom line – it makes no clinical sense at all. Why would I try a novel compound that Joan’s never had and one to which I have no idea how she’ll respond when I could keep her on the same compound knowing that she tolerates it just fine?
Past experience tells me insurance companies rarely budge on this, and appealing the decision would just introduce even more delay of care, so I begrudgingly change the prescription and send it again to the pharmacy. I message Joan to let her know that her insurance won’t cover my drug of choice and that we have to try this older one first.
A few hours later, Joan sends me a message: “My pharmacy says it’s going to take A WEEK to get the long-acting medicine!”
In the meantime, Joan has been using her short-acting opioid faster than anticipated because of her escalating pain – so she’s now running low on that as well.
I write for more of her short-acting opioid and e-script it to her pharmacy.
Within a few hours, we get another automatic response from her insurance that we’re going to need a prior authorization for additional short-acting opioid because she’s exceeded “quantity limitations,” which as far as I can tell is a completely arbitrary number not based on clinical evidence.
The prior auth team jumps on it and submits to override the quantity limit – successfully – and sends the override code to her pharmacy to reprocess the prescription.
But now the pharmacist tells Joan that they won’t fill the Rx anyway because it’s “too early.” They tell her that “state laws” prevent them from filling the scrip.
Is this true? I have no idea. I’m not an expert on California pharmacy law. All I know is that my patient is in pain and something needs to happen quickly.
I write for a second short-acting opioid – again a completely different compound. Ironically, this Rx goes through instantly without need for prior authorization. But now Joan has to switch to another new drug for no good medical reason.
If you’re still with me this far into the weeds, I’m grateful. In all it took a combined 4 hours of work (between myself and the prior auth team) to get two opioid Rx’s filled – and these were completely different medications than the ones I originally wrote for. I also had to move her prescriptions to the hospital’s pharmacy (another inconvenience for Joan and her family) so that she could get the medications in a timely manner. All this work to ensure that a single patient had adequate and timely pain relief and to prevent her from having to make an unnecessary visit to the emergency department for pain crisis.
This is just a regular day in outpatient palliative care in the era of the opioid epidemic.
The epidemic has caused tremendous pain and suffering for millions of people over the past 2 decades – namely those lost to opioid overdoses and their loved ones. And for the most part, tightening access to opioids for routine aches and pains among a relatively healthy population is not wrong, in my opinion, as long as those restrictions are based in good faith on robust evidence.
But the hidden cost of the Centers for Disease Control and Prevention’s 2016 opioid prescribing guidelines for nonmalignant pain, as well as the flurry of restrictive state laws they generated, is felt every day by patients with serious illness even though the guidelines were never meant to affect them. Patients with active cancer, receiving palliative care services, or at the end of life, were supposed to be exempted from these guidelines since good evidence supports the use of opioids in these populations.
Instead of preserving access to desperately needed pain medicine for those suffering with serious illness, states and insurers have aggressively sought to gatekeep opioids from everyone, resulting in stigma, delays, and needless suffering.
Several recent studies have revealed the effects of this gatekeeping on patients with cancer.
A qualitative study with 26 advanced cancer patients described the demoralization and stigma many patients felt when taking opioids, which they directly tied to media messaging around the opioid epidemic. Even when they reluctantly agreed to take opioids to treat cancer-related pain, there were systemic impediments to achieving adequate pain relief – similar to my experience with Joan – that were directly caused by insurance and pharmacy constraints.
Those of us who care for oncology patients also appear to be undertreating cancer-related pain. Another recent study that found the amount of opioid medications prescribed to an advanced cancer patient near the end of life dropped by 38% between 2007 and 2017. The authors suggest that a direct consequence of this decline in appropriate opioid prescribing is an observed 50% rise in emergency department visits over the same time period by cancer patients for pain-related reasons.
This makes sense – if patients aren’t routinely prescribed the opioids they need to manage their cancer-related pain; or, if the stigma against using opioids is so harsh that it causes patients to shun opioids; or, if there are so many system barriers in place to prevent patients from obtaining opioids in a timely manner – then patients’ pain will crescendo, leaving them with little alternative but to head to the emergency department.
This undertreatment is corroborated by another study that examined data from the Centers for Medicare & Medicaid Services Part D prescriber database between 2013 and 2017, finding that both oncologists and nononcologists prescribed about 21% fewer opioids to Medicare beneficiaries during that time, compared with the period prior to 2013.
Interestingly, the researchers also found that opioid prescribing by palliative care providers increased by 15% over the same period. On a positive note, this suggests the presence of a growing outpatient palliative care workforce. But it may also reflect growing unease among oncologists with the perceived liability for prescribing opioids and a desire to ask other specialists to take on this liability. At the same time, it may reflect the very real and ever-increasing administrative burden associated with prescribing opioids and the fact that busy oncologists may not have time to spend on this aspect of cancer care. Thus, as palliative care clinicians become more visible and numerous in the outpatient arena, oncologists may increasingly ask palliative care clinicians like myself to take this on.
The problem with this is that merely handing off the administrative burden to another clinician doesn’t address the underlying problem. Anecdotal evidence suggests (and my own experiences corroborate) this administrative burden can cause real harm. A survey of 1,000 physicians conducted by the American Medical Association in 2021 found that 93% of respondents reported a delay in patient care due to prior authorization burden and 34% of respondents reported that their patients had suffered a “serious adverse event” due to prior authorization requirements.
The CDC recently announced it will take steps to revise the 2016 opioid prescribing guidelines for chronic pain after hearing from members of the medical community as well as patients living with chronic pain about the harsh, unintended consequences of the guidelines. I can only hope that insurance companies will follow suit, revising their opioid prior authorization requirements to finally come into alignment with the rational, safe use of opioids in patients with advanced cancer. It’s too bad that any improvement in the future will be too late for the millions of patients who have suffered irreversible iatrogenic harms due to delays in achieving adequate pain relief.
Sarah F. D’Ambruoso, NP, is a palliative care nurse practitioner in Santa Monica, Calif.
Pain management is a huge part of how we in palliative care help patients – and most of the time, I think we do it well, but in the regulatory environment of the opioid epidemic,
A patient – let’s call her Joan – calls me in distress. She is a 62-year-old woman with widespread metastatic breast cancer. Her pain is mainly due to bone metastases, but she also has discomfort due to the cancer’s invasion of the thin membranes that line her lungs and abdomen.
She was started on a combination opioid and acetaminophen tablet about 2 months ago by her oncologist, but is now requiring it around the clock, nearing the ceiling dose for this particular medication.
Given that her pain is escalating, Joan and I discuss starting a long-acting opioid to better manage the peak and trough effect of short-acting opioids, which can make a patient feel that the pain is relieved only for a few hours at a time, with sharp spikes throughout the day that mandate the next dose of short-acting opioid. This tethers the patient to the clock, having to take as many as six or eight doses of medication per day, and can be very disruptive to daily life.
I send an e-prescription for the same opioid Joan’s currently taking, but in a long-acting format that will slow-release over 8-10 hours, relieving her of the need to take a medication every 3-4 hours. I have learned over the years that nearly every long-acting opioid automatically generates a prior authorization request from the patient’s insurance company and so I immediately email our prior authorization team to submit to Joan’s insurance right away to avoid this extra delay.
Our prior authorization team is exceptionally responsive and submits these requests with urgency every time – they understand that cancer pain is a serious problem and we can’t wait 5 business days for answers. They are typically able to obtain an approved prior authorization for nearly every long-acting opioid I write within 24-48 hours.
But here’s where things go sideways.
First, the insurance company denies the prior authorization request, demanding that I revise the prescription from the long-acting version of the opioid she is currently taking to a cheaper, older opioid that she’s never tried before. In other words, they won’t cover the drug I requested without Joan first trying a completely different drug and failing it. This only makes sense for the insurance company’s bottom line – it makes no clinical sense at all. Why would I try a novel compound that Joan’s never had and one to which I have no idea how she’ll respond when I could keep her on the same compound knowing that she tolerates it just fine?
Past experience tells me insurance companies rarely budge on this, and appealing the decision would just introduce even more delay of care, so I begrudgingly change the prescription and send it again to the pharmacy. I message Joan to let her know that her insurance won’t cover my drug of choice and that we have to try this older one first.
A few hours later, Joan sends me a message: “My pharmacy says it’s going to take A WEEK to get the long-acting medicine!”
In the meantime, Joan has been using her short-acting opioid faster than anticipated because of her escalating pain – so she’s now running low on that as well.
I write for more of her short-acting opioid and e-script it to her pharmacy.
Within a few hours, we get another automatic response from her insurance that we’re going to need a prior authorization for additional short-acting opioid because she’s exceeded “quantity limitations,” which as far as I can tell is a completely arbitrary number not based on clinical evidence.
The prior auth team jumps on it and submits to override the quantity limit – successfully – and sends the override code to her pharmacy to reprocess the prescription.
But now the pharmacist tells Joan that they won’t fill the Rx anyway because it’s “too early.” They tell her that “state laws” prevent them from filling the scrip.
Is this true? I have no idea. I’m not an expert on California pharmacy law. All I know is that my patient is in pain and something needs to happen quickly.
I write for a second short-acting opioid – again a completely different compound. Ironically, this Rx goes through instantly without need for prior authorization. But now Joan has to switch to another new drug for no good medical reason.
If you’re still with me this far into the weeds, I’m grateful. In all it took a combined 4 hours of work (between myself and the prior auth team) to get two opioid Rx’s filled – and these were completely different medications than the ones I originally wrote for. I also had to move her prescriptions to the hospital’s pharmacy (another inconvenience for Joan and her family) so that she could get the medications in a timely manner. All this work to ensure that a single patient had adequate and timely pain relief and to prevent her from having to make an unnecessary visit to the emergency department for pain crisis.
This is just a regular day in outpatient palliative care in the era of the opioid epidemic.
The epidemic has caused tremendous pain and suffering for millions of people over the past 2 decades – namely those lost to opioid overdoses and their loved ones. And for the most part, tightening access to opioids for routine aches and pains among a relatively healthy population is not wrong, in my opinion, as long as those restrictions are based in good faith on robust evidence.
But the hidden cost of the Centers for Disease Control and Prevention’s 2016 opioid prescribing guidelines for nonmalignant pain, as well as the flurry of restrictive state laws they generated, is felt every day by patients with serious illness even though the guidelines were never meant to affect them. Patients with active cancer, receiving palliative care services, or at the end of life, were supposed to be exempted from these guidelines since good evidence supports the use of opioids in these populations.
Instead of preserving access to desperately needed pain medicine for those suffering with serious illness, states and insurers have aggressively sought to gatekeep opioids from everyone, resulting in stigma, delays, and needless suffering.
Several recent studies have revealed the effects of this gatekeeping on patients with cancer.
A qualitative study with 26 advanced cancer patients described the demoralization and stigma many patients felt when taking opioids, which they directly tied to media messaging around the opioid epidemic. Even when they reluctantly agreed to take opioids to treat cancer-related pain, there were systemic impediments to achieving adequate pain relief – similar to my experience with Joan – that were directly caused by insurance and pharmacy constraints.
Those of us who care for oncology patients also appear to be undertreating cancer-related pain. Another recent study that found the amount of opioid medications prescribed to an advanced cancer patient near the end of life dropped by 38% between 2007 and 2017. The authors suggest that a direct consequence of this decline in appropriate opioid prescribing is an observed 50% rise in emergency department visits over the same time period by cancer patients for pain-related reasons.
This makes sense – if patients aren’t routinely prescribed the opioids they need to manage their cancer-related pain; or, if the stigma against using opioids is so harsh that it causes patients to shun opioids; or, if there are so many system barriers in place to prevent patients from obtaining opioids in a timely manner – then patients’ pain will crescendo, leaving them with little alternative but to head to the emergency department.
This undertreatment is corroborated by another study that examined data from the Centers for Medicare & Medicaid Services Part D prescriber database between 2013 and 2017, finding that both oncologists and nononcologists prescribed about 21% fewer opioids to Medicare beneficiaries during that time, compared with the period prior to 2013.
Interestingly, the researchers also found that opioid prescribing by palliative care providers increased by 15% over the same period. On a positive note, this suggests the presence of a growing outpatient palliative care workforce. But it may also reflect growing unease among oncologists with the perceived liability for prescribing opioids and a desire to ask other specialists to take on this liability. At the same time, it may reflect the very real and ever-increasing administrative burden associated with prescribing opioids and the fact that busy oncologists may not have time to spend on this aspect of cancer care. Thus, as palliative care clinicians become more visible and numerous in the outpatient arena, oncologists may increasingly ask palliative care clinicians like myself to take this on.
The problem with this is that merely handing off the administrative burden to another clinician doesn’t address the underlying problem. Anecdotal evidence suggests (and my own experiences corroborate) this administrative burden can cause real harm. A survey of 1,000 physicians conducted by the American Medical Association in 2021 found that 93% of respondents reported a delay in patient care due to prior authorization burden and 34% of respondents reported that their patients had suffered a “serious adverse event” due to prior authorization requirements.
The CDC recently announced it will take steps to revise the 2016 opioid prescribing guidelines for chronic pain after hearing from members of the medical community as well as patients living with chronic pain about the harsh, unintended consequences of the guidelines. I can only hope that insurance companies will follow suit, revising their opioid prior authorization requirements to finally come into alignment with the rational, safe use of opioids in patients with advanced cancer. It’s too bad that any improvement in the future will be too late for the millions of patients who have suffered irreversible iatrogenic harms due to delays in achieving adequate pain relief.
Sarah F. D’Ambruoso, NP, is a palliative care nurse practitioner in Santa Monica, Calif.
FDA approves neoadjuvant nivolumab/chemo for early-stage NSCLC
in combination with platinum-doublet chemotherapy, regardless of PDL-1 status.
Nivolumab is the first immune checkpoint inhibitor to be approved for resectable NSCLC; its three prior NSCLC indications are for metastatic disease, the agency said in its announcement.
Approval was based on the CheckMate 816 trial, which randomized 358 patients evenly to either nivolumab plus platinum doublets or to platinum doublets alone every 3 weeks for up to 3 cycles.
Trial participants had histologically confirmed stage IB, II, or IIIA disease, which was measurable by RECIST criteria. They were enrolled regardless of tumor PD-L1 status.
At surgery, the pathologic complete response rate was 24% in the nivolumab arm versus 2.2% in the chemotherapy-alone group.
Median event-free survival was 31.6 months with nivolumab but 20.8 months without it, which translated to a 37% reduction in the risk for progression, recurrence, or death following surgery. A trend toward better overall survival was not statistically significant, Bristol Myers Squibb said in its own announcement.
Nivolumab’s new neoadjuvant indication is for adult patients with resectable NSCLC (tumors greater than or equal to 4 cm or node positive). The recommended dosage is 360 mg in combination with platinum-doublet chemotherapy on the same day every 3 weeks for three cycles.
In a press release from Bristol Myers Squibb, CheckMate 816 investigator and Dana-Farber Cancer Institute thoracic oncologist Mark Awad, MD, PhD, called the approval “a turning point in how we treat resectable NSCLC.”
Patients with known EGFR mutations or ALK translocations, grade 2 or higher peripheral neuropathy, active autoimmune disease, or medical conditions requiring systemic immunosuppression were excluded.
There were no fatal adverse events in the nivolumab arm, but 30% of participants had serious adverse events, most commonly pneumonia and vomiting.
The most common side effects across all grades were nausea (38%), constipation (34%), fatigue (26%), decreased appetite (20%), and rash (20%). Surgical complications and hospital lengths were similar between the two study groups.
Rival checkpoint inhibitor pembrolizumab is also being investigated for neoadjuvant NSCLC.
A version of this article first appeared on Medscape.com.
in combination with platinum-doublet chemotherapy, regardless of PDL-1 status.
Nivolumab is the first immune checkpoint inhibitor to be approved for resectable NSCLC; its three prior NSCLC indications are for metastatic disease, the agency said in its announcement.
Approval was based on the CheckMate 816 trial, which randomized 358 patients evenly to either nivolumab plus platinum doublets or to platinum doublets alone every 3 weeks for up to 3 cycles.
Trial participants had histologically confirmed stage IB, II, or IIIA disease, which was measurable by RECIST criteria. They were enrolled regardless of tumor PD-L1 status.
At surgery, the pathologic complete response rate was 24% in the nivolumab arm versus 2.2% in the chemotherapy-alone group.
Median event-free survival was 31.6 months with nivolumab but 20.8 months without it, which translated to a 37% reduction in the risk for progression, recurrence, or death following surgery. A trend toward better overall survival was not statistically significant, Bristol Myers Squibb said in its own announcement.
Nivolumab’s new neoadjuvant indication is for adult patients with resectable NSCLC (tumors greater than or equal to 4 cm or node positive). The recommended dosage is 360 mg in combination with platinum-doublet chemotherapy on the same day every 3 weeks for three cycles.
In a press release from Bristol Myers Squibb, CheckMate 816 investigator and Dana-Farber Cancer Institute thoracic oncologist Mark Awad, MD, PhD, called the approval “a turning point in how we treat resectable NSCLC.”
Patients with known EGFR mutations or ALK translocations, grade 2 or higher peripheral neuropathy, active autoimmune disease, or medical conditions requiring systemic immunosuppression were excluded.
There were no fatal adverse events in the nivolumab arm, but 30% of participants had serious adverse events, most commonly pneumonia and vomiting.
The most common side effects across all grades were nausea (38%), constipation (34%), fatigue (26%), decreased appetite (20%), and rash (20%). Surgical complications and hospital lengths were similar between the two study groups.
Rival checkpoint inhibitor pembrolizumab is also being investigated for neoadjuvant NSCLC.
A version of this article first appeared on Medscape.com.
in combination with platinum-doublet chemotherapy, regardless of PDL-1 status.
Nivolumab is the first immune checkpoint inhibitor to be approved for resectable NSCLC; its three prior NSCLC indications are for metastatic disease, the agency said in its announcement.
Approval was based on the CheckMate 816 trial, which randomized 358 patients evenly to either nivolumab plus platinum doublets or to platinum doublets alone every 3 weeks for up to 3 cycles.
Trial participants had histologically confirmed stage IB, II, or IIIA disease, which was measurable by RECIST criteria. They were enrolled regardless of tumor PD-L1 status.
At surgery, the pathologic complete response rate was 24% in the nivolumab arm versus 2.2% in the chemotherapy-alone group.
Median event-free survival was 31.6 months with nivolumab but 20.8 months without it, which translated to a 37% reduction in the risk for progression, recurrence, or death following surgery. A trend toward better overall survival was not statistically significant, Bristol Myers Squibb said in its own announcement.
Nivolumab’s new neoadjuvant indication is for adult patients with resectable NSCLC (tumors greater than or equal to 4 cm or node positive). The recommended dosage is 360 mg in combination with platinum-doublet chemotherapy on the same day every 3 weeks for three cycles.
In a press release from Bristol Myers Squibb, CheckMate 816 investigator and Dana-Farber Cancer Institute thoracic oncologist Mark Awad, MD, PhD, called the approval “a turning point in how we treat resectable NSCLC.”
Patients with known EGFR mutations or ALK translocations, grade 2 or higher peripheral neuropathy, active autoimmune disease, or medical conditions requiring systemic immunosuppression were excluded.
There were no fatal adverse events in the nivolumab arm, but 30% of participants had serious adverse events, most commonly pneumonia and vomiting.
The most common side effects across all grades were nausea (38%), constipation (34%), fatigue (26%), decreased appetite (20%), and rash (20%). Surgical complications and hospital lengths were similar between the two study groups.
Rival checkpoint inhibitor pembrolizumab is also being investigated for neoadjuvant NSCLC.
A version of this article first appeared on Medscape.com.
Lung cancer now a growing public health threat
, according to estimates of lung cancer incident cases, deaths, and their age-standardized rates.
The findings, based on recently released data from GLOBOCAN 2020 projected to the year 2050, suggest that the lung cancer epidemic will continue to unfold, according to Rajesh Sharma, PhD, et al., in a study published in the International Journal of Clinical Oncology. GLOBOCAN 2020 is an online database produced by the International Agency for Research on Cancer. It provides global cancer statistics from 185 countries for 36 cancer types.
The increase in lung cancer, the leading cancer worldwide in terms of deaths, is generally attributed to increases in cigarette smoking, Sharma et al. wrote. They point out that, while cigarette smoking is expected to have peaked in industrialized countries in the latter half of the twentieth century, the tobacco smoking epidemic is unfolding in regions of Asia and Africa with concomitant increases in lung cancer burden in several countries. Smoking is the most significant lung cancer risk factor, followed by air pollution (especially particulate matter, passive smoking, and occupational exposure to radon and asbestos).
The authors investigated bivariate associations between smoking prevalence and age-standardized rates of lung cancer, and projected lung cancer incident cases and deaths to 2050. They also looked at mortality-to-incidence, considered to be a proxy indicator of 5-year survival, and at human development index, a measure including life expectancy at birth, years of schooling, and standard of living. The results, they state, are expected to aid in policy formulation to combat the lung cancer burden at global, regional, and national levels.
Tobacco smoking prevalence was 21.9% worldwide in 2016, with tobacco smoking prevalence exceeding 25% in 57/149 countries. It was high in European countries with 5 of the top-10 countries among the 149 countries within Europe. Prevalence was greater than 10% in all European countries. Notably, 11/33 countries in Africa had a smoking prevalence less than 10%.
Analysis showed 2.21 million new lung cancer cases and 1.8 million deaths attributed to lung cancer worldwide in 2020, with males accounting for about two-thirds of the burden. The analysis projection for 2050 was for 3.8 million incident cases of lung cancer and 3.2 million lung cancer deaths globally. In 2050, lung cancer cases and deaths are projected to be more than 100,000 in 10/21 regions, led by Eastern Asia, projected to record 1.7 million incident cases and 1.5 million deaths.
The burden of lung cancer in regions of Asia and Africa is expected to increase at least twofold from 2020 to 2050, surpassing European regions that are expected to have the smallest increases. Also, while incident cases will remain much higher in Northern America than in Southeastern Asia and South-Central Asia, the number of lives lost is projected to be similar. The age-specific incidence and death rates rose with age such that the oldest age groups had the highest age-specific rates. With the human development index, mortality-to-incidence showed a negative correlation.
The authors wrote that worsening smoking and pollution levels in developing countries may push the future lung cancer burden much higher than these projections. Unless reversed, cases and death will grow unabated.
“Countering the burden of lung cancer also requires curtailment of other risk factors such as air pollution and exposure to carcinogens,” the authors wrote.
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors. The authors stated that they have no conflicts of interest.
, according to estimates of lung cancer incident cases, deaths, and their age-standardized rates.
The findings, based on recently released data from GLOBOCAN 2020 projected to the year 2050, suggest that the lung cancer epidemic will continue to unfold, according to Rajesh Sharma, PhD, et al., in a study published in the International Journal of Clinical Oncology. GLOBOCAN 2020 is an online database produced by the International Agency for Research on Cancer. It provides global cancer statistics from 185 countries for 36 cancer types.
The increase in lung cancer, the leading cancer worldwide in terms of deaths, is generally attributed to increases in cigarette smoking, Sharma et al. wrote. They point out that, while cigarette smoking is expected to have peaked in industrialized countries in the latter half of the twentieth century, the tobacco smoking epidemic is unfolding in regions of Asia and Africa with concomitant increases in lung cancer burden in several countries. Smoking is the most significant lung cancer risk factor, followed by air pollution (especially particulate matter, passive smoking, and occupational exposure to radon and asbestos).
The authors investigated bivariate associations between smoking prevalence and age-standardized rates of lung cancer, and projected lung cancer incident cases and deaths to 2050. They also looked at mortality-to-incidence, considered to be a proxy indicator of 5-year survival, and at human development index, a measure including life expectancy at birth, years of schooling, and standard of living. The results, they state, are expected to aid in policy formulation to combat the lung cancer burden at global, regional, and national levels.
Tobacco smoking prevalence was 21.9% worldwide in 2016, with tobacco smoking prevalence exceeding 25% in 57/149 countries. It was high in European countries with 5 of the top-10 countries among the 149 countries within Europe. Prevalence was greater than 10% in all European countries. Notably, 11/33 countries in Africa had a smoking prevalence less than 10%.
Analysis showed 2.21 million new lung cancer cases and 1.8 million deaths attributed to lung cancer worldwide in 2020, with males accounting for about two-thirds of the burden. The analysis projection for 2050 was for 3.8 million incident cases of lung cancer and 3.2 million lung cancer deaths globally. In 2050, lung cancer cases and deaths are projected to be more than 100,000 in 10/21 regions, led by Eastern Asia, projected to record 1.7 million incident cases and 1.5 million deaths.
The burden of lung cancer in regions of Asia and Africa is expected to increase at least twofold from 2020 to 2050, surpassing European regions that are expected to have the smallest increases. Also, while incident cases will remain much higher in Northern America than in Southeastern Asia and South-Central Asia, the number of lives lost is projected to be similar. The age-specific incidence and death rates rose with age such that the oldest age groups had the highest age-specific rates. With the human development index, mortality-to-incidence showed a negative correlation.
The authors wrote that worsening smoking and pollution levels in developing countries may push the future lung cancer burden much higher than these projections. Unless reversed, cases and death will grow unabated.
“Countering the burden of lung cancer also requires curtailment of other risk factors such as air pollution and exposure to carcinogens,” the authors wrote.
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors. The authors stated that they have no conflicts of interest.
, according to estimates of lung cancer incident cases, deaths, and their age-standardized rates.
The findings, based on recently released data from GLOBOCAN 2020 projected to the year 2050, suggest that the lung cancer epidemic will continue to unfold, according to Rajesh Sharma, PhD, et al., in a study published in the International Journal of Clinical Oncology. GLOBOCAN 2020 is an online database produced by the International Agency for Research on Cancer. It provides global cancer statistics from 185 countries for 36 cancer types.
The increase in lung cancer, the leading cancer worldwide in terms of deaths, is generally attributed to increases in cigarette smoking, Sharma et al. wrote. They point out that, while cigarette smoking is expected to have peaked in industrialized countries in the latter half of the twentieth century, the tobacco smoking epidemic is unfolding in regions of Asia and Africa with concomitant increases in lung cancer burden in several countries. Smoking is the most significant lung cancer risk factor, followed by air pollution (especially particulate matter, passive smoking, and occupational exposure to radon and asbestos).
The authors investigated bivariate associations between smoking prevalence and age-standardized rates of lung cancer, and projected lung cancer incident cases and deaths to 2050. They also looked at mortality-to-incidence, considered to be a proxy indicator of 5-year survival, and at human development index, a measure including life expectancy at birth, years of schooling, and standard of living. The results, they state, are expected to aid in policy formulation to combat the lung cancer burden at global, regional, and national levels.
Tobacco smoking prevalence was 21.9% worldwide in 2016, with tobacco smoking prevalence exceeding 25% in 57/149 countries. It was high in European countries with 5 of the top-10 countries among the 149 countries within Europe. Prevalence was greater than 10% in all European countries. Notably, 11/33 countries in Africa had a smoking prevalence less than 10%.
Analysis showed 2.21 million new lung cancer cases and 1.8 million deaths attributed to lung cancer worldwide in 2020, with males accounting for about two-thirds of the burden. The analysis projection for 2050 was for 3.8 million incident cases of lung cancer and 3.2 million lung cancer deaths globally. In 2050, lung cancer cases and deaths are projected to be more than 100,000 in 10/21 regions, led by Eastern Asia, projected to record 1.7 million incident cases and 1.5 million deaths.
The burden of lung cancer in regions of Asia and Africa is expected to increase at least twofold from 2020 to 2050, surpassing European regions that are expected to have the smallest increases. Also, while incident cases will remain much higher in Northern America than in Southeastern Asia and South-Central Asia, the number of lives lost is projected to be similar. The age-specific incidence and death rates rose with age such that the oldest age groups had the highest age-specific rates. With the human development index, mortality-to-incidence showed a negative correlation.
The authors wrote that worsening smoking and pollution levels in developing countries may push the future lung cancer burden much higher than these projections. Unless reversed, cases and death will grow unabated.
“Countering the burden of lung cancer also requires curtailment of other risk factors such as air pollution and exposure to carcinogens,” the authors wrote.
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors. The authors stated that they have no conflicts of interest.
FROM INTERNATIONAL JOURNAL OF CLINICAL ONCOLOGY
ARBs and cancer risk: New meta-analysis raises questions again
The debate on whether the popular class of antihypertensive drugs, angiotensin receptor blockers (ARBs), may be associated with an increased risk for cancer has been reopened with the publication of a new meta-analysis.
The analysis found an increasing risk for cancer, and specifically lung cancer, with increasing cumulative exposure to these drugs.
The findings are reported in a study published online in PLOS ONE.
The author of this new meta-analysis is Ilke Sipahi, MD, a cardiologist from Acibadem University Medical School, Istanbul, who previously raised this issue in an initial meta-analysis published in 2010.
“The new meta-analysis is important because it is the first study to investigate whether there is a dose response in the association between ARBs and cancer,” Dr. Sipahi told this news organization.
“I found a clear signal of increased risk of cancer as exposure to ARBs increased, and the association started to become significant when the maximum dose was taken for 3 years,” he added.
Dr. Sipahi explained that in the first meta-analysis published in Lancet Oncology, he and his colleagues reported an increased cancer risk with ARBs based on observations from high-exposure trials – those that included higher doses of ARBs with a long duration of follow-up.
Following this publication, an investigation by the U.S. Food and Drug Administration refuted the risk, and a collaboration of ARB trial investigators also performed an analysis published in the Journal of Hypertension (2011. doi: 10.1097/HJH.0b013e328344a7de), which again did not show an increased risk for cancer with use of ARBs.
Dr. Sipahi claims that those analyses by the FDA and the ARB Trialists Collaboration, which were all trial-level meta-analyses, diluted the “high exposure” data (including higher doses taken for longer periods of time) with a large amount of other data on much lower exposures (lower doses and/or shorter time periods).
“The overall risk would then inevitably become nonsignificant. These analyses also did not look at different exposure levels,” he says.
“For cancer, the degree of exposure is obviously very important. The risk associated with smoking 2 or 3 cigarettes a day for a year is very different from that of smoking 2 packs a day for 40 years. The same principle applies to taking a medication,” Dr. Sipahi asserts.
From these latest data, he estimates that 120 patients needed to be treated with the maximal daily dose of an ARB for 4.7 years for one excess cancer diagnosis, and 464 patients needed to be treated for one excess lung cancer.
“Given that at least 200 million individuals are being treated with an ARB globally, approximately 1.7 million excess cancers (and 430,000 lung cancers) in 4.6 years could be potentially caused by this class of drugs,” he suggests.
For the current analysis, Dr. Sipahi used trial-level data taken from the paper by the ARB Trialists Collaboration and investigated the effect of exposure to ARBs – including both the dose taken and the length of treatment – on risk for cancer. He performed metaregression analyses that he says has not been done before.
“I mathematically quantitated the degree of exposure in each trial. And when the degree of exposure was correlated with risk of cancer, there was a significant association.”
The new meta-analysis includes 15 randomized controlled trials. The two coprimary outcomes were the relationship between cumulative exposure to ARBs and risk for all cancers combined and the relationship between cumulative exposure and risk for lung cancer.
In the trials, 74,021 patients were randomly assigned to an ARB, resulting in a total cumulative exposure of 172,389 person-years of exposure to daily high dose (or equivalent), and 61,197 patients were randomly assigned to control.
Results showed a highly significant correlation between the degree of cumulative exposure to ARBs and risk for all cancers combined (slope = 0.07; 95% confidence interval, 0.03-0.11; P < .001) and also lung cancer (slope = 0.16; 95% CI, 0.05-0.27; P = .003).
In trials where the cumulative exposure was greater than 3 years of exposure to daily high dose, there was a statistically significant increase in risk for all cancers combined (risk ratio, 1.11; 95% CI, 1.03-1.19; P = .006).
There was also a statistically significant increase in risk for lung cancers in trials where the cumulative exposure was greater than 2.5 years (RR, 1.21; 95% CI, 1.02-1.44; P = .03).
In trials with lower cumulative exposure to ARBs, there was no increased risk either for all cancers combined or lung cancer.
Dr. Sipahi reports that the cumulative exposure-risk relationship with ARBs was independent of background angiotensin-converting enzyme (ACE) inhibitor treatment or the type of control (placebo or nonplacebo control).
But he acknowledges that since this is a trial-level analysis, the effects of patient characteristics such as age and smoking status could not be examined because of lack of patient-level data.
Dr. Sipahi says he does not know the mechanism behind these findings, but he draws attention to the recent withdrawal of several thousand lots of ARB formulations because of the presence of potentially carcinogenic impurities that have been suggested to be a byproduct of ARB synthesis.
He also claims that unlike some other classes of antihypertensives, ARBs have not been shown to reduce the risk for MI, leading him to conclude that “other classes of antihypertensives with good safety and efficacy data (such as ACE-inhibitors, calcium-channel blockers or others) should become the preferred first-line agents in the treatment of hypertension.”
Dr. Sipahi wants the FDA to reinvestigate the issue of ARBs and cancer risk using individual patient data. “They already have the patient-level data from the trials. They should look at it more carefully and look at exposure levels and how they relate to cancer risk,” he said. “And the fact that there have been studies linking high ARB exposure levels to increased cancer risk should at least get a warning on the drug labels.”
A ‘clear increase’ in risk
Dr. Sipahi also points out that a link between ARBs and cancer has been found in another meta-analysis performed in 2013 by senior FDA analyst Thomas Marciniak, MD.
“Because he worked at the FDA, [Dr.] Marciniak had access to individual patent data. This is the best type of analysis and generally produces more accurate results than a trial-level meta-analysis,” Dr. Sipahi commented.
Dr. Marciniak’s analysis, which is available on the FDA website as part of another document, was not officially published elsewhere, and no further action has been taken on the issue.
Contacted by this news organization, Dr. Marciniak, who has now retired from the FDA, said he not only conducted a patient-level meta-analysis but also followed up adverse effects reported in the trials that could have been a symptom of cancer to establish further whether the patient was later diagnosed with cancer or not.
“I used every scrap of information sent in, including serious adverse event reports. I saw a clear increase in lung cancer risk with the ARBs,” Dr. Marciniak said. He did not, however, perform a dose-response relationship analysis.
Asked why his analysis and those from Dr. Sipahi reach different conclusions to those from the ARB Trialists Collaboration and the official FDA investigations, Dr. Marciniak said: “It may be that there were too many low-exposure trials that just washed out the difference. But trial data generally do not capture adverse events such as cancer, which takes a long time to develop, very well, and if you’re not really looking for it, you’re probably not going to find it.”
Dr. Marciniak said that Dr. Sipahi’s current findings are in line with his results. “Finding a dose response, to me, is extremely compelling, and I think the signal here is real,” he commented. “I think this new paper from Dr. Sipahi verifies what I found. I think the FDA should now release all individual patient data it has.”
Contacted for comment, an FDA spokesperson said, “Generally the FDA does not comment on specific studies but evaluates them as part of the body of evidence to further our understanding about a particular issue and assist in our mission to protect public health.”
They added: “The FDA has ongoing assessment, surveillance, compliance, and pharmaceutical quality efforts across every product area, and we will continue to work with drug manufacturers to ensure safe, effective, and high-quality drugs for the American public. When we identify new and previously unrecognized risks to safety and quality, we react swiftly to resolve the problem, as we have done in responding to the recent findings of nitrosamines in certain medicines.”
Analysis ‘should be taken seriously’
Commenting on this new study, Steve Nissen, MD, a key figure in analyzing such complex data and who has himself uncovered problems with high-profile drugs in the past, says the current analysis should be taken seriously.
Dr. Nissen, who was Dr. Sipahi’s senior during his post-doc position at the Cleveland Clinic, wrote an editorial accompanying Dr. Sipahi’s first paper and calling for urgent regulatory review of the evidence.
He says the new findings add to previous evidence suggesting a possible risk for cancer with ARBs.
“[Dr.] Sipahi is a capable researcher, and this analysis needs to be taken seriously, but it needs to be verified. It is not possible to draw a strong conclusion on this analysis, as it is not based on individual patient data, but I don’t think it should be ignored,” Dr. Nissen stated.
“I will say again what I said 12 years ago – that the regulatory agencies need to carefully review all their data in a very detailed way. The FDA and EMA have access to the individual patient data and are both very capable of doing the required analyses.”
Limitations of trial-level analysis
Asked to evaluate the statistics in the current paper, Andrew Althouse, PhD, an assistant professor of medicine at the University of Pittsburgh, and a clinical trial statistician, explained that the best way to do a thorough analysis of the relationship between ARB exposure and risk for incident cancer would involve the use of patient-level data.
“As such data were not available to Dr. Sipahi, I believe he is doing as well as he can. But without full access to individual patient-level data from the respective trials, it is difficult to support any firm conclusions,” Dr. Althouse said in an interview.
He suggested that the meta-regression analyses used in the paper were unable to properly estimate the relationship between ARB exposure and risk for incident cancer.
“Taken at face value, the current analysis suggests that [in] trials with longer follow-up duration (and therefore greater cumulative exposure to ARB for the treatment group), the risk of developing cancer for patients in the ARB group versus the non-ARB group was progressively higher. But this study doesn’t take into account the actual amount of follow-up time for individual patients or potential differences in the amount of follow-up time between the two groups in each trial,” he noted.
Dr. Althouse says this raises the possibility of “competing risks” or the idea that if ARBs reduce cardiovascular disease and cardiovascular death, then there would be more patients remaining in that arm who could go on to develop cancer. “So a crude count of the number of cancer cases may look as though patients receiving ARBs are ‘more likely’ to develop cancer, but this is a mirage.”
He added: “When there are some patients dying during the study, the only way to tell whether the intervention actually increased the risk of other health-related complications is to have an analysis that properly accounts for each patient’s time-at-risk of the outcome. Unfortunately, properly analyzing this requires the use of patient-level data.”
Cardiologists skeptical?
Cardiology experts asked for thoughts on the new meta-analysis were also cautious to read too much into the findings.
Franz Messerli, MD, professor of medicine at the University of Bern, Switzerland, commented: “Perhaps one would simply ignore this rambling, cherrypicking-based condemnation of ARBs if it were not for the powerful negative connotation of the word cancer. Thus, the meta-analysis of Dr. Sipahi purporting that ARBs could be increasing the development of cancers in a cumulative way is of concern to both physicians and patients.”
But, raising a similar point to Dr. Althouse about competing risks, Dr. Messerli said: “We have to consider that as one gets older, the cardiovascular disease state and cancer state will compete with each other for the outcome of death. The better that therapies protect against cardiovascular death, the more they will increase life expectancy and thus the risk of cancer.”
He also added that “in head-to-head comparisons with ACE inhibitors, ARBs showed similar efficacy in terms of death, CV mortality, MI, stroke, and end-stage kidney disease, so can we agree that the attempt of Dr. Sipahi to disparage ARBs as a class is much ado about nothing?”
Dr. Nissen, however, said he views the idea of competing risk as “a bit of a stretch” in this case. “Although ARBs are effective antihypertensive drugs, I would say there is very little evidence that they would prolong survival versus other antihypertensives.”
Dr. Sipahi also claims that this argument is not relevant to the current analysis. “ARBs did not increase survival in any of the high-exposure trials that showed an excess in cancers. Therefore, competing outcomes, or ‘survival bias’ to be more specific, is not a possibility here,” he says.
George Bakris, MD, professor of medicine at the University of Chicago Medicine, noted that while the current study shows a slight increase in cancer incidence, especially lung cancer, among those taking ARBs for more than 3 years, it “totally ignores the overwhelming cardiovascular risk reduction seen in the trials.”
“Moreover,” he adds, “the author notes that the findings were independent of ACE-inhibitors, but he can’t rule out smoking and age as factors, two major risk factors for cancer and lung cancer, specifically. Thus, as typical of these types of analyses, the associations are probably true/true unrelated or, at best, partially related.”
Dr. Bakris referred to the potentially carcinogenic nitrosamine and azido compounds found in several ARB formulations that have resulted in recalls.
“At any stage of drug synthesis throughout each product’s lifetime, these impurities may evolve if an amine reacts with a nitrosating agent coexisting under appropriate conditions,” he said. “Drug regulatory authorities worldwide have established stringent guidelines on nitrosamine contamination for all drug products. The studies noted in the author’s analysis were done well before these guidelines were implemented. Hence, many of the issues raised by the authors using trials from 10-20 years ago are not of significant concern.”
Still, the cardiology experts all agreed on one thing – that patients should continue to take ARBs as prescribed.
Noting that worldwide authorities are now addressing the issue of possible carcinogen contamination, Dr. Bakris stressed that patients “should not panic and should not stop their meds.”
Dr. Nissen added: “What we don’t want is for patents who are taking ARBs to stop taking these medications – hypertension is a deadly disorder, and these drugs have proven cardiovascular benefits.”
Dr. Sipahi received no specific funding for this work. He reports receiving lecture honoraria from Novartis, Boehringer Ingelheim, Sanofi, Sandoz, Bristol-Myers Squibb, Bayer, Pfizer, Ranbaxy, Servier, and ARIS and served on advisory boards for Novartis, Sanofi, Servier, Bristol-Myers Squibb, Pfizer, Bayer and I.E. Ulagay. The other commenters do not report any relevant disclosures.
A version of this article first appeared on Medscape.com.
The debate on whether the popular class of antihypertensive drugs, angiotensin receptor blockers (ARBs), may be associated with an increased risk for cancer has been reopened with the publication of a new meta-analysis.
The analysis found an increasing risk for cancer, and specifically lung cancer, with increasing cumulative exposure to these drugs.
The findings are reported in a study published online in PLOS ONE.
The author of this new meta-analysis is Ilke Sipahi, MD, a cardiologist from Acibadem University Medical School, Istanbul, who previously raised this issue in an initial meta-analysis published in 2010.
“The new meta-analysis is important because it is the first study to investigate whether there is a dose response in the association between ARBs and cancer,” Dr. Sipahi told this news organization.
“I found a clear signal of increased risk of cancer as exposure to ARBs increased, and the association started to become significant when the maximum dose was taken for 3 years,” he added.
Dr. Sipahi explained that in the first meta-analysis published in Lancet Oncology, he and his colleagues reported an increased cancer risk with ARBs based on observations from high-exposure trials – those that included higher doses of ARBs with a long duration of follow-up.
Following this publication, an investigation by the U.S. Food and Drug Administration refuted the risk, and a collaboration of ARB trial investigators also performed an analysis published in the Journal of Hypertension (2011. doi: 10.1097/HJH.0b013e328344a7de), which again did not show an increased risk for cancer with use of ARBs.
Dr. Sipahi claims that those analyses by the FDA and the ARB Trialists Collaboration, which were all trial-level meta-analyses, diluted the “high exposure” data (including higher doses taken for longer periods of time) with a large amount of other data on much lower exposures (lower doses and/or shorter time periods).
“The overall risk would then inevitably become nonsignificant. These analyses also did not look at different exposure levels,” he says.
“For cancer, the degree of exposure is obviously very important. The risk associated with smoking 2 or 3 cigarettes a day for a year is very different from that of smoking 2 packs a day for 40 years. The same principle applies to taking a medication,” Dr. Sipahi asserts.
From these latest data, he estimates that 120 patients needed to be treated with the maximal daily dose of an ARB for 4.7 years for one excess cancer diagnosis, and 464 patients needed to be treated for one excess lung cancer.
“Given that at least 200 million individuals are being treated with an ARB globally, approximately 1.7 million excess cancers (and 430,000 lung cancers) in 4.6 years could be potentially caused by this class of drugs,” he suggests.
For the current analysis, Dr. Sipahi used trial-level data taken from the paper by the ARB Trialists Collaboration and investigated the effect of exposure to ARBs – including both the dose taken and the length of treatment – on risk for cancer. He performed metaregression analyses that he says has not been done before.
“I mathematically quantitated the degree of exposure in each trial. And when the degree of exposure was correlated with risk of cancer, there was a significant association.”
The new meta-analysis includes 15 randomized controlled trials. The two coprimary outcomes were the relationship between cumulative exposure to ARBs and risk for all cancers combined and the relationship between cumulative exposure and risk for lung cancer.
In the trials, 74,021 patients were randomly assigned to an ARB, resulting in a total cumulative exposure of 172,389 person-years of exposure to daily high dose (or equivalent), and 61,197 patients were randomly assigned to control.
Results showed a highly significant correlation between the degree of cumulative exposure to ARBs and risk for all cancers combined (slope = 0.07; 95% confidence interval, 0.03-0.11; P < .001) and also lung cancer (slope = 0.16; 95% CI, 0.05-0.27; P = .003).
In trials where the cumulative exposure was greater than 3 years of exposure to daily high dose, there was a statistically significant increase in risk for all cancers combined (risk ratio, 1.11; 95% CI, 1.03-1.19; P = .006).
There was also a statistically significant increase in risk for lung cancers in trials where the cumulative exposure was greater than 2.5 years (RR, 1.21; 95% CI, 1.02-1.44; P = .03).
In trials with lower cumulative exposure to ARBs, there was no increased risk either for all cancers combined or lung cancer.
Dr. Sipahi reports that the cumulative exposure-risk relationship with ARBs was independent of background angiotensin-converting enzyme (ACE) inhibitor treatment or the type of control (placebo or nonplacebo control).
But he acknowledges that since this is a trial-level analysis, the effects of patient characteristics such as age and smoking status could not be examined because of lack of patient-level data.
Dr. Sipahi says he does not know the mechanism behind these findings, but he draws attention to the recent withdrawal of several thousand lots of ARB formulations because of the presence of potentially carcinogenic impurities that have been suggested to be a byproduct of ARB synthesis.
He also claims that unlike some other classes of antihypertensives, ARBs have not been shown to reduce the risk for MI, leading him to conclude that “other classes of antihypertensives with good safety and efficacy data (such as ACE-inhibitors, calcium-channel blockers or others) should become the preferred first-line agents in the treatment of hypertension.”
Dr. Sipahi wants the FDA to reinvestigate the issue of ARBs and cancer risk using individual patient data. “They already have the patient-level data from the trials. They should look at it more carefully and look at exposure levels and how they relate to cancer risk,” he said. “And the fact that there have been studies linking high ARB exposure levels to increased cancer risk should at least get a warning on the drug labels.”
A ‘clear increase’ in risk
Dr. Sipahi also points out that a link between ARBs and cancer has been found in another meta-analysis performed in 2013 by senior FDA analyst Thomas Marciniak, MD.
“Because he worked at the FDA, [Dr.] Marciniak had access to individual patent data. This is the best type of analysis and generally produces more accurate results than a trial-level meta-analysis,” Dr. Sipahi commented.
Dr. Marciniak’s analysis, which is available on the FDA website as part of another document, was not officially published elsewhere, and no further action has been taken on the issue.
Contacted by this news organization, Dr. Marciniak, who has now retired from the FDA, said he not only conducted a patient-level meta-analysis but also followed up adverse effects reported in the trials that could have been a symptom of cancer to establish further whether the patient was later diagnosed with cancer or not.
“I used every scrap of information sent in, including serious adverse event reports. I saw a clear increase in lung cancer risk with the ARBs,” Dr. Marciniak said. He did not, however, perform a dose-response relationship analysis.
Asked why his analysis and those from Dr. Sipahi reach different conclusions to those from the ARB Trialists Collaboration and the official FDA investigations, Dr. Marciniak said: “It may be that there were too many low-exposure trials that just washed out the difference. But trial data generally do not capture adverse events such as cancer, which takes a long time to develop, very well, and if you’re not really looking for it, you’re probably not going to find it.”
Dr. Marciniak said that Dr. Sipahi’s current findings are in line with his results. “Finding a dose response, to me, is extremely compelling, and I think the signal here is real,” he commented. “I think this new paper from Dr. Sipahi verifies what I found. I think the FDA should now release all individual patient data it has.”
Contacted for comment, an FDA spokesperson said, “Generally the FDA does not comment on specific studies but evaluates them as part of the body of evidence to further our understanding about a particular issue and assist in our mission to protect public health.”
They added: “The FDA has ongoing assessment, surveillance, compliance, and pharmaceutical quality efforts across every product area, and we will continue to work with drug manufacturers to ensure safe, effective, and high-quality drugs for the American public. When we identify new and previously unrecognized risks to safety and quality, we react swiftly to resolve the problem, as we have done in responding to the recent findings of nitrosamines in certain medicines.”
Analysis ‘should be taken seriously’
Commenting on this new study, Steve Nissen, MD, a key figure in analyzing such complex data and who has himself uncovered problems with high-profile drugs in the past, says the current analysis should be taken seriously.
Dr. Nissen, who was Dr. Sipahi’s senior during his post-doc position at the Cleveland Clinic, wrote an editorial accompanying Dr. Sipahi’s first paper and calling for urgent regulatory review of the evidence.
He says the new findings add to previous evidence suggesting a possible risk for cancer with ARBs.
“[Dr.] Sipahi is a capable researcher, and this analysis needs to be taken seriously, but it needs to be verified. It is not possible to draw a strong conclusion on this analysis, as it is not based on individual patient data, but I don’t think it should be ignored,” Dr. Nissen stated.
“I will say again what I said 12 years ago – that the regulatory agencies need to carefully review all their data in a very detailed way. The FDA and EMA have access to the individual patient data and are both very capable of doing the required analyses.”
Limitations of trial-level analysis
Asked to evaluate the statistics in the current paper, Andrew Althouse, PhD, an assistant professor of medicine at the University of Pittsburgh, and a clinical trial statistician, explained that the best way to do a thorough analysis of the relationship between ARB exposure and risk for incident cancer would involve the use of patient-level data.
“As such data were not available to Dr. Sipahi, I believe he is doing as well as he can. But without full access to individual patient-level data from the respective trials, it is difficult to support any firm conclusions,” Dr. Althouse said in an interview.
He suggested that the meta-regression analyses used in the paper were unable to properly estimate the relationship between ARB exposure and risk for incident cancer.
“Taken at face value, the current analysis suggests that [in] trials with longer follow-up duration (and therefore greater cumulative exposure to ARB for the treatment group), the risk of developing cancer for patients in the ARB group versus the non-ARB group was progressively higher. But this study doesn’t take into account the actual amount of follow-up time for individual patients or potential differences in the amount of follow-up time between the two groups in each trial,” he noted.
Dr. Althouse says this raises the possibility of “competing risks” or the idea that if ARBs reduce cardiovascular disease and cardiovascular death, then there would be more patients remaining in that arm who could go on to develop cancer. “So a crude count of the number of cancer cases may look as though patients receiving ARBs are ‘more likely’ to develop cancer, but this is a mirage.”
He added: “When there are some patients dying during the study, the only way to tell whether the intervention actually increased the risk of other health-related complications is to have an analysis that properly accounts for each patient’s time-at-risk of the outcome. Unfortunately, properly analyzing this requires the use of patient-level data.”
Cardiologists skeptical?
Cardiology experts asked for thoughts on the new meta-analysis were also cautious to read too much into the findings.
Franz Messerli, MD, professor of medicine at the University of Bern, Switzerland, commented: “Perhaps one would simply ignore this rambling, cherrypicking-based condemnation of ARBs if it were not for the powerful negative connotation of the word cancer. Thus, the meta-analysis of Dr. Sipahi purporting that ARBs could be increasing the development of cancers in a cumulative way is of concern to both physicians and patients.”
But, raising a similar point to Dr. Althouse about competing risks, Dr. Messerli said: “We have to consider that as one gets older, the cardiovascular disease state and cancer state will compete with each other for the outcome of death. The better that therapies protect against cardiovascular death, the more they will increase life expectancy and thus the risk of cancer.”
He also added that “in head-to-head comparisons with ACE inhibitors, ARBs showed similar efficacy in terms of death, CV mortality, MI, stroke, and end-stage kidney disease, so can we agree that the attempt of Dr. Sipahi to disparage ARBs as a class is much ado about nothing?”
Dr. Nissen, however, said he views the idea of competing risk as “a bit of a stretch” in this case. “Although ARBs are effective antihypertensive drugs, I would say there is very little evidence that they would prolong survival versus other antihypertensives.”
Dr. Sipahi also claims that this argument is not relevant to the current analysis. “ARBs did not increase survival in any of the high-exposure trials that showed an excess in cancers. Therefore, competing outcomes, or ‘survival bias’ to be more specific, is not a possibility here,” he says.
George Bakris, MD, professor of medicine at the University of Chicago Medicine, noted that while the current study shows a slight increase in cancer incidence, especially lung cancer, among those taking ARBs for more than 3 years, it “totally ignores the overwhelming cardiovascular risk reduction seen in the trials.”
“Moreover,” he adds, “the author notes that the findings were independent of ACE-inhibitors, but he can’t rule out smoking and age as factors, two major risk factors for cancer and lung cancer, specifically. Thus, as typical of these types of analyses, the associations are probably true/true unrelated or, at best, partially related.”
Dr. Bakris referred to the potentially carcinogenic nitrosamine and azido compounds found in several ARB formulations that have resulted in recalls.
“At any stage of drug synthesis throughout each product’s lifetime, these impurities may evolve if an amine reacts with a nitrosating agent coexisting under appropriate conditions,” he said. “Drug regulatory authorities worldwide have established stringent guidelines on nitrosamine contamination for all drug products. The studies noted in the author’s analysis were done well before these guidelines were implemented. Hence, many of the issues raised by the authors using trials from 10-20 years ago are not of significant concern.”
Still, the cardiology experts all agreed on one thing – that patients should continue to take ARBs as prescribed.
Noting that worldwide authorities are now addressing the issue of possible carcinogen contamination, Dr. Bakris stressed that patients “should not panic and should not stop their meds.”
Dr. Nissen added: “What we don’t want is for patents who are taking ARBs to stop taking these medications – hypertension is a deadly disorder, and these drugs have proven cardiovascular benefits.”
Dr. Sipahi received no specific funding for this work. He reports receiving lecture honoraria from Novartis, Boehringer Ingelheim, Sanofi, Sandoz, Bristol-Myers Squibb, Bayer, Pfizer, Ranbaxy, Servier, and ARIS and served on advisory boards for Novartis, Sanofi, Servier, Bristol-Myers Squibb, Pfizer, Bayer and I.E. Ulagay. The other commenters do not report any relevant disclosures.
A version of this article first appeared on Medscape.com.
The debate on whether the popular class of antihypertensive drugs, angiotensin receptor blockers (ARBs), may be associated with an increased risk for cancer has been reopened with the publication of a new meta-analysis.
The analysis found an increasing risk for cancer, and specifically lung cancer, with increasing cumulative exposure to these drugs.
The findings are reported in a study published online in PLOS ONE.
The author of this new meta-analysis is Ilke Sipahi, MD, a cardiologist from Acibadem University Medical School, Istanbul, who previously raised this issue in an initial meta-analysis published in 2010.
“The new meta-analysis is important because it is the first study to investigate whether there is a dose response in the association between ARBs and cancer,” Dr. Sipahi told this news organization.
“I found a clear signal of increased risk of cancer as exposure to ARBs increased, and the association started to become significant when the maximum dose was taken for 3 years,” he added.
Dr. Sipahi explained that in the first meta-analysis published in Lancet Oncology, he and his colleagues reported an increased cancer risk with ARBs based on observations from high-exposure trials – those that included higher doses of ARBs with a long duration of follow-up.
Following this publication, an investigation by the U.S. Food and Drug Administration refuted the risk, and a collaboration of ARB trial investigators also performed an analysis published in the Journal of Hypertension (2011. doi: 10.1097/HJH.0b013e328344a7de), which again did not show an increased risk for cancer with use of ARBs.
Dr. Sipahi claims that those analyses by the FDA and the ARB Trialists Collaboration, which were all trial-level meta-analyses, diluted the “high exposure” data (including higher doses taken for longer periods of time) with a large amount of other data on much lower exposures (lower doses and/or shorter time periods).
“The overall risk would then inevitably become nonsignificant. These analyses also did not look at different exposure levels,” he says.
“For cancer, the degree of exposure is obviously very important. The risk associated with smoking 2 or 3 cigarettes a day for a year is very different from that of smoking 2 packs a day for 40 years. The same principle applies to taking a medication,” Dr. Sipahi asserts.
From these latest data, he estimates that 120 patients needed to be treated with the maximal daily dose of an ARB for 4.7 years for one excess cancer diagnosis, and 464 patients needed to be treated for one excess lung cancer.
“Given that at least 200 million individuals are being treated with an ARB globally, approximately 1.7 million excess cancers (and 430,000 lung cancers) in 4.6 years could be potentially caused by this class of drugs,” he suggests.
For the current analysis, Dr. Sipahi used trial-level data taken from the paper by the ARB Trialists Collaboration and investigated the effect of exposure to ARBs – including both the dose taken and the length of treatment – on risk for cancer. He performed metaregression analyses that he says has not been done before.
“I mathematically quantitated the degree of exposure in each trial. And when the degree of exposure was correlated with risk of cancer, there was a significant association.”
The new meta-analysis includes 15 randomized controlled trials. The two coprimary outcomes were the relationship between cumulative exposure to ARBs and risk for all cancers combined and the relationship between cumulative exposure and risk for lung cancer.
In the trials, 74,021 patients were randomly assigned to an ARB, resulting in a total cumulative exposure of 172,389 person-years of exposure to daily high dose (or equivalent), and 61,197 patients were randomly assigned to control.
Results showed a highly significant correlation between the degree of cumulative exposure to ARBs and risk for all cancers combined (slope = 0.07; 95% confidence interval, 0.03-0.11; P < .001) and also lung cancer (slope = 0.16; 95% CI, 0.05-0.27; P = .003).
In trials where the cumulative exposure was greater than 3 years of exposure to daily high dose, there was a statistically significant increase in risk for all cancers combined (risk ratio, 1.11; 95% CI, 1.03-1.19; P = .006).
There was also a statistically significant increase in risk for lung cancers in trials where the cumulative exposure was greater than 2.5 years (RR, 1.21; 95% CI, 1.02-1.44; P = .03).
In trials with lower cumulative exposure to ARBs, there was no increased risk either for all cancers combined or lung cancer.
Dr. Sipahi reports that the cumulative exposure-risk relationship with ARBs was independent of background angiotensin-converting enzyme (ACE) inhibitor treatment or the type of control (placebo or nonplacebo control).
But he acknowledges that since this is a trial-level analysis, the effects of patient characteristics such as age and smoking status could not be examined because of lack of patient-level data.
Dr. Sipahi says he does not know the mechanism behind these findings, but he draws attention to the recent withdrawal of several thousand lots of ARB formulations because of the presence of potentially carcinogenic impurities that have been suggested to be a byproduct of ARB synthesis.
He also claims that unlike some other classes of antihypertensives, ARBs have not been shown to reduce the risk for MI, leading him to conclude that “other classes of antihypertensives with good safety and efficacy data (such as ACE-inhibitors, calcium-channel blockers or others) should become the preferred first-line agents in the treatment of hypertension.”
Dr. Sipahi wants the FDA to reinvestigate the issue of ARBs and cancer risk using individual patient data. “They already have the patient-level data from the trials. They should look at it more carefully and look at exposure levels and how they relate to cancer risk,” he said. “And the fact that there have been studies linking high ARB exposure levels to increased cancer risk should at least get a warning on the drug labels.”
A ‘clear increase’ in risk
Dr. Sipahi also points out that a link between ARBs and cancer has been found in another meta-analysis performed in 2013 by senior FDA analyst Thomas Marciniak, MD.
“Because he worked at the FDA, [Dr.] Marciniak had access to individual patent data. This is the best type of analysis and generally produces more accurate results than a trial-level meta-analysis,” Dr. Sipahi commented.
Dr. Marciniak’s analysis, which is available on the FDA website as part of another document, was not officially published elsewhere, and no further action has been taken on the issue.
Contacted by this news organization, Dr. Marciniak, who has now retired from the FDA, said he not only conducted a patient-level meta-analysis but also followed up adverse effects reported in the trials that could have been a symptom of cancer to establish further whether the patient was later diagnosed with cancer or not.
“I used every scrap of information sent in, including serious adverse event reports. I saw a clear increase in lung cancer risk with the ARBs,” Dr. Marciniak said. He did not, however, perform a dose-response relationship analysis.
Asked why his analysis and those from Dr. Sipahi reach different conclusions to those from the ARB Trialists Collaboration and the official FDA investigations, Dr. Marciniak said: “It may be that there were too many low-exposure trials that just washed out the difference. But trial data generally do not capture adverse events such as cancer, which takes a long time to develop, very well, and if you’re not really looking for it, you’re probably not going to find it.”
Dr. Marciniak said that Dr. Sipahi’s current findings are in line with his results. “Finding a dose response, to me, is extremely compelling, and I think the signal here is real,” he commented. “I think this new paper from Dr. Sipahi verifies what I found. I think the FDA should now release all individual patient data it has.”
Contacted for comment, an FDA spokesperson said, “Generally the FDA does not comment on specific studies but evaluates them as part of the body of evidence to further our understanding about a particular issue and assist in our mission to protect public health.”
They added: “The FDA has ongoing assessment, surveillance, compliance, and pharmaceutical quality efforts across every product area, and we will continue to work with drug manufacturers to ensure safe, effective, and high-quality drugs for the American public. When we identify new and previously unrecognized risks to safety and quality, we react swiftly to resolve the problem, as we have done in responding to the recent findings of nitrosamines in certain medicines.”
Analysis ‘should be taken seriously’
Commenting on this new study, Steve Nissen, MD, a key figure in analyzing such complex data and who has himself uncovered problems with high-profile drugs in the past, says the current analysis should be taken seriously.
Dr. Nissen, who was Dr. Sipahi’s senior during his post-doc position at the Cleveland Clinic, wrote an editorial accompanying Dr. Sipahi’s first paper and calling for urgent regulatory review of the evidence.
He says the new findings add to previous evidence suggesting a possible risk for cancer with ARBs.
“[Dr.] Sipahi is a capable researcher, and this analysis needs to be taken seriously, but it needs to be verified. It is not possible to draw a strong conclusion on this analysis, as it is not based on individual patient data, but I don’t think it should be ignored,” Dr. Nissen stated.
“I will say again what I said 12 years ago – that the regulatory agencies need to carefully review all their data in a very detailed way. The FDA and EMA have access to the individual patient data and are both very capable of doing the required analyses.”
Limitations of trial-level analysis
Asked to evaluate the statistics in the current paper, Andrew Althouse, PhD, an assistant professor of medicine at the University of Pittsburgh, and a clinical trial statistician, explained that the best way to do a thorough analysis of the relationship between ARB exposure and risk for incident cancer would involve the use of patient-level data.
“As such data were not available to Dr. Sipahi, I believe he is doing as well as he can. But without full access to individual patient-level data from the respective trials, it is difficult to support any firm conclusions,” Dr. Althouse said in an interview.
He suggested that the meta-regression analyses used in the paper were unable to properly estimate the relationship between ARB exposure and risk for incident cancer.
“Taken at face value, the current analysis suggests that [in] trials with longer follow-up duration (and therefore greater cumulative exposure to ARB for the treatment group), the risk of developing cancer for patients in the ARB group versus the non-ARB group was progressively higher. But this study doesn’t take into account the actual amount of follow-up time for individual patients or potential differences in the amount of follow-up time between the two groups in each trial,” he noted.
Dr. Althouse says this raises the possibility of “competing risks” or the idea that if ARBs reduce cardiovascular disease and cardiovascular death, then there would be more patients remaining in that arm who could go on to develop cancer. “So a crude count of the number of cancer cases may look as though patients receiving ARBs are ‘more likely’ to develop cancer, but this is a mirage.”
He added: “When there are some patients dying during the study, the only way to tell whether the intervention actually increased the risk of other health-related complications is to have an analysis that properly accounts for each patient’s time-at-risk of the outcome. Unfortunately, properly analyzing this requires the use of patient-level data.”
Cardiologists skeptical?
Cardiology experts asked for thoughts on the new meta-analysis were also cautious to read too much into the findings.
Franz Messerli, MD, professor of medicine at the University of Bern, Switzerland, commented: “Perhaps one would simply ignore this rambling, cherrypicking-based condemnation of ARBs if it were not for the powerful negative connotation of the word cancer. Thus, the meta-analysis of Dr. Sipahi purporting that ARBs could be increasing the development of cancers in a cumulative way is of concern to both physicians and patients.”
But, raising a similar point to Dr. Althouse about competing risks, Dr. Messerli said: “We have to consider that as one gets older, the cardiovascular disease state and cancer state will compete with each other for the outcome of death. The better that therapies protect against cardiovascular death, the more they will increase life expectancy and thus the risk of cancer.”
He also added that “in head-to-head comparisons with ACE inhibitors, ARBs showed similar efficacy in terms of death, CV mortality, MI, stroke, and end-stage kidney disease, so can we agree that the attempt of Dr. Sipahi to disparage ARBs as a class is much ado about nothing?”
Dr. Nissen, however, said he views the idea of competing risk as “a bit of a stretch” in this case. “Although ARBs are effective antihypertensive drugs, I would say there is very little evidence that they would prolong survival versus other antihypertensives.”
Dr. Sipahi also claims that this argument is not relevant to the current analysis. “ARBs did not increase survival in any of the high-exposure trials that showed an excess in cancers. Therefore, competing outcomes, or ‘survival bias’ to be more specific, is not a possibility here,” he says.
George Bakris, MD, professor of medicine at the University of Chicago Medicine, noted that while the current study shows a slight increase in cancer incidence, especially lung cancer, among those taking ARBs for more than 3 years, it “totally ignores the overwhelming cardiovascular risk reduction seen in the trials.”
“Moreover,” he adds, “the author notes that the findings were independent of ACE-inhibitors, but he can’t rule out smoking and age as factors, two major risk factors for cancer and lung cancer, specifically. Thus, as typical of these types of analyses, the associations are probably true/true unrelated or, at best, partially related.”
Dr. Bakris referred to the potentially carcinogenic nitrosamine and azido compounds found in several ARB formulations that have resulted in recalls.
“At any stage of drug synthesis throughout each product’s lifetime, these impurities may evolve if an amine reacts with a nitrosating agent coexisting under appropriate conditions,” he said. “Drug regulatory authorities worldwide have established stringent guidelines on nitrosamine contamination for all drug products. The studies noted in the author’s analysis were done well before these guidelines were implemented. Hence, many of the issues raised by the authors using trials from 10-20 years ago are not of significant concern.”
Still, the cardiology experts all agreed on one thing – that patients should continue to take ARBs as prescribed.
Noting that worldwide authorities are now addressing the issue of possible carcinogen contamination, Dr. Bakris stressed that patients “should not panic and should not stop their meds.”
Dr. Nissen added: “What we don’t want is for patents who are taking ARBs to stop taking these medications – hypertension is a deadly disorder, and these drugs have proven cardiovascular benefits.”
Dr. Sipahi received no specific funding for this work. He reports receiving lecture honoraria from Novartis, Boehringer Ingelheim, Sanofi, Sandoz, Bristol-Myers Squibb, Bayer, Pfizer, Ranbaxy, Servier, and ARIS and served on advisory boards for Novartis, Sanofi, Servier, Bristol-Myers Squibb, Pfizer, Bayer and I.E. Ulagay. The other commenters do not report any relevant disclosures.
A version of this article first appeared on Medscape.com.
Is a progression-free survival benefit alone really worth $10,000 a month?
In the field of lung cancer, and more broadly in oncology, many of our biggest advances in 2021 have come as clinically meaningful improvements in surrogate endpoints – disease-free survival, progression-free survival, and sometimes even pathologic complete response rate.
I have historically been most compelled to consider new findings to be practice-changing when they improve overall survival or quality of life – the endpoints that translate to direct benefits for patients. However, I also feel it is appropriate to call surrogate endpoints practice-changing when they can predict improvements in overall survival or quality of life.
Take the PACIFIC trial, which assessed maintenance durvalumab after concurrent chemoradiation for unresectable stage III non–small cell lung cancer (NSCLC).
Back in 2017, I was initially unconvinced by the interim phase 3 data that were presented in a press release that highlighted the disease-free survival benefit. However, after examining additional data more closely, I saw the dramatic improvement in time to distant relapse or death was overwhelmingly likely to predict an improvement in overall survival – a benefit that the data subsequently bore out.
More recently, the disease-free survival results for adjuvant osimertinib in resected endothelial growth factor receptor mutation–positive NSCLC and adjuvant atezolizumab in resected programmed death-ligand 1–positive stage II-IIIA NSCLC have led to excitement about Food and Drug Administration approvals for these therapies. Although there is reason to be cautious about the likelihood of an overall survival benefit with either therapy – particularly for patients with low programmed death-ligand 1 who receive atezolizumab – I think that the results are promising enough to discuss these treatment options with appropriate patients.
Some argue, however, that overall survival is not necessarily a critical goal and that certain surrogate endpoints are inherently beneficial. Patients and oncologists may, for instance, view delaying disease progression as a win, even if overall survival remains the same.
I appreciate the view that favorable scan results are an achievement, even without a survival benefit. Patients appreciate the good news, and it is gratifying for us to deliver it. However, what remains unspoken is whether the benefit can be provided at a reasonable value given the financial costs associated with the new treatment.
In the United States, we consider the physician-patient relationship to be autonomous and even revered, but we conveniently ignore the fact that both are deciding on treatments that are funded by people who are not represented in the room. And in a health care system that fails to cover basic cancer care needs as well as other critical, high-value interventions for both the uninsured and underinsured, we should acknowledge that our decisions redirect limited resources from others.
Is it the best use of $10,000 per month for a new drug that improves disease-free survival but not overall survival?
At the same time, we also have to remain vigilant and reflect on whether we are echoing the marketing messages of the companies selling these treatments. Having recently watched the excellent Hulu series Dopesick, which realistically portrays the medical community’s egregious overuse of Oxycontin at the behest of Purdue Pharmaceuticals, it is striking to see how effectively the pharmaceutical industry can co-opt stakeholders. Very few physicians or patients have expertise in health care policy with broad societal perspective, yet subspecialists offer edicts as if society should dedicate unlimited resources first and foremost to our career focus or personal cause.
I certainly appreciate the appeal of surrogate endpoints in a world in which we hope to offer novel therapies to patients in a timely fashion. In the next few years, some of our most promising data in oncology will demand that we consider whether surrogate endpoints are practice-changing. We are facing a fundamental question: Are we using these surrogate endpoints to predict overall survival or quality of life or do these endpoints stand on their own as practice-changing metrics?
We need to acknowledge that our primary clinical focus is not the only one that deserves our attention, particularly when our treatment decisions are, in fact, spending other people’s money. We should be asking not whether we prefer to deliver good news after a scan, but whether that alone is enough to justify the high cost of a new treatment without an overall survival benefit.
Dr. West disclosed serving as a director, officer, partner, employee, adviser, consultant, or trustee for Ariad/Takeda, Bristol-Myers Squibb, Boehringer Ingelheim, Spectrum, AstraZeneca, Celgene, Genentech/Roche, Pfizer, and Merck; serving as a speaker or a member of a speakers bureau for Ariad/Takeda, AstraZeneca, and Genentech/Roche; and receiving income from Eli Lilly. A version of this article first appeared on Medscape.com.
In the field of lung cancer, and more broadly in oncology, many of our biggest advances in 2021 have come as clinically meaningful improvements in surrogate endpoints – disease-free survival, progression-free survival, and sometimes even pathologic complete response rate.
I have historically been most compelled to consider new findings to be practice-changing when they improve overall survival or quality of life – the endpoints that translate to direct benefits for patients. However, I also feel it is appropriate to call surrogate endpoints practice-changing when they can predict improvements in overall survival or quality of life.
Take the PACIFIC trial, which assessed maintenance durvalumab after concurrent chemoradiation for unresectable stage III non–small cell lung cancer (NSCLC).
Back in 2017, I was initially unconvinced by the interim phase 3 data that were presented in a press release that highlighted the disease-free survival benefit. However, after examining additional data more closely, I saw the dramatic improvement in time to distant relapse or death was overwhelmingly likely to predict an improvement in overall survival – a benefit that the data subsequently bore out.
More recently, the disease-free survival results for adjuvant osimertinib in resected endothelial growth factor receptor mutation–positive NSCLC and adjuvant atezolizumab in resected programmed death-ligand 1–positive stage II-IIIA NSCLC have led to excitement about Food and Drug Administration approvals for these therapies. Although there is reason to be cautious about the likelihood of an overall survival benefit with either therapy – particularly for patients with low programmed death-ligand 1 who receive atezolizumab – I think that the results are promising enough to discuss these treatment options with appropriate patients.
Some argue, however, that overall survival is not necessarily a critical goal and that certain surrogate endpoints are inherently beneficial. Patients and oncologists may, for instance, view delaying disease progression as a win, even if overall survival remains the same.
I appreciate the view that favorable scan results are an achievement, even without a survival benefit. Patients appreciate the good news, and it is gratifying for us to deliver it. However, what remains unspoken is whether the benefit can be provided at a reasonable value given the financial costs associated with the new treatment.
In the United States, we consider the physician-patient relationship to be autonomous and even revered, but we conveniently ignore the fact that both are deciding on treatments that are funded by people who are not represented in the room. And in a health care system that fails to cover basic cancer care needs as well as other critical, high-value interventions for both the uninsured and underinsured, we should acknowledge that our decisions redirect limited resources from others.
Is it the best use of $10,000 per month for a new drug that improves disease-free survival but not overall survival?
At the same time, we also have to remain vigilant and reflect on whether we are echoing the marketing messages of the companies selling these treatments. Having recently watched the excellent Hulu series Dopesick, which realistically portrays the medical community’s egregious overuse of Oxycontin at the behest of Purdue Pharmaceuticals, it is striking to see how effectively the pharmaceutical industry can co-opt stakeholders. Very few physicians or patients have expertise in health care policy with broad societal perspective, yet subspecialists offer edicts as if society should dedicate unlimited resources first and foremost to our career focus or personal cause.
I certainly appreciate the appeal of surrogate endpoints in a world in which we hope to offer novel therapies to patients in a timely fashion. In the next few years, some of our most promising data in oncology will demand that we consider whether surrogate endpoints are practice-changing. We are facing a fundamental question: Are we using these surrogate endpoints to predict overall survival or quality of life or do these endpoints stand on their own as practice-changing metrics?
We need to acknowledge that our primary clinical focus is not the only one that deserves our attention, particularly when our treatment decisions are, in fact, spending other people’s money. We should be asking not whether we prefer to deliver good news after a scan, but whether that alone is enough to justify the high cost of a new treatment without an overall survival benefit.
Dr. West disclosed serving as a director, officer, partner, employee, adviser, consultant, or trustee for Ariad/Takeda, Bristol-Myers Squibb, Boehringer Ingelheim, Spectrum, AstraZeneca, Celgene, Genentech/Roche, Pfizer, and Merck; serving as a speaker or a member of a speakers bureau for Ariad/Takeda, AstraZeneca, and Genentech/Roche; and receiving income from Eli Lilly. A version of this article first appeared on Medscape.com.
In the field of lung cancer, and more broadly in oncology, many of our biggest advances in 2021 have come as clinically meaningful improvements in surrogate endpoints – disease-free survival, progression-free survival, and sometimes even pathologic complete response rate.
I have historically been most compelled to consider new findings to be practice-changing when they improve overall survival or quality of life – the endpoints that translate to direct benefits for patients. However, I also feel it is appropriate to call surrogate endpoints practice-changing when they can predict improvements in overall survival or quality of life.
Take the PACIFIC trial, which assessed maintenance durvalumab after concurrent chemoradiation for unresectable stage III non–small cell lung cancer (NSCLC).
Back in 2017, I was initially unconvinced by the interim phase 3 data that were presented in a press release that highlighted the disease-free survival benefit. However, after examining additional data more closely, I saw the dramatic improvement in time to distant relapse or death was overwhelmingly likely to predict an improvement in overall survival – a benefit that the data subsequently bore out.
More recently, the disease-free survival results for adjuvant osimertinib in resected endothelial growth factor receptor mutation–positive NSCLC and adjuvant atezolizumab in resected programmed death-ligand 1–positive stage II-IIIA NSCLC have led to excitement about Food and Drug Administration approvals for these therapies. Although there is reason to be cautious about the likelihood of an overall survival benefit with either therapy – particularly for patients with low programmed death-ligand 1 who receive atezolizumab – I think that the results are promising enough to discuss these treatment options with appropriate patients.
Some argue, however, that overall survival is not necessarily a critical goal and that certain surrogate endpoints are inherently beneficial. Patients and oncologists may, for instance, view delaying disease progression as a win, even if overall survival remains the same.
I appreciate the view that favorable scan results are an achievement, even without a survival benefit. Patients appreciate the good news, and it is gratifying for us to deliver it. However, what remains unspoken is whether the benefit can be provided at a reasonable value given the financial costs associated with the new treatment.
In the United States, we consider the physician-patient relationship to be autonomous and even revered, but we conveniently ignore the fact that both are deciding on treatments that are funded by people who are not represented in the room. And in a health care system that fails to cover basic cancer care needs as well as other critical, high-value interventions for both the uninsured and underinsured, we should acknowledge that our decisions redirect limited resources from others.
Is it the best use of $10,000 per month for a new drug that improves disease-free survival but not overall survival?
At the same time, we also have to remain vigilant and reflect on whether we are echoing the marketing messages of the companies selling these treatments. Having recently watched the excellent Hulu series Dopesick, which realistically portrays the medical community’s egregious overuse of Oxycontin at the behest of Purdue Pharmaceuticals, it is striking to see how effectively the pharmaceutical industry can co-opt stakeholders. Very few physicians or patients have expertise in health care policy with broad societal perspective, yet subspecialists offer edicts as if society should dedicate unlimited resources first and foremost to our career focus or personal cause.
I certainly appreciate the appeal of surrogate endpoints in a world in which we hope to offer novel therapies to patients in a timely fashion. In the next few years, some of our most promising data in oncology will demand that we consider whether surrogate endpoints are practice-changing. We are facing a fundamental question: Are we using these surrogate endpoints to predict overall survival or quality of life or do these endpoints stand on their own as practice-changing metrics?
We need to acknowledge that our primary clinical focus is not the only one that deserves our attention, particularly when our treatment decisions are, in fact, spending other people’s money. We should be asking not whether we prefer to deliver good news after a scan, but whether that alone is enough to justify the high cost of a new treatment without an overall survival benefit.
Dr. West disclosed serving as a director, officer, partner, employee, adviser, consultant, or trustee for Ariad/Takeda, Bristol-Myers Squibb, Boehringer Ingelheim, Spectrum, AstraZeneca, Celgene, Genentech/Roche, Pfizer, and Merck; serving as a speaker or a member of a speakers bureau for Ariad/Takeda, AstraZeneca, and Genentech/Roche; and receiving income from Eli Lilly. A version of this article first appeared on Medscape.com.