User login
COVID-19 in pregnancy raises risk of preterm birth and severe disease
based on data from two studies published in the Centers for Disease Control and Prevention’s Morbidity and Mortality Weekly Report.
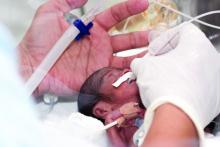
In a study of birth and infant outcomes, rates of preterm birth (less than 37 weeks’ gestational age) were higher among women with confirmed SARS-CoV-2 infections compared with the national average (12.9% vs. 10.2%) wrote Kate R. Woodworth, MD, and colleagues of the CDC COVID-19 Response Pregnancy and Linked Outcomes Team.
The researchers collected information on pregnancy and infant outcomes from 16 jurisdictions through the Surveillance for Emerging Threats to Mothers and Babies Network (SET-NET). The study included 5,252 women with laboratory-confirmed SARS-CoV-2 infection reported during March 29–Oct. 14, 2020.
Overall, 12.9% of the 3,912 live births with known gestational age were preterm. A total of 610 infants were tested for SARS-CoV-2, and 2.6% were positive. Most of these perinatal infections (85%) occurred among infants born to women with SARS-CoV-2 infection within 1 week of delivery.
Half of the infants with positive test results were preterm, possibly reflecting higher screening rates in the ICU, the researchers said. “These findings also support the growing evidence that although severe COVID-19 does occur in neonates the majority of term neonates experience asymptomatic infection or mild disease; however, information on long term outcomes among exposed infants is unknown.”
Address disparities that amplify risk
The study findings were limited by several factors including inconsistent symptom reporting, overrepresentation of Hispanic women, and incomplete information on pregnancy loss, Dr. Woodworth and associates noted. However, the results add to the knowledge about the impact of COVID-19 disease on pregnancy by providing a large, population-based cohort with completed pregnancy outcomes as well as infant testing.
“SET-NET will continue to follow pregnancies affected by SARS-CoV-2 through completion of pregnancy and infants until age 6 months to guide clinical and public health practice,” the researchers noted. “Longer-term investigation into solutions to alleviate underlying inequities in social determinants of health associated with disparities in maternal morbidity, mortality, and adverse pregnancy outcomes, and effectively addressing these inequities, could reduce the prevalence of conditions and experiences that might amplify risks from COVID-19,” they added.
Severe disease and death increased in pregnant women
In a second study published in the MMWR, Laura D. Zambrano, PhD, and colleagues, also of the CDC COVID-19 Response Pregnancy and Linked Outcomes Team, compared data on 23,434 reportedly pregnant and 386,028 nonpregnant women of reproductive age (15-44 years) with confirmed and symptomatic SARS-CoV-2 infections reported to the CDC between Jan. 22, 2020, and Oct. 3, 2020.
After adjustment for age, race, and underlying medical conditions, pregnant women with COVID-19 disease were significantly more likely than were nonpregnant women to be admitted to intensive care (10.5 per 1,000 cases vs. 3.9 per 1,000 cases), to receive invasive ventilation (2.9 vs. 1.1), receive extracorporeal membrane oxygenation (0.7 vs. 0.3) and to die (1.5 vs. 1.2).
“Irrespective of pregnancy status, ICU admissions, receipt of invasive ventilation, and death occurred more often among women aged 35-44 years than among those aged 15-24 years,” Dr. Zambrano and associates noted. In addition, non-Hispanic Black and Black women comprised 14.1% of the study population but accounted for 36.6% of deaths overall (9 in pregnant women and 167 in nonpregnant women).
The findings in the study of characteristics were limited by several factors including the voluntary reporting of COVID-19 cases, potential reporting bias, and inadequate time to assess severe cases, the researchers noted. However, “data from previous influenza pandemics, including 2009 H1N1, have shown that pregnant women are at increased risk for severe outcomes including death and the absolute risks for severe outcomes were higher than in this study of COVID-19 during pregnancy.”
“Pregnant women should be informed of their risk for severe COVID-19–associated illness and the warning signs of severe COVID-19,” Dr. Zambrano and associates said. “Providers who care for pregnant women should be familiar with guidelines for medical management of COVID-19, including considerations for management of COVID-19 in pregnancy.”
More data needed for informed counseling
“It is important to conduct research trials involving pregnant women so that we have reliable data regarding outcomes with which to counsel women,” Angela Bianco, MD, a maternal fetal medicine specialist at Mount Sinai Hospital in New York, said in an interview.
“Often pregnant women are excluded from research trials, but the impact of the current public health crisis affects all persons regardless of pregnancy status,” she said.
Dr. Bianco said that she was not surprised by the findings of either study. “In fact, our own research produced similar results.”
“These recent publications found that age-matched pregnant versus nonpregnant women had more severe manifestations of COVID-19, and specifically that pregnant women had a higher risk of requiring ventilation and intensive care admission, as well as higher risk of death,” she said. “Previous studies examining the effect of other SARS viruses have demonstrated that pregnancy is associated with worse outcomes; these findings are likely attributable to the relative state of immunosuppression in pregnancy.” Also, “one of these trials found a greater risk of premature birth in women with COVID-19; this may largely be attributable to iatrogenic delivery due to maternal illness as opposed to spontaneous preterm birth,” Dr. Bianco explained.
“Data are emerging regarding the impact of SARS-CoV-2 on pregnancy outcomes, however information remains limited,” Dr. Bianco noted. “Clinicians need to make patients aware that SARS-CoV-2 infection during pregnancy is associated with a greater risk of severe illness requiring intensive care and/or ventilatory support and even death; however, the precise rates remain unknown. “COVID-19 during pregnancy may result in a preterm birth, but at this time the rate of fetal infection remains unknown,” she said. “Clinicians need to reinforce the importance of physical distancing, mask use, and proper hand hygiene, particularly in this vulnerable population.”
Dr. Bianco emphasized: “Longitudinal studies assessing the impact of SARS-CoV-2 infection at various gestational age periods are needed, as at this time most of the available data includes women with SARS-CoV-2 infection around the time of delivery. Long-term infant outcomes are needed, as well as studies assessing the risk of fetal infection.”
The studies were supported by the Centers for Disease Control and Prevention. The researchers had no financial conflicts to disclose. Dr. Bianco had no relevant financial disclosures.
SOURCE: Woodworth KR et al. MMWR. 2020 Nov 2. doi: 10.15585/mmwr.mm6944e2; Zambrano LD et al. MMWR. 2020 Nov 2. doi: 10.15585/mmwr.mm6944e3.
based on data from two studies published in the Centers for Disease Control and Prevention’s Morbidity and Mortality Weekly Report.
In a study of birth and infant outcomes, rates of preterm birth (less than 37 weeks’ gestational age) were higher among women with confirmed SARS-CoV-2 infections compared with the national average (12.9% vs. 10.2%) wrote Kate R. Woodworth, MD, and colleagues of the CDC COVID-19 Response Pregnancy and Linked Outcomes Team.
The researchers collected information on pregnancy and infant outcomes from 16 jurisdictions through the Surveillance for Emerging Threats to Mothers and Babies Network (SET-NET). The study included 5,252 women with laboratory-confirmed SARS-CoV-2 infection reported during March 29–Oct. 14, 2020.
Overall, 12.9% of the 3,912 live births with known gestational age were preterm. A total of 610 infants were tested for SARS-CoV-2, and 2.6% were positive. Most of these perinatal infections (85%) occurred among infants born to women with SARS-CoV-2 infection within 1 week of delivery.
Half of the infants with positive test results were preterm, possibly reflecting higher screening rates in the ICU, the researchers said. “These findings also support the growing evidence that although severe COVID-19 does occur in neonates the majority of term neonates experience asymptomatic infection or mild disease; however, information on long term outcomes among exposed infants is unknown.”
Address disparities that amplify risk
The study findings were limited by several factors including inconsistent symptom reporting, overrepresentation of Hispanic women, and incomplete information on pregnancy loss, Dr. Woodworth and associates noted. However, the results add to the knowledge about the impact of COVID-19 disease on pregnancy by providing a large, population-based cohort with completed pregnancy outcomes as well as infant testing.
“SET-NET will continue to follow pregnancies affected by SARS-CoV-2 through completion of pregnancy and infants until age 6 months to guide clinical and public health practice,” the researchers noted. “Longer-term investigation into solutions to alleviate underlying inequities in social determinants of health associated with disparities in maternal morbidity, mortality, and adverse pregnancy outcomes, and effectively addressing these inequities, could reduce the prevalence of conditions and experiences that might amplify risks from COVID-19,” they added.
Severe disease and death increased in pregnant women
In a second study published in the MMWR, Laura D. Zambrano, PhD, and colleagues, also of the CDC COVID-19 Response Pregnancy and Linked Outcomes Team, compared data on 23,434 reportedly pregnant and 386,028 nonpregnant women of reproductive age (15-44 years) with confirmed and symptomatic SARS-CoV-2 infections reported to the CDC between Jan. 22, 2020, and Oct. 3, 2020.
After adjustment for age, race, and underlying medical conditions, pregnant women with COVID-19 disease were significantly more likely than were nonpregnant women to be admitted to intensive care (10.5 per 1,000 cases vs. 3.9 per 1,000 cases), to receive invasive ventilation (2.9 vs. 1.1), receive extracorporeal membrane oxygenation (0.7 vs. 0.3) and to die (1.5 vs. 1.2).
“Irrespective of pregnancy status, ICU admissions, receipt of invasive ventilation, and death occurred more often among women aged 35-44 years than among those aged 15-24 years,” Dr. Zambrano and associates noted. In addition, non-Hispanic Black and Black women comprised 14.1% of the study population but accounted for 36.6% of deaths overall (9 in pregnant women and 167 in nonpregnant women).
The findings in the study of characteristics were limited by several factors including the voluntary reporting of COVID-19 cases, potential reporting bias, and inadequate time to assess severe cases, the researchers noted. However, “data from previous influenza pandemics, including 2009 H1N1, have shown that pregnant women are at increased risk for severe outcomes including death and the absolute risks for severe outcomes were higher than in this study of COVID-19 during pregnancy.”
“Pregnant women should be informed of their risk for severe COVID-19–associated illness and the warning signs of severe COVID-19,” Dr. Zambrano and associates said. “Providers who care for pregnant women should be familiar with guidelines for medical management of COVID-19, including considerations for management of COVID-19 in pregnancy.”
More data needed for informed counseling
“It is important to conduct research trials involving pregnant women so that we have reliable data regarding outcomes with which to counsel women,” Angela Bianco, MD, a maternal fetal medicine specialist at Mount Sinai Hospital in New York, said in an interview.
“Often pregnant women are excluded from research trials, but the impact of the current public health crisis affects all persons regardless of pregnancy status,” she said.
Dr. Bianco said that she was not surprised by the findings of either study. “In fact, our own research produced similar results.”
“These recent publications found that age-matched pregnant versus nonpregnant women had more severe manifestations of COVID-19, and specifically that pregnant women had a higher risk of requiring ventilation and intensive care admission, as well as higher risk of death,” she said. “Previous studies examining the effect of other SARS viruses have demonstrated that pregnancy is associated with worse outcomes; these findings are likely attributable to the relative state of immunosuppression in pregnancy.” Also, “one of these trials found a greater risk of premature birth in women with COVID-19; this may largely be attributable to iatrogenic delivery due to maternal illness as opposed to spontaneous preterm birth,” Dr. Bianco explained.
“Data are emerging regarding the impact of SARS-CoV-2 on pregnancy outcomes, however information remains limited,” Dr. Bianco noted. “Clinicians need to make patients aware that SARS-CoV-2 infection during pregnancy is associated with a greater risk of severe illness requiring intensive care and/or ventilatory support and even death; however, the precise rates remain unknown. “COVID-19 during pregnancy may result in a preterm birth, but at this time the rate of fetal infection remains unknown,” she said. “Clinicians need to reinforce the importance of physical distancing, mask use, and proper hand hygiene, particularly in this vulnerable population.”
Dr. Bianco emphasized: “Longitudinal studies assessing the impact of SARS-CoV-2 infection at various gestational age periods are needed, as at this time most of the available data includes women with SARS-CoV-2 infection around the time of delivery. Long-term infant outcomes are needed, as well as studies assessing the risk of fetal infection.”
The studies were supported by the Centers for Disease Control and Prevention. The researchers had no financial conflicts to disclose. Dr. Bianco had no relevant financial disclosures.
SOURCE: Woodworth KR et al. MMWR. 2020 Nov 2. doi: 10.15585/mmwr.mm6944e2; Zambrano LD et al. MMWR. 2020 Nov 2. doi: 10.15585/mmwr.mm6944e3.
based on data from two studies published in the Centers for Disease Control and Prevention’s Morbidity and Mortality Weekly Report.
In a study of birth and infant outcomes, rates of preterm birth (less than 37 weeks’ gestational age) were higher among women with confirmed SARS-CoV-2 infections compared with the national average (12.9% vs. 10.2%) wrote Kate R. Woodworth, MD, and colleagues of the CDC COVID-19 Response Pregnancy and Linked Outcomes Team.
The researchers collected information on pregnancy and infant outcomes from 16 jurisdictions through the Surveillance for Emerging Threats to Mothers and Babies Network (SET-NET). The study included 5,252 women with laboratory-confirmed SARS-CoV-2 infection reported during March 29–Oct. 14, 2020.
Overall, 12.9% of the 3,912 live births with known gestational age were preterm. A total of 610 infants were tested for SARS-CoV-2, and 2.6% were positive. Most of these perinatal infections (85%) occurred among infants born to women with SARS-CoV-2 infection within 1 week of delivery.
Half of the infants with positive test results were preterm, possibly reflecting higher screening rates in the ICU, the researchers said. “These findings also support the growing evidence that although severe COVID-19 does occur in neonates the majority of term neonates experience asymptomatic infection or mild disease; however, information on long term outcomes among exposed infants is unknown.”
Address disparities that amplify risk
The study findings were limited by several factors including inconsistent symptom reporting, overrepresentation of Hispanic women, and incomplete information on pregnancy loss, Dr. Woodworth and associates noted. However, the results add to the knowledge about the impact of COVID-19 disease on pregnancy by providing a large, population-based cohort with completed pregnancy outcomes as well as infant testing.
“SET-NET will continue to follow pregnancies affected by SARS-CoV-2 through completion of pregnancy and infants until age 6 months to guide clinical and public health practice,” the researchers noted. “Longer-term investigation into solutions to alleviate underlying inequities in social determinants of health associated with disparities in maternal morbidity, mortality, and adverse pregnancy outcomes, and effectively addressing these inequities, could reduce the prevalence of conditions and experiences that might amplify risks from COVID-19,” they added.
Severe disease and death increased in pregnant women
In a second study published in the MMWR, Laura D. Zambrano, PhD, and colleagues, also of the CDC COVID-19 Response Pregnancy and Linked Outcomes Team, compared data on 23,434 reportedly pregnant and 386,028 nonpregnant women of reproductive age (15-44 years) with confirmed and symptomatic SARS-CoV-2 infections reported to the CDC between Jan. 22, 2020, and Oct. 3, 2020.
After adjustment for age, race, and underlying medical conditions, pregnant women with COVID-19 disease were significantly more likely than were nonpregnant women to be admitted to intensive care (10.5 per 1,000 cases vs. 3.9 per 1,000 cases), to receive invasive ventilation (2.9 vs. 1.1), receive extracorporeal membrane oxygenation (0.7 vs. 0.3) and to die (1.5 vs. 1.2).
“Irrespective of pregnancy status, ICU admissions, receipt of invasive ventilation, and death occurred more often among women aged 35-44 years than among those aged 15-24 years,” Dr. Zambrano and associates noted. In addition, non-Hispanic Black and Black women comprised 14.1% of the study population but accounted for 36.6% of deaths overall (9 in pregnant women and 167 in nonpregnant women).
The findings in the study of characteristics were limited by several factors including the voluntary reporting of COVID-19 cases, potential reporting bias, and inadequate time to assess severe cases, the researchers noted. However, “data from previous influenza pandemics, including 2009 H1N1, have shown that pregnant women are at increased risk for severe outcomes including death and the absolute risks for severe outcomes were higher than in this study of COVID-19 during pregnancy.”
“Pregnant women should be informed of their risk for severe COVID-19–associated illness and the warning signs of severe COVID-19,” Dr. Zambrano and associates said. “Providers who care for pregnant women should be familiar with guidelines for medical management of COVID-19, including considerations for management of COVID-19 in pregnancy.”
More data needed for informed counseling
“It is important to conduct research trials involving pregnant women so that we have reliable data regarding outcomes with which to counsel women,” Angela Bianco, MD, a maternal fetal medicine specialist at Mount Sinai Hospital in New York, said in an interview.
“Often pregnant women are excluded from research trials, but the impact of the current public health crisis affects all persons regardless of pregnancy status,” she said.
Dr. Bianco said that she was not surprised by the findings of either study. “In fact, our own research produced similar results.”
“These recent publications found that age-matched pregnant versus nonpregnant women had more severe manifestations of COVID-19, and specifically that pregnant women had a higher risk of requiring ventilation and intensive care admission, as well as higher risk of death,” she said. “Previous studies examining the effect of other SARS viruses have demonstrated that pregnancy is associated with worse outcomes; these findings are likely attributable to the relative state of immunosuppression in pregnancy.” Also, “one of these trials found a greater risk of premature birth in women with COVID-19; this may largely be attributable to iatrogenic delivery due to maternal illness as opposed to spontaneous preterm birth,” Dr. Bianco explained.
“Data are emerging regarding the impact of SARS-CoV-2 on pregnancy outcomes, however information remains limited,” Dr. Bianco noted. “Clinicians need to make patients aware that SARS-CoV-2 infection during pregnancy is associated with a greater risk of severe illness requiring intensive care and/or ventilatory support and even death; however, the precise rates remain unknown. “COVID-19 during pregnancy may result in a preterm birth, but at this time the rate of fetal infection remains unknown,” she said. “Clinicians need to reinforce the importance of physical distancing, mask use, and proper hand hygiene, particularly in this vulnerable population.”
Dr. Bianco emphasized: “Longitudinal studies assessing the impact of SARS-CoV-2 infection at various gestational age periods are needed, as at this time most of the available data includes women with SARS-CoV-2 infection around the time of delivery. Long-term infant outcomes are needed, as well as studies assessing the risk of fetal infection.”
The studies were supported by the Centers for Disease Control and Prevention. The researchers had no financial conflicts to disclose. Dr. Bianco had no relevant financial disclosures.
SOURCE: Woodworth KR et al. MMWR. 2020 Nov 2. doi: 10.15585/mmwr.mm6944e2; Zambrano LD et al. MMWR. 2020 Nov 2. doi: 10.15585/mmwr.mm6944e3.
FROM MMWR
New case suggestive of in utero SARS-CoV-2 transmission
A new report of mother-to-fetus transmission of SARS-CoV-2 through umbilical cord blood adds to a small but growing body of evidence that the virus can be transmitted in utero.
Further,
The data
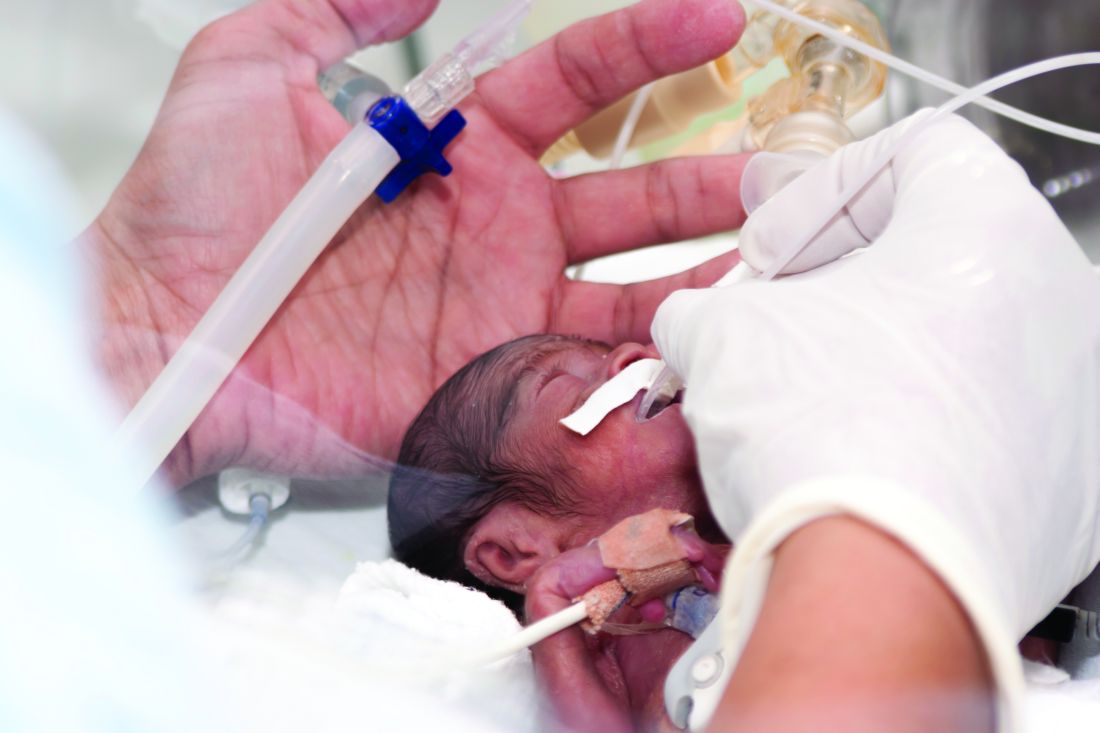
In a report published in the Journal of The Pediatric Infectious Diseases Society, Isabelle Von Kohorn, MD, PhD, of Holy Cross Health in Silver Spring, Md., and colleagues, described a case of neonatal infection with SARS-CoV-2 in a boy delivered by C-section at 34 weeks to a mother diagnosed with COVID-19 some 14 hours before. The newborn was immediately removed to a neonatal ICU and reunited with his mother a week later, once the mother had recovered.
Dr. Von Kohorn and colleagues reported that, while the infant’s nasopharyngeal swab test for SARS-CoV-2 was negative at 24 hours after birth, repeat molecular tests (using different assays) from 49 hours on were positive and indicated an increasing viral burden, although the infant never developed symptoms of COVID-19. In addition to being found in the nasopharynx, viral RNA also was detected in cord blood and in urine. No viral RNA was found in the placenta.
The circumstances of the birth, and the care taken to keep mother and her infant at a safe distance along with masking of the mother, made it “extremely unlikely” that the infant acquired his infection by the respiratory route, Dr. Von Kohorn and colleagues wrote.
“While we cannot rule out microscopic maternal blood contamination of cord blood in this or any other delivery, cord blood collection procedures are designed to avoid gross contamination with maternal blood. Microscopic contamination would not explain the RNA levels observed in our patient’s cord blood,” they wrote.
Clinicians should note that a neonate born to a mother with COVID-19 may take time to test positive for SARS-CoV-2 , the investigators argued, though the current recommendation of the American Academy of Pediatrics is to test nasopharyngeal secretions of well newborns at 24 and 48 hours but not again in the absence of symptoms. “This case suggests that some cases of SARS-CoV-2 in newborns may be detectable only after 48 hours of life.”
The authors hypothesized that virus transmitted by cord blood “seeded the nasopharynx and required 2 days for incubation and replication sufficient for detection.”
Some perspective
In an interview, Andrea Edlow, MD, A maternal-fetal medicine specialist at Massachusetts General Hospital in Boston, called the findings provocative if not definitive in establishing in utero or vertical transmission of SARS-CoV-2 in the same way that a Nature Communications case report did in July 2020. In that case, of a baby born to a mother with COVID-19, virus was seen at high levels in the placenta.
With the current case, “the absence of detectable virus in the placenta is certainly inconsistent/confusing if the authors claim hematogenous spread from mother to baby,” Dr. Edlow commented, “but the authors do offer plausible explanations, such as examination of limited areas within the placenta (when we know infection is likely to be patchy) and possible degradation of RNA prior to attempting to measure placental viral presence.”
Dr. Von Kohorn and colleagues’ study was funded by the National Institutes of Health, and the investigators disclosed no financial conflicts of interest. Dr. Edlow had no relevant financial disclosures.
SOURCE: Von Kohorn I et al. J Pediat Inf Dis Soc. 2020 Oct 22. doi: 10.1093/jpids/piaa127
A new report of mother-to-fetus transmission of SARS-CoV-2 through umbilical cord blood adds to a small but growing body of evidence that the virus can be transmitted in utero.
Further,
The data
In a report published in the Journal of The Pediatric Infectious Diseases Society, Isabelle Von Kohorn, MD, PhD, of Holy Cross Health in Silver Spring, Md., and colleagues, described a case of neonatal infection with SARS-CoV-2 in a boy delivered by C-section at 34 weeks to a mother diagnosed with COVID-19 some 14 hours before. The newborn was immediately removed to a neonatal ICU and reunited with his mother a week later, once the mother had recovered.
Dr. Von Kohorn and colleagues reported that, while the infant’s nasopharyngeal swab test for SARS-CoV-2 was negative at 24 hours after birth, repeat molecular tests (using different assays) from 49 hours on were positive and indicated an increasing viral burden, although the infant never developed symptoms of COVID-19. In addition to being found in the nasopharynx, viral RNA also was detected in cord blood and in urine. No viral RNA was found in the placenta.
The circumstances of the birth, and the care taken to keep mother and her infant at a safe distance along with masking of the mother, made it “extremely unlikely” that the infant acquired his infection by the respiratory route, Dr. Von Kohorn and colleagues wrote.
“While we cannot rule out microscopic maternal blood contamination of cord blood in this or any other delivery, cord blood collection procedures are designed to avoid gross contamination with maternal blood. Microscopic contamination would not explain the RNA levels observed in our patient’s cord blood,” they wrote.
Clinicians should note that a neonate born to a mother with COVID-19 may take time to test positive for SARS-CoV-2 , the investigators argued, though the current recommendation of the American Academy of Pediatrics is to test nasopharyngeal secretions of well newborns at 24 and 48 hours but not again in the absence of symptoms. “This case suggests that some cases of SARS-CoV-2 in newborns may be detectable only after 48 hours of life.”
The authors hypothesized that virus transmitted by cord blood “seeded the nasopharynx and required 2 days for incubation and replication sufficient for detection.”
Some perspective
In an interview, Andrea Edlow, MD, A maternal-fetal medicine specialist at Massachusetts General Hospital in Boston, called the findings provocative if not definitive in establishing in utero or vertical transmission of SARS-CoV-2 in the same way that a Nature Communications case report did in July 2020. In that case, of a baby born to a mother with COVID-19, virus was seen at high levels in the placenta.
With the current case, “the absence of detectable virus in the placenta is certainly inconsistent/confusing if the authors claim hematogenous spread from mother to baby,” Dr. Edlow commented, “but the authors do offer plausible explanations, such as examination of limited areas within the placenta (when we know infection is likely to be patchy) and possible degradation of RNA prior to attempting to measure placental viral presence.”
Dr. Von Kohorn and colleagues’ study was funded by the National Institutes of Health, and the investigators disclosed no financial conflicts of interest. Dr. Edlow had no relevant financial disclosures.
SOURCE: Von Kohorn I et al. J Pediat Inf Dis Soc. 2020 Oct 22. doi: 10.1093/jpids/piaa127
A new report of mother-to-fetus transmission of SARS-CoV-2 through umbilical cord blood adds to a small but growing body of evidence that the virus can be transmitted in utero.
Further,
The data
In a report published in the Journal of The Pediatric Infectious Diseases Society, Isabelle Von Kohorn, MD, PhD, of Holy Cross Health in Silver Spring, Md., and colleagues, described a case of neonatal infection with SARS-CoV-2 in a boy delivered by C-section at 34 weeks to a mother diagnosed with COVID-19 some 14 hours before. The newborn was immediately removed to a neonatal ICU and reunited with his mother a week later, once the mother had recovered.
Dr. Von Kohorn and colleagues reported that, while the infant’s nasopharyngeal swab test for SARS-CoV-2 was negative at 24 hours after birth, repeat molecular tests (using different assays) from 49 hours on were positive and indicated an increasing viral burden, although the infant never developed symptoms of COVID-19. In addition to being found in the nasopharynx, viral RNA also was detected in cord blood and in urine. No viral RNA was found in the placenta.
The circumstances of the birth, and the care taken to keep mother and her infant at a safe distance along with masking of the mother, made it “extremely unlikely” that the infant acquired his infection by the respiratory route, Dr. Von Kohorn and colleagues wrote.
“While we cannot rule out microscopic maternal blood contamination of cord blood in this or any other delivery, cord blood collection procedures are designed to avoid gross contamination with maternal blood. Microscopic contamination would not explain the RNA levels observed in our patient’s cord blood,” they wrote.
Clinicians should note that a neonate born to a mother with COVID-19 may take time to test positive for SARS-CoV-2 , the investigators argued, though the current recommendation of the American Academy of Pediatrics is to test nasopharyngeal secretions of well newborns at 24 and 48 hours but not again in the absence of symptoms. “This case suggests that some cases of SARS-CoV-2 in newborns may be detectable only after 48 hours of life.”
The authors hypothesized that virus transmitted by cord blood “seeded the nasopharynx and required 2 days for incubation and replication sufficient for detection.”
Some perspective
In an interview, Andrea Edlow, MD, A maternal-fetal medicine specialist at Massachusetts General Hospital in Boston, called the findings provocative if not definitive in establishing in utero or vertical transmission of SARS-CoV-2 in the same way that a Nature Communications case report did in July 2020. In that case, of a baby born to a mother with COVID-19, virus was seen at high levels in the placenta.
With the current case, “the absence of detectable virus in the placenta is certainly inconsistent/confusing if the authors claim hematogenous spread from mother to baby,” Dr. Edlow commented, “but the authors do offer plausible explanations, such as examination of limited areas within the placenta (when we know infection is likely to be patchy) and possible degradation of RNA prior to attempting to measure placental viral presence.”
Dr. Von Kohorn and colleagues’ study was funded by the National Institutes of Health, and the investigators disclosed no financial conflicts of interest. Dr. Edlow had no relevant financial disclosures.
SOURCE: Von Kohorn I et al. J Pediat Inf Dis Soc. 2020 Oct 22. doi: 10.1093/jpids/piaa127
FROM THE JOURNAL OF THE PEDIATRIC INFECTIOUS DISEASES SOCIETY
Primary care workforce expanding, but mostly in cities
researchers say.
The finding may provide some reassurance for those who have worried about a shortage of health care workers and whether they will be able to meet the nation’s growing burden of chronic diseases.
“Access to primary care doctors is critical to population health and to reduce health care disparities in this country,” said Donglan Zhang, PhD, an assistant professor of public health at the University of Georgia, Athens.
However, many counties remain underserved, Dr. Zhang said in an interview. The need for primary care in the United States is increasing not only with population growth but because the population is aging.
Dr. Zhang and colleagues published the finding in JAMA Network Open.
Many previous reports have warned of a shortage in primary care providers. To examine recent trends in the primary care workforce, Dr. Zhang and colleagues obtained data on all the primary care clinicians registered with the Centers for Medicare & Medicaid Services from 2009 to 2017.
For the study, the researchers included general practitioners, family physicians and internists without subspecialties, nurse practitioners, and physician assistants. They then compared the number of providers with the number of residents in each county as recorded by the US Census, using urban or rural classifications for each county from the Centers for Disease Control and Prevention.
Because the U.S. Health Resources and Services Administration defines a primary care “shortage” as fewer than 1 primary care practitioner per 3,500 people, the researchers focused on this ratio. They found that the number of nurse practitioners and physician assistants was increasing much faster than the number of primary care physicians. This was true especially in rural areas, but the percentage increase for both nurse practitioners and physician assistants was lower in rural areas versus urban.
The researchers also found that there were more primary care physicians per capita in counties with higher household incomes, a higher proportion of Asian residents, and a higher proportion of college graduates.
They didn’t find a significant association between the median household income and per capita number of nurse practitioners.
They found that counties with a higher proportion of Black and Asian residents had a higher number of nurse practitioners per capita. But they found an opposite association between the proportion of Black residents and the number of physician assistants per capita.
The authors hypothesized that health care reform, particularly the passage of the Affordable Care Act in 2010, may explain the recent increase in the primary care workforce. The legislation expanded the number of people with health insurance and provided incentives for primary and preventive care.
Another factor behind the increase in the primary care workforce could be state laws that have expanded the scope of practice for nurse practitioners and primary care providers, she said.
Numbers may overestimate available care
The gap between rural and urban areas could be even wider than this study suggests, Ada D. Stewart, MD, president of the American Academy of Family Physicians, said in an interview. Many nurse practitioners and physician assistants don’t actually practice primary care, but instead assist physicians in other specialties such as orthopedics or general surgery.

“They are part of a team and I don’t want to diminish that at all, but especially when we talk about infant and maternal mortality, family physicians need to be there themselves providing primary care,” she said. “We’re there in hospitals and emergency rooms, and not just taking care of diabetes and hypertension.”
In addition, the primary care workforce may have been reduced since the conclusion of the study period (Dec. 31, 2017) as a result of the COVID-19 pandemic forcing some primary care physicians into retirement, Dr. Stewart said.
Measures that could help reduce the disparity include a more robust system of teaching health centers in rural counties, higher reimbursement for primary care, a lower cost of medical education, and recruiting more people from rural areas to become physicians, Dr. Stewart said.
Telehealth can enhance health care in rural areas, but many people in rural areas lack internet or cellular service, or don’t have access to computers. “We don’t want to create another healthcare disparity,” she said.
And physicians can get to know their patients’ needs better in a face-to-face visit, she said. “Telehealth does have a place, but it does not replace that person-to-person visit.”
This study was funded by National Institute on Minority Health and Health Disparities. Dr. Zhang and Dr. Stewart disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
researchers say.
The finding may provide some reassurance for those who have worried about a shortage of health care workers and whether they will be able to meet the nation’s growing burden of chronic diseases.
“Access to primary care doctors is critical to population health and to reduce health care disparities in this country,” said Donglan Zhang, PhD, an assistant professor of public health at the University of Georgia, Athens.
However, many counties remain underserved, Dr. Zhang said in an interview. The need for primary care in the United States is increasing not only with population growth but because the population is aging.
Dr. Zhang and colleagues published the finding in JAMA Network Open.
Many previous reports have warned of a shortage in primary care providers. To examine recent trends in the primary care workforce, Dr. Zhang and colleagues obtained data on all the primary care clinicians registered with the Centers for Medicare & Medicaid Services from 2009 to 2017.
For the study, the researchers included general practitioners, family physicians and internists without subspecialties, nurse practitioners, and physician assistants. They then compared the number of providers with the number of residents in each county as recorded by the US Census, using urban or rural classifications for each county from the Centers for Disease Control and Prevention.
Because the U.S. Health Resources and Services Administration defines a primary care “shortage” as fewer than 1 primary care practitioner per 3,500 people, the researchers focused on this ratio. They found that the number of nurse practitioners and physician assistants was increasing much faster than the number of primary care physicians. This was true especially in rural areas, but the percentage increase for both nurse practitioners and physician assistants was lower in rural areas versus urban.
The researchers also found that there were more primary care physicians per capita in counties with higher household incomes, a higher proportion of Asian residents, and a higher proportion of college graduates.
They didn’t find a significant association between the median household income and per capita number of nurse practitioners.
They found that counties with a higher proportion of Black and Asian residents had a higher number of nurse practitioners per capita. But they found an opposite association between the proportion of Black residents and the number of physician assistants per capita.
The authors hypothesized that health care reform, particularly the passage of the Affordable Care Act in 2010, may explain the recent increase in the primary care workforce. The legislation expanded the number of people with health insurance and provided incentives for primary and preventive care.
Another factor behind the increase in the primary care workforce could be state laws that have expanded the scope of practice for nurse practitioners and primary care providers, she said.
Numbers may overestimate available care
The gap between rural and urban areas could be even wider than this study suggests, Ada D. Stewart, MD, president of the American Academy of Family Physicians, said in an interview. Many nurse practitioners and physician assistants don’t actually practice primary care, but instead assist physicians in other specialties such as orthopedics or general surgery.

“They are part of a team and I don’t want to diminish that at all, but especially when we talk about infant and maternal mortality, family physicians need to be there themselves providing primary care,” she said. “We’re there in hospitals and emergency rooms, and not just taking care of diabetes and hypertension.”
In addition, the primary care workforce may have been reduced since the conclusion of the study period (Dec. 31, 2017) as a result of the COVID-19 pandemic forcing some primary care physicians into retirement, Dr. Stewart said.
Measures that could help reduce the disparity include a more robust system of teaching health centers in rural counties, higher reimbursement for primary care, a lower cost of medical education, and recruiting more people from rural areas to become physicians, Dr. Stewart said.
Telehealth can enhance health care in rural areas, but many people in rural areas lack internet or cellular service, or don’t have access to computers. “We don’t want to create another healthcare disparity,” she said.
And physicians can get to know their patients’ needs better in a face-to-face visit, she said. “Telehealth does have a place, but it does not replace that person-to-person visit.”
This study was funded by National Institute on Minority Health and Health Disparities. Dr. Zhang and Dr. Stewart disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
researchers say.
The finding may provide some reassurance for those who have worried about a shortage of health care workers and whether they will be able to meet the nation’s growing burden of chronic diseases.
“Access to primary care doctors is critical to population health and to reduce health care disparities in this country,” said Donglan Zhang, PhD, an assistant professor of public health at the University of Georgia, Athens.
However, many counties remain underserved, Dr. Zhang said in an interview. The need for primary care in the United States is increasing not only with population growth but because the population is aging.
Dr. Zhang and colleagues published the finding in JAMA Network Open.
Many previous reports have warned of a shortage in primary care providers. To examine recent trends in the primary care workforce, Dr. Zhang and colleagues obtained data on all the primary care clinicians registered with the Centers for Medicare & Medicaid Services from 2009 to 2017.
For the study, the researchers included general practitioners, family physicians and internists without subspecialties, nurse practitioners, and physician assistants. They then compared the number of providers with the number of residents in each county as recorded by the US Census, using urban or rural classifications for each county from the Centers for Disease Control and Prevention.
Because the U.S. Health Resources and Services Administration defines a primary care “shortage” as fewer than 1 primary care practitioner per 3,500 people, the researchers focused on this ratio. They found that the number of nurse practitioners and physician assistants was increasing much faster than the number of primary care physicians. This was true especially in rural areas, but the percentage increase for both nurse practitioners and physician assistants was lower in rural areas versus urban.
The researchers also found that there were more primary care physicians per capita in counties with higher household incomes, a higher proportion of Asian residents, and a higher proportion of college graduates.
They didn’t find a significant association between the median household income and per capita number of nurse practitioners.
They found that counties with a higher proportion of Black and Asian residents had a higher number of nurse practitioners per capita. But they found an opposite association between the proportion of Black residents and the number of physician assistants per capita.
The authors hypothesized that health care reform, particularly the passage of the Affordable Care Act in 2010, may explain the recent increase in the primary care workforce. The legislation expanded the number of people with health insurance and provided incentives for primary and preventive care.
Another factor behind the increase in the primary care workforce could be state laws that have expanded the scope of practice for nurse practitioners and primary care providers, she said.
Numbers may overestimate available care
The gap between rural and urban areas could be even wider than this study suggests, Ada D. Stewart, MD, president of the American Academy of Family Physicians, said in an interview. Many nurse practitioners and physician assistants don’t actually practice primary care, but instead assist physicians in other specialties such as orthopedics or general surgery.

“They are part of a team and I don’t want to diminish that at all, but especially when we talk about infant and maternal mortality, family physicians need to be there themselves providing primary care,” she said. “We’re there in hospitals and emergency rooms, and not just taking care of diabetes and hypertension.”
In addition, the primary care workforce may have been reduced since the conclusion of the study period (Dec. 31, 2017) as a result of the COVID-19 pandemic forcing some primary care physicians into retirement, Dr. Stewart said.
Measures that could help reduce the disparity include a more robust system of teaching health centers in rural counties, higher reimbursement for primary care, a lower cost of medical education, and recruiting more people from rural areas to become physicians, Dr. Stewart said.
Telehealth can enhance health care in rural areas, but many people in rural areas lack internet or cellular service, or don’t have access to computers. “We don’t want to create another healthcare disparity,” she said.
And physicians can get to know their patients’ needs better in a face-to-face visit, she said. “Telehealth does have a place, but it does not replace that person-to-person visit.”
This study was funded by National Institute on Minority Health and Health Disparities. Dr. Zhang and Dr. Stewart disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Proposed withdrawal of approval of preterm drug: Two opposing views
The Oct. 5, 2020 move by the Food and Drug Administration’s Center for Drug Evaluation and Research (CDER) suggesting the withdrawal of the approval of Makena incited some opposition.
Amag Pharmaceuticals’ 17 alpha-hydroxyprogesterone caproate (17OHP) injection received accelerated approval in 2011 to reduce the risk of recurrent preterm birth in women with previous unexplained preterm birth. Makena is the only drug approved for preventing recurrent preterm birth.
The back story
The approval was based on findings from a randomized, placebo-controlled trial that demonstrated a 34% relative risk reduction in births before 37 weeks – from 55% in the placebo arm to 36% in the 17OHP-treated arm.
The trial was not designed to measure neonatal outcomes, with the surrogate outcome of recurrent preterm birth being determined as “reasonably likely” to predict benefit to the neonate.
Subsequently, results of the required postapproval confirmatory PROLONG trialproduced conflicting results, failing to show a benefit of 17OHP on either preterm birth or neonatal outcome, which prompted the proposed withdrawal of the drug’s approval.
The CDER advisory committee agreed unanimously that the PROLONG trial did not support the clinical benefit of 17OHP, but the committee was not unanimous in deciding what to do. Of the 16 members, 9 voted to withdraw the drug’s approval, while seven voted to retain it and require another confirmatory trial.
When CDER recommends withdrawal, the company can request a public hearing, which it has done. The FDA commissioner will recommend whether to grant this request.
In the meantime, one from a group of three doctors who are against it and the other from the CDER.
Arguments from the opposing views
“We sympathize with women who are at risk for recurrent preterm birth that could result in death or significant lifelong health effects in neonates, but retaining on the market a drug not shown to be effective for this use does not protect or promote their health,” wrote Christina Chang, MD, MPH and associates from CDER.
On the other hand, “the widespread use of 17OHP after accelerated approval has not uncovered important safety signals,” countered Michael F. Greene, MD, from Massachusetts General Hospital, Boston; David Harrington, PhD, from the Harvard T. Chan School of Public Health, Boston; and Mark A. Klebanoff, MD, MPH, who was coauthor on the original preapproval study and is with Nationwide Children’s Hospital, the Ohio State University College of Medicine, and Ohio State University College of Public Health, all in Columbus. “Withdrawal of the approval for 17OHP, as imperfect as it may be, will leave a very vulnerable demographic group of U.S. women at high risk for this complication of pregnancy with absolutely no available therapeutic option.”
While both the preapproval study and postapproval PROLONG trial had the same enrollment criteria – namely women with a singleton pregnancy and previous singleton spontaneous preterm birth – all parties acknowledged that the studies ended up with very different cohorts. Approval of the drug in the United States made it difficult to recruit U.S. participants for the second trial “because of a lack of equipoise perceived by health care providers and patients,” noted Dr. Greene and associates, resulting in 75% of the PROLONG study’s cohort coming from Europe. This meant that 59% of those in the first study were non-Hispanic black compared with just 6.6% in the PROLONG study, a difference that is important because of the increased risk of preterm birth in Black women.
“Black women are generally underrepresented in U.S. clinical trials, and they are clearly underrepresented in the PROLONG study,” noted Dr. Greene and colleagues, adding that “the total number of qualifying composite neonatal outcome events among Blacks or African Americans in the entire PROLONG study population of 1,700 participants was 9 (6 of 69 in the 17OHP group and 3 of 40 in the placebo group). This is not a robust database from which to conclude that there is no effect in Black women.”
But, Dr. Chang and the CDER group argued, while the first study showed 17OHP “reduced the risk of recurrent preterm birth in both Black and non-Black participants, the lack of even a trend toward efficacy among either Black or non-Black women in [the PROLONG study] argues that the smaller proportion of Black women [in the PROLONG study] does not explain the lack of efficacy.”
In addition to race, there were other risk factors for preterm birth, such as tobacco, alcohol, and street drug use; marital status; and age that differed between the two study cohorts. Even after subcategorizing PROLONG trial participants into higher or lower risk for preterm birth based on these risk factors, Dr. Chang and associates still found no evidence of benefit to 17OHP treatment in any risk group.
Withdrawing approval of 17OHP for a recurrent preterm indication would still allow off-label prescribing, but would most likely end insurance coverage and eventually manufacturing of the drug, noted Dr. Greene and associates.
“When the majority of a population achieves little benefit from a drug, but a minority demographic group at greatest risk for a serious medical problem appears to obtain significant benefit, any decision that will ultimately make it impossible to obtain the drug should be undertaken cautiously,” they warned. “This issue is particularly pressing when that minority group may be the least able to find and financially afford work-arounds to obtain the needed medication in our complex medical system that has a history of failing to serve them well.”
Dr. Chang and associates reported they had no relevant financial disclosures. Dr. Greene and associates reported that they had no relevant conflicts of interest or financial disclosures. Dr. Greene reported he is employed by the New England Journal of Medicine as associate editor. Dr. Harrington reported being employed by the journal as statistical consultant. Dr. Klebanoff reported he was an author of the original article about 17OHP published in the journal and referenced in this article.
The Oct. 5, 2020 move by the Food and Drug Administration’s Center for Drug Evaluation and Research (CDER) suggesting the withdrawal of the approval of Makena incited some opposition.
Amag Pharmaceuticals’ 17 alpha-hydroxyprogesterone caproate (17OHP) injection received accelerated approval in 2011 to reduce the risk of recurrent preterm birth in women with previous unexplained preterm birth. Makena is the only drug approved for preventing recurrent preterm birth.
The back story
The approval was based on findings from a randomized, placebo-controlled trial that demonstrated a 34% relative risk reduction in births before 37 weeks – from 55% in the placebo arm to 36% in the 17OHP-treated arm.
The trial was not designed to measure neonatal outcomes, with the surrogate outcome of recurrent preterm birth being determined as “reasonably likely” to predict benefit to the neonate.
Subsequently, results of the required postapproval confirmatory PROLONG trialproduced conflicting results, failing to show a benefit of 17OHP on either preterm birth or neonatal outcome, which prompted the proposed withdrawal of the drug’s approval.
The CDER advisory committee agreed unanimously that the PROLONG trial did not support the clinical benefit of 17OHP, but the committee was not unanimous in deciding what to do. Of the 16 members, 9 voted to withdraw the drug’s approval, while seven voted to retain it and require another confirmatory trial.
When CDER recommends withdrawal, the company can request a public hearing, which it has done. The FDA commissioner will recommend whether to grant this request.
In the meantime, one from a group of three doctors who are against it and the other from the CDER.
Arguments from the opposing views
“We sympathize with women who are at risk for recurrent preterm birth that could result in death or significant lifelong health effects in neonates, but retaining on the market a drug not shown to be effective for this use does not protect or promote their health,” wrote Christina Chang, MD, MPH and associates from CDER.
On the other hand, “the widespread use of 17OHP after accelerated approval has not uncovered important safety signals,” countered Michael F. Greene, MD, from Massachusetts General Hospital, Boston; David Harrington, PhD, from the Harvard T. Chan School of Public Health, Boston; and Mark A. Klebanoff, MD, MPH, who was coauthor on the original preapproval study and is with Nationwide Children’s Hospital, the Ohio State University College of Medicine, and Ohio State University College of Public Health, all in Columbus. “Withdrawal of the approval for 17OHP, as imperfect as it may be, will leave a very vulnerable demographic group of U.S. women at high risk for this complication of pregnancy with absolutely no available therapeutic option.”
While both the preapproval study and postapproval PROLONG trial had the same enrollment criteria – namely women with a singleton pregnancy and previous singleton spontaneous preterm birth – all parties acknowledged that the studies ended up with very different cohorts. Approval of the drug in the United States made it difficult to recruit U.S. participants for the second trial “because of a lack of equipoise perceived by health care providers and patients,” noted Dr. Greene and associates, resulting in 75% of the PROLONG study’s cohort coming from Europe. This meant that 59% of those in the first study were non-Hispanic black compared with just 6.6% in the PROLONG study, a difference that is important because of the increased risk of preterm birth in Black women.
“Black women are generally underrepresented in U.S. clinical trials, and they are clearly underrepresented in the PROLONG study,” noted Dr. Greene and colleagues, adding that “the total number of qualifying composite neonatal outcome events among Blacks or African Americans in the entire PROLONG study population of 1,700 participants was 9 (6 of 69 in the 17OHP group and 3 of 40 in the placebo group). This is not a robust database from which to conclude that there is no effect in Black women.”
But, Dr. Chang and the CDER group argued, while the first study showed 17OHP “reduced the risk of recurrent preterm birth in both Black and non-Black participants, the lack of even a trend toward efficacy among either Black or non-Black women in [the PROLONG study] argues that the smaller proportion of Black women [in the PROLONG study] does not explain the lack of efficacy.”
In addition to race, there were other risk factors for preterm birth, such as tobacco, alcohol, and street drug use; marital status; and age that differed between the two study cohorts. Even after subcategorizing PROLONG trial participants into higher or lower risk for preterm birth based on these risk factors, Dr. Chang and associates still found no evidence of benefit to 17OHP treatment in any risk group.
Withdrawing approval of 17OHP for a recurrent preterm indication would still allow off-label prescribing, but would most likely end insurance coverage and eventually manufacturing of the drug, noted Dr. Greene and associates.
“When the majority of a population achieves little benefit from a drug, but a minority demographic group at greatest risk for a serious medical problem appears to obtain significant benefit, any decision that will ultimately make it impossible to obtain the drug should be undertaken cautiously,” they warned. “This issue is particularly pressing when that minority group may be the least able to find and financially afford work-arounds to obtain the needed medication in our complex medical system that has a history of failing to serve them well.”
Dr. Chang and associates reported they had no relevant financial disclosures. Dr. Greene and associates reported that they had no relevant conflicts of interest or financial disclosures. Dr. Greene reported he is employed by the New England Journal of Medicine as associate editor. Dr. Harrington reported being employed by the journal as statistical consultant. Dr. Klebanoff reported he was an author of the original article about 17OHP published in the journal and referenced in this article.
The Oct. 5, 2020 move by the Food and Drug Administration’s Center for Drug Evaluation and Research (CDER) suggesting the withdrawal of the approval of Makena incited some opposition.
Amag Pharmaceuticals’ 17 alpha-hydroxyprogesterone caproate (17OHP) injection received accelerated approval in 2011 to reduce the risk of recurrent preterm birth in women with previous unexplained preterm birth. Makena is the only drug approved for preventing recurrent preterm birth.
The back story
The approval was based on findings from a randomized, placebo-controlled trial that demonstrated a 34% relative risk reduction in births before 37 weeks – from 55% in the placebo arm to 36% in the 17OHP-treated arm.
The trial was not designed to measure neonatal outcomes, with the surrogate outcome of recurrent preterm birth being determined as “reasonably likely” to predict benefit to the neonate.
Subsequently, results of the required postapproval confirmatory PROLONG trialproduced conflicting results, failing to show a benefit of 17OHP on either preterm birth or neonatal outcome, which prompted the proposed withdrawal of the drug’s approval.
The CDER advisory committee agreed unanimously that the PROLONG trial did not support the clinical benefit of 17OHP, but the committee was not unanimous in deciding what to do. Of the 16 members, 9 voted to withdraw the drug’s approval, while seven voted to retain it and require another confirmatory trial.
When CDER recommends withdrawal, the company can request a public hearing, which it has done. The FDA commissioner will recommend whether to grant this request.
In the meantime, one from a group of three doctors who are against it and the other from the CDER.
Arguments from the opposing views
“We sympathize with women who are at risk for recurrent preterm birth that could result in death or significant lifelong health effects in neonates, but retaining on the market a drug not shown to be effective for this use does not protect or promote their health,” wrote Christina Chang, MD, MPH and associates from CDER.
On the other hand, “the widespread use of 17OHP after accelerated approval has not uncovered important safety signals,” countered Michael F. Greene, MD, from Massachusetts General Hospital, Boston; David Harrington, PhD, from the Harvard T. Chan School of Public Health, Boston; and Mark A. Klebanoff, MD, MPH, who was coauthor on the original preapproval study and is with Nationwide Children’s Hospital, the Ohio State University College of Medicine, and Ohio State University College of Public Health, all in Columbus. “Withdrawal of the approval for 17OHP, as imperfect as it may be, will leave a very vulnerable demographic group of U.S. women at high risk for this complication of pregnancy with absolutely no available therapeutic option.”
While both the preapproval study and postapproval PROLONG trial had the same enrollment criteria – namely women with a singleton pregnancy and previous singleton spontaneous preterm birth – all parties acknowledged that the studies ended up with very different cohorts. Approval of the drug in the United States made it difficult to recruit U.S. participants for the second trial “because of a lack of equipoise perceived by health care providers and patients,” noted Dr. Greene and associates, resulting in 75% of the PROLONG study’s cohort coming from Europe. This meant that 59% of those in the first study were non-Hispanic black compared with just 6.6% in the PROLONG study, a difference that is important because of the increased risk of preterm birth in Black women.
“Black women are generally underrepresented in U.S. clinical trials, and they are clearly underrepresented in the PROLONG study,” noted Dr. Greene and colleagues, adding that “the total number of qualifying composite neonatal outcome events among Blacks or African Americans in the entire PROLONG study population of 1,700 participants was 9 (6 of 69 in the 17OHP group and 3 of 40 in the placebo group). This is not a robust database from which to conclude that there is no effect in Black women.”
But, Dr. Chang and the CDER group argued, while the first study showed 17OHP “reduced the risk of recurrent preterm birth in both Black and non-Black participants, the lack of even a trend toward efficacy among either Black or non-Black women in [the PROLONG study] argues that the smaller proportion of Black women [in the PROLONG study] does not explain the lack of efficacy.”
In addition to race, there were other risk factors for preterm birth, such as tobacco, alcohol, and street drug use; marital status; and age that differed between the two study cohorts. Even after subcategorizing PROLONG trial participants into higher or lower risk for preterm birth based on these risk factors, Dr. Chang and associates still found no evidence of benefit to 17OHP treatment in any risk group.
Withdrawing approval of 17OHP for a recurrent preterm indication would still allow off-label prescribing, but would most likely end insurance coverage and eventually manufacturing of the drug, noted Dr. Greene and associates.
“When the majority of a population achieves little benefit from a drug, but a minority demographic group at greatest risk for a serious medical problem appears to obtain significant benefit, any decision that will ultimately make it impossible to obtain the drug should be undertaken cautiously,” they warned. “This issue is particularly pressing when that minority group may be the least able to find and financially afford work-arounds to obtain the needed medication in our complex medical system that has a history of failing to serve them well.”
Dr. Chang and associates reported they had no relevant financial disclosures. Dr. Greene and associates reported that they had no relevant conflicts of interest or financial disclosures. Dr. Greene reported he is employed by the New England Journal of Medicine as associate editor. Dr. Harrington reported being employed by the journal as statistical consultant. Dr. Klebanoff reported he was an author of the original article about 17OHP published in the journal and referenced in this article.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Maternal oxygen in labor: False reassurance?
False reassurance?
CASE Heart rate tracing suggests fetal distress
Ms. M. presents for elective induction of labor at 39 weeks’ gestation. During the course of her labor, a Category II fetal heart rate (FHR) tracing is noted, and maternal oxygen is administered as part of the intrauterine resuscitative efforts. Her infant ultimately was delivered vaginally with an arterial cord blood pH of 7.1 and Apgar scores of 5 and 7.
Should intrauterine resuscitation include maternal oxygen administration?
It is a common sight on labor and delivery: An FHR monitoring strip is noted to be a Category II tracing. There may be fetal tachycardia, late decelerations, or perhaps decreased variability. The nurse or physician goes to the laboring mother’s room, checks cervical dilation, changes the patient’s position, and puts an oxygen mask over her face.
The American College of Obstetricians and Gynecologists (ACOG) lists maternal oxygen administration, most commonly at 10 L/min via a nonrebreather face mask, as an intrauterine resuscitative measure for Category II or Category III FHR tracings.1 Maternal oxygen is used to treat abnormal FHR tracings in approximately half of all births in the United States.2 Despite these recommendations and the frequency of its use, however, evidence is limited that maternal oxygenation improves neonatal outcome. In fact, there is emerging evidence of potential harm.
Why use oxygen?
The use of maternal oxygen supplementation intuitively makes sense. We know that certain abnormalities in FHR tracings can signal fetal hypoxia. Left untreated, the hypoxia could lead to fetal acidemia and associated neonatal sequelae. Theoretically, the administration of maternal oxygen should lead to improved fetal oxygenation and improved fetal outcome. This is supported by studies from the 1960s that demonstrate improved FHR tracings after maternal oxygen administration.3
This idea was further supported by studies that demonstrated an increase in fetal oxygen levels when maternal oxygen is administered. Haydon and colleagues evaluated the administration of maternal oxygen in women with nonreassuring FHR tracings.4 Their data showed that maternal oxygen administration increased fetal oxygen as measured by fetal pulse oximetry. The lower the initial fetal oxygen levels prior to oxygen administration, the greater the increase.
Despite these findings, evidence for improved neonatal outcomes is lacking.5 While heart rate tracings and fetal oxygen saturation may be improved with maternal oxygen supplementation, neonatal morbidity appears to remain unchanged (FIGURE). In fact, newer research suggests potential harm. Although an improved FHR tracing may be comforting to the clinician, the end result may be less so. Given these findings on maternal oxygen supplementation, it is time to break this practice habit.
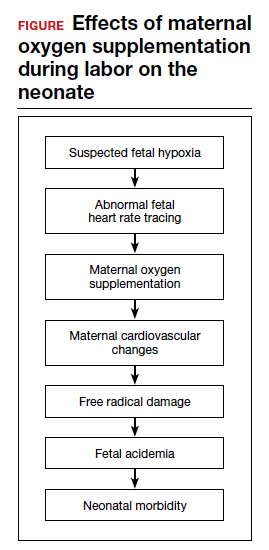
Maternal cardiovascular effects
Most of the literature on maternal hyperoxygenation focuses on fetal response. Before examining the effects on the fetus, however, we must consider the effect on the mother. Cardiovascular changes occur during and after maternal oxygen administration that should be taken into account.
McHugh and colleagues measured the hemodynamic changes in 46 pregnant and 20 nonpregnant women before, immediately, and 10 minutes after a 30-minute period of high-flow oxygen administration.6 While there were no changes in the nonpregnant women’s parameters, in the pregnant women heart rate and stroke volume were decreased after oxygen administration. Additionally, systemic vascular resistance increased and did not return to baseline by 10 minutes postadministration.
Since the purpose of the maternal oxygen administration is to increase oxygen to the fetus, this decrease in cardiac output and increase in systemic vascular resistance is concerning. These results may negate the intended effect of increased oxygen delivery to the fetus.
Continue to: Maternal and fetal oxidative stress...
Maternal and fetal oxidative stress
Assuming that the abnormal FHR tracing in our case patient is actually due to fetal hypoxia, it would seem prudent to increase fetal oxygenation. However, fetal hyperoxygenation may lead to free radical damage that could worsen neonatal outcomes. Oxidative stress, which can be caused by both hypoxia and hyperoxia, can lead to endothelial and cell receptor damage. This is known to contribute to the cerebral damage of hypoxic-ischemic encephalopathy.
In a randomized trial, Khaw and colleagues measured lipid peroxidases as a “free radical footprint” in women undergoing elective cesarean delivery who were administered oxygen or room air.7 Maternal and fetal oxygen levels were higher in the oxygen-supplementation group, but lipid peroxidases also were elevated. This finding suggests that the excess oxygen results in free radical formation and potentially negative effects on the neonate.
Although maternal oxygen supplementation frequently is viewed as harmless, this research shows that free radical damage may occur in the mother as well.
Additional research shows that longer durations of oxygen administration are correlated with worsening neonatal outcomes. In a study of liberal versus indicated oxygen use, the average time was approximately 90 minutes.8 Use for longer than 176 minutes was associated with lower oxygen levels in fetal blood. A proposed mechanism for this response is placental vasoconstriction thought to protect the fetus from free radical damage.
Again, if the goal is to increase oxygenation, prolonged maternal oxygen supplementation appears to produce the opposite effect.
Fetal acidemia and neonatal morbidity
If a fetus with an abnormal FHR tracing is thought to be hypoxic or acidemic, adding the potentially harmful effects of free radicals could worsen this condition. This is exactly what Raghuraman and colleagues demonstrated in a large prospective cohort analysis.9 While there was no difference in neonatal morbidity between those receiving oxygen and those on room air, there was a significant difference among infants with acidemia and hyperoxia. Composite morbidity (mechanical ventilation, hypothermic therapy, meconium aspiration, and death) was significantly increased in neonates with both hyperoxia and acidemia compared with nonacidemic hyperoxic infants.9 This is further supported by reports of an increased need for neonatal resuscitation and a fourfold increase in umbilical cord pH of less than 7.2.10
While intrauterine and extrauterine life certainly differ, these findings align with the pediatric literature that supports neonatal resuscitation with room air rather than 100% oxygen.11 Additionally, the intrauterine environment is relatively hypoxic, which may make free radical damage more severe.
Continue to: Oxygen use during the COVID-19 pandemic...
Oxygen use during the COVID-19 pandemic
While high-flow oxygen by mask is not considered an aerosol-generating procedure according to the Centers for Disease Control and Prevention, data are limited regarding the cleaning and filtering of oxygen. It is unknown if high-flow oxygen by mask increases the risk of infectious disease transmission to care providers. Therefore, in the midst of the COVID-19 pandemic, ACOG currently recommends against using supplemental oxygen for Category II and Category III tracings, since the benefits are not well established and the possibility of harm to providers may be increased.12 Oxygen supplementation still should be used in mothers with hypoxia.
Other intrauterine resuscitation options
Maternal oxygen administration does not appear beneficial for neonatal outcomes, but other methods can be used. An intravenous fluid bolus and lateral positioning of the mother, for example, are both associated with increased fetal oxygenation. Reducing uterine activity by discontinuing oxytocin or cervical ripening agents or by administering a tocolytic also can improve FHR abnormalities. Oxygen use should be reserved for patients with maternal hypoxia.
The bottom line
The liberal use of maternal oxygenation for the management of abnormal FHR tracings should be stopped. Clear evidence of its benefit is lacking, and the real possibility of fetal and maternal harm remains. This may be especially true during the COVID-19 pandemic. ●
- American College of Obstetricians and Gynecologists. Practice bulletin No. 116. Management of intrapartum fetal heart rate tracings. Obstet Gynecol. 2010;116:1232-1240.
- Hamel MS, Anderson BL, Rouse DJ. Oxygen for intrauterine resuscitation: of unproved benefit and potentially harmful. Am J Obstet Gynecol. 2014;211:124-127.
- Althabe O, Schwarcz RL, Pose SV, et al. Effects on fetal heart rate and fetal pO2 of oxygen administration to the mother. Am J Obstet Gynecol. 1967;98:858-870.
- Haydon ML, Gorenberg DM, Nageotte MP, et al. The effect of maternal oxygen administration on fetal pulse oximetry during labor in fetuses with nonreassuring fetal heart rate patterns. Am J Obstet Gynecol. 2006;195:735-738.
- Fawole B, Hofmeyr GJ. Maternal oxygen administration for fetal distress. Cochrane Database Syst Rev. 2012;12:CD0000136.
- McHugh A, El-Khuffash A, Bussmann N, et al. Hyperoxygenation in pregnancy exerts a more profound effect on cardiovascular hemodynamics than is observed in the nonpregnant state. Am J Obstet Gynecol. 2019;220:397.e1-397.e8.
- Khaw KS, Wang CC, Ngan Kee WD, et al. Effects of high inspired oxygen fraction during elective caesarean section under spinal anaesthesia on maternal and fetal oxygenation and lipid peroxidation. Br J Anaesth. 2002;88:18-23.
- Watkins VY, Martin S, Macones GA, et al. The duration of intrapartum supplemental oxygen administration and umbilical cord oxygen content. Am J Obstet Gynecol. 2020;223:440.e1-440.e7.
- Raghuraman N, Temming LA, Stout MJ, et al. Intrauterine hyperoxemia and risk of neonatal morbidity. Obstet Gynecol. 2017;129:676-682.
- Thorp JA, Trobough T, Evans R, et al. The effect of maternal oxygen administration during the second stage of labor on umbilical cord blood gas values: a randomized controlled prospective trial. Am J Obstet Gynecol. 1995;172(2 pt 1):465-474.
- Rabi Y, Rabi D, Yee W. Room air resuscitation of the depressed newborn: a systematic review and meta-analysis. Resuscitation. 2007;72:353-363.
- COVID-19 FAQs for Obstetrician-Gynecologists, Obstetrics. https://www.acog.org/clinical-information/physician-faqs/covid-19-faqs-for-ob-gyns-obstetrics. Accessed October 15, 2020.
CASE Heart rate tracing suggests fetal distress
Ms. M. presents for elective induction of labor at 39 weeks’ gestation. During the course of her labor, a Category II fetal heart rate (FHR) tracing is noted, and maternal oxygen is administered as part of the intrauterine resuscitative efforts. Her infant ultimately was delivered vaginally with an arterial cord blood pH of 7.1 and Apgar scores of 5 and 7.
Should intrauterine resuscitation include maternal oxygen administration?
It is a common sight on labor and delivery: An FHR monitoring strip is noted to be a Category II tracing. There may be fetal tachycardia, late decelerations, or perhaps decreased variability. The nurse or physician goes to the laboring mother’s room, checks cervical dilation, changes the patient’s position, and puts an oxygen mask over her face.
The American College of Obstetricians and Gynecologists (ACOG) lists maternal oxygen administration, most commonly at 10 L/min via a nonrebreather face mask, as an intrauterine resuscitative measure for Category II or Category III FHR tracings.1 Maternal oxygen is used to treat abnormal FHR tracings in approximately half of all births in the United States.2 Despite these recommendations and the frequency of its use, however, evidence is limited that maternal oxygenation improves neonatal outcome. In fact, there is emerging evidence of potential harm.
Why use oxygen?
The use of maternal oxygen supplementation intuitively makes sense. We know that certain abnormalities in FHR tracings can signal fetal hypoxia. Left untreated, the hypoxia could lead to fetal acidemia and associated neonatal sequelae. Theoretically, the administration of maternal oxygen should lead to improved fetal oxygenation and improved fetal outcome. This is supported by studies from the 1960s that demonstrate improved FHR tracings after maternal oxygen administration.3
This idea was further supported by studies that demonstrated an increase in fetal oxygen levels when maternal oxygen is administered. Haydon and colleagues evaluated the administration of maternal oxygen in women with nonreassuring FHR tracings.4 Their data showed that maternal oxygen administration increased fetal oxygen as measured by fetal pulse oximetry. The lower the initial fetal oxygen levels prior to oxygen administration, the greater the increase.
Despite these findings, evidence for improved neonatal outcomes is lacking.5 While heart rate tracings and fetal oxygen saturation may be improved with maternal oxygen supplementation, neonatal morbidity appears to remain unchanged (FIGURE). In fact, newer research suggests potential harm. Although an improved FHR tracing may be comforting to the clinician, the end result may be less so. Given these findings on maternal oxygen supplementation, it is time to break this practice habit.

Maternal cardiovascular effects
Most of the literature on maternal hyperoxygenation focuses on fetal response. Before examining the effects on the fetus, however, we must consider the effect on the mother. Cardiovascular changes occur during and after maternal oxygen administration that should be taken into account.
McHugh and colleagues measured the hemodynamic changes in 46 pregnant and 20 nonpregnant women before, immediately, and 10 minutes after a 30-minute period of high-flow oxygen administration.6 While there were no changes in the nonpregnant women’s parameters, in the pregnant women heart rate and stroke volume were decreased after oxygen administration. Additionally, systemic vascular resistance increased and did not return to baseline by 10 minutes postadministration.
Since the purpose of the maternal oxygen administration is to increase oxygen to the fetus, this decrease in cardiac output and increase in systemic vascular resistance is concerning. These results may negate the intended effect of increased oxygen delivery to the fetus.
Continue to: Maternal and fetal oxidative stress...
Maternal and fetal oxidative stress
Assuming that the abnormal FHR tracing in our case patient is actually due to fetal hypoxia, it would seem prudent to increase fetal oxygenation. However, fetal hyperoxygenation may lead to free radical damage that could worsen neonatal outcomes. Oxidative stress, which can be caused by both hypoxia and hyperoxia, can lead to endothelial and cell receptor damage. This is known to contribute to the cerebral damage of hypoxic-ischemic encephalopathy.
In a randomized trial, Khaw and colleagues measured lipid peroxidases as a “free radical footprint” in women undergoing elective cesarean delivery who were administered oxygen or room air.7 Maternal and fetal oxygen levels were higher in the oxygen-supplementation group, but lipid peroxidases also were elevated. This finding suggests that the excess oxygen results in free radical formation and potentially negative effects on the neonate.
Although maternal oxygen supplementation frequently is viewed as harmless, this research shows that free radical damage may occur in the mother as well.
Additional research shows that longer durations of oxygen administration are correlated with worsening neonatal outcomes. In a study of liberal versus indicated oxygen use, the average time was approximately 90 minutes.8 Use for longer than 176 minutes was associated with lower oxygen levels in fetal blood. A proposed mechanism for this response is placental vasoconstriction thought to protect the fetus from free radical damage.
Again, if the goal is to increase oxygenation, prolonged maternal oxygen supplementation appears to produce the opposite effect.
Fetal acidemia and neonatal morbidity
If a fetus with an abnormal FHR tracing is thought to be hypoxic or acidemic, adding the potentially harmful effects of free radicals could worsen this condition. This is exactly what Raghuraman and colleagues demonstrated in a large prospective cohort analysis.9 While there was no difference in neonatal morbidity between those receiving oxygen and those on room air, there was a significant difference among infants with acidemia and hyperoxia. Composite morbidity (mechanical ventilation, hypothermic therapy, meconium aspiration, and death) was significantly increased in neonates with both hyperoxia and acidemia compared with nonacidemic hyperoxic infants.9 This is further supported by reports of an increased need for neonatal resuscitation and a fourfold increase in umbilical cord pH of less than 7.2.10
While intrauterine and extrauterine life certainly differ, these findings align with the pediatric literature that supports neonatal resuscitation with room air rather than 100% oxygen.11 Additionally, the intrauterine environment is relatively hypoxic, which may make free radical damage more severe.
Continue to: Oxygen use during the COVID-19 pandemic...
Oxygen use during the COVID-19 pandemic
While high-flow oxygen by mask is not considered an aerosol-generating procedure according to the Centers for Disease Control and Prevention, data are limited regarding the cleaning and filtering of oxygen. It is unknown if high-flow oxygen by mask increases the risk of infectious disease transmission to care providers. Therefore, in the midst of the COVID-19 pandemic, ACOG currently recommends against using supplemental oxygen for Category II and Category III tracings, since the benefits are not well established and the possibility of harm to providers may be increased.12 Oxygen supplementation still should be used in mothers with hypoxia.
Other intrauterine resuscitation options
Maternal oxygen administration does not appear beneficial for neonatal outcomes, but other methods can be used. An intravenous fluid bolus and lateral positioning of the mother, for example, are both associated with increased fetal oxygenation. Reducing uterine activity by discontinuing oxytocin or cervical ripening agents or by administering a tocolytic also can improve FHR abnormalities. Oxygen use should be reserved for patients with maternal hypoxia.
The bottom line
The liberal use of maternal oxygenation for the management of abnormal FHR tracings should be stopped. Clear evidence of its benefit is lacking, and the real possibility of fetal and maternal harm remains. This may be especially true during the COVID-19 pandemic. ●
CASE Heart rate tracing suggests fetal distress
Ms. M. presents for elective induction of labor at 39 weeks’ gestation. During the course of her labor, a Category II fetal heart rate (FHR) tracing is noted, and maternal oxygen is administered as part of the intrauterine resuscitative efforts. Her infant ultimately was delivered vaginally with an arterial cord blood pH of 7.1 and Apgar scores of 5 and 7.
Should intrauterine resuscitation include maternal oxygen administration?
It is a common sight on labor and delivery: An FHR monitoring strip is noted to be a Category II tracing. There may be fetal tachycardia, late decelerations, or perhaps decreased variability. The nurse or physician goes to the laboring mother’s room, checks cervical dilation, changes the patient’s position, and puts an oxygen mask over her face.
The American College of Obstetricians and Gynecologists (ACOG) lists maternal oxygen administration, most commonly at 10 L/min via a nonrebreather face mask, as an intrauterine resuscitative measure for Category II or Category III FHR tracings.1 Maternal oxygen is used to treat abnormal FHR tracings in approximately half of all births in the United States.2 Despite these recommendations and the frequency of its use, however, evidence is limited that maternal oxygenation improves neonatal outcome. In fact, there is emerging evidence of potential harm.
Why use oxygen?
The use of maternal oxygen supplementation intuitively makes sense. We know that certain abnormalities in FHR tracings can signal fetal hypoxia. Left untreated, the hypoxia could lead to fetal acidemia and associated neonatal sequelae. Theoretically, the administration of maternal oxygen should lead to improved fetal oxygenation and improved fetal outcome. This is supported by studies from the 1960s that demonstrate improved FHR tracings after maternal oxygen administration.3
This idea was further supported by studies that demonstrated an increase in fetal oxygen levels when maternal oxygen is administered. Haydon and colleagues evaluated the administration of maternal oxygen in women with nonreassuring FHR tracings.4 Their data showed that maternal oxygen administration increased fetal oxygen as measured by fetal pulse oximetry. The lower the initial fetal oxygen levels prior to oxygen administration, the greater the increase.
Despite these findings, evidence for improved neonatal outcomes is lacking.5 While heart rate tracings and fetal oxygen saturation may be improved with maternal oxygen supplementation, neonatal morbidity appears to remain unchanged (FIGURE). In fact, newer research suggests potential harm. Although an improved FHR tracing may be comforting to the clinician, the end result may be less so. Given these findings on maternal oxygen supplementation, it is time to break this practice habit.

Maternal cardiovascular effects
Most of the literature on maternal hyperoxygenation focuses on fetal response. Before examining the effects on the fetus, however, we must consider the effect on the mother. Cardiovascular changes occur during and after maternal oxygen administration that should be taken into account.
McHugh and colleagues measured the hemodynamic changes in 46 pregnant and 20 nonpregnant women before, immediately, and 10 minutes after a 30-minute period of high-flow oxygen administration.6 While there were no changes in the nonpregnant women’s parameters, in the pregnant women heart rate and stroke volume were decreased after oxygen administration. Additionally, systemic vascular resistance increased and did not return to baseline by 10 minutes postadministration.
Since the purpose of the maternal oxygen administration is to increase oxygen to the fetus, this decrease in cardiac output and increase in systemic vascular resistance is concerning. These results may negate the intended effect of increased oxygen delivery to the fetus.
Continue to: Maternal and fetal oxidative stress...
Maternal and fetal oxidative stress
Assuming that the abnormal FHR tracing in our case patient is actually due to fetal hypoxia, it would seem prudent to increase fetal oxygenation. However, fetal hyperoxygenation may lead to free radical damage that could worsen neonatal outcomes. Oxidative stress, which can be caused by both hypoxia and hyperoxia, can lead to endothelial and cell receptor damage. This is known to contribute to the cerebral damage of hypoxic-ischemic encephalopathy.
In a randomized trial, Khaw and colleagues measured lipid peroxidases as a “free radical footprint” in women undergoing elective cesarean delivery who were administered oxygen or room air.7 Maternal and fetal oxygen levels were higher in the oxygen-supplementation group, but lipid peroxidases also were elevated. This finding suggests that the excess oxygen results in free radical formation and potentially negative effects on the neonate.
Although maternal oxygen supplementation frequently is viewed as harmless, this research shows that free radical damage may occur in the mother as well.
Additional research shows that longer durations of oxygen administration are correlated with worsening neonatal outcomes. In a study of liberal versus indicated oxygen use, the average time was approximately 90 minutes.8 Use for longer than 176 minutes was associated with lower oxygen levels in fetal blood. A proposed mechanism for this response is placental vasoconstriction thought to protect the fetus from free radical damage.
Again, if the goal is to increase oxygenation, prolonged maternal oxygen supplementation appears to produce the opposite effect.
Fetal acidemia and neonatal morbidity
If a fetus with an abnormal FHR tracing is thought to be hypoxic or acidemic, adding the potentially harmful effects of free radicals could worsen this condition. This is exactly what Raghuraman and colleagues demonstrated in a large prospective cohort analysis.9 While there was no difference in neonatal morbidity between those receiving oxygen and those on room air, there was a significant difference among infants with acidemia and hyperoxia. Composite morbidity (mechanical ventilation, hypothermic therapy, meconium aspiration, and death) was significantly increased in neonates with both hyperoxia and acidemia compared with nonacidemic hyperoxic infants.9 This is further supported by reports of an increased need for neonatal resuscitation and a fourfold increase in umbilical cord pH of less than 7.2.10
While intrauterine and extrauterine life certainly differ, these findings align with the pediatric literature that supports neonatal resuscitation with room air rather than 100% oxygen.11 Additionally, the intrauterine environment is relatively hypoxic, which may make free radical damage more severe.
Continue to: Oxygen use during the COVID-19 pandemic...
Oxygen use during the COVID-19 pandemic
While high-flow oxygen by mask is not considered an aerosol-generating procedure according to the Centers for Disease Control and Prevention, data are limited regarding the cleaning and filtering of oxygen. It is unknown if high-flow oxygen by mask increases the risk of infectious disease transmission to care providers. Therefore, in the midst of the COVID-19 pandemic, ACOG currently recommends against using supplemental oxygen for Category II and Category III tracings, since the benefits are not well established and the possibility of harm to providers may be increased.12 Oxygen supplementation still should be used in mothers with hypoxia.
Other intrauterine resuscitation options
Maternal oxygen administration does not appear beneficial for neonatal outcomes, but other methods can be used. An intravenous fluid bolus and lateral positioning of the mother, for example, are both associated with increased fetal oxygenation. Reducing uterine activity by discontinuing oxytocin or cervical ripening agents or by administering a tocolytic also can improve FHR abnormalities. Oxygen use should be reserved for patients with maternal hypoxia.
The bottom line
The liberal use of maternal oxygenation for the management of abnormal FHR tracings should be stopped. Clear evidence of its benefit is lacking, and the real possibility of fetal and maternal harm remains. This may be especially true during the COVID-19 pandemic. ●
- American College of Obstetricians and Gynecologists. Practice bulletin No. 116. Management of intrapartum fetal heart rate tracings. Obstet Gynecol. 2010;116:1232-1240.
- Hamel MS, Anderson BL, Rouse DJ. Oxygen for intrauterine resuscitation: of unproved benefit and potentially harmful. Am J Obstet Gynecol. 2014;211:124-127.
- Althabe O, Schwarcz RL, Pose SV, et al. Effects on fetal heart rate and fetal pO2 of oxygen administration to the mother. Am J Obstet Gynecol. 1967;98:858-870.
- Haydon ML, Gorenberg DM, Nageotte MP, et al. The effect of maternal oxygen administration on fetal pulse oximetry during labor in fetuses with nonreassuring fetal heart rate patterns. Am J Obstet Gynecol. 2006;195:735-738.
- Fawole B, Hofmeyr GJ. Maternal oxygen administration for fetal distress. Cochrane Database Syst Rev. 2012;12:CD0000136.
- McHugh A, El-Khuffash A, Bussmann N, et al. Hyperoxygenation in pregnancy exerts a more profound effect on cardiovascular hemodynamics than is observed in the nonpregnant state. Am J Obstet Gynecol. 2019;220:397.e1-397.e8.
- Khaw KS, Wang CC, Ngan Kee WD, et al. Effects of high inspired oxygen fraction during elective caesarean section under spinal anaesthesia on maternal and fetal oxygenation and lipid peroxidation. Br J Anaesth. 2002;88:18-23.
- Watkins VY, Martin S, Macones GA, et al. The duration of intrapartum supplemental oxygen administration and umbilical cord oxygen content. Am J Obstet Gynecol. 2020;223:440.e1-440.e7.
- Raghuraman N, Temming LA, Stout MJ, et al. Intrauterine hyperoxemia and risk of neonatal morbidity. Obstet Gynecol. 2017;129:676-682.
- Thorp JA, Trobough T, Evans R, et al. The effect of maternal oxygen administration during the second stage of labor on umbilical cord blood gas values: a randomized controlled prospective trial. Am J Obstet Gynecol. 1995;172(2 pt 1):465-474.
- Rabi Y, Rabi D, Yee W. Room air resuscitation of the depressed newborn: a systematic review and meta-analysis. Resuscitation. 2007;72:353-363.
- COVID-19 FAQs for Obstetrician-Gynecologists, Obstetrics. https://www.acog.org/clinical-information/physician-faqs/covid-19-faqs-for-ob-gyns-obstetrics. Accessed October 15, 2020.
- American College of Obstetricians and Gynecologists. Practice bulletin No. 116. Management of intrapartum fetal heart rate tracings. Obstet Gynecol. 2010;116:1232-1240.
- Hamel MS, Anderson BL, Rouse DJ. Oxygen for intrauterine resuscitation: of unproved benefit and potentially harmful. Am J Obstet Gynecol. 2014;211:124-127.
- Althabe O, Schwarcz RL, Pose SV, et al. Effects on fetal heart rate and fetal pO2 of oxygen administration to the mother. Am J Obstet Gynecol. 1967;98:858-870.
- Haydon ML, Gorenberg DM, Nageotte MP, et al. The effect of maternal oxygen administration on fetal pulse oximetry during labor in fetuses with nonreassuring fetal heart rate patterns. Am J Obstet Gynecol. 2006;195:735-738.
- Fawole B, Hofmeyr GJ. Maternal oxygen administration for fetal distress. Cochrane Database Syst Rev. 2012;12:CD0000136.
- McHugh A, El-Khuffash A, Bussmann N, et al. Hyperoxygenation in pregnancy exerts a more profound effect on cardiovascular hemodynamics than is observed in the nonpregnant state. Am J Obstet Gynecol. 2019;220:397.e1-397.e8.
- Khaw KS, Wang CC, Ngan Kee WD, et al. Effects of high inspired oxygen fraction during elective caesarean section under spinal anaesthesia on maternal and fetal oxygenation and lipid peroxidation. Br J Anaesth. 2002;88:18-23.
- Watkins VY, Martin S, Macones GA, et al. The duration of intrapartum supplemental oxygen administration and umbilical cord oxygen content. Am J Obstet Gynecol. 2020;223:440.e1-440.e7.
- Raghuraman N, Temming LA, Stout MJ, et al. Intrauterine hyperoxemia and risk of neonatal morbidity. Obstet Gynecol. 2017;129:676-682.
- Thorp JA, Trobough T, Evans R, et al. The effect of maternal oxygen administration during the second stage of labor on umbilical cord blood gas values: a randomized controlled prospective trial. Am J Obstet Gynecol. 1995;172(2 pt 1):465-474.
- Rabi Y, Rabi D, Yee W. Room air resuscitation of the depressed newborn: a systematic review and meta-analysis. Resuscitation. 2007;72:353-363.
- COVID-19 FAQs for Obstetrician-Gynecologists, Obstetrics. https://www.acog.org/clinical-information/physician-faqs/covid-19-faqs-for-ob-gyns-obstetrics. Accessed October 15, 2020.
False reassurance?
False reassurance?
Syphilis: Cutting risk through primary prevention and prenatal screening
CASE Pregnant woman with positive Treponema pallidum antibody test
A 30-year-old primigravida at 10 weeks and 4 days of gestation by her last menstrual period presents to your office for her initial prenatal visit. She expresses no concerns. You order the standard set of laboratory tests, including a sexually transmitted infection (STI) screening panel. Consistent with your institution’s use of the reverse algorithm for syphilis screening, you obtain a Treponema pallidum antibody test, which reflexes to the rapid plasma reagin (RPR) test. Three days later, you receive a notification that this patient’s T pallidum antibody result was positive, followed by negative RPR test results. The follow-up T pallidum particle agglutination (TP-PA) test also was negative. Given these findings, you consider:
- What is the correct interpretation of the patient’s sequence of test results?
- Is she infected, and does she require treatment?
Meet our perpetrator
Syphilis has plagued society since the late 15th century, although its causative agent, the spirochete T pallidum, was not recognized until 1905.1,2T pallidum bacteria are transmitted via sexual contact, as well as through vertical transmission during pregnancy or delivery. Infection with syphilis is reported in 50% to 60% of sexual partners after a single exposure to an infected individual with early syphilis, and the mean incubation period is 21 days.3T pallidum can cross the placenta and infect a fetus as early as the sixth week of gestation.3 Congenital syphilis infections occur in the neonates of 50% to 80% of women with untreated primary, secondary, or early latent syphilis infections; maternal syphilis is associated with a 21% increased risk of stillbirth, a 6% increased risk of preterm delivery, and a 9% increased risk of neonatal death.4,5 Additionally, syphilis infection is associated with a high risk of HIV infection, as well as coinfection with other STIs.1
Given the highly infective nature of T pallidum, as well as the severity of the potential consequences of infection for both mothers and babies, primary prevention, education of at-risk populations, and early recognition of clinical features of syphilis infection are of utmost importance in preventing morbidity and mortality. In this article, we review the epidemiology and extensive clinical manifestations of syphilis, as well as current screening recommendations and treatment for pregnant women.
The extent of the problem today
Although US rates of syphilis have ebbed and flowed for the past several decades, the current incidence has grown exponentially in recent years, with the number of cases reported to the Centers for Disease Control and Prevention (CDC) increasing by 71% from 2014 to 2018.6 During this time period, reported cases of primary and secondary syphilis in women more than doubled (172.7% and 165.4%, respectively) according to CDC data, accompanied by a parallel rise in reported cases of congenital syphilis in both live and stillborn infants.6 In 2018, the CDC reported a national rate of congenital syphilis of 33.1 cases per 100,000 live births, a 39.7% rise compared with data from 2017.6
Those most at risk. Risk factors for syphilis infection include age younger than 30 years, low socioeconomic status, substance abuse, HIV infection, concurrent STIs, and high-risk sexual activity (sex with multiple high-risk partners).3 Additionally, reported rates of primary and secondary syphilis infections, as well as congenital syphilis infections, are more elevated among women who identify as Black, American Indian/Alaska Native, and/or Hispanic.6 Congenital infections in the United States are correlated with a lack of prenatal care, which has been similarly linked with racial and socioeconomic disparities, as well as with untreated mental health and substance use disorders and recent immigration to the United States.5,7
Continue to: The many phases of syphilis...
The many phases of syphilis
The characteristic lesion of primary syphilis is a chancre, which is a painless, ulcerative lesion with raised borders and a clean, indurated base appearing at the site of spirochete entry (FIGURE 1). Chancres most commonly appear in the genital area, with the most frequent sites in females being within the vaginal canal or on the cervix. Primary chancres tend to heal spontaneously within 3 to 6 weeks, even without treatment, and frequently are accompanied by painless inguinal lymphadenopathy. Given that the most common chancre sites are not immediately apparent, primary infections in women often go undetected.3 In fact, it is essential for clinicians to recognize that, in our routine practice, most patients with syphilis will not be symptomatic at all, and the diagnosis will only be made by serologic screening.
Following resolution of the primary phase, the patient may enter the secondary stage of T pallidum infection. During this stage, spirochetes may disseminate throughout the bloodstream to infect all major organ systems. The principal manifestations of secondary syphilis include a diffuse maculopapular rash that begins on the trunk and proximal extremities and spreads to include the palms and soles (FIGURE 2); mucosal lesions, such as mucous patches and condyloma lata (FIGURE 3); nonscarring alopecia; periostitis; generalized lymphadenopathy; and, in some cases, hepatitis or nephritis.1,3
Secondary syphilis usually clears within 2 to 6 weeks, with the patient then entering the early latent stage of syphilis. During this period, up to 25% of patients are subject to flares of secondary syphilitic lesions but otherwise are asymptomatic.1,3,4 These recurrences tend to occur within 1 year, hence the distinction between early and late latent stages. Once a year has passed, patients are not contagious by sexual transmission and are unlikely to suffer a relapse of secondary symptoms.1,3 However, late latent syphilis is characterized by periods of intermittent bacteremia that allow for seeding of the placenta and infection in about 10% of fetuses.5
Untreated, about 40% of patients will progress to the tertiary stage of syphilis, which is characterized by gummas affecting the skin and mucous membranes (FIGURE 4) and cardiovascular manifestations including arterial aneurysms and aortic insufficiency.3
Neurologic manifestations of syphilis may arise during any of the above stages, though the most characteristic manifestations tend to appear decades after the primary infection. Early neurosyphilis may present as meningitis, with or without concomitant ocular syphilis (uveitis, retinitis) and/or as otic syphilis (hearing loss, persistent tinnitus).1,5 Patients with late (tertiary) neurosyphilis tend to exhibit meningovascular symptoms similar to stroke (aphasia, hemiplegia, seizures) and/or parenchymal effects such as general paresis. Tabes dorsalis (manifestations of which include urinary and rectal incontinence, lightning pains, and ataxia) is a late-onset manifestation.1,3
Congenital syphilis can be subdivided into an early and late stage. The first stage, in which clinical findings occur within the first 2 years of life, commonly features a desquamating rash, hepatomegaly, and rhinitis. Anemia, thrombocytopenia, periostitis, and osteomyelitis also have been documented.5 Of note, two-thirds of infants are asymptomatic at birth and may not develop such clinical manifestations for 3 to 8 weeks.3 If untreated, early congenital infection may progress to late manifestations, such as Hutchinson teeth, mulberry molars, interstitial keratitis, deafness, saddle nose, saber shins, and such neurologic abnormalities as developmental delay and general paresis.3
Continue to: Prenatal screening and diagnosis...
Prenatal screening and diagnosis
Current recommendations issued by the CDC and the American College of Obstetricians and Gynecologists state that all pregnant women should be screened for syphilis infection at their first presentation to care, with repeat screening between 28 and 32 weeks of gestation and at birth, for women living in areas with a high prevalence of syphilis and/or with any of the aforementioned risk factors.3,5 Given that providers may be unfamiliar with the prevalence of syphilis in their area, and that patients may acquire or develop an infection later on in their pregnancy, researchers have begun to investigate the feasibility of universal third-trimester screening. While the cost-effectiveness of such a protocol is disputed, recent studies suggest that it may result in a substantial decrease in adverse maternal and fetal outcomes.8,9
Diagnostic tests
The traditional algorithm for the diagnosis of syphilis infection begins with a nontreponemal screening test, such as the RPR or the Venereal Disease Research Laboratory test. If positive, these screening tests are followed by a confirmatory treponemal test, such as the

The “reverse” screening algorithm begins with the FTA and, if positive, reflexes to the RPR. A reactive RPR indicates an active infection, and the patient should be treated. A negative RPR should be followed by the TP-PA to rule out a false-positive immunoglobulin G test. If the TP-PA test result is positive, the diagnosis of syphilis is confirmed (FIGURE 6). It is crucial to understand, however, that treponemal antibodies will remain positive for a patient’s lifetime, and someone who may have been treated for syphilis in the past also will screen positive. Once 2 treponemal tests are positive, physicians should take a careful history to assess prior infection risk and treatment status. A negative TP-PA excludes a diagnosis of syphilis.
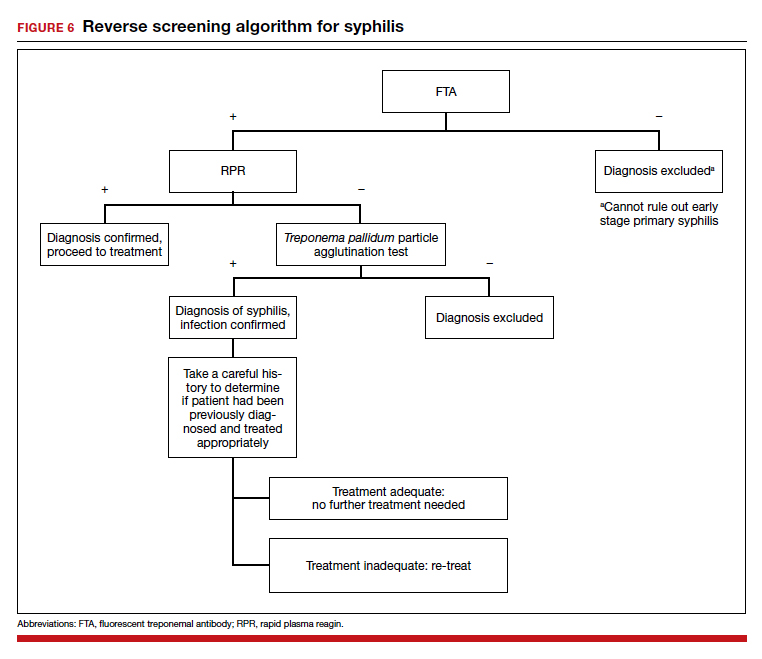
Advantages of the reverse screening algorithm. Nontreponemal tests are inexpensive and easy to perform, and titers allow for identification of a baseline to evaluate response to treatment.11 However, given the fluctuation of RPR sensitivity (depending on stage of disease and a decreased ability to detect primary and latent stages of syphilis), there has been a resurgence of interest in the reverse algorithm.11 While reverse screening has been found to incur higher costs, and may result in overtreatment and increased stress due to false-positive results,12 there is evidence to suggest that this algorithm is more sensitive for primary and latent infections.8,11,13-15
Given the rise in prevalence of syphilis infections in the United States over the past decade, and therefore a higher pretest probability of syphilis in the population, we favor the reverse screening algorithm in obstetrics, particularly given the risks of adverse maternal and fetal outcomes.
Treating syphilis in pregnancy
Parenteral benzathine penicillin G is the only currently recommended medication for the treatment of syphilis in pregnancy. This drug is effective in treating maternal infection and in preventing fetal infections, as well as in treating established fetal infections.3,5 Regimens differ depending on the stage of syphilis infection (TABLE). Treatment for presumed early syphilis is recommended for women who have had sexual contact with a partner diagnosed with primary, secondary, or early latent syphilis within 3 months of their current pregnancy.5 Any patient with diagnosed syphilis who demonstrates clinical signs of neurologic involvement should undergo lumbar puncture to assess for evidence of neurosyphilis.3 CDC guidelines recommend that patients who report an allergy to penicillin undergo desensitization therapy in a controlled setting, as other antibiotics that have been investigated in the treatment of syphilis are either not appropriate due to teratogenicity or due to suboptimal fetal treatment.3,5
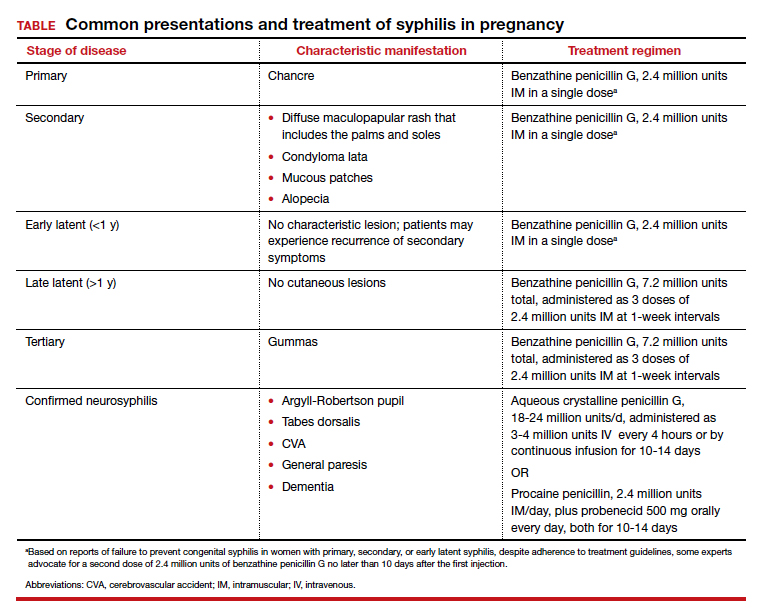
Syphilotherapy may lead to the Jarisch-Herxheimer reaction, which is an acute systemic reaction to inflammatory cytokines produced in response to lipopolysaccharide released by dying spirochetes.5 This reaction is characterized by fever, chills, myalgia, headache, hypotension, and worsening of cutaneous lesions. Preterm labor and delivery and fetal heart rate tracing abnormalities also have been documented in pregnant women experiencing this reaction, particularly during the second half of pregnancy.16 Prior to the start of treatment, a detailed sonographic assessment should be performed to assess the fetus for signs of early syphilis, including hepatomegaly, elevated peak systolic velocity of the middle cerebral artery (indicative of fetal anemia), polyhydramnios, placentomegaly, or hydrops.5,7
CASE Resolved
The combination of the patient’s test results—positive FTA, negative RPR, and negative TP-PA—suggest a false-positive treponemal assay. This sequence of tests excludes a diagnosis of syphilis; therefore, no treatment is necessary. Depending on the prevalence of syphilis in the patient’s geographic location, as well as her sexual history, rescreening between 28 and 32 weeks may be warranted. ●
- Ghanem KG, Ram S, Rice PA. The modern epidemic of syphilis. N Engl J Med. 2020;382:845-854.
- Barnett R. Syphilis. Lancet. 2018;391:1471.
- Duff P. Maternal and fetal infections. In: Resnik R, Lockwood CJ, Moore T, et al. Creasy and Resnik's Maternal-Fetal Medicine: Principles and Practice. 8th ed. Philadelphia, PA: Elsevier; 2018:862-919.
- Gomez GB, Kamb ML, Newman LM, et al. Untreated maternal syphilis and adverse outcomes of pregnancy: a systematic review and meta-analysis. Bull World Health Organ. 2013;91:217-226.
- Adhikari EH. Syphilis in pregnancy. Obstet Gynecol. 2020;135:1121-1135.
- Syphilis. CDC website. https://www.cdc.gov/std/stats18/syphilis.htm. Published October 1, 2019. Accessed October 6, 2020.
- Rac MF, Revell PA, Eppes CS. Syphilis during pregnancy: a preventable threat to maternal-fetal health. Am J Obstet Gynecol. 2017;4:352-363.
- Dunseth CD, Ford BA, Krasowski MD. Traditional versus reverse syphilis algorithms: a comparison at a large academic medical center. Pract Lab Med. 2017;8:52-59.
- Hersh AR, Megli CJ, Caughey AB. Repeat screening for syphilis in the third trimester of pregnancy: a cost-effectiveness analysis. Obstet Gynecol. 2018;132:699-706.
- Albright CM, Emerson JB, Werner EF, et al. Third trimester prenatal syphilis screening: a cost-effectiveness analysis. Obstet Gynecol. 2015;126:479-485.
- Seña AC, White BL, Sparling PF. Novel Treponema pallidum serologic tests: a paradigm shift in syphilis screening for the 21st century. Clin Infect Dis. 2010;51:700-708.
- Owusu-Edusei K Jr, Peterman TA, Ballard RC. Serologic testing for syphilis in the United States: a cost-effectiveness analysis of two screening algorithms. Sex Transm Dis. 2011;38:1-7.
- Huh HJ, Chung JW, Park SY, et al. Comparison of automated treponemal and nontreponemal test algorithms as first-line syphilis screening assays. Ann Lab Med. 2016;36:23-27.
- Centers for Disease Control and Prevention. Syphilis testing algorithms using treponemal test for initial screening-four laboratories. New York City, 2005-2006. MMWR Morb Mortal Wkly Rep. 2008;57:872-875.
- Mishra S, Boily MC, Ng V, et al. The laboratory impact of changing syphilis screening from the rapid-plasma reagin to a treponemal enzyme immunoassay: a case-study from the greater Toronto area. Sex Transm Dis. 2011;38:190-196.
- Klein VR, Cox SM, Mitchell MD, et al. The Jarisch-Herzheimer reaction complicating syphilotherapy in pregnancy. Obstet Gynecol. 1990;75:375-380.
CASE Pregnant woman with positive Treponema pallidum antibody test
A 30-year-old primigravida at 10 weeks and 4 days of gestation by her last menstrual period presents to your office for her initial prenatal visit. She expresses no concerns. You order the standard set of laboratory tests, including a sexually transmitted infection (STI) screening panel. Consistent with your institution’s use of the reverse algorithm for syphilis screening, you obtain a Treponema pallidum antibody test, which reflexes to the rapid plasma reagin (RPR) test. Three days later, you receive a notification that this patient’s T pallidum antibody result was positive, followed by negative RPR test results. The follow-up T pallidum particle agglutination (TP-PA) test also was negative. Given these findings, you consider:
- What is the correct interpretation of the patient’s sequence of test results?
- Is she infected, and does she require treatment?
Meet our perpetrator
Syphilis has plagued society since the late 15th century, although its causative agent, the spirochete T pallidum, was not recognized until 1905.1,2T pallidum bacteria are transmitted via sexual contact, as well as through vertical transmission during pregnancy or delivery. Infection with syphilis is reported in 50% to 60% of sexual partners after a single exposure to an infected individual with early syphilis, and the mean incubation period is 21 days.3T pallidum can cross the placenta and infect a fetus as early as the sixth week of gestation.3 Congenital syphilis infections occur in the neonates of 50% to 80% of women with untreated primary, secondary, or early latent syphilis infections; maternal syphilis is associated with a 21% increased risk of stillbirth, a 6% increased risk of preterm delivery, and a 9% increased risk of neonatal death.4,5 Additionally, syphilis infection is associated with a high risk of HIV infection, as well as coinfection with other STIs.1
Given the highly infective nature of T pallidum, as well as the severity of the potential consequences of infection for both mothers and babies, primary prevention, education of at-risk populations, and early recognition of clinical features of syphilis infection are of utmost importance in preventing morbidity and mortality. In this article, we review the epidemiology and extensive clinical manifestations of syphilis, as well as current screening recommendations and treatment for pregnant women.
The extent of the problem today
Although US rates of syphilis have ebbed and flowed for the past several decades, the current incidence has grown exponentially in recent years, with the number of cases reported to the Centers for Disease Control and Prevention (CDC) increasing by 71% from 2014 to 2018.6 During this time period, reported cases of primary and secondary syphilis in women more than doubled (172.7% and 165.4%, respectively) according to CDC data, accompanied by a parallel rise in reported cases of congenital syphilis in both live and stillborn infants.6 In 2018, the CDC reported a national rate of congenital syphilis of 33.1 cases per 100,000 live births, a 39.7% rise compared with data from 2017.6
Those most at risk. Risk factors for syphilis infection include age younger than 30 years, low socioeconomic status, substance abuse, HIV infection, concurrent STIs, and high-risk sexual activity (sex with multiple high-risk partners).3 Additionally, reported rates of primary and secondary syphilis infections, as well as congenital syphilis infections, are more elevated among women who identify as Black, American Indian/Alaska Native, and/or Hispanic.6 Congenital infections in the United States are correlated with a lack of prenatal care, which has been similarly linked with racial and socioeconomic disparities, as well as with untreated mental health and substance use disorders and recent immigration to the United States.5,7
Continue to: The many phases of syphilis...
The many phases of syphilis
The characteristic lesion of primary syphilis is a chancre, which is a painless, ulcerative lesion with raised borders and a clean, indurated base appearing at the site of spirochete entry (FIGURE 1). Chancres most commonly appear in the genital area, with the most frequent sites in females being within the vaginal canal or on the cervix. Primary chancres tend to heal spontaneously within 3 to 6 weeks, even without treatment, and frequently are accompanied by painless inguinal lymphadenopathy. Given that the most common chancre sites are not immediately apparent, primary infections in women often go undetected.3 In fact, it is essential for clinicians to recognize that, in our routine practice, most patients with syphilis will not be symptomatic at all, and the diagnosis will only be made by serologic screening.

Following resolution of the primary phase, the patient may enter the secondary stage of T pallidum infection. During this stage, spirochetes may disseminate throughout the bloodstream to infect all major organ systems. The principal manifestations of secondary syphilis include a diffuse maculopapular rash that begins on the trunk and proximal extremities and spreads to include the palms and soles (FIGURE 2); mucosal lesions, such as mucous patches and condyloma lata (FIGURE 3); nonscarring alopecia; periostitis; generalized lymphadenopathy; and, in some cases, hepatitis or nephritis.1,3

Secondary syphilis usually clears within 2 to 6 weeks, with the patient then entering the early latent stage of syphilis. During this period, up to 25% of patients are subject to flares of secondary syphilitic lesions but otherwise are asymptomatic.1,3,4 These recurrences tend to occur within 1 year, hence the distinction between early and late latent stages. Once a year has passed, patients are not contagious by sexual transmission and are unlikely to suffer a relapse of secondary symptoms.1,3 However, late latent syphilis is characterized by periods of intermittent bacteremia that allow for seeding of the placenta and infection in about 10% of fetuses.5
Untreated, about 40% of patients will progress to the tertiary stage of syphilis, which is characterized by gummas affecting the skin and mucous membranes (FIGURE 4) and cardiovascular manifestations including arterial aneurysms and aortic insufficiency.3
Neurologic manifestations of syphilis may arise during any of the above stages, though the most characteristic manifestations tend to appear decades after the primary infection. Early neurosyphilis may present as meningitis, with or without concomitant ocular syphilis (uveitis, retinitis) and/or as otic syphilis (hearing loss, persistent tinnitus).1,5 Patients with late (tertiary) neurosyphilis tend to exhibit meningovascular symptoms similar to stroke (aphasia, hemiplegia, seizures) and/or parenchymal effects such as general paresis. Tabes dorsalis (manifestations of which include urinary and rectal incontinence, lightning pains, and ataxia) is a late-onset manifestation.1,3
Congenital syphilis can be subdivided into an early and late stage. The first stage, in which clinical findings occur within the first 2 years of life, commonly features a desquamating rash, hepatomegaly, and rhinitis. Anemia, thrombocytopenia, periostitis, and osteomyelitis also have been documented.5 Of note, two-thirds of infants are asymptomatic at birth and may not develop such clinical manifestations for 3 to 8 weeks.3 If untreated, early congenital infection may progress to late manifestations, such as Hutchinson teeth, mulberry molars, interstitial keratitis, deafness, saddle nose, saber shins, and such neurologic abnormalities as developmental delay and general paresis.3
Continue to: Prenatal screening and diagnosis...
Prenatal screening and diagnosis
Current recommendations issued by the CDC and the American College of Obstetricians and Gynecologists state that all pregnant women should be screened for syphilis infection at their first presentation to care, with repeat screening between 28 and 32 weeks of gestation and at birth, for women living in areas with a high prevalence of syphilis and/or with any of the aforementioned risk factors.3,5 Given that providers may be unfamiliar with the prevalence of syphilis in their area, and that patients may acquire or develop an infection later on in their pregnancy, researchers have begun to investigate the feasibility of universal third-trimester screening. While the cost-effectiveness of such a protocol is disputed, recent studies suggest that it may result in a substantial decrease in adverse maternal and fetal outcomes.8,9
Diagnostic tests
The traditional algorithm for the diagnosis of syphilis infection begins with a nontreponemal screening test, such as the RPR or the Venereal Disease Research Laboratory test. If positive, these screening tests are followed by a confirmatory treponemal test, such as the

The “reverse” screening algorithm begins with the FTA and, if positive, reflexes to the RPR. A reactive RPR indicates an active infection, and the patient should be treated. A negative RPR should be followed by the TP-PA to rule out a false-positive immunoglobulin G test. If the TP-PA test result is positive, the diagnosis of syphilis is confirmed (FIGURE 6). It is crucial to understand, however, that treponemal antibodies will remain positive for a patient’s lifetime, and someone who may have been treated for syphilis in the past also will screen positive. Once 2 treponemal tests are positive, physicians should take a careful history to assess prior infection risk and treatment status. A negative TP-PA excludes a diagnosis of syphilis.

Advantages of the reverse screening algorithm. Nontreponemal tests are inexpensive and easy to perform, and titers allow for identification of a baseline to evaluate response to treatment.11 However, given the fluctuation of RPR sensitivity (depending on stage of disease and a decreased ability to detect primary and latent stages of syphilis), there has been a resurgence of interest in the reverse algorithm.11 While reverse screening has been found to incur higher costs, and may result in overtreatment and increased stress due to false-positive results,12 there is evidence to suggest that this algorithm is more sensitive for primary and latent infections.8,11,13-15
Given the rise in prevalence of syphilis infections in the United States over the past decade, and therefore a higher pretest probability of syphilis in the population, we favor the reverse screening algorithm in obstetrics, particularly given the risks of adverse maternal and fetal outcomes.
Treating syphilis in pregnancy
Parenteral benzathine penicillin G is the only currently recommended medication for the treatment of syphilis in pregnancy. This drug is effective in treating maternal infection and in preventing fetal infections, as well as in treating established fetal infections.3,5 Regimens differ depending on the stage of syphilis infection (TABLE). Treatment for presumed early syphilis is recommended for women who have had sexual contact with a partner diagnosed with primary, secondary, or early latent syphilis within 3 months of their current pregnancy.5 Any patient with diagnosed syphilis who demonstrates clinical signs of neurologic involvement should undergo lumbar puncture to assess for evidence of neurosyphilis.3 CDC guidelines recommend that patients who report an allergy to penicillin undergo desensitization therapy in a controlled setting, as other antibiotics that have been investigated in the treatment of syphilis are either not appropriate due to teratogenicity or due to suboptimal fetal treatment.3,5

Syphilotherapy may lead to the Jarisch-Herxheimer reaction, which is an acute systemic reaction to inflammatory cytokines produced in response to lipopolysaccharide released by dying spirochetes.5 This reaction is characterized by fever, chills, myalgia, headache, hypotension, and worsening of cutaneous lesions. Preterm labor and delivery and fetal heart rate tracing abnormalities also have been documented in pregnant women experiencing this reaction, particularly during the second half of pregnancy.16 Prior to the start of treatment, a detailed sonographic assessment should be performed to assess the fetus for signs of early syphilis, including hepatomegaly, elevated peak systolic velocity of the middle cerebral artery (indicative of fetal anemia), polyhydramnios, placentomegaly, or hydrops.5,7
CASE Resolved
The combination of the patient’s test results—positive FTA, negative RPR, and negative TP-PA—suggest a false-positive treponemal assay. This sequence of tests excludes a diagnosis of syphilis; therefore, no treatment is necessary. Depending on the prevalence of syphilis in the patient’s geographic location, as well as her sexual history, rescreening between 28 and 32 weeks may be warranted. ●
CASE Pregnant woman with positive Treponema pallidum antibody test
A 30-year-old primigravida at 10 weeks and 4 days of gestation by her last menstrual period presents to your office for her initial prenatal visit. She expresses no concerns. You order the standard set of laboratory tests, including a sexually transmitted infection (STI) screening panel. Consistent with your institution’s use of the reverse algorithm for syphilis screening, you obtain a Treponema pallidum antibody test, which reflexes to the rapid plasma reagin (RPR) test. Three days later, you receive a notification that this patient’s T pallidum antibody result was positive, followed by negative RPR test results. The follow-up T pallidum particle agglutination (TP-PA) test also was negative. Given these findings, you consider:
- What is the correct interpretation of the patient’s sequence of test results?
- Is she infected, and does she require treatment?
Meet our perpetrator
Syphilis has plagued society since the late 15th century, although its causative agent, the spirochete T pallidum, was not recognized until 1905.1,2T pallidum bacteria are transmitted via sexual contact, as well as through vertical transmission during pregnancy or delivery. Infection with syphilis is reported in 50% to 60% of sexual partners after a single exposure to an infected individual with early syphilis, and the mean incubation period is 21 days.3T pallidum can cross the placenta and infect a fetus as early as the sixth week of gestation.3 Congenital syphilis infections occur in the neonates of 50% to 80% of women with untreated primary, secondary, or early latent syphilis infections; maternal syphilis is associated with a 21% increased risk of stillbirth, a 6% increased risk of preterm delivery, and a 9% increased risk of neonatal death.4,5 Additionally, syphilis infection is associated with a high risk of HIV infection, as well as coinfection with other STIs.1
Given the highly infective nature of T pallidum, as well as the severity of the potential consequences of infection for both mothers and babies, primary prevention, education of at-risk populations, and early recognition of clinical features of syphilis infection are of utmost importance in preventing morbidity and mortality. In this article, we review the epidemiology and extensive clinical manifestations of syphilis, as well as current screening recommendations and treatment for pregnant women.
The extent of the problem today
Although US rates of syphilis have ebbed and flowed for the past several decades, the current incidence has grown exponentially in recent years, with the number of cases reported to the Centers for Disease Control and Prevention (CDC) increasing by 71% from 2014 to 2018.6 During this time period, reported cases of primary and secondary syphilis in women more than doubled (172.7% and 165.4%, respectively) according to CDC data, accompanied by a parallel rise in reported cases of congenital syphilis in both live and stillborn infants.6 In 2018, the CDC reported a national rate of congenital syphilis of 33.1 cases per 100,000 live births, a 39.7% rise compared with data from 2017.6
Those most at risk. Risk factors for syphilis infection include age younger than 30 years, low socioeconomic status, substance abuse, HIV infection, concurrent STIs, and high-risk sexual activity (sex with multiple high-risk partners).3 Additionally, reported rates of primary and secondary syphilis infections, as well as congenital syphilis infections, are more elevated among women who identify as Black, American Indian/Alaska Native, and/or Hispanic.6 Congenital infections in the United States are correlated with a lack of prenatal care, which has been similarly linked with racial and socioeconomic disparities, as well as with untreated mental health and substance use disorders and recent immigration to the United States.5,7
Continue to: The many phases of syphilis...
The many phases of syphilis
The characteristic lesion of primary syphilis is a chancre, which is a painless, ulcerative lesion with raised borders and a clean, indurated base appearing at the site of spirochete entry (FIGURE 1). Chancres most commonly appear in the genital area, with the most frequent sites in females being within the vaginal canal or on the cervix. Primary chancres tend to heal spontaneously within 3 to 6 weeks, even without treatment, and frequently are accompanied by painless inguinal lymphadenopathy. Given that the most common chancre sites are not immediately apparent, primary infections in women often go undetected.3 In fact, it is essential for clinicians to recognize that, in our routine practice, most patients with syphilis will not be symptomatic at all, and the diagnosis will only be made by serologic screening.

Following resolution of the primary phase, the patient may enter the secondary stage of T pallidum infection. During this stage, spirochetes may disseminate throughout the bloodstream to infect all major organ systems. The principal manifestations of secondary syphilis include a diffuse maculopapular rash that begins on the trunk and proximal extremities and spreads to include the palms and soles (FIGURE 2); mucosal lesions, such as mucous patches and condyloma lata (FIGURE 3); nonscarring alopecia; periostitis; generalized lymphadenopathy; and, in some cases, hepatitis or nephritis.1,3

Secondary syphilis usually clears within 2 to 6 weeks, with the patient then entering the early latent stage of syphilis. During this period, up to 25% of patients are subject to flares of secondary syphilitic lesions but otherwise are asymptomatic.1,3,4 These recurrences tend to occur within 1 year, hence the distinction between early and late latent stages. Once a year has passed, patients are not contagious by sexual transmission and are unlikely to suffer a relapse of secondary symptoms.1,3 However, late latent syphilis is characterized by periods of intermittent bacteremia that allow for seeding of the placenta and infection in about 10% of fetuses.5
Untreated, about 40% of patients will progress to the tertiary stage of syphilis, which is characterized by gummas affecting the skin and mucous membranes (FIGURE 4) and cardiovascular manifestations including arterial aneurysms and aortic insufficiency.3
Neurologic manifestations of syphilis may arise during any of the above stages, though the most characteristic manifestations tend to appear decades after the primary infection. Early neurosyphilis may present as meningitis, with or without concomitant ocular syphilis (uveitis, retinitis) and/or as otic syphilis (hearing loss, persistent tinnitus).1,5 Patients with late (tertiary) neurosyphilis tend to exhibit meningovascular symptoms similar to stroke (aphasia, hemiplegia, seizures) and/or parenchymal effects such as general paresis. Tabes dorsalis (manifestations of which include urinary and rectal incontinence, lightning pains, and ataxia) is a late-onset manifestation.1,3
Congenital syphilis can be subdivided into an early and late stage. The first stage, in which clinical findings occur within the first 2 years of life, commonly features a desquamating rash, hepatomegaly, and rhinitis. Anemia, thrombocytopenia, periostitis, and osteomyelitis also have been documented.5 Of note, two-thirds of infants are asymptomatic at birth and may not develop such clinical manifestations for 3 to 8 weeks.3 If untreated, early congenital infection may progress to late manifestations, such as Hutchinson teeth, mulberry molars, interstitial keratitis, deafness, saddle nose, saber shins, and such neurologic abnormalities as developmental delay and general paresis.3
Continue to: Prenatal screening and diagnosis...
Prenatal screening and diagnosis
Current recommendations issued by the CDC and the American College of Obstetricians and Gynecologists state that all pregnant women should be screened for syphilis infection at their first presentation to care, with repeat screening between 28 and 32 weeks of gestation and at birth, for women living in areas with a high prevalence of syphilis and/or with any of the aforementioned risk factors.3,5 Given that providers may be unfamiliar with the prevalence of syphilis in their area, and that patients may acquire or develop an infection later on in their pregnancy, researchers have begun to investigate the feasibility of universal third-trimester screening. While the cost-effectiveness of such a protocol is disputed, recent studies suggest that it may result in a substantial decrease in adverse maternal and fetal outcomes.8,9
Diagnostic tests
The traditional algorithm for the diagnosis of syphilis infection begins with a nontreponemal screening test, such as the RPR or the Venereal Disease Research Laboratory test. If positive, these screening tests are followed by a confirmatory treponemal test, such as the

The “reverse” screening algorithm begins with the FTA and, if positive, reflexes to the RPR. A reactive RPR indicates an active infection, and the patient should be treated. A negative RPR should be followed by the TP-PA to rule out a false-positive immunoglobulin G test. If the TP-PA test result is positive, the diagnosis of syphilis is confirmed (FIGURE 6). It is crucial to understand, however, that treponemal antibodies will remain positive for a patient’s lifetime, and someone who may have been treated for syphilis in the past also will screen positive. Once 2 treponemal tests are positive, physicians should take a careful history to assess prior infection risk and treatment status. A negative TP-PA excludes a diagnosis of syphilis.

Advantages of the reverse screening algorithm. Nontreponemal tests are inexpensive and easy to perform, and titers allow for identification of a baseline to evaluate response to treatment.11 However, given the fluctuation of RPR sensitivity (depending on stage of disease and a decreased ability to detect primary and latent stages of syphilis), there has been a resurgence of interest in the reverse algorithm.11 While reverse screening has been found to incur higher costs, and may result in overtreatment and increased stress due to false-positive results,12 there is evidence to suggest that this algorithm is more sensitive for primary and latent infections.8,11,13-15
Given the rise in prevalence of syphilis infections in the United States over the past decade, and therefore a higher pretest probability of syphilis in the population, we favor the reverse screening algorithm in obstetrics, particularly given the risks of adverse maternal and fetal outcomes.
Treating syphilis in pregnancy
Parenteral benzathine penicillin G is the only currently recommended medication for the treatment of syphilis in pregnancy. This drug is effective in treating maternal infection and in preventing fetal infections, as well as in treating established fetal infections.3,5 Regimens differ depending on the stage of syphilis infection (TABLE). Treatment for presumed early syphilis is recommended for women who have had sexual contact with a partner diagnosed with primary, secondary, or early latent syphilis within 3 months of their current pregnancy.5 Any patient with diagnosed syphilis who demonstrates clinical signs of neurologic involvement should undergo lumbar puncture to assess for evidence of neurosyphilis.3 CDC guidelines recommend that patients who report an allergy to penicillin undergo desensitization therapy in a controlled setting, as other antibiotics that have been investigated in the treatment of syphilis are either not appropriate due to teratogenicity or due to suboptimal fetal treatment.3,5

Syphilotherapy may lead to the Jarisch-Herxheimer reaction, which is an acute systemic reaction to inflammatory cytokines produced in response to lipopolysaccharide released by dying spirochetes.5 This reaction is characterized by fever, chills, myalgia, headache, hypotension, and worsening of cutaneous lesions. Preterm labor and delivery and fetal heart rate tracing abnormalities also have been documented in pregnant women experiencing this reaction, particularly during the second half of pregnancy.16 Prior to the start of treatment, a detailed sonographic assessment should be performed to assess the fetus for signs of early syphilis, including hepatomegaly, elevated peak systolic velocity of the middle cerebral artery (indicative of fetal anemia), polyhydramnios, placentomegaly, or hydrops.5,7
CASE Resolved
The combination of the patient’s test results—positive FTA, negative RPR, and negative TP-PA—suggest a false-positive treponemal assay. This sequence of tests excludes a diagnosis of syphilis; therefore, no treatment is necessary. Depending on the prevalence of syphilis in the patient’s geographic location, as well as her sexual history, rescreening between 28 and 32 weeks may be warranted. ●
- Ghanem KG, Ram S, Rice PA. The modern epidemic of syphilis. N Engl J Med. 2020;382:845-854.
- Barnett R. Syphilis. Lancet. 2018;391:1471.
- Duff P. Maternal and fetal infections. In: Resnik R, Lockwood CJ, Moore T, et al. Creasy and Resnik's Maternal-Fetal Medicine: Principles and Practice. 8th ed. Philadelphia, PA: Elsevier; 2018:862-919.
- Gomez GB, Kamb ML, Newman LM, et al. Untreated maternal syphilis and adverse outcomes of pregnancy: a systematic review and meta-analysis. Bull World Health Organ. 2013;91:217-226.
- Adhikari EH. Syphilis in pregnancy. Obstet Gynecol. 2020;135:1121-1135.
- Syphilis. CDC website. https://www.cdc.gov/std/stats18/syphilis.htm. Published October 1, 2019. Accessed October 6, 2020.
- Rac MF, Revell PA, Eppes CS. Syphilis during pregnancy: a preventable threat to maternal-fetal health. Am J Obstet Gynecol. 2017;4:352-363.
- Dunseth CD, Ford BA, Krasowski MD. Traditional versus reverse syphilis algorithms: a comparison at a large academic medical center. Pract Lab Med. 2017;8:52-59.
- Hersh AR, Megli CJ, Caughey AB. Repeat screening for syphilis in the third trimester of pregnancy: a cost-effectiveness analysis. Obstet Gynecol. 2018;132:699-706.
- Albright CM, Emerson JB, Werner EF, et al. Third trimester prenatal syphilis screening: a cost-effectiveness analysis. Obstet Gynecol. 2015;126:479-485.
- Seña AC, White BL, Sparling PF. Novel Treponema pallidum serologic tests: a paradigm shift in syphilis screening for the 21st century. Clin Infect Dis. 2010;51:700-708.
- Owusu-Edusei K Jr, Peterman TA, Ballard RC. Serologic testing for syphilis in the United States: a cost-effectiveness analysis of two screening algorithms. Sex Transm Dis. 2011;38:1-7.
- Huh HJ, Chung JW, Park SY, et al. Comparison of automated treponemal and nontreponemal test algorithms as first-line syphilis screening assays. Ann Lab Med. 2016;36:23-27.
- Centers for Disease Control and Prevention. Syphilis testing algorithms using treponemal test for initial screening-four laboratories. New York City, 2005-2006. MMWR Morb Mortal Wkly Rep. 2008;57:872-875.
- Mishra S, Boily MC, Ng V, et al. The laboratory impact of changing syphilis screening from the rapid-plasma reagin to a treponemal enzyme immunoassay: a case-study from the greater Toronto area. Sex Transm Dis. 2011;38:190-196.
- Klein VR, Cox SM, Mitchell MD, et al. The Jarisch-Herzheimer reaction complicating syphilotherapy in pregnancy. Obstet Gynecol. 1990;75:375-380.
- Ghanem KG, Ram S, Rice PA. The modern epidemic of syphilis. N Engl J Med. 2020;382:845-854.
- Barnett R. Syphilis. Lancet. 2018;391:1471.
- Duff P. Maternal and fetal infections. In: Resnik R, Lockwood CJ, Moore T, et al. Creasy and Resnik's Maternal-Fetal Medicine: Principles and Practice. 8th ed. Philadelphia, PA: Elsevier; 2018:862-919.
- Gomez GB, Kamb ML, Newman LM, et al. Untreated maternal syphilis and adverse outcomes of pregnancy: a systematic review and meta-analysis. Bull World Health Organ. 2013;91:217-226.
- Adhikari EH. Syphilis in pregnancy. Obstet Gynecol. 2020;135:1121-1135.
- Syphilis. CDC website. https://www.cdc.gov/std/stats18/syphilis.htm. Published October 1, 2019. Accessed October 6, 2020.
- Rac MF, Revell PA, Eppes CS. Syphilis during pregnancy: a preventable threat to maternal-fetal health. Am J Obstet Gynecol. 2017;4:352-363.
- Dunseth CD, Ford BA, Krasowski MD. Traditional versus reverse syphilis algorithms: a comparison at a large academic medical center. Pract Lab Med. 2017;8:52-59.
- Hersh AR, Megli CJ, Caughey AB. Repeat screening for syphilis in the third trimester of pregnancy: a cost-effectiveness analysis. Obstet Gynecol. 2018;132:699-706.
- Albright CM, Emerson JB, Werner EF, et al. Third trimester prenatal syphilis screening: a cost-effectiveness analysis. Obstet Gynecol. 2015;126:479-485.
- Seña AC, White BL, Sparling PF. Novel Treponema pallidum serologic tests: a paradigm shift in syphilis screening for the 21st century. Clin Infect Dis. 2010;51:700-708.
- Owusu-Edusei K Jr, Peterman TA, Ballard RC. Serologic testing for syphilis in the United States: a cost-effectiveness analysis of two screening algorithms. Sex Transm Dis. 2011;38:1-7.
- Huh HJ, Chung JW, Park SY, et al. Comparison of automated treponemal and nontreponemal test algorithms as first-line syphilis screening assays. Ann Lab Med. 2016;36:23-27.
- Centers for Disease Control and Prevention. Syphilis testing algorithms using treponemal test for initial screening-four laboratories. New York City, 2005-2006. MMWR Morb Mortal Wkly Rep. 2008;57:872-875.
- Mishra S, Boily MC, Ng V, et al. The laboratory impact of changing syphilis screening from the rapid-plasma reagin to a treponemal enzyme immunoassay: a case-study from the greater Toronto area. Sex Transm Dis. 2011;38:190-196.
- Klein VR, Cox SM, Mitchell MD, et al. The Jarisch-Herzheimer reaction complicating syphilotherapy in pregnancy. Obstet Gynecol. 1990;75:375-380.
Unrecognized placenta accreta spectrum: Intraoperative management
CASE Concerning finding on repeat CD
A 30-year-old woman with a history of 1 prior cesarean delivery (CD) presents to labor and delivery at 38 weeks of gestation with symptoms of mild cramping. Her prenatal care was uncomplicated. The covering team made a decision to proceed with a repeat CD. A Pfannenstiel incision is made to enter the abdomen, and inspection of the lower uterine segment is concerning for a placenta accreta spectrum (PAS) (FIGURE).
What would be your next steps?

Placenta accreta spectrum describes the range of disorders of placental implantation, including placenta accreta, increta, and percreta. PAS is a significant cause of severe maternal morbidity and mortality, primarily due to massive hemorrhage at the time of delivery. The incidence of PAS continues to rise along with the CD rate. The authors of a recent meta-analysis reported a pooled prevalence rate of 1 in 588 women.1 Notably, in women with PAS, the rate of hysterectomy is 52.2%, and the transfusion-dependent hemorrhage rate is 46.9%.1
Ideally, PAS should be diagnosed or at least suspected antenatally during prenatal ultrasonography, leading to delivery planning by a multidisciplinary team.2 The presence of a multidisciplinary team—in addition to the primary obstetric and surgical teams—composed of experienced anesthesiologists, a blood bank able to respond to massive transfusion needs, critical care specialists, and interventional radiologists is associated with improved outcomes.3-5
Occasionally, a patient is found to have an advanced PAS (increta or percreta) at the time of delivery. In these situations, it is paramount that the appropriate resources be assembled as expeditiously as possible to optimize maternal outcomes. Surgical management can be challenging even for experienced pelvic surgeons, and appropriate resuscitation cannot be provided by a single anesthesiologist working alone. A cavalier attitude of proceeding with the delivery “as usual” in the face of an unexpected PAS situation can lead to disastrous consequences, including maternal death.
In this article, we review the important steps to take when faced with the unexpected situation of a PAS at the time of CD.
Continue to: Stop and collect your multidisciplinary team...
Stop and collect your multidisciplinary team
Once the diagnosis of an advanced PAS is suspected, the first step is to stop and request the presence of your institution’s multidisciplinary surgical team. This team typically includes a maternal-fetal specialist or, if not available, an experienced obstetrician, and an expert pelvic surgeon, which varies by institution (gynecologic oncologist, trauma surgeon, urologist, urogynecologist, vascular surgeon). An interventional radiology team is an additional useful resource that can assist with the control of pelvic hemorrhage using embolization techniques.
In our opinion, it is not appropriate to have a surgical backup team available only as needed at a certain distance from the hospital or even in the building. Because of the acuity and magnitude of bleeding that can occur in a short time, the most appropriate approach is to have your surgical team scrubbed and ready to assist or take over the procedure immediately if indicated.
Additional support staff also may be required. A single circulating nurse may not be sufficient, and available nursing staff may need to be called. The surgical technician scrubbed on the case may be familiar only with uncomplicated CDs and can be overwhelmed during a PAS case. Having a more experienced surgical technologist can optimize the availability of the appropriate instruments for the surgical team.
If a multidisciplinary surgical team with PAS management expertise is not available at your institution and the patient is stable, it is appropriate to consider transferring her to the nearest center that can meet the high-risk needs of this situation.6
Prepare for resuscitation
While you are calling your multidisciplinary team members, implement plans for resuscitation by notifying the anesthesiologist about the PAS findings. This will allow the gathering of needed resources that may include calling on additional anesthesiologists with experience in high-risk obstetrics, trauma, or critical care.
Placing large-bore intravenous lines or a central line to allow rapid transfusion is essential. Strongly consider inserting an arterial line for hemodynamic monitoring and intraoperative blood draws to monitor blood loss, blood gases, electrolytes, and coagulation parameters, which can guide resuscitative efforts and replacement therapies.
Simultaneously, inform the blood bank to prepare blood and blood products for possible activation of a massive transfusion protocol. It is imperative to have the products available in the operating room (OR) prior to proceeding with the surgery. Our current practice is to have 10 units of packed red blood cells and fresh frozen plasma available in the OR for all our prenatally diagnosed electively planned PAS cases.
Optimize exposure of the surgical field
Appropriate exposure of the surgical field is essential and should include exposure of the uterine fundus and the pelvic sidewalls. The uterine incision should avoid the placenta; typically it is placed at the level of the uterine fundus. Exposure of the pelvic sidewalls is needed to open the retroperitoneum and identify the ureter and the iliac vessels.
Vertical extension of the fascial incision probably will be needed to achieve appropriate exposure. Although at times this can be done without a concomitant vertical skin incision, often an inverted T incision is required. Be mindful that PAS is a life-threatening condition and that aesthetics are not a priority. After extending the fascial incision, adequate exposure can be achieved with any of the commonly used retractors or wound protectors (depending on institutional availability and surgeon preference) or by the surgical assistants using body wall retractors.
We routinely place the patient in lithotomy position. This allows us to monitor for vaginal bleeding (often a site of unrecognized massive hemorrhage) during the surgery, facilitate retrograde bladder filling, and provide a vaginal access to the pelvis. In addition, the lithotomy position allows for cystoscopy and placement of ureteral stents, which can be performed before starting the surgery to help prevent urinary tract injuries or at the end of the procedure in case one is suspected.7
Continue to: Performing the hysterectomy...
Performing the hysterectomy
A complete review of all surgical techniques for managing PAS is beyond the scope of this article. However, we briefly cover important procedural steps and offer suggestions on how to minimize the risk of bleeding.
In our experience. The areas with the highest risk of massive bleeding that can be difficult to control include the pelvic sidewall when there is lateral extension of the PAS, the vesicouterine space, and placenta previa vaginally. Be mindful of these areas at risk and have a plan in place in case of bleeding.
Uterine incision
Avoid the placenta when making the uterine incision, which is typically done in the fundal part of the uterus. Cut and tie the cord and return it to the uterine cavity. Close the incision in a single layer. Use of a surgical stapler can be used for the hysterotomy and can decrease the amount of blood loss.8
Superior attachments of the uterus
The superior attachments of the uterus include the round ligament, the utero-ovarian ligament, and the fallopian tubes. With meticulous dissection, develop an avascular space underneath these structures and, in turn, individually divide and suture ligate; this is typically achieved with minimal blood loss.
In addition, isolate the engorged veins of the broad ligament and divide them in a similar fashion.
In our experience. Use of a vessel-sealing device can facilitate division of all the former structures. Simply excise the fallopian tubes with the vessel-sealing device either at this time or after the uterus is removed.
Pelvic sidewall
Once the superior attachments of the uterus have been divided, the next step involves exposing the pelvic sidewall structures, that is, the ureter and the pelvic vessels. Expose the ureter from the pelvic brim to the level of the uterine artery. The hypogastric artery is exposed as well in this process and the pararectal space developed.
When the PAS has extended laterally, perform stepwise division of the lateral attachments of the placenta to the pelvic sidewall using a combination of electrocautery, hemoclips, and the vessel-sealing device. In laterally extended PAS cases, it often is necessary to divide the uterine artery either at its origin or at the level of the ureter to allow for the completion of the separation of the placenta from the pelvic sidewall.
In our experience. During this lateral dissection, significant bleeding may be encountered from the neovascular network that has developed in the pelvic sidewall. The bleeding may be diffuse and difficult to control with the methods described above. In this situation, we have found that placing hemostatic agents in this area and packing the sidewall with laparotomy pads can achieve hemostasis in most cases, thus allowing the surgery to proceed.
1. Stop and collect your multidisciplinary team. If required resources are not available at your institution and the patient is stable, consider transferring her to the nearest center of expertise
2. Prepare for resuscitation: Have blood products available in the operating room and optimize IV access and arterial line
3. Optimize exposure of the surgical field: place in lithotomy position, extend fascial incision, perform hysterotomy to avoid the placenta, and expose pelvic sidewall and ureters
4. Be mindful of likely sources of massive bleeding: pelvic sidewall, bladder/vesicouterine space, and/or placenta previa vaginally
5. Proceed with meticulous dissection to minimize the risk of hemorrhage, retrograde fill the bladder, be mindful of the utility of packing
6. Be prepared to move to an expeditious hysterectomy in case of massive bleeding
Continue to: Bladder dissection...
Bladder dissection
The next critical part of the surgery involves developing the vesicovaginal space to mobilize the bladder. Prior to initiating the bladder dissection, we routinely retrograde fill the bladder with 180 to 240 mL of saline mixed with methylene blue. This delineates the superior edge of the bladder and indicates the appropriate level to start the dissection. Then slowly develop the vesicouterine space using a combination of electrocautery and a vessel-sealing device until the bladder is mobilized to the level of the anterior vaginal wall. Many vascular connections are encountered at that level, and meticulous dissection and patience is required to systematically divide them all.
In our experience. This part of the surgery presents several challenges. The bladder wall may be invaded by the placenta, which will lead to an increased risk of bleeding and cystotomy during the dissection. In these cases, it might be preferable to create an intentional cystotomy to guide the dissection; at times, a limited excision of the involved bladder wall may be required. In other cases, even in the absence of bladder wall invasion, the bladder may be densely adherent to the uterine wall (usually due to a history of prior CDs), and bladder mobilization may be complicated by bleeding from the neovascular network that has developed between the placenta and bladder.
Uterine arteries and cervix
Once the placenta is separated from its lateral attachments and the bladder is mobilized, the next steps are similar to those in a standard abdominal hysterectomy. If the uterine arteries were not yet divided during the pelvic sidewall dissection, they are clamped, divided, and suture ligated at the level of the uterine isthmus. The decision whether to perform a supracervical or total hysterectomy depends on the level of cervical involvement by the placenta, surgeon preference, anatomic distortion, and bleeding from the cervix and anterior vaginal wall.
Responding to massive bleeding
Not uncommonly, and despite the best efforts to avoid it, massive bleeding may develop from the areas at risk as noted above. If the bleeding is significant and originates from multiple areas (including vaginal bleeding from placenta previa), we recommend proceeding with an expeditious hysterectomy to remove the specimen, and then reassess the pelvic field for hemostatic control and any organ damage that may have occurred.
The challenge of PAS
Surgical management of PAS is one the most challenging procedures in pelvic surgery. Successful outcomes require a multidisciplinary team approach and an experienced team dedicated to the management of this condition.9 By contrast, proceeding “as usual” in the face of an unexpected PAS situation can lead to disastrous consequences in terms of maternal morbidity and mortality. ●
- Jauniaux E, Bunce C, Gronbeck L, et al. Prevalence and main outcomes of placenta accreta spectrum: a systematic review and meta-analysis. Am J Obstet Gynecol. 2019;221:208-218.
- Society of Gynecologic Oncology, American College of Obstetricians and Gynecologists, Society for Maternal-Fetal Medicine, et al. Placenta accreta spectrum. Am J Obstet Gynecol. 2018;219:B2-B16.
- Eller AG, Bennett MA, Sharshiner M, et al. Maternal morbidity in cases of placenta accreta managed by a multidisciplinary care team compared with standard obstetric care. Obstet Gynecol. 2011;117(2 pt 1):331-337.
- Shamshirsaz AA, Fox KA, Salmanian B, et al. Maternal morbidity in patients with morbidly adherent placenta treated with and without a standardized multidisciplinary approach. Am J Obstet Gynecol. 2015;212:218.e1-9.
- Collins SL, Alemdar B, van Beekhuizen HJ, et al; International Society for Abnormally Invasive Placenta. Evidence-based guidelines for the management of abnormally invasive placenta: recommendations from the International Society for Abnormally Invasive Placenta. Am J Obstet Gynecol. 2019;220:511-526.
- Silver RM, Fox KA, Barton JR, et al. Center of excellence for placenta accreta. Am J Obstet Gynecol. 2015;212:561-568.
- Tam Tam KB, Dozier J, Martin JN Jr. Approaches to reduce urinary tract injury during management of placenta accreta, increta, and percreta: a systematic review. J Matern Fetal Neonatal Med. 2012;25:329-334.
- Belfort MA, Shamshiraz AA, Fox K. Minimizing blood loss at cesarean-hysterectomy for placenta previa percreta. Am J Obstet Gynecol. 2017;216:78.e1-78.e2.
- Shamshirsaz AA, Fox KA, Erfani H, et al. Multidisciplinary team learning in the management of the morbidly adherent placenta: outcome improvements over time. Am J Obstet Gynecol. 2017;216:612.e1-612.e5.
CASE Concerning finding on repeat CD
A 30-year-old woman with a history of 1 prior cesarean delivery (CD) presents to labor and delivery at 38 weeks of gestation with symptoms of mild cramping. Her prenatal care was uncomplicated. The covering team made a decision to proceed with a repeat CD. A Pfannenstiel incision is made to enter the abdomen, and inspection of the lower uterine segment is concerning for a placenta accreta spectrum (PAS) (FIGURE).
What would be your next steps?

Placenta accreta spectrum describes the range of disorders of placental implantation, including placenta accreta, increta, and percreta. PAS is a significant cause of severe maternal morbidity and mortality, primarily due to massive hemorrhage at the time of delivery. The incidence of PAS continues to rise along with the CD rate. The authors of a recent meta-analysis reported a pooled prevalence rate of 1 in 588 women.1 Notably, in women with PAS, the rate of hysterectomy is 52.2%, and the transfusion-dependent hemorrhage rate is 46.9%.1
Ideally, PAS should be diagnosed or at least suspected antenatally during prenatal ultrasonography, leading to delivery planning by a multidisciplinary team.2 The presence of a multidisciplinary team—in addition to the primary obstetric and surgical teams—composed of experienced anesthesiologists, a blood bank able to respond to massive transfusion needs, critical care specialists, and interventional radiologists is associated with improved outcomes.3-5
Occasionally, a patient is found to have an advanced PAS (increta or percreta) at the time of delivery. In these situations, it is paramount that the appropriate resources be assembled as expeditiously as possible to optimize maternal outcomes. Surgical management can be challenging even for experienced pelvic surgeons, and appropriate resuscitation cannot be provided by a single anesthesiologist working alone. A cavalier attitude of proceeding with the delivery “as usual” in the face of an unexpected PAS situation can lead to disastrous consequences, including maternal death.
In this article, we review the important steps to take when faced with the unexpected situation of a PAS at the time of CD.
Continue to: Stop and collect your multidisciplinary team...
Stop and collect your multidisciplinary team
Once the diagnosis of an advanced PAS is suspected, the first step is to stop and request the presence of your institution’s multidisciplinary surgical team. This team typically includes a maternal-fetal specialist or, if not available, an experienced obstetrician, and an expert pelvic surgeon, which varies by institution (gynecologic oncologist, trauma surgeon, urologist, urogynecologist, vascular surgeon). An interventional radiology team is an additional useful resource that can assist with the control of pelvic hemorrhage using embolization techniques.
In our opinion, it is not appropriate to have a surgical backup team available only as needed at a certain distance from the hospital or even in the building. Because of the acuity and magnitude of bleeding that can occur in a short time, the most appropriate approach is to have your surgical team scrubbed and ready to assist or take over the procedure immediately if indicated.
Additional support staff also may be required. A single circulating nurse may not be sufficient, and available nursing staff may need to be called. The surgical technician scrubbed on the case may be familiar only with uncomplicated CDs and can be overwhelmed during a PAS case. Having a more experienced surgical technologist can optimize the availability of the appropriate instruments for the surgical team.
If a multidisciplinary surgical team with PAS management expertise is not available at your institution and the patient is stable, it is appropriate to consider transferring her to the nearest center that can meet the high-risk needs of this situation.6

Prepare for resuscitation
While you are calling your multidisciplinary team members, implement plans for resuscitation by notifying the anesthesiologist about the PAS findings. This will allow the gathering of needed resources that may include calling on additional anesthesiologists with experience in high-risk obstetrics, trauma, or critical care.
Placing large-bore intravenous lines or a central line to allow rapid transfusion is essential. Strongly consider inserting an arterial line for hemodynamic monitoring and intraoperative blood draws to monitor blood loss, blood gases, electrolytes, and coagulation parameters, which can guide resuscitative efforts and replacement therapies.
Simultaneously, inform the blood bank to prepare blood and blood products for possible activation of a massive transfusion protocol. It is imperative to have the products available in the operating room (OR) prior to proceeding with the surgery. Our current practice is to have 10 units of packed red blood cells and fresh frozen plasma available in the OR for all our prenatally diagnosed electively planned PAS cases.
Optimize exposure of the surgical field
Appropriate exposure of the surgical field is essential and should include exposure of the uterine fundus and the pelvic sidewalls. The uterine incision should avoid the placenta; typically it is placed at the level of the uterine fundus. Exposure of the pelvic sidewalls is needed to open the retroperitoneum and identify the ureter and the iliac vessels.
Vertical extension of the fascial incision probably will be needed to achieve appropriate exposure. Although at times this can be done without a concomitant vertical skin incision, often an inverted T incision is required. Be mindful that PAS is a life-threatening condition and that aesthetics are not a priority. After extending the fascial incision, adequate exposure can be achieved with any of the commonly used retractors or wound protectors (depending on institutional availability and surgeon preference) or by the surgical assistants using body wall retractors.
We routinely place the patient in lithotomy position. This allows us to monitor for vaginal bleeding (often a site of unrecognized massive hemorrhage) during the surgery, facilitate retrograde bladder filling, and provide a vaginal access to the pelvis. In addition, the lithotomy position allows for cystoscopy and placement of ureteral stents, which can be performed before starting the surgery to help prevent urinary tract injuries or at the end of the procedure in case one is suspected.7
Continue to: Performing the hysterectomy...
Performing the hysterectomy
A complete review of all surgical techniques for managing PAS is beyond the scope of this article. However, we briefly cover important procedural steps and offer suggestions on how to minimize the risk of bleeding.
In our experience. The areas with the highest risk of massive bleeding that can be difficult to control include the pelvic sidewall when there is lateral extension of the PAS, the vesicouterine space, and placenta previa vaginally. Be mindful of these areas at risk and have a plan in place in case of bleeding.
Uterine incision
Avoid the placenta when making the uterine incision, which is typically done in the fundal part of the uterus. Cut and tie the cord and return it to the uterine cavity. Close the incision in a single layer. Use of a surgical stapler can be used for the hysterotomy and can decrease the amount of blood loss.8
Superior attachments of the uterus
The superior attachments of the uterus include the round ligament, the utero-ovarian ligament, and the fallopian tubes. With meticulous dissection, develop an avascular space underneath these structures and, in turn, individually divide and suture ligate; this is typically achieved with minimal blood loss.
In addition, isolate the engorged veins of the broad ligament and divide them in a similar fashion.
In our experience. Use of a vessel-sealing device can facilitate division of all the former structures. Simply excise the fallopian tubes with the vessel-sealing device either at this time or after the uterus is removed.
Pelvic sidewall
Once the superior attachments of the uterus have been divided, the next step involves exposing the pelvic sidewall structures, that is, the ureter and the pelvic vessels. Expose the ureter from the pelvic brim to the level of the uterine artery. The hypogastric artery is exposed as well in this process and the pararectal space developed.
When the PAS has extended laterally, perform stepwise division of the lateral attachments of the placenta to the pelvic sidewall using a combination of electrocautery, hemoclips, and the vessel-sealing device. In laterally extended PAS cases, it often is necessary to divide the uterine artery either at its origin or at the level of the ureter to allow for the completion of the separation of the placenta from the pelvic sidewall.
In our experience. During this lateral dissection, significant bleeding may be encountered from the neovascular network that has developed in the pelvic sidewall. The bleeding may be diffuse and difficult to control with the methods described above. In this situation, we have found that placing hemostatic agents in this area and packing the sidewall with laparotomy pads can achieve hemostasis in most cases, thus allowing the surgery to proceed.
1. Stop and collect your multidisciplinary team. If required resources are not available at your institution and the patient is stable, consider transferring her to the nearest center of expertise
2. Prepare for resuscitation: Have blood products available in the operating room and optimize IV access and arterial line
3. Optimize exposure of the surgical field: place in lithotomy position, extend fascial incision, perform hysterotomy to avoid the placenta, and expose pelvic sidewall and ureters
4. Be mindful of likely sources of massive bleeding: pelvic sidewall, bladder/vesicouterine space, and/or placenta previa vaginally
5. Proceed with meticulous dissection to minimize the risk of hemorrhage, retrograde fill the bladder, be mindful of the utility of packing
6. Be prepared to move to an expeditious hysterectomy in case of massive bleeding
Continue to: Bladder dissection...
Bladder dissection
The next critical part of the surgery involves developing the vesicovaginal space to mobilize the bladder. Prior to initiating the bladder dissection, we routinely retrograde fill the bladder with 180 to 240 mL of saline mixed with methylene blue. This delineates the superior edge of the bladder and indicates the appropriate level to start the dissection. Then slowly develop the vesicouterine space using a combination of electrocautery and a vessel-sealing device until the bladder is mobilized to the level of the anterior vaginal wall. Many vascular connections are encountered at that level, and meticulous dissection and patience is required to systematically divide them all.
In our experience. This part of the surgery presents several challenges. The bladder wall may be invaded by the placenta, which will lead to an increased risk of bleeding and cystotomy during the dissection. In these cases, it might be preferable to create an intentional cystotomy to guide the dissection; at times, a limited excision of the involved bladder wall may be required. In other cases, even in the absence of bladder wall invasion, the bladder may be densely adherent to the uterine wall (usually due to a history of prior CDs), and bladder mobilization may be complicated by bleeding from the neovascular network that has developed between the placenta and bladder.
Uterine arteries and cervix
Once the placenta is separated from its lateral attachments and the bladder is mobilized, the next steps are similar to those in a standard abdominal hysterectomy. If the uterine arteries were not yet divided during the pelvic sidewall dissection, they are clamped, divided, and suture ligated at the level of the uterine isthmus. The decision whether to perform a supracervical or total hysterectomy depends on the level of cervical involvement by the placenta, surgeon preference, anatomic distortion, and bleeding from the cervix and anterior vaginal wall.
Responding to massive bleeding
Not uncommonly, and despite the best efforts to avoid it, massive bleeding may develop from the areas at risk as noted above. If the bleeding is significant and originates from multiple areas (including vaginal bleeding from placenta previa), we recommend proceeding with an expeditious hysterectomy to remove the specimen, and then reassess the pelvic field for hemostatic control and any organ damage that may have occurred.
The challenge of PAS
Surgical management of PAS is one the most challenging procedures in pelvic surgery. Successful outcomes require a multidisciplinary team approach and an experienced team dedicated to the management of this condition.9 By contrast, proceeding “as usual” in the face of an unexpected PAS situation can lead to disastrous consequences in terms of maternal morbidity and mortality. ●
CASE Concerning finding on repeat CD
A 30-year-old woman with a history of 1 prior cesarean delivery (CD) presents to labor and delivery at 38 weeks of gestation with symptoms of mild cramping. Her prenatal care was uncomplicated. The covering team made a decision to proceed with a repeat CD. A Pfannenstiel incision is made to enter the abdomen, and inspection of the lower uterine segment is concerning for a placenta accreta spectrum (PAS) (FIGURE).
What would be your next steps?

Placenta accreta spectrum describes the range of disorders of placental implantation, including placenta accreta, increta, and percreta. PAS is a significant cause of severe maternal morbidity and mortality, primarily due to massive hemorrhage at the time of delivery. The incidence of PAS continues to rise along with the CD rate. The authors of a recent meta-analysis reported a pooled prevalence rate of 1 in 588 women.1 Notably, in women with PAS, the rate of hysterectomy is 52.2%, and the transfusion-dependent hemorrhage rate is 46.9%.1
Ideally, PAS should be diagnosed or at least suspected antenatally during prenatal ultrasonography, leading to delivery planning by a multidisciplinary team.2 The presence of a multidisciplinary team—in addition to the primary obstetric and surgical teams—composed of experienced anesthesiologists, a blood bank able to respond to massive transfusion needs, critical care specialists, and interventional radiologists is associated with improved outcomes.3-5
Occasionally, a patient is found to have an advanced PAS (increta or percreta) at the time of delivery. In these situations, it is paramount that the appropriate resources be assembled as expeditiously as possible to optimize maternal outcomes. Surgical management can be challenging even for experienced pelvic surgeons, and appropriate resuscitation cannot be provided by a single anesthesiologist working alone. A cavalier attitude of proceeding with the delivery “as usual” in the face of an unexpected PAS situation can lead to disastrous consequences, including maternal death.
In this article, we review the important steps to take when faced with the unexpected situation of a PAS at the time of CD.
Continue to: Stop and collect your multidisciplinary team...
Stop and collect your multidisciplinary team
Once the diagnosis of an advanced PAS is suspected, the first step is to stop and request the presence of your institution’s multidisciplinary surgical team. This team typically includes a maternal-fetal specialist or, if not available, an experienced obstetrician, and an expert pelvic surgeon, which varies by institution (gynecologic oncologist, trauma surgeon, urologist, urogynecologist, vascular surgeon). An interventional radiology team is an additional useful resource that can assist with the control of pelvic hemorrhage using embolization techniques.
In our opinion, it is not appropriate to have a surgical backup team available only as needed at a certain distance from the hospital or even in the building. Because of the acuity and magnitude of bleeding that can occur in a short time, the most appropriate approach is to have your surgical team scrubbed and ready to assist or take over the procedure immediately if indicated.
Additional support staff also may be required. A single circulating nurse may not be sufficient, and available nursing staff may need to be called. The surgical technician scrubbed on the case may be familiar only with uncomplicated CDs and can be overwhelmed during a PAS case. Having a more experienced surgical technologist can optimize the availability of the appropriate instruments for the surgical team.
If a multidisciplinary surgical team with PAS management expertise is not available at your institution and the patient is stable, it is appropriate to consider transferring her to the nearest center that can meet the high-risk needs of this situation.6

Prepare for resuscitation
While you are calling your multidisciplinary team members, implement plans for resuscitation by notifying the anesthesiologist about the PAS findings. This will allow the gathering of needed resources that may include calling on additional anesthesiologists with experience in high-risk obstetrics, trauma, or critical care.
Placing large-bore intravenous lines or a central line to allow rapid transfusion is essential. Strongly consider inserting an arterial line for hemodynamic monitoring and intraoperative blood draws to monitor blood loss, blood gases, electrolytes, and coagulation parameters, which can guide resuscitative efforts and replacement therapies.
Simultaneously, inform the blood bank to prepare blood and blood products for possible activation of a massive transfusion protocol. It is imperative to have the products available in the operating room (OR) prior to proceeding with the surgery. Our current practice is to have 10 units of packed red blood cells and fresh frozen plasma available in the OR for all our prenatally diagnosed electively planned PAS cases.
Optimize exposure of the surgical field
Appropriate exposure of the surgical field is essential and should include exposure of the uterine fundus and the pelvic sidewalls. The uterine incision should avoid the placenta; typically it is placed at the level of the uterine fundus. Exposure of the pelvic sidewalls is needed to open the retroperitoneum and identify the ureter and the iliac vessels.
Vertical extension of the fascial incision probably will be needed to achieve appropriate exposure. Although at times this can be done without a concomitant vertical skin incision, often an inverted T incision is required. Be mindful that PAS is a life-threatening condition and that aesthetics are not a priority. After extending the fascial incision, adequate exposure can be achieved with any of the commonly used retractors or wound protectors (depending on institutional availability and surgeon preference) or by the surgical assistants using body wall retractors.
We routinely place the patient in lithotomy position. This allows us to monitor for vaginal bleeding (often a site of unrecognized massive hemorrhage) during the surgery, facilitate retrograde bladder filling, and provide a vaginal access to the pelvis. In addition, the lithotomy position allows for cystoscopy and placement of ureteral stents, which can be performed before starting the surgery to help prevent urinary tract injuries or at the end of the procedure in case one is suspected.7
Continue to: Performing the hysterectomy...
Performing the hysterectomy
A complete review of all surgical techniques for managing PAS is beyond the scope of this article. However, we briefly cover important procedural steps and offer suggestions on how to minimize the risk of bleeding.
In our experience. The areas with the highest risk of massive bleeding that can be difficult to control include the pelvic sidewall when there is lateral extension of the PAS, the vesicouterine space, and placenta previa vaginally. Be mindful of these areas at risk and have a plan in place in case of bleeding.
Uterine incision
Avoid the placenta when making the uterine incision, which is typically done in the fundal part of the uterus. Cut and tie the cord and return it to the uterine cavity. Close the incision in a single layer. Use of a surgical stapler can be used for the hysterotomy and can decrease the amount of blood loss.8
Superior attachments of the uterus
The superior attachments of the uterus include the round ligament, the utero-ovarian ligament, and the fallopian tubes. With meticulous dissection, develop an avascular space underneath these structures and, in turn, individually divide and suture ligate; this is typically achieved with minimal blood loss.
In addition, isolate the engorged veins of the broad ligament and divide them in a similar fashion.
In our experience. Use of a vessel-sealing device can facilitate division of all the former structures. Simply excise the fallopian tubes with the vessel-sealing device either at this time or after the uterus is removed.
Pelvic sidewall
Once the superior attachments of the uterus have been divided, the next step involves exposing the pelvic sidewall structures, that is, the ureter and the pelvic vessels. Expose the ureter from the pelvic brim to the level of the uterine artery. The hypogastric artery is exposed as well in this process and the pararectal space developed.
When the PAS has extended laterally, perform stepwise division of the lateral attachments of the placenta to the pelvic sidewall using a combination of electrocautery, hemoclips, and the vessel-sealing device. In laterally extended PAS cases, it often is necessary to divide the uterine artery either at its origin or at the level of the ureter to allow for the completion of the separation of the placenta from the pelvic sidewall.
In our experience. During this lateral dissection, significant bleeding may be encountered from the neovascular network that has developed in the pelvic sidewall. The bleeding may be diffuse and difficult to control with the methods described above. In this situation, we have found that placing hemostatic agents in this area and packing the sidewall with laparotomy pads can achieve hemostasis in most cases, thus allowing the surgery to proceed.
1. Stop and collect your multidisciplinary team. If required resources are not available at your institution and the patient is stable, consider transferring her to the nearest center of expertise
2. Prepare for resuscitation: Have blood products available in the operating room and optimize IV access and arterial line
3. Optimize exposure of the surgical field: place in lithotomy position, extend fascial incision, perform hysterotomy to avoid the placenta, and expose pelvic sidewall and ureters
4. Be mindful of likely sources of massive bleeding: pelvic sidewall, bladder/vesicouterine space, and/or placenta previa vaginally
5. Proceed with meticulous dissection to minimize the risk of hemorrhage, retrograde fill the bladder, be mindful of the utility of packing
6. Be prepared to move to an expeditious hysterectomy in case of massive bleeding
Continue to: Bladder dissection...
Bladder dissection
The next critical part of the surgery involves developing the vesicovaginal space to mobilize the bladder. Prior to initiating the bladder dissection, we routinely retrograde fill the bladder with 180 to 240 mL of saline mixed with methylene blue. This delineates the superior edge of the bladder and indicates the appropriate level to start the dissection. Then slowly develop the vesicouterine space using a combination of electrocautery and a vessel-sealing device until the bladder is mobilized to the level of the anterior vaginal wall. Many vascular connections are encountered at that level, and meticulous dissection and patience is required to systematically divide them all.
In our experience. This part of the surgery presents several challenges. The bladder wall may be invaded by the placenta, which will lead to an increased risk of bleeding and cystotomy during the dissection. In these cases, it might be preferable to create an intentional cystotomy to guide the dissection; at times, a limited excision of the involved bladder wall may be required. In other cases, even in the absence of bladder wall invasion, the bladder may be densely adherent to the uterine wall (usually due to a history of prior CDs), and bladder mobilization may be complicated by bleeding from the neovascular network that has developed between the placenta and bladder.
Uterine arteries and cervix
Once the placenta is separated from its lateral attachments and the bladder is mobilized, the next steps are similar to those in a standard abdominal hysterectomy. If the uterine arteries were not yet divided during the pelvic sidewall dissection, they are clamped, divided, and suture ligated at the level of the uterine isthmus. The decision whether to perform a supracervical or total hysterectomy depends on the level of cervical involvement by the placenta, surgeon preference, anatomic distortion, and bleeding from the cervix and anterior vaginal wall.
Responding to massive bleeding
Not uncommonly, and despite the best efforts to avoid it, massive bleeding may develop from the areas at risk as noted above. If the bleeding is significant and originates from multiple areas (including vaginal bleeding from placenta previa), we recommend proceeding with an expeditious hysterectomy to remove the specimen, and then reassess the pelvic field for hemostatic control and any organ damage that may have occurred.
The challenge of PAS
Surgical management of PAS is one the most challenging procedures in pelvic surgery. Successful outcomes require a multidisciplinary team approach and an experienced team dedicated to the management of this condition.9 By contrast, proceeding “as usual” in the face of an unexpected PAS situation can lead to disastrous consequences in terms of maternal morbidity and mortality. ●
- Jauniaux E, Bunce C, Gronbeck L, et al. Prevalence and main outcomes of placenta accreta spectrum: a systematic review and meta-analysis. Am J Obstet Gynecol. 2019;221:208-218.
- Society of Gynecologic Oncology, American College of Obstetricians and Gynecologists, Society for Maternal-Fetal Medicine, et al. Placenta accreta spectrum. Am J Obstet Gynecol. 2018;219:B2-B16.
- Eller AG, Bennett MA, Sharshiner M, et al. Maternal morbidity in cases of placenta accreta managed by a multidisciplinary care team compared with standard obstetric care. Obstet Gynecol. 2011;117(2 pt 1):331-337.
- Shamshirsaz AA, Fox KA, Salmanian B, et al. Maternal morbidity in patients with morbidly adherent placenta treated with and without a standardized multidisciplinary approach. Am J Obstet Gynecol. 2015;212:218.e1-9.
- Collins SL, Alemdar B, van Beekhuizen HJ, et al; International Society for Abnormally Invasive Placenta. Evidence-based guidelines for the management of abnormally invasive placenta: recommendations from the International Society for Abnormally Invasive Placenta. Am J Obstet Gynecol. 2019;220:511-526.
- Silver RM, Fox KA, Barton JR, et al. Center of excellence for placenta accreta. Am J Obstet Gynecol. 2015;212:561-568.
- Tam Tam KB, Dozier J, Martin JN Jr. Approaches to reduce urinary tract injury during management of placenta accreta, increta, and percreta: a systematic review. J Matern Fetal Neonatal Med. 2012;25:329-334.
- Belfort MA, Shamshiraz AA, Fox K. Minimizing blood loss at cesarean-hysterectomy for placenta previa percreta. Am J Obstet Gynecol. 2017;216:78.e1-78.e2.
- Shamshirsaz AA, Fox KA, Erfani H, et al. Multidisciplinary team learning in the management of the morbidly adherent placenta: outcome improvements over time. Am J Obstet Gynecol. 2017;216:612.e1-612.e5.
- Jauniaux E, Bunce C, Gronbeck L, et al. Prevalence and main outcomes of placenta accreta spectrum: a systematic review and meta-analysis. Am J Obstet Gynecol. 2019;221:208-218.
- Society of Gynecologic Oncology, American College of Obstetricians and Gynecologists, Society for Maternal-Fetal Medicine, et al. Placenta accreta spectrum. Am J Obstet Gynecol. 2018;219:B2-B16.
- Eller AG, Bennett MA, Sharshiner M, et al. Maternal morbidity in cases of placenta accreta managed by a multidisciplinary care team compared with standard obstetric care. Obstet Gynecol. 2011;117(2 pt 1):331-337.
- Shamshirsaz AA, Fox KA, Salmanian B, et al. Maternal morbidity in patients with morbidly adherent placenta treated with and without a standardized multidisciplinary approach. Am J Obstet Gynecol. 2015;212:218.e1-9.
- Collins SL, Alemdar B, van Beekhuizen HJ, et al; International Society for Abnormally Invasive Placenta. Evidence-based guidelines for the management of abnormally invasive placenta: recommendations from the International Society for Abnormally Invasive Placenta. Am J Obstet Gynecol. 2019;220:511-526.
- Silver RM, Fox KA, Barton JR, et al. Center of excellence for placenta accreta. Am J Obstet Gynecol. 2015;212:561-568.
- Tam Tam KB, Dozier J, Martin JN Jr. Approaches to reduce urinary tract injury during management of placenta accreta, increta, and percreta: a systematic review. J Matern Fetal Neonatal Med. 2012;25:329-334.
- Belfort MA, Shamshiraz AA, Fox K. Minimizing blood loss at cesarean-hysterectomy for placenta previa percreta. Am J Obstet Gynecol. 2017;216:78.e1-78.e2.
- Shamshirsaz AA, Fox KA, Erfani H, et al. Multidisciplinary team learning in the management of the morbidly adherent placenta: outcome improvements over time. Am J Obstet Gynecol. 2017;216:612.e1-612.e5.
Apps for applying to ObGyn residency programs in the era of virtual interviews
The coronavirus disease 2019 (COVID-19) pandemic has upended the traditional 2020–2021 application season for ObGyn residency programs. In May 2020, the 2 national ObGyn education organizations, the Association of Professors of Gynecology and Obstetrics (APGO) and Council on Resident Education in ObGyn (CREOG), issued guidelines to ensure a fair and equitable application process.1 These guidelines are consistent with recommendations from the Association of American Medical Colleges (AAMC) and the Coalition for Physician Accountability. Important recommendations include:
- limiting away rotations
- being flexible in the number of specialty-specific letters of recommendation required
- encouraging residency programs to develop alternate means of conveying information about their curriculum.
In addition, these statements provide timing on when programs should release interview offers and when to begin interviews. Finally, programs are required to commit to online interviews and virtual visits for all applicants, including local students, rather than in-person interviews.
Here, we focus on identifying apps that students can use to help them with the application process—apps for the nuts and bolts of applying and interviewing and apps to learn more about individual programs.
Students must use the Electronic Residency Application Service (ERAS) platform from AAMC to enter their information and register with the National Resident Matching Program (NRMP). Students also must use the ERAS to submit their applications to their selected residency programs. The ERAS platform does not include an app to aid in the completion or submission of an application. The NRMP has developed the MATCH PRISM app, but this does not allow students to register for the match or submit their rank list. To learn about how to schedule interviews, residency programs may use one of the following sources: ERAS, Interview Broker, or Thalamus. Moreover, APGO/CREOG has partnered with Thalamus for the upcoming application cycle, which provides residency programs and applicants tools for application management, interview scheduling, and itinerary building. Thalamus offers a free app.
This year offers some unique challenges. The application process for ObGyn residencies is likely to be more competitive, and students face the added stress of having to navigate the interview season:
- without away rotations (audition interviews)
- without in-person visits of the city/hospital/program or social events before or after interview day
- with an all-virtual interview day.
Continue to: To find information on individual residency programs...
To find information on individual residency programs, the APGO website lists the FREIDA and APGO Residency Directories, which are not apps. Students are also aware of the Doximity Residency Navigator, which does include an app. The NRMP MATCH PRISM app is another resource, as it provides students with a directory of residency programs and information about each program.
The American College of Obstetricians and Gynecologists (ACOG) recognizes that residency program websites and social media will be crucial in helping applicants learn about individual programs, faculty, and residents. As such, ACOG hosted a Virtual Residency Showcase in September 2020 in which programs posted content on Instagram and Twitter using the hashtag #ACOG-ResWeek20.2 Similarly, APGO and CREOG produced a report containing a social media directory, which lists individual residency programs and whether or not they have a social media handle/account.3 In a recent webinar,4 Drs. Sarah Santiago and Elizabeth Southworth noted that the number of residency programs that have an Instagram account more than doubled (from 60 to 128) between May and September 2020.

We present 2 tables describing the important features and caveats of apps available to students to assist them with residency applications this year—TABLE 1 summarizes apps to aid with applications and interviews; TABLE 2 lists apps designed for students to learn more about individual residency programs. We wish all of this year’s students every success in their search for the right program. ●
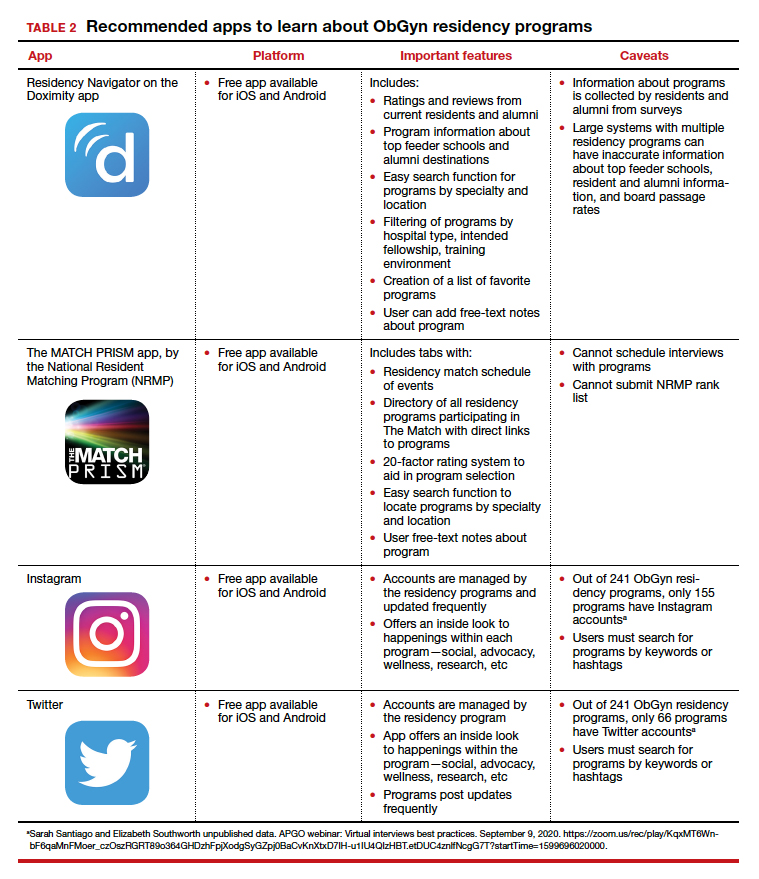
- Association of Professors of Gynecology and Obstetrics, Council on Resident Education in ObGyn. Updated APGO and CREOG Residency Application Response to COVID-19. https://www.apgo.org/wp-content/uploads/2020/05 /Updated-APGO-CREOG-Residency-Response-to -COVID-19-.pdf. Accessed October 27, 2020.
- https://www.acog.org/education-and-events/webinars /virtual-residency-showcase. Accessed October 4, 2020.
- Social media directory-ObGyn. https://docs.google.com /spreadsheets/d/e/2PACX-1vQ6boyn7FWV9tEhfQp1o3 XJgNIPNBQ3qCYf4IpV-rOPcd212J-HNR84p0r85nXrAz MvOmcNlgjywDP/pubhtml?gid=1472916499&single =true. Accessed October 27, 2020.
- APGO webinar: Virtual interviews best practices. September 9, 2020. https://zoom.us/rec/play/KqxMT6Wnb F6qaMnFMoer_czOszRGRT89o364GHDzhFpjXodgSyGZpj 0BaCvKnXtxD7IH-u1IU4QIzHBT.etDUC4znlfNcgG7T?start Time=1599696020000. Accessed October 4, 2020.
The coronavirus disease 2019 (COVID-19) pandemic has upended the traditional 2020–2021 application season for ObGyn residency programs. In May 2020, the 2 national ObGyn education organizations, the Association of Professors of Gynecology and Obstetrics (APGO) and Council on Resident Education in ObGyn (CREOG), issued guidelines to ensure a fair and equitable application process.1 These guidelines are consistent with recommendations from the Association of American Medical Colleges (AAMC) and the Coalition for Physician Accountability. Important recommendations include:
- limiting away rotations
- being flexible in the number of specialty-specific letters of recommendation required
- encouraging residency programs to develop alternate means of conveying information about their curriculum.
In addition, these statements provide timing on when programs should release interview offers and when to begin interviews. Finally, programs are required to commit to online interviews and virtual visits for all applicants, including local students, rather than in-person interviews.
Here, we focus on identifying apps that students can use to help them with the application process—apps for the nuts and bolts of applying and interviewing and apps to learn more about individual programs.
Students must use the Electronic Residency Application Service (ERAS) platform from AAMC to enter their information and register with the National Resident Matching Program (NRMP). Students also must use the ERAS to submit their applications to their selected residency programs. The ERAS platform does not include an app to aid in the completion or submission of an application. The NRMP has developed the MATCH PRISM app, but this does not allow students to register for the match or submit their rank list. To learn about how to schedule interviews, residency programs may use one of the following sources: ERAS, Interview Broker, or Thalamus. Moreover, APGO/CREOG has partnered with Thalamus for the upcoming application cycle, which provides residency programs and applicants tools for application management, interview scheduling, and itinerary building. Thalamus offers a free app.
This year offers some unique challenges. The application process for ObGyn residencies is likely to be more competitive, and students face the added stress of having to navigate the interview season:
- without away rotations (audition interviews)
- without in-person visits of the city/hospital/program or social events before or after interview day
- with an all-virtual interview day.
Continue to: To find information on individual residency programs...
To find information on individual residency programs, the APGO website lists the FREIDA and APGO Residency Directories, which are not apps. Students are also aware of the Doximity Residency Navigator, which does include an app. The NRMP MATCH PRISM app is another resource, as it provides students with a directory of residency programs and information about each program.
The American College of Obstetricians and Gynecologists (ACOG) recognizes that residency program websites and social media will be crucial in helping applicants learn about individual programs, faculty, and residents. As such, ACOG hosted a Virtual Residency Showcase in September 2020 in which programs posted content on Instagram and Twitter using the hashtag #ACOG-ResWeek20.2 Similarly, APGO and CREOG produced a report containing a social media directory, which lists individual residency programs and whether or not they have a social media handle/account.3 In a recent webinar,4 Drs. Sarah Santiago and Elizabeth Southworth noted that the number of residency programs that have an Instagram account more than doubled (from 60 to 128) between May and September 2020.

We present 2 tables describing the important features and caveats of apps available to students to assist them with residency applications this year—TABLE 1 summarizes apps to aid with applications and interviews; TABLE 2 lists apps designed for students to learn more about individual residency programs. We wish all of this year’s students every success in their search for the right program. ●

The coronavirus disease 2019 (COVID-19) pandemic has upended the traditional 2020–2021 application season for ObGyn residency programs. In May 2020, the 2 national ObGyn education organizations, the Association of Professors of Gynecology and Obstetrics (APGO) and Council on Resident Education in ObGyn (CREOG), issued guidelines to ensure a fair and equitable application process.1 These guidelines are consistent with recommendations from the Association of American Medical Colleges (AAMC) and the Coalition for Physician Accountability. Important recommendations include:
- limiting away rotations
- being flexible in the number of specialty-specific letters of recommendation required
- encouraging residency programs to develop alternate means of conveying information about their curriculum.
In addition, these statements provide timing on when programs should release interview offers and when to begin interviews. Finally, programs are required to commit to online interviews and virtual visits for all applicants, including local students, rather than in-person interviews.
Here, we focus on identifying apps that students can use to help them with the application process—apps for the nuts and bolts of applying and interviewing and apps to learn more about individual programs.
Students must use the Electronic Residency Application Service (ERAS) platform from AAMC to enter their information and register with the National Resident Matching Program (NRMP). Students also must use the ERAS to submit their applications to their selected residency programs. The ERAS platform does not include an app to aid in the completion or submission of an application. The NRMP has developed the MATCH PRISM app, but this does not allow students to register for the match or submit their rank list. To learn about how to schedule interviews, residency programs may use one of the following sources: ERAS, Interview Broker, or Thalamus. Moreover, APGO/CREOG has partnered with Thalamus for the upcoming application cycle, which provides residency programs and applicants tools for application management, interview scheduling, and itinerary building. Thalamus offers a free app.
This year offers some unique challenges. The application process for ObGyn residencies is likely to be more competitive, and students face the added stress of having to navigate the interview season:
- without away rotations (audition interviews)
- without in-person visits of the city/hospital/program or social events before or after interview day
- with an all-virtual interview day.
Continue to: To find information on individual residency programs...
To find information on individual residency programs, the APGO website lists the FREIDA and APGO Residency Directories, which are not apps. Students are also aware of the Doximity Residency Navigator, which does include an app. The NRMP MATCH PRISM app is another resource, as it provides students with a directory of residency programs and information about each program.
The American College of Obstetricians and Gynecologists (ACOG) recognizes that residency program websites and social media will be crucial in helping applicants learn about individual programs, faculty, and residents. As such, ACOG hosted a Virtual Residency Showcase in September 2020 in which programs posted content on Instagram and Twitter using the hashtag #ACOG-ResWeek20.2 Similarly, APGO and CREOG produced a report containing a social media directory, which lists individual residency programs and whether or not they have a social media handle/account.3 In a recent webinar,4 Drs. Sarah Santiago and Elizabeth Southworth noted that the number of residency programs that have an Instagram account more than doubled (from 60 to 128) between May and September 2020.

We present 2 tables describing the important features and caveats of apps available to students to assist them with residency applications this year—TABLE 1 summarizes apps to aid with applications and interviews; TABLE 2 lists apps designed for students to learn more about individual residency programs. We wish all of this year’s students every success in their search for the right program. ●

- Association of Professors of Gynecology and Obstetrics, Council on Resident Education in ObGyn. Updated APGO and CREOG Residency Application Response to COVID-19. https://www.apgo.org/wp-content/uploads/2020/05 /Updated-APGO-CREOG-Residency-Response-to -COVID-19-.pdf. Accessed October 27, 2020.
- https://www.acog.org/education-and-events/webinars /virtual-residency-showcase. Accessed October 4, 2020.
- Social media directory-ObGyn. https://docs.google.com /spreadsheets/d/e/2PACX-1vQ6boyn7FWV9tEhfQp1o3 XJgNIPNBQ3qCYf4IpV-rOPcd212J-HNR84p0r85nXrAz MvOmcNlgjywDP/pubhtml?gid=1472916499&single =true. Accessed October 27, 2020.
- APGO webinar: Virtual interviews best practices. September 9, 2020. https://zoom.us/rec/play/KqxMT6Wnb F6qaMnFMoer_czOszRGRT89o364GHDzhFpjXodgSyGZpj 0BaCvKnXtxD7IH-u1IU4QIzHBT.etDUC4znlfNcgG7T?start Time=1599696020000. Accessed October 4, 2020.
- Association of Professors of Gynecology and Obstetrics, Council on Resident Education in ObGyn. Updated APGO and CREOG Residency Application Response to COVID-19. https://www.apgo.org/wp-content/uploads/2020/05 /Updated-APGO-CREOG-Residency-Response-to -COVID-19-.pdf. Accessed October 27, 2020.
- https://www.acog.org/education-and-events/webinars /virtual-residency-showcase. Accessed October 4, 2020.
- Social media directory-ObGyn. https://docs.google.com /spreadsheets/d/e/2PACX-1vQ6boyn7FWV9tEhfQp1o3 XJgNIPNBQ3qCYf4IpV-rOPcd212J-HNR84p0r85nXrAz MvOmcNlgjywDP/pubhtml?gid=1472916499&single =true. Accessed October 27, 2020.
- APGO webinar: Virtual interviews best practices. September 9, 2020. https://zoom.us/rec/play/KqxMT6Wnb F6qaMnFMoer_czOszRGRT89o364GHDzhFpjXodgSyGZpj 0BaCvKnXtxD7IH-u1IU4QIzHBT.etDUC4znlfNcgG7T?start Time=1599696020000. Accessed October 4, 2020.
'Cardio-obstetrics' tied to better outcome in pregnancy with CVD
A multidisciplinary cardio-obstetrics team-based care model may help improve cardiovascular care for pregnant women with cardiovascular disease (CVD), according to a recent study.
“We sought to describe clinical characteristics, maternal and fetal outcomes, and cardiovascular readmissions in a cohort of pregnant women with underlying CVD followed by a cardio-obstetrics team,” wrote Ella Magun, MD, of Columbia University, New York, and coauthors. Their report is in the Journal of the American College of Cardiology.
The researchers reported the outcomes of a retrospective cohort analysis involving 306 pregnant women with CVD, who were treated at a quaternary care hospital in New York City.
They defined cardio-obstetrics as a team-based collaborative approach to maternal care that includes maternal fetal medicine, cardiology, anesthesiology, neonatology, nursing, social work, and pharmacy.
More than half of the women in the cohort (53%) were Hispanic and Latino, and 74% were receiving Medicaid, suggesting low socioeconomic status. Key outcomes of interest were cardiovascular readmissions at 30 days, 90 days, and 1 year. Secondary endpoints included maternal death, need for a left ventricular assist device or heart transplantation, and fetal demise.
The most frequently observed forms of CVD were arrhythmias (29%), cardiomyopathy (24%), congenital heart disease (24%), valvular disease (16%), and coronary artery disease (4%). The median Cardiac Disease in Pregnancy (CARPREG II) score was 3, and 43% of women had a CARPREG II score of 4 or higher.
After a median follow-up of 2.6 years, the 30-day and 90-day cardiovascular readmission rates were 1.9% and 4.6%, which was lower than the national 30-day postpartum rate of readmission (3.6%). One maternal death (0.3%) occurred within a year of delivery (woman with Eisenmenger syndrome).
“Despite high CARPREG II scores in this patient population, we found low rates of maternal and fetal complications with a low rate of 30- and 90-day readmissions following delivery,” the researchers wrote.
Experts weigh in
“We’re seeing widely increasing interest in the implementation of cardio-obstetrics models for multidisciplinary collaborative care and initial studies suggest these team-based models improve pregnancy and postpartum outcomes for women with cardiac disease,” said Lisa M. Hollier, MD, past president of the American College of Obstetricians and Gynecologists and professor at Baylor College of Medicine in Houston.
Dr. Magun and colleagues acknowledged that a key limitation of the present study was the retrospective, single-center design.
“With program expansions over the next 2-3 years, I expect to see an increasing number of prospective studies with larger sample sizes evaluating the impact of cardio-obstetrics teams on maternal morbidity and mortality,” Dr. Hollier said.
“These findings suggest that our cardio-obstetrics program may help provide improved cardiovascular care to an otherwise underserved population,” the authors concluded.
In an editorial accompanying the reports, Pamela Ouyang, MBBS, and Garima Sharma, MD, wrote that, although this study wasn’t designed to assess the benefit of cardio-obstetric teams relative to standard of care, its implementation of a multidisciplinary team-based care model showed excellent long-term outcomes.
The importance of coordinated postpartum follow-up with both cardiologists and obstetricians is becoming increasingly recognized, especially for women with poor pregnancy outcomes and with CVD that arises during pregnancy, such as pregnancy-associated spontaneous coronary artery dissection and peripartum cardiomyopathy, wrote Dr. Ouyang and Dr. Sharma, both with Johns Hopkins University in Baltimore.
“I’m very excited about the growing recognition of the importance of cardio-obstetrics and the emergence of many of these models of care at various institutions,” Melinda Davis, MD, of the University of Michigan in Ann Arbor, said in an interview.
“Over the next few years, I expect we will see several studies that show the benefits of the cardio-obstetrics model of care,” she explained. “Multicenter collaboration will be very important for learning about the optimal way to manage high-risk conditions during pregnancy.”
No funding sources were reported. The authors of this paper disclosed no conflicts of interest.
SOURCE: Magun E et al. JACC. 2020 Nov 3. doi: 10.1016/j.jacc.2020.08.071.
A multidisciplinary cardio-obstetrics team-based care model may help improve cardiovascular care for pregnant women with cardiovascular disease (CVD), according to a recent study.
“We sought to describe clinical characteristics, maternal and fetal outcomes, and cardiovascular readmissions in a cohort of pregnant women with underlying CVD followed by a cardio-obstetrics team,” wrote Ella Magun, MD, of Columbia University, New York, and coauthors. Their report is in the Journal of the American College of Cardiology.
The researchers reported the outcomes of a retrospective cohort analysis involving 306 pregnant women with CVD, who were treated at a quaternary care hospital in New York City.
They defined cardio-obstetrics as a team-based collaborative approach to maternal care that includes maternal fetal medicine, cardiology, anesthesiology, neonatology, nursing, social work, and pharmacy.
More than half of the women in the cohort (53%) were Hispanic and Latino, and 74% were receiving Medicaid, suggesting low socioeconomic status. Key outcomes of interest were cardiovascular readmissions at 30 days, 90 days, and 1 year. Secondary endpoints included maternal death, need for a left ventricular assist device or heart transplantation, and fetal demise.
The most frequently observed forms of CVD were arrhythmias (29%), cardiomyopathy (24%), congenital heart disease (24%), valvular disease (16%), and coronary artery disease (4%). The median Cardiac Disease in Pregnancy (CARPREG II) score was 3, and 43% of women had a CARPREG II score of 4 or higher.
After a median follow-up of 2.6 years, the 30-day and 90-day cardiovascular readmission rates were 1.9% and 4.6%, which was lower than the national 30-day postpartum rate of readmission (3.6%). One maternal death (0.3%) occurred within a year of delivery (woman with Eisenmenger syndrome).
“Despite high CARPREG II scores in this patient population, we found low rates of maternal and fetal complications with a low rate of 30- and 90-day readmissions following delivery,” the researchers wrote.
Experts weigh in
“We’re seeing widely increasing interest in the implementation of cardio-obstetrics models for multidisciplinary collaborative care and initial studies suggest these team-based models improve pregnancy and postpartum outcomes for women with cardiac disease,” said Lisa M. Hollier, MD, past president of the American College of Obstetricians and Gynecologists and professor at Baylor College of Medicine in Houston.
Dr. Magun and colleagues acknowledged that a key limitation of the present study was the retrospective, single-center design.
“With program expansions over the next 2-3 years, I expect to see an increasing number of prospective studies with larger sample sizes evaluating the impact of cardio-obstetrics teams on maternal morbidity and mortality,” Dr. Hollier said.
“These findings suggest that our cardio-obstetrics program may help provide improved cardiovascular care to an otherwise underserved population,” the authors concluded.
In an editorial accompanying the reports, Pamela Ouyang, MBBS, and Garima Sharma, MD, wrote that, although this study wasn’t designed to assess the benefit of cardio-obstetric teams relative to standard of care, its implementation of a multidisciplinary team-based care model showed excellent long-term outcomes.
The importance of coordinated postpartum follow-up with both cardiologists and obstetricians is becoming increasingly recognized, especially for women with poor pregnancy outcomes and with CVD that arises during pregnancy, such as pregnancy-associated spontaneous coronary artery dissection and peripartum cardiomyopathy, wrote Dr. Ouyang and Dr. Sharma, both with Johns Hopkins University in Baltimore.
“I’m very excited about the growing recognition of the importance of cardio-obstetrics and the emergence of many of these models of care at various institutions,” Melinda Davis, MD, of the University of Michigan in Ann Arbor, said in an interview.
“Over the next few years, I expect we will see several studies that show the benefits of the cardio-obstetrics model of care,” she explained. “Multicenter collaboration will be very important for learning about the optimal way to manage high-risk conditions during pregnancy.”
No funding sources were reported. The authors of this paper disclosed no conflicts of interest.
SOURCE: Magun E et al. JACC. 2020 Nov 3. doi: 10.1016/j.jacc.2020.08.071.
A multidisciplinary cardio-obstetrics team-based care model may help improve cardiovascular care for pregnant women with cardiovascular disease (CVD), according to a recent study.
“We sought to describe clinical characteristics, maternal and fetal outcomes, and cardiovascular readmissions in a cohort of pregnant women with underlying CVD followed by a cardio-obstetrics team,” wrote Ella Magun, MD, of Columbia University, New York, and coauthors. Their report is in the Journal of the American College of Cardiology.
The researchers reported the outcomes of a retrospective cohort analysis involving 306 pregnant women with CVD, who were treated at a quaternary care hospital in New York City.
They defined cardio-obstetrics as a team-based collaborative approach to maternal care that includes maternal fetal medicine, cardiology, anesthesiology, neonatology, nursing, social work, and pharmacy.
More than half of the women in the cohort (53%) were Hispanic and Latino, and 74% were receiving Medicaid, suggesting low socioeconomic status. Key outcomes of interest were cardiovascular readmissions at 30 days, 90 days, and 1 year. Secondary endpoints included maternal death, need for a left ventricular assist device or heart transplantation, and fetal demise.
The most frequently observed forms of CVD were arrhythmias (29%), cardiomyopathy (24%), congenital heart disease (24%), valvular disease (16%), and coronary artery disease (4%). The median Cardiac Disease in Pregnancy (CARPREG II) score was 3, and 43% of women had a CARPREG II score of 4 or higher.
After a median follow-up of 2.6 years, the 30-day and 90-day cardiovascular readmission rates were 1.9% and 4.6%, which was lower than the national 30-day postpartum rate of readmission (3.6%). One maternal death (0.3%) occurred within a year of delivery (woman with Eisenmenger syndrome).
“Despite high CARPREG II scores in this patient population, we found low rates of maternal and fetal complications with a low rate of 30- and 90-day readmissions following delivery,” the researchers wrote.
Experts weigh in
“We’re seeing widely increasing interest in the implementation of cardio-obstetrics models for multidisciplinary collaborative care and initial studies suggest these team-based models improve pregnancy and postpartum outcomes for women with cardiac disease,” said Lisa M. Hollier, MD, past president of the American College of Obstetricians and Gynecologists and professor at Baylor College of Medicine in Houston.
Dr. Magun and colleagues acknowledged that a key limitation of the present study was the retrospective, single-center design.
“With program expansions over the next 2-3 years, I expect to see an increasing number of prospective studies with larger sample sizes evaluating the impact of cardio-obstetrics teams on maternal morbidity and mortality,” Dr. Hollier said.
“These findings suggest that our cardio-obstetrics program may help provide improved cardiovascular care to an otherwise underserved population,” the authors concluded.
In an editorial accompanying the reports, Pamela Ouyang, MBBS, and Garima Sharma, MD, wrote that, although this study wasn’t designed to assess the benefit of cardio-obstetric teams relative to standard of care, its implementation of a multidisciplinary team-based care model showed excellent long-term outcomes.
The importance of coordinated postpartum follow-up with both cardiologists and obstetricians is becoming increasingly recognized, especially for women with poor pregnancy outcomes and with CVD that arises during pregnancy, such as pregnancy-associated spontaneous coronary artery dissection and peripartum cardiomyopathy, wrote Dr. Ouyang and Dr. Sharma, both with Johns Hopkins University in Baltimore.
“I’m very excited about the growing recognition of the importance of cardio-obstetrics and the emergence of many of these models of care at various institutions,” Melinda Davis, MD, of the University of Michigan in Ann Arbor, said in an interview.
“Over the next few years, I expect we will see several studies that show the benefits of the cardio-obstetrics model of care,” she explained. “Multicenter collaboration will be very important for learning about the optimal way to manage high-risk conditions during pregnancy.”
No funding sources were reported. The authors of this paper disclosed no conflicts of interest.
SOURCE: Magun E et al. JACC. 2020 Nov 3. doi: 10.1016/j.jacc.2020.08.071.
FROM THE JOURNAL OF THE AMERICAN COLLEGE OF CARDIOLOGY
Few women hospitalized for influenza have been vaccinated
Researchers analyzed data from 9,652 women ages 15-44 who were hospitalized with laboratory-confirmed influenza from October through April during the 2010-2019 influenza seasons. Data were pulled from the U.S. Influenza Hospitalization Surveillance Network (FluSurv-NET).
Of those women, 2,697 (28%) were pregnant. Median age was 28 and median gestational age was 32 weeks. Those studied included 36% who were non-Hispanic White; 29% non-Hispanic Black; and 20% Hispanic women.
Some 89% of the women, pregnant and nonpregnant, received antivirals while in the hospital but only 31% reported they had received the flu vaccine in the current season, despite guideline recommendations citing clear evidence that vaccination is safe for mother and baby.
Rachel Holstein, MPH, an epidemiology and information science fellow at the Centers for Disease Control and Prevention, who presented her team’s work as part of IDWeek 2020, explained that the mother’s vaccination can help protect the baby from flu infection for several months after birth, before the baby can be vaccinated.
She noted that pregnant women are at high risk for influenza-associated hospitalization.
“Changes in the immune system, heart, and lungs during pregnancy make pregnant women, and women up to 2 weeks post partum, more prone to severe illness from flu, including illness resulting in hospitalization,” she said in an interview
“Vaccination has been shown to reduce the risk of flu-associated acute respiratory infection in pregnant women by up to one-half,” she said. “A 2018 study showed that getting a flu shot reduced a pregnant woman’s risk of being hospitalized with flu by an average of 40%.»
FluSurv-NET data show hospitalizations were more common in the third trimester of pregnancy compared with the first and second, Holstein said. The most common underlying conditions among these women were asthma (23%) and obesity (10%), and 12% were current tobacco smokers. Overall, 5% of pregnant women with flu required ICU admission, 2% needed mechanical ventilation, and 6% developed pneumonia.
Vaccine uptake lowest in first two trimesters
Holstein said vaccine coverage was lowest among women in their first or second trimesters for all 9 seasons, and overall vaccination coverage increased significantly over time.
Uptake also differed by age. The data showed coverage was lower among women aged 15-34 years, compared with women 35 years and older (34% vs. 50%).
“It was as low as 15% among pregnant women aged 15-34 years in the 2011-12 season,” she added.
Jeanne Sheffield, MD, director of the division of maternal-fetal medicine at Johns Hopkins Medicine, Baltimore, said in an interview the low uptake of vaccine shown in this study is both familiar and frustrating.
She said education from health care providers has improved, but women are nonetheless frequently fearful. She pointed out the widespread phenomenon of vaccine hesitancy in the general population.
Coverage was 45.3% among adults in the 2018-2019 flu season, 8.2 percentage points higher than coverage during the 2017-18 season (37.1%) according to CDC estimates.
Added to that, she said, is further hesitancy when women believe vaccination could harm the unborn baby, despite “very good data that flu vaccine is safe in pregnancy, acceptable in pregnancy in all trimesters, and is optimal standard of care.”
Holstein added, “We know from past research that a range of factors – including negative attitudes and beliefs about vaccines, less knowledge about and access to vaccines, and a lack of trust in healthcare providers and vaccines – can contribute to lower vaccination rates.”
Healthcare providers play a key role in increasing flu vaccinations among pregnant women, she said.
“A provider recommendation, combined with an offer to administer a flu vaccine at the time of visit, remains one of the best ways to accomplish this,” Holstein said.
Holstein and Sheffield have disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
Researchers analyzed data from 9,652 women ages 15-44 who were hospitalized with laboratory-confirmed influenza from October through April during the 2010-2019 influenza seasons. Data were pulled from the U.S. Influenza Hospitalization Surveillance Network (FluSurv-NET).
Of those women, 2,697 (28%) were pregnant. Median age was 28 and median gestational age was 32 weeks. Those studied included 36% who were non-Hispanic White; 29% non-Hispanic Black; and 20% Hispanic women.
Some 89% of the women, pregnant and nonpregnant, received antivirals while in the hospital but only 31% reported they had received the flu vaccine in the current season, despite guideline recommendations citing clear evidence that vaccination is safe for mother and baby.
Rachel Holstein, MPH, an epidemiology and information science fellow at the Centers for Disease Control and Prevention, who presented her team’s work as part of IDWeek 2020, explained that the mother’s vaccination can help protect the baby from flu infection for several months after birth, before the baby can be vaccinated.
She noted that pregnant women are at high risk for influenza-associated hospitalization.
“Changes in the immune system, heart, and lungs during pregnancy make pregnant women, and women up to 2 weeks post partum, more prone to severe illness from flu, including illness resulting in hospitalization,” she said in an interview
“Vaccination has been shown to reduce the risk of flu-associated acute respiratory infection in pregnant women by up to one-half,” she said. “A 2018 study showed that getting a flu shot reduced a pregnant woman’s risk of being hospitalized with flu by an average of 40%.»
FluSurv-NET data show hospitalizations were more common in the third trimester of pregnancy compared with the first and second, Holstein said. The most common underlying conditions among these women were asthma (23%) and obesity (10%), and 12% were current tobacco smokers. Overall, 5% of pregnant women with flu required ICU admission, 2% needed mechanical ventilation, and 6% developed pneumonia.
Vaccine uptake lowest in first two trimesters
Holstein said vaccine coverage was lowest among women in their first or second trimesters for all 9 seasons, and overall vaccination coverage increased significantly over time.
Uptake also differed by age. The data showed coverage was lower among women aged 15-34 years, compared with women 35 years and older (34% vs. 50%).
“It was as low as 15% among pregnant women aged 15-34 years in the 2011-12 season,” she added.
Jeanne Sheffield, MD, director of the division of maternal-fetal medicine at Johns Hopkins Medicine, Baltimore, said in an interview the low uptake of vaccine shown in this study is both familiar and frustrating.
She said education from health care providers has improved, but women are nonetheless frequently fearful. She pointed out the widespread phenomenon of vaccine hesitancy in the general population.
Coverage was 45.3% among adults in the 2018-2019 flu season, 8.2 percentage points higher than coverage during the 2017-18 season (37.1%) according to CDC estimates.
Added to that, she said, is further hesitancy when women believe vaccination could harm the unborn baby, despite “very good data that flu vaccine is safe in pregnancy, acceptable in pregnancy in all trimesters, and is optimal standard of care.”
Holstein added, “We know from past research that a range of factors – including negative attitudes and beliefs about vaccines, less knowledge about and access to vaccines, and a lack of trust in healthcare providers and vaccines – can contribute to lower vaccination rates.”
Healthcare providers play a key role in increasing flu vaccinations among pregnant women, she said.
“A provider recommendation, combined with an offer to administer a flu vaccine at the time of visit, remains one of the best ways to accomplish this,” Holstein said.
Holstein and Sheffield have disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
Researchers analyzed data from 9,652 women ages 15-44 who were hospitalized with laboratory-confirmed influenza from October through April during the 2010-2019 influenza seasons. Data were pulled from the U.S. Influenza Hospitalization Surveillance Network (FluSurv-NET).
Of those women, 2,697 (28%) were pregnant. Median age was 28 and median gestational age was 32 weeks. Those studied included 36% who were non-Hispanic White; 29% non-Hispanic Black; and 20% Hispanic women.
Some 89% of the women, pregnant and nonpregnant, received antivirals while in the hospital but only 31% reported they had received the flu vaccine in the current season, despite guideline recommendations citing clear evidence that vaccination is safe for mother and baby.
Rachel Holstein, MPH, an epidemiology and information science fellow at the Centers for Disease Control and Prevention, who presented her team’s work as part of IDWeek 2020, explained that the mother’s vaccination can help protect the baby from flu infection for several months after birth, before the baby can be vaccinated.
She noted that pregnant women are at high risk for influenza-associated hospitalization.
“Changes in the immune system, heart, and lungs during pregnancy make pregnant women, and women up to 2 weeks post partum, more prone to severe illness from flu, including illness resulting in hospitalization,” she said in an interview
“Vaccination has been shown to reduce the risk of flu-associated acute respiratory infection in pregnant women by up to one-half,” she said. “A 2018 study showed that getting a flu shot reduced a pregnant woman’s risk of being hospitalized with flu by an average of 40%.»
FluSurv-NET data show hospitalizations were more common in the third trimester of pregnancy compared with the first and second, Holstein said. The most common underlying conditions among these women were asthma (23%) and obesity (10%), and 12% were current tobacco smokers. Overall, 5% of pregnant women with flu required ICU admission, 2% needed mechanical ventilation, and 6% developed pneumonia.
Vaccine uptake lowest in first two trimesters
Holstein said vaccine coverage was lowest among women in their first or second trimesters for all 9 seasons, and overall vaccination coverage increased significantly over time.
Uptake also differed by age. The data showed coverage was lower among women aged 15-34 years, compared with women 35 years and older (34% vs. 50%).
“It was as low as 15% among pregnant women aged 15-34 years in the 2011-12 season,” she added.
Jeanne Sheffield, MD, director of the division of maternal-fetal medicine at Johns Hopkins Medicine, Baltimore, said in an interview the low uptake of vaccine shown in this study is both familiar and frustrating.
She said education from health care providers has improved, but women are nonetheless frequently fearful. She pointed out the widespread phenomenon of vaccine hesitancy in the general population.
Coverage was 45.3% among adults in the 2018-2019 flu season, 8.2 percentage points higher than coverage during the 2017-18 season (37.1%) according to CDC estimates.
Added to that, she said, is further hesitancy when women believe vaccination could harm the unborn baby, despite “very good data that flu vaccine is safe in pregnancy, acceptable in pregnancy in all trimesters, and is optimal standard of care.”
Holstein added, “We know from past research that a range of factors – including negative attitudes and beliefs about vaccines, less knowledge about and access to vaccines, and a lack of trust in healthcare providers and vaccines – can contribute to lower vaccination rates.”
Healthcare providers play a key role in increasing flu vaccinations among pregnant women, she said.
“A provider recommendation, combined with an offer to administer a flu vaccine at the time of visit, remains one of the best ways to accomplish this,” Holstein said.
Holstein and Sheffield have disclosed no relevant financial relationships.
This article first appeared on Medscape.com.