User login
Cannabis use in pregnancy and lactation: A changing landscape
National survey data from 2007-2012 of more than 93,000 pregnant women suggest that around 7% of pregnant respondents reported any cannabis use in the last 2-12 months; of those, 16% reported daily or almost daily use. Among pregnant past-year users in the same survey, 70% perceived slight or no risk of harm from cannabis use 1-2 times a week in pregnancy.1
Data from the Kaiser Northern California health plan involving more than 279,000 pregnancies followed during 2009-2016 suggest that there has been a significant upward trend in use of cannabis during pregnancy, from 4% to 7%, as reported by the mother and/or identified by routine urine screening. The highest prevalence in that study was seen among 18- to 24-year-old pregnant women, increasing from 13% to 22% over the 7-year study period. Importantly, more than 50% of cannabis users in the sample were identified by toxicology screening alone.2,3 Common reasons given for use of cannabis in pregnancy include anxiety, pain, and nausea and vomiting of pregnancy.4
With respect to adverse perinatal outcomes, several case-control studies have examined risks for major birth defects with maternal self-report of cannabis use. Some have noted very modest increased risks for selected major birth defects (odds ratios less than 2); however, data still are very limited.5,6
A number of prospective studies have addressed risks of preterm birth and growth restriction, accounting for mother’s concomitant tobacco use.7-11 Some of these studies have suggested about a twofold to threefold increased risk for preterm delivery and an increased risk for reduced birth weight – particularly with heavier or regular cannabis use – but study findings have not been entirely consistent.
Given its psychoactive properties, there has been high interest in understanding whether there are any short- or long-term neurodevelopmental effects on children prenatally exposed to cannabis. These outcomes have been studied in two small older cohorts in the United States and Canada and one more recent cohort in the Netherlands.12-15 Deficits in several measures of cognition and behavior were noted in follow-up of those children from birth to adulthood. However, it is unclear to what extent these findings may have been influenced by heredity, environment, or other factors.
There have been limitations in almost all studies published to date, including small sample sizes, no biomarker validation of maternal report of dose and gestational timing of cannabis use, and lack of detailed data on common coexposures, such as alcohol, tobacco, and other drugs. In addition, newer studies of pregnancy outcomes in women who use currently available cannabis products are needed, given the substantial increase in the potency of cannabis used today, compared with that of 20 years ago. For example, the tetrahydrocannabinol (THC) concentration in commonly cultivated marijuana plants has increased threefold from 4% to 12% between 1995 and 2014.16
There are very limited data on the presence of cannabis in breast milk and the potential effects of exposure to THC and other metabolites for breastfed infants. However, two recent studies have demonstrated there are low but measurable levels of some cannabis metabolites in breast milk.17-18 Further work is needed to determine if these metabolites accumulate in milk and if at a given dose and age of the breastfed infant, there are any growth, neurodevelopmental, or other clinically important adverse effects.
Related questions, such as potential differences in the effects of exposure during pregnancy or lactation based on the route of administration (edible vs. inhaled) and the use of cannabidiol (CBD) products, have not been studied.
At the present time, the American College of Obstetricians and Gynecologists recommends that women who are pregnant or contemplating pregnancy be encouraged to discontinue marijuana use. With respect to lactation and breastfeeding, ACOG concludes there are insufficient data to evaluate the effects on infants, and in the absence of such data, marijuana use is discouraged. Similarly, the American Academy of Pediatrics recommends women of childbearing age abstain from marijuana use while pregnant or breastfeeding because of potential adverse consequences to the fetus, infant, or child.
In August 2019, the U.S. Surgeon General issued an advisory regarding potential harm to developing brains from the use of marijuana during pregnancy and lactation. The Food and Drug Administration issued a similar statement in October 2019 strongly advising against the use of CBD, THC, and marijuana in any form during pregnancy or while breastfeeding.
Dr. Chambers is professor of pediatrics and director of clinical research at Rady Children’s Hospital and associate director of the Clinical and Translational Research Institute at the University of California, San Diego. She is also director of MotherToBaby California, president of the Organization of Teratology Information Specialists, and past president of the Teratology Society.
References
1. Am J Obstet Gynecol. 2015 Aug;213(2):201.e1-10.
2. JAMA. 2017 Dec 26;318(24):2490-1.
3. JAMA. 2017 Jan 10;317(2):207-9.
4. Complement Ther Clin Pract. 2009 Nov;15(4)242-6.
5. Paediatr Perinat Epidemiol. 2014 Sep; 28(5): 424-33.
6. J Toxicol Environ Health A. 2007 Jan;70(1):7-18.
7. Am J Obstet Gynecol. 1983 Aug 15;146(8):992-4.
8. Clin Perinatol. 1991 Mar;18(1):77-91.
9. Am J Epidemiol. 1986 Dec;124(6):986-93.
10. Pediatr Res. 2012 Feb;71(2):215-9.
11. Reprod Toxicol. 2016;62:77-86.
12. Neurotoxicol Teratol. 1987 Jan-Feb;9(1):1-7.
13. Neurotoxicol Teratol. 1994 Mar-Apr;16(2):169-75.
14. Biol Psychiatry. 2016 Jun 15;79(12):971-9.
15. Pharmacol Ther. 2018 Feb;182:133-51.
16. Biol Psychiatry. 2016 Apr 1;79(7):613-9.
17. Obstet Gynecol. 2018 May;131(5):783-8.
18. Pediatrics. 2018 Sep;142(3):e20181076.
National survey data from 2007-2012 of more than 93,000 pregnant women suggest that around 7% of pregnant respondents reported any cannabis use in the last 2-12 months; of those, 16% reported daily or almost daily use. Among pregnant past-year users in the same survey, 70% perceived slight or no risk of harm from cannabis use 1-2 times a week in pregnancy.1
Data from the Kaiser Northern California health plan involving more than 279,000 pregnancies followed during 2009-2016 suggest that there has been a significant upward trend in use of cannabis during pregnancy, from 4% to 7%, as reported by the mother and/or identified by routine urine screening. The highest prevalence in that study was seen among 18- to 24-year-old pregnant women, increasing from 13% to 22% over the 7-year study period. Importantly, more than 50% of cannabis users in the sample were identified by toxicology screening alone.2,3 Common reasons given for use of cannabis in pregnancy include anxiety, pain, and nausea and vomiting of pregnancy.4
With respect to adverse perinatal outcomes, several case-control studies have examined risks for major birth defects with maternal self-report of cannabis use. Some have noted very modest increased risks for selected major birth defects (odds ratios less than 2); however, data still are very limited.5,6
A number of prospective studies have addressed risks of preterm birth and growth restriction, accounting for mother’s concomitant tobacco use.7-11 Some of these studies have suggested about a twofold to threefold increased risk for preterm delivery and an increased risk for reduced birth weight – particularly with heavier or regular cannabis use – but study findings have not been entirely consistent.
Given its psychoactive properties, there has been high interest in understanding whether there are any short- or long-term neurodevelopmental effects on children prenatally exposed to cannabis. These outcomes have been studied in two small older cohorts in the United States and Canada and one more recent cohort in the Netherlands.12-15 Deficits in several measures of cognition and behavior were noted in follow-up of those children from birth to adulthood. However, it is unclear to what extent these findings may have been influenced by heredity, environment, or other factors.
There have been limitations in almost all studies published to date, including small sample sizes, no biomarker validation of maternal report of dose and gestational timing of cannabis use, and lack of detailed data on common coexposures, such as alcohol, tobacco, and other drugs. In addition, newer studies of pregnancy outcomes in women who use currently available cannabis products are needed, given the substantial increase in the potency of cannabis used today, compared with that of 20 years ago. For example, the tetrahydrocannabinol (THC) concentration in commonly cultivated marijuana plants has increased threefold from 4% to 12% between 1995 and 2014.16
There are very limited data on the presence of cannabis in breast milk and the potential effects of exposure to THC and other metabolites for breastfed infants. However, two recent studies have demonstrated there are low but measurable levels of some cannabis metabolites in breast milk.17-18 Further work is needed to determine if these metabolites accumulate in milk and if at a given dose and age of the breastfed infant, there are any growth, neurodevelopmental, or other clinically important adverse effects.
Related questions, such as potential differences in the effects of exposure during pregnancy or lactation based on the route of administration (edible vs. inhaled) and the use of cannabidiol (CBD) products, have not been studied.
At the present time, the American College of Obstetricians and Gynecologists recommends that women who are pregnant or contemplating pregnancy be encouraged to discontinue marijuana use. With respect to lactation and breastfeeding, ACOG concludes there are insufficient data to evaluate the effects on infants, and in the absence of such data, marijuana use is discouraged. Similarly, the American Academy of Pediatrics recommends women of childbearing age abstain from marijuana use while pregnant or breastfeeding because of potential adverse consequences to the fetus, infant, or child.
In August 2019, the U.S. Surgeon General issued an advisory regarding potential harm to developing brains from the use of marijuana during pregnancy and lactation. The Food and Drug Administration issued a similar statement in October 2019 strongly advising against the use of CBD, THC, and marijuana in any form during pregnancy or while breastfeeding.
Dr. Chambers is professor of pediatrics and director of clinical research at Rady Children’s Hospital and associate director of the Clinical and Translational Research Institute at the University of California, San Diego. She is also director of MotherToBaby California, president of the Organization of Teratology Information Specialists, and past president of the Teratology Society.
References
1. Am J Obstet Gynecol. 2015 Aug;213(2):201.e1-10.
2. JAMA. 2017 Dec 26;318(24):2490-1.
3. JAMA. 2017 Jan 10;317(2):207-9.
4. Complement Ther Clin Pract. 2009 Nov;15(4)242-6.
5. Paediatr Perinat Epidemiol. 2014 Sep; 28(5): 424-33.
6. J Toxicol Environ Health A. 2007 Jan;70(1):7-18.
7. Am J Obstet Gynecol. 1983 Aug 15;146(8):992-4.
8. Clin Perinatol. 1991 Mar;18(1):77-91.
9. Am J Epidemiol. 1986 Dec;124(6):986-93.
10. Pediatr Res. 2012 Feb;71(2):215-9.
11. Reprod Toxicol. 2016;62:77-86.
12. Neurotoxicol Teratol. 1987 Jan-Feb;9(1):1-7.
13. Neurotoxicol Teratol. 1994 Mar-Apr;16(2):169-75.
14. Biol Psychiatry. 2016 Jun 15;79(12):971-9.
15. Pharmacol Ther. 2018 Feb;182:133-51.
16. Biol Psychiatry. 2016 Apr 1;79(7):613-9.
17. Obstet Gynecol. 2018 May;131(5):783-8.
18. Pediatrics. 2018 Sep;142(3):e20181076.
National survey data from 2007-2012 of more than 93,000 pregnant women suggest that around 7% of pregnant respondents reported any cannabis use in the last 2-12 months; of those, 16% reported daily or almost daily use. Among pregnant past-year users in the same survey, 70% perceived slight or no risk of harm from cannabis use 1-2 times a week in pregnancy.1
Data from the Kaiser Northern California health plan involving more than 279,000 pregnancies followed during 2009-2016 suggest that there has been a significant upward trend in use of cannabis during pregnancy, from 4% to 7%, as reported by the mother and/or identified by routine urine screening. The highest prevalence in that study was seen among 18- to 24-year-old pregnant women, increasing from 13% to 22% over the 7-year study period. Importantly, more than 50% of cannabis users in the sample were identified by toxicology screening alone.2,3 Common reasons given for use of cannabis in pregnancy include anxiety, pain, and nausea and vomiting of pregnancy.4
With respect to adverse perinatal outcomes, several case-control studies have examined risks for major birth defects with maternal self-report of cannabis use. Some have noted very modest increased risks for selected major birth defects (odds ratios less than 2); however, data still are very limited.5,6
A number of prospective studies have addressed risks of preterm birth and growth restriction, accounting for mother’s concomitant tobacco use.7-11 Some of these studies have suggested about a twofold to threefold increased risk for preterm delivery and an increased risk for reduced birth weight – particularly with heavier or regular cannabis use – but study findings have not been entirely consistent.
Given its psychoactive properties, there has been high interest in understanding whether there are any short- or long-term neurodevelopmental effects on children prenatally exposed to cannabis. These outcomes have been studied in two small older cohorts in the United States and Canada and one more recent cohort in the Netherlands.12-15 Deficits in several measures of cognition and behavior were noted in follow-up of those children from birth to adulthood. However, it is unclear to what extent these findings may have been influenced by heredity, environment, or other factors.
There have been limitations in almost all studies published to date, including small sample sizes, no biomarker validation of maternal report of dose and gestational timing of cannabis use, and lack of detailed data on common coexposures, such as alcohol, tobacco, and other drugs. In addition, newer studies of pregnancy outcomes in women who use currently available cannabis products are needed, given the substantial increase in the potency of cannabis used today, compared with that of 20 years ago. For example, the tetrahydrocannabinol (THC) concentration in commonly cultivated marijuana plants has increased threefold from 4% to 12% between 1995 and 2014.16
There are very limited data on the presence of cannabis in breast milk and the potential effects of exposure to THC and other metabolites for breastfed infants. However, two recent studies have demonstrated there are low but measurable levels of some cannabis metabolites in breast milk.17-18 Further work is needed to determine if these metabolites accumulate in milk and if at a given dose and age of the breastfed infant, there are any growth, neurodevelopmental, or other clinically important adverse effects.
Related questions, such as potential differences in the effects of exposure during pregnancy or lactation based on the route of administration (edible vs. inhaled) and the use of cannabidiol (CBD) products, have not been studied.
At the present time, the American College of Obstetricians and Gynecologists recommends that women who are pregnant or contemplating pregnancy be encouraged to discontinue marijuana use. With respect to lactation and breastfeeding, ACOG concludes there are insufficient data to evaluate the effects on infants, and in the absence of such data, marijuana use is discouraged. Similarly, the American Academy of Pediatrics recommends women of childbearing age abstain from marijuana use while pregnant or breastfeeding because of potential adverse consequences to the fetus, infant, or child.
In August 2019, the U.S. Surgeon General issued an advisory regarding potential harm to developing brains from the use of marijuana during pregnancy and lactation. The Food and Drug Administration issued a similar statement in October 2019 strongly advising against the use of CBD, THC, and marijuana in any form during pregnancy or while breastfeeding.
Dr. Chambers is professor of pediatrics and director of clinical research at Rady Children’s Hospital and associate director of the Clinical and Translational Research Institute at the University of California, San Diego. She is also director of MotherToBaby California, president of the Organization of Teratology Information Specialists, and past president of the Teratology Society.
References
1. Am J Obstet Gynecol. 2015 Aug;213(2):201.e1-10.
2. JAMA. 2017 Dec 26;318(24):2490-1.
3. JAMA. 2017 Jan 10;317(2):207-9.
4. Complement Ther Clin Pract. 2009 Nov;15(4)242-6.
5. Paediatr Perinat Epidemiol. 2014 Sep; 28(5): 424-33.
6. J Toxicol Environ Health A. 2007 Jan;70(1):7-18.
7. Am J Obstet Gynecol. 1983 Aug 15;146(8):992-4.
8. Clin Perinatol. 1991 Mar;18(1):77-91.
9. Am J Epidemiol. 1986 Dec;124(6):986-93.
10. Pediatr Res. 2012 Feb;71(2):215-9.
11. Reprod Toxicol. 2016;62:77-86.
12. Neurotoxicol Teratol. 1987 Jan-Feb;9(1):1-7.
13. Neurotoxicol Teratol. 1994 Mar-Apr;16(2):169-75.
14. Biol Psychiatry. 2016 Jun 15;79(12):971-9.
15. Pharmacol Ther. 2018 Feb;182:133-51.
16. Biol Psychiatry. 2016 Apr 1;79(7):613-9.
17. Obstet Gynecol. 2018 May;131(5):783-8.
18. Pediatrics. 2018 Sep;142(3):e20181076.
Suicide rate higher than average for female clinicians
The suicide rate for women who provide health care is higher than that of all women of working age, while male health care practitioners are less likely to end their lives than working-age men as a whole, according to the Centers for Disease Control and Prevention.
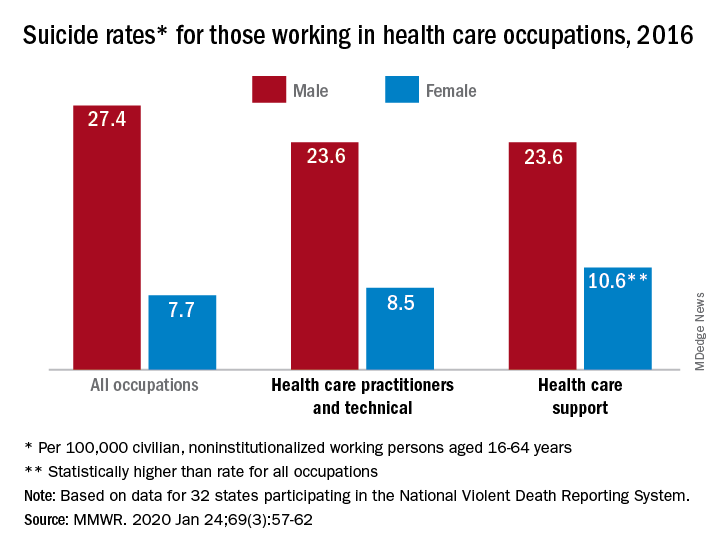
In 2016, the suicide rate for women classified as “healthcare practitioners and technical” – a category that includes physicians and surgeons, as well as chiropractors, physician assistants, and nurse practitioners – was 8.5 per 100,000 population, compared with 7.7 per 100,000 for all working women aged 16-64 years. That difference, however, was not statistically significant, Cora Peterson, PhD, and associates at the CDC said in the Morbidity and Mortality Weekly Report.
For females classified as “healthcare support” – medical assistants and transcriptionists, phlebotomists, and pharmacy aides – the suicide rate of 10.6 per 100,000 was significantly higher than that of all working women, the investigators noted.
The suicide rate for males in each of the two occupation categories was 23.6 per 100,000 population in 2016, lower than the rate of 27.4 per 100,000 for males of all occupations, they said, based on data from 32 states that participated in the 2016 National Violent Death Reporting System.
For males, the highest suicide rates in occupations meeting criteria for sample size were “construction and extraction” (49.4 per 100,000); “installation, maintenance, and repair” (36.9); and “arts, design, entertainment, sports, and media” (32.0). Among females, the highest rates were seen in “construction and extraction” (25.5 per 100,000), “protective service” (14.0), and “transportation and material moving” (12.5), with healthcare support next, Dr. Peterson and associates reported.
“Although relative comparisons of suicide rates in this manner are useful for prevention purposes, Therefore, all industry sectors and occupational groups can contribute to reducing suicide incidence,” they wrote.
SOURCE: Peterson C et al. MMWR. 2020 Jan 24;69(3):57-62.
The suicide rate for women who provide health care is higher than that of all women of working age, while male health care practitioners are less likely to end their lives than working-age men as a whole, according to the Centers for Disease Control and Prevention.

In 2016, the suicide rate for women classified as “healthcare practitioners and technical” – a category that includes physicians and surgeons, as well as chiropractors, physician assistants, and nurse practitioners – was 8.5 per 100,000 population, compared with 7.7 per 100,000 for all working women aged 16-64 years. That difference, however, was not statistically significant, Cora Peterson, PhD, and associates at the CDC said in the Morbidity and Mortality Weekly Report.
For females classified as “healthcare support” – medical assistants and transcriptionists, phlebotomists, and pharmacy aides – the suicide rate of 10.6 per 100,000 was significantly higher than that of all working women, the investigators noted.
The suicide rate for males in each of the two occupation categories was 23.6 per 100,000 population in 2016, lower than the rate of 27.4 per 100,000 for males of all occupations, they said, based on data from 32 states that participated in the 2016 National Violent Death Reporting System.
For males, the highest suicide rates in occupations meeting criteria for sample size were “construction and extraction” (49.4 per 100,000); “installation, maintenance, and repair” (36.9); and “arts, design, entertainment, sports, and media” (32.0). Among females, the highest rates were seen in “construction and extraction” (25.5 per 100,000), “protective service” (14.0), and “transportation and material moving” (12.5), with healthcare support next, Dr. Peterson and associates reported.
“Although relative comparisons of suicide rates in this manner are useful for prevention purposes, Therefore, all industry sectors and occupational groups can contribute to reducing suicide incidence,” they wrote.
SOURCE: Peterson C et al. MMWR. 2020 Jan 24;69(3):57-62.
The suicide rate for women who provide health care is higher than that of all women of working age, while male health care practitioners are less likely to end their lives than working-age men as a whole, according to the Centers for Disease Control and Prevention.

In 2016, the suicide rate for women classified as “healthcare practitioners and technical” – a category that includes physicians and surgeons, as well as chiropractors, physician assistants, and nurse practitioners – was 8.5 per 100,000 population, compared with 7.7 per 100,000 for all working women aged 16-64 years. That difference, however, was not statistically significant, Cora Peterson, PhD, and associates at the CDC said in the Morbidity and Mortality Weekly Report.
For females classified as “healthcare support” – medical assistants and transcriptionists, phlebotomists, and pharmacy aides – the suicide rate of 10.6 per 100,000 was significantly higher than that of all working women, the investigators noted.
The suicide rate for males in each of the two occupation categories was 23.6 per 100,000 population in 2016, lower than the rate of 27.4 per 100,000 for males of all occupations, they said, based on data from 32 states that participated in the 2016 National Violent Death Reporting System.
For males, the highest suicide rates in occupations meeting criteria for sample size were “construction and extraction” (49.4 per 100,000); “installation, maintenance, and repair” (36.9); and “arts, design, entertainment, sports, and media” (32.0). Among females, the highest rates were seen in “construction and extraction” (25.5 per 100,000), “protective service” (14.0), and “transportation and material moving” (12.5), with healthcare support next, Dr. Peterson and associates reported.
“Although relative comparisons of suicide rates in this manner are useful for prevention purposes, Therefore, all industry sectors and occupational groups can contribute to reducing suicide incidence,” they wrote.
SOURCE: Peterson C et al. MMWR. 2020 Jan 24;69(3):57-62.
FROM MMWR
Start of myeloma therapy may be delayed for women, minorities
Women and racial minorities with multiple myeloma may be at increased risk of delayed treatment, a situation that should be addressed urgently, according to authors of a recent analysis of a clinical oncology database.
By contrast, patients receiving myeloma treatment sooner after diagnosis included patients who were over 80 years of age, had multiple comorbidities, were treated at specialized cancer programs or in areas other than the Northeast, and had Medicaid or did not have private insurance, the authors reported.
Contrary to what was expected, levels of education and income did not significantly affect the timeliness of treatment in this analysis by Vivek Kumar, MD, of Dana-Farber Cancer Institute in Boston and coinvestigators.
While results of studies to date are “conflicting” as to whether timeliness of myeloma therapy will affect patient outcomes, recent studies in breast cancer and other tumor types suggest earlier treatment intervention may reduce morbidity, improve quality of life, and possibly prolong survival, according to Dr. Kumar and colleagues.
Moreover, the focus of myeloma treatment has shifted toward earlier treatment in light of the superiority of today’s treatment options, which was demonstrated in the 2014 update of the International Myeloma Working Group (IMWG) diagnostic criteria, according to the investigators.
“The definition of active MM [multiple myeloma] has been updated so that patients who may have been considered to have smoldering MM previously are now treated sooner to prevent end-organ damage whenever possible,” said Dr. Kumar and coauthors in their report in JCO Oncology Practice.
The analysis of timely myeloma treatment was based on for 74,722 patients in the National Cancer Database who received a diagnosis of multiple myeloma between 2004 and 2015 and went on to receive systemic treatment within the first year of diagnosis.
Delay in treatment, defined as receiving antimyeloma therapy 40 or more days after diagnosis, occurred in 18,375 of those patients, or about one-quarter of the study cohort. The mean time from diagnosis to start of treatment in that group was 63 days.
Compared with patients who received treatment within 7 days of diagnosis, patients with delays in treatment were more likely to be women (odds ratio, 1.15; 95% confidence interval, 1.1-1.2) and more likely to be non-Hispanic black (OR, 1.21; 95% CI, 1.14-1.28), the investigators reported.
A previous analysis of the SEER-Medicare database suggested that certain antimyeloma agents are used later in racial and ethnic minorities, including Hispanic patients, who had the highest median time to first dose of bortezomib, Dr. Kumar and colleagues noted.
However, no report before the present one had looked at the time to overall initial treatment in racial and ethnic minorities, they added.
Patients diagnosed in more recent years had higher odds of treatment delay, though this could have been caused by an increase in the number of patients diagnosed early; prior to the 2014 IMWG diagnostic criteria revision, many would have been offered therapy only when signs of end-organ damage were present, while patients without end-organ damage would have been said to have smoldering disease, authors said.
Patients 80 years of age and older and those with a higher Charlson comorbidity score had a lower likelihood of treatment delay in this analysis, possibly reflecting the frailty of those patients and an urgent need for treatment, according to investigators.
Uninsured patients and those with Medicaid were less likely than insured patients to experience treatment delay, according to the report.
“This may be associated with the fact that, for these insurances, prior authorization is typically not required before initiating treatment,” said Dr. Kumar and colleagues. “However, this could also depend on several other possible factors, including availability of caregiver support and seeking medical care later.”
Dr. Kumar reported no conflicts of interest related to the analysis. Coauthors reported disclosures with Takeda, Guardant Health, and other pharmaceutical companies.
SOURCE: Kumar V et al. JCO Oncology Practice. 2020 Jan 21. doi: 10.1200/JOP.19.00309.
Women and racial minorities with multiple myeloma may be at increased risk of delayed treatment, a situation that should be addressed urgently, according to authors of a recent analysis of a clinical oncology database.
By contrast, patients receiving myeloma treatment sooner after diagnosis included patients who were over 80 years of age, had multiple comorbidities, were treated at specialized cancer programs or in areas other than the Northeast, and had Medicaid or did not have private insurance, the authors reported.
Contrary to what was expected, levels of education and income did not significantly affect the timeliness of treatment in this analysis by Vivek Kumar, MD, of Dana-Farber Cancer Institute in Boston and coinvestigators.
While results of studies to date are “conflicting” as to whether timeliness of myeloma therapy will affect patient outcomes, recent studies in breast cancer and other tumor types suggest earlier treatment intervention may reduce morbidity, improve quality of life, and possibly prolong survival, according to Dr. Kumar and colleagues.
Moreover, the focus of myeloma treatment has shifted toward earlier treatment in light of the superiority of today’s treatment options, which was demonstrated in the 2014 update of the International Myeloma Working Group (IMWG) diagnostic criteria, according to the investigators.
“The definition of active MM [multiple myeloma] has been updated so that patients who may have been considered to have smoldering MM previously are now treated sooner to prevent end-organ damage whenever possible,” said Dr. Kumar and coauthors in their report in JCO Oncology Practice.
The analysis of timely myeloma treatment was based on for 74,722 patients in the National Cancer Database who received a diagnosis of multiple myeloma between 2004 and 2015 and went on to receive systemic treatment within the first year of diagnosis.
Delay in treatment, defined as receiving antimyeloma therapy 40 or more days after diagnosis, occurred in 18,375 of those patients, or about one-quarter of the study cohort. The mean time from diagnosis to start of treatment in that group was 63 days.
Compared with patients who received treatment within 7 days of diagnosis, patients with delays in treatment were more likely to be women (odds ratio, 1.15; 95% confidence interval, 1.1-1.2) and more likely to be non-Hispanic black (OR, 1.21; 95% CI, 1.14-1.28), the investigators reported.
A previous analysis of the SEER-Medicare database suggested that certain antimyeloma agents are used later in racial and ethnic minorities, including Hispanic patients, who had the highest median time to first dose of bortezomib, Dr. Kumar and colleagues noted.
However, no report before the present one had looked at the time to overall initial treatment in racial and ethnic minorities, they added.
Patients diagnosed in more recent years had higher odds of treatment delay, though this could have been caused by an increase in the number of patients diagnosed early; prior to the 2014 IMWG diagnostic criteria revision, many would have been offered therapy only when signs of end-organ damage were present, while patients without end-organ damage would have been said to have smoldering disease, authors said.
Patients 80 years of age and older and those with a higher Charlson comorbidity score had a lower likelihood of treatment delay in this analysis, possibly reflecting the frailty of those patients and an urgent need for treatment, according to investigators.
Uninsured patients and those with Medicaid were less likely than insured patients to experience treatment delay, according to the report.
“This may be associated with the fact that, for these insurances, prior authorization is typically not required before initiating treatment,” said Dr. Kumar and colleagues. “However, this could also depend on several other possible factors, including availability of caregiver support and seeking medical care later.”
Dr. Kumar reported no conflicts of interest related to the analysis. Coauthors reported disclosures with Takeda, Guardant Health, and other pharmaceutical companies.
SOURCE: Kumar V et al. JCO Oncology Practice. 2020 Jan 21. doi: 10.1200/JOP.19.00309.
Women and racial minorities with multiple myeloma may be at increased risk of delayed treatment, a situation that should be addressed urgently, according to authors of a recent analysis of a clinical oncology database.
By contrast, patients receiving myeloma treatment sooner after diagnosis included patients who were over 80 years of age, had multiple comorbidities, were treated at specialized cancer programs or in areas other than the Northeast, and had Medicaid or did not have private insurance, the authors reported.
Contrary to what was expected, levels of education and income did not significantly affect the timeliness of treatment in this analysis by Vivek Kumar, MD, of Dana-Farber Cancer Institute in Boston and coinvestigators.
While results of studies to date are “conflicting” as to whether timeliness of myeloma therapy will affect patient outcomes, recent studies in breast cancer and other tumor types suggest earlier treatment intervention may reduce morbidity, improve quality of life, and possibly prolong survival, according to Dr. Kumar and colleagues.
Moreover, the focus of myeloma treatment has shifted toward earlier treatment in light of the superiority of today’s treatment options, which was demonstrated in the 2014 update of the International Myeloma Working Group (IMWG) diagnostic criteria, according to the investigators.
“The definition of active MM [multiple myeloma] has been updated so that patients who may have been considered to have smoldering MM previously are now treated sooner to prevent end-organ damage whenever possible,” said Dr. Kumar and coauthors in their report in JCO Oncology Practice.
The analysis of timely myeloma treatment was based on for 74,722 patients in the National Cancer Database who received a diagnosis of multiple myeloma between 2004 and 2015 and went on to receive systemic treatment within the first year of diagnosis.
Delay in treatment, defined as receiving antimyeloma therapy 40 or more days after diagnosis, occurred in 18,375 of those patients, or about one-quarter of the study cohort. The mean time from diagnosis to start of treatment in that group was 63 days.
Compared with patients who received treatment within 7 days of diagnosis, patients with delays in treatment were more likely to be women (odds ratio, 1.15; 95% confidence interval, 1.1-1.2) and more likely to be non-Hispanic black (OR, 1.21; 95% CI, 1.14-1.28), the investigators reported.
A previous analysis of the SEER-Medicare database suggested that certain antimyeloma agents are used later in racial and ethnic minorities, including Hispanic patients, who had the highest median time to first dose of bortezomib, Dr. Kumar and colleagues noted.
However, no report before the present one had looked at the time to overall initial treatment in racial and ethnic minorities, they added.
Patients diagnosed in more recent years had higher odds of treatment delay, though this could have been caused by an increase in the number of patients diagnosed early; prior to the 2014 IMWG diagnostic criteria revision, many would have been offered therapy only when signs of end-organ damage were present, while patients without end-organ damage would have been said to have smoldering disease, authors said.
Patients 80 years of age and older and those with a higher Charlson comorbidity score had a lower likelihood of treatment delay in this analysis, possibly reflecting the frailty of those patients and an urgent need for treatment, according to investigators.
Uninsured patients and those with Medicaid were less likely than insured patients to experience treatment delay, according to the report.
“This may be associated with the fact that, for these insurances, prior authorization is typically not required before initiating treatment,” said Dr. Kumar and colleagues. “However, this could also depend on several other possible factors, including availability of caregiver support and seeking medical care later.”
Dr. Kumar reported no conflicts of interest related to the analysis. Coauthors reported disclosures with Takeda, Guardant Health, and other pharmaceutical companies.
SOURCE: Kumar V et al. JCO Oncology Practice. 2020 Jan 21. doi: 10.1200/JOP.19.00309.
FROM JCO ONCOLOGY PRACTICE
Key clinical point:
Major finding: Patients with delays in treatment were more likely to be women (odds ratio, 1.15) and more likely to be non-Hispanic blacks (OR, 1.21).
Study details: Retrospective analysis of 74,722 patients in the National Cancer Database diagnosed with multiple myeloma between 2004 and 2015.
Disclosures: Dr. Kumar reported no conflicts of interest related to the analysis. Coauthors reported disclosures with Takeda, Guardant Health, and other pharmaceutical companies.
Source: Kumar V et al. JCO Oncology Practice. 2020 Jan 21. doi: 10.1200/JOP.19.00309.
Methotrexate gives durable remission from idiopathic granulomatous mastitis
ATLANTA – Methotrexate, in combination with prednisone, might be emerging as the go-to option for idiopathic granulomatous mastitis, according to investigators from Oregon Health & Science University, Portland.
Idiopathic granulomatous mastitis (IGM) is an inflammatory disease in which granulomas form in breast tissue. It strikes mostly young to middle-aged women with painful, firm breast masses, sometimes with redness and drainage. Diagnosis is by biopsy with rule-out of known causes.
IGM does not respond to antibiotics. Prednisone and surgery have been the traditional approaches, but masses can recur after surgery, and a year or more of prednisone, with the weight gain and side effects, is problematic. As a result, cases are increasingly being referred to rheumatologists for other options, said lead investigator Sarah Ringsted, MD, a rheumatology fellow at the university.
A study she presented at the annual meeting of the American College of Rheumatology and previous work from others builds a case for methotrexate, which often seems to put the disease in remission and allows for shorter glucocorticoid courses. These days, “I offer this to patients as a great option. It’s really nice to have, instead of having women go on months and months of high-dose steroids, and I think we can save patients from unnecessary” surgery, Dr. Ringsted said.
Her usual regimen these days is methotrexate 15-20 mg/week for 12-18 months, with high-dose prednisone (greater than 20 mg/day) for the first 3 months, followed by a taper.
Dr. Ringsted and associates compared 23 women treated at the university during 2007-2018. Just 5 of the 12 women (42%) treated with high-dose prednisone alone went into remission and did not relapse over a mean follow-up of 27 months. Two out of three women who had both high-dose glucocorticoids and surgery achieved remission without relapse, as did all three women who received methotrexate and high-dose glucocorticoids (one also had surgery). Five other patients were treated with other options; just two had a durable remission.
The numbers are small, but they add to two previous reports. Among 19 women who had failed other treatments, 94% improved and 75% went into remission with 15 months of methotrexate in a review from Stanford (Calif.) University. An Iranian study of 17 patients treated with methotrexate, and also glucocorticoids in some, had a relapse rate of only 17.8%.
There were several cases of both inflammatory arthritis and erythema nodosum in the Oregon series, a higher incidence than what has been reported before for IGM. “It’s interesting because it makes me think of sarcoidosis. There have been cases of sarcoidosis causing mastitis, but mostly in patients with other features” of the disease. “It makes me wonder if any of these women will develop sarcoidosis later on; I think that’s an interesting question,” Dr. Ringsted said.
Women in the study were an average age of 32 years, and over half were Hispanic, which is associated with a higher risk for IGM. Almost all the women had been pregnant before and had breast fed in the previous 5 years. Cancer, tuberculosis, and fungal infections were among the things ruled out before mastitis was deemed idiopathic.
Women with IGM tend to be of childbearing age, and must be cautioned against the teratogenic effects of methotrexate, Dr. Ringsted noted.
There was no external funding, and the investigators didn’t report any disclosures.
SOURCE: Ringsted S et al. Arthritis Rheumatol. 2019;71(suppl 10), Abstract 386.
ATLANTA – Methotrexate, in combination with prednisone, might be emerging as the go-to option for idiopathic granulomatous mastitis, according to investigators from Oregon Health & Science University, Portland.
Idiopathic granulomatous mastitis (IGM) is an inflammatory disease in which granulomas form in breast tissue. It strikes mostly young to middle-aged women with painful, firm breast masses, sometimes with redness and drainage. Diagnosis is by biopsy with rule-out of known causes.
IGM does not respond to antibiotics. Prednisone and surgery have been the traditional approaches, but masses can recur after surgery, and a year or more of prednisone, with the weight gain and side effects, is problematic. As a result, cases are increasingly being referred to rheumatologists for other options, said lead investigator Sarah Ringsted, MD, a rheumatology fellow at the university.
A study she presented at the annual meeting of the American College of Rheumatology and previous work from others builds a case for methotrexate, which often seems to put the disease in remission and allows for shorter glucocorticoid courses. These days, “I offer this to patients as a great option. It’s really nice to have, instead of having women go on months and months of high-dose steroids, and I think we can save patients from unnecessary” surgery, Dr. Ringsted said.
Her usual regimen these days is methotrexate 15-20 mg/week for 12-18 months, with high-dose prednisone (greater than 20 mg/day) for the first 3 months, followed by a taper.
Dr. Ringsted and associates compared 23 women treated at the university during 2007-2018. Just 5 of the 12 women (42%) treated with high-dose prednisone alone went into remission and did not relapse over a mean follow-up of 27 months. Two out of three women who had both high-dose glucocorticoids and surgery achieved remission without relapse, as did all three women who received methotrexate and high-dose glucocorticoids (one also had surgery). Five other patients were treated with other options; just two had a durable remission.
The numbers are small, but they add to two previous reports. Among 19 women who had failed other treatments, 94% improved and 75% went into remission with 15 months of methotrexate in a review from Stanford (Calif.) University. An Iranian study of 17 patients treated with methotrexate, and also glucocorticoids in some, had a relapse rate of only 17.8%.
There were several cases of both inflammatory arthritis and erythema nodosum in the Oregon series, a higher incidence than what has been reported before for IGM. “It’s interesting because it makes me think of sarcoidosis. There have been cases of sarcoidosis causing mastitis, but mostly in patients with other features” of the disease. “It makes me wonder if any of these women will develop sarcoidosis later on; I think that’s an interesting question,” Dr. Ringsted said.
Women in the study were an average age of 32 years, and over half were Hispanic, which is associated with a higher risk for IGM. Almost all the women had been pregnant before and had breast fed in the previous 5 years. Cancer, tuberculosis, and fungal infections were among the things ruled out before mastitis was deemed idiopathic.
Women with IGM tend to be of childbearing age, and must be cautioned against the teratogenic effects of methotrexate, Dr. Ringsted noted.
There was no external funding, and the investigators didn’t report any disclosures.
SOURCE: Ringsted S et al. Arthritis Rheumatol. 2019;71(suppl 10), Abstract 386.
ATLANTA – Methotrexate, in combination with prednisone, might be emerging as the go-to option for idiopathic granulomatous mastitis, according to investigators from Oregon Health & Science University, Portland.
Idiopathic granulomatous mastitis (IGM) is an inflammatory disease in which granulomas form in breast tissue. It strikes mostly young to middle-aged women with painful, firm breast masses, sometimes with redness and drainage. Diagnosis is by biopsy with rule-out of known causes.
IGM does not respond to antibiotics. Prednisone and surgery have been the traditional approaches, but masses can recur after surgery, and a year or more of prednisone, with the weight gain and side effects, is problematic. As a result, cases are increasingly being referred to rheumatologists for other options, said lead investigator Sarah Ringsted, MD, a rheumatology fellow at the university.
A study she presented at the annual meeting of the American College of Rheumatology and previous work from others builds a case for methotrexate, which often seems to put the disease in remission and allows for shorter glucocorticoid courses. These days, “I offer this to patients as a great option. It’s really nice to have, instead of having women go on months and months of high-dose steroids, and I think we can save patients from unnecessary” surgery, Dr. Ringsted said.
Her usual regimen these days is methotrexate 15-20 mg/week for 12-18 months, with high-dose prednisone (greater than 20 mg/day) for the first 3 months, followed by a taper.
Dr. Ringsted and associates compared 23 women treated at the university during 2007-2018. Just 5 of the 12 women (42%) treated with high-dose prednisone alone went into remission and did not relapse over a mean follow-up of 27 months. Two out of three women who had both high-dose glucocorticoids and surgery achieved remission without relapse, as did all three women who received methotrexate and high-dose glucocorticoids (one also had surgery). Five other patients were treated with other options; just two had a durable remission.
The numbers are small, but they add to two previous reports. Among 19 women who had failed other treatments, 94% improved and 75% went into remission with 15 months of methotrexate in a review from Stanford (Calif.) University. An Iranian study of 17 patients treated with methotrexate, and also glucocorticoids in some, had a relapse rate of only 17.8%.
There were several cases of both inflammatory arthritis and erythema nodosum in the Oregon series, a higher incidence than what has been reported before for IGM. “It’s interesting because it makes me think of sarcoidosis. There have been cases of sarcoidosis causing mastitis, but mostly in patients with other features” of the disease. “It makes me wonder if any of these women will develop sarcoidosis later on; I think that’s an interesting question,” Dr. Ringsted said.
Women in the study were an average age of 32 years, and over half were Hispanic, which is associated with a higher risk for IGM. Almost all the women had been pregnant before and had breast fed in the previous 5 years. Cancer, tuberculosis, and fungal infections were among the things ruled out before mastitis was deemed idiopathic.
Women with IGM tend to be of childbearing age, and must be cautioned against the teratogenic effects of methotrexate, Dr. Ringsted noted.
There was no external funding, and the investigators didn’t report any disclosures.
SOURCE: Ringsted S et al. Arthritis Rheumatol. 2019;71(suppl 10), Abstract 386.
REPORTING FROM ACR 2019
Unnecessary pelvic exams, Pap tests common in young women
according to estimates from a study published online in JAMA Internal Medicine.
Approximately 2.6 million young women – about a quarter of those in this age group – reported receiving a pelvic exam in the previous year even though fewer than 10% were pregnant or receiving treatment for a sexually transmitted infection (STI) at the time.
Similarly, an estimated three in four Pap tests given to women aged 15-20 years likely were unnecessary. Based on Medicare payments for screening Pap tests and pelvic exams, the unnecessary procedures represented an estimated $123 million in a year.
“The American College of Obstetricians and Gynecologists recognizes that no evidence supports routine speculum examination or BPE in healthy, asymptomatic women younger than 21 years and recommends that these examinations be performed only when medically indicated,” said Jin Qin, ScD, of the Centers for Disease Control and Prevention and colleagues.
“Our results showed that, despite the recommendation, many young women without discernible medical indication received potentially unnecessary BPE or Pap tests, which may be a reflection of a long-standing clinical practice in the United States.”
These findings “demonstrate what happens to vulnerable populations (in this case, girls and young women) when clinicians do not keep up with or do not adhere to new guidelines,” Melissa A. Simon, MD, MPH, wrote in an invited commentary. She acknowledged the challenges of keeping up with new guidelines but noted the potential for harm from unnecessary screening. Dr. Simon is vice chair for clinical research in the department of obstetrics and gynecology at Northwestern University, Chicago.
The researchers analyzed responses from 3,410 young women aged 15-20 years in the National Survey of Family Growth (NSFG) during 2011-2017 and extrapolated the results to estimate nationwide statistics. The researchers found that 23% of young women – 2.6 million in the United States – had received a bimanual pelvic exam during the previous year.
“This analysis focused on the bimanual component of the pelvic examination because it is the most invasive of the pelvic examination components and less likely to be confused with a speculum examination for cervical cancer or STI screening,” the authors note.
More than half of these pelvic exams (54%) – an estimated 1.4 million exams – potentially were unnecessary. The authors classified these pelvic exams as potentially unnecessary if it was not indicated for pregnancy, intrauterine device (IUD) use, or STI treatment in the past 12 months or for another medical problem.
Among the respondents, 5% were pregnant, 22% had been tested for an STI, and 5% had been treated for an STI during the previous year. About a third of respondents (33%) had used at least one type of hormonal contraception besides an IUD in the past year, but only 2% had used an IUD.
Dr. Simon said that some have advocated for routine bimanual pelvic exams to prompt women to see their provider every year, but without evidence to support the practice.
“In fact, many women (younger and older) associate the bimanual pelvic and speculum examinations with fear, anxiety, embarrassment, discomfort, and pain,” Dr. Simon emphasized. “Girls and women with a history of sexual violence may be more vulnerable to these harms. In addition, adolescent girls may delay starting contraception use or obtaining screening for sexually transmitted infections because of fear of pelvic examination, which thus creates unnecessary barriers to obtaining important screening and family-planning methods.”
The researchers also found that 19% of young women, about 2.2 million, had received a Pap test in the previous year. The majority of these (72%) likely were unnecessary, they wrote, explaining that cervical cancer screening is not recommended for those younger than 21 years unless they are HIV positive and sexually active.
“Because HIV infection status is not available in the NSFG, we estimated prevalence of Pap tests performed as part of a routine examination and considered them potentially unnecessary,” the authors explained.
Young women were seven times more likely to have undergone a bimanual pelvic exam if they received a Pap test (adjusted prevalence ratio [aPR], 7.12). In fact, the authors reported that nearly all potentially unnecessary bimanual pelvic exams (98%) occurred during the same visit as a Pap test that was potentially unnecessary as well.
Young women also were more likely to receive a bimanual pelvic exam if they underwent STI testing or used any hormonal contraception besides an IUD (aPR, 1.6 and 1.31, respectively). Those with public insurance or no insurance were less likely to receive a pelvic exam compared with those who had private insurance, although no associations were found with race/ethnicity.
Young women were about four times more likely to have a Pap test if they had STI testing (aPR, 3.77). Odds of a Pap test also were greater among those aged 18-20 years (aPR, 1.54), those with a pregnancy (aPR, 2.31), those with an IUD (aPR, 1.54), and those using any non-IUD hormonal contraception (aPR, 1.75).
Staying up to date on current guidelines and consistently delivering evidence-based care according to those guidelines “is not easy,” Dr. Simon commented. It involves building and maintaining a trusting clinician-patient relationship that centers on shared decision making, keeping up with research, and “unlearn[ing] deeply ingrained practices,” which is difficult.
“Clinicians are not well instructed on how to pivot or unlearn a practice,” Dr. Simon continued. “The science of deimplementation, especially with respect to guideline-concordant care, is in its infancy.” She also noted the value of annual visits, even without routine pelvic exams.
“Rethinking the goals of the annual health examination for young women and learning to unlearn will not put anyone out of business,” Dr. Simon concluded. “Rather, change can increase patients’ connectivity, trust, and engagement with primary care clinicians and, most importantly, avoid harms, especially to those who are most vulnerable.”
No external funding was used. The study authors and Dr. Simon have disclosed no relevant financial relationships.
SOURCE: Qin J et al. JAMA Intern Med. 2019 Jan 6. doi: 10.1001/jamainternmed.2019.5727.
An earlier version of this story appeared on Medscape.com.
A call for shared decision making
The experts who wrote American College of Obstetricians and Gynecologists’ clinical guideline on the pelvic exam (Obstet Gynecol. 2018 Oct;132[4]:e174-80) reviewed available evidence and found insufficient evidence to support routine screening for asymptomatic nonpregnant women who have no increased risk for specific gynecologic conditions (e.g., history of gynecologic cancer). Hence, ACOG recommends routine screening based on a shared decision between the asymptomatic woman and her doctor keeping in mind her medical and family history and her preference. This decision should be made after reviewing the limitations of the exam with regard to insufficient evidence to support its accuracy in screening for ovarian cancer, bacterial vaginosis, trichomoniasis, and genital herpes, plus lack of evidence for other gynecologic conditions.
In addition, we physicians must educate women, especially vulnerable populations, that deferring a pelvic exam for asymptomatic women entails judicious care. Deferring an exam does not mean that we are withholding medical care. If she wants an exam, understanding its limitations, then this preference is an indication itself for the exam as stated in our guideline.
It is important to emphasize to patients that we are deferring Pap smears until age 21 years per ACOG and the American Society for Colposcopy and Cervical Pathology, and that there is no need for a pelvic exam for sexually transmitted infection screening per the Centers for Disease Control and Prevention. Likewise, there is no need for a pelvic exam prior initiation of contraception except for intrauterine device insertion also according to the CDC.
Catherine Cansino, MD, MPH , is associate clinical professor of obstetrics and gynecology at the University of California, Davis. She was asked to comment on the Qin et al. article. Dr. Cansino is a coauthor of the ACOG 2018 guideline on the utility of pelvic exam. She also is a member of the Ob.Gyn. News editorial advisory board. She reported no relevant financial disclosures.
A call for shared decision making
The experts who wrote American College of Obstetricians and Gynecologists’ clinical guideline on the pelvic exam (Obstet Gynecol. 2018 Oct;132[4]:e174-80) reviewed available evidence and found insufficient evidence to support routine screening for asymptomatic nonpregnant women who have no increased risk for specific gynecologic conditions (e.g., history of gynecologic cancer). Hence, ACOG recommends routine screening based on a shared decision between the asymptomatic woman and her doctor keeping in mind her medical and family history and her preference. This decision should be made after reviewing the limitations of the exam with regard to insufficient evidence to support its accuracy in screening for ovarian cancer, bacterial vaginosis, trichomoniasis, and genital herpes, plus lack of evidence for other gynecologic conditions.
In addition, we physicians must educate women, especially vulnerable populations, that deferring a pelvic exam for asymptomatic women entails judicious care. Deferring an exam does not mean that we are withholding medical care. If she wants an exam, understanding its limitations, then this preference is an indication itself for the exam as stated in our guideline.
It is important to emphasize to patients that we are deferring Pap smears until age 21 years per ACOG and the American Society for Colposcopy and Cervical Pathology, and that there is no need for a pelvic exam for sexually transmitted infection screening per the Centers for Disease Control and Prevention. Likewise, there is no need for a pelvic exam prior initiation of contraception except for intrauterine device insertion also according to the CDC.
Catherine Cansino, MD, MPH , is associate clinical professor of obstetrics and gynecology at the University of California, Davis. She was asked to comment on the Qin et al. article. Dr. Cansino is a coauthor of the ACOG 2018 guideline on the utility of pelvic exam. She also is a member of the Ob.Gyn. News editorial advisory board. She reported no relevant financial disclosures.
A call for shared decision making
The experts who wrote American College of Obstetricians and Gynecologists’ clinical guideline on the pelvic exam (Obstet Gynecol. 2018 Oct;132[4]:e174-80) reviewed available evidence and found insufficient evidence to support routine screening for asymptomatic nonpregnant women who have no increased risk for specific gynecologic conditions (e.g., history of gynecologic cancer). Hence, ACOG recommends routine screening based on a shared decision between the asymptomatic woman and her doctor keeping in mind her medical and family history and her preference. This decision should be made after reviewing the limitations of the exam with regard to insufficient evidence to support its accuracy in screening for ovarian cancer, bacterial vaginosis, trichomoniasis, and genital herpes, plus lack of evidence for other gynecologic conditions.
In addition, we physicians must educate women, especially vulnerable populations, that deferring a pelvic exam for asymptomatic women entails judicious care. Deferring an exam does not mean that we are withholding medical care. If she wants an exam, understanding its limitations, then this preference is an indication itself for the exam as stated in our guideline.
It is important to emphasize to patients that we are deferring Pap smears until age 21 years per ACOG and the American Society for Colposcopy and Cervical Pathology, and that there is no need for a pelvic exam for sexually transmitted infection screening per the Centers for Disease Control and Prevention. Likewise, there is no need for a pelvic exam prior initiation of contraception except for intrauterine device insertion also according to the CDC.
Catherine Cansino, MD, MPH , is associate clinical professor of obstetrics and gynecology at the University of California, Davis. She was asked to comment on the Qin et al. article. Dr. Cansino is a coauthor of the ACOG 2018 guideline on the utility of pelvic exam. She also is a member of the Ob.Gyn. News editorial advisory board. She reported no relevant financial disclosures.
according to estimates from a study published online in JAMA Internal Medicine.
Approximately 2.6 million young women – about a quarter of those in this age group – reported receiving a pelvic exam in the previous year even though fewer than 10% were pregnant or receiving treatment for a sexually transmitted infection (STI) at the time.
Similarly, an estimated three in four Pap tests given to women aged 15-20 years likely were unnecessary. Based on Medicare payments for screening Pap tests and pelvic exams, the unnecessary procedures represented an estimated $123 million in a year.
“The American College of Obstetricians and Gynecologists recognizes that no evidence supports routine speculum examination or BPE in healthy, asymptomatic women younger than 21 years and recommends that these examinations be performed only when medically indicated,” said Jin Qin, ScD, of the Centers for Disease Control and Prevention and colleagues.
“Our results showed that, despite the recommendation, many young women without discernible medical indication received potentially unnecessary BPE or Pap tests, which may be a reflection of a long-standing clinical practice in the United States.”
These findings “demonstrate what happens to vulnerable populations (in this case, girls and young women) when clinicians do not keep up with or do not adhere to new guidelines,” Melissa A. Simon, MD, MPH, wrote in an invited commentary. She acknowledged the challenges of keeping up with new guidelines but noted the potential for harm from unnecessary screening. Dr. Simon is vice chair for clinical research in the department of obstetrics and gynecology at Northwestern University, Chicago.
The researchers analyzed responses from 3,410 young women aged 15-20 years in the National Survey of Family Growth (NSFG) during 2011-2017 and extrapolated the results to estimate nationwide statistics. The researchers found that 23% of young women – 2.6 million in the United States – had received a bimanual pelvic exam during the previous year.
“This analysis focused on the bimanual component of the pelvic examination because it is the most invasive of the pelvic examination components and less likely to be confused with a speculum examination for cervical cancer or STI screening,” the authors note.
More than half of these pelvic exams (54%) – an estimated 1.4 million exams – potentially were unnecessary. The authors classified these pelvic exams as potentially unnecessary if it was not indicated for pregnancy, intrauterine device (IUD) use, or STI treatment in the past 12 months or for another medical problem.
Among the respondents, 5% were pregnant, 22% had been tested for an STI, and 5% had been treated for an STI during the previous year. About a third of respondents (33%) had used at least one type of hormonal contraception besides an IUD in the past year, but only 2% had used an IUD.
Dr. Simon said that some have advocated for routine bimanual pelvic exams to prompt women to see their provider every year, but without evidence to support the practice.
“In fact, many women (younger and older) associate the bimanual pelvic and speculum examinations with fear, anxiety, embarrassment, discomfort, and pain,” Dr. Simon emphasized. “Girls and women with a history of sexual violence may be more vulnerable to these harms. In addition, adolescent girls may delay starting contraception use or obtaining screening for sexually transmitted infections because of fear of pelvic examination, which thus creates unnecessary barriers to obtaining important screening and family-planning methods.”
The researchers also found that 19% of young women, about 2.2 million, had received a Pap test in the previous year. The majority of these (72%) likely were unnecessary, they wrote, explaining that cervical cancer screening is not recommended for those younger than 21 years unless they are HIV positive and sexually active.
“Because HIV infection status is not available in the NSFG, we estimated prevalence of Pap tests performed as part of a routine examination and considered them potentially unnecessary,” the authors explained.
Young women were seven times more likely to have undergone a bimanual pelvic exam if they received a Pap test (adjusted prevalence ratio [aPR], 7.12). In fact, the authors reported that nearly all potentially unnecessary bimanual pelvic exams (98%) occurred during the same visit as a Pap test that was potentially unnecessary as well.
Young women also were more likely to receive a bimanual pelvic exam if they underwent STI testing or used any hormonal contraception besides an IUD (aPR, 1.6 and 1.31, respectively). Those with public insurance or no insurance were less likely to receive a pelvic exam compared with those who had private insurance, although no associations were found with race/ethnicity.
Young women were about four times more likely to have a Pap test if they had STI testing (aPR, 3.77). Odds of a Pap test also were greater among those aged 18-20 years (aPR, 1.54), those with a pregnancy (aPR, 2.31), those with an IUD (aPR, 1.54), and those using any non-IUD hormonal contraception (aPR, 1.75).
Staying up to date on current guidelines and consistently delivering evidence-based care according to those guidelines “is not easy,” Dr. Simon commented. It involves building and maintaining a trusting clinician-patient relationship that centers on shared decision making, keeping up with research, and “unlearn[ing] deeply ingrained practices,” which is difficult.
“Clinicians are not well instructed on how to pivot or unlearn a practice,” Dr. Simon continued. “The science of deimplementation, especially with respect to guideline-concordant care, is in its infancy.” She also noted the value of annual visits, even without routine pelvic exams.
“Rethinking the goals of the annual health examination for young women and learning to unlearn will not put anyone out of business,” Dr. Simon concluded. “Rather, change can increase patients’ connectivity, trust, and engagement with primary care clinicians and, most importantly, avoid harms, especially to those who are most vulnerable.”
No external funding was used. The study authors and Dr. Simon have disclosed no relevant financial relationships.
SOURCE: Qin J et al. JAMA Intern Med. 2019 Jan 6. doi: 10.1001/jamainternmed.2019.5727.
An earlier version of this story appeared on Medscape.com.
according to estimates from a study published online in JAMA Internal Medicine.
Approximately 2.6 million young women – about a quarter of those in this age group – reported receiving a pelvic exam in the previous year even though fewer than 10% were pregnant or receiving treatment for a sexually transmitted infection (STI) at the time.
Similarly, an estimated three in four Pap tests given to women aged 15-20 years likely were unnecessary. Based on Medicare payments for screening Pap tests and pelvic exams, the unnecessary procedures represented an estimated $123 million in a year.
“The American College of Obstetricians and Gynecologists recognizes that no evidence supports routine speculum examination or BPE in healthy, asymptomatic women younger than 21 years and recommends that these examinations be performed only when medically indicated,” said Jin Qin, ScD, of the Centers for Disease Control and Prevention and colleagues.
“Our results showed that, despite the recommendation, many young women without discernible medical indication received potentially unnecessary BPE or Pap tests, which may be a reflection of a long-standing clinical practice in the United States.”
These findings “demonstrate what happens to vulnerable populations (in this case, girls and young women) when clinicians do not keep up with or do not adhere to new guidelines,” Melissa A. Simon, MD, MPH, wrote in an invited commentary. She acknowledged the challenges of keeping up with new guidelines but noted the potential for harm from unnecessary screening. Dr. Simon is vice chair for clinical research in the department of obstetrics and gynecology at Northwestern University, Chicago.
The researchers analyzed responses from 3,410 young women aged 15-20 years in the National Survey of Family Growth (NSFG) during 2011-2017 and extrapolated the results to estimate nationwide statistics. The researchers found that 23% of young women – 2.6 million in the United States – had received a bimanual pelvic exam during the previous year.
“This analysis focused on the bimanual component of the pelvic examination because it is the most invasive of the pelvic examination components and less likely to be confused with a speculum examination for cervical cancer or STI screening,” the authors note.
More than half of these pelvic exams (54%) – an estimated 1.4 million exams – potentially were unnecessary. The authors classified these pelvic exams as potentially unnecessary if it was not indicated for pregnancy, intrauterine device (IUD) use, or STI treatment in the past 12 months or for another medical problem.
Among the respondents, 5% were pregnant, 22% had been tested for an STI, and 5% had been treated for an STI during the previous year. About a third of respondents (33%) had used at least one type of hormonal contraception besides an IUD in the past year, but only 2% had used an IUD.
Dr. Simon said that some have advocated for routine bimanual pelvic exams to prompt women to see their provider every year, but without evidence to support the practice.
“In fact, many women (younger and older) associate the bimanual pelvic and speculum examinations with fear, anxiety, embarrassment, discomfort, and pain,” Dr. Simon emphasized. “Girls and women with a history of sexual violence may be more vulnerable to these harms. In addition, adolescent girls may delay starting contraception use or obtaining screening for sexually transmitted infections because of fear of pelvic examination, which thus creates unnecessary barriers to obtaining important screening and family-planning methods.”
The researchers also found that 19% of young women, about 2.2 million, had received a Pap test in the previous year. The majority of these (72%) likely were unnecessary, they wrote, explaining that cervical cancer screening is not recommended for those younger than 21 years unless they are HIV positive and sexually active.
“Because HIV infection status is not available in the NSFG, we estimated prevalence of Pap tests performed as part of a routine examination and considered them potentially unnecessary,” the authors explained.
Young women were seven times more likely to have undergone a bimanual pelvic exam if they received a Pap test (adjusted prevalence ratio [aPR], 7.12). In fact, the authors reported that nearly all potentially unnecessary bimanual pelvic exams (98%) occurred during the same visit as a Pap test that was potentially unnecessary as well.
Young women also were more likely to receive a bimanual pelvic exam if they underwent STI testing or used any hormonal contraception besides an IUD (aPR, 1.6 and 1.31, respectively). Those with public insurance or no insurance were less likely to receive a pelvic exam compared with those who had private insurance, although no associations were found with race/ethnicity.
Young women were about four times more likely to have a Pap test if they had STI testing (aPR, 3.77). Odds of a Pap test also were greater among those aged 18-20 years (aPR, 1.54), those with a pregnancy (aPR, 2.31), those with an IUD (aPR, 1.54), and those using any non-IUD hormonal contraception (aPR, 1.75).
Staying up to date on current guidelines and consistently delivering evidence-based care according to those guidelines “is not easy,” Dr. Simon commented. It involves building and maintaining a trusting clinician-patient relationship that centers on shared decision making, keeping up with research, and “unlearn[ing] deeply ingrained practices,” which is difficult.
“Clinicians are not well instructed on how to pivot or unlearn a practice,” Dr. Simon continued. “The science of deimplementation, especially with respect to guideline-concordant care, is in its infancy.” She also noted the value of annual visits, even without routine pelvic exams.
“Rethinking the goals of the annual health examination for young women and learning to unlearn will not put anyone out of business,” Dr. Simon concluded. “Rather, change can increase patients’ connectivity, trust, and engagement with primary care clinicians and, most importantly, avoid harms, especially to those who are most vulnerable.”
No external funding was used. The study authors and Dr. Simon have disclosed no relevant financial relationships.
SOURCE: Qin J et al. JAMA Intern Med. 2019 Jan 6. doi: 10.1001/jamainternmed.2019.5727.
An earlier version of this story appeared on Medscape.com.
Provide appropriate sexual, reproductive health care for transgender patients
I recently was on a panel of experts discussing how to prevent HIV among transgender youth. Preventing HIV among transgender youth, especially transgender youth of color, remains a challenge for multiple reasons – racism, poverty, stigma, marginalization, and discrimination play a role in the HIV epidemic. A barrier to preventing HIV infections among transgender youth is a lack of knowledge on how to provide them with comprehensive sexual and reproductive health care. Here are some tips and resources that can help you ensure that transgender youth are safe and healthy.
One of the challenges of obtaining a sexual history is asking the right questions For example, if you have a transgender male assigned female at birth, ask whether their partners produce sperm instead of asking about the sex of their partners. A transgender male’s partner may identify as female but is assigned male at birth and uses her penis during sex. Furthermore, a transgender male may be on testosterone, but he still can get pregnant. Asking how they use their organs is just as important. A transgender male who has condomless penile-vaginal sex with multiple partners is at a higher risk for HIV infection than is a transgender male who shares sex toys with his only partner.
Normalizing that you ask a comprehensive sexual history to all your patients regardless of gender identity may put the patient at ease. Many transgender people are reluctant to disclose their gender identity to their provider because they are afraid that the provider may fixate on their sexuality once they do. Stating that you ask sexual health questions to all your patients may prevent the transgender patient from feeling singled out.
Finally, you don’t have to ask a sexual history with every transgender patient, just as you wouldn’t for your cisgender patients. If a patient is complaining of a sprained ankle, a sexual history may not be helpful, compared with obtaining one when a patient comes in with pelvic pain. Many transgender patients avoid care because they are frequently asked about their sexual history or gender identity when these are not relevant to their chief complaint.
Here are some helpful questions to ask when taking a sexual history, according to the University of California, San Francisco, Transgender Care & Treatment Guidelines.1
- Are you having sex? How many sex partners have you had in the past year?
- Who are you having sex with? What types of sex are you having? What parts of your anatomy do you use for sex?
- How do you protect yourself from STIs?
- What STIs have you had in the past, if any? When were you last tested for STIs?
- Has your partner(s) ever been diagnosed with any STIs?
- Do you use alcohol or any drugs when you have sex?
- Do you exchange sex for money, drugs, or a place to stay?
Also, use a trauma-informed approach when working with transgender patients. Many have been victims of sexual trauma. Always have a chaperone accompany you during the exam, explain to the patient what you plan to do and why it is necessary, and allow them to decline (and document their declining the physical exam). Also consider having your patient self-swab for STI screening if appropriate.1
Like obtaining a sexual history, routine screenings for certain types of cancers will be based on the organs the patient has. For example, a transgender woman assigned male at birth will not need a cervical cancer screening, but a transgender man assigned female at birth may need one – if the patient still has a cervix. Cervical cancer screening guidelines are similar for transgender men as it is for nontransgender women, and one should use the same guidelines endorsed by the American Cancer Society, American Society of Colposcopy and Cervical Pathology, American Society of Clinical Pathologists, U.S. Preventive Services Task Force, and the World Health Organization.2-4
Cervical screenings should never be a requirement for testosterone therapy, and no transgender male under the age of 21 years will need cervical screening. The University of California guidelines offers tips on how to make transgender men more comfortable during cervical cancer screening.5
Contraception and menstrual management also are important for transgender patients. Testosterone can induce amenorrhea for transgender men, but it is not good birth control. If a transgender male patient has sex with partners that produce sperm, then the physician should discuss effective birth control options. There is no ideal birth control option for transgender men. One must consider multiple factors including the patient’s desire for pregnancy, desire to cease periods, ease of administration, and risk for thrombosis.
Most transgender men may balk at the idea of taking estrogen-containing contraception, but it is more effective than oral progestin-only pills. Intrauterine devices are highly effective in pregnancy prevention and can achieve amenorrhea in 50% of users within 1 year,but some transmen may become dysphoric with the procedure. 6 The etonogestrel implants also are highly effective birth control, but irregular periods are common, leading to discontinuation. Depot medroxyprogesterone is highly effective in preventing pregnancy and can induce amenorrhea in 70% of users within 1 year and 80% of users in 2 years, but also is associated with weight gain in one-third of users.7 Finally, pubertal blockers can rapidly stop periods for transmen who are severely dysphoric from their menses; however, before achieving amenorrhea, a flare bleed can occur 4-6 weeks after administration.8 Support from a mental health therapist during this time is critical. Pubertal blockers, nevertheless, are not suitable birth control.
When providing affirming sexual and reproductive health care for transgender patients, key principles include focusing on organs and activities over identity. Additionally, screening for certain types of cancers also is dependent on organs. Finally, do not neglect the importance of contraception among transgender men. Taking these principles in consideration will help you provide excellent care for transgender youth.
Dr. Montano is an assistant professor of pediatrics at the University of Pittsburgh and an adolescent medicine physician at the Children’s Hospital of Pittsburgh. He said he had no relevant financial disclosures. Email him at [email protected].
References
1. Transgender people and sexually transmitted infections (https://transcare.ucsf.edu/guidelines/stis).
2. CA Cancer J Clin. 2012 May-Jun;62(3):147-72.
3. Ann Intern Med. 2012;156(12):880-91.
4. Cervical cancer screening in developing countries: Report of a WHO consultation. 2002. World Health Organization, Geneva.
5. Screening for cervical cancer for transgender men (https://transcare.ucsf.edu/guidelines/cervical-cancer).
6. Contraception. 2002 Feb;65(2):129-32.
7. Rev Endocr Metab Disord. 2011 Jun;12(2):93-106.
8. Int J Womens Health. 2014 Jun 23;6:631-7.
Resources
Breast cancer screening in transgender men. (https://transcare.ucsf.edu/guidelines/breast-cancer-men).
Screening for breast cancer in transgender women. (https://transcare.ucsf.edu/guidelines/breast-cancer-women).
Transgender health and HIV (https://transcare.ucsf.edu/guidelines/hiv).
Centers for Disease Control and Prevention: HIV and Transgender People (https://www.cdc.gov/hiv/group/gender/transgender/index.html).
I recently was on a panel of experts discussing how to prevent HIV among transgender youth. Preventing HIV among transgender youth, especially transgender youth of color, remains a challenge for multiple reasons – racism, poverty, stigma, marginalization, and discrimination play a role in the HIV epidemic. A barrier to preventing HIV infections among transgender youth is a lack of knowledge on how to provide them with comprehensive sexual and reproductive health care. Here are some tips and resources that can help you ensure that transgender youth are safe and healthy.
One of the challenges of obtaining a sexual history is asking the right questions For example, if you have a transgender male assigned female at birth, ask whether their partners produce sperm instead of asking about the sex of their partners. A transgender male’s partner may identify as female but is assigned male at birth and uses her penis during sex. Furthermore, a transgender male may be on testosterone, but he still can get pregnant. Asking how they use their organs is just as important. A transgender male who has condomless penile-vaginal sex with multiple partners is at a higher risk for HIV infection than is a transgender male who shares sex toys with his only partner.
Normalizing that you ask a comprehensive sexual history to all your patients regardless of gender identity may put the patient at ease. Many transgender people are reluctant to disclose their gender identity to their provider because they are afraid that the provider may fixate on their sexuality once they do. Stating that you ask sexual health questions to all your patients may prevent the transgender patient from feeling singled out.
Finally, you don’t have to ask a sexual history with every transgender patient, just as you wouldn’t for your cisgender patients. If a patient is complaining of a sprained ankle, a sexual history may not be helpful, compared with obtaining one when a patient comes in with pelvic pain. Many transgender patients avoid care because they are frequently asked about their sexual history or gender identity when these are not relevant to their chief complaint.
Here are some helpful questions to ask when taking a sexual history, according to the University of California, San Francisco, Transgender Care & Treatment Guidelines.1
- Are you having sex? How many sex partners have you had in the past year?
- Who are you having sex with? What types of sex are you having? What parts of your anatomy do you use for sex?
- How do you protect yourself from STIs?
- What STIs have you had in the past, if any? When were you last tested for STIs?
- Has your partner(s) ever been diagnosed with any STIs?
- Do you use alcohol or any drugs when you have sex?
- Do you exchange sex for money, drugs, or a place to stay?
Also, use a trauma-informed approach when working with transgender patients. Many have been victims of sexual trauma. Always have a chaperone accompany you during the exam, explain to the patient what you plan to do and why it is necessary, and allow them to decline (and document their declining the physical exam). Also consider having your patient self-swab for STI screening if appropriate.1
Like obtaining a sexual history, routine screenings for certain types of cancers will be based on the organs the patient has. For example, a transgender woman assigned male at birth will not need a cervical cancer screening, but a transgender man assigned female at birth may need one – if the patient still has a cervix. Cervical cancer screening guidelines are similar for transgender men as it is for nontransgender women, and one should use the same guidelines endorsed by the American Cancer Society, American Society of Colposcopy and Cervical Pathology, American Society of Clinical Pathologists, U.S. Preventive Services Task Force, and the World Health Organization.2-4
Cervical screenings should never be a requirement for testosterone therapy, and no transgender male under the age of 21 years will need cervical screening. The University of California guidelines offers tips on how to make transgender men more comfortable during cervical cancer screening.5
Contraception and menstrual management also are important for transgender patients. Testosterone can induce amenorrhea for transgender men, but it is not good birth control. If a transgender male patient has sex with partners that produce sperm, then the physician should discuss effective birth control options. There is no ideal birth control option for transgender men. One must consider multiple factors including the patient’s desire for pregnancy, desire to cease periods, ease of administration, and risk for thrombosis.
Most transgender men may balk at the idea of taking estrogen-containing contraception, but it is more effective than oral progestin-only pills. Intrauterine devices are highly effective in pregnancy prevention and can achieve amenorrhea in 50% of users within 1 year,but some transmen may become dysphoric with the procedure. 6 The etonogestrel implants also are highly effective birth control, but irregular periods are common, leading to discontinuation. Depot medroxyprogesterone is highly effective in preventing pregnancy and can induce amenorrhea in 70% of users within 1 year and 80% of users in 2 years, but also is associated with weight gain in one-third of users.7 Finally, pubertal blockers can rapidly stop periods for transmen who are severely dysphoric from their menses; however, before achieving amenorrhea, a flare bleed can occur 4-6 weeks after administration.8 Support from a mental health therapist during this time is critical. Pubertal blockers, nevertheless, are not suitable birth control.
When providing affirming sexual and reproductive health care for transgender patients, key principles include focusing on organs and activities over identity. Additionally, screening for certain types of cancers also is dependent on organs. Finally, do not neglect the importance of contraception among transgender men. Taking these principles in consideration will help you provide excellent care for transgender youth.
Dr. Montano is an assistant professor of pediatrics at the University of Pittsburgh and an adolescent medicine physician at the Children’s Hospital of Pittsburgh. He said he had no relevant financial disclosures. Email him at [email protected].
References
1. Transgender people and sexually transmitted infections (https://transcare.ucsf.edu/guidelines/stis).
2. CA Cancer J Clin. 2012 May-Jun;62(3):147-72.
3. Ann Intern Med. 2012;156(12):880-91.
4. Cervical cancer screening in developing countries: Report of a WHO consultation. 2002. World Health Organization, Geneva.
5. Screening for cervical cancer for transgender men (https://transcare.ucsf.edu/guidelines/cervical-cancer).
6. Contraception. 2002 Feb;65(2):129-32.
7. Rev Endocr Metab Disord. 2011 Jun;12(2):93-106.
8. Int J Womens Health. 2014 Jun 23;6:631-7.
Resources
Breast cancer screening in transgender men. (https://transcare.ucsf.edu/guidelines/breast-cancer-men).
Screening for breast cancer in transgender women. (https://transcare.ucsf.edu/guidelines/breast-cancer-women).
Transgender health and HIV (https://transcare.ucsf.edu/guidelines/hiv).
Centers for Disease Control and Prevention: HIV and Transgender People (https://www.cdc.gov/hiv/group/gender/transgender/index.html).
I recently was on a panel of experts discussing how to prevent HIV among transgender youth. Preventing HIV among transgender youth, especially transgender youth of color, remains a challenge for multiple reasons – racism, poverty, stigma, marginalization, and discrimination play a role in the HIV epidemic. A barrier to preventing HIV infections among transgender youth is a lack of knowledge on how to provide them with comprehensive sexual and reproductive health care. Here are some tips and resources that can help you ensure that transgender youth are safe and healthy.
One of the challenges of obtaining a sexual history is asking the right questions For example, if you have a transgender male assigned female at birth, ask whether their partners produce sperm instead of asking about the sex of their partners. A transgender male’s partner may identify as female but is assigned male at birth and uses her penis during sex. Furthermore, a transgender male may be on testosterone, but he still can get pregnant. Asking how they use their organs is just as important. A transgender male who has condomless penile-vaginal sex with multiple partners is at a higher risk for HIV infection than is a transgender male who shares sex toys with his only partner.
Normalizing that you ask a comprehensive sexual history to all your patients regardless of gender identity may put the patient at ease. Many transgender people are reluctant to disclose their gender identity to their provider because they are afraid that the provider may fixate on their sexuality once they do. Stating that you ask sexual health questions to all your patients may prevent the transgender patient from feeling singled out.
Finally, you don’t have to ask a sexual history with every transgender patient, just as you wouldn’t for your cisgender patients. If a patient is complaining of a sprained ankle, a sexual history may not be helpful, compared with obtaining one when a patient comes in with pelvic pain. Many transgender patients avoid care because they are frequently asked about their sexual history or gender identity when these are not relevant to their chief complaint.
Here are some helpful questions to ask when taking a sexual history, according to the University of California, San Francisco, Transgender Care & Treatment Guidelines.1
- Are you having sex? How many sex partners have you had in the past year?
- Who are you having sex with? What types of sex are you having? What parts of your anatomy do you use for sex?
- How do you protect yourself from STIs?
- What STIs have you had in the past, if any? When were you last tested for STIs?
- Has your partner(s) ever been diagnosed with any STIs?
- Do you use alcohol or any drugs when you have sex?
- Do you exchange sex for money, drugs, or a place to stay?
Also, use a trauma-informed approach when working with transgender patients. Many have been victims of sexual trauma. Always have a chaperone accompany you during the exam, explain to the patient what you plan to do and why it is necessary, and allow them to decline (and document their declining the physical exam). Also consider having your patient self-swab for STI screening if appropriate.1
Like obtaining a sexual history, routine screenings for certain types of cancers will be based on the organs the patient has. For example, a transgender woman assigned male at birth will not need a cervical cancer screening, but a transgender man assigned female at birth may need one – if the patient still has a cervix. Cervical cancer screening guidelines are similar for transgender men as it is for nontransgender women, and one should use the same guidelines endorsed by the American Cancer Society, American Society of Colposcopy and Cervical Pathology, American Society of Clinical Pathologists, U.S. Preventive Services Task Force, and the World Health Organization.2-4
Cervical screenings should never be a requirement for testosterone therapy, and no transgender male under the age of 21 years will need cervical screening. The University of California guidelines offers tips on how to make transgender men more comfortable during cervical cancer screening.5
Contraception and menstrual management also are important for transgender patients. Testosterone can induce amenorrhea for transgender men, but it is not good birth control. If a transgender male patient has sex with partners that produce sperm, then the physician should discuss effective birth control options. There is no ideal birth control option for transgender men. One must consider multiple factors including the patient’s desire for pregnancy, desire to cease periods, ease of administration, and risk for thrombosis.
Most transgender men may balk at the idea of taking estrogen-containing contraception, but it is more effective than oral progestin-only pills. Intrauterine devices are highly effective in pregnancy prevention and can achieve amenorrhea in 50% of users within 1 year,but some transmen may become dysphoric with the procedure. 6 The etonogestrel implants also are highly effective birth control, but irregular periods are common, leading to discontinuation. Depot medroxyprogesterone is highly effective in preventing pregnancy and can induce amenorrhea in 70% of users within 1 year and 80% of users in 2 years, but also is associated with weight gain in one-third of users.7 Finally, pubertal blockers can rapidly stop periods for transmen who are severely dysphoric from their menses; however, before achieving amenorrhea, a flare bleed can occur 4-6 weeks after administration.8 Support from a mental health therapist during this time is critical. Pubertal blockers, nevertheless, are not suitable birth control.
When providing affirming sexual and reproductive health care for transgender patients, key principles include focusing on organs and activities over identity. Additionally, screening for certain types of cancers also is dependent on organs. Finally, do not neglect the importance of contraception among transgender men. Taking these principles in consideration will help you provide excellent care for transgender youth.
Dr. Montano is an assistant professor of pediatrics at the University of Pittsburgh and an adolescent medicine physician at the Children’s Hospital of Pittsburgh. He said he had no relevant financial disclosures. Email him at [email protected].
References
1. Transgender people and sexually transmitted infections (https://transcare.ucsf.edu/guidelines/stis).
2. CA Cancer J Clin. 2012 May-Jun;62(3):147-72.
3. Ann Intern Med. 2012;156(12):880-91.
4. Cervical cancer screening in developing countries: Report of a WHO consultation. 2002. World Health Organization, Geneva.
5. Screening for cervical cancer for transgender men (https://transcare.ucsf.edu/guidelines/cervical-cancer).
6. Contraception. 2002 Feb;65(2):129-32.
7. Rev Endocr Metab Disord. 2011 Jun;12(2):93-106.
8. Int J Womens Health. 2014 Jun 23;6:631-7.
Resources
Breast cancer screening in transgender men. (https://transcare.ucsf.edu/guidelines/breast-cancer-men).
Screening for breast cancer in transgender women. (https://transcare.ucsf.edu/guidelines/breast-cancer-women).
Transgender health and HIV (https://transcare.ucsf.edu/guidelines/hiv).
Centers for Disease Control and Prevention: HIV and Transgender People (https://www.cdc.gov/hiv/group/gender/transgender/index.html).
Cognitive problems after extremely preterm birth persist
Cognitive and neuropsychological impairment associated with extremely preterm (EP) birth persists into young adulthood, according to findings from the 1995 EPICure cohort.
Of note, intellectual impairment increased significantly after the age of 11 years among 19-year-olds in the cohort of individuals born EP, Helen O’Reilly, PhD, of the Institute for Women’s Health at University College London and colleagues reported in Pediatrics.
Neuropsychological assessment to examine general cognitive abilities, visuomotor abilities, prospective memory, and certain aspects of executive functioning and language in 127 cases and 64 term-born controls showed significantly lower scores across all tests in those born EP.
Impairment in at least one neuropsychological domain was present in 60% of EP birth cases (compared with 21% of controls), with 35% having impairment in at least four domains. Most deficits occurred in general cognitive function and/or visuomotor abilities.
Further, and those with cognitive impairment at 11 years were at increased risk of deficit at 19 years (RR, 3.56), even after adjustment for sex and socioeconomic status, the authors wrote.
None of the term-born controls had a cognitive impairment at 11 years, and two (3%) had impairment at 19 years.
Studies of adults born very preterm have revealed that these individuals are at risk for neuropsychological impairment, but the extent of such impairment in individuals with EP birth, defined as birth before 26 weeks’ gestation, had not previously been studied in the long term.
Assessments in the EPICure cohort of individuals born EP in 1995 previously showed scores at 1.1-1.6 standard deviations lower on measures of general cognitive function, compared with standardized norms and/or term-born controls, at age 2.5, 6, and 11 years, Dr. O’Reilly and colleagues explained.
The current findings indicate that general cognitive and neuropsychological functioning problems associated with EP birth persist and can increase into early adulthood, and they “highlight the need for early and ongoing neuropsychological and educational assessment in EP children to ensure these children receive appropriate support in school and for planned educational pathways,” the investigators concluded.
In an accompanying editorial, Louis A. Schmidt, PhD, and Saroj Saigal, MD, of McMaster University, Hamilton, Ont., wrote that these findings “provide compelling evidence for persistent effects of cognitive impairments” in individuals born EP.
They highlighted three lessons from the study:
- It is important to control for anxiety in future studies like this “to eliminate potential confounding influences of anxiety when examining performance-based measures in the laboratory setting,” as individuals born EP are known to exhibit anxiety.
- Group heterogeneity also should be considered, as all survivors of prematurity are not alike.
- Measurement equivalency should be established between groups.
With respect to the latter, “although many of the measures used by O’Reilly et al. have been normed, issues of measurement invariance have not been established between EP and control groups on some of the measures reported,” Dr. Schmidt and Dr. Saigal wrote, noting that “many other studies [also] fail to consider this fundamental measurement property.”
“Considering issues of measurement equivalency is of critical importance to ensuring unbiased interpretations of findings,” they added, concluding that the findings by O’Reilly et al. represent an important contribution and confirm findings from many prior studies of extreme prematurity, which “informs how we effectively manage these problems.”
“As the percentage of preterm birth continues to rise worldwide, coupled with reduced morbidity and mortality, and with more EP infants reaching adulthood, there is a need for prospective, long-term outcome studies of extreme prematurity,” Dr. Schmidt and Dr. Saigal added.
The study was funded by the Medical Research Council United Kingdom. The authors reported having no relevant financial disclosures. The editorial by Dr. Schmidt and Dr. Saigal, who also reported having no relevant financial disclosures, was supported by the Canadian Institutes of Health Research.
SOURCES: O’Reilly H et al. Pediatrics. 2020;145(2):e20192087; Schmidt LA, Saigal S. Pediatrics. 2020;145(2):e20193359.
Cognitive and neuropsychological impairment associated with extremely preterm (EP) birth persists into young adulthood, according to findings from the 1995 EPICure cohort.
Of note, intellectual impairment increased significantly after the age of 11 years among 19-year-olds in the cohort of individuals born EP, Helen O’Reilly, PhD, of the Institute for Women’s Health at University College London and colleagues reported in Pediatrics.
Neuropsychological assessment to examine general cognitive abilities, visuomotor abilities, prospective memory, and certain aspects of executive functioning and language in 127 cases and 64 term-born controls showed significantly lower scores across all tests in those born EP.
Impairment in at least one neuropsychological domain was present in 60% of EP birth cases (compared with 21% of controls), with 35% having impairment in at least four domains. Most deficits occurred in general cognitive function and/or visuomotor abilities.
Further, and those with cognitive impairment at 11 years were at increased risk of deficit at 19 years (RR, 3.56), even after adjustment for sex and socioeconomic status, the authors wrote.
None of the term-born controls had a cognitive impairment at 11 years, and two (3%) had impairment at 19 years.
Studies of adults born very preterm have revealed that these individuals are at risk for neuropsychological impairment, but the extent of such impairment in individuals with EP birth, defined as birth before 26 weeks’ gestation, had not previously been studied in the long term.
Assessments in the EPICure cohort of individuals born EP in 1995 previously showed scores at 1.1-1.6 standard deviations lower on measures of general cognitive function, compared with standardized norms and/or term-born controls, at age 2.5, 6, and 11 years, Dr. O’Reilly and colleagues explained.
The current findings indicate that general cognitive and neuropsychological functioning problems associated with EP birth persist and can increase into early adulthood, and they “highlight the need for early and ongoing neuropsychological and educational assessment in EP children to ensure these children receive appropriate support in school and for planned educational pathways,” the investigators concluded.
In an accompanying editorial, Louis A. Schmidt, PhD, and Saroj Saigal, MD, of McMaster University, Hamilton, Ont., wrote that these findings “provide compelling evidence for persistent effects of cognitive impairments” in individuals born EP.
They highlighted three lessons from the study:
- It is important to control for anxiety in future studies like this “to eliminate potential confounding influences of anxiety when examining performance-based measures in the laboratory setting,” as individuals born EP are known to exhibit anxiety.
- Group heterogeneity also should be considered, as all survivors of prematurity are not alike.
- Measurement equivalency should be established between groups.
With respect to the latter, “although many of the measures used by O’Reilly et al. have been normed, issues of measurement invariance have not been established between EP and control groups on some of the measures reported,” Dr. Schmidt and Dr. Saigal wrote, noting that “many other studies [also] fail to consider this fundamental measurement property.”
“Considering issues of measurement equivalency is of critical importance to ensuring unbiased interpretations of findings,” they added, concluding that the findings by O’Reilly et al. represent an important contribution and confirm findings from many prior studies of extreme prematurity, which “informs how we effectively manage these problems.”
“As the percentage of preterm birth continues to rise worldwide, coupled with reduced morbidity and mortality, and with more EP infants reaching adulthood, there is a need for prospective, long-term outcome studies of extreme prematurity,” Dr. Schmidt and Dr. Saigal added.
The study was funded by the Medical Research Council United Kingdom. The authors reported having no relevant financial disclosures. The editorial by Dr. Schmidt and Dr. Saigal, who also reported having no relevant financial disclosures, was supported by the Canadian Institutes of Health Research.
SOURCES: O’Reilly H et al. Pediatrics. 2020;145(2):e20192087; Schmidt LA, Saigal S. Pediatrics. 2020;145(2):e20193359.
Cognitive and neuropsychological impairment associated with extremely preterm (EP) birth persists into young adulthood, according to findings from the 1995 EPICure cohort.
Of note, intellectual impairment increased significantly after the age of 11 years among 19-year-olds in the cohort of individuals born EP, Helen O’Reilly, PhD, of the Institute for Women’s Health at University College London and colleagues reported in Pediatrics.
Neuropsychological assessment to examine general cognitive abilities, visuomotor abilities, prospective memory, and certain aspects of executive functioning and language in 127 cases and 64 term-born controls showed significantly lower scores across all tests in those born EP.
Impairment in at least one neuropsychological domain was present in 60% of EP birth cases (compared with 21% of controls), with 35% having impairment in at least four domains. Most deficits occurred in general cognitive function and/or visuomotor abilities.
Further, and those with cognitive impairment at 11 years were at increased risk of deficit at 19 years (RR, 3.56), even after adjustment for sex and socioeconomic status, the authors wrote.
None of the term-born controls had a cognitive impairment at 11 years, and two (3%) had impairment at 19 years.
Studies of adults born very preterm have revealed that these individuals are at risk for neuropsychological impairment, but the extent of such impairment in individuals with EP birth, defined as birth before 26 weeks’ gestation, had not previously been studied in the long term.
Assessments in the EPICure cohort of individuals born EP in 1995 previously showed scores at 1.1-1.6 standard deviations lower on measures of general cognitive function, compared with standardized norms and/or term-born controls, at age 2.5, 6, and 11 years, Dr. O’Reilly and colleagues explained.
The current findings indicate that general cognitive and neuropsychological functioning problems associated with EP birth persist and can increase into early adulthood, and they “highlight the need for early and ongoing neuropsychological and educational assessment in EP children to ensure these children receive appropriate support in school and for planned educational pathways,” the investigators concluded.
In an accompanying editorial, Louis A. Schmidt, PhD, and Saroj Saigal, MD, of McMaster University, Hamilton, Ont., wrote that these findings “provide compelling evidence for persistent effects of cognitive impairments” in individuals born EP.
They highlighted three lessons from the study:
- It is important to control for anxiety in future studies like this “to eliminate potential confounding influences of anxiety when examining performance-based measures in the laboratory setting,” as individuals born EP are known to exhibit anxiety.
- Group heterogeneity also should be considered, as all survivors of prematurity are not alike.
- Measurement equivalency should be established between groups.
With respect to the latter, “although many of the measures used by O’Reilly et al. have been normed, issues of measurement invariance have not been established between EP and control groups on some of the measures reported,” Dr. Schmidt and Dr. Saigal wrote, noting that “many other studies [also] fail to consider this fundamental measurement property.”
“Considering issues of measurement equivalency is of critical importance to ensuring unbiased interpretations of findings,” they added, concluding that the findings by O’Reilly et al. represent an important contribution and confirm findings from many prior studies of extreme prematurity, which “informs how we effectively manage these problems.”
“As the percentage of preterm birth continues to rise worldwide, coupled with reduced morbidity and mortality, and with more EP infants reaching adulthood, there is a need for prospective, long-term outcome studies of extreme prematurity,” Dr. Schmidt and Dr. Saigal added.
The study was funded by the Medical Research Council United Kingdom. The authors reported having no relevant financial disclosures. The editorial by Dr. Schmidt and Dr. Saigal, who also reported having no relevant financial disclosures, was supported by the Canadian Institutes of Health Research.
SOURCES: O’Reilly H et al. Pediatrics. 2020;145(2):e20192087; Schmidt LA, Saigal S. Pediatrics. 2020;145(2):e20193359.
FROM PEDIATRICS
Experts in Europe issue guidance on atopic dermatitis in pregnancy
MADRID – European atopic dermatitis experts have issued formal guidance on a seriously neglected topic: treatment of the disease during pregnancy, breastfeeding, and in men planning to father children.
The impetus for the project was clear: “,” Christian Vestergaard, MD, PhD, declared at a meeting of the European Task Force on Atopic Dermatitis held in conjunction with the annual congress of the European Academy of Dermatology and Venereology.
He presented highlights of the task force’s position paper on the topic, for which he served as first author. The group’s recommendations are based on expert opinion, since randomized clinical trial literature in this area is nonexistent because of ethical concerns. But the task force, comprising a who’s who in European dermatology, drew on a wealth of collective clinical experience in this area.
“We have all of Europe involved in doing this position statement. It’s meant as what we think is proper treatment and what we can say about the different drugs,” explained Dr. Vestergaard, a dermatologist at the University of Aarhus (Denmark).
Most nonobstetricians are intimidated by atopic dermatitis (AD) in pregnancy, and are concerned about the potential for treatment-related harm to the fetus. As a consequence, they are reluctant to recommend anything beyond weak class I topical corticosteroids and emollients. That’s clearly insufficient in light of the vast scope of need, he asserted. After all, AD affects 15%-20% of all children and persists or reappears in adulthood in one out of five of them. Half of those adults are women, many of whom will at some point wish to become pregnant. And many men with AD will eventually want to father children.
A key message from the task force is that untreated AD in pregnancy potentially places the mother and fetus at risk of serious complications, including Staphylococcus aureus infection and eczema herpeticum.
“If you take one thing away from our position paper, it’s that you can use class II or III topical corticosteroids in pregnant women as first-line therapy,” Dr. Vestergaard said.
This stance contradicts a longstanding widely held concern that topical steroids in pregnancy might increase the risk of facial cleft in the offspring, a worry that has been convincingly debunked in a Cochrane systematic review of 14 studies including more than 1.6 million pregnancies. The report concluded there was no association between topical corticosteroids of any potency with preterm delivery, birth defects, or low Apgar scores (Cochrane Database Syst Rev. 2015 Oct 26. doi: 10.1002/14651858.CD007346.pub3).
The task force recommends that if class II or III topical corticosteroid use in pregnancy exceeds 200 g/month, it’s worth considering add-on UV therapy, with narrow band UVB-311 nm as the regimen of choice; it can be used liberally. UV therapy with psoralens is not advised because of a theoretical risk of mutagenicity.
Product labeling for the topical calcineurin inhibitors declares that the agents should not be used during pregnancy. However, the European task force position paper takes issue with that and declares that topical tacrolimus (Protopic) can be considered an off-label first-line therapy in pregnant women with an insufficient response to liberal use of emollients. The same holds true for breastfeeding patients with AD. Just as when topical corticosteroids are used in the nipple area, topical tacrolimus should be applied after nursing, and the nipple area should be gently cleaned before nursing.
The rationale behind recommending topical tacrolimus as a first-line treatment is that systemic absorption of the drug is trivial. Plus, observational studies of oral tacrolimus in pregnant women who have received a solid organ transplant have shown no increase in congenital malformations.
The task force recommends against the use of topical pimecrolimus (Elidel) or crisaborole (Eucrisa) in pregnancy or lactation due to lack of clinical experience in these settings, Dr. Vestergaard continued.
The task force position is that chlorhexidine and other topical antiseptics – with the notable exception of triclosan – can be used in pregnancy to prevent recurrent skin infections. Aminoglycosides should be avoided, but topical fusidic acid is a reasonable antibiotic for treatment of small areas of clinically infected atopic dermatitis in pregnancy.
Systemic therapies
If disease control is insufficient with topical therapy, it’s appropriate to engage in shared decision-making with the patient regarding systemic treatment. She needs to understand up front that the worldwide overall background stillbirth rate in the general population is about 3%, and that severe congenital malformations are present in up to 6% of all live births.
“You need to inform them that they can have systemic therapy and give birth to a child with congenital defects which have nothing to do with the medication,” noted Dr. Vestergaard.
That said, the task force recommends cyclosporine as the off-label, first-line systemic therapy in pregnancy and lactation when long-term treatment is required. This guidance is based largely upon reassuring evidence in solid organ transplant recipients.
The recommended second-line therapy is systemic corticosteroids, but it’s a qualified recommendation. Dr. Vestergaard and colleagues find that systemic corticosteroid therapy is only rarely needed in pregnant AD patients, and the task force recommendation is to limit the use to less than 2-3 weeks and no more than 0.5 mg/kg per day of prednisone. Dexamethasone is not recommended.
Azathioprine should not be started in pregnancy, according to the task force, but when no other options are available, it may be continued in women already on the drug, albeit at half of the prepregnancy dose.
Dupilumab (Dupixent) is to be avoided in pregnant women with AD until more clinical experience becomes available.
Treatment of prospective fathers with AD
The European task force recommends that topical therapies can be prescribed in prospective fathers without any special concerns. The same is true for systemic corticosteroids. Methotrexate should be halted 3 months before planned pregnancy, as is the case for mycophenolate mofetil (CellCept). Azathioprine is recommended when other options have failed. Cyclosporine is deemed a reasonable option in the treatment of men with severe AD at the time of conception if other treatments have failed; of note, neither the Food and Drug Administration nor the European regulatory agency have issued contraindications for the use of the drug in men who wish to become fathers.
Mycophenolate mofetil carries a theoretical risk of teratogenicity. The European task force recommends that men should use condoms while on the drug and for at least 90 days afterward.
Unplanned pregnancy in women on systemic therapy
The recommended course of action is to immediately stop systemic therapy, intensify appropriate topical therapy in anticipation of worsening AD, and refer the patient to an obstetrician and a teratology information center for an individualized risk assessment. Methotrexate and mycophenolate mofetil are known teratogens.
The full 16-page task force position paper was published shortly before EADV 2019 (J Eur Acad Dermatol Venereol. 2019 Sep;33[9]:1644-59).
The report was developed without commercial sponsorship. Dr. Vestergaard indicated he has received research grants from and/or serves as a consultant to eight pharmaceutical companies.
Jenny Murase, MD, is with the department of dermatology, University of California, San Francisco, and is the director of medical consultative dermatology at the Palo Alto Foundation Medical Group, Mountain View, Calif. She has served on advisory boards for Dermira, Sanofi, and UCB; performed dermatologic consulting for UpToDate and Ferndale, and given nonbranded lectures for disease state management awareness for Regeneron and UCB.
Jenny Murase, MD, is with the department of dermatology, University of California, San Francisco, and is the director of medical consultative dermatology at the Palo Alto Foundation Medical Group, Mountain View, Calif. She has served on advisory boards for Dermira, Sanofi, and UCB; performed dermatologic consulting for UpToDate and Ferndale, and given nonbranded lectures for disease state management awareness for Regeneron and UCB.
Jenny Murase, MD, is with the department of dermatology, University of California, San Francisco, and is the director of medical consultative dermatology at the Palo Alto Foundation Medical Group, Mountain View, Calif. She has served on advisory boards for Dermira, Sanofi, and UCB; performed dermatologic consulting for UpToDate and Ferndale, and given nonbranded lectures for disease state management awareness for Regeneron and UCB.
MADRID – European atopic dermatitis experts have issued formal guidance on a seriously neglected topic: treatment of the disease during pregnancy, breastfeeding, and in men planning to father children.
The impetus for the project was clear: “,” Christian Vestergaard, MD, PhD, declared at a meeting of the European Task Force on Atopic Dermatitis held in conjunction with the annual congress of the European Academy of Dermatology and Venereology.
He presented highlights of the task force’s position paper on the topic, for which he served as first author. The group’s recommendations are based on expert opinion, since randomized clinical trial literature in this area is nonexistent because of ethical concerns. But the task force, comprising a who’s who in European dermatology, drew on a wealth of collective clinical experience in this area.
“We have all of Europe involved in doing this position statement. It’s meant as what we think is proper treatment and what we can say about the different drugs,” explained Dr. Vestergaard, a dermatologist at the University of Aarhus (Denmark).
Most nonobstetricians are intimidated by atopic dermatitis (AD) in pregnancy, and are concerned about the potential for treatment-related harm to the fetus. As a consequence, they are reluctant to recommend anything beyond weak class I topical corticosteroids and emollients. That’s clearly insufficient in light of the vast scope of need, he asserted. After all, AD affects 15%-20% of all children and persists or reappears in adulthood in one out of five of them. Half of those adults are women, many of whom will at some point wish to become pregnant. And many men with AD will eventually want to father children.
A key message from the task force is that untreated AD in pregnancy potentially places the mother and fetus at risk of serious complications, including Staphylococcus aureus infection and eczema herpeticum.
“If you take one thing away from our position paper, it’s that you can use class II or III topical corticosteroids in pregnant women as first-line therapy,” Dr. Vestergaard said.
This stance contradicts a longstanding widely held concern that topical steroids in pregnancy might increase the risk of facial cleft in the offspring, a worry that has been convincingly debunked in a Cochrane systematic review of 14 studies including more than 1.6 million pregnancies. The report concluded there was no association between topical corticosteroids of any potency with preterm delivery, birth defects, or low Apgar scores (Cochrane Database Syst Rev. 2015 Oct 26. doi: 10.1002/14651858.CD007346.pub3).
The task force recommends that if class II or III topical corticosteroid use in pregnancy exceeds 200 g/month, it’s worth considering add-on UV therapy, with narrow band UVB-311 nm as the regimen of choice; it can be used liberally. UV therapy with psoralens is not advised because of a theoretical risk of mutagenicity.
Product labeling for the topical calcineurin inhibitors declares that the agents should not be used during pregnancy. However, the European task force position paper takes issue with that and declares that topical tacrolimus (Protopic) can be considered an off-label first-line therapy in pregnant women with an insufficient response to liberal use of emollients. The same holds true for breastfeeding patients with AD. Just as when topical corticosteroids are used in the nipple area, topical tacrolimus should be applied after nursing, and the nipple area should be gently cleaned before nursing.
The rationale behind recommending topical tacrolimus as a first-line treatment is that systemic absorption of the drug is trivial. Plus, observational studies of oral tacrolimus in pregnant women who have received a solid organ transplant have shown no increase in congenital malformations.
The task force recommends against the use of topical pimecrolimus (Elidel) or crisaborole (Eucrisa) in pregnancy or lactation due to lack of clinical experience in these settings, Dr. Vestergaard continued.
The task force position is that chlorhexidine and other topical antiseptics – with the notable exception of triclosan – can be used in pregnancy to prevent recurrent skin infections. Aminoglycosides should be avoided, but topical fusidic acid is a reasonable antibiotic for treatment of small areas of clinically infected atopic dermatitis in pregnancy.
Systemic therapies
If disease control is insufficient with topical therapy, it’s appropriate to engage in shared decision-making with the patient regarding systemic treatment. She needs to understand up front that the worldwide overall background stillbirth rate in the general population is about 3%, and that severe congenital malformations are present in up to 6% of all live births.
“You need to inform them that they can have systemic therapy and give birth to a child with congenital defects which have nothing to do with the medication,” noted Dr. Vestergaard.
That said, the task force recommends cyclosporine as the off-label, first-line systemic therapy in pregnancy and lactation when long-term treatment is required. This guidance is based largely upon reassuring evidence in solid organ transplant recipients.
The recommended second-line therapy is systemic corticosteroids, but it’s a qualified recommendation. Dr. Vestergaard and colleagues find that systemic corticosteroid therapy is only rarely needed in pregnant AD patients, and the task force recommendation is to limit the use to less than 2-3 weeks and no more than 0.5 mg/kg per day of prednisone. Dexamethasone is not recommended.
Azathioprine should not be started in pregnancy, according to the task force, but when no other options are available, it may be continued in women already on the drug, albeit at half of the prepregnancy dose.
Dupilumab (Dupixent) is to be avoided in pregnant women with AD until more clinical experience becomes available.
Treatment of prospective fathers with AD
The European task force recommends that topical therapies can be prescribed in prospective fathers without any special concerns. The same is true for systemic corticosteroids. Methotrexate should be halted 3 months before planned pregnancy, as is the case for mycophenolate mofetil (CellCept). Azathioprine is recommended when other options have failed. Cyclosporine is deemed a reasonable option in the treatment of men with severe AD at the time of conception if other treatments have failed; of note, neither the Food and Drug Administration nor the European regulatory agency have issued contraindications for the use of the drug in men who wish to become fathers.
Mycophenolate mofetil carries a theoretical risk of teratogenicity. The European task force recommends that men should use condoms while on the drug and for at least 90 days afterward.
Unplanned pregnancy in women on systemic therapy
The recommended course of action is to immediately stop systemic therapy, intensify appropriate topical therapy in anticipation of worsening AD, and refer the patient to an obstetrician and a teratology information center for an individualized risk assessment. Methotrexate and mycophenolate mofetil are known teratogens.
The full 16-page task force position paper was published shortly before EADV 2019 (J Eur Acad Dermatol Venereol. 2019 Sep;33[9]:1644-59).
The report was developed without commercial sponsorship. Dr. Vestergaard indicated he has received research grants from and/or serves as a consultant to eight pharmaceutical companies.
MADRID – European atopic dermatitis experts have issued formal guidance on a seriously neglected topic: treatment of the disease during pregnancy, breastfeeding, and in men planning to father children.
The impetus for the project was clear: “,” Christian Vestergaard, MD, PhD, declared at a meeting of the European Task Force on Atopic Dermatitis held in conjunction with the annual congress of the European Academy of Dermatology and Venereology.
He presented highlights of the task force’s position paper on the topic, for which he served as first author. The group’s recommendations are based on expert opinion, since randomized clinical trial literature in this area is nonexistent because of ethical concerns. But the task force, comprising a who’s who in European dermatology, drew on a wealth of collective clinical experience in this area.
“We have all of Europe involved in doing this position statement. It’s meant as what we think is proper treatment and what we can say about the different drugs,” explained Dr. Vestergaard, a dermatologist at the University of Aarhus (Denmark).
Most nonobstetricians are intimidated by atopic dermatitis (AD) in pregnancy, and are concerned about the potential for treatment-related harm to the fetus. As a consequence, they are reluctant to recommend anything beyond weak class I topical corticosteroids and emollients. That’s clearly insufficient in light of the vast scope of need, he asserted. After all, AD affects 15%-20% of all children and persists or reappears in adulthood in one out of five of them. Half of those adults are women, many of whom will at some point wish to become pregnant. And many men with AD will eventually want to father children.
A key message from the task force is that untreated AD in pregnancy potentially places the mother and fetus at risk of serious complications, including Staphylococcus aureus infection and eczema herpeticum.
“If you take one thing away from our position paper, it’s that you can use class II or III topical corticosteroids in pregnant women as first-line therapy,” Dr. Vestergaard said.
This stance contradicts a longstanding widely held concern that topical steroids in pregnancy might increase the risk of facial cleft in the offspring, a worry that has been convincingly debunked in a Cochrane systematic review of 14 studies including more than 1.6 million pregnancies. The report concluded there was no association between topical corticosteroids of any potency with preterm delivery, birth defects, or low Apgar scores (Cochrane Database Syst Rev. 2015 Oct 26. doi: 10.1002/14651858.CD007346.pub3).
The task force recommends that if class II or III topical corticosteroid use in pregnancy exceeds 200 g/month, it’s worth considering add-on UV therapy, with narrow band UVB-311 nm as the regimen of choice; it can be used liberally. UV therapy with psoralens is not advised because of a theoretical risk of mutagenicity.
Product labeling for the topical calcineurin inhibitors declares that the agents should not be used during pregnancy. However, the European task force position paper takes issue with that and declares that topical tacrolimus (Protopic) can be considered an off-label first-line therapy in pregnant women with an insufficient response to liberal use of emollients. The same holds true for breastfeeding patients with AD. Just as when topical corticosteroids are used in the nipple area, topical tacrolimus should be applied after nursing, and the nipple area should be gently cleaned before nursing.
The rationale behind recommending topical tacrolimus as a first-line treatment is that systemic absorption of the drug is trivial. Plus, observational studies of oral tacrolimus in pregnant women who have received a solid organ transplant have shown no increase in congenital malformations.
The task force recommends against the use of topical pimecrolimus (Elidel) or crisaborole (Eucrisa) in pregnancy or lactation due to lack of clinical experience in these settings, Dr. Vestergaard continued.
The task force position is that chlorhexidine and other topical antiseptics – with the notable exception of triclosan – can be used in pregnancy to prevent recurrent skin infections. Aminoglycosides should be avoided, but topical fusidic acid is a reasonable antibiotic for treatment of small areas of clinically infected atopic dermatitis in pregnancy.
Systemic therapies
If disease control is insufficient with topical therapy, it’s appropriate to engage in shared decision-making with the patient regarding systemic treatment. She needs to understand up front that the worldwide overall background stillbirth rate in the general population is about 3%, and that severe congenital malformations are present in up to 6% of all live births.
“You need to inform them that they can have systemic therapy and give birth to a child with congenital defects which have nothing to do with the medication,” noted Dr. Vestergaard.
That said, the task force recommends cyclosporine as the off-label, first-line systemic therapy in pregnancy and lactation when long-term treatment is required. This guidance is based largely upon reassuring evidence in solid organ transplant recipients.
The recommended second-line therapy is systemic corticosteroids, but it’s a qualified recommendation. Dr. Vestergaard and colleagues find that systemic corticosteroid therapy is only rarely needed in pregnant AD patients, and the task force recommendation is to limit the use to less than 2-3 weeks and no more than 0.5 mg/kg per day of prednisone. Dexamethasone is not recommended.
Azathioprine should not be started in pregnancy, according to the task force, but when no other options are available, it may be continued in women already on the drug, albeit at half of the prepregnancy dose.
Dupilumab (Dupixent) is to be avoided in pregnant women with AD until more clinical experience becomes available.
Treatment of prospective fathers with AD
The European task force recommends that topical therapies can be prescribed in prospective fathers without any special concerns. The same is true for systemic corticosteroids. Methotrexate should be halted 3 months before planned pregnancy, as is the case for mycophenolate mofetil (CellCept). Azathioprine is recommended when other options have failed. Cyclosporine is deemed a reasonable option in the treatment of men with severe AD at the time of conception if other treatments have failed; of note, neither the Food and Drug Administration nor the European regulatory agency have issued contraindications for the use of the drug in men who wish to become fathers.
Mycophenolate mofetil carries a theoretical risk of teratogenicity. The European task force recommends that men should use condoms while on the drug and for at least 90 days afterward.
Unplanned pregnancy in women on systemic therapy
The recommended course of action is to immediately stop systemic therapy, intensify appropriate topical therapy in anticipation of worsening AD, and refer the patient to an obstetrician and a teratology information center for an individualized risk assessment. Methotrexate and mycophenolate mofetil are known teratogens.
The full 16-page task force position paper was published shortly before EADV 2019 (J Eur Acad Dermatol Venereol. 2019 Sep;33[9]:1644-59).
The report was developed without commercial sponsorship. Dr. Vestergaard indicated he has received research grants from and/or serves as a consultant to eight pharmaceutical companies.
REPORTING FROM THE EADV CONGRESS
Ideal management of RA in pregnancy improves outcomes
Women whose rheumatoid arthritis is carefully managed before and during pregnancy have a significantly lower risk of adverse pregnancy outcomes, including miscarriage or perinatal death, new research suggests.
A study published in Arthritis Care & Research presents the outcomes of a retrospective, observational study examining health care data from 443 first pregnancies in women with RA and 6,097 women without the disease.
First author Alessandra Bortoluzzi, MD, PhD, from the Rheumatology Unit at the University of Ferrara (Italy) and coauthors looked at seven diagnostic, therapeutic, and follow-up health care quality indicators during the prepregnancy and perinatal period. They included having at least one blood test in the 18 months before conception and during pregnancy, preconception musculoskeletal imaging, no exposure or wash-out from teratogenic drugs, and no exposure to biologic drugs between conception and delivery or end of pregnancy.
An ideal clinical pathway included at least one element from each of the diagnostic, therapeutic, and prenatal follow-up quality indicators.
Overall, women with RA had a significantly higher rate of thyroid diseases, adverse pregnancy outcomes, and miscarriage or perinatal death when compared with controls. However, those who followed the ideal clinical pathway for management of their disease during pregnancy had a 40% lower odds of adverse pregnancy outcomes (odds ratio, 0.60; 95% confidence interval, 0.39-0.94) and a 60% lower odds of miscarriage or perinatal death (OR, 0.40; 95% CI, 0.24-0.69) in comparison with women with RA who were not managed to the same standard. The researchers adjusted both comparisons for age, Charlson comorbidity index, and thyroid diseases.
Women with RA who met diagnostic, therapeutic, and prenatal follow-up quality indicators showed no significant differences from the general population in terms of the risk of adverse pregnancy outcomes, miscarriage, or perinatal death after adjusting for hypertension in addition to the same variables as before.
When researchers looked at some of the individual health care quality indicators, they found that testing for antiphospholipid (aPL) antibodies within 18 months of conception or pregnancy was associated with a 44% lower rate of adverse pregnancy outcomes. Similarly, antinuclear antibody or anti–extractable nuclear antigen antibody testing was associated with 36% lower odds of adverse pregnancy outcomes.
Dr. Bortoluzzi and her coauthors wrote that their findings pointed to the value of testing for aPL antibodies in women with RA who wish to get pregnant.
“In fact, despite the absence of formal recommendation or validated health care quality indicators focused on stratification of preconceptional obstetric risk in patients with RA, we started from the basic and universally accepted assumption that aPL antibodies are pathogenic autoantibodies and therefore recognized risk factors for adverse pregnancy outcome,” they wrote.
Women with RA who had either no exposure to methotrexate or leflunomide or who had a washout period from 6 months prior to conception had 72% lower odds of adverse pregnancy outcomes.
The authors also looked at the effects of drugs such as aspirin, glucocorticoids, and low-molecular-weight heparin that are used during pregnancy. They found that the relative risk of adverse pregnancy outcomes was 40% higher in women with RA who were taking glucocorticoids, compared with those with the disease but not taking that type of medication. However, low-molecular-weight heparin use was associated with an 80% lower relative risk of miscarriage or perinatal death in comparison with those not taking it. Researchers saw no significant effects of aspirin or conventional synthetic disease-modifying antirheumatic drugs on either adverse pregnancy outcomes or the risk of miscarriage or perinatal death.
“This reinforces the importance of adjustment of therapy for RA before conception and throughout pregnancy, because medication use could affect pregnancy course not only influencing maternal disease activity but also the gestational outcome,” the authors wrote. “Although this is a study conducted on administrative data, we can hypothesize that exposure to therapy represents a marker of high RA disease activity and severity. In our setting, it is possible that, the more active the disease, the greater the probability of being included in the ideal clinical pathway, but in any case, this resulted in a lower odds ratio of adverse pregnancy outcome and miscarriage/perinatal death.”
The study was supported by the Italian Society for Rheumatology. No conflicts of interest were declared.
SOURCE: Bortoluzzi A et al. Arthritis Care Res. 2020 Jan 8. doi: 10.1002/ACR.24116.
Women whose rheumatoid arthritis is carefully managed before and during pregnancy have a significantly lower risk of adverse pregnancy outcomes, including miscarriage or perinatal death, new research suggests.
A study published in Arthritis Care & Research presents the outcomes of a retrospective, observational study examining health care data from 443 first pregnancies in women with RA and 6,097 women without the disease.
First author Alessandra Bortoluzzi, MD, PhD, from the Rheumatology Unit at the University of Ferrara (Italy) and coauthors looked at seven diagnostic, therapeutic, and follow-up health care quality indicators during the prepregnancy and perinatal period. They included having at least one blood test in the 18 months before conception and during pregnancy, preconception musculoskeletal imaging, no exposure or wash-out from teratogenic drugs, and no exposure to biologic drugs between conception and delivery or end of pregnancy.
An ideal clinical pathway included at least one element from each of the diagnostic, therapeutic, and prenatal follow-up quality indicators.
Overall, women with RA had a significantly higher rate of thyroid diseases, adverse pregnancy outcomes, and miscarriage or perinatal death when compared with controls. However, those who followed the ideal clinical pathway for management of their disease during pregnancy had a 40% lower odds of adverse pregnancy outcomes (odds ratio, 0.60; 95% confidence interval, 0.39-0.94) and a 60% lower odds of miscarriage or perinatal death (OR, 0.40; 95% CI, 0.24-0.69) in comparison with women with RA who were not managed to the same standard. The researchers adjusted both comparisons for age, Charlson comorbidity index, and thyroid diseases.
Women with RA who met diagnostic, therapeutic, and prenatal follow-up quality indicators showed no significant differences from the general population in terms of the risk of adverse pregnancy outcomes, miscarriage, or perinatal death after adjusting for hypertension in addition to the same variables as before.
When researchers looked at some of the individual health care quality indicators, they found that testing for antiphospholipid (aPL) antibodies within 18 months of conception or pregnancy was associated with a 44% lower rate of adverse pregnancy outcomes. Similarly, antinuclear antibody or anti–extractable nuclear antigen antibody testing was associated with 36% lower odds of adverse pregnancy outcomes.
Dr. Bortoluzzi and her coauthors wrote that their findings pointed to the value of testing for aPL antibodies in women with RA who wish to get pregnant.
“In fact, despite the absence of formal recommendation or validated health care quality indicators focused on stratification of preconceptional obstetric risk in patients with RA, we started from the basic and universally accepted assumption that aPL antibodies are pathogenic autoantibodies and therefore recognized risk factors for adverse pregnancy outcome,” they wrote.
Women with RA who had either no exposure to methotrexate or leflunomide or who had a washout period from 6 months prior to conception had 72% lower odds of adverse pregnancy outcomes.
The authors also looked at the effects of drugs such as aspirin, glucocorticoids, and low-molecular-weight heparin that are used during pregnancy. They found that the relative risk of adverse pregnancy outcomes was 40% higher in women with RA who were taking glucocorticoids, compared with those with the disease but not taking that type of medication. However, low-molecular-weight heparin use was associated with an 80% lower relative risk of miscarriage or perinatal death in comparison with those not taking it. Researchers saw no significant effects of aspirin or conventional synthetic disease-modifying antirheumatic drugs on either adverse pregnancy outcomes or the risk of miscarriage or perinatal death.
“This reinforces the importance of adjustment of therapy for RA before conception and throughout pregnancy, because medication use could affect pregnancy course not only influencing maternal disease activity but also the gestational outcome,” the authors wrote. “Although this is a study conducted on administrative data, we can hypothesize that exposure to therapy represents a marker of high RA disease activity and severity. In our setting, it is possible that, the more active the disease, the greater the probability of being included in the ideal clinical pathway, but in any case, this resulted in a lower odds ratio of adverse pregnancy outcome and miscarriage/perinatal death.”
The study was supported by the Italian Society for Rheumatology. No conflicts of interest were declared.
SOURCE: Bortoluzzi A et al. Arthritis Care Res. 2020 Jan 8. doi: 10.1002/ACR.24116.
Women whose rheumatoid arthritis is carefully managed before and during pregnancy have a significantly lower risk of adverse pregnancy outcomes, including miscarriage or perinatal death, new research suggests.
A study published in Arthritis Care & Research presents the outcomes of a retrospective, observational study examining health care data from 443 first pregnancies in women with RA and 6,097 women without the disease.
First author Alessandra Bortoluzzi, MD, PhD, from the Rheumatology Unit at the University of Ferrara (Italy) and coauthors looked at seven diagnostic, therapeutic, and follow-up health care quality indicators during the prepregnancy and perinatal period. They included having at least one blood test in the 18 months before conception and during pregnancy, preconception musculoskeletal imaging, no exposure or wash-out from teratogenic drugs, and no exposure to biologic drugs between conception and delivery or end of pregnancy.
An ideal clinical pathway included at least one element from each of the diagnostic, therapeutic, and prenatal follow-up quality indicators.
Overall, women with RA had a significantly higher rate of thyroid diseases, adverse pregnancy outcomes, and miscarriage or perinatal death when compared with controls. However, those who followed the ideal clinical pathway for management of their disease during pregnancy had a 40% lower odds of adverse pregnancy outcomes (odds ratio, 0.60; 95% confidence interval, 0.39-0.94) and a 60% lower odds of miscarriage or perinatal death (OR, 0.40; 95% CI, 0.24-0.69) in comparison with women with RA who were not managed to the same standard. The researchers adjusted both comparisons for age, Charlson comorbidity index, and thyroid diseases.
Women with RA who met diagnostic, therapeutic, and prenatal follow-up quality indicators showed no significant differences from the general population in terms of the risk of adverse pregnancy outcomes, miscarriage, or perinatal death after adjusting for hypertension in addition to the same variables as before.
When researchers looked at some of the individual health care quality indicators, they found that testing for antiphospholipid (aPL) antibodies within 18 months of conception or pregnancy was associated with a 44% lower rate of adverse pregnancy outcomes. Similarly, antinuclear antibody or anti–extractable nuclear antigen antibody testing was associated with 36% lower odds of adverse pregnancy outcomes.
Dr. Bortoluzzi and her coauthors wrote that their findings pointed to the value of testing for aPL antibodies in women with RA who wish to get pregnant.
“In fact, despite the absence of formal recommendation or validated health care quality indicators focused on stratification of preconceptional obstetric risk in patients with RA, we started from the basic and universally accepted assumption that aPL antibodies are pathogenic autoantibodies and therefore recognized risk factors for adverse pregnancy outcome,” they wrote.
Women with RA who had either no exposure to methotrexate or leflunomide or who had a washout period from 6 months prior to conception had 72% lower odds of adverse pregnancy outcomes.
The authors also looked at the effects of drugs such as aspirin, glucocorticoids, and low-molecular-weight heparin that are used during pregnancy. They found that the relative risk of adverse pregnancy outcomes was 40% higher in women with RA who were taking glucocorticoids, compared with those with the disease but not taking that type of medication. However, low-molecular-weight heparin use was associated with an 80% lower relative risk of miscarriage or perinatal death in comparison with those not taking it. Researchers saw no significant effects of aspirin or conventional synthetic disease-modifying antirheumatic drugs on either adverse pregnancy outcomes or the risk of miscarriage or perinatal death.
“This reinforces the importance of adjustment of therapy for RA before conception and throughout pregnancy, because medication use could affect pregnancy course not only influencing maternal disease activity but also the gestational outcome,” the authors wrote. “Although this is a study conducted on administrative data, we can hypothesize that exposure to therapy represents a marker of high RA disease activity and severity. In our setting, it is possible that, the more active the disease, the greater the probability of being included in the ideal clinical pathway, but in any case, this resulted in a lower odds ratio of adverse pregnancy outcome and miscarriage/perinatal death.”
The study was supported by the Italian Society for Rheumatology. No conflicts of interest were declared.
SOURCE: Bortoluzzi A et al. Arthritis Care Res. 2020 Jan 8. doi: 10.1002/ACR.24116.
FROM ARTHRITIS CARE & RESEARCH
Key clinical point: Well-managed rheumatoid arthritis during preconception and pregnancy is associated with improved pregnancy outcomes.
Major finding: Women who adhered to an ideal clinical pathway for their RA had significantly lower risk of adverse pregnancy outcomes and miscarriage and/or perinatal death.
Study details: Retrospective, observational study of 443 first pregnancies in women with RA and 6,097 women without.
Disclosures: The study was supported by the Italian Society for Rheumatology. No conflicts of interest were declared.
Source: Bortoluzzi A et al. Arthritis Care Res. 2020 Jan 8. doi: 10.1002/ACR.24116.
Barrett’s esophagus risk factors vary by patient sex
In males but not females, hyperinsulinemia, insulin resistance, and metabolic syndrome are independently associated with a higher risk of Barrett’s esophagus (BE), based on a recent case-control study.
These findings offer some insight into why men have higher rates of BE and esophageal adenocarcinoma than do women, reported lead author Bradley J. Kendall, MBBS, PhD, of the University of Queensland in Brisbane, Australia, and colleagues.
“Esophageal adenocarcinoma (EA) and its precursor lesion, Barrett’s esophagus (BE), are more common in males than females,” the investigators wrote in the Journal of Clinical Gastroenterology. “In contrast, gastroesophageal reflux (GER), the major risk factor for BE and EA occurs at similar frequencies in both sexes. In addition, 10% to 20% of BE and EA cases report no history of GER symptoms. This suggests that non-GER factors are important in the development of BE and EA.”
To examine risk factors more closely, the investigators enrolled 227 patients with BE and 241 age- and sex-matched controls. Data were drawn from self-reported questionnaires, interviews, blood pressure readings, and anthropometric measurements, the latter of which included weight, height, and waist circumference. These patient characteristics were supplemented with fasted blood assays. The investigators looked for associations between BE and insulin level, Homeostatic Model Assessment of Insulin Resistance, metabolic syndrome, type 2 diabetes mellitus, insulin-like growth factors, and interleukin-6 (IL-6).
Across the entire population, independent associations were detected between BE and hyperinsulinemia (highest vs. lowest tertile; odds ratio, 1.9; P = .003), insulin resistance (OR, 1.9; P = .006) and metabolic syndrome (OR, 1.8; P = .004). For those with metabolic syndrome, risk of BE increased by 20% for each additional syndrome criterion (P = .02).
When stratifying by sex, however, all of the above risk factors remained statistically significant in men, but not in women. For male patients, compared with the population as a whole, risks were relatively higher for all three factors: hyperinsulinemia (OR, 2.1; P = .007), insulin resistance (OR, 2.1; P = .01), and metabolic syndrome (OR, 2.3; P = .001). Similarly, for men with metabolic syndrome, each additional syndrome criterion increased risk of BE by 40% (P = .005).
Regardless of sex stratification, the other evaluated characteristics (type 2 diabetes mellitus, insulin-like growth factors, and IL-6) were not associated with BE risk.
The investigators offered some possible mechanistic explanations for their findings.
“Hyperinsulinemia ... can result in increased insulin signaling, increased cellular proliferation, reduced apoptosis, oncogenic pathway activation, and enhanced cellular invasion,” they wrote. “In addition, abdominal obesity, which is the driver of these disorders of insulin homeostasis, also alters adipocytokine profiles and induces a chronic systemic inflammatory state. The inflammatory state can result in oncoprotein activation, angiogenesis, cellular proliferation, apoptosis, and metastasis.”
Concerning the difference between sexes, the investigators pointed to patterns of abdominal obesity. “[A]bdominal obesity and the associated metabolic sequelae are more common in males than females,” they wrote. “These observations give rise to the notion that the metabolic syndrome potentiates the inflammatory effects of the gastric refluxate in the distal esophagus and this may play a role in the male predominance of BE and EA.”
While these findings offer insight into the underlying processes that precipitate BE, the investigators suggested that more research is needed.
“The interactions of obesity hormones, GER, and the cells of the esophageal mucosa warrant further investigation,” they concluded.
The study was funded by the Queensland Cancer Fund, Queensland Government Smart State Fund, and the Princess Alexandra Hospital Research Foundation, and others. The investigators reported no disclosures.
SOURCE: Kendall BJ et al. J Clin Gastroenterol. 2019 Dec 24. doi: 10.1097/MCG.0000000000001307.
In males but not females, hyperinsulinemia, insulin resistance, and metabolic syndrome are independently associated with a higher risk of Barrett’s esophagus (BE), based on a recent case-control study.
These findings offer some insight into why men have higher rates of BE and esophageal adenocarcinoma than do women, reported lead author Bradley J. Kendall, MBBS, PhD, of the University of Queensland in Brisbane, Australia, and colleagues.
“Esophageal adenocarcinoma (EA) and its precursor lesion, Barrett’s esophagus (BE), are more common in males than females,” the investigators wrote in the Journal of Clinical Gastroenterology. “In contrast, gastroesophageal reflux (GER), the major risk factor for BE and EA occurs at similar frequencies in both sexes. In addition, 10% to 20% of BE and EA cases report no history of GER symptoms. This suggests that non-GER factors are important in the development of BE and EA.”
To examine risk factors more closely, the investigators enrolled 227 patients with BE and 241 age- and sex-matched controls. Data were drawn from self-reported questionnaires, interviews, blood pressure readings, and anthropometric measurements, the latter of which included weight, height, and waist circumference. These patient characteristics were supplemented with fasted blood assays. The investigators looked for associations between BE and insulin level, Homeostatic Model Assessment of Insulin Resistance, metabolic syndrome, type 2 diabetes mellitus, insulin-like growth factors, and interleukin-6 (IL-6).
Across the entire population, independent associations were detected between BE and hyperinsulinemia (highest vs. lowest tertile; odds ratio, 1.9; P = .003), insulin resistance (OR, 1.9; P = .006) and metabolic syndrome (OR, 1.8; P = .004). For those with metabolic syndrome, risk of BE increased by 20% for each additional syndrome criterion (P = .02).
When stratifying by sex, however, all of the above risk factors remained statistically significant in men, but not in women. For male patients, compared with the population as a whole, risks were relatively higher for all three factors: hyperinsulinemia (OR, 2.1; P = .007), insulin resistance (OR, 2.1; P = .01), and metabolic syndrome (OR, 2.3; P = .001). Similarly, for men with metabolic syndrome, each additional syndrome criterion increased risk of BE by 40% (P = .005).
Regardless of sex stratification, the other evaluated characteristics (type 2 diabetes mellitus, insulin-like growth factors, and IL-6) were not associated with BE risk.
The investigators offered some possible mechanistic explanations for their findings.
“Hyperinsulinemia ... can result in increased insulin signaling, increased cellular proliferation, reduced apoptosis, oncogenic pathway activation, and enhanced cellular invasion,” they wrote. “In addition, abdominal obesity, which is the driver of these disorders of insulin homeostasis, also alters adipocytokine profiles and induces a chronic systemic inflammatory state. The inflammatory state can result in oncoprotein activation, angiogenesis, cellular proliferation, apoptosis, and metastasis.”
Concerning the difference between sexes, the investigators pointed to patterns of abdominal obesity. “[A]bdominal obesity and the associated metabolic sequelae are more common in males than females,” they wrote. “These observations give rise to the notion that the metabolic syndrome potentiates the inflammatory effects of the gastric refluxate in the distal esophagus and this may play a role in the male predominance of BE and EA.”
While these findings offer insight into the underlying processes that precipitate BE, the investigators suggested that more research is needed.
“The interactions of obesity hormones, GER, and the cells of the esophageal mucosa warrant further investigation,” they concluded.
The study was funded by the Queensland Cancer Fund, Queensland Government Smart State Fund, and the Princess Alexandra Hospital Research Foundation, and others. The investigators reported no disclosures.
SOURCE: Kendall BJ et al. J Clin Gastroenterol. 2019 Dec 24. doi: 10.1097/MCG.0000000000001307.
In males but not females, hyperinsulinemia, insulin resistance, and metabolic syndrome are independently associated with a higher risk of Barrett’s esophagus (BE), based on a recent case-control study.
These findings offer some insight into why men have higher rates of BE and esophageal adenocarcinoma than do women, reported lead author Bradley J. Kendall, MBBS, PhD, of the University of Queensland in Brisbane, Australia, and colleagues.
“Esophageal adenocarcinoma (EA) and its precursor lesion, Barrett’s esophagus (BE), are more common in males than females,” the investigators wrote in the Journal of Clinical Gastroenterology. “In contrast, gastroesophageal reflux (GER), the major risk factor for BE and EA occurs at similar frequencies in both sexes. In addition, 10% to 20% of BE and EA cases report no history of GER symptoms. This suggests that non-GER factors are important in the development of BE and EA.”
To examine risk factors more closely, the investigators enrolled 227 patients with BE and 241 age- and sex-matched controls. Data were drawn from self-reported questionnaires, interviews, blood pressure readings, and anthropometric measurements, the latter of which included weight, height, and waist circumference. These patient characteristics were supplemented with fasted blood assays. The investigators looked for associations between BE and insulin level, Homeostatic Model Assessment of Insulin Resistance, metabolic syndrome, type 2 diabetes mellitus, insulin-like growth factors, and interleukin-6 (IL-6).
Across the entire population, independent associations were detected between BE and hyperinsulinemia (highest vs. lowest tertile; odds ratio, 1.9; P = .003), insulin resistance (OR, 1.9; P = .006) and metabolic syndrome (OR, 1.8; P = .004). For those with metabolic syndrome, risk of BE increased by 20% for each additional syndrome criterion (P = .02).
When stratifying by sex, however, all of the above risk factors remained statistically significant in men, but not in women. For male patients, compared with the population as a whole, risks were relatively higher for all three factors: hyperinsulinemia (OR, 2.1; P = .007), insulin resistance (OR, 2.1; P = .01), and metabolic syndrome (OR, 2.3; P = .001). Similarly, for men with metabolic syndrome, each additional syndrome criterion increased risk of BE by 40% (P = .005).
Regardless of sex stratification, the other evaluated characteristics (type 2 diabetes mellitus, insulin-like growth factors, and IL-6) were not associated with BE risk.
The investigators offered some possible mechanistic explanations for their findings.
“Hyperinsulinemia ... can result in increased insulin signaling, increased cellular proliferation, reduced apoptosis, oncogenic pathway activation, and enhanced cellular invasion,” they wrote. “In addition, abdominal obesity, which is the driver of these disorders of insulin homeostasis, also alters adipocytokine profiles and induces a chronic systemic inflammatory state. The inflammatory state can result in oncoprotein activation, angiogenesis, cellular proliferation, apoptosis, and metastasis.”
Concerning the difference between sexes, the investigators pointed to patterns of abdominal obesity. “[A]bdominal obesity and the associated metabolic sequelae are more common in males than females,” they wrote. “These observations give rise to the notion that the metabolic syndrome potentiates the inflammatory effects of the gastric refluxate in the distal esophagus and this may play a role in the male predominance of BE and EA.”
While these findings offer insight into the underlying processes that precipitate BE, the investigators suggested that more research is needed.
“The interactions of obesity hormones, GER, and the cells of the esophageal mucosa warrant further investigation,” they concluded.
The study was funded by the Queensland Cancer Fund, Queensland Government Smart State Fund, and the Princess Alexandra Hospital Research Foundation, and others. The investigators reported no disclosures.
SOURCE: Kendall BJ et al. J Clin Gastroenterol. 2019 Dec 24. doi: 10.1097/MCG.0000000000001307.
FROM JOURNAL OF CLINICAL GASTROENTEROLOGY