User login
Troubling trend as both diabetes types rise among U.S. youth
The incidence of type 1 and type 2 diabetes continues to rise among children and adolescents in the United States, new data from the SEARCH for Diabetes in Youth study show.
The SEARCH data demonstrate an increase in the youth population aged 0-19 diagnosed with type 1 or type 2 diabetes in five representative U.S. centers. Between 2002 and 2018, the annual incidence rose by about 2% per year for type 1 diabetes and 5% per year for type 2 diabetes. The rates of increase for both types were greater among non-White than White youth.
These increases “will result in an expanding population of young adults at risk of developing early complications of diabetes whose health care needs will exceed those of their peers,” write Lynne E. Wagenknecht, DrPH, of Wake Forest University School of Medicine, Winston-Salem, N.C., and colleagues in their article, recently published in The Lancet Diabetes & Endocrinology.
In an accompanying editorial, Jonathan E. Shaw, MD, and Dianna J. Magliano, PhD, both at the Baker Heart and Diabetes Institute, Melbourne, write that one of the most “concerning findings” was a 7%-9% annual increase in the incidence of type 2 diabetes among Hispanic, Asian, and Pacific Islander populations.
“This is a health care crisis in the making. ...Youth and young-adult-onset type 2 diabetes are growing problems leading to poor outcomes and to widening social inequality, adversely affecting a population that might already be disadvantaged. Better information about its natural history, prevention, and management is urgently needed,” they write.
Upward trends in both diabetes types
Overall, 18,169 children and adolescents with type 1 diabetes and 5,293 with type 2 diabetes were identified over the 17-year study period in SEARCH. After adjustment for age, sex, and race/ethnicity, there was a significant increase in type 1 diabetes incidence from 19.5 cases/100,000 population in 2002-2003 to 22.2/100,000 in 2017-2018, a 2.02% annual increase.
The upward trend was even greater for type 2 diabetes, from 9.0/100,000 in 2002-2003 to 17.9/100,000 in 2017-2018, a 5.31% annual increase.
The annual rate of increase in type 1 diabetes was highest among Asian/Pacific Islander youth (4.84%), followed by Hispanic (4.14%) and Black youth (2.93%): All significantly rose over the 17 years.
For type 2 diabetes, significant annual rates of increase were also highest for Asian/Pacific Islanders (8.92%), followed by Hispanic (7.17%) and Black youth (5.99%).
Among youth aged 15-19 years, the overall incidence of type 2 diabetes exceeded that of type 1 diabetes (19.7 vs. 14.6/100,000).
The incidence of type 2 diabetes may be rising because of increased rates of obesity, as well as increased screening of at-risk youth, the authors say.
And, the editorialists note, obesity is also a risk factor for type 1 diabetes.
Peak incidence of type 1 diabetes occurred at age 10 years, while for type 2 diabetes, the peak was at 16 years. There were also seasonal peaks, occurring in January for type 1 diabetes and in August for type 2 diabetes. Those seasonal patterns have been previously reported; they are possibly because of increased viral infections and decreased sun exposure for the former, and increased physical exams in preparation for school in the latter, the authors speculate.
Dr. Shaw and Dr. Magliano note that the reduced incidence after age 16 years “might simply reflect a failure to diagnose,” suggesting that there will likely be an upturn in incidence in the subsequent decade.
The editorialists also point out: “Not only does the long duration of diabetes that youth-onset leads to cause a large burden of fatal and nonfatal complications, but it magnifies intergenerational effects.”
“When type 2 diabetes is already present before pregnancy, birth outcomes are worse, and the long-term metabolic health of the offspring is adversely affected. This does not bode well for the epidemic of diabetes and its complications.”
The study was funded by the Centers for Disease Control and Prevention and National Institutes of Health. The authors and Dr. Magliano have reported no relevant financial relationships. Dr. Shaw has reported receiving honoraria for lectures and for advisory boards and grants from AstraZeneca, Boehringer Ingelheim, Pfizer, Eli Lilly, Sanofi, Roche, Mylan, and Zuellig Pharma.
A version of this article originally appeared on Medscape.com.
The incidence of type 1 and type 2 diabetes continues to rise among children and adolescents in the United States, new data from the SEARCH for Diabetes in Youth study show.
The SEARCH data demonstrate an increase in the youth population aged 0-19 diagnosed with type 1 or type 2 diabetes in five representative U.S. centers. Between 2002 and 2018, the annual incidence rose by about 2% per year for type 1 diabetes and 5% per year for type 2 diabetes. The rates of increase for both types were greater among non-White than White youth.
These increases “will result in an expanding population of young adults at risk of developing early complications of diabetes whose health care needs will exceed those of their peers,” write Lynne E. Wagenknecht, DrPH, of Wake Forest University School of Medicine, Winston-Salem, N.C., and colleagues in their article, recently published in The Lancet Diabetes & Endocrinology.
In an accompanying editorial, Jonathan E. Shaw, MD, and Dianna J. Magliano, PhD, both at the Baker Heart and Diabetes Institute, Melbourne, write that one of the most “concerning findings” was a 7%-9% annual increase in the incidence of type 2 diabetes among Hispanic, Asian, and Pacific Islander populations.
“This is a health care crisis in the making. ...Youth and young-adult-onset type 2 diabetes are growing problems leading to poor outcomes and to widening social inequality, adversely affecting a population that might already be disadvantaged. Better information about its natural history, prevention, and management is urgently needed,” they write.
Upward trends in both diabetes types
Overall, 18,169 children and adolescents with type 1 diabetes and 5,293 with type 2 diabetes were identified over the 17-year study period in SEARCH. After adjustment for age, sex, and race/ethnicity, there was a significant increase in type 1 diabetes incidence from 19.5 cases/100,000 population in 2002-2003 to 22.2/100,000 in 2017-2018, a 2.02% annual increase.
The upward trend was even greater for type 2 diabetes, from 9.0/100,000 in 2002-2003 to 17.9/100,000 in 2017-2018, a 5.31% annual increase.
The annual rate of increase in type 1 diabetes was highest among Asian/Pacific Islander youth (4.84%), followed by Hispanic (4.14%) and Black youth (2.93%): All significantly rose over the 17 years.
For type 2 diabetes, significant annual rates of increase were also highest for Asian/Pacific Islanders (8.92%), followed by Hispanic (7.17%) and Black youth (5.99%).
Among youth aged 15-19 years, the overall incidence of type 2 diabetes exceeded that of type 1 diabetes (19.7 vs. 14.6/100,000).
The incidence of type 2 diabetes may be rising because of increased rates of obesity, as well as increased screening of at-risk youth, the authors say.
And, the editorialists note, obesity is also a risk factor for type 1 diabetes.
Peak incidence of type 1 diabetes occurred at age 10 years, while for type 2 diabetes, the peak was at 16 years. There were also seasonal peaks, occurring in January for type 1 diabetes and in August for type 2 diabetes. Those seasonal patterns have been previously reported; they are possibly because of increased viral infections and decreased sun exposure for the former, and increased physical exams in preparation for school in the latter, the authors speculate.
Dr. Shaw and Dr. Magliano note that the reduced incidence after age 16 years “might simply reflect a failure to diagnose,” suggesting that there will likely be an upturn in incidence in the subsequent decade.
The editorialists also point out: “Not only does the long duration of diabetes that youth-onset leads to cause a large burden of fatal and nonfatal complications, but it magnifies intergenerational effects.”
“When type 2 diabetes is already present before pregnancy, birth outcomes are worse, and the long-term metabolic health of the offspring is adversely affected. This does not bode well for the epidemic of diabetes and its complications.”
The study was funded by the Centers for Disease Control and Prevention and National Institutes of Health. The authors and Dr. Magliano have reported no relevant financial relationships. Dr. Shaw has reported receiving honoraria for lectures and for advisory boards and grants from AstraZeneca, Boehringer Ingelheim, Pfizer, Eli Lilly, Sanofi, Roche, Mylan, and Zuellig Pharma.
A version of this article originally appeared on Medscape.com.
The incidence of type 1 and type 2 diabetes continues to rise among children and adolescents in the United States, new data from the SEARCH for Diabetes in Youth study show.
The SEARCH data demonstrate an increase in the youth population aged 0-19 diagnosed with type 1 or type 2 diabetes in five representative U.S. centers. Between 2002 and 2018, the annual incidence rose by about 2% per year for type 1 diabetes and 5% per year for type 2 diabetes. The rates of increase for both types were greater among non-White than White youth.
These increases “will result in an expanding population of young adults at risk of developing early complications of diabetes whose health care needs will exceed those of their peers,” write Lynne E. Wagenknecht, DrPH, of Wake Forest University School of Medicine, Winston-Salem, N.C., and colleagues in their article, recently published in The Lancet Diabetes & Endocrinology.
In an accompanying editorial, Jonathan E. Shaw, MD, and Dianna J. Magliano, PhD, both at the Baker Heart and Diabetes Institute, Melbourne, write that one of the most “concerning findings” was a 7%-9% annual increase in the incidence of type 2 diabetes among Hispanic, Asian, and Pacific Islander populations.
“This is a health care crisis in the making. ...Youth and young-adult-onset type 2 diabetes are growing problems leading to poor outcomes and to widening social inequality, adversely affecting a population that might already be disadvantaged. Better information about its natural history, prevention, and management is urgently needed,” they write.
Upward trends in both diabetes types
Overall, 18,169 children and adolescents with type 1 diabetes and 5,293 with type 2 diabetes were identified over the 17-year study period in SEARCH. After adjustment for age, sex, and race/ethnicity, there was a significant increase in type 1 diabetes incidence from 19.5 cases/100,000 population in 2002-2003 to 22.2/100,000 in 2017-2018, a 2.02% annual increase.
The upward trend was even greater for type 2 diabetes, from 9.0/100,000 in 2002-2003 to 17.9/100,000 in 2017-2018, a 5.31% annual increase.
The annual rate of increase in type 1 diabetes was highest among Asian/Pacific Islander youth (4.84%), followed by Hispanic (4.14%) and Black youth (2.93%): All significantly rose over the 17 years.
For type 2 diabetes, significant annual rates of increase were also highest for Asian/Pacific Islanders (8.92%), followed by Hispanic (7.17%) and Black youth (5.99%).
Among youth aged 15-19 years, the overall incidence of type 2 diabetes exceeded that of type 1 diabetes (19.7 vs. 14.6/100,000).
The incidence of type 2 diabetes may be rising because of increased rates of obesity, as well as increased screening of at-risk youth, the authors say.
And, the editorialists note, obesity is also a risk factor for type 1 diabetes.
Peak incidence of type 1 diabetes occurred at age 10 years, while for type 2 diabetes, the peak was at 16 years. There were also seasonal peaks, occurring in January for type 1 diabetes and in August for type 2 diabetes. Those seasonal patterns have been previously reported; they are possibly because of increased viral infections and decreased sun exposure for the former, and increased physical exams in preparation for school in the latter, the authors speculate.
Dr. Shaw and Dr. Magliano note that the reduced incidence after age 16 years “might simply reflect a failure to diagnose,” suggesting that there will likely be an upturn in incidence in the subsequent decade.
The editorialists also point out: “Not only does the long duration of diabetes that youth-onset leads to cause a large burden of fatal and nonfatal complications, but it magnifies intergenerational effects.”
“When type 2 diabetes is already present before pregnancy, birth outcomes are worse, and the long-term metabolic health of the offspring is adversely affected. This does not bode well for the epidemic of diabetes and its complications.”
The study was funded by the Centers for Disease Control and Prevention and National Institutes of Health. The authors and Dr. Magliano have reported no relevant financial relationships. Dr. Shaw has reported receiving honoraria for lectures and for advisory boards and grants from AstraZeneca, Boehringer Ingelheim, Pfizer, Eli Lilly, Sanofi, Roche, Mylan, and Zuellig Pharma.
A version of this article originally appeared on Medscape.com.
FROM THE LANCET DIABETES & ENDOCRINOLOGY
New insight into preventing antipsychotic-induced weight gain
In the first dose-response meta-analysis focusing on antipsychotic-induced weight gain, researchers provide data on the trajectory of this risk associated with individual agents.
Investigators analyzed 52 randomized controlled trials (RCTs) encompassing more than 22,500 participants with schizophrenia treated with antipsychotics. They found that, with the exception of aripiprazole long-acting injectable (LAI), all of the other antipsychotics has significant dose-response effect on weight gain. Furthermore, weight gain occurred with some antipsychotics even at relatively low doses.
“We found significant dose-response associations for weight and metabolic variables, with a unique signature for each antipsychotic,” write the investigators, led by Michel Sabé, MD, of the division of adult psychiatry, department of psychiatry, Geneva University Hospitals.
“Despite several limitations, including the limited number of available studies, our results may provide useful information for preventing weight gain and metabolic disturbances by adapting antipsychotic doses,” they add.
The study was published online in The Journal of Clinical Psychiatry.
Balancing risks and benefits
Antipsychotics are first-line therapy for schizophrenia and are associated with weight gain, lipid disturbances, and glucose dysregulation – especially second-generation antipsychotics (SGAs), which can lead to obesity, type 2 diabetes, and metabolic syndrome.
Given that people with schizophrenia also tend to have lifestyle-related cardiovascular risk factors, it’s important to find “a balance between beneficial and adverse effects of antipsychotics,” the investigators note
The question of whether weight gain and metabolic dysregulation are dose-dependent “remains controversial.” The effect of specific SGAs on weight gain has been investigated, but only one study has been conducted using a dose-response meta-analysis, and that study did not address metabolic disturbance.
The investigators conducted a systematic review and a dose-response meta-analysis of fixed-dose randomized controlled trials (RCTs) investigating antipsychotic-induced weight gain and metabolic disturbance in adults with acute schizophrenia.
To be included in the analysis, RCTs had to focus on adult patients with schizophrenia or related disorders and include a placebo as a comparator to the drug.
Studies involved only short-term administration of antipsychotics (2-13 weeks) rather than maintenance therapy.
The mean (SD) change in weight (body weight and/or body mass index) between baseline and the study endpoint constituted the primary outcome, with secondary outcomes including changes in metabolic parameters.
The researchers characterized the dose-response relationship using a nonlinear restricted cubic spline model, with three “knots” located at the 10th, 50th, and 90th percentiles of overall dose distribution.
They also calculated dose-response curves and estimated 50% and 95% effective doses (ED50 and ED95, respectively), extracted from the estimated dose-response curves for each antipsychotic.
The researchers then calculated the weight gain at each effective dose (ED50 and ED95) in milligrams and the weight gain corresponding to the ED95 value in kilograms.
Shared decision-making
Of 6,812 citations, the researchers selected 52 RCTs that met inclusion criteria (n = 22,588 participants, with 16,311 receiving antipsychotics and 6,277 receiving placebo; mean age, 38.5 years, 69.2% male). The studies were conducted between1996 and 2021.
The risk for bias in most studies was “low,” although 21% of the studies “presented a high risk.”
With the exception of aripiprazole LAI, all of the other antipsychotics had a “significant dose-response” association with weight.
For example, oral aripiprazole exhibited a significant dose-response association for weight, but there was no significant association found for aripiprazole LAI (c2 = 8.744; P = .0126 vs. c2 = 3.107; P = .2115). However, both curves were still ascending at maximum doses, the authors note.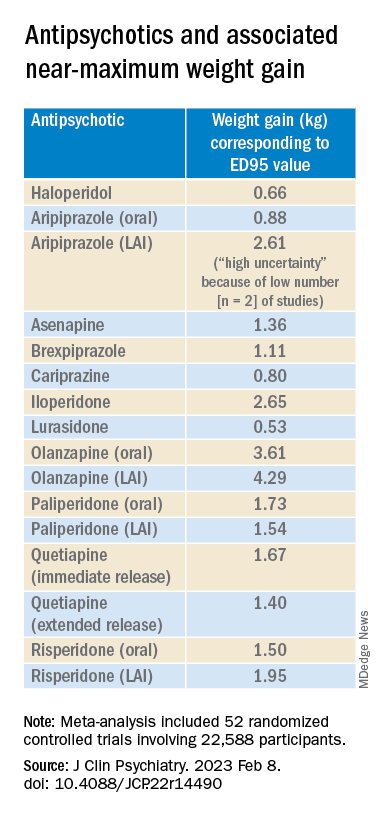
Metabolically neutral
Antipsychotics with a decreasing or quasi-parabolic dose-response curve for weight included brexpiprazole, cariprazine, haloperidol, lurasidone, and quetiapine ER: for these antipsychotics, the ED95 weight gain ranged from 0.53 kg to 1.40 kg.
These antipsychotics “reach their weight gain ED95 at relatively low median effective doses, and higher doses, which mostly correspond to near-maximum effective doses, may even be associated with less weight gain,” the authors note.
In addition, only doses higher than the near-maximum effective dose of brexpiprazole were associated with a small increase in total cholesterol. And cariprazine presented “significantly decreasing curves” at higher doses for LDL cholesterol.
With the exception of quetiapine, this group of medications might be regarded as “metabolically neutral” in terms of weight gain and metabolic disturbances.
Antipsychotics with a plateau-shaped curve were asenapine, iloperidone, paliperidone LAI, quetiapine IR, and risperidone, with a weight gain ED95 ranging from 1.36 to 2.65 kg.
Aripiprazole and olanzapine (oral and LAI formulations), as well as risperidone LAI and oral paliperidone, presented weight gain curves that continued climbing at higher doses (especially olanzapine). However, the drugs have different metabolic profiles, ranging from 0.88 kg ED95 for oral aripiprazole to 4.29 kg for olanzapine LAI.
Olanzapine had the most pronounced weight gain, in addition to associations with all metabolic outcomes.
For some drugs with important metabolic side effects, “a lower dose might provide a better combination of high efficacy and reduced metabolic side effects,” the authors write.
The findings might “provide additional information for clinicians aiming to determine the most suitable dose to prevent weight gain and metabolic disturbance in a shared decision-making process with their patients,” they note.
The results add to “existing concerns about the use of olanzapine as a first-line drug,” they add.
Lowest effective dose
Commenting on the study, Roger S. McIntyre, MD, professor of psychiatry and pharmacology, University of Toronto, and head of the mood disorders psychopharmacology unit, said clinicians “not infrequently increase doses to achieve better symptom control, [but] this decision should be informed by the additional observation herein that the increase in those could be accompanied by weight increase.”
Moreover, many patients “take concomitant medications that could possibly increase the bioavailability of antipsychotics, which may also increase the risk for weight gain,” said Dr. McIntyre, chairman and executive director of the Brain and Cognitive Discover Foundation, Toronto. He was not involved with this study.
“These data provide a reason to believe that for many people antipsychotic-associated weight gain could be mitigated by using the lowest effective dose, and rather than censor the use of some medications out of concern for weight gain, perhaps using the lowest effective dose of the medication will provide the opportunity for mitigation,” he added. “So I think it really guides clinicians to provide the lowest effective dose as a potential therapeutic and preventive strategy.”
The study received no financial support. Dr. Sabé reports no relevant financial relationships. Three coauthors report relationships with industry; the full list is contained in the original article.
Dr. McIntyre is a CEO of Braxia Scientific Corp. He has received research grant support from CIHR/GACD/National Natural Science Foundation of China (NSFC) and the Milken Institute; speaker/consultation fees from Lundbeck, Janssen, Alkermes, Neumora Therapeutics, Boehringer Ingelheim, Sage, Biogen, Mitsubishi Tanabe, Purdue, Pfizer, Otsuka, Takeda, Neurocrine, Sunovion, Bausch Health, Axsome, Novo Nordisk, Kris, Sanofi, Eisai, Intra-Cellular, NewBridge Pharmaceuticals, Viatris, Abbvie, and Atai Life Sciences.
A version of this article first appeared on Medscape.com.
In the first dose-response meta-analysis focusing on antipsychotic-induced weight gain, researchers provide data on the trajectory of this risk associated with individual agents.
Investigators analyzed 52 randomized controlled trials (RCTs) encompassing more than 22,500 participants with schizophrenia treated with antipsychotics. They found that, with the exception of aripiprazole long-acting injectable (LAI), all of the other antipsychotics has significant dose-response effect on weight gain. Furthermore, weight gain occurred with some antipsychotics even at relatively low doses.
“We found significant dose-response associations for weight and metabolic variables, with a unique signature for each antipsychotic,” write the investigators, led by Michel Sabé, MD, of the division of adult psychiatry, department of psychiatry, Geneva University Hospitals.
“Despite several limitations, including the limited number of available studies, our results may provide useful information for preventing weight gain and metabolic disturbances by adapting antipsychotic doses,” they add.
The study was published online in The Journal of Clinical Psychiatry.
Balancing risks and benefits
Antipsychotics are first-line therapy for schizophrenia and are associated with weight gain, lipid disturbances, and glucose dysregulation – especially second-generation antipsychotics (SGAs), which can lead to obesity, type 2 diabetes, and metabolic syndrome.
Given that people with schizophrenia also tend to have lifestyle-related cardiovascular risk factors, it’s important to find “a balance between beneficial and adverse effects of antipsychotics,” the investigators note
The question of whether weight gain and metabolic dysregulation are dose-dependent “remains controversial.” The effect of specific SGAs on weight gain has been investigated, but only one study has been conducted using a dose-response meta-analysis, and that study did not address metabolic disturbance.
The investigators conducted a systematic review and a dose-response meta-analysis of fixed-dose randomized controlled trials (RCTs) investigating antipsychotic-induced weight gain and metabolic disturbance in adults with acute schizophrenia.
To be included in the analysis, RCTs had to focus on adult patients with schizophrenia or related disorders and include a placebo as a comparator to the drug.
Studies involved only short-term administration of antipsychotics (2-13 weeks) rather than maintenance therapy.
The mean (SD) change in weight (body weight and/or body mass index) between baseline and the study endpoint constituted the primary outcome, with secondary outcomes including changes in metabolic parameters.
The researchers characterized the dose-response relationship using a nonlinear restricted cubic spline model, with three “knots” located at the 10th, 50th, and 90th percentiles of overall dose distribution.
They also calculated dose-response curves and estimated 50% and 95% effective doses (ED50 and ED95, respectively), extracted from the estimated dose-response curves for each antipsychotic.
The researchers then calculated the weight gain at each effective dose (ED50 and ED95) in milligrams and the weight gain corresponding to the ED95 value in kilograms.
Shared decision-making
Of 6,812 citations, the researchers selected 52 RCTs that met inclusion criteria (n = 22,588 participants, with 16,311 receiving antipsychotics and 6,277 receiving placebo; mean age, 38.5 years, 69.2% male). The studies were conducted between1996 and 2021.
The risk for bias in most studies was “low,” although 21% of the studies “presented a high risk.”
With the exception of aripiprazole LAI, all of the other antipsychotics had a “significant dose-response” association with weight.
For example, oral aripiprazole exhibited a significant dose-response association for weight, but there was no significant association found for aripiprazole LAI (c2 = 8.744; P = .0126 vs. c2 = 3.107; P = .2115). However, both curves were still ascending at maximum doses, the authors note.
Metabolically neutral
Antipsychotics with a decreasing or quasi-parabolic dose-response curve for weight included brexpiprazole, cariprazine, haloperidol, lurasidone, and quetiapine ER: for these antipsychotics, the ED95 weight gain ranged from 0.53 kg to 1.40 kg.
These antipsychotics “reach their weight gain ED95 at relatively low median effective doses, and higher doses, which mostly correspond to near-maximum effective doses, may even be associated with less weight gain,” the authors note.
In addition, only doses higher than the near-maximum effective dose of brexpiprazole were associated with a small increase in total cholesterol. And cariprazine presented “significantly decreasing curves” at higher doses for LDL cholesterol.
With the exception of quetiapine, this group of medications might be regarded as “metabolically neutral” in terms of weight gain and metabolic disturbances.
Antipsychotics with a plateau-shaped curve were asenapine, iloperidone, paliperidone LAI, quetiapine IR, and risperidone, with a weight gain ED95 ranging from 1.36 to 2.65 kg.
Aripiprazole and olanzapine (oral and LAI formulations), as well as risperidone LAI and oral paliperidone, presented weight gain curves that continued climbing at higher doses (especially olanzapine). However, the drugs have different metabolic profiles, ranging from 0.88 kg ED95 for oral aripiprazole to 4.29 kg for olanzapine LAI.
Olanzapine had the most pronounced weight gain, in addition to associations with all metabolic outcomes.
For some drugs with important metabolic side effects, “a lower dose might provide a better combination of high efficacy and reduced metabolic side effects,” the authors write.
The findings might “provide additional information for clinicians aiming to determine the most suitable dose to prevent weight gain and metabolic disturbance in a shared decision-making process with their patients,” they note.
The results add to “existing concerns about the use of olanzapine as a first-line drug,” they add.
Lowest effective dose
Commenting on the study, Roger S. McIntyre, MD, professor of psychiatry and pharmacology, University of Toronto, and head of the mood disorders psychopharmacology unit, said clinicians “not infrequently increase doses to achieve better symptom control, [but] this decision should be informed by the additional observation herein that the increase in those could be accompanied by weight increase.”
Moreover, many patients “take concomitant medications that could possibly increase the bioavailability of antipsychotics, which may also increase the risk for weight gain,” said Dr. McIntyre, chairman and executive director of the Brain and Cognitive Discover Foundation, Toronto. He was not involved with this study.
“These data provide a reason to believe that for many people antipsychotic-associated weight gain could be mitigated by using the lowest effective dose, and rather than censor the use of some medications out of concern for weight gain, perhaps using the lowest effective dose of the medication will provide the opportunity for mitigation,” he added. “So I think it really guides clinicians to provide the lowest effective dose as a potential therapeutic and preventive strategy.”
The study received no financial support. Dr. Sabé reports no relevant financial relationships. Three coauthors report relationships with industry; the full list is contained in the original article.
Dr. McIntyre is a CEO of Braxia Scientific Corp. He has received research grant support from CIHR/GACD/National Natural Science Foundation of China (NSFC) and the Milken Institute; speaker/consultation fees from Lundbeck, Janssen, Alkermes, Neumora Therapeutics, Boehringer Ingelheim, Sage, Biogen, Mitsubishi Tanabe, Purdue, Pfizer, Otsuka, Takeda, Neurocrine, Sunovion, Bausch Health, Axsome, Novo Nordisk, Kris, Sanofi, Eisai, Intra-Cellular, NewBridge Pharmaceuticals, Viatris, Abbvie, and Atai Life Sciences.
A version of this article first appeared on Medscape.com.
In the first dose-response meta-analysis focusing on antipsychotic-induced weight gain, researchers provide data on the trajectory of this risk associated with individual agents.
Investigators analyzed 52 randomized controlled trials (RCTs) encompassing more than 22,500 participants with schizophrenia treated with antipsychotics. They found that, with the exception of aripiprazole long-acting injectable (LAI), all of the other antipsychotics has significant dose-response effect on weight gain. Furthermore, weight gain occurred with some antipsychotics even at relatively low doses.
“We found significant dose-response associations for weight and metabolic variables, with a unique signature for each antipsychotic,” write the investigators, led by Michel Sabé, MD, of the division of adult psychiatry, department of psychiatry, Geneva University Hospitals.
“Despite several limitations, including the limited number of available studies, our results may provide useful information for preventing weight gain and metabolic disturbances by adapting antipsychotic doses,” they add.
The study was published online in The Journal of Clinical Psychiatry.
Balancing risks and benefits
Antipsychotics are first-line therapy for schizophrenia and are associated with weight gain, lipid disturbances, and glucose dysregulation – especially second-generation antipsychotics (SGAs), which can lead to obesity, type 2 diabetes, and metabolic syndrome.
Given that people with schizophrenia also tend to have lifestyle-related cardiovascular risk factors, it’s important to find “a balance between beneficial and adverse effects of antipsychotics,” the investigators note
The question of whether weight gain and metabolic dysregulation are dose-dependent “remains controversial.” The effect of specific SGAs on weight gain has been investigated, but only one study has been conducted using a dose-response meta-analysis, and that study did not address metabolic disturbance.
The investigators conducted a systematic review and a dose-response meta-analysis of fixed-dose randomized controlled trials (RCTs) investigating antipsychotic-induced weight gain and metabolic disturbance in adults with acute schizophrenia.
To be included in the analysis, RCTs had to focus on adult patients with schizophrenia or related disorders and include a placebo as a comparator to the drug.
Studies involved only short-term administration of antipsychotics (2-13 weeks) rather than maintenance therapy.
The mean (SD) change in weight (body weight and/or body mass index) between baseline and the study endpoint constituted the primary outcome, with secondary outcomes including changes in metabolic parameters.
The researchers characterized the dose-response relationship using a nonlinear restricted cubic spline model, with three “knots” located at the 10th, 50th, and 90th percentiles of overall dose distribution.
They also calculated dose-response curves and estimated 50% and 95% effective doses (ED50 and ED95, respectively), extracted from the estimated dose-response curves for each antipsychotic.
The researchers then calculated the weight gain at each effective dose (ED50 and ED95) in milligrams and the weight gain corresponding to the ED95 value in kilograms.
Shared decision-making
Of 6,812 citations, the researchers selected 52 RCTs that met inclusion criteria (n = 22,588 participants, with 16,311 receiving antipsychotics and 6,277 receiving placebo; mean age, 38.5 years, 69.2% male). The studies were conducted between1996 and 2021.
The risk for bias in most studies was “low,” although 21% of the studies “presented a high risk.”
With the exception of aripiprazole LAI, all of the other antipsychotics had a “significant dose-response” association with weight.
For example, oral aripiprazole exhibited a significant dose-response association for weight, but there was no significant association found for aripiprazole LAI (c2 = 8.744; P = .0126 vs. c2 = 3.107; P = .2115). However, both curves were still ascending at maximum doses, the authors note.
Metabolically neutral
Antipsychotics with a decreasing or quasi-parabolic dose-response curve for weight included brexpiprazole, cariprazine, haloperidol, lurasidone, and quetiapine ER: for these antipsychotics, the ED95 weight gain ranged from 0.53 kg to 1.40 kg.
These antipsychotics “reach their weight gain ED95 at relatively low median effective doses, and higher doses, which mostly correspond to near-maximum effective doses, may even be associated with less weight gain,” the authors note.
In addition, only doses higher than the near-maximum effective dose of brexpiprazole were associated with a small increase in total cholesterol. And cariprazine presented “significantly decreasing curves” at higher doses for LDL cholesterol.
With the exception of quetiapine, this group of medications might be regarded as “metabolically neutral” in terms of weight gain and metabolic disturbances.
Antipsychotics with a plateau-shaped curve were asenapine, iloperidone, paliperidone LAI, quetiapine IR, and risperidone, with a weight gain ED95 ranging from 1.36 to 2.65 kg.
Aripiprazole and olanzapine (oral and LAI formulations), as well as risperidone LAI and oral paliperidone, presented weight gain curves that continued climbing at higher doses (especially olanzapine). However, the drugs have different metabolic profiles, ranging from 0.88 kg ED95 for oral aripiprazole to 4.29 kg for olanzapine LAI.
Olanzapine had the most pronounced weight gain, in addition to associations with all metabolic outcomes.
For some drugs with important metabolic side effects, “a lower dose might provide a better combination of high efficacy and reduced metabolic side effects,” the authors write.
The findings might “provide additional information for clinicians aiming to determine the most suitable dose to prevent weight gain and metabolic disturbance in a shared decision-making process with their patients,” they note.
The results add to “existing concerns about the use of olanzapine as a first-line drug,” they add.
Lowest effective dose
Commenting on the study, Roger S. McIntyre, MD, professor of psychiatry and pharmacology, University of Toronto, and head of the mood disorders psychopharmacology unit, said clinicians “not infrequently increase doses to achieve better symptom control, [but] this decision should be informed by the additional observation herein that the increase in those could be accompanied by weight increase.”
Moreover, many patients “take concomitant medications that could possibly increase the bioavailability of antipsychotics, which may also increase the risk for weight gain,” said Dr. McIntyre, chairman and executive director of the Brain and Cognitive Discover Foundation, Toronto. He was not involved with this study.
“These data provide a reason to believe that for many people antipsychotic-associated weight gain could be mitigated by using the lowest effective dose, and rather than censor the use of some medications out of concern for weight gain, perhaps using the lowest effective dose of the medication will provide the opportunity for mitigation,” he added. “So I think it really guides clinicians to provide the lowest effective dose as a potential therapeutic and preventive strategy.”
The study received no financial support. Dr. Sabé reports no relevant financial relationships. Three coauthors report relationships with industry; the full list is contained in the original article.
Dr. McIntyre is a CEO of Braxia Scientific Corp. He has received research grant support from CIHR/GACD/National Natural Science Foundation of China (NSFC) and the Milken Institute; speaker/consultation fees from Lundbeck, Janssen, Alkermes, Neumora Therapeutics, Boehringer Ingelheim, Sage, Biogen, Mitsubishi Tanabe, Purdue, Pfizer, Otsuka, Takeda, Neurocrine, Sunovion, Bausch Health, Axsome, Novo Nordisk, Kris, Sanofi, Eisai, Intra-Cellular, NewBridge Pharmaceuticals, Viatris, Abbvie, and Atai Life Sciences.
A version of this article first appeared on Medscape.com.
FROM THE JOURNAL OF CLINICAL PSYCHIATRY
Keto/paleo diets ‘lower quality than others,’ and bad for planet
suggests a new analysis that reveals meat-based, as well as keto and paleo diets, to be the worst on both measures.
The research was published online in The American Journal of Clinical Nutrition.
To obtain a real-world view on the environmental and health impact of diets as consumed by U.S. adults, the team examined a nationally representative survey of the 1-day eating habits of more than 16,000 individuals.
This revealed that the best quality diet was pescatarian, followed by vegetarian and vegan diets. Omnivore diets, although less healthy, tended to score better than keto and paleo diets, which were the lowest ranked.
Both keto and paleo diets tend to be higher in animal foods and lower in plant foods than other popular diets, the researchers explain in their study, and they both have been associated with negative effects on blood lipids, specifically increased LDL cholesterol, raising concern about the long-term health outcomes associated with these diets.”
Analysis of the environmental impact of the different eating patterns showed that the vegan diet had the lowest carbon footprint, followed by the vegetarian and pescatarian diets. The omnivore, paleo, and keto diets had a far higher carbon footprint, with that of the keto diet more than four times greater than that for a vegan diet.
“Climate change is arguably one of the most pressing problems of our time, and a lot of people are interested in moving to a plant-based diet,” said senior author Diego Rose, PhD, MPH, RD, in a press release.
“Based on our results, that would reduce your footprint and be generally healthy,” noted Dr. Rose, nutrition program director, Tulane University, New Orleans.
To determine the carbon footprint and quality of popular diets as they are consumed by U.S. adults, Keelia O’Malley, PhD, MPH, Amelia Willits-Smith, PhD, MSc, and Dr. Rose, all with Tulane University, studied 24-hour recall data from the ongoing, nationally representative National Health and Nutrition Examination Survey (NHANES) for the years 2005-2010.
The data, which was captured by trained interviewers using a validated tool, was matched with the U.S. Department of Agriculture Food Patterns Equivalents Database to categorize the participants into one of six mutually exclusive categories: vegan, vegetarian, pescatarian, keto, paleo, or omnivore.
The omnivore category included anyone who did not fit into any of the preceding categories.
The environmental impact of the diets was then calculated by matching the established greenhouse gas emissions (GHGE) of over 300 commodities to foods listed on the NHANES, which was then summarized for each individual to give a carbon footprint for their 1-day diet.
Finally, the quality of their diet was estimated using the 2010 versions of the Healthy Eating Index and the Alternate Healthy Eating Index, both of which award a score to food components based on their impact on health.
Overall, 16,412 individuals were included in the analysis, of whom 52.1% were female.
The most common diet was omnivore, which was followed by 83.6% of respondents, followed by vegetarian (7.5%), pescatarian (4.7%), vegan (0.7%), keto (0.4%), and paleo diets (0.3%).
The lowest carbon footprint was seen with a vegan diet, at an average of 0.69 kg of carbon dioxide equivalents per 1,000 kcal consumed, followed by a vegetarian diet (1.16 kg of carbon dioxide equivalents per 1,000 kcal) and pescatarian diet (1.66 kg of carbon dioxide equivalents per 1,000 kcal).
The highest carbon footprints were observed with the omnivore (2.23 kg of carbon dioxide equivalents per 1,000 kcal), paleo (2.62 kg of carbon dioxide equivalents per 1,000 kcal), and keto diets (2.91 kg of carbon dioxide equivalents per 1,000 kcal).
In terms of diet quality, the pescatarian diet was ranked the highest on both eating index scores, followed by the vegetarian, then vegan, diets. The order of the three lowest scores depended on the index used, with either the keto or paleo diet deemed to be the worst quality.
Analysis of individuals following an omnivore diet suggested that those who ate in line with the DASH or Mediterranean diets had higher diet quality, as well as a lower environmental impact, than other people within the group.
Hence, Dr. Rose observed, “Our research ... shows there is a way to improve your health and footprint without giving up meat entirely.”
The researchers acknowledge that the use of 1-day diets has limitations, including that whatever individuals may have eaten during those 24 hours may not correspond to their overall day-in, day-out diet.
The study was supported by the Wellcome Trust. Dr. Rose declares relationships with the Center for Biological Diversity, the NCI, and the Health Resources and Services Administration. Dr. Willits-Smith has received funding from CBD and NCI. Dr. O’Malley has received funding from HRSA.
A version of this article first appeared on Medscape.com.
suggests a new analysis that reveals meat-based, as well as keto and paleo diets, to be the worst on both measures.
The research was published online in The American Journal of Clinical Nutrition.
To obtain a real-world view on the environmental and health impact of diets as consumed by U.S. adults, the team examined a nationally representative survey of the 1-day eating habits of more than 16,000 individuals.
This revealed that the best quality diet was pescatarian, followed by vegetarian and vegan diets. Omnivore diets, although less healthy, tended to score better than keto and paleo diets, which were the lowest ranked.
Both keto and paleo diets tend to be higher in animal foods and lower in plant foods than other popular diets, the researchers explain in their study, and they both have been associated with negative effects on blood lipids, specifically increased LDL cholesterol, raising concern about the long-term health outcomes associated with these diets.”
Analysis of the environmental impact of the different eating patterns showed that the vegan diet had the lowest carbon footprint, followed by the vegetarian and pescatarian diets. The omnivore, paleo, and keto diets had a far higher carbon footprint, with that of the keto diet more than four times greater than that for a vegan diet.
“Climate change is arguably one of the most pressing problems of our time, and a lot of people are interested in moving to a plant-based diet,” said senior author Diego Rose, PhD, MPH, RD, in a press release.
“Based on our results, that would reduce your footprint and be generally healthy,” noted Dr. Rose, nutrition program director, Tulane University, New Orleans.
To determine the carbon footprint and quality of popular diets as they are consumed by U.S. adults, Keelia O’Malley, PhD, MPH, Amelia Willits-Smith, PhD, MSc, and Dr. Rose, all with Tulane University, studied 24-hour recall data from the ongoing, nationally representative National Health and Nutrition Examination Survey (NHANES) for the years 2005-2010.
The data, which was captured by trained interviewers using a validated tool, was matched with the U.S. Department of Agriculture Food Patterns Equivalents Database to categorize the participants into one of six mutually exclusive categories: vegan, vegetarian, pescatarian, keto, paleo, or omnivore.
The omnivore category included anyone who did not fit into any of the preceding categories.
The environmental impact of the diets was then calculated by matching the established greenhouse gas emissions (GHGE) of over 300 commodities to foods listed on the NHANES, which was then summarized for each individual to give a carbon footprint for their 1-day diet.
Finally, the quality of their diet was estimated using the 2010 versions of the Healthy Eating Index and the Alternate Healthy Eating Index, both of which award a score to food components based on their impact on health.
Overall, 16,412 individuals were included in the analysis, of whom 52.1% were female.
The most common diet was omnivore, which was followed by 83.6% of respondents, followed by vegetarian (7.5%), pescatarian (4.7%), vegan (0.7%), keto (0.4%), and paleo diets (0.3%).
The lowest carbon footprint was seen with a vegan diet, at an average of 0.69 kg of carbon dioxide equivalents per 1,000 kcal consumed, followed by a vegetarian diet (1.16 kg of carbon dioxide equivalents per 1,000 kcal) and pescatarian diet (1.66 kg of carbon dioxide equivalents per 1,000 kcal).
The highest carbon footprints were observed with the omnivore (2.23 kg of carbon dioxide equivalents per 1,000 kcal), paleo (2.62 kg of carbon dioxide equivalents per 1,000 kcal), and keto diets (2.91 kg of carbon dioxide equivalents per 1,000 kcal).
In terms of diet quality, the pescatarian diet was ranked the highest on both eating index scores, followed by the vegetarian, then vegan, diets. The order of the three lowest scores depended on the index used, with either the keto or paleo diet deemed to be the worst quality.
Analysis of individuals following an omnivore diet suggested that those who ate in line with the DASH or Mediterranean diets had higher diet quality, as well as a lower environmental impact, than other people within the group.
Hence, Dr. Rose observed, “Our research ... shows there is a way to improve your health and footprint without giving up meat entirely.”
The researchers acknowledge that the use of 1-day diets has limitations, including that whatever individuals may have eaten during those 24 hours may not correspond to their overall day-in, day-out diet.
The study was supported by the Wellcome Trust. Dr. Rose declares relationships with the Center for Biological Diversity, the NCI, and the Health Resources and Services Administration. Dr. Willits-Smith has received funding from CBD and NCI. Dr. O’Malley has received funding from HRSA.
A version of this article first appeared on Medscape.com.
suggests a new analysis that reveals meat-based, as well as keto and paleo diets, to be the worst on both measures.
The research was published online in The American Journal of Clinical Nutrition.
To obtain a real-world view on the environmental and health impact of diets as consumed by U.S. adults, the team examined a nationally representative survey of the 1-day eating habits of more than 16,000 individuals.
This revealed that the best quality diet was pescatarian, followed by vegetarian and vegan diets. Omnivore diets, although less healthy, tended to score better than keto and paleo diets, which were the lowest ranked.
Both keto and paleo diets tend to be higher in animal foods and lower in plant foods than other popular diets, the researchers explain in their study, and they both have been associated with negative effects on blood lipids, specifically increased LDL cholesterol, raising concern about the long-term health outcomes associated with these diets.”
Analysis of the environmental impact of the different eating patterns showed that the vegan diet had the lowest carbon footprint, followed by the vegetarian and pescatarian diets. The omnivore, paleo, and keto diets had a far higher carbon footprint, with that of the keto diet more than four times greater than that for a vegan diet.
“Climate change is arguably one of the most pressing problems of our time, and a lot of people are interested in moving to a plant-based diet,” said senior author Diego Rose, PhD, MPH, RD, in a press release.
“Based on our results, that would reduce your footprint and be generally healthy,” noted Dr. Rose, nutrition program director, Tulane University, New Orleans.
To determine the carbon footprint and quality of popular diets as they are consumed by U.S. adults, Keelia O’Malley, PhD, MPH, Amelia Willits-Smith, PhD, MSc, and Dr. Rose, all with Tulane University, studied 24-hour recall data from the ongoing, nationally representative National Health and Nutrition Examination Survey (NHANES) for the years 2005-2010.
The data, which was captured by trained interviewers using a validated tool, was matched with the U.S. Department of Agriculture Food Patterns Equivalents Database to categorize the participants into one of six mutually exclusive categories: vegan, vegetarian, pescatarian, keto, paleo, or omnivore.
The omnivore category included anyone who did not fit into any of the preceding categories.
The environmental impact of the diets was then calculated by matching the established greenhouse gas emissions (GHGE) of over 300 commodities to foods listed on the NHANES, which was then summarized for each individual to give a carbon footprint for their 1-day diet.
Finally, the quality of their diet was estimated using the 2010 versions of the Healthy Eating Index and the Alternate Healthy Eating Index, both of which award a score to food components based on their impact on health.
Overall, 16,412 individuals were included in the analysis, of whom 52.1% were female.
The most common diet was omnivore, which was followed by 83.6% of respondents, followed by vegetarian (7.5%), pescatarian (4.7%), vegan (0.7%), keto (0.4%), and paleo diets (0.3%).
The lowest carbon footprint was seen with a vegan diet, at an average of 0.69 kg of carbon dioxide equivalents per 1,000 kcal consumed, followed by a vegetarian diet (1.16 kg of carbon dioxide equivalents per 1,000 kcal) and pescatarian diet (1.66 kg of carbon dioxide equivalents per 1,000 kcal).
The highest carbon footprints were observed with the omnivore (2.23 kg of carbon dioxide equivalents per 1,000 kcal), paleo (2.62 kg of carbon dioxide equivalents per 1,000 kcal), and keto diets (2.91 kg of carbon dioxide equivalents per 1,000 kcal).
In terms of diet quality, the pescatarian diet was ranked the highest on both eating index scores, followed by the vegetarian, then vegan, diets. The order of the three lowest scores depended on the index used, with either the keto or paleo diet deemed to be the worst quality.
Analysis of individuals following an omnivore diet suggested that those who ate in line with the DASH or Mediterranean diets had higher diet quality, as well as a lower environmental impact, than other people within the group.
Hence, Dr. Rose observed, “Our research ... shows there is a way to improve your health and footprint without giving up meat entirely.”
The researchers acknowledge that the use of 1-day diets has limitations, including that whatever individuals may have eaten during those 24 hours may not correspond to their overall day-in, day-out diet.
The study was supported by the Wellcome Trust. Dr. Rose declares relationships with the Center for Biological Diversity, the NCI, and the Health Resources and Services Administration. Dr. Willits-Smith has received funding from CBD and NCI. Dr. O’Malley has received funding from HRSA.
A version of this article first appeared on Medscape.com.
FROM THE AMERICAN JOURNAL OF CLINICAL NUTRITION
44-year-old man • elevated total cholesterol • chest pains • ketogenic diet • Dx?
THE CASE
A 44-year-old man with a history of morbid obesity reestablished care in our clinic. He had been treated in our health care system about 5 years previously, and prior lab testing showed a total cholesterol of 203 mg/dL; triglycerides, 191 mg/dL; high-density lipoprotein (HDL), 56 mg/dL; and low-density lipoprotein (LDL), 109 mg/dL. At that time, he weighed 299 lbs (BMI, 39.4). He then started a strict ketogenic diet and a regular exercise program (running ~ 16 miles per week and lifting weights), which he maintained for several years. He had experienced remarkable weight loss; upon reestablishing care, he weighed 199 lbs (BMI, 26.33).
However, lipid testing revealed a severely elevated total cholesterol of 334 mg/dL; LDL, 248 mg/dL; HDL, 67 mg/dL; and triglycerides, 95 mg/dL. He was advised to start statin therapy and to stop his ketogenic diet, but he was hesitant to take either step. He elected to have his lab work reevaluated in 6 months.
About 4 months later, he presented with new and increasing burning pain in his mid chest and upper abdomen. He rated the pain 6/10 in severity and said it occurred during exertion or at night when lying down. Resting would relieve the pain. Reduced intake of spicy foods and caffeine had also helped. He denied dyspnea, diaphoresis, palpitations, or nausea.
The patient was a nonsmoker but did have a strong family history of cardiovascular disease. His vital signs and physical examination were unremarkable, apart from mild epigastric and periumbilical tenderness on palpation.
THE DIAGNOSIS
The patient’s chest pain had features of both gastroesophageal reflux disease (GERD) and coronary artery disease (CAD) with exertional angina. His high-fat diet, nightly symptoms, and the partial relief he achieved by cutting back on spicy foods and caffeine suggested GERD, but the exertional nature of the chest pain and gradual relief with rest was highly suggestive of angina, so an outpatient electrocardiogram treadmill stress test was ordered.
The stress test was markedly abnormal, showing worsening ST depressions and T-wave inversions with exertion, and he experienced chest pain during testing. An urgent left heart catheterization was performed, showing severe multivessel CAD. He subsequently underwent 3-vessel coronary artery bypass grafting. A familial hypercholesterolemia panel failed to reveal any significant variants.
As a result of these findings, the patient received a diagnosis of severe ketogenic diet–associated hypercholesterolemia and early-onset CAD.
Continue to: DISCUSSION
DISCUSSION
Low-carbohydrate (low-carb) and ketogenic diets have grown in popularity throughout the United States over the past decade, particularly for weight loss, and the diet has entered the popular consciousness with several celebrities publicly supporting it.1 Simultaneously, there also has been a growing interest in these diets for the treatment of chronic diseases, such as type 2 diabetes.2 However, the long-term cardiovascular effects of low-carb diets are not well studied, and there is significant heterogeneity among these diets.
Low-carb vs low-fat. Multiple meta-analyses comparing low-carb diets to low-fat diets have found that those following low-carb diets have significantly higher total cholesterol and LDL levels.3,4,5 The National Lipid Association’s review of evidence determined that LDL and total cholesterol responses vary in individuals following a low-carb diet, but that increasing LDL levels in particular were concerning enough to warrant lipid monitoring of patients on low-carb diets.6 Another meta-analysis evaluated the difference in estimated atherosclerotic cardiovascular disease (ASCVD) risk between low-carb and low-fat diets, finding those following a low-carb diet to have a lower estimated ASCVD risk but higher LDL levels.7
Weighing the benefits and harms. Since our patient’s dramatic weight loss and greatly increased exercise level would be expected to lower his LDL levels, the severe worsening of his LDL levels was likely related to his ketogenic diet and was a factor in the early onset of CAD. The benefits of low-carb diets for weight loss, contrasted with the consistent worsening of LDL levels, has prompted a debate about which parameters should be considered in estimating the long-term risk of these diets for patients. Diamond et al8 posit that these diets have beneficial effects on “the most reliable [cardiovascular disease] risk factors,” but long-term, patient-oriented outcome data are lacking, and these diets may not be appropriate for certain patients, as our case demonstrates.
A reasonable strategy for patients contemplating a low-carb diet specifically for weight loss would be to use such a diet for 3 to 6 months to achieve initial and rapid results, then continue with a heart-healthy diet and increased exercise levels to maintain weight loss and reduce long-term cardiovascular risk.
Our patient was started on a postoperative medication regimen of aspirin 81 mg/d, evolocumab 140 mg every 14 days, metoprolol tartrate 25 mg bid, and rosuvastatin 10 mg/d. A year later, he was able to resume a high level of physical activity (6-mile runs) without chest pain. His follow-up lipid panel showed a total cholesterol of 153 mg/dL; LDL, 53 mg/dL; HDL, 89 mg/dL; and triglycerides, 55 mg/dL. He had also switched to a regular diet and had been able to maintain his weight loss.
THE TAKEAWAY
Growing evidence suggests that low-carb diets may have a significant and detrimental effect on LDL levels. The long-term safety of these diets hasn’t been well studied, particularly regarding cardiovascular outcomes. At a minimum, patients who initiate low-carb diets should be counseled on general dietary recommendations regarding saturated fat and cholesterol intake, and they should have a follow-up lipid screening to evaluate for any significant worsening in total cholesterol and LDL levels.
CORRESPONDENCE
Samuel Dickmann, MD, 13611 NW 1st Lane, Suite 200, Newberry, FL 32669; [email protected]
1. Gorin A. What is the keto diet – and is it right for you? NBC News BETTER. February 22, 2018. Accessed February 3, 2023. www.nbcnews.com/better/health/what-keto-diet-it-right-you-ncna847256
2. Tinguely D, Gross J, Kosinski, C. Efficacy of ketogenic diets on type 2 diabetes: a systematic review. Current Diabetes Reports. 2021;21:32. doi: 10.1007/s11892-021-01399-z
3. Mansoor N, Vinknes KJ, Veierod MB, et al. Effects of low-carbohydrate diets v. low-fat diets on body weight and cardiovascular risk factors a meta-analysis of randomised controlled trials. Br J Nutr. 2016;115:466-479. doi: 10.1017/S0007114515004699
4. Bueno NB, de Melo ISV, de Oliveira SL, et al. Very-low-carbohydrate ketogenic diet v. low-fat diet for long-term weight loss: a meta-analysis of randomised controlled trials. Br J Nutr. 2013;110:1178-1187. doi: 10.1017/S0007114513000548
5. Chawla S, Tessarolo Silva F, Amaral Medeiros S, et al. The effect of low-fat and low-carbohydrate diets on weight loss and lipid levels: a systematic review and meta-analysis. Nutrients. 2020;12:3774. doi: 10.3390/nu12123774
6. Kirkpatrick CF, Bolick JP, Kris-Etherton PM, et al. Review of current evidence and clinical recommendations on the effects of low-carbohydrate and very-low-carbohydrate (including ketogenic) diets for the management of body weight and other cardiometabolic risk factors: a scientific statement from the National Lipid Association Nutrition and Lifestyle Task Force. J Clin Lipidol. 2019;13:689-711.e1. doi: 10.1016/j.jacl.2019.08.003
7. Sackner-Bernstein J, Kanter D, Kaul S. Dietary intervention for overweight and obese adults: comparison of low-carbohydrate and low-fat diets. a meta-analysis. PLoS One. 2015;10:e0139817. doi: 10.1371/journal.pone.0139817
8. Diamond DM, O’Neill BJ, Volek JS. Low carbohydrate diet: are concerns with saturated fat, lipids, and cardiovascular disease risk justified? Curr Opin Endocrinol Diabetes Obes. 2020;27:291-300. doi: 10.1097/MED.0000000000000568
THE CASE
A 44-year-old man with a history of morbid obesity reestablished care in our clinic. He had been treated in our health care system about 5 years previously, and prior lab testing showed a total cholesterol of 203 mg/dL; triglycerides, 191 mg/dL; high-density lipoprotein (HDL), 56 mg/dL; and low-density lipoprotein (LDL), 109 mg/dL. At that time, he weighed 299 lbs (BMI, 39.4). He then started a strict ketogenic diet and a regular exercise program (running ~ 16 miles per week and lifting weights), which he maintained for several years. He had experienced remarkable weight loss; upon reestablishing care, he weighed 199 lbs (BMI, 26.33).
However, lipid testing revealed a severely elevated total cholesterol of 334 mg/dL; LDL, 248 mg/dL; HDL, 67 mg/dL; and triglycerides, 95 mg/dL. He was advised to start statin therapy and to stop his ketogenic diet, but he was hesitant to take either step. He elected to have his lab work reevaluated in 6 months.
About 4 months later, he presented with new and increasing burning pain in his mid chest and upper abdomen. He rated the pain 6/10 in severity and said it occurred during exertion or at night when lying down. Resting would relieve the pain. Reduced intake of spicy foods and caffeine had also helped. He denied dyspnea, diaphoresis, palpitations, or nausea.
The patient was a nonsmoker but did have a strong family history of cardiovascular disease. His vital signs and physical examination were unremarkable, apart from mild epigastric and periumbilical tenderness on palpation.
THE DIAGNOSIS
The patient’s chest pain had features of both gastroesophageal reflux disease (GERD) and coronary artery disease (CAD) with exertional angina. His high-fat diet, nightly symptoms, and the partial relief he achieved by cutting back on spicy foods and caffeine suggested GERD, but the exertional nature of the chest pain and gradual relief with rest was highly suggestive of angina, so an outpatient electrocardiogram treadmill stress test was ordered.
The stress test was markedly abnormal, showing worsening ST depressions and T-wave inversions with exertion, and he experienced chest pain during testing. An urgent left heart catheterization was performed, showing severe multivessel CAD. He subsequently underwent 3-vessel coronary artery bypass grafting. A familial hypercholesterolemia panel failed to reveal any significant variants.
As a result of these findings, the patient received a diagnosis of severe ketogenic diet–associated hypercholesterolemia and early-onset CAD.
Continue to: DISCUSSION
DISCUSSION
Low-carbohydrate (low-carb) and ketogenic diets have grown in popularity throughout the United States over the past decade, particularly for weight loss, and the diet has entered the popular consciousness with several celebrities publicly supporting it.1 Simultaneously, there also has been a growing interest in these diets for the treatment of chronic diseases, such as type 2 diabetes.2 However, the long-term cardiovascular effects of low-carb diets are not well studied, and there is significant heterogeneity among these diets.
Low-carb vs low-fat. Multiple meta-analyses comparing low-carb diets to low-fat diets have found that those following low-carb diets have significantly higher total cholesterol and LDL levels.3,4,5 The National Lipid Association’s review of evidence determined that LDL and total cholesterol responses vary in individuals following a low-carb diet, but that increasing LDL levels in particular were concerning enough to warrant lipid monitoring of patients on low-carb diets.6 Another meta-analysis evaluated the difference in estimated atherosclerotic cardiovascular disease (ASCVD) risk between low-carb and low-fat diets, finding those following a low-carb diet to have a lower estimated ASCVD risk but higher LDL levels.7
Weighing the benefits and harms. Since our patient’s dramatic weight loss and greatly increased exercise level would be expected to lower his LDL levels, the severe worsening of his LDL levels was likely related to his ketogenic diet and was a factor in the early onset of CAD. The benefits of low-carb diets for weight loss, contrasted with the consistent worsening of LDL levels, has prompted a debate about which parameters should be considered in estimating the long-term risk of these diets for patients. Diamond et al8 posit that these diets have beneficial effects on “the most reliable [cardiovascular disease] risk factors,” but long-term, patient-oriented outcome data are lacking, and these diets may not be appropriate for certain patients, as our case demonstrates.
A reasonable strategy for patients contemplating a low-carb diet specifically for weight loss would be to use such a diet for 3 to 6 months to achieve initial and rapid results, then continue with a heart-healthy diet and increased exercise levels to maintain weight loss and reduce long-term cardiovascular risk.
Our patient was started on a postoperative medication regimen of aspirin 81 mg/d, evolocumab 140 mg every 14 days, metoprolol tartrate 25 mg bid, and rosuvastatin 10 mg/d. A year later, he was able to resume a high level of physical activity (6-mile runs) without chest pain. His follow-up lipid panel showed a total cholesterol of 153 mg/dL; LDL, 53 mg/dL; HDL, 89 mg/dL; and triglycerides, 55 mg/dL. He had also switched to a regular diet and had been able to maintain his weight loss.
THE TAKEAWAY
Growing evidence suggests that low-carb diets may have a significant and detrimental effect on LDL levels. The long-term safety of these diets hasn’t been well studied, particularly regarding cardiovascular outcomes. At a minimum, patients who initiate low-carb diets should be counseled on general dietary recommendations regarding saturated fat and cholesterol intake, and they should have a follow-up lipid screening to evaluate for any significant worsening in total cholesterol and LDL levels.
CORRESPONDENCE
Samuel Dickmann, MD, 13611 NW 1st Lane, Suite 200, Newberry, FL 32669; [email protected]
THE CASE
A 44-year-old man with a history of morbid obesity reestablished care in our clinic. He had been treated in our health care system about 5 years previously, and prior lab testing showed a total cholesterol of 203 mg/dL; triglycerides, 191 mg/dL; high-density lipoprotein (HDL), 56 mg/dL; and low-density lipoprotein (LDL), 109 mg/dL. At that time, he weighed 299 lbs (BMI, 39.4). He then started a strict ketogenic diet and a regular exercise program (running ~ 16 miles per week and lifting weights), which he maintained for several years. He had experienced remarkable weight loss; upon reestablishing care, he weighed 199 lbs (BMI, 26.33).
However, lipid testing revealed a severely elevated total cholesterol of 334 mg/dL; LDL, 248 mg/dL; HDL, 67 mg/dL; and triglycerides, 95 mg/dL. He was advised to start statin therapy and to stop his ketogenic diet, but he was hesitant to take either step. He elected to have his lab work reevaluated in 6 months.
About 4 months later, he presented with new and increasing burning pain in his mid chest and upper abdomen. He rated the pain 6/10 in severity and said it occurred during exertion or at night when lying down. Resting would relieve the pain. Reduced intake of spicy foods and caffeine had also helped. He denied dyspnea, diaphoresis, palpitations, or nausea.
The patient was a nonsmoker but did have a strong family history of cardiovascular disease. His vital signs and physical examination were unremarkable, apart from mild epigastric and periumbilical tenderness on palpation.
THE DIAGNOSIS
The patient’s chest pain had features of both gastroesophageal reflux disease (GERD) and coronary artery disease (CAD) with exertional angina. His high-fat diet, nightly symptoms, and the partial relief he achieved by cutting back on spicy foods and caffeine suggested GERD, but the exertional nature of the chest pain and gradual relief with rest was highly suggestive of angina, so an outpatient electrocardiogram treadmill stress test was ordered.
The stress test was markedly abnormal, showing worsening ST depressions and T-wave inversions with exertion, and he experienced chest pain during testing. An urgent left heart catheterization was performed, showing severe multivessel CAD. He subsequently underwent 3-vessel coronary artery bypass grafting. A familial hypercholesterolemia panel failed to reveal any significant variants.
As a result of these findings, the patient received a diagnosis of severe ketogenic diet–associated hypercholesterolemia and early-onset CAD.
Continue to: DISCUSSION
DISCUSSION
Low-carbohydrate (low-carb) and ketogenic diets have grown in popularity throughout the United States over the past decade, particularly for weight loss, and the diet has entered the popular consciousness with several celebrities publicly supporting it.1 Simultaneously, there also has been a growing interest in these diets for the treatment of chronic diseases, such as type 2 diabetes.2 However, the long-term cardiovascular effects of low-carb diets are not well studied, and there is significant heterogeneity among these diets.
Low-carb vs low-fat. Multiple meta-analyses comparing low-carb diets to low-fat diets have found that those following low-carb diets have significantly higher total cholesterol and LDL levels.3,4,5 The National Lipid Association’s review of evidence determined that LDL and total cholesterol responses vary in individuals following a low-carb diet, but that increasing LDL levels in particular were concerning enough to warrant lipid monitoring of patients on low-carb diets.6 Another meta-analysis evaluated the difference in estimated atherosclerotic cardiovascular disease (ASCVD) risk between low-carb and low-fat diets, finding those following a low-carb diet to have a lower estimated ASCVD risk but higher LDL levels.7
Weighing the benefits and harms. Since our patient’s dramatic weight loss and greatly increased exercise level would be expected to lower his LDL levels, the severe worsening of his LDL levels was likely related to his ketogenic diet and was a factor in the early onset of CAD. The benefits of low-carb diets for weight loss, contrasted with the consistent worsening of LDL levels, has prompted a debate about which parameters should be considered in estimating the long-term risk of these diets for patients. Diamond et al8 posit that these diets have beneficial effects on “the most reliable [cardiovascular disease] risk factors,” but long-term, patient-oriented outcome data are lacking, and these diets may not be appropriate for certain patients, as our case demonstrates.
A reasonable strategy for patients contemplating a low-carb diet specifically for weight loss would be to use such a diet for 3 to 6 months to achieve initial and rapid results, then continue with a heart-healthy diet and increased exercise levels to maintain weight loss and reduce long-term cardiovascular risk.
Our patient was started on a postoperative medication regimen of aspirin 81 mg/d, evolocumab 140 mg every 14 days, metoprolol tartrate 25 mg bid, and rosuvastatin 10 mg/d. A year later, he was able to resume a high level of physical activity (6-mile runs) without chest pain. His follow-up lipid panel showed a total cholesterol of 153 mg/dL; LDL, 53 mg/dL; HDL, 89 mg/dL; and triglycerides, 55 mg/dL. He had also switched to a regular diet and had been able to maintain his weight loss.
THE TAKEAWAY
Growing evidence suggests that low-carb diets may have a significant and detrimental effect on LDL levels. The long-term safety of these diets hasn’t been well studied, particularly regarding cardiovascular outcomes. At a minimum, patients who initiate low-carb diets should be counseled on general dietary recommendations regarding saturated fat and cholesterol intake, and they should have a follow-up lipid screening to evaluate for any significant worsening in total cholesterol and LDL levels.
CORRESPONDENCE
Samuel Dickmann, MD, 13611 NW 1st Lane, Suite 200, Newberry, FL 32669; [email protected]
1. Gorin A. What is the keto diet – and is it right for you? NBC News BETTER. February 22, 2018. Accessed February 3, 2023. www.nbcnews.com/better/health/what-keto-diet-it-right-you-ncna847256
2. Tinguely D, Gross J, Kosinski, C. Efficacy of ketogenic diets on type 2 diabetes: a systematic review. Current Diabetes Reports. 2021;21:32. doi: 10.1007/s11892-021-01399-z
3. Mansoor N, Vinknes KJ, Veierod MB, et al. Effects of low-carbohydrate diets v. low-fat diets on body weight and cardiovascular risk factors a meta-analysis of randomised controlled trials. Br J Nutr. 2016;115:466-479. doi: 10.1017/S0007114515004699
4. Bueno NB, de Melo ISV, de Oliveira SL, et al. Very-low-carbohydrate ketogenic diet v. low-fat diet for long-term weight loss: a meta-analysis of randomised controlled trials. Br J Nutr. 2013;110:1178-1187. doi: 10.1017/S0007114513000548
5. Chawla S, Tessarolo Silva F, Amaral Medeiros S, et al. The effect of low-fat and low-carbohydrate diets on weight loss and lipid levels: a systematic review and meta-analysis. Nutrients. 2020;12:3774. doi: 10.3390/nu12123774
6. Kirkpatrick CF, Bolick JP, Kris-Etherton PM, et al. Review of current evidence and clinical recommendations on the effects of low-carbohydrate and very-low-carbohydrate (including ketogenic) diets for the management of body weight and other cardiometabolic risk factors: a scientific statement from the National Lipid Association Nutrition and Lifestyle Task Force. J Clin Lipidol. 2019;13:689-711.e1. doi: 10.1016/j.jacl.2019.08.003
7. Sackner-Bernstein J, Kanter D, Kaul S. Dietary intervention for overweight and obese adults: comparison of low-carbohydrate and low-fat diets. a meta-analysis. PLoS One. 2015;10:e0139817. doi: 10.1371/journal.pone.0139817
8. Diamond DM, O’Neill BJ, Volek JS. Low carbohydrate diet: are concerns with saturated fat, lipids, and cardiovascular disease risk justified? Curr Opin Endocrinol Diabetes Obes. 2020;27:291-300. doi: 10.1097/MED.0000000000000568
1. Gorin A. What is the keto diet – and is it right for you? NBC News BETTER. February 22, 2018. Accessed February 3, 2023. www.nbcnews.com/better/health/what-keto-diet-it-right-you-ncna847256
2. Tinguely D, Gross J, Kosinski, C. Efficacy of ketogenic diets on type 2 diabetes: a systematic review. Current Diabetes Reports. 2021;21:32. doi: 10.1007/s11892-021-01399-z
3. Mansoor N, Vinknes KJ, Veierod MB, et al. Effects of low-carbohydrate diets v. low-fat diets on body weight and cardiovascular risk factors a meta-analysis of randomised controlled trials. Br J Nutr. 2016;115:466-479. doi: 10.1017/S0007114515004699
4. Bueno NB, de Melo ISV, de Oliveira SL, et al. Very-low-carbohydrate ketogenic diet v. low-fat diet for long-term weight loss: a meta-analysis of randomised controlled trials. Br J Nutr. 2013;110:1178-1187. doi: 10.1017/S0007114513000548
5. Chawla S, Tessarolo Silva F, Amaral Medeiros S, et al. The effect of low-fat and low-carbohydrate diets on weight loss and lipid levels: a systematic review and meta-analysis. Nutrients. 2020;12:3774. doi: 10.3390/nu12123774
6. Kirkpatrick CF, Bolick JP, Kris-Etherton PM, et al. Review of current evidence and clinical recommendations on the effects of low-carbohydrate and very-low-carbohydrate (including ketogenic) diets for the management of body weight and other cardiometabolic risk factors: a scientific statement from the National Lipid Association Nutrition and Lifestyle Task Force. J Clin Lipidol. 2019;13:689-711.e1. doi: 10.1016/j.jacl.2019.08.003
7. Sackner-Bernstein J, Kanter D, Kaul S. Dietary intervention for overweight and obese adults: comparison of low-carbohydrate and low-fat diets. a meta-analysis. PLoS One. 2015;10:e0139817. doi: 10.1371/journal.pone.0139817
8. Diamond DM, O’Neill BJ, Volek JS. Low carbohydrate diet: are concerns with saturated fat, lipids, and cardiovascular disease risk justified? Curr Opin Endocrinol Diabetes Obes. 2020;27:291-300. doi: 10.1097/MED.0000000000000568
What happens if we sit for more than 8 hours per day?
according to a recent Latin American study published in BMC Public Health.
These data come from almost 8,000 people aged 20-65 years (half of whom are women) who participated in the Latin American Study on Nutrition and Health (ELANS). The cross-sectional survey included representative samples from urban populations in Argentina, Brazil, Chile, Colombia, Costa Rica, Ecuador, Peru, and Venezuela. The average time spent sitting was 420 min/d. Ecuador had the lowest time (300 min/day), and Argentina and Peru had the highest (480 min/day).
No amount of sitting time has been associated with a greater health risk, but the World Health Organization recommends that sitting time be minimal.
“We used to believe that any intense physical exercise could compensate for a sedentary life. But now we know that a sedentary lifestyle in general and sitting time in particular have a direct effect and are an independent risk factor for chronic diseases,” said study author Irina Kovalskys, PhD, a pediatric specialist in nutrition and a professor of nutrition at the Catholic University of Argentina, Buenos Aires, and a principal investigator of ELANS.
Dr. Kovalskys stated that the 420-min average sitting time is worrying in a population such as the one studied, in which 60% of adults are obese and there are high rates of cardiometabolic risk factors. She affirmed that it is important to raise awareness among the population and focus on adolescents.
Felipe Lobelo, PhD, is a Colombian physician, an associate professor of global health at Emory University and director of epidemiology at Kaiser Permanente Georgia, both in Atlanta. He did not participate in this study but promotes the concept of exercise in medicine. The activity of the patient must be included in a clinical setting, and improving the level of physical activity can have a positive impact on health prognosis, he said.
“To make public health recommendations or even advise patients, a cutoff point is needed. Guidelines recommend 150 minutes per week of moderate to vigorous physical activity, and some countries have started to indicate that we should be concerned about people’s sitting time. There is still no equivalent to the 150 minutes, therefore, these studies are important, especially in the Latin American population,” said Dr. Lobelo.
He explained that the concept of an increased risk of death or chronic disease because of a lack of physical activity arose in the past 50 years, but only in the past 2 decades have we started thinking about sitting time.
“Spending more than 8 hours sitting per day clearly causes a much higher risk of chronic diseases, including obesity and diabetes. It may be a continuous and progressive association, and the point at which this increase becomes exponential is clearly between 6 and 8 hours of sitting time,” Dr. Lobelo added.
The authors expected to find a linear association with risk for being overweight or obese after 4 hours, but they did not find one. “This study has limitations. Among them was that other indicators were not considered, such as health indicators. Collaborations are starting with other research groups, and other studies are being designed,” said study author Gerson Ferrari, PhD, an associate professor at Santiago de Chile University.
Comparing indicators
The Latin American study tried to establish a sitting cutoff time after which the risk of becoming overweight or obese increases. It used three indicators of excess weight: body mass index (BMI), waist circumference, and neck circumference.
Sitting for more than 8 hours increased the chances of excess weight by 10% when measured by BMI and by 13% when neck circumference was used.
Dr. Ferrari stated that the result obtained measuring BMI is the one that should be considered, because it is used in public policy. Neck circumference is a more recent measurement of detection and it is less studied, but it is a valid indicator, with good sensitivity and advantages over others, such as ease of measurement and lack of variation over time.
According to the results of this study, measuring neck circumference may be the most sensitive method of the three. Neck circumference was proportionally greater in people who sat for at least 4, at least 6, and at least 8 hours/day than in those who sat for less than 4, less than 6, and less than 8 hours/day. This relationship was not observed with the other indicators.
Broaching the topic
“What is important is uninterrupted sitting time. The recommendation is to break up those sitting times with active periods. Health professionals have already incorporated the concept of moderate to vigorous physical exercise, but nonintense activities are sufficient to reduce sitting time. Yoga may not be vigorous, but it is valuable at reducing sitting time,” said Dr. Kovalskys.
Dr. Ferrari recommended giving patients concrete messages so that they spend as little time possible sitting. “It is better to stand on the bus or the subway even when there is a place to sit. Are you going to talk on the phone? It is better to do it while walking or at least standing instead of sitting.”
A recent literature review conducted by investigators of the University of Birmingham (England) studied the possible molecular and physiologic mechanisms of inactivity time, health consequences, and protection strategies. It offers an evaluation of interventions that can compensate for the immediate negative consequences of inactivity.
Physical activity
Some studies suggest that more than 60 min/day of moderate-intensity exercise or more than 150 min/week of moderate to vigorous exercise may be effective at mitigating the increased risk for mortality associated with sitting time, but reduced intensity may not be enough.
Active pauses
Interrupting sitting every 30-60 min to walk or cycle (2-10 min), performing 3 minutes of simple resistance activities every 30 minutes, such as calf or knee lifts, performing intermittent leg movements (1 minute of activity for every 4 minutes of inactivity during a 3-hour protocol session), or pausing to climb stairs (5 minutes every hour) may be beneficial for vascular health. However, not all studies have demonstrated these positive effects, therefore, some populations may need exercise of greater intensity or duration to counteract the negative vascular effects of acute inactivity periods.
Standing workstations
Standing workstations are effective at reducing sitting time in offices but may be ineffective at reducing vascular alterations related to sitting time. Although some experimental studies indicate vascular benefits, epidemiologic studies suggest that long periods of standing can be harmful to vascular health, especially for venous diseases. Recommendations for use should be accompanied by specific regimens on the frequency and duration of the position to attain the maximum benefits and minimize other vascular complications.
One problem that Dr. Lobelo noted is that some doctors ask their patients how active they are, but they do so in a nonstandardized manner. This observation led him to publish, together with the American Heart Association, an article on the importance for health systems of considering physical activity as a vital sign and including it in records in a standardized manner.
He said that “one advantage of having physical activity as a vital sign in patient medical records is that it allows us to identify individuals who are at greater risk.”
Kaiser Permanente asks the following questions: how many minutes of physical activity do you perform regularly per week, and what is the average intensity of that activity? Patients can be classified into the following three groups: those who follow the recommendations, those with almost no activity, and those who perform some physical activity but do not meet the recommended 150 min/week of moderate to vigorous activity.
Recording sitting time is more difficult. Dr. Lobelo indicated that “it is easier for a person to remember how much time they spent running than how long they were sitting.” Regarding the use of technology, he commented that most watches provide a good estimate. Without technology, it can be estimated by asking how much time is spent in the car, on the bus, or in front of the computer or television and then adding up these times.
Dr. Lobelo emphasized that the two behaviors, lack of physical activity and excessive sitting time, have independent associations with health outcomes. But if both are combined, the risk of obesity, diabetes, and cardiovascular diseases is not just added but rather is multiplied. These behaviors contribute to the epidemic of obesity and diabetes, since most people do not follow either of the two recommendations.
“Studies show that of the two behaviors, the more negative for health would be not following the physical activity recommendations,” said Dr. Lobelo. “If the recommendation of 150 min/week of moderate to vigorous physical activity is followed, the associated risk of sitting too much declines by 80%-90%. Additionally, we can prevent, help to manage, and decrease the risk of complications in more than 100 diseases, including infections. During the pandemic, it was observed that more active people had a lower risk of dying or of being hospitalized due to COVID-19 than less active people, independently of other factors, such as hypertension, diabetes, and obesity.”
Moreover, Dr. Lobelo believes in “practicing what you preach” and advocates that doctors become healthy models.
Dr. Lobelo, Dr. Ferrari, and Dr. Kovalskys disclosed no relevant financial relationships.
This article was translated from the Medscape Spanish edition. A version appeared on Medscape.com.
according to a recent Latin American study published in BMC Public Health.
These data come from almost 8,000 people aged 20-65 years (half of whom are women) who participated in the Latin American Study on Nutrition and Health (ELANS). The cross-sectional survey included representative samples from urban populations in Argentina, Brazil, Chile, Colombia, Costa Rica, Ecuador, Peru, and Venezuela. The average time spent sitting was 420 min/d. Ecuador had the lowest time (300 min/day), and Argentina and Peru had the highest (480 min/day).
No amount of sitting time has been associated with a greater health risk, but the World Health Organization recommends that sitting time be minimal.
“We used to believe that any intense physical exercise could compensate for a sedentary life. But now we know that a sedentary lifestyle in general and sitting time in particular have a direct effect and are an independent risk factor for chronic diseases,” said study author Irina Kovalskys, PhD, a pediatric specialist in nutrition and a professor of nutrition at the Catholic University of Argentina, Buenos Aires, and a principal investigator of ELANS.
Dr. Kovalskys stated that the 420-min average sitting time is worrying in a population such as the one studied, in which 60% of adults are obese and there are high rates of cardiometabolic risk factors. She affirmed that it is important to raise awareness among the population and focus on adolescents.
Felipe Lobelo, PhD, is a Colombian physician, an associate professor of global health at Emory University and director of epidemiology at Kaiser Permanente Georgia, both in Atlanta. He did not participate in this study but promotes the concept of exercise in medicine. The activity of the patient must be included in a clinical setting, and improving the level of physical activity can have a positive impact on health prognosis, he said.
“To make public health recommendations or even advise patients, a cutoff point is needed. Guidelines recommend 150 minutes per week of moderate to vigorous physical activity, and some countries have started to indicate that we should be concerned about people’s sitting time. There is still no equivalent to the 150 minutes, therefore, these studies are important, especially in the Latin American population,” said Dr. Lobelo.
He explained that the concept of an increased risk of death or chronic disease because of a lack of physical activity arose in the past 50 years, but only in the past 2 decades have we started thinking about sitting time.
“Spending more than 8 hours sitting per day clearly causes a much higher risk of chronic diseases, including obesity and diabetes. It may be a continuous and progressive association, and the point at which this increase becomes exponential is clearly between 6 and 8 hours of sitting time,” Dr. Lobelo added.
The authors expected to find a linear association with risk for being overweight or obese after 4 hours, but they did not find one. “This study has limitations. Among them was that other indicators were not considered, such as health indicators. Collaborations are starting with other research groups, and other studies are being designed,” said study author Gerson Ferrari, PhD, an associate professor at Santiago de Chile University.
Comparing indicators
The Latin American study tried to establish a sitting cutoff time after which the risk of becoming overweight or obese increases. It used three indicators of excess weight: body mass index (BMI), waist circumference, and neck circumference.
Sitting for more than 8 hours increased the chances of excess weight by 10% when measured by BMI and by 13% when neck circumference was used.
Dr. Ferrari stated that the result obtained measuring BMI is the one that should be considered, because it is used in public policy. Neck circumference is a more recent measurement of detection and it is less studied, but it is a valid indicator, with good sensitivity and advantages over others, such as ease of measurement and lack of variation over time.
According to the results of this study, measuring neck circumference may be the most sensitive method of the three. Neck circumference was proportionally greater in people who sat for at least 4, at least 6, and at least 8 hours/day than in those who sat for less than 4, less than 6, and less than 8 hours/day. This relationship was not observed with the other indicators.
Broaching the topic
“What is important is uninterrupted sitting time. The recommendation is to break up those sitting times with active periods. Health professionals have already incorporated the concept of moderate to vigorous physical exercise, but nonintense activities are sufficient to reduce sitting time. Yoga may not be vigorous, but it is valuable at reducing sitting time,” said Dr. Kovalskys.
Dr. Ferrari recommended giving patients concrete messages so that they spend as little time possible sitting. “It is better to stand on the bus or the subway even when there is a place to sit. Are you going to talk on the phone? It is better to do it while walking or at least standing instead of sitting.”
A recent literature review conducted by investigators of the University of Birmingham (England) studied the possible molecular and physiologic mechanisms of inactivity time, health consequences, and protection strategies. It offers an evaluation of interventions that can compensate for the immediate negative consequences of inactivity.
Physical activity
Some studies suggest that more than 60 min/day of moderate-intensity exercise or more than 150 min/week of moderate to vigorous exercise may be effective at mitigating the increased risk for mortality associated with sitting time, but reduced intensity may not be enough.
Active pauses
Interrupting sitting every 30-60 min to walk or cycle (2-10 min), performing 3 minutes of simple resistance activities every 30 minutes, such as calf or knee lifts, performing intermittent leg movements (1 minute of activity for every 4 minutes of inactivity during a 3-hour protocol session), or pausing to climb stairs (5 minutes every hour) may be beneficial for vascular health. However, not all studies have demonstrated these positive effects, therefore, some populations may need exercise of greater intensity or duration to counteract the negative vascular effects of acute inactivity periods.
Standing workstations
Standing workstations are effective at reducing sitting time in offices but may be ineffective at reducing vascular alterations related to sitting time. Although some experimental studies indicate vascular benefits, epidemiologic studies suggest that long periods of standing can be harmful to vascular health, especially for venous diseases. Recommendations for use should be accompanied by specific regimens on the frequency and duration of the position to attain the maximum benefits and minimize other vascular complications.
One problem that Dr. Lobelo noted is that some doctors ask their patients how active they are, but they do so in a nonstandardized manner. This observation led him to publish, together with the American Heart Association, an article on the importance for health systems of considering physical activity as a vital sign and including it in records in a standardized manner.
He said that “one advantage of having physical activity as a vital sign in patient medical records is that it allows us to identify individuals who are at greater risk.”
Kaiser Permanente asks the following questions: how many minutes of physical activity do you perform regularly per week, and what is the average intensity of that activity? Patients can be classified into the following three groups: those who follow the recommendations, those with almost no activity, and those who perform some physical activity but do not meet the recommended 150 min/week of moderate to vigorous activity.
Recording sitting time is more difficult. Dr. Lobelo indicated that “it is easier for a person to remember how much time they spent running than how long they were sitting.” Regarding the use of technology, he commented that most watches provide a good estimate. Without technology, it can be estimated by asking how much time is spent in the car, on the bus, or in front of the computer or television and then adding up these times.
Dr. Lobelo emphasized that the two behaviors, lack of physical activity and excessive sitting time, have independent associations with health outcomes. But if both are combined, the risk of obesity, diabetes, and cardiovascular diseases is not just added but rather is multiplied. These behaviors contribute to the epidemic of obesity and diabetes, since most people do not follow either of the two recommendations.
“Studies show that of the two behaviors, the more negative for health would be not following the physical activity recommendations,” said Dr. Lobelo. “If the recommendation of 150 min/week of moderate to vigorous physical activity is followed, the associated risk of sitting too much declines by 80%-90%. Additionally, we can prevent, help to manage, and decrease the risk of complications in more than 100 diseases, including infections. During the pandemic, it was observed that more active people had a lower risk of dying or of being hospitalized due to COVID-19 than less active people, independently of other factors, such as hypertension, diabetes, and obesity.”
Moreover, Dr. Lobelo believes in “practicing what you preach” and advocates that doctors become healthy models.
Dr. Lobelo, Dr. Ferrari, and Dr. Kovalskys disclosed no relevant financial relationships.
This article was translated from the Medscape Spanish edition. A version appeared on Medscape.com.
according to a recent Latin American study published in BMC Public Health.
These data come from almost 8,000 people aged 20-65 years (half of whom are women) who participated in the Latin American Study on Nutrition and Health (ELANS). The cross-sectional survey included representative samples from urban populations in Argentina, Brazil, Chile, Colombia, Costa Rica, Ecuador, Peru, and Venezuela. The average time spent sitting was 420 min/d. Ecuador had the lowest time (300 min/day), and Argentina and Peru had the highest (480 min/day).
No amount of sitting time has been associated with a greater health risk, but the World Health Organization recommends that sitting time be minimal.
“We used to believe that any intense physical exercise could compensate for a sedentary life. But now we know that a sedentary lifestyle in general and sitting time in particular have a direct effect and are an independent risk factor for chronic diseases,” said study author Irina Kovalskys, PhD, a pediatric specialist in nutrition and a professor of nutrition at the Catholic University of Argentina, Buenos Aires, and a principal investigator of ELANS.
Dr. Kovalskys stated that the 420-min average sitting time is worrying in a population such as the one studied, in which 60% of adults are obese and there are high rates of cardiometabolic risk factors. She affirmed that it is important to raise awareness among the population and focus on adolescents.
Felipe Lobelo, PhD, is a Colombian physician, an associate professor of global health at Emory University and director of epidemiology at Kaiser Permanente Georgia, both in Atlanta. He did not participate in this study but promotes the concept of exercise in medicine. The activity of the patient must be included in a clinical setting, and improving the level of physical activity can have a positive impact on health prognosis, he said.
“To make public health recommendations or even advise patients, a cutoff point is needed. Guidelines recommend 150 minutes per week of moderate to vigorous physical activity, and some countries have started to indicate that we should be concerned about people’s sitting time. There is still no equivalent to the 150 minutes, therefore, these studies are important, especially in the Latin American population,” said Dr. Lobelo.
He explained that the concept of an increased risk of death or chronic disease because of a lack of physical activity arose in the past 50 years, but only in the past 2 decades have we started thinking about sitting time.
“Spending more than 8 hours sitting per day clearly causes a much higher risk of chronic diseases, including obesity and diabetes. It may be a continuous and progressive association, and the point at which this increase becomes exponential is clearly between 6 and 8 hours of sitting time,” Dr. Lobelo added.
The authors expected to find a linear association with risk for being overweight or obese after 4 hours, but they did not find one. “This study has limitations. Among them was that other indicators were not considered, such as health indicators. Collaborations are starting with other research groups, and other studies are being designed,” said study author Gerson Ferrari, PhD, an associate professor at Santiago de Chile University.
Comparing indicators
The Latin American study tried to establish a sitting cutoff time after which the risk of becoming overweight or obese increases. It used three indicators of excess weight: body mass index (BMI), waist circumference, and neck circumference.
Sitting for more than 8 hours increased the chances of excess weight by 10% when measured by BMI and by 13% when neck circumference was used.
Dr. Ferrari stated that the result obtained measuring BMI is the one that should be considered, because it is used in public policy. Neck circumference is a more recent measurement of detection and it is less studied, but it is a valid indicator, with good sensitivity and advantages over others, such as ease of measurement and lack of variation over time.
According to the results of this study, measuring neck circumference may be the most sensitive method of the three. Neck circumference was proportionally greater in people who sat for at least 4, at least 6, and at least 8 hours/day than in those who sat for less than 4, less than 6, and less than 8 hours/day. This relationship was not observed with the other indicators.
Broaching the topic
“What is important is uninterrupted sitting time. The recommendation is to break up those sitting times with active periods. Health professionals have already incorporated the concept of moderate to vigorous physical exercise, but nonintense activities are sufficient to reduce sitting time. Yoga may not be vigorous, but it is valuable at reducing sitting time,” said Dr. Kovalskys.
Dr. Ferrari recommended giving patients concrete messages so that they spend as little time possible sitting. “It is better to stand on the bus or the subway even when there is a place to sit. Are you going to talk on the phone? It is better to do it while walking or at least standing instead of sitting.”
A recent literature review conducted by investigators of the University of Birmingham (England) studied the possible molecular and physiologic mechanisms of inactivity time, health consequences, and protection strategies. It offers an evaluation of interventions that can compensate for the immediate negative consequences of inactivity.
Physical activity
Some studies suggest that more than 60 min/day of moderate-intensity exercise or more than 150 min/week of moderate to vigorous exercise may be effective at mitigating the increased risk for mortality associated with sitting time, but reduced intensity may not be enough.
Active pauses
Interrupting sitting every 30-60 min to walk or cycle (2-10 min), performing 3 minutes of simple resistance activities every 30 minutes, such as calf or knee lifts, performing intermittent leg movements (1 minute of activity for every 4 minutes of inactivity during a 3-hour protocol session), or pausing to climb stairs (5 minutes every hour) may be beneficial for vascular health. However, not all studies have demonstrated these positive effects, therefore, some populations may need exercise of greater intensity or duration to counteract the negative vascular effects of acute inactivity periods.
Standing workstations
Standing workstations are effective at reducing sitting time in offices but may be ineffective at reducing vascular alterations related to sitting time. Although some experimental studies indicate vascular benefits, epidemiologic studies suggest that long periods of standing can be harmful to vascular health, especially for venous diseases. Recommendations for use should be accompanied by specific regimens on the frequency and duration of the position to attain the maximum benefits and minimize other vascular complications.
One problem that Dr. Lobelo noted is that some doctors ask their patients how active they are, but they do so in a nonstandardized manner. This observation led him to publish, together with the American Heart Association, an article on the importance for health systems of considering physical activity as a vital sign and including it in records in a standardized manner.
He said that “one advantage of having physical activity as a vital sign in patient medical records is that it allows us to identify individuals who are at greater risk.”
Kaiser Permanente asks the following questions: how many minutes of physical activity do you perform regularly per week, and what is the average intensity of that activity? Patients can be classified into the following three groups: those who follow the recommendations, those with almost no activity, and those who perform some physical activity but do not meet the recommended 150 min/week of moderate to vigorous activity.
Recording sitting time is more difficult. Dr. Lobelo indicated that “it is easier for a person to remember how much time they spent running than how long they were sitting.” Regarding the use of technology, he commented that most watches provide a good estimate. Without technology, it can be estimated by asking how much time is spent in the car, on the bus, or in front of the computer or television and then adding up these times.
Dr. Lobelo emphasized that the two behaviors, lack of physical activity and excessive sitting time, have independent associations with health outcomes. But if both are combined, the risk of obesity, diabetes, and cardiovascular diseases is not just added but rather is multiplied. These behaviors contribute to the epidemic of obesity and diabetes, since most people do not follow either of the two recommendations.
“Studies show that of the two behaviors, the more negative for health would be not following the physical activity recommendations,” said Dr. Lobelo. “If the recommendation of 150 min/week of moderate to vigorous physical activity is followed, the associated risk of sitting too much declines by 80%-90%. Additionally, we can prevent, help to manage, and decrease the risk of complications in more than 100 diseases, including infections. During the pandemic, it was observed that more active people had a lower risk of dying or of being hospitalized due to COVID-19 than less active people, independently of other factors, such as hypertension, diabetes, and obesity.”
Moreover, Dr. Lobelo believes in “practicing what you preach” and advocates that doctors become healthy models.
Dr. Lobelo, Dr. Ferrari, and Dr. Kovalskys disclosed no relevant financial relationships.
This article was translated from the Medscape Spanish edition. A version appeared on Medscape.com.
FROM BMC PUBLIC HEALTH
Fewer than 10% of eligible type 2 diabetes patients get new, pricey drugs
Fewer than 10% of American adults with type 2 diabetes who qualified for treatment with newer agents – such as an SGLT2 inhibitor or GLP-1 agonist – actually received treatment with at least one drug from drug class in 2017-2020, based on a new analysis of just over a thousand adults who participated in a representative, biannual survey and self-reported a diabetes diagnosis.
The cost of these agents, and their uncertain cost-effectiveness at current prices, is likely a key driver of the low usage rate, say the authors of a brief report published in Annals of Internal Medicine.
“Clinical studies have shown that both GLP-1 [glucagonlike peptide–1] receptor agonists and SGLT2 [sodium-glucose cotransporter 2] inhibitors yield additional clinical benefits, compared with older treatments in reducing body weight and progression of cardiovascular disease and chronic kidney disease,” write Shichao Tang, PhD, from the U.S. Centers for Disease Control and Prevention, Atlanta, and colleagues.
“However, these medications come at a substantially higher cost,” they stress.
Dr. Tang explained in an interview that the new study “points to prior studies about the high cost of these medications as a potential barrier to use, but more research is needed to understand cost-effectiveness and any potential barriers to use, including cost.”
The work “did not include research into cost-effectiveness or why the percentage of people already using these medications was low,” he emphasized.
Dr. Tang and colleagues used data collected by the U.S. National Health and Nutrition Examination Survey during two 2-year cycles between 2017 and 2020 that included 1,417 people who self-identified a diagnosis of diabetes.
Excluding those who likely had type 1 diabetes and those with incomplete data left 1,330 survey participants, including 1,133 (85%) who fit criteria for the treatment of type 2 diabetes with an agent from one of the two studied classes, as recommended in 2022 by a panel representing the American Diabetes Association and the European Association for the Study of Diabetes.
Among these 1,133 people – who represent more than 22 million American adults with type 2 diabetes who fit the 2022 criteria – a scant 3.7% were actually taking a GLP-1 agonist and 5.3% were taking an SGLT2 inhibitor.
“While it’s important to note that our data predate the 2022 recommendations, these drugs were offered as second-line therapy for patients with certain diabetes-related complications in 2017-2020” and hence provide potentially useful insights, noted Dr. Tang, a health economist with the CDC National Center for Chronic Disease Prevention and Health Promotion.
Based on retail prices listed on a United States–based website, a 30-day supply of an oral SGLT2 inhibitor can cost about $550-$600 per month, while common subcutaneously injected GLP-1 receptor agonists can run from a few hundred dollars for a daily injection or close to $1,000 for a formulation administered weekly.
“Cost-effectiveness was not formally considered in the current guideline, but an assessment of cost-effectiveness may assist better targeting of interventions to achieve the greatest effect at a sustainable cost,” the researchers conclude.
The study received no commercial funding. None of the authors had relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Fewer than 10% of American adults with type 2 diabetes who qualified for treatment with newer agents – such as an SGLT2 inhibitor or GLP-1 agonist – actually received treatment with at least one drug from drug class in 2017-2020, based on a new analysis of just over a thousand adults who participated in a representative, biannual survey and self-reported a diabetes diagnosis.
The cost of these agents, and their uncertain cost-effectiveness at current prices, is likely a key driver of the low usage rate, say the authors of a brief report published in Annals of Internal Medicine.
“Clinical studies have shown that both GLP-1 [glucagonlike peptide–1] receptor agonists and SGLT2 [sodium-glucose cotransporter 2] inhibitors yield additional clinical benefits, compared with older treatments in reducing body weight and progression of cardiovascular disease and chronic kidney disease,” write Shichao Tang, PhD, from the U.S. Centers for Disease Control and Prevention, Atlanta, and colleagues.
“However, these medications come at a substantially higher cost,” they stress.
Dr. Tang explained in an interview that the new study “points to prior studies about the high cost of these medications as a potential barrier to use, but more research is needed to understand cost-effectiveness and any potential barriers to use, including cost.”
The work “did not include research into cost-effectiveness or why the percentage of people already using these medications was low,” he emphasized.
Dr. Tang and colleagues used data collected by the U.S. National Health and Nutrition Examination Survey during two 2-year cycles between 2017 and 2020 that included 1,417 people who self-identified a diagnosis of diabetes.
Excluding those who likely had type 1 diabetes and those with incomplete data left 1,330 survey participants, including 1,133 (85%) who fit criteria for the treatment of type 2 diabetes with an agent from one of the two studied classes, as recommended in 2022 by a panel representing the American Diabetes Association and the European Association for the Study of Diabetes.
Among these 1,133 people – who represent more than 22 million American adults with type 2 diabetes who fit the 2022 criteria – a scant 3.7% were actually taking a GLP-1 agonist and 5.3% were taking an SGLT2 inhibitor.
“While it’s important to note that our data predate the 2022 recommendations, these drugs were offered as second-line therapy for patients with certain diabetes-related complications in 2017-2020” and hence provide potentially useful insights, noted Dr. Tang, a health economist with the CDC National Center for Chronic Disease Prevention and Health Promotion.
Based on retail prices listed on a United States–based website, a 30-day supply of an oral SGLT2 inhibitor can cost about $550-$600 per month, while common subcutaneously injected GLP-1 receptor agonists can run from a few hundred dollars for a daily injection or close to $1,000 for a formulation administered weekly.
“Cost-effectiveness was not formally considered in the current guideline, but an assessment of cost-effectiveness may assist better targeting of interventions to achieve the greatest effect at a sustainable cost,” the researchers conclude.
The study received no commercial funding. None of the authors had relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Fewer than 10% of American adults with type 2 diabetes who qualified for treatment with newer agents – such as an SGLT2 inhibitor or GLP-1 agonist – actually received treatment with at least one drug from drug class in 2017-2020, based on a new analysis of just over a thousand adults who participated in a representative, biannual survey and self-reported a diabetes diagnosis.
The cost of these agents, and their uncertain cost-effectiveness at current prices, is likely a key driver of the low usage rate, say the authors of a brief report published in Annals of Internal Medicine.
“Clinical studies have shown that both GLP-1 [glucagonlike peptide–1] receptor agonists and SGLT2 [sodium-glucose cotransporter 2] inhibitors yield additional clinical benefits, compared with older treatments in reducing body weight and progression of cardiovascular disease and chronic kidney disease,” write Shichao Tang, PhD, from the U.S. Centers for Disease Control and Prevention, Atlanta, and colleagues.
“However, these medications come at a substantially higher cost,” they stress.
Dr. Tang explained in an interview that the new study “points to prior studies about the high cost of these medications as a potential barrier to use, but more research is needed to understand cost-effectiveness and any potential barriers to use, including cost.”
The work “did not include research into cost-effectiveness or why the percentage of people already using these medications was low,” he emphasized.
Dr. Tang and colleagues used data collected by the U.S. National Health and Nutrition Examination Survey during two 2-year cycles between 2017 and 2020 that included 1,417 people who self-identified a diagnosis of diabetes.
Excluding those who likely had type 1 diabetes and those with incomplete data left 1,330 survey participants, including 1,133 (85%) who fit criteria for the treatment of type 2 diabetes with an agent from one of the two studied classes, as recommended in 2022 by a panel representing the American Diabetes Association and the European Association for the Study of Diabetes.
Among these 1,133 people – who represent more than 22 million American adults with type 2 diabetes who fit the 2022 criteria – a scant 3.7% were actually taking a GLP-1 agonist and 5.3% were taking an SGLT2 inhibitor.
“While it’s important to note that our data predate the 2022 recommendations, these drugs were offered as second-line therapy for patients with certain diabetes-related complications in 2017-2020” and hence provide potentially useful insights, noted Dr. Tang, a health economist with the CDC National Center for Chronic Disease Prevention and Health Promotion.
Based on retail prices listed on a United States–based website, a 30-day supply of an oral SGLT2 inhibitor can cost about $550-$600 per month, while common subcutaneously injected GLP-1 receptor agonists can run from a few hundred dollars for a daily injection or close to $1,000 for a formulation administered weekly.
“Cost-effectiveness was not formally considered in the current guideline, but an assessment of cost-effectiveness may assist better targeting of interventions to achieve the greatest effect at a sustainable cost,” the researchers conclude.
The study received no commercial funding. None of the authors had relevant financial relationships.
A version of this article originally appeared on Medscape.com.
FROM ANNALS OF INTERNAL MEDICINE
Swelling of the lower extremities
The patient is sent for lymphoscintigraphy, which showed impaired lymphatic drainage of both lower extremities consistent with obesity-induced lymphedema.
Lymphedema is a chronic condition caused by the abnormal development of the lymphatic system (primary lymphedema) or injury to lymphatic vasculature (secondary lymphedema). Chronic interstitial fluid accumulation may lead to fibrosis, persistent inflammation, and adipose deposition, which often results in massive hypertrophy. Obesity, which affects approximately 40% of the US population, is a rising cause of secondary lymphedema. Obesity-induced lymphedema (OIL) is a result of external compression of the lymphatic system by adipose tissue and increased production of lymph, which results in direct injury to the lymphatic endothelium. As BMI increases, lymphedema worsens and ambulation becomes more difficult, placing patients in an unfavorable cycle of weight gain and lymphatic injury.
The diagnosis of OIL is made by history and physical and is confirmed with diagnostic imaging. The classic presentation of lymphedema is edema of the lower extremities and a positive Stemmer sign. The Stemmer sign is positive if the examiner is unable to grab the dorsal skin between the thumb and index finger. The gold standard for lymphatic imaging is radionuclide lymphoscintigraphy. It involves injecting a tracer protein into the distal extremity, which should be taken up by the lymphatic vasculature and visualized in patients with normal lymphatic function. Lymphoscintigraphy has a high sensitivity (96%) and specificity (100%) for the diagnosis of lymphedema.
The risk for lymphatic dysfunction increases with elevated BMI. A BMI threshold appears to exist between 53 and 59, at which point lower-extremity lymphatic dysfunction begins to occur and is almost universal when BMI exceeds 60. Sixty percent of patients with OIL will develop massive localized lymphedema; the higher the BMI, the greater the risk for massive localized lymphedema. Typical areas of localized lymphedema include the lower extremities, genitals, and abdominal wall. In addition, patients with OIL are at increased risk for infections, such as cellulitis. Other complications of OIL include functional disabilities, psychosocial morbidity, and malignant transformation.
Patients at risk for OIL should be counseled and educated about weight management interventions, such as intensive treatment programs, lifestyle changes, and medications before their BMI reaches 50, a threshold where irreversible lower-extremity lymphedema may occur. Although weight loss may reduce symptoms of OIL, irreversible lymphatic dysfunction also may occur. Individuals at risk for OIL are often referred to a bariatric surgical weight management center.
Lymphedema management consists of compression regimens, physiotherapy, and manual lymphatic drainage. Adipose deposition in the late stages of lymphedema may decrease the response to such manual treatments. Operative procedures are typically used when these treatment options have been inadequate. Patients with chronic advanced lymphedema, in whom lymphatic impairment is accompanied by deposition of fibroadipose tissue, may also require debulking surgery.
Courtney Whittle, MD, MSW, Diplomate of ABOM, Pediatric Lead, Obesity Champion, TSPMG, Weight A Minute Clinic, Atlanta, Georgia.
Courtney Whittle, MD, MSW, Diplomate of ABOM, has disclosed no relevant financial relationships.
Image Quizzes are fictional or fictionalized clinical scenarios intended to provide evidence-based educational takeaways.
The patient is sent for lymphoscintigraphy, which showed impaired lymphatic drainage of both lower extremities consistent with obesity-induced lymphedema.
Lymphedema is a chronic condition caused by the abnormal development of the lymphatic system (primary lymphedema) or injury to lymphatic vasculature (secondary lymphedema). Chronic interstitial fluid accumulation may lead to fibrosis, persistent inflammation, and adipose deposition, which often results in massive hypertrophy. Obesity, which affects approximately 40% of the US population, is a rising cause of secondary lymphedema. Obesity-induced lymphedema (OIL) is a result of external compression of the lymphatic system by adipose tissue and increased production of lymph, which results in direct injury to the lymphatic endothelium. As BMI increases, lymphedema worsens and ambulation becomes more difficult, placing patients in an unfavorable cycle of weight gain and lymphatic injury.
The diagnosis of OIL is made by history and physical and is confirmed with diagnostic imaging. The classic presentation of lymphedema is edema of the lower extremities and a positive Stemmer sign. The Stemmer sign is positive if the examiner is unable to grab the dorsal skin between the thumb and index finger. The gold standard for lymphatic imaging is radionuclide lymphoscintigraphy. It involves injecting a tracer protein into the distal extremity, which should be taken up by the lymphatic vasculature and visualized in patients with normal lymphatic function. Lymphoscintigraphy has a high sensitivity (96%) and specificity (100%) for the diagnosis of lymphedema.
The risk for lymphatic dysfunction increases with elevated BMI. A BMI threshold appears to exist between 53 and 59, at which point lower-extremity lymphatic dysfunction begins to occur and is almost universal when BMI exceeds 60. Sixty percent of patients with OIL will develop massive localized lymphedema; the higher the BMI, the greater the risk for massive localized lymphedema. Typical areas of localized lymphedema include the lower extremities, genitals, and abdominal wall. In addition, patients with OIL are at increased risk for infections, such as cellulitis. Other complications of OIL include functional disabilities, psychosocial morbidity, and malignant transformation.
Patients at risk for OIL should be counseled and educated about weight management interventions, such as intensive treatment programs, lifestyle changes, and medications before their BMI reaches 50, a threshold where irreversible lower-extremity lymphedema may occur. Although weight loss may reduce symptoms of OIL, irreversible lymphatic dysfunction also may occur. Individuals at risk for OIL are often referred to a bariatric surgical weight management center.
Lymphedema management consists of compression regimens, physiotherapy, and manual lymphatic drainage. Adipose deposition in the late stages of lymphedema may decrease the response to such manual treatments. Operative procedures are typically used when these treatment options have been inadequate. Patients with chronic advanced lymphedema, in whom lymphatic impairment is accompanied by deposition of fibroadipose tissue, may also require debulking surgery.
Courtney Whittle, MD, MSW, Diplomate of ABOM, Pediatric Lead, Obesity Champion, TSPMG, Weight A Minute Clinic, Atlanta, Georgia.
Courtney Whittle, MD, MSW, Diplomate of ABOM, has disclosed no relevant financial relationships.
Image Quizzes are fictional or fictionalized clinical scenarios intended to provide evidence-based educational takeaways.
The patient is sent for lymphoscintigraphy, which showed impaired lymphatic drainage of both lower extremities consistent with obesity-induced lymphedema.
Lymphedema is a chronic condition caused by the abnormal development of the lymphatic system (primary lymphedema) or injury to lymphatic vasculature (secondary lymphedema). Chronic interstitial fluid accumulation may lead to fibrosis, persistent inflammation, and adipose deposition, which often results in massive hypertrophy. Obesity, which affects approximately 40% of the US population, is a rising cause of secondary lymphedema. Obesity-induced lymphedema (OIL) is a result of external compression of the lymphatic system by adipose tissue and increased production of lymph, which results in direct injury to the lymphatic endothelium. As BMI increases, lymphedema worsens and ambulation becomes more difficult, placing patients in an unfavorable cycle of weight gain and lymphatic injury.
The diagnosis of OIL is made by history and physical and is confirmed with diagnostic imaging. The classic presentation of lymphedema is edema of the lower extremities and a positive Stemmer sign. The Stemmer sign is positive if the examiner is unable to grab the dorsal skin between the thumb and index finger. The gold standard for lymphatic imaging is radionuclide lymphoscintigraphy. It involves injecting a tracer protein into the distal extremity, which should be taken up by the lymphatic vasculature and visualized in patients with normal lymphatic function. Lymphoscintigraphy has a high sensitivity (96%) and specificity (100%) for the diagnosis of lymphedema.
The risk for lymphatic dysfunction increases with elevated BMI. A BMI threshold appears to exist between 53 and 59, at which point lower-extremity lymphatic dysfunction begins to occur and is almost universal when BMI exceeds 60. Sixty percent of patients with OIL will develop massive localized lymphedema; the higher the BMI, the greater the risk for massive localized lymphedema. Typical areas of localized lymphedema include the lower extremities, genitals, and abdominal wall. In addition, patients with OIL are at increased risk for infections, such as cellulitis. Other complications of OIL include functional disabilities, psychosocial morbidity, and malignant transformation.
Patients at risk for OIL should be counseled and educated about weight management interventions, such as intensive treatment programs, lifestyle changes, and medications before their BMI reaches 50, a threshold where irreversible lower-extremity lymphedema may occur. Although weight loss may reduce symptoms of OIL, irreversible lymphatic dysfunction also may occur. Individuals at risk for OIL are often referred to a bariatric surgical weight management center.
Lymphedema management consists of compression regimens, physiotherapy, and manual lymphatic drainage. Adipose deposition in the late stages of lymphedema may decrease the response to such manual treatments. Operative procedures are typically used when these treatment options have been inadequate. Patients with chronic advanced lymphedema, in whom lymphatic impairment is accompanied by deposition of fibroadipose tissue, may also require debulking surgery.
Courtney Whittle, MD, MSW, Diplomate of ABOM, Pediatric Lead, Obesity Champion, TSPMG, Weight A Minute Clinic, Atlanta, Georgia.
Courtney Whittle, MD, MSW, Diplomate of ABOM, has disclosed no relevant financial relationships.
Image Quizzes are fictional or fictionalized clinical scenarios intended to provide evidence-based educational takeaways.
A 52-year-old woman presents with bilateral pain and swelling of the lower extremities. She is 5 ft 6 in tall and weighs 376 lb; her BMI is 60.7. Four years ago, the patient was injured in a car accident, which limited her mobility; at that time her BMI was 39. She complains of difficulty using her legs and has developed periodic infections of the lower limbs, which have been treated at the wound clinic. Past medical history is significant for diabetes and hypertension, managed with metformin and lisinopril. She reports no history of penetrating trauma, lymphadenectomy, recent travel, or radiation. On physical examination, there is pitting edema of the lower extremities and a positive Stemmer sign bilaterally.
New AHA statement urges focus on CV risk before pregnancy
Increased public health and research efforts to optimize prepregnancy cardiovascular health are needed, particularly among those in under-represented racial and ethnic groups, according to a new scientific statement from the American Heart Association.
“We have released this statement at this time because there is a maternal health crisis in the U.S. with rising maternal morbidity and mortality rates, which are the highest among high-income countries,” chair of the scientific statement writing group, Sadiya S. Khan, MD, told this news organization.
Cardiovascular disease (CVD) is the leading cause of death during pregnancy and the postpartum period and represents 26.5% of pregnancy-related deaths, the statement reports.
“While there is a lot of emphasis in trying to reduce cardiovascular risk during the period of actual pregnancy, much of that risk has often already developed and the women have been living with it for some time, so interventions during pregnancy may be too late,” Dr. Khan, assistant professor of medicine and preventive medicine at Northwestern University, Chicago, said.
“We wanted to try and emphasize the importance of starting to reduce cardiovascular risk earlier before pregnancy. In terms of improving cardiovascular health, this should have benefits both for the mother and the child,” she added.
The statement, “Optimizing Prepregnancy Cardiovascular Health to Improve Outcomes in Pregnant and Postpartum Individuals and Offspring” was published online in a “Go Red For Women” spotlight issue of the AHA publication Circulation.
Currently, nearly one in five births are complicated by such an adverse pregnancy outcome, and there is a strong association between these complications and risk for subsequent cardiovascular disease.
Prepregnancy window
Over the past decade, rates of adverse pregnancy outcomes have increased significantly in the United States, with a near doubling in rates of hypertensive disorders of pregnancy, and there are persistent disparities, with Black individuals significantly more likely to experience adverse pregnancy outcomes, the statement notes.
Emerging data suggest that these complications have, at least in part, prepregnancy origins. Thus, the prepregnancy period may be a critical window during which interventions have a great potential for benefit in both women and their offspring, it says.
The authors suggest a life-course approach to measure, modify, and monitor prepregnancy cardiovascular health, with all clinicians who interact with pregnancy-capable individuals emphasizing optimization of cardiovascular health beginning early in childhood.
“Leveraging these opportunities to target cardiovascular health has the potential to improve health across the life course and for subsequent generations,” they add.
Critical research gap
Despite the evidence linking an individual’s prepregnancy health to their offspring’s health, there are no large trials to test whether improving overall cardiovascular health before pregnancy will reduce pregnancy complications, pregnancy-related cardiovascular death, or cardiovascular risk for offspring. The statement authors suggest that such a trial should be considered.
“This would be a big undertaking, but it could be feasible and could be really impactful,” Dr. Khan said. “Of course it would be challenging to recruit women who are planning a pregnancy and to follow them to see if they do get pregnant and consider interventions and outcomes, but given the importance of the need, we think this is something that should be invested in.”
She pointed out that the main way to improve the cardiovascular health of this cohort would be through behavioral counseling on physical activity and diet. “We need to develop strategies tailored to this age group – young women and those who may already have young children – and often the last thing they are thinking about is themselves and their own health.”
She explained that while it is presumed that controlling cardiovascular risk factors will be beneficial, the bigger question is how that can be achieved. “Behavioral interventions are difficult to achieve and often have low adherence, so the focus of the trials should be on strategies on how to deliver behavioral counseling to achieve better cardiovascular health in this population.”
Dr. Khan stressed that any approaches to improving prepregnancy cardiovascular health must address the current racial disparities that are present. “We must make sure that our policies are successful not just in improving cardiovascular health but to ensure it is done equitably. We must find ways to ensure all individuals can access care.”
A version of this article first appeared on Medscape.com.
Increased public health and research efforts to optimize prepregnancy cardiovascular health are needed, particularly among those in under-represented racial and ethnic groups, according to a new scientific statement from the American Heart Association.
“We have released this statement at this time because there is a maternal health crisis in the U.S. with rising maternal morbidity and mortality rates, which are the highest among high-income countries,” chair of the scientific statement writing group, Sadiya S. Khan, MD, told this news organization.
Cardiovascular disease (CVD) is the leading cause of death during pregnancy and the postpartum period and represents 26.5% of pregnancy-related deaths, the statement reports.
“While there is a lot of emphasis in trying to reduce cardiovascular risk during the period of actual pregnancy, much of that risk has often already developed and the women have been living with it for some time, so interventions during pregnancy may be too late,” Dr. Khan, assistant professor of medicine and preventive medicine at Northwestern University, Chicago, said.
“We wanted to try and emphasize the importance of starting to reduce cardiovascular risk earlier before pregnancy. In terms of improving cardiovascular health, this should have benefits both for the mother and the child,” she added.
The statement, “Optimizing Prepregnancy Cardiovascular Health to Improve Outcomes in Pregnant and Postpartum Individuals and Offspring” was published online in a “Go Red For Women” spotlight issue of the AHA publication Circulation.
Currently, nearly one in five births are complicated by such an adverse pregnancy outcome, and there is a strong association between these complications and risk for subsequent cardiovascular disease.
Prepregnancy window
Over the past decade, rates of adverse pregnancy outcomes have increased significantly in the United States, with a near doubling in rates of hypertensive disorders of pregnancy, and there are persistent disparities, with Black individuals significantly more likely to experience adverse pregnancy outcomes, the statement notes.
Emerging data suggest that these complications have, at least in part, prepregnancy origins. Thus, the prepregnancy period may be a critical window during which interventions have a great potential for benefit in both women and their offspring, it says.
The authors suggest a life-course approach to measure, modify, and monitor prepregnancy cardiovascular health, with all clinicians who interact with pregnancy-capable individuals emphasizing optimization of cardiovascular health beginning early in childhood.
“Leveraging these opportunities to target cardiovascular health has the potential to improve health across the life course and for subsequent generations,” they add.
Critical research gap
Despite the evidence linking an individual’s prepregnancy health to their offspring’s health, there are no large trials to test whether improving overall cardiovascular health before pregnancy will reduce pregnancy complications, pregnancy-related cardiovascular death, or cardiovascular risk for offspring. The statement authors suggest that such a trial should be considered.
“This would be a big undertaking, but it could be feasible and could be really impactful,” Dr. Khan said. “Of course it would be challenging to recruit women who are planning a pregnancy and to follow them to see if they do get pregnant and consider interventions and outcomes, but given the importance of the need, we think this is something that should be invested in.”
She pointed out that the main way to improve the cardiovascular health of this cohort would be through behavioral counseling on physical activity and diet. “We need to develop strategies tailored to this age group – young women and those who may already have young children – and often the last thing they are thinking about is themselves and their own health.”
She explained that while it is presumed that controlling cardiovascular risk factors will be beneficial, the bigger question is how that can be achieved. “Behavioral interventions are difficult to achieve and often have low adherence, so the focus of the trials should be on strategies on how to deliver behavioral counseling to achieve better cardiovascular health in this population.”
Dr. Khan stressed that any approaches to improving prepregnancy cardiovascular health must address the current racial disparities that are present. “We must make sure that our policies are successful not just in improving cardiovascular health but to ensure it is done equitably. We must find ways to ensure all individuals can access care.”
A version of this article first appeared on Medscape.com.
Increased public health and research efforts to optimize prepregnancy cardiovascular health are needed, particularly among those in under-represented racial and ethnic groups, according to a new scientific statement from the American Heart Association.
“We have released this statement at this time because there is a maternal health crisis in the U.S. with rising maternal morbidity and mortality rates, which are the highest among high-income countries,” chair of the scientific statement writing group, Sadiya S. Khan, MD, told this news organization.
Cardiovascular disease (CVD) is the leading cause of death during pregnancy and the postpartum period and represents 26.5% of pregnancy-related deaths, the statement reports.
“While there is a lot of emphasis in trying to reduce cardiovascular risk during the period of actual pregnancy, much of that risk has often already developed and the women have been living with it for some time, so interventions during pregnancy may be too late,” Dr. Khan, assistant professor of medicine and preventive medicine at Northwestern University, Chicago, said.
“We wanted to try and emphasize the importance of starting to reduce cardiovascular risk earlier before pregnancy. In terms of improving cardiovascular health, this should have benefits both for the mother and the child,” she added.
The statement, “Optimizing Prepregnancy Cardiovascular Health to Improve Outcomes in Pregnant and Postpartum Individuals and Offspring” was published online in a “Go Red For Women” spotlight issue of the AHA publication Circulation.
Currently, nearly one in five births are complicated by such an adverse pregnancy outcome, and there is a strong association between these complications and risk for subsequent cardiovascular disease.
Prepregnancy window
Over the past decade, rates of adverse pregnancy outcomes have increased significantly in the United States, with a near doubling in rates of hypertensive disorders of pregnancy, and there are persistent disparities, with Black individuals significantly more likely to experience adverse pregnancy outcomes, the statement notes.
Emerging data suggest that these complications have, at least in part, prepregnancy origins. Thus, the prepregnancy period may be a critical window during which interventions have a great potential for benefit in both women and their offspring, it says.
The authors suggest a life-course approach to measure, modify, and monitor prepregnancy cardiovascular health, with all clinicians who interact with pregnancy-capable individuals emphasizing optimization of cardiovascular health beginning early in childhood.
“Leveraging these opportunities to target cardiovascular health has the potential to improve health across the life course and for subsequent generations,” they add.
Critical research gap
Despite the evidence linking an individual’s prepregnancy health to their offspring’s health, there are no large trials to test whether improving overall cardiovascular health before pregnancy will reduce pregnancy complications, pregnancy-related cardiovascular death, or cardiovascular risk for offspring. The statement authors suggest that such a trial should be considered.
“This would be a big undertaking, but it could be feasible and could be really impactful,” Dr. Khan said. “Of course it would be challenging to recruit women who are planning a pregnancy and to follow them to see if they do get pregnant and consider interventions and outcomes, but given the importance of the need, we think this is something that should be invested in.”
She pointed out that the main way to improve the cardiovascular health of this cohort would be through behavioral counseling on physical activity and diet. “We need to develop strategies tailored to this age group – young women and those who may already have young children – and often the last thing they are thinking about is themselves and their own health.”
She explained that while it is presumed that controlling cardiovascular risk factors will be beneficial, the bigger question is how that can be achieved. “Behavioral interventions are difficult to achieve and often have low adherence, so the focus of the trials should be on strategies on how to deliver behavioral counseling to achieve better cardiovascular health in this population.”
Dr. Khan stressed that any approaches to improving prepregnancy cardiovascular health must address the current racial disparities that are present. “We must make sure that our policies are successful not just in improving cardiovascular health but to ensure it is done equitably. We must find ways to ensure all individuals can access care.”
A version of this article first appeared on Medscape.com.
FROM CIRCULATION
Myths about smoking, diet, alcohol, and cancer persist
FRANCE – Conducted every 5 years since 2005, the Cancer Survey documents the knowledge, perceptions, and way of life of the French people in relation to cancer. The researchers analyzed responses to telephone interviews of a representative sample of almost 5,000 individuals aged 15-85 years.
This study shows how thinking has changed over time and how difficult it is to alter preconceived notions.
Is cancer hereditary?
The report shows that 67.7% of respondents believe that cancer is a hereditary disease. Respondents were asked to explain their answer. “Data show that medical practices for cancer treatment substantiate this belief [that cancer is hereditary],” wrote the authors of the report.
“Indeed, health care professionals almost systematically ask questions about family history of breast cancer and, when a family member has been diagnosed with cancer, medical monitoring of other family members is often sought out, thus reinforcing the belief that cancer is hereditary,” they said.
Furthermore, there seems to be confusion regarding the role of genes in the development of cancer. A person can inherit cancer-predisposing genes, not cancer itself. The authors highlighted their concern that this confusion may “lead people to think that prevention measures are unnecessary because cancer is inherited.”
Misconceptions about smoking
About 41% of smokers think that the length of time one has been smoking is the biggest determining factor for developing cancer; 58.1% think the number of cigarettes smoked per day has a bigger impact.
Experts at InCA and SPF put the debate to rest, stating that prolonged exposure to carcinogenic substances is far more toxic. As for the danger threshold concerning the number of cigarettes smoked per day, respondents believed this to be 9.2 cigarettes per day, on average. They believed that the danger threshold for the number of years as an active smoker is 13.4, on average.
“The [survey] respondents clearly understand that smoking carries a risk, but many smokers think that light smoking or smoking for a short period of time doesn’t carry any risks.” Yet it is understood that even occasional tobacco consumption increases mortality.
This was not the only misconception regarding smoking and its relationship with cancer. About 34% of survey respondents agreed with the following statement: “Smoking doesn’t cause cancer unless you’re a heavy smoker and have smoked for a long time.” Furthermore, 43.3% agreed with the statement, “Pollution is more likely to cause cancer than smoking,” 54.6% think that “exercising cleans your lungs of tobacco,” and 61.6% think that “a smoker can prevent developing cancer caused by smoking if they know to quit on time.”
Overweight and obesity
Although diet and excess weight represent the third and fourth biggest avoidable cancer risk factors, after smoking and alcohol, only 30% of survey respondents knew of this link.
“Among the causes of cancer known and cited by respondents without prompting, excessive weight and obesity were mentioned only 100 times out of 12,558 responses,” highlighted the authors of the report. The explanation put forward by the authors is that discourse about diet has been more focused on diet as a protective health factor, especially in preventing cardiovascular diseases. “The link between cancer and diet is less prominent in the public space,” they noted.
Breastfeeding and cancer
About 63% of survey respondents, which for the first time included both women and men, believe that breastfeeding does not affect mothers’ risk of breast cancer, but this is a misconception. And almost 1 in 3 respondents said that breastfeeding provides health benefits for the mother.
Artificial UV rays
Exposure to UV rays, whether of natural or artificial origin, is a major risk factor for skin cancer. However, 1 in 5 people (20.9%) think that a session in a tanning bed is less harmful than sun exposure.
Daily stress
Regarding psychological factors linked to cancer, the authors noted that risk factors not supported by scientific evidence were, ironically, cited more often by respondents than proven risk factors. There is a real knowledge gap between scientific data and the beliefs of the French people. For example, “working at night” is largely not seen as a risk factor, but data show that it presents a clear risk. However, “not being able to express one’s feelings,” “having been weakened by traumatic experiences,” and “being exposed to the stress of modern life” are seen as risk factors of cancer, without any scientific evidence.
Cigarettes and e-cigarettes
About 53% of respondents agreed that “e-cigarettes are just as harmful or more harmful than traditional cigarettes.” Nicotine and the flavors in e-cigarettes are largely perceived as “very” or “extremely” harmful to the health of a person. However, the authors note that “no published study on nicotine substitutes has shown harmful effects on the health of a person, let alone determined it a risk factor for cancer. The nicotine doses in e-cigarettes are similar to traditional nicotine substitutes, and no cytotoxic effect of nicotine in its inhaled form has been found.” There seems to be confusion between dependence and risk of cancer.
Alcohol consumption
Eight of 10 respondents believe that “some people can drink a lot of alcohol all their life without ever getting cancer,” which goes against the scientific literature. The authors of the report state that the negative effects of alcohol on health seem poorly understood. Although alcohol is the second biggest cause of cancer, only a third of survey respondents cited it without having been prompted as one of the main causes of cancer. And 23.5% even think that “in terms of decreasing your risk of cancer, it’s better to drink a little wine than to drink no wine at all.”
This article was translated from the Medscape French edition. A version of this article appeared on Medscape.com.
FRANCE – Conducted every 5 years since 2005, the Cancer Survey documents the knowledge, perceptions, and way of life of the French people in relation to cancer. The researchers analyzed responses to telephone interviews of a representative sample of almost 5,000 individuals aged 15-85 years.
This study shows how thinking has changed over time and how difficult it is to alter preconceived notions.
Is cancer hereditary?
The report shows that 67.7% of respondents believe that cancer is a hereditary disease. Respondents were asked to explain their answer. “Data show that medical practices for cancer treatment substantiate this belief [that cancer is hereditary],” wrote the authors of the report.
“Indeed, health care professionals almost systematically ask questions about family history of breast cancer and, when a family member has been diagnosed with cancer, medical monitoring of other family members is often sought out, thus reinforcing the belief that cancer is hereditary,” they said.
Furthermore, there seems to be confusion regarding the role of genes in the development of cancer. A person can inherit cancer-predisposing genes, not cancer itself. The authors highlighted their concern that this confusion may “lead people to think that prevention measures are unnecessary because cancer is inherited.”
Misconceptions about smoking
About 41% of smokers think that the length of time one has been smoking is the biggest determining factor for developing cancer; 58.1% think the number of cigarettes smoked per day has a bigger impact.
Experts at InCA and SPF put the debate to rest, stating that prolonged exposure to carcinogenic substances is far more toxic. As for the danger threshold concerning the number of cigarettes smoked per day, respondents believed this to be 9.2 cigarettes per day, on average. They believed that the danger threshold for the number of years as an active smoker is 13.4, on average.
“The [survey] respondents clearly understand that smoking carries a risk, but many smokers think that light smoking or smoking for a short period of time doesn’t carry any risks.” Yet it is understood that even occasional tobacco consumption increases mortality.
This was not the only misconception regarding smoking and its relationship with cancer. About 34% of survey respondents agreed with the following statement: “Smoking doesn’t cause cancer unless you’re a heavy smoker and have smoked for a long time.” Furthermore, 43.3% agreed with the statement, “Pollution is more likely to cause cancer than smoking,” 54.6% think that “exercising cleans your lungs of tobacco,” and 61.6% think that “a smoker can prevent developing cancer caused by smoking if they know to quit on time.”
Overweight and obesity
Although diet and excess weight represent the third and fourth biggest avoidable cancer risk factors, after smoking and alcohol, only 30% of survey respondents knew of this link.
“Among the causes of cancer known and cited by respondents without prompting, excessive weight and obesity were mentioned only 100 times out of 12,558 responses,” highlighted the authors of the report. The explanation put forward by the authors is that discourse about diet has been more focused on diet as a protective health factor, especially in preventing cardiovascular diseases. “The link between cancer and diet is less prominent in the public space,” they noted.
Breastfeeding and cancer
About 63% of survey respondents, which for the first time included both women and men, believe that breastfeeding does not affect mothers’ risk of breast cancer, but this is a misconception. And almost 1 in 3 respondents said that breastfeeding provides health benefits for the mother.
Artificial UV rays
Exposure to UV rays, whether of natural or artificial origin, is a major risk factor for skin cancer. However, 1 in 5 people (20.9%) think that a session in a tanning bed is less harmful than sun exposure.
Daily stress
Regarding psychological factors linked to cancer, the authors noted that risk factors not supported by scientific evidence were, ironically, cited more often by respondents than proven risk factors. There is a real knowledge gap between scientific data and the beliefs of the French people. For example, “working at night” is largely not seen as a risk factor, but data show that it presents a clear risk. However, “not being able to express one’s feelings,” “having been weakened by traumatic experiences,” and “being exposed to the stress of modern life” are seen as risk factors of cancer, without any scientific evidence.
Cigarettes and e-cigarettes
About 53% of respondents agreed that “e-cigarettes are just as harmful or more harmful than traditional cigarettes.” Nicotine and the flavors in e-cigarettes are largely perceived as “very” or “extremely” harmful to the health of a person. However, the authors note that “no published study on nicotine substitutes has shown harmful effects on the health of a person, let alone determined it a risk factor for cancer. The nicotine doses in e-cigarettes are similar to traditional nicotine substitutes, and no cytotoxic effect of nicotine in its inhaled form has been found.” There seems to be confusion between dependence and risk of cancer.
Alcohol consumption
Eight of 10 respondents believe that “some people can drink a lot of alcohol all their life without ever getting cancer,” which goes against the scientific literature. The authors of the report state that the negative effects of alcohol on health seem poorly understood. Although alcohol is the second biggest cause of cancer, only a third of survey respondents cited it without having been prompted as one of the main causes of cancer. And 23.5% even think that “in terms of decreasing your risk of cancer, it’s better to drink a little wine than to drink no wine at all.”
This article was translated from the Medscape French edition. A version of this article appeared on Medscape.com.
FRANCE – Conducted every 5 years since 2005, the Cancer Survey documents the knowledge, perceptions, and way of life of the French people in relation to cancer. The researchers analyzed responses to telephone interviews of a representative sample of almost 5,000 individuals aged 15-85 years.
This study shows how thinking has changed over time and how difficult it is to alter preconceived notions.
Is cancer hereditary?
The report shows that 67.7% of respondents believe that cancer is a hereditary disease. Respondents were asked to explain their answer. “Data show that medical practices for cancer treatment substantiate this belief [that cancer is hereditary],” wrote the authors of the report.
“Indeed, health care professionals almost systematically ask questions about family history of breast cancer and, when a family member has been diagnosed with cancer, medical monitoring of other family members is often sought out, thus reinforcing the belief that cancer is hereditary,” they said.
Furthermore, there seems to be confusion regarding the role of genes in the development of cancer. A person can inherit cancer-predisposing genes, not cancer itself. The authors highlighted their concern that this confusion may “lead people to think that prevention measures are unnecessary because cancer is inherited.”
Misconceptions about smoking
About 41% of smokers think that the length of time one has been smoking is the biggest determining factor for developing cancer; 58.1% think the number of cigarettes smoked per day has a bigger impact.
Experts at InCA and SPF put the debate to rest, stating that prolonged exposure to carcinogenic substances is far more toxic. As for the danger threshold concerning the number of cigarettes smoked per day, respondents believed this to be 9.2 cigarettes per day, on average. They believed that the danger threshold for the number of years as an active smoker is 13.4, on average.
“The [survey] respondents clearly understand that smoking carries a risk, but many smokers think that light smoking or smoking for a short period of time doesn’t carry any risks.” Yet it is understood that even occasional tobacco consumption increases mortality.
This was not the only misconception regarding smoking and its relationship with cancer. About 34% of survey respondents agreed with the following statement: “Smoking doesn’t cause cancer unless you’re a heavy smoker and have smoked for a long time.” Furthermore, 43.3% agreed with the statement, “Pollution is more likely to cause cancer than smoking,” 54.6% think that “exercising cleans your lungs of tobacco,” and 61.6% think that “a smoker can prevent developing cancer caused by smoking if they know to quit on time.”
Overweight and obesity
Although diet and excess weight represent the third and fourth biggest avoidable cancer risk factors, after smoking and alcohol, only 30% of survey respondents knew of this link.
“Among the causes of cancer known and cited by respondents without prompting, excessive weight and obesity were mentioned only 100 times out of 12,558 responses,” highlighted the authors of the report. The explanation put forward by the authors is that discourse about diet has been more focused on diet as a protective health factor, especially in preventing cardiovascular diseases. “The link between cancer and diet is less prominent in the public space,” they noted.
Breastfeeding and cancer
About 63% of survey respondents, which for the first time included both women and men, believe that breastfeeding does not affect mothers’ risk of breast cancer, but this is a misconception. And almost 1 in 3 respondents said that breastfeeding provides health benefits for the mother.
Artificial UV rays
Exposure to UV rays, whether of natural or artificial origin, is a major risk factor for skin cancer. However, 1 in 5 people (20.9%) think that a session in a tanning bed is less harmful than sun exposure.
Daily stress
Regarding psychological factors linked to cancer, the authors noted that risk factors not supported by scientific evidence were, ironically, cited more often by respondents than proven risk factors. There is a real knowledge gap between scientific data and the beliefs of the French people. For example, “working at night” is largely not seen as a risk factor, but data show that it presents a clear risk. However, “not being able to express one’s feelings,” “having been weakened by traumatic experiences,” and “being exposed to the stress of modern life” are seen as risk factors of cancer, without any scientific evidence.
Cigarettes and e-cigarettes
About 53% of respondents agreed that “e-cigarettes are just as harmful or more harmful than traditional cigarettes.” Nicotine and the flavors in e-cigarettes are largely perceived as “very” or “extremely” harmful to the health of a person. However, the authors note that “no published study on nicotine substitutes has shown harmful effects on the health of a person, let alone determined it a risk factor for cancer. The nicotine doses in e-cigarettes are similar to traditional nicotine substitutes, and no cytotoxic effect of nicotine in its inhaled form has been found.” There seems to be confusion between dependence and risk of cancer.
Alcohol consumption
Eight of 10 respondents believe that “some people can drink a lot of alcohol all their life without ever getting cancer,” which goes against the scientific literature. The authors of the report state that the negative effects of alcohol on health seem poorly understood. Although alcohol is the second biggest cause of cancer, only a third of survey respondents cited it without having been prompted as one of the main causes of cancer. And 23.5% even think that “in terms of decreasing your risk of cancer, it’s better to drink a little wine than to drink no wine at all.”
This article was translated from the Medscape French edition. A version of this article appeared on Medscape.com.
Toxic chemicals we consume without knowing it
Life expectancy is falling precipitously. Three-fourths of Americans are overweight or obese, half have diabetes or prediabetes, and a majority are metabolically unhealthy. Furthermore, the rates of allergic, inflammatory, and autoimmune diseases are rising at rates of 3%-9% per year in the West, far faster than the speed of genetic change in this population.
Of course, diet and lifestyle are major factors behind such trends, but a grossly underappreciated driver in what ails us is the role of environmental toxins and endocrine-disrupting chemicals. In years past, these factors have largely evaded the traditional Western medical establishment; however, mounting evidence now supports their significance in fertility, metabolic health, and cancer.
Although several industrial chemicals and toxins have been identified as carcinogens and have subsequently been regulated, many more remain persistent in the environment and continue to be freely used. It is therefore incumbent upon both the general public and clinicians to be knowledgeable about these exposures. Here, we review some of the most common exposures and the substantial health risks associated with them, along with some general guidance around best practices for how to minimize exposure.
Microplastics
“Microplastics” is a term used to describe small fragments or particles of plastic breakdown or microbeads from household or personal care products, measuring less than 5 mm in length.
Plastic waste is accumulating at alarming and devastating proportions – by 2050, it is estimated that by weight, there will be more plastic than fish in the oceans. That translates into hundreds of thousands of tons of microplastics and trillions of these particles in the seas. A recent study demonstrated that microplastics were present in the bloodstream in the majority of 22 otherwise healthy participants.
Since the 1950s, plastic exposure has been shown to promote tumorigenesis in animal studies, and in vitro studies have demonstrated the toxicity of microplastics at the cellular level. However, it is not well known whether the plastic itself is toxic or if it simply serves as a carrier for other environmental toxins to bioaccumulate.
According to Tasha Stoiber, a senior scientist at the Environmental Working Group, “Microplastics have been widely detected in fish and seafood, as well as other products like bottled water, beer, honey, and tap water.” The EWG states there are no formal advisories on fish consumption to avoid exposure to microplastics at the moment.
Pressure also is mounting for a ban on microbeads in personal care products.
Until such bans are put in place, it is advised to avoid single-use plastics, favor reusable tote bags for grocery shopping rather than plastic bags, and opt for loose leaf tea or paper tea bags rather than mesh-based alternatives.
Phthalates
Phthalates are chemicals used to make plastics soft and durable, as well as to bind fragrances. They are commonly found in household items such as vinyl (for example, flooring, shower curtains) and fragrances, air fresheners, and perfumes.
Phthalates are known hormone-disrupting chemicals, exposure to which has been associated with abnormal sexual and brain development in children, as well as lower levels of testosterone in men. Exposures are thought to occur via inhalation, ingestion, and skin contact; however, fasting studies demonstrate that a majority of exposure is probably food related.
To avoid phthalate exposures, recommendations include avoiding polyvinyl chloride plastics (particularly food containers, plastic wrap, and children’s toys), which are identifiable by the recycle code number 3, as well as air fresheners and fragranced products.
The EWG’s Skin Deep database provides an important resource on phthalate-free personal care products.
Despite pressure from consumer advocacy groups, the U.S. Food and Drug Administration has not yet banned phthalates in food packaging.
Bisphenol A (BPA)
BPA is a chemical additive used to make clear and hard polycarbonate plastics, as well as epoxy and thermal papers. BPA is one of the highest-volume chemicals, with roughly 6 billion pounds produced each year. BPA is traditionally found in many clear plastic bottles and sippy cups, as well as in the lining of canned foods.
Structurally, BPA acts as an estrogen mimetic and has been associated with cardiovascular disease, obesity, and male sexual dysfunction. Since 2012, BPA has been banned in sippy cups and baby bottles, but there is some debate as to whether its replacements (bisphenol S and bisphenol F) are any safer; they appear to have similar hormonal effects as BPA.
As with phthalates, the majority of ingestion is thought to be food related. BPA has been found in more than 90% of a representative study population in the United States.
Guidance advises avoiding polycarbonate plastics (identifiable with the recycling code number 7), as well as avoiding handling thermal papers such as tickets and receipts, if possible. Food and beverages should be stored in glass or stainless steel. If plastic must be used, opt for polycarbonate- and polyvinyl chloride–free plastics, and food and beverages should never be reheated in plastic containers or wrapping. Canned foods should ideally be avoided, particularly canned tunas and condensed soups. If canned products are bought, they should ideally be BPA free.
Dioxins and polychlorinated biphenyls (PCBs)
Dioxins are mainly the byproducts of industrial practices; they are released after incineration, trash burning, and fires. PCBs, which are somewhat structurally related to dioxins, were previously found in products such as flame retardants and coolants. Dioxins and PCBs are often grouped in the same category under the umbrella term “persistent organic pollutants” because they break down slowly and remain in the environment even after emissions have been curbed.
Tetrachlorodibenzodioxin, perhaps the best-known dioxin, is a known carcinogen. Dioxins also have been associated with a host of health implications in development, immunity, and reproductive and endocrine systems. Higher levels of PCB exposure have also been associated with an increased risk for mortality from cardiovascular disease.
Notably, dioxin emissions have been reduced by 90% since the 1980s, and the U.S. Environmental Protection Agency has banned the use of PCBs in industrial manufacturing since 1979. However, environmental dioxins and PCBs still enter the food chain and accumulate in fat.
The best ways to avoid exposures are through limiting meat, fish, and dairy consumption and trimming the skin and fat from meats. The level of dioxins and PCBs found in meat, eggs, fish, and dairy are approximately 5-10 times higher than they are in plant-based foods. Research has shown that farmed salmon is likely to be the most PCB-contaminated protein source in the U.S. diet; however, newer forms of land-based and sustainable aquaculture probably avoid this exposure.
Pesticides
The growth of modern monoculture agriculture in the United States over the past century has coincided with a dramatic surge in the use of industrial pesticides. In fact, over 90% of the U.S. population have pesticides in their urine and blood, regardless of where they live. Exposures are thought to be food related.
Approximately 1 billion pounds of pesticides are used annually in the United States, including nearly 300 million pounds of glyphosate, which has been identified as a probable carcinogen by European agencies. The EPA has not yet reached this conclusion, although the matter is currently being litigated.
A large European prospective cohort trial demonstrated a lower risk for cancer in those with a greater frequency of self-reported organic food consumption. In addition to cancer risk, relatively elevated blood levels of a pesticide known as beta-hexachlorocyclohexane (B-HCH) are associated with higher all-cause mortality. Also, exposure to DDE – a metabolite of DDT, a chlorinated pesticide heavily used in the 1940s-1960s that still persists in the environment today – has been shown to increase the risk for Alzheimer’s-type dementia as well as overall cognitive decline.
Because these chlorinated pesticides are often fat soluble, they seem to accumulate in animal products. Therefore, people consuming a vegetarian diet have been found to have lower levels of B-HCH. This has led to the recommendation that consumers of produce should favor organic over conventional, if possible. Here too, the EWG provides an important resource to consumers in the form of shopper guides regarding pesticides in produce.
Per- and polyfluoroalkyl substances (PFAS)
PFAS are a group of fluorinated compounds discovered in the 1930s. Their chemical composition includes a durable carbon-fluoride bond, giving them a persistence within the environment that has led to their being referred to as “forever chemicals.”
PFAS have been detected in the blood of 98% of Americans, and in the rainwater of locations as far afield as Tibet and Antarctica. Even low levels of exposure have been associated with an increased risk for cancer, liver disease, low birth weight, and hormonal disruption.
The properties of PFAS also make them both durable at very high heat and water repellent. Notoriously, the chemical was used by 3M to make Scotchgard for carpets and fabrics and by Dupont to make Teflon for nonstick coating of pots and pans. Although perfluorooctanoic acid (PFOA) was removed from nonstick cookware in 2013, PFAS – a family of thousands of synthetic compounds – remain common in fast-food packaging, water- and stain-repellent clothing, firefighting foam, and personal care products. PFAS are released into the environment during the breakdown of these consumer and industrial products, as well as from dumping from waste facilities.
Alarmingly, the EWG notes that up to 200 million Americans may be exposed to PFAS in their drinking water. In March 2021, the EPA announced that they will be regulating PFAS in drinking water; however, the regulations have not been finalized. Currently, it is up to individual states to test for its presence in the water. The EWG has compiled a map of all known PFAS contamination sites.
To avoid or prevent exposures from PFAS, recommendations include filtering tap water with either reverse osmosis or activated carbon filters, as well as avoiding fast food and carry-out food, if possible, and consumer products labeled as “water resistant,” “stain-resistant,” and “nonstick.”
In a testament to how harmful these chemicals are, the EPA recently revised their lifetime health advisories for PFAS, such as PFOA, to 0.004 parts per trillion, which is more than 10,000 times smaller than the previous limit of 70 parts per trillion. The EPA also has proposed formally designating certain PFAS chemicals as “hazardous substances.”
Dr. Goel, clinical assistant professor of medicine at Weill Cornell Medicine, New York, has disclosed no relevant financial relationships. A version of this article originally appeared on Medscape.com.
Life expectancy is falling precipitously. Three-fourths of Americans are overweight or obese, half have diabetes or prediabetes, and a majority are metabolically unhealthy. Furthermore, the rates of allergic, inflammatory, and autoimmune diseases are rising at rates of 3%-9% per year in the West, far faster than the speed of genetic change in this population.
Of course, diet and lifestyle are major factors behind such trends, but a grossly underappreciated driver in what ails us is the role of environmental toxins and endocrine-disrupting chemicals. In years past, these factors have largely evaded the traditional Western medical establishment; however, mounting evidence now supports their significance in fertility, metabolic health, and cancer.
Although several industrial chemicals and toxins have been identified as carcinogens and have subsequently been regulated, many more remain persistent in the environment and continue to be freely used. It is therefore incumbent upon both the general public and clinicians to be knowledgeable about these exposures. Here, we review some of the most common exposures and the substantial health risks associated with them, along with some general guidance around best practices for how to minimize exposure.
Microplastics
“Microplastics” is a term used to describe small fragments or particles of plastic breakdown or microbeads from household or personal care products, measuring less than 5 mm in length.
Plastic waste is accumulating at alarming and devastating proportions – by 2050, it is estimated that by weight, there will be more plastic than fish in the oceans. That translates into hundreds of thousands of tons of microplastics and trillions of these particles in the seas. A recent study demonstrated that microplastics were present in the bloodstream in the majority of 22 otherwise healthy participants.
Since the 1950s, plastic exposure has been shown to promote tumorigenesis in animal studies, and in vitro studies have demonstrated the toxicity of microplastics at the cellular level. However, it is not well known whether the plastic itself is toxic or if it simply serves as a carrier for other environmental toxins to bioaccumulate.
According to Tasha Stoiber, a senior scientist at the Environmental Working Group, “Microplastics have been widely detected in fish and seafood, as well as other products like bottled water, beer, honey, and tap water.” The EWG states there are no formal advisories on fish consumption to avoid exposure to microplastics at the moment.
Pressure also is mounting for a ban on microbeads in personal care products.
Until such bans are put in place, it is advised to avoid single-use plastics, favor reusable tote bags for grocery shopping rather than plastic bags, and opt for loose leaf tea or paper tea bags rather than mesh-based alternatives.
Phthalates
Phthalates are chemicals used to make plastics soft and durable, as well as to bind fragrances. They are commonly found in household items such as vinyl (for example, flooring, shower curtains) and fragrances, air fresheners, and perfumes.
Phthalates are known hormone-disrupting chemicals, exposure to which has been associated with abnormal sexual and brain development in children, as well as lower levels of testosterone in men. Exposures are thought to occur via inhalation, ingestion, and skin contact; however, fasting studies demonstrate that a majority of exposure is probably food related.
To avoid phthalate exposures, recommendations include avoiding polyvinyl chloride plastics (particularly food containers, plastic wrap, and children’s toys), which are identifiable by the recycle code number 3, as well as air fresheners and fragranced products.
The EWG’s Skin Deep database provides an important resource on phthalate-free personal care products.
Despite pressure from consumer advocacy groups, the U.S. Food and Drug Administration has not yet banned phthalates in food packaging.
Bisphenol A (BPA)
BPA is a chemical additive used to make clear and hard polycarbonate plastics, as well as epoxy and thermal papers. BPA is one of the highest-volume chemicals, with roughly 6 billion pounds produced each year. BPA is traditionally found in many clear plastic bottles and sippy cups, as well as in the lining of canned foods.
Structurally, BPA acts as an estrogen mimetic and has been associated with cardiovascular disease, obesity, and male sexual dysfunction. Since 2012, BPA has been banned in sippy cups and baby bottles, but there is some debate as to whether its replacements (bisphenol S and bisphenol F) are any safer; they appear to have similar hormonal effects as BPA.
As with phthalates, the majority of ingestion is thought to be food related. BPA has been found in more than 90% of a representative study population in the United States.
Guidance advises avoiding polycarbonate plastics (identifiable with the recycling code number 7), as well as avoiding handling thermal papers such as tickets and receipts, if possible. Food and beverages should be stored in glass or stainless steel. If plastic must be used, opt for polycarbonate- and polyvinyl chloride–free plastics, and food and beverages should never be reheated in plastic containers or wrapping. Canned foods should ideally be avoided, particularly canned tunas and condensed soups. If canned products are bought, they should ideally be BPA free.
Dioxins and polychlorinated biphenyls (PCBs)
Dioxins are mainly the byproducts of industrial practices; they are released after incineration, trash burning, and fires. PCBs, which are somewhat structurally related to dioxins, were previously found in products such as flame retardants and coolants. Dioxins and PCBs are often grouped in the same category under the umbrella term “persistent organic pollutants” because they break down slowly and remain in the environment even after emissions have been curbed.
Tetrachlorodibenzodioxin, perhaps the best-known dioxin, is a known carcinogen. Dioxins also have been associated with a host of health implications in development, immunity, and reproductive and endocrine systems. Higher levels of PCB exposure have also been associated with an increased risk for mortality from cardiovascular disease.
Notably, dioxin emissions have been reduced by 90% since the 1980s, and the U.S. Environmental Protection Agency has banned the use of PCBs in industrial manufacturing since 1979. However, environmental dioxins and PCBs still enter the food chain and accumulate in fat.
The best ways to avoid exposures are through limiting meat, fish, and dairy consumption and trimming the skin and fat from meats. The level of dioxins and PCBs found in meat, eggs, fish, and dairy are approximately 5-10 times higher than they are in plant-based foods. Research has shown that farmed salmon is likely to be the most PCB-contaminated protein source in the U.S. diet; however, newer forms of land-based and sustainable aquaculture probably avoid this exposure.
Pesticides
The growth of modern monoculture agriculture in the United States over the past century has coincided with a dramatic surge in the use of industrial pesticides. In fact, over 90% of the U.S. population have pesticides in their urine and blood, regardless of where they live. Exposures are thought to be food related.
Approximately 1 billion pounds of pesticides are used annually in the United States, including nearly 300 million pounds of glyphosate, which has been identified as a probable carcinogen by European agencies. The EPA has not yet reached this conclusion, although the matter is currently being litigated.
A large European prospective cohort trial demonstrated a lower risk for cancer in those with a greater frequency of self-reported organic food consumption. In addition to cancer risk, relatively elevated blood levels of a pesticide known as beta-hexachlorocyclohexane (B-HCH) are associated with higher all-cause mortality. Also, exposure to DDE – a metabolite of DDT, a chlorinated pesticide heavily used in the 1940s-1960s that still persists in the environment today – has been shown to increase the risk for Alzheimer’s-type dementia as well as overall cognitive decline.
Because these chlorinated pesticides are often fat soluble, they seem to accumulate in animal products. Therefore, people consuming a vegetarian diet have been found to have lower levels of B-HCH. This has led to the recommendation that consumers of produce should favor organic over conventional, if possible. Here too, the EWG provides an important resource to consumers in the form of shopper guides regarding pesticides in produce.
Per- and polyfluoroalkyl substances (PFAS)
PFAS are a group of fluorinated compounds discovered in the 1930s. Their chemical composition includes a durable carbon-fluoride bond, giving them a persistence within the environment that has led to their being referred to as “forever chemicals.”
PFAS have been detected in the blood of 98% of Americans, and in the rainwater of locations as far afield as Tibet and Antarctica. Even low levels of exposure have been associated with an increased risk for cancer, liver disease, low birth weight, and hormonal disruption.
The properties of PFAS also make them both durable at very high heat and water repellent. Notoriously, the chemical was used by 3M to make Scotchgard for carpets and fabrics and by Dupont to make Teflon for nonstick coating of pots and pans. Although perfluorooctanoic acid (PFOA) was removed from nonstick cookware in 2013, PFAS – a family of thousands of synthetic compounds – remain common in fast-food packaging, water- and stain-repellent clothing, firefighting foam, and personal care products. PFAS are released into the environment during the breakdown of these consumer and industrial products, as well as from dumping from waste facilities.
Alarmingly, the EWG notes that up to 200 million Americans may be exposed to PFAS in their drinking water. In March 2021, the EPA announced that they will be regulating PFAS in drinking water; however, the regulations have not been finalized. Currently, it is up to individual states to test for its presence in the water. The EWG has compiled a map of all known PFAS contamination sites.
To avoid or prevent exposures from PFAS, recommendations include filtering tap water with either reverse osmosis or activated carbon filters, as well as avoiding fast food and carry-out food, if possible, and consumer products labeled as “water resistant,” “stain-resistant,” and “nonstick.”
In a testament to how harmful these chemicals are, the EPA recently revised their lifetime health advisories for PFAS, such as PFOA, to 0.004 parts per trillion, which is more than 10,000 times smaller than the previous limit of 70 parts per trillion. The EPA also has proposed formally designating certain PFAS chemicals as “hazardous substances.”
Dr. Goel, clinical assistant professor of medicine at Weill Cornell Medicine, New York, has disclosed no relevant financial relationships. A version of this article originally appeared on Medscape.com.
Life expectancy is falling precipitously. Three-fourths of Americans are overweight or obese, half have diabetes or prediabetes, and a majority are metabolically unhealthy. Furthermore, the rates of allergic, inflammatory, and autoimmune diseases are rising at rates of 3%-9% per year in the West, far faster than the speed of genetic change in this population.
Of course, diet and lifestyle are major factors behind such trends, but a grossly underappreciated driver in what ails us is the role of environmental toxins and endocrine-disrupting chemicals. In years past, these factors have largely evaded the traditional Western medical establishment; however, mounting evidence now supports their significance in fertility, metabolic health, and cancer.
Although several industrial chemicals and toxins have been identified as carcinogens and have subsequently been regulated, many more remain persistent in the environment and continue to be freely used. It is therefore incumbent upon both the general public and clinicians to be knowledgeable about these exposures. Here, we review some of the most common exposures and the substantial health risks associated with them, along with some general guidance around best practices for how to minimize exposure.
Microplastics
“Microplastics” is a term used to describe small fragments or particles of plastic breakdown or microbeads from household or personal care products, measuring less than 5 mm in length.
Plastic waste is accumulating at alarming and devastating proportions – by 2050, it is estimated that by weight, there will be more plastic than fish in the oceans. That translates into hundreds of thousands of tons of microplastics and trillions of these particles in the seas. A recent study demonstrated that microplastics were present in the bloodstream in the majority of 22 otherwise healthy participants.
Since the 1950s, plastic exposure has been shown to promote tumorigenesis in animal studies, and in vitro studies have demonstrated the toxicity of microplastics at the cellular level. However, it is not well known whether the plastic itself is toxic or if it simply serves as a carrier for other environmental toxins to bioaccumulate.
According to Tasha Stoiber, a senior scientist at the Environmental Working Group, “Microplastics have been widely detected in fish and seafood, as well as other products like bottled water, beer, honey, and tap water.” The EWG states there are no formal advisories on fish consumption to avoid exposure to microplastics at the moment.
Pressure also is mounting for a ban on microbeads in personal care products.
Until such bans are put in place, it is advised to avoid single-use plastics, favor reusable tote bags for grocery shopping rather than plastic bags, and opt for loose leaf tea or paper tea bags rather than mesh-based alternatives.
Phthalates
Phthalates are chemicals used to make plastics soft and durable, as well as to bind fragrances. They are commonly found in household items such as vinyl (for example, flooring, shower curtains) and fragrances, air fresheners, and perfumes.
Phthalates are known hormone-disrupting chemicals, exposure to which has been associated with abnormal sexual and brain development in children, as well as lower levels of testosterone in men. Exposures are thought to occur via inhalation, ingestion, and skin contact; however, fasting studies demonstrate that a majority of exposure is probably food related.
To avoid phthalate exposures, recommendations include avoiding polyvinyl chloride plastics (particularly food containers, plastic wrap, and children’s toys), which are identifiable by the recycle code number 3, as well as air fresheners and fragranced products.
The EWG’s Skin Deep database provides an important resource on phthalate-free personal care products.
Despite pressure from consumer advocacy groups, the U.S. Food and Drug Administration has not yet banned phthalates in food packaging.
Bisphenol A (BPA)
BPA is a chemical additive used to make clear and hard polycarbonate plastics, as well as epoxy and thermal papers. BPA is one of the highest-volume chemicals, with roughly 6 billion pounds produced each year. BPA is traditionally found in many clear plastic bottles and sippy cups, as well as in the lining of canned foods.
Structurally, BPA acts as an estrogen mimetic and has been associated with cardiovascular disease, obesity, and male sexual dysfunction. Since 2012, BPA has been banned in sippy cups and baby bottles, but there is some debate as to whether its replacements (bisphenol S and bisphenol F) are any safer; they appear to have similar hormonal effects as BPA.
As with phthalates, the majority of ingestion is thought to be food related. BPA has been found in more than 90% of a representative study population in the United States.
Guidance advises avoiding polycarbonate plastics (identifiable with the recycling code number 7), as well as avoiding handling thermal papers such as tickets and receipts, if possible. Food and beverages should be stored in glass or stainless steel. If plastic must be used, opt for polycarbonate- and polyvinyl chloride–free plastics, and food and beverages should never be reheated in plastic containers or wrapping. Canned foods should ideally be avoided, particularly canned tunas and condensed soups. If canned products are bought, they should ideally be BPA free.
Dioxins and polychlorinated biphenyls (PCBs)
Dioxins are mainly the byproducts of industrial practices; they are released after incineration, trash burning, and fires. PCBs, which are somewhat structurally related to dioxins, were previously found in products such as flame retardants and coolants. Dioxins and PCBs are often grouped in the same category under the umbrella term “persistent organic pollutants” because they break down slowly and remain in the environment even after emissions have been curbed.
Tetrachlorodibenzodioxin, perhaps the best-known dioxin, is a known carcinogen. Dioxins also have been associated with a host of health implications in development, immunity, and reproductive and endocrine systems. Higher levels of PCB exposure have also been associated with an increased risk for mortality from cardiovascular disease.
Notably, dioxin emissions have been reduced by 90% since the 1980s, and the U.S. Environmental Protection Agency has banned the use of PCBs in industrial manufacturing since 1979. However, environmental dioxins and PCBs still enter the food chain and accumulate in fat.
The best ways to avoid exposures are through limiting meat, fish, and dairy consumption and trimming the skin and fat from meats. The level of dioxins and PCBs found in meat, eggs, fish, and dairy are approximately 5-10 times higher than they are in plant-based foods. Research has shown that farmed salmon is likely to be the most PCB-contaminated protein source in the U.S. diet; however, newer forms of land-based and sustainable aquaculture probably avoid this exposure.
Pesticides
The growth of modern monoculture agriculture in the United States over the past century has coincided with a dramatic surge in the use of industrial pesticides. In fact, over 90% of the U.S. population have pesticides in their urine and blood, regardless of where they live. Exposures are thought to be food related.
Approximately 1 billion pounds of pesticides are used annually in the United States, including nearly 300 million pounds of glyphosate, which has been identified as a probable carcinogen by European agencies. The EPA has not yet reached this conclusion, although the matter is currently being litigated.
A large European prospective cohort trial demonstrated a lower risk for cancer in those with a greater frequency of self-reported organic food consumption. In addition to cancer risk, relatively elevated blood levels of a pesticide known as beta-hexachlorocyclohexane (B-HCH) are associated with higher all-cause mortality. Also, exposure to DDE – a metabolite of DDT, a chlorinated pesticide heavily used in the 1940s-1960s that still persists in the environment today – has been shown to increase the risk for Alzheimer’s-type dementia as well as overall cognitive decline.
Because these chlorinated pesticides are often fat soluble, they seem to accumulate in animal products. Therefore, people consuming a vegetarian diet have been found to have lower levels of B-HCH. This has led to the recommendation that consumers of produce should favor organic over conventional, if possible. Here too, the EWG provides an important resource to consumers in the form of shopper guides regarding pesticides in produce.
Per- and polyfluoroalkyl substances (PFAS)
PFAS are a group of fluorinated compounds discovered in the 1930s. Their chemical composition includes a durable carbon-fluoride bond, giving them a persistence within the environment that has led to their being referred to as “forever chemicals.”
PFAS have been detected in the blood of 98% of Americans, and in the rainwater of locations as far afield as Tibet and Antarctica. Even low levels of exposure have been associated with an increased risk for cancer, liver disease, low birth weight, and hormonal disruption.
The properties of PFAS also make them both durable at very high heat and water repellent. Notoriously, the chemical was used by 3M to make Scotchgard for carpets and fabrics and by Dupont to make Teflon for nonstick coating of pots and pans. Although perfluorooctanoic acid (PFOA) was removed from nonstick cookware in 2013, PFAS – a family of thousands of synthetic compounds – remain common in fast-food packaging, water- and stain-repellent clothing, firefighting foam, and personal care products. PFAS are released into the environment during the breakdown of these consumer and industrial products, as well as from dumping from waste facilities.
Alarmingly, the EWG notes that up to 200 million Americans may be exposed to PFAS in their drinking water. In March 2021, the EPA announced that they will be regulating PFAS in drinking water; however, the regulations have not been finalized. Currently, it is up to individual states to test for its presence in the water. The EWG has compiled a map of all known PFAS contamination sites.
To avoid or prevent exposures from PFAS, recommendations include filtering tap water with either reverse osmosis or activated carbon filters, as well as avoiding fast food and carry-out food, if possible, and consumer products labeled as “water resistant,” “stain-resistant,” and “nonstick.”
In a testament to how harmful these chemicals are, the EPA recently revised their lifetime health advisories for PFAS, such as PFOA, to 0.004 parts per trillion, which is more than 10,000 times smaller than the previous limit of 70 parts per trillion. The EPA also has proposed formally designating certain PFAS chemicals as “hazardous substances.”
Dr. Goel, clinical assistant professor of medicine at Weill Cornell Medicine, New York, has disclosed no relevant financial relationships. A version of this article originally appeared on Medscape.com.