User login
Vemurafenib's Efficacy in Melanoma Wanes Over Time
CHICAGO – Vemurafenib has a persistent survival benefit when given as first-line therapy for advanced BRAF-mutant melanoma, but the magnitude of benefit diminishes over time, according to an update of the BRIM-3 randomized trial.
With a median follow-up of about a year, vemurafenib was associated with a 30% relative reduction in the risk of death, relative to dacarbazine, presenting author Dr. Paul Chapman reported at the annual meeting of the American Society of Clinical Oncology. This is roughly half of the 63% relative risk reduction seen at a median follow-up of about 3 months, as reported at last year’s meeting.
The safety profile of vemurafenib was largely consistent with the earlier experience; the main high-grade adverse effects were skin toxicity (seen in about a third of patients) and liver toxicity (seen in about a tenth). But a few patients have developed second primary melanomas.
The observed temporal trend for overall survival benefit suggests "perhaps that most of the benefit in overall survival may be due to decreasing early deaths," said Dr. Chapman, an oncologist at Memorial Sloan-Kettering Cancer Center in New York.
Discussant Dr. Michael B. Atkins of the Lombardi Comprehensive Cancer Center at Georgetown University in Washington maintained that "the mature overall survival data [confirm] that vemurafenib is better than dacarbazine," and noted that it was "reassuring" that findings were similar, whether patients who crossed over to the vemurafenib arm were censored or not, "probably the most stringent test of survival."
However, he called attention to the fact that the median survival of 13-plus months with vemurafenib seen in BRIM-3 falls short of the 16-plus months seen in BRIM-2, a phase II trial in patients with previously treated advanced melanoma. "Did first-line patients have more aggressive disease and therefore fall off early?" he wondered. "Alternatively, was there less continuation of vemurafenib after progressive disease in the BRIM-3 trial relative to the BRIM-2 trial? If that was the case, some sort of confirmatory analysis should be done to see whether that can impact overall survival."
The decrease in vemurafenib efficacy seen over time might have several explanations, Dr. Atkins proposed. "Is it that the early, more robust hazard ratio was a manifestation of the so-called Lazarus effect, that these targeted therapies can cause patients who are really in bad shape and would have a short survival to have their survival prolonged, even though it was less than the survival of patients with less-aggressive tumors?" he wondered, echoing Dr. Chapman’s suggestion. Alternatively, maturation of the survival data might have contributed to the change in efficacy in patients with stage IIIC, M1a, and M1b disease who did not appear to benefit from vemurafenib.
In the larger context, the findings raise an important question pertaining to the clinical trials process generally, according to Dr. Atkins: "What does this say about the early analysis of trials like this, and reporting forever more that the hazard ratio is 0.35 or so, rather than what the more accurate 0.70 is?"
A Genentech spokesperson said that "due to differences in the study populations and trial designs, the results from these trials cannot be directly compared. BRIM3 was a global, randomized, Phase III trial comparing Zelboraf (vemurafenib) to DTIC chemotherapy in people with newly diagnosed BRAF V600 mutation-positive metastatic melanoma, while BRIM2 was a Phase II study that evaluated patients with previously treated disease."
The 675 patients in BRIM-3 had stage IIIC or IV melanoma harboring the BRAF V600E mutation. They were assigned evenly to intravenous dacarbazine or oral vemurafenib, the only targeted agent approved specifically for treating BRAF-mutant melanoma.
Large proportions of patients in both the vemurafenib and dacarbazine arms (36% and 44%, respectively) received other anticancer therapies after coming off the trial, Dr. Chapman reported. About a fifth of all patients in each arm went on to receive ipilimumab, and a fourth in the dacarbazine arm crossed over to vemurafenib.
Updated results – after a median follow-up of 9.5 months in the dacarbazine arm and 12.5 months in the vemurafenib arm – showed persistence of a progression-free survival benefit in favor of vemurafenib with censoring of patients at crossover (6.9 vs. 1.6 months; hazard ratio, 0.38; P less than .001).
There was also still an improvement in overall survival, regardless of whether patients were censored at crossover (13.6 vs. 9.7 months; HR, 0.70; P less than .001) or not (13.6 vs. 10.3 months; HR, 0.76; P less than .01).
The leading grade 3 or worse adverse events with vemurafenib remained cutaneous squamous cell carcinomas (seen in 19% of patients), keratoacanthomas (10%), and elevations of liver function tests (10%). In addition, eight patients developed new primary melanomas.
Dr. Chapman disclosed that he receives honoraria and research funding from Roche/Genentech. The trial was sponsored by Hoffman-LaRoche. Dr. Atkins disclosed that he is a consultant to Bristol-Myers Squibb, Celgene, Curetech, Genentech, Merck, Novartis, and Prometheus.
This article was updated on July 3, 2012.
CHICAGO – Vemurafenib has a persistent survival benefit when given as first-line therapy for advanced BRAF-mutant melanoma, but the magnitude of benefit diminishes over time, according to an update of the BRIM-3 randomized trial.
With a median follow-up of about a year, vemurafenib was associated with a 30% relative reduction in the risk of death, relative to dacarbazine, presenting author Dr. Paul Chapman reported at the annual meeting of the American Society of Clinical Oncology. This is roughly half of the 63% relative risk reduction seen at a median follow-up of about 3 months, as reported at last year’s meeting.
The safety profile of vemurafenib was largely consistent with the earlier experience; the main high-grade adverse effects were skin toxicity (seen in about a third of patients) and liver toxicity (seen in about a tenth). But a few patients have developed second primary melanomas.
The observed temporal trend for overall survival benefit suggests "perhaps that most of the benefit in overall survival may be due to decreasing early deaths," said Dr. Chapman, an oncologist at Memorial Sloan-Kettering Cancer Center in New York.
Discussant Dr. Michael B. Atkins of the Lombardi Comprehensive Cancer Center at Georgetown University in Washington maintained that "the mature overall survival data [confirm] that vemurafenib is better than dacarbazine," and noted that it was "reassuring" that findings were similar, whether patients who crossed over to the vemurafenib arm were censored or not, "probably the most stringent test of survival."
However, he called attention to the fact that the median survival of 13-plus months with vemurafenib seen in BRIM-3 falls short of the 16-plus months seen in BRIM-2, a phase II trial in patients with previously treated advanced melanoma. "Did first-line patients have more aggressive disease and therefore fall off early?" he wondered. "Alternatively, was there less continuation of vemurafenib after progressive disease in the BRIM-3 trial relative to the BRIM-2 trial? If that was the case, some sort of confirmatory analysis should be done to see whether that can impact overall survival."
The decrease in vemurafenib efficacy seen over time might have several explanations, Dr. Atkins proposed. "Is it that the early, more robust hazard ratio was a manifestation of the so-called Lazarus effect, that these targeted therapies can cause patients who are really in bad shape and would have a short survival to have their survival prolonged, even though it was less than the survival of patients with less-aggressive tumors?" he wondered, echoing Dr. Chapman’s suggestion. Alternatively, maturation of the survival data might have contributed to the change in efficacy in patients with stage IIIC, M1a, and M1b disease who did not appear to benefit from vemurafenib.
In the larger context, the findings raise an important question pertaining to the clinical trials process generally, according to Dr. Atkins: "What does this say about the early analysis of trials like this, and reporting forever more that the hazard ratio is 0.35 or so, rather than what the more accurate 0.70 is?"
A Genentech spokesperson said that "due to differences in the study populations and trial designs, the results from these trials cannot be directly compared. BRIM3 was a global, randomized, Phase III trial comparing Zelboraf (vemurafenib) to DTIC chemotherapy in people with newly diagnosed BRAF V600 mutation-positive metastatic melanoma, while BRIM2 was a Phase II study that evaluated patients with previously treated disease."
The 675 patients in BRIM-3 had stage IIIC or IV melanoma harboring the BRAF V600E mutation. They were assigned evenly to intravenous dacarbazine or oral vemurafenib, the only targeted agent approved specifically for treating BRAF-mutant melanoma.
Large proportions of patients in both the vemurafenib and dacarbazine arms (36% and 44%, respectively) received other anticancer therapies after coming off the trial, Dr. Chapman reported. About a fifth of all patients in each arm went on to receive ipilimumab, and a fourth in the dacarbazine arm crossed over to vemurafenib.
Updated results – after a median follow-up of 9.5 months in the dacarbazine arm and 12.5 months in the vemurafenib arm – showed persistence of a progression-free survival benefit in favor of vemurafenib with censoring of patients at crossover (6.9 vs. 1.6 months; hazard ratio, 0.38; P less than .001).
There was also still an improvement in overall survival, regardless of whether patients were censored at crossover (13.6 vs. 9.7 months; HR, 0.70; P less than .001) or not (13.6 vs. 10.3 months; HR, 0.76; P less than .01).
The leading grade 3 or worse adverse events with vemurafenib remained cutaneous squamous cell carcinomas (seen in 19% of patients), keratoacanthomas (10%), and elevations of liver function tests (10%). In addition, eight patients developed new primary melanomas.
Dr. Chapman disclosed that he receives honoraria and research funding from Roche/Genentech. The trial was sponsored by Hoffman-LaRoche. Dr. Atkins disclosed that he is a consultant to Bristol-Myers Squibb, Celgene, Curetech, Genentech, Merck, Novartis, and Prometheus.
This article was updated on July 3, 2012.
CHICAGO – Vemurafenib has a persistent survival benefit when given as first-line therapy for advanced BRAF-mutant melanoma, but the magnitude of benefit diminishes over time, according to an update of the BRIM-3 randomized trial.
With a median follow-up of about a year, vemurafenib was associated with a 30% relative reduction in the risk of death, relative to dacarbazine, presenting author Dr. Paul Chapman reported at the annual meeting of the American Society of Clinical Oncology. This is roughly half of the 63% relative risk reduction seen at a median follow-up of about 3 months, as reported at last year’s meeting.
The safety profile of vemurafenib was largely consistent with the earlier experience; the main high-grade adverse effects were skin toxicity (seen in about a third of patients) and liver toxicity (seen in about a tenth). But a few patients have developed second primary melanomas.
The observed temporal trend for overall survival benefit suggests "perhaps that most of the benefit in overall survival may be due to decreasing early deaths," said Dr. Chapman, an oncologist at Memorial Sloan-Kettering Cancer Center in New York.
Discussant Dr. Michael B. Atkins of the Lombardi Comprehensive Cancer Center at Georgetown University in Washington maintained that "the mature overall survival data [confirm] that vemurafenib is better than dacarbazine," and noted that it was "reassuring" that findings were similar, whether patients who crossed over to the vemurafenib arm were censored or not, "probably the most stringent test of survival."
However, he called attention to the fact that the median survival of 13-plus months with vemurafenib seen in BRIM-3 falls short of the 16-plus months seen in BRIM-2, a phase II trial in patients with previously treated advanced melanoma. "Did first-line patients have more aggressive disease and therefore fall off early?" he wondered. "Alternatively, was there less continuation of vemurafenib after progressive disease in the BRIM-3 trial relative to the BRIM-2 trial? If that was the case, some sort of confirmatory analysis should be done to see whether that can impact overall survival."
The decrease in vemurafenib efficacy seen over time might have several explanations, Dr. Atkins proposed. "Is it that the early, more robust hazard ratio was a manifestation of the so-called Lazarus effect, that these targeted therapies can cause patients who are really in bad shape and would have a short survival to have their survival prolonged, even though it was less than the survival of patients with less-aggressive tumors?" he wondered, echoing Dr. Chapman’s suggestion. Alternatively, maturation of the survival data might have contributed to the change in efficacy in patients with stage IIIC, M1a, and M1b disease who did not appear to benefit from vemurafenib.
In the larger context, the findings raise an important question pertaining to the clinical trials process generally, according to Dr. Atkins: "What does this say about the early analysis of trials like this, and reporting forever more that the hazard ratio is 0.35 or so, rather than what the more accurate 0.70 is?"
A Genentech spokesperson said that "due to differences in the study populations and trial designs, the results from these trials cannot be directly compared. BRIM3 was a global, randomized, Phase III trial comparing Zelboraf (vemurafenib) to DTIC chemotherapy in people with newly diagnosed BRAF V600 mutation-positive metastatic melanoma, while BRIM2 was a Phase II study that evaluated patients with previously treated disease."
The 675 patients in BRIM-3 had stage IIIC or IV melanoma harboring the BRAF V600E mutation. They were assigned evenly to intravenous dacarbazine or oral vemurafenib, the only targeted agent approved specifically for treating BRAF-mutant melanoma.
Large proportions of patients in both the vemurafenib and dacarbazine arms (36% and 44%, respectively) received other anticancer therapies after coming off the trial, Dr. Chapman reported. About a fifth of all patients in each arm went on to receive ipilimumab, and a fourth in the dacarbazine arm crossed over to vemurafenib.
Updated results – after a median follow-up of 9.5 months in the dacarbazine arm and 12.5 months in the vemurafenib arm – showed persistence of a progression-free survival benefit in favor of vemurafenib with censoring of patients at crossover (6.9 vs. 1.6 months; hazard ratio, 0.38; P less than .001).
There was also still an improvement in overall survival, regardless of whether patients were censored at crossover (13.6 vs. 9.7 months; HR, 0.70; P less than .001) or not (13.6 vs. 10.3 months; HR, 0.76; P less than .01).
The leading grade 3 or worse adverse events with vemurafenib remained cutaneous squamous cell carcinomas (seen in 19% of patients), keratoacanthomas (10%), and elevations of liver function tests (10%). In addition, eight patients developed new primary melanomas.
Dr. Chapman disclosed that he receives honoraria and research funding from Roche/Genentech. The trial was sponsored by Hoffman-LaRoche. Dr. Atkins disclosed that he is a consultant to Bristol-Myers Squibb, Celgene, Curetech, Genentech, Merck, Novartis, and Prometheus.
This article was updated on July 3, 2012.
AT THE ANNUAL MEETING OF THE AMERICAN SOCIETY OF CLINICAL ONCOLOGY
Major Finding: At a median follow-up of 9.5-12.5 months, vemurafenib conferred a 30% reduction in the risk of death vs. dacarbazine, about half the 63% reduction reported at a median follow-up of 2.3-3.8 months.
Data Source: The phase III, randomized BRIM-3 trial randomized 675 patients with V600E BRAF-mutant melanoma.
Disclosures: Dr. Chapman disclosed that he receives honoraria and research funding from Roche/Genentech. The trial was sponsored by Hoffman-LaRoche. Dr. Atkins disclosed that he is a consultant to Bristol-Myers Squibb, Celgene, Curetech, Genentech, Merck, Novartis, and Prometheus.
Last Case
Dear Intern:
Well, your elective is over. So is medical school, since this is your last rotation. I get nostalgic this time of year, having graduated 40 years ago, which I’m sure you’ll agree is a ridiculously long time. Because you matched at your first choice, you are now well and truly launched.
One of my recurring sentimental notions is to note a student’s last case in medical school. Will this encounter be noteworthy; or will the whole thing end, so to speak, not with a bang but a whimper?
Looking for significance in a last case is similar to quoting people’s last words. The famous ones you hear about are usually edited, interpolated from some earlier occasion, or made up altogether. If you’ve seen people breathe their last, you know that most are not able to say much of anything, nor are their families, should they happen to be there, in any frame of mind to absorb enduring wisdom.
But lo and behold – our last encounter together on this elective has indeed turned out to be unusual, perhaps even memorable. We should discuss it.
A biopsy report just came back via fax, with the heading, "Crucial Clinical Information." We saw this patient together yesterday. Truong was the Vietnamese man in his mid-20s. A mole on his back started to grow. It got itchy, he said. It bled twice.
We examined the mole in question, along with the rest of him. As you recall, he had several moles, all black like the one in question. The irritated one was regular in outline, except for a scab in the center, consistent with having been scratched.
You remember that I was not very concerned. I said that raised moles often worry patients but aren’t usually a problem. Bleeding, moreover, is mostly a sign of minor, unrecalled trauma, a scratch while sleeping or something like that. As you’ve seen this month, patients troop in daily to complain that this mole is itchy or that one has bled. I often take these lesions off to reduce anxiety as to make the diagnosis. As a prospective primary physician, you will have to make judgments about itchy moles all the time.
I asked Truong how much the irritation bothered him. He waffled. I did the biopsy.
Here is the report: malignant melanoma, level IV, thickness at least 2.1 mm.
This is what runs through my head as I read this: At this point in my career, I am as good at judging moles as I am ever going to be. I am as clinically astute as I am ever going to be. And yet, here I am, still managing to almost miss a diagnosis in which the patient’s well-being, even his life, depends on my getting it right. In how many other cases have I (and the patient) not been so lucky? How would I know?
What lessons can I draw from this experience, and which ones can I pass on to you at your very different stage of clinical life? That because certainty is unattainable, we should take off every mole just to be sure? Every mole a patient points to? If we did that, would we be better doctors? Would we be treating patients, or our own insecurity?
It has always seemed to me that in medical practice and, in general, it is best to avoid the extremes, to be neither cavalier nor paranoid. Every passerby may assault you, every neighbor (or patient) may sue you, but it’s no good going through life treating everybody as a potential mugger or litigant. You won’t have many friends if you do that, and your patients will have a lot of unnecessary scars on their skin.
There really is something to the old saying: smart is good, lucky is better. Difficult or atypical cases can make the clever and diligent doctor look like a fool, or worse.
So perhaps your last case in medical school will come to mind from time to time as your career advances. Work hard, study hard, pay attention; become as good as you possibly can. But don’t ever get too comfortable. We try our best, but nobody always gets it right.
Good luck!
To respond to this column, e-mail Dr. Rockoff at our editorial offices at [email protected].
Dear Intern:
Well, your elective is over. So is medical school, since this is your last rotation. I get nostalgic this time of year, having graduated 40 years ago, which I’m sure you’ll agree is a ridiculously long time. Because you matched at your first choice, you are now well and truly launched.
One of my recurring sentimental notions is to note a student’s last case in medical school. Will this encounter be noteworthy; or will the whole thing end, so to speak, not with a bang but a whimper?
Looking for significance in a last case is similar to quoting people’s last words. The famous ones you hear about are usually edited, interpolated from some earlier occasion, or made up altogether. If you’ve seen people breathe their last, you know that most are not able to say much of anything, nor are their families, should they happen to be there, in any frame of mind to absorb enduring wisdom.
But lo and behold – our last encounter together on this elective has indeed turned out to be unusual, perhaps even memorable. We should discuss it.
A biopsy report just came back via fax, with the heading, "Crucial Clinical Information." We saw this patient together yesterday. Truong was the Vietnamese man in his mid-20s. A mole on his back started to grow. It got itchy, he said. It bled twice.
We examined the mole in question, along with the rest of him. As you recall, he had several moles, all black like the one in question. The irritated one was regular in outline, except for a scab in the center, consistent with having been scratched.
You remember that I was not very concerned. I said that raised moles often worry patients but aren’t usually a problem. Bleeding, moreover, is mostly a sign of minor, unrecalled trauma, a scratch while sleeping or something like that. As you’ve seen this month, patients troop in daily to complain that this mole is itchy or that one has bled. I often take these lesions off to reduce anxiety as to make the diagnosis. As a prospective primary physician, you will have to make judgments about itchy moles all the time.
I asked Truong how much the irritation bothered him. He waffled. I did the biopsy.
Here is the report: malignant melanoma, level IV, thickness at least 2.1 mm.
This is what runs through my head as I read this: At this point in my career, I am as good at judging moles as I am ever going to be. I am as clinically astute as I am ever going to be. And yet, here I am, still managing to almost miss a diagnosis in which the patient’s well-being, even his life, depends on my getting it right. In how many other cases have I (and the patient) not been so lucky? How would I know?
What lessons can I draw from this experience, and which ones can I pass on to you at your very different stage of clinical life? That because certainty is unattainable, we should take off every mole just to be sure? Every mole a patient points to? If we did that, would we be better doctors? Would we be treating patients, or our own insecurity?
It has always seemed to me that in medical practice and, in general, it is best to avoid the extremes, to be neither cavalier nor paranoid. Every passerby may assault you, every neighbor (or patient) may sue you, but it’s no good going through life treating everybody as a potential mugger or litigant. You won’t have many friends if you do that, and your patients will have a lot of unnecessary scars on their skin.
There really is something to the old saying: smart is good, lucky is better. Difficult or atypical cases can make the clever and diligent doctor look like a fool, or worse.
So perhaps your last case in medical school will come to mind from time to time as your career advances. Work hard, study hard, pay attention; become as good as you possibly can. But don’t ever get too comfortable. We try our best, but nobody always gets it right.
Good luck!
To respond to this column, e-mail Dr. Rockoff at our editorial offices at [email protected].
Dear Intern:
Well, your elective is over. So is medical school, since this is your last rotation. I get nostalgic this time of year, having graduated 40 years ago, which I’m sure you’ll agree is a ridiculously long time. Because you matched at your first choice, you are now well and truly launched.
One of my recurring sentimental notions is to note a student’s last case in medical school. Will this encounter be noteworthy; or will the whole thing end, so to speak, not with a bang but a whimper?
Looking for significance in a last case is similar to quoting people’s last words. The famous ones you hear about are usually edited, interpolated from some earlier occasion, or made up altogether. If you’ve seen people breathe their last, you know that most are not able to say much of anything, nor are their families, should they happen to be there, in any frame of mind to absorb enduring wisdom.
But lo and behold – our last encounter together on this elective has indeed turned out to be unusual, perhaps even memorable. We should discuss it.
A biopsy report just came back via fax, with the heading, "Crucial Clinical Information." We saw this patient together yesterday. Truong was the Vietnamese man in his mid-20s. A mole on his back started to grow. It got itchy, he said. It bled twice.
We examined the mole in question, along with the rest of him. As you recall, he had several moles, all black like the one in question. The irritated one was regular in outline, except for a scab in the center, consistent with having been scratched.
You remember that I was not very concerned. I said that raised moles often worry patients but aren’t usually a problem. Bleeding, moreover, is mostly a sign of minor, unrecalled trauma, a scratch while sleeping or something like that. As you’ve seen this month, patients troop in daily to complain that this mole is itchy or that one has bled. I often take these lesions off to reduce anxiety as to make the diagnosis. As a prospective primary physician, you will have to make judgments about itchy moles all the time.
I asked Truong how much the irritation bothered him. He waffled. I did the biopsy.
Here is the report: malignant melanoma, level IV, thickness at least 2.1 mm.
This is what runs through my head as I read this: At this point in my career, I am as good at judging moles as I am ever going to be. I am as clinically astute as I am ever going to be. And yet, here I am, still managing to almost miss a diagnosis in which the patient’s well-being, even his life, depends on my getting it right. In how many other cases have I (and the patient) not been so lucky? How would I know?
What lessons can I draw from this experience, and which ones can I pass on to you at your very different stage of clinical life? That because certainty is unattainable, we should take off every mole just to be sure? Every mole a patient points to? If we did that, would we be better doctors? Would we be treating patients, or our own insecurity?
It has always seemed to me that in medical practice and, in general, it is best to avoid the extremes, to be neither cavalier nor paranoid. Every passerby may assault you, every neighbor (or patient) may sue you, but it’s no good going through life treating everybody as a potential mugger or litigant. You won’t have many friends if you do that, and your patients will have a lot of unnecessary scars on their skin.
There really is something to the old saying: smart is good, lucky is better. Difficult or atypical cases can make the clever and diligent doctor look like a fool, or worse.
So perhaps your last case in medical school will come to mind from time to time as your career advances. Work hard, study hard, pay attention; become as good as you possibly can. But don’t ever get too comfortable. We try our best, but nobody always gets it right.
Good luck!
To respond to this column, e-mail Dr. Rockoff at our editorial offices at [email protected].
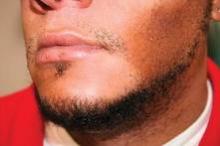
Melanoma in the Skin of Color Population
Dr. Rossi discusses melanoma in the skin of color population including the incidence and presentation of melanoma as well as factors contributing to delayed diagnosis in these patients. He also gives recommendations to physicians to help educate their patients. For more information, read Dr. Rossi's article in the May 2012 issue, "Melanoma in Skin of Color."
Dr. Rossi discusses melanoma in the skin of color population including the incidence and presentation of melanoma as well as factors contributing to delayed diagnosis in these patients. He also gives recommendations to physicians to help educate their patients. For more information, read Dr. Rossi's article in the May 2012 issue, "Melanoma in Skin of Color."
Dr. Rossi discusses melanoma in the skin of color population including the incidence and presentation of melanoma as well as factors contributing to delayed diagnosis in these patients. He also gives recommendations to physicians to help educate their patients. For more information, read Dr. Rossi's article in the May 2012 issue, "Melanoma in Skin of Color."
Rhode Island Passes Tan Ban
Rhode Island joins California and Vermont to become the third state to institute restrictions on the use of tanning beds by minors.
There had been an effort to completely ban the use of tanning beds by anyone under the age of 18, but a compromise was struck. In the final bill, which passed into law without the governor's signature, minors cannot use indoor tanning beds without a prescription from a physician for ultraviolet radiation therapy or a signed consent from a parent or guardian. The parental consent has to be signed in the presence of a tanning salon employee, and it essentially has the parent acknowledge that UV radiation is a carcinogen and that exposure increases the probability of developing melanoma.
The American Academy of Dermatology applauded the state's action, and noted that in Rhode Island the incidence of melanoma in women 15 years and older increased by 24% between 2004 and 2008.
"Prevention is one of the most valuable tools that we have as dermatologists. We need to continue educating patients about the risks of indoor tanning and encouraging healthy decisions to help prevent skin cancer," Dr. Daniel M. Siegel, president of the AAD, said in a statement.
"This law is similar to bans in California and Vermont that will protect children and adolescents from the health hazards of indoor tanning and sends a strong message from the state that tanning is a dangerous behavior and should be avoided," said Dr. Siegel.
Rhode Island joins California and Vermont to become the third state to institute restrictions on the use of tanning beds by minors.
There had been an effort to completely ban the use of tanning beds by anyone under the age of 18, but a compromise was struck. In the final bill, which passed into law without the governor's signature, minors cannot use indoor tanning beds without a prescription from a physician for ultraviolet radiation therapy or a signed consent from a parent or guardian. The parental consent has to be signed in the presence of a tanning salon employee, and it essentially has the parent acknowledge that UV radiation is a carcinogen and that exposure increases the probability of developing melanoma.
The American Academy of Dermatology applauded the state's action, and noted that in Rhode Island the incidence of melanoma in women 15 years and older increased by 24% between 2004 and 2008.
"Prevention is one of the most valuable tools that we have as dermatologists. We need to continue educating patients about the risks of indoor tanning and encouraging healthy decisions to help prevent skin cancer," Dr. Daniel M. Siegel, president of the AAD, said in a statement.
"This law is similar to bans in California and Vermont that will protect children and adolescents from the health hazards of indoor tanning and sends a strong message from the state that tanning is a dangerous behavior and should be avoided," said Dr. Siegel.
Rhode Island joins California and Vermont to become the third state to institute restrictions on the use of tanning beds by minors.
There had been an effort to completely ban the use of tanning beds by anyone under the age of 18, but a compromise was struck. In the final bill, which passed into law without the governor's signature, minors cannot use indoor tanning beds without a prescription from a physician for ultraviolet radiation therapy or a signed consent from a parent or guardian. The parental consent has to be signed in the presence of a tanning salon employee, and it essentially has the parent acknowledge that UV radiation is a carcinogen and that exposure increases the probability of developing melanoma.
The American Academy of Dermatology applauded the state's action, and noted that in Rhode Island the incidence of melanoma in women 15 years and older increased by 24% between 2004 and 2008.
"Prevention is one of the most valuable tools that we have as dermatologists. We need to continue educating patients about the risks of indoor tanning and encouraging healthy decisions to help prevent skin cancer," Dr. Daniel M. Siegel, president of the AAD, said in a statement.
"This law is similar to bans in California and Vermont that will protect children and adolescents from the health hazards of indoor tanning and sends a strong message from the state that tanning is a dangerous behavior and should be avoided," said Dr. Siegel.
Don't Disregard High-Dose Brachytherapy for Skin Cancer
ORLANDO – Although Mohs surgery remains a mainstay of skin cancer treatment, some patients benefit from targeted brachytherapy of their basal or squamous cell carcinoma lesions, according to Dr. Michael E. Kasper.
Patients who are elderly, infirm, or on blood thinners are good candidates for noninvasive brachytherapy using high-dose, small surface applicators, Dr. Kasper said. This therapeutic strategy also works well for treating lesions in anatomic locations at risk for delayed surgical healing.
"We are treating various small lesions with these surface applicators," Dr. Kasper said. For example, superficial squamous cell carcinoma (SCC) lesions up to 2 cm can be targeted "where we feel comfortable about the visible margins," he said at the annual meeting of the Florida Society of Dermatology and Dermatologic Surgery.
"We also see patients who are tired of [invasive resection] or who are poor candidates for surgery, and that is really the bulk of our patients," Dr. Kasper said. He assured meeting attendees that his goal as a radiation oncologist is not to take skin cancer patients away from dermatologists. "We’re not interested in treating 40- or 50-year-olds and really competing," said Dr. Kasper of Lynn Cancer Institute at Boca Raton (Fla.) Regional Hospital. "We are interested in working with dermatologists and really helping you with those patients who might be neglected or who are at high risk of developing a serious recurrence."
Neglected patients may include nursing home residents who do not get medical attention for their skin cancer in its earlier stages, he said.
Available data point to good local control and cosmesis for a majority of patients, Dr. Kasper said. A typical patient might experience acute effects such as crusting and some mild erythema about 10 days to 2 weeks after brachytherapy of a well differentiated SCC lesion of the lower extremity. More brisk erythema also occurs in about 10%-15% of patients, Dr. Kasper said.
Late hypopigmentation also develops in about 10% of patients, he said. "We are also seeing a few telangiectasias, but it’s fairly mild."
Interpret postradiation therapy biopsies with caution, Dr. Kasper warned. Of the 240 patients treated to date at his institution over about 6.5 years, there were three documented recurrences, including a couple at 3 and 4 months. "While we are counting those as recurrences, they probably aren’t. That’s way too early to biopsy these lesions." Use discretion and ideally wait until you see clear progression prior to performing a postradiation biopsy, he said. "There certainly are false positives that occur due to delayed tumor regression. The cancer cells die when they reproduce and many of these are slow growing tumors. So we would not expect them to all be completely resolved at 3 or 4 months although clinically, on the surface, they can appear that way."
Historically brachytherapy was delivered as a low-dose treatment over a long period of time at many sessions. Low-dose brachytherapy is typically in the 0.4 to 2.0 Gy/hr range, medium dose is greater than 2 and up to 12 Gy/hr, and the high dose exceeds 12 Gy/hr.
Availability of high-dose rate brachytherapy was a "major breakthrough" because patients no longer had to lie in the hospital all weekend to receive treatment. "It was a bit controversial at first, but now there are really good data to show there are radiobiologic reasons why the high dose rate may actually be advantageous in killing cancer cells compared to this low trickle effect that we were using with low-dose regimens."
Dr. Kasper determined that up to 30 sessions of low-dose rate brachytherapy can be a real impediment to patient compliance and worked to design a safe and effective regimen delivered in fewer sessions. "We wanted to be at six treatments and worked backwards." Striking a balance between the dose-response rate and the potential late side effects was another consideration.
A meeting attendee asked about the relative cost of brachytherapy, compared with other treatment modalities. Brachytherapy is generally more expensive than Mohs surgery, Dr. Kasper replied. The avoidance of cancer recurrences will hopefully justify the higher initial costs, he said. He added that brachytherapy costs are coming down, more so in outpatient centers, compared with hospital settings.
Dr. Kasper said he receives consulting fees from Nucletron/Elekta.
ORLANDO – Although Mohs surgery remains a mainstay of skin cancer treatment, some patients benefit from targeted brachytherapy of their basal or squamous cell carcinoma lesions, according to Dr. Michael E. Kasper.
Patients who are elderly, infirm, or on blood thinners are good candidates for noninvasive brachytherapy using high-dose, small surface applicators, Dr. Kasper said. This therapeutic strategy also works well for treating lesions in anatomic locations at risk for delayed surgical healing.
"We are treating various small lesions with these surface applicators," Dr. Kasper said. For example, superficial squamous cell carcinoma (SCC) lesions up to 2 cm can be targeted "where we feel comfortable about the visible margins," he said at the annual meeting of the Florida Society of Dermatology and Dermatologic Surgery.
"We also see patients who are tired of [invasive resection] or who are poor candidates for surgery, and that is really the bulk of our patients," Dr. Kasper said. He assured meeting attendees that his goal as a radiation oncologist is not to take skin cancer patients away from dermatologists. "We’re not interested in treating 40- or 50-year-olds and really competing," said Dr. Kasper of Lynn Cancer Institute at Boca Raton (Fla.) Regional Hospital. "We are interested in working with dermatologists and really helping you with those patients who might be neglected or who are at high risk of developing a serious recurrence."
Neglected patients may include nursing home residents who do not get medical attention for their skin cancer in its earlier stages, he said.
Available data point to good local control and cosmesis for a majority of patients, Dr. Kasper said. A typical patient might experience acute effects such as crusting and some mild erythema about 10 days to 2 weeks after brachytherapy of a well differentiated SCC lesion of the lower extremity. More brisk erythema also occurs in about 10%-15% of patients, Dr. Kasper said.
Late hypopigmentation also develops in about 10% of patients, he said. "We are also seeing a few telangiectasias, but it’s fairly mild."
Interpret postradiation therapy biopsies with caution, Dr. Kasper warned. Of the 240 patients treated to date at his institution over about 6.5 years, there were three documented recurrences, including a couple at 3 and 4 months. "While we are counting those as recurrences, they probably aren’t. That’s way too early to biopsy these lesions." Use discretion and ideally wait until you see clear progression prior to performing a postradiation biopsy, he said. "There certainly are false positives that occur due to delayed tumor regression. The cancer cells die when they reproduce and many of these are slow growing tumors. So we would not expect them to all be completely resolved at 3 or 4 months although clinically, on the surface, they can appear that way."
Historically brachytherapy was delivered as a low-dose treatment over a long period of time at many sessions. Low-dose brachytherapy is typically in the 0.4 to 2.0 Gy/hr range, medium dose is greater than 2 and up to 12 Gy/hr, and the high dose exceeds 12 Gy/hr.
Availability of high-dose rate brachytherapy was a "major breakthrough" because patients no longer had to lie in the hospital all weekend to receive treatment. "It was a bit controversial at first, but now there are really good data to show there are radiobiologic reasons why the high dose rate may actually be advantageous in killing cancer cells compared to this low trickle effect that we were using with low-dose regimens."
Dr. Kasper determined that up to 30 sessions of low-dose rate brachytherapy can be a real impediment to patient compliance and worked to design a safe and effective regimen delivered in fewer sessions. "We wanted to be at six treatments and worked backwards." Striking a balance between the dose-response rate and the potential late side effects was another consideration.
A meeting attendee asked about the relative cost of brachytherapy, compared with other treatment modalities. Brachytherapy is generally more expensive than Mohs surgery, Dr. Kasper replied. The avoidance of cancer recurrences will hopefully justify the higher initial costs, he said. He added that brachytherapy costs are coming down, more so in outpatient centers, compared with hospital settings.
Dr. Kasper said he receives consulting fees from Nucletron/Elekta.
ORLANDO – Although Mohs surgery remains a mainstay of skin cancer treatment, some patients benefit from targeted brachytherapy of their basal or squamous cell carcinoma lesions, according to Dr. Michael E. Kasper.
Patients who are elderly, infirm, or on blood thinners are good candidates for noninvasive brachytherapy using high-dose, small surface applicators, Dr. Kasper said. This therapeutic strategy also works well for treating lesions in anatomic locations at risk for delayed surgical healing.
"We are treating various small lesions with these surface applicators," Dr. Kasper said. For example, superficial squamous cell carcinoma (SCC) lesions up to 2 cm can be targeted "where we feel comfortable about the visible margins," he said at the annual meeting of the Florida Society of Dermatology and Dermatologic Surgery.
"We also see patients who are tired of [invasive resection] or who are poor candidates for surgery, and that is really the bulk of our patients," Dr. Kasper said. He assured meeting attendees that his goal as a radiation oncologist is not to take skin cancer patients away from dermatologists. "We’re not interested in treating 40- or 50-year-olds and really competing," said Dr. Kasper of Lynn Cancer Institute at Boca Raton (Fla.) Regional Hospital. "We are interested in working with dermatologists and really helping you with those patients who might be neglected or who are at high risk of developing a serious recurrence."
Neglected patients may include nursing home residents who do not get medical attention for their skin cancer in its earlier stages, he said.
Available data point to good local control and cosmesis for a majority of patients, Dr. Kasper said. A typical patient might experience acute effects such as crusting and some mild erythema about 10 days to 2 weeks after brachytherapy of a well differentiated SCC lesion of the lower extremity. More brisk erythema also occurs in about 10%-15% of patients, Dr. Kasper said.
Late hypopigmentation also develops in about 10% of patients, he said. "We are also seeing a few telangiectasias, but it’s fairly mild."
Interpret postradiation therapy biopsies with caution, Dr. Kasper warned. Of the 240 patients treated to date at his institution over about 6.5 years, there were three documented recurrences, including a couple at 3 and 4 months. "While we are counting those as recurrences, they probably aren’t. That’s way too early to biopsy these lesions." Use discretion and ideally wait until you see clear progression prior to performing a postradiation biopsy, he said. "There certainly are false positives that occur due to delayed tumor regression. The cancer cells die when they reproduce and many of these are slow growing tumors. So we would not expect them to all be completely resolved at 3 or 4 months although clinically, on the surface, they can appear that way."
Historically brachytherapy was delivered as a low-dose treatment over a long period of time at many sessions. Low-dose brachytherapy is typically in the 0.4 to 2.0 Gy/hr range, medium dose is greater than 2 and up to 12 Gy/hr, and the high dose exceeds 12 Gy/hr.
Availability of high-dose rate brachytherapy was a "major breakthrough" because patients no longer had to lie in the hospital all weekend to receive treatment. "It was a bit controversial at first, but now there are really good data to show there are radiobiologic reasons why the high dose rate may actually be advantageous in killing cancer cells compared to this low trickle effect that we were using with low-dose regimens."
Dr. Kasper determined that up to 30 sessions of low-dose rate brachytherapy can be a real impediment to patient compliance and worked to design a safe and effective regimen delivered in fewer sessions. "We wanted to be at six treatments and worked backwards." Striking a balance between the dose-response rate and the potential late side effects was another consideration.
A meeting attendee asked about the relative cost of brachytherapy, compared with other treatment modalities. Brachytherapy is generally more expensive than Mohs surgery, Dr. Kasper replied. The avoidance of cancer recurrences will hopefully justify the higher initial costs, he said. He added that brachytherapy costs are coming down, more so in outpatient centers, compared with hospital settings.
Dr. Kasper said he receives consulting fees from Nucletron/Elekta.
EXPERT ANALYSIS FROM THE ANNUAL MEETING OF THE FLORIDA SOCIETY OF DERMATOLOGY AND DERMATOLOGIC SURGERY
Most Cancers Not Elevated in Vasculitis
BERLIN – Nonmelanoma skin cancer was the only type of malignancy found to occur at an increased rate in new 5-year follow-up data on 535 patients treated for systemic small-vessel vasculitis in four major randomized trials sponsored by the European Vasculitis Society.
With 2,650 person-years of prospective observation of 281 clinical trial participants treated for granulomatosis with polyangiitis (formerly called Wegener’s granulomatosis) and 254 treated for microscopic polyangiitis, patients with circulating antineutrophil cytoplasmic antibody (ANCA)-associated vasculitis had a 2.78-fold greater incidence of nonmelanoma skin cancer than did the matched general population, based upon data from the European cancer registry. But no other type of cancer was significantly increased, Dr. Kerstin W.A. Westman reported at the annual European Congress of Rheumatology.
This finding came as a pleasant surprise. Earlier publications by other investigators have reported broadly elevated malignancy risks in the years following administration of the various toxic, largely cyclophosphamide-based therapeutic regimens for ANCA-associated vasculitis, noted Dr. Westman, a nephrologist at Lund (Sweden) University.
For example, Danish investigators reported significantly increased rates of bladder and prostate cancer and acute myeloid leukemia as well as nonmelanoma skin cancer in a study featuring 2,121 person-years of follow-up (J. Rheumatol.2008;35:100-5). However, the Danish patients were treated during 1973-1999, while participants in the four European Vasculitis Society randomized trials underwent treatment as recently as 2007, at which time, the research focus had shifted to trying to decrease the burden of immunosuppression while maintaining effectiveness, Dr. Westman observed. For this reason, she and her coworkers are cautiously optimistic about the limited cancer risk seen to date in their analysis.
"This may be caused by too short a period of time of follow-up or else, as we would like to think, maybe we have improved the treatment by lowering the cumulative dose of cyclophosphamide, making it less toxic. But that we don’t really know yet," she said.
Several audience members rose to relate that their own clinical experience has been that lymphoma, bladder cancer, and other malignancies crop up not within 5 years post treatment for ANCA-associated vasculitis, but 10-15 years later or even more.
"I completely agree," Dr. Westman replied. "It’s important for us to arrange longer-term follow-up to say anything conclusive about malignancy. We’re aiming for 10-year follow-up in these patients."
Still, she added, there is reason to be hopeful that some of the more recently evaluated treatment regimens are less oncogenic. For one thing, the earlier Danish study found increased rates of several types of cancer within 5 years.
Also, she cited a recent report on 445 patients with granulomatosis with polyangiitis treated during the past 4 decades at a single large German academic rheumatology center. The German investigators found no increase in malignancies. Moreover, the standardized mortality ratio – essentially, the risk of all-cause mortality compared to that of the matched general population – fell from 2.1 in patients treated during 1966-1993, to 1.41 in those treated in 1994-1998, to 1.03, meaning no increased risk, in patients treated in 1999-2002. Relapse rates during those three time periods dropped from 61% to 51% to 35% (Arthritis Rheum. 2011;63:257-66).
The four-study European Vasculitis Society cohort followed prospectively for 5 years had a median age of 61 years at the time of enrollment in the randomized trials. Of the 535 patients, 53% had antiproteinase-3 antibody positive ANCA (PR3-ANCA) and 38% had antimyeloperoxidase-positive ANCA.
Overall survival at 2 and 5 years was 85% and 78%, respectively. Mortality was 2.6-fold higher than in the age- and sex-matched general population. The mortality risk was greatest during the first year of follow up, with deaths mainly from infection or active vasculitis. After year 1, most deaths were caused by infection, cardiovascular events, or cancer.
Relapse, defined as new or worsened manifestations of ANCA-associated vasculitis requiring a change in therapy, occurred in 38% of patients. The two major risk factors for relapse were being PR3-ANCA positive or having cardiovascular involvement at enrollment.
The best 5-year overall survival was in patients younger than age 50 years at enrollment in their randomized trial. Survival was significantly worse in stepwise fashion in 50- to 60-year-olds, worse yet in 60- to 70-year-olds, and lowest of all in patients above age 70 years.
"Age matters," Dr. Westman declared.
One or more cardiovascular events occurred in 14% of the subjects during 5 years of follow-up. There were 32 cardiovascular deaths, 25 nonfatal strokes, and 42 nonfatal myocardial infarctions or revascularization procedures. Not surprisingly, older age was an independent risk factor for cardiovascular events. But PR3-ANCA–positive status proved to be an independent protector against cardiovascular events; it was associated with a 59% reduction in risk.
Also, in a new analysis of the CYCLOPS study, the most recent of the four European Vasculitis Society clinical trials, the risk of relapse was independently related to a participant’s PR3-ANCA status. The lowest relapse rate occurred in PR3-ANCA–negative patients randomized to daily oral cyclophosphamide for remission induction, while the worst relapse rate was in PR3-ANCA–positive patients assigned to pulse cyclophosphamide (Ann. Rheum. Dis. 2012;71:955-60). These findings may bring closer the day when individualized tailoring of immunosuppression might become possible.
Of patients with granulomatosis with polyangiitis, 49% experienced otolaryngologic involvement during 5 years of follow up, and in 65% of affected patients the otolaryngologic damage appeared to be permanent. An important study observation was that patients with frequent relapses were at increased risk of permanent otolaryngologic damage, Dr. Westman continued.
The four European Vasculitis Society randomized trials that tested various induction and maintenance-of-remission treatment regimens were known as NORAM, CYCAZAREM, MEPEX, and CYCLOPS.
The 5-year follow-up analysis of the four trials was funded by the European League Against Rheumatism. Dr. Westman reported having no financial conflicts.
BERLIN – Nonmelanoma skin cancer was the only type of malignancy found to occur at an increased rate in new 5-year follow-up data on 535 patients treated for systemic small-vessel vasculitis in four major randomized trials sponsored by the European Vasculitis Society.
With 2,650 person-years of prospective observation of 281 clinical trial participants treated for granulomatosis with polyangiitis (formerly called Wegener’s granulomatosis) and 254 treated for microscopic polyangiitis, patients with circulating antineutrophil cytoplasmic antibody (ANCA)-associated vasculitis had a 2.78-fold greater incidence of nonmelanoma skin cancer than did the matched general population, based upon data from the European cancer registry. But no other type of cancer was significantly increased, Dr. Kerstin W.A. Westman reported at the annual European Congress of Rheumatology.
This finding came as a pleasant surprise. Earlier publications by other investigators have reported broadly elevated malignancy risks in the years following administration of the various toxic, largely cyclophosphamide-based therapeutic regimens for ANCA-associated vasculitis, noted Dr. Westman, a nephrologist at Lund (Sweden) University.
For example, Danish investigators reported significantly increased rates of bladder and prostate cancer and acute myeloid leukemia as well as nonmelanoma skin cancer in a study featuring 2,121 person-years of follow-up (J. Rheumatol.2008;35:100-5). However, the Danish patients were treated during 1973-1999, while participants in the four European Vasculitis Society randomized trials underwent treatment as recently as 2007, at which time, the research focus had shifted to trying to decrease the burden of immunosuppression while maintaining effectiveness, Dr. Westman observed. For this reason, she and her coworkers are cautiously optimistic about the limited cancer risk seen to date in their analysis.
"This may be caused by too short a period of time of follow-up or else, as we would like to think, maybe we have improved the treatment by lowering the cumulative dose of cyclophosphamide, making it less toxic. But that we don’t really know yet," she said.
Several audience members rose to relate that their own clinical experience has been that lymphoma, bladder cancer, and other malignancies crop up not within 5 years post treatment for ANCA-associated vasculitis, but 10-15 years later or even more.
"I completely agree," Dr. Westman replied. "It’s important for us to arrange longer-term follow-up to say anything conclusive about malignancy. We’re aiming for 10-year follow-up in these patients."
Still, she added, there is reason to be hopeful that some of the more recently evaluated treatment regimens are less oncogenic. For one thing, the earlier Danish study found increased rates of several types of cancer within 5 years.
Also, she cited a recent report on 445 patients with granulomatosis with polyangiitis treated during the past 4 decades at a single large German academic rheumatology center. The German investigators found no increase in malignancies. Moreover, the standardized mortality ratio – essentially, the risk of all-cause mortality compared to that of the matched general population – fell from 2.1 in patients treated during 1966-1993, to 1.41 in those treated in 1994-1998, to 1.03, meaning no increased risk, in patients treated in 1999-2002. Relapse rates during those three time periods dropped from 61% to 51% to 35% (Arthritis Rheum. 2011;63:257-66).
The four-study European Vasculitis Society cohort followed prospectively for 5 years had a median age of 61 years at the time of enrollment in the randomized trials. Of the 535 patients, 53% had antiproteinase-3 antibody positive ANCA (PR3-ANCA) and 38% had antimyeloperoxidase-positive ANCA.
Overall survival at 2 and 5 years was 85% and 78%, respectively. Mortality was 2.6-fold higher than in the age- and sex-matched general population. The mortality risk was greatest during the first year of follow up, with deaths mainly from infection or active vasculitis. After year 1, most deaths were caused by infection, cardiovascular events, or cancer.
Relapse, defined as new or worsened manifestations of ANCA-associated vasculitis requiring a change in therapy, occurred in 38% of patients. The two major risk factors for relapse were being PR3-ANCA positive or having cardiovascular involvement at enrollment.
The best 5-year overall survival was in patients younger than age 50 years at enrollment in their randomized trial. Survival was significantly worse in stepwise fashion in 50- to 60-year-olds, worse yet in 60- to 70-year-olds, and lowest of all in patients above age 70 years.
"Age matters," Dr. Westman declared.
One or more cardiovascular events occurred in 14% of the subjects during 5 years of follow-up. There were 32 cardiovascular deaths, 25 nonfatal strokes, and 42 nonfatal myocardial infarctions or revascularization procedures. Not surprisingly, older age was an independent risk factor for cardiovascular events. But PR3-ANCA–positive status proved to be an independent protector against cardiovascular events; it was associated with a 59% reduction in risk.
Also, in a new analysis of the CYCLOPS study, the most recent of the four European Vasculitis Society clinical trials, the risk of relapse was independently related to a participant’s PR3-ANCA status. The lowest relapse rate occurred in PR3-ANCA–negative patients randomized to daily oral cyclophosphamide for remission induction, while the worst relapse rate was in PR3-ANCA–positive patients assigned to pulse cyclophosphamide (Ann. Rheum. Dis. 2012;71:955-60). These findings may bring closer the day when individualized tailoring of immunosuppression might become possible.
Of patients with granulomatosis with polyangiitis, 49% experienced otolaryngologic involvement during 5 years of follow up, and in 65% of affected patients the otolaryngologic damage appeared to be permanent. An important study observation was that patients with frequent relapses were at increased risk of permanent otolaryngologic damage, Dr. Westman continued.
The four European Vasculitis Society randomized trials that tested various induction and maintenance-of-remission treatment regimens were known as NORAM, CYCAZAREM, MEPEX, and CYCLOPS.
The 5-year follow-up analysis of the four trials was funded by the European League Against Rheumatism. Dr. Westman reported having no financial conflicts.
BERLIN – Nonmelanoma skin cancer was the only type of malignancy found to occur at an increased rate in new 5-year follow-up data on 535 patients treated for systemic small-vessel vasculitis in four major randomized trials sponsored by the European Vasculitis Society.
With 2,650 person-years of prospective observation of 281 clinical trial participants treated for granulomatosis with polyangiitis (formerly called Wegener’s granulomatosis) and 254 treated for microscopic polyangiitis, patients with circulating antineutrophil cytoplasmic antibody (ANCA)-associated vasculitis had a 2.78-fold greater incidence of nonmelanoma skin cancer than did the matched general population, based upon data from the European cancer registry. But no other type of cancer was significantly increased, Dr. Kerstin W.A. Westman reported at the annual European Congress of Rheumatology.
This finding came as a pleasant surprise. Earlier publications by other investigators have reported broadly elevated malignancy risks in the years following administration of the various toxic, largely cyclophosphamide-based therapeutic regimens for ANCA-associated vasculitis, noted Dr. Westman, a nephrologist at Lund (Sweden) University.
For example, Danish investigators reported significantly increased rates of bladder and prostate cancer and acute myeloid leukemia as well as nonmelanoma skin cancer in a study featuring 2,121 person-years of follow-up (J. Rheumatol.2008;35:100-5). However, the Danish patients were treated during 1973-1999, while participants in the four European Vasculitis Society randomized trials underwent treatment as recently as 2007, at which time, the research focus had shifted to trying to decrease the burden of immunosuppression while maintaining effectiveness, Dr. Westman observed. For this reason, she and her coworkers are cautiously optimistic about the limited cancer risk seen to date in their analysis.
"This may be caused by too short a period of time of follow-up or else, as we would like to think, maybe we have improved the treatment by lowering the cumulative dose of cyclophosphamide, making it less toxic. But that we don’t really know yet," she said.
Several audience members rose to relate that their own clinical experience has been that lymphoma, bladder cancer, and other malignancies crop up not within 5 years post treatment for ANCA-associated vasculitis, but 10-15 years later or even more.
"I completely agree," Dr. Westman replied. "It’s important for us to arrange longer-term follow-up to say anything conclusive about malignancy. We’re aiming for 10-year follow-up in these patients."
Still, she added, there is reason to be hopeful that some of the more recently evaluated treatment regimens are less oncogenic. For one thing, the earlier Danish study found increased rates of several types of cancer within 5 years.
Also, she cited a recent report on 445 patients with granulomatosis with polyangiitis treated during the past 4 decades at a single large German academic rheumatology center. The German investigators found no increase in malignancies. Moreover, the standardized mortality ratio – essentially, the risk of all-cause mortality compared to that of the matched general population – fell from 2.1 in patients treated during 1966-1993, to 1.41 in those treated in 1994-1998, to 1.03, meaning no increased risk, in patients treated in 1999-2002. Relapse rates during those three time periods dropped from 61% to 51% to 35% (Arthritis Rheum. 2011;63:257-66).
The four-study European Vasculitis Society cohort followed prospectively for 5 years had a median age of 61 years at the time of enrollment in the randomized trials. Of the 535 patients, 53% had antiproteinase-3 antibody positive ANCA (PR3-ANCA) and 38% had antimyeloperoxidase-positive ANCA.
Overall survival at 2 and 5 years was 85% and 78%, respectively. Mortality was 2.6-fold higher than in the age- and sex-matched general population. The mortality risk was greatest during the first year of follow up, with deaths mainly from infection or active vasculitis. After year 1, most deaths were caused by infection, cardiovascular events, or cancer.
Relapse, defined as new or worsened manifestations of ANCA-associated vasculitis requiring a change in therapy, occurred in 38% of patients. The two major risk factors for relapse were being PR3-ANCA positive or having cardiovascular involvement at enrollment.
The best 5-year overall survival was in patients younger than age 50 years at enrollment in their randomized trial. Survival was significantly worse in stepwise fashion in 50- to 60-year-olds, worse yet in 60- to 70-year-olds, and lowest of all in patients above age 70 years.
"Age matters," Dr. Westman declared.
One or more cardiovascular events occurred in 14% of the subjects during 5 years of follow-up. There were 32 cardiovascular deaths, 25 nonfatal strokes, and 42 nonfatal myocardial infarctions or revascularization procedures. Not surprisingly, older age was an independent risk factor for cardiovascular events. But PR3-ANCA–positive status proved to be an independent protector against cardiovascular events; it was associated with a 59% reduction in risk.
Also, in a new analysis of the CYCLOPS study, the most recent of the four European Vasculitis Society clinical trials, the risk of relapse was independently related to a participant’s PR3-ANCA status. The lowest relapse rate occurred in PR3-ANCA–negative patients randomized to daily oral cyclophosphamide for remission induction, while the worst relapse rate was in PR3-ANCA–positive patients assigned to pulse cyclophosphamide (Ann. Rheum. Dis. 2012;71:955-60). These findings may bring closer the day when individualized tailoring of immunosuppression might become possible.
Of patients with granulomatosis with polyangiitis, 49% experienced otolaryngologic involvement during 5 years of follow up, and in 65% of affected patients the otolaryngologic damage appeared to be permanent. An important study observation was that patients with frequent relapses were at increased risk of permanent otolaryngologic damage, Dr. Westman continued.
The four European Vasculitis Society randomized trials that tested various induction and maintenance-of-remission treatment regimens were known as NORAM, CYCAZAREM, MEPEX, and CYCLOPS.
The 5-year follow-up analysis of the four trials was funded by the European League Against Rheumatism. Dr. Westman reported having no financial conflicts.
AT THE ANNUAL EUROPEAN CONGRESS OF RHEUMATOLOGY
Major Finding: Patients with circulating antineutrophil cytoplasmic antibody (ANCA)-associated vasculitis had a 2.78-fold greater incidence of nonmelanoma skin cancer than did the matched general population, based upon European cancer registry data. But no other type of cancer was significantly increased in patients treated for ANCA-associated vasculitis. Patients in four European Vasculitis Society–sponsored randomized trials had an increased incidence of only one type of malignancy – nonmelanoma skin cancer – during 5 years of prospective follow-up.
Data Source: This finding is based on an analysis of data from a prospective follow-up of 281 patients with granulomatosis with polyangiitis and 254 with microscopic polyangiitis.
Disclosures: The long-term follow-up was funded by the European League Against Rheumatism. The investigators reported having no financial conflicts.
Baggy Eye Solutions: The Skinny Podcast
In this month's program, Miami dermatologist Dr. Jill Waibel shares her excitement about the effectiveness of fractional lasers to treat burn scars.
Then, Dr. Brett Coldiron, president of the American College of Mohs Surgery, explains why Mohs surgery rates are on the rise.
Health and Human Services Secretary Kathleen Sebelius asks for help and support in implementing the affordable care act.
And finally, Cosmetic Counter host Dr. Lily Talakoub offers tips for treating under-eye circles.
Don't miss another episode of The Skinny Podcast; subscribe on iTunes!
In this month's program, Miami dermatologist Dr. Jill Waibel shares her excitement about the effectiveness of fractional lasers to treat burn scars.
Then, Dr. Brett Coldiron, president of the American College of Mohs Surgery, explains why Mohs surgery rates are on the rise.
Health and Human Services Secretary Kathleen Sebelius asks for help and support in implementing the affordable care act.
And finally, Cosmetic Counter host Dr. Lily Talakoub offers tips for treating under-eye circles.
Don't miss another episode of The Skinny Podcast; subscribe on iTunes!
In this month's program, Miami dermatologist Dr. Jill Waibel shares her excitement about the effectiveness of fractional lasers to treat burn scars.
Then, Dr. Brett Coldiron, president of the American College of Mohs Surgery, explains why Mohs surgery rates are on the rise.
Health and Human Services Secretary Kathleen Sebelius asks for help and support in implementing the affordable care act.
And finally, Cosmetic Counter host Dr. Lily Talakoub offers tips for treating under-eye circles.
Don't miss another episode of The Skinny Podcast; subscribe on iTunes!
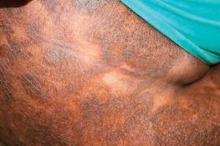
CTCL Proves Challenging in Skin of Color Patients
Over the past 4 decades, cutaneous T-cell lymphoma diagnoses have been on the rise, especially among black and other skin of color patients. Unfortunately, this form of skin cancer can be a diagnostic challenge.
Regarding diagnosis, cutaneous T-cell lymphoma (CTCL) is the wild card of dermatology; it is a great mimicker of many skin disorders, and can look like almost anything. However, the most striking feature of the condition – and the key to diagnosis in ethnic skin – is its polymorphous pigmentation that is unique to skin of color.
CTCL lesions may be flat or raised, and may mimic other skin disorders associated with pigment change. These can include tinea versicolor, vitiligo, pityriasis rosea, and psoriasis. Lichen planus pigmentosus and progressive macular hypomelanosis are other important CTCL mimickers. Consider all of these conditions in the differential diagnosis.
An excellent resource for photos and descriptions of CTCL, as well as CTCL look-alikes, is an article by Dr. Ginette A. Hinds and Dr. Peter Heald (J. Am. Acad. Dermatol. 2009;60:359-75). The article also highlights the clinical variants within the mycosis fungoides subtype, the most common form of CTCL among blacks.
For example, look out for the "herald patch" of unilesional mycosis fungoides, which takes the form of a single large, dyspigmented patch. According to the authors, this variant has an excellent prognosis, possibly because of an active immune response that limits the initial spread of disease and contributes to preventing relapse.
Another variant is pigmented purpuric mycosis fungoides, which can often be confused with the benign pigmented purpura syndromes of Schamberg’s disease (progressive pigmented purpuric dermatitis), Gougerot-Blum syndrome (pigmented purpuric lichenoid dermatitis), Majocchi’s disease (purpura annularis telangiectodes), Doucas and Kapetanakis (lymphocytic capillaritis of unknown cause), and lichen aureus. The key to diagnosis in this case is recalling that "the lesions of the benign syndromes rarely assume the morphology and distribution found with mycosis fungoides," Dr. Hinds and Dr. Heald wrote.
Not only is mycosis fungoides difficult to diagnose, but it is especially prevalent among black patients. For instance, a 1988 study looking at Surveillance, Epidemiology, and End Results (SEER) data from 1973 through 1984 found that the incidence among black patients was twice that of white patients: 0.52/100,000 vs. 0.26 (JAMA 1988;260:42-46).
Similarly, in another study of 132 black patients with skin cancer, mycosis fungoides represented 12.1% of all skin neoplasms (Dermatol. Clin. 1988;6:397-405).
Mycosis fungoides is the fourth most common skin cancer among Japanese patients, representing approximately 5% of all skin malignancies in the population.
The incidence of CTCL and its subtypes is only increasing. A 2007 study that extended the SEER data mentioned above from 1973 to 2002 and included all CTCL cases found an incidence rate of 9.0 for black patients, per 1 million person-years, compared with 6.1 for white patients (Arch. Dermatol. 2007;143:854-9).
As in many skin cancers, early diagnosis can make the difference in successful outcomes. So add CTCL, and especially the mycosis fungoides subtype, to your differential when confronting pigmentation disorders in skin of color patients.
Dr. Roberts is past president of the Women’s Dermatologic Society (WDS) as well as past president of the California Society of Dermatology and Dermatologic Surgery (Calderm). She was a founding director of dermatopathology at the Loma Linda University Medical Center (Calif.). She currently runs a private practice in Rancho Mirage, Calif.
She disclosed financial relationships with Allergan, L’Oréal/La Roche Posay, Skin Medica, and Valeant Pharmaceuticals.
Over the past 4 decades, cutaneous T-cell lymphoma diagnoses have been on the rise, especially among black and other skin of color patients. Unfortunately, this form of skin cancer can be a diagnostic challenge.
Regarding diagnosis, cutaneous T-cell lymphoma (CTCL) is the wild card of dermatology; it is a great mimicker of many skin disorders, and can look like almost anything. However, the most striking feature of the condition – and the key to diagnosis in ethnic skin – is its polymorphous pigmentation that is unique to skin of color.
CTCL lesions may be flat or raised, and may mimic other skin disorders associated with pigment change. These can include tinea versicolor, vitiligo, pityriasis rosea, and psoriasis. Lichen planus pigmentosus and progressive macular hypomelanosis are other important CTCL mimickers. Consider all of these conditions in the differential diagnosis.
An excellent resource for photos and descriptions of CTCL, as well as CTCL look-alikes, is an article by Dr. Ginette A. Hinds and Dr. Peter Heald (J. Am. Acad. Dermatol. 2009;60:359-75). The article also highlights the clinical variants within the mycosis fungoides subtype, the most common form of CTCL among blacks.
For example, look out for the "herald patch" of unilesional mycosis fungoides, which takes the form of a single large, dyspigmented patch. According to the authors, this variant has an excellent prognosis, possibly because of an active immune response that limits the initial spread of disease and contributes to preventing relapse.
Another variant is pigmented purpuric mycosis fungoides, which can often be confused with the benign pigmented purpura syndromes of Schamberg’s disease (progressive pigmented purpuric dermatitis), Gougerot-Blum syndrome (pigmented purpuric lichenoid dermatitis), Majocchi’s disease (purpura annularis telangiectodes), Doucas and Kapetanakis (lymphocytic capillaritis of unknown cause), and lichen aureus. The key to diagnosis in this case is recalling that "the lesions of the benign syndromes rarely assume the morphology and distribution found with mycosis fungoides," Dr. Hinds and Dr. Heald wrote.
Not only is mycosis fungoides difficult to diagnose, but it is especially prevalent among black patients. For instance, a 1988 study looking at Surveillance, Epidemiology, and End Results (SEER) data from 1973 through 1984 found that the incidence among black patients was twice that of white patients: 0.52/100,000 vs. 0.26 (JAMA 1988;260:42-46).
Similarly, in another study of 132 black patients with skin cancer, mycosis fungoides represented 12.1% of all skin neoplasms (Dermatol. Clin. 1988;6:397-405).
Mycosis fungoides is the fourth most common skin cancer among Japanese patients, representing approximately 5% of all skin malignancies in the population.
The incidence of CTCL and its subtypes is only increasing. A 2007 study that extended the SEER data mentioned above from 1973 to 2002 and included all CTCL cases found an incidence rate of 9.0 for black patients, per 1 million person-years, compared with 6.1 for white patients (Arch. Dermatol. 2007;143:854-9).
As in many skin cancers, early diagnosis can make the difference in successful outcomes. So add CTCL, and especially the mycosis fungoides subtype, to your differential when confronting pigmentation disorders in skin of color patients.
Dr. Roberts is past president of the Women’s Dermatologic Society (WDS) as well as past president of the California Society of Dermatology and Dermatologic Surgery (Calderm). She was a founding director of dermatopathology at the Loma Linda University Medical Center (Calif.). She currently runs a private practice in Rancho Mirage, Calif.
She disclosed financial relationships with Allergan, L’Oréal/La Roche Posay, Skin Medica, and Valeant Pharmaceuticals.
Over the past 4 decades, cutaneous T-cell lymphoma diagnoses have been on the rise, especially among black and other skin of color patients. Unfortunately, this form of skin cancer can be a diagnostic challenge.
Regarding diagnosis, cutaneous T-cell lymphoma (CTCL) is the wild card of dermatology; it is a great mimicker of many skin disorders, and can look like almost anything. However, the most striking feature of the condition – and the key to diagnosis in ethnic skin – is its polymorphous pigmentation that is unique to skin of color.
CTCL lesions may be flat or raised, and may mimic other skin disorders associated with pigment change. These can include tinea versicolor, vitiligo, pityriasis rosea, and psoriasis. Lichen planus pigmentosus and progressive macular hypomelanosis are other important CTCL mimickers. Consider all of these conditions in the differential diagnosis.
An excellent resource for photos and descriptions of CTCL, as well as CTCL look-alikes, is an article by Dr. Ginette A. Hinds and Dr. Peter Heald (J. Am. Acad. Dermatol. 2009;60:359-75). The article also highlights the clinical variants within the mycosis fungoides subtype, the most common form of CTCL among blacks.
For example, look out for the "herald patch" of unilesional mycosis fungoides, which takes the form of a single large, dyspigmented patch. According to the authors, this variant has an excellent prognosis, possibly because of an active immune response that limits the initial spread of disease and contributes to preventing relapse.
Another variant is pigmented purpuric mycosis fungoides, which can often be confused with the benign pigmented purpura syndromes of Schamberg’s disease (progressive pigmented purpuric dermatitis), Gougerot-Blum syndrome (pigmented purpuric lichenoid dermatitis), Majocchi’s disease (purpura annularis telangiectodes), Doucas and Kapetanakis (lymphocytic capillaritis of unknown cause), and lichen aureus. The key to diagnosis in this case is recalling that "the lesions of the benign syndromes rarely assume the morphology and distribution found with mycosis fungoides," Dr. Hinds and Dr. Heald wrote.
Not only is mycosis fungoides difficult to diagnose, but it is especially prevalent among black patients. For instance, a 1988 study looking at Surveillance, Epidemiology, and End Results (SEER) data from 1973 through 1984 found that the incidence among black patients was twice that of white patients: 0.52/100,000 vs. 0.26 (JAMA 1988;260:42-46).
Similarly, in another study of 132 black patients with skin cancer, mycosis fungoides represented 12.1% of all skin neoplasms (Dermatol. Clin. 1988;6:397-405).
Mycosis fungoides is the fourth most common skin cancer among Japanese patients, representing approximately 5% of all skin malignancies in the population.
The incidence of CTCL and its subtypes is only increasing. A 2007 study that extended the SEER data mentioned above from 1973 to 2002 and included all CTCL cases found an incidence rate of 9.0 for black patients, per 1 million person-years, compared with 6.1 for white patients (Arch. Dermatol. 2007;143:854-9).
As in many skin cancers, early diagnosis can make the difference in successful outcomes. So add CTCL, and especially the mycosis fungoides subtype, to your differential when confronting pigmentation disorders in skin of color patients.
Dr. Roberts is past president of the Women’s Dermatologic Society (WDS) as well as past president of the California Society of Dermatology and Dermatologic Surgery (Calderm). She was a founding director of dermatopathology at the Loma Linda University Medical Center (Calif.). She currently runs a private practice in Rancho Mirage, Calif.
She disclosed financial relationships with Allergan, L’Oréal/La Roche Posay, Skin Medica, and Valeant Pharmaceuticals.
Aspirin Found to Protect Against Melanoma
RALEIGH, N.C. – Postmenopausal women who used aspirin regularly had a significantly reduced risk of developing melanoma during long-term prospective follow-up, according to a Women’s Health Initiative observational study analysis.
The magnitude of this risk reduction grew with greater duration of aspirin use, reported Christina A. Gamba, a medical student at Stanford (Calif.) University.
During 12 years of prospective follow-up, women who at the time of study enrollment reported they had been taking aspirin regularly for less than a year had roughly an 11% reduction in the likelihood of developing incident melanoma, compared with aspirin nonusers. Those who had been using aspirin regularly for 1-4 years at enrollment went on to have a 20% risk reduction. And women on aspirin for 5 years or longer at enrollment were 30% less likely to develop melanoma than nonusers.
These relative risk figures were fully adjusted for age, education, income, skin type, melanoma risk factors, physical activity, vitamin D intake, smoking status, body mass index, sunburn history, time spent outdoors, sunscreen use, and medical indications for aspirin use, such as cardiovascular disease, she noted.
The findings suggest a possible chemopreventive effect for aspirin against the development of melanoma. As such, these data are consistent with other studies that have linked NSAIDs with decreased risks of breast, colorectal, and gastric cancer. The specific mechanism of benefit is thought to involve inhibition of cyclo-oxygenase-2, which is proinflammatory.
Ms. Gamba explained that her Women’s Health Initiative observational study analysis included 59,806 postmenopausal white women for whom complete pertinent data were available. During nearly 12 years of follow-up, 548 incident melanomas occurred among the study population: 255 were invasive; the rest were in situ.
At enrollment, 25% of women reported using aspirin regularly; their melanoma incidence during follow-up was 0.76%. Fifteen percent of subjects used nonaspirin NSAIDs; their melanoma incidence was 0.97%, which was identical to the rate in the 60% of women who were nonusers of any NSAIDs. Regular users of acetaminophen, a drug that relieves pain but doesn’t inhibit cyclo-oxygenase-2, had the same melanoma incidence as nonusers of aspirin and other NSAIDs.
Three-quarters of the aspirin users were on regular- or extrastrength formulations. There were too few women on low-dose aspirin to be able to draw any conclusions as to whether it, too, protected against melanoma, she said at the annual meeting of the Society of Investigative Dermatology.
The study included one measure of consistency: At 3 years of follow-up, 60% of women who reported taking aspirin regularly at baseline were still doing so, as were 35% of those who had been on nonaspirin NSAIDs at enrollment.
Audience members posed this question: If aspirin’s presumed chemoprevention mechanism involves the inhibition of cyclooxygenase-2, why didn’t users of nonaspirin NSAIDs enjoy a similar benefit? Ms. Gamba offered two possible theories. One is that users of nonaspirin NSAIDs took the medication less frequently, in contrast to aspirin users who generally took the drug on a daily basis.
The other possibility, she continued, is that aspirin curbs tumorigenesis through cyclo-oxygenase-independent mechanisms – perhaps by promoting apoptotic genes and/or inhibiting tumor-promoting genes.
All cases of melanoma in the Women’s Health Initiative were pathologically confirmed. Ms. Gamba and her investigators are now going back and examining the pathology reports to learn what specific types of melanoma were involved. They are also reanalyzing their aspirin/NSAID usage data, this time restricting the analysis to cases of in situ melanoma.
The Women’s Health Initiative Observational Study was sponsored by the National Institutes of Health. Ms. Gamba reported having no relevant financial conflicts.
RALEIGH, N.C. – Postmenopausal women who used aspirin regularly had a significantly reduced risk of developing melanoma during long-term prospective follow-up, according to a Women’s Health Initiative observational study analysis.
The magnitude of this risk reduction grew with greater duration of aspirin use, reported Christina A. Gamba, a medical student at Stanford (Calif.) University.
During 12 years of prospective follow-up, women who at the time of study enrollment reported they had been taking aspirin regularly for less than a year had roughly an 11% reduction in the likelihood of developing incident melanoma, compared with aspirin nonusers. Those who had been using aspirin regularly for 1-4 years at enrollment went on to have a 20% risk reduction. And women on aspirin for 5 years or longer at enrollment were 30% less likely to develop melanoma than nonusers.
These relative risk figures were fully adjusted for age, education, income, skin type, melanoma risk factors, physical activity, vitamin D intake, smoking status, body mass index, sunburn history, time spent outdoors, sunscreen use, and medical indications for aspirin use, such as cardiovascular disease, she noted.
The findings suggest a possible chemopreventive effect for aspirin against the development of melanoma. As such, these data are consistent with other studies that have linked NSAIDs with decreased risks of breast, colorectal, and gastric cancer. The specific mechanism of benefit is thought to involve inhibition of cyclo-oxygenase-2, which is proinflammatory.
Ms. Gamba explained that her Women’s Health Initiative observational study analysis included 59,806 postmenopausal white women for whom complete pertinent data were available. During nearly 12 years of follow-up, 548 incident melanomas occurred among the study population: 255 were invasive; the rest were in situ.
At enrollment, 25% of women reported using aspirin regularly; their melanoma incidence during follow-up was 0.76%. Fifteen percent of subjects used nonaspirin NSAIDs; their melanoma incidence was 0.97%, which was identical to the rate in the 60% of women who were nonusers of any NSAIDs. Regular users of acetaminophen, a drug that relieves pain but doesn’t inhibit cyclo-oxygenase-2, had the same melanoma incidence as nonusers of aspirin and other NSAIDs.
Three-quarters of the aspirin users were on regular- or extrastrength formulations. There were too few women on low-dose aspirin to be able to draw any conclusions as to whether it, too, protected against melanoma, she said at the annual meeting of the Society of Investigative Dermatology.
The study included one measure of consistency: At 3 years of follow-up, 60% of women who reported taking aspirin regularly at baseline were still doing so, as were 35% of those who had been on nonaspirin NSAIDs at enrollment.
Audience members posed this question: If aspirin’s presumed chemoprevention mechanism involves the inhibition of cyclooxygenase-2, why didn’t users of nonaspirin NSAIDs enjoy a similar benefit? Ms. Gamba offered two possible theories. One is that users of nonaspirin NSAIDs took the medication less frequently, in contrast to aspirin users who generally took the drug on a daily basis.
The other possibility, she continued, is that aspirin curbs tumorigenesis through cyclo-oxygenase-independent mechanisms – perhaps by promoting apoptotic genes and/or inhibiting tumor-promoting genes.
All cases of melanoma in the Women’s Health Initiative were pathologically confirmed. Ms. Gamba and her investigators are now going back and examining the pathology reports to learn what specific types of melanoma were involved. They are also reanalyzing their aspirin/NSAID usage data, this time restricting the analysis to cases of in situ melanoma.
The Women’s Health Initiative Observational Study was sponsored by the National Institutes of Health. Ms. Gamba reported having no relevant financial conflicts.
RALEIGH, N.C. – Postmenopausal women who used aspirin regularly had a significantly reduced risk of developing melanoma during long-term prospective follow-up, according to a Women’s Health Initiative observational study analysis.
The magnitude of this risk reduction grew with greater duration of aspirin use, reported Christina A. Gamba, a medical student at Stanford (Calif.) University.
During 12 years of prospective follow-up, women who at the time of study enrollment reported they had been taking aspirin regularly for less than a year had roughly an 11% reduction in the likelihood of developing incident melanoma, compared with aspirin nonusers. Those who had been using aspirin regularly for 1-4 years at enrollment went on to have a 20% risk reduction. And women on aspirin for 5 years or longer at enrollment were 30% less likely to develop melanoma than nonusers.
These relative risk figures were fully adjusted for age, education, income, skin type, melanoma risk factors, physical activity, vitamin D intake, smoking status, body mass index, sunburn history, time spent outdoors, sunscreen use, and medical indications for aspirin use, such as cardiovascular disease, she noted.
The findings suggest a possible chemopreventive effect for aspirin against the development of melanoma. As such, these data are consistent with other studies that have linked NSAIDs with decreased risks of breast, colorectal, and gastric cancer. The specific mechanism of benefit is thought to involve inhibition of cyclo-oxygenase-2, which is proinflammatory.
Ms. Gamba explained that her Women’s Health Initiative observational study analysis included 59,806 postmenopausal white women for whom complete pertinent data were available. During nearly 12 years of follow-up, 548 incident melanomas occurred among the study population: 255 were invasive; the rest were in situ.
At enrollment, 25% of women reported using aspirin regularly; their melanoma incidence during follow-up was 0.76%. Fifteen percent of subjects used nonaspirin NSAIDs; their melanoma incidence was 0.97%, which was identical to the rate in the 60% of women who were nonusers of any NSAIDs. Regular users of acetaminophen, a drug that relieves pain but doesn’t inhibit cyclo-oxygenase-2, had the same melanoma incidence as nonusers of aspirin and other NSAIDs.
Three-quarters of the aspirin users were on regular- or extrastrength formulations. There were too few women on low-dose aspirin to be able to draw any conclusions as to whether it, too, protected against melanoma, she said at the annual meeting of the Society of Investigative Dermatology.
The study included one measure of consistency: At 3 years of follow-up, 60% of women who reported taking aspirin regularly at baseline were still doing so, as were 35% of those who had been on nonaspirin NSAIDs at enrollment.
Audience members posed this question: If aspirin’s presumed chemoprevention mechanism involves the inhibition of cyclooxygenase-2, why didn’t users of nonaspirin NSAIDs enjoy a similar benefit? Ms. Gamba offered two possible theories. One is that users of nonaspirin NSAIDs took the medication less frequently, in contrast to aspirin users who generally took the drug on a daily basis.
The other possibility, she continued, is that aspirin curbs tumorigenesis through cyclo-oxygenase-independent mechanisms – perhaps by promoting apoptotic genes and/or inhibiting tumor-promoting genes.
All cases of melanoma in the Women’s Health Initiative were pathologically confirmed. Ms. Gamba and her investigators are now going back and examining the pathology reports to learn what specific types of melanoma were involved. They are also reanalyzing their aspirin/NSAID usage data, this time restricting the analysis to cases of in situ melanoma.
The Women’s Health Initiative Observational Study was sponsored by the National Institutes of Health. Ms. Gamba reported having no relevant financial conflicts.
FROM THE ANNUAL MEETING OF THE SOCIETY FOR INVESTIGATIVE DERMATOLOGY
Major Finding: Postmenopausal women who reported using aspirin regularly at baseline went on to have an 11%-30% reduction in the risk for developing melanoma, compared with aspirin nonusers.
Data Source: Data were from an analysis of nearly 60,000 postmenopausal white participants in the Women’s Health Initiative observational study.
Disclosures: The Women’s Health Initiative study was funded by the National Institutes of Health. Ms. Gamba reported having no relevant financial conflicts.
Vismodegib Continues to Prove BCC Effectiveness
Two small, early-phase studies found that vismodegib is effective for the treatment of basal cell nevus syndrome and advanced basal cell carcinoma, according to results published June 6 in the New England Journal of Medicine.
Basal cell malignancies are among the most common nonmelanoma skin cancers in the United States, accounting for at least 80% of the 2.1 million cases. It is a highly disfiguring cancer but is only sometimes life threatening. Almost all basal cell carcinomas (BCCs) are caused by alterations in the hedgehog signaling pathway.
Vismodegib (Erivedge) was approved by the Food and Drug Administration in January for treating locally advanced and metastatic BCC. Vismodegib (manufactured by Genentech) is a small molecule inhibitor that steps in to repair the hedgehog pathway.
Study 1: Locally Advanced and Metastatic BCC
In the first study, conducted before vismodegib received FDA approval for locally advanced and metastatic BCC, Dr. Aleksandar Sekulic and colleagues conducted a phase II, nonrandomized, two-cohort trial at multiple sites around the United States and overseas. Vismodegib was given to 33 patients with metastatic BCC and 63 patients with locally advanced disease. Both cohorts had inoperable disease or were not eligible for surgery.
All the patients were treated with 150 mg of oral vismodegib daily. The median age of patients was 62 years and all were white, noted Dr. Sekulic of the Mayo Clinic in Scottsdale, Ariz.
The patients were treated with vismodegib until disease progression, until side effects became intolerable, or until end of study. Patients could stop the drug for up to a month if side effects were intolerable.
There was no standard end point for measuring response in locally advanced BCC when the study was designed. The definition used was a decrease of 30% or more of the externally visible or radiographic dimension or complete resolution of ulceration. Progressive disease was an increase of 20% or more in the externally visible or radiographic dimension, new ulceration, or a new lesion.
The response rate for the metastatic group treated with vismodegib was 30%. All of the responses were partial, defined as an absence of residual BCC in a biopsy specimen. In locally advanced disease, the response rate was 43%. Thirteen of the 63 patients had a complete response. The median duration of response was 7.6 months for metastatic and locally advanced BCC patients (N. Engl. J. Med. 2012;366:2171-9).
Half of the patients stopped the drug early, with 18% of metastatic patients discontinuing because of disease progression. Twenty-five percent of locally advanced patients decided on their own to stop therapy, for unknown reasons, according to the investigators. Adverse events – primarily muscle spasms, weight loss, fatigue, and loss of appetite – took a toll on patients. A quarter reported serious adverse events.
Seven patients died, but the relation to vismodegib is not clear at this point. "The deaths were considered by the site investigator to be unrelated to vismodegib," the investigators wrote.
The locally advanced and metastatic BCC trial was funded by Genentech and was jointly designed by Genetech and Dr. Sekulic. A majority of his colleagues reported being employees and/or receiving grants or financial support from the company.
In an interview, Dr. Ali Hendi, assistant clinical professor of dermatology at Georgetown University Medical Center, Washington, said that he felt the investigators in the study were overly subjective in defining locally advanced tumors. "These are tumors that are aggressive, but that does not make them inoperable in the right hands," said Dr. Hendi, who is also a Mohs surgeon.
He also noted the slim complete response rate, which, when coupled with the toxicity and the high cost of the therapy – which might run as much as $70,000 per treatment course – make it a less desirable therapy for many patients.
"For locally advanced [disease] I think the utility is limited if there is good surgical care available," he said. Vismodegib may be more useful for BCC nevus syndrome and for metastatic disease, where it may be the only therapy available, he added.
Dr. Hendi reported no conflicts.
Study 2: Basal Cell Nevus Syndrome
In a second randomized, double-blind, placebo-controlled trial of 41 patients with basal cell nevus syndrome, Dr. Jean Y. Tang and colleagues found that vismodegib reduced BCC tumors and blocked the development of new BCCs.
Basal cell nevus syndrome is rare, but it causes hundreds or thousands of carcinomas in each patient. There is no therapy, and often patients have to undergo multiple surgeries to remove the tumors.
Patients with the condition have one defective copy of the tumor suppressor gene (PTCH1), which inhibits the hedgehog signaling pathway, noted Dr. Tang of Stanford University (Calif.), and her colleagues.
The study enrolled patients from Sept. 2009 through Jan. 2011, but in Dec. 2010 the data safety and monitoring board decided to end the placebo treatment because the results so highly favored vismodegib. Patients were randomized to placebo or 150 mg daily of the drug for a planned 18 months.
The primary end point was the comparative rate of appearance of new BCCs that were eligible for surgery. Patients were followed for a mean of 8 months. Those who took vismodegib had a much lower rate of surgically eligible disease: 2 vs. 29 cases for placebo. Existing carcinomas shrunk by 65% in vismodegib-treated patients, compared with only 11% for placebo patients. Vismodegib patients also had fewer surgeries than patients receiving placebo: 0.31 per patient compared with 4.4 per patient, respectively.
At 1 month, there was a 90% reduction in hedgehog target-gene expression. There was no residual carcinoma in 83% of the biopsy samples taken from sites where the disease had been judged to be clinically regressed (N. Engl. J. Med. 2012;366:2180-8).
Fifty-four percent of study patients discontinued treatment because of adverse events. Patients treated with vismodegib were more likely to have grade 1 or 2 dysgeusia, muscle cramps, hair loss, and weight loss. They also had more grade 3 or 4 adverse events. The authors said that these side effects were similar to those reported in phase 1 and 2 trials of vismodegib, and also in other studies of therapies that inhibit the hedgehog pathway.
The authors concluded that vismodegib reduces tumor burden and blocks new tumor growth, but whether some of the clinically regressed tumors still harbor residual tumor cells cannot be ruled out – which could explain why there is tumor regrowth after therapy is stopped.
However, the "findings confirm the essential role of the hedgehog pathway in basal cell carcinomas," they wrote.
The trial was funded by Genentech and by grants from the National Institutes of Health, the National Institute of Arthritis and Musculoskeletal and Skin Diseases, the National Cancer Institute, the Swim Across America Foundation, and the Michael J. Rainen Family Foundation. Dr. Tang reported receiving consulting fees from Genentech. Several coauthors of the study reported receiving grants or fees from Genentech.
The two studies show that vismodegib is highly effective. "It is a landmark day for patients with basal cell carcinoma and all those involved in their care – the greatest advance in therapy yet seen for this disease," Dr. John T. Lear wrote in an accompanying editorial.
The response rates were impressive, especially in patients with basal cell nevus syndrome.
"However, the side effects are considerable and frequent, resulting in high rates of drug discontinuation, and these rates will probably be even higher in clinical practice," he said.
Another question raised is whether hedgehog pathway inhibition truly clears the BCC or whether it leaves residual resistant cells that could give rise to a recurrence. Follow-up studies are needed to address this question, Dr. Lear said (N. Engl. J. Med. 2012;366:2225-6).
Intermittent dosing might allow more patients to benefit by preventing new lesions. This would be especially important for BCC nevus syndrome. And for patients with large, symptomatic lesions, "the usefulness of local control in improving quality of life should not be underestimated," he noted.
Dr. Lear is affiliated with the Manchester Academic Health Science Centre at Manchester (England) University and the departments of dermatology at Salford Royal Hospital and Central Manchester National Health Service Foundation Trusts. He has received consulting fees from Novartis for an ongoing BCC study.
The two studies show that vismodegib is highly effective. "It is a landmark day for patients with basal cell carcinoma and all those involved in their care – the greatest advance in therapy yet seen for this disease," Dr. John T. Lear wrote in an accompanying editorial.
The response rates were impressive, especially in patients with basal cell nevus syndrome.
"However, the side effects are considerable and frequent, resulting in high rates of drug discontinuation, and these rates will probably be even higher in clinical practice," he said.
Another question raised is whether hedgehog pathway inhibition truly clears the BCC or whether it leaves residual resistant cells that could give rise to a recurrence. Follow-up studies are needed to address this question, Dr. Lear said (N. Engl. J. Med. 2012;366:2225-6).
Intermittent dosing might allow more patients to benefit by preventing new lesions. This would be especially important for BCC nevus syndrome. And for patients with large, symptomatic lesions, "the usefulness of local control in improving quality of life should not be underestimated," he noted.
Dr. Lear is affiliated with the Manchester Academic Health Science Centre at Manchester (England) University and the departments of dermatology at Salford Royal Hospital and Central Manchester National Health Service Foundation Trusts. He has received consulting fees from Novartis for an ongoing BCC study.
The two studies show that vismodegib is highly effective. "It is a landmark day for patients with basal cell carcinoma and all those involved in their care – the greatest advance in therapy yet seen for this disease," Dr. John T. Lear wrote in an accompanying editorial.
The response rates were impressive, especially in patients with basal cell nevus syndrome.
"However, the side effects are considerable and frequent, resulting in high rates of drug discontinuation, and these rates will probably be even higher in clinical practice," he said.
Another question raised is whether hedgehog pathway inhibition truly clears the BCC or whether it leaves residual resistant cells that could give rise to a recurrence. Follow-up studies are needed to address this question, Dr. Lear said (N. Engl. J. Med. 2012;366:2225-6).
Intermittent dosing might allow more patients to benefit by preventing new lesions. This would be especially important for BCC nevus syndrome. And for patients with large, symptomatic lesions, "the usefulness of local control in improving quality of life should not be underestimated," he noted.
Dr. Lear is affiliated with the Manchester Academic Health Science Centre at Manchester (England) University and the departments of dermatology at Salford Royal Hospital and Central Manchester National Health Service Foundation Trusts. He has received consulting fees from Novartis for an ongoing BCC study.
Two small, early-phase studies found that vismodegib is effective for the treatment of basal cell nevus syndrome and advanced basal cell carcinoma, according to results published June 6 in the New England Journal of Medicine.
Basal cell malignancies are among the most common nonmelanoma skin cancers in the United States, accounting for at least 80% of the 2.1 million cases. It is a highly disfiguring cancer but is only sometimes life threatening. Almost all basal cell carcinomas (BCCs) are caused by alterations in the hedgehog signaling pathway.
Vismodegib (Erivedge) was approved by the Food and Drug Administration in January for treating locally advanced and metastatic BCC. Vismodegib (manufactured by Genentech) is a small molecule inhibitor that steps in to repair the hedgehog pathway.
Study 1: Locally Advanced and Metastatic BCC
In the first study, conducted before vismodegib received FDA approval for locally advanced and metastatic BCC, Dr. Aleksandar Sekulic and colleagues conducted a phase II, nonrandomized, two-cohort trial at multiple sites around the United States and overseas. Vismodegib was given to 33 patients with metastatic BCC and 63 patients with locally advanced disease. Both cohorts had inoperable disease or were not eligible for surgery.
All the patients were treated with 150 mg of oral vismodegib daily. The median age of patients was 62 years and all were white, noted Dr. Sekulic of the Mayo Clinic in Scottsdale, Ariz.
The patients were treated with vismodegib until disease progression, until side effects became intolerable, or until end of study. Patients could stop the drug for up to a month if side effects were intolerable.
There was no standard end point for measuring response in locally advanced BCC when the study was designed. The definition used was a decrease of 30% or more of the externally visible or radiographic dimension or complete resolution of ulceration. Progressive disease was an increase of 20% or more in the externally visible or radiographic dimension, new ulceration, or a new lesion.
The response rate for the metastatic group treated with vismodegib was 30%. All of the responses were partial, defined as an absence of residual BCC in a biopsy specimen. In locally advanced disease, the response rate was 43%. Thirteen of the 63 patients had a complete response. The median duration of response was 7.6 months for metastatic and locally advanced BCC patients (N. Engl. J. Med. 2012;366:2171-9).
Half of the patients stopped the drug early, with 18% of metastatic patients discontinuing because of disease progression. Twenty-five percent of locally advanced patients decided on their own to stop therapy, for unknown reasons, according to the investigators. Adverse events – primarily muscle spasms, weight loss, fatigue, and loss of appetite – took a toll on patients. A quarter reported serious adverse events.
Seven patients died, but the relation to vismodegib is not clear at this point. "The deaths were considered by the site investigator to be unrelated to vismodegib," the investigators wrote.
The locally advanced and metastatic BCC trial was funded by Genentech and was jointly designed by Genetech and Dr. Sekulic. A majority of his colleagues reported being employees and/or receiving grants or financial support from the company.
In an interview, Dr. Ali Hendi, assistant clinical professor of dermatology at Georgetown University Medical Center, Washington, said that he felt the investigators in the study were overly subjective in defining locally advanced tumors. "These are tumors that are aggressive, but that does not make them inoperable in the right hands," said Dr. Hendi, who is also a Mohs surgeon.
He also noted the slim complete response rate, which, when coupled with the toxicity and the high cost of the therapy – which might run as much as $70,000 per treatment course – make it a less desirable therapy for many patients.
"For locally advanced [disease] I think the utility is limited if there is good surgical care available," he said. Vismodegib may be more useful for BCC nevus syndrome and for metastatic disease, where it may be the only therapy available, he added.
Dr. Hendi reported no conflicts.
Study 2: Basal Cell Nevus Syndrome
In a second randomized, double-blind, placebo-controlled trial of 41 patients with basal cell nevus syndrome, Dr. Jean Y. Tang and colleagues found that vismodegib reduced BCC tumors and blocked the development of new BCCs.
Basal cell nevus syndrome is rare, but it causes hundreds or thousands of carcinomas in each patient. There is no therapy, and often patients have to undergo multiple surgeries to remove the tumors.
Patients with the condition have one defective copy of the tumor suppressor gene (PTCH1), which inhibits the hedgehog signaling pathway, noted Dr. Tang of Stanford University (Calif.), and her colleagues.
The study enrolled patients from Sept. 2009 through Jan. 2011, but in Dec. 2010 the data safety and monitoring board decided to end the placebo treatment because the results so highly favored vismodegib. Patients were randomized to placebo or 150 mg daily of the drug for a planned 18 months.
The primary end point was the comparative rate of appearance of new BCCs that were eligible for surgery. Patients were followed for a mean of 8 months. Those who took vismodegib had a much lower rate of surgically eligible disease: 2 vs. 29 cases for placebo. Existing carcinomas shrunk by 65% in vismodegib-treated patients, compared with only 11% for placebo patients. Vismodegib patients also had fewer surgeries than patients receiving placebo: 0.31 per patient compared with 4.4 per patient, respectively.
At 1 month, there was a 90% reduction in hedgehog target-gene expression. There was no residual carcinoma in 83% of the biopsy samples taken from sites where the disease had been judged to be clinically regressed (N. Engl. J. Med. 2012;366:2180-8).
Fifty-four percent of study patients discontinued treatment because of adverse events. Patients treated with vismodegib were more likely to have grade 1 or 2 dysgeusia, muscle cramps, hair loss, and weight loss. They also had more grade 3 or 4 adverse events. The authors said that these side effects were similar to those reported in phase 1 and 2 trials of vismodegib, and also in other studies of therapies that inhibit the hedgehog pathway.
The authors concluded that vismodegib reduces tumor burden and blocks new tumor growth, but whether some of the clinically regressed tumors still harbor residual tumor cells cannot be ruled out – which could explain why there is tumor regrowth after therapy is stopped.
However, the "findings confirm the essential role of the hedgehog pathway in basal cell carcinomas," they wrote.
The trial was funded by Genentech and by grants from the National Institutes of Health, the National Institute of Arthritis and Musculoskeletal and Skin Diseases, the National Cancer Institute, the Swim Across America Foundation, and the Michael J. Rainen Family Foundation. Dr. Tang reported receiving consulting fees from Genentech. Several coauthors of the study reported receiving grants or fees from Genentech.
Two small, early-phase studies found that vismodegib is effective for the treatment of basal cell nevus syndrome and advanced basal cell carcinoma, according to results published June 6 in the New England Journal of Medicine.
Basal cell malignancies are among the most common nonmelanoma skin cancers in the United States, accounting for at least 80% of the 2.1 million cases. It is a highly disfiguring cancer but is only sometimes life threatening. Almost all basal cell carcinomas (BCCs) are caused by alterations in the hedgehog signaling pathway.
Vismodegib (Erivedge) was approved by the Food and Drug Administration in January for treating locally advanced and metastatic BCC. Vismodegib (manufactured by Genentech) is a small molecule inhibitor that steps in to repair the hedgehog pathway.
Study 1: Locally Advanced and Metastatic BCC
In the first study, conducted before vismodegib received FDA approval for locally advanced and metastatic BCC, Dr. Aleksandar Sekulic and colleagues conducted a phase II, nonrandomized, two-cohort trial at multiple sites around the United States and overseas. Vismodegib was given to 33 patients with metastatic BCC and 63 patients with locally advanced disease. Both cohorts had inoperable disease or were not eligible for surgery.
All the patients were treated with 150 mg of oral vismodegib daily. The median age of patients was 62 years and all were white, noted Dr. Sekulic of the Mayo Clinic in Scottsdale, Ariz.
The patients were treated with vismodegib until disease progression, until side effects became intolerable, or until end of study. Patients could stop the drug for up to a month if side effects were intolerable.
There was no standard end point for measuring response in locally advanced BCC when the study was designed. The definition used was a decrease of 30% or more of the externally visible or radiographic dimension or complete resolution of ulceration. Progressive disease was an increase of 20% or more in the externally visible or radiographic dimension, new ulceration, or a new lesion.
The response rate for the metastatic group treated with vismodegib was 30%. All of the responses were partial, defined as an absence of residual BCC in a biopsy specimen. In locally advanced disease, the response rate was 43%. Thirteen of the 63 patients had a complete response. The median duration of response was 7.6 months for metastatic and locally advanced BCC patients (N. Engl. J. Med. 2012;366:2171-9).
Half of the patients stopped the drug early, with 18% of metastatic patients discontinuing because of disease progression. Twenty-five percent of locally advanced patients decided on their own to stop therapy, for unknown reasons, according to the investigators. Adverse events – primarily muscle spasms, weight loss, fatigue, and loss of appetite – took a toll on patients. A quarter reported serious adverse events.
Seven patients died, but the relation to vismodegib is not clear at this point. "The deaths were considered by the site investigator to be unrelated to vismodegib," the investigators wrote.
The locally advanced and metastatic BCC trial was funded by Genentech and was jointly designed by Genetech and Dr. Sekulic. A majority of his colleagues reported being employees and/or receiving grants or financial support from the company.
In an interview, Dr. Ali Hendi, assistant clinical professor of dermatology at Georgetown University Medical Center, Washington, said that he felt the investigators in the study were overly subjective in defining locally advanced tumors. "These are tumors that are aggressive, but that does not make them inoperable in the right hands," said Dr. Hendi, who is also a Mohs surgeon.
He also noted the slim complete response rate, which, when coupled with the toxicity and the high cost of the therapy – which might run as much as $70,000 per treatment course – make it a less desirable therapy for many patients.
"For locally advanced [disease] I think the utility is limited if there is good surgical care available," he said. Vismodegib may be more useful for BCC nevus syndrome and for metastatic disease, where it may be the only therapy available, he added.
Dr. Hendi reported no conflicts.
Study 2: Basal Cell Nevus Syndrome
In a second randomized, double-blind, placebo-controlled trial of 41 patients with basal cell nevus syndrome, Dr. Jean Y. Tang and colleagues found that vismodegib reduced BCC tumors and blocked the development of new BCCs.
Basal cell nevus syndrome is rare, but it causes hundreds or thousands of carcinomas in each patient. There is no therapy, and often patients have to undergo multiple surgeries to remove the tumors.
Patients with the condition have one defective copy of the tumor suppressor gene (PTCH1), which inhibits the hedgehog signaling pathway, noted Dr. Tang of Stanford University (Calif.), and her colleagues.
The study enrolled patients from Sept. 2009 through Jan. 2011, but in Dec. 2010 the data safety and monitoring board decided to end the placebo treatment because the results so highly favored vismodegib. Patients were randomized to placebo or 150 mg daily of the drug for a planned 18 months.
The primary end point was the comparative rate of appearance of new BCCs that were eligible for surgery. Patients were followed for a mean of 8 months. Those who took vismodegib had a much lower rate of surgically eligible disease: 2 vs. 29 cases for placebo. Existing carcinomas shrunk by 65% in vismodegib-treated patients, compared with only 11% for placebo patients. Vismodegib patients also had fewer surgeries than patients receiving placebo: 0.31 per patient compared with 4.4 per patient, respectively.
At 1 month, there was a 90% reduction in hedgehog target-gene expression. There was no residual carcinoma in 83% of the biopsy samples taken from sites where the disease had been judged to be clinically regressed (N. Engl. J. Med. 2012;366:2180-8).
Fifty-four percent of study patients discontinued treatment because of adverse events. Patients treated with vismodegib were more likely to have grade 1 or 2 dysgeusia, muscle cramps, hair loss, and weight loss. They also had more grade 3 or 4 adverse events. The authors said that these side effects were similar to those reported in phase 1 and 2 trials of vismodegib, and also in other studies of therapies that inhibit the hedgehog pathway.
The authors concluded that vismodegib reduces tumor burden and blocks new tumor growth, but whether some of the clinically regressed tumors still harbor residual tumor cells cannot be ruled out – which could explain why there is tumor regrowth after therapy is stopped.
However, the "findings confirm the essential role of the hedgehog pathway in basal cell carcinomas," they wrote.
The trial was funded by Genentech and by grants from the National Institutes of Health, the National Institute of Arthritis and Musculoskeletal and Skin Diseases, the National Cancer Institute, the Swim Across America Foundation, and the Michael J. Rainen Family Foundation. Dr. Tang reported receiving consulting fees from Genentech. Several coauthors of the study reported receiving grants or fees from Genentech.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Major Finding: In the first trial, the response rate for vismodegib-treated patients was 30% for the metastatic group and 43% for the locally advanced BCC group. In the basal cell nevus syndrome trial, vismodegib-treated patients had a lower rate of surgically eligible disease: 2 cases vs. 29 cases for placebo.
Data Source: In the first trial, the data came from a nonrandomized, dual-cohort, multicenter, phase II trial of 96 patients. The data for the second trial came from a randomized, placebo-controlled study of 41 patients.
Disclosures: The locally advanced and metastatic BCC trial was funded by Genentech and was jointly designed by Genentech and Dr. Sekulic. A majority of his colleagues reported being employees and/or receiving grants or financial support from the company. The basal cell nevus syndrome trial was funded by Genentech and by grants from the National Institutes of Health, the National Institute of Arthritis and Musculoskeletal and Skin Diseases, the National Cancer Institute, the Swim Across America Foundation, and the Michael J. Rainen Family Foundation. Dr. Tang reported receiving consulting fees from Genentech. Several coauthors of the study reported receiving grants or fees from Genentech.