User login
EMPEROR-Preserved findings confirmed in ‘true’ HFpEF patients
Main results from the landmark EMPEROR-Preserved trial, reported in August, established for the first time that treatment with a drug, the sodium-glucose cotransporter 2 inhibitor empagliflozin, could clearly benefit patients with heart failure with preserved ejection fraction (HFpEF).
The only caveat was that EMPEROR-Preserved enrolled patients with a left ventricular ejection fraction of at least 41%, while “true” HFpEF means patients with heart failure and an LVEF of at least 50%, according to recent definitions. About one-third of the 5,988 patients enrolled in EMPEROR-Preserved had an LVEF of 41%-49%, heart failure with mildly reduced ejection fraction.
Secondary analysis from the EMPEROR-Preserved trial has now resolved this ambiguity by showing that, among the 4,005 patients (67%) enrolled in the trial with an LVEF of at least 50%, treatment with empagliflozin (Jardiance) reduced the study’s primary endpoint – cardiovascular death or first hospitalization for heart failure – by a significant 17%, relative to patients who received placebo, dismissing any doubt about the relevance of the overall finding to the subgroup of patients with unmitigated HFpEF.
“This is the first large-scale trial to document meaningful and significant improvements associated with drug therapy in patients with ‘true’ HFpEF,” Stefan D. Anker, MD, said in presenting the results at the American Heart Association scientific sessions.
Streamlining heart failure treatment
The demonstration that empagliflozin is an effective – and safe – treatment for patients with HFpEF not only provides a new treatment for a disorder that until now had no evidence-based intervention, but also streamlines the management approach for treating patients with heart failure with an agent from empagliflozin’s class, the SGLT2 inhibitors, commented Mary Norine Walsh, MD, medical director of the heart failure and cardiac transplantation programs at Ascension St. Vincent Heart Center in Indianapolis.
That’s because empagliflozin has shown significant and consistent benefit across essentially the full range of LVEFs seen in patients with heart failure based on its performance in EMPEROR-Preserved as well as in a mirror-image trial, EMPEROR-Reduced, run in patients with heart failure with reduced ejection fraction.
“Clinicians do not need to stop and assess LVEF with echocardiography or other imaging before they decide on how to treat heart failure patients” with an SGLT2 inhibitor, noted Dr. Walsh, a designated discussant for the report. “Clinicians who are busy can now refer less to LVEF than to the patient’s phenotype.”
Treatment prevents hospitalization for heart failure
The more-detailed data reported by Dr. Anker also strengthened the case that the benefit from empagliflozin in patients with an LVEF of at least 50% mostly came from a reduction in hospitalizations for heart failure (HHF), which dropped following start of empagliflozin treatment by a relative 22%, compared with placebo for first HHF, a significant decline, and by a relative 17% for total HHF, a reduction that missed significance in this secondary analysis. The other half of the primary endpoint, cardiovascular death, declined by a nonsignificant 11% with empagliflozin treatment, compared with placebo in patients with clear-cut HFpEF.
The significant reduction in first HHF is, by itself, sufficient reason to use empagliflozin (or possibly a different SGLT2 inhibitor) in patients with HFpEF, maintained Clyde W. Yancy, MD, professor and chief of cardiology at Northwestern Medicine in Chicago.
“Attenuated HHF is a meaningful outcome,” stressed Dr. Yancy, also a discussant for the study. “This is the first time we’ve had evidence supporting that we can change the natural history of patients with HFpEF. While we still need to find interventions that save lives, we cannot overlook that this treatment can improve morbidity, and we cannot overlook that patient quality of life is better.”
Further benefits in patients with an LVEF of at least 50%
Dr. Anker, professor of cardiology and metabolism at Charité Medical University in Berlin, also reported results from several other analyses that further defined the effect of empagliflozin on clinical outcomes of patients with “true” HFpEF:
- The impact of empagliflozin, compared with placebo, for reducing both the study’s combined, primary outcome as well as total HHF was statistically consistent across all strata of LVEF, from 50% to greater than 70%. However, both outcome measures also showed a puzzling loss of benefit among patients with an LVEF of 65%-69%. In prior reports, a researcher on the EMPEROR-Preserved team, Milton Packer, MD, speculated that some patients in this LVEF stratum might not actually have had heart failure but instead had a different disorder that mimicked heart failure in clinical presentation, such as atrial fibrillation.
- Patients’ quality of life as measured by the Kansas City Cardiomyopathy Questionnaire showed a consistent benefit from empagliflozin treatment, compared with placebo, both in patients with an LVEF of at least 50% as well as in those with an LVEF of 41%-49%. In both subgroups the adjusted mean difference from placebo was significant and about 1.5 points.
- Patients showed a significant improvement in average New York Heart Association functional class while on treatment, and a strong trend toward less deterioration in functional class while on treatment.
- Deterioration of renal function on treatment slowed by an average 1.24 mL/min per 1.73 m2 per year in patients on empagliflozin, compared with placebo, in the subgroup with an LVEF of at least 50%.
Dr. Anker also reported the primary outcome and component results for the subgroup of patients with a baseline LVEF of 41%-49%. These patients had what looked like a “bigger magnitude” of effect from treatment, he noted, showing a significant 29% relative decline in the primary endpoint, compared with placebo-treated patients, and a significant 42% relative drop in first HHF and a significant 43% relative decline in total HHF, compared with placebo.
The primary analysis from EMPEROR-Preserved, which included all 5,988 randomized patients with heart failure and an LVEF of 41% or greater, showed a significant reduction in the combined, primary endpoint with empagliflozin treatment of 21%, compared with control patients during a median follow-up of about 26 months. The absolute rate reduction of the combined primary endpoint was 3.3% during 26-months’ follow-up. Statistical tests have shown no heterogeneity of this effect by diabetes status (49% of patients had diabetes), nor by renal function down to an estimated glomerular filtration rate at entry as low as 20 mL/min per 1.73 m2.
EMPEROR-Preserved was sponsored by Boehringer Ingelheim and Lilly, the two companies that market empagliflozin (Jardiance). Dr. Anker has been a consultant to Boehringer Ingelheim as well as to Abbott Vascular, Bayer, Brahms, Cardiac Dimensions, Cordio, Novartis, Servier, and Vifor. Dr. Walsh and Dr. Yancy had no disclosures.
Main results from the landmark EMPEROR-Preserved trial, reported in August, established for the first time that treatment with a drug, the sodium-glucose cotransporter 2 inhibitor empagliflozin, could clearly benefit patients with heart failure with preserved ejection fraction (HFpEF).
The only caveat was that EMPEROR-Preserved enrolled patients with a left ventricular ejection fraction of at least 41%, while “true” HFpEF means patients with heart failure and an LVEF of at least 50%, according to recent definitions. About one-third of the 5,988 patients enrolled in EMPEROR-Preserved had an LVEF of 41%-49%, heart failure with mildly reduced ejection fraction.
Secondary analysis from the EMPEROR-Preserved trial has now resolved this ambiguity by showing that, among the 4,005 patients (67%) enrolled in the trial with an LVEF of at least 50%, treatment with empagliflozin (Jardiance) reduced the study’s primary endpoint – cardiovascular death or first hospitalization for heart failure – by a significant 17%, relative to patients who received placebo, dismissing any doubt about the relevance of the overall finding to the subgroup of patients with unmitigated HFpEF.
“This is the first large-scale trial to document meaningful and significant improvements associated with drug therapy in patients with ‘true’ HFpEF,” Stefan D. Anker, MD, said in presenting the results at the American Heart Association scientific sessions.
Streamlining heart failure treatment
The demonstration that empagliflozin is an effective – and safe – treatment for patients with HFpEF not only provides a new treatment for a disorder that until now had no evidence-based intervention, but also streamlines the management approach for treating patients with heart failure with an agent from empagliflozin’s class, the SGLT2 inhibitors, commented Mary Norine Walsh, MD, medical director of the heart failure and cardiac transplantation programs at Ascension St. Vincent Heart Center in Indianapolis.
That’s because empagliflozin has shown significant and consistent benefit across essentially the full range of LVEFs seen in patients with heart failure based on its performance in EMPEROR-Preserved as well as in a mirror-image trial, EMPEROR-Reduced, run in patients with heart failure with reduced ejection fraction.
“Clinicians do not need to stop and assess LVEF with echocardiography or other imaging before they decide on how to treat heart failure patients” with an SGLT2 inhibitor, noted Dr. Walsh, a designated discussant for the report. “Clinicians who are busy can now refer less to LVEF than to the patient’s phenotype.”
Treatment prevents hospitalization for heart failure
The more-detailed data reported by Dr. Anker also strengthened the case that the benefit from empagliflozin in patients with an LVEF of at least 50% mostly came from a reduction in hospitalizations for heart failure (HHF), which dropped following start of empagliflozin treatment by a relative 22%, compared with placebo for first HHF, a significant decline, and by a relative 17% for total HHF, a reduction that missed significance in this secondary analysis. The other half of the primary endpoint, cardiovascular death, declined by a nonsignificant 11% with empagliflozin treatment, compared with placebo in patients with clear-cut HFpEF.
The significant reduction in first HHF is, by itself, sufficient reason to use empagliflozin (or possibly a different SGLT2 inhibitor) in patients with HFpEF, maintained Clyde W. Yancy, MD, professor and chief of cardiology at Northwestern Medicine in Chicago.
“Attenuated HHF is a meaningful outcome,” stressed Dr. Yancy, also a discussant for the study. “This is the first time we’ve had evidence supporting that we can change the natural history of patients with HFpEF. While we still need to find interventions that save lives, we cannot overlook that this treatment can improve morbidity, and we cannot overlook that patient quality of life is better.”
Further benefits in patients with an LVEF of at least 50%
Dr. Anker, professor of cardiology and metabolism at Charité Medical University in Berlin, also reported results from several other analyses that further defined the effect of empagliflozin on clinical outcomes of patients with “true” HFpEF:
- The impact of empagliflozin, compared with placebo, for reducing both the study’s combined, primary outcome as well as total HHF was statistically consistent across all strata of LVEF, from 50% to greater than 70%. However, both outcome measures also showed a puzzling loss of benefit among patients with an LVEF of 65%-69%. In prior reports, a researcher on the EMPEROR-Preserved team, Milton Packer, MD, speculated that some patients in this LVEF stratum might not actually have had heart failure but instead had a different disorder that mimicked heart failure in clinical presentation, such as atrial fibrillation.
- Patients’ quality of life as measured by the Kansas City Cardiomyopathy Questionnaire showed a consistent benefit from empagliflozin treatment, compared with placebo, both in patients with an LVEF of at least 50% as well as in those with an LVEF of 41%-49%. In both subgroups the adjusted mean difference from placebo was significant and about 1.5 points.
- Patients showed a significant improvement in average New York Heart Association functional class while on treatment, and a strong trend toward less deterioration in functional class while on treatment.
- Deterioration of renal function on treatment slowed by an average 1.24 mL/min per 1.73 m2 per year in patients on empagliflozin, compared with placebo, in the subgroup with an LVEF of at least 50%.
Dr. Anker also reported the primary outcome and component results for the subgroup of patients with a baseline LVEF of 41%-49%. These patients had what looked like a “bigger magnitude” of effect from treatment, he noted, showing a significant 29% relative decline in the primary endpoint, compared with placebo-treated patients, and a significant 42% relative drop in first HHF and a significant 43% relative decline in total HHF, compared with placebo.
The primary analysis from EMPEROR-Preserved, which included all 5,988 randomized patients with heart failure and an LVEF of 41% or greater, showed a significant reduction in the combined, primary endpoint with empagliflozin treatment of 21%, compared with control patients during a median follow-up of about 26 months. The absolute rate reduction of the combined primary endpoint was 3.3% during 26-months’ follow-up. Statistical tests have shown no heterogeneity of this effect by diabetes status (49% of patients had diabetes), nor by renal function down to an estimated glomerular filtration rate at entry as low as 20 mL/min per 1.73 m2.
EMPEROR-Preserved was sponsored by Boehringer Ingelheim and Lilly, the two companies that market empagliflozin (Jardiance). Dr. Anker has been a consultant to Boehringer Ingelheim as well as to Abbott Vascular, Bayer, Brahms, Cardiac Dimensions, Cordio, Novartis, Servier, and Vifor. Dr. Walsh and Dr. Yancy had no disclosures.
Main results from the landmark EMPEROR-Preserved trial, reported in August, established for the first time that treatment with a drug, the sodium-glucose cotransporter 2 inhibitor empagliflozin, could clearly benefit patients with heart failure with preserved ejection fraction (HFpEF).
The only caveat was that EMPEROR-Preserved enrolled patients with a left ventricular ejection fraction of at least 41%, while “true” HFpEF means patients with heart failure and an LVEF of at least 50%, according to recent definitions. About one-third of the 5,988 patients enrolled in EMPEROR-Preserved had an LVEF of 41%-49%, heart failure with mildly reduced ejection fraction.
Secondary analysis from the EMPEROR-Preserved trial has now resolved this ambiguity by showing that, among the 4,005 patients (67%) enrolled in the trial with an LVEF of at least 50%, treatment with empagliflozin (Jardiance) reduced the study’s primary endpoint – cardiovascular death or first hospitalization for heart failure – by a significant 17%, relative to patients who received placebo, dismissing any doubt about the relevance of the overall finding to the subgroup of patients with unmitigated HFpEF.
“This is the first large-scale trial to document meaningful and significant improvements associated with drug therapy in patients with ‘true’ HFpEF,” Stefan D. Anker, MD, said in presenting the results at the American Heart Association scientific sessions.
Streamlining heart failure treatment
The demonstration that empagliflozin is an effective – and safe – treatment for patients with HFpEF not only provides a new treatment for a disorder that until now had no evidence-based intervention, but also streamlines the management approach for treating patients with heart failure with an agent from empagliflozin’s class, the SGLT2 inhibitors, commented Mary Norine Walsh, MD, medical director of the heart failure and cardiac transplantation programs at Ascension St. Vincent Heart Center in Indianapolis.
That’s because empagliflozin has shown significant and consistent benefit across essentially the full range of LVEFs seen in patients with heart failure based on its performance in EMPEROR-Preserved as well as in a mirror-image trial, EMPEROR-Reduced, run in patients with heart failure with reduced ejection fraction.
“Clinicians do not need to stop and assess LVEF with echocardiography or other imaging before they decide on how to treat heart failure patients” with an SGLT2 inhibitor, noted Dr. Walsh, a designated discussant for the report. “Clinicians who are busy can now refer less to LVEF than to the patient’s phenotype.”
Treatment prevents hospitalization for heart failure
The more-detailed data reported by Dr. Anker also strengthened the case that the benefit from empagliflozin in patients with an LVEF of at least 50% mostly came from a reduction in hospitalizations for heart failure (HHF), which dropped following start of empagliflozin treatment by a relative 22%, compared with placebo for first HHF, a significant decline, and by a relative 17% for total HHF, a reduction that missed significance in this secondary analysis. The other half of the primary endpoint, cardiovascular death, declined by a nonsignificant 11% with empagliflozin treatment, compared with placebo in patients with clear-cut HFpEF.
The significant reduction in first HHF is, by itself, sufficient reason to use empagliflozin (or possibly a different SGLT2 inhibitor) in patients with HFpEF, maintained Clyde W. Yancy, MD, professor and chief of cardiology at Northwestern Medicine in Chicago.
“Attenuated HHF is a meaningful outcome,” stressed Dr. Yancy, also a discussant for the study. “This is the first time we’ve had evidence supporting that we can change the natural history of patients with HFpEF. While we still need to find interventions that save lives, we cannot overlook that this treatment can improve morbidity, and we cannot overlook that patient quality of life is better.”
Further benefits in patients with an LVEF of at least 50%
Dr. Anker, professor of cardiology and metabolism at Charité Medical University in Berlin, also reported results from several other analyses that further defined the effect of empagliflozin on clinical outcomes of patients with “true” HFpEF:
- The impact of empagliflozin, compared with placebo, for reducing both the study’s combined, primary outcome as well as total HHF was statistically consistent across all strata of LVEF, from 50% to greater than 70%. However, both outcome measures also showed a puzzling loss of benefit among patients with an LVEF of 65%-69%. In prior reports, a researcher on the EMPEROR-Preserved team, Milton Packer, MD, speculated that some patients in this LVEF stratum might not actually have had heart failure but instead had a different disorder that mimicked heart failure in clinical presentation, such as atrial fibrillation.
- Patients’ quality of life as measured by the Kansas City Cardiomyopathy Questionnaire showed a consistent benefit from empagliflozin treatment, compared with placebo, both in patients with an LVEF of at least 50% as well as in those with an LVEF of 41%-49%. In both subgroups the adjusted mean difference from placebo was significant and about 1.5 points.
- Patients showed a significant improvement in average New York Heart Association functional class while on treatment, and a strong trend toward less deterioration in functional class while on treatment.
- Deterioration of renal function on treatment slowed by an average 1.24 mL/min per 1.73 m2 per year in patients on empagliflozin, compared with placebo, in the subgroup with an LVEF of at least 50%.
Dr. Anker also reported the primary outcome and component results for the subgroup of patients with a baseline LVEF of 41%-49%. These patients had what looked like a “bigger magnitude” of effect from treatment, he noted, showing a significant 29% relative decline in the primary endpoint, compared with placebo-treated patients, and a significant 42% relative drop in first HHF and a significant 43% relative decline in total HHF, compared with placebo.
The primary analysis from EMPEROR-Preserved, which included all 5,988 randomized patients with heart failure and an LVEF of 41% or greater, showed a significant reduction in the combined, primary endpoint with empagliflozin treatment of 21%, compared with control patients during a median follow-up of about 26 months. The absolute rate reduction of the combined primary endpoint was 3.3% during 26-months’ follow-up. Statistical tests have shown no heterogeneity of this effect by diabetes status (49% of patients had diabetes), nor by renal function down to an estimated glomerular filtration rate at entry as low as 20 mL/min per 1.73 m2.
EMPEROR-Preserved was sponsored by Boehringer Ingelheim and Lilly, the two companies that market empagliflozin (Jardiance). Dr. Anker has been a consultant to Boehringer Ingelheim as well as to Abbott Vascular, Bayer, Brahms, Cardiac Dimensions, Cordio, Novartis, Servier, and Vifor. Dr. Walsh and Dr. Yancy had no disclosures.
FROM AHA2021
Specific blood pressure-lowering drugs prevent onset of new diabetes
results from a new meta-analysis show.
Angiotensin-converting enzyme (ACE) inhibitors and angiotensin II receptor blockers (ARB) – so-called renin-angiotensin system (RAS) blockers – showed the strongest association with preventive effects, while conversely, beta-blocker and thiazide diuretic antihypertensives were linked to an increased risk of new-onset diabetes.
“This study suggests that blood pressure lowering can help prevent diabetes in addition to its well-established beneficial effects in reducing cardiovascular events,” write Milad Nazarzadeh and colleagues with the Blood Pressure Lowering Treatment Trialists’ Collaboration in their article published in The Lancet.
“The differing effects of the drug classes support decision-making for antihypertensive drug choice according to an individual’s risk profile,” note Mr. Nazarzadeh, of Deep Medicine, Oxford Martin School, University of Oxford, U.K., and colleagues.
“In particular, [RAS inhibitors], ACE inhibitors and ARBs, should become the drugs of choice when clinical risk of diabetes is of concern, whereas beta blockers and thiazide diuretics should be avoided where possible,” they add.
In an accompanying editorial, Matthew A. Cavender, MD, MPH, and Robert C. Wirka, MD, of the University of North Carolina at Chapel Hill, agree that the new findings, along with the bulk of previous evidence, point to an important role of RAS-inhibiting drugs in diabetes prevention.
“Based on the accumulated evidence, including the results of these analyses, blood pressure control, particularly with RAS inhibition, should be considered as a possible strategy to reduce the risk of developing diabetes,” they write.
They note that, while “the absolute risk reduction found in this meta-analysis is modest, interventions with small benefits can have an outsized effect when applied to conditions as common as hypertension.”
And commenting on the findings to the U.K. Science & Media Centre, Marc George, MBChB, PhD, blood pressure clinical lead for University College London Hospital, U.K., said: “Lowering blood pressure prevents heart attacks, strokes, and kidney failure, and this new large and comprehensive study published in The Lancet also shows that it lowers the risk of developing diabetes. Until now this effect was not clear.”
Kevin McConway, PhD, emeritus professor of applied statistics, The Open University, U.K., similarly concurs: “Though there is good evidence that lowering people’s blood pressure, if it is too high, can have important health benefits in reducing the risk of heart attacks and strokes, it hasn’t been clear whether lowering blood pressure can reduce the chance of developing type 2 diabetes in the future. This is an impressive study.”
RAS blockers associated with lower diabetes risk
The findings are from an individual data meta-analysis of 19 randomized, placebo-controlled trials conducted between 1973 and 2008 and involving five major classes of antihypertensive drugs: ACE inhibitors, ARBs, beta-blockers, thiazide diuretics, and calcium channel blockers.
Overall, the studies included 145,939 participants, of whom 60.6% were men.
Over a median follow-up of 4.5 years, 9,883 of the study participants developed new-onset type 2 diabetes.
Those treated with ACE inhibitors or ARBs had a reduced relative risk of new-onset diabetes that was nearly identical (risk reduction, 0.84 for both) versus placebo.
However, treatment with beta-blockers or thiazide diuretics was associated with an increased risk of type 2 diabetes (RR, 1.48 and 1.20, respectively), consistent with previous evidence that, specifically, second-line thiazide diuretics and third-line beta blockers increase the risk of diabetes.
No significant reduction or increase in risk was observed with calcium channel blockers (RR, 1.02).
For the reductions with ACE inhibitors and ARBs, each reduction in systolic blood pressure of 5-mm Hg was associated with an 11% reduced risk of developing diabetes.
“The relative magnitude of reduction per 5-mm Hg systolic blood pressure lowering was similar to those reported for prevention of major cardiovascular events,” the authors say.
“[This] will strengthen the case for blood pressure reduction through lifestyle interventions known to reduce blood pressure, and blood pressure lowering treatments with drugs, and possibly device therapies,” they say.
In the opposite direction, research has suggested that each 20-mm Hg increase in systolic blood pressure is associated with as much as a 77% increased risk of type 2 diabetes; however, the causality of that association is uncertain, the authors note.
Results fill gap in evidence for guidelines
The meta-analysis findings were further validated in a supplemental mendelian randomization analysis, which used data from the International Consortium for Blood Pressure genome-wide association study and the UK Biobank. The analysis showed that people with genetic variants that have a similar effect on the RAS pathway as ACE inhibitors and ARBs also had a reduced risk of diabetes.
On this point, Dipender Gill, BMBCh, PhD, lecturer in clinical pharmacology and therapeutics at St. George’s, University of London, told the U.K. Science and Media Centre: “This is a comprehensive study triangulating clinical trial and genetic data to find support for effects of blood pressure reduction through particular pharmacological targets on glycemic control and risk of type 2 diabetes.”
Mr. Nazarzadeh and colleagues say that uncertainty regarding whether the reduction in diabetes risk is caused by blood pressure lowering itself, or by some other effect of the antihypertensive drugs, has meant that guideline recommendations on the role of antihypertensive drugs have been lacking.
However, the authors assert that “our study fills this gap in evidence using individual participant data from randomized controlled trials and assessing effects for a standardized fixed degree of blood pressure reduction.”
“With consistent results from both randomized controlled trials and genetic analyses, we have shown that elevated blood pressure is indeed a modifiable risk factor for new-onset type 2 diabetes in people without a diagnosis of diabetes, with a relative effect size similar to those seen for the prevention of major cardiovascular disease,” they state.
Authors of U.S. hypertension guidelines should follow lead of ESC
Under the European Society of Cardiology (ESC) guidelines, RAS inhibitors (in combination with a calcium channel blocker or thiazide diuretic) have a class 1 recommendation for the treatment of hypertension; however, diabetes and cardiology societies in the United States only recommend a preference for a RAS inhibitor over other agents among those with concomitant albuminuria.
But with an estimated 13% of Americans having diabetes and a striking 34.5% having prediabetes, the need for more measures to tackle the problem is urgent, say Dr. Cavender and Dr. Wirka in their editorial.
“Perhaps these data are enough to encourage the writers of the hypertension guidelines in the U.S. to follow the lead of the ESC to make RAS inhibitors the first-line hypertension treatment for all patients and not just in those with albuminuria,” they state.
Dr. Cavender has reported receiving research support from Amgen, AstraZeneca, Boehringer-Ingelheim, CSL Behring, and Novartis, and consulting fees from Amgen, AstraZeneca, Bayer, Boehringer-Ingelheim, Boston Scientific, Edwards Lifesciences, Merck, and Novo Nordisk. Disclosures for the other authors are listed with the article. Dr. Wirka and Dr. George have reported no relevant financial relationships. Dr. McConway is a trustee of the SMC and member of its advisory committee. Dr. Gill is employed part-time by Novo Nordisk.
A version of this article first appeared on Medscape.com.
results from a new meta-analysis show.
Angiotensin-converting enzyme (ACE) inhibitors and angiotensin II receptor blockers (ARB) – so-called renin-angiotensin system (RAS) blockers – showed the strongest association with preventive effects, while conversely, beta-blocker and thiazide diuretic antihypertensives were linked to an increased risk of new-onset diabetes.
“This study suggests that blood pressure lowering can help prevent diabetes in addition to its well-established beneficial effects in reducing cardiovascular events,” write Milad Nazarzadeh and colleagues with the Blood Pressure Lowering Treatment Trialists’ Collaboration in their article published in The Lancet.
“The differing effects of the drug classes support decision-making for antihypertensive drug choice according to an individual’s risk profile,” note Mr. Nazarzadeh, of Deep Medicine, Oxford Martin School, University of Oxford, U.K., and colleagues.
“In particular, [RAS inhibitors], ACE inhibitors and ARBs, should become the drugs of choice when clinical risk of diabetes is of concern, whereas beta blockers and thiazide diuretics should be avoided where possible,” they add.
In an accompanying editorial, Matthew A. Cavender, MD, MPH, and Robert C. Wirka, MD, of the University of North Carolina at Chapel Hill, agree that the new findings, along with the bulk of previous evidence, point to an important role of RAS-inhibiting drugs in diabetes prevention.
“Based on the accumulated evidence, including the results of these analyses, blood pressure control, particularly with RAS inhibition, should be considered as a possible strategy to reduce the risk of developing diabetes,” they write.
They note that, while “the absolute risk reduction found in this meta-analysis is modest, interventions with small benefits can have an outsized effect when applied to conditions as common as hypertension.”
And commenting on the findings to the U.K. Science & Media Centre, Marc George, MBChB, PhD, blood pressure clinical lead for University College London Hospital, U.K., said: “Lowering blood pressure prevents heart attacks, strokes, and kidney failure, and this new large and comprehensive study published in The Lancet also shows that it lowers the risk of developing diabetes. Until now this effect was not clear.”
Kevin McConway, PhD, emeritus professor of applied statistics, The Open University, U.K., similarly concurs: “Though there is good evidence that lowering people’s blood pressure, if it is too high, can have important health benefits in reducing the risk of heart attacks and strokes, it hasn’t been clear whether lowering blood pressure can reduce the chance of developing type 2 diabetes in the future. This is an impressive study.”
RAS blockers associated with lower diabetes risk
The findings are from an individual data meta-analysis of 19 randomized, placebo-controlled trials conducted between 1973 and 2008 and involving five major classes of antihypertensive drugs: ACE inhibitors, ARBs, beta-blockers, thiazide diuretics, and calcium channel blockers.
Overall, the studies included 145,939 participants, of whom 60.6% were men.
Over a median follow-up of 4.5 years, 9,883 of the study participants developed new-onset type 2 diabetes.
Those treated with ACE inhibitors or ARBs had a reduced relative risk of new-onset diabetes that was nearly identical (risk reduction, 0.84 for both) versus placebo.
However, treatment with beta-blockers or thiazide diuretics was associated with an increased risk of type 2 diabetes (RR, 1.48 and 1.20, respectively), consistent with previous evidence that, specifically, second-line thiazide diuretics and third-line beta blockers increase the risk of diabetes.
No significant reduction or increase in risk was observed with calcium channel blockers (RR, 1.02).
For the reductions with ACE inhibitors and ARBs, each reduction in systolic blood pressure of 5-mm Hg was associated with an 11% reduced risk of developing diabetes.
“The relative magnitude of reduction per 5-mm Hg systolic blood pressure lowering was similar to those reported for prevention of major cardiovascular events,” the authors say.
“[This] will strengthen the case for blood pressure reduction through lifestyle interventions known to reduce blood pressure, and blood pressure lowering treatments with drugs, and possibly device therapies,” they say.
In the opposite direction, research has suggested that each 20-mm Hg increase in systolic blood pressure is associated with as much as a 77% increased risk of type 2 diabetes; however, the causality of that association is uncertain, the authors note.
Results fill gap in evidence for guidelines
The meta-analysis findings were further validated in a supplemental mendelian randomization analysis, which used data from the International Consortium for Blood Pressure genome-wide association study and the UK Biobank. The analysis showed that people with genetic variants that have a similar effect on the RAS pathway as ACE inhibitors and ARBs also had a reduced risk of diabetes.
On this point, Dipender Gill, BMBCh, PhD, lecturer in clinical pharmacology and therapeutics at St. George’s, University of London, told the U.K. Science and Media Centre: “This is a comprehensive study triangulating clinical trial and genetic data to find support for effects of blood pressure reduction through particular pharmacological targets on glycemic control and risk of type 2 diabetes.”
Mr. Nazarzadeh and colleagues say that uncertainty regarding whether the reduction in diabetes risk is caused by blood pressure lowering itself, or by some other effect of the antihypertensive drugs, has meant that guideline recommendations on the role of antihypertensive drugs have been lacking.
However, the authors assert that “our study fills this gap in evidence using individual participant data from randomized controlled trials and assessing effects for a standardized fixed degree of blood pressure reduction.”
“With consistent results from both randomized controlled trials and genetic analyses, we have shown that elevated blood pressure is indeed a modifiable risk factor for new-onset type 2 diabetes in people without a diagnosis of diabetes, with a relative effect size similar to those seen for the prevention of major cardiovascular disease,” they state.
Authors of U.S. hypertension guidelines should follow lead of ESC
Under the European Society of Cardiology (ESC) guidelines, RAS inhibitors (in combination with a calcium channel blocker or thiazide diuretic) have a class 1 recommendation for the treatment of hypertension; however, diabetes and cardiology societies in the United States only recommend a preference for a RAS inhibitor over other agents among those with concomitant albuminuria.
But with an estimated 13% of Americans having diabetes and a striking 34.5% having prediabetes, the need for more measures to tackle the problem is urgent, say Dr. Cavender and Dr. Wirka in their editorial.
“Perhaps these data are enough to encourage the writers of the hypertension guidelines in the U.S. to follow the lead of the ESC to make RAS inhibitors the first-line hypertension treatment for all patients and not just in those with albuminuria,” they state.
Dr. Cavender has reported receiving research support from Amgen, AstraZeneca, Boehringer-Ingelheim, CSL Behring, and Novartis, and consulting fees from Amgen, AstraZeneca, Bayer, Boehringer-Ingelheim, Boston Scientific, Edwards Lifesciences, Merck, and Novo Nordisk. Disclosures for the other authors are listed with the article. Dr. Wirka and Dr. George have reported no relevant financial relationships. Dr. McConway is a trustee of the SMC and member of its advisory committee. Dr. Gill is employed part-time by Novo Nordisk.
A version of this article first appeared on Medscape.com.
results from a new meta-analysis show.
Angiotensin-converting enzyme (ACE) inhibitors and angiotensin II receptor blockers (ARB) – so-called renin-angiotensin system (RAS) blockers – showed the strongest association with preventive effects, while conversely, beta-blocker and thiazide diuretic antihypertensives were linked to an increased risk of new-onset diabetes.
“This study suggests that blood pressure lowering can help prevent diabetes in addition to its well-established beneficial effects in reducing cardiovascular events,” write Milad Nazarzadeh and colleagues with the Blood Pressure Lowering Treatment Trialists’ Collaboration in their article published in The Lancet.
“The differing effects of the drug classes support decision-making for antihypertensive drug choice according to an individual’s risk profile,” note Mr. Nazarzadeh, of Deep Medicine, Oxford Martin School, University of Oxford, U.K., and colleagues.
“In particular, [RAS inhibitors], ACE inhibitors and ARBs, should become the drugs of choice when clinical risk of diabetes is of concern, whereas beta blockers and thiazide diuretics should be avoided where possible,” they add.
In an accompanying editorial, Matthew A. Cavender, MD, MPH, and Robert C. Wirka, MD, of the University of North Carolina at Chapel Hill, agree that the new findings, along with the bulk of previous evidence, point to an important role of RAS-inhibiting drugs in diabetes prevention.
“Based on the accumulated evidence, including the results of these analyses, blood pressure control, particularly with RAS inhibition, should be considered as a possible strategy to reduce the risk of developing diabetes,” they write.
They note that, while “the absolute risk reduction found in this meta-analysis is modest, interventions with small benefits can have an outsized effect when applied to conditions as common as hypertension.”
And commenting on the findings to the U.K. Science & Media Centre, Marc George, MBChB, PhD, blood pressure clinical lead for University College London Hospital, U.K., said: “Lowering blood pressure prevents heart attacks, strokes, and kidney failure, and this new large and comprehensive study published in The Lancet also shows that it lowers the risk of developing diabetes. Until now this effect was not clear.”
Kevin McConway, PhD, emeritus professor of applied statistics, The Open University, U.K., similarly concurs: “Though there is good evidence that lowering people’s blood pressure, if it is too high, can have important health benefits in reducing the risk of heart attacks and strokes, it hasn’t been clear whether lowering blood pressure can reduce the chance of developing type 2 diabetes in the future. This is an impressive study.”
RAS blockers associated with lower diabetes risk
The findings are from an individual data meta-analysis of 19 randomized, placebo-controlled trials conducted between 1973 and 2008 and involving five major classes of antihypertensive drugs: ACE inhibitors, ARBs, beta-blockers, thiazide diuretics, and calcium channel blockers.
Overall, the studies included 145,939 participants, of whom 60.6% were men.
Over a median follow-up of 4.5 years, 9,883 of the study participants developed new-onset type 2 diabetes.
Those treated with ACE inhibitors or ARBs had a reduced relative risk of new-onset diabetes that was nearly identical (risk reduction, 0.84 for both) versus placebo.
However, treatment with beta-blockers or thiazide diuretics was associated with an increased risk of type 2 diabetes (RR, 1.48 and 1.20, respectively), consistent with previous evidence that, specifically, second-line thiazide diuretics and third-line beta blockers increase the risk of diabetes.
No significant reduction or increase in risk was observed with calcium channel blockers (RR, 1.02).
For the reductions with ACE inhibitors and ARBs, each reduction in systolic blood pressure of 5-mm Hg was associated with an 11% reduced risk of developing diabetes.
“The relative magnitude of reduction per 5-mm Hg systolic blood pressure lowering was similar to those reported for prevention of major cardiovascular events,” the authors say.
“[This] will strengthen the case for blood pressure reduction through lifestyle interventions known to reduce blood pressure, and blood pressure lowering treatments with drugs, and possibly device therapies,” they say.
In the opposite direction, research has suggested that each 20-mm Hg increase in systolic blood pressure is associated with as much as a 77% increased risk of type 2 diabetes; however, the causality of that association is uncertain, the authors note.
Results fill gap in evidence for guidelines
The meta-analysis findings were further validated in a supplemental mendelian randomization analysis, which used data from the International Consortium for Blood Pressure genome-wide association study and the UK Biobank. The analysis showed that people with genetic variants that have a similar effect on the RAS pathway as ACE inhibitors and ARBs also had a reduced risk of diabetes.
On this point, Dipender Gill, BMBCh, PhD, lecturer in clinical pharmacology and therapeutics at St. George’s, University of London, told the U.K. Science and Media Centre: “This is a comprehensive study triangulating clinical trial and genetic data to find support for effects of blood pressure reduction through particular pharmacological targets on glycemic control and risk of type 2 diabetes.”
Mr. Nazarzadeh and colleagues say that uncertainty regarding whether the reduction in diabetes risk is caused by blood pressure lowering itself, or by some other effect of the antihypertensive drugs, has meant that guideline recommendations on the role of antihypertensive drugs have been lacking.
However, the authors assert that “our study fills this gap in evidence using individual participant data from randomized controlled trials and assessing effects for a standardized fixed degree of blood pressure reduction.”
“With consistent results from both randomized controlled trials and genetic analyses, we have shown that elevated blood pressure is indeed a modifiable risk factor for new-onset type 2 diabetes in people without a diagnosis of diabetes, with a relative effect size similar to those seen for the prevention of major cardiovascular disease,” they state.
Authors of U.S. hypertension guidelines should follow lead of ESC
Under the European Society of Cardiology (ESC) guidelines, RAS inhibitors (in combination with a calcium channel blocker or thiazide diuretic) have a class 1 recommendation for the treatment of hypertension; however, diabetes and cardiology societies in the United States only recommend a preference for a RAS inhibitor over other agents among those with concomitant albuminuria.
But with an estimated 13% of Americans having diabetes and a striking 34.5% having prediabetes, the need for more measures to tackle the problem is urgent, say Dr. Cavender and Dr. Wirka in their editorial.
“Perhaps these data are enough to encourage the writers of the hypertension guidelines in the U.S. to follow the lead of the ESC to make RAS inhibitors the first-line hypertension treatment for all patients and not just in those with albuminuria,” they state.
Dr. Cavender has reported receiving research support from Amgen, AstraZeneca, Boehringer-Ingelheim, CSL Behring, and Novartis, and consulting fees from Amgen, AstraZeneca, Bayer, Boehringer-Ingelheim, Boston Scientific, Edwards Lifesciences, Merck, and Novo Nordisk. Disclosures for the other authors are listed with the article. Dr. Wirka and Dr. George have reported no relevant financial relationships. Dr. McConway is a trustee of the SMC and member of its advisory committee. Dr. Gill is employed part-time by Novo Nordisk.
A version of this article first appeared on Medscape.com.
FROM THE LANCET
ASCEND: Aspirin shows hint of dementia protection in T2D
A regimen of daily, low-dose aspirin failed to produce a significant reduction in the incidence of dementia or cognitive impairment in ASCEND, a randomized, multicenter trial with more than 15,000 people with diabetes followed for an average of more than 9 years, but the results hinted at enough of a benefit to warrant further study, some experts said.
“The question remains open,” said Jane Armitage, MBBS, FRCP, as she presented the findings at the American Heart Association scientific sessions. “The rate ratios suggest some benefit. It’s encouraging,” added Dr. Armitage, professor of clinical trials and epidemiology at Oxford (England) University.
The study tallied dementia outcomes three different ways: It applied a narrow definition that relied on a specific diagnosis of dementia in a person’s EHR or in their death record. (Dr. Armitage and her associates tracked outcomes for 99% of the enrolled participants by linking to their U.K. national health records and death records.)
A second metric used a broader outcome definition that tracked EHR entries for not only dementia but also diagnoses of cognitive impairment, delirium, confusion, prescription of dementia medications, and referral to a memory clinic or geriatric psychiatry. The third assessment was a cognitive-function test given to participants at the end of follow-up, but only 58% of enrolled participants completed this part of the study, and it’s also possible that some subjects missed this assessment because of dementia onset. These limitations hamper clear interpretation of this third metric, Dr. Armitage said.
The main findings for the other two, more reliable measures of incident dementia or cognitive deterioration showed a nonsignificant 9% relative risk reduction linked with aspirin use compared with placebo for the more inclusive endpoint, and a nonsignificant 11% relative risk reduction with aspirin using the narrow definition for dementia only, she reported. The third method, a directly administered assessment of dementia and cognition, also showed a small, nonsignificant effect from daily aspirin use relative to placebo.
Results can’t rule out modest aspirin effect
Dr. Armitage highlighted that the two more reliable measures both appeared to rule out risk for neurologic harm from aspirin because the upper limit of the 95% confidence interval for relative effect reached only 1.02 using the broad outcomes, and 1.06 for the narrower endpoint of dementia only. On the other hand, focus on the low end of the 95% confidence interval suggested potentially meaningful benefits, with a possible reduction by aspirin in events relative to placebo of as much as 19% by the broad outcome definition and by 25% with the narrow definition.
“Even if it was only a 15% relative risk reduction, that would be important,” given the high dementia incidence worldwide, Dr. Armitage said during a press briefing. “It’s entirely possible, with our results, that a modest benefit exists.”
This take on the findings won some support. Further studies with more people, longer follow-up, and perhaps enrolling a more selected, higher risk cohort may better address potential neurologic benefit from aspirin, suggested Amytis Towfighi, MD, a stroke neurologist and professor of neurology at the University of Southern California, Los Angeles, and a designated discussant for the report.
The result “was rather encouraging. I was a little surprised” by the findings, commented Chrystie M. Ballantyne, MD, professor and director of the Center for Cardiometabolic Disease Prevention at Baylor College of Medicine, Houston, also a discussant.
The results “don’t mean that no one benefits from aspirin. Perhaps certain people at risk would benefit from dementia protection. It’s an open question,” commented Erin D. Michos, MD, director of Women’s Cardiovascular Health at Johns Hopkins Medicine, Baltimore.
But others saw the findings as more unequivocally neutral. “This gives us an early, preliminary answer, that aspirin does not seem to improve dementia,” commented Amit Khera, MD, professor and director of Preventive Cardiology at UT Southwestern Medical Center, Dallas, and a third discussant at the meeting.
Evidence against routine, widespread primary prevention with aspirin
ASCEND had the primary goal of assessing a daily, 100-mg aspirin dose for its safety and efficacy for preventing vascular events such as MIs and ischemic strokes in 15,480 people with diabetes who were at least 40 years old at enrollment and had no history of cardiovascular disease. The main results came out in 2018 and showed that while aspirin produced a significant benefit by reducing thrombotic events, it also resulted in significantly more major bleeding events compared with placebo, and overall the magnitude of benefit roughly matched magnitude of risk.
These findings, along with similar results from two other high-profile aspirin studies reported at about the same time (ASPREE, and ARRIVE), led to recommendations from groups like the U.S. Preventive Services Task Force and from the American College of Cardiology and American Heart Association that caution against widespread, routine aspirin use for primary prevention of atherosclerotic cardiovascular disease events in most adults.
The groups instead endorsed a tailored strategy of targeting aspirin to people with a higher than average risk for ischemic thrombotic events and a lower than average bleeding risk. (The most recent aspirin recommendations from the USPSTF, currently in draft form, substantially curtail aspirin’s appropriate use, eliminating it in those over age 60 years.)
However, experts and prevailing practice recommendations continue to endorse routine aspirin use for secondary prevention in patients with an established history of cardiovascular disease.
The new findings reported by Dr. Armitage came from additional analyses of dementia and cognitive impairment overlaid on the main ASCEND outcome analyses. ASCEND actively treated and followed study participants for an average of 7.4 years, then researchers tracked further dementia outcomes based on medical-record entries for an average of another 1.8 years.
ASCEND received partial funding or support from Abbott, Bayer, Mylan, and Solvay. Dr. Armitage had no disclosures. Dr. Towfighi, Dr. Khera, and Dr. Michos had no disclosures. Dr. Ballantyne has had financial relationships with numerous companies.
A regimen of daily, low-dose aspirin failed to produce a significant reduction in the incidence of dementia or cognitive impairment in ASCEND, a randomized, multicenter trial with more than 15,000 people with diabetes followed for an average of more than 9 years, but the results hinted at enough of a benefit to warrant further study, some experts said.
“The question remains open,” said Jane Armitage, MBBS, FRCP, as she presented the findings at the American Heart Association scientific sessions. “The rate ratios suggest some benefit. It’s encouraging,” added Dr. Armitage, professor of clinical trials and epidemiology at Oxford (England) University.
The study tallied dementia outcomes three different ways: It applied a narrow definition that relied on a specific diagnosis of dementia in a person’s EHR or in their death record. (Dr. Armitage and her associates tracked outcomes for 99% of the enrolled participants by linking to their U.K. national health records and death records.)
A second metric used a broader outcome definition that tracked EHR entries for not only dementia but also diagnoses of cognitive impairment, delirium, confusion, prescription of dementia medications, and referral to a memory clinic or geriatric psychiatry. The third assessment was a cognitive-function test given to participants at the end of follow-up, but only 58% of enrolled participants completed this part of the study, and it’s also possible that some subjects missed this assessment because of dementia onset. These limitations hamper clear interpretation of this third metric, Dr. Armitage said.
The main findings for the other two, more reliable measures of incident dementia or cognitive deterioration showed a nonsignificant 9% relative risk reduction linked with aspirin use compared with placebo for the more inclusive endpoint, and a nonsignificant 11% relative risk reduction with aspirin using the narrow definition for dementia only, she reported. The third method, a directly administered assessment of dementia and cognition, also showed a small, nonsignificant effect from daily aspirin use relative to placebo.
Results can’t rule out modest aspirin effect
Dr. Armitage highlighted that the two more reliable measures both appeared to rule out risk for neurologic harm from aspirin because the upper limit of the 95% confidence interval for relative effect reached only 1.02 using the broad outcomes, and 1.06 for the narrower endpoint of dementia only. On the other hand, focus on the low end of the 95% confidence interval suggested potentially meaningful benefits, with a possible reduction by aspirin in events relative to placebo of as much as 19% by the broad outcome definition and by 25% with the narrow definition.
“Even if it was only a 15% relative risk reduction, that would be important,” given the high dementia incidence worldwide, Dr. Armitage said during a press briefing. “It’s entirely possible, with our results, that a modest benefit exists.”
This take on the findings won some support. Further studies with more people, longer follow-up, and perhaps enrolling a more selected, higher risk cohort may better address potential neurologic benefit from aspirin, suggested Amytis Towfighi, MD, a stroke neurologist and professor of neurology at the University of Southern California, Los Angeles, and a designated discussant for the report.
The result “was rather encouraging. I was a little surprised” by the findings, commented Chrystie M. Ballantyne, MD, professor and director of the Center for Cardiometabolic Disease Prevention at Baylor College of Medicine, Houston, also a discussant.
The results “don’t mean that no one benefits from aspirin. Perhaps certain people at risk would benefit from dementia protection. It’s an open question,” commented Erin D. Michos, MD, director of Women’s Cardiovascular Health at Johns Hopkins Medicine, Baltimore.
But others saw the findings as more unequivocally neutral. “This gives us an early, preliminary answer, that aspirin does not seem to improve dementia,” commented Amit Khera, MD, professor and director of Preventive Cardiology at UT Southwestern Medical Center, Dallas, and a third discussant at the meeting.
Evidence against routine, widespread primary prevention with aspirin
ASCEND had the primary goal of assessing a daily, 100-mg aspirin dose for its safety and efficacy for preventing vascular events such as MIs and ischemic strokes in 15,480 people with diabetes who were at least 40 years old at enrollment and had no history of cardiovascular disease. The main results came out in 2018 and showed that while aspirin produced a significant benefit by reducing thrombotic events, it also resulted in significantly more major bleeding events compared with placebo, and overall the magnitude of benefit roughly matched magnitude of risk.
These findings, along with similar results from two other high-profile aspirin studies reported at about the same time (ASPREE, and ARRIVE), led to recommendations from groups like the U.S. Preventive Services Task Force and from the American College of Cardiology and American Heart Association that caution against widespread, routine aspirin use for primary prevention of atherosclerotic cardiovascular disease events in most adults.
The groups instead endorsed a tailored strategy of targeting aspirin to people with a higher than average risk for ischemic thrombotic events and a lower than average bleeding risk. (The most recent aspirin recommendations from the USPSTF, currently in draft form, substantially curtail aspirin’s appropriate use, eliminating it in those over age 60 years.)
However, experts and prevailing practice recommendations continue to endorse routine aspirin use for secondary prevention in patients with an established history of cardiovascular disease.
The new findings reported by Dr. Armitage came from additional analyses of dementia and cognitive impairment overlaid on the main ASCEND outcome analyses. ASCEND actively treated and followed study participants for an average of 7.4 years, then researchers tracked further dementia outcomes based on medical-record entries for an average of another 1.8 years.
ASCEND received partial funding or support from Abbott, Bayer, Mylan, and Solvay. Dr. Armitage had no disclosures. Dr. Towfighi, Dr. Khera, and Dr. Michos had no disclosures. Dr. Ballantyne has had financial relationships with numerous companies.
A regimen of daily, low-dose aspirin failed to produce a significant reduction in the incidence of dementia or cognitive impairment in ASCEND, a randomized, multicenter trial with more than 15,000 people with diabetes followed for an average of more than 9 years, but the results hinted at enough of a benefit to warrant further study, some experts said.
“The question remains open,” said Jane Armitage, MBBS, FRCP, as she presented the findings at the American Heart Association scientific sessions. “The rate ratios suggest some benefit. It’s encouraging,” added Dr. Armitage, professor of clinical trials and epidemiology at Oxford (England) University.
The study tallied dementia outcomes three different ways: It applied a narrow definition that relied on a specific diagnosis of dementia in a person’s EHR or in their death record. (Dr. Armitage and her associates tracked outcomes for 99% of the enrolled participants by linking to their U.K. national health records and death records.)
A second metric used a broader outcome definition that tracked EHR entries for not only dementia but also diagnoses of cognitive impairment, delirium, confusion, prescription of dementia medications, and referral to a memory clinic or geriatric psychiatry. The third assessment was a cognitive-function test given to participants at the end of follow-up, but only 58% of enrolled participants completed this part of the study, and it’s also possible that some subjects missed this assessment because of dementia onset. These limitations hamper clear interpretation of this third metric, Dr. Armitage said.
The main findings for the other two, more reliable measures of incident dementia or cognitive deterioration showed a nonsignificant 9% relative risk reduction linked with aspirin use compared with placebo for the more inclusive endpoint, and a nonsignificant 11% relative risk reduction with aspirin using the narrow definition for dementia only, she reported. The third method, a directly administered assessment of dementia and cognition, also showed a small, nonsignificant effect from daily aspirin use relative to placebo.
Results can’t rule out modest aspirin effect
Dr. Armitage highlighted that the two more reliable measures both appeared to rule out risk for neurologic harm from aspirin because the upper limit of the 95% confidence interval for relative effect reached only 1.02 using the broad outcomes, and 1.06 for the narrower endpoint of dementia only. On the other hand, focus on the low end of the 95% confidence interval suggested potentially meaningful benefits, with a possible reduction by aspirin in events relative to placebo of as much as 19% by the broad outcome definition and by 25% with the narrow definition.
“Even if it was only a 15% relative risk reduction, that would be important,” given the high dementia incidence worldwide, Dr. Armitage said during a press briefing. “It’s entirely possible, with our results, that a modest benefit exists.”
This take on the findings won some support. Further studies with more people, longer follow-up, and perhaps enrolling a more selected, higher risk cohort may better address potential neurologic benefit from aspirin, suggested Amytis Towfighi, MD, a stroke neurologist and professor of neurology at the University of Southern California, Los Angeles, and a designated discussant for the report.
The result “was rather encouraging. I was a little surprised” by the findings, commented Chrystie M. Ballantyne, MD, professor and director of the Center for Cardiometabolic Disease Prevention at Baylor College of Medicine, Houston, also a discussant.
The results “don’t mean that no one benefits from aspirin. Perhaps certain people at risk would benefit from dementia protection. It’s an open question,” commented Erin D. Michos, MD, director of Women’s Cardiovascular Health at Johns Hopkins Medicine, Baltimore.
But others saw the findings as more unequivocally neutral. “This gives us an early, preliminary answer, that aspirin does not seem to improve dementia,” commented Amit Khera, MD, professor and director of Preventive Cardiology at UT Southwestern Medical Center, Dallas, and a third discussant at the meeting.
Evidence against routine, widespread primary prevention with aspirin
ASCEND had the primary goal of assessing a daily, 100-mg aspirin dose for its safety and efficacy for preventing vascular events such as MIs and ischemic strokes in 15,480 people with diabetes who were at least 40 years old at enrollment and had no history of cardiovascular disease. The main results came out in 2018 and showed that while aspirin produced a significant benefit by reducing thrombotic events, it also resulted in significantly more major bleeding events compared with placebo, and overall the magnitude of benefit roughly matched magnitude of risk.
These findings, along with similar results from two other high-profile aspirin studies reported at about the same time (ASPREE, and ARRIVE), led to recommendations from groups like the U.S. Preventive Services Task Force and from the American College of Cardiology and American Heart Association that caution against widespread, routine aspirin use for primary prevention of atherosclerotic cardiovascular disease events in most adults.
The groups instead endorsed a tailored strategy of targeting aspirin to people with a higher than average risk for ischemic thrombotic events and a lower than average bleeding risk. (The most recent aspirin recommendations from the USPSTF, currently in draft form, substantially curtail aspirin’s appropriate use, eliminating it in those over age 60 years.)
However, experts and prevailing practice recommendations continue to endorse routine aspirin use for secondary prevention in patients with an established history of cardiovascular disease.
The new findings reported by Dr. Armitage came from additional analyses of dementia and cognitive impairment overlaid on the main ASCEND outcome analyses. ASCEND actively treated and followed study participants for an average of 7.4 years, then researchers tracked further dementia outcomes based on medical-record entries for an average of another 1.8 years.
ASCEND received partial funding or support from Abbott, Bayer, Mylan, and Solvay. Dr. Armitage had no disclosures. Dr. Towfighi, Dr. Khera, and Dr. Michos had no disclosures. Dr. Ballantyne has had financial relationships with numerous companies.
FROM AHA 2021
In diabetes, fast-growing pancreatic cysts may be a red flag
LAS VEGAS – New results from a single center, retrospective analysis suggest that individuals with diabetes and pancreatic cysts have larger cyst sizes at diagnosis, and a faster subsequent cyst growth rate. Smoking was independently associated with faster growth rate.
Most pancreatic cancer patients were previously diagnosed with hyperglycemia and diabetes, and pancreatic cancer can cause diabetes. “This sort of dual causality raises questions as to whether or not hyperglycemia, or the new diagnosis of diabetes itself, could be a harbinger of cancer or precancer. And should these patients be more closely monitored?” David Robbins, MD, said in an interview.
Dr. Robbins, associate professor of medicine and program director in gastroenterology in the Northwell Health System, New York, presented the study at the annual meeting of the American College of Gastroenterology.
Faster growth rates of pancreatic cysts in the presence of diabetes are important because they represent a potential mark for cyst aggressiveness. “So the question really is, in the setting of diabetes, are there factors perhaps circulating in the bloodstream, or other intrinsic factors, that make these cysts more dangerous and require a different surveillance approach than someone who doesn’t have diabetes? We have (surveillance) guidelines that address the average population, but they don’t really hone in on what do you do with (individuals with diabetes),” Dr. Robbins said during the presentation.
The study could have implications for screening, said session moderator Dayna Early, MD, professor of medicine at Washington University and director of endoscopy at Barnes Jewish Hospital, both in St. Louis. “I think this is important information to guide us to look more closely at patients with diabetes who do have pancreatic cysts,” she said in an interview.
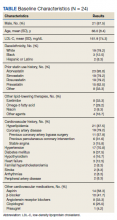
The study included 177 adults with pancreatic cysts or abnormal imaging results between 2013 and 2020. Sixty-five percent were female, and the mean age was 65.4 years; 64% were White, 10% were Black, and 8.5% were Asian. Among the participants, 24.8% were smokers and 32.2% had type 2 diabetes.
Patients with diabetes had larger cyst sizes (2.23 cm versus 2.76 cm), as well as a higher annual cyst growth rate (1.90 cm versus 1.30 cm). Cyst size and growth rate were similar between patients with controlled and uncontrolled diabetes. Smoking was associated with a larger cyst size overall (2.2 cm versus 1.81 cm), and were larger still among patients with diabetes who smoked (2.35 cm).
Seventy-one patients went on to have pathologic confirmation by endoscopic ultrasound-guided fine needle aspiration. “In the diabetic group, two developed adenocarcinoma, six of the nondiabetics developed adenocarcinoma, and there was no difference in CEA or serum CA 19-9,” Dr. Robbins said during his presentation.
Of 28 patients diagnosed with pancreatic cancer, 13 had type 2 diabetes.
Defining danger
There remains uncertainty about what cyst growth rate is most dangerous. Some guidelines recommend that individuals with new-onset or worsening diabetes and intraductal papillary mucinous neoplasm or mucinous cystic neoplasm cysts, or cysts alone that are growing faster than 3 mm per year, may be at significantly increased risk of pancreatic cancer. These guidelines recommend that they be screened with short-interval magnetic resonance imaging or endoscopic ultrasound (EUS) fine needle aspiration. However, this recommendation is conditional and is backed by a very low level of evidence.
Other reports have shown varying risks at different growth rates. “It’s not really clear at this point. And that’s why I think, while our study is small and exploratory, this is a particular area that is relatively easy to evaluate. We have huge databases of pancreatic cyst evolution, and we know that 30 million Americans have diabetes. So, the next obvious study is to do a more systematic look at that, and work towards refining and making sense of these divergent guidelines, all of which are saying the same thing but using different threshold numbers,” said Dr. Robbins.
The next step is do larger, multicenter studies in the context of other risk factors such as family history and smoking, but the current finding represents an opportunity to catch at least some pancreatic cancers earlier, according to Dr. Robbins. He suggested that individuals with diabetes who are diagnosed with a pancreatic cyst should be referred to a gastroenterologist or another specialist to track cyst growth. “That is going to miss a lot of folks who didn’t get imaging for whatever reason (and so don’t have a cyst identified), but it is an early opportunity, and it’s better than what we’re doing now.”
During the talk, Dr. Robbins said, “Given the ease, availability and low cost of diabetes screening in the general clinic population, we encourage the inclusion of HbA1c and fasting glucose in algorithms for pancreatic cyst surveillance.”
Dr. Early found the suggestion intriguing, but wasn’t ready to lend full support. “I think looking at the suggestion of possibly monitoring hemoglobin A1c levels was novel. I don’t know that we’ll necessarily adopt that as standard practice, but that’s something I think that could be looked at in the future as a way to help risk stratify whether patients need to be surveyed more frequently,” she said.
Dr. Robbins and Dr. Early have no relevant financial disclosures.
LAS VEGAS – New results from a single center, retrospective analysis suggest that individuals with diabetes and pancreatic cysts have larger cyst sizes at diagnosis, and a faster subsequent cyst growth rate. Smoking was independently associated with faster growth rate.
Most pancreatic cancer patients were previously diagnosed with hyperglycemia and diabetes, and pancreatic cancer can cause diabetes. “This sort of dual causality raises questions as to whether or not hyperglycemia, or the new diagnosis of diabetes itself, could be a harbinger of cancer or precancer. And should these patients be more closely monitored?” David Robbins, MD, said in an interview.
Dr. Robbins, associate professor of medicine and program director in gastroenterology in the Northwell Health System, New York, presented the study at the annual meeting of the American College of Gastroenterology.
Faster growth rates of pancreatic cysts in the presence of diabetes are important because they represent a potential mark for cyst aggressiveness. “So the question really is, in the setting of diabetes, are there factors perhaps circulating in the bloodstream, or other intrinsic factors, that make these cysts more dangerous and require a different surveillance approach than someone who doesn’t have diabetes? We have (surveillance) guidelines that address the average population, but they don’t really hone in on what do you do with (individuals with diabetes),” Dr. Robbins said during the presentation.
The study could have implications for screening, said session moderator Dayna Early, MD, professor of medicine at Washington University and director of endoscopy at Barnes Jewish Hospital, both in St. Louis. “I think this is important information to guide us to look more closely at patients with diabetes who do have pancreatic cysts,” she said in an interview.
The study included 177 adults with pancreatic cysts or abnormal imaging results between 2013 and 2020. Sixty-five percent were female, and the mean age was 65.4 years; 64% were White, 10% were Black, and 8.5% were Asian. Among the participants, 24.8% were smokers and 32.2% had type 2 diabetes.
Patients with diabetes had larger cyst sizes (2.23 cm versus 2.76 cm), as well as a higher annual cyst growth rate (1.90 cm versus 1.30 cm). Cyst size and growth rate were similar between patients with controlled and uncontrolled diabetes. Smoking was associated with a larger cyst size overall (2.2 cm versus 1.81 cm), and were larger still among patients with diabetes who smoked (2.35 cm).
Seventy-one patients went on to have pathologic confirmation by endoscopic ultrasound-guided fine needle aspiration. “In the diabetic group, two developed adenocarcinoma, six of the nondiabetics developed adenocarcinoma, and there was no difference in CEA or serum CA 19-9,” Dr. Robbins said during his presentation.
Of 28 patients diagnosed with pancreatic cancer, 13 had type 2 diabetes.
Defining danger
There remains uncertainty about what cyst growth rate is most dangerous. Some guidelines recommend that individuals with new-onset or worsening diabetes and intraductal papillary mucinous neoplasm or mucinous cystic neoplasm cysts, or cysts alone that are growing faster than 3 mm per year, may be at significantly increased risk of pancreatic cancer. These guidelines recommend that they be screened with short-interval magnetic resonance imaging or endoscopic ultrasound (EUS) fine needle aspiration. However, this recommendation is conditional and is backed by a very low level of evidence.
Other reports have shown varying risks at different growth rates. “It’s not really clear at this point. And that’s why I think, while our study is small and exploratory, this is a particular area that is relatively easy to evaluate. We have huge databases of pancreatic cyst evolution, and we know that 30 million Americans have diabetes. So, the next obvious study is to do a more systematic look at that, and work towards refining and making sense of these divergent guidelines, all of which are saying the same thing but using different threshold numbers,” said Dr. Robbins.
The next step is do larger, multicenter studies in the context of other risk factors such as family history and smoking, but the current finding represents an opportunity to catch at least some pancreatic cancers earlier, according to Dr. Robbins. He suggested that individuals with diabetes who are diagnosed with a pancreatic cyst should be referred to a gastroenterologist or another specialist to track cyst growth. “That is going to miss a lot of folks who didn’t get imaging for whatever reason (and so don’t have a cyst identified), but it is an early opportunity, and it’s better than what we’re doing now.”
During the talk, Dr. Robbins said, “Given the ease, availability and low cost of diabetes screening in the general clinic population, we encourage the inclusion of HbA1c and fasting glucose in algorithms for pancreatic cyst surveillance.”
Dr. Early found the suggestion intriguing, but wasn’t ready to lend full support. “I think looking at the suggestion of possibly monitoring hemoglobin A1c levels was novel. I don’t know that we’ll necessarily adopt that as standard practice, but that’s something I think that could be looked at in the future as a way to help risk stratify whether patients need to be surveyed more frequently,” she said.
Dr. Robbins and Dr. Early have no relevant financial disclosures.
LAS VEGAS – New results from a single center, retrospective analysis suggest that individuals with diabetes and pancreatic cysts have larger cyst sizes at diagnosis, and a faster subsequent cyst growth rate. Smoking was independently associated with faster growth rate.
Most pancreatic cancer patients were previously diagnosed with hyperglycemia and diabetes, and pancreatic cancer can cause diabetes. “This sort of dual causality raises questions as to whether or not hyperglycemia, or the new diagnosis of diabetes itself, could be a harbinger of cancer or precancer. And should these patients be more closely monitored?” David Robbins, MD, said in an interview.
Dr. Robbins, associate professor of medicine and program director in gastroenterology in the Northwell Health System, New York, presented the study at the annual meeting of the American College of Gastroenterology.
Faster growth rates of pancreatic cysts in the presence of diabetes are important because they represent a potential mark for cyst aggressiveness. “So the question really is, in the setting of diabetes, are there factors perhaps circulating in the bloodstream, or other intrinsic factors, that make these cysts more dangerous and require a different surveillance approach than someone who doesn’t have diabetes? We have (surveillance) guidelines that address the average population, but they don’t really hone in on what do you do with (individuals with diabetes),” Dr. Robbins said during the presentation.
The study could have implications for screening, said session moderator Dayna Early, MD, professor of medicine at Washington University and director of endoscopy at Barnes Jewish Hospital, both in St. Louis. “I think this is important information to guide us to look more closely at patients with diabetes who do have pancreatic cysts,” she said in an interview.
The study included 177 adults with pancreatic cysts or abnormal imaging results between 2013 and 2020. Sixty-five percent were female, and the mean age was 65.4 years; 64% were White, 10% were Black, and 8.5% were Asian. Among the participants, 24.8% were smokers and 32.2% had type 2 diabetes.
Patients with diabetes had larger cyst sizes (2.23 cm versus 2.76 cm), as well as a higher annual cyst growth rate (1.90 cm versus 1.30 cm). Cyst size and growth rate were similar between patients with controlled and uncontrolled diabetes. Smoking was associated with a larger cyst size overall (2.2 cm versus 1.81 cm), and were larger still among patients with diabetes who smoked (2.35 cm).
Seventy-one patients went on to have pathologic confirmation by endoscopic ultrasound-guided fine needle aspiration. “In the diabetic group, two developed adenocarcinoma, six of the nondiabetics developed adenocarcinoma, and there was no difference in CEA or serum CA 19-9,” Dr. Robbins said during his presentation.
Of 28 patients diagnosed with pancreatic cancer, 13 had type 2 diabetes.
Defining danger
There remains uncertainty about what cyst growth rate is most dangerous. Some guidelines recommend that individuals with new-onset or worsening diabetes and intraductal papillary mucinous neoplasm or mucinous cystic neoplasm cysts, or cysts alone that are growing faster than 3 mm per year, may be at significantly increased risk of pancreatic cancer. These guidelines recommend that they be screened with short-interval magnetic resonance imaging or endoscopic ultrasound (EUS) fine needle aspiration. However, this recommendation is conditional and is backed by a very low level of evidence.
Other reports have shown varying risks at different growth rates. “It’s not really clear at this point. And that’s why I think, while our study is small and exploratory, this is a particular area that is relatively easy to evaluate. We have huge databases of pancreatic cyst evolution, and we know that 30 million Americans have diabetes. So, the next obvious study is to do a more systematic look at that, and work towards refining and making sense of these divergent guidelines, all of which are saying the same thing but using different threshold numbers,” said Dr. Robbins.
The next step is do larger, multicenter studies in the context of other risk factors such as family history and smoking, but the current finding represents an opportunity to catch at least some pancreatic cancers earlier, according to Dr. Robbins. He suggested that individuals with diabetes who are diagnosed with a pancreatic cyst should be referred to a gastroenterologist or another specialist to track cyst growth. “That is going to miss a lot of folks who didn’t get imaging for whatever reason (and so don’t have a cyst identified), but it is an early opportunity, and it’s better than what we’re doing now.”
During the talk, Dr. Robbins said, “Given the ease, availability and low cost of diabetes screening in the general clinic population, we encourage the inclusion of HbA1c and fasting glucose in algorithms for pancreatic cyst surveillance.”
Dr. Early found the suggestion intriguing, but wasn’t ready to lend full support. “I think looking at the suggestion of possibly monitoring hemoglobin A1c levels was novel. I don’t know that we’ll necessarily adopt that as standard practice, but that’s something I think that could be looked at in the future as a way to help risk stratify whether patients need to be surveyed more frequently,” she said.
Dr. Robbins and Dr. Early have no relevant financial disclosures.
AT ACG 2021
Finerenone, sotagliflozin exert heart failure benefits despite renal dysfunction
New analyses of trial results for the cardiorenal agents finerenone and sotagliflozin continued the pattern showing that they exert consistent heart failure benefits in patients who span a broad spectrum of renal function, further disproving the notion that more severe stages of chronic kidney disease preclude aggressive medical management.
Analysis of combined data from two pivotal trials of the nonsteroidal mineralocorticoid receptor antagonist (MRA) finerenone (Kerendia), which together enrolled more than 13,000 patients with type 2 diabetes and chronic kidney disease, showed in greater detail that .
That spectrum included patients with estimated glomerular filtration rates (eGFR) as low as 25 mL/min per 1.73m2 and patients with micro- or macroalbuminuria, as well as those with normal urinary albumin levels, Gerasimos Filippatos, MD, reported at the American Heart Association scientific sessions.
And in a separate, unrelated report, combined data from the two pivotal trials, with a total of nearly 12,000 patients with type 2 diabetes, for sotagliflozin (Zynquista), a novel and still unapproved agent that inhibits both the sodium-glucose cotransporter (SGLT) 1 and 2 enzymes, showed a consistent effect significantly reducing cardiovascular death, hospitalization for heart failure, or urgent heart failure outpatient events in patients with eGFR rates as low as 25 mL/min per 1.73m2, Deepak L. Bhatt, MD, reported at the meeting.
These two reports follow a third, presented just a week earlier during Kidney Week, that showed the benefit from the SGLT2 inhibitor empagliflozin (Jardiance) for preventing heart failure hospitalizations or cardiovascular death in patients with heart failure with preserved ejection fraction remained consistent even in patients with an eGFR as low as 20 mL/min/1.73m2 in results from the EMPEROR-Preserved trial. Similar findings for empagliflozin in patients with heart failure with reduced ejection fraction in the EMPEROR-Reduced trial came out nearly a year ago.
A message to clinicians from these reports is, “don’t wait for patients to develop heart failure” to start these drugs, according to Dipti Itchhaporia, MD, director of disease management for the Hoag Heart and Vascular Institute in Newport Beach, Calif. “It’s time to start using these drugs upstream to have fewer patients with heart failure downstream,” she said in an interview.
Finerenone works differently than spironolactone
The new finerenone analysis included 5,734 patients enrolled in the FIDELIO-DKD trial, and 7,437 in the FIGARO-DKD trial, two very similar trials that differed by transposing the primary endpoint of one to the secondary endpoint of the other, and vice versa. The combined analysis is known as FIDELITY.
Expanding on a report that he first gave at the European Society of Cardiology annual congress in August 2021, Dr. Filippatos provided a few additional details on the analysis that showed a consistent effect of finerenone on preventing hospitalizations for heart failure, and on preventing a combined endpoint of hospitalizations for heart failure and cardiovascular death regardless of the severity of chronic kidney disease down to 25 mL/min per 1.73 m2. Statistical analysis showed no hint of an interaction between finerenone’s effect on these outcomes in patients with an eGFR of 60 mL/min per 1.73 m2 or greater and those with reduced renal function. Analyses also showed no interaction based on urinary albumin-to-creatinine ratio, be it more or less than 300 mg/g, reported Dr. Filippatos, professor and director of the heart failure unit at Attikon University Hospital in Athens.
“We use MRAs [such as spironolactone] in heart failure patients, but it’s difficult to use because of the risk of patients developing hyperkalemia,” noted Dr. Itchhaporia, who added that reluctance to use spironolactone is especially high for patients with depressed renal function, which could exacerbate a hyperkalemic effect. Evidence shows that finerenone poses a substantially reduced risk for raising serum potassium levels, making finerenone a more attractive agent to use in patients with CKD who have an elevated risk for heart failure events as well as an increased risk for hyperkalemia, like those enrolled in the two finerenone trials, she said.
Sotagliflozin uniquely inhibits SGLT1 and SGLT2
The new sotagliflozin analyses reported by Dr. Bhatt combined data for more than 11,800 patients randomized into either of two trials, SCORED, which randomized more than 10,000 patients with type 2 diabetes and chronic kidney disease, and SOLOIST, which randomized more than 1,000 patients with type 2 diabetes who were recently hospitalized for worsening heart failure.
A prespecified analysis for the combined data from both studies looked at the impact of sotagliflozin treatment on the combined outcome of cardiovascular death, hospitalization for heart failure, or an urgent outpatient visit because of heart failure based on kidney function at baseline. The analysis showed that sotagliflozin was at least as effective in the 8% of study patients who at baseline had an eGFR of 25-29 mL/min per 1.73 m2 as it was in patients with more preserved renal function.
Benefit from sotagliflozin treatment “was consistent across the full range of eGFR,” said Dr. Bhatt, professor at Harvard Medical School in Boston and executive director of interventional cardiovascular programs at Brigham and Women’s Hospital in Boston.
Results from a second analysis that he reported also showed a consistent effect of sotagliflozin on reducing hemoglobin A1c levels in the enrolled patients, even those with the lowest levels of renal function, an effect not previously seen with the related class of SGLT2 inhibitors (which includes empagliflozin, canagliflozin [Invokana], and dapagliflozin [Farxiga]). Dr. Bhatt suggested that, while SGLT2 inhibitors act entirely in the kidneys and hence their effect on glycemic control is blunted by renal dysfunction, sotagliflozin also inhibits the SGLT1 enzyme, which functions in the gut to transport glucose out of the digestive tract and into the blood, a glycemic control pathway that’s independent of renal function.
FIDELIO-DKD, FIGARO-DKD, and FIDELITY were sponsored by Bayer, the company that markets finerenone (Kerendia). SCORED and SOLOIST were sponsored by Sanofi, and later by Lexicon, the companies developing sotagliflozin (Zynquista). EMPEROR-Preserved and EMPEROR-Reduced were sponsored by Boehringer-Ingelheim and Lilly, the companies that market empagliflozin (Jardiance). Dr. Filippatos has had financial relationships with Bayer and Boehringer-Ingelheim, as well as with Amgen, Medtronic, Novartis, Servier, and Vifor. Dr. Bhatt has received research funding from Sanofi, Lexicon, Bayer, and Boehringer-Ingelheim, Lilly, and numerous other companies, and he has been an adviser to Boehringer-Ingelheim and several other companies.
New analyses of trial results for the cardiorenal agents finerenone and sotagliflozin continued the pattern showing that they exert consistent heart failure benefits in patients who span a broad spectrum of renal function, further disproving the notion that more severe stages of chronic kidney disease preclude aggressive medical management.
Analysis of combined data from two pivotal trials of the nonsteroidal mineralocorticoid receptor antagonist (MRA) finerenone (Kerendia), which together enrolled more than 13,000 patients with type 2 diabetes and chronic kidney disease, showed in greater detail that .
That spectrum included patients with estimated glomerular filtration rates (eGFR) as low as 25 mL/min per 1.73m2 and patients with micro- or macroalbuminuria, as well as those with normal urinary albumin levels, Gerasimos Filippatos, MD, reported at the American Heart Association scientific sessions.
And in a separate, unrelated report, combined data from the two pivotal trials, with a total of nearly 12,000 patients with type 2 diabetes, for sotagliflozin (Zynquista), a novel and still unapproved agent that inhibits both the sodium-glucose cotransporter (SGLT) 1 and 2 enzymes, showed a consistent effect significantly reducing cardiovascular death, hospitalization for heart failure, or urgent heart failure outpatient events in patients with eGFR rates as low as 25 mL/min per 1.73m2, Deepak L. Bhatt, MD, reported at the meeting.
These two reports follow a third, presented just a week earlier during Kidney Week, that showed the benefit from the SGLT2 inhibitor empagliflozin (Jardiance) for preventing heart failure hospitalizations or cardiovascular death in patients with heart failure with preserved ejection fraction remained consistent even in patients with an eGFR as low as 20 mL/min/1.73m2 in results from the EMPEROR-Preserved trial. Similar findings for empagliflozin in patients with heart failure with reduced ejection fraction in the EMPEROR-Reduced trial came out nearly a year ago.
A message to clinicians from these reports is, “don’t wait for patients to develop heart failure” to start these drugs, according to Dipti Itchhaporia, MD, director of disease management for the Hoag Heart and Vascular Institute in Newport Beach, Calif. “It’s time to start using these drugs upstream to have fewer patients with heart failure downstream,” she said in an interview.
Finerenone works differently than spironolactone
The new finerenone analysis included 5,734 patients enrolled in the FIDELIO-DKD trial, and 7,437 in the FIGARO-DKD trial, two very similar trials that differed by transposing the primary endpoint of one to the secondary endpoint of the other, and vice versa. The combined analysis is known as FIDELITY.
Expanding on a report that he first gave at the European Society of Cardiology annual congress in August 2021, Dr. Filippatos provided a few additional details on the analysis that showed a consistent effect of finerenone on preventing hospitalizations for heart failure, and on preventing a combined endpoint of hospitalizations for heart failure and cardiovascular death regardless of the severity of chronic kidney disease down to 25 mL/min per 1.73 m2. Statistical analysis showed no hint of an interaction between finerenone’s effect on these outcomes in patients with an eGFR of 60 mL/min per 1.73 m2 or greater and those with reduced renal function. Analyses also showed no interaction based on urinary albumin-to-creatinine ratio, be it more or less than 300 mg/g, reported Dr. Filippatos, professor and director of the heart failure unit at Attikon University Hospital in Athens.
“We use MRAs [such as spironolactone] in heart failure patients, but it’s difficult to use because of the risk of patients developing hyperkalemia,” noted Dr. Itchhaporia, who added that reluctance to use spironolactone is especially high for patients with depressed renal function, which could exacerbate a hyperkalemic effect. Evidence shows that finerenone poses a substantially reduced risk for raising serum potassium levels, making finerenone a more attractive agent to use in patients with CKD who have an elevated risk for heart failure events as well as an increased risk for hyperkalemia, like those enrolled in the two finerenone trials, she said.
Sotagliflozin uniquely inhibits SGLT1 and SGLT2
The new sotagliflozin analyses reported by Dr. Bhatt combined data for more than 11,800 patients randomized into either of two trials, SCORED, which randomized more than 10,000 patients with type 2 diabetes and chronic kidney disease, and SOLOIST, which randomized more than 1,000 patients with type 2 diabetes who were recently hospitalized for worsening heart failure.
A prespecified analysis for the combined data from both studies looked at the impact of sotagliflozin treatment on the combined outcome of cardiovascular death, hospitalization for heart failure, or an urgent outpatient visit because of heart failure based on kidney function at baseline. The analysis showed that sotagliflozin was at least as effective in the 8% of study patients who at baseline had an eGFR of 25-29 mL/min per 1.73 m2 as it was in patients with more preserved renal function.
Benefit from sotagliflozin treatment “was consistent across the full range of eGFR,” said Dr. Bhatt, professor at Harvard Medical School in Boston and executive director of interventional cardiovascular programs at Brigham and Women’s Hospital in Boston.
Results from a second analysis that he reported also showed a consistent effect of sotagliflozin on reducing hemoglobin A1c levels in the enrolled patients, even those with the lowest levels of renal function, an effect not previously seen with the related class of SGLT2 inhibitors (which includes empagliflozin, canagliflozin [Invokana], and dapagliflozin [Farxiga]). Dr. Bhatt suggested that, while SGLT2 inhibitors act entirely in the kidneys and hence their effect on glycemic control is blunted by renal dysfunction, sotagliflozin also inhibits the SGLT1 enzyme, which functions in the gut to transport glucose out of the digestive tract and into the blood, a glycemic control pathway that’s independent of renal function.
FIDELIO-DKD, FIGARO-DKD, and FIDELITY were sponsored by Bayer, the company that markets finerenone (Kerendia). SCORED and SOLOIST were sponsored by Sanofi, and later by Lexicon, the companies developing sotagliflozin (Zynquista). EMPEROR-Preserved and EMPEROR-Reduced were sponsored by Boehringer-Ingelheim and Lilly, the companies that market empagliflozin (Jardiance). Dr. Filippatos has had financial relationships with Bayer and Boehringer-Ingelheim, as well as with Amgen, Medtronic, Novartis, Servier, and Vifor. Dr. Bhatt has received research funding from Sanofi, Lexicon, Bayer, and Boehringer-Ingelheim, Lilly, and numerous other companies, and he has been an adviser to Boehringer-Ingelheim and several other companies.
New analyses of trial results for the cardiorenal agents finerenone and sotagliflozin continued the pattern showing that they exert consistent heart failure benefits in patients who span a broad spectrum of renal function, further disproving the notion that more severe stages of chronic kidney disease preclude aggressive medical management.
Analysis of combined data from two pivotal trials of the nonsteroidal mineralocorticoid receptor antagonist (MRA) finerenone (Kerendia), which together enrolled more than 13,000 patients with type 2 diabetes and chronic kidney disease, showed in greater detail that .
That spectrum included patients with estimated glomerular filtration rates (eGFR) as low as 25 mL/min per 1.73m2 and patients with micro- or macroalbuminuria, as well as those with normal urinary albumin levels, Gerasimos Filippatos, MD, reported at the American Heart Association scientific sessions.
And in a separate, unrelated report, combined data from the two pivotal trials, with a total of nearly 12,000 patients with type 2 diabetes, for sotagliflozin (Zynquista), a novel and still unapproved agent that inhibits both the sodium-glucose cotransporter (SGLT) 1 and 2 enzymes, showed a consistent effect significantly reducing cardiovascular death, hospitalization for heart failure, or urgent heart failure outpatient events in patients with eGFR rates as low as 25 mL/min per 1.73m2, Deepak L. Bhatt, MD, reported at the meeting.
These two reports follow a third, presented just a week earlier during Kidney Week, that showed the benefit from the SGLT2 inhibitor empagliflozin (Jardiance) for preventing heart failure hospitalizations or cardiovascular death in patients with heart failure with preserved ejection fraction remained consistent even in patients with an eGFR as low as 20 mL/min/1.73m2 in results from the EMPEROR-Preserved trial. Similar findings for empagliflozin in patients with heart failure with reduced ejection fraction in the EMPEROR-Reduced trial came out nearly a year ago.
A message to clinicians from these reports is, “don’t wait for patients to develop heart failure” to start these drugs, according to Dipti Itchhaporia, MD, director of disease management for the Hoag Heart and Vascular Institute in Newport Beach, Calif. “It’s time to start using these drugs upstream to have fewer patients with heart failure downstream,” she said in an interview.
Finerenone works differently than spironolactone
The new finerenone analysis included 5,734 patients enrolled in the FIDELIO-DKD trial, and 7,437 in the FIGARO-DKD trial, two very similar trials that differed by transposing the primary endpoint of one to the secondary endpoint of the other, and vice versa. The combined analysis is known as FIDELITY.
Expanding on a report that he first gave at the European Society of Cardiology annual congress in August 2021, Dr. Filippatos provided a few additional details on the analysis that showed a consistent effect of finerenone on preventing hospitalizations for heart failure, and on preventing a combined endpoint of hospitalizations for heart failure and cardiovascular death regardless of the severity of chronic kidney disease down to 25 mL/min per 1.73 m2. Statistical analysis showed no hint of an interaction between finerenone’s effect on these outcomes in patients with an eGFR of 60 mL/min per 1.73 m2 or greater and those with reduced renal function. Analyses also showed no interaction based on urinary albumin-to-creatinine ratio, be it more or less than 300 mg/g, reported Dr. Filippatos, professor and director of the heart failure unit at Attikon University Hospital in Athens.
“We use MRAs [such as spironolactone] in heart failure patients, but it’s difficult to use because of the risk of patients developing hyperkalemia,” noted Dr. Itchhaporia, who added that reluctance to use spironolactone is especially high for patients with depressed renal function, which could exacerbate a hyperkalemic effect. Evidence shows that finerenone poses a substantially reduced risk for raising serum potassium levels, making finerenone a more attractive agent to use in patients with CKD who have an elevated risk for heart failure events as well as an increased risk for hyperkalemia, like those enrolled in the two finerenone trials, she said.
Sotagliflozin uniquely inhibits SGLT1 and SGLT2
The new sotagliflozin analyses reported by Dr. Bhatt combined data for more than 11,800 patients randomized into either of two trials, SCORED, which randomized more than 10,000 patients with type 2 diabetes and chronic kidney disease, and SOLOIST, which randomized more than 1,000 patients with type 2 diabetes who were recently hospitalized for worsening heart failure.
A prespecified analysis for the combined data from both studies looked at the impact of sotagliflozin treatment on the combined outcome of cardiovascular death, hospitalization for heart failure, or an urgent outpatient visit because of heart failure based on kidney function at baseline. The analysis showed that sotagliflozin was at least as effective in the 8% of study patients who at baseline had an eGFR of 25-29 mL/min per 1.73 m2 as it was in patients with more preserved renal function.
Benefit from sotagliflozin treatment “was consistent across the full range of eGFR,” said Dr. Bhatt, professor at Harvard Medical School in Boston and executive director of interventional cardiovascular programs at Brigham and Women’s Hospital in Boston.
Results from a second analysis that he reported also showed a consistent effect of sotagliflozin on reducing hemoglobin A1c levels in the enrolled patients, even those with the lowest levels of renal function, an effect not previously seen with the related class of SGLT2 inhibitors (which includes empagliflozin, canagliflozin [Invokana], and dapagliflozin [Farxiga]). Dr. Bhatt suggested that, while SGLT2 inhibitors act entirely in the kidneys and hence their effect on glycemic control is blunted by renal dysfunction, sotagliflozin also inhibits the SGLT1 enzyme, which functions in the gut to transport glucose out of the digestive tract and into the blood, a glycemic control pathway that’s independent of renal function.
FIDELIO-DKD, FIGARO-DKD, and FIDELITY were sponsored by Bayer, the company that markets finerenone (Kerendia). SCORED and SOLOIST were sponsored by Sanofi, and later by Lexicon, the companies developing sotagliflozin (Zynquista). EMPEROR-Preserved and EMPEROR-Reduced were sponsored by Boehringer-Ingelheim and Lilly, the companies that market empagliflozin (Jardiance). Dr. Filippatos has had financial relationships with Bayer and Boehringer-Ingelheim, as well as with Amgen, Medtronic, Novartis, Servier, and Vifor. Dr. Bhatt has received research funding from Sanofi, Lexicon, Bayer, and Boehringer-Ingelheim, Lilly, and numerous other companies, and he has been an adviser to Boehringer-Ingelheim and several other companies.
FROM AHA 2021
More STEP data: Semaglutide cuts weight, cravings, beats liraglutide
The STEP 5 clinical trial extends favorable weight loss from 1 year out to 2 years for the glucagon-like peptide-1 (GLP-1) agonist semaglutide (Wegovy, Novo Nordisk), given as a once-weekly 2.4-mg subcutaneous injection, and some food cravings were improved in a subgroup analysis.
In another study, STEP 8, weight loss was greater at 68 weeks with semaglutide subcutaneous injection than with a 3-mg daily subcutaneous injection of another GLP-1 agonist, liraglutide (Saxenda, Novo Nordisk), approved earlier for weight loss.
Researchers presented these promising outcomes, with no new safety signals, at ObesityWeek® 2021.
However, there is more to learn about the drug class, researchers agree. Follow-up is still relatively short for a chronic disease and many patients have gastrointestinal side effects with semaglutide, one expert cautions.
The key findings were:
In STEP 5, combined with lifestyle intervention (a reduced-calorie meal plan and advice about physical activity), weekly injection of 2.4 mg semaglutide led to:
- 15.2% weight loss, compared with 2.6% weight loss with placebo at 2 years (P < .0001);
- 77% of patients losing at least 5% of their weight, compared with 34% of patients in the placebo group at 2 years (P < .0001);
- Significantly greater improvement in overall control of cravings, and craving for savory foods, in a subset of patients, versus placebo, but questionnaire scores for positive mood and craving for sweet foods were similar in both groups.
- In STEP 8, mean body weight at 68 weeks was 15.8% lower with 2.4 mg/week subcutaneous semaglutide plus lifestyle changes versus 6.4% lower with 3.0 mg/day subcutaneous liraglutide plus lifestyle changes (P < .001).
Can treat to a target weight-loss range
The undiminished weight loss efficacy in the 2-year data for STEP 5 “portends well,” said W. Timothy Garvey, MD, following his presentation of the results.
“I think this is a new era in obesity care,” said Dr. Garvey, director of the diabetes research center at the University of Alabama at Birmingham. Semaglutide “essentially doubles weight loss efficacy” compared to the other approved pharmacotherapies for obesity.
With this degree of potential weight loss, clinicians “can use weight as a biomarker and treat to a target [weight-loss] range,” he said.
Expounding on this in an interview, Dr. Garvey noted that, as stated in the 2016 American Association of Clinical Endocrinologists (AACE) and American College of Endocrinology (ACE) clinical practice guidelines for medical care of patients with obesity, of which he was lead author, “the objective of care in obesity is to increase health of patients and prevent or treat complications.”
Semaglutide “can treat to a range of weight loss of 10% to 20% in the majority of patients,” which is associated with improvements in cardiovascular and metabolic risk factors.
In STEP 5, of the 51% of patients in the semaglutide group who had prediabetes at enrollment, 80% had normal glycemia at 2 years; however, the trial was not powered nor designed to investigate this.
More data are needed to inform long-term care decisions. The ongoing SELECT cardiovascular outcomes trial of semaglutide, with expected primary study completion on Sept. 28, 2023, should provide more information.
Weight loss plus reduced cravings
In another presentation, Sean Wharton, MD, PharmD, said, “In adults with overweight or obesity, substantial weight loss with semaglutide 2.4 mg was accompanied by short- and long-term improvements in control of eating.”
“Most patients living with obesity who are attempting to decrease calories will have food cravings, based on the biological parameters of weight preservation,” Dr. Wharton, medical director at the Wharton Medical Clinic, in Hamilton, Ont., explained in an email.
The degree of craving varies from patient to patient, likely based on genetics, he added. Research in this field is still emerging.
“I believe that semaglutide 2.4 mg is a game-changer in the field of weight management, and it will change the dialogue for insurance plans and with policymakers regarding coverage for this medication,” said Dr. Wharton.
“The data from the STEP programs are very strong. I am certainly hoping for a change to bias against covering these medications that we have seen in the past,” he said.
Clinically meaningful weight loss
When presenting the STEP 8 findings, Domenica M. Rubino, MD, said: “Participants were significantly more likely to achieve clinically meaningful weight loss thresholds with semaglutide 2.4 mg versus liraglutide 3.0 mg, accompanied by greater improvements in cardiometabolic risk factors.”
For example, patients can have better mobility, which is important for quality of life, Dr. Rubino, director of the Washington Center for Weight Management and Research, Arlington, Virginia, noted.
A smaller percentage of patients respond to liraglutide, she added. Clinicians need to individualize treatment.
When asked, “How do you choose which medical therapy?” Dr. Rubino responded: “We sit and talk.” Finding the medical therapy that fits the patient depends on things such as the patient’s insurance coverage and ability to tolerate side effects such as dehydration, diarrhea, and nausea.
When asked, “How do you switch from liraglutide to semaglutide?” she noted that there are no current guidelines for this. “You have to be careful. Start on the lowest dose of Wegovy. Be cautious, conservative.”
Still early days, caveats remain
“The STEP trials as a group appear to be making the case that obesity may now be considered a medically manageable disease, based on the experience with semaglutide,” Julie R. Ingelfinger, MD, who was not involved with the research, commented in an email.
“STEP 5 and 8 may suggest that weight loss occurs and is sustainable in overweight persons without diabetes with one or more comorbidities or in obese persons without diabetes,” added Dr. Ingelfinger, professor of pediatrics, Harvard Medical School, consultant in pediatric nephrology, Massachusetts General Hospital, Boston, and deputy editor, The New England Journal of Medicine.
However, “even 2 years, in the case of STEP 5, and ~68 weeks in the case of STEP 8, may not be long enough to know whether semaglutide is as promising as these brief summaries (abstracts) suggest,” she cautioned.
“Obesity is a chronic condition, and very long-term therapy and management are required,” Dr. Ingelfinger continued.
“Further, it is hard to generalize when gastrointestinal adverse events are common in a study,” she said. For example, in STEP 8, they were just as common with semaglutide as with the comparator liraglutide, she noted.
“The racial and ethnic representativeness of these studies does not reflect population distributions in the U.S., limiting generalization,” she continued.
“So, there remain caveats in interpreting these data.”
STEP 5 weight loss efficacy and safety at 2 years
Garvey reported that STEP 5 was a phase 3b trial that randomized 304 adults in the United States, Canada, Hungary, Italy, and Spain, who were 18 years and older, with a body mass index (BMI) ≥27 kg/m2 with at least one weight-related comorbidity (hypertension, dyslipidemia, obstructive sleep apnea, or cardiovascular disease) or a BMI ≥30 kg/m2, without type 2 diabetes, to receive semaglutide or placebo plus lifestyle intervention.
Most participants were women (78%) and White (93%). On average, they were 47 years old, weighed 106 kg (223.7 pounds), had a BMI of 38.5 kg/m2, a waist circumference of 115.7 cm (45.6 inches), and an A1c of 5.7%.
A total of 87% of patients in the semaglutide group and 73% of patients in the placebo group completed the trial.
At 104 weeks, participants were more likely to lose ≥10%, ≥15%, and ≥20% of body weight with semaglutide versus placebo (61.8% vs. 13.3%, 52.1% vs. 7.0%, and 36.1% vs. 2.3%, respectively; P < .0001 for all).
Patients in the semaglutide group had greater health improvements in cardiovascular risk factors (waist circumference, systolic and diastolic blood pressure, and C-reactive protein) and metabolic risk factors (A1c, fasting plasma glucose, fasting serum insulin, and triglycerides) than those in the placebo group (P < .05 for all).
Safety and tolerability were consistent with adverse events seen with this drug class, with no new safety signals.
Control of eating questionnaire findings at 2 years in STEP 5
Dr. Wharton and colleagues assessed changes in responses to the Control of Eating questionnaire at baseline and at 20, 52, and 104 weeks in patients from the U.S. and Canada in the STEP 5 trial (88 patients in the semaglutide group and 86 patients in the placebo group).
The questionnaire consisted of 19 questions grouped into four categories: control of food cravings, craving for savory foods (salty and spicy, dairy, or starchy foods), craving for sweet foods (chocolate, sweet foods, or fruit/fruit juice), and positive mood.
At week 104, patients in the semaglutide group had significantly greater improvements in scores for craving for salty and spicy, dairy, and starchy foods, and resisting cravings.
Semaglutide versus liraglutide, 68-week efficacy and safety in STEP 8
STEP 8 randomized 338 U.S. adults without diabetes and a BMI of ≥27 kg/m2 plus one or more weight-related comorbidities or a BMI of ≥30 kg/m2 3:1 to semaglutide 2.4 mg once weekly (n = 126) or matching placebo, or 3:1 liraglutide 3.0 mg once daily (n = 127) or matching placebo, plus lifestyle intervention.
Most participants were women (78%) and were a mean age of 49, had a mean body weight of 104.5 kg, and had a mean BMI of 37.5 kg/m2.
In STEP 8, more participants achieved ≥10%, ≥15%, and ≥20% weight loss with semaglutide than with liraglutide (70.9% vs. 25.6%, 55.6% vs. 12.0%, and 38.5% vs. 6.0%, respectively; P < .001 for all odds ratios).
Semaglutide improved waist circumference, A1c, and C-reactive protein versus liraglutide (unadjusted P < .001 for all).
Gastrointestinal adverse events were reported by 84% and 83% of participants receiving semaglutide and liraglutide, respectively. Most events were mild/moderate and transient, with prevalence declining over time.
Fewer participants stopped treatment due to adverse events with semaglutide than liraglutide (3.2% vs. 12.6%).
Dr. Garvey has reported serving as a site principal investigator for multicentered clinical trials sponsored by his university and funded by Eli Lilly, Novo Nordisk, and Pfizer. Dr. Wharton has reported financial ties to Novo Nordisk, Bausch Health Canada, Eli Lily, and Boehringer Ingelheim Canada. Dr. Rubino has reported ties to Boehringer Ingelheim and AstraZeneca. Dr. Ingelfinger has reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The STEP 5 clinical trial extends favorable weight loss from 1 year out to 2 years for the glucagon-like peptide-1 (GLP-1) agonist semaglutide (Wegovy, Novo Nordisk), given as a once-weekly 2.4-mg subcutaneous injection, and some food cravings were improved in a subgroup analysis.
In another study, STEP 8, weight loss was greater at 68 weeks with semaglutide subcutaneous injection than with a 3-mg daily subcutaneous injection of another GLP-1 agonist, liraglutide (Saxenda, Novo Nordisk), approved earlier for weight loss.
Researchers presented these promising outcomes, with no new safety signals, at ObesityWeek® 2021.
However, there is more to learn about the drug class, researchers agree. Follow-up is still relatively short for a chronic disease and many patients have gastrointestinal side effects with semaglutide, one expert cautions.
The key findings were:
In STEP 5, combined with lifestyle intervention (a reduced-calorie meal plan and advice about physical activity), weekly injection of 2.4 mg semaglutide led to:
- 15.2% weight loss, compared with 2.6% weight loss with placebo at 2 years (P < .0001);
- 77% of patients losing at least 5% of their weight, compared with 34% of patients in the placebo group at 2 years (P < .0001);
- Significantly greater improvement in overall control of cravings, and craving for savory foods, in a subset of patients, versus placebo, but questionnaire scores for positive mood and craving for sweet foods were similar in both groups.
- In STEP 8, mean body weight at 68 weeks was 15.8% lower with 2.4 mg/week subcutaneous semaglutide plus lifestyle changes versus 6.4% lower with 3.0 mg/day subcutaneous liraglutide plus lifestyle changes (P < .001).
Can treat to a target weight-loss range
The undiminished weight loss efficacy in the 2-year data for STEP 5 “portends well,” said W. Timothy Garvey, MD, following his presentation of the results.
“I think this is a new era in obesity care,” said Dr. Garvey, director of the diabetes research center at the University of Alabama at Birmingham. Semaglutide “essentially doubles weight loss efficacy” compared to the other approved pharmacotherapies for obesity.
With this degree of potential weight loss, clinicians “can use weight as a biomarker and treat to a target [weight-loss] range,” he said.
Expounding on this in an interview, Dr. Garvey noted that, as stated in the 2016 American Association of Clinical Endocrinologists (AACE) and American College of Endocrinology (ACE) clinical practice guidelines for medical care of patients with obesity, of which he was lead author, “the objective of care in obesity is to increase health of patients and prevent or treat complications.”
Semaglutide “can treat to a range of weight loss of 10% to 20% in the majority of patients,” which is associated with improvements in cardiovascular and metabolic risk factors.
In STEP 5, of the 51% of patients in the semaglutide group who had prediabetes at enrollment, 80% had normal glycemia at 2 years; however, the trial was not powered nor designed to investigate this.
More data are needed to inform long-term care decisions. The ongoing SELECT cardiovascular outcomes trial of semaglutide, with expected primary study completion on Sept. 28, 2023, should provide more information.
Weight loss plus reduced cravings
In another presentation, Sean Wharton, MD, PharmD, said, “In adults with overweight or obesity, substantial weight loss with semaglutide 2.4 mg was accompanied by short- and long-term improvements in control of eating.”
“Most patients living with obesity who are attempting to decrease calories will have food cravings, based on the biological parameters of weight preservation,” Dr. Wharton, medical director at the Wharton Medical Clinic, in Hamilton, Ont., explained in an email.
The degree of craving varies from patient to patient, likely based on genetics, he added. Research in this field is still emerging.
“I believe that semaglutide 2.4 mg is a game-changer in the field of weight management, and it will change the dialogue for insurance plans and with policymakers regarding coverage for this medication,” said Dr. Wharton.
“The data from the STEP programs are very strong. I am certainly hoping for a change to bias against covering these medications that we have seen in the past,” he said.
Clinically meaningful weight loss
When presenting the STEP 8 findings, Domenica M. Rubino, MD, said: “Participants were significantly more likely to achieve clinically meaningful weight loss thresholds with semaglutide 2.4 mg versus liraglutide 3.0 mg, accompanied by greater improvements in cardiometabolic risk factors.”
For example, patients can have better mobility, which is important for quality of life, Dr. Rubino, director of the Washington Center for Weight Management and Research, Arlington, Virginia, noted.
A smaller percentage of patients respond to liraglutide, she added. Clinicians need to individualize treatment.
When asked, “How do you choose which medical therapy?” Dr. Rubino responded: “We sit and talk.” Finding the medical therapy that fits the patient depends on things such as the patient’s insurance coverage and ability to tolerate side effects such as dehydration, diarrhea, and nausea.
When asked, “How do you switch from liraglutide to semaglutide?” she noted that there are no current guidelines for this. “You have to be careful. Start on the lowest dose of Wegovy. Be cautious, conservative.”
Still early days, caveats remain
“The STEP trials as a group appear to be making the case that obesity may now be considered a medically manageable disease, based on the experience with semaglutide,” Julie R. Ingelfinger, MD, who was not involved with the research, commented in an email.
“STEP 5 and 8 may suggest that weight loss occurs and is sustainable in overweight persons without diabetes with one or more comorbidities or in obese persons without diabetes,” added Dr. Ingelfinger, professor of pediatrics, Harvard Medical School, consultant in pediatric nephrology, Massachusetts General Hospital, Boston, and deputy editor, The New England Journal of Medicine.
However, “even 2 years, in the case of STEP 5, and ~68 weeks in the case of STEP 8, may not be long enough to know whether semaglutide is as promising as these brief summaries (abstracts) suggest,” she cautioned.
“Obesity is a chronic condition, and very long-term therapy and management are required,” Dr. Ingelfinger continued.
“Further, it is hard to generalize when gastrointestinal adverse events are common in a study,” she said. For example, in STEP 8, they were just as common with semaglutide as with the comparator liraglutide, she noted.
“The racial and ethnic representativeness of these studies does not reflect population distributions in the U.S., limiting generalization,” she continued.
“So, there remain caveats in interpreting these data.”
STEP 5 weight loss efficacy and safety at 2 years
Garvey reported that STEP 5 was a phase 3b trial that randomized 304 adults in the United States, Canada, Hungary, Italy, and Spain, who were 18 years and older, with a body mass index (BMI) ≥27 kg/m2 with at least one weight-related comorbidity (hypertension, dyslipidemia, obstructive sleep apnea, or cardiovascular disease) or a BMI ≥30 kg/m2, without type 2 diabetes, to receive semaglutide or placebo plus lifestyle intervention.
Most participants were women (78%) and White (93%). On average, they were 47 years old, weighed 106 kg (223.7 pounds), had a BMI of 38.5 kg/m2, a waist circumference of 115.7 cm (45.6 inches), and an A1c of 5.7%.
A total of 87% of patients in the semaglutide group and 73% of patients in the placebo group completed the trial.
At 104 weeks, participants were more likely to lose ≥10%, ≥15%, and ≥20% of body weight with semaglutide versus placebo (61.8% vs. 13.3%, 52.1% vs. 7.0%, and 36.1% vs. 2.3%, respectively; P < .0001 for all).
Patients in the semaglutide group had greater health improvements in cardiovascular risk factors (waist circumference, systolic and diastolic blood pressure, and C-reactive protein) and metabolic risk factors (A1c, fasting plasma glucose, fasting serum insulin, and triglycerides) than those in the placebo group (P < .05 for all).
Safety and tolerability were consistent with adverse events seen with this drug class, with no new safety signals.
Control of eating questionnaire findings at 2 years in STEP 5
Dr. Wharton and colleagues assessed changes in responses to the Control of Eating questionnaire at baseline and at 20, 52, and 104 weeks in patients from the U.S. and Canada in the STEP 5 trial (88 patients in the semaglutide group and 86 patients in the placebo group).
The questionnaire consisted of 19 questions grouped into four categories: control of food cravings, craving for savory foods (salty and spicy, dairy, or starchy foods), craving for sweet foods (chocolate, sweet foods, or fruit/fruit juice), and positive mood.
At week 104, patients in the semaglutide group had significantly greater improvements in scores for craving for salty and spicy, dairy, and starchy foods, and resisting cravings.
Semaglutide versus liraglutide, 68-week efficacy and safety in STEP 8
STEP 8 randomized 338 U.S. adults without diabetes and a BMI of ≥27 kg/m2 plus one or more weight-related comorbidities or a BMI of ≥30 kg/m2 3:1 to semaglutide 2.4 mg once weekly (n = 126) or matching placebo, or 3:1 liraglutide 3.0 mg once daily (n = 127) or matching placebo, plus lifestyle intervention.
Most participants were women (78%) and were a mean age of 49, had a mean body weight of 104.5 kg, and had a mean BMI of 37.5 kg/m2.
In STEP 8, more participants achieved ≥10%, ≥15%, and ≥20% weight loss with semaglutide than with liraglutide (70.9% vs. 25.6%, 55.6% vs. 12.0%, and 38.5% vs. 6.0%, respectively; P < .001 for all odds ratios).
Semaglutide improved waist circumference, A1c, and C-reactive protein versus liraglutide (unadjusted P < .001 for all).
Gastrointestinal adverse events were reported by 84% and 83% of participants receiving semaglutide and liraglutide, respectively. Most events were mild/moderate and transient, with prevalence declining over time.
Fewer participants stopped treatment due to adverse events with semaglutide than liraglutide (3.2% vs. 12.6%).
Dr. Garvey has reported serving as a site principal investigator for multicentered clinical trials sponsored by his university and funded by Eli Lilly, Novo Nordisk, and Pfizer. Dr. Wharton has reported financial ties to Novo Nordisk, Bausch Health Canada, Eli Lily, and Boehringer Ingelheim Canada. Dr. Rubino has reported ties to Boehringer Ingelheim and AstraZeneca. Dr. Ingelfinger has reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The STEP 5 clinical trial extends favorable weight loss from 1 year out to 2 years for the glucagon-like peptide-1 (GLP-1) agonist semaglutide (Wegovy, Novo Nordisk), given as a once-weekly 2.4-mg subcutaneous injection, and some food cravings were improved in a subgroup analysis.
In another study, STEP 8, weight loss was greater at 68 weeks with semaglutide subcutaneous injection than with a 3-mg daily subcutaneous injection of another GLP-1 agonist, liraglutide (Saxenda, Novo Nordisk), approved earlier for weight loss.
Researchers presented these promising outcomes, with no new safety signals, at ObesityWeek® 2021.
However, there is more to learn about the drug class, researchers agree. Follow-up is still relatively short for a chronic disease and many patients have gastrointestinal side effects with semaglutide, one expert cautions.
The key findings were:
In STEP 5, combined with lifestyle intervention (a reduced-calorie meal plan and advice about physical activity), weekly injection of 2.4 mg semaglutide led to:
- 15.2% weight loss, compared with 2.6% weight loss with placebo at 2 years (P < .0001);
- 77% of patients losing at least 5% of their weight, compared with 34% of patients in the placebo group at 2 years (P < .0001);
- Significantly greater improvement in overall control of cravings, and craving for savory foods, in a subset of patients, versus placebo, but questionnaire scores for positive mood and craving for sweet foods were similar in both groups.
- In STEP 8, mean body weight at 68 weeks was 15.8% lower with 2.4 mg/week subcutaneous semaglutide plus lifestyle changes versus 6.4% lower with 3.0 mg/day subcutaneous liraglutide plus lifestyle changes (P < .001).
Can treat to a target weight-loss range
The undiminished weight loss efficacy in the 2-year data for STEP 5 “portends well,” said W. Timothy Garvey, MD, following his presentation of the results.
“I think this is a new era in obesity care,” said Dr. Garvey, director of the diabetes research center at the University of Alabama at Birmingham. Semaglutide “essentially doubles weight loss efficacy” compared to the other approved pharmacotherapies for obesity.
With this degree of potential weight loss, clinicians “can use weight as a biomarker and treat to a target [weight-loss] range,” he said.
Expounding on this in an interview, Dr. Garvey noted that, as stated in the 2016 American Association of Clinical Endocrinologists (AACE) and American College of Endocrinology (ACE) clinical practice guidelines for medical care of patients with obesity, of which he was lead author, “the objective of care in obesity is to increase health of patients and prevent or treat complications.”
Semaglutide “can treat to a range of weight loss of 10% to 20% in the majority of patients,” which is associated with improvements in cardiovascular and metabolic risk factors.
In STEP 5, of the 51% of patients in the semaglutide group who had prediabetes at enrollment, 80% had normal glycemia at 2 years; however, the trial was not powered nor designed to investigate this.
More data are needed to inform long-term care decisions. The ongoing SELECT cardiovascular outcomes trial of semaglutide, with expected primary study completion on Sept. 28, 2023, should provide more information.
Weight loss plus reduced cravings
In another presentation, Sean Wharton, MD, PharmD, said, “In adults with overweight or obesity, substantial weight loss with semaglutide 2.4 mg was accompanied by short- and long-term improvements in control of eating.”
“Most patients living with obesity who are attempting to decrease calories will have food cravings, based on the biological parameters of weight preservation,” Dr. Wharton, medical director at the Wharton Medical Clinic, in Hamilton, Ont., explained in an email.
The degree of craving varies from patient to patient, likely based on genetics, he added. Research in this field is still emerging.
“I believe that semaglutide 2.4 mg is a game-changer in the field of weight management, and it will change the dialogue for insurance plans and with policymakers regarding coverage for this medication,” said Dr. Wharton.
“The data from the STEP programs are very strong. I am certainly hoping for a change to bias against covering these medications that we have seen in the past,” he said.
Clinically meaningful weight loss
When presenting the STEP 8 findings, Domenica M. Rubino, MD, said: “Participants were significantly more likely to achieve clinically meaningful weight loss thresholds with semaglutide 2.4 mg versus liraglutide 3.0 mg, accompanied by greater improvements in cardiometabolic risk factors.”
For example, patients can have better mobility, which is important for quality of life, Dr. Rubino, director of the Washington Center for Weight Management and Research, Arlington, Virginia, noted.
A smaller percentage of patients respond to liraglutide, she added. Clinicians need to individualize treatment.
When asked, “How do you choose which medical therapy?” Dr. Rubino responded: “We sit and talk.” Finding the medical therapy that fits the patient depends on things such as the patient’s insurance coverage and ability to tolerate side effects such as dehydration, diarrhea, and nausea.
When asked, “How do you switch from liraglutide to semaglutide?” she noted that there are no current guidelines for this. “You have to be careful. Start on the lowest dose of Wegovy. Be cautious, conservative.”
Still early days, caveats remain
“The STEP trials as a group appear to be making the case that obesity may now be considered a medically manageable disease, based on the experience with semaglutide,” Julie R. Ingelfinger, MD, who was not involved with the research, commented in an email.
“STEP 5 and 8 may suggest that weight loss occurs and is sustainable in overweight persons without diabetes with one or more comorbidities or in obese persons without diabetes,” added Dr. Ingelfinger, professor of pediatrics, Harvard Medical School, consultant in pediatric nephrology, Massachusetts General Hospital, Boston, and deputy editor, The New England Journal of Medicine.
However, “even 2 years, in the case of STEP 5, and ~68 weeks in the case of STEP 8, may not be long enough to know whether semaglutide is as promising as these brief summaries (abstracts) suggest,” she cautioned.
“Obesity is a chronic condition, and very long-term therapy and management are required,” Dr. Ingelfinger continued.
“Further, it is hard to generalize when gastrointestinal adverse events are common in a study,” she said. For example, in STEP 8, they were just as common with semaglutide as with the comparator liraglutide, she noted.
“The racial and ethnic representativeness of these studies does not reflect population distributions in the U.S., limiting generalization,” she continued.
“So, there remain caveats in interpreting these data.”
STEP 5 weight loss efficacy and safety at 2 years
Garvey reported that STEP 5 was a phase 3b trial that randomized 304 adults in the United States, Canada, Hungary, Italy, and Spain, who were 18 years and older, with a body mass index (BMI) ≥27 kg/m2 with at least one weight-related comorbidity (hypertension, dyslipidemia, obstructive sleep apnea, or cardiovascular disease) or a BMI ≥30 kg/m2, without type 2 diabetes, to receive semaglutide or placebo plus lifestyle intervention.
Most participants were women (78%) and White (93%). On average, they were 47 years old, weighed 106 kg (223.7 pounds), had a BMI of 38.5 kg/m2, a waist circumference of 115.7 cm (45.6 inches), and an A1c of 5.7%.
A total of 87% of patients in the semaglutide group and 73% of patients in the placebo group completed the trial.
At 104 weeks, participants were more likely to lose ≥10%, ≥15%, and ≥20% of body weight with semaglutide versus placebo (61.8% vs. 13.3%, 52.1% vs. 7.0%, and 36.1% vs. 2.3%, respectively; P < .0001 for all).
Patients in the semaglutide group had greater health improvements in cardiovascular risk factors (waist circumference, systolic and diastolic blood pressure, and C-reactive protein) and metabolic risk factors (A1c, fasting plasma glucose, fasting serum insulin, and triglycerides) than those in the placebo group (P < .05 for all).
Safety and tolerability were consistent with adverse events seen with this drug class, with no new safety signals.
Control of eating questionnaire findings at 2 years in STEP 5
Dr. Wharton and colleagues assessed changes in responses to the Control of Eating questionnaire at baseline and at 20, 52, and 104 weeks in patients from the U.S. and Canada in the STEP 5 trial (88 patients in the semaglutide group and 86 patients in the placebo group).
The questionnaire consisted of 19 questions grouped into four categories: control of food cravings, craving for savory foods (salty and spicy, dairy, or starchy foods), craving for sweet foods (chocolate, sweet foods, or fruit/fruit juice), and positive mood.
At week 104, patients in the semaglutide group had significantly greater improvements in scores for craving for salty and spicy, dairy, and starchy foods, and resisting cravings.
Semaglutide versus liraglutide, 68-week efficacy and safety in STEP 8
STEP 8 randomized 338 U.S. adults without diabetes and a BMI of ≥27 kg/m2 plus one or more weight-related comorbidities or a BMI of ≥30 kg/m2 3:1 to semaglutide 2.4 mg once weekly (n = 126) or matching placebo, or 3:1 liraglutide 3.0 mg once daily (n = 127) or matching placebo, plus lifestyle intervention.
Most participants were women (78%) and were a mean age of 49, had a mean body weight of 104.5 kg, and had a mean BMI of 37.5 kg/m2.
In STEP 8, more participants achieved ≥10%, ≥15%, and ≥20% weight loss with semaglutide than with liraglutide (70.9% vs. 25.6%, 55.6% vs. 12.0%, and 38.5% vs. 6.0%, respectively; P < .001 for all odds ratios).
Semaglutide improved waist circumference, A1c, and C-reactive protein versus liraglutide (unadjusted P < .001 for all).
Gastrointestinal adverse events were reported by 84% and 83% of participants receiving semaglutide and liraglutide, respectively. Most events were mild/moderate and transient, with prevalence declining over time.
Fewer participants stopped treatment due to adverse events with semaglutide than liraglutide (3.2% vs. 12.6%).
Dr. Garvey has reported serving as a site principal investigator for multicentered clinical trials sponsored by his university and funded by Eli Lilly, Novo Nordisk, and Pfizer. Dr. Wharton has reported financial ties to Novo Nordisk, Bausch Health Canada, Eli Lily, and Boehringer Ingelheim Canada. Dr. Rubino has reported ties to Boehringer Ingelheim and AstraZeneca. Dr. Ingelfinger has reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Contact allergens in medical devices: A cause for concern?
Despite the clinical value of medical devices, there is a potential for these products to cause adverse skin reactions in some patients. highlighting the possibility of a high prevalence of contact allergens in these devices.
“We found it important to publish these findings, because up until now no clear figures have been reported regarding this particular clinical problem,” said study author Olivier Aerts, MD, a researcher in the contact allergy unit at the University Hospital Antwerp, Belgium, in an interview with this news organization.
For the study, Dr. Aerts and colleagues conducted a retrospective analysis of medical device users with suspected allergic contact dermatitis. All patients had been patch tested at a tertiary European clinic between 2018 and 2020.
The cohort included patients who experienced suspected contact allergy from medical adhesives (n = 57), gloves (n = 38), topical and surface medical devices (n = 38), glucose sensors and insulin pumps (n = 74), and prostheses (n = 75). Other medical products associated with contact allergy in another 44 patients included surgical glues, face masks, compression stockings, condoms, and suture materials.
Overall, 326 patients had been patch-tested during the 30-month study period. Approximately 25.8% of all patients – including 299 adults and 27 children – were referred for contact allergy associated with medical devices.
Acrylates were the most frequently encountered contact allergens and were found in diabetes devices and medical adhesives. Potential skin sensitizers included colophonium-related substances, D-limonene, isothiazolinone derivatives, salicylates, and sulphites, all of which were identified across most products.
According to the investigators, many of the labels for the medical devices made no mention of the potential skin sensitizers, except in the cases of some topical and surface disinfectants. And many topical products are often marketed as medical devices rather than cosmetics, further complicating labeling issues, according to Dr. Aerts.
“What should be done to help any patient suffering from allergic contact due to medical devices is that these devices should be labeled with all their components, or at the very least with the potential skin sensitizers these may contain,” Dr. Aerts explained. He added that manufacturers should “establish more cooperation with physicians/dermatologists who evaluate such patients,” a cooperation that often exists with cosmetic companies.
Dr. Aerts noted that while it’s important for patch testers and dermatologists to be aware of the prevalence of allergic contact dermatitis in medical device users, companies producing these devices should also be aware of these potential issues. “Additionally, legislators/regulators should perhaps focus some more on the cutaneous side effects these products may provoke,” he said, “as this awareness may hopefully also serve as a stimulant to perform more clinical allergy research in this field.”
Leonard Bielory, MD, an allergist at Robert Wood Johnson University Hospital in Rahway, New Jersey, told this news organization that the findings are “alarming” and should heighten clinicians’ awareness of the possibility of allergic contact dermatitis among medical device users.
Dr. Bielory, who wasn’t involved in the research, noted that the findings from this study may not be entirely generalizable to the U.S., given the study was performed in Europe. “In contrast to other countries, the U.S. is very conscientious about allergic responses to items being used in hospitals,” he added, “or such that the issue here is that many of these things would be an adverse reaction, which you have to report.” He suggested that further research in this field is needed to determine the prevalence of possible skin sensitizers in products specifically developed and marketed in the U.S.
The study had no specific funding. Dr. Aerts and Dr. Bielory have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Despite the clinical value of medical devices, there is a potential for these products to cause adverse skin reactions in some patients. highlighting the possibility of a high prevalence of contact allergens in these devices.
“We found it important to publish these findings, because up until now no clear figures have been reported regarding this particular clinical problem,” said study author Olivier Aerts, MD, a researcher in the contact allergy unit at the University Hospital Antwerp, Belgium, in an interview with this news organization.
For the study, Dr. Aerts and colleagues conducted a retrospective analysis of medical device users with suspected allergic contact dermatitis. All patients had been patch tested at a tertiary European clinic between 2018 and 2020.
The cohort included patients who experienced suspected contact allergy from medical adhesives (n = 57), gloves (n = 38), topical and surface medical devices (n = 38), glucose sensors and insulin pumps (n = 74), and prostheses (n = 75). Other medical products associated with contact allergy in another 44 patients included surgical glues, face masks, compression stockings, condoms, and suture materials.
Overall, 326 patients had been patch-tested during the 30-month study period. Approximately 25.8% of all patients – including 299 adults and 27 children – were referred for contact allergy associated with medical devices.
Acrylates were the most frequently encountered contact allergens and were found in diabetes devices and medical adhesives. Potential skin sensitizers included colophonium-related substances, D-limonene, isothiazolinone derivatives, salicylates, and sulphites, all of which were identified across most products.
According to the investigators, many of the labels for the medical devices made no mention of the potential skin sensitizers, except in the cases of some topical and surface disinfectants. And many topical products are often marketed as medical devices rather than cosmetics, further complicating labeling issues, according to Dr. Aerts.
“What should be done to help any patient suffering from allergic contact due to medical devices is that these devices should be labeled with all their components, or at the very least with the potential skin sensitizers these may contain,” Dr. Aerts explained. He added that manufacturers should “establish more cooperation with physicians/dermatologists who evaluate such patients,” a cooperation that often exists with cosmetic companies.
Dr. Aerts noted that while it’s important for patch testers and dermatologists to be aware of the prevalence of allergic contact dermatitis in medical device users, companies producing these devices should also be aware of these potential issues. “Additionally, legislators/regulators should perhaps focus some more on the cutaneous side effects these products may provoke,” he said, “as this awareness may hopefully also serve as a stimulant to perform more clinical allergy research in this field.”
Leonard Bielory, MD, an allergist at Robert Wood Johnson University Hospital in Rahway, New Jersey, told this news organization that the findings are “alarming” and should heighten clinicians’ awareness of the possibility of allergic contact dermatitis among medical device users.
Dr. Bielory, who wasn’t involved in the research, noted that the findings from this study may not be entirely generalizable to the U.S., given the study was performed in Europe. “In contrast to other countries, the U.S. is very conscientious about allergic responses to items being used in hospitals,” he added, “or such that the issue here is that many of these things would be an adverse reaction, which you have to report.” He suggested that further research in this field is needed to determine the prevalence of possible skin sensitizers in products specifically developed and marketed in the U.S.
The study had no specific funding. Dr. Aerts and Dr. Bielory have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Despite the clinical value of medical devices, there is a potential for these products to cause adverse skin reactions in some patients. highlighting the possibility of a high prevalence of contact allergens in these devices.
“We found it important to publish these findings, because up until now no clear figures have been reported regarding this particular clinical problem,” said study author Olivier Aerts, MD, a researcher in the contact allergy unit at the University Hospital Antwerp, Belgium, in an interview with this news organization.
For the study, Dr. Aerts and colleagues conducted a retrospective analysis of medical device users with suspected allergic contact dermatitis. All patients had been patch tested at a tertiary European clinic between 2018 and 2020.
The cohort included patients who experienced suspected contact allergy from medical adhesives (n = 57), gloves (n = 38), topical and surface medical devices (n = 38), glucose sensors and insulin pumps (n = 74), and prostheses (n = 75). Other medical products associated with contact allergy in another 44 patients included surgical glues, face masks, compression stockings, condoms, and suture materials.
Overall, 326 patients had been patch-tested during the 30-month study period. Approximately 25.8% of all patients – including 299 adults and 27 children – were referred for contact allergy associated with medical devices.
Acrylates were the most frequently encountered contact allergens and were found in diabetes devices and medical adhesives. Potential skin sensitizers included colophonium-related substances, D-limonene, isothiazolinone derivatives, salicylates, and sulphites, all of which were identified across most products.
According to the investigators, many of the labels for the medical devices made no mention of the potential skin sensitizers, except in the cases of some topical and surface disinfectants. And many topical products are often marketed as medical devices rather than cosmetics, further complicating labeling issues, according to Dr. Aerts.
“What should be done to help any patient suffering from allergic contact due to medical devices is that these devices should be labeled with all their components, or at the very least with the potential skin sensitizers these may contain,” Dr. Aerts explained. He added that manufacturers should “establish more cooperation with physicians/dermatologists who evaluate such patients,” a cooperation that often exists with cosmetic companies.
Dr. Aerts noted that while it’s important for patch testers and dermatologists to be aware of the prevalence of allergic contact dermatitis in medical device users, companies producing these devices should also be aware of these potential issues. “Additionally, legislators/regulators should perhaps focus some more on the cutaneous side effects these products may provoke,” he said, “as this awareness may hopefully also serve as a stimulant to perform more clinical allergy research in this field.”
Leonard Bielory, MD, an allergist at Robert Wood Johnson University Hospital in Rahway, New Jersey, told this news organization that the findings are “alarming” and should heighten clinicians’ awareness of the possibility of allergic contact dermatitis among medical device users.
Dr. Bielory, who wasn’t involved in the research, noted that the findings from this study may not be entirely generalizable to the U.S., given the study was performed in Europe. “In contrast to other countries, the U.S. is very conscientious about allergic responses to items being used in hospitals,” he added, “or such that the issue here is that many of these things would be an adverse reaction, which you have to report.” He suggested that further research in this field is needed to determine the prevalence of possible skin sensitizers in products specifically developed and marketed in the U.S.
The study had no specific funding. Dr. Aerts and Dr. Bielory have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
AHA 2021 puts scientific dialogue, health equity center stage
Virtual platforms democratized scientific meetings during the COVID-19 pandemic but, as any meeting-goer will tell you, it’s the questions from the floor and the back-and-forth of an expert panel that often reveal the importance of and/or problems with a presentation. It’s the scrutiny that makes the science resonate, especially in this postfactual era.
The all-virtual American Heart Association Scientific Sessions 2021 is looking to recreate the engagement of an in-person meeting by offering more live interactive events. They range from seven late-breaking science (LBS) sessions to Saturday’s fireside chat on the Pfizer and Moderna COVID-19 vaccines and Monday’s dive into the controversial new AHA/American College of Cardiology Chest Pain guidelines.
To help digest the latest science, attendees will be able to have their questions answered in real-time via Slido, meet with the trialists, and hear live commentary from key opinion leaders after the live events. A networking function will also allow attendees and exhibitors to chat or meet virtually.
“In this day and age, many people pretty quickly can get access to the science but it’s what I call the IC sort of phenomenon – the presentation of the information, the context of the information, putting it into how I’m going to use it in my practice, and then the critical appraisal – that’s what most people want at the Scientific Sessions,” program committee chair Manesh R. Patel, MD, of Duke University School of Medicine, said in an interview. “We’re all craving ways in which we can interact with one another to put things in context.”
Plans for a hybrid in-person meeting in Boston were scuttled in September because of the Delta variant surge, but the theme of the meeting remained: “One World. Together for Science.” Attendees will be able to access more than 500 live and on-demand sessions including 117 oral abstracts, 286 poster sessions, 59 moderated digital posters, and over a dozen sessions focused on strategies to promote health equity.
“Last year there was a Presidential Session and a statement on structural racism, so we wanted to take the next step and say, What are the ways in which people are starting to interact and do things to make a difference?” explained Dr. Patel. “So, this year, you’ll see different versions of that from the Main Event session, which has some case vignettes and a panel discussion, to other health equity sessions that describe not just COVID care, but blood pressure care, maternal-fetal medicine, and congenital kids. Wherever we can, we’ve tried to infuse it throughout the sessions and will continue to.”
Late-breaking science
The LBS sessions kick off at 9:30 a.m. ET Saturday with AVATAR, a randomized trial of aortic valve replacement vs. watchful waiting in severe aortic stenosis proved asymptomatic through exercise testing.
“The findings of that trial, depending on what they are, could certainly impact clinical practice because it’s a very common scenario in which we have elderly patients with aortic valve stenosis that might be severe but they may not be symptomatic,” he said.
It’s followed by a randomized trial from the Cardiothoracic Surgical Trials Network, examining whether tricuspid repair at the time of mitral valve surgery leads to beneficial outcomes. “I think it’s a pretty important study,” Dr. Patel said, “because it’ll again affect how we think about our clinical practice.”
Rounding out the LBS.01 session is RAPID CABG, comparing early vs. delayed coronary bypass graft surgery (CABG) in patients with acute coronary syndromes on ticagrelor, and the pivotal U.S. VEST trial of an external support device already approved in Europe for saphenous vein grafts during CABG.
Saturday’s LBS.02 at 3:00 p.m. ET is devoted to hypertension and looks at how the COVID-19 pandemic affected blood pressure control. There’s also a study of remotely delivered hypertension and lipid management in 10,000 patients across the Partners Healthcare System and a cluster randomized trial of a village doctor–led blood pressure intervention in rural China.
Sunday’s LBS.03 at 8:00 a.m. ET is focused on atrial arrhythmias, starting with the CRAVE trial examining the effect of caffeine consumption on cardiac ectopy burden in 108 patients using an N-of-1 design and 2-day blocks on and off caffeine. “There’s an ability to identify a dose response that you get arrhythmias when you increase the amount of coffee you drink vs. not in an individual, so I think that will be likely discussed a lot and worth paying attention to,” Dr. Patel said.
The session also includes GIRAF, a comparison of cognitive outcomes with dabigatran (Pradaxa) vs. warfarin (Coumadin) in nonvalvular atrial fibrillation (AF); PALACS, a randomized trial examining whether left-sided pericardiotomy prevents AF after cardiac surgery; and AMAZE, which study sponsor AtriCure revealed missed its primary efficacy endpoint of freedom from AF with the LARIAT suture delivery device for left atrial appendage closure plus pulmonary vein isolation.
LBS.04 at 3:30 p.m. ET Sunday takes on digital health, with results from the nonrandomized Fitbit Heart Study on AF notifications from 450,000 participants wearing a single-lead ECG patch. “A lot of technologies claim that they can detect things, and we should ask that people go through the rigorous evaluation to see if they in fact do. So, in that respect, I think it›s an important step,” observed Dr. Patel.
Also on tap is I-STOP-AFib, another N-of-1 study using mobile apps and the AliveCor device to identify individual AF triggers; and REVeAL-HF, a 4,000-patient study examining whether electronic alerts that provide clinicians with prognostic information on their heart failure (HF) patients will reduce mortality and 30-day HF hospitalizations.
LBS.05 at 5:00 p.m. ET provides new information from EMPEROR-Preserved in HF with preserved ejection fraction and main results from EMPULSE, also using the sodium-glucose cotransporter 2 (SGLT2) inhibitor empagliflozin (Jardiance) in 530 patients hospitalized for acute HF.
The session also features CHIEF-HF, a randomized trial leveraging mobile technologies to test whether 12 weeks of another SGLT2 inhibitor, canagliflozin (Invokana), is superior to placebo for improving HF symptoms; and DREAM-HF, a comparison of transendocardial delivery of allogeneic mesenchymal precursor cells vs. a sham comparator in chronic HF as a result of left ventricular systolic dysfunction.
Monday’s LBS.06 at 8:00 a.m. ET details the safety and cholesterol-lowering efficacy of MK-0616, an investigational oral PCSK9 inhibitor. “It’s just a phase 2 [trial], but there’s interest in an oral PCSK9 inhibitor, given that the current ones are subcutaneous,” Dr. Patel said.
Results will also be presented from PREPARE-IT 2, which tested icosapent ethyl vs. placebo in outpatients with COVID-19. In the recently reported PREPARE-IT 1, a loading dose of icosapent ethyl failed to reduce the risk of hospitalization with SARS-CoV-2 infection among at-risk individuals.
LBS.07 at 11:00 a.m. Monday completes the late-breakers with new results from ASCEND, this time examining the effect of aspirin on dementia and cognitive impairment in patients with diabetes.
Next up is a look at the effectiveness of P2Y12 inhibitors in hospitalized patients with COVID-19 in the adaptive ACTIV-4a trial, followed by results of the pivotal phase 3 REVERSE-IT trial of bentracimab, a recombinant human monoclonal antibody antigen fragment designed to reverse the antiplatelet activity of ticagrelor in the event of major bleeding or when urgent surgery is needed.
Closing out the session is AXIOMATIC-TKR, a double-blind comparison of the safety and efficacy of the investigational oral factor XI anticoagulant JNJ-70033093 vs. subcutaneous enoxaparin (Lovenox) in elective total knee replacement.
For those searching for more AHA-related science online, the Resuscitation Science Symposium (ReSS) will run from this Friday through Sunday and the Quality of Care and Outcomes Research (QCOR) Scientific Sessions will take the stage next Monday, Nov. 15.
A version of this article first appeared on Medscape.com.
Virtual platforms democratized scientific meetings during the COVID-19 pandemic but, as any meeting-goer will tell you, it’s the questions from the floor and the back-and-forth of an expert panel that often reveal the importance of and/or problems with a presentation. It’s the scrutiny that makes the science resonate, especially in this postfactual era.
The all-virtual American Heart Association Scientific Sessions 2021 is looking to recreate the engagement of an in-person meeting by offering more live interactive events. They range from seven late-breaking science (LBS) sessions to Saturday’s fireside chat on the Pfizer and Moderna COVID-19 vaccines and Monday’s dive into the controversial new AHA/American College of Cardiology Chest Pain guidelines.
To help digest the latest science, attendees will be able to have their questions answered in real-time via Slido, meet with the trialists, and hear live commentary from key opinion leaders after the live events. A networking function will also allow attendees and exhibitors to chat or meet virtually.
“In this day and age, many people pretty quickly can get access to the science but it’s what I call the IC sort of phenomenon – the presentation of the information, the context of the information, putting it into how I’m going to use it in my practice, and then the critical appraisal – that’s what most people want at the Scientific Sessions,” program committee chair Manesh R. Patel, MD, of Duke University School of Medicine, said in an interview. “We’re all craving ways in which we can interact with one another to put things in context.”
Plans for a hybrid in-person meeting in Boston were scuttled in September because of the Delta variant surge, but the theme of the meeting remained: “One World. Together for Science.” Attendees will be able to access more than 500 live and on-demand sessions including 117 oral abstracts, 286 poster sessions, 59 moderated digital posters, and over a dozen sessions focused on strategies to promote health equity.
“Last year there was a Presidential Session and a statement on structural racism, so we wanted to take the next step and say, What are the ways in which people are starting to interact and do things to make a difference?” explained Dr. Patel. “So, this year, you’ll see different versions of that from the Main Event session, which has some case vignettes and a panel discussion, to other health equity sessions that describe not just COVID care, but blood pressure care, maternal-fetal medicine, and congenital kids. Wherever we can, we’ve tried to infuse it throughout the sessions and will continue to.”
Late-breaking science
The LBS sessions kick off at 9:30 a.m. ET Saturday with AVATAR, a randomized trial of aortic valve replacement vs. watchful waiting in severe aortic stenosis proved asymptomatic through exercise testing.
“The findings of that trial, depending on what they are, could certainly impact clinical practice because it’s a very common scenario in which we have elderly patients with aortic valve stenosis that might be severe but they may not be symptomatic,” he said.
It’s followed by a randomized trial from the Cardiothoracic Surgical Trials Network, examining whether tricuspid repair at the time of mitral valve surgery leads to beneficial outcomes. “I think it’s a pretty important study,” Dr. Patel said, “because it’ll again affect how we think about our clinical practice.”
Rounding out the LBS.01 session is RAPID CABG, comparing early vs. delayed coronary bypass graft surgery (CABG) in patients with acute coronary syndromes on ticagrelor, and the pivotal U.S. VEST trial of an external support device already approved in Europe for saphenous vein grafts during CABG.
Saturday’s LBS.02 at 3:00 p.m. ET is devoted to hypertension and looks at how the COVID-19 pandemic affected blood pressure control. There’s also a study of remotely delivered hypertension and lipid management in 10,000 patients across the Partners Healthcare System and a cluster randomized trial of a village doctor–led blood pressure intervention in rural China.
Sunday’s LBS.03 at 8:00 a.m. ET is focused on atrial arrhythmias, starting with the CRAVE trial examining the effect of caffeine consumption on cardiac ectopy burden in 108 patients using an N-of-1 design and 2-day blocks on and off caffeine. “There’s an ability to identify a dose response that you get arrhythmias when you increase the amount of coffee you drink vs. not in an individual, so I think that will be likely discussed a lot and worth paying attention to,” Dr. Patel said.
The session also includes GIRAF, a comparison of cognitive outcomes with dabigatran (Pradaxa) vs. warfarin (Coumadin) in nonvalvular atrial fibrillation (AF); PALACS, a randomized trial examining whether left-sided pericardiotomy prevents AF after cardiac surgery; and AMAZE, which study sponsor AtriCure revealed missed its primary efficacy endpoint of freedom from AF with the LARIAT suture delivery device for left atrial appendage closure plus pulmonary vein isolation.
LBS.04 at 3:30 p.m. ET Sunday takes on digital health, with results from the nonrandomized Fitbit Heart Study on AF notifications from 450,000 participants wearing a single-lead ECG patch. “A lot of technologies claim that they can detect things, and we should ask that people go through the rigorous evaluation to see if they in fact do. So, in that respect, I think it›s an important step,” observed Dr. Patel.
Also on tap is I-STOP-AFib, another N-of-1 study using mobile apps and the AliveCor device to identify individual AF triggers; and REVeAL-HF, a 4,000-patient study examining whether electronic alerts that provide clinicians with prognostic information on their heart failure (HF) patients will reduce mortality and 30-day HF hospitalizations.
LBS.05 at 5:00 p.m. ET provides new information from EMPEROR-Preserved in HF with preserved ejection fraction and main results from EMPULSE, also using the sodium-glucose cotransporter 2 (SGLT2) inhibitor empagliflozin (Jardiance) in 530 patients hospitalized for acute HF.
The session also features CHIEF-HF, a randomized trial leveraging mobile technologies to test whether 12 weeks of another SGLT2 inhibitor, canagliflozin (Invokana), is superior to placebo for improving HF symptoms; and DREAM-HF, a comparison of transendocardial delivery of allogeneic mesenchymal precursor cells vs. a sham comparator in chronic HF as a result of left ventricular systolic dysfunction.
Monday’s LBS.06 at 8:00 a.m. ET details the safety and cholesterol-lowering efficacy of MK-0616, an investigational oral PCSK9 inhibitor. “It’s just a phase 2 [trial], but there’s interest in an oral PCSK9 inhibitor, given that the current ones are subcutaneous,” Dr. Patel said.
Results will also be presented from PREPARE-IT 2, which tested icosapent ethyl vs. placebo in outpatients with COVID-19. In the recently reported PREPARE-IT 1, a loading dose of icosapent ethyl failed to reduce the risk of hospitalization with SARS-CoV-2 infection among at-risk individuals.
LBS.07 at 11:00 a.m. Monday completes the late-breakers with new results from ASCEND, this time examining the effect of aspirin on dementia and cognitive impairment in patients with diabetes.
Next up is a look at the effectiveness of P2Y12 inhibitors in hospitalized patients with COVID-19 in the adaptive ACTIV-4a trial, followed by results of the pivotal phase 3 REVERSE-IT trial of bentracimab, a recombinant human monoclonal antibody antigen fragment designed to reverse the antiplatelet activity of ticagrelor in the event of major bleeding or when urgent surgery is needed.
Closing out the session is AXIOMATIC-TKR, a double-blind comparison of the safety and efficacy of the investigational oral factor XI anticoagulant JNJ-70033093 vs. subcutaneous enoxaparin (Lovenox) in elective total knee replacement.
For those searching for more AHA-related science online, the Resuscitation Science Symposium (ReSS) will run from this Friday through Sunday and the Quality of Care and Outcomes Research (QCOR) Scientific Sessions will take the stage next Monday, Nov. 15.
A version of this article first appeared on Medscape.com.
Virtual platforms democratized scientific meetings during the COVID-19 pandemic but, as any meeting-goer will tell you, it’s the questions from the floor and the back-and-forth of an expert panel that often reveal the importance of and/or problems with a presentation. It’s the scrutiny that makes the science resonate, especially in this postfactual era.
The all-virtual American Heart Association Scientific Sessions 2021 is looking to recreate the engagement of an in-person meeting by offering more live interactive events. They range from seven late-breaking science (LBS) sessions to Saturday’s fireside chat on the Pfizer and Moderna COVID-19 vaccines and Monday’s dive into the controversial new AHA/American College of Cardiology Chest Pain guidelines.
To help digest the latest science, attendees will be able to have their questions answered in real-time via Slido, meet with the trialists, and hear live commentary from key opinion leaders after the live events. A networking function will also allow attendees and exhibitors to chat or meet virtually.
“In this day and age, many people pretty quickly can get access to the science but it’s what I call the IC sort of phenomenon – the presentation of the information, the context of the information, putting it into how I’m going to use it in my practice, and then the critical appraisal – that’s what most people want at the Scientific Sessions,” program committee chair Manesh R. Patel, MD, of Duke University School of Medicine, said in an interview. “We’re all craving ways in which we can interact with one another to put things in context.”
Plans for a hybrid in-person meeting in Boston were scuttled in September because of the Delta variant surge, but the theme of the meeting remained: “One World. Together for Science.” Attendees will be able to access more than 500 live and on-demand sessions including 117 oral abstracts, 286 poster sessions, 59 moderated digital posters, and over a dozen sessions focused on strategies to promote health equity.
“Last year there was a Presidential Session and a statement on structural racism, so we wanted to take the next step and say, What are the ways in which people are starting to interact and do things to make a difference?” explained Dr. Patel. “So, this year, you’ll see different versions of that from the Main Event session, which has some case vignettes and a panel discussion, to other health equity sessions that describe not just COVID care, but blood pressure care, maternal-fetal medicine, and congenital kids. Wherever we can, we’ve tried to infuse it throughout the sessions and will continue to.”
Late-breaking science
The LBS sessions kick off at 9:30 a.m. ET Saturday with AVATAR, a randomized trial of aortic valve replacement vs. watchful waiting in severe aortic stenosis proved asymptomatic through exercise testing.
“The findings of that trial, depending on what they are, could certainly impact clinical practice because it’s a very common scenario in which we have elderly patients with aortic valve stenosis that might be severe but they may not be symptomatic,” he said.
It’s followed by a randomized trial from the Cardiothoracic Surgical Trials Network, examining whether tricuspid repair at the time of mitral valve surgery leads to beneficial outcomes. “I think it’s a pretty important study,” Dr. Patel said, “because it’ll again affect how we think about our clinical practice.”
Rounding out the LBS.01 session is RAPID CABG, comparing early vs. delayed coronary bypass graft surgery (CABG) in patients with acute coronary syndromes on ticagrelor, and the pivotal U.S. VEST trial of an external support device already approved in Europe for saphenous vein grafts during CABG.
Saturday’s LBS.02 at 3:00 p.m. ET is devoted to hypertension and looks at how the COVID-19 pandemic affected blood pressure control. There’s also a study of remotely delivered hypertension and lipid management in 10,000 patients across the Partners Healthcare System and a cluster randomized trial of a village doctor–led blood pressure intervention in rural China.
Sunday’s LBS.03 at 8:00 a.m. ET is focused on atrial arrhythmias, starting with the CRAVE trial examining the effect of caffeine consumption on cardiac ectopy burden in 108 patients using an N-of-1 design and 2-day blocks on and off caffeine. “There’s an ability to identify a dose response that you get arrhythmias when you increase the amount of coffee you drink vs. not in an individual, so I think that will be likely discussed a lot and worth paying attention to,” Dr. Patel said.
The session also includes GIRAF, a comparison of cognitive outcomes with dabigatran (Pradaxa) vs. warfarin (Coumadin) in nonvalvular atrial fibrillation (AF); PALACS, a randomized trial examining whether left-sided pericardiotomy prevents AF after cardiac surgery; and AMAZE, which study sponsor AtriCure revealed missed its primary efficacy endpoint of freedom from AF with the LARIAT suture delivery device for left atrial appendage closure plus pulmonary vein isolation.
LBS.04 at 3:30 p.m. ET Sunday takes on digital health, with results from the nonrandomized Fitbit Heart Study on AF notifications from 450,000 participants wearing a single-lead ECG patch. “A lot of technologies claim that they can detect things, and we should ask that people go through the rigorous evaluation to see if they in fact do. So, in that respect, I think it›s an important step,” observed Dr. Patel.
Also on tap is I-STOP-AFib, another N-of-1 study using mobile apps and the AliveCor device to identify individual AF triggers; and REVeAL-HF, a 4,000-patient study examining whether electronic alerts that provide clinicians with prognostic information on their heart failure (HF) patients will reduce mortality and 30-day HF hospitalizations.
LBS.05 at 5:00 p.m. ET provides new information from EMPEROR-Preserved in HF with preserved ejection fraction and main results from EMPULSE, also using the sodium-glucose cotransporter 2 (SGLT2) inhibitor empagliflozin (Jardiance) in 530 patients hospitalized for acute HF.
The session also features CHIEF-HF, a randomized trial leveraging mobile technologies to test whether 12 weeks of another SGLT2 inhibitor, canagliflozin (Invokana), is superior to placebo for improving HF symptoms; and DREAM-HF, a comparison of transendocardial delivery of allogeneic mesenchymal precursor cells vs. a sham comparator in chronic HF as a result of left ventricular systolic dysfunction.
Monday’s LBS.06 at 8:00 a.m. ET details the safety and cholesterol-lowering efficacy of MK-0616, an investigational oral PCSK9 inhibitor. “It’s just a phase 2 [trial], but there’s interest in an oral PCSK9 inhibitor, given that the current ones are subcutaneous,” Dr. Patel said.
Results will also be presented from PREPARE-IT 2, which tested icosapent ethyl vs. placebo in outpatients with COVID-19. In the recently reported PREPARE-IT 1, a loading dose of icosapent ethyl failed to reduce the risk of hospitalization with SARS-CoV-2 infection among at-risk individuals.
LBS.07 at 11:00 a.m. Monday completes the late-breakers with new results from ASCEND, this time examining the effect of aspirin on dementia and cognitive impairment in patients with diabetes.
Next up is a look at the effectiveness of P2Y12 inhibitors in hospitalized patients with COVID-19 in the adaptive ACTIV-4a trial, followed by results of the pivotal phase 3 REVERSE-IT trial of bentracimab, a recombinant human monoclonal antibody antigen fragment designed to reverse the antiplatelet activity of ticagrelor in the event of major bleeding or when urgent surgery is needed.
Closing out the session is AXIOMATIC-TKR, a double-blind comparison of the safety and efficacy of the investigational oral factor XI anticoagulant JNJ-70033093 vs. subcutaneous enoxaparin (Lovenox) in elective total knee replacement.
For those searching for more AHA-related science online, the Resuscitation Science Symposium (ReSS) will run from this Friday through Sunday and the Quality of Care and Outcomes Research (QCOR) Scientific Sessions will take the stage next Monday, Nov. 15.
A version of this article first appeared on Medscape.com.
FROM AHA 2021
Evaluation of the Effectiveness and Safety of Alirocumab Use in Statin-Intolerant Veterans
In 2016, 17.6 million deaths occurred globally due to cardiovascular disease (CVD) with coronary artery disease (CAD) and ischemic stroke as top contributors.1 Elevated low-density lipoprotein cholesterol (LDL-C) has been linked to greater risk of atherosclerotic cardiovascular disease (ASCVD); therefore, LDL-C reduction is imperative to decrease risk of cardiovascular (CV) morbidity and mortality.2 Since 1987, statin therapy has been the mainstay of treatment for hypercholesterolemia, and current practice guidelines recommend statins as first-line therapy given demonstrated reductions in LDL-C and CV mortality reduction in robust clinical trials.2-4 Although generally safe and well tolerated, muscle-related adverse events (AEs) limit optimal use of statins in up to 20% of individuals who have an indication for statin therapy.5 As a consequence, these patients receive suboptimal statin doses or no statin therapy and are at a higher risk for ASCVD.5
Proprotein convertase subtilisin/kexin type 9 (PCSK9) inhibitors have been shown to significantly lower LDL-C when used as monotherapy or in combination with statins and/or other lipid-lowering therapies.5 These agents are currently approved by the US Food and Drug Administration as an adjunct to diet with or without other lipid-lowering therapies for the management of primary hypercholesterolemia (including heterozygous familial hypercholesterolemia), homozygous familial hypercholesterolemia (evolocumab only), and for use in patients with established CVD unable to achieve their lipid-lowering goals with maximally tolerated statin doses and ezetimibe.4 With the ability to reduce LDL-C by up to 65%, PCSK9 inhibitors offer an alternative option for LDL-C and potentially CV risk reduction in statin-intolerant patients.5
Alirocumab, the formulary preferred PCSK9 inhibitor at the Michael E. DeBakey Veterans Affairs Medical Center (MEDVAMC) in Houston, Texas, has been increasingly used in high-risk statin-intolerant veterans. The primary objective of this case series was to assess LDL-C reduction associated with alirocumab use in statin-intolerant veterans at the MEDVAMC. The secondary objective was to assess the incidence of CV events. This study was approved by the MEDVAMC Quality Assurance and Regulatory Affairs committee.
Methods
In this single-center case series, a retrospective chart review was conducted to identify statin-intolerant veterans who were initiated on treatment with alirocumab for LDL-C and/or CV risk reduction between June 2017 and May 2019. Adult veterans with a diagnosis of primary hypercholesterolemia (including heterozygous familial hypercholesterolemia) and/or CAD with documented statin intolerance were included in the study. Statin intolerance was defined in accordance with the National Lipid Association (NLA) definition as aninability to tolerate ≥ 2 statins with a trial of at least 1 statin at its lowest daily dose.5 Veterans who previously received treatment with evolocumab, those prescribed concurrent statin therapies, and those missing follow-up lipid panels at 24 weeks were excluded from the study. To assess LDL-C reduction, LDL-C at baseline was compared with LDL-C at 4 and 24 weeks. Incident CV events before and after alirocumab initiation were documented. The US Department of Veteran Affairs (VA) Computerized Patient Record System was used to collect patient data.
Data Collection, Measures, and Analysis
Electronic health records of all eligible patients who received alirocumab were reviewed, and basic demographics (patient age, sex, and race/ethnicity) as well as medical characteristics at baseline were collected. To confirm statin intolerance, each veteran’s history of statin use and use of additional lipid-lowering agents was documented. CV history was measured with an index of categorical measures for hypertension, confirmed CAD, hyperlipidemia, heart failure, arrhythmias, peripheral artery disease, stroke, diabetes mellitus, and hypothyroidism. Additionally, concomitant medications, such as aspirin, P2Y12 inhibitors, β-blockers, angiotensin-converting enzyme inhibitors, and angiotensin II receptor blockers that patients were taking also were collected. Each veteran’s lipid panel at baseline, and at 4 and 24 weeks posttreatment initiation, also was extracted. Continuous variables were summarized with means (SD), and categorical variables were summarized with frequencies and proportions. The paired Wilcoxon signed rank test was used to compare LDL-C at 4 and 24 weeks after alirocumab initiation with patients’ baseline LDL-C.
Results
Between June 2017 and May 2019, 122 veterans were initiated on alirocumab. Of these veterans, 98 were excluded: 35 concurrently received statin therapy, 33 missed follow-up lipid panels, 21 had previously received evolocumab, 6 failed to meet the NLA definition for statin intolerance, 2 did not fill active alirocumab prescriptions, and 1 had an incalculable LDL-C with a baseline triglyceride level of 3079 mg/dL. This resulted in 24 veterans included in the analysis.
Most participants were male (87.5%) and White veterans (79.2%) with a mean (SD) age of 66.0 (8.4) years and mean (SD) baseline LDL-C of 161.9 (74.3) mg/dL. At baseline, 21 veterans had a history of primary hyperlipidemia, 19 had a history of CAD, and 2 had a history of heterozygous familial hypercholesterolemia. Of the 24 patients included, the most trialed statins before alirocumab initiation were atorvastatin (95.8%), simvastatin (79.2%), rosuvastatin (79.2%), and pravastatin (62.5%) (Table).
LDL-C Reduction
Veterans were initially treated with alirocumab 75 mg administered subcutaneously every 2 weeks; however, 11 veterans required a dose increase to 150 mg every 2 weeks. At treatment week 4, the median LDL-C reduction was 78.5 mg/dL (IQR, 28.0-107.3; P < .01), and at treatment week 24, the median LDL-C reduction was 55.6 mg/dL (IQR, 18.6-85.3; P < .01). This equated to median LDL-C reductions from baseline of 48.5% at week 4 and 34.3% at week 24. A total of 3 veterans experienced LDL-C increases following initiation of alirocumab. At week 4, 9 veterans were noted to have an LDL-C reduction > 50%, 7 veterans had an LDL-C reduction between 30% and 50%, and 5 veterans had an LDL-C reduction of < 30%. At week 24, 6 had an LDL-C reduction > 50%, 9 veterans had an LDL-C reduction between 30% and 50%, and 6 had a LDL-C reduction < 30%.
Cardiovascular Events
Before alirocumab initiation, 22 CV events and interventions were reported in 16 veterans: 12 percutaneous coronary interventions, 5 coronary artery bypass surgeries (CABG), 4 myocardial infarctions, and 1 transient ischemic attack. One month following alirocumab initiation, 1 veteran underwent a CABG after a non-ST-elevation myocardial infarction (NSTEMI).
Safety and Tolerability
Alirocumab was discontinued in 5 veterans due to 4 cases of intolerance (reported memory loss, lethargy, myalgias, and body aches with dyspnea) and 1 case of persistent LDL-C of < 40 mg/dL. Alirocumab was discontinued after 1 year in 2 patients (persistent LDL-C < 40 mg/dL and reported memory loss) and after 6 months in the veteran who reported lethargy. Alirocumab was discontinued after 4 months in the veteran with myalgias and within 2 months in the veteran with body aches and dyspnea. No other AEs were reported.
Discussion
The Efficacy and Safety of Alirocumab vs Ezetimibe in Statin-Intolerant Veterans With a Statin Rechallenge Arm trial is the first clinical trial to examine the efficacy and safety of alirocumab use in statin-intolerant patients. In the trial, 314 patients were randomized to receive alirocumab, ezetimibe, or an atorvastatin rechallenge.6 At 24 weeks, alirocumab reduced mean (SE) LDL-C by 45.0% (2.2%) vs 14.6% (2.2%) with ezetimibe (mean difference 30.4% [3.1%], P < .01).6 Fewer skeletal-muscle-related events also were noted with alirocumab vs atorvastatin (hazard ratio, 0.61; 95% CI, 0.38-0.99; P = .04).6
In this case series, an LDL-C reduction of > 50% was observed in 9 veterans (42.9%) following 4 weeks of treatment; however, LDL-C reduction of > 50% compared with baseline was sustained in only 6 veterans (28.6%) at week 24. Additionally, LDL-C increases from baseline were observed in 3 veterans; the reasoning for the observed increase was unclear, but this may have been due to nonadherence and dietary factors.4 Although a majority of patients saw a significant and clinically meaningful reduction in LDL-C, the group of patients with an increase in the same may have benefitted from targeted intervention to improve medication and dietary adherence. PCSK9 inhibitor resistance also may have contributed to an increase in LDL-C during treatment.7
Of the 24 patients included, 4 reported AEs resulted in therapy discontinuation. Memory impairment, a rare AE of alirocumab, was reported 1 year following alirocumab initiation. Additionally, lethargy was reported after 6 months of treatment. Myalgia also was reported in a veteran 4 months following treatment, and 1 veteran experienced body aches and dyspnea < 2 months following treatment. The most common AEs associated with alirocumab, as noted in previous safety and efficacy clinical trials, included: nasopharyngitis, injection site reaction, influenza, urinary tract infection, and myalgias.8 Many of these more common AEs may be subclinical and underreported. This small case series, however, detected 4 events severe enough to lead to therapy discontinuation. Although this sample is not representative of all statin-intolerant patients who receive treatment with alirocumab, our findings suggest the need for patient education on potential AEs before therapy initiation and clinician monitoring at follow-up visits.
The ODYSSEY OUTCOMES trial established a CV benefit associated with alirocumab; however, patients included had a recent acute coronary syndrome event and were receiving a high-intensity statin.9 This case series is unique in that before alirocumab initiation, 22 CV events/interventions were reported in the sample of 24 patients. After therapy initiation, 1 patient underwent a CABG after an NSTEMI in the month following initiation. This suggests that cardiac complications are possible after PCSK-9 initiation; however, little information can be gained from 1 patient. Nevertheless, early therapy failure should be investigated in the context of real-world use in statin-intolerant patients. This is a complex task, however, given the difficulties of achieving a balanced study design. Statin intolerance is a clear source of selection bias into treatment with alirocumab as patients in this population have already initiated and failed statin therapy. The prevalence of prior CV events and the time-dependent association between prior and future CV events stand as another complex confounder. Although there is a clear and pressing need to understand the risks and benefits of PCSK9 therapy in statin-intolerant patients, future research in this area will need to cautiously address these important sources of bias.
Overall, the results of this case series support LDL-C reduction associated with alirocumab in the absence of statin therapy. Despite favorable results, use of alirocumab may be limited by cost and its subcutaneous route of administration. Bempedoic acid, an oral, once-daily lipid-lowering agent poses an alternative to PCSK9 inhibitors, but further data regarding CV outcomes with this agent is needed.10,11 Robust randomized controlled trials also are needed to evaluate CV outcomes for alirocumab use in statin-intolerant veterans.
Limitations
Only 24 veterans were included in the study, reflecting 20% of the charts reviewed (80% exclusion rate), and in this small sample, only 1 CV event was observed. Both of these serve as threats to external validity. As the study information was extracted from chart review, the results may be limited by coding or historical bias. Medical information from outside institutions may be missing from medical records. Additionally, results may be skewed by possible documentation errors. Furthermore, the period between previous CV events and alirocumab initiation is unclear as event dates were often not recorded if treatment was received at an outside institution.
Due to the short follow-up period, the case series is limited in its assessment of CV outcomes and safety outcomes. Larger studies over an extended period are needed to assess CV outcomes and safety of alirocumab use in statin-intolerant patients. Also, medication adherence was not assessed. Given the impact of medication adherence on LDL-C reduction, it is unclear what role medication adherence played in the LDL-C reduction observed in this study.4
Conclusions
Alirocumab use in 24 statin-intolerant veterans resulted in a significant reduction in LDL-C at 4 and 24 weeks after initiation. In addition, 1 CV event/intervention was observed following alirocumab initiation, although this should be interpreted with caution due to the retrospective nature of this case series, small sample size, and short follow-up period. Large, long-term studies would better evaluate the CV benefit associated with alirocumab therapy in a veteran population.
1. Benjamin EJ, Munter P, Alonso A, et al; American Heart Association Council on Epidemiology and Prevention Statistics Committee and Stroke Statistics Subcommittee. Heart disease and stroke statistics—2019 update: a report from the American Heart Association. Circulation. 2019;139(10):e56-e528. doi:10.1161/CIR.0000000000000659
2. Stone NJ, Robinson JG, Lichtenstein AH, et al; American College of Cardiology/American Heart Association Task Force on Practice Guidelines. 2013 ACC/AHA guideline on the treatment of blood cholesterol to reduce atherosclerotic cardiovascular risk in adults: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. Circulation. 2014 Jun 24;129(25)(suppl 2):S1-S45. doi:10.1016/j.jacc.2013.11.002
3. Hajar R. Statins: past and present. Heart Views. 2011;12(3): 121-127. doi:10.4103/1995-705X.95070
4. Grundy SM, Stone NJ, Bailey AL, et al. 2018 AHA/ACC/AACVPR/AAPA/ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA guideline on the management of blood cholesterol: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. J Am Coll Cardiol 2019;73(4):3168-3209. doi:10.1016/j.jacc.2018.11.002
5. Toth PH, Patti AM, Giglio RV, et al. Management of statin intolerance in 2018: still more questions than answers. Am J Cardiovasc Drugs. 2018;18(3):157-173. doi:10.1007/s40256-017-0259-7
6. Moriarty PM, Thompson PD, Cannon CP, et al; ODYSSEY ALTERNATIVE Investigators. Efficacy and safety of alirocumab vs ezetimibe in statin-intolerant patients, with a statin rechallenge arm: The ODYSSEY ALTERNATIVE randomized trial. J Clin Lipidol. 2015;9(6):758-769. doi:10.1016/j.jacl.2015.08.006
7. Shapiro MD, Miles J, Tavori H, Fazio S. Diagnosing resistance to a proprotein convertase subtilisin/kexin type 9 inhibitor. Ann Intern Med. 2018;168(5):376-379. doi:10.7326/M17-2485
8. Raedler LA. Praluent (alirocumab): first PCSK9 inhibitor approved by the FDA for hypercholesterolemia. Am Health Drug Benefits. 2016;9:123-126.
9. Schwartz GC, Steg PC, Szarek M, et al; ODYSSEY OUTCOMES Committees and Investigators. Alirocumab and cardiovascular outcomes after acute coronary syndrome. N Engl J Med. 2018;379(22):2097-2107. doi:10.1056/NEJMoa1801174
10. Nexletol. Package insert. Esperion Therapeutics Inc; 2020.
11. Laufs U, Banach M, Mancini GBJ, et al. Efficacy and safety of bempedoic acid in patients with hypercholesterolemia and statin intolerance. J Am Heart Assoc. 2019;8(7):e011662. doi:10.1161/JAHA.118.011662
In 2016, 17.6 million deaths occurred globally due to cardiovascular disease (CVD) with coronary artery disease (CAD) and ischemic stroke as top contributors.1 Elevated low-density lipoprotein cholesterol (LDL-C) has been linked to greater risk of atherosclerotic cardiovascular disease (ASCVD); therefore, LDL-C reduction is imperative to decrease risk of cardiovascular (CV) morbidity and mortality.2 Since 1987, statin therapy has been the mainstay of treatment for hypercholesterolemia, and current practice guidelines recommend statins as first-line therapy given demonstrated reductions in LDL-C and CV mortality reduction in robust clinical trials.2-4 Although generally safe and well tolerated, muscle-related adverse events (AEs) limit optimal use of statins in up to 20% of individuals who have an indication for statin therapy.5 As a consequence, these patients receive suboptimal statin doses or no statin therapy and are at a higher risk for ASCVD.5
Proprotein convertase subtilisin/kexin type 9 (PCSK9) inhibitors have been shown to significantly lower LDL-C when used as monotherapy or in combination with statins and/or other lipid-lowering therapies.5 These agents are currently approved by the US Food and Drug Administration as an adjunct to diet with or without other lipid-lowering therapies for the management of primary hypercholesterolemia (including heterozygous familial hypercholesterolemia), homozygous familial hypercholesterolemia (evolocumab only), and for use in patients with established CVD unable to achieve their lipid-lowering goals with maximally tolerated statin doses and ezetimibe.4 With the ability to reduce LDL-C by up to 65%, PCSK9 inhibitors offer an alternative option for LDL-C and potentially CV risk reduction in statin-intolerant patients.5
Alirocumab, the formulary preferred PCSK9 inhibitor at the Michael E. DeBakey Veterans Affairs Medical Center (MEDVAMC) in Houston, Texas, has been increasingly used in high-risk statin-intolerant veterans. The primary objective of this case series was to assess LDL-C reduction associated with alirocumab use in statin-intolerant veterans at the MEDVAMC. The secondary objective was to assess the incidence of CV events. This study was approved by the MEDVAMC Quality Assurance and Regulatory Affairs committee.
Methods
In this single-center case series, a retrospective chart review was conducted to identify statin-intolerant veterans who were initiated on treatment with alirocumab for LDL-C and/or CV risk reduction between June 2017 and May 2019. Adult veterans with a diagnosis of primary hypercholesterolemia (including heterozygous familial hypercholesterolemia) and/or CAD with documented statin intolerance were included in the study. Statin intolerance was defined in accordance with the National Lipid Association (NLA) definition as aninability to tolerate ≥ 2 statins with a trial of at least 1 statin at its lowest daily dose.5 Veterans who previously received treatment with evolocumab, those prescribed concurrent statin therapies, and those missing follow-up lipid panels at 24 weeks were excluded from the study. To assess LDL-C reduction, LDL-C at baseline was compared with LDL-C at 4 and 24 weeks. Incident CV events before and after alirocumab initiation were documented. The US Department of Veteran Affairs (VA) Computerized Patient Record System was used to collect patient data.
Data Collection, Measures, and Analysis
Electronic health records of all eligible patients who received alirocumab were reviewed, and basic demographics (patient age, sex, and race/ethnicity) as well as medical characteristics at baseline were collected. To confirm statin intolerance, each veteran’s history of statin use and use of additional lipid-lowering agents was documented. CV history was measured with an index of categorical measures for hypertension, confirmed CAD, hyperlipidemia, heart failure, arrhythmias, peripheral artery disease, stroke, diabetes mellitus, and hypothyroidism. Additionally, concomitant medications, such as aspirin, P2Y12 inhibitors, β-blockers, angiotensin-converting enzyme inhibitors, and angiotensin II receptor blockers that patients were taking also were collected. Each veteran’s lipid panel at baseline, and at 4 and 24 weeks posttreatment initiation, also was extracted. Continuous variables were summarized with means (SD), and categorical variables were summarized with frequencies and proportions. The paired Wilcoxon signed rank test was used to compare LDL-C at 4 and 24 weeks after alirocumab initiation with patients’ baseline LDL-C.
Results
Between June 2017 and May 2019, 122 veterans were initiated on alirocumab. Of these veterans, 98 were excluded: 35 concurrently received statin therapy, 33 missed follow-up lipid panels, 21 had previously received evolocumab, 6 failed to meet the NLA definition for statin intolerance, 2 did not fill active alirocumab prescriptions, and 1 had an incalculable LDL-C with a baseline triglyceride level of 3079 mg/dL. This resulted in 24 veterans included in the analysis.
Most participants were male (87.5%) and White veterans (79.2%) with a mean (SD) age of 66.0 (8.4) years and mean (SD) baseline LDL-C of 161.9 (74.3) mg/dL. At baseline, 21 veterans had a history of primary hyperlipidemia, 19 had a history of CAD, and 2 had a history of heterozygous familial hypercholesterolemia. Of the 24 patients included, the most trialed statins before alirocumab initiation were atorvastatin (95.8%), simvastatin (79.2%), rosuvastatin (79.2%), and pravastatin (62.5%) (Table).
LDL-C Reduction
Veterans were initially treated with alirocumab 75 mg administered subcutaneously every 2 weeks; however, 11 veterans required a dose increase to 150 mg every 2 weeks. At treatment week 4, the median LDL-C reduction was 78.5 mg/dL (IQR, 28.0-107.3; P < .01), and at treatment week 24, the median LDL-C reduction was 55.6 mg/dL (IQR, 18.6-85.3; P < .01). This equated to median LDL-C reductions from baseline of 48.5% at week 4 and 34.3% at week 24. A total of 3 veterans experienced LDL-C increases following initiation of alirocumab. At week 4, 9 veterans were noted to have an LDL-C reduction > 50%, 7 veterans had an LDL-C reduction between 30% and 50%, and 5 veterans had an LDL-C reduction of < 30%. At week 24, 6 had an LDL-C reduction > 50%, 9 veterans had an LDL-C reduction between 30% and 50%, and 6 had a LDL-C reduction < 30%.
Cardiovascular Events
Before alirocumab initiation, 22 CV events and interventions were reported in 16 veterans: 12 percutaneous coronary interventions, 5 coronary artery bypass surgeries (CABG), 4 myocardial infarctions, and 1 transient ischemic attack. One month following alirocumab initiation, 1 veteran underwent a CABG after a non-ST-elevation myocardial infarction (NSTEMI).
Safety and Tolerability
Alirocumab was discontinued in 5 veterans due to 4 cases of intolerance (reported memory loss, lethargy, myalgias, and body aches with dyspnea) and 1 case of persistent LDL-C of < 40 mg/dL. Alirocumab was discontinued after 1 year in 2 patients (persistent LDL-C < 40 mg/dL and reported memory loss) and after 6 months in the veteran who reported lethargy. Alirocumab was discontinued after 4 months in the veteran with myalgias and within 2 months in the veteran with body aches and dyspnea. No other AEs were reported.
Discussion
The Efficacy and Safety of Alirocumab vs Ezetimibe in Statin-Intolerant Veterans With a Statin Rechallenge Arm trial is the first clinical trial to examine the efficacy and safety of alirocumab use in statin-intolerant patients. In the trial, 314 patients were randomized to receive alirocumab, ezetimibe, or an atorvastatin rechallenge.6 At 24 weeks, alirocumab reduced mean (SE) LDL-C by 45.0% (2.2%) vs 14.6% (2.2%) with ezetimibe (mean difference 30.4% [3.1%], P < .01).6 Fewer skeletal-muscle-related events also were noted with alirocumab vs atorvastatin (hazard ratio, 0.61; 95% CI, 0.38-0.99; P = .04).6
In this case series, an LDL-C reduction of > 50% was observed in 9 veterans (42.9%) following 4 weeks of treatment; however, LDL-C reduction of > 50% compared with baseline was sustained in only 6 veterans (28.6%) at week 24. Additionally, LDL-C increases from baseline were observed in 3 veterans; the reasoning for the observed increase was unclear, but this may have been due to nonadherence and dietary factors.4 Although a majority of patients saw a significant and clinically meaningful reduction in LDL-C, the group of patients with an increase in the same may have benefitted from targeted intervention to improve medication and dietary adherence. PCSK9 inhibitor resistance also may have contributed to an increase in LDL-C during treatment.7
Of the 24 patients included, 4 reported AEs resulted in therapy discontinuation. Memory impairment, a rare AE of alirocumab, was reported 1 year following alirocumab initiation. Additionally, lethargy was reported after 6 months of treatment. Myalgia also was reported in a veteran 4 months following treatment, and 1 veteran experienced body aches and dyspnea < 2 months following treatment. The most common AEs associated with alirocumab, as noted in previous safety and efficacy clinical trials, included: nasopharyngitis, injection site reaction, influenza, urinary tract infection, and myalgias.8 Many of these more common AEs may be subclinical and underreported. This small case series, however, detected 4 events severe enough to lead to therapy discontinuation. Although this sample is not representative of all statin-intolerant patients who receive treatment with alirocumab, our findings suggest the need for patient education on potential AEs before therapy initiation and clinician monitoring at follow-up visits.
The ODYSSEY OUTCOMES trial established a CV benefit associated with alirocumab; however, patients included had a recent acute coronary syndrome event and were receiving a high-intensity statin.9 This case series is unique in that before alirocumab initiation, 22 CV events/interventions were reported in the sample of 24 patients. After therapy initiation, 1 patient underwent a CABG after an NSTEMI in the month following initiation. This suggests that cardiac complications are possible after PCSK-9 initiation; however, little information can be gained from 1 patient. Nevertheless, early therapy failure should be investigated in the context of real-world use in statin-intolerant patients. This is a complex task, however, given the difficulties of achieving a balanced study design. Statin intolerance is a clear source of selection bias into treatment with alirocumab as patients in this population have already initiated and failed statin therapy. The prevalence of prior CV events and the time-dependent association between prior and future CV events stand as another complex confounder. Although there is a clear and pressing need to understand the risks and benefits of PCSK9 therapy in statin-intolerant patients, future research in this area will need to cautiously address these important sources of bias.
Overall, the results of this case series support LDL-C reduction associated with alirocumab in the absence of statin therapy. Despite favorable results, use of alirocumab may be limited by cost and its subcutaneous route of administration. Bempedoic acid, an oral, once-daily lipid-lowering agent poses an alternative to PCSK9 inhibitors, but further data regarding CV outcomes with this agent is needed.10,11 Robust randomized controlled trials also are needed to evaluate CV outcomes for alirocumab use in statin-intolerant veterans.
Limitations
Only 24 veterans were included in the study, reflecting 20% of the charts reviewed (80% exclusion rate), and in this small sample, only 1 CV event was observed. Both of these serve as threats to external validity. As the study information was extracted from chart review, the results may be limited by coding or historical bias. Medical information from outside institutions may be missing from medical records. Additionally, results may be skewed by possible documentation errors. Furthermore, the period between previous CV events and alirocumab initiation is unclear as event dates were often not recorded if treatment was received at an outside institution.
Due to the short follow-up period, the case series is limited in its assessment of CV outcomes and safety outcomes. Larger studies over an extended period are needed to assess CV outcomes and safety of alirocumab use in statin-intolerant patients. Also, medication adherence was not assessed. Given the impact of medication adherence on LDL-C reduction, it is unclear what role medication adherence played in the LDL-C reduction observed in this study.4
Conclusions
Alirocumab use in 24 statin-intolerant veterans resulted in a significant reduction in LDL-C at 4 and 24 weeks after initiation. In addition, 1 CV event/intervention was observed following alirocumab initiation, although this should be interpreted with caution due to the retrospective nature of this case series, small sample size, and short follow-up period. Large, long-term studies would better evaluate the CV benefit associated with alirocumab therapy in a veteran population.
In 2016, 17.6 million deaths occurred globally due to cardiovascular disease (CVD) with coronary artery disease (CAD) and ischemic stroke as top contributors.1 Elevated low-density lipoprotein cholesterol (LDL-C) has been linked to greater risk of atherosclerotic cardiovascular disease (ASCVD); therefore, LDL-C reduction is imperative to decrease risk of cardiovascular (CV) morbidity and mortality.2 Since 1987, statin therapy has been the mainstay of treatment for hypercholesterolemia, and current practice guidelines recommend statins as first-line therapy given demonstrated reductions in LDL-C and CV mortality reduction in robust clinical trials.2-4 Although generally safe and well tolerated, muscle-related adverse events (AEs) limit optimal use of statins in up to 20% of individuals who have an indication for statin therapy.5 As a consequence, these patients receive suboptimal statin doses or no statin therapy and are at a higher risk for ASCVD.5
Proprotein convertase subtilisin/kexin type 9 (PCSK9) inhibitors have been shown to significantly lower LDL-C when used as monotherapy or in combination with statins and/or other lipid-lowering therapies.5 These agents are currently approved by the US Food and Drug Administration as an adjunct to diet with or without other lipid-lowering therapies for the management of primary hypercholesterolemia (including heterozygous familial hypercholesterolemia), homozygous familial hypercholesterolemia (evolocumab only), and for use in patients with established CVD unable to achieve their lipid-lowering goals with maximally tolerated statin doses and ezetimibe.4 With the ability to reduce LDL-C by up to 65%, PCSK9 inhibitors offer an alternative option for LDL-C and potentially CV risk reduction in statin-intolerant patients.5
Alirocumab, the formulary preferred PCSK9 inhibitor at the Michael E. DeBakey Veterans Affairs Medical Center (MEDVAMC) in Houston, Texas, has been increasingly used in high-risk statin-intolerant veterans. The primary objective of this case series was to assess LDL-C reduction associated with alirocumab use in statin-intolerant veterans at the MEDVAMC. The secondary objective was to assess the incidence of CV events. This study was approved by the MEDVAMC Quality Assurance and Regulatory Affairs committee.
Methods
In this single-center case series, a retrospective chart review was conducted to identify statin-intolerant veterans who were initiated on treatment with alirocumab for LDL-C and/or CV risk reduction between June 2017 and May 2019. Adult veterans with a diagnosis of primary hypercholesterolemia (including heterozygous familial hypercholesterolemia) and/or CAD with documented statin intolerance were included in the study. Statin intolerance was defined in accordance with the National Lipid Association (NLA) definition as aninability to tolerate ≥ 2 statins with a trial of at least 1 statin at its lowest daily dose.5 Veterans who previously received treatment with evolocumab, those prescribed concurrent statin therapies, and those missing follow-up lipid panels at 24 weeks were excluded from the study. To assess LDL-C reduction, LDL-C at baseline was compared with LDL-C at 4 and 24 weeks. Incident CV events before and after alirocumab initiation were documented. The US Department of Veteran Affairs (VA) Computerized Patient Record System was used to collect patient data.
Data Collection, Measures, and Analysis
Electronic health records of all eligible patients who received alirocumab were reviewed, and basic demographics (patient age, sex, and race/ethnicity) as well as medical characteristics at baseline were collected. To confirm statin intolerance, each veteran’s history of statin use and use of additional lipid-lowering agents was documented. CV history was measured with an index of categorical measures for hypertension, confirmed CAD, hyperlipidemia, heart failure, arrhythmias, peripheral artery disease, stroke, diabetes mellitus, and hypothyroidism. Additionally, concomitant medications, such as aspirin, P2Y12 inhibitors, β-blockers, angiotensin-converting enzyme inhibitors, and angiotensin II receptor blockers that patients were taking also were collected. Each veteran’s lipid panel at baseline, and at 4 and 24 weeks posttreatment initiation, also was extracted. Continuous variables were summarized with means (SD), and categorical variables were summarized with frequencies and proportions. The paired Wilcoxon signed rank test was used to compare LDL-C at 4 and 24 weeks after alirocumab initiation with patients’ baseline LDL-C.
Results
Between June 2017 and May 2019, 122 veterans were initiated on alirocumab. Of these veterans, 98 were excluded: 35 concurrently received statin therapy, 33 missed follow-up lipid panels, 21 had previously received evolocumab, 6 failed to meet the NLA definition for statin intolerance, 2 did not fill active alirocumab prescriptions, and 1 had an incalculable LDL-C with a baseline triglyceride level of 3079 mg/dL. This resulted in 24 veterans included in the analysis.
Most participants were male (87.5%) and White veterans (79.2%) with a mean (SD) age of 66.0 (8.4) years and mean (SD) baseline LDL-C of 161.9 (74.3) mg/dL. At baseline, 21 veterans had a history of primary hyperlipidemia, 19 had a history of CAD, and 2 had a history of heterozygous familial hypercholesterolemia. Of the 24 patients included, the most trialed statins before alirocumab initiation were atorvastatin (95.8%), simvastatin (79.2%), rosuvastatin (79.2%), and pravastatin (62.5%) (Table).
LDL-C Reduction
Veterans were initially treated with alirocumab 75 mg administered subcutaneously every 2 weeks; however, 11 veterans required a dose increase to 150 mg every 2 weeks. At treatment week 4, the median LDL-C reduction was 78.5 mg/dL (IQR, 28.0-107.3; P < .01), and at treatment week 24, the median LDL-C reduction was 55.6 mg/dL (IQR, 18.6-85.3; P < .01). This equated to median LDL-C reductions from baseline of 48.5% at week 4 and 34.3% at week 24. A total of 3 veterans experienced LDL-C increases following initiation of alirocumab. At week 4, 9 veterans were noted to have an LDL-C reduction > 50%, 7 veterans had an LDL-C reduction between 30% and 50%, and 5 veterans had an LDL-C reduction of < 30%. At week 24, 6 had an LDL-C reduction > 50%, 9 veterans had an LDL-C reduction between 30% and 50%, and 6 had a LDL-C reduction < 30%.
Cardiovascular Events
Before alirocumab initiation, 22 CV events and interventions were reported in 16 veterans: 12 percutaneous coronary interventions, 5 coronary artery bypass surgeries (CABG), 4 myocardial infarctions, and 1 transient ischemic attack. One month following alirocumab initiation, 1 veteran underwent a CABG after a non-ST-elevation myocardial infarction (NSTEMI).
Safety and Tolerability
Alirocumab was discontinued in 5 veterans due to 4 cases of intolerance (reported memory loss, lethargy, myalgias, and body aches with dyspnea) and 1 case of persistent LDL-C of < 40 mg/dL. Alirocumab was discontinued after 1 year in 2 patients (persistent LDL-C < 40 mg/dL and reported memory loss) and after 6 months in the veteran who reported lethargy. Alirocumab was discontinued after 4 months in the veteran with myalgias and within 2 months in the veteran with body aches and dyspnea. No other AEs were reported.
Discussion
The Efficacy and Safety of Alirocumab vs Ezetimibe in Statin-Intolerant Veterans With a Statin Rechallenge Arm trial is the first clinical trial to examine the efficacy and safety of alirocumab use in statin-intolerant patients. In the trial, 314 patients were randomized to receive alirocumab, ezetimibe, or an atorvastatin rechallenge.6 At 24 weeks, alirocumab reduced mean (SE) LDL-C by 45.0% (2.2%) vs 14.6% (2.2%) with ezetimibe (mean difference 30.4% [3.1%], P < .01).6 Fewer skeletal-muscle-related events also were noted with alirocumab vs atorvastatin (hazard ratio, 0.61; 95% CI, 0.38-0.99; P = .04).6
In this case series, an LDL-C reduction of > 50% was observed in 9 veterans (42.9%) following 4 weeks of treatment; however, LDL-C reduction of > 50% compared with baseline was sustained in only 6 veterans (28.6%) at week 24. Additionally, LDL-C increases from baseline were observed in 3 veterans; the reasoning for the observed increase was unclear, but this may have been due to nonadherence and dietary factors.4 Although a majority of patients saw a significant and clinically meaningful reduction in LDL-C, the group of patients with an increase in the same may have benefitted from targeted intervention to improve medication and dietary adherence. PCSK9 inhibitor resistance also may have contributed to an increase in LDL-C during treatment.7
Of the 24 patients included, 4 reported AEs resulted in therapy discontinuation. Memory impairment, a rare AE of alirocumab, was reported 1 year following alirocumab initiation. Additionally, lethargy was reported after 6 months of treatment. Myalgia also was reported in a veteran 4 months following treatment, and 1 veteran experienced body aches and dyspnea < 2 months following treatment. The most common AEs associated with alirocumab, as noted in previous safety and efficacy clinical trials, included: nasopharyngitis, injection site reaction, influenza, urinary tract infection, and myalgias.8 Many of these more common AEs may be subclinical and underreported. This small case series, however, detected 4 events severe enough to lead to therapy discontinuation. Although this sample is not representative of all statin-intolerant patients who receive treatment with alirocumab, our findings suggest the need for patient education on potential AEs before therapy initiation and clinician monitoring at follow-up visits.
The ODYSSEY OUTCOMES trial established a CV benefit associated with alirocumab; however, patients included had a recent acute coronary syndrome event and were receiving a high-intensity statin.9 This case series is unique in that before alirocumab initiation, 22 CV events/interventions were reported in the sample of 24 patients. After therapy initiation, 1 patient underwent a CABG after an NSTEMI in the month following initiation. This suggests that cardiac complications are possible after PCSK-9 initiation; however, little information can be gained from 1 patient. Nevertheless, early therapy failure should be investigated in the context of real-world use in statin-intolerant patients. This is a complex task, however, given the difficulties of achieving a balanced study design. Statin intolerance is a clear source of selection bias into treatment with alirocumab as patients in this population have already initiated and failed statin therapy. The prevalence of prior CV events and the time-dependent association between prior and future CV events stand as another complex confounder. Although there is a clear and pressing need to understand the risks and benefits of PCSK9 therapy in statin-intolerant patients, future research in this area will need to cautiously address these important sources of bias.
Overall, the results of this case series support LDL-C reduction associated with alirocumab in the absence of statin therapy. Despite favorable results, use of alirocumab may be limited by cost and its subcutaneous route of administration. Bempedoic acid, an oral, once-daily lipid-lowering agent poses an alternative to PCSK9 inhibitors, but further data regarding CV outcomes with this agent is needed.10,11 Robust randomized controlled trials also are needed to evaluate CV outcomes for alirocumab use in statin-intolerant veterans.
Limitations
Only 24 veterans were included in the study, reflecting 20% of the charts reviewed (80% exclusion rate), and in this small sample, only 1 CV event was observed. Both of these serve as threats to external validity. As the study information was extracted from chart review, the results may be limited by coding or historical bias. Medical information from outside institutions may be missing from medical records. Additionally, results may be skewed by possible documentation errors. Furthermore, the period between previous CV events and alirocumab initiation is unclear as event dates were often not recorded if treatment was received at an outside institution.
Due to the short follow-up period, the case series is limited in its assessment of CV outcomes and safety outcomes. Larger studies over an extended period are needed to assess CV outcomes and safety of alirocumab use in statin-intolerant patients. Also, medication adherence was not assessed. Given the impact of medication adherence on LDL-C reduction, it is unclear what role medication adherence played in the LDL-C reduction observed in this study.4
Conclusions
Alirocumab use in 24 statin-intolerant veterans resulted in a significant reduction in LDL-C at 4 and 24 weeks after initiation. In addition, 1 CV event/intervention was observed following alirocumab initiation, although this should be interpreted with caution due to the retrospective nature of this case series, small sample size, and short follow-up period. Large, long-term studies would better evaluate the CV benefit associated with alirocumab therapy in a veteran population.
1. Benjamin EJ, Munter P, Alonso A, et al; American Heart Association Council on Epidemiology and Prevention Statistics Committee and Stroke Statistics Subcommittee. Heart disease and stroke statistics—2019 update: a report from the American Heart Association. Circulation. 2019;139(10):e56-e528. doi:10.1161/CIR.0000000000000659
2. Stone NJ, Robinson JG, Lichtenstein AH, et al; American College of Cardiology/American Heart Association Task Force on Practice Guidelines. 2013 ACC/AHA guideline on the treatment of blood cholesterol to reduce atherosclerotic cardiovascular risk in adults: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. Circulation. 2014 Jun 24;129(25)(suppl 2):S1-S45. doi:10.1016/j.jacc.2013.11.002
3. Hajar R. Statins: past and present. Heart Views. 2011;12(3): 121-127. doi:10.4103/1995-705X.95070
4. Grundy SM, Stone NJ, Bailey AL, et al. 2018 AHA/ACC/AACVPR/AAPA/ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA guideline on the management of blood cholesterol: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. J Am Coll Cardiol 2019;73(4):3168-3209. doi:10.1016/j.jacc.2018.11.002
5. Toth PH, Patti AM, Giglio RV, et al. Management of statin intolerance in 2018: still more questions than answers. Am J Cardiovasc Drugs. 2018;18(3):157-173. doi:10.1007/s40256-017-0259-7
6. Moriarty PM, Thompson PD, Cannon CP, et al; ODYSSEY ALTERNATIVE Investigators. Efficacy and safety of alirocumab vs ezetimibe in statin-intolerant patients, with a statin rechallenge arm: The ODYSSEY ALTERNATIVE randomized trial. J Clin Lipidol. 2015;9(6):758-769. doi:10.1016/j.jacl.2015.08.006
7. Shapiro MD, Miles J, Tavori H, Fazio S. Diagnosing resistance to a proprotein convertase subtilisin/kexin type 9 inhibitor. Ann Intern Med. 2018;168(5):376-379. doi:10.7326/M17-2485
8. Raedler LA. Praluent (alirocumab): first PCSK9 inhibitor approved by the FDA for hypercholesterolemia. Am Health Drug Benefits. 2016;9:123-126.
9. Schwartz GC, Steg PC, Szarek M, et al; ODYSSEY OUTCOMES Committees and Investigators. Alirocumab and cardiovascular outcomes after acute coronary syndrome. N Engl J Med. 2018;379(22):2097-2107. doi:10.1056/NEJMoa1801174
10. Nexletol. Package insert. Esperion Therapeutics Inc; 2020.
11. Laufs U, Banach M, Mancini GBJ, et al. Efficacy and safety of bempedoic acid in patients with hypercholesterolemia and statin intolerance. J Am Heart Assoc. 2019;8(7):e011662. doi:10.1161/JAHA.118.011662
1. Benjamin EJ, Munter P, Alonso A, et al; American Heart Association Council on Epidemiology and Prevention Statistics Committee and Stroke Statistics Subcommittee. Heart disease and stroke statistics—2019 update: a report from the American Heart Association. Circulation. 2019;139(10):e56-e528. doi:10.1161/CIR.0000000000000659
2. Stone NJ, Robinson JG, Lichtenstein AH, et al; American College of Cardiology/American Heart Association Task Force on Practice Guidelines. 2013 ACC/AHA guideline on the treatment of blood cholesterol to reduce atherosclerotic cardiovascular risk in adults: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. Circulation. 2014 Jun 24;129(25)(suppl 2):S1-S45. doi:10.1016/j.jacc.2013.11.002
3. Hajar R. Statins: past and present. Heart Views. 2011;12(3): 121-127. doi:10.4103/1995-705X.95070
4. Grundy SM, Stone NJ, Bailey AL, et al. 2018 AHA/ACC/AACVPR/AAPA/ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA guideline on the management of blood cholesterol: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. J Am Coll Cardiol 2019;73(4):3168-3209. doi:10.1016/j.jacc.2018.11.002
5. Toth PH, Patti AM, Giglio RV, et al. Management of statin intolerance in 2018: still more questions than answers. Am J Cardiovasc Drugs. 2018;18(3):157-173. doi:10.1007/s40256-017-0259-7
6. Moriarty PM, Thompson PD, Cannon CP, et al; ODYSSEY ALTERNATIVE Investigators. Efficacy and safety of alirocumab vs ezetimibe in statin-intolerant patients, with a statin rechallenge arm: The ODYSSEY ALTERNATIVE randomized trial. J Clin Lipidol. 2015;9(6):758-769. doi:10.1016/j.jacl.2015.08.006
7. Shapiro MD, Miles J, Tavori H, Fazio S. Diagnosing resistance to a proprotein convertase subtilisin/kexin type 9 inhibitor. Ann Intern Med. 2018;168(5):376-379. doi:10.7326/M17-2485
8. Raedler LA. Praluent (alirocumab): first PCSK9 inhibitor approved by the FDA for hypercholesterolemia. Am Health Drug Benefits. 2016;9:123-126.
9. Schwartz GC, Steg PC, Szarek M, et al; ODYSSEY OUTCOMES Committees and Investigators. Alirocumab and cardiovascular outcomes after acute coronary syndrome. N Engl J Med. 2018;379(22):2097-2107. doi:10.1056/NEJMoa1801174
10. Nexletol. Package insert. Esperion Therapeutics Inc; 2020.
11. Laufs U, Banach M, Mancini GBJ, et al. Efficacy and safety of bempedoic acid in patients with hypercholesterolemia and statin intolerance. J Am Heart Assoc. 2019;8(7):e011662. doi:10.1161/JAHA.118.011662
Continuous Blood Glucose Monitoring Outcomes in Veterans With Type 2 Diabetes
Nearly 25% of patients served in the US Department of Veterans Affairs (VA) have been diagnosed with type 2 diabetes mellitus (T2DM), although the prevalence among adults in the United States is 9%.1 Patients with DM typically monitor their blood glucose using intermittent fingerstick self-testing. Continuous glucose monitoring (CGM) might offer a more comprehensive picture of glucose control to improve disease management. Within the VA, criteria for CGM use varies among facilities, but generally veterans prescribed at least 3 daily insulin injections and 4 daily blood glucose checks qualify.2
CGM therapy has been extensively researched for type 1 DM (T1DM); however, outcomes of CGM use among older adults with T2DM have not been fully evaluated. In a 2018 review of randomized clinical trials evaluating CGM use, 17 trials examined only patients with T1DM (2009 participants), 4 included only patients with T2DM patients (547 patients), 3 evaluated patients with T1DM or T2DM (655 patients), and 3 included women with gestational diabetes (585 patients).3 Of 27 studies that included change in hemoglobin A1c (HbA1c) as an endpoint, 15 found a statistically significant reduction in HbA1c for the CGM group. Four trials evaluated CGM use in adults with T2DM and 3 found no difference in HbA1c overall. However, 1 study found a difference in HbA1c only in individuals aged < 65 years, and another study found a greater improvement in the CGM group (approximately 0.5%).4,5 These mixed results indicate a need for further subgroup analysis in specific populations to determine the optimal use of CGM in adults with T2DM. Although this study was not designed to measure changes in hypoglycemic episodes or the relative efficacy of different CGM products, it establishes a baseline from which to conduct additional research.
Our primary objective was to determine change in HbA1c in each patient from the year before CGM initiation to the year after. Secondary objectives included changes in blood pressure (BP), weight, and diabetes-related hospital and clinic visits during the same time frame. We also completed subanalysis comparing primary outcomes in engaged or adherent patients compared with the entire study group. This study was completed as a quality improvement project with approval from the Lexington Veterans Affairs Health Care System in Kentucky information security office and was exempted from institutional review board review.
Methods
This project was a retrospective evaluation using the VA database of patient records. Rather than using a control group, our study used a pre–post model to determine the impact of CGM for each patient. For the primary outcome, average HbA1c values were calculated for the year before and year after CGM initiation. Hemoglobin and hematocrit values were included if reported within 3 months of the HbA1c values to ensure validity of HbA1c results. Average HbA1c was 13.37 g/dL (range, 10.5-17.3), and average hematocrit was 43.3% (range, 36-52). Change in average HbA1c was recorded for each patient. Based on research by Taylor and colleagues, a change in HbA1c of 0.8% was considered clinically significant for this project.6
Mean BP and weight were calculated for the years before and after CGM initiation. Only values for routine clinic visits were included; values taken during an acute health incident, inpatient stay, infusion clinic appointments, or home readings were excluded. Changes were recorded for each patient. Patient encounter notes were used to determine the number of DM-related hospital, emergency department, and clinic visits, such as nephrology, podiatry, vascular medicine, or infectious disease clinic or inpatient encounters during the study period. Routine endocrinology or primary care visits were not included, and patient care notes were consulted to ensure that the encounters were related to a DM complication. The change in number of visits was calculated for each patient.
Adherence was defined as patients receiving active medication management, documented treatment regimen adherence, and > 4 annual endocrinology clinic visits. Active medication management was defined as having > 1 dosage or medication change for oral or noninsulin antihyperglycemics, initiation, or adjustment of insulin dosages according to the patient records. Treatment adherence was determined based on medication reconciliation notes and refill request history. Only endocrinology clinic visits at VA outpatient clinics were included.
Study Population
A sample of 166 patients was needed to detect an HbA1c change of 0.8 per power analysis. The normal approximation method using the z statistic was used, with 2-tailed α = 0.05, β = 0.05, E = 0.8, and S = 1.2. We randomly selected 175 patients among all individuals with an active prescription for CGM in 2018 and 2019, who had a diagnosis of T2DM, and were managed by VA endocrinology clinics (including endocrine clinics, diabetes clinics, and patient aligned care team clinics) at 87 VA medical centers. Patients with types of DM other than T2DM were excluded, as well as those with a diagnosed hemoglobinopathy or HbA1c < 10 g/dL. The adherent subgroup included 40 patients of the 175 sample population (Table 1).
Results
Both the total population and the adherent subgroup showed reduction in HbA1c, the primary endpoint. The complete population showed a HbA1c change of –0.3 (95% CI, –0.4 to –0.2), and the adherent subgroup had a change of –1.3 (95% CI, –1.5 to –1.2). The total survey population had a mean change in weight of –1.9 lb (–0.9 kg) (95% CI, –3.7 to –0.1) and the adherent subgroup had an average change of –8.0 lb (–3.6 kg) (95% CI, –12.3 to –3.8). Average systolic BP changes were –0.1 mm Hg (95% CI, –1.6 to 1.5) in the total population and +3.3 mm Hg (95% CI, –0.01 to 6.22) in the adherent subgroup. A decrease in total encounters for DM complications was observed in the population (–0.3 total encounters per patient, 95% CI, –0.5 to –0.2) and the adherent subgroup (–0.6 total encounters per patient, 95% CI, –1.0 to –0.1) (Table 2).
Before the study, 107 (61.1%) patients were taking oral or noninsulin DM medication only, 4 (2.3%) were on insulin only, and 64 (36.6%) were prescribed both insulin and oral/noninsulin antihyperglycemics. Noninsulin and oral antihyperglycemic regimens included combinations of biguanide, dipeptidyl peptidase- 4 inhibitor, sodium-glucose cotransporter-2 inhibitor, sulfonylurea, meglitinide, β-glucosidase inhibitor, glucagon-like peptide-1 (GLP-1) analog, and thiazolidinedione drug classes. Nearly 70% (122) had no reported changes in DM treatment beyond dosage titrations. Among these patients, 18 (10.3%) were on an insulin pump for the duration of the study. Among the 53 (30.3%) patients who had changes in treatment, 31 (17.7%) transitioned from insulin injections to an insulin pump, 13 (7.4%) changed from 1 insulin injection to another (ie, addition of long-acting insulin, transition to u500 insulin, changing from 1 insulin category or brand to another), 8 (4.6%) began an oral/noninsulin antihyperglycemic, 4 (2.3%) began insulin injections, 13 (7.4%) discontinued noninsulin or oral antihyperglycemics, and 2 (1.1%) discontinued insulin during the study period.
Data showed that 113 (64.5%) patients had no changes in antihypertensives. The remaining 62 (35.4%) had the following adjustments: 14 (8%) increased dose of current medication(s), 9 (5.1%) decreased dose of current medication(s), 8 (4.6%) discontinued all antihypertensive medications, 10 (5.7%) switched to a different antihypertensive class, and 16 (9.1%) added additional antihypertensive medication(s) to their existing regimen during the study period.
Patients in the study group used 7 different types of CGM sensors. Chart review revealed that 84 (47.7%) patients used Medtronic devices, with 26 (14.8%) using first-generation Guardian sensors, 50 (28.4%) using Enlite sensors, and 8 (4.5) using Guardian 3 sensors. We found that 81 (46.0%) veterans were prescribed Dexcom devices, with 5 (2.8%) using SEVEN PLUS sensors, 68 (38.6%) using G4-5 sensors, and 8 (4.5%) using G6 sensors. The remaining 10 (5.7%) patients were using Freestyle Libre sensors during the study period.
Discussion
CGM did not correspond with clinically significant reductions in HbA1c. However, veterans with increased health care engagement were likely to achieve clinically significant HbA1c improvements. The veterans in the adherent subgroup had a higher baseline HbA1c, which could be because of a variety of factors mentioned in patient care notes, including insulin resistance, poor dietary habits, and exercise regimen nonadherence. These patients might have had more room to improve their glycemic control without concern of hypoglycemia, and their higher baseline HbA1c could have provided increased motivation for improving their health during the study period.
Adherent patients also had a greater reduction in weight and hospital or clinic visits with CGM compared with the total population. These veterans’ increased involvement in their health care might have led to better dietary and exercise adherence, which would have decreased insulin dosing and contributed to weight loss. Only 1 patient in the adherent subgroup initiated a GLP-1 agonist during the study period, making it unlikely that medication changes had a significant impact on weight loss in the subgroup analysis. This improvement in overall health status might have contributed to the reduction in hospital or clinic visits observed in this population.
Average systolic BP data decreased minimally in the total survey population and increased in the adherent subgroup over the course of the study. These results were determined to be statistically significant. Changes in systolic BP readings were minimal, indicating that it is unlikely that these changes contributed meaningfully to the patients’ overall health status.
Although not related to the study objectives, the adherent population required less antihypertensive adjustments with similar BP control. This could be explained by improved overall health or better adherence and engagement in therapy. The results of this project show that despite limited medication changes, T2DM management improved among adherent patients using CGM. The general study population, which was more likely to have documented nonadherence with treatment or clinic appointments, had minimal benefit. CGM technology in the T2DM veteran population is more likely to have significant clinical benefit in patients who are adherent with their medication regimens and follow-up appointments compared with the larger study population.
The results of this study are in line with previous studies on CGM use in the T2DM patient population. We agree with the previously published research that CGM alone does not have a meaningful impact on HbA1c reduction. Our study population also was older than those in previous studies, adding to the Haak and colleagues conclusion that patients aged < 65 years might have better outcomes with CGM.4
Strengths of this study include specificity to the veteran population using VA resources, as well as including nondiabetes outcomes. This allows for specific application to the veteran population and could provide broader evidence for CGM use. Demonstrated decreases in HbA1c, weight, and clinic visits in the adherent population suggest that providing veterans with CGM therapy with frequent endocrinology follow-up improves health outcomes and could decrease overall health spending.
Limitations
Limitations of this study include retrospective design, a small sample size, and solely focusing on T2DM. As a retrospective study, we cannot rule out the influence of outside factors, such as participation in a non-VA weight loss program. This study lacked the power to assess the impact of the different CGM brands. The study did not include data on severe hypoglycemic or hyperglycemic episodes as veterans might have needed emergent care at non-VA facilities. Future research will evaluate the impact of CGM on symptomatic and severe hypoglycemic episodes and use of insulin vs oral or noninsulin antihyperglycemics and the comparative efficacy of different CGM brands among veterans.
Conclusions
CGM did not correspond with clinically significant reductions in HbA1c. However, veterans with increased health care engagement were likely to achieve clinically significant HbA1c improvements. Adherent patients also had more reduction in weight and hospital or clinic visits with CGM compared with the total population. These veterans’ increased involvement in their health care might have led to better dietary and exercise adherence, which would have decreased insulin dosing and contributed to weight loss.
1. Liu Y, Sayam S, Shao X, et al. Prevalence of and trends in diabetes among veterans, United States, 2005-2014. Prev Chronic Dis. 2017;14:E135. Published 2017 Dec 14. doi:10.5888/pcd14.170230
2. Hackett M. VA pharmacies now carry the Dexcom G6 CGM at no cost for qualifying patients. September 23, 2020. Accessed September 28, 2021. https://www.mobihealthnews.com/news/va-pharmacies-now-carry-dexcom-g6-cgm-no-cost-qualifying-patients
3. Peters AL. The evidence base for continuous glucose monitoring. In: Role of Continuous Glucose Monitoring in Diabetes Treatment. Arlington (VA): American Diabetes Association; August 2018.3-7. doi:10.2337/db20181-3
4. Haak T, Hanaire H, Ajjan R, Hermanns N, Riveline JP, Rayman G. Flash glucose-sensing technology as a replacement for blood glucose monitoring for the management of insulin-treated type 2 diabetes: a multicenter, open-label randomized controlled trial. Diabetes Ther. 2017;8(1):55-73. doi:10.1007/s13300-016-0223-6
5. Yoo HJ, An HG, Park SY, et al. Use of a real time continuous glucose monitoring system as a motivational device for poorly controlled type 2 diabetes. Diabetes Res Clin Pract. 2008;82(1):73-79. doi:10.1016/j.diabres.2008.06.015
6. Taylor PJ, Thompson CH, Brinkworth GD. Effectiveness and acceptability of continuous glucose monitoring for type 2 diabetes management: A narrative review. J Diabetes Investig. 2018;9(4):713-725. doi:10.1111/jdi.12807
Nearly 25% of patients served in the US Department of Veterans Affairs (VA) have been diagnosed with type 2 diabetes mellitus (T2DM), although the prevalence among adults in the United States is 9%.1 Patients with DM typically monitor their blood glucose using intermittent fingerstick self-testing. Continuous glucose monitoring (CGM) might offer a more comprehensive picture of glucose control to improve disease management. Within the VA, criteria for CGM use varies among facilities, but generally veterans prescribed at least 3 daily insulin injections and 4 daily blood glucose checks qualify.2
CGM therapy has been extensively researched for type 1 DM (T1DM); however, outcomes of CGM use among older adults with T2DM have not been fully evaluated. In a 2018 review of randomized clinical trials evaluating CGM use, 17 trials examined only patients with T1DM (2009 participants), 4 included only patients with T2DM patients (547 patients), 3 evaluated patients with T1DM or T2DM (655 patients), and 3 included women with gestational diabetes (585 patients).3 Of 27 studies that included change in hemoglobin A1c (HbA1c) as an endpoint, 15 found a statistically significant reduction in HbA1c for the CGM group. Four trials evaluated CGM use in adults with T2DM and 3 found no difference in HbA1c overall. However, 1 study found a difference in HbA1c only in individuals aged < 65 years, and another study found a greater improvement in the CGM group (approximately 0.5%).4,5 These mixed results indicate a need for further subgroup analysis in specific populations to determine the optimal use of CGM in adults with T2DM. Although this study was not designed to measure changes in hypoglycemic episodes or the relative efficacy of different CGM products, it establishes a baseline from which to conduct additional research.
Our primary objective was to determine change in HbA1c in each patient from the year before CGM initiation to the year after. Secondary objectives included changes in blood pressure (BP), weight, and diabetes-related hospital and clinic visits during the same time frame. We also completed subanalysis comparing primary outcomes in engaged or adherent patients compared with the entire study group. This study was completed as a quality improvement project with approval from the Lexington Veterans Affairs Health Care System in Kentucky information security office and was exempted from institutional review board review.
Methods
This project was a retrospective evaluation using the VA database of patient records. Rather than using a control group, our study used a pre–post model to determine the impact of CGM for each patient. For the primary outcome, average HbA1c values were calculated for the year before and year after CGM initiation. Hemoglobin and hematocrit values were included if reported within 3 months of the HbA1c values to ensure validity of HbA1c results. Average HbA1c was 13.37 g/dL (range, 10.5-17.3), and average hematocrit was 43.3% (range, 36-52). Change in average HbA1c was recorded for each patient. Based on research by Taylor and colleagues, a change in HbA1c of 0.8% was considered clinically significant for this project.6
Mean BP and weight were calculated for the years before and after CGM initiation. Only values for routine clinic visits were included; values taken during an acute health incident, inpatient stay, infusion clinic appointments, or home readings were excluded. Changes were recorded for each patient. Patient encounter notes were used to determine the number of DM-related hospital, emergency department, and clinic visits, such as nephrology, podiatry, vascular medicine, or infectious disease clinic or inpatient encounters during the study period. Routine endocrinology or primary care visits were not included, and patient care notes were consulted to ensure that the encounters were related to a DM complication. The change in number of visits was calculated for each patient.
Adherence was defined as patients receiving active medication management, documented treatment regimen adherence, and > 4 annual endocrinology clinic visits. Active medication management was defined as having > 1 dosage or medication change for oral or noninsulin antihyperglycemics, initiation, or adjustment of insulin dosages according to the patient records. Treatment adherence was determined based on medication reconciliation notes and refill request history. Only endocrinology clinic visits at VA outpatient clinics were included.
Study Population
A sample of 166 patients was needed to detect an HbA1c change of 0.8 per power analysis. The normal approximation method using the z statistic was used, with 2-tailed α = 0.05, β = 0.05, E = 0.8, and S = 1.2. We randomly selected 175 patients among all individuals with an active prescription for CGM in 2018 and 2019, who had a diagnosis of T2DM, and were managed by VA endocrinology clinics (including endocrine clinics, diabetes clinics, and patient aligned care team clinics) at 87 VA medical centers. Patients with types of DM other than T2DM were excluded, as well as those with a diagnosed hemoglobinopathy or HbA1c < 10 g/dL. The adherent subgroup included 40 patients of the 175 sample population (Table 1).
Results
Both the total population and the adherent subgroup showed reduction in HbA1c, the primary endpoint. The complete population showed a HbA1c change of –0.3 (95% CI, –0.4 to –0.2), and the adherent subgroup had a change of –1.3 (95% CI, –1.5 to –1.2). The total survey population had a mean change in weight of –1.9 lb (–0.9 kg) (95% CI, –3.7 to –0.1) and the adherent subgroup had an average change of –8.0 lb (–3.6 kg) (95% CI, –12.3 to –3.8). Average systolic BP changes were –0.1 mm Hg (95% CI, –1.6 to 1.5) in the total population and +3.3 mm Hg (95% CI, –0.01 to 6.22) in the adherent subgroup. A decrease in total encounters for DM complications was observed in the population (–0.3 total encounters per patient, 95% CI, –0.5 to –0.2) and the adherent subgroup (–0.6 total encounters per patient, 95% CI, –1.0 to –0.1) (Table 2).
Before the study, 107 (61.1%) patients were taking oral or noninsulin DM medication only, 4 (2.3%) were on insulin only, and 64 (36.6%) were prescribed both insulin and oral/noninsulin antihyperglycemics. Noninsulin and oral antihyperglycemic regimens included combinations of biguanide, dipeptidyl peptidase- 4 inhibitor, sodium-glucose cotransporter-2 inhibitor, sulfonylurea, meglitinide, β-glucosidase inhibitor, glucagon-like peptide-1 (GLP-1) analog, and thiazolidinedione drug classes. Nearly 70% (122) had no reported changes in DM treatment beyond dosage titrations. Among these patients, 18 (10.3%) were on an insulin pump for the duration of the study. Among the 53 (30.3%) patients who had changes in treatment, 31 (17.7%) transitioned from insulin injections to an insulin pump, 13 (7.4%) changed from 1 insulin injection to another (ie, addition of long-acting insulin, transition to u500 insulin, changing from 1 insulin category or brand to another), 8 (4.6%) began an oral/noninsulin antihyperglycemic, 4 (2.3%) began insulin injections, 13 (7.4%) discontinued noninsulin or oral antihyperglycemics, and 2 (1.1%) discontinued insulin during the study period.
Data showed that 113 (64.5%) patients had no changes in antihypertensives. The remaining 62 (35.4%) had the following adjustments: 14 (8%) increased dose of current medication(s), 9 (5.1%) decreased dose of current medication(s), 8 (4.6%) discontinued all antihypertensive medications, 10 (5.7%) switched to a different antihypertensive class, and 16 (9.1%) added additional antihypertensive medication(s) to their existing regimen during the study period.
Patients in the study group used 7 different types of CGM sensors. Chart review revealed that 84 (47.7%) patients used Medtronic devices, with 26 (14.8%) using first-generation Guardian sensors, 50 (28.4%) using Enlite sensors, and 8 (4.5) using Guardian 3 sensors. We found that 81 (46.0%) veterans were prescribed Dexcom devices, with 5 (2.8%) using SEVEN PLUS sensors, 68 (38.6%) using G4-5 sensors, and 8 (4.5%) using G6 sensors. The remaining 10 (5.7%) patients were using Freestyle Libre sensors during the study period.
Discussion
CGM did not correspond with clinically significant reductions in HbA1c. However, veterans with increased health care engagement were likely to achieve clinically significant HbA1c improvements. The veterans in the adherent subgroup had a higher baseline HbA1c, which could be because of a variety of factors mentioned in patient care notes, including insulin resistance, poor dietary habits, and exercise regimen nonadherence. These patients might have had more room to improve their glycemic control without concern of hypoglycemia, and their higher baseline HbA1c could have provided increased motivation for improving their health during the study period.
Adherent patients also had a greater reduction in weight and hospital or clinic visits with CGM compared with the total population. These veterans’ increased involvement in their health care might have led to better dietary and exercise adherence, which would have decreased insulin dosing and contributed to weight loss. Only 1 patient in the adherent subgroup initiated a GLP-1 agonist during the study period, making it unlikely that medication changes had a significant impact on weight loss in the subgroup analysis. This improvement in overall health status might have contributed to the reduction in hospital or clinic visits observed in this population.
Average systolic BP data decreased minimally in the total survey population and increased in the adherent subgroup over the course of the study. These results were determined to be statistically significant. Changes in systolic BP readings were minimal, indicating that it is unlikely that these changes contributed meaningfully to the patients’ overall health status.
Although not related to the study objectives, the adherent population required less antihypertensive adjustments with similar BP control. This could be explained by improved overall health or better adherence and engagement in therapy. The results of this project show that despite limited medication changes, T2DM management improved among adherent patients using CGM. The general study population, which was more likely to have documented nonadherence with treatment or clinic appointments, had minimal benefit. CGM technology in the T2DM veteran population is more likely to have significant clinical benefit in patients who are adherent with their medication regimens and follow-up appointments compared with the larger study population.
The results of this study are in line with previous studies on CGM use in the T2DM patient population. We agree with the previously published research that CGM alone does not have a meaningful impact on HbA1c reduction. Our study population also was older than those in previous studies, adding to the Haak and colleagues conclusion that patients aged < 65 years might have better outcomes with CGM.4
Strengths of this study include specificity to the veteran population using VA resources, as well as including nondiabetes outcomes. This allows for specific application to the veteran population and could provide broader evidence for CGM use. Demonstrated decreases in HbA1c, weight, and clinic visits in the adherent population suggest that providing veterans with CGM therapy with frequent endocrinology follow-up improves health outcomes and could decrease overall health spending.
Limitations
Limitations of this study include retrospective design, a small sample size, and solely focusing on T2DM. As a retrospective study, we cannot rule out the influence of outside factors, such as participation in a non-VA weight loss program. This study lacked the power to assess the impact of the different CGM brands. The study did not include data on severe hypoglycemic or hyperglycemic episodes as veterans might have needed emergent care at non-VA facilities. Future research will evaluate the impact of CGM on symptomatic and severe hypoglycemic episodes and use of insulin vs oral or noninsulin antihyperglycemics and the comparative efficacy of different CGM brands among veterans.
Conclusions
CGM did not correspond with clinically significant reductions in HbA1c. However, veterans with increased health care engagement were likely to achieve clinically significant HbA1c improvements. Adherent patients also had more reduction in weight and hospital or clinic visits with CGM compared with the total population. These veterans’ increased involvement in their health care might have led to better dietary and exercise adherence, which would have decreased insulin dosing and contributed to weight loss.
Nearly 25% of patients served in the US Department of Veterans Affairs (VA) have been diagnosed with type 2 diabetes mellitus (T2DM), although the prevalence among adults in the United States is 9%.1 Patients with DM typically monitor their blood glucose using intermittent fingerstick self-testing. Continuous glucose monitoring (CGM) might offer a more comprehensive picture of glucose control to improve disease management. Within the VA, criteria for CGM use varies among facilities, but generally veterans prescribed at least 3 daily insulin injections and 4 daily blood glucose checks qualify.2
CGM therapy has been extensively researched for type 1 DM (T1DM); however, outcomes of CGM use among older adults with T2DM have not been fully evaluated. In a 2018 review of randomized clinical trials evaluating CGM use, 17 trials examined only patients with T1DM (2009 participants), 4 included only patients with T2DM patients (547 patients), 3 evaluated patients with T1DM or T2DM (655 patients), and 3 included women with gestational diabetes (585 patients).3 Of 27 studies that included change in hemoglobin A1c (HbA1c) as an endpoint, 15 found a statistically significant reduction in HbA1c for the CGM group. Four trials evaluated CGM use in adults with T2DM and 3 found no difference in HbA1c overall. However, 1 study found a difference in HbA1c only in individuals aged < 65 years, and another study found a greater improvement in the CGM group (approximately 0.5%).4,5 These mixed results indicate a need for further subgroup analysis in specific populations to determine the optimal use of CGM in adults with T2DM. Although this study was not designed to measure changes in hypoglycemic episodes or the relative efficacy of different CGM products, it establishes a baseline from which to conduct additional research.
Our primary objective was to determine change in HbA1c in each patient from the year before CGM initiation to the year after. Secondary objectives included changes in blood pressure (BP), weight, and diabetes-related hospital and clinic visits during the same time frame. We also completed subanalysis comparing primary outcomes in engaged or adherent patients compared with the entire study group. This study was completed as a quality improvement project with approval from the Lexington Veterans Affairs Health Care System in Kentucky information security office and was exempted from institutional review board review.
Methods
This project was a retrospective evaluation using the VA database of patient records. Rather than using a control group, our study used a pre–post model to determine the impact of CGM for each patient. For the primary outcome, average HbA1c values were calculated for the year before and year after CGM initiation. Hemoglobin and hematocrit values were included if reported within 3 months of the HbA1c values to ensure validity of HbA1c results. Average HbA1c was 13.37 g/dL (range, 10.5-17.3), and average hematocrit was 43.3% (range, 36-52). Change in average HbA1c was recorded for each patient. Based on research by Taylor and colleagues, a change in HbA1c of 0.8% was considered clinically significant for this project.6
Mean BP and weight were calculated for the years before and after CGM initiation. Only values for routine clinic visits were included; values taken during an acute health incident, inpatient stay, infusion clinic appointments, or home readings were excluded. Changes were recorded for each patient. Patient encounter notes were used to determine the number of DM-related hospital, emergency department, and clinic visits, such as nephrology, podiatry, vascular medicine, or infectious disease clinic or inpatient encounters during the study period. Routine endocrinology or primary care visits were not included, and patient care notes were consulted to ensure that the encounters were related to a DM complication. The change in number of visits was calculated for each patient.
Adherence was defined as patients receiving active medication management, documented treatment regimen adherence, and > 4 annual endocrinology clinic visits. Active medication management was defined as having > 1 dosage or medication change for oral or noninsulin antihyperglycemics, initiation, or adjustment of insulin dosages according to the patient records. Treatment adherence was determined based on medication reconciliation notes and refill request history. Only endocrinology clinic visits at VA outpatient clinics were included.
Study Population
A sample of 166 patients was needed to detect an HbA1c change of 0.8 per power analysis. The normal approximation method using the z statistic was used, with 2-tailed α = 0.05, β = 0.05, E = 0.8, and S = 1.2. We randomly selected 175 patients among all individuals with an active prescription for CGM in 2018 and 2019, who had a diagnosis of T2DM, and were managed by VA endocrinology clinics (including endocrine clinics, diabetes clinics, and patient aligned care team clinics) at 87 VA medical centers. Patients with types of DM other than T2DM were excluded, as well as those with a diagnosed hemoglobinopathy or HbA1c < 10 g/dL. The adherent subgroup included 40 patients of the 175 sample population (Table 1).
Results
Both the total population and the adherent subgroup showed reduction in HbA1c, the primary endpoint. The complete population showed a HbA1c change of –0.3 (95% CI, –0.4 to –0.2), and the adherent subgroup had a change of –1.3 (95% CI, –1.5 to –1.2). The total survey population had a mean change in weight of –1.9 lb (–0.9 kg) (95% CI, –3.7 to –0.1) and the adherent subgroup had an average change of –8.0 lb (–3.6 kg) (95% CI, –12.3 to –3.8). Average systolic BP changes were –0.1 mm Hg (95% CI, –1.6 to 1.5) in the total population and +3.3 mm Hg (95% CI, –0.01 to 6.22) in the adherent subgroup. A decrease in total encounters for DM complications was observed in the population (–0.3 total encounters per patient, 95% CI, –0.5 to –0.2) and the adherent subgroup (–0.6 total encounters per patient, 95% CI, –1.0 to –0.1) (Table 2).
Before the study, 107 (61.1%) patients were taking oral or noninsulin DM medication only, 4 (2.3%) were on insulin only, and 64 (36.6%) were prescribed both insulin and oral/noninsulin antihyperglycemics. Noninsulin and oral antihyperglycemic regimens included combinations of biguanide, dipeptidyl peptidase- 4 inhibitor, sodium-glucose cotransporter-2 inhibitor, sulfonylurea, meglitinide, β-glucosidase inhibitor, glucagon-like peptide-1 (GLP-1) analog, and thiazolidinedione drug classes. Nearly 70% (122) had no reported changes in DM treatment beyond dosage titrations. Among these patients, 18 (10.3%) were on an insulin pump for the duration of the study. Among the 53 (30.3%) patients who had changes in treatment, 31 (17.7%) transitioned from insulin injections to an insulin pump, 13 (7.4%) changed from 1 insulin injection to another (ie, addition of long-acting insulin, transition to u500 insulin, changing from 1 insulin category or brand to another), 8 (4.6%) began an oral/noninsulin antihyperglycemic, 4 (2.3%) began insulin injections, 13 (7.4%) discontinued noninsulin or oral antihyperglycemics, and 2 (1.1%) discontinued insulin during the study period.
Data showed that 113 (64.5%) patients had no changes in antihypertensives. The remaining 62 (35.4%) had the following adjustments: 14 (8%) increased dose of current medication(s), 9 (5.1%) decreased dose of current medication(s), 8 (4.6%) discontinued all antihypertensive medications, 10 (5.7%) switched to a different antihypertensive class, and 16 (9.1%) added additional antihypertensive medication(s) to their existing regimen during the study period.
Patients in the study group used 7 different types of CGM sensors. Chart review revealed that 84 (47.7%) patients used Medtronic devices, with 26 (14.8%) using first-generation Guardian sensors, 50 (28.4%) using Enlite sensors, and 8 (4.5) using Guardian 3 sensors. We found that 81 (46.0%) veterans were prescribed Dexcom devices, with 5 (2.8%) using SEVEN PLUS sensors, 68 (38.6%) using G4-5 sensors, and 8 (4.5%) using G6 sensors. The remaining 10 (5.7%) patients were using Freestyle Libre sensors during the study period.
Discussion
CGM did not correspond with clinically significant reductions in HbA1c. However, veterans with increased health care engagement were likely to achieve clinically significant HbA1c improvements. The veterans in the adherent subgroup had a higher baseline HbA1c, which could be because of a variety of factors mentioned in patient care notes, including insulin resistance, poor dietary habits, and exercise regimen nonadherence. These patients might have had more room to improve their glycemic control without concern of hypoglycemia, and their higher baseline HbA1c could have provided increased motivation for improving their health during the study period.
Adherent patients also had a greater reduction in weight and hospital or clinic visits with CGM compared with the total population. These veterans’ increased involvement in their health care might have led to better dietary and exercise adherence, which would have decreased insulin dosing and contributed to weight loss. Only 1 patient in the adherent subgroup initiated a GLP-1 agonist during the study period, making it unlikely that medication changes had a significant impact on weight loss in the subgroup analysis. This improvement in overall health status might have contributed to the reduction in hospital or clinic visits observed in this population.
Average systolic BP data decreased minimally in the total survey population and increased in the adherent subgroup over the course of the study. These results were determined to be statistically significant. Changes in systolic BP readings were minimal, indicating that it is unlikely that these changes contributed meaningfully to the patients’ overall health status.
Although not related to the study objectives, the adherent population required less antihypertensive adjustments with similar BP control. This could be explained by improved overall health or better adherence and engagement in therapy. The results of this project show that despite limited medication changes, T2DM management improved among adherent patients using CGM. The general study population, which was more likely to have documented nonadherence with treatment or clinic appointments, had minimal benefit. CGM technology in the T2DM veteran population is more likely to have significant clinical benefit in patients who are adherent with their medication regimens and follow-up appointments compared with the larger study population.
The results of this study are in line with previous studies on CGM use in the T2DM patient population. We agree with the previously published research that CGM alone does not have a meaningful impact on HbA1c reduction. Our study population also was older than those in previous studies, adding to the Haak and colleagues conclusion that patients aged < 65 years might have better outcomes with CGM.4
Strengths of this study include specificity to the veteran population using VA resources, as well as including nondiabetes outcomes. This allows for specific application to the veteran population and could provide broader evidence for CGM use. Demonstrated decreases in HbA1c, weight, and clinic visits in the adherent population suggest that providing veterans with CGM therapy with frequent endocrinology follow-up improves health outcomes and could decrease overall health spending.
Limitations
Limitations of this study include retrospective design, a small sample size, and solely focusing on T2DM. As a retrospective study, we cannot rule out the influence of outside factors, such as participation in a non-VA weight loss program. This study lacked the power to assess the impact of the different CGM brands. The study did not include data on severe hypoglycemic or hyperglycemic episodes as veterans might have needed emergent care at non-VA facilities. Future research will evaluate the impact of CGM on symptomatic and severe hypoglycemic episodes and use of insulin vs oral or noninsulin antihyperglycemics and the comparative efficacy of different CGM brands among veterans.
Conclusions
CGM did not correspond with clinically significant reductions in HbA1c. However, veterans with increased health care engagement were likely to achieve clinically significant HbA1c improvements. Adherent patients also had more reduction in weight and hospital or clinic visits with CGM compared with the total population. These veterans’ increased involvement in their health care might have led to better dietary and exercise adherence, which would have decreased insulin dosing and contributed to weight loss.
1. Liu Y, Sayam S, Shao X, et al. Prevalence of and trends in diabetes among veterans, United States, 2005-2014. Prev Chronic Dis. 2017;14:E135. Published 2017 Dec 14. doi:10.5888/pcd14.170230
2. Hackett M. VA pharmacies now carry the Dexcom G6 CGM at no cost for qualifying patients. September 23, 2020. Accessed September 28, 2021. https://www.mobihealthnews.com/news/va-pharmacies-now-carry-dexcom-g6-cgm-no-cost-qualifying-patients
3. Peters AL. The evidence base for continuous glucose monitoring. In: Role of Continuous Glucose Monitoring in Diabetes Treatment. Arlington (VA): American Diabetes Association; August 2018.3-7. doi:10.2337/db20181-3
4. Haak T, Hanaire H, Ajjan R, Hermanns N, Riveline JP, Rayman G. Flash glucose-sensing technology as a replacement for blood glucose monitoring for the management of insulin-treated type 2 diabetes: a multicenter, open-label randomized controlled trial. Diabetes Ther. 2017;8(1):55-73. doi:10.1007/s13300-016-0223-6
5. Yoo HJ, An HG, Park SY, et al. Use of a real time continuous glucose monitoring system as a motivational device for poorly controlled type 2 diabetes. Diabetes Res Clin Pract. 2008;82(1):73-79. doi:10.1016/j.diabres.2008.06.015
6. Taylor PJ, Thompson CH, Brinkworth GD. Effectiveness and acceptability of continuous glucose monitoring for type 2 diabetes management: A narrative review. J Diabetes Investig. 2018;9(4):713-725. doi:10.1111/jdi.12807
1. Liu Y, Sayam S, Shao X, et al. Prevalence of and trends in diabetes among veterans, United States, 2005-2014. Prev Chronic Dis. 2017;14:E135. Published 2017 Dec 14. doi:10.5888/pcd14.170230
2. Hackett M. VA pharmacies now carry the Dexcom G6 CGM at no cost for qualifying patients. September 23, 2020. Accessed September 28, 2021. https://www.mobihealthnews.com/news/va-pharmacies-now-carry-dexcom-g6-cgm-no-cost-qualifying-patients
3. Peters AL. The evidence base for continuous glucose monitoring. In: Role of Continuous Glucose Monitoring in Diabetes Treatment. Arlington (VA): American Diabetes Association; August 2018.3-7. doi:10.2337/db20181-3
4. Haak T, Hanaire H, Ajjan R, Hermanns N, Riveline JP, Rayman G. Flash glucose-sensing technology as a replacement for blood glucose monitoring for the management of insulin-treated type 2 diabetes: a multicenter, open-label randomized controlled trial. Diabetes Ther. 2017;8(1):55-73. doi:10.1007/s13300-016-0223-6
5. Yoo HJ, An HG, Park SY, et al. Use of a real time continuous glucose monitoring system as a motivational device for poorly controlled type 2 diabetes. Diabetes Res Clin Pract. 2008;82(1):73-79. doi:10.1016/j.diabres.2008.06.015
6. Taylor PJ, Thompson CH, Brinkworth GD. Effectiveness and acceptability of continuous glucose monitoring for type 2 diabetes management: A narrative review. J Diabetes Investig. 2018;9(4):713-725. doi:10.1111/jdi.12807