User login
Young lupus patients need more than medications
SAN FRANCISCO – – and therein lies the importance of introducing interventions beyond simply prescribing appropriate medications, Hermine I. Brunner, MD, asserted at an international congress on systemic lupus erythematosus.
Pilot studies conducted by her research group as well as others suggest that brief cognitive-behavioral interventions, web-based patient and caregiver education, and social media interactions significantly improve the fatigue and depression, poor quality of life, and lack of adherence to medication that are pervasive in young patients with SLE, according to Dr. Brunner, director of the division of rheumatology and professor of pediatrics at the University of Cincinnati and scientific director of the Pediatric Rheumatology Collaborative Study Group.
“Don’t misunderstand: I don’t think we can treat lupus simply with a psychological intervention at the bedside. However, I think doctors would be well advised to offer both psychological interventions and medication when they see young lupus patients, because without the psychological intervention the patients may not feel sufficiently at ease to take their medication. They will not get the benefit of the medications you’ve prescribed,” she said.
Patients with SLE take an average of eight medications daily. Their medication adherence rate is comparable to that of patients with diabetes or many other chronic diseases: that is to say, lousy. When investigators at the University of Texas MD Anderson Cancer Center, Houston, utilized an electronic monitoring system to chart adherence to prescribed oral medications in adults with SLE, they found that over the course of 2 years of follow-up only one-fourth of them had an adherence rate of 80% or better, which is the standard definition of adherence (Lupus. 2012 Oct;21[11]:1158-65).
Treatment adherence is particularly problematic in adolescents and young adults with SLE. They often have great difficulty in mastering the self-management skills required to stay on top of their disease when they have so much else going on during what is a vulnerable and challenging period of development, even for healthy youths.
The texting intervention
Dr. Brunner and her colleagues at Cincinnati Children’s Hospital Medical Center recognized the scope of the nonadherence problem early on. Years ago they started sending text messaging reminders of pending clinic visits to their patients who had a poor track record of showing up for appointments.
“We texted patients 2 weeks before their scheduled visit, 1 week before, and then again the day before the visit,” she explained.
This simple intervention resulted in a 47% reduction in missed appointments, compared with a control group. Also, text recipients were more likely to cancel appointments instead of simply not showing up, an important benefit from a practice management and scheduling standpoint (J Rheumatol. 2012 Jan;39[1]:174-9). Disappointingly, however, the text messaging intervention had no impact on adherence to prescribed use of hydroxychloroquine. This led the investigators to conduct a deeper dive into the roots of the nonadherence problem in childhood-onset lupus.
Disease control, quality of life
Dr. Brunner and her coworkers conducted an in-depth assessment of health-related quality of life in 50 patients with childhood-onset SLE over the course of 6 months. The results were surprising.
“When we looked at the correlation between disease control and quality of life, actually there was none,” according to the pediatric rheumatologist.
Instead, the investigators found that young patients with persistently low quality of life despite objectively measured good disease control scored high for fatigue and depressive symptoms (Lupus. 2018 Jan;27[1]:124-33). This led Dr. Brunner and her coinvestigators to consider developing a practical behavioral intervention to address these potentially modifiable predictors of impaired health-related quality of life in their patient population.
The need for novel approaches was highlighted in focus groups conducted by the investigators, in which patients and their primary caregivers emphasized that current therapeutic strategies don’t adequately address key problems of living with lupus, especially the prominent fatigue, pain, and depressed mood that hamper daily function and personal relationships. Patients said they don’t feel an immediate benefit from taking their medications, so why bother? And parents expressed frustration about how difficult it is to get their teenagers to understand the consequences of nonadherence when they’re at an age when they don’t yet even grasp the concept of their own mortality (Lupus. 2019 Mar. doi: 10.1177/0961203319839478. These observations spurred the Cincinnati investigators to develop a modified cognitive-behavioral therapy (CBT) protocol, known as TEACH, which they believe is the first CBT intervention to specifically target psychological problems in young people with childhood-onset SLE.
The TEACH program
TEACH (Treatment and Education Approach for Childhood-Onset Lupus) is a six-session program that teaches patients and caregivers self-advocacy, relaxation techniques, how to improve sleep hygiene, the importance of engaging in planned pleasant activities, and why taking medications matters. The program content differs depending upon whether the patient is an adolescent or young adult.
Results of a recently published small feasibility study were highly encouraging, showing that 83% of people who enrolled in the program completed it. Posttreatment assessment showed that patients had a marked decrease in depressive symptoms as measured by both the Children’s Depression Inventory and the Beck Depression Inventory. They also showed a significant reduction in fatigue. However, while favorable trends in terms of reduced pain and anxiety symptoms were noted, they didn’t achieve statistical significance (Pediatr Rheumatol Online J. 2019 Feb 18. doi: 10.1186/s12969-019-0307-8). The next step in this project is a planned controlled randomized trial.
A web-based medication adherence program
Researchers at Pennsylvania State University took a different approach. They created a publicly available educational website, www.facinglupustogether.com, aimed at improving self-management skills – and especially medication adherence – in teens and young adults with SLE.
The website contains eight modules: Making the transition and taking charge of my medications, Learning about lupus, Learning about lupus medications, Managing symptoms of lupus, How do I handle lupus and my family, How do I handle lupus and my friends, Lupus and stress, and My personal goals and how I will achieve them. Each takes about 10 minutes to complete.
In a pilot study, 37 patients tackled one module per week and were randomized to respond to questions about the weekly topic either in a journal or by discussing the key points in an online social media forum with other young people with SLE. The idea was to create an intervention that capitalizes on the excellent social media skills possessed by today’s youth. And indeed, incorporation of social media proved to be a winning strategy. Medication adherence for hydroxychloroquine in the group randomized to social media participation jumped from 50% in the 3 months prior to starting the program to 92% in the first 3 months post completion, whereas medication adherence didn’t change significantly in the other study arm. The social media group also experienced significant improvements in self-efficacy, sense of community, acceptance of illness, optimism and control over the future, and other measures of empowerment. The control group did not show significant change in any of these domains (Pediatr Rheumatol Online J. 2018 Mar 14. doi: 10.1186/s12969-018-0232-2).
The TEACH study was sponsored by the National Institutes of Health. The web-based medication adherence program pilot study was supported by the Lupus Foundation of America. What the two approaches share in common is a conviction that, when it comes to addressing pain, fatigue, diminished quality of life, and poor medication adherence in young patients with SLE: “Our medication prescription alone doesn’t do it,” Dr. Brunner said.
She reported having no financial conflicts regarding her presentation.
SAN FRANCISCO – – and therein lies the importance of introducing interventions beyond simply prescribing appropriate medications, Hermine I. Brunner, MD, asserted at an international congress on systemic lupus erythematosus.
Pilot studies conducted by her research group as well as others suggest that brief cognitive-behavioral interventions, web-based patient and caregiver education, and social media interactions significantly improve the fatigue and depression, poor quality of life, and lack of adherence to medication that are pervasive in young patients with SLE, according to Dr. Brunner, director of the division of rheumatology and professor of pediatrics at the University of Cincinnati and scientific director of the Pediatric Rheumatology Collaborative Study Group.
“Don’t misunderstand: I don’t think we can treat lupus simply with a psychological intervention at the bedside. However, I think doctors would be well advised to offer both psychological interventions and medication when they see young lupus patients, because without the psychological intervention the patients may not feel sufficiently at ease to take their medication. They will not get the benefit of the medications you’ve prescribed,” she said.
Patients with SLE take an average of eight medications daily. Their medication adherence rate is comparable to that of patients with diabetes or many other chronic diseases: that is to say, lousy. When investigators at the University of Texas MD Anderson Cancer Center, Houston, utilized an electronic monitoring system to chart adherence to prescribed oral medications in adults with SLE, they found that over the course of 2 years of follow-up only one-fourth of them had an adherence rate of 80% or better, which is the standard definition of adherence (Lupus. 2012 Oct;21[11]:1158-65).
Treatment adherence is particularly problematic in adolescents and young adults with SLE. They often have great difficulty in mastering the self-management skills required to stay on top of their disease when they have so much else going on during what is a vulnerable and challenging period of development, even for healthy youths.
The texting intervention
Dr. Brunner and her colleagues at Cincinnati Children’s Hospital Medical Center recognized the scope of the nonadherence problem early on. Years ago they started sending text messaging reminders of pending clinic visits to their patients who had a poor track record of showing up for appointments.
“We texted patients 2 weeks before their scheduled visit, 1 week before, and then again the day before the visit,” she explained.
This simple intervention resulted in a 47% reduction in missed appointments, compared with a control group. Also, text recipients were more likely to cancel appointments instead of simply not showing up, an important benefit from a practice management and scheduling standpoint (J Rheumatol. 2012 Jan;39[1]:174-9). Disappointingly, however, the text messaging intervention had no impact on adherence to prescribed use of hydroxychloroquine. This led the investigators to conduct a deeper dive into the roots of the nonadherence problem in childhood-onset lupus.
Disease control, quality of life
Dr. Brunner and her coworkers conducted an in-depth assessment of health-related quality of life in 50 patients with childhood-onset SLE over the course of 6 months. The results were surprising.
“When we looked at the correlation between disease control and quality of life, actually there was none,” according to the pediatric rheumatologist.
Instead, the investigators found that young patients with persistently low quality of life despite objectively measured good disease control scored high for fatigue and depressive symptoms (Lupus. 2018 Jan;27[1]:124-33). This led Dr. Brunner and her coinvestigators to consider developing a practical behavioral intervention to address these potentially modifiable predictors of impaired health-related quality of life in their patient population.
The need for novel approaches was highlighted in focus groups conducted by the investigators, in which patients and their primary caregivers emphasized that current therapeutic strategies don’t adequately address key problems of living with lupus, especially the prominent fatigue, pain, and depressed mood that hamper daily function and personal relationships. Patients said they don’t feel an immediate benefit from taking their medications, so why bother? And parents expressed frustration about how difficult it is to get their teenagers to understand the consequences of nonadherence when they’re at an age when they don’t yet even grasp the concept of their own mortality (Lupus. 2019 Mar. doi: 10.1177/0961203319839478. These observations spurred the Cincinnati investigators to develop a modified cognitive-behavioral therapy (CBT) protocol, known as TEACH, which they believe is the first CBT intervention to specifically target psychological problems in young people with childhood-onset SLE.
The TEACH program
TEACH (Treatment and Education Approach for Childhood-Onset Lupus) is a six-session program that teaches patients and caregivers self-advocacy, relaxation techniques, how to improve sleep hygiene, the importance of engaging in planned pleasant activities, and why taking medications matters. The program content differs depending upon whether the patient is an adolescent or young adult.
Results of a recently published small feasibility study were highly encouraging, showing that 83% of people who enrolled in the program completed it. Posttreatment assessment showed that patients had a marked decrease in depressive symptoms as measured by both the Children’s Depression Inventory and the Beck Depression Inventory. They also showed a significant reduction in fatigue. However, while favorable trends in terms of reduced pain and anxiety symptoms were noted, they didn’t achieve statistical significance (Pediatr Rheumatol Online J. 2019 Feb 18. doi: 10.1186/s12969-019-0307-8). The next step in this project is a planned controlled randomized trial.
A web-based medication adherence program
Researchers at Pennsylvania State University took a different approach. They created a publicly available educational website, www.facinglupustogether.com, aimed at improving self-management skills – and especially medication adherence – in teens and young adults with SLE.
The website contains eight modules: Making the transition and taking charge of my medications, Learning about lupus, Learning about lupus medications, Managing symptoms of lupus, How do I handle lupus and my family, How do I handle lupus and my friends, Lupus and stress, and My personal goals and how I will achieve them. Each takes about 10 minutes to complete.
In a pilot study, 37 patients tackled one module per week and were randomized to respond to questions about the weekly topic either in a journal or by discussing the key points in an online social media forum with other young people with SLE. The idea was to create an intervention that capitalizes on the excellent social media skills possessed by today’s youth. And indeed, incorporation of social media proved to be a winning strategy. Medication adherence for hydroxychloroquine in the group randomized to social media participation jumped from 50% in the 3 months prior to starting the program to 92% in the first 3 months post completion, whereas medication adherence didn’t change significantly in the other study arm. The social media group also experienced significant improvements in self-efficacy, sense of community, acceptance of illness, optimism and control over the future, and other measures of empowerment. The control group did not show significant change in any of these domains (Pediatr Rheumatol Online J. 2018 Mar 14. doi: 10.1186/s12969-018-0232-2).
The TEACH study was sponsored by the National Institutes of Health. The web-based medication adherence program pilot study was supported by the Lupus Foundation of America. What the two approaches share in common is a conviction that, when it comes to addressing pain, fatigue, diminished quality of life, and poor medication adherence in young patients with SLE: “Our medication prescription alone doesn’t do it,” Dr. Brunner said.
She reported having no financial conflicts regarding her presentation.
SAN FRANCISCO – – and therein lies the importance of introducing interventions beyond simply prescribing appropriate medications, Hermine I. Brunner, MD, asserted at an international congress on systemic lupus erythematosus.
Pilot studies conducted by her research group as well as others suggest that brief cognitive-behavioral interventions, web-based patient and caregiver education, and social media interactions significantly improve the fatigue and depression, poor quality of life, and lack of adherence to medication that are pervasive in young patients with SLE, according to Dr. Brunner, director of the division of rheumatology and professor of pediatrics at the University of Cincinnati and scientific director of the Pediatric Rheumatology Collaborative Study Group.
“Don’t misunderstand: I don’t think we can treat lupus simply with a psychological intervention at the bedside. However, I think doctors would be well advised to offer both psychological interventions and medication when they see young lupus patients, because without the psychological intervention the patients may not feel sufficiently at ease to take their medication. They will not get the benefit of the medications you’ve prescribed,” she said.
Patients with SLE take an average of eight medications daily. Their medication adherence rate is comparable to that of patients with diabetes or many other chronic diseases: that is to say, lousy. When investigators at the University of Texas MD Anderson Cancer Center, Houston, utilized an electronic monitoring system to chart adherence to prescribed oral medications in adults with SLE, they found that over the course of 2 years of follow-up only one-fourth of them had an adherence rate of 80% or better, which is the standard definition of adherence (Lupus. 2012 Oct;21[11]:1158-65).
Treatment adherence is particularly problematic in adolescents and young adults with SLE. They often have great difficulty in mastering the self-management skills required to stay on top of their disease when they have so much else going on during what is a vulnerable and challenging period of development, even for healthy youths.
The texting intervention
Dr. Brunner and her colleagues at Cincinnati Children’s Hospital Medical Center recognized the scope of the nonadherence problem early on. Years ago they started sending text messaging reminders of pending clinic visits to their patients who had a poor track record of showing up for appointments.
“We texted patients 2 weeks before their scheduled visit, 1 week before, and then again the day before the visit,” she explained.
This simple intervention resulted in a 47% reduction in missed appointments, compared with a control group. Also, text recipients were more likely to cancel appointments instead of simply not showing up, an important benefit from a practice management and scheduling standpoint (J Rheumatol. 2012 Jan;39[1]:174-9). Disappointingly, however, the text messaging intervention had no impact on adherence to prescribed use of hydroxychloroquine. This led the investigators to conduct a deeper dive into the roots of the nonadherence problem in childhood-onset lupus.
Disease control, quality of life
Dr. Brunner and her coworkers conducted an in-depth assessment of health-related quality of life in 50 patients with childhood-onset SLE over the course of 6 months. The results were surprising.
“When we looked at the correlation between disease control and quality of life, actually there was none,” according to the pediatric rheumatologist.
Instead, the investigators found that young patients with persistently low quality of life despite objectively measured good disease control scored high for fatigue and depressive symptoms (Lupus. 2018 Jan;27[1]:124-33). This led Dr. Brunner and her coinvestigators to consider developing a practical behavioral intervention to address these potentially modifiable predictors of impaired health-related quality of life in their patient population.
The need for novel approaches was highlighted in focus groups conducted by the investigators, in which patients and their primary caregivers emphasized that current therapeutic strategies don’t adequately address key problems of living with lupus, especially the prominent fatigue, pain, and depressed mood that hamper daily function and personal relationships. Patients said they don’t feel an immediate benefit from taking their medications, so why bother? And parents expressed frustration about how difficult it is to get their teenagers to understand the consequences of nonadherence when they’re at an age when they don’t yet even grasp the concept of their own mortality (Lupus. 2019 Mar. doi: 10.1177/0961203319839478. These observations spurred the Cincinnati investigators to develop a modified cognitive-behavioral therapy (CBT) protocol, known as TEACH, which they believe is the first CBT intervention to specifically target psychological problems in young people with childhood-onset SLE.
The TEACH program
TEACH (Treatment and Education Approach for Childhood-Onset Lupus) is a six-session program that teaches patients and caregivers self-advocacy, relaxation techniques, how to improve sleep hygiene, the importance of engaging in planned pleasant activities, and why taking medications matters. The program content differs depending upon whether the patient is an adolescent or young adult.
Results of a recently published small feasibility study were highly encouraging, showing that 83% of people who enrolled in the program completed it. Posttreatment assessment showed that patients had a marked decrease in depressive symptoms as measured by both the Children’s Depression Inventory and the Beck Depression Inventory. They also showed a significant reduction in fatigue. However, while favorable trends in terms of reduced pain and anxiety symptoms were noted, they didn’t achieve statistical significance (Pediatr Rheumatol Online J. 2019 Feb 18. doi: 10.1186/s12969-019-0307-8). The next step in this project is a planned controlled randomized trial.
A web-based medication adherence program
Researchers at Pennsylvania State University took a different approach. They created a publicly available educational website, www.facinglupustogether.com, aimed at improving self-management skills – and especially medication adherence – in teens and young adults with SLE.
The website contains eight modules: Making the transition and taking charge of my medications, Learning about lupus, Learning about lupus medications, Managing symptoms of lupus, How do I handle lupus and my family, How do I handle lupus and my friends, Lupus and stress, and My personal goals and how I will achieve them. Each takes about 10 minutes to complete.
In a pilot study, 37 patients tackled one module per week and were randomized to respond to questions about the weekly topic either in a journal or by discussing the key points in an online social media forum with other young people with SLE. The idea was to create an intervention that capitalizes on the excellent social media skills possessed by today’s youth. And indeed, incorporation of social media proved to be a winning strategy. Medication adherence for hydroxychloroquine in the group randomized to social media participation jumped from 50% in the 3 months prior to starting the program to 92% in the first 3 months post completion, whereas medication adherence didn’t change significantly in the other study arm. The social media group also experienced significant improvements in self-efficacy, sense of community, acceptance of illness, optimism and control over the future, and other measures of empowerment. The control group did not show significant change in any of these domains (Pediatr Rheumatol Online J. 2018 Mar 14. doi: 10.1186/s12969-018-0232-2).
The TEACH study was sponsored by the National Institutes of Health. The web-based medication adherence program pilot study was supported by the Lupus Foundation of America. What the two approaches share in common is a conviction that, when it comes to addressing pain, fatigue, diminished quality of life, and poor medication adherence in young patients with SLE: “Our medication prescription alone doesn’t do it,” Dr. Brunner said.
She reported having no financial conflicts regarding her presentation.
REPORTING FROM LUPUS 2019
Identifying CMV infection in asymptomatic newborns – one step closer?
Cytomegalovirus (CMV) infection is the most common congenital viral infection in U.S. children, with a frequency between 0.5% and 1% of newborn infants resulting in approximately 30,000 infected children annually. A small minority (approximately 10%) can be identified in the neonatal period as symptomatic with jaundice (from direct hyperbilirubinemia), petechiae (from thrombocytopenia), hepatosplenomegaly, microcephaly, or other manifestations. The vast majority are asymptomatic at birth, yet 15% will have or develop sensorineural hearing loss (SNHL) during the first few years of life; others (1%-2%) will develop vision loss associated with retinal scars. Congenital CMV accounts for 20% of those with SNHL detected at birth and 25% of children with SNHL at 4 years of age.
Screening for congenital CMV has been an ongoing subject of debate. The challenges of implementing screening programs are related both to the diagnostics (collecting urine samples on newborns) as well as with the question of whether we have treatment and interventions to offer babies diagnosed with congenital CMV across the complete spectrum of clinical presentations.
Current screening programs implemented in some hospitals, called “targeted screening,” in which babies who fail newborn screening programs are tested for CMV, are not sufficient to achieve the goal of identifying babies who will need follow-up for early detection of SNHL or vision abnormalities, or possibly early antiviral therapy (Valcyte; valganciclovir), because only a small portion of those who eventually develop SNHL are currently identified by the targeted screening programs.1
However, its availability only has added to the debate as to whether the time has arrived for universal screening.
Vertical transmission of CMV occurs in utero (during any of the trimesters), at birth by passage through the birth canal, or postnatally by ingestion of breast milk. Neonatal infection (in utero and postnatal) occurs in both mothers with primary CMV infection during gestation and in those with recurrent infection (from a different viral strain) or reactivation of infection. Severe clinically symptomatic disease and sequelae is associated with primary maternal infection and early transmission to the fetus. However, it is estimated that nonprimary maternal infection accounts for 75% of neonatal infections. Transmission by breast milk to full-term, healthy infants does not appear to be associated with clinical illness or sequelae; however, preterm infants or those with birth weights less than 1,500 g have a small risk of developing clinical disease.
The polymerase chain reaction–based saliva CMV test (Alethia CMV Assay Test System) was licensed by the Food and Drug Administration in November 2018 after studies demonstrated high sensitivity and specificity, compared with viral culture (the gold standard). In one study, 17,327 infants were screened with the liquid-saliva PCR assay, and 0.5% tested positive for CMV on both the saliva test and culture. Sensitivity and specificity of the liquid-saliva PCR assay were 100% and 99.9%, respectively.2 The availability of an approved saliva-based assay that is both highly sensitive and specific overcomes the challenge of collecting urine, which has been a limiting factor in development of pragmatic universal screening programs. To date, most of the focus in identification of congenital CMV infection has been linking newborn hearing testing programs with CMV testing. For some, these have been labeled “targeted screening programs for CMV.” To us, these appear to be best practice for medical evaluations of an infant with identified SNHL. The availability of saliva-based CMV testing should enable virtually all children who fail newborn screening to be tested for CMV. In multiple studies,3,4 6% of infants with confirmed hearing screen failure tested positive for CMV. A recent study5 identified only 1 infant among the 171 infants who failed newborn screening, however only approximately 15% of the infants were eventually confirmed as hearing impaired at audiology follow-up, suggesting that programmatically testing for CMV might be limited to those with confirmed hearing loss if such can be accomplished within a narrow window of time.
The major challenge with linking CMV testing with newborn hearing screening is whether treatment with valganciclovir would be of value in congenital CMV infection and isolated hearing loss. Studies of children with symptomatic central nervous system congenital CMV disease provide evidence of improvement (or lack of progression) in hearing loss in those treated with valganciclovir. Few, if any of these children had isolated hearing loss in this pivotal study.6 An observational study reported improved outcomes in 55 of 59 (93%) children with congenital CMV and isolated SNHL treated with valganciclovir between birth to 12 weeks of life.7 Hearing improved in nearly 70% of ears, 27% showed no change, and only 3% demonstrated progression of hearing loss; most of the improved ears returned to normal hearing. Currently, a National Institutes of Health study (ValEAR) is recruiting CMV-infected infants with isolated SNHL and randomizing them to treatment with valganciclovir or placebo. The goal is to determine if infants treated with valganciclovir will have better hearing and language outcomes.
Linking CMV testing to those who fail newborn hearing screening programs is an important step, as it appears such children are at least five times more likely to be infected with CMV than is the overall birth cohort. However, such strategies fall short of identifying the majority of newborns with congenital CMV infection, who are completely asymptomatic yet are at risk for development of complications that potentially have substantial impact on their quality of life. Although the availability of sensitive and specific PCR testing in saliva provides a pragmatic approach to identify infected children, many questions remain. First, would a confirmatory test be necessary, such as urine PCR (now considered the gold standard by many CMV experts)? Second, once identified, what regimen for follow-up testing would be indicated to identify those with early SNHL or retinopathy, and until what age? Third, is there a role for treatment in asymptomatic infection? Would that treatment be prophylactic, prior to the development of clinical signs, or implemented once early evidence of SNHL or retinopathy is present?
The Valgan Toddler study – sponsored by NIH and the University of Alabama as part of the Collaborative Antiviral Study Group – will enroll children who are aged 1 month through 3 years and who had a recent diagnosis of hearing loss (within the prior 12 weeks) and evidence of congenital CMV infection. The purpose of this study is to compare the effect on hearing and neurologic outcomes in infants aged 1 month through 4 years with recent onset SNHL who receive 6 weeks of valganciclovir versus children who do not receive this drug. The results of such studies will be critical for the development of best practices.
In summary, the licensure of a rapid PCR-based tool for diagnosis of CMV infection from saliva adds to our ability to develop screening programs to detect asymptomatic infants with congenital CMV infection. The ability to link newborns who fail hearing screening programs with CMV testing will lead to more detection of CMV-infected neonates, both with isolated hearing loss, and subsequently with no signs or symptoms of infection. There is an urgent need for evidence from randomized clinical trials to enable the development of best practices for such infants.
Dr. Pelton is professor of pediatrics and epidemiology at Boston University and senior attending physician at Boston Medical Center. Dr. Lapidot is a senior fellow in pediatric infectious diseases, Boston Medical Center. Neither Dr. Pelton nor Dr. Lapidot have any relevant financial disclosures. Email them at [email protected].
References
1. J Pediatric Infect Dis Soc. 2019 Mar 28;8(1):55-9.
2. N Engl J Med 2011 Jun 2; 364:2111-8.
3. Pediatrics. 2008 May;121(5):970-5
4. J Clin Virol. 2018 May;102:110-5.
5. J Pediatric Infect Dis Soc. 2019 Mar;8(1):55-9.
6. J Pediatr. 2003 Jul;143(1):16-25.
7. J Pediatr. 2018 Aug;199:166-70.
Cytomegalovirus (CMV) infection is the most common congenital viral infection in U.S. children, with a frequency between 0.5% and 1% of newborn infants resulting in approximately 30,000 infected children annually. A small minority (approximately 10%) can be identified in the neonatal period as symptomatic with jaundice (from direct hyperbilirubinemia), petechiae (from thrombocytopenia), hepatosplenomegaly, microcephaly, or other manifestations. The vast majority are asymptomatic at birth, yet 15% will have or develop sensorineural hearing loss (SNHL) during the first few years of life; others (1%-2%) will develop vision loss associated with retinal scars. Congenital CMV accounts for 20% of those with SNHL detected at birth and 25% of children with SNHL at 4 years of age.
Screening for congenital CMV has been an ongoing subject of debate. The challenges of implementing screening programs are related both to the diagnostics (collecting urine samples on newborns) as well as with the question of whether we have treatment and interventions to offer babies diagnosed with congenital CMV across the complete spectrum of clinical presentations.
Current screening programs implemented in some hospitals, called “targeted screening,” in which babies who fail newborn screening programs are tested for CMV, are not sufficient to achieve the goal of identifying babies who will need follow-up for early detection of SNHL or vision abnormalities, or possibly early antiviral therapy (Valcyte; valganciclovir), because only a small portion of those who eventually develop SNHL are currently identified by the targeted screening programs.1
However, its availability only has added to the debate as to whether the time has arrived for universal screening.
Vertical transmission of CMV occurs in utero (during any of the trimesters), at birth by passage through the birth canal, or postnatally by ingestion of breast milk. Neonatal infection (in utero and postnatal) occurs in both mothers with primary CMV infection during gestation and in those with recurrent infection (from a different viral strain) or reactivation of infection. Severe clinically symptomatic disease and sequelae is associated with primary maternal infection and early transmission to the fetus. However, it is estimated that nonprimary maternal infection accounts for 75% of neonatal infections. Transmission by breast milk to full-term, healthy infants does not appear to be associated with clinical illness or sequelae; however, preterm infants or those with birth weights less than 1,500 g have a small risk of developing clinical disease.
The polymerase chain reaction–based saliva CMV test (Alethia CMV Assay Test System) was licensed by the Food and Drug Administration in November 2018 after studies demonstrated high sensitivity and specificity, compared with viral culture (the gold standard). In one study, 17,327 infants were screened with the liquid-saliva PCR assay, and 0.5% tested positive for CMV on both the saliva test and culture. Sensitivity and specificity of the liquid-saliva PCR assay were 100% and 99.9%, respectively.2 The availability of an approved saliva-based assay that is both highly sensitive and specific overcomes the challenge of collecting urine, which has been a limiting factor in development of pragmatic universal screening programs. To date, most of the focus in identification of congenital CMV infection has been linking newborn hearing testing programs with CMV testing. For some, these have been labeled “targeted screening programs for CMV.” To us, these appear to be best practice for medical evaluations of an infant with identified SNHL. The availability of saliva-based CMV testing should enable virtually all children who fail newborn screening to be tested for CMV. In multiple studies,3,4 6% of infants with confirmed hearing screen failure tested positive for CMV. A recent study5 identified only 1 infant among the 171 infants who failed newborn screening, however only approximately 15% of the infants were eventually confirmed as hearing impaired at audiology follow-up, suggesting that programmatically testing for CMV might be limited to those with confirmed hearing loss if such can be accomplished within a narrow window of time.
The major challenge with linking CMV testing with newborn hearing screening is whether treatment with valganciclovir would be of value in congenital CMV infection and isolated hearing loss. Studies of children with symptomatic central nervous system congenital CMV disease provide evidence of improvement (or lack of progression) in hearing loss in those treated with valganciclovir. Few, if any of these children had isolated hearing loss in this pivotal study.6 An observational study reported improved outcomes in 55 of 59 (93%) children with congenital CMV and isolated SNHL treated with valganciclovir between birth to 12 weeks of life.7 Hearing improved in nearly 70% of ears, 27% showed no change, and only 3% demonstrated progression of hearing loss; most of the improved ears returned to normal hearing. Currently, a National Institutes of Health study (ValEAR) is recruiting CMV-infected infants with isolated SNHL and randomizing them to treatment with valganciclovir or placebo. The goal is to determine if infants treated with valganciclovir will have better hearing and language outcomes.
Linking CMV testing to those who fail newborn hearing screening programs is an important step, as it appears such children are at least five times more likely to be infected with CMV than is the overall birth cohort. However, such strategies fall short of identifying the majority of newborns with congenital CMV infection, who are completely asymptomatic yet are at risk for development of complications that potentially have substantial impact on their quality of life. Although the availability of sensitive and specific PCR testing in saliva provides a pragmatic approach to identify infected children, many questions remain. First, would a confirmatory test be necessary, such as urine PCR (now considered the gold standard by many CMV experts)? Second, once identified, what regimen for follow-up testing would be indicated to identify those with early SNHL or retinopathy, and until what age? Third, is there a role for treatment in asymptomatic infection? Would that treatment be prophylactic, prior to the development of clinical signs, or implemented once early evidence of SNHL or retinopathy is present?
The Valgan Toddler study – sponsored by NIH and the University of Alabama as part of the Collaborative Antiviral Study Group – will enroll children who are aged 1 month through 3 years and who had a recent diagnosis of hearing loss (within the prior 12 weeks) and evidence of congenital CMV infection. The purpose of this study is to compare the effect on hearing and neurologic outcomes in infants aged 1 month through 4 years with recent onset SNHL who receive 6 weeks of valganciclovir versus children who do not receive this drug. The results of such studies will be critical for the development of best practices.
In summary, the licensure of a rapid PCR-based tool for diagnosis of CMV infection from saliva adds to our ability to develop screening programs to detect asymptomatic infants with congenital CMV infection. The ability to link newborns who fail hearing screening programs with CMV testing will lead to more detection of CMV-infected neonates, both with isolated hearing loss, and subsequently with no signs or symptoms of infection. There is an urgent need for evidence from randomized clinical trials to enable the development of best practices for such infants.
Dr. Pelton is professor of pediatrics and epidemiology at Boston University and senior attending physician at Boston Medical Center. Dr. Lapidot is a senior fellow in pediatric infectious diseases, Boston Medical Center. Neither Dr. Pelton nor Dr. Lapidot have any relevant financial disclosures. Email them at [email protected].
References
1. J Pediatric Infect Dis Soc. 2019 Mar 28;8(1):55-9.
2. N Engl J Med 2011 Jun 2; 364:2111-8.
3. Pediatrics. 2008 May;121(5):970-5
4. J Clin Virol. 2018 May;102:110-5.
5. J Pediatric Infect Dis Soc. 2019 Mar;8(1):55-9.
6. J Pediatr. 2003 Jul;143(1):16-25.
7. J Pediatr. 2018 Aug;199:166-70.
Cytomegalovirus (CMV) infection is the most common congenital viral infection in U.S. children, with a frequency between 0.5% and 1% of newborn infants resulting in approximately 30,000 infected children annually. A small minority (approximately 10%) can be identified in the neonatal period as symptomatic with jaundice (from direct hyperbilirubinemia), petechiae (from thrombocytopenia), hepatosplenomegaly, microcephaly, or other manifestations. The vast majority are asymptomatic at birth, yet 15% will have or develop sensorineural hearing loss (SNHL) during the first few years of life; others (1%-2%) will develop vision loss associated with retinal scars. Congenital CMV accounts for 20% of those with SNHL detected at birth and 25% of children with SNHL at 4 years of age.
Screening for congenital CMV has been an ongoing subject of debate. The challenges of implementing screening programs are related both to the diagnostics (collecting urine samples on newborns) as well as with the question of whether we have treatment and interventions to offer babies diagnosed with congenital CMV across the complete spectrum of clinical presentations.
Current screening programs implemented in some hospitals, called “targeted screening,” in which babies who fail newborn screening programs are tested for CMV, are not sufficient to achieve the goal of identifying babies who will need follow-up for early detection of SNHL or vision abnormalities, or possibly early antiviral therapy (Valcyte; valganciclovir), because only a small portion of those who eventually develop SNHL are currently identified by the targeted screening programs.1
However, its availability only has added to the debate as to whether the time has arrived for universal screening.
Vertical transmission of CMV occurs in utero (during any of the trimesters), at birth by passage through the birth canal, or postnatally by ingestion of breast milk. Neonatal infection (in utero and postnatal) occurs in both mothers with primary CMV infection during gestation and in those with recurrent infection (from a different viral strain) or reactivation of infection. Severe clinically symptomatic disease and sequelae is associated with primary maternal infection and early transmission to the fetus. However, it is estimated that nonprimary maternal infection accounts for 75% of neonatal infections. Transmission by breast milk to full-term, healthy infants does not appear to be associated with clinical illness or sequelae; however, preterm infants or those with birth weights less than 1,500 g have a small risk of developing clinical disease.
The polymerase chain reaction–based saliva CMV test (Alethia CMV Assay Test System) was licensed by the Food and Drug Administration in November 2018 after studies demonstrated high sensitivity and specificity, compared with viral culture (the gold standard). In one study, 17,327 infants were screened with the liquid-saliva PCR assay, and 0.5% tested positive for CMV on both the saliva test and culture. Sensitivity and specificity of the liquid-saliva PCR assay were 100% and 99.9%, respectively.2 The availability of an approved saliva-based assay that is both highly sensitive and specific overcomes the challenge of collecting urine, which has been a limiting factor in development of pragmatic universal screening programs. To date, most of the focus in identification of congenital CMV infection has been linking newborn hearing testing programs with CMV testing. For some, these have been labeled “targeted screening programs for CMV.” To us, these appear to be best practice for medical evaluations of an infant with identified SNHL. The availability of saliva-based CMV testing should enable virtually all children who fail newborn screening to be tested for CMV. In multiple studies,3,4 6% of infants with confirmed hearing screen failure tested positive for CMV. A recent study5 identified only 1 infant among the 171 infants who failed newborn screening, however only approximately 15% of the infants were eventually confirmed as hearing impaired at audiology follow-up, suggesting that programmatically testing for CMV might be limited to those with confirmed hearing loss if such can be accomplished within a narrow window of time.
The major challenge with linking CMV testing with newborn hearing screening is whether treatment with valganciclovir would be of value in congenital CMV infection and isolated hearing loss. Studies of children with symptomatic central nervous system congenital CMV disease provide evidence of improvement (or lack of progression) in hearing loss in those treated with valganciclovir. Few, if any of these children had isolated hearing loss in this pivotal study.6 An observational study reported improved outcomes in 55 of 59 (93%) children with congenital CMV and isolated SNHL treated with valganciclovir between birth to 12 weeks of life.7 Hearing improved in nearly 70% of ears, 27% showed no change, and only 3% demonstrated progression of hearing loss; most of the improved ears returned to normal hearing. Currently, a National Institutes of Health study (ValEAR) is recruiting CMV-infected infants with isolated SNHL and randomizing them to treatment with valganciclovir or placebo. The goal is to determine if infants treated with valganciclovir will have better hearing and language outcomes.
Linking CMV testing to those who fail newborn hearing screening programs is an important step, as it appears such children are at least five times more likely to be infected with CMV than is the overall birth cohort. However, such strategies fall short of identifying the majority of newborns with congenital CMV infection, who are completely asymptomatic yet are at risk for development of complications that potentially have substantial impact on their quality of life. Although the availability of sensitive and specific PCR testing in saliva provides a pragmatic approach to identify infected children, many questions remain. First, would a confirmatory test be necessary, such as urine PCR (now considered the gold standard by many CMV experts)? Second, once identified, what regimen for follow-up testing would be indicated to identify those with early SNHL or retinopathy, and until what age? Third, is there a role for treatment in asymptomatic infection? Would that treatment be prophylactic, prior to the development of clinical signs, or implemented once early evidence of SNHL or retinopathy is present?
The Valgan Toddler study – sponsored by NIH and the University of Alabama as part of the Collaborative Antiviral Study Group – will enroll children who are aged 1 month through 3 years and who had a recent diagnosis of hearing loss (within the prior 12 weeks) and evidence of congenital CMV infection. The purpose of this study is to compare the effect on hearing and neurologic outcomes in infants aged 1 month through 4 years with recent onset SNHL who receive 6 weeks of valganciclovir versus children who do not receive this drug. The results of such studies will be critical for the development of best practices.
In summary, the licensure of a rapid PCR-based tool for diagnosis of CMV infection from saliva adds to our ability to develop screening programs to detect asymptomatic infants with congenital CMV infection. The ability to link newborns who fail hearing screening programs with CMV testing will lead to more detection of CMV-infected neonates, both with isolated hearing loss, and subsequently with no signs or symptoms of infection. There is an urgent need for evidence from randomized clinical trials to enable the development of best practices for such infants.
Dr. Pelton is professor of pediatrics and epidemiology at Boston University and senior attending physician at Boston Medical Center. Dr. Lapidot is a senior fellow in pediatric infectious diseases, Boston Medical Center. Neither Dr. Pelton nor Dr. Lapidot have any relevant financial disclosures. Email them at [email protected].
References
1. J Pediatric Infect Dis Soc. 2019 Mar 28;8(1):55-9.
2. N Engl J Med 2011 Jun 2; 364:2111-8.
3. Pediatrics. 2008 May;121(5):970-5
4. J Clin Virol. 2018 May;102:110-5.
5. J Pediatric Infect Dis Soc. 2019 Mar;8(1):55-9.
6. J Pediatr. 2003 Jul;143(1):16-25.
7. J Pediatr. 2018 Aug;199:166-70.
USPSTF finds the evidence inconclusive for lead screening in young children, pregnant women
according to a recommendation from the U.S. Preventive Services Task Force.
Elevated blood lead levels are associated with potentially irreversible neurologic problems in children and with organ system impairment and adverse perinatal effects in pregnant women, according to the statement.
“Thus, the primary benefit of screening may be in preventing future exposures or exposure of others to environmental sources,” the task force members wrote in JAMA Pediatrics.
However, the task force issued I statements, meaning that “the current evidence is insufficient to assess the balance of benefits and harms of screening for elevated blood lead levels” in asymptomatic children aged 5 years and younger and in asymptomatic pregnant women.
The task force cited evidence that questionnaires and other clinical prediction tools are inaccurate at identifying elevated blood lead levels in asymptomatic children and pregnant women. In addition, the task force found adequate evidence that capillary blood testing identified elevated blood lead levels in children, but found inadequate evidence that treating elevated blood lead levels was effective in asymptomatic children aged 5 years and younger or in pregnant women.
In the evidence report accompanying the recommendation statement in JAMA Pediatrics, Amy G. Cantor, MD, MPH, of Oregon Health & Science University, Portland, and her colleagues reviewed data from a total of 24 studies including 11,433 individuals.
None of the studies evaluated the risks or benefits of blood lead screening in children. However, in three of four studies, capillary blood lead testing showed sensitivities ranging from 87% to 91% and specificities from 92% to 99%, based on a blood lead level cutoff of 10 mcg/dL or less.
“Evidence indicates that capillary sampling is slightly less sensitive than venous sampling, with comparable specificity,” Dr. Cantor and her colleagues wrote. “Both methods require confirmation.”
There is only limited evidence on whether intervening when children present with elevated blood lead levels results in better neurodevelopmental outcomes. One trial showed beneficial effects of dimercaptosuccinic acid chelation of lowering elevated blood lead levels (20-44 mcg/dL) at 1 year versus placebo, but no clear effect on longer term blood lead levels or neurodevelopmental outcomes, they reported.
For residential interventions, again evidence is limited and blood lead concentrations were not clearly affected. Evidence on calcium and iron interventions was poor quality and insufficient to tell if there was an effect on blood lead levels or clinical outcomes, Dr. Cantor and her colleagues wrote.
No studies of screening for elevated lead levels in pregnant women were identified, nor were studies of health outcomes after interventions to reduce blood lead levels in asymptomatic pregnant women, they noted.
Studies involving pregnant women were limited, and included data on the diagnostic accuracy of a clinical questionnaire and the effects of nutritional intervention during pregnancy, Dr. Cantor and her colleagues wrote.
“This update confirms there are no clear effects of interventions for lowering elevated blood levels in affected children or to improve neurodevelopmental outcomes,” they concluded. “Evidence to determine benefits and harms of screening or treating elevated lead levels during pregnancy remains extremely limited.”
The recommendation updates the last version issued in 2006. The USPSTF is supported by the Agency for Healthcare Research and Quality. The researchers for both articles reported no relevant financial disclosures.
SOURCE: Curry SJ et al. JAMA Pediatr. 2019 Apr 16. doi: 10.1001/jama.2019.3326; Cantor AG et al. JAMA Pediatr. 2019 Apr 16. doi: 10.1001/jama.2019.1004.
“The inconclusive findings of the new USPSTF [U.S. Preventive Services Task Force] recommendation does not mean that screening children for elevated lead levels is not necessary, nor does it shed light on whether screening should be targeted to children at high risk or whether it should be universally done,” Michael Weitzman, MD, wrote in an editorial in response to the USPSTF recommendations.
Dr. Weitzman noted that the recommendation is a consequence of the lack of quality studies on lead level screening, and wrote that, although the recommendations apply to asymptomatic children at both average risk and increased risk, the USPSTF does not recommend for or against screening or that screening be abandoned.
It is standard pediatric practice to counsel parents on lead exposure and screening for elevated blood lead levels in children aged 1-5 years, he wrote, adding that “the American Academy of Pediatrics, Bright Futures, the Centers for Disease Control and Prevention, and Medicaid all recommend universal blood lead screening or the screening of selected children believed to be at especially high risk of exposure at approximately age 1 and 2 years.”
More rigorous research is needed to make definitive recommendations, but in the meantime, clinicians should continue to work with local health departments, housing authorities, and schools to provide care for children with elevated lead levels while continuing with the screening practices recommended by the AAP and other organizations, and advocating for prevention of lead exposure, Dr. Weitzman wrote.
Dr. Weitzman is professor of pediatrics and professor of environmental medicine at New York University. This is a summary of the editorial Dr. Weitzman wrote to accompany the published USPSTF recommendation (JAMA Pediatr. 2019 Apr 16. doi:10.1001/jamapediatrics.2019.0855). He reported no relevant financial disclosures.
“The inconclusive findings of the new USPSTF [U.S. Preventive Services Task Force] recommendation does not mean that screening children for elevated lead levels is not necessary, nor does it shed light on whether screening should be targeted to children at high risk or whether it should be universally done,” Michael Weitzman, MD, wrote in an editorial in response to the USPSTF recommendations.
Dr. Weitzman noted that the recommendation is a consequence of the lack of quality studies on lead level screening, and wrote that, although the recommendations apply to asymptomatic children at both average risk and increased risk, the USPSTF does not recommend for or against screening or that screening be abandoned.
It is standard pediatric practice to counsel parents on lead exposure and screening for elevated blood lead levels in children aged 1-5 years, he wrote, adding that “the American Academy of Pediatrics, Bright Futures, the Centers for Disease Control and Prevention, and Medicaid all recommend universal blood lead screening or the screening of selected children believed to be at especially high risk of exposure at approximately age 1 and 2 years.”
More rigorous research is needed to make definitive recommendations, but in the meantime, clinicians should continue to work with local health departments, housing authorities, and schools to provide care for children with elevated lead levels while continuing with the screening practices recommended by the AAP and other organizations, and advocating for prevention of lead exposure, Dr. Weitzman wrote.
Dr. Weitzman is professor of pediatrics and professor of environmental medicine at New York University. This is a summary of the editorial Dr. Weitzman wrote to accompany the published USPSTF recommendation (JAMA Pediatr. 2019 Apr 16. doi:10.1001/jamapediatrics.2019.0855). He reported no relevant financial disclosures.
“The inconclusive findings of the new USPSTF [U.S. Preventive Services Task Force] recommendation does not mean that screening children for elevated lead levels is not necessary, nor does it shed light on whether screening should be targeted to children at high risk or whether it should be universally done,” Michael Weitzman, MD, wrote in an editorial in response to the USPSTF recommendations.
Dr. Weitzman noted that the recommendation is a consequence of the lack of quality studies on lead level screening, and wrote that, although the recommendations apply to asymptomatic children at both average risk and increased risk, the USPSTF does not recommend for or against screening or that screening be abandoned.
It is standard pediatric practice to counsel parents on lead exposure and screening for elevated blood lead levels in children aged 1-5 years, he wrote, adding that “the American Academy of Pediatrics, Bright Futures, the Centers for Disease Control and Prevention, and Medicaid all recommend universal blood lead screening or the screening of selected children believed to be at especially high risk of exposure at approximately age 1 and 2 years.”
More rigorous research is needed to make definitive recommendations, but in the meantime, clinicians should continue to work with local health departments, housing authorities, and schools to provide care for children with elevated lead levels while continuing with the screening practices recommended by the AAP and other organizations, and advocating for prevention of lead exposure, Dr. Weitzman wrote.
Dr. Weitzman is professor of pediatrics and professor of environmental medicine at New York University. This is a summary of the editorial Dr. Weitzman wrote to accompany the published USPSTF recommendation (JAMA Pediatr. 2019 Apr 16. doi:10.1001/jamapediatrics.2019.0855). He reported no relevant financial disclosures.
according to a recommendation from the U.S. Preventive Services Task Force.
Elevated blood lead levels are associated with potentially irreversible neurologic problems in children and with organ system impairment and adverse perinatal effects in pregnant women, according to the statement.
“Thus, the primary benefit of screening may be in preventing future exposures or exposure of others to environmental sources,” the task force members wrote in JAMA Pediatrics.
However, the task force issued I statements, meaning that “the current evidence is insufficient to assess the balance of benefits and harms of screening for elevated blood lead levels” in asymptomatic children aged 5 years and younger and in asymptomatic pregnant women.
The task force cited evidence that questionnaires and other clinical prediction tools are inaccurate at identifying elevated blood lead levels in asymptomatic children and pregnant women. In addition, the task force found adequate evidence that capillary blood testing identified elevated blood lead levels in children, but found inadequate evidence that treating elevated blood lead levels was effective in asymptomatic children aged 5 years and younger or in pregnant women.
In the evidence report accompanying the recommendation statement in JAMA Pediatrics, Amy G. Cantor, MD, MPH, of Oregon Health & Science University, Portland, and her colleagues reviewed data from a total of 24 studies including 11,433 individuals.
None of the studies evaluated the risks or benefits of blood lead screening in children. However, in three of four studies, capillary blood lead testing showed sensitivities ranging from 87% to 91% and specificities from 92% to 99%, based on a blood lead level cutoff of 10 mcg/dL or less.
“Evidence indicates that capillary sampling is slightly less sensitive than venous sampling, with comparable specificity,” Dr. Cantor and her colleagues wrote. “Both methods require confirmation.”
There is only limited evidence on whether intervening when children present with elevated blood lead levels results in better neurodevelopmental outcomes. One trial showed beneficial effects of dimercaptosuccinic acid chelation of lowering elevated blood lead levels (20-44 mcg/dL) at 1 year versus placebo, but no clear effect on longer term blood lead levels or neurodevelopmental outcomes, they reported.
For residential interventions, again evidence is limited and blood lead concentrations were not clearly affected. Evidence on calcium and iron interventions was poor quality and insufficient to tell if there was an effect on blood lead levels or clinical outcomes, Dr. Cantor and her colleagues wrote.
No studies of screening for elevated lead levels in pregnant women were identified, nor were studies of health outcomes after interventions to reduce blood lead levels in asymptomatic pregnant women, they noted.
Studies involving pregnant women were limited, and included data on the diagnostic accuracy of a clinical questionnaire and the effects of nutritional intervention during pregnancy, Dr. Cantor and her colleagues wrote.
“This update confirms there are no clear effects of interventions for lowering elevated blood levels in affected children or to improve neurodevelopmental outcomes,” they concluded. “Evidence to determine benefits and harms of screening or treating elevated lead levels during pregnancy remains extremely limited.”
The recommendation updates the last version issued in 2006. The USPSTF is supported by the Agency for Healthcare Research and Quality. The researchers for both articles reported no relevant financial disclosures.
SOURCE: Curry SJ et al. JAMA Pediatr. 2019 Apr 16. doi: 10.1001/jama.2019.3326; Cantor AG et al. JAMA Pediatr. 2019 Apr 16. doi: 10.1001/jama.2019.1004.
according to a recommendation from the U.S. Preventive Services Task Force.
Elevated blood lead levels are associated with potentially irreversible neurologic problems in children and with organ system impairment and adverse perinatal effects in pregnant women, according to the statement.
“Thus, the primary benefit of screening may be in preventing future exposures or exposure of others to environmental sources,” the task force members wrote in JAMA Pediatrics.
However, the task force issued I statements, meaning that “the current evidence is insufficient to assess the balance of benefits and harms of screening for elevated blood lead levels” in asymptomatic children aged 5 years and younger and in asymptomatic pregnant women.
The task force cited evidence that questionnaires and other clinical prediction tools are inaccurate at identifying elevated blood lead levels in asymptomatic children and pregnant women. In addition, the task force found adequate evidence that capillary blood testing identified elevated blood lead levels in children, but found inadequate evidence that treating elevated blood lead levels was effective in asymptomatic children aged 5 years and younger or in pregnant women.
In the evidence report accompanying the recommendation statement in JAMA Pediatrics, Amy G. Cantor, MD, MPH, of Oregon Health & Science University, Portland, and her colleagues reviewed data from a total of 24 studies including 11,433 individuals.
None of the studies evaluated the risks or benefits of blood lead screening in children. However, in three of four studies, capillary blood lead testing showed sensitivities ranging from 87% to 91% and specificities from 92% to 99%, based on a blood lead level cutoff of 10 mcg/dL or less.
“Evidence indicates that capillary sampling is slightly less sensitive than venous sampling, with comparable specificity,” Dr. Cantor and her colleagues wrote. “Both methods require confirmation.”
There is only limited evidence on whether intervening when children present with elevated blood lead levels results in better neurodevelopmental outcomes. One trial showed beneficial effects of dimercaptosuccinic acid chelation of lowering elevated blood lead levels (20-44 mcg/dL) at 1 year versus placebo, but no clear effect on longer term blood lead levels or neurodevelopmental outcomes, they reported.
For residential interventions, again evidence is limited and blood lead concentrations were not clearly affected. Evidence on calcium and iron interventions was poor quality and insufficient to tell if there was an effect on blood lead levels or clinical outcomes, Dr. Cantor and her colleagues wrote.
No studies of screening for elevated lead levels in pregnant women were identified, nor were studies of health outcomes after interventions to reduce blood lead levels in asymptomatic pregnant women, they noted.
Studies involving pregnant women were limited, and included data on the diagnostic accuracy of a clinical questionnaire and the effects of nutritional intervention during pregnancy, Dr. Cantor and her colleagues wrote.
“This update confirms there are no clear effects of interventions for lowering elevated blood levels in affected children or to improve neurodevelopmental outcomes,” they concluded. “Evidence to determine benefits and harms of screening or treating elevated lead levels during pregnancy remains extremely limited.”
The recommendation updates the last version issued in 2006. The USPSTF is supported by the Agency for Healthcare Research and Quality. The researchers for both articles reported no relevant financial disclosures.
SOURCE: Curry SJ et al. JAMA Pediatr. 2019 Apr 16. doi: 10.1001/jama.2019.3326; Cantor AG et al. JAMA Pediatr. 2019 Apr 16. doi: 10.1001/jama.2019.1004.
FROM JAMA PEDIATRICS
Busiest week yet brings 2019 measles total to 555 cases
according to the Centers for Disease Control and Prevention.
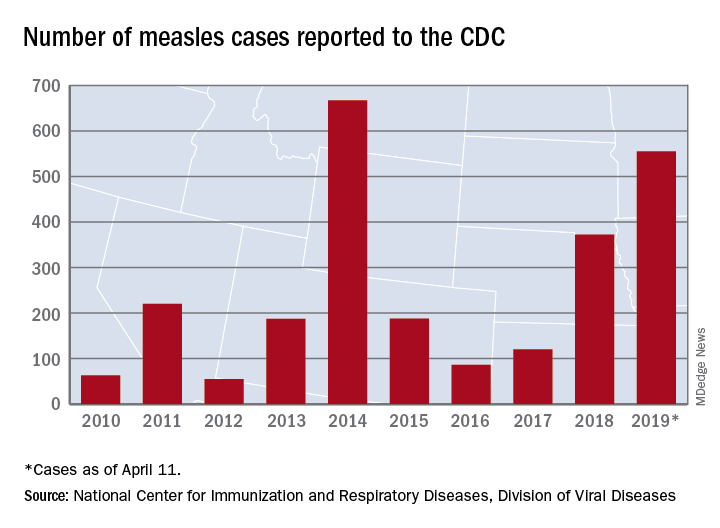
The 90 measles cases reported during the week ending April 11 mark the third consecutive weekly high for 2019, topping the 78 recorded during the week of April 4 and the 73 reported during the week of March 28. Meanwhile, this year’s total trails only the 667 cases reported in 2014 for the highest in the postelimination era, the CDC said April 15.
New York reported 26 new cases in Brooklyn’s Williamsburg neighborhood last week, which puts the borough at 227 for the year, with another two occurring in the Flushing section of Queens. A public health emergency declared on April 9 covers several zip codes in Williamsburg and requires unvaccinated individuals who may have been exposed to measles to receive “the measles-mumps-rubella vaccine in order to protect others in the community and help curtail the ongoing outbreak,” the city’s health department said in a written statement.
Maryland became the 20th state to report a measles case this year, and the state’s department of health said it was notifying those in the vicinity of a medical office building in Pikesville about possible exposure on April 2.
The recent outbreak in Michigan’s Oakland County did not result in any new patients over the last week and remains at 38 cases, with the state reporting one additional case in Wayne County. More recent reports of a case in Washtenaw County and another in Oakland County were reversed after additional testing, the state health department reported.
according to the Centers for Disease Control and Prevention.

The 90 measles cases reported during the week ending April 11 mark the third consecutive weekly high for 2019, topping the 78 recorded during the week of April 4 and the 73 reported during the week of March 28. Meanwhile, this year’s total trails only the 667 cases reported in 2014 for the highest in the postelimination era, the CDC said April 15.
New York reported 26 new cases in Brooklyn’s Williamsburg neighborhood last week, which puts the borough at 227 for the year, with another two occurring in the Flushing section of Queens. A public health emergency declared on April 9 covers several zip codes in Williamsburg and requires unvaccinated individuals who may have been exposed to measles to receive “the measles-mumps-rubella vaccine in order to protect others in the community and help curtail the ongoing outbreak,” the city’s health department said in a written statement.
Maryland became the 20th state to report a measles case this year, and the state’s department of health said it was notifying those in the vicinity of a medical office building in Pikesville about possible exposure on April 2.
The recent outbreak in Michigan’s Oakland County did not result in any new patients over the last week and remains at 38 cases, with the state reporting one additional case in Wayne County. More recent reports of a case in Washtenaw County and another in Oakland County were reversed after additional testing, the state health department reported.
according to the Centers for Disease Control and Prevention.

The 90 measles cases reported during the week ending April 11 mark the third consecutive weekly high for 2019, topping the 78 recorded during the week of April 4 and the 73 reported during the week of March 28. Meanwhile, this year’s total trails only the 667 cases reported in 2014 for the highest in the postelimination era, the CDC said April 15.
New York reported 26 new cases in Brooklyn’s Williamsburg neighborhood last week, which puts the borough at 227 for the year, with another two occurring in the Flushing section of Queens. A public health emergency declared on April 9 covers several zip codes in Williamsburg and requires unvaccinated individuals who may have been exposed to measles to receive “the measles-mumps-rubella vaccine in order to protect others in the community and help curtail the ongoing outbreak,” the city’s health department said in a written statement.
Maryland became the 20th state to report a measles case this year, and the state’s department of health said it was notifying those in the vicinity of a medical office building in Pikesville about possible exposure on April 2.
The recent outbreak in Michigan’s Oakland County did not result in any new patients over the last week and remains at 38 cases, with the state reporting one additional case in Wayne County. More recent reports of a case in Washtenaw County and another in Oakland County were reversed after additional testing, the state health department reported.
Bariatric surgery viable for teens with cognitive disabilities
Adolescents with severe obesity and cognitive impairment or developmental delay (CI/DD) lost as much weight, and at a similar rate, as their typically developing peers following laparoscopic sleeve gastrectomy (LSG), according in an observational study.
“On the basis of these new data, LSG appears to be a viable and successful short-term weight-management tool for adolescents with CI/DD, who are established as particularly vulnerable to obesity and secondary health concerns,” wrote Sarah E. Hornack, PhD, a psychologist with Children’s National Health System and George Washington University, both in Washington, and her associates.
“In fact, there may be advantages to undergoing surgery during adolescence rather than waiting until adulthood for this population,” they wrote in the journal Pediatrics. With more supports likely in place for teens undergoing this surgery, they won’t be “going it alone,” the authors noted, which “could translate to better cooperation with parental guidance regarding surgery requirements, including diet and exercise recommendations.”
Despite higher rates of obesity and related medical issues among youth with CI/DD, little research explores effective interventions in this population, the authors wrote.
They therefore compared outcomes among a group of 63 teens with obesity who underwent bariatric surgery during 2010-2017. The adolescents, who had a body mass index (BMI) of at least 40 kg/m2 or one of at least 35 kg/m2 with a medical comorbidity, first underwent preoperative psychological evaluations involving a cognitive assessment. The 17 adolescents with an IQ less than 80 were classified as having CI/DD, leaving 46 without CI/DD. Three teens had Down syndrome.
Age, sex, and BMI before surgery were similar in those with CI/DD versus those without. The majority of participants overall were female (65%) and black (57%) with an average age of 17 years and an average BMI of 51.2. Whites comprised 24% of participants while 17% were Hispanic and 1% another race/ethnicity.
The findings revealed that IQ did not predict weight loss. The percentage of excess BMI lost (%EBMIL) and rate of excess weight loss remained similar between those with and without CI/DD, though “a trend for a higher rate of change in %EBMIL for those individuals with CI/DD” suggested “they may experience greater rates of weight loss over time than their typically developing peers,” the authors reported. However, the proportion of participants assessed decreased with each follow-up, from 59 at 3 months to 14 at 24 months.
In addition to the small population, short-term follow-up and loss to follow-up, another study limitation is the lack of a control group of CI/DD patients who did not undergo bariatric surgery and instead received a behavioral intervention or other therapy.
But the authors noted existing evidence that “younger children respond better to behavioral interventions than adolescents do, suggesting that older youth may require a different treatment approach.” In addition, “bariatric surgery performed earlier in the trajectory of large weight gain has also been shown to lead to greater resolution of obesity, suggesting that waiting for adulthood can be detrimental,” they wrote.
SOURCE: Hornack SE et al. Pediatrics. 2019 Apr 15. doi: 10.1542/peds.2018-2908.
Despite increasing evidence to support the safety and effectiveness of bariatric surgery in confronting the challenge of increasing obesity rates among adolescents, access to care remains limited for many such teens.
Prominent examples include a significant disparity in insurance authorization for bariatric surgical care when comparing pediatric patients to their adult counterparts, low rates of referral from primary caregivers, and general uncertainty regarding potential exclusionary criteria.
The researchers should be commended for exploring bariatric surgery outcomes in an understudied population. However, both the likely importance of social supports to the participants’ success and, especially, the need to approach the issue of informed thoughtfully, perhaps with additional institutional guidance are crucial to success.
Although literature addressing ethical concerns specifically associated with bariatric surgery for children with intellectual or developmental disability is limited, previous attempts to offer a logical clinical framework highlight the importance of using a case-by-case approach predicated on the need to establish a well-defined risk/benefit ratio.
As an important part of efforts to tackle such challenges, bariatric surgical care providers should strongly consider the routine use of available resources (i.e., institutional ethics committees) to assist in complex medical decision making.”
These comments are adapted from an accompanying editorial by Marc P. Michalsky, MD, of the Ohio State University and Nationwide Children’s Hospital, both in Columbus (Pediatrics. 15 April 2019; doi: 10.1542/peds.2018-4112). He reported having no disclosures.
Despite increasing evidence to support the safety and effectiveness of bariatric surgery in confronting the challenge of increasing obesity rates among adolescents, access to care remains limited for many such teens.
Prominent examples include a significant disparity in insurance authorization for bariatric surgical care when comparing pediatric patients to their adult counterparts, low rates of referral from primary caregivers, and general uncertainty regarding potential exclusionary criteria.
The researchers should be commended for exploring bariatric surgery outcomes in an understudied population. However, both the likely importance of social supports to the participants’ success and, especially, the need to approach the issue of informed thoughtfully, perhaps with additional institutional guidance are crucial to success.
Although literature addressing ethical concerns specifically associated with bariatric surgery for children with intellectual or developmental disability is limited, previous attempts to offer a logical clinical framework highlight the importance of using a case-by-case approach predicated on the need to establish a well-defined risk/benefit ratio.
As an important part of efforts to tackle such challenges, bariatric surgical care providers should strongly consider the routine use of available resources (i.e., institutional ethics committees) to assist in complex medical decision making.”
These comments are adapted from an accompanying editorial by Marc P. Michalsky, MD, of the Ohio State University and Nationwide Children’s Hospital, both in Columbus (Pediatrics. 15 April 2019; doi: 10.1542/peds.2018-4112). He reported having no disclosures.
Despite increasing evidence to support the safety and effectiveness of bariatric surgery in confronting the challenge of increasing obesity rates among adolescents, access to care remains limited for many such teens.
Prominent examples include a significant disparity in insurance authorization for bariatric surgical care when comparing pediatric patients to their adult counterparts, low rates of referral from primary caregivers, and general uncertainty regarding potential exclusionary criteria.
The researchers should be commended for exploring bariatric surgery outcomes in an understudied population. However, both the likely importance of social supports to the participants’ success and, especially, the need to approach the issue of informed thoughtfully, perhaps with additional institutional guidance are crucial to success.
Although literature addressing ethical concerns specifically associated with bariatric surgery for children with intellectual or developmental disability is limited, previous attempts to offer a logical clinical framework highlight the importance of using a case-by-case approach predicated on the need to establish a well-defined risk/benefit ratio.
As an important part of efforts to tackle such challenges, bariatric surgical care providers should strongly consider the routine use of available resources (i.e., institutional ethics committees) to assist in complex medical decision making.”
These comments are adapted from an accompanying editorial by Marc P. Michalsky, MD, of the Ohio State University and Nationwide Children’s Hospital, both in Columbus (Pediatrics. 15 April 2019; doi: 10.1542/peds.2018-4112). He reported having no disclosures.
Adolescents with severe obesity and cognitive impairment or developmental delay (CI/DD) lost as much weight, and at a similar rate, as their typically developing peers following laparoscopic sleeve gastrectomy (LSG), according in an observational study.
“On the basis of these new data, LSG appears to be a viable and successful short-term weight-management tool for adolescents with CI/DD, who are established as particularly vulnerable to obesity and secondary health concerns,” wrote Sarah E. Hornack, PhD, a psychologist with Children’s National Health System and George Washington University, both in Washington, and her associates.
“In fact, there may be advantages to undergoing surgery during adolescence rather than waiting until adulthood for this population,” they wrote in the journal Pediatrics. With more supports likely in place for teens undergoing this surgery, they won’t be “going it alone,” the authors noted, which “could translate to better cooperation with parental guidance regarding surgery requirements, including diet and exercise recommendations.”
Despite higher rates of obesity and related medical issues among youth with CI/DD, little research explores effective interventions in this population, the authors wrote.
They therefore compared outcomes among a group of 63 teens with obesity who underwent bariatric surgery during 2010-2017. The adolescents, who had a body mass index (BMI) of at least 40 kg/m2 or one of at least 35 kg/m2 with a medical comorbidity, first underwent preoperative psychological evaluations involving a cognitive assessment. The 17 adolescents with an IQ less than 80 were classified as having CI/DD, leaving 46 without CI/DD. Three teens had Down syndrome.
Age, sex, and BMI before surgery were similar in those with CI/DD versus those without. The majority of participants overall were female (65%) and black (57%) with an average age of 17 years and an average BMI of 51.2. Whites comprised 24% of participants while 17% were Hispanic and 1% another race/ethnicity.
The findings revealed that IQ did not predict weight loss. The percentage of excess BMI lost (%EBMIL) and rate of excess weight loss remained similar between those with and without CI/DD, though “a trend for a higher rate of change in %EBMIL for those individuals with CI/DD” suggested “they may experience greater rates of weight loss over time than their typically developing peers,” the authors reported. However, the proportion of participants assessed decreased with each follow-up, from 59 at 3 months to 14 at 24 months.
In addition to the small population, short-term follow-up and loss to follow-up, another study limitation is the lack of a control group of CI/DD patients who did not undergo bariatric surgery and instead received a behavioral intervention or other therapy.
But the authors noted existing evidence that “younger children respond better to behavioral interventions than adolescents do, suggesting that older youth may require a different treatment approach.” In addition, “bariatric surgery performed earlier in the trajectory of large weight gain has also been shown to lead to greater resolution of obesity, suggesting that waiting for adulthood can be detrimental,” they wrote.
SOURCE: Hornack SE et al. Pediatrics. 2019 Apr 15. doi: 10.1542/peds.2018-2908.
Adolescents with severe obesity and cognitive impairment or developmental delay (CI/DD) lost as much weight, and at a similar rate, as their typically developing peers following laparoscopic sleeve gastrectomy (LSG), according in an observational study.
“On the basis of these new data, LSG appears to be a viable and successful short-term weight-management tool for adolescents with CI/DD, who are established as particularly vulnerable to obesity and secondary health concerns,” wrote Sarah E. Hornack, PhD, a psychologist with Children’s National Health System and George Washington University, both in Washington, and her associates.
“In fact, there may be advantages to undergoing surgery during adolescence rather than waiting until adulthood for this population,” they wrote in the journal Pediatrics. With more supports likely in place for teens undergoing this surgery, they won’t be “going it alone,” the authors noted, which “could translate to better cooperation with parental guidance regarding surgery requirements, including diet and exercise recommendations.”
Despite higher rates of obesity and related medical issues among youth with CI/DD, little research explores effective interventions in this population, the authors wrote.
They therefore compared outcomes among a group of 63 teens with obesity who underwent bariatric surgery during 2010-2017. The adolescents, who had a body mass index (BMI) of at least 40 kg/m2 or one of at least 35 kg/m2 with a medical comorbidity, first underwent preoperative psychological evaluations involving a cognitive assessment. The 17 adolescents with an IQ less than 80 were classified as having CI/DD, leaving 46 without CI/DD. Three teens had Down syndrome.
Age, sex, and BMI before surgery were similar in those with CI/DD versus those without. The majority of participants overall were female (65%) and black (57%) with an average age of 17 years and an average BMI of 51.2. Whites comprised 24% of participants while 17% were Hispanic and 1% another race/ethnicity.
The findings revealed that IQ did not predict weight loss. The percentage of excess BMI lost (%EBMIL) and rate of excess weight loss remained similar between those with and without CI/DD, though “a trend for a higher rate of change in %EBMIL for those individuals with CI/DD” suggested “they may experience greater rates of weight loss over time than their typically developing peers,” the authors reported. However, the proportion of participants assessed decreased with each follow-up, from 59 at 3 months to 14 at 24 months.
In addition to the small population, short-term follow-up and loss to follow-up, another study limitation is the lack of a control group of CI/DD patients who did not undergo bariatric surgery and instead received a behavioral intervention or other therapy.
But the authors noted existing evidence that “younger children respond better to behavioral interventions than adolescents do, suggesting that older youth may require a different treatment approach.” In addition, “bariatric surgery performed earlier in the trajectory of large weight gain has also been shown to lead to greater resolution of obesity, suggesting that waiting for adulthood can be detrimental,” they wrote.
SOURCE: Hornack SE et al. Pediatrics. 2019 Apr 15. doi: 10.1542/peds.2018-2908.
FROM PEDIATRICS
FDA warns of possible temporary shortage of trach tube
manufactured by Smiths Medical caused by the closure of a large ethylene oxide sterilization facilities in Willowbrook, Ill., and the future planned closure of a similar facility.
The shortage may affect pediatric use because, although tubes are used for both adults and children, there are fewer alternative products on the market for pediatric patients. Parents and caregivers of children who use the Bivona tube are encouraged to check with Smiths Medical about available inventory and with their health care providers about alternative products.
Jeff Shuren, MD, director of the Center for Devices and Radiological Health, wrote in a press release, “I want to assure you that the FDA is working closely with the company to quickly resolve their sterilization challenges and bring these critical devices to the patients who need them as quickly as possible, which we anticipate will be made available again beginning the week of April 22.”
For patients currently using the Bivona tubes, Dr. Shuren noted, “The closure of the Willowbrook facility does not impact tubes already in use by patients at home or in health care settings. The company is communicating with patients about the tubes and how patients and caregivers can mitigate any potential impact, including reusing and cleaning tubes in accordance with the manufacturer’s instructions for use.”
Read the entire announcement at the FDA website.
manufactured by Smiths Medical caused by the closure of a large ethylene oxide sterilization facilities in Willowbrook, Ill., and the future planned closure of a similar facility.
The shortage may affect pediatric use because, although tubes are used for both adults and children, there are fewer alternative products on the market for pediatric patients. Parents and caregivers of children who use the Bivona tube are encouraged to check with Smiths Medical about available inventory and with their health care providers about alternative products.
Jeff Shuren, MD, director of the Center for Devices and Radiological Health, wrote in a press release, “I want to assure you that the FDA is working closely with the company to quickly resolve their sterilization challenges and bring these critical devices to the patients who need them as quickly as possible, which we anticipate will be made available again beginning the week of April 22.”
For patients currently using the Bivona tubes, Dr. Shuren noted, “The closure of the Willowbrook facility does not impact tubes already in use by patients at home or in health care settings. The company is communicating with patients about the tubes and how patients and caregivers can mitigate any potential impact, including reusing and cleaning tubes in accordance with the manufacturer’s instructions for use.”
Read the entire announcement at the FDA website.
manufactured by Smiths Medical caused by the closure of a large ethylene oxide sterilization facilities in Willowbrook, Ill., and the future planned closure of a similar facility.
The shortage may affect pediatric use because, although tubes are used for both adults and children, there are fewer alternative products on the market for pediatric patients. Parents and caregivers of children who use the Bivona tube are encouraged to check with Smiths Medical about available inventory and with their health care providers about alternative products.
Jeff Shuren, MD, director of the Center for Devices and Radiological Health, wrote in a press release, “I want to assure you that the FDA is working closely with the company to quickly resolve their sterilization challenges and bring these critical devices to the patients who need them as quickly as possible, which we anticipate will be made available again beginning the week of April 22.”
For patients currently using the Bivona tubes, Dr. Shuren noted, “The closure of the Willowbrook facility does not impact tubes already in use by patients at home or in health care settings. The company is communicating with patients about the tubes and how patients and caregivers can mitigate any potential impact, including reusing and cleaning tubes in accordance with the manufacturer’s instructions for use.”
Read the entire announcement at the FDA website.
A chance to unite
Is America coming apart at the seams? According to the press, there are more things that divide us than bind us together. It’s red state versus blue state, it’s the privileged versus the disadvantaged, people of color versus the white majority. Could the great melting pot have cooled and its contents settled out into a dozen stratified layers?
Despite the image of a divided America that we see portrayed in the newspapers and on television, I continue to believe that there is more that we share in common than separate us, but it’s a struggle. The media operate on the assumption that conflict draws more readers than good news about cooperation and compromise. The situation is compounded by the apparent absence of a leader from either party who wants to unite us.
However, when one scratches the surface, there is surprising amount of agreement among Americans. For example, according to John Gramlich (“7 facts about guns in the U.S.,” Pew Research Center, Dec. 27, 2018), 89% of both Republicans and Democrats feel that people with mental illness should not be allowed to purchase a gun. And 79% of Republicans and 91% of Democrats favor background checks at gun shows and for private sales for purchase of a gun. As of 2018, 58% of Americans feel that abortion should be legal in all or most cases, and only 37% feel it should be illegal in all or most cases. (“Public Opinion on Abortion,” Pew Research Center, Oct. 15, 2018).
At the core of many of our struggles to unite is a question that has bedeviled democracies for millennia: How does one balance a citizen’s freedom of choice with the health and safety of the society in which that person lives? While resolutions on gun control and abortion seem unlikely in the foreseeable future, the current outbreaks of measles offer America a rare opportunity to unite on an issue that pits personal freedom against societal safety.
According to Virginia Villa (“5 facts about vaccines in the U.S.,” Pew Research Center, Mar. 19, 2019), 82% of adults in the United States believe that the MMR vaccine should be required for public school attendance, while only 17% believe that parents should be allowed to leave their child unvaccinated even if their decision creates a health risk for other children and adults.
Why should we expect the government to respond to protect the population from the risk posed by the unvaccinated minority when it has done very little to further gun control? Obviously a key difference is that the antivaccination minority lacks the financial resources and political muscle of a large organization such as the National Rifle Association. While we must never underestimate the power of social media, the publicity surfacing from the mainstream media as the measles outbreaks in the United States have continued has prompted several states to rethink their policies regarding vaccination requirements and school attendance. Here in Maine, there has been strong support among the legislature for eliminating exemptions for philosophic or religious exemptions.
It is probably unrealistic to expect the federal government to act on the health threat caused by the antivaccine movement. However, it is encouraging that, at least at the local level, there is hope for closing one of the wounds that divide us. As providers who care for children, we should seize this opportunity created by the measles outbreaks to promote legislation and policies that strike a sensible balance between the right of the individual and the safety of the society at large.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Email him at [email protected].
Is America coming apart at the seams? According to the press, there are more things that divide us than bind us together. It’s red state versus blue state, it’s the privileged versus the disadvantaged, people of color versus the white majority. Could the great melting pot have cooled and its contents settled out into a dozen stratified layers?
Despite the image of a divided America that we see portrayed in the newspapers and on television, I continue to believe that there is more that we share in common than separate us, but it’s a struggle. The media operate on the assumption that conflict draws more readers than good news about cooperation and compromise. The situation is compounded by the apparent absence of a leader from either party who wants to unite us.
However, when one scratches the surface, there is surprising amount of agreement among Americans. For example, according to John Gramlich (“7 facts about guns in the U.S.,” Pew Research Center, Dec. 27, 2018), 89% of both Republicans and Democrats feel that people with mental illness should not be allowed to purchase a gun. And 79% of Republicans and 91% of Democrats favor background checks at gun shows and for private sales for purchase of a gun. As of 2018, 58% of Americans feel that abortion should be legal in all or most cases, and only 37% feel it should be illegal in all or most cases. (“Public Opinion on Abortion,” Pew Research Center, Oct. 15, 2018).
At the core of many of our struggles to unite is a question that has bedeviled democracies for millennia: How does one balance a citizen’s freedom of choice with the health and safety of the society in which that person lives? While resolutions on gun control and abortion seem unlikely in the foreseeable future, the current outbreaks of measles offer America a rare opportunity to unite on an issue that pits personal freedom against societal safety.
According to Virginia Villa (“5 facts about vaccines in the U.S.,” Pew Research Center, Mar. 19, 2019), 82% of adults in the United States believe that the MMR vaccine should be required for public school attendance, while only 17% believe that parents should be allowed to leave their child unvaccinated even if their decision creates a health risk for other children and adults.
Why should we expect the government to respond to protect the population from the risk posed by the unvaccinated minority when it has done very little to further gun control? Obviously a key difference is that the antivaccination minority lacks the financial resources and political muscle of a large organization such as the National Rifle Association. While we must never underestimate the power of social media, the publicity surfacing from the mainstream media as the measles outbreaks in the United States have continued has prompted several states to rethink their policies regarding vaccination requirements and school attendance. Here in Maine, there has been strong support among the legislature for eliminating exemptions for philosophic or religious exemptions.
It is probably unrealistic to expect the federal government to act on the health threat caused by the antivaccine movement. However, it is encouraging that, at least at the local level, there is hope for closing one of the wounds that divide us. As providers who care for children, we should seize this opportunity created by the measles outbreaks to promote legislation and policies that strike a sensible balance between the right of the individual and the safety of the society at large.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Email him at [email protected].
Is America coming apart at the seams? According to the press, there are more things that divide us than bind us together. It’s red state versus blue state, it’s the privileged versus the disadvantaged, people of color versus the white majority. Could the great melting pot have cooled and its contents settled out into a dozen stratified layers?
Despite the image of a divided America that we see portrayed in the newspapers and on television, I continue to believe that there is more that we share in common than separate us, but it’s a struggle. The media operate on the assumption that conflict draws more readers than good news about cooperation and compromise. The situation is compounded by the apparent absence of a leader from either party who wants to unite us.
However, when one scratches the surface, there is surprising amount of agreement among Americans. For example, according to John Gramlich (“7 facts about guns in the U.S.,” Pew Research Center, Dec. 27, 2018), 89% of both Republicans and Democrats feel that people with mental illness should not be allowed to purchase a gun. And 79% of Republicans and 91% of Democrats favor background checks at gun shows and for private sales for purchase of a gun. As of 2018, 58% of Americans feel that abortion should be legal in all or most cases, and only 37% feel it should be illegal in all or most cases. (“Public Opinion on Abortion,” Pew Research Center, Oct. 15, 2018).
At the core of many of our struggles to unite is a question that has bedeviled democracies for millennia: How does one balance a citizen’s freedom of choice with the health and safety of the society in which that person lives? While resolutions on gun control and abortion seem unlikely in the foreseeable future, the current outbreaks of measles offer America a rare opportunity to unite on an issue that pits personal freedom against societal safety.
According to Virginia Villa (“5 facts about vaccines in the U.S.,” Pew Research Center, Mar. 19, 2019), 82% of adults in the United States believe that the MMR vaccine should be required for public school attendance, while only 17% believe that parents should be allowed to leave their child unvaccinated even if their decision creates a health risk for other children and adults.
Why should we expect the government to respond to protect the population from the risk posed by the unvaccinated minority when it has done very little to further gun control? Obviously a key difference is that the antivaccination minority lacks the financial resources and political muscle of a large organization such as the National Rifle Association. While we must never underestimate the power of social media, the publicity surfacing from the mainstream media as the measles outbreaks in the United States have continued has prompted several states to rethink their policies regarding vaccination requirements and school attendance. Here in Maine, there has been strong support among the legislature for eliminating exemptions for philosophic or religious exemptions.
It is probably unrealistic to expect the federal government to act on the health threat caused by the antivaccine movement. However, it is encouraging that, at least at the local level, there is hope for closing one of the wounds that divide us. As providers who care for children, we should seize this opportunity created by the measles outbreaks to promote legislation and policies that strike a sensible balance between the right of the individual and the safety of the society at large.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Email him at [email protected].
Rate of objects ingested by young children increased over last two decades
– from an estimated 9 cases per 10,000 children to 18 cases per 10,000 (R2 = 0.90; P less than .001) – according to an analysis in Pediatrics.
The analysis was conducted by Danielle Orsagh-Yentis, MD, of Vanderbilt University, Nashville, Tenn., and her colleagues. They estimated that, during the study period, 759,074 children younger than 6 years of age were evaluated in U.S. EDs for suspected or confirmed foreign-body ingestions. These estimates were based on data for 29,893 actual cases taken from the National Electronic Injury Surveillance System (NEISS), which represents about 100 hospitals. Each case in this system is given a sample weight by the Consumer Product Safety Commission using a validated method, and the estimates are based on this weighting.
The analysis showed that children aged 1 year (21%) and boys (53%) were the most likely to ingest foreign bodies. Coins were the most frequently ingested objects, at 62%. Among cases which had the location noted (59%), most ingestions occurred in the home (97%).
The authors noted that, although batteries and magnets represented only 7% and 2% of all cases, respectively, “they can both enact considerable damage when ingested.” For example, despite being only the fourth mostly likely object to be ingested, batteries were the second mostly likely to be implicated among hospitalized patients.
The authors noted that the NEISS captures patients in the ED only; the total number of foreign-body ingestions, then, was likely underestimated. Despite this, the authors felt the long study period and large sample were strengths of their analysis.
Dr. Orsagh-Yentis and her associates disclosed no potential conflicts of interest.
SOURCE: Orsagh-Yentis D et al. Pediatrics. 2019 Apr 12. doi: 10.1542/peds.2018-1988.
– from an estimated 9 cases per 10,000 children to 18 cases per 10,000 (R2 = 0.90; P less than .001) – according to an analysis in Pediatrics.
The analysis was conducted by Danielle Orsagh-Yentis, MD, of Vanderbilt University, Nashville, Tenn., and her colleagues. They estimated that, during the study period, 759,074 children younger than 6 years of age were evaluated in U.S. EDs for suspected or confirmed foreign-body ingestions. These estimates were based on data for 29,893 actual cases taken from the National Electronic Injury Surveillance System (NEISS), which represents about 100 hospitals. Each case in this system is given a sample weight by the Consumer Product Safety Commission using a validated method, and the estimates are based on this weighting.
The analysis showed that children aged 1 year (21%) and boys (53%) were the most likely to ingest foreign bodies. Coins were the most frequently ingested objects, at 62%. Among cases which had the location noted (59%), most ingestions occurred in the home (97%).
The authors noted that, although batteries and magnets represented only 7% and 2% of all cases, respectively, “they can both enact considerable damage when ingested.” For example, despite being only the fourth mostly likely object to be ingested, batteries were the second mostly likely to be implicated among hospitalized patients.
The authors noted that the NEISS captures patients in the ED only; the total number of foreign-body ingestions, then, was likely underestimated. Despite this, the authors felt the long study period and large sample were strengths of their analysis.
Dr. Orsagh-Yentis and her associates disclosed no potential conflicts of interest.
SOURCE: Orsagh-Yentis D et al. Pediatrics. 2019 Apr 12. doi: 10.1542/peds.2018-1988.
– from an estimated 9 cases per 10,000 children to 18 cases per 10,000 (R2 = 0.90; P less than .001) – according to an analysis in Pediatrics.
The analysis was conducted by Danielle Orsagh-Yentis, MD, of Vanderbilt University, Nashville, Tenn., and her colleagues. They estimated that, during the study period, 759,074 children younger than 6 years of age were evaluated in U.S. EDs for suspected or confirmed foreign-body ingestions. These estimates were based on data for 29,893 actual cases taken from the National Electronic Injury Surveillance System (NEISS), which represents about 100 hospitals. Each case in this system is given a sample weight by the Consumer Product Safety Commission using a validated method, and the estimates are based on this weighting.
The analysis showed that children aged 1 year (21%) and boys (53%) were the most likely to ingest foreign bodies. Coins were the most frequently ingested objects, at 62%. Among cases which had the location noted (59%), most ingestions occurred in the home (97%).
The authors noted that, although batteries and magnets represented only 7% and 2% of all cases, respectively, “they can both enact considerable damage when ingested.” For example, despite being only the fourth mostly likely object to be ingested, batteries were the second mostly likely to be implicated among hospitalized patients.
The authors noted that the NEISS captures patients in the ED only; the total number of foreign-body ingestions, then, was likely underestimated. Despite this, the authors felt the long study period and large sample were strengths of their analysis.
Dr. Orsagh-Yentis and her associates disclosed no potential conflicts of interest.
SOURCE: Orsagh-Yentis D et al. Pediatrics. 2019 Apr 12. doi: 10.1542/peds.2018-1988.
FROM PEDIATRICS
No clear benefit seen for postdischarge oxygen in preemies with BPD
Preterm infants with bronchopulmonary dysplasia (BPD) discharged with supplemental oxygen showed slightly better weight and significantly improved weight-for-length scores, but were more likely to use medical resources and had rates of neurodevelopmental impairment similar to those of infants not discharged with oxygen, according to research published in Pediatrics.
“With this study, we provide important and novel information that may aid the decision of whether to discharge an infant with supplemental oxygen, particularly for those infants who might be weaned off by some clinicians and not by others,” wrote Sara B. DeMauro, MD, MSCE, of University of Pennsylvania, Philadelphia, and Children’s Hospital of Philadelphia, and her colleagues. “This study helps to clarify, both for clinicians and parents, the potential benefits and harms that might be expected from home oxygen therapy among the subset of infants for whom the best course of action is unclear.”
Dr. DeMauro and her colleagues examined 1,039 preterm infants with BPD given supplemental oxygen by nasal cannula between January 2006 and December 2014, who were propensity matched to infants in a control group with a similar severity of BPD who were not discharged with oxygen. The infants were born at less than 27 weeks’ gestation and began receiving oxygen therapy or respiratory support at 36 weeks’ postmenstrual age. These infants were then measured for growth, neurodevelopment, and resource use from discharge to follow-up at 22-26 months corrected age.
At follow-up, infants discharged with oxygen showed marginal weight improvement scores (adjusted mean difference, 0.11) and significantly improved weight-for-length scores (adjusted mean difference, 0.13), but they had rates of neurodevelopmental impairment similar to those of infants with BPD discharged without supplemental oxygen. In addition, infants discharged with oxygen had a greater likelihood of rehospitalization due to respiratory illness (adjusted relative risk, 1.33), use of asthma or BPD medication (adjusted RR, 1.30), and use of medical equipment such as a pulse oximeter (adjusted RR, 2.94).
The researchers noted that their study’s design prevented them from examining all infants with BPD discharged with supplemental oxygen and what factors influenced discharge of infants with supplemental oxygen, as well as the effects of various durations of supplemental oxygen exposure.
“Definitive evaluation of the risk/benefit ratio of this therapy will require prospective controlled trials,” Dr. DeMauro and her colleagues wrote. “Such research will facilitate a more evidence-based approach to clinical decisions about postdischarge care of infants with BPD.”
This study received funding from the Eunice Kennedy Shriver National Institute of Child Health and Human Development Neonatal Research Network and the National Institutes of Health. The authors reported no relevant financial disclosures.
SOURCE: DeMauro SB et al. Pediatrics. 2019 Apr 11. doi: 10.1542/peds.2018-2956.
While oxygen use recommendations for preterm infants in the delivery room and neonatal ICU have changed, postdischarge oxygen instructions have largely not, with variations among practices and evidence for its use not well established.
The results from DeMauro et al., while not establishing causality, can instead be used to design a prospective trial to identify which preterm infants with BPD require oxygen post discharge, Reese H. Clark, MD, and Veeral N. Tolia, MD, wrote in a related editorial.
Supplemental oxygen also was associated with greater resource use among infants in the study, and they were more likely to require medications for asthma and BPD, procedures such as tracheotomy, and rehospitalization, which is in line with previous clinical studies analyzing oxygen use in the NICU, they noted.
The findings by DeMauro et al. could be used to improve the design and safety of a prospective study. For example, “it may not be feasible or ethical to include some infants with more severe BPD in future trials,” they noted. “Once again, we are challenged to reevaluate our clinical beliefs and biases about the use of oxygen,” said Dr. Clark and Dr. Tolia. “Now we must collaborate to design and implement a trial to help us determine which infants should receive oxygen after discharge. We look forward to seeing those results.”
Dr. Clark is from the Center for Research and Education at MEDNAX in Sunrise, Fla., and Dr. Tolia is at Baylor University Medical Center and Pediatrix Medical Group in Dallas. This is a summary of the editorial accompanying the report by DeMauro et al. (Pediatrics. 2019 Apr 11. doi: 10.1542/peds.2019-0372). They reported no relevant financial disclosures or external funding.
While oxygen use recommendations for preterm infants in the delivery room and neonatal ICU have changed, postdischarge oxygen instructions have largely not, with variations among practices and evidence for its use not well established.
The results from DeMauro et al., while not establishing causality, can instead be used to design a prospective trial to identify which preterm infants with BPD require oxygen post discharge, Reese H. Clark, MD, and Veeral N. Tolia, MD, wrote in a related editorial.
Supplemental oxygen also was associated with greater resource use among infants in the study, and they were more likely to require medications for asthma and BPD, procedures such as tracheotomy, and rehospitalization, which is in line with previous clinical studies analyzing oxygen use in the NICU, they noted.
The findings by DeMauro et al. could be used to improve the design and safety of a prospective study. For example, “it may not be feasible or ethical to include some infants with more severe BPD in future trials,” they noted. “Once again, we are challenged to reevaluate our clinical beliefs and biases about the use of oxygen,” said Dr. Clark and Dr. Tolia. “Now we must collaborate to design and implement a trial to help us determine which infants should receive oxygen after discharge. We look forward to seeing those results.”
Dr. Clark is from the Center for Research and Education at MEDNAX in Sunrise, Fla., and Dr. Tolia is at Baylor University Medical Center and Pediatrix Medical Group in Dallas. This is a summary of the editorial accompanying the report by DeMauro et al. (Pediatrics. 2019 Apr 11. doi: 10.1542/peds.2019-0372). They reported no relevant financial disclosures or external funding.
While oxygen use recommendations for preterm infants in the delivery room and neonatal ICU have changed, postdischarge oxygen instructions have largely not, with variations among practices and evidence for its use not well established.
The results from DeMauro et al., while not establishing causality, can instead be used to design a prospective trial to identify which preterm infants with BPD require oxygen post discharge, Reese H. Clark, MD, and Veeral N. Tolia, MD, wrote in a related editorial.
Supplemental oxygen also was associated with greater resource use among infants in the study, and they were more likely to require medications for asthma and BPD, procedures such as tracheotomy, and rehospitalization, which is in line with previous clinical studies analyzing oxygen use in the NICU, they noted.
The findings by DeMauro et al. could be used to improve the design and safety of a prospective study. For example, “it may not be feasible or ethical to include some infants with more severe BPD in future trials,” they noted. “Once again, we are challenged to reevaluate our clinical beliefs and biases about the use of oxygen,” said Dr. Clark and Dr. Tolia. “Now we must collaborate to design and implement a trial to help us determine which infants should receive oxygen after discharge. We look forward to seeing those results.”
Dr. Clark is from the Center for Research and Education at MEDNAX in Sunrise, Fla., and Dr. Tolia is at Baylor University Medical Center and Pediatrix Medical Group in Dallas. This is a summary of the editorial accompanying the report by DeMauro et al. (Pediatrics. 2019 Apr 11. doi: 10.1542/peds.2019-0372). They reported no relevant financial disclosures or external funding.
Preterm infants with bronchopulmonary dysplasia (BPD) discharged with supplemental oxygen showed slightly better weight and significantly improved weight-for-length scores, but were more likely to use medical resources and had rates of neurodevelopmental impairment similar to those of infants not discharged with oxygen, according to research published in Pediatrics.
“With this study, we provide important and novel information that may aid the decision of whether to discharge an infant with supplemental oxygen, particularly for those infants who might be weaned off by some clinicians and not by others,” wrote Sara B. DeMauro, MD, MSCE, of University of Pennsylvania, Philadelphia, and Children’s Hospital of Philadelphia, and her colleagues. “This study helps to clarify, both for clinicians and parents, the potential benefits and harms that might be expected from home oxygen therapy among the subset of infants for whom the best course of action is unclear.”
Dr. DeMauro and her colleagues examined 1,039 preterm infants with BPD given supplemental oxygen by nasal cannula between January 2006 and December 2014, who were propensity matched to infants in a control group with a similar severity of BPD who were not discharged with oxygen. The infants were born at less than 27 weeks’ gestation and began receiving oxygen therapy or respiratory support at 36 weeks’ postmenstrual age. These infants were then measured for growth, neurodevelopment, and resource use from discharge to follow-up at 22-26 months corrected age.
At follow-up, infants discharged with oxygen showed marginal weight improvement scores (adjusted mean difference, 0.11) and significantly improved weight-for-length scores (adjusted mean difference, 0.13), but they had rates of neurodevelopmental impairment similar to those of infants with BPD discharged without supplemental oxygen. In addition, infants discharged with oxygen had a greater likelihood of rehospitalization due to respiratory illness (adjusted relative risk, 1.33), use of asthma or BPD medication (adjusted RR, 1.30), and use of medical equipment such as a pulse oximeter (adjusted RR, 2.94).
The researchers noted that their study’s design prevented them from examining all infants with BPD discharged with supplemental oxygen and what factors influenced discharge of infants with supplemental oxygen, as well as the effects of various durations of supplemental oxygen exposure.
“Definitive evaluation of the risk/benefit ratio of this therapy will require prospective controlled trials,” Dr. DeMauro and her colleagues wrote. “Such research will facilitate a more evidence-based approach to clinical decisions about postdischarge care of infants with BPD.”
This study received funding from the Eunice Kennedy Shriver National Institute of Child Health and Human Development Neonatal Research Network and the National Institutes of Health. The authors reported no relevant financial disclosures.
SOURCE: DeMauro SB et al. Pediatrics. 2019 Apr 11. doi: 10.1542/peds.2018-2956.
Preterm infants with bronchopulmonary dysplasia (BPD) discharged with supplemental oxygen showed slightly better weight and significantly improved weight-for-length scores, but were more likely to use medical resources and had rates of neurodevelopmental impairment similar to those of infants not discharged with oxygen, according to research published in Pediatrics.
“With this study, we provide important and novel information that may aid the decision of whether to discharge an infant with supplemental oxygen, particularly for those infants who might be weaned off by some clinicians and not by others,” wrote Sara B. DeMauro, MD, MSCE, of University of Pennsylvania, Philadelphia, and Children’s Hospital of Philadelphia, and her colleagues. “This study helps to clarify, both for clinicians and parents, the potential benefits and harms that might be expected from home oxygen therapy among the subset of infants for whom the best course of action is unclear.”
Dr. DeMauro and her colleagues examined 1,039 preterm infants with BPD given supplemental oxygen by nasal cannula between January 2006 and December 2014, who were propensity matched to infants in a control group with a similar severity of BPD who were not discharged with oxygen. The infants were born at less than 27 weeks’ gestation and began receiving oxygen therapy or respiratory support at 36 weeks’ postmenstrual age. These infants were then measured for growth, neurodevelopment, and resource use from discharge to follow-up at 22-26 months corrected age.
At follow-up, infants discharged with oxygen showed marginal weight improvement scores (adjusted mean difference, 0.11) and significantly improved weight-for-length scores (adjusted mean difference, 0.13), but they had rates of neurodevelopmental impairment similar to those of infants with BPD discharged without supplemental oxygen. In addition, infants discharged with oxygen had a greater likelihood of rehospitalization due to respiratory illness (adjusted relative risk, 1.33), use of asthma or BPD medication (adjusted RR, 1.30), and use of medical equipment such as a pulse oximeter (adjusted RR, 2.94).
The researchers noted that their study’s design prevented them from examining all infants with BPD discharged with supplemental oxygen and what factors influenced discharge of infants with supplemental oxygen, as well as the effects of various durations of supplemental oxygen exposure.
“Definitive evaluation of the risk/benefit ratio of this therapy will require prospective controlled trials,” Dr. DeMauro and her colleagues wrote. “Such research will facilitate a more evidence-based approach to clinical decisions about postdischarge care of infants with BPD.”
This study received funding from the Eunice Kennedy Shriver National Institute of Child Health and Human Development Neonatal Research Network and the National Institutes of Health. The authors reported no relevant financial disclosures.
SOURCE: DeMauro SB et al. Pediatrics. 2019 Apr 11. doi: 10.1542/peds.2018-2956.
FROM PEDIATRICS
Key clinical point: compared with controls.
Major finding: At 22-26 months of age, infants discharged with oxygen showed marginal improvement in weight z scores (adjusted mean difference, 0.11) and significantly improved weight-for-length z scores (adjusted mean difference, 0.13), but similar rates of neurodevelopmental impairment.
Study details: A retrospective propensity-matched cohort study of 1,039 preterm infants given supplemental oxygen by nasal cannula between January 2006 and December 2014 and analyzed over 2 years of life.
Disclosures: This study received funding from the Eunice Kennedy Shriver National Institute of Child Health and Human Development Neonatal Research Network and the National Institutes of Health. The authors reported no relevant financial disclosures.
Source: DeMauro SB et al. Pediatrics. 2019 Apr 11. doi: 10.1542/peds.2018-2956.
Mucus buildup precedes lung damage in children with CF
according to a cross-sectional cohort study.
It has been difficult for researchers to pinpoint the mechanisms that initiate lung disease in people with CF, because it is challenging to study young people with the disease and “CF animal models often fail to recapitulate aspects of human CF disease and yield disparate findings,” wrote Charles R. Esther Jr., MD, of the division of pediatric pulmonology at the University of North Carolina at Chapel Hill and his colleagues in Science Translational Medicine.
The researchers studied 46 clinically stable young children (aged 3.3 years, plus or minus 1.7 years) with CF and 16 age-matched controls who did not have CF, but had respiratory symptoms (aged 3.2 years, plus or minus 2.0 years) using chest CT imaging and bronchoalveolar lavage fluid. BALF samples in CF patients were collected over 62 study visits and subsequently cultured for detection and quantification of pathogens. The children with CF were enrolled in the Australian Respiratory Early Surveillance Team for Cystic Fibrosis (AREST CF) program.
“We analyzed the relationships between airway mucus, inflammation, and bacterial culture/microbiome,” the researchers wrote.
BALF total mucin levels were higher in CF samples versus non-CF controls. In addition, Dr. Esther and his colleagues found that these results were the same regardless of infection status and that increased densities of mucus flakes were also seen in samples from the CF patients. “Elevated total mucin concentrations and inflammatory markers were observed in children with CF despite a low incidence of pathogens identified by culture or molecular microbiology. This muco-inflammatory state also characterized our CF population with the earliest lung disease [without substantial CT-defined structural changes] in the setting of little or no pathogen infection,” they wrote.
Based on the findings, the investigators postulated that the airways of children with CF may show distinct defects in the clearance of recently created mucins, which could contribute to early CF lung disease.
A key limitation of the study was the prophylactic use of intermittent antibiotics. As a result, bacterial infection could have contributed to the development of early CF lung disease.
“Agents designed to remove permanent mucus covering airway surfaces of young children with CF appear to be rational strategies to prevent bacterial infection and disease progression,” they concluded.
The study was supported by the National Heart, Lung, and Blood Institute; the North Carolina Translational and Clinical Sciences Institute; the National Health and Medical Research Council; and the Cystic Fibrosis Foundation. Two coauthors reported financial affiliations with Parion Sciences.
SOURCE: Esther CR et al. Sci Transl Med. 2019 Apr 3. doi: 10.1126/scitranslmed.aav3488.
according to a cross-sectional cohort study.
It has been difficult for researchers to pinpoint the mechanisms that initiate lung disease in people with CF, because it is challenging to study young people with the disease and “CF animal models often fail to recapitulate aspects of human CF disease and yield disparate findings,” wrote Charles R. Esther Jr., MD, of the division of pediatric pulmonology at the University of North Carolina at Chapel Hill and his colleagues in Science Translational Medicine.
The researchers studied 46 clinically stable young children (aged 3.3 years, plus or minus 1.7 years) with CF and 16 age-matched controls who did not have CF, but had respiratory symptoms (aged 3.2 years, plus or minus 2.0 years) using chest CT imaging and bronchoalveolar lavage fluid. BALF samples in CF patients were collected over 62 study visits and subsequently cultured for detection and quantification of pathogens. The children with CF were enrolled in the Australian Respiratory Early Surveillance Team for Cystic Fibrosis (AREST CF) program.
“We analyzed the relationships between airway mucus, inflammation, and bacterial culture/microbiome,” the researchers wrote.
BALF total mucin levels were higher in CF samples versus non-CF controls. In addition, Dr. Esther and his colleagues found that these results were the same regardless of infection status and that increased densities of mucus flakes were also seen in samples from the CF patients. “Elevated total mucin concentrations and inflammatory markers were observed in children with CF despite a low incidence of pathogens identified by culture or molecular microbiology. This muco-inflammatory state also characterized our CF population with the earliest lung disease [without substantial CT-defined structural changes] in the setting of little or no pathogen infection,” they wrote.
Based on the findings, the investigators postulated that the airways of children with CF may show distinct defects in the clearance of recently created mucins, which could contribute to early CF lung disease.
A key limitation of the study was the prophylactic use of intermittent antibiotics. As a result, bacterial infection could have contributed to the development of early CF lung disease.
“Agents designed to remove permanent mucus covering airway surfaces of young children with CF appear to be rational strategies to prevent bacterial infection and disease progression,” they concluded.
The study was supported by the National Heart, Lung, and Blood Institute; the North Carolina Translational and Clinical Sciences Institute; the National Health and Medical Research Council; and the Cystic Fibrosis Foundation. Two coauthors reported financial affiliations with Parion Sciences.
SOURCE: Esther CR et al. Sci Transl Med. 2019 Apr 3. doi: 10.1126/scitranslmed.aav3488.
according to a cross-sectional cohort study.
It has been difficult for researchers to pinpoint the mechanisms that initiate lung disease in people with CF, because it is challenging to study young people with the disease and “CF animal models often fail to recapitulate aspects of human CF disease and yield disparate findings,” wrote Charles R. Esther Jr., MD, of the division of pediatric pulmonology at the University of North Carolina at Chapel Hill and his colleagues in Science Translational Medicine.
The researchers studied 46 clinically stable young children (aged 3.3 years, plus or minus 1.7 years) with CF and 16 age-matched controls who did not have CF, but had respiratory symptoms (aged 3.2 years, plus or minus 2.0 years) using chest CT imaging and bronchoalveolar lavage fluid. BALF samples in CF patients were collected over 62 study visits and subsequently cultured for detection and quantification of pathogens. The children with CF were enrolled in the Australian Respiratory Early Surveillance Team for Cystic Fibrosis (AREST CF) program.
“We analyzed the relationships between airway mucus, inflammation, and bacterial culture/microbiome,” the researchers wrote.
BALF total mucin levels were higher in CF samples versus non-CF controls. In addition, Dr. Esther and his colleagues found that these results were the same regardless of infection status and that increased densities of mucus flakes were also seen in samples from the CF patients. “Elevated total mucin concentrations and inflammatory markers were observed in children with CF despite a low incidence of pathogens identified by culture or molecular microbiology. This muco-inflammatory state also characterized our CF population with the earliest lung disease [without substantial CT-defined structural changes] in the setting of little or no pathogen infection,” they wrote.
Based on the findings, the investigators postulated that the airways of children with CF may show distinct defects in the clearance of recently created mucins, which could contribute to early CF lung disease.
A key limitation of the study was the prophylactic use of intermittent antibiotics. As a result, bacterial infection could have contributed to the development of early CF lung disease.
“Agents designed to remove permanent mucus covering airway surfaces of young children with CF appear to be rational strategies to prevent bacterial infection and disease progression,” they concluded.
The study was supported by the National Heart, Lung, and Blood Institute; the North Carolina Translational and Clinical Sciences Institute; the National Health and Medical Research Council; and the Cystic Fibrosis Foundation. Two coauthors reported financial affiliations with Parion Sciences.
SOURCE: Esther CR et al. Sci Transl Med. 2019 Apr 3. doi: 10.1126/scitranslmed.aav3488.
FROM SCIENCE TRANSLATIONAL MEDICINE