User login
COVID-19 vaccines are safe and effective for patients with migraine
according to a presentation at the American Headache Society’s 2021 annual meeting.
Amy Gelfand, MD, director of pediatric headache at University of California, San Francisco, reviewed common concerns migraine patients or their clinicians might have related any of the three vaccines, starting with a review of how the vaccines work – by targeting the spike protein of the SARS-CoV-2 virus.
“The vaccines induce response to that protein, but only that protein, so there’s no reason to think they’re going to cause the body to produce neutralizing antibodies against any of our migraine therapeutics,” Dr. Gelfand said. She added that the phase 3 clinical trials included participants from a wide range of ages and comorbidities, so there were likely many people in the trials who have migraine, though no subgroup analyses have been performed for this group or are likely to be performed.
Common questions
The two treatments people have the most questions about concerning the COVID-19 vaccine are onabotulinumtoxinA and CGRP pathway monoclonal antibodies (mAbs), likely because both of these are injections, as is the vaccine, Dr. Gelfand said. First, she reminded attendees that onabotulinumtoxinA is not a dermal filler, since some reports following administration of the Moderna vaccine suggested that some people with dermal fillers had swelling in those areas after vaccination.
In addition, “there’s no reason to think the onabotulinumtoxinA would influence our body’s immune response to any vaccine, so there’s no need to retime the onabotulinumtoxinA injections around COVID-19 vaccine administration,” Dr. Gelfand said.
Regarding mAbs, she acknowledged that some white blood cells have CGRP receptors, which may have a pro- or anti-inflammatory role, but clinical trials of mAbs did not show any evidence of being immunosuppressive or myelosuppressive.
“The monoclonal antibodies themselves have undergone engineering so that they are just going after their one target,” Dr. Gelfand said. “They’re not going to be expected to bind to anything else outside of their targets, so I don’t think there’s anything there to make us retime the monoclonal antibody administration relative to the COVID-19 vaccine.”
She did note that patients who choose to get mAbs injections in their arm instead of their thigh or abdomen may want to receive it in the opposite arm than they one they have gotten or will get the vaccine in since the vaccine can cause discomfort.
The other common question patients may have is whether taking any NSAIDs or acetaminophen before getting the COVID-19 vaccine will reduce their immune response to the vaccination. This concern arises because of past evidence showing that some infants tended to have lower immunologic responses when they received acetaminophen after their primary vaccines’ series, but the clinical significance of those reduced responses is not clear since they still had strong responses. Further, this effect was not seen with booster shots, suggesting it’s an age-dependent effect.
During the clinical trials of the AstraZeneca vaccine, several sites gave prophylactic paracetamol without any apparent detrimental effect on antibody response, Dr. Gelfand said. Further, the mRNA and adenovirus-vectored vaccines appear to induce antibodies far above what many believe is needed for protection.
“Even if there were a slight decrease, it’s not clear that that would have any kind of clinical significance for that person in terms of their level of protection against COVID-19,” she said. “Bottom line, it’s fine for patients to use either of these after administration of the COVID-19 vaccine.” The Centers for Disease Control and Prevention doesn’t recommend it prophylactically beforehand, but it’s fine to take it for a fever, aches or headache after getting the vaccine.
Migraine or vaccine reaction?
Dr. Gelfand then addressed whether it should affect physicians’ headache differential if seeing a patient who recently received an adenovirus-vectored vaccine, such as the Johnson & Johnson or AstraZeneca vaccines. The question relates to the discovery of a very rare potential adverse event from these vaccines: cerebral venous sinus thrombosis (CVST) with thrombocytopenia and thromboses in other major vessels, together called thrombosis thrombocytopenia syndrome (TTS). No TTS cases have been reported following mRNA vaccines.
TTS’s mechanism appears similar to autoimmune heparin-induced thrombocytopenia, where the body produces platelet-activating antibodies. TTS currently has three diagnostic criteria: new-onset thrombocytopenia (<150,000/microliter) without evidence of platelet clumping, venous or arterial thrombosis, and absence of prior exposure to heparin.
So far, TTS has been limited only to the vaccines that use an adenovirus vector. One male clinical trial participant experienced CVST with thrombocytopenia in Johnson & Johnson phase 3 trials, and 12 cases out of approximately 8 million Johnson & Johnson doses were reported to the Vaccine Adverse Event Reporting System between March 2 and April 21, 2021. Three TTS more cases followed these, resulting in 15 TTS events per 8 million doses.
In terms of clinical features, all 15 cases were females under age 60, mostly white, and all 11 who were tested were positive for the heparin-platelet factor 4 antibody test. TTS occurred 6-15 days after vaccination for these cases, and all but one had a headache. Their platelet count was 9,000-127,000. None were pregnant or postpartum.
“For us, as headache clinicians, the epidemiology of TTS overlaps with the epidemiology of migraine – they’re happening to the same group of patients,” Dr. Gelfand said. Most of the cases occurred in women aged 30-39 years, while the estimated incidence in women aged 50 or older is 0.9 cases per million doses.
The CDC has proceeded with the Johnson & Johnson vaccine because a risk-benefit analysis revealed that use of the vaccine will result in fewer hospitalization and deaths from COVID-19, compared with adverse events from the vaccine, Dr. Gelfand explained. However, the CDC notes that “women younger than 50 years old should be made aware of a rare risk of blood clots with low platelets following vaccination and the availability of other COVID-19 vaccines where this risk has not been observed.”
For clinicians, the existence of TTS raises a question when patients with a history of migraine call after having received the Johnson & Johnson vaccine, Dr. Gelfand said: “How do we know if this is a spontaneous attack, if it’s a headache provoked by receiving the vaccine, or they have one of these rare cases of [TTS]?”
Three things help with this differential, she said: timing, epidemiology, and headache phenotype. Headache after a vaccine is very common, but it usually happens within the first couple of hours or days after the vaccine. By day 4 after vaccination, few people had headaches in the clinical trials. Since TTS requires production of antibodies, a headache within a few hours of vaccination should not raise concerns about TTS. It should be considered, however, for patients who experience a headache within a week or 2 after vaccination.
Then consider the epidemiology: If it’s a woman between ages 18 and49 calling, the risk is higher than if it’s a male over age 50. Then consider whether there are any unusual headache features, positionality, encephalopathy, or clinical features that could suggest clots in other parts of the body, such as abdominal pain, shortness of breath, or pain in the legs.
“At the end of the day, if it’s a person who’s in this epidemiological window and they’re calling a week or 2 out from the Johnson & Johnson vaccine, we may just need to work it up and see,” Dr. Gelfand said. Work-up involves a CBC, a platelet count to see if they’re thrombocytopenic, and perhaps imaging, preferentially using MRI/MRV over CT since it’s a younger population. Treatment for CVST with thrombocytopenia is a nonheparin anticoagulant, and platelet transfusion should not occur before consulting with hematology.
Continue to vaccinate
“The big take home is that we should continue to vaccinate patients with migraine and that your current therapies do not interfere with the vaccine working and that the vaccine does not interact with our therapies,” Brian D. Loftus, MD, BSChE, immediate past president of the Southern Headache Society and a neurologist at Bellaire (Pa.) Neurology, said of the presentation. He also felt it was helpful to know that NSAIDs likely have no impact on the vaccines’ effectiveness as well.
“The most important new information for me was that the median onset of the CSVT was 8 days post vaccine,” Dr. Loftus said. “Typically, postvaccine headache is seen much sooner, within 1-2 days, so this is a useful clinical feature to separate out who needs to closer follow-up and possible neuroimaging.”
Given the epidemiology of those most likely to have TTS, Dr. Loftus said he would advise his female patients younger than 60 to simply get the Pfizer or Moderna vaccine since they appear safer for this demographic.
Dr. Gelfand is editor of the journal Headache but has no industry disclosures. Her spouse has received clinical trial grant support from Genentech and honoraria for editorial work from Dynamed Plus. Dr. Loftus has received grants or fees from Teva, Amgen, Abbvie, and Biohaven.
according to a presentation at the American Headache Society’s 2021 annual meeting.
Amy Gelfand, MD, director of pediatric headache at University of California, San Francisco, reviewed common concerns migraine patients or their clinicians might have related any of the three vaccines, starting with a review of how the vaccines work – by targeting the spike protein of the SARS-CoV-2 virus.
“The vaccines induce response to that protein, but only that protein, so there’s no reason to think they’re going to cause the body to produce neutralizing antibodies against any of our migraine therapeutics,” Dr. Gelfand said. She added that the phase 3 clinical trials included participants from a wide range of ages and comorbidities, so there were likely many people in the trials who have migraine, though no subgroup analyses have been performed for this group or are likely to be performed.
Common questions
The two treatments people have the most questions about concerning the COVID-19 vaccine are onabotulinumtoxinA and CGRP pathway monoclonal antibodies (mAbs), likely because both of these are injections, as is the vaccine, Dr. Gelfand said. First, she reminded attendees that onabotulinumtoxinA is not a dermal filler, since some reports following administration of the Moderna vaccine suggested that some people with dermal fillers had swelling in those areas after vaccination.
In addition, “there’s no reason to think the onabotulinumtoxinA would influence our body’s immune response to any vaccine, so there’s no need to retime the onabotulinumtoxinA injections around COVID-19 vaccine administration,” Dr. Gelfand said.
Regarding mAbs, she acknowledged that some white blood cells have CGRP receptors, which may have a pro- or anti-inflammatory role, but clinical trials of mAbs did not show any evidence of being immunosuppressive or myelosuppressive.
“The monoclonal antibodies themselves have undergone engineering so that they are just going after their one target,” Dr. Gelfand said. “They’re not going to be expected to bind to anything else outside of their targets, so I don’t think there’s anything there to make us retime the monoclonal antibody administration relative to the COVID-19 vaccine.”
She did note that patients who choose to get mAbs injections in their arm instead of their thigh or abdomen may want to receive it in the opposite arm than they one they have gotten or will get the vaccine in since the vaccine can cause discomfort.
The other common question patients may have is whether taking any NSAIDs or acetaminophen before getting the COVID-19 vaccine will reduce their immune response to the vaccination. This concern arises because of past evidence showing that some infants tended to have lower immunologic responses when they received acetaminophen after their primary vaccines’ series, but the clinical significance of those reduced responses is not clear since they still had strong responses. Further, this effect was not seen with booster shots, suggesting it’s an age-dependent effect.
During the clinical trials of the AstraZeneca vaccine, several sites gave prophylactic paracetamol without any apparent detrimental effect on antibody response, Dr. Gelfand said. Further, the mRNA and adenovirus-vectored vaccines appear to induce antibodies far above what many believe is needed for protection.
“Even if there were a slight decrease, it’s not clear that that would have any kind of clinical significance for that person in terms of their level of protection against COVID-19,” she said. “Bottom line, it’s fine for patients to use either of these after administration of the COVID-19 vaccine.” The Centers for Disease Control and Prevention doesn’t recommend it prophylactically beforehand, but it’s fine to take it for a fever, aches or headache after getting the vaccine.
Migraine or vaccine reaction?
Dr. Gelfand then addressed whether it should affect physicians’ headache differential if seeing a patient who recently received an adenovirus-vectored vaccine, such as the Johnson & Johnson or AstraZeneca vaccines. The question relates to the discovery of a very rare potential adverse event from these vaccines: cerebral venous sinus thrombosis (CVST) with thrombocytopenia and thromboses in other major vessels, together called thrombosis thrombocytopenia syndrome (TTS). No TTS cases have been reported following mRNA vaccines.
TTS’s mechanism appears similar to autoimmune heparin-induced thrombocytopenia, where the body produces platelet-activating antibodies. TTS currently has three diagnostic criteria: new-onset thrombocytopenia (<150,000/microliter) without evidence of platelet clumping, venous or arterial thrombosis, and absence of prior exposure to heparin.
So far, TTS has been limited only to the vaccines that use an adenovirus vector. One male clinical trial participant experienced CVST with thrombocytopenia in Johnson & Johnson phase 3 trials, and 12 cases out of approximately 8 million Johnson & Johnson doses were reported to the Vaccine Adverse Event Reporting System between March 2 and April 21, 2021. Three TTS more cases followed these, resulting in 15 TTS events per 8 million doses.
In terms of clinical features, all 15 cases were females under age 60, mostly white, and all 11 who were tested were positive for the heparin-platelet factor 4 antibody test. TTS occurred 6-15 days after vaccination for these cases, and all but one had a headache. Their platelet count was 9,000-127,000. None were pregnant or postpartum.
“For us, as headache clinicians, the epidemiology of TTS overlaps with the epidemiology of migraine – they’re happening to the same group of patients,” Dr. Gelfand said. Most of the cases occurred in women aged 30-39 years, while the estimated incidence in women aged 50 or older is 0.9 cases per million doses.
The CDC has proceeded with the Johnson & Johnson vaccine because a risk-benefit analysis revealed that use of the vaccine will result in fewer hospitalization and deaths from COVID-19, compared with adverse events from the vaccine, Dr. Gelfand explained. However, the CDC notes that “women younger than 50 years old should be made aware of a rare risk of blood clots with low platelets following vaccination and the availability of other COVID-19 vaccines where this risk has not been observed.”
For clinicians, the existence of TTS raises a question when patients with a history of migraine call after having received the Johnson & Johnson vaccine, Dr. Gelfand said: “How do we know if this is a spontaneous attack, if it’s a headache provoked by receiving the vaccine, or they have one of these rare cases of [TTS]?”
Three things help with this differential, she said: timing, epidemiology, and headache phenotype. Headache after a vaccine is very common, but it usually happens within the first couple of hours or days after the vaccine. By day 4 after vaccination, few people had headaches in the clinical trials. Since TTS requires production of antibodies, a headache within a few hours of vaccination should not raise concerns about TTS. It should be considered, however, for patients who experience a headache within a week or 2 after vaccination.
Then consider the epidemiology: If it’s a woman between ages 18 and49 calling, the risk is higher than if it’s a male over age 50. Then consider whether there are any unusual headache features, positionality, encephalopathy, or clinical features that could suggest clots in other parts of the body, such as abdominal pain, shortness of breath, or pain in the legs.
“At the end of the day, if it’s a person who’s in this epidemiological window and they’re calling a week or 2 out from the Johnson & Johnson vaccine, we may just need to work it up and see,” Dr. Gelfand said. Work-up involves a CBC, a platelet count to see if they’re thrombocytopenic, and perhaps imaging, preferentially using MRI/MRV over CT since it’s a younger population. Treatment for CVST with thrombocytopenia is a nonheparin anticoagulant, and platelet transfusion should not occur before consulting with hematology.
Continue to vaccinate
“The big take home is that we should continue to vaccinate patients with migraine and that your current therapies do not interfere with the vaccine working and that the vaccine does not interact with our therapies,” Brian D. Loftus, MD, BSChE, immediate past president of the Southern Headache Society and a neurologist at Bellaire (Pa.) Neurology, said of the presentation. He also felt it was helpful to know that NSAIDs likely have no impact on the vaccines’ effectiveness as well.
“The most important new information for me was that the median onset of the CSVT was 8 days post vaccine,” Dr. Loftus said. “Typically, postvaccine headache is seen much sooner, within 1-2 days, so this is a useful clinical feature to separate out who needs to closer follow-up and possible neuroimaging.”
Given the epidemiology of those most likely to have TTS, Dr. Loftus said he would advise his female patients younger than 60 to simply get the Pfizer or Moderna vaccine since they appear safer for this demographic.
Dr. Gelfand is editor of the journal Headache but has no industry disclosures. Her spouse has received clinical trial grant support from Genentech and honoraria for editorial work from Dynamed Plus. Dr. Loftus has received grants or fees from Teva, Amgen, Abbvie, and Biohaven.
according to a presentation at the American Headache Society’s 2021 annual meeting.
Amy Gelfand, MD, director of pediatric headache at University of California, San Francisco, reviewed common concerns migraine patients or their clinicians might have related any of the three vaccines, starting with a review of how the vaccines work – by targeting the spike protein of the SARS-CoV-2 virus.
“The vaccines induce response to that protein, but only that protein, so there’s no reason to think they’re going to cause the body to produce neutralizing antibodies against any of our migraine therapeutics,” Dr. Gelfand said. She added that the phase 3 clinical trials included participants from a wide range of ages and comorbidities, so there were likely many people in the trials who have migraine, though no subgroup analyses have been performed for this group or are likely to be performed.
Common questions
The two treatments people have the most questions about concerning the COVID-19 vaccine are onabotulinumtoxinA and CGRP pathway monoclonal antibodies (mAbs), likely because both of these are injections, as is the vaccine, Dr. Gelfand said. First, she reminded attendees that onabotulinumtoxinA is not a dermal filler, since some reports following administration of the Moderna vaccine suggested that some people with dermal fillers had swelling in those areas after vaccination.
In addition, “there’s no reason to think the onabotulinumtoxinA would influence our body’s immune response to any vaccine, so there’s no need to retime the onabotulinumtoxinA injections around COVID-19 vaccine administration,” Dr. Gelfand said.
Regarding mAbs, she acknowledged that some white blood cells have CGRP receptors, which may have a pro- or anti-inflammatory role, but clinical trials of mAbs did not show any evidence of being immunosuppressive or myelosuppressive.
“The monoclonal antibodies themselves have undergone engineering so that they are just going after their one target,” Dr. Gelfand said. “They’re not going to be expected to bind to anything else outside of their targets, so I don’t think there’s anything there to make us retime the monoclonal antibody administration relative to the COVID-19 vaccine.”
She did note that patients who choose to get mAbs injections in their arm instead of their thigh or abdomen may want to receive it in the opposite arm than they one they have gotten or will get the vaccine in since the vaccine can cause discomfort.
The other common question patients may have is whether taking any NSAIDs or acetaminophen before getting the COVID-19 vaccine will reduce their immune response to the vaccination. This concern arises because of past evidence showing that some infants tended to have lower immunologic responses when they received acetaminophen after their primary vaccines’ series, but the clinical significance of those reduced responses is not clear since they still had strong responses. Further, this effect was not seen with booster shots, suggesting it’s an age-dependent effect.
During the clinical trials of the AstraZeneca vaccine, several sites gave prophylactic paracetamol without any apparent detrimental effect on antibody response, Dr. Gelfand said. Further, the mRNA and adenovirus-vectored vaccines appear to induce antibodies far above what many believe is needed for protection.
“Even if there were a slight decrease, it’s not clear that that would have any kind of clinical significance for that person in terms of their level of protection against COVID-19,” she said. “Bottom line, it’s fine for patients to use either of these after administration of the COVID-19 vaccine.” The Centers for Disease Control and Prevention doesn’t recommend it prophylactically beforehand, but it’s fine to take it for a fever, aches or headache after getting the vaccine.
Migraine or vaccine reaction?
Dr. Gelfand then addressed whether it should affect physicians’ headache differential if seeing a patient who recently received an adenovirus-vectored vaccine, such as the Johnson & Johnson or AstraZeneca vaccines. The question relates to the discovery of a very rare potential adverse event from these vaccines: cerebral venous sinus thrombosis (CVST) with thrombocytopenia and thromboses in other major vessels, together called thrombosis thrombocytopenia syndrome (TTS). No TTS cases have been reported following mRNA vaccines.
TTS’s mechanism appears similar to autoimmune heparin-induced thrombocytopenia, where the body produces platelet-activating antibodies. TTS currently has three diagnostic criteria: new-onset thrombocytopenia (<150,000/microliter) without evidence of platelet clumping, venous or arterial thrombosis, and absence of prior exposure to heparin.
So far, TTS has been limited only to the vaccines that use an adenovirus vector. One male clinical trial participant experienced CVST with thrombocytopenia in Johnson & Johnson phase 3 trials, and 12 cases out of approximately 8 million Johnson & Johnson doses were reported to the Vaccine Adverse Event Reporting System between March 2 and April 21, 2021. Three TTS more cases followed these, resulting in 15 TTS events per 8 million doses.
In terms of clinical features, all 15 cases were females under age 60, mostly white, and all 11 who were tested were positive for the heparin-platelet factor 4 antibody test. TTS occurred 6-15 days after vaccination for these cases, and all but one had a headache. Their platelet count was 9,000-127,000. None were pregnant or postpartum.
“For us, as headache clinicians, the epidemiology of TTS overlaps with the epidemiology of migraine – they’re happening to the same group of patients,” Dr. Gelfand said. Most of the cases occurred in women aged 30-39 years, while the estimated incidence in women aged 50 or older is 0.9 cases per million doses.
The CDC has proceeded with the Johnson & Johnson vaccine because a risk-benefit analysis revealed that use of the vaccine will result in fewer hospitalization and deaths from COVID-19, compared with adverse events from the vaccine, Dr. Gelfand explained. However, the CDC notes that “women younger than 50 years old should be made aware of a rare risk of blood clots with low platelets following vaccination and the availability of other COVID-19 vaccines where this risk has not been observed.”
For clinicians, the existence of TTS raises a question when patients with a history of migraine call after having received the Johnson & Johnson vaccine, Dr. Gelfand said: “How do we know if this is a spontaneous attack, if it’s a headache provoked by receiving the vaccine, or they have one of these rare cases of [TTS]?”
Three things help with this differential, she said: timing, epidemiology, and headache phenotype. Headache after a vaccine is very common, but it usually happens within the first couple of hours or days after the vaccine. By day 4 after vaccination, few people had headaches in the clinical trials. Since TTS requires production of antibodies, a headache within a few hours of vaccination should not raise concerns about TTS. It should be considered, however, for patients who experience a headache within a week or 2 after vaccination.
Then consider the epidemiology: If it’s a woman between ages 18 and49 calling, the risk is higher than if it’s a male over age 50. Then consider whether there are any unusual headache features, positionality, encephalopathy, or clinical features that could suggest clots in other parts of the body, such as abdominal pain, shortness of breath, or pain in the legs.
“At the end of the day, if it’s a person who’s in this epidemiological window and they’re calling a week or 2 out from the Johnson & Johnson vaccine, we may just need to work it up and see,” Dr. Gelfand said. Work-up involves a CBC, a platelet count to see if they’re thrombocytopenic, and perhaps imaging, preferentially using MRI/MRV over CT since it’s a younger population. Treatment for CVST with thrombocytopenia is a nonheparin anticoagulant, and platelet transfusion should not occur before consulting with hematology.
Continue to vaccinate
“The big take home is that we should continue to vaccinate patients with migraine and that your current therapies do not interfere with the vaccine working and that the vaccine does not interact with our therapies,” Brian D. Loftus, MD, BSChE, immediate past president of the Southern Headache Society and a neurologist at Bellaire (Pa.) Neurology, said of the presentation. He also felt it was helpful to know that NSAIDs likely have no impact on the vaccines’ effectiveness as well.
“The most important new information for me was that the median onset of the CSVT was 8 days post vaccine,” Dr. Loftus said. “Typically, postvaccine headache is seen much sooner, within 1-2 days, so this is a useful clinical feature to separate out who needs to closer follow-up and possible neuroimaging.”
Given the epidemiology of those most likely to have TTS, Dr. Loftus said he would advise his female patients younger than 60 to simply get the Pfizer or Moderna vaccine since they appear safer for this demographic.
Dr. Gelfand is editor of the journal Headache but has no industry disclosures. Her spouse has received clinical trial grant support from Genentech and honoraria for editorial work from Dynamed Plus. Dr. Loftus has received grants or fees from Teva, Amgen, Abbvie, and Biohaven.
FROM AHS 2021
Difficult patient, or something else? A review of personality disorders
Specific behaviors or expressed thoughts may signal a need for screening. Take into account an individual’s strengths and limitations when designing a Tx approach.
THE CASES
Winston S* is a 23-year-old man referred by a psychiatrist colleague for primary care. He works delivering papers in the early morning hours and spends his day alone in his apartment mainly eating frozen pizza. He has worked solitary jobs his entire life and says he prefers it that way. His answers to questions lack emotion. He doesn’t seem to have any friends or regular contact with family. He follows the medical advice he receives but can’t seem to get out of the house to exercise or socialize. His psychiatrist was treating him with a selective serotonin reuptake inhibitor for depression when he was referred.
Denise L* is a 37-year-old woman who transferred to your practice because she says the previous practice’s office manager was disrespectful and the doctor did not listen to her. She has been “very appreciative” of you and your “well-run office.” You have addressed her fibromyalgia and she has shared several personal details about her life. In the following weeks, you receive several phone calls and messages from her. At a follow-up visit, she asks questions about your family and seems agitated when you hesitate to answer. She questions whether you remember details of her history. She pushes, “Did you remember that, doctor?” She also mentions that your front desk staff seems rude to her.
Ruth B* is an 82-year-old woman whose blood pressure measured in your office is 176/94 mm Hg. When you recommend starting a medication and getting blood tests, she responds with a litany of fearful questions. She seems immobilized by worries about treatment and equally so about the risks of nontreatment. You can’t seem to get past the anxiety to decide on a satisfactory plan. She has to write everything down on a notepad and worries if she does not get every detail.
●
* This patient’s name has been changed to protect his identity. The other 2 patients are an amalgam of patients for whom the authors have provided care.
According to a survey of practicing primary care physicians, as many as 15% of patient encounters can be difficult.1 Demanding, intrusive, or angry patients who reject health care interventions are often-cited sources of these difficulties.2,3 While it is true that patient, physician, and environmental factors may contribute to challenging interactions, some patients who are “difficult” may actually have a personality disorder that requires a distinctive approach to care. Recognizing these patients can help empower physicians to provide compassionate and effective care, reduce team angst, and minimize burnout.
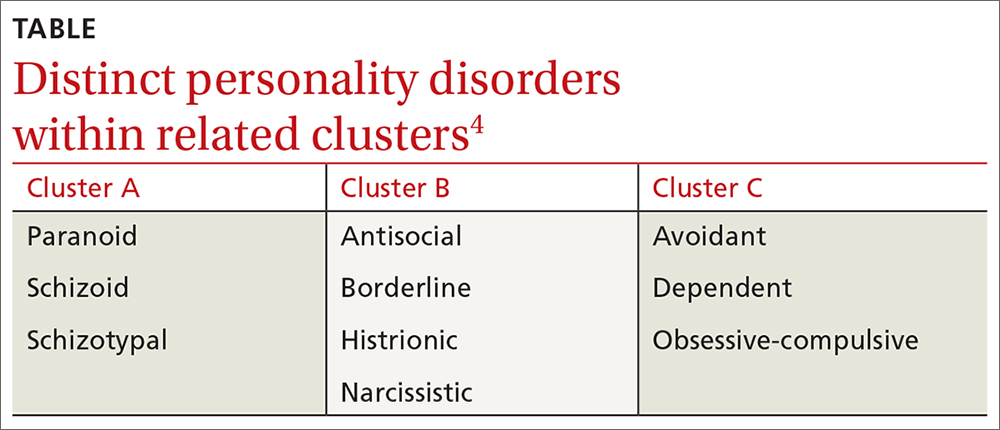
❚ What qualifies as a personality disorder? A personality disorder is an enduring pattern of inner experience and behavior that deviates markedly from the expectations of the individual’s culture, is pervasive and inflexible, has an onset in adolescence or early adulthood, is unchanging over time, and leads to distress or impairment in social or occupational functioning.4 The prevalence of any personality disorder seems to have increased over the past decade from 9.1%4 to 12.16%.5 The Diagnostic and Statistical Manual of Mental Disorders (DSM-5) classifies personality disorders in 3 clusters—A, B, and C (TABLE4)—with prevalence rates at 7.23%, 5.53%, and 6.7%, respectively.5 The review below will focus on the distinct personality disorders exhibited by the patients described in the opening cases.
Continue to: A closer look at the clusters...
A closer look at the clusters
Cluster A disorders
Paranoid, schizoid, and schizotypal disorders are part of this cluster. These patients exhibit odd or eccentric thinking and behavior. Individuals with schizoid personality disorder, for instance, usually lack relationships and lack the desire to acquire and maintain relationships.4 They often organize their lives to remain isolated and will choose occupations that require little social interaction. They sometimes view themselves as observers rather than participants in their own lives.6
Cluster B disorders
Dramatic, overly emotional, or unpredictable thinking and behavior are characteristic of individuals who have antisocial, borderline, histrionic, or narcissistic disorders. Patients with borderline personality disorder (BPD), for example, demonstrate a longstanding pattern of instability in affect, self-image, and relationships.4 Patients with BPD often display extreme interpersonal hypersensitivity and make frantic efforts to avoid real or imagined abandonment. Identity disturbance, feelings of emptiness, and efforts to avoid abandonment have all been associated with increased suicide risk.7
In a primary care setting, such a patient may display extremely strong reactions to minor disappointments. When the physician is unavailable for a last-minute appointment or to authorize an unscheduled medication refill or to receive an after-hours phone call, the patient may become irate. The physician, who previously was idealized by the patient as “the only person who understands me,” is now devalued as “the worst doctor I’ve ever had.”8
Cluster C disorders
With these individuals, anxious or fearful thinking and behavior predominate. Avoidant, dependent, and obsessive-compulsive disorders are included in this cluster.
Dependent personality disorder (DPD) is characterized by a pervasive and extreme need to be taken care of. Submissive and clingy behavior and fear of separation are excessive. This patient may have difficulty making everyday decisions, being assertive, or expressing disagreement with others.4
Obsessive-compulsive personality disorder falls in this cluster and is typified by a pervasive preoccupation with orderliness, perfectionism, and control, at the price of flexibility and efficiency. This individual may be reluctant to get rid of sentimental objects, have rigid moral beliefs, and have significant difficulty working with others who do not follow their rules.4
Continue to: These clues may suggest...
These clues may suggest a personality disorder
If you find that encounters with a particular patient are growing increasingly difficult, consider whether the following behaviors, attitudes, and patterns of thinking are coming into play. If they are, you may want to consider using a screening tool, which we’ll discuss in a moment.
❚ Clues to cluster A disorders
- The patient has no peer relationships outside immediate family.
- The patient almost always chooses solitary activities for work and personal enjoyment.
❚ Cluster B clues
- Hypersensitivity to treatment disagreements or cancelled appointments are common (and likely experienced as rejection).
- Mood changes occur very quickly, even during a single visit.
- There is a history of many failed relationships with providers and others.
- The patient will describe an individual as both “wonderful” and “terrible” (ie, splitting) and may do so during the course of one visit.
- The patient may also split groups (eg, medical staff) by affective extremes (eg, adoration and hatred).
- The patient may hint at suicide or acts of self-harm.7
❚ Cluster C clues
- There is an excessive dependency on family, friends, or providers.
- Significant anxiety is experienced when the patient has to make an independent decision.
- There is a fear of relationship loss and resultant vulnerability to exploitation or abuse.
- Pervasive perfectionism makes treatment planning or course changes difficult.
- Anxiety and fear are unrelieved despite support and ample information.
Consider these screening tools
Several screening tools for personality disorders can be used to follow up on your initial clinical impressions. We also highly recommend you consider concurrent screening for substance abuse, as addiction is a common comorbidity with personality disorders.
❚
❚ A sampling of screening tools. The Standardised Assessment of Personality Abbreviated Scale (SAPAS)9 is an 8-item measure that correlates well with disorders in clusters A and C.
BPD (cluster B) has many brief scale options, including the McLean Screening Instrument for Borderline Personality Disorder (MSI-BPD).10 This 10-item questionnaire demonstrates sensitivity and specificity for BPD.
The International Personality Disorder Examination (IPDE) includes a 15-minute screening tool to help identify patients who may have any personality disorder, regardless of cluster.11
Improve patient encounters with these Tx pearls
In the family medicine clinic, a collaborative primary care and behavioral health team can be extremely helpful in the diagnosis and management of patients with personality disorders.12 First-line treatment of these disorders is psychotherapy, whereas medications are mainly used for symptom management. See Black and colleagues’ work for a thorough discussion on psychopharmacology considerations with personality disorders. 13
The following tips can help you to improve your interactions with patients who have personality disorders.
❚ Cluster A approaches
- Recommend treatment that respects the patient’s need for relative isolation.14
- Don’t be personally offended by your patient’s flat or disinterested affect or concrete thinking; don’t let it diminish the emotional support you provide.6
- Consult with a health psychologist (who has expertise in physical health conditions, brief treatments, and the medical system) to connect the patient with a long-term therapist. It is better to focus on fundamental changes, rather than employing brief behavioral techniques, for symptom relief. Patients with personality disorders tend to have better outcomes with long-term psychological care.15
❚ Cluster B approaches
- Set boundaries—eg, specific time limits for visits—and keep them.8
- Schedule brief, more frequent, appointments to reduce perceived feelings of abandonment.
- Coordinate plans with the entire clinic team to avoid splitting and blaming.16
- Avoid providing patients with personal information, as it may provide fodder for splitting behavior. 8
- Do not take things personally. Let patients “own” their own distress. These patients often take an emotional toll on the provider.16
- Engage the help of a health psychologist to reduce burnout and for more long-term continuity of care. A health psychologist who specializes in dialectical behavioral therapy to work on emotion regulation, distress tolerance, and interpersonal effectiveness would be ideal.17
Continue to: Cluster C approaches...
❚
❚ Cluster C approaches
- Engage the help of family and other trusted individuals in supporting treatment plans.18,19
- Try to provide just 2 treatment choices to the patient and reinforce his or her responsibility to help make the decision collaboratively. This step is important since it is difficult to enhance autonomy in these patients.20
- Engage the help of a cognitive behavioral therapist who can work on assertiveness and problem-solving skills.19
- Be empathetic with the patient and patiently build a trusting relationship, rather than “arguing” with the patient about each specific worry.20
- Make only one change at a time. Give small assignments to the patient, such as monitoring symptoms or reading up on their condition. These can help the patient feel more in control.21
- Present information in brief, clear terms. Avoid “grey areas” to reduce anxiety.21
- Engage a behavioral health provider to reduce rigid expectations and ideally increase feelings of self-esteem; this has been shown to predict better treatment outcomes.22
CASES
Mr. S displays cluster-A characteristics of schizoid personality disorder in addition to the depression he is being treated for. His physician was not put off by his flat affect and respected his limitations with social activities. Use of a stationary bike was recommended for exercise rather than walks outdoors. He also preferred phone calls to in-person encounters, so his follow-up visits were conducted by phone.
Ms. L exhibits cluster-B characteristics of BPD. You begin the tricky dance of setting limits, keeping communication clear, and not blaming yourself or others on your team for Ms. L’s feelings. You schedule regular visits with explicit time limits and discuss with your entire team how to avoid splitting. You involve a psychologist, familiar with treating BPD, who helps the patient learn positive interpersonal coping skills.
Ms. B displays cluster-C characteristics of dependent and obsessive-compulsive personality disorders. At her follow-up visit, you provide a great deal of empathy and try not to argue her out of each worry that she brings up. You make one change at a time and enlist the help of her daughter in giving her pills at home and offering reassurance. You collaborate with a cognitive behavioral therapist who works on exposing her to moderately anxiety-provoking situations/decisions.
1. Hull SK, Broquet K. How to manage difficult patient encounters. Fam Pract Manag. 2007;14:30-34.
2. Groves JE. Taking care of the hateful patient. N Engl J Med.1978;298: 883-887.
3. O’Dowd TC. Five years of heartsink patients in primary care. BMJ. 1988;297:528-530.
4. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 5th edition (DSM-5). American Psychiatric Publishing; 2013.
5. Volkert J, Gablonski TC, Rabung S. Prevalence of personality disorders in the general adult population in Western countries: systematic review and meta-analysis. Br J Psychiatry. 2018;213:709-715.
6. Esterberg ML, Goulding SM, Walker EF. Cluster A personality disorders: schizotypal, schizoid and paranoid personality disorders in childhood and adolescence. J Psychopathol Behav Assess. 2010;32:515-528.
7. Yen S, Peters JR, Nishar S, et al. Association of borderline personality disorder criteria with suicide attempts: findings from the collaborative longitudinal study of personality disorders over 10 years of follow-up. JAMA Psychiatry. 2021;78:187-194.
8. Dubovsky AN, Kiefer MM. Borderline personality disorder in the primary care setting. Med Clin North Am. 2014;98:1049-1064.
9. Hesse M, Moran P. (2010). Screening for personality disorder with the Standardised Assessment of Personality: Abbreviated Scale (SAPAS): further evidence of concurrent validity. BMC Psychiatry. 2010;10:10.
10. Zanarini MC, Vujanovic AA, Parachini EA, et al. A screening measure for BPD: the McLean screening instrument for borderline personality disorder (MSI-BPD). J Pers Disord. 2003;17:568-573.
11. Loranger AW, Sartorius N, Andreoli A, et al. The International Personality Disorder Examination. The World Health Organization/Alcohol, Drug Abuse, and Mental Health Administration international pilot study of personality disorders. Arch Gen Psychiatry. 1994;51:215-224.12. Nelson KJ, Skodol A, Friedman M. Pharmacotherapy for personality disorders. UpToDate. Accessed April 22, 2021. www.uptodate.com/contents/pharmacotherapy-for-personality-disorders
13. Black D, Paris J, Schulz C. Evidence-based integrated biopsychosocial treatment of borderline personality disorder. In: Muse M (ed). Cognitive Behavioral Psychopharmacology: the Clinical Practice of Evidence-Based Biopsychosocial Integration. Wiley; 2017:137-166.
14. Beck AT, Davis DD, Freeman A. Cognitive Therapy of Personality Disorders. 3rd ed. The Guilford Press; 2015.
15. Thylstrup B, Hesse M. “I am not complaining”–ambivalence construct in schizoid personality disorder. Am J Psychother. 2009;63:147-167.
16. Ricke AK, Lee MJ, Chambers JE. The difficult patient: borderline personality disorder in the obstetrical and gynecological patient. Obstet Gynecol Surv. 2012;67:495-502.
17. Seow LLY, Page AC, Hooke GR. Severity of borderline personality disorder symptoms as a moderator of the association between the use of dialectical behaviour therapy skills and treatment outcomes. Psychother Res. 2020;30:920-933.
18. Nichols WC. Integrative marital and family treatment of dependent personality disorders. In: MacFarlane MM (Ed.) Family Treatment of Personality Disorders: Advances in Clinical Practice. Haworth Clinical Practice Press; 2004:173-204.
19. Disney KL. Dependent personality disorder: a critical review. Clin Psychol Rev. 2013;33:1184-1196.
20. Bender DS. The therapeutic alliance in the treatment of personality disorders. J Psychiatr Pract. 2005;11:73-87.
21. Ward RK. Assessment and management of personality disorders. Am Fam Physician. 2004;70:1505-1512.
22. Cummings JA, Hayes AM, Cardaciotto L, et al. The dynamics of self-esteem in cognitive therapy for avoidant and obsessive-compulsive personality disorders: an adaptive role of self-esteem variability? Cognit Ther Res. 2012;36:272-281.
Specific behaviors or expressed thoughts may signal a need for screening. Take into account an individual’s strengths and limitations when designing a Tx approach.
Specific behaviors or expressed thoughts may signal a need for screening. Take into account an individual’s strengths and limitations when designing a Tx approach.
THE CASES
Winston S* is a 23-year-old man referred by a psychiatrist colleague for primary care. He works delivering papers in the early morning hours and spends his day alone in his apartment mainly eating frozen pizza. He has worked solitary jobs his entire life and says he prefers it that way. His answers to questions lack emotion. He doesn’t seem to have any friends or regular contact with family. He follows the medical advice he receives but can’t seem to get out of the house to exercise or socialize. His psychiatrist was treating him with a selective serotonin reuptake inhibitor for depression when he was referred.
Denise L* is a 37-year-old woman who transferred to your practice because she says the previous practice’s office manager was disrespectful and the doctor did not listen to her. She has been “very appreciative” of you and your “well-run office.” You have addressed her fibromyalgia and she has shared several personal details about her life. In the following weeks, you receive several phone calls and messages from her. At a follow-up visit, she asks questions about your family and seems agitated when you hesitate to answer. She questions whether you remember details of her history. She pushes, “Did you remember that, doctor?” She also mentions that your front desk staff seems rude to her.
Ruth B* is an 82-year-old woman whose blood pressure measured in your office is 176/94 mm Hg. When you recommend starting a medication and getting blood tests, she responds with a litany of fearful questions. She seems immobilized by worries about treatment and equally so about the risks of nontreatment. You can’t seem to get past the anxiety to decide on a satisfactory plan. She has to write everything down on a notepad and worries if she does not get every detail.
●
* This patient’s name has been changed to protect his identity. The other 2 patients are an amalgam of patients for whom the authors have provided care.
According to a survey of practicing primary care physicians, as many as 15% of patient encounters can be difficult.1 Demanding, intrusive, or angry patients who reject health care interventions are often-cited sources of these difficulties.2,3 While it is true that patient, physician, and environmental factors may contribute to challenging interactions, some patients who are “difficult” may actually have a personality disorder that requires a distinctive approach to care. Recognizing these patients can help empower physicians to provide compassionate and effective care, reduce team angst, and minimize burnout.
❚ What qualifies as a personality disorder? A personality disorder is an enduring pattern of inner experience and behavior that deviates markedly from the expectations of the individual’s culture, is pervasive and inflexible, has an onset in adolescence or early adulthood, is unchanging over time, and leads to distress or impairment in social or occupational functioning.4 The prevalence of any personality disorder seems to have increased over the past decade from 9.1%4 to 12.16%.5 The Diagnostic and Statistical Manual of Mental Disorders (DSM-5) classifies personality disorders in 3 clusters—A, B, and C (TABLE4)—with prevalence rates at 7.23%, 5.53%, and 6.7%, respectively.5 The review below will focus on the distinct personality disorders exhibited by the patients described in the opening cases.

Continue to: A closer look at the clusters...
A closer look at the clusters
Cluster A disorders
Paranoid, schizoid, and schizotypal disorders are part of this cluster. These patients exhibit odd or eccentric thinking and behavior. Individuals with schizoid personality disorder, for instance, usually lack relationships and lack the desire to acquire and maintain relationships.4 They often organize their lives to remain isolated and will choose occupations that require little social interaction. They sometimes view themselves as observers rather than participants in their own lives.6
Cluster B disorders
Dramatic, overly emotional, or unpredictable thinking and behavior are characteristic of individuals who have antisocial, borderline, histrionic, or narcissistic disorders. Patients with borderline personality disorder (BPD), for example, demonstrate a longstanding pattern of instability in affect, self-image, and relationships.4 Patients with BPD often display extreme interpersonal hypersensitivity and make frantic efforts to avoid real or imagined abandonment. Identity disturbance, feelings of emptiness, and efforts to avoid abandonment have all been associated with increased suicide risk.7
In a primary care setting, such a patient may display extremely strong reactions to minor disappointments. When the physician is unavailable for a last-minute appointment or to authorize an unscheduled medication refill or to receive an after-hours phone call, the patient may become irate. The physician, who previously was idealized by the patient as “the only person who understands me,” is now devalued as “the worst doctor I’ve ever had.”8
Cluster C disorders
With these individuals, anxious or fearful thinking and behavior predominate. Avoidant, dependent, and obsessive-compulsive disorders are included in this cluster.
Dependent personality disorder (DPD) is characterized by a pervasive and extreme need to be taken care of. Submissive and clingy behavior and fear of separation are excessive. This patient may have difficulty making everyday decisions, being assertive, or expressing disagreement with others.4
Obsessive-compulsive personality disorder falls in this cluster and is typified by a pervasive preoccupation with orderliness, perfectionism, and control, at the price of flexibility and efficiency. This individual may be reluctant to get rid of sentimental objects, have rigid moral beliefs, and have significant difficulty working with others who do not follow their rules.4
Continue to: These clues may suggest...
These clues may suggest a personality disorder
If you find that encounters with a particular patient are growing increasingly difficult, consider whether the following behaviors, attitudes, and patterns of thinking are coming into play. If they are, you may want to consider using a screening tool, which we’ll discuss in a moment.
❚ Clues to cluster A disorders
- The patient has no peer relationships outside immediate family.
- The patient almost always chooses solitary activities for work and personal enjoyment.
❚ Cluster B clues
- Hypersensitivity to treatment disagreements or cancelled appointments are common (and likely experienced as rejection).
- Mood changes occur very quickly, even during a single visit.
- There is a history of many failed relationships with providers and others.
- The patient will describe an individual as both “wonderful” and “terrible” (ie, splitting) and may do so during the course of one visit.
- The patient may also split groups (eg, medical staff) by affective extremes (eg, adoration and hatred).
- The patient may hint at suicide or acts of self-harm.7
❚ Cluster C clues
- There is an excessive dependency on family, friends, or providers.
- Significant anxiety is experienced when the patient has to make an independent decision.
- There is a fear of relationship loss and resultant vulnerability to exploitation or abuse.
- Pervasive perfectionism makes treatment planning or course changes difficult.
- Anxiety and fear are unrelieved despite support and ample information.
Consider these screening tools
Several screening tools for personality disorders can be used to follow up on your initial clinical impressions. We also highly recommend you consider concurrent screening for substance abuse, as addiction is a common comorbidity with personality disorders.
❚
❚ A sampling of screening tools. The Standardised Assessment of Personality Abbreviated Scale (SAPAS)9 is an 8-item measure that correlates well with disorders in clusters A and C.
BPD (cluster B) has many brief scale options, including the McLean Screening Instrument for Borderline Personality Disorder (MSI-BPD).10 This 10-item questionnaire demonstrates sensitivity and specificity for BPD.
The International Personality Disorder Examination (IPDE) includes a 15-minute screening tool to help identify patients who may have any personality disorder, regardless of cluster.11
Improve patient encounters with these Tx pearls
In the family medicine clinic, a collaborative primary care and behavioral health team can be extremely helpful in the diagnosis and management of patients with personality disorders.12 First-line treatment of these disorders is psychotherapy, whereas medications are mainly used for symptom management. See Black and colleagues’ work for a thorough discussion on psychopharmacology considerations with personality disorders. 13
The following tips can help you to improve your interactions with patients who have personality disorders.
❚ Cluster A approaches
- Recommend treatment that respects the patient’s need for relative isolation.14
- Don’t be personally offended by your patient’s flat or disinterested affect or concrete thinking; don’t let it diminish the emotional support you provide.6
- Consult with a health psychologist (who has expertise in physical health conditions, brief treatments, and the medical system) to connect the patient with a long-term therapist. It is better to focus on fundamental changes, rather than employing brief behavioral techniques, for symptom relief. Patients with personality disorders tend to have better outcomes with long-term psychological care.15
❚ Cluster B approaches
- Set boundaries—eg, specific time limits for visits—and keep them.8
- Schedule brief, more frequent, appointments to reduce perceived feelings of abandonment.
- Coordinate plans with the entire clinic team to avoid splitting and blaming.16
- Avoid providing patients with personal information, as it may provide fodder for splitting behavior. 8
- Do not take things personally. Let patients “own” their own distress. These patients often take an emotional toll on the provider.16
- Engage the help of a health psychologist to reduce burnout and for more long-term continuity of care. A health psychologist who specializes in dialectical behavioral therapy to work on emotion regulation, distress tolerance, and interpersonal effectiveness would be ideal.17
Continue to: Cluster C approaches...
❚
❚ Cluster C approaches
- Engage the help of family and other trusted individuals in supporting treatment plans.18,19
- Try to provide just 2 treatment choices to the patient and reinforce his or her responsibility to help make the decision collaboratively. This step is important since it is difficult to enhance autonomy in these patients.20
- Engage the help of a cognitive behavioral therapist who can work on assertiveness and problem-solving skills.19
- Be empathetic with the patient and patiently build a trusting relationship, rather than “arguing” with the patient about each specific worry.20
- Make only one change at a time. Give small assignments to the patient, such as monitoring symptoms or reading up on their condition. These can help the patient feel more in control.21
- Present information in brief, clear terms. Avoid “grey areas” to reduce anxiety.21
- Engage a behavioral health provider to reduce rigid expectations and ideally increase feelings of self-esteem; this has been shown to predict better treatment outcomes.22
CASES
Mr. S displays cluster-A characteristics of schizoid personality disorder in addition to the depression he is being treated for. His physician was not put off by his flat affect and respected his limitations with social activities. Use of a stationary bike was recommended for exercise rather than walks outdoors. He also preferred phone calls to in-person encounters, so his follow-up visits were conducted by phone.
Ms. L exhibits cluster-B characteristics of BPD. You begin the tricky dance of setting limits, keeping communication clear, and not blaming yourself or others on your team for Ms. L’s feelings. You schedule regular visits with explicit time limits and discuss with your entire team how to avoid splitting. You involve a psychologist, familiar with treating BPD, who helps the patient learn positive interpersonal coping skills.
Ms. B displays cluster-C characteristics of dependent and obsessive-compulsive personality disorders. At her follow-up visit, you provide a great deal of empathy and try not to argue her out of each worry that she brings up. You make one change at a time and enlist the help of her daughter in giving her pills at home and offering reassurance. You collaborate with a cognitive behavioral therapist who works on exposing her to moderately anxiety-provoking situations/decisions.
THE CASES
Winston S* is a 23-year-old man referred by a psychiatrist colleague for primary care. He works delivering papers in the early morning hours and spends his day alone in his apartment mainly eating frozen pizza. He has worked solitary jobs his entire life and says he prefers it that way. His answers to questions lack emotion. He doesn’t seem to have any friends or regular contact with family. He follows the medical advice he receives but can’t seem to get out of the house to exercise or socialize. His psychiatrist was treating him with a selective serotonin reuptake inhibitor for depression when he was referred.
Denise L* is a 37-year-old woman who transferred to your practice because she says the previous practice’s office manager was disrespectful and the doctor did not listen to her. She has been “very appreciative” of you and your “well-run office.” You have addressed her fibromyalgia and she has shared several personal details about her life. In the following weeks, you receive several phone calls and messages from her. At a follow-up visit, she asks questions about your family and seems agitated when you hesitate to answer. She questions whether you remember details of her history. She pushes, “Did you remember that, doctor?” She also mentions that your front desk staff seems rude to her.
Ruth B* is an 82-year-old woman whose blood pressure measured in your office is 176/94 mm Hg. When you recommend starting a medication and getting blood tests, she responds with a litany of fearful questions. She seems immobilized by worries about treatment and equally so about the risks of nontreatment. You can’t seem to get past the anxiety to decide on a satisfactory plan. She has to write everything down on a notepad and worries if she does not get every detail.
●
* This patient’s name has been changed to protect his identity. The other 2 patients are an amalgam of patients for whom the authors have provided care.
According to a survey of practicing primary care physicians, as many as 15% of patient encounters can be difficult.1 Demanding, intrusive, or angry patients who reject health care interventions are often-cited sources of these difficulties.2,3 While it is true that patient, physician, and environmental factors may contribute to challenging interactions, some patients who are “difficult” may actually have a personality disorder that requires a distinctive approach to care. Recognizing these patients can help empower physicians to provide compassionate and effective care, reduce team angst, and minimize burnout.
❚ What qualifies as a personality disorder? A personality disorder is an enduring pattern of inner experience and behavior that deviates markedly from the expectations of the individual’s culture, is pervasive and inflexible, has an onset in adolescence or early adulthood, is unchanging over time, and leads to distress or impairment in social or occupational functioning.4 The prevalence of any personality disorder seems to have increased over the past decade from 9.1%4 to 12.16%.5 The Diagnostic and Statistical Manual of Mental Disorders (DSM-5) classifies personality disorders in 3 clusters—A, B, and C (TABLE4)—with prevalence rates at 7.23%, 5.53%, and 6.7%, respectively.5 The review below will focus on the distinct personality disorders exhibited by the patients described in the opening cases.

Continue to: A closer look at the clusters...
A closer look at the clusters
Cluster A disorders
Paranoid, schizoid, and schizotypal disorders are part of this cluster. These patients exhibit odd or eccentric thinking and behavior. Individuals with schizoid personality disorder, for instance, usually lack relationships and lack the desire to acquire and maintain relationships.4 They often organize their lives to remain isolated and will choose occupations that require little social interaction. They sometimes view themselves as observers rather than participants in their own lives.6
Cluster B disorders
Dramatic, overly emotional, or unpredictable thinking and behavior are characteristic of individuals who have antisocial, borderline, histrionic, or narcissistic disorders. Patients with borderline personality disorder (BPD), for example, demonstrate a longstanding pattern of instability in affect, self-image, and relationships.4 Patients with BPD often display extreme interpersonal hypersensitivity and make frantic efforts to avoid real or imagined abandonment. Identity disturbance, feelings of emptiness, and efforts to avoid abandonment have all been associated with increased suicide risk.7
In a primary care setting, such a patient may display extremely strong reactions to minor disappointments. When the physician is unavailable for a last-minute appointment or to authorize an unscheduled medication refill or to receive an after-hours phone call, the patient may become irate. The physician, who previously was idealized by the patient as “the only person who understands me,” is now devalued as “the worst doctor I’ve ever had.”8
Cluster C disorders
With these individuals, anxious or fearful thinking and behavior predominate. Avoidant, dependent, and obsessive-compulsive disorders are included in this cluster.
Dependent personality disorder (DPD) is characterized by a pervasive and extreme need to be taken care of. Submissive and clingy behavior and fear of separation are excessive. This patient may have difficulty making everyday decisions, being assertive, or expressing disagreement with others.4
Obsessive-compulsive personality disorder falls in this cluster and is typified by a pervasive preoccupation with orderliness, perfectionism, and control, at the price of flexibility and efficiency. This individual may be reluctant to get rid of sentimental objects, have rigid moral beliefs, and have significant difficulty working with others who do not follow their rules.4
Continue to: These clues may suggest...
These clues may suggest a personality disorder
If you find that encounters with a particular patient are growing increasingly difficult, consider whether the following behaviors, attitudes, and patterns of thinking are coming into play. If they are, you may want to consider using a screening tool, which we’ll discuss in a moment.
❚ Clues to cluster A disorders
- The patient has no peer relationships outside immediate family.
- The patient almost always chooses solitary activities for work and personal enjoyment.
❚ Cluster B clues
- Hypersensitivity to treatment disagreements or cancelled appointments are common (and likely experienced as rejection).
- Mood changes occur very quickly, even during a single visit.
- There is a history of many failed relationships with providers and others.
- The patient will describe an individual as both “wonderful” and “terrible” (ie, splitting) and may do so during the course of one visit.
- The patient may also split groups (eg, medical staff) by affective extremes (eg, adoration and hatred).
- The patient may hint at suicide or acts of self-harm.7
❚ Cluster C clues
- There is an excessive dependency on family, friends, or providers.
- Significant anxiety is experienced when the patient has to make an independent decision.
- There is a fear of relationship loss and resultant vulnerability to exploitation or abuse.
- Pervasive perfectionism makes treatment planning or course changes difficult.
- Anxiety and fear are unrelieved despite support and ample information.
Consider these screening tools
Several screening tools for personality disorders can be used to follow up on your initial clinical impressions. We also highly recommend you consider concurrent screening for substance abuse, as addiction is a common comorbidity with personality disorders.
❚
❚ A sampling of screening tools. The Standardised Assessment of Personality Abbreviated Scale (SAPAS)9 is an 8-item measure that correlates well with disorders in clusters A and C.
BPD (cluster B) has many brief scale options, including the McLean Screening Instrument for Borderline Personality Disorder (MSI-BPD).10 This 10-item questionnaire demonstrates sensitivity and specificity for BPD.
The International Personality Disorder Examination (IPDE) includes a 15-minute screening tool to help identify patients who may have any personality disorder, regardless of cluster.11
Improve patient encounters with these Tx pearls
In the family medicine clinic, a collaborative primary care and behavioral health team can be extremely helpful in the diagnosis and management of patients with personality disorders.12 First-line treatment of these disorders is psychotherapy, whereas medications are mainly used for symptom management. See Black and colleagues’ work for a thorough discussion on psychopharmacology considerations with personality disorders. 13
The following tips can help you to improve your interactions with patients who have personality disorders.
❚ Cluster A approaches
- Recommend treatment that respects the patient’s need for relative isolation.14
- Don’t be personally offended by your patient’s flat or disinterested affect or concrete thinking; don’t let it diminish the emotional support you provide.6
- Consult with a health psychologist (who has expertise in physical health conditions, brief treatments, and the medical system) to connect the patient with a long-term therapist. It is better to focus on fundamental changes, rather than employing brief behavioral techniques, for symptom relief. Patients with personality disorders tend to have better outcomes with long-term psychological care.15
❚ Cluster B approaches
- Set boundaries—eg, specific time limits for visits—and keep them.8
- Schedule brief, more frequent, appointments to reduce perceived feelings of abandonment.
- Coordinate plans with the entire clinic team to avoid splitting and blaming.16
- Avoid providing patients with personal information, as it may provide fodder for splitting behavior. 8
- Do not take things personally. Let patients “own” their own distress. These patients often take an emotional toll on the provider.16
- Engage the help of a health psychologist to reduce burnout and for more long-term continuity of care. A health psychologist who specializes in dialectical behavioral therapy to work on emotion regulation, distress tolerance, and interpersonal effectiveness would be ideal.17
Continue to: Cluster C approaches...
❚
❚ Cluster C approaches
- Engage the help of family and other trusted individuals in supporting treatment plans.18,19
- Try to provide just 2 treatment choices to the patient and reinforce his or her responsibility to help make the decision collaboratively. This step is important since it is difficult to enhance autonomy in these patients.20
- Engage the help of a cognitive behavioral therapist who can work on assertiveness and problem-solving skills.19
- Be empathetic with the patient and patiently build a trusting relationship, rather than “arguing” with the patient about each specific worry.20
- Make only one change at a time. Give small assignments to the patient, such as monitoring symptoms or reading up on their condition. These can help the patient feel more in control.21
- Present information in brief, clear terms. Avoid “grey areas” to reduce anxiety.21
- Engage a behavioral health provider to reduce rigid expectations and ideally increase feelings of self-esteem; this has been shown to predict better treatment outcomes.22
CASES
Mr. S displays cluster-A characteristics of schizoid personality disorder in addition to the depression he is being treated for. His physician was not put off by his flat affect and respected his limitations with social activities. Use of a stationary bike was recommended for exercise rather than walks outdoors. He also preferred phone calls to in-person encounters, so his follow-up visits were conducted by phone.
Ms. L exhibits cluster-B characteristics of BPD. You begin the tricky dance of setting limits, keeping communication clear, and not blaming yourself or others on your team for Ms. L’s feelings. You schedule regular visits with explicit time limits and discuss with your entire team how to avoid splitting. You involve a psychologist, familiar with treating BPD, who helps the patient learn positive interpersonal coping skills.
Ms. B displays cluster-C characteristics of dependent and obsessive-compulsive personality disorders. At her follow-up visit, you provide a great deal of empathy and try not to argue her out of each worry that she brings up. You make one change at a time and enlist the help of her daughter in giving her pills at home and offering reassurance. You collaborate with a cognitive behavioral therapist who works on exposing her to moderately anxiety-provoking situations/decisions.
1. Hull SK, Broquet K. How to manage difficult patient encounters. Fam Pract Manag. 2007;14:30-34.
2. Groves JE. Taking care of the hateful patient. N Engl J Med.1978;298: 883-887.
3. O’Dowd TC. Five years of heartsink patients in primary care. BMJ. 1988;297:528-530.
4. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 5th edition (DSM-5). American Psychiatric Publishing; 2013.
5. Volkert J, Gablonski TC, Rabung S. Prevalence of personality disorders in the general adult population in Western countries: systematic review and meta-analysis. Br J Psychiatry. 2018;213:709-715.
6. Esterberg ML, Goulding SM, Walker EF. Cluster A personality disorders: schizotypal, schizoid and paranoid personality disorders in childhood and adolescence. J Psychopathol Behav Assess. 2010;32:515-528.
7. Yen S, Peters JR, Nishar S, et al. Association of borderline personality disorder criteria with suicide attempts: findings from the collaborative longitudinal study of personality disorders over 10 years of follow-up. JAMA Psychiatry. 2021;78:187-194.
8. Dubovsky AN, Kiefer MM. Borderline personality disorder in the primary care setting. Med Clin North Am. 2014;98:1049-1064.
9. Hesse M, Moran P. (2010). Screening for personality disorder with the Standardised Assessment of Personality: Abbreviated Scale (SAPAS): further evidence of concurrent validity. BMC Psychiatry. 2010;10:10.
10. Zanarini MC, Vujanovic AA, Parachini EA, et al. A screening measure for BPD: the McLean screening instrument for borderline personality disorder (MSI-BPD). J Pers Disord. 2003;17:568-573.
11. Loranger AW, Sartorius N, Andreoli A, et al. The International Personality Disorder Examination. The World Health Organization/Alcohol, Drug Abuse, and Mental Health Administration international pilot study of personality disorders. Arch Gen Psychiatry. 1994;51:215-224.12. Nelson KJ, Skodol A, Friedman M. Pharmacotherapy for personality disorders. UpToDate. Accessed April 22, 2021. www.uptodate.com/contents/pharmacotherapy-for-personality-disorders
13. Black D, Paris J, Schulz C. Evidence-based integrated biopsychosocial treatment of borderline personality disorder. In: Muse M (ed). Cognitive Behavioral Psychopharmacology: the Clinical Practice of Evidence-Based Biopsychosocial Integration. Wiley; 2017:137-166.
14. Beck AT, Davis DD, Freeman A. Cognitive Therapy of Personality Disorders. 3rd ed. The Guilford Press; 2015.
15. Thylstrup B, Hesse M. “I am not complaining”–ambivalence construct in schizoid personality disorder. Am J Psychother. 2009;63:147-167.
16. Ricke AK, Lee MJ, Chambers JE. The difficult patient: borderline personality disorder in the obstetrical and gynecological patient. Obstet Gynecol Surv. 2012;67:495-502.
17. Seow LLY, Page AC, Hooke GR. Severity of borderline personality disorder symptoms as a moderator of the association between the use of dialectical behaviour therapy skills and treatment outcomes. Psychother Res. 2020;30:920-933.
18. Nichols WC. Integrative marital and family treatment of dependent personality disorders. In: MacFarlane MM (Ed.) Family Treatment of Personality Disorders: Advances in Clinical Practice. Haworth Clinical Practice Press; 2004:173-204.
19. Disney KL. Dependent personality disorder: a critical review. Clin Psychol Rev. 2013;33:1184-1196.
20. Bender DS. The therapeutic alliance in the treatment of personality disorders. J Psychiatr Pract. 2005;11:73-87.
21. Ward RK. Assessment and management of personality disorders. Am Fam Physician. 2004;70:1505-1512.
22. Cummings JA, Hayes AM, Cardaciotto L, et al. The dynamics of self-esteem in cognitive therapy for avoidant and obsessive-compulsive personality disorders: an adaptive role of self-esteem variability? Cognit Ther Res. 2012;36:272-281.
1. Hull SK, Broquet K. How to manage difficult patient encounters. Fam Pract Manag. 2007;14:30-34.
2. Groves JE. Taking care of the hateful patient. N Engl J Med.1978;298: 883-887.
3. O’Dowd TC. Five years of heartsink patients in primary care. BMJ. 1988;297:528-530.
4. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 5th edition (DSM-5). American Psychiatric Publishing; 2013.
5. Volkert J, Gablonski TC, Rabung S. Prevalence of personality disorders in the general adult population in Western countries: systematic review and meta-analysis. Br J Psychiatry. 2018;213:709-715.
6. Esterberg ML, Goulding SM, Walker EF. Cluster A personality disorders: schizotypal, schizoid and paranoid personality disorders in childhood and adolescence. J Psychopathol Behav Assess. 2010;32:515-528.
7. Yen S, Peters JR, Nishar S, et al. Association of borderline personality disorder criteria with suicide attempts: findings from the collaborative longitudinal study of personality disorders over 10 years of follow-up. JAMA Psychiatry. 2021;78:187-194.
8. Dubovsky AN, Kiefer MM. Borderline personality disorder in the primary care setting. Med Clin North Am. 2014;98:1049-1064.
9. Hesse M, Moran P. (2010). Screening for personality disorder with the Standardised Assessment of Personality: Abbreviated Scale (SAPAS): further evidence of concurrent validity. BMC Psychiatry. 2010;10:10.
10. Zanarini MC, Vujanovic AA, Parachini EA, et al. A screening measure for BPD: the McLean screening instrument for borderline personality disorder (MSI-BPD). J Pers Disord. 2003;17:568-573.
11. Loranger AW, Sartorius N, Andreoli A, et al. The International Personality Disorder Examination. The World Health Organization/Alcohol, Drug Abuse, and Mental Health Administration international pilot study of personality disorders. Arch Gen Psychiatry. 1994;51:215-224.12. Nelson KJ, Skodol A, Friedman M. Pharmacotherapy for personality disorders. UpToDate. Accessed April 22, 2021. www.uptodate.com/contents/pharmacotherapy-for-personality-disorders
13. Black D, Paris J, Schulz C. Evidence-based integrated biopsychosocial treatment of borderline personality disorder. In: Muse M (ed). Cognitive Behavioral Psychopharmacology: the Clinical Practice of Evidence-Based Biopsychosocial Integration. Wiley; 2017:137-166.
14. Beck AT, Davis DD, Freeman A. Cognitive Therapy of Personality Disorders. 3rd ed. The Guilford Press; 2015.
15. Thylstrup B, Hesse M. “I am not complaining”–ambivalence construct in schizoid personality disorder. Am J Psychother. 2009;63:147-167.
16. Ricke AK, Lee MJ, Chambers JE. The difficult patient: borderline personality disorder in the obstetrical and gynecological patient. Obstet Gynecol Surv. 2012;67:495-502.
17. Seow LLY, Page AC, Hooke GR. Severity of borderline personality disorder symptoms as a moderator of the association between the use of dialectical behaviour therapy skills and treatment outcomes. Psychother Res. 2020;30:920-933.
18. Nichols WC. Integrative marital and family treatment of dependent personality disorders. In: MacFarlane MM (Ed.) Family Treatment of Personality Disorders: Advances in Clinical Practice. Haworth Clinical Practice Press; 2004:173-204.
19. Disney KL. Dependent personality disorder: a critical review. Clin Psychol Rev. 2013;33:1184-1196.
20. Bender DS. The therapeutic alliance in the treatment of personality disorders. J Psychiatr Pract. 2005;11:73-87.
21. Ward RK. Assessment and management of personality disorders. Am Fam Physician. 2004;70:1505-1512.
22. Cummings JA, Hayes AM, Cardaciotto L, et al. The dynamics of self-esteem in cognitive therapy for avoidant and obsessive-compulsive personality disorders: an adaptive role of self-esteem variability? Cognit Ther Res. 2012;36:272-281.
Treating sleep apnea lowers MI and stroke risk
particularly for patients with moderate to severe OSA and those who are more adherent to CPAP therapy, a new study suggests.
“Most clinical trials on the effect of CPAP on CV diseases to date have focused on secondary CV prevention. This study contributes another piece of evidence about the role of CPAP therapy to prevent CV diseases,” said Diego R. Mazzotti, PhD, an assistant professor at the University of Kansas Medical Center, Kansas City.
“Our study, while observational, suggests that clinical trials focused on understanding how to sustain long-term CPAP adherence in obstructive sleep apnea patients are necessary and could be critical for optimizing comorbidity risk reduction,” Dr. Mazzotti said.
The study was presented at the virtual annual meeting of the Associated Professional Sleep Societies.
Good adherence important
The researchers analyzed the electronic health records of adults referred for a sleep study through the Kaiser Permanente Southern California health system. The sample included 11,145 adults without OSA, 13,898 with OSA who used CPAP, and 20,884 adults with OSA who did not use CPAP. None of them had CV disease at baseline. Median follow-up was 262 days.
The primary outcome was first occurrence of myocardial infarction, stroke, unstable angina, heart failure, or death caused by CV disease.
In adjusted models, adults with moderate to severe OSA (apnea-hypopnea index ≥15) who did not use CPAP were 71% more likely than those without OSA to have a first CV event (hazard ratio, 1.71; 95% CI, 1.11-2.64). However, the risk for a CV event during follow-up was 32% lower among OSA patients with any CPAP use (HR, 0.68; 95% CI, 0.50-0.93; P = .016).
The effect was mostly driven by those who used CPAP for at least 4 hours per night (HR, 0.60; 95% CI, 0.39-0.95). This association was stronger for those with moderate to severe OSA (HR, 0.56; 95% CI, 0.39-0.81).
“This study highlights the importance of long-term management of CPAP therapy in patients with moderate-severe OSA,” Dr. Mazzotti said in an interview.
“It suggests that maintaining good CPAP adherence might be beneficial for cardiovascular health, besides the already established benefits on quality of life, sleepiness, and other cardiometabolic functions,” he said.
Dr. Mazzotti said several mechanisms might explain the association between CPAP use and lower risk for CV events. “CPAP treats OSA by preventing respiratory pauses that occur during sleep, therefore preventing arousals, sleep fragmentation, and decreases in blood oxygen. These improved cardiorespiratory functions can be beneficial to avoid certain molecular changes that are known to contribute to cardiovascular risk, such as oxidative stress and inflammation,” he explained.
“However, specific studies fully understanding these mechanisms are necessary,” Dr. Mazzotti added.
In a comment, Nitun Verma, MD, a spokesperson for the American Academy of Sleep Medicine, said that “the frequent decreases in oxygen levels and fragmented sleep from apnea are associated with cardiovascular disorders. We know this from multiple studies. This, however, was a large study and strengthens the association between improving apnea and reduced serious cardiovascular events.”
Funding for the study was provided by the American Academy of Sleep Medicine Foundation and the American Heart Association. Dr. Mazzotti and Dr. Verma disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
particularly for patients with moderate to severe OSA and those who are more adherent to CPAP therapy, a new study suggests.
“Most clinical trials on the effect of CPAP on CV diseases to date have focused on secondary CV prevention. This study contributes another piece of evidence about the role of CPAP therapy to prevent CV diseases,” said Diego R. Mazzotti, PhD, an assistant professor at the University of Kansas Medical Center, Kansas City.
“Our study, while observational, suggests that clinical trials focused on understanding how to sustain long-term CPAP adherence in obstructive sleep apnea patients are necessary and could be critical for optimizing comorbidity risk reduction,” Dr. Mazzotti said.
The study was presented at the virtual annual meeting of the Associated Professional Sleep Societies.
Good adherence important
The researchers analyzed the electronic health records of adults referred for a sleep study through the Kaiser Permanente Southern California health system. The sample included 11,145 adults without OSA, 13,898 with OSA who used CPAP, and 20,884 adults with OSA who did not use CPAP. None of them had CV disease at baseline. Median follow-up was 262 days.
The primary outcome was first occurrence of myocardial infarction, stroke, unstable angina, heart failure, or death caused by CV disease.
In adjusted models, adults with moderate to severe OSA (apnea-hypopnea index ≥15) who did not use CPAP were 71% more likely than those without OSA to have a first CV event (hazard ratio, 1.71; 95% CI, 1.11-2.64). However, the risk for a CV event during follow-up was 32% lower among OSA patients with any CPAP use (HR, 0.68; 95% CI, 0.50-0.93; P = .016).
The effect was mostly driven by those who used CPAP for at least 4 hours per night (HR, 0.60; 95% CI, 0.39-0.95). This association was stronger for those with moderate to severe OSA (HR, 0.56; 95% CI, 0.39-0.81).
“This study highlights the importance of long-term management of CPAP therapy in patients with moderate-severe OSA,” Dr. Mazzotti said in an interview.
“It suggests that maintaining good CPAP adherence might be beneficial for cardiovascular health, besides the already established benefits on quality of life, sleepiness, and other cardiometabolic functions,” he said.
Dr. Mazzotti said several mechanisms might explain the association between CPAP use and lower risk for CV events. “CPAP treats OSA by preventing respiratory pauses that occur during sleep, therefore preventing arousals, sleep fragmentation, and decreases in blood oxygen. These improved cardiorespiratory functions can be beneficial to avoid certain molecular changes that are known to contribute to cardiovascular risk, such as oxidative stress and inflammation,” he explained.
“However, specific studies fully understanding these mechanisms are necessary,” Dr. Mazzotti added.
In a comment, Nitun Verma, MD, a spokesperson for the American Academy of Sleep Medicine, said that “the frequent decreases in oxygen levels and fragmented sleep from apnea are associated with cardiovascular disorders. We know this from multiple studies. This, however, was a large study and strengthens the association between improving apnea and reduced serious cardiovascular events.”
Funding for the study was provided by the American Academy of Sleep Medicine Foundation and the American Heart Association. Dr. Mazzotti and Dr. Verma disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
particularly for patients with moderate to severe OSA and those who are more adherent to CPAP therapy, a new study suggests.
“Most clinical trials on the effect of CPAP on CV diseases to date have focused on secondary CV prevention. This study contributes another piece of evidence about the role of CPAP therapy to prevent CV diseases,” said Diego R. Mazzotti, PhD, an assistant professor at the University of Kansas Medical Center, Kansas City.
“Our study, while observational, suggests that clinical trials focused on understanding how to sustain long-term CPAP adherence in obstructive sleep apnea patients are necessary and could be critical for optimizing comorbidity risk reduction,” Dr. Mazzotti said.
The study was presented at the virtual annual meeting of the Associated Professional Sleep Societies.
Good adherence important
The researchers analyzed the electronic health records of adults referred for a sleep study through the Kaiser Permanente Southern California health system. The sample included 11,145 adults without OSA, 13,898 with OSA who used CPAP, and 20,884 adults with OSA who did not use CPAP. None of them had CV disease at baseline. Median follow-up was 262 days.
The primary outcome was first occurrence of myocardial infarction, stroke, unstable angina, heart failure, or death caused by CV disease.
In adjusted models, adults with moderate to severe OSA (apnea-hypopnea index ≥15) who did not use CPAP were 71% more likely than those without OSA to have a first CV event (hazard ratio, 1.71; 95% CI, 1.11-2.64). However, the risk for a CV event during follow-up was 32% lower among OSA patients with any CPAP use (HR, 0.68; 95% CI, 0.50-0.93; P = .016).
The effect was mostly driven by those who used CPAP for at least 4 hours per night (HR, 0.60; 95% CI, 0.39-0.95). This association was stronger for those with moderate to severe OSA (HR, 0.56; 95% CI, 0.39-0.81).
“This study highlights the importance of long-term management of CPAP therapy in patients with moderate-severe OSA,” Dr. Mazzotti said in an interview.
“It suggests that maintaining good CPAP adherence might be beneficial for cardiovascular health, besides the already established benefits on quality of life, sleepiness, and other cardiometabolic functions,” he said.
Dr. Mazzotti said several mechanisms might explain the association between CPAP use and lower risk for CV events. “CPAP treats OSA by preventing respiratory pauses that occur during sleep, therefore preventing arousals, sleep fragmentation, and decreases in blood oxygen. These improved cardiorespiratory functions can be beneficial to avoid certain molecular changes that are known to contribute to cardiovascular risk, such as oxidative stress and inflammation,” he explained.
“However, specific studies fully understanding these mechanisms are necessary,” Dr. Mazzotti added.
In a comment, Nitun Verma, MD, a spokesperson for the American Academy of Sleep Medicine, said that “the frequent decreases in oxygen levels and fragmented sleep from apnea are associated with cardiovascular disorders. We know this from multiple studies. This, however, was a large study and strengthens the association between improving apnea and reduced serious cardiovascular events.”
Funding for the study was provided by the American Academy of Sleep Medicine Foundation and the American Heart Association. Dr. Mazzotti and Dr. Verma disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM SLEEP 2021
Memory benefit seen with antihypertensives crossing blood-brain barrier
Over a 3-year period, cognitively normal older adults taking BBB-crossing antihypertensives demonstrated superior verbal memory, compared with similar individuals receiving non–BBB-crossing antihypertensives, reported lead author Jean K. Ho, PhD, of the Institute for Memory Impairments and Neurological Disorders at the University of California, Irvine, and colleagues.
According to the investigators, the findings add color to a known link between hypertension and neurologic degeneration, and may aid the search for new therapeutic targets.
“Hypertension is a well-established risk factor for cognitive decline and dementia, possibly through its effects on both cerebrovascular disease and Alzheimer’s disease,” Dr. Ho and colleagues wrote in Hypertension. “Studies of antihypertensive treatments have reported possible salutary effects on cognition and cerebrovascular disease, as well as Alzheimer’s disease neuropathology.”
In a previous study, individuals younger than 75 years exposed to antihypertensives had an 8% decreased risk of dementia per year of use, while another trial showed that intensive blood pressure–lowering therapy reduced mild cognitive impairment by 19%.
“Despite these encouraging findings ... larger meta-analytic studies have been hampered by the fact that pharmacokinetic properties are typically not considered in existing studies or routine clinical practice,” wrote Dr. Ho and colleagues. “The present study sought to fill this gap [in that it was] a large and longitudinal meta-analytic study of existing data recoded to assess the effects of BBB-crossing potential in renin-angiotensin system [RAS] treatments among hypertensive adults.”
Methods and results
The meta-analysis included randomized clinical trials, prospective cohort studies, and retrospective observational studies. The researchers assessed data on 12,849 individuals from 14 cohorts that received either BBB-crossing or non–BBB-crossing antihypertensives.
The BBB-crossing properties of RAS treatments were identified by a literature review. Of ACE inhibitors, captopril, fosinopril, lisinopril, perindopril, ramipril, and trandolapril were classified as BBB crossing, and benazepril, enalapril, moexipril, and quinapril were classified as non–BBB-crossing. Of ARBs, telmisartan and candesartan were considered BBB-crossing, and olmesartan, eprosartan, irbesartan, and losartan were tagged as non–BBB-crossing.
Cognition was assessed via the following seven domains: executive function, attention, verbal memory learning, language, mental status, recall, and processing speed.
Compared with individuals taking non–BBB-crossing antihypertensives, those taking BBB-crossing agents had significantly superior verbal memory (recall), with a maximum effect size of 0.07 (P = .03).
According to the investigators, this finding was particularly noteworthy, as the BBB-crossing group had relatively higher vascular risk burden and lower mean education level.
“These differences make it all the more remarkable that the BBB-crossing group displayed better memory ability over time despite these cognitive disadvantages,” the investigators wrote.
Still, not all the findings favored BBB-crossing agents. Individuals in the BBB-crossing group had relatively inferior attention ability, with a minimum effect size of –0.17 (P = .02).
The other cognitive measures were not significantly different between groups.
Clinicians may consider findings after accounting for other factors
Principal investigator Daniel A. Nation, PhD, associate professor of psychological science and a faculty member of the Institute for Memory Impairments and Neurological Disorders at the University of California, Irvine, suggested that the small difference in verbal memory between groups could be clinically significant over a longer period of time.
“Although the overall effect size was pretty small, if you look at how long it would take for someone [with dementia] to progress over many years of decline, it would actually end up being a pretty big effect,” Dr. Nation said in an interview. “Small effect sizes could actually end up preventing a lot of cases of dementia,” he added.
The conflicting results in the BBB-crossing group – better verbal memory but worse attention ability – were “surprising,” he noted.
“I sort of didn’t believe it at first,” Dr. Nation said, “because the memory finding is sort of replication – we’d observed the same exact effect on memory in a smaller sample in another study. ... The attention [finding], going another way, was a new thing.”
Dr. Nation suggested that the intergroup differences in attention ability may stem from idiosyncrasies of the tests used to measure that domain, which can be impacted by cardiovascular or brain vascular disease. Or it could be caused by something else entirely, he said, noting that further investigation is needed.
He added that the improvements in verbal memory within the BBB-crossing group could be caused by direct effects on the brain. He pointed out that certain ACE polymorphisms have been linked with Alzheimer’s disease risk, and those same polymorphisms, in animal models, lead to neurodegeneration, with reversal possible through administration of ACE inhibitors.
“It could be that what we’re observing has nothing really to do with blood pressure,” Dr. Nation explained. “This could be a neuronal effect on learning memory systems.”
He went on to suggest that clinicians may consider these findings when selecting antihypertensive agents for their patients, with the caveat that all other prescribing factors have already been taking to account.
“In the event that you’re going to give an ACE inhibitor or an angiotensin receptor blocker anyway, and it ends up being a somewhat arbitrary decision in terms of which specific drug you’re going to give, then perhaps this is a piece of information you would take into account – that one gets in the brain and one doesn’t – in somebody at risk for cognitive decline,” Dr. Nation said.
Exact mechanisms of action unknown
Hélène Girouard, PhD, assistant professor of pharmacology and physiology at the University of Montreal, said in an interview that the findings are “of considerable importance, knowing that brain alterations could begin as much as 30 years before manifestation of dementia.”
Since 2003, Dr. Girouard has been studying the cognitive effects of antihypertensive medications. She noted that previous studies involving rodents “have shown beneficial effects [of BBB-crossing antihypertensive drugs] on cognition independent of their effects on blood pressure.”
The drugs’ exact mechanisms of action, however, remain elusive, according to Dr. Girouard, who offered several possible explanations, including amelioration of BBB disruption, brain inflammation, cerebral blood flow dysregulation, cholinergic dysfunction, and neurologic deficits. “Whether these mechanisms may explain Ho and colleagues’ observations remains to be established,” she added.
Andrea L. Schneider, MD, PhD, assistant professor of neurology at the University of Pennsylvania, Philadelphia, applauded the study, but ultimately suggested that more research is needed to impact clinical decision-making.
“The results of this important and well-done study suggest that further investigation into targeted mechanism-based approaches to selecting hypertension treatment agents, with a specific focus on cognitive outcomes, is warranted,” Dr. Schneider said in an interview. “Before changing clinical practice, further work is necessary to disentangle contributions of medication mechanism, comorbid vascular risk factors, and achieved blood pressure reduction, among others.”
The investigators disclosed support from the National Institutes of Health, the Alzheimer’s Association, the Waksman Foundation of Japan, and others. The interviewees reported no relevant conflicts of interest.
Over a 3-year period, cognitively normal older adults taking BBB-crossing antihypertensives demonstrated superior verbal memory, compared with similar individuals receiving non–BBB-crossing antihypertensives, reported lead author Jean K. Ho, PhD, of the Institute for Memory Impairments and Neurological Disorders at the University of California, Irvine, and colleagues.
According to the investigators, the findings add color to a known link between hypertension and neurologic degeneration, and may aid the search for new therapeutic targets.
“Hypertension is a well-established risk factor for cognitive decline and dementia, possibly through its effects on both cerebrovascular disease and Alzheimer’s disease,” Dr. Ho and colleagues wrote in Hypertension. “Studies of antihypertensive treatments have reported possible salutary effects on cognition and cerebrovascular disease, as well as Alzheimer’s disease neuropathology.”
In a previous study, individuals younger than 75 years exposed to antihypertensives had an 8% decreased risk of dementia per year of use, while another trial showed that intensive blood pressure–lowering therapy reduced mild cognitive impairment by 19%.
“Despite these encouraging findings ... larger meta-analytic studies have been hampered by the fact that pharmacokinetic properties are typically not considered in existing studies or routine clinical practice,” wrote Dr. Ho and colleagues. “The present study sought to fill this gap [in that it was] a large and longitudinal meta-analytic study of existing data recoded to assess the effects of BBB-crossing potential in renin-angiotensin system [RAS] treatments among hypertensive adults.”
Methods and results
The meta-analysis included randomized clinical trials, prospective cohort studies, and retrospective observational studies. The researchers assessed data on 12,849 individuals from 14 cohorts that received either BBB-crossing or non–BBB-crossing antihypertensives.
The BBB-crossing properties of RAS treatments were identified by a literature review. Of ACE inhibitors, captopril, fosinopril, lisinopril, perindopril, ramipril, and trandolapril were classified as BBB crossing, and benazepril, enalapril, moexipril, and quinapril were classified as non–BBB-crossing. Of ARBs, telmisartan and candesartan were considered BBB-crossing, and olmesartan, eprosartan, irbesartan, and losartan were tagged as non–BBB-crossing.
Cognition was assessed via the following seven domains: executive function, attention, verbal memory learning, language, mental status, recall, and processing speed.
Compared with individuals taking non–BBB-crossing antihypertensives, those taking BBB-crossing agents had significantly superior verbal memory (recall), with a maximum effect size of 0.07 (P = .03).
According to the investigators, this finding was particularly noteworthy, as the BBB-crossing group had relatively higher vascular risk burden and lower mean education level.
“These differences make it all the more remarkable that the BBB-crossing group displayed better memory ability over time despite these cognitive disadvantages,” the investigators wrote.
Still, not all the findings favored BBB-crossing agents. Individuals in the BBB-crossing group had relatively inferior attention ability, with a minimum effect size of –0.17 (P = .02).
The other cognitive measures were not significantly different between groups.
Clinicians may consider findings after accounting for other factors
Principal investigator Daniel A. Nation, PhD, associate professor of psychological science and a faculty member of the Institute for Memory Impairments and Neurological Disorders at the University of California, Irvine, suggested that the small difference in verbal memory between groups could be clinically significant over a longer period of time.
“Although the overall effect size was pretty small, if you look at how long it would take for someone [with dementia] to progress over many years of decline, it would actually end up being a pretty big effect,” Dr. Nation said in an interview. “Small effect sizes could actually end up preventing a lot of cases of dementia,” he added.
The conflicting results in the BBB-crossing group – better verbal memory but worse attention ability – were “surprising,” he noted.
“I sort of didn’t believe it at first,” Dr. Nation said, “because the memory finding is sort of replication – we’d observed the same exact effect on memory in a smaller sample in another study. ... The attention [finding], going another way, was a new thing.”
Dr. Nation suggested that the intergroup differences in attention ability may stem from idiosyncrasies of the tests used to measure that domain, which can be impacted by cardiovascular or brain vascular disease. Or it could be caused by something else entirely, he said, noting that further investigation is needed.
He added that the improvements in verbal memory within the BBB-crossing group could be caused by direct effects on the brain. He pointed out that certain ACE polymorphisms have been linked with Alzheimer’s disease risk, and those same polymorphisms, in animal models, lead to neurodegeneration, with reversal possible through administration of ACE inhibitors.
“It could be that what we’re observing has nothing really to do with blood pressure,” Dr. Nation explained. “This could be a neuronal effect on learning memory systems.”
He went on to suggest that clinicians may consider these findings when selecting antihypertensive agents for their patients, with the caveat that all other prescribing factors have already been taking to account.
“In the event that you’re going to give an ACE inhibitor or an angiotensin receptor blocker anyway, and it ends up being a somewhat arbitrary decision in terms of which specific drug you’re going to give, then perhaps this is a piece of information you would take into account – that one gets in the brain and one doesn’t – in somebody at risk for cognitive decline,” Dr. Nation said.
Exact mechanisms of action unknown
Hélène Girouard, PhD, assistant professor of pharmacology and physiology at the University of Montreal, said in an interview that the findings are “of considerable importance, knowing that brain alterations could begin as much as 30 years before manifestation of dementia.”
Since 2003, Dr. Girouard has been studying the cognitive effects of antihypertensive medications. She noted that previous studies involving rodents “have shown beneficial effects [of BBB-crossing antihypertensive drugs] on cognition independent of their effects on blood pressure.”
The drugs’ exact mechanisms of action, however, remain elusive, according to Dr. Girouard, who offered several possible explanations, including amelioration of BBB disruption, brain inflammation, cerebral blood flow dysregulation, cholinergic dysfunction, and neurologic deficits. “Whether these mechanisms may explain Ho and colleagues’ observations remains to be established,” she added.
Andrea L. Schneider, MD, PhD, assistant professor of neurology at the University of Pennsylvania, Philadelphia, applauded the study, but ultimately suggested that more research is needed to impact clinical decision-making.
“The results of this important and well-done study suggest that further investigation into targeted mechanism-based approaches to selecting hypertension treatment agents, with a specific focus on cognitive outcomes, is warranted,” Dr. Schneider said in an interview. “Before changing clinical practice, further work is necessary to disentangle contributions of medication mechanism, comorbid vascular risk factors, and achieved blood pressure reduction, among others.”
The investigators disclosed support from the National Institutes of Health, the Alzheimer’s Association, the Waksman Foundation of Japan, and others. The interviewees reported no relevant conflicts of interest.
Over a 3-year period, cognitively normal older adults taking BBB-crossing antihypertensives demonstrated superior verbal memory, compared with similar individuals receiving non–BBB-crossing antihypertensives, reported lead author Jean K. Ho, PhD, of the Institute for Memory Impairments and Neurological Disorders at the University of California, Irvine, and colleagues.
According to the investigators, the findings add color to a known link between hypertension and neurologic degeneration, and may aid the search for new therapeutic targets.
“Hypertension is a well-established risk factor for cognitive decline and dementia, possibly through its effects on both cerebrovascular disease and Alzheimer’s disease,” Dr. Ho and colleagues wrote in Hypertension. “Studies of antihypertensive treatments have reported possible salutary effects on cognition and cerebrovascular disease, as well as Alzheimer’s disease neuropathology.”
In a previous study, individuals younger than 75 years exposed to antihypertensives had an 8% decreased risk of dementia per year of use, while another trial showed that intensive blood pressure–lowering therapy reduced mild cognitive impairment by 19%.
“Despite these encouraging findings ... larger meta-analytic studies have been hampered by the fact that pharmacokinetic properties are typically not considered in existing studies or routine clinical practice,” wrote Dr. Ho and colleagues. “The present study sought to fill this gap [in that it was] a large and longitudinal meta-analytic study of existing data recoded to assess the effects of BBB-crossing potential in renin-angiotensin system [RAS] treatments among hypertensive adults.”
Methods and results
The meta-analysis included randomized clinical trials, prospective cohort studies, and retrospective observational studies. The researchers assessed data on 12,849 individuals from 14 cohorts that received either BBB-crossing or non–BBB-crossing antihypertensives.
The BBB-crossing properties of RAS treatments were identified by a literature review. Of ACE inhibitors, captopril, fosinopril, lisinopril, perindopril, ramipril, and trandolapril were classified as BBB crossing, and benazepril, enalapril, moexipril, and quinapril were classified as non–BBB-crossing. Of ARBs, telmisartan and candesartan were considered BBB-crossing, and olmesartan, eprosartan, irbesartan, and losartan were tagged as non–BBB-crossing.
Cognition was assessed via the following seven domains: executive function, attention, verbal memory learning, language, mental status, recall, and processing speed.
Compared with individuals taking non–BBB-crossing antihypertensives, those taking BBB-crossing agents had significantly superior verbal memory (recall), with a maximum effect size of 0.07 (P = .03).
According to the investigators, this finding was particularly noteworthy, as the BBB-crossing group had relatively higher vascular risk burden and lower mean education level.
“These differences make it all the more remarkable that the BBB-crossing group displayed better memory ability over time despite these cognitive disadvantages,” the investigators wrote.
Still, not all the findings favored BBB-crossing agents. Individuals in the BBB-crossing group had relatively inferior attention ability, with a minimum effect size of –0.17 (P = .02).
The other cognitive measures were not significantly different between groups.
Clinicians may consider findings after accounting for other factors
Principal investigator Daniel A. Nation, PhD, associate professor of psychological science and a faculty member of the Institute for Memory Impairments and Neurological Disorders at the University of California, Irvine, suggested that the small difference in verbal memory between groups could be clinically significant over a longer period of time.
“Although the overall effect size was pretty small, if you look at how long it would take for someone [with dementia] to progress over many years of decline, it would actually end up being a pretty big effect,” Dr. Nation said in an interview. “Small effect sizes could actually end up preventing a lot of cases of dementia,” he added.
The conflicting results in the BBB-crossing group – better verbal memory but worse attention ability – were “surprising,” he noted.
“I sort of didn’t believe it at first,” Dr. Nation said, “because the memory finding is sort of replication – we’d observed the same exact effect on memory in a smaller sample in another study. ... The attention [finding], going another way, was a new thing.”
Dr. Nation suggested that the intergroup differences in attention ability may stem from idiosyncrasies of the tests used to measure that domain, which can be impacted by cardiovascular or brain vascular disease. Or it could be caused by something else entirely, he said, noting that further investigation is needed.
He added that the improvements in verbal memory within the BBB-crossing group could be caused by direct effects on the brain. He pointed out that certain ACE polymorphisms have been linked with Alzheimer’s disease risk, and those same polymorphisms, in animal models, lead to neurodegeneration, with reversal possible through administration of ACE inhibitors.
“It could be that what we’re observing has nothing really to do with blood pressure,” Dr. Nation explained. “This could be a neuronal effect on learning memory systems.”
He went on to suggest that clinicians may consider these findings when selecting antihypertensive agents for their patients, with the caveat that all other prescribing factors have already been taking to account.
“In the event that you’re going to give an ACE inhibitor or an angiotensin receptor blocker anyway, and it ends up being a somewhat arbitrary decision in terms of which specific drug you’re going to give, then perhaps this is a piece of information you would take into account – that one gets in the brain and one doesn’t – in somebody at risk for cognitive decline,” Dr. Nation said.
Exact mechanisms of action unknown
Hélène Girouard, PhD, assistant professor of pharmacology and physiology at the University of Montreal, said in an interview that the findings are “of considerable importance, knowing that brain alterations could begin as much as 30 years before manifestation of dementia.”
Since 2003, Dr. Girouard has been studying the cognitive effects of antihypertensive medications. She noted that previous studies involving rodents “have shown beneficial effects [of BBB-crossing antihypertensive drugs] on cognition independent of their effects on blood pressure.”
The drugs’ exact mechanisms of action, however, remain elusive, according to Dr. Girouard, who offered several possible explanations, including amelioration of BBB disruption, brain inflammation, cerebral blood flow dysregulation, cholinergic dysfunction, and neurologic deficits. “Whether these mechanisms may explain Ho and colleagues’ observations remains to be established,” she added.
Andrea L. Schneider, MD, PhD, assistant professor of neurology at the University of Pennsylvania, Philadelphia, applauded the study, but ultimately suggested that more research is needed to impact clinical decision-making.
“The results of this important and well-done study suggest that further investigation into targeted mechanism-based approaches to selecting hypertension treatment agents, with a specific focus on cognitive outcomes, is warranted,” Dr. Schneider said in an interview. “Before changing clinical practice, further work is necessary to disentangle contributions of medication mechanism, comorbid vascular risk factors, and achieved blood pressure reduction, among others.”
The investigators disclosed support from the National Institutes of Health, the Alzheimer’s Association, the Waksman Foundation of Japan, and others. The interviewees reported no relevant conflicts of interest.
FROM HYPERTENSION
Is trouble falling asleep a modifiable risk factor for dementia?
, new research suggests.
Trouble falling asleep “may be a modifiable risk factor for later-life cognitive impairment and dementia,” said lead author Afsara Zaheed, a PhD candidate in clinical science, department of psychology, University of Michigan, Ann Arbor.
“Patients should also be aware of the importance of insomnia on cognitive functioning so that they can bring up these concerns with their providers early,” she said.
The findings were presented at Virtual SLEEP 2021, the 35th Annual Meeting of the Associated Professional Sleep Societies.
Poor sleep common with age
As many as one-half of older adults report having poor sleep quality and insomnia, and growing evidence suggests that insomnia may be a unique risk factor for cognitive decline in later life, Ms. Zaheed explained.
To investigate further, the researchers analyzed data on 2,496 adults aged 51 years and older who were participants in the Health and Retirement Study, a longitudinal study of aging in a nationally representative population of older adults.
In 2002, participants were asked how often they had trouble falling asleep, woke up during the night, woke up too early, and were not able to fall asleep again and how often they felt really rested when they woke up in the morning.
In 2016, participants’ cognition was assessed using a battery of neuropsychological tests that gauged episodic memory, executive function, language, visuospatial/construction, and processing speed.
Analyses controlled for sociodemographics, baseline global cognitive performance, and the influence of depressive symptoms and vascular disease.
Compared with other insomnia symptoms, having difficulty falling asleep in 2002 was the main insomnia symptom that was predictive of cognitive impairment 14 years later, in 2016.
More frequent trouble falling asleep was predictive of poorer episodic memory, executive function, language, processing speed, and visuospatial performance.
The associations between sleep initiation and later cognitive impairment were partially explained by depressive symptoms and vascular disease burden for all domains except episodic memory, which was only partially explained by depressive symptoms.
Unclear mechanism
Ms. Zaheed said research is needed to uncover neurophysiologic mechanisms underlying the observed associations. “It may be that chronic difficulty with falling asleep is associated with inflammatory or metabolic processes that negatively affect brain structure and function over time,” she said.
“Insomnia has also been linked with higher accumulation of protein aggregates in the brain that disrupt cell communication and are characteristic of late-life disorders such as Alzheimer’s disease,” she added.
“While our project did not directly investigate these potential causal pathways between insomnia and cognition, our results suggest that investigating these potential mechanisms is an important area for future research,” Ms. Zaheed said.
“While additional intervention research is needed to determine whether targeting insomnia in older patients can have lasting cognitive benefits, results from this study suggest that discussing insomnia symptoms at the primary care level may be beneficial for both doctors and patients,” she added.
“By targeting insomnia – for example, through an evidence-based cognitive–behavioral therapy approach – individuals may improve various mental and physical health outcomes in addition to improving their sleep quality,” Ms. Zaheed said.
Reached for comment, Shaheen E. Lakhan, MD, PhD, neurologist in Newton, Massachusetts, said, “There is a strong link between chronic sleep disturbances and cognitive impairment, including dementia.”
“This study further supports this link and specifically calls out initiating sleep (as opposed to staying asleep) as the culprit. It also raises the hypothesis that the link is primarily mediated by depression and vascular disease; however, the verdict is still out,” said Dr. Lakhan.
The study was funded by the National Institute on Aging. Ms. Zaheed and Dr. Lakhan have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, new research suggests.
Trouble falling asleep “may be a modifiable risk factor for later-life cognitive impairment and dementia,” said lead author Afsara Zaheed, a PhD candidate in clinical science, department of psychology, University of Michigan, Ann Arbor.
“Patients should also be aware of the importance of insomnia on cognitive functioning so that they can bring up these concerns with their providers early,” she said.
The findings were presented at Virtual SLEEP 2021, the 35th Annual Meeting of the Associated Professional Sleep Societies.
Poor sleep common with age
As many as one-half of older adults report having poor sleep quality and insomnia, and growing evidence suggests that insomnia may be a unique risk factor for cognitive decline in later life, Ms. Zaheed explained.
To investigate further, the researchers analyzed data on 2,496 adults aged 51 years and older who were participants in the Health and Retirement Study, a longitudinal study of aging in a nationally representative population of older adults.
In 2002, participants were asked how often they had trouble falling asleep, woke up during the night, woke up too early, and were not able to fall asleep again and how often they felt really rested when they woke up in the morning.
In 2016, participants’ cognition was assessed using a battery of neuropsychological tests that gauged episodic memory, executive function, language, visuospatial/construction, and processing speed.
Analyses controlled for sociodemographics, baseline global cognitive performance, and the influence of depressive symptoms and vascular disease.
Compared with other insomnia symptoms, having difficulty falling asleep in 2002 was the main insomnia symptom that was predictive of cognitive impairment 14 years later, in 2016.
More frequent trouble falling asleep was predictive of poorer episodic memory, executive function, language, processing speed, and visuospatial performance.
The associations between sleep initiation and later cognitive impairment were partially explained by depressive symptoms and vascular disease burden for all domains except episodic memory, which was only partially explained by depressive symptoms.
Unclear mechanism
Ms. Zaheed said research is needed to uncover neurophysiologic mechanisms underlying the observed associations. “It may be that chronic difficulty with falling asleep is associated with inflammatory or metabolic processes that negatively affect brain structure and function over time,” she said.
“Insomnia has also been linked with higher accumulation of protein aggregates in the brain that disrupt cell communication and are characteristic of late-life disorders such as Alzheimer’s disease,” she added.
“While our project did not directly investigate these potential causal pathways between insomnia and cognition, our results suggest that investigating these potential mechanisms is an important area for future research,” Ms. Zaheed said.
“While additional intervention research is needed to determine whether targeting insomnia in older patients can have lasting cognitive benefits, results from this study suggest that discussing insomnia symptoms at the primary care level may be beneficial for both doctors and patients,” she added.
“By targeting insomnia – for example, through an evidence-based cognitive–behavioral therapy approach – individuals may improve various mental and physical health outcomes in addition to improving their sleep quality,” Ms. Zaheed said.
Reached for comment, Shaheen E. Lakhan, MD, PhD, neurologist in Newton, Massachusetts, said, “There is a strong link between chronic sleep disturbances and cognitive impairment, including dementia.”
“This study further supports this link and specifically calls out initiating sleep (as opposed to staying asleep) as the culprit. It also raises the hypothesis that the link is primarily mediated by depression and vascular disease; however, the verdict is still out,” said Dr. Lakhan.
The study was funded by the National Institute on Aging. Ms. Zaheed and Dr. Lakhan have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, new research suggests.
Trouble falling asleep “may be a modifiable risk factor for later-life cognitive impairment and dementia,” said lead author Afsara Zaheed, a PhD candidate in clinical science, department of psychology, University of Michigan, Ann Arbor.
“Patients should also be aware of the importance of insomnia on cognitive functioning so that they can bring up these concerns with their providers early,” she said.
The findings were presented at Virtual SLEEP 2021, the 35th Annual Meeting of the Associated Professional Sleep Societies.
Poor sleep common with age
As many as one-half of older adults report having poor sleep quality and insomnia, and growing evidence suggests that insomnia may be a unique risk factor for cognitive decline in later life, Ms. Zaheed explained.
To investigate further, the researchers analyzed data on 2,496 adults aged 51 years and older who were participants in the Health and Retirement Study, a longitudinal study of aging in a nationally representative population of older adults.
In 2002, participants were asked how often they had trouble falling asleep, woke up during the night, woke up too early, and were not able to fall asleep again and how often they felt really rested when they woke up in the morning.
In 2016, participants’ cognition was assessed using a battery of neuropsychological tests that gauged episodic memory, executive function, language, visuospatial/construction, and processing speed.
Analyses controlled for sociodemographics, baseline global cognitive performance, and the influence of depressive symptoms and vascular disease.
Compared with other insomnia symptoms, having difficulty falling asleep in 2002 was the main insomnia symptom that was predictive of cognitive impairment 14 years later, in 2016.
More frequent trouble falling asleep was predictive of poorer episodic memory, executive function, language, processing speed, and visuospatial performance.
The associations between sleep initiation and later cognitive impairment were partially explained by depressive symptoms and vascular disease burden for all domains except episodic memory, which was only partially explained by depressive symptoms.
Unclear mechanism
Ms. Zaheed said research is needed to uncover neurophysiologic mechanisms underlying the observed associations. “It may be that chronic difficulty with falling asleep is associated with inflammatory or metabolic processes that negatively affect brain structure and function over time,” she said.
“Insomnia has also been linked with higher accumulation of protein aggregates in the brain that disrupt cell communication and are characteristic of late-life disorders such as Alzheimer’s disease,” she added.
“While our project did not directly investigate these potential causal pathways between insomnia and cognition, our results suggest that investigating these potential mechanisms is an important area for future research,” Ms. Zaheed said.
“While additional intervention research is needed to determine whether targeting insomnia in older patients can have lasting cognitive benefits, results from this study suggest that discussing insomnia symptoms at the primary care level may be beneficial for both doctors and patients,” she added.
“By targeting insomnia – for example, through an evidence-based cognitive–behavioral therapy approach – individuals may improve various mental and physical health outcomes in addition to improving their sleep quality,” Ms. Zaheed said.
Reached for comment, Shaheen E. Lakhan, MD, PhD, neurologist in Newton, Massachusetts, said, “There is a strong link between chronic sleep disturbances and cognitive impairment, including dementia.”
“This study further supports this link and specifically calls out initiating sleep (as opposed to staying asleep) as the culprit. It also raises the hypothesis that the link is primarily mediated by depression and vascular disease; however, the verdict is still out,” said Dr. Lakhan.
The study was funded by the National Institute on Aging. Ms. Zaheed and Dr. Lakhan have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Insomnia in children tied to mood and anxiety disorders in adulthood
, new research indicates. However, insomnia symptoms in childhood that remit in the transition to adolescence do not confer increased risk of mood or anxiety disorders later on, the study found.
“As insomnia symptoms may precipitate or maintain internalizing disorders, our findings further reinforce the need for early sleep interventions to prevent future mental health disorders,” said lead investigator Julio Fernandez-Mendoza, PhD, associate professor at Penn State University, Hershey.
He presented his research at Virtual SLEEP 2021, the 35th annual meeting of the Associated Professional Sleep Societies.
Results ‘very clear’
The findings are based on data from the Penn State Child Cohort, a longitudinal, population-based sample of 700 children with a median age of 9 years, including 421 who were followed up 8 years later as adolescents (median age, 16 years) and 502 who were followed up 15 years later as young adults (median age, 24 years).
The data are “very clear that the risk of having internalizing disorders in young adulthood associated with having persistent insomnia symptoms, since childhood through adolescence into young adulthood,” Dr. Fernandez-Mendoza said in his presentation.
A persistent developmental trajectory was associated with a threefold increased risk of adult internalizing disorder (hazard ratio, 3.19).
The risk of having an internalizing disorder in young adulthood associated with newly developing (incident) insomnia symptoms is about twofold higher (HR, 1.94), whereas the risk associated with the waxing and waning pattern of insomnia is 1.5-fold (HR, 1.53) higher and only marginally significant, he reported.
An equally important finding, said Dr. Fernandez-Mendoza, is that those who had remitted insomnia symptoms in the transition to adolescence and throughout young adulthood were not at increased risk of having an internalizing disorder in young adulthood.
“Insomnia symptoms in a persistent manner associated with long-term adverse mental health outcomes, but remission of those insomnia symptoms associated with a good prognosis,” he said.
It’s also important to note, he said, that about 40% of children do not outgrow their insomnia symptoms in the transition to adolescence and are at risk of developing mental health disorders later on during early adulthood.
Reached for comment, Nitun Verma, MD, a spokesperson for the American Academy of Sleep Medicine, said: “There is a connection with mood and anxiety disorders with sleep, especially insomnia. This is a good reminder that reviewing someone’s sleep habits should always be a part of assessing someone’s mental health.”
A version of this article first appeared on Medscape.com.
, new research indicates. However, insomnia symptoms in childhood that remit in the transition to adolescence do not confer increased risk of mood or anxiety disorders later on, the study found.
“As insomnia symptoms may precipitate or maintain internalizing disorders, our findings further reinforce the need for early sleep interventions to prevent future mental health disorders,” said lead investigator Julio Fernandez-Mendoza, PhD, associate professor at Penn State University, Hershey.
He presented his research at Virtual SLEEP 2021, the 35th annual meeting of the Associated Professional Sleep Societies.
Results ‘very clear’
The findings are based on data from the Penn State Child Cohort, a longitudinal, population-based sample of 700 children with a median age of 9 years, including 421 who were followed up 8 years later as adolescents (median age, 16 years) and 502 who were followed up 15 years later as young adults (median age, 24 years).
The data are “very clear that the risk of having internalizing disorders in young adulthood associated with having persistent insomnia symptoms, since childhood through adolescence into young adulthood,” Dr. Fernandez-Mendoza said in his presentation.
A persistent developmental trajectory was associated with a threefold increased risk of adult internalizing disorder (hazard ratio, 3.19).
The risk of having an internalizing disorder in young adulthood associated with newly developing (incident) insomnia symptoms is about twofold higher (HR, 1.94), whereas the risk associated with the waxing and waning pattern of insomnia is 1.5-fold (HR, 1.53) higher and only marginally significant, he reported.
An equally important finding, said Dr. Fernandez-Mendoza, is that those who had remitted insomnia symptoms in the transition to adolescence and throughout young adulthood were not at increased risk of having an internalizing disorder in young adulthood.
“Insomnia symptoms in a persistent manner associated with long-term adverse mental health outcomes, but remission of those insomnia symptoms associated with a good prognosis,” he said.
It’s also important to note, he said, that about 40% of children do not outgrow their insomnia symptoms in the transition to adolescence and are at risk of developing mental health disorders later on during early adulthood.
Reached for comment, Nitun Verma, MD, a spokesperson for the American Academy of Sleep Medicine, said: “There is a connection with mood and anxiety disorders with sleep, especially insomnia. This is a good reminder that reviewing someone’s sleep habits should always be a part of assessing someone’s mental health.”
A version of this article first appeared on Medscape.com.
, new research indicates. However, insomnia symptoms in childhood that remit in the transition to adolescence do not confer increased risk of mood or anxiety disorders later on, the study found.
“As insomnia symptoms may precipitate or maintain internalizing disorders, our findings further reinforce the need for early sleep interventions to prevent future mental health disorders,” said lead investigator Julio Fernandez-Mendoza, PhD, associate professor at Penn State University, Hershey.
He presented his research at Virtual SLEEP 2021, the 35th annual meeting of the Associated Professional Sleep Societies.
Results ‘very clear’
The findings are based on data from the Penn State Child Cohort, a longitudinal, population-based sample of 700 children with a median age of 9 years, including 421 who were followed up 8 years later as adolescents (median age, 16 years) and 502 who were followed up 15 years later as young adults (median age, 24 years).
The data are “very clear that the risk of having internalizing disorders in young adulthood associated with having persistent insomnia symptoms, since childhood through adolescence into young adulthood,” Dr. Fernandez-Mendoza said in his presentation.
A persistent developmental trajectory was associated with a threefold increased risk of adult internalizing disorder (hazard ratio, 3.19).
The risk of having an internalizing disorder in young adulthood associated with newly developing (incident) insomnia symptoms is about twofold higher (HR, 1.94), whereas the risk associated with the waxing and waning pattern of insomnia is 1.5-fold (HR, 1.53) higher and only marginally significant, he reported.
An equally important finding, said Dr. Fernandez-Mendoza, is that those who had remitted insomnia symptoms in the transition to adolescence and throughout young adulthood were not at increased risk of having an internalizing disorder in young adulthood.
“Insomnia symptoms in a persistent manner associated with long-term adverse mental health outcomes, but remission of those insomnia symptoms associated with a good prognosis,” he said.
It’s also important to note, he said, that about 40% of children do not outgrow their insomnia symptoms in the transition to adolescence and are at risk of developing mental health disorders later on during early adulthood.
Reached for comment, Nitun Verma, MD, a spokesperson for the American Academy of Sleep Medicine, said: “There is a connection with mood and anxiety disorders with sleep, especially insomnia. This is a good reminder that reviewing someone’s sleep habits should always be a part of assessing someone’s mental health.”
A version of this article first appeared on Medscape.com.
Neurodegeneration complicates psychiatric care for Parkinson’s patients
Managing depression and anxiety in Parkinson’s disease should start with a review of medications and involve multidisciplinary care, according to a recent summary of evidence.
“Depression and anxiety have a complex relationship with the disease and while the exact mechanism for this association is unknown, both disturbances occur with increased prevalence across the disease course and when present earlier in life, increase the risk of PD by about twofold,” wrote Gregory M. Pontone, MD, of Johns Hopkins University, Baltimore, and colleagues.
Randomized trials to guide treatment of anxiety and depression in patients with Parkinson’s disease (PD) are limited, the researchers noted. However, data from a longitudinal study showed that PD patients whose depression remitted spontaneously or responded to treatment were able to attain a level of function similar to that of never-depressed PD patients, Dr. Pontone and colleagues said.
The researchers offered a pair of treatment algorithms to help guide clinicians in managing depression and anxiety in PD. However, a caveat to keep in mind is that “the benefit of antidepressant medications, used for depression or anxiety, can be confounded when motor symptoms are not optimally treated,” the researchers emphasized.
For depression, the researchers advised starting with some lab work; “at a minimum we suggest checking a complete blood count, metabolic panel, TSH, B12, and folate,” they noted. They recommended an antidepressant, cognitive-behavioral therapy, or both, as a first-line treatment, such as monotherapy with selective norepinephrine reuptake inhibitors or selective serotonin reuptake inhibitors. They advised titrating the chosen monotherapy to a minimum effective dose over a 2- to 3-week period to assess response.
“We recommend continuing antidepressant therapy for at least 1 year based on literature in non-PD populations and anecdotal clinical experience. At 1 year, if not in remission, consider continuing treatment or augmenting to improve response,” the researchers said.
, and they recommended using anxiety rating scales to diagnose anxiety in PD. “Given the high prevalence of atypical anxiety syndromes in PD and their potential association with both motor and nonmotor symptoms of the disease, working with the neurologist to achieve optimal control of PD is an essential first step to alleviating anxiety,” they emphasized.
The researchers also advised addressing comorbidities, including cardiovascular disease, chronic pain, diabetes, gastrointestinal issues, hyperthyroidism, and lung disease, all of which can be associated with anxiety. Once comorbidities are addressed, they advised caution given the lack of evidence for efficacy of both pharmacologic and nonpharmacologic anxiety treatments for PD patients. However, first-tier treatment for anxiety could include monotherapy with serotonin-norepinephrine reuptake inhibitors or selective serotonin reuptake inhibitors, they said.
PD patients with depression and anxiety also may benefit from nonpharmacologic interventions, including exercise, mindfulness, relaxation therapy, and cognitive behavioral therapy the researchers said.
Although the algorithm may not differ significantly from current treatment protocols, it highlights aspects unique to PD patients, the researchers said. In particular, the algorithm shows “that interventions used for motor symptoms, for example, dopamine agonists, may be especially potent for mood in the PD population and that augmentation strategies, such as antipsychotics and lithium, may not be well tolerated given their outsized risk of adverse events in PD,” they said.
“While an article of this kind cannot hope to address the gap in knowledge on comparative efficacy between interventions, it can guide readers on the best strategies for implementation and risk mitigation in PD – essentially focusing more on effectiveness,” they concluded.
The study received no outside funding. Dr. Pontone disclosed serving as a consultant for Acadia Pharmaceuticals and Concert Pharmaceuticals.
Managing depression and anxiety in Parkinson’s disease should start with a review of medications and involve multidisciplinary care, according to a recent summary of evidence.
“Depression and anxiety have a complex relationship with the disease and while the exact mechanism for this association is unknown, both disturbances occur with increased prevalence across the disease course and when present earlier in life, increase the risk of PD by about twofold,” wrote Gregory M. Pontone, MD, of Johns Hopkins University, Baltimore, and colleagues.
Randomized trials to guide treatment of anxiety and depression in patients with Parkinson’s disease (PD) are limited, the researchers noted. However, data from a longitudinal study showed that PD patients whose depression remitted spontaneously or responded to treatment were able to attain a level of function similar to that of never-depressed PD patients, Dr. Pontone and colleagues said.
The researchers offered a pair of treatment algorithms to help guide clinicians in managing depression and anxiety in PD. However, a caveat to keep in mind is that “the benefit of antidepressant medications, used for depression or anxiety, can be confounded when motor symptoms are not optimally treated,” the researchers emphasized.
For depression, the researchers advised starting with some lab work; “at a minimum we suggest checking a complete blood count, metabolic panel, TSH, B12, and folate,” they noted. They recommended an antidepressant, cognitive-behavioral therapy, or both, as a first-line treatment, such as monotherapy with selective norepinephrine reuptake inhibitors or selective serotonin reuptake inhibitors. They advised titrating the chosen monotherapy to a minimum effective dose over a 2- to 3-week period to assess response.
“We recommend continuing antidepressant therapy for at least 1 year based on literature in non-PD populations and anecdotal clinical experience. At 1 year, if not in remission, consider continuing treatment or augmenting to improve response,” the researchers said.
, and they recommended using anxiety rating scales to diagnose anxiety in PD. “Given the high prevalence of atypical anxiety syndromes in PD and their potential association with both motor and nonmotor symptoms of the disease, working with the neurologist to achieve optimal control of PD is an essential first step to alleviating anxiety,” they emphasized.
The researchers also advised addressing comorbidities, including cardiovascular disease, chronic pain, diabetes, gastrointestinal issues, hyperthyroidism, and lung disease, all of which can be associated with anxiety. Once comorbidities are addressed, they advised caution given the lack of evidence for efficacy of both pharmacologic and nonpharmacologic anxiety treatments for PD patients. However, first-tier treatment for anxiety could include monotherapy with serotonin-norepinephrine reuptake inhibitors or selective serotonin reuptake inhibitors, they said.
PD patients with depression and anxiety also may benefit from nonpharmacologic interventions, including exercise, mindfulness, relaxation therapy, and cognitive behavioral therapy the researchers said.
Although the algorithm may not differ significantly from current treatment protocols, it highlights aspects unique to PD patients, the researchers said. In particular, the algorithm shows “that interventions used for motor symptoms, for example, dopamine agonists, may be especially potent for mood in the PD population and that augmentation strategies, such as antipsychotics and lithium, may not be well tolerated given their outsized risk of adverse events in PD,” they said.
“While an article of this kind cannot hope to address the gap in knowledge on comparative efficacy between interventions, it can guide readers on the best strategies for implementation and risk mitigation in PD – essentially focusing more on effectiveness,” they concluded.
The study received no outside funding. Dr. Pontone disclosed serving as a consultant for Acadia Pharmaceuticals and Concert Pharmaceuticals.
Managing depression and anxiety in Parkinson’s disease should start with a review of medications and involve multidisciplinary care, according to a recent summary of evidence.
“Depression and anxiety have a complex relationship with the disease and while the exact mechanism for this association is unknown, both disturbances occur with increased prevalence across the disease course and when present earlier in life, increase the risk of PD by about twofold,” wrote Gregory M. Pontone, MD, of Johns Hopkins University, Baltimore, and colleagues.
Randomized trials to guide treatment of anxiety and depression in patients with Parkinson’s disease (PD) are limited, the researchers noted. However, data from a longitudinal study showed that PD patients whose depression remitted spontaneously or responded to treatment were able to attain a level of function similar to that of never-depressed PD patients, Dr. Pontone and colleagues said.
The researchers offered a pair of treatment algorithms to help guide clinicians in managing depression and anxiety in PD. However, a caveat to keep in mind is that “the benefit of antidepressant medications, used for depression or anxiety, can be confounded when motor symptoms are not optimally treated,” the researchers emphasized.
For depression, the researchers advised starting with some lab work; “at a minimum we suggest checking a complete blood count, metabolic panel, TSH, B12, and folate,” they noted. They recommended an antidepressant, cognitive-behavioral therapy, or both, as a first-line treatment, such as monotherapy with selective norepinephrine reuptake inhibitors or selective serotonin reuptake inhibitors. They advised titrating the chosen monotherapy to a minimum effective dose over a 2- to 3-week period to assess response.
“We recommend continuing antidepressant therapy for at least 1 year based on literature in non-PD populations and anecdotal clinical experience. At 1 year, if not in remission, consider continuing treatment or augmenting to improve response,” the researchers said.
, and they recommended using anxiety rating scales to diagnose anxiety in PD. “Given the high prevalence of atypical anxiety syndromes in PD and their potential association with both motor and nonmotor symptoms of the disease, working with the neurologist to achieve optimal control of PD is an essential first step to alleviating anxiety,” they emphasized.
The researchers also advised addressing comorbidities, including cardiovascular disease, chronic pain, diabetes, gastrointestinal issues, hyperthyroidism, and lung disease, all of which can be associated with anxiety. Once comorbidities are addressed, they advised caution given the lack of evidence for efficacy of both pharmacologic and nonpharmacologic anxiety treatments for PD patients. However, first-tier treatment for anxiety could include monotherapy with serotonin-norepinephrine reuptake inhibitors or selective serotonin reuptake inhibitors, they said.
PD patients with depression and anxiety also may benefit from nonpharmacologic interventions, including exercise, mindfulness, relaxation therapy, and cognitive behavioral therapy the researchers said.
Although the algorithm may not differ significantly from current treatment protocols, it highlights aspects unique to PD patients, the researchers said. In particular, the algorithm shows “that interventions used for motor symptoms, for example, dopamine agonists, may be especially potent for mood in the PD population and that augmentation strategies, such as antipsychotics and lithium, may not be well tolerated given their outsized risk of adverse events in PD,” they said.
“While an article of this kind cannot hope to address the gap in knowledge on comparative efficacy between interventions, it can guide readers on the best strategies for implementation and risk mitigation in PD – essentially focusing more on effectiveness,” they concluded.
The study received no outside funding. Dr. Pontone disclosed serving as a consultant for Acadia Pharmaceuticals and Concert Pharmaceuticals.
FROM THE AMERICAN JOURNAL OF GERIATRIC PSYCHIATRY
Bariatric surgery tied to 22% lower 5-year stroke risk
Patients with obesity who underwent bariatric surgery had 46% lower odds of stroke 1 year later, similar odds of stroke 3 years later, and 22% lower odds of stroke 5 years later, compared with matched control patients, in new research.
Michael D. Williams, MD, presented the study findings (abstract A002) at the annual meeting of the American Society for Metabolic & Bariatric Surgery.
The findings are “very good news,” even though the protection against stroke declined further out from the surgery, John D. Scott, MD, scientific program chair of the ASMBS meeting, told this news organization.
The investigators matched more than 56,000 patients with obesity who had bariatric surgery with an equal number of similar patients who did not have this surgery, from a large national insurance database, in what they believe is the largest study of this to date.
“Any intervention that decreases your risk of [cardiovascular] events is good news,” said Dr. Scott, a clinical professor of surgery at the University of South Carolina, Greenville, and metabolic and bariatric surgery director at Prisma Health in Greenville, S.C. “And having a 22%-45% chance of reduction in stroke risk is a very worthwhile intervention.”
Asked how this would change the way clinicians inform patients of what to expect from bariatric surgery, he said: “I would advise patients that studies like this show that surgery would not increase your risk of having a stroke.
“This is consistent with many studies that show that the risks of all macrovascular events decrease after the comorbidity reductions seen after surgery.”
According to Dr. Scott, “the next steps might include a prospective randomized trial of medical treatment versus surgery alone for [cardiovascular]/stroke outcomes, but this is unlikely.”
Similarly, Dr. Williams told this news organization that “I would tell [patients] that surgery is an effective and durable method for weight loss. It also can improve comorbid conditions, particularly diabetes and hypertension.”
Even with this study, “I’m not sure it’s appropriate to say that bariatric surgery will reduce the risk of stroke,” he cautioned.
“However, as we continue to investigate the effects of bariatric surgery, this study contributes to the greater body of knowledge that suggests that reduction in ischemic stroke risk is yet another benefit of bariatric surgery.”
The assigned discussant, Corrigan L. McBride, MD, MBA wanted to know if the lower odds ratio at 1 year might be because preoperative patient selection might eliminate patients at high risk of poor cardiovascular outcomes.
Dr. Williams, a resident at Rush Medical College, Chicago, replied that it is difficult to eliminate potential selection bias, despite best efforts, but this study shows that he can tell patients: “Having surgery is not going to increases your risk of stroke.”
“This is an important study,” Dr. McBride, professor and chief of minimally invasive surgery and bariatric surgery, University of Nebraska Medical Center, Omaha, told this news organization.
“It is the first large study to show a decreased [or no increased] risk of stroke 1, 3, and 5 years after bariatric surgery compared to matched patients, and it had enough data to look at stroke as a standalone endpoint,” Dr. McBride said. “It is important too, for patients and their physicians to understand that there is a lower chance of them having a stroke if they have surgery than if they do not.”
‘Important,’ ‘good news’ for stroke risk after bariatric surgery
The impact of bariatric surgery on remission of type 2 diabetes is well known, Dr. Williams noted, and other studies have reported how bariatric surgery affects the risk of major adverse cardiovascular events – a composite of stroke, myocardial infarction, coronary artery disease, and all-cause death – including a study presented in the same meeting session.
However, a very large sample size is needed to be able to demonstrate the effect of bariatric surgery on stroke, since stroke is a rare event.
The researchers analyzed data from the Mariner (PearlDiver.) all-payer insurance national claims database of patients in the United States.
They matched 56,514 patients with a body mass index over 35 kg/m2 and comorbidities or a BMI of more than 40 who underwent sleeve gastrectomy or Roux-en-Y gastric bypass during 2010-2019 with 56,514 control patients who did not undergo bariatric surgery.
A year after bariatric surgery, patients in that group had a lower stroke rate than patients in the control group (0.6% vs. 1.2%), and they had close to 50% lower odds of having a stroke (odds ratio, 0.54; 95% CI, 0.47-0.61).
Three years after bariatric surgery, there were 44,948 patients in each group; the rate of stroke was 2.1% in the surgery group and 2.2% in the control group, and there was no significant difference in the odds of having a stroke (OR, 0.96; 95% CI, 0.91-1.00).
Five years after bariatric surgery, there were 27,619 patients in each group; the stroke rate was lower in the bariatric surgery group than in the control group (2.8% vs 3.6%), but reduced odds of stroke was not as great as after 1 year (OR, 0.78; 95% CI, 0.65-0.90).
Dr. Williams has no relevant financial disclosures. Dr. McBride and Dr. Scott disclosed that they are speakers/trainers/faculty advisers for Gore. Dr. Scott is also a consultant for C-SATS (part of Johnson & Johnson).
Patients with obesity who underwent bariatric surgery had 46% lower odds of stroke 1 year later, similar odds of stroke 3 years later, and 22% lower odds of stroke 5 years later, compared with matched control patients, in new research.
Michael D. Williams, MD, presented the study findings (abstract A002) at the annual meeting of the American Society for Metabolic & Bariatric Surgery.
The findings are “very good news,” even though the protection against stroke declined further out from the surgery, John D. Scott, MD, scientific program chair of the ASMBS meeting, told this news organization.
The investigators matched more than 56,000 patients with obesity who had bariatric surgery with an equal number of similar patients who did not have this surgery, from a large national insurance database, in what they believe is the largest study of this to date.
“Any intervention that decreases your risk of [cardiovascular] events is good news,” said Dr. Scott, a clinical professor of surgery at the University of South Carolina, Greenville, and metabolic and bariatric surgery director at Prisma Health in Greenville, S.C. “And having a 22%-45% chance of reduction in stroke risk is a very worthwhile intervention.”
Asked how this would change the way clinicians inform patients of what to expect from bariatric surgery, he said: “I would advise patients that studies like this show that surgery would not increase your risk of having a stroke.
“This is consistent with many studies that show that the risks of all macrovascular events decrease after the comorbidity reductions seen after surgery.”
According to Dr. Scott, “the next steps might include a prospective randomized trial of medical treatment versus surgery alone for [cardiovascular]/stroke outcomes, but this is unlikely.”
Similarly, Dr. Williams told this news organization that “I would tell [patients] that surgery is an effective and durable method for weight loss. It also can improve comorbid conditions, particularly diabetes and hypertension.”
Even with this study, “I’m not sure it’s appropriate to say that bariatric surgery will reduce the risk of stroke,” he cautioned.
“However, as we continue to investigate the effects of bariatric surgery, this study contributes to the greater body of knowledge that suggests that reduction in ischemic stroke risk is yet another benefit of bariatric surgery.”
The assigned discussant, Corrigan L. McBride, MD, MBA wanted to know if the lower odds ratio at 1 year might be because preoperative patient selection might eliminate patients at high risk of poor cardiovascular outcomes.
Dr. Williams, a resident at Rush Medical College, Chicago, replied that it is difficult to eliminate potential selection bias, despite best efforts, but this study shows that he can tell patients: “Having surgery is not going to increases your risk of stroke.”
“This is an important study,” Dr. McBride, professor and chief of minimally invasive surgery and bariatric surgery, University of Nebraska Medical Center, Omaha, told this news organization.
“It is the first large study to show a decreased [or no increased] risk of stroke 1, 3, and 5 years after bariatric surgery compared to matched patients, and it had enough data to look at stroke as a standalone endpoint,” Dr. McBride said. “It is important too, for patients and their physicians to understand that there is a lower chance of them having a stroke if they have surgery than if they do not.”
‘Important,’ ‘good news’ for stroke risk after bariatric surgery
The impact of bariatric surgery on remission of type 2 diabetes is well known, Dr. Williams noted, and other studies have reported how bariatric surgery affects the risk of major adverse cardiovascular events – a composite of stroke, myocardial infarction, coronary artery disease, and all-cause death – including a study presented in the same meeting session.
However, a very large sample size is needed to be able to demonstrate the effect of bariatric surgery on stroke, since stroke is a rare event.
The researchers analyzed data from the Mariner (PearlDiver.) all-payer insurance national claims database of patients in the United States.
They matched 56,514 patients with a body mass index over 35 kg/m2 and comorbidities or a BMI of more than 40 who underwent sleeve gastrectomy or Roux-en-Y gastric bypass during 2010-2019 with 56,514 control patients who did not undergo bariatric surgery.
A year after bariatric surgery, patients in that group had a lower stroke rate than patients in the control group (0.6% vs. 1.2%), and they had close to 50% lower odds of having a stroke (odds ratio, 0.54; 95% CI, 0.47-0.61).
Three years after bariatric surgery, there were 44,948 patients in each group; the rate of stroke was 2.1% in the surgery group and 2.2% in the control group, and there was no significant difference in the odds of having a stroke (OR, 0.96; 95% CI, 0.91-1.00).
Five years after bariatric surgery, there were 27,619 patients in each group; the stroke rate was lower in the bariatric surgery group than in the control group (2.8% vs 3.6%), but reduced odds of stroke was not as great as after 1 year (OR, 0.78; 95% CI, 0.65-0.90).
Dr. Williams has no relevant financial disclosures. Dr. McBride and Dr. Scott disclosed that they are speakers/trainers/faculty advisers for Gore. Dr. Scott is also a consultant for C-SATS (part of Johnson & Johnson).
Patients with obesity who underwent bariatric surgery had 46% lower odds of stroke 1 year later, similar odds of stroke 3 years later, and 22% lower odds of stroke 5 years later, compared with matched control patients, in new research.
Michael D. Williams, MD, presented the study findings (abstract A002) at the annual meeting of the American Society for Metabolic & Bariatric Surgery.
The findings are “very good news,” even though the protection against stroke declined further out from the surgery, John D. Scott, MD, scientific program chair of the ASMBS meeting, told this news organization.
The investigators matched more than 56,000 patients with obesity who had bariatric surgery with an equal number of similar patients who did not have this surgery, from a large national insurance database, in what they believe is the largest study of this to date.
“Any intervention that decreases your risk of [cardiovascular] events is good news,” said Dr. Scott, a clinical professor of surgery at the University of South Carolina, Greenville, and metabolic and bariatric surgery director at Prisma Health in Greenville, S.C. “And having a 22%-45% chance of reduction in stroke risk is a very worthwhile intervention.”
Asked how this would change the way clinicians inform patients of what to expect from bariatric surgery, he said: “I would advise patients that studies like this show that surgery would not increase your risk of having a stroke.
“This is consistent with many studies that show that the risks of all macrovascular events decrease after the comorbidity reductions seen after surgery.”
According to Dr. Scott, “the next steps might include a prospective randomized trial of medical treatment versus surgery alone for [cardiovascular]/stroke outcomes, but this is unlikely.”
Similarly, Dr. Williams told this news organization that “I would tell [patients] that surgery is an effective and durable method for weight loss. It also can improve comorbid conditions, particularly diabetes and hypertension.”
Even with this study, “I’m not sure it’s appropriate to say that bariatric surgery will reduce the risk of stroke,” he cautioned.
“However, as we continue to investigate the effects of bariatric surgery, this study contributes to the greater body of knowledge that suggests that reduction in ischemic stroke risk is yet another benefit of bariatric surgery.”
The assigned discussant, Corrigan L. McBride, MD, MBA wanted to know if the lower odds ratio at 1 year might be because preoperative patient selection might eliminate patients at high risk of poor cardiovascular outcomes.
Dr. Williams, a resident at Rush Medical College, Chicago, replied that it is difficult to eliminate potential selection bias, despite best efforts, but this study shows that he can tell patients: “Having surgery is not going to increases your risk of stroke.”
“This is an important study,” Dr. McBride, professor and chief of minimally invasive surgery and bariatric surgery, University of Nebraska Medical Center, Omaha, told this news organization.
“It is the first large study to show a decreased [or no increased] risk of stroke 1, 3, and 5 years after bariatric surgery compared to matched patients, and it had enough data to look at stroke as a standalone endpoint,” Dr. McBride said. “It is important too, for patients and their physicians to understand that there is a lower chance of them having a stroke if they have surgery than if they do not.”
‘Important,’ ‘good news’ for stroke risk after bariatric surgery
The impact of bariatric surgery on remission of type 2 diabetes is well known, Dr. Williams noted, and other studies have reported how bariatric surgery affects the risk of major adverse cardiovascular events – a composite of stroke, myocardial infarction, coronary artery disease, and all-cause death – including a study presented in the same meeting session.
However, a very large sample size is needed to be able to demonstrate the effect of bariatric surgery on stroke, since stroke is a rare event.
The researchers analyzed data from the Mariner (PearlDiver.) all-payer insurance national claims database of patients in the United States.
They matched 56,514 patients with a body mass index over 35 kg/m2 and comorbidities or a BMI of more than 40 who underwent sleeve gastrectomy or Roux-en-Y gastric bypass during 2010-2019 with 56,514 control patients who did not undergo bariatric surgery.
A year after bariatric surgery, patients in that group had a lower stroke rate than patients in the control group (0.6% vs. 1.2%), and they had close to 50% lower odds of having a stroke (odds ratio, 0.54; 95% CI, 0.47-0.61).
Three years after bariatric surgery, there were 44,948 patients in each group; the rate of stroke was 2.1% in the surgery group and 2.2% in the control group, and there was no significant difference in the odds of having a stroke (OR, 0.96; 95% CI, 0.91-1.00).
Five years after bariatric surgery, there were 27,619 patients in each group; the stroke rate was lower in the bariatric surgery group than in the control group (2.8% vs 3.6%), but reduced odds of stroke was not as great as after 1 year (OR, 0.78; 95% CI, 0.65-0.90).
Dr. Williams has no relevant financial disclosures. Dr. McBride and Dr. Scott disclosed that they are speakers/trainers/faculty advisers for Gore. Dr. Scott is also a consultant for C-SATS (part of Johnson & Johnson).
FROM ASMBS 2021
Reversal agents curb DOAC-related bleeding but deaths still high
Agents that reverse the effect of direct oral anticoagulants (DOACs) are highly effective in patients with severe bleeding, but mortality rates remain high despite their use, a meta-analysis shows.
Effective hemostasis was achieved in 78.5% of patients treated with a reversal agent, whereas failure to achieve hemostasis was associated with more than a threefold higher relative risk for death (relative risk, 3.63; 95% confidence interval, 2.56-5.16).
“This has implications in practice because it emphasizes the need for achieving effective hemostasis, if not with only one agent, trying other agents or treatment modalities, because it is a strong predictor of survival,” lead author Antonio Gómez-Outes, MD, PhD, said in an interview.
The bad news, he said, is that the mortality rate was still significant, at 17.7%, and approximately half of patients with DOAC-related severe intracranial bleeding survived with long-term moderate/severe disability.
“The lesson is to prevent these bleeding events because once they appear, even if you give an antidote, the outcome is poor, particularly for intracranial bleeding,” said Dr. Gómez-Outes, division of pharmacology and clinical drug evaluation, Spanish Agency for Medicines and Medical Devices, Madrid.
To put this in context, mortality rates were close to 50% after intracranial bleeding a decade ago when there were no antidotes or reversal agents, he observed. “So to some extent, patient care has improved, and the outcome has improved, but there is a long road to improve regarding disability.”
More than 100,000 DOAC-related major bleeding cases occur each year in the United States and European Union, Dr. Gómez-Outes said, and about half are severe enough to require hospitalization and potentially the use of a reversal agent. These include idarucizumab (Praxbind) for dabigatran reversal and prothombin complex concentrates (4CCC) or andexanet alpha (Andexxa) for reversal of direct factor Xa inhibitors like rivaroxaban, apixaban, and edoxaban.
As reported in the June 22 issue of the Journal of the American College of Cardiology, the meta-analysis comprised 4,735 patients (mean age, 77 years; 57% male) with severe DOAC-related bleeding who received 4PCC (n = 2,688), idarucizumab (n = 1,111), or andexanet (n = 936) in 60 studies between January 2010 and December 2020.
Atrial fibrillation (AFib) was the most common reason for use of a DOAC (82%), followed by venous thromboembolism (14%). Rivaroxaban was used in 36%, apixaban in 32%, dabigatran in 31%, and edoxaban in 1%.
The index bleeding event was intracranial hemorrhage (ICH) in 55%. Anticoagulation was restarted in 57% of patients an average of 11 days after admission.
Mortality rates were 20.2% in patients with ICH and 15.4% in those with extracranial bleeding. There were no differences in death rates by reversal agent used, type of study, risk for bias, or study sponsorship in meta-regression analysis.
Rebleeding occurred in 13.2% of patients; 82.0% of these events were described as an ICH, and 78.0% occurred after anticoagulation was restarted.
The overall rate of thromboembolism was 4.6%. The risk was particularly high with andexanet, at 10.7%, and relatively low with idarucizumab (3.8%) and 4PCC (4.3%), the authors note.
“Our meta-analysis suggests specific reversal with andexanet is not superior to unspecific reversal with 4PCC, and that’s good news because many centers, in many countries, have no access to specific antidotes that are more costly,” Dr. Gómez-Outes said. “4PCC is an effective and relatively safe drug, so it’s still a good option for these patients.”
Labeling for andexanet includes a warning for thromboembolic events, but in the absence of direct comparisons, the findings should be interpreted with caution, he added. Further insights are expected from an ongoing randomized trial of andexanet and standard of care in 900 patients who present with acute ICH less than 15 hours after taking an oral factor Xa inhibitor. The preliminary completion date is set for 2023.
“The meta-analysis raises awareness about the rates of mortality and thromboembolism after reversal agent administration, although understanding the implications of these data is challenging,” Christopher Granger, MD, and Sean P. Pokomey, MD, MBA, Duke University Medical Center, Durham, N.C., say in an accompanying editorial.
The fact that failure to achieve hemostasis was associated with death is expected and might be related to the way hemostasis was defined, rather than the actual failure of the hemostatic treatments, they suggest. “The prothrombotic effects of each agent, including andexanet, need to be better understood, as clinicians work toward including reversal agents into algorithms for bleeding management.”
Effective hemostasis was defined in the studies through various methods as: “Excellent/good” using the Sarode and ANNEXA-4 scales; “yes” in the International Society on Thrombosis and Hemostasis Scale; and with other scales and through clinical judgment.
Although the size of the meta-analysis dwarfs previous reviews, the editorialists and authors point out that 47 of the 60 studies were retrospective, only two had control groups, and 45 had a high risk for bias.
In general, there was also poor reporting of key clinical data, such as postbleeding anticoagulation management, and a limitation of the mortality analysis is that it was based in selected patients with effective hemostasis assessed within 48 hours, which may not capture early deaths, the authors note.
“The morbidity and mortality from ischemic strokes as a result of undertreatment of stroke prevention in patients with AFib continue to dwarf the bleeding related mortality among patients with AFib and on DOACs, and thus the number one priority is to treat nearly all patients with AFib with a DOAC,” Dr. Granger and Dr. Pokomey conclude. “The availability of reversal agents for DOACs should provide reassurance, with another tool in our armamentarium, to providers to prescribe OACs for stroke prevention.”
No funding/grant support was received to conduct the study. Coauthor Ramón Lecumberri has received personal fees from Boehringer Ingelheim and Bristol Myers Squibb outside the submitted work. All other authors report no relevant financial relationships. Dr. Granger has received research and consulting fees from Bristol Myers Squibb, Pfizer, Boehringer Ingelheim, Bayer, Janssen, Boston Scientific, Apple, AstraZeneca, Novartis, AbbVie, Biomed, CeleCor, GSK, Novartis, Medtronic, Merck, Novo Nordisk, Philips, Rho, and the U.S. Food and Drug Administration. Dr. Pokomey has received modest consulting support from Bristol Myers Squibb, Pfizer, Boston Scientific, Medtronic, Janssen, and Zoll; modest research support from Gilead, Boston Scientific, Bristol Myers Squibb, Pfizer, and Janssen; and significant research support from the FDA.
A version of this article first appeared on Medscape.com.
Agents that reverse the effect of direct oral anticoagulants (DOACs) are highly effective in patients with severe bleeding, but mortality rates remain high despite their use, a meta-analysis shows.
Effective hemostasis was achieved in 78.5% of patients treated with a reversal agent, whereas failure to achieve hemostasis was associated with more than a threefold higher relative risk for death (relative risk, 3.63; 95% confidence interval, 2.56-5.16).
“This has implications in practice because it emphasizes the need for achieving effective hemostasis, if not with only one agent, trying other agents or treatment modalities, because it is a strong predictor of survival,” lead author Antonio Gómez-Outes, MD, PhD, said in an interview.
The bad news, he said, is that the mortality rate was still significant, at 17.7%, and approximately half of patients with DOAC-related severe intracranial bleeding survived with long-term moderate/severe disability.
“The lesson is to prevent these bleeding events because once they appear, even if you give an antidote, the outcome is poor, particularly for intracranial bleeding,” said Dr. Gómez-Outes, division of pharmacology and clinical drug evaluation, Spanish Agency for Medicines and Medical Devices, Madrid.
To put this in context, mortality rates were close to 50% after intracranial bleeding a decade ago when there were no antidotes or reversal agents, he observed. “So to some extent, patient care has improved, and the outcome has improved, but there is a long road to improve regarding disability.”
More than 100,000 DOAC-related major bleeding cases occur each year in the United States and European Union, Dr. Gómez-Outes said, and about half are severe enough to require hospitalization and potentially the use of a reversal agent. These include idarucizumab (Praxbind) for dabigatran reversal and prothombin complex concentrates (4CCC) or andexanet alpha (Andexxa) for reversal of direct factor Xa inhibitors like rivaroxaban, apixaban, and edoxaban.
As reported in the June 22 issue of the Journal of the American College of Cardiology, the meta-analysis comprised 4,735 patients (mean age, 77 years; 57% male) with severe DOAC-related bleeding who received 4PCC (n = 2,688), idarucizumab (n = 1,111), or andexanet (n = 936) in 60 studies between January 2010 and December 2020.
Atrial fibrillation (AFib) was the most common reason for use of a DOAC (82%), followed by venous thromboembolism (14%). Rivaroxaban was used in 36%, apixaban in 32%, dabigatran in 31%, and edoxaban in 1%.
The index bleeding event was intracranial hemorrhage (ICH) in 55%. Anticoagulation was restarted in 57% of patients an average of 11 days after admission.
Mortality rates were 20.2% in patients with ICH and 15.4% in those with extracranial bleeding. There were no differences in death rates by reversal agent used, type of study, risk for bias, or study sponsorship in meta-regression analysis.
Rebleeding occurred in 13.2% of patients; 82.0% of these events were described as an ICH, and 78.0% occurred after anticoagulation was restarted.
The overall rate of thromboembolism was 4.6%. The risk was particularly high with andexanet, at 10.7%, and relatively low with idarucizumab (3.8%) and 4PCC (4.3%), the authors note.
“Our meta-analysis suggests specific reversal with andexanet is not superior to unspecific reversal with 4PCC, and that’s good news because many centers, in many countries, have no access to specific antidotes that are more costly,” Dr. Gómez-Outes said. “4PCC is an effective and relatively safe drug, so it’s still a good option for these patients.”
Labeling for andexanet includes a warning for thromboembolic events, but in the absence of direct comparisons, the findings should be interpreted with caution, he added. Further insights are expected from an ongoing randomized trial of andexanet and standard of care in 900 patients who present with acute ICH less than 15 hours after taking an oral factor Xa inhibitor. The preliminary completion date is set for 2023.
“The meta-analysis raises awareness about the rates of mortality and thromboembolism after reversal agent administration, although understanding the implications of these data is challenging,” Christopher Granger, MD, and Sean P. Pokomey, MD, MBA, Duke University Medical Center, Durham, N.C., say in an accompanying editorial.
The fact that failure to achieve hemostasis was associated with death is expected and might be related to the way hemostasis was defined, rather than the actual failure of the hemostatic treatments, they suggest. “The prothrombotic effects of each agent, including andexanet, need to be better understood, as clinicians work toward including reversal agents into algorithms for bleeding management.”
Effective hemostasis was defined in the studies through various methods as: “Excellent/good” using the Sarode and ANNEXA-4 scales; “yes” in the International Society on Thrombosis and Hemostasis Scale; and with other scales and through clinical judgment.
Although the size of the meta-analysis dwarfs previous reviews, the editorialists and authors point out that 47 of the 60 studies were retrospective, only two had control groups, and 45 had a high risk for bias.
In general, there was also poor reporting of key clinical data, such as postbleeding anticoagulation management, and a limitation of the mortality analysis is that it was based in selected patients with effective hemostasis assessed within 48 hours, which may not capture early deaths, the authors note.
“The morbidity and mortality from ischemic strokes as a result of undertreatment of stroke prevention in patients with AFib continue to dwarf the bleeding related mortality among patients with AFib and on DOACs, and thus the number one priority is to treat nearly all patients with AFib with a DOAC,” Dr. Granger and Dr. Pokomey conclude. “The availability of reversal agents for DOACs should provide reassurance, with another tool in our armamentarium, to providers to prescribe OACs for stroke prevention.”
No funding/grant support was received to conduct the study. Coauthor Ramón Lecumberri has received personal fees from Boehringer Ingelheim and Bristol Myers Squibb outside the submitted work. All other authors report no relevant financial relationships. Dr. Granger has received research and consulting fees from Bristol Myers Squibb, Pfizer, Boehringer Ingelheim, Bayer, Janssen, Boston Scientific, Apple, AstraZeneca, Novartis, AbbVie, Biomed, CeleCor, GSK, Novartis, Medtronic, Merck, Novo Nordisk, Philips, Rho, and the U.S. Food and Drug Administration. Dr. Pokomey has received modest consulting support from Bristol Myers Squibb, Pfizer, Boston Scientific, Medtronic, Janssen, and Zoll; modest research support from Gilead, Boston Scientific, Bristol Myers Squibb, Pfizer, and Janssen; and significant research support from the FDA.
A version of this article first appeared on Medscape.com.
Agents that reverse the effect of direct oral anticoagulants (DOACs) are highly effective in patients with severe bleeding, but mortality rates remain high despite their use, a meta-analysis shows.
Effective hemostasis was achieved in 78.5% of patients treated with a reversal agent, whereas failure to achieve hemostasis was associated with more than a threefold higher relative risk for death (relative risk, 3.63; 95% confidence interval, 2.56-5.16).
“This has implications in practice because it emphasizes the need for achieving effective hemostasis, if not with only one agent, trying other agents or treatment modalities, because it is a strong predictor of survival,” lead author Antonio Gómez-Outes, MD, PhD, said in an interview.
The bad news, he said, is that the mortality rate was still significant, at 17.7%, and approximately half of patients with DOAC-related severe intracranial bleeding survived with long-term moderate/severe disability.
“The lesson is to prevent these bleeding events because once they appear, even if you give an antidote, the outcome is poor, particularly for intracranial bleeding,” said Dr. Gómez-Outes, division of pharmacology and clinical drug evaluation, Spanish Agency for Medicines and Medical Devices, Madrid.
To put this in context, mortality rates were close to 50% after intracranial bleeding a decade ago when there were no antidotes or reversal agents, he observed. “So to some extent, patient care has improved, and the outcome has improved, but there is a long road to improve regarding disability.”
More than 100,000 DOAC-related major bleeding cases occur each year in the United States and European Union, Dr. Gómez-Outes said, and about half are severe enough to require hospitalization and potentially the use of a reversal agent. These include idarucizumab (Praxbind) for dabigatran reversal and prothombin complex concentrates (4CCC) or andexanet alpha (Andexxa) for reversal of direct factor Xa inhibitors like rivaroxaban, apixaban, and edoxaban.
As reported in the June 22 issue of the Journal of the American College of Cardiology, the meta-analysis comprised 4,735 patients (mean age, 77 years; 57% male) with severe DOAC-related bleeding who received 4PCC (n = 2,688), idarucizumab (n = 1,111), or andexanet (n = 936) in 60 studies between January 2010 and December 2020.
Atrial fibrillation (AFib) was the most common reason for use of a DOAC (82%), followed by venous thromboembolism (14%). Rivaroxaban was used in 36%, apixaban in 32%, dabigatran in 31%, and edoxaban in 1%.
The index bleeding event was intracranial hemorrhage (ICH) in 55%. Anticoagulation was restarted in 57% of patients an average of 11 days after admission.
Mortality rates were 20.2% in patients with ICH and 15.4% in those with extracranial bleeding. There were no differences in death rates by reversal agent used, type of study, risk for bias, or study sponsorship in meta-regression analysis.
Rebleeding occurred in 13.2% of patients; 82.0% of these events were described as an ICH, and 78.0% occurred after anticoagulation was restarted.
The overall rate of thromboembolism was 4.6%. The risk was particularly high with andexanet, at 10.7%, and relatively low with idarucizumab (3.8%) and 4PCC (4.3%), the authors note.
“Our meta-analysis suggests specific reversal with andexanet is not superior to unspecific reversal with 4PCC, and that’s good news because many centers, in many countries, have no access to specific antidotes that are more costly,” Dr. Gómez-Outes said. “4PCC is an effective and relatively safe drug, so it’s still a good option for these patients.”
Labeling for andexanet includes a warning for thromboembolic events, but in the absence of direct comparisons, the findings should be interpreted with caution, he added. Further insights are expected from an ongoing randomized trial of andexanet and standard of care in 900 patients who present with acute ICH less than 15 hours after taking an oral factor Xa inhibitor. The preliminary completion date is set for 2023.
“The meta-analysis raises awareness about the rates of mortality and thromboembolism after reversal agent administration, although understanding the implications of these data is challenging,” Christopher Granger, MD, and Sean P. Pokomey, MD, MBA, Duke University Medical Center, Durham, N.C., say in an accompanying editorial.
The fact that failure to achieve hemostasis was associated with death is expected and might be related to the way hemostasis was defined, rather than the actual failure of the hemostatic treatments, they suggest. “The prothrombotic effects of each agent, including andexanet, need to be better understood, as clinicians work toward including reversal agents into algorithms for bleeding management.”
Effective hemostasis was defined in the studies through various methods as: “Excellent/good” using the Sarode and ANNEXA-4 scales; “yes” in the International Society on Thrombosis and Hemostasis Scale; and with other scales and through clinical judgment.
Although the size of the meta-analysis dwarfs previous reviews, the editorialists and authors point out that 47 of the 60 studies were retrospective, only two had control groups, and 45 had a high risk for bias.
In general, there was also poor reporting of key clinical data, such as postbleeding anticoagulation management, and a limitation of the mortality analysis is that it was based in selected patients with effective hemostasis assessed within 48 hours, which may not capture early deaths, the authors note.
“The morbidity and mortality from ischemic strokes as a result of undertreatment of stroke prevention in patients with AFib continue to dwarf the bleeding related mortality among patients with AFib and on DOACs, and thus the number one priority is to treat nearly all patients with AFib with a DOAC,” Dr. Granger and Dr. Pokomey conclude. “The availability of reversal agents for DOACs should provide reassurance, with another tool in our armamentarium, to providers to prescribe OACs for stroke prevention.”
No funding/grant support was received to conduct the study. Coauthor Ramón Lecumberri has received personal fees from Boehringer Ingelheim and Bristol Myers Squibb outside the submitted work. All other authors report no relevant financial relationships. Dr. Granger has received research and consulting fees from Bristol Myers Squibb, Pfizer, Boehringer Ingelheim, Bayer, Janssen, Boston Scientific, Apple, AstraZeneca, Novartis, AbbVie, Biomed, CeleCor, GSK, Novartis, Medtronic, Merck, Novo Nordisk, Philips, Rho, and the U.S. Food and Drug Administration. Dr. Pokomey has received modest consulting support from Bristol Myers Squibb, Pfizer, Boston Scientific, Medtronic, Janssen, and Zoll; modest research support from Gilead, Boston Scientific, Bristol Myers Squibb, Pfizer, and Janssen; and significant research support from the FDA.
A version of this article first appeared on Medscape.com.
Watchdog group demands removal of FDA leaders after aducanumab approval
In a letter to the U.S. Department of Health and Human Services Secretary Xavier Becerra, Michael A. Carome, MD, director of Public Citizen’s Health Research Group, said: “The FDA’s decision to approve aducanumab for anyone with Alzheimer’s disease, regardless of severity, showed a stunning disregard for science, eviscerated the agency’s standards for approving new drugs, and ranks as one of the most irresponsible and egregious decisions in the history of the agency.”
Public Citizen urged Mr. Becerra to seek the resignations or the removal of the three FDA officials it said were most responsible for the approval – Acting FDA Commissioner Janet Woodcock, MD; Center for Drug Evaluation and Research (CDER) Director Patrizia Cavazzoni, MD; and CDER’s Office of Neuroscience Director Billy Dunn, MD.
“This decision is a disastrous blow to the agency’s credibility, public health, and the financial sustainability of the Medicare program,” writes Dr. Carome, noting that Biogen said it would charge $56,000 annually for the infusion.
Aaron Kesselheim, MD, one of three FDA Peripheral and Central Nervous System Drugs advisory committee members who resigned in the wake of the approval, agreed with Public Citizen that the agency’s credibility is suffering.
“The aducanumab decision is the worst example yet of the FDA’s movement away from its high standards,” Dr. Kesselheim, a professor of medicine at Harvard Medical School, Boston, and Harvard colleague Jerry Avorn, MD, wrote in the New York Times on June 15.
“As physicians, we know well that Alzheimer’s disease is a terrible condition,” they wrote. However, they added, “approving a drug that has such poor evidence that it works and causes such worrisome side effects is not the solution.”
In his resignation letter, Dr. Kesselheim said he had also been dismayed by the agency’s 2016 approval of eteplirsen (Exondys 51, Sarepta Therapeutics) for Duchenne muscular dystrophy. In both the eteplirsen and aducanumab approvals, the agency went against its advisers’ recommendations, Dr. Kesselheim said.
Advocates who backed approval decry cost
Aducanumab had a rocky road to approval but had unwavering backing from the Alzheimer’s Association and at least one other organization, UsAgainstAlzheimer’s.
The Alzheimer’s Association was particularly outspoken in its support and, in March, was accused of potential conflict of interest by Public Citizen and several neurologists because the association accepted at least $1.4 million from Biogen and its partner Eisai since fiscal year 2018.
The association applauded the FDA approval but, a few days later, expressed outrage over the $56,000-a-year price tag.
“This price is simply unacceptable,” the Alzheimer’s Association said in the statement. “For many, this price will pose an insurmountable barrier to access, it complicates and jeopardizes sustainable access to this treatment, and may further deepen issues of health equity,” the association said, adding, “We call on Biogen to change this price.”
UsAgainstAlzheimer’s also expressed concerns about access, even before it knew aducanumab’s price.
“Shockingly, Medicare does not reimburse patients for the expensive PET scans important to determine whether someone is appropriate for this drug,” noted George Vradenburg, chairman and cofounder of the group, in a June 7 statement. “We intend to work with Biogen and Medicare to make access to this drug affordable for every American who needs it,” Mr. Vradenburg said.
Dr. Carome said the advocates’ complaints were hard to fathom.
“This should not have come as a surprise to anyone,” Dr. Carome said, adding that “it’s essentially the ballpark figure the company threw out weeks ago.”
“Fifty-six-thousand-dollars is particularly egregiously overpriced for a drug that doesn’t work,” Dr. Carome said. “If the [Alzheimer’s Association] truly finds this objectionable, hopefully they’ll stop accepting money from Biogen and its partner Eisai,” he added.
“The Alzheimer’s Association is recognizing that the genie is out of the bottle and that they are going to have trouble reining in the inevitable run-away costs,” said Mike Greicius, MD, MPH, associate professor of neurology at Stanford University’s Wu Tsai Neurosciences Institute, Stanford, California.
“In addition to the eye-popping annual cost that Biogen has invented, I hope the Alzheimer’s Association is also concerned about the dangerously loose and broad FDA labeling which does not require screening for amyloid-positivity and does not restrict use to the milder forms of disease studied in the Phase 3 trials,” Dr. Greicius said.
Another advocacy group, Patients For Affordable Drugs, commended the Alzheimer’s Association. Its statement “was nothing short of courageous, especially in light of the Alzheimer’s Association’s reliance on funding from drug corporations, including Biogen,” said David Mitchell, a cancer patient and founder of Patients For Affordable Drugs, in a statement.
Mr. Mitchell said his members “stand with the Alzheimer’s Association in its denunciation of the price set by Biogen” and called for a new law that would allow Medicare to negotiate drug prices.
A version of this article first appeared on Medscape.com.
In a letter to the U.S. Department of Health and Human Services Secretary Xavier Becerra, Michael A. Carome, MD, director of Public Citizen’s Health Research Group, said: “The FDA’s decision to approve aducanumab for anyone with Alzheimer’s disease, regardless of severity, showed a stunning disregard for science, eviscerated the agency’s standards for approving new drugs, and ranks as one of the most irresponsible and egregious decisions in the history of the agency.”
Public Citizen urged Mr. Becerra to seek the resignations or the removal of the three FDA officials it said were most responsible for the approval – Acting FDA Commissioner Janet Woodcock, MD; Center for Drug Evaluation and Research (CDER) Director Patrizia Cavazzoni, MD; and CDER’s Office of Neuroscience Director Billy Dunn, MD.
“This decision is a disastrous blow to the agency’s credibility, public health, and the financial sustainability of the Medicare program,” writes Dr. Carome, noting that Biogen said it would charge $56,000 annually for the infusion.
Aaron Kesselheim, MD, one of three FDA Peripheral and Central Nervous System Drugs advisory committee members who resigned in the wake of the approval, agreed with Public Citizen that the agency’s credibility is suffering.
“The aducanumab decision is the worst example yet of the FDA’s movement away from its high standards,” Dr. Kesselheim, a professor of medicine at Harvard Medical School, Boston, and Harvard colleague Jerry Avorn, MD, wrote in the New York Times on June 15.
“As physicians, we know well that Alzheimer’s disease is a terrible condition,” they wrote. However, they added, “approving a drug that has such poor evidence that it works and causes such worrisome side effects is not the solution.”
In his resignation letter, Dr. Kesselheim said he had also been dismayed by the agency’s 2016 approval of eteplirsen (Exondys 51, Sarepta Therapeutics) for Duchenne muscular dystrophy. In both the eteplirsen and aducanumab approvals, the agency went against its advisers’ recommendations, Dr. Kesselheim said.
Advocates who backed approval decry cost
Aducanumab had a rocky road to approval but had unwavering backing from the Alzheimer’s Association and at least one other organization, UsAgainstAlzheimer’s.
The Alzheimer’s Association was particularly outspoken in its support and, in March, was accused of potential conflict of interest by Public Citizen and several neurologists because the association accepted at least $1.4 million from Biogen and its partner Eisai since fiscal year 2018.
The association applauded the FDA approval but, a few days later, expressed outrage over the $56,000-a-year price tag.
“This price is simply unacceptable,” the Alzheimer’s Association said in the statement. “For many, this price will pose an insurmountable barrier to access, it complicates and jeopardizes sustainable access to this treatment, and may further deepen issues of health equity,” the association said, adding, “We call on Biogen to change this price.”
UsAgainstAlzheimer’s also expressed concerns about access, even before it knew aducanumab’s price.
“Shockingly, Medicare does not reimburse patients for the expensive PET scans important to determine whether someone is appropriate for this drug,” noted George Vradenburg, chairman and cofounder of the group, in a June 7 statement. “We intend to work with Biogen and Medicare to make access to this drug affordable for every American who needs it,” Mr. Vradenburg said.
Dr. Carome said the advocates’ complaints were hard to fathom.
“This should not have come as a surprise to anyone,” Dr. Carome said, adding that “it’s essentially the ballpark figure the company threw out weeks ago.”
“Fifty-six-thousand-dollars is particularly egregiously overpriced for a drug that doesn’t work,” Dr. Carome said. “If the [Alzheimer’s Association] truly finds this objectionable, hopefully they’ll stop accepting money from Biogen and its partner Eisai,” he added.
“The Alzheimer’s Association is recognizing that the genie is out of the bottle and that they are going to have trouble reining in the inevitable run-away costs,” said Mike Greicius, MD, MPH, associate professor of neurology at Stanford University’s Wu Tsai Neurosciences Institute, Stanford, California.
“In addition to the eye-popping annual cost that Biogen has invented, I hope the Alzheimer’s Association is also concerned about the dangerously loose and broad FDA labeling which does not require screening for amyloid-positivity and does not restrict use to the milder forms of disease studied in the Phase 3 trials,” Dr. Greicius said.
Another advocacy group, Patients For Affordable Drugs, commended the Alzheimer’s Association. Its statement “was nothing short of courageous, especially in light of the Alzheimer’s Association’s reliance on funding from drug corporations, including Biogen,” said David Mitchell, a cancer patient and founder of Patients For Affordable Drugs, in a statement.
Mr. Mitchell said his members “stand with the Alzheimer’s Association in its denunciation of the price set by Biogen” and called for a new law that would allow Medicare to negotiate drug prices.
A version of this article first appeared on Medscape.com.
In a letter to the U.S. Department of Health and Human Services Secretary Xavier Becerra, Michael A. Carome, MD, director of Public Citizen’s Health Research Group, said: “The FDA’s decision to approve aducanumab for anyone with Alzheimer’s disease, regardless of severity, showed a stunning disregard for science, eviscerated the agency’s standards for approving new drugs, and ranks as one of the most irresponsible and egregious decisions in the history of the agency.”
Public Citizen urged Mr. Becerra to seek the resignations or the removal of the three FDA officials it said were most responsible for the approval – Acting FDA Commissioner Janet Woodcock, MD; Center for Drug Evaluation and Research (CDER) Director Patrizia Cavazzoni, MD; and CDER’s Office of Neuroscience Director Billy Dunn, MD.
“This decision is a disastrous blow to the agency’s credibility, public health, and the financial sustainability of the Medicare program,” writes Dr. Carome, noting that Biogen said it would charge $56,000 annually for the infusion.
Aaron Kesselheim, MD, one of three FDA Peripheral and Central Nervous System Drugs advisory committee members who resigned in the wake of the approval, agreed with Public Citizen that the agency’s credibility is suffering.
“The aducanumab decision is the worst example yet of the FDA’s movement away from its high standards,” Dr. Kesselheim, a professor of medicine at Harvard Medical School, Boston, and Harvard colleague Jerry Avorn, MD, wrote in the New York Times on June 15.
“As physicians, we know well that Alzheimer’s disease is a terrible condition,” they wrote. However, they added, “approving a drug that has such poor evidence that it works and causes such worrisome side effects is not the solution.”
In his resignation letter, Dr. Kesselheim said he had also been dismayed by the agency’s 2016 approval of eteplirsen (Exondys 51, Sarepta Therapeutics) for Duchenne muscular dystrophy. In both the eteplirsen and aducanumab approvals, the agency went against its advisers’ recommendations, Dr. Kesselheim said.
Advocates who backed approval decry cost
Aducanumab had a rocky road to approval but had unwavering backing from the Alzheimer’s Association and at least one other organization, UsAgainstAlzheimer’s.
The Alzheimer’s Association was particularly outspoken in its support and, in March, was accused of potential conflict of interest by Public Citizen and several neurologists because the association accepted at least $1.4 million from Biogen and its partner Eisai since fiscal year 2018.
The association applauded the FDA approval but, a few days later, expressed outrage over the $56,000-a-year price tag.
“This price is simply unacceptable,” the Alzheimer’s Association said in the statement. “For many, this price will pose an insurmountable barrier to access, it complicates and jeopardizes sustainable access to this treatment, and may further deepen issues of health equity,” the association said, adding, “We call on Biogen to change this price.”
UsAgainstAlzheimer’s also expressed concerns about access, even before it knew aducanumab’s price.
“Shockingly, Medicare does not reimburse patients for the expensive PET scans important to determine whether someone is appropriate for this drug,” noted George Vradenburg, chairman and cofounder of the group, in a June 7 statement. “We intend to work with Biogen and Medicare to make access to this drug affordable for every American who needs it,” Mr. Vradenburg said.
Dr. Carome said the advocates’ complaints were hard to fathom.
“This should not have come as a surprise to anyone,” Dr. Carome said, adding that “it’s essentially the ballpark figure the company threw out weeks ago.”
“Fifty-six-thousand-dollars is particularly egregiously overpriced for a drug that doesn’t work,” Dr. Carome said. “If the [Alzheimer’s Association] truly finds this objectionable, hopefully they’ll stop accepting money from Biogen and its partner Eisai,” he added.
“The Alzheimer’s Association is recognizing that the genie is out of the bottle and that they are going to have trouble reining in the inevitable run-away costs,” said Mike Greicius, MD, MPH, associate professor of neurology at Stanford University’s Wu Tsai Neurosciences Institute, Stanford, California.
“In addition to the eye-popping annual cost that Biogen has invented, I hope the Alzheimer’s Association is also concerned about the dangerously loose and broad FDA labeling which does not require screening for amyloid-positivity and does not restrict use to the milder forms of disease studied in the Phase 3 trials,” Dr. Greicius said.
Another advocacy group, Patients For Affordable Drugs, commended the Alzheimer’s Association. Its statement “was nothing short of courageous, especially in light of the Alzheimer’s Association’s reliance on funding from drug corporations, including Biogen,” said David Mitchell, a cancer patient and founder of Patients For Affordable Drugs, in a statement.
Mr. Mitchell said his members “stand with the Alzheimer’s Association in its denunciation of the price set by Biogen” and called for a new law that would allow Medicare to negotiate drug prices.
A version of this article first appeared on Medscape.com.