User login
Long QT and Cardiac Arrest After Symptomatic Improvement of Pulmonary Edema
A case of extreme QT prolongation induced following symptomatic resolution of acute pulmonary edema is both relatively unknown and poorly understood.
Abnormalities in the T-wave morphology of an electrocardiogram (ECG) are classically attributed to ischemic cardiac disease. However, these changes can be seen in a variety of other etiologies, including noncardiac pathology, which should be considered whenever reviewing an ECG: central nervous system disease, including stroke and subarachnoid hemorrhage; hypothermia; pulmonary disease, such as pulmonary embolism or chronic obstructive pulmonary disease; myopericarditis; drug effects; and electrolyte abnormalities.
Prolongation of the QT interval, on the other hand, can be precipitated by medications, metabolic derangements, or genetic phenotypes. The QT interval is measured from the beginning of the QRS complex to the termination of the T wave and represents the total time for ventricular depolarization and repolarization. The QT interval must be corrected based on the patient’s heart rate, known as the QTc. As the QTc interval lengthens, there is increased risk of R-on-T phenomena, which may result in Torsades de Pointes (TdP). Typical features of TdP include an antecedent prolonged QTc, cyclic polymorphic ventricular tachycardia on the surface ECG, and either a short-lived spontaneously terminating course or degeneration into ventricular fibrillation (VF) and sudden cardiac death.1 These dysrhythmias become more likely as the QTc interval exceeds 500 msec.2
The combination of new-onset global T-wave inversions with prolongation of the QT interval has been reported in only a few limited conditions. Some known causes of these QT T changes include cardiac ischemia, status epilepticus, pheochromocytoma, and acute cocaine intoxication.3 One uncommon and rarely reported cause of extreme QT prolongation and T-wave inversion is acute pulmonary edema. The ECG findings are not present on initial patient presentation; rather the dynamic changes occur after resolution of the pulmonary symptoms. Despite significant ECG changes, all prior reported cases describe ECG normalization without significant morbidity.4,5 We report a case of extreme QT prolongation following acute pulmonary edema that resulted in cardiac arrest secondary to VF.
Case Presentation
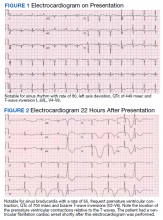
A 72-year-old male with medical history of combined systolic and diastolic heart failure, ischemic cardiomyopathy, coronary artery disease, cerebral vascular accident, hypertension, hyperlipidemia, type 2 diabetes mellitus, and tobacco dependence presented to the emergency department (ED) by emergency medical services after awaking with acute onset of dyspnea and diaphoresis. On arrival at the ED, the patient was noted to be in respiratory distress (ie, unable to speak single words) and was extremely diaphoretic. His initial vital signs included blood pressure, 186/113 mm Hg, heart rate, 104 beats per minute, respiratory rate, 40 breaths per minute, and temperature, 36.4 °C. The patient was quickly placed on bilevel positive airway pressure and given sublingual nitroglycerin followed by transdermal nitroglycerin with a single dose of 40 mg IV furosemide, which improved his respiratory status. A chest X-ray was consistent with pulmonary edema, and his brain natriuretic peptide was 1654 pg/mL. An ECG demonstrated new T-wave inversions, and his troponin increased from 0.04 to 0.24 ng/mL during his ED stay (Figure 1). He was started on a heparin infusion and admitted to the hospital for hypertensive emergency with presumed acute decompensated heart failure and non-ST-elevated myocardial infarction.
Throughout the patient’s first night, the troponin level started to down-trend after peaking at 0.24 ng/mL, and his oxygen requirements decreased allowing transition to nasal cannula. However, his repeat ECGs demonstrated significant T-wave abnormalities, new premature ventricular contractions, bradycardia, and a prolonging QTc interval to 703 msec (Figure 2). At this time, the patient’s electrolytes were normal, specifically a potassium level of 4.4 mEq/L, calcium 8.8 mg/dL, magnesium 2.0 mg/dL, and phosphorus 2.6 mg/dL. Given the worsening ECG changes, a computed tomography scan of his head was ordered to rule out intracranial pathology. While in the scanner, the patient went into pulseless VF, prompting defibrillation with 200 J. In addition, he was given 75 mg IV lidocaine, 2 g IV magnesium, and 1 ampule of both calcium chloride and sodium bicarbonate. With treatment, he had return of spontaneous circulation and was taken promptly to cardiac catheterization. The catheterization showed no significant obstructive coronary artery disease, and no interventions were performed. The patient was transferred to the cardiac intensive care unit for continued care.
During his course in the intensive care unit, the patient’s potassium and magnesium levels were maintained at high-normal levels. The patient was started on a dobutamine infusion to increase his heart rate and attempt to decrease his QTc. The patient also underwent cardiac magnetic resonance imaging (MRI) to evaluate for possible myocarditis, which showed no evidence of acute inflammation. Echocardiogram demonstrated an ejection fraction of 40% and global hypokinesis but no specific regional abnormalities and no change from prior echocardiogram performed 1 year earlier. Over the course of 3 days, his ECG normalized and his QTc shortened to 477 msec. Genetic testing was performed and did not reveal any mutations associated with long QT syndrome. Ultimately, an automated internal cardiac defibrillator (AICD) was placed, and the patient was discharged home.
Over the 2 years since his initial event, the patient has not experienced recurrent VF and his AICD has not fired. The patient continues to have ED presentations for heart-failure symptoms, though he has been stable from an electrophysiologic standpoint and his QTc remains less than 500 msec.
Discussion
Prolongation of the QT interval as a result of deep, global T-wave inversions after resolution of acute pulmonary edema has been minimally reported.4,5 This phenomenon has been described in the cardiology literature but has not been discussed in the emergency medicine literature and bears consideration in this case.4,5 As noted, an extensive evaluation did not reveal another cause of QTc prolongation. The patient had normal electrolytes and temperature, his neurologic examination and computed tomography were not remarkable. The patient had no obstructive coronary artery disease on catheterization, no evidence of acute myocarditis on cardiac MRI, no prescribed medications associated with QT prolongation, and no evidence of genetic mutations associated with QT prolongation on testing. The minimal troponin elevation was felt to represent a type II myocardial infarction related to ischemia due to supply-demand mismatch rather than acute plaque rupture.
Littmann published a case series of 9 cases of delayed onset T-wave inversion and extreme QTc prolongation in the 24 to 48 hours following treatment and symptomatic improvement in acute pulmonary edema.4 In each of his patients, an ischemic cardiac insult was ruled out as the etiology of the pulmonary edema by laboratory assessment, echocardiography, and left heart catheterization.All of the patients in this case series recovered without incident and with normalization of the QTc interval.4 Similarly, in our patient, significant QT T changes occurred approximately 22 hours after presentation and with resolution of symptoms of pulmonary edema. Pascale and colleagues also published a series of 3 patients developing similar ECG patterns following a hypertensive crisis with resolution of ECG findings and without any morbidity.5 In contrast, our patient experienced significant morbidity secondary to the extreme QTc prolongation.
Conclusions
We believe this is the first reported case of excessive prolongation of the QTc with VF arrest secondary to resolution of acute pulmonary edema. The pattern observed in our patient follows the patterns outlined in the previous case series—patients present with acute pulmonary edema and hypertensive crisis but develop significant ECG abnormalities about 24 hours after the resolution of the high catecholamine state. Our patient did have a history of prior cardiac insult, given the QTc changes developed acutely, with frequent premature ventricular contractions, and the cardiac arrest occurred at maximal QTc prolongation, yet after resolution of the high catecholamine state, the treatment team felt there was likely an uncaptured and short-lived episode of TdP that degenerated into VF. This theory is further supported by the lack of recurrent VF episodes, confirmed by AICD interrogation, after normalization of the QTc in our patient.
1. Passman R, Kadish A. Polymorphic ventricular tachycardia, long Q-T syndrome, and torsades de pointes. Med Clin North Am. 2001;85(2):321-341. doi:10.1016/s0025-7125(05)70318-7
2. Kallergis EM, Goudis CA, Simantirakis EN, Kochiadakis GE, Vardas PE. Mechanisms, risk factors, and management of acquired long QT syndrome: a comprehensive review. ScientificWorldJournal. 2012;2012:212178. doi:10.1100/2012/212178
3. Miller MA, Elmariah S, Fischer A. Giant T-wave inversions and extreme QT prolongation. Circ Arrhythm Electrophysiol. 2009;2(6):e42-e43. doi:10.1161/CIRCEP.108.825729
4. Littmann L. Large T wave inversion and QT prolongation associated with pulmonary edema: a report of nine cases. J Am Coll Cardiol. 1999;34(4):1106-1110. doi:10.1016/s0735-1097(99)00311-3
5. Pascale P, Quartenoud B, Stauffer JC. Isolated large inverted T wave in pulmonary edema due to hypertensive crisis: a novel electrocardiographic phenomenon mimicking ischemia?. Clin Res Cardiol. 2007;96(5):288-294. doi:10.1007/s00392-007-0504-1
A case of extreme QT prolongation induced following symptomatic resolution of acute pulmonary edema is both relatively unknown and poorly understood.
A case of extreme QT prolongation induced following symptomatic resolution of acute pulmonary edema is both relatively unknown and poorly understood.
Abnormalities in the T-wave morphology of an electrocardiogram (ECG) are classically attributed to ischemic cardiac disease. However, these changes can be seen in a variety of other etiologies, including noncardiac pathology, which should be considered whenever reviewing an ECG: central nervous system disease, including stroke and subarachnoid hemorrhage; hypothermia; pulmonary disease, such as pulmonary embolism or chronic obstructive pulmonary disease; myopericarditis; drug effects; and electrolyte abnormalities.
Prolongation of the QT interval, on the other hand, can be precipitated by medications, metabolic derangements, or genetic phenotypes. The QT interval is measured from the beginning of the QRS complex to the termination of the T wave and represents the total time for ventricular depolarization and repolarization. The QT interval must be corrected based on the patient’s heart rate, known as the QTc. As the QTc interval lengthens, there is increased risk of R-on-T phenomena, which may result in Torsades de Pointes (TdP). Typical features of TdP include an antecedent prolonged QTc, cyclic polymorphic ventricular tachycardia on the surface ECG, and either a short-lived spontaneously terminating course or degeneration into ventricular fibrillation (VF) and sudden cardiac death.1 These dysrhythmias become more likely as the QTc interval exceeds 500 msec.2
The combination of new-onset global T-wave inversions with prolongation of the QT interval has been reported in only a few limited conditions. Some known causes of these QT T changes include cardiac ischemia, status epilepticus, pheochromocytoma, and acute cocaine intoxication.3 One uncommon and rarely reported cause of extreme QT prolongation and T-wave inversion is acute pulmonary edema. The ECG findings are not present on initial patient presentation; rather the dynamic changes occur after resolution of the pulmonary symptoms. Despite significant ECG changes, all prior reported cases describe ECG normalization without significant morbidity.4,5 We report a case of extreme QT prolongation following acute pulmonary edema that resulted in cardiac arrest secondary to VF.
Case Presentation
A 72-year-old male with medical history of combined systolic and diastolic heart failure, ischemic cardiomyopathy, coronary artery disease, cerebral vascular accident, hypertension, hyperlipidemia, type 2 diabetes mellitus, and tobacco dependence presented to the emergency department (ED) by emergency medical services after awaking with acute onset of dyspnea and diaphoresis. On arrival at the ED, the patient was noted to be in respiratory distress (ie, unable to speak single words) and was extremely diaphoretic. His initial vital signs included blood pressure, 186/113 mm Hg, heart rate, 104 beats per minute, respiratory rate, 40 breaths per minute, and temperature, 36.4 °C. The patient was quickly placed on bilevel positive airway pressure and given sublingual nitroglycerin followed by transdermal nitroglycerin with a single dose of 40 mg IV furosemide, which improved his respiratory status. A chest X-ray was consistent with pulmonary edema, and his brain natriuretic peptide was 1654 pg/mL. An ECG demonstrated new T-wave inversions, and his troponin increased from 0.04 to 0.24 ng/mL during his ED stay (Figure 1). He was started on a heparin infusion and admitted to the hospital for hypertensive emergency with presumed acute decompensated heart failure and non-ST-elevated myocardial infarction.
Throughout the patient’s first night, the troponin level started to down-trend after peaking at 0.24 ng/mL, and his oxygen requirements decreased allowing transition to nasal cannula. However, his repeat ECGs demonstrated significant T-wave abnormalities, new premature ventricular contractions, bradycardia, and a prolonging QTc interval to 703 msec (Figure 2). At this time, the patient’s electrolytes were normal, specifically a potassium level of 4.4 mEq/L, calcium 8.8 mg/dL, magnesium 2.0 mg/dL, and phosphorus 2.6 mg/dL. Given the worsening ECG changes, a computed tomography scan of his head was ordered to rule out intracranial pathology. While in the scanner, the patient went into pulseless VF, prompting defibrillation with 200 J. In addition, he was given 75 mg IV lidocaine, 2 g IV magnesium, and 1 ampule of both calcium chloride and sodium bicarbonate. With treatment, he had return of spontaneous circulation and was taken promptly to cardiac catheterization. The catheterization showed no significant obstructive coronary artery disease, and no interventions were performed. The patient was transferred to the cardiac intensive care unit for continued care.
During his course in the intensive care unit, the patient’s potassium and magnesium levels were maintained at high-normal levels. The patient was started on a dobutamine infusion to increase his heart rate and attempt to decrease his QTc. The patient also underwent cardiac magnetic resonance imaging (MRI) to evaluate for possible myocarditis, which showed no evidence of acute inflammation. Echocardiogram demonstrated an ejection fraction of 40% and global hypokinesis but no specific regional abnormalities and no change from prior echocardiogram performed 1 year earlier. Over the course of 3 days, his ECG normalized and his QTc shortened to 477 msec. Genetic testing was performed and did not reveal any mutations associated with long QT syndrome. Ultimately, an automated internal cardiac defibrillator (AICD) was placed, and the patient was discharged home.
Over the 2 years since his initial event, the patient has not experienced recurrent VF and his AICD has not fired. The patient continues to have ED presentations for heart-failure symptoms, though he has been stable from an electrophysiologic standpoint and his QTc remains less than 500 msec.
Discussion
Prolongation of the QT interval as a result of deep, global T-wave inversions after resolution of acute pulmonary edema has been minimally reported.4,5 This phenomenon has been described in the cardiology literature but has not been discussed in the emergency medicine literature and bears consideration in this case.4,5 As noted, an extensive evaluation did not reveal another cause of QTc prolongation. The patient had normal electrolytes and temperature, his neurologic examination and computed tomography were not remarkable. The patient had no obstructive coronary artery disease on catheterization, no evidence of acute myocarditis on cardiac MRI, no prescribed medications associated with QT prolongation, and no evidence of genetic mutations associated with QT prolongation on testing. The minimal troponin elevation was felt to represent a type II myocardial infarction related to ischemia due to supply-demand mismatch rather than acute plaque rupture.
Littmann published a case series of 9 cases of delayed onset T-wave inversion and extreme QTc prolongation in the 24 to 48 hours following treatment and symptomatic improvement in acute pulmonary edema.4 In each of his patients, an ischemic cardiac insult was ruled out as the etiology of the pulmonary edema by laboratory assessment, echocardiography, and left heart catheterization.All of the patients in this case series recovered without incident and with normalization of the QTc interval.4 Similarly, in our patient, significant QT T changes occurred approximately 22 hours after presentation and with resolution of symptoms of pulmonary edema. Pascale and colleagues also published a series of 3 patients developing similar ECG patterns following a hypertensive crisis with resolution of ECG findings and without any morbidity.5 In contrast, our patient experienced significant morbidity secondary to the extreme QTc prolongation.
Conclusions
We believe this is the first reported case of excessive prolongation of the QTc with VF arrest secondary to resolution of acute pulmonary edema. The pattern observed in our patient follows the patterns outlined in the previous case series—patients present with acute pulmonary edema and hypertensive crisis but develop significant ECG abnormalities about 24 hours after the resolution of the high catecholamine state. Our patient did have a history of prior cardiac insult, given the QTc changes developed acutely, with frequent premature ventricular contractions, and the cardiac arrest occurred at maximal QTc prolongation, yet after resolution of the high catecholamine state, the treatment team felt there was likely an uncaptured and short-lived episode of TdP that degenerated into VF. This theory is further supported by the lack of recurrent VF episodes, confirmed by AICD interrogation, after normalization of the QTc in our patient.
Abnormalities in the T-wave morphology of an electrocardiogram (ECG) are classically attributed to ischemic cardiac disease. However, these changes can be seen in a variety of other etiologies, including noncardiac pathology, which should be considered whenever reviewing an ECG: central nervous system disease, including stroke and subarachnoid hemorrhage; hypothermia; pulmonary disease, such as pulmonary embolism or chronic obstructive pulmonary disease; myopericarditis; drug effects; and electrolyte abnormalities.
Prolongation of the QT interval, on the other hand, can be precipitated by medications, metabolic derangements, or genetic phenotypes. The QT interval is measured from the beginning of the QRS complex to the termination of the T wave and represents the total time for ventricular depolarization and repolarization. The QT interval must be corrected based on the patient’s heart rate, known as the QTc. As the QTc interval lengthens, there is increased risk of R-on-T phenomena, which may result in Torsades de Pointes (TdP). Typical features of TdP include an antecedent prolonged QTc, cyclic polymorphic ventricular tachycardia on the surface ECG, and either a short-lived spontaneously terminating course or degeneration into ventricular fibrillation (VF) and sudden cardiac death.1 These dysrhythmias become more likely as the QTc interval exceeds 500 msec.2
The combination of new-onset global T-wave inversions with prolongation of the QT interval has been reported in only a few limited conditions. Some known causes of these QT T changes include cardiac ischemia, status epilepticus, pheochromocytoma, and acute cocaine intoxication.3 One uncommon and rarely reported cause of extreme QT prolongation and T-wave inversion is acute pulmonary edema. The ECG findings are not present on initial patient presentation; rather the dynamic changes occur after resolution of the pulmonary symptoms. Despite significant ECG changes, all prior reported cases describe ECG normalization without significant morbidity.4,5 We report a case of extreme QT prolongation following acute pulmonary edema that resulted in cardiac arrest secondary to VF.
Case Presentation
A 72-year-old male with medical history of combined systolic and diastolic heart failure, ischemic cardiomyopathy, coronary artery disease, cerebral vascular accident, hypertension, hyperlipidemia, type 2 diabetes mellitus, and tobacco dependence presented to the emergency department (ED) by emergency medical services after awaking with acute onset of dyspnea and diaphoresis. On arrival at the ED, the patient was noted to be in respiratory distress (ie, unable to speak single words) and was extremely diaphoretic. His initial vital signs included blood pressure, 186/113 mm Hg, heart rate, 104 beats per minute, respiratory rate, 40 breaths per minute, and temperature, 36.4 °C. The patient was quickly placed on bilevel positive airway pressure and given sublingual nitroglycerin followed by transdermal nitroglycerin with a single dose of 40 mg IV furosemide, which improved his respiratory status. A chest X-ray was consistent with pulmonary edema, and his brain natriuretic peptide was 1654 pg/mL. An ECG demonstrated new T-wave inversions, and his troponin increased from 0.04 to 0.24 ng/mL during his ED stay (Figure 1). He was started on a heparin infusion and admitted to the hospital for hypertensive emergency with presumed acute decompensated heart failure and non-ST-elevated myocardial infarction.
Throughout the patient’s first night, the troponin level started to down-trend after peaking at 0.24 ng/mL, and his oxygen requirements decreased allowing transition to nasal cannula. However, his repeat ECGs demonstrated significant T-wave abnormalities, new premature ventricular contractions, bradycardia, and a prolonging QTc interval to 703 msec (Figure 2). At this time, the patient’s electrolytes were normal, specifically a potassium level of 4.4 mEq/L, calcium 8.8 mg/dL, magnesium 2.0 mg/dL, and phosphorus 2.6 mg/dL. Given the worsening ECG changes, a computed tomography scan of his head was ordered to rule out intracranial pathology. While in the scanner, the patient went into pulseless VF, prompting defibrillation with 200 J. In addition, he was given 75 mg IV lidocaine, 2 g IV magnesium, and 1 ampule of both calcium chloride and sodium bicarbonate. With treatment, he had return of spontaneous circulation and was taken promptly to cardiac catheterization. The catheterization showed no significant obstructive coronary artery disease, and no interventions were performed. The patient was transferred to the cardiac intensive care unit for continued care.
During his course in the intensive care unit, the patient’s potassium and magnesium levels were maintained at high-normal levels. The patient was started on a dobutamine infusion to increase his heart rate and attempt to decrease his QTc. The patient also underwent cardiac magnetic resonance imaging (MRI) to evaluate for possible myocarditis, which showed no evidence of acute inflammation. Echocardiogram demonstrated an ejection fraction of 40% and global hypokinesis but no specific regional abnormalities and no change from prior echocardiogram performed 1 year earlier. Over the course of 3 days, his ECG normalized and his QTc shortened to 477 msec. Genetic testing was performed and did not reveal any mutations associated with long QT syndrome. Ultimately, an automated internal cardiac defibrillator (AICD) was placed, and the patient was discharged home.
Over the 2 years since his initial event, the patient has not experienced recurrent VF and his AICD has not fired. The patient continues to have ED presentations for heart-failure symptoms, though he has been stable from an electrophysiologic standpoint and his QTc remains less than 500 msec.
Discussion
Prolongation of the QT interval as a result of deep, global T-wave inversions after resolution of acute pulmonary edema has been minimally reported.4,5 This phenomenon has been described in the cardiology literature but has not been discussed in the emergency medicine literature and bears consideration in this case.4,5 As noted, an extensive evaluation did not reveal another cause of QTc prolongation. The patient had normal electrolytes and temperature, his neurologic examination and computed tomography were not remarkable. The patient had no obstructive coronary artery disease on catheterization, no evidence of acute myocarditis on cardiac MRI, no prescribed medications associated with QT prolongation, and no evidence of genetic mutations associated with QT prolongation on testing. The minimal troponin elevation was felt to represent a type II myocardial infarction related to ischemia due to supply-demand mismatch rather than acute plaque rupture.
Littmann published a case series of 9 cases of delayed onset T-wave inversion and extreme QTc prolongation in the 24 to 48 hours following treatment and symptomatic improvement in acute pulmonary edema.4 In each of his patients, an ischemic cardiac insult was ruled out as the etiology of the pulmonary edema by laboratory assessment, echocardiography, and left heart catheterization.All of the patients in this case series recovered without incident and with normalization of the QTc interval.4 Similarly, in our patient, significant QT T changes occurred approximately 22 hours after presentation and with resolution of symptoms of pulmonary edema. Pascale and colleagues also published a series of 3 patients developing similar ECG patterns following a hypertensive crisis with resolution of ECG findings and without any morbidity.5 In contrast, our patient experienced significant morbidity secondary to the extreme QTc prolongation.
Conclusions
We believe this is the first reported case of excessive prolongation of the QTc with VF arrest secondary to resolution of acute pulmonary edema. The pattern observed in our patient follows the patterns outlined in the previous case series—patients present with acute pulmonary edema and hypertensive crisis but develop significant ECG abnormalities about 24 hours after the resolution of the high catecholamine state. Our patient did have a history of prior cardiac insult, given the QTc changes developed acutely, with frequent premature ventricular contractions, and the cardiac arrest occurred at maximal QTc prolongation, yet after resolution of the high catecholamine state, the treatment team felt there was likely an uncaptured and short-lived episode of TdP that degenerated into VF. This theory is further supported by the lack of recurrent VF episodes, confirmed by AICD interrogation, after normalization of the QTc in our patient.
1. Passman R, Kadish A. Polymorphic ventricular tachycardia, long Q-T syndrome, and torsades de pointes. Med Clin North Am. 2001;85(2):321-341. doi:10.1016/s0025-7125(05)70318-7
2. Kallergis EM, Goudis CA, Simantirakis EN, Kochiadakis GE, Vardas PE. Mechanisms, risk factors, and management of acquired long QT syndrome: a comprehensive review. ScientificWorldJournal. 2012;2012:212178. doi:10.1100/2012/212178
3. Miller MA, Elmariah S, Fischer A. Giant T-wave inversions and extreme QT prolongation. Circ Arrhythm Electrophysiol. 2009;2(6):e42-e43. doi:10.1161/CIRCEP.108.825729
4. Littmann L. Large T wave inversion and QT prolongation associated with pulmonary edema: a report of nine cases. J Am Coll Cardiol. 1999;34(4):1106-1110. doi:10.1016/s0735-1097(99)00311-3
5. Pascale P, Quartenoud B, Stauffer JC. Isolated large inverted T wave in pulmonary edema due to hypertensive crisis: a novel electrocardiographic phenomenon mimicking ischemia?. Clin Res Cardiol. 2007;96(5):288-294. doi:10.1007/s00392-007-0504-1
1. Passman R, Kadish A. Polymorphic ventricular tachycardia, long Q-T syndrome, and torsades de pointes. Med Clin North Am. 2001;85(2):321-341. doi:10.1016/s0025-7125(05)70318-7
2. Kallergis EM, Goudis CA, Simantirakis EN, Kochiadakis GE, Vardas PE. Mechanisms, risk factors, and management of acquired long QT syndrome: a comprehensive review. ScientificWorldJournal. 2012;2012:212178. doi:10.1100/2012/212178
3. Miller MA, Elmariah S, Fischer A. Giant T-wave inversions and extreme QT prolongation. Circ Arrhythm Electrophysiol. 2009;2(6):e42-e43. doi:10.1161/CIRCEP.108.825729
4. Littmann L. Large T wave inversion and QT prolongation associated with pulmonary edema: a report of nine cases. J Am Coll Cardiol. 1999;34(4):1106-1110. doi:10.1016/s0735-1097(99)00311-3
5. Pascale P, Quartenoud B, Stauffer JC. Isolated large inverted T wave in pulmonary edema due to hypertensive crisis: a novel electrocardiographic phenomenon mimicking ischemia?. Clin Res Cardiol. 2007;96(5):288-294. doi:10.1007/s00392-007-0504-1
Emphysematous Aortitis due to Klebsiella Pneumoniae in a Patient With Poorly Controlled Diabetes Mellitus
Patients with poorly controlled diabetes mellitus and an infectious source can be predisposed to infectious aortitis.
Aortitis is the all-encompassing term ascribed to the inflammatory process in the aortic wall that can be either infective or noninfective in origin, commonly autoimmune or inflammatory large-vessel vasculitis.1 Infectious aortitis, also known as bacterial, microbial, or cryptogenic aortitis, as well as mycotic or infected aneurysm, is a rare entity in the current antibiotic era but potentially a life-threatening disorder.2 The potential complications of infectious aortitis include emphysematous aortitis (EA), pseudoaneurysm, aortic rupture, septic emboli, and fistula formation (eg, aorto-enteric fistula).2,3
EA is a rare but serious inflammatory condition of the aorta with a nonspecific clinical presentation associated with high morbidity and mortality.2-6 The condition is characterized by a localized collection of gas and purulent exudate at the aortic wall.1,3 A few cases of EA have previously been reported; however, no known cases have been reported in the literature due to Klebsiella pneumoniae (K pneumoniae).
The pathophysiology of EA is the presence of underlying damage to the arterial wall caused by a hematogenously inoculated gas-producing organism.2,3 Most reported cases of EA are due to endovascular graft complications. Under normal circumstances, the aortic intima is highly resistant to infectious pathogens; however, certain risk factors, such as diabetes mellitus (DM), atherosclerotic disease, preexisting aneurysm, cystic medial necrosis, vascular malformation, presence of medical devices, surgery, or impaired immunity can alter the integrity of the aortic intimal layer and predispose the aortic intima to infection.1,4-7 Bacteria are the most common causative organisms that can infect the aorta, especially Staphylococcus, Enterococcus, Streptococcus, Salmonella, and spirochete Treponema pallidum (syphilis).1,2,4,8 The site of the primary infection remains unclear in some patients.2,3,5,6 Infection of the aorta can arise by several mechanisms: direct extension of a local infection to an existing intimal injury or atherosclerotic plaque (the most common mechanism), septic embolism from endocarditis, direct bacterial inoculation from traumatic contamination, contiguous infection extending to the aorta wall, or a distant source of bacteremia.2,3
Clinical manifestations of EA depend on the site and the extent of infection. The diagnosis should be considered in patients with atherosclerosis, fever, abdominal pain, and leukocytosis.2,4-8 The differential diagnosis for EA includes (1) noninfective causes of aortitis, including rheumatoid arthritis and systemic lupus erythematosus; (2) tuberculous aortitis; (3) syphilitic aortitis; and (4) idiopathic isolated aortitis. Establishing an early diagnosis of infectious aortitis is extremely important because this condition is associated with a high rate of morbidity and mortality secondary to aortic rupture.2-7
Imaging is critical for a reliable and quick diagnosis of acute aortic pathology. Noninvasive cross-sectional imaging modalities, such as contrast-enhanced computed tomography (CT), magnetic resonance imaging, nuclear medicine, or positron emission tomography, are used for both the initial diagnosis and follow-up of aortitis.1 CT is the primary imaging method in most medical centers because it is widely available with short acquisition time in critically ill patients.3 CT allows rapid detection of abnormalities in wall thickness, diameter, and density, and enhancement of periaortic structures, enabling reliable exclusion of other aortic pathologies that may resemble acute aortitis. Also, CT aids in planning the optimal therapeutic approach.1,3,5-8
This case illustrates EA associated with infection by K pneumoniae in a patient with poorly controlled type 2 DM (T2DM). In this single case, our patient presented to the Bay Pines Veterans Affairs Healthcare System (BPVAHS) in Florida with recent superficial soft tissue injury, severe hyperglycemia, worsening abdominal pain, and leukocytosis without fever or chills. The correct diagnosis of EA was confirmed by characteristic CT findings.
Case Presentation
A 72-year-old male with a history of T2DM, hypertension, atherosclerotic vascular disease, obstructive lung disease, and smoking 1.5 packs per day for 40 years presented with diabetic ketoacidosis, a urinary tract infection, and abdominal pain of 1-week duration that started to worsen the morning he arrived at the BPVAHS emergency department. He reported no nausea, vomiting, diarrhea, constipation, chest pain, shortness of breath, fever, chills, fatigue, or dysuria. He had a nonhealing laceration on his left medial foot that occurred 18 days before admission and was treated at an outside hospital.
The patient’s surgical history included a left common femoral endarterectomy and a left femoral popliteal above-knee reverse saphenous vein bypass 4 years ago for severe critical limb ischemia due to occlusion of his left superficial femoral artery with distal embolization to the first and fifth toes. About 6 months later, he developed disabling claudication in his left lower extremity due to distal popliteal artery occlusion and had another bypass surgery to the below-knee popliteal artery with a reverse saphenous vein graft harvested from the right thigh.
On initial examination, his vital signs were within normal limits except for a blood pressure of 177/87 mm Hg. His physical examination demonstrated a nondistended abdomen with normal bowel sounds, mild lower quadrant tenderness on the left more than on the right, intermittent abdominal pain located around umbilicus with radiation to the back, and a negative psoas sign. His left medial foot had a nonhealing laceration with black sutures in place, with minimal erythema in the surrounding tissue and scab formation. He also had mild costovertebral tenderness on the left.
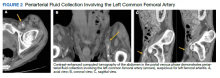
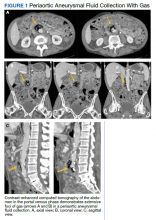
Initial laboratory investigation results were notable for a glucose level of 609 mg/dL and a white blood cell count of 14.6 × 103 cells/mcL with 86.5% of neutrophils. A CT scan of his abdomen revealed extensive atherosclerosis of the abdominal aorta and periaortic aneurysmal fluid collection with multiple foci of gas (Figure 1). Additionally, the aneurysmal fluid collection involved the proximal segment of the left common femoral artery, suspicious for left femoral arteritis (Figure 2). The patient was started on broad-spectrum antibiotics, morphine, and an insulin drip. Both urine and blood cultures were positive for K pneumoniae susceptible to multiple antibiotics. He was transferred to a tertiary medical center and was referred for a vascular surgery consultation.
The patient underwent surgical resection of the infected infrarenal EA and infected left common femoral artery with right axillary-bifemoral bypass with an 8-mm PTFE (polytetrafluoroethylene) graft. During the surgery, excision of the wall of the left common femoral artery and infrarenal aorta revealed frank pus with purulent fluid, which was sent to cytology for analysis and culture. His intraoperative cultures grew K pneumoniae sensitive to multiple antibiotics, including ceftriaxone, sulfamethoxazole/trimethoprim, and ampicillin/sulbactam. The vascular surgery team recommended inpatient admission and administration of 6 weeks of IV antibiotics postoperatively with ceftriaxone, followed by outpatient oral suppression therapy after discharge. The patient tolerated the surgery well and was discharged after 6 weeks of IV ceftriaxone followed by outpatient oral suppression therapy. However, the patient was transferred back to BPVAHS for continued care and rehabilitation placement.
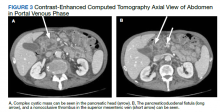
The patient’s subsequent course was complicated by multiple hospital admissions, including aspiration pneumonia, hypoglycemia, diarrhea, and anemia. On one of his CT abdomen/pelvic examinations, a cysticlike mass was noted in the pancreatic head with a possible pancreatic duodenal fistula (this mass was not mentioned on the initial presurgical CT, although it can be seen in retrospect (Figure 3). Gastroenterology was consulted.
An upper endoscopy was performed that confirmed the fistula at the second portion of the duodenum. Findings from an endoscopic ultrasonography performed at an outside institution were concerning for a main duct intraductal papillary mucinous neoplasm (IPMN) with fistula, with biopsy results pending.
Discussion
This case contributes to the evidence that poorly controlled T2DM can be a predisposing factor for multiple vascular complications, including the infection of the aortic wall with progression to EA. Klebsiella species are considered opportunistic, Gram-negative pathogens that may disseminate to other tissues, causing life-threatening infections, including pneumonia, UTIs, bacteremia, and sepsis.9K pneumoniae infections are particularly challenging in neonates, the elderly, and immunocompromised individuals.9 CT is sensitive and specific in the detection of this pathologic entity.1,3 In patients with a suspected infectious etiology, the presence of foci of gas on CT in solid organ tissues is usually associated with an anaerobic infection. Gas can also be produced by Gram-negative facultative anaerobes that can ferment glucose in necrotic tissues.9
Although any microorganism can infect the aorta, K pneumoniae cultured from the blood specimen, urine culture, and intraoperative specimens in our patient was responsible for the formed gas in the aortic wall. Occurrence of spontaneous gas by this microorganism is usually associated with conditions leading to either increased vulnerability to infections and/or enhanced bacterial virulence.9 Although a relationship between EA and T2DM has not been proved, it is well known that patients with T2DM have a defect in their host-defense mechanisms, making them more susceptible to infections such as EA. Furthermore, because patients with T2DM are prone to the development of Gram-negative sepsis, organisms such as K pneumoniae would tend to emerge. Patients with poorly controlled T2DM and the presence of an infectious source can be predisposed to infectious aortitis, eventually leading to a gas-forming infection of the aorta.5,7
We postulate that the hematogenous spread of bacteria from a laceration in the leg as well as the presence of the pancreaticoduodenal fistula was likely the cause of the infectious EA in this case, considering the patient’s underlying uncontrolled T2DM. The patient’s prior left lower extremity vascular graft also may have provided a nidus for spreading to the aorta. Other reported underlying diseases of EA include aortic atherosclerosis, T2DM, diverticulitis, colon cancer, underlying aneurysm, immune-compromised status, and the presence of a medical device or open surgery.4-7,9
To our knowledge, this is the first case of EA associated with a pancreaticoduodenal fistula related to intraductal papillary mucinous neoplasm (IPMN). Fistulation of a main duct IPMN is rare, occurring in just 6.6% of cases.10 It can occur both before and after malignant degeneration.
EA requires rapid diagnosis, antibiotic therapy, and consultation with a vascular surgeon for immediate resection of the infected artery and graft bypass. The initial treatment of suspected infectious aortitis is IV antibiotics with broad antimicrobial coverage of the most likely pathologic organisms, particularly staphylococcal species and Gram-negative rods. Surgical debridement and revascularization should be completed early because of the high mortality rate of this condition. The intent of surgery is to control sepsis and reconstruct the arterial vasculature. Patients should remain on parenteral or oral antibiotics for at least 6 weeks to ensure full clearance of the infection.8 They should be followed up closely with serial blood cultures and CT scans.8 The rarity of the disorder, low level of awareness, varying presentations, and lack of evidence delineating pathogenesis and causality contribute to the challenge of recognizing, diagnosing, and treating EA in patients with T2DM and inflammation.
Conclusions
This case report can help bring awareness of this rare and potentially life-threatening condition in patients with T2DM. Clinicians should be aware of the risk of AE, particularly in patients with several additional risk factors: recent skin/soft tissue trauma, prior vascular graft surgery, and an underlying pancreatic mass. CT is the imaging method of choice that helps to rapidly choose a necessary emergent treatment approach.
1. Litmanovich DE, Yıldırım A, Bankier AA. Insights into imaging of aortitis. Insights Imaging. 2012;3(6):545-560. doi:10.1007/s13244-012-0192-x
2. Lopes RJ, Almeida J, Dias PJ, Pinho P, Maciel MJ. Infectious thoracic aortitis: a literature review. Clin Cardiol. 2009;32(9):488-490. doi:10.1002/clc.20578
3. Murphy DJ, Keraliya AR, Agrawal MD, Aghayev A, Steigner ML. Cross-sectional imaging of aortic infections. Insights Imaging. 2016;7(6):801-818. doi:10.1007/s13244-016-0522-5
4. Md Noh MSF, Abdul Rashid AM, Ar A, B N, Mohammed Y, A RE. Emphysematous aortitis: report of two cases and CT imaging findings. BJR Case Rep. 2017;3(3):20170006. doi:10.1259/bjrcr.20170006
5. Harris C, Geffen J, Rizg K, et al. A rare report of infectious emphysematous aortitis secondary to Clostridium septicum without prior vascular intervention. Case Rep Vasc Med. 2017;2017:4984325. doi:10.1155/2017/4984325
6. Ito F, Inokuchi R, Matsumoto A, et al. Presence of periaortic gas in Clostridium septicum-infected aortic aneurysm aids in early diagnosis: a case report and systematic review of the literature. J Med Case Rep. 2017;11(1):268. doi:10.1186/s13256-017-1422-0
7. Urgiles S, Matos-Casano H, Win KZ, Berardo J, Bhatt U, Shah J. Emphysematous aortitis due to Clostridium septicum in an 89-year-old female with ileus. Case Rep Infect Dis. 2019;2019:1094837. doi:10.1155/2019/1094837
8. Foote EA, Postier RG, Greenfield RA, Bronze MS. Infectious aortitis. Curr Treat Options Cardiovasc Med. 2005;7(2):89-97. doi:10.1007/s11936-005-0010-6
9. Paczosa MK, Mecsas J. Klebsiella pneumoniae: going on the offense with a strong defense. Microbiol Mol Biol Rev. 2016;80(3):629-661. doi:10.1128/mmbr.00078-15
10. Kobayashi G, Fujita N, Noda Y, et al. Intraductal papillary mucinous neoplasms of the pancreas showing fistula formation into other organs. J Gastroenterol. 2010;45(10):1080-1089. doi:10.1007/s00535-010-0263-z
Patients with poorly controlled diabetes mellitus and an infectious source can be predisposed to infectious aortitis.
Patients with poorly controlled diabetes mellitus and an infectious source can be predisposed to infectious aortitis.
Aortitis is the all-encompassing term ascribed to the inflammatory process in the aortic wall that can be either infective or noninfective in origin, commonly autoimmune or inflammatory large-vessel vasculitis.1 Infectious aortitis, also known as bacterial, microbial, or cryptogenic aortitis, as well as mycotic or infected aneurysm, is a rare entity in the current antibiotic era but potentially a life-threatening disorder.2 The potential complications of infectious aortitis include emphysematous aortitis (EA), pseudoaneurysm, aortic rupture, septic emboli, and fistula formation (eg, aorto-enteric fistula).2,3
EA is a rare but serious inflammatory condition of the aorta with a nonspecific clinical presentation associated with high morbidity and mortality.2-6 The condition is characterized by a localized collection of gas and purulent exudate at the aortic wall.1,3 A few cases of EA have previously been reported; however, no known cases have been reported in the literature due to Klebsiella pneumoniae (K pneumoniae).
The pathophysiology of EA is the presence of underlying damage to the arterial wall caused by a hematogenously inoculated gas-producing organism.2,3 Most reported cases of EA are due to endovascular graft complications. Under normal circumstances, the aortic intima is highly resistant to infectious pathogens; however, certain risk factors, such as diabetes mellitus (DM), atherosclerotic disease, preexisting aneurysm, cystic medial necrosis, vascular malformation, presence of medical devices, surgery, or impaired immunity can alter the integrity of the aortic intimal layer and predispose the aortic intima to infection.1,4-7 Bacteria are the most common causative organisms that can infect the aorta, especially Staphylococcus, Enterococcus, Streptococcus, Salmonella, and spirochete Treponema pallidum (syphilis).1,2,4,8 The site of the primary infection remains unclear in some patients.2,3,5,6 Infection of the aorta can arise by several mechanisms: direct extension of a local infection to an existing intimal injury or atherosclerotic plaque (the most common mechanism), septic embolism from endocarditis, direct bacterial inoculation from traumatic contamination, contiguous infection extending to the aorta wall, or a distant source of bacteremia.2,3
Clinical manifestations of EA depend on the site and the extent of infection. The diagnosis should be considered in patients with atherosclerosis, fever, abdominal pain, and leukocytosis.2,4-8 The differential diagnosis for EA includes (1) noninfective causes of aortitis, including rheumatoid arthritis and systemic lupus erythematosus; (2) tuberculous aortitis; (3) syphilitic aortitis; and (4) idiopathic isolated aortitis. Establishing an early diagnosis of infectious aortitis is extremely important because this condition is associated with a high rate of morbidity and mortality secondary to aortic rupture.2-7
Imaging is critical for a reliable and quick diagnosis of acute aortic pathology. Noninvasive cross-sectional imaging modalities, such as contrast-enhanced computed tomography (CT), magnetic resonance imaging, nuclear medicine, or positron emission tomography, are used for both the initial diagnosis and follow-up of aortitis.1 CT is the primary imaging method in most medical centers because it is widely available with short acquisition time in critically ill patients.3 CT allows rapid detection of abnormalities in wall thickness, diameter, and density, and enhancement of periaortic structures, enabling reliable exclusion of other aortic pathologies that may resemble acute aortitis. Also, CT aids in planning the optimal therapeutic approach.1,3,5-8
This case illustrates EA associated with infection by K pneumoniae in a patient with poorly controlled type 2 DM (T2DM). In this single case, our patient presented to the Bay Pines Veterans Affairs Healthcare System (BPVAHS) in Florida with recent superficial soft tissue injury, severe hyperglycemia, worsening abdominal pain, and leukocytosis without fever or chills. The correct diagnosis of EA was confirmed by characteristic CT findings.
Case Presentation
A 72-year-old male with a history of T2DM, hypertension, atherosclerotic vascular disease, obstructive lung disease, and smoking 1.5 packs per day for 40 years presented with diabetic ketoacidosis, a urinary tract infection, and abdominal pain of 1-week duration that started to worsen the morning he arrived at the BPVAHS emergency department. He reported no nausea, vomiting, diarrhea, constipation, chest pain, shortness of breath, fever, chills, fatigue, or dysuria. He had a nonhealing laceration on his left medial foot that occurred 18 days before admission and was treated at an outside hospital.
The patient’s surgical history included a left common femoral endarterectomy and a left femoral popliteal above-knee reverse saphenous vein bypass 4 years ago for severe critical limb ischemia due to occlusion of his left superficial femoral artery with distal embolization to the first and fifth toes. About 6 months later, he developed disabling claudication in his left lower extremity due to distal popliteal artery occlusion and had another bypass surgery to the below-knee popliteal artery with a reverse saphenous vein graft harvested from the right thigh.
On initial examination, his vital signs were within normal limits except for a blood pressure of 177/87 mm Hg. His physical examination demonstrated a nondistended abdomen with normal bowel sounds, mild lower quadrant tenderness on the left more than on the right, intermittent abdominal pain located around umbilicus with radiation to the back, and a negative psoas sign. His left medial foot had a nonhealing laceration with black sutures in place, with minimal erythema in the surrounding tissue and scab formation. He also had mild costovertebral tenderness on the left.
Initial laboratory investigation results were notable for a glucose level of 609 mg/dL and a white blood cell count of 14.6 × 103 cells/mcL with 86.5% of neutrophils. A CT scan of his abdomen revealed extensive atherosclerosis of the abdominal aorta and periaortic aneurysmal fluid collection with multiple foci of gas (Figure 1). Additionally, the aneurysmal fluid collection involved the proximal segment of the left common femoral artery, suspicious for left femoral arteritis (Figure 2). The patient was started on broad-spectrum antibiotics, morphine, and an insulin drip. Both urine and blood cultures were positive for K pneumoniae susceptible to multiple antibiotics. He was transferred to a tertiary medical center and was referred for a vascular surgery consultation.
The patient underwent surgical resection of the infected infrarenal EA and infected left common femoral artery with right axillary-bifemoral bypass with an 8-mm PTFE (polytetrafluoroethylene) graft. During the surgery, excision of the wall of the left common femoral artery and infrarenal aorta revealed frank pus with purulent fluid, which was sent to cytology for analysis and culture. His intraoperative cultures grew K pneumoniae sensitive to multiple antibiotics, including ceftriaxone, sulfamethoxazole/trimethoprim, and ampicillin/sulbactam. The vascular surgery team recommended inpatient admission and administration of 6 weeks of IV antibiotics postoperatively with ceftriaxone, followed by outpatient oral suppression therapy after discharge. The patient tolerated the surgery well and was discharged after 6 weeks of IV ceftriaxone followed by outpatient oral suppression therapy. However, the patient was transferred back to BPVAHS for continued care and rehabilitation placement.
The patient’s subsequent course was complicated by multiple hospital admissions, including aspiration pneumonia, hypoglycemia, diarrhea, and anemia. On one of his CT abdomen/pelvic examinations, a cysticlike mass was noted in the pancreatic head with a possible pancreatic duodenal fistula (this mass was not mentioned on the initial presurgical CT, although it can be seen in retrospect (Figure 3). Gastroenterology was consulted.
An upper endoscopy was performed that confirmed the fistula at the second portion of the duodenum. Findings from an endoscopic ultrasonography performed at an outside institution were concerning for a main duct intraductal papillary mucinous neoplasm (IPMN) with fistula, with biopsy results pending.
Discussion
This case contributes to the evidence that poorly controlled T2DM can be a predisposing factor for multiple vascular complications, including the infection of the aortic wall with progression to EA. Klebsiella species are considered opportunistic, Gram-negative pathogens that may disseminate to other tissues, causing life-threatening infections, including pneumonia, UTIs, bacteremia, and sepsis.9K pneumoniae infections are particularly challenging in neonates, the elderly, and immunocompromised individuals.9 CT is sensitive and specific in the detection of this pathologic entity.1,3 In patients with a suspected infectious etiology, the presence of foci of gas on CT in solid organ tissues is usually associated with an anaerobic infection. Gas can also be produced by Gram-negative facultative anaerobes that can ferment glucose in necrotic tissues.9
Although any microorganism can infect the aorta, K pneumoniae cultured from the blood specimen, urine culture, and intraoperative specimens in our patient was responsible for the formed gas in the aortic wall. Occurrence of spontaneous gas by this microorganism is usually associated with conditions leading to either increased vulnerability to infections and/or enhanced bacterial virulence.9 Although a relationship between EA and T2DM has not been proved, it is well known that patients with T2DM have a defect in their host-defense mechanisms, making them more susceptible to infections such as EA. Furthermore, because patients with T2DM are prone to the development of Gram-negative sepsis, organisms such as K pneumoniae would tend to emerge. Patients with poorly controlled T2DM and the presence of an infectious source can be predisposed to infectious aortitis, eventually leading to a gas-forming infection of the aorta.5,7
We postulate that the hematogenous spread of bacteria from a laceration in the leg as well as the presence of the pancreaticoduodenal fistula was likely the cause of the infectious EA in this case, considering the patient’s underlying uncontrolled T2DM. The patient’s prior left lower extremity vascular graft also may have provided a nidus for spreading to the aorta. Other reported underlying diseases of EA include aortic atherosclerosis, T2DM, diverticulitis, colon cancer, underlying aneurysm, immune-compromised status, and the presence of a medical device or open surgery.4-7,9
To our knowledge, this is the first case of EA associated with a pancreaticoduodenal fistula related to intraductal papillary mucinous neoplasm (IPMN). Fistulation of a main duct IPMN is rare, occurring in just 6.6% of cases.10 It can occur both before and after malignant degeneration.
EA requires rapid diagnosis, antibiotic therapy, and consultation with a vascular surgeon for immediate resection of the infected artery and graft bypass. The initial treatment of suspected infectious aortitis is IV antibiotics with broad antimicrobial coverage of the most likely pathologic organisms, particularly staphylococcal species and Gram-negative rods. Surgical debridement and revascularization should be completed early because of the high mortality rate of this condition. The intent of surgery is to control sepsis and reconstruct the arterial vasculature. Patients should remain on parenteral or oral antibiotics for at least 6 weeks to ensure full clearance of the infection.8 They should be followed up closely with serial blood cultures and CT scans.8 The rarity of the disorder, low level of awareness, varying presentations, and lack of evidence delineating pathogenesis and causality contribute to the challenge of recognizing, diagnosing, and treating EA in patients with T2DM and inflammation.
Conclusions
This case report can help bring awareness of this rare and potentially life-threatening condition in patients with T2DM. Clinicians should be aware of the risk of AE, particularly in patients with several additional risk factors: recent skin/soft tissue trauma, prior vascular graft surgery, and an underlying pancreatic mass. CT is the imaging method of choice that helps to rapidly choose a necessary emergent treatment approach.
Aortitis is the all-encompassing term ascribed to the inflammatory process in the aortic wall that can be either infective or noninfective in origin, commonly autoimmune or inflammatory large-vessel vasculitis.1 Infectious aortitis, also known as bacterial, microbial, or cryptogenic aortitis, as well as mycotic or infected aneurysm, is a rare entity in the current antibiotic era but potentially a life-threatening disorder.2 The potential complications of infectious aortitis include emphysematous aortitis (EA), pseudoaneurysm, aortic rupture, septic emboli, and fistula formation (eg, aorto-enteric fistula).2,3
EA is a rare but serious inflammatory condition of the aorta with a nonspecific clinical presentation associated with high morbidity and mortality.2-6 The condition is characterized by a localized collection of gas and purulent exudate at the aortic wall.1,3 A few cases of EA have previously been reported; however, no known cases have been reported in the literature due to Klebsiella pneumoniae (K pneumoniae).
The pathophysiology of EA is the presence of underlying damage to the arterial wall caused by a hematogenously inoculated gas-producing organism.2,3 Most reported cases of EA are due to endovascular graft complications. Under normal circumstances, the aortic intima is highly resistant to infectious pathogens; however, certain risk factors, such as diabetes mellitus (DM), atherosclerotic disease, preexisting aneurysm, cystic medial necrosis, vascular malformation, presence of medical devices, surgery, or impaired immunity can alter the integrity of the aortic intimal layer and predispose the aortic intima to infection.1,4-7 Bacteria are the most common causative organisms that can infect the aorta, especially Staphylococcus, Enterococcus, Streptococcus, Salmonella, and spirochete Treponema pallidum (syphilis).1,2,4,8 The site of the primary infection remains unclear in some patients.2,3,5,6 Infection of the aorta can arise by several mechanisms: direct extension of a local infection to an existing intimal injury or atherosclerotic plaque (the most common mechanism), septic embolism from endocarditis, direct bacterial inoculation from traumatic contamination, contiguous infection extending to the aorta wall, or a distant source of bacteremia.2,3
Clinical manifestations of EA depend on the site and the extent of infection. The diagnosis should be considered in patients with atherosclerosis, fever, abdominal pain, and leukocytosis.2,4-8 The differential diagnosis for EA includes (1) noninfective causes of aortitis, including rheumatoid arthritis and systemic lupus erythematosus; (2) tuberculous aortitis; (3) syphilitic aortitis; and (4) idiopathic isolated aortitis. Establishing an early diagnosis of infectious aortitis is extremely important because this condition is associated with a high rate of morbidity and mortality secondary to aortic rupture.2-7
Imaging is critical for a reliable and quick diagnosis of acute aortic pathology. Noninvasive cross-sectional imaging modalities, such as contrast-enhanced computed tomography (CT), magnetic resonance imaging, nuclear medicine, or positron emission tomography, are used for both the initial diagnosis and follow-up of aortitis.1 CT is the primary imaging method in most medical centers because it is widely available with short acquisition time in critically ill patients.3 CT allows rapid detection of abnormalities in wall thickness, diameter, and density, and enhancement of periaortic structures, enabling reliable exclusion of other aortic pathologies that may resemble acute aortitis. Also, CT aids in planning the optimal therapeutic approach.1,3,5-8
This case illustrates EA associated with infection by K pneumoniae in a patient with poorly controlled type 2 DM (T2DM). In this single case, our patient presented to the Bay Pines Veterans Affairs Healthcare System (BPVAHS) in Florida with recent superficial soft tissue injury, severe hyperglycemia, worsening abdominal pain, and leukocytosis without fever or chills. The correct diagnosis of EA was confirmed by characteristic CT findings.
Case Presentation
A 72-year-old male with a history of T2DM, hypertension, atherosclerotic vascular disease, obstructive lung disease, and smoking 1.5 packs per day for 40 years presented with diabetic ketoacidosis, a urinary tract infection, and abdominal pain of 1-week duration that started to worsen the morning he arrived at the BPVAHS emergency department. He reported no nausea, vomiting, diarrhea, constipation, chest pain, shortness of breath, fever, chills, fatigue, or dysuria. He had a nonhealing laceration on his left medial foot that occurred 18 days before admission and was treated at an outside hospital.
The patient’s surgical history included a left common femoral endarterectomy and a left femoral popliteal above-knee reverse saphenous vein bypass 4 years ago for severe critical limb ischemia due to occlusion of his left superficial femoral artery with distal embolization to the first and fifth toes. About 6 months later, he developed disabling claudication in his left lower extremity due to distal popliteal artery occlusion and had another bypass surgery to the below-knee popliteal artery with a reverse saphenous vein graft harvested from the right thigh.
On initial examination, his vital signs were within normal limits except for a blood pressure of 177/87 mm Hg. His physical examination demonstrated a nondistended abdomen with normal bowel sounds, mild lower quadrant tenderness on the left more than on the right, intermittent abdominal pain located around umbilicus with radiation to the back, and a negative psoas sign. His left medial foot had a nonhealing laceration with black sutures in place, with minimal erythema in the surrounding tissue and scab formation. He also had mild costovertebral tenderness on the left.
Initial laboratory investigation results were notable for a glucose level of 609 mg/dL and a white blood cell count of 14.6 × 103 cells/mcL with 86.5% of neutrophils. A CT scan of his abdomen revealed extensive atherosclerosis of the abdominal aorta and periaortic aneurysmal fluid collection with multiple foci of gas (Figure 1). Additionally, the aneurysmal fluid collection involved the proximal segment of the left common femoral artery, suspicious for left femoral arteritis (Figure 2). The patient was started on broad-spectrum antibiotics, morphine, and an insulin drip. Both urine and blood cultures were positive for K pneumoniae susceptible to multiple antibiotics. He was transferred to a tertiary medical center and was referred for a vascular surgery consultation.
The patient underwent surgical resection of the infected infrarenal EA and infected left common femoral artery with right axillary-bifemoral bypass with an 8-mm PTFE (polytetrafluoroethylene) graft. During the surgery, excision of the wall of the left common femoral artery and infrarenal aorta revealed frank pus with purulent fluid, which was sent to cytology for analysis and culture. His intraoperative cultures grew K pneumoniae sensitive to multiple antibiotics, including ceftriaxone, sulfamethoxazole/trimethoprim, and ampicillin/sulbactam. The vascular surgery team recommended inpatient admission and administration of 6 weeks of IV antibiotics postoperatively with ceftriaxone, followed by outpatient oral suppression therapy after discharge. The patient tolerated the surgery well and was discharged after 6 weeks of IV ceftriaxone followed by outpatient oral suppression therapy. However, the patient was transferred back to BPVAHS for continued care and rehabilitation placement.
The patient’s subsequent course was complicated by multiple hospital admissions, including aspiration pneumonia, hypoglycemia, diarrhea, and anemia. On one of his CT abdomen/pelvic examinations, a cysticlike mass was noted in the pancreatic head with a possible pancreatic duodenal fistula (this mass was not mentioned on the initial presurgical CT, although it can be seen in retrospect (Figure 3). Gastroenterology was consulted.
An upper endoscopy was performed that confirmed the fistula at the second portion of the duodenum. Findings from an endoscopic ultrasonography performed at an outside institution were concerning for a main duct intraductal papillary mucinous neoplasm (IPMN) with fistula, with biopsy results pending.
Discussion
This case contributes to the evidence that poorly controlled T2DM can be a predisposing factor for multiple vascular complications, including the infection of the aortic wall with progression to EA. Klebsiella species are considered opportunistic, Gram-negative pathogens that may disseminate to other tissues, causing life-threatening infections, including pneumonia, UTIs, bacteremia, and sepsis.9K pneumoniae infections are particularly challenging in neonates, the elderly, and immunocompromised individuals.9 CT is sensitive and specific in the detection of this pathologic entity.1,3 In patients with a suspected infectious etiology, the presence of foci of gas on CT in solid organ tissues is usually associated with an anaerobic infection. Gas can also be produced by Gram-negative facultative anaerobes that can ferment glucose in necrotic tissues.9
Although any microorganism can infect the aorta, K pneumoniae cultured from the blood specimen, urine culture, and intraoperative specimens in our patient was responsible for the formed gas in the aortic wall. Occurrence of spontaneous gas by this microorganism is usually associated with conditions leading to either increased vulnerability to infections and/or enhanced bacterial virulence.9 Although a relationship between EA and T2DM has not been proved, it is well known that patients with T2DM have a defect in their host-defense mechanisms, making them more susceptible to infections such as EA. Furthermore, because patients with T2DM are prone to the development of Gram-negative sepsis, organisms such as K pneumoniae would tend to emerge. Patients with poorly controlled T2DM and the presence of an infectious source can be predisposed to infectious aortitis, eventually leading to a gas-forming infection of the aorta.5,7
We postulate that the hematogenous spread of bacteria from a laceration in the leg as well as the presence of the pancreaticoduodenal fistula was likely the cause of the infectious EA in this case, considering the patient’s underlying uncontrolled T2DM. The patient’s prior left lower extremity vascular graft also may have provided a nidus for spreading to the aorta. Other reported underlying diseases of EA include aortic atherosclerosis, T2DM, diverticulitis, colon cancer, underlying aneurysm, immune-compromised status, and the presence of a medical device or open surgery.4-7,9
To our knowledge, this is the first case of EA associated with a pancreaticoduodenal fistula related to intraductal papillary mucinous neoplasm (IPMN). Fistulation of a main duct IPMN is rare, occurring in just 6.6% of cases.10 It can occur both before and after malignant degeneration.
EA requires rapid diagnosis, antibiotic therapy, and consultation with a vascular surgeon for immediate resection of the infected artery and graft bypass. The initial treatment of suspected infectious aortitis is IV antibiotics with broad antimicrobial coverage of the most likely pathologic organisms, particularly staphylococcal species and Gram-negative rods. Surgical debridement and revascularization should be completed early because of the high mortality rate of this condition. The intent of surgery is to control sepsis and reconstruct the arterial vasculature. Patients should remain on parenteral or oral antibiotics for at least 6 weeks to ensure full clearance of the infection.8 They should be followed up closely with serial blood cultures and CT scans.8 The rarity of the disorder, low level of awareness, varying presentations, and lack of evidence delineating pathogenesis and causality contribute to the challenge of recognizing, diagnosing, and treating EA in patients with T2DM and inflammation.
Conclusions
This case report can help bring awareness of this rare and potentially life-threatening condition in patients with T2DM. Clinicians should be aware of the risk of AE, particularly in patients with several additional risk factors: recent skin/soft tissue trauma, prior vascular graft surgery, and an underlying pancreatic mass. CT is the imaging method of choice that helps to rapidly choose a necessary emergent treatment approach.
1. Litmanovich DE, Yıldırım A, Bankier AA. Insights into imaging of aortitis. Insights Imaging. 2012;3(6):545-560. doi:10.1007/s13244-012-0192-x
2. Lopes RJ, Almeida J, Dias PJ, Pinho P, Maciel MJ. Infectious thoracic aortitis: a literature review. Clin Cardiol. 2009;32(9):488-490. doi:10.1002/clc.20578
3. Murphy DJ, Keraliya AR, Agrawal MD, Aghayev A, Steigner ML. Cross-sectional imaging of aortic infections. Insights Imaging. 2016;7(6):801-818. doi:10.1007/s13244-016-0522-5
4. Md Noh MSF, Abdul Rashid AM, Ar A, B N, Mohammed Y, A RE. Emphysematous aortitis: report of two cases and CT imaging findings. BJR Case Rep. 2017;3(3):20170006. doi:10.1259/bjrcr.20170006
5. Harris C, Geffen J, Rizg K, et al. A rare report of infectious emphysematous aortitis secondary to Clostridium septicum without prior vascular intervention. Case Rep Vasc Med. 2017;2017:4984325. doi:10.1155/2017/4984325
6. Ito F, Inokuchi R, Matsumoto A, et al. Presence of periaortic gas in Clostridium septicum-infected aortic aneurysm aids in early diagnosis: a case report and systematic review of the literature. J Med Case Rep. 2017;11(1):268. doi:10.1186/s13256-017-1422-0
7. Urgiles S, Matos-Casano H, Win KZ, Berardo J, Bhatt U, Shah J. Emphysematous aortitis due to Clostridium septicum in an 89-year-old female with ileus. Case Rep Infect Dis. 2019;2019:1094837. doi:10.1155/2019/1094837
8. Foote EA, Postier RG, Greenfield RA, Bronze MS. Infectious aortitis. Curr Treat Options Cardiovasc Med. 2005;7(2):89-97. doi:10.1007/s11936-005-0010-6
9. Paczosa MK, Mecsas J. Klebsiella pneumoniae: going on the offense with a strong defense. Microbiol Mol Biol Rev. 2016;80(3):629-661. doi:10.1128/mmbr.00078-15
10. Kobayashi G, Fujita N, Noda Y, et al. Intraductal papillary mucinous neoplasms of the pancreas showing fistula formation into other organs. J Gastroenterol. 2010;45(10):1080-1089. doi:10.1007/s00535-010-0263-z
1. Litmanovich DE, Yıldırım A, Bankier AA. Insights into imaging of aortitis. Insights Imaging. 2012;3(6):545-560. doi:10.1007/s13244-012-0192-x
2. Lopes RJ, Almeida J, Dias PJ, Pinho P, Maciel MJ. Infectious thoracic aortitis: a literature review. Clin Cardiol. 2009;32(9):488-490. doi:10.1002/clc.20578
3. Murphy DJ, Keraliya AR, Agrawal MD, Aghayev A, Steigner ML. Cross-sectional imaging of aortic infections. Insights Imaging. 2016;7(6):801-818. doi:10.1007/s13244-016-0522-5
4. Md Noh MSF, Abdul Rashid AM, Ar A, B N, Mohammed Y, A RE. Emphysematous aortitis: report of two cases and CT imaging findings. BJR Case Rep. 2017;3(3):20170006. doi:10.1259/bjrcr.20170006
5. Harris C, Geffen J, Rizg K, et al. A rare report of infectious emphysematous aortitis secondary to Clostridium septicum without prior vascular intervention. Case Rep Vasc Med. 2017;2017:4984325. doi:10.1155/2017/4984325
6. Ito F, Inokuchi R, Matsumoto A, et al. Presence of periaortic gas in Clostridium septicum-infected aortic aneurysm aids in early diagnosis: a case report and systematic review of the literature. J Med Case Rep. 2017;11(1):268. doi:10.1186/s13256-017-1422-0
7. Urgiles S, Matos-Casano H, Win KZ, Berardo J, Bhatt U, Shah J. Emphysematous aortitis due to Clostridium septicum in an 89-year-old female with ileus. Case Rep Infect Dis. 2019;2019:1094837. doi:10.1155/2019/1094837
8. Foote EA, Postier RG, Greenfield RA, Bronze MS. Infectious aortitis. Curr Treat Options Cardiovasc Med. 2005;7(2):89-97. doi:10.1007/s11936-005-0010-6
9. Paczosa MK, Mecsas J. Klebsiella pneumoniae: going on the offense with a strong defense. Microbiol Mol Biol Rev. 2016;80(3):629-661. doi:10.1128/mmbr.00078-15
10. Kobayashi G, Fujita N, Noda Y, et al. Intraductal papillary mucinous neoplasms of the pancreas showing fistula formation into other organs. J Gastroenterol. 2010;45(10):1080-1089. doi:10.1007/s00535-010-0263-z
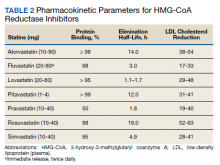
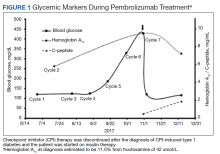
Rosuvastatin-Induced Rhabdomyolysis, Pancreatitis, Transaminitis, and Acute Kidney Injury
Changing medications within a drug class requires considering the indication and dosage, possible adverse effects, and drug-drug interactions.
Attention should be paid to changing a tolerated medication to another within its class. Many drugs approved by the US Food and Drug Administration (FDA), have equivalent therapeutic properties as existing drugs. Rarely do such medications share the same potency and adverse effect (AE) profile.
Case Presentation
A 77-year-old man presented to the emergency department (ED) at the Raymond G. Murphy Medical Center in Albuquerque, New Mexico, with a 1-month history of progressive muscle weakness, which was so severe that he required assistance rising from chairs. The symptoms began when he switched from atorvastatin 40 mg daily to rosuvastatin 40 mg daily. A nephrology consultation was requested for an elevated plasma creatinine.
The patient reported strict adherence to his prescribed medications. In the days following the switch to rosuvastatin, he noticed that his urine turned black. He described the color as “like burnt coffee.” The color gradually cleared before his ED presentation. The patient stopped taking rosuvastatin the day prior to presentation and noted improvement of his symptoms. Review of symptoms was significant for lower extremity paresthesia and numbness the day he started rosuvastatin. He had no symptoms of decompensated heart failure and no recent exacerbations requiring alteration of his diuretic regimen.
The patient’s medical history was significant for traumatic brain injury with complex partial seizures, carpal tunnel syndrome, dyslipidemia, coronary artery disease with percutaneous intervention to the right coronary artery in the late 1990s, atrial fibrillation and ventricular tachycardia, status post implantable cardioverter defibrillator, heart failure with reduced ejection fraction (25%) attributed to ischemic cardiomyopathy, hypertension, lower urinary tract symptoms/prostatism, and previous bladder cancer. In the mid-1960s, the patient served in the US Army and had been deployed to South Korea. After the service, he worked for the local city government. He was retired for about 15 years. He reported no tobacco, alcohol, or recreational drug use and no tattoos. He did not require prior blood or blood product transfusions. None of his family members—parents, siblings, or children—had any history of kidney disease.
The patient’s outpatient medications included levetiracetam 750 mg twice daily, melatonin 9 mg at night, menthol 16%/methyl-salicylate 30% topically up to 4 times per day as needed, aspirin 81 mg once daily, fish oil 1000 mg twice daily, amiodarone 400 mg twice daily, hydralazine 20 mg 3 times daily, isosorbide mononitrate 60 mg daily, metoprolol succinate 100 mg daily, and tamsulosin 0.4 mg at night. His vital signs were stable: afebrile (97.5 ºF), normocardic (74 beats per minute), normotensive (118/78 mm Hg), and normoxic (98% on room air). On examination, he appeared elderly, somewhat frail, and chronically ill but in no acute distress. Affect was pleasant and appropriate, attention was high, and his thought process was logical. He had sparse, grey scalp hair. Extraocular movements were intact. Oral mucosa was pink and moist. His back was nontender, and there was no costovertebral tenderness bilaterally. The patient was in no respiratory distress, with a slightly hyperresonant chest to percussion bilaterally, very faint inspiratory basilar crepitant rales (that cleared with repeat inspiration), and was otherwise clear to auscultation throughout. An outline of an implanted pacemaker was evident on the chest under his left clavicle, with a laterally displaced apical impulse. The rate was normal and the rhythm was regular. Upper extremities demonstrated papyraceous skin but without cyanosis, clubbing, or edema. Radial pulses were slightly diminished. He had no lower extremity edema.
His laboratory values are provided in Table 1. Kidney function was stable months prior to admission. Of note, the blood urea nitrogen and plasma creatinine were increased from his baseline up to 47 and 5.89 mg/dL, respectively. The serum glutamic-oxaloacetic transaminase and serum glutamic pyruvic transaminase were 1051 U/L and 408 U/L, respectively. Plasma amylase and lipase levels also were elevated, 230 U/L and 892 U/L, respectively. Creatine kinase was 41,099 U/L. Urinalysis demonstrated a specific gravity of 1.017, pH of 5, and a large amount of blood (92 red blood cells/high power field).
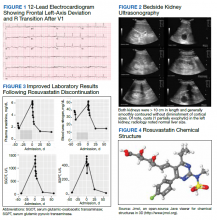
A 12-lead electrocardiogram demonstrated a sinus rhythm, PR interval of 0.20 ms, narrow QRS with a leftward frontal axis deviation, R-transition between precordial leads V1 and V2, and flattening of the ST segments in III, V1-V3 (Figure 1). A portable chest X-ray demonstrated clear lung fields, no evidence of effusion in the costophrenic area. Ultrasonography was conducted at the time of the examination (Figure 2). The kidneys were smoothly contoured, each measuring > 10 cm; there was an exophytic cyst on the left. Otherwise, the cortices, perhaps slightly echogenic, did not appear diminished. The bladder was not abnormally enlarged.
Rosuvastatin-induced rhabdomyolysis, pancreatitis, transaminitis, and drug-induced acute kidney injury were considered high among the diagnostic differentials. The 3-hydroxy-3-methyl-glutaryl-CoA (HMG-CoA) reductase inhibitor was stopped, and he was prescribed an acute renal insufficiency diet. All laboratory parameters improved with this change (Figure 3). Two months after presentation (and with rosuvastatin added to his list of adverse reactions), all symptoms resolved and his plasma creatinine reached a nadir of 1.22 mg/dL.
Discussion
Statin-class drugs inhibit the HMG-CoA reductase (Table 2). Upregulation of low-density lipoprotein cholesterol (LDL-C) receptors in the liver result in increased LDL-C uptake and cholesterol catabolism.1 Prescribed inhibitors of the HMG-CoA reductase—statins—are known to reduce mortality due to cardiovascular disease (CVD). Much like any other pharmaceutical agent with any measurable potency, HMG-CoA inhibitors can have AEs. Statin therapy has been associated with pancreatitis.2 Muscle toxicity is a complication of HMG-CoA reductase inhibitors, and statin-associated symptoms are a leading cause of nonadherence.3 Rosuvastatin had higher AE and drug reactions compared with that of atorvastatin and pitavastatin (35.6%, 8.7%, and 22.2%, respectively) in clinical trials for approval.4 We have reported concomitant adermatopathic dermatomyositis with statin-induced myopathy in a 48-year-old man from simvastatin (40 to 80 mg daily).1
Toxin-induced myopathy should be considered early in the differential diagnosis of weakness.5 All HMG-CoA inhibitors have been associated with acute kidney injury, particularly at high doses and also are known to induce myopathies, sometimes with inclusion bodies.1 Muscle-related AEs correlate with the potency of an HMG-CoA reductase inhibitor according to an analysis using the FDA AE Reporting System (AERS).6 Myalgia and rhabdomyolysis are well-known AEs of this class of medications. Furthermore, type II muscle atrophy—particularly in the proximal limb muscles—has been reported.5 Patients may have difficulty rising from chairs.1 Rosuvastatin had the strongest signal for muscular AEs (eg, myalgia, rhabdomyolysis, increased creatine phosphokinase level) from an FDA analysis of AERS.7
Rosuvastatin is the only HMG-CoA reductase inhibitor that causes dose-dependent increases in proteinuria and hematuria (Figure 4).8 Rosuvastatin at a 5-mg dose may induce 4 times the proteinuria as a placebo. Typically, other statins potentially reduce proteinuria (without hematuria). Proteinuria may be induced by rosuvastatin even at low doses.8 Proteinuria is attributed to how rosuvastatin impacts proximal tubular function.9 The drug is transported into the proximal tubule by the organic anion transporter-3. Acute kidney injury has been associated with several statins, including rosuvastatin.7,10 This may be associated with denuded tubular epithelia, active urinary sediment, acute tubular toxicity, vacuolated epithelial cells, and tubular cell casts. Unlike atorvastatin, the increase in proteinuria and hematuria also is dose dependent.
In patients with renal insufficiency (short of end-stage renal disease [ESRD]), most statins other than rosuvastatin are well tolerated and recommended for reduction of overall and CVD mortality risk. However, these benefits seem to diminish once ESRD is reached. Atorvastatin did not impact CVD mortality in patients with type 2 diabetes mellitus (T2DM) and ESRD (despite decreasing LDL-C).11 The AURORA study randomized 10 mg of statin vs placebo in 2776 maintenance dialysis patients aged 50 to 80 years. Rosuvastatin lowered the LDL-C but did not affect all-cause mortality (13.5 vs 14.0 events per 100 patient-years). Patients randomized to rosuvastatin had more than twice as many unclassified strokes (9 vs 4). Rosuvastatin, although efficacious in reducing LDL-C, had no impact on CVD mortality, nonfatal myocardial infarction, or nonfatal stroke.12 Post hoc analysis demonstrated that in patients with T2DM with ESRD the hazard ratio for hemorrhagic stroke was 5.2.13
Rosuvastatin ranked lower than lovastatin, pravastatin, simvastatin, atorvastatin, and fluvastatin with respect to reduction of all-cause mortality in trials of participants with or without prior coronary artery disease.14 AEs, such as rhabdomyolysis, proteinuria, nephropathy, renal failure, liver, and muscle toxicity are higher with rosuvastatin than other medications in its class.15
Conclusions
For patients with existing CVD, standard clinical practice is to encourage increased and regular physical activity, cholesterol-lowering diets, weight loss, and smoking cessation. Hypertension should be treated. Glycemia should be well controlled in the setting of T2DM. β-blockers may be beneficial in those with histories of myocardial infarction or heart failure with reduced systolic function. Statins are a valuable tool in the treatment of dyslipidemia.
Statin-induced muscle symptoms are a major reason for discontinuation and nonadherence.16 Statin-induced myalgia, myositis, and myopathy have been used interchangeably.17 Rhabdomyolysis, myalgia, increased creatine kinase, statin myopathy, and immune-mediated necrotizing myopathy are among the clinical phenotypes caused by statins.17 There are 33,695 serious cases—1808 deaths—reported with rosuvastatin in the FDA AERS as of June 30, 2021. Myalgia, pain in extremity, muscle spasms, pain, and arthralgia top the list of AEs. When statin-induced symptoms occur, adherence is rarely improved by dismissive clinicians.18
Drugs in the same class often have common therapeutic properties. Potencies and AE profiles are seldom uniform. The decision to add or change the brand of medication within a class should be balanced with considerations for the indication, duplications, simplification, AEs, appropriate dosage, and drug-drug interactions.
Acknowledgments
Brent Wagner is funded by a US Department of Veterans Affairs Merit Award (I01 BX001958), a National Institutes of Health R01 grant (DK-102085), Dialysis Clinic, Inc., and partially supported by the University of New Mexico Brain and Behavioral Health Institute (BBHI 2018-1008, 2020-21-002) and in part by the University of New Mexico’s Signature Program in Cardiovascular and Metabolic Disease (CVMD); and the University of New Mexico School of Medicine Research Allocation Committee (C-2459-RAC, New Mexico Medical Trust). Brent Wagner is an Associate Member to the University of New Mexico Health Sciences Center Autophagy, Inflammation, and Metabolism Center of Biomedical Research Excellence (AIM CoBRE) supported by NIH grant P20GM121176.
Funding
National Institutes of Health Grant R01 DK-102085, Dialysis Clinic Inc., VA Merit Award I01 BX001958, Center for Integrated Nanotechnologies User Agreement 2019AU0120, Brain & Behavioral Health Institute (grants 2018-1008, 2020-21-002), University of New Mexico’s Signature Program in Cardiovascular and Metabolic Disease (CVMD), the University of New Mexico School of Medicine Research Allocation Committee (C-2459-RAC, New Mexico Medical Trust) and a metabolomics voucher from the AIM Center (NIH P20GM121176).
1. Wagner B, Kagan-Hallet KS, Russell IJ. Concomitant presentation of adermatopathic dermatomyositis, statin myopathy, fibromyalgia syndrome, piriformis muscle myofascial pain and diabetic neuropathy. J Musculoskeletal Pain. 2003;11(2):25-30. doi:10.1300/J094v11n02_05
2. Collins R, Reith C, Emberson J, et al. Interpretation of the evidence for the efficacy and safety of statin therapy [published correction appears in Lancet. 2017 Feb 11;389(10069):602]. Lancet. 2016;388(10059):2532-2561. doi:10.1016/S0140-6736(16)31357-5
3. Stroes ES, Thompson PD, Corsini A, et al. Statin-associated muscle symptoms: impact on statin therapy-European Atherosclerosis Society Consensus Panel Statement on Assessment, Aetiology and Management. Eur Heart J. 2015;36(17):1012-1022. doi:10.1093/eurheartj/ehv043
4. Saku K, Zhang B, Noda K; PATROL Trial Investigators. Randomized head-to-head comparison of pitavastatin, atorvastatin, and rosuvastatin for safety and efficacy (quantity and quality of LDL): the PATROL trial. Circ J. 2011;75(6):1493-1505. doi:10.1253/circj.cj-10-1281
5. Wald JJ. The effects of toxins on muscle. Neurol Clin. 2000;18(3):695-718. doi:10.1016/s0733-8619(05)70219-x
6. Hoffman KB, Kraus C, Dimbil M, Golomb BA. A survey of the FDA’s AERS database regarding muscle and tendon adverse events linked to the statin drug class. PLoS One. 2012;7(8):e42866. doi:10.1371/journal.pone.0042866
7. Sakaeda T, Kadoyama K, Okuno Y. Statin-associated muscular and renal adverse events: data mining of the public version of the FDA adverse event reporting system. PLoS One. 2011;6(12):e28124. doi:10.1371/journal.pone.0028124
8. Tiwari A. An overview of statin-associated proteinuria. Drug Discov Today. 2006;11(9-10):458-464. doi:10.1016/j.drudis.2006.03.017
9. Verhulst A, Sayer R, De Broe ME, D’Haese PC, Brown CD. Human proximal tubular epithelium actively secretes but does not retain rosuvastatin. Mol Pharmacol. 2008;74(4):1084-1091. doi:10.1124/mol.108.047647
10. Jones PH, Davidson MH, Stein EA, et al. Comparison of the efficacy and safety of rosuvastatin versus atorvastatin, simvastatin, and pravastatin across doses (STELLAR* Trial). Am J Cardiol. 2003;92(2):152-160. doi:10.1016/s0002-9149(03)00530-7
11. Wanner C, Krane V, März W, et al. Atorvastatin in patients with type 2 diabetes mellitus undergoing hemodialysis [published correction appears in N Engl J Med. 2005 Oct 13;353(15):1640]. N Engl J Med. 2005;353(3):238-248. doi:10.1056/NEJMoa043545
12. Fellström BC, Jardine AG, Schmieder RE, et al. Rosuvastatin and cardiovascular events in patients undergoing hemodialysis [published correction appears in N Engl J Med. 2010 Apr 15;362(15):1450]. N Engl J Med. 2009;360(14):1395-1407. doi:10.1056/NEJMoa0810177
13. Holdaas H, Holme I, Schmieder RE, et al. Rosuvastatin in diabetic hemodialysis patients. J Am Soc Nephrol. 2011;22(7):1335-1341. doi:10.1681/ASN.2010090987
14. Naci H, Brugts JJ, Fleurence R, Tsoi B, Toor H, Ades AE. Comparative benefits of statins in the primary and secondary prevention of major coronary events and all-cause mortality: a network meta-analysis of placebo-controlled and active-comparator trials. Eur J Prev Cardiol. 2013;20(4):641-657. doi:10.1177/2047487313480435
15. Alsheikh-Ali AA, Ambrose MS, Kuvin JT, Karas RH. The safety of rosuvastatin as used in common clinical practice: a postmarketing analysis. Circulation. 2005;111(23):3051-3057. doi:10.1161/CIRCULATIONAHA.105.555482
16. Ward NC, Watts GF, Eckel RH. Statin toxicity. Circ Res. 2019;124(2):328-350. doi:10.1161/CIRCRESAHA.118.312782
17. Selva-O’Callaghan A, Alvarado-Cardenas M, Pinal-Fernández I, et al. Statin-induced myalgia and myositis: an update on pathogenesis and clinical recommendations. Expert Rev Clin Immunol. 2018;14(3):215-224. doi:10.1080/1744666X.2018.1440206
18. Koslik HJ, Meskimen AH, Golomb BA. Physicians’ Experiences as patients with statin side effects: a case series. Drug Saf Case Rep. 2017;4(1):3. doi:10.1007/s40800-017-0045-0
Changing medications within a drug class requires considering the indication and dosage, possible adverse effects, and drug-drug interactions.
Changing medications within a drug class requires considering the indication and dosage, possible adverse effects, and drug-drug interactions.
Attention should be paid to changing a tolerated medication to another within its class. Many drugs approved by the US Food and Drug Administration (FDA), have equivalent therapeutic properties as existing drugs. Rarely do such medications share the same potency and adverse effect (AE) profile.
Case Presentation
A 77-year-old man presented to the emergency department (ED) at the Raymond G. Murphy Medical Center in Albuquerque, New Mexico, with a 1-month history of progressive muscle weakness, which was so severe that he required assistance rising from chairs. The symptoms began when he switched from atorvastatin 40 mg daily to rosuvastatin 40 mg daily. A nephrology consultation was requested for an elevated plasma creatinine.
The patient reported strict adherence to his prescribed medications. In the days following the switch to rosuvastatin, he noticed that his urine turned black. He described the color as “like burnt coffee.” The color gradually cleared before his ED presentation. The patient stopped taking rosuvastatin the day prior to presentation and noted improvement of his symptoms. Review of symptoms was significant for lower extremity paresthesia and numbness the day he started rosuvastatin. He had no symptoms of decompensated heart failure and no recent exacerbations requiring alteration of his diuretic regimen.
The patient’s medical history was significant for traumatic brain injury with complex partial seizures, carpal tunnel syndrome, dyslipidemia, coronary artery disease with percutaneous intervention to the right coronary artery in the late 1990s, atrial fibrillation and ventricular tachycardia, status post implantable cardioverter defibrillator, heart failure with reduced ejection fraction (25%) attributed to ischemic cardiomyopathy, hypertension, lower urinary tract symptoms/prostatism, and previous bladder cancer. In the mid-1960s, the patient served in the US Army and had been deployed to South Korea. After the service, he worked for the local city government. He was retired for about 15 years. He reported no tobacco, alcohol, or recreational drug use and no tattoos. He did not require prior blood or blood product transfusions. None of his family members—parents, siblings, or children—had any history of kidney disease.
The patient’s outpatient medications included levetiracetam 750 mg twice daily, melatonin 9 mg at night, menthol 16%/methyl-salicylate 30% topically up to 4 times per day as needed, aspirin 81 mg once daily, fish oil 1000 mg twice daily, amiodarone 400 mg twice daily, hydralazine 20 mg 3 times daily, isosorbide mononitrate 60 mg daily, metoprolol succinate 100 mg daily, and tamsulosin 0.4 mg at night. His vital signs were stable: afebrile (97.5 ºF), normocardic (74 beats per minute), normotensive (118/78 mm Hg), and normoxic (98% on room air). On examination, he appeared elderly, somewhat frail, and chronically ill but in no acute distress. Affect was pleasant and appropriate, attention was high, and his thought process was logical. He had sparse, grey scalp hair. Extraocular movements were intact. Oral mucosa was pink and moist. His back was nontender, and there was no costovertebral tenderness bilaterally. The patient was in no respiratory distress, with a slightly hyperresonant chest to percussion bilaterally, very faint inspiratory basilar crepitant rales (that cleared with repeat inspiration), and was otherwise clear to auscultation throughout. An outline of an implanted pacemaker was evident on the chest under his left clavicle, with a laterally displaced apical impulse. The rate was normal and the rhythm was regular. Upper extremities demonstrated papyraceous skin but without cyanosis, clubbing, or edema. Radial pulses were slightly diminished. He had no lower extremity edema.
His laboratory values are provided in Table 1. Kidney function was stable months prior to admission. Of note, the blood urea nitrogen and plasma creatinine were increased from his baseline up to 47 and 5.89 mg/dL, respectively. The serum glutamic-oxaloacetic transaminase and serum glutamic pyruvic transaminase were 1051 U/L and 408 U/L, respectively. Plasma amylase and lipase levels also were elevated, 230 U/L and 892 U/L, respectively. Creatine kinase was 41,099 U/L. Urinalysis demonstrated a specific gravity of 1.017, pH of 5, and a large amount of blood (92 red blood cells/high power field).
A 12-lead electrocardiogram demonstrated a sinus rhythm, PR interval of 0.20 ms, narrow QRS with a leftward frontal axis deviation, R-transition between precordial leads V1 and V2, and flattening of the ST segments in III, V1-V3 (Figure 1). A portable chest X-ray demonstrated clear lung fields, no evidence of effusion in the costophrenic area. Ultrasonography was conducted at the time of the examination (Figure 2). The kidneys were smoothly contoured, each measuring > 10 cm; there was an exophytic cyst on the left. Otherwise, the cortices, perhaps slightly echogenic, did not appear diminished. The bladder was not abnormally enlarged.
Rosuvastatin-induced rhabdomyolysis, pancreatitis, transaminitis, and drug-induced acute kidney injury were considered high among the diagnostic differentials. The 3-hydroxy-3-methyl-glutaryl-CoA (HMG-CoA) reductase inhibitor was stopped, and he was prescribed an acute renal insufficiency diet. All laboratory parameters improved with this change (Figure 3). Two months after presentation (and with rosuvastatin added to his list of adverse reactions), all symptoms resolved and his plasma creatinine reached a nadir of 1.22 mg/dL.
Discussion
Statin-class drugs inhibit the HMG-CoA reductase (Table 2). Upregulation of low-density lipoprotein cholesterol (LDL-C) receptors in the liver result in increased LDL-C uptake and cholesterol catabolism.1 Prescribed inhibitors of the HMG-CoA reductase—statins—are known to reduce mortality due to cardiovascular disease (CVD). Much like any other pharmaceutical agent with any measurable potency, HMG-CoA inhibitors can have AEs. Statin therapy has been associated with pancreatitis.2 Muscle toxicity is a complication of HMG-CoA reductase inhibitors, and statin-associated symptoms are a leading cause of nonadherence.3 Rosuvastatin had higher AE and drug reactions compared with that of atorvastatin and pitavastatin (35.6%, 8.7%, and 22.2%, respectively) in clinical trials for approval.4 We have reported concomitant adermatopathic dermatomyositis with statin-induced myopathy in a 48-year-old man from simvastatin (40 to 80 mg daily).1
Toxin-induced myopathy should be considered early in the differential diagnosis of weakness.5 All HMG-CoA inhibitors have been associated with acute kidney injury, particularly at high doses and also are known to induce myopathies, sometimes with inclusion bodies.1 Muscle-related AEs correlate with the potency of an HMG-CoA reductase inhibitor according to an analysis using the FDA AE Reporting System (AERS).6 Myalgia and rhabdomyolysis are well-known AEs of this class of medications. Furthermore, type II muscle atrophy—particularly in the proximal limb muscles—has been reported.5 Patients may have difficulty rising from chairs.1 Rosuvastatin had the strongest signal for muscular AEs (eg, myalgia, rhabdomyolysis, increased creatine phosphokinase level) from an FDA analysis of AERS.7
Rosuvastatin is the only HMG-CoA reductase inhibitor that causes dose-dependent increases in proteinuria and hematuria (Figure 4).8 Rosuvastatin at a 5-mg dose may induce 4 times the proteinuria as a placebo. Typically, other statins potentially reduce proteinuria (without hematuria). Proteinuria may be induced by rosuvastatin even at low doses.8 Proteinuria is attributed to how rosuvastatin impacts proximal tubular function.9 The drug is transported into the proximal tubule by the organic anion transporter-3. Acute kidney injury has been associated with several statins, including rosuvastatin.7,10 This may be associated with denuded tubular epithelia, active urinary sediment, acute tubular toxicity, vacuolated epithelial cells, and tubular cell casts. Unlike atorvastatin, the increase in proteinuria and hematuria also is dose dependent.
In patients with renal insufficiency (short of end-stage renal disease [ESRD]), most statins other than rosuvastatin are well tolerated and recommended for reduction of overall and CVD mortality risk. However, these benefits seem to diminish once ESRD is reached. Atorvastatin did not impact CVD mortality in patients with type 2 diabetes mellitus (T2DM) and ESRD (despite decreasing LDL-C).11 The AURORA study randomized 10 mg of statin vs placebo in 2776 maintenance dialysis patients aged 50 to 80 years. Rosuvastatin lowered the LDL-C but did not affect all-cause mortality (13.5 vs 14.0 events per 100 patient-years). Patients randomized to rosuvastatin had more than twice as many unclassified strokes (9 vs 4). Rosuvastatin, although efficacious in reducing LDL-C, had no impact on CVD mortality, nonfatal myocardial infarction, or nonfatal stroke.12 Post hoc analysis demonstrated that in patients with T2DM with ESRD the hazard ratio for hemorrhagic stroke was 5.2.13
Rosuvastatin ranked lower than lovastatin, pravastatin, simvastatin, atorvastatin, and fluvastatin with respect to reduction of all-cause mortality in trials of participants with or without prior coronary artery disease.14 AEs, such as rhabdomyolysis, proteinuria, nephropathy, renal failure, liver, and muscle toxicity are higher with rosuvastatin than other medications in its class.15
Conclusions
For patients with existing CVD, standard clinical practice is to encourage increased and regular physical activity, cholesterol-lowering diets, weight loss, and smoking cessation. Hypertension should be treated. Glycemia should be well controlled in the setting of T2DM. β-blockers may be beneficial in those with histories of myocardial infarction or heart failure with reduced systolic function. Statins are a valuable tool in the treatment of dyslipidemia.
Statin-induced muscle symptoms are a major reason for discontinuation and nonadherence.16 Statin-induced myalgia, myositis, and myopathy have been used interchangeably.17 Rhabdomyolysis, myalgia, increased creatine kinase, statin myopathy, and immune-mediated necrotizing myopathy are among the clinical phenotypes caused by statins.17 There are 33,695 serious cases—1808 deaths—reported with rosuvastatin in the FDA AERS as of June 30, 2021. Myalgia, pain in extremity, muscle spasms, pain, and arthralgia top the list of AEs. When statin-induced symptoms occur, adherence is rarely improved by dismissive clinicians.18
Drugs in the same class often have common therapeutic properties. Potencies and AE profiles are seldom uniform. The decision to add or change the brand of medication within a class should be balanced with considerations for the indication, duplications, simplification, AEs, appropriate dosage, and drug-drug interactions.
Acknowledgments
Brent Wagner is funded by a US Department of Veterans Affairs Merit Award (I01 BX001958), a National Institutes of Health R01 grant (DK-102085), Dialysis Clinic, Inc., and partially supported by the University of New Mexico Brain and Behavioral Health Institute (BBHI 2018-1008, 2020-21-002) and in part by the University of New Mexico’s Signature Program in Cardiovascular and Metabolic Disease (CVMD); and the University of New Mexico School of Medicine Research Allocation Committee (C-2459-RAC, New Mexico Medical Trust). Brent Wagner is an Associate Member to the University of New Mexico Health Sciences Center Autophagy, Inflammation, and Metabolism Center of Biomedical Research Excellence (AIM CoBRE) supported by NIH grant P20GM121176.
Funding
National Institutes of Health Grant R01 DK-102085, Dialysis Clinic Inc., VA Merit Award I01 BX001958, Center for Integrated Nanotechnologies User Agreement 2019AU0120, Brain & Behavioral Health Institute (grants 2018-1008, 2020-21-002), University of New Mexico’s Signature Program in Cardiovascular and Metabolic Disease (CVMD), the University of New Mexico School of Medicine Research Allocation Committee (C-2459-RAC, New Mexico Medical Trust) and a metabolomics voucher from the AIM Center (NIH P20GM121176).
Attention should be paid to changing a tolerated medication to another within its class. Many drugs approved by the US Food and Drug Administration (FDA), have equivalent therapeutic properties as existing drugs. Rarely do such medications share the same potency and adverse effect (AE) profile.
Case Presentation
A 77-year-old man presented to the emergency department (ED) at the Raymond G. Murphy Medical Center in Albuquerque, New Mexico, with a 1-month history of progressive muscle weakness, which was so severe that he required assistance rising from chairs. The symptoms began when he switched from atorvastatin 40 mg daily to rosuvastatin 40 mg daily. A nephrology consultation was requested for an elevated plasma creatinine.
The patient reported strict adherence to his prescribed medications. In the days following the switch to rosuvastatin, he noticed that his urine turned black. He described the color as “like burnt coffee.” The color gradually cleared before his ED presentation. The patient stopped taking rosuvastatin the day prior to presentation and noted improvement of his symptoms. Review of symptoms was significant for lower extremity paresthesia and numbness the day he started rosuvastatin. He had no symptoms of decompensated heart failure and no recent exacerbations requiring alteration of his diuretic regimen.
The patient’s medical history was significant for traumatic brain injury with complex partial seizures, carpal tunnel syndrome, dyslipidemia, coronary artery disease with percutaneous intervention to the right coronary artery in the late 1990s, atrial fibrillation and ventricular tachycardia, status post implantable cardioverter defibrillator, heart failure with reduced ejection fraction (25%) attributed to ischemic cardiomyopathy, hypertension, lower urinary tract symptoms/prostatism, and previous bladder cancer. In the mid-1960s, the patient served in the US Army and had been deployed to South Korea. After the service, he worked for the local city government. He was retired for about 15 years. He reported no tobacco, alcohol, or recreational drug use and no tattoos. He did not require prior blood or blood product transfusions. None of his family members—parents, siblings, or children—had any history of kidney disease.
The patient’s outpatient medications included levetiracetam 750 mg twice daily, melatonin 9 mg at night, menthol 16%/methyl-salicylate 30% topically up to 4 times per day as needed, aspirin 81 mg once daily, fish oil 1000 mg twice daily, amiodarone 400 mg twice daily, hydralazine 20 mg 3 times daily, isosorbide mononitrate 60 mg daily, metoprolol succinate 100 mg daily, and tamsulosin 0.4 mg at night. His vital signs were stable: afebrile (97.5 ºF), normocardic (74 beats per minute), normotensive (118/78 mm Hg), and normoxic (98% on room air). On examination, he appeared elderly, somewhat frail, and chronically ill but in no acute distress. Affect was pleasant and appropriate, attention was high, and his thought process was logical. He had sparse, grey scalp hair. Extraocular movements were intact. Oral mucosa was pink and moist. His back was nontender, and there was no costovertebral tenderness bilaterally. The patient was in no respiratory distress, with a slightly hyperresonant chest to percussion bilaterally, very faint inspiratory basilar crepitant rales (that cleared with repeat inspiration), and was otherwise clear to auscultation throughout. An outline of an implanted pacemaker was evident on the chest under his left clavicle, with a laterally displaced apical impulse. The rate was normal and the rhythm was regular. Upper extremities demonstrated papyraceous skin but without cyanosis, clubbing, or edema. Radial pulses were slightly diminished. He had no lower extremity edema.
His laboratory values are provided in Table 1. Kidney function was stable months prior to admission. Of note, the blood urea nitrogen and plasma creatinine were increased from his baseline up to 47 and 5.89 mg/dL, respectively. The serum glutamic-oxaloacetic transaminase and serum glutamic pyruvic transaminase were 1051 U/L and 408 U/L, respectively. Plasma amylase and lipase levels also were elevated, 230 U/L and 892 U/L, respectively. Creatine kinase was 41,099 U/L. Urinalysis demonstrated a specific gravity of 1.017, pH of 5, and a large amount of blood (92 red blood cells/high power field).
A 12-lead electrocardiogram demonstrated a sinus rhythm, PR interval of 0.20 ms, narrow QRS with a leftward frontal axis deviation, R-transition between precordial leads V1 and V2, and flattening of the ST segments in III, V1-V3 (Figure 1). A portable chest X-ray demonstrated clear lung fields, no evidence of effusion in the costophrenic area. Ultrasonography was conducted at the time of the examination (Figure 2). The kidneys were smoothly contoured, each measuring > 10 cm; there was an exophytic cyst on the left. Otherwise, the cortices, perhaps slightly echogenic, did not appear diminished. The bladder was not abnormally enlarged.
Rosuvastatin-induced rhabdomyolysis, pancreatitis, transaminitis, and drug-induced acute kidney injury were considered high among the diagnostic differentials. The 3-hydroxy-3-methyl-glutaryl-CoA (HMG-CoA) reductase inhibitor was stopped, and he was prescribed an acute renal insufficiency diet. All laboratory parameters improved with this change (Figure 3). Two months after presentation (and with rosuvastatin added to his list of adverse reactions), all symptoms resolved and his plasma creatinine reached a nadir of 1.22 mg/dL.
Discussion
Statin-class drugs inhibit the HMG-CoA reductase (Table 2). Upregulation of low-density lipoprotein cholesterol (LDL-C) receptors in the liver result in increased LDL-C uptake and cholesterol catabolism.1 Prescribed inhibitors of the HMG-CoA reductase—statins—are known to reduce mortality due to cardiovascular disease (CVD). Much like any other pharmaceutical agent with any measurable potency, HMG-CoA inhibitors can have AEs. Statin therapy has been associated with pancreatitis.2 Muscle toxicity is a complication of HMG-CoA reductase inhibitors, and statin-associated symptoms are a leading cause of nonadherence.3 Rosuvastatin had higher AE and drug reactions compared with that of atorvastatin and pitavastatin (35.6%, 8.7%, and 22.2%, respectively) in clinical trials for approval.4 We have reported concomitant adermatopathic dermatomyositis with statin-induced myopathy in a 48-year-old man from simvastatin (40 to 80 mg daily).1
Toxin-induced myopathy should be considered early in the differential diagnosis of weakness.5 All HMG-CoA inhibitors have been associated with acute kidney injury, particularly at high doses and also are known to induce myopathies, sometimes with inclusion bodies.1 Muscle-related AEs correlate with the potency of an HMG-CoA reductase inhibitor according to an analysis using the FDA AE Reporting System (AERS).6 Myalgia and rhabdomyolysis are well-known AEs of this class of medications. Furthermore, type II muscle atrophy—particularly in the proximal limb muscles—has been reported.5 Patients may have difficulty rising from chairs.1 Rosuvastatin had the strongest signal for muscular AEs (eg, myalgia, rhabdomyolysis, increased creatine phosphokinase level) from an FDA analysis of AERS.7
Rosuvastatin is the only HMG-CoA reductase inhibitor that causes dose-dependent increases in proteinuria and hematuria (Figure 4).8 Rosuvastatin at a 5-mg dose may induce 4 times the proteinuria as a placebo. Typically, other statins potentially reduce proteinuria (without hematuria). Proteinuria may be induced by rosuvastatin even at low doses.8 Proteinuria is attributed to how rosuvastatin impacts proximal tubular function.9 The drug is transported into the proximal tubule by the organic anion transporter-3. Acute kidney injury has been associated with several statins, including rosuvastatin.7,10 This may be associated with denuded tubular epithelia, active urinary sediment, acute tubular toxicity, vacuolated epithelial cells, and tubular cell casts. Unlike atorvastatin, the increase in proteinuria and hematuria also is dose dependent.
In patients with renal insufficiency (short of end-stage renal disease [ESRD]), most statins other than rosuvastatin are well tolerated and recommended for reduction of overall and CVD mortality risk. However, these benefits seem to diminish once ESRD is reached. Atorvastatin did not impact CVD mortality in patients with type 2 diabetes mellitus (T2DM) and ESRD (despite decreasing LDL-C).11 The AURORA study randomized 10 mg of statin vs placebo in 2776 maintenance dialysis patients aged 50 to 80 years. Rosuvastatin lowered the LDL-C but did not affect all-cause mortality (13.5 vs 14.0 events per 100 patient-years). Patients randomized to rosuvastatin had more than twice as many unclassified strokes (9 vs 4). Rosuvastatin, although efficacious in reducing LDL-C, had no impact on CVD mortality, nonfatal myocardial infarction, or nonfatal stroke.12 Post hoc analysis demonstrated that in patients with T2DM with ESRD the hazard ratio for hemorrhagic stroke was 5.2.13
Rosuvastatin ranked lower than lovastatin, pravastatin, simvastatin, atorvastatin, and fluvastatin with respect to reduction of all-cause mortality in trials of participants with or without prior coronary artery disease.14 AEs, such as rhabdomyolysis, proteinuria, nephropathy, renal failure, liver, and muscle toxicity are higher with rosuvastatin than other medications in its class.15
Conclusions
For patients with existing CVD, standard clinical practice is to encourage increased and regular physical activity, cholesterol-lowering diets, weight loss, and smoking cessation. Hypertension should be treated. Glycemia should be well controlled in the setting of T2DM. β-blockers may be beneficial in those with histories of myocardial infarction or heart failure with reduced systolic function. Statins are a valuable tool in the treatment of dyslipidemia.
Statin-induced muscle symptoms are a major reason for discontinuation and nonadherence.16 Statin-induced myalgia, myositis, and myopathy have been used interchangeably.17 Rhabdomyolysis, myalgia, increased creatine kinase, statin myopathy, and immune-mediated necrotizing myopathy are among the clinical phenotypes caused by statins.17 There are 33,695 serious cases—1808 deaths—reported with rosuvastatin in the FDA AERS as of June 30, 2021. Myalgia, pain in extremity, muscle spasms, pain, and arthralgia top the list of AEs. When statin-induced symptoms occur, adherence is rarely improved by dismissive clinicians.18
Drugs in the same class often have common therapeutic properties. Potencies and AE profiles are seldom uniform. The decision to add or change the brand of medication within a class should be balanced with considerations for the indication, duplications, simplification, AEs, appropriate dosage, and drug-drug interactions.
Acknowledgments
Brent Wagner is funded by a US Department of Veterans Affairs Merit Award (I01 BX001958), a National Institutes of Health R01 grant (DK-102085), Dialysis Clinic, Inc., and partially supported by the University of New Mexico Brain and Behavioral Health Institute (BBHI 2018-1008, 2020-21-002) and in part by the University of New Mexico’s Signature Program in Cardiovascular and Metabolic Disease (CVMD); and the University of New Mexico School of Medicine Research Allocation Committee (C-2459-RAC, New Mexico Medical Trust). Brent Wagner is an Associate Member to the University of New Mexico Health Sciences Center Autophagy, Inflammation, and Metabolism Center of Biomedical Research Excellence (AIM CoBRE) supported by NIH grant P20GM121176.
Funding
National Institutes of Health Grant R01 DK-102085, Dialysis Clinic Inc., VA Merit Award I01 BX001958, Center for Integrated Nanotechnologies User Agreement 2019AU0120, Brain & Behavioral Health Institute (grants 2018-1008, 2020-21-002), University of New Mexico’s Signature Program in Cardiovascular and Metabolic Disease (CVMD), the University of New Mexico School of Medicine Research Allocation Committee (C-2459-RAC, New Mexico Medical Trust) and a metabolomics voucher from the AIM Center (NIH P20GM121176).
1. Wagner B, Kagan-Hallet KS, Russell IJ. Concomitant presentation of adermatopathic dermatomyositis, statin myopathy, fibromyalgia syndrome, piriformis muscle myofascial pain and diabetic neuropathy. J Musculoskeletal Pain. 2003;11(2):25-30. doi:10.1300/J094v11n02_05
2. Collins R, Reith C, Emberson J, et al. Interpretation of the evidence for the efficacy and safety of statin therapy [published correction appears in Lancet. 2017 Feb 11;389(10069):602]. Lancet. 2016;388(10059):2532-2561. doi:10.1016/S0140-6736(16)31357-5
3. Stroes ES, Thompson PD, Corsini A, et al. Statin-associated muscle symptoms: impact on statin therapy-European Atherosclerosis Society Consensus Panel Statement on Assessment, Aetiology and Management. Eur Heart J. 2015;36(17):1012-1022. doi:10.1093/eurheartj/ehv043
4. Saku K, Zhang B, Noda K; PATROL Trial Investigators. Randomized head-to-head comparison of pitavastatin, atorvastatin, and rosuvastatin for safety and efficacy (quantity and quality of LDL): the PATROL trial. Circ J. 2011;75(6):1493-1505. doi:10.1253/circj.cj-10-1281
5. Wald JJ. The effects of toxins on muscle. Neurol Clin. 2000;18(3):695-718. doi:10.1016/s0733-8619(05)70219-x
6. Hoffman KB, Kraus C, Dimbil M, Golomb BA. A survey of the FDA’s AERS database regarding muscle and tendon adverse events linked to the statin drug class. PLoS One. 2012;7(8):e42866. doi:10.1371/journal.pone.0042866
7. Sakaeda T, Kadoyama K, Okuno Y. Statin-associated muscular and renal adverse events: data mining of the public version of the FDA adverse event reporting system. PLoS One. 2011;6(12):e28124. doi:10.1371/journal.pone.0028124
8. Tiwari A. An overview of statin-associated proteinuria. Drug Discov Today. 2006;11(9-10):458-464. doi:10.1016/j.drudis.2006.03.017
9. Verhulst A, Sayer R, De Broe ME, D’Haese PC, Brown CD. Human proximal tubular epithelium actively secretes but does not retain rosuvastatin. Mol Pharmacol. 2008;74(4):1084-1091. doi:10.1124/mol.108.047647
10. Jones PH, Davidson MH, Stein EA, et al. Comparison of the efficacy and safety of rosuvastatin versus atorvastatin, simvastatin, and pravastatin across doses (STELLAR* Trial). Am J Cardiol. 2003;92(2):152-160. doi:10.1016/s0002-9149(03)00530-7
11. Wanner C, Krane V, März W, et al. Atorvastatin in patients with type 2 diabetes mellitus undergoing hemodialysis [published correction appears in N Engl J Med. 2005 Oct 13;353(15):1640]. N Engl J Med. 2005;353(3):238-248. doi:10.1056/NEJMoa043545
12. Fellström BC, Jardine AG, Schmieder RE, et al. Rosuvastatin and cardiovascular events in patients undergoing hemodialysis [published correction appears in N Engl J Med. 2010 Apr 15;362(15):1450]. N Engl J Med. 2009;360(14):1395-1407. doi:10.1056/NEJMoa0810177
13. Holdaas H, Holme I, Schmieder RE, et al. Rosuvastatin in diabetic hemodialysis patients. J Am Soc Nephrol. 2011;22(7):1335-1341. doi:10.1681/ASN.2010090987
14. Naci H, Brugts JJ, Fleurence R, Tsoi B, Toor H, Ades AE. Comparative benefits of statins in the primary and secondary prevention of major coronary events and all-cause mortality: a network meta-analysis of placebo-controlled and active-comparator trials. Eur J Prev Cardiol. 2013;20(4):641-657. doi:10.1177/2047487313480435
15. Alsheikh-Ali AA, Ambrose MS, Kuvin JT, Karas RH. The safety of rosuvastatin as used in common clinical practice: a postmarketing analysis. Circulation. 2005;111(23):3051-3057. doi:10.1161/CIRCULATIONAHA.105.555482
16. Ward NC, Watts GF, Eckel RH. Statin toxicity. Circ Res. 2019;124(2):328-350. doi:10.1161/CIRCRESAHA.118.312782
17. Selva-O’Callaghan A, Alvarado-Cardenas M, Pinal-Fernández I, et al. Statin-induced myalgia and myositis: an update on pathogenesis and clinical recommendations. Expert Rev Clin Immunol. 2018;14(3):215-224. doi:10.1080/1744666X.2018.1440206
18. Koslik HJ, Meskimen AH, Golomb BA. Physicians’ Experiences as patients with statin side effects: a case series. Drug Saf Case Rep. 2017;4(1):3. doi:10.1007/s40800-017-0045-0
1. Wagner B, Kagan-Hallet KS, Russell IJ. Concomitant presentation of adermatopathic dermatomyositis, statin myopathy, fibromyalgia syndrome, piriformis muscle myofascial pain and diabetic neuropathy. J Musculoskeletal Pain. 2003;11(2):25-30. doi:10.1300/J094v11n02_05
2. Collins R, Reith C, Emberson J, et al. Interpretation of the evidence for the efficacy and safety of statin therapy [published correction appears in Lancet. 2017 Feb 11;389(10069):602]. Lancet. 2016;388(10059):2532-2561. doi:10.1016/S0140-6736(16)31357-5
3. Stroes ES, Thompson PD, Corsini A, et al. Statin-associated muscle symptoms: impact on statin therapy-European Atherosclerosis Society Consensus Panel Statement on Assessment, Aetiology and Management. Eur Heart J. 2015;36(17):1012-1022. doi:10.1093/eurheartj/ehv043
4. Saku K, Zhang B, Noda K; PATROL Trial Investigators. Randomized head-to-head comparison of pitavastatin, atorvastatin, and rosuvastatin for safety and efficacy (quantity and quality of LDL): the PATROL trial. Circ J. 2011;75(6):1493-1505. doi:10.1253/circj.cj-10-1281
5. Wald JJ. The effects of toxins on muscle. Neurol Clin. 2000;18(3):695-718. doi:10.1016/s0733-8619(05)70219-x
6. Hoffman KB, Kraus C, Dimbil M, Golomb BA. A survey of the FDA’s AERS database regarding muscle and tendon adverse events linked to the statin drug class. PLoS One. 2012;7(8):e42866. doi:10.1371/journal.pone.0042866
7. Sakaeda T, Kadoyama K, Okuno Y. Statin-associated muscular and renal adverse events: data mining of the public version of the FDA adverse event reporting system. PLoS One. 2011;6(12):e28124. doi:10.1371/journal.pone.0028124
8. Tiwari A. An overview of statin-associated proteinuria. Drug Discov Today. 2006;11(9-10):458-464. doi:10.1016/j.drudis.2006.03.017
9. Verhulst A, Sayer R, De Broe ME, D’Haese PC, Brown CD. Human proximal tubular epithelium actively secretes but does not retain rosuvastatin. Mol Pharmacol. 2008;74(4):1084-1091. doi:10.1124/mol.108.047647
10. Jones PH, Davidson MH, Stein EA, et al. Comparison of the efficacy and safety of rosuvastatin versus atorvastatin, simvastatin, and pravastatin across doses (STELLAR* Trial). Am J Cardiol. 2003;92(2):152-160. doi:10.1016/s0002-9149(03)00530-7
11. Wanner C, Krane V, März W, et al. Atorvastatin in patients with type 2 diabetes mellitus undergoing hemodialysis [published correction appears in N Engl J Med. 2005 Oct 13;353(15):1640]. N Engl J Med. 2005;353(3):238-248. doi:10.1056/NEJMoa043545
12. Fellström BC, Jardine AG, Schmieder RE, et al. Rosuvastatin and cardiovascular events in patients undergoing hemodialysis [published correction appears in N Engl J Med. 2010 Apr 15;362(15):1450]. N Engl J Med. 2009;360(14):1395-1407. doi:10.1056/NEJMoa0810177
13. Holdaas H, Holme I, Schmieder RE, et al. Rosuvastatin in diabetic hemodialysis patients. J Am Soc Nephrol. 2011;22(7):1335-1341. doi:10.1681/ASN.2010090987
14. Naci H, Brugts JJ, Fleurence R, Tsoi B, Toor H, Ades AE. Comparative benefits of statins in the primary and secondary prevention of major coronary events and all-cause mortality: a network meta-analysis of placebo-controlled and active-comparator trials. Eur J Prev Cardiol. 2013;20(4):641-657. doi:10.1177/2047487313480435
15. Alsheikh-Ali AA, Ambrose MS, Kuvin JT, Karas RH. The safety of rosuvastatin as used in common clinical practice: a postmarketing analysis. Circulation. 2005;111(23):3051-3057. doi:10.1161/CIRCULATIONAHA.105.555482
16. Ward NC, Watts GF, Eckel RH. Statin toxicity. Circ Res. 2019;124(2):328-350. doi:10.1161/CIRCRESAHA.118.312782
17. Selva-O’Callaghan A, Alvarado-Cardenas M, Pinal-Fernández I, et al. Statin-induced myalgia and myositis: an update on pathogenesis and clinical recommendations. Expert Rev Clin Immunol. 2018;14(3):215-224. doi:10.1080/1744666X.2018.1440206
18. Koslik HJ, Meskimen AH, Golomb BA. Physicians’ Experiences as patients with statin side effects: a case series. Drug Saf Case Rep. 2017;4(1):3. doi:10.1007/s40800-017-0045-0
Liraglutide effective against weight regain after gastric bypass
The glucagon-like peptide-1 (GLP-1) receptor agonist liraglutide (Saxenda, Novo Nordisk) was safe and effective for treating weight regain after Roux-en-Y gastric bypass (RYGB), in a randomized controlled trial.
That is, 132 patients who had lost at least 25% of their initial weight after RYGB and then gained at least 10% back were randomized 2:1 to receive liraglutide plus frequent lifestyle advice from a registered dietitian or lifestyle advice alone.
After a year, 69%, 48%, and 24% of patients who had received liraglutide lost at least 5%, 10%, and 15% of their study entry weight, respectively. In contrast, only 5% of patients in the control group lost at least 5% of their weight and none lost at least 10% of their weight.
“Liraglutide 3.0 mg/day, with lifestyle modification, was significantly more effective than placebo in treating weight regain after RYGB without increased risk of serious adverse events,” Holly F. Lofton, MD, summarized this week in an oral session at ObesityWeek®, the annual meeting of The Obesity Society.
Dr. Lofton, a clinical associate professor of surgery and medicine, and director, weight management program, NYU, Langone Health, explained to this news organization that she initiated the study after attending a “packed” session about post bariatric surgery weight regain at a prior American Society of Metabolic and Bariatric Surgery conference.
“The lecturers recommended conservative measures (such as reiterating the diet recommendations, exercise, [and] counseling), and revisional surgeries,” she said in an email, but at the time “there was no literature that provided direction on which pharmacotherapies are best for this population.”
It was known that decreases in endogenous GLP-1 levels coincide with weight regain, and liraglutide (Saxenda) was the only GLP-1 agonist approved for chronic weight management at the time, so she devised the current study protocol.
The findings are especially helpful for patients who are not candidates for bariatric surgery revisions, she noted. Further research is needed to investigate the effect of newer GLP-1 agonists, such as semaglutide (Wegovy), on weight regain following different types of bariatric surgery.
Asked to comment, Wendy C. King, PhD, who was not involved with this research, said that more than two-thirds of patients treated with 3 mg/day subcutaneous liraglutide injections in the current study lost at least 5% of their initial weight a year later, and 20% of them attained a weight as low as, or lower than, their lowest weight after bariatric surgery (nadir weight).
“The fact that both groups received lifestyle counseling from registered dietitians for just over a year, but only patients in the liraglutide group lost weight, on average, speaks to the difficulty of losing weight following weight regain post–bariatric surgery,” added Dr. King, an associate professor of epidemiology at the University of Pittsburgh, Pennsylvania.
This study “provides data that may help clinicians and patients understand the potential effect of adding liraglutide 3.0 mg/day to their weight loss efforts,” she told this news organization in an email.
However, “given that 42% of those on liraglutide reported gastrointestinal-related side effects, patients should also be counseled on this potential outcome and given suggestions for how to minimize such side effects,” Dr. King suggested.
Weight regain common, repeat surgery entails risk
Weight regain is common even years after bariatric surgery. Repeat surgery entails some risk, and lifestyle approaches alone are rarely successful in reversing weight regain, Dr. Lofton told the audience.
The researchers enrolled 132 adults who had a mean weight of 134 kg (295 pounds) when they underwent RYGB, and who lost at least 25% of their initial weight (mean weight loss of 38%) after the surgery, but who also regained at least 10% of their initial weight.
At enrollment of the current study (baseline), the patients had had RYGB 18 months to 10 years earlier (mean 5.7 years earlier) and now had a mean weight of 99 kg (218 pounds) and a mean BMI of 35.6 kg/m2. None of the patients had diabetes.
The patients were randomized to receive liraglutide (n = 89, 84% women) or placebo (n = 43, 88% women) for 56 weeks.
They were a mean age of 48 years, and about 59% were White and 25% were Black.
All patients had clinic visits every 3 months where they received lifestyle counseling from a registered dietitian.
At 12 months, patients in the liraglutide group had lost a mean of 8.8% of their baseline weight, whereas those in the placebo group had gained a mean of 1.48% of their baseline weight.
There were no significant between-group differences in cardiometabolic variables.
None of the patients in the control group attained a weight that was as low as their nadir weight after RYGB.
The rates of nausea (25%), constipation (16%), and abdominal pain (10%) in the liraglutide group were higher than in the placebo group (7%, 14%, and 5%, respectively) but similar to rates of gastrointestinal side effects in other trials of this agent.
Dr. Lofton has disclosed receiving consulting fees and being on a speaker bureau for Novo Nordisk and receiving research funds from Boehringer Ingelheim, Eli Lilly, and Novo Nordisk. Dr. King has reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The glucagon-like peptide-1 (GLP-1) receptor agonist liraglutide (Saxenda, Novo Nordisk) was safe and effective for treating weight regain after Roux-en-Y gastric bypass (RYGB), in a randomized controlled trial.
That is, 132 patients who had lost at least 25% of their initial weight after RYGB and then gained at least 10% back were randomized 2:1 to receive liraglutide plus frequent lifestyle advice from a registered dietitian or lifestyle advice alone.
After a year, 69%, 48%, and 24% of patients who had received liraglutide lost at least 5%, 10%, and 15% of their study entry weight, respectively. In contrast, only 5% of patients in the control group lost at least 5% of their weight and none lost at least 10% of their weight.
“Liraglutide 3.0 mg/day, with lifestyle modification, was significantly more effective than placebo in treating weight regain after RYGB without increased risk of serious adverse events,” Holly F. Lofton, MD, summarized this week in an oral session at ObesityWeek®, the annual meeting of The Obesity Society.
Dr. Lofton, a clinical associate professor of surgery and medicine, and director, weight management program, NYU, Langone Health, explained to this news organization that she initiated the study after attending a “packed” session about post bariatric surgery weight regain at a prior American Society of Metabolic and Bariatric Surgery conference.
“The lecturers recommended conservative measures (such as reiterating the diet recommendations, exercise, [and] counseling), and revisional surgeries,” she said in an email, but at the time “there was no literature that provided direction on which pharmacotherapies are best for this population.”
It was known that decreases in endogenous GLP-1 levels coincide with weight regain, and liraglutide (Saxenda) was the only GLP-1 agonist approved for chronic weight management at the time, so she devised the current study protocol.
The findings are especially helpful for patients who are not candidates for bariatric surgery revisions, she noted. Further research is needed to investigate the effect of newer GLP-1 agonists, such as semaglutide (Wegovy), on weight regain following different types of bariatric surgery.
Asked to comment, Wendy C. King, PhD, who was not involved with this research, said that more than two-thirds of patients treated with 3 mg/day subcutaneous liraglutide injections in the current study lost at least 5% of their initial weight a year later, and 20% of them attained a weight as low as, or lower than, their lowest weight after bariatric surgery (nadir weight).
“The fact that both groups received lifestyle counseling from registered dietitians for just over a year, but only patients in the liraglutide group lost weight, on average, speaks to the difficulty of losing weight following weight regain post–bariatric surgery,” added Dr. King, an associate professor of epidemiology at the University of Pittsburgh, Pennsylvania.
This study “provides data that may help clinicians and patients understand the potential effect of adding liraglutide 3.0 mg/day to their weight loss efforts,” she told this news organization in an email.
However, “given that 42% of those on liraglutide reported gastrointestinal-related side effects, patients should also be counseled on this potential outcome and given suggestions for how to minimize such side effects,” Dr. King suggested.
Weight regain common, repeat surgery entails risk
Weight regain is common even years after bariatric surgery. Repeat surgery entails some risk, and lifestyle approaches alone are rarely successful in reversing weight regain, Dr. Lofton told the audience.
The researchers enrolled 132 adults who had a mean weight of 134 kg (295 pounds) when they underwent RYGB, and who lost at least 25% of their initial weight (mean weight loss of 38%) after the surgery, but who also regained at least 10% of their initial weight.
At enrollment of the current study (baseline), the patients had had RYGB 18 months to 10 years earlier (mean 5.7 years earlier) and now had a mean weight of 99 kg (218 pounds) and a mean BMI of 35.6 kg/m2. None of the patients had diabetes.
The patients were randomized to receive liraglutide (n = 89, 84% women) or placebo (n = 43, 88% women) for 56 weeks.
They were a mean age of 48 years, and about 59% were White and 25% were Black.
All patients had clinic visits every 3 months where they received lifestyle counseling from a registered dietitian.
At 12 months, patients in the liraglutide group had lost a mean of 8.8% of their baseline weight, whereas those in the placebo group had gained a mean of 1.48% of their baseline weight.
There were no significant between-group differences in cardiometabolic variables.
None of the patients in the control group attained a weight that was as low as their nadir weight after RYGB.
The rates of nausea (25%), constipation (16%), and abdominal pain (10%) in the liraglutide group were higher than in the placebo group (7%, 14%, and 5%, respectively) but similar to rates of gastrointestinal side effects in other trials of this agent.
Dr. Lofton has disclosed receiving consulting fees and being on a speaker bureau for Novo Nordisk and receiving research funds from Boehringer Ingelheim, Eli Lilly, and Novo Nordisk. Dr. King has reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The glucagon-like peptide-1 (GLP-1) receptor agonist liraglutide (Saxenda, Novo Nordisk) was safe and effective for treating weight regain after Roux-en-Y gastric bypass (RYGB), in a randomized controlled trial.
That is, 132 patients who had lost at least 25% of their initial weight after RYGB and then gained at least 10% back were randomized 2:1 to receive liraglutide plus frequent lifestyle advice from a registered dietitian or lifestyle advice alone.
After a year, 69%, 48%, and 24% of patients who had received liraglutide lost at least 5%, 10%, and 15% of their study entry weight, respectively. In contrast, only 5% of patients in the control group lost at least 5% of their weight and none lost at least 10% of their weight.
“Liraglutide 3.0 mg/day, with lifestyle modification, was significantly more effective than placebo in treating weight regain after RYGB without increased risk of serious adverse events,” Holly F. Lofton, MD, summarized this week in an oral session at ObesityWeek®, the annual meeting of The Obesity Society.
Dr. Lofton, a clinical associate professor of surgery and medicine, and director, weight management program, NYU, Langone Health, explained to this news organization that she initiated the study after attending a “packed” session about post bariatric surgery weight regain at a prior American Society of Metabolic and Bariatric Surgery conference.
“The lecturers recommended conservative measures (such as reiterating the diet recommendations, exercise, [and] counseling), and revisional surgeries,” she said in an email, but at the time “there was no literature that provided direction on which pharmacotherapies are best for this population.”
It was known that decreases in endogenous GLP-1 levels coincide with weight regain, and liraglutide (Saxenda) was the only GLP-1 agonist approved for chronic weight management at the time, so she devised the current study protocol.
The findings are especially helpful for patients who are not candidates for bariatric surgery revisions, she noted. Further research is needed to investigate the effect of newer GLP-1 agonists, such as semaglutide (Wegovy), on weight regain following different types of bariatric surgery.
Asked to comment, Wendy C. King, PhD, who was not involved with this research, said that more than two-thirds of patients treated with 3 mg/day subcutaneous liraglutide injections in the current study lost at least 5% of their initial weight a year later, and 20% of them attained a weight as low as, or lower than, their lowest weight after bariatric surgery (nadir weight).
“The fact that both groups received lifestyle counseling from registered dietitians for just over a year, but only patients in the liraglutide group lost weight, on average, speaks to the difficulty of losing weight following weight regain post–bariatric surgery,” added Dr. King, an associate professor of epidemiology at the University of Pittsburgh, Pennsylvania.
This study “provides data that may help clinicians and patients understand the potential effect of adding liraglutide 3.0 mg/day to their weight loss efforts,” she told this news organization in an email.
However, “given that 42% of those on liraglutide reported gastrointestinal-related side effects, patients should also be counseled on this potential outcome and given suggestions for how to minimize such side effects,” Dr. King suggested.
Weight regain common, repeat surgery entails risk
Weight regain is common even years after bariatric surgery. Repeat surgery entails some risk, and lifestyle approaches alone are rarely successful in reversing weight regain, Dr. Lofton told the audience.
The researchers enrolled 132 adults who had a mean weight of 134 kg (295 pounds) when they underwent RYGB, and who lost at least 25% of their initial weight (mean weight loss of 38%) after the surgery, but who also regained at least 10% of their initial weight.
At enrollment of the current study (baseline), the patients had had RYGB 18 months to 10 years earlier (mean 5.7 years earlier) and now had a mean weight of 99 kg (218 pounds) and a mean BMI of 35.6 kg/m2. None of the patients had diabetes.
The patients were randomized to receive liraglutide (n = 89, 84% women) or placebo (n = 43, 88% women) for 56 weeks.
They were a mean age of 48 years, and about 59% were White and 25% were Black.
All patients had clinic visits every 3 months where they received lifestyle counseling from a registered dietitian.
At 12 months, patients in the liraglutide group had lost a mean of 8.8% of their baseline weight, whereas those in the placebo group had gained a mean of 1.48% of their baseline weight.
There were no significant between-group differences in cardiometabolic variables.
None of the patients in the control group attained a weight that was as low as their nadir weight after RYGB.
The rates of nausea (25%), constipation (16%), and abdominal pain (10%) in the liraglutide group were higher than in the placebo group (7%, 14%, and 5%, respectively) but similar to rates of gastrointestinal side effects in other trials of this agent.
Dr. Lofton has disclosed receiving consulting fees and being on a speaker bureau for Novo Nordisk and receiving research funds from Boehringer Ingelheim, Eli Lilly, and Novo Nordisk. Dr. King has reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM OBESITY WEEK 2021
Statins’ effects on CVD outweigh risk for diabetes in RA
The use of statins by patients with rheumatoid arthritis appears to provide an overall net benefit on cardiovascular disease outcomes that outweighs the risk of type 2 diabetes mellitus (T2DM) seen with the drugs in the general population, according to evidence from a cohort study of more than 16,000 people in the United Kingdom that was presented at the virtual annual meeting of the American College of Rheumatology.
“Our study emphasizes that RA patients should be assessed for statin initiation to improve CVD risk,” lead study author Gulsen Ozen, MD, a third-year resident at the University of Nebraska, Omaha, said in an interview. Because the risk of T2DM with statin use is no worse in patients with RA than in the general population, statin initiation “is actually a great opportunity to address the risk factors for T2DM such as activity and exercise, obesity and weight loss, and [use of glucocorticoids], which have other important health effects,” she said.
“Also, importantly, even if [patients] develop T2DM, statins still work on CVD and mortality outcomes as in patients without diabetes,” Dr. Ozen added. “Given all, the benefits of statins way outweigh the hazards.”
Dr. Ozen said this was the first large cohort study to evaluate CVD mortality and T2DM risks with statins in patients with RA, a claim with which rheumatologist Elena Myasoedova, MD, PhD, of the Mayo Clinic in Rochester, Minn., concurred.
Dr. Myasoedova, professor of rheumatology and epidemiology at Mayo, said in an interview that the study was “methodologically rigorous” using time-conditional propensity score (TCPS) matching and a prevalent new-user design, “thus addressing the immortal time bias” found in the design of studies in which patients enter a cohort but do not start a treatment before developing the outcome of interest and are assigned to the untreated group or when the period of delay from when patients enter the cohort to when they are treated is excluded from the analysis. An earlier study from the same authors did not use TCPS matching, she said.
“The study findings suggest that patients with RA can benefit from statin use in terms of CVD outcomes and mortality but physicians should use vigilance regarding increased T2DM risk and discuss this possibility with patients,” Dr. Myasoedova said. “Identifying patients who are at higher risk of developing T2DM after statin initiation would be important to personalize the approach to statin therapy.”
Study details
The study accessed records from the U.K. Clinical Practice Research Datalink and linked Hospital Episode Statistics and Office of National Statistics databases. It analyzed adult patients with RA who were diagnosed during 1989-2018 in two cohorts: One for CVD and all-cause mortality, consisting of 1,768 statin initiators and 3,528 TCPS-matched nonusers; and a T2DM cohort with 3,608 statin initiators and 7,208 TCPS-matched nonusers.
In the entire cohort, statin use was associated with a 32% reduction in CV events (composite endpoint of the nonfatal or fatal MI, stroke, hospitalized heart failure, or CVD mortality), a 54% reduction in all-cause mortality, and a 33% increase in risk for T2DM, Dr. Ozen said. Results were similar in both sexes, although CV event reduction with statins in men did not reach statistical significance, likely because of a smaller sample size, she said.
Patients with and without a history of CVD had a similar reduction in CV events and all-cause mortality, and risk for T2DM increased with statins, but the latter reached statistical significance only in patients without a history of CVD, Dr. Ozen said.
Patients with RA who are at risk for T2DM and who are taking statins require blood glucose monitoring, which is typically done in patients with RA on disease-modifying antirheumatic drugs, and hemoglobin A1c testing when glucose levels are impaired, she said. “Any concerns for T2DM would be also communicated by the primary care providers of the patients to initiate further assessment and management,” she said.
But Dr. Ozen noted that confusion exists among primary care physicians and rheumatologists about who’s responsible for prescribing statins in these patients. “I would like to remind you that instead of assigning this role to a certain specialty, just good communication could improve this care gap of statin underutilization in RA,” she said. “Also, for rheumatologists, given that all-cause mortality reduction with statins was as high as CV event reduction, statins may be reducing other causes of mortality through improving disease activity.”
Bristol-Myers Squibb provided funding for the study. Dr. Ozen and Dr. Myasoedova have no relevant disclosures.
The use of statins by patients with rheumatoid arthritis appears to provide an overall net benefit on cardiovascular disease outcomes that outweighs the risk of type 2 diabetes mellitus (T2DM) seen with the drugs in the general population, according to evidence from a cohort study of more than 16,000 people in the United Kingdom that was presented at the virtual annual meeting of the American College of Rheumatology.
“Our study emphasizes that RA patients should be assessed for statin initiation to improve CVD risk,” lead study author Gulsen Ozen, MD, a third-year resident at the University of Nebraska, Omaha, said in an interview. Because the risk of T2DM with statin use is no worse in patients with RA than in the general population, statin initiation “is actually a great opportunity to address the risk factors for T2DM such as activity and exercise, obesity and weight loss, and [use of glucocorticoids], which have other important health effects,” she said.
“Also, importantly, even if [patients] develop T2DM, statins still work on CVD and mortality outcomes as in patients without diabetes,” Dr. Ozen added. “Given all, the benefits of statins way outweigh the hazards.”
Dr. Ozen said this was the first large cohort study to evaluate CVD mortality and T2DM risks with statins in patients with RA, a claim with which rheumatologist Elena Myasoedova, MD, PhD, of the Mayo Clinic in Rochester, Minn., concurred.
Dr. Myasoedova, professor of rheumatology and epidemiology at Mayo, said in an interview that the study was “methodologically rigorous” using time-conditional propensity score (TCPS) matching and a prevalent new-user design, “thus addressing the immortal time bias” found in the design of studies in which patients enter a cohort but do not start a treatment before developing the outcome of interest and are assigned to the untreated group or when the period of delay from when patients enter the cohort to when they are treated is excluded from the analysis. An earlier study from the same authors did not use TCPS matching, she said.
“The study findings suggest that patients with RA can benefit from statin use in terms of CVD outcomes and mortality but physicians should use vigilance regarding increased T2DM risk and discuss this possibility with patients,” Dr. Myasoedova said. “Identifying patients who are at higher risk of developing T2DM after statin initiation would be important to personalize the approach to statin therapy.”
Study details
The study accessed records from the U.K. Clinical Practice Research Datalink and linked Hospital Episode Statistics and Office of National Statistics databases. It analyzed adult patients with RA who were diagnosed during 1989-2018 in two cohorts: One for CVD and all-cause mortality, consisting of 1,768 statin initiators and 3,528 TCPS-matched nonusers; and a T2DM cohort with 3,608 statin initiators and 7,208 TCPS-matched nonusers.
In the entire cohort, statin use was associated with a 32% reduction in CV events (composite endpoint of the nonfatal or fatal MI, stroke, hospitalized heart failure, or CVD mortality), a 54% reduction in all-cause mortality, and a 33% increase in risk for T2DM, Dr. Ozen said. Results were similar in both sexes, although CV event reduction with statins in men did not reach statistical significance, likely because of a smaller sample size, she said.
Patients with and without a history of CVD had a similar reduction in CV events and all-cause mortality, and risk for T2DM increased with statins, but the latter reached statistical significance only in patients without a history of CVD, Dr. Ozen said.
Patients with RA who are at risk for T2DM and who are taking statins require blood glucose monitoring, which is typically done in patients with RA on disease-modifying antirheumatic drugs, and hemoglobin A1c testing when glucose levels are impaired, she said. “Any concerns for T2DM would be also communicated by the primary care providers of the patients to initiate further assessment and management,” she said.
But Dr. Ozen noted that confusion exists among primary care physicians and rheumatologists about who’s responsible for prescribing statins in these patients. “I would like to remind you that instead of assigning this role to a certain specialty, just good communication could improve this care gap of statin underutilization in RA,” she said. “Also, for rheumatologists, given that all-cause mortality reduction with statins was as high as CV event reduction, statins may be reducing other causes of mortality through improving disease activity.”
Bristol-Myers Squibb provided funding for the study. Dr. Ozen and Dr. Myasoedova have no relevant disclosures.
The use of statins by patients with rheumatoid arthritis appears to provide an overall net benefit on cardiovascular disease outcomes that outweighs the risk of type 2 diabetes mellitus (T2DM) seen with the drugs in the general population, according to evidence from a cohort study of more than 16,000 people in the United Kingdom that was presented at the virtual annual meeting of the American College of Rheumatology.
“Our study emphasizes that RA patients should be assessed for statin initiation to improve CVD risk,” lead study author Gulsen Ozen, MD, a third-year resident at the University of Nebraska, Omaha, said in an interview. Because the risk of T2DM with statin use is no worse in patients with RA than in the general population, statin initiation “is actually a great opportunity to address the risk factors for T2DM such as activity and exercise, obesity and weight loss, and [use of glucocorticoids], which have other important health effects,” she said.
“Also, importantly, even if [patients] develop T2DM, statins still work on CVD and mortality outcomes as in patients without diabetes,” Dr. Ozen added. “Given all, the benefits of statins way outweigh the hazards.”
Dr. Ozen said this was the first large cohort study to evaluate CVD mortality and T2DM risks with statins in patients with RA, a claim with which rheumatologist Elena Myasoedova, MD, PhD, of the Mayo Clinic in Rochester, Minn., concurred.
Dr. Myasoedova, professor of rheumatology and epidemiology at Mayo, said in an interview that the study was “methodologically rigorous” using time-conditional propensity score (TCPS) matching and a prevalent new-user design, “thus addressing the immortal time bias” found in the design of studies in which patients enter a cohort but do not start a treatment before developing the outcome of interest and are assigned to the untreated group or when the period of delay from when patients enter the cohort to when they are treated is excluded from the analysis. An earlier study from the same authors did not use TCPS matching, she said.
“The study findings suggest that patients with RA can benefit from statin use in terms of CVD outcomes and mortality but physicians should use vigilance regarding increased T2DM risk and discuss this possibility with patients,” Dr. Myasoedova said. “Identifying patients who are at higher risk of developing T2DM after statin initiation would be important to personalize the approach to statin therapy.”
Study details
The study accessed records from the U.K. Clinical Practice Research Datalink and linked Hospital Episode Statistics and Office of National Statistics databases. It analyzed adult patients with RA who were diagnosed during 1989-2018 in two cohorts: One for CVD and all-cause mortality, consisting of 1,768 statin initiators and 3,528 TCPS-matched nonusers; and a T2DM cohort with 3,608 statin initiators and 7,208 TCPS-matched nonusers.
In the entire cohort, statin use was associated with a 32% reduction in CV events (composite endpoint of the nonfatal or fatal MI, stroke, hospitalized heart failure, or CVD mortality), a 54% reduction in all-cause mortality, and a 33% increase in risk for T2DM, Dr. Ozen said. Results were similar in both sexes, although CV event reduction with statins in men did not reach statistical significance, likely because of a smaller sample size, she said.
Patients with and without a history of CVD had a similar reduction in CV events and all-cause mortality, and risk for T2DM increased with statins, but the latter reached statistical significance only in patients without a history of CVD, Dr. Ozen said.
Patients with RA who are at risk for T2DM and who are taking statins require blood glucose monitoring, which is typically done in patients with RA on disease-modifying antirheumatic drugs, and hemoglobin A1c testing when glucose levels are impaired, she said. “Any concerns for T2DM would be also communicated by the primary care providers of the patients to initiate further assessment and management,” she said.
But Dr. Ozen noted that confusion exists among primary care physicians and rheumatologists about who’s responsible for prescribing statins in these patients. “I would like to remind you that instead of assigning this role to a certain specialty, just good communication could improve this care gap of statin underutilization in RA,” she said. “Also, for rheumatologists, given that all-cause mortality reduction with statins was as high as CV event reduction, statins may be reducing other causes of mortality through improving disease activity.”
Bristol-Myers Squibb provided funding for the study. Dr. Ozen and Dr. Myasoedova have no relevant disclosures.
FROM ACR 2021
‘If obesity were diabetes or cancer, how would you approach it?’
“When considering the challenges of obesity, ask yourself: ‘If it were diabetes, cancer, HIV, or Alzheimer’s, how would you discuss it, approach it, assess it, treat it?’” Lee M. Kaplan, MD, PhD, asked the audience of health care professionals during ObesityWeek®, the annual meeting of The Obesity Society.
“And then do it for obesity, using the full spectrum of tools at our disposal,” he advised.
This was the takeaway that Dr. Kaplan, director of the Obesity, Metabolism, and Nutrition Institute at Massachusetts General Hospital and associate professor, Harvard Medical School, Boston, left the audience with at the end of his lecture entitled, “What does the future of obesity care look like?”
Invited to summarize his main points, Dr. Kaplan told this news organization in an interview that practitioners caring for patients with obesity need to first “recognize that obesity is a disease” caused by dysfunction of the metabolic system that regulates body fat – in the same way immune dysregulation can lead to asthma.
Second, “we are finally developing noninvasive therapies that are more effective,” he noted, referring to the recently approved semaglutide, and even more potent weight-loss therapies that could be on the market within 3 years, so that weight-loss outcomes with pharmacotherapy are approaching those with bariatric surgery.
Third, it is important that patients with obesity get “broad and equitable access” to treatment, and health care practitioners need to be on the same page and have a “shared understanding” of which treatments are appropriate for individual patients, “just as we do for other diseases.”
Need for a shared understanding
“Dr. Kaplan really brought home the idea that we all need a shared understanding of what obesity is – and what it is not,” agreed symposium moderator Donna H. Ryan, MD, in an email.
“He underscored the biologic basis of obesity,” noted Dr. Ryan, professor emerita at Pennington Biomedical Research Center in Baton Rouge, Louisiana, and associate editor-in-chief of Obesity, the official journal of The Obesity Society.
“It is a dysregulation of the body’s weight (especially adipose tissue) regulatory system,” she continued. “The body responds to powerful environmental pressures that produce excess energy balance, and we store that as fat and defend our highest fat mass. This makes obesity a disease, a chronic disease that requires a medical approach to reverse. It’s not a cosmetic problem, it’s a medical problem,” she emphasized.
There is so much misinformation out there about obesity, according to Dr. Ryan.
“People think it’s a lack of willpower, and even patients blame themselves for not being able to lose weight and keep it off. It’s not their fault! It’s biology.”
Although the supplement industry and fad diets falsely promise fast results, there is no magic diet, she continued.
“But we have made progress based on understanding the biologic basis of obesity and have new medications that offer real hope for patients.”
“With 42% of U.S. adults having a BMI that qualifies as obesity, we need a concerted and broad effort to address this problem, and that starts with everybody on the same page as to what obesity is ... a shared understanding of the biologic basis of obesity. It’s time to take obesity seriously,” she summarized, echoing Dr. Kaplan.
A question of biology
“Obesity results from inappropriate pathophysiological regulation of body fat mass,” when the body defends adiposity, Dr. Kaplan explained at the start of his lecture.
The treatment strategy for obesity has always been a stepwise approach starting with lifestyle changes, then pharmacotherapy, then possibly bariatric surgery – each step with a potentially greater chance of weight loss. But now, he explained, medicine is on the verge of having an armamentarium of more potent weight-loss medications.
Compared with phentermine/topiramate, orlistat, naltrexone/bupropion, and liraglutide – which roughly might provide 5% to 10% weight loss, the glucagon-like peptide-1 (GLP-1) agonist semaglutide 2.4 mg/week (Wegovy, Novo Nordisk), approved by the U.S. Food and Drug Association in June, provides almost double this potential weight loss.
And two new agents that could provide “never seen before weight loss” of 25% could potentially enter the marketplace by 2025: the amylin agonist cagrilintide (Novo Nordisk) and the twincretin tirzepatide (Eli Lilly) (a combined glucose-dependent insulinotropic polypeptide [GIP] and GLP-1 receptor agonist).
In addition, when liraglutide comes off patent, a generic version could potentially be introduced, and combined generic liraglutide plus generic phentermine/topiramate could be a less expensive weight-loss treatment option in the future, he noted.
One size does not fit all
Importantly, weight loss varies widely among individual patients.
A graph of potential weight loss with different treatments (for example, bariatric surgery or liraglutide) versus the percentage of patients that attain the weight losses is roughly bell-shaped, Dr. Kaplan explained. For example, in the STEP1 trial of semaglutide, roughly 7.1% of patients lost less than 5% of their initial weight, 25% of patients lost 20% to 30%, and 10.8% of patients lost 30% or more; that is, patients at the higher end had weight loss comparable to that seen with bariatric surgery
Adding pharmacotherapy after bariatric surgery could be synergistic. For example, in the GRAVITAS study of patients with type 2 diabetes who had gastric bypass surgery, those who received liraglutide after surgery had augmented weight loss compared with those who received placebo.
People at a cocktail party might come up to him and say, “I’d like to lose 5 pounds, 10 pounds,” Dr. Kaplan related in the Q&A session.
“That’s not obesity,” he emphasized. Obesity is excess body fat that poses a risk to health. A person with obesity may have 50 or more excess pounds, and the body is trying to defend this weight.
“If we want to treat obesity more effectively, we have to fully understand why it is a disease and how that disease differs from the cultural desire for thinness,” he reiterated.
“We have to keep the needs and goals of all people living with obesity foremost in our minds, even if many of them have been previously misled by the bias, stigma, blame, and discrimination that surrounds them.”
“We need to re-evaluate what we think we know about obesity and open our minds to new ideas,” he added.
Dr. Kaplan has reported financial ties to Eli Lilly, Gelesis, GI Dynamics, IntelliHealth, Johnson & Johnson, Novo Nordisk, Pfizer, and Rhythm Pharmaceuticals. Dr. Ryan has ties to numerous Novo Nordisk, Pfizer, and several other pharmaceutical companies, including having an ownership interest in Gila Therapeutics, Xeno Biosciences, Epitomee, Calibrate, Roman, and Scientific Intake.
A version of this article first appeared on Medscape.com.
“When considering the challenges of obesity, ask yourself: ‘If it were diabetes, cancer, HIV, or Alzheimer’s, how would you discuss it, approach it, assess it, treat it?’” Lee M. Kaplan, MD, PhD, asked the audience of health care professionals during ObesityWeek®, the annual meeting of The Obesity Society.
“And then do it for obesity, using the full spectrum of tools at our disposal,” he advised.
This was the takeaway that Dr. Kaplan, director of the Obesity, Metabolism, and Nutrition Institute at Massachusetts General Hospital and associate professor, Harvard Medical School, Boston, left the audience with at the end of his lecture entitled, “What does the future of obesity care look like?”
Invited to summarize his main points, Dr. Kaplan told this news organization in an interview that practitioners caring for patients with obesity need to first “recognize that obesity is a disease” caused by dysfunction of the metabolic system that regulates body fat – in the same way immune dysregulation can lead to asthma.
Second, “we are finally developing noninvasive therapies that are more effective,” he noted, referring to the recently approved semaglutide, and even more potent weight-loss therapies that could be on the market within 3 years, so that weight-loss outcomes with pharmacotherapy are approaching those with bariatric surgery.
Third, it is important that patients with obesity get “broad and equitable access” to treatment, and health care practitioners need to be on the same page and have a “shared understanding” of which treatments are appropriate for individual patients, “just as we do for other diseases.”
Need for a shared understanding
“Dr. Kaplan really brought home the idea that we all need a shared understanding of what obesity is – and what it is not,” agreed symposium moderator Donna H. Ryan, MD, in an email.
“He underscored the biologic basis of obesity,” noted Dr. Ryan, professor emerita at Pennington Biomedical Research Center in Baton Rouge, Louisiana, and associate editor-in-chief of Obesity, the official journal of The Obesity Society.
“It is a dysregulation of the body’s weight (especially adipose tissue) regulatory system,” she continued. “The body responds to powerful environmental pressures that produce excess energy balance, and we store that as fat and defend our highest fat mass. This makes obesity a disease, a chronic disease that requires a medical approach to reverse. It’s not a cosmetic problem, it’s a medical problem,” she emphasized.
There is so much misinformation out there about obesity, according to Dr. Ryan.
“People think it’s a lack of willpower, and even patients blame themselves for not being able to lose weight and keep it off. It’s not their fault! It’s biology.”
Although the supplement industry and fad diets falsely promise fast results, there is no magic diet, she continued.
“But we have made progress based on understanding the biologic basis of obesity and have new medications that offer real hope for patients.”
“With 42% of U.S. adults having a BMI that qualifies as obesity, we need a concerted and broad effort to address this problem, and that starts with everybody on the same page as to what obesity is ... a shared understanding of the biologic basis of obesity. It’s time to take obesity seriously,” she summarized, echoing Dr. Kaplan.
A question of biology
“Obesity results from inappropriate pathophysiological regulation of body fat mass,” when the body defends adiposity, Dr. Kaplan explained at the start of his lecture.
The treatment strategy for obesity has always been a stepwise approach starting with lifestyle changes, then pharmacotherapy, then possibly bariatric surgery – each step with a potentially greater chance of weight loss. But now, he explained, medicine is on the verge of having an armamentarium of more potent weight-loss medications.
Compared with phentermine/topiramate, orlistat, naltrexone/bupropion, and liraglutide – which roughly might provide 5% to 10% weight loss, the glucagon-like peptide-1 (GLP-1) agonist semaglutide 2.4 mg/week (Wegovy, Novo Nordisk), approved by the U.S. Food and Drug Association in June, provides almost double this potential weight loss.
And two new agents that could provide “never seen before weight loss” of 25% could potentially enter the marketplace by 2025: the amylin agonist cagrilintide (Novo Nordisk) and the twincretin tirzepatide (Eli Lilly) (a combined glucose-dependent insulinotropic polypeptide [GIP] and GLP-1 receptor agonist).
In addition, when liraglutide comes off patent, a generic version could potentially be introduced, and combined generic liraglutide plus generic phentermine/topiramate could be a less expensive weight-loss treatment option in the future, he noted.
One size does not fit all
Importantly, weight loss varies widely among individual patients.
A graph of potential weight loss with different treatments (for example, bariatric surgery or liraglutide) versus the percentage of patients that attain the weight losses is roughly bell-shaped, Dr. Kaplan explained. For example, in the STEP1 trial of semaglutide, roughly 7.1% of patients lost less than 5% of their initial weight, 25% of patients lost 20% to 30%, and 10.8% of patients lost 30% or more; that is, patients at the higher end had weight loss comparable to that seen with bariatric surgery
Adding pharmacotherapy after bariatric surgery could be synergistic. For example, in the GRAVITAS study of patients with type 2 diabetes who had gastric bypass surgery, those who received liraglutide after surgery had augmented weight loss compared with those who received placebo.
People at a cocktail party might come up to him and say, “I’d like to lose 5 pounds, 10 pounds,” Dr. Kaplan related in the Q&A session.
“That’s not obesity,” he emphasized. Obesity is excess body fat that poses a risk to health. A person with obesity may have 50 or more excess pounds, and the body is trying to defend this weight.
“If we want to treat obesity more effectively, we have to fully understand why it is a disease and how that disease differs from the cultural desire for thinness,” he reiterated.
“We have to keep the needs and goals of all people living with obesity foremost in our minds, even if many of them have been previously misled by the bias, stigma, blame, and discrimination that surrounds them.”
“We need to re-evaluate what we think we know about obesity and open our minds to new ideas,” he added.
Dr. Kaplan has reported financial ties to Eli Lilly, Gelesis, GI Dynamics, IntelliHealth, Johnson & Johnson, Novo Nordisk, Pfizer, and Rhythm Pharmaceuticals. Dr. Ryan has ties to numerous Novo Nordisk, Pfizer, and several other pharmaceutical companies, including having an ownership interest in Gila Therapeutics, Xeno Biosciences, Epitomee, Calibrate, Roman, and Scientific Intake.
A version of this article first appeared on Medscape.com.
“When considering the challenges of obesity, ask yourself: ‘If it were diabetes, cancer, HIV, or Alzheimer’s, how would you discuss it, approach it, assess it, treat it?’” Lee M. Kaplan, MD, PhD, asked the audience of health care professionals during ObesityWeek®, the annual meeting of The Obesity Society.
“And then do it for obesity, using the full spectrum of tools at our disposal,” he advised.
This was the takeaway that Dr. Kaplan, director of the Obesity, Metabolism, and Nutrition Institute at Massachusetts General Hospital and associate professor, Harvard Medical School, Boston, left the audience with at the end of his lecture entitled, “What does the future of obesity care look like?”
Invited to summarize his main points, Dr. Kaplan told this news organization in an interview that practitioners caring for patients with obesity need to first “recognize that obesity is a disease” caused by dysfunction of the metabolic system that regulates body fat – in the same way immune dysregulation can lead to asthma.
Second, “we are finally developing noninvasive therapies that are more effective,” he noted, referring to the recently approved semaglutide, and even more potent weight-loss therapies that could be on the market within 3 years, so that weight-loss outcomes with pharmacotherapy are approaching those with bariatric surgery.
Third, it is important that patients with obesity get “broad and equitable access” to treatment, and health care practitioners need to be on the same page and have a “shared understanding” of which treatments are appropriate for individual patients, “just as we do for other diseases.”
Need for a shared understanding
“Dr. Kaplan really brought home the idea that we all need a shared understanding of what obesity is – and what it is not,” agreed symposium moderator Donna H. Ryan, MD, in an email.
“He underscored the biologic basis of obesity,” noted Dr. Ryan, professor emerita at Pennington Biomedical Research Center in Baton Rouge, Louisiana, and associate editor-in-chief of Obesity, the official journal of The Obesity Society.
“It is a dysregulation of the body’s weight (especially adipose tissue) regulatory system,” she continued. “The body responds to powerful environmental pressures that produce excess energy balance, and we store that as fat and defend our highest fat mass. This makes obesity a disease, a chronic disease that requires a medical approach to reverse. It’s not a cosmetic problem, it’s a medical problem,” she emphasized.
There is so much misinformation out there about obesity, according to Dr. Ryan.
“People think it’s a lack of willpower, and even patients blame themselves for not being able to lose weight and keep it off. It’s not their fault! It’s biology.”
Although the supplement industry and fad diets falsely promise fast results, there is no magic diet, she continued.
“But we have made progress based on understanding the biologic basis of obesity and have new medications that offer real hope for patients.”
“With 42% of U.S. adults having a BMI that qualifies as obesity, we need a concerted and broad effort to address this problem, and that starts with everybody on the same page as to what obesity is ... a shared understanding of the biologic basis of obesity. It’s time to take obesity seriously,” she summarized, echoing Dr. Kaplan.
A question of biology
“Obesity results from inappropriate pathophysiological regulation of body fat mass,” when the body defends adiposity, Dr. Kaplan explained at the start of his lecture.
The treatment strategy for obesity has always been a stepwise approach starting with lifestyle changes, then pharmacotherapy, then possibly bariatric surgery – each step with a potentially greater chance of weight loss. But now, he explained, medicine is on the verge of having an armamentarium of more potent weight-loss medications.
Compared with phentermine/topiramate, orlistat, naltrexone/bupropion, and liraglutide – which roughly might provide 5% to 10% weight loss, the glucagon-like peptide-1 (GLP-1) agonist semaglutide 2.4 mg/week (Wegovy, Novo Nordisk), approved by the U.S. Food and Drug Association in June, provides almost double this potential weight loss.
And two new agents that could provide “never seen before weight loss” of 25% could potentially enter the marketplace by 2025: the amylin agonist cagrilintide (Novo Nordisk) and the twincretin tirzepatide (Eli Lilly) (a combined glucose-dependent insulinotropic polypeptide [GIP] and GLP-1 receptor agonist).
In addition, when liraglutide comes off patent, a generic version could potentially be introduced, and combined generic liraglutide plus generic phentermine/topiramate could be a less expensive weight-loss treatment option in the future, he noted.
One size does not fit all
Importantly, weight loss varies widely among individual patients.
A graph of potential weight loss with different treatments (for example, bariatric surgery or liraglutide) versus the percentage of patients that attain the weight losses is roughly bell-shaped, Dr. Kaplan explained. For example, in the STEP1 trial of semaglutide, roughly 7.1% of patients lost less than 5% of their initial weight, 25% of patients lost 20% to 30%, and 10.8% of patients lost 30% or more; that is, patients at the higher end had weight loss comparable to that seen with bariatric surgery
Adding pharmacotherapy after bariatric surgery could be synergistic. For example, in the GRAVITAS study of patients with type 2 diabetes who had gastric bypass surgery, those who received liraglutide after surgery had augmented weight loss compared with those who received placebo.
People at a cocktail party might come up to him and say, “I’d like to lose 5 pounds, 10 pounds,” Dr. Kaplan related in the Q&A session.
“That’s not obesity,” he emphasized. Obesity is excess body fat that poses a risk to health. A person with obesity may have 50 or more excess pounds, and the body is trying to defend this weight.
“If we want to treat obesity more effectively, we have to fully understand why it is a disease and how that disease differs from the cultural desire for thinness,” he reiterated.
“We have to keep the needs and goals of all people living with obesity foremost in our minds, even if many of them have been previously misled by the bias, stigma, blame, and discrimination that surrounds them.”
“We need to re-evaluate what we think we know about obesity and open our minds to new ideas,” he added.
Dr. Kaplan has reported financial ties to Eli Lilly, Gelesis, GI Dynamics, IntelliHealth, Johnson & Johnson, Novo Nordisk, Pfizer, and Rhythm Pharmaceuticals. Dr. Ryan has ties to numerous Novo Nordisk, Pfizer, and several other pharmaceutical companies, including having an ownership interest in Gila Therapeutics, Xeno Biosciences, Epitomee, Calibrate, Roman, and Scientific Intake.
A version of this article first appeared on Medscape.com.
FROM OBESITY WEEK 2020
Pembrolizumab-Induced Type 1 Diabetes in a 95-Year-Old Veteran With Metastatic Melanoma
Immune checkpoint inhibitors (CPIs) have revolutionized cancer therapy and improved the prognosis for a variety of advanced solid tumors and Hodgkin lymphoma, but evidence is growing regarding severe endocrine disturbances.1,2 CPIs block inhibitory molecules on activated T cells to increase tumor cell destruction but also can breach normal tolerance, resulting in a spectrum of immune-related adverse events (irAE).1,2 Programmed cell death-1 (PD-1) inhibitors are one type of CPIs. Pembrolizumab is a humanized monoclonal antibody that targets the PD-1 checkpoint pathway and is approved for the treatment of malignant melanoma and non-small cell lung cancer.3,4 When the PD-1 checkpoint pathway is inhibited, T cells targeting cancer are activated, as are autoreactive T cells, such as those regulating pancreatic islet cell survival, which can lead to type 1 diabetes mellitus (T1DM).5
Case Presentation
A 95-year-old male veteran with long-standing, stable prediabetes was treated with pembrolizumab for stage 4 melanoma. Four months after treatment initiation and 3 weeks after completion of his sixth treatment cycle of pembrolizumab (2 mg/kg every 3 weeks), he presented for surveillance positron emission tomography (PET) and was incidentally found to have a serum glucose of 423 mg/dL. Hypothesis-driven history taking revealed polyuria, polydipsia, and a 12-lb weight loss during the previous 3 months. The patient reported no abdominal pain, nausea, or vomiting. He showed no evidence of pancreatic metastases on recent imaging. His family history was notable for a daughter with T1DM diagnosed at a young age.
On examination, the patient’s vital signs were normal aside from a blood pressure of 80/40 mm Hg. His body mass index was 30. He was alert and oriented with comfortable respirations and no Kussmaul breathing. He exhibited dry mucous membranes and poor skin turgor. Laboratory studies revealed 135 mmol/L sodium (reference, 135-145), 4.6 mmol/L potassium (reference, 3.6-5.2), 100 mmol/L chloride (reference, 99-106), bicarbonate of 26.5 mmol/L (reference, 23-29), serum blood urea nitrogen 27 mg/dL (reference, 6-24), 1.06 mg/dL creatinine (reference, 0.74-1.35), and 423 mg/dL glucose (reference, 70-100), with negative urine ketones. Further studies demonstrated 462 µmol/L fructosamine (reference, 190-270), correlating with hemoglobin A1c (
Dicussion
Immunotherapy is now an integral part of cancer treatment and can result in endocrine disturbances.1,2 Life-threatening irAEs are rare and may mimic more common conditions; thus, there is growing recognition of the need to educate health care professionals in appropriate screening and management of these conditions. CPI-induced T1DM is an uncommon but clinically significant event with an incidence of 0.4 to 1.27% and a median onset of 20 weeks after initiation of therapy (range, 1-228 weeks).8-12In case seriesfrom 3 academic centers, 59 to81% of patients with CPI-induced T1DM presented with diabetic ketoacidosis (DKA), and only 40 to 71% of patients were autoantibody positive.13-16 These patients are older than those presenting with classic T1DM, often require intensive care unit admission, and nearly invariably require exogenous insulin injections for metabolic control.13-16
Based on the later age of onset of cancers that may be treated with CPI, patients with CPI-induced T1DM may be misdiagnosed with T2DM or hyperglycemia from other causes, such as medications or acute illness in the outpatient setting, risking suboptimal treatment.
Given the infrequent incidence and lack of controlled trials, screening and treatment recommendations for CPI-induced T1DM are based on principles derived from case series and expert opinion. Development of polyuria, polydipsia, weight loss, nausea, and/or vomiting should prompt investigation for possible development or worsening of hyperglycemia, suggestive of development of T1DM.17 American Society of Clinical Oncology (ASCO) guidelines recommend that serum glucose be assessed at baseline and with each treatment cycle during induction for 12 weeks, then every 3 to 6 weeks thereafter.17 There is no reported association between the number of CPI treatments and the development of DM.8,9 Following our patient’s fifth pembrolizumab cycle, a random glucose reading was noted to be 186 mg/dL (Figure 1). Under the ASCO guidelines, ideally the patient would have received close clinical follow-up given the striking increase in plasma glucose compared with prior baseline lower values and perhaps been further evaluated with an anti-GAD antibody titer to screen for T1DM.17
This patient's case adds to the published reports of CPI-induced T1DM without DKA and represents the oldest patient experiencing this irAE in the literature.13-16 The degree of elevation of his initial fructosamine, which is comparable to an average plasma glucose of approximately 270 mg/dL, belied the rapid rate of rise of his recent plasma glucose. Given the trajectory of glycemic markers and symptoms, one could certainly be concerned about imminent decompensation to DKA. However, fortuitous point-of-care glucose reading prior to surveillance PET resulted in a new critical diagnosis and initiation of treatment.
Assessing the need for inpatient evaluation includes obtaining urine ketones and acid-base status as screening for DKA.17 Antibodies and C-peptide can be sent to support diagnosis of new onset T1DM, although the initiation of therapy should not be delayed for these results.17 As noted before, many of these patients also are antibody negative.13-16 Low C-peptide levels should prompt a high suspicion for CPI-induced T1DM, and initiation of insulin therapy should be strongly considered.17 In a case series of 27 patients, 85% exhibited a rapid loss of β-cell function, evidenced by the acute progression to hyperglycemia and low or undetectable levels of C-peptide at diagnosis.9 Likewise, our patient had a low C-peptide level and negative anti-GAD antibody titer but was treated before these results were available. Inpatient admission for close glycemic monitoring may be reasonable; several cases reported prompt diagnosis and avoidance of DKA in this setting.17
In contrast to other irAEs, there is no available evidence that high-dose corticosteroids alter the course of pembrolizumab-induced T2DM.18 Depending on the degree of hyperglycemia, endocrinology consultation and insulin treatment are appropriate where the diagnosis of T1DM is suspected even without evidence of DKA.17 For patients with T2DM, there may be a positive synergistic effect of metformin in combination with CPIs in tumor control.19 Our patient’s C-peptide improved with insulin treatment, consistent with correction of glucose toxicity and a honeymoon period in his course. However, in patients reported with pembrolizumab-induced T1DM, insulin requirement for treatment generally persists despite cessation of pembrolizumab therapy.13-16
Conclusions
Pembrolizumab-induced T1DM is a rare, but potentially life-threatening irAE. The acute risk of DKA requires early recognition and prompt treatment of patients taking CPIs. More than 90% of primary care physicians (PCPs) fulfill general medical care roles for patients with cancer; therefore, they play an essential role in evaluating symptoms during therapy.20 Further studies evaluating the role of PCPs and outcomes when PCPs are involved in oncologic care should be conducted.
With increased index of suspicion, this clinical scenario presents an opportunity for PCPs that may help reduce irAE-associated morbidity and mortality of patients on CPIs, like pembrolizumab. Figure 2 illustrates an example addendum that can be used to alert and tag a PCP of a mutual patient after initiation of CPI therapy. Determining the optimal interface between PCPs, oncologists, and endocrinologists in delivering and coordinating high-quality cancer care in the setting of immunotherapy is an important area for ongoing quality improvement.
Acknowledgment
The authors thank all the staff and health care professionals at VA Greater Los Angeles Healthcare System who were involved in the care of this patient.
1. Puzanov I, Diab A, Abdallah K, et al; Society for Immunotherapy of Cancer Toxicity Management Working Group. Managing toxicities associated with immune checkpoint inhibitors: consensus recommendations from the Society for Immunotherapy of Cancer (SITC) Toxicity Management Working Group. J Immunother Cancer. 2017;5(1):95. doi:10.1186/s40425-017-0300-z
2. Villa NM, Farahmand A, Du L, et al. Endocrinopathies with use of cancer immunotherapies. Clin Endocrinol (Oxf). 2018;88(2):327-332. doi:10.1111/cen.13483
3. Schachter J, Ribas A, Long GV, et al. Pembrolizumab versus ipilimumab for advanced melanoma: final overall survival results of a multicentre, randomised, open-label phase 3 study (KEYNOTE-006). Lancet. 2017;390(10105):1853-1862. doi:10.1016/S0140-6736(17)31601-X
4. Garon EB, Hellmann MD, Rizvi NA, et al. Five-year overall survival for patients with advanced non-small-cell lung cancer treated with pembrolizumab: results from the phase I KEYNOTE-001 Study. J Clin Oncol. 2019;37(28):2518-2527. doi:10.1200/JCO.19.00934
5. Ribas A. Tumor immunotherapy directed at PD-1. N Engl J Med. 2012;366(26):2517-2519. doi:10.1056/NEJMe1205943
6. Malmstrom H, Walldius G, Grill V, Jungner I, Gudbjomsdottir S, Hammar N. Frustosamine is a useful indicator of hyperglycemia and glucose control in clinical and epidemiological studies- cross-sectional and longitudinal experience from the AMORIS cohort. PLoS One. 2014;9(10):e111463. doi:10.1371/journal.pone.0111463
7. Skinner S, Diaw M, Mbaye MN, et al. Evaluation of agreement between hemoglobin A1c, fasting glucose, and fructosamine in Senagalese individuals with and without sickle-cell trait. PLoS One. 2019;14(2):e0212552. doi:10.1371/journal.pone.0212552
8. Byun DJ, Wolchok JD, Rosenberg LM, Girotra M. Cancer immunotherapy-immune checkpoint blockade and associated endocrinopathies. Nat Rev Endocrinol. 2017;13(4):195-207. doi:10.1038/nrendo.2016.205
9. Stamatouli AM, Quandt Z, Perdigoto AL, et al. Collateral damage: insulin-dependent diabetes induced with checkpoint inhibitors. Diabetes. 2018;67(8):1471-1480. doi:10.2337/dbi18-0002
10. Liu J, Zhou H, Zhang Y, et al. Reporting of immune checkpoint inhibitor therapy-associated diabetes, 2015-2019. Diabetes Care. 2020;43(7):e79-e80. [Published online ahead of print, 2020 May 11]. doi:10.2337/dc20-0459
11. Barroso-Sousa R, Barry WT, Garrido-Castro AC, et al. Incidence of endocrine dysfunction following the use of different immune checkpoint inhibitor regimens: a systematic review and meta-analysis. JAMA Oncol. 2018;4(2):173-182. doi:10.1001/jamaoncol.2017.3064
12. de Filette J, Andreescu CE, Cools F, Bravenboer B, Velkeniers B. A systematic review and meta-analysis of endocrine-related adverse events associated with immune checkpoint inhibitors. Horm Metab Res. 2019;51(3):145-156. doi:10.1055/a-0843-3366
13. Hughes J, Vudattu N, Sznol M, et al. Precipitation of autoimmune diabetes with anti-PD-1 immunotherapy. Diabetes Care. 2015;38(4):e55-e57. doi:10.2337/dc14-2349
14. Clotman K, Janssens K, Specenier P, Weets I, De block CEM. Programmed cell death-1 inhibitor-induced type 1 diabetes mellitus. J Clin Endocrinol Metab. 2018;103(9):3144-3154. doi:10.1210/jc.2018-00728
15. Kotwal A, Haddox C, Block M, Kudva YC. Immune checkpoint inhibitors: an emerging cause of insulin-dependent diabetes. BMJ Open Diabetes Res Care. 2019;7(1):e000591. doi:10.1136/bmjdrc-2018-000591
16. Chang LS, Barroso-Sousa R, Tolaney SM, Hodi FS, Kaiser UB, Min L. Endocrine toxicity of cancer immunotherapy targeting immune checkpoints. Endocr Rev. 2019;40(1):17-65. doi:10.1210/er.2018-00006
17. Brahmer JR, Lacchetti C, Schneider BJ, et al; National Comprehensive Cancer Network. Management of immune-related adverse events in patients treated with immune checkpoint inhibitor therapy: American Society of Clinical Oncology Clinical Practice Guideline. J Clin Oncol. 2018;36(17):1714-1768. doi:10.1200/JCO.2017.77.6385
18. Aleksova J, Lau PK, Soldatos G, Mcarthur G. Glucocorticoids did not reverse type 1 diabetes mellitus secondary to pembrolizumab in a patient with metastatic melanoma. BMJ Case Rep. 2016;2016:bcr2016217454. doi:10.1136/bcr-2016-217454
19. Afzal MZ, Mercado RR, Shirai K. Efficacy of metformin in combination with immune checkpoint inhibitors (anti-PD-1/anti-CTLA-4) in metastatic malignant melanoma. J Immunother Cancer. 2018;6(1):64. doi:10.1186/s40425-018-0375-1
20. Klabunde CN, Ambs A, Keating NL, et al. The role of primary care physicians in cancer care. J Gen Intern Med. 2009;24(9):1029-1036. doi:10.1007/s11606-009-1058-x
Immune checkpoint inhibitors (CPIs) have revolutionized cancer therapy and improved the prognosis for a variety of advanced solid tumors and Hodgkin lymphoma, but evidence is growing regarding severe endocrine disturbances.1,2 CPIs block inhibitory molecules on activated T cells to increase tumor cell destruction but also can breach normal tolerance, resulting in a spectrum of immune-related adverse events (irAE).1,2 Programmed cell death-1 (PD-1) inhibitors are one type of CPIs. Pembrolizumab is a humanized monoclonal antibody that targets the PD-1 checkpoint pathway and is approved for the treatment of malignant melanoma and non-small cell lung cancer.3,4 When the PD-1 checkpoint pathway is inhibited, T cells targeting cancer are activated, as are autoreactive T cells, such as those regulating pancreatic islet cell survival, which can lead to type 1 diabetes mellitus (T1DM).5
Case Presentation
A 95-year-old male veteran with long-standing, stable prediabetes was treated with pembrolizumab for stage 4 melanoma. Four months after treatment initiation and 3 weeks after completion of his sixth treatment cycle of pembrolizumab (2 mg/kg every 3 weeks), he presented for surveillance positron emission tomography (PET) and was incidentally found to have a serum glucose of 423 mg/dL. Hypothesis-driven history taking revealed polyuria, polydipsia, and a 12-lb weight loss during the previous 3 months. The patient reported no abdominal pain, nausea, or vomiting. He showed no evidence of pancreatic metastases on recent imaging. His family history was notable for a daughter with T1DM diagnosed at a young age.
On examination, the patient’s vital signs were normal aside from a blood pressure of 80/40 mm Hg. His body mass index was 30. He was alert and oriented with comfortable respirations and no Kussmaul breathing. He exhibited dry mucous membranes and poor skin turgor. Laboratory studies revealed 135 mmol/L sodium (reference, 135-145), 4.6 mmol/L potassium (reference, 3.6-5.2), 100 mmol/L chloride (reference, 99-106), bicarbonate of 26.5 mmol/L (reference, 23-29), serum blood urea nitrogen 27 mg/dL (reference, 6-24), 1.06 mg/dL creatinine (reference, 0.74-1.35), and 423 mg/dL glucose (reference, 70-100), with negative urine ketones. Further studies demonstrated 462 µmol/L fructosamine (reference, 190-270), correlating with hemoglobin A1c (
Dicussion
Immunotherapy is now an integral part of cancer treatment and can result in endocrine disturbances.1,2 Life-threatening irAEs are rare and may mimic more common conditions; thus, there is growing recognition of the need to educate health care professionals in appropriate screening and management of these conditions. CPI-induced T1DM is an uncommon but clinically significant event with an incidence of 0.4 to 1.27% and a median onset of 20 weeks after initiation of therapy (range, 1-228 weeks).8-12In case seriesfrom 3 academic centers, 59 to81% of patients with CPI-induced T1DM presented with diabetic ketoacidosis (DKA), and only 40 to 71% of patients were autoantibody positive.13-16 These patients are older than those presenting with classic T1DM, often require intensive care unit admission, and nearly invariably require exogenous insulin injections for metabolic control.13-16
Based on the later age of onset of cancers that may be treated with CPI, patients with CPI-induced T1DM may be misdiagnosed with T2DM or hyperglycemia from other causes, such as medications or acute illness in the outpatient setting, risking suboptimal treatment.
Given the infrequent incidence and lack of controlled trials, screening and treatment recommendations for CPI-induced T1DM are based on principles derived from case series and expert opinion. Development of polyuria, polydipsia, weight loss, nausea, and/or vomiting should prompt investigation for possible development or worsening of hyperglycemia, suggestive of development of T1DM.17 American Society of Clinical Oncology (ASCO) guidelines recommend that serum glucose be assessed at baseline and with each treatment cycle during induction for 12 weeks, then every 3 to 6 weeks thereafter.17 There is no reported association between the number of CPI treatments and the development of DM.8,9 Following our patient’s fifth pembrolizumab cycle, a random glucose reading was noted to be 186 mg/dL (Figure 1). Under the ASCO guidelines, ideally the patient would have received close clinical follow-up given the striking increase in plasma glucose compared with prior baseline lower values and perhaps been further evaluated with an anti-GAD antibody titer to screen for T1DM.17
This patient's case adds to the published reports of CPI-induced T1DM without DKA and represents the oldest patient experiencing this irAE in the literature.13-16 The degree of elevation of his initial fructosamine, which is comparable to an average plasma glucose of approximately 270 mg/dL, belied the rapid rate of rise of his recent plasma glucose. Given the trajectory of glycemic markers and symptoms, one could certainly be concerned about imminent decompensation to DKA. However, fortuitous point-of-care glucose reading prior to surveillance PET resulted in a new critical diagnosis and initiation of treatment.
Assessing the need for inpatient evaluation includes obtaining urine ketones and acid-base status as screening for DKA.17 Antibodies and C-peptide can be sent to support diagnosis of new onset T1DM, although the initiation of therapy should not be delayed for these results.17 As noted before, many of these patients also are antibody negative.13-16 Low C-peptide levels should prompt a high suspicion for CPI-induced T1DM, and initiation of insulin therapy should be strongly considered.17 In a case series of 27 patients, 85% exhibited a rapid loss of β-cell function, evidenced by the acute progression to hyperglycemia and low or undetectable levels of C-peptide at diagnosis.9 Likewise, our patient had a low C-peptide level and negative anti-GAD antibody titer but was treated before these results were available. Inpatient admission for close glycemic monitoring may be reasonable; several cases reported prompt diagnosis and avoidance of DKA in this setting.17
In contrast to other irAEs, there is no available evidence that high-dose corticosteroids alter the course of pembrolizumab-induced T2DM.18 Depending on the degree of hyperglycemia, endocrinology consultation and insulin treatment are appropriate where the diagnosis of T1DM is suspected even without evidence of DKA.17 For patients with T2DM, there may be a positive synergistic effect of metformin in combination with CPIs in tumor control.19 Our patient’s C-peptide improved with insulin treatment, consistent with correction of glucose toxicity and a honeymoon period in his course. However, in patients reported with pembrolizumab-induced T1DM, insulin requirement for treatment generally persists despite cessation of pembrolizumab therapy.13-16
Conclusions
Pembrolizumab-induced T1DM is a rare, but potentially life-threatening irAE. The acute risk of DKA requires early recognition and prompt treatment of patients taking CPIs. More than 90% of primary care physicians (PCPs) fulfill general medical care roles for patients with cancer; therefore, they play an essential role in evaluating symptoms during therapy.20 Further studies evaluating the role of PCPs and outcomes when PCPs are involved in oncologic care should be conducted.
With increased index of suspicion, this clinical scenario presents an opportunity for PCPs that may help reduce irAE-associated morbidity and mortality of patients on CPIs, like pembrolizumab. Figure 2 illustrates an example addendum that can be used to alert and tag a PCP of a mutual patient after initiation of CPI therapy. Determining the optimal interface between PCPs, oncologists, and endocrinologists in delivering and coordinating high-quality cancer care in the setting of immunotherapy is an important area for ongoing quality improvement.
Acknowledgment
The authors thank all the staff and health care professionals at VA Greater Los Angeles Healthcare System who were involved in the care of this patient.
Immune checkpoint inhibitors (CPIs) have revolutionized cancer therapy and improved the prognosis for a variety of advanced solid tumors and Hodgkin lymphoma, but evidence is growing regarding severe endocrine disturbances.1,2 CPIs block inhibitory molecules on activated T cells to increase tumor cell destruction but also can breach normal tolerance, resulting in a spectrum of immune-related adverse events (irAE).1,2 Programmed cell death-1 (PD-1) inhibitors are one type of CPIs. Pembrolizumab is a humanized monoclonal antibody that targets the PD-1 checkpoint pathway and is approved for the treatment of malignant melanoma and non-small cell lung cancer.3,4 When the PD-1 checkpoint pathway is inhibited, T cells targeting cancer are activated, as are autoreactive T cells, such as those regulating pancreatic islet cell survival, which can lead to type 1 diabetes mellitus (T1DM).5
Case Presentation
A 95-year-old male veteran with long-standing, stable prediabetes was treated with pembrolizumab for stage 4 melanoma. Four months after treatment initiation and 3 weeks after completion of his sixth treatment cycle of pembrolizumab (2 mg/kg every 3 weeks), he presented for surveillance positron emission tomography (PET) and was incidentally found to have a serum glucose of 423 mg/dL. Hypothesis-driven history taking revealed polyuria, polydipsia, and a 12-lb weight loss during the previous 3 months. The patient reported no abdominal pain, nausea, or vomiting. He showed no evidence of pancreatic metastases on recent imaging. His family history was notable for a daughter with T1DM diagnosed at a young age.
On examination, the patient’s vital signs were normal aside from a blood pressure of 80/40 mm Hg. His body mass index was 30. He was alert and oriented with comfortable respirations and no Kussmaul breathing. He exhibited dry mucous membranes and poor skin turgor. Laboratory studies revealed 135 mmol/L sodium (reference, 135-145), 4.6 mmol/L potassium (reference, 3.6-5.2), 100 mmol/L chloride (reference, 99-106), bicarbonate of 26.5 mmol/L (reference, 23-29), serum blood urea nitrogen 27 mg/dL (reference, 6-24), 1.06 mg/dL creatinine (reference, 0.74-1.35), and 423 mg/dL glucose (reference, 70-100), with negative urine ketones. Further studies demonstrated 462 µmol/L fructosamine (reference, 190-270), correlating with hemoglobin A1c (
Dicussion
Immunotherapy is now an integral part of cancer treatment and can result in endocrine disturbances.1,2 Life-threatening irAEs are rare and may mimic more common conditions; thus, there is growing recognition of the need to educate health care professionals in appropriate screening and management of these conditions. CPI-induced T1DM is an uncommon but clinically significant event with an incidence of 0.4 to 1.27% and a median onset of 20 weeks after initiation of therapy (range, 1-228 weeks).8-12In case seriesfrom 3 academic centers, 59 to81% of patients with CPI-induced T1DM presented with diabetic ketoacidosis (DKA), and only 40 to 71% of patients were autoantibody positive.13-16 These patients are older than those presenting with classic T1DM, often require intensive care unit admission, and nearly invariably require exogenous insulin injections for metabolic control.13-16
Based on the later age of onset of cancers that may be treated with CPI, patients with CPI-induced T1DM may be misdiagnosed with T2DM or hyperglycemia from other causes, such as medications or acute illness in the outpatient setting, risking suboptimal treatment.
Given the infrequent incidence and lack of controlled trials, screening and treatment recommendations for CPI-induced T1DM are based on principles derived from case series and expert opinion. Development of polyuria, polydipsia, weight loss, nausea, and/or vomiting should prompt investigation for possible development or worsening of hyperglycemia, suggestive of development of T1DM.17 American Society of Clinical Oncology (ASCO) guidelines recommend that serum glucose be assessed at baseline and with each treatment cycle during induction for 12 weeks, then every 3 to 6 weeks thereafter.17 There is no reported association between the number of CPI treatments and the development of DM.8,9 Following our patient’s fifth pembrolizumab cycle, a random glucose reading was noted to be 186 mg/dL (Figure 1). Under the ASCO guidelines, ideally the patient would have received close clinical follow-up given the striking increase in plasma glucose compared with prior baseline lower values and perhaps been further evaluated with an anti-GAD antibody titer to screen for T1DM.17
This patient's case adds to the published reports of CPI-induced T1DM without DKA and represents the oldest patient experiencing this irAE in the literature.13-16 The degree of elevation of his initial fructosamine, which is comparable to an average plasma glucose of approximately 270 mg/dL, belied the rapid rate of rise of his recent plasma glucose. Given the trajectory of glycemic markers and symptoms, one could certainly be concerned about imminent decompensation to DKA. However, fortuitous point-of-care glucose reading prior to surveillance PET resulted in a new critical diagnosis and initiation of treatment.
Assessing the need for inpatient evaluation includes obtaining urine ketones and acid-base status as screening for DKA.17 Antibodies and C-peptide can be sent to support diagnosis of new onset T1DM, although the initiation of therapy should not be delayed for these results.17 As noted before, many of these patients also are antibody negative.13-16 Low C-peptide levels should prompt a high suspicion for CPI-induced T1DM, and initiation of insulin therapy should be strongly considered.17 In a case series of 27 patients, 85% exhibited a rapid loss of β-cell function, evidenced by the acute progression to hyperglycemia and low or undetectable levels of C-peptide at diagnosis.9 Likewise, our patient had a low C-peptide level and negative anti-GAD antibody titer but was treated before these results were available. Inpatient admission for close glycemic monitoring may be reasonable; several cases reported prompt diagnosis and avoidance of DKA in this setting.17
In contrast to other irAEs, there is no available evidence that high-dose corticosteroids alter the course of pembrolizumab-induced T2DM.18 Depending on the degree of hyperglycemia, endocrinology consultation and insulin treatment are appropriate where the diagnosis of T1DM is suspected even without evidence of DKA.17 For patients with T2DM, there may be a positive synergistic effect of metformin in combination with CPIs in tumor control.19 Our patient’s C-peptide improved with insulin treatment, consistent with correction of glucose toxicity and a honeymoon period in his course. However, in patients reported with pembrolizumab-induced T1DM, insulin requirement for treatment generally persists despite cessation of pembrolizumab therapy.13-16
Conclusions
Pembrolizumab-induced T1DM is a rare, but potentially life-threatening irAE. The acute risk of DKA requires early recognition and prompt treatment of patients taking CPIs. More than 90% of primary care physicians (PCPs) fulfill general medical care roles for patients with cancer; therefore, they play an essential role in evaluating symptoms during therapy.20 Further studies evaluating the role of PCPs and outcomes when PCPs are involved in oncologic care should be conducted.
With increased index of suspicion, this clinical scenario presents an opportunity for PCPs that may help reduce irAE-associated morbidity and mortality of patients on CPIs, like pembrolizumab. Figure 2 illustrates an example addendum that can be used to alert and tag a PCP of a mutual patient after initiation of CPI therapy. Determining the optimal interface between PCPs, oncologists, and endocrinologists in delivering and coordinating high-quality cancer care in the setting of immunotherapy is an important area for ongoing quality improvement.
Acknowledgment
The authors thank all the staff and health care professionals at VA Greater Los Angeles Healthcare System who were involved in the care of this patient.
1. Puzanov I, Diab A, Abdallah K, et al; Society for Immunotherapy of Cancer Toxicity Management Working Group. Managing toxicities associated with immune checkpoint inhibitors: consensus recommendations from the Society for Immunotherapy of Cancer (SITC) Toxicity Management Working Group. J Immunother Cancer. 2017;5(1):95. doi:10.1186/s40425-017-0300-z
2. Villa NM, Farahmand A, Du L, et al. Endocrinopathies with use of cancer immunotherapies. Clin Endocrinol (Oxf). 2018;88(2):327-332. doi:10.1111/cen.13483
3. Schachter J, Ribas A, Long GV, et al. Pembrolizumab versus ipilimumab for advanced melanoma: final overall survival results of a multicentre, randomised, open-label phase 3 study (KEYNOTE-006). Lancet. 2017;390(10105):1853-1862. doi:10.1016/S0140-6736(17)31601-X
4. Garon EB, Hellmann MD, Rizvi NA, et al. Five-year overall survival for patients with advanced non-small-cell lung cancer treated with pembrolizumab: results from the phase I KEYNOTE-001 Study. J Clin Oncol. 2019;37(28):2518-2527. doi:10.1200/JCO.19.00934
5. Ribas A. Tumor immunotherapy directed at PD-1. N Engl J Med. 2012;366(26):2517-2519. doi:10.1056/NEJMe1205943
6. Malmstrom H, Walldius G, Grill V, Jungner I, Gudbjomsdottir S, Hammar N. Frustosamine is a useful indicator of hyperglycemia and glucose control in clinical and epidemiological studies- cross-sectional and longitudinal experience from the AMORIS cohort. PLoS One. 2014;9(10):e111463. doi:10.1371/journal.pone.0111463
7. Skinner S, Diaw M, Mbaye MN, et al. Evaluation of agreement between hemoglobin A1c, fasting glucose, and fructosamine in Senagalese individuals with and without sickle-cell trait. PLoS One. 2019;14(2):e0212552. doi:10.1371/journal.pone.0212552
8. Byun DJ, Wolchok JD, Rosenberg LM, Girotra M. Cancer immunotherapy-immune checkpoint blockade and associated endocrinopathies. Nat Rev Endocrinol. 2017;13(4):195-207. doi:10.1038/nrendo.2016.205
9. Stamatouli AM, Quandt Z, Perdigoto AL, et al. Collateral damage: insulin-dependent diabetes induced with checkpoint inhibitors. Diabetes. 2018;67(8):1471-1480. doi:10.2337/dbi18-0002
10. Liu J, Zhou H, Zhang Y, et al. Reporting of immune checkpoint inhibitor therapy-associated diabetes, 2015-2019. Diabetes Care. 2020;43(7):e79-e80. [Published online ahead of print, 2020 May 11]. doi:10.2337/dc20-0459
11. Barroso-Sousa R, Barry WT, Garrido-Castro AC, et al. Incidence of endocrine dysfunction following the use of different immune checkpoint inhibitor regimens: a systematic review and meta-analysis. JAMA Oncol. 2018;4(2):173-182. doi:10.1001/jamaoncol.2017.3064
12. de Filette J, Andreescu CE, Cools F, Bravenboer B, Velkeniers B. A systematic review and meta-analysis of endocrine-related adverse events associated with immune checkpoint inhibitors. Horm Metab Res. 2019;51(3):145-156. doi:10.1055/a-0843-3366
13. Hughes J, Vudattu N, Sznol M, et al. Precipitation of autoimmune diabetes with anti-PD-1 immunotherapy. Diabetes Care. 2015;38(4):e55-e57. doi:10.2337/dc14-2349
14. Clotman K, Janssens K, Specenier P, Weets I, De block CEM. Programmed cell death-1 inhibitor-induced type 1 diabetes mellitus. J Clin Endocrinol Metab. 2018;103(9):3144-3154. doi:10.1210/jc.2018-00728
15. Kotwal A, Haddox C, Block M, Kudva YC. Immune checkpoint inhibitors: an emerging cause of insulin-dependent diabetes. BMJ Open Diabetes Res Care. 2019;7(1):e000591. doi:10.1136/bmjdrc-2018-000591
16. Chang LS, Barroso-Sousa R, Tolaney SM, Hodi FS, Kaiser UB, Min L. Endocrine toxicity of cancer immunotherapy targeting immune checkpoints. Endocr Rev. 2019;40(1):17-65. doi:10.1210/er.2018-00006
17. Brahmer JR, Lacchetti C, Schneider BJ, et al; National Comprehensive Cancer Network. Management of immune-related adverse events in patients treated with immune checkpoint inhibitor therapy: American Society of Clinical Oncology Clinical Practice Guideline. J Clin Oncol. 2018;36(17):1714-1768. doi:10.1200/JCO.2017.77.6385
18. Aleksova J, Lau PK, Soldatos G, Mcarthur G. Glucocorticoids did not reverse type 1 diabetes mellitus secondary to pembrolizumab in a patient with metastatic melanoma. BMJ Case Rep. 2016;2016:bcr2016217454. doi:10.1136/bcr-2016-217454
19. Afzal MZ, Mercado RR, Shirai K. Efficacy of metformin in combination with immune checkpoint inhibitors (anti-PD-1/anti-CTLA-4) in metastatic malignant melanoma. J Immunother Cancer. 2018;6(1):64. doi:10.1186/s40425-018-0375-1
20. Klabunde CN, Ambs A, Keating NL, et al. The role of primary care physicians in cancer care. J Gen Intern Med. 2009;24(9):1029-1036. doi:10.1007/s11606-009-1058-x
1. Puzanov I, Diab A, Abdallah K, et al; Society for Immunotherapy of Cancer Toxicity Management Working Group. Managing toxicities associated with immune checkpoint inhibitors: consensus recommendations from the Society for Immunotherapy of Cancer (SITC) Toxicity Management Working Group. J Immunother Cancer. 2017;5(1):95. doi:10.1186/s40425-017-0300-z
2. Villa NM, Farahmand A, Du L, et al. Endocrinopathies with use of cancer immunotherapies. Clin Endocrinol (Oxf). 2018;88(2):327-332. doi:10.1111/cen.13483
3. Schachter J, Ribas A, Long GV, et al. Pembrolizumab versus ipilimumab for advanced melanoma: final overall survival results of a multicentre, randomised, open-label phase 3 study (KEYNOTE-006). Lancet. 2017;390(10105):1853-1862. doi:10.1016/S0140-6736(17)31601-X
4. Garon EB, Hellmann MD, Rizvi NA, et al. Five-year overall survival for patients with advanced non-small-cell lung cancer treated with pembrolizumab: results from the phase I KEYNOTE-001 Study. J Clin Oncol. 2019;37(28):2518-2527. doi:10.1200/JCO.19.00934
5. Ribas A. Tumor immunotherapy directed at PD-1. N Engl J Med. 2012;366(26):2517-2519. doi:10.1056/NEJMe1205943
6. Malmstrom H, Walldius G, Grill V, Jungner I, Gudbjomsdottir S, Hammar N. Frustosamine is a useful indicator of hyperglycemia and glucose control in clinical and epidemiological studies- cross-sectional and longitudinal experience from the AMORIS cohort. PLoS One. 2014;9(10):e111463. doi:10.1371/journal.pone.0111463
7. Skinner S, Diaw M, Mbaye MN, et al. Evaluation of agreement between hemoglobin A1c, fasting glucose, and fructosamine in Senagalese individuals with and without sickle-cell trait. PLoS One. 2019;14(2):e0212552. doi:10.1371/journal.pone.0212552
8. Byun DJ, Wolchok JD, Rosenberg LM, Girotra M. Cancer immunotherapy-immune checkpoint blockade and associated endocrinopathies. Nat Rev Endocrinol. 2017;13(4):195-207. doi:10.1038/nrendo.2016.205
9. Stamatouli AM, Quandt Z, Perdigoto AL, et al. Collateral damage: insulin-dependent diabetes induced with checkpoint inhibitors. Diabetes. 2018;67(8):1471-1480. doi:10.2337/dbi18-0002
10. Liu J, Zhou H, Zhang Y, et al. Reporting of immune checkpoint inhibitor therapy-associated diabetes, 2015-2019. Diabetes Care. 2020;43(7):e79-e80. [Published online ahead of print, 2020 May 11]. doi:10.2337/dc20-0459
11. Barroso-Sousa R, Barry WT, Garrido-Castro AC, et al. Incidence of endocrine dysfunction following the use of different immune checkpoint inhibitor regimens: a systematic review and meta-analysis. JAMA Oncol. 2018;4(2):173-182. doi:10.1001/jamaoncol.2017.3064
12. de Filette J, Andreescu CE, Cools F, Bravenboer B, Velkeniers B. A systematic review and meta-analysis of endocrine-related adverse events associated with immune checkpoint inhibitors. Horm Metab Res. 2019;51(3):145-156. doi:10.1055/a-0843-3366
13. Hughes J, Vudattu N, Sznol M, et al. Precipitation of autoimmune diabetes with anti-PD-1 immunotherapy. Diabetes Care. 2015;38(4):e55-e57. doi:10.2337/dc14-2349
14. Clotman K, Janssens K, Specenier P, Weets I, De block CEM. Programmed cell death-1 inhibitor-induced type 1 diabetes mellitus. J Clin Endocrinol Metab. 2018;103(9):3144-3154. doi:10.1210/jc.2018-00728
15. Kotwal A, Haddox C, Block M, Kudva YC. Immune checkpoint inhibitors: an emerging cause of insulin-dependent diabetes. BMJ Open Diabetes Res Care. 2019;7(1):e000591. doi:10.1136/bmjdrc-2018-000591
16. Chang LS, Barroso-Sousa R, Tolaney SM, Hodi FS, Kaiser UB, Min L. Endocrine toxicity of cancer immunotherapy targeting immune checkpoints. Endocr Rev. 2019;40(1):17-65. doi:10.1210/er.2018-00006
17. Brahmer JR, Lacchetti C, Schneider BJ, et al; National Comprehensive Cancer Network. Management of immune-related adverse events in patients treated with immune checkpoint inhibitor therapy: American Society of Clinical Oncology Clinical Practice Guideline. J Clin Oncol. 2018;36(17):1714-1768. doi:10.1200/JCO.2017.77.6385
18. Aleksova J, Lau PK, Soldatos G, Mcarthur G. Glucocorticoids did not reverse type 1 diabetes mellitus secondary to pembrolizumab in a patient with metastatic melanoma. BMJ Case Rep. 2016;2016:bcr2016217454. doi:10.1136/bcr-2016-217454
19. Afzal MZ, Mercado RR, Shirai K. Efficacy of metformin in combination with immune checkpoint inhibitors (anti-PD-1/anti-CTLA-4) in metastatic malignant melanoma. J Immunother Cancer. 2018;6(1):64. doi:10.1186/s40425-018-0375-1
20. Klabunde CN, Ambs A, Keating NL, et al. The role of primary care physicians in cancer care. J Gen Intern Med. 2009;24(9):1029-1036. doi:10.1007/s11606-009-1058-x
SUGAR trial finds superior stent for those with diabetes and CAD
Superiority shown on TLF endpoint
Designed to show noninferiority for treatment of coronary artery disease (CAD) in patients with diabetes, a head-to-head comparison of contemporary stents ended up showing that one was superior to the for the primary endpoint of target lesion failure (TLF).
In the superiority analysis, the 35% relative reduction in the risk of TLF at 1 year for the Cre8 EVO (Alvimedica) stent relative to the Resolute Onyx (Medtronic) device reached significance, according to Rafael Romaguera, MD, PhD, an interventional cardiologist at the Bellvitge University Hospital, Barcelona.
At 1 year, the rates of TLF were 7.2% and 10.5% for the Cre8 EVO and Resolute Onyx stents, respectively. On the basis of noninferiority, the 3.73% reduction in TLF at 1 year among those receiving the Cre8 EVO device provided a highly significant confirmation of noninferiority (P < .001) and triggered the preplanned superiority analysis.
When the significant advantage on the TLF endpoint (P = .03) was broken down into its components, the Cre8 EVO stent was linked to numerically lower rates of cardiac death (2.1% vs. 2.7%), target vessel MI (5.3% vs. 7.2%), and target lesion revascularization (2.4% vs. 3.9%), according to the SUGAR (Second-Generation Drug-Eluting Stents in Diabetes) trial results presented at the Transcatheter Cardiovascular Therapeutics annual meeting, held virtually and live in Orlando and sponsored by the Cardiovascular Research Foundation.
In a previous study comparing these devices, called the ReCre8 trial, the rates of TLF in an all-comer CAD population were similar at 1 year. When an updated 3-year analysis was presented earlier in 2021 at the Cardiovascular Research Technologies meeting, they remained similar.
Diabetes-centered trial was unmet need
The rationale for conducting a new trial limited to patients with diabetes was based on the greater risk in this population, according to Dr. Romaguera. He cited data that indicate the risk of major adverse cardiac events are about two times higher 2 years after stent implantation in patients with diabetes relative to those without, even when contemporary drug-eluting stents are used.
Both the Cre8 EVO and Resolute Onyx stent are drug eluting and employ contemporary architecture that provides the basis for marketing claims that they are suitable for complex patients; but they have differences.
“There are three features that I think differentiate the Cre8 EVO stent,” Dr. Romaguera reported at the meeting, sponsored by the Cardiovascular Research Foundation.
One is the absence of polymer, which contrasts with the permanent polymer of the Resolute device. This feature affects the dissolution of the anti-inflammatory drug and might be one explanation for the greater protection from ischemic events, according to Dr. Romaguera.
Another is the thickness of the struts, which range from 70 to 80 mm for the Cre8 EVO device and from 92 to 102 mm for the Resolute Onyx device. In experimental studies, strut thickness has been associated with greater risk of thrombus formation, although it is unclear if this modest difference is clinically significant.
Also important, the Cre8 EVO device employs sirolimus for an anti-inflammatory effect, while the Resolute Onyx elutes zotarolimus. Again, experimental evidence suggests a greater anti-inflammatory effect reduces the need for dual-antiplatelet therapy (DAPT); that might offer a relative advantage in patients with an elevated risk of bleeding.
It is not clear whether all of these features contribute to the better results observed in this trial in diabetes patients, but Dr. Romaguera indicated that the lower risk of TLF with Cre8 EVO is not just statistically significant but also clinically meaningful.
In SUGAR, which included 23 centers in Spain, 1,175 patients with confirmed diabetes scheduled for percutaneous intervention (PCI) were randomized to one of the two stents. The study was purposely designed with very few exclusion criteria.
SUGAR trial employed all-comer design
“This was an all-comer design and there was no limitation in regard to clinical presentation, complexity, number of lesions, or other disease features,” said Dr. Romaguera. The major exclusions were a life expectancy of less than 2 years and a contraindication to taking DAPT for at least 1 month,
The patients were almost equally divided between those who had a non–ST-segment elevation MI) and those with chronic coronary artery disease, but patients with a STEMI, representing about 12% of the population, were included. Almost all of the patients (about 95%) had type 2 diabetes; nearly one-third were on insulin at the time of randomization.
According to Dr. Romaguera, “SUGAR is the first powered trial to compare new-generation drug-eluting stents in patients with diabetes,” and he emphasized the all-comer design in supporting its clinical relevance.
Several of those participating in discussion of the trial during the late-breaker session agreed. Although the moderator, Gregg Stone, MD, of the Icahn School of Medicine at Mount Sinai, New York, expressed surprise that the trial “actually demonstrated superiority” given the difficulty of showing a difference between modern stents, he called the findings “remarkable.”
Others seemed to suggest that it would alter their practice.
“This study is sweet like sugar for us, because now we have a stent that is dedicated and fitted for the diabetic population,” said Gennaro Sardella, MD, of Sapienza University of Rome.
For Marc Etienne Jolicoeur, MD, an interventional cardiologist associated with Duke University, Durham, N.C., one of the impressive findings was the early separation of the curves in favor of Cre8 EVO. Calling SUGAR a “fantastic trial,” he indicated that the progressive advantage over time reinforced his impression that the difference is real.
However, David Kandzari, MD, director of interventional cardiology, Piedmont Hart Institute, Atlanta, was more circumspect. He did not express any criticisms of the trial, but he called for “a larger evidence base” before declaring the Cre8 EVO device a standard of care for patients with diabetes undergoing PCI.
The SUGAR results were published in the European Heart Journal at the time of presentation at the meeting.
The trial was funded by the Spanish Society of Cardiology. Dr. Romaguera reported financial relationships with Biotronik and Boston Scientific. Dr. Stone, has financial relationships with more than 10 pharmaceutical companies, including those developing devices used in PCI. Dr. Sardella and Dr. Jolicoeur reported no financial relationships relevant to this topic. Dr. Kandzari reported financial relationships with Ablative Solutions and Medtronic.
Superiority shown on TLF endpoint
Superiority shown on TLF endpoint
Designed to show noninferiority for treatment of coronary artery disease (CAD) in patients with diabetes, a head-to-head comparison of contemporary stents ended up showing that one was superior to the for the primary endpoint of target lesion failure (TLF).
In the superiority analysis, the 35% relative reduction in the risk of TLF at 1 year for the Cre8 EVO (Alvimedica) stent relative to the Resolute Onyx (Medtronic) device reached significance, according to Rafael Romaguera, MD, PhD, an interventional cardiologist at the Bellvitge University Hospital, Barcelona.
At 1 year, the rates of TLF were 7.2% and 10.5% for the Cre8 EVO and Resolute Onyx stents, respectively. On the basis of noninferiority, the 3.73% reduction in TLF at 1 year among those receiving the Cre8 EVO device provided a highly significant confirmation of noninferiority (P < .001) and triggered the preplanned superiority analysis.
When the significant advantage on the TLF endpoint (P = .03) was broken down into its components, the Cre8 EVO stent was linked to numerically lower rates of cardiac death (2.1% vs. 2.7%), target vessel MI (5.3% vs. 7.2%), and target lesion revascularization (2.4% vs. 3.9%), according to the SUGAR (Second-Generation Drug-Eluting Stents in Diabetes) trial results presented at the Transcatheter Cardiovascular Therapeutics annual meeting, held virtually and live in Orlando and sponsored by the Cardiovascular Research Foundation.
In a previous study comparing these devices, called the ReCre8 trial, the rates of TLF in an all-comer CAD population were similar at 1 year. When an updated 3-year analysis was presented earlier in 2021 at the Cardiovascular Research Technologies meeting, they remained similar.
Diabetes-centered trial was unmet need
The rationale for conducting a new trial limited to patients with diabetes was based on the greater risk in this population, according to Dr. Romaguera. He cited data that indicate the risk of major adverse cardiac events are about two times higher 2 years after stent implantation in patients with diabetes relative to those without, even when contemporary drug-eluting stents are used.
Both the Cre8 EVO and Resolute Onyx stent are drug eluting and employ contemporary architecture that provides the basis for marketing claims that they are suitable for complex patients; but they have differences.
“There are three features that I think differentiate the Cre8 EVO stent,” Dr. Romaguera reported at the meeting, sponsored by the Cardiovascular Research Foundation.
One is the absence of polymer, which contrasts with the permanent polymer of the Resolute device. This feature affects the dissolution of the anti-inflammatory drug and might be one explanation for the greater protection from ischemic events, according to Dr. Romaguera.
Another is the thickness of the struts, which range from 70 to 80 mm for the Cre8 EVO device and from 92 to 102 mm for the Resolute Onyx device. In experimental studies, strut thickness has been associated with greater risk of thrombus formation, although it is unclear if this modest difference is clinically significant.
Also important, the Cre8 EVO device employs sirolimus for an anti-inflammatory effect, while the Resolute Onyx elutes zotarolimus. Again, experimental evidence suggests a greater anti-inflammatory effect reduces the need for dual-antiplatelet therapy (DAPT); that might offer a relative advantage in patients with an elevated risk of bleeding.
It is not clear whether all of these features contribute to the better results observed in this trial in diabetes patients, but Dr. Romaguera indicated that the lower risk of TLF with Cre8 EVO is not just statistically significant but also clinically meaningful.
In SUGAR, which included 23 centers in Spain, 1,175 patients with confirmed diabetes scheduled for percutaneous intervention (PCI) were randomized to one of the two stents. The study was purposely designed with very few exclusion criteria.
SUGAR trial employed all-comer design
“This was an all-comer design and there was no limitation in regard to clinical presentation, complexity, number of lesions, or other disease features,” said Dr. Romaguera. The major exclusions were a life expectancy of less than 2 years and a contraindication to taking DAPT for at least 1 month,
The patients were almost equally divided between those who had a non–ST-segment elevation MI) and those with chronic coronary artery disease, but patients with a STEMI, representing about 12% of the population, were included. Almost all of the patients (about 95%) had type 2 diabetes; nearly one-third were on insulin at the time of randomization.
According to Dr. Romaguera, “SUGAR is the first powered trial to compare new-generation drug-eluting stents in patients with diabetes,” and he emphasized the all-comer design in supporting its clinical relevance.
Several of those participating in discussion of the trial during the late-breaker session agreed. Although the moderator, Gregg Stone, MD, of the Icahn School of Medicine at Mount Sinai, New York, expressed surprise that the trial “actually demonstrated superiority” given the difficulty of showing a difference between modern stents, he called the findings “remarkable.”
Others seemed to suggest that it would alter their practice.
“This study is sweet like sugar for us, because now we have a stent that is dedicated and fitted for the diabetic population,” said Gennaro Sardella, MD, of Sapienza University of Rome.
For Marc Etienne Jolicoeur, MD, an interventional cardiologist associated with Duke University, Durham, N.C., one of the impressive findings was the early separation of the curves in favor of Cre8 EVO. Calling SUGAR a “fantastic trial,” he indicated that the progressive advantage over time reinforced his impression that the difference is real.
However, David Kandzari, MD, director of interventional cardiology, Piedmont Hart Institute, Atlanta, was more circumspect. He did not express any criticisms of the trial, but he called for “a larger evidence base” before declaring the Cre8 EVO device a standard of care for patients with diabetes undergoing PCI.
The SUGAR results were published in the European Heart Journal at the time of presentation at the meeting.
The trial was funded by the Spanish Society of Cardiology. Dr. Romaguera reported financial relationships with Biotronik and Boston Scientific. Dr. Stone, has financial relationships with more than 10 pharmaceutical companies, including those developing devices used in PCI. Dr. Sardella and Dr. Jolicoeur reported no financial relationships relevant to this topic. Dr. Kandzari reported financial relationships with Ablative Solutions and Medtronic.
Designed to show noninferiority for treatment of coronary artery disease (CAD) in patients with diabetes, a head-to-head comparison of contemporary stents ended up showing that one was superior to the for the primary endpoint of target lesion failure (TLF).
In the superiority analysis, the 35% relative reduction in the risk of TLF at 1 year for the Cre8 EVO (Alvimedica) stent relative to the Resolute Onyx (Medtronic) device reached significance, according to Rafael Romaguera, MD, PhD, an interventional cardiologist at the Bellvitge University Hospital, Barcelona.
At 1 year, the rates of TLF were 7.2% and 10.5% for the Cre8 EVO and Resolute Onyx stents, respectively. On the basis of noninferiority, the 3.73% reduction in TLF at 1 year among those receiving the Cre8 EVO device provided a highly significant confirmation of noninferiority (P < .001) and triggered the preplanned superiority analysis.
When the significant advantage on the TLF endpoint (P = .03) was broken down into its components, the Cre8 EVO stent was linked to numerically lower rates of cardiac death (2.1% vs. 2.7%), target vessel MI (5.3% vs. 7.2%), and target lesion revascularization (2.4% vs. 3.9%), according to the SUGAR (Second-Generation Drug-Eluting Stents in Diabetes) trial results presented at the Transcatheter Cardiovascular Therapeutics annual meeting, held virtually and live in Orlando and sponsored by the Cardiovascular Research Foundation.
In a previous study comparing these devices, called the ReCre8 trial, the rates of TLF in an all-comer CAD population were similar at 1 year. When an updated 3-year analysis was presented earlier in 2021 at the Cardiovascular Research Technologies meeting, they remained similar.
Diabetes-centered trial was unmet need
The rationale for conducting a new trial limited to patients with diabetes was based on the greater risk in this population, according to Dr. Romaguera. He cited data that indicate the risk of major adverse cardiac events are about two times higher 2 years after stent implantation in patients with diabetes relative to those without, even when contemporary drug-eluting stents are used.
Both the Cre8 EVO and Resolute Onyx stent are drug eluting and employ contemporary architecture that provides the basis for marketing claims that they are suitable for complex patients; but they have differences.
“There are three features that I think differentiate the Cre8 EVO stent,” Dr. Romaguera reported at the meeting, sponsored by the Cardiovascular Research Foundation.
One is the absence of polymer, which contrasts with the permanent polymer of the Resolute device. This feature affects the dissolution of the anti-inflammatory drug and might be one explanation for the greater protection from ischemic events, according to Dr. Romaguera.
Another is the thickness of the struts, which range from 70 to 80 mm for the Cre8 EVO device and from 92 to 102 mm for the Resolute Onyx device. In experimental studies, strut thickness has been associated with greater risk of thrombus formation, although it is unclear if this modest difference is clinically significant.
Also important, the Cre8 EVO device employs sirolimus for an anti-inflammatory effect, while the Resolute Onyx elutes zotarolimus. Again, experimental evidence suggests a greater anti-inflammatory effect reduces the need for dual-antiplatelet therapy (DAPT); that might offer a relative advantage in patients with an elevated risk of bleeding.
It is not clear whether all of these features contribute to the better results observed in this trial in diabetes patients, but Dr. Romaguera indicated that the lower risk of TLF with Cre8 EVO is not just statistically significant but also clinically meaningful.
In SUGAR, which included 23 centers in Spain, 1,175 patients with confirmed diabetes scheduled for percutaneous intervention (PCI) were randomized to one of the two stents. The study was purposely designed with very few exclusion criteria.
SUGAR trial employed all-comer design
“This was an all-comer design and there was no limitation in regard to clinical presentation, complexity, number of lesions, or other disease features,” said Dr. Romaguera. The major exclusions were a life expectancy of less than 2 years and a contraindication to taking DAPT for at least 1 month,
The patients were almost equally divided between those who had a non–ST-segment elevation MI) and those with chronic coronary artery disease, but patients with a STEMI, representing about 12% of the population, were included. Almost all of the patients (about 95%) had type 2 diabetes; nearly one-third were on insulin at the time of randomization.
According to Dr. Romaguera, “SUGAR is the first powered trial to compare new-generation drug-eluting stents in patients with diabetes,” and he emphasized the all-comer design in supporting its clinical relevance.
Several of those participating in discussion of the trial during the late-breaker session agreed. Although the moderator, Gregg Stone, MD, of the Icahn School of Medicine at Mount Sinai, New York, expressed surprise that the trial “actually demonstrated superiority” given the difficulty of showing a difference between modern stents, he called the findings “remarkable.”
Others seemed to suggest that it would alter their practice.
“This study is sweet like sugar for us, because now we have a stent that is dedicated and fitted for the diabetic population,” said Gennaro Sardella, MD, of Sapienza University of Rome.
For Marc Etienne Jolicoeur, MD, an interventional cardiologist associated with Duke University, Durham, N.C., one of the impressive findings was the early separation of the curves in favor of Cre8 EVO. Calling SUGAR a “fantastic trial,” he indicated that the progressive advantage over time reinforced his impression that the difference is real.
However, David Kandzari, MD, director of interventional cardiology, Piedmont Hart Institute, Atlanta, was more circumspect. He did not express any criticisms of the trial, but he called for “a larger evidence base” before declaring the Cre8 EVO device a standard of care for patients with diabetes undergoing PCI.
The SUGAR results were published in the European Heart Journal at the time of presentation at the meeting.
The trial was funded by the Spanish Society of Cardiology. Dr. Romaguera reported financial relationships with Biotronik and Boston Scientific. Dr. Stone, has financial relationships with more than 10 pharmaceutical companies, including those developing devices used in PCI. Dr. Sardella and Dr. Jolicoeur reported no financial relationships relevant to this topic. Dr. Kandzari reported financial relationships with Ablative Solutions and Medtronic.
FROM TCT 2021
COVID-19 has brought more complex, longer office visits
Evidence of this came from the latest Primary Care Collaborative (PCC) survey, which found that primary care clinicians are seeing more complex patients requiring longer appointments in the wake of COVID-19.
The PCC with the Larry A. Green Center regularly surveys primary care clinicians. This round of questions came August 14-17 and included 1,263 respondents from 49 states, the District of Columbia, and two territories.
More than 7 in 10 (71%) respondents said their patients are more complex and nearly the same percentage said appointments are taking more time.
Ann Greiner, president and CEO of the PCC, said in an interview that 55% of respondents reported that clinicians are struggling to keep up with pent-up demand after patients have delayed or canceled care. Sixty-five percent in the survey said they had seen a rise in children’s mental health issues, and 58% said they were unsure how to help their patients with long COVID.
In addition, primary care clinicians are having repeated conversations with patients on why they should get a vaccine and which one.
“I think that’s adding to the complexity. There is a lot going on here with patient trust,” Ms. Greiner said.
‘We’re going to be playing catch-up’
Jacqueline Fincher, MD, an internist in Thompson, Ga., said in an interview that appointments have gotten longer and more complex in the wake of the pandemic – “no question.”
The immediate past president of the American College of Physicians is seeing patients with chronic disease that has gone untreated for sometimes a year or more, she said.
“Their blood pressure was not under good control, they were under more stress, their sugars were up and weren’t being followed as closely for conditions such as congestive heart failure,” she said.
Dr. Fincher, who works in a rural practice 40 miles from Augusta, Ga., with her physician husband and two other physicians, said patients are ready to come back in, “but I don’t have enough slots for them.”
She said she prioritizes what to help patients with first and schedules the next tier for the next appointment, but added, “honestly, over the next 2 years we’re going to be playing catch-up.”
At the same time, the CDC has estimated that 45% of U.S. adults are at increased risk for complications from COVID-19 because of cardiovascular disease, diabetes, respiratory disease, hypertension, or cancer. Rates ranged from 19.8% for people 18-29 years old to 80.7% for people over 80 years of age.
Long COVID could overwhelm existing health care capacity
Primary care physicians are also having to diagnose sometimes “invisible” symptoms after people have recovered from acute COVID-19 infection. Diagnosing takes intent listening to patients who describe symptoms that tests can’t confirm.
As this news organization has previously reported, half of COVID-19 survivors report postacute sequelae of COVID-19 (PASC) lasting longer than 6 months.
“These long-term PASC effects occur on a scale that could overwhelm existing health care capacity, particularly in low- and middle-income countries,” the authors wrote.
Anxiety, depression ‘have gone off the charts’
Danielle Loeb, MD, MPH, associate professor of internal medicine at the University of Colorado in Denver, who studies complexity in primary care, said in the wake of COVID-19, more patients have developed “new, serious anxiety.”
“That got extremely exacerbated during the pandemic. Anxiety and depression have gone off the charts,” said Dr. Loeb, who prefers the pronoun “they.”
Dr. Loeb cares for a large number of transgender patients. As offices reopen, some patients are having trouble reintegrating into the workplace and resuming social contacts. The primary care doctor says appointments can get longer because of the need to complete tasks, such as filling out forms for Family Medical Leave Act for those not yet ready to return to work.
COVID-19–related fears are keeping many patients from coming into the office, Dr. Loeb said, either from fear of exposure or because they have mental health issues that keep them from feeling safe leaving the house.
“That really affects my ability to care for them,” they said.
Loss of employment in the pandemic or fear of job loss and subsequent changing of insurance has complicated primary care in terms of treatment and administrative tasks, according to Dr. Loeb.
To help treat patients with acute mental health issues and manage other patients, Dr. Loeb’s practice has brought in a social worker and a therapist.
Team-based care is key in the survival of primary care practices, though providing that is difficult in the smaller clinics because of the critical mass of patients needed to make it viable, they said.
“It’s the only answer. It’s the only way you don’t drown,” Dr. Loeb added. “I’m not drowning, and I credit that to my clinic having the help to support the mental health piece of things.”
Rethinking workflow
Tricia McGinnis, MPP, MPH, executive vice president of the nonprofit Center for Health Care Strategies (CHCS) says complexity has forced rethinking workflow.
“A lot of the trends we’re seeing in primary care were there pre-COVID, but COVID has exacerbated those trends,” she said in an interview.
“The good news ... is that it was already becoming clear that primary care needed to provide basic mental health services and integrate with behavioral health. It had also become clear that effective primary care needed to address social issues that keep patients from accessing health care,” she said.
Expanding care teams, as Dr. Loeb mentioned, is a key strategy, according to Ms. McGinnis. Potential teams would include the clinical staff, but also social workers and community health workers – people who come from the community primary care is serving who can help build trust with patients and connect the patient to the primary care team.
“There’s a lot that needs to happen that the clinician doesn’t need to do,” she said.
Telehealth can be a big factor in coordinating the team, Ms. McGinnis added.
“It’s thinking less about who’s doing the work, but more about the work that needs to be done to keep people healthy. Then let’s think about the type of workers best suited to perform those tasks,” she said.
As for reimbursing more complex care, population-based, up-front capitated payments linked to high-quality care and better outcomes will need to replace fee-for-service models, according to Ms. McGinnis.
That will provide reliable incomes for primary care offices, but also flexibility in how each patient with different levels of complexity is managed, she said.
Ms. Greiner, Dr. Fincher, Dr. Loeb, and Ms. McGinnis have no relevant financial relationships.
Evidence of this came from the latest Primary Care Collaborative (PCC) survey, which found that primary care clinicians are seeing more complex patients requiring longer appointments in the wake of COVID-19.
The PCC with the Larry A. Green Center regularly surveys primary care clinicians. This round of questions came August 14-17 and included 1,263 respondents from 49 states, the District of Columbia, and two territories.
More than 7 in 10 (71%) respondents said their patients are more complex and nearly the same percentage said appointments are taking more time.
Ann Greiner, president and CEO of the PCC, said in an interview that 55% of respondents reported that clinicians are struggling to keep up with pent-up demand after patients have delayed or canceled care. Sixty-five percent in the survey said they had seen a rise in children’s mental health issues, and 58% said they were unsure how to help their patients with long COVID.
In addition, primary care clinicians are having repeated conversations with patients on why they should get a vaccine and which one.
“I think that’s adding to the complexity. There is a lot going on here with patient trust,” Ms. Greiner said.
‘We’re going to be playing catch-up’
Jacqueline Fincher, MD, an internist in Thompson, Ga., said in an interview that appointments have gotten longer and more complex in the wake of the pandemic – “no question.”
The immediate past president of the American College of Physicians is seeing patients with chronic disease that has gone untreated for sometimes a year or more, she said.
“Their blood pressure was not under good control, they were under more stress, their sugars were up and weren’t being followed as closely for conditions such as congestive heart failure,” she said.
Dr. Fincher, who works in a rural practice 40 miles from Augusta, Ga., with her physician husband and two other physicians, said patients are ready to come back in, “but I don’t have enough slots for them.”
She said she prioritizes what to help patients with first and schedules the next tier for the next appointment, but added, “honestly, over the next 2 years we’re going to be playing catch-up.”
At the same time, the CDC has estimated that 45% of U.S. adults are at increased risk for complications from COVID-19 because of cardiovascular disease, diabetes, respiratory disease, hypertension, or cancer. Rates ranged from 19.8% for people 18-29 years old to 80.7% for people over 80 years of age.
Long COVID could overwhelm existing health care capacity
Primary care physicians are also having to diagnose sometimes “invisible” symptoms after people have recovered from acute COVID-19 infection. Diagnosing takes intent listening to patients who describe symptoms that tests can’t confirm.
As this news organization has previously reported, half of COVID-19 survivors report postacute sequelae of COVID-19 (PASC) lasting longer than 6 months.
“These long-term PASC effects occur on a scale that could overwhelm existing health care capacity, particularly in low- and middle-income countries,” the authors wrote.
Anxiety, depression ‘have gone off the charts’
Danielle Loeb, MD, MPH, associate professor of internal medicine at the University of Colorado in Denver, who studies complexity in primary care, said in the wake of COVID-19, more patients have developed “new, serious anxiety.”
“That got extremely exacerbated during the pandemic. Anxiety and depression have gone off the charts,” said Dr. Loeb, who prefers the pronoun “they.”
Dr. Loeb cares for a large number of transgender patients. As offices reopen, some patients are having trouble reintegrating into the workplace and resuming social contacts. The primary care doctor says appointments can get longer because of the need to complete tasks, such as filling out forms for Family Medical Leave Act for those not yet ready to return to work.
COVID-19–related fears are keeping many patients from coming into the office, Dr. Loeb said, either from fear of exposure or because they have mental health issues that keep them from feeling safe leaving the house.
“That really affects my ability to care for them,” they said.
Loss of employment in the pandemic or fear of job loss and subsequent changing of insurance has complicated primary care in terms of treatment and administrative tasks, according to Dr. Loeb.
To help treat patients with acute mental health issues and manage other patients, Dr. Loeb’s practice has brought in a social worker and a therapist.
Team-based care is key in the survival of primary care practices, though providing that is difficult in the smaller clinics because of the critical mass of patients needed to make it viable, they said.
“It’s the only answer. It’s the only way you don’t drown,” Dr. Loeb added. “I’m not drowning, and I credit that to my clinic having the help to support the mental health piece of things.”
Rethinking workflow
Tricia McGinnis, MPP, MPH, executive vice president of the nonprofit Center for Health Care Strategies (CHCS) says complexity has forced rethinking workflow.
“A lot of the trends we’re seeing in primary care were there pre-COVID, but COVID has exacerbated those trends,” she said in an interview.
“The good news ... is that it was already becoming clear that primary care needed to provide basic mental health services and integrate with behavioral health. It had also become clear that effective primary care needed to address social issues that keep patients from accessing health care,” she said.
Expanding care teams, as Dr. Loeb mentioned, is a key strategy, according to Ms. McGinnis. Potential teams would include the clinical staff, but also social workers and community health workers – people who come from the community primary care is serving who can help build trust with patients and connect the patient to the primary care team.
“There’s a lot that needs to happen that the clinician doesn’t need to do,” she said.
Telehealth can be a big factor in coordinating the team, Ms. McGinnis added.
“It’s thinking less about who’s doing the work, but more about the work that needs to be done to keep people healthy. Then let’s think about the type of workers best suited to perform those tasks,” she said.
As for reimbursing more complex care, population-based, up-front capitated payments linked to high-quality care and better outcomes will need to replace fee-for-service models, according to Ms. McGinnis.
That will provide reliable incomes for primary care offices, but also flexibility in how each patient with different levels of complexity is managed, she said.
Ms. Greiner, Dr. Fincher, Dr. Loeb, and Ms. McGinnis have no relevant financial relationships.
Evidence of this came from the latest Primary Care Collaborative (PCC) survey, which found that primary care clinicians are seeing more complex patients requiring longer appointments in the wake of COVID-19.
The PCC with the Larry A. Green Center regularly surveys primary care clinicians. This round of questions came August 14-17 and included 1,263 respondents from 49 states, the District of Columbia, and two territories.
More than 7 in 10 (71%) respondents said their patients are more complex and nearly the same percentage said appointments are taking more time.
Ann Greiner, president and CEO of the PCC, said in an interview that 55% of respondents reported that clinicians are struggling to keep up with pent-up demand after patients have delayed or canceled care. Sixty-five percent in the survey said they had seen a rise in children’s mental health issues, and 58% said they were unsure how to help their patients with long COVID.
In addition, primary care clinicians are having repeated conversations with patients on why they should get a vaccine and which one.
“I think that’s adding to the complexity. There is a lot going on here with patient trust,” Ms. Greiner said.
‘We’re going to be playing catch-up’
Jacqueline Fincher, MD, an internist in Thompson, Ga., said in an interview that appointments have gotten longer and more complex in the wake of the pandemic – “no question.”
The immediate past president of the American College of Physicians is seeing patients with chronic disease that has gone untreated for sometimes a year or more, she said.
“Their blood pressure was not under good control, they were under more stress, their sugars were up and weren’t being followed as closely for conditions such as congestive heart failure,” she said.
Dr. Fincher, who works in a rural practice 40 miles from Augusta, Ga., with her physician husband and two other physicians, said patients are ready to come back in, “but I don’t have enough slots for them.”
She said she prioritizes what to help patients with first and schedules the next tier for the next appointment, but added, “honestly, over the next 2 years we’re going to be playing catch-up.”
At the same time, the CDC has estimated that 45% of U.S. adults are at increased risk for complications from COVID-19 because of cardiovascular disease, diabetes, respiratory disease, hypertension, or cancer. Rates ranged from 19.8% for people 18-29 years old to 80.7% for people over 80 years of age.
Long COVID could overwhelm existing health care capacity
Primary care physicians are also having to diagnose sometimes “invisible” symptoms after people have recovered from acute COVID-19 infection. Diagnosing takes intent listening to patients who describe symptoms that tests can’t confirm.
As this news organization has previously reported, half of COVID-19 survivors report postacute sequelae of COVID-19 (PASC) lasting longer than 6 months.
“These long-term PASC effects occur on a scale that could overwhelm existing health care capacity, particularly in low- and middle-income countries,” the authors wrote.
Anxiety, depression ‘have gone off the charts’
Danielle Loeb, MD, MPH, associate professor of internal medicine at the University of Colorado in Denver, who studies complexity in primary care, said in the wake of COVID-19, more patients have developed “new, serious anxiety.”
“That got extremely exacerbated during the pandemic. Anxiety and depression have gone off the charts,” said Dr. Loeb, who prefers the pronoun “they.”
Dr. Loeb cares for a large number of transgender patients. As offices reopen, some patients are having trouble reintegrating into the workplace and resuming social contacts. The primary care doctor says appointments can get longer because of the need to complete tasks, such as filling out forms for Family Medical Leave Act for those not yet ready to return to work.
COVID-19–related fears are keeping many patients from coming into the office, Dr. Loeb said, either from fear of exposure or because they have mental health issues that keep them from feeling safe leaving the house.
“That really affects my ability to care for them,” they said.
Loss of employment in the pandemic or fear of job loss and subsequent changing of insurance has complicated primary care in terms of treatment and administrative tasks, according to Dr. Loeb.
To help treat patients with acute mental health issues and manage other patients, Dr. Loeb’s practice has brought in a social worker and a therapist.
Team-based care is key in the survival of primary care practices, though providing that is difficult in the smaller clinics because of the critical mass of patients needed to make it viable, they said.
“It’s the only answer. It’s the only way you don’t drown,” Dr. Loeb added. “I’m not drowning, and I credit that to my clinic having the help to support the mental health piece of things.”
Rethinking workflow
Tricia McGinnis, MPP, MPH, executive vice president of the nonprofit Center for Health Care Strategies (CHCS) says complexity has forced rethinking workflow.
“A lot of the trends we’re seeing in primary care were there pre-COVID, but COVID has exacerbated those trends,” she said in an interview.
“The good news ... is that it was already becoming clear that primary care needed to provide basic mental health services and integrate with behavioral health. It had also become clear that effective primary care needed to address social issues that keep patients from accessing health care,” she said.
Expanding care teams, as Dr. Loeb mentioned, is a key strategy, according to Ms. McGinnis. Potential teams would include the clinical staff, but also social workers and community health workers – people who come from the community primary care is serving who can help build trust with patients and connect the patient to the primary care team.
“There’s a lot that needs to happen that the clinician doesn’t need to do,” she said.
Telehealth can be a big factor in coordinating the team, Ms. McGinnis added.
“It’s thinking less about who’s doing the work, but more about the work that needs to be done to keep people healthy. Then let’s think about the type of workers best suited to perform those tasks,” she said.
As for reimbursing more complex care, population-based, up-front capitated payments linked to high-quality care and better outcomes will need to replace fee-for-service models, according to Ms. McGinnis.
That will provide reliable incomes for primary care offices, but also flexibility in how each patient with different levels of complexity is managed, she said.
Ms. Greiner, Dr. Fincher, Dr. Loeb, and Ms. McGinnis have no relevant financial relationships.
New single-button blood glucose monitor available in U.S.
The POGO Automatic Blood Glucose Monitoring System (Intuity Medical) has been cleared by the U.S. Food and Drug Administration for people with diabetes aged 13 years and older.
It contains a 10-test cartridge, and once loaded and the monitor is turned on, the user only has to press their finger on a button to activate POGO Automatic, which then does all the work of lancing and blood collection, followed by a 4-second countdown and a result. Users only need to carry the monitor and not separate lancets or strips.
An app called Patterns is available for iOS and Android that allows the results from the device to automatically sync via Bluetooth. It visually presents glucose trends and enables data sharing with health care providers.
“We know that people with diabetes are more effective at managing their diabetes when they regularly check their blood glucose and use the information to take action,” said Daniel Einhorn, MD, medical director of Scripps Whittier Diabetes Institute, president of Diabetes and Endocrine Associates, and chairperson of the Intuity Medical Scientific Advisory Board, in a company statement.
“My patients and millions of others with diabetes have struggled for decades with the burden of checking their glucose because it’s complicated, there’s a lot to carry around, and it’s intrusive,” he added. “What they’ve needed is a simple, quick, and truly discreet way to check their blood glucose, so they’ll actually do it.”
How does POGO compare with CGM?
Continuous glucose monitors (CGMs), such as the Abbott FreeStyle Libre, Dexcom G6, and Eversense implant, are increasingly employed by people with type 1 diabetes, and some with type 2 diabetes, to keep a close eye on their blood glucose levels.
Asked how the POGO device compares with CGM systems, Intuity Chief Commercial Officer Dean Zikria said: “While [CGM] is certainly an important option for a subset of people with diabetes, CGM is a very different technology, requiring a user to wear a sensor and transmitter on their body.”
“Patients also need to obtain a prescription in order to use CGM.”
“Conversely, POGO Automatic is available with or without a prescription. POGO Automatic also gives people who do not want to wear a device on their body a new choice other than traditional blood glucose monitoring,” Mr. Zikria added.
The POGO system is available at U.S. pharmacies, including CVS and Walgreens, and can also be purchased online.
The device costs $68 from the company website and a pack of 5 cartridges (each containing 10 tests, with an aim of people performing 1-2 tests per day) costs a further $32 as a one-off, or $32 per month as a subscription.
The product is also eligible for purchase using Flexible Spending Accounts and Health Savings Accounts.
A version of this article first appeared on Medscape.com.
The POGO Automatic Blood Glucose Monitoring System (Intuity Medical) has been cleared by the U.S. Food and Drug Administration for people with diabetes aged 13 years and older.
It contains a 10-test cartridge, and once loaded and the monitor is turned on, the user only has to press their finger on a button to activate POGO Automatic, which then does all the work of lancing and blood collection, followed by a 4-second countdown and a result. Users only need to carry the monitor and not separate lancets or strips.
An app called Patterns is available for iOS and Android that allows the results from the device to automatically sync via Bluetooth. It visually presents glucose trends and enables data sharing with health care providers.
“We know that people with diabetes are more effective at managing their diabetes when they regularly check their blood glucose and use the information to take action,” said Daniel Einhorn, MD, medical director of Scripps Whittier Diabetes Institute, president of Diabetes and Endocrine Associates, and chairperson of the Intuity Medical Scientific Advisory Board, in a company statement.
“My patients and millions of others with diabetes have struggled for decades with the burden of checking their glucose because it’s complicated, there’s a lot to carry around, and it’s intrusive,” he added. “What they’ve needed is a simple, quick, and truly discreet way to check their blood glucose, so they’ll actually do it.”
How does POGO compare with CGM?
Continuous glucose monitors (CGMs), such as the Abbott FreeStyle Libre, Dexcom G6, and Eversense implant, are increasingly employed by people with type 1 diabetes, and some with type 2 diabetes, to keep a close eye on their blood glucose levels.
Asked how the POGO device compares with CGM systems, Intuity Chief Commercial Officer Dean Zikria said: “While [CGM] is certainly an important option for a subset of people with diabetes, CGM is a very different technology, requiring a user to wear a sensor and transmitter on their body.”
“Patients also need to obtain a prescription in order to use CGM.”
“Conversely, POGO Automatic is available with or without a prescription. POGO Automatic also gives people who do not want to wear a device on their body a new choice other than traditional blood glucose monitoring,” Mr. Zikria added.
The POGO system is available at U.S. pharmacies, including CVS and Walgreens, and can also be purchased online.
The device costs $68 from the company website and a pack of 5 cartridges (each containing 10 tests, with an aim of people performing 1-2 tests per day) costs a further $32 as a one-off, or $32 per month as a subscription.
The product is also eligible for purchase using Flexible Spending Accounts and Health Savings Accounts.
A version of this article first appeared on Medscape.com.
The POGO Automatic Blood Glucose Monitoring System (Intuity Medical) has been cleared by the U.S. Food and Drug Administration for people with diabetes aged 13 years and older.
It contains a 10-test cartridge, and once loaded and the monitor is turned on, the user only has to press their finger on a button to activate POGO Automatic, which then does all the work of lancing and blood collection, followed by a 4-second countdown and a result. Users only need to carry the monitor and not separate lancets or strips.
An app called Patterns is available for iOS and Android that allows the results from the device to automatically sync via Bluetooth. It visually presents glucose trends and enables data sharing with health care providers.
“We know that people with diabetes are more effective at managing their diabetes when they regularly check their blood glucose and use the information to take action,” said Daniel Einhorn, MD, medical director of Scripps Whittier Diabetes Institute, president of Diabetes and Endocrine Associates, and chairperson of the Intuity Medical Scientific Advisory Board, in a company statement.
“My patients and millions of others with diabetes have struggled for decades with the burden of checking their glucose because it’s complicated, there’s a lot to carry around, and it’s intrusive,” he added. “What they’ve needed is a simple, quick, and truly discreet way to check their blood glucose, so they’ll actually do it.”
How does POGO compare with CGM?
Continuous glucose monitors (CGMs), such as the Abbott FreeStyle Libre, Dexcom G6, and Eversense implant, are increasingly employed by people with type 1 diabetes, and some with type 2 diabetes, to keep a close eye on their blood glucose levels.
Asked how the POGO device compares with CGM systems, Intuity Chief Commercial Officer Dean Zikria said: “While [CGM] is certainly an important option for a subset of people with diabetes, CGM is a very different technology, requiring a user to wear a sensor and transmitter on their body.”
“Patients also need to obtain a prescription in order to use CGM.”
“Conversely, POGO Automatic is available with or without a prescription. POGO Automatic also gives people who do not want to wear a device on their body a new choice other than traditional blood glucose monitoring,” Mr. Zikria added.
The POGO system is available at U.S. pharmacies, including CVS and Walgreens, and can also be purchased online.
The device costs $68 from the company website and a pack of 5 cartridges (each containing 10 tests, with an aim of people performing 1-2 tests per day) costs a further $32 as a one-off, or $32 per month as a subscription.
The product is also eligible for purchase using Flexible Spending Accounts and Health Savings Accounts.
A version of this article first appeared on Medscape.com.