User login
Children with uncontrolled asthma at higher risk of being bullied
The risk of bullying and teasing is higher in children and young people with poorer asthma control, an international study reported. Published online in the Archives of Disease in Childhood, the Room to Breathe survey of 943 children in six countries found 9.9% had experienced asthma-related bullying or teasing (n = 93).
Children with well-controlled disease, however, were less likely to report being victimized by asthma-related bullying/teasing: odds ratio, 0.51; 95% confidence interval, 0.23-0.84; P = .006).
“It’s important for pediatricians to recognize that children and young people with asthma commonly report bullying or teasing as a result of their condition,” Will Carroll, MD, of the Paediatric Respiratory Service at Staffordshire Children’s Hospital at Royal Stoke, Stoke-on-Trent, England, told this news organization. “Pediatricians should talk to children themselves with asthma about this and not just their parents, and efforts should be made to improve asthma control whenever possible.”
Though common and potentially long-lasting in its effects, bullying is rarely addressed by health care professionals, the U.K. authors said.
But things may differ in the United States. According to Mark Welles, MD, a pediatrician at Cohen Children’s Medical Center at Northwell Health in Queen’s, N.Y., and regional cochair of the American Academy of Pediatrics antibullying committee, young doctors here are trained to ask about bullying when seeing a child, no matter what the reason for the visit. “It’s important to build a rapport with the child, and you need to ask about the disease they may have but also generally ask, ‘How are things at school? Is everyone nice to you?’ It is becoming more common practice to ask this,” said Dr. Welles, who was not involved with the U.K. research.
The U.K. study drew on unpublished data from the Room to Breathe survey conducted by Dr. Carroll’s group during 2008-2009 in Canada, the United Kingdom, Greece, Hungary, South Africa, and the Netherlands. Only 358 of 930 (38.5%) children were found to be well controlled according to current Global Initiative for Asthma symptom-control criteria.
The analysis also found a highly significant association (P < .0001) between Childhood Asthma Control Test (C-ACT) score and reported bullying/teasing, with bullied children having lower scores. C-ACT–defined controlled asthma scores of 20 or higher were significantly associated with a lower risk of bullying (OR, 0.46; 95% CI, 0.28-0.76; P = .001).
In other study findings, harassment was more common in children whose asthma was serious enough to entail activity restriction (OR, 1.74; 95% CI, 1.11-2.75; P = .010) and who described their asthma as “bad” (OR, 3.02; 95% CI, 1.86-4.85; P < .001), as well as those whose parents reported ongoing asthma-related health worries (OR, 1.64; 95% CI, 1.04-2.58; P = .024).
“When a child is clearly different from others, such as having bad asthma or being limited in activities due to asthma, they stand out more and are more frequently bullied,” said Tracy Evian Waasdorp, PhD, MSEd, director of research for school-based bullying and social-emotional learning at Children’s Hospital of Philadelphia, and also not a participant in the U.K. study.
In contrast to the 10% bullying rate in Dr. Carroll’s study, Dr. Waasdorp referred to a CHOP analysis of more than 64,000 youth from a Northeastern state in which those with asthma were 40% more likely to be victims of in-person bullying and 70% were more likely to be cyberbullied than youth without asthma. “Having a medical condition can therefore put you at risk of being bullied regardless of what country you live in,” she said.
CHOP policy encourages practitioners to routinely ask about bullying and to provide handouts and resources for parents, she added.
Interestingly, the U.K. investigators found that open public use of spacers was not associated with asthma-related bullying, nor was parental worry at diagnosis or parental concern about steroid use.
But according to Dr. Welles, “Kids may be using the inhaler in front of other kids, and they may be embarrassed and not want to be seen as different. So they may not use the inhaler when needed for gym class or sports, forcing them to sit out and then potentially be bullied again. It’s a vicious cycle.”
Previous research has identified the bullying and teasing of children with food allergies.
Behaviors have included allergy-specific harassment such as smearing peanut butter on a youngster’s forehead or putting peanut butter cookie crumbs in a child’s lunch box.
“In our survey we asked the question ‘Have you been teased or bullied because of your asthma?’ but we didn’t ask what form this took,” Dr. Carroll said. “But we were surprised at just how many children said yes. It’s time for more research, I think.”
“There are never enough studies around this,” added Dr. Welles. “Bullying, whether because of asthma or otherwise, has the potential for long-term effects well into adulthood.”
In the meantime, asthma consultations should incorporate specific questions about bullying. They should also be child focused in order to gain a representative appreciation of asthma control and its effect on the child’s life.
“As pediatricians, we need to be continuously supporting parents and find the help they need to address any mental health issues,” Dr. Welles said. “Every pediatrician and parent needs to be aware and recognize when something is different in their child’s life. Please don’t ignore it.”
Dr. Waasdorp stressed that school and other communities should be aware that children with asthma may be at increased risk for aggression and harmful interactions related to their asthma. “Programming to reduce bullying should focus broadly on shifting the climate so that bullying is not perceived to be normative and on improving ‘upstander,’ or positive bystander, responses.” she said.
The original survey was funded by Nycomed (Zurich). No additional funding was requested for the current analysis. Dr. Carroll reported personal fees from GlaxoSmithKline, Novartis, and Trudell Medical International outside the submitted work. Dr. Welles and Dr. Waasdorp disclosed no competing interests relevant to their comments.
The risk of bullying and teasing is higher in children and young people with poorer asthma control, an international study reported. Published online in the Archives of Disease in Childhood, the Room to Breathe survey of 943 children in six countries found 9.9% had experienced asthma-related bullying or teasing (n = 93).
Children with well-controlled disease, however, were less likely to report being victimized by asthma-related bullying/teasing: odds ratio, 0.51; 95% confidence interval, 0.23-0.84; P = .006).
“It’s important for pediatricians to recognize that children and young people with asthma commonly report bullying or teasing as a result of their condition,” Will Carroll, MD, of the Paediatric Respiratory Service at Staffordshire Children’s Hospital at Royal Stoke, Stoke-on-Trent, England, told this news organization. “Pediatricians should talk to children themselves with asthma about this and not just their parents, and efforts should be made to improve asthma control whenever possible.”
Though common and potentially long-lasting in its effects, bullying is rarely addressed by health care professionals, the U.K. authors said.
But things may differ in the United States. According to Mark Welles, MD, a pediatrician at Cohen Children’s Medical Center at Northwell Health in Queen’s, N.Y., and regional cochair of the American Academy of Pediatrics antibullying committee, young doctors here are trained to ask about bullying when seeing a child, no matter what the reason for the visit. “It’s important to build a rapport with the child, and you need to ask about the disease they may have but also generally ask, ‘How are things at school? Is everyone nice to you?’ It is becoming more common practice to ask this,” said Dr. Welles, who was not involved with the U.K. research.
The U.K. study drew on unpublished data from the Room to Breathe survey conducted by Dr. Carroll’s group during 2008-2009 in Canada, the United Kingdom, Greece, Hungary, South Africa, and the Netherlands. Only 358 of 930 (38.5%) children were found to be well controlled according to current Global Initiative for Asthma symptom-control criteria.
The analysis also found a highly significant association (P < .0001) between Childhood Asthma Control Test (C-ACT) score and reported bullying/teasing, with bullied children having lower scores. C-ACT–defined controlled asthma scores of 20 or higher were significantly associated with a lower risk of bullying (OR, 0.46; 95% CI, 0.28-0.76; P = .001).
In other study findings, harassment was more common in children whose asthma was serious enough to entail activity restriction (OR, 1.74; 95% CI, 1.11-2.75; P = .010) and who described their asthma as “bad” (OR, 3.02; 95% CI, 1.86-4.85; P < .001), as well as those whose parents reported ongoing asthma-related health worries (OR, 1.64; 95% CI, 1.04-2.58; P = .024).
“When a child is clearly different from others, such as having bad asthma or being limited in activities due to asthma, they stand out more and are more frequently bullied,” said Tracy Evian Waasdorp, PhD, MSEd, director of research for school-based bullying and social-emotional learning at Children’s Hospital of Philadelphia, and also not a participant in the U.K. study.
In contrast to the 10% bullying rate in Dr. Carroll’s study, Dr. Waasdorp referred to a CHOP analysis of more than 64,000 youth from a Northeastern state in which those with asthma were 40% more likely to be victims of in-person bullying and 70% were more likely to be cyberbullied than youth without asthma. “Having a medical condition can therefore put you at risk of being bullied regardless of what country you live in,” she said.
CHOP policy encourages practitioners to routinely ask about bullying and to provide handouts and resources for parents, she added.
Interestingly, the U.K. investigators found that open public use of spacers was not associated with asthma-related bullying, nor was parental worry at diagnosis or parental concern about steroid use.
But according to Dr. Welles, “Kids may be using the inhaler in front of other kids, and they may be embarrassed and not want to be seen as different. So they may not use the inhaler when needed for gym class or sports, forcing them to sit out and then potentially be bullied again. It’s a vicious cycle.”
Previous research has identified the bullying and teasing of children with food allergies.
Behaviors have included allergy-specific harassment such as smearing peanut butter on a youngster’s forehead or putting peanut butter cookie crumbs in a child’s lunch box.
“In our survey we asked the question ‘Have you been teased or bullied because of your asthma?’ but we didn’t ask what form this took,” Dr. Carroll said. “But we were surprised at just how many children said yes. It’s time for more research, I think.”
“There are never enough studies around this,” added Dr. Welles. “Bullying, whether because of asthma or otherwise, has the potential for long-term effects well into adulthood.”
In the meantime, asthma consultations should incorporate specific questions about bullying. They should also be child focused in order to gain a representative appreciation of asthma control and its effect on the child’s life.
“As pediatricians, we need to be continuously supporting parents and find the help they need to address any mental health issues,” Dr. Welles said. “Every pediatrician and parent needs to be aware and recognize when something is different in their child’s life. Please don’t ignore it.”
Dr. Waasdorp stressed that school and other communities should be aware that children with asthma may be at increased risk for aggression and harmful interactions related to their asthma. “Programming to reduce bullying should focus broadly on shifting the climate so that bullying is not perceived to be normative and on improving ‘upstander,’ or positive bystander, responses.” she said.
The original survey was funded by Nycomed (Zurich). No additional funding was requested for the current analysis. Dr. Carroll reported personal fees from GlaxoSmithKline, Novartis, and Trudell Medical International outside the submitted work. Dr. Welles and Dr. Waasdorp disclosed no competing interests relevant to their comments.
The risk of bullying and teasing is higher in children and young people with poorer asthma control, an international study reported. Published online in the Archives of Disease in Childhood, the Room to Breathe survey of 943 children in six countries found 9.9% had experienced asthma-related bullying or teasing (n = 93).
Children with well-controlled disease, however, were less likely to report being victimized by asthma-related bullying/teasing: odds ratio, 0.51; 95% confidence interval, 0.23-0.84; P = .006).
“It’s important for pediatricians to recognize that children and young people with asthma commonly report bullying or teasing as a result of their condition,” Will Carroll, MD, of the Paediatric Respiratory Service at Staffordshire Children’s Hospital at Royal Stoke, Stoke-on-Trent, England, told this news organization. “Pediatricians should talk to children themselves with asthma about this and not just their parents, and efforts should be made to improve asthma control whenever possible.”
Though common and potentially long-lasting in its effects, bullying is rarely addressed by health care professionals, the U.K. authors said.
But things may differ in the United States. According to Mark Welles, MD, a pediatrician at Cohen Children’s Medical Center at Northwell Health in Queen’s, N.Y., and regional cochair of the American Academy of Pediatrics antibullying committee, young doctors here are trained to ask about bullying when seeing a child, no matter what the reason for the visit. “It’s important to build a rapport with the child, and you need to ask about the disease they may have but also generally ask, ‘How are things at school? Is everyone nice to you?’ It is becoming more common practice to ask this,” said Dr. Welles, who was not involved with the U.K. research.
The U.K. study drew on unpublished data from the Room to Breathe survey conducted by Dr. Carroll’s group during 2008-2009 in Canada, the United Kingdom, Greece, Hungary, South Africa, and the Netherlands. Only 358 of 930 (38.5%) children were found to be well controlled according to current Global Initiative for Asthma symptom-control criteria.
The analysis also found a highly significant association (P < .0001) between Childhood Asthma Control Test (C-ACT) score and reported bullying/teasing, with bullied children having lower scores. C-ACT–defined controlled asthma scores of 20 or higher were significantly associated with a lower risk of bullying (OR, 0.46; 95% CI, 0.28-0.76; P = .001).
In other study findings, harassment was more common in children whose asthma was serious enough to entail activity restriction (OR, 1.74; 95% CI, 1.11-2.75; P = .010) and who described their asthma as “bad” (OR, 3.02; 95% CI, 1.86-4.85; P < .001), as well as those whose parents reported ongoing asthma-related health worries (OR, 1.64; 95% CI, 1.04-2.58; P = .024).
“When a child is clearly different from others, such as having bad asthma or being limited in activities due to asthma, they stand out more and are more frequently bullied,” said Tracy Evian Waasdorp, PhD, MSEd, director of research for school-based bullying and social-emotional learning at Children’s Hospital of Philadelphia, and also not a participant in the U.K. study.
In contrast to the 10% bullying rate in Dr. Carroll’s study, Dr. Waasdorp referred to a CHOP analysis of more than 64,000 youth from a Northeastern state in which those with asthma were 40% more likely to be victims of in-person bullying and 70% were more likely to be cyberbullied than youth without asthma. “Having a medical condition can therefore put you at risk of being bullied regardless of what country you live in,” she said.
CHOP policy encourages practitioners to routinely ask about bullying and to provide handouts and resources for parents, she added.
Interestingly, the U.K. investigators found that open public use of spacers was not associated with asthma-related bullying, nor was parental worry at diagnosis or parental concern about steroid use.
But according to Dr. Welles, “Kids may be using the inhaler in front of other kids, and they may be embarrassed and not want to be seen as different. So they may not use the inhaler when needed for gym class or sports, forcing them to sit out and then potentially be bullied again. It’s a vicious cycle.”
Previous research has identified the bullying and teasing of children with food allergies.
Behaviors have included allergy-specific harassment such as smearing peanut butter on a youngster’s forehead or putting peanut butter cookie crumbs in a child’s lunch box.
“In our survey we asked the question ‘Have you been teased or bullied because of your asthma?’ but we didn’t ask what form this took,” Dr. Carroll said. “But we were surprised at just how many children said yes. It’s time for more research, I think.”
“There are never enough studies around this,” added Dr. Welles. “Bullying, whether because of asthma or otherwise, has the potential for long-term effects well into adulthood.”
In the meantime, asthma consultations should incorporate specific questions about bullying. They should also be child focused in order to gain a representative appreciation of asthma control and its effect on the child’s life.
“As pediatricians, we need to be continuously supporting parents and find the help they need to address any mental health issues,” Dr. Welles said. “Every pediatrician and parent needs to be aware and recognize when something is different in their child’s life. Please don’t ignore it.”
Dr. Waasdorp stressed that school and other communities should be aware that children with asthma may be at increased risk for aggression and harmful interactions related to their asthma. “Programming to reduce bullying should focus broadly on shifting the climate so that bullying is not perceived to be normative and on improving ‘upstander,’ or positive bystander, responses.” she said.
The original survey was funded by Nycomed (Zurich). No additional funding was requested for the current analysis. Dr. Carroll reported personal fees from GlaxoSmithKline, Novartis, and Trudell Medical International outside the submitted work. Dr. Welles and Dr. Waasdorp disclosed no competing interests relevant to their comments.
FROM ARCHIVES OF DISEASE IN CHILDHOOD
DRESS Syndrome Due to Cefdinir Mimicking Superinfected Eczema in a Pediatric Patient
To the Editor:
Drug reaction with eosinophilia and systemic symptoms (DRESS) syndrome, or drug-induced hypersensitivity syndrome, is a serious and potentially fatal multiorgan drug hypersensitivity reaction. Drug reaction with eosinophilia and systemic symptoms syndrome shares many clinical features with viral exanthems and may be difficult to diagnose in the setting of atopic dermatitis (AD) in which children may have baseline eosinophilia from an atopic diathesis. The cutaneous exanthema also may be variable in presentation, further complicating diagnosis.1,2
A 3-year-old boy with AD since infancy and a history of anaphylaxis to peanuts presented to the emergency department with reported fever, rash, sore throat, and decreased oral intake. Ten days prior, the patient was treated for cellulitis of the left foot with a 7-day course of cefdinir with complete resolution of symptoms. Four days prior to admission, the patient started developing “bumps” on the face and fevers. He was seen at an outside facility, where a rapid test for Streptococcus was negative, and the patient was treated with ibuprofen and fluids for a presumed viral exanthem. The rash subsequently spread to involve the trunk and extremities. On the day of admission, the patient had a positive rapid test for Streptococcus and was referred to the emergency department with concern for superinfected eczema and eczema herpeticum. The patient recently traveled to Puerto Rico, where he had contact with an aunt with active herpes zoster but no other sick contacts. The patient’s immunizations were reported to be up-to-date.
Physical examination revealed the patient was afebrile but irritable and had erythematous crusted papules and patches on the face, arms, and legs, as well as erythematous dry patches on the chest, abdomen, and back (Figure). There were no conjunctival erythematous or oral erosions. The patient was admitted to the hospital for presumed superinfected AD and possible eczema herpeticum. He was started on intravenous clindamycin and acyclovir.

The following day, the patient had new facial edema and fever (temperature, 102.8 °F [39.36 °C]) in addition to palpable mobile cervical, axillary, and inguinal lymphadenopathy. He also was noted to have notably worsening eosinophilia from 1288 (14%) to 2570 (29.2%) cells/µL (reference range, 0%–5%) and new-onset transaminitis. Herpes and varicella-zoster direct fluorescent antibody tests, culture, and serum polymerase chain reaction were all negative, and acyclovir was discontinued. Repeat laboratory tests 12 hours later showed a continued uptrend in transaminitis. Serologies for acute and chronic cytomegalovirus; Epstein-Barr virus; and hepatitis A, B, and C were all nonreactive. The patient was started on intravenous methylprednisolone 1 mg/kg daily for suspected DRESS syndrome likely due to cefdinir.
The patient’s eosinophilia completely resolved (from approximately 2600 to 100 cells/µL) after 1 dose of steroids, and his transaminitis trended down over the next few days. He remained afebrile for the remainder of his admission, and his facial swelling and rash continued to improve. Bacterial culture from the skin grew oxacillin-susceptible Staphylococcus aureus and group A Streptococcus pyogenes. A blood culture was negative. The patient was discharged home to complete a 10-day course of clindamycin and was given topical steroids for the eczema. He continued on oral prednisolone 1 mg/kg daily for 10 days, after which the dose was tapered down for a total 1-month course of systemic corticosteroids. At 1-month follow-up after completing the course of steroids, he was doing well with normal hepatic enzyme levels and no recurrence of fever, facial edema, or rash. He continues to be followed for management of the AD.
Drug reaction with eosinophilia and systemic symptoms syndrome is a serious systemic adverse drug reaction, with high morbidity and even mortality, estimated at 10% in the adult population, though more specific pediatric mortality data are not available.1,2 The exact pathogenesis of DRESS syndrome has not been elucidated. Certain human leukocyte antigen class I alleles are predisposed to the development of DRESS syndrome, but there has not been a human leukocyte antigen subtype identified with beta-lactam–associated DRESS syndrome. Some studies have demonstrated a reactivation of human herpesvirus 6, human herpesvirus 7, and Epstein-Barr virus.3 One study involving 40 patients with DRESS syndrome identified viremia in 76% (29/38) of patients and identified CD8+ T-cell populations directed toward viral epitopes.3 Finally, DRESS syndrome may be related to the slow detoxification and elimination of intermediary products of offending medications that serve as an immunogenic stimulus for the inflammatory cascade.2
In adults, DRESS syndrome was first identified in association with phenytoin, but more recently other drugs have been identified, including other aromatic anticonvulsants (ie, lamotrigine, phenobarbital, carbamazepine), allopurinol, sulfonamides, antiretrovirals (particularly abacavir), and minocycline.2 In a 3-year pediatric prospective study, 11 cases of DRESS syndrome were identified: 4 cases due to lamotrigine, and 3 caused by penicillins.4 The trigger in our patient’s case was the beta-lactam, third-generation cephalosporin cefdinir, and his symptoms developed within 6 days of starting the medication. Many articles report that beta-lactams are a rare cause of DRESS syndrome, with only a handful of cases reported.1,5,6
The diagnosis of DRESS syndrome often can be delayed, as children present acutely febrile and toxic appearing. Unlike many adverse drug reactions, DRESS syndrome does not show rapid resolution with withdrawal of the causative agent, further complicating the diagnosis. The typical onset of DRESS syndrome generally ranges from 2 to 6 weeks after the initiation of the offending drug; however, faster onset of symptoms, similar to our case, has been noted in antibiotic-triggered cases. In the prospective pediatric series by Sasidharanpillai et al,4 the average time to onset among 3 antibiotic-triggered DRESS cases was 5.8 days vs 23.9 days among the 4 cases of lamotrigine-associated DRESS syndrome.
Our patient demonstrated the classic features of DRESS syndrome, including fever, rash, lymphadenopathy, facial edema, peripheral eosinophilia, atypical lymphocytosis, and hepatitis. Based on the proposed RegiSCAR scoring system, our patient was classified as a “definite” case of DRESS syndrome.1,7 Other hematologic findings in DRESS syndrome may include thrombocytopenia and anemia. The liver is the most commonly affected internal organ in DRESS syndrome, with pneumonitis, carditis, and nephritis reported less frequently.1 The pattern of liver injury in our patient was mixed (hepatocellular and cholestatic), the second most common pattern in patients with DRESS syndrome (the cholestatic pattern is most common).8
The exanthem of DRESS syndrome can vary in morphology, with up to 7% of patients reported to have eczemalike lesions in the multinational prospective RegiSCAR study.1 Other entities in the differential diagnosis for our patient included Kawasaki disease, where conjunctivitis and strawberry tongue are classically present, as well as erythrodermic AD, where internal organ involvement is not common.2 Our patient’s exanthem initially was considered to be a flare of AD with superimposed bacterial infection and possible eczema herpeticum. Although bacterial cultures did grow Staphylococcus and Streptococcus, viral studies were all negative, and this alone would not have explained the facial edema, rapidly rising eosinophil count, and transaminitis. The dramatic drop in his eosinophil count and decrease in hepatic enzymes after 1 dose of intravenous methylprednisolone also supported the diagnosis of DRESS syndrome.
Treatment recommendations remain largely anecdotal. Early systemic steroids generally are accepted as the first line of therapy, with a slow taper. Although the average required duration of systemic steroids in 1 series of adults was reported at 50.1 days,9 the duration was shorter (21–35 days) in a series of pediatric patients.4 Our patient’s clinical symptoms and laboratory values normalized after completing a 1-month steroid taper. Other therapies have been tried for recalcitrant cases, including intravenous immunoglobulin, plasmapheresis, rituximab, and valganciclovir.2
Early clinical recognition of the signs and symptoms of DRESS syndrome in the setting of a new medication can decrease morbidity and mortality. Although DRESS syndrome in pediatric patients presents with many similar clinical features as in adults, it may be a greater diagnostic challenge. As in adult cases, timely administration of systemic corticosteroids and tapering based on clinical signs and symptoms can lead to resolution of the hypersensitivity syndrome.
- Kardaun SH, Sekula P, Valeyrie-Allanore L, et al. Drug reaction with eosinophilia and systemic symptoms (DRESS): an original multisystem adverse drug reaction. results from the prospective RegiSCAR study. Br J Dermatol. 2013;169:1071-1080.
- Fernando SL. Drug-reaction eosinophilia and systemic symptoms and drug-induced hypersensitivity syndrome. Australas J Dermatol. 2014;55:15-23.
- Picard D, Janela B, Descamps V, et al. Drug reaction with eosinophilia and systemic symptoms (DRESS): a multiorgan antiviral T cell response. Sci Transl Med. 2010;2:46ra62.
- Sasidharanpillai S, Sabitha S, Riyaz N, et al. Drug reaction with eosinophilia and systemic symptoms in children: a prospective study. Pediatr Dermatol. 2016;33:E162-E165.
- Aouam K, Chaabane A, Toumi A, et al. Drug rash with eosinophilia and systemic symptoms (DRESS) probably induced by cefotaxime: a report of two cases. Clin Med Res. 2012;10:32-35.
- Guleria VS, Dhillon M, Gill S, et al. Ceftriaxone induced drug rash with eosinophilia and systemic symptoms. J Res Pharm Pract. 2014;3:72-74.
- Kardaun SH, Sidoroff A, Valeyrie-Allanore L, et al. Variability in the clinical pattern of cutaneous side-effects of drugs with systemic symptoms: does a DRESS syndrome really exist? Br J Dermatol. 2007;156:609-611.
- Lin IC, Yang HC, Strong C, et al. Liver injury in patients with DRESS: a clinical study of 72 cases. J Am Acad Dermatol. 2015;72:984-991.
- Ang CC, Wang YS, Yoosuff EL, et al. Retrospective analysis of drug-induced hypersensitivity syndrome: a study of 27 patients. J Am Acad Dermatol. 2010;63:219-227.
To the Editor:
Drug reaction with eosinophilia and systemic symptoms (DRESS) syndrome, or drug-induced hypersensitivity syndrome, is a serious and potentially fatal multiorgan drug hypersensitivity reaction. Drug reaction with eosinophilia and systemic symptoms syndrome shares many clinical features with viral exanthems and may be difficult to diagnose in the setting of atopic dermatitis (AD) in which children may have baseline eosinophilia from an atopic diathesis. The cutaneous exanthema also may be variable in presentation, further complicating diagnosis.1,2
A 3-year-old boy with AD since infancy and a history of anaphylaxis to peanuts presented to the emergency department with reported fever, rash, sore throat, and decreased oral intake. Ten days prior, the patient was treated for cellulitis of the left foot with a 7-day course of cefdinir with complete resolution of symptoms. Four days prior to admission, the patient started developing “bumps” on the face and fevers. He was seen at an outside facility, where a rapid test for Streptococcus was negative, and the patient was treated with ibuprofen and fluids for a presumed viral exanthem. The rash subsequently spread to involve the trunk and extremities. On the day of admission, the patient had a positive rapid test for Streptococcus and was referred to the emergency department with concern for superinfected eczema and eczema herpeticum. The patient recently traveled to Puerto Rico, where he had contact with an aunt with active herpes zoster but no other sick contacts. The patient’s immunizations were reported to be up-to-date.
Physical examination revealed the patient was afebrile but irritable and had erythematous crusted papules and patches on the face, arms, and legs, as well as erythematous dry patches on the chest, abdomen, and back (Figure). There were no conjunctival erythematous or oral erosions. The patient was admitted to the hospital for presumed superinfected AD and possible eczema herpeticum. He was started on intravenous clindamycin and acyclovir.

The following day, the patient had new facial edema and fever (temperature, 102.8 °F [39.36 °C]) in addition to palpable mobile cervical, axillary, and inguinal lymphadenopathy. He also was noted to have notably worsening eosinophilia from 1288 (14%) to 2570 (29.2%) cells/µL (reference range, 0%–5%) and new-onset transaminitis. Herpes and varicella-zoster direct fluorescent antibody tests, culture, and serum polymerase chain reaction were all negative, and acyclovir was discontinued. Repeat laboratory tests 12 hours later showed a continued uptrend in transaminitis. Serologies for acute and chronic cytomegalovirus; Epstein-Barr virus; and hepatitis A, B, and C were all nonreactive. The patient was started on intravenous methylprednisolone 1 mg/kg daily for suspected DRESS syndrome likely due to cefdinir.
The patient’s eosinophilia completely resolved (from approximately 2600 to 100 cells/µL) after 1 dose of steroids, and his transaminitis trended down over the next few days. He remained afebrile for the remainder of his admission, and his facial swelling and rash continued to improve. Bacterial culture from the skin grew oxacillin-susceptible Staphylococcus aureus and group A Streptococcus pyogenes. A blood culture was negative. The patient was discharged home to complete a 10-day course of clindamycin and was given topical steroids for the eczema. He continued on oral prednisolone 1 mg/kg daily for 10 days, after which the dose was tapered down for a total 1-month course of systemic corticosteroids. At 1-month follow-up after completing the course of steroids, he was doing well with normal hepatic enzyme levels and no recurrence of fever, facial edema, or rash. He continues to be followed for management of the AD.
Drug reaction with eosinophilia and systemic symptoms syndrome is a serious systemic adverse drug reaction, with high morbidity and even mortality, estimated at 10% in the adult population, though more specific pediatric mortality data are not available.1,2 The exact pathogenesis of DRESS syndrome has not been elucidated. Certain human leukocyte antigen class I alleles are predisposed to the development of DRESS syndrome, but there has not been a human leukocyte antigen subtype identified with beta-lactam–associated DRESS syndrome. Some studies have demonstrated a reactivation of human herpesvirus 6, human herpesvirus 7, and Epstein-Barr virus.3 One study involving 40 patients with DRESS syndrome identified viremia in 76% (29/38) of patients and identified CD8+ T-cell populations directed toward viral epitopes.3 Finally, DRESS syndrome may be related to the slow detoxification and elimination of intermediary products of offending medications that serve as an immunogenic stimulus for the inflammatory cascade.2
In adults, DRESS syndrome was first identified in association with phenytoin, but more recently other drugs have been identified, including other aromatic anticonvulsants (ie, lamotrigine, phenobarbital, carbamazepine), allopurinol, sulfonamides, antiretrovirals (particularly abacavir), and minocycline.2 In a 3-year pediatric prospective study, 11 cases of DRESS syndrome were identified: 4 cases due to lamotrigine, and 3 caused by penicillins.4 The trigger in our patient’s case was the beta-lactam, third-generation cephalosporin cefdinir, and his symptoms developed within 6 days of starting the medication. Many articles report that beta-lactams are a rare cause of DRESS syndrome, with only a handful of cases reported.1,5,6
The diagnosis of DRESS syndrome often can be delayed, as children present acutely febrile and toxic appearing. Unlike many adverse drug reactions, DRESS syndrome does not show rapid resolution with withdrawal of the causative agent, further complicating the diagnosis. The typical onset of DRESS syndrome generally ranges from 2 to 6 weeks after the initiation of the offending drug; however, faster onset of symptoms, similar to our case, has been noted in antibiotic-triggered cases. In the prospective pediatric series by Sasidharanpillai et al,4 the average time to onset among 3 antibiotic-triggered DRESS cases was 5.8 days vs 23.9 days among the 4 cases of lamotrigine-associated DRESS syndrome.
Our patient demonstrated the classic features of DRESS syndrome, including fever, rash, lymphadenopathy, facial edema, peripheral eosinophilia, atypical lymphocytosis, and hepatitis. Based on the proposed RegiSCAR scoring system, our patient was classified as a “definite” case of DRESS syndrome.1,7 Other hematologic findings in DRESS syndrome may include thrombocytopenia and anemia. The liver is the most commonly affected internal organ in DRESS syndrome, with pneumonitis, carditis, and nephritis reported less frequently.1 The pattern of liver injury in our patient was mixed (hepatocellular and cholestatic), the second most common pattern in patients with DRESS syndrome (the cholestatic pattern is most common).8
The exanthem of DRESS syndrome can vary in morphology, with up to 7% of patients reported to have eczemalike lesions in the multinational prospective RegiSCAR study.1 Other entities in the differential diagnosis for our patient included Kawasaki disease, where conjunctivitis and strawberry tongue are classically present, as well as erythrodermic AD, where internal organ involvement is not common.2 Our patient’s exanthem initially was considered to be a flare of AD with superimposed bacterial infection and possible eczema herpeticum. Although bacterial cultures did grow Staphylococcus and Streptococcus, viral studies were all negative, and this alone would not have explained the facial edema, rapidly rising eosinophil count, and transaminitis. The dramatic drop in his eosinophil count and decrease in hepatic enzymes after 1 dose of intravenous methylprednisolone also supported the diagnosis of DRESS syndrome.
Treatment recommendations remain largely anecdotal. Early systemic steroids generally are accepted as the first line of therapy, with a slow taper. Although the average required duration of systemic steroids in 1 series of adults was reported at 50.1 days,9 the duration was shorter (21–35 days) in a series of pediatric patients.4 Our patient’s clinical symptoms and laboratory values normalized after completing a 1-month steroid taper. Other therapies have been tried for recalcitrant cases, including intravenous immunoglobulin, plasmapheresis, rituximab, and valganciclovir.2
Early clinical recognition of the signs and symptoms of DRESS syndrome in the setting of a new medication can decrease morbidity and mortality. Although DRESS syndrome in pediatric patients presents with many similar clinical features as in adults, it may be a greater diagnostic challenge. As in adult cases, timely administration of systemic corticosteroids and tapering based on clinical signs and symptoms can lead to resolution of the hypersensitivity syndrome.
To the Editor:
Drug reaction with eosinophilia and systemic symptoms (DRESS) syndrome, or drug-induced hypersensitivity syndrome, is a serious and potentially fatal multiorgan drug hypersensitivity reaction. Drug reaction with eosinophilia and systemic symptoms syndrome shares many clinical features with viral exanthems and may be difficult to diagnose in the setting of atopic dermatitis (AD) in which children may have baseline eosinophilia from an atopic diathesis. The cutaneous exanthema also may be variable in presentation, further complicating diagnosis.1,2
A 3-year-old boy with AD since infancy and a history of anaphylaxis to peanuts presented to the emergency department with reported fever, rash, sore throat, and decreased oral intake. Ten days prior, the patient was treated for cellulitis of the left foot with a 7-day course of cefdinir with complete resolution of symptoms. Four days prior to admission, the patient started developing “bumps” on the face and fevers. He was seen at an outside facility, where a rapid test for Streptococcus was negative, and the patient was treated with ibuprofen and fluids for a presumed viral exanthem. The rash subsequently spread to involve the trunk and extremities. On the day of admission, the patient had a positive rapid test for Streptococcus and was referred to the emergency department with concern for superinfected eczema and eczema herpeticum. The patient recently traveled to Puerto Rico, where he had contact with an aunt with active herpes zoster but no other sick contacts. The patient’s immunizations were reported to be up-to-date.
Physical examination revealed the patient was afebrile but irritable and had erythematous crusted papules and patches on the face, arms, and legs, as well as erythematous dry patches on the chest, abdomen, and back (Figure). There were no conjunctival erythematous or oral erosions. The patient was admitted to the hospital for presumed superinfected AD and possible eczema herpeticum. He was started on intravenous clindamycin and acyclovir.

The following day, the patient had new facial edema and fever (temperature, 102.8 °F [39.36 °C]) in addition to palpable mobile cervical, axillary, and inguinal lymphadenopathy. He also was noted to have notably worsening eosinophilia from 1288 (14%) to 2570 (29.2%) cells/µL (reference range, 0%–5%) and new-onset transaminitis. Herpes and varicella-zoster direct fluorescent antibody tests, culture, and serum polymerase chain reaction were all negative, and acyclovir was discontinued. Repeat laboratory tests 12 hours later showed a continued uptrend in transaminitis. Serologies for acute and chronic cytomegalovirus; Epstein-Barr virus; and hepatitis A, B, and C were all nonreactive. The patient was started on intravenous methylprednisolone 1 mg/kg daily for suspected DRESS syndrome likely due to cefdinir.
The patient’s eosinophilia completely resolved (from approximately 2600 to 100 cells/µL) after 1 dose of steroids, and his transaminitis trended down over the next few days. He remained afebrile for the remainder of his admission, and his facial swelling and rash continued to improve. Bacterial culture from the skin grew oxacillin-susceptible Staphylococcus aureus and group A Streptococcus pyogenes. A blood culture was negative. The patient was discharged home to complete a 10-day course of clindamycin and was given topical steroids for the eczema. He continued on oral prednisolone 1 mg/kg daily for 10 days, after which the dose was tapered down for a total 1-month course of systemic corticosteroids. At 1-month follow-up after completing the course of steroids, he was doing well with normal hepatic enzyme levels and no recurrence of fever, facial edema, or rash. He continues to be followed for management of the AD.
Drug reaction with eosinophilia and systemic symptoms syndrome is a serious systemic adverse drug reaction, with high morbidity and even mortality, estimated at 10% in the adult population, though more specific pediatric mortality data are not available.1,2 The exact pathogenesis of DRESS syndrome has not been elucidated. Certain human leukocyte antigen class I alleles are predisposed to the development of DRESS syndrome, but there has not been a human leukocyte antigen subtype identified with beta-lactam–associated DRESS syndrome. Some studies have demonstrated a reactivation of human herpesvirus 6, human herpesvirus 7, and Epstein-Barr virus.3 One study involving 40 patients with DRESS syndrome identified viremia in 76% (29/38) of patients and identified CD8+ T-cell populations directed toward viral epitopes.3 Finally, DRESS syndrome may be related to the slow detoxification and elimination of intermediary products of offending medications that serve as an immunogenic stimulus for the inflammatory cascade.2
In adults, DRESS syndrome was first identified in association with phenytoin, but more recently other drugs have been identified, including other aromatic anticonvulsants (ie, lamotrigine, phenobarbital, carbamazepine), allopurinol, sulfonamides, antiretrovirals (particularly abacavir), and minocycline.2 In a 3-year pediatric prospective study, 11 cases of DRESS syndrome were identified: 4 cases due to lamotrigine, and 3 caused by penicillins.4 The trigger in our patient’s case was the beta-lactam, third-generation cephalosporin cefdinir, and his symptoms developed within 6 days of starting the medication. Many articles report that beta-lactams are a rare cause of DRESS syndrome, with only a handful of cases reported.1,5,6
The diagnosis of DRESS syndrome often can be delayed, as children present acutely febrile and toxic appearing. Unlike many adverse drug reactions, DRESS syndrome does not show rapid resolution with withdrawal of the causative agent, further complicating the diagnosis. The typical onset of DRESS syndrome generally ranges from 2 to 6 weeks after the initiation of the offending drug; however, faster onset of symptoms, similar to our case, has been noted in antibiotic-triggered cases. In the prospective pediatric series by Sasidharanpillai et al,4 the average time to onset among 3 antibiotic-triggered DRESS cases was 5.8 days vs 23.9 days among the 4 cases of lamotrigine-associated DRESS syndrome.
Our patient demonstrated the classic features of DRESS syndrome, including fever, rash, lymphadenopathy, facial edema, peripheral eosinophilia, atypical lymphocytosis, and hepatitis. Based on the proposed RegiSCAR scoring system, our patient was classified as a “definite” case of DRESS syndrome.1,7 Other hematologic findings in DRESS syndrome may include thrombocytopenia and anemia. The liver is the most commonly affected internal organ in DRESS syndrome, with pneumonitis, carditis, and nephritis reported less frequently.1 The pattern of liver injury in our patient was mixed (hepatocellular and cholestatic), the second most common pattern in patients with DRESS syndrome (the cholestatic pattern is most common).8
The exanthem of DRESS syndrome can vary in morphology, with up to 7% of patients reported to have eczemalike lesions in the multinational prospective RegiSCAR study.1 Other entities in the differential diagnosis for our patient included Kawasaki disease, where conjunctivitis and strawberry tongue are classically present, as well as erythrodermic AD, where internal organ involvement is not common.2 Our patient’s exanthem initially was considered to be a flare of AD with superimposed bacterial infection and possible eczema herpeticum. Although bacterial cultures did grow Staphylococcus and Streptococcus, viral studies were all negative, and this alone would not have explained the facial edema, rapidly rising eosinophil count, and transaminitis. The dramatic drop in his eosinophil count and decrease in hepatic enzymes after 1 dose of intravenous methylprednisolone also supported the diagnosis of DRESS syndrome.
Treatment recommendations remain largely anecdotal. Early systemic steroids generally are accepted as the first line of therapy, with a slow taper. Although the average required duration of systemic steroids in 1 series of adults was reported at 50.1 days,9 the duration was shorter (21–35 days) in a series of pediatric patients.4 Our patient’s clinical symptoms and laboratory values normalized after completing a 1-month steroid taper. Other therapies have been tried for recalcitrant cases, including intravenous immunoglobulin, plasmapheresis, rituximab, and valganciclovir.2
Early clinical recognition of the signs and symptoms of DRESS syndrome in the setting of a new medication can decrease morbidity and mortality. Although DRESS syndrome in pediatric patients presents with many similar clinical features as in adults, it may be a greater diagnostic challenge. As in adult cases, timely administration of systemic corticosteroids and tapering based on clinical signs and symptoms can lead to resolution of the hypersensitivity syndrome.
- Kardaun SH, Sekula P, Valeyrie-Allanore L, et al. Drug reaction with eosinophilia and systemic symptoms (DRESS): an original multisystem adverse drug reaction. results from the prospective RegiSCAR study. Br J Dermatol. 2013;169:1071-1080.
- Fernando SL. Drug-reaction eosinophilia and systemic symptoms and drug-induced hypersensitivity syndrome. Australas J Dermatol. 2014;55:15-23.
- Picard D, Janela B, Descamps V, et al. Drug reaction with eosinophilia and systemic symptoms (DRESS): a multiorgan antiviral T cell response. Sci Transl Med. 2010;2:46ra62.
- Sasidharanpillai S, Sabitha S, Riyaz N, et al. Drug reaction with eosinophilia and systemic symptoms in children: a prospective study. Pediatr Dermatol. 2016;33:E162-E165.
- Aouam K, Chaabane A, Toumi A, et al. Drug rash with eosinophilia and systemic symptoms (DRESS) probably induced by cefotaxime: a report of two cases. Clin Med Res. 2012;10:32-35.
- Guleria VS, Dhillon M, Gill S, et al. Ceftriaxone induced drug rash with eosinophilia and systemic symptoms. J Res Pharm Pract. 2014;3:72-74.
- Kardaun SH, Sidoroff A, Valeyrie-Allanore L, et al. Variability in the clinical pattern of cutaneous side-effects of drugs with systemic symptoms: does a DRESS syndrome really exist? Br J Dermatol. 2007;156:609-611.
- Lin IC, Yang HC, Strong C, et al. Liver injury in patients with DRESS: a clinical study of 72 cases. J Am Acad Dermatol. 2015;72:984-991.
- Ang CC, Wang YS, Yoosuff EL, et al. Retrospective analysis of drug-induced hypersensitivity syndrome: a study of 27 patients. J Am Acad Dermatol. 2010;63:219-227.
- Kardaun SH, Sekula P, Valeyrie-Allanore L, et al. Drug reaction with eosinophilia and systemic symptoms (DRESS): an original multisystem adverse drug reaction. results from the prospective RegiSCAR study. Br J Dermatol. 2013;169:1071-1080.
- Fernando SL. Drug-reaction eosinophilia and systemic symptoms and drug-induced hypersensitivity syndrome. Australas J Dermatol. 2014;55:15-23.
- Picard D, Janela B, Descamps V, et al. Drug reaction with eosinophilia and systemic symptoms (DRESS): a multiorgan antiviral T cell response. Sci Transl Med. 2010;2:46ra62.
- Sasidharanpillai S, Sabitha S, Riyaz N, et al. Drug reaction with eosinophilia and systemic symptoms in children: a prospective study. Pediatr Dermatol. 2016;33:E162-E165.
- Aouam K, Chaabane A, Toumi A, et al. Drug rash with eosinophilia and systemic symptoms (DRESS) probably induced by cefotaxime: a report of two cases. Clin Med Res. 2012;10:32-35.
- Guleria VS, Dhillon M, Gill S, et al. Ceftriaxone induced drug rash with eosinophilia and systemic symptoms. J Res Pharm Pract. 2014;3:72-74.
- Kardaun SH, Sidoroff A, Valeyrie-Allanore L, et al. Variability in the clinical pattern of cutaneous side-effects of drugs with systemic symptoms: does a DRESS syndrome really exist? Br J Dermatol. 2007;156:609-611.
- Lin IC, Yang HC, Strong C, et al. Liver injury in patients with DRESS: a clinical study of 72 cases. J Am Acad Dermatol. 2015;72:984-991.
- Ang CC, Wang YS, Yoosuff EL, et al. Retrospective analysis of drug-induced hypersensitivity syndrome: a study of 27 patients. J Am Acad Dermatol. 2010;63:219-227.
Practice Points
- Drug reaction with eosinophilia and systemic symptoms (DRESS) syndrome shares many clinical features with viral exanthems and may be difficult to diagnose in the setting of atopic dermatitis in which children may have baseline eosinophilia from an atopic diathesis.
- Early clinical recognition of the signs and symptoms of DRESS syndrome in the setting of a new medication can decrease morbidity and mortality.
Genomic screening of healthy newborns gets more popular
Even before their baby is born, parents face some tough questions: Home birth or hospital? Cloth or disposable diapers? Breast, bottle, or both? But advances in genetic sequencing technology mean that parents will soon face yet another choice: whether to sequence their newborn’s DNA for an overview of the baby’s entire genome.
Genetic testing has been used for decades to diagnose conditions even before birth. But DNA sequencing technologies, once expensive and tough to access, are now rapid and cheap enough that doctors could order genomic screening for any infant, regardless of health status.
The possibility has raised many questions about the ethical, legal, and social repercussions of doing so. One of the biggest sticking points of sequencing newborns is the potential psychosocial fallout for families of such wide-scale use of genetic screening.
“There’s a narrative of catastrophic distress,” says Robert Green, MD, a geneticist at Harvard Medical School and lead investigator on the BabySeq study, which is evaluating the medical, social, and economic consequences of newborn genetic screening. The concern is that parents learning that their child carries a gene variant related to cancer or heart disease will become “incredibly anxious and distressed,” he says. “And it’s not an unreasonable speculation.”
But Dr. Green’s team found no evidence of such anxiety in the results from a randomized trial it conducted, published in JAMA Pediatrics. In the meantime, Genomics England announced it would begin a pilot study involving whole-genome sequencing of up to 200,000 babies. The first goal is to identify severe disease that starts in childhood, but the information would also be stored and used to detect drug sensitivities and conditions that come up later in life.
The large U.K. project is a bold move, according to David Amor, PhD, a pediatric geneticist at Murdoch Children’s Research Institute in Australia, who says its time has come. Geneticists have been accused of thinking their field involves unique pitfalls, compared with the rest of medicine, he points out, and that doctors need to protect patients and families from the potential harm genetic testing poses.
“But it is becoming apparent that that’s not really the case,” he says, and “maybe there’s not a whole lot special about genetics – it’s just medicine.”
When a first-draft copy of the human genome was published in 2001, scientists and doctors hailed the start of a new era of precision medicine. Knowing our genome sequence was expected to lead to a better grasp on our individual disease risks. Yet even as technologies advanced, clinical genetics remained focused on diagnosis rather than screening, according to Lilian Downie, a clinical genetics PhD candidate at the University of Melbourne. She calls the difference subtle but important.
Diagnostic genetic testing confirms whether a person has a specific condition, whereas genetic screening tests evaluate someone’s risk of getting an illness. Both approaches use sequencing, but they answer different questions, explains Ms. Downie.
Diagnosing disease versus predicting future illness
Genetic testing is on the upswing for both purposes, whether clinically for diagnosis or through direct-to-consumer screening-oriented services like 23andMe. Scientists began to note that many people carried disease-related genetic variants without having signs of disease. In some cases, a variant that is mathematically linked to a disease simply doesn’t cause it. In other cases, though, even if the gene variant contributes to a disease, not everyone who carries the genetic change will get the condition.
This potential disconnect between having a variant and developing the condition is a big problem, says Katie Stoll, a genetic counselor and executive director of the Genetic Support Foundation in Olympia, WA.
“It’s more complicated than just looking at one gene variant and one outcome,” she says. Without a sure link between the two, this information could unnecessarily entail “some pretty big emotional and financial costs.”
Ms. Stoll and others in the genetics field who share similar concerns are one reason the BabySeq project was first funded back in 2015. Although the overall aim of the initiative is to answer questions about the value of genomic sequencing in newborn screening, the media and scientific attention has focused on the psychosocial impact of healthy newborn sequencing, says Dr. Green. In the study published in JAMA Pediatrics, his group focused on these issues, too.
For that randomized trial, they enrolled 325 families, 257 with healthy babies and 68 whose babies had spent time in neonatal intensive care. Enrolled infants were randomly given standard care alone or standard care with genomic sequencing added on. The genomic sequencing report contained information about the presence of genetic variants associated with disease that start in childhood. Parents also could choose whether to learn about genetic risks for conditions that start in adulthood, such as cancer.
Boston-based Tina Moniz was one of those parents. When her first daughter was born in Jan. 2016, someone from the BabySeq study asked her and her husband if they would like to take part. The decision was simple for the couple.
“I didn’t hesitate,” she says. “To me, knowledge is power.”
Using screening tools for parental and marital distress and parent-child bonding, the research evaluated BabySeq families at 3 and 10 months after parents received the sequencing results. The investigators found no significant differences in any of these measures between screened and unscreened families. Ms. Moniz learned that her daughter’s only concerning result was being a carrier for cystic fibrosis. Rather than finding this information anxiety-provoking, Ms. Moniz considered it to be reassuring.
“My mom brain worries about so many things, but at least I know I don’t have to add genetic disease to the list,” she says.
But Ms. Stoll, who wasn’t involved in the BabySeq study, isn’t as convinced. She says that less than 10% of the families approached about the trial ultimately agreed to take part, suggesting potential bias in the selection process. Most participants were white, well-educated, and well-off, making it hard to generalize the study’s results.
What’s more, the standard care involved meeting with a genetic counselor and giving a detailed family history, neither of which is routinely offered to new parents, Ms. Stoll says. These study features leave her unconvinced that healthy newborn genetic screening is beneficial.
“We can’t assume these psychosocial consequences will be true for everyone,” she says.
Follow-up and treatment needed
Traditional newborn screening relies on blood biochemical tests to detect and diagnose metabolic diseases. This approach still outperforms DNA sequencing in trials, says Cynthia Powell, MD, a pediatric geneticist at the University of North Carolina at Chapel Hill, who wasn’t involved with the BabySeq study. Despite the enthusiasm for genomics, this kind of screening won’t replace newborn biochemical screening anytime soon, she says.
“There are some states that have only one geneticist available, so should we really be doing this if we can’t provide the necessary follow-up and treatment for these babies?” she asks.
Still, Dr. Powell says, the BabySeq study helps advance understanding of what the infrastructure needs are for widespread use of DNA sequencing in newborns. She says those needs include appropriate consent processes, access to genetic counselors to discuss testing, and referrals for further testing and treatment in those babies with concerning results.
The BabySeq program will also guide new initiatives, like the pilot program that Genomics England launched in Sept. 2021. As part of that project, the U.K. group intends to look into how practical whole-genome sequencing for newborn screening would be and look at the risks, benefits, and limits of its widespread use.
“For the first time, we’re putting real data into these questions that people have basically just speculated and hypothesized and created narratives about,” Dr. Green says.
But for now, the findings on the psychosocial effects of general newborn genomic screening show that “we should consider genetics to be just one more arrow in our medical quiver.”
A version of this article first appeared on WebMD.com.
Even before their baby is born, parents face some tough questions: Home birth or hospital? Cloth or disposable diapers? Breast, bottle, or both? But advances in genetic sequencing technology mean that parents will soon face yet another choice: whether to sequence their newborn’s DNA for an overview of the baby’s entire genome.
Genetic testing has been used for decades to diagnose conditions even before birth. But DNA sequencing technologies, once expensive and tough to access, are now rapid and cheap enough that doctors could order genomic screening for any infant, regardless of health status.
The possibility has raised many questions about the ethical, legal, and social repercussions of doing so. One of the biggest sticking points of sequencing newborns is the potential psychosocial fallout for families of such wide-scale use of genetic screening.
“There’s a narrative of catastrophic distress,” says Robert Green, MD, a geneticist at Harvard Medical School and lead investigator on the BabySeq study, which is evaluating the medical, social, and economic consequences of newborn genetic screening. The concern is that parents learning that their child carries a gene variant related to cancer or heart disease will become “incredibly anxious and distressed,” he says. “And it’s not an unreasonable speculation.”
But Dr. Green’s team found no evidence of such anxiety in the results from a randomized trial it conducted, published in JAMA Pediatrics. In the meantime, Genomics England announced it would begin a pilot study involving whole-genome sequencing of up to 200,000 babies. The first goal is to identify severe disease that starts in childhood, but the information would also be stored and used to detect drug sensitivities and conditions that come up later in life.
The large U.K. project is a bold move, according to David Amor, PhD, a pediatric geneticist at Murdoch Children’s Research Institute in Australia, who says its time has come. Geneticists have been accused of thinking their field involves unique pitfalls, compared with the rest of medicine, he points out, and that doctors need to protect patients and families from the potential harm genetic testing poses.
“But it is becoming apparent that that’s not really the case,” he says, and “maybe there’s not a whole lot special about genetics – it’s just medicine.”
When a first-draft copy of the human genome was published in 2001, scientists and doctors hailed the start of a new era of precision medicine. Knowing our genome sequence was expected to lead to a better grasp on our individual disease risks. Yet even as technologies advanced, clinical genetics remained focused on diagnosis rather than screening, according to Lilian Downie, a clinical genetics PhD candidate at the University of Melbourne. She calls the difference subtle but important.
Diagnostic genetic testing confirms whether a person has a specific condition, whereas genetic screening tests evaluate someone’s risk of getting an illness. Both approaches use sequencing, but they answer different questions, explains Ms. Downie.
Diagnosing disease versus predicting future illness
Genetic testing is on the upswing for both purposes, whether clinically for diagnosis or through direct-to-consumer screening-oriented services like 23andMe. Scientists began to note that many people carried disease-related genetic variants without having signs of disease. In some cases, a variant that is mathematically linked to a disease simply doesn’t cause it. In other cases, though, even if the gene variant contributes to a disease, not everyone who carries the genetic change will get the condition.
This potential disconnect between having a variant and developing the condition is a big problem, says Katie Stoll, a genetic counselor and executive director of the Genetic Support Foundation in Olympia, WA.
“It’s more complicated than just looking at one gene variant and one outcome,” she says. Without a sure link between the two, this information could unnecessarily entail “some pretty big emotional and financial costs.”
Ms. Stoll and others in the genetics field who share similar concerns are one reason the BabySeq project was first funded back in 2015. Although the overall aim of the initiative is to answer questions about the value of genomic sequencing in newborn screening, the media and scientific attention has focused on the psychosocial impact of healthy newborn sequencing, says Dr. Green. In the study published in JAMA Pediatrics, his group focused on these issues, too.
For that randomized trial, they enrolled 325 families, 257 with healthy babies and 68 whose babies had spent time in neonatal intensive care. Enrolled infants were randomly given standard care alone or standard care with genomic sequencing added on. The genomic sequencing report contained information about the presence of genetic variants associated with disease that start in childhood. Parents also could choose whether to learn about genetic risks for conditions that start in adulthood, such as cancer.
Boston-based Tina Moniz was one of those parents. When her first daughter was born in Jan. 2016, someone from the BabySeq study asked her and her husband if they would like to take part. The decision was simple for the couple.
“I didn’t hesitate,” she says. “To me, knowledge is power.”
Using screening tools for parental and marital distress and parent-child bonding, the research evaluated BabySeq families at 3 and 10 months after parents received the sequencing results. The investigators found no significant differences in any of these measures between screened and unscreened families. Ms. Moniz learned that her daughter’s only concerning result was being a carrier for cystic fibrosis. Rather than finding this information anxiety-provoking, Ms. Moniz considered it to be reassuring.
“My mom brain worries about so many things, but at least I know I don’t have to add genetic disease to the list,” she says.
But Ms. Stoll, who wasn’t involved in the BabySeq study, isn’t as convinced. She says that less than 10% of the families approached about the trial ultimately agreed to take part, suggesting potential bias in the selection process. Most participants were white, well-educated, and well-off, making it hard to generalize the study’s results.
What’s more, the standard care involved meeting with a genetic counselor and giving a detailed family history, neither of which is routinely offered to new parents, Ms. Stoll says. These study features leave her unconvinced that healthy newborn genetic screening is beneficial.
“We can’t assume these psychosocial consequences will be true for everyone,” she says.
Follow-up and treatment needed
Traditional newborn screening relies on blood biochemical tests to detect and diagnose metabolic diseases. This approach still outperforms DNA sequencing in trials, says Cynthia Powell, MD, a pediatric geneticist at the University of North Carolina at Chapel Hill, who wasn’t involved with the BabySeq study. Despite the enthusiasm for genomics, this kind of screening won’t replace newborn biochemical screening anytime soon, she says.
“There are some states that have only one geneticist available, so should we really be doing this if we can’t provide the necessary follow-up and treatment for these babies?” she asks.
Still, Dr. Powell says, the BabySeq study helps advance understanding of what the infrastructure needs are for widespread use of DNA sequencing in newborns. She says those needs include appropriate consent processes, access to genetic counselors to discuss testing, and referrals for further testing and treatment in those babies with concerning results.
The BabySeq program will also guide new initiatives, like the pilot program that Genomics England launched in Sept. 2021. As part of that project, the U.K. group intends to look into how practical whole-genome sequencing for newborn screening would be and look at the risks, benefits, and limits of its widespread use.
“For the first time, we’re putting real data into these questions that people have basically just speculated and hypothesized and created narratives about,” Dr. Green says.
But for now, the findings on the psychosocial effects of general newborn genomic screening show that “we should consider genetics to be just one more arrow in our medical quiver.”
A version of this article first appeared on WebMD.com.
Even before their baby is born, parents face some tough questions: Home birth or hospital? Cloth or disposable diapers? Breast, bottle, or both? But advances in genetic sequencing technology mean that parents will soon face yet another choice: whether to sequence their newborn’s DNA for an overview of the baby’s entire genome.
Genetic testing has been used for decades to diagnose conditions even before birth. But DNA sequencing technologies, once expensive and tough to access, are now rapid and cheap enough that doctors could order genomic screening for any infant, regardless of health status.
The possibility has raised many questions about the ethical, legal, and social repercussions of doing so. One of the biggest sticking points of sequencing newborns is the potential psychosocial fallout for families of such wide-scale use of genetic screening.
“There’s a narrative of catastrophic distress,” says Robert Green, MD, a geneticist at Harvard Medical School and lead investigator on the BabySeq study, which is evaluating the medical, social, and economic consequences of newborn genetic screening. The concern is that parents learning that their child carries a gene variant related to cancer or heart disease will become “incredibly anxious and distressed,” he says. “And it’s not an unreasonable speculation.”
But Dr. Green’s team found no evidence of such anxiety in the results from a randomized trial it conducted, published in JAMA Pediatrics. In the meantime, Genomics England announced it would begin a pilot study involving whole-genome sequencing of up to 200,000 babies. The first goal is to identify severe disease that starts in childhood, but the information would also be stored and used to detect drug sensitivities and conditions that come up later in life.
The large U.K. project is a bold move, according to David Amor, PhD, a pediatric geneticist at Murdoch Children’s Research Institute in Australia, who says its time has come. Geneticists have been accused of thinking their field involves unique pitfalls, compared with the rest of medicine, he points out, and that doctors need to protect patients and families from the potential harm genetic testing poses.
“But it is becoming apparent that that’s not really the case,” he says, and “maybe there’s not a whole lot special about genetics – it’s just medicine.”
When a first-draft copy of the human genome was published in 2001, scientists and doctors hailed the start of a new era of precision medicine. Knowing our genome sequence was expected to lead to a better grasp on our individual disease risks. Yet even as technologies advanced, clinical genetics remained focused on diagnosis rather than screening, according to Lilian Downie, a clinical genetics PhD candidate at the University of Melbourne. She calls the difference subtle but important.
Diagnostic genetic testing confirms whether a person has a specific condition, whereas genetic screening tests evaluate someone’s risk of getting an illness. Both approaches use sequencing, but they answer different questions, explains Ms. Downie.
Diagnosing disease versus predicting future illness
Genetic testing is on the upswing for both purposes, whether clinically for diagnosis or through direct-to-consumer screening-oriented services like 23andMe. Scientists began to note that many people carried disease-related genetic variants without having signs of disease. In some cases, a variant that is mathematically linked to a disease simply doesn’t cause it. In other cases, though, even if the gene variant contributes to a disease, not everyone who carries the genetic change will get the condition.
This potential disconnect between having a variant and developing the condition is a big problem, says Katie Stoll, a genetic counselor and executive director of the Genetic Support Foundation in Olympia, WA.
“It’s more complicated than just looking at one gene variant and one outcome,” she says. Without a sure link between the two, this information could unnecessarily entail “some pretty big emotional and financial costs.”
Ms. Stoll and others in the genetics field who share similar concerns are one reason the BabySeq project was first funded back in 2015. Although the overall aim of the initiative is to answer questions about the value of genomic sequencing in newborn screening, the media and scientific attention has focused on the psychosocial impact of healthy newborn sequencing, says Dr. Green. In the study published in JAMA Pediatrics, his group focused on these issues, too.
For that randomized trial, they enrolled 325 families, 257 with healthy babies and 68 whose babies had spent time in neonatal intensive care. Enrolled infants were randomly given standard care alone or standard care with genomic sequencing added on. The genomic sequencing report contained information about the presence of genetic variants associated with disease that start in childhood. Parents also could choose whether to learn about genetic risks for conditions that start in adulthood, such as cancer.
Boston-based Tina Moniz was one of those parents. When her first daughter was born in Jan. 2016, someone from the BabySeq study asked her and her husband if they would like to take part. The decision was simple for the couple.
“I didn’t hesitate,” she says. “To me, knowledge is power.”
Using screening tools for parental and marital distress and parent-child bonding, the research evaluated BabySeq families at 3 and 10 months after parents received the sequencing results. The investigators found no significant differences in any of these measures between screened and unscreened families. Ms. Moniz learned that her daughter’s only concerning result was being a carrier for cystic fibrosis. Rather than finding this information anxiety-provoking, Ms. Moniz considered it to be reassuring.
“My mom brain worries about so many things, but at least I know I don’t have to add genetic disease to the list,” she says.
But Ms. Stoll, who wasn’t involved in the BabySeq study, isn’t as convinced. She says that less than 10% of the families approached about the trial ultimately agreed to take part, suggesting potential bias in the selection process. Most participants were white, well-educated, and well-off, making it hard to generalize the study’s results.
What’s more, the standard care involved meeting with a genetic counselor and giving a detailed family history, neither of which is routinely offered to new parents, Ms. Stoll says. These study features leave her unconvinced that healthy newborn genetic screening is beneficial.
“We can’t assume these psychosocial consequences will be true for everyone,” she says.
Follow-up and treatment needed
Traditional newborn screening relies on blood biochemical tests to detect and diagnose metabolic diseases. This approach still outperforms DNA sequencing in trials, says Cynthia Powell, MD, a pediatric geneticist at the University of North Carolina at Chapel Hill, who wasn’t involved with the BabySeq study. Despite the enthusiasm for genomics, this kind of screening won’t replace newborn biochemical screening anytime soon, she says.
“There are some states that have only one geneticist available, so should we really be doing this if we can’t provide the necessary follow-up and treatment for these babies?” she asks.
Still, Dr. Powell says, the BabySeq study helps advance understanding of what the infrastructure needs are for widespread use of DNA sequencing in newborns. She says those needs include appropriate consent processes, access to genetic counselors to discuss testing, and referrals for further testing and treatment in those babies with concerning results.
The BabySeq program will also guide new initiatives, like the pilot program that Genomics England launched in Sept. 2021. As part of that project, the U.K. group intends to look into how practical whole-genome sequencing for newborn screening would be and look at the risks, benefits, and limits of its widespread use.
“For the first time, we’re putting real data into these questions that people have basically just speculated and hypothesized and created narratives about,” Dr. Green says.
But for now, the findings on the psychosocial effects of general newborn genomic screening show that “we should consider genetics to be just one more arrow in our medical quiver.”
A version of this article first appeared on WebMD.com.
Expert shares top five atopic dermatitis–related questions he fields
Will my child outgrow the eczema?
That is perhaps the No. 1 atopic dermatitis–related question that Lawrence F. Eichenfield, MD, fields from parents in his role as chief of pediatric and adolescent dermatology at Rady’s Children’s Hospital, San Diego.
The answer “is pretty tricky,” he said during MedscapeLive’s annual Las Vegas Dermatology Seminar. “We used to say, ‘yeah, your kid will probably outgrow the disease,’ but we now have good data that show there are variable courses.”
Using data from the birth study cohort known as the Avon Longitudinal Study of Parents and Children, researchers in the United Kingdom investigated the existence of different longitudinal phenotypes of AD among 9,894 children. They found that 58% of the children in the cohort were unaffected or had transient AD, while 12.9% had early-onset/early-resolving AD. The remaining AD phenotypes consisted of 7%-8% patients each (early-onset persistent, early-onset late-resolving, mid-onset resolving, and late-onset resolving).
“There have been several studies that looked at the natural course of AD,” said Dr. Eichenfield, distinguished professor of dermatology and pediatrics at the University of California, San Diego. “A cohort study from Thailand showed that 50% of patients with childhood AD lost their AD diagnosis about 5 years into it, while there was an increase in allergic rhino-conjunctivitis and asthma, similar to what’s been seen in atopic march studies,” he noted.
A separate group of investigators analyzed records from The Health Improvement Network in the UK to determine the prevalence of AD among more than 8 million patients seen in primary care between 1994 and 2013. They found that the cumulative lifetime prevalence of atopic eczema was 9.9% and the highest rates of active disease were among children and older adults. “The takeaway was markedly inconsistent in terms of whether AD went away over time or increased over time, so it’s really not especially helpful prevalence data,” Dr. Eichenfield said. “Overall, you have a high prevalence in the first years of life, it decreases, and it may increase again when people are 60 years and older. Whether that’s truly AD or xerotic eczema isn’t known in this data set.”
A separate meta-analysis of 17 studies reported that 26% of adults with AD said they had adult-onset disease, which is characterized by more atopy, more foot dermatitis, and less flexural involvement.
Dr. Eichenfield tells parents, “there’s a really good chance (depending on disease severity) that 60% to 70% of children will outgrow their eczema or most of it,” he said. “If you ask me when, I won’t tell you. The important thing is to treat it to minimize its impact. We want minimal rash, minimal itch, and minimal sleep disturbance. Sometimes I say, ‘that might improve the chance of the eczema getting better over time.’ ”
Following are four other common questions parents and patients ask him:
Can we figure out the allergies causing the eczema? “This is probably one of the most unnerving questions I get asked,” he said. “It’s a loaded question. My answer is that allergies are intertwined with AD. Searching for the secret allergy causing the atopic dermatitis is rarely successful.” Sensitization is much more common with AD, he added, meaning specific IgE testing, whether it be blood testing or skin prick testing. “The more severe your eczema is, the more chance you’ll have of real food allergy,” he said. “About 15% of milder eczema patients will have at least one food allergy, but when you get to the more moderate to severe cases, about 40% will have a true food allergy.”
Food reactions may not cause eczema, though. Food reactions can cause urticaria, angioedema, eczematous dermatitis, allergic contact dermatitis, contact urticaria, and respiratory findings. According to National Institutes of Health guidelines for food allergy, skin prick tests and serum IgE tests are recommended to assist in identification of foods that may be provoking IgE-mediated food reactions, but are not diagnostic of food allergy.
“There’s a huge literature showing that there’s a lot of food allergy testing that’s just not helpful,” he said. In one study, 89% of food challenges administered in patients who were listed as being allergic based on skin prick tests or serum IgE tests did not have a true food allergy.
“Empiric elimination diets aren’t especially useful. However, we occasionally see children who do have AD exacerbated by food allergies in the first year of life,” he said. NIH guidelines suggest that children younger than 5 years of age with moderate to severe AD be considered for food allergy evaluation for milk, egg, peanut, wheat, and soy, if at least one of the following conditions is met: the child has persistent AD in spite of optimized management and topical therapy, and/or the child has a reliable history of an immediate reaction after ingestion of a specific food.
“We do know that there are high rates of comorbid allergic processes, besides food allergy, associated with atopic dermatitis, including allergic rhinitis and asthma both in children and adults,” Dr. Eichenfield said. “I do discuss allergy triggers and their importance in the life of the individual, though not necessarily as factors in AD. There are a variety of environmental allergens and/or environmental triggers that can significantly impact AD. Recently, we have seen studies discussing air pollution and wildfires as exacerbators of AD.”
How should I bathe and moisturize? There are no standard guidelines for the frequency, type, or duration of bathing in patients with AD, he said, though in more severe disease, frequent bathing can be helpful along with standard anti-inflammatory topical medicines. “I keep my general recommendations vague,” Dr. Eichenfield said. “I do explain that we don’t want to use harsh soaps; we want to be gentle in our washing. I usually recommend daily to every other day bathing. It’s important to pat the skin dry and then apply a moisturizer. Applying a moisturizer 2-3 minutes after bathing is important and limited significant cleanser use can be helpful.”
Moisturizers and emollients are a standard of care in U.S. guidelines published in 2013 and 2014, and international guidelines, and are steroid-sparing and useful for both prevention and maintenance. “I tell parents and patients that there is no reason to avoid bathing because of AD as long as you moisturize after,” he said.
Do I have to use topical [name of drug]? “I try to explain that there is skin barrier dysfunction that stimulates the inflammatory milieu, and that inflammation in the skin or blood in AD negatively impacts skin barrier function,” Dr. Eichenfield said. “I explain that if inflammation doesn’t get better with good skin care, moisturizers, and avoidance of triggers, we need anti-inflammatory medication. Then we discuss what the options are, the significant variation in strengths of topical corticosteroids, and topical nonsteroid options.”
When he counsels parents and patients on the use of topical corticosteroids, he tells them that cortisone is a naturally-occurring metabolite, and that “we can work together to let you know how much medicine to use, and how a safe amount is a powerful tool to fix the eczema.” He often says that topical steroids “are like hammers. We have tiny hammers, like over-the-counter hydrocortisone, and sledgehammers like clobetasol. We also have ‘screwdrivers’ and ‘pliers’ with nonsteroidal topical calcineurin and PDE-4 inhibitors, which are especially useful for maintenance therapy. Topical ruxolitinib is a new medicine that we may use for patients as well. The label includes discussion of side effects from oral JAK inhibitors as well as from the drug development program, so it takes some time to talk through.”
Is it time for a stronger systemic medicine? Any conversation about this topic should support the concept that the AD is multifactorial. “We have the rash of eczema,” he said. “We have the itch. We have impact on sleep disturbance. We have the comorbidities. We have other physical changes, which can happen with bacterial infections and other immune system or cardiovascular changes. We have the impact on quality of life and impact on school and work. When we recognize that if patients have significant enough disease that it is not getting better with topicals and is having a negative impact on their lives, we can move our discussion to systemic therapy.”
When counseling patients about systemic therapy, Dr. Eichenfield will conduct a body surface area assessment and document how bad the itch is. “But I’m not just recording the information; I’m bringing it out in the room,” he said. “I’ll do a BSA assessment and say, for example, ‘oh, you have 32% of your body involved with eczema.’ I ask about sleep disturbance, to get the answer ‘out in the room.’ ” He also asks questions such as: “When was the last time your skin was last totally clear? Are there activities that you or your family don’t do because of your eczema, or that you’re living your life around it? Is there anxiety or depression?” Documenting both the impact on quality of life and the severity of disease “makes it easier to discuss systemic therapy,” Dr. Eichenfield said. “Meanwhile, as the provider, I am trying to figure out if the patient should ‘go into the topical therapy bucket’ or into the ‘systemic therapy bucket.’ ”
Counseling about systemic therapy includes shared decision-making regarding the choice of biologics versus oral JAK inhibitors versus traditional systemic agent or phototherapy. Factors to consider in the decision making include patient age, sex, severity, comorbidities, prior therapy, risk aversion, duration, medication access, and desired efficacy. “Evolving therapies can change the conversation, the questions, and the outcomes, but the overarching desired outcome is long-term disease control, minimal eczematous rash, minimal pruritus, and minimal sleep disturbance,” he said.
Dr. Eichenfield disclosed that he has served as a consultant to or investigator for AbbVie; Almirall; Arcutis; Arena; Asana; Termagant; Dermira; Forte Biosciences; Galderma Laboratories; Glenmark/Chinos; Incyte; Kyowa Kirin; Leo Pharma; Eli Lilly and Company; Novartis; Ortho Dermatology; Otsuka; Pfizer; Freestone; Regeneron, and Sanofi Genzyme.
MedscapeLive and this news organization are owned by the same parent company.
Will my child outgrow the eczema?
That is perhaps the No. 1 atopic dermatitis–related question that Lawrence F. Eichenfield, MD, fields from parents in his role as chief of pediatric and adolescent dermatology at Rady’s Children’s Hospital, San Diego.
The answer “is pretty tricky,” he said during MedscapeLive’s annual Las Vegas Dermatology Seminar. “We used to say, ‘yeah, your kid will probably outgrow the disease,’ but we now have good data that show there are variable courses.”
Using data from the birth study cohort known as the Avon Longitudinal Study of Parents and Children, researchers in the United Kingdom investigated the existence of different longitudinal phenotypes of AD among 9,894 children. They found that 58% of the children in the cohort were unaffected or had transient AD, while 12.9% had early-onset/early-resolving AD. The remaining AD phenotypes consisted of 7%-8% patients each (early-onset persistent, early-onset late-resolving, mid-onset resolving, and late-onset resolving).
“There have been several studies that looked at the natural course of AD,” said Dr. Eichenfield, distinguished professor of dermatology and pediatrics at the University of California, San Diego. “A cohort study from Thailand showed that 50% of patients with childhood AD lost their AD diagnosis about 5 years into it, while there was an increase in allergic rhino-conjunctivitis and asthma, similar to what’s been seen in atopic march studies,” he noted.
A separate group of investigators analyzed records from The Health Improvement Network in the UK to determine the prevalence of AD among more than 8 million patients seen in primary care between 1994 and 2013. They found that the cumulative lifetime prevalence of atopic eczema was 9.9% and the highest rates of active disease were among children and older adults. “The takeaway was markedly inconsistent in terms of whether AD went away over time or increased over time, so it’s really not especially helpful prevalence data,” Dr. Eichenfield said. “Overall, you have a high prevalence in the first years of life, it decreases, and it may increase again when people are 60 years and older. Whether that’s truly AD or xerotic eczema isn’t known in this data set.”
A separate meta-analysis of 17 studies reported that 26% of adults with AD said they had adult-onset disease, which is characterized by more atopy, more foot dermatitis, and less flexural involvement.
Dr. Eichenfield tells parents, “there’s a really good chance (depending on disease severity) that 60% to 70% of children will outgrow their eczema or most of it,” he said. “If you ask me when, I won’t tell you. The important thing is to treat it to minimize its impact. We want minimal rash, minimal itch, and minimal sleep disturbance. Sometimes I say, ‘that might improve the chance of the eczema getting better over time.’ ”
Following are four other common questions parents and patients ask him:
Can we figure out the allergies causing the eczema? “This is probably one of the most unnerving questions I get asked,” he said. “It’s a loaded question. My answer is that allergies are intertwined with AD. Searching for the secret allergy causing the atopic dermatitis is rarely successful.” Sensitization is much more common with AD, he added, meaning specific IgE testing, whether it be blood testing or skin prick testing. “The more severe your eczema is, the more chance you’ll have of real food allergy,” he said. “About 15% of milder eczema patients will have at least one food allergy, but when you get to the more moderate to severe cases, about 40% will have a true food allergy.”
Food reactions may not cause eczema, though. Food reactions can cause urticaria, angioedema, eczematous dermatitis, allergic contact dermatitis, contact urticaria, and respiratory findings. According to National Institutes of Health guidelines for food allergy, skin prick tests and serum IgE tests are recommended to assist in identification of foods that may be provoking IgE-mediated food reactions, but are not diagnostic of food allergy.
“There’s a huge literature showing that there’s a lot of food allergy testing that’s just not helpful,” he said. In one study, 89% of food challenges administered in patients who were listed as being allergic based on skin prick tests or serum IgE tests did not have a true food allergy.
“Empiric elimination diets aren’t especially useful. However, we occasionally see children who do have AD exacerbated by food allergies in the first year of life,” he said. NIH guidelines suggest that children younger than 5 years of age with moderate to severe AD be considered for food allergy evaluation for milk, egg, peanut, wheat, and soy, if at least one of the following conditions is met: the child has persistent AD in spite of optimized management and topical therapy, and/or the child has a reliable history of an immediate reaction after ingestion of a specific food.
“We do know that there are high rates of comorbid allergic processes, besides food allergy, associated with atopic dermatitis, including allergic rhinitis and asthma both in children and adults,” Dr. Eichenfield said. “I do discuss allergy triggers and their importance in the life of the individual, though not necessarily as factors in AD. There are a variety of environmental allergens and/or environmental triggers that can significantly impact AD. Recently, we have seen studies discussing air pollution and wildfires as exacerbators of AD.”
How should I bathe and moisturize? There are no standard guidelines for the frequency, type, or duration of bathing in patients with AD, he said, though in more severe disease, frequent bathing can be helpful along with standard anti-inflammatory topical medicines. “I keep my general recommendations vague,” Dr. Eichenfield said. “I do explain that we don’t want to use harsh soaps; we want to be gentle in our washing. I usually recommend daily to every other day bathing. It’s important to pat the skin dry and then apply a moisturizer. Applying a moisturizer 2-3 minutes after bathing is important and limited significant cleanser use can be helpful.”
Moisturizers and emollients are a standard of care in U.S. guidelines published in 2013 and 2014, and international guidelines, and are steroid-sparing and useful for both prevention and maintenance. “I tell parents and patients that there is no reason to avoid bathing because of AD as long as you moisturize after,” he said.
Do I have to use topical [name of drug]? “I try to explain that there is skin barrier dysfunction that stimulates the inflammatory milieu, and that inflammation in the skin or blood in AD negatively impacts skin barrier function,” Dr. Eichenfield said. “I explain that if inflammation doesn’t get better with good skin care, moisturizers, and avoidance of triggers, we need anti-inflammatory medication. Then we discuss what the options are, the significant variation in strengths of topical corticosteroids, and topical nonsteroid options.”
When he counsels parents and patients on the use of topical corticosteroids, he tells them that cortisone is a naturally-occurring metabolite, and that “we can work together to let you know how much medicine to use, and how a safe amount is a powerful tool to fix the eczema.” He often says that topical steroids “are like hammers. We have tiny hammers, like over-the-counter hydrocortisone, and sledgehammers like clobetasol. We also have ‘screwdrivers’ and ‘pliers’ with nonsteroidal topical calcineurin and PDE-4 inhibitors, which are especially useful for maintenance therapy. Topical ruxolitinib is a new medicine that we may use for patients as well. The label includes discussion of side effects from oral JAK inhibitors as well as from the drug development program, so it takes some time to talk through.”
Is it time for a stronger systemic medicine? Any conversation about this topic should support the concept that the AD is multifactorial. “We have the rash of eczema,” he said. “We have the itch. We have impact on sleep disturbance. We have the comorbidities. We have other physical changes, which can happen with bacterial infections and other immune system or cardiovascular changes. We have the impact on quality of life and impact on school and work. When we recognize that if patients have significant enough disease that it is not getting better with topicals and is having a negative impact on their lives, we can move our discussion to systemic therapy.”
When counseling patients about systemic therapy, Dr. Eichenfield will conduct a body surface area assessment and document how bad the itch is. “But I’m not just recording the information; I’m bringing it out in the room,” he said. “I’ll do a BSA assessment and say, for example, ‘oh, you have 32% of your body involved with eczema.’ I ask about sleep disturbance, to get the answer ‘out in the room.’ ” He also asks questions such as: “When was the last time your skin was last totally clear? Are there activities that you or your family don’t do because of your eczema, or that you’re living your life around it? Is there anxiety or depression?” Documenting both the impact on quality of life and the severity of disease “makes it easier to discuss systemic therapy,” Dr. Eichenfield said. “Meanwhile, as the provider, I am trying to figure out if the patient should ‘go into the topical therapy bucket’ or into the ‘systemic therapy bucket.’ ”
Counseling about systemic therapy includes shared decision-making regarding the choice of biologics versus oral JAK inhibitors versus traditional systemic agent or phototherapy. Factors to consider in the decision making include patient age, sex, severity, comorbidities, prior therapy, risk aversion, duration, medication access, and desired efficacy. “Evolving therapies can change the conversation, the questions, and the outcomes, but the overarching desired outcome is long-term disease control, minimal eczematous rash, minimal pruritus, and minimal sleep disturbance,” he said.
Dr. Eichenfield disclosed that he has served as a consultant to or investigator for AbbVie; Almirall; Arcutis; Arena; Asana; Termagant; Dermira; Forte Biosciences; Galderma Laboratories; Glenmark/Chinos; Incyte; Kyowa Kirin; Leo Pharma; Eli Lilly and Company; Novartis; Ortho Dermatology; Otsuka; Pfizer; Freestone; Regeneron, and Sanofi Genzyme.
MedscapeLive and this news organization are owned by the same parent company.
Will my child outgrow the eczema?
That is perhaps the No. 1 atopic dermatitis–related question that Lawrence F. Eichenfield, MD, fields from parents in his role as chief of pediatric and adolescent dermatology at Rady’s Children’s Hospital, San Diego.
The answer “is pretty tricky,” he said during MedscapeLive’s annual Las Vegas Dermatology Seminar. “We used to say, ‘yeah, your kid will probably outgrow the disease,’ but we now have good data that show there are variable courses.”
Using data from the birth study cohort known as the Avon Longitudinal Study of Parents and Children, researchers in the United Kingdom investigated the existence of different longitudinal phenotypes of AD among 9,894 children. They found that 58% of the children in the cohort were unaffected or had transient AD, while 12.9% had early-onset/early-resolving AD. The remaining AD phenotypes consisted of 7%-8% patients each (early-onset persistent, early-onset late-resolving, mid-onset resolving, and late-onset resolving).
“There have been several studies that looked at the natural course of AD,” said Dr. Eichenfield, distinguished professor of dermatology and pediatrics at the University of California, San Diego. “A cohort study from Thailand showed that 50% of patients with childhood AD lost their AD diagnosis about 5 years into it, while there was an increase in allergic rhino-conjunctivitis and asthma, similar to what’s been seen in atopic march studies,” he noted.
A separate group of investigators analyzed records from The Health Improvement Network in the UK to determine the prevalence of AD among more than 8 million patients seen in primary care between 1994 and 2013. They found that the cumulative lifetime prevalence of atopic eczema was 9.9% and the highest rates of active disease were among children and older adults. “The takeaway was markedly inconsistent in terms of whether AD went away over time or increased over time, so it’s really not especially helpful prevalence data,” Dr. Eichenfield said. “Overall, you have a high prevalence in the first years of life, it decreases, and it may increase again when people are 60 years and older. Whether that’s truly AD or xerotic eczema isn’t known in this data set.”
A separate meta-analysis of 17 studies reported that 26% of adults with AD said they had adult-onset disease, which is characterized by more atopy, more foot dermatitis, and less flexural involvement.
Dr. Eichenfield tells parents, “there’s a really good chance (depending on disease severity) that 60% to 70% of children will outgrow their eczema or most of it,” he said. “If you ask me when, I won’t tell you. The important thing is to treat it to minimize its impact. We want minimal rash, minimal itch, and minimal sleep disturbance. Sometimes I say, ‘that might improve the chance of the eczema getting better over time.’ ”
Following are four other common questions parents and patients ask him:
Can we figure out the allergies causing the eczema? “This is probably one of the most unnerving questions I get asked,” he said. “It’s a loaded question. My answer is that allergies are intertwined with AD. Searching for the secret allergy causing the atopic dermatitis is rarely successful.” Sensitization is much more common with AD, he added, meaning specific IgE testing, whether it be blood testing or skin prick testing. “The more severe your eczema is, the more chance you’ll have of real food allergy,” he said. “About 15% of milder eczema patients will have at least one food allergy, but when you get to the more moderate to severe cases, about 40% will have a true food allergy.”
Food reactions may not cause eczema, though. Food reactions can cause urticaria, angioedema, eczematous dermatitis, allergic contact dermatitis, contact urticaria, and respiratory findings. According to National Institutes of Health guidelines for food allergy, skin prick tests and serum IgE tests are recommended to assist in identification of foods that may be provoking IgE-mediated food reactions, but are not diagnostic of food allergy.
“There’s a huge literature showing that there’s a lot of food allergy testing that’s just not helpful,” he said. In one study, 89% of food challenges administered in patients who were listed as being allergic based on skin prick tests or serum IgE tests did not have a true food allergy.
“Empiric elimination diets aren’t especially useful. However, we occasionally see children who do have AD exacerbated by food allergies in the first year of life,” he said. NIH guidelines suggest that children younger than 5 years of age with moderate to severe AD be considered for food allergy evaluation for milk, egg, peanut, wheat, and soy, if at least one of the following conditions is met: the child has persistent AD in spite of optimized management and topical therapy, and/or the child has a reliable history of an immediate reaction after ingestion of a specific food.
“We do know that there are high rates of comorbid allergic processes, besides food allergy, associated with atopic dermatitis, including allergic rhinitis and asthma both in children and adults,” Dr. Eichenfield said. “I do discuss allergy triggers and their importance in the life of the individual, though not necessarily as factors in AD. There are a variety of environmental allergens and/or environmental triggers that can significantly impact AD. Recently, we have seen studies discussing air pollution and wildfires as exacerbators of AD.”
How should I bathe and moisturize? There are no standard guidelines for the frequency, type, or duration of bathing in patients with AD, he said, though in more severe disease, frequent bathing can be helpful along with standard anti-inflammatory topical medicines. “I keep my general recommendations vague,” Dr. Eichenfield said. “I do explain that we don’t want to use harsh soaps; we want to be gentle in our washing. I usually recommend daily to every other day bathing. It’s important to pat the skin dry and then apply a moisturizer. Applying a moisturizer 2-3 minutes after bathing is important and limited significant cleanser use can be helpful.”
Moisturizers and emollients are a standard of care in U.S. guidelines published in 2013 and 2014, and international guidelines, and are steroid-sparing and useful for both prevention and maintenance. “I tell parents and patients that there is no reason to avoid bathing because of AD as long as you moisturize after,” he said.
Do I have to use topical [name of drug]? “I try to explain that there is skin barrier dysfunction that stimulates the inflammatory milieu, and that inflammation in the skin or blood in AD negatively impacts skin barrier function,” Dr. Eichenfield said. “I explain that if inflammation doesn’t get better with good skin care, moisturizers, and avoidance of triggers, we need anti-inflammatory medication. Then we discuss what the options are, the significant variation in strengths of topical corticosteroids, and topical nonsteroid options.”
When he counsels parents and patients on the use of topical corticosteroids, he tells them that cortisone is a naturally-occurring metabolite, and that “we can work together to let you know how much medicine to use, and how a safe amount is a powerful tool to fix the eczema.” He often says that topical steroids “are like hammers. We have tiny hammers, like over-the-counter hydrocortisone, and sledgehammers like clobetasol. We also have ‘screwdrivers’ and ‘pliers’ with nonsteroidal topical calcineurin and PDE-4 inhibitors, which are especially useful for maintenance therapy. Topical ruxolitinib is a new medicine that we may use for patients as well. The label includes discussion of side effects from oral JAK inhibitors as well as from the drug development program, so it takes some time to talk through.”
Is it time for a stronger systemic medicine? Any conversation about this topic should support the concept that the AD is multifactorial. “We have the rash of eczema,” he said. “We have the itch. We have impact on sleep disturbance. We have the comorbidities. We have other physical changes, which can happen with bacterial infections and other immune system or cardiovascular changes. We have the impact on quality of life and impact on school and work. When we recognize that if patients have significant enough disease that it is not getting better with topicals and is having a negative impact on their lives, we can move our discussion to systemic therapy.”
When counseling patients about systemic therapy, Dr. Eichenfield will conduct a body surface area assessment and document how bad the itch is. “But I’m not just recording the information; I’m bringing it out in the room,” he said. “I’ll do a BSA assessment and say, for example, ‘oh, you have 32% of your body involved with eczema.’ I ask about sleep disturbance, to get the answer ‘out in the room.’ ” He also asks questions such as: “When was the last time your skin was last totally clear? Are there activities that you or your family don’t do because of your eczema, or that you’re living your life around it? Is there anxiety or depression?” Documenting both the impact on quality of life and the severity of disease “makes it easier to discuss systemic therapy,” Dr. Eichenfield said. “Meanwhile, as the provider, I am trying to figure out if the patient should ‘go into the topical therapy bucket’ or into the ‘systemic therapy bucket.’ ”
Counseling about systemic therapy includes shared decision-making regarding the choice of biologics versus oral JAK inhibitors versus traditional systemic agent or phototherapy. Factors to consider in the decision making include patient age, sex, severity, comorbidities, prior therapy, risk aversion, duration, medication access, and desired efficacy. “Evolving therapies can change the conversation, the questions, and the outcomes, but the overarching desired outcome is long-term disease control, minimal eczematous rash, minimal pruritus, and minimal sleep disturbance,” he said.
Dr. Eichenfield disclosed that he has served as a consultant to or investigator for AbbVie; Almirall; Arcutis; Arena; Asana; Termagant; Dermira; Forte Biosciences; Galderma Laboratories; Glenmark/Chinos; Incyte; Kyowa Kirin; Leo Pharma; Eli Lilly and Company; Novartis; Ortho Dermatology; Otsuka; Pfizer; Freestone; Regeneron, and Sanofi Genzyme.
MedscapeLive and this news organization are owned by the same parent company.
FROM THE MEDSCAPELIVE LAS VEGAS DERMATOLOGY SEMINAR
Retinopathy risk in children higher in T2D than T1D
Children diagnosed with type 2 diabetes (T2D) appear significantly more likely to develop retinopathy and other ocular complications over time than children who are diagnosed with type 1 diabetes (T1D), researchers report.
Among a population-based cohort of children (defined as younger than 22 years), the risk of diabetic retinopathy was 88% greater in those with T2D than T1D within the first 15 years of disease diagnosis.
“The purpose of this study was to assess the risk of developing diabetes-associated ocular complications among a population-based cohort of children diagnosed with either T1D or T2D during a 50-year period,” lead author Patricia Bai, BA, of Mayo Clinic, Phoenix, and colleagues reported in JAMA Ophthalmology.
The researchers retrospectively reviewed medical records from all residents of Olmsted County, Minn., from 1970 to 2019. The study cohort included 606 children with a confirmed a diagnosis of T1D or T2D, 525 (87%) of whom had at least one ocular examination.
The mean age at diabetes diagnosis was 12 years (standard deviation, 5.4 years); most participants were White (95.7% in 1990), and half (50%) were male. Diabetes-associated ocular complications occurred in 31.9% and 26.6% of children with T1D and T2D, respectively.
The hazard ratios illustrating the risk between T2D and T1D rates were 1.88 (95% confidence interval, 1.13-3.12; P = .02) for any diabetic retinopathy, 2.33 (95% CI, 0.99-5.50; P = .048) for proliferative diabetic retinopathy, 1.49 (95% CI, 0.46-4.89; P = .50) for diabetic macular edema, 2.43 (95% CI, 0.54-11.07; P = .24) for a visually significant cataract, and 4.06 (95% CI, 1.34-12.33; P = .007) for requiring pars plana vitrectomy within the first 15 years of diagnosis.
These results suggest that earlier surveillance and intervention may help prevent vision-threatening complications, the researchers explained.
“After adjusting for race using self-identified categories of White or not White, the adjusted HR of developing any retinopathy was 1.63 (95% CI, 0.96-2.79; P = .07), and the adjusted HR of developing proliferative diabetic retinopathy was 2.02 (95% CI, 0.79-5.16; P = .14)” in T2D versus T1D patients, the researchers wrote.
“We would expect the reported rate of type 2 diabetes to be potentially underestimated in our study cohort,” Ms. Bai commented in an interview. “Race has been suggested to be a surrogate for other social determinants of health, such as lower rates of optimal follow-up care received by racial and ethnic minorities, which could influence subsequent retinopathy rates.”
Understanding retinopathy outcomes in youth
In an accompanying editorial, Jennifer K. Sun, MD, MPH, from Harvard Medical School, Boston, wrote that the present study indicates the natural history of retinopathy may differ between patients with T1D and T2D.
While the pathophysiology of diabetic retinopathy in T1D and T2D appears similar, other patient-related factors such as lipid profiles, the presence of hypertension, and body mass index may differ between the two disease states.
She wrote that “there is a particular need to document retinopathy outcomes and risk factors for advanced disease in youth with T2D, for whom there is a paucity of information.”
Ms. Bai and colleagues acknowledged that a key limitation of the study was the retrospective design. As a result, irregular follow-up and incomplete data may limit the applicability of the findings.
“Some children with milder forms of diabetes may have eluded detection, a limitation that is more likely to affect T2D, which may exist undetected for years before a diagnosis,” Bai explained.
Dr. Sun recommended that further epidemiologic studies are needed to help optimize guidelines for screening and follow-up for young people diagnosed with diabetes. “Such efforts may potentially lead to increased understanding of the mechanistic differences between pathology in T1D versus T2D,” she concluded.
This study used the resources of the Rochester Epidemiology Project (REP) medical records linkage system, which is supported by grant funding from the National Institute on Aging, the Mayo Clinic Research Committee, and by fees paid annually by REP users. The study authors disclosed no conflicts of interest.
Children diagnosed with type 2 diabetes (T2D) appear significantly more likely to develop retinopathy and other ocular complications over time than children who are diagnosed with type 1 diabetes (T1D), researchers report.
Among a population-based cohort of children (defined as younger than 22 years), the risk of diabetic retinopathy was 88% greater in those with T2D than T1D within the first 15 years of disease diagnosis.
“The purpose of this study was to assess the risk of developing diabetes-associated ocular complications among a population-based cohort of children diagnosed with either T1D or T2D during a 50-year period,” lead author Patricia Bai, BA, of Mayo Clinic, Phoenix, and colleagues reported in JAMA Ophthalmology.
The researchers retrospectively reviewed medical records from all residents of Olmsted County, Minn., from 1970 to 2019. The study cohort included 606 children with a confirmed a diagnosis of T1D or T2D, 525 (87%) of whom had at least one ocular examination.
The mean age at diabetes diagnosis was 12 years (standard deviation, 5.4 years); most participants were White (95.7% in 1990), and half (50%) were male. Diabetes-associated ocular complications occurred in 31.9% and 26.6% of children with T1D and T2D, respectively.
The hazard ratios illustrating the risk between T2D and T1D rates were 1.88 (95% confidence interval, 1.13-3.12; P = .02) for any diabetic retinopathy, 2.33 (95% CI, 0.99-5.50; P = .048) for proliferative diabetic retinopathy, 1.49 (95% CI, 0.46-4.89; P = .50) for diabetic macular edema, 2.43 (95% CI, 0.54-11.07; P = .24) for a visually significant cataract, and 4.06 (95% CI, 1.34-12.33; P = .007) for requiring pars plana vitrectomy within the first 15 years of diagnosis.
These results suggest that earlier surveillance and intervention may help prevent vision-threatening complications, the researchers explained.
“After adjusting for race using self-identified categories of White or not White, the adjusted HR of developing any retinopathy was 1.63 (95% CI, 0.96-2.79; P = .07), and the adjusted HR of developing proliferative diabetic retinopathy was 2.02 (95% CI, 0.79-5.16; P = .14)” in T2D versus T1D patients, the researchers wrote.
“We would expect the reported rate of type 2 diabetes to be potentially underestimated in our study cohort,” Ms. Bai commented in an interview. “Race has been suggested to be a surrogate for other social determinants of health, such as lower rates of optimal follow-up care received by racial and ethnic minorities, which could influence subsequent retinopathy rates.”
Understanding retinopathy outcomes in youth
In an accompanying editorial, Jennifer K. Sun, MD, MPH, from Harvard Medical School, Boston, wrote that the present study indicates the natural history of retinopathy may differ between patients with T1D and T2D.
While the pathophysiology of diabetic retinopathy in T1D and T2D appears similar, other patient-related factors such as lipid profiles, the presence of hypertension, and body mass index may differ between the two disease states.
She wrote that “there is a particular need to document retinopathy outcomes and risk factors for advanced disease in youth with T2D, for whom there is a paucity of information.”
Ms. Bai and colleagues acknowledged that a key limitation of the study was the retrospective design. As a result, irregular follow-up and incomplete data may limit the applicability of the findings.
“Some children with milder forms of diabetes may have eluded detection, a limitation that is more likely to affect T2D, which may exist undetected for years before a diagnosis,” Bai explained.
Dr. Sun recommended that further epidemiologic studies are needed to help optimize guidelines for screening and follow-up for young people diagnosed with diabetes. “Such efforts may potentially lead to increased understanding of the mechanistic differences between pathology in T1D versus T2D,” she concluded.
This study used the resources of the Rochester Epidemiology Project (REP) medical records linkage system, which is supported by grant funding from the National Institute on Aging, the Mayo Clinic Research Committee, and by fees paid annually by REP users. The study authors disclosed no conflicts of interest.
Children diagnosed with type 2 diabetes (T2D) appear significantly more likely to develop retinopathy and other ocular complications over time than children who are diagnosed with type 1 diabetes (T1D), researchers report.
Among a population-based cohort of children (defined as younger than 22 years), the risk of diabetic retinopathy was 88% greater in those with T2D than T1D within the first 15 years of disease diagnosis.
“The purpose of this study was to assess the risk of developing diabetes-associated ocular complications among a population-based cohort of children diagnosed with either T1D or T2D during a 50-year period,” lead author Patricia Bai, BA, of Mayo Clinic, Phoenix, and colleagues reported in JAMA Ophthalmology.
The researchers retrospectively reviewed medical records from all residents of Olmsted County, Minn., from 1970 to 2019. The study cohort included 606 children with a confirmed a diagnosis of T1D or T2D, 525 (87%) of whom had at least one ocular examination.
The mean age at diabetes diagnosis was 12 years (standard deviation, 5.4 years); most participants were White (95.7% in 1990), and half (50%) were male. Diabetes-associated ocular complications occurred in 31.9% and 26.6% of children with T1D and T2D, respectively.
The hazard ratios illustrating the risk between T2D and T1D rates were 1.88 (95% confidence interval, 1.13-3.12; P = .02) for any diabetic retinopathy, 2.33 (95% CI, 0.99-5.50; P = .048) for proliferative diabetic retinopathy, 1.49 (95% CI, 0.46-4.89; P = .50) for diabetic macular edema, 2.43 (95% CI, 0.54-11.07; P = .24) for a visually significant cataract, and 4.06 (95% CI, 1.34-12.33; P = .007) for requiring pars plana vitrectomy within the first 15 years of diagnosis.
These results suggest that earlier surveillance and intervention may help prevent vision-threatening complications, the researchers explained.
“After adjusting for race using self-identified categories of White or not White, the adjusted HR of developing any retinopathy was 1.63 (95% CI, 0.96-2.79; P = .07), and the adjusted HR of developing proliferative diabetic retinopathy was 2.02 (95% CI, 0.79-5.16; P = .14)” in T2D versus T1D patients, the researchers wrote.
“We would expect the reported rate of type 2 diabetes to be potentially underestimated in our study cohort,” Ms. Bai commented in an interview. “Race has been suggested to be a surrogate for other social determinants of health, such as lower rates of optimal follow-up care received by racial and ethnic minorities, which could influence subsequent retinopathy rates.”
Understanding retinopathy outcomes in youth
In an accompanying editorial, Jennifer K. Sun, MD, MPH, from Harvard Medical School, Boston, wrote that the present study indicates the natural history of retinopathy may differ between patients with T1D and T2D.
While the pathophysiology of diabetic retinopathy in T1D and T2D appears similar, other patient-related factors such as lipid profiles, the presence of hypertension, and body mass index may differ between the two disease states.
She wrote that “there is a particular need to document retinopathy outcomes and risk factors for advanced disease in youth with T2D, for whom there is a paucity of information.”
Ms. Bai and colleagues acknowledged that a key limitation of the study was the retrospective design. As a result, irregular follow-up and incomplete data may limit the applicability of the findings.
“Some children with milder forms of diabetes may have eluded detection, a limitation that is more likely to affect T2D, which may exist undetected for years before a diagnosis,” Bai explained.
Dr. Sun recommended that further epidemiologic studies are needed to help optimize guidelines for screening and follow-up for young people diagnosed with diabetes. “Such efforts may potentially lead to increased understanding of the mechanistic differences between pathology in T1D versus T2D,” she concluded.
This study used the resources of the Rochester Epidemiology Project (REP) medical records linkage system, which is supported by grant funding from the National Institute on Aging, the Mayo Clinic Research Committee, and by fees paid annually by REP users. The study authors disclosed no conflicts of interest.
FROM JAMA OPHTHALMOLOGY
Poorly controlled asthma predicts COVID-19 hospitalization in children
Children and adolescents with poorly controlled asthma were three to six times more likely to be hospitalized with COVID-19 infections, based on data from a national study of more than 750,000 children in Scotland.
Although the majority of COVID-19 cases in children have been mild, some children require hospitalization, wrote Ting Shi, PhD, of the University of Edinburgh (Scotland) and colleagues.
Vaccination policies to potentially reduce infection and hospitalization of children remain inconsistent, the researchers said. Identifying which school-age children would derive the greatest benefit from vaccination “could help to reduce the risk of infection and consequently the need for children to have time off school; and might also reduce the risk of spread of SARS-CoV-2 within schools and households,” but the potential benefits of vaccination for children with asthma in particular have not been well studied, they wrote.
The United Kingdom’s Joint Commission on Vaccination and Immunisation commissioned research on the rates of hospitalization among children with poorly controlled asthma. In a national incidence cohort study published in The Lancet Respiratory Medicine, the researchers reviewed data from all children aged 5-17 years in Scotland who were enrolled in the linked dataset of Early Pandemic Evaluation and Enhanced Surveillance of COVID-19 (EAVE II). The total number of children in the dataset was 752,867, and 63,463 (8.4%) of these had diagnosed asthma. Among the children with asthma, 4,339 (6.8%) had confirmed COVID-19 infections between March 1, 2020, and July 27, 2021. A total of 67 infected children were hospitalized. Of the 689,404 children without asthma, 40,231 (5.8%) had confirmed COVID-19 infections, and 382 (0.9%) of these children were hospitalized.
Overall, hospital admission rates for COVID-19 were significantly higher among children with asthma, compared to those without asthma (adjusted hazard ratio, 1.49), and the rates increased among children with poorly controlled asthma.
The researchers used previous hospital admission for asthma as a measure of uncontrolled asthma, and found that hospitalization was at least six times as likely for children with poorly controlled asthma, compared with those with no asthma (aHR, 6.40), although children with well-controlled asthma also had an increased risk of hospitalization, compared with those with no asthma (aHR, 1.36).
When the researchers used oral corticosteroid prescriptions as an indicator of uncontrolled asthma, the adjusted hazard ratios were 3.38, 3.53, 1.52, and 1.34 for children with prescribed corticosteroid courses of three or more, two, one, and none, respectively, compared with children with no asthma.
These hazard ratios remained significant after controlling for factors including age, sex, socioeconomic status, comorbidity, and previous hospital admission, the researchers wrote.
In an age-based analysis, results were similar for children aged 12-17 years, but in children aged 5-11 years, the hospitalization risk decreased for those with one course of corticosteroids and reached the highest rate for those with three or more courses, rather than two courses.
The study findings were limited by several factors including the relatively small numbers of COVID-19 hospitalizations, ICU admissions, and deaths in children with asthma, the researchers noted. Other limitations include potential changes in asthma control over the study period, and lack of data on certain confounders such as tobacco use, unsuitable housing, and ethnicity, they noted. However, the results were strengthened by the use of a large, national dataset, and access to electronic health records, they said.
The findings reflect data from previous studies suggesting increased risk of hospitalization for patients with respiratory illness who develop COVID-19 infections, the researchers wrote.
The results emphasize the importance of good asthma control to protect children from severe COVID-19, and careful monitoring of children with poorly controlled asthma who do become infected, they added.
“The findings from this linkage of multiple data sources have helped inform the prioritisation of school-aged children with poorly controlled asthma for vaccines,” they concluded.
Findings support value of vaccination for children with asthma
“Pediatricians see many children who suffer from asthma, and although one could assume that these children would have more serious consequences from contracting COVID-19, the current study examines a large database in a way not possible in the United States to address the severity question,” said Suzanne C. Boulter, MD, of the Geisel School of Medicine at Dartmouth, Hanover, N.H. “The authors used prior hospitalization rate or two prescriptions for oral corticosteroids as markers of asthma severity prior to the onset of COVID-19 in Scotland, and they collected retrospective data for 16 months of the pandemic through July of 2021, showing a significant increase in hospitalization for those children,” she said. Dr. Boulter said she was not surprised by this finding, given the impact of COVID-19 on the respiratory system.
“Pediatricians have found significant challenges from some groups of parents when discussing the indications and need for vaccination in their patients,” said Dr. Boulter. “Having this data on the increased risk of morbidity and mortality in children with asthma might help parents who are uncertain about the risk/benefit ratio of the vaccine make their decision,” she said.
Dr. Boulter said she hoped that additional studies will yield ongoing information about hospitalization rates for COVID-19 not only about asthma, but also other diagnoses affecting children in the United States and worldwide.
“It would also be important to see a breakdown of ethnic factors and adverse childhood experiences and how they relate to hospitalization and death from COVID-19,” Dr. Boulter said.
“The results of this study are not surprising, as we have known for a long time that children with severe asthma are more susceptible to severe respiratory viruses,” Francis E. Rushton, MD, a pediatrician in Beaufort, S.C., said in an interview. “But the study is still important, as it helps us determine which children are most urgently in need of protection from COVID-19 in any of its forms,” he emphasized. In particular, the current study underlines the importance of vaccinating children with unstable asthma, Dr. Rushton said.
Going forward, “it would be interesting to do additional studies looking at other markers for poor asthma control that could guide our vaccine efforts so that they are focused on those most at risk,” he added.
The study was supported by the UK Research and Innovation (Medical Research Council), Research and Innovation Industrial Strategy Challenge Fund, Health Data Research UK, and the Scottish Government. Lead author Dr. Shi had no financial conflicts to disclose. Dr. Rushton and Dr. Boulter had no financial conflicts to disclose, but each serves on the Editorial Advisory Board of Pediatric News.
Children and adolescents with poorly controlled asthma were three to six times more likely to be hospitalized with COVID-19 infections, based on data from a national study of more than 750,000 children in Scotland.
Although the majority of COVID-19 cases in children have been mild, some children require hospitalization, wrote Ting Shi, PhD, of the University of Edinburgh (Scotland) and colleagues.
Vaccination policies to potentially reduce infection and hospitalization of children remain inconsistent, the researchers said. Identifying which school-age children would derive the greatest benefit from vaccination “could help to reduce the risk of infection and consequently the need for children to have time off school; and might also reduce the risk of spread of SARS-CoV-2 within schools and households,” but the potential benefits of vaccination for children with asthma in particular have not been well studied, they wrote.
The United Kingdom’s Joint Commission on Vaccination and Immunisation commissioned research on the rates of hospitalization among children with poorly controlled asthma. In a national incidence cohort study published in The Lancet Respiratory Medicine, the researchers reviewed data from all children aged 5-17 years in Scotland who were enrolled in the linked dataset of Early Pandemic Evaluation and Enhanced Surveillance of COVID-19 (EAVE II). The total number of children in the dataset was 752,867, and 63,463 (8.4%) of these had diagnosed asthma. Among the children with asthma, 4,339 (6.8%) had confirmed COVID-19 infections between March 1, 2020, and July 27, 2021. A total of 67 infected children were hospitalized. Of the 689,404 children without asthma, 40,231 (5.8%) had confirmed COVID-19 infections, and 382 (0.9%) of these children were hospitalized.
Overall, hospital admission rates for COVID-19 were significantly higher among children with asthma, compared to those without asthma (adjusted hazard ratio, 1.49), and the rates increased among children with poorly controlled asthma.
The researchers used previous hospital admission for asthma as a measure of uncontrolled asthma, and found that hospitalization was at least six times as likely for children with poorly controlled asthma, compared with those with no asthma (aHR, 6.40), although children with well-controlled asthma also had an increased risk of hospitalization, compared with those with no asthma (aHR, 1.36).
When the researchers used oral corticosteroid prescriptions as an indicator of uncontrolled asthma, the adjusted hazard ratios were 3.38, 3.53, 1.52, and 1.34 for children with prescribed corticosteroid courses of three or more, two, one, and none, respectively, compared with children with no asthma.
These hazard ratios remained significant after controlling for factors including age, sex, socioeconomic status, comorbidity, and previous hospital admission, the researchers wrote.
In an age-based analysis, results were similar for children aged 12-17 years, but in children aged 5-11 years, the hospitalization risk decreased for those with one course of corticosteroids and reached the highest rate for those with three or more courses, rather than two courses.
The study findings were limited by several factors including the relatively small numbers of COVID-19 hospitalizations, ICU admissions, and deaths in children with asthma, the researchers noted. Other limitations include potential changes in asthma control over the study period, and lack of data on certain confounders such as tobacco use, unsuitable housing, and ethnicity, they noted. However, the results were strengthened by the use of a large, national dataset, and access to electronic health records, they said.
The findings reflect data from previous studies suggesting increased risk of hospitalization for patients with respiratory illness who develop COVID-19 infections, the researchers wrote.
The results emphasize the importance of good asthma control to protect children from severe COVID-19, and careful monitoring of children with poorly controlled asthma who do become infected, they added.
“The findings from this linkage of multiple data sources have helped inform the prioritisation of school-aged children with poorly controlled asthma for vaccines,” they concluded.
Findings support value of vaccination for children with asthma
“Pediatricians see many children who suffer from asthma, and although one could assume that these children would have more serious consequences from contracting COVID-19, the current study examines a large database in a way not possible in the United States to address the severity question,” said Suzanne C. Boulter, MD, of the Geisel School of Medicine at Dartmouth, Hanover, N.H. “The authors used prior hospitalization rate or two prescriptions for oral corticosteroids as markers of asthma severity prior to the onset of COVID-19 in Scotland, and they collected retrospective data for 16 months of the pandemic through July of 2021, showing a significant increase in hospitalization for those children,” she said. Dr. Boulter said she was not surprised by this finding, given the impact of COVID-19 on the respiratory system.
“Pediatricians have found significant challenges from some groups of parents when discussing the indications and need for vaccination in their patients,” said Dr. Boulter. “Having this data on the increased risk of morbidity and mortality in children with asthma might help parents who are uncertain about the risk/benefit ratio of the vaccine make their decision,” she said.
Dr. Boulter said she hoped that additional studies will yield ongoing information about hospitalization rates for COVID-19 not only about asthma, but also other diagnoses affecting children in the United States and worldwide.
“It would also be important to see a breakdown of ethnic factors and adverse childhood experiences and how they relate to hospitalization and death from COVID-19,” Dr. Boulter said.
“The results of this study are not surprising, as we have known for a long time that children with severe asthma are more susceptible to severe respiratory viruses,” Francis E. Rushton, MD, a pediatrician in Beaufort, S.C., said in an interview. “But the study is still important, as it helps us determine which children are most urgently in need of protection from COVID-19 in any of its forms,” he emphasized. In particular, the current study underlines the importance of vaccinating children with unstable asthma, Dr. Rushton said.
Going forward, “it would be interesting to do additional studies looking at other markers for poor asthma control that could guide our vaccine efforts so that they are focused on those most at risk,” he added.
The study was supported by the UK Research and Innovation (Medical Research Council), Research and Innovation Industrial Strategy Challenge Fund, Health Data Research UK, and the Scottish Government. Lead author Dr. Shi had no financial conflicts to disclose. Dr. Rushton and Dr. Boulter had no financial conflicts to disclose, but each serves on the Editorial Advisory Board of Pediatric News.
Children and adolescents with poorly controlled asthma were three to six times more likely to be hospitalized with COVID-19 infections, based on data from a national study of more than 750,000 children in Scotland.
Although the majority of COVID-19 cases in children have been mild, some children require hospitalization, wrote Ting Shi, PhD, of the University of Edinburgh (Scotland) and colleagues.
Vaccination policies to potentially reduce infection and hospitalization of children remain inconsistent, the researchers said. Identifying which school-age children would derive the greatest benefit from vaccination “could help to reduce the risk of infection and consequently the need for children to have time off school; and might also reduce the risk of spread of SARS-CoV-2 within schools and households,” but the potential benefits of vaccination for children with asthma in particular have not been well studied, they wrote.
The United Kingdom’s Joint Commission on Vaccination and Immunisation commissioned research on the rates of hospitalization among children with poorly controlled asthma. In a national incidence cohort study published in The Lancet Respiratory Medicine, the researchers reviewed data from all children aged 5-17 years in Scotland who were enrolled in the linked dataset of Early Pandemic Evaluation and Enhanced Surveillance of COVID-19 (EAVE II). The total number of children in the dataset was 752,867, and 63,463 (8.4%) of these had diagnosed asthma. Among the children with asthma, 4,339 (6.8%) had confirmed COVID-19 infections between March 1, 2020, and July 27, 2021. A total of 67 infected children were hospitalized. Of the 689,404 children without asthma, 40,231 (5.8%) had confirmed COVID-19 infections, and 382 (0.9%) of these children were hospitalized.
Overall, hospital admission rates for COVID-19 were significantly higher among children with asthma, compared to those without asthma (adjusted hazard ratio, 1.49), and the rates increased among children with poorly controlled asthma.
The researchers used previous hospital admission for asthma as a measure of uncontrolled asthma, and found that hospitalization was at least six times as likely for children with poorly controlled asthma, compared with those with no asthma (aHR, 6.40), although children with well-controlled asthma also had an increased risk of hospitalization, compared with those with no asthma (aHR, 1.36).
When the researchers used oral corticosteroid prescriptions as an indicator of uncontrolled asthma, the adjusted hazard ratios were 3.38, 3.53, 1.52, and 1.34 for children with prescribed corticosteroid courses of three or more, two, one, and none, respectively, compared with children with no asthma.
These hazard ratios remained significant after controlling for factors including age, sex, socioeconomic status, comorbidity, and previous hospital admission, the researchers wrote.
In an age-based analysis, results were similar for children aged 12-17 years, but in children aged 5-11 years, the hospitalization risk decreased for those with one course of corticosteroids and reached the highest rate for those with three or more courses, rather than two courses.
The study findings were limited by several factors including the relatively small numbers of COVID-19 hospitalizations, ICU admissions, and deaths in children with asthma, the researchers noted. Other limitations include potential changes in asthma control over the study period, and lack of data on certain confounders such as tobacco use, unsuitable housing, and ethnicity, they noted. However, the results were strengthened by the use of a large, national dataset, and access to electronic health records, they said.
The findings reflect data from previous studies suggesting increased risk of hospitalization for patients with respiratory illness who develop COVID-19 infections, the researchers wrote.
The results emphasize the importance of good asthma control to protect children from severe COVID-19, and careful monitoring of children with poorly controlled asthma who do become infected, they added.
“The findings from this linkage of multiple data sources have helped inform the prioritisation of school-aged children with poorly controlled asthma for vaccines,” they concluded.
Findings support value of vaccination for children with asthma
“Pediatricians see many children who suffer from asthma, and although one could assume that these children would have more serious consequences from contracting COVID-19, the current study examines a large database in a way not possible in the United States to address the severity question,” said Suzanne C. Boulter, MD, of the Geisel School of Medicine at Dartmouth, Hanover, N.H. “The authors used prior hospitalization rate or two prescriptions for oral corticosteroids as markers of asthma severity prior to the onset of COVID-19 in Scotland, and they collected retrospective data for 16 months of the pandemic through July of 2021, showing a significant increase in hospitalization for those children,” she said. Dr. Boulter said she was not surprised by this finding, given the impact of COVID-19 on the respiratory system.
“Pediatricians have found significant challenges from some groups of parents when discussing the indications and need for vaccination in their patients,” said Dr. Boulter. “Having this data on the increased risk of morbidity and mortality in children with asthma might help parents who are uncertain about the risk/benefit ratio of the vaccine make their decision,” she said.
Dr. Boulter said she hoped that additional studies will yield ongoing information about hospitalization rates for COVID-19 not only about asthma, but also other diagnoses affecting children in the United States and worldwide.
“It would also be important to see a breakdown of ethnic factors and adverse childhood experiences and how they relate to hospitalization and death from COVID-19,” Dr. Boulter said.
“The results of this study are not surprising, as we have known for a long time that children with severe asthma are more susceptible to severe respiratory viruses,” Francis E. Rushton, MD, a pediatrician in Beaufort, S.C., said in an interview. “But the study is still important, as it helps us determine which children are most urgently in need of protection from COVID-19 in any of its forms,” he emphasized. In particular, the current study underlines the importance of vaccinating children with unstable asthma, Dr. Rushton said.
Going forward, “it would be interesting to do additional studies looking at other markers for poor asthma control that could guide our vaccine efforts so that they are focused on those most at risk,” he added.
The study was supported by the UK Research and Innovation (Medical Research Council), Research and Innovation Industrial Strategy Challenge Fund, Health Data Research UK, and the Scottish Government. Lead author Dr. Shi had no financial conflicts to disclose. Dr. Rushton and Dr. Boulter had no financial conflicts to disclose, but each serves on the Editorial Advisory Board of Pediatric News.
FROM THE LANCET
‘Top’ surgery for trans youth: Advance or dangerous medicine?
Is the gender-affirmative treatment approach an example of “medicine continuing on its progressive march of improving human life” or “a manifestation of dangerous medicine that ... will cause more harm than benefit to vulnerable youths?” wonders an Australian psychiatrist in a newly published letter that addresses the controversial procedure of masculinizing chest surgery – a double mastectomy – in young people with gender dysphoria (GD).
Alison Clayton, MBBS, explores the evidence for masculinizing chest surgery and looks back at examples of “dangerous medicine” in the past century while looking forward, wondering how future medics will retrospectively view gender affirmative treatment, especially so-called “top” or masculinizing chest surgery, which is in actual fact a double mastectomy, in a letter published Nov. 22 in the Archives of Sexual Behavior.
“It is surprising that clinicians and researchers claim chest surgery for GD youth is an evidence-based intervention, rather than acknowledging it is an experimental treatment that requires more rigorous and human research ethics committee [HREC] approved research,” she writes.
“The medical profession needs to consider whether, in its championing of the gender-affirmative approach for GD youth, it is also acting brashly and making mistakes that will negatively impact some young people for the rest of their lives,” she continues.
Ms. Clayton, after many years of experience as a psychiatrist, has recently returned to postgraduate research into the history of 20th-century psychiatry at the School of Historical and Philosophical Studies, University of Melbourne.
Meanwhile, the authors of a viewpoint published online Dec. 1 in JAMA Surgery, agree with Ms. Clayton on the issue of a lack of long-term studies on which to base decisions, particularly when it comes to insurance coverage for gender surgeries in the United States.
Nnenaya Agochukwu-Mmonu, MD, and colleagues recommend use of the coverage with evidence development (CED) approach, which would, they say, provide a “rigorous evidence base for gender-affirming interventions and surgery while simultaneously allowing access and provisional coverage for these services.”
Threefold increase in gender-affirming surgeries in past decade
There has been a threefold rise in the rate of gender-affirming surgeries in the United States in the past decade, which can be attributed to increased recognition of gender dysphoria, decreasing social stigma toward these individuals, greater clinical experience, and expanding insurance coverage, according to Dr. Agochukwu-Mmonu, of the department of urology, NYU School of Medicine, and coauthors.
Ms. Clayton meanwhile notes that of the increasing number of adolescents being referred for treatment for gender dysphoria in the Western world, most were born female and many have “a history of psychiatric illness or neurodevelopmental disorders.”
Many of these youngsters also show a “high demand” for surgical removal of breasts, she adds, noting that this operation is being undertaken as routine treatment in patients as young as 13, with some clinicians arguing that “this surgery is an evidence-based intervention that improves mental health outcomes, and that it is discriminatory for it not to be available.”
She also notes that “chest dysphoria” is “a recently created term meaning discomfort with one’s breasts.” The term “breast” is therefore largely absent in publications talking about this surgery as it “may cause distress for transgender males,” to quote one source, Ms. Clayton says, and “this seems part of a broader pattern of removing this term from clinical language,” according to another article on the subject.
Ms. Clayton also says, “There are only a handful of published studies focusing on the potential benefits of masculinizing chest surgery,” and notes that these mostly report on surgery for individuals younger than 21 years old.
Significant methodological flaws in existing research
One study of 14 postsurgical youth (nine of whom were under 18 years) found that “all reported high aesthetic satisfaction and most self-reported low complication rates and improvement in mood.”
Another cross-sectional retrospective survey looked at 68 postsurgical transmasculine youth (72% of the eligible postsurgical population); 49% had surgery when younger than age 18, with the youngest being age 13 and the oldest age 24. At the time of the survey, only 14% of participants were more than 2 years postsurgery. The postsurgical participants were found to have reduced chest dysphoria (the outcome) compared with a convenience and nonmatched comparison sample of nonsurgical transmasculine youth.
And a 2021 qualitative study of 30 transmale youth – about half of whom had undergone chest surgery – concluded that the postsurgical cohort experienced “tremendous” benefits in chest dysphoria and a range of psychological outcomes.
On this particular study, Ms. Clayton notes that “in my opinion, they did not provide enough detail for the reader to make an informed judgment regarding this latter claim.”
She goes on to discuss genital surgery, sometimes called full gender-affirming surgery (or “bottom surgery”), and says proponents of these operations point out that the main objections to them in minors is to “surgical sterilization, and people get super worked up about that ... it is a barrier we have to overcome, and I think we are going to.”
Ms. Clayton asserts that it seems “this barrier is already being overcome, as it has been reported that in the United States, genital surgery is being undertaken on gender dysphoric minors as young as 15 years old.”
Reflecting on the available evidence, Ms. Clayton highlights the significant methodological flaws that limit the extent to which surgery can be linked to short-term improved mental health outcomes and adds that information on long-term outcomes and rates of regret is unavailable.
She also asserts that the research fails to assess “a role for psychological interventions which could be utilized, as a least-harm intervention, until maturity is reached.”
Historical examples of experimental medicine
Ms. Clayton goes on to draw parallels with experimental medicine performed on homosexuals in the 20th century, highlighting the medical and surgical interventions, which included metrazol convulsive therapy, chemical castration with estrogens, surgical castration, clitoridectomy, brain operations, and aversive electrotherapy.
She also refers to the historical practice of hormonal treatment for “tall girls” and “short boys” between the 1960s and 1980s. Hormones were given to young people who did not have any medical reason underpinning their stature but were distressed, and society considered their height to have a negative social impact.
“With the encouragement of physicians and school nurses, enthusiastic media promotion, and pharmaceutical companies’ advertising, parents sought hormonal interventions,” she writes, adding that, at the time the hormones were considered safe, but long-term adverse effects emerged, including impaired fertility and increased risk of cancers.
“This seems another part of the story of medicine acting to reinforce society’s sex stereotypes, and for some patients it came at disastrous personal cost,” writes Ms. Clayton.
The gender-affirming approach is based on endorsing the adolescent’s stated gender identity with minimal questioning and “that they should be supported to undertake social transition, medical transition, masculinizing chest surgery, and, some also argue, genital surgery,” she writes.
Objectors to this approach pinpoint the “limited and low-quality evidence base for the benefits” but also “the irreversible and long-term adverse impacts of these treatments on fertility and sexual function, as well as on bone, brain, and cardiovascular functioning.”
Current studies of gender-affirming surgeries lack standardization
In their viewpoint, Dr. Agochukwu-Mmonu and colleagues state that use of a CED would not only help provide an evidence base but would also ensure better-informed policy access and coverage decisions to help standardize approaches to gender surgery in the United States.
Currently, they note, “Studies examining the mental health benefit for patients undergoing gender-affirming surgeries include measures that lack standardization, evaluate different interventions (that is, surgeries are rarely done with concurrent hormone administration), include dissimilar patient populations, and use different study designs.”
This difference in study design leads to variation in reported outcomes. Although many studies have shown benefit, others report that patients have unrealistic expectations or experience regret, Dr. Agochukwu-Mmonu and coauthors conclude.
CED provides an option that would enable informed decisions. “It allows the deliberate use of innovative therapies, explicit integration of transgender and nonbinary patient input, and ongoing systematic evaluation aimed to identify specific patient groups who would or would not benefit from their use.”
This leads back to Ms. Clayton’s central question around whether the gender-affirmative approach is a medical advance or dangerous medicine.
“Why are these experimental interventions, with inherent risks and scarce, low-quality evidence for benefits being implemented outside HREC-regulated clinical trial settings?’” she wonders.
Ms. Clayton has declared no conflicts of interest.
A version of this article first appeared on Medscape.com.
Is the gender-affirmative treatment approach an example of “medicine continuing on its progressive march of improving human life” or “a manifestation of dangerous medicine that ... will cause more harm than benefit to vulnerable youths?” wonders an Australian psychiatrist in a newly published letter that addresses the controversial procedure of masculinizing chest surgery – a double mastectomy – in young people with gender dysphoria (GD).
Alison Clayton, MBBS, explores the evidence for masculinizing chest surgery and looks back at examples of “dangerous medicine” in the past century while looking forward, wondering how future medics will retrospectively view gender affirmative treatment, especially so-called “top” or masculinizing chest surgery, which is in actual fact a double mastectomy, in a letter published Nov. 22 in the Archives of Sexual Behavior.
“It is surprising that clinicians and researchers claim chest surgery for GD youth is an evidence-based intervention, rather than acknowledging it is an experimental treatment that requires more rigorous and human research ethics committee [HREC] approved research,” she writes.
“The medical profession needs to consider whether, in its championing of the gender-affirmative approach for GD youth, it is also acting brashly and making mistakes that will negatively impact some young people for the rest of their lives,” she continues.
Ms. Clayton, after many years of experience as a psychiatrist, has recently returned to postgraduate research into the history of 20th-century psychiatry at the School of Historical and Philosophical Studies, University of Melbourne.
Meanwhile, the authors of a viewpoint published online Dec. 1 in JAMA Surgery, agree with Ms. Clayton on the issue of a lack of long-term studies on which to base decisions, particularly when it comes to insurance coverage for gender surgeries in the United States.
Nnenaya Agochukwu-Mmonu, MD, and colleagues recommend use of the coverage with evidence development (CED) approach, which would, they say, provide a “rigorous evidence base for gender-affirming interventions and surgery while simultaneously allowing access and provisional coverage for these services.”
Threefold increase in gender-affirming surgeries in past decade
There has been a threefold rise in the rate of gender-affirming surgeries in the United States in the past decade, which can be attributed to increased recognition of gender dysphoria, decreasing social stigma toward these individuals, greater clinical experience, and expanding insurance coverage, according to Dr. Agochukwu-Mmonu, of the department of urology, NYU School of Medicine, and coauthors.
Ms. Clayton meanwhile notes that of the increasing number of adolescents being referred for treatment for gender dysphoria in the Western world, most were born female and many have “a history of psychiatric illness or neurodevelopmental disorders.”
Many of these youngsters also show a “high demand” for surgical removal of breasts, she adds, noting that this operation is being undertaken as routine treatment in patients as young as 13, with some clinicians arguing that “this surgery is an evidence-based intervention that improves mental health outcomes, and that it is discriminatory for it not to be available.”
She also notes that “chest dysphoria” is “a recently created term meaning discomfort with one’s breasts.” The term “breast” is therefore largely absent in publications talking about this surgery as it “may cause distress for transgender males,” to quote one source, Ms. Clayton says, and “this seems part of a broader pattern of removing this term from clinical language,” according to another article on the subject.
Ms. Clayton also says, “There are only a handful of published studies focusing on the potential benefits of masculinizing chest surgery,” and notes that these mostly report on surgery for individuals younger than 21 years old.
Significant methodological flaws in existing research
One study of 14 postsurgical youth (nine of whom were under 18 years) found that “all reported high aesthetic satisfaction and most self-reported low complication rates and improvement in mood.”
Another cross-sectional retrospective survey looked at 68 postsurgical transmasculine youth (72% of the eligible postsurgical population); 49% had surgery when younger than age 18, with the youngest being age 13 and the oldest age 24. At the time of the survey, only 14% of participants were more than 2 years postsurgery. The postsurgical participants were found to have reduced chest dysphoria (the outcome) compared with a convenience and nonmatched comparison sample of nonsurgical transmasculine youth.
And a 2021 qualitative study of 30 transmale youth – about half of whom had undergone chest surgery – concluded that the postsurgical cohort experienced “tremendous” benefits in chest dysphoria and a range of psychological outcomes.
On this particular study, Ms. Clayton notes that “in my opinion, they did not provide enough detail for the reader to make an informed judgment regarding this latter claim.”
She goes on to discuss genital surgery, sometimes called full gender-affirming surgery (or “bottom surgery”), and says proponents of these operations point out that the main objections to them in minors is to “surgical sterilization, and people get super worked up about that ... it is a barrier we have to overcome, and I think we are going to.”
Ms. Clayton asserts that it seems “this barrier is already being overcome, as it has been reported that in the United States, genital surgery is being undertaken on gender dysphoric minors as young as 15 years old.”
Reflecting on the available evidence, Ms. Clayton highlights the significant methodological flaws that limit the extent to which surgery can be linked to short-term improved mental health outcomes and adds that information on long-term outcomes and rates of regret is unavailable.
She also asserts that the research fails to assess “a role for psychological interventions which could be utilized, as a least-harm intervention, until maturity is reached.”
Historical examples of experimental medicine
Ms. Clayton goes on to draw parallels with experimental medicine performed on homosexuals in the 20th century, highlighting the medical and surgical interventions, which included metrazol convulsive therapy, chemical castration with estrogens, surgical castration, clitoridectomy, brain operations, and aversive electrotherapy.
She also refers to the historical practice of hormonal treatment for “tall girls” and “short boys” between the 1960s and 1980s. Hormones were given to young people who did not have any medical reason underpinning their stature but were distressed, and society considered their height to have a negative social impact.
“With the encouragement of physicians and school nurses, enthusiastic media promotion, and pharmaceutical companies’ advertising, parents sought hormonal interventions,” she writes, adding that, at the time the hormones were considered safe, but long-term adverse effects emerged, including impaired fertility and increased risk of cancers.
“This seems another part of the story of medicine acting to reinforce society’s sex stereotypes, and for some patients it came at disastrous personal cost,” writes Ms. Clayton.
The gender-affirming approach is based on endorsing the adolescent’s stated gender identity with minimal questioning and “that they should be supported to undertake social transition, medical transition, masculinizing chest surgery, and, some also argue, genital surgery,” she writes.
Objectors to this approach pinpoint the “limited and low-quality evidence base for the benefits” but also “the irreversible and long-term adverse impacts of these treatments on fertility and sexual function, as well as on bone, brain, and cardiovascular functioning.”
Current studies of gender-affirming surgeries lack standardization
In their viewpoint, Dr. Agochukwu-Mmonu and colleagues state that use of a CED would not only help provide an evidence base but would also ensure better-informed policy access and coverage decisions to help standardize approaches to gender surgery in the United States.
Currently, they note, “Studies examining the mental health benefit for patients undergoing gender-affirming surgeries include measures that lack standardization, evaluate different interventions (that is, surgeries are rarely done with concurrent hormone administration), include dissimilar patient populations, and use different study designs.”
This difference in study design leads to variation in reported outcomes. Although many studies have shown benefit, others report that patients have unrealistic expectations or experience regret, Dr. Agochukwu-Mmonu and coauthors conclude.
CED provides an option that would enable informed decisions. “It allows the deliberate use of innovative therapies, explicit integration of transgender and nonbinary patient input, and ongoing systematic evaluation aimed to identify specific patient groups who would or would not benefit from their use.”
This leads back to Ms. Clayton’s central question around whether the gender-affirmative approach is a medical advance or dangerous medicine.
“Why are these experimental interventions, with inherent risks and scarce, low-quality evidence for benefits being implemented outside HREC-regulated clinical trial settings?’” she wonders.
Ms. Clayton has declared no conflicts of interest.
A version of this article first appeared on Medscape.com.
Is the gender-affirmative treatment approach an example of “medicine continuing on its progressive march of improving human life” or “a manifestation of dangerous medicine that ... will cause more harm than benefit to vulnerable youths?” wonders an Australian psychiatrist in a newly published letter that addresses the controversial procedure of masculinizing chest surgery – a double mastectomy – in young people with gender dysphoria (GD).
Alison Clayton, MBBS, explores the evidence for masculinizing chest surgery and looks back at examples of “dangerous medicine” in the past century while looking forward, wondering how future medics will retrospectively view gender affirmative treatment, especially so-called “top” or masculinizing chest surgery, which is in actual fact a double mastectomy, in a letter published Nov. 22 in the Archives of Sexual Behavior.
“It is surprising that clinicians and researchers claim chest surgery for GD youth is an evidence-based intervention, rather than acknowledging it is an experimental treatment that requires more rigorous and human research ethics committee [HREC] approved research,” she writes.
“The medical profession needs to consider whether, in its championing of the gender-affirmative approach for GD youth, it is also acting brashly and making mistakes that will negatively impact some young people for the rest of their lives,” she continues.
Ms. Clayton, after many years of experience as a psychiatrist, has recently returned to postgraduate research into the history of 20th-century psychiatry at the School of Historical and Philosophical Studies, University of Melbourne.
Meanwhile, the authors of a viewpoint published online Dec. 1 in JAMA Surgery, agree with Ms. Clayton on the issue of a lack of long-term studies on which to base decisions, particularly when it comes to insurance coverage for gender surgeries in the United States.
Nnenaya Agochukwu-Mmonu, MD, and colleagues recommend use of the coverage with evidence development (CED) approach, which would, they say, provide a “rigorous evidence base for gender-affirming interventions and surgery while simultaneously allowing access and provisional coverage for these services.”
Threefold increase in gender-affirming surgeries in past decade
There has been a threefold rise in the rate of gender-affirming surgeries in the United States in the past decade, which can be attributed to increased recognition of gender dysphoria, decreasing social stigma toward these individuals, greater clinical experience, and expanding insurance coverage, according to Dr. Agochukwu-Mmonu, of the department of urology, NYU School of Medicine, and coauthors.
Ms. Clayton meanwhile notes that of the increasing number of adolescents being referred for treatment for gender dysphoria in the Western world, most were born female and many have “a history of psychiatric illness or neurodevelopmental disorders.”
Many of these youngsters also show a “high demand” for surgical removal of breasts, she adds, noting that this operation is being undertaken as routine treatment in patients as young as 13, with some clinicians arguing that “this surgery is an evidence-based intervention that improves mental health outcomes, and that it is discriminatory for it not to be available.”
She also notes that “chest dysphoria” is “a recently created term meaning discomfort with one’s breasts.” The term “breast” is therefore largely absent in publications talking about this surgery as it “may cause distress for transgender males,” to quote one source, Ms. Clayton says, and “this seems part of a broader pattern of removing this term from clinical language,” according to another article on the subject.
Ms. Clayton also says, “There are only a handful of published studies focusing on the potential benefits of masculinizing chest surgery,” and notes that these mostly report on surgery for individuals younger than 21 years old.
Significant methodological flaws in existing research
One study of 14 postsurgical youth (nine of whom were under 18 years) found that “all reported high aesthetic satisfaction and most self-reported low complication rates and improvement in mood.”
Another cross-sectional retrospective survey looked at 68 postsurgical transmasculine youth (72% of the eligible postsurgical population); 49% had surgery when younger than age 18, with the youngest being age 13 and the oldest age 24. At the time of the survey, only 14% of participants were more than 2 years postsurgery. The postsurgical participants were found to have reduced chest dysphoria (the outcome) compared with a convenience and nonmatched comparison sample of nonsurgical transmasculine youth.
And a 2021 qualitative study of 30 transmale youth – about half of whom had undergone chest surgery – concluded that the postsurgical cohort experienced “tremendous” benefits in chest dysphoria and a range of psychological outcomes.
On this particular study, Ms. Clayton notes that “in my opinion, they did not provide enough detail for the reader to make an informed judgment regarding this latter claim.”
She goes on to discuss genital surgery, sometimes called full gender-affirming surgery (or “bottom surgery”), and says proponents of these operations point out that the main objections to them in minors is to “surgical sterilization, and people get super worked up about that ... it is a barrier we have to overcome, and I think we are going to.”
Ms. Clayton asserts that it seems “this barrier is already being overcome, as it has been reported that in the United States, genital surgery is being undertaken on gender dysphoric minors as young as 15 years old.”
Reflecting on the available evidence, Ms. Clayton highlights the significant methodological flaws that limit the extent to which surgery can be linked to short-term improved mental health outcomes and adds that information on long-term outcomes and rates of regret is unavailable.
She also asserts that the research fails to assess “a role for psychological interventions which could be utilized, as a least-harm intervention, until maturity is reached.”
Historical examples of experimental medicine
Ms. Clayton goes on to draw parallels with experimental medicine performed on homosexuals in the 20th century, highlighting the medical and surgical interventions, which included metrazol convulsive therapy, chemical castration with estrogens, surgical castration, clitoridectomy, brain operations, and aversive electrotherapy.
She also refers to the historical practice of hormonal treatment for “tall girls” and “short boys” between the 1960s and 1980s. Hormones were given to young people who did not have any medical reason underpinning their stature but were distressed, and society considered their height to have a negative social impact.
“With the encouragement of physicians and school nurses, enthusiastic media promotion, and pharmaceutical companies’ advertising, parents sought hormonal interventions,” she writes, adding that, at the time the hormones were considered safe, but long-term adverse effects emerged, including impaired fertility and increased risk of cancers.
“This seems another part of the story of medicine acting to reinforce society’s sex stereotypes, and for some patients it came at disastrous personal cost,” writes Ms. Clayton.
The gender-affirming approach is based on endorsing the adolescent’s stated gender identity with minimal questioning and “that they should be supported to undertake social transition, medical transition, masculinizing chest surgery, and, some also argue, genital surgery,” she writes.
Objectors to this approach pinpoint the “limited and low-quality evidence base for the benefits” but also “the irreversible and long-term adverse impacts of these treatments on fertility and sexual function, as well as on bone, brain, and cardiovascular functioning.”
Current studies of gender-affirming surgeries lack standardization
In their viewpoint, Dr. Agochukwu-Mmonu and colleagues state that use of a CED would not only help provide an evidence base but would also ensure better-informed policy access and coverage decisions to help standardize approaches to gender surgery in the United States.
Currently, they note, “Studies examining the mental health benefit for patients undergoing gender-affirming surgeries include measures that lack standardization, evaluate different interventions (that is, surgeries are rarely done with concurrent hormone administration), include dissimilar patient populations, and use different study designs.”
This difference in study design leads to variation in reported outcomes. Although many studies have shown benefit, others report that patients have unrealistic expectations or experience regret, Dr. Agochukwu-Mmonu and coauthors conclude.
CED provides an option that would enable informed decisions. “It allows the deliberate use of innovative therapies, explicit integration of transgender and nonbinary patient input, and ongoing systematic evaluation aimed to identify specific patient groups who would or would not benefit from their use.”
This leads back to Ms. Clayton’s central question around whether the gender-affirmative approach is a medical advance or dangerous medicine.
“Why are these experimental interventions, with inherent risks and scarce, low-quality evidence for benefits being implemented outside HREC-regulated clinical trial settings?’” she wonders.
Ms. Clayton has declared no conflicts of interest.
A version of this article first appeared on Medscape.com.
FROM JAMA SURGERY
Children and COVID: New cases, vaccinations both decline
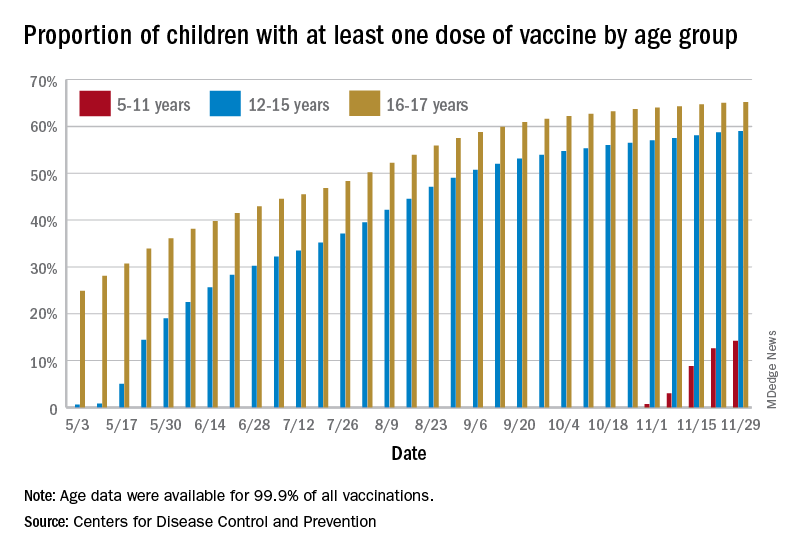
States reported 131,828 new pediatric cases for the week of Nov. 19-25, a decline of 7.1% over the previous week but still enough to surpass 100,000 for the 16th consecutive week. The weekly count had risen for 3 straight weeks since the last decrease in late October, the American Academy of Pediatrics and the Children’s Hospital Association said Nov. 30 in their weekly COVID report.
The AAP/CHA analysis, based on data from state and territorial health departments, puts the total number of cases in children at 6.9 million since the pandemic began, representing 17.0% of cases in Americans of all ages. The Centers for Disease Control and Prevention, which uses an age limit of 18 years to define a child, unlike some states, reports numbers of 6.1 million and 15.5%.
New vaccinations among the youngest eligible children, those aged 5-11 years, were down for the second week in a row after reaching almost 1.7 million during the first full week after approval on Nov. 2. Since then, the vaccination counts have been 1.2 million (Nov. 16-22) and 333,000 (Nov. 23-29), the CDC said on its COVID Data Tracker. A similar drop in the last week – from 127,000 to just 50,000 – also was seen for those aged 12-17 years.
Altogether, 14.2% of children aged 5-11, almost 4.1 million individuals, have received at least one dose of the vaccine, compared with 59.0% (10 million) of the 12- to 15-year-olds and 65.2% (5.5 million) of those aged 16-17. Just under 1% of the youngest group has been fully vaccinated, versus 49.0% and 55.8% for the older children, the CDC said.
It has been reported that Pfizer and BioNTech, which produce the only COVID vaccine approved for children, are planning to apply to the Food and Drug Administration during the first week of December for authorization for a booster dose for 16- and 17-year-olds.

States reported 131,828 new pediatric cases for the week of Nov. 19-25, a decline of 7.1% over the previous week but still enough to surpass 100,000 for the 16th consecutive week. The weekly count had risen for 3 straight weeks since the last decrease in late October, the American Academy of Pediatrics and the Children’s Hospital Association said Nov. 30 in their weekly COVID report.
The AAP/CHA analysis, based on data from state and territorial health departments, puts the total number of cases in children at 6.9 million since the pandemic began, representing 17.0% of cases in Americans of all ages. The Centers for Disease Control and Prevention, which uses an age limit of 18 years to define a child, unlike some states, reports numbers of 6.1 million and 15.5%.
New vaccinations among the youngest eligible children, those aged 5-11 years, were down for the second week in a row after reaching almost 1.7 million during the first full week after approval on Nov. 2. Since then, the vaccination counts have been 1.2 million (Nov. 16-22) and 333,000 (Nov. 23-29), the CDC said on its COVID Data Tracker. A similar drop in the last week – from 127,000 to just 50,000 – also was seen for those aged 12-17 years.
Altogether, 14.2% of children aged 5-11, almost 4.1 million individuals, have received at least one dose of the vaccine, compared with 59.0% (10 million) of the 12- to 15-year-olds and 65.2% (5.5 million) of those aged 16-17. Just under 1% of the youngest group has been fully vaccinated, versus 49.0% and 55.8% for the older children, the CDC said.
It has been reported that Pfizer and BioNTech, which produce the only COVID vaccine approved for children, are planning to apply to the Food and Drug Administration during the first week of December for authorization for a booster dose for 16- and 17-year-olds.

States reported 131,828 new pediatric cases for the week of Nov. 19-25, a decline of 7.1% over the previous week but still enough to surpass 100,000 for the 16th consecutive week. The weekly count had risen for 3 straight weeks since the last decrease in late October, the American Academy of Pediatrics and the Children’s Hospital Association said Nov. 30 in their weekly COVID report.
The AAP/CHA analysis, based on data from state and territorial health departments, puts the total number of cases in children at 6.9 million since the pandemic began, representing 17.0% of cases in Americans of all ages. The Centers for Disease Control and Prevention, which uses an age limit of 18 years to define a child, unlike some states, reports numbers of 6.1 million and 15.5%.
New vaccinations among the youngest eligible children, those aged 5-11 years, were down for the second week in a row after reaching almost 1.7 million during the first full week after approval on Nov. 2. Since then, the vaccination counts have been 1.2 million (Nov. 16-22) and 333,000 (Nov. 23-29), the CDC said on its COVID Data Tracker. A similar drop in the last week – from 127,000 to just 50,000 – also was seen for those aged 12-17 years.
Altogether, 14.2% of children aged 5-11, almost 4.1 million individuals, have received at least one dose of the vaccine, compared with 59.0% (10 million) of the 12- to 15-year-olds and 65.2% (5.5 million) of those aged 16-17. Just under 1% of the youngest group has been fully vaccinated, versus 49.0% and 55.8% for the older children, the CDC said.
It has been reported that Pfizer and BioNTech, which produce the only COVID vaccine approved for children, are planning to apply to the Food and Drug Administration during the first week of December for authorization for a booster dose for 16- and 17-year-olds.
Association of height, BMI, and AD in young children may be transient
The , according to a large cohort study published online in JAMA Dermatology.
“The potential for ‘catch up’ in height for children with atopic dermatitis observed in our study may be explained with resolution of atopic dermatitis or successful treatment,” write senior author Aaron M. Drucker, MD, ScM, from the division of dermatology, University of Toronto, and Women’s College Hospital in Toronto, and colleagues. They postulated that, while the association between AD and shorter height is “is likely multifactorial,” it may be driven in part by sleep loss caused by AD, or corticosteroid treatment of AD, both of which can result in growth retardation and subsequent increased BMI.
The researchers used data from TARGet Kids!, a prospective, longitudinal cohort study designed to study multiple health conditions in children from general pediatric and family practices across Toronto. Their study included 10,611 children for whom there was data on height, weight, BMI, and standardized z scores, which account for age and sex differences in anthropometric characteristics. Clinically relevant covariates that were collected included child age, sex, birth weight, history of asthma, family income, maternal and paternal ethnicity, and maternal height and BMI.
The mean age of the children in the study at cohort entry was 23 months, and they were followed for a median of 28.5 months, during which time they had a median of two visits. At baseline, 947 (8.9%) children had parent-reported AD, with this number rising to 1,834 (17.3%) during follow-up.
After adjusting for covariates, AD was associated with lower mean z-height (P < .001), higher mean z-BMI (P = .008), but lower mean z-weight (P < .001), compared with children without AD. Using World Health Organization growth tables, the researchers estimated that “children with atopic dermatitis were, on average, approximately 0.5 cm shorter at age 2 years and 0.6 cm shorter at age 5 years than children without atopic dermatitis” after adjusting for covariates. They also estimated that children with AD were “on average, approximately 0.2 more BMI units at age 2 years” than children without AD. The associations between AD and height diminished by age 14 years, as did the association between AD and BMI by age 5.5 years.
“Given that we found children with atopic dermatitis to be somewhat less heavy, as measured by z-weight, than children without atopic dermatitis and that this association did not attenuate with age, it is possible that our findings for BMI, and perhaps those of previous studies, are explained mainly by differences in height,” the authors write. “This distinction has obvious clinical importance – rather than a focus on obesity and obesogenic behaviors being problematic in children with atopic dermatitis, research might be better directed at understanding the association between atopic dermatitis and initially shorter stature.”
Asked to comment on the study results, Jonathan Silverberg, MD, PhD, MPH, associate professor of dermatology, George Washington University, Washington, told this news organization he would have preferred using the wording “in addition to focusing on obesity,” rather than “focus on obesity.”
“We should not ignore diet and sedentary activity as important factors,” he said, pointing to another recent study that found higher rates of eating disorders associated with AD.
Dr. Silverberg said that he was not familiar enough with the cohort sample to comment on how representative it is of the Canadian population, or on how generalizable the results are to other regions and populations. Generalizability, he added, “is an important issue, as we previously found regional differences with respect to the association between AD and obesity.”
In addition, he noted that in the study AD was defined as an “ever history” of disease rather than “in the past year or currently,” so, even though it is a longitudinal study, “it is really looking at how AD at any point in patients’ lives is related to weight or stature,” he explained. But, he added, “many cases of childhood AD ‘burn out’ or become milder/clear as the children get older. So, if the AD clears, then one would expect to see attenuation of associations as the children get older. However, this doesn’t tell us about how persistent AD into later childhood or adolescence is related to height or weight.”
Previous studies found that short stature and obesity were particularly associated with moderate – and even more to severe – atopic dermatitis, Dr. Silverberg said. It is likely that most patients in this primary care cohort had mild disease, he noted, so the effect sizes are likely diluted by mostly mild disease “and not relevant to the more persistent and severe AD patients encountered in the dermatology practice setting.”
The study was supported by the department of medicine, Women’s College Hospital, and the Canadian Institutes of Health Research.
One author reported receiving compensation from the British Journal of Dermatology, the American Academy of Dermatology, and the National Eczema Association and has served as a paid consultant for the Canadian Agency for Drugs and Technologies in Health outside the submitted work. No other disclosures were reported. Dr. Silverberg has disclosed no relevant financial relationships.
Commentary by Robert Sidbury, MD, MPH
Among the more puzzling “associations” to emerge in recent literature has been the association between atopic dermatitis (AD) and obesity. I see many children with severe AD every day and my gestalt “association” is a thinner, shorter child rather than an overweight one. Dr. Drucker and colleagues’ data has helped me understand this dissonance. Children with AD do in fact, on average, weigh less but they are also shorter, possibly explaining their higher body mass index (BMI). More important, these findings are transient, with height differences dissipating by 14 years of age, and BMI differences by kindergarten. This information should train providers’ sights on optimal AD treatment and optimal nutritional and lifestyle support without undue concern for obesity or obesogenic behaviors.
Dr. Sidbury is chief of dermatology at Seattle Children's Hospital and professor, department of pediatrics, University of Washington, Seattle. He is a site principal investigator for dupilumab trials, for which the hospital has a contract with Regeneron.
A version of this article first appeared on Medscape.com.
This article was updated 6/18/22.
The , according to a large cohort study published online in JAMA Dermatology.
“The potential for ‘catch up’ in height for children with atopic dermatitis observed in our study may be explained with resolution of atopic dermatitis or successful treatment,” write senior author Aaron M. Drucker, MD, ScM, from the division of dermatology, University of Toronto, and Women’s College Hospital in Toronto, and colleagues. They postulated that, while the association between AD and shorter height is “is likely multifactorial,” it may be driven in part by sleep loss caused by AD, or corticosteroid treatment of AD, both of which can result in growth retardation and subsequent increased BMI.
The researchers used data from TARGet Kids!, a prospective, longitudinal cohort study designed to study multiple health conditions in children from general pediatric and family practices across Toronto. Their study included 10,611 children for whom there was data on height, weight, BMI, and standardized z scores, which account for age and sex differences in anthropometric characteristics. Clinically relevant covariates that were collected included child age, sex, birth weight, history of asthma, family income, maternal and paternal ethnicity, and maternal height and BMI.
The mean age of the children in the study at cohort entry was 23 months, and they were followed for a median of 28.5 months, during which time they had a median of two visits. At baseline, 947 (8.9%) children had parent-reported AD, with this number rising to 1,834 (17.3%) during follow-up.
After adjusting for covariates, AD was associated with lower mean z-height (P < .001), higher mean z-BMI (P = .008), but lower mean z-weight (P < .001), compared with children without AD. Using World Health Organization growth tables, the researchers estimated that “children with atopic dermatitis were, on average, approximately 0.5 cm shorter at age 2 years and 0.6 cm shorter at age 5 years than children without atopic dermatitis” after adjusting for covariates. They also estimated that children with AD were “on average, approximately 0.2 more BMI units at age 2 years” than children without AD. The associations between AD and height diminished by age 14 years, as did the association between AD and BMI by age 5.5 years.
“Given that we found children with atopic dermatitis to be somewhat less heavy, as measured by z-weight, than children without atopic dermatitis and that this association did not attenuate with age, it is possible that our findings for BMI, and perhaps those of previous studies, are explained mainly by differences in height,” the authors write. “This distinction has obvious clinical importance – rather than a focus on obesity and obesogenic behaviors being problematic in children with atopic dermatitis, research might be better directed at understanding the association between atopic dermatitis and initially shorter stature.”
Asked to comment on the study results, Jonathan Silverberg, MD, PhD, MPH, associate professor of dermatology, George Washington University, Washington, told this news organization he would have preferred using the wording “in addition to focusing on obesity,” rather than “focus on obesity.”
“We should not ignore diet and sedentary activity as important factors,” he said, pointing to another recent study that found higher rates of eating disorders associated with AD.
Dr. Silverberg said that he was not familiar enough with the cohort sample to comment on how representative it is of the Canadian population, or on how generalizable the results are to other regions and populations. Generalizability, he added, “is an important issue, as we previously found regional differences with respect to the association between AD and obesity.”
In addition, he noted that in the study AD was defined as an “ever history” of disease rather than “in the past year or currently,” so, even though it is a longitudinal study, “it is really looking at how AD at any point in patients’ lives is related to weight or stature,” he explained. But, he added, “many cases of childhood AD ‘burn out’ or become milder/clear as the children get older. So, if the AD clears, then one would expect to see attenuation of associations as the children get older. However, this doesn’t tell us about how persistent AD into later childhood or adolescence is related to height or weight.”
Previous studies found that short stature and obesity were particularly associated with moderate – and even more to severe – atopic dermatitis, Dr. Silverberg said. It is likely that most patients in this primary care cohort had mild disease, he noted, so the effect sizes are likely diluted by mostly mild disease “and not relevant to the more persistent and severe AD patients encountered in the dermatology practice setting.”
The study was supported by the department of medicine, Women’s College Hospital, and the Canadian Institutes of Health Research.
One author reported receiving compensation from the British Journal of Dermatology, the American Academy of Dermatology, and the National Eczema Association and has served as a paid consultant for the Canadian Agency for Drugs and Technologies in Health outside the submitted work. No other disclosures were reported. Dr. Silverberg has disclosed no relevant financial relationships.
Commentary by Robert Sidbury, MD, MPH
Among the more puzzling “associations” to emerge in recent literature has been the association between atopic dermatitis (AD) and obesity. I see many children with severe AD every day and my gestalt “association” is a thinner, shorter child rather than an overweight one. Dr. Drucker and colleagues’ data has helped me understand this dissonance. Children with AD do in fact, on average, weigh less but they are also shorter, possibly explaining their higher body mass index (BMI). More important, these findings are transient, with height differences dissipating by 14 years of age, and BMI differences by kindergarten. This information should train providers’ sights on optimal AD treatment and optimal nutritional and lifestyle support without undue concern for obesity or obesogenic behaviors.
Dr. Sidbury is chief of dermatology at Seattle Children's Hospital and professor, department of pediatrics, University of Washington, Seattle. He is a site principal investigator for dupilumab trials, for which the hospital has a contract with Regeneron.
A version of this article first appeared on Medscape.com.
This article was updated 6/18/22.
The , according to a large cohort study published online in JAMA Dermatology.
“The potential for ‘catch up’ in height for children with atopic dermatitis observed in our study may be explained with resolution of atopic dermatitis or successful treatment,” write senior author Aaron M. Drucker, MD, ScM, from the division of dermatology, University of Toronto, and Women’s College Hospital in Toronto, and colleagues. They postulated that, while the association between AD and shorter height is “is likely multifactorial,” it may be driven in part by sleep loss caused by AD, or corticosteroid treatment of AD, both of which can result in growth retardation and subsequent increased BMI.
The researchers used data from TARGet Kids!, a prospective, longitudinal cohort study designed to study multiple health conditions in children from general pediatric and family practices across Toronto. Their study included 10,611 children for whom there was data on height, weight, BMI, and standardized z scores, which account for age and sex differences in anthropometric characteristics. Clinically relevant covariates that were collected included child age, sex, birth weight, history of asthma, family income, maternal and paternal ethnicity, and maternal height and BMI.
The mean age of the children in the study at cohort entry was 23 months, and they were followed for a median of 28.5 months, during which time they had a median of two visits. At baseline, 947 (8.9%) children had parent-reported AD, with this number rising to 1,834 (17.3%) during follow-up.
After adjusting for covariates, AD was associated with lower mean z-height (P < .001), higher mean z-BMI (P = .008), but lower mean z-weight (P < .001), compared with children without AD. Using World Health Organization growth tables, the researchers estimated that “children with atopic dermatitis were, on average, approximately 0.5 cm shorter at age 2 years and 0.6 cm shorter at age 5 years than children without atopic dermatitis” after adjusting for covariates. They also estimated that children with AD were “on average, approximately 0.2 more BMI units at age 2 years” than children without AD. The associations between AD and height diminished by age 14 years, as did the association between AD and BMI by age 5.5 years.
“Given that we found children with atopic dermatitis to be somewhat less heavy, as measured by z-weight, than children without atopic dermatitis and that this association did not attenuate with age, it is possible that our findings for BMI, and perhaps those of previous studies, are explained mainly by differences in height,” the authors write. “This distinction has obvious clinical importance – rather than a focus on obesity and obesogenic behaviors being problematic in children with atopic dermatitis, research might be better directed at understanding the association between atopic dermatitis and initially shorter stature.”
Asked to comment on the study results, Jonathan Silverberg, MD, PhD, MPH, associate professor of dermatology, George Washington University, Washington, told this news organization he would have preferred using the wording “in addition to focusing on obesity,” rather than “focus on obesity.”
“We should not ignore diet and sedentary activity as important factors,” he said, pointing to another recent study that found higher rates of eating disorders associated with AD.
Dr. Silverberg said that he was not familiar enough with the cohort sample to comment on how representative it is of the Canadian population, or on how generalizable the results are to other regions and populations. Generalizability, he added, “is an important issue, as we previously found regional differences with respect to the association between AD and obesity.”
In addition, he noted that in the study AD was defined as an “ever history” of disease rather than “in the past year or currently,” so, even though it is a longitudinal study, “it is really looking at how AD at any point in patients’ lives is related to weight or stature,” he explained. But, he added, “many cases of childhood AD ‘burn out’ or become milder/clear as the children get older. So, if the AD clears, then one would expect to see attenuation of associations as the children get older. However, this doesn’t tell us about how persistent AD into later childhood or adolescence is related to height or weight.”
Previous studies found that short stature and obesity were particularly associated with moderate – and even more to severe – atopic dermatitis, Dr. Silverberg said. It is likely that most patients in this primary care cohort had mild disease, he noted, so the effect sizes are likely diluted by mostly mild disease “and not relevant to the more persistent and severe AD patients encountered in the dermatology practice setting.”
The study was supported by the department of medicine, Women’s College Hospital, and the Canadian Institutes of Health Research.
One author reported receiving compensation from the British Journal of Dermatology, the American Academy of Dermatology, and the National Eczema Association and has served as a paid consultant for the Canadian Agency for Drugs and Technologies in Health outside the submitted work. No other disclosures were reported. Dr. Silverberg has disclosed no relevant financial relationships.
Commentary by Robert Sidbury, MD, MPH
Among the more puzzling “associations” to emerge in recent literature has been the association between atopic dermatitis (AD) and obesity. I see many children with severe AD every day and my gestalt “association” is a thinner, shorter child rather than an overweight one. Dr. Drucker and colleagues’ data has helped me understand this dissonance. Children with AD do in fact, on average, weigh less but they are also shorter, possibly explaining their higher body mass index (BMI). More important, these findings are transient, with height differences dissipating by 14 years of age, and BMI differences by kindergarten. This information should train providers’ sights on optimal AD treatment and optimal nutritional and lifestyle support without undue concern for obesity or obesogenic behaviors.
Dr. Sidbury is chief of dermatology at Seattle Children's Hospital and professor, department of pediatrics, University of Washington, Seattle. He is a site principal investigator for dupilumab trials, for which the hospital has a contract with Regeneron.
A version of this article first appeared on Medscape.com.
This article was updated 6/18/22.
FROM JAMA DERMATOLOGY
Sickle cell raises risk for stillbirth
Both sickle cell trait and sickle cell disease were significantly associated with an increased risk of stillbirth, based on data from more than 50,000 women.
Pregnant women with sickle cell disease (SCD) are at increased risk of complications, including stillbirth, but many women with the disease in the United States lack access to specialty care, Silvia P. Canelón, PhD, of the University of Pennsylvania, Philadelphia, and colleagues wrote. Sickle cell trait (SCT), defined as one abnormal allele of the hemoglobin gene, is not considered a disease state because many carriers are asymptomatic, and therefore even less likely to be assessed for potential complications. “However, it is possible for people with SCT to experience sickling of red blood cells under severe hypoxia, dehydration, and hyperthermia. This condition can lead to severe medical complications for sickle cell carriers, including fetal loss, splenic infarction, exercise-related sudden death, and others,” they noted.
In a study published in JAMA Network Open, the researchers reviewed data from 63,334 deliveries in 50,560 women between Jan. 1, 2010, and Aug. 15, 2017, at four quaternary academic medical centers in Pennsylvania. Of these, 1,904 had SCT but not SCD, and 164 had SCD. The mean age of the women was 29.5 years, and approximately 56% were single at the time of delivery. A majority (87%) of the study population was Rhesus-factor positive, 47.0% were Black or African American, 33.7% were White, and 45.2% had ABO blood type O.
Risk factors for stillbirth used in the analysis included SCD, numbers of pain crises and blood transfusions before delivery, delivery episode (to represent parity), history of cesarean delivery, multiple gestation, age, marital status, race and ethnicity, ABO blood type, Rhesus factor, and year of delivery.
Overall, the prevalence of stillbirth in women with SCT was 1.1%, compared with 0.8% in the general study population, and was significantly associated with increased risk of stillbirth after controlling for multiple risk factors. The adjusted odds ratio was 8.94 for stillbirth risk in women with SCT, compared with women without SCT (P = .045), although the risk was greater among women with SCD, compared with those without SCD (aOR, 26.40).
“In addition, the stratified analysis found Black or African American patients with SCD to be at higher risk of stillbirth, compared with Black or African American patients without SCD (aOR, 3.59),” but no significant association was noted between stillbirth and SCT, the researchers wrote. Stillbirth rates were 1.1% in Black or African American women overall, 2.7% in those with SCD, and 1.0% in those with SCT. Overall, multiple gestation was associated with an increased risk of stillbirth (aOR, 4.68), while a history of cesarean delivery and being married at the time of delivery were associated with decreased risk (aOR, 0.44 and 0.72, respectively).
The lack of association between stillbirth and SCT in Black or African American patients supports some previous research, but contradicts other studies, the researchers wrote. “Ultimately, it may be impossible to disentangle the risks due to the disease and those due to disparities associated with the disease that have resulted from longstanding inequity and stigma,” they said. The findings also suggest that biological mechanisms of SCT may contribute to severe clinical complications, and therefore “invite a more critical examination of the assumption that SCT is not a disease state.”
The study findings were limited by several factors including the lack of assessment of SCT independent of other comorbidities, such as hypertension, preeclampsia, diabetes, and obesity, and by the use of billing codes that could misclassify patients, the researchers noted.
However, the results support some findings from previous studies of the potential health complications for pregnant SCT patients. The large study population highlights the need to identify women’s SCT status during obstetric care, and to provide both pregnancy guidance for SCT patients and systemic support of comprehensive care for SCD and SCT patients, they concluded.
Disparities may drive stillbirth in sickle cell trait women
“There is a paucity of research evaluating sickle cell trait and the risk of adverse pregnancy outcomes such as stillbirth,” Iris Krishna, MD, of Emory University, Atlanta, said in an interview. “Prior studies evaluating the risk of stillbirth have yielded mixed results, and an increased risk of stillbirth in women with sickle cell trait has not been established. This study is unique in that it attempts to address how racial inequities and health disparities may contribute to risk of stillbirth in women with sickle cell trait.”
Although the study findings suggest an increased risk of stillbirth in women with sickle cell trait, an analysis stratified for Black or African American patients showed no association, Dr. Krishna said. “The prevalence of stillbirth was noted to be 1% among Black or African American patients with sickle cell trait compared to the prevalence of stillbirth of 1.1% among Black or African American women with no sickle cell trait or disease. Although, sickle cell trait or sickle cell disease can be found in any racial or ethnic group, it disproportionately affects Black or African Americans, with a sickle cell trait carrier rate of approximately 1 in 10. The mixed findings in this study amongst racial/ethnic groups further suggest that there is more research needed before an association between stillbirth and sickle cell trait can be supported.”
As for clinical implications, “it is well established that for women with sickle cell trait there is an increased risk of urinary tract infections in pregnancy,” said Dr. Krishna. “Women with sickle cell trait should have a urine culture performed at their first prenatal visit and each trimester. At this time, studies evaluating risk of stillbirth in women with sickle cell trait have yielded conflicting results, and current consensus is that women with sickle cell trait are not at increased risk. In comparison, women with sickle cell disease are at increased risk for stillbirth and adverse pregnancy outcomes. Women with sickle cell disease should be followed closely during pregnancy and fetal surveillance implemented at 32 weeks, if not sooner, to reduce risk of stillbirth.
“Prior studies evaluating risk of stillbirth in women with sickle cell trait consist of retrospective cohorts with small study populations,” Dr. Krishna added. Notably, the current study was limited by the inability to adjust for comorbidities including diabetes, hypertension, and obesity, that are not only associated with an increased risk for stillbirth, but also disproportionately common among Black women.
“More studies are needed evaluating the relationship between these comorbidities as well as studies specifically evaluating how race affects care and pregnancy outcomes,” Dr. Krisha emphasized.
The study was funded by the University of Pennsylvania department of biostatistics, epidemiology, and informatics. Lead author Dr. Canelón disclosed grants from the Centers for Disease Control and Prevention, Clinical and Translational Science Awards, and grants from the National Institutes of Health outside the submitted work. Dr. Krishna had no financial conflicts to disclose, but serves on the editorial advisory board of Ob.Gyn News.
Both sickle cell trait and sickle cell disease were significantly associated with an increased risk of stillbirth, based on data from more than 50,000 women.
Pregnant women with sickle cell disease (SCD) are at increased risk of complications, including stillbirth, but many women with the disease in the United States lack access to specialty care, Silvia P. Canelón, PhD, of the University of Pennsylvania, Philadelphia, and colleagues wrote. Sickle cell trait (SCT), defined as one abnormal allele of the hemoglobin gene, is not considered a disease state because many carriers are asymptomatic, and therefore even less likely to be assessed for potential complications. “However, it is possible for people with SCT to experience sickling of red blood cells under severe hypoxia, dehydration, and hyperthermia. This condition can lead to severe medical complications for sickle cell carriers, including fetal loss, splenic infarction, exercise-related sudden death, and others,” they noted.
In a study published in JAMA Network Open, the researchers reviewed data from 63,334 deliveries in 50,560 women between Jan. 1, 2010, and Aug. 15, 2017, at four quaternary academic medical centers in Pennsylvania. Of these, 1,904 had SCT but not SCD, and 164 had SCD. The mean age of the women was 29.5 years, and approximately 56% were single at the time of delivery. A majority (87%) of the study population was Rhesus-factor positive, 47.0% were Black or African American, 33.7% were White, and 45.2% had ABO blood type O.
Risk factors for stillbirth used in the analysis included SCD, numbers of pain crises and blood transfusions before delivery, delivery episode (to represent parity), history of cesarean delivery, multiple gestation, age, marital status, race and ethnicity, ABO blood type, Rhesus factor, and year of delivery.
Overall, the prevalence of stillbirth in women with SCT was 1.1%, compared with 0.8% in the general study population, and was significantly associated with increased risk of stillbirth after controlling for multiple risk factors. The adjusted odds ratio was 8.94 for stillbirth risk in women with SCT, compared with women without SCT (P = .045), although the risk was greater among women with SCD, compared with those without SCD (aOR, 26.40).
“In addition, the stratified analysis found Black or African American patients with SCD to be at higher risk of stillbirth, compared with Black or African American patients without SCD (aOR, 3.59),” but no significant association was noted between stillbirth and SCT, the researchers wrote. Stillbirth rates were 1.1% in Black or African American women overall, 2.7% in those with SCD, and 1.0% in those with SCT. Overall, multiple gestation was associated with an increased risk of stillbirth (aOR, 4.68), while a history of cesarean delivery and being married at the time of delivery were associated with decreased risk (aOR, 0.44 and 0.72, respectively).
The lack of association between stillbirth and SCT in Black or African American patients supports some previous research, but contradicts other studies, the researchers wrote. “Ultimately, it may be impossible to disentangle the risks due to the disease and those due to disparities associated with the disease that have resulted from longstanding inequity and stigma,” they said. The findings also suggest that biological mechanisms of SCT may contribute to severe clinical complications, and therefore “invite a more critical examination of the assumption that SCT is not a disease state.”
The study findings were limited by several factors including the lack of assessment of SCT independent of other comorbidities, such as hypertension, preeclampsia, diabetes, and obesity, and by the use of billing codes that could misclassify patients, the researchers noted.
However, the results support some findings from previous studies of the potential health complications for pregnant SCT patients. The large study population highlights the need to identify women’s SCT status during obstetric care, and to provide both pregnancy guidance for SCT patients and systemic support of comprehensive care for SCD and SCT patients, they concluded.
Disparities may drive stillbirth in sickle cell trait women
“There is a paucity of research evaluating sickle cell trait and the risk of adverse pregnancy outcomes such as stillbirth,” Iris Krishna, MD, of Emory University, Atlanta, said in an interview. “Prior studies evaluating the risk of stillbirth have yielded mixed results, and an increased risk of stillbirth in women with sickle cell trait has not been established. This study is unique in that it attempts to address how racial inequities and health disparities may contribute to risk of stillbirth in women with sickle cell trait.”
Although the study findings suggest an increased risk of stillbirth in women with sickle cell trait, an analysis stratified for Black or African American patients showed no association, Dr. Krishna said. “The prevalence of stillbirth was noted to be 1% among Black or African American patients with sickle cell trait compared to the prevalence of stillbirth of 1.1% among Black or African American women with no sickle cell trait or disease. Although, sickle cell trait or sickle cell disease can be found in any racial or ethnic group, it disproportionately affects Black or African Americans, with a sickle cell trait carrier rate of approximately 1 in 10. The mixed findings in this study amongst racial/ethnic groups further suggest that there is more research needed before an association between stillbirth and sickle cell trait can be supported.”
As for clinical implications, “it is well established that for women with sickle cell trait there is an increased risk of urinary tract infections in pregnancy,” said Dr. Krishna. “Women with sickle cell trait should have a urine culture performed at their first prenatal visit and each trimester. At this time, studies evaluating risk of stillbirth in women with sickle cell trait have yielded conflicting results, and current consensus is that women with sickle cell trait are not at increased risk. In comparison, women with sickle cell disease are at increased risk for stillbirth and adverse pregnancy outcomes. Women with sickle cell disease should be followed closely during pregnancy and fetal surveillance implemented at 32 weeks, if not sooner, to reduce risk of stillbirth.
“Prior studies evaluating risk of stillbirth in women with sickle cell trait consist of retrospective cohorts with small study populations,” Dr. Krishna added. Notably, the current study was limited by the inability to adjust for comorbidities including diabetes, hypertension, and obesity, that are not only associated with an increased risk for stillbirth, but also disproportionately common among Black women.
“More studies are needed evaluating the relationship between these comorbidities as well as studies specifically evaluating how race affects care and pregnancy outcomes,” Dr. Krisha emphasized.
The study was funded by the University of Pennsylvania department of biostatistics, epidemiology, and informatics. Lead author Dr. Canelón disclosed grants from the Centers for Disease Control and Prevention, Clinical and Translational Science Awards, and grants from the National Institutes of Health outside the submitted work. Dr. Krishna had no financial conflicts to disclose, but serves on the editorial advisory board of Ob.Gyn News.
Both sickle cell trait and sickle cell disease were significantly associated with an increased risk of stillbirth, based on data from more than 50,000 women.
Pregnant women with sickle cell disease (SCD) are at increased risk of complications, including stillbirth, but many women with the disease in the United States lack access to specialty care, Silvia P. Canelón, PhD, of the University of Pennsylvania, Philadelphia, and colleagues wrote. Sickle cell trait (SCT), defined as one abnormal allele of the hemoglobin gene, is not considered a disease state because many carriers are asymptomatic, and therefore even less likely to be assessed for potential complications. “However, it is possible for people with SCT to experience sickling of red blood cells under severe hypoxia, dehydration, and hyperthermia. This condition can lead to severe medical complications for sickle cell carriers, including fetal loss, splenic infarction, exercise-related sudden death, and others,” they noted.
In a study published in JAMA Network Open, the researchers reviewed data from 63,334 deliveries in 50,560 women between Jan. 1, 2010, and Aug. 15, 2017, at four quaternary academic medical centers in Pennsylvania. Of these, 1,904 had SCT but not SCD, and 164 had SCD. The mean age of the women was 29.5 years, and approximately 56% were single at the time of delivery. A majority (87%) of the study population was Rhesus-factor positive, 47.0% were Black or African American, 33.7% were White, and 45.2% had ABO blood type O.
Risk factors for stillbirth used in the analysis included SCD, numbers of pain crises and blood transfusions before delivery, delivery episode (to represent parity), history of cesarean delivery, multiple gestation, age, marital status, race and ethnicity, ABO blood type, Rhesus factor, and year of delivery.
Overall, the prevalence of stillbirth in women with SCT was 1.1%, compared with 0.8% in the general study population, and was significantly associated with increased risk of stillbirth after controlling for multiple risk factors. The adjusted odds ratio was 8.94 for stillbirth risk in women with SCT, compared with women without SCT (P = .045), although the risk was greater among women with SCD, compared with those without SCD (aOR, 26.40).
“In addition, the stratified analysis found Black or African American patients with SCD to be at higher risk of stillbirth, compared with Black or African American patients without SCD (aOR, 3.59),” but no significant association was noted between stillbirth and SCT, the researchers wrote. Stillbirth rates were 1.1% in Black or African American women overall, 2.7% in those with SCD, and 1.0% in those with SCT. Overall, multiple gestation was associated with an increased risk of stillbirth (aOR, 4.68), while a history of cesarean delivery and being married at the time of delivery were associated with decreased risk (aOR, 0.44 and 0.72, respectively).
The lack of association between stillbirth and SCT in Black or African American patients supports some previous research, but contradicts other studies, the researchers wrote. “Ultimately, it may be impossible to disentangle the risks due to the disease and those due to disparities associated with the disease that have resulted from longstanding inequity and stigma,” they said. The findings also suggest that biological mechanisms of SCT may contribute to severe clinical complications, and therefore “invite a more critical examination of the assumption that SCT is not a disease state.”
The study findings were limited by several factors including the lack of assessment of SCT independent of other comorbidities, such as hypertension, preeclampsia, diabetes, and obesity, and by the use of billing codes that could misclassify patients, the researchers noted.
However, the results support some findings from previous studies of the potential health complications for pregnant SCT patients. The large study population highlights the need to identify women’s SCT status during obstetric care, and to provide both pregnancy guidance for SCT patients and systemic support of comprehensive care for SCD and SCT patients, they concluded.
Disparities may drive stillbirth in sickle cell trait women
“There is a paucity of research evaluating sickle cell trait and the risk of adverse pregnancy outcomes such as stillbirth,” Iris Krishna, MD, of Emory University, Atlanta, said in an interview. “Prior studies evaluating the risk of stillbirth have yielded mixed results, and an increased risk of stillbirth in women with sickle cell trait has not been established. This study is unique in that it attempts to address how racial inequities and health disparities may contribute to risk of stillbirth in women with sickle cell trait.”
Although the study findings suggest an increased risk of stillbirth in women with sickle cell trait, an analysis stratified for Black or African American patients showed no association, Dr. Krishna said. “The prevalence of stillbirth was noted to be 1% among Black or African American patients with sickle cell trait compared to the prevalence of stillbirth of 1.1% among Black or African American women with no sickle cell trait or disease. Although, sickle cell trait or sickle cell disease can be found in any racial or ethnic group, it disproportionately affects Black or African Americans, with a sickle cell trait carrier rate of approximately 1 in 10. The mixed findings in this study amongst racial/ethnic groups further suggest that there is more research needed before an association between stillbirth and sickle cell trait can be supported.”
As for clinical implications, “it is well established that for women with sickle cell trait there is an increased risk of urinary tract infections in pregnancy,” said Dr. Krishna. “Women with sickle cell trait should have a urine culture performed at their first prenatal visit and each trimester. At this time, studies evaluating risk of stillbirth in women with sickle cell trait have yielded conflicting results, and current consensus is that women with sickle cell trait are not at increased risk. In comparison, women with sickle cell disease are at increased risk for stillbirth and adverse pregnancy outcomes. Women with sickle cell disease should be followed closely during pregnancy and fetal surveillance implemented at 32 weeks, if not sooner, to reduce risk of stillbirth.
“Prior studies evaluating risk of stillbirth in women with sickle cell trait consist of retrospective cohorts with small study populations,” Dr. Krishna added. Notably, the current study was limited by the inability to adjust for comorbidities including diabetes, hypertension, and obesity, that are not only associated with an increased risk for stillbirth, but also disproportionately common among Black women.
“More studies are needed evaluating the relationship between these comorbidities as well as studies specifically evaluating how race affects care and pregnancy outcomes,” Dr. Krisha emphasized.
The study was funded by the University of Pennsylvania department of biostatistics, epidemiology, and informatics. Lead author Dr. Canelón disclosed grants from the Centers for Disease Control and Prevention, Clinical and Translational Science Awards, and grants from the National Institutes of Health outside the submitted work. Dr. Krishna had no financial conflicts to disclose, but serves on the editorial advisory board of Ob.Gyn News.
FROM JAMA NETWORK OPEN