User login
Myocardial infarction in women younger than 50: Lessons to learn
Young women (under 50) are increasingly having heart attacks without doctors really knowing why. This is where the Young Women Presenting Acute Myocardial Infarction in France (WAMIF) study comes in, the results of which were presented in an e-poster at the annual congress of the European Society of Cardiology by Stéphane Manzo-Silberman, MD, Institute of Cardiology, Pitié-Salpétrière, Paris. The results (yet to be published) fight several of the preconceived ideas on the topic, Dr. Manzo-Silberman commented in an interview.
Significantly higher hospital death rates in women
“Cardiovascular disease is the main cause of death in women, killing seven times more than breast cancer,” notes Dr. Manzo-Silberman. The hospital death rate is significantly higher in women and, despite going down, is significantly higher than in men (more than double), particularly in women under 50. What’s more, in addition to the typical risk factors, women present specific risk factors related to hormone changes, high-risk inflammatory profiles, and thrombophilia.”
The WAMIF study was designed to determine the clinical, biological, and morphological features linked to hospital mortality after 12 months in women under 50. The prospective, observational study included all women in this age range from 30 sites in France between May 2017 and June 2019.
90% with retrosternal chest pain
The age of the 314 women enrolled was 44.9 years on average. Nearly two-thirds (192) presented with ST-segment elevation myocardial infarction and the other 122 without. In terms of symptoms, 91.6% of these women presented with typical chest pain, and 59.7% had related symptoms.
“With more than 90% having retrosternal pain, the idea that myocardial infarction presents with atypical symptoms in women has been widely challenged, despite the fact that more than half present with related symptoms and it isn’t known in which order these symptoms occur, Dr. Manzo-Silberman said in an interview. But what we can say is that if at any point a young woman mentions chest pain, even when occurring as part of several other symptoms, MI must be deemed a possibility until it has been ruled out.”
The risk profile revealed that 75.5% were smokers, 35% had a family history of heart disease, 33% had pregnancy complications, and 55% had recently experienced a stressful situation. The analysis also showed that cannabis use and oral contraception were primary risk factors in women younger than 35.
“With regard to risk factors, when designing this study we expected that lots of these young women would have largely atypical autoimmune conditions, with high levels of inflammation. We looked for everything, but this was not actually the case. Instead, we found very many women to have classic risk factors; three-quarters were smokers, a modifiable risk factor, which can largely be prevented. The other aspect concerns contraception, and it’s why I insist that gynecologists must be involved insofar as they must inform their patients how to manage their risk factors and tweak their contraception.”
Coronary angiography findings showed that only 1% received a normal result, 29.3% had vessel damage, and 14.6% had aortic dissection. “We were surprised again here because we expected that with young women we would see lots of heart attacks without obstruction, [in other words] normal coronary arteries, atypical forms of MI,” commented Dr. Manzo-Silberman. “In fact, most presented with atheroma, often obstructive lesions, or even triple-vessel disease, in nearly a third of the cohort. So that’s another misconception dispelled – we can’t just think that because a woman is young, nothing will be found. Coronary catheterization should be considered, and the diagnostic process should be completed in full.”
After 1 year, there had been two cancer-related deaths and 25 patients had undergone several angioplasty procedures. Nevertheless, 90.4% had not experienced any type of CV event, and 72% had not even had any symptoms.
“The final surprise was prognosis,” he said. “Previous studies, especially some authored by Viola Vaccarino, MD, PhD, showed an excess hospital rate in women and we had expected this to be the case here, but no hospital deaths were recorded. However, not far off 10% of women attended (at least once) the emergency department in the year following for recurrent chest pain which was not ischemic – ECG normal, troponin normal – so something was missing in their education as a patient.”
“So, there are improvements to be made in terms of secondary prevention, follow-up, and in the education of these young female patients who have experienced the major event that is a myocardial infarction,” concluded Dr. Manzo-Silberman.
This content was originally published on Medscape French edition.
Young women (under 50) are increasingly having heart attacks without doctors really knowing why. This is where the Young Women Presenting Acute Myocardial Infarction in France (WAMIF) study comes in, the results of which were presented in an e-poster at the annual congress of the European Society of Cardiology by Stéphane Manzo-Silberman, MD, Institute of Cardiology, Pitié-Salpétrière, Paris. The results (yet to be published) fight several of the preconceived ideas on the topic, Dr. Manzo-Silberman commented in an interview.
Significantly higher hospital death rates in women
“Cardiovascular disease is the main cause of death in women, killing seven times more than breast cancer,” notes Dr. Manzo-Silberman. The hospital death rate is significantly higher in women and, despite going down, is significantly higher than in men (more than double), particularly in women under 50. What’s more, in addition to the typical risk factors, women present specific risk factors related to hormone changes, high-risk inflammatory profiles, and thrombophilia.”
The WAMIF study was designed to determine the clinical, biological, and morphological features linked to hospital mortality after 12 months in women under 50. The prospective, observational study included all women in this age range from 30 sites in France between May 2017 and June 2019.
90% with retrosternal chest pain
The age of the 314 women enrolled was 44.9 years on average. Nearly two-thirds (192) presented with ST-segment elevation myocardial infarction and the other 122 without. In terms of symptoms, 91.6% of these women presented with typical chest pain, and 59.7% had related symptoms.
“With more than 90% having retrosternal pain, the idea that myocardial infarction presents with atypical symptoms in women has been widely challenged, despite the fact that more than half present with related symptoms and it isn’t known in which order these symptoms occur, Dr. Manzo-Silberman said in an interview. But what we can say is that if at any point a young woman mentions chest pain, even when occurring as part of several other symptoms, MI must be deemed a possibility until it has been ruled out.”
The risk profile revealed that 75.5% were smokers, 35% had a family history of heart disease, 33% had pregnancy complications, and 55% had recently experienced a stressful situation. The analysis also showed that cannabis use and oral contraception were primary risk factors in women younger than 35.
“With regard to risk factors, when designing this study we expected that lots of these young women would have largely atypical autoimmune conditions, with high levels of inflammation. We looked for everything, but this was not actually the case. Instead, we found very many women to have classic risk factors; three-quarters were smokers, a modifiable risk factor, which can largely be prevented. The other aspect concerns contraception, and it’s why I insist that gynecologists must be involved insofar as they must inform their patients how to manage their risk factors and tweak their contraception.”
Coronary angiography findings showed that only 1% received a normal result, 29.3% had vessel damage, and 14.6% had aortic dissection. “We were surprised again here because we expected that with young women we would see lots of heart attacks without obstruction, [in other words] normal coronary arteries, atypical forms of MI,” commented Dr. Manzo-Silberman. “In fact, most presented with atheroma, often obstructive lesions, or even triple-vessel disease, in nearly a third of the cohort. So that’s another misconception dispelled – we can’t just think that because a woman is young, nothing will be found. Coronary catheterization should be considered, and the diagnostic process should be completed in full.”
After 1 year, there had been two cancer-related deaths and 25 patients had undergone several angioplasty procedures. Nevertheless, 90.4% had not experienced any type of CV event, and 72% had not even had any symptoms.
“The final surprise was prognosis,” he said. “Previous studies, especially some authored by Viola Vaccarino, MD, PhD, showed an excess hospital rate in women and we had expected this to be the case here, but no hospital deaths were recorded. However, not far off 10% of women attended (at least once) the emergency department in the year following for recurrent chest pain which was not ischemic – ECG normal, troponin normal – so something was missing in their education as a patient.”
“So, there are improvements to be made in terms of secondary prevention, follow-up, and in the education of these young female patients who have experienced the major event that is a myocardial infarction,” concluded Dr. Manzo-Silberman.
This content was originally published on Medscape French edition.
Young women (under 50) are increasingly having heart attacks without doctors really knowing why. This is where the Young Women Presenting Acute Myocardial Infarction in France (WAMIF) study comes in, the results of which were presented in an e-poster at the annual congress of the European Society of Cardiology by Stéphane Manzo-Silberman, MD, Institute of Cardiology, Pitié-Salpétrière, Paris. The results (yet to be published) fight several of the preconceived ideas on the topic, Dr. Manzo-Silberman commented in an interview.
Significantly higher hospital death rates in women
“Cardiovascular disease is the main cause of death in women, killing seven times more than breast cancer,” notes Dr. Manzo-Silberman. The hospital death rate is significantly higher in women and, despite going down, is significantly higher than in men (more than double), particularly in women under 50. What’s more, in addition to the typical risk factors, women present specific risk factors related to hormone changes, high-risk inflammatory profiles, and thrombophilia.”
The WAMIF study was designed to determine the clinical, biological, and morphological features linked to hospital mortality after 12 months in women under 50. The prospective, observational study included all women in this age range from 30 sites in France between May 2017 and June 2019.
90% with retrosternal chest pain
The age of the 314 women enrolled was 44.9 years on average. Nearly two-thirds (192) presented with ST-segment elevation myocardial infarction and the other 122 without. In terms of symptoms, 91.6% of these women presented with typical chest pain, and 59.7% had related symptoms.
“With more than 90% having retrosternal pain, the idea that myocardial infarction presents with atypical symptoms in women has been widely challenged, despite the fact that more than half present with related symptoms and it isn’t known in which order these symptoms occur, Dr. Manzo-Silberman said in an interview. But what we can say is that if at any point a young woman mentions chest pain, even when occurring as part of several other symptoms, MI must be deemed a possibility until it has been ruled out.”
The risk profile revealed that 75.5% were smokers, 35% had a family history of heart disease, 33% had pregnancy complications, and 55% had recently experienced a stressful situation. The analysis also showed that cannabis use and oral contraception were primary risk factors in women younger than 35.
“With regard to risk factors, when designing this study we expected that lots of these young women would have largely atypical autoimmune conditions, with high levels of inflammation. We looked for everything, but this was not actually the case. Instead, we found very many women to have classic risk factors; three-quarters were smokers, a modifiable risk factor, which can largely be prevented. The other aspect concerns contraception, and it’s why I insist that gynecologists must be involved insofar as they must inform their patients how to manage their risk factors and tweak their contraception.”
Coronary angiography findings showed that only 1% received a normal result, 29.3% had vessel damage, and 14.6% had aortic dissection. “We were surprised again here because we expected that with young women we would see lots of heart attacks without obstruction, [in other words] normal coronary arteries, atypical forms of MI,” commented Dr. Manzo-Silberman. “In fact, most presented with atheroma, often obstructive lesions, or even triple-vessel disease, in nearly a third of the cohort. So that’s another misconception dispelled – we can’t just think that because a woman is young, nothing will be found. Coronary catheterization should be considered, and the diagnostic process should be completed in full.”
After 1 year, there had been two cancer-related deaths and 25 patients had undergone several angioplasty procedures. Nevertheless, 90.4% had not experienced any type of CV event, and 72% had not even had any symptoms.
“The final surprise was prognosis,” he said. “Previous studies, especially some authored by Viola Vaccarino, MD, PhD, showed an excess hospital rate in women and we had expected this to be the case here, but no hospital deaths were recorded. However, not far off 10% of women attended (at least once) the emergency department in the year following for recurrent chest pain which was not ischemic – ECG normal, troponin normal – so something was missing in their education as a patient.”
“So, there are improvements to be made in terms of secondary prevention, follow-up, and in the education of these young female patients who have experienced the major event that is a myocardial infarction,” concluded Dr. Manzo-Silberman.
This content was originally published on Medscape French edition.
FROM ESC CONGRESS 2022
Integrase inhibitors and gestational weight gain: Should women worry?
In recent years, increased use of integrase strand transferase inhibitor (INSTI) antiviral treatment (ART) has raised concerns about weight gain and adverse outcomes in patients with HIV. This is especially true regarding possible excessive gestational weight gain, which in women without HIV has been associated with maternal gestational diabetes, hypertensive and liver conditions, as well as related risks for preterm birth, fetal macrosomia, and higher weight after birth.
Unfortunately, few studies in pregnant women with HIV have moved out of the controlled environment into real-world settings, potentially limiting current knowledge about the impact of gestational weight gain – as well as strategies to both prevent it and the associated adverse outcomes.
That is what a team of infectious disease specialists at the Hospital Federal dos Servidores do Estado in Rio de Janeiro recently sought to answer among a cohort of INSTI-experienced and INSTI-naive women with BMIs less than 25 kg/m2 (underweight/normal weight) and higher than 25 kg/m2.
Surprising findings
The investigators determined that rates of excessive weight gain were significantly higher in INSTI-naive women with BMI less than 25 who experienced rates as high as 31.6%, compared with approximately 12% of women who conceived while on INSTIs, regardless of BMI values at baseline (P = .004).
However, rates of unfavorable pregnancy outcomes (for example, small for gestational age, preterm birth, stillbirth, death) appeared to be low overall and similar among all the study groups.
“We had some discussions when we were working on this and thought that the weight gain might have adverse effects,” Trevon Fuller, PhD, lead author and a postdoctoral student at the Hospital Federal dos Servidores do Estado, told this news organization.
“But it looked like the weight gain might actually be good, to the extent that we didn’t see any harm to the mom or the baby of those underweight or normal weight women who were naive to INSTIs,” he explained.
Dr. Fuller and his team enrolled 198 pregnant women living with HIV who sought care at the Hospital Federal dos Servidores do Estado – a national reference center for USAID’s Prevention of Mother to Child Transmission strategic program – between October 2014 and October 2021.
Participants were divided into two primary cohorts: BMI less than 25 at enrollment (n = 74) or BMI of 25 or higher (n = 124), then further divided by timing of INSTI-based combined ART:
- INSTI-naive: women using INSTI-based ART (raltegravir [Isentress] 400 mg twice per day or dolutegravir [Tivicay] 50 mg/day plus 2 non-nucleoside reverse transcriptase inhibitors – lamivudine plus tenofovir disoproxil fumarate or lamivudine plus zidovudine) for 4 weeks between baseline and near delivery.
- INSTI-experienced: women who became pregnant while using INSTIs for at least 6 months before conception.
Among underweight/normal weight participants, 77% (n = 57) were INSTI-naive and 23% (n = 17) INSTI-experienced, and among overweight/obese participants, 81.5% (n = 101) were INSTI-naive, and 18.5% (n = 23) were experienced.
Maternal age, which did not differ significantly by BMI or treatment experience, was a median of 28 years, and most participants were non-White. All participants were virally suppressed near delivery.
Study findings, which were published online in HIV Medicine, highlighted that median weight near delivery in participants who were overweight/obese at baseline was similar regardless of whether they were treatment-experienced (90 kg [198 lb]) or treatment-naive (82.3 kg [181 lb]), P = .026.
However, participants who were underweight/normal weight who were INSTI-naive had significantly higher rates of gestational weight gain (31.5%, 18/57), compared with those of underweight/normal weight who were INSTI-experienced (11.8%, 2/17), P = .004. Notably, this gain was significant in all categories of change (that is, low < 0.18 kg/week, normal 0.18-0.59 kg/week), and high > 0.59 kg/week).
“One of the things that we took away was that this weight gain is primarily happening with women who are starting INSTIs,” said Dr. Fuller.
“The data suggest that [it] might be temporary in the sense that there’s not going to be continuous weight gain but that it will probably approach some type of horizontal asymptote,” he added.
Although obstetric and neonatal outcomes were secondary measures, the investigators did not observe any significantly different outcomes when comparing the groups, and there were no stillbirths, neonatal deaths, or macrosomia.
Preterm delivery rates in underweight/normal weight participants who were INSTI-experienced (11.8%, 2/17) and INSTI-naive (5.3%, 3/57) were similar to overweight/obese participants who were INSTI-experienced (13%, 3/23) and INSTI-naive (6.9%, 7/101).
The same was true for low birthweight.
Still, the study appears to raise more questions than it answers, Sigal Yawetz, MD, an infectious disease specialist at Brigham and Women’s Hospital, Boston, said in an interview – a factor that she said is common also in some of the more recent randomized controlled studies, such as IMPAACT PROMISE.
Dr. Yawetz, who was not involved in the study, also noted, “The groups were small, so comparisons within the groups are difficult, and so many people were excluded that it’s hard to know if there were adverse outcomes related to this ... It’s very confounded.”
The World Health Organization estimates that there are roughly 1.3 million pregnant women with HIV, 81% of whom are on antiretroviral therapy. Although the literature continues to evolve, data suggest that in general, Black women are at greater risk for gestational weight gain.
“We have to remember that women who gain excess weight in pregnancy are still going to be with this weight following pregnancy as well,” Dr. Yawetz said. “So, it might impact their pregnancy but also their health after delivery and for subsequent pregnancies, which we don’t have data for yet.”
Dr. Fuller agrees that more data are needed and mentioned that the team plans to study this further, ideally with larger sample sizes.
Yet, despite the lingering questions, there is a silver lining, one that Dr. Yawetz was emphatic about.
“I really welcome people doing studies on this because we really need the data. By far, integrase inhibitors are the first-line regimen all over the world for pregnant women, and if you look at the gestalt or full picture, this is the best regimen to give pregnant women,” she said.
Dr. Fuller and Dr. Yawetz report no relevant financial relationships. The study was independently supported.
A version of this article first appeared on Medscape.com.
In recent years, increased use of integrase strand transferase inhibitor (INSTI) antiviral treatment (ART) has raised concerns about weight gain and adverse outcomes in patients with HIV. This is especially true regarding possible excessive gestational weight gain, which in women without HIV has been associated with maternal gestational diabetes, hypertensive and liver conditions, as well as related risks for preterm birth, fetal macrosomia, and higher weight after birth.
Unfortunately, few studies in pregnant women with HIV have moved out of the controlled environment into real-world settings, potentially limiting current knowledge about the impact of gestational weight gain – as well as strategies to both prevent it and the associated adverse outcomes.
That is what a team of infectious disease specialists at the Hospital Federal dos Servidores do Estado in Rio de Janeiro recently sought to answer among a cohort of INSTI-experienced and INSTI-naive women with BMIs less than 25 kg/m2 (underweight/normal weight) and higher than 25 kg/m2.
Surprising findings
The investigators determined that rates of excessive weight gain were significantly higher in INSTI-naive women with BMI less than 25 who experienced rates as high as 31.6%, compared with approximately 12% of women who conceived while on INSTIs, regardless of BMI values at baseline (P = .004).
However, rates of unfavorable pregnancy outcomes (for example, small for gestational age, preterm birth, stillbirth, death) appeared to be low overall and similar among all the study groups.
“We had some discussions when we were working on this and thought that the weight gain might have adverse effects,” Trevon Fuller, PhD, lead author and a postdoctoral student at the Hospital Federal dos Servidores do Estado, told this news organization.
“But it looked like the weight gain might actually be good, to the extent that we didn’t see any harm to the mom or the baby of those underweight or normal weight women who were naive to INSTIs,” he explained.
Dr. Fuller and his team enrolled 198 pregnant women living with HIV who sought care at the Hospital Federal dos Servidores do Estado – a national reference center for USAID’s Prevention of Mother to Child Transmission strategic program – between October 2014 and October 2021.
Participants were divided into two primary cohorts: BMI less than 25 at enrollment (n = 74) or BMI of 25 or higher (n = 124), then further divided by timing of INSTI-based combined ART:
- INSTI-naive: women using INSTI-based ART (raltegravir [Isentress] 400 mg twice per day or dolutegravir [Tivicay] 50 mg/day plus 2 non-nucleoside reverse transcriptase inhibitors – lamivudine plus tenofovir disoproxil fumarate or lamivudine plus zidovudine) for 4 weeks between baseline and near delivery.
- INSTI-experienced: women who became pregnant while using INSTIs for at least 6 months before conception.
Among underweight/normal weight participants, 77% (n = 57) were INSTI-naive and 23% (n = 17) INSTI-experienced, and among overweight/obese participants, 81.5% (n = 101) were INSTI-naive, and 18.5% (n = 23) were experienced.
Maternal age, which did not differ significantly by BMI or treatment experience, was a median of 28 years, and most participants were non-White. All participants were virally suppressed near delivery.
Study findings, which were published online in HIV Medicine, highlighted that median weight near delivery in participants who were overweight/obese at baseline was similar regardless of whether they were treatment-experienced (90 kg [198 lb]) or treatment-naive (82.3 kg [181 lb]), P = .026.
However, participants who were underweight/normal weight who were INSTI-naive had significantly higher rates of gestational weight gain (31.5%, 18/57), compared with those of underweight/normal weight who were INSTI-experienced (11.8%, 2/17), P = .004. Notably, this gain was significant in all categories of change (that is, low < 0.18 kg/week, normal 0.18-0.59 kg/week), and high > 0.59 kg/week).
“One of the things that we took away was that this weight gain is primarily happening with women who are starting INSTIs,” said Dr. Fuller.
“The data suggest that [it] might be temporary in the sense that there’s not going to be continuous weight gain but that it will probably approach some type of horizontal asymptote,” he added.
Although obstetric and neonatal outcomes were secondary measures, the investigators did not observe any significantly different outcomes when comparing the groups, and there were no stillbirths, neonatal deaths, or macrosomia.
Preterm delivery rates in underweight/normal weight participants who were INSTI-experienced (11.8%, 2/17) and INSTI-naive (5.3%, 3/57) were similar to overweight/obese participants who were INSTI-experienced (13%, 3/23) and INSTI-naive (6.9%, 7/101).
The same was true for low birthweight.
Still, the study appears to raise more questions than it answers, Sigal Yawetz, MD, an infectious disease specialist at Brigham and Women’s Hospital, Boston, said in an interview – a factor that she said is common also in some of the more recent randomized controlled studies, such as IMPAACT PROMISE.
Dr. Yawetz, who was not involved in the study, also noted, “The groups were small, so comparisons within the groups are difficult, and so many people were excluded that it’s hard to know if there were adverse outcomes related to this ... It’s very confounded.”
The World Health Organization estimates that there are roughly 1.3 million pregnant women with HIV, 81% of whom are on antiretroviral therapy. Although the literature continues to evolve, data suggest that in general, Black women are at greater risk for gestational weight gain.
“We have to remember that women who gain excess weight in pregnancy are still going to be with this weight following pregnancy as well,” Dr. Yawetz said. “So, it might impact their pregnancy but also their health after delivery and for subsequent pregnancies, which we don’t have data for yet.”
Dr. Fuller agrees that more data are needed and mentioned that the team plans to study this further, ideally with larger sample sizes.
Yet, despite the lingering questions, there is a silver lining, one that Dr. Yawetz was emphatic about.
“I really welcome people doing studies on this because we really need the data. By far, integrase inhibitors are the first-line regimen all over the world for pregnant women, and if you look at the gestalt or full picture, this is the best regimen to give pregnant women,” she said.
Dr. Fuller and Dr. Yawetz report no relevant financial relationships. The study was independently supported.
A version of this article first appeared on Medscape.com.
In recent years, increased use of integrase strand transferase inhibitor (INSTI) antiviral treatment (ART) has raised concerns about weight gain and adverse outcomes in patients with HIV. This is especially true regarding possible excessive gestational weight gain, which in women without HIV has been associated with maternal gestational diabetes, hypertensive and liver conditions, as well as related risks for preterm birth, fetal macrosomia, and higher weight after birth.
Unfortunately, few studies in pregnant women with HIV have moved out of the controlled environment into real-world settings, potentially limiting current knowledge about the impact of gestational weight gain – as well as strategies to both prevent it and the associated adverse outcomes.
That is what a team of infectious disease specialists at the Hospital Federal dos Servidores do Estado in Rio de Janeiro recently sought to answer among a cohort of INSTI-experienced and INSTI-naive women with BMIs less than 25 kg/m2 (underweight/normal weight) and higher than 25 kg/m2.
Surprising findings
The investigators determined that rates of excessive weight gain were significantly higher in INSTI-naive women with BMI less than 25 who experienced rates as high as 31.6%, compared with approximately 12% of women who conceived while on INSTIs, regardless of BMI values at baseline (P = .004).
However, rates of unfavorable pregnancy outcomes (for example, small for gestational age, preterm birth, stillbirth, death) appeared to be low overall and similar among all the study groups.
“We had some discussions when we were working on this and thought that the weight gain might have adverse effects,” Trevon Fuller, PhD, lead author and a postdoctoral student at the Hospital Federal dos Servidores do Estado, told this news organization.
“But it looked like the weight gain might actually be good, to the extent that we didn’t see any harm to the mom or the baby of those underweight or normal weight women who were naive to INSTIs,” he explained.
Dr. Fuller and his team enrolled 198 pregnant women living with HIV who sought care at the Hospital Federal dos Servidores do Estado – a national reference center for USAID’s Prevention of Mother to Child Transmission strategic program – between October 2014 and October 2021.
Participants were divided into two primary cohorts: BMI less than 25 at enrollment (n = 74) or BMI of 25 or higher (n = 124), then further divided by timing of INSTI-based combined ART:
- INSTI-naive: women using INSTI-based ART (raltegravir [Isentress] 400 mg twice per day or dolutegravir [Tivicay] 50 mg/day plus 2 non-nucleoside reverse transcriptase inhibitors – lamivudine plus tenofovir disoproxil fumarate or lamivudine plus zidovudine) for 4 weeks between baseline and near delivery.
- INSTI-experienced: women who became pregnant while using INSTIs for at least 6 months before conception.
Among underweight/normal weight participants, 77% (n = 57) were INSTI-naive and 23% (n = 17) INSTI-experienced, and among overweight/obese participants, 81.5% (n = 101) were INSTI-naive, and 18.5% (n = 23) were experienced.
Maternal age, which did not differ significantly by BMI or treatment experience, was a median of 28 years, and most participants were non-White. All participants were virally suppressed near delivery.
Study findings, which were published online in HIV Medicine, highlighted that median weight near delivery in participants who were overweight/obese at baseline was similar regardless of whether they were treatment-experienced (90 kg [198 lb]) or treatment-naive (82.3 kg [181 lb]), P = .026.
However, participants who were underweight/normal weight who were INSTI-naive had significantly higher rates of gestational weight gain (31.5%, 18/57), compared with those of underweight/normal weight who were INSTI-experienced (11.8%, 2/17), P = .004. Notably, this gain was significant in all categories of change (that is, low < 0.18 kg/week, normal 0.18-0.59 kg/week), and high > 0.59 kg/week).
“One of the things that we took away was that this weight gain is primarily happening with women who are starting INSTIs,” said Dr. Fuller.
“The data suggest that [it] might be temporary in the sense that there’s not going to be continuous weight gain but that it will probably approach some type of horizontal asymptote,” he added.
Although obstetric and neonatal outcomes were secondary measures, the investigators did not observe any significantly different outcomes when comparing the groups, and there were no stillbirths, neonatal deaths, or macrosomia.
Preterm delivery rates in underweight/normal weight participants who were INSTI-experienced (11.8%, 2/17) and INSTI-naive (5.3%, 3/57) were similar to overweight/obese participants who were INSTI-experienced (13%, 3/23) and INSTI-naive (6.9%, 7/101).
The same was true for low birthweight.
Still, the study appears to raise more questions than it answers, Sigal Yawetz, MD, an infectious disease specialist at Brigham and Women’s Hospital, Boston, said in an interview – a factor that she said is common also in some of the more recent randomized controlled studies, such as IMPAACT PROMISE.
Dr. Yawetz, who was not involved in the study, also noted, “The groups were small, so comparisons within the groups are difficult, and so many people were excluded that it’s hard to know if there were adverse outcomes related to this ... It’s very confounded.”
The World Health Organization estimates that there are roughly 1.3 million pregnant women with HIV, 81% of whom are on antiretroviral therapy. Although the literature continues to evolve, data suggest that in general, Black women are at greater risk for gestational weight gain.
“We have to remember that women who gain excess weight in pregnancy are still going to be with this weight following pregnancy as well,” Dr. Yawetz said. “So, it might impact their pregnancy but also their health after delivery and for subsequent pregnancies, which we don’t have data for yet.”
Dr. Fuller agrees that more data are needed and mentioned that the team plans to study this further, ideally with larger sample sizes.
Yet, despite the lingering questions, there is a silver lining, one that Dr. Yawetz was emphatic about.
“I really welcome people doing studies on this because we really need the data. By far, integrase inhibitors are the first-line regimen all over the world for pregnant women, and if you look at the gestalt or full picture, this is the best regimen to give pregnant women,” she said.
Dr. Fuller and Dr. Yawetz report no relevant financial relationships. The study was independently supported.
A version of this article first appeared on Medscape.com.
Weight gain during pregnancy may play role in child ADHD risk
Obesity in women of reproductive age has emerged as one of the main risk factors associated with neonatal complications and long-term neuropsychiatric consequences in offspring, including attention-deficit/hyperactivity disorder.
Research has also linked pregestational diabetes and gestational diabetes mellitus (GDM) to an increased risk for ADHD in offspring. Now, an observational study of 1,036 singleton births at one hospital between 1998 and 2008 suggests that in the presence of GDM, maternal obesity combined with excessive weight gain during pregnancy may be jointly associated with increased risk of offspring ADHD. The median follow-up was 17.7 years.
Maternal obesity was independently associated with ADHD (adjusted hazard ratio, 1.66; 95% confidence interval: 1.07-2.60), but excessive weight gain during pregnancy and maternal overweight were not, reported Verónica Perea, MD, PhD, of the Hospital Universitari Mútua de Terrassa, Barcelona, and colleagues in the Journal of Clinical Endocrinology & Metabolism.
However, in women with pregestation obesity who gained more weight than recommended by the National Academy of Medicine (NAM), the risk of offspring ADHD was higher, compared with women of normal weight whose pregnancy weight stayed within NAM guidelines (adjusted hazard ratio, 2.13; 95% confidence interval: 1.14-4.01).
“The results of this study suggest that the negative repercussions of excessive weight gain on children within the setting of a high-risk population with GDM and obesity were not only observed during the prenatal period but also years later with a development of ADHD,” the researchers wrote.
The study also showed that when maternal weight gain did not exceed NAM guidelines, maternal obesity was no longer independently associated with ADHD in offspring (aHR, 1.36; 95% CI: 0.78-2.36). This finding conflicts with earlier studies focusing primarily on the role of pregestational maternal weight, the researchers said. A 2018 nationwide Finnish cohort study in newborns showed an increased long-term risk of ADHD in those born to women with GDM, compared with the nondiabetic population. This long-term risk of ADHD increased in the presence of pregestational obesity (HR, 1.64).
Similarly, evidence from systematic reviews and meta-analyses has demonstrated that antenatal lifestyle interventions to prevent excessive weight gain during pregnancy were associated with a reduction in adverse pregnancy outcomes. However, evidence on offspring mental health was lacking, especially in high-risk pregnancies with gestational diabetes, the study authors said.
Although causal inferences can’t be drawn from the current observational study, “it seems that the higher risk [of ADHD] observed would be explained by the role of gestational weight gain during the antenatal period,” Dr. Perea said in an interview. Importantly, the study highlights a window of opportunity for promoting healthy weight gain during pregnancy, Dr. Perea said. ”This should be a priority in the current management of gestation.”
Fatima Cody Stanford, MD, MPH, an associate professor of medicine and pediatrics at Harvard Medical School, Boston, agreed. “I think one of the key issues is that there’s very little attention paid to how weight gain is regulated during pregnancy,” she said in an interview. On many other points, however, Dr. Stanford, who is a specialist in obesity medicine at Massachusetts General Hospital Weight Center, did not agree.
The association between ADHD and obesity has already been well established by a 2019 meta-analysis and systematic review of studies over the last 10 years, she emphasized. “These studies were able to show a much stronger association between maternal obesity and ADHD in offspring because they were powered to detect differences.”
The current study does not say “anything new or novel,” Dr. Stanford added. “Maternal obesity and the association with an increased risk of ADHD in offspring is the main issue. I don’t think there was any appreciable increase when weight gain during pregnancy was factored in. It’s mild at best.”
Eran Bornstein, MD, vice-chair of obstetrics and gynecology at Lenox Hill Hospital, New York, expressed a similar point of view. Although the study findings “add to the current literature,” they should be interpreted “cautiously,” Dr. Bornstein said in an interview.
The size of the effect on ADHD risk attributable to maternal weight gain during pregnancy “was not clear,” he said. “Cohort studies of this sort are excellent for finding associations which help us generate the hypothesis, but this doesn’t demonstrate a cause and effect or a magnitude for this effect.”
Physicians should follow cumulative data suggesting that maternal obesity is associated with a number of pregnancy complications and neonatal outcomes in women with and without diabetes, Dr. Bornstein suggested. “Optimizing maternal weight prior to pregnancy and adhering to recommendations regarding weight gain has the potential to improve some of these outcomes.”
Treating obesity prior to conception mitigates GDM risk, agreed Dr. Stanford. “The issue,” she explained, “is that all of the drugs approved for the treatment of obesity are contraindicated in pregnancy and lifestyle modification fails in 96% of cases, even when there is no pregnancy.” Drugs such as metformin are being used off-label to treat obesity and to safely manage gestational weight gain, she said. “Those of us who practice obesity medicine know that metformin can be safely used throughout pregnancy with no harm to the fetus.”
This study was partially funded by Fundació Docència i Recerca MútuaTerrassa. Dr. Perea and study coauthors reporting have no conflicts of interest. Dr. Stanford disclosed relationships with Novo Nordisk, Eli Lilly, Boehringer Ingelheim, Gelesis, Pfizer, Currax, and Rhythm. Dr. Bornstein reported having no conflicts of interest.
This story was updated on 11/7/2022.
Obesity in women of reproductive age has emerged as one of the main risk factors associated with neonatal complications and long-term neuropsychiatric consequences in offspring, including attention-deficit/hyperactivity disorder.
Research has also linked pregestational diabetes and gestational diabetes mellitus (GDM) to an increased risk for ADHD in offspring. Now, an observational study of 1,036 singleton births at one hospital between 1998 and 2008 suggests that in the presence of GDM, maternal obesity combined with excessive weight gain during pregnancy may be jointly associated with increased risk of offspring ADHD. The median follow-up was 17.7 years.
Maternal obesity was independently associated with ADHD (adjusted hazard ratio, 1.66; 95% confidence interval: 1.07-2.60), but excessive weight gain during pregnancy and maternal overweight were not, reported Verónica Perea, MD, PhD, of the Hospital Universitari Mútua de Terrassa, Barcelona, and colleagues in the Journal of Clinical Endocrinology & Metabolism.
However, in women with pregestation obesity who gained more weight than recommended by the National Academy of Medicine (NAM), the risk of offspring ADHD was higher, compared with women of normal weight whose pregnancy weight stayed within NAM guidelines (adjusted hazard ratio, 2.13; 95% confidence interval: 1.14-4.01).
“The results of this study suggest that the negative repercussions of excessive weight gain on children within the setting of a high-risk population with GDM and obesity were not only observed during the prenatal period but also years later with a development of ADHD,” the researchers wrote.
The study also showed that when maternal weight gain did not exceed NAM guidelines, maternal obesity was no longer independently associated with ADHD in offspring (aHR, 1.36; 95% CI: 0.78-2.36). This finding conflicts with earlier studies focusing primarily on the role of pregestational maternal weight, the researchers said. A 2018 nationwide Finnish cohort study in newborns showed an increased long-term risk of ADHD in those born to women with GDM, compared with the nondiabetic population. This long-term risk of ADHD increased in the presence of pregestational obesity (HR, 1.64).
Similarly, evidence from systematic reviews and meta-analyses has demonstrated that antenatal lifestyle interventions to prevent excessive weight gain during pregnancy were associated with a reduction in adverse pregnancy outcomes. However, evidence on offspring mental health was lacking, especially in high-risk pregnancies with gestational diabetes, the study authors said.
Although causal inferences can’t be drawn from the current observational study, “it seems that the higher risk [of ADHD] observed would be explained by the role of gestational weight gain during the antenatal period,” Dr. Perea said in an interview. Importantly, the study highlights a window of opportunity for promoting healthy weight gain during pregnancy, Dr. Perea said. ”This should be a priority in the current management of gestation.”
Fatima Cody Stanford, MD, MPH, an associate professor of medicine and pediatrics at Harvard Medical School, Boston, agreed. “I think one of the key issues is that there’s very little attention paid to how weight gain is regulated during pregnancy,” she said in an interview. On many other points, however, Dr. Stanford, who is a specialist in obesity medicine at Massachusetts General Hospital Weight Center, did not agree.
The association between ADHD and obesity has already been well established by a 2019 meta-analysis and systematic review of studies over the last 10 years, she emphasized. “These studies were able to show a much stronger association between maternal obesity and ADHD in offspring because they were powered to detect differences.”
The current study does not say “anything new or novel,” Dr. Stanford added. “Maternal obesity and the association with an increased risk of ADHD in offspring is the main issue. I don’t think there was any appreciable increase when weight gain during pregnancy was factored in. It’s mild at best.”
Eran Bornstein, MD, vice-chair of obstetrics and gynecology at Lenox Hill Hospital, New York, expressed a similar point of view. Although the study findings “add to the current literature,” they should be interpreted “cautiously,” Dr. Bornstein said in an interview.
The size of the effect on ADHD risk attributable to maternal weight gain during pregnancy “was not clear,” he said. “Cohort studies of this sort are excellent for finding associations which help us generate the hypothesis, but this doesn’t demonstrate a cause and effect or a magnitude for this effect.”
Physicians should follow cumulative data suggesting that maternal obesity is associated with a number of pregnancy complications and neonatal outcomes in women with and without diabetes, Dr. Bornstein suggested. “Optimizing maternal weight prior to pregnancy and adhering to recommendations regarding weight gain has the potential to improve some of these outcomes.”
Treating obesity prior to conception mitigates GDM risk, agreed Dr. Stanford. “The issue,” she explained, “is that all of the drugs approved for the treatment of obesity are contraindicated in pregnancy and lifestyle modification fails in 96% of cases, even when there is no pregnancy.” Drugs such as metformin are being used off-label to treat obesity and to safely manage gestational weight gain, she said. “Those of us who practice obesity medicine know that metformin can be safely used throughout pregnancy with no harm to the fetus.”
This study was partially funded by Fundació Docència i Recerca MútuaTerrassa. Dr. Perea and study coauthors reporting have no conflicts of interest. Dr. Stanford disclosed relationships with Novo Nordisk, Eli Lilly, Boehringer Ingelheim, Gelesis, Pfizer, Currax, and Rhythm. Dr. Bornstein reported having no conflicts of interest.
This story was updated on 11/7/2022.
Obesity in women of reproductive age has emerged as one of the main risk factors associated with neonatal complications and long-term neuropsychiatric consequences in offspring, including attention-deficit/hyperactivity disorder.
Research has also linked pregestational diabetes and gestational diabetes mellitus (GDM) to an increased risk for ADHD in offspring. Now, an observational study of 1,036 singleton births at one hospital between 1998 and 2008 suggests that in the presence of GDM, maternal obesity combined with excessive weight gain during pregnancy may be jointly associated with increased risk of offspring ADHD. The median follow-up was 17.7 years.
Maternal obesity was independently associated with ADHD (adjusted hazard ratio, 1.66; 95% confidence interval: 1.07-2.60), but excessive weight gain during pregnancy and maternal overweight were not, reported Verónica Perea, MD, PhD, of the Hospital Universitari Mútua de Terrassa, Barcelona, and colleagues in the Journal of Clinical Endocrinology & Metabolism.
However, in women with pregestation obesity who gained more weight than recommended by the National Academy of Medicine (NAM), the risk of offspring ADHD was higher, compared with women of normal weight whose pregnancy weight stayed within NAM guidelines (adjusted hazard ratio, 2.13; 95% confidence interval: 1.14-4.01).
“The results of this study suggest that the negative repercussions of excessive weight gain on children within the setting of a high-risk population with GDM and obesity were not only observed during the prenatal period but also years later with a development of ADHD,” the researchers wrote.
The study also showed that when maternal weight gain did not exceed NAM guidelines, maternal obesity was no longer independently associated with ADHD in offspring (aHR, 1.36; 95% CI: 0.78-2.36). This finding conflicts with earlier studies focusing primarily on the role of pregestational maternal weight, the researchers said. A 2018 nationwide Finnish cohort study in newborns showed an increased long-term risk of ADHD in those born to women with GDM, compared with the nondiabetic population. This long-term risk of ADHD increased in the presence of pregestational obesity (HR, 1.64).
Similarly, evidence from systematic reviews and meta-analyses has demonstrated that antenatal lifestyle interventions to prevent excessive weight gain during pregnancy were associated with a reduction in adverse pregnancy outcomes. However, evidence on offspring mental health was lacking, especially in high-risk pregnancies with gestational diabetes, the study authors said.
Although causal inferences can’t be drawn from the current observational study, “it seems that the higher risk [of ADHD] observed would be explained by the role of gestational weight gain during the antenatal period,” Dr. Perea said in an interview. Importantly, the study highlights a window of opportunity for promoting healthy weight gain during pregnancy, Dr. Perea said. ”This should be a priority in the current management of gestation.”
Fatima Cody Stanford, MD, MPH, an associate professor of medicine and pediatrics at Harvard Medical School, Boston, agreed. “I think one of the key issues is that there’s very little attention paid to how weight gain is regulated during pregnancy,” she said in an interview. On many other points, however, Dr. Stanford, who is a specialist in obesity medicine at Massachusetts General Hospital Weight Center, did not agree.
The association between ADHD and obesity has already been well established by a 2019 meta-analysis and systematic review of studies over the last 10 years, she emphasized. “These studies were able to show a much stronger association between maternal obesity and ADHD in offspring because they were powered to detect differences.”
The current study does not say “anything new or novel,” Dr. Stanford added. “Maternal obesity and the association with an increased risk of ADHD in offspring is the main issue. I don’t think there was any appreciable increase when weight gain during pregnancy was factored in. It’s mild at best.”
Eran Bornstein, MD, vice-chair of obstetrics and gynecology at Lenox Hill Hospital, New York, expressed a similar point of view. Although the study findings “add to the current literature,” they should be interpreted “cautiously,” Dr. Bornstein said in an interview.
The size of the effect on ADHD risk attributable to maternal weight gain during pregnancy “was not clear,” he said. “Cohort studies of this sort are excellent for finding associations which help us generate the hypothesis, but this doesn’t demonstrate a cause and effect or a magnitude for this effect.”
Physicians should follow cumulative data suggesting that maternal obesity is associated with a number of pregnancy complications and neonatal outcomes in women with and without diabetes, Dr. Bornstein suggested. “Optimizing maternal weight prior to pregnancy and adhering to recommendations regarding weight gain has the potential to improve some of these outcomes.”
Treating obesity prior to conception mitigates GDM risk, agreed Dr. Stanford. “The issue,” she explained, “is that all of the drugs approved for the treatment of obesity are contraindicated in pregnancy and lifestyle modification fails in 96% of cases, even when there is no pregnancy.” Drugs such as metformin are being used off-label to treat obesity and to safely manage gestational weight gain, she said. “Those of us who practice obesity medicine know that metformin can be safely used throughout pregnancy with no harm to the fetus.”
This study was partially funded by Fundació Docència i Recerca MútuaTerrassa. Dr. Perea and study coauthors reporting have no conflicts of interest. Dr. Stanford disclosed relationships with Novo Nordisk, Eli Lilly, Boehringer Ingelheim, Gelesis, Pfizer, Currax, and Rhythm. Dr. Bornstein reported having no conflicts of interest.
This story was updated on 11/7/2022.
The Journal of Clinical Endocrinology & Metabolism
Intimate partner violence: Opening the door to a safer future
THE CASE
Louise T* is a 42-year-old woman who presented to her family medicine office for a routine annual visit. During the exam, her physician noticed bruises on Ms. T’s arms and back. Upon further inquiry, Ms. T reported that she and her husband had argued the night before the appointment. With some hesitancy, she went on to say that this was not the first time this had happened. She said that she and her husband had been arguing frequently for several years and that 6 months earlier, when he lost his job, he began hitting and pushing her.
●
*The patient’s name has been changed to protect her identity.
Intimate partner violence (IPV) includes physical, sexual, or psychological aggression or stalking perpetrated by a current or former relationship partner.1 IPV affects more than 12 million men and women living in the United States each year.2 According to a national survey of IPV, approximately one-third (35.6%) of women and one-quarter (28.5%) of men living in the United States experience rape, physical violence, or stalking by an intimate partner during their lifetime.2 Lifetime exposure to psychological IPV is even more prevalent, affecting nearly half of women and men (48.4% and 48.8%, respectively).2
Lifetime prevalence of any form of IPV is higher among women who identify as bisexual (59.8%) and lesbian (46.3%) compared with those who identify as heterosexual (37.2%); rates are comparable among men who identify as heterosexual (31.9%), bisexual (35.3%), and gay (35.1%).3 Preliminary data suggest that IPV may have increased in frequency and severity during the COVID-19 pandemic, particularly in the context of mandated shelter-in-place and stay-at-home orders.4-6
IPV is associated with numerous negative health consequences. They include fear and concern for safety, mental health disorders such as posttraumatic stress disorder (PTSD), and physical health problems including physical injury, chronic pain, sleep disturbance, and frequent headaches.2 IPV is also associated with a greater number of missed days from school and work and increased utilization of legal, health care, and housing services.2,7 The overall annual cost of IPV against women is estimated at $5.8 billion, with health care costs accounting for approximately $4.1 billion.7 Family physicians can play an important role in curbing the devastating effects of IPV by screening patients and providing resources when needed.
Facilitate disclosure using screening tools and protocol
In Ms. T’s case, evidence of violence was clearly visible. However, not all instances of IPV leave physical marks. The US Preventive Services Task Force (USPSTF) recommends that all women of childbearing age be screened for IPV, whether or not they exhibit signs of violence.8 While the USPSTF has only published recommendations regarding screening women for IPV, there has been a recent push to screen all patients given that men also experience high rates of IPV.9
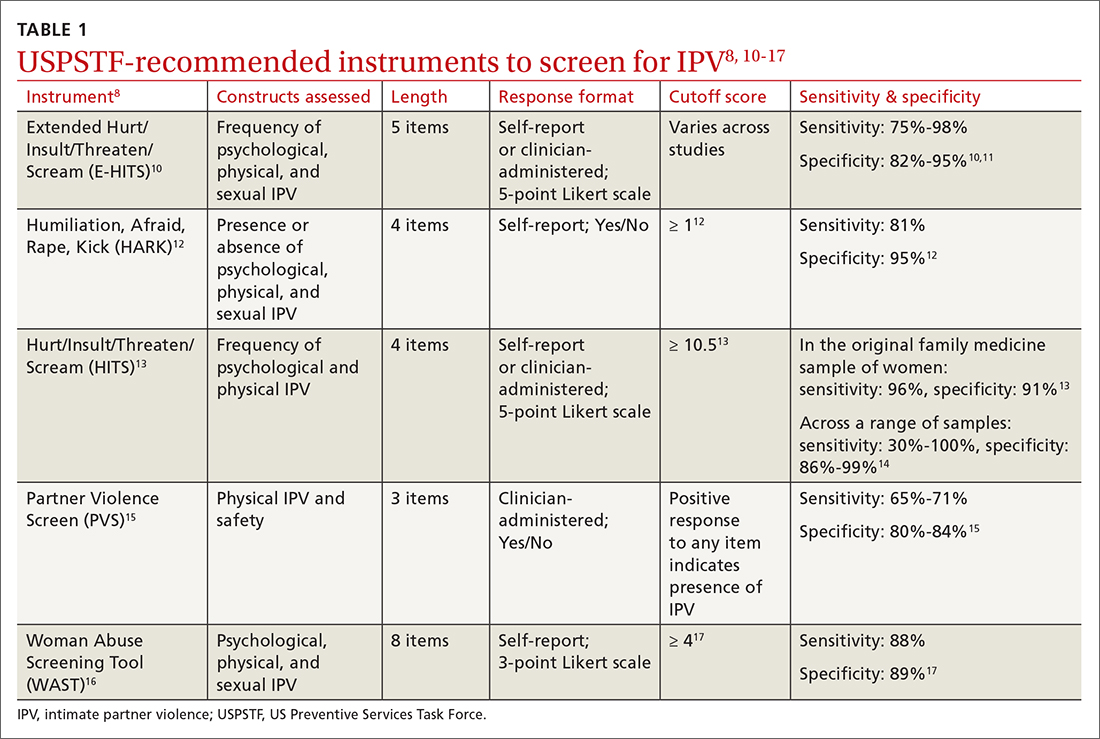
Utilize a brief screening tool. Directly ask patients about IPV; this can help reduce stigma, facilitate disclosure, and initiate the process of connecting patients to potentially lifesaving resources. The USPSTF lists several brief screening measures that can be used in primary care settings to assess exposure to IPV (TABLE 18,10-17). The brevity of these screening tools makes them well suited for busy physicians; cutoff scores facilitate the rapid identification of positive screens. While the USPSTF has not made specific recommendations regarding a screening interval, many studies examining the utility of these measures have reported on annual screenings.8 While there is limited evidence that brief screening alone leads to reductions in IPV,8 discussing IPV in a supportive and empathic manner and connecting patients to resources, such as supportive counseling, does have an important benefit: It can reduce symptoms of depression.18
Continue to: Screen patients in private; this protocol can help
Screen patients in private; this protocol can help. Given the sensitive nature of IPV and the potential danger some patients may be facing, it is important to screen patients in a safe and supportive environment.19,20 Screening should be conducted by the primary care clinician, ideally when a trusting relationship already has been formed. Screen patients only when they are alone in a private room; avoid screening in public spaces such as clinic waiting rooms or in the vicinity of the patient’s partner or children older than age 2 years.19,20
To provide all patients with an opportunity for private and safe IPV screening, clinics are encouraged to develop a clinic-wide policy whereby patients are routinely escorted to the exam room alone for the first portion of their visit, after which any accompanying individuals may be invited to join.21 Clinic staff can inform patients and accompanying individuals of this policy when they first arrive. Once in the exam room, and before the screening process begins, clearly state reporting requirements to ensure that patients can make an informed decision about whether to disclose IPV.19
Set a receptive tone. The manner in which clinicians discuss IPV with their patients is just as important as the setting. Demonstrating sensitivity and genuine concern for the patient’s safety and well-being may increase the patient’s comfort level throughout the screening process and may facilitate disclosures of IPV.19,22 When screening patients for IPV, sit face to face rather than standing over them, maintain warm and open body language, and speak in a soft tone of voice.22
Patients may feel more comfortable if you ask screening questions in a straightforward, nonjudgmental manner, as this helps to normalize the screening experience. We also recommend using behaviorally specific language (eg, “Do arguments [with your partner] ever result in hitting, kicking, or pushing?”16 or “How often does your partner scream or curse at you?”),13 as some patients who have experienced IPV will not label their experiences as “abuse” or “violence.” Not every patient who experiences IPV will be ready to disclose these events; however, maintaining a positive and supportive relationship during routine IPV screening and throughout the remainder of the medical visit may help facilitate future disclosures if, and when, a patient is ready to seek support.19
CRITICAL INTERVENTION ELEMENTS: EMPATHY AND SAFETY
A physician’s response to an IPV disclosure can have a lasting impact on the patient. We encourage family physicians to respond to IPV disclosures with empathy. Maintain eye contact and warm body language, validate the patient’s experiences (“I am sorry this happened to you,” “that must have been terrifying”), tell the patient that the violence was not their fault, and thank the patient for disclosing.23
Continue to: Assess patient safety
Assess patient safety. Another critical component of intervention is to assess the patient’s safety and engage in safety planning. If the patient agrees to this next step, you may wish to provide a warm handoff to a trained social worker, nurse, or psychologist in the clinic who can spend more time covering this information with the patient. Some key components of a safety assessment include determining whether the violence or threat of violence is ongoing and identifying who lives in the home (eg, the partner, children, and any pets). You and the patient can also discuss red flags that would indicate elevated risk. You should discuss red flags that are unique to the patient’s relationship as well as common factors that have been found to heighten risk for IPV (eg, partner engaging in heavy alcohol use).1
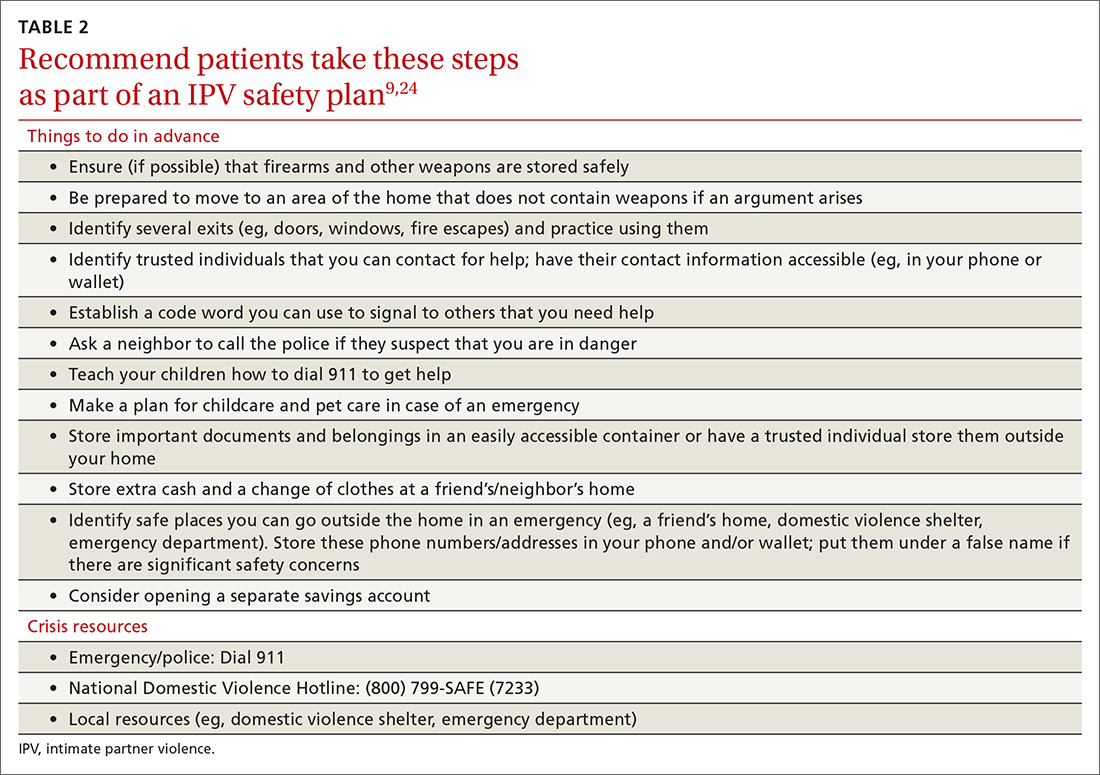
With the patient’s permission, collaboratively construct a safety plan that details how the patient can stay safe on a daily basis and how to safely leave should a dangerous situation arise (TABLE 29,24). The interactive safety planning tool available on the National Domestic Violence Hotline’s website can be a valuable resource (www.thehotline.org/plan-for-safety/).24 Finally, if a patient is experiencing mental health concerns associated with IPV (eg, PTSD, depression, substance misuse, suicidal ideation), consider a referral to a domestic violence counseling center or mental health provider.
Move at the patient’s pace. Even if patients are willing to disclose IPV, they will differ in their readiness to discuss psychoeducation, safety planning, and referrals. Similarly, even if a patient is experiencing severe violence, they may not be ready to leave the relationship. Thus, it’s important to ask the patient for permission before initiating each successive step of the follow-up intervention. You and the patient may wish to schedule additional appointments to discuss this information at a pace the patient finds appropriate.
You may need to spend some time helping the patient recognize the severity of their situation and to feel empowered to take action. In addition, offer information and resources to all patients, even those who do not disclose IPV. Some patients may want to receive this information even if they do not feel comfortable sharing their experiences during the appointment.20 You can also inform patients that they are welcome to bring up issues related to IPV at any future appointments in order to leave the door open to future disclosures.
THE CASE
The physician determined that Ms. T had been experiencing physical and psychological IPV in her current relationship. After responding empathically and obtaining the patient’s consent, the physician provided a warm handoff to the psychologist in the clinic. With Ms. T’s permission, the psychologist provided psychoeducation about IPV, and they discussed Ms. T’s current situation and risk level. They determined that Ms. T was at risk for subsequent episodes of IPV and they collaborated on a safety plan, making sure to discuss contact information for local and national crisis resources.
Continue to: Ms. T saved the phone number...
Ms. T saved the phone number for her local domestic violence shelter in her phone under a false name in case her husband looked through her phone. She said she planned to work on several safety plan items when her husband was away from the house and it was safe to do so. For example, she planned to identify additional ways to exit the house in an emergency and she was going to put together a bag with a change of clothes and some money and drop it off at a trusted friend’s house.
Ms. T and the psychologist agreed to follow up with an office visit in 1 week to discuss any additional safety concerns and to determine whether Ms. T could benefit from a referral to domestic violence counseling services or mental health treatment. The psychologist provided a summary of the topics she and Ms. T had discussed to the physician. The physician scheduled a follow-up appointment with Ms. T in 3 weeks to assess her current safety, troubleshoot any difficulties in implementing her safety plan, and offer additional resources, as needed.
CORRESPONDENCE
Andrea Massa, PhD, 125 Doughty Street, Suite 300, Charleston, SC 29403; [email protected]
1. CDC. National Center for Injury Prevention and Control. Preventing intimate partner violence. 2021. Accessed June 27, 2022. www.cdc.gov/violenceprevention/intimatepartnerviolence/fastfact.html
2. CDC. Black MC, Basile KC, Breiding MJ, et al. The National Intimate Partner and Sexual Violence Survey: 2010 Summary Report. Accessed June 27, 2022. www.cdc.gov/violenceprevention/pdf/nisvs_executive_summary-a.pdf
3. Chen J, Walters ML, Gilbert LK, et al. Sexual violence, stalking, and intimate partner violence by sexual orientation, United States. Psychol Violence. 2020;10:110-119. doi:10.1037/vio0000252
4. Kofman YB, Garfin DR. Home is not always a haven: the domestic violence crisis amid the COVID-19 pandemic. Psychol Trauma. 2020;12:S199-S201. doi:10.1037/tra0000866
5. Lyons M, Brewer G. Experiences of intimate partner violence during lockdown and the COVID-19 pandemic. J Fam Violence. 2021:1-9. doi:10.1007/s10896-021-00260-x
6. Parrott DJ, Halmos MB, Stappenbeck CA, et al. Intimate partner aggression during the COVID-19 pandemic: associations with stress and heavy drinking. Psychol Violence. 2021;12:95-103. doi:10.1037/vio0000395
7. CDC. National Center for Injury Prevention and
8. US Preventive Services Task Force. Screening for intimate partner violence, elder abuse, and abuse of vulnerable adults: US Preventive Services Task Force final recommendation statement. JAMA. 2018;320:1678-1687. doi:10.1001/jama.2018.14741
9. Sprunger JG, Schumacher JA, Coffey SF, et al. It’s time to start asking all patients about intimate partner violence. J Fam Pract. 2019;68:152-161.
10. Chan CC, Chan YC, Au A, et al. Reliability and validity of the “Extended - Hurt, Insult, Threaten, Scream” (E-HITS) screening tool in detecting intimate partner violence in hospital emergency departments in Hong Kong. Hong Kong J Emerg Med. 2010;17:109-117. doi:10.1177/102490791001700202
11. Iverson KM, King MW, Gerber MR, et al. Accuracy of an intimate partner violence screening tool for female VHA patients: a replication and extension. J Trauma Stress. 2015;28:79-82. doi:10.1002/jts.21985
12. Sohal H, Eldridge S, Feder G. The sensitivity and specificity of four questions (HARK) to identify intimate partner violence: a diagnostic accuracy study in general practice. BMC Fam Pract. 2007;8:49. doi:10.1186/1471-2296-8-49
13. Sherin KM, Sinacore JM, Li X, et al. HITS: a short domestic violence screening tool for use in a family practice setting. Fam Med. 1998;30:508-512.
14. Rabin RF, Jennings JM, Campbell JC, et al. Intimate partner violence screening tools: a systematic review. Am J Prev Med. 2009;36:439-445.e4. doi:10.1016/j.amepre.2009.01.024
15. Feldhaus KM, Koziol-McLain J, Amsbury HL, et al. Accuracy of 3 brief screening questions for detecting partner violence in the emergency department. JAMA. 1997;277:1357-1361. doi:10.1001/jama.1997.03540410035027
16. Brown JB, Lent B, Schmidt G, et al. Application of the Woman Abuse Screening Tool (WAST) and WAST-short in the family practice setting. J Fam Pract. 2000;49:896-903.
17. Wathen CN, Jamieson E, MacMillan HL, MVAWRG. Who is identified by screening for intimate partner violence? Womens Health Issues. 2008;18:423-432. doi:10.1016/j.whi.2008.08.003
18. Hegarty K, O’Doherty L, Taft A, et al. Screening and counselling in the primary care setting for women who have experienced intimate partner violence (WEAVE): a cluster randomised controlled trial. Lancet. 2013;382:249-258. doi: 10.1016/S0140-6736(13)60052-5
19. Correa NP, Cain CM, Bertenthal M, et al. Women’s experiences of being screened for intimate partner violence in the health care setting. Nurs Womens Health. 2020;24:185-196. doi:10.1016/j.nwh.2020.04.002
20. Chang JC, Decker MR, Moracco KE, et al. Asking about intimate partner violence: advice from female survivors to health care providers. Patient Educ Couns. 2005;59:141-147. doi:10.1016/j.pec.2004.10.008
21. Paterno MT, Draughon JE. Screening for intimate partner violence. J Midwifery Womens Health. 2016;61:370-375. doi:10.1111/jmwh.12443
22. Iverson KM, Huang K, Wells SY, et al. Women veterans’ preferences for intimate partner violence screening and response procedures within the Veterans Health Administration. Res Nurs Health. 2014;37:302-311. doi:10.1002/nur.21602
23. National Sexual Violence Research Center. Assessing patients for sexual violence: A guide for health care providers. 2011. Accessed June 28, 2022. www.nsvrc.org/publications/assessing-patients-sexual-violence-guide-health-care-providers
24. National Domestic Violence Hotline. Interactive guide to safety planning. Accessed August 22, 2022. https://www.thehotline.org/plan-for-safety/create-a-safety-plan/
THE CASE
Louise T* is a 42-year-old woman who presented to her family medicine office for a routine annual visit. During the exam, her physician noticed bruises on Ms. T’s arms and back. Upon further inquiry, Ms. T reported that she and her husband had argued the night before the appointment. With some hesitancy, she went on to say that this was not the first time this had happened. She said that she and her husband had been arguing frequently for several years and that 6 months earlier, when he lost his job, he began hitting and pushing her.
●
*The patient’s name has been changed to protect her identity.
Intimate partner violence (IPV) includes physical, sexual, or psychological aggression or stalking perpetrated by a current or former relationship partner.1 IPV affects more than 12 million men and women living in the United States each year.2 According to a national survey of IPV, approximately one-third (35.6%) of women and one-quarter (28.5%) of men living in the United States experience rape, physical violence, or stalking by an intimate partner during their lifetime.2 Lifetime exposure to psychological IPV is even more prevalent, affecting nearly half of women and men (48.4% and 48.8%, respectively).2
Lifetime prevalence of any form of IPV is higher among women who identify as bisexual (59.8%) and lesbian (46.3%) compared with those who identify as heterosexual (37.2%); rates are comparable among men who identify as heterosexual (31.9%), bisexual (35.3%), and gay (35.1%).3 Preliminary data suggest that IPV may have increased in frequency and severity during the COVID-19 pandemic, particularly in the context of mandated shelter-in-place and stay-at-home orders.4-6
IPV is associated with numerous negative health consequences. They include fear and concern for safety, mental health disorders such as posttraumatic stress disorder (PTSD), and physical health problems including physical injury, chronic pain, sleep disturbance, and frequent headaches.2 IPV is also associated with a greater number of missed days from school and work and increased utilization of legal, health care, and housing services.2,7 The overall annual cost of IPV against women is estimated at $5.8 billion, with health care costs accounting for approximately $4.1 billion.7 Family physicians can play an important role in curbing the devastating effects of IPV by screening patients and providing resources when needed.
Facilitate disclosure using screening tools and protocol
In Ms. T’s case, evidence of violence was clearly visible. However, not all instances of IPV leave physical marks. The US Preventive Services Task Force (USPSTF) recommends that all women of childbearing age be screened for IPV, whether or not they exhibit signs of violence.8 While the USPSTF has only published recommendations regarding screening women for IPV, there has been a recent push to screen all patients given that men also experience high rates of IPV.9
Utilize a brief screening tool. Directly ask patients about IPV; this can help reduce stigma, facilitate disclosure, and initiate the process of connecting patients to potentially lifesaving resources. The USPSTF lists several brief screening measures that can be used in primary care settings to assess exposure to IPV (TABLE 18,10-17). The brevity of these screening tools makes them well suited for busy physicians; cutoff scores facilitate the rapid identification of positive screens. While the USPSTF has not made specific recommendations regarding a screening interval, many studies examining the utility of these measures have reported on annual screenings.8 While there is limited evidence that brief screening alone leads to reductions in IPV,8 discussing IPV in a supportive and empathic manner and connecting patients to resources, such as supportive counseling, does have an important benefit: It can reduce symptoms of depression.18

Continue to: Screen patients in private; this protocol can help
Screen patients in private; this protocol can help. Given the sensitive nature of IPV and the potential danger some patients may be facing, it is important to screen patients in a safe and supportive environment.19,20 Screening should be conducted by the primary care clinician, ideally when a trusting relationship already has been formed. Screen patients only when they are alone in a private room; avoid screening in public spaces such as clinic waiting rooms or in the vicinity of the patient’s partner or children older than age 2 years.19,20
To provide all patients with an opportunity for private and safe IPV screening, clinics are encouraged to develop a clinic-wide policy whereby patients are routinely escorted to the exam room alone for the first portion of their visit, after which any accompanying individuals may be invited to join.21 Clinic staff can inform patients and accompanying individuals of this policy when they first arrive. Once in the exam room, and before the screening process begins, clearly state reporting requirements to ensure that patients can make an informed decision about whether to disclose IPV.19
Set a receptive tone. The manner in which clinicians discuss IPV with their patients is just as important as the setting. Demonstrating sensitivity and genuine concern for the patient’s safety and well-being may increase the patient’s comfort level throughout the screening process and may facilitate disclosures of IPV.19,22 When screening patients for IPV, sit face to face rather than standing over them, maintain warm and open body language, and speak in a soft tone of voice.22
Patients may feel more comfortable if you ask screening questions in a straightforward, nonjudgmental manner, as this helps to normalize the screening experience. We also recommend using behaviorally specific language (eg, “Do arguments [with your partner] ever result in hitting, kicking, or pushing?”16 or “How often does your partner scream or curse at you?”),13 as some patients who have experienced IPV will not label their experiences as “abuse” or “violence.” Not every patient who experiences IPV will be ready to disclose these events; however, maintaining a positive and supportive relationship during routine IPV screening and throughout the remainder of the medical visit may help facilitate future disclosures if, and when, a patient is ready to seek support.19
CRITICAL INTERVENTION ELEMENTS: EMPATHY AND SAFETY
A physician’s response to an IPV disclosure can have a lasting impact on the patient. We encourage family physicians to respond to IPV disclosures with empathy. Maintain eye contact and warm body language, validate the patient’s experiences (“I am sorry this happened to you,” “that must have been terrifying”), tell the patient that the violence was not their fault, and thank the patient for disclosing.23
Continue to: Assess patient safety
Assess patient safety. Another critical component of intervention is to assess the patient’s safety and engage in safety planning. If the patient agrees to this next step, you may wish to provide a warm handoff to a trained social worker, nurse, or psychologist in the clinic who can spend more time covering this information with the patient. Some key components of a safety assessment include determining whether the violence or threat of violence is ongoing and identifying who lives in the home (eg, the partner, children, and any pets). You and the patient can also discuss red flags that would indicate elevated risk. You should discuss red flags that are unique to the patient’s relationship as well as common factors that have been found to heighten risk for IPV (eg, partner engaging in heavy alcohol use).1
With the patient’s permission, collaboratively construct a safety plan that details how the patient can stay safe on a daily basis and how to safely leave should a dangerous situation arise (TABLE 29,24). The interactive safety planning tool available on the National Domestic Violence Hotline’s website can be a valuable resource (www.thehotline.org/plan-for-safety/).24 Finally, if a patient is experiencing mental health concerns associated with IPV (eg, PTSD, depression, substance misuse, suicidal ideation), consider a referral to a domestic violence counseling center or mental health provider.

Move at the patient’s pace. Even if patients are willing to disclose IPV, they will differ in their readiness to discuss psychoeducation, safety planning, and referrals. Similarly, even if a patient is experiencing severe violence, they may not be ready to leave the relationship. Thus, it’s important to ask the patient for permission before initiating each successive step of the follow-up intervention. You and the patient may wish to schedule additional appointments to discuss this information at a pace the patient finds appropriate.
You may need to spend some time helping the patient recognize the severity of their situation and to feel empowered to take action. In addition, offer information and resources to all patients, even those who do not disclose IPV. Some patients may want to receive this information even if they do not feel comfortable sharing their experiences during the appointment.20 You can also inform patients that they are welcome to bring up issues related to IPV at any future appointments in order to leave the door open to future disclosures.
THE CASE
The physician determined that Ms. T had been experiencing physical and psychological IPV in her current relationship. After responding empathically and obtaining the patient’s consent, the physician provided a warm handoff to the psychologist in the clinic. With Ms. T’s permission, the psychologist provided psychoeducation about IPV, and they discussed Ms. T’s current situation and risk level. They determined that Ms. T was at risk for subsequent episodes of IPV and they collaborated on a safety plan, making sure to discuss contact information for local and national crisis resources.
Continue to: Ms. T saved the phone number...
Ms. T saved the phone number for her local domestic violence shelter in her phone under a false name in case her husband looked through her phone. She said she planned to work on several safety plan items when her husband was away from the house and it was safe to do so. For example, she planned to identify additional ways to exit the house in an emergency and she was going to put together a bag with a change of clothes and some money and drop it off at a trusted friend’s house.
Ms. T and the psychologist agreed to follow up with an office visit in 1 week to discuss any additional safety concerns and to determine whether Ms. T could benefit from a referral to domestic violence counseling services or mental health treatment. The psychologist provided a summary of the topics she and Ms. T had discussed to the physician. The physician scheduled a follow-up appointment with Ms. T in 3 weeks to assess her current safety, troubleshoot any difficulties in implementing her safety plan, and offer additional resources, as needed.
CORRESPONDENCE
Andrea Massa, PhD, 125 Doughty Street, Suite 300, Charleston, SC 29403; [email protected]
THE CASE
Louise T* is a 42-year-old woman who presented to her family medicine office for a routine annual visit. During the exam, her physician noticed bruises on Ms. T’s arms and back. Upon further inquiry, Ms. T reported that she and her husband had argued the night before the appointment. With some hesitancy, she went on to say that this was not the first time this had happened. She said that she and her husband had been arguing frequently for several years and that 6 months earlier, when he lost his job, he began hitting and pushing her.
●
*The patient’s name has been changed to protect her identity.
Intimate partner violence (IPV) includes physical, sexual, or psychological aggression or stalking perpetrated by a current or former relationship partner.1 IPV affects more than 12 million men and women living in the United States each year.2 According to a national survey of IPV, approximately one-third (35.6%) of women and one-quarter (28.5%) of men living in the United States experience rape, physical violence, or stalking by an intimate partner during their lifetime.2 Lifetime exposure to psychological IPV is even more prevalent, affecting nearly half of women and men (48.4% and 48.8%, respectively).2
Lifetime prevalence of any form of IPV is higher among women who identify as bisexual (59.8%) and lesbian (46.3%) compared with those who identify as heterosexual (37.2%); rates are comparable among men who identify as heterosexual (31.9%), bisexual (35.3%), and gay (35.1%).3 Preliminary data suggest that IPV may have increased in frequency and severity during the COVID-19 pandemic, particularly in the context of mandated shelter-in-place and stay-at-home orders.4-6
IPV is associated with numerous negative health consequences. They include fear and concern for safety, mental health disorders such as posttraumatic stress disorder (PTSD), and physical health problems including physical injury, chronic pain, sleep disturbance, and frequent headaches.2 IPV is also associated with a greater number of missed days from school and work and increased utilization of legal, health care, and housing services.2,7 The overall annual cost of IPV against women is estimated at $5.8 billion, with health care costs accounting for approximately $4.1 billion.7 Family physicians can play an important role in curbing the devastating effects of IPV by screening patients and providing resources when needed.
Facilitate disclosure using screening tools and protocol
In Ms. T’s case, evidence of violence was clearly visible. However, not all instances of IPV leave physical marks. The US Preventive Services Task Force (USPSTF) recommends that all women of childbearing age be screened for IPV, whether or not they exhibit signs of violence.8 While the USPSTF has only published recommendations regarding screening women for IPV, there has been a recent push to screen all patients given that men also experience high rates of IPV.9
Utilize a brief screening tool. Directly ask patients about IPV; this can help reduce stigma, facilitate disclosure, and initiate the process of connecting patients to potentially lifesaving resources. The USPSTF lists several brief screening measures that can be used in primary care settings to assess exposure to IPV (TABLE 18,10-17). The brevity of these screening tools makes them well suited for busy physicians; cutoff scores facilitate the rapid identification of positive screens. While the USPSTF has not made specific recommendations regarding a screening interval, many studies examining the utility of these measures have reported on annual screenings.8 While there is limited evidence that brief screening alone leads to reductions in IPV,8 discussing IPV in a supportive and empathic manner and connecting patients to resources, such as supportive counseling, does have an important benefit: It can reduce symptoms of depression.18

Continue to: Screen patients in private; this protocol can help
Screen patients in private; this protocol can help. Given the sensitive nature of IPV and the potential danger some patients may be facing, it is important to screen patients in a safe and supportive environment.19,20 Screening should be conducted by the primary care clinician, ideally when a trusting relationship already has been formed. Screen patients only when they are alone in a private room; avoid screening in public spaces such as clinic waiting rooms or in the vicinity of the patient’s partner or children older than age 2 years.19,20
To provide all patients with an opportunity for private and safe IPV screening, clinics are encouraged to develop a clinic-wide policy whereby patients are routinely escorted to the exam room alone for the first portion of their visit, after which any accompanying individuals may be invited to join.21 Clinic staff can inform patients and accompanying individuals of this policy when they first arrive. Once in the exam room, and before the screening process begins, clearly state reporting requirements to ensure that patients can make an informed decision about whether to disclose IPV.19
Set a receptive tone. The manner in which clinicians discuss IPV with their patients is just as important as the setting. Demonstrating sensitivity and genuine concern for the patient’s safety and well-being may increase the patient’s comfort level throughout the screening process and may facilitate disclosures of IPV.19,22 When screening patients for IPV, sit face to face rather than standing over them, maintain warm and open body language, and speak in a soft tone of voice.22
Patients may feel more comfortable if you ask screening questions in a straightforward, nonjudgmental manner, as this helps to normalize the screening experience. We also recommend using behaviorally specific language (eg, “Do arguments [with your partner] ever result in hitting, kicking, or pushing?”16 or “How often does your partner scream or curse at you?”),13 as some patients who have experienced IPV will not label their experiences as “abuse” or “violence.” Not every patient who experiences IPV will be ready to disclose these events; however, maintaining a positive and supportive relationship during routine IPV screening and throughout the remainder of the medical visit may help facilitate future disclosures if, and when, a patient is ready to seek support.19
CRITICAL INTERVENTION ELEMENTS: EMPATHY AND SAFETY
A physician’s response to an IPV disclosure can have a lasting impact on the patient. We encourage family physicians to respond to IPV disclosures with empathy. Maintain eye contact and warm body language, validate the patient’s experiences (“I am sorry this happened to you,” “that must have been terrifying”), tell the patient that the violence was not their fault, and thank the patient for disclosing.23
Continue to: Assess patient safety
Assess patient safety. Another critical component of intervention is to assess the patient’s safety and engage in safety planning. If the patient agrees to this next step, you may wish to provide a warm handoff to a trained social worker, nurse, or psychologist in the clinic who can spend more time covering this information with the patient. Some key components of a safety assessment include determining whether the violence or threat of violence is ongoing and identifying who lives in the home (eg, the partner, children, and any pets). You and the patient can also discuss red flags that would indicate elevated risk. You should discuss red flags that are unique to the patient’s relationship as well as common factors that have been found to heighten risk for IPV (eg, partner engaging in heavy alcohol use).1
With the patient’s permission, collaboratively construct a safety plan that details how the patient can stay safe on a daily basis and how to safely leave should a dangerous situation arise (TABLE 29,24). The interactive safety planning tool available on the National Domestic Violence Hotline’s website can be a valuable resource (www.thehotline.org/plan-for-safety/).24 Finally, if a patient is experiencing mental health concerns associated with IPV (eg, PTSD, depression, substance misuse, suicidal ideation), consider a referral to a domestic violence counseling center or mental health provider.

Move at the patient’s pace. Even if patients are willing to disclose IPV, they will differ in their readiness to discuss psychoeducation, safety planning, and referrals. Similarly, even if a patient is experiencing severe violence, they may not be ready to leave the relationship. Thus, it’s important to ask the patient for permission before initiating each successive step of the follow-up intervention. You and the patient may wish to schedule additional appointments to discuss this information at a pace the patient finds appropriate.
You may need to spend some time helping the patient recognize the severity of their situation and to feel empowered to take action. In addition, offer information and resources to all patients, even those who do not disclose IPV. Some patients may want to receive this information even if they do not feel comfortable sharing their experiences during the appointment.20 You can also inform patients that they are welcome to bring up issues related to IPV at any future appointments in order to leave the door open to future disclosures.
THE CASE
The physician determined that Ms. T had been experiencing physical and psychological IPV in her current relationship. After responding empathically and obtaining the patient’s consent, the physician provided a warm handoff to the psychologist in the clinic. With Ms. T’s permission, the psychologist provided psychoeducation about IPV, and they discussed Ms. T’s current situation and risk level. They determined that Ms. T was at risk for subsequent episodes of IPV and they collaborated on a safety plan, making sure to discuss contact information for local and national crisis resources.
Continue to: Ms. T saved the phone number...
Ms. T saved the phone number for her local domestic violence shelter in her phone under a false name in case her husband looked through her phone. She said she planned to work on several safety plan items when her husband was away from the house and it was safe to do so. For example, she planned to identify additional ways to exit the house in an emergency and she was going to put together a bag with a change of clothes and some money and drop it off at a trusted friend’s house.
Ms. T and the psychologist agreed to follow up with an office visit in 1 week to discuss any additional safety concerns and to determine whether Ms. T could benefit from a referral to domestic violence counseling services or mental health treatment. The psychologist provided a summary of the topics she and Ms. T had discussed to the physician. The physician scheduled a follow-up appointment with Ms. T in 3 weeks to assess her current safety, troubleshoot any difficulties in implementing her safety plan, and offer additional resources, as needed.
CORRESPONDENCE
Andrea Massa, PhD, 125 Doughty Street, Suite 300, Charleston, SC 29403; [email protected]
1. CDC. National Center for Injury Prevention and Control. Preventing intimate partner violence. 2021. Accessed June 27, 2022. www.cdc.gov/violenceprevention/intimatepartnerviolence/fastfact.html
2. CDC. Black MC, Basile KC, Breiding MJ, et al. The National Intimate Partner and Sexual Violence Survey: 2010 Summary Report. Accessed June 27, 2022. www.cdc.gov/violenceprevention/pdf/nisvs_executive_summary-a.pdf
3. Chen J, Walters ML, Gilbert LK, et al. Sexual violence, stalking, and intimate partner violence by sexual orientation, United States. Psychol Violence. 2020;10:110-119. doi:10.1037/vio0000252
4. Kofman YB, Garfin DR. Home is not always a haven: the domestic violence crisis amid the COVID-19 pandemic. Psychol Trauma. 2020;12:S199-S201. doi:10.1037/tra0000866
5. Lyons M, Brewer G. Experiences of intimate partner violence during lockdown and the COVID-19 pandemic. J Fam Violence. 2021:1-9. doi:10.1007/s10896-021-00260-x
6. Parrott DJ, Halmos MB, Stappenbeck CA, et al. Intimate partner aggression during the COVID-19 pandemic: associations with stress and heavy drinking. Psychol Violence. 2021;12:95-103. doi:10.1037/vio0000395
7. CDC. National Center for Injury Prevention and
8. US Preventive Services Task Force. Screening for intimate partner violence, elder abuse, and abuse of vulnerable adults: US Preventive Services Task Force final recommendation statement. JAMA. 2018;320:1678-1687. doi:10.1001/jama.2018.14741
9. Sprunger JG, Schumacher JA, Coffey SF, et al. It’s time to start asking all patients about intimate partner violence. J Fam Pract. 2019;68:152-161.
10. Chan CC, Chan YC, Au A, et al. Reliability and validity of the “Extended - Hurt, Insult, Threaten, Scream” (E-HITS) screening tool in detecting intimate partner violence in hospital emergency departments in Hong Kong. Hong Kong J Emerg Med. 2010;17:109-117. doi:10.1177/102490791001700202
11. Iverson KM, King MW, Gerber MR, et al. Accuracy of an intimate partner violence screening tool for female VHA patients: a replication and extension. J Trauma Stress. 2015;28:79-82. doi:10.1002/jts.21985
12. Sohal H, Eldridge S, Feder G. The sensitivity and specificity of four questions (HARK) to identify intimate partner violence: a diagnostic accuracy study in general practice. BMC Fam Pract. 2007;8:49. doi:10.1186/1471-2296-8-49
13. Sherin KM, Sinacore JM, Li X, et al. HITS: a short domestic violence screening tool for use in a family practice setting. Fam Med. 1998;30:508-512.
14. Rabin RF, Jennings JM, Campbell JC, et al. Intimate partner violence screening tools: a systematic review. Am J Prev Med. 2009;36:439-445.e4. doi:10.1016/j.amepre.2009.01.024
15. Feldhaus KM, Koziol-McLain J, Amsbury HL, et al. Accuracy of 3 brief screening questions for detecting partner violence in the emergency department. JAMA. 1997;277:1357-1361. doi:10.1001/jama.1997.03540410035027
16. Brown JB, Lent B, Schmidt G, et al. Application of the Woman Abuse Screening Tool (WAST) and WAST-short in the family practice setting. J Fam Pract. 2000;49:896-903.
17. Wathen CN, Jamieson E, MacMillan HL, MVAWRG. Who is identified by screening for intimate partner violence? Womens Health Issues. 2008;18:423-432. doi:10.1016/j.whi.2008.08.003
18. Hegarty K, O’Doherty L, Taft A, et al. Screening and counselling in the primary care setting for women who have experienced intimate partner violence (WEAVE): a cluster randomised controlled trial. Lancet. 2013;382:249-258. doi: 10.1016/S0140-6736(13)60052-5
19. Correa NP, Cain CM, Bertenthal M, et al. Women’s experiences of being screened for intimate partner violence in the health care setting. Nurs Womens Health. 2020;24:185-196. doi:10.1016/j.nwh.2020.04.002
20. Chang JC, Decker MR, Moracco KE, et al. Asking about intimate partner violence: advice from female survivors to health care providers. Patient Educ Couns. 2005;59:141-147. doi:10.1016/j.pec.2004.10.008
21. Paterno MT, Draughon JE. Screening for intimate partner violence. J Midwifery Womens Health. 2016;61:370-375. doi:10.1111/jmwh.12443
22. Iverson KM, Huang K, Wells SY, et al. Women veterans’ preferences for intimate partner violence screening and response procedures within the Veterans Health Administration. Res Nurs Health. 2014;37:302-311. doi:10.1002/nur.21602
23. National Sexual Violence Research Center. Assessing patients for sexual violence: A guide for health care providers. 2011. Accessed June 28, 2022. www.nsvrc.org/publications/assessing-patients-sexual-violence-guide-health-care-providers
24. National Domestic Violence Hotline. Interactive guide to safety planning. Accessed August 22, 2022. https://www.thehotline.org/plan-for-safety/create-a-safety-plan/
1. CDC. National Center for Injury Prevention and Control. Preventing intimate partner violence. 2021. Accessed June 27, 2022. www.cdc.gov/violenceprevention/intimatepartnerviolence/fastfact.html
2. CDC. Black MC, Basile KC, Breiding MJ, et al. The National Intimate Partner and Sexual Violence Survey: 2010 Summary Report. Accessed June 27, 2022. www.cdc.gov/violenceprevention/pdf/nisvs_executive_summary-a.pdf
3. Chen J, Walters ML, Gilbert LK, et al. Sexual violence, stalking, and intimate partner violence by sexual orientation, United States. Psychol Violence. 2020;10:110-119. doi:10.1037/vio0000252
4. Kofman YB, Garfin DR. Home is not always a haven: the domestic violence crisis amid the COVID-19 pandemic. Psychol Trauma. 2020;12:S199-S201. doi:10.1037/tra0000866
5. Lyons M, Brewer G. Experiences of intimate partner violence during lockdown and the COVID-19 pandemic. J Fam Violence. 2021:1-9. doi:10.1007/s10896-021-00260-x
6. Parrott DJ, Halmos MB, Stappenbeck CA, et al. Intimate partner aggression during the COVID-19 pandemic: associations with stress and heavy drinking. Psychol Violence. 2021;12:95-103. doi:10.1037/vio0000395
7. CDC. National Center for Injury Prevention and
8. US Preventive Services Task Force. Screening for intimate partner violence, elder abuse, and abuse of vulnerable adults: US Preventive Services Task Force final recommendation statement. JAMA. 2018;320:1678-1687. doi:10.1001/jama.2018.14741
9. Sprunger JG, Schumacher JA, Coffey SF, et al. It’s time to start asking all patients about intimate partner violence. J Fam Pract. 2019;68:152-161.
10. Chan CC, Chan YC, Au A, et al. Reliability and validity of the “Extended - Hurt, Insult, Threaten, Scream” (E-HITS) screening tool in detecting intimate partner violence in hospital emergency departments in Hong Kong. Hong Kong J Emerg Med. 2010;17:109-117. doi:10.1177/102490791001700202
11. Iverson KM, King MW, Gerber MR, et al. Accuracy of an intimate partner violence screening tool for female VHA patients: a replication and extension. J Trauma Stress. 2015;28:79-82. doi:10.1002/jts.21985
12. Sohal H, Eldridge S, Feder G. The sensitivity and specificity of four questions (HARK) to identify intimate partner violence: a diagnostic accuracy study in general practice. BMC Fam Pract. 2007;8:49. doi:10.1186/1471-2296-8-49
13. Sherin KM, Sinacore JM, Li X, et al. HITS: a short domestic violence screening tool for use in a family practice setting. Fam Med. 1998;30:508-512.
14. Rabin RF, Jennings JM, Campbell JC, et al. Intimate partner violence screening tools: a systematic review. Am J Prev Med. 2009;36:439-445.e4. doi:10.1016/j.amepre.2009.01.024
15. Feldhaus KM, Koziol-McLain J, Amsbury HL, et al. Accuracy of 3 brief screening questions for detecting partner violence in the emergency department. JAMA. 1997;277:1357-1361. doi:10.1001/jama.1997.03540410035027
16. Brown JB, Lent B, Schmidt G, et al. Application of the Woman Abuse Screening Tool (WAST) and WAST-short in the family practice setting. J Fam Pract. 2000;49:896-903.
17. Wathen CN, Jamieson E, MacMillan HL, MVAWRG. Who is identified by screening for intimate partner violence? Womens Health Issues. 2008;18:423-432. doi:10.1016/j.whi.2008.08.003
18. Hegarty K, O’Doherty L, Taft A, et al. Screening and counselling in the primary care setting for women who have experienced intimate partner violence (WEAVE): a cluster randomised controlled trial. Lancet. 2013;382:249-258. doi: 10.1016/S0140-6736(13)60052-5
19. Correa NP, Cain CM, Bertenthal M, et al. Women’s experiences of being screened for intimate partner violence in the health care setting. Nurs Womens Health. 2020;24:185-196. doi:10.1016/j.nwh.2020.04.002
20. Chang JC, Decker MR, Moracco KE, et al. Asking about intimate partner violence: advice from female survivors to health care providers. Patient Educ Couns. 2005;59:141-147. doi:10.1016/j.pec.2004.10.008
21. Paterno MT, Draughon JE. Screening for intimate partner violence. J Midwifery Womens Health. 2016;61:370-375. doi:10.1111/jmwh.12443
22. Iverson KM, Huang K, Wells SY, et al. Women veterans’ preferences for intimate partner violence screening and response procedures within the Veterans Health Administration. Res Nurs Health. 2014;37:302-311. doi:10.1002/nur.21602
23. National Sexual Violence Research Center. Assessing patients for sexual violence: A guide for health care providers. 2011. Accessed June 28, 2022. www.nsvrc.org/publications/assessing-patients-sexual-violence-guide-health-care-providers
24. National Domestic Violence Hotline. Interactive guide to safety planning. Accessed August 22, 2022. https://www.thehotline.org/plan-for-safety/create-a-safety-plan/
Congenital cytomegalovirus declined in wake of COVID-19
Congenital cytomegalovirus cases declined significantly during the COVID-19 pandemic, compared with a period before the pandemic, based on data from nearly 20,000 newborns.
A study originated to explore racial and ethnic differences in congenital cytomegalovirus (cCMV) began in 2016, but was halted in April 2020 because of the COVID-19 pandemic, wrote Mark R. Schleiss, MD, of the University of Minnesota, Minneapolis, and colleagues. The study resumed for a period from August 2020 to December 2021, and the researchers compared data on cCMV before and during the pandemic. The prepandemic period included data from April 2016 to March 2020.
“We have been screening for congenital CMV infection in Minnesota for 6 years as a part of a multicenter collaborative study that I lead as the primary investigator,” Dr. Schleiss said in an interview. “Our efforts have contributed to the decision, vetted through the Minnesota Legislature and signed into law in 2021 (the “Vivian Act”), to begin universal screening for all newborns in Minnesota in 2023. In the context of this ongoing screening/surveillance study, it was important and scientifically very interesting to examine the impact of the COVID-19 pandemic on the risk of congenital CMV infection,” he explained.
The findings were published in a research letter in JAMA Network Open. A total of 15,697 newborns were screened before the pandemic and 4,222 were screened during the pandemic period at six hospitals. The majority of the mothers participating during the prepandemic and pandemic periods were non-Hispanic White (71% and 60%, respectively).
Overall, the percentage screened prevalence for cCMV was 79% in the prepandemic period and 21% during the pandemic, with rates of 4.5 per 1,000 and 1.4 per 1,000, respectively.
Although the highest percentage of cCMV cases occurred in newborns of mothers aged 25 years and older (86%), the prevalence was highest among newborns of mothers aged 24 years and younger (6.0 per 1,000). The prevalence of cCMV overall was higher in infants of non-Hispanic Black mothers vs. non-Hispanic White mothers, but not significantly different (5.1 per 1,000 vs. 4.6 per 1,000) and among second newborns vs. first newborns (6.0 vs. 3.2 per 1,000, respectively).
Factors related to COVID-19, including reduced day care attendance, behavioral changes, and mitigation measures at childcare facilities such as smaller classes and increased hand hygiene and disinfection may have contributed to this decrease in cCMV in the pandemic period, the researchers wrote in their discussion.
The comparable prevalence in newborns of non-Hispanic Black and White mothers contrasts with previous studies showing a higher prevalence in children of non-Hispanic Black mothers, the researchers noted in their discussion.
The study was limited by several factors, including the variation in time points for enrollment at different sites and the exclusion of families in the newborn nursery with positive COVID-19 results during the pandemic, they wrote. More research is needed on the potential effects of behavioral interventions to reduce CMV risk during pregnancy, as well as future CMV vaccination for childbearing-aged women and young children, they concluded.
However, the researchers were surprised by the impact of COVID-19 on the prevalence of cCMV, Dr. Schleiss said in an interview. “We have had the knowledge for many years that CMV infections in young women are commonly acquired through interactions with their toddlers. These interactions – sharing food, wiping drool and nasal discharge from the toddler’s nose, changing diapers, kissing the child on the mouth – can transmit CMV,” he said. In addition, toddlers may acquire CMV from group day care; the child then sheds CMV and transmits the virus to their pregnant mother, who then transmits the virus across the placenta, leading to cCMV infection in the newborn, Dr. Schleiss explained.
Although the researchers expected a decrease in CMV in the wake of closures of group day care, increased home schooling, decreased interactions among children, hygienic precautions, and social isolation, the decrease exceeded their expectations, said Dr. Schleiss. “Our previous work showed that in the 5-year period leading up to the pandemic, about one baby in every 200 births was born with CMV. Between August 2020 and December 2021, the number decreased to one baby in every 1,000 births,” a difference he and his team found striking.
The message from the study is that CMV can be prevented, said Dr. Schleiss. “Hygienic precautions during pregnancy had a big impact. Since congenital CMV infection is the most common congenital infection in the United States, and probably globally, that causes disabilities in children, the implications are highly significant,” he said. “The hygienic precautions we all have engaged in during the pandemic, such as masking, handwashing, and infection prevention behaviors, were almost certainly responsible for the reduction in CMV transmission, which in turn protected mothers and newborns from the potentially devastating effects of the CMV virus,” he noted.
Looking ahead, “Vaccines are moving forward in clinical trials that aim to confer immunity on young women of childbearing age to protect future pregnancies against transmission of CMV to the newborn infant; it would be very important to examine in future studies whether hygienic precautions would have the same impact as a potential vaccine,” Dr. Schleiss said. More research is needed to examine the effect of education of women about CMV transmission, he added. “We think it is very important to share this knowledge from our study with the pediatric community, since pediatricians can be important in counseling women about future pregnancies and the risks of CMV acquisition and transmission,” he noted.
Implications for other viruses
Although CMV poses minimal risk for healthy populations, irreversible complications for infants born with congenital CMV, especially hearing loss, are very concerning, said Catherine Haut, DNP, CPNP-AC/PC, a pediatric nurse practitioner in Rehoboth Beach, Del., in an interview.
“The study of viral transmission during a time of isolation, masking, and other mitigation procedures for COVID-19 assists in awareness that other viruses may also be limited with the use of these measures,” she said.
Dr. Haut was not surprised by the findings, given that CMV is transmitted primarily through direct contact with body fluids and that more than 50% of American adults have been infected by age 40, according to the Centers for Disease Control and Prevention, she said.
The take-home message for pediatricians, Dr. Haut said, is measures to prevent transmission of viral infection can yield significant positive health outcomes for the pediatric population; however, the effect of isolation, which has been associated with a higher rate of mental health problems, should not be ignored.
“Despite appropriate statistical analyses and presentation of findings in this study, the population sampled during the pandemic was less than 30% of the pre-COVID sampling, representing a study limitation,” and conducting research in a single state limits generalizability, Dr. Haut noted. “I agree with the authors that additional study is necessary to better understand prevention measures and apply these methods to reduce CMV transmission. Pursuit of CMV immunization opportunities is also needed,” she said.
The study was supported by the Centers for Disease Control and Prevention, the National Vaccine Program Office, the Minnesota Department of Health Newborn Screening Program, and the University of South Carolina Disability Research and Dissemination Center. Lead author Dr. Schleiss disclosed grants from the CDC, the National Institutes of Health, and the DRDC during the conduct of the study; he also disclosed receiving personal fees from Moderna, Sanofi, GlaxoSmithKline, and Merck unrelated to the study. Dr. Haut had no financial conflicts to disclose and serves on the Editorial Advisory Board of Pediatric News.
Congenital cytomegalovirus cases declined significantly during the COVID-19 pandemic, compared with a period before the pandemic, based on data from nearly 20,000 newborns.
A study originated to explore racial and ethnic differences in congenital cytomegalovirus (cCMV) began in 2016, but was halted in April 2020 because of the COVID-19 pandemic, wrote Mark R. Schleiss, MD, of the University of Minnesota, Minneapolis, and colleagues. The study resumed for a period from August 2020 to December 2021, and the researchers compared data on cCMV before and during the pandemic. The prepandemic period included data from April 2016 to March 2020.
“We have been screening for congenital CMV infection in Minnesota for 6 years as a part of a multicenter collaborative study that I lead as the primary investigator,” Dr. Schleiss said in an interview. “Our efforts have contributed to the decision, vetted through the Minnesota Legislature and signed into law in 2021 (the “Vivian Act”), to begin universal screening for all newborns in Minnesota in 2023. In the context of this ongoing screening/surveillance study, it was important and scientifically very interesting to examine the impact of the COVID-19 pandemic on the risk of congenital CMV infection,” he explained.
The findings were published in a research letter in JAMA Network Open. A total of 15,697 newborns were screened before the pandemic and 4,222 were screened during the pandemic period at six hospitals. The majority of the mothers participating during the prepandemic and pandemic periods were non-Hispanic White (71% and 60%, respectively).
Overall, the percentage screened prevalence for cCMV was 79% in the prepandemic period and 21% during the pandemic, with rates of 4.5 per 1,000 and 1.4 per 1,000, respectively.
Although the highest percentage of cCMV cases occurred in newborns of mothers aged 25 years and older (86%), the prevalence was highest among newborns of mothers aged 24 years and younger (6.0 per 1,000). The prevalence of cCMV overall was higher in infants of non-Hispanic Black mothers vs. non-Hispanic White mothers, but not significantly different (5.1 per 1,000 vs. 4.6 per 1,000) and among second newborns vs. first newborns (6.0 vs. 3.2 per 1,000, respectively).
Factors related to COVID-19, including reduced day care attendance, behavioral changes, and mitigation measures at childcare facilities such as smaller classes and increased hand hygiene and disinfection may have contributed to this decrease in cCMV in the pandemic period, the researchers wrote in their discussion.
The comparable prevalence in newborns of non-Hispanic Black and White mothers contrasts with previous studies showing a higher prevalence in children of non-Hispanic Black mothers, the researchers noted in their discussion.
The study was limited by several factors, including the variation in time points for enrollment at different sites and the exclusion of families in the newborn nursery with positive COVID-19 results during the pandemic, they wrote. More research is needed on the potential effects of behavioral interventions to reduce CMV risk during pregnancy, as well as future CMV vaccination for childbearing-aged women and young children, they concluded.
However, the researchers were surprised by the impact of COVID-19 on the prevalence of cCMV, Dr. Schleiss said in an interview. “We have had the knowledge for many years that CMV infections in young women are commonly acquired through interactions with their toddlers. These interactions – sharing food, wiping drool and nasal discharge from the toddler’s nose, changing diapers, kissing the child on the mouth – can transmit CMV,” he said. In addition, toddlers may acquire CMV from group day care; the child then sheds CMV and transmits the virus to their pregnant mother, who then transmits the virus across the placenta, leading to cCMV infection in the newborn, Dr. Schleiss explained.
Although the researchers expected a decrease in CMV in the wake of closures of group day care, increased home schooling, decreased interactions among children, hygienic precautions, and social isolation, the decrease exceeded their expectations, said Dr. Schleiss. “Our previous work showed that in the 5-year period leading up to the pandemic, about one baby in every 200 births was born with CMV. Between August 2020 and December 2021, the number decreased to one baby in every 1,000 births,” a difference he and his team found striking.
The message from the study is that CMV can be prevented, said Dr. Schleiss. “Hygienic precautions during pregnancy had a big impact. Since congenital CMV infection is the most common congenital infection in the United States, and probably globally, that causes disabilities in children, the implications are highly significant,” he said. “The hygienic precautions we all have engaged in during the pandemic, such as masking, handwashing, and infection prevention behaviors, were almost certainly responsible for the reduction in CMV transmission, which in turn protected mothers and newborns from the potentially devastating effects of the CMV virus,” he noted.
Looking ahead, “Vaccines are moving forward in clinical trials that aim to confer immunity on young women of childbearing age to protect future pregnancies against transmission of CMV to the newborn infant; it would be very important to examine in future studies whether hygienic precautions would have the same impact as a potential vaccine,” Dr. Schleiss said. More research is needed to examine the effect of education of women about CMV transmission, he added. “We think it is very important to share this knowledge from our study with the pediatric community, since pediatricians can be important in counseling women about future pregnancies and the risks of CMV acquisition and transmission,” he noted.
Implications for other viruses
Although CMV poses minimal risk for healthy populations, irreversible complications for infants born with congenital CMV, especially hearing loss, are very concerning, said Catherine Haut, DNP, CPNP-AC/PC, a pediatric nurse practitioner in Rehoboth Beach, Del., in an interview.
“The study of viral transmission during a time of isolation, masking, and other mitigation procedures for COVID-19 assists in awareness that other viruses may also be limited with the use of these measures,” she said.
Dr. Haut was not surprised by the findings, given that CMV is transmitted primarily through direct contact with body fluids and that more than 50% of American adults have been infected by age 40, according to the Centers for Disease Control and Prevention, she said.
The take-home message for pediatricians, Dr. Haut said, is measures to prevent transmission of viral infection can yield significant positive health outcomes for the pediatric population; however, the effect of isolation, which has been associated with a higher rate of mental health problems, should not be ignored.
“Despite appropriate statistical analyses and presentation of findings in this study, the population sampled during the pandemic was less than 30% of the pre-COVID sampling, representing a study limitation,” and conducting research in a single state limits generalizability, Dr. Haut noted. “I agree with the authors that additional study is necessary to better understand prevention measures and apply these methods to reduce CMV transmission. Pursuit of CMV immunization opportunities is also needed,” she said.
The study was supported by the Centers for Disease Control and Prevention, the National Vaccine Program Office, the Minnesota Department of Health Newborn Screening Program, and the University of South Carolina Disability Research and Dissemination Center. Lead author Dr. Schleiss disclosed grants from the CDC, the National Institutes of Health, and the DRDC during the conduct of the study; he also disclosed receiving personal fees from Moderna, Sanofi, GlaxoSmithKline, and Merck unrelated to the study. Dr. Haut had no financial conflicts to disclose and serves on the Editorial Advisory Board of Pediatric News.
Congenital cytomegalovirus cases declined significantly during the COVID-19 pandemic, compared with a period before the pandemic, based on data from nearly 20,000 newborns.
A study originated to explore racial and ethnic differences in congenital cytomegalovirus (cCMV) began in 2016, but was halted in April 2020 because of the COVID-19 pandemic, wrote Mark R. Schleiss, MD, of the University of Minnesota, Minneapolis, and colleagues. The study resumed for a period from August 2020 to December 2021, and the researchers compared data on cCMV before and during the pandemic. The prepandemic period included data from April 2016 to March 2020.
“We have been screening for congenital CMV infection in Minnesota for 6 years as a part of a multicenter collaborative study that I lead as the primary investigator,” Dr. Schleiss said in an interview. “Our efforts have contributed to the decision, vetted through the Minnesota Legislature and signed into law in 2021 (the “Vivian Act”), to begin universal screening for all newborns in Minnesota in 2023. In the context of this ongoing screening/surveillance study, it was important and scientifically very interesting to examine the impact of the COVID-19 pandemic on the risk of congenital CMV infection,” he explained.
The findings were published in a research letter in JAMA Network Open. A total of 15,697 newborns were screened before the pandemic and 4,222 were screened during the pandemic period at six hospitals. The majority of the mothers participating during the prepandemic and pandemic periods were non-Hispanic White (71% and 60%, respectively).
Overall, the percentage screened prevalence for cCMV was 79% in the prepandemic period and 21% during the pandemic, with rates of 4.5 per 1,000 and 1.4 per 1,000, respectively.
Although the highest percentage of cCMV cases occurred in newborns of mothers aged 25 years and older (86%), the prevalence was highest among newborns of mothers aged 24 years and younger (6.0 per 1,000). The prevalence of cCMV overall was higher in infants of non-Hispanic Black mothers vs. non-Hispanic White mothers, but not significantly different (5.1 per 1,000 vs. 4.6 per 1,000) and among second newborns vs. first newborns (6.0 vs. 3.2 per 1,000, respectively).
Factors related to COVID-19, including reduced day care attendance, behavioral changes, and mitigation measures at childcare facilities such as smaller classes and increased hand hygiene and disinfection may have contributed to this decrease in cCMV in the pandemic period, the researchers wrote in their discussion.
The comparable prevalence in newborns of non-Hispanic Black and White mothers contrasts with previous studies showing a higher prevalence in children of non-Hispanic Black mothers, the researchers noted in their discussion.
The study was limited by several factors, including the variation in time points for enrollment at different sites and the exclusion of families in the newborn nursery with positive COVID-19 results during the pandemic, they wrote. More research is needed on the potential effects of behavioral interventions to reduce CMV risk during pregnancy, as well as future CMV vaccination for childbearing-aged women and young children, they concluded.
However, the researchers were surprised by the impact of COVID-19 on the prevalence of cCMV, Dr. Schleiss said in an interview. “We have had the knowledge for many years that CMV infections in young women are commonly acquired through interactions with their toddlers. These interactions – sharing food, wiping drool and nasal discharge from the toddler’s nose, changing diapers, kissing the child on the mouth – can transmit CMV,” he said. In addition, toddlers may acquire CMV from group day care; the child then sheds CMV and transmits the virus to their pregnant mother, who then transmits the virus across the placenta, leading to cCMV infection in the newborn, Dr. Schleiss explained.
Although the researchers expected a decrease in CMV in the wake of closures of group day care, increased home schooling, decreased interactions among children, hygienic precautions, and social isolation, the decrease exceeded their expectations, said Dr. Schleiss. “Our previous work showed that in the 5-year period leading up to the pandemic, about one baby in every 200 births was born with CMV. Between August 2020 and December 2021, the number decreased to one baby in every 1,000 births,” a difference he and his team found striking.
The message from the study is that CMV can be prevented, said Dr. Schleiss. “Hygienic precautions during pregnancy had a big impact. Since congenital CMV infection is the most common congenital infection in the United States, and probably globally, that causes disabilities in children, the implications are highly significant,” he said. “The hygienic precautions we all have engaged in during the pandemic, such as masking, handwashing, and infection prevention behaviors, were almost certainly responsible for the reduction in CMV transmission, which in turn protected mothers and newborns from the potentially devastating effects of the CMV virus,” he noted.
Looking ahead, “Vaccines are moving forward in clinical trials that aim to confer immunity on young women of childbearing age to protect future pregnancies against transmission of CMV to the newborn infant; it would be very important to examine in future studies whether hygienic precautions would have the same impact as a potential vaccine,” Dr. Schleiss said. More research is needed to examine the effect of education of women about CMV transmission, he added. “We think it is very important to share this knowledge from our study with the pediatric community, since pediatricians can be important in counseling women about future pregnancies and the risks of CMV acquisition and transmission,” he noted.
Implications for other viruses
Although CMV poses minimal risk for healthy populations, irreversible complications for infants born with congenital CMV, especially hearing loss, are very concerning, said Catherine Haut, DNP, CPNP-AC/PC, a pediatric nurse practitioner in Rehoboth Beach, Del., in an interview.
“The study of viral transmission during a time of isolation, masking, and other mitigation procedures for COVID-19 assists in awareness that other viruses may also be limited with the use of these measures,” she said.
Dr. Haut was not surprised by the findings, given that CMV is transmitted primarily through direct contact with body fluids and that more than 50% of American adults have been infected by age 40, according to the Centers for Disease Control and Prevention, she said.
The take-home message for pediatricians, Dr. Haut said, is measures to prevent transmission of viral infection can yield significant positive health outcomes for the pediatric population; however, the effect of isolation, which has been associated with a higher rate of mental health problems, should not be ignored.
“Despite appropriate statistical analyses and presentation of findings in this study, the population sampled during the pandemic was less than 30% of the pre-COVID sampling, representing a study limitation,” and conducting research in a single state limits generalizability, Dr. Haut noted. “I agree with the authors that additional study is necessary to better understand prevention measures and apply these methods to reduce CMV transmission. Pursuit of CMV immunization opportunities is also needed,” she said.
The study was supported by the Centers for Disease Control and Prevention, the National Vaccine Program Office, the Minnesota Department of Health Newborn Screening Program, and the University of South Carolina Disability Research and Dissemination Center. Lead author Dr. Schleiss disclosed grants from the CDC, the National Institutes of Health, and the DRDC during the conduct of the study; he also disclosed receiving personal fees from Moderna, Sanofi, GlaxoSmithKline, and Merck unrelated to the study. Dr. Haut had no financial conflicts to disclose and serves on the Editorial Advisory Board of Pediatric News.
FROM JAMA NETWORK OPEN
Isotretinoin prescribers need better education on emergency contraception
, in a survey of 57 clinicians.
Pregnancies among patients on isotretinoin have declined since the iPLEDGE risk management program was introduced in 2005, but from 2011 to 2017, 210 to 310 pregnancies were reported to the Food and Drug Administration every year, wrote Catherine E. Smiley of Penn State University, Hershey, Pa., and coauthors Melissa Butt, DrPH, and Andrea L. Zaenglein, MD, of Penn State.
For patients on isotretinoin, EC “becomes critical when abstinence fails or contraception is not used properly,” but EC merits only a brief mention in iPLEDGE materials for patients and providers, they noted.
Patients on isotretinoin who choose abstinence as their form of birth control are the group at greatest risk for pregnancy, Dr. Zaenglein, professor of dermatology and pediatric dermatology, Penn State University, said in an interview. “However, the iPLEDGE program fails to educate patients adequately on emergency contraception,” she explained.
To assess pediatric dermatologists’ understanding of EC and their contraception counseling practices for isotretinoin patients, the researchers surveyed 57 pediatric dermatologists who prescribed isotretinoin as part of their practices. The findings were published in Pediatric Dermatology.Respondents included 53 practicing dermatologists, 2 residents, and 2 fellows. Approximately one-third (31.6%) had been in practice for 6-10 years, almost 23% had been in practice for 3-5 years, and almost 20% had been in practice for 21 or more years. Almost two-thirds practiced pediatric dermatology only.
Overall, 58% of the respondents strongly agreed that they provided contraception counseling to patients at their initial visit for isotretinoin, but only 7% and 3.5% reported providing EC counseling at initial and follow-up visits, respectively. More than half (58%) said they did not counsel patients on the side effects of EC.
As for provider education, 7.1% of respondents said they had received formal education on EC counseling, 25% reported receiving informal education on EC counseling, and 68% said they received no education on EC counseling.
A total of 32% of respondents said they were at least somewhat confident in how to obtain EC in their state.
EC is an effective form of contraception if used after unprotected intercourse, and discounts can reduce the price to as low as $9.69, the researchers wrote in their discussion. “Given that most providers in this study did not receive formal education on EC, and most do not provide EC counseling to their patients of reproductive potential on isotretinoin, EC education should be a core competency in dermatology residency education on isotretinoin prescribing,” the researchers noted. In addition, EC counseling in the iPLEDGE program should be improved by including more information in education materials and reminding patients that EC is an option, they said.
The study findings were limited by several factors including the small sample size and the multiple-choice format that prevented respondents to share rationales for their responses, the researchers noted.
However, the results highlight the need to improve EC education among pediatric dermatologists to better inform patients considering isotretinoin, especially those choosing abstinence as a method of birth control, they emphasized.
“This study is very important at this specific time for two reasons,” Dr. Zaenglein said in an interview. “The first is that with the recent disastrous rollout of the new iPLEDGE changes, there have been many calls to reform the REMS program. For the first time in the 22-year history of the program, the isotretinoin manufacturers, who manage the iPLEDGE program as an unidentified group (the IPMG), have been forced by the FDA to meet with the AAD iPLEDGE Task Force,” said Dr. Zaenglein, a member of the task force.
“The task force is currently advocating for common sense changes to iPLEDGE and I think enhancing education on emergency contraception is vital to the goal of the program, stated as ‘to manage the risk of isotretinoin’s teratogenicity and to minimize fetal exposure,’ ” she added. For many patients who previously became pregnant on isotretinoin, Plan B, an over-the-counter, FDA-approved form of contraception, might have prevented that pregnancy if the patients received adequate education on EC, she said.
The current study is especially relevant now, said Dr. Zaenglein. “With the reversal of Roe v. Wade, access to abortion is restricted or completely banned in many states, which makes educating our patients on how to prevent pregnancy even more important.”
Dr. Zaenglein said she was “somewhat surprised” by how many respondents were not educating their isotretinoin patients on EC. “However, these results follow a known trend among dermatologists. Only 50% of dermatologists prescribe oral contraceptives for acne, despite its being an FDA-approved treatment for the most common dermatologic condition we see in adolescents and young adults,” she noted.
“In general, dermatologists, and subsequently dermatology residents, are poorly educated on issues of reproductive health and how they are relevant to dermatologic care,” she added.
Dr. Zaenglein’s take home message: “Dermatologists should educate all patients of childbearing potential taking isotretinoin on how to acquire and use emergency contraception at every visit.” As for additional research, she said that since the study was conducted with pediatric dermatologists, “it would be very interesting to see if general dermatologists had the same lack of comfort in educating patients on emergency contraception and what their standard counseling practices are.”
The study received no outside funding. Dr. Zaenglein is a member of the AAD’s iPLEDGE Work Group and serves as an editor-in-chief of Pediatric Dermatology.
, in a survey of 57 clinicians.
Pregnancies among patients on isotretinoin have declined since the iPLEDGE risk management program was introduced in 2005, but from 2011 to 2017, 210 to 310 pregnancies were reported to the Food and Drug Administration every year, wrote Catherine E. Smiley of Penn State University, Hershey, Pa., and coauthors Melissa Butt, DrPH, and Andrea L. Zaenglein, MD, of Penn State.
For patients on isotretinoin, EC “becomes critical when abstinence fails or contraception is not used properly,” but EC merits only a brief mention in iPLEDGE materials for patients and providers, they noted.
Patients on isotretinoin who choose abstinence as their form of birth control are the group at greatest risk for pregnancy, Dr. Zaenglein, professor of dermatology and pediatric dermatology, Penn State University, said in an interview. “However, the iPLEDGE program fails to educate patients adequately on emergency contraception,” she explained.
To assess pediatric dermatologists’ understanding of EC and their contraception counseling practices for isotretinoin patients, the researchers surveyed 57 pediatric dermatologists who prescribed isotretinoin as part of their practices. The findings were published in Pediatric Dermatology.Respondents included 53 practicing dermatologists, 2 residents, and 2 fellows. Approximately one-third (31.6%) had been in practice for 6-10 years, almost 23% had been in practice for 3-5 years, and almost 20% had been in practice for 21 or more years. Almost two-thirds practiced pediatric dermatology only.
Overall, 58% of the respondents strongly agreed that they provided contraception counseling to patients at their initial visit for isotretinoin, but only 7% and 3.5% reported providing EC counseling at initial and follow-up visits, respectively. More than half (58%) said they did not counsel patients on the side effects of EC.
As for provider education, 7.1% of respondents said they had received formal education on EC counseling, 25% reported receiving informal education on EC counseling, and 68% said they received no education on EC counseling.
A total of 32% of respondents said they were at least somewhat confident in how to obtain EC in their state.
EC is an effective form of contraception if used after unprotected intercourse, and discounts can reduce the price to as low as $9.69, the researchers wrote in their discussion. “Given that most providers in this study did not receive formal education on EC, and most do not provide EC counseling to their patients of reproductive potential on isotretinoin, EC education should be a core competency in dermatology residency education on isotretinoin prescribing,” the researchers noted. In addition, EC counseling in the iPLEDGE program should be improved by including more information in education materials and reminding patients that EC is an option, they said.
The study findings were limited by several factors including the small sample size and the multiple-choice format that prevented respondents to share rationales for their responses, the researchers noted.
However, the results highlight the need to improve EC education among pediatric dermatologists to better inform patients considering isotretinoin, especially those choosing abstinence as a method of birth control, they emphasized.
“This study is very important at this specific time for two reasons,” Dr. Zaenglein said in an interview. “The first is that with the recent disastrous rollout of the new iPLEDGE changes, there have been many calls to reform the REMS program. For the first time in the 22-year history of the program, the isotretinoin manufacturers, who manage the iPLEDGE program as an unidentified group (the IPMG), have been forced by the FDA to meet with the AAD iPLEDGE Task Force,” said Dr. Zaenglein, a member of the task force.
“The task force is currently advocating for common sense changes to iPLEDGE and I think enhancing education on emergency contraception is vital to the goal of the program, stated as ‘to manage the risk of isotretinoin’s teratogenicity and to minimize fetal exposure,’ ” she added. For many patients who previously became pregnant on isotretinoin, Plan B, an over-the-counter, FDA-approved form of contraception, might have prevented that pregnancy if the patients received adequate education on EC, she said.
The current study is especially relevant now, said Dr. Zaenglein. “With the reversal of Roe v. Wade, access to abortion is restricted or completely banned in many states, which makes educating our patients on how to prevent pregnancy even more important.”
Dr. Zaenglein said she was “somewhat surprised” by how many respondents were not educating their isotretinoin patients on EC. “However, these results follow a known trend among dermatologists. Only 50% of dermatologists prescribe oral contraceptives for acne, despite its being an FDA-approved treatment for the most common dermatologic condition we see in adolescents and young adults,” she noted.
“In general, dermatologists, and subsequently dermatology residents, are poorly educated on issues of reproductive health and how they are relevant to dermatologic care,” she added.
Dr. Zaenglein’s take home message: “Dermatologists should educate all patients of childbearing potential taking isotretinoin on how to acquire and use emergency contraception at every visit.” As for additional research, she said that since the study was conducted with pediatric dermatologists, “it would be very interesting to see if general dermatologists had the same lack of comfort in educating patients on emergency contraception and what their standard counseling practices are.”
The study received no outside funding. Dr. Zaenglein is a member of the AAD’s iPLEDGE Work Group and serves as an editor-in-chief of Pediatric Dermatology.
, in a survey of 57 clinicians.
Pregnancies among patients on isotretinoin have declined since the iPLEDGE risk management program was introduced in 2005, but from 2011 to 2017, 210 to 310 pregnancies were reported to the Food and Drug Administration every year, wrote Catherine E. Smiley of Penn State University, Hershey, Pa., and coauthors Melissa Butt, DrPH, and Andrea L. Zaenglein, MD, of Penn State.
For patients on isotretinoin, EC “becomes critical when abstinence fails or contraception is not used properly,” but EC merits only a brief mention in iPLEDGE materials for patients and providers, they noted.
Patients on isotretinoin who choose abstinence as their form of birth control are the group at greatest risk for pregnancy, Dr. Zaenglein, professor of dermatology and pediatric dermatology, Penn State University, said in an interview. “However, the iPLEDGE program fails to educate patients adequately on emergency contraception,” she explained.
To assess pediatric dermatologists’ understanding of EC and their contraception counseling practices for isotretinoin patients, the researchers surveyed 57 pediatric dermatologists who prescribed isotretinoin as part of their practices. The findings were published in Pediatric Dermatology.Respondents included 53 practicing dermatologists, 2 residents, and 2 fellows. Approximately one-third (31.6%) had been in practice for 6-10 years, almost 23% had been in practice for 3-5 years, and almost 20% had been in practice for 21 or more years. Almost two-thirds practiced pediatric dermatology only.
Overall, 58% of the respondents strongly agreed that they provided contraception counseling to patients at their initial visit for isotretinoin, but only 7% and 3.5% reported providing EC counseling at initial and follow-up visits, respectively. More than half (58%) said they did not counsel patients on the side effects of EC.
As for provider education, 7.1% of respondents said they had received formal education on EC counseling, 25% reported receiving informal education on EC counseling, and 68% said they received no education on EC counseling.
A total of 32% of respondents said they were at least somewhat confident in how to obtain EC in their state.
EC is an effective form of contraception if used after unprotected intercourse, and discounts can reduce the price to as low as $9.69, the researchers wrote in their discussion. “Given that most providers in this study did not receive formal education on EC, and most do not provide EC counseling to their patients of reproductive potential on isotretinoin, EC education should be a core competency in dermatology residency education on isotretinoin prescribing,” the researchers noted. In addition, EC counseling in the iPLEDGE program should be improved by including more information in education materials and reminding patients that EC is an option, they said.
The study findings were limited by several factors including the small sample size and the multiple-choice format that prevented respondents to share rationales for their responses, the researchers noted.
However, the results highlight the need to improve EC education among pediatric dermatologists to better inform patients considering isotretinoin, especially those choosing abstinence as a method of birth control, they emphasized.
“This study is very important at this specific time for two reasons,” Dr. Zaenglein said in an interview. “The first is that with the recent disastrous rollout of the new iPLEDGE changes, there have been many calls to reform the REMS program. For the first time in the 22-year history of the program, the isotretinoin manufacturers, who manage the iPLEDGE program as an unidentified group (the IPMG), have been forced by the FDA to meet with the AAD iPLEDGE Task Force,” said Dr. Zaenglein, a member of the task force.
“The task force is currently advocating for common sense changes to iPLEDGE and I think enhancing education on emergency contraception is vital to the goal of the program, stated as ‘to manage the risk of isotretinoin’s teratogenicity and to minimize fetal exposure,’ ” she added. For many patients who previously became pregnant on isotretinoin, Plan B, an over-the-counter, FDA-approved form of contraception, might have prevented that pregnancy if the patients received adequate education on EC, she said.
The current study is especially relevant now, said Dr. Zaenglein. “With the reversal of Roe v. Wade, access to abortion is restricted or completely banned in many states, which makes educating our patients on how to prevent pregnancy even more important.”
Dr. Zaenglein said she was “somewhat surprised” by how many respondents were not educating their isotretinoin patients on EC. “However, these results follow a known trend among dermatologists. Only 50% of dermatologists prescribe oral contraceptives for acne, despite its being an FDA-approved treatment for the most common dermatologic condition we see in adolescents and young adults,” she noted.
“In general, dermatologists, and subsequently dermatology residents, are poorly educated on issues of reproductive health and how they are relevant to dermatologic care,” she added.
Dr. Zaenglein’s take home message: “Dermatologists should educate all patients of childbearing potential taking isotretinoin on how to acquire and use emergency contraception at every visit.” As for additional research, she said that since the study was conducted with pediatric dermatologists, “it would be very interesting to see if general dermatologists had the same lack of comfort in educating patients on emergency contraception and what their standard counseling practices are.”
The study received no outside funding. Dr. Zaenglein is a member of the AAD’s iPLEDGE Work Group and serves as an editor-in-chief of Pediatric Dermatology.
FROM PEDIATRIC DERMATOLOGY
New study supports safety of COVID-19 boosters during pregnancy
Doctors and health professionals continue to recommend COVID-19 vaccine boosters or third doses for adolescents and adults more than 5 months after their initial vaccinations with the Pfizer-BioNTech BNT162b2 or Moderna mRNA-1273 primary vaccine series or more than 2 months after receiving the Janssen JNJ-78436735 vaccine, Alisa Kachikis, MD, of the University of Washington, Seattle, and colleagues wrote in JAMA Network Open.
Although multiple studies have shown that the COVID-19 primary series is safe and well tolerated in pregnant and lactating women, information on the safety and tolerability of boosters are lacking, the researchers noted.
“COVID-19 will be with us for a while, and it is important to continue to provide data on COVID-19 vaccines in these groups, particularly because there still are many questions about the vaccine, and because pregnant individuals have been, understandably, more hesitant to receive COVID-19 vaccines,” Dr. Kachikis said in an interview. “The findings of this study that COVID-19 booster doses are well tolerated among pregnant and lactating individuals are especially pertinent with the new COVID-19 boosters available this fall.”
In the new study, the researchers reviewed data from 17,014 participants who were part of an ongoing online prospective study of COVID-19 vaccines in pregnant and lactating individuals. Data were collected between October 2021 and April 2022 through an online survey.
The study population included 2,009 participants (11.8%) who were pregnant at the time of their booster or third dose, 10,279 (60.4%) who were lactating, and 4,726 (27.8%) who were neither pregnant nor lactating. The mean age of the participants was 33.3 years; 92.1% self-identified as White, 94.5% self-identified as non-Hispanic, and 99.7% self-identified as female.
The receipt of a booster was similar across trimesters; 26.4%, 36.5%, and 37.1% of participants received boosters or third doses in the first, second, and third trimester, respectively. The primary outcome was self-reported vaccine reactions within 24 hours of the dose.
Overall, 82.8% of the respondents reported a reaction at the site of the injection, such as redness, pain, or swelling, and 67.9% reported at least one systemic symptom, such as aches and pains, headache, chills, or fever. The most frequently reported symptoms across all groups were injection-site pain (82.2%) and fatigue (54.4%).
The pregnant women were significantly more likely than nonpregnant or nonlactating individuals to report any local reaction at the injection site (adjusted odds ratio, 1.2; P = .01), but less likely to report any systemic reaction (aOR, 0.7; P < .001).
The majority (97.6%) of the pregnant respondents and 96.0% of those lactating reported no obstetric or lactation concerns after vaccination.
Overall, a majority of the respondents reported that recommendations from public health authorities were helpful in their decision to receive a COVID-19 booster or third dose (90.0% of pregnant respondents, 89.9% of lactating respondents, and 88.1% of those neither pregnant nor lactating).
Although vaccine uptake in the current study population was high (91.1% overall and 95.0% of those pregnant), “the importance of the health care professional’s recommendation is pertinent given the ongoing increased vaccine hesitancy among pregnant individuals in the context of the COVID-19 vaccine,” the researchers emphasized.
The study findings were limited by several factors including the reliance on self-reports and a convenience sample composed mainly of health care workers because of their vaccine eligibility at the time the study started, which limits generalizability, the researchers noted. Analyses on the pregnancy outcomes of those who were pregnant when vaccinated are in progress.
The results were strengthened by the large study population that included participants from all 50 states and several territories, and ability to compare results between pregnant and lactating individuals with those who were neither pregnant nor lactating, but were of childbearing age, they said.
The results support the safety of COVID-19 boosters for pregnant and breastfeeding individuals, and these data are important to inform discussions between patients and clinicians to boost vaccine uptake and acceptance in this population, they concluded.
“Our earlier data analysis showed that pregnant and lactating individuals did very well with the initial COVID-19 vaccine series, so it was not very surprising that they also did well with COVID-19 booster or third doses,” Dr. Kachikis said in an interview.
There are two takeaway messages for clinicians, she said: “First, pregnant and lactating individuals tolerated the COVID-19 booster well. The second is that clinicians are very important when it comes to vaccine acceptance.”
“In our study, we found that, while pregnant participants were more likely to report that they were hesitant to receive the booster, they also were more likely to have discussed the COVID-19 booster with their health care provider, and to have received a recommendation to receive the booster. So, spending a little bit of extra time with patients discussing COVID-19 boosters and recommending them can make a significant difference,” she said.
The message of the study is highly reassuring for pregnant and lactating individuals, Dr. Kachikis added. “Most of the participants reported that they had fewer symptoms with the COVID-19 booster compared to the primary vaccine series, which is good news, especially since a new COVID-19 booster is being recommended for the fall.”
Reassuring findings for doctors and patients
The current study is especially timely, as updated COVID-19 boosters have now been recommended for most individuals by the Centers for Disease Control and Prevention, Martina L. Badell, MD, a maternal-fetal medicine specialist at Emory University, Atlanta, said in an interview.
The findings support previous studies on the tolerability of COVID-19 vaccinations in pregnant and lactating persons, said Dr. Badell, who was not involved in the study.
The reassuring message for clinicians is that COVID-19 booster vaccinations are similarly well tolerated in pregnancy and lactation as they are in nonpregnant individuals, said Dr. Badell. “Given the risks of COVID infections in pregnancy and neonates, reassuring data on the tolerability and safety of vaccination in this population is very important.” Also, the researchers found that all three cohorts reported that recommendations from public or medical health authorities helped them make a decision about vaccination; “thus the more data to support these recommendations, the better,” she emphasized.
If you are pregnant or breastfeeding, the message from the study is that COVID-19 booster vaccinations are similarly well tolerated by those who are pregnant or breastfeeding and those who are not, said Dr. Badell.
“This study provides additional support for the strong recommendation to encourage not only COVID-19 vaccination in pregnancy and lactation, but booster vaccinations specifically,” and pregnant and breastfeeding individuals should not be excluded from the new CDC recommendations for COVID-19 boosters, she said.
Future research suggestions
Next steps for research include evaluating the obstetrical and neonatal outcomes in pregnancy and lactation following COVID- 19 boosters, Dr. Badell added.
Dr. Kachikis suggested studies try to answer the remaining questions about COVID-19 vaccines and the immunity of pregnant and lactating persons, particularly since they were excluded from the early clinical trials in 2020.
The study was supported by the National Institute of Allergy and Infectious Diseases, a Women’s Reproductive Health Research Award, and the National Center for Advancing Translational Sciences of the National Institutes of Health. \Dr. Kachikis disclosed serving as a research consultant for Pfizer and GlaxoSmithKline and as an unpaid consultant for GlaxoSmithKline unrelated to the current study, as well as grant support from Merck and Pfizer unrelated to the current study. Dr. Badell had no financial conflicts to disclose.
Doctors and health professionals continue to recommend COVID-19 vaccine boosters or third doses for adolescents and adults more than 5 months after their initial vaccinations with the Pfizer-BioNTech BNT162b2 or Moderna mRNA-1273 primary vaccine series or more than 2 months after receiving the Janssen JNJ-78436735 vaccine, Alisa Kachikis, MD, of the University of Washington, Seattle, and colleagues wrote in JAMA Network Open.
Although multiple studies have shown that the COVID-19 primary series is safe and well tolerated in pregnant and lactating women, information on the safety and tolerability of boosters are lacking, the researchers noted.
“COVID-19 will be with us for a while, and it is important to continue to provide data on COVID-19 vaccines in these groups, particularly because there still are many questions about the vaccine, and because pregnant individuals have been, understandably, more hesitant to receive COVID-19 vaccines,” Dr. Kachikis said in an interview. “The findings of this study that COVID-19 booster doses are well tolerated among pregnant and lactating individuals are especially pertinent with the new COVID-19 boosters available this fall.”
In the new study, the researchers reviewed data from 17,014 participants who were part of an ongoing online prospective study of COVID-19 vaccines in pregnant and lactating individuals. Data were collected between October 2021 and April 2022 through an online survey.
The study population included 2,009 participants (11.8%) who were pregnant at the time of their booster or third dose, 10,279 (60.4%) who were lactating, and 4,726 (27.8%) who were neither pregnant nor lactating. The mean age of the participants was 33.3 years; 92.1% self-identified as White, 94.5% self-identified as non-Hispanic, and 99.7% self-identified as female.
The receipt of a booster was similar across trimesters; 26.4%, 36.5%, and 37.1% of participants received boosters or third doses in the first, second, and third trimester, respectively. The primary outcome was self-reported vaccine reactions within 24 hours of the dose.
Overall, 82.8% of the respondents reported a reaction at the site of the injection, such as redness, pain, or swelling, and 67.9% reported at least one systemic symptom, such as aches and pains, headache, chills, or fever. The most frequently reported symptoms across all groups were injection-site pain (82.2%) and fatigue (54.4%).
The pregnant women were significantly more likely than nonpregnant or nonlactating individuals to report any local reaction at the injection site (adjusted odds ratio, 1.2; P = .01), but less likely to report any systemic reaction (aOR, 0.7; P < .001).
The majority (97.6%) of the pregnant respondents and 96.0% of those lactating reported no obstetric or lactation concerns after vaccination.
Overall, a majority of the respondents reported that recommendations from public health authorities were helpful in their decision to receive a COVID-19 booster or third dose (90.0% of pregnant respondents, 89.9% of lactating respondents, and 88.1% of those neither pregnant nor lactating).
Although vaccine uptake in the current study population was high (91.1% overall and 95.0% of those pregnant), “the importance of the health care professional’s recommendation is pertinent given the ongoing increased vaccine hesitancy among pregnant individuals in the context of the COVID-19 vaccine,” the researchers emphasized.
The study findings were limited by several factors including the reliance on self-reports and a convenience sample composed mainly of health care workers because of their vaccine eligibility at the time the study started, which limits generalizability, the researchers noted. Analyses on the pregnancy outcomes of those who were pregnant when vaccinated are in progress.
The results were strengthened by the large study population that included participants from all 50 states and several territories, and ability to compare results between pregnant and lactating individuals with those who were neither pregnant nor lactating, but were of childbearing age, they said.
The results support the safety of COVID-19 boosters for pregnant and breastfeeding individuals, and these data are important to inform discussions between patients and clinicians to boost vaccine uptake and acceptance in this population, they concluded.
“Our earlier data analysis showed that pregnant and lactating individuals did very well with the initial COVID-19 vaccine series, so it was not very surprising that they also did well with COVID-19 booster or third doses,” Dr. Kachikis said in an interview.
There are two takeaway messages for clinicians, she said: “First, pregnant and lactating individuals tolerated the COVID-19 booster well. The second is that clinicians are very important when it comes to vaccine acceptance.”
“In our study, we found that, while pregnant participants were more likely to report that they were hesitant to receive the booster, they also were more likely to have discussed the COVID-19 booster with their health care provider, and to have received a recommendation to receive the booster. So, spending a little bit of extra time with patients discussing COVID-19 boosters and recommending them can make a significant difference,” she said.
The message of the study is highly reassuring for pregnant and lactating individuals, Dr. Kachikis added. “Most of the participants reported that they had fewer symptoms with the COVID-19 booster compared to the primary vaccine series, which is good news, especially since a new COVID-19 booster is being recommended for the fall.”
Reassuring findings for doctors and patients
The current study is especially timely, as updated COVID-19 boosters have now been recommended for most individuals by the Centers for Disease Control and Prevention, Martina L. Badell, MD, a maternal-fetal medicine specialist at Emory University, Atlanta, said in an interview.
The findings support previous studies on the tolerability of COVID-19 vaccinations in pregnant and lactating persons, said Dr. Badell, who was not involved in the study.
The reassuring message for clinicians is that COVID-19 booster vaccinations are similarly well tolerated in pregnancy and lactation as they are in nonpregnant individuals, said Dr. Badell. “Given the risks of COVID infections in pregnancy and neonates, reassuring data on the tolerability and safety of vaccination in this population is very important.” Also, the researchers found that all three cohorts reported that recommendations from public or medical health authorities helped them make a decision about vaccination; “thus the more data to support these recommendations, the better,” she emphasized.
If you are pregnant or breastfeeding, the message from the study is that COVID-19 booster vaccinations are similarly well tolerated by those who are pregnant or breastfeeding and those who are not, said Dr. Badell.
“This study provides additional support for the strong recommendation to encourage not only COVID-19 vaccination in pregnancy and lactation, but booster vaccinations specifically,” and pregnant and breastfeeding individuals should not be excluded from the new CDC recommendations for COVID-19 boosters, she said.
Future research suggestions
Next steps for research include evaluating the obstetrical and neonatal outcomes in pregnancy and lactation following COVID- 19 boosters, Dr. Badell added.
Dr. Kachikis suggested studies try to answer the remaining questions about COVID-19 vaccines and the immunity of pregnant and lactating persons, particularly since they were excluded from the early clinical trials in 2020.
The study was supported by the National Institute of Allergy and Infectious Diseases, a Women’s Reproductive Health Research Award, and the National Center for Advancing Translational Sciences of the National Institutes of Health. \Dr. Kachikis disclosed serving as a research consultant for Pfizer and GlaxoSmithKline and as an unpaid consultant for GlaxoSmithKline unrelated to the current study, as well as grant support from Merck and Pfizer unrelated to the current study. Dr. Badell had no financial conflicts to disclose.
Doctors and health professionals continue to recommend COVID-19 vaccine boosters or third doses for adolescents and adults more than 5 months after their initial vaccinations with the Pfizer-BioNTech BNT162b2 or Moderna mRNA-1273 primary vaccine series or more than 2 months after receiving the Janssen JNJ-78436735 vaccine, Alisa Kachikis, MD, of the University of Washington, Seattle, and colleagues wrote in JAMA Network Open.
Although multiple studies have shown that the COVID-19 primary series is safe and well tolerated in pregnant and lactating women, information on the safety and tolerability of boosters are lacking, the researchers noted.
“COVID-19 will be with us for a while, and it is important to continue to provide data on COVID-19 vaccines in these groups, particularly because there still are many questions about the vaccine, and because pregnant individuals have been, understandably, more hesitant to receive COVID-19 vaccines,” Dr. Kachikis said in an interview. “The findings of this study that COVID-19 booster doses are well tolerated among pregnant and lactating individuals are especially pertinent with the new COVID-19 boosters available this fall.”
In the new study, the researchers reviewed data from 17,014 participants who were part of an ongoing online prospective study of COVID-19 vaccines in pregnant and lactating individuals. Data were collected between October 2021 and April 2022 through an online survey.
The study population included 2,009 participants (11.8%) who were pregnant at the time of their booster or third dose, 10,279 (60.4%) who were lactating, and 4,726 (27.8%) who were neither pregnant nor lactating. The mean age of the participants was 33.3 years; 92.1% self-identified as White, 94.5% self-identified as non-Hispanic, and 99.7% self-identified as female.
The receipt of a booster was similar across trimesters; 26.4%, 36.5%, and 37.1% of participants received boosters or third doses in the first, second, and third trimester, respectively. The primary outcome was self-reported vaccine reactions within 24 hours of the dose.
Overall, 82.8% of the respondents reported a reaction at the site of the injection, such as redness, pain, or swelling, and 67.9% reported at least one systemic symptom, such as aches and pains, headache, chills, or fever. The most frequently reported symptoms across all groups were injection-site pain (82.2%) and fatigue (54.4%).
The pregnant women were significantly more likely than nonpregnant or nonlactating individuals to report any local reaction at the injection site (adjusted odds ratio, 1.2; P = .01), but less likely to report any systemic reaction (aOR, 0.7; P < .001).
The majority (97.6%) of the pregnant respondents and 96.0% of those lactating reported no obstetric or lactation concerns after vaccination.
Overall, a majority of the respondents reported that recommendations from public health authorities were helpful in their decision to receive a COVID-19 booster or third dose (90.0% of pregnant respondents, 89.9% of lactating respondents, and 88.1% of those neither pregnant nor lactating).
Although vaccine uptake in the current study population was high (91.1% overall and 95.0% of those pregnant), “the importance of the health care professional’s recommendation is pertinent given the ongoing increased vaccine hesitancy among pregnant individuals in the context of the COVID-19 vaccine,” the researchers emphasized.
The study findings were limited by several factors including the reliance on self-reports and a convenience sample composed mainly of health care workers because of their vaccine eligibility at the time the study started, which limits generalizability, the researchers noted. Analyses on the pregnancy outcomes of those who were pregnant when vaccinated are in progress.
The results were strengthened by the large study population that included participants from all 50 states and several territories, and ability to compare results between pregnant and lactating individuals with those who were neither pregnant nor lactating, but were of childbearing age, they said.
The results support the safety of COVID-19 boosters for pregnant and breastfeeding individuals, and these data are important to inform discussions between patients and clinicians to boost vaccine uptake and acceptance in this population, they concluded.
“Our earlier data analysis showed that pregnant and lactating individuals did very well with the initial COVID-19 vaccine series, so it was not very surprising that they also did well with COVID-19 booster or third doses,” Dr. Kachikis said in an interview.
There are two takeaway messages for clinicians, she said: “First, pregnant and lactating individuals tolerated the COVID-19 booster well. The second is that clinicians are very important when it comes to vaccine acceptance.”
“In our study, we found that, while pregnant participants were more likely to report that they were hesitant to receive the booster, they also were more likely to have discussed the COVID-19 booster with their health care provider, and to have received a recommendation to receive the booster. So, spending a little bit of extra time with patients discussing COVID-19 boosters and recommending them can make a significant difference,” she said.
The message of the study is highly reassuring for pregnant and lactating individuals, Dr. Kachikis added. “Most of the participants reported that they had fewer symptoms with the COVID-19 booster compared to the primary vaccine series, which is good news, especially since a new COVID-19 booster is being recommended for the fall.”
Reassuring findings for doctors and patients
The current study is especially timely, as updated COVID-19 boosters have now been recommended for most individuals by the Centers for Disease Control and Prevention, Martina L. Badell, MD, a maternal-fetal medicine specialist at Emory University, Atlanta, said in an interview.
The findings support previous studies on the tolerability of COVID-19 vaccinations in pregnant and lactating persons, said Dr. Badell, who was not involved in the study.
The reassuring message for clinicians is that COVID-19 booster vaccinations are similarly well tolerated in pregnancy and lactation as they are in nonpregnant individuals, said Dr. Badell. “Given the risks of COVID infections in pregnancy and neonates, reassuring data on the tolerability and safety of vaccination in this population is very important.” Also, the researchers found that all three cohorts reported that recommendations from public or medical health authorities helped them make a decision about vaccination; “thus the more data to support these recommendations, the better,” she emphasized.
If you are pregnant or breastfeeding, the message from the study is that COVID-19 booster vaccinations are similarly well tolerated by those who are pregnant or breastfeeding and those who are not, said Dr. Badell.
“This study provides additional support for the strong recommendation to encourage not only COVID-19 vaccination in pregnancy and lactation, but booster vaccinations specifically,” and pregnant and breastfeeding individuals should not be excluded from the new CDC recommendations for COVID-19 boosters, she said.
Future research suggestions
Next steps for research include evaluating the obstetrical and neonatal outcomes in pregnancy and lactation following COVID- 19 boosters, Dr. Badell added.
Dr. Kachikis suggested studies try to answer the remaining questions about COVID-19 vaccines and the immunity of pregnant and lactating persons, particularly since they were excluded from the early clinical trials in 2020.
The study was supported by the National Institute of Allergy and Infectious Diseases, a Women’s Reproductive Health Research Award, and the National Center for Advancing Translational Sciences of the National Institutes of Health. \Dr. Kachikis disclosed serving as a research consultant for Pfizer and GlaxoSmithKline and as an unpaid consultant for GlaxoSmithKline unrelated to the current study, as well as grant support from Merck and Pfizer unrelated to the current study. Dr. Badell had no financial conflicts to disclose.
FROM JAMA NETWORK OPEN
WIC review finds broad benefits, knowledge gaps
How exactly the national program achieves these outcomes, however, remains unclear, and study quality shows room for improvement, reported co–lead authors Maya Venkataramani, MD, MPH and S. Michelle Ogunwole, MD, PhD of Johns Hopkins University, Baltimore, and colleagues.
The WIC program, which has been serving low-income women and young children since 1974, “provides supplemental foods, nutrition education and breastfeeding support, screening and referrals to medical and social services, and support for high-risk pregnancies,” the investigators wrote in Annals of Internal Medicine. The U.S. Food and Nutrition Service administers the program.
The authors conducted a systematic review of 20 observational studies aimed at determining the impacts of WIC participation on maternal, neonatal-birth, and infant-child health outcomes.
All studies included in the review began in or after 2009, when the WIC food package was revised to better address diet-related chronic diseases. For inclusion in the review, studies were required to have a WIC-eligible comparison group. Included research also evaluated the relationship between WIC participation and the prespecified health outcomes.
“We found only 20 studies that fulfilled our rigorous study inclusion criteria for these specific outcomes,” the investigators wrote. “In some areas, the evidence was absent, and in others, the strength of evidence (SOE) was moderate or low.”
Six outcome categories were assessed: maternal morbidity, maternal pregnancy outcomes, maternal health behaviors, maternal health care utilization, child morbidity, and childhood health care utilization. Of these, maternal health care utilization had the most robust body of evidence, while data from studies evaluating maternal morbidity and child morbidity were deemed insufficient.
Based on eligible studies, WIC participation was associated with reduced risks of insufficient weight gain in pregnancy, preterm birth, low infant birthweight, and infant mortality. Participation was also associated with an increased likelihood of infant and child health care utilization, such as routine immunizations.
Growing evidence should drive enrollment
“Growing evidence points to WIC as a way to reduce risk of preterm birth and other adverse outcomes,” said Laura Jelliffe-Pawlowski, PhD, MS, professor at the University of California, San Francisco and a director for the UCSF California Preterm Birth Initiative.
Dr. Jelliffe-Pawlowski, who conducted a California-based study included in the paper, said the review is noteworthy because it shows that WIC-associated benefits are observed across locations.
“It’s not just in California; it’s across the country,” she said. “It’s a national call to action – where there’s partnership between national-, state- and community-level WIC programs – to make WIC as accessible as possible, and reflect community wants and needs, so that more people enroll, and more people stay enrolled.”
Dr. Jelliffe-Pawlowski’s coauthor on the California study, Rita Hamad, MD, PhD, associate professor of family & community medicine at UCSF and associate director of the UCSF Center for Health Equity, encouraged health care providers to drive WIC enrollment, noting that, presently, only one in four eligible 4-year-olds participates.
“Physicians and other health care stakeholders can help patients benefit from this program by encouraging them to sign up, and even by providing sign-up support in the form of a social worker or other staff member,” Dr. Hamad said. “There is also literature on the types of interventions that improve take-up of safety net programs that providers can look to.”
Goals of future research
Optimizing WIC operations, however, is only half the battle, considering the evidence gaps revealed by the review.
“We still need stronger studies that use more rigorous study designs ... to provide more convincing evidence to policymakers, as well as more evidence on long-term impacts,” Dr. Hamad said. “We also need to better understand why take-up is low in these programs despite these potential health benefits. Then we can make sure that economically disadvantaged families receive the benefits for which they are eligible through interventions to improve participation rates.”
Ideally, WIC programs would receive additional funding for independent parties to evaluate health outcomes, according to Ashwini Lakshmanan, MD, MS, MPH, associate professor in the department of health systems science at Kaiser Permanente Bernard J. Tyson School of Medicine, Pasadena, Calif.
Dr. Lakshmanan, who previously evaluated the benefits of WIC participation for high-risk infants, noted that randomized clinical trials would be unethical in this setting, yet data collection can still be “very conscientious and intentional,” with a focus on policy-shaping outcome metrics like immunizations and pediatric health care visits.
“The main point is thinking about it at the forefront, and not retrospectively,” Dr. Lakshmanan said.
Dr. Ogunwole, who led the present review, suggested in a written comment that future studies “could employ robust statistical methods (propensity matching, fixed effects models, etc.) to help reduce bias.”
She also recommended evaluating innovations in WIC programs; for example, adding a health coach, or conducting a cooking skills intervention.
Studies are also needed to better understand the various obstacles to WIC success, such as misconceptions about the program, discrimination, and barriers to enrollment, Dr. Ogunwole added.
“WIC enrollment has been decreasing for a number of years, and this was occurring prepandemic as well,” she said. “More work needs to be done to understand this issue.”
The study was supported by the Agency for Healthcare Research and Quality, U.S. Department of Health & Human Services. The investigators and interviewees disclosed no conflicts of interest.
How exactly the national program achieves these outcomes, however, remains unclear, and study quality shows room for improvement, reported co–lead authors Maya Venkataramani, MD, MPH and S. Michelle Ogunwole, MD, PhD of Johns Hopkins University, Baltimore, and colleagues.
The WIC program, which has been serving low-income women and young children since 1974, “provides supplemental foods, nutrition education and breastfeeding support, screening and referrals to medical and social services, and support for high-risk pregnancies,” the investigators wrote in Annals of Internal Medicine. The U.S. Food and Nutrition Service administers the program.
The authors conducted a systematic review of 20 observational studies aimed at determining the impacts of WIC participation on maternal, neonatal-birth, and infant-child health outcomes.
All studies included in the review began in or after 2009, when the WIC food package was revised to better address diet-related chronic diseases. For inclusion in the review, studies were required to have a WIC-eligible comparison group. Included research also evaluated the relationship between WIC participation and the prespecified health outcomes.
“We found only 20 studies that fulfilled our rigorous study inclusion criteria for these specific outcomes,” the investigators wrote. “In some areas, the evidence was absent, and in others, the strength of evidence (SOE) was moderate or low.”
Six outcome categories were assessed: maternal morbidity, maternal pregnancy outcomes, maternal health behaviors, maternal health care utilization, child morbidity, and childhood health care utilization. Of these, maternal health care utilization had the most robust body of evidence, while data from studies evaluating maternal morbidity and child morbidity were deemed insufficient.
Based on eligible studies, WIC participation was associated with reduced risks of insufficient weight gain in pregnancy, preterm birth, low infant birthweight, and infant mortality. Participation was also associated with an increased likelihood of infant and child health care utilization, such as routine immunizations.
Growing evidence should drive enrollment
“Growing evidence points to WIC as a way to reduce risk of preterm birth and other adverse outcomes,” said Laura Jelliffe-Pawlowski, PhD, MS, professor at the University of California, San Francisco and a director for the UCSF California Preterm Birth Initiative.
Dr. Jelliffe-Pawlowski, who conducted a California-based study included in the paper, said the review is noteworthy because it shows that WIC-associated benefits are observed across locations.
“It’s not just in California; it’s across the country,” she said. “It’s a national call to action – where there’s partnership between national-, state- and community-level WIC programs – to make WIC as accessible as possible, and reflect community wants and needs, so that more people enroll, and more people stay enrolled.”
Dr. Jelliffe-Pawlowski’s coauthor on the California study, Rita Hamad, MD, PhD, associate professor of family & community medicine at UCSF and associate director of the UCSF Center for Health Equity, encouraged health care providers to drive WIC enrollment, noting that, presently, only one in four eligible 4-year-olds participates.
“Physicians and other health care stakeholders can help patients benefit from this program by encouraging them to sign up, and even by providing sign-up support in the form of a social worker or other staff member,” Dr. Hamad said. “There is also literature on the types of interventions that improve take-up of safety net programs that providers can look to.”
Goals of future research
Optimizing WIC operations, however, is only half the battle, considering the evidence gaps revealed by the review.
“We still need stronger studies that use more rigorous study designs ... to provide more convincing evidence to policymakers, as well as more evidence on long-term impacts,” Dr. Hamad said. “We also need to better understand why take-up is low in these programs despite these potential health benefits. Then we can make sure that economically disadvantaged families receive the benefits for which they are eligible through interventions to improve participation rates.”
Ideally, WIC programs would receive additional funding for independent parties to evaluate health outcomes, according to Ashwini Lakshmanan, MD, MS, MPH, associate professor in the department of health systems science at Kaiser Permanente Bernard J. Tyson School of Medicine, Pasadena, Calif.
Dr. Lakshmanan, who previously evaluated the benefits of WIC participation for high-risk infants, noted that randomized clinical trials would be unethical in this setting, yet data collection can still be “very conscientious and intentional,” with a focus on policy-shaping outcome metrics like immunizations and pediatric health care visits.
“The main point is thinking about it at the forefront, and not retrospectively,” Dr. Lakshmanan said.
Dr. Ogunwole, who led the present review, suggested in a written comment that future studies “could employ robust statistical methods (propensity matching, fixed effects models, etc.) to help reduce bias.”
She also recommended evaluating innovations in WIC programs; for example, adding a health coach, or conducting a cooking skills intervention.
Studies are also needed to better understand the various obstacles to WIC success, such as misconceptions about the program, discrimination, and barriers to enrollment, Dr. Ogunwole added.
“WIC enrollment has been decreasing for a number of years, and this was occurring prepandemic as well,” she said. “More work needs to be done to understand this issue.”
The study was supported by the Agency for Healthcare Research and Quality, U.S. Department of Health & Human Services. The investigators and interviewees disclosed no conflicts of interest.
How exactly the national program achieves these outcomes, however, remains unclear, and study quality shows room for improvement, reported co–lead authors Maya Venkataramani, MD, MPH and S. Michelle Ogunwole, MD, PhD of Johns Hopkins University, Baltimore, and colleagues.
The WIC program, which has been serving low-income women and young children since 1974, “provides supplemental foods, nutrition education and breastfeeding support, screening and referrals to medical and social services, and support for high-risk pregnancies,” the investigators wrote in Annals of Internal Medicine. The U.S. Food and Nutrition Service administers the program.
The authors conducted a systematic review of 20 observational studies aimed at determining the impacts of WIC participation on maternal, neonatal-birth, and infant-child health outcomes.
All studies included in the review began in or after 2009, when the WIC food package was revised to better address diet-related chronic diseases. For inclusion in the review, studies were required to have a WIC-eligible comparison group. Included research also evaluated the relationship between WIC participation and the prespecified health outcomes.
“We found only 20 studies that fulfilled our rigorous study inclusion criteria for these specific outcomes,” the investigators wrote. “In some areas, the evidence was absent, and in others, the strength of evidence (SOE) was moderate or low.”
Six outcome categories were assessed: maternal morbidity, maternal pregnancy outcomes, maternal health behaviors, maternal health care utilization, child morbidity, and childhood health care utilization. Of these, maternal health care utilization had the most robust body of evidence, while data from studies evaluating maternal morbidity and child morbidity were deemed insufficient.
Based on eligible studies, WIC participation was associated with reduced risks of insufficient weight gain in pregnancy, preterm birth, low infant birthweight, and infant mortality. Participation was also associated with an increased likelihood of infant and child health care utilization, such as routine immunizations.
Growing evidence should drive enrollment
“Growing evidence points to WIC as a way to reduce risk of preterm birth and other adverse outcomes,” said Laura Jelliffe-Pawlowski, PhD, MS, professor at the University of California, San Francisco and a director for the UCSF California Preterm Birth Initiative.
Dr. Jelliffe-Pawlowski, who conducted a California-based study included in the paper, said the review is noteworthy because it shows that WIC-associated benefits are observed across locations.
“It’s not just in California; it’s across the country,” she said. “It’s a national call to action – where there’s partnership between national-, state- and community-level WIC programs – to make WIC as accessible as possible, and reflect community wants and needs, so that more people enroll, and more people stay enrolled.”
Dr. Jelliffe-Pawlowski’s coauthor on the California study, Rita Hamad, MD, PhD, associate professor of family & community medicine at UCSF and associate director of the UCSF Center for Health Equity, encouraged health care providers to drive WIC enrollment, noting that, presently, only one in four eligible 4-year-olds participates.
“Physicians and other health care stakeholders can help patients benefit from this program by encouraging them to sign up, and even by providing sign-up support in the form of a social worker or other staff member,” Dr. Hamad said. “There is also literature on the types of interventions that improve take-up of safety net programs that providers can look to.”
Goals of future research
Optimizing WIC operations, however, is only half the battle, considering the evidence gaps revealed by the review.
“We still need stronger studies that use more rigorous study designs ... to provide more convincing evidence to policymakers, as well as more evidence on long-term impacts,” Dr. Hamad said. “We also need to better understand why take-up is low in these programs despite these potential health benefits. Then we can make sure that economically disadvantaged families receive the benefits for which they are eligible through interventions to improve participation rates.”
Ideally, WIC programs would receive additional funding for independent parties to evaluate health outcomes, according to Ashwini Lakshmanan, MD, MS, MPH, associate professor in the department of health systems science at Kaiser Permanente Bernard J. Tyson School of Medicine, Pasadena, Calif.
Dr. Lakshmanan, who previously evaluated the benefits of WIC participation for high-risk infants, noted that randomized clinical trials would be unethical in this setting, yet data collection can still be “very conscientious and intentional,” with a focus on policy-shaping outcome metrics like immunizations and pediatric health care visits.
“The main point is thinking about it at the forefront, and not retrospectively,” Dr. Lakshmanan said.
Dr. Ogunwole, who led the present review, suggested in a written comment that future studies “could employ robust statistical methods (propensity matching, fixed effects models, etc.) to help reduce bias.”
She also recommended evaluating innovations in WIC programs; for example, adding a health coach, or conducting a cooking skills intervention.
Studies are also needed to better understand the various obstacles to WIC success, such as misconceptions about the program, discrimination, and barriers to enrollment, Dr. Ogunwole added.
“WIC enrollment has been decreasing for a number of years, and this was occurring prepandemic as well,” she said. “More work needs to be done to understand this issue.”
The study was supported by the Agency for Healthcare Research and Quality, U.S. Department of Health & Human Services. The investigators and interviewees disclosed no conflicts of interest.
FROM ANNALS OF INTERNAL MEDICINE
In the rush to curtail abortion, states adopt a jumbled stew of definitions for human life
As life-preserving medical technology advanced in the second half of the 20th century, doctors and families were faced with a thorny decision, one with weighty legal and moral implications: How should we define when life ends? Cardiopulmonary bypass machines could keep the blood pumping and ventilators could maintain breathing long after a patient’s natural ability to perform those vital functions had ceased.
After decades of deliberations involving physicians, bioethicists, attorneys, and theologians, a U.S. presidential commission in 1981 settled on a scientifically derived dividing line between life and death that has endured, more or less, ever since: A person was considered dead when the entire brain – including the brain stem, its most primitive portion – was no longer functioning, even if other vital functions could be maintained indefinitely through artificial life support.
In the decades since, the committee’s criteria have served as a foundation for laws in most states adopting brain death as a standard for legal death.
Now, with the overturning of Roe v. Wade and dozens of states rushing to impose abortion restrictions, At conception, the hint of a heartbeat, a first breath, the ability to survive outside the womb with the help of the latest technology?
That we’ve been able to devise and apply uniform clinical standards for when life ends, but not when it begins, is due largely to the legal and political maelstrom around abortion. And in the 2 months since the U.S. Supreme Court issued its opinion in Dobbs v. Jackson Women’s Health Organization, eliminating a longstanding federal right to abortion, state legislators are eagerly bounding into that void, looking to codify into law assorted definitions of life that carry profound repercussions for abortion rights, birth control, and assisted reproduction, as well as civil and criminal law.
“The court said that when life begins is up to whoever is running your state – whether they are wrong or not, or you agree with them or not,” said Mary Ziegler, a law professor at the University of California, Davis, who has written several books on the history of abortion.
Unlike the debate over death, which delved into exquisite medical and scientific detail, the legislative scramble to determine when life’s building blocks reach a threshold that warrants government protection as human life has generally ignored the input of mainstream medical professionals.
Instead, red states across much of the South and portions of the Midwest are adopting language drafted by elected officials that is informed by conservative Christian doctrine, often with little scientific underpinning.
A handful of Republican-led states, including Arkansas, Kentucky, Missouri, and Oklahoma, have passed laws declaring that life begins at fertilization, a contention that opens the door to a host of pregnancy-related litigation. This includes wrongful death lawsuits brought on behalf of the estate of an embryo by disgruntled ex-partners against physicians and women who end a pregnancy or even miscarry. (One such lawsuit is underway in Arizona. Another reached the Alabama Supreme Court.)
In Kentucky, the law outlawing abortion uses morally explosive terms to define pregnancy as “the human female reproductive condition of having a living unborn human being within her body throughout the entire embryonic and fetal stages of the unborn child from fertilization to full gestation and childbirth.”
Several other states, including Georgia, have adopted measures equating life with the point at which an embryo’s nascent cardiac activity can be detected by an ultrasound, at around 6 weeks of gestation. Many such laws mischaracterize the flickering electrical impulses detectable at that stage as a heartbeat, including in Georgia, whose Department of Revenue recently announced that “any unborn child with a detectable human heartbeat” can be claimed as a dependent.
The Supreme Court’s 1973 decision in Roe v. Wade that established a constitutional right to abortion did not define a moment when life begins. The opinion, written by Justice Harry Blackmun, observed that the Constitution does not provide a definition of “person,” though it extends protections to those born or naturalized in the United States. The court majority made note of the many disparate views among religions and scientists on when life begins, and concluded it was not up to the states to adopt one theory of life.
Instead, Roe created a framework intended to balance a pregnant woman’s right to make decisions about her body with a public interest in protecting potential human life. That decision and a key ruling that followed generally recognized a woman’s right to abortion up to the point medical professionals judge a fetus viable to survive outside the uterus, at about 24 weeks of gestation.
In decisively overturning Roe in June, the Supreme Court’s conservative majority drew on legal arguments that have shaped another contentious end-of-life issue. The legal standard employed in Dobbs – that there is no right to abortion in the federal Constitution and that states can decide on their own – is the same rationale used in 1997 when the Supreme Court said terminally ill people did not have a constitutional right to medically assisted death. That decision, Washington v. Glucksberg, is mentioned 15 times in the majority opinion for Dobbs and a concurrence by Justice Clarence Thomas.
Often, the same groups that have led the fight to outlaw abortion have also challenged medical aid-in-dying laws. Even after Dobbs, so-called right-to-die laws remain far less common than those codifying state abortion rights. Ten states allow physicians to prescribe lethal doses of medicine for terminally ill patients. Doctors are still prohibited from administering the drugs.
James Bopp, general counsel for the National Right to Life Committee who has been central to the efforts to outlaw abortion, said that both abortion and medically assisted death, which he refers to as physician-assisted suicide, endanger society.
“Every individual human life has inherent value and is sacred,” said Mr. Bopp. “The government has the duty to protect that life.”
Both issues raise profound societal questions: Can the government keep a patient on life support against his wishes, or force a woman to give birth? Can states bar their own residents from going to other states to end a pregnancy, or prohibit out-of-state patients from coming in to seek medically assisted death? And who gets to decide, particularly if the answer imposes a singular religious viewpoint?
Just as there are legal implications that flow from determining a person’s death, from organ donation to inheritance, the implied rights held by a legally recognized zygote are potentially vast. Will death certificates be issued for every lost pregnancy? Will miscarriages be investigated? When will Social Security numbers be issued? How will census counts be tallied and congressional districts drawn?
Medical professionals and bioethicists caution that both the beginning and end of life are complicated biological processes that are not defined by a single identifiable moment – and are ill suited to the political arena.
“Unfortunately, biological occurrences are not events, they are processes,” said David Magnus, PhD, director of the Stanford (Calif.) Center for Biomedical Ethics.
Moreover, asking doctors “What is life?” or “What is death?” may miss the point, said Dr. Magnus: “Medicine can answer the question ‘When does a biological organism cease to exist?’ But they can’t answer the question ‘When does a person begin or end?’ because those are metaphysical issues.”
Ben Sarbey, a doctoral candidate in the department of philosophy at Duke University, Durham, N.C., who studies medical ethics, echoed that perspective, recounting the Paradox of the Heap, a thought experiment that involves placing grains of sand one on top of the next. The philosophical quandary is this: At what point do those grains of sand become something more – a heap?
“We’re going to have a rough time placing a dividing line that this counts as a person and this does not count as a person,” he said. “Many things count as life – a sperm counts as life, a person in a persistent vegetative state counts as life – but does that constitute a person that we should be protecting?”
Even as debate over the court’s abortion decision percolates, the 1981 federal statute that grew out of the presidential committee’s findings, the Uniform Determination of Death Act, is also under review. In 2022, the Uniform Law Commission, a nonpartisan group of legal experts that drafts laws intended for adoption in multiple states, has taken up the work to revisit the definition of death.
The group will consider sharpening the medical standards for brain death in light of advances in the understanding of brain function. And they will look to address lingering questions raised in recent years as families and religious groups have waged heated legal battles over terminating artificial life support for patients with no brain wave activity.
Mr. Bopp, with the National Right to Life Committee, is among those serving on advisory panels for the effort, along with an array of doctors, philosophers, and medical ethicists. The concept of “personhood” that infuses the antiabortion movement’s broader push for fetal rights is expected to be an underlying topic, albeit in mirror image: When does a life form cease being a person?
Dr. Magnus, who is also serving on an advisory panel, has no doubt the commission will reach a consensus, a sober resolution rooted in science. What’s less clear, he said, is whether in today’s political environment that updated definition will hold the same sway, an enduring legal standard embraced across states.
KHN (Kaiser Health News) is a national newsroom that produces in-depth journalism about health issues. Together with Policy Analysis and Polling, KHN is one of the three major operating programs at KFF (Kaiser Family Foundation). KFF is an endowed nonprofit organization providing information on health issues to the nation.
As life-preserving medical technology advanced in the second half of the 20th century, doctors and families were faced with a thorny decision, one with weighty legal and moral implications: How should we define when life ends? Cardiopulmonary bypass machines could keep the blood pumping and ventilators could maintain breathing long after a patient’s natural ability to perform those vital functions had ceased.
After decades of deliberations involving physicians, bioethicists, attorneys, and theologians, a U.S. presidential commission in 1981 settled on a scientifically derived dividing line between life and death that has endured, more or less, ever since: A person was considered dead when the entire brain – including the brain stem, its most primitive portion – was no longer functioning, even if other vital functions could be maintained indefinitely through artificial life support.
In the decades since, the committee’s criteria have served as a foundation for laws in most states adopting brain death as a standard for legal death.
Now, with the overturning of Roe v. Wade and dozens of states rushing to impose abortion restrictions, At conception, the hint of a heartbeat, a first breath, the ability to survive outside the womb with the help of the latest technology?
That we’ve been able to devise and apply uniform clinical standards for when life ends, but not when it begins, is due largely to the legal and political maelstrom around abortion. And in the 2 months since the U.S. Supreme Court issued its opinion in Dobbs v. Jackson Women’s Health Organization, eliminating a longstanding federal right to abortion, state legislators are eagerly bounding into that void, looking to codify into law assorted definitions of life that carry profound repercussions for abortion rights, birth control, and assisted reproduction, as well as civil and criminal law.
“The court said that when life begins is up to whoever is running your state – whether they are wrong or not, or you agree with them or not,” said Mary Ziegler, a law professor at the University of California, Davis, who has written several books on the history of abortion.
Unlike the debate over death, which delved into exquisite medical and scientific detail, the legislative scramble to determine when life’s building blocks reach a threshold that warrants government protection as human life has generally ignored the input of mainstream medical professionals.
Instead, red states across much of the South and portions of the Midwest are adopting language drafted by elected officials that is informed by conservative Christian doctrine, often with little scientific underpinning.
A handful of Republican-led states, including Arkansas, Kentucky, Missouri, and Oklahoma, have passed laws declaring that life begins at fertilization, a contention that opens the door to a host of pregnancy-related litigation. This includes wrongful death lawsuits brought on behalf of the estate of an embryo by disgruntled ex-partners against physicians and women who end a pregnancy or even miscarry. (One such lawsuit is underway in Arizona. Another reached the Alabama Supreme Court.)
In Kentucky, the law outlawing abortion uses morally explosive terms to define pregnancy as “the human female reproductive condition of having a living unborn human being within her body throughout the entire embryonic and fetal stages of the unborn child from fertilization to full gestation and childbirth.”
Several other states, including Georgia, have adopted measures equating life with the point at which an embryo’s nascent cardiac activity can be detected by an ultrasound, at around 6 weeks of gestation. Many such laws mischaracterize the flickering electrical impulses detectable at that stage as a heartbeat, including in Georgia, whose Department of Revenue recently announced that “any unborn child with a detectable human heartbeat” can be claimed as a dependent.
The Supreme Court’s 1973 decision in Roe v. Wade that established a constitutional right to abortion did not define a moment when life begins. The opinion, written by Justice Harry Blackmun, observed that the Constitution does not provide a definition of “person,” though it extends protections to those born or naturalized in the United States. The court majority made note of the many disparate views among religions and scientists on when life begins, and concluded it was not up to the states to adopt one theory of life.
Instead, Roe created a framework intended to balance a pregnant woman’s right to make decisions about her body with a public interest in protecting potential human life. That decision and a key ruling that followed generally recognized a woman’s right to abortion up to the point medical professionals judge a fetus viable to survive outside the uterus, at about 24 weeks of gestation.
In decisively overturning Roe in June, the Supreme Court’s conservative majority drew on legal arguments that have shaped another contentious end-of-life issue. The legal standard employed in Dobbs – that there is no right to abortion in the federal Constitution and that states can decide on their own – is the same rationale used in 1997 when the Supreme Court said terminally ill people did not have a constitutional right to medically assisted death. That decision, Washington v. Glucksberg, is mentioned 15 times in the majority opinion for Dobbs and a concurrence by Justice Clarence Thomas.
Often, the same groups that have led the fight to outlaw abortion have also challenged medical aid-in-dying laws. Even after Dobbs, so-called right-to-die laws remain far less common than those codifying state abortion rights. Ten states allow physicians to prescribe lethal doses of medicine for terminally ill patients. Doctors are still prohibited from administering the drugs.
James Bopp, general counsel for the National Right to Life Committee who has been central to the efforts to outlaw abortion, said that both abortion and medically assisted death, which he refers to as physician-assisted suicide, endanger society.
“Every individual human life has inherent value and is sacred,” said Mr. Bopp. “The government has the duty to protect that life.”
Both issues raise profound societal questions: Can the government keep a patient on life support against his wishes, or force a woman to give birth? Can states bar their own residents from going to other states to end a pregnancy, or prohibit out-of-state patients from coming in to seek medically assisted death? And who gets to decide, particularly if the answer imposes a singular religious viewpoint?
Just as there are legal implications that flow from determining a person’s death, from organ donation to inheritance, the implied rights held by a legally recognized zygote are potentially vast. Will death certificates be issued for every lost pregnancy? Will miscarriages be investigated? When will Social Security numbers be issued? How will census counts be tallied and congressional districts drawn?
Medical professionals and bioethicists caution that both the beginning and end of life are complicated biological processes that are not defined by a single identifiable moment – and are ill suited to the political arena.
“Unfortunately, biological occurrences are not events, they are processes,” said David Magnus, PhD, director of the Stanford (Calif.) Center for Biomedical Ethics.
Moreover, asking doctors “What is life?” or “What is death?” may miss the point, said Dr. Magnus: “Medicine can answer the question ‘When does a biological organism cease to exist?’ But they can’t answer the question ‘When does a person begin or end?’ because those are metaphysical issues.”
Ben Sarbey, a doctoral candidate in the department of philosophy at Duke University, Durham, N.C., who studies medical ethics, echoed that perspective, recounting the Paradox of the Heap, a thought experiment that involves placing grains of sand one on top of the next. The philosophical quandary is this: At what point do those grains of sand become something more – a heap?
“We’re going to have a rough time placing a dividing line that this counts as a person and this does not count as a person,” he said. “Many things count as life – a sperm counts as life, a person in a persistent vegetative state counts as life – but does that constitute a person that we should be protecting?”
Even as debate over the court’s abortion decision percolates, the 1981 federal statute that grew out of the presidential committee’s findings, the Uniform Determination of Death Act, is also under review. In 2022, the Uniform Law Commission, a nonpartisan group of legal experts that drafts laws intended for adoption in multiple states, has taken up the work to revisit the definition of death.
The group will consider sharpening the medical standards for brain death in light of advances in the understanding of brain function. And they will look to address lingering questions raised in recent years as families and religious groups have waged heated legal battles over terminating artificial life support for patients with no brain wave activity.
Mr. Bopp, with the National Right to Life Committee, is among those serving on advisory panels for the effort, along with an array of doctors, philosophers, and medical ethicists. The concept of “personhood” that infuses the antiabortion movement’s broader push for fetal rights is expected to be an underlying topic, albeit in mirror image: When does a life form cease being a person?
Dr. Magnus, who is also serving on an advisory panel, has no doubt the commission will reach a consensus, a sober resolution rooted in science. What’s less clear, he said, is whether in today’s political environment that updated definition will hold the same sway, an enduring legal standard embraced across states.
KHN (Kaiser Health News) is a national newsroom that produces in-depth journalism about health issues. Together with Policy Analysis and Polling, KHN is one of the three major operating programs at KFF (Kaiser Family Foundation). KFF is an endowed nonprofit organization providing information on health issues to the nation.
As life-preserving medical technology advanced in the second half of the 20th century, doctors and families were faced with a thorny decision, one with weighty legal and moral implications: How should we define when life ends? Cardiopulmonary bypass machines could keep the blood pumping and ventilators could maintain breathing long after a patient’s natural ability to perform those vital functions had ceased.
After decades of deliberations involving physicians, bioethicists, attorneys, and theologians, a U.S. presidential commission in 1981 settled on a scientifically derived dividing line between life and death that has endured, more or less, ever since: A person was considered dead when the entire brain – including the brain stem, its most primitive portion – was no longer functioning, even if other vital functions could be maintained indefinitely through artificial life support.
In the decades since, the committee’s criteria have served as a foundation for laws in most states adopting brain death as a standard for legal death.
Now, with the overturning of Roe v. Wade and dozens of states rushing to impose abortion restrictions, At conception, the hint of a heartbeat, a first breath, the ability to survive outside the womb with the help of the latest technology?
That we’ve been able to devise and apply uniform clinical standards for when life ends, but not when it begins, is due largely to the legal and political maelstrom around abortion. And in the 2 months since the U.S. Supreme Court issued its opinion in Dobbs v. Jackson Women’s Health Organization, eliminating a longstanding federal right to abortion, state legislators are eagerly bounding into that void, looking to codify into law assorted definitions of life that carry profound repercussions for abortion rights, birth control, and assisted reproduction, as well as civil and criminal law.
“The court said that when life begins is up to whoever is running your state – whether they are wrong or not, or you agree with them or not,” said Mary Ziegler, a law professor at the University of California, Davis, who has written several books on the history of abortion.
Unlike the debate over death, which delved into exquisite medical and scientific detail, the legislative scramble to determine when life’s building blocks reach a threshold that warrants government protection as human life has generally ignored the input of mainstream medical professionals.
Instead, red states across much of the South and portions of the Midwest are adopting language drafted by elected officials that is informed by conservative Christian doctrine, often with little scientific underpinning.
A handful of Republican-led states, including Arkansas, Kentucky, Missouri, and Oklahoma, have passed laws declaring that life begins at fertilization, a contention that opens the door to a host of pregnancy-related litigation. This includes wrongful death lawsuits brought on behalf of the estate of an embryo by disgruntled ex-partners against physicians and women who end a pregnancy or even miscarry. (One such lawsuit is underway in Arizona. Another reached the Alabama Supreme Court.)
In Kentucky, the law outlawing abortion uses morally explosive terms to define pregnancy as “the human female reproductive condition of having a living unborn human being within her body throughout the entire embryonic and fetal stages of the unborn child from fertilization to full gestation and childbirth.”
Several other states, including Georgia, have adopted measures equating life with the point at which an embryo’s nascent cardiac activity can be detected by an ultrasound, at around 6 weeks of gestation. Many such laws mischaracterize the flickering electrical impulses detectable at that stage as a heartbeat, including in Georgia, whose Department of Revenue recently announced that “any unborn child with a detectable human heartbeat” can be claimed as a dependent.
The Supreme Court’s 1973 decision in Roe v. Wade that established a constitutional right to abortion did not define a moment when life begins. The opinion, written by Justice Harry Blackmun, observed that the Constitution does not provide a definition of “person,” though it extends protections to those born or naturalized in the United States. The court majority made note of the many disparate views among religions and scientists on when life begins, and concluded it was not up to the states to adopt one theory of life.
Instead, Roe created a framework intended to balance a pregnant woman’s right to make decisions about her body with a public interest in protecting potential human life. That decision and a key ruling that followed generally recognized a woman’s right to abortion up to the point medical professionals judge a fetus viable to survive outside the uterus, at about 24 weeks of gestation.
In decisively overturning Roe in June, the Supreme Court’s conservative majority drew on legal arguments that have shaped another contentious end-of-life issue. The legal standard employed in Dobbs – that there is no right to abortion in the federal Constitution and that states can decide on their own – is the same rationale used in 1997 when the Supreme Court said terminally ill people did not have a constitutional right to medically assisted death. That decision, Washington v. Glucksberg, is mentioned 15 times in the majority opinion for Dobbs and a concurrence by Justice Clarence Thomas.
Often, the same groups that have led the fight to outlaw abortion have also challenged medical aid-in-dying laws. Even after Dobbs, so-called right-to-die laws remain far less common than those codifying state abortion rights. Ten states allow physicians to prescribe lethal doses of medicine for terminally ill patients. Doctors are still prohibited from administering the drugs.
James Bopp, general counsel for the National Right to Life Committee who has been central to the efforts to outlaw abortion, said that both abortion and medically assisted death, which he refers to as physician-assisted suicide, endanger society.
“Every individual human life has inherent value and is sacred,” said Mr. Bopp. “The government has the duty to protect that life.”
Both issues raise profound societal questions: Can the government keep a patient on life support against his wishes, or force a woman to give birth? Can states bar their own residents from going to other states to end a pregnancy, or prohibit out-of-state patients from coming in to seek medically assisted death? And who gets to decide, particularly if the answer imposes a singular religious viewpoint?
Just as there are legal implications that flow from determining a person’s death, from organ donation to inheritance, the implied rights held by a legally recognized zygote are potentially vast. Will death certificates be issued for every lost pregnancy? Will miscarriages be investigated? When will Social Security numbers be issued? How will census counts be tallied and congressional districts drawn?
Medical professionals and bioethicists caution that both the beginning and end of life are complicated biological processes that are not defined by a single identifiable moment – and are ill suited to the political arena.
“Unfortunately, biological occurrences are not events, they are processes,” said David Magnus, PhD, director of the Stanford (Calif.) Center for Biomedical Ethics.
Moreover, asking doctors “What is life?” or “What is death?” may miss the point, said Dr. Magnus: “Medicine can answer the question ‘When does a biological organism cease to exist?’ But they can’t answer the question ‘When does a person begin or end?’ because those are metaphysical issues.”
Ben Sarbey, a doctoral candidate in the department of philosophy at Duke University, Durham, N.C., who studies medical ethics, echoed that perspective, recounting the Paradox of the Heap, a thought experiment that involves placing grains of sand one on top of the next. The philosophical quandary is this: At what point do those grains of sand become something more – a heap?
“We’re going to have a rough time placing a dividing line that this counts as a person and this does not count as a person,” he said. “Many things count as life – a sperm counts as life, a person in a persistent vegetative state counts as life – but does that constitute a person that we should be protecting?”
Even as debate over the court’s abortion decision percolates, the 1981 federal statute that grew out of the presidential committee’s findings, the Uniform Determination of Death Act, is also under review. In 2022, the Uniform Law Commission, a nonpartisan group of legal experts that drafts laws intended for adoption in multiple states, has taken up the work to revisit the definition of death.
The group will consider sharpening the medical standards for brain death in light of advances in the understanding of brain function. And they will look to address lingering questions raised in recent years as families and religious groups have waged heated legal battles over terminating artificial life support for patients with no brain wave activity.
Mr. Bopp, with the National Right to Life Committee, is among those serving on advisory panels for the effort, along with an array of doctors, philosophers, and medical ethicists. The concept of “personhood” that infuses the antiabortion movement’s broader push for fetal rights is expected to be an underlying topic, albeit in mirror image: When does a life form cease being a person?
Dr. Magnus, who is also serving on an advisory panel, has no doubt the commission will reach a consensus, a sober resolution rooted in science. What’s less clear, he said, is whether in today’s political environment that updated definition will hold the same sway, an enduring legal standard embraced across states.
KHN (Kaiser Health News) is a national newsroom that produces in-depth journalism about health issues. Together with Policy Analysis and Polling, KHN is one of the three major operating programs at KFF (Kaiser Family Foundation). KFF is an endowed nonprofit organization providing information on health issues to the nation.
VA Amends Rule on Abortions
The US Department of Veterans Affairs (VA) has submitted an interim final rule amending its medical regulations to remove the exclusion on abortion counseling and establish exceptions to the exclusion on abortions.
With this rule, the VA will now offer veterans abortion counseling and abortions—specifically, when the life or health of the pregnant veteran would be endangered if the pregnancy were carried to term, or in cases of rape or incest. Beneficiaries enrolled in CHAMPVA will have the same access to care.
“As abortion bans come into force across the country,” the interim final rule indicates, “veterans in many states are no longer assured access to abortion services in their communities, even when those services are needed… Unless VA removes its existing prohibitions on abortion-related care and makes clear that needed abortion-related care is authorized, these veterans will face serious threats to their life and health.”
“This is a patient safety decision,” said Denis McDonough, Secretary of Veterans Affairs. “Pregnant veterans and VA beneficiaries deserve to have access to world-class reproductive care when they need it most. That’s what our nation owes them, and that’s what we at VA will deliver.”
The rule is the VA’s latest response to the June 24, 2022, Dobbs v Jackson Women’s Health Organization Supreme Court decision, which overruled Roe v Wade and Planned Parenthood of Southeastern Pennsylvania v Casey. “After Dobbs,” according to the rule summary, “certain States have begun to enforce existing abortion bans and restrictions on care, and are proposing and enacting new ones, creating urgent risks to the lives and health of pregnant veterans and CHAMPVA beneficiaries in these states.” The VA is “acting to help to ensure that, irrespective of what laws or policies States may impose” veterans will be able to receive needed care.
Restricting access to abortion care has well-documented adverse health consequences, including a higher risk of loss of future fertility, significant morbidity, or death. Veterans are also at greater risk of experiencing pregnancy-related complications due to increased rates of chronic health conditions. “We came to this decision after listening to VA health care providers and veterans across the country, who sounded the alarm that abortion restrictions are creating a medical emergency for those we serve,” said Under Secretary for Health Shereef Elnahal, MD, MBA. “Offering this care will save veterans’ health and lives, and there is nothing more important than that.”
Services will be authorized immediately after the interim final rule is published. VA is taking steps to guarantee abortion-related care anywhere in the country. VA employees, when working within the scope of their federal employment, may provide authorized services regardless of state restrictions.
The determination of whether the “life and health of the pregnant veteran would be endangered if the pregnancy were carried to term” will be made on a case-by-case basis after “careful consultation” between VA health care professionals and their patients, the VA says. In cases of rape or incest, self-reporting from a veteran or VA beneficiary will constitute sufficient evidence that an act of rape or incest occurred.
The interim final rule will be available for public comment for 30 days after it is published.
The US Department of Veterans Affairs (VA) has submitted an interim final rule amending its medical regulations to remove the exclusion on abortion counseling and establish exceptions to the exclusion on abortions.
With this rule, the VA will now offer veterans abortion counseling and abortions—specifically, when the life or health of the pregnant veteran would be endangered if the pregnancy were carried to term, or in cases of rape or incest. Beneficiaries enrolled in CHAMPVA will have the same access to care.
“As abortion bans come into force across the country,” the interim final rule indicates, “veterans in many states are no longer assured access to abortion services in their communities, even when those services are needed… Unless VA removes its existing prohibitions on abortion-related care and makes clear that needed abortion-related care is authorized, these veterans will face serious threats to their life and health.”
“This is a patient safety decision,” said Denis McDonough, Secretary of Veterans Affairs. “Pregnant veterans and VA beneficiaries deserve to have access to world-class reproductive care when they need it most. That’s what our nation owes them, and that’s what we at VA will deliver.”
The rule is the VA’s latest response to the June 24, 2022, Dobbs v Jackson Women’s Health Organization Supreme Court decision, which overruled Roe v Wade and Planned Parenthood of Southeastern Pennsylvania v Casey. “After Dobbs,” according to the rule summary, “certain States have begun to enforce existing abortion bans and restrictions on care, and are proposing and enacting new ones, creating urgent risks to the lives and health of pregnant veterans and CHAMPVA beneficiaries in these states.” The VA is “acting to help to ensure that, irrespective of what laws or policies States may impose” veterans will be able to receive needed care.
Restricting access to abortion care has well-documented adverse health consequences, including a higher risk of loss of future fertility, significant morbidity, or death. Veterans are also at greater risk of experiencing pregnancy-related complications due to increased rates of chronic health conditions. “We came to this decision after listening to VA health care providers and veterans across the country, who sounded the alarm that abortion restrictions are creating a medical emergency for those we serve,” said Under Secretary for Health Shereef Elnahal, MD, MBA. “Offering this care will save veterans’ health and lives, and there is nothing more important than that.”
Services will be authorized immediately after the interim final rule is published. VA is taking steps to guarantee abortion-related care anywhere in the country. VA employees, when working within the scope of their federal employment, may provide authorized services regardless of state restrictions.
The determination of whether the “life and health of the pregnant veteran would be endangered if the pregnancy were carried to term” will be made on a case-by-case basis after “careful consultation” between VA health care professionals and their patients, the VA says. In cases of rape or incest, self-reporting from a veteran or VA beneficiary will constitute sufficient evidence that an act of rape or incest occurred.
The interim final rule will be available for public comment for 30 days after it is published.
The US Department of Veterans Affairs (VA) has submitted an interim final rule amending its medical regulations to remove the exclusion on abortion counseling and establish exceptions to the exclusion on abortions.
With this rule, the VA will now offer veterans abortion counseling and abortions—specifically, when the life or health of the pregnant veteran would be endangered if the pregnancy were carried to term, or in cases of rape or incest. Beneficiaries enrolled in CHAMPVA will have the same access to care.
“As abortion bans come into force across the country,” the interim final rule indicates, “veterans in many states are no longer assured access to abortion services in their communities, even when those services are needed… Unless VA removes its existing prohibitions on abortion-related care and makes clear that needed abortion-related care is authorized, these veterans will face serious threats to their life and health.”
“This is a patient safety decision,” said Denis McDonough, Secretary of Veterans Affairs. “Pregnant veterans and VA beneficiaries deserve to have access to world-class reproductive care when they need it most. That’s what our nation owes them, and that’s what we at VA will deliver.”
The rule is the VA’s latest response to the June 24, 2022, Dobbs v Jackson Women’s Health Organization Supreme Court decision, which overruled Roe v Wade and Planned Parenthood of Southeastern Pennsylvania v Casey. “After Dobbs,” according to the rule summary, “certain States have begun to enforce existing abortion bans and restrictions on care, and are proposing and enacting new ones, creating urgent risks to the lives and health of pregnant veterans and CHAMPVA beneficiaries in these states.” The VA is “acting to help to ensure that, irrespective of what laws or policies States may impose” veterans will be able to receive needed care.
Restricting access to abortion care has well-documented adverse health consequences, including a higher risk of loss of future fertility, significant morbidity, or death. Veterans are also at greater risk of experiencing pregnancy-related complications due to increased rates of chronic health conditions. “We came to this decision after listening to VA health care providers and veterans across the country, who sounded the alarm that abortion restrictions are creating a medical emergency for those we serve,” said Under Secretary for Health Shereef Elnahal, MD, MBA. “Offering this care will save veterans’ health and lives, and there is nothing more important than that.”
Services will be authorized immediately after the interim final rule is published. VA is taking steps to guarantee abortion-related care anywhere in the country. VA employees, when working within the scope of their federal employment, may provide authorized services regardless of state restrictions.
The determination of whether the “life and health of the pregnant veteran would be endangered if the pregnancy were carried to term” will be made on a case-by-case basis after “careful consultation” between VA health care professionals and their patients, the VA says. In cases of rape or incest, self-reporting from a veteran or VA beneficiary will constitute sufficient evidence that an act of rape or incest occurred.
The interim final rule will be available for public comment for 30 days after it is published.






