User login
HM19: Pediatric medical and surgical co-management
Anticipatory and prevention-heavy approach
Presenter
Erin Shaughnessy, MD, MSHCM
Session title
Reaching Across the Aisle: Pediatric Co-Management with Surgery and Subspecialists
Session summary
Dr. Shaughnessy articulated a balanced approach to the importance of careful selection of patients needing to be co-managed by pediatric hospitalists. She compared two personal and very different experiences.
She initially managed a well-developed surgical co-management service at a quaternary, academic, free-standing children’s hospital, in which surgeons and subspecialists also admitted and managed patients to their own services. Currently, Dr. Shaughnessy is a division chief at Phoenix Children’s Hospital, a free-standing children’s hospital with a community hospital background, in which hospitalists admit most, if not all the patients, while subspecialty services have been transitioning only recently to having their own admitting services and employing the ideas of limited co-management.
She reminded the HM19 audience of the essential principles of co-management: shared responsibility, authority and accountability for the care of a hospitalized patient, discussing the scenarios, both from literature and real life, in which the line could become blurry at times.
Many pediatric programs are moving away from a traditional consultation model, Dr. Shaughnessy said, in which a consult is called for a new or a persistent problem with a patient, and where a consulting team signs off upon the resolved issue.
The more modern co-management model infuses a need for anticipatory and prevention-heavy approach, intertwined with fiscally responsible ideas that must be palatable for all: administration, hospitalists, and patients.
Dr. Shaughnessy reviewed a number of articles from both adult and pediatric literature with varied results, some that have shown decreased length of stay, decreased number of medical complications, decreased readmissions, decreased number of tests, but some that have also shown an increase in median hospital costs, emphasizing perhaps the importance of context in which one practices.
Finally, she identified patient selection, collaborative relationships, clear roles delineation, and excellence in communication as four main factors deciding the faith of a co-management model.
Key takeaways for HM
1. Careful selection of patients to be co-managed is essential and can prevent potential increase in costs and negative outcomes.
2. Success in medical and surgical co-management relies on well-delineated roles, collaborative culture, and immaculate communication.
Dr. Giordano is a pediatric neurosurgery hospitalist and assistant professor in pediatrics at Columbia University Irving Medical Center in New York.
Anticipatory and prevention-heavy approach
Anticipatory and prevention-heavy approach
Presenter
Erin Shaughnessy, MD, MSHCM
Session title
Reaching Across the Aisle: Pediatric Co-Management with Surgery and Subspecialists
Session summary
Dr. Shaughnessy articulated a balanced approach to the importance of careful selection of patients needing to be co-managed by pediatric hospitalists. She compared two personal and very different experiences.
She initially managed a well-developed surgical co-management service at a quaternary, academic, free-standing children’s hospital, in which surgeons and subspecialists also admitted and managed patients to their own services. Currently, Dr. Shaughnessy is a division chief at Phoenix Children’s Hospital, a free-standing children’s hospital with a community hospital background, in which hospitalists admit most, if not all the patients, while subspecialty services have been transitioning only recently to having their own admitting services and employing the ideas of limited co-management.
She reminded the HM19 audience of the essential principles of co-management: shared responsibility, authority and accountability for the care of a hospitalized patient, discussing the scenarios, both from literature and real life, in which the line could become blurry at times.
Many pediatric programs are moving away from a traditional consultation model, Dr. Shaughnessy said, in which a consult is called for a new or a persistent problem with a patient, and where a consulting team signs off upon the resolved issue.
The more modern co-management model infuses a need for anticipatory and prevention-heavy approach, intertwined with fiscally responsible ideas that must be palatable for all: administration, hospitalists, and patients.
Dr. Shaughnessy reviewed a number of articles from both adult and pediatric literature with varied results, some that have shown decreased length of stay, decreased number of medical complications, decreased readmissions, decreased number of tests, but some that have also shown an increase in median hospital costs, emphasizing perhaps the importance of context in which one practices.
Finally, she identified patient selection, collaborative relationships, clear roles delineation, and excellence in communication as four main factors deciding the faith of a co-management model.
Key takeaways for HM
1. Careful selection of patients to be co-managed is essential and can prevent potential increase in costs and negative outcomes.
2. Success in medical and surgical co-management relies on well-delineated roles, collaborative culture, and immaculate communication.
Dr. Giordano is a pediatric neurosurgery hospitalist and assistant professor in pediatrics at Columbia University Irving Medical Center in New York.
Presenter
Erin Shaughnessy, MD, MSHCM
Session title
Reaching Across the Aisle: Pediatric Co-Management with Surgery and Subspecialists
Session summary
Dr. Shaughnessy articulated a balanced approach to the importance of careful selection of patients needing to be co-managed by pediatric hospitalists. She compared two personal and very different experiences.
She initially managed a well-developed surgical co-management service at a quaternary, academic, free-standing children’s hospital, in which surgeons and subspecialists also admitted and managed patients to their own services. Currently, Dr. Shaughnessy is a division chief at Phoenix Children’s Hospital, a free-standing children’s hospital with a community hospital background, in which hospitalists admit most, if not all the patients, while subspecialty services have been transitioning only recently to having their own admitting services and employing the ideas of limited co-management.
She reminded the HM19 audience of the essential principles of co-management: shared responsibility, authority and accountability for the care of a hospitalized patient, discussing the scenarios, both from literature and real life, in which the line could become blurry at times.
Many pediatric programs are moving away from a traditional consultation model, Dr. Shaughnessy said, in which a consult is called for a new or a persistent problem with a patient, and where a consulting team signs off upon the resolved issue.
The more modern co-management model infuses a need for anticipatory and prevention-heavy approach, intertwined with fiscally responsible ideas that must be palatable for all: administration, hospitalists, and patients.
Dr. Shaughnessy reviewed a number of articles from both adult and pediatric literature with varied results, some that have shown decreased length of stay, decreased number of medical complications, decreased readmissions, decreased number of tests, but some that have also shown an increase in median hospital costs, emphasizing perhaps the importance of context in which one practices.
Finally, she identified patient selection, collaborative relationships, clear roles delineation, and excellence in communication as four main factors deciding the faith of a co-management model.
Key takeaways for HM
1. Careful selection of patients to be co-managed is essential and can prevent potential increase in costs and negative outcomes.
2. Success in medical and surgical co-management relies on well-delineated roles, collaborative culture, and immaculate communication.
Dr. Giordano is a pediatric neurosurgery hospitalist and assistant professor in pediatrics at Columbia University Irving Medical Center in New York.
Is childhood cancer associated with assisted reproductive technology?
Recently, two studies were published addressing the potential association of childhood cancer and assisted reproductive technology. For more than a decade and a half, it has been acknowledged that ART is associated with increased concern both with structural birth defects, as well as imprinting disorders. As both of these issues have been linked to greater cancer risk in children, it is important to decipher the impact of ART on childhood cancer risk.
Published online April 1 in JAMA Pediatrics, the study, “Association of in vitro fertilization [IVF] with childhood cancer in the United States,”1 by LG Spector et al. looked retrospectively at birth and cancer registries in 14 states with 8 years of data on 275,686 children were conceived via ART through 2013, who were compared with 2,266,847 children selected randomly.
The overall cancer rate per 1,000,000 person-years was low in both groups: 252 for the IVF group and 193 for the control group, for an overall hazard risk of 1.17. Of note, the rate of hepatic tumors was higher among the IVF group than the non-IVF group (18 vs. 5.7; hazard ratio, 2.46). There appeared to be no association with specific IVF treatments, whether children were conceived by donor egg vs. autologous egg; frozen embryos vs. fresh embryos; use of intracytoplasmic sperm injection (ICSI) vs. none; assisted hatching vs. none; and day-3 vs. day-5 transfer. The researchers concluded that the “increased rate of embryonal cancers, particularly hepatic tumors, that could not be attributed to IVF rather than to underlying infertility.”
This first and largest cohort study of association between IVF and the risk of childhood cancer ever published showed little evidence of excess risk of most cancers, including more common cancers such as leukemia.
The authors did note limitations in their study. Mothers who conceived via IVF were more likely to be white, non-Hispanic, more educated, and older. Could this patient population undergoing ART be at greater risk of producing offspring with cancer concerns? If that were the case – and not great risk of childhood cancer in ART, per se – one therefore would extrapolate that couples undergoing ART vs. alternative infertility treatment should not show a treatment-biased risk (i.e., ART vs. non-ART).
This was demonstrated recently in the study, “Risk of cancer in children and young adults conceived by assisted reproductive technology.”2 This Dutch historical cohort study with prospective follow-up of a median 21 years evaluated 47,690 live-born children, of which 24,269 were ART conceived, 13,761 naturally conceived, and 9,660 conceived naturally or with fertility drugs but not by ART.
Overall, cancer risk was not increased in ART-conceived children, compared with naturally conceived subfertile women or even the general population. A nonsignificant increased risk was observed in children conceived by ICSI or cryopreservation.
On the basis of these two studies, there appears to be no significant increased risk of cancer in children conceived through fertility treatment, including ART.
Although these studies do not support the conclusion reached by a 2013 meta-analysis of 9 studies that specifically looked at ART and 16 other studies that looked at other types of medically assisted reproduction (such medically assisted reproduction as reproduction achieved through ovulation induction; controlled ovarian stimulation; ovulation triggering; intrauterine, intracervical, or intravaginal insemination) which reported a significant increased risk of overall cancers (1.33), including leukemia, CNS cancer, and neuroblastoma,3 they do agree more closely with two prospective studies conducted in the United Kingdom and Nordic countries.
In the U.K. study,4 there was no overall increased risk of cancer associated with ART, but two types of cancer were noted to be higher in the ART-conceived group – hepatoblastoma (3.27 risk) and rhabdomyosarcoma (2.62 risk) – but the absolute risk of these two types of cancer was small in this 17-year study of 106,013 children. This, of course, would be consistent with the JAMA Pediatrics study. In the Nordic study,5 similar to the Dutch Study, IVF was not associated with a significant increased risk of cancer (1.08). The Nordic study included 91,796 children born of ART-assisted pregnancies, compared with 358,419 children born after spontaneous conceptions.
The evidence so far shows that there appears to be no significant increased risk of cancer overall associated with fertility treatments, including IVF.
Dr. Miller is a clinical associate professor at the University of Illinois in Chicago and past president of the AAGL. He is a reproductive endocrinologist and minimally invasive gynecologic surgeon in metropolitan Chicago and the director of minimally invasive gynecologic surgery at Advocate Lutheran General Hospital, Park Ridge, Ill. He also is a member of Ob.Gyn. News editorial advisory board. Dr. Miller disclosed that he is president of the Advanced IVF Institute in Park Ridge and Naperville, Ill.
References
1. JAMA Pediatr. 2019 Apr 1. doi: 10.1001/jamapediatrics.2019.0392.
2. Hum Reprod. 2019 Apr 1;34(4):740-50.
3. Fertil Steril. 2013 Jul. doi: 10.1016/j.fertnstert.2013.03.017.
4. N Engl J Med. 2013 Nov 7;369(19):1819-27.
5. Hum Reprod. 2014 Sep;29(9):2050-7.
Recently, two studies were published addressing the potential association of childhood cancer and assisted reproductive technology. For more than a decade and a half, it has been acknowledged that ART is associated with increased concern both with structural birth defects, as well as imprinting disorders. As both of these issues have been linked to greater cancer risk in children, it is important to decipher the impact of ART on childhood cancer risk.
Published online April 1 in JAMA Pediatrics, the study, “Association of in vitro fertilization [IVF] with childhood cancer in the United States,”1 by LG Spector et al. looked retrospectively at birth and cancer registries in 14 states with 8 years of data on 275,686 children were conceived via ART through 2013, who were compared with 2,266,847 children selected randomly.
The overall cancer rate per 1,000,000 person-years was low in both groups: 252 for the IVF group and 193 for the control group, for an overall hazard risk of 1.17. Of note, the rate of hepatic tumors was higher among the IVF group than the non-IVF group (18 vs. 5.7; hazard ratio, 2.46). There appeared to be no association with specific IVF treatments, whether children were conceived by donor egg vs. autologous egg; frozen embryos vs. fresh embryos; use of intracytoplasmic sperm injection (ICSI) vs. none; assisted hatching vs. none; and day-3 vs. day-5 transfer. The researchers concluded that the “increased rate of embryonal cancers, particularly hepatic tumors, that could not be attributed to IVF rather than to underlying infertility.”
This first and largest cohort study of association between IVF and the risk of childhood cancer ever published showed little evidence of excess risk of most cancers, including more common cancers such as leukemia.
The authors did note limitations in their study. Mothers who conceived via IVF were more likely to be white, non-Hispanic, more educated, and older. Could this patient population undergoing ART be at greater risk of producing offspring with cancer concerns? If that were the case – and not great risk of childhood cancer in ART, per se – one therefore would extrapolate that couples undergoing ART vs. alternative infertility treatment should not show a treatment-biased risk (i.e., ART vs. non-ART).
This was demonstrated recently in the study, “Risk of cancer in children and young adults conceived by assisted reproductive technology.”2 This Dutch historical cohort study with prospective follow-up of a median 21 years evaluated 47,690 live-born children, of which 24,269 were ART conceived, 13,761 naturally conceived, and 9,660 conceived naturally or with fertility drugs but not by ART.
Overall, cancer risk was not increased in ART-conceived children, compared with naturally conceived subfertile women or even the general population. A nonsignificant increased risk was observed in children conceived by ICSI or cryopreservation.
On the basis of these two studies, there appears to be no significant increased risk of cancer in children conceived through fertility treatment, including ART.
Although these studies do not support the conclusion reached by a 2013 meta-analysis of 9 studies that specifically looked at ART and 16 other studies that looked at other types of medically assisted reproduction (such medically assisted reproduction as reproduction achieved through ovulation induction; controlled ovarian stimulation; ovulation triggering; intrauterine, intracervical, or intravaginal insemination) which reported a significant increased risk of overall cancers (1.33), including leukemia, CNS cancer, and neuroblastoma,3 they do agree more closely with two prospective studies conducted in the United Kingdom and Nordic countries.
In the U.K. study,4 there was no overall increased risk of cancer associated with ART, but two types of cancer were noted to be higher in the ART-conceived group – hepatoblastoma (3.27 risk) and rhabdomyosarcoma (2.62 risk) – but the absolute risk of these two types of cancer was small in this 17-year study of 106,013 children. This, of course, would be consistent with the JAMA Pediatrics study. In the Nordic study,5 similar to the Dutch Study, IVF was not associated with a significant increased risk of cancer (1.08). The Nordic study included 91,796 children born of ART-assisted pregnancies, compared with 358,419 children born after spontaneous conceptions.
The evidence so far shows that there appears to be no significant increased risk of cancer overall associated with fertility treatments, including IVF.
Dr. Miller is a clinical associate professor at the University of Illinois in Chicago and past president of the AAGL. He is a reproductive endocrinologist and minimally invasive gynecologic surgeon in metropolitan Chicago and the director of minimally invasive gynecologic surgery at Advocate Lutheran General Hospital, Park Ridge, Ill. He also is a member of Ob.Gyn. News editorial advisory board. Dr. Miller disclosed that he is president of the Advanced IVF Institute in Park Ridge and Naperville, Ill.
References
1. JAMA Pediatr. 2019 Apr 1. doi: 10.1001/jamapediatrics.2019.0392.
2. Hum Reprod. 2019 Apr 1;34(4):740-50.
3. Fertil Steril. 2013 Jul. doi: 10.1016/j.fertnstert.2013.03.017.
4. N Engl J Med. 2013 Nov 7;369(19):1819-27.
5. Hum Reprod. 2014 Sep;29(9):2050-7.
Recently, two studies were published addressing the potential association of childhood cancer and assisted reproductive technology. For more than a decade and a half, it has been acknowledged that ART is associated with increased concern both with structural birth defects, as well as imprinting disorders. As both of these issues have been linked to greater cancer risk in children, it is important to decipher the impact of ART on childhood cancer risk.
Published online April 1 in JAMA Pediatrics, the study, “Association of in vitro fertilization [IVF] with childhood cancer in the United States,”1 by LG Spector et al. looked retrospectively at birth and cancer registries in 14 states with 8 years of data on 275,686 children were conceived via ART through 2013, who were compared with 2,266,847 children selected randomly.
The overall cancer rate per 1,000,000 person-years was low in both groups: 252 for the IVF group and 193 for the control group, for an overall hazard risk of 1.17. Of note, the rate of hepatic tumors was higher among the IVF group than the non-IVF group (18 vs. 5.7; hazard ratio, 2.46). There appeared to be no association with specific IVF treatments, whether children were conceived by donor egg vs. autologous egg; frozen embryos vs. fresh embryos; use of intracytoplasmic sperm injection (ICSI) vs. none; assisted hatching vs. none; and day-3 vs. day-5 transfer. The researchers concluded that the “increased rate of embryonal cancers, particularly hepatic tumors, that could not be attributed to IVF rather than to underlying infertility.”
This first and largest cohort study of association between IVF and the risk of childhood cancer ever published showed little evidence of excess risk of most cancers, including more common cancers such as leukemia.
The authors did note limitations in their study. Mothers who conceived via IVF were more likely to be white, non-Hispanic, more educated, and older. Could this patient population undergoing ART be at greater risk of producing offspring with cancer concerns? If that were the case – and not great risk of childhood cancer in ART, per se – one therefore would extrapolate that couples undergoing ART vs. alternative infertility treatment should not show a treatment-biased risk (i.e., ART vs. non-ART).
This was demonstrated recently in the study, “Risk of cancer in children and young adults conceived by assisted reproductive technology.”2 This Dutch historical cohort study with prospective follow-up of a median 21 years evaluated 47,690 live-born children, of which 24,269 were ART conceived, 13,761 naturally conceived, and 9,660 conceived naturally or with fertility drugs but not by ART.
Overall, cancer risk was not increased in ART-conceived children, compared with naturally conceived subfertile women or even the general population. A nonsignificant increased risk was observed in children conceived by ICSI or cryopreservation.
On the basis of these two studies, there appears to be no significant increased risk of cancer in children conceived through fertility treatment, including ART.
Although these studies do not support the conclusion reached by a 2013 meta-analysis of 9 studies that specifically looked at ART and 16 other studies that looked at other types of medically assisted reproduction (such medically assisted reproduction as reproduction achieved through ovulation induction; controlled ovarian stimulation; ovulation triggering; intrauterine, intracervical, or intravaginal insemination) which reported a significant increased risk of overall cancers (1.33), including leukemia, CNS cancer, and neuroblastoma,3 they do agree more closely with two prospective studies conducted in the United Kingdom and Nordic countries.
In the U.K. study,4 there was no overall increased risk of cancer associated with ART, but two types of cancer were noted to be higher in the ART-conceived group – hepatoblastoma (3.27 risk) and rhabdomyosarcoma (2.62 risk) – but the absolute risk of these two types of cancer was small in this 17-year study of 106,013 children. This, of course, would be consistent with the JAMA Pediatrics study. In the Nordic study,5 similar to the Dutch Study, IVF was not associated with a significant increased risk of cancer (1.08). The Nordic study included 91,796 children born of ART-assisted pregnancies, compared with 358,419 children born after spontaneous conceptions.
The evidence so far shows that there appears to be no significant increased risk of cancer overall associated with fertility treatments, including IVF.
Dr. Miller is a clinical associate professor at the University of Illinois in Chicago and past president of the AAGL. He is a reproductive endocrinologist and minimally invasive gynecologic surgeon in metropolitan Chicago and the director of minimally invasive gynecologic surgery at Advocate Lutheran General Hospital, Park Ridge, Ill. He also is a member of Ob.Gyn. News editorial advisory board. Dr. Miller disclosed that he is president of the Advanced IVF Institute in Park Ridge and Naperville, Ill.
References
1. JAMA Pediatr. 2019 Apr 1. doi: 10.1001/jamapediatrics.2019.0392.
2. Hum Reprod. 2019 Apr 1;34(4):740-50.
3. Fertil Steril. 2013 Jul. doi: 10.1016/j.fertnstert.2013.03.017.
4. N Engl J Med. 2013 Nov 7;369(19):1819-27.
5. Hum Reprod. 2014 Sep;29(9):2050-7.
Papules and Telangiectases on the Distal Fingers of a Child
The Diagnosis: Juvenile Dermatomyositis
Juvenile dermatomyositis (JDM) is a rare idiopathic inflammatory myopathy of childhood that is autoimmune in nature with an annual incidence ranging from 2.5 to 4.1 cases per million children. Its peak incidence is between 5 and 10 years of age, and it affects girls more than boys at a 2-fold to 5-fold greater rate.1 Juvenile dermatomyositis is characterized by skeletal muscle weakness in the presence of distinctive rashes, including Gottron papules and heliotrope erythema. Muscle weakness typically is proximal and symmetrical, and eventually patients may have trouble rising from a seated position or lifting objects overhead. Other skin manifestations include nail fold capillary changes, calcinosis cutis, and less commonly ulcerations signifying vasculopathy of the skin.2 A subset of patients will present with juvenile amyopathic dermatomyositis. These children have the characteristic skin changes without the muscle weakness or elevated muscle enzymes for more than 6 months; however, one-quarter may go on to develop mysositis.3
Diagnosis of JDM traditionally was based on the following 5 diagnostic criteria: characteristic skin rash, proximal muscle weakness, elevated muscle enzymes, myopathic changes on electromyogram, and typical muscle biopsy.1 Current practice shows a broadening of diagnostic criteria using new techniques in the diagnosis of JDM. To make the diagnosis, the patient must have the characteristic skin manifestations with a minimum of 3 other criteria.4 A 2006 international consensus survey expanded the list of criteria to include typical findings on magnetic resonance imaging (MRI), nail fold capillaroscopy abnormalities, calcinosis, and
dysphonia.5
To assess muscle disease, MRI is utilized because it is a reliable noninvasive tool to assess muscle inflammation. Muscle biopsy is only recommended if the diagnosis is unclear.5 The results of the MRI in our patient displayed symmetric mild fatty atrophy of the gluteus maximus muscle, as well as edema in the right rectus femoris and left vastus lateralis muscles, suggesting early findings of myositis. Muscle enzymes may not be diagnostic because they are not always elevated at diagnosis. Our patient had a normal creatinine kinase level (92 U/L [reference range, <190 U/L]), and both aldolase and lactate dehydrogenase also were within reference range. Conversely, antinuclear antibodies frequently are positive in patients with JDM, such as in our patient at a 1:320 dilution, but are nonspecific and nondiagnostic. It is recommended to include nail fold capillaroscopy to evaluate periungual capillary changes because nailfold capillary density is a sensitive measure of both skin and muscle disease.5 Using dermoscopy, nail fold capillary dilation was observed in our patient.
Other differential diagnoses can have somewhat similar clinical features to JDM. Infantile papular acrodermatitis, commonly referred to as Gianotti-Crosti syndrome, is a viral exanthem that affects children (median age, 2 years).6 The rash appears as monomorphous, flat-topped, pink to brown papules affecting the face, buttocks, and arms; it typically spontaneously resolves in 10 days.6
Juvenile-onset lupus is a chronic autoimmune disorder that can involve any organ system and typically affects children aged 11 to 12 years with a female preponderance. Skin manifestations are similar to adult-onset lupus and include malar rash, discoid rash, oral ulcerations, petechiae, palpable purpura, and digital telangiectasia and ulcers. 7
Juvenile scleroderma is rare connective-tissue disorder that also has multiple organ involvement. Cutaneous involvement can range from isolated morphealike plaques to diffuse sclerotic lesions with growth disturbances, contractures, and facial atrophy.8
Verrucae planae, commonly referred to as flat warts, are papules caused primarily by human papillomavirus types 3, 10, 28, and 41. Children and young adults commonly are affected, and warts can appear on the hands, as in our patient.6
Treatment of JDM depends on disease severity at initial presentation and requires a multidisciplinary approach. The mainstay of treatment is high-dose oral prednisone in combination with disease-modifying drugs such as methotrexate and cyclosporin A. Patients with more severe presentations (eg, ulcerative skin disease) or life-threatening organ involvement are treated with cyclophosphamide, usually in combination with high-dose glucocorticoids.9
Early detection with aggressive treatment is vital to reduce morbidity and mortality from organ damage and disease complications. Mortality rates have dropped to 3%10 in recent decades with the use of systemic glucocorticoids. Delayed treatment is associated with a prolonged disease course and poorer outcomes. Disease complications in children with JDM include osteoporosis, calcinosis, and intestinal perforation; however, with early treatment, children with JDM can expect full recovery and to live a normal life as compared to adults with dermatomyositis.10
Prior to our patient's diagnosis, the family was assigned to move to an overseas location through the US Military with no direct access to advanced medical care. Early detection and diagnosis of JDM through an astute clinical examination allowed the patient and her family to remain in the continental United States to continue receiving specialty care.
- Mendez EP, Lipton R, Ramsey-Goldman R, et al. US incidence of juvenile dermatomyositis,1995-1998: results from the National Institute of Arthritis and Musculoskeletal and Skin Diseases Registry. Arthritis Rheum. 2003;49:300-305.
- Shah M, Mamyrova G, Targoff IN, et al. The clinical phenotypes of the juvenile idiopathic inflammatory myopathies. Medicine. 2013;92:25-41.
- Gerami P, Walling HW, Lewis J, et al. A systematic review of juvenile-onset clinically amyopathic dermatomyositis. Br J Dermatol. 2007;57:637-644.
- Enders FB, Bader-Meunier B, Baildam E, et al. Consensus-based recommendations for the management of juvenile dermatomyositis. Ann Rheum Dis. 2017;76:329-340.
- Brown VE, Pilkington CA, Feldman BM, et al. An international consensus survey of the diagnostic criteria for juvenile dermatomyositis (JDM). Rheumatology (Oxford). 2006;45:990-993.
- William JD, Berger TG, Elston DM. Viral diseases. In: William JD, Berger TG, Elston DM. Andrews' Diseases of the Skin: Clinical Dermatology. 11th ed. China: Saunders Elsevier; 2011:360-413.
- Levy DM, Kamphuis S. Systemic lupus erythematosus in children and adolescents. Pediatr Clin North Am. 2012;59:345-364.
- Li SC, Torok KS, Pope E, et al; Childhood Arthritis and Rheumatology Research Alliance (CARRA) Localized Scleroderma Workgroup. Development of consensus treatment plans for juvenile localized scleroderma: a roadmap toward comparative effectiveness studies in juvenile localized scleroderma. Arthritis Care Res (Hoboken). 2012;64:1175-1185.
- Stringer E, Ota S, Bohnsack J, et al. Treatment approaches to juvenile dermatomyositis (JDM) across North America: the Childhood Arthritis and Rheumatology Research Alliance (CARRA) JDM treatment study. J Rhematol. 2010;37:S1953-S1961.
- Huber AM, Feldman BM. Long-term outcomes in juvenile dermatomyositis: how did we get here and where are we going? Curr Rheumatol Rep. 2005;7:441-446.
The Diagnosis: Juvenile Dermatomyositis
Juvenile dermatomyositis (JDM) is a rare idiopathic inflammatory myopathy of childhood that is autoimmune in nature with an annual incidence ranging from 2.5 to 4.1 cases per million children. Its peak incidence is between 5 and 10 years of age, and it affects girls more than boys at a 2-fold to 5-fold greater rate.1 Juvenile dermatomyositis is characterized by skeletal muscle weakness in the presence of distinctive rashes, including Gottron papules and heliotrope erythema. Muscle weakness typically is proximal and symmetrical, and eventually patients may have trouble rising from a seated position or lifting objects overhead. Other skin manifestations include nail fold capillary changes, calcinosis cutis, and less commonly ulcerations signifying vasculopathy of the skin.2 A subset of patients will present with juvenile amyopathic dermatomyositis. These children have the characteristic skin changes without the muscle weakness or elevated muscle enzymes for more than 6 months; however, one-quarter may go on to develop mysositis.3
Diagnosis of JDM traditionally was based on the following 5 diagnostic criteria: characteristic skin rash, proximal muscle weakness, elevated muscle enzymes, myopathic changes on electromyogram, and typical muscle biopsy.1 Current practice shows a broadening of diagnostic criteria using new techniques in the diagnosis of JDM. To make the diagnosis, the patient must have the characteristic skin manifestations with a minimum of 3 other criteria.4 A 2006 international consensus survey expanded the list of criteria to include typical findings on magnetic resonance imaging (MRI), nail fold capillaroscopy abnormalities, calcinosis, and
dysphonia.5
To assess muscle disease, MRI is utilized because it is a reliable noninvasive tool to assess muscle inflammation. Muscle biopsy is only recommended if the diagnosis is unclear.5 The results of the MRI in our patient displayed symmetric mild fatty atrophy of the gluteus maximus muscle, as well as edema in the right rectus femoris and left vastus lateralis muscles, suggesting early findings of myositis. Muscle enzymes may not be diagnostic because they are not always elevated at diagnosis. Our patient had a normal creatinine kinase level (92 U/L [reference range, <190 U/L]), and both aldolase and lactate dehydrogenase also were within reference range. Conversely, antinuclear antibodies frequently are positive in patients with JDM, such as in our patient at a 1:320 dilution, but are nonspecific and nondiagnostic. It is recommended to include nail fold capillaroscopy to evaluate periungual capillary changes because nailfold capillary density is a sensitive measure of both skin and muscle disease.5 Using dermoscopy, nail fold capillary dilation was observed in our patient.
Other differential diagnoses can have somewhat similar clinical features to JDM. Infantile papular acrodermatitis, commonly referred to as Gianotti-Crosti syndrome, is a viral exanthem that affects children (median age, 2 years).6 The rash appears as monomorphous, flat-topped, pink to brown papules affecting the face, buttocks, and arms; it typically spontaneously resolves in 10 days.6
Juvenile-onset lupus is a chronic autoimmune disorder that can involve any organ system and typically affects children aged 11 to 12 years with a female preponderance. Skin manifestations are similar to adult-onset lupus and include malar rash, discoid rash, oral ulcerations, petechiae, palpable purpura, and digital telangiectasia and ulcers. 7
Juvenile scleroderma is rare connective-tissue disorder that also has multiple organ involvement. Cutaneous involvement can range from isolated morphealike plaques to diffuse sclerotic lesions with growth disturbances, contractures, and facial atrophy.8
Verrucae planae, commonly referred to as flat warts, are papules caused primarily by human papillomavirus types 3, 10, 28, and 41. Children and young adults commonly are affected, and warts can appear on the hands, as in our patient.6
Treatment of JDM depends on disease severity at initial presentation and requires a multidisciplinary approach. The mainstay of treatment is high-dose oral prednisone in combination with disease-modifying drugs such as methotrexate and cyclosporin A. Patients with more severe presentations (eg, ulcerative skin disease) or life-threatening organ involvement are treated with cyclophosphamide, usually in combination with high-dose glucocorticoids.9
Early detection with aggressive treatment is vital to reduce morbidity and mortality from organ damage and disease complications. Mortality rates have dropped to 3%10 in recent decades with the use of systemic glucocorticoids. Delayed treatment is associated with a prolonged disease course and poorer outcomes. Disease complications in children with JDM include osteoporosis, calcinosis, and intestinal perforation; however, with early treatment, children with JDM can expect full recovery and to live a normal life as compared to adults with dermatomyositis.10
Prior to our patient's diagnosis, the family was assigned to move to an overseas location through the US Military with no direct access to advanced medical care. Early detection and diagnosis of JDM through an astute clinical examination allowed the patient and her family to remain in the continental United States to continue receiving specialty care.
The Diagnosis: Juvenile Dermatomyositis
Juvenile dermatomyositis (JDM) is a rare idiopathic inflammatory myopathy of childhood that is autoimmune in nature with an annual incidence ranging from 2.5 to 4.1 cases per million children. Its peak incidence is between 5 and 10 years of age, and it affects girls more than boys at a 2-fold to 5-fold greater rate.1 Juvenile dermatomyositis is characterized by skeletal muscle weakness in the presence of distinctive rashes, including Gottron papules and heliotrope erythema. Muscle weakness typically is proximal and symmetrical, and eventually patients may have trouble rising from a seated position or lifting objects overhead. Other skin manifestations include nail fold capillary changes, calcinosis cutis, and less commonly ulcerations signifying vasculopathy of the skin.2 A subset of patients will present with juvenile amyopathic dermatomyositis. These children have the characteristic skin changes without the muscle weakness or elevated muscle enzymes for more than 6 months; however, one-quarter may go on to develop mysositis.3
Diagnosis of JDM traditionally was based on the following 5 diagnostic criteria: characteristic skin rash, proximal muscle weakness, elevated muscle enzymes, myopathic changes on electromyogram, and typical muscle biopsy.1 Current practice shows a broadening of diagnostic criteria using new techniques in the diagnosis of JDM. To make the diagnosis, the patient must have the characteristic skin manifestations with a minimum of 3 other criteria.4 A 2006 international consensus survey expanded the list of criteria to include typical findings on magnetic resonance imaging (MRI), nail fold capillaroscopy abnormalities, calcinosis, and
dysphonia.5
To assess muscle disease, MRI is utilized because it is a reliable noninvasive tool to assess muscle inflammation. Muscle biopsy is only recommended if the diagnosis is unclear.5 The results of the MRI in our patient displayed symmetric mild fatty atrophy of the gluteus maximus muscle, as well as edema in the right rectus femoris and left vastus lateralis muscles, suggesting early findings of myositis. Muscle enzymes may not be diagnostic because they are not always elevated at diagnosis. Our patient had a normal creatinine kinase level (92 U/L [reference range, <190 U/L]), and both aldolase and lactate dehydrogenase also were within reference range. Conversely, antinuclear antibodies frequently are positive in patients with JDM, such as in our patient at a 1:320 dilution, but are nonspecific and nondiagnostic. It is recommended to include nail fold capillaroscopy to evaluate periungual capillary changes because nailfold capillary density is a sensitive measure of both skin and muscle disease.5 Using dermoscopy, nail fold capillary dilation was observed in our patient.
Other differential diagnoses can have somewhat similar clinical features to JDM. Infantile papular acrodermatitis, commonly referred to as Gianotti-Crosti syndrome, is a viral exanthem that affects children (median age, 2 years).6 The rash appears as monomorphous, flat-topped, pink to brown papules affecting the face, buttocks, and arms; it typically spontaneously resolves in 10 days.6
Juvenile-onset lupus is a chronic autoimmune disorder that can involve any organ system and typically affects children aged 11 to 12 years with a female preponderance. Skin manifestations are similar to adult-onset lupus and include malar rash, discoid rash, oral ulcerations, petechiae, palpable purpura, and digital telangiectasia and ulcers. 7
Juvenile scleroderma is rare connective-tissue disorder that also has multiple organ involvement. Cutaneous involvement can range from isolated morphealike plaques to diffuse sclerotic lesions with growth disturbances, contractures, and facial atrophy.8
Verrucae planae, commonly referred to as flat warts, are papules caused primarily by human papillomavirus types 3, 10, 28, and 41. Children and young adults commonly are affected, and warts can appear on the hands, as in our patient.6
Treatment of JDM depends on disease severity at initial presentation and requires a multidisciplinary approach. The mainstay of treatment is high-dose oral prednisone in combination with disease-modifying drugs such as methotrexate and cyclosporin A. Patients with more severe presentations (eg, ulcerative skin disease) or life-threatening organ involvement are treated with cyclophosphamide, usually in combination with high-dose glucocorticoids.9
Early detection with aggressive treatment is vital to reduce morbidity and mortality from organ damage and disease complications. Mortality rates have dropped to 3%10 in recent decades with the use of systemic glucocorticoids. Delayed treatment is associated with a prolonged disease course and poorer outcomes. Disease complications in children with JDM include osteoporosis, calcinosis, and intestinal perforation; however, with early treatment, children with JDM can expect full recovery and to live a normal life as compared to adults with dermatomyositis.10
Prior to our patient's diagnosis, the family was assigned to move to an overseas location through the US Military with no direct access to advanced medical care. Early detection and diagnosis of JDM through an astute clinical examination allowed the patient and her family to remain in the continental United States to continue receiving specialty care.
- Mendez EP, Lipton R, Ramsey-Goldman R, et al. US incidence of juvenile dermatomyositis,1995-1998: results from the National Institute of Arthritis and Musculoskeletal and Skin Diseases Registry. Arthritis Rheum. 2003;49:300-305.
- Shah M, Mamyrova G, Targoff IN, et al. The clinical phenotypes of the juvenile idiopathic inflammatory myopathies. Medicine. 2013;92:25-41.
- Gerami P, Walling HW, Lewis J, et al. A systematic review of juvenile-onset clinically amyopathic dermatomyositis. Br J Dermatol. 2007;57:637-644.
- Enders FB, Bader-Meunier B, Baildam E, et al. Consensus-based recommendations for the management of juvenile dermatomyositis. Ann Rheum Dis. 2017;76:329-340.
- Brown VE, Pilkington CA, Feldman BM, et al. An international consensus survey of the diagnostic criteria for juvenile dermatomyositis (JDM). Rheumatology (Oxford). 2006;45:990-993.
- William JD, Berger TG, Elston DM. Viral diseases. In: William JD, Berger TG, Elston DM. Andrews' Diseases of the Skin: Clinical Dermatology. 11th ed. China: Saunders Elsevier; 2011:360-413.
- Levy DM, Kamphuis S. Systemic lupus erythematosus in children and adolescents. Pediatr Clin North Am. 2012;59:345-364.
- Li SC, Torok KS, Pope E, et al; Childhood Arthritis and Rheumatology Research Alliance (CARRA) Localized Scleroderma Workgroup. Development of consensus treatment plans for juvenile localized scleroderma: a roadmap toward comparative effectiveness studies in juvenile localized scleroderma. Arthritis Care Res (Hoboken). 2012;64:1175-1185.
- Stringer E, Ota S, Bohnsack J, et al. Treatment approaches to juvenile dermatomyositis (JDM) across North America: the Childhood Arthritis and Rheumatology Research Alliance (CARRA) JDM treatment study. J Rhematol. 2010;37:S1953-S1961.
- Huber AM, Feldman BM. Long-term outcomes in juvenile dermatomyositis: how did we get here and where are we going? Curr Rheumatol Rep. 2005;7:441-446.
- Mendez EP, Lipton R, Ramsey-Goldman R, et al. US incidence of juvenile dermatomyositis,1995-1998: results from the National Institute of Arthritis and Musculoskeletal and Skin Diseases Registry. Arthritis Rheum. 2003;49:300-305.
- Shah M, Mamyrova G, Targoff IN, et al. The clinical phenotypes of the juvenile idiopathic inflammatory myopathies. Medicine. 2013;92:25-41.
- Gerami P, Walling HW, Lewis J, et al. A systematic review of juvenile-onset clinically amyopathic dermatomyositis. Br J Dermatol. 2007;57:637-644.
- Enders FB, Bader-Meunier B, Baildam E, et al. Consensus-based recommendations for the management of juvenile dermatomyositis. Ann Rheum Dis. 2017;76:329-340.
- Brown VE, Pilkington CA, Feldman BM, et al. An international consensus survey of the diagnostic criteria for juvenile dermatomyositis (JDM). Rheumatology (Oxford). 2006;45:990-993.
- William JD, Berger TG, Elston DM. Viral diseases. In: William JD, Berger TG, Elston DM. Andrews' Diseases of the Skin: Clinical Dermatology. 11th ed. China: Saunders Elsevier; 2011:360-413.
- Levy DM, Kamphuis S. Systemic lupus erythematosus in children and adolescents. Pediatr Clin North Am. 2012;59:345-364.
- Li SC, Torok KS, Pope E, et al; Childhood Arthritis and Rheumatology Research Alliance (CARRA) Localized Scleroderma Workgroup. Development of consensus treatment plans for juvenile localized scleroderma: a roadmap toward comparative effectiveness studies in juvenile localized scleroderma. Arthritis Care Res (Hoboken). 2012;64:1175-1185.
- Stringer E, Ota S, Bohnsack J, et al. Treatment approaches to juvenile dermatomyositis (JDM) across North America: the Childhood Arthritis and Rheumatology Research Alliance (CARRA) JDM treatment study. J Rhematol. 2010;37:S1953-S1961.
- Huber AM, Feldman BM. Long-term outcomes in juvenile dermatomyositis: how did we get here and where are we going? Curr Rheumatol Rep. 2005;7:441-446.
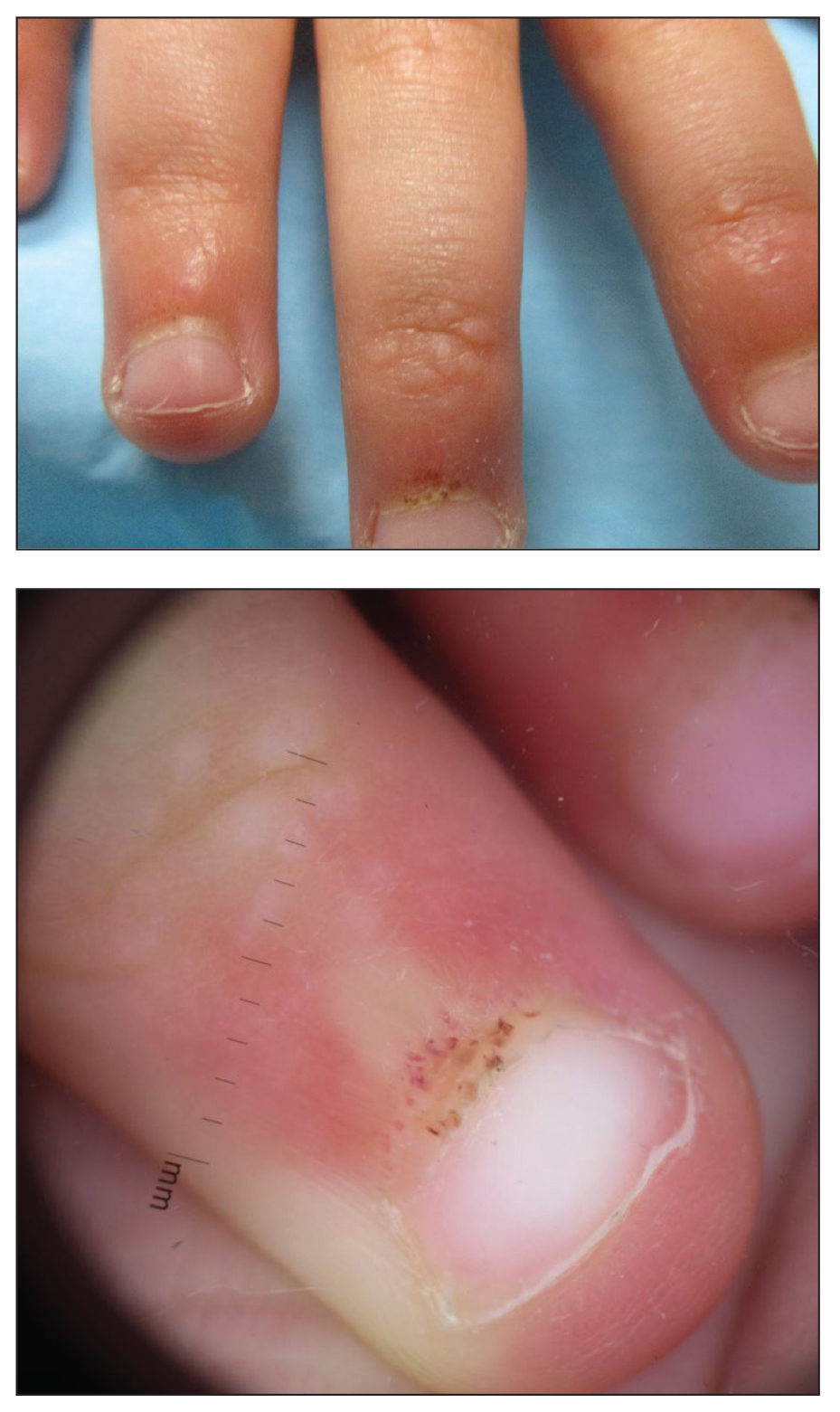
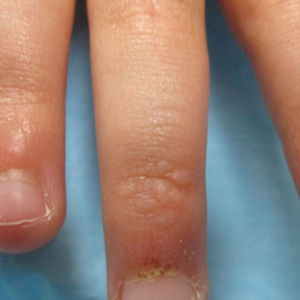
A 4-year-old girl presented to our dermatology clinic with asymptomatic flesh-colored bumps on the fingers of 2 to 3 months’ duration. Prior to presentation the patient was otherwise healthy with normal growth and development. She was referred to dermatology for recommended treatment options for suspected flat warts. On physical examination, grouped 1- to 3-mm, smooth, flat-topped papules were found on the dorsal aspects of the distal interphalangeal joints of all fingers (top). The papules were nonpruritic. Additionally, there were nail findings of ragged cuticles and dilated capillary loops in the proximal nail folds (bottom). The patient did not bite her nails, per the mother’s report, and no other rashes were noted. There were no systemic symptoms or reports of muscle fatigue. She was positive for antinuclear antibodies at 1:320 dilution. Magnetic resonance imaging of the thighs and pelvis was ordered.
Large measles outbreak reported in Michigan
A new measles outbreak in Michigan has already resulted in 39 cases, and four more states reported their first cases of 2019 during the week ending April 4, according to the Centers for Disease Control and Prevention
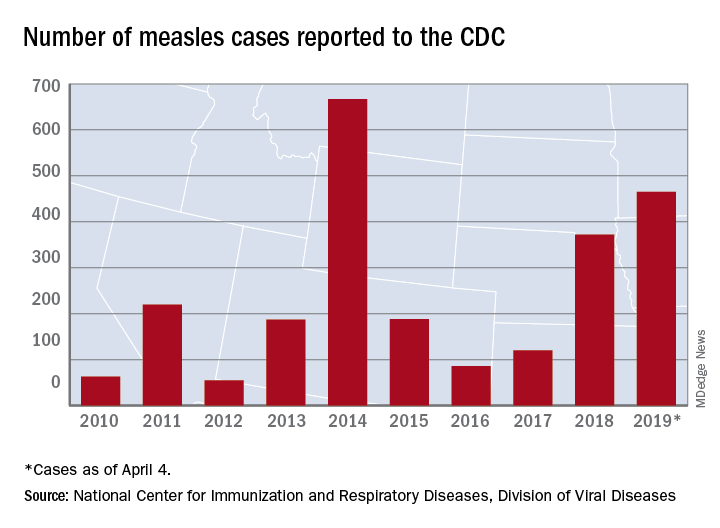
The measles virus has now infected individuals in Florida, Indiana, Massachusetts, and Nevada, which means that 19 states have now reported a total of 465 cases this year, and that is the second-highest total “reported in the U.S. since measles was eliminated in 2000,” the CDC said April 8.
The Michigan outbreak is mostly concentrated in Oakland County, where 38 cases have occurred. The county has posted an up-to-date list of exposure locations.
Not to be outdone, New York reported 45 new cases last week: 44 in Brooklyn and 1 in Queens. There have been 259 confirmed cases in the two boroughs since the outbreak began in October of last year.
Besides Michigan and New York City, there are five other outbreaks ongoing in the United States: Rockland County, N.Y.; Washington State (no new cases since March 22); Butte County, Calif.; Santa Cruz County, Calif.; and New Jersey, the CDC reported.
A judge in New York State temporarily blocked an order banning unimmunized children from public spaces in Rockland County and has set a hearing date of April 19, CNN reported. The ban, ordered by Rockland County Executive Ed Day, went into effect on March 27.
On April 2, the Maine Center for Disease Control & Prevention announced that an out-of-state resident with a confirmed case of measles had visited two health care offices – one in Falmouth and one in Westbrook – on March 27. No cases in Maine residents have been reported yet.
On a vaccine-related note, the Washington State Senate’s Health and Long Term Care Committee approved a proposal on April 1 that would “end the personal exemption for parents who don’t want their children vaccinated against measles,” the Spokane Spokesman-Review said. The bill, which would still allow medical and religious exemptions, has already passed the state’s House of Representatives and goes next to the full senate.
A new measles outbreak in Michigan has already resulted in 39 cases, and four more states reported their first cases of 2019 during the week ending April 4, according to the Centers for Disease Control and Prevention

The measles virus has now infected individuals in Florida, Indiana, Massachusetts, and Nevada, which means that 19 states have now reported a total of 465 cases this year, and that is the second-highest total “reported in the U.S. since measles was eliminated in 2000,” the CDC said April 8.
The Michigan outbreak is mostly concentrated in Oakland County, where 38 cases have occurred. The county has posted an up-to-date list of exposure locations.
Not to be outdone, New York reported 45 new cases last week: 44 in Brooklyn and 1 in Queens. There have been 259 confirmed cases in the two boroughs since the outbreak began in October of last year.
Besides Michigan and New York City, there are five other outbreaks ongoing in the United States: Rockland County, N.Y.; Washington State (no new cases since March 22); Butte County, Calif.; Santa Cruz County, Calif.; and New Jersey, the CDC reported.
A judge in New York State temporarily blocked an order banning unimmunized children from public spaces in Rockland County and has set a hearing date of April 19, CNN reported. The ban, ordered by Rockland County Executive Ed Day, went into effect on March 27.
On April 2, the Maine Center for Disease Control & Prevention announced that an out-of-state resident with a confirmed case of measles had visited two health care offices – one in Falmouth and one in Westbrook – on March 27. No cases in Maine residents have been reported yet.
On a vaccine-related note, the Washington State Senate’s Health and Long Term Care Committee approved a proposal on April 1 that would “end the personal exemption for parents who don’t want their children vaccinated against measles,” the Spokane Spokesman-Review said. The bill, which would still allow medical and religious exemptions, has already passed the state’s House of Representatives and goes next to the full senate.
A new measles outbreak in Michigan has already resulted in 39 cases, and four more states reported their first cases of 2019 during the week ending April 4, according to the Centers for Disease Control and Prevention

The measles virus has now infected individuals in Florida, Indiana, Massachusetts, and Nevada, which means that 19 states have now reported a total of 465 cases this year, and that is the second-highest total “reported in the U.S. since measles was eliminated in 2000,” the CDC said April 8.
The Michigan outbreak is mostly concentrated in Oakland County, where 38 cases have occurred. The county has posted an up-to-date list of exposure locations.
Not to be outdone, New York reported 45 new cases last week: 44 in Brooklyn and 1 in Queens. There have been 259 confirmed cases in the two boroughs since the outbreak began in October of last year.
Besides Michigan and New York City, there are five other outbreaks ongoing in the United States: Rockland County, N.Y.; Washington State (no new cases since March 22); Butte County, Calif.; Santa Cruz County, Calif.; and New Jersey, the CDC reported.
A judge in New York State temporarily blocked an order banning unimmunized children from public spaces in Rockland County and has set a hearing date of April 19, CNN reported. The ban, ordered by Rockland County Executive Ed Day, went into effect on March 27.
On April 2, the Maine Center for Disease Control & Prevention announced that an out-of-state resident with a confirmed case of measles had visited two health care offices – one in Falmouth and one in Westbrook – on March 27. No cases in Maine residents have been reported yet.
On a vaccine-related note, the Washington State Senate’s Health and Long Term Care Committee approved a proposal on April 1 that would “end the personal exemption for parents who don’t want their children vaccinated against measles,” the Spokane Spokesman-Review said. The bill, which would still allow medical and religious exemptions, has already passed the state’s House of Representatives and goes next to the full senate.
Direct-to-consumer telemedicine visits may lead to pediatric antibiotic overprescribing
(ARIs), according to a study of antibiotic prescriptions for ARIs across 3 clinical settings.
“These differences in antibiotic prescribing for children contrast with previous studies of DTC telemedicine quality among adult patients in which quality differences have been smaller or nonexistent,” wrote Kristin N. Ray, MD, of Children’s Hospital of Pittsburgh, and her coauthors. The study was published in Pediatrics.
To determine quality of care during pediatric DTC telemedicine visits, the researchers embarked on a retrospective cohort study using 2015–2016 claims data from a large national commercial health plan. They identified visits for ARIs and matched them across 3 settings: DTC telemedicine, urgent care, and PCP offices. The matched sample included 4,604 DTC telemedicine visits, 38,408 urgent care visits, and 485,201 PCP visits.
Their analysis showed that children were more likely to be prescribed antibiotics at DTC telemedicine visits than in other settings (52% versus 42% for urgent care and 31% for PCP, P less than .001). In addition, they were less likely to receive guideline-concordant antibiotic management (59% versus 67% and 78%, P less than .001). This was primarily attributed to “antibiotic prescribing for visits with viral ARI diagnoses that do not warrant antibiotics,” antibiotics were appropriately not prescribed in only 54% of those DTC telemedicine visits, compared with 66% for urgent care and 80% for PCP (P less than .001).
The authors shared the limitations of their study, including a lack of sociodemographic or clinical data stemming from a reliance on insurance claims. They also noted that their analysis was limited to a specific health plan and its contracted DTC telemedicine vendor, recognizing that “antibiotic prescribing among other DTC telemedicine companies, models, and populations may differ.”
The study was funded by the National Institutes of Health and supported in part by grants from the Eunice Kennedy Shriver National Institute of Child Health and Human Development and gifts from Melvin Hall. The authors reported no conflicts of interest.
SOURCE: Ray KN et al. Pediatrics. 2019 Apr 8. doi: 10.1542/peds.2018-2491.
These findings from this study illustrate the issues with direct-to-consumer (DTC) telemedicine, especially when treating children, according to Jeffrey S. Gerber, MD, medical director of the antimicrobial stewardship program at Children’s Hospital of Philadelphia.
The best way to get a 5-star rating after a DTC telemedicine visit is to prescribe an antibiotic, Dr. Gerber wrote, so it shouldn’t be surprising that doctors are handing them out at a higher rate than after an urgent care or a primary care visit. It should also be noted that this study covers a very specific privately insured population and that DTC telemedicine remains a “small piece of the pie,” for now, in terms of patient care.
But, he added, the most problematic element of this study may be that none of the 3 most common pediatric acute respiratory tract infection (ARTI) diagnoses should be followed with an immediate prescription, especially after a virtual visit.
“It could be argued that essentially no ARTI encounters should lead to antibiotic prescriptions solely on the basis of a DTC telemedicine visit,” he wrote, recognizing that – though there may be value for telemedicine in a screening capacity – the DTC version seems to be a “low quality encounter” at best and “a vehicle for antibiotic overuse” at worst.
These comments are adapted from an accompanying editorial (Pediatrics. 2019 Apr 8. doi: 10.1542/peds.2019-0631 ). Dr. Gerber reported receiving personal fees from Medtronic outside the submitted work.
These findings from this study illustrate the issues with direct-to-consumer (DTC) telemedicine, especially when treating children, according to Jeffrey S. Gerber, MD, medical director of the antimicrobial stewardship program at Children’s Hospital of Philadelphia.
The best way to get a 5-star rating after a DTC telemedicine visit is to prescribe an antibiotic, Dr. Gerber wrote, so it shouldn’t be surprising that doctors are handing them out at a higher rate than after an urgent care or a primary care visit. It should also be noted that this study covers a very specific privately insured population and that DTC telemedicine remains a “small piece of the pie,” for now, in terms of patient care.
But, he added, the most problematic element of this study may be that none of the 3 most common pediatric acute respiratory tract infection (ARTI) diagnoses should be followed with an immediate prescription, especially after a virtual visit.
“It could be argued that essentially no ARTI encounters should lead to antibiotic prescriptions solely on the basis of a DTC telemedicine visit,” he wrote, recognizing that – though there may be value for telemedicine in a screening capacity – the DTC version seems to be a “low quality encounter” at best and “a vehicle for antibiotic overuse” at worst.
These comments are adapted from an accompanying editorial (Pediatrics. 2019 Apr 8. doi: 10.1542/peds.2019-0631 ). Dr. Gerber reported receiving personal fees from Medtronic outside the submitted work.
These findings from this study illustrate the issues with direct-to-consumer (DTC) telemedicine, especially when treating children, according to Jeffrey S. Gerber, MD, medical director of the antimicrobial stewardship program at Children’s Hospital of Philadelphia.
The best way to get a 5-star rating after a DTC telemedicine visit is to prescribe an antibiotic, Dr. Gerber wrote, so it shouldn’t be surprising that doctors are handing them out at a higher rate than after an urgent care or a primary care visit. It should also be noted that this study covers a very specific privately insured population and that DTC telemedicine remains a “small piece of the pie,” for now, in terms of patient care.
But, he added, the most problematic element of this study may be that none of the 3 most common pediatric acute respiratory tract infection (ARTI) diagnoses should be followed with an immediate prescription, especially after a virtual visit.
“It could be argued that essentially no ARTI encounters should lead to antibiotic prescriptions solely on the basis of a DTC telemedicine visit,” he wrote, recognizing that – though there may be value for telemedicine in a screening capacity – the DTC version seems to be a “low quality encounter” at best and “a vehicle for antibiotic overuse” at worst.
These comments are adapted from an accompanying editorial (Pediatrics. 2019 Apr 8. doi: 10.1542/peds.2019-0631 ). Dr. Gerber reported receiving personal fees from Medtronic outside the submitted work.
(ARIs), according to a study of antibiotic prescriptions for ARIs across 3 clinical settings.
“These differences in antibiotic prescribing for children contrast with previous studies of DTC telemedicine quality among adult patients in which quality differences have been smaller or nonexistent,” wrote Kristin N. Ray, MD, of Children’s Hospital of Pittsburgh, and her coauthors. The study was published in Pediatrics.
To determine quality of care during pediatric DTC telemedicine visits, the researchers embarked on a retrospective cohort study using 2015–2016 claims data from a large national commercial health plan. They identified visits for ARIs and matched them across 3 settings: DTC telemedicine, urgent care, and PCP offices. The matched sample included 4,604 DTC telemedicine visits, 38,408 urgent care visits, and 485,201 PCP visits.
Their analysis showed that children were more likely to be prescribed antibiotics at DTC telemedicine visits than in other settings (52% versus 42% for urgent care and 31% for PCP, P less than .001). In addition, they were less likely to receive guideline-concordant antibiotic management (59% versus 67% and 78%, P less than .001). This was primarily attributed to “antibiotic prescribing for visits with viral ARI diagnoses that do not warrant antibiotics,” antibiotics were appropriately not prescribed in only 54% of those DTC telemedicine visits, compared with 66% for urgent care and 80% for PCP (P less than .001).
The authors shared the limitations of their study, including a lack of sociodemographic or clinical data stemming from a reliance on insurance claims. They also noted that their analysis was limited to a specific health plan and its contracted DTC telemedicine vendor, recognizing that “antibiotic prescribing among other DTC telemedicine companies, models, and populations may differ.”
The study was funded by the National Institutes of Health and supported in part by grants from the Eunice Kennedy Shriver National Institute of Child Health and Human Development and gifts from Melvin Hall. The authors reported no conflicts of interest.
SOURCE: Ray KN et al. Pediatrics. 2019 Apr 8. doi: 10.1542/peds.2018-2491.
(ARIs), according to a study of antibiotic prescriptions for ARIs across 3 clinical settings.
“These differences in antibiotic prescribing for children contrast with previous studies of DTC telemedicine quality among adult patients in which quality differences have been smaller or nonexistent,” wrote Kristin N. Ray, MD, of Children’s Hospital of Pittsburgh, and her coauthors. The study was published in Pediatrics.
To determine quality of care during pediatric DTC telemedicine visits, the researchers embarked on a retrospective cohort study using 2015–2016 claims data from a large national commercial health plan. They identified visits for ARIs and matched them across 3 settings: DTC telemedicine, urgent care, and PCP offices. The matched sample included 4,604 DTC telemedicine visits, 38,408 urgent care visits, and 485,201 PCP visits.
Their analysis showed that children were more likely to be prescribed antibiotics at DTC telemedicine visits than in other settings (52% versus 42% for urgent care and 31% for PCP, P less than .001). In addition, they were less likely to receive guideline-concordant antibiotic management (59% versus 67% and 78%, P less than .001). This was primarily attributed to “antibiotic prescribing for visits with viral ARI diagnoses that do not warrant antibiotics,” antibiotics were appropriately not prescribed in only 54% of those DTC telemedicine visits, compared with 66% for urgent care and 80% for PCP (P less than .001).
The authors shared the limitations of their study, including a lack of sociodemographic or clinical data stemming from a reliance on insurance claims. They also noted that their analysis was limited to a specific health plan and its contracted DTC telemedicine vendor, recognizing that “antibiotic prescribing among other DTC telemedicine companies, models, and populations may differ.”
The study was funded by the National Institutes of Health and supported in part by grants from the Eunice Kennedy Shriver National Institute of Child Health and Human Development and gifts from Melvin Hall. The authors reported no conflicts of interest.
SOURCE: Ray KN et al. Pediatrics. 2019 Apr 8. doi: 10.1542/peds.2018-2491.
FROM PEDIATRICS
Key clinical point: For children diagnosed with acute respiratory infections, antibiotic prescribing was higher and guideline-concordant antibiotic management was lower at direct-to-consumer (DTC) telemedicine visits.
Major finding: Children at DTC telemedicine visits were prescribed antibiotics for respiratory infections 52% of the time, compared with 42% at urgent care visits and 31% at primary care provider visits.
Study details: A retrospective cohort study of DTC telemedicine, urgent care, and primary care provider visits for acute respiratory infections and subsequent antibiotic prescriptions.
Disclosures: The study was funded by the National Institutes of Health and supported in part by grants from the Eunice Kennedy Shriver National Institute of Child Health and Human Development and gifts from Melvin Hall. The authors reported no conflicts of interest.
Source: Ray KN et al. Pediatrics. 2019 Apr 8. doi: 10.1542/peds.2018-2491.
Oscillatory ventilation reduced reintubation risk for preterm infants
Nasal high-frequency oscillatory ventilation, in a randomized trial of 206 preterm infants with respiratory failure.
Previous studies have supported the use of NHFOV as more effective for reducing CO2 and for lowering the risk of reintubation compared with NCPAP. But no randomized, controlled trials had compared the outcomes for preterm infants in particular, wrote Long Chen, MD, PhD, of Children’s Hospital of Chongqing Medical University, Chongqing, China, and colleagues.
Their study, published in Chest, was conducted at a single tertiary NICU in China between May 2017 and May 2018, and randomized infants with a gestational age less than 37 weeks to NHFOV (103 infants) or NCPAP (103 infants). Infants with major congenital abnormalities were excluded. The infants included 127 (61.7%) diagnosed with respiratory distress syndrome (RDS), 53 (25.7%) diagnosed with acute RDS (ARDS), and 26 (12.6%) diagnosed with both RDS and ARDS.
Overall, the reintubation rate within 6 hours was significantly lower among infants treated with NHFOV compared with those treated with NCPAP (15.5% vs. 34%, P = .002), and in the subset of infants with ARDS (23.5% vs. 52.6%, P = .032). Among infants with a gestational age of 32 weeks or less, reintuibation rates were also significantly lower among those treated with NHFOV (26.1% vs. 55.6%, P = .004).
In addition, PCO2 levels, 6 hours after extubation, were significantly lower among infants on NHFOV, compared with those on NCPAP (49.6 vs. 56.9 P = .00). The hospital stay, a secondary outcome, was significantly shorter among the infants treated with NHFOV, than those treated with NCPAP (22 days, vs. 27.6 days, P =.011).
Although the researchers observed some nasal trauma in NHFOV-treated patients, and intestinal dilation in both groups similar to side effects seen in previous studies, no feeding intolerance or skin lesions were associated with NHFOV. The study findings were consistent with those from previous studies, and suggested that the causes of respiratory failure might account for the differences between the treatment groups, they noted.
“RDS is primarily restrictive in the acute phase, and the high frequency oscillation over CPAP does not therefore bring any benefit. However, ARDS is both restrictive and obstructive in the acute phase due to the nature of ARDS,” and NHFOV is “able to improve oxygenation,” they added.
The study findings were limited by several factors including the use of data from a single center and the small number of infants younger than 28 weeks’ gestation, the researchers noted. However, they added, two international, multicenter, randomized controlled trials are in the works.
The study was supported by Social Livelihood Program of 38 Chongqing Science and Technology Commission, China. The researchers had no financial conflicts to disclose.
SOURCE: Long C et al. Chest. 2019; 155(4): 740-8.
Nasal high-frequency oscillatory ventilation, in a randomized trial of 206 preterm infants with respiratory failure.
Previous studies have supported the use of NHFOV as more effective for reducing CO2 and for lowering the risk of reintubation compared with NCPAP. But no randomized, controlled trials had compared the outcomes for preterm infants in particular, wrote Long Chen, MD, PhD, of Children’s Hospital of Chongqing Medical University, Chongqing, China, and colleagues.
Their study, published in Chest, was conducted at a single tertiary NICU in China between May 2017 and May 2018, and randomized infants with a gestational age less than 37 weeks to NHFOV (103 infants) or NCPAP (103 infants). Infants with major congenital abnormalities were excluded. The infants included 127 (61.7%) diagnosed with respiratory distress syndrome (RDS), 53 (25.7%) diagnosed with acute RDS (ARDS), and 26 (12.6%) diagnosed with both RDS and ARDS.
Overall, the reintubation rate within 6 hours was significantly lower among infants treated with NHFOV compared with those treated with NCPAP (15.5% vs. 34%, P = .002), and in the subset of infants with ARDS (23.5% vs. 52.6%, P = .032). Among infants with a gestational age of 32 weeks or less, reintuibation rates were also significantly lower among those treated with NHFOV (26.1% vs. 55.6%, P = .004).
In addition, PCO2 levels, 6 hours after extubation, were significantly lower among infants on NHFOV, compared with those on NCPAP (49.6 vs. 56.9 P = .00). The hospital stay, a secondary outcome, was significantly shorter among the infants treated with NHFOV, than those treated with NCPAP (22 days, vs. 27.6 days, P =.011).
Although the researchers observed some nasal trauma in NHFOV-treated patients, and intestinal dilation in both groups similar to side effects seen in previous studies, no feeding intolerance or skin lesions were associated with NHFOV. The study findings were consistent with those from previous studies, and suggested that the causes of respiratory failure might account for the differences between the treatment groups, they noted.
“RDS is primarily restrictive in the acute phase, and the high frequency oscillation over CPAP does not therefore bring any benefit. However, ARDS is both restrictive and obstructive in the acute phase due to the nature of ARDS,” and NHFOV is “able to improve oxygenation,” they added.
The study findings were limited by several factors including the use of data from a single center and the small number of infants younger than 28 weeks’ gestation, the researchers noted. However, they added, two international, multicenter, randomized controlled trials are in the works.
The study was supported by Social Livelihood Program of 38 Chongqing Science and Technology Commission, China. The researchers had no financial conflicts to disclose.
SOURCE: Long C et al. Chest. 2019; 155(4): 740-8.
Nasal high-frequency oscillatory ventilation, in a randomized trial of 206 preterm infants with respiratory failure.
Previous studies have supported the use of NHFOV as more effective for reducing CO2 and for lowering the risk of reintubation compared with NCPAP. But no randomized, controlled trials had compared the outcomes for preterm infants in particular, wrote Long Chen, MD, PhD, of Children’s Hospital of Chongqing Medical University, Chongqing, China, and colleagues.
Their study, published in Chest, was conducted at a single tertiary NICU in China between May 2017 and May 2018, and randomized infants with a gestational age less than 37 weeks to NHFOV (103 infants) or NCPAP (103 infants). Infants with major congenital abnormalities were excluded. The infants included 127 (61.7%) diagnosed with respiratory distress syndrome (RDS), 53 (25.7%) diagnosed with acute RDS (ARDS), and 26 (12.6%) diagnosed with both RDS and ARDS.
Overall, the reintubation rate within 6 hours was significantly lower among infants treated with NHFOV compared with those treated with NCPAP (15.5% vs. 34%, P = .002), and in the subset of infants with ARDS (23.5% vs. 52.6%, P = .032). Among infants with a gestational age of 32 weeks or less, reintuibation rates were also significantly lower among those treated with NHFOV (26.1% vs. 55.6%, P = .004).
In addition, PCO2 levels, 6 hours after extubation, were significantly lower among infants on NHFOV, compared with those on NCPAP (49.6 vs. 56.9 P = .00). The hospital stay, a secondary outcome, was significantly shorter among the infants treated with NHFOV, than those treated with NCPAP (22 days, vs. 27.6 days, P =.011).
Although the researchers observed some nasal trauma in NHFOV-treated patients, and intestinal dilation in both groups similar to side effects seen in previous studies, no feeding intolerance or skin lesions were associated with NHFOV. The study findings were consistent with those from previous studies, and suggested that the causes of respiratory failure might account for the differences between the treatment groups, they noted.
“RDS is primarily restrictive in the acute phase, and the high frequency oscillation over CPAP does not therefore bring any benefit. However, ARDS is both restrictive and obstructive in the acute phase due to the nature of ARDS,” and NHFOV is “able to improve oxygenation,” they added.
The study findings were limited by several factors including the use of data from a single center and the small number of infants younger than 28 weeks’ gestation, the researchers noted. However, they added, two international, multicenter, randomized controlled trials are in the works.
The study was supported by Social Livelihood Program of 38 Chongqing Science and Technology Commission, China. The researchers had no financial conflicts to disclose.
SOURCE: Long C et al. Chest. 2019; 155(4): 740-8.
FROM CHEST
Addressing anxiety helps youth with functional abdominal pain disorders
MILWAUKEE – A stepped-care approach to youth with functional abdominal pain disorders may be effective in targeting those with comorbid anxiety, according to ongoing research.
A study of 79 pediatric patients with a functional abdominal pain disorder (FAPD) and co-occurring anxiety found that those who received cognitive behavioral therapy (CBT) that included a component to address anxiety had less functional disability and anxiety than those who received treatment as usual. Pain scores also dropped, though the difference was not statistically significant.
The patients, aged 9-14 years and mostly white and female, were randomized to treatment allocation. Functional disability scores were significantly lower post-treatment for those who received the stepped therapy compared with the treatment as usual group (P less than .05, Cohen’s D = .49). This indicates a moderate effect size, said Natoshia Cunningham, PhD, speaking at the scientific meeting of the American Pain Society.
Mean scores on an anxiety rating scale also dropped below the threshold for clinical anxiety for those receiving the stepped therapy; on average, the treatment as usual group still scored above the clinical anxiety threshold after treatment (P for difference = .05).
The study, part of ongoing research, tests a hybrid online intervention, dubbed Aim to Decrease Anxiety and Pain Treatment, or ADAPT. The ADAPT program includes some common elements of CBT for anxiety that were not previously included in the pediatric pain CBT in use for the FAPD patients, she said.
The hybrid program began with two in-person sessions, each lasting one hour. These were followed by up to four web-based sessions. Patients viewed videos, read some material online, and complete activities with follow-up assessments. The web-based component was structured so that providers can see how patients fare on assessments – and even see which activities had been opened or completed. This, said Dr. Cunningham, allowed the treating provider to tailor what’s addressed in the associated weekly phone checks that accompany the online content.
Parents were also given practical, evidence-based advice to help manage their child’s FAPD. These include encouraging children to be independent in pain management, stopping “status checks,” encouraging normal school and social activities, and avoiding special privileges when pain interferes with activities.
Overall, up to 40% of pediatric functional abdominal pain patients may not respond to CBT, the most efficacious treatment known, said Dr. Cunningham, a pediatric psychologist at the University of Cincinnati. Her research indicates that comorbid anxiety may predict poor response, and that addressing anxiety improves pain and disability in this complex, common disorder.
With a brief psychosocial screening that identifies patients with anxiety, Dr. Cunningham and her colleagues can implement the targeted, partially web-based therapy strategy that tackles anxiety along with CBT for functional abdominal pain.
“Anxiety is common and related to poor outcomes,” noted Dr. Cunningham, She added that overall, half or more of individuals with chronic pain also have anxiety. Among children with FAPD, “Clinical anxiety predicts disability and poor treatment response.”
The first step, she said, was identifying the patients with FAPD who had anxiety, including those with subclinical anxiety.
At intake, children coming to the Cincinnati Children’s Hospital’s gastroenterology clinic complete anxiety screening via the Screen for Child Anxiety Related Emotional Disorders (SCARED) (Depress Anxiety. 2000;12[2]:85-91). Disability and pain are assessed by the Functional Disability Inventory and the Numeric Rating Scale (J Pediatr Psychol. 1991 Feb;16[1]:39-58).
In earlier research, Dr. Cunningham and her collaborators found a significant association between anxiety and both higher pain levels and more disability. And, clinically significant anxiety was more likely among the FAPD patients with persistent disability after six months of treatment.
A surprising finding from the screenings, said Dr. Cunningham, is that youth endorsed more anxiety symptoms in self-assessment than their parents observed. “Children are often their own best informants of their internalizing symptoms,” she said. “Not only do their parents not notice it, it may not be obvious to their providers, either.”
Since many children with FAPD have anxiety, the next question was “How do we better enhance their treatments?” she continued. To answer that question, she took one step back: “How do these youth respond to our current best practice?”
Looking at Cincinnati Children’s patients with FAPD who did – or did not – have anxiety, Dr. Cunningham found that “those who have clinical levels of anxiety don’t respond as well to CBT.” Pain-directed therapy alone, she said, “is insufficient to treat these patients.”
Together with brief screening, stepped therapy delivered via ADAPT offers promise to boost the efficacy of FAPD treatment, perhaps even in a primary care setting, said Dr. Cunningham. She and her collaborators are continuing to study comorbid anxiety and pain in youth; current work is using functional magnetic resonance imaging to examine cognitive and affective changes in patients receiving the ADAPT intervention.
The study was funded by the American Pain Society Sharon S. Keller Chronic Pain Research Grant, Cincinnati Children’s Hospital, and the National Institutes of Health. Dr. Cunningham reported no relevant conflicts of interest.
[email protected]
SOURCE: Cunningham N. et al. APS 2019.
MILWAUKEE – A stepped-care approach to youth with functional abdominal pain disorders may be effective in targeting those with comorbid anxiety, according to ongoing research.
A study of 79 pediatric patients with a functional abdominal pain disorder (FAPD) and co-occurring anxiety found that those who received cognitive behavioral therapy (CBT) that included a component to address anxiety had less functional disability and anxiety than those who received treatment as usual. Pain scores also dropped, though the difference was not statistically significant.
The patients, aged 9-14 years and mostly white and female, were randomized to treatment allocation. Functional disability scores were significantly lower post-treatment for those who received the stepped therapy compared with the treatment as usual group (P less than .05, Cohen’s D = .49). This indicates a moderate effect size, said Natoshia Cunningham, PhD, speaking at the scientific meeting of the American Pain Society.
Mean scores on an anxiety rating scale also dropped below the threshold for clinical anxiety for those receiving the stepped therapy; on average, the treatment as usual group still scored above the clinical anxiety threshold after treatment (P for difference = .05).
The study, part of ongoing research, tests a hybrid online intervention, dubbed Aim to Decrease Anxiety and Pain Treatment, or ADAPT. The ADAPT program includes some common elements of CBT for anxiety that were not previously included in the pediatric pain CBT in use for the FAPD patients, she said.
The hybrid program began with two in-person sessions, each lasting one hour. These were followed by up to four web-based sessions. Patients viewed videos, read some material online, and complete activities with follow-up assessments. The web-based component was structured so that providers can see how patients fare on assessments – and even see which activities had been opened or completed. This, said Dr. Cunningham, allowed the treating provider to tailor what’s addressed in the associated weekly phone checks that accompany the online content.
Parents were also given practical, evidence-based advice to help manage their child’s FAPD. These include encouraging children to be independent in pain management, stopping “status checks,” encouraging normal school and social activities, and avoiding special privileges when pain interferes with activities.
Overall, up to 40% of pediatric functional abdominal pain patients may not respond to CBT, the most efficacious treatment known, said Dr. Cunningham, a pediatric psychologist at the University of Cincinnati. Her research indicates that comorbid anxiety may predict poor response, and that addressing anxiety improves pain and disability in this complex, common disorder.
With a brief psychosocial screening that identifies patients with anxiety, Dr. Cunningham and her colleagues can implement the targeted, partially web-based therapy strategy that tackles anxiety along with CBT for functional abdominal pain.
“Anxiety is common and related to poor outcomes,” noted Dr. Cunningham, She added that overall, half or more of individuals with chronic pain also have anxiety. Among children with FAPD, “Clinical anxiety predicts disability and poor treatment response.”
The first step, she said, was identifying the patients with FAPD who had anxiety, including those with subclinical anxiety.
At intake, children coming to the Cincinnati Children’s Hospital’s gastroenterology clinic complete anxiety screening via the Screen for Child Anxiety Related Emotional Disorders (SCARED) (Depress Anxiety. 2000;12[2]:85-91). Disability and pain are assessed by the Functional Disability Inventory and the Numeric Rating Scale (J Pediatr Psychol. 1991 Feb;16[1]:39-58).
In earlier research, Dr. Cunningham and her collaborators found a significant association between anxiety and both higher pain levels and more disability. And, clinically significant anxiety was more likely among the FAPD patients with persistent disability after six months of treatment.
A surprising finding from the screenings, said Dr. Cunningham, is that youth endorsed more anxiety symptoms in self-assessment than their parents observed. “Children are often their own best informants of their internalizing symptoms,” she said. “Not only do their parents not notice it, it may not be obvious to their providers, either.”
Since many children with FAPD have anxiety, the next question was “How do we better enhance their treatments?” she continued. To answer that question, she took one step back: “How do these youth respond to our current best practice?”
Looking at Cincinnati Children’s patients with FAPD who did – or did not – have anxiety, Dr. Cunningham found that “those who have clinical levels of anxiety don’t respond as well to CBT.” Pain-directed therapy alone, she said, “is insufficient to treat these patients.”
Together with brief screening, stepped therapy delivered via ADAPT offers promise to boost the efficacy of FAPD treatment, perhaps even in a primary care setting, said Dr. Cunningham. She and her collaborators are continuing to study comorbid anxiety and pain in youth; current work is using functional magnetic resonance imaging to examine cognitive and affective changes in patients receiving the ADAPT intervention.
The study was funded by the American Pain Society Sharon S. Keller Chronic Pain Research Grant, Cincinnati Children’s Hospital, and the National Institutes of Health. Dr. Cunningham reported no relevant conflicts of interest.
[email protected]
SOURCE: Cunningham N. et al. APS 2019.
MILWAUKEE – A stepped-care approach to youth with functional abdominal pain disorders may be effective in targeting those with comorbid anxiety, according to ongoing research.
A study of 79 pediatric patients with a functional abdominal pain disorder (FAPD) and co-occurring anxiety found that those who received cognitive behavioral therapy (CBT) that included a component to address anxiety had less functional disability and anxiety than those who received treatment as usual. Pain scores also dropped, though the difference was not statistically significant.
The patients, aged 9-14 years and mostly white and female, were randomized to treatment allocation. Functional disability scores were significantly lower post-treatment for those who received the stepped therapy compared with the treatment as usual group (P less than .05, Cohen’s D = .49). This indicates a moderate effect size, said Natoshia Cunningham, PhD, speaking at the scientific meeting of the American Pain Society.
Mean scores on an anxiety rating scale also dropped below the threshold for clinical anxiety for those receiving the stepped therapy; on average, the treatment as usual group still scored above the clinical anxiety threshold after treatment (P for difference = .05).
The study, part of ongoing research, tests a hybrid online intervention, dubbed Aim to Decrease Anxiety and Pain Treatment, or ADAPT. The ADAPT program includes some common elements of CBT for anxiety that were not previously included in the pediatric pain CBT in use for the FAPD patients, she said.
The hybrid program began with two in-person sessions, each lasting one hour. These were followed by up to four web-based sessions. Patients viewed videos, read some material online, and complete activities with follow-up assessments. The web-based component was structured so that providers can see how patients fare on assessments – and even see which activities had been opened or completed. This, said Dr. Cunningham, allowed the treating provider to tailor what’s addressed in the associated weekly phone checks that accompany the online content.
Parents were also given practical, evidence-based advice to help manage their child’s FAPD. These include encouraging children to be independent in pain management, stopping “status checks,” encouraging normal school and social activities, and avoiding special privileges when pain interferes with activities.
Overall, up to 40% of pediatric functional abdominal pain patients may not respond to CBT, the most efficacious treatment known, said Dr. Cunningham, a pediatric psychologist at the University of Cincinnati. Her research indicates that comorbid anxiety may predict poor response, and that addressing anxiety improves pain and disability in this complex, common disorder.
With a brief psychosocial screening that identifies patients with anxiety, Dr. Cunningham and her colleagues can implement the targeted, partially web-based therapy strategy that tackles anxiety along with CBT for functional abdominal pain.
“Anxiety is common and related to poor outcomes,” noted Dr. Cunningham, She added that overall, half or more of individuals with chronic pain also have anxiety. Among children with FAPD, “Clinical anxiety predicts disability and poor treatment response.”
The first step, she said, was identifying the patients with FAPD who had anxiety, including those with subclinical anxiety.
At intake, children coming to the Cincinnati Children’s Hospital’s gastroenterology clinic complete anxiety screening via the Screen for Child Anxiety Related Emotional Disorders (SCARED) (Depress Anxiety. 2000;12[2]:85-91). Disability and pain are assessed by the Functional Disability Inventory and the Numeric Rating Scale (J Pediatr Psychol. 1991 Feb;16[1]:39-58).
In earlier research, Dr. Cunningham and her collaborators found a significant association between anxiety and both higher pain levels and more disability. And, clinically significant anxiety was more likely among the FAPD patients with persistent disability after six months of treatment.
A surprising finding from the screenings, said Dr. Cunningham, is that youth endorsed more anxiety symptoms in self-assessment than their parents observed. “Children are often their own best informants of their internalizing symptoms,” she said. “Not only do their parents not notice it, it may not be obvious to their providers, either.”
Since many children with FAPD have anxiety, the next question was “How do we better enhance their treatments?” she continued. To answer that question, she took one step back: “How do these youth respond to our current best practice?”
Looking at Cincinnati Children’s patients with FAPD who did – or did not – have anxiety, Dr. Cunningham found that “those who have clinical levels of anxiety don’t respond as well to CBT.” Pain-directed therapy alone, she said, “is insufficient to treat these patients.”
Together with brief screening, stepped therapy delivered via ADAPT offers promise to boost the efficacy of FAPD treatment, perhaps even in a primary care setting, said Dr. Cunningham. She and her collaborators are continuing to study comorbid anxiety and pain in youth; current work is using functional magnetic resonance imaging to examine cognitive and affective changes in patients receiving the ADAPT intervention.
The study was funded by the American Pain Society Sharon S. Keller Chronic Pain Research Grant, Cincinnati Children’s Hospital, and the National Institutes of Health. Dr. Cunningham reported no relevant conflicts of interest.
[email protected]
SOURCE: Cunningham N. et al. APS 2019.
REPORTING FROM APS 2019
Flu activity falling but still elevated
Measures of influenza activity fell again as the flu season continues to make its later-than-usual departure this year, according to the Centers for Disease Control and Prevention.
On the geographic front, the map of influenza-like illness (ILI) activity for the week ending March 30 shows that only 6 states are at level 10 on the CDC’s 1-10 scale, compared with 11 for the previous week, and that those same 6 states make up the entire membership of the high range of levels 8-10, which is down from 20 states a week ago, data from the CDC’s Outpatient ILI Surveillance Network show.
The proportion of outpatient visits for ILI, now at 3.2%, dropped for the sixth consecutive week after reaching its season high of 5.1% back in mid-February. The outpatient rate has now been at or above the national baseline of 2.2% for 19 weeks this season, the CDC’s influenza division said April 5, noting that the average for the past five seasons is 16 weeks.
Six flu-related pediatric deaths were reported in the week ending March 30, and the total is now 82 for the 2018-2019 season. Five of the six occurred during previous weeks of this season, and one occurred in the 2017-2018 season, the CDC said.
Measures of influenza activity fell again as the flu season continues to make its later-than-usual departure this year, according to the Centers for Disease Control and Prevention.

On the geographic front, the map of influenza-like illness (ILI) activity for the week ending March 30 shows that only 6 states are at level 10 on the CDC’s 1-10 scale, compared with 11 for the previous week, and that those same 6 states make up the entire membership of the high range of levels 8-10, which is down from 20 states a week ago, data from the CDC’s Outpatient ILI Surveillance Network show.
The proportion of outpatient visits for ILI, now at 3.2%, dropped for the sixth consecutive week after reaching its season high of 5.1% back in mid-February. The outpatient rate has now been at or above the national baseline of 2.2% for 19 weeks this season, the CDC’s influenza division said April 5, noting that the average for the past five seasons is 16 weeks.
Six flu-related pediatric deaths were reported in the week ending March 30, and the total is now 82 for the 2018-2019 season. Five of the six occurred during previous weeks of this season, and one occurred in the 2017-2018 season, the CDC said.
Measures of influenza activity fell again as the flu season continues to make its later-than-usual departure this year, according to the Centers for Disease Control and Prevention.

On the geographic front, the map of influenza-like illness (ILI) activity for the week ending March 30 shows that only 6 states are at level 10 on the CDC’s 1-10 scale, compared with 11 for the previous week, and that those same 6 states make up the entire membership of the high range of levels 8-10, which is down from 20 states a week ago, data from the CDC’s Outpatient ILI Surveillance Network show.
The proportion of outpatient visits for ILI, now at 3.2%, dropped for the sixth consecutive week after reaching its season high of 5.1% back in mid-February. The outpatient rate has now been at or above the national baseline of 2.2% for 19 weeks this season, the CDC’s influenza division said April 5, noting that the average for the past five seasons is 16 weeks.
Six flu-related pediatric deaths were reported in the week ending March 30, and the total is now 82 for the 2018-2019 season. Five of the six occurred during previous weeks of this season, and one occurred in the 2017-2018 season, the CDC said.
Tackling the challenges of pediatric localized scleroderma
MAUI, HAWAII – One of the most important steps to take when a child has received a biopsy-confirmed diagnosis of localized scleroderma is to sit down with the family and explain the rationale for the aggressive therapies to come, Anne M. Stevens, MD, PhD, said at the 2019 Rheumatology Winter Clinical Symposium.
It can be a tough sell at first, especially when a child has only a small red streak on the nose and perhaps a subtle linear lesion on the forehead or scalp. But the family has to come to understand that this is a serious, chronic, progressive fibrotic disease.
“Talk about what a big impact this disease can have on growth of a limb and the normal life of a child because of the cosmetic appearance. Explain that the length of treatment course is based on the long-term outcomes and quality of life. This discussion is usually sufficient” to convince people to give their children “these pretty serious medications,” said Dr. Stevens, professor of pediatrics and head of the division of pediatric rheumatology at the University of Washington, Seattle.
“The treatment goal is to control inflammation and prevent damage in these patients, who we like to catch very early, when it’s a subtle lesion,” she added.
The biggest problem
The biggest contributors to poor quality of life in patients with juvenile localized scleroderma are the extracutaneous manifestations, which occur in up to 50% of cases. Joint pain occurs in roughly 20% of patients, joint contractures due to fibrosis of skin and/or tendons in 30%, and myalgia with or without myositis in 15%. Muscle atrophy due to the deep component of the scleroderma can occur. Moreover, growth problems – especially leg or arm length discrepancies – happen in about 20% of patients in prospective studies. These growth problems may not be obvious until a child enters a growth spurt, at which point there is a limited ability to achieve improvement. That’s why Dr. Stevens recommends that every child with localized scleroderma should get a full joint exam at every visit, with measurement and photos of lesions and recording of all erythematous, violaceous, and waxy-hued areas. And if there are lesions on the head, annual eye exams are warranted.
The prevalence of juvenile localized scleroderma in the United States is about 3 per 100,000, with a mean age of onset of 8.2 years. That makes it 100-fold more common than pediatric systemic sclerosis.
The treatment ladder
There are no Food and Drug Administration–approved medications for localized scleroderma in children. It’s all off label. That being said, there is strong consensus among members of the Childhood Arthritis and Rheumatology Research Alliance that the first-line therapy is methotrexate at 15 mg/m2 or a maximum of 20 mg/week plus intravenous corticosteroids weaned over the course of 3-6 months. This is the treatment regimen with the best supporting evidence of safety and efficacy, including a single Italian randomized, double-blind, placebo-controlled clinical trial (Arthritis Rheum. 2011 Jul;63[7]:1998-2006) and an accompanying long-term, open-label follow-up study (J Am Acad Dermatol. 2012 Dec;67[6]:1151-6).
All of the other treatments she uses for juvenile localized scleroderma – mycophenolate mofetil (CellCept), abatacept (Orencia), tocilizumab (Actemra), and occasionally others – are backed only by a smattering of small case series. However, given the serious potential trajectory of this disease, that modest evidence base has been sufficient for her to receive insurance coverage approval of these agents.
In the randomized trial of first-line methotrexate, 48 of 65 patients treated with methotrexate plus steroid (74%) were responders. And among those 48 responders, 35 (73%) maintained a clinical remission for a mean of 25 months off-drug, while another 13 (27%) were in clinical remission on methotrexate. Twenty-eight patients developed side effects that were generally mild; no one required treatment discontinuation. At the 5-year mark, after an average of an initial 2 years on methotrexate, half of the patients were in a sustained clinical remission, which Dr. Stevens deemed “pretty good” considering the well established and manageable safety profile of the drug.
If a patient fails to respond to methotrexate plus corticosteroids within a few months or later experiences disease progression, Dr. Stevens’ second-line therapy is mycophenolate mofetil in conjunction with corticosteroids. Its use in arresting juvenile localized scleroderma is supported by two favorable published case series, the largest of which includes 10 patients (Rheumatology [Oxford]. 2009 Nov;48[11]:1410-3).
Dr. Stevens’ third-line therapy is intravenous abatacept at 10 mg/kg monthly along with intravenous methylprednisolone at 500 mg/week. There are five published case series, the most recent and largest of which included 13 adult patients, two of whom had en coup de sabre lesions (Acta Derm Venereol. 2018 Apr 16;98[4]:465-6). The biologic also shows promise in patients with advanced severe disease with deep tissue involvement (Semin Arthritis Rheum. 2017 Jun;46[6]:775-81). And abatacept has a plausible mechanism of action in localized scleroderma: French investigators have shown it induces regression of skin fibrosis in a mouse model of the disease (Ann Rheum Dis. 2016 Dec;75[12]:2142-9).
Her fourth-line strategy is the anti-interleukin-6 agent tocilizumab, again in conjunction with corticosteroids. In a translational study, tocilizumab has been shown to normalize dermal fibroblasts and collagen in patients with systemic sclerosis (Ann Rheum Dis. 2018 Sep;77[9]:1362-71). And there have been two promising small retrospective case series as well. A more definitive clinical trial is planned.
Dr. Stevens said that when starting a biologic agent in a child with localized scleroderma, she routinely adds methotrexate until the disease is under control.
Drugs supported by case reports and worth considering on an individual basis as a last resort are hydroxychloroquine, azathioprine, cyclosporine, and imatinib mesylate (Gleevec).
For mild, superficial lesions that don’t cross joints, ultraviolet light A phototherapy is a therapeutic option. It displayed significant benefit in a systematic review and meta-analysis of 19 studies comparing it to methotrexate, although the results with methotrexate were deemed superior (Semin Arthritis Rheum. 2018 Dec;48[3]:495-503).
The pros and cons of getting a baseline brain MRI
Children with localized scleroderma have increased rates of severe headache, peripheral neuropathy, complex partial seizures, and stroke. So it had been Dr. Stevens’ routine practice to obtain an initial brain MRI at the time of diagnosis. Of late, though, she has reconsidered that practice.
“The problem is that some patients with abnormal MRI lesions have no CNS disease at all, and there are also a fair number of patients with a normal MRI who have CNS symptoms. So in our practice we’re pulling back on doing screening MRIs because we don’t know what to do with the findings, and it just makes everybody worried,” she said.
However, if a child with localized scleroderma develops headaches, seizures, neuropathies, or other CNS symptoms, then by all means get an MRI, and if it shows findings such as brain atrophy, white matter lesions, calcifications, or leptomeningeal enhancement, consider treatment, she added.
Dr. Stevens reported receiving research funding from Kineta and Seattle Genetics.
MAUI, HAWAII – One of the most important steps to take when a child has received a biopsy-confirmed diagnosis of localized scleroderma is to sit down with the family and explain the rationale for the aggressive therapies to come, Anne M. Stevens, MD, PhD, said at the 2019 Rheumatology Winter Clinical Symposium.
It can be a tough sell at first, especially when a child has only a small red streak on the nose and perhaps a subtle linear lesion on the forehead or scalp. But the family has to come to understand that this is a serious, chronic, progressive fibrotic disease.
“Talk about what a big impact this disease can have on growth of a limb and the normal life of a child because of the cosmetic appearance. Explain that the length of treatment course is based on the long-term outcomes and quality of life. This discussion is usually sufficient” to convince people to give their children “these pretty serious medications,” said Dr. Stevens, professor of pediatrics and head of the division of pediatric rheumatology at the University of Washington, Seattle.
“The treatment goal is to control inflammation and prevent damage in these patients, who we like to catch very early, when it’s a subtle lesion,” she added.
The biggest problem
The biggest contributors to poor quality of life in patients with juvenile localized scleroderma are the extracutaneous manifestations, which occur in up to 50% of cases. Joint pain occurs in roughly 20% of patients, joint contractures due to fibrosis of skin and/or tendons in 30%, and myalgia with or without myositis in 15%. Muscle atrophy due to the deep component of the scleroderma can occur. Moreover, growth problems – especially leg or arm length discrepancies – happen in about 20% of patients in prospective studies. These growth problems may not be obvious until a child enters a growth spurt, at which point there is a limited ability to achieve improvement. That’s why Dr. Stevens recommends that every child with localized scleroderma should get a full joint exam at every visit, with measurement and photos of lesions and recording of all erythematous, violaceous, and waxy-hued areas. And if there are lesions on the head, annual eye exams are warranted.
The prevalence of juvenile localized scleroderma in the United States is about 3 per 100,000, with a mean age of onset of 8.2 years. That makes it 100-fold more common than pediatric systemic sclerosis.
The treatment ladder
There are no Food and Drug Administration–approved medications for localized scleroderma in children. It’s all off label. That being said, there is strong consensus among members of the Childhood Arthritis and Rheumatology Research Alliance that the first-line therapy is methotrexate at 15 mg/m2 or a maximum of 20 mg/week plus intravenous corticosteroids weaned over the course of 3-6 months. This is the treatment regimen with the best supporting evidence of safety and efficacy, including a single Italian randomized, double-blind, placebo-controlled clinical trial (Arthritis Rheum. 2011 Jul;63[7]:1998-2006) and an accompanying long-term, open-label follow-up study (J Am Acad Dermatol. 2012 Dec;67[6]:1151-6).
All of the other treatments she uses for juvenile localized scleroderma – mycophenolate mofetil (CellCept), abatacept (Orencia), tocilizumab (Actemra), and occasionally others – are backed only by a smattering of small case series. However, given the serious potential trajectory of this disease, that modest evidence base has been sufficient for her to receive insurance coverage approval of these agents.
In the randomized trial of first-line methotrexate, 48 of 65 patients treated with methotrexate plus steroid (74%) were responders. And among those 48 responders, 35 (73%) maintained a clinical remission for a mean of 25 months off-drug, while another 13 (27%) were in clinical remission on methotrexate. Twenty-eight patients developed side effects that were generally mild; no one required treatment discontinuation. At the 5-year mark, after an average of an initial 2 years on methotrexate, half of the patients were in a sustained clinical remission, which Dr. Stevens deemed “pretty good” considering the well established and manageable safety profile of the drug.
If a patient fails to respond to methotrexate plus corticosteroids within a few months or later experiences disease progression, Dr. Stevens’ second-line therapy is mycophenolate mofetil in conjunction with corticosteroids. Its use in arresting juvenile localized scleroderma is supported by two favorable published case series, the largest of which includes 10 patients (Rheumatology [Oxford]. 2009 Nov;48[11]:1410-3).
Dr. Stevens’ third-line therapy is intravenous abatacept at 10 mg/kg monthly along with intravenous methylprednisolone at 500 mg/week. There are five published case series, the most recent and largest of which included 13 adult patients, two of whom had en coup de sabre lesions (Acta Derm Venereol. 2018 Apr 16;98[4]:465-6). The biologic also shows promise in patients with advanced severe disease with deep tissue involvement (Semin Arthritis Rheum. 2017 Jun;46[6]:775-81). And abatacept has a plausible mechanism of action in localized scleroderma: French investigators have shown it induces regression of skin fibrosis in a mouse model of the disease (Ann Rheum Dis. 2016 Dec;75[12]:2142-9).
Her fourth-line strategy is the anti-interleukin-6 agent tocilizumab, again in conjunction with corticosteroids. In a translational study, tocilizumab has been shown to normalize dermal fibroblasts and collagen in patients with systemic sclerosis (Ann Rheum Dis. 2018 Sep;77[9]:1362-71). And there have been two promising small retrospective case series as well. A more definitive clinical trial is planned.
Dr. Stevens said that when starting a biologic agent in a child with localized scleroderma, she routinely adds methotrexate until the disease is under control.
Drugs supported by case reports and worth considering on an individual basis as a last resort are hydroxychloroquine, azathioprine, cyclosporine, and imatinib mesylate (Gleevec).
For mild, superficial lesions that don’t cross joints, ultraviolet light A phototherapy is a therapeutic option. It displayed significant benefit in a systematic review and meta-analysis of 19 studies comparing it to methotrexate, although the results with methotrexate were deemed superior (Semin Arthritis Rheum. 2018 Dec;48[3]:495-503).
The pros and cons of getting a baseline brain MRI
Children with localized scleroderma have increased rates of severe headache, peripheral neuropathy, complex partial seizures, and stroke. So it had been Dr. Stevens’ routine practice to obtain an initial brain MRI at the time of diagnosis. Of late, though, she has reconsidered that practice.
“The problem is that some patients with abnormal MRI lesions have no CNS disease at all, and there are also a fair number of patients with a normal MRI who have CNS symptoms. So in our practice we’re pulling back on doing screening MRIs because we don’t know what to do with the findings, and it just makes everybody worried,” she said.
However, if a child with localized scleroderma develops headaches, seizures, neuropathies, or other CNS symptoms, then by all means get an MRI, and if it shows findings such as brain atrophy, white matter lesions, calcifications, or leptomeningeal enhancement, consider treatment, she added.
Dr. Stevens reported receiving research funding from Kineta and Seattle Genetics.
MAUI, HAWAII – One of the most important steps to take when a child has received a biopsy-confirmed diagnosis of localized scleroderma is to sit down with the family and explain the rationale for the aggressive therapies to come, Anne M. Stevens, MD, PhD, said at the 2019 Rheumatology Winter Clinical Symposium.
It can be a tough sell at first, especially when a child has only a small red streak on the nose and perhaps a subtle linear lesion on the forehead or scalp. But the family has to come to understand that this is a serious, chronic, progressive fibrotic disease.
“Talk about what a big impact this disease can have on growth of a limb and the normal life of a child because of the cosmetic appearance. Explain that the length of treatment course is based on the long-term outcomes and quality of life. This discussion is usually sufficient” to convince people to give their children “these pretty serious medications,” said Dr. Stevens, professor of pediatrics and head of the division of pediatric rheumatology at the University of Washington, Seattle.
“The treatment goal is to control inflammation and prevent damage in these patients, who we like to catch very early, when it’s a subtle lesion,” she added.
The biggest problem
The biggest contributors to poor quality of life in patients with juvenile localized scleroderma are the extracutaneous manifestations, which occur in up to 50% of cases. Joint pain occurs in roughly 20% of patients, joint contractures due to fibrosis of skin and/or tendons in 30%, and myalgia with or without myositis in 15%. Muscle atrophy due to the deep component of the scleroderma can occur. Moreover, growth problems – especially leg or arm length discrepancies – happen in about 20% of patients in prospective studies. These growth problems may not be obvious until a child enters a growth spurt, at which point there is a limited ability to achieve improvement. That’s why Dr. Stevens recommends that every child with localized scleroderma should get a full joint exam at every visit, with measurement and photos of lesions and recording of all erythematous, violaceous, and waxy-hued areas. And if there are lesions on the head, annual eye exams are warranted.
The prevalence of juvenile localized scleroderma in the United States is about 3 per 100,000, with a mean age of onset of 8.2 years. That makes it 100-fold more common than pediatric systemic sclerosis.
The treatment ladder
There are no Food and Drug Administration–approved medications for localized scleroderma in children. It’s all off label. That being said, there is strong consensus among members of the Childhood Arthritis and Rheumatology Research Alliance that the first-line therapy is methotrexate at 15 mg/m2 or a maximum of 20 mg/week plus intravenous corticosteroids weaned over the course of 3-6 months. This is the treatment regimen with the best supporting evidence of safety and efficacy, including a single Italian randomized, double-blind, placebo-controlled clinical trial (Arthritis Rheum. 2011 Jul;63[7]:1998-2006) and an accompanying long-term, open-label follow-up study (J Am Acad Dermatol. 2012 Dec;67[6]:1151-6).
All of the other treatments she uses for juvenile localized scleroderma – mycophenolate mofetil (CellCept), abatacept (Orencia), tocilizumab (Actemra), and occasionally others – are backed only by a smattering of small case series. However, given the serious potential trajectory of this disease, that modest evidence base has been sufficient for her to receive insurance coverage approval of these agents.
In the randomized trial of first-line methotrexate, 48 of 65 patients treated with methotrexate plus steroid (74%) were responders. And among those 48 responders, 35 (73%) maintained a clinical remission for a mean of 25 months off-drug, while another 13 (27%) were in clinical remission on methotrexate. Twenty-eight patients developed side effects that were generally mild; no one required treatment discontinuation. At the 5-year mark, after an average of an initial 2 years on methotrexate, half of the patients were in a sustained clinical remission, which Dr. Stevens deemed “pretty good” considering the well established and manageable safety profile of the drug.
If a patient fails to respond to methotrexate plus corticosteroids within a few months or later experiences disease progression, Dr. Stevens’ second-line therapy is mycophenolate mofetil in conjunction with corticosteroids. Its use in arresting juvenile localized scleroderma is supported by two favorable published case series, the largest of which includes 10 patients (Rheumatology [Oxford]. 2009 Nov;48[11]:1410-3).
Dr. Stevens’ third-line therapy is intravenous abatacept at 10 mg/kg monthly along with intravenous methylprednisolone at 500 mg/week. There are five published case series, the most recent and largest of which included 13 adult patients, two of whom had en coup de sabre lesions (Acta Derm Venereol. 2018 Apr 16;98[4]:465-6). The biologic also shows promise in patients with advanced severe disease with deep tissue involvement (Semin Arthritis Rheum. 2017 Jun;46[6]:775-81). And abatacept has a plausible mechanism of action in localized scleroderma: French investigators have shown it induces regression of skin fibrosis in a mouse model of the disease (Ann Rheum Dis. 2016 Dec;75[12]:2142-9).
Her fourth-line strategy is the anti-interleukin-6 agent tocilizumab, again in conjunction with corticosteroids. In a translational study, tocilizumab has been shown to normalize dermal fibroblasts and collagen in patients with systemic sclerosis (Ann Rheum Dis. 2018 Sep;77[9]:1362-71). And there have been two promising small retrospective case series as well. A more definitive clinical trial is planned.
Dr. Stevens said that when starting a biologic agent in a child with localized scleroderma, she routinely adds methotrexate until the disease is under control.
Drugs supported by case reports and worth considering on an individual basis as a last resort are hydroxychloroquine, azathioprine, cyclosporine, and imatinib mesylate (Gleevec).
For mild, superficial lesions that don’t cross joints, ultraviolet light A phototherapy is a therapeutic option. It displayed significant benefit in a systematic review and meta-analysis of 19 studies comparing it to methotrexate, although the results with methotrexate were deemed superior (Semin Arthritis Rheum. 2018 Dec;48[3]:495-503).
The pros and cons of getting a baseline brain MRI
Children with localized scleroderma have increased rates of severe headache, peripheral neuropathy, complex partial seizures, and stroke. So it had been Dr. Stevens’ routine practice to obtain an initial brain MRI at the time of diagnosis. Of late, though, she has reconsidered that practice.
“The problem is that some patients with abnormal MRI lesions have no CNS disease at all, and there are also a fair number of patients with a normal MRI who have CNS symptoms. So in our practice we’re pulling back on doing screening MRIs because we don’t know what to do with the findings, and it just makes everybody worried,” she said.
However, if a child with localized scleroderma develops headaches, seizures, neuropathies, or other CNS symptoms, then by all means get an MRI, and if it shows findings such as brain atrophy, white matter lesions, calcifications, or leptomeningeal enhancement, consider treatment, she added.
Dr. Stevens reported receiving research funding from Kineta and Seattle Genetics.
REPORTING FROM RWCS 2019
Keeping caries at bay in breastfeeding babies
Early childhood caries (ECCs) are a preventable public health challenge. Breastfeeding may provide early protection from ECCs. In addition, oral hygiene that begins in infancy, regular dental care visits, and a healthy diet can minimize ECC risk.
In this article we review the critical role of the family physician (FP) in reducing ECCs by promoting breastfeeding and infant oral health and addressing dental health concerns.
How ECCs develop
ECCs represent decayed, missing, or filled areas in the primary dentition of the tooth surface. The bacteria that cause them (most often Streptococcus mutans1) strongly adhere to teeth and produce acids as waste products of fermentable carbohydrate metabolism that demineralize tooth enamel and progress into the dentin. Weakened enamel and dentin can result in cavitation (ie, a dental cavity). Left untreated, caries can extend to the pulp and destroy the entire tooth. ECCs are a risk factor not only for dental caries in primary teeth, but in permanent dentition as well.
ECCs are the most common chronic disease affecting young children.1 Dental disease may begin soon after tooth eruption with detrimental effects on oral development. Almost half of children have dental caries by 5 years of age.2
ECCs represent a complex and multifactorial disease that is impacted by biomedical factors and unmet social needs. Children who are most at risk include those with low socioeconomic status, a high-sugar diet, exposure to household smoke, and limited dental care access.3 In addition, women with low education, poor oral health, and/or a lack of fluoride exposure are more likely to have children with ECCs.3 This is partly because of vertical transmission of cariogenic bacteria from caregiver to child. Horizontal transmission in daycare settings can also occur. Paternal and child oral health have not been linked.
Support breastfeeding; keep oral microbiome changes in mind
The American Academy of Pediatrics (AAP) recommends exclusive breastfeeding for the first 6 months of life, a combination of breastfeeding and complementary foods until 12 months of age, and continued breastfeeding for as long as mutually desired by mother and baby.4 The World Health Organization (WHO) recommends continued breastfeeding until 2 years of age or beyond.5 In fact, the WHO global nutrition targets for 2025 include increasing the rate of exclusive breastfeeding in the first 6 months of life to at least 50%.6
In addition to maternal, financial, and societal benefits, human milk offers nutritional and other health-related advantages for children that optimize growth and development into adulthood.4 Breastfed infants may benefit from reduction in infections and diseases, including asthma, diabetes mellitus, childhood cancer, and obesity.7 Improved neurocognitive development, intelligence, and education attainment in adulthood have also been described.8 And the rich microbiome of human milk helps to establish oral and intestinal floras9 and may mediate protection from ECCs.3
Continue to: However, as a child's oral microbiome changes...
However, as a child’s oral microbiome changes with the emergence of primary teeth and exposure to more and varied bacteria and dietary sugars, the natural sugars in human milk may become the substrate for cariogenic bacteria.3 ECCs can develop and progress rapidly. Importantly, both the practice of breastfeeding and ECC risk are modified by socioeconomic status, maternal oral health and education, and exposure to household smoking.3,7 Understanding these relationships may help you better target risk assessment and counseling efforts.
What the research tells us about breastfeeding and ECCs
Breastfeeding is hypothesized to be one of many factors that influence ECC development. However, studies on this association have had conflicting results and have not adequately controlled for major confounders, such as dietary composition, maternal and infant oral hygiene, and maternal oral health status.
So here is what we know.
Breastfeeding during the first year. In one meta-analysis involving children who breastfed for up to 12 months, those who breastfed longer within the 12-month period had a reduced risk of ECCs compared with those who breastfed for a shorter period of time,3 which implies that breast milk may be protective in the first year of life.3
Further, a 2014 study with about 500 participants found that children were more likely to have caries by 5 years of age if they breastfed for <6 months than if they breastfed for at least 6 months.10
Continue to: After the first year
After the first year. A Canadian study found an increased risk of ECCs associated with breastfeeding for longer periods of time. The study of healthy urban children reported that breastfeeding for >24 months was associated with a 2- to 3-fold increased odds of ECCs compared with shorter breastfeeding duration.11
No relationship? Lastly, a US study using National Health and Nutrition Examination Survey data found there was no evidence to suggest that breastfeeding duration was an independent risk factor for ECCs.12
A possible explanation for a link
An initial protective effect of breastfeeding against ECCs may be related to breast milk’s immunomodulatory factors and rich microbiome. Breast milk contains Lactobacilli and substances, including human casein and secretory IgA, that inhibit growth and attachment of bacteria,9 particularly the caries pathogen S mutans. Early defense against ECCs may be mediated through the establishment of a healthy oral and gut microbiome that results from exposure to breastfeeding and contact with skin, gut, and breast milk microbiomes. Later on, the child’s oral microbiome changes with the emergence of teeth and the introduction of complementary foods andother drinks.
A look at the role vitamin D plays
Vitamin D status may influence childhood dental health.13 Low maternal vitamin D levels have been associated with ECCs,14 and mothers with higher prenatal vitamin D intakes were more likely to report that their children were caries-free compared with women who had lower vitamin D intake.15 Additionally, children with severe ECCs were found to have lower vitamin D levels than cavity-free children.16 Unfortunately, only a minority of infants who are predominantly breastfed for > 6 months receive vitamin D supplementation.17
Other factors at work: Carbohydrate exposure, nocturnal feedings
Exposure to carbohydrates—the essential substrate for cariogenic bacteria—is a key factor in ECC development. Refined sugars contribute considerably to tooth decay. Frequency of feeding and feeding practices, such as prolonged nocturnal feeding (either breast or bottle) may increase ECC risk.3 Further, a major determinant of ECC risk is colonization of the infant’s mouth by cariogenic bacteria. Finally, ECC risk depends on socioeconomic status, oral hygiene, exposure to fluoride, and the mother’s oral health, education, and smoking status.3 Even birth order plays a role, with those born first having lower risk than subsequent children.18
Continue to: Breastfeeding and another area of oral health...
Breastfeeding and another area of oral health: Malocclusion
In addition to its relationship with ECCs, breastfeeding promotes adequate development of craniofacial structures (comprising the tongue, facial muscles, and jaw), which are important for smiling, emotion, and social contact. Breastfeeding may prevent the development of malocclusion (ie, a misalignment of the teeth) in primary dentition, which is a risk factor for malocclusion in adulthood.7 Although previous studies had conflicting results, a large prospective study found that breastfeeding significantly reduced the risk of moderate and severe malocclusion; however, this effect was nullified by nonnutritive sucking and pacifier use.19
Oral health recommendations: The FP’s role
ECCs are theoretically preventable. To optimize the benefits of breastfeeding and minimize ECC risk, parents should follow recommendations for their children regarding proper oral hygiene, appropriate fluoride exposure, regular dental visits, and a healthy diet.1
Be sure to advise parents to:
- avoid saliva-sharing behaviors (eg, sharing utensils with their children or cleaning a pacifier with their mouth), as these may increase early colonization of S mutans in infants;
- seek regular preventive dental care and attend to caries—both for their children and themselves; and
- use antimicrobial oral care products including xylitol-containing chewing gum to lower levels of cariogenic microorganisms in themselves and, in turn, reduce mother–child vertical transmission of S mutans.1
In addition, make sure your prenatal counseling includes a discussion of the importance of good maternal oral health and diet—including an adequate vitamin D intake—to prevent ECCs in their children.
It’s never too early to start
Providing guidance on children’s oral health can start with the first well-infant visit. FPs should perform an oral health risk assessment by 6 months of age (see the AAP’s Oral Health Risk Assessment Tool at https://www.aap.org/en-us/Documents/oralhealth_RiskAssessmentTool.pdf) and evaluate fluoride exposure. Advise parents to establish a dental home by the time the child is 12 months of age; to clean their children’s mouths after feedings (before teeth arrive) with a clean, wet, soft washcloth; and to brush their children’s teeth, once they erupt, twice daily using a soft toothbrush (TABLE 120).

Continue to: Talk to parents about...
Talk to parents about how to provide optimal exposure to fluoride, which is known to be safe and effective for the prevention of ECCs.1 Use of fluoridated toothpaste in small amounts provides the benefits of fluoride without increasing the risk of fluorosis, especially for children at risk for caries (see TABLE 221).

The US Preventive Services Task Force recommends that primary care practitioners apply fluoride varnish biannually for at least 2 years to the primary teeth of all children up to 5 years of age (Grade B evidence).22 This is particularly important for high-risk children, such as those with low-income or minority status. However, practitioners should also take into account that high cumulative fluoride intake can lead to dental fluorosis.1 Finally, tell parents to avoid giving their children sugar-containing snacks and drinks to reduce ECC risk.
CORRESPONDENCE
Peter D. Wong, MD, 303-89 Humber College Boulevard, Toronto, Ontario, Canada M9V 4B8; [email protected].
1. American Academy of Pediatric Dentistry. Guideline on infant oral health care. Pediatr Dent. 2012;34:e148-e152.
2. Dye BA, Thornton-Evans G, Li X, et al. Dental caries and sealant prevalence in children and adolescents in the United States, 2011-2012. NCHS Data Brief. No. 191, March 2015. US Department of Health and Human Services, Centers for Disease Control and Prevention, National Center for Health Statistics; 2015. https://www.cdc.gov/nchs/data/databriefs/db191.pdf. Accessed January 25, 2019.
3. Tham R, Bowatte G, Dharmage SC, et al. Breastfeeding and the risk of dental caries: a systematic review and meta-analysis. Acta Paediatr. 2015;104:62-84.
4. Eidelman AI, Schanler RJ, Johnston M, et al. Breastfeeding and the use of human milk (section on breastfeeding). Pediatrics. 2012;129:e827-e841.
5. World Health Organization. 55th World Health Assembly. Agenda Item 13.10: Infant and young child nutrition. https://www.who.int/nutrition/topics/WHA55.25_iycn_en.pdf?ua=1. May 18, 2002. Accessed January 25, 2019.
6. World Health Organization. Global Nutrition Targets 2025 Breastfeeding Policy Brief 5. http://apps.who.int/iris/bitstream/10665/149022/1/WHO_NMH_NHD_14.7_eng.pdf?ua=1. Accessed April 1, 2019.
7. Victora CG, Bahl R, Barros AJ, et al. Breastfeeding in the 21st century: epidemiology, mechanisms, and lifelong effect. Lancet. 2016;387:475-490.
8. Victora CG, Horta BL, de Mola CL, et al. Association between breastfeeding and intelligence, educational attainment, and income at 30 years of age: a prospective birth cohort study from Brazil. Lancet Glob Health. 2015;3:e199-e205.
9. Jain N, Walker WA. Diet and host-microbial crosstalk in postnatal intestinal immune homeostasis. Nat Rev Gastroenterol Hepatol. 2015;12:14-25.
10. Hong L, Levy SM, Warren JJ, et al. Infant breast-feeding and childhood caries: a nine-year study. Pediatr Dent. 2014;36:342-347.
11. Wong PD, Birken CS, Parkin PC, et al. Total breast-feeding duration and dental caries in healthy urban children. Acad Pediatr. 2017;17:310-315.
12. Iida H, Auinger P, Billings RJ, et al. Association between infant breastfeeding and early childhood caries in the United States. Pediatrics. 2007;120:e944-e952.
13. Schroth R, Rabbani R, Loewen G, et al. Vitamin D and dental caries in children. J Dent Res. 2016;95:173-179.
14. Schroth RJ, Lavelle C, Tate R, et al. Prenatal vitamin D and dental caries in infants. Pediatrics. 2014;133:e1277-e1284.
15. Tanaka K, Hitsumoto S, Miyake Y, et al. Higher vitamin D intake during pregnancy is associated with reduced risk of dental caries in young Japanese children. Ann Epidemiol. 2015;25:620-625.
16. Schroth RJ, Halchuk S, Star L. Prevalence and risk factors of caregiver reported severe early childhood caries in Manitoba First Nations children: results from the RHS Phase 2 (2008-2010). Int J Circumpolar Health. 2013;72.
17. Taylor JA, Geyer LJ, Feldman KW. Use of supplemental vitamin D among infants breastfed for prolonged periods. Pediatrics. 2010;125:105-111.
18. Nicolau B, Marcenes W, Bartley M, et al. A life course approach to assessing causes of dental caries experience: the relationship between biological, behavioural, socio-economic and psychological conditions and caries in adolescents. Caries Res. 2003;37:319-326.
19. Peres KG, Cascaes AM, Peres MA, et al. Exclusive breastfeeding and risk of dental malocclusion. Pediatrics. 2015;136:e60-e67.
20. Tinanoff N, Reisin S. Update on early childhood caries since the Surgeon General’s Report. Acad Pediatr. 2009;9:396-403.
21. Canadian Dental Association. CDA Position on Use of Fluorides in Caries Prevention. 2012. https://www.cda-adc.ca/_files/position_statements/fluoride.pdf. Accessed January 25, 2019.
22. US Preventive Services Task Force. USPSTF A and B Recommendations. https://www.uspreventiveservicestaskforce.org/Page/Name/uspstf-a-and-b-recommendations/. Accessed April 1, 2019.
Early childhood caries (ECCs) are a preventable public health challenge. Breastfeeding may provide early protection from ECCs. In addition, oral hygiene that begins in infancy, regular dental care visits, and a healthy diet can minimize ECC risk.
In this article we review the critical role of the family physician (FP) in reducing ECCs by promoting breastfeeding and infant oral health and addressing dental health concerns.
How ECCs develop
ECCs represent decayed, missing, or filled areas in the primary dentition of the tooth surface. The bacteria that cause them (most often Streptococcus mutans1) strongly adhere to teeth and produce acids as waste products of fermentable carbohydrate metabolism that demineralize tooth enamel and progress into the dentin. Weakened enamel and dentin can result in cavitation (ie, a dental cavity). Left untreated, caries can extend to the pulp and destroy the entire tooth. ECCs are a risk factor not only for dental caries in primary teeth, but in permanent dentition as well.
ECCs are the most common chronic disease affecting young children.1 Dental disease may begin soon after tooth eruption with detrimental effects on oral development. Almost half of children have dental caries by 5 years of age.2
ECCs represent a complex and multifactorial disease that is impacted by biomedical factors and unmet social needs. Children who are most at risk include those with low socioeconomic status, a high-sugar diet, exposure to household smoke, and limited dental care access.3 In addition, women with low education, poor oral health, and/or a lack of fluoride exposure are more likely to have children with ECCs.3 This is partly because of vertical transmission of cariogenic bacteria from caregiver to child. Horizontal transmission in daycare settings can also occur. Paternal and child oral health have not been linked.
Support breastfeeding; keep oral microbiome changes in mind
The American Academy of Pediatrics (AAP) recommends exclusive breastfeeding for the first 6 months of life, a combination of breastfeeding and complementary foods until 12 months of age, and continued breastfeeding for as long as mutually desired by mother and baby.4 The World Health Organization (WHO) recommends continued breastfeeding until 2 years of age or beyond.5 In fact, the WHO global nutrition targets for 2025 include increasing the rate of exclusive breastfeeding in the first 6 months of life to at least 50%.6
In addition to maternal, financial, and societal benefits, human milk offers nutritional and other health-related advantages for children that optimize growth and development into adulthood.4 Breastfed infants may benefit from reduction in infections and diseases, including asthma, diabetes mellitus, childhood cancer, and obesity.7 Improved neurocognitive development, intelligence, and education attainment in adulthood have also been described.8 And the rich microbiome of human milk helps to establish oral and intestinal floras9 and may mediate protection from ECCs.3
Continue to: However, as a child's oral microbiome changes...
However, as a child’s oral microbiome changes with the emergence of primary teeth and exposure to more and varied bacteria and dietary sugars, the natural sugars in human milk may become the substrate for cariogenic bacteria.3 ECCs can develop and progress rapidly. Importantly, both the practice of breastfeeding and ECC risk are modified by socioeconomic status, maternal oral health and education, and exposure to household smoking.3,7 Understanding these relationships may help you better target risk assessment and counseling efforts.
What the research tells us about breastfeeding and ECCs
Breastfeeding is hypothesized to be one of many factors that influence ECC development. However, studies on this association have had conflicting results and have not adequately controlled for major confounders, such as dietary composition, maternal and infant oral hygiene, and maternal oral health status.
So here is what we know.
Breastfeeding during the first year. In one meta-analysis involving children who breastfed for up to 12 months, those who breastfed longer within the 12-month period had a reduced risk of ECCs compared with those who breastfed for a shorter period of time,3 which implies that breast milk may be protective in the first year of life.3
Further, a 2014 study with about 500 participants found that children were more likely to have caries by 5 years of age if they breastfed for <6 months than if they breastfed for at least 6 months.10
Continue to: After the first year
After the first year. A Canadian study found an increased risk of ECCs associated with breastfeeding for longer periods of time. The study of healthy urban children reported that breastfeeding for >24 months was associated with a 2- to 3-fold increased odds of ECCs compared with shorter breastfeeding duration.11
No relationship? Lastly, a US study using National Health and Nutrition Examination Survey data found there was no evidence to suggest that breastfeeding duration was an independent risk factor for ECCs.12
A possible explanation for a link
An initial protective effect of breastfeeding against ECCs may be related to breast milk’s immunomodulatory factors and rich microbiome. Breast milk contains Lactobacilli and substances, including human casein and secretory IgA, that inhibit growth and attachment of bacteria,9 particularly the caries pathogen S mutans. Early defense against ECCs may be mediated through the establishment of a healthy oral and gut microbiome that results from exposure to breastfeeding and contact with skin, gut, and breast milk microbiomes. Later on, the child’s oral microbiome changes with the emergence of teeth and the introduction of complementary foods andother drinks.
A look at the role vitamin D plays
Vitamin D status may influence childhood dental health.13 Low maternal vitamin D levels have been associated with ECCs,14 and mothers with higher prenatal vitamin D intakes were more likely to report that their children were caries-free compared with women who had lower vitamin D intake.15 Additionally, children with severe ECCs were found to have lower vitamin D levels than cavity-free children.16 Unfortunately, only a minority of infants who are predominantly breastfed for > 6 months receive vitamin D supplementation.17
Other factors at work: Carbohydrate exposure, nocturnal feedings
Exposure to carbohydrates—the essential substrate for cariogenic bacteria—is a key factor in ECC development. Refined sugars contribute considerably to tooth decay. Frequency of feeding and feeding practices, such as prolonged nocturnal feeding (either breast or bottle) may increase ECC risk.3 Further, a major determinant of ECC risk is colonization of the infant’s mouth by cariogenic bacteria. Finally, ECC risk depends on socioeconomic status, oral hygiene, exposure to fluoride, and the mother’s oral health, education, and smoking status.3 Even birth order plays a role, with those born first having lower risk than subsequent children.18
Continue to: Breastfeeding and another area of oral health...
Breastfeeding and another area of oral health: Malocclusion
In addition to its relationship with ECCs, breastfeeding promotes adequate development of craniofacial structures (comprising the tongue, facial muscles, and jaw), which are important for smiling, emotion, and social contact. Breastfeeding may prevent the development of malocclusion (ie, a misalignment of the teeth) in primary dentition, which is a risk factor for malocclusion in adulthood.7 Although previous studies had conflicting results, a large prospective study found that breastfeeding significantly reduced the risk of moderate and severe malocclusion; however, this effect was nullified by nonnutritive sucking and pacifier use.19
Oral health recommendations: The FP’s role
ECCs are theoretically preventable. To optimize the benefits of breastfeeding and minimize ECC risk, parents should follow recommendations for their children regarding proper oral hygiene, appropriate fluoride exposure, regular dental visits, and a healthy diet.1
Be sure to advise parents to:
- avoid saliva-sharing behaviors (eg, sharing utensils with their children or cleaning a pacifier with their mouth), as these may increase early colonization of S mutans in infants;
- seek regular preventive dental care and attend to caries—both for their children and themselves; and
- use antimicrobial oral care products including xylitol-containing chewing gum to lower levels of cariogenic microorganisms in themselves and, in turn, reduce mother–child vertical transmission of S mutans.1
In addition, make sure your prenatal counseling includes a discussion of the importance of good maternal oral health and diet—including an adequate vitamin D intake—to prevent ECCs in their children.
It’s never too early to start
Providing guidance on children’s oral health can start with the first well-infant visit. FPs should perform an oral health risk assessment by 6 months of age (see the AAP’s Oral Health Risk Assessment Tool at https://www.aap.org/en-us/Documents/oralhealth_RiskAssessmentTool.pdf) and evaluate fluoride exposure. Advise parents to establish a dental home by the time the child is 12 months of age; to clean their children’s mouths after feedings (before teeth arrive) with a clean, wet, soft washcloth; and to brush their children’s teeth, once they erupt, twice daily using a soft toothbrush (TABLE 120).

Continue to: Talk to parents about...
Talk to parents about how to provide optimal exposure to fluoride, which is known to be safe and effective for the prevention of ECCs.1 Use of fluoridated toothpaste in small amounts provides the benefits of fluoride without increasing the risk of fluorosis, especially for children at risk for caries (see TABLE 221).

The US Preventive Services Task Force recommends that primary care practitioners apply fluoride varnish biannually for at least 2 years to the primary teeth of all children up to 5 years of age (Grade B evidence).22 This is particularly important for high-risk children, such as those with low-income or minority status. However, practitioners should also take into account that high cumulative fluoride intake can lead to dental fluorosis.1 Finally, tell parents to avoid giving their children sugar-containing snacks and drinks to reduce ECC risk.
CORRESPONDENCE
Peter D. Wong, MD, 303-89 Humber College Boulevard, Toronto, Ontario, Canada M9V 4B8; [email protected].
Early childhood caries (ECCs) are a preventable public health challenge. Breastfeeding may provide early protection from ECCs. In addition, oral hygiene that begins in infancy, regular dental care visits, and a healthy diet can minimize ECC risk.
In this article we review the critical role of the family physician (FP) in reducing ECCs by promoting breastfeeding and infant oral health and addressing dental health concerns.
How ECCs develop
ECCs represent decayed, missing, or filled areas in the primary dentition of the tooth surface. The bacteria that cause them (most often Streptococcus mutans1) strongly adhere to teeth and produce acids as waste products of fermentable carbohydrate metabolism that demineralize tooth enamel and progress into the dentin. Weakened enamel and dentin can result in cavitation (ie, a dental cavity). Left untreated, caries can extend to the pulp and destroy the entire tooth. ECCs are a risk factor not only for dental caries in primary teeth, but in permanent dentition as well.
ECCs are the most common chronic disease affecting young children.1 Dental disease may begin soon after tooth eruption with detrimental effects on oral development. Almost half of children have dental caries by 5 years of age.2
ECCs represent a complex and multifactorial disease that is impacted by biomedical factors and unmet social needs. Children who are most at risk include those with low socioeconomic status, a high-sugar diet, exposure to household smoke, and limited dental care access.3 In addition, women with low education, poor oral health, and/or a lack of fluoride exposure are more likely to have children with ECCs.3 This is partly because of vertical transmission of cariogenic bacteria from caregiver to child. Horizontal transmission in daycare settings can also occur. Paternal and child oral health have not been linked.
Support breastfeeding; keep oral microbiome changes in mind
The American Academy of Pediatrics (AAP) recommends exclusive breastfeeding for the first 6 months of life, a combination of breastfeeding and complementary foods until 12 months of age, and continued breastfeeding for as long as mutually desired by mother and baby.4 The World Health Organization (WHO) recommends continued breastfeeding until 2 years of age or beyond.5 In fact, the WHO global nutrition targets for 2025 include increasing the rate of exclusive breastfeeding in the first 6 months of life to at least 50%.6
In addition to maternal, financial, and societal benefits, human milk offers nutritional and other health-related advantages for children that optimize growth and development into adulthood.4 Breastfed infants may benefit from reduction in infections and diseases, including asthma, diabetes mellitus, childhood cancer, and obesity.7 Improved neurocognitive development, intelligence, and education attainment in adulthood have also been described.8 And the rich microbiome of human milk helps to establish oral and intestinal floras9 and may mediate protection from ECCs.3
Continue to: However, as a child's oral microbiome changes...
However, as a child’s oral microbiome changes with the emergence of primary teeth and exposure to more and varied bacteria and dietary sugars, the natural sugars in human milk may become the substrate for cariogenic bacteria.3 ECCs can develop and progress rapidly. Importantly, both the practice of breastfeeding and ECC risk are modified by socioeconomic status, maternal oral health and education, and exposure to household smoking.3,7 Understanding these relationships may help you better target risk assessment and counseling efforts.
What the research tells us about breastfeeding and ECCs
Breastfeeding is hypothesized to be one of many factors that influence ECC development. However, studies on this association have had conflicting results and have not adequately controlled for major confounders, such as dietary composition, maternal and infant oral hygiene, and maternal oral health status.
So here is what we know.
Breastfeeding during the first year. In one meta-analysis involving children who breastfed for up to 12 months, those who breastfed longer within the 12-month period had a reduced risk of ECCs compared with those who breastfed for a shorter period of time,3 which implies that breast milk may be protective in the first year of life.3
Further, a 2014 study with about 500 participants found that children were more likely to have caries by 5 years of age if they breastfed for <6 months than if they breastfed for at least 6 months.10
Continue to: After the first year
After the first year. A Canadian study found an increased risk of ECCs associated with breastfeeding for longer periods of time. The study of healthy urban children reported that breastfeeding for >24 months was associated with a 2- to 3-fold increased odds of ECCs compared with shorter breastfeeding duration.11
No relationship? Lastly, a US study using National Health and Nutrition Examination Survey data found there was no evidence to suggest that breastfeeding duration was an independent risk factor for ECCs.12
A possible explanation for a link
An initial protective effect of breastfeeding against ECCs may be related to breast milk’s immunomodulatory factors and rich microbiome. Breast milk contains Lactobacilli and substances, including human casein and secretory IgA, that inhibit growth and attachment of bacteria,9 particularly the caries pathogen S mutans. Early defense against ECCs may be mediated through the establishment of a healthy oral and gut microbiome that results from exposure to breastfeeding and contact with skin, gut, and breast milk microbiomes. Later on, the child’s oral microbiome changes with the emergence of teeth and the introduction of complementary foods andother drinks.
A look at the role vitamin D plays
Vitamin D status may influence childhood dental health.13 Low maternal vitamin D levels have been associated with ECCs,14 and mothers with higher prenatal vitamin D intakes were more likely to report that their children were caries-free compared with women who had lower vitamin D intake.15 Additionally, children with severe ECCs were found to have lower vitamin D levels than cavity-free children.16 Unfortunately, only a minority of infants who are predominantly breastfed for > 6 months receive vitamin D supplementation.17
Other factors at work: Carbohydrate exposure, nocturnal feedings
Exposure to carbohydrates—the essential substrate for cariogenic bacteria—is a key factor in ECC development. Refined sugars contribute considerably to tooth decay. Frequency of feeding and feeding practices, such as prolonged nocturnal feeding (either breast or bottle) may increase ECC risk.3 Further, a major determinant of ECC risk is colonization of the infant’s mouth by cariogenic bacteria. Finally, ECC risk depends on socioeconomic status, oral hygiene, exposure to fluoride, and the mother’s oral health, education, and smoking status.3 Even birth order plays a role, with those born first having lower risk than subsequent children.18
Continue to: Breastfeeding and another area of oral health...
Breastfeeding and another area of oral health: Malocclusion
In addition to its relationship with ECCs, breastfeeding promotes adequate development of craniofacial structures (comprising the tongue, facial muscles, and jaw), which are important for smiling, emotion, and social contact. Breastfeeding may prevent the development of malocclusion (ie, a misalignment of the teeth) in primary dentition, which is a risk factor for malocclusion in adulthood.7 Although previous studies had conflicting results, a large prospective study found that breastfeeding significantly reduced the risk of moderate and severe malocclusion; however, this effect was nullified by nonnutritive sucking and pacifier use.19
Oral health recommendations: The FP’s role
ECCs are theoretically preventable. To optimize the benefits of breastfeeding and minimize ECC risk, parents should follow recommendations for their children regarding proper oral hygiene, appropriate fluoride exposure, regular dental visits, and a healthy diet.1
Be sure to advise parents to:
- avoid saliva-sharing behaviors (eg, sharing utensils with their children or cleaning a pacifier with their mouth), as these may increase early colonization of S mutans in infants;
- seek regular preventive dental care and attend to caries—both for their children and themselves; and
- use antimicrobial oral care products including xylitol-containing chewing gum to lower levels of cariogenic microorganisms in themselves and, in turn, reduce mother–child vertical transmission of S mutans.1
In addition, make sure your prenatal counseling includes a discussion of the importance of good maternal oral health and diet—including an adequate vitamin D intake—to prevent ECCs in their children.
It’s never too early to start
Providing guidance on children’s oral health can start with the first well-infant visit. FPs should perform an oral health risk assessment by 6 months of age (see the AAP’s Oral Health Risk Assessment Tool at https://www.aap.org/en-us/Documents/oralhealth_RiskAssessmentTool.pdf) and evaluate fluoride exposure. Advise parents to establish a dental home by the time the child is 12 months of age; to clean their children’s mouths after feedings (before teeth arrive) with a clean, wet, soft washcloth; and to brush their children’s teeth, once they erupt, twice daily using a soft toothbrush (TABLE 120).

Continue to: Talk to parents about...
Talk to parents about how to provide optimal exposure to fluoride, which is known to be safe and effective for the prevention of ECCs.1 Use of fluoridated toothpaste in small amounts provides the benefits of fluoride without increasing the risk of fluorosis, especially for children at risk for caries (see TABLE 221).

The US Preventive Services Task Force recommends that primary care practitioners apply fluoride varnish biannually for at least 2 years to the primary teeth of all children up to 5 years of age (Grade B evidence).22 This is particularly important for high-risk children, such as those with low-income or minority status. However, practitioners should also take into account that high cumulative fluoride intake can lead to dental fluorosis.1 Finally, tell parents to avoid giving their children sugar-containing snacks and drinks to reduce ECC risk.
CORRESPONDENCE
Peter D. Wong, MD, 303-89 Humber College Boulevard, Toronto, Ontario, Canada M9V 4B8; [email protected].
1. American Academy of Pediatric Dentistry. Guideline on infant oral health care. Pediatr Dent. 2012;34:e148-e152.
2. Dye BA, Thornton-Evans G, Li X, et al. Dental caries and sealant prevalence in children and adolescents in the United States, 2011-2012. NCHS Data Brief. No. 191, March 2015. US Department of Health and Human Services, Centers for Disease Control and Prevention, National Center for Health Statistics; 2015. https://www.cdc.gov/nchs/data/databriefs/db191.pdf. Accessed January 25, 2019.
3. Tham R, Bowatte G, Dharmage SC, et al. Breastfeeding and the risk of dental caries: a systematic review and meta-analysis. Acta Paediatr. 2015;104:62-84.
4. Eidelman AI, Schanler RJ, Johnston M, et al. Breastfeeding and the use of human milk (section on breastfeeding). Pediatrics. 2012;129:e827-e841.
5. World Health Organization. 55th World Health Assembly. Agenda Item 13.10: Infant and young child nutrition. https://www.who.int/nutrition/topics/WHA55.25_iycn_en.pdf?ua=1. May 18, 2002. Accessed January 25, 2019.
6. World Health Organization. Global Nutrition Targets 2025 Breastfeeding Policy Brief 5. http://apps.who.int/iris/bitstream/10665/149022/1/WHO_NMH_NHD_14.7_eng.pdf?ua=1. Accessed April 1, 2019.
7. Victora CG, Bahl R, Barros AJ, et al. Breastfeeding in the 21st century: epidemiology, mechanisms, and lifelong effect. Lancet. 2016;387:475-490.
8. Victora CG, Horta BL, de Mola CL, et al. Association between breastfeeding and intelligence, educational attainment, and income at 30 years of age: a prospective birth cohort study from Brazil. Lancet Glob Health. 2015;3:e199-e205.
9. Jain N, Walker WA. Diet and host-microbial crosstalk in postnatal intestinal immune homeostasis. Nat Rev Gastroenterol Hepatol. 2015;12:14-25.
10. Hong L, Levy SM, Warren JJ, et al. Infant breast-feeding and childhood caries: a nine-year study. Pediatr Dent. 2014;36:342-347.
11. Wong PD, Birken CS, Parkin PC, et al. Total breast-feeding duration and dental caries in healthy urban children. Acad Pediatr. 2017;17:310-315.
12. Iida H, Auinger P, Billings RJ, et al. Association between infant breastfeeding and early childhood caries in the United States. Pediatrics. 2007;120:e944-e952.
13. Schroth R, Rabbani R, Loewen G, et al. Vitamin D and dental caries in children. J Dent Res. 2016;95:173-179.
14. Schroth RJ, Lavelle C, Tate R, et al. Prenatal vitamin D and dental caries in infants. Pediatrics. 2014;133:e1277-e1284.
15. Tanaka K, Hitsumoto S, Miyake Y, et al. Higher vitamin D intake during pregnancy is associated with reduced risk of dental caries in young Japanese children. Ann Epidemiol. 2015;25:620-625.
16. Schroth RJ, Halchuk S, Star L. Prevalence and risk factors of caregiver reported severe early childhood caries in Manitoba First Nations children: results from the RHS Phase 2 (2008-2010). Int J Circumpolar Health. 2013;72.
17. Taylor JA, Geyer LJ, Feldman KW. Use of supplemental vitamin D among infants breastfed for prolonged periods. Pediatrics. 2010;125:105-111.
18. Nicolau B, Marcenes W, Bartley M, et al. A life course approach to assessing causes of dental caries experience: the relationship between biological, behavioural, socio-economic and psychological conditions and caries in adolescents. Caries Res. 2003;37:319-326.
19. Peres KG, Cascaes AM, Peres MA, et al. Exclusive breastfeeding and risk of dental malocclusion. Pediatrics. 2015;136:e60-e67.
20. Tinanoff N, Reisin S. Update on early childhood caries since the Surgeon General’s Report. Acad Pediatr. 2009;9:396-403.
21. Canadian Dental Association. CDA Position on Use of Fluorides in Caries Prevention. 2012. https://www.cda-adc.ca/_files/position_statements/fluoride.pdf. Accessed January 25, 2019.
22. US Preventive Services Task Force. USPSTF A and B Recommendations. https://www.uspreventiveservicestaskforce.org/Page/Name/uspstf-a-and-b-recommendations/. Accessed April 1, 2019.
1. American Academy of Pediatric Dentistry. Guideline on infant oral health care. Pediatr Dent. 2012;34:e148-e152.
2. Dye BA, Thornton-Evans G, Li X, et al. Dental caries and sealant prevalence in children and adolescents in the United States, 2011-2012. NCHS Data Brief. No. 191, March 2015. US Department of Health and Human Services, Centers for Disease Control and Prevention, National Center for Health Statistics; 2015. https://www.cdc.gov/nchs/data/databriefs/db191.pdf. Accessed January 25, 2019.
3. Tham R, Bowatte G, Dharmage SC, et al. Breastfeeding and the risk of dental caries: a systematic review and meta-analysis. Acta Paediatr. 2015;104:62-84.
4. Eidelman AI, Schanler RJ, Johnston M, et al. Breastfeeding and the use of human milk (section on breastfeeding). Pediatrics. 2012;129:e827-e841.
5. World Health Organization. 55th World Health Assembly. Agenda Item 13.10: Infant and young child nutrition. https://www.who.int/nutrition/topics/WHA55.25_iycn_en.pdf?ua=1. May 18, 2002. Accessed January 25, 2019.
6. World Health Organization. Global Nutrition Targets 2025 Breastfeeding Policy Brief 5. http://apps.who.int/iris/bitstream/10665/149022/1/WHO_NMH_NHD_14.7_eng.pdf?ua=1. Accessed April 1, 2019.
7. Victora CG, Bahl R, Barros AJ, et al. Breastfeeding in the 21st century: epidemiology, mechanisms, and lifelong effect. Lancet. 2016;387:475-490.
8. Victora CG, Horta BL, de Mola CL, et al. Association between breastfeeding and intelligence, educational attainment, and income at 30 years of age: a prospective birth cohort study from Brazil. Lancet Glob Health. 2015;3:e199-e205.
9. Jain N, Walker WA. Diet and host-microbial crosstalk in postnatal intestinal immune homeostasis. Nat Rev Gastroenterol Hepatol. 2015;12:14-25.
10. Hong L, Levy SM, Warren JJ, et al. Infant breast-feeding and childhood caries: a nine-year study. Pediatr Dent. 2014;36:342-347.
11. Wong PD, Birken CS, Parkin PC, et al. Total breast-feeding duration and dental caries in healthy urban children. Acad Pediatr. 2017;17:310-315.
12. Iida H, Auinger P, Billings RJ, et al. Association between infant breastfeeding and early childhood caries in the United States. Pediatrics. 2007;120:e944-e952.
13. Schroth R, Rabbani R, Loewen G, et al. Vitamin D and dental caries in children. J Dent Res. 2016;95:173-179.
14. Schroth RJ, Lavelle C, Tate R, et al. Prenatal vitamin D and dental caries in infants. Pediatrics. 2014;133:e1277-e1284.
15. Tanaka K, Hitsumoto S, Miyake Y, et al. Higher vitamin D intake during pregnancy is associated with reduced risk of dental caries in young Japanese children. Ann Epidemiol. 2015;25:620-625.
16. Schroth RJ, Halchuk S, Star L. Prevalence and risk factors of caregiver reported severe early childhood caries in Manitoba First Nations children: results from the RHS Phase 2 (2008-2010). Int J Circumpolar Health. 2013;72.
17. Taylor JA, Geyer LJ, Feldman KW. Use of supplemental vitamin D among infants breastfed for prolonged periods. Pediatrics. 2010;125:105-111.
18. Nicolau B, Marcenes W, Bartley M, et al. A life course approach to assessing causes of dental caries experience: the relationship between biological, behavioural, socio-economic and psychological conditions and caries in adolescents. Caries Res. 2003;37:319-326.
19. Peres KG, Cascaes AM, Peres MA, et al. Exclusive breastfeeding and risk of dental malocclusion. Pediatrics. 2015;136:e60-e67.
20. Tinanoff N, Reisin S. Update on early childhood caries since the Surgeon General’s Report. Acad Pediatr. 2009;9:396-403.
21. Canadian Dental Association. CDA Position on Use of Fluorides in Caries Prevention. 2012. https://www.cda-adc.ca/_files/position_statements/fluoride.pdf. Accessed January 25, 2019.
22. US Preventive Services Task Force. USPSTF A and B Recommendations. https://www.uspreventiveservicestaskforce.org/Page/Name/uspstf-a-and-b-recommendations/. Accessed April 1, 2019.
PRACTICE RECOMMENDATIONS
› Promote breastfeeding as the preferred method of feeding infants. A
› Optimize pediatric oral health by reducing risk factors for dental disease and by providing parents with anticipatory guidance to prevent early childhood caries. B
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series