User login
Immune checkpoint inhibitors don’t increase COVID-19 incidence or mortality, studies suggest
Cytokine storm plays a major role in the pathogenesis of COVID-19, according to research published in The Lancet Respiratory Medicine. This has generated concern about using ICIs during the pandemic, given their immunostimulatory activity and the risk of immune-related adverse effects.
However, two retrospective studies suggest ICIs do not increase the risk of developing COVID-19 or dying from the disease.
In a study of 1,545 cancer patients prescribed ICIs and 20,418 matched controls, the incidence of COVID-19 was 1.4% with ICI therapy and 1.0% without it (odds ratio, 1.38; P = .15).
In a case-control study of 50 patients with cancer and COVID-19, 28% of patients who had received ICIs died from COVID-19, compared with 36% of patients who had not received ICIs (OR, 0.36; P = .23).
Vartan Pahalyants and Kevin Tyan, both students in Harvard University’s joint MD/MBA program in Boston, presented these studies at the meeting.
COVID-19 incidence with ICIs
Mr. Pahalyants and colleagues analyzed data from cancer patients treated in the Mass General Brigham health care system. The researchers compared 1,545 patients with at least one ICI prescription between July 1, 2019, and Feb. 29, 2020, with 20,418 matched cancer patients not prescribed ICIs. The team assessed COVID-19 incidence based on positive test results through June 19, 2020, from public health data.
The incidence of COVID-19 was low in both groups – 1.4% in the ICI group and 1.0% in the matched control group (P = .16). Among COVID-19–positive patients, the all-cause death rate was 40.9% in the ICI group and 28.6% in the control group (P = .23).
In multivariate analysis, patients prescribed ICIs did not have a significantly elevated risk for COVID-19 relative to peers not prescribed ICIs (OR, 1.38; P = .15). However, risk was significantly increased for female patients (OR, 1.74; P < .001), those living in a town or county with higher COVID-19 positivity rate (OR, 1.59; P < .001), and those with severe comorbidity (vs. mild or moderate; OR, 9.77; P = .02).
Among COVID-19–positive patients, those prescribed ICIs did not have a significantly elevated risk for all-cause mortality (OR, 1.60; P = .71), but male sex and lower income were associated with an increased risk of death.
“We did not identify an increased risk of [COVID-19] diagnosis among patients prescribed ICIs compared to the controls,” Mr. Pahalyants said. “This information may assist patients and their providers in decision-making around continuation of therapy during this protracted pandemic. However, more research needs to be conducted to determine potential behavioral and testing factors that may have affected COVID-19 diagnosis susceptibility among patients included in the study.”
COVID-19 mortality with ICIs
For their study, Mr. Tyan and colleagues identified 25 cancer patients who had received ICIs in the year before a COVID-19 diagnosis between March 20, 2020, and June 3, 2020, at the Dana-Farber Cancer Institute and Mass General Brigham network. The researchers then matched each patient with a cancer patient having a COVID-19 diagnosis who had not received ICIs during the preceding year.
Overall, 28% of patients who had received ICIs before their COVID-19 diagnosis died from COVID-19, compared with 36% of those who had not received ICIs.
In multivariate analysis, ICI therapy did not predict COVID-19 mortality (OR, 0.36; P = .23). However, the risk of death from COVID-19 increased with age (OR, 1.14; P = .01) and for patients with chronic obstructive pulmonary disease (OR, 12.26; P = .01), and risk was lower for statin users (OR, 0.08; P = .02). Findings were similar in an analysis restricted to hospitalized patients in the ICI group and their matched controls.
Two ICI-treated patients with COVID-19 had persistent immune-related adverse events (hypophysitis in both cases), and one ICI-treated patient developed a new immune-related adverse event (hypothyroidism).
At COVID-19 presentation, relative to counterparts who had not received ICIs, patients who had received ICIs had higher platelet counts (P = .017) and higher D-dimer levels (P = .037). In the context of similar levels of other biomarkers, this finding is “of unclear significance, as all deaths in the cohort were due to respiratory failure as opposed to hypercoagulability,” Mr. Tyan said.
The patients treated with ICIs were more likely to die from COVID-19 if they had elevated troponin levels (P = .01), whereas no such association was seen for those not treated with ICIs.
“We found that ICI therapy is not associated with greater risk for COVID-19 mortality. Our period of follow-up was relatively short, but we did not observe a high incidence of new or persistent immune-related adverse events among our patients taking ICIs,” Mr. Tyan said.
“While larger prospective trials are needed to evaluate long-term safety in the context of COVID-19 infection, our findings support the continuation of ICI therapy during the pandemic as it does not appear to worsen outcomes for cancer patients,” he concluded.
ICI therapy can continue, with precautions
“The question of susceptibility to COVID-19 has been unclear as ICIs do not necessarily cause immunosuppression but certainly result in modulation of a patient’s immune system,” said Deborah Doroshow, MD, PhD, assistant professor at the Tisch Cancer Institute Icahn School of Medicine at Mount Sinai, New York. She was not involved in these studies.
“The findings of the study by Pahalyants and colleagues, which used a very large sample size, appear to convincingly demonstrate that ICI receipt is not associated with an increased susceptibility to COVID-19,” Dr. Doroshow said in an interview.
However, the findings of the study by Tyan and colleagues are more “thought-provoking,” Dr. Doroshow said. She noted that a large study published in Nature Medicine showed previous ICI therapy in cancer patients with COVID-19 increased the risk for hospitalization or severe COVID-19 requiring high-flow oxygen or mechanical ventilation. The new study was much smaller and did not perform statistical comparisons for outcomes such as oxygen requirements.
“I would feel comfortable telling patients that the data suggests that ICI treatment does not increase their risk of COVID-19. However, if they were to be diagnosed with COVID-19, it is unclear whether their previous ICI treatment increases their risk for poor outcomes,” Dr. Doroshow said.
“I would feel comfortable continuing to treat patients with ICIs at this time, but because we know that patients with cancer are generally more likely to develop COVID-19 and have poor outcomes, it is critical that our patients be educated about social distancing and mask wearing to the extent that their living and working situations permit,” she added.
Mr. Pahalyants disclosed no relevant conflicts of interest, and his study did not receive any specific funding. Mr. Tyan disclosed that he is cofounder and chief science officer of Kinnos, and his study did not receive any specific funding. Dr. Doroshow disclosed no relevant conflicts of interest.
SOURCE: Pahalyants V et al. SITC 2020, Abstract 826. Tyan K et al. SITC 2020, Abstract 481.
Cytokine storm plays a major role in the pathogenesis of COVID-19, according to research published in The Lancet Respiratory Medicine. This has generated concern about using ICIs during the pandemic, given their immunostimulatory activity and the risk of immune-related adverse effects.
However, two retrospective studies suggest ICIs do not increase the risk of developing COVID-19 or dying from the disease.
In a study of 1,545 cancer patients prescribed ICIs and 20,418 matched controls, the incidence of COVID-19 was 1.4% with ICI therapy and 1.0% without it (odds ratio, 1.38; P = .15).
In a case-control study of 50 patients with cancer and COVID-19, 28% of patients who had received ICIs died from COVID-19, compared with 36% of patients who had not received ICIs (OR, 0.36; P = .23).
Vartan Pahalyants and Kevin Tyan, both students in Harvard University’s joint MD/MBA program in Boston, presented these studies at the meeting.
COVID-19 incidence with ICIs
Mr. Pahalyants and colleagues analyzed data from cancer patients treated in the Mass General Brigham health care system. The researchers compared 1,545 patients with at least one ICI prescription between July 1, 2019, and Feb. 29, 2020, with 20,418 matched cancer patients not prescribed ICIs. The team assessed COVID-19 incidence based on positive test results through June 19, 2020, from public health data.
The incidence of COVID-19 was low in both groups – 1.4% in the ICI group and 1.0% in the matched control group (P = .16). Among COVID-19–positive patients, the all-cause death rate was 40.9% in the ICI group and 28.6% in the control group (P = .23).
In multivariate analysis, patients prescribed ICIs did not have a significantly elevated risk for COVID-19 relative to peers not prescribed ICIs (OR, 1.38; P = .15). However, risk was significantly increased for female patients (OR, 1.74; P < .001), those living in a town or county with higher COVID-19 positivity rate (OR, 1.59; P < .001), and those with severe comorbidity (vs. mild or moderate; OR, 9.77; P = .02).
Among COVID-19–positive patients, those prescribed ICIs did not have a significantly elevated risk for all-cause mortality (OR, 1.60; P = .71), but male sex and lower income were associated with an increased risk of death.
“We did not identify an increased risk of [COVID-19] diagnosis among patients prescribed ICIs compared to the controls,” Mr. Pahalyants said. “This information may assist patients and their providers in decision-making around continuation of therapy during this protracted pandemic. However, more research needs to be conducted to determine potential behavioral and testing factors that may have affected COVID-19 diagnosis susceptibility among patients included in the study.”
COVID-19 mortality with ICIs
For their study, Mr. Tyan and colleagues identified 25 cancer patients who had received ICIs in the year before a COVID-19 diagnosis between March 20, 2020, and June 3, 2020, at the Dana-Farber Cancer Institute and Mass General Brigham network. The researchers then matched each patient with a cancer patient having a COVID-19 diagnosis who had not received ICIs during the preceding year.
Overall, 28% of patients who had received ICIs before their COVID-19 diagnosis died from COVID-19, compared with 36% of those who had not received ICIs.
In multivariate analysis, ICI therapy did not predict COVID-19 mortality (OR, 0.36; P = .23). However, the risk of death from COVID-19 increased with age (OR, 1.14; P = .01) and for patients with chronic obstructive pulmonary disease (OR, 12.26; P = .01), and risk was lower for statin users (OR, 0.08; P = .02). Findings were similar in an analysis restricted to hospitalized patients in the ICI group and their matched controls.
Two ICI-treated patients with COVID-19 had persistent immune-related adverse events (hypophysitis in both cases), and one ICI-treated patient developed a new immune-related adverse event (hypothyroidism).
At COVID-19 presentation, relative to counterparts who had not received ICIs, patients who had received ICIs had higher platelet counts (P = .017) and higher D-dimer levels (P = .037). In the context of similar levels of other biomarkers, this finding is “of unclear significance, as all deaths in the cohort were due to respiratory failure as opposed to hypercoagulability,” Mr. Tyan said.
The patients treated with ICIs were more likely to die from COVID-19 if they had elevated troponin levels (P = .01), whereas no such association was seen for those not treated with ICIs.
“We found that ICI therapy is not associated with greater risk for COVID-19 mortality. Our period of follow-up was relatively short, but we did not observe a high incidence of new or persistent immune-related adverse events among our patients taking ICIs,” Mr. Tyan said.
“While larger prospective trials are needed to evaluate long-term safety in the context of COVID-19 infection, our findings support the continuation of ICI therapy during the pandemic as it does not appear to worsen outcomes for cancer patients,” he concluded.
ICI therapy can continue, with precautions
“The question of susceptibility to COVID-19 has been unclear as ICIs do not necessarily cause immunosuppression but certainly result in modulation of a patient’s immune system,” said Deborah Doroshow, MD, PhD, assistant professor at the Tisch Cancer Institute Icahn School of Medicine at Mount Sinai, New York. She was not involved in these studies.
“The findings of the study by Pahalyants and colleagues, which used a very large sample size, appear to convincingly demonstrate that ICI receipt is not associated with an increased susceptibility to COVID-19,” Dr. Doroshow said in an interview.
However, the findings of the study by Tyan and colleagues are more “thought-provoking,” Dr. Doroshow said. She noted that a large study published in Nature Medicine showed previous ICI therapy in cancer patients with COVID-19 increased the risk for hospitalization or severe COVID-19 requiring high-flow oxygen or mechanical ventilation. The new study was much smaller and did not perform statistical comparisons for outcomes such as oxygen requirements.
“I would feel comfortable telling patients that the data suggests that ICI treatment does not increase their risk of COVID-19. However, if they were to be diagnosed with COVID-19, it is unclear whether their previous ICI treatment increases their risk for poor outcomes,” Dr. Doroshow said.
“I would feel comfortable continuing to treat patients with ICIs at this time, but because we know that patients with cancer are generally more likely to develop COVID-19 and have poor outcomes, it is critical that our patients be educated about social distancing and mask wearing to the extent that their living and working situations permit,” she added.
Mr. Pahalyants disclosed no relevant conflicts of interest, and his study did not receive any specific funding. Mr. Tyan disclosed that he is cofounder and chief science officer of Kinnos, and his study did not receive any specific funding. Dr. Doroshow disclosed no relevant conflicts of interest.
SOURCE: Pahalyants V et al. SITC 2020, Abstract 826. Tyan K et al. SITC 2020, Abstract 481.
Cytokine storm plays a major role in the pathogenesis of COVID-19, according to research published in The Lancet Respiratory Medicine. This has generated concern about using ICIs during the pandemic, given their immunostimulatory activity and the risk of immune-related adverse effects.
However, two retrospective studies suggest ICIs do not increase the risk of developing COVID-19 or dying from the disease.
In a study of 1,545 cancer patients prescribed ICIs and 20,418 matched controls, the incidence of COVID-19 was 1.4% with ICI therapy and 1.0% without it (odds ratio, 1.38; P = .15).
In a case-control study of 50 patients with cancer and COVID-19, 28% of patients who had received ICIs died from COVID-19, compared with 36% of patients who had not received ICIs (OR, 0.36; P = .23).
Vartan Pahalyants and Kevin Tyan, both students in Harvard University’s joint MD/MBA program in Boston, presented these studies at the meeting.
COVID-19 incidence with ICIs
Mr. Pahalyants and colleagues analyzed data from cancer patients treated in the Mass General Brigham health care system. The researchers compared 1,545 patients with at least one ICI prescription between July 1, 2019, and Feb. 29, 2020, with 20,418 matched cancer patients not prescribed ICIs. The team assessed COVID-19 incidence based on positive test results through June 19, 2020, from public health data.
The incidence of COVID-19 was low in both groups – 1.4% in the ICI group and 1.0% in the matched control group (P = .16). Among COVID-19–positive patients, the all-cause death rate was 40.9% in the ICI group and 28.6% in the control group (P = .23).
In multivariate analysis, patients prescribed ICIs did not have a significantly elevated risk for COVID-19 relative to peers not prescribed ICIs (OR, 1.38; P = .15). However, risk was significantly increased for female patients (OR, 1.74; P < .001), those living in a town or county with higher COVID-19 positivity rate (OR, 1.59; P < .001), and those with severe comorbidity (vs. mild or moderate; OR, 9.77; P = .02).
Among COVID-19–positive patients, those prescribed ICIs did not have a significantly elevated risk for all-cause mortality (OR, 1.60; P = .71), but male sex and lower income were associated with an increased risk of death.
“We did not identify an increased risk of [COVID-19] diagnosis among patients prescribed ICIs compared to the controls,” Mr. Pahalyants said. “This information may assist patients and their providers in decision-making around continuation of therapy during this protracted pandemic. However, more research needs to be conducted to determine potential behavioral and testing factors that may have affected COVID-19 diagnosis susceptibility among patients included in the study.”
COVID-19 mortality with ICIs
For their study, Mr. Tyan and colleagues identified 25 cancer patients who had received ICIs in the year before a COVID-19 diagnosis between March 20, 2020, and June 3, 2020, at the Dana-Farber Cancer Institute and Mass General Brigham network. The researchers then matched each patient with a cancer patient having a COVID-19 diagnosis who had not received ICIs during the preceding year.
Overall, 28% of patients who had received ICIs before their COVID-19 diagnosis died from COVID-19, compared with 36% of those who had not received ICIs.
In multivariate analysis, ICI therapy did not predict COVID-19 mortality (OR, 0.36; P = .23). However, the risk of death from COVID-19 increased with age (OR, 1.14; P = .01) and for patients with chronic obstructive pulmonary disease (OR, 12.26; P = .01), and risk was lower for statin users (OR, 0.08; P = .02). Findings were similar in an analysis restricted to hospitalized patients in the ICI group and their matched controls.
Two ICI-treated patients with COVID-19 had persistent immune-related adverse events (hypophysitis in both cases), and one ICI-treated patient developed a new immune-related adverse event (hypothyroidism).
At COVID-19 presentation, relative to counterparts who had not received ICIs, patients who had received ICIs had higher platelet counts (P = .017) and higher D-dimer levels (P = .037). In the context of similar levels of other biomarkers, this finding is “of unclear significance, as all deaths in the cohort were due to respiratory failure as opposed to hypercoagulability,” Mr. Tyan said.
The patients treated with ICIs were more likely to die from COVID-19 if they had elevated troponin levels (P = .01), whereas no such association was seen for those not treated with ICIs.
“We found that ICI therapy is not associated with greater risk for COVID-19 mortality. Our period of follow-up was relatively short, but we did not observe a high incidence of new or persistent immune-related adverse events among our patients taking ICIs,” Mr. Tyan said.
“While larger prospective trials are needed to evaluate long-term safety in the context of COVID-19 infection, our findings support the continuation of ICI therapy during the pandemic as it does not appear to worsen outcomes for cancer patients,” he concluded.
ICI therapy can continue, with precautions
“The question of susceptibility to COVID-19 has been unclear as ICIs do not necessarily cause immunosuppression but certainly result in modulation of a patient’s immune system,” said Deborah Doroshow, MD, PhD, assistant professor at the Tisch Cancer Institute Icahn School of Medicine at Mount Sinai, New York. She was not involved in these studies.
“The findings of the study by Pahalyants and colleagues, which used a very large sample size, appear to convincingly demonstrate that ICI receipt is not associated with an increased susceptibility to COVID-19,” Dr. Doroshow said in an interview.
However, the findings of the study by Tyan and colleagues are more “thought-provoking,” Dr. Doroshow said. She noted that a large study published in Nature Medicine showed previous ICI therapy in cancer patients with COVID-19 increased the risk for hospitalization or severe COVID-19 requiring high-flow oxygen or mechanical ventilation. The new study was much smaller and did not perform statistical comparisons for outcomes such as oxygen requirements.
“I would feel comfortable telling patients that the data suggests that ICI treatment does not increase their risk of COVID-19. However, if they were to be diagnosed with COVID-19, it is unclear whether their previous ICI treatment increases their risk for poor outcomes,” Dr. Doroshow said.
“I would feel comfortable continuing to treat patients with ICIs at this time, but because we know that patients with cancer are generally more likely to develop COVID-19 and have poor outcomes, it is critical that our patients be educated about social distancing and mask wearing to the extent that their living and working situations permit,” she added.
Mr. Pahalyants disclosed no relevant conflicts of interest, and his study did not receive any specific funding. Mr. Tyan disclosed that he is cofounder and chief science officer of Kinnos, and his study did not receive any specific funding. Dr. Doroshow disclosed no relevant conflicts of interest.
SOURCE: Pahalyants V et al. SITC 2020, Abstract 826. Tyan K et al. SITC 2020, Abstract 481.
FROM SITC 2020
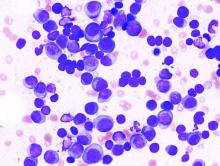
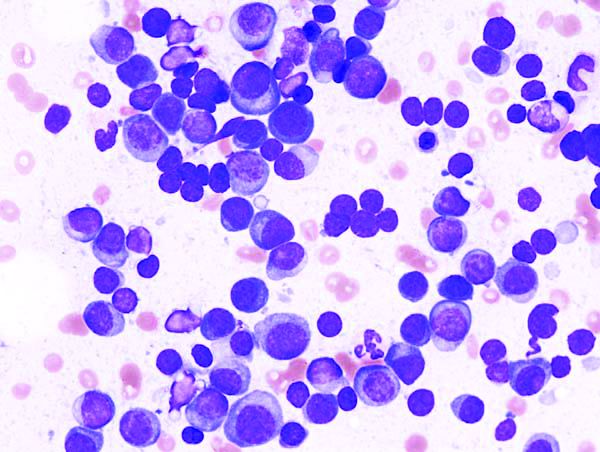
Circulating miRNA could be a potential biomarker for early diagnosis of MM
Circulating microRNAs (miRNAs) could be a potential noninvasive biomarker for early diagnosis of multiple myeloma (MM), according to the results of a meta-analysis published online in the Journal of Bone Oncology.
In recent years, because of the rise of the miRNA technique, many scholars have studied its value in the diagnosis of MM, and have obtained good but inconsistent results, according to Shuai-Shuai Gao, of the Xi’an (China) Daxing Hospital, and colleagues. For this reason, they conducted the meta-analysis in order to more clearly determine the role of miRNA in the early diagnosis of MM. The meta-analysis ultimately assessed 32 studies from 15 articles comprising 2,053 MM patients and 1,118 healthy controls.
All the included studies involved newly diagnosed MM patients and healthy controls; the obtained miRNA was derived from serum or plasma specimens; and the report contained relevant statistics such as sensitivity, specificity, and area-under-the-curve values.
High sensitivity and specificity
The researchers found that the overall sensitivity and specificity of using miRNAs for the diagnosis of MM were 0.81 and 0.85, respectively. In addition, the overall positive likelihood ratio, negative likelihood ratio, diagnostic odds ratio, and area under the curve were 5.5, 0.22, 25 and 0.90, respectively.
A subgroup analysis showed that the down-regulation of miRNA clusters with larger samples size of plasma type could carry out a better diagnostic accuracy of MM patients, according to the authors.
“[Circulating miRNAs] not only had high sensitivity and strong specificity, but also had noninvasive and no radiation risks. It is worth continuing to optimize its practicality. In the future, multicenter, more rigorous, and high-quality case-control studies are still needed in clinical practice to improve the efficacy of circulating miRNA in the early diagnosis of MM,” the researchers concluded.
The study did not receive any outside funding and the researchers reported that they had no conflicts.
SOURCE: Gao S-S et al. J Bone Oncol. 2020 Oct 21. doi: 10.1016/j.jbo.2020.100327.
Circulating microRNAs (miRNAs) could be a potential noninvasive biomarker for early diagnosis of multiple myeloma (MM), according to the results of a meta-analysis published online in the Journal of Bone Oncology.
In recent years, because of the rise of the miRNA technique, many scholars have studied its value in the diagnosis of MM, and have obtained good but inconsistent results, according to Shuai-Shuai Gao, of the Xi’an (China) Daxing Hospital, and colleagues. For this reason, they conducted the meta-analysis in order to more clearly determine the role of miRNA in the early diagnosis of MM. The meta-analysis ultimately assessed 32 studies from 15 articles comprising 2,053 MM patients and 1,118 healthy controls.
All the included studies involved newly diagnosed MM patients and healthy controls; the obtained miRNA was derived from serum or plasma specimens; and the report contained relevant statistics such as sensitivity, specificity, and area-under-the-curve values.
High sensitivity and specificity
The researchers found that the overall sensitivity and specificity of using miRNAs for the diagnosis of MM were 0.81 and 0.85, respectively. In addition, the overall positive likelihood ratio, negative likelihood ratio, diagnostic odds ratio, and area under the curve were 5.5, 0.22, 25 and 0.90, respectively.
A subgroup analysis showed that the down-regulation of miRNA clusters with larger samples size of plasma type could carry out a better diagnostic accuracy of MM patients, according to the authors.
“[Circulating miRNAs] not only had high sensitivity and strong specificity, but also had noninvasive and no radiation risks. It is worth continuing to optimize its practicality. In the future, multicenter, more rigorous, and high-quality case-control studies are still needed in clinical practice to improve the efficacy of circulating miRNA in the early diagnosis of MM,” the researchers concluded.
The study did not receive any outside funding and the researchers reported that they had no conflicts.
SOURCE: Gao S-S et al. J Bone Oncol. 2020 Oct 21. doi: 10.1016/j.jbo.2020.100327.
Circulating microRNAs (miRNAs) could be a potential noninvasive biomarker for early diagnosis of multiple myeloma (MM), according to the results of a meta-analysis published online in the Journal of Bone Oncology.
In recent years, because of the rise of the miRNA technique, many scholars have studied its value in the diagnosis of MM, and have obtained good but inconsistent results, according to Shuai-Shuai Gao, of the Xi’an (China) Daxing Hospital, and colleagues. For this reason, they conducted the meta-analysis in order to more clearly determine the role of miRNA in the early diagnosis of MM. The meta-analysis ultimately assessed 32 studies from 15 articles comprising 2,053 MM patients and 1,118 healthy controls.
All the included studies involved newly diagnosed MM patients and healthy controls; the obtained miRNA was derived from serum or plasma specimens; and the report contained relevant statistics such as sensitivity, specificity, and area-under-the-curve values.
High sensitivity and specificity
The researchers found that the overall sensitivity and specificity of using miRNAs for the diagnosis of MM were 0.81 and 0.85, respectively. In addition, the overall positive likelihood ratio, negative likelihood ratio, diagnostic odds ratio, and area under the curve were 5.5, 0.22, 25 and 0.90, respectively.
A subgroup analysis showed that the down-regulation of miRNA clusters with larger samples size of plasma type could carry out a better diagnostic accuracy of MM patients, according to the authors.
“[Circulating miRNAs] not only had high sensitivity and strong specificity, but also had noninvasive and no radiation risks. It is worth continuing to optimize its practicality. In the future, multicenter, more rigorous, and high-quality case-control studies are still needed in clinical practice to improve the efficacy of circulating miRNA in the early diagnosis of MM,” the researchers concluded.
The study did not receive any outside funding and the researchers reported that they had no conflicts.
SOURCE: Gao S-S et al. J Bone Oncol. 2020 Oct 21. doi: 10.1016/j.jbo.2020.100327.
FROM THE JOURNAL OF BONE ONCOLOGY
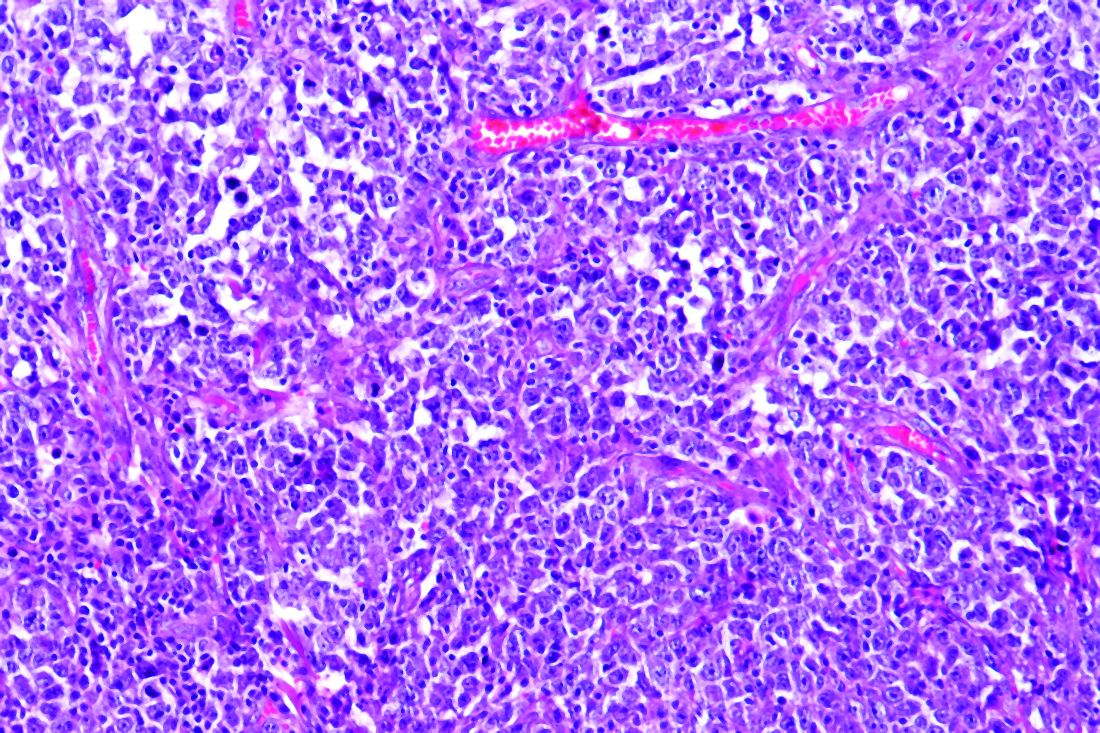
Pigment traits, sun sensitivity associated with risk of non-Hodgkin lymphomas and chronic lymphocytic leukemia
Risk factors for keratinocyte carcinomas, primarily pigment traits and sun sensitivity, were associated with the risk of developing non-Hodgkin lymphomas (NHL) and chronic lymphocytic leukemia (CLL) in an analysis of 92,097 women in France.
The presence of “many or very many nevi [moles]” was particularly associated with the risk of CLL among individuals in the E3N cohort, according to a report published online in Cancer Medicine. E3N is a prospective cohort of French women aged 40-65 years at inclusion in 1990. Researchers collected cancer data at baseline and every 2-3 years.
Hazard ratios and 95% confidence intervals for associations between patients pigmentary traits and sun exposure and their risk for CLL/NHL were estimated using Cox models, according to study author Louis-Marie Garcin, MD, of the Université Paris-Saclay, Villejuif, and colleagues.
Common etiology?
Among the 92,097 women included in the study, 622 incident cases of CLL/NHL were observed over a median of 24-years’ follow-up.
The presence of nevi was associated with CLL/NHL risk. The HR for “many or very many nevi” relative to “no nevi” was 1.56. The association with number of nevi was strongest for the risk of CLL, with an HR for “many or very many nevi” of 3.00 vs. 1.32 for NHL. In addition, the researchers found that women whose skin was highly sensitive to sunburn also had a higher risk of CLL (HR, 1.96), while no increased risk of NHL was observed. All HR values were within their respective 95% confidence intervals.
Relevant characteristics that were found to not be associated with added CLL/NHL risk were skin or hair color, number of freckles, and average daily UV dose during spring and summer in the location of residence at birth or at inclusion.
These observations suggest that CLL in particular may share some constitutional risk factors with keratinocyte cancers, according to the researchers.
“We report an association between nevi frequency and CLL/NHL risk, suggesting a partly common genetic etiology of these tumors. Future research should investigate common pathophysiological pathways that could promote the development of both skin carcinoma and CLL/NHL,” the researchers concluded.
The study was sponsored by the French government. The authors stated that they had no conflicts of interest.
SOURCE: Garcin L-M et al. Cancer Med. 2020. doi: 10.1002/cam4.3586.
Risk factors for keratinocyte carcinomas, primarily pigment traits and sun sensitivity, were associated with the risk of developing non-Hodgkin lymphomas (NHL) and chronic lymphocytic leukemia (CLL) in an analysis of 92,097 women in France.
The presence of “many or very many nevi [moles]” was particularly associated with the risk of CLL among individuals in the E3N cohort, according to a report published online in Cancer Medicine. E3N is a prospective cohort of French women aged 40-65 years at inclusion in 1990. Researchers collected cancer data at baseline and every 2-3 years.
Hazard ratios and 95% confidence intervals for associations between patients pigmentary traits and sun exposure and their risk for CLL/NHL were estimated using Cox models, according to study author Louis-Marie Garcin, MD, of the Université Paris-Saclay, Villejuif, and colleagues.
Common etiology?
Among the 92,097 women included in the study, 622 incident cases of CLL/NHL were observed over a median of 24-years’ follow-up.
The presence of nevi was associated with CLL/NHL risk. The HR for “many or very many nevi” relative to “no nevi” was 1.56. The association with number of nevi was strongest for the risk of CLL, with an HR for “many or very many nevi” of 3.00 vs. 1.32 for NHL. In addition, the researchers found that women whose skin was highly sensitive to sunburn also had a higher risk of CLL (HR, 1.96), while no increased risk of NHL was observed. All HR values were within their respective 95% confidence intervals.
Relevant characteristics that were found to not be associated with added CLL/NHL risk were skin or hair color, number of freckles, and average daily UV dose during spring and summer in the location of residence at birth or at inclusion.
These observations suggest that CLL in particular may share some constitutional risk factors with keratinocyte cancers, according to the researchers.
“We report an association between nevi frequency and CLL/NHL risk, suggesting a partly common genetic etiology of these tumors. Future research should investigate common pathophysiological pathways that could promote the development of both skin carcinoma and CLL/NHL,” the researchers concluded.
The study was sponsored by the French government. The authors stated that they had no conflicts of interest.
SOURCE: Garcin L-M et al. Cancer Med. 2020. doi: 10.1002/cam4.3586.
Risk factors for keratinocyte carcinomas, primarily pigment traits and sun sensitivity, were associated with the risk of developing non-Hodgkin lymphomas (NHL) and chronic lymphocytic leukemia (CLL) in an analysis of 92,097 women in France.
The presence of “many or very many nevi [moles]” was particularly associated with the risk of CLL among individuals in the E3N cohort, according to a report published online in Cancer Medicine. E3N is a prospective cohort of French women aged 40-65 years at inclusion in 1990. Researchers collected cancer data at baseline and every 2-3 years.
Hazard ratios and 95% confidence intervals for associations between patients pigmentary traits and sun exposure and their risk for CLL/NHL were estimated using Cox models, according to study author Louis-Marie Garcin, MD, of the Université Paris-Saclay, Villejuif, and colleagues.
Common etiology?
Among the 92,097 women included in the study, 622 incident cases of CLL/NHL were observed over a median of 24-years’ follow-up.
The presence of nevi was associated with CLL/NHL risk. The HR for “many or very many nevi” relative to “no nevi” was 1.56. The association with number of nevi was strongest for the risk of CLL, with an HR for “many or very many nevi” of 3.00 vs. 1.32 for NHL. In addition, the researchers found that women whose skin was highly sensitive to sunburn also had a higher risk of CLL (HR, 1.96), while no increased risk of NHL was observed. All HR values were within their respective 95% confidence intervals.
Relevant characteristics that were found to not be associated with added CLL/NHL risk were skin or hair color, number of freckles, and average daily UV dose during spring and summer in the location of residence at birth or at inclusion.
These observations suggest that CLL in particular may share some constitutional risk factors with keratinocyte cancers, according to the researchers.
“We report an association between nevi frequency and CLL/NHL risk, suggesting a partly common genetic etiology of these tumors. Future research should investigate common pathophysiological pathways that could promote the development of both skin carcinoma and CLL/NHL,” the researchers concluded.
The study was sponsored by the French government. The authors stated that they had no conflicts of interest.
SOURCE: Garcin L-M et al. Cancer Med. 2020. doi: 10.1002/cam4.3586.
FROM CANCER MEDICINE
Using telehealth to deliver palliative care to cancer patients
Traditional delivery of palliative care to outpatients with cancer is associated with many challenges.
Telehealth can eliminate some of these challenges but comes with issues of its own, according to results of the REACH PC trial.
Jennifer S. Temel, MD, of Massachusetts General Hospital in Boston, discussed the use of telemedicine in palliative care, including results from REACH PC, during an educational session at the ASCO Virtual Quality Care Symposium 2020.
Dr. Temel noted that, for cancer patients, an in-person visit with a palliative care specialist can cost time, induce fatigue, and increase financial burden from transportation and parking expenses.
For caregivers and family, an in-person visit may necessitate absence from family and/or work, require complex scheduling to coordinate with other office visits, and result in additional transportation and/or parking expenses.
For health care systems, to have a dedicated palliative care clinic requires precious space and financial expenditures for office personnel and other resources.
These issues make it attractive to consider whether telehealth could be used for palliative care services.
Scarcity of palliative care specialists
In the United States, there is roughly 1 palliative care physician for every 20,000 older adults with a life-limiting illness, according to research published in Annual Review of Public Health in 2014.
In its 2019 state-by-state report card, the Center to Advance Palliative Care noted that only 72% of U.S. hospitals with 50 or more beds have a palliative care team.
For patients with serious illnesses and those who are socioeconomically or geographically disadvantaged, palliative care is often inaccessible.
Inefficiencies in the current system are an additional impediment. Palliative care specialists frequently see patients during a portion of the patient’s routine visit to subspecialty or primary care clinics. This limits the palliative care specialist’s ability to perform comprehensive assessments and provide patient-centered care efficiently.
Special considerations regarding telehealth for palliative care
As a specialty, palliative care involves interactions that could make the use of telehealth problematic. For example, conveyance of interest, warmth, and touch are challenging or impossible in a video format.
Palliative care specialists engage with patients regarding relatively serious topics such as prognosis and end-of-life preferences. There is uncertainty about how those discussions would be received by patients and their caregivers via video.
Furthermore, there are logistical impediments such as prescribing opioids with video or across state lines.
Despite these concerns, the ENABLE study showed that supplementing usual oncology care with weekly (transitioning to monthly) telephone-based educational palliative care produced higher quality of life and mood than did usual oncology care alone. These results were published in JAMA in 2009.
REACH PC study demonstrates feasibility of telehealth model
Dr. Temel described the ongoing REACH PC trial in which palliative care is delivered via video visits and compared with in-person palliative care for patients with advanced non–small cell lung cancer.
The primary aim of REACH PC is to determine whether telehealth palliative care is equivalent to traditional palliative care in improving quality of life as a supplement to routine oncology care.
Currently, REACH PC has enrolled 581 patients at its 20 sites, spanning a geographically diverse area. Just over half of patients approached about REACH PC agreed to enroll in it. Ultimately, 1,250 enrollees are sought.
Among patients who declined to participate, 7.6% indicated “discomfort with technology” as the reason. Most refusals were due to lack of interest in research (35.1%) and/or palliative care (22.9%).
Older adults were prominent among enrollees. More than 60% were older than 60 years of age, and more than one-third were older than 70 years.
Among patients who began the trial, there were slightly more withdrawals in the telehealth participants, in comparison with in-person participants (13.6% versus 9.1%).
When palliative care clinicians were queried about video visits, 64.3% said there were no challenges. This is comparable to the 65.5% of clinicians who had no challenges with in-person visits.
When problems occurred with video visits, they were most frequently technical (19.1%). Only 1.4% of clinicians reported difficulty addressing topics that felt uncomfortable over video, and 1.5% reported difficulty establishing rapport.
The success rates of video and in-person visits were similar. About 80% of visits accomplished planned goals.
‘Webside’ manner
Strategies such as reflective listening and summarizing what patients say (to verify an accurate understanding of the patient’s perspective) are key to successful palliative care visits, regardless of the setting.
For telehealth visits, Dr. Temel described techniques she defined as “webside manner,” to compensate for the inability of the clinician to touch a patient. These techniques include leaning in toward the camera, nodding, and pausing to be certain the patient has finished speaking before the clinician speaks again.
Is telehealth the future of palliative care?
I include myself among those oncologists who have voiced concern about moving from face-to-face to remote visits for complicated consultations such as those required for palliative care. Nonetheless, from the preliminary results of the REACH PC trial, it appears that telehealth could be a valuable tool.
To minimize differences between in-person and remote delivery of palliative care, practical strategies for ensuring rapport and facilitating a trusting relationship should be defined further and disseminated.
In addition, we need to be vigilant for widening inequities of care from rapid movement to the use of technology (i.e., an equity gap). In their telehealth experience during the COVID-19 pandemic, investigators at Houston Methodist Cancer Center found that patients declining virtual visits tended to be older, lower-income, and less likely to have commercial insurance. These results were recently published in JCO Oncology Practice.
For the foregoing reasons, hybrid systems for palliative care services will probably always be needed.
Going forward, we should heed the advice of Alvin Toffler in his book Future Shock. Mr. Toffler said, “The illiterate of the 21st century will not be those who cannot read and write, but those who cannot learn, unlearn, and relearn.”
The traditional model for delivering palliative care will almost certainly need to be reimagined and relearned.
Dr. Temel disclosed institutional research funding from Pfizer.
Dr. Lyss was a community-based medical oncologist and clinical researcher for more than 35 years before his recent retirement. His clinical and research interests were focused on breast and lung cancers, as well as expanding clinical trial access to medically underserved populations. He is based in St. Louis. He has no conflicts of interest.
Traditional delivery of palliative care to outpatients with cancer is associated with many challenges.
Telehealth can eliminate some of these challenges but comes with issues of its own, according to results of the REACH PC trial.
Jennifer S. Temel, MD, of Massachusetts General Hospital in Boston, discussed the use of telemedicine in palliative care, including results from REACH PC, during an educational session at the ASCO Virtual Quality Care Symposium 2020.
Dr. Temel noted that, for cancer patients, an in-person visit with a palliative care specialist can cost time, induce fatigue, and increase financial burden from transportation and parking expenses.
For caregivers and family, an in-person visit may necessitate absence from family and/or work, require complex scheduling to coordinate with other office visits, and result in additional transportation and/or parking expenses.
For health care systems, to have a dedicated palliative care clinic requires precious space and financial expenditures for office personnel and other resources.
These issues make it attractive to consider whether telehealth could be used for palliative care services.
Scarcity of palliative care specialists
In the United States, there is roughly 1 palliative care physician for every 20,000 older adults with a life-limiting illness, according to research published in Annual Review of Public Health in 2014.
In its 2019 state-by-state report card, the Center to Advance Palliative Care noted that only 72% of U.S. hospitals with 50 or more beds have a palliative care team.
For patients with serious illnesses and those who are socioeconomically or geographically disadvantaged, palliative care is often inaccessible.
Inefficiencies in the current system are an additional impediment. Palliative care specialists frequently see patients during a portion of the patient’s routine visit to subspecialty or primary care clinics. This limits the palliative care specialist’s ability to perform comprehensive assessments and provide patient-centered care efficiently.
Special considerations regarding telehealth for palliative care
As a specialty, palliative care involves interactions that could make the use of telehealth problematic. For example, conveyance of interest, warmth, and touch are challenging or impossible in a video format.
Palliative care specialists engage with patients regarding relatively serious topics such as prognosis and end-of-life preferences. There is uncertainty about how those discussions would be received by patients and their caregivers via video.
Furthermore, there are logistical impediments such as prescribing opioids with video or across state lines.
Despite these concerns, the ENABLE study showed that supplementing usual oncology care with weekly (transitioning to monthly) telephone-based educational palliative care produced higher quality of life and mood than did usual oncology care alone. These results were published in JAMA in 2009.
REACH PC study demonstrates feasibility of telehealth model
Dr. Temel described the ongoing REACH PC trial in which palliative care is delivered via video visits and compared with in-person palliative care for patients with advanced non–small cell lung cancer.
The primary aim of REACH PC is to determine whether telehealth palliative care is equivalent to traditional palliative care in improving quality of life as a supplement to routine oncology care.
Currently, REACH PC has enrolled 581 patients at its 20 sites, spanning a geographically diverse area. Just over half of patients approached about REACH PC agreed to enroll in it. Ultimately, 1,250 enrollees are sought.
Among patients who declined to participate, 7.6% indicated “discomfort with technology” as the reason. Most refusals were due to lack of interest in research (35.1%) and/or palliative care (22.9%).
Older adults were prominent among enrollees. More than 60% were older than 60 years of age, and more than one-third were older than 70 years.
Among patients who began the trial, there were slightly more withdrawals in the telehealth participants, in comparison with in-person participants (13.6% versus 9.1%).
When palliative care clinicians were queried about video visits, 64.3% said there were no challenges. This is comparable to the 65.5% of clinicians who had no challenges with in-person visits.
When problems occurred with video visits, they were most frequently technical (19.1%). Only 1.4% of clinicians reported difficulty addressing topics that felt uncomfortable over video, and 1.5% reported difficulty establishing rapport.
The success rates of video and in-person visits were similar. About 80% of visits accomplished planned goals.
‘Webside’ manner
Strategies such as reflective listening and summarizing what patients say (to verify an accurate understanding of the patient’s perspective) are key to successful palliative care visits, regardless of the setting.
For telehealth visits, Dr. Temel described techniques she defined as “webside manner,” to compensate for the inability of the clinician to touch a patient. These techniques include leaning in toward the camera, nodding, and pausing to be certain the patient has finished speaking before the clinician speaks again.
Is telehealth the future of palliative care?
I include myself among those oncologists who have voiced concern about moving from face-to-face to remote visits for complicated consultations such as those required for palliative care. Nonetheless, from the preliminary results of the REACH PC trial, it appears that telehealth could be a valuable tool.
To minimize differences between in-person and remote delivery of palliative care, practical strategies for ensuring rapport and facilitating a trusting relationship should be defined further and disseminated.
In addition, we need to be vigilant for widening inequities of care from rapid movement to the use of technology (i.e., an equity gap). In their telehealth experience during the COVID-19 pandemic, investigators at Houston Methodist Cancer Center found that patients declining virtual visits tended to be older, lower-income, and less likely to have commercial insurance. These results were recently published in JCO Oncology Practice.
For the foregoing reasons, hybrid systems for palliative care services will probably always be needed.
Going forward, we should heed the advice of Alvin Toffler in his book Future Shock. Mr. Toffler said, “The illiterate of the 21st century will not be those who cannot read and write, but those who cannot learn, unlearn, and relearn.”
The traditional model for delivering palliative care will almost certainly need to be reimagined and relearned.
Dr. Temel disclosed institutional research funding from Pfizer.
Dr. Lyss was a community-based medical oncologist and clinical researcher for more than 35 years before his recent retirement. His clinical and research interests were focused on breast and lung cancers, as well as expanding clinical trial access to medically underserved populations. He is based in St. Louis. He has no conflicts of interest.
Traditional delivery of palliative care to outpatients with cancer is associated with many challenges.
Telehealth can eliminate some of these challenges but comes with issues of its own, according to results of the REACH PC trial.
Jennifer S. Temel, MD, of Massachusetts General Hospital in Boston, discussed the use of telemedicine in palliative care, including results from REACH PC, during an educational session at the ASCO Virtual Quality Care Symposium 2020.
Dr. Temel noted that, for cancer patients, an in-person visit with a palliative care specialist can cost time, induce fatigue, and increase financial burden from transportation and parking expenses.
For caregivers and family, an in-person visit may necessitate absence from family and/or work, require complex scheduling to coordinate with other office visits, and result in additional transportation and/or parking expenses.
For health care systems, to have a dedicated palliative care clinic requires precious space and financial expenditures for office personnel and other resources.
These issues make it attractive to consider whether telehealth could be used for palliative care services.
Scarcity of palliative care specialists
In the United States, there is roughly 1 palliative care physician for every 20,000 older adults with a life-limiting illness, according to research published in Annual Review of Public Health in 2014.
In its 2019 state-by-state report card, the Center to Advance Palliative Care noted that only 72% of U.S. hospitals with 50 or more beds have a palliative care team.
For patients with serious illnesses and those who are socioeconomically or geographically disadvantaged, palliative care is often inaccessible.
Inefficiencies in the current system are an additional impediment. Palliative care specialists frequently see patients during a portion of the patient’s routine visit to subspecialty or primary care clinics. This limits the palliative care specialist’s ability to perform comprehensive assessments and provide patient-centered care efficiently.
Special considerations regarding telehealth for palliative care
As a specialty, palliative care involves interactions that could make the use of telehealth problematic. For example, conveyance of interest, warmth, and touch are challenging or impossible in a video format.
Palliative care specialists engage with patients regarding relatively serious topics such as prognosis and end-of-life preferences. There is uncertainty about how those discussions would be received by patients and their caregivers via video.
Furthermore, there are logistical impediments such as prescribing opioids with video or across state lines.
Despite these concerns, the ENABLE study showed that supplementing usual oncology care with weekly (transitioning to monthly) telephone-based educational palliative care produced higher quality of life and mood than did usual oncology care alone. These results were published in JAMA in 2009.
REACH PC study demonstrates feasibility of telehealth model
Dr. Temel described the ongoing REACH PC trial in which palliative care is delivered via video visits and compared with in-person palliative care for patients with advanced non–small cell lung cancer.
The primary aim of REACH PC is to determine whether telehealth palliative care is equivalent to traditional palliative care in improving quality of life as a supplement to routine oncology care.
Currently, REACH PC has enrolled 581 patients at its 20 sites, spanning a geographically diverse area. Just over half of patients approached about REACH PC agreed to enroll in it. Ultimately, 1,250 enrollees are sought.
Among patients who declined to participate, 7.6% indicated “discomfort with technology” as the reason. Most refusals were due to lack of interest in research (35.1%) and/or palliative care (22.9%).
Older adults were prominent among enrollees. More than 60% were older than 60 years of age, and more than one-third were older than 70 years.
Among patients who began the trial, there were slightly more withdrawals in the telehealth participants, in comparison with in-person participants (13.6% versus 9.1%).
When palliative care clinicians were queried about video visits, 64.3% said there were no challenges. This is comparable to the 65.5% of clinicians who had no challenges with in-person visits.
When problems occurred with video visits, they were most frequently technical (19.1%). Only 1.4% of clinicians reported difficulty addressing topics that felt uncomfortable over video, and 1.5% reported difficulty establishing rapport.
The success rates of video and in-person visits were similar. About 80% of visits accomplished planned goals.
‘Webside’ manner
Strategies such as reflective listening and summarizing what patients say (to verify an accurate understanding of the patient’s perspective) are key to successful palliative care visits, regardless of the setting.
For telehealth visits, Dr. Temel described techniques she defined as “webside manner,” to compensate for the inability of the clinician to touch a patient. These techniques include leaning in toward the camera, nodding, and pausing to be certain the patient has finished speaking before the clinician speaks again.
Is telehealth the future of palliative care?
I include myself among those oncologists who have voiced concern about moving from face-to-face to remote visits for complicated consultations such as those required for palliative care. Nonetheless, from the preliminary results of the REACH PC trial, it appears that telehealth could be a valuable tool.
To minimize differences between in-person and remote delivery of palliative care, practical strategies for ensuring rapport and facilitating a trusting relationship should be defined further and disseminated.
In addition, we need to be vigilant for widening inequities of care from rapid movement to the use of technology (i.e., an equity gap). In their telehealth experience during the COVID-19 pandemic, investigators at Houston Methodist Cancer Center found that patients declining virtual visits tended to be older, lower-income, and less likely to have commercial insurance. These results were recently published in JCO Oncology Practice.
For the foregoing reasons, hybrid systems for palliative care services will probably always be needed.
Going forward, we should heed the advice of Alvin Toffler in his book Future Shock. Mr. Toffler said, “The illiterate of the 21st century will not be those who cannot read and write, but those who cannot learn, unlearn, and relearn.”
The traditional model for delivering palliative care will almost certainly need to be reimagined and relearned.
Dr. Temel disclosed institutional research funding from Pfizer.
Dr. Lyss was a community-based medical oncologist and clinical researcher for more than 35 years before his recent retirement. His clinical and research interests were focused on breast and lung cancers, as well as expanding clinical trial access to medically underserved populations. He is based in St. Louis. He has no conflicts of interest.
FROM ASCO QUALITY CARE SYMPOSIUM 2020
New cancer drugs may have saved more than 1.2 million Americans
Reductions in mortality were most notable for tumor types with relatively more approvals, including lung and breast cancer, melanoma, lymphoma, and leukemia.
A report from the American Cancer Society (ACS) estimated that, from 1991 to 2017, there were 2,902,200 total cancer deaths avoided from improvements in mortality from all potential sources.
The new findings, reported in the Journal of Medical Economics, suggest that drugs approved between 2000 and 2016 to treat the 15 most common cancer types helped to reduce mortality by 24% per 100,000 people.
“This study provides evidence that a significant share of that reduction from 2000 to 2016 was associated with the introduction of new therapies. The ACS report and other studies demonstrate that the improvements in lung cancer specifically are likely due to new treatments,” said lead study author Joanna P. MacEwan, MD, of PRECISIONheor in Los Angeles.
The findings contribute to a better understanding of whether increased spending on cancer drugs are worth the investment, according to the study authors.
“We provide evidence that the gains in survival measured in clinical trials are translating into health benefits for patients in the real world and confirm previous research that has also shown that new pharmaceutical treatments are associated with improved real-world survival outcomes for patients,” Dr. MacEwan said.
Full effect not yet observed
The researchers used a series of national data sets from sources including the Centers for Disease Control and Prevention; the U.S. Mortality Files by the National Center of Health Statistics; Survival, Epidemiology and End Results program; and United States Cancer Statistics.
The team calculated age-adjusted cancer mortality rates per year for the 15 most common tumor types and also looked at incident cases of cancer by tumor type, represented as per 100,000 people, for all ages, races, and genders.
The researchers then translated the change in cancer mortality in the U.S. from 2000 to 2016 associated with treatment stocks in each year into deaths averted per year.
Across the 16 years, mortality was down by 1,291,769 deaths. The following cancers had significant reductions in mortality: breast (n = 127,874), colorectal (n = 46,705), lung (n = 375,256), prostate (n = 476,210), gastric (n = 758), and renal (n = 739) cancers, as well as non-Hodgkin lymphoma (n = 48,836) and leukemia (n = 4,011).
Estimated mortality increased by 825 deaths in patients with thyroid cancer and 7,768 deaths for those with bladder cancer. These rises are likely due to the result of sparse drug approvals during this period – five for thyroid cancer and three for bladder cancer – Dr. MacEwan said. There were no approvals in liver or uterine cancer and few approvals in pancreatic and oral cancer.
The full effect of new drug introductions may not have been observed yet, Dr. MacEwan noted.
“There are fewer patients using the treatments for drugs approved in the later years of our study and less follow-up time to measure outcomes,” she said. “Over time, utilization of the newer therapies will likely increase and the full effect on mortality will be observed.”
Other factors at play
Multiple factors have led to the declines in mortality, said William G. Cance, MD, chief medical and scientific officer for the ACS, who was not involved in this study. “We are slowly sorting out the explanations in greater granularity.”
Dr. MacEwan said improved cancer screening may partially explain the decline in mortality in some tumor types.
“If screening in a particular tumor type improved during the study period and tumors were diagnosed earlier, then mortality for that tumor type may decline,” she said. “However, we did not find strong evidence to suggest that there were significant changes in screening during our study period. Breast cancer screening rates, for example, were stable over our study period.”
Cancer screening is not as strong an influence as it should be, Dr. Cance said.
“The lung cancer screening rate is low. In breast and colorectal cancers, we need to double down on earlier screening,” he said, noting that less than one-quarter of adults between ages 45 and 50 years are currently screened for colorectal cancer. The ACS recommends that people at average risk of colorectal cancer start regular screening at age 45.
More research is necessary to evaluate the relationship between drug approvals and cancer mortality, Dr. MacEwan said.
“Research directly linking utilization of new therapies to improved survival or reduced mortality in the real-world setting would more definitively demonstrate the impact of new treatments,” she said. “New therapies have improved outcomes for many patients and should continue to be considered as key elements of cancer treatment.”
“We need to continue to reduce tobacco smoking and improve on modifiable behaviors at the same time as we work on getting new drugs to cancer patients,” Dr. Cance said. “We are coming into an era of multiple new therapeutics, including targeted therapies, immunotherapies, and cellular therapies. Clinicians need to look closely at the trial data of new drugs and pay close attention to those that have the most mortality impact.”
“We also need equitable distribution of newer drugs,” Dr. Cance added. “They should be distributed to everybody who deserves them. Mortality is often impacted by social determinants of health.”
Funding for this research was provided by Pfizer. Study authors disclosed relationships, including employment, with Pfizer. Dr. Cance had no disclosures.
SOURCE: MacEwan JP et al. J Med Econ. 2020 Nov 9;1-12.
Reductions in mortality were most notable for tumor types with relatively more approvals, including lung and breast cancer, melanoma, lymphoma, and leukemia.
A report from the American Cancer Society (ACS) estimated that, from 1991 to 2017, there were 2,902,200 total cancer deaths avoided from improvements in mortality from all potential sources.
The new findings, reported in the Journal of Medical Economics, suggest that drugs approved between 2000 and 2016 to treat the 15 most common cancer types helped to reduce mortality by 24% per 100,000 people.
“This study provides evidence that a significant share of that reduction from 2000 to 2016 was associated with the introduction of new therapies. The ACS report and other studies demonstrate that the improvements in lung cancer specifically are likely due to new treatments,” said lead study author Joanna P. MacEwan, MD, of PRECISIONheor in Los Angeles.
The findings contribute to a better understanding of whether increased spending on cancer drugs are worth the investment, according to the study authors.
“We provide evidence that the gains in survival measured in clinical trials are translating into health benefits for patients in the real world and confirm previous research that has also shown that new pharmaceutical treatments are associated with improved real-world survival outcomes for patients,” Dr. MacEwan said.
Full effect not yet observed
The researchers used a series of national data sets from sources including the Centers for Disease Control and Prevention; the U.S. Mortality Files by the National Center of Health Statistics; Survival, Epidemiology and End Results program; and United States Cancer Statistics.
The team calculated age-adjusted cancer mortality rates per year for the 15 most common tumor types and also looked at incident cases of cancer by tumor type, represented as per 100,000 people, for all ages, races, and genders.
The researchers then translated the change in cancer mortality in the U.S. from 2000 to 2016 associated with treatment stocks in each year into deaths averted per year.
Across the 16 years, mortality was down by 1,291,769 deaths. The following cancers had significant reductions in mortality: breast (n = 127,874), colorectal (n = 46,705), lung (n = 375,256), prostate (n = 476,210), gastric (n = 758), and renal (n = 739) cancers, as well as non-Hodgkin lymphoma (n = 48,836) and leukemia (n = 4,011).
Estimated mortality increased by 825 deaths in patients with thyroid cancer and 7,768 deaths for those with bladder cancer. These rises are likely due to the result of sparse drug approvals during this period – five for thyroid cancer and three for bladder cancer – Dr. MacEwan said. There were no approvals in liver or uterine cancer and few approvals in pancreatic and oral cancer.
The full effect of new drug introductions may not have been observed yet, Dr. MacEwan noted.
“There are fewer patients using the treatments for drugs approved in the later years of our study and less follow-up time to measure outcomes,” she said. “Over time, utilization of the newer therapies will likely increase and the full effect on mortality will be observed.”
Other factors at play
Multiple factors have led to the declines in mortality, said William G. Cance, MD, chief medical and scientific officer for the ACS, who was not involved in this study. “We are slowly sorting out the explanations in greater granularity.”
Dr. MacEwan said improved cancer screening may partially explain the decline in mortality in some tumor types.
“If screening in a particular tumor type improved during the study period and tumors were diagnosed earlier, then mortality for that tumor type may decline,” she said. “However, we did not find strong evidence to suggest that there were significant changes in screening during our study period. Breast cancer screening rates, for example, were stable over our study period.”
Cancer screening is not as strong an influence as it should be, Dr. Cance said.
“The lung cancer screening rate is low. In breast and colorectal cancers, we need to double down on earlier screening,” he said, noting that less than one-quarter of adults between ages 45 and 50 years are currently screened for colorectal cancer. The ACS recommends that people at average risk of colorectal cancer start regular screening at age 45.
More research is necessary to evaluate the relationship between drug approvals and cancer mortality, Dr. MacEwan said.
“Research directly linking utilization of new therapies to improved survival or reduced mortality in the real-world setting would more definitively demonstrate the impact of new treatments,” she said. “New therapies have improved outcomes for many patients and should continue to be considered as key elements of cancer treatment.”
“We need to continue to reduce tobacco smoking and improve on modifiable behaviors at the same time as we work on getting new drugs to cancer patients,” Dr. Cance said. “We are coming into an era of multiple new therapeutics, including targeted therapies, immunotherapies, and cellular therapies. Clinicians need to look closely at the trial data of new drugs and pay close attention to those that have the most mortality impact.”
“We also need equitable distribution of newer drugs,” Dr. Cance added. “They should be distributed to everybody who deserves them. Mortality is often impacted by social determinants of health.”
Funding for this research was provided by Pfizer. Study authors disclosed relationships, including employment, with Pfizer. Dr. Cance had no disclosures.
SOURCE: MacEwan JP et al. J Med Econ. 2020 Nov 9;1-12.
Reductions in mortality were most notable for tumor types with relatively more approvals, including lung and breast cancer, melanoma, lymphoma, and leukemia.
A report from the American Cancer Society (ACS) estimated that, from 1991 to 2017, there were 2,902,200 total cancer deaths avoided from improvements in mortality from all potential sources.
The new findings, reported in the Journal of Medical Economics, suggest that drugs approved between 2000 and 2016 to treat the 15 most common cancer types helped to reduce mortality by 24% per 100,000 people.
“This study provides evidence that a significant share of that reduction from 2000 to 2016 was associated with the introduction of new therapies. The ACS report and other studies demonstrate that the improvements in lung cancer specifically are likely due to new treatments,” said lead study author Joanna P. MacEwan, MD, of PRECISIONheor in Los Angeles.
The findings contribute to a better understanding of whether increased spending on cancer drugs are worth the investment, according to the study authors.
“We provide evidence that the gains in survival measured in clinical trials are translating into health benefits for patients in the real world and confirm previous research that has also shown that new pharmaceutical treatments are associated with improved real-world survival outcomes for patients,” Dr. MacEwan said.
Full effect not yet observed
The researchers used a series of national data sets from sources including the Centers for Disease Control and Prevention; the U.S. Mortality Files by the National Center of Health Statistics; Survival, Epidemiology and End Results program; and United States Cancer Statistics.
The team calculated age-adjusted cancer mortality rates per year for the 15 most common tumor types and also looked at incident cases of cancer by tumor type, represented as per 100,000 people, for all ages, races, and genders.
The researchers then translated the change in cancer mortality in the U.S. from 2000 to 2016 associated with treatment stocks in each year into deaths averted per year.
Across the 16 years, mortality was down by 1,291,769 deaths. The following cancers had significant reductions in mortality: breast (n = 127,874), colorectal (n = 46,705), lung (n = 375,256), prostate (n = 476,210), gastric (n = 758), and renal (n = 739) cancers, as well as non-Hodgkin lymphoma (n = 48,836) and leukemia (n = 4,011).
Estimated mortality increased by 825 deaths in patients with thyroid cancer and 7,768 deaths for those with bladder cancer. These rises are likely due to the result of sparse drug approvals during this period – five for thyroid cancer and three for bladder cancer – Dr. MacEwan said. There were no approvals in liver or uterine cancer and few approvals in pancreatic and oral cancer.
The full effect of new drug introductions may not have been observed yet, Dr. MacEwan noted.
“There are fewer patients using the treatments for drugs approved in the later years of our study and less follow-up time to measure outcomes,” she said. “Over time, utilization of the newer therapies will likely increase and the full effect on mortality will be observed.”
Other factors at play
Multiple factors have led to the declines in mortality, said William G. Cance, MD, chief medical and scientific officer for the ACS, who was not involved in this study. “We are slowly sorting out the explanations in greater granularity.”
Dr. MacEwan said improved cancer screening may partially explain the decline in mortality in some tumor types.
“If screening in a particular tumor type improved during the study period and tumors were diagnosed earlier, then mortality for that tumor type may decline,” she said. “However, we did not find strong evidence to suggest that there were significant changes in screening during our study period. Breast cancer screening rates, for example, were stable over our study period.”
Cancer screening is not as strong an influence as it should be, Dr. Cance said.
“The lung cancer screening rate is low. In breast and colorectal cancers, we need to double down on earlier screening,” he said, noting that less than one-quarter of adults between ages 45 and 50 years are currently screened for colorectal cancer. The ACS recommends that people at average risk of colorectal cancer start regular screening at age 45.
More research is necessary to evaluate the relationship between drug approvals and cancer mortality, Dr. MacEwan said.
“Research directly linking utilization of new therapies to improved survival or reduced mortality in the real-world setting would more definitively demonstrate the impact of new treatments,” she said. “New therapies have improved outcomes for many patients and should continue to be considered as key elements of cancer treatment.”
“We need to continue to reduce tobacco smoking and improve on modifiable behaviors at the same time as we work on getting new drugs to cancer patients,” Dr. Cance said. “We are coming into an era of multiple new therapeutics, including targeted therapies, immunotherapies, and cellular therapies. Clinicians need to look closely at the trial data of new drugs and pay close attention to those that have the most mortality impact.”
“We also need equitable distribution of newer drugs,” Dr. Cance added. “They should be distributed to everybody who deserves them. Mortality is often impacted by social determinants of health.”
Funding for this research was provided by Pfizer. Study authors disclosed relationships, including employment, with Pfizer. Dr. Cance had no disclosures.
SOURCE: MacEwan JP et al. J Med Econ. 2020 Nov 9;1-12.
FROM JOURNAL OF MEDICAL ECONOMICS
Home care for bortezomib safe and reduces hospital visits in myeloma patients
Home administration of bortezomib (Velcade), as a once or twice-weekly subcutaneous self-injection is safe in patients with myeloma, significantly reducing hospital visits, and likely improving quality of life, a study shows.
The majority (43 of 52 patients) successfully self-administered bortezomib and completed the course. Also, hospital visits for those on the so-called Homecare programme reduced by 50%, with most visits comprising a fortnightly drug pickup from the drive-through pharmacy.
The work was presented as a poster by lead author and researcher, Kanchana De Abrew, hematology consultant at University Hospital Southampton NHS Foundation Trust, at this year’s virtual British Society of Haematology (BSH) meeting. De Abrew conducted the study while at Queen Alexandra Hospital, Portsmouth.
“We wanted to minimize patient visits to hospital because with travel time and waiting time, patients can easily find a visit takes up a whole morning, so this relates to their quality of life as well as having financial implications for patients,” Dr. De Abrew said in an interview. It also reduced the impact on day units and improved capacity for other services.
Dr. De Abrew noted that the study was conducted in the pre-COVID-19 era, but that the current enhanced threat of infection only served to reinforce the benefits of self-administration at home and avoiding unnecessary hospital visits.
“This project could easily be set up in other hospitals and some other centers have already contacted us about this. It might suit rural areas,” she added.
‘Safe and effective’
Dr. Matthew Jenner, consultant hematologist for University Hospital Southampton NHS Foundation Trust, who was not involved in the study, remarked that the study demonstrated another way to deliver bortezomib outside of hospital in addition to home care services that require trained nurses to administer treatment. “With a modest amount of training of the patient and family, it is both a safe and effective way of delivering treatment. This reduces hospital visits for the patient and frees up much needed capacity for heavily stretched chemotherapy units, creating space for other newer treatments that require hospital attendance.
“It is of benefit all round to both the patients undertaking self-administration and those who benefit from improved capacity,” added Dr. Jenner.
Avoiding hospital visits
Myeloma patients are already immunosuppressed prior to treatment and then this worsens once on treatment. Once they are sitting in a clinic environment they are surrounded by similarly immunosuppressed patients, so their risk is heightened further.
Figures suggest myeloma cases are on the increase. Annually, the United Kingdom sees around 5,800 new cases of myeloma and incidence increased by a significant 32% between the periods of 1993-1995 and 2015-2017. These figures were reflected in the patient numbers at the Queen Alexandra Hospital where the study was carried out. Many patients receive bortezomib, which forms the backbone of four National Institute for Health and Care Excellence (NICE) approved regimens.
“Patients are living longer so in the early 2000s patients had a life expectancy of 2-3 years, whereas now patients live for around 5 years. Also, the scope and lines of treatments have increased a lot. Over 50% of patients are likely to have bortezomib at some point in their management,” explained Dr. De Abrew.
Bortezomib is given once or twice weekly as a subcutaneous injection, and this usually continues for approximately 6-8 months with four to six cycles. Administering the drug in hospital requires around a half-hour slot placing considerable burden on the hematology day unit resources, and this can adversely affect the patient experience with waiting times and the need for frequent hospital visits.
Patient or relatives taught to self-administer at home
In 2017, clinical nurse specialists taught suitable patients to self-administer bortezomib in the Homecare protocol. Patients collected a 2-week supply of the drug. The protocol aimed to improve patient quality of life by reducing hospital visits, and increasing capacity in the hematology day unit. Since the start of the programme in 2017, the majority (71) of myeloma patients at Portsmouth have been treated through the Homecare program.
Dr. De Abrew conducted a retrospective review of patients who received bortezomib between January and October 2019 aimed at determining the effectiveness of the Homecare programme. To this end, she measured the proportion able to commence the Homecare protocol; the proportion successful in completing treatment on the Homecare protocol; the amount of additional clinical nurse specialist time required to support the Homecare protocol; and the number of associated adverse incidents.
A total of 52 bortezomib-treated patients were included in the study. Patients were excluded if they were on a different combination of drugs that required hospital visits, or inpatient care for other reasons. Three patients ceased the drug – two because of toxicity, and one because of rapid progression. The average age of patients was 74 years, and 55.8% were using bortezomib as first-line, 36.5% second-line, and the remainder third-line or more.
The vast majority started the Homecare protocol (45/52), and 25 self-administered and 17 received a relative’s help. A total of 43 completed the self-administration protocol with two reverting to hospital assistance. Bortezomib was given for four to six cycles lasting around 6-8 months.
Clinical nurse specialists trained 38 patients for home care, with an average training time of 43 minutes. Two of these patients were considered unsuitable for self-administration. The remainder were trained by ward nurses or did not require training having received bortezomib previously.
A total of 20 patients required additional clinical nurse specialist time requiring an average of 55 minutes. Of those requiring additional support: Seven needed retraining; two needed the first dose delivered by a nurse specialist; two requested help from the hematology unit; and nine wanted general extra support – for example, help with injection site queries (usually administered to the abdominal area), reassurance during administration, syringe queries, administrative queries, and queries around spillages.
“Importantly, patients always have the phone number of the nurse specialist at hand. But most people managed okay, and even if they needed additional support they still got there,” remarked Dr. De Abrew.
In terms of adverse events, there were six in total. These included three reported spillages (with no harm caused), and three experienced injection site incidents (rash, pain). “We found a low number of reported adverse events,” she said.
Dr. De Abrew added that generally, many more medications were being converted to subcutaneous formulations in myeloma and other hematology conditions. “Perhaps these results could inform self-administration of other drugs. In hematology, we get so many new drugs come through every year, but we don’t get the increased resources to manage this in the day units. Broadening self-administration could really help with capacity as well as improve quality of life for the patients.
“These results show that it can be done!” she said.
Dr. De Abrew declared no relevant conflicts of interest. Dr. Jenner declared receiving honoraria from Janssen, which manufactures branded Velcade (bortezomib).
A version of this story originally appeared on Medscape.com.
Home administration of bortezomib (Velcade), as a once or twice-weekly subcutaneous self-injection is safe in patients with myeloma, significantly reducing hospital visits, and likely improving quality of life, a study shows.
The majority (43 of 52 patients) successfully self-administered bortezomib and completed the course. Also, hospital visits for those on the so-called Homecare programme reduced by 50%, with most visits comprising a fortnightly drug pickup from the drive-through pharmacy.
The work was presented as a poster by lead author and researcher, Kanchana De Abrew, hematology consultant at University Hospital Southampton NHS Foundation Trust, at this year’s virtual British Society of Haematology (BSH) meeting. De Abrew conducted the study while at Queen Alexandra Hospital, Portsmouth.
“We wanted to minimize patient visits to hospital because with travel time and waiting time, patients can easily find a visit takes up a whole morning, so this relates to their quality of life as well as having financial implications for patients,” Dr. De Abrew said in an interview. It also reduced the impact on day units and improved capacity for other services.
Dr. De Abrew noted that the study was conducted in the pre-COVID-19 era, but that the current enhanced threat of infection only served to reinforce the benefits of self-administration at home and avoiding unnecessary hospital visits.
“This project could easily be set up in other hospitals and some other centers have already contacted us about this. It might suit rural areas,” she added.
‘Safe and effective’
Dr. Matthew Jenner, consultant hematologist for University Hospital Southampton NHS Foundation Trust, who was not involved in the study, remarked that the study demonstrated another way to deliver bortezomib outside of hospital in addition to home care services that require trained nurses to administer treatment. “With a modest amount of training of the patient and family, it is both a safe and effective way of delivering treatment. This reduces hospital visits for the patient and frees up much needed capacity for heavily stretched chemotherapy units, creating space for other newer treatments that require hospital attendance.
“It is of benefit all round to both the patients undertaking self-administration and those who benefit from improved capacity,” added Dr. Jenner.
Avoiding hospital visits
Myeloma patients are already immunosuppressed prior to treatment and then this worsens once on treatment. Once they are sitting in a clinic environment they are surrounded by similarly immunosuppressed patients, so their risk is heightened further.
Figures suggest myeloma cases are on the increase. Annually, the United Kingdom sees around 5,800 new cases of myeloma and incidence increased by a significant 32% between the periods of 1993-1995 and 2015-2017. These figures were reflected in the patient numbers at the Queen Alexandra Hospital where the study was carried out. Many patients receive bortezomib, which forms the backbone of four National Institute for Health and Care Excellence (NICE) approved regimens.
“Patients are living longer so in the early 2000s patients had a life expectancy of 2-3 years, whereas now patients live for around 5 years. Also, the scope and lines of treatments have increased a lot. Over 50% of patients are likely to have bortezomib at some point in their management,” explained Dr. De Abrew.
Bortezomib is given once or twice weekly as a subcutaneous injection, and this usually continues for approximately 6-8 months with four to six cycles. Administering the drug in hospital requires around a half-hour slot placing considerable burden on the hematology day unit resources, and this can adversely affect the patient experience with waiting times and the need for frequent hospital visits.
Patient or relatives taught to self-administer at home
In 2017, clinical nurse specialists taught suitable patients to self-administer bortezomib in the Homecare protocol. Patients collected a 2-week supply of the drug. The protocol aimed to improve patient quality of life by reducing hospital visits, and increasing capacity in the hematology day unit. Since the start of the programme in 2017, the majority (71) of myeloma patients at Portsmouth have been treated through the Homecare program.
Dr. De Abrew conducted a retrospective review of patients who received bortezomib between January and October 2019 aimed at determining the effectiveness of the Homecare programme. To this end, she measured the proportion able to commence the Homecare protocol; the proportion successful in completing treatment on the Homecare protocol; the amount of additional clinical nurse specialist time required to support the Homecare protocol; and the number of associated adverse incidents.
A total of 52 bortezomib-treated patients were included in the study. Patients were excluded if they were on a different combination of drugs that required hospital visits, or inpatient care for other reasons. Three patients ceased the drug – two because of toxicity, and one because of rapid progression. The average age of patients was 74 years, and 55.8% were using bortezomib as first-line, 36.5% second-line, and the remainder third-line or more.
The vast majority started the Homecare protocol (45/52), and 25 self-administered and 17 received a relative’s help. A total of 43 completed the self-administration protocol with two reverting to hospital assistance. Bortezomib was given for four to six cycles lasting around 6-8 months.
Clinical nurse specialists trained 38 patients for home care, with an average training time of 43 minutes. Two of these patients were considered unsuitable for self-administration. The remainder were trained by ward nurses or did not require training having received bortezomib previously.
A total of 20 patients required additional clinical nurse specialist time requiring an average of 55 minutes. Of those requiring additional support: Seven needed retraining; two needed the first dose delivered by a nurse specialist; two requested help from the hematology unit; and nine wanted general extra support – for example, help with injection site queries (usually administered to the abdominal area), reassurance during administration, syringe queries, administrative queries, and queries around spillages.
“Importantly, patients always have the phone number of the nurse specialist at hand. But most people managed okay, and even if they needed additional support they still got there,” remarked Dr. De Abrew.
In terms of adverse events, there were six in total. These included three reported spillages (with no harm caused), and three experienced injection site incidents (rash, pain). “We found a low number of reported adverse events,” she said.
Dr. De Abrew added that generally, many more medications were being converted to subcutaneous formulations in myeloma and other hematology conditions. “Perhaps these results could inform self-administration of other drugs. In hematology, we get so many new drugs come through every year, but we don’t get the increased resources to manage this in the day units. Broadening self-administration could really help with capacity as well as improve quality of life for the patients.
“These results show that it can be done!” she said.
Dr. De Abrew declared no relevant conflicts of interest. Dr. Jenner declared receiving honoraria from Janssen, which manufactures branded Velcade (bortezomib).
A version of this story originally appeared on Medscape.com.
Home administration of bortezomib (Velcade), as a once or twice-weekly subcutaneous self-injection is safe in patients with myeloma, significantly reducing hospital visits, and likely improving quality of life, a study shows.
The majority (43 of 52 patients) successfully self-administered bortezomib and completed the course. Also, hospital visits for those on the so-called Homecare programme reduced by 50%, with most visits comprising a fortnightly drug pickup from the drive-through pharmacy.
The work was presented as a poster by lead author and researcher, Kanchana De Abrew, hematology consultant at University Hospital Southampton NHS Foundation Trust, at this year’s virtual British Society of Haematology (BSH) meeting. De Abrew conducted the study while at Queen Alexandra Hospital, Portsmouth.
“We wanted to minimize patient visits to hospital because with travel time and waiting time, patients can easily find a visit takes up a whole morning, so this relates to their quality of life as well as having financial implications for patients,” Dr. De Abrew said in an interview. It also reduced the impact on day units and improved capacity for other services.
Dr. De Abrew noted that the study was conducted in the pre-COVID-19 era, but that the current enhanced threat of infection only served to reinforce the benefits of self-administration at home and avoiding unnecessary hospital visits.
“This project could easily be set up in other hospitals and some other centers have already contacted us about this. It might suit rural areas,” she added.
‘Safe and effective’
Dr. Matthew Jenner, consultant hematologist for University Hospital Southampton NHS Foundation Trust, who was not involved in the study, remarked that the study demonstrated another way to deliver bortezomib outside of hospital in addition to home care services that require trained nurses to administer treatment. “With a modest amount of training of the patient and family, it is both a safe and effective way of delivering treatment. This reduces hospital visits for the patient and frees up much needed capacity for heavily stretched chemotherapy units, creating space for other newer treatments that require hospital attendance.
“It is of benefit all round to both the patients undertaking self-administration and those who benefit from improved capacity,” added Dr. Jenner.
Avoiding hospital visits
Myeloma patients are already immunosuppressed prior to treatment and then this worsens once on treatment. Once they are sitting in a clinic environment they are surrounded by similarly immunosuppressed patients, so their risk is heightened further.
Figures suggest myeloma cases are on the increase. Annually, the United Kingdom sees around 5,800 new cases of myeloma and incidence increased by a significant 32% between the periods of 1993-1995 and 2015-2017. These figures were reflected in the patient numbers at the Queen Alexandra Hospital where the study was carried out. Many patients receive bortezomib, which forms the backbone of four National Institute for Health and Care Excellence (NICE) approved regimens.
“Patients are living longer so in the early 2000s patients had a life expectancy of 2-3 years, whereas now patients live for around 5 years. Also, the scope and lines of treatments have increased a lot. Over 50% of patients are likely to have bortezomib at some point in their management,” explained Dr. De Abrew.
Bortezomib is given once or twice weekly as a subcutaneous injection, and this usually continues for approximately 6-8 months with four to six cycles. Administering the drug in hospital requires around a half-hour slot placing considerable burden on the hematology day unit resources, and this can adversely affect the patient experience with waiting times and the need for frequent hospital visits.
Patient or relatives taught to self-administer at home
In 2017, clinical nurse specialists taught suitable patients to self-administer bortezomib in the Homecare protocol. Patients collected a 2-week supply of the drug. The protocol aimed to improve patient quality of life by reducing hospital visits, and increasing capacity in the hematology day unit. Since the start of the programme in 2017, the majority (71) of myeloma patients at Portsmouth have been treated through the Homecare program.
Dr. De Abrew conducted a retrospective review of patients who received bortezomib between January and October 2019 aimed at determining the effectiveness of the Homecare programme. To this end, she measured the proportion able to commence the Homecare protocol; the proportion successful in completing treatment on the Homecare protocol; the amount of additional clinical nurse specialist time required to support the Homecare protocol; and the number of associated adverse incidents.
A total of 52 bortezomib-treated patients were included in the study. Patients were excluded if they were on a different combination of drugs that required hospital visits, or inpatient care for other reasons. Three patients ceased the drug – two because of toxicity, and one because of rapid progression. The average age of patients was 74 years, and 55.8% were using bortezomib as first-line, 36.5% second-line, and the remainder third-line or more.
The vast majority started the Homecare protocol (45/52), and 25 self-administered and 17 received a relative’s help. A total of 43 completed the self-administration protocol with two reverting to hospital assistance. Bortezomib was given for four to six cycles lasting around 6-8 months.
Clinical nurse specialists trained 38 patients for home care, with an average training time of 43 minutes. Two of these patients were considered unsuitable for self-administration. The remainder were trained by ward nurses or did not require training having received bortezomib previously.
A total of 20 patients required additional clinical nurse specialist time requiring an average of 55 minutes. Of those requiring additional support: Seven needed retraining; two needed the first dose delivered by a nurse specialist; two requested help from the hematology unit; and nine wanted general extra support – for example, help with injection site queries (usually administered to the abdominal area), reassurance during administration, syringe queries, administrative queries, and queries around spillages.
“Importantly, patients always have the phone number of the nurse specialist at hand. But most people managed okay, and even if they needed additional support they still got there,” remarked Dr. De Abrew.
In terms of adverse events, there were six in total. These included three reported spillages (with no harm caused), and three experienced injection site incidents (rash, pain). “We found a low number of reported adverse events,” she said.
Dr. De Abrew added that generally, many more medications were being converted to subcutaneous formulations in myeloma and other hematology conditions. “Perhaps these results could inform self-administration of other drugs. In hematology, we get so many new drugs come through every year, but we don’t get the increased resources to manage this in the day units. Broadening self-administration could really help with capacity as well as improve quality of life for the patients.
“These results show that it can be done!” she said.
Dr. De Abrew declared no relevant conflicts of interest. Dr. Jenner declared receiving honoraria from Janssen, which manufactures branded Velcade (bortezomib).
A version of this story originally appeared on Medscape.com.
Lower BP and better tumor control with drug combo?
It’s not ready for the clinic, but new research suggests that angiotensin receptor II blockers (ARBs) widely used to treat hypertension may improve responses to cancer immunotherapy agents targeted against the programmed death-1/ligand-1 (PD-1/PD-L1) pathway.
That conclusion comes from an observational study of 597 patients with more than 3 dozen different cancer types treated in clinical trials at the US National Institutes of Health. Investigators found that both objective response rates and 3-year overall survival (OS) rates were significantly higher for patients treated with a PD-1/PD-L1 inhibitor who were on ARBs, compared with patients who weren’t taking the antihypertensive agents.
An association was also seen between higher ORR and OS rates for patients taking ACE inhibitors, but it was not statistically significant, reported Julius Strauss, MD, from the Center for Cancer Research at the National Cancer Institute in Bethesda, Md.
All study patients received PD-1/PD-L1 inhibitors, and the ORR for patients treated with ARBs was 33.8%, compared with 19.5% for those treated with ACE inhibitors, and 17% for those who took neither drug. The respective complete response (CR) rates were 11.3%, 3.7%, and 3.1%.
Strauss discussed the data during an online briefing prior to his presentation of the findings during the 32nd EORTC-NCI-AACR Symposium on Molecular Targets and Cancer Therapeutics, which is taking place virtually.
Several early studies have suggested that angiotensin II, in addition to its effect on blood pressure, can also affect cancer growth by leading to downstream production of two proteins: vascular endothelial growth factor (VEGF) and transforming growth factor–beta (TGF-beta), he explained.
“Both of these [proteins] have been linked to cancer growth and cancer resistance to immune system attack,” Strauss observed.
He also discussed the mechanics of possible effects. Angiotensin II increases VEGF and TGF-beta through binding to the AT1 receptor, but has the opposite effect when it binds to the AT2 receptor, resulting in a decrease in both of the growth factors, he added.
ACE inhibitors prevent the conversion of angiotensin I to angiotensin II, with the result being that the drugs indirectly block both the AT1 and AT2 receptors.
In contrast, ARBs block only the AT1 receptor and leave the AT2 counter-regulatory receptor alone, said Strauss.
More data, including on overall survival
Strauss and colleagues examined whether ACE inhibitors and/or ARBs could have an effect on the response to PD-1/PD-L1 immune checkpoint inhibitors delivered with or without other immunotherapies, such as anti-cytotoxic T-lymphocyte-associated protein 4 (CTLA-4) checkpoint inhibitors, or targeted agents such as tyrosine kinase inhibitors (TKIs).
They pooled data on 597 patients receiving PD-1/PD-L1 inhibitors in clinical trials for various cancers, including 71 receiving concomitant ARBs, 82 receiving an ACE inhibitor, and 444 who were not receiving either class of antihypertensives.
The above-mentioned improvement in ORR with ARBs compared with patients not receiving the drug was statistically significant (P = .001), as was the improvement in CR rates (P = .002). In contrast, neither ORR nor CR were significantly better with patients on ACE inhibitors compared with patients not taking these drugs.
In multiple regression analysis controlling for age, gender, body mass index (BMI), tumor type, and additional therapies given, the superior ORR and CR rates with ARBs remained (P = .039 and .002, respectively), while there continued to be no significant additional benefit with ACE inhibitors.
The median overall survival was 35.2 months for patients on ARBs, 26.2 months for those on ACE inhibitors, and 18.8 months for patients on neither drug. The respective 3-year OS rates were 48.1%, 37.2%, and 31.5%, with the difference between the ARB and no-drug groups being significant (P = .0078).
In regression analysis controlling for the factors mentioned before, the OS advantage with ARBs but not ACE inhibitors remained significant (P = .006 for ARBs, and .078 for ACE inhibitors).
Strauss emphasized that further study is needed to determine if AT1 blockade can improve outcomes when combined anti-PD-1/PD-L1-based therapy.
It might be reasonable for patients who are taking ACE inhibitors to control blood pressure and are also receiving immunotherapy with a PD-1/PD-L1 inhibitor to be switched to an ARB if it is deemed safe and if further research bears it out, said Strauss in response to a question from Medscape Medical News.
Hypothesis-generating study
Meeting cochair Emiliano Calvo, MD, PhD, from Hospital de Madrid Norte Sanchinarro in Madrid, who attended the media briefing but was not involved in the study, commented that hypothesis-generating research using drugs already on the market for other indications adds important value to cancer therapy.
James Gulley, MD, PhD, from the Center for Cancer Research at the NCI, also a meeting cochair, agreed with Calvo.
“Thinking about utilizing the data that already exists to really get hypothesis-generating questions, it also opens up the possibility for real-world data, real-world evidence from these big datasets from [electronic medical records] that we could really interrogate and understand what we might see and get these hypothesis-generating findings that we could then prospectively evaluate,” Gulley said.
The research was funded by the National Cancer Institute. Strauss and Gulley are National Cancer Institute employees. Calvo disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
It’s not ready for the clinic, but new research suggests that angiotensin receptor II blockers (ARBs) widely used to treat hypertension may improve responses to cancer immunotherapy agents targeted against the programmed death-1/ligand-1 (PD-1/PD-L1) pathway.
That conclusion comes from an observational study of 597 patients with more than 3 dozen different cancer types treated in clinical trials at the US National Institutes of Health. Investigators found that both objective response rates and 3-year overall survival (OS) rates were significantly higher for patients treated with a PD-1/PD-L1 inhibitor who were on ARBs, compared with patients who weren’t taking the antihypertensive agents.
An association was also seen between higher ORR and OS rates for patients taking ACE inhibitors, but it was not statistically significant, reported Julius Strauss, MD, from the Center for Cancer Research at the National Cancer Institute in Bethesda, Md.
All study patients received PD-1/PD-L1 inhibitors, and the ORR for patients treated with ARBs was 33.8%, compared with 19.5% for those treated with ACE inhibitors, and 17% for those who took neither drug. The respective complete response (CR) rates were 11.3%, 3.7%, and 3.1%.
Strauss discussed the data during an online briefing prior to his presentation of the findings during the 32nd EORTC-NCI-AACR Symposium on Molecular Targets and Cancer Therapeutics, which is taking place virtually.
Several early studies have suggested that angiotensin II, in addition to its effect on blood pressure, can also affect cancer growth by leading to downstream production of two proteins: vascular endothelial growth factor (VEGF) and transforming growth factor–beta (TGF-beta), he explained.
“Both of these [proteins] have been linked to cancer growth and cancer resistance to immune system attack,” Strauss observed.
He also discussed the mechanics of possible effects. Angiotensin II increases VEGF and TGF-beta through binding to the AT1 receptor, but has the opposite effect when it binds to the AT2 receptor, resulting in a decrease in both of the growth factors, he added.
ACE inhibitors prevent the conversion of angiotensin I to angiotensin II, with the result being that the drugs indirectly block both the AT1 and AT2 receptors.
In contrast, ARBs block only the AT1 receptor and leave the AT2 counter-regulatory receptor alone, said Strauss.
More data, including on overall survival
Strauss and colleagues examined whether ACE inhibitors and/or ARBs could have an effect on the response to PD-1/PD-L1 immune checkpoint inhibitors delivered with or without other immunotherapies, such as anti-cytotoxic T-lymphocyte-associated protein 4 (CTLA-4) checkpoint inhibitors, or targeted agents such as tyrosine kinase inhibitors (TKIs).
They pooled data on 597 patients receiving PD-1/PD-L1 inhibitors in clinical trials for various cancers, including 71 receiving concomitant ARBs, 82 receiving an ACE inhibitor, and 444 who were not receiving either class of antihypertensives.
The above-mentioned improvement in ORR with ARBs compared with patients not receiving the drug was statistically significant (P = .001), as was the improvement in CR rates (P = .002). In contrast, neither ORR nor CR were significantly better with patients on ACE inhibitors compared with patients not taking these drugs.
In multiple regression analysis controlling for age, gender, body mass index (BMI), tumor type, and additional therapies given, the superior ORR and CR rates with ARBs remained (P = .039 and .002, respectively), while there continued to be no significant additional benefit with ACE inhibitors.
The median overall survival was 35.2 months for patients on ARBs, 26.2 months for those on ACE inhibitors, and 18.8 months for patients on neither drug. The respective 3-year OS rates were 48.1%, 37.2%, and 31.5%, with the difference between the ARB and no-drug groups being significant (P = .0078).
In regression analysis controlling for the factors mentioned before, the OS advantage with ARBs but not ACE inhibitors remained significant (P = .006 for ARBs, and .078 for ACE inhibitors).
Strauss emphasized that further study is needed to determine if AT1 blockade can improve outcomes when combined anti-PD-1/PD-L1-based therapy.
It might be reasonable for patients who are taking ACE inhibitors to control blood pressure and are also receiving immunotherapy with a PD-1/PD-L1 inhibitor to be switched to an ARB if it is deemed safe and if further research bears it out, said Strauss in response to a question from Medscape Medical News.
Hypothesis-generating study
Meeting cochair Emiliano Calvo, MD, PhD, from Hospital de Madrid Norte Sanchinarro in Madrid, who attended the media briefing but was not involved in the study, commented that hypothesis-generating research using drugs already on the market for other indications adds important value to cancer therapy.
James Gulley, MD, PhD, from the Center for Cancer Research at the NCI, also a meeting cochair, agreed with Calvo.
“Thinking about utilizing the data that already exists to really get hypothesis-generating questions, it also opens up the possibility for real-world data, real-world evidence from these big datasets from [electronic medical records] that we could really interrogate and understand what we might see and get these hypothesis-generating findings that we could then prospectively evaluate,” Gulley said.
The research was funded by the National Cancer Institute. Strauss and Gulley are National Cancer Institute employees. Calvo disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
It’s not ready for the clinic, but new research suggests that angiotensin receptor II blockers (ARBs) widely used to treat hypertension may improve responses to cancer immunotherapy agents targeted against the programmed death-1/ligand-1 (PD-1/PD-L1) pathway.
That conclusion comes from an observational study of 597 patients with more than 3 dozen different cancer types treated in clinical trials at the US National Institutes of Health. Investigators found that both objective response rates and 3-year overall survival (OS) rates were significantly higher for patients treated with a PD-1/PD-L1 inhibitor who were on ARBs, compared with patients who weren’t taking the antihypertensive agents.
An association was also seen between higher ORR and OS rates for patients taking ACE inhibitors, but it was not statistically significant, reported Julius Strauss, MD, from the Center for Cancer Research at the National Cancer Institute in Bethesda, Md.
All study patients received PD-1/PD-L1 inhibitors, and the ORR for patients treated with ARBs was 33.8%, compared with 19.5% for those treated with ACE inhibitors, and 17% for those who took neither drug. The respective complete response (CR) rates were 11.3%, 3.7%, and 3.1%.
Strauss discussed the data during an online briefing prior to his presentation of the findings during the 32nd EORTC-NCI-AACR Symposium on Molecular Targets and Cancer Therapeutics, which is taking place virtually.
Several early studies have suggested that angiotensin II, in addition to its effect on blood pressure, can also affect cancer growth by leading to downstream production of two proteins: vascular endothelial growth factor (VEGF) and transforming growth factor–beta (TGF-beta), he explained.
“Both of these [proteins] have been linked to cancer growth and cancer resistance to immune system attack,” Strauss observed.
He also discussed the mechanics of possible effects. Angiotensin II increases VEGF and TGF-beta through binding to the AT1 receptor, but has the opposite effect when it binds to the AT2 receptor, resulting in a decrease in both of the growth factors, he added.
ACE inhibitors prevent the conversion of angiotensin I to angiotensin II, with the result being that the drugs indirectly block both the AT1 and AT2 receptors.
In contrast, ARBs block only the AT1 receptor and leave the AT2 counter-regulatory receptor alone, said Strauss.
More data, including on overall survival
Strauss and colleagues examined whether ACE inhibitors and/or ARBs could have an effect on the response to PD-1/PD-L1 immune checkpoint inhibitors delivered with or without other immunotherapies, such as anti-cytotoxic T-lymphocyte-associated protein 4 (CTLA-4) checkpoint inhibitors, or targeted agents such as tyrosine kinase inhibitors (TKIs).
They pooled data on 597 patients receiving PD-1/PD-L1 inhibitors in clinical trials for various cancers, including 71 receiving concomitant ARBs, 82 receiving an ACE inhibitor, and 444 who were not receiving either class of antihypertensives.
The above-mentioned improvement in ORR with ARBs compared with patients not receiving the drug was statistically significant (P = .001), as was the improvement in CR rates (P = .002). In contrast, neither ORR nor CR were significantly better with patients on ACE inhibitors compared with patients not taking these drugs.
In multiple regression analysis controlling for age, gender, body mass index (BMI), tumor type, and additional therapies given, the superior ORR and CR rates with ARBs remained (P = .039 and .002, respectively), while there continued to be no significant additional benefit with ACE inhibitors.
The median overall survival was 35.2 months for patients on ARBs, 26.2 months for those on ACE inhibitors, and 18.8 months for patients on neither drug. The respective 3-year OS rates were 48.1%, 37.2%, and 31.5%, with the difference between the ARB and no-drug groups being significant (P = .0078).
In regression analysis controlling for the factors mentioned before, the OS advantage with ARBs but not ACE inhibitors remained significant (P = .006 for ARBs, and .078 for ACE inhibitors).
Strauss emphasized that further study is needed to determine if AT1 blockade can improve outcomes when combined anti-PD-1/PD-L1-based therapy.
It might be reasonable for patients who are taking ACE inhibitors to control blood pressure and are also receiving immunotherapy with a PD-1/PD-L1 inhibitor to be switched to an ARB if it is deemed safe and if further research bears it out, said Strauss in response to a question from Medscape Medical News.
Hypothesis-generating study
Meeting cochair Emiliano Calvo, MD, PhD, from Hospital de Madrid Norte Sanchinarro in Madrid, who attended the media briefing but was not involved in the study, commented that hypothesis-generating research using drugs already on the market for other indications adds important value to cancer therapy.
James Gulley, MD, PhD, from the Center for Cancer Research at the NCI, also a meeting cochair, agreed with Calvo.
“Thinking about utilizing the data that already exists to really get hypothesis-generating questions, it also opens up the possibility for real-world data, real-world evidence from these big datasets from [electronic medical records] that we could really interrogate and understand what we might see and get these hypothesis-generating findings that we could then prospectively evaluate,” Gulley said.
The research was funded by the National Cancer Institute. Strauss and Gulley are National Cancer Institute employees. Calvo disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
Socioeconomic factors affect survival of multiple myeloma patients
Disparities driven by socioeconomic factors have been shown to affect outcomes for patients with a variety of cancer types. Researchers found that this was also true for patients with multiple myeloma, according to a report published in Hematology/Oncology and Stem Cell Therapy.
In particular, survival was affected by a variety of socioeconomic factors.
Researchers conducting the study queried the National Cancer Database for patients diagnosed with multiple myeloma between 2004 and 2016. Only those 56,102 patients who received systemic therapy as the first-line treatment were included, according to Thejus T. Jayakrishnan, MD, of Allegheny Health Network, Pittsburgh, and colleagues.
Enrollment rates for therapy were calculated using receiving systemic therapy as the incident event of interest (numerator) over time to initiation of therapy (denominator). The incident rate ratios were analyzed using Poisson regression. A multivariate Cox proportional hazards model was used for survival analysis of 50,543 patients, and differences were determined as hazard ratios.
Significant differences
The study showed that therapy enrollment was significantly affected by race and sex (P < .005), with the enrollment rate for women and for non-Hispanic Blacks both being lower versus men and non-Hispanic Whites, respectively.
Advanced age, earlier year of diagnosis, lack of insurance or Medicaid, and higher comorbidity were found to be associated with poor survival (HR >1), whereas being a woman or a non-Hispanic Black (who were speculated to have more favorable cytogenetic profiles), having a higher income, and having treatment at an academic center were all associated with improved survival (each category at HR <1).
“Disparities in [multiple myeloma] exist and are caused by a complex interplay of multiple factors, with socioeconomic factors such as insurance and income playing a dominant role. The disparities not only exact high human cost but also negatively impact the economics of health care,” the researchers concluded.
The study was not funded and the authors reported that they had no relevant disclosures.
SOURCE: Jayakrishnan TT et al. Hematol Oncol Stem Cell Ther. 2020 Oct 10. doi: 10.1016/j.hemonc.2020.09.005.
Disparities driven by socioeconomic factors have been shown to affect outcomes for patients with a variety of cancer types. Researchers found that this was also true for patients with multiple myeloma, according to a report published in Hematology/Oncology and Stem Cell Therapy.
In particular, survival was affected by a variety of socioeconomic factors.
Researchers conducting the study queried the National Cancer Database for patients diagnosed with multiple myeloma between 2004 and 2016. Only those 56,102 patients who received systemic therapy as the first-line treatment were included, according to Thejus T. Jayakrishnan, MD, of Allegheny Health Network, Pittsburgh, and colleagues.
Enrollment rates for therapy were calculated using receiving systemic therapy as the incident event of interest (numerator) over time to initiation of therapy (denominator). The incident rate ratios were analyzed using Poisson regression. A multivariate Cox proportional hazards model was used for survival analysis of 50,543 patients, and differences were determined as hazard ratios.
Significant differences
The study showed that therapy enrollment was significantly affected by race and sex (P < .005), with the enrollment rate for women and for non-Hispanic Blacks both being lower versus men and non-Hispanic Whites, respectively.
Advanced age, earlier year of diagnosis, lack of insurance or Medicaid, and higher comorbidity were found to be associated with poor survival (HR >1), whereas being a woman or a non-Hispanic Black (who were speculated to have more favorable cytogenetic profiles), having a higher income, and having treatment at an academic center were all associated with improved survival (each category at HR <1).
“Disparities in [multiple myeloma] exist and are caused by a complex interplay of multiple factors, with socioeconomic factors such as insurance and income playing a dominant role. The disparities not only exact high human cost but also negatively impact the economics of health care,” the researchers concluded.
The study was not funded and the authors reported that they had no relevant disclosures.
SOURCE: Jayakrishnan TT et al. Hematol Oncol Stem Cell Ther. 2020 Oct 10. doi: 10.1016/j.hemonc.2020.09.005.
Disparities driven by socioeconomic factors have been shown to affect outcomes for patients with a variety of cancer types. Researchers found that this was also true for patients with multiple myeloma, according to a report published in Hematology/Oncology and Stem Cell Therapy.
In particular, survival was affected by a variety of socioeconomic factors.
Researchers conducting the study queried the National Cancer Database for patients diagnosed with multiple myeloma between 2004 and 2016. Only those 56,102 patients who received systemic therapy as the first-line treatment were included, according to Thejus T. Jayakrishnan, MD, of Allegheny Health Network, Pittsburgh, and colleagues.
Enrollment rates for therapy were calculated using receiving systemic therapy as the incident event of interest (numerator) over time to initiation of therapy (denominator). The incident rate ratios were analyzed using Poisson regression. A multivariate Cox proportional hazards model was used for survival analysis of 50,543 patients, and differences were determined as hazard ratios.
Significant differences
The study showed that therapy enrollment was significantly affected by race and sex (P < .005), with the enrollment rate for women and for non-Hispanic Blacks both being lower versus men and non-Hispanic Whites, respectively.
Advanced age, earlier year of diagnosis, lack of insurance or Medicaid, and higher comorbidity were found to be associated with poor survival (HR >1), whereas being a woman or a non-Hispanic Black (who were speculated to have more favorable cytogenetic profiles), having a higher income, and having treatment at an academic center were all associated with improved survival (each category at HR <1).
“Disparities in [multiple myeloma] exist and are caused by a complex interplay of multiple factors, with socioeconomic factors such as insurance and income playing a dominant role. The disparities not only exact high human cost but also negatively impact the economics of health care,” the researchers concluded.
The study was not funded and the authors reported that they had no relevant disclosures.
SOURCE: Jayakrishnan TT et al. Hematol Oncol Stem Cell Ther. 2020 Oct 10. doi: 10.1016/j.hemonc.2020.09.005.
FROM HEMATOLOGY/ONCOLOGY AND STEM CELL THERAPY
Are oncologists ready to confront a second wave of COVID-19?
Canceled appointments, postponed surgeries, and delayed cancer diagnoses – all are a recipe for exhaustion for oncologists around the world, struggling to reach and treat their patients during the pandemic. Physicians and their teams felt the pain as COVID-19 took its initial march around the globe.
“We saw the distress of people with cancer who could no longer get to anyone on the phone. Their medical visit was usually canceled. Their radiotherapy session was postponed or modified, and chemotherapy postponed,” says Axel Kahn, MD, chairman of the board of directors of La Ligue Nationale Contre le Cancer (National League Against Cancer). “In the vast majority of cases, cancer treatment can be postponed or readjusted, without affecting the patient’s chances of survival, but there has been a lot of anxiety because the patients do not know that.”
The stay-at-home factor was one that played out across many months during the first wave.
“I believe that the ‘stay-home’ message that we transmitted was rigorously followed by patients who should have come to the emergency room much earlier and who, therefore, were admitted with a much more deteriorated general condition than in non-COVID-19 times,” says Benjamín Domingo Arrué, MD, from the department of medical oncology at Hospital Universitari i Politècnic La Fe in Valencia, Spain.
And in Brazil, some of the impact from the initial hit of COVID-19 on oncology is only now being felt, according to Laura Testa, MD, head of breast medical oncology, Instituto do Câncer do Estado de São Paulo.
“We are starting to see a lot of cancer cases that didn’t show up at the beginning of the pandemic, but now they are arriving to us already in advanced stages,” she said. “These patients need hospital care. If the situation worsens and goes back to what we saw at the peak of the curve, I fear the public system won’t be able to treat properly the oncology patients that need hospital care and the patients with cancer who also have COVID-19.”
But even as health care worker fatigue and concerns linger, oncologists say that what they have learned in the last 6 months has helped them prepare as COVID-19 cases increase and a second global wave kicks up.
Lessons from the first wave
In the United States, COVID-19 hit different regions at different times and to different degrees. One of the areas hit first was Seattle.
“We jumped on top of this, we were evidence based, we put things in place very, very quickly,” said Julie Gralow, MD, professor at the University of Washington and the Fred Hutchinson Cancer Research Center, both in Seattle.
“We did a really good job keeping COVID out of our cancer centers,” Dr. Gralow said. “We learned how to be super safe, and to keep symptomatic people out of the building, and to limit the extra people they could bring with them. It’s all about the number of contacts you have.”
The story was different, though, for oncologists in several other countries, and sometimes it varied immensely within each nation.
“We treated fewer patients with cancer during the first wave,” says Dirk Arnold, MD, medical director of the Asklepios Tumor Center Hamburg (Germany), in an interview. “In part, this was because staff were quarantined and because we had a completely different infrastructure in all of the hospitals. But also fewer patients with cancer came to the clinic at all. A lot of resources were directed toward COVID-19.”
In Spain, telemedicine helped keep up with visits, but other areas felt the effect of COVID-19 patient loads.
“At least in the oncology department of our center, we have practically maintained 100% of visits, mostly by telephone,” says Dr. Arrué, “but the reality is that our country has not yet been prepared for telemedicine.”
Laura Mezquita, MD, of the department of medical oncology at Hospital Clinic de Barcelona, describes a more dramatic situation: “We have seen how some of our patients, especially with metastatic disease, have been dismissed for intensive care and life-support treatments, as well as specific treatments against COVID-19 (tocilizumab, remdesivir, etc.) due to the general health collapse of the former wave,” she said. She adds that specific oncologic populations, such as those with thoracic tumors, have been more affected.
Distress among oncologists
Many oncologists are still feeling stressed and fatigued after the first wave, just as a second string of outbreaks is on its way.
A survey presented at last month’s ESMO 2020 Congress found that, in July-August, moral distress was reported by one-third of the oncologists who responded, and more than half reported a feeling of exhaustion.
“The tiredness and team exhaustion is noticeable,” said Dr. Arnold. “We recently had a task force discussion about what will happen when we have a second wave and how the department and our services will adapt. It was clear that those who were at the very front in the first wave had only a limited desire to do that again in the second wave.”
Another concern: COVID-19’s effect on staffing levels.
“We have a population of young caregivers who are affected by the COVID-19 disease with an absenteeism rate that is quite unprecedented,” said Sophie Beaupère, general delegate of Unicancer since January.
She said that, in general, the absenteeism rate in the cancer centers averages 5%-6%, depending on the year. But that rate is now skyrocketing.
Stop-start cycle for surgery
As caregivers quarantined around the world, more than 10% of patients with cancer had treatment canceled or delayed during the first wave of the pandemic, according to another survey from ESMO, involving 109 oncologists from 18 countries.
Difficulties were reported for surgeries by 34% of the centers, but also difficulties with delivering chemotherapy (22% of centers), radiotherapy (13.7%), and therapy with checkpoint inhibitors (9.1%), monoclonal antibodies (9%), and oral targeted therapy (3.7%).
Stopping surgery is a real concern in France, noted Dr. Kahn, the National League Against Cancer chair. He says that in regions that were badly hit by COVID-19, “it was not possible to have access to the operating room for people who absolutely needed surgery; for example, patients with lung cancer that was still operable. Most of the recovery rooms were mobilized for resuscitation.”
There may be some solutions, suggested Thierry Breton, director general of the National Institute of Cancer in France. “We are getting prepared, with the health ministry, for a possible increase in hospital tension, which would lead to a situation where we would have to reschedule operations. Nationally, regionally, and locally, we are seeing how we can resume and prioritize surgeries that have not been done.”
Delays in cancer diagnosis
While COVID-19 affected treatment, many oncologists say the major impact of the first wave was a delay in diagnosing cancer. Some of this was a result of the suspension of cancer screening programs, but there was also fear among the general public about visiting clinics and hospitals during a pandemic.
“We didn’t do so well with cancer during the first wave here in the U.K.,” said Karol Sikora, PhD, MBBChir, professor of cancer medicine and founding dean at the University of Buckingham Medical School, London. “Cancer diagnostic pathways virtually stalled partly because patients didn’t seek help, but getting scans and biopsies was also very difficult. Even patients referred urgently under the ‘2-weeks-wait’ rule were turned down.”
In France, “the delay in diagnosis is indisputable,” said Dr. Kahn. “About 50% of the cancer diagnoses one would expect during this period were missed.”
“I am worried that there remains a major traffic jam that has not been caught up with, and, in the meantime, the health crisis is worsening,” he added.
In Seattle, Dr. Gralow said the first COVID-19 wave had little impact on treatment for breast cancer, but it was in screening for breast cancer “where things really got messed up.”
“Even though we’ve been fully ramped up again,” she said, concerns remain. To ensure that screening mammography is maintained, “we have spaced out the visits to keep our waiting rooms less populated, with a longer time between using the machine so we can clean it. To do this, we have extended operating hours and are now opening on Saturday.
“So we’re actually at 100% of our capacity, but I’m really nervous, though, that a lot of people put off their screening mammogram and aren’t going to come in and get it.
“Not only did people get the message to stay home and not do nonessential things, but I think a lot of people lost their health insurance when they lost their jobs,” she said, and without health insurance, they are not covered for cancer screening.
Looking ahead, with a plan
Many oncologists agree that access to care can and must be improved – and there were some positive moves.
“Some regimens changed during the first months of the pandemic, and I don’t see them going back to the way they were anytime soon,” said Dr. Testa. “The changes/adaptations that were made to minimize the chance of SARS-CoV-2 infection are still in place and will go on for a while. In this context, telemedicine helped a lot. The pandemic forced the stakeholders to step up and put it in place in March. And now it’s here to stay.”
The experience gained in the last several months has driven preparation for the next wave.
“We are not going to see the disorganization that we saw during the first wave,” said Florence Joly, MD, PhD, head of medical oncology at the Centre François Baclesse in Caen, France. “The difference between now and earlier this year is that COVID diagnostic tests are available. That was one of the problems in the first wave. We had no way to diagnose.”
On the East Coast of the United States, medical oncologist Charu Aggarwal, MD, MPH, is also optimistic: “I think we’re at a place where we can manage.”
“I believe if there was going to be a new wave of COVID-19 cases we would be: better psychologically prepared and better organized,” said Dr. Aggarwal, assistant professor of medicine in the hematology-oncology division at the University of Pennsylvania, Philadelphia. “We already have experience with all of the tools, we have telemedicine available, we have screening protocols available, we have testing, we are already universally masking, everyone’s hand-washing, so I do think that means we would be okay.”
Dr. Arnold agreed that “we are much better prepared than for the first wave, but … we have immense tasks in the area of patient management, the digitization of patient care, the clear allocation of resources when there is a second or third wave. In many areas of preparation, I believe, unfortunately, we are not as well positioned as we had actually hoped.”
The first wave of COVID hit cancer services in the United Kingdom particularly hard: One modeling study suggested that delays in cancer referrals will lead to thousands of additional deaths and tens of thousands of life-years lost.
“Cancer services are working at near normal levels now, but they are still fragile and could be severely compromised again if the NHS [National Health Service] gets flooded by COVID patients,” said Dr. Sikora.
The second wave may be different. “Although the number of infections has increased, the hospitalizations have only risen a little. Let’s see what happens,” he said in an interview. Since then, however, infections have continued to rise, and there has been an increase in hospitalizations. New social distancing measures in the United Kingdom were put into place on Oct. 12, with the aim of protecting the NHS from overload.
Dr. Arrué describes it this way: “The reality is that the ‘second wave’ has left behind the initial grief and shock that both patients and health professionals experienced when faced with something that, until now, we had only seen in the movies.” The second wave has led to new restrictions – including a partial lockdown since the beginning of October.
Dr. Aggarwal says her department recently had a conference with Anthony Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, about the impact of COVID-19 on oncology.
“I asked him what advice he’d give oncologists, and he said to go back to as much screening as you were doing previously as quickly as possible. That’s what must be relayed to our oncologists in the community – and also to primary care physicians – because they are often the ones who are ordering and championing the screening efforts.”
This article was originated by Aude Lecrubier, Medscape French edition, and developed by Zosia Chustecka, Medscape Oncology. With additional reporting by Kate Johnson, freelance medical journalist, Claudia Gottschling for Medscape Germany, Leoleli Schwartz for Medscape em português, Tim Locke for Medscape United Kingdom, and Carla Nieto Martínez, freelance medical journalist for Medscape Spanish edition.
This article first appeared on Medscape.com.
Canceled appointments, postponed surgeries, and delayed cancer diagnoses – all are a recipe for exhaustion for oncologists around the world, struggling to reach and treat their patients during the pandemic. Physicians and their teams felt the pain as COVID-19 took its initial march around the globe.
“We saw the distress of people with cancer who could no longer get to anyone on the phone. Their medical visit was usually canceled. Their radiotherapy session was postponed or modified, and chemotherapy postponed,” says Axel Kahn, MD, chairman of the board of directors of La Ligue Nationale Contre le Cancer (National League Against Cancer). “In the vast majority of cases, cancer treatment can be postponed or readjusted, without affecting the patient’s chances of survival, but there has been a lot of anxiety because the patients do not know that.”
The stay-at-home factor was one that played out across many months during the first wave.
“I believe that the ‘stay-home’ message that we transmitted was rigorously followed by patients who should have come to the emergency room much earlier and who, therefore, were admitted with a much more deteriorated general condition than in non-COVID-19 times,” says Benjamín Domingo Arrué, MD, from the department of medical oncology at Hospital Universitari i Politècnic La Fe in Valencia, Spain.
And in Brazil, some of the impact from the initial hit of COVID-19 on oncology is only now being felt, according to Laura Testa, MD, head of breast medical oncology, Instituto do Câncer do Estado de São Paulo.
“We are starting to see a lot of cancer cases that didn’t show up at the beginning of the pandemic, but now they are arriving to us already in advanced stages,” she said. “These patients need hospital care. If the situation worsens and goes back to what we saw at the peak of the curve, I fear the public system won’t be able to treat properly the oncology patients that need hospital care and the patients with cancer who also have COVID-19.”
But even as health care worker fatigue and concerns linger, oncologists say that what they have learned in the last 6 months has helped them prepare as COVID-19 cases increase and a second global wave kicks up.
Lessons from the first wave
In the United States, COVID-19 hit different regions at different times and to different degrees. One of the areas hit first was Seattle.
“We jumped on top of this, we were evidence based, we put things in place very, very quickly,” said Julie Gralow, MD, professor at the University of Washington and the Fred Hutchinson Cancer Research Center, both in Seattle.
“We did a really good job keeping COVID out of our cancer centers,” Dr. Gralow said. “We learned how to be super safe, and to keep symptomatic people out of the building, and to limit the extra people they could bring with them. It’s all about the number of contacts you have.”
The story was different, though, for oncologists in several other countries, and sometimes it varied immensely within each nation.
“We treated fewer patients with cancer during the first wave,” says Dirk Arnold, MD, medical director of the Asklepios Tumor Center Hamburg (Germany), in an interview. “In part, this was because staff were quarantined and because we had a completely different infrastructure in all of the hospitals. But also fewer patients with cancer came to the clinic at all. A lot of resources were directed toward COVID-19.”
In Spain, telemedicine helped keep up with visits, but other areas felt the effect of COVID-19 patient loads.
“At least in the oncology department of our center, we have practically maintained 100% of visits, mostly by telephone,” says Dr. Arrué, “but the reality is that our country has not yet been prepared for telemedicine.”
Laura Mezquita, MD, of the department of medical oncology at Hospital Clinic de Barcelona, describes a more dramatic situation: “We have seen how some of our patients, especially with metastatic disease, have been dismissed for intensive care and life-support treatments, as well as specific treatments against COVID-19 (tocilizumab, remdesivir, etc.) due to the general health collapse of the former wave,” she said. She adds that specific oncologic populations, such as those with thoracic tumors, have been more affected.
Distress among oncologists
Many oncologists are still feeling stressed and fatigued after the first wave, just as a second string of outbreaks is on its way.
A survey presented at last month’s ESMO 2020 Congress found that, in July-August, moral distress was reported by one-third of the oncologists who responded, and more than half reported a feeling of exhaustion.
“The tiredness and team exhaustion is noticeable,” said Dr. Arnold. “We recently had a task force discussion about what will happen when we have a second wave and how the department and our services will adapt. It was clear that those who were at the very front in the first wave had only a limited desire to do that again in the second wave.”
Another concern: COVID-19’s effect on staffing levels.
“We have a population of young caregivers who are affected by the COVID-19 disease with an absenteeism rate that is quite unprecedented,” said Sophie Beaupère, general delegate of Unicancer since January.
She said that, in general, the absenteeism rate in the cancer centers averages 5%-6%, depending on the year. But that rate is now skyrocketing.
Stop-start cycle for surgery
As caregivers quarantined around the world, more than 10% of patients with cancer had treatment canceled or delayed during the first wave of the pandemic, according to another survey from ESMO, involving 109 oncologists from 18 countries.
Difficulties were reported for surgeries by 34% of the centers, but also difficulties with delivering chemotherapy (22% of centers), radiotherapy (13.7%), and therapy with checkpoint inhibitors (9.1%), monoclonal antibodies (9%), and oral targeted therapy (3.7%).
Stopping surgery is a real concern in France, noted Dr. Kahn, the National League Against Cancer chair. He says that in regions that were badly hit by COVID-19, “it was not possible to have access to the operating room for people who absolutely needed surgery; for example, patients with lung cancer that was still operable. Most of the recovery rooms were mobilized for resuscitation.”
There may be some solutions, suggested Thierry Breton, director general of the National Institute of Cancer in France. “We are getting prepared, with the health ministry, for a possible increase in hospital tension, which would lead to a situation where we would have to reschedule operations. Nationally, regionally, and locally, we are seeing how we can resume and prioritize surgeries that have not been done.”
Delays in cancer diagnosis
While COVID-19 affected treatment, many oncologists say the major impact of the first wave was a delay in diagnosing cancer. Some of this was a result of the suspension of cancer screening programs, but there was also fear among the general public about visiting clinics and hospitals during a pandemic.
“We didn’t do so well with cancer during the first wave here in the U.K.,” said Karol Sikora, PhD, MBBChir, professor of cancer medicine and founding dean at the University of Buckingham Medical School, London. “Cancer diagnostic pathways virtually stalled partly because patients didn’t seek help, but getting scans and biopsies was also very difficult. Even patients referred urgently under the ‘2-weeks-wait’ rule were turned down.”
In France, “the delay in diagnosis is indisputable,” said Dr. Kahn. “About 50% of the cancer diagnoses one would expect during this period were missed.”
“I am worried that there remains a major traffic jam that has not been caught up with, and, in the meantime, the health crisis is worsening,” he added.
In Seattle, Dr. Gralow said the first COVID-19 wave had little impact on treatment for breast cancer, but it was in screening for breast cancer “where things really got messed up.”
“Even though we’ve been fully ramped up again,” she said, concerns remain. To ensure that screening mammography is maintained, “we have spaced out the visits to keep our waiting rooms less populated, with a longer time between using the machine so we can clean it. To do this, we have extended operating hours and are now opening on Saturday.
“So we’re actually at 100% of our capacity, but I’m really nervous, though, that a lot of people put off their screening mammogram and aren’t going to come in and get it.
“Not only did people get the message to stay home and not do nonessential things, but I think a lot of people lost their health insurance when they lost their jobs,” she said, and without health insurance, they are not covered for cancer screening.
Looking ahead, with a plan
Many oncologists agree that access to care can and must be improved – and there were some positive moves.
“Some regimens changed during the first months of the pandemic, and I don’t see them going back to the way they were anytime soon,” said Dr. Testa. “The changes/adaptations that were made to minimize the chance of SARS-CoV-2 infection are still in place and will go on for a while. In this context, telemedicine helped a lot. The pandemic forced the stakeholders to step up and put it in place in March. And now it’s here to stay.”
The experience gained in the last several months has driven preparation for the next wave.
“We are not going to see the disorganization that we saw during the first wave,” said Florence Joly, MD, PhD, head of medical oncology at the Centre François Baclesse in Caen, France. “The difference between now and earlier this year is that COVID diagnostic tests are available. That was one of the problems in the first wave. We had no way to diagnose.”
On the East Coast of the United States, medical oncologist Charu Aggarwal, MD, MPH, is also optimistic: “I think we’re at a place where we can manage.”
“I believe if there was going to be a new wave of COVID-19 cases we would be: better psychologically prepared and better organized,” said Dr. Aggarwal, assistant professor of medicine in the hematology-oncology division at the University of Pennsylvania, Philadelphia. “We already have experience with all of the tools, we have telemedicine available, we have screening protocols available, we have testing, we are already universally masking, everyone’s hand-washing, so I do think that means we would be okay.”
Dr. Arnold agreed that “we are much better prepared than for the first wave, but … we have immense tasks in the area of patient management, the digitization of patient care, the clear allocation of resources when there is a second or third wave. In many areas of preparation, I believe, unfortunately, we are not as well positioned as we had actually hoped.”
The first wave of COVID hit cancer services in the United Kingdom particularly hard: One modeling study suggested that delays in cancer referrals will lead to thousands of additional deaths and tens of thousands of life-years lost.
“Cancer services are working at near normal levels now, but they are still fragile and could be severely compromised again if the NHS [National Health Service] gets flooded by COVID patients,” said Dr. Sikora.
The second wave may be different. “Although the number of infections has increased, the hospitalizations have only risen a little. Let’s see what happens,” he said in an interview. Since then, however, infections have continued to rise, and there has been an increase in hospitalizations. New social distancing measures in the United Kingdom were put into place on Oct. 12, with the aim of protecting the NHS from overload.
Dr. Arrué describes it this way: “The reality is that the ‘second wave’ has left behind the initial grief and shock that both patients and health professionals experienced when faced with something that, until now, we had only seen in the movies.” The second wave has led to new restrictions – including a partial lockdown since the beginning of October.
Dr. Aggarwal says her department recently had a conference with Anthony Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, about the impact of COVID-19 on oncology.
“I asked him what advice he’d give oncologists, and he said to go back to as much screening as you were doing previously as quickly as possible. That’s what must be relayed to our oncologists in the community – and also to primary care physicians – because they are often the ones who are ordering and championing the screening efforts.”
This article was originated by Aude Lecrubier, Medscape French edition, and developed by Zosia Chustecka, Medscape Oncology. With additional reporting by Kate Johnson, freelance medical journalist, Claudia Gottschling for Medscape Germany, Leoleli Schwartz for Medscape em português, Tim Locke for Medscape United Kingdom, and Carla Nieto Martínez, freelance medical journalist for Medscape Spanish edition.
This article first appeared on Medscape.com.
Canceled appointments, postponed surgeries, and delayed cancer diagnoses – all are a recipe for exhaustion for oncologists around the world, struggling to reach and treat their patients during the pandemic. Physicians and their teams felt the pain as COVID-19 took its initial march around the globe.
“We saw the distress of people with cancer who could no longer get to anyone on the phone. Their medical visit was usually canceled. Their radiotherapy session was postponed or modified, and chemotherapy postponed,” says Axel Kahn, MD, chairman of the board of directors of La Ligue Nationale Contre le Cancer (National League Against Cancer). “In the vast majority of cases, cancer treatment can be postponed or readjusted, without affecting the patient’s chances of survival, but there has been a lot of anxiety because the patients do not know that.”
The stay-at-home factor was one that played out across many months during the first wave.
“I believe that the ‘stay-home’ message that we transmitted was rigorously followed by patients who should have come to the emergency room much earlier and who, therefore, were admitted with a much more deteriorated general condition than in non-COVID-19 times,” says Benjamín Domingo Arrué, MD, from the department of medical oncology at Hospital Universitari i Politècnic La Fe in Valencia, Spain.
And in Brazil, some of the impact from the initial hit of COVID-19 on oncology is only now being felt, according to Laura Testa, MD, head of breast medical oncology, Instituto do Câncer do Estado de São Paulo.
“We are starting to see a lot of cancer cases that didn’t show up at the beginning of the pandemic, but now they are arriving to us already in advanced stages,” she said. “These patients need hospital care. If the situation worsens and goes back to what we saw at the peak of the curve, I fear the public system won’t be able to treat properly the oncology patients that need hospital care and the patients with cancer who also have COVID-19.”
But even as health care worker fatigue and concerns linger, oncologists say that what they have learned in the last 6 months has helped them prepare as COVID-19 cases increase and a second global wave kicks up.
Lessons from the first wave
In the United States, COVID-19 hit different regions at different times and to different degrees. One of the areas hit first was Seattle.
“We jumped on top of this, we were evidence based, we put things in place very, very quickly,” said Julie Gralow, MD, professor at the University of Washington and the Fred Hutchinson Cancer Research Center, both in Seattle.
“We did a really good job keeping COVID out of our cancer centers,” Dr. Gralow said. “We learned how to be super safe, and to keep symptomatic people out of the building, and to limit the extra people they could bring with them. It’s all about the number of contacts you have.”
The story was different, though, for oncologists in several other countries, and sometimes it varied immensely within each nation.
“We treated fewer patients with cancer during the first wave,” says Dirk Arnold, MD, medical director of the Asklepios Tumor Center Hamburg (Germany), in an interview. “In part, this was because staff were quarantined and because we had a completely different infrastructure in all of the hospitals. But also fewer patients with cancer came to the clinic at all. A lot of resources were directed toward COVID-19.”
In Spain, telemedicine helped keep up with visits, but other areas felt the effect of COVID-19 patient loads.
“At least in the oncology department of our center, we have practically maintained 100% of visits, mostly by telephone,” says Dr. Arrué, “but the reality is that our country has not yet been prepared for telemedicine.”
Laura Mezquita, MD, of the department of medical oncology at Hospital Clinic de Barcelona, describes a more dramatic situation: “We have seen how some of our patients, especially with metastatic disease, have been dismissed for intensive care and life-support treatments, as well as specific treatments against COVID-19 (tocilizumab, remdesivir, etc.) due to the general health collapse of the former wave,” she said. She adds that specific oncologic populations, such as those with thoracic tumors, have been more affected.
Distress among oncologists
Many oncologists are still feeling stressed and fatigued after the first wave, just as a second string of outbreaks is on its way.
A survey presented at last month’s ESMO 2020 Congress found that, in July-August, moral distress was reported by one-third of the oncologists who responded, and more than half reported a feeling of exhaustion.
“The tiredness and team exhaustion is noticeable,” said Dr. Arnold. “We recently had a task force discussion about what will happen when we have a second wave and how the department and our services will adapt. It was clear that those who were at the very front in the first wave had only a limited desire to do that again in the second wave.”
Another concern: COVID-19’s effect on staffing levels.
“We have a population of young caregivers who are affected by the COVID-19 disease with an absenteeism rate that is quite unprecedented,” said Sophie Beaupère, general delegate of Unicancer since January.
She said that, in general, the absenteeism rate in the cancer centers averages 5%-6%, depending on the year. But that rate is now skyrocketing.
Stop-start cycle for surgery
As caregivers quarantined around the world, more than 10% of patients with cancer had treatment canceled or delayed during the first wave of the pandemic, according to another survey from ESMO, involving 109 oncologists from 18 countries.
Difficulties were reported for surgeries by 34% of the centers, but also difficulties with delivering chemotherapy (22% of centers), radiotherapy (13.7%), and therapy with checkpoint inhibitors (9.1%), monoclonal antibodies (9%), and oral targeted therapy (3.7%).
Stopping surgery is a real concern in France, noted Dr. Kahn, the National League Against Cancer chair. He says that in regions that were badly hit by COVID-19, “it was not possible to have access to the operating room for people who absolutely needed surgery; for example, patients with lung cancer that was still operable. Most of the recovery rooms were mobilized for resuscitation.”
There may be some solutions, suggested Thierry Breton, director general of the National Institute of Cancer in France. “We are getting prepared, with the health ministry, for a possible increase in hospital tension, which would lead to a situation where we would have to reschedule operations. Nationally, regionally, and locally, we are seeing how we can resume and prioritize surgeries that have not been done.”
Delays in cancer diagnosis
While COVID-19 affected treatment, many oncologists say the major impact of the first wave was a delay in diagnosing cancer. Some of this was a result of the suspension of cancer screening programs, but there was also fear among the general public about visiting clinics and hospitals during a pandemic.
“We didn’t do so well with cancer during the first wave here in the U.K.,” said Karol Sikora, PhD, MBBChir, professor of cancer medicine and founding dean at the University of Buckingham Medical School, London. “Cancer diagnostic pathways virtually stalled partly because patients didn’t seek help, but getting scans and biopsies was also very difficult. Even patients referred urgently under the ‘2-weeks-wait’ rule were turned down.”
In France, “the delay in diagnosis is indisputable,” said Dr. Kahn. “About 50% of the cancer diagnoses one would expect during this period were missed.”
“I am worried that there remains a major traffic jam that has not been caught up with, and, in the meantime, the health crisis is worsening,” he added.
In Seattle, Dr. Gralow said the first COVID-19 wave had little impact on treatment for breast cancer, but it was in screening for breast cancer “where things really got messed up.”
“Even though we’ve been fully ramped up again,” she said, concerns remain. To ensure that screening mammography is maintained, “we have spaced out the visits to keep our waiting rooms less populated, with a longer time between using the machine so we can clean it. To do this, we have extended operating hours and are now opening on Saturday.
“So we’re actually at 100% of our capacity, but I’m really nervous, though, that a lot of people put off their screening mammogram and aren’t going to come in and get it.
“Not only did people get the message to stay home and not do nonessential things, but I think a lot of people lost their health insurance when they lost their jobs,” she said, and without health insurance, they are not covered for cancer screening.
Looking ahead, with a plan
Many oncologists agree that access to care can and must be improved – and there were some positive moves.
“Some regimens changed during the first months of the pandemic, and I don’t see them going back to the way they were anytime soon,” said Dr. Testa. “The changes/adaptations that were made to minimize the chance of SARS-CoV-2 infection are still in place and will go on for a while. In this context, telemedicine helped a lot. The pandemic forced the stakeholders to step up and put it in place in March. And now it’s here to stay.”
The experience gained in the last several months has driven preparation for the next wave.
“We are not going to see the disorganization that we saw during the first wave,” said Florence Joly, MD, PhD, head of medical oncology at the Centre François Baclesse in Caen, France. “The difference between now and earlier this year is that COVID diagnostic tests are available. That was one of the problems in the first wave. We had no way to diagnose.”
On the East Coast of the United States, medical oncologist Charu Aggarwal, MD, MPH, is also optimistic: “I think we’re at a place where we can manage.”
“I believe if there was going to be a new wave of COVID-19 cases we would be: better psychologically prepared and better organized,” said Dr. Aggarwal, assistant professor of medicine in the hematology-oncology division at the University of Pennsylvania, Philadelphia. “We already have experience with all of the tools, we have telemedicine available, we have screening protocols available, we have testing, we are already universally masking, everyone’s hand-washing, so I do think that means we would be okay.”
Dr. Arnold agreed that “we are much better prepared than for the first wave, but … we have immense tasks in the area of patient management, the digitization of patient care, the clear allocation of resources when there is a second or third wave. In many areas of preparation, I believe, unfortunately, we are not as well positioned as we had actually hoped.”
The first wave of COVID hit cancer services in the United Kingdom particularly hard: One modeling study suggested that delays in cancer referrals will lead to thousands of additional deaths and tens of thousands of life-years lost.
“Cancer services are working at near normal levels now, but they are still fragile and could be severely compromised again if the NHS [National Health Service] gets flooded by COVID patients,” said Dr. Sikora.
The second wave may be different. “Although the number of infections has increased, the hospitalizations have only risen a little. Let’s see what happens,” he said in an interview. Since then, however, infections have continued to rise, and there has been an increase in hospitalizations. New social distancing measures in the United Kingdom were put into place on Oct. 12, with the aim of protecting the NHS from overload.
Dr. Arrué describes it this way: “The reality is that the ‘second wave’ has left behind the initial grief and shock that both patients and health professionals experienced when faced with something that, until now, we had only seen in the movies.” The second wave has led to new restrictions – including a partial lockdown since the beginning of October.
Dr. Aggarwal says her department recently had a conference with Anthony Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, about the impact of COVID-19 on oncology.
“I asked him what advice he’d give oncologists, and he said to go back to as much screening as you were doing previously as quickly as possible. That’s what must be relayed to our oncologists in the community – and also to primary care physicians – because they are often the ones who are ordering and championing the screening efforts.”
This article was originated by Aude Lecrubier, Medscape French edition, and developed by Zosia Chustecka, Medscape Oncology. With additional reporting by Kate Johnson, freelance medical journalist, Claudia Gottschling for Medscape Germany, Leoleli Schwartz for Medscape em português, Tim Locke for Medscape United Kingdom, and Carla Nieto Martínez, freelance medical journalist for Medscape Spanish edition.
This article first appeared on Medscape.com.
Older age, r/r disease in lymphoma patients tied to increased COVID-19 death rate
Patients with B-cell lymphoma are immunocompromised because of the disease and its treatments. This presents the question of their outcomes upon infection with SARS-CoV-2. Researchers assessed the characteristics of patients with lymphoma hospitalized for COVID-19 and analyzed determinants of mortality in a retrospective database study. The investigators looked at data from adult patients with lymphoma who were hospitalized for COVID-19 in March and April 2020 in three French regions.
Older age and relapsed/refractory (r/r) disease in B-cell lymphoma patients were both found to be independent risk factors of increased death rate from COVID-19, according to the online report in EClinicalMedicine, published by The Lancet.
These results encourage “the application of standard Covid-19 treatment, including intubation, for lymphoma patients with Covid-19 lymphoma diagnosis, under first- or second-line chemotherapy, or in remission,” according to Sylvain Lamure, MD, of Montellier (France) University, and colleagues.
The study examined a series of 89 consecutive patients from three French regions who had lymphoma and were hospitalized for COVID-19 in March and April 2020. The population was homogeneous; most patients were diagnosed with B-cell non-Hodgkin lymphoma (NHL) and had been treated for their lymphoma within 1 year.
Promising results for many
There were a significant associations between 30-day mortality and increasing age (over age 70 years) and r/r lymphoma. However, in the absence of those factors, mortality of the lymphoma patients with COVID-19 was comparable with that of the reference French COVID-19 population. In addition, there was no significant impact of active lymphoma treatment that had been given within 1 year, except for those patients who received bendamustine, which was associated with greater mortality, according to the researchers.
With a median follow-up of 33 days from admission, the Kaplan-Meier estimate of 30-day overall survival was 71% (95% confidence interval, 62%-81%). According to histological type of the lymphoma, 30-day overall survival rates were 80% (95% CI, 45%-100%) for Hodgkin lymphoma, 71% (95% CI, 61%-82%) for B-cell non-Hodgkin Lymphoma, and 71% (95% CI, 38%-100%) for T-cell non-Hodgkin Lymphoma.
The main factors associated with mortality were age 70 years and older (hazard ratio, 3.78; 95% CI, 1.73-8.25; P = .0009), hypertension (HR, 2.20; 95% CI, 1.06-4.59; P = .03), previous cancer (HR, 2.11; 95% CI, 0.90-4.92; P = .08), use of bendamustine within 12 months before admission to hospital (HR, 3.05; 95% CI, 1.31-7.11; P = .01), and r/r lymphoma (HR, 2.62; 95% CI, 1.20-5.72; P = .02).
Overall, the Kaplan-Meier estimates of 30-day overall survival were 61% for patients with r/r lymphoma, 52% in patients age 70 years with non–r/r lymphoma, and 88% for patients younger than 70 years with non–r/r, which was comparable with general population survival data among French populations, according to the researchers.
“Longer term clinical follow-up and biological monitoring of immune responses is warranted to explore the impact of lymphoma and its treatment on the immunity and prolonged outcome of Covid-19 patients,” they concluded.
The study was unsponsored. Several of the authors reported financial relationships with a number of biotechnology and pharmaceutical companies.
SOURCE: Lamure S et al. EClinicalMedicine. 2020 Oct 12. doi: 10.1016/j.eclinm.2020.100549.
Patients with B-cell lymphoma are immunocompromised because of the disease and its treatments. This presents the question of their outcomes upon infection with SARS-CoV-2. Researchers assessed the characteristics of patients with lymphoma hospitalized for COVID-19 and analyzed determinants of mortality in a retrospective database study. The investigators looked at data from adult patients with lymphoma who were hospitalized for COVID-19 in March and April 2020 in three French regions.
Older age and relapsed/refractory (r/r) disease in B-cell lymphoma patients were both found to be independent risk factors of increased death rate from COVID-19, according to the online report in EClinicalMedicine, published by The Lancet.
These results encourage “the application of standard Covid-19 treatment, including intubation, for lymphoma patients with Covid-19 lymphoma diagnosis, under first- or second-line chemotherapy, or in remission,” according to Sylvain Lamure, MD, of Montellier (France) University, and colleagues.
The study examined a series of 89 consecutive patients from three French regions who had lymphoma and were hospitalized for COVID-19 in March and April 2020. The population was homogeneous; most patients were diagnosed with B-cell non-Hodgkin lymphoma (NHL) and had been treated for their lymphoma within 1 year.
Promising results for many
There were a significant associations between 30-day mortality and increasing age (over age 70 years) and r/r lymphoma. However, in the absence of those factors, mortality of the lymphoma patients with COVID-19 was comparable with that of the reference French COVID-19 population. In addition, there was no significant impact of active lymphoma treatment that had been given within 1 year, except for those patients who received bendamustine, which was associated with greater mortality, according to the researchers.
With a median follow-up of 33 days from admission, the Kaplan-Meier estimate of 30-day overall survival was 71% (95% confidence interval, 62%-81%). According to histological type of the lymphoma, 30-day overall survival rates were 80% (95% CI, 45%-100%) for Hodgkin lymphoma, 71% (95% CI, 61%-82%) for B-cell non-Hodgkin Lymphoma, and 71% (95% CI, 38%-100%) for T-cell non-Hodgkin Lymphoma.
The main factors associated with mortality were age 70 years and older (hazard ratio, 3.78; 95% CI, 1.73-8.25; P = .0009), hypertension (HR, 2.20; 95% CI, 1.06-4.59; P = .03), previous cancer (HR, 2.11; 95% CI, 0.90-4.92; P = .08), use of bendamustine within 12 months before admission to hospital (HR, 3.05; 95% CI, 1.31-7.11; P = .01), and r/r lymphoma (HR, 2.62; 95% CI, 1.20-5.72; P = .02).
Overall, the Kaplan-Meier estimates of 30-day overall survival were 61% for patients with r/r lymphoma, 52% in patients age 70 years with non–r/r lymphoma, and 88% for patients younger than 70 years with non–r/r, which was comparable with general population survival data among French populations, according to the researchers.
“Longer term clinical follow-up and biological monitoring of immune responses is warranted to explore the impact of lymphoma and its treatment on the immunity and prolonged outcome of Covid-19 patients,” they concluded.
The study was unsponsored. Several of the authors reported financial relationships with a number of biotechnology and pharmaceutical companies.
SOURCE: Lamure S et al. EClinicalMedicine. 2020 Oct 12. doi: 10.1016/j.eclinm.2020.100549.
Patients with B-cell lymphoma are immunocompromised because of the disease and its treatments. This presents the question of their outcomes upon infection with SARS-CoV-2. Researchers assessed the characteristics of patients with lymphoma hospitalized for COVID-19 and analyzed determinants of mortality in a retrospective database study. The investigators looked at data from adult patients with lymphoma who were hospitalized for COVID-19 in March and April 2020 in three French regions.
Older age and relapsed/refractory (r/r) disease in B-cell lymphoma patients were both found to be independent risk factors of increased death rate from COVID-19, according to the online report in EClinicalMedicine, published by The Lancet.
These results encourage “the application of standard Covid-19 treatment, including intubation, for lymphoma patients with Covid-19 lymphoma diagnosis, under first- or second-line chemotherapy, or in remission,” according to Sylvain Lamure, MD, of Montellier (France) University, and colleagues.
The study examined a series of 89 consecutive patients from three French regions who had lymphoma and were hospitalized for COVID-19 in March and April 2020. The population was homogeneous; most patients were diagnosed with B-cell non-Hodgkin lymphoma (NHL) and had been treated for their lymphoma within 1 year.
Promising results for many
There were a significant associations between 30-day mortality and increasing age (over age 70 years) and r/r lymphoma. However, in the absence of those factors, mortality of the lymphoma patients with COVID-19 was comparable with that of the reference French COVID-19 population. In addition, there was no significant impact of active lymphoma treatment that had been given within 1 year, except for those patients who received bendamustine, which was associated with greater mortality, according to the researchers.
With a median follow-up of 33 days from admission, the Kaplan-Meier estimate of 30-day overall survival was 71% (95% confidence interval, 62%-81%). According to histological type of the lymphoma, 30-day overall survival rates were 80% (95% CI, 45%-100%) for Hodgkin lymphoma, 71% (95% CI, 61%-82%) for B-cell non-Hodgkin Lymphoma, and 71% (95% CI, 38%-100%) for T-cell non-Hodgkin Lymphoma.
The main factors associated with mortality were age 70 years and older (hazard ratio, 3.78; 95% CI, 1.73-8.25; P = .0009), hypertension (HR, 2.20; 95% CI, 1.06-4.59; P = .03), previous cancer (HR, 2.11; 95% CI, 0.90-4.92; P = .08), use of bendamustine within 12 months before admission to hospital (HR, 3.05; 95% CI, 1.31-7.11; P = .01), and r/r lymphoma (HR, 2.62; 95% CI, 1.20-5.72; P = .02).
Overall, the Kaplan-Meier estimates of 30-day overall survival were 61% for patients with r/r lymphoma, 52% in patients age 70 years with non–r/r lymphoma, and 88% for patients younger than 70 years with non–r/r, which was comparable with general population survival data among French populations, according to the researchers.
“Longer term clinical follow-up and biological monitoring of immune responses is warranted to explore the impact of lymphoma and its treatment on the immunity and prolonged outcome of Covid-19 patients,” they concluded.
The study was unsponsored. Several of the authors reported financial relationships with a number of biotechnology and pharmaceutical companies.
SOURCE: Lamure S et al. EClinicalMedicine. 2020 Oct 12. doi: 10.1016/j.eclinm.2020.100549.
FROM ECLINICALMEDICINE