User login
Tofacitinib succeeds as ulcerative colitis induction therapy
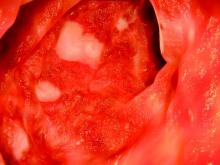
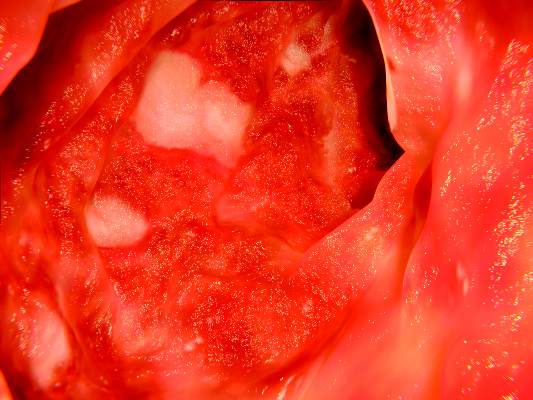
A 10-mg dose of tofacitinib twice daily significantly improved remission, mucosal healing, and clinical response in adults with active ulcerative colitis (UC), based on data from a pair of identical phase III studies including nearly 900 patients. The findings were presented at the European Crohn’s and Colitis Organisation conference in Amsterdam.
“In up to one-third of patients with UC, treatment is not completely successful or complications arise,” study coauthor Dr. Geert D’Haens of the University of Amsterdam said in an interview. The goal of the studies was to evaluate the safety and efficacy of a 10-mg dose of oral tofacitinib twice daily in inducing remission in UC patients, he added. The OCTAVE (Oral Clinical Trials for Tofacitinib in Ulcerative Colitis) Induction 1 study included 476 patients taking tofacitinib and 122 patients taking placebo; the OCTAVE Induction 2 study included 429 patients on tofacitinib and 112 patients on placebo.
Overall, significantly more patients receiving tofacitinib 10 mg twice daily achieved remission, mucosal healing, and clinical response in both studies, compared with the placebo, at 8 weeks. In the OCTAVE Induction 1 and Induction 2 studies, remission at 8 weeks for tofacitinib compared with placebo was 19% vs. 8% and 17% vs. 4%, respectively. Mucosal healing rates in the Induction 1 and 2 studies for tofacitinib compared with placebo were 31% vs. 16% and 28% vs. 12%, respectively, and clinical response rates were 60% vs. 33% and 55% vs. 29%, respectively. Efficacy was similar for patients previously treated with tumor necrosis factor inhibitors and those who were not.
The incidence of adverse events and serious adverse events was not significantly different between treatment and placebo groups in either study. However, tofacitinib treatment was associated with increases in serum lipid (total cholesterol, low-density and high-density lipoprotein), and creatine kinase levels.
“The clinical trial data confirm our blinded observations,” Dr. D’Haens said. “Even when all other drugs have failed, tofacitinib can be effective. Since Janus kinase inhibitors reduce the production of many proinflammatory cytokines, the clinical findings are in line with what we expected. Fortunately, adverse events were limited and allowed prolonged treatment with this agent,” he noted.
Research on tofacitinib and UC is ongoing, said Dr. D’Haens. The two studies reported here, OCTAVE Induction 1 and 2, are part of the global OCTAVE program, he said. Other related studies include a third phase III study, OCTAVE Sustain, and a long-term extension trial called OCTAVE Open. “OCTAVE Sustain is a phase III placebo-controlled study evaluating oral tofacitinib 10 mg and 5 mg b.i.d. as maintenance therapy in adult patients with moderately to severely active UC. Top-line results for this study are anticipated at the end of this year,” he said. “OCTAVE Open is an ongoing open-label extension study designed to assess the safety and tolerability of tofacitinib 10 mg and 5 mg b.i.d. in patients who have completed or who have had treatment failure in OCTAVE Sustain or who were nonresponders upon completing OCTAVE Induction 1 or 2,” he added.
The study was supported in part by Pfizer. Dr. D’Haens disclosed financial relationships with multiple companies, including Pfizer.
A 10-mg dose of tofacitinib twice daily significantly improved remission, mucosal healing, and clinical response in adults with active ulcerative colitis (UC), based on data from a pair of identical phase III studies including nearly 900 patients. The findings were presented at the European Crohn’s and Colitis Organisation conference in Amsterdam.
“In up to one-third of patients with UC, treatment is not completely successful or complications arise,” study coauthor Dr. Geert D’Haens of the University of Amsterdam said in an interview. The goal of the studies was to evaluate the safety and efficacy of a 10-mg dose of oral tofacitinib twice daily in inducing remission in UC patients, he added. The OCTAVE (Oral Clinical Trials for Tofacitinib in Ulcerative Colitis) Induction 1 study included 476 patients taking tofacitinib and 122 patients taking placebo; the OCTAVE Induction 2 study included 429 patients on tofacitinib and 112 patients on placebo.
Overall, significantly more patients receiving tofacitinib 10 mg twice daily achieved remission, mucosal healing, and clinical response in both studies, compared with the placebo, at 8 weeks. In the OCTAVE Induction 1 and Induction 2 studies, remission at 8 weeks for tofacitinib compared with placebo was 19% vs. 8% and 17% vs. 4%, respectively. Mucosal healing rates in the Induction 1 and 2 studies for tofacitinib compared with placebo were 31% vs. 16% and 28% vs. 12%, respectively, and clinical response rates were 60% vs. 33% and 55% vs. 29%, respectively. Efficacy was similar for patients previously treated with tumor necrosis factor inhibitors and those who were not.
The incidence of adverse events and serious adverse events was not significantly different between treatment and placebo groups in either study. However, tofacitinib treatment was associated with increases in serum lipid (total cholesterol, low-density and high-density lipoprotein), and creatine kinase levels.
“The clinical trial data confirm our blinded observations,” Dr. D’Haens said. “Even when all other drugs have failed, tofacitinib can be effective. Since Janus kinase inhibitors reduce the production of many proinflammatory cytokines, the clinical findings are in line with what we expected. Fortunately, adverse events were limited and allowed prolonged treatment with this agent,” he noted.
Research on tofacitinib and UC is ongoing, said Dr. D’Haens. The two studies reported here, OCTAVE Induction 1 and 2, are part of the global OCTAVE program, he said. Other related studies include a third phase III study, OCTAVE Sustain, and a long-term extension trial called OCTAVE Open. “OCTAVE Sustain is a phase III placebo-controlled study evaluating oral tofacitinib 10 mg and 5 mg b.i.d. as maintenance therapy in adult patients with moderately to severely active UC. Top-line results for this study are anticipated at the end of this year,” he said. “OCTAVE Open is an ongoing open-label extension study designed to assess the safety and tolerability of tofacitinib 10 mg and 5 mg b.i.d. in patients who have completed or who have had treatment failure in OCTAVE Sustain or who were nonresponders upon completing OCTAVE Induction 1 or 2,” he added.
The study was supported in part by Pfizer. Dr. D’Haens disclosed financial relationships with multiple companies, including Pfizer.
A 10-mg dose of tofacitinib twice daily significantly improved remission, mucosal healing, and clinical response in adults with active ulcerative colitis (UC), based on data from a pair of identical phase III studies including nearly 900 patients. The findings were presented at the European Crohn’s and Colitis Organisation conference in Amsterdam.
“In up to one-third of patients with UC, treatment is not completely successful or complications arise,” study coauthor Dr. Geert D’Haens of the University of Amsterdam said in an interview. The goal of the studies was to evaluate the safety and efficacy of a 10-mg dose of oral tofacitinib twice daily in inducing remission in UC patients, he added. The OCTAVE (Oral Clinical Trials for Tofacitinib in Ulcerative Colitis) Induction 1 study included 476 patients taking tofacitinib and 122 patients taking placebo; the OCTAVE Induction 2 study included 429 patients on tofacitinib and 112 patients on placebo.
Overall, significantly more patients receiving tofacitinib 10 mg twice daily achieved remission, mucosal healing, and clinical response in both studies, compared with the placebo, at 8 weeks. In the OCTAVE Induction 1 and Induction 2 studies, remission at 8 weeks for tofacitinib compared with placebo was 19% vs. 8% and 17% vs. 4%, respectively. Mucosal healing rates in the Induction 1 and 2 studies for tofacitinib compared with placebo were 31% vs. 16% and 28% vs. 12%, respectively, and clinical response rates were 60% vs. 33% and 55% vs. 29%, respectively. Efficacy was similar for patients previously treated with tumor necrosis factor inhibitors and those who were not.
The incidence of adverse events and serious adverse events was not significantly different between treatment and placebo groups in either study. However, tofacitinib treatment was associated with increases in serum lipid (total cholesterol, low-density and high-density lipoprotein), and creatine kinase levels.
“The clinical trial data confirm our blinded observations,” Dr. D’Haens said. “Even when all other drugs have failed, tofacitinib can be effective. Since Janus kinase inhibitors reduce the production of many proinflammatory cytokines, the clinical findings are in line with what we expected. Fortunately, adverse events were limited and allowed prolonged treatment with this agent,” he noted.
Research on tofacitinib and UC is ongoing, said Dr. D’Haens. The two studies reported here, OCTAVE Induction 1 and 2, are part of the global OCTAVE program, he said. Other related studies include a third phase III study, OCTAVE Sustain, and a long-term extension trial called OCTAVE Open. “OCTAVE Sustain is a phase III placebo-controlled study evaluating oral tofacitinib 10 mg and 5 mg b.i.d. as maintenance therapy in adult patients with moderately to severely active UC. Top-line results for this study are anticipated at the end of this year,” he said. “OCTAVE Open is an ongoing open-label extension study designed to assess the safety and tolerability of tofacitinib 10 mg and 5 mg b.i.d. in patients who have completed or who have had treatment failure in OCTAVE Sustain or who were nonresponders upon completing OCTAVE Induction 1 or 2,” he added.
The study was supported in part by Pfizer. Dr. D’Haens disclosed financial relationships with multiple companies, including Pfizer.
EXPERT ANALYSIS FROM ECCO
Key clinical point: Oral tofacitinib reduced the symptoms of moderate to severe ulcerative colitis and induced remission and healing of the diseased colonic mucosa in patients previously treated with tumor necrosis factor inhibitors and those who were not.
Major finding: In the OCTAVE Induction 1 and Induction 2 studies, remission at 8 weeks for tofacitinib patients compared with placebo patients was 19% vs. 8% and 17% vs. 4%, respectively.
Data source: A pair of identical phase III studies including nearly 900 patients.
Disclosures: The study was supported in part by Pfizer. Dr. Geert D’Haens disclosed financial relationships with multiple companies, including Pfizer.
Ustekinumab improves outlook for complex Crohn’s disease
Ustekinumab induced a clinical response and remission in adults with moderate to severe Crohn’s disease who had failed or failed to tolerate other anti–tumor necrosis factor therapies in a phase III induction study of 741 patients (UNITI-1).
The findings were presented at the European Crohn’s and Colitis Organization (ECCO) conference in Amsterdam.
“There is great need for improved therapy for Crohn’s disease, since many patients are failing conventional therapies and also TNF [tumor necrosis factor] antagonists,” lead author Dr. Paul Rutgeerts said in an interview. “Blockade of IL-12/23 is a new mode of action in IBD [inflammatory bowel disease], although this therapy has already been proven effective in the treatment of psoriasis and psoriatic arthropathy,” he said. Data from prior large-scale studies show that ustekinumab is effective in patients with refractory Crohn’s disease, noted Dr. Rutgeerts of University Hospital Gasthuisberg, in Leuven, the Netherlands.
Ustekinumab is approved for treating moderate to severe plaque psoriasis and psoriatic arthritis, and was tested in a phase IIb study for Crohn’s disease in which intravenous ustekinumab induction was followed by surveillance colonoscopy maintenance. This study reviewed the safety and efficacy of intravenous ustekinumab in the same patient population.
The patients were randomized to a 130-mg dose of ustekinumab, weight-based dosing of ustekinumab at 6 mg/kg, or an intravenous placebo. The patients had an average disease duration of 10 years, a baseline median Crohn’s Disease Activity Index (CDAI) score of 317, and a baseline C-reactive protein level of 9.9 mg/L.
After 6 weeks, 34% of patients treated with 6 mg/kg or 130 mg of ustekinumab showed a clinical response, vs. 22% of placebo patients. Clinical response was defined as a reduction from baseline of the CDAI score of at least 100 points. At 8 weeks, patients entered an ongoing maintenance study or were followed up to 20 weeks. Clinical remission (defined as a CDAI score of less than 150 points) at 8 weeks was significantly higher in the 6-mg/kg group (21%) and 130-mg group (16%), compared with the placebo group (7%).
In addition, patients in both ustekinumab groups showed significant improvements in C-reactive protein (CRP), fecal lactoferrin, and calprotectin, and in scores on the Inflammatory Bowel Disease Questionnaire, compared with placebo patients.
The incidence of adverse events, serious adverse events, and infections was not significantly different among the treatment and placebo groups, and no deaths, malignancies, or major adverse events were reported among ustekinumab patients through the 20-week follow-up period.
“The clinical and biologic efficacy of the drug is robust both in patients who have failed conventional drugs and patients who have already failed one, two, or three anti-TNF biologicals,” Dr. Rutgeerts noted. “The approval of Stelara for Crohn’s disease would represent a new treatment option for the more than 5 million people worldwide, including nearly 700,000 Americans, who are living with an inflammatory bowel disease,” he said.
Data from the ongoing IM-UNITI study will demonstrate the long-term efficacy of ustekinumab for Crohn’s patients, but another key step for research would be a head-to-head comparison between ustekinumab and an anti-TNF antibody, Dr. Rutgeerts added.
Dr. Rutgeerts disclosed receiving personal fees from multiple companies including Johnson & Johnson, Amgen, AstraZeneca, Medimmune, Merck, and UCB. The study was supported in part by Janssen.
Ustekinumab induced a clinical response and remission in adults with moderate to severe Crohn’s disease who had failed or failed to tolerate other anti–tumor necrosis factor therapies in a phase III induction study of 741 patients (UNITI-1).
The findings were presented at the European Crohn’s and Colitis Organization (ECCO) conference in Amsterdam.
“There is great need for improved therapy for Crohn’s disease, since many patients are failing conventional therapies and also TNF [tumor necrosis factor] antagonists,” lead author Dr. Paul Rutgeerts said in an interview. “Blockade of IL-12/23 is a new mode of action in IBD [inflammatory bowel disease], although this therapy has already been proven effective in the treatment of psoriasis and psoriatic arthropathy,” he said. Data from prior large-scale studies show that ustekinumab is effective in patients with refractory Crohn’s disease, noted Dr. Rutgeerts of University Hospital Gasthuisberg, in Leuven, the Netherlands.
Ustekinumab is approved for treating moderate to severe plaque psoriasis and psoriatic arthritis, and was tested in a phase IIb study for Crohn’s disease in which intravenous ustekinumab induction was followed by surveillance colonoscopy maintenance. This study reviewed the safety and efficacy of intravenous ustekinumab in the same patient population.
The patients were randomized to a 130-mg dose of ustekinumab, weight-based dosing of ustekinumab at 6 mg/kg, or an intravenous placebo. The patients had an average disease duration of 10 years, a baseline median Crohn’s Disease Activity Index (CDAI) score of 317, and a baseline C-reactive protein level of 9.9 mg/L.
After 6 weeks, 34% of patients treated with 6 mg/kg or 130 mg of ustekinumab showed a clinical response, vs. 22% of placebo patients. Clinical response was defined as a reduction from baseline of the CDAI score of at least 100 points. At 8 weeks, patients entered an ongoing maintenance study or were followed up to 20 weeks. Clinical remission (defined as a CDAI score of less than 150 points) at 8 weeks was significantly higher in the 6-mg/kg group (21%) and 130-mg group (16%), compared with the placebo group (7%).
In addition, patients in both ustekinumab groups showed significant improvements in C-reactive protein (CRP), fecal lactoferrin, and calprotectin, and in scores on the Inflammatory Bowel Disease Questionnaire, compared with placebo patients.
The incidence of adverse events, serious adverse events, and infections was not significantly different among the treatment and placebo groups, and no deaths, malignancies, or major adverse events were reported among ustekinumab patients through the 20-week follow-up period.
“The clinical and biologic efficacy of the drug is robust both in patients who have failed conventional drugs and patients who have already failed one, two, or three anti-TNF biologicals,” Dr. Rutgeerts noted. “The approval of Stelara for Crohn’s disease would represent a new treatment option for the more than 5 million people worldwide, including nearly 700,000 Americans, who are living with an inflammatory bowel disease,” he said.
Data from the ongoing IM-UNITI study will demonstrate the long-term efficacy of ustekinumab for Crohn’s patients, but another key step for research would be a head-to-head comparison between ustekinumab and an anti-TNF antibody, Dr. Rutgeerts added.
Dr. Rutgeerts disclosed receiving personal fees from multiple companies including Johnson & Johnson, Amgen, AstraZeneca, Medimmune, Merck, and UCB. The study was supported in part by Janssen.
Ustekinumab induced a clinical response and remission in adults with moderate to severe Crohn’s disease who had failed or failed to tolerate other anti–tumor necrosis factor therapies in a phase III induction study of 741 patients (UNITI-1).
The findings were presented at the European Crohn’s and Colitis Organization (ECCO) conference in Amsterdam.
“There is great need for improved therapy for Crohn’s disease, since many patients are failing conventional therapies and also TNF [tumor necrosis factor] antagonists,” lead author Dr. Paul Rutgeerts said in an interview. “Blockade of IL-12/23 is a new mode of action in IBD [inflammatory bowel disease], although this therapy has already been proven effective in the treatment of psoriasis and psoriatic arthropathy,” he said. Data from prior large-scale studies show that ustekinumab is effective in patients with refractory Crohn’s disease, noted Dr. Rutgeerts of University Hospital Gasthuisberg, in Leuven, the Netherlands.
Ustekinumab is approved for treating moderate to severe plaque psoriasis and psoriatic arthritis, and was tested in a phase IIb study for Crohn’s disease in which intravenous ustekinumab induction was followed by surveillance colonoscopy maintenance. This study reviewed the safety and efficacy of intravenous ustekinumab in the same patient population.
The patients were randomized to a 130-mg dose of ustekinumab, weight-based dosing of ustekinumab at 6 mg/kg, or an intravenous placebo. The patients had an average disease duration of 10 years, a baseline median Crohn’s Disease Activity Index (CDAI) score of 317, and a baseline C-reactive protein level of 9.9 mg/L.
After 6 weeks, 34% of patients treated with 6 mg/kg or 130 mg of ustekinumab showed a clinical response, vs. 22% of placebo patients. Clinical response was defined as a reduction from baseline of the CDAI score of at least 100 points. At 8 weeks, patients entered an ongoing maintenance study or were followed up to 20 weeks. Clinical remission (defined as a CDAI score of less than 150 points) at 8 weeks was significantly higher in the 6-mg/kg group (21%) and 130-mg group (16%), compared with the placebo group (7%).
In addition, patients in both ustekinumab groups showed significant improvements in C-reactive protein (CRP), fecal lactoferrin, and calprotectin, and in scores on the Inflammatory Bowel Disease Questionnaire, compared with placebo patients.
The incidence of adverse events, serious adverse events, and infections was not significantly different among the treatment and placebo groups, and no deaths, malignancies, or major adverse events were reported among ustekinumab patients through the 20-week follow-up period.
“The clinical and biologic efficacy of the drug is robust both in patients who have failed conventional drugs and patients who have already failed one, two, or three anti-TNF biologicals,” Dr. Rutgeerts noted. “The approval of Stelara for Crohn’s disease would represent a new treatment option for the more than 5 million people worldwide, including nearly 700,000 Americans, who are living with an inflammatory bowel disease,” he said.
Data from the ongoing IM-UNITI study will demonstrate the long-term efficacy of ustekinumab for Crohn’s patients, but another key step for research would be a head-to-head comparison between ustekinumab and an anti-TNF antibody, Dr. Rutgeerts added.
Dr. Rutgeerts disclosed receiving personal fees from multiple companies including Johnson & Johnson, Amgen, AstraZeneca, Medimmune, Merck, and UCB. The study was supported in part by Janssen.
EXPERT ANALYSIS FROM ECCO
Key clinical point: Ustekinumab is effective both as first-line biologic treatment in patients who have failed conventional therapy and in those who have failed previous anti-TNF therapies.
Major finding: After 6 weeks, 34% of patients treated with approximately 6 mg/kg or 130 mg of ustekinumab showed a clinical response vs. 22% of placebo patients.
Data source: A randomized, phase III induction study of 741 adults with moderate to severe Crohn’s disease.
Disclosures: Dr. Rutgeerts disclosed receiving personal fees from multiple companies including Johnson & Johnson (Janssen), Amgen, AstraZeneca, Medimmune, Merck, and UCB. The study was supported in part by Janssen.
Fatty liver risk rises in years after transplant
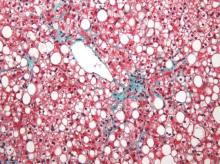
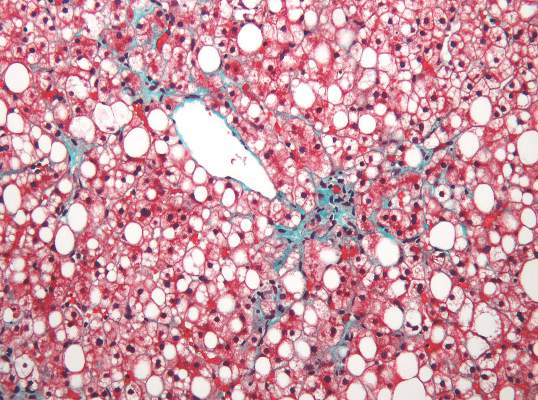
Steatosis may be present in at least half of liver transplant recipients, and the prevalence increases significantly over time, according to data from a retrospective study of 548 adult patients.
Although steatosis is common after transplantation, the prevalence, risk factors, and impact on patient survival has not been well studied, wrote Dr. Irena Hejlova of the Institute for Clinical and Experimental Medicine in Prague, Czech Republic, and her colleagues.
“Our study was the first to document that the prevalence of steatosis in LT [liver transplant] recipients may be far higher than previously reported,” they said.
The researchers reviewed liver biopsies and patient survival data and found steatosis in 309 (56%) of the patients, including 93 (17%) with significant steatosis (defined as greater than 33%). Pretransplant factors associated with significant steatosis included cirrhosis caused by alcohol consumption as well as a high body mass index. Post-transplant risk factors associated with increased risk of significant steatosis included increased body mass index, increased serum triglycerides, alcohol consumption, and type 2 diabetes. However, “Although patients transplanted for alcoholic cirrhosis are at an increased risk, the vast majority of post-transplant steatosis is nonalcohol-related,” the researchers noted.
The overall prevalence of steatosis increased from 30% at 1 year after transplant to 48% at 10 years after transplant. Post-transplant steatosis was not associated with worse patient survival in the short term, but the long-term survival of patients with significant steatosis tended to be worse.
Read the full study here (Liver Transpl. 2016 Apr 5. doi: 10.1002/lt.24393).
Steatosis may be present in at least half of liver transplant recipients, and the prevalence increases significantly over time, according to data from a retrospective study of 548 adult patients.
Although steatosis is common after transplantation, the prevalence, risk factors, and impact on patient survival has not been well studied, wrote Dr. Irena Hejlova of the Institute for Clinical and Experimental Medicine in Prague, Czech Republic, and her colleagues.
“Our study was the first to document that the prevalence of steatosis in LT [liver transplant] recipients may be far higher than previously reported,” they said.
The researchers reviewed liver biopsies and patient survival data and found steatosis in 309 (56%) of the patients, including 93 (17%) with significant steatosis (defined as greater than 33%). Pretransplant factors associated with significant steatosis included cirrhosis caused by alcohol consumption as well as a high body mass index. Post-transplant risk factors associated with increased risk of significant steatosis included increased body mass index, increased serum triglycerides, alcohol consumption, and type 2 diabetes. However, “Although patients transplanted for alcoholic cirrhosis are at an increased risk, the vast majority of post-transplant steatosis is nonalcohol-related,” the researchers noted.
The overall prevalence of steatosis increased from 30% at 1 year after transplant to 48% at 10 years after transplant. Post-transplant steatosis was not associated with worse patient survival in the short term, but the long-term survival of patients with significant steatosis tended to be worse.
Read the full study here (Liver Transpl. 2016 Apr 5. doi: 10.1002/lt.24393).
Steatosis may be present in at least half of liver transplant recipients, and the prevalence increases significantly over time, according to data from a retrospective study of 548 adult patients.
Although steatosis is common after transplantation, the prevalence, risk factors, and impact on patient survival has not been well studied, wrote Dr. Irena Hejlova of the Institute for Clinical and Experimental Medicine in Prague, Czech Republic, and her colleagues.
“Our study was the first to document that the prevalence of steatosis in LT [liver transplant] recipients may be far higher than previously reported,” they said.
The researchers reviewed liver biopsies and patient survival data and found steatosis in 309 (56%) of the patients, including 93 (17%) with significant steatosis (defined as greater than 33%). Pretransplant factors associated with significant steatosis included cirrhosis caused by alcohol consumption as well as a high body mass index. Post-transplant risk factors associated with increased risk of significant steatosis included increased body mass index, increased serum triglycerides, alcohol consumption, and type 2 diabetes. However, “Although patients transplanted for alcoholic cirrhosis are at an increased risk, the vast majority of post-transplant steatosis is nonalcohol-related,” the researchers noted.
The overall prevalence of steatosis increased from 30% at 1 year after transplant to 48% at 10 years after transplant. Post-transplant steatosis was not associated with worse patient survival in the short term, but the long-term survival of patients with significant steatosis tended to be worse.
Read the full study here (Liver Transpl. 2016 Apr 5. doi: 10.1002/lt.24393).
FROM LIVER TRANSPLANTATION
Experts offer insight into changing payment models for new technologies
BOSTON – Many developers of innovative medical technologies don’t realize that insurers are going to want to see evidence that a new technology improves health outcomes before they will consider paying for it, according to Dr. Louis Jacques, senior vice president and chief clinical officer for ADVI, a health care advisory services firm in Washington.
The Centers for Medicare & Medicaid Services generally seeks “adequate evidence to conclude that the item or service improves clinically meaningful health outcomes for the Medicare population” when determining Medicare coverage, Dr. Jacques said in a presentation at the 2016 AGA Tech Summit, which is sponsored by the AGA Center for GI Innovation and Technology.
Medicare is less likely to pay if there is something controversial about the available evidence, he said in an interview. “The controversy is usually about inadequate evidence or lack of applicability to the affected Medicare beneficiary population,” Dr. Jacques said, noting that the typical Medicare patient, a 74-year-old woman with comorbid conditions taking multiple medications, often is not the type of person included in clinical trials that would generate such evidence.
Medicare is looking for “adequate evidence that a treatment strategy using the new therapeutic technology, compared to alternatives, leads to improved clinically meaningful health outcomes in Medicare beneficiaries,” he said.
Similarly, new diagnostic technologies should provide the physician with additional information that can alter treatment recommendations, resulting in therapeutic changes that improve outcomes.
Dr. Jacques noted that over the next several years, the health care system will move away from fee for service and begin more and more to pay for value, with physicians becoming more accountable for outcomes and cost.
He advised those developing a new device or treatment to think about how they would justify a particular treatment approach to their colleagues. They need to ask themselves: “What is the value proposition that supports what they are recommending? Are they choosing it because it has better outcomes, is less likely to have an adverse event, is more efficiently provided, or is it something that is less expensive?” he said.
Two major health reform laws will change how insurers pay for new medical devices, though the impact of these changes remains to be seen. According to Harry Glorikian, a health care consultant based in Lexington, Mass., the Affordable Care Act and the Medicaid and CHIP Reauthorization Act (MACRA) put many items into play, most notably fundamental changes in Medicare/Medicaid and the shift from fee-for-service to value-based payment.
“It’s a crazy time because of the need for virtually all hospital-based organizations to operate under both fee-for-service and non–fee-for-service payment arrangements right now,” he said in an interview.
Along with those two reform laws, the Health & Human Services department has set a schedule for transitioning to paying for the value – rather than the volume – of care. The initial goal – 30% of Medicare payments based on value by the end of 2016 – was reached in early March.
“HHS also set a goal of tying 85% of all traditional Medicare payments to quality or value by 2016 and 90% by 2018 through programs such as the Hospital Value-Based Purchasing and the Hospital Readmissions Reduction programs. This will be huge, and its impact will be tremendous,” he added.
One tool physicians can leverage to show value in this new payment landscape is the AGA Digestive Health Recognition Program, a quality improvement program and clinical data registry that allows clinicians to demonstrate quality of care for several disease states (colorectal cancer, hepatitis C virus, and irritable bowel disease) (www.gastro.org/DHRP).
Over the next 5-10 years, watch for changes in value-based payment models and a long-term move to a single-payer system, said Mr. Glorikian. In addition, expect changes in the workforce in terms of who delivers care and how.
Finally, physicians will need to use big data to manage patients, prescribe therapy, and integrate all aspects of medical practice, he said.
Dr. Jacques disclosed that he has financial relationships with Exact Sciences and Medtronic.
BOSTON – Many developers of innovative medical technologies don’t realize that insurers are going to want to see evidence that a new technology improves health outcomes before they will consider paying for it, according to Dr. Louis Jacques, senior vice president and chief clinical officer for ADVI, a health care advisory services firm in Washington.
The Centers for Medicare & Medicaid Services generally seeks “adequate evidence to conclude that the item or service improves clinically meaningful health outcomes for the Medicare population” when determining Medicare coverage, Dr. Jacques said in a presentation at the 2016 AGA Tech Summit, which is sponsored by the AGA Center for GI Innovation and Technology.
Medicare is less likely to pay if there is something controversial about the available evidence, he said in an interview. “The controversy is usually about inadequate evidence or lack of applicability to the affected Medicare beneficiary population,” Dr. Jacques said, noting that the typical Medicare patient, a 74-year-old woman with comorbid conditions taking multiple medications, often is not the type of person included in clinical trials that would generate such evidence.
Medicare is looking for “adequate evidence that a treatment strategy using the new therapeutic technology, compared to alternatives, leads to improved clinically meaningful health outcomes in Medicare beneficiaries,” he said.
Similarly, new diagnostic technologies should provide the physician with additional information that can alter treatment recommendations, resulting in therapeutic changes that improve outcomes.
Dr. Jacques noted that over the next several years, the health care system will move away from fee for service and begin more and more to pay for value, with physicians becoming more accountable for outcomes and cost.
He advised those developing a new device or treatment to think about how they would justify a particular treatment approach to their colleagues. They need to ask themselves: “What is the value proposition that supports what they are recommending? Are they choosing it because it has better outcomes, is less likely to have an adverse event, is more efficiently provided, or is it something that is less expensive?” he said.
Two major health reform laws will change how insurers pay for new medical devices, though the impact of these changes remains to be seen. According to Harry Glorikian, a health care consultant based in Lexington, Mass., the Affordable Care Act and the Medicaid and CHIP Reauthorization Act (MACRA) put many items into play, most notably fundamental changes in Medicare/Medicaid and the shift from fee-for-service to value-based payment.
“It’s a crazy time because of the need for virtually all hospital-based organizations to operate under both fee-for-service and non–fee-for-service payment arrangements right now,” he said in an interview.
Along with those two reform laws, the Health & Human Services department has set a schedule for transitioning to paying for the value – rather than the volume – of care. The initial goal – 30% of Medicare payments based on value by the end of 2016 – was reached in early March.
“HHS also set a goal of tying 85% of all traditional Medicare payments to quality or value by 2016 and 90% by 2018 through programs such as the Hospital Value-Based Purchasing and the Hospital Readmissions Reduction programs. This will be huge, and its impact will be tremendous,” he added.
One tool physicians can leverage to show value in this new payment landscape is the AGA Digestive Health Recognition Program, a quality improvement program and clinical data registry that allows clinicians to demonstrate quality of care for several disease states (colorectal cancer, hepatitis C virus, and irritable bowel disease) (www.gastro.org/DHRP).
Over the next 5-10 years, watch for changes in value-based payment models and a long-term move to a single-payer system, said Mr. Glorikian. In addition, expect changes in the workforce in terms of who delivers care and how.
Finally, physicians will need to use big data to manage patients, prescribe therapy, and integrate all aspects of medical practice, he said.
Dr. Jacques disclosed that he has financial relationships with Exact Sciences and Medtronic.
BOSTON – Many developers of innovative medical technologies don’t realize that insurers are going to want to see evidence that a new technology improves health outcomes before they will consider paying for it, according to Dr. Louis Jacques, senior vice president and chief clinical officer for ADVI, a health care advisory services firm in Washington.
The Centers for Medicare & Medicaid Services generally seeks “adequate evidence to conclude that the item or service improves clinically meaningful health outcomes for the Medicare population” when determining Medicare coverage, Dr. Jacques said in a presentation at the 2016 AGA Tech Summit, which is sponsored by the AGA Center for GI Innovation and Technology.
Medicare is less likely to pay if there is something controversial about the available evidence, he said in an interview. “The controversy is usually about inadequate evidence or lack of applicability to the affected Medicare beneficiary population,” Dr. Jacques said, noting that the typical Medicare patient, a 74-year-old woman with comorbid conditions taking multiple medications, often is not the type of person included in clinical trials that would generate such evidence.
Medicare is looking for “adequate evidence that a treatment strategy using the new therapeutic technology, compared to alternatives, leads to improved clinically meaningful health outcomes in Medicare beneficiaries,” he said.
Similarly, new diagnostic technologies should provide the physician with additional information that can alter treatment recommendations, resulting in therapeutic changes that improve outcomes.
Dr. Jacques noted that over the next several years, the health care system will move away from fee for service and begin more and more to pay for value, with physicians becoming more accountable for outcomes and cost.
He advised those developing a new device or treatment to think about how they would justify a particular treatment approach to their colleagues. They need to ask themselves: “What is the value proposition that supports what they are recommending? Are they choosing it because it has better outcomes, is less likely to have an adverse event, is more efficiently provided, or is it something that is less expensive?” he said.
Two major health reform laws will change how insurers pay for new medical devices, though the impact of these changes remains to be seen. According to Harry Glorikian, a health care consultant based in Lexington, Mass., the Affordable Care Act and the Medicaid and CHIP Reauthorization Act (MACRA) put many items into play, most notably fundamental changes in Medicare/Medicaid and the shift from fee-for-service to value-based payment.
“It’s a crazy time because of the need for virtually all hospital-based organizations to operate under both fee-for-service and non–fee-for-service payment arrangements right now,” he said in an interview.
Along with those two reform laws, the Health & Human Services department has set a schedule for transitioning to paying for the value – rather than the volume – of care. The initial goal – 30% of Medicare payments based on value by the end of 2016 – was reached in early March.
“HHS also set a goal of tying 85% of all traditional Medicare payments to quality or value by 2016 and 90% by 2018 through programs such as the Hospital Value-Based Purchasing and the Hospital Readmissions Reduction programs. This will be huge, and its impact will be tremendous,” he added.
One tool physicians can leverage to show value in this new payment landscape is the AGA Digestive Health Recognition Program, a quality improvement program and clinical data registry that allows clinicians to demonstrate quality of care for several disease states (colorectal cancer, hepatitis C virus, and irritable bowel disease) (www.gastro.org/DHRP).
Over the next 5-10 years, watch for changes in value-based payment models and a long-term move to a single-payer system, said Mr. Glorikian. In addition, expect changes in the workforce in terms of who delivers care and how.
Finally, physicians will need to use big data to manage patients, prescribe therapy, and integrate all aspects of medical practice, he said.
Dr. Jacques disclosed that he has financial relationships with Exact Sciences and Medtronic.
EXPERT ANALYSIS FROM THE 2016 AGA TECH SUMMIT
April 2016: Click for Credit
Here are 4 articles in the April issue of Clinician Reviews (individual articles are valid for one year from date of publication—expiration dates below):
1. Later Menopause Lowers Risk for Later Depression
To take the posttest, go to: http://bit.ly/1U7I7f3
Expires January 6, 2017
VITALS
Key clinical point: Later menopause, with its longer estrogen exposure, appears tied to a lower risk of postmenopausal depression.
Major finding: The risk of depression decreased by 2% for each 2 premenopausal years after age 40.
Data source: The meta-analysis comprised 14 studies with more than 67,700 women.
Disclosures: Neither Dr. Georgakis nor any of the coauthors declared any financial conflicts.
2. Preschool ASD Prevalence Estimates Lower Than Grade School Estimates
To take the posttest, go to: http://bit.ly/24Mec0X
Expires January 5, 2017
VITALS
Key clinical point: The prevalence of autism spectrum disorders among 4-year-olds is about 30% lower than among 8-year-olds.
Major finding: Prevalence of ASD among 4-year-olds was 13/1,000 children across five U.S. states.
Data source: A comparison of health and medical records for nationally representative cohorts involving 58,467 4-year-olds and 56,727 8-year-olds in five U.S. states in 2010.
Disclosures: The Centers for Disease Control and Prevention funded the research. Dr. Christensen and her associates reported no disclosures.
3. Long-term PPI Use Linked to Increased Risk for Dementia
To take the posttest, go to: http://bit.ly/1nrCdsb
Expires February 24, 2017
VITALS
Key clinical point: Proton pump inhibitors may add to the risk of dementia in older adults.
Major finding: The risk of incident dementia was 44% higher in adults who used PPIs long term, compared with those who did not.
Data source: The prospective cohort study included 73,679 adults aged 75 years and older.
Disclosures: The researchers had no financial conflicts to disclose.
4. Elevated Cardiovascular Risks Linked to Hidradenitis Suppurativa
To take the posttest, go to: http://bit.ly/1nrEFz3
Expires February 17, 2017
VITALS
Key clinical point: Hidradenitis suppurativa is associated with a significantly increased risk of adverse cardiovascular events and all-cause mortality.
Major finding: Individuals with hidradenitis suppurativa had a 57% greater risk of myocardial infarction and 33% greater risk of ischemic stroke, compared with the general population.
Data source: A population-based cohort study in 5,964 patients with hidradenitis suppurativa.
Disclosures: No conflicts of interest were declared.
Here are 4 articles in the April issue of Clinician Reviews (individual articles are valid for one year from date of publication—expiration dates below):
1. Later Menopause Lowers Risk for Later Depression
To take the posttest, go to: http://bit.ly/1U7I7f3
Expires January 6, 2017
VITALS
Key clinical point: Later menopause, with its longer estrogen exposure, appears tied to a lower risk of postmenopausal depression.
Major finding: The risk of depression decreased by 2% for each 2 premenopausal years after age 40.
Data source: The meta-analysis comprised 14 studies with more than 67,700 women.
Disclosures: Neither Dr. Georgakis nor any of the coauthors declared any financial conflicts.
2. Preschool ASD Prevalence Estimates Lower Than Grade School Estimates
To take the posttest, go to: http://bit.ly/24Mec0X
Expires January 5, 2017
VITALS
Key clinical point: The prevalence of autism spectrum disorders among 4-year-olds is about 30% lower than among 8-year-olds.
Major finding: Prevalence of ASD among 4-year-olds was 13/1,000 children across five U.S. states.
Data source: A comparison of health and medical records for nationally representative cohorts involving 58,467 4-year-olds and 56,727 8-year-olds in five U.S. states in 2010.
Disclosures: The Centers for Disease Control and Prevention funded the research. Dr. Christensen and her associates reported no disclosures.
3. Long-term PPI Use Linked to Increased Risk for Dementia
To take the posttest, go to: http://bit.ly/1nrCdsb
Expires February 24, 2017
VITALS
Key clinical point: Proton pump inhibitors may add to the risk of dementia in older adults.
Major finding: The risk of incident dementia was 44% higher in adults who used PPIs long term, compared with those who did not.
Data source: The prospective cohort study included 73,679 adults aged 75 years and older.
Disclosures: The researchers had no financial conflicts to disclose.
4. Elevated Cardiovascular Risks Linked to Hidradenitis Suppurativa
To take the posttest, go to: http://bit.ly/1nrEFz3
Expires February 17, 2017
VITALS
Key clinical point: Hidradenitis suppurativa is associated with a significantly increased risk of adverse cardiovascular events and all-cause mortality.
Major finding: Individuals with hidradenitis suppurativa had a 57% greater risk of myocardial infarction and 33% greater risk of ischemic stroke, compared with the general population.
Data source: A population-based cohort study in 5,964 patients with hidradenitis suppurativa.
Disclosures: No conflicts of interest were declared.
Here are 4 articles in the April issue of Clinician Reviews (individual articles are valid for one year from date of publication—expiration dates below):
1. Later Menopause Lowers Risk for Later Depression
To take the posttest, go to: http://bit.ly/1U7I7f3
Expires January 6, 2017
VITALS
Key clinical point: Later menopause, with its longer estrogen exposure, appears tied to a lower risk of postmenopausal depression.
Major finding: The risk of depression decreased by 2% for each 2 premenopausal years after age 40.
Data source: The meta-analysis comprised 14 studies with more than 67,700 women.
Disclosures: Neither Dr. Georgakis nor any of the coauthors declared any financial conflicts.
2. Preschool ASD Prevalence Estimates Lower Than Grade School Estimates
To take the posttest, go to: http://bit.ly/24Mec0X
Expires January 5, 2017
VITALS
Key clinical point: The prevalence of autism spectrum disorders among 4-year-olds is about 30% lower than among 8-year-olds.
Major finding: Prevalence of ASD among 4-year-olds was 13/1,000 children across five U.S. states.
Data source: A comparison of health and medical records for nationally representative cohorts involving 58,467 4-year-olds and 56,727 8-year-olds in five U.S. states in 2010.
Disclosures: The Centers for Disease Control and Prevention funded the research. Dr. Christensen and her associates reported no disclosures.
3. Long-term PPI Use Linked to Increased Risk for Dementia
To take the posttest, go to: http://bit.ly/1nrCdsb
Expires February 24, 2017
VITALS
Key clinical point: Proton pump inhibitors may add to the risk of dementia in older adults.
Major finding: The risk of incident dementia was 44% higher in adults who used PPIs long term, compared with those who did not.
Data source: The prospective cohort study included 73,679 adults aged 75 years and older.
Disclosures: The researchers had no financial conflicts to disclose.
4. Elevated Cardiovascular Risks Linked to Hidradenitis Suppurativa
To take the posttest, go to: http://bit.ly/1nrEFz3
Expires February 17, 2017
VITALS
Key clinical point: Hidradenitis suppurativa is associated with a significantly increased risk of adverse cardiovascular events and all-cause mortality.
Major finding: Individuals with hidradenitis suppurativa had a 57% greater risk of myocardial infarction and 33% greater risk of ischemic stroke, compared with the general population.
Data source: A population-based cohort study in 5,964 patients with hidradenitis suppurativa.
Disclosures: No conflicts of interest were declared.
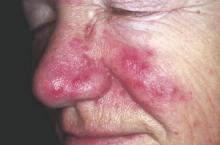
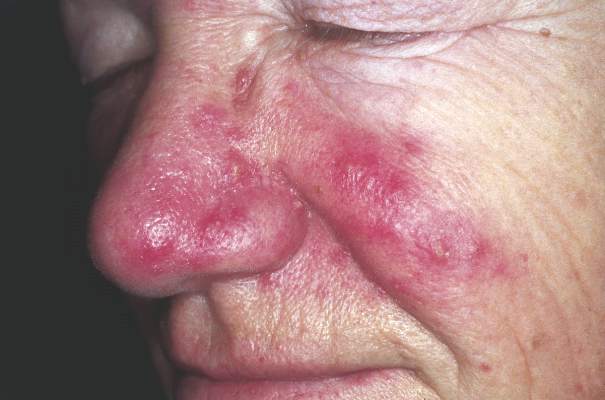
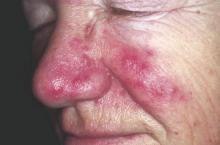
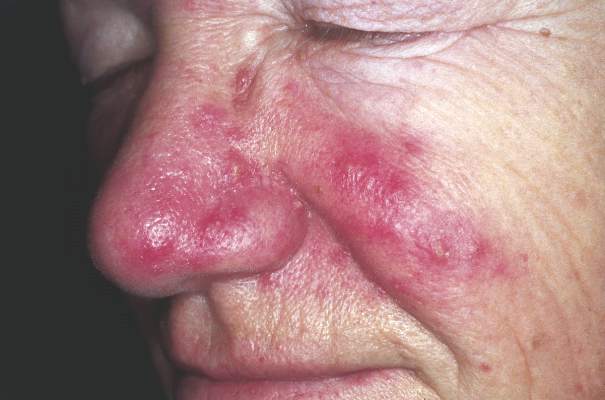
Rosacea Linked to Increased Parkinson’s Disease Risk
Individuals with rosacea were nearly twice as likely to develop Parkinson’s disease as those without rosacea, based on an analysis of results of a nationwide cohort study of 5.4 million people conducted in Denmark. The findings were published online March 21 in JAMA Neurology.
Data from previous studies suggest a link between rosacea and Parkinson’s disease (PD), as both may stem from elevated activity of matrix metalloproteinases, wrote Dr. Alexander Egeberg of the University of Copenhagen, and his associates.
To explore the possible link between rosacea and Parkinson’s disease, the researchers reviewed data on about 5.4 million adults in a Danish database of all citizens aged 18 and older, over a 15-year period from January 1, 1997, to December 31, 2011. A total of 22,387 individuals were diagnosed with Parkinson’s disease and 68,053 were diagnosed with rosacea (JAMA Neurol. 2016 Mar 21. doi: 10.1001/jamaneurol.2016.0022).
The incidence of Parkinson’s disease was 7.62 per 10,000 person-years among rosacea patients vs. 3.54 per 10,000 person-years in the general population, the researchers noted.
Overall, the Parkinson’s disease risk was significantly higher among rosacea patients, with an adjusted incidence rate ratio (IRR) of 1.71 for rosacea patients compared with the general population. The IRR was even higher in patients with ocular rosacea (adjusted IRR, 2.03).
However, treatment with tetracycline appeared to reduce the Parkinson’s risk in the rosacea patients: After controlling for multiple variables, having filled a prescription for tetracycline was associated with a 2% drop in the risk of PD (adjusted IRR, 0.98).
A sensitivity analysis showed no dose-dependent relationship with disease severity; the IRRs for Parkinson’s disease in patients with mild rosacea and moderate to severe rosacea were 1.82 and 1.84, respectively. However, moderate to severe rosacea was defined as cases where tetracyclines were used, and the neuroprotective effect of tetracyclines might impact the relationship between rosacea and Parkinson’s disease, the researchers noted.
“It is tempting to speculate that, in patients with coexistent rosacea, Parkinson’s disease may display other phenotypic characteristics, and it is possible that rosacea-associated features, such as facial flushing, may contribute to support a Parkinson’s disease diagnosis at an early phase of the disease,” the researchers noted.
Limitations of the study include its observational design, which does not allow the establishment of causation, the researchers noted, and additional research is needed to confirm the observations and clinical consequences.
Lead author Dr. Egeberg was employed by Pfizer at the time of the study; one of the coauthors was supported by an unrestricted research scholarship from the Novo Nordisk Foundation.
An intriguing aspect of the study was the lack of disease severity impact on the incidence rate ratio (IRR) on Parkinson’s disease, Dr. Thomas S. Wingo wrote in an accompanying editorial. He added: “In other words, people with moderate to severe rosacea have the same IRR for PD as do those who have moderate disease. To explain this result, the authors hypothesized that a disease-severity effect might be blunted by the treatment of moderate to severe rosacea with tetracycline. The reason for this possible effect is that tetracycline is chemically similar to minocycline, which has shown evidence for exerting a protective effect in animal models of PD, although this effect has not been consistently seen.”
Dr. Wingo noted that the “intriguing finding that increased tetracycline use is associated with a small but appreciable reduction in the risk of PD should be further explored. Of particular interest would be to understand the temporal association between the use of tetracycline and effect on PD risk” (JAMA Neurol. 2016 Mar 21. doi: 10.1001/jamaneurol.2016.0291).
Dr. Wingo is a member of the departments of neurology and human genetics at Emory University, Atlanta. He had no financial conflicts to disclose.
An intriguing aspect of the study was the lack of disease severity impact on the incidence rate ratio (IRR) on Parkinson’s disease, Dr. Thomas S. Wingo wrote in an accompanying editorial. He added: “In other words, people with moderate to severe rosacea have the same IRR for PD as do those who have moderate disease. To explain this result, the authors hypothesized that a disease-severity effect might be blunted by the treatment of moderate to severe rosacea with tetracycline. The reason for this possible effect is that tetracycline is chemically similar to minocycline, which has shown evidence for exerting a protective effect in animal models of PD, although this effect has not been consistently seen.”
Dr. Wingo noted that the “intriguing finding that increased tetracycline use is associated with a small but appreciable reduction in the risk of PD should be further explored. Of particular interest would be to understand the temporal association between the use of tetracycline and effect on PD risk” (JAMA Neurol. 2016 Mar 21. doi: 10.1001/jamaneurol.2016.0291).
Dr. Wingo is a member of the departments of neurology and human genetics at Emory University, Atlanta. He had no financial conflicts to disclose.
An intriguing aspect of the study was the lack of disease severity impact on the incidence rate ratio (IRR) on Parkinson’s disease, Dr. Thomas S. Wingo wrote in an accompanying editorial. He added: “In other words, people with moderate to severe rosacea have the same IRR for PD as do those who have moderate disease. To explain this result, the authors hypothesized that a disease-severity effect might be blunted by the treatment of moderate to severe rosacea with tetracycline. The reason for this possible effect is that tetracycline is chemically similar to minocycline, which has shown evidence for exerting a protective effect in animal models of PD, although this effect has not been consistently seen.”
Dr. Wingo noted that the “intriguing finding that increased tetracycline use is associated with a small but appreciable reduction in the risk of PD should be further explored. Of particular interest would be to understand the temporal association between the use of tetracycline and effect on PD risk” (JAMA Neurol. 2016 Mar 21. doi: 10.1001/jamaneurol.2016.0291).
Dr. Wingo is a member of the departments of neurology and human genetics at Emory University, Atlanta. He had no financial conflicts to disclose.
Individuals with rosacea were nearly twice as likely to develop Parkinson’s disease as those without rosacea, based on an analysis of results of a nationwide cohort study of 5.4 million people conducted in Denmark. The findings were published online March 21 in JAMA Neurology.
Data from previous studies suggest a link between rosacea and Parkinson’s disease (PD), as both may stem from elevated activity of matrix metalloproteinases, wrote Dr. Alexander Egeberg of the University of Copenhagen, and his associates.
To explore the possible link between rosacea and Parkinson’s disease, the researchers reviewed data on about 5.4 million adults in a Danish database of all citizens aged 18 and older, over a 15-year period from January 1, 1997, to December 31, 2011. A total of 22,387 individuals were diagnosed with Parkinson’s disease and 68,053 were diagnosed with rosacea (JAMA Neurol. 2016 Mar 21. doi: 10.1001/jamaneurol.2016.0022).
The incidence of Parkinson’s disease was 7.62 per 10,000 person-years among rosacea patients vs. 3.54 per 10,000 person-years in the general population, the researchers noted.
Overall, the Parkinson’s disease risk was significantly higher among rosacea patients, with an adjusted incidence rate ratio (IRR) of 1.71 for rosacea patients compared with the general population. The IRR was even higher in patients with ocular rosacea (adjusted IRR, 2.03).
However, treatment with tetracycline appeared to reduce the Parkinson’s risk in the rosacea patients: After controlling for multiple variables, having filled a prescription for tetracycline was associated with a 2% drop in the risk of PD (adjusted IRR, 0.98).
A sensitivity analysis showed no dose-dependent relationship with disease severity; the IRRs for Parkinson’s disease in patients with mild rosacea and moderate to severe rosacea were 1.82 and 1.84, respectively. However, moderate to severe rosacea was defined as cases where tetracyclines were used, and the neuroprotective effect of tetracyclines might impact the relationship between rosacea and Parkinson’s disease, the researchers noted.
“It is tempting to speculate that, in patients with coexistent rosacea, Parkinson’s disease may display other phenotypic characteristics, and it is possible that rosacea-associated features, such as facial flushing, may contribute to support a Parkinson’s disease diagnosis at an early phase of the disease,” the researchers noted.
Limitations of the study include its observational design, which does not allow the establishment of causation, the researchers noted, and additional research is needed to confirm the observations and clinical consequences.
Lead author Dr. Egeberg was employed by Pfizer at the time of the study; one of the coauthors was supported by an unrestricted research scholarship from the Novo Nordisk Foundation.
Individuals with rosacea were nearly twice as likely to develop Parkinson’s disease as those without rosacea, based on an analysis of results of a nationwide cohort study of 5.4 million people conducted in Denmark. The findings were published online March 21 in JAMA Neurology.
Data from previous studies suggest a link between rosacea and Parkinson’s disease (PD), as both may stem from elevated activity of matrix metalloproteinases, wrote Dr. Alexander Egeberg of the University of Copenhagen, and his associates.
To explore the possible link between rosacea and Parkinson’s disease, the researchers reviewed data on about 5.4 million adults in a Danish database of all citizens aged 18 and older, over a 15-year period from January 1, 1997, to December 31, 2011. A total of 22,387 individuals were diagnosed with Parkinson’s disease and 68,053 were diagnosed with rosacea (JAMA Neurol. 2016 Mar 21. doi: 10.1001/jamaneurol.2016.0022).
The incidence of Parkinson’s disease was 7.62 per 10,000 person-years among rosacea patients vs. 3.54 per 10,000 person-years in the general population, the researchers noted.
Overall, the Parkinson’s disease risk was significantly higher among rosacea patients, with an adjusted incidence rate ratio (IRR) of 1.71 for rosacea patients compared with the general population. The IRR was even higher in patients with ocular rosacea (adjusted IRR, 2.03).
However, treatment with tetracycline appeared to reduce the Parkinson’s risk in the rosacea patients: After controlling for multiple variables, having filled a prescription for tetracycline was associated with a 2% drop in the risk of PD (adjusted IRR, 0.98).
A sensitivity analysis showed no dose-dependent relationship with disease severity; the IRRs for Parkinson’s disease in patients with mild rosacea and moderate to severe rosacea were 1.82 and 1.84, respectively. However, moderate to severe rosacea was defined as cases where tetracyclines were used, and the neuroprotective effect of tetracyclines might impact the relationship between rosacea and Parkinson’s disease, the researchers noted.
“It is tempting to speculate that, in patients with coexistent rosacea, Parkinson’s disease may display other phenotypic characteristics, and it is possible that rosacea-associated features, such as facial flushing, may contribute to support a Parkinson’s disease diagnosis at an early phase of the disease,” the researchers noted.
Limitations of the study include its observational design, which does not allow the establishment of causation, the researchers noted, and additional research is needed to confirm the observations and clinical consequences.
Lead author Dr. Egeberg was employed by Pfizer at the time of the study; one of the coauthors was supported by an unrestricted research scholarship from the Novo Nordisk Foundation.
FROM JAMA NEUROLOGY
Rosacea linked to increased Parkinson’s disease risk
Individuals with rosacea were nearly twice as likely to develop Parkinson’s disease as those without rosacea, based on an analysis of results of a nationwide cohort study of 5.4 million people conducted in Denmark. The findings were published online March 21 in JAMA Neurology.
Data from previous studies suggest a link between rosacea and Parkinson’s disease (PD), as both may stem from elevated activity of matrix metalloproteinases, wrote Dr. Alexander Egeberg of the University of Copenhagen, and his associates.
To explore the possible link between rosacea and Parkinson’s disease, the researchers reviewed data on about 5.4 million adults in a Danish database of all citizens aged 18 and older, over a 15-year period from January 1, 1997, to December 31, 2011. A total of 22,387 individuals were diagnosed with Parkinson’s disease and 68,053 were diagnosed with rosacea (JAMA Neurol. 2016 Mar 21. doi: 10.1001/jamaneurol.2016.0022).
The incidence of Parkinson’s disease was 7.62 per 10,000 person-years among rosacea patients vs. 3.54 per 10,000 person-years in the general population, the researchers noted.
Overall, the Parkinson’s disease risk was significantly higher among rosacea patients, with an adjusted incidence rate ratio (IRR) of 1.71 for rosacea patients compared with the general population. The IRR was even higher in patients with ocular rosacea (adjusted IRR, 2.03).
However, treatment with tetracycline appeared to reduce the Parkinson’s risk in the rosacea patients: After controlling for multiple variables, having filled a prescription for tetracycline was associated with a 2% drop in the risk of PD (adjusted IRR, 0.98).
A sensitivity analysis showed no dose-dependent relationship with disease severity; the IRRs for Parkinson’s disease in patients with mild rosacea and moderate to severe rosacea were 1.82 and 1.84, respectively. However, moderate to severe rosacea was defined as cases where tetracyclines were used, and the neuroprotective effect of tetracyclines might impact the relationship between rosacea and Parkinson’s disease, the researchers noted.
“It is tempting to speculate that, in patients with coexistent rosacea, Parkinson’s disease may display other phenotypic characteristics, and it is possible that rosacea-associated features, such as facial flushing, may contribute to support a Parkinson’s disease diagnosis at an early phase of the disease,” the researchers noted.
Limitations of the study include its observational design, which does not allow the establishment of causation, the researchers noted, and additional research is needed to confirm the observations and clinical consequences.
Lead author Dr. Egeberg was employed by Pfizer at the time of the study; one of the coauthors was supported by an unrestricted research scholarship from the Novo Nordisk Foundation.
An intriguing aspect of the study was the lack of disease severity impact on the incidence rate ratio (IRR) on Parkinson’s disease, Dr. Thomas S. Wingo wrote in an accompanying editorial. He added: “In other words, people with moderate to severe rosacea have the same IRR for PD as do those who have moderate disease. To explain this result, the authors hypothesized that a disease-severity effect might be blunted by the treatment of moderate to severe rosacea with tetracycline. The reason for this possible effect is that tetracycline is chemically similar to minocycline, which has shown evidence for exerting a protective effect in animal models of PD, although this effect has not been consistently seen.”
Dr. Wingo noted that the “intriguing finding that increased tetracycline use is associated with a small but appreciable reduction in the risk of PD should be further explored. Of particular interest would be to understand the temporal association between the use of tetracycline and effect on PD risk” (JAMA Neurol. 2016 Mar 21. doi: 10.1001/jamaneurol.2016.0291).
Dr. Wingo is a member of the departments of neurology and human genetics at Emory University, Atlanta. He had no financial conflicts to disclose.
An intriguing aspect of the study was the lack of disease severity impact on the incidence rate ratio (IRR) on Parkinson’s disease, Dr. Thomas S. Wingo wrote in an accompanying editorial. He added: “In other words, people with moderate to severe rosacea have the same IRR for PD as do those who have moderate disease. To explain this result, the authors hypothesized that a disease-severity effect might be blunted by the treatment of moderate to severe rosacea with tetracycline. The reason for this possible effect is that tetracycline is chemically similar to minocycline, which has shown evidence for exerting a protective effect in animal models of PD, although this effect has not been consistently seen.”
Dr. Wingo noted that the “intriguing finding that increased tetracycline use is associated with a small but appreciable reduction in the risk of PD should be further explored. Of particular interest would be to understand the temporal association between the use of tetracycline and effect on PD risk” (JAMA Neurol. 2016 Mar 21. doi: 10.1001/jamaneurol.2016.0291).
Dr. Wingo is a member of the departments of neurology and human genetics at Emory University, Atlanta. He had no financial conflicts to disclose.
An intriguing aspect of the study was the lack of disease severity impact on the incidence rate ratio (IRR) on Parkinson’s disease, Dr. Thomas S. Wingo wrote in an accompanying editorial. He added: “In other words, people with moderate to severe rosacea have the same IRR for PD as do those who have moderate disease. To explain this result, the authors hypothesized that a disease-severity effect might be blunted by the treatment of moderate to severe rosacea with tetracycline. The reason for this possible effect is that tetracycline is chemically similar to minocycline, which has shown evidence for exerting a protective effect in animal models of PD, although this effect has not been consistently seen.”
Dr. Wingo noted that the “intriguing finding that increased tetracycline use is associated with a small but appreciable reduction in the risk of PD should be further explored. Of particular interest would be to understand the temporal association between the use of tetracycline and effect on PD risk” (JAMA Neurol. 2016 Mar 21. doi: 10.1001/jamaneurol.2016.0291).
Dr. Wingo is a member of the departments of neurology and human genetics at Emory University, Atlanta. He had no financial conflicts to disclose.
Individuals with rosacea were nearly twice as likely to develop Parkinson’s disease as those without rosacea, based on an analysis of results of a nationwide cohort study of 5.4 million people conducted in Denmark. The findings were published online March 21 in JAMA Neurology.
Data from previous studies suggest a link between rosacea and Parkinson’s disease (PD), as both may stem from elevated activity of matrix metalloproteinases, wrote Dr. Alexander Egeberg of the University of Copenhagen, and his associates.
To explore the possible link between rosacea and Parkinson’s disease, the researchers reviewed data on about 5.4 million adults in a Danish database of all citizens aged 18 and older, over a 15-year period from January 1, 1997, to December 31, 2011. A total of 22,387 individuals were diagnosed with Parkinson’s disease and 68,053 were diagnosed with rosacea (JAMA Neurol. 2016 Mar 21. doi: 10.1001/jamaneurol.2016.0022).
The incidence of Parkinson’s disease was 7.62 per 10,000 person-years among rosacea patients vs. 3.54 per 10,000 person-years in the general population, the researchers noted.
Overall, the Parkinson’s disease risk was significantly higher among rosacea patients, with an adjusted incidence rate ratio (IRR) of 1.71 for rosacea patients compared with the general population. The IRR was even higher in patients with ocular rosacea (adjusted IRR, 2.03).
However, treatment with tetracycline appeared to reduce the Parkinson’s risk in the rosacea patients: After controlling for multiple variables, having filled a prescription for tetracycline was associated with a 2% drop in the risk of PD (adjusted IRR, 0.98).
A sensitivity analysis showed no dose-dependent relationship with disease severity; the IRRs for Parkinson’s disease in patients with mild rosacea and moderate to severe rosacea were 1.82 and 1.84, respectively. However, moderate to severe rosacea was defined as cases where tetracyclines were used, and the neuroprotective effect of tetracyclines might impact the relationship between rosacea and Parkinson’s disease, the researchers noted.
“It is tempting to speculate that, in patients with coexistent rosacea, Parkinson’s disease may display other phenotypic characteristics, and it is possible that rosacea-associated features, such as facial flushing, may contribute to support a Parkinson’s disease diagnosis at an early phase of the disease,” the researchers noted.
Limitations of the study include its observational design, which does not allow the establishment of causation, the researchers noted, and additional research is needed to confirm the observations and clinical consequences.
Lead author Dr. Egeberg was employed by Pfizer at the time of the study; one of the coauthors was supported by an unrestricted research scholarship from the Novo Nordisk Foundation.
Individuals with rosacea were nearly twice as likely to develop Parkinson’s disease as those without rosacea, based on an analysis of results of a nationwide cohort study of 5.4 million people conducted in Denmark. The findings were published online March 21 in JAMA Neurology.
Data from previous studies suggest a link between rosacea and Parkinson’s disease (PD), as both may stem from elevated activity of matrix metalloproteinases, wrote Dr. Alexander Egeberg of the University of Copenhagen, and his associates.
To explore the possible link between rosacea and Parkinson’s disease, the researchers reviewed data on about 5.4 million adults in a Danish database of all citizens aged 18 and older, over a 15-year period from January 1, 1997, to December 31, 2011. A total of 22,387 individuals were diagnosed with Parkinson’s disease and 68,053 were diagnosed with rosacea (JAMA Neurol. 2016 Mar 21. doi: 10.1001/jamaneurol.2016.0022).
The incidence of Parkinson’s disease was 7.62 per 10,000 person-years among rosacea patients vs. 3.54 per 10,000 person-years in the general population, the researchers noted.
Overall, the Parkinson’s disease risk was significantly higher among rosacea patients, with an adjusted incidence rate ratio (IRR) of 1.71 for rosacea patients compared with the general population. The IRR was even higher in patients with ocular rosacea (adjusted IRR, 2.03).
However, treatment with tetracycline appeared to reduce the Parkinson’s risk in the rosacea patients: After controlling for multiple variables, having filled a prescription for tetracycline was associated with a 2% drop in the risk of PD (adjusted IRR, 0.98).
A sensitivity analysis showed no dose-dependent relationship with disease severity; the IRRs for Parkinson’s disease in patients with mild rosacea and moderate to severe rosacea were 1.82 and 1.84, respectively. However, moderate to severe rosacea was defined as cases where tetracyclines were used, and the neuroprotective effect of tetracyclines might impact the relationship between rosacea and Parkinson’s disease, the researchers noted.
“It is tempting to speculate that, in patients with coexistent rosacea, Parkinson’s disease may display other phenotypic characteristics, and it is possible that rosacea-associated features, such as facial flushing, may contribute to support a Parkinson’s disease diagnosis at an early phase of the disease,” the researchers noted.
Limitations of the study include its observational design, which does not allow the establishment of causation, the researchers noted, and additional research is needed to confirm the observations and clinical consequences.
Lead author Dr. Egeberg was employed by Pfizer at the time of the study; one of the coauthors was supported by an unrestricted research scholarship from the Novo Nordisk Foundation.
FROM JAMA NEUROLOGY
Key clinical point: Rosacea was found to be an independent risk factor for Parkinson’s disease, an effect reduced by the use of tetracycline.
Major finding: The incidence of Parkinson’s disease was 7.62 per 10,000 person-years among people with rosacea vs. 3.54 per 10,000 person-years in the general population over a 15-year follow-up period.
Data source: The data come from a nationwide cohort study conducted in Denmark that included 5.4 million adults aged 18 years and older.
Disclosures: Lead author Dr. Alexander Egeberg was employed by Pfizer at the time of the study; a study coauthor was supported by an unrestricted research scholarship from the Novo Nordisk Foundation.
Options to Treat Hyperhidrosis Are Increasing
Clinicians have an expanding range of options to offer patients with persistent hyperhidrosis, Dr. David M. Pariser said at the Caribbean Dermatology Symposium.
Hyperhidrosis currently affects an estimated 7.8 million individuals in the United States. About half have axillary hyperhidrosis, and include about 1.3 million people who report hyperhidrosis that is “barely tolerable” or “intolerable,” Dr. Pariser said at the meeting, provided by Global Academy for Medical Education.
The treatment options include next generation antiperspirants that contain aluminum zirconium trichlorohydrex, which he said are more effective and less irritating than previously available products.
Although many patients apply antiperspirant as part of their morning routines, topical antiperspirants with aluminum chloride are effective when applied overnight, or for at least 6-8 hours, according to Dr. Pariser, professor of dermatology at Eastern Virginia Medical School, Norfolk, Va. To avoid irritating acid formation, patients should make sure the skin is dry before application, but if irritation occurs, he recommends that it should be washed off in the morning “before sweating begins.”
Aluminum chloride products should be applied nightly until patients see improvement, and then they can decrease the frequency of use. Concerns about a potential link between aluminum and Alzheimer’s disease stem from a 1960s study that was never successfully replicated, he pointed out.
In general, topical agents are a useful adjunct to other hyperhidrosis treatments, such as onabotulinumtoxinA, Dr. Pariser said. “Many insurance companies consider treatment of hyperhidrosis with iontophoresis or botulinum toxin medically necessary when topical aluminum chloride or other extra strength antiperspirants are ineffective or result in irritation,” he noted.
Other new topical products on the horizon for hyperhidrosis include glycopyrrolate wipes and gel, oxybutynin gel, and topical botulinum toxins, he added.
Patients suffering from generalized hyperhidrosis or hyperhidrosis of a spinal cord injury may benefit from systemic treatment, although no systemic agents are currently approved by the Food and Drug Administration for this purpose, Dr. Pariser said.
Glycopyrrolate is the preferred systemic medication for off-label hyperhidrosis treatment, and patients typically start at 1 mg twice daily, increasing the dose by 1 mg a day each week until they achieve the desired improvement or are unable to tolerate adverse effects, he said. Data also support the use of oral oxybutynin for hyperhidrosis, he noted.
Patients seeking more “permanent” treatment for hyperhidrosis might consider microwave thermolysis with an FDA-cleared device designed to stop sweating by using microwaves to destroy the eccrine glands, which do not regenerate, Dr. Pariser said. The device works by delivering microwave energy precisely in the dermal-fat interface region. Deeper tissue is unaffected, and contact cooling protects the epidermis and upper dermis.
Side effects from microwave thermolysis are usually minimal and transient, he noted. “The most common side effects are swelling and tenderness in the treated area, which can last up to a few weeks.”
By contrast, the effects of botulinum toxin on hyperhidrosis are not permanent, but patients report a high level of satisfaction and improved quality of life with this treatment option, he said. Duration of treatment varies, but data from a 16-month, randomized, double-blind trial of 207 patients showed a mean duration of 7 months between treatments.
Overall, treatment of patients with focal hyperhidrosis “leads to greater improvement of a patient’s quality of life than treatment of any other dermatologic disorder,” Dr. Pariser said. Treatments are relatively easy to learn, economically viable, and easily incorporated into a routine office practice, he said.
Dr. Pariser disclosed ties with Allergan, Dermira, Watson Labs, Ulthera, Brickell Biotech, Revance, Anterios, and Theravida.
Global Academy and this news organization are owned by the same parent company.
Clinicians have an expanding range of options to offer patients with persistent hyperhidrosis, Dr. David M. Pariser said at the Caribbean Dermatology Symposium.
Hyperhidrosis currently affects an estimated 7.8 million individuals in the United States. About half have axillary hyperhidrosis, and include about 1.3 million people who report hyperhidrosis that is “barely tolerable” or “intolerable,” Dr. Pariser said at the meeting, provided by Global Academy for Medical Education.
The treatment options include next generation antiperspirants that contain aluminum zirconium trichlorohydrex, which he said are more effective and less irritating than previously available products.
Although many patients apply antiperspirant as part of their morning routines, topical antiperspirants with aluminum chloride are effective when applied overnight, or for at least 6-8 hours, according to Dr. Pariser, professor of dermatology at Eastern Virginia Medical School, Norfolk, Va. To avoid irritating acid formation, patients should make sure the skin is dry before application, but if irritation occurs, he recommends that it should be washed off in the morning “before sweating begins.”
Aluminum chloride products should be applied nightly until patients see improvement, and then they can decrease the frequency of use. Concerns about a potential link between aluminum and Alzheimer’s disease stem from a 1960s study that was never successfully replicated, he pointed out.
In general, topical agents are a useful adjunct to other hyperhidrosis treatments, such as onabotulinumtoxinA, Dr. Pariser said. “Many insurance companies consider treatment of hyperhidrosis with iontophoresis or botulinum toxin medically necessary when topical aluminum chloride or other extra strength antiperspirants are ineffective or result in irritation,” he noted.
Other new topical products on the horizon for hyperhidrosis include glycopyrrolate wipes and gel, oxybutynin gel, and topical botulinum toxins, he added.
Patients suffering from generalized hyperhidrosis or hyperhidrosis of a spinal cord injury may benefit from systemic treatment, although no systemic agents are currently approved by the Food and Drug Administration for this purpose, Dr. Pariser said.
Glycopyrrolate is the preferred systemic medication for off-label hyperhidrosis treatment, and patients typically start at 1 mg twice daily, increasing the dose by 1 mg a day each week until they achieve the desired improvement or are unable to tolerate adverse effects, he said. Data also support the use of oral oxybutynin for hyperhidrosis, he noted.
Patients seeking more “permanent” treatment for hyperhidrosis might consider microwave thermolysis with an FDA-cleared device designed to stop sweating by using microwaves to destroy the eccrine glands, which do not regenerate, Dr. Pariser said. The device works by delivering microwave energy precisely in the dermal-fat interface region. Deeper tissue is unaffected, and contact cooling protects the epidermis and upper dermis.
Side effects from microwave thermolysis are usually minimal and transient, he noted. “The most common side effects are swelling and tenderness in the treated area, which can last up to a few weeks.”
By contrast, the effects of botulinum toxin on hyperhidrosis are not permanent, but patients report a high level of satisfaction and improved quality of life with this treatment option, he said. Duration of treatment varies, but data from a 16-month, randomized, double-blind trial of 207 patients showed a mean duration of 7 months between treatments.
Overall, treatment of patients with focal hyperhidrosis “leads to greater improvement of a patient’s quality of life than treatment of any other dermatologic disorder,” Dr. Pariser said. Treatments are relatively easy to learn, economically viable, and easily incorporated into a routine office practice, he said.
Dr. Pariser disclosed ties with Allergan, Dermira, Watson Labs, Ulthera, Brickell Biotech, Revance, Anterios, and Theravida.
Global Academy and this news organization are owned by the same parent company.
Clinicians have an expanding range of options to offer patients with persistent hyperhidrosis, Dr. David M. Pariser said at the Caribbean Dermatology Symposium.
Hyperhidrosis currently affects an estimated 7.8 million individuals in the United States. About half have axillary hyperhidrosis, and include about 1.3 million people who report hyperhidrosis that is “barely tolerable” or “intolerable,” Dr. Pariser said at the meeting, provided by Global Academy for Medical Education.
The treatment options include next generation antiperspirants that contain aluminum zirconium trichlorohydrex, which he said are more effective and less irritating than previously available products.
Although many patients apply antiperspirant as part of their morning routines, topical antiperspirants with aluminum chloride are effective when applied overnight, or for at least 6-8 hours, according to Dr. Pariser, professor of dermatology at Eastern Virginia Medical School, Norfolk, Va. To avoid irritating acid formation, patients should make sure the skin is dry before application, but if irritation occurs, he recommends that it should be washed off in the morning “before sweating begins.”
Aluminum chloride products should be applied nightly until patients see improvement, and then they can decrease the frequency of use. Concerns about a potential link between aluminum and Alzheimer’s disease stem from a 1960s study that was never successfully replicated, he pointed out.
In general, topical agents are a useful adjunct to other hyperhidrosis treatments, such as onabotulinumtoxinA, Dr. Pariser said. “Many insurance companies consider treatment of hyperhidrosis with iontophoresis or botulinum toxin medically necessary when topical aluminum chloride or other extra strength antiperspirants are ineffective or result in irritation,” he noted.
Other new topical products on the horizon for hyperhidrosis include glycopyrrolate wipes and gel, oxybutynin gel, and topical botulinum toxins, he added.
Patients suffering from generalized hyperhidrosis or hyperhidrosis of a spinal cord injury may benefit from systemic treatment, although no systemic agents are currently approved by the Food and Drug Administration for this purpose, Dr. Pariser said.
Glycopyrrolate is the preferred systemic medication for off-label hyperhidrosis treatment, and patients typically start at 1 mg twice daily, increasing the dose by 1 mg a day each week until they achieve the desired improvement or are unable to tolerate adverse effects, he said. Data also support the use of oral oxybutynin for hyperhidrosis, he noted.
Patients seeking more “permanent” treatment for hyperhidrosis might consider microwave thermolysis with an FDA-cleared device designed to stop sweating by using microwaves to destroy the eccrine glands, which do not regenerate, Dr. Pariser said. The device works by delivering microwave energy precisely in the dermal-fat interface region. Deeper tissue is unaffected, and contact cooling protects the epidermis and upper dermis.
Side effects from microwave thermolysis are usually minimal and transient, he noted. “The most common side effects are swelling and tenderness in the treated area, which can last up to a few weeks.”
By contrast, the effects of botulinum toxin on hyperhidrosis are not permanent, but patients report a high level of satisfaction and improved quality of life with this treatment option, he said. Duration of treatment varies, but data from a 16-month, randomized, double-blind trial of 207 patients showed a mean duration of 7 months between treatments.
Overall, treatment of patients with focal hyperhidrosis “leads to greater improvement of a patient’s quality of life than treatment of any other dermatologic disorder,” Dr. Pariser said. Treatments are relatively easy to learn, economically viable, and easily incorporated into a routine office practice, he said.
Dr. Pariser disclosed ties with Allergan, Dermira, Watson Labs, Ulthera, Brickell Biotech, Revance, Anterios, and Theravida.
Global Academy and this news organization are owned by the same parent company.
EXPERT ANALYSIS FROM THE CARIBBEAN DERMATOLOGY SYMPOSIUM
Options to treat hyperhidrosis are increasing
Clinicians have an expanding range of options to offer patients with persistent hyperhidrosis, Dr. David M. Pariser said at the Caribbean Dermatology Symposium.
Hyperhidrosis currently affects an estimated 7.8 million individuals in the United States. About half have axillary hyperhidrosis, and include about 1.3 million people who report hyperhidrosis that is “barely tolerable” or “intolerable,” Dr. Pariser said at the meeting, provided by Global Academy for Medical Education.
The treatment options include next generation antiperspirants that contain aluminum zirconium trichlorohydrex, which he said are more effective and less irritating than previously available products.
Although many patients apply antiperspirant as part of their morning routines, topical antiperspirants with aluminum chloride are effective when applied overnight, or for at least 6-8 hours, according to Dr. Pariser, professor of dermatology at Eastern Virginia Medical School, Norfolk, Va. To avoid irritating acid formation, patients should make sure the skin is dry before application, but if irritation occurs, he recommends that it should be washed off in the morning “before sweating begins.”
Aluminum chloride products should be applied nightly until patients see improvement, and then they can decrease the frequency of use. Concerns about a potential link between aluminum and Alzheimer’s disease stem from a 1960s study that was never successfully replicated, he pointed out.
In general, topical agents are a useful adjunct to other hyperhidrosis treatments, such as onabotulinumtoxinA, Dr. Pariser said. “Many insurance companies consider treatment of hyperhidrosis with iontophoresis or botulinum toxin medically necessary when topical aluminum chloride or other extra strength antiperspirants are ineffective or result in irritation,” he noted.
Other new topical products on the horizon for hyperhidrosis include glycopyrrolate wipes and gel, oxybutynin gel, and topical botulinum toxins, he added.
Patients suffering from generalized hyperhidrosis or hyperhidrosis of a spinal cord injury may benefit from systemic treatment, although no systemic agents are currently approved by the Food and Drug Administration for this purpose, Dr. Pariser said.
Glycopyrrolate is the preferred systemic medication for off-label hyperhidrosis treatment, and patients typically start at 1 mg twice daily, increasing the dose by 1 mg a day each week until they achieve the desired improvement or are unable to tolerate adverse effects, he said. Data also support the use of oral oxybutynin for hyperhidrosis, he noted.
Patients seeking more “permanent” treatment for hyperhidrosis might consider microwave thermolysis with an FDA-cleared device designed to stop sweating by using microwaves to destroy the eccrine glands, which do not regenerate, Dr. Pariser said. The device works by delivering microwave energy precisely in the dermal-fat interface region. Deeper tissue is unaffected, and contact cooling protects the epidermis and upper dermis.
Side effects from microwave thermolysis are usually minimal and transient, he noted. “The most common side effects are swelling and tenderness in the treated area, which can last up to a few weeks.”
By contrast, the effects of botulinum toxin on hyperhidrosis are not permanent, but patients report a high level of satisfaction and improved quality of life with this treatment option, he said. Duration of treatment varies, but data from a 16-month, randomized, double-blind trial of 207 patients showed a mean duration of 7 months between treatments.
Overall, treatment of patients with focal hyperhidrosis “leads to greater improvement of a patient’s quality of life than treatment of any other dermatologic disorder,” Dr. Pariser said. Treatments are relatively easy to learn, economically viable, and easily incorporated into a routine office practice, he said.
Dr. Pariser disclosed ties with Allergan, Dermira, Watson Labs, Ulthera, Brickell Biotech, Revance, Anterios, and Theravida.
Global Academy and this news organization are owned by the same parent company.
Clinicians have an expanding range of options to offer patients with persistent hyperhidrosis, Dr. David M. Pariser said at the Caribbean Dermatology Symposium.
Hyperhidrosis currently affects an estimated 7.8 million individuals in the United States. About half have axillary hyperhidrosis, and include about 1.3 million people who report hyperhidrosis that is “barely tolerable” or “intolerable,” Dr. Pariser said at the meeting, provided by Global Academy for Medical Education.
The treatment options include next generation antiperspirants that contain aluminum zirconium trichlorohydrex, which he said are more effective and less irritating than previously available products.
Although many patients apply antiperspirant as part of their morning routines, topical antiperspirants with aluminum chloride are effective when applied overnight, or for at least 6-8 hours, according to Dr. Pariser, professor of dermatology at Eastern Virginia Medical School, Norfolk, Va. To avoid irritating acid formation, patients should make sure the skin is dry before application, but if irritation occurs, he recommends that it should be washed off in the morning “before sweating begins.”
Aluminum chloride products should be applied nightly until patients see improvement, and then they can decrease the frequency of use. Concerns about a potential link between aluminum and Alzheimer’s disease stem from a 1960s study that was never successfully replicated, he pointed out.
In general, topical agents are a useful adjunct to other hyperhidrosis treatments, such as onabotulinumtoxinA, Dr. Pariser said. “Many insurance companies consider treatment of hyperhidrosis with iontophoresis or botulinum toxin medically necessary when topical aluminum chloride or other extra strength antiperspirants are ineffective or result in irritation,” he noted.
Other new topical products on the horizon for hyperhidrosis include glycopyrrolate wipes and gel, oxybutynin gel, and topical botulinum toxins, he added.
Patients suffering from generalized hyperhidrosis or hyperhidrosis of a spinal cord injury may benefit from systemic treatment, although no systemic agents are currently approved by the Food and Drug Administration for this purpose, Dr. Pariser said.
Glycopyrrolate is the preferred systemic medication for off-label hyperhidrosis treatment, and patients typically start at 1 mg twice daily, increasing the dose by 1 mg a day each week until they achieve the desired improvement or are unable to tolerate adverse effects, he said. Data also support the use of oral oxybutynin for hyperhidrosis, he noted.
Patients seeking more “permanent” treatment for hyperhidrosis might consider microwave thermolysis with an FDA-cleared device designed to stop sweating by using microwaves to destroy the eccrine glands, which do not regenerate, Dr. Pariser said. The device works by delivering microwave energy precisely in the dermal-fat interface region. Deeper tissue is unaffected, and contact cooling protects the epidermis and upper dermis.
Side effects from microwave thermolysis are usually minimal and transient, he noted. “The most common side effects are swelling and tenderness in the treated area, which can last up to a few weeks.”
By contrast, the effects of botulinum toxin on hyperhidrosis are not permanent, but patients report a high level of satisfaction and improved quality of life with this treatment option, he said. Duration of treatment varies, but data from a 16-month, randomized, double-blind trial of 207 patients showed a mean duration of 7 months between treatments.
Overall, treatment of patients with focal hyperhidrosis “leads to greater improvement of a patient’s quality of life than treatment of any other dermatologic disorder,” Dr. Pariser said. Treatments are relatively easy to learn, economically viable, and easily incorporated into a routine office practice, he said.
Dr. Pariser disclosed ties with Allergan, Dermira, Watson Labs, Ulthera, Brickell Biotech, Revance, Anterios, and Theravida.
Global Academy and this news organization are owned by the same parent company.
Clinicians have an expanding range of options to offer patients with persistent hyperhidrosis, Dr. David M. Pariser said at the Caribbean Dermatology Symposium.
Hyperhidrosis currently affects an estimated 7.8 million individuals in the United States. About half have axillary hyperhidrosis, and include about 1.3 million people who report hyperhidrosis that is “barely tolerable” or “intolerable,” Dr. Pariser said at the meeting, provided by Global Academy for Medical Education.
The treatment options include next generation antiperspirants that contain aluminum zirconium trichlorohydrex, which he said are more effective and less irritating than previously available products.
Although many patients apply antiperspirant as part of their morning routines, topical antiperspirants with aluminum chloride are effective when applied overnight, or for at least 6-8 hours, according to Dr. Pariser, professor of dermatology at Eastern Virginia Medical School, Norfolk, Va. To avoid irritating acid formation, patients should make sure the skin is dry before application, but if irritation occurs, he recommends that it should be washed off in the morning “before sweating begins.”
Aluminum chloride products should be applied nightly until patients see improvement, and then they can decrease the frequency of use. Concerns about a potential link between aluminum and Alzheimer’s disease stem from a 1960s study that was never successfully replicated, he pointed out.
In general, topical agents are a useful adjunct to other hyperhidrosis treatments, such as onabotulinumtoxinA, Dr. Pariser said. “Many insurance companies consider treatment of hyperhidrosis with iontophoresis or botulinum toxin medically necessary when topical aluminum chloride or other extra strength antiperspirants are ineffective or result in irritation,” he noted.
Other new topical products on the horizon for hyperhidrosis include glycopyrrolate wipes and gel, oxybutynin gel, and topical botulinum toxins, he added.
Patients suffering from generalized hyperhidrosis or hyperhidrosis of a spinal cord injury may benefit from systemic treatment, although no systemic agents are currently approved by the Food and Drug Administration for this purpose, Dr. Pariser said.
Glycopyrrolate is the preferred systemic medication for off-label hyperhidrosis treatment, and patients typically start at 1 mg twice daily, increasing the dose by 1 mg a day each week until they achieve the desired improvement or are unable to tolerate adverse effects, he said. Data also support the use of oral oxybutynin for hyperhidrosis, he noted.
Patients seeking more “permanent” treatment for hyperhidrosis might consider microwave thermolysis with an FDA-cleared device designed to stop sweating by using microwaves to destroy the eccrine glands, which do not regenerate, Dr. Pariser said. The device works by delivering microwave energy precisely in the dermal-fat interface region. Deeper tissue is unaffected, and contact cooling protects the epidermis and upper dermis.
Side effects from microwave thermolysis are usually minimal and transient, he noted. “The most common side effects are swelling and tenderness in the treated area, which can last up to a few weeks.”
By contrast, the effects of botulinum toxin on hyperhidrosis are not permanent, but patients report a high level of satisfaction and improved quality of life with this treatment option, he said. Duration of treatment varies, but data from a 16-month, randomized, double-blind trial of 207 patients showed a mean duration of 7 months between treatments.
Overall, treatment of patients with focal hyperhidrosis “leads to greater improvement of a patient’s quality of life than treatment of any other dermatologic disorder,” Dr. Pariser said. Treatments are relatively easy to learn, economically viable, and easily incorporated into a routine office practice, he said.
Dr. Pariser disclosed ties with Allergan, Dermira, Watson Labs, Ulthera, Brickell Biotech, Revance, Anterios, and Theravida.
Global Academy and this news organization are owned by the same parent company.
EXPERT ANALYSIS FROM THE CARIBBEAN DERMATOLOGY SYMPOSIUM
FDA invites involvement in guidance plans
The Food and Drug Administration wants to become more transparent and informative in its drug regulatory decision making, Dr. Jonathan Wilkin said at the Caribbean Dermatology Symposium.
“Part of this approach is the structured benefit-risk assessment tool,” he said in an interview.
The Prescription Drug User Fee Act (PDUFA) is reauthorized every 5 years, most recently in 2013, Dr. Wilkin said at the meeting, provided by Global Academy for Medical Education.
Two notable changes to PDUFA V (the fifth authorization of PDUFA) from previous versions are a focus on getting input from patients and their families on what constitutes a clinically meaningful improvement, and the publication of the FDA’s draft document on Structured Benefit-Risk Assessment, which is written for FDA staff and regulated drug manufacturers, explained Dr. Wilkin, a dermatologist in the Cayman Islands.
These changes are playing out with specific steps to increase patient participation and informing into the FDA’s decision-making process, said Dr. Wilkin, a former director of the Food and Drug Administration’s Division of Dermatologic and Dental Drug Products.
“Patients, their families, and patient advocacy groups are being invited to attend meetings with FDA to discuss patient needs in specific diseases, such as psoriasis and alopecia areata. This is expected to lead to perspectives on the benefit-risk assessments by the patients, for example, what risks they are willing to accept for what level of direct benefit, and their relationship to patient-reported outcome measures in clinical trials,” he said. “In the future, these patient reported outcome measures may be in package inserts and could be useful in physicians' discussions of therapeutic options with patients.”
The FDA’s guidance documents are designed to represent the current thinking of the FDA on a particular topic, said Dr. Wilkin. The guidance documents are written to be generally applicable, and are not binding. “Other approaches may be used as long as they comply with governing statutes and regulations,” he said at the meeting.
In addition, it may even be possible that under some circumstances FDA guidance documents may be deemed inappropriate, and the FDA reserves the right to adopt a different approach if the circumstances require it, he added.
Clinicians need not stay on the sidelines when it comes to guidance documents, said Dr. Wilkin, who highlighted several ways dermatologists can get involved in the process:
• Offer input on works in progress: Clinicians can provide input on FDA guidance documents under development.
• Propose something new: Clinicians can make a case for why a guidance document is needed in a particular area.
• Draft your own: Clinicians can even submit drafts of their proposed guidance documents for consideration by the FDA. To submit a draft for the FDA’s consideration, mark the document “Guidance Document Submission,” and send it to: Division of Dockets Management (HFA-305), 5630 Fishers Lane, Room 1061, Rockville, MD, 20852.
• Suggest a revision: Clinicians can, at any time, suggest that the FDA revise or withdraw an already existing guidance document. The suggestion should address why the revision or withdrawal is needed, and how it should be revised, if applicable.
• Leave a comment: Once a year, the FDA publishes a list of potential topics for guidance document development or revision in the next year. The list is published in the Federal Register and online. Clinicians can comment on the list by proposing alternative topics or making recommendations related to the topics being considered.
“To participate in the development and issuance of guidance documents through one of the mechanisms described, contact the center or office that is responsible for the regulatory activity covered by the guidance document,” Dr. Wilkin said.
Global Academy and this news organization are owned by the same parent company. Dr. Wilkin had no relevant financial conflicts to disclose.
This article was updated 3/21/16.
The Food and Drug Administration wants to become more transparent and informative in its drug regulatory decision making, Dr. Jonathan Wilkin said at the Caribbean Dermatology Symposium.
“Part of this approach is the structured benefit-risk assessment tool,” he said in an interview.
The Prescription Drug User Fee Act (PDUFA) is reauthorized every 5 years, most recently in 2013, Dr. Wilkin said at the meeting, provided by Global Academy for Medical Education.
Two notable changes to PDUFA V (the fifth authorization of PDUFA) from previous versions are a focus on getting input from patients and their families on what constitutes a clinically meaningful improvement, and the publication of the FDA’s draft document on Structured Benefit-Risk Assessment, which is written for FDA staff and regulated drug manufacturers, explained Dr. Wilkin, a dermatologist in the Cayman Islands.
These changes are playing out with specific steps to increase patient participation and informing into the FDA’s decision-making process, said Dr. Wilkin, a former director of the Food and Drug Administration’s Division of Dermatologic and Dental Drug Products.
“Patients, their families, and patient advocacy groups are being invited to attend meetings with FDA to discuss patient needs in specific diseases, such as psoriasis and alopecia areata. This is expected to lead to perspectives on the benefit-risk assessments by the patients, for example, what risks they are willing to accept for what level of direct benefit, and their relationship to patient-reported outcome measures in clinical trials,” he said. “In the future, these patient reported outcome measures may be in package inserts and could be useful in physicians' discussions of therapeutic options with patients.”
The FDA’s guidance documents are designed to represent the current thinking of the FDA on a particular topic, said Dr. Wilkin. The guidance documents are written to be generally applicable, and are not binding. “Other approaches may be used as long as they comply with governing statutes and regulations,” he said at the meeting.
In addition, it may even be possible that under some circumstances FDA guidance documents may be deemed inappropriate, and the FDA reserves the right to adopt a different approach if the circumstances require it, he added.
Clinicians need not stay on the sidelines when it comes to guidance documents, said Dr. Wilkin, who highlighted several ways dermatologists can get involved in the process:
• Offer input on works in progress: Clinicians can provide input on FDA guidance documents under development.
• Propose something new: Clinicians can make a case for why a guidance document is needed in a particular area.
• Draft your own: Clinicians can even submit drafts of their proposed guidance documents for consideration by the FDA. To submit a draft for the FDA’s consideration, mark the document “Guidance Document Submission,” and send it to: Division of Dockets Management (HFA-305), 5630 Fishers Lane, Room 1061, Rockville, MD, 20852.
• Suggest a revision: Clinicians can, at any time, suggest that the FDA revise or withdraw an already existing guidance document. The suggestion should address why the revision or withdrawal is needed, and how it should be revised, if applicable.
• Leave a comment: Once a year, the FDA publishes a list of potential topics for guidance document development or revision in the next year. The list is published in the Federal Register and online. Clinicians can comment on the list by proposing alternative topics or making recommendations related to the topics being considered.
“To participate in the development and issuance of guidance documents through one of the mechanisms described, contact the center or office that is responsible for the regulatory activity covered by the guidance document,” Dr. Wilkin said.
Global Academy and this news organization are owned by the same parent company. Dr. Wilkin had no relevant financial conflicts to disclose.
This article was updated 3/21/16.
The Food and Drug Administration wants to become more transparent and informative in its drug regulatory decision making, Dr. Jonathan Wilkin said at the Caribbean Dermatology Symposium.
“Part of this approach is the structured benefit-risk assessment tool,” he said in an interview.
The Prescription Drug User Fee Act (PDUFA) is reauthorized every 5 years, most recently in 2013, Dr. Wilkin said at the meeting, provided by Global Academy for Medical Education.
Two notable changes to PDUFA V (the fifth authorization of PDUFA) from previous versions are a focus on getting input from patients and their families on what constitutes a clinically meaningful improvement, and the publication of the FDA’s draft document on Structured Benefit-Risk Assessment, which is written for FDA staff and regulated drug manufacturers, explained Dr. Wilkin, a dermatologist in the Cayman Islands.
These changes are playing out with specific steps to increase patient participation and informing into the FDA’s decision-making process, said Dr. Wilkin, a former director of the Food and Drug Administration’s Division of Dermatologic and Dental Drug Products.
“Patients, their families, and patient advocacy groups are being invited to attend meetings with FDA to discuss patient needs in specific diseases, such as psoriasis and alopecia areata. This is expected to lead to perspectives on the benefit-risk assessments by the patients, for example, what risks they are willing to accept for what level of direct benefit, and their relationship to patient-reported outcome measures in clinical trials,” he said. “In the future, these patient reported outcome measures may be in package inserts and could be useful in physicians' discussions of therapeutic options with patients.”
The FDA’s guidance documents are designed to represent the current thinking of the FDA on a particular topic, said Dr. Wilkin. The guidance documents are written to be generally applicable, and are not binding. “Other approaches may be used as long as they comply with governing statutes and regulations,” he said at the meeting.
In addition, it may even be possible that under some circumstances FDA guidance documents may be deemed inappropriate, and the FDA reserves the right to adopt a different approach if the circumstances require it, he added.
Clinicians need not stay on the sidelines when it comes to guidance documents, said Dr. Wilkin, who highlighted several ways dermatologists can get involved in the process:
• Offer input on works in progress: Clinicians can provide input on FDA guidance documents under development.
• Propose something new: Clinicians can make a case for why a guidance document is needed in a particular area.
• Draft your own: Clinicians can even submit drafts of their proposed guidance documents for consideration by the FDA. To submit a draft for the FDA’s consideration, mark the document “Guidance Document Submission,” and send it to: Division of Dockets Management (HFA-305), 5630 Fishers Lane, Room 1061, Rockville, MD, 20852.
• Suggest a revision: Clinicians can, at any time, suggest that the FDA revise or withdraw an already existing guidance document. The suggestion should address why the revision or withdrawal is needed, and how it should be revised, if applicable.
• Leave a comment: Once a year, the FDA publishes a list of potential topics for guidance document development or revision in the next year. The list is published in the Federal Register and online. Clinicians can comment on the list by proposing alternative topics or making recommendations related to the topics being considered.
“To participate in the development and issuance of guidance documents through one of the mechanisms described, contact the center or office that is responsible for the regulatory activity covered by the guidance document,” Dr. Wilkin said.
Global Academy and this news organization are owned by the same parent company. Dr. Wilkin had no relevant financial conflicts to disclose.
This article was updated 3/21/16.
EXPERT ANALYSIS FROM THE CARIBBEAN DERMATOLOGY SYMPOSIUM