User login
Most Lymphomatoid Papulosis Has Benign Course
LAS VEGAS – Although certain characteristics of lymphomatoid papulosis appear be associated with progression to lymphoma, the majority of patients will have a benign course of disease.
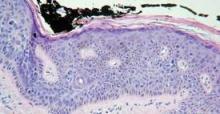
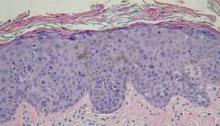
The recurrent papulonodular skin eruption lymphomatoid papulosis (LyP) can be a confusing dermatologic entity because it appears malignant histologically, but it usually follows a clinically benign and indolent course (Arch. Dermatol. 1968;97:23-30). And even the 10%-20% of patients who do progress to lymphoma tend to have less aggressive disease, said Dr. Lawrence E. Gibson, professor of dermatology at the Mayo Clinic in Rochester, Minn. The reasons for this disconnect are not yet known.
"Patients with LyP look like lymphoma under the microscope but have clinically indolent cutaneous disease. ... It is an example of a very important clinical-pathological relationship that really needs to be made to help us understand how to treat patients better," he said.
"The bottom line is most of our patients who have LyP do not go on to have aggressive lymphomas."
To identify which LyP patients are more likely to progress to lymphoma, Dr. Gibson, Dr. Rokea A. el-Azhary, Dr. Aieska de Souza, and their associates conducted a retrospective analysis of 123 patients seen at the Mayo Clinic between 1991 and 2008. The patients were followed for a mean of 4 years (range, 2 months to 14 years). The 65 males and 58 females had a mean age of 47 years (range, 1-83 years), and a mean of 14 lesions (range, 1-100). Most (88%) of the lesions were papules, with a reported mean duration of 5.5 weeks. Pruritis was present in 38% and scar formation in 58%, the researchers reported (J. Am. Acad. Dermatol. 2011 [doi:10.1016/j.jaad.2011.07.012]).
Hematologic malignancies were present in 17 patients (14%). Of those, 10 were cutaneous lymphomas – 8 mycosis fungoides (MF) and 2 anaplastic large-cell lymphomas (ALCL). Hodgkin lymphoma was present in three patients (including the two with ALCL), multiple myeloma or monoclonal gammopathy in three, and myelodysplastic syndrome in one.
"The bottom line is most of our patients who have LyP do not go on to have aggressive lymphomas. They certainly don’t have cytotoxic lymphomas. And if they have lymphoma, they usually have MF. I think that’s somewhat reassuring to us. And for the most part, most of the patients don’t have anything. They have a normal life," Dr. Gibson said at the seminar sponsored by Skin Disease Education Foundation (SDEF).
Of 97 LyP patients for whom original biopsy slides were available, the majority (69) had World Health Organization/European Organization for Research and Treatment of Cancer histologic classification type A, including 35 with immunophenotypic subtype CD8 and 34 with subtype CD4. Another 13 patients had type B lesions (8 CD4, 5 CD8), and 6 had type C, all of which were CD4. The other 9 patients had more than one histologic type (A, B, or C), and/or more than one immunophenotypic subtype (CD4 or CD8). They were designated mixed type.
Clinically, there were no distinguishing features among the subtypes. This finding contrasts with some previous reports that the CD8 subtype might predispose to worse disease outcome (Am. J. Pathol. 1999;155:483-92).
"Our findings indicated that the LyP subtype CD8 does not signify more aggressive disease, a poor prognosis, or an association with malignancy," Dr. Gibson and his colleagues wrote.
Hematologic malignancies were present in 5 of the 9 mixed-type patients (55.5%), compared with 4 of the 34 with A/CD4 (12%), 4 of the 35 A/CD8 (11.5%), 1 of the 8 B/CD4 patients (12.5%), and 1 of the 5 B/CD8 patients (20%). (Two of the 17 malignancies were excluded from analysis because original slides were not available.) The odds ratio for malignancy for the patients with mixed-type LyP versus all other types was a statistically significant 4.33 (P = .03).
In a molecular genetics substudy of 84 LyP lesions from 76 patients, 42 (50%) were positive for clonal T-cell receptor gene rearrangement (TCRGR), 34 (40%) were negative, and 8 (10%) showed equivocal results or had insufficient DNA for analysis. Among the LyP patients who had a hematologic malignancy, 9 of 11 (82%) had positive TCRGR, compared with 30 of 68 (44%) LyP patients without malignancy. That association was also significant, with an odds ratio of 5.7 (P = .02), noted the investigators.
"A positive T-cell receptor gene rearrangement or having more than one type of LyP may have a higher risk of progression to lymphoma, but the evidence is not hard and fast. ... The take-home message is most of these patients do just fine," Dr. Gibson said.
Dr. Gibson stated that he had no relevant financial disclosures or conflicts of interest. SDEF and this news organization are owned by Elsevier.
LAS VEGAS – Although certain characteristics of lymphomatoid papulosis appear be associated with progression to lymphoma, the majority of patients will have a benign course of disease.
The recurrent papulonodular skin eruption lymphomatoid papulosis (LyP) can be a confusing dermatologic entity because it appears malignant histologically, but it usually follows a clinically benign and indolent course (Arch. Dermatol. 1968;97:23-30). And even the 10%-20% of patients who do progress to lymphoma tend to have less aggressive disease, said Dr. Lawrence E. Gibson, professor of dermatology at the Mayo Clinic in Rochester, Minn. The reasons for this disconnect are not yet known.
"Patients with LyP look like lymphoma under the microscope but have clinically indolent cutaneous disease. ... It is an example of a very important clinical-pathological relationship that really needs to be made to help us understand how to treat patients better," he said.
"The bottom line is most of our patients who have LyP do not go on to have aggressive lymphomas."
To identify which LyP patients are more likely to progress to lymphoma, Dr. Gibson, Dr. Rokea A. el-Azhary, Dr. Aieska de Souza, and their associates conducted a retrospective analysis of 123 patients seen at the Mayo Clinic between 1991 and 2008. The patients were followed for a mean of 4 years (range, 2 months to 14 years). The 65 males and 58 females had a mean age of 47 years (range, 1-83 years), and a mean of 14 lesions (range, 1-100). Most (88%) of the lesions were papules, with a reported mean duration of 5.5 weeks. Pruritis was present in 38% and scar formation in 58%, the researchers reported (J. Am. Acad. Dermatol. 2011 [doi:10.1016/j.jaad.2011.07.012]).
Hematologic malignancies were present in 17 patients (14%). Of those, 10 were cutaneous lymphomas – 8 mycosis fungoides (MF) and 2 anaplastic large-cell lymphomas (ALCL). Hodgkin lymphoma was present in three patients (including the two with ALCL), multiple myeloma or monoclonal gammopathy in three, and myelodysplastic syndrome in one.
"The bottom line is most of our patients who have LyP do not go on to have aggressive lymphomas. They certainly don’t have cytotoxic lymphomas. And if they have lymphoma, they usually have MF. I think that’s somewhat reassuring to us. And for the most part, most of the patients don’t have anything. They have a normal life," Dr. Gibson said at the seminar sponsored by Skin Disease Education Foundation (SDEF).
Of 97 LyP patients for whom original biopsy slides were available, the majority (69) had World Health Organization/European Organization for Research and Treatment of Cancer histologic classification type A, including 35 with immunophenotypic subtype CD8 and 34 with subtype CD4. Another 13 patients had type B lesions (8 CD4, 5 CD8), and 6 had type C, all of which were CD4. The other 9 patients had more than one histologic type (A, B, or C), and/or more than one immunophenotypic subtype (CD4 or CD8). They were designated mixed type.
Clinically, there were no distinguishing features among the subtypes. This finding contrasts with some previous reports that the CD8 subtype might predispose to worse disease outcome (Am. J. Pathol. 1999;155:483-92).
"Our findings indicated that the LyP subtype CD8 does not signify more aggressive disease, a poor prognosis, or an association with malignancy," Dr. Gibson and his colleagues wrote.
Hematologic malignancies were present in 5 of the 9 mixed-type patients (55.5%), compared with 4 of the 34 with A/CD4 (12%), 4 of the 35 A/CD8 (11.5%), 1 of the 8 B/CD4 patients (12.5%), and 1 of the 5 B/CD8 patients (20%). (Two of the 17 malignancies were excluded from analysis because original slides were not available.) The odds ratio for malignancy for the patients with mixed-type LyP versus all other types was a statistically significant 4.33 (P = .03).
In a molecular genetics substudy of 84 LyP lesions from 76 patients, 42 (50%) were positive for clonal T-cell receptor gene rearrangement (TCRGR), 34 (40%) were negative, and 8 (10%) showed equivocal results or had insufficient DNA for analysis. Among the LyP patients who had a hematologic malignancy, 9 of 11 (82%) had positive TCRGR, compared with 30 of 68 (44%) LyP patients without malignancy. That association was also significant, with an odds ratio of 5.7 (P = .02), noted the investigators.
"A positive T-cell receptor gene rearrangement or having more than one type of LyP may have a higher risk of progression to lymphoma, but the evidence is not hard and fast. ... The take-home message is most of these patients do just fine," Dr. Gibson said.
Dr. Gibson stated that he had no relevant financial disclosures or conflicts of interest. SDEF and this news organization are owned by Elsevier.
LAS VEGAS – Although certain characteristics of lymphomatoid papulosis appear be associated with progression to lymphoma, the majority of patients will have a benign course of disease.
The recurrent papulonodular skin eruption lymphomatoid papulosis (LyP) can be a confusing dermatologic entity because it appears malignant histologically, but it usually follows a clinically benign and indolent course (Arch. Dermatol. 1968;97:23-30). And even the 10%-20% of patients who do progress to lymphoma tend to have less aggressive disease, said Dr. Lawrence E. Gibson, professor of dermatology at the Mayo Clinic in Rochester, Minn. The reasons for this disconnect are not yet known.
"Patients with LyP look like lymphoma under the microscope but have clinically indolent cutaneous disease. ... It is an example of a very important clinical-pathological relationship that really needs to be made to help us understand how to treat patients better," he said.
"The bottom line is most of our patients who have LyP do not go on to have aggressive lymphomas."
To identify which LyP patients are more likely to progress to lymphoma, Dr. Gibson, Dr. Rokea A. el-Azhary, Dr. Aieska de Souza, and their associates conducted a retrospective analysis of 123 patients seen at the Mayo Clinic between 1991 and 2008. The patients were followed for a mean of 4 years (range, 2 months to 14 years). The 65 males and 58 females had a mean age of 47 years (range, 1-83 years), and a mean of 14 lesions (range, 1-100). Most (88%) of the lesions were papules, with a reported mean duration of 5.5 weeks. Pruritis was present in 38% and scar formation in 58%, the researchers reported (J. Am. Acad. Dermatol. 2011 [doi:10.1016/j.jaad.2011.07.012]).
Hematologic malignancies were present in 17 patients (14%). Of those, 10 were cutaneous lymphomas – 8 mycosis fungoides (MF) and 2 anaplastic large-cell lymphomas (ALCL). Hodgkin lymphoma was present in three patients (including the two with ALCL), multiple myeloma or monoclonal gammopathy in three, and myelodysplastic syndrome in one.
"The bottom line is most of our patients who have LyP do not go on to have aggressive lymphomas. They certainly don’t have cytotoxic lymphomas. And if they have lymphoma, they usually have MF. I think that’s somewhat reassuring to us. And for the most part, most of the patients don’t have anything. They have a normal life," Dr. Gibson said at the seminar sponsored by Skin Disease Education Foundation (SDEF).
Of 97 LyP patients for whom original biopsy slides were available, the majority (69) had World Health Organization/European Organization for Research and Treatment of Cancer histologic classification type A, including 35 with immunophenotypic subtype CD8 and 34 with subtype CD4. Another 13 patients had type B lesions (8 CD4, 5 CD8), and 6 had type C, all of which were CD4. The other 9 patients had more than one histologic type (A, B, or C), and/or more than one immunophenotypic subtype (CD4 or CD8). They were designated mixed type.
Clinically, there were no distinguishing features among the subtypes. This finding contrasts with some previous reports that the CD8 subtype might predispose to worse disease outcome (Am. J. Pathol. 1999;155:483-92).
"Our findings indicated that the LyP subtype CD8 does not signify more aggressive disease, a poor prognosis, or an association with malignancy," Dr. Gibson and his colleagues wrote.
Hematologic malignancies were present in 5 of the 9 mixed-type patients (55.5%), compared with 4 of the 34 with A/CD4 (12%), 4 of the 35 A/CD8 (11.5%), 1 of the 8 B/CD4 patients (12.5%), and 1 of the 5 B/CD8 patients (20%). (Two of the 17 malignancies were excluded from analysis because original slides were not available.) The odds ratio for malignancy for the patients with mixed-type LyP versus all other types was a statistically significant 4.33 (P = .03).
In a molecular genetics substudy of 84 LyP lesions from 76 patients, 42 (50%) were positive for clonal T-cell receptor gene rearrangement (TCRGR), 34 (40%) were negative, and 8 (10%) showed equivocal results or had insufficient DNA for analysis. Among the LyP patients who had a hematologic malignancy, 9 of 11 (82%) had positive TCRGR, compared with 30 of 68 (44%) LyP patients without malignancy. That association was also significant, with an odds ratio of 5.7 (P = .02), noted the investigators.
"A positive T-cell receptor gene rearrangement or having more than one type of LyP may have a higher risk of progression to lymphoma, but the evidence is not hard and fast. ... The take-home message is most of these patients do just fine," Dr. Gibson said.
Dr. Gibson stated that he had no relevant financial disclosures or conflicts of interest. SDEF and this news organization are owned by Elsevier.
FROM THE SDEF LAS VEGAS DERMATOLOGY SEMINAR
Major Finding: Hematologic malignancies were present in 17 patients (14%).
Data Source: Retrospective analysis of 123 LyP patients seen at the Mayo Clinic between 1991 and 2008.
Disclosures: Dr. Gibson has no relevant financial disclosures or conflicts of interest. SDEF and this news organization are owned by Elsevier.
Are At-Home Devices a Threat?: The Skinny Podcast
In this month's podcast, Reporter Naseem Miller interviews Dr. Ashley Wysong, a former six-time NCAA All-American and national champion in middle distance running, about a recent survey that found nearly half of collegiate athletes don't use sunscreen.
Dr. Sheila Fallon Friedlander discusses who should be receiving vitamin D supplementation, and Dr. Ilona Frieden says that growing evidence suggests propranolol is a "remarkably effective treatment in shrinking hemangioma growth, even in children whose hemangiomas are already fully grown in some cases."
In this month's Cosmetic Counter segment, Dr. Lily Talakoub discusses what at-home devices are worth your patient's investment. And last but not least, Dr. Alan Rockoff shares more patient-story humor.
Don't miss another episode of The Skinny Podcast; subscribe on iTunes!
In this month's podcast, Reporter Naseem Miller interviews Dr. Ashley Wysong, a former six-time NCAA All-American and national champion in middle distance running, about a recent survey that found nearly half of collegiate athletes don't use sunscreen.
Dr. Sheila Fallon Friedlander discusses who should be receiving vitamin D supplementation, and Dr. Ilona Frieden says that growing evidence suggests propranolol is a "remarkably effective treatment in shrinking hemangioma growth, even in children whose hemangiomas are already fully grown in some cases."
In this month's Cosmetic Counter segment, Dr. Lily Talakoub discusses what at-home devices are worth your patient's investment. And last but not least, Dr. Alan Rockoff shares more patient-story humor.
Don't miss another episode of The Skinny Podcast; subscribe on iTunes!
In this month's podcast, Reporter Naseem Miller interviews Dr. Ashley Wysong, a former six-time NCAA All-American and national champion in middle distance running, about a recent survey that found nearly half of collegiate athletes don't use sunscreen.
Dr. Sheila Fallon Friedlander discusses who should be receiving vitamin D supplementation, and Dr. Ilona Frieden says that growing evidence suggests propranolol is a "remarkably effective treatment in shrinking hemangioma growth, even in children whose hemangiomas are already fully grown in some cases."
In this month's Cosmetic Counter segment, Dr. Lily Talakoub discusses what at-home devices are worth your patient's investment. And last but not least, Dr. Alan Rockoff shares more patient-story humor.
Don't miss another episode of The Skinny Podcast; subscribe on iTunes!
Hospital Setting May Pose Collegial Challenges in Mohs
SAN DIEGO – It would be somewhat of an understatement to say that Mohs surgery was not particularly welcomed at hospitals where Dr. Rainer Sachse used to practice.
As a plastic and reconstructive surgeon, he had hoped to utilize the technique to treat extensive, high-stage tumors in high-risk patients within a hospital setting, believing that the literature convincingly suggested that Mohs offered the best hope for cancer control in such patients.
Nurses, he recalled, considered Mohs a "slow, mutilating, expensive" procedure. Surgeons felt it was unnecessary. Pathologists thought their traditional methods "worked perfectly well."
Dermatologists seemed preoccupied by turf battles between the American Society for Mohs Surgery (ASMS) and the American College of Mohs Surgery. Dermatology residents had no time, and plastic surgery residents were uninterested, Dr. Sachse said at the meeting sponsored by the ASMS.
He pressed on, though, inspired by cases in which conventional excisions and standard therapy mutilated patients or led to their untimely deaths from tumors with continuous growth patterns.
He observed colleagues and sought out preceptorships, pored over the literature, and took a Mohs surgery course before beginning to try small cases with a pathologist who had received dermatopathology training. Finally, he began to change minds and take on more challenging cases.
Seeing the results, "a pathologist who was initially very skeptical eventually became a strong supporter," said Dr. Sachse, who is in private practice in Fort Lauderdale, Fla.
Hospital-based Mohs surgery has a role in the treatment of "psychologically problematic" patients, including those with severe anxiety and/or dementia; patients with high-risk medical conditions, including ischemic heart disease, severe arrhythmias, significant pulmonary insufficiencies, or coagulopathies; the physical handicapped; and the morbidly obese, he said.
Additionally, some hospitalized patients require urgent care, what Dr. Sachse terms "emergency Mohs surgery." He offered, as an example, a leukemia patient with skin necrosis of the scalp that was doubling in size every 24 hours. A biopsy revealed that the patient had a zygomycete fungal infection.
Mohs micrographic surgery was followed by topical and oral antifungal therapy and, after a delay, a skin graft, for a clinically and aesthetically acceptable result, he said.
With unusual and complex cases, it is important to build teamwork within the hospital since a multispecialty approach is necessary for cases that invade multiple structures of the head and neck, said Dr. Sachse.
In such cases, the concept of complete margin control must be emphasized, since many colleagues in otolaryngology, plastic surgery, and pathology will be unfamiliar with the basic tenets of the approach and the precision required to achieving those goals, he noted.
He learned to counter perceptions of Mohs surgery as tedious and slow by using careful planning and documentation of results in patients who might have been previously considered inoperable.
Education helps, as does realistic scheduling of operating rooms for the time required for extensive debridement, meticulous staging, excision, and repair.
Presenting Mohs cases to a hospital tumor board can be illuminating to the uninitiated, and the cases themselves are "very rewarding," he said.
Dr. Sachse cautioned that surgeons taking on highly complex cases should "be prepared to meet a patient you may not be able to cure." That said, "Mohs is a surgical tool which can and should be used for very extensive tumors. The complexity of the margins may increase exponentially, but you can always cut quicker than any tumor can grow."
Dr. Sachse said that he had no relevant disclosures.
plastic and reconstructive surgeon, high-stage tumors, high-risk patients, hospital setting, cancer control, Dermatologists, American Society for Mohs Surgery, ASMS, the American College of Mohs Surgery,
SAN DIEGO – It would be somewhat of an understatement to say that Mohs surgery was not particularly welcomed at hospitals where Dr. Rainer Sachse used to practice.
As a plastic and reconstructive surgeon, he had hoped to utilize the technique to treat extensive, high-stage tumors in high-risk patients within a hospital setting, believing that the literature convincingly suggested that Mohs offered the best hope for cancer control in such patients.
Nurses, he recalled, considered Mohs a "slow, mutilating, expensive" procedure. Surgeons felt it was unnecessary. Pathologists thought their traditional methods "worked perfectly well."
Dermatologists seemed preoccupied by turf battles between the American Society for Mohs Surgery (ASMS) and the American College of Mohs Surgery. Dermatology residents had no time, and plastic surgery residents were uninterested, Dr. Sachse said at the meeting sponsored by the ASMS.
He pressed on, though, inspired by cases in which conventional excisions and standard therapy mutilated patients or led to their untimely deaths from tumors with continuous growth patterns.
He observed colleagues and sought out preceptorships, pored over the literature, and took a Mohs surgery course before beginning to try small cases with a pathologist who had received dermatopathology training. Finally, he began to change minds and take on more challenging cases.
Seeing the results, "a pathologist who was initially very skeptical eventually became a strong supporter," said Dr. Sachse, who is in private practice in Fort Lauderdale, Fla.
Hospital-based Mohs surgery has a role in the treatment of "psychologically problematic" patients, including those with severe anxiety and/or dementia; patients with high-risk medical conditions, including ischemic heart disease, severe arrhythmias, significant pulmonary insufficiencies, or coagulopathies; the physical handicapped; and the morbidly obese, he said.
Additionally, some hospitalized patients require urgent care, what Dr. Sachse terms "emergency Mohs surgery." He offered, as an example, a leukemia patient with skin necrosis of the scalp that was doubling in size every 24 hours. A biopsy revealed that the patient had a zygomycete fungal infection.
Mohs micrographic surgery was followed by topical and oral antifungal therapy and, after a delay, a skin graft, for a clinically and aesthetically acceptable result, he said.
With unusual and complex cases, it is important to build teamwork within the hospital since a multispecialty approach is necessary for cases that invade multiple structures of the head and neck, said Dr. Sachse.
In such cases, the concept of complete margin control must be emphasized, since many colleagues in otolaryngology, plastic surgery, and pathology will be unfamiliar with the basic tenets of the approach and the precision required to achieving those goals, he noted.
He learned to counter perceptions of Mohs surgery as tedious and slow by using careful planning and documentation of results in patients who might have been previously considered inoperable.
Education helps, as does realistic scheduling of operating rooms for the time required for extensive debridement, meticulous staging, excision, and repair.
Presenting Mohs cases to a hospital tumor board can be illuminating to the uninitiated, and the cases themselves are "very rewarding," he said.
Dr. Sachse cautioned that surgeons taking on highly complex cases should "be prepared to meet a patient you may not be able to cure." That said, "Mohs is a surgical tool which can and should be used for very extensive tumors. The complexity of the margins may increase exponentially, but you can always cut quicker than any tumor can grow."
Dr. Sachse said that he had no relevant disclosures.
SAN DIEGO – It would be somewhat of an understatement to say that Mohs surgery was not particularly welcomed at hospitals where Dr. Rainer Sachse used to practice.
As a plastic and reconstructive surgeon, he had hoped to utilize the technique to treat extensive, high-stage tumors in high-risk patients within a hospital setting, believing that the literature convincingly suggested that Mohs offered the best hope for cancer control in such patients.
Nurses, he recalled, considered Mohs a "slow, mutilating, expensive" procedure. Surgeons felt it was unnecessary. Pathologists thought their traditional methods "worked perfectly well."
Dermatologists seemed preoccupied by turf battles between the American Society for Mohs Surgery (ASMS) and the American College of Mohs Surgery. Dermatology residents had no time, and plastic surgery residents were uninterested, Dr. Sachse said at the meeting sponsored by the ASMS.
He pressed on, though, inspired by cases in which conventional excisions and standard therapy mutilated patients or led to their untimely deaths from tumors with continuous growth patterns.
He observed colleagues and sought out preceptorships, pored over the literature, and took a Mohs surgery course before beginning to try small cases with a pathologist who had received dermatopathology training. Finally, he began to change minds and take on more challenging cases.
Seeing the results, "a pathologist who was initially very skeptical eventually became a strong supporter," said Dr. Sachse, who is in private practice in Fort Lauderdale, Fla.
Hospital-based Mohs surgery has a role in the treatment of "psychologically problematic" patients, including those with severe anxiety and/or dementia; patients with high-risk medical conditions, including ischemic heart disease, severe arrhythmias, significant pulmonary insufficiencies, or coagulopathies; the physical handicapped; and the morbidly obese, he said.
Additionally, some hospitalized patients require urgent care, what Dr. Sachse terms "emergency Mohs surgery." He offered, as an example, a leukemia patient with skin necrosis of the scalp that was doubling in size every 24 hours. A biopsy revealed that the patient had a zygomycete fungal infection.
Mohs micrographic surgery was followed by topical and oral antifungal therapy and, after a delay, a skin graft, for a clinically and aesthetically acceptable result, he said.
With unusual and complex cases, it is important to build teamwork within the hospital since a multispecialty approach is necessary for cases that invade multiple structures of the head and neck, said Dr. Sachse.
In such cases, the concept of complete margin control must be emphasized, since many colleagues in otolaryngology, plastic surgery, and pathology will be unfamiliar with the basic tenets of the approach and the precision required to achieving those goals, he noted.
He learned to counter perceptions of Mohs surgery as tedious and slow by using careful planning and documentation of results in patients who might have been previously considered inoperable.
Education helps, as does realistic scheduling of operating rooms for the time required for extensive debridement, meticulous staging, excision, and repair.
Presenting Mohs cases to a hospital tumor board can be illuminating to the uninitiated, and the cases themselves are "very rewarding," he said.
Dr. Sachse cautioned that surgeons taking on highly complex cases should "be prepared to meet a patient you may not be able to cure." That said, "Mohs is a surgical tool which can and should be used for very extensive tumors. The complexity of the margins may increase exponentially, but you can always cut quicker than any tumor can grow."
Dr. Sachse said that he had no relevant disclosures.
plastic and reconstructive surgeon, high-stage tumors, high-risk patients, hospital setting, cancer control, Dermatologists, American Society for Mohs Surgery, ASMS, the American College of Mohs Surgery,
plastic and reconstructive surgeon, high-stage tumors, high-risk patients, hospital setting, cancer control, Dermatologists, American Society for Mohs Surgery, ASMS, the American College of Mohs Surgery,
EXPERT ANALYSIS FROM A MEETING SPONSORED BY THE AMERICAN SOCIETY FOR MOHS SURGERY
Lines Blur Between Dysplasia, Carcinoma in Situ
SAN DIEGO – It’s not getting any easier to make the histological distinction between carcinoma in situ and simple dysplasia in a sun-damaged epidermis, but it may be getting less critical to get it right, noted Dr. John B. Campbell.
During his presentation at a meeting sponsored by the American Society for Mohs Surgery, Dr. Campbell, a Mohs pathologist in private practice in San Diego, showed slides depicting disorderly cells that typify dysplasia extending high up into the epidermis along the edges of specimens containing in situ carcinoma. "There are a lot of things that go along with dysplastic changes," he said. "The nuclei become larger; they stain darker; they have less cytoplasm so that the N/C [nuclear to cytoplasmic] ratio increases."
"At some place along the edges of the specimen you’re going to have to draw a line between in situ carcinoma and simple dysplasia. There are gradations that are somewhat subjective."
Pathologists commonly envision a line midway through the epidermis. If the dark and disorganized cells reach that point, they call it "moderate dysplasia." If they’re clumped in the lower third of the epidermis, it merits a call of "mild dysplasia," and if they extend to near the surface, it’s "severe."
"Dysplastic changes that are wall-to-wall, top-to-bottom are carcinoma in situ," he said. "You’ll need to develop criteria on your own."
In the meantime, the pressure to make the right call may be easing.
"What I’ve seen happening in the last few years is that we’re becoming less and less sensitive about dysplasia at the edges of in situ carcinomas because we can treat them so easily topically," said Dr. Campbell. Beyond watchful waiting or freezing, such regions can now be well managed with topical therapies such as Aldara (imiquimod) or Efudex (fluorouracil).
"Once we’ve cleared the unequivocal carcinoma in situ, people are letting a significant amount of dysplasia remain and then treating it in follow-up.
"There’s really no downside to [conservative or topical management] as long as the patient is reliable," he said.
Taking a careful look at the site during follow-up visits will easily reveal any cell changes that might prompt a fresh biopsy. One finding of note in such cases is the proclivity of dysplastic cells to take a downward course along appendage structures such as eccrine ducts and follicles in the epithelium. "They can go down quite deep ... maybe 2 mm deep to the surface," he said.
In his practice, Dr. Campbell and his colleagues characterize such findings as "advanced actinic keratoses," and note the presence of dysplastic cells along appendage structures.
Dr. Campbell reported no relevant disclosures.
SAN DIEGO – It’s not getting any easier to make the histological distinction between carcinoma in situ and simple dysplasia in a sun-damaged epidermis, but it may be getting less critical to get it right, noted Dr. John B. Campbell.
During his presentation at a meeting sponsored by the American Society for Mohs Surgery, Dr. Campbell, a Mohs pathologist in private practice in San Diego, showed slides depicting disorderly cells that typify dysplasia extending high up into the epidermis along the edges of specimens containing in situ carcinoma. "There are a lot of things that go along with dysplastic changes," he said. "The nuclei become larger; they stain darker; they have less cytoplasm so that the N/C [nuclear to cytoplasmic] ratio increases."
"At some place along the edges of the specimen you’re going to have to draw a line between in situ carcinoma and simple dysplasia. There are gradations that are somewhat subjective."
Pathologists commonly envision a line midway through the epidermis. If the dark and disorganized cells reach that point, they call it "moderate dysplasia." If they’re clumped in the lower third of the epidermis, it merits a call of "mild dysplasia," and if they extend to near the surface, it’s "severe."
"Dysplastic changes that are wall-to-wall, top-to-bottom are carcinoma in situ," he said. "You’ll need to develop criteria on your own."
In the meantime, the pressure to make the right call may be easing.
"What I’ve seen happening in the last few years is that we’re becoming less and less sensitive about dysplasia at the edges of in situ carcinomas because we can treat them so easily topically," said Dr. Campbell. Beyond watchful waiting or freezing, such regions can now be well managed with topical therapies such as Aldara (imiquimod) or Efudex (fluorouracil).
"Once we’ve cleared the unequivocal carcinoma in situ, people are letting a significant amount of dysplasia remain and then treating it in follow-up.
"There’s really no downside to [conservative or topical management] as long as the patient is reliable," he said.
Taking a careful look at the site during follow-up visits will easily reveal any cell changes that might prompt a fresh biopsy. One finding of note in such cases is the proclivity of dysplastic cells to take a downward course along appendage structures such as eccrine ducts and follicles in the epithelium. "They can go down quite deep ... maybe 2 mm deep to the surface," he said.
In his practice, Dr. Campbell and his colleagues characterize such findings as "advanced actinic keratoses," and note the presence of dysplastic cells along appendage structures.
Dr. Campbell reported no relevant disclosures.
SAN DIEGO – It’s not getting any easier to make the histological distinction between carcinoma in situ and simple dysplasia in a sun-damaged epidermis, but it may be getting less critical to get it right, noted Dr. John B. Campbell.
During his presentation at a meeting sponsored by the American Society for Mohs Surgery, Dr. Campbell, a Mohs pathologist in private practice in San Diego, showed slides depicting disorderly cells that typify dysplasia extending high up into the epidermis along the edges of specimens containing in situ carcinoma. "There are a lot of things that go along with dysplastic changes," he said. "The nuclei become larger; they stain darker; they have less cytoplasm so that the N/C [nuclear to cytoplasmic] ratio increases."
"At some place along the edges of the specimen you’re going to have to draw a line between in situ carcinoma and simple dysplasia. There are gradations that are somewhat subjective."
Pathologists commonly envision a line midway through the epidermis. If the dark and disorganized cells reach that point, they call it "moderate dysplasia." If they’re clumped in the lower third of the epidermis, it merits a call of "mild dysplasia," and if they extend to near the surface, it’s "severe."
"Dysplastic changes that are wall-to-wall, top-to-bottom are carcinoma in situ," he said. "You’ll need to develop criteria on your own."
In the meantime, the pressure to make the right call may be easing.
"What I’ve seen happening in the last few years is that we’re becoming less and less sensitive about dysplasia at the edges of in situ carcinomas because we can treat them so easily topically," said Dr. Campbell. Beyond watchful waiting or freezing, such regions can now be well managed with topical therapies such as Aldara (imiquimod) or Efudex (fluorouracil).
"Once we’ve cleared the unequivocal carcinoma in situ, people are letting a significant amount of dysplasia remain and then treating it in follow-up.
"There’s really no downside to [conservative or topical management] as long as the patient is reliable," he said.
Taking a careful look at the site during follow-up visits will easily reveal any cell changes that might prompt a fresh biopsy. One finding of note in such cases is the proclivity of dysplastic cells to take a downward course along appendage structures such as eccrine ducts and follicles in the epithelium. "They can go down quite deep ... maybe 2 mm deep to the surface," he said.
In his practice, Dr. Campbell and his colleagues characterize such findings as "advanced actinic keratoses," and note the presence of dysplastic cells along appendage structures.
Dr. Campbell reported no relevant disclosures.
EXPERT ANALYSIS FROM A MEETING SPONSORED BY THE AMERICAN SOCIETY FOR MOHS SURGERY
Mohs Microscope Shopping? Expert Describes Must Haves
SAN DIEGO – Like choosing a cell phone, stereo system, or new car, shopping for a microscope suitable for a Mohs surgery practice can be a delicate balance between what is needed, what is wanted, and how much money there is to spend.
Consumer Reports doesn’t have a special Mohs edition to help out on the microscope hunt, but Dr. Kenneth G. Gross offered his perspective on must-haves, don’t-wants, and "highly desirable features" to look for in a microscope designated for the special needs inherent in Mohs.
For starters, he emphasized that an appropriate microscope is a necessity, not a luxury.
"That little student microscope you used in medical school is really not the kind of microscope you want to use in a Mohs practice. You don’t bring a knife to a gun fight," he said at a meeting sponsored by the American Society for Mohs Surgery.
Dr. Gross, a dermatologic surgeon practicing in San Diego, explained that several competitive companies market technical microscopes that work well for Mohs and that all make quality products. He stated no personal preference but recommended shopping for a microscope that contains certain specifications, no matter the manufacturer.
Binocular or Trinocular?
Mohs surgeons really need a microscope with a teaching head – a binocular design – and ideally, the addition of a trinocular scope for photography.
A dual head is valuable not only for teaching or consultation with a resident, colleague, or pathologist, but it also allows for demonstrating to a technician the errors that can thwart a complete and accurate view of a specimen.
A third head for a camera is not 100% necessary, but is very nice to have, according to Dr. Gross.
"Photography is so easy nowadays, it is really stone simple. Everything is through the viewfinder and through the lens," automatically optimizing the F-stop, speed, and color balance.
"That little student microscope you used in medical school is really not the kind of microscope you want to use in a Mohs practice."
"Really, there’s no trick to it," said Dr. Gross. "You just attach a digital camera on top and shoot."
Point it Out
An adjustable, lighted pointer further assists communication and identification of focal regions on a slide.
"If there’s something I don’t see, [the pathologist] can flip on the pointer and say, "There you are, man. There it is."
Focusing on Lenses
Objective lenses, those closest to the specimen, come in three quality levels, and the middle level is acceptable for Mohs, according to Dr. Gross.
The lowest power objective lens should be no bigger than a 2.5x; a 2x is preferable. A 1x or 1.5x is fine as well, but not necessary, he said. With a 2.5x objective lens, "You can orient yourself to a pretty big specimen ... without getting lost."
He compared the view of a large specimen with a 10x objective lens to looking at ink dots and then trying to figure out how they combine to form letters on a book page.
Nose pieces hold five objective lenses, with a 2x, 4x, 10x, 20x, and 40x of middle-quality a good selection.
"You do not need an oil immersion lens on your microscope," he said.
Proper lighting with different lenses is achieved by using swing-out condensers, the best of which "clunk" into place like a solid car door.
The ones that freely slide from side to side are "kind of a piece of junk in my opinion," he said, "because they get out of focus easily."
When it comes to ocular lenses, pony up for the focusable, highest-quality, wide-angle options available, Dr. Gross said.
Each person viewing the specimen should be able to separately focus the image to accommodate individual differences in visual acuity.
Angle for Tilt Heads
Opting for a system with tilt heads isn’t imperative, but is wise if more than one doctor is sharing the microscope, according to Dr. Gross.
"Unless you’re identical twins, you’re going to [have one doctor who is] taller or shorter, sits up straight or slumped, use[s] different style chairs. If you have tilt heads, they’re really easy to adjust."
You can economize, though, by foregoing an option that allows the microscope heads to push in or out. "That’s a waste of money," he said.
Dr. Gross said he had no financial disclosures with regard to any company that manufactures or maintains microscopes used in Mohs surgery.
SAN DIEGO – Like choosing a cell phone, stereo system, or new car, shopping for a microscope suitable for a Mohs surgery practice can be a delicate balance between what is needed, what is wanted, and how much money there is to spend.
Consumer Reports doesn’t have a special Mohs edition to help out on the microscope hunt, but Dr. Kenneth G. Gross offered his perspective on must-haves, don’t-wants, and "highly desirable features" to look for in a microscope designated for the special needs inherent in Mohs.
For starters, he emphasized that an appropriate microscope is a necessity, not a luxury.
"That little student microscope you used in medical school is really not the kind of microscope you want to use in a Mohs practice. You don’t bring a knife to a gun fight," he said at a meeting sponsored by the American Society for Mohs Surgery.
Dr. Gross, a dermatologic surgeon practicing in San Diego, explained that several competitive companies market technical microscopes that work well for Mohs and that all make quality products. He stated no personal preference but recommended shopping for a microscope that contains certain specifications, no matter the manufacturer.
Binocular or Trinocular?
Mohs surgeons really need a microscope with a teaching head – a binocular design – and ideally, the addition of a trinocular scope for photography.
A dual head is valuable not only for teaching or consultation with a resident, colleague, or pathologist, but it also allows for demonstrating to a technician the errors that can thwart a complete and accurate view of a specimen.
A third head for a camera is not 100% necessary, but is very nice to have, according to Dr. Gross.
"Photography is so easy nowadays, it is really stone simple. Everything is through the viewfinder and through the lens," automatically optimizing the F-stop, speed, and color balance.
"That little student microscope you used in medical school is really not the kind of microscope you want to use in a Mohs practice."
"Really, there’s no trick to it," said Dr. Gross. "You just attach a digital camera on top and shoot."
Point it Out
An adjustable, lighted pointer further assists communication and identification of focal regions on a slide.
"If there’s something I don’t see, [the pathologist] can flip on the pointer and say, "There you are, man. There it is."
Focusing on Lenses
Objective lenses, those closest to the specimen, come in three quality levels, and the middle level is acceptable for Mohs, according to Dr. Gross.
The lowest power objective lens should be no bigger than a 2.5x; a 2x is preferable. A 1x or 1.5x is fine as well, but not necessary, he said. With a 2.5x objective lens, "You can orient yourself to a pretty big specimen ... without getting lost."
He compared the view of a large specimen with a 10x objective lens to looking at ink dots and then trying to figure out how they combine to form letters on a book page.
Nose pieces hold five objective lenses, with a 2x, 4x, 10x, 20x, and 40x of middle-quality a good selection.
"You do not need an oil immersion lens on your microscope," he said.
Proper lighting with different lenses is achieved by using swing-out condensers, the best of which "clunk" into place like a solid car door.
The ones that freely slide from side to side are "kind of a piece of junk in my opinion," he said, "because they get out of focus easily."
When it comes to ocular lenses, pony up for the focusable, highest-quality, wide-angle options available, Dr. Gross said.
Each person viewing the specimen should be able to separately focus the image to accommodate individual differences in visual acuity.
Angle for Tilt Heads
Opting for a system with tilt heads isn’t imperative, but is wise if more than one doctor is sharing the microscope, according to Dr. Gross.
"Unless you’re identical twins, you’re going to [have one doctor who is] taller or shorter, sits up straight or slumped, use[s] different style chairs. If you have tilt heads, they’re really easy to adjust."
You can economize, though, by foregoing an option that allows the microscope heads to push in or out. "That’s a waste of money," he said.
Dr. Gross said he had no financial disclosures with regard to any company that manufactures or maintains microscopes used in Mohs surgery.
SAN DIEGO – Like choosing a cell phone, stereo system, or new car, shopping for a microscope suitable for a Mohs surgery practice can be a delicate balance between what is needed, what is wanted, and how much money there is to spend.
Consumer Reports doesn’t have a special Mohs edition to help out on the microscope hunt, but Dr. Kenneth G. Gross offered his perspective on must-haves, don’t-wants, and "highly desirable features" to look for in a microscope designated for the special needs inherent in Mohs.
For starters, he emphasized that an appropriate microscope is a necessity, not a luxury.
"That little student microscope you used in medical school is really not the kind of microscope you want to use in a Mohs practice. You don’t bring a knife to a gun fight," he said at a meeting sponsored by the American Society for Mohs Surgery.
Dr. Gross, a dermatologic surgeon practicing in San Diego, explained that several competitive companies market technical microscopes that work well for Mohs and that all make quality products. He stated no personal preference but recommended shopping for a microscope that contains certain specifications, no matter the manufacturer.
Binocular or Trinocular?
Mohs surgeons really need a microscope with a teaching head – a binocular design – and ideally, the addition of a trinocular scope for photography.
A dual head is valuable not only for teaching or consultation with a resident, colleague, or pathologist, but it also allows for demonstrating to a technician the errors that can thwart a complete and accurate view of a specimen.
A third head for a camera is not 100% necessary, but is very nice to have, according to Dr. Gross.
"Photography is so easy nowadays, it is really stone simple. Everything is through the viewfinder and through the lens," automatically optimizing the F-stop, speed, and color balance.
"That little student microscope you used in medical school is really not the kind of microscope you want to use in a Mohs practice."
"Really, there’s no trick to it," said Dr. Gross. "You just attach a digital camera on top and shoot."
Point it Out
An adjustable, lighted pointer further assists communication and identification of focal regions on a slide.
"If there’s something I don’t see, [the pathologist] can flip on the pointer and say, "There you are, man. There it is."
Focusing on Lenses
Objective lenses, those closest to the specimen, come in three quality levels, and the middle level is acceptable for Mohs, according to Dr. Gross.
The lowest power objective lens should be no bigger than a 2.5x; a 2x is preferable. A 1x or 1.5x is fine as well, but not necessary, he said. With a 2.5x objective lens, "You can orient yourself to a pretty big specimen ... without getting lost."
He compared the view of a large specimen with a 10x objective lens to looking at ink dots and then trying to figure out how they combine to form letters on a book page.
Nose pieces hold five objective lenses, with a 2x, 4x, 10x, 20x, and 40x of middle-quality a good selection.
"You do not need an oil immersion lens on your microscope," he said.
Proper lighting with different lenses is achieved by using swing-out condensers, the best of which "clunk" into place like a solid car door.
The ones that freely slide from side to side are "kind of a piece of junk in my opinion," he said, "because they get out of focus easily."
When it comes to ocular lenses, pony up for the focusable, highest-quality, wide-angle options available, Dr. Gross said.
Each person viewing the specimen should be able to separately focus the image to accommodate individual differences in visual acuity.
Angle for Tilt Heads
Opting for a system with tilt heads isn’t imperative, but is wise if more than one doctor is sharing the microscope, according to Dr. Gross.
"Unless you’re identical twins, you’re going to [have one doctor who is] taller or shorter, sits up straight or slumped, use[s] different style chairs. If you have tilt heads, they’re really easy to adjust."
You can economize, though, by foregoing an option that allows the microscope heads to push in or out. "That’s a waste of money," he said.
Dr. Gross said he had no financial disclosures with regard to any company that manufactures or maintains microscopes used in Mohs surgery.
EXPERT ANALYSIS FROM A MEETING SPONSORED BY THE AMERICAN SOCIETY FOR MOHS SURGERY
‘Keep it Simple,’ for a Satisfactory ASMS Grade
SAN DIEGO – If you want to receive a coveted "satisfactory" grade from peer reviewers of the American Society for Mohs Surgery, "keep it simple," advised Dr. Sharon F. Tiefenbrunn, a member of the ASMS peer review committee..
"The goal of this is to show us that you can recognize a perfect case and can produce a perfect case," she said at a meeting sponsored by the ASMS.
Highly complex or controversial cases are frequently "unreviewable," she noted.
"We don’t want your greatest case, where you worked until almost midnight, and it took 10 stages to clear, and the patient had to have a free flap to repair the defect, and you were ready to tear out your hair and swear off Mohs forever," stressed Dr. Tiefenbrunn, a procedural dermatologist in private practice in St. Louis.
Instead, peer reviewers want to see a stage II to III Mohs case, with tumor evident on stage I and a tumor-free final stage, she said.
Dr. Tiefenbrunn explained that the peer review program was launched in 2000, reviewing cases from the previous year. Its purpose is to improve the quality of Mohs surgery, track the organization’s success in teaching Mohs techniques, and provide practitioners with one of the two episodes of peer review required by the Clinical Laboratory Improvement Act (CLIA).
Cases are submitted to the ASMS administrative office and cycled to reviewers, who classify them as satisfactory, unreviewable, or "comments only."
A typical comment might be: "not enough overlap to determine clear margin." Such cases are sent to the peer review chairman, who forwards the case, along with a critique, back to the presenter.
In essence, a "comments" case fails to meet peer review standards, while an "unreviewable" case potentially might be corrected according to reviewers’ comments and resubmitted, said Dr. Tiefenbrunn.
Satisfactory cases demonstrate use of standard Mohs technique, have a minimal number of slides, and represent noncontroversial histology.
Some common reasons that cases are judged "unreviewable" include problems with the tumor map and ink legend, failure to mark the tumor on the map, unlabeled sections, confusing slide labeling, use of a nonstandard Mohs technique, bubbles, or a failure to identify tumor on stage I.
Dr. Tiefenbrunn reminded attendees of several cardinal rules, among them the need to include a complete skin edge (at least 90%), a complete deep margin, visible structural details, an adequate stain, visible ink for orientation, and a stage II showing a 2.5-mm margin in every direction.
Reviewers appreciate a legible presenter form; an orderly, precisely drawn map; well-packaged and well-organized slides; and simple explanations for anything on histology, such as actinic keratoses or nevi that might be mistaken for a tumor, said Dr. Tiefenbrunn.
"Presentation matters," she said. "Reviewers are your colleagues who are just as busy as you. We want to whip through these cases and just circle ‘satisfactory’ and go home."
Dr. Tiefenbrunn reported no relevant financial disclosures.
SAN DIEGO – If you want to receive a coveted "satisfactory" grade from peer reviewers of the American Society for Mohs Surgery, "keep it simple," advised Dr. Sharon F. Tiefenbrunn, a member of the ASMS peer review committee..
"The goal of this is to show us that you can recognize a perfect case and can produce a perfect case," she said at a meeting sponsored by the ASMS.
Highly complex or controversial cases are frequently "unreviewable," she noted.
"We don’t want your greatest case, where you worked until almost midnight, and it took 10 stages to clear, and the patient had to have a free flap to repair the defect, and you were ready to tear out your hair and swear off Mohs forever," stressed Dr. Tiefenbrunn, a procedural dermatologist in private practice in St. Louis.
Instead, peer reviewers want to see a stage II to III Mohs case, with tumor evident on stage I and a tumor-free final stage, she said.
Dr. Tiefenbrunn explained that the peer review program was launched in 2000, reviewing cases from the previous year. Its purpose is to improve the quality of Mohs surgery, track the organization’s success in teaching Mohs techniques, and provide practitioners with one of the two episodes of peer review required by the Clinical Laboratory Improvement Act (CLIA).
Cases are submitted to the ASMS administrative office and cycled to reviewers, who classify them as satisfactory, unreviewable, or "comments only."
A typical comment might be: "not enough overlap to determine clear margin." Such cases are sent to the peer review chairman, who forwards the case, along with a critique, back to the presenter.
In essence, a "comments" case fails to meet peer review standards, while an "unreviewable" case potentially might be corrected according to reviewers’ comments and resubmitted, said Dr. Tiefenbrunn.
Satisfactory cases demonstrate use of standard Mohs technique, have a minimal number of slides, and represent noncontroversial histology.
Some common reasons that cases are judged "unreviewable" include problems with the tumor map and ink legend, failure to mark the tumor on the map, unlabeled sections, confusing slide labeling, use of a nonstandard Mohs technique, bubbles, or a failure to identify tumor on stage I.
Dr. Tiefenbrunn reminded attendees of several cardinal rules, among them the need to include a complete skin edge (at least 90%), a complete deep margin, visible structural details, an adequate stain, visible ink for orientation, and a stage II showing a 2.5-mm margin in every direction.
Reviewers appreciate a legible presenter form; an orderly, precisely drawn map; well-packaged and well-organized slides; and simple explanations for anything on histology, such as actinic keratoses or nevi that might be mistaken for a tumor, said Dr. Tiefenbrunn.
"Presentation matters," she said. "Reviewers are your colleagues who are just as busy as you. We want to whip through these cases and just circle ‘satisfactory’ and go home."
Dr. Tiefenbrunn reported no relevant financial disclosures.
SAN DIEGO – If you want to receive a coveted "satisfactory" grade from peer reviewers of the American Society for Mohs Surgery, "keep it simple," advised Dr. Sharon F. Tiefenbrunn, a member of the ASMS peer review committee..
"The goal of this is to show us that you can recognize a perfect case and can produce a perfect case," she said at a meeting sponsored by the ASMS.
Highly complex or controversial cases are frequently "unreviewable," she noted.
"We don’t want your greatest case, where you worked until almost midnight, and it took 10 stages to clear, and the patient had to have a free flap to repair the defect, and you were ready to tear out your hair and swear off Mohs forever," stressed Dr. Tiefenbrunn, a procedural dermatologist in private practice in St. Louis.
Instead, peer reviewers want to see a stage II to III Mohs case, with tumor evident on stage I and a tumor-free final stage, she said.
Dr. Tiefenbrunn explained that the peer review program was launched in 2000, reviewing cases from the previous year. Its purpose is to improve the quality of Mohs surgery, track the organization’s success in teaching Mohs techniques, and provide practitioners with one of the two episodes of peer review required by the Clinical Laboratory Improvement Act (CLIA).
Cases are submitted to the ASMS administrative office and cycled to reviewers, who classify them as satisfactory, unreviewable, or "comments only."
A typical comment might be: "not enough overlap to determine clear margin." Such cases are sent to the peer review chairman, who forwards the case, along with a critique, back to the presenter.
In essence, a "comments" case fails to meet peer review standards, while an "unreviewable" case potentially might be corrected according to reviewers’ comments and resubmitted, said Dr. Tiefenbrunn.
Satisfactory cases demonstrate use of standard Mohs technique, have a minimal number of slides, and represent noncontroversial histology.
Some common reasons that cases are judged "unreviewable" include problems with the tumor map and ink legend, failure to mark the tumor on the map, unlabeled sections, confusing slide labeling, use of a nonstandard Mohs technique, bubbles, or a failure to identify tumor on stage I.
Dr. Tiefenbrunn reminded attendees of several cardinal rules, among them the need to include a complete skin edge (at least 90%), a complete deep margin, visible structural details, an adequate stain, visible ink for orientation, and a stage II showing a 2.5-mm margin in every direction.
Reviewers appreciate a legible presenter form; an orderly, precisely drawn map; well-packaged and well-organized slides; and simple explanations for anything on histology, such as actinic keratoses or nevi that might be mistaken for a tumor, said Dr. Tiefenbrunn.
"Presentation matters," she said. "Reviewers are your colleagues who are just as busy as you. We want to whip through these cases and just circle ‘satisfactory’ and go home."
Dr. Tiefenbrunn reported no relevant financial disclosures.
EXPERT ANALYSIS FROM A MEETING SPONSORED BY THE AMERICAN SOCIETY FOR MOHS SURGERY
Phototherapy in the Age of Biologics
Daniel Walker, BS, and Heidi Jacobe, MD, MSCS
Dermatologists are presented with a diversity of therapeutic modalities for the treatment of inflammatory, sclerosing, and neoplastic conditions, but with the development of various new irradiation devices that utilize specific parts of the electromagnetic spectrum, phototherapy has become a more viable, accessible, and efficacious option in the treatment of these conditions. The ultraviolet (UV) range (10-400 nm) is further subdivided into UVA and UVB, each of which has been particularly useful in a number of skin conditions. The most commonly used forms of UV irradiation are UVA1, psoralen plus UVA (PUVA), and narrowband (NB) UVB. Each of these modalities differ in their mechanism of action, indications, and side effect profiles, and it is important that clinicians be familiar with these differences. Today, phototherapy is a valuable option in the treatment of many nonpsoriatic conditions including atopic dermatitis, sclerosing skin conditions such as morphea, vitiligo, and mycosis fungoides. Due to its relative safety, phototherapy may be used in most populations, including children and pregnant women. However, contraindications and side effects are known and should be considered before patients begin a phototherapeutic regimen.
*For a PDF of the full article, click on the link to the left of this introduction.
Daniel Walker, BS, and Heidi Jacobe, MD, MSCS
Dermatologists are presented with a diversity of therapeutic modalities for the treatment of inflammatory, sclerosing, and neoplastic conditions, but with the development of various new irradiation devices that utilize specific parts of the electromagnetic spectrum, phototherapy has become a more viable, accessible, and efficacious option in the treatment of these conditions. The ultraviolet (UV) range (10-400 nm) is further subdivided into UVA and UVB, each of which has been particularly useful in a number of skin conditions. The most commonly used forms of UV irradiation are UVA1, psoralen plus UVA (PUVA), and narrowband (NB) UVB. Each of these modalities differ in their mechanism of action, indications, and side effect profiles, and it is important that clinicians be familiar with these differences. Today, phototherapy is a valuable option in the treatment of many nonpsoriatic conditions including atopic dermatitis, sclerosing skin conditions such as morphea, vitiligo, and mycosis fungoides. Due to its relative safety, phototherapy may be used in most populations, including children and pregnant women. However, contraindications and side effects are known and should be considered before patients begin a phototherapeutic regimen.
*For a PDF of the full article, click on the link to the left of this introduction.
Daniel Walker, BS, and Heidi Jacobe, MD, MSCS
Dermatologists are presented with a diversity of therapeutic modalities for the treatment of inflammatory, sclerosing, and neoplastic conditions, but with the development of various new irradiation devices that utilize specific parts of the electromagnetic spectrum, phototherapy has become a more viable, accessible, and efficacious option in the treatment of these conditions. The ultraviolet (UV) range (10-400 nm) is further subdivided into UVA and UVB, each of which has been particularly useful in a number of skin conditions. The most commonly used forms of UV irradiation are UVA1, psoralen plus UVA (PUVA), and narrowband (NB) UVB. Each of these modalities differ in their mechanism of action, indications, and side effect profiles, and it is important that clinicians be familiar with these differences. Today, phototherapy is a valuable option in the treatment of many nonpsoriatic conditions including atopic dermatitis, sclerosing skin conditions such as morphea, vitiligo, and mycosis fungoides. Due to its relative safety, phototherapy may be used in most populations, including children and pregnant women. However, contraindications and side effects are known and should be considered before patients begin a phototherapeutic regimen.
*For a PDF of the full article, click on the link to the left of this introduction.
Photodynamic Therapy: Current Evidence and Applications in Dermatology
Yoojin Lee, BA, and Elma D. Baron, MD
Photodynamic therapy (PDT) involves the activation of a photosensitizing drug, which preferentially localizes to diseased skin, by irradiation with light to cause selective cytotoxic damage. Since its discovery in the early 20th century and the development of topical photosensitizers 2 decades ago, PDT is increasingly being used in dermatology for a wide range of neoplastic, inflammatory, and infectious cutaneous conditions. Topical 5-aminolevulinic acid and methyl aminolevulinic acid, the most commonly used agents in PDT, have received Food and Drug Administration approval for the treatment of actinic keratoses, and many second-generation photosensitizers are under investigation. Compared with conventional therapies, PDT has the advantage of being noninvasive and capable of field treatment. It is also associated with quicker recovery periods and excellent cosmetic results. Because of these benefits, PDT is being evaluated as a potential treatment option for many dermatologic conditions and has been shown to be effective for certain nonmelanoma skin cancers. Although research is still limited, PDT might also have a therapeutic benefit for cutaneous T-cell lymphoma, acne, psoriasis, leishmaniasis, and warts, among others. This article is a review of the clinical applications of PDT in dermatology and summarizes the current evidence in literature describing its efficacy, safety, and cosmetic outcome.Photodynamic therapy (PDT) involves the activation of a photosensitizing drug, which preferentially localizes to diseased skin, by irradiation with light to cause selective cytotoxic damage. Since its discovery in the early 20th century and the development of topical photosensitizers 2 decades ago, PDT is increasingly being used in dermatology for a wide range of neoplastic, inflammatory, and infectious cutaneous conditions. Topical 5-aminolevulinic acid and methyl aminolevulinic acid, the most commonly used agents in PDT, have received Food and Drug Administration approval for the treatment of actinic keratoses, and many second-generation photosensitizers are under investigation. Compared with conventional therapies, PDT has the advantage of being noninvasive and capable of field treatment. It is also associated with quicker recovery periods and excellent cosmetic results. Because of these benefits, PDT is being evaluated as a potential treatment option for many dermatologic conditions and has been shown to be effective for certain nonmelanoma skin cancers. Although research is still limited, PDT might also have a therapeutic benefit for cutaneous T-cell lymphoma, acne, psoriasis, leishmaniasis, and warts, among others. This article is a review of the clinical applications of PDT in dermatology and summarizes the current evidence in literature describing its efficacy, safety, and cosmetic outcome.
*For a PDF of the full article, click on the link to the left of this introduction.
Yoojin Lee, BA, and Elma D. Baron, MD
Photodynamic therapy (PDT) involves the activation of a photosensitizing drug, which preferentially localizes to diseased skin, by irradiation with light to cause selective cytotoxic damage. Since its discovery in the early 20th century and the development of topical photosensitizers 2 decades ago, PDT is increasingly being used in dermatology for a wide range of neoplastic, inflammatory, and infectious cutaneous conditions. Topical 5-aminolevulinic acid and methyl aminolevulinic acid, the most commonly used agents in PDT, have received Food and Drug Administration approval for the treatment of actinic keratoses, and many second-generation photosensitizers are under investigation. Compared with conventional therapies, PDT has the advantage of being noninvasive and capable of field treatment. It is also associated with quicker recovery periods and excellent cosmetic results. Because of these benefits, PDT is being evaluated as a potential treatment option for many dermatologic conditions and has been shown to be effective for certain nonmelanoma skin cancers. Although research is still limited, PDT might also have a therapeutic benefit for cutaneous T-cell lymphoma, acne, psoriasis, leishmaniasis, and warts, among others. This article is a review of the clinical applications of PDT in dermatology and summarizes the current evidence in literature describing its efficacy, safety, and cosmetic outcome.Photodynamic therapy (PDT) involves the activation of a photosensitizing drug, which preferentially localizes to diseased skin, by irradiation with light to cause selective cytotoxic damage. Since its discovery in the early 20th century and the development of topical photosensitizers 2 decades ago, PDT is increasingly being used in dermatology for a wide range of neoplastic, inflammatory, and infectious cutaneous conditions. Topical 5-aminolevulinic acid and methyl aminolevulinic acid, the most commonly used agents in PDT, have received Food and Drug Administration approval for the treatment of actinic keratoses, and many second-generation photosensitizers are under investigation. Compared with conventional therapies, PDT has the advantage of being noninvasive and capable of field treatment. It is also associated with quicker recovery periods and excellent cosmetic results. Because of these benefits, PDT is being evaluated as a potential treatment option for many dermatologic conditions and has been shown to be effective for certain nonmelanoma skin cancers. Although research is still limited, PDT might also have a therapeutic benefit for cutaneous T-cell lymphoma, acne, psoriasis, leishmaniasis, and warts, among others. This article is a review of the clinical applications of PDT in dermatology and summarizes the current evidence in literature describing its efficacy, safety, and cosmetic outcome.
*For a PDF of the full article, click on the link to the left of this introduction.
Yoojin Lee, BA, and Elma D. Baron, MD
Photodynamic therapy (PDT) involves the activation of a photosensitizing drug, which preferentially localizes to diseased skin, by irradiation with light to cause selective cytotoxic damage. Since its discovery in the early 20th century and the development of topical photosensitizers 2 decades ago, PDT is increasingly being used in dermatology for a wide range of neoplastic, inflammatory, and infectious cutaneous conditions. Topical 5-aminolevulinic acid and methyl aminolevulinic acid, the most commonly used agents in PDT, have received Food and Drug Administration approval for the treatment of actinic keratoses, and many second-generation photosensitizers are under investigation. Compared with conventional therapies, PDT has the advantage of being noninvasive and capable of field treatment. It is also associated with quicker recovery periods and excellent cosmetic results. Because of these benefits, PDT is being evaluated as a potential treatment option for many dermatologic conditions and has been shown to be effective for certain nonmelanoma skin cancers. Although research is still limited, PDT might also have a therapeutic benefit for cutaneous T-cell lymphoma, acne, psoriasis, leishmaniasis, and warts, among others. This article is a review of the clinical applications of PDT in dermatology and summarizes the current evidence in literature describing its efficacy, safety, and cosmetic outcome.Photodynamic therapy (PDT) involves the activation of a photosensitizing drug, which preferentially localizes to diseased skin, by irradiation with light to cause selective cytotoxic damage. Since its discovery in the early 20th century and the development of topical photosensitizers 2 decades ago, PDT is increasingly being used in dermatology for a wide range of neoplastic, inflammatory, and infectious cutaneous conditions. Topical 5-aminolevulinic acid and methyl aminolevulinic acid, the most commonly used agents in PDT, have received Food and Drug Administration approval for the treatment of actinic keratoses, and many second-generation photosensitizers are under investigation. Compared with conventional therapies, PDT has the advantage of being noninvasive and capable of field treatment. It is also associated with quicker recovery periods and excellent cosmetic results. Because of these benefits, PDT is being evaluated as a potential treatment option for many dermatologic conditions and has been shown to be effective for certain nonmelanoma skin cancers. Although research is still limited, PDT might also have a therapeutic benefit for cutaneous T-cell lymphoma, acne, psoriasis, leishmaniasis, and warts, among others. This article is a review of the clinical applications of PDT in dermatology and summarizes the current evidence in literature describing its efficacy, safety, and cosmetic outcome.
*For a PDF of the full article, click on the link to the left of this introduction.
Photoprotection in the Era of Nanotechnology
Steven Q. Wang, MD, and Ian R. Tooley, PhD
Commercial sunscreen based on nano-sized titanium dioxide (TiO2) and zinc oxide (ZnO) delivers superior UV protection and reduces whitening on skin compared to the older generations of inorganic sunscreens. This review discusses the historical use of nano-sized TiO2 and ZnO in sunscreen and the relationship between UV attenuation and the primary particles, aggregates and agglomerates that make up these inorganic oxides. In addition we reviewed the recent safety concerns surrounding these materials, specifically, percutaneous penetration of TiO2 and ZnO nanoparticles through human skin and their potential to cause phototoxicity.
*For a PDF of the full article, click on the link to the left of this introduction.
Steven Q. Wang, MD, and Ian R. Tooley, PhD
Commercial sunscreen based on nano-sized titanium dioxide (TiO2) and zinc oxide (ZnO) delivers superior UV protection and reduces whitening on skin compared to the older generations of inorganic sunscreens. This review discusses the historical use of nano-sized TiO2 and ZnO in sunscreen and the relationship between UV attenuation and the primary particles, aggregates and agglomerates that make up these inorganic oxides. In addition we reviewed the recent safety concerns surrounding these materials, specifically, percutaneous penetration of TiO2 and ZnO nanoparticles through human skin and their potential to cause phototoxicity.
*For a PDF of the full article, click on the link to the left of this introduction.
Steven Q. Wang, MD, and Ian R. Tooley, PhD
Commercial sunscreen based on nano-sized titanium dioxide (TiO2) and zinc oxide (ZnO) delivers superior UV protection and reduces whitening on skin compared to the older generations of inorganic sunscreens. This review discusses the historical use of nano-sized TiO2 and ZnO in sunscreen and the relationship between UV attenuation and the primary particles, aggregates and agglomerates that make up these inorganic oxides. In addition we reviewed the recent safety concerns surrounding these materials, specifically, percutaneous penetration of TiO2 and ZnO nanoparticles through human skin and their potential to cause phototoxicity.
*For a PDF of the full article, click on the link to the left of this introduction.
Ultraviolet A Radiation: Its Role in Immunosuppression and Carcinogenesis
Gary M. Halliday, PhD, DSc, Scott N. Byrne, PhD, and Diona L. Damian, MBBS, FACD, PhD
Ultraviolet A (UVA) radiation is immunosuppressive and mutagenic in humans and carcinogenic in animals. UVA suppresses immunity with a bell-shaped dose response. At doses equivalent to 15-20 minutes of sun exposure at noon, UVA contributes to approximately 75% of sunlight-induced immunosuppression. A recent action spectrum, indicating that 360-380 nm but not 320-350 nm UVA suppresses immunity in humans, suggests an important role for reactive oxygen species. UVA also causes an energy crisis in cells, and normalization of adenosine triphosphate with nicotinamide prevents UVA immunosuppression. UVA activation of the alternative complement pathway and defects in memory T-cell development are also involved. Human skin cancers contain mutations in the p53 and BRM genes that are consistent with being induced by UVA. UVA is also mutagenic in human skin equivalents. The basal layer of human skin is more susceptible to UVA-induced mutations than the upper layers. Because skin cancers arise from these basal proliferating cells, this finding is likely to be important and could be attributable to low levels of the DNA repair enzyme OGG1 in basal cells. UVA is therefore likely to make a larger contribution to UVA-induced skin carcinogenesis in humans than is predicted by small animal models as the result of being immunosuppressive and mutagenic for basal keratinocytes.
*For a PDF of the full article, click on the link to the left of this introduction.
Gary M. Halliday, PhD, DSc, Scott N. Byrne, PhD, and Diona L. Damian, MBBS, FACD, PhD
Ultraviolet A (UVA) radiation is immunosuppressive and mutagenic in humans and carcinogenic in animals. UVA suppresses immunity with a bell-shaped dose response. At doses equivalent to 15-20 minutes of sun exposure at noon, UVA contributes to approximately 75% of sunlight-induced immunosuppression. A recent action spectrum, indicating that 360-380 nm but not 320-350 nm UVA suppresses immunity in humans, suggests an important role for reactive oxygen species. UVA also causes an energy crisis in cells, and normalization of adenosine triphosphate with nicotinamide prevents UVA immunosuppression. UVA activation of the alternative complement pathway and defects in memory T-cell development are also involved. Human skin cancers contain mutations in the p53 and BRM genes that are consistent with being induced by UVA. UVA is also mutagenic in human skin equivalents. The basal layer of human skin is more susceptible to UVA-induced mutations than the upper layers. Because skin cancers arise from these basal proliferating cells, this finding is likely to be important and could be attributable to low levels of the DNA repair enzyme OGG1 in basal cells. UVA is therefore likely to make a larger contribution to UVA-induced skin carcinogenesis in humans than is predicted by small animal models as the result of being immunosuppressive and mutagenic for basal keratinocytes.
*For a PDF of the full article, click on the link to the left of this introduction.
Gary M. Halliday, PhD, DSc, Scott N. Byrne, PhD, and Diona L. Damian, MBBS, FACD, PhD
Ultraviolet A (UVA) radiation is immunosuppressive and mutagenic in humans and carcinogenic in animals. UVA suppresses immunity with a bell-shaped dose response. At doses equivalent to 15-20 minutes of sun exposure at noon, UVA contributes to approximately 75% of sunlight-induced immunosuppression. A recent action spectrum, indicating that 360-380 nm but not 320-350 nm UVA suppresses immunity in humans, suggests an important role for reactive oxygen species. UVA also causes an energy crisis in cells, and normalization of adenosine triphosphate with nicotinamide prevents UVA immunosuppression. UVA activation of the alternative complement pathway and defects in memory T-cell development are also involved. Human skin cancers contain mutations in the p53 and BRM genes that are consistent with being induced by UVA. UVA is also mutagenic in human skin equivalents. The basal layer of human skin is more susceptible to UVA-induced mutations than the upper layers. Because skin cancers arise from these basal proliferating cells, this finding is likely to be important and could be attributable to low levels of the DNA repair enzyme OGG1 in basal cells. UVA is therefore likely to make a larger contribution to UVA-induced skin carcinogenesis in humans than is predicted by small animal models as the result of being immunosuppressive and mutagenic for basal keratinocytes.
*For a PDF of the full article, click on the link to the left of this introduction.