User login
Monoclonal Antibodies in MS
A 19-year-old man was diagnosed with relapsing multiple sclerosis (MS) at age 7 and is currently being treated with an infusible monoclonal antibody (mAb) therapy. Early in the day, he receives an infusion at an outpatient clinic. That night, he begins to experience numbness and tingling in his right upper extremity, which prompts a visit to an urgent care clinic. There, the clinician administers IV fluids to the patient. After his symptoms improve, the patient is discharged home.
The next morning, he has a new onset of left-side shoulder and neck pain with a pulsating headache. The patient reports his symptoms to his primary care provider (PCP), who instructs him to visit the emergency department (ED) for evaluation and treatment of a possible infection.
EXAMINATION
The patient arrives at the ED with a 102.4°F fever, vomiting, cough, mild congestion, diaphoresis, generalized myalgias, and chills. He also reports depression and anxiety, saying that for the past 7 days, “I haven’t felt like my normal self.”
Medical history includes moderate persistent asthma that is not well controlled, status asthmaticus, and use of an electronic vaporizing device for inhaling nicotine and marijuana/tetrahydrocannabinol (THC). Besides mAb infusions, his medications include hydrocodone/acetaminophen, prochlorperazine, gabapentin, hydroxyzine, trazodone, albuterol, and montelukast.
Examination reveals vital signs within normal limits. Lab work confirms elevated white blood cell count and absolute neutrophil count. Chest x-ray shows diffuse bilateral interstitial and patchy airspace opacities. He is diagnosed with bilateral pneumonia and is admitted and started on an IV antibiotic.
Within 24 hours, a new chest x-ray shows worsening symptoms. CT of the chest with contrast reveals diffuse bilateral ground-glass and airspace opacities suggestive of acute respiratory distress syndrome; bilateral thickening of the pulmonary interstitium; trace bilateral pleural effusions; increased caliber of the main pulmonary artery; and mediastinal and right hilar lymphadenopathy.
Subsequently, the patient developed sepsis and went into acute hypoxemic respiratory failure. He is transferred to the ICU, and pulmonology is consulted. A bronchoscopy with bronchoalveolar lavage (BAL) reveals neutrophil predominance; fungal, bacterial, and viral cultures are negative. The patient is started on broad-spectrum IV antibiotics and high-dose IV steroids. After 4 days, he begins to improve and is transferred out of the ICU. He is discharged with oral steroids and antibiotics.
Continue to: DISCUSSION
DISCUSSION
Fortunately, the PCP and the ED provider identified risk factors that contributed to the patient’s pneumonia and its subsequent worsening to sepsis and acute hypoxemic respiratory failure. The immunosuppressive/immunomodulatory effect of mAb therapy increased the patient’s risk for infection and the severity of infection, which is why vigilant safety monitoring and surveillance is essential with mAb treatment.1 Bloodwork should be performed at least every 6 months and include a complete blood count, complete metabolic panel with differential, and JC virus antibody test. Additionally, urinalysis should be performed prior to every mAb infusion. All testing recommended in the package insert for the patient’s prescribed therapy should be performed.
The patient’s history of asthma and his chronic vaping predisposed him to respiratory infections. In mice studies, exposure to e-cigarette vapor has been shown to be cytotoxic to airway cells and to decrease macrophage and neutrophil antimicrobial function.2 Exposure also alters immunomodulating cytokines in the airway, increases inflammatory markers seen in BAL and serum samples, and increases the virulence of Staphylococcus aureus
TREATMENT AND PATIENT EDUCATION
The PCP’s treatment plan included patient education about the importance of infection control measures when receiving a mAb; this includes practicing good hand and environmental hygiene, maintaining vaccinations, avoiding or reducing exposure to individuals who have infections or colds, avoiding large crowds (especially during flu season), and following recommendations for nutrition and hydration. The PCP also discussed how to recognize the early signs and symptoms of an infection—and the need for vigilant safety monitoring. The PCP described available options for smoking cessation, including nicotine replacement products, prescription non-nicotine medications, behavioral therapy, and/or counseling (individual, group or telephone) and discussed the risks associated with consuming nicotine and/or marijuana/THC and using electronic vaporizing devices.
The PCP emphasized the importance of completing the entire course of the oral antibiotics prescribed at discharge. The patient and the PCP agreed to the following plan of care: appointments with a pulmonologist and a neurologist within the next 2 weeks, and follow-up visits with the
1. Celius EG. Infections in patients with multiple sclerosis: implications for disease-modifying therapy. Acta Neurol Scand. 2017;136(suppl 201):34-36.
2. Hwang JH, Lyes M, Sladewski K, et al. Electronic cigarette inhalation alters innate immunity and airway cytokines while increasing the virulence of colonizing bacteria. J Mol Med (Berl). 2016;94(6):667-679.
A 19-year-old man was diagnosed with relapsing multiple sclerosis (MS) at age 7 and is currently being treated with an infusible monoclonal antibody (mAb) therapy. Early in the day, he receives an infusion at an outpatient clinic. That night, he begins to experience numbness and tingling in his right upper extremity, which prompts a visit to an urgent care clinic. There, the clinician administers IV fluids to the patient. After his symptoms improve, the patient is discharged home.
The next morning, he has a new onset of left-side shoulder and neck pain with a pulsating headache. The patient reports his symptoms to his primary care provider (PCP), who instructs him to visit the emergency department (ED) for evaluation and treatment of a possible infection.
EXAMINATION
The patient arrives at the ED with a 102.4°F fever, vomiting, cough, mild congestion, diaphoresis, generalized myalgias, and chills. He also reports depression and anxiety, saying that for the past 7 days, “I haven’t felt like my normal self.”
Medical history includes moderate persistent asthma that is not well controlled, status asthmaticus, and use of an electronic vaporizing device for inhaling nicotine and marijuana/tetrahydrocannabinol (THC). Besides mAb infusions, his medications include hydrocodone/acetaminophen, prochlorperazine, gabapentin, hydroxyzine, trazodone, albuterol, and montelukast.
Examination reveals vital signs within normal limits. Lab work confirms elevated white blood cell count and absolute neutrophil count. Chest x-ray shows diffuse bilateral interstitial and patchy airspace opacities. He is diagnosed with bilateral pneumonia and is admitted and started on an IV antibiotic.
Within 24 hours, a new chest x-ray shows worsening symptoms. CT of the chest with contrast reveals diffuse bilateral ground-glass and airspace opacities suggestive of acute respiratory distress syndrome; bilateral thickening of the pulmonary interstitium; trace bilateral pleural effusions; increased caliber of the main pulmonary artery; and mediastinal and right hilar lymphadenopathy.
Subsequently, the patient developed sepsis and went into acute hypoxemic respiratory failure. He is transferred to the ICU, and pulmonology is consulted. A bronchoscopy with bronchoalveolar lavage (BAL) reveals neutrophil predominance; fungal, bacterial, and viral cultures are negative. The patient is started on broad-spectrum IV antibiotics and high-dose IV steroids. After 4 days, he begins to improve and is transferred out of the ICU. He is discharged with oral steroids and antibiotics.
Continue to: DISCUSSION
DISCUSSION
Fortunately, the PCP and the ED provider identified risk factors that contributed to the patient’s pneumonia and its subsequent worsening to sepsis and acute hypoxemic respiratory failure. The immunosuppressive/immunomodulatory effect of mAb therapy increased the patient’s risk for infection and the severity of infection, which is why vigilant safety monitoring and surveillance is essential with mAb treatment.1 Bloodwork should be performed at least every 6 months and include a complete blood count, complete metabolic panel with differential, and JC virus antibody test. Additionally, urinalysis should be performed prior to every mAb infusion. All testing recommended in the package insert for the patient’s prescribed therapy should be performed.
The patient’s history of asthma and his chronic vaping predisposed him to respiratory infections. In mice studies, exposure to e-cigarette vapor has been shown to be cytotoxic to airway cells and to decrease macrophage and neutrophil antimicrobial function.2 Exposure also alters immunomodulating cytokines in the airway, increases inflammatory markers seen in BAL and serum samples, and increases the virulence of Staphylococcus aureus
TREATMENT AND PATIENT EDUCATION
The PCP’s treatment plan included patient education about the importance of infection control measures when receiving a mAb; this includes practicing good hand and environmental hygiene, maintaining vaccinations, avoiding or reducing exposure to individuals who have infections or colds, avoiding large crowds (especially during flu season), and following recommendations for nutrition and hydration. The PCP also discussed how to recognize the early signs and symptoms of an infection—and the need for vigilant safety monitoring. The PCP described available options for smoking cessation, including nicotine replacement products, prescription non-nicotine medications, behavioral therapy, and/or counseling (individual, group or telephone) and discussed the risks associated with consuming nicotine and/or marijuana/THC and using electronic vaporizing devices.
The PCP emphasized the importance of completing the entire course of the oral antibiotics prescribed at discharge. The patient and the PCP agreed to the following plan of care: appointments with a pulmonologist and a neurologist within the next 2 weeks, and follow-up visits with the
A 19-year-old man was diagnosed with relapsing multiple sclerosis (MS) at age 7 and is currently being treated with an infusible monoclonal antibody (mAb) therapy. Early in the day, he receives an infusion at an outpatient clinic. That night, he begins to experience numbness and tingling in his right upper extremity, which prompts a visit to an urgent care clinic. There, the clinician administers IV fluids to the patient. After his symptoms improve, the patient is discharged home.
The next morning, he has a new onset of left-side shoulder and neck pain with a pulsating headache. The patient reports his symptoms to his primary care provider (PCP), who instructs him to visit the emergency department (ED) for evaluation and treatment of a possible infection.
EXAMINATION
The patient arrives at the ED with a 102.4°F fever, vomiting, cough, mild congestion, diaphoresis, generalized myalgias, and chills. He also reports depression and anxiety, saying that for the past 7 days, “I haven’t felt like my normal self.”
Medical history includes moderate persistent asthma that is not well controlled, status asthmaticus, and use of an electronic vaporizing device for inhaling nicotine and marijuana/tetrahydrocannabinol (THC). Besides mAb infusions, his medications include hydrocodone/acetaminophen, prochlorperazine, gabapentin, hydroxyzine, trazodone, albuterol, and montelukast.
Examination reveals vital signs within normal limits. Lab work confirms elevated white blood cell count and absolute neutrophil count. Chest x-ray shows diffuse bilateral interstitial and patchy airspace opacities. He is diagnosed with bilateral pneumonia and is admitted and started on an IV antibiotic.
Within 24 hours, a new chest x-ray shows worsening symptoms. CT of the chest with contrast reveals diffuse bilateral ground-glass and airspace opacities suggestive of acute respiratory distress syndrome; bilateral thickening of the pulmonary interstitium; trace bilateral pleural effusions; increased caliber of the main pulmonary artery; and mediastinal and right hilar lymphadenopathy.
Subsequently, the patient developed sepsis and went into acute hypoxemic respiratory failure. He is transferred to the ICU, and pulmonology is consulted. A bronchoscopy with bronchoalveolar lavage (BAL) reveals neutrophil predominance; fungal, bacterial, and viral cultures are negative. The patient is started on broad-spectrum IV antibiotics and high-dose IV steroids. After 4 days, he begins to improve and is transferred out of the ICU. He is discharged with oral steroids and antibiotics.
Continue to: DISCUSSION
DISCUSSION
Fortunately, the PCP and the ED provider identified risk factors that contributed to the patient’s pneumonia and its subsequent worsening to sepsis and acute hypoxemic respiratory failure. The immunosuppressive/immunomodulatory effect of mAb therapy increased the patient’s risk for infection and the severity of infection, which is why vigilant safety monitoring and surveillance is essential with mAb treatment.1 Bloodwork should be performed at least every 6 months and include a complete blood count, complete metabolic panel with differential, and JC virus antibody test. Additionally, urinalysis should be performed prior to every mAb infusion. All testing recommended in the package insert for the patient’s prescribed therapy should be performed.
The patient’s history of asthma and his chronic vaping predisposed him to respiratory infections. In mice studies, exposure to e-cigarette vapor has been shown to be cytotoxic to airway cells and to decrease macrophage and neutrophil antimicrobial function.2 Exposure also alters immunomodulating cytokines in the airway, increases inflammatory markers seen in BAL and serum samples, and increases the virulence of Staphylococcus aureus
TREATMENT AND PATIENT EDUCATION
The PCP’s treatment plan included patient education about the importance of infection control measures when receiving a mAb; this includes practicing good hand and environmental hygiene, maintaining vaccinations, avoiding or reducing exposure to individuals who have infections or colds, avoiding large crowds (especially during flu season), and following recommendations for nutrition and hydration. The PCP also discussed how to recognize the early signs and symptoms of an infection—and the need for vigilant safety monitoring. The PCP described available options for smoking cessation, including nicotine replacement products, prescription non-nicotine medications, behavioral therapy, and/or counseling (individual, group or telephone) and discussed the risks associated with consuming nicotine and/or marijuana/THC and using electronic vaporizing devices.
The PCP emphasized the importance of completing the entire course of the oral antibiotics prescribed at discharge. The patient and the PCP agreed to the following plan of care: appointments with a pulmonologist and a neurologist within the next 2 weeks, and follow-up visits with the
1. Celius EG. Infections in patients with multiple sclerosis: implications for disease-modifying therapy. Acta Neurol Scand. 2017;136(suppl 201):34-36.
2. Hwang JH, Lyes M, Sladewski K, et al. Electronic cigarette inhalation alters innate immunity and airway cytokines while increasing the virulence of colonizing bacteria. J Mol Med (Berl). 2016;94(6):667-679.
1. Celius EG. Infections in patients with multiple sclerosis: implications for disease-modifying therapy. Acta Neurol Scand. 2017;136(suppl 201):34-36.
2. Hwang JH, Lyes M, Sladewski K, et al. Electronic cigarette inhalation alters innate immunity and airway cytokines while increasing the virulence of colonizing bacteria. J Mol Med (Berl). 2016;94(6):667-679.
Reappraising standard treatment of comorbid insomnia/depression
COPENHAGEN – The traditional treatment paradigm for patients with comorbid depression and insomnia has been to focus on the depression in expectation that the sleep problems will fade away with the depressive symptoms.
Big mistake, Kerstin Blom, PhD, said during the annual congress of the European College of Neuropsychopharmacology.
That treatment strategy is insufficient, because untreated insomnia seldom improves. It hinders recovery from depression, increases the risk of new depressive episodes, and causes continued suffering because of poor sleep, asserted Dr. Blom, a clinical psychologist and researcher at the Internet Psychiatry Clinic at the Karolinska Institute in Stockholm.
She presented highlights of a series of three randomized, controlled trials for which she was first author. The take-home message: Insomnia with comorbid depression is not merely a symptom of depression; it requires specific treatment.
“Insomnia needs to be treated according to guidelines – that is, with cognitive-behavioral therapy – when it’s comorbid with depression,” she declared. “Insomnia therapy also treats comorbid depression, but it’s not so much the other way around. There are some effects on insomnia when you treat depression, but they’re not very large.”
The first study in her series included 43 adults with psychiatrist-diagnosed comorbid insomnia and major depression who were randomized to an 8-week course of psychologist-guided, Internet-delivered cognitive-behavioral therapy (ICBT) for one disorder or the other. At 6- and 12-month follow-up, patients who received ICBT for insomnia had significantly greater improvement in their insomnia as measured by the self-rated Insomnia Severity Index than did those who got ICBT for depression, while both forms of treatment were similarly effective in reducing depression severity as reflected in Montgomery-Åsberg Depression Rating Scale (MADRS) scores (Sleep. 2015 Feb 1;38[2]:267-77).
At 3-year follow-up, the beneficial impact of ICBT for insomnia remained strong, with recipients reporting less need for additional sleep treatment and less use of sleep medication than did the patients who got ICBT for depression. Both groups were left with mild depression, pointing to the need to develop a combined form of CBT that would simultaneously address both disorders in patients with comorbid depression and insomnia (Sleep. 2017 Aug 1;40[8]. doi. 10.1093/sleep/zsx108).
The Swedish investigators went on to create a 9-week course of psychologist-guided combination ICBT for both insomnia and depression. Then they randomized 126 dual-diagnosis patients to that treatment program or to therapist-guided ICBT for depression plus a placebo sleep intervention, which included education about sleep hygiene, stress management, and use of a sleep diary. At 6 months of follow-up, the dual-target ICBT group had a significantly greater reduction in Insomnia Severity Index scores than those who got ICBT for depression plus placebo. No between-group difference were found in the reduction in MADRS scores.
Dr. Blom observed. Follow-up out to 36 months is ongoing.
The third study included 148 nondepressed adults with insomnia who were randomized to the 8-week ICBT insomnia intervention or an active control treatment, which again included patient education, stress management, and a sleep diary. At 6 months, the active CBT-insomnia group had significantly lower Insomnia Severity Index scores than controls. However, at 12 and 36 months, the control group caught up, and there was no longer a between-group difference, with 74% of participants no longer meeting diagnostic criteria for insomnia at 36 months. The explanation for the catch-up? The control group used significantly more hypnotic sleep medications and more frequently sought additional insomnia treatments, including yoga and mindfulness, outside of the study setting during follow-up (Sleep. 2016 Jun 1;39[6]:1267-74).
It was Dr. Blom’s intent to also use this randomized, controlled trials to test the hypothesis that improving insomnia in nondepressed patients prevents future episodes of depression. She was thwarted in this attempt.
“After they got our low-intensity control version of a sleep intervention, they went out and got more treatment and that seems to have helped them, which is great,” Dr. Blom said. “But it sort of ruined our prediction study.”
However, in a post hoc analysis, study participants who were poor sleepers at 12 months had significantly more depressive symptoms at 36 months than did those with improved sleep at 12 months. The effect size was quite large, with a between-group 5.5-point difference in MADRS scores at 36 months in a study population that was nondepressed at baseline.
“So improved sleep may prevent depression long term,” Dr. Blom said. “The jury is still out on that one.”
She reported having no financial conflicts regarding her studies, which were supported by government research funding.
COPENHAGEN – The traditional treatment paradigm for patients with comorbid depression and insomnia has been to focus on the depression in expectation that the sleep problems will fade away with the depressive symptoms.
Big mistake, Kerstin Blom, PhD, said during the annual congress of the European College of Neuropsychopharmacology.
That treatment strategy is insufficient, because untreated insomnia seldom improves. It hinders recovery from depression, increases the risk of new depressive episodes, and causes continued suffering because of poor sleep, asserted Dr. Blom, a clinical psychologist and researcher at the Internet Psychiatry Clinic at the Karolinska Institute in Stockholm.
She presented highlights of a series of three randomized, controlled trials for which she was first author. The take-home message: Insomnia with comorbid depression is not merely a symptom of depression; it requires specific treatment.
“Insomnia needs to be treated according to guidelines – that is, with cognitive-behavioral therapy – when it’s comorbid with depression,” she declared. “Insomnia therapy also treats comorbid depression, but it’s not so much the other way around. There are some effects on insomnia when you treat depression, but they’re not very large.”
The first study in her series included 43 adults with psychiatrist-diagnosed comorbid insomnia and major depression who were randomized to an 8-week course of psychologist-guided, Internet-delivered cognitive-behavioral therapy (ICBT) for one disorder or the other. At 6- and 12-month follow-up, patients who received ICBT for insomnia had significantly greater improvement in their insomnia as measured by the self-rated Insomnia Severity Index than did those who got ICBT for depression, while both forms of treatment were similarly effective in reducing depression severity as reflected in Montgomery-Åsberg Depression Rating Scale (MADRS) scores (Sleep. 2015 Feb 1;38[2]:267-77).
At 3-year follow-up, the beneficial impact of ICBT for insomnia remained strong, with recipients reporting less need for additional sleep treatment and less use of sleep medication than did the patients who got ICBT for depression. Both groups were left with mild depression, pointing to the need to develop a combined form of CBT that would simultaneously address both disorders in patients with comorbid depression and insomnia (Sleep. 2017 Aug 1;40[8]. doi. 10.1093/sleep/zsx108).
The Swedish investigators went on to create a 9-week course of psychologist-guided combination ICBT for both insomnia and depression. Then they randomized 126 dual-diagnosis patients to that treatment program or to therapist-guided ICBT for depression plus a placebo sleep intervention, which included education about sleep hygiene, stress management, and use of a sleep diary. At 6 months of follow-up, the dual-target ICBT group had a significantly greater reduction in Insomnia Severity Index scores than those who got ICBT for depression plus placebo. No between-group difference were found in the reduction in MADRS scores.
Dr. Blom observed. Follow-up out to 36 months is ongoing.
The third study included 148 nondepressed adults with insomnia who were randomized to the 8-week ICBT insomnia intervention or an active control treatment, which again included patient education, stress management, and a sleep diary. At 6 months, the active CBT-insomnia group had significantly lower Insomnia Severity Index scores than controls. However, at 12 and 36 months, the control group caught up, and there was no longer a between-group difference, with 74% of participants no longer meeting diagnostic criteria for insomnia at 36 months. The explanation for the catch-up? The control group used significantly more hypnotic sleep medications and more frequently sought additional insomnia treatments, including yoga and mindfulness, outside of the study setting during follow-up (Sleep. 2016 Jun 1;39[6]:1267-74).
It was Dr. Blom’s intent to also use this randomized, controlled trials to test the hypothesis that improving insomnia in nondepressed patients prevents future episodes of depression. She was thwarted in this attempt.
“After they got our low-intensity control version of a sleep intervention, they went out and got more treatment and that seems to have helped them, which is great,” Dr. Blom said. “But it sort of ruined our prediction study.”
However, in a post hoc analysis, study participants who were poor sleepers at 12 months had significantly more depressive symptoms at 36 months than did those with improved sleep at 12 months. The effect size was quite large, with a between-group 5.5-point difference in MADRS scores at 36 months in a study population that was nondepressed at baseline.
“So improved sleep may prevent depression long term,” Dr. Blom said. “The jury is still out on that one.”
She reported having no financial conflicts regarding her studies, which were supported by government research funding.
COPENHAGEN – The traditional treatment paradigm for patients with comorbid depression and insomnia has been to focus on the depression in expectation that the sleep problems will fade away with the depressive symptoms.
Big mistake, Kerstin Blom, PhD, said during the annual congress of the European College of Neuropsychopharmacology.
That treatment strategy is insufficient, because untreated insomnia seldom improves. It hinders recovery from depression, increases the risk of new depressive episodes, and causes continued suffering because of poor sleep, asserted Dr. Blom, a clinical psychologist and researcher at the Internet Psychiatry Clinic at the Karolinska Institute in Stockholm.
She presented highlights of a series of three randomized, controlled trials for which she was first author. The take-home message: Insomnia with comorbid depression is not merely a symptom of depression; it requires specific treatment.
“Insomnia needs to be treated according to guidelines – that is, with cognitive-behavioral therapy – when it’s comorbid with depression,” she declared. “Insomnia therapy also treats comorbid depression, but it’s not so much the other way around. There are some effects on insomnia when you treat depression, but they’re not very large.”
The first study in her series included 43 adults with psychiatrist-diagnosed comorbid insomnia and major depression who were randomized to an 8-week course of psychologist-guided, Internet-delivered cognitive-behavioral therapy (ICBT) for one disorder or the other. At 6- and 12-month follow-up, patients who received ICBT for insomnia had significantly greater improvement in their insomnia as measured by the self-rated Insomnia Severity Index than did those who got ICBT for depression, while both forms of treatment were similarly effective in reducing depression severity as reflected in Montgomery-Åsberg Depression Rating Scale (MADRS) scores (Sleep. 2015 Feb 1;38[2]:267-77).
At 3-year follow-up, the beneficial impact of ICBT for insomnia remained strong, with recipients reporting less need for additional sleep treatment and less use of sleep medication than did the patients who got ICBT for depression. Both groups were left with mild depression, pointing to the need to develop a combined form of CBT that would simultaneously address both disorders in patients with comorbid depression and insomnia (Sleep. 2017 Aug 1;40[8]. doi. 10.1093/sleep/zsx108).
The Swedish investigators went on to create a 9-week course of psychologist-guided combination ICBT for both insomnia and depression. Then they randomized 126 dual-diagnosis patients to that treatment program or to therapist-guided ICBT for depression plus a placebo sleep intervention, which included education about sleep hygiene, stress management, and use of a sleep diary. At 6 months of follow-up, the dual-target ICBT group had a significantly greater reduction in Insomnia Severity Index scores than those who got ICBT for depression plus placebo. No between-group difference were found in the reduction in MADRS scores.
Dr. Blom observed. Follow-up out to 36 months is ongoing.
The third study included 148 nondepressed adults with insomnia who were randomized to the 8-week ICBT insomnia intervention or an active control treatment, which again included patient education, stress management, and a sleep diary. At 6 months, the active CBT-insomnia group had significantly lower Insomnia Severity Index scores than controls. However, at 12 and 36 months, the control group caught up, and there was no longer a between-group difference, with 74% of participants no longer meeting diagnostic criteria for insomnia at 36 months. The explanation for the catch-up? The control group used significantly more hypnotic sleep medications and more frequently sought additional insomnia treatments, including yoga and mindfulness, outside of the study setting during follow-up (Sleep. 2016 Jun 1;39[6]:1267-74).
It was Dr. Blom’s intent to also use this randomized, controlled trials to test the hypothesis that improving insomnia in nondepressed patients prevents future episodes of depression. She was thwarted in this attempt.
“After they got our low-intensity control version of a sleep intervention, they went out and got more treatment and that seems to have helped them, which is great,” Dr. Blom said. “But it sort of ruined our prediction study.”
However, in a post hoc analysis, study participants who were poor sleepers at 12 months had significantly more depressive symptoms at 36 months than did those with improved sleep at 12 months. The effect size was quite large, with a between-group 5.5-point difference in MADRS scores at 36 months in a study population that was nondepressed at baseline.
“So improved sleep may prevent depression long term,” Dr. Blom said. “The jury is still out on that one.”
She reported having no financial conflicts regarding her studies, which were supported by government research funding.
REPORTING FROM ECNP 2019
Fentanyl-related deaths show strong regional pattern
Fentanyl was involved in more overdose deaths than any other drug in 2017, and the death rate in New England was 15 times higher than in regions of the Midwest and West, according to the National Center for Health Statistics.
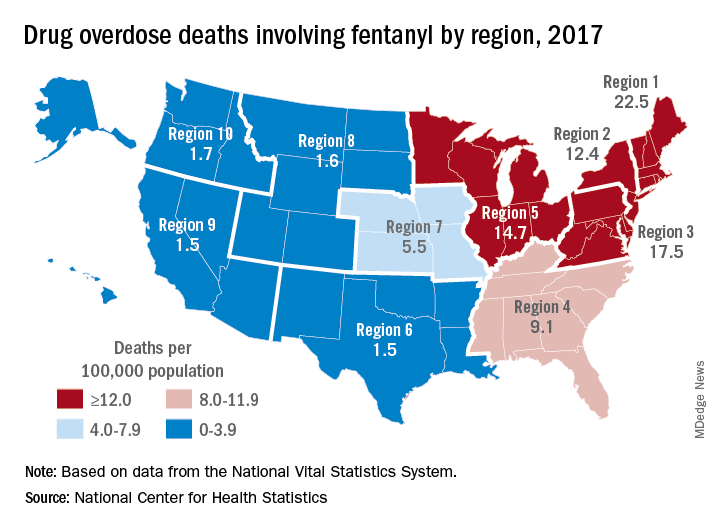
Nationally, fentanyl was involved in 39% of all drug overdose deaths and had an age-adjusted death rate of 8.7/100,000 standard population in 2017. In 2016, when fentanyl also was the most involved drug in the United States, the corresponding figures were 29% and 5.9/100,000, the agency said in a recent report.
Fentanyl was the most involved drug in overdose deaths for 6 of the country’s 10 public health regions in 2017, with a clear pattern of decreasing use from east to west. The highest death rate (22.5/100,000) occurred in Region 1 (New England) and the lowest rates (1.5/100,000) came in Region 6 (Arkansas, Louisiana, New Mexico, Oklahoma, and Texas) and Region 9 (Arizona, California, Hawaii, and Nevada), the researchers said.
A somewhat similar pattern was seen for heroin, which was second nationally on the list of drugs most frequently involved in overdose deaths (23%), except that New England was somewhat below three other regions in the East and upper Midwest. The highest heroin death rate (8.6/100,000) was seen in Region 2 (New Jersey and New York) and the lowest (2.2) occurred in Region 9, they said, based on data from the National Vital Statistics System’s mortality files.
The fentanyl pattern was even more closely repeated with cocaine, third in involvement nationally at 21% of overdose deaths in 2017. The high in overdose deaths (9.5/100,000) came in Region 1 again, and the low in Region 9 (1.3), along with Region 7 (Iowa, Kansas, Missouri, and Nebraska) and Region 10 (Alaska, Idaho, Oregon, and Washington), the report showed.
The regional pattern of overdose deaths for methamphetamine, which was fourth nationally in involvement (13.3%), basically reversed the other three drugs: highest in the West and lowest in the Northeast. Region 9 had the highest death rate (5.2/100,000) and Region 2 the lowest (0.4), with Region 1 just ahead at 0.6.
Fentanyl was involved in more overdose deaths than any other drug in 2017, and the death rate in New England was 15 times higher than in regions of the Midwest and West, according to the National Center for Health Statistics.

Nationally, fentanyl was involved in 39% of all drug overdose deaths and had an age-adjusted death rate of 8.7/100,000 standard population in 2017. In 2016, when fentanyl also was the most involved drug in the United States, the corresponding figures were 29% and 5.9/100,000, the agency said in a recent report.
Fentanyl was the most involved drug in overdose deaths for 6 of the country’s 10 public health regions in 2017, with a clear pattern of decreasing use from east to west. The highest death rate (22.5/100,000) occurred in Region 1 (New England) and the lowest rates (1.5/100,000) came in Region 6 (Arkansas, Louisiana, New Mexico, Oklahoma, and Texas) and Region 9 (Arizona, California, Hawaii, and Nevada), the researchers said.
A somewhat similar pattern was seen for heroin, which was second nationally on the list of drugs most frequently involved in overdose deaths (23%), except that New England was somewhat below three other regions in the East and upper Midwest. The highest heroin death rate (8.6/100,000) was seen in Region 2 (New Jersey and New York) and the lowest (2.2) occurred in Region 9, they said, based on data from the National Vital Statistics System’s mortality files.
The fentanyl pattern was even more closely repeated with cocaine, third in involvement nationally at 21% of overdose deaths in 2017. The high in overdose deaths (9.5/100,000) came in Region 1 again, and the low in Region 9 (1.3), along with Region 7 (Iowa, Kansas, Missouri, and Nebraska) and Region 10 (Alaska, Idaho, Oregon, and Washington), the report showed.
The regional pattern of overdose deaths for methamphetamine, which was fourth nationally in involvement (13.3%), basically reversed the other three drugs: highest in the West and lowest in the Northeast. Region 9 had the highest death rate (5.2/100,000) and Region 2 the lowest (0.4), with Region 1 just ahead at 0.6.
Fentanyl was involved in more overdose deaths than any other drug in 2017, and the death rate in New England was 15 times higher than in regions of the Midwest and West, according to the National Center for Health Statistics.

Nationally, fentanyl was involved in 39% of all drug overdose deaths and had an age-adjusted death rate of 8.7/100,000 standard population in 2017. In 2016, when fentanyl also was the most involved drug in the United States, the corresponding figures were 29% and 5.9/100,000, the agency said in a recent report.
Fentanyl was the most involved drug in overdose deaths for 6 of the country’s 10 public health regions in 2017, with a clear pattern of decreasing use from east to west. The highest death rate (22.5/100,000) occurred in Region 1 (New England) and the lowest rates (1.5/100,000) came in Region 6 (Arkansas, Louisiana, New Mexico, Oklahoma, and Texas) and Region 9 (Arizona, California, Hawaii, and Nevada), the researchers said.
A somewhat similar pattern was seen for heroin, which was second nationally on the list of drugs most frequently involved in overdose deaths (23%), except that New England was somewhat below three other regions in the East and upper Midwest. The highest heroin death rate (8.6/100,000) was seen in Region 2 (New Jersey and New York) and the lowest (2.2) occurred in Region 9, they said, based on data from the National Vital Statistics System’s mortality files.
The fentanyl pattern was even more closely repeated with cocaine, third in involvement nationally at 21% of overdose deaths in 2017. The high in overdose deaths (9.5/100,000) came in Region 1 again, and the low in Region 9 (1.3), along with Region 7 (Iowa, Kansas, Missouri, and Nebraska) and Region 10 (Alaska, Idaho, Oregon, and Washington), the report showed.
The regional pattern of overdose deaths for methamphetamine, which was fourth nationally in involvement (13.3%), basically reversed the other three drugs: highest in the West and lowest in the Northeast. Region 9 had the highest death rate (5.2/100,000) and Region 2 the lowest (0.4), with Region 1 just ahead at 0.6.
MS-related disability may be decreasing
STOCKHOLM – , according to an overview provided at the annual congress of the European Committee for Treatment and Research in Multiple Sclerosis. Data consistently indicate that the time that elapses before a patient requires a cane for ambulation has increased, and survival has likewise improved. “Some of the improvement can be attributed confidently to treatment effect,” said Ilya Kister, MD, associate professor of neurology at NYU Langone Health in New York. “We hope to see an even greater change with newer therapies.”
At the same time, neurologists appear to be diagnosing more cases of MS than they previously did, said Dr. Kister, which suggests that neurologists probably are diagnosing milder cases. The overall societal burden of MS remains high.
The relative prevalence of mild disability has increased
About 25 years have elapsed since the first disease-modifying treatment (DMT) for MS became available, and treatment has become widespread during that time. Dr. Kister and colleagues sought to determine whether the current clinical population of patients with MS, who for the most part receive DMTs, has less disability than do untreated patients or patients from natural history studies do. They identified the MS Severity Score (MSSS) as a measure with which to compare populations. The MSSS assigns a patient a ranking according to his or her level of disability, using a reference population of patients with the same disease duration for comparison. “MSSS can be conceptualized as rate of disability accumulation,” said Dr. Kister. “Lower MSSS corresponds to relatively slower disability accumulation, and higher MSSS to higher disability accumulation.”
The MSSS was developed using the Expanded Disability Status Scale (EDSS) score as a measure of disability. Because many neurologists do not routinely obtain EDSS scores for their patients, Dr. Kister and colleagues used the Patient-Determined Disease Steps (PDDS) to measure disability. As its name implies, the PDDS is a patient-reported outcome measure that mainly measures ambulation. It correlates strongly with EDSS, said Dr. Kister. He and colleagues used the PDDS to develop a reference table of MS disability, which they called the Patient-Derived MSSS.
The investigators examined a large sample of patients at NYU MS Center and Barnabas MS Center in Livingston, N.J. They grouped patients into sextiles according to their Patient-Derived MSSS. Dr. Kister and colleagues found that, rather than arriving at sextiles that contained equal numbers of patients, as would be expected if disability were distributed as in the reference population, they had significantly more patients in the two lowest sextiles and significantly fewer patients in the two highest sextiles. “This [result] suggests that the disability curve has indeed shifted toward the more benign end of the spectrum in the contemporary clinic population,” said Dr. Kister.
Other researchers have observed a similar phenomenon. George et al. published the results of a large, international collaboration in Neurology Genetics in 2016. After examining more than 7,000 patients, the investigators noted a similar overrepresentation of patients with milder severity scores and underrepresentation of patients with higher severity scores. These results support the hypothesis of a shift toward milder disability, said Dr. Kister.
Trend toward milder disability
The investigators next examined whether the rate of accumulation of disability among patients with MS had changed from year to year since DMTs were introduced. They conducted a univariate analysis of MSSS for 6,238 patients who were enrolled in the N.Y. State MS Consortium during 1996-2007. They found that patients who were enrolled in more recent years had significantly lower MSSS than patients who were enrolled in earlier years, regardless of disease duration. When Dr. Kister and colleagues replicated their analysis using EDSS, they found significantly lower levels of disability for patients enrolled in more recent years, except for patients with disease duration of 26-30 years. A multivariate analysis showed that the median MSSS of enrollees into the N.Y. State MS Consortium decreased from 5.04 in 1996 to 3.78 in 2006.
In a subsequent study, Dr. Kister and colleagues examined the age at which patients in the MSBase registry reached various disability milestones (e.g., EDSS of 6, which indicates the need of a cane to walk outdoors), according to their year of enrollment in the registry. They found a significant increase in age at milestone achievement with each subsequent calendar year. For example, for every consecutive year of enrollment, the age at which patients attained an EDSS of 6 increased by 0.38 years. These analyses were confirmed for the subgroups of patients diagnosed according to the Poser and McDonald criteria. The increase in age “is probably not just related to the shift in diagnostic criteria,” said Dr. Kister. When the researchers calculated the net average gains in years over the 13-year follow-up period, they found that patients who entered at the end of the enrollment period were 4.9 years older when they reached an EDSS of 6, compared with patients with an EDSS of 6 who entered at the beginning of the enrollment period.
International data show similar trends
Research conducted around the world shows similar trends, said Dr. Kister. In 2009, Veugelers et al. published the results of a study that included 1,752 patients with MS in Nova Scotia. Before the 1998 introduction of a drug insurance program that provides DMTs, the time to an EDSS of 6 was 14.4 years. After the introduction of this program, the time to EDSS of 6 was 18.6 years.
More recently, Capra et al. examined 1,324 patients with MS who attended an MS center in Brescia, Italy, during 1980-2010. They found that the age at which 50% of patients reached an EDSS of 6 was approximately 55 years in 1990. By 2010, the age at achieving this milestone had increased to approximately 63 years.
In a prospective study, Cree et al. examined the evolution of disability in 448 actively treated patients with relapsing-remitting MS and 69 patients with progressive MS. Approximately 45% of patients had no disability worsening during a 10-year follow-up period. Furthermore, a comparatively low 11% of patients had reached an EDSS of 6 at 10 years. The average disease duration of the cohort at that time was 17 years, said Dr. Kister. The results indicated that about 50% of patients would be expected to reach an EDSS of 6 after a disease duration of approximately 38 years, “which is much longer than in the natural history studies,” he added.
In 2019, Beiki et al. found that among patients with relapsing-remitting MS, the risk of reaching an EDSS of 6 decreased by 7% with each subsequent calendar year of diagnosis. The researchers did not observe a similar trend among patients with progressive MS. Their population-based, retrospective study included 7,331 patients in Sweden.
Two additional studies in Scandinavian populations add to the evidence of decreasing disability. In their examination of Swedish patients with MS who received a diagnosis of MS during 1968-2012, Burkill et al. found that the risk of death decreased over time. The hazard ratio of mortality for patients with MS, compared with a non-MS comparator group, decreased from 6.52 among those diagnosed during 1968-1980 to 2.08 for patients diagnosed during 2001-2012. The decrease in the risk of mortality was greater among patients with MS than in a matched comparator population. Similarly, in a nationwide, population-based study, Koch-Henriksen et al. found that all-cause excess mortality in Danish patients with MS decreased from 1950 through 1999.
The role of DMTs
The evidence suggests that DMTs are affecting the long-term progression of MS, said Dr. Kister. Palace et al. compared patients with MS in the UK who received treatment with interferon-beta with a modeled untreated cohort of patients in British Columbia. They found that treated patients reached an EDSS of 6 4 years later than did untreated patients.
Furthermore, an analysis by Brown et al. showed that the time to conversion to secondary progressive MS was longer among treated patients, compared with untreated patients. The risk of conversion was lower for patients treated with newer, more effective therapies (i.e., fingolimod, alemtuzumab, or natalizumab) than for those treated with glatiramer acetate or interferon beta.
Finally, Kingwell and colleagues examined the effect of treatment with interferon-beta on survival using an international cohort of approximately 6,000 patients with relapsing-remitting MS. They found that exposure to interferon-beta for more than 3 years was associated with a 32% reduction in the risk of mortality. They observed no similar risk reduction among patients exposed to interferon-beta for 6 months to 3 years.
Although these data are encouraging, other evidence indicates that the prevalence of MS in the United States has increased considerably in the past 40 years. Researchers estimate that 1 million Americans have MS, which “suggests that we are diagnosing many more mild cases,” said Dr. Kister. The burden of the disease remains high, he concluded.
Dr. Kister reported receiving consulting fees or research grants from Biogen, Roche, Genzyme and Genentech.
SOURCE: Kister I et al. ECTRIMS 2019. Abstract 281754.
STOCKHOLM – , according to an overview provided at the annual congress of the European Committee for Treatment and Research in Multiple Sclerosis. Data consistently indicate that the time that elapses before a patient requires a cane for ambulation has increased, and survival has likewise improved. “Some of the improvement can be attributed confidently to treatment effect,” said Ilya Kister, MD, associate professor of neurology at NYU Langone Health in New York. “We hope to see an even greater change with newer therapies.”
At the same time, neurologists appear to be diagnosing more cases of MS than they previously did, said Dr. Kister, which suggests that neurologists probably are diagnosing milder cases. The overall societal burden of MS remains high.
The relative prevalence of mild disability has increased
About 25 years have elapsed since the first disease-modifying treatment (DMT) for MS became available, and treatment has become widespread during that time. Dr. Kister and colleagues sought to determine whether the current clinical population of patients with MS, who for the most part receive DMTs, has less disability than do untreated patients or patients from natural history studies do. They identified the MS Severity Score (MSSS) as a measure with which to compare populations. The MSSS assigns a patient a ranking according to his or her level of disability, using a reference population of patients with the same disease duration for comparison. “MSSS can be conceptualized as rate of disability accumulation,” said Dr. Kister. “Lower MSSS corresponds to relatively slower disability accumulation, and higher MSSS to higher disability accumulation.”
The MSSS was developed using the Expanded Disability Status Scale (EDSS) score as a measure of disability. Because many neurologists do not routinely obtain EDSS scores for their patients, Dr. Kister and colleagues used the Patient-Determined Disease Steps (PDDS) to measure disability. As its name implies, the PDDS is a patient-reported outcome measure that mainly measures ambulation. It correlates strongly with EDSS, said Dr. Kister. He and colleagues used the PDDS to develop a reference table of MS disability, which they called the Patient-Derived MSSS.
The investigators examined a large sample of patients at NYU MS Center and Barnabas MS Center in Livingston, N.J. They grouped patients into sextiles according to their Patient-Derived MSSS. Dr. Kister and colleagues found that, rather than arriving at sextiles that contained equal numbers of patients, as would be expected if disability were distributed as in the reference population, they had significantly more patients in the two lowest sextiles and significantly fewer patients in the two highest sextiles. “This [result] suggests that the disability curve has indeed shifted toward the more benign end of the spectrum in the contemporary clinic population,” said Dr. Kister.
Other researchers have observed a similar phenomenon. George et al. published the results of a large, international collaboration in Neurology Genetics in 2016. After examining more than 7,000 patients, the investigators noted a similar overrepresentation of patients with milder severity scores and underrepresentation of patients with higher severity scores. These results support the hypothesis of a shift toward milder disability, said Dr. Kister.
Trend toward milder disability
The investigators next examined whether the rate of accumulation of disability among patients with MS had changed from year to year since DMTs were introduced. They conducted a univariate analysis of MSSS for 6,238 patients who were enrolled in the N.Y. State MS Consortium during 1996-2007. They found that patients who were enrolled in more recent years had significantly lower MSSS than patients who were enrolled in earlier years, regardless of disease duration. When Dr. Kister and colleagues replicated their analysis using EDSS, they found significantly lower levels of disability for patients enrolled in more recent years, except for patients with disease duration of 26-30 years. A multivariate analysis showed that the median MSSS of enrollees into the N.Y. State MS Consortium decreased from 5.04 in 1996 to 3.78 in 2006.
In a subsequent study, Dr. Kister and colleagues examined the age at which patients in the MSBase registry reached various disability milestones (e.g., EDSS of 6, which indicates the need of a cane to walk outdoors), according to their year of enrollment in the registry. They found a significant increase in age at milestone achievement with each subsequent calendar year. For example, for every consecutive year of enrollment, the age at which patients attained an EDSS of 6 increased by 0.38 years. These analyses were confirmed for the subgroups of patients diagnosed according to the Poser and McDonald criteria. The increase in age “is probably not just related to the shift in diagnostic criteria,” said Dr. Kister. When the researchers calculated the net average gains in years over the 13-year follow-up period, they found that patients who entered at the end of the enrollment period were 4.9 years older when they reached an EDSS of 6, compared with patients with an EDSS of 6 who entered at the beginning of the enrollment period.
International data show similar trends
Research conducted around the world shows similar trends, said Dr. Kister. In 2009, Veugelers et al. published the results of a study that included 1,752 patients with MS in Nova Scotia. Before the 1998 introduction of a drug insurance program that provides DMTs, the time to an EDSS of 6 was 14.4 years. After the introduction of this program, the time to EDSS of 6 was 18.6 years.
More recently, Capra et al. examined 1,324 patients with MS who attended an MS center in Brescia, Italy, during 1980-2010. They found that the age at which 50% of patients reached an EDSS of 6 was approximately 55 years in 1990. By 2010, the age at achieving this milestone had increased to approximately 63 years.
In a prospective study, Cree et al. examined the evolution of disability in 448 actively treated patients with relapsing-remitting MS and 69 patients with progressive MS. Approximately 45% of patients had no disability worsening during a 10-year follow-up period. Furthermore, a comparatively low 11% of patients had reached an EDSS of 6 at 10 years. The average disease duration of the cohort at that time was 17 years, said Dr. Kister. The results indicated that about 50% of patients would be expected to reach an EDSS of 6 after a disease duration of approximately 38 years, “which is much longer than in the natural history studies,” he added.
In 2019, Beiki et al. found that among patients with relapsing-remitting MS, the risk of reaching an EDSS of 6 decreased by 7% with each subsequent calendar year of diagnosis. The researchers did not observe a similar trend among patients with progressive MS. Their population-based, retrospective study included 7,331 patients in Sweden.
Two additional studies in Scandinavian populations add to the evidence of decreasing disability. In their examination of Swedish patients with MS who received a diagnosis of MS during 1968-2012, Burkill et al. found that the risk of death decreased over time. The hazard ratio of mortality for patients with MS, compared with a non-MS comparator group, decreased from 6.52 among those diagnosed during 1968-1980 to 2.08 for patients diagnosed during 2001-2012. The decrease in the risk of mortality was greater among patients with MS than in a matched comparator population. Similarly, in a nationwide, population-based study, Koch-Henriksen et al. found that all-cause excess mortality in Danish patients with MS decreased from 1950 through 1999.
The role of DMTs
The evidence suggests that DMTs are affecting the long-term progression of MS, said Dr. Kister. Palace et al. compared patients with MS in the UK who received treatment with interferon-beta with a modeled untreated cohort of patients in British Columbia. They found that treated patients reached an EDSS of 6 4 years later than did untreated patients.
Furthermore, an analysis by Brown et al. showed that the time to conversion to secondary progressive MS was longer among treated patients, compared with untreated patients. The risk of conversion was lower for patients treated with newer, more effective therapies (i.e., fingolimod, alemtuzumab, or natalizumab) than for those treated with glatiramer acetate or interferon beta.
Finally, Kingwell and colleagues examined the effect of treatment with interferon-beta on survival using an international cohort of approximately 6,000 patients with relapsing-remitting MS. They found that exposure to interferon-beta for more than 3 years was associated with a 32% reduction in the risk of mortality. They observed no similar risk reduction among patients exposed to interferon-beta for 6 months to 3 years.
Although these data are encouraging, other evidence indicates that the prevalence of MS in the United States has increased considerably in the past 40 years. Researchers estimate that 1 million Americans have MS, which “suggests that we are diagnosing many more mild cases,” said Dr. Kister. The burden of the disease remains high, he concluded.
Dr. Kister reported receiving consulting fees or research grants from Biogen, Roche, Genzyme and Genentech.
SOURCE: Kister I et al. ECTRIMS 2019. Abstract 281754.
STOCKHOLM – , according to an overview provided at the annual congress of the European Committee for Treatment and Research in Multiple Sclerosis. Data consistently indicate that the time that elapses before a patient requires a cane for ambulation has increased, and survival has likewise improved. “Some of the improvement can be attributed confidently to treatment effect,” said Ilya Kister, MD, associate professor of neurology at NYU Langone Health in New York. “We hope to see an even greater change with newer therapies.”
At the same time, neurologists appear to be diagnosing more cases of MS than they previously did, said Dr. Kister, which suggests that neurologists probably are diagnosing milder cases. The overall societal burden of MS remains high.
The relative prevalence of mild disability has increased
About 25 years have elapsed since the first disease-modifying treatment (DMT) for MS became available, and treatment has become widespread during that time. Dr. Kister and colleagues sought to determine whether the current clinical population of patients with MS, who for the most part receive DMTs, has less disability than do untreated patients or patients from natural history studies do. They identified the MS Severity Score (MSSS) as a measure with which to compare populations. The MSSS assigns a patient a ranking according to his or her level of disability, using a reference population of patients with the same disease duration for comparison. “MSSS can be conceptualized as rate of disability accumulation,” said Dr. Kister. “Lower MSSS corresponds to relatively slower disability accumulation, and higher MSSS to higher disability accumulation.”
The MSSS was developed using the Expanded Disability Status Scale (EDSS) score as a measure of disability. Because many neurologists do not routinely obtain EDSS scores for their patients, Dr. Kister and colleagues used the Patient-Determined Disease Steps (PDDS) to measure disability. As its name implies, the PDDS is a patient-reported outcome measure that mainly measures ambulation. It correlates strongly with EDSS, said Dr. Kister. He and colleagues used the PDDS to develop a reference table of MS disability, which they called the Patient-Derived MSSS.
The investigators examined a large sample of patients at NYU MS Center and Barnabas MS Center in Livingston, N.J. They grouped patients into sextiles according to their Patient-Derived MSSS. Dr. Kister and colleagues found that, rather than arriving at sextiles that contained equal numbers of patients, as would be expected if disability were distributed as in the reference population, they had significantly more patients in the two lowest sextiles and significantly fewer patients in the two highest sextiles. “This [result] suggests that the disability curve has indeed shifted toward the more benign end of the spectrum in the contemporary clinic population,” said Dr. Kister.
Other researchers have observed a similar phenomenon. George et al. published the results of a large, international collaboration in Neurology Genetics in 2016. After examining more than 7,000 patients, the investigators noted a similar overrepresentation of patients with milder severity scores and underrepresentation of patients with higher severity scores. These results support the hypothesis of a shift toward milder disability, said Dr. Kister.
Trend toward milder disability
The investigators next examined whether the rate of accumulation of disability among patients with MS had changed from year to year since DMTs were introduced. They conducted a univariate analysis of MSSS for 6,238 patients who were enrolled in the N.Y. State MS Consortium during 1996-2007. They found that patients who were enrolled in more recent years had significantly lower MSSS than patients who were enrolled in earlier years, regardless of disease duration. When Dr. Kister and colleagues replicated their analysis using EDSS, they found significantly lower levels of disability for patients enrolled in more recent years, except for patients with disease duration of 26-30 years. A multivariate analysis showed that the median MSSS of enrollees into the N.Y. State MS Consortium decreased from 5.04 in 1996 to 3.78 in 2006.
In a subsequent study, Dr. Kister and colleagues examined the age at which patients in the MSBase registry reached various disability milestones (e.g., EDSS of 6, which indicates the need of a cane to walk outdoors), according to their year of enrollment in the registry. They found a significant increase in age at milestone achievement with each subsequent calendar year. For example, for every consecutive year of enrollment, the age at which patients attained an EDSS of 6 increased by 0.38 years. These analyses were confirmed for the subgroups of patients diagnosed according to the Poser and McDonald criteria. The increase in age “is probably not just related to the shift in diagnostic criteria,” said Dr. Kister. When the researchers calculated the net average gains in years over the 13-year follow-up period, they found that patients who entered at the end of the enrollment period were 4.9 years older when they reached an EDSS of 6, compared with patients with an EDSS of 6 who entered at the beginning of the enrollment period.
International data show similar trends
Research conducted around the world shows similar trends, said Dr. Kister. In 2009, Veugelers et al. published the results of a study that included 1,752 patients with MS in Nova Scotia. Before the 1998 introduction of a drug insurance program that provides DMTs, the time to an EDSS of 6 was 14.4 years. After the introduction of this program, the time to EDSS of 6 was 18.6 years.
More recently, Capra et al. examined 1,324 patients with MS who attended an MS center in Brescia, Italy, during 1980-2010. They found that the age at which 50% of patients reached an EDSS of 6 was approximately 55 years in 1990. By 2010, the age at achieving this milestone had increased to approximately 63 years.
In a prospective study, Cree et al. examined the evolution of disability in 448 actively treated patients with relapsing-remitting MS and 69 patients with progressive MS. Approximately 45% of patients had no disability worsening during a 10-year follow-up period. Furthermore, a comparatively low 11% of patients had reached an EDSS of 6 at 10 years. The average disease duration of the cohort at that time was 17 years, said Dr. Kister. The results indicated that about 50% of patients would be expected to reach an EDSS of 6 after a disease duration of approximately 38 years, “which is much longer than in the natural history studies,” he added.
In 2019, Beiki et al. found that among patients with relapsing-remitting MS, the risk of reaching an EDSS of 6 decreased by 7% with each subsequent calendar year of diagnosis. The researchers did not observe a similar trend among patients with progressive MS. Their population-based, retrospective study included 7,331 patients in Sweden.
Two additional studies in Scandinavian populations add to the evidence of decreasing disability. In their examination of Swedish patients with MS who received a diagnosis of MS during 1968-2012, Burkill et al. found that the risk of death decreased over time. The hazard ratio of mortality for patients with MS, compared with a non-MS comparator group, decreased from 6.52 among those diagnosed during 1968-1980 to 2.08 for patients diagnosed during 2001-2012. The decrease in the risk of mortality was greater among patients with MS than in a matched comparator population. Similarly, in a nationwide, population-based study, Koch-Henriksen et al. found that all-cause excess mortality in Danish patients with MS decreased from 1950 through 1999.
The role of DMTs
The evidence suggests that DMTs are affecting the long-term progression of MS, said Dr. Kister. Palace et al. compared patients with MS in the UK who received treatment with interferon-beta with a modeled untreated cohort of patients in British Columbia. They found that treated patients reached an EDSS of 6 4 years later than did untreated patients.
Furthermore, an analysis by Brown et al. showed that the time to conversion to secondary progressive MS was longer among treated patients, compared with untreated patients. The risk of conversion was lower for patients treated with newer, more effective therapies (i.e., fingolimod, alemtuzumab, or natalizumab) than for those treated with glatiramer acetate or interferon beta.
Finally, Kingwell and colleagues examined the effect of treatment with interferon-beta on survival using an international cohort of approximately 6,000 patients with relapsing-remitting MS. They found that exposure to interferon-beta for more than 3 years was associated with a 32% reduction in the risk of mortality. They observed no similar risk reduction among patients exposed to interferon-beta for 6 months to 3 years.
Although these data are encouraging, other evidence indicates that the prevalence of MS in the United States has increased considerably in the past 40 years. Researchers estimate that 1 million Americans have MS, which “suggests that we are diagnosing many more mild cases,” said Dr. Kister. The burden of the disease remains high, he concluded.
Dr. Kister reported receiving consulting fees or research grants from Biogen, Roche, Genzyme and Genentech.
SOURCE: Kister I et al. ECTRIMS 2019. Abstract 281754.
EXPERT ANALYSIS FROM ECTRIMS 2019
Medicare beneficiaries pay most for Alzheimer’s
according to the Kaiser Family Foundation.
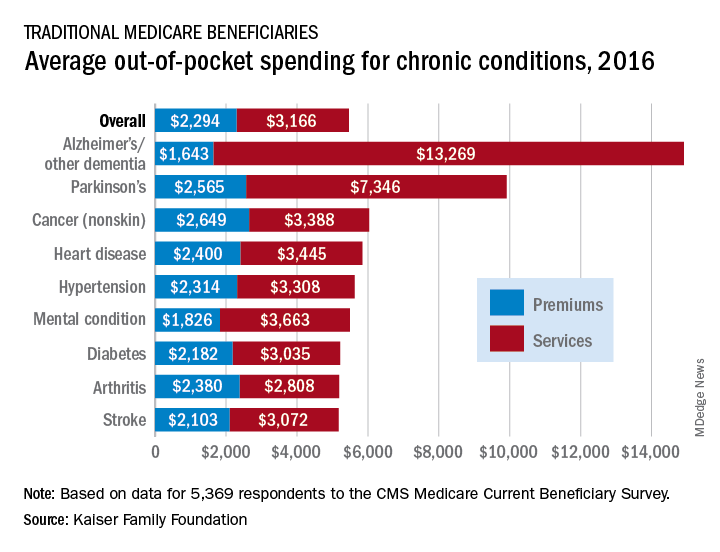
Out-of-pocket spending for Alzheimer’s disease or other dementia was higher than any other chronic condition, averaging $14,913 in 2016 (the latest year for which data are available), compared with $5,460 for all beneficiaries in traditional Medicare, Kaiser investigators said in a recent report based on data for 5,369 respondents to the Medicare Current Beneficiary Survey.
Those totals were divided between services – including long-term care facilities, medical providers and supplies, and prescription drugs – and premiums for Medicare and other types of supplemental insurance. The premium associated with Alzheimer’s, $1,643, was the lowest of any major chronic condition, but the average cost for services, $13,269, was almost twice as high as the next most expensive condition, Parkinson’s disease, and more than four times higher than the overall Medicare average, Juliette Cubanski, PhD, and associates said.
Out-of-pocket costs are higher for patients with Alzheimer’s and Parkinson’s because “these beneficiaries are more likely to reside in a long-term care facility than those with other conditions,” they said. In 2016, out-of-pocket spending on long-term care facility services averaged over $27,000 for Medicare beneficiaries with Alzheimer’s and other dementia and over $28,000 for those with Parkinson’s disease. For all traditional Medicare beneficiaries, average out-of-pocket spending on such services was $1,014.
“The fact that traditional Medicare does not have an annual out-of-pocket limit and does not cover certain services that older adults are more likely to need may undermine the financial security that Medicare provides, especially for people with significant needs and limited incomes. Addressing these gaps would help to alleviate the financial burden of health care for people with Medicare, although doing so would also increase federal spending and taxes,” Dr. Cubanski and associates wrote.
according to the Kaiser Family Foundation.

Out-of-pocket spending for Alzheimer’s disease or other dementia was higher than any other chronic condition, averaging $14,913 in 2016 (the latest year for which data are available), compared with $5,460 for all beneficiaries in traditional Medicare, Kaiser investigators said in a recent report based on data for 5,369 respondents to the Medicare Current Beneficiary Survey.
Those totals were divided between services – including long-term care facilities, medical providers and supplies, and prescription drugs – and premiums for Medicare and other types of supplemental insurance. The premium associated with Alzheimer’s, $1,643, was the lowest of any major chronic condition, but the average cost for services, $13,269, was almost twice as high as the next most expensive condition, Parkinson’s disease, and more than four times higher than the overall Medicare average, Juliette Cubanski, PhD, and associates said.
Out-of-pocket costs are higher for patients with Alzheimer’s and Parkinson’s because “these beneficiaries are more likely to reside in a long-term care facility than those with other conditions,” they said. In 2016, out-of-pocket spending on long-term care facility services averaged over $27,000 for Medicare beneficiaries with Alzheimer’s and other dementia and over $28,000 for those with Parkinson’s disease. For all traditional Medicare beneficiaries, average out-of-pocket spending on such services was $1,014.
“The fact that traditional Medicare does not have an annual out-of-pocket limit and does not cover certain services that older adults are more likely to need may undermine the financial security that Medicare provides, especially for people with significant needs and limited incomes. Addressing these gaps would help to alleviate the financial burden of health care for people with Medicare, although doing so would also increase federal spending and taxes,” Dr. Cubanski and associates wrote.
according to the Kaiser Family Foundation.

Out-of-pocket spending for Alzheimer’s disease or other dementia was higher than any other chronic condition, averaging $14,913 in 2016 (the latest year for which data are available), compared with $5,460 for all beneficiaries in traditional Medicare, Kaiser investigators said in a recent report based on data for 5,369 respondents to the Medicare Current Beneficiary Survey.
Those totals were divided between services – including long-term care facilities, medical providers and supplies, and prescription drugs – and premiums for Medicare and other types of supplemental insurance. The premium associated with Alzheimer’s, $1,643, was the lowest of any major chronic condition, but the average cost for services, $13,269, was almost twice as high as the next most expensive condition, Parkinson’s disease, and more than four times higher than the overall Medicare average, Juliette Cubanski, PhD, and associates said.
Out-of-pocket costs are higher for patients with Alzheimer’s and Parkinson’s because “these beneficiaries are more likely to reside in a long-term care facility than those with other conditions,” they said. In 2016, out-of-pocket spending on long-term care facility services averaged over $27,000 for Medicare beneficiaries with Alzheimer’s and other dementia and over $28,000 for those with Parkinson’s disease. For all traditional Medicare beneficiaries, average out-of-pocket spending on such services was $1,014.
“The fact that traditional Medicare does not have an annual out-of-pocket limit and does not cover certain services that older adults are more likely to need may undermine the financial security that Medicare provides, especially for people with significant needs and limited incomes. Addressing these gaps would help to alleviate the financial burden of health care for people with Medicare, although doing so would also increase federal spending and taxes,” Dr. Cubanski and associates wrote.
Worsening nausea, vomiting, and dizziness • 20-pound weight loss in 2 months • mild hearing loss • reoccurring episodes of falls • Dx?
THE CASE
A 26-year-old Hispanic/African American woman presented to our clinic with a 2-month history of nausea and vomiting, along with dizziness. The nausea and vomiting persistently worsened, and she was only able to tolerate apples and berries. During this 2-month period, she lost 20 pounds and her symptoms progressed to include pruritus, ataxia, and mild hearing loss, with reoccurring episodes of falls.
THE DIAGNOSIS
On examination, she was found to be bradycardic with a heart rate of 47 beats/min, right- axis deviation, and inverted T waves in leads I, II, and augmented vector left. Her family history included the death of an aunt who was in her early 30s due to an unknown heart condition.
Echocardiogram identified mild mitral valve regurgitation with an ejection fraction of 55% to 60% (reference range: 55%-70%). Cardiology determined that her bradycardia was not the source of her symptoms. A neurologic exam identified 3+ hyperreflexia (indicating the reflex was increased), tandem gait instability, and left oculomotor dysfunction.
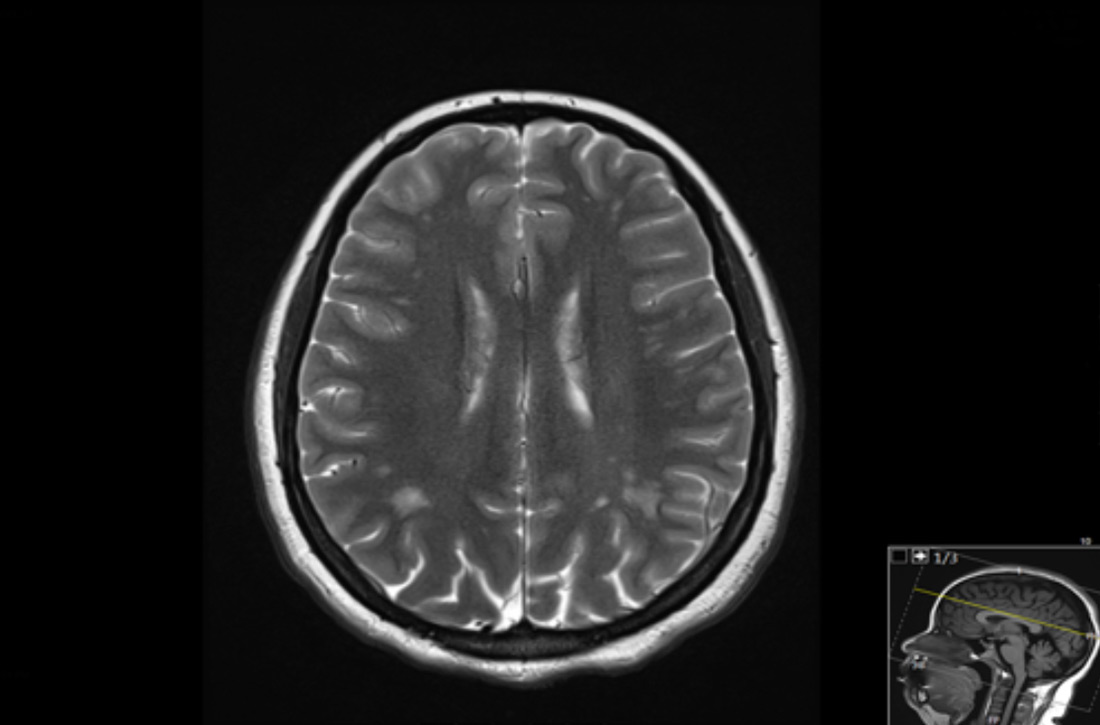
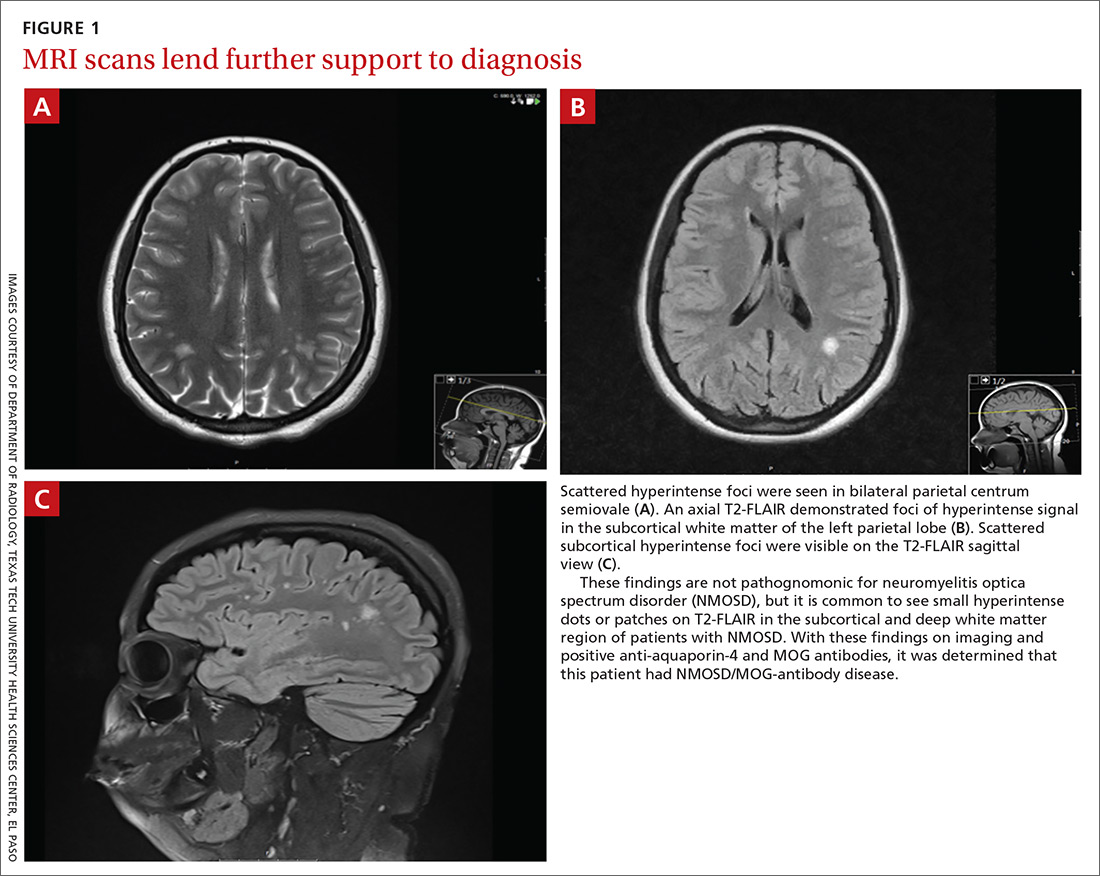
Brain magnetic resonance imaging (MRI) identified bilateral parietal white matter lesions where a demyelinating process could not be excluded (FIGURE 1A). The patient’s symptoms of nausea and vomiting continued, and she only tolerated peanuts and liquids. An MRI of the spine was negative.
Laboratory testing revealed that the patient was negative for human immunodeficiency virus (HIV), syphilis, Lyme disease, and lupus. Her thyroid-stimulating hormone level was 1.7 mIU/L (reference range: 0.4-4.2 mIU/L), and her vitamin B12 level was 504 pg/mL (reference range: 160-950 pg/mL).
The patient’s lumbar puncture was negative for oligoclonal bands. The IgG synthesis rate/index cerebrospinal fluid (CSF) was –3.9, ruling out multiple sclerosis. Her CSF culture was negative, with a glucose level of 42 mg/dL (reference range: 70-110 mg/dL), colorless appearance, 1 white blood cell, and spinal albumin of 12.2 mg/dL (reference range: 8-42 mg/dL). The visual evoked potential was negative. The aquaporin-4 (AQP4) antibody was positive at 3.4 U/mL, and the myelin oligodendrocyte glycoprotein (MOG) antibody was positive.
Gastroenterology concluded a normal gastric accommodation and unremarkable computed tomography (CT) enterography. Moderate erosions were identified in the stomach with an erythematous gastropathy. The patient was placed on a proton pump inhibitor.
Continue to: Following the examination...
Following the examination and laboratory testing, the patient was admitted under our family medicine service for neuromyelitis optica (NMO) affecting the area postrema. NMO, also known as Devic’s disease, is an autoimmune disorder that affects the spinal cord and optic nerves. Autoantibodies against AQP4 are created in the periphery and are directed against astrocytes in the central nervous system. These antibodies bind to the foot processes of astrocytes, inducing complement-mediated cell damage and granulocyte infiltration.1-5
Intravenous methylprednisolone was initiated at 250 mg every 6 hours for 3 days. A repeat brain MRI demonstrated nonspecific multiple scattered foci of hyperintense signal involving the subcortical supratentorial white matter without abnormal enhancement, most likely representing nonactive demyelinating plaques (FIGURES 1B and 1C).
Dx is revisited. Our patient was referred to an NMO clinic for evaluation. After further testing (including a repeat MRI based on the neurologist’s specifications, anti-aquaporin antibody testing, and MOG-antibody testing) and case discussion, it was determined that the patient had MOG-antibody disease. This disease, along with NMO, comprise a spectrum of disorders referred to as neuromyelitis optica spectrum disorder (NMOSD).
The patient was subsequently prescribed a rituximab infusion, 500 mg/50 mL, to treat the current attack. One infusion was to be completed weekly for 2 weeks with plans to repeat treatment every 6 months to prevent flares of NMO. During the first dose, the patient had a reaction to the treatment, which caused pruritus and chest tightness. She was able to complete the infusion after being treated with diphenhydramine.
Tx continued. In order to complete the second of 2 infusions of rituximab, the patient was pretreated with oral methylprednisolone the night before the infusion, along with diphenhydramine and acetaminophen on the day of treatment. Fortunately, the patient tolerated the infusion well with no adverse effects or reactions.
Continue to: DISCUSSION
DISCUSSION
Within the NMO spectrum, the MOG antibody is positive in up to 42% of AQP4-seronegative cases.6 MOG is a minor myelin component that is expressed exclusively in the central nervous system on the surface of myelin and oligodendrocyte processes. The role of this glycoprotein is not well understood but is hypothesized to function as a cell surface receptor or cell adhesion molecule.7
Among a cohort of 252 patients from the United Kingdom who tested positive for the MOG-IgG1 antibody, optic neuritis was seen in 55%, while 18% experienced transverse myelitis, and 15% had a history of area postrema syndrome. A brain MRI identified lesions in all areas of the brain including the brain stem, cerebellum, and cerebral hemispheres.8
Risk factors for NMOSD include female gender, Asian and African ethnicities, Epstein Barr virus seropositivity, and tobacco abuse.
Differential diagnosis. Many diseases or conditions that are inflammatory, autoimmune, infectious, or neoplastic can involve the central nervous system and mimic the clinical and radiologic phenotypes of NMOSD-AQP4. They include lupus, SjÖgren’s syndrome, multiple sclerosis, sarcoidosis, acute disseminated encephalomyelitis, HIV, and vitamin B12 deficiency.
Treatment. The standard treatment is intravenous methylprednisolone, 1 g/d for 3 to 5 days followed by a steroid taper. Therapeutic plasma exchange is recommended for refractory cases and in patients with spinal cord demyelination.9-11 Rituximab is the first-line therapy for attack prevention12-15 in NMOSD broadly and may be effective in MOG antibody disease, as well. In an open-label study of patients with NMOSD treated with rituximab, 64% were relapse free at follow-up, which ranged from 12 to 67 months.13 In a long-term study of patients treated with rituximab, 87% maintained a reduced relapse rate and 93% had improvement or stability over a 5-year follow-up.14
Continue to: Our patient
Our patient. After her diagnosis of NMOSD/MOG-antibody disease, our patient’s symptoms progressed to include vertigo, vestibular ataxia, pruritus, left foot drop, lower extremity numbness, and decreased hearing. After the second rituximab infusion her symptoms continued, but over time stabilized and have not worsened. She currently receives gabapentin 300 mg every 8 hours, as needed, for extremity numbness (which has been working well) along with sertraline 100 mg/d for depression.
Subsequent office visits have showed no further weight loss. Based on the current response to the rituximab, her prognosis is undetermined by Neurology as they continue to monitor for progression.
THE TAKEAWAY
Vestibular ataxia, foot drop, pruritus, vertigo, decreased hearing, numbness, and oculomotor dysfunction in the presence of nausea and vomiting should raise suspicion for NMOSD. The presence of AQP4 antibodies along with demyelinating central nervous system lesions, is highly indicative of NMO. The presence of MOG antibodies may indicate NMOSD/MOG-antibody disease. The initial treatment of NMOSD is intravenous methylprednisolone, which can be followed by treatment with rituximab to achieve remission.
CORRESPONDENCE
Daniel Murphy, MD, FAAFP, Department of Family and Community Medicine, Texas Tech University Health Science Center El Paso, 9849 Kenworthy Street, El Paso, Texas 79924; [email protected]
1. Hinson SR, Pittock SJ, Lucchinetti CF, et al. Pathogenic potential of IgG binding to water channel extracellular domain in neuromyelitis optica. Neurology. 2007;69:2221-2231.
2. Ratelade J, Zhang H, Saadoun S, et al. Neuromyelitis optica IgG and natural killer cells Produce NMO lesions in mice without myelin loss. Acta Neuropathol. 2012;123:861-872.
3. Saadoun S, Waters P, Bell BA, et al. Intra-cerebral injection of neuromyelitis optica immunoglobulin G and human complement produces neuromyelitis optica lesions in mice. Brain. 2010;133:349-361.
4. Takahashi T, Fujihara K, Nakashima I, et al. Anti-aquaporin-4 antibody is involved in the pathogenesis of NMO: a study on antibody titer. Brain. 2007;130:1235-1243.
5. Jarius S, Aboul-Enein F, Waters P, et al. Antibody to aquaporin-4 in the long-term course of neuromyelitis optica. Brain. 2008;131:3072-3080.
6. Hamid SHM, Whittam D, Mutch K, et al. What proportion of AQP4-IgG-negative NMO spectrum disorder patients are Mog-IgG positive? A cross sectional study of 132 patients. J Neurol. 2017; 264:2088-2094.
7. Peschl P, Bradi M, Hoftberger R, et al. Myelin oligodendrocyte glycoprotein: deciphering a target in inflammatory demyelinating diseases. Front Immunol. 2017;8:529.
8. Jurynczyk M, Messina S, Woodhall MR, et al. Clinical presentation and prognosis in MOG-antibody disease: a UK study. Brain. 2017;140:3128-3138.
9. Sellner J, Boggild M, Clanet M, et al. EFNS Guidelines on diagnosis and management of neuromyelitis optica. Eur J Neurol. 2010;17:1019-1032.
10. Kleiter I, Gahlen A, Borisow N, et al. Neuromyelitis optica: evaluation of 871 attacks and 1,153 treatment courses. Ann Neurol. 2016;79:206-216.
11. Watanabe S, Nakashima I, Misu T, et al. Therapeutic efficacy of plasma exchange in NMO-IgG-positive patients with neuromyelitis optica. Mult Scler. 2007;13:128-132.
12. Collongues N, Brassat D, Maillart E, et al. Efficacy of rituximab in refractory neuromyelitis optica. Mult Scler. 2016;22:955-959.
13. Collongues N, de Seze J. An update on the evidence for the efficacy and safety of rituximab in the management of neuromyelitis optica. Ther Adv Neurol Disord. 2016;9:180-188.
14. Kim SH, Huh SY, Lee SJ, et al. A 5-year follow-up of rituximab treatment in patients with neuromyelitis optica spectrum disorder. JAMA Neurol. 2013;70:1110-1117.
15. Kim SH, Kim W, Li XF, et al. Repeated treatment with rituximab based on the assessment of peripheral circulating memory B cells in patients with relapsing neuromyelitis optica over 2 years. Arch Neurol. 2011;68:1412-1420.
THE CASE
A 26-year-old Hispanic/African American woman presented to our clinic with a 2-month history of nausea and vomiting, along with dizziness. The nausea and vomiting persistently worsened, and she was only able to tolerate apples and berries. During this 2-month period, she lost 20 pounds and her symptoms progressed to include pruritus, ataxia, and mild hearing loss, with reoccurring episodes of falls.
THE DIAGNOSIS
On examination, she was found to be bradycardic with a heart rate of 47 beats/min, right- axis deviation, and inverted T waves in leads I, II, and augmented vector left. Her family history included the death of an aunt who was in her early 30s due to an unknown heart condition.
Echocardiogram identified mild mitral valve regurgitation with an ejection fraction of 55% to 60% (reference range: 55%-70%). Cardiology determined that her bradycardia was not the source of her symptoms. A neurologic exam identified 3+ hyperreflexia (indicating the reflex was increased), tandem gait instability, and left oculomotor dysfunction.
Brain magnetic resonance imaging (MRI) identified bilateral parietal white matter lesions where a demyelinating process could not be excluded (FIGURE 1A). The patient’s symptoms of nausea and vomiting continued, and she only tolerated peanuts and liquids. An MRI of the spine was negative.

Laboratory testing revealed that the patient was negative for human immunodeficiency virus (HIV), syphilis, Lyme disease, and lupus. Her thyroid-stimulating hormone level was 1.7 mIU/L (reference range: 0.4-4.2 mIU/L), and her vitamin B12 level was 504 pg/mL (reference range: 160-950 pg/mL).
The patient’s lumbar puncture was negative for oligoclonal bands. The IgG synthesis rate/index cerebrospinal fluid (CSF) was –3.9, ruling out multiple sclerosis. Her CSF culture was negative, with a glucose level of 42 mg/dL (reference range: 70-110 mg/dL), colorless appearance, 1 white blood cell, and spinal albumin of 12.2 mg/dL (reference range: 8-42 mg/dL). The visual evoked potential was negative. The aquaporin-4 (AQP4) antibody was positive at 3.4 U/mL, and the myelin oligodendrocyte glycoprotein (MOG) antibody was positive.
Gastroenterology concluded a normal gastric accommodation and unremarkable computed tomography (CT) enterography. Moderate erosions were identified in the stomach with an erythematous gastropathy. The patient was placed on a proton pump inhibitor.
Continue to: Following the examination...
Following the examination and laboratory testing, the patient was admitted under our family medicine service for neuromyelitis optica (NMO) affecting the area postrema. NMO, also known as Devic’s disease, is an autoimmune disorder that affects the spinal cord and optic nerves. Autoantibodies against AQP4 are created in the periphery and are directed against astrocytes in the central nervous system. These antibodies bind to the foot processes of astrocytes, inducing complement-mediated cell damage and granulocyte infiltration.1-5
Intravenous methylprednisolone was initiated at 250 mg every 6 hours for 3 days. A repeat brain MRI demonstrated nonspecific multiple scattered foci of hyperintense signal involving the subcortical supratentorial white matter without abnormal enhancement, most likely representing nonactive demyelinating plaques (FIGURES 1B and 1C).
Dx is revisited. Our patient was referred to an NMO clinic for evaluation. After further testing (including a repeat MRI based on the neurologist’s specifications, anti-aquaporin antibody testing, and MOG-antibody testing) and case discussion, it was determined that the patient had MOG-antibody disease. This disease, along with NMO, comprise a spectrum of disorders referred to as neuromyelitis optica spectrum disorder (NMOSD).
The patient was subsequently prescribed a rituximab infusion, 500 mg/50 mL, to treat the current attack. One infusion was to be completed weekly for 2 weeks with plans to repeat treatment every 6 months to prevent flares of NMO. During the first dose, the patient had a reaction to the treatment, which caused pruritus and chest tightness. She was able to complete the infusion after being treated with diphenhydramine.
Tx continued. In order to complete the second of 2 infusions of rituximab, the patient was pretreated with oral methylprednisolone the night before the infusion, along with diphenhydramine and acetaminophen on the day of treatment. Fortunately, the patient tolerated the infusion well with no adverse effects or reactions.
Continue to: DISCUSSION
DISCUSSION
Within the NMO spectrum, the MOG antibody is positive in up to 42% of AQP4-seronegative cases.6 MOG is a minor myelin component that is expressed exclusively in the central nervous system on the surface of myelin and oligodendrocyte processes. The role of this glycoprotein is not well understood but is hypothesized to function as a cell surface receptor or cell adhesion molecule.7
Among a cohort of 252 patients from the United Kingdom who tested positive for the MOG-IgG1 antibody, optic neuritis was seen in 55%, while 18% experienced transverse myelitis, and 15% had a history of area postrema syndrome. A brain MRI identified lesions in all areas of the brain including the brain stem, cerebellum, and cerebral hemispheres.8
Risk factors for NMOSD include female gender, Asian and African ethnicities, Epstein Barr virus seropositivity, and tobacco abuse.
Differential diagnosis. Many diseases or conditions that are inflammatory, autoimmune, infectious, or neoplastic can involve the central nervous system and mimic the clinical and radiologic phenotypes of NMOSD-AQP4. They include lupus, SjÖgren’s syndrome, multiple sclerosis, sarcoidosis, acute disseminated encephalomyelitis, HIV, and vitamin B12 deficiency.
Treatment. The standard treatment is intravenous methylprednisolone, 1 g/d for 3 to 5 days followed by a steroid taper. Therapeutic plasma exchange is recommended for refractory cases and in patients with spinal cord demyelination.9-11 Rituximab is the first-line therapy for attack prevention12-15 in NMOSD broadly and may be effective in MOG antibody disease, as well. In an open-label study of patients with NMOSD treated with rituximab, 64% were relapse free at follow-up, which ranged from 12 to 67 months.13 In a long-term study of patients treated with rituximab, 87% maintained a reduced relapse rate and 93% had improvement or stability over a 5-year follow-up.14
Continue to: Our patient
Our patient. After her diagnosis of NMOSD/MOG-antibody disease, our patient’s symptoms progressed to include vertigo, vestibular ataxia, pruritus, left foot drop, lower extremity numbness, and decreased hearing. After the second rituximab infusion her symptoms continued, but over time stabilized and have not worsened. She currently receives gabapentin 300 mg every 8 hours, as needed, for extremity numbness (which has been working well) along with sertraline 100 mg/d for depression.
Subsequent office visits have showed no further weight loss. Based on the current response to the rituximab, her prognosis is undetermined by Neurology as they continue to monitor for progression.
THE TAKEAWAY
Vestibular ataxia, foot drop, pruritus, vertigo, decreased hearing, numbness, and oculomotor dysfunction in the presence of nausea and vomiting should raise suspicion for NMOSD. The presence of AQP4 antibodies along with demyelinating central nervous system lesions, is highly indicative of NMO. The presence of MOG antibodies may indicate NMOSD/MOG-antibody disease. The initial treatment of NMOSD is intravenous methylprednisolone, which can be followed by treatment with rituximab to achieve remission.
CORRESPONDENCE
Daniel Murphy, MD, FAAFP, Department of Family and Community Medicine, Texas Tech University Health Science Center El Paso, 9849 Kenworthy Street, El Paso, Texas 79924; [email protected]
THE CASE
A 26-year-old Hispanic/African American woman presented to our clinic with a 2-month history of nausea and vomiting, along with dizziness. The nausea and vomiting persistently worsened, and she was only able to tolerate apples and berries. During this 2-month period, she lost 20 pounds and her symptoms progressed to include pruritus, ataxia, and mild hearing loss, with reoccurring episodes of falls.
THE DIAGNOSIS
On examination, she was found to be bradycardic with a heart rate of 47 beats/min, right- axis deviation, and inverted T waves in leads I, II, and augmented vector left. Her family history included the death of an aunt who was in her early 30s due to an unknown heart condition.
Echocardiogram identified mild mitral valve regurgitation with an ejection fraction of 55% to 60% (reference range: 55%-70%). Cardiology determined that her bradycardia was not the source of her symptoms. A neurologic exam identified 3+ hyperreflexia (indicating the reflex was increased), tandem gait instability, and left oculomotor dysfunction.
Brain magnetic resonance imaging (MRI) identified bilateral parietal white matter lesions where a demyelinating process could not be excluded (FIGURE 1A). The patient’s symptoms of nausea and vomiting continued, and she only tolerated peanuts and liquids. An MRI of the spine was negative.

Laboratory testing revealed that the patient was negative for human immunodeficiency virus (HIV), syphilis, Lyme disease, and lupus. Her thyroid-stimulating hormone level was 1.7 mIU/L (reference range: 0.4-4.2 mIU/L), and her vitamin B12 level was 504 pg/mL (reference range: 160-950 pg/mL).
The patient’s lumbar puncture was negative for oligoclonal bands. The IgG synthesis rate/index cerebrospinal fluid (CSF) was –3.9, ruling out multiple sclerosis. Her CSF culture was negative, with a glucose level of 42 mg/dL (reference range: 70-110 mg/dL), colorless appearance, 1 white blood cell, and spinal albumin of 12.2 mg/dL (reference range: 8-42 mg/dL). The visual evoked potential was negative. The aquaporin-4 (AQP4) antibody was positive at 3.4 U/mL, and the myelin oligodendrocyte glycoprotein (MOG) antibody was positive.
Gastroenterology concluded a normal gastric accommodation and unremarkable computed tomography (CT) enterography. Moderate erosions were identified in the stomach with an erythematous gastropathy. The patient was placed on a proton pump inhibitor.
Continue to: Following the examination...
Following the examination and laboratory testing, the patient was admitted under our family medicine service for neuromyelitis optica (NMO) affecting the area postrema. NMO, also known as Devic’s disease, is an autoimmune disorder that affects the spinal cord and optic nerves. Autoantibodies against AQP4 are created in the periphery and are directed against astrocytes in the central nervous system. These antibodies bind to the foot processes of astrocytes, inducing complement-mediated cell damage and granulocyte infiltration.1-5
Intravenous methylprednisolone was initiated at 250 mg every 6 hours for 3 days. A repeat brain MRI demonstrated nonspecific multiple scattered foci of hyperintense signal involving the subcortical supratentorial white matter without abnormal enhancement, most likely representing nonactive demyelinating plaques (FIGURES 1B and 1C).
Dx is revisited. Our patient was referred to an NMO clinic for evaluation. After further testing (including a repeat MRI based on the neurologist’s specifications, anti-aquaporin antibody testing, and MOG-antibody testing) and case discussion, it was determined that the patient had MOG-antibody disease. This disease, along with NMO, comprise a spectrum of disorders referred to as neuromyelitis optica spectrum disorder (NMOSD).
The patient was subsequently prescribed a rituximab infusion, 500 mg/50 mL, to treat the current attack. One infusion was to be completed weekly for 2 weeks with plans to repeat treatment every 6 months to prevent flares of NMO. During the first dose, the patient had a reaction to the treatment, which caused pruritus and chest tightness. She was able to complete the infusion after being treated with diphenhydramine.
Tx continued. In order to complete the second of 2 infusions of rituximab, the patient was pretreated with oral methylprednisolone the night before the infusion, along with diphenhydramine and acetaminophen on the day of treatment. Fortunately, the patient tolerated the infusion well with no adverse effects or reactions.
Continue to: DISCUSSION
DISCUSSION
Within the NMO spectrum, the MOG antibody is positive in up to 42% of AQP4-seronegative cases.6 MOG is a minor myelin component that is expressed exclusively in the central nervous system on the surface of myelin and oligodendrocyte processes. The role of this glycoprotein is not well understood but is hypothesized to function as a cell surface receptor or cell adhesion molecule.7
Among a cohort of 252 patients from the United Kingdom who tested positive for the MOG-IgG1 antibody, optic neuritis was seen in 55%, while 18% experienced transverse myelitis, and 15% had a history of area postrema syndrome. A brain MRI identified lesions in all areas of the brain including the brain stem, cerebellum, and cerebral hemispheres.8
Risk factors for NMOSD include female gender, Asian and African ethnicities, Epstein Barr virus seropositivity, and tobacco abuse.
Differential diagnosis. Many diseases or conditions that are inflammatory, autoimmune, infectious, or neoplastic can involve the central nervous system and mimic the clinical and radiologic phenotypes of NMOSD-AQP4. They include lupus, SjÖgren’s syndrome, multiple sclerosis, sarcoidosis, acute disseminated encephalomyelitis, HIV, and vitamin B12 deficiency.
Treatment. The standard treatment is intravenous methylprednisolone, 1 g/d for 3 to 5 days followed by a steroid taper. Therapeutic plasma exchange is recommended for refractory cases and in patients with spinal cord demyelination.9-11 Rituximab is the first-line therapy for attack prevention12-15 in NMOSD broadly and may be effective in MOG antibody disease, as well. In an open-label study of patients with NMOSD treated with rituximab, 64% were relapse free at follow-up, which ranged from 12 to 67 months.13 In a long-term study of patients treated with rituximab, 87% maintained a reduced relapse rate and 93% had improvement or stability over a 5-year follow-up.14
Continue to: Our patient
Our patient. After her diagnosis of NMOSD/MOG-antibody disease, our patient’s symptoms progressed to include vertigo, vestibular ataxia, pruritus, left foot drop, lower extremity numbness, and decreased hearing. After the second rituximab infusion her symptoms continued, but over time stabilized and have not worsened. She currently receives gabapentin 300 mg every 8 hours, as needed, for extremity numbness (which has been working well) along with sertraline 100 mg/d for depression.
Subsequent office visits have showed no further weight loss. Based on the current response to the rituximab, her prognosis is undetermined by Neurology as they continue to monitor for progression.
THE TAKEAWAY
Vestibular ataxia, foot drop, pruritus, vertigo, decreased hearing, numbness, and oculomotor dysfunction in the presence of nausea and vomiting should raise suspicion for NMOSD. The presence of AQP4 antibodies along with demyelinating central nervous system lesions, is highly indicative of NMO. The presence of MOG antibodies may indicate NMOSD/MOG-antibody disease. The initial treatment of NMOSD is intravenous methylprednisolone, which can be followed by treatment with rituximab to achieve remission.
CORRESPONDENCE
Daniel Murphy, MD, FAAFP, Department of Family and Community Medicine, Texas Tech University Health Science Center El Paso, 9849 Kenworthy Street, El Paso, Texas 79924; [email protected]
1. Hinson SR, Pittock SJ, Lucchinetti CF, et al. Pathogenic potential of IgG binding to water channel extracellular domain in neuromyelitis optica. Neurology. 2007;69:2221-2231.
2. Ratelade J, Zhang H, Saadoun S, et al. Neuromyelitis optica IgG and natural killer cells Produce NMO lesions in mice without myelin loss. Acta Neuropathol. 2012;123:861-872.
3. Saadoun S, Waters P, Bell BA, et al. Intra-cerebral injection of neuromyelitis optica immunoglobulin G and human complement produces neuromyelitis optica lesions in mice. Brain. 2010;133:349-361.
4. Takahashi T, Fujihara K, Nakashima I, et al. Anti-aquaporin-4 antibody is involved in the pathogenesis of NMO: a study on antibody titer. Brain. 2007;130:1235-1243.
5. Jarius S, Aboul-Enein F, Waters P, et al. Antibody to aquaporin-4 in the long-term course of neuromyelitis optica. Brain. 2008;131:3072-3080.
6. Hamid SHM, Whittam D, Mutch K, et al. What proportion of AQP4-IgG-negative NMO spectrum disorder patients are Mog-IgG positive? A cross sectional study of 132 patients. J Neurol. 2017; 264:2088-2094.
7. Peschl P, Bradi M, Hoftberger R, et al. Myelin oligodendrocyte glycoprotein: deciphering a target in inflammatory demyelinating diseases. Front Immunol. 2017;8:529.
8. Jurynczyk M, Messina S, Woodhall MR, et al. Clinical presentation and prognosis in MOG-antibody disease: a UK study. Brain. 2017;140:3128-3138.
9. Sellner J, Boggild M, Clanet M, et al. EFNS Guidelines on diagnosis and management of neuromyelitis optica. Eur J Neurol. 2010;17:1019-1032.
10. Kleiter I, Gahlen A, Borisow N, et al. Neuromyelitis optica: evaluation of 871 attacks and 1,153 treatment courses. Ann Neurol. 2016;79:206-216.
11. Watanabe S, Nakashima I, Misu T, et al. Therapeutic efficacy of plasma exchange in NMO-IgG-positive patients with neuromyelitis optica. Mult Scler. 2007;13:128-132.
12. Collongues N, Brassat D, Maillart E, et al. Efficacy of rituximab in refractory neuromyelitis optica. Mult Scler. 2016;22:955-959.
13. Collongues N, de Seze J. An update on the evidence for the efficacy and safety of rituximab in the management of neuromyelitis optica. Ther Adv Neurol Disord. 2016;9:180-188.
14. Kim SH, Huh SY, Lee SJ, et al. A 5-year follow-up of rituximab treatment in patients with neuromyelitis optica spectrum disorder. JAMA Neurol. 2013;70:1110-1117.
15. Kim SH, Kim W, Li XF, et al. Repeated treatment with rituximab based on the assessment of peripheral circulating memory B cells in patients with relapsing neuromyelitis optica over 2 years. Arch Neurol. 2011;68:1412-1420.
1. Hinson SR, Pittock SJ, Lucchinetti CF, et al. Pathogenic potential of IgG binding to water channel extracellular domain in neuromyelitis optica. Neurology. 2007;69:2221-2231.
2. Ratelade J, Zhang H, Saadoun S, et al. Neuromyelitis optica IgG and natural killer cells Produce NMO lesions in mice without myelin loss. Acta Neuropathol. 2012;123:861-872.
3. Saadoun S, Waters P, Bell BA, et al. Intra-cerebral injection of neuromyelitis optica immunoglobulin G and human complement produces neuromyelitis optica lesions in mice. Brain. 2010;133:349-361.
4. Takahashi T, Fujihara K, Nakashima I, et al. Anti-aquaporin-4 antibody is involved in the pathogenesis of NMO: a study on antibody titer. Brain. 2007;130:1235-1243.
5. Jarius S, Aboul-Enein F, Waters P, et al. Antibody to aquaporin-4 in the long-term course of neuromyelitis optica. Brain. 2008;131:3072-3080.
6. Hamid SHM, Whittam D, Mutch K, et al. What proportion of AQP4-IgG-negative NMO spectrum disorder patients are Mog-IgG positive? A cross sectional study of 132 patients. J Neurol. 2017; 264:2088-2094.
7. Peschl P, Bradi M, Hoftberger R, et al. Myelin oligodendrocyte glycoprotein: deciphering a target in inflammatory demyelinating diseases. Front Immunol. 2017;8:529.
8. Jurynczyk M, Messina S, Woodhall MR, et al. Clinical presentation and prognosis in MOG-antibody disease: a UK study. Brain. 2017;140:3128-3138.
9. Sellner J, Boggild M, Clanet M, et al. EFNS Guidelines on diagnosis and management of neuromyelitis optica. Eur J Neurol. 2010;17:1019-1032.
10. Kleiter I, Gahlen A, Borisow N, et al. Neuromyelitis optica: evaluation of 871 attacks and 1,153 treatment courses. Ann Neurol. 2016;79:206-216.
11. Watanabe S, Nakashima I, Misu T, et al. Therapeutic efficacy of plasma exchange in NMO-IgG-positive patients with neuromyelitis optica. Mult Scler. 2007;13:128-132.
12. Collongues N, Brassat D, Maillart E, et al. Efficacy of rituximab in refractory neuromyelitis optica. Mult Scler. 2016;22:955-959.
13. Collongues N, de Seze J. An update on the evidence for the efficacy and safety of rituximab in the management of neuromyelitis optica. Ther Adv Neurol Disord. 2016;9:180-188.
14. Kim SH, Huh SY, Lee SJ, et al. A 5-year follow-up of rituximab treatment in patients with neuromyelitis optica spectrum disorder. JAMA Neurol. 2013;70:1110-1117.
15. Kim SH, Kim W, Li XF, et al. Repeated treatment with rituximab based on the assessment of peripheral circulating memory B cells in patients with relapsing neuromyelitis optica over 2 years. Arch Neurol. 2011;68:1412-1420.
Insomnia symptoms increase likelihood of stroke and heart disease
, according to a large cohort study of adults in China. A greater number of insomnia symptoms is associated with increased risk, and this relationship is more evident in younger adults and in adults without hypertension at baseline, researchers reported Nov. 6 in Neurology.
“These results suggest that, if we can target people who are having trouble sleeping with behavioral therapies, it’s possible that we could reduce the number of cases of stroke, heart attack, and other diseases later down the line,” study author Liming Li, MD, professor of epidemiology at Peking University, Beijing, said in a news release.
To clarify the relationships between individual insomnia symptoms, cardiocerebral vascular diseases, and potential effect modifiers, Dr. Li and colleagues analyzed data from the China Kadoorie Biobank Study. For this study, more than 500,000 adults in China aged 30-79 years completed a baseline survey during 2004-2008. The present analysis included data from 487,200 participants who did not have a history of stroke, coronary heart disease, or cancer at baseline.
For the baseline survey, participants answered questions about whether specific insomnia symptoms occurred at least 3 days per week during the past month. The symptoms included difficulty initiating or maintaining sleep (that is, sleep onset latency of 30 minutes or more after going to bed or waking up in the middle of the night); waking too early and being unable to fall back asleep; and trouble functioning during the day because of bad sleep.
The researchers assessed the incidence of cardiocerebral vascular diseases through 2016 by examining disease registries, national health insurance claims databases, and local records. Investigators identified participants with any cardiocerebral vascular disease and assessed the incidence of ischemic heart disease, acute myocardial infarction, hemorrhagic stroke, and ischemic stroke. The researchers followed each participant until the diagnosis of a cardiocerebral vascular disease outcome, death from any cause, loss to follow-up, or Dec. 31, 2016. The researchers used Cox proportional hazard models to estimate hazard ratios for the association between each insomnia symptom and cardiocerebral vascular disease outcomes. They adjusted the models for established and potential confounding factors, including age, income, smoking status, diet, and physical activity.
More than 16% had any insomnia symptom
Of the 487,200 participants, 11.3% had difficulty initiating or maintaining sleep, 10.4% had early morning awakening, and 2.2% had daytime dysfunction attributed to poor sleep. Compared with participants without insomnia symptoms, participants with insomnia symptoms tended to be older and were more likely to be female, not married, and from a rural area. In addition, those with insomnia symptoms were more likely have depression or anxiety symptoms, lower education level, lower household income, and lower body mass index. They also were more likely to have a history of diabetes mellitus. During a median follow-up of 9.6 years, 130,032 cases of cardiocerebral vascular disease occurred, including 40,348 cases of ischemic heart disease and 45,316 cases of stroke.
After adjustment for potential confounders, each insomnia symptom was associated with greater risk of cardiocerebral vascular disease. For difficulty initiating or maintaining sleep, the hazard ratio was 1.09. For early-morning awakening, the HR was 1.07. For daytime dysfunction, the HR was 1.13. Each insomnia symptom was associated with increased risk of ischemic heart disease and ischemic stroke, whereas only difficulty initiating or maintaining sleep was associated with increased risk of acute MI.
In all, 16.4% of participants reported any insomnia symptom; 10% had one symptom, 5.2% had two symptoms, and 1.2% had three symptoms. “Compared with those without any insomnia symptoms, participants with one, two, or three symptoms had a 7%, 10%, or 18% higher risk of total [cardiocerebral vascular disease] incidence, respectively,” the authors wrote. “Our study is the first large-scale cohort study that identified positive dose-response relationships between the number of insomnia symptoms and risks of [cardiocerebral vascular diseases, ischemic heart disease] and stroke incidence.”
Opportunity for intervention
Compared with clinical diagnostic criteria for insomnia, “individual insomnia symptoms are better defined and more feasible to assess with questionnaires in large-scale population studies and clinical practice,” Dr. Li and colleagues wrote. “Moreover, it is reasonable that insomnia symptoms are more modifiable and precisely targetable through behavioral therapies before developing into clinically significant insomnia disorder. Therefore, future clinical trials or community-based intervention studies should be conducted to test whether lifestyle or sleep hygiene interventions for insomnia symptoms can reduce subsequent [cardiocerebral vascular disease] risks.”
The results suggest that efforts aimed at early detection and intervention should include a focus on younger adults and people who do not have high blood pressure, Dr. Li said.
The self-reported insomnia symptoms used in this study have not been fully validated, the investigators noted. The researchers also lacked information about potential confounders, such as shift work and obstructive sleep apnea, that are risk factors for coronary heart disease or stroke and may interfere with insomnia symptoms. In addition, the study did not capture changes in insomnia symptoms over time.
This study was supported by the National Key Research and Development Program of China, the Chinese Ministry of Science and Technology, and the National Natural Science Foundation of China. The China Kadoorie Biobank surveys were supported by grants from the Kadoorie Charitable Foundation and the U.K. Wellcome Trust. The authors had no relevant disclosures.
SOURCE: Zheng B et al. Neurology. 2019 Nov 6. doi: 10.1212/WNL.0000000000008581.
, according to a large cohort study of adults in China. A greater number of insomnia symptoms is associated with increased risk, and this relationship is more evident in younger adults and in adults without hypertension at baseline, researchers reported Nov. 6 in Neurology.
“These results suggest that, if we can target people who are having trouble sleeping with behavioral therapies, it’s possible that we could reduce the number of cases of stroke, heart attack, and other diseases later down the line,” study author Liming Li, MD, professor of epidemiology at Peking University, Beijing, said in a news release.
To clarify the relationships between individual insomnia symptoms, cardiocerebral vascular diseases, and potential effect modifiers, Dr. Li and colleagues analyzed data from the China Kadoorie Biobank Study. For this study, more than 500,000 adults in China aged 30-79 years completed a baseline survey during 2004-2008. The present analysis included data from 487,200 participants who did not have a history of stroke, coronary heart disease, or cancer at baseline.
For the baseline survey, participants answered questions about whether specific insomnia symptoms occurred at least 3 days per week during the past month. The symptoms included difficulty initiating or maintaining sleep (that is, sleep onset latency of 30 minutes or more after going to bed or waking up in the middle of the night); waking too early and being unable to fall back asleep; and trouble functioning during the day because of bad sleep.
The researchers assessed the incidence of cardiocerebral vascular diseases through 2016 by examining disease registries, national health insurance claims databases, and local records. Investigators identified participants with any cardiocerebral vascular disease and assessed the incidence of ischemic heart disease, acute myocardial infarction, hemorrhagic stroke, and ischemic stroke. The researchers followed each participant until the diagnosis of a cardiocerebral vascular disease outcome, death from any cause, loss to follow-up, or Dec. 31, 2016. The researchers used Cox proportional hazard models to estimate hazard ratios for the association between each insomnia symptom and cardiocerebral vascular disease outcomes. They adjusted the models for established and potential confounding factors, including age, income, smoking status, diet, and physical activity.
More than 16% had any insomnia symptom
Of the 487,200 participants, 11.3% had difficulty initiating or maintaining sleep, 10.4% had early morning awakening, and 2.2% had daytime dysfunction attributed to poor sleep. Compared with participants without insomnia symptoms, participants with insomnia symptoms tended to be older and were more likely to be female, not married, and from a rural area. In addition, those with insomnia symptoms were more likely have depression or anxiety symptoms, lower education level, lower household income, and lower body mass index. They also were more likely to have a history of diabetes mellitus. During a median follow-up of 9.6 years, 130,032 cases of cardiocerebral vascular disease occurred, including 40,348 cases of ischemic heart disease and 45,316 cases of stroke.
After adjustment for potential confounders, each insomnia symptom was associated with greater risk of cardiocerebral vascular disease. For difficulty initiating or maintaining sleep, the hazard ratio was 1.09. For early-morning awakening, the HR was 1.07. For daytime dysfunction, the HR was 1.13. Each insomnia symptom was associated with increased risk of ischemic heart disease and ischemic stroke, whereas only difficulty initiating or maintaining sleep was associated with increased risk of acute MI.
In all, 16.4% of participants reported any insomnia symptom; 10% had one symptom, 5.2% had two symptoms, and 1.2% had three symptoms. “Compared with those without any insomnia symptoms, participants with one, two, or three symptoms had a 7%, 10%, or 18% higher risk of total [cardiocerebral vascular disease] incidence, respectively,” the authors wrote. “Our study is the first large-scale cohort study that identified positive dose-response relationships between the number of insomnia symptoms and risks of [cardiocerebral vascular diseases, ischemic heart disease] and stroke incidence.”
Opportunity for intervention
Compared with clinical diagnostic criteria for insomnia, “individual insomnia symptoms are better defined and more feasible to assess with questionnaires in large-scale population studies and clinical practice,” Dr. Li and colleagues wrote. “Moreover, it is reasonable that insomnia symptoms are more modifiable and precisely targetable through behavioral therapies before developing into clinically significant insomnia disorder. Therefore, future clinical trials or community-based intervention studies should be conducted to test whether lifestyle or sleep hygiene interventions for insomnia symptoms can reduce subsequent [cardiocerebral vascular disease] risks.”
The results suggest that efforts aimed at early detection and intervention should include a focus on younger adults and people who do not have high blood pressure, Dr. Li said.
The self-reported insomnia symptoms used in this study have not been fully validated, the investigators noted. The researchers also lacked information about potential confounders, such as shift work and obstructive sleep apnea, that are risk factors for coronary heart disease or stroke and may interfere with insomnia symptoms. In addition, the study did not capture changes in insomnia symptoms over time.
This study was supported by the National Key Research and Development Program of China, the Chinese Ministry of Science and Technology, and the National Natural Science Foundation of China. The China Kadoorie Biobank surveys were supported by grants from the Kadoorie Charitable Foundation and the U.K. Wellcome Trust. The authors had no relevant disclosures.
SOURCE: Zheng B et al. Neurology. 2019 Nov 6. doi: 10.1212/WNL.0000000000008581.
, according to a large cohort study of adults in China. A greater number of insomnia symptoms is associated with increased risk, and this relationship is more evident in younger adults and in adults without hypertension at baseline, researchers reported Nov. 6 in Neurology.
“These results suggest that, if we can target people who are having trouble sleeping with behavioral therapies, it’s possible that we could reduce the number of cases of stroke, heart attack, and other diseases later down the line,” study author Liming Li, MD, professor of epidemiology at Peking University, Beijing, said in a news release.
To clarify the relationships between individual insomnia symptoms, cardiocerebral vascular diseases, and potential effect modifiers, Dr. Li and colleagues analyzed data from the China Kadoorie Biobank Study. For this study, more than 500,000 adults in China aged 30-79 years completed a baseline survey during 2004-2008. The present analysis included data from 487,200 participants who did not have a history of stroke, coronary heart disease, or cancer at baseline.
For the baseline survey, participants answered questions about whether specific insomnia symptoms occurred at least 3 days per week during the past month. The symptoms included difficulty initiating or maintaining sleep (that is, sleep onset latency of 30 minutes or more after going to bed or waking up in the middle of the night); waking too early and being unable to fall back asleep; and trouble functioning during the day because of bad sleep.
The researchers assessed the incidence of cardiocerebral vascular diseases through 2016 by examining disease registries, national health insurance claims databases, and local records. Investigators identified participants with any cardiocerebral vascular disease and assessed the incidence of ischemic heart disease, acute myocardial infarction, hemorrhagic stroke, and ischemic stroke. The researchers followed each participant until the diagnosis of a cardiocerebral vascular disease outcome, death from any cause, loss to follow-up, or Dec. 31, 2016. The researchers used Cox proportional hazard models to estimate hazard ratios for the association between each insomnia symptom and cardiocerebral vascular disease outcomes. They adjusted the models for established and potential confounding factors, including age, income, smoking status, diet, and physical activity.
More than 16% had any insomnia symptom
Of the 487,200 participants, 11.3% had difficulty initiating or maintaining sleep, 10.4% had early morning awakening, and 2.2% had daytime dysfunction attributed to poor sleep. Compared with participants without insomnia symptoms, participants with insomnia symptoms tended to be older and were more likely to be female, not married, and from a rural area. In addition, those with insomnia symptoms were more likely have depression or anxiety symptoms, lower education level, lower household income, and lower body mass index. They also were more likely to have a history of diabetes mellitus. During a median follow-up of 9.6 years, 130,032 cases of cardiocerebral vascular disease occurred, including 40,348 cases of ischemic heart disease and 45,316 cases of stroke.
After adjustment for potential confounders, each insomnia symptom was associated with greater risk of cardiocerebral vascular disease. For difficulty initiating or maintaining sleep, the hazard ratio was 1.09. For early-morning awakening, the HR was 1.07. For daytime dysfunction, the HR was 1.13. Each insomnia symptom was associated with increased risk of ischemic heart disease and ischemic stroke, whereas only difficulty initiating or maintaining sleep was associated with increased risk of acute MI.
In all, 16.4% of participants reported any insomnia symptom; 10% had one symptom, 5.2% had two symptoms, and 1.2% had three symptoms. “Compared with those without any insomnia symptoms, participants with one, two, or three symptoms had a 7%, 10%, or 18% higher risk of total [cardiocerebral vascular disease] incidence, respectively,” the authors wrote. “Our study is the first large-scale cohort study that identified positive dose-response relationships between the number of insomnia symptoms and risks of [cardiocerebral vascular diseases, ischemic heart disease] and stroke incidence.”
Opportunity for intervention
Compared with clinical diagnostic criteria for insomnia, “individual insomnia symptoms are better defined and more feasible to assess with questionnaires in large-scale population studies and clinical practice,” Dr. Li and colleagues wrote. “Moreover, it is reasonable that insomnia symptoms are more modifiable and precisely targetable through behavioral therapies before developing into clinically significant insomnia disorder. Therefore, future clinical trials or community-based intervention studies should be conducted to test whether lifestyle or sleep hygiene interventions for insomnia symptoms can reduce subsequent [cardiocerebral vascular disease] risks.”
The results suggest that efforts aimed at early detection and intervention should include a focus on younger adults and people who do not have high blood pressure, Dr. Li said.
The self-reported insomnia symptoms used in this study have not been fully validated, the investigators noted. The researchers also lacked information about potential confounders, such as shift work and obstructive sleep apnea, that are risk factors for coronary heart disease or stroke and may interfere with insomnia symptoms. In addition, the study did not capture changes in insomnia symptoms over time.
This study was supported by the National Key Research and Development Program of China, the Chinese Ministry of Science and Technology, and the National Natural Science Foundation of China. The China Kadoorie Biobank surveys were supported by grants from the Kadoorie Charitable Foundation and the U.K. Wellcome Trust. The authors had no relevant disclosures.
SOURCE: Zheng B et al. Neurology. 2019 Nov 6. doi: 10.1212/WNL.0000000000008581.
FROM NEUROLOGY
Key clinical point: The presence of insomnia symptoms increases the likelihood of cardiovascular or cerebrovascular disease during approximately 10 years of follow-up.
Major finding: After adjustment for potential confounders, each insomnia symptom was associated with greater risk of cardiocerebral vascular disease. For difficulty initiating or maintaining sleep, the hazard ratio was 1.09. For early-morning awakening, the HR was 1.07. For daytime dysfunction, the HR was 1.13.
Study details: An analysis of data from 487,200 adults in China aged 30-79 years who completed a baseline survey during 2004-2008 and were followed through 2016.
Disclosures: This study was supported by the National Key Research and Development Program of China, the Chinese Ministry of Science and Technology, and the National Natural Science Foundation of China. The China Kadoorie Biobank surveys were supported by grants from the Kadoorie Charitable Foundation and the U.K. Wellcome Trust. The authors had no relevant disclosures.
Source: Zheng B et al. Neurology. 2019 Nov 6. doi: 10.1212/WNL.0000000000008581.
New score predicts benefits of prolonged cardiac monitoring for TIA, stroke patients
Background: Identifying paroxysmal atrial fibrillation (AFib) as the etiology of a transient ischemic attack (TIA) or stroke has implications for treatment as well as secondary prevention. Currently, there is not a universal, practical way to help determine which patients would benefit from prolonged cardiac monitoring to establish the diagnosis of AFib.
Study design: Logistic regression analysis of three prospective multicenter trials examining TIA and stroke patients who received Holter-ECG monitoring.
Setting: Patients who presented with TIA or stroke in Central Europe.
Synopsis: Using data from 1,556 patients, the authors identified age and NIH stroke scale score as being predictive of which patients were at highest risk for AFib detection within 72 hours of Holter-ECG monitor initiation. The authors developed a formula, titled AS5F; this formula scores each year of age as 0.76 points and then an NIH stroke scale score of 5 or less as 9 points or greater than 5 as 21 points. The authors found that the high-risk group (defined as those with AS5F scores of 67.5 or higher) had a predicted risk of 5.2%-40.8%, with a number needed to screen of 3. Given that a majority of the European patients included in the study were white, generalizability to other populations is unclear.
Bottom line: AS5F score may be able to predict those TIA and stroke patients who are most likely to be diagnosed with AFib with 72-hour cardiac monitoring.
Citation: Uphaus T et al. Development and validation of a score to detect paroxysmal atrial fibrillation after stroke. Neurology. 2019 Jan 8. doi. 10.1212/WNL.0000000000006727.
Dr. Phillips is a hospitalist at Beth Israel Deaconess Medical Center and instructor in medicine at Harvard Medical School.
Background: Identifying paroxysmal atrial fibrillation (AFib) as the etiology of a transient ischemic attack (TIA) or stroke has implications for treatment as well as secondary prevention. Currently, there is not a universal, practical way to help determine which patients would benefit from prolonged cardiac monitoring to establish the diagnosis of AFib.
Study design: Logistic regression analysis of three prospective multicenter trials examining TIA and stroke patients who received Holter-ECG monitoring.
Setting: Patients who presented with TIA or stroke in Central Europe.
Synopsis: Using data from 1,556 patients, the authors identified age and NIH stroke scale score as being predictive of which patients were at highest risk for AFib detection within 72 hours of Holter-ECG monitor initiation. The authors developed a formula, titled AS5F; this formula scores each year of age as 0.76 points and then an NIH stroke scale score of 5 or less as 9 points or greater than 5 as 21 points. The authors found that the high-risk group (defined as those with AS5F scores of 67.5 or higher) had a predicted risk of 5.2%-40.8%, with a number needed to screen of 3. Given that a majority of the European patients included in the study were white, generalizability to other populations is unclear.
Bottom line: AS5F score may be able to predict those TIA and stroke patients who are most likely to be diagnosed with AFib with 72-hour cardiac monitoring.
Citation: Uphaus T et al. Development and validation of a score to detect paroxysmal atrial fibrillation after stroke. Neurology. 2019 Jan 8. doi. 10.1212/WNL.0000000000006727.
Dr. Phillips is a hospitalist at Beth Israel Deaconess Medical Center and instructor in medicine at Harvard Medical School.
Background: Identifying paroxysmal atrial fibrillation (AFib) as the etiology of a transient ischemic attack (TIA) or stroke has implications for treatment as well as secondary prevention. Currently, there is not a universal, practical way to help determine which patients would benefit from prolonged cardiac monitoring to establish the diagnosis of AFib.
Study design: Logistic regression analysis of three prospective multicenter trials examining TIA and stroke patients who received Holter-ECG monitoring.
Setting: Patients who presented with TIA or stroke in Central Europe.
Synopsis: Using data from 1,556 patients, the authors identified age and NIH stroke scale score as being predictive of which patients were at highest risk for AFib detection within 72 hours of Holter-ECG monitor initiation. The authors developed a formula, titled AS5F; this formula scores each year of age as 0.76 points and then an NIH stroke scale score of 5 or less as 9 points or greater than 5 as 21 points. The authors found that the high-risk group (defined as those with AS5F scores of 67.5 or higher) had a predicted risk of 5.2%-40.8%, with a number needed to screen of 3. Given that a majority of the European patients included in the study were white, generalizability to other populations is unclear.
Bottom line: AS5F score may be able to predict those TIA and stroke patients who are most likely to be diagnosed with AFib with 72-hour cardiac monitoring.
Citation: Uphaus T et al. Development and validation of a score to detect paroxysmal atrial fibrillation after stroke. Neurology. 2019 Jan 8. doi. 10.1212/WNL.0000000000006727.
Dr. Phillips is a hospitalist at Beth Israel Deaconess Medical Center and instructor in medicine at Harvard Medical School.
Click for Credit: Long-term antibiotics & stroke, CHD; Postvaccination seizures; more
Here are 5 articles from the November issue of Clinician Reviews (individual articles are valid for one year from date of publication—expiration dates below):
1. Poor response to statins hikes risk of cardiovascular events
To take the posttest, go to: https://bit.ly/2MVHlDR
Expires April 17, 2020
2. Postvaccination febrile seizures are no more severe than other febrile seizures
To take the posttest, go to: https://bit.ly/2VUJzaE
Expires April 19, 2020
3. Hydroxychloroquine adherence in SLE: worse than you thought
To take the posttest, go to: https://bit.ly/2oT00Z9
Expires April 22, 2020
4. Long-term antibiotic use may heighten stroke, CHD risk
To take the posttest, go to: https://bit.ly/2OUUVu5
Expires April 28, 2020
5. Knowledge gaps about long-term osteoporosis drug therapy benefits, risks remain large
To take the posttest, go to: https://bit.ly/2Msgqkb
Expires May 1, 2020
Here are 5 articles from the November issue of Clinician Reviews (individual articles are valid for one year from date of publication—expiration dates below):
1. Poor response to statins hikes risk of cardiovascular events
To take the posttest, go to: https://bit.ly/2MVHlDR
Expires April 17, 2020
2. Postvaccination febrile seizures are no more severe than other febrile seizures
To take the posttest, go to: https://bit.ly/2VUJzaE
Expires April 19, 2020
3. Hydroxychloroquine adherence in SLE: worse than you thought
To take the posttest, go to: https://bit.ly/2oT00Z9
Expires April 22, 2020
4. Long-term antibiotic use may heighten stroke, CHD risk
To take the posttest, go to: https://bit.ly/2OUUVu5
Expires April 28, 2020
5. Knowledge gaps about long-term osteoporosis drug therapy benefits, risks remain large
To take the posttest, go to: https://bit.ly/2Msgqkb
Expires May 1, 2020
Here are 5 articles from the November issue of Clinician Reviews (individual articles are valid for one year from date of publication—expiration dates below):
1. Poor response to statins hikes risk of cardiovascular events
To take the posttest, go to: https://bit.ly/2MVHlDR
Expires April 17, 2020
2. Postvaccination febrile seizures are no more severe than other febrile seizures
To take the posttest, go to: https://bit.ly/2VUJzaE
Expires April 19, 2020
3. Hydroxychloroquine adherence in SLE: worse than you thought
To take the posttest, go to: https://bit.ly/2oT00Z9
Expires April 22, 2020
4. Long-term antibiotic use may heighten stroke, CHD risk
To take the posttest, go to: https://bit.ly/2OUUVu5
Expires April 28, 2020
5. Knowledge gaps about long-term osteoporosis drug therapy benefits, risks remain large
To take the posttest, go to: https://bit.ly/2Msgqkb
Expires May 1, 2020
Brain abscess with lung infection? Think Nocardia
ST. LOUIS – according to University of California, San Francisco, investigators.
Nocardia – an ubiquitous gram-positive rod normally found in standing water, decaying plants, and soil, that can cause problems when it is inhaled as dust or introduced through a nick in the skin – is an underappreciated cause of brain abscess that is not covered by standard empiric therapy targeting the more common causes: Staphylococcus and Streptococcus bacteria, said senior investigator Megan Richie, MD, an assistant neurology professor at UCSF.
“Patients that have a lung infection with a new brain abscess should be started on empiric therapy not just for pyogenic organisms, but also for Nocardia pending biopsy and operative culture data, especially given that empiric therapy of high-dose Bactrim for Nocardia is relatively benign,” she said at the annual meeting of the American Neurological Association.
The advice comes from a comparison of 14 Nocardia cases with 42 randomly selected Staph/Strep cases in a university radiologic database. Nine Nocardia cases were confirmed by operative specimen culture, the rest by lung, blood, or other tissue cultures.
Dr. Richie and colleagues suspected an association with lung infection, which has been reported anecdotally in the literature. The researchers wanted to take a quantitative look to see if it held up statistically after pushback on a brain abscess patient with a lung infection. “We were concerned this patient had Nocardia, but it took quite some time to convince other doctors that we really needed to start [Bactrim]. The patient was not immunocompromised and the infectious disease team said ‘Nocardia brain infections don’t happen in immunocompetent patients,’” Dr. Richie said,
The man did, however, turn out to have Nocardia, and of the 14 cases in the series, four patients (29%) were not immunosuppressed. “I think this would surprise [physicians] who have a little bit less experience with this organism,” Dr. Richie said.Patients with a Nocardia brain abscess were far more likely to have a concomitant lung infection (86% vs. 2%; odds ratio, 246; 95% confidence interval, 21-2953; P less than .0001). Staph/Strep brain abscess patients were more likely to have concomitant ear or sinus infections (40% versus 0%; P = .005). Immunosuppression did turn out to be more common in the Nocardia group, as well (71% vs. 19%; OR, 11; 95% CI, 3-43; P = .001), as did diabetes (36% vs. 10%; P = .03).
Nocardia patients were older (median age, 61 yrs vs. 46 yrs: P = .01) and more likely to be Hispanic (36% vs. 10%; P = .04). There were no differences in sex; neurosurgery history; intravenous drug use; or endocarditis.
On imaging, Nocardia brain abscesses were poorly circumscribed and tended to have multiple lobes, “often two in a figure-eight pattern,” Dr. Richie said. Nocardia diagnosis took longer (median, 7 vs. 4 days; P = .04), “which makes sense because it is a harder diagnosis to make,” she said.
Operative specimen culture was the most potent diagnostic tool. Blood cultures were positive in just one Nocardia patient and a few controls.
There was no external funding, and the investigators did not have any relevant disclosures.
ST. LOUIS – according to University of California, San Francisco, investigators.
Nocardia – an ubiquitous gram-positive rod normally found in standing water, decaying plants, and soil, that can cause problems when it is inhaled as dust or introduced through a nick in the skin – is an underappreciated cause of brain abscess that is not covered by standard empiric therapy targeting the more common causes: Staphylococcus and Streptococcus bacteria, said senior investigator Megan Richie, MD, an assistant neurology professor at UCSF.
“Patients that have a lung infection with a new brain abscess should be started on empiric therapy not just for pyogenic organisms, but also for Nocardia pending biopsy and operative culture data, especially given that empiric therapy of high-dose Bactrim for Nocardia is relatively benign,” she said at the annual meeting of the American Neurological Association.
The advice comes from a comparison of 14 Nocardia cases with 42 randomly selected Staph/Strep cases in a university radiologic database. Nine Nocardia cases were confirmed by operative specimen culture, the rest by lung, blood, or other tissue cultures.
Dr. Richie and colleagues suspected an association with lung infection, which has been reported anecdotally in the literature. The researchers wanted to take a quantitative look to see if it held up statistically after pushback on a brain abscess patient with a lung infection. “We were concerned this patient had Nocardia, but it took quite some time to convince other doctors that we really needed to start [Bactrim]. The patient was not immunocompromised and the infectious disease team said ‘Nocardia brain infections don’t happen in immunocompetent patients,’” Dr. Richie said,
The man did, however, turn out to have Nocardia, and of the 14 cases in the series, four patients (29%) were not immunosuppressed. “I think this would surprise [physicians] who have a little bit less experience with this organism,” Dr. Richie said.Patients with a Nocardia brain abscess were far more likely to have a concomitant lung infection (86% vs. 2%; odds ratio, 246; 95% confidence interval, 21-2953; P less than .0001). Staph/Strep brain abscess patients were more likely to have concomitant ear or sinus infections (40% versus 0%; P = .005). Immunosuppression did turn out to be more common in the Nocardia group, as well (71% vs. 19%; OR, 11; 95% CI, 3-43; P = .001), as did diabetes (36% vs. 10%; P = .03).
Nocardia patients were older (median age, 61 yrs vs. 46 yrs: P = .01) and more likely to be Hispanic (36% vs. 10%; P = .04). There were no differences in sex; neurosurgery history; intravenous drug use; or endocarditis.
On imaging, Nocardia brain abscesses were poorly circumscribed and tended to have multiple lobes, “often two in a figure-eight pattern,” Dr. Richie said. Nocardia diagnosis took longer (median, 7 vs. 4 days; P = .04), “which makes sense because it is a harder diagnosis to make,” she said.
Operative specimen culture was the most potent diagnostic tool. Blood cultures were positive in just one Nocardia patient and a few controls.
There was no external funding, and the investigators did not have any relevant disclosures.
ST. LOUIS – according to University of California, San Francisco, investigators.
Nocardia – an ubiquitous gram-positive rod normally found in standing water, decaying plants, and soil, that can cause problems when it is inhaled as dust or introduced through a nick in the skin – is an underappreciated cause of brain abscess that is not covered by standard empiric therapy targeting the more common causes: Staphylococcus and Streptococcus bacteria, said senior investigator Megan Richie, MD, an assistant neurology professor at UCSF.
“Patients that have a lung infection with a new brain abscess should be started on empiric therapy not just for pyogenic organisms, but also for Nocardia pending biopsy and operative culture data, especially given that empiric therapy of high-dose Bactrim for Nocardia is relatively benign,” she said at the annual meeting of the American Neurological Association.
The advice comes from a comparison of 14 Nocardia cases with 42 randomly selected Staph/Strep cases in a university radiologic database. Nine Nocardia cases were confirmed by operative specimen culture, the rest by lung, blood, or other tissue cultures.
Dr. Richie and colleagues suspected an association with lung infection, which has been reported anecdotally in the literature. The researchers wanted to take a quantitative look to see if it held up statistically after pushback on a brain abscess patient with a lung infection. “We were concerned this patient had Nocardia, but it took quite some time to convince other doctors that we really needed to start [Bactrim]. The patient was not immunocompromised and the infectious disease team said ‘Nocardia brain infections don’t happen in immunocompetent patients,’” Dr. Richie said,
The man did, however, turn out to have Nocardia, and of the 14 cases in the series, four patients (29%) were not immunosuppressed. “I think this would surprise [physicians] who have a little bit less experience with this organism,” Dr. Richie said.Patients with a Nocardia brain abscess were far more likely to have a concomitant lung infection (86% vs. 2%; odds ratio, 246; 95% confidence interval, 21-2953; P less than .0001). Staph/Strep brain abscess patients were more likely to have concomitant ear or sinus infections (40% versus 0%; P = .005). Immunosuppression did turn out to be more common in the Nocardia group, as well (71% vs. 19%; OR, 11; 95% CI, 3-43; P = .001), as did diabetes (36% vs. 10%; P = .03).
Nocardia patients were older (median age, 61 yrs vs. 46 yrs: P = .01) and more likely to be Hispanic (36% vs. 10%; P = .04). There were no differences in sex; neurosurgery history; intravenous drug use; or endocarditis.
On imaging, Nocardia brain abscesses were poorly circumscribed and tended to have multiple lobes, “often two in a figure-eight pattern,” Dr. Richie said. Nocardia diagnosis took longer (median, 7 vs. 4 days; P = .04), “which makes sense because it is a harder diagnosis to make,” she said.
Operative specimen culture was the most potent diagnostic tool. Blood cultures were positive in just one Nocardia patient and a few controls.
There was no external funding, and the investigators did not have any relevant disclosures.
REPORTING FROM ANA 2019