User login
Prior antibiotic use lowers checkpoint inhibitor response and survival
Prior antibiotic use may be associated with a reduced treatment response to checkpoint inhibitors, and worse outcomes, in patients with cancer, according to investigators.
In a prospective cohort study, researchers followed 196 patients with cancer who were treated with immune checkpoint inhibitors in routine clinical practice.
A total of 22 patients had been treated with a 7-day or less course of broad-spectrum beta-lactam–based antibiotics in the 30 days prior to starting immune checkpoint inhibitor therapy, and 68 patients were concurrently taking broad-spectrum beta-lactam–based antibiotics with their checkpoint inhibitor therapy.
The analysis revealed that prior antibiotic therapy was associated with nearly a 100% greater likelihood of poor response to checkpoint inhibitor therapy (P less than .001) and significantly worse overall survival (2 vs. 26 months). Patients who had been on prior antibiotic therapy were also more likely to stop checkpoint inhibitor therapy because their disease had progressed, and were more likely to die of progressive disease while on checkpoint inhibitors.
However, concurrent antibiotic use did not appear to affect either treatment response to checkpoint inhibitors or overall survival.
The most common indication for both prior and concurrent antibiotic use was respiratory tract infections. Researchers examined whether cancer type might play a role in contributing to the association; for example, chronic airway disease in lung cancer might mean higher likelihood of antibiotic use but also lower treatment response and survival.
They found that the association between prior antibiotic therapy and overall survival was consistent across the 119 patients with non–small cell lung cancer, the 38 patients with melanoma, and the 39 patients with other tumor types.
The association was also independent of the class of antibiotic used, the patient’s performance status, and their corticosteroid use.
“Broad-spectrum ATB [antibiotic] use can cause prolonged disruption of the gut ecosystem and impair the effectiveness of the cytotoxic T-cell response against cancer, strengthening the biologic plausibility underlying the adverse effect of ATB therapy on immunotherapy outcomes,” wrote Dr. David J. Pinato, from Imperial College London, and coauthors in JAMA Oncology.
Addressing the question of whether comorbidities might be the mediating factor, the authors pointed out that the use of antibiotics during checkpoint inhibitor therapy – which was a potential indicator of patients’ status worsening during treatment – was not associated with reduced response to treatment or lower overall survival.
“Although provision of cATB [concurrent antibiotic] therapy appears to be safe in the context of immunotherapy, clinicians should carefully weigh the pros and cons of prescribing broad-spectrum ATBs prior to ICI [immune checkpoint inhibitor] treatment,” they wrote.
The study was supported by the Imperial College National Institute for Health Research Biomedical Research Centre, the Imperial College Tissue Bank, the Imperial Cancer Research U.K. Centre, the National Institute for Health Research, and the Wellcome Trust Strategic Fund. Two authors reported receiving grant funding and personal fees from the pharmaceutical sector unrelated to the study.
SOURCE: Pinato D et al. JAMA Oncol. 2019 Sep 12. doi: 10.1001/jamaoncol.2019.2785.
Prior antibiotic use may be associated with a reduced treatment response to checkpoint inhibitors, and worse outcomes, in patients with cancer, according to investigators.
In a prospective cohort study, researchers followed 196 patients with cancer who were treated with immune checkpoint inhibitors in routine clinical practice.
A total of 22 patients had been treated with a 7-day or less course of broad-spectrum beta-lactam–based antibiotics in the 30 days prior to starting immune checkpoint inhibitor therapy, and 68 patients were concurrently taking broad-spectrum beta-lactam–based antibiotics with their checkpoint inhibitor therapy.
The analysis revealed that prior antibiotic therapy was associated with nearly a 100% greater likelihood of poor response to checkpoint inhibitor therapy (P less than .001) and significantly worse overall survival (2 vs. 26 months). Patients who had been on prior antibiotic therapy were also more likely to stop checkpoint inhibitor therapy because their disease had progressed, and were more likely to die of progressive disease while on checkpoint inhibitors.
However, concurrent antibiotic use did not appear to affect either treatment response to checkpoint inhibitors or overall survival.
The most common indication for both prior and concurrent antibiotic use was respiratory tract infections. Researchers examined whether cancer type might play a role in contributing to the association; for example, chronic airway disease in lung cancer might mean higher likelihood of antibiotic use but also lower treatment response and survival.
They found that the association between prior antibiotic therapy and overall survival was consistent across the 119 patients with non–small cell lung cancer, the 38 patients with melanoma, and the 39 patients with other tumor types.
The association was also independent of the class of antibiotic used, the patient’s performance status, and their corticosteroid use.
“Broad-spectrum ATB [antibiotic] use can cause prolonged disruption of the gut ecosystem and impair the effectiveness of the cytotoxic T-cell response against cancer, strengthening the biologic plausibility underlying the adverse effect of ATB therapy on immunotherapy outcomes,” wrote Dr. David J. Pinato, from Imperial College London, and coauthors in JAMA Oncology.
Addressing the question of whether comorbidities might be the mediating factor, the authors pointed out that the use of antibiotics during checkpoint inhibitor therapy – which was a potential indicator of patients’ status worsening during treatment – was not associated with reduced response to treatment or lower overall survival.
“Although provision of cATB [concurrent antibiotic] therapy appears to be safe in the context of immunotherapy, clinicians should carefully weigh the pros and cons of prescribing broad-spectrum ATBs prior to ICI [immune checkpoint inhibitor] treatment,” they wrote.
The study was supported by the Imperial College National Institute for Health Research Biomedical Research Centre, the Imperial College Tissue Bank, the Imperial Cancer Research U.K. Centre, the National Institute for Health Research, and the Wellcome Trust Strategic Fund. Two authors reported receiving grant funding and personal fees from the pharmaceutical sector unrelated to the study.
SOURCE: Pinato D et al. JAMA Oncol. 2019 Sep 12. doi: 10.1001/jamaoncol.2019.2785.
Prior antibiotic use may be associated with a reduced treatment response to checkpoint inhibitors, and worse outcomes, in patients with cancer, according to investigators.
In a prospective cohort study, researchers followed 196 patients with cancer who were treated with immune checkpoint inhibitors in routine clinical practice.
A total of 22 patients had been treated with a 7-day or less course of broad-spectrum beta-lactam–based antibiotics in the 30 days prior to starting immune checkpoint inhibitor therapy, and 68 patients were concurrently taking broad-spectrum beta-lactam–based antibiotics with their checkpoint inhibitor therapy.
The analysis revealed that prior antibiotic therapy was associated with nearly a 100% greater likelihood of poor response to checkpoint inhibitor therapy (P less than .001) and significantly worse overall survival (2 vs. 26 months). Patients who had been on prior antibiotic therapy were also more likely to stop checkpoint inhibitor therapy because their disease had progressed, and were more likely to die of progressive disease while on checkpoint inhibitors.
However, concurrent antibiotic use did not appear to affect either treatment response to checkpoint inhibitors or overall survival.
The most common indication for both prior and concurrent antibiotic use was respiratory tract infections. Researchers examined whether cancer type might play a role in contributing to the association; for example, chronic airway disease in lung cancer might mean higher likelihood of antibiotic use but also lower treatment response and survival.
They found that the association between prior antibiotic therapy and overall survival was consistent across the 119 patients with non–small cell lung cancer, the 38 patients with melanoma, and the 39 patients with other tumor types.
The association was also independent of the class of antibiotic used, the patient’s performance status, and their corticosteroid use.
“Broad-spectrum ATB [antibiotic] use can cause prolonged disruption of the gut ecosystem and impair the effectiveness of the cytotoxic T-cell response against cancer, strengthening the biologic plausibility underlying the adverse effect of ATB therapy on immunotherapy outcomes,” wrote Dr. David J. Pinato, from Imperial College London, and coauthors in JAMA Oncology.
Addressing the question of whether comorbidities might be the mediating factor, the authors pointed out that the use of antibiotics during checkpoint inhibitor therapy – which was a potential indicator of patients’ status worsening during treatment – was not associated with reduced response to treatment or lower overall survival.
“Although provision of cATB [concurrent antibiotic] therapy appears to be safe in the context of immunotherapy, clinicians should carefully weigh the pros and cons of prescribing broad-spectrum ATBs prior to ICI [immune checkpoint inhibitor] treatment,” they wrote.
The study was supported by the Imperial College National Institute for Health Research Biomedical Research Centre, the Imperial College Tissue Bank, the Imperial Cancer Research U.K. Centre, the National Institute for Health Research, and the Wellcome Trust Strategic Fund. Two authors reported receiving grant funding and personal fees from the pharmaceutical sector unrelated to the study.
SOURCE: Pinato D et al. JAMA Oncol. 2019 Sep 12. doi: 10.1001/jamaoncol.2019.2785.
FROM JAMA ONCOLOGY
Key clinical point: People who take antibiotics prior to checkpoint inhibitor therapy have lower treatment response and overall survival.
Major finding: Prior antibiotic use is associated with a nearly a 100% greater likelihood of poor response to checkpoint inhibitor therapy.
Study details: A prospective cohort study involving 196 patients receiving checkpoint inhibitor therapy for cancer.
Disclosures: The study was supported by the Imperial College National Institute for Health Research Biomedical Research Centre, the Imperial College Tissue Bank, the Imperial Cancer Research U.K. Centre, the National Institute for Health Research, and the Wellcome Trust Strategic Fund. Two authors reported receiving grant funding and personal fees from the pharmaceutical sector unrelated to the study.
Source: Pinato D et al. JAMA Oncol. 2019 Sep 12. doi: 10.1001/jamaoncol.2019.2785.
Sniffing Out Malignant Melanoma: A Case of Canine Olfactory Detection
To the Editor:
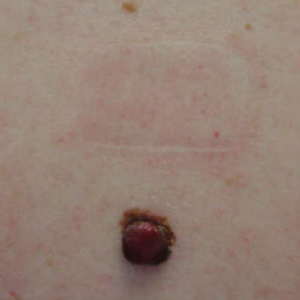
A 43-year-old woman presented with a mole on the central back that had been present since childhood and had changed and grown over the last few years. The patient reported that her 2-year-old rescue dog frequently sniffed the mole and would subsequently get agitated and try to scratch and bite the lesion. This behavior prompted the patient to visit a dermatologist.
She reported no personal history of melanoma or nonmelanoma skin cancer, tanning booth exposure, blistering sunburns, or use of immunosuppressant medications. Her family history was remarkable for basal cell carcinoma in her father but no family history of melanoma. Physical examination revealed a 1.2×1.5-cm brown patch along with a 1×1-cm ulcerated nodule on the lower aspect of the lesion (Figure 1). Dermoscopy showed a blue-white veil and an irregular vascular pattern (Figure 2). No cervical, axillary, or inguinal lymphadenopathy was appreciated on physical examination. Reflectance confocal microscopy showed pagetoid spread of atypical round melanocytes as well as melanocytes in the stratum corneum (Figure 3).
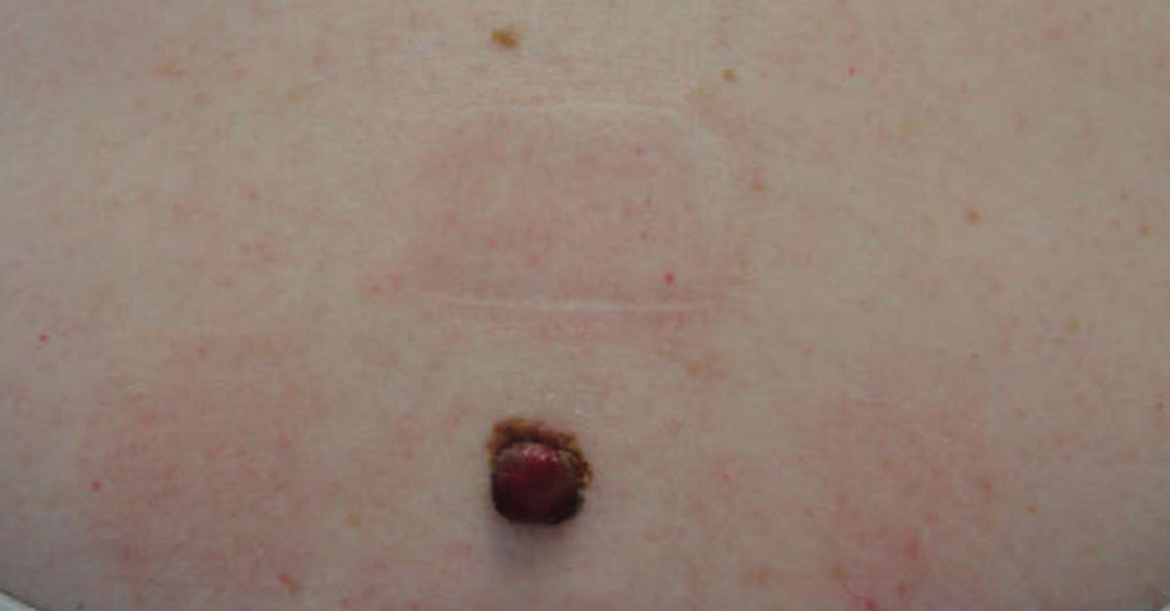
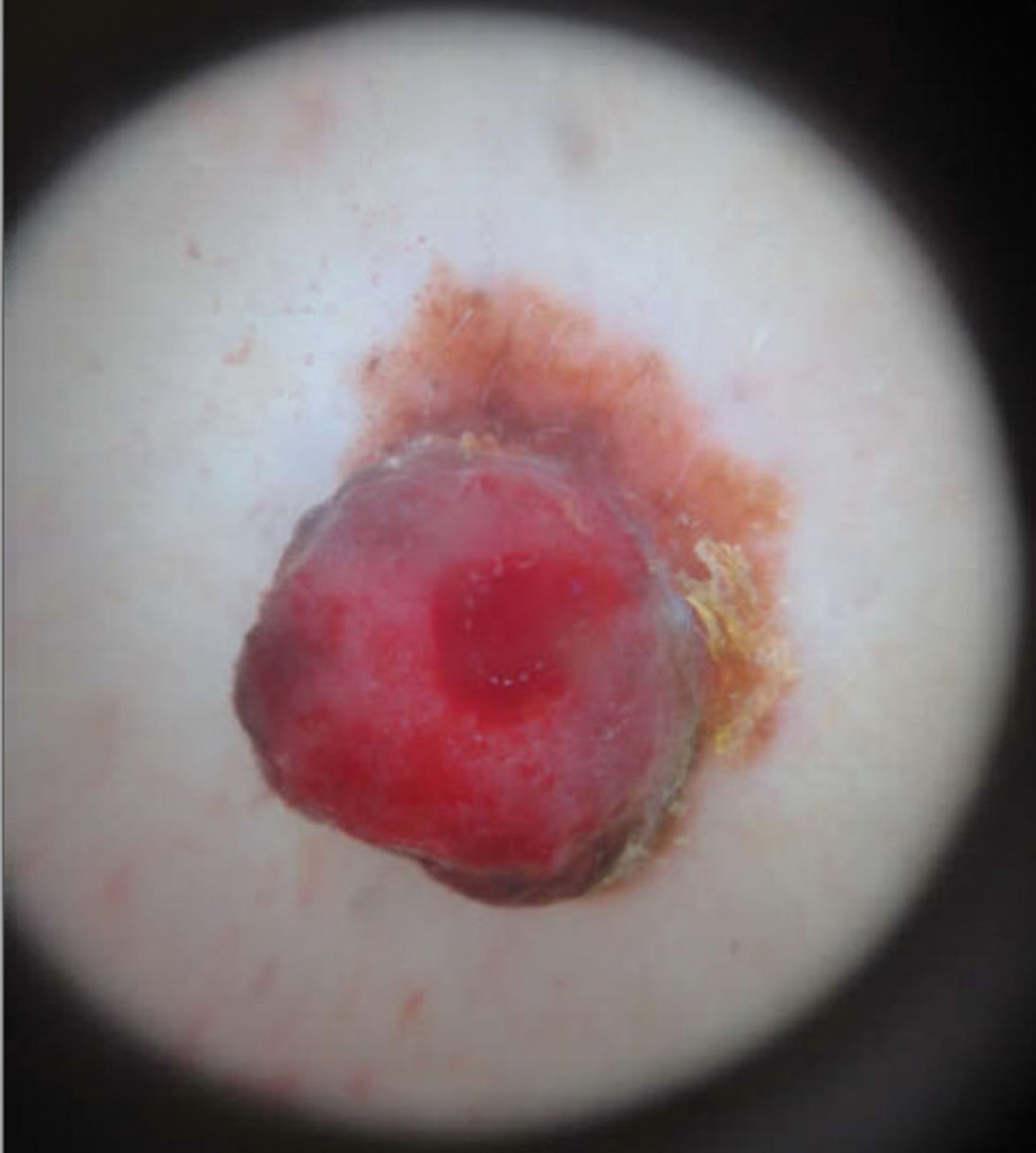

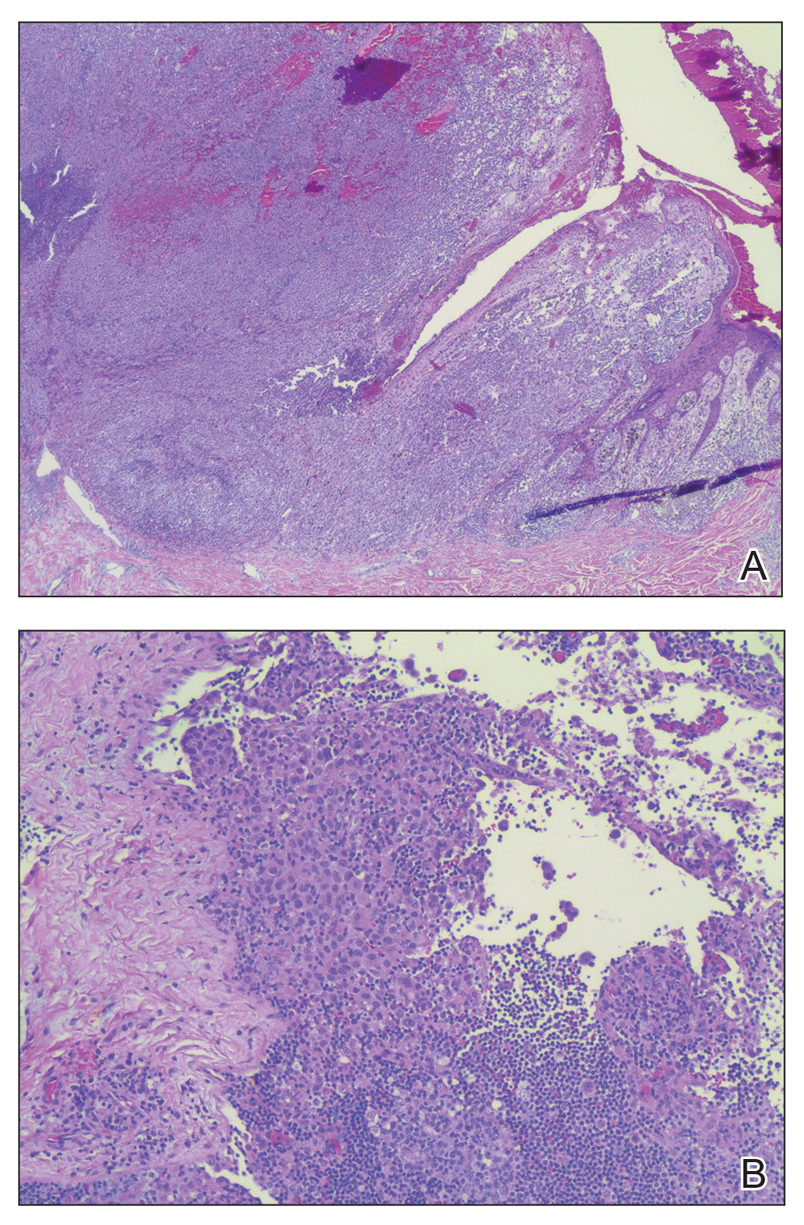
The patient was referred to a surgical oncologist for wide local excision and sentinel lymph node biopsy. Pathology showed a 4-mm-thick melanoma with numerous positive lymph nodes (Figure 4). The patient subsequently underwent a right axillary lymphadenectomy and was diagnosed with stage IIIB malignant melanoma. After surgery, the patient reported that her dog would now sniff her back and calmly rest his head in her lap.
She was treated with ipilimumab but subsequently developed panhypopituitarism, so she was taken off the ipilimumab. Currently, the patient is doing well. She follows up annually for full-body skin examinations and has not had any recurrence in the last 7 years. The patient credits her dog for prompting her to see a dermatologist and saving her life.
Both anecdotal and systematic evidence have emerged on the role of canine olfaction in the detection of lung, breast, colorectal, ovarian, prostate, and skin cancers, including malignant melanoma.1-6 A 1989 case report described a woman who was prompted to seek dermatologic evaluation of a pigmented lesion because her dog consistently targeted the lesion. Excision and subsequent histopathologic examination of the lesion revealed that it was malignant melanoma.5 Another case report described a patient whose dog, which was not trained to detect cancers in humans, persistently licked a lesion behind the patient’s ear that eventually was found to be malignant melanoma.6 These reports have inspired considerable research interest regarding canine olfaction as a potential method to noninvasively screen for and even diagnose malignant melanomas in humans.
Both physiologic and pathologic metabolic processes result in the production of volatile organic compounds (VOCs), or small odorant molecules that evaporate at normal temperatures and pressures.1 Individual cells release VOCs in extremely low concentrations into the blood, urine, feces, and breath, as well as onto the skin’s surface, but there are methods for detecting these VOCs, including gas chromatography–mass spectrometry and canine olfaction.7,8 Pathologic processes, such as infection and malignancy, result in irregular protein synthesis and metabolism, producing new VOCs or differing concentrations of VOCs as compared to normal processes.1
Dimethyl disulfide and dimethyl trisulfide compounds have been identified in malignant melanoma, and these compounds are not produced by normal melanocytes.7 Furthermore, malignant melanoma produces differing quantities of these compounds as compared to normal melanocytes, including isovaleric acid, 2-methylbutyric acid, isoamyl alcohol (3-methyl-1-butanol), and 2-methyl-1-butanol, resulting in a distinct odorant profile that previously has been detected via canine olfaction.7 Canine olfaction can identify odorant molecules at up to 1 part per trillion (a magnitude more sensitive than the currently available gas chromatography–mass spectrometry technologies) and can detect the production of new VOCs or altered VOC ratios due to pathologic processes.1 Systematic studies with dogs that are trained to detect cancers in humans have shown that canine olfaction correctly identified malignant melanomas against healthy skin, benign nevi, and even basal cell carcinomas at higher rates than what would have been expected by chance alone.2,3
Canine olfaction can identify new or altered ratios of odorant VOCs associated with pathologic metabolic processes, and canines can be trained to target odor profiles associated with specific diseases.1 Canine olfaction for melanoma screening and diagnosis may seem appealing, as it provides an easily transportable, real-time, low-cost method compared to other techniques such as gas chromatography–mass spectrometry.1 Although preliminary results have shown that canine olfaction detects melanoma at higher rates than would be expected by chance alone, these findings have not approached clinical utility for the widespread use of canine olfaction as a screening method for melanoma.2,3,9 Further studies are needed to understand the role of canine olfaction in melanoma screening and diagnosis as well as to explore methods to optimize sensitivity and specificity. Until then, patients and dermatologists should not ignore the behavior of dogs toward skin lesions. Dogs may be beneficial in the detection of melanoma and help save lives, as was seen in our case.
- Angle C, Waggoner LP, Ferrando A, et al. Canine detection of the volatilome: a review of implications for pathogen and disease detection. Front Vet Sci. 2016;3:47.
- Pickel D, Mauncy GP, Walker DB, et al. Evidence for canine olfactory detection of melanoma. Applied Animal Behaviour Science. 2004;89:107-116.
- Willis CM, Britton LE, Swindells MA, et al. Invasive melanoma in vivo can be distinguished from basal cell carcinoma, benign naevi and healthy skin by canine olfaction: a proof‐of‐principle study of differential volatile organic compound emission. Br J Dermatol. 2016;175:1020-1029.
- Jezierski T, Walczak M, Ligor T, et al. Study of the art: canine olfaction used for cancer detection on the basis of breath odour. perspectives and limitations. J Breath Res. 2015;9:027001.
- Williams H, Pembroke A. Sniffer dogs in the melanoma clinic? Lancet. 1989;1:734.
- Campbell LF, Farmery L, George SM, et al. Canine olfactory detection of malignant melanoma. BMJ Case Rep. 2013. doi:10.1136/bcr-2013-008566.
- Kwak J, Gallagher M, Ozdener MH, et al. Volatile biomarkers from human melanoma cells. J Chromotogr B Analyt Technol Biomed Life Sci. 2013;931:90-96.
- D’Amico A, Bono R, Pennazza G, et al. Identification of melanoma with a gas sensor array. Skin Res Technol. 2008;14:226-236.
- Elliker KR, Williams HC. Detection of skin cancer odours using dogs: a step forward in melanoma detection training and research methodologies. Br J Dermatol. 2016;175:851-852.
To the Editor:
A 43-year-old woman presented with a mole on the central back that had been present since childhood and had changed and grown over the last few years. The patient reported that her 2-year-old rescue dog frequently sniffed the mole and would subsequently get agitated and try to scratch and bite the lesion. This behavior prompted the patient to visit a dermatologist.
She reported no personal history of melanoma or nonmelanoma skin cancer, tanning booth exposure, blistering sunburns, or use of immunosuppressant medications. Her family history was remarkable for basal cell carcinoma in her father but no family history of melanoma. Physical examination revealed a 1.2×1.5-cm brown patch along with a 1×1-cm ulcerated nodule on the lower aspect of the lesion (Figure 1). Dermoscopy showed a blue-white veil and an irregular vascular pattern (Figure 2). No cervical, axillary, or inguinal lymphadenopathy was appreciated on physical examination. Reflectance confocal microscopy showed pagetoid spread of atypical round melanocytes as well as melanocytes in the stratum corneum (Figure 3).



The patient was referred to a surgical oncologist for wide local excision and sentinel lymph node biopsy. Pathology showed a 4-mm-thick melanoma with numerous positive lymph nodes (Figure 4). The patient subsequently underwent a right axillary lymphadenectomy and was diagnosed with stage IIIB malignant melanoma. After surgery, the patient reported that her dog would now sniff her back and calmly rest his head in her lap.

She was treated with ipilimumab but subsequently developed panhypopituitarism, so she was taken off the ipilimumab. Currently, the patient is doing well. She follows up annually for full-body skin examinations and has not had any recurrence in the last 7 years. The patient credits her dog for prompting her to see a dermatologist and saving her life.
Both anecdotal and systematic evidence have emerged on the role of canine olfaction in the detection of lung, breast, colorectal, ovarian, prostate, and skin cancers, including malignant melanoma.1-6 A 1989 case report described a woman who was prompted to seek dermatologic evaluation of a pigmented lesion because her dog consistently targeted the lesion. Excision and subsequent histopathologic examination of the lesion revealed that it was malignant melanoma.5 Another case report described a patient whose dog, which was not trained to detect cancers in humans, persistently licked a lesion behind the patient’s ear that eventually was found to be malignant melanoma.6 These reports have inspired considerable research interest regarding canine olfaction as a potential method to noninvasively screen for and even diagnose malignant melanomas in humans.
Both physiologic and pathologic metabolic processes result in the production of volatile organic compounds (VOCs), or small odorant molecules that evaporate at normal temperatures and pressures.1 Individual cells release VOCs in extremely low concentrations into the blood, urine, feces, and breath, as well as onto the skin’s surface, but there are methods for detecting these VOCs, including gas chromatography–mass spectrometry and canine olfaction.7,8 Pathologic processes, such as infection and malignancy, result in irregular protein synthesis and metabolism, producing new VOCs or differing concentrations of VOCs as compared to normal processes.1
Dimethyl disulfide and dimethyl trisulfide compounds have been identified in malignant melanoma, and these compounds are not produced by normal melanocytes.7 Furthermore, malignant melanoma produces differing quantities of these compounds as compared to normal melanocytes, including isovaleric acid, 2-methylbutyric acid, isoamyl alcohol (3-methyl-1-butanol), and 2-methyl-1-butanol, resulting in a distinct odorant profile that previously has been detected via canine olfaction.7 Canine olfaction can identify odorant molecules at up to 1 part per trillion (a magnitude more sensitive than the currently available gas chromatography–mass spectrometry technologies) and can detect the production of new VOCs or altered VOC ratios due to pathologic processes.1 Systematic studies with dogs that are trained to detect cancers in humans have shown that canine olfaction correctly identified malignant melanomas against healthy skin, benign nevi, and even basal cell carcinomas at higher rates than what would have been expected by chance alone.2,3
Canine olfaction can identify new or altered ratios of odorant VOCs associated with pathologic metabolic processes, and canines can be trained to target odor profiles associated with specific diseases.1 Canine olfaction for melanoma screening and diagnosis may seem appealing, as it provides an easily transportable, real-time, low-cost method compared to other techniques such as gas chromatography–mass spectrometry.1 Although preliminary results have shown that canine olfaction detects melanoma at higher rates than would be expected by chance alone, these findings have not approached clinical utility for the widespread use of canine olfaction as a screening method for melanoma.2,3,9 Further studies are needed to understand the role of canine olfaction in melanoma screening and diagnosis as well as to explore methods to optimize sensitivity and specificity. Until then, patients and dermatologists should not ignore the behavior of dogs toward skin lesions. Dogs may be beneficial in the detection of melanoma and help save lives, as was seen in our case.
To the Editor:
A 43-year-old woman presented with a mole on the central back that had been present since childhood and had changed and grown over the last few years. The patient reported that her 2-year-old rescue dog frequently sniffed the mole and would subsequently get agitated and try to scratch and bite the lesion. This behavior prompted the patient to visit a dermatologist.
She reported no personal history of melanoma or nonmelanoma skin cancer, tanning booth exposure, blistering sunburns, or use of immunosuppressant medications. Her family history was remarkable for basal cell carcinoma in her father but no family history of melanoma. Physical examination revealed a 1.2×1.5-cm brown patch along with a 1×1-cm ulcerated nodule on the lower aspect of the lesion (Figure 1). Dermoscopy showed a blue-white veil and an irregular vascular pattern (Figure 2). No cervical, axillary, or inguinal lymphadenopathy was appreciated on physical examination. Reflectance confocal microscopy showed pagetoid spread of atypical round melanocytes as well as melanocytes in the stratum corneum (Figure 3).



The patient was referred to a surgical oncologist for wide local excision and sentinel lymph node biopsy. Pathology showed a 4-mm-thick melanoma with numerous positive lymph nodes (Figure 4). The patient subsequently underwent a right axillary lymphadenectomy and was diagnosed with stage IIIB malignant melanoma. After surgery, the patient reported that her dog would now sniff her back and calmly rest his head in her lap.

She was treated with ipilimumab but subsequently developed panhypopituitarism, so she was taken off the ipilimumab. Currently, the patient is doing well. She follows up annually for full-body skin examinations and has not had any recurrence in the last 7 years. The patient credits her dog for prompting her to see a dermatologist and saving her life.
Both anecdotal and systematic evidence have emerged on the role of canine olfaction in the detection of lung, breast, colorectal, ovarian, prostate, and skin cancers, including malignant melanoma.1-6 A 1989 case report described a woman who was prompted to seek dermatologic evaluation of a pigmented lesion because her dog consistently targeted the lesion. Excision and subsequent histopathologic examination of the lesion revealed that it was malignant melanoma.5 Another case report described a patient whose dog, which was not trained to detect cancers in humans, persistently licked a lesion behind the patient’s ear that eventually was found to be malignant melanoma.6 These reports have inspired considerable research interest regarding canine olfaction as a potential method to noninvasively screen for and even diagnose malignant melanomas in humans.
Both physiologic and pathologic metabolic processes result in the production of volatile organic compounds (VOCs), or small odorant molecules that evaporate at normal temperatures and pressures.1 Individual cells release VOCs in extremely low concentrations into the blood, urine, feces, and breath, as well as onto the skin’s surface, but there are methods for detecting these VOCs, including gas chromatography–mass spectrometry and canine olfaction.7,8 Pathologic processes, such as infection and malignancy, result in irregular protein synthesis and metabolism, producing new VOCs or differing concentrations of VOCs as compared to normal processes.1
Dimethyl disulfide and dimethyl trisulfide compounds have been identified in malignant melanoma, and these compounds are not produced by normal melanocytes.7 Furthermore, malignant melanoma produces differing quantities of these compounds as compared to normal melanocytes, including isovaleric acid, 2-methylbutyric acid, isoamyl alcohol (3-methyl-1-butanol), and 2-methyl-1-butanol, resulting in a distinct odorant profile that previously has been detected via canine olfaction.7 Canine olfaction can identify odorant molecules at up to 1 part per trillion (a magnitude more sensitive than the currently available gas chromatography–mass spectrometry technologies) and can detect the production of new VOCs or altered VOC ratios due to pathologic processes.1 Systematic studies with dogs that are trained to detect cancers in humans have shown that canine olfaction correctly identified malignant melanomas against healthy skin, benign nevi, and even basal cell carcinomas at higher rates than what would have been expected by chance alone.2,3
Canine olfaction can identify new or altered ratios of odorant VOCs associated with pathologic metabolic processes, and canines can be trained to target odor profiles associated with specific diseases.1 Canine olfaction for melanoma screening and diagnosis may seem appealing, as it provides an easily transportable, real-time, low-cost method compared to other techniques such as gas chromatography–mass spectrometry.1 Although preliminary results have shown that canine olfaction detects melanoma at higher rates than would be expected by chance alone, these findings have not approached clinical utility for the widespread use of canine olfaction as a screening method for melanoma.2,3,9 Further studies are needed to understand the role of canine olfaction in melanoma screening and diagnosis as well as to explore methods to optimize sensitivity and specificity. Until then, patients and dermatologists should not ignore the behavior of dogs toward skin lesions. Dogs may be beneficial in the detection of melanoma and help save lives, as was seen in our case.
- Angle C, Waggoner LP, Ferrando A, et al. Canine detection of the volatilome: a review of implications for pathogen and disease detection. Front Vet Sci. 2016;3:47.
- Pickel D, Mauncy GP, Walker DB, et al. Evidence for canine olfactory detection of melanoma. Applied Animal Behaviour Science. 2004;89:107-116.
- Willis CM, Britton LE, Swindells MA, et al. Invasive melanoma in vivo can be distinguished from basal cell carcinoma, benign naevi and healthy skin by canine olfaction: a proof‐of‐principle study of differential volatile organic compound emission. Br J Dermatol. 2016;175:1020-1029.
- Jezierski T, Walczak M, Ligor T, et al. Study of the art: canine olfaction used for cancer detection on the basis of breath odour. perspectives and limitations. J Breath Res. 2015;9:027001.
- Williams H, Pembroke A. Sniffer dogs in the melanoma clinic? Lancet. 1989;1:734.
- Campbell LF, Farmery L, George SM, et al. Canine olfactory detection of malignant melanoma. BMJ Case Rep. 2013. doi:10.1136/bcr-2013-008566.
- Kwak J, Gallagher M, Ozdener MH, et al. Volatile biomarkers from human melanoma cells. J Chromotogr B Analyt Technol Biomed Life Sci. 2013;931:90-96.
- D’Amico A, Bono R, Pennazza G, et al. Identification of melanoma with a gas sensor array. Skin Res Technol. 2008;14:226-236.
- Elliker KR, Williams HC. Detection of skin cancer odours using dogs: a step forward in melanoma detection training and research methodologies. Br J Dermatol. 2016;175:851-852.
- Angle C, Waggoner LP, Ferrando A, et al. Canine detection of the volatilome: a review of implications for pathogen and disease detection. Front Vet Sci. 2016;3:47.
- Pickel D, Mauncy GP, Walker DB, et al. Evidence for canine olfactory detection of melanoma. Applied Animal Behaviour Science. 2004;89:107-116.
- Willis CM, Britton LE, Swindells MA, et al. Invasive melanoma in vivo can be distinguished from basal cell carcinoma, benign naevi and healthy skin by canine olfaction: a proof‐of‐principle study of differential volatile organic compound emission. Br J Dermatol. 2016;175:1020-1029.
- Jezierski T, Walczak M, Ligor T, et al. Study of the art: canine olfaction used for cancer detection on the basis of breath odour. perspectives and limitations. J Breath Res. 2015;9:027001.
- Williams H, Pembroke A. Sniffer dogs in the melanoma clinic? Lancet. 1989;1:734.
- Campbell LF, Farmery L, George SM, et al. Canine olfactory detection of malignant melanoma. BMJ Case Rep. 2013. doi:10.1136/bcr-2013-008566.
- Kwak J, Gallagher M, Ozdener MH, et al. Volatile biomarkers from human melanoma cells. J Chromotogr B Analyt Technol Biomed Life Sci. 2013;931:90-96.
- D’Amico A, Bono R, Pennazza G, et al. Identification of melanoma with a gas sensor array. Skin Res Technol. 2008;14:226-236.
- Elliker KR, Williams HC. Detection of skin cancer odours using dogs: a step forward in melanoma detection training and research methodologies. Br J Dermatol. 2016;175:851-852.
Practice Points
- Physiologic and pathologic processes produce volatile organic compounds in the skin and other tissues.
- Malignant melanocytes release unique volatile organic compounds (VOCs) as well as differing combinations and quantities of VOCs as compared to normal melanocytes.
- Volatile organic compounds released at the skin’s surface can be detected by various methods, including canine olfaction; therefore, unusual canine behavior toward skin lesions should not be ignored.
Characterization of Adverse Reactions to ‘4-week’ Nivolumab Dosing
Background: Nivolumab was recently approved for a new flat-dose schedule 480 mg IV every 4 weeks (“480 Q4w”) using data from pharmacokinetics simulations without being first tested directly in humans. We noted several unusual adverse drug reactions (ADRs) using the new dosing and hypothesized that this new dose schedule might generate more ADRs than prior dosing schedules.
Methods: This study attempts to summarize and characterize the types of ADRs seen on the new 480 Q4w dosing. We conducted a retrospective, descriptive chart review and case series including patients at the San Antonio VA Hematology/Oncology clinic treated with at least one dose of Nivolumab 480 mg between 2/1/18 and 10/1/18. We tracked whether these patients developed ADRs, and if so, the highest CTCAE 4.03 grade of reaction, the number of treatments before the reaction developed, and whether the reaction influenced treatment (hold treatment, stop treatment, dose change).
Results: 18 patients matched this criterion (all male, average age 67.6 years). 6 patients experienced an ADR during treatment with the 480 Q4w dose. Grade 1 toxicities included pruritis, abdominal pain, skin rash, fatigue, fever, cramping, myalgia, and diarrhea. There was a Grade 3 case of encephalopathy and a Grade 2 case of diplopia. Of the 6 patients who experienced an adverse drug reaction, 2 (with only Grade 1 toxicities) continued treatment at their same dose frequency; the others changed to 240 mg Q2w. All 4 patients who experienced an ADR and had their dose changed to 240 mg Q2w experienced resolution or improvement in their symptoms except for 1 patient’s complaint of abdominal pain.
Conclusion: 480 Q4w dosing of Nivolumab may have a different ADR profile from prior dose regimens; further quantitative analysis will be required to answer this question. Dose frequency change may present an opportunity to relieve toxicities while allowing patients to continue treatment.
Background: Nivolumab was recently approved for a new flat-dose schedule 480 mg IV every 4 weeks (“480 Q4w”) using data from pharmacokinetics simulations without being first tested directly in humans. We noted several unusual adverse drug reactions (ADRs) using the new dosing and hypothesized that this new dose schedule might generate more ADRs than prior dosing schedules.
Methods: This study attempts to summarize and characterize the types of ADRs seen on the new 480 Q4w dosing. We conducted a retrospective, descriptive chart review and case series including patients at the San Antonio VA Hematology/Oncology clinic treated with at least one dose of Nivolumab 480 mg between 2/1/18 and 10/1/18. We tracked whether these patients developed ADRs, and if so, the highest CTCAE 4.03 grade of reaction, the number of treatments before the reaction developed, and whether the reaction influenced treatment (hold treatment, stop treatment, dose change).
Results: 18 patients matched this criterion (all male, average age 67.6 years). 6 patients experienced an ADR during treatment with the 480 Q4w dose. Grade 1 toxicities included pruritis, abdominal pain, skin rash, fatigue, fever, cramping, myalgia, and diarrhea. There was a Grade 3 case of encephalopathy and a Grade 2 case of diplopia. Of the 6 patients who experienced an adverse drug reaction, 2 (with only Grade 1 toxicities) continued treatment at their same dose frequency; the others changed to 240 mg Q2w. All 4 patients who experienced an ADR and had their dose changed to 240 mg Q2w experienced resolution or improvement in their symptoms except for 1 patient’s complaint of abdominal pain.
Conclusion: 480 Q4w dosing of Nivolumab may have a different ADR profile from prior dose regimens; further quantitative analysis will be required to answer this question. Dose frequency change may present an opportunity to relieve toxicities while allowing patients to continue treatment.
Background: Nivolumab was recently approved for a new flat-dose schedule 480 mg IV every 4 weeks (“480 Q4w”) using data from pharmacokinetics simulations without being first tested directly in humans. We noted several unusual adverse drug reactions (ADRs) using the new dosing and hypothesized that this new dose schedule might generate more ADRs than prior dosing schedules.
Methods: This study attempts to summarize and characterize the types of ADRs seen on the new 480 Q4w dosing. We conducted a retrospective, descriptive chart review and case series including patients at the San Antonio VA Hematology/Oncology clinic treated with at least one dose of Nivolumab 480 mg between 2/1/18 and 10/1/18. We tracked whether these patients developed ADRs, and if so, the highest CTCAE 4.03 grade of reaction, the number of treatments before the reaction developed, and whether the reaction influenced treatment (hold treatment, stop treatment, dose change).
Results: 18 patients matched this criterion (all male, average age 67.6 years). 6 patients experienced an ADR during treatment with the 480 Q4w dose. Grade 1 toxicities included pruritis, abdominal pain, skin rash, fatigue, fever, cramping, myalgia, and diarrhea. There was a Grade 3 case of encephalopathy and a Grade 2 case of diplopia. Of the 6 patients who experienced an adverse drug reaction, 2 (with only Grade 1 toxicities) continued treatment at their same dose frequency; the others changed to 240 mg Q2w. All 4 patients who experienced an ADR and had their dose changed to 240 mg Q2w experienced resolution or improvement in their symptoms except for 1 patient’s complaint of abdominal pain.
Conclusion: 480 Q4w dosing of Nivolumab may have a different ADR profile from prior dose regimens; further quantitative analysis will be required to answer this question. Dose frequency change may present an opportunity to relieve toxicities while allowing patients to continue treatment.
Melanoma of Unknown Primary Presenting as a Parotid Gland Mass
Background: Malignant melanoma is an aggressive malignancy that can present as a poorly differentiated neoplasm. Loss of S100 and melanA antigenicity can make pathologic identification difficult, especially in those patients who lack a cutaneous primary lesion. Immunostaining with SOX10, a key nuclear transcription factor in the differentiation of neural crest progenitor cells to melanocytes, has a high reported sensitivity and specificity for pathologic identification of melanoma in difficult cases.
Case Report: A 69-year-old male with a history of heavy tobacco use presented to the otolaryngology clinic with a left parotid mass. He underwent a parotid gland biopsy, which was significant for a high grade, poorly differentiated malignancy of unclear primary source. A staging PET/CT demonstrated localized hypermetabolic activity in the draining left cervical lymph node basins. He underwent a left modified radical neck dissection and parotidectomy. Pathologic assessment demonstrated a 3.9 × 1.6 × 1.6 cm3 poorly differentiated carcinoma with perineural invasion and 8/85 lymph nodes involved. Morphologically, it had features of a high grade epithelioid tumor with spindle cell features. Immunohistochemical (IHC) stains were negative for epithelial markers (AE1/3, EMA, CK5/6, CAM5.2), smooth muscle actin, CD34, S100, and melanA. Given the concern for a spindle cell melanoma that lost its antigenicity for S100 and melanA, a SOX10 IHC stain was performed.
The SOX10 immunostain demonstrated strong, diffuse positivity which secured the diagnosis of malignant melanoma. Molecular testing for BRAF and KIT mutations was negative. The nal diagnosis was a stage IVA (pT2pN2bM0) malignant melanoma of the parotid gland without a cutaneous primary lesion. The patient received a course of adjuvant radiation to a total dose of 66Gy and will complete one year of adjuvant immunotherapy with Nivolumab.
Conclusion: Malignant melanoma can present as a poorly differentiated malignancy and may be difficult to diagnose by providers, especially in the absence of a typical clinical history and a primary cutaneous lesion. In cases where the standard melanoma immunostains are negative, IHC staining with SOX10 can help secure the diagnosis with high sensitivity and specificity.
Background: Malignant melanoma is an aggressive malignancy that can present as a poorly differentiated neoplasm. Loss of S100 and melanA antigenicity can make pathologic identification difficult, especially in those patients who lack a cutaneous primary lesion. Immunostaining with SOX10, a key nuclear transcription factor in the differentiation of neural crest progenitor cells to melanocytes, has a high reported sensitivity and specificity for pathologic identification of melanoma in difficult cases.
Case Report: A 69-year-old male with a history of heavy tobacco use presented to the otolaryngology clinic with a left parotid mass. He underwent a parotid gland biopsy, which was significant for a high grade, poorly differentiated malignancy of unclear primary source. A staging PET/CT demonstrated localized hypermetabolic activity in the draining left cervical lymph node basins. He underwent a left modified radical neck dissection and parotidectomy. Pathologic assessment demonstrated a 3.9 × 1.6 × 1.6 cm3 poorly differentiated carcinoma with perineural invasion and 8/85 lymph nodes involved. Morphologically, it had features of a high grade epithelioid tumor with spindle cell features. Immunohistochemical (IHC) stains were negative for epithelial markers (AE1/3, EMA, CK5/6, CAM5.2), smooth muscle actin, CD34, S100, and melanA. Given the concern for a spindle cell melanoma that lost its antigenicity for S100 and melanA, a SOX10 IHC stain was performed.
The SOX10 immunostain demonstrated strong, diffuse positivity which secured the diagnosis of malignant melanoma. Molecular testing for BRAF and KIT mutations was negative. The nal diagnosis was a stage IVA (pT2pN2bM0) malignant melanoma of the parotid gland without a cutaneous primary lesion. The patient received a course of adjuvant radiation to a total dose of 66Gy and will complete one year of adjuvant immunotherapy with Nivolumab.
Conclusion: Malignant melanoma can present as a poorly differentiated malignancy and may be difficult to diagnose by providers, especially in the absence of a typical clinical history and a primary cutaneous lesion. In cases where the standard melanoma immunostains are negative, IHC staining with SOX10 can help secure the diagnosis with high sensitivity and specificity.
Background: Malignant melanoma is an aggressive malignancy that can present as a poorly differentiated neoplasm. Loss of S100 and melanA antigenicity can make pathologic identification difficult, especially in those patients who lack a cutaneous primary lesion. Immunostaining with SOX10, a key nuclear transcription factor in the differentiation of neural crest progenitor cells to melanocytes, has a high reported sensitivity and specificity for pathologic identification of melanoma in difficult cases.
Case Report: A 69-year-old male with a history of heavy tobacco use presented to the otolaryngology clinic with a left parotid mass. He underwent a parotid gland biopsy, which was significant for a high grade, poorly differentiated malignancy of unclear primary source. A staging PET/CT demonstrated localized hypermetabolic activity in the draining left cervical lymph node basins. He underwent a left modified radical neck dissection and parotidectomy. Pathologic assessment demonstrated a 3.9 × 1.6 × 1.6 cm3 poorly differentiated carcinoma with perineural invasion and 8/85 lymph nodes involved. Morphologically, it had features of a high grade epithelioid tumor with spindle cell features. Immunohistochemical (IHC) stains were negative for epithelial markers (AE1/3, EMA, CK5/6, CAM5.2), smooth muscle actin, CD34, S100, and melanA. Given the concern for a spindle cell melanoma that lost its antigenicity for S100 and melanA, a SOX10 IHC stain was performed.
The SOX10 immunostain demonstrated strong, diffuse positivity which secured the diagnosis of malignant melanoma. Molecular testing for BRAF and KIT mutations was negative. The nal diagnosis was a stage IVA (pT2pN2bM0) malignant melanoma of the parotid gland without a cutaneous primary lesion. The patient received a course of adjuvant radiation to a total dose of 66Gy and will complete one year of adjuvant immunotherapy with Nivolumab.
Conclusion: Malignant melanoma can present as a poorly differentiated malignancy and may be difficult to diagnose by providers, especially in the absence of a typical clinical history and a primary cutaneous lesion. In cases where the standard melanoma immunostains are negative, IHC staining with SOX10 can help secure the diagnosis with high sensitivity and specificity.
Severe lipodystrophy linked to pembrolizumab
according to a case study.
A 47-year-old woman received pembrolizumab to treat metastatic melanoma and developed immune-related generalized acquired lipodystrophy. The condition has persisted 12 months after she stopped taking pembrolizumab.
Julie Delyon, MD, PhD, of Saint-Louis Hospital in Paris, and colleagues described this case in the British Journal of Dermatology.
The patient was diagnosed with BRAF-mutated, stage IV melanoma with bone and lymph node metastases. She received pembrolizumab at 2 mg/kg every 3 weeks as first-line treatment. She achieved a complete response and was still in remission at last follow-up.
The patient was obese at baseline, with a body mass index of 40 kg/m2, but she did not have diabetes, hepatic steatosis, or dyslipidemia.
Within 2 months of starting pembrolizumab, the patient observed “some major changes to her physical appearance,” according to Dr. Delyon and colleagues. At 10 months, the patient had severe lipodystrophy.
The patient experienced a change in fat distribution – namely, severe peripheral lipoatrophy and an accumulation of trunk fat. Imaging revealed a decrease in subcutaneous adipose tissue, which contrasted with an increase in visceral fat observed from baseline.
The patient also developed “moderate hirsutism, facial and limb atrophy, and prominent forearm and leg muscles and veins,” according to the authors. She had impaired glucose tolerance with insulin resistance, reduced concentrations of leptin and adiponectin, hypertriglyceridemia, a low level of HDL cholesterol, and hepatic steatosis.
Analyses of subcutaneous fat revealed adipose tissue atrophy with edema, lipophages, and CD3+/CD4+ T-cell infiltration of the fat and vessel walls. This suggested that the lipodystrophy had an autoimmune origin, according to the authors.
The patient had no family history of autoimmune disease or lipodystrophy. She tested negative for mutations in 23 genes associated with generalized lipodystrophy.
The patient also tested negative for HIV, antinuclear antibodies, native anti-DNA, and anti-insulin receptor antibodies. There were no signs of panniculitis, and the authors noted that “there were no arguments in favor of hyperthyroidism, Cushing syndrome, or acromegaly.”
In an attempt to reverse the lipodystrophy, the researchers stopped pembrolizumab treatment. The patient was treated for diabetes and hypertriglyceridemia as well. She could not receive corticosteroids because of the risk of severe metabolic complications, and she didn’t receive metreleptin because it wasn’t available. The patient still had lipodystrophy 12 months after stopping pembrolizumab.
Dr. Delyon and colleagues wrote that this case suggests pembrolizumab, and perhaps other anti–programmed death 1 therapies, may cause lipodystrophy with severe metabolic complications. “The long-term side effects of such metabolic adverse events, although rare, are unknown and will probably become a topic of utmost importance, considering the increasing rate of remission following ICIs and their use in the adjuvant setting.”
Two coauthors reported relationships with Merck, which markets pembrolizumab as Keytruda. The authors also reported relationships with Bristol-Myers Squibb, Pierre Fabre, Takeda, Innate Pharma, LEO Pharma, Roche, GlaxoSmithKline, Novartis, and Amgen.
SOURCE: Delyon J et al. Br J Dermatol. 2019 May 11. doi: 10.1111/bjd.18124.
according to a case study.
A 47-year-old woman received pembrolizumab to treat metastatic melanoma and developed immune-related generalized acquired lipodystrophy. The condition has persisted 12 months after she stopped taking pembrolizumab.
Julie Delyon, MD, PhD, of Saint-Louis Hospital in Paris, and colleagues described this case in the British Journal of Dermatology.
The patient was diagnosed with BRAF-mutated, stage IV melanoma with bone and lymph node metastases. She received pembrolizumab at 2 mg/kg every 3 weeks as first-line treatment. She achieved a complete response and was still in remission at last follow-up.
The patient was obese at baseline, with a body mass index of 40 kg/m2, but she did not have diabetes, hepatic steatosis, or dyslipidemia.
Within 2 months of starting pembrolizumab, the patient observed “some major changes to her physical appearance,” according to Dr. Delyon and colleagues. At 10 months, the patient had severe lipodystrophy.
The patient experienced a change in fat distribution – namely, severe peripheral lipoatrophy and an accumulation of trunk fat. Imaging revealed a decrease in subcutaneous adipose tissue, which contrasted with an increase in visceral fat observed from baseline.
The patient also developed “moderate hirsutism, facial and limb atrophy, and prominent forearm and leg muscles and veins,” according to the authors. She had impaired glucose tolerance with insulin resistance, reduced concentrations of leptin and adiponectin, hypertriglyceridemia, a low level of HDL cholesterol, and hepatic steatosis.
Analyses of subcutaneous fat revealed adipose tissue atrophy with edema, lipophages, and CD3+/CD4+ T-cell infiltration of the fat and vessel walls. This suggested that the lipodystrophy had an autoimmune origin, according to the authors.
The patient had no family history of autoimmune disease or lipodystrophy. She tested negative for mutations in 23 genes associated with generalized lipodystrophy.
The patient also tested negative for HIV, antinuclear antibodies, native anti-DNA, and anti-insulin receptor antibodies. There were no signs of panniculitis, and the authors noted that “there were no arguments in favor of hyperthyroidism, Cushing syndrome, or acromegaly.”
In an attempt to reverse the lipodystrophy, the researchers stopped pembrolizumab treatment. The patient was treated for diabetes and hypertriglyceridemia as well. She could not receive corticosteroids because of the risk of severe metabolic complications, and she didn’t receive metreleptin because it wasn’t available. The patient still had lipodystrophy 12 months after stopping pembrolizumab.
Dr. Delyon and colleagues wrote that this case suggests pembrolizumab, and perhaps other anti–programmed death 1 therapies, may cause lipodystrophy with severe metabolic complications. “The long-term side effects of such metabolic adverse events, although rare, are unknown and will probably become a topic of utmost importance, considering the increasing rate of remission following ICIs and their use in the adjuvant setting.”
Two coauthors reported relationships with Merck, which markets pembrolizumab as Keytruda. The authors also reported relationships with Bristol-Myers Squibb, Pierre Fabre, Takeda, Innate Pharma, LEO Pharma, Roche, GlaxoSmithKline, Novartis, and Amgen.
SOURCE: Delyon J et al. Br J Dermatol. 2019 May 11. doi: 10.1111/bjd.18124.
according to a case study.
A 47-year-old woman received pembrolizumab to treat metastatic melanoma and developed immune-related generalized acquired lipodystrophy. The condition has persisted 12 months after she stopped taking pembrolizumab.
Julie Delyon, MD, PhD, of Saint-Louis Hospital in Paris, and colleagues described this case in the British Journal of Dermatology.
The patient was diagnosed with BRAF-mutated, stage IV melanoma with bone and lymph node metastases. She received pembrolizumab at 2 mg/kg every 3 weeks as first-line treatment. She achieved a complete response and was still in remission at last follow-up.
The patient was obese at baseline, with a body mass index of 40 kg/m2, but she did not have diabetes, hepatic steatosis, or dyslipidemia.
Within 2 months of starting pembrolizumab, the patient observed “some major changes to her physical appearance,” according to Dr. Delyon and colleagues. At 10 months, the patient had severe lipodystrophy.
The patient experienced a change in fat distribution – namely, severe peripheral lipoatrophy and an accumulation of trunk fat. Imaging revealed a decrease in subcutaneous adipose tissue, which contrasted with an increase in visceral fat observed from baseline.
The patient also developed “moderate hirsutism, facial and limb atrophy, and prominent forearm and leg muscles and veins,” according to the authors. She had impaired glucose tolerance with insulin resistance, reduced concentrations of leptin and adiponectin, hypertriglyceridemia, a low level of HDL cholesterol, and hepatic steatosis.
Analyses of subcutaneous fat revealed adipose tissue atrophy with edema, lipophages, and CD3+/CD4+ T-cell infiltration of the fat and vessel walls. This suggested that the lipodystrophy had an autoimmune origin, according to the authors.
The patient had no family history of autoimmune disease or lipodystrophy. She tested negative for mutations in 23 genes associated with generalized lipodystrophy.
The patient also tested negative for HIV, antinuclear antibodies, native anti-DNA, and anti-insulin receptor antibodies. There were no signs of panniculitis, and the authors noted that “there were no arguments in favor of hyperthyroidism, Cushing syndrome, or acromegaly.”
In an attempt to reverse the lipodystrophy, the researchers stopped pembrolizumab treatment. The patient was treated for diabetes and hypertriglyceridemia as well. She could not receive corticosteroids because of the risk of severe metabolic complications, and she didn’t receive metreleptin because it wasn’t available. The patient still had lipodystrophy 12 months after stopping pembrolizumab.
Dr. Delyon and colleagues wrote that this case suggests pembrolizumab, and perhaps other anti–programmed death 1 therapies, may cause lipodystrophy with severe metabolic complications. “The long-term side effects of such metabolic adverse events, although rare, are unknown and will probably become a topic of utmost importance, considering the increasing rate of remission following ICIs and their use in the adjuvant setting.”
Two coauthors reported relationships with Merck, which markets pembrolizumab as Keytruda. The authors also reported relationships with Bristol-Myers Squibb, Pierre Fabre, Takeda, Innate Pharma, LEO Pharma, Roche, GlaxoSmithKline, Novartis, and Amgen.
SOURCE: Delyon J et al. Br J Dermatol. 2019 May 11. doi: 10.1111/bjd.18124.
FROM THE BRITISH JOURNAL OF DERMATOLOGY
Dabrafenib plus trametinib yields long-term benefit in melanoma patients
Dabrafenib plus trametinib treatment was associated with a 5-year overall survival rate of 34% in patients with melanoma harboring a BRAF V600E or V600K mutation, according to a combined analysis of two trials.
The 5-year progression-free survival rate was 19% in the long-term, pooled analysis of the COMBI-d and COMBI-v trials, which included at total of 563 patients with previously untreated, unresectable or metastatic melanoma who received combined treatment with the BRAF inhibitor dabrafenib and the MEK inhibitor trametinib.
Previously reported 5-year progression-free survival rates for patients treated with anti–programmed death-1 checkpoint inhibitors, either nivolumab or pembrolizumab, “appear to be similar” to these results for dabrafenib plus trametinib, investigators said in a report on the analysis appearing in the New England Journal of Medicine.
To date, however, 5-year survival data have not been reported for other BRAF-targeted therapies, according to the investigators, who were led by Caroline Robert, MD, PhD, of Institut Gustave Roussy and Paris-Sud-Paris-Saclay University, Villejuif, France.
“These data will be critical to assess the potential of therapy to exert long-term disease control through analysis of survival plateaus and to understand factors predictive of long-term survival,” Dr. Robert and coauthors wrote in their report.
A total of 211 patients in the COMBI-d trial were randomly allocated to receive the combination of dabrafenib plus trametinib, while in COMBI-v, 352 received this combination therapy, according to investigators.
Notably, the survival curves for dabrafenib plus trametinib appear to plateau starting at 3 years, investigators reported. In a previously published report on pooled COMBI-d and COMBI-v data, the 3-year progression-free survival rate was 23%, and the 3-year overall survival rate was 44%.
In this more recent analysis, progression-free survival rates were 21% at 4 years and 19% at 5 years, while overall survival rates were 37% at 4 years and 34% at 5 years.
“This finding suggests stabilization of rates of progression-free survival and overall survival over time in this population,” Dr. Robert and colleagues wrote.
Survival rates were higher in patients with normal lactate dehydrogenase (LDH) levels at baseline, and they were especially high in those with normal LDH and three or fewer disease sites at baseline, according to the report. Specifically, the reported 5-year rates of progression-free and overall survival were 31% and 55%, respectively.
Other factors associated with prolonged progression-free survival included female sex, older age, better performance status, and BRAF V600E genotype, according to results of a multivariate analysis that investigators said confirmed findings from the previously reported 3-year data.
The study was supported by GlaxoSmithKline and Novartis. Dr. Robert provided disclosures related to BMS, Pierre Fabre, Novartis, Amgen, Merck, Roche, MSD, and Sanofi.
SOURCE: Robert C et al. N Engl J Med. 2019 Aug 15. doi: 10.1056/NEJMoa1904059
Dabrafenib plus trametinib treatment was associated with a 5-year overall survival rate of 34% in patients with melanoma harboring a BRAF V600E or V600K mutation, according to a combined analysis of two trials.
The 5-year progression-free survival rate was 19% in the long-term, pooled analysis of the COMBI-d and COMBI-v trials, which included at total of 563 patients with previously untreated, unresectable or metastatic melanoma who received combined treatment with the BRAF inhibitor dabrafenib and the MEK inhibitor trametinib.
Previously reported 5-year progression-free survival rates for patients treated with anti–programmed death-1 checkpoint inhibitors, either nivolumab or pembrolizumab, “appear to be similar” to these results for dabrafenib plus trametinib, investigators said in a report on the analysis appearing in the New England Journal of Medicine.
To date, however, 5-year survival data have not been reported for other BRAF-targeted therapies, according to the investigators, who were led by Caroline Robert, MD, PhD, of Institut Gustave Roussy and Paris-Sud-Paris-Saclay University, Villejuif, France.
“These data will be critical to assess the potential of therapy to exert long-term disease control through analysis of survival plateaus and to understand factors predictive of long-term survival,” Dr. Robert and coauthors wrote in their report.
A total of 211 patients in the COMBI-d trial were randomly allocated to receive the combination of dabrafenib plus trametinib, while in COMBI-v, 352 received this combination therapy, according to investigators.
Notably, the survival curves for dabrafenib plus trametinib appear to plateau starting at 3 years, investigators reported. In a previously published report on pooled COMBI-d and COMBI-v data, the 3-year progression-free survival rate was 23%, and the 3-year overall survival rate was 44%.
In this more recent analysis, progression-free survival rates were 21% at 4 years and 19% at 5 years, while overall survival rates were 37% at 4 years and 34% at 5 years.
“This finding suggests stabilization of rates of progression-free survival and overall survival over time in this population,” Dr. Robert and colleagues wrote.
Survival rates were higher in patients with normal lactate dehydrogenase (LDH) levels at baseline, and they were especially high in those with normal LDH and three or fewer disease sites at baseline, according to the report. Specifically, the reported 5-year rates of progression-free and overall survival were 31% and 55%, respectively.
Other factors associated with prolonged progression-free survival included female sex, older age, better performance status, and BRAF V600E genotype, according to results of a multivariate analysis that investigators said confirmed findings from the previously reported 3-year data.
The study was supported by GlaxoSmithKline and Novartis. Dr. Robert provided disclosures related to BMS, Pierre Fabre, Novartis, Amgen, Merck, Roche, MSD, and Sanofi.
SOURCE: Robert C et al. N Engl J Med. 2019 Aug 15. doi: 10.1056/NEJMoa1904059
Dabrafenib plus trametinib treatment was associated with a 5-year overall survival rate of 34% in patients with melanoma harboring a BRAF V600E or V600K mutation, according to a combined analysis of two trials.
The 5-year progression-free survival rate was 19% in the long-term, pooled analysis of the COMBI-d and COMBI-v trials, which included at total of 563 patients with previously untreated, unresectable or metastatic melanoma who received combined treatment with the BRAF inhibitor dabrafenib and the MEK inhibitor trametinib.
Previously reported 5-year progression-free survival rates for patients treated with anti–programmed death-1 checkpoint inhibitors, either nivolumab or pembrolizumab, “appear to be similar” to these results for dabrafenib plus trametinib, investigators said in a report on the analysis appearing in the New England Journal of Medicine.
To date, however, 5-year survival data have not been reported for other BRAF-targeted therapies, according to the investigators, who were led by Caroline Robert, MD, PhD, of Institut Gustave Roussy and Paris-Sud-Paris-Saclay University, Villejuif, France.
“These data will be critical to assess the potential of therapy to exert long-term disease control through analysis of survival plateaus and to understand factors predictive of long-term survival,” Dr. Robert and coauthors wrote in their report.
A total of 211 patients in the COMBI-d trial were randomly allocated to receive the combination of dabrafenib plus trametinib, while in COMBI-v, 352 received this combination therapy, according to investigators.
Notably, the survival curves for dabrafenib plus trametinib appear to plateau starting at 3 years, investigators reported. In a previously published report on pooled COMBI-d and COMBI-v data, the 3-year progression-free survival rate was 23%, and the 3-year overall survival rate was 44%.
In this more recent analysis, progression-free survival rates were 21% at 4 years and 19% at 5 years, while overall survival rates were 37% at 4 years and 34% at 5 years.
“This finding suggests stabilization of rates of progression-free survival and overall survival over time in this population,” Dr. Robert and colleagues wrote.
Survival rates were higher in patients with normal lactate dehydrogenase (LDH) levels at baseline, and they were especially high in those with normal LDH and three or fewer disease sites at baseline, according to the report. Specifically, the reported 5-year rates of progression-free and overall survival were 31% and 55%, respectively.
Other factors associated with prolonged progression-free survival included female sex, older age, better performance status, and BRAF V600E genotype, according to results of a multivariate analysis that investigators said confirmed findings from the previously reported 3-year data.
The study was supported by GlaxoSmithKline and Novartis. Dr. Robert provided disclosures related to BMS, Pierre Fabre, Novartis, Amgen, Merck, Roche, MSD, and Sanofi.
SOURCE: Robert C et al. N Engl J Med. 2019 Aug 15. doi: 10.1056/NEJMoa1904059
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Key clinical point: A long-term survival benefit was seen in about a third of patients with metastatic or unresectable melanoma who underwent first-line treatment with dabrafenib and trametinib.
Major finding: The 5-year rates of progression-free survival and overall survival were 19% and 34%, respectively.
Study details: Pooled analysis including 563 patients randomly allocated to the combination treatment in two randomized trials (COMBI-d and COMBI-v).
Disclosures: The study was supported by GlaxoSmithKline and Novartis. The first author provided disclosures related to BMS, Pierre Fabre, Novartis, Amgen, Merck, Roche, MSD, and Sanofi.
Source: Robert C et al. N Engl J Med. 2019 Aug 15. doi: 10.1056/NEJMoa1904059
Noninvasive Imaging Tools in Dermatology
Traditionally, diagnosis of skin disease relies on clinical inspection, often followed by biopsy and histopathologic examination. In recent years, new noninvasive tools have emerged that can aid in clinical diagnosis and reduce the number of unnecessary benign biopsies. Although there has been a surge in noninvasive diagnostic technologies, many tools are still in research and development phases, with few tools widely adopted and used in regular clinical practice. In this article, we discuss the use of dermoscopy, reflectance confocal microscopy (RCM), and optical coherence tomography (OCT) in the diagnosis and management of skin disease.
Dermoscopy
Dermoscopy, also known as epiluminescence light microscopy and previously known as dermatoscopy, utilizes a ×10 to ×100 microscope objective with a light source to magnify and visualize structures present below the skin’s surface, such as melanin and blood vessels. There are 3 types of dermoscopy: conventional nonpolarized dermoscopy, polarized contact dermoscopy, and nonpolarized contact dermoscopy (Figure 1). Traditional nonpolarized dermoscopy requires a liquid medium and direct contact with the skin, and it relies on light reflection and refraction properties.1 Cross-polarized light sources allow visualization of deeper structures, either with or without a liquid medium and contact with the skin surface. Although there is overall concurrence among the different types of dermoscopy, subtle differences in the appearance of color, features, and structure are present.1
Dermoscopy offers many benefits for dermatologists and other providers. It can be used to aid in the diagnosis of cutaneous neoplasms and other skin diseases. Numerous low-cost dermatoscopes currently are commercially available. The handheld, easily transportable nature of dermatoscopes have resulted in widespread practice integration. Approximately 84% of attending dermatologists in US academic settings reported using dermoscopy, and many refer to the dermatoscope as “the dermatologist’s stethoscope.”2 In addition, 6% to 15% of other US providers, including family physicians, internal medicine physicians, and plastic surgeons, have reported using dermoscopy in their clinical practices. Limitations of dermoscopy include visualization of the skin surface only and not deeper structures within the tissue, the need for training for adequate interpretation of dermoscopic images, and lack of reimbursement for dermoscopic examination.3
Many dermoscopic structures that correspond well with histopathology have been described. Dermoscopy has a sensitivity of 79% to 96% and specificity of 69% to 99% in the diagnosis of melanoma.4 There is variable data on the specificity of dermoscopy in the diagnosis of melanoma, with one meta-analysis finding no statistically significant difference in specificity compared to naked eye examination,5 while other studies report increased specificity and subsequent reduction in biopsy of benign lesions.6,7 Dermoscopy also can aid in the diagnosis of keratinocytic neoplasms, and dermoscopy also results in a sensitivity of 78.6% to 100% and a specificity of 53.8% to 100% in the diagnosis of basal cell carcinoma (BCC).8 Limitations of dermoscopy include false-positive diagnoses, commonly seborrheic keratoses and nevi, resulting in unnecessary biopsies, as well as false-negative diagnoses, commonly amelanotic and nevoid melanoma, resulting in delays in skin cancer diagnosis and resultant poor outcomes.9 Dermoscopy also is used to aid in the diagnosis of inflammatory and infectious skin diseases, as well as scalp, hair, and nail disorders.10
Reflectance Confocal Microscopy
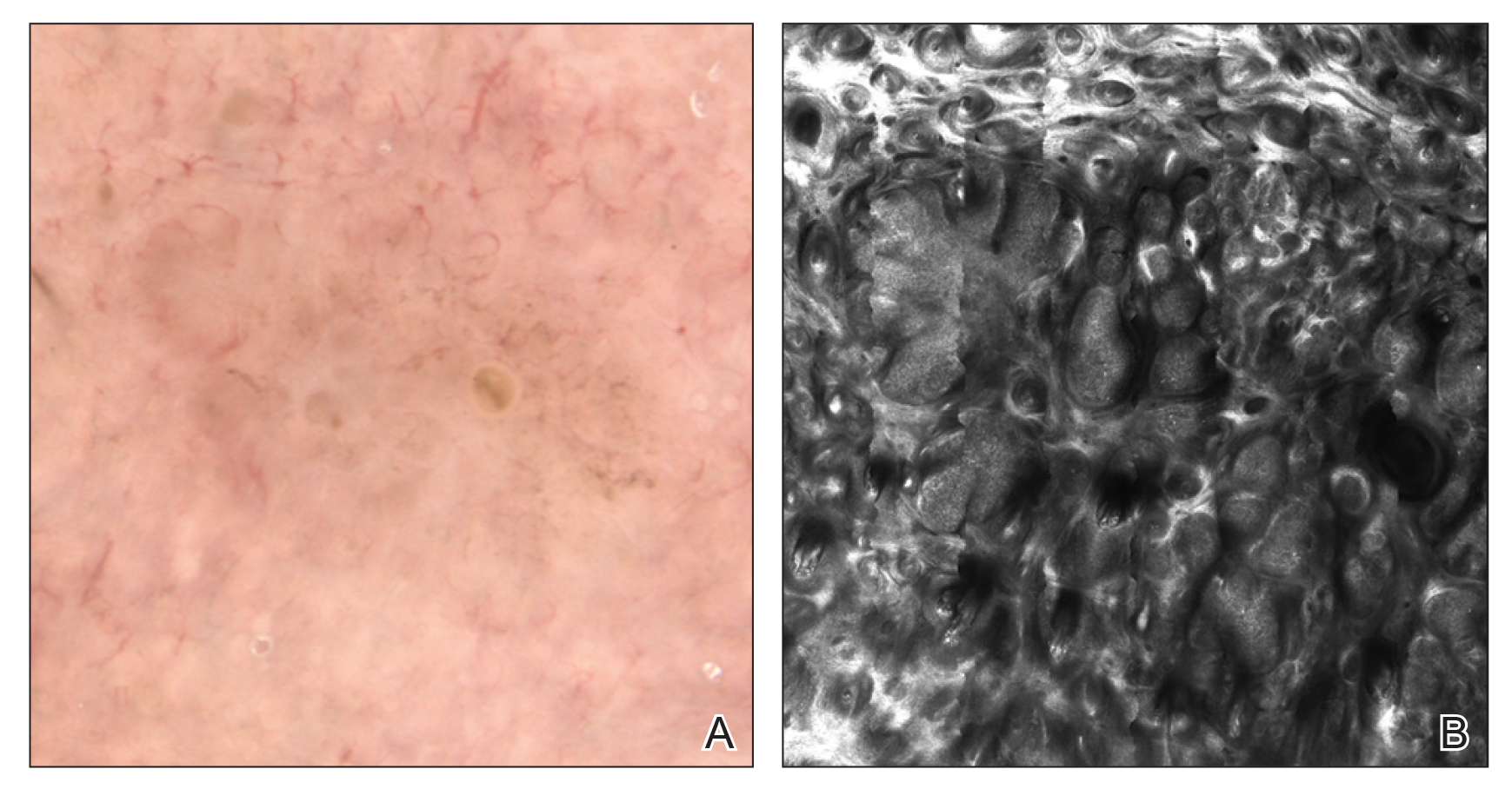
Reflectance confocal microscopy utilizes an 830-nm laser to capture horizontal en face images of the skin with high resolution. Different structures of the skin have varying indices of refraction: keratin, melanin, and collagen appear bright white, while other components appear dark, generating black-and-white RCM images.11 Currently, there are 2 reflectance confocal microscopes that are commercially available in the United States. The Vivascope 1500 (Caliber ID) is the traditional model that captures 8×8-mm images, and the Vivascope 3000 (Caliber ID) is a smaller handheld model that captures 0.5×0.5-mm images. The traditional model provides the advantages of higher-resolution images and the ability to capture larger surface areas but is best suited to image flat areas of skin to which a square window can be adhered. The handheld model allows improved contact with the varying topography of skin; does not require an adhesive window; and can be used to image cartilaginous, mucosal, and sensitive surfaces. However, it can be difficult to correlate individual images captured by the handheld RCM with the location relative to the lesion, as it is exquisitely sensitive to motion and also is operator dependent. Although complex algorithms are under development to stitch individual images to provide better correlation with the geography of the lesion, such programs are not yet widely available.12
Reflectance confocal microscopy affords many benefits for patients and providers. It is noninvasive and painless and is capable of imaging in vivo live skin as compared to clinical examination and dermoscopy, which only allow for visualization of the skin’s surface. Reflectance confocal microscopy also is time efficient, as imaging of a single lesion can be completed in 10 to 15 minutes. This technology generates high-resolution images, and RCM diagnosis has consistently demonstrated high sensitivity and specificity when compared to histopathology.13 Additionally, RCM imaging can spare biopsy and resultant scarring on cosmetically sensitive areas. Recently, RCM imaging of the skin has been granted Category I Current Procedural Terminology reimbursement codes that allow provider reimbursement and integration of RCM into daily practice14; however, private insurance coverage in the United States is variable. Limitations of RCM include a maximum depth of 200 to 300 µm, high cost to procure a reflectance confocal microscope, and the need for considerable training and practice to accurately interpret grayscale en face images.15
There has been extensive research regarding the use of RCM in the evaluation of cutaneous neoplasms and other skin diseases. Numerous features and patterns have been identified and described that correspond with different skin diseases and correspond well with histopathology (Figure 2).13,16,17 Reflectance confocal microscopy has demonstrated consistently high accuracy in the diagnosis of melanocytic lesions, with a sensitivity of 93% to 100% and a specificity of 75% to 99%.18-21 Reflectance confocal microscopy is especially useful in the evaluation of clinically or dermoscopically equivocal pigmented lesions due to greater specificity, resulting in a reduction of unnecessary biopsies.22,23 It also has high accuracy in the diagnosis of keratinocytic neoplasms, with a sensitivity of 82% to 100% and a specificity of 78% to 97% in the diagnosis of BCC,24 and a sensitivity of 74% to 100% and specificity of 78% to 100% in the diagnosis of squamous cell carcinoma (SCC).25,26 Evaluation of SCC and actinic keratosis (AK) using RCM may be limited by considerable hyperkeratosis and ulceration. In addition, it can be challenging to differentiate AK and SCC on RCM, and considerable expertise is required to accurately grade cytologic and architectural atypia.27 However, RCM has been used to discriminate between in situ and invasive proliferations.28 Reflectance confocal microscopy has wide applications in the diagnosis and management of cutaneous infections29,30 and inflammatory skin diseases.29,31-33 Recent RCM research explored the use of RCM to identify biopsy sites,34 delineate presurgical tumor margins,35,36 and monitor response to noninvasive treatments.37,38
Optical Coherence Tomography
Optical coherence tomography is an imaging modality that utilizes light backscatter from infrared light to produce grayscale cross-sectional or vertical images and horizontal en face images.39 Optical coherence tomography can visualize structures in the epidermis, dermoepidermal junction, and upper dermis.40 It can image boundaries of structures but cannot visualize individual cells.
There are different types of OCT devices available, including frequency-domain OCT (FD-OCT), or conventional OCT, and high-definition OCT (HD-OCT). With FD-OCT, images are captured at a maximum depth of 1 to 2 mm but with limited resolution. High-definition OCT has superior resolution compared to FD-OCT but is restricted to a shallower depth of 750 μm.39 The main advantage of OCT is the ability to noninvasively image live tissue and visualize 2- to 5-times greater depth as compared to RCM. Several OCT devices have obtained US Food and Drug Administration approval; however, OCT has not been widely adopted into clinical practice and is available only in tertiary academic centers. Additionally, OCT imaging in dermatology is rarely reimbursed. Other limitations of OCT include poor resolution of images, high cost to procure an OCT device, and the need for advanced training and experience to accurately interpret images.40,41
Optical coherence tomography primarily is used to diagnose cutaneous neoplasms. The best evidence of the diagnostic accuracy of OCT is in the setting of BCC, with a recent systematic review reporting a sensitivity of 66% to 96% and a specificity of 75% to 86% for conventional FD-OCT.42 The use of FD-OCT results in an increase in specificity without a significant change in sensitivity when compared to dermoscopy in the diagnosis of BCC.43 Melanoma is difficult to diagnose via FD-OCT, as the visualization of architectural features often is limited by poor resolution.44 A study of HD-OCT in the diagnosis of melanoma with a limited sample size reported a sensitivity of 74% to 80% and a specificity of 92% to 93%.45 Similarly, a study of HD-OCT used in the diagnosis of AK and SCC revealed a sensitivity and specificity of 81.6% and 92.6%, respectively, for AK and 93.8% and 98.9%, respectively, for SCC.46
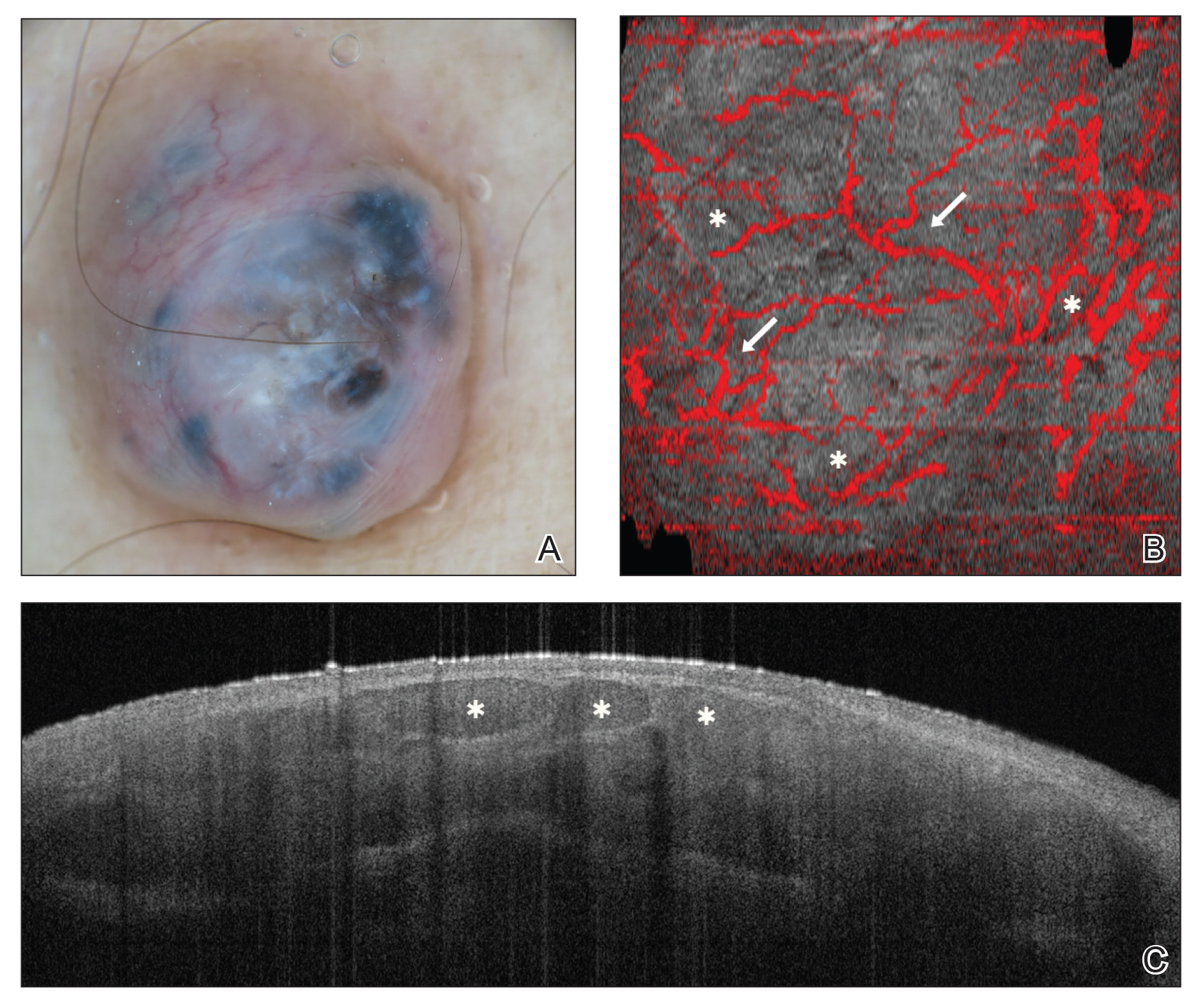
Numerous algorithms and scoring systems have been developed to further explore the utility of OCT in the diagnosis of cutaneous neoplasms.47,48 Recent research investigated the utility of dynamic OCT, which can evaluate microvasculature in the diagnosis of cutaneous neoplasms (Figure 3)49; the combination of OCT with other imaging modalities50,51; the use of OCT to delineate presurgical margins52,53; and the role of OCT in the diagnosis and monitoring of inflammatory and infectious skin diseases.54,55
Final Thoughts
In recent years, there has been a surge of interest in noninvasive techniques for diagnosis and management of skin diseases; however, noninvasive tools exist on a spectrum in dermatology. Dermoscopy provides low-cost imaging of the skin’s surface and has been widely adopted by dermatologists and other providers to aid in clinical diagnosis. Reflectance confocal microscopy provides reimbursable in vivo imaging of live tissue with cellular-level resolution but is limited by depth, cost, and need for advanced training; thus, RCM has only been adopted in some clinical practices. Optical coherence tomography offers in vivo imaging of live tissue with substantial depth but poor resolution, high cost, need for advanced training, and rare reimbursement for providers. Future directions include combination of complementary imaging modalities, increased clinical practice integration, and education and reimbursement for providers.
- Benvenuto-Andrade C, Dusza SW, Agero AL, et al. Differences between polarized light dermoscopy and immersion contact dermoscopy for the evaluation of skin lesions. Arch Dermatol. 2007;143:329-338.
- Terushkin V, Oliveria SA, Marghoob AA, et al. Use of and beliefs about total body photography and dermatoscopy among US dermatology training programs: an update. J Am Acad Dermatol. 2010;62:794-803.
- Morris JB, Alfonso SV, Hernandez N, et al. Use of and intentions to use dermoscopy among physicians in the United States. Dermatol Pract Concept. 2017;7:7-16.
- Yélamos O, Braun RP, Liopyris K, et al. Dermoscopy and dermatopathology correlates of cutaneous neoplasms. J Am Acad Dermatol. 2019;80:341-363.
- Vestergaard ME, Macaskill P, Holt PE, et al. Dermoscopy compared with naked eye examination for the diagnosis of primary melanoma: a meta-analysis of studies performed in a clinical setting. Br J Dermatol. 2008;159:669-676.
- Carli P, de Giorgi V, Chiarugi A, et al. Addition of dermoscopy to conventional naked-eye examination in melanoma screening: a randomized study. J Am Acad Dermatol. 2004;50:683-668.
- Lallas A, Zalaudek I, Argenziano G, et al. Dermoscopy in general dermatology. Dermatol Clin. 2013;31:679-694.
- Reiter O, Mimouni I, Gdalvevich M, et al. The diagnostic accuracy of dermoscopy for basal cell carcinoma: a systematic review and meta-analysis. J Am Acad Dermatol. 2019;80:1380-1388.
- Papageorgiou V, Apalla Z, Sotiriou E, et al. The limitations of dermoscopy: false-positive and false-negative tumours. J Eur Acad Dermatol Venereol. 2018;32:879-888.
- Micali G, Verzì AE, Lacarrubba F. Alternative uses of dermoscopy in daily clinical practice: an update. J Am Acad Dermatol. 2018;79:1117-1132.e1.
- Rajadhyaksha M, Grossman M, Esterowitz D, et al. In vivo confocal scanning laser microscopy of human skin: melanin provides strong contrast. J Invest Dermatol. 1995;104:946-952.
- Kose K, Gou M, Yélamos O, et al. Automated video-mosaicking approach for confocal microscopic imaging in vivo: an approach to address challenges in imaging living tissue and extend field of view. Sci Rep. 2017;7:10759.
- Rao BK, John AM, Francisco G, et al. Diagnostic accuracy of reflectance confocal microscopy for diagnosis of skin lesions [published online October 8, 2018]. Arch Pathol Lab Med. 2019;143:326-329.
- Current Procedural Terminology, Professional Edition. Chicago IL: American Medical Association; 2016. The preliminary physician fee schedule for 2017 is available at https://www.cms.gov/Medicare/Medicare-Fee-for-Service-Payment/PhysicianFeeSched/PFS-Federal-Regulation-Notices-Items/CMS-1654-P.html.
- Jain M, Pulijal SV, Rajadhyaksha M, et al. Evaluation of bedside diagnostic accuracy, learning curve, and challenges for a novice reflectance confocal microscopy reader for skin cancer detection in vivo. JAMA Dermatol. 2018;154:962-965.
- Rao BK, Pellacani G. Atlas of Confocal Microscopy in Dermatology: Clinical, Confocal, and Histological Images. New York, NY: NIDIskin LLC; 2013.
- Scope A, Benvenuto-Andrande C, Agero AL, et al. In vivo reflectance confocal microscopy imaging of melanocytic skin lesions: consensus terminology glossary and illustrative images. J Am Acad Dermatol. 2007;57:644-658.
- Gerger A, Hofmann-Wellenhof R, Langsenlehner U, et al. In vivo confocal laser scanning microscopy of melanocytic skin tumours: diagnostic applicability using unselected tumour images. Br J Dermatol. 2008;158:329-333.
- Stevenson AD, Mickan S, Mallett S, et al. Systematic review of diagnostic accuracy of reflectance confocal microscopy for melanoma diagnosis in patients with clinically equivocal skin lesions. Dermatol Pract Concept. 2013;3:19-27.
- Alarcon I, Carrera C, Palou J, et al. Impact of in vivo reflectance confocal microscopy on the number needed to treat melanoma in doubtful lesions. Br J Dermatol. 2014;170:802-808.
- Lovatto L, Carrera C, Salerni G, et al. In vivo reflectance confocal microscopy of equivocal melanocytic lesions detected by digital dermoscopy follow-up. J Eur Acad Dermatol Venereol. 2015;29:1918-1925.
- Guitera P, Pellacani G, Longo C, et al. In vivo reflectance confocal microscopy enhances secondary evaluation of melanocytic lesions. J Invest Dermatol. 2009;129:131-138.
- Xiong YQ, Ma SJ, Mo Y, et al. Comparison of dermoscopy and reflectance confocal microscopy for the diagnosis of malignant skin tumours: a meta-analysis. J Cancer Res Clin Oncol. 2017;143:1627-1635.
- Kadouch DJ, Schram ME, Leeflang MM, et al. In vivo confocal microscopy of basal cell carcinoma: a systematic review of diagnostic accuracy. J Eur Acad Dermatol Venereol. 2015;29:1890-1897.
- Dinnes J, Deeks JJ, Chuchu N, et al; Cochrane Skin Cancer Diagnostic Test Accuracy Group. Reflectance confocal microscopy for diagnosing keratinocyte skin cancers in adults. Cochrane Database Syst Rev. 2018;12:CD013191.
- Nguyen KP, Peppelman M, Hoogedoorn L, et al. The current role of in vivo reflectance confocal microscopy within the continuum of actinic keratosis and squamous cell carcinoma: a systematic review. Eur J Dermatol. 2016;26:549-565.
- Pellacani G, Ulrich M, Casari A, et al. Grading keratinocyte atypia in actinic keratosis: a correlation of reflectance confocal microscopy and histopathology. J Eur Acad Dermatol Venereol. 2015;29:2216-2221.
- Manfredini M, Longo C, Ferrari B, et al. Dermoscopic and reflectance confocal microscopy features of cutaneous squamous cell carcinoma. J Eur Acad Dermatol Venereol. 2017;31:1828-1833.
- Hoogedoorn L, Peppelman M, van de Kerkhof PC, et al. The value of in vivo reflectance confocal microscopy in the diagnosis and monitoring of inflammatory and infectious skin diseases: a systematic review. Br J Dermatol. 2015;172:1222-1248.
- Cinotti E, Perrot JL, Labeille B, et al. Reflectance confocal microscopy for cutaneous infections and infestations. J Eur Acad Dermatol Venereol. 2016;30:754-763.
- Ardigo M, Longo C, Gonzalez S; International Confocal Working Group Inflammatory Skin Diseases Project. Multicentre study on inflammatory skin diseases from The International Confocal Working Group: specific confocal microscopy features and an algorithmic method of diagnosis. Br J Dermatol. 2016;175:364-374.
- Ardigo M, Agozzino M, Franceschini C, et al. Reflectance confocal microscopy algorithms for inflammatory and hair diseases. Dermatol Clin. 2016;34:487-496.
- Manfredini M, Bettoli V, Sacripanti G, et al. The evolution of healthy skin to acne lesions: a longitudinal, in vivo evaluation with reflectance confocal microscopy and optical coherence tomography [published online April 26, 2019]. J Eur Acad Dermatol Venereol. doi:10.1111/jdv.15641.
- Navarrete-Dechent C, Mori S, Cordova M, et al. Reflectance confocal microscopy as a novel tool for presurgical identification of basal cell carcinoma biopsy site. J Am Acad Dermatol. 2019;80:e7-e8.
- Pan ZY, Lin JR, Cheng TT, et al. In vivo reflectance confocal microscopy of basal cell carcinoma: feasibility of preoperative mapping of cancer margins. Dermatol Surg. 2012;38:1945-1950.
- Venturini M, Gualdi G, Zanca A, et al. A new approach for presurgical margin assessment by reflectance confocal microscopy of basal cell carcinoma. Br J Dermatol. 2016;174:380-385.
- Sierra H, Yélamos O, Cordova M, et al. Reflectance confocal microscopy‐guided laser ablation of basal cell carcinomas: initial clinical experience. J Biomed Opt. 2017;22:1-13.
- Maier T, Kulichova D, Ruzicka T, et al. Noninvasive monitoring of basal cell carcinomas treated with systemic hedgehog inhibitors: pseudocysts as a sign of tumor regression. J Am Acad Dermatol. 2014;71:725-730.
- Levine A, Wang K, Markowitz O. Optical coherence tomography in the diagnosis of skin cancer. Dermatol Clin. 2017;35:465-488.
- Schneider SL, Kohli I, Hamzavi IH, et al. Emerging imaging technologies in dermatology: part I: basic principles. J Am Acad Dermatol. 2019;80:1114-1120.
- Mogensen M, Joergensen TM, Nümberg BM, et al. Assessment of optical coherence tomography imaging in the diagnosis of non‐melanoma skin cancer and benign lesions versus normal skin: observer‐blinded evaluation by dermatologists and pathologists. Dermatol Surg. 2009;35:965-972.
- Ferrante di Ruffano L, Dinnes J, Deeks JJ, et al. Optical coherence tomography for diagnosing skin cancer in adults. Cochrane Database Syst Rev. 2018;12:CD013189.
- Ulrich M, von Braunmuehl T, Kurzen H, et al. The sensitivity and specificity of optical coherence tomography for the assisted diagnosis of nonpigmented basal cell carcinoma: an observational study. Br J Dermatol. 2015;173:428-435.
- Wessels R, de Bruin DM, Relyveld GM, et al. Functional optical coherence tomography of pigmented lesions. J Eur Acad Dermatol Venereol. 2015;29:738‐744.
- Gambichler T, Schmid-Wendtner MH, Plura I, et al. A multicentre pilot study investigating high‐definition optical coherence tomography in the differentiation of cutaneous melanoma and melanocytic naevi. J Eur Acad Dermatol Venereol. 2015;29:537‐541.
- Marneffe A, Suppa M, Miyamoto M, et al. Validation of a diagnostic algorithm for the discrimination of actinic keratosis from normal skin and squamous cell carcinoma by means of high-definition optical coherence tomography. Exp Dermatol. 2016;25:684-687.
- Boone MA, Suppa M, Dhaenens F, et al. In vivo assessment of optical properties of melanocytic skin lesions and differentiation of melanoma from non-malignant lesions by high-definition optical coherence tomography. Arch Dermatol Res. 2016;308:7-20.
- Boone MA, Suppa M, Marneffe A, et al. A new algorithm for the discrimination of actinic keratosis from normal skin and squamous cell carcinoma based on in vivo analysis of optical properties by high-definition optical coherence tomography. J Eur Acad Dermatol Venereol. 2016;30:1714-1725.
- Themstrup L, Pellacani G, Welzel J, et al. In vivo microvascular imaging of cutaneous actinic keratosis, Bowen’s disease and squamous cell carcinoma using dynamic optical coherence tomography. J Eur Acad Dermatol Venereol. 2017;31:1655-1662.
- Alex A, Weingast J, Weinigel M, et al. Three-dimensional multiphoton/optical coherence tomography for diagnostic applications in dermatology. J Biophotonics. 2013;6:352-362.
- Iftimia N, Yélamos O, Chen CJ, et al. Handheld optical coherence tomography-reflectance confocal microscopy probe for detection of basal cell carcinoma and delineation of margins. J Biomed Opt. 2017;22:76006.
- Wang KX, Meekings A, Fluhr JW, et al. Optical coherence tomography-based optimization of Mohs micrographic surgery of basal cell carcinoma: a pilot study. Dermatol Surg. 2013;39:627-633.
- Chan CS, Rohrer TE. Optical coherence tomography and its role in Mohs micrographic surgery: a case report. Case Rep Dermatol. 2012;4:269-274.
- Gambichler T, Jaedicke V, Terras S. Optical coherence tomography in dermatology: technical and clinical aspects. Arch Dermatol Res. 2011;303:457-473.
- Manfredini M, Greco M, Farnetani F, et al. Acne: morphologic and vascular study of lesions and surrounding skin by means of optical coherence tomography. J Eur Acad Dermatol Venereol. 2017;31:1541-1546.
Traditionally, diagnosis of skin disease relies on clinical inspection, often followed by biopsy and histopathologic examination. In recent years, new noninvasive tools have emerged that can aid in clinical diagnosis and reduce the number of unnecessary benign biopsies. Although there has been a surge in noninvasive diagnostic technologies, many tools are still in research and development phases, with few tools widely adopted and used in regular clinical practice. In this article, we discuss the use of dermoscopy, reflectance confocal microscopy (RCM), and optical coherence tomography (OCT) in the diagnosis and management of skin disease.
Dermoscopy
Dermoscopy, also known as epiluminescence light microscopy and previously known as dermatoscopy, utilizes a ×10 to ×100 microscope objective with a light source to magnify and visualize structures present below the skin’s surface, such as melanin and blood vessels. There are 3 types of dermoscopy: conventional nonpolarized dermoscopy, polarized contact dermoscopy, and nonpolarized contact dermoscopy (Figure 1). Traditional nonpolarized dermoscopy requires a liquid medium and direct contact with the skin, and it relies on light reflection and refraction properties.1 Cross-polarized light sources allow visualization of deeper structures, either with or without a liquid medium and contact with the skin surface. Although there is overall concurrence among the different types of dermoscopy, subtle differences in the appearance of color, features, and structure are present.1

Dermoscopy offers many benefits for dermatologists and other providers. It can be used to aid in the diagnosis of cutaneous neoplasms and other skin diseases. Numerous low-cost dermatoscopes currently are commercially available. The handheld, easily transportable nature of dermatoscopes have resulted in widespread practice integration. Approximately 84% of attending dermatologists in US academic settings reported using dermoscopy, and many refer to the dermatoscope as “the dermatologist’s stethoscope.”2 In addition, 6% to 15% of other US providers, including family physicians, internal medicine physicians, and plastic surgeons, have reported using dermoscopy in their clinical practices. Limitations of dermoscopy include visualization of the skin surface only and not deeper structures within the tissue, the need for training for adequate interpretation of dermoscopic images, and lack of reimbursement for dermoscopic examination.3
Many dermoscopic structures that correspond well with histopathology have been described. Dermoscopy has a sensitivity of 79% to 96% and specificity of 69% to 99% in the diagnosis of melanoma.4 There is variable data on the specificity of dermoscopy in the diagnosis of melanoma, with one meta-analysis finding no statistically significant difference in specificity compared to naked eye examination,5 while other studies report increased specificity and subsequent reduction in biopsy of benign lesions.6,7 Dermoscopy also can aid in the diagnosis of keratinocytic neoplasms, and dermoscopy also results in a sensitivity of 78.6% to 100% and a specificity of 53.8% to 100% in the diagnosis of basal cell carcinoma (BCC).8 Limitations of dermoscopy include false-positive diagnoses, commonly seborrheic keratoses and nevi, resulting in unnecessary biopsies, as well as false-negative diagnoses, commonly amelanotic and nevoid melanoma, resulting in delays in skin cancer diagnosis and resultant poor outcomes.9 Dermoscopy also is used to aid in the diagnosis of inflammatory and infectious skin diseases, as well as scalp, hair, and nail disorders.10
Reflectance Confocal Microscopy
Reflectance confocal microscopy utilizes an 830-nm laser to capture horizontal en face images of the skin with high resolution. Different structures of the skin have varying indices of refraction: keratin, melanin, and collagen appear bright white, while other components appear dark, generating black-and-white RCM images.11 Currently, there are 2 reflectance confocal microscopes that are commercially available in the United States. The Vivascope 1500 (Caliber ID) is the traditional model that captures 8×8-mm images, and the Vivascope 3000 (Caliber ID) is a smaller handheld model that captures 0.5×0.5-mm images. The traditional model provides the advantages of higher-resolution images and the ability to capture larger surface areas but is best suited to image flat areas of skin to which a square window can be adhered. The handheld model allows improved contact with the varying topography of skin; does not require an adhesive window; and can be used to image cartilaginous, mucosal, and sensitive surfaces. However, it can be difficult to correlate individual images captured by the handheld RCM with the location relative to the lesion, as it is exquisitely sensitive to motion and also is operator dependent. Although complex algorithms are under development to stitch individual images to provide better correlation with the geography of the lesion, such programs are not yet widely available.12
Reflectance confocal microscopy affords many benefits for patients and providers. It is noninvasive and painless and is capable of imaging in vivo live skin as compared to clinical examination and dermoscopy, which only allow for visualization of the skin’s surface. Reflectance confocal microscopy also is time efficient, as imaging of a single lesion can be completed in 10 to 15 minutes. This technology generates high-resolution images, and RCM diagnosis has consistently demonstrated high sensitivity and specificity when compared to histopathology.13 Additionally, RCM imaging can spare biopsy and resultant scarring on cosmetically sensitive areas. Recently, RCM imaging of the skin has been granted Category I Current Procedural Terminology reimbursement codes that allow provider reimbursement and integration of RCM into daily practice14; however, private insurance coverage in the United States is variable. Limitations of RCM include a maximum depth of 200 to 300 µm, high cost to procure a reflectance confocal microscope, and the need for considerable training and practice to accurately interpret grayscale en face images.15
There has been extensive research regarding the use of RCM in the evaluation of cutaneous neoplasms and other skin diseases. Numerous features and patterns have been identified and described that correspond with different skin diseases and correspond well with histopathology (Figure 2).13,16,17 Reflectance confocal microscopy has demonstrated consistently high accuracy in the diagnosis of melanocytic lesions, with a sensitivity of 93% to 100% and a specificity of 75% to 99%.18-21 Reflectance confocal microscopy is especially useful in the evaluation of clinically or dermoscopically equivocal pigmented lesions due to greater specificity, resulting in a reduction of unnecessary biopsies.22,23 It also has high accuracy in the diagnosis of keratinocytic neoplasms, with a sensitivity of 82% to 100% and a specificity of 78% to 97% in the diagnosis of BCC,24 and a sensitivity of 74% to 100% and specificity of 78% to 100% in the diagnosis of squamous cell carcinoma (SCC).25,26 Evaluation of SCC and actinic keratosis (AK) using RCM may be limited by considerable hyperkeratosis and ulceration. In addition, it can be challenging to differentiate AK and SCC on RCM, and considerable expertise is required to accurately grade cytologic and architectural atypia.27 However, RCM has been used to discriminate between in situ and invasive proliferations.28 Reflectance confocal microscopy has wide applications in the diagnosis and management of cutaneous infections29,30 and inflammatory skin diseases.29,31-33 Recent RCM research explored the use of RCM to identify biopsy sites,34 delineate presurgical tumor margins,35,36 and monitor response to noninvasive treatments.37,38

Optical Coherence Tomography
Optical coherence tomography is an imaging modality that utilizes light backscatter from infrared light to produce grayscale cross-sectional or vertical images and horizontal en face images.39 Optical coherence tomography can visualize structures in the epidermis, dermoepidermal junction, and upper dermis.40 It can image boundaries of structures but cannot visualize individual cells.
There are different types of OCT devices available, including frequency-domain OCT (FD-OCT), or conventional OCT, and high-definition OCT (HD-OCT). With FD-OCT, images are captured at a maximum depth of 1 to 2 mm but with limited resolution. High-definition OCT has superior resolution compared to FD-OCT but is restricted to a shallower depth of 750 μm.39 The main advantage of OCT is the ability to noninvasively image live tissue and visualize 2- to 5-times greater depth as compared to RCM. Several OCT devices have obtained US Food and Drug Administration approval; however, OCT has not been widely adopted into clinical practice and is available only in tertiary academic centers. Additionally, OCT imaging in dermatology is rarely reimbursed. Other limitations of OCT include poor resolution of images, high cost to procure an OCT device, and the need for advanced training and experience to accurately interpret images.40,41
Optical coherence tomography primarily is used to diagnose cutaneous neoplasms. The best evidence of the diagnostic accuracy of OCT is in the setting of BCC, with a recent systematic review reporting a sensitivity of 66% to 96% and a specificity of 75% to 86% for conventional FD-OCT.42 The use of FD-OCT results in an increase in specificity without a significant change in sensitivity when compared to dermoscopy in the diagnosis of BCC.43 Melanoma is difficult to diagnose via FD-OCT, as the visualization of architectural features often is limited by poor resolution.44 A study of HD-OCT in the diagnosis of melanoma with a limited sample size reported a sensitivity of 74% to 80% and a specificity of 92% to 93%.45 Similarly, a study of HD-OCT used in the diagnosis of AK and SCC revealed a sensitivity and specificity of 81.6% and 92.6%, respectively, for AK and 93.8% and 98.9%, respectively, for SCC.46
Numerous algorithms and scoring systems have been developed to further explore the utility of OCT in the diagnosis of cutaneous neoplasms.47,48 Recent research investigated the utility of dynamic OCT, which can evaluate microvasculature in the diagnosis of cutaneous neoplasms (Figure 3)49; the combination of OCT with other imaging modalities50,51; the use of OCT to delineate presurgical margins52,53; and the role of OCT in the diagnosis and monitoring of inflammatory and infectious skin diseases.54,55

Final Thoughts
In recent years, there has been a surge of interest in noninvasive techniques for diagnosis and management of skin diseases; however, noninvasive tools exist on a spectrum in dermatology. Dermoscopy provides low-cost imaging of the skin’s surface and has been widely adopted by dermatologists and other providers to aid in clinical diagnosis. Reflectance confocal microscopy provides reimbursable in vivo imaging of live tissue with cellular-level resolution but is limited by depth, cost, and need for advanced training; thus, RCM has only been adopted in some clinical practices. Optical coherence tomography offers in vivo imaging of live tissue with substantial depth but poor resolution, high cost, need for advanced training, and rare reimbursement for providers. Future directions include combination of complementary imaging modalities, increased clinical practice integration, and education and reimbursement for providers.
Traditionally, diagnosis of skin disease relies on clinical inspection, often followed by biopsy and histopathologic examination. In recent years, new noninvasive tools have emerged that can aid in clinical diagnosis and reduce the number of unnecessary benign biopsies. Although there has been a surge in noninvasive diagnostic technologies, many tools are still in research and development phases, with few tools widely adopted and used in regular clinical practice. In this article, we discuss the use of dermoscopy, reflectance confocal microscopy (RCM), and optical coherence tomography (OCT) in the diagnosis and management of skin disease.
Dermoscopy
Dermoscopy, also known as epiluminescence light microscopy and previously known as dermatoscopy, utilizes a ×10 to ×100 microscope objective with a light source to magnify and visualize structures present below the skin’s surface, such as melanin and blood vessels. There are 3 types of dermoscopy: conventional nonpolarized dermoscopy, polarized contact dermoscopy, and nonpolarized contact dermoscopy (Figure 1). Traditional nonpolarized dermoscopy requires a liquid medium and direct contact with the skin, and it relies on light reflection and refraction properties.1 Cross-polarized light sources allow visualization of deeper structures, either with or without a liquid medium and contact with the skin surface. Although there is overall concurrence among the different types of dermoscopy, subtle differences in the appearance of color, features, and structure are present.1

Dermoscopy offers many benefits for dermatologists and other providers. It can be used to aid in the diagnosis of cutaneous neoplasms and other skin diseases. Numerous low-cost dermatoscopes currently are commercially available. The handheld, easily transportable nature of dermatoscopes have resulted in widespread practice integration. Approximately 84% of attending dermatologists in US academic settings reported using dermoscopy, and many refer to the dermatoscope as “the dermatologist’s stethoscope.”2 In addition, 6% to 15% of other US providers, including family physicians, internal medicine physicians, and plastic surgeons, have reported using dermoscopy in their clinical practices. Limitations of dermoscopy include visualization of the skin surface only and not deeper structures within the tissue, the need for training for adequate interpretation of dermoscopic images, and lack of reimbursement for dermoscopic examination.3
Many dermoscopic structures that correspond well with histopathology have been described. Dermoscopy has a sensitivity of 79% to 96% and specificity of 69% to 99% in the diagnosis of melanoma.4 There is variable data on the specificity of dermoscopy in the diagnosis of melanoma, with one meta-analysis finding no statistically significant difference in specificity compared to naked eye examination,5 while other studies report increased specificity and subsequent reduction in biopsy of benign lesions.6,7 Dermoscopy also can aid in the diagnosis of keratinocytic neoplasms, and dermoscopy also results in a sensitivity of 78.6% to 100% and a specificity of 53.8% to 100% in the diagnosis of basal cell carcinoma (BCC).8 Limitations of dermoscopy include false-positive diagnoses, commonly seborrheic keratoses and nevi, resulting in unnecessary biopsies, as well as false-negative diagnoses, commonly amelanotic and nevoid melanoma, resulting in delays in skin cancer diagnosis and resultant poor outcomes.9 Dermoscopy also is used to aid in the diagnosis of inflammatory and infectious skin diseases, as well as scalp, hair, and nail disorders.10
Reflectance Confocal Microscopy
Reflectance confocal microscopy utilizes an 830-nm laser to capture horizontal en face images of the skin with high resolution. Different structures of the skin have varying indices of refraction: keratin, melanin, and collagen appear bright white, while other components appear dark, generating black-and-white RCM images.11 Currently, there are 2 reflectance confocal microscopes that are commercially available in the United States. The Vivascope 1500 (Caliber ID) is the traditional model that captures 8×8-mm images, and the Vivascope 3000 (Caliber ID) is a smaller handheld model that captures 0.5×0.5-mm images. The traditional model provides the advantages of higher-resolution images and the ability to capture larger surface areas but is best suited to image flat areas of skin to which a square window can be adhered. The handheld model allows improved contact with the varying topography of skin; does not require an adhesive window; and can be used to image cartilaginous, mucosal, and sensitive surfaces. However, it can be difficult to correlate individual images captured by the handheld RCM with the location relative to the lesion, as it is exquisitely sensitive to motion and also is operator dependent. Although complex algorithms are under development to stitch individual images to provide better correlation with the geography of the lesion, such programs are not yet widely available.12
Reflectance confocal microscopy affords many benefits for patients and providers. It is noninvasive and painless and is capable of imaging in vivo live skin as compared to clinical examination and dermoscopy, which only allow for visualization of the skin’s surface. Reflectance confocal microscopy also is time efficient, as imaging of a single lesion can be completed in 10 to 15 minutes. This technology generates high-resolution images, and RCM diagnosis has consistently demonstrated high sensitivity and specificity when compared to histopathology.13 Additionally, RCM imaging can spare biopsy and resultant scarring on cosmetically sensitive areas. Recently, RCM imaging of the skin has been granted Category I Current Procedural Terminology reimbursement codes that allow provider reimbursement and integration of RCM into daily practice14; however, private insurance coverage in the United States is variable. Limitations of RCM include a maximum depth of 200 to 300 µm, high cost to procure a reflectance confocal microscope, and the need for considerable training and practice to accurately interpret grayscale en face images.15
There has been extensive research regarding the use of RCM in the evaluation of cutaneous neoplasms and other skin diseases. Numerous features and patterns have been identified and described that correspond with different skin diseases and correspond well with histopathology (Figure 2).13,16,17 Reflectance confocal microscopy has demonstrated consistently high accuracy in the diagnosis of melanocytic lesions, with a sensitivity of 93% to 100% and a specificity of 75% to 99%.18-21 Reflectance confocal microscopy is especially useful in the evaluation of clinically or dermoscopically equivocal pigmented lesions due to greater specificity, resulting in a reduction of unnecessary biopsies.22,23 It also has high accuracy in the diagnosis of keratinocytic neoplasms, with a sensitivity of 82% to 100% and a specificity of 78% to 97% in the diagnosis of BCC,24 and a sensitivity of 74% to 100% and specificity of 78% to 100% in the diagnosis of squamous cell carcinoma (SCC).25,26 Evaluation of SCC and actinic keratosis (AK) using RCM may be limited by considerable hyperkeratosis and ulceration. In addition, it can be challenging to differentiate AK and SCC on RCM, and considerable expertise is required to accurately grade cytologic and architectural atypia.27 However, RCM has been used to discriminate between in situ and invasive proliferations.28 Reflectance confocal microscopy has wide applications in the diagnosis and management of cutaneous infections29,30 and inflammatory skin diseases.29,31-33 Recent RCM research explored the use of RCM to identify biopsy sites,34 delineate presurgical tumor margins,35,36 and monitor response to noninvasive treatments.37,38

Optical Coherence Tomography
Optical coherence tomography is an imaging modality that utilizes light backscatter from infrared light to produce grayscale cross-sectional or vertical images and horizontal en face images.39 Optical coherence tomography can visualize structures in the epidermis, dermoepidermal junction, and upper dermis.40 It can image boundaries of structures but cannot visualize individual cells.
There are different types of OCT devices available, including frequency-domain OCT (FD-OCT), or conventional OCT, and high-definition OCT (HD-OCT). With FD-OCT, images are captured at a maximum depth of 1 to 2 mm but with limited resolution. High-definition OCT has superior resolution compared to FD-OCT but is restricted to a shallower depth of 750 μm.39 The main advantage of OCT is the ability to noninvasively image live tissue and visualize 2- to 5-times greater depth as compared to RCM. Several OCT devices have obtained US Food and Drug Administration approval; however, OCT has not been widely adopted into clinical practice and is available only in tertiary academic centers. Additionally, OCT imaging in dermatology is rarely reimbursed. Other limitations of OCT include poor resolution of images, high cost to procure an OCT device, and the need for advanced training and experience to accurately interpret images.40,41
Optical coherence tomography primarily is used to diagnose cutaneous neoplasms. The best evidence of the diagnostic accuracy of OCT is in the setting of BCC, with a recent systematic review reporting a sensitivity of 66% to 96% and a specificity of 75% to 86% for conventional FD-OCT.42 The use of FD-OCT results in an increase in specificity without a significant change in sensitivity when compared to dermoscopy in the diagnosis of BCC.43 Melanoma is difficult to diagnose via FD-OCT, as the visualization of architectural features often is limited by poor resolution.44 A study of HD-OCT in the diagnosis of melanoma with a limited sample size reported a sensitivity of 74% to 80% and a specificity of 92% to 93%.45 Similarly, a study of HD-OCT used in the diagnosis of AK and SCC revealed a sensitivity and specificity of 81.6% and 92.6%, respectively, for AK and 93.8% and 98.9%, respectively, for SCC.46
Numerous algorithms and scoring systems have been developed to further explore the utility of OCT in the diagnosis of cutaneous neoplasms.47,48 Recent research investigated the utility of dynamic OCT, which can evaluate microvasculature in the diagnosis of cutaneous neoplasms (Figure 3)49; the combination of OCT with other imaging modalities50,51; the use of OCT to delineate presurgical margins52,53; and the role of OCT in the diagnosis and monitoring of inflammatory and infectious skin diseases.54,55

Final Thoughts
In recent years, there has been a surge of interest in noninvasive techniques for diagnosis and management of skin diseases; however, noninvasive tools exist on a spectrum in dermatology. Dermoscopy provides low-cost imaging of the skin’s surface and has been widely adopted by dermatologists and other providers to aid in clinical diagnosis. Reflectance confocal microscopy provides reimbursable in vivo imaging of live tissue with cellular-level resolution but is limited by depth, cost, and need for advanced training; thus, RCM has only been adopted in some clinical practices. Optical coherence tomography offers in vivo imaging of live tissue with substantial depth but poor resolution, high cost, need for advanced training, and rare reimbursement for providers. Future directions include combination of complementary imaging modalities, increased clinical practice integration, and education and reimbursement for providers.
- Benvenuto-Andrade C, Dusza SW, Agero AL, et al. Differences between polarized light dermoscopy and immersion contact dermoscopy for the evaluation of skin lesions. Arch Dermatol. 2007;143:329-338.
- Terushkin V, Oliveria SA, Marghoob AA, et al. Use of and beliefs about total body photography and dermatoscopy among US dermatology training programs: an update. J Am Acad Dermatol. 2010;62:794-803.
- Morris JB, Alfonso SV, Hernandez N, et al. Use of and intentions to use dermoscopy among physicians in the United States. Dermatol Pract Concept. 2017;7:7-16.
- Yélamos O, Braun RP, Liopyris K, et al. Dermoscopy and dermatopathology correlates of cutaneous neoplasms. J Am Acad Dermatol. 2019;80:341-363.
- Vestergaard ME, Macaskill P, Holt PE, et al. Dermoscopy compared with naked eye examination for the diagnosis of primary melanoma: a meta-analysis of studies performed in a clinical setting. Br J Dermatol. 2008;159:669-676.
- Carli P, de Giorgi V, Chiarugi A, et al. Addition of dermoscopy to conventional naked-eye examination in melanoma screening: a randomized study. J Am Acad Dermatol. 2004;50:683-668.
- Lallas A, Zalaudek I, Argenziano G, et al. Dermoscopy in general dermatology. Dermatol Clin. 2013;31:679-694.
- Reiter O, Mimouni I, Gdalvevich M, et al. The diagnostic accuracy of dermoscopy for basal cell carcinoma: a systematic review and meta-analysis. J Am Acad Dermatol. 2019;80:1380-1388.
- Papageorgiou V, Apalla Z, Sotiriou E, et al. The limitations of dermoscopy: false-positive and false-negative tumours. J Eur Acad Dermatol Venereol. 2018;32:879-888.
- Micali G, Verzì AE, Lacarrubba F. Alternative uses of dermoscopy in daily clinical practice: an update. J Am Acad Dermatol. 2018;79:1117-1132.e1.
- Rajadhyaksha M, Grossman M, Esterowitz D, et al. In vivo confocal scanning laser microscopy of human skin: melanin provides strong contrast. J Invest Dermatol. 1995;104:946-952.
- Kose K, Gou M, Yélamos O, et al. Automated video-mosaicking approach for confocal microscopic imaging in vivo: an approach to address challenges in imaging living tissue and extend field of view. Sci Rep. 2017;7:10759.
- Rao BK, John AM, Francisco G, et al. Diagnostic accuracy of reflectance confocal microscopy for diagnosis of skin lesions [published online October 8, 2018]. Arch Pathol Lab Med. 2019;143:326-329.
- Current Procedural Terminology, Professional Edition. Chicago IL: American Medical Association; 2016. The preliminary physician fee schedule for 2017 is available at https://www.cms.gov/Medicare/Medicare-Fee-for-Service-Payment/PhysicianFeeSched/PFS-Federal-Regulation-Notices-Items/CMS-1654-P.html.
- Jain M, Pulijal SV, Rajadhyaksha M, et al. Evaluation of bedside diagnostic accuracy, learning curve, and challenges for a novice reflectance confocal microscopy reader for skin cancer detection in vivo. JAMA Dermatol. 2018;154:962-965.
- Rao BK, Pellacani G. Atlas of Confocal Microscopy in Dermatology: Clinical, Confocal, and Histological Images. New York, NY: NIDIskin LLC; 2013.
- Scope A, Benvenuto-Andrande C, Agero AL, et al. In vivo reflectance confocal microscopy imaging of melanocytic skin lesions: consensus terminology glossary and illustrative images. J Am Acad Dermatol. 2007;57:644-658.
- Gerger A, Hofmann-Wellenhof R, Langsenlehner U, et al. In vivo confocal laser scanning microscopy of melanocytic skin tumours: diagnostic applicability using unselected tumour images. Br J Dermatol. 2008;158:329-333.
- Stevenson AD, Mickan S, Mallett S, et al. Systematic review of diagnostic accuracy of reflectance confocal microscopy for melanoma diagnosis in patients with clinically equivocal skin lesions. Dermatol Pract Concept. 2013;3:19-27.
- Alarcon I, Carrera C, Palou J, et al. Impact of in vivo reflectance confocal microscopy on the number needed to treat melanoma in doubtful lesions. Br J Dermatol. 2014;170:802-808.
- Lovatto L, Carrera C, Salerni G, et al. In vivo reflectance confocal microscopy of equivocal melanocytic lesions detected by digital dermoscopy follow-up. J Eur Acad Dermatol Venereol. 2015;29:1918-1925.
- Guitera P, Pellacani G, Longo C, et al. In vivo reflectance confocal microscopy enhances secondary evaluation of melanocytic lesions. J Invest Dermatol. 2009;129:131-138.
- Xiong YQ, Ma SJ, Mo Y, et al. Comparison of dermoscopy and reflectance confocal microscopy for the diagnosis of malignant skin tumours: a meta-analysis. J Cancer Res Clin Oncol. 2017;143:1627-1635.
- Kadouch DJ, Schram ME, Leeflang MM, et al. In vivo confocal microscopy of basal cell carcinoma: a systematic review of diagnostic accuracy. J Eur Acad Dermatol Venereol. 2015;29:1890-1897.
- Dinnes J, Deeks JJ, Chuchu N, et al; Cochrane Skin Cancer Diagnostic Test Accuracy Group. Reflectance confocal microscopy for diagnosing keratinocyte skin cancers in adults. Cochrane Database Syst Rev. 2018;12:CD013191.
- Nguyen KP, Peppelman M, Hoogedoorn L, et al. The current role of in vivo reflectance confocal microscopy within the continuum of actinic keratosis and squamous cell carcinoma: a systematic review. Eur J Dermatol. 2016;26:549-565.
- Pellacani G, Ulrich M, Casari A, et al. Grading keratinocyte atypia in actinic keratosis: a correlation of reflectance confocal microscopy and histopathology. J Eur Acad Dermatol Venereol. 2015;29:2216-2221.
- Manfredini M, Longo C, Ferrari B, et al. Dermoscopic and reflectance confocal microscopy features of cutaneous squamous cell carcinoma. J Eur Acad Dermatol Venereol. 2017;31:1828-1833.
- Hoogedoorn L, Peppelman M, van de Kerkhof PC, et al. The value of in vivo reflectance confocal microscopy in the diagnosis and monitoring of inflammatory and infectious skin diseases: a systematic review. Br J Dermatol. 2015;172:1222-1248.
- Cinotti E, Perrot JL, Labeille B, et al. Reflectance confocal microscopy for cutaneous infections and infestations. J Eur Acad Dermatol Venereol. 2016;30:754-763.
- Ardigo M, Longo C, Gonzalez S; International Confocal Working Group Inflammatory Skin Diseases Project. Multicentre study on inflammatory skin diseases from The International Confocal Working Group: specific confocal microscopy features and an algorithmic method of diagnosis. Br J Dermatol. 2016;175:364-374.
- Ardigo M, Agozzino M, Franceschini C, et al. Reflectance confocal microscopy algorithms for inflammatory and hair diseases. Dermatol Clin. 2016;34:487-496.
- Manfredini M, Bettoli V, Sacripanti G, et al. The evolution of healthy skin to acne lesions: a longitudinal, in vivo evaluation with reflectance confocal microscopy and optical coherence tomography [published online April 26, 2019]. J Eur Acad Dermatol Venereol. doi:10.1111/jdv.15641.
- Navarrete-Dechent C, Mori S, Cordova M, et al. Reflectance confocal microscopy as a novel tool for presurgical identification of basal cell carcinoma biopsy site. J Am Acad Dermatol. 2019;80:e7-e8.
- Pan ZY, Lin JR, Cheng TT, et al. In vivo reflectance confocal microscopy of basal cell carcinoma: feasibility of preoperative mapping of cancer margins. Dermatol Surg. 2012;38:1945-1950.
- Venturini M, Gualdi G, Zanca A, et al. A new approach for presurgical margin assessment by reflectance confocal microscopy of basal cell carcinoma. Br J Dermatol. 2016;174:380-385.
- Sierra H, Yélamos O, Cordova M, et al. Reflectance confocal microscopy‐guided laser ablation of basal cell carcinomas: initial clinical experience. J Biomed Opt. 2017;22:1-13.
- Maier T, Kulichova D, Ruzicka T, et al. Noninvasive monitoring of basal cell carcinomas treated with systemic hedgehog inhibitors: pseudocysts as a sign of tumor regression. J Am Acad Dermatol. 2014;71:725-730.
- Levine A, Wang K, Markowitz O. Optical coherence tomography in the diagnosis of skin cancer. Dermatol Clin. 2017;35:465-488.
- Schneider SL, Kohli I, Hamzavi IH, et al. Emerging imaging technologies in dermatology: part I: basic principles. J Am Acad Dermatol. 2019;80:1114-1120.
- Mogensen M, Joergensen TM, Nümberg BM, et al. Assessment of optical coherence tomography imaging in the diagnosis of non‐melanoma skin cancer and benign lesions versus normal skin: observer‐blinded evaluation by dermatologists and pathologists. Dermatol Surg. 2009;35:965-972.
- Ferrante di Ruffano L, Dinnes J, Deeks JJ, et al. Optical coherence tomography for diagnosing skin cancer in adults. Cochrane Database Syst Rev. 2018;12:CD013189.
- Ulrich M, von Braunmuehl T, Kurzen H, et al. The sensitivity and specificity of optical coherence tomography for the assisted diagnosis of nonpigmented basal cell carcinoma: an observational study. Br J Dermatol. 2015;173:428-435.
- Wessels R, de Bruin DM, Relyveld GM, et al. Functional optical coherence tomography of pigmented lesions. J Eur Acad Dermatol Venereol. 2015;29:738‐744.
- Gambichler T, Schmid-Wendtner MH, Plura I, et al. A multicentre pilot study investigating high‐definition optical coherence tomography in the differentiation of cutaneous melanoma and melanocytic naevi. J Eur Acad Dermatol Venereol. 2015;29:537‐541.
- Marneffe A, Suppa M, Miyamoto M, et al. Validation of a diagnostic algorithm for the discrimination of actinic keratosis from normal skin and squamous cell carcinoma by means of high-definition optical coherence tomography. Exp Dermatol. 2016;25:684-687.
- Boone MA, Suppa M, Dhaenens F, et al. In vivo assessment of optical properties of melanocytic skin lesions and differentiation of melanoma from non-malignant lesions by high-definition optical coherence tomography. Arch Dermatol Res. 2016;308:7-20.
- Boone MA, Suppa M, Marneffe A, et al. A new algorithm for the discrimination of actinic keratosis from normal skin and squamous cell carcinoma based on in vivo analysis of optical properties by high-definition optical coherence tomography. J Eur Acad Dermatol Venereol. 2016;30:1714-1725.
- Themstrup L, Pellacani G, Welzel J, et al. In vivo microvascular imaging of cutaneous actinic keratosis, Bowen’s disease and squamous cell carcinoma using dynamic optical coherence tomography. J Eur Acad Dermatol Venereol. 2017;31:1655-1662.
- Alex A, Weingast J, Weinigel M, et al. Three-dimensional multiphoton/optical coherence tomography for diagnostic applications in dermatology. J Biophotonics. 2013;6:352-362.
- Iftimia N, Yélamos O, Chen CJ, et al. Handheld optical coherence tomography-reflectance confocal microscopy probe for detection of basal cell carcinoma and delineation of margins. J Biomed Opt. 2017;22:76006.
- Wang KX, Meekings A, Fluhr JW, et al. Optical coherence tomography-based optimization of Mohs micrographic surgery of basal cell carcinoma: a pilot study. Dermatol Surg. 2013;39:627-633.
- Chan CS, Rohrer TE. Optical coherence tomography and its role in Mohs micrographic surgery: a case report. Case Rep Dermatol. 2012;4:269-274.
- Gambichler T, Jaedicke V, Terras S. Optical coherence tomography in dermatology: technical and clinical aspects. Arch Dermatol Res. 2011;303:457-473.
- Manfredini M, Greco M, Farnetani F, et al. Acne: morphologic and vascular study of lesions and surrounding skin by means of optical coherence tomography. J Eur Acad Dermatol Venereol. 2017;31:1541-1546.
- Benvenuto-Andrade C, Dusza SW, Agero AL, et al. Differences between polarized light dermoscopy and immersion contact dermoscopy for the evaluation of skin lesions. Arch Dermatol. 2007;143:329-338.
- Terushkin V, Oliveria SA, Marghoob AA, et al. Use of and beliefs about total body photography and dermatoscopy among US dermatology training programs: an update. J Am Acad Dermatol. 2010;62:794-803.
- Morris JB, Alfonso SV, Hernandez N, et al. Use of and intentions to use dermoscopy among physicians in the United States. Dermatol Pract Concept. 2017;7:7-16.
- Yélamos O, Braun RP, Liopyris K, et al. Dermoscopy and dermatopathology correlates of cutaneous neoplasms. J Am Acad Dermatol. 2019;80:341-363.
- Vestergaard ME, Macaskill P, Holt PE, et al. Dermoscopy compared with naked eye examination for the diagnosis of primary melanoma: a meta-analysis of studies performed in a clinical setting. Br J Dermatol. 2008;159:669-676.
- Carli P, de Giorgi V, Chiarugi A, et al. Addition of dermoscopy to conventional naked-eye examination in melanoma screening: a randomized study. J Am Acad Dermatol. 2004;50:683-668.
- Lallas A, Zalaudek I, Argenziano G, et al. Dermoscopy in general dermatology. Dermatol Clin. 2013;31:679-694.
- Reiter O, Mimouni I, Gdalvevich M, et al. The diagnostic accuracy of dermoscopy for basal cell carcinoma: a systematic review and meta-analysis. J Am Acad Dermatol. 2019;80:1380-1388.
- Papageorgiou V, Apalla Z, Sotiriou E, et al. The limitations of dermoscopy: false-positive and false-negative tumours. J Eur Acad Dermatol Venereol. 2018;32:879-888.
- Micali G, Verzì AE, Lacarrubba F. Alternative uses of dermoscopy in daily clinical practice: an update. J Am Acad Dermatol. 2018;79:1117-1132.e1.
- Rajadhyaksha M, Grossman M, Esterowitz D, et al. In vivo confocal scanning laser microscopy of human skin: melanin provides strong contrast. J Invest Dermatol. 1995;104:946-952.
- Kose K, Gou M, Yélamos O, et al. Automated video-mosaicking approach for confocal microscopic imaging in vivo: an approach to address challenges in imaging living tissue and extend field of view. Sci Rep. 2017;7:10759.
- Rao BK, John AM, Francisco G, et al. Diagnostic accuracy of reflectance confocal microscopy for diagnosis of skin lesions [published online October 8, 2018]. Arch Pathol Lab Med. 2019;143:326-329.
- Current Procedural Terminology, Professional Edition. Chicago IL: American Medical Association; 2016. The preliminary physician fee schedule for 2017 is available at https://www.cms.gov/Medicare/Medicare-Fee-for-Service-Payment/PhysicianFeeSched/PFS-Federal-Regulation-Notices-Items/CMS-1654-P.html.
- Jain M, Pulijal SV, Rajadhyaksha M, et al. Evaluation of bedside diagnostic accuracy, learning curve, and challenges for a novice reflectance confocal microscopy reader for skin cancer detection in vivo. JAMA Dermatol. 2018;154:962-965.
- Rao BK, Pellacani G. Atlas of Confocal Microscopy in Dermatology: Clinical, Confocal, and Histological Images. New York, NY: NIDIskin LLC; 2013.
- Scope A, Benvenuto-Andrande C, Agero AL, et al. In vivo reflectance confocal microscopy imaging of melanocytic skin lesions: consensus terminology glossary and illustrative images. J Am Acad Dermatol. 2007;57:644-658.
- Gerger A, Hofmann-Wellenhof R, Langsenlehner U, et al. In vivo confocal laser scanning microscopy of melanocytic skin tumours: diagnostic applicability using unselected tumour images. Br J Dermatol. 2008;158:329-333.
- Stevenson AD, Mickan S, Mallett S, et al. Systematic review of diagnostic accuracy of reflectance confocal microscopy for melanoma diagnosis in patients with clinically equivocal skin lesions. Dermatol Pract Concept. 2013;3:19-27.
- Alarcon I, Carrera C, Palou J, et al. Impact of in vivo reflectance confocal microscopy on the number needed to treat melanoma in doubtful lesions. Br J Dermatol. 2014;170:802-808.
- Lovatto L, Carrera C, Salerni G, et al. In vivo reflectance confocal microscopy of equivocal melanocytic lesions detected by digital dermoscopy follow-up. J Eur Acad Dermatol Venereol. 2015;29:1918-1925.
- Guitera P, Pellacani G, Longo C, et al. In vivo reflectance confocal microscopy enhances secondary evaluation of melanocytic lesions. J Invest Dermatol. 2009;129:131-138.
- Xiong YQ, Ma SJ, Mo Y, et al. Comparison of dermoscopy and reflectance confocal microscopy for the diagnosis of malignant skin tumours: a meta-analysis. J Cancer Res Clin Oncol. 2017;143:1627-1635.
- Kadouch DJ, Schram ME, Leeflang MM, et al. In vivo confocal microscopy of basal cell carcinoma: a systematic review of diagnostic accuracy. J Eur Acad Dermatol Venereol. 2015;29:1890-1897.
- Dinnes J, Deeks JJ, Chuchu N, et al; Cochrane Skin Cancer Diagnostic Test Accuracy Group. Reflectance confocal microscopy for diagnosing keratinocyte skin cancers in adults. Cochrane Database Syst Rev. 2018;12:CD013191.
- Nguyen KP, Peppelman M, Hoogedoorn L, et al. The current role of in vivo reflectance confocal microscopy within the continuum of actinic keratosis and squamous cell carcinoma: a systematic review. Eur J Dermatol. 2016;26:549-565.
- Pellacani G, Ulrich M, Casari A, et al. Grading keratinocyte atypia in actinic keratosis: a correlation of reflectance confocal microscopy and histopathology. J Eur Acad Dermatol Venereol. 2015;29:2216-2221.
- Manfredini M, Longo C, Ferrari B, et al. Dermoscopic and reflectance confocal microscopy features of cutaneous squamous cell carcinoma. J Eur Acad Dermatol Venereol. 2017;31:1828-1833.
- Hoogedoorn L, Peppelman M, van de Kerkhof PC, et al. The value of in vivo reflectance confocal microscopy in the diagnosis and monitoring of inflammatory and infectious skin diseases: a systematic review. Br J Dermatol. 2015;172:1222-1248.
- Cinotti E, Perrot JL, Labeille B, et al. Reflectance confocal microscopy for cutaneous infections and infestations. J Eur Acad Dermatol Venereol. 2016;30:754-763.
- Ardigo M, Longo C, Gonzalez S; International Confocal Working Group Inflammatory Skin Diseases Project. Multicentre study on inflammatory skin diseases from The International Confocal Working Group: specific confocal microscopy features and an algorithmic method of diagnosis. Br J Dermatol. 2016;175:364-374.
- Ardigo M, Agozzino M, Franceschini C, et al. Reflectance confocal microscopy algorithms for inflammatory and hair diseases. Dermatol Clin. 2016;34:487-496.
- Manfredini M, Bettoli V, Sacripanti G, et al. The evolution of healthy skin to acne lesions: a longitudinal, in vivo evaluation with reflectance confocal microscopy and optical coherence tomography [published online April 26, 2019]. J Eur Acad Dermatol Venereol. doi:10.1111/jdv.15641.
- Navarrete-Dechent C, Mori S, Cordova M, et al. Reflectance confocal microscopy as a novel tool for presurgical identification of basal cell carcinoma biopsy site. J Am Acad Dermatol. 2019;80:e7-e8.
- Pan ZY, Lin JR, Cheng TT, et al. In vivo reflectance confocal microscopy of basal cell carcinoma: feasibility of preoperative mapping of cancer margins. Dermatol Surg. 2012;38:1945-1950.
- Venturini M, Gualdi G, Zanca A, et al. A new approach for presurgical margin assessment by reflectance confocal microscopy of basal cell carcinoma. Br J Dermatol. 2016;174:380-385.
- Sierra H, Yélamos O, Cordova M, et al. Reflectance confocal microscopy‐guided laser ablation of basal cell carcinomas: initial clinical experience. J Biomed Opt. 2017;22:1-13.
- Maier T, Kulichova D, Ruzicka T, et al. Noninvasive monitoring of basal cell carcinomas treated with systemic hedgehog inhibitors: pseudocysts as a sign of tumor regression. J Am Acad Dermatol. 2014;71:725-730.
- Levine A, Wang K, Markowitz O. Optical coherence tomography in the diagnosis of skin cancer. Dermatol Clin. 2017;35:465-488.
- Schneider SL, Kohli I, Hamzavi IH, et al. Emerging imaging technologies in dermatology: part I: basic principles. J Am Acad Dermatol. 2019;80:1114-1120.
- Mogensen M, Joergensen TM, Nümberg BM, et al. Assessment of optical coherence tomography imaging in the diagnosis of non‐melanoma skin cancer and benign lesions versus normal skin: observer‐blinded evaluation by dermatologists and pathologists. Dermatol Surg. 2009;35:965-972.
- Ferrante di Ruffano L, Dinnes J, Deeks JJ, et al. Optical coherence tomography for diagnosing skin cancer in adults. Cochrane Database Syst Rev. 2018;12:CD013189.
- Ulrich M, von Braunmuehl T, Kurzen H, et al. The sensitivity and specificity of optical coherence tomography for the assisted diagnosis of nonpigmented basal cell carcinoma: an observational study. Br J Dermatol. 2015;173:428-435.
- Wessels R, de Bruin DM, Relyveld GM, et al. Functional optical coherence tomography of pigmented lesions. J Eur Acad Dermatol Venereol. 2015;29:738‐744.
- Gambichler T, Schmid-Wendtner MH, Plura I, et al. A multicentre pilot study investigating high‐definition optical coherence tomography in the differentiation of cutaneous melanoma and melanocytic naevi. J Eur Acad Dermatol Venereol. 2015;29:537‐541.
- Marneffe A, Suppa M, Miyamoto M, et al. Validation of a diagnostic algorithm for the discrimination of actinic keratosis from normal skin and squamous cell carcinoma by means of high-definition optical coherence tomography. Exp Dermatol. 2016;25:684-687.
- Boone MA, Suppa M, Dhaenens F, et al. In vivo assessment of optical properties of melanocytic skin lesions and differentiation of melanoma from non-malignant lesions by high-definition optical coherence tomography. Arch Dermatol Res. 2016;308:7-20.
- Boone MA, Suppa M, Marneffe A, et al. A new algorithm for the discrimination of actinic keratosis from normal skin and squamous cell carcinoma based on in vivo analysis of optical properties by high-definition optical coherence tomography. J Eur Acad Dermatol Venereol. 2016;30:1714-1725.
- Themstrup L, Pellacani G, Welzel J, et al. In vivo microvascular imaging of cutaneous actinic keratosis, Bowen’s disease and squamous cell carcinoma using dynamic optical coherence tomography. J Eur Acad Dermatol Venereol. 2017;31:1655-1662.
- Alex A, Weingast J, Weinigel M, et al. Three-dimensional multiphoton/optical coherence tomography for diagnostic applications in dermatology. J Biophotonics. 2013;6:352-362.
- Iftimia N, Yélamos O, Chen CJ, et al. Handheld optical coherence tomography-reflectance confocal microscopy probe for detection of basal cell carcinoma and delineation of margins. J Biomed Opt. 2017;22:76006.
- Wang KX, Meekings A, Fluhr JW, et al. Optical coherence tomography-based optimization of Mohs micrographic surgery of basal cell carcinoma: a pilot study. Dermatol Surg. 2013;39:627-633.
- Chan CS, Rohrer TE. Optical coherence tomography and its role in Mohs micrographic surgery: a case report. Case Rep Dermatol. 2012;4:269-274.
- Gambichler T, Jaedicke V, Terras S. Optical coherence tomography in dermatology: technical and clinical aspects. Arch Dermatol Res. 2011;303:457-473.
- Manfredini M, Greco M, Farnetani F, et al. Acne: morphologic and vascular study of lesions and surrounding skin by means of optical coherence tomography. J Eur Acad Dermatol Venereol. 2017;31:1541-1546.
Practice Points
- There are several new noninvasive imaging tools in dermatology that can be utilized to aid in the diagnosis and management of skin disease, including dermoscopy, reflectance confocal microscopy, and optical coherence tomography.
- Among these tools, there are several differences in cost, clinical integration, reimbursement, and accuracy.
Darkening and Eruptive Nevi During Treatment With Erlotinib
To the Editor:
Erlotinib is a small-molecule selective tyrosine kinase inhibitor that functions by blocking the intracellular portion of the epidermal growth factor receptor (EGFR)1,2; EGFR normally is expressed in the basal layer of the epidermis, sweat glands, and hair follicles, and is overexpressed in some cancers.1,3 Normal activation of EGFR leads to signal transduction through the mitogen-activated protein kinase (MAPK) signaling pathway, which stimulates cell survival and proliferation.4,5 Erlotinib-induced inhibition of EGFR prevents tyrosine kinase phosphorylation and aims to decrease cell proliferation in these tumors.
Erlotinib is indicated as once-daily oral monotherapy for the treatment of advanced-stage non–small cell lung cancer (NSCLCA) and in combination with gemcitabine for treatment of advanced-stage pancreatic cancer.1 A number of cutaneous side effects have been reported, including acneform eruption, xerosis, paronychia, and pruritus.6 Other tyrosine kinase inhibitors, which also decrease signal transduction through the MAPK pathway, have some overlapping side effects; among these are vemurafenib, a selective BRAF inhibitor, and sorafenib, a multikinase inhibitor.7,8
A 70-year-old man with NSCLCA presented with eruptive nevi and darkening of existing nevi 3 months after starting monotherapy with erlotinib. Physical examination demonstrated the simultaneous appearance of scattered acneform papules and pustules; diffuse xerosis; and numerous dark brown to black nevi on the trunk, arms, and legs. Compared to prior clinical photographs taken in our office, darkening of existing medium brown nevi was noted, and new nevi developed in areas where no prior nevi had been visible (Figure 1).
The patient’s medical history included 3 invasive melanomas, all of which were diagnosed at least 7 years prior to the initiation of erlotinib and were treated by surgical excision alone. Prior treatment of NSCLCA consisted of a left lower lobectomy followed by docetaxel, carboplatin, pegfilgrastim, dexamethasone, and pemetrexed. A thorough review of all of the patient’s medications revealed no associations with changes in nevi.
A review of the patient’s treatment timeline revealed that all other chemotherapeutic medications had been discontinued a minimum of 5 weeks before starting erlotinib. A complete cutaneous examination performed in our office after completion of these chemotherapeutic agents and prior to initiation of erlotinib was unremarkable for abnormally dark or eruptive nevi.
Since starting erlotinib treatment, the patient underwent 10 biopsies of clinically suspicious dark nevi performed by a dermatologist in our office. Two of these were diagnosed as melanoma in situ and one as an atypical nevus. A temporal association of the darkening and eruptive nevi with erlotinib treatment was established; however, because erlotinib was essential to his NSCLCA treatment, he continued erlotinib with frequent complete cutaneous examinations.
A number of cutaneous side effects have been described during treatment with erlotinib, the most common being acneform eruption.6 The incidence and severity of acneform eruptions have been positively correlated to survival in patients with NSCLCA.3,5,6 Other common side effects include xerosis, paronychia, and pruritus.1,5,6 Less common side effects include periungual pyogenic granulomas and hair growth abnormalities.1
Eruptive nevi previously were reported in a patient who was treated with erlotinib.1 Other tyrosine kinase inhibitors that also decrease signal transduction through the MAPK pathway, including sorafenib and vemurafenib, have been reported to cause eruptive nevi. There are 7 reports of eruptive nevi with sorafenib and 5 reports with vemurafenib.7-9 Development of nevi were noted within a few months of initiating treatment with these medications.7
A PubMed search of articles indexed for MEDLINE using the terms erlotinib and melanoma and erlotinib and nevi yielded no prior reports of darkening of existing nevi or the development of melanoma during treatment with erlotinib. However, vemurafenib has been reported to cause dysplastic nevi, melanomas, and darkening of existing nevi, in addition to eruptive nevi.8-10 The side effects of vemurafenib have been ascribed to a paradoxical upregulation of MAPK in BRAF wild-type cells. This effect has been well documented and demonstrated in vivo.8,10 Perhaps erlotinib has a similar potential to paradoxically upregulate the MAPK pathway, thus stimulating cellular proliferation and survival.
Another tyrosine kinase receptor, c-KIT, is found on the cell membrane of melanocytes along with EGFR.11,12 The c-KIT receptor also activates the MAPK pathway and is critical to the development, migration, and survival of melanocytes.11,13 Stimulation of the c-KIT tyrosine kinase receptor also can induce melanocyte proliferation and melanogenesis.11 The c-KIT receptor is encoded by the KIT gene (KIT proto-oncogene receptor tyrosine kinase). Mutations in this gene are associated with melanocytic disorders. Inherited KIT mutation leading to c-KIT receptor deficiency is associated with piebaldism. Acquired activating KIT mutations increasing c-KIT expression are associated with acral and mucosal melanomas as well as melanomas in chronically sun-damaged skin.13
We hypothesized that erlotinib-induced inhibition of the MAPK pathway could lead to a reactive increase in expression of c-KIT and thus stimulate melanocyte proliferation and pigment production. Similar feedback upregulation of an MAPK pathway stimulating receptor during downstream MAPK inhibition has been demonstrated in colon adenocarcinoma; in this setting, BRAF inhibitors blocking the MAPK pathway leads to upregulation of EGFR.14 In our patient, c-KIT immunostaining revealed a mild to moderate increase in intensity (ie, the darkness of the staining) in nevi and melanomas during treatment with erlotinib compared to nevi biopsied before erlotinib treatment (Figure 2). The increased intensity of c-KIT immunostaining was further confirmed via semiquantitative digital image analysis. Using this method, a darkened nevus biopsied during treatment with erlotinib demonstrated 43.16% of cells (N=31,451) had very strong c-KIT staining, while a nevus biopsied before treatment with erlotinib demonstrated only 3.32% of cells (N=7507) with very strong c-KIT staining. Increased expression of c-KIT, possibly reactive to downstream inhibition the MAPK pathway from erlotinib, could be implicated in our case of eruptive nevi.
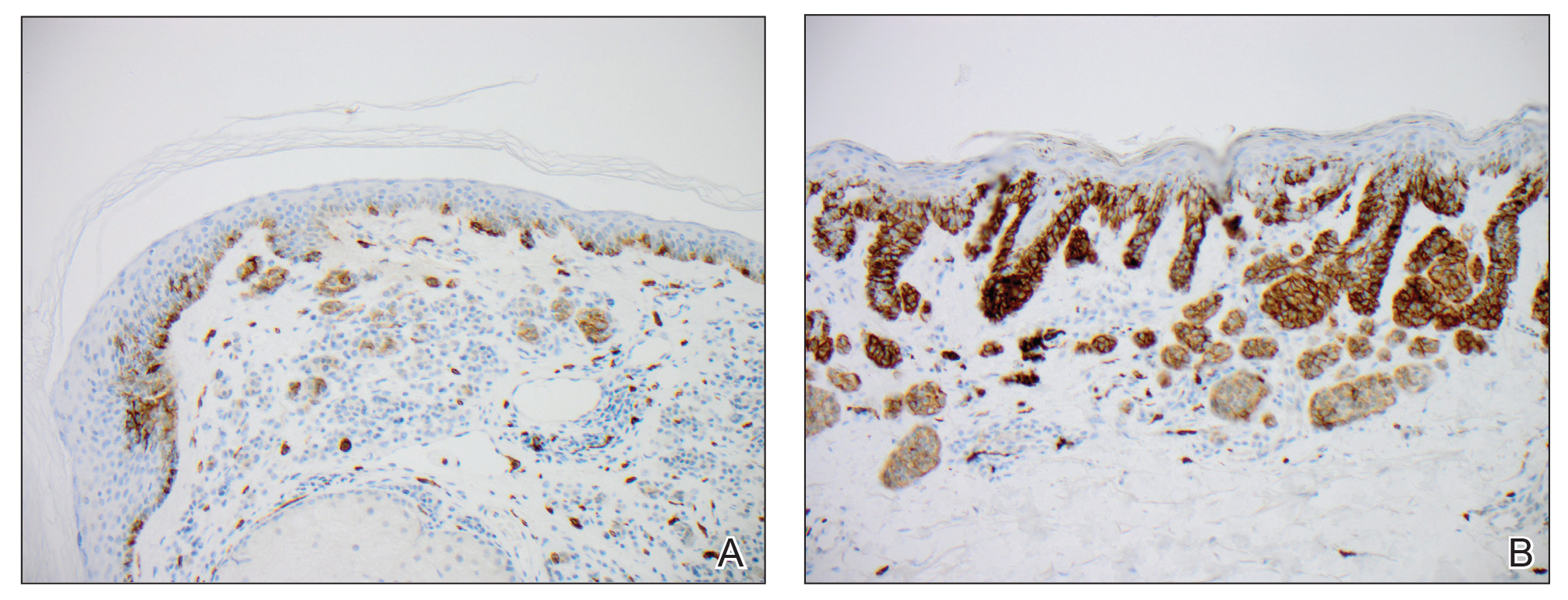
In summary, we report a rare case of darkening of existing nevi and development of melanoma in situ during treatment with erlotinib. The patient’s therapeutic timeline and concurrence of other well-documented side effects provided support for erlotinib as the causative agent in our patient. Additional support is provided through reports of other medications affecting the same pathway as erlotinib causing eruptive nevi, darkening of existing nevi, and melanoma in situ.7-10 Through c-KIT immunostaining, we demonstrated that increased expression of c-KIT might be responsible for the changes in nevi in our patient. We, therefore, suggest frequent full-body skin examinations in patients treated with erlotinib to monitor for the possible development of malignant melanomas.
- Santiago F, Goncalo M, Reis J, et al. Adverse cutaneous reactions to epidermal growth factor receptor inhibitors: a study of 14 patients. An Bras Dermatol 2011;86:483-490.
- Lubbe J, Masouye I, Dietrich P. Generalized xerotic dermatitis with neutrophilic spongiosis induced by erlotinib (Tarceva). Dermatology. 2008;216:247-249.
- Dessinioti C, Antoniou C, Katsambas A. Acneiform eruptions. Clin Dermatol. 2014;32:24-34.
- Herbst R, Fukuoka M, Baselga J. Gefitinib—a novel targeted approach to treating cancer. Nat Rev Cancer. 2004;4:979-987.
- Brodell L, Hepper D, Lind A, et al. Histopathology of acneiform eruptions in patients treated with epidermal growth factor receptor inhibitors. J Cutan Pathol. 2013;40:865-870.
- Kiyohara Y, Yamazaki N, Kishi A. Erlotinib-related skin toxicities: treatment strategies in patients with metastatic non-small cell lung cancer. J Am Acad Dermatol 2013;69:463-472.
- Uhlenhake E, Watson A, Aronson P. Sorafenib induced eruptive melanocytic lesions. Dermatol Online J. 2013;19:181-84.
- Chu E, Wanat K, Miller C, et al. Diverse cutaneous side effects associated with BRAF inhibitor therapy: a clinicopathologic study. J Am Acad Dermatol 2012;67:1265-1272.
- Boussemart L, Routier E, Mateus C, et al. Prospective study of cutaneous side-effects associated with the BRAF inhibitor vemurafenib: a study of 42 patients. Ann Oncol. 2013;24:1691-1697.
- Cohen P, Bedikian A, Kim K. Appearance of new vemurafenib-associated melanocytic nevi on normal-appearing skin: case series and a review of changing or new pigmented lesions in patients with metastatic malignant melanoma after initiating treatment with vemurafenib. J Clin Aesthet Dermatol. 2013;6:27-37.
- Longley B, Tyrrell L, Lu S, et al. Somatic c-KIT activating mutation in urticaria pigmentosa and aggressive mastocytosis: establishment of clonality in a human mast cell neoplasm. Nat Genet. 1996;12:312-314.
- Yun W, Bang S, Min K, et al. Epidermal growth factor and epidermal growth factor signaling attenuate laser-induced melanogenesis. Dermatol Surg. 2013;39:1903-1911.
- Swick J, Maize J. Molecular biology of melanoma. J Am Acad Dermatol. 2012;67:1049-1054.
- Sun C, Wang L, Huang S, et al. Reversible and adaptive resistance to BRAF(V600E) inhibition in melanoma. Nature. 2014;508:118-122.
To the Editor:
Erlotinib is a small-molecule selective tyrosine kinase inhibitor that functions by blocking the intracellular portion of the epidermal growth factor receptor (EGFR)1,2; EGFR normally is expressed in the basal layer of the epidermis, sweat glands, and hair follicles, and is overexpressed in some cancers.1,3 Normal activation of EGFR leads to signal transduction through the mitogen-activated protein kinase (MAPK) signaling pathway, which stimulates cell survival and proliferation.4,5 Erlotinib-induced inhibition of EGFR prevents tyrosine kinase phosphorylation and aims to decrease cell proliferation in these tumors.
Erlotinib is indicated as once-daily oral monotherapy for the treatment of advanced-stage non–small cell lung cancer (NSCLCA) and in combination with gemcitabine for treatment of advanced-stage pancreatic cancer.1 A number of cutaneous side effects have been reported, including acneform eruption, xerosis, paronychia, and pruritus.6 Other tyrosine kinase inhibitors, which also decrease signal transduction through the MAPK pathway, have some overlapping side effects; among these are vemurafenib, a selective BRAF inhibitor, and sorafenib, a multikinase inhibitor.7,8
A 70-year-old man with NSCLCA presented with eruptive nevi and darkening of existing nevi 3 months after starting monotherapy with erlotinib. Physical examination demonstrated the simultaneous appearance of scattered acneform papules and pustules; diffuse xerosis; and numerous dark brown to black nevi on the trunk, arms, and legs. Compared to prior clinical photographs taken in our office, darkening of existing medium brown nevi was noted, and new nevi developed in areas where no prior nevi had been visible (Figure 1).

The patient’s medical history included 3 invasive melanomas, all of which were diagnosed at least 7 years prior to the initiation of erlotinib and were treated by surgical excision alone. Prior treatment of NSCLCA consisted of a left lower lobectomy followed by docetaxel, carboplatin, pegfilgrastim, dexamethasone, and pemetrexed. A thorough review of all of the patient’s medications revealed no associations with changes in nevi.
A review of the patient’s treatment timeline revealed that all other chemotherapeutic medications had been discontinued a minimum of 5 weeks before starting erlotinib. A complete cutaneous examination performed in our office after completion of these chemotherapeutic agents and prior to initiation of erlotinib was unremarkable for abnormally dark or eruptive nevi.
Since starting erlotinib treatment, the patient underwent 10 biopsies of clinically suspicious dark nevi performed by a dermatologist in our office. Two of these were diagnosed as melanoma in situ and one as an atypical nevus. A temporal association of the darkening and eruptive nevi with erlotinib treatment was established; however, because erlotinib was essential to his NSCLCA treatment, he continued erlotinib with frequent complete cutaneous examinations.
A number of cutaneous side effects have been described during treatment with erlotinib, the most common being acneform eruption.6 The incidence and severity of acneform eruptions have been positively correlated to survival in patients with NSCLCA.3,5,6 Other common side effects include xerosis, paronychia, and pruritus.1,5,6 Less common side effects include periungual pyogenic granulomas and hair growth abnormalities.1
Eruptive nevi previously were reported in a patient who was treated with erlotinib.1 Other tyrosine kinase inhibitors that also decrease signal transduction through the MAPK pathway, including sorafenib and vemurafenib, have been reported to cause eruptive nevi. There are 7 reports of eruptive nevi with sorafenib and 5 reports with vemurafenib.7-9 Development of nevi were noted within a few months of initiating treatment with these medications.7
A PubMed search of articles indexed for MEDLINE using the terms erlotinib and melanoma and erlotinib and nevi yielded no prior reports of darkening of existing nevi or the development of melanoma during treatment with erlotinib. However, vemurafenib has been reported to cause dysplastic nevi, melanomas, and darkening of existing nevi, in addition to eruptive nevi.8-10 The side effects of vemurafenib have been ascribed to a paradoxical upregulation of MAPK in BRAF wild-type cells. This effect has been well documented and demonstrated in vivo.8,10 Perhaps erlotinib has a similar potential to paradoxically upregulate the MAPK pathway, thus stimulating cellular proliferation and survival.
Another tyrosine kinase receptor, c-KIT, is found on the cell membrane of melanocytes along with EGFR.11,12 The c-KIT receptor also activates the MAPK pathway and is critical to the development, migration, and survival of melanocytes.11,13 Stimulation of the c-KIT tyrosine kinase receptor also can induce melanocyte proliferation and melanogenesis.11 The c-KIT receptor is encoded by the KIT gene (KIT proto-oncogene receptor tyrosine kinase). Mutations in this gene are associated with melanocytic disorders. Inherited KIT mutation leading to c-KIT receptor deficiency is associated with piebaldism. Acquired activating KIT mutations increasing c-KIT expression are associated with acral and mucosal melanomas as well as melanomas in chronically sun-damaged skin.13
We hypothesized that erlotinib-induced inhibition of the MAPK pathway could lead to a reactive increase in expression of c-KIT and thus stimulate melanocyte proliferation and pigment production. Similar feedback upregulation of an MAPK pathway stimulating receptor during downstream MAPK inhibition has been demonstrated in colon adenocarcinoma; in this setting, BRAF inhibitors blocking the MAPK pathway leads to upregulation of EGFR.14 In our patient, c-KIT immunostaining revealed a mild to moderate increase in intensity (ie, the darkness of the staining) in nevi and melanomas during treatment with erlotinib compared to nevi biopsied before erlotinib treatment (Figure 2). The increased intensity of c-KIT immunostaining was further confirmed via semiquantitative digital image analysis. Using this method, a darkened nevus biopsied during treatment with erlotinib demonstrated 43.16% of cells (N=31,451) had very strong c-KIT staining, while a nevus biopsied before treatment with erlotinib demonstrated only 3.32% of cells (N=7507) with very strong c-KIT staining. Increased expression of c-KIT, possibly reactive to downstream inhibition the MAPK pathway from erlotinib, could be implicated in our case of eruptive nevi.

In summary, we report a rare case of darkening of existing nevi and development of melanoma in situ during treatment with erlotinib. The patient’s therapeutic timeline and concurrence of other well-documented side effects provided support for erlotinib as the causative agent in our patient. Additional support is provided through reports of other medications affecting the same pathway as erlotinib causing eruptive nevi, darkening of existing nevi, and melanoma in situ.7-10 Through c-KIT immunostaining, we demonstrated that increased expression of c-KIT might be responsible for the changes in nevi in our patient. We, therefore, suggest frequent full-body skin examinations in patients treated with erlotinib to monitor for the possible development of malignant melanomas.
To the Editor:
Erlotinib is a small-molecule selective tyrosine kinase inhibitor that functions by blocking the intracellular portion of the epidermal growth factor receptor (EGFR)1,2; EGFR normally is expressed in the basal layer of the epidermis, sweat glands, and hair follicles, and is overexpressed in some cancers.1,3 Normal activation of EGFR leads to signal transduction through the mitogen-activated protein kinase (MAPK) signaling pathway, which stimulates cell survival and proliferation.4,5 Erlotinib-induced inhibition of EGFR prevents tyrosine kinase phosphorylation and aims to decrease cell proliferation in these tumors.
Erlotinib is indicated as once-daily oral monotherapy for the treatment of advanced-stage non–small cell lung cancer (NSCLCA) and in combination with gemcitabine for treatment of advanced-stage pancreatic cancer.1 A number of cutaneous side effects have been reported, including acneform eruption, xerosis, paronychia, and pruritus.6 Other tyrosine kinase inhibitors, which also decrease signal transduction through the MAPK pathway, have some overlapping side effects; among these are vemurafenib, a selective BRAF inhibitor, and sorafenib, a multikinase inhibitor.7,8
A 70-year-old man with NSCLCA presented with eruptive nevi and darkening of existing nevi 3 months after starting monotherapy with erlotinib. Physical examination demonstrated the simultaneous appearance of scattered acneform papules and pustules; diffuse xerosis; and numerous dark brown to black nevi on the trunk, arms, and legs. Compared to prior clinical photographs taken in our office, darkening of existing medium brown nevi was noted, and new nevi developed in areas where no prior nevi had been visible (Figure 1).

The patient’s medical history included 3 invasive melanomas, all of which were diagnosed at least 7 years prior to the initiation of erlotinib and were treated by surgical excision alone. Prior treatment of NSCLCA consisted of a left lower lobectomy followed by docetaxel, carboplatin, pegfilgrastim, dexamethasone, and pemetrexed. A thorough review of all of the patient’s medications revealed no associations with changes in nevi.
A review of the patient’s treatment timeline revealed that all other chemotherapeutic medications had been discontinued a minimum of 5 weeks before starting erlotinib. A complete cutaneous examination performed in our office after completion of these chemotherapeutic agents and prior to initiation of erlotinib was unremarkable for abnormally dark or eruptive nevi.
Since starting erlotinib treatment, the patient underwent 10 biopsies of clinically suspicious dark nevi performed by a dermatologist in our office. Two of these were diagnosed as melanoma in situ and one as an atypical nevus. A temporal association of the darkening and eruptive nevi with erlotinib treatment was established; however, because erlotinib was essential to his NSCLCA treatment, he continued erlotinib with frequent complete cutaneous examinations.
A number of cutaneous side effects have been described during treatment with erlotinib, the most common being acneform eruption.6 The incidence and severity of acneform eruptions have been positively correlated to survival in patients with NSCLCA.3,5,6 Other common side effects include xerosis, paronychia, and pruritus.1,5,6 Less common side effects include periungual pyogenic granulomas and hair growth abnormalities.1
Eruptive nevi previously were reported in a patient who was treated with erlotinib.1 Other tyrosine kinase inhibitors that also decrease signal transduction through the MAPK pathway, including sorafenib and vemurafenib, have been reported to cause eruptive nevi. There are 7 reports of eruptive nevi with sorafenib and 5 reports with vemurafenib.7-9 Development of nevi were noted within a few months of initiating treatment with these medications.7
A PubMed search of articles indexed for MEDLINE using the terms erlotinib and melanoma and erlotinib and nevi yielded no prior reports of darkening of existing nevi or the development of melanoma during treatment with erlotinib. However, vemurafenib has been reported to cause dysplastic nevi, melanomas, and darkening of existing nevi, in addition to eruptive nevi.8-10 The side effects of vemurafenib have been ascribed to a paradoxical upregulation of MAPK in BRAF wild-type cells. This effect has been well documented and demonstrated in vivo.8,10 Perhaps erlotinib has a similar potential to paradoxically upregulate the MAPK pathway, thus stimulating cellular proliferation and survival.
Another tyrosine kinase receptor, c-KIT, is found on the cell membrane of melanocytes along with EGFR.11,12 The c-KIT receptor also activates the MAPK pathway and is critical to the development, migration, and survival of melanocytes.11,13 Stimulation of the c-KIT tyrosine kinase receptor also can induce melanocyte proliferation and melanogenesis.11 The c-KIT receptor is encoded by the KIT gene (KIT proto-oncogene receptor tyrosine kinase). Mutations in this gene are associated with melanocytic disorders. Inherited KIT mutation leading to c-KIT receptor deficiency is associated with piebaldism. Acquired activating KIT mutations increasing c-KIT expression are associated with acral and mucosal melanomas as well as melanomas in chronically sun-damaged skin.13
We hypothesized that erlotinib-induced inhibition of the MAPK pathway could lead to a reactive increase in expression of c-KIT and thus stimulate melanocyte proliferation and pigment production. Similar feedback upregulation of an MAPK pathway stimulating receptor during downstream MAPK inhibition has been demonstrated in colon adenocarcinoma; in this setting, BRAF inhibitors blocking the MAPK pathway leads to upregulation of EGFR.14 In our patient, c-KIT immunostaining revealed a mild to moderate increase in intensity (ie, the darkness of the staining) in nevi and melanomas during treatment with erlotinib compared to nevi biopsied before erlotinib treatment (Figure 2). The increased intensity of c-KIT immunostaining was further confirmed via semiquantitative digital image analysis. Using this method, a darkened nevus biopsied during treatment with erlotinib demonstrated 43.16% of cells (N=31,451) had very strong c-KIT staining, while a nevus biopsied before treatment with erlotinib demonstrated only 3.32% of cells (N=7507) with very strong c-KIT staining. Increased expression of c-KIT, possibly reactive to downstream inhibition the MAPK pathway from erlotinib, could be implicated in our case of eruptive nevi.

In summary, we report a rare case of darkening of existing nevi and development of melanoma in situ during treatment with erlotinib. The patient’s therapeutic timeline and concurrence of other well-documented side effects provided support for erlotinib as the causative agent in our patient. Additional support is provided through reports of other medications affecting the same pathway as erlotinib causing eruptive nevi, darkening of existing nevi, and melanoma in situ.7-10 Through c-KIT immunostaining, we demonstrated that increased expression of c-KIT might be responsible for the changes in nevi in our patient. We, therefore, suggest frequent full-body skin examinations in patients treated with erlotinib to monitor for the possible development of malignant melanomas.
- Santiago F, Goncalo M, Reis J, et al. Adverse cutaneous reactions to epidermal growth factor receptor inhibitors: a study of 14 patients. An Bras Dermatol 2011;86:483-490.
- Lubbe J, Masouye I, Dietrich P. Generalized xerotic dermatitis with neutrophilic spongiosis induced by erlotinib (Tarceva). Dermatology. 2008;216:247-249.
- Dessinioti C, Antoniou C, Katsambas A. Acneiform eruptions. Clin Dermatol. 2014;32:24-34.
- Herbst R, Fukuoka M, Baselga J. Gefitinib—a novel targeted approach to treating cancer. Nat Rev Cancer. 2004;4:979-987.
- Brodell L, Hepper D, Lind A, et al. Histopathology of acneiform eruptions in patients treated with epidermal growth factor receptor inhibitors. J Cutan Pathol. 2013;40:865-870.
- Kiyohara Y, Yamazaki N, Kishi A. Erlotinib-related skin toxicities: treatment strategies in patients with metastatic non-small cell lung cancer. J Am Acad Dermatol 2013;69:463-472.
- Uhlenhake E, Watson A, Aronson P. Sorafenib induced eruptive melanocytic lesions. Dermatol Online J. 2013;19:181-84.
- Chu E, Wanat K, Miller C, et al. Diverse cutaneous side effects associated with BRAF inhibitor therapy: a clinicopathologic study. J Am Acad Dermatol 2012;67:1265-1272.
- Boussemart L, Routier E, Mateus C, et al. Prospective study of cutaneous side-effects associated with the BRAF inhibitor vemurafenib: a study of 42 patients. Ann Oncol. 2013;24:1691-1697.
- Cohen P, Bedikian A, Kim K. Appearance of new vemurafenib-associated melanocytic nevi on normal-appearing skin: case series and a review of changing or new pigmented lesions in patients with metastatic malignant melanoma after initiating treatment with vemurafenib. J Clin Aesthet Dermatol. 2013;6:27-37.
- Longley B, Tyrrell L, Lu S, et al. Somatic c-KIT activating mutation in urticaria pigmentosa and aggressive mastocytosis: establishment of clonality in a human mast cell neoplasm. Nat Genet. 1996;12:312-314.
- Yun W, Bang S, Min K, et al. Epidermal growth factor and epidermal growth factor signaling attenuate laser-induced melanogenesis. Dermatol Surg. 2013;39:1903-1911.
- Swick J, Maize J. Molecular biology of melanoma. J Am Acad Dermatol. 2012;67:1049-1054.
- Sun C, Wang L, Huang S, et al. Reversible and adaptive resistance to BRAF(V600E) inhibition in melanoma. Nature. 2014;508:118-122.
- Santiago F, Goncalo M, Reis J, et al. Adverse cutaneous reactions to epidermal growth factor receptor inhibitors: a study of 14 patients. An Bras Dermatol 2011;86:483-490.
- Lubbe J, Masouye I, Dietrich P. Generalized xerotic dermatitis with neutrophilic spongiosis induced by erlotinib (Tarceva). Dermatology. 2008;216:247-249.
- Dessinioti C, Antoniou C, Katsambas A. Acneiform eruptions. Clin Dermatol. 2014;32:24-34.
- Herbst R, Fukuoka M, Baselga J. Gefitinib—a novel targeted approach to treating cancer. Nat Rev Cancer. 2004;4:979-987.
- Brodell L, Hepper D, Lind A, et al. Histopathology of acneiform eruptions in patients treated with epidermal growth factor receptor inhibitors. J Cutan Pathol. 2013;40:865-870.
- Kiyohara Y, Yamazaki N, Kishi A. Erlotinib-related skin toxicities: treatment strategies in patients with metastatic non-small cell lung cancer. J Am Acad Dermatol 2013;69:463-472.
- Uhlenhake E, Watson A, Aronson P. Sorafenib induced eruptive melanocytic lesions. Dermatol Online J. 2013;19:181-84.
- Chu E, Wanat K, Miller C, et al. Diverse cutaneous side effects associated with BRAF inhibitor therapy: a clinicopathologic study. J Am Acad Dermatol 2012;67:1265-1272.
- Boussemart L, Routier E, Mateus C, et al. Prospective study of cutaneous side-effects associated with the BRAF inhibitor vemurafenib: a study of 42 patients. Ann Oncol. 2013;24:1691-1697.
- Cohen P, Bedikian A, Kim K. Appearance of new vemurafenib-associated melanocytic nevi on normal-appearing skin: case series and a review of changing or new pigmented lesions in patients with metastatic malignant melanoma after initiating treatment with vemurafenib. J Clin Aesthet Dermatol. 2013;6:27-37.
- Longley B, Tyrrell L, Lu S, et al. Somatic c-KIT activating mutation in urticaria pigmentosa and aggressive mastocytosis: establishment of clonality in a human mast cell neoplasm. Nat Genet. 1996;12:312-314.
- Yun W, Bang S, Min K, et al. Epidermal growth factor and epidermal growth factor signaling attenuate laser-induced melanogenesis. Dermatol Surg. 2013;39:1903-1911.
- Swick J, Maize J. Molecular biology of melanoma. J Am Acad Dermatol. 2012;67:1049-1054.
- Sun C, Wang L, Huang S, et al. Reversible and adaptive resistance to BRAF(V600E) inhibition in melanoma. Nature. 2014;508:118-122.
Practice Points
- Cutaneous side effects of erlotinib include acneform eruption, xerosis, paronychia, and pruritus.
- Clinicians should monitor patients for darkening and/or eruptive nevi as well as melanoma during treatment with erlotinib.
Study outlines survival factors with nivolumab
Predictors of long-term survival of patients with advanced melanoma, renal cell carcinoma (RCC), non–small cell lung cancer (NSCLC), and other malignancies treated with nivolumab include the absence of liver or bone metastases, excellent baseline performance status, and the presence of grade 3 or greater treatment-related adverse events, investigators have found.
A secondary analysis of the phase 1 CA209-003 trial with expansion cohorts showed that, among 270 heavily pretreated patients with melanoma, RCC, and NSCLC who received single-agent nivolumab (Opdivo) during this trial, those with liver or bone metastases had a 69% higher risk for death within 5 years.
In contrast, patients with Eastern Cooperative Oncology Group (ECOG) performance status of 0 had a nearly threefold higher chance for survival, compared with patients with less favorable performance status scores, reported Suzanne L. Topalian, MD, from the Sidney Kimmel Comprehensive Cancer Center at Johns Hopkins University in Baltimore and colleagues.
“The results of this study suggest that survival benefits reported in the more limited follow-up of recent nivolumab randomized clinical trials may persist for prolonged periods in some patients, extending to at least 5 years,” they wrote in JAMA Oncology.
In the CA209-003 trial, investigators enrolled patients 18 years or older with documented evidence of advanced melanoma, RCC, NSCLC, castration-resistant prostate cancer, or colorectal cancer. To be eligible, patients needed to have received 1-5 previous systemic therapies for advanced or recurrent cancer, measurable disease by Response Evaluation Criteria in Solid Tumors (RECIST) version 1.0, and an ECOG performance status of 0-2. The current survival analysis included data on 107 patients with melanoma, 34 with RCC, and 129 with NSCLC.
Estimated 5-year overall survival rates were 34.2% for patients with melanoma, 27.7% for patients with RCC, and 15.6% for patients with NSCLC. A multivariable analysis controlling for age, sex, performance status, metastatic disease, and number of prior therapies showed that the presence of either liver or bone metastases was associated with an odds ratio for 5-year survival of 0.31 (P = .02 and .04, respectively).
One factor favorably associated with survival included ECOG performance status 0 (OR, 2.74; P = .003). The investigators also found that treatment-related adverse events (AEs) were associated with longer overall survival, with a median of 19.8 months for patients with any grade of treatment-related event and 20.3 months for patients with grade 3 or greater events, compared with a median of 5.8 months for patients with no treatment-related events (P less than .001 for each comparison based on hazard ratios).
“Of note, patients in our study who developed treatment-related AEs, regardless of whether the AEs were deemed to have an immune-mediated causality, had significantly higher ORRs [overall response rates] and prolonged 5-year OS. These findings are reminiscent of some reports of anti–CTLA-4 therapy and align with other studies of anti–PD-1 therapies, “ Dr. Topalian and associates wrote.
The study and the secondary analysis were supported by Bristol-Myers Squibb. Dr. Topalian disclosed grants and travel reimbursements from Bristol-Myers Squibb and consulting fees with other entities. Multiple co-authors reported similar relationships. Four of the co-authors are Bristol-Myers Squibb employees.
SOURCE: Topalian SL et al. JAMA Oncology. 2019 Jul 25. doi: 10.1001/jamaoncol.2019.2187.
Although the existence of a subset of patients experiencing long-term survival certainly substantiates the role of PD-1/ PD-L1 checkpoint blockade in cancer immunotherapy, it is noteworthy to consider that these agents as monotherapy have not yielded sufficient activity and efficacy to replace standard-of-care therapy in the first line of therapy in advanced solid tumors, with the exception of NSCLC expressing high levels of PD-L1 and melanoma; emerging results also restrict monotherapy to stringently defined subsets of patients with gastric, esophageal, head and neck, and bladder cancers. Baseline predictive biomarkers have demonstrated distinct shortcomings, the first being their poor discriminatory ability and low negative predictive value. The clinician keen on securing the best possible outcome for his patients is thus left with the necessity for indiscriminate administration of PD-1/PD-L1 checkpoint inhibitors.
Unsurprisingly, the field of combination therapies using PD-1/PD-L1 checkpoint blockade as a backbone has been growing exponentially; a recent review shows more than 2,250 immunotherapy trials, 1,716 of which are investigating PD-1/ PD-L1 checkpoint inhibitors with more than 240 combination partners. Analysis of the pipeline also reveals a 67% increase in the number of active agents, amounting to more than 3,300, between September 2017 and September 2018. A noteworthy development is a 113% increase in cell therapies, and an increase of agents targeting neoantigens identified through bioinformatics analysis of an individual patient’s tumor, suggesting a shift toward increased personalization of immunotherapy. The observation that clinical development of immunotherapy agents has outstripped our understanding of the cancer-immune interactions advocates for renewed collective efforts in standardizing immune monitoring methods in clinical trials to identify immune evasion pathways that are dominant and to build novel trial designs able to efficiently enhance matching of patients with therapy.
Stefan Zimmermann, MD, and Solange Peters, MD, PhD, are from the Centre Hospitalier Universitaire Vaudois in Lausanne,Switzerland. Their remarks are excerpted and adapted from an editorial accompanying the study (JAMA Oncol. 2019 Jul 25. doi: 10.1001/jamaoncol.2019.2186). Dr. Zimmerman disclosed fees for advisory roles, travel grants, and clinical research support from Bristol-Myers Squibb and others. Dr. Peters disclosed fees for advisory board participation and/or lectures from Bristol-Myers Squibb and others.
Although the existence of a subset of patients experiencing long-term survival certainly substantiates the role of PD-1/ PD-L1 checkpoint blockade in cancer immunotherapy, it is noteworthy to consider that these agents as monotherapy have not yielded sufficient activity and efficacy to replace standard-of-care therapy in the first line of therapy in advanced solid tumors, with the exception of NSCLC expressing high levels of PD-L1 and melanoma; emerging results also restrict monotherapy to stringently defined subsets of patients with gastric, esophageal, head and neck, and bladder cancers. Baseline predictive biomarkers have demonstrated distinct shortcomings, the first being their poor discriminatory ability and low negative predictive value. The clinician keen on securing the best possible outcome for his patients is thus left with the necessity for indiscriminate administration of PD-1/PD-L1 checkpoint inhibitors.
Unsurprisingly, the field of combination therapies using PD-1/PD-L1 checkpoint blockade as a backbone has been growing exponentially; a recent review shows more than 2,250 immunotherapy trials, 1,716 of which are investigating PD-1/ PD-L1 checkpoint inhibitors with more than 240 combination partners. Analysis of the pipeline also reveals a 67% increase in the number of active agents, amounting to more than 3,300, between September 2017 and September 2018. A noteworthy development is a 113% increase in cell therapies, and an increase of agents targeting neoantigens identified through bioinformatics analysis of an individual patient’s tumor, suggesting a shift toward increased personalization of immunotherapy. The observation that clinical development of immunotherapy agents has outstripped our understanding of the cancer-immune interactions advocates for renewed collective efforts in standardizing immune monitoring methods in clinical trials to identify immune evasion pathways that are dominant and to build novel trial designs able to efficiently enhance matching of patients with therapy.
Stefan Zimmermann, MD, and Solange Peters, MD, PhD, are from the Centre Hospitalier Universitaire Vaudois in Lausanne,Switzerland. Their remarks are excerpted and adapted from an editorial accompanying the study (JAMA Oncol. 2019 Jul 25. doi: 10.1001/jamaoncol.2019.2186). Dr. Zimmerman disclosed fees for advisory roles, travel grants, and clinical research support from Bristol-Myers Squibb and others. Dr. Peters disclosed fees for advisory board participation and/or lectures from Bristol-Myers Squibb and others.
Although the existence of a subset of patients experiencing long-term survival certainly substantiates the role of PD-1/ PD-L1 checkpoint blockade in cancer immunotherapy, it is noteworthy to consider that these agents as monotherapy have not yielded sufficient activity and efficacy to replace standard-of-care therapy in the first line of therapy in advanced solid tumors, with the exception of NSCLC expressing high levels of PD-L1 and melanoma; emerging results also restrict monotherapy to stringently defined subsets of patients with gastric, esophageal, head and neck, and bladder cancers. Baseline predictive biomarkers have demonstrated distinct shortcomings, the first being their poor discriminatory ability and low negative predictive value. The clinician keen on securing the best possible outcome for his patients is thus left with the necessity for indiscriminate administration of PD-1/PD-L1 checkpoint inhibitors.
Unsurprisingly, the field of combination therapies using PD-1/PD-L1 checkpoint blockade as a backbone has been growing exponentially; a recent review shows more than 2,250 immunotherapy trials, 1,716 of which are investigating PD-1/ PD-L1 checkpoint inhibitors with more than 240 combination partners. Analysis of the pipeline also reveals a 67% increase in the number of active agents, amounting to more than 3,300, between September 2017 and September 2018. A noteworthy development is a 113% increase in cell therapies, and an increase of agents targeting neoantigens identified through bioinformatics analysis of an individual patient’s tumor, suggesting a shift toward increased personalization of immunotherapy. The observation that clinical development of immunotherapy agents has outstripped our understanding of the cancer-immune interactions advocates for renewed collective efforts in standardizing immune monitoring methods in clinical trials to identify immune evasion pathways that are dominant and to build novel trial designs able to efficiently enhance matching of patients with therapy.
Stefan Zimmermann, MD, and Solange Peters, MD, PhD, are from the Centre Hospitalier Universitaire Vaudois in Lausanne,Switzerland. Their remarks are excerpted and adapted from an editorial accompanying the study (JAMA Oncol. 2019 Jul 25. doi: 10.1001/jamaoncol.2019.2186). Dr. Zimmerman disclosed fees for advisory roles, travel grants, and clinical research support from Bristol-Myers Squibb and others. Dr. Peters disclosed fees for advisory board participation and/or lectures from Bristol-Myers Squibb and others.
Predictors of long-term survival of patients with advanced melanoma, renal cell carcinoma (RCC), non–small cell lung cancer (NSCLC), and other malignancies treated with nivolumab include the absence of liver or bone metastases, excellent baseline performance status, and the presence of grade 3 or greater treatment-related adverse events, investigators have found.
A secondary analysis of the phase 1 CA209-003 trial with expansion cohorts showed that, among 270 heavily pretreated patients with melanoma, RCC, and NSCLC who received single-agent nivolumab (Opdivo) during this trial, those with liver or bone metastases had a 69% higher risk for death within 5 years.
In contrast, patients with Eastern Cooperative Oncology Group (ECOG) performance status of 0 had a nearly threefold higher chance for survival, compared with patients with less favorable performance status scores, reported Suzanne L. Topalian, MD, from the Sidney Kimmel Comprehensive Cancer Center at Johns Hopkins University in Baltimore and colleagues.
“The results of this study suggest that survival benefits reported in the more limited follow-up of recent nivolumab randomized clinical trials may persist for prolonged periods in some patients, extending to at least 5 years,” they wrote in JAMA Oncology.
In the CA209-003 trial, investigators enrolled patients 18 years or older with documented evidence of advanced melanoma, RCC, NSCLC, castration-resistant prostate cancer, or colorectal cancer. To be eligible, patients needed to have received 1-5 previous systemic therapies for advanced or recurrent cancer, measurable disease by Response Evaluation Criteria in Solid Tumors (RECIST) version 1.0, and an ECOG performance status of 0-2. The current survival analysis included data on 107 patients with melanoma, 34 with RCC, and 129 with NSCLC.
Estimated 5-year overall survival rates were 34.2% for patients with melanoma, 27.7% for patients with RCC, and 15.6% for patients with NSCLC. A multivariable analysis controlling for age, sex, performance status, metastatic disease, and number of prior therapies showed that the presence of either liver or bone metastases was associated with an odds ratio for 5-year survival of 0.31 (P = .02 and .04, respectively).
One factor favorably associated with survival included ECOG performance status 0 (OR, 2.74; P = .003). The investigators also found that treatment-related adverse events (AEs) were associated with longer overall survival, with a median of 19.8 months for patients with any grade of treatment-related event and 20.3 months for patients with grade 3 or greater events, compared with a median of 5.8 months for patients with no treatment-related events (P less than .001 for each comparison based on hazard ratios).
“Of note, patients in our study who developed treatment-related AEs, regardless of whether the AEs were deemed to have an immune-mediated causality, had significantly higher ORRs [overall response rates] and prolonged 5-year OS. These findings are reminiscent of some reports of anti–CTLA-4 therapy and align with other studies of anti–PD-1 therapies, “ Dr. Topalian and associates wrote.
The study and the secondary analysis were supported by Bristol-Myers Squibb. Dr. Topalian disclosed grants and travel reimbursements from Bristol-Myers Squibb and consulting fees with other entities. Multiple co-authors reported similar relationships. Four of the co-authors are Bristol-Myers Squibb employees.
SOURCE: Topalian SL et al. JAMA Oncology. 2019 Jul 25. doi: 10.1001/jamaoncol.2019.2187.
Predictors of long-term survival of patients with advanced melanoma, renal cell carcinoma (RCC), non–small cell lung cancer (NSCLC), and other malignancies treated with nivolumab include the absence of liver or bone metastases, excellent baseline performance status, and the presence of grade 3 or greater treatment-related adverse events, investigators have found.
A secondary analysis of the phase 1 CA209-003 trial with expansion cohorts showed that, among 270 heavily pretreated patients with melanoma, RCC, and NSCLC who received single-agent nivolumab (Opdivo) during this trial, those with liver or bone metastases had a 69% higher risk for death within 5 years.
In contrast, patients with Eastern Cooperative Oncology Group (ECOG) performance status of 0 had a nearly threefold higher chance for survival, compared with patients with less favorable performance status scores, reported Suzanne L. Topalian, MD, from the Sidney Kimmel Comprehensive Cancer Center at Johns Hopkins University in Baltimore and colleagues.
“The results of this study suggest that survival benefits reported in the more limited follow-up of recent nivolumab randomized clinical trials may persist for prolonged periods in some patients, extending to at least 5 years,” they wrote in JAMA Oncology.
In the CA209-003 trial, investigators enrolled patients 18 years or older with documented evidence of advanced melanoma, RCC, NSCLC, castration-resistant prostate cancer, or colorectal cancer. To be eligible, patients needed to have received 1-5 previous systemic therapies for advanced or recurrent cancer, measurable disease by Response Evaluation Criteria in Solid Tumors (RECIST) version 1.0, and an ECOG performance status of 0-2. The current survival analysis included data on 107 patients with melanoma, 34 with RCC, and 129 with NSCLC.
Estimated 5-year overall survival rates were 34.2% for patients with melanoma, 27.7% for patients with RCC, and 15.6% for patients with NSCLC. A multivariable analysis controlling for age, sex, performance status, metastatic disease, and number of prior therapies showed that the presence of either liver or bone metastases was associated with an odds ratio for 5-year survival of 0.31 (P = .02 and .04, respectively).
One factor favorably associated with survival included ECOG performance status 0 (OR, 2.74; P = .003). The investigators also found that treatment-related adverse events (AEs) were associated with longer overall survival, with a median of 19.8 months for patients with any grade of treatment-related event and 20.3 months for patients with grade 3 or greater events, compared with a median of 5.8 months for patients with no treatment-related events (P less than .001 for each comparison based on hazard ratios).
“Of note, patients in our study who developed treatment-related AEs, regardless of whether the AEs were deemed to have an immune-mediated causality, had significantly higher ORRs [overall response rates] and prolonged 5-year OS. These findings are reminiscent of some reports of anti–CTLA-4 therapy and align with other studies of anti–PD-1 therapies, “ Dr. Topalian and associates wrote.
The study and the secondary analysis were supported by Bristol-Myers Squibb. Dr. Topalian disclosed grants and travel reimbursements from Bristol-Myers Squibb and consulting fees with other entities. Multiple co-authors reported similar relationships. Four of the co-authors are Bristol-Myers Squibb employees.
SOURCE: Topalian SL et al. JAMA Oncology. 2019 Jul 25. doi: 10.1001/jamaoncol.2019.2187.
FROM JAMA ONCOLOGY
Cases of pediatric invasive melanoma have declined since 2002, study finds
AUSTIN – The compared with females. The risk of death is also significantly increased in black patients, other nonwhite patients, and in cases where surgery was not performed.
Those are key findings from a study that set out to investigate the incidence of pediatric melanoma over the last 2 decades and factors influencing survival. At the annual meeting of the Society for Pediatric Dermatology, one of the study authors, Spandana Maddukuri, said that pediatric melanoma is the most common skin cancer in the pediatric population, accounting for 1-3% of all pediatric malignancies and 1%-4% of all cases of melanoma (Pediatr Surg. 2013;48[11]:2207-13).
“Nonmodifiable risk factors are similar to those in adult melanoma and include fair skin, light hair and eye color, increased number of congenital nevi, and family history of melanoma,” said Ms. Maddukuri, a third-year student at New Jersey Medical School, Newark. “Environmental risk factors are similar to those in adult melanoma and include exposure to UV radiation. About 60% of children do not meet standard ABCDE [asymmetrical, border, color, diameter, evolving] diagnosis criteria, which often leads to delayed diagnosis.”
Some of the characteristics that are more commonly found in pediatric lesions include amelanosis, bleeding, uniform color, and variable diameter (J Am Acad Dermatol. 2013; 68[6]:913-25).
Ms. Maddukuri and colleagues queried the National Cancer Institute Surveillance, Epidemiology, and End Results (SEER) database for cases of malignant melanoma that were diagnosed in individuals aged younger than 20 years between 2002 and 2015. After excluding all cases of adult melanoma and all cases of in situ melanoma, they included 1,620 patients in the final analysis and divided them into five age groups: less than 1 year, 1-4 years, 5-9 years, 10-14 years, and 15-19 years. They calculated the overall incidence rate per 100,000 population of pediatric melanoma based on data from the 2000 U.S. Census. Age-, sex-, and race-specific incidence rates were also calculated. Kaplan-Meier and Cox regression analyses to investigate disease-specific survival and risk factors.
With each successive age group, the investigators observed that incidence rate was significantly higher than that of the previous age group (P less than .005). “However, the most striking increase in incidence occurs between the age group of 10-14 and 15-19,” she said. “Sex also influenced incidence rates. Males had an incidence rate of 0.396 per 100,000 population while females had an incidence rate of 0.579 per 100,000 population.”
Race also influenced incidence rates. White patients had the highest incidence rate of 0.605 per 100,000 population, while blacks had the lowest incident rate at 0.042 per 100,000 population. American Indian and Alaska Native patients had incidence rates of 0.046 per 100,000 population, while Asians and Pacific Islanders had an incidence rate of 0.127 per 100,000 population.
The researchers found that increased survival was associated with white race, female sex, treatment with surgical intervention, and age older than 5 years. No differences in survival were observed regarding the primary anatomic location or extent of disease. The hazard ratio of death from invasive melanoma was significantly increased in males (HR, 2.34), black patients (HR, 3.96), other nonwhite patients (HR, 3.64), and in cases where surgery was not performed (HR, 6.04).
“It is surprising that, although incidence is significantly higher in white patients and females, compared to black patients and males, respectively, the risk of dying from melanoma is much higher in black patients and males,” Ms. Maddukuri said in an interview at the meeting. “Overall, the dermatologic community is on the right track in screening and diagnosing pediatric melanoma, as seen by the decreased incidence over the last 2 decades. However, increased awareness regarding pediatric melanoma is still encouraged. I believe we were able to identify certain populations that need more attention in terms of screening, diagnosis, and treatment, which are patients less than 5 years old, black and other nonwhite patients, and males.”
She acknowledged certain shortcomings of the study, including a limited clinical history of the patient population because of the nature of the database. She also said that further studies are required to investigate the contributing factors to decreasing incidence and to evaluate the relationship of the favorable prognostic factors to increased survival. The researchers are currently working on correlating incidence rates with UV exposure and geographical location.
They reported having no financial disclosures.
AUSTIN – The compared with females. The risk of death is also significantly increased in black patients, other nonwhite patients, and in cases where surgery was not performed.
Those are key findings from a study that set out to investigate the incidence of pediatric melanoma over the last 2 decades and factors influencing survival. At the annual meeting of the Society for Pediatric Dermatology, one of the study authors, Spandana Maddukuri, said that pediatric melanoma is the most common skin cancer in the pediatric population, accounting for 1-3% of all pediatric malignancies and 1%-4% of all cases of melanoma (Pediatr Surg. 2013;48[11]:2207-13).
“Nonmodifiable risk factors are similar to those in adult melanoma and include fair skin, light hair and eye color, increased number of congenital nevi, and family history of melanoma,” said Ms. Maddukuri, a third-year student at New Jersey Medical School, Newark. “Environmental risk factors are similar to those in adult melanoma and include exposure to UV radiation. About 60% of children do not meet standard ABCDE [asymmetrical, border, color, diameter, evolving] diagnosis criteria, which often leads to delayed diagnosis.”
Some of the characteristics that are more commonly found in pediatric lesions include amelanosis, bleeding, uniform color, and variable diameter (J Am Acad Dermatol. 2013; 68[6]:913-25).
Ms. Maddukuri and colleagues queried the National Cancer Institute Surveillance, Epidemiology, and End Results (SEER) database for cases of malignant melanoma that were diagnosed in individuals aged younger than 20 years between 2002 and 2015. After excluding all cases of adult melanoma and all cases of in situ melanoma, they included 1,620 patients in the final analysis and divided them into five age groups: less than 1 year, 1-4 years, 5-9 years, 10-14 years, and 15-19 years. They calculated the overall incidence rate per 100,000 population of pediatric melanoma based on data from the 2000 U.S. Census. Age-, sex-, and race-specific incidence rates were also calculated. Kaplan-Meier and Cox regression analyses to investigate disease-specific survival and risk factors.
With each successive age group, the investigators observed that incidence rate was significantly higher than that of the previous age group (P less than .005). “However, the most striking increase in incidence occurs between the age group of 10-14 and 15-19,” she said. “Sex also influenced incidence rates. Males had an incidence rate of 0.396 per 100,000 population while females had an incidence rate of 0.579 per 100,000 population.”
Race also influenced incidence rates. White patients had the highest incidence rate of 0.605 per 100,000 population, while blacks had the lowest incident rate at 0.042 per 100,000 population. American Indian and Alaska Native patients had incidence rates of 0.046 per 100,000 population, while Asians and Pacific Islanders had an incidence rate of 0.127 per 100,000 population.
The researchers found that increased survival was associated with white race, female sex, treatment with surgical intervention, and age older than 5 years. No differences in survival were observed regarding the primary anatomic location or extent of disease. The hazard ratio of death from invasive melanoma was significantly increased in males (HR, 2.34), black patients (HR, 3.96), other nonwhite patients (HR, 3.64), and in cases where surgery was not performed (HR, 6.04).
“It is surprising that, although incidence is significantly higher in white patients and females, compared to black patients and males, respectively, the risk of dying from melanoma is much higher in black patients and males,” Ms. Maddukuri said in an interview at the meeting. “Overall, the dermatologic community is on the right track in screening and diagnosing pediatric melanoma, as seen by the decreased incidence over the last 2 decades. However, increased awareness regarding pediatric melanoma is still encouraged. I believe we were able to identify certain populations that need more attention in terms of screening, diagnosis, and treatment, which are patients less than 5 years old, black and other nonwhite patients, and males.”
She acknowledged certain shortcomings of the study, including a limited clinical history of the patient population because of the nature of the database. She also said that further studies are required to investigate the contributing factors to decreasing incidence and to evaluate the relationship of the favorable prognostic factors to increased survival. The researchers are currently working on correlating incidence rates with UV exposure and geographical location.
They reported having no financial disclosures.
AUSTIN – The compared with females. The risk of death is also significantly increased in black patients, other nonwhite patients, and in cases where surgery was not performed.
Those are key findings from a study that set out to investigate the incidence of pediatric melanoma over the last 2 decades and factors influencing survival. At the annual meeting of the Society for Pediatric Dermatology, one of the study authors, Spandana Maddukuri, said that pediatric melanoma is the most common skin cancer in the pediatric population, accounting for 1-3% of all pediatric malignancies and 1%-4% of all cases of melanoma (Pediatr Surg. 2013;48[11]:2207-13).
“Nonmodifiable risk factors are similar to those in adult melanoma and include fair skin, light hair and eye color, increased number of congenital nevi, and family history of melanoma,” said Ms. Maddukuri, a third-year student at New Jersey Medical School, Newark. “Environmental risk factors are similar to those in adult melanoma and include exposure to UV radiation. About 60% of children do not meet standard ABCDE [asymmetrical, border, color, diameter, evolving] diagnosis criteria, which often leads to delayed diagnosis.”
Some of the characteristics that are more commonly found in pediatric lesions include amelanosis, bleeding, uniform color, and variable diameter (J Am Acad Dermatol. 2013; 68[6]:913-25).
Ms. Maddukuri and colleagues queried the National Cancer Institute Surveillance, Epidemiology, and End Results (SEER) database for cases of malignant melanoma that were diagnosed in individuals aged younger than 20 years between 2002 and 2015. After excluding all cases of adult melanoma and all cases of in situ melanoma, they included 1,620 patients in the final analysis and divided them into five age groups: less than 1 year, 1-4 years, 5-9 years, 10-14 years, and 15-19 years. They calculated the overall incidence rate per 100,000 population of pediatric melanoma based on data from the 2000 U.S. Census. Age-, sex-, and race-specific incidence rates were also calculated. Kaplan-Meier and Cox regression analyses to investigate disease-specific survival and risk factors.
With each successive age group, the investigators observed that incidence rate was significantly higher than that of the previous age group (P less than .005). “However, the most striking increase in incidence occurs between the age group of 10-14 and 15-19,” she said. “Sex also influenced incidence rates. Males had an incidence rate of 0.396 per 100,000 population while females had an incidence rate of 0.579 per 100,000 population.”
Race also influenced incidence rates. White patients had the highest incidence rate of 0.605 per 100,000 population, while blacks had the lowest incident rate at 0.042 per 100,000 population. American Indian and Alaska Native patients had incidence rates of 0.046 per 100,000 population, while Asians and Pacific Islanders had an incidence rate of 0.127 per 100,000 population.
The researchers found that increased survival was associated with white race, female sex, treatment with surgical intervention, and age older than 5 years. No differences in survival were observed regarding the primary anatomic location or extent of disease. The hazard ratio of death from invasive melanoma was significantly increased in males (HR, 2.34), black patients (HR, 3.96), other nonwhite patients (HR, 3.64), and in cases where surgery was not performed (HR, 6.04).
“It is surprising that, although incidence is significantly higher in white patients and females, compared to black patients and males, respectively, the risk of dying from melanoma is much higher in black patients and males,” Ms. Maddukuri said in an interview at the meeting. “Overall, the dermatologic community is on the right track in screening and diagnosing pediatric melanoma, as seen by the decreased incidence over the last 2 decades. However, increased awareness regarding pediatric melanoma is still encouraged. I believe we were able to identify certain populations that need more attention in terms of screening, diagnosis, and treatment, which are patients less than 5 years old, black and other nonwhite patients, and males.”
She acknowledged certain shortcomings of the study, including a limited clinical history of the patient population because of the nature of the database. She also said that further studies are required to investigate the contributing factors to decreasing incidence and to evaluate the relationship of the favorable prognostic factors to increased survival. The researchers are currently working on correlating incidence rates with UV exposure and geographical location.
They reported having no financial disclosures.
REPORTING FROM SPD 2019