User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'main-prefix')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
div[contains(@class, 'view-medstat-quiz-listing-panes')]
div[contains(@class, 'pane-article-sidebar-latest-news')]
FDA approves dapagliflozin (Farxiga) for chronic kidney disease
The Food and Drug Administration has approved dapagliflozin (Farxiga, AstraZeneca) to reduce the risk for kidney function decline, kidney failure, cardiovascular death, and hospitalization for heart failure in adult patients with chronic kidney disease (CKD) at risk for disease progression.
“Chronic kidney disease is an important public health issue, and there is a significant unmet need for therapies that slow disease progression and improve outcomes,” said Aliza Thompson, MD, deputy director of the division of cardiology and nephrology at the FDA’s Center for Drug Evaluation and Research. “Today’s approval of Farxiga for the treatment of chronic kidney disease is an important step forward in helping people living with kidney disease.”
Dapagliflozin was approved in 2014 to improve glycemic control in patients with diabetes mellitus, and approval was expanded in 2020 to include treatment of patients with heart failure and reduced ejection fraction, based on results of the DAPA-HF trial.
This new approval in chronic kidney disease was based on results of the DAPA-CKD trial that was stopped early in March 2020 because of efficacy of the treatment.
DAPA-CKD randomly assigned 4,304 patients with CKD but without diabetes to receive either dapagliflozin or placebo. The full study results, reported at the 2020 annual congress of the European Society of Cardiology and simultaneously published in the New England Journal of Medicine, showed that, during a median of 2.4 years, treatment with dapagliflozin led to a significant 31% relative reduction, compared with placebo in the study’s primary outcome, a composite that included at least a 50% drop in estimated glomerular filtration rate, compared with baseline, end-stage kidney disease, kidney transplant, renal death, or cardiovascular death.
Dapagliflozin treatment also cut all-cause mortality by a statistically significant relative reduction of 31%, and another secondary-endpoint analysis showed a statistically significant 29% relative reduction in the rate of cardiovascular death or heart failure hospitalization.
“Farxiga was not studied, nor is expected to be effective, in treating chronic kidney disease among patients with autosomal dominant or recessive polycystic (characterized by multiple cysts) kidney disease or among patients who require or have recently used immunosuppressive therapy to treat kidney disease,” the FDA statement noted.
Dapagliflozin should not be used by patients with a history of serious hypersensitivity reactions to this medication, or who are on dialysis, the agency added. “Serious, life-threatening cases of Fournier’s Gangrene have occurred in patients with diabetes taking Farxiga.”
Patients should consider taking a lower dose of insulin or insulin secretagogue to reduce hypoglycemic risk if they are also taking dapagliflozin. Treatment can also cause dehydration, serious urinary tract infections, genital yeast infections, and metabolic acidosis, the announcement said. “Patients should be assessed for their volume status and kidney function before starting Farxiga.”
Dapagliflozin previously received Fast Track, Breakthrough Therapy, and Priority Review designations for this new indication.
A version of this article first appeared on Medscape.com.
The Food and Drug Administration has approved dapagliflozin (Farxiga, AstraZeneca) to reduce the risk for kidney function decline, kidney failure, cardiovascular death, and hospitalization for heart failure in adult patients with chronic kidney disease (CKD) at risk for disease progression.
“Chronic kidney disease is an important public health issue, and there is a significant unmet need for therapies that slow disease progression and improve outcomes,” said Aliza Thompson, MD, deputy director of the division of cardiology and nephrology at the FDA’s Center for Drug Evaluation and Research. “Today’s approval of Farxiga for the treatment of chronic kidney disease is an important step forward in helping people living with kidney disease.”
Dapagliflozin was approved in 2014 to improve glycemic control in patients with diabetes mellitus, and approval was expanded in 2020 to include treatment of patients with heart failure and reduced ejection fraction, based on results of the DAPA-HF trial.
This new approval in chronic kidney disease was based on results of the DAPA-CKD trial that was stopped early in March 2020 because of efficacy of the treatment.
DAPA-CKD randomly assigned 4,304 patients with CKD but without diabetes to receive either dapagliflozin or placebo. The full study results, reported at the 2020 annual congress of the European Society of Cardiology and simultaneously published in the New England Journal of Medicine, showed that, during a median of 2.4 years, treatment with dapagliflozin led to a significant 31% relative reduction, compared with placebo in the study’s primary outcome, a composite that included at least a 50% drop in estimated glomerular filtration rate, compared with baseline, end-stage kidney disease, kidney transplant, renal death, or cardiovascular death.
Dapagliflozin treatment also cut all-cause mortality by a statistically significant relative reduction of 31%, and another secondary-endpoint analysis showed a statistically significant 29% relative reduction in the rate of cardiovascular death or heart failure hospitalization.
“Farxiga was not studied, nor is expected to be effective, in treating chronic kidney disease among patients with autosomal dominant or recessive polycystic (characterized by multiple cysts) kidney disease or among patients who require or have recently used immunosuppressive therapy to treat kidney disease,” the FDA statement noted.
Dapagliflozin should not be used by patients with a history of serious hypersensitivity reactions to this medication, or who are on dialysis, the agency added. “Serious, life-threatening cases of Fournier’s Gangrene have occurred in patients with diabetes taking Farxiga.”
Patients should consider taking a lower dose of insulin or insulin secretagogue to reduce hypoglycemic risk if they are also taking dapagliflozin. Treatment can also cause dehydration, serious urinary tract infections, genital yeast infections, and metabolic acidosis, the announcement said. “Patients should be assessed for their volume status and kidney function before starting Farxiga.”
Dapagliflozin previously received Fast Track, Breakthrough Therapy, and Priority Review designations for this new indication.
A version of this article first appeared on Medscape.com.
The Food and Drug Administration has approved dapagliflozin (Farxiga, AstraZeneca) to reduce the risk for kidney function decline, kidney failure, cardiovascular death, and hospitalization for heart failure in adult patients with chronic kidney disease (CKD) at risk for disease progression.
“Chronic kidney disease is an important public health issue, and there is a significant unmet need for therapies that slow disease progression and improve outcomes,” said Aliza Thompson, MD, deputy director of the division of cardiology and nephrology at the FDA’s Center for Drug Evaluation and Research. “Today’s approval of Farxiga for the treatment of chronic kidney disease is an important step forward in helping people living with kidney disease.”
Dapagliflozin was approved in 2014 to improve glycemic control in patients with diabetes mellitus, and approval was expanded in 2020 to include treatment of patients with heart failure and reduced ejection fraction, based on results of the DAPA-HF trial.
This new approval in chronic kidney disease was based on results of the DAPA-CKD trial that was stopped early in March 2020 because of efficacy of the treatment.
DAPA-CKD randomly assigned 4,304 patients with CKD but without diabetes to receive either dapagliflozin or placebo. The full study results, reported at the 2020 annual congress of the European Society of Cardiology and simultaneously published in the New England Journal of Medicine, showed that, during a median of 2.4 years, treatment with dapagliflozin led to a significant 31% relative reduction, compared with placebo in the study’s primary outcome, a composite that included at least a 50% drop in estimated glomerular filtration rate, compared with baseline, end-stage kidney disease, kidney transplant, renal death, or cardiovascular death.
Dapagliflozin treatment also cut all-cause mortality by a statistically significant relative reduction of 31%, and another secondary-endpoint analysis showed a statistically significant 29% relative reduction in the rate of cardiovascular death or heart failure hospitalization.
“Farxiga was not studied, nor is expected to be effective, in treating chronic kidney disease among patients with autosomal dominant or recessive polycystic (characterized by multiple cysts) kidney disease or among patients who require or have recently used immunosuppressive therapy to treat kidney disease,” the FDA statement noted.
Dapagliflozin should not be used by patients with a history of serious hypersensitivity reactions to this medication, or who are on dialysis, the agency added. “Serious, life-threatening cases of Fournier’s Gangrene have occurred in patients with diabetes taking Farxiga.”
Patients should consider taking a lower dose of insulin or insulin secretagogue to reduce hypoglycemic risk if they are also taking dapagliflozin. Treatment can also cause dehydration, serious urinary tract infections, genital yeast infections, and metabolic acidosis, the announcement said. “Patients should be assessed for their volume status and kidney function before starting Farxiga.”
Dapagliflozin previously received Fast Track, Breakthrough Therapy, and Priority Review designations for this new indication.
A version of this article first appeared on Medscape.com.
AHA issues new advice on managing stage 1 hypertension
Clinicians should consider the use of medication for adults with untreated stage 1 hypertension (130-139/80-89 mm Hg) whose 10-year risk for atherosclerotic cardiovascular disease is <10% and who fail to meet the blood pressure goal of <130/80 mm Hg after 6 months of guideline-based lifestyle therapy, the American Heart Association (AHA) advises in new scientific statement.
The statement was published online April 29 in Hypertension.
The recommendation complements the 2017 American College of Cardiology/American Heart Association Blood Pressure Management Guidelines, which do not fully address how to manage untreated stage 1 hypertension, the AHA says.
“There are no treatment recommendations in current guidelines for patients who are at relatively low short-term risk of heart disease when blood pressure does not drop below 130 mm Hg after six months of recommended lifestyle changes. This statement fills that gap,” Daniel W. Jones, MD, chair of the statement writing group and a past president of the AHA, said in a news release.
If after 6 months with lifestyle changes, blood pressure does not improve, lifestyle therapy should be continued and “clinicians should consider adding medications to control blood pressure,” said Dr. Jones, professor and dean emeritus, University of Mississippi, Jackson.
Healthy lifestyle changes to lower blood pressure include achieving ideal body weight, exercising (30 min of moderate to vigorous physical activity on most days, if possible), limiting dietary sodium, enhancing potassium intake, and following the Dietary Approaches to Stop Hypertension (DASH) diet, which is plentiful in fruits and vegetables with low-fat dairy products and reduced saturated fat and total fat. In addition, patients should be advised to limit alcohol intake and to not smoke.
The writing group acknowledges that these goals can be hard to achieve and maintain over time.
“It is very hard in America and most industrialized countries to limit sodium sufficiently to lower blood pressure, and it is difficult for all of us to maintain a healthy weight in what I refer to as a toxic food environment,” Dr. Jones said.
“We want clinicians to advise patients to take healthy lifestyle changes seriously and do their best. We certainly prefer to achieve blood pressure goals without adding medication; however, successfully treating high blood pressure does extend both years and quality of life,” said Dr. Jones.
The AHA statement also addresses cases in which adults were found to have hypertension during adolescence or childhood and were prescribed antihypertensive drug therapy.
In this patient population, clinicians should consider the original indications for starting antihypertensive drug treatment and the need to continue the medication and lifestyle therapy as young adults, the AHA advises.
“In young adults with stage 1 hypertension who are not controlled with lifestyle therapy, special consideration should be given to use of antihypertensive medication in individuals with a family history of premature CVD, a history of hypertension during pregnancy, or a personal history of premature birth,” the AHA states.
The scientific statement was prepared by the volunteer writing group on behalf of the AHA Council on Hypertension; the Council on the Kidney in Cardiovascular Disease; the Council on Arteriosclerosis, Thrombosis and Vascular Biology; the Council on Cardiovascular Radiology and Intervention; the Council on Lifelong Congenital Heart Disease and Heart Health in the Young; and the Stroke Council.
The authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Clinicians should consider the use of medication for adults with untreated stage 1 hypertension (130-139/80-89 mm Hg) whose 10-year risk for atherosclerotic cardiovascular disease is <10% and who fail to meet the blood pressure goal of <130/80 mm Hg after 6 months of guideline-based lifestyle therapy, the American Heart Association (AHA) advises in new scientific statement.
The statement was published online April 29 in Hypertension.
The recommendation complements the 2017 American College of Cardiology/American Heart Association Blood Pressure Management Guidelines, which do not fully address how to manage untreated stage 1 hypertension, the AHA says.
“There are no treatment recommendations in current guidelines for patients who are at relatively low short-term risk of heart disease when blood pressure does not drop below 130 mm Hg after six months of recommended lifestyle changes. This statement fills that gap,” Daniel W. Jones, MD, chair of the statement writing group and a past president of the AHA, said in a news release.
If after 6 months with lifestyle changes, blood pressure does not improve, lifestyle therapy should be continued and “clinicians should consider adding medications to control blood pressure,” said Dr. Jones, professor and dean emeritus, University of Mississippi, Jackson.
Healthy lifestyle changes to lower blood pressure include achieving ideal body weight, exercising (30 min of moderate to vigorous physical activity on most days, if possible), limiting dietary sodium, enhancing potassium intake, and following the Dietary Approaches to Stop Hypertension (DASH) diet, which is plentiful in fruits and vegetables with low-fat dairy products and reduced saturated fat and total fat. In addition, patients should be advised to limit alcohol intake and to not smoke.
The writing group acknowledges that these goals can be hard to achieve and maintain over time.
“It is very hard in America and most industrialized countries to limit sodium sufficiently to lower blood pressure, and it is difficult for all of us to maintain a healthy weight in what I refer to as a toxic food environment,” Dr. Jones said.
“We want clinicians to advise patients to take healthy lifestyle changes seriously and do their best. We certainly prefer to achieve blood pressure goals without adding medication; however, successfully treating high blood pressure does extend both years and quality of life,” said Dr. Jones.
The AHA statement also addresses cases in which adults were found to have hypertension during adolescence or childhood and were prescribed antihypertensive drug therapy.
In this patient population, clinicians should consider the original indications for starting antihypertensive drug treatment and the need to continue the medication and lifestyle therapy as young adults, the AHA advises.
“In young adults with stage 1 hypertension who are not controlled with lifestyle therapy, special consideration should be given to use of antihypertensive medication in individuals with a family history of premature CVD, a history of hypertension during pregnancy, or a personal history of premature birth,” the AHA states.
The scientific statement was prepared by the volunteer writing group on behalf of the AHA Council on Hypertension; the Council on the Kidney in Cardiovascular Disease; the Council on Arteriosclerosis, Thrombosis and Vascular Biology; the Council on Cardiovascular Radiology and Intervention; the Council on Lifelong Congenital Heart Disease and Heart Health in the Young; and the Stroke Council.
The authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Clinicians should consider the use of medication for adults with untreated stage 1 hypertension (130-139/80-89 mm Hg) whose 10-year risk for atherosclerotic cardiovascular disease is <10% and who fail to meet the blood pressure goal of <130/80 mm Hg after 6 months of guideline-based lifestyle therapy, the American Heart Association (AHA) advises in new scientific statement.
The statement was published online April 29 in Hypertension.
The recommendation complements the 2017 American College of Cardiology/American Heart Association Blood Pressure Management Guidelines, which do not fully address how to manage untreated stage 1 hypertension, the AHA says.
“There are no treatment recommendations in current guidelines for patients who are at relatively low short-term risk of heart disease when blood pressure does not drop below 130 mm Hg after six months of recommended lifestyle changes. This statement fills that gap,” Daniel W. Jones, MD, chair of the statement writing group and a past president of the AHA, said in a news release.
If after 6 months with lifestyle changes, blood pressure does not improve, lifestyle therapy should be continued and “clinicians should consider adding medications to control blood pressure,” said Dr. Jones, professor and dean emeritus, University of Mississippi, Jackson.
Healthy lifestyle changes to lower blood pressure include achieving ideal body weight, exercising (30 min of moderate to vigorous physical activity on most days, if possible), limiting dietary sodium, enhancing potassium intake, and following the Dietary Approaches to Stop Hypertension (DASH) diet, which is plentiful in fruits and vegetables with low-fat dairy products and reduced saturated fat and total fat. In addition, patients should be advised to limit alcohol intake and to not smoke.
The writing group acknowledges that these goals can be hard to achieve and maintain over time.
“It is very hard in America and most industrialized countries to limit sodium sufficiently to lower blood pressure, and it is difficult for all of us to maintain a healthy weight in what I refer to as a toxic food environment,” Dr. Jones said.
“We want clinicians to advise patients to take healthy lifestyle changes seriously and do their best. We certainly prefer to achieve blood pressure goals without adding medication; however, successfully treating high blood pressure does extend both years and quality of life,” said Dr. Jones.
The AHA statement also addresses cases in which adults were found to have hypertension during adolescence or childhood and were prescribed antihypertensive drug therapy.
In this patient population, clinicians should consider the original indications for starting antihypertensive drug treatment and the need to continue the medication and lifestyle therapy as young adults, the AHA advises.
“In young adults with stage 1 hypertension who are not controlled with lifestyle therapy, special consideration should be given to use of antihypertensive medication in individuals with a family history of premature CVD, a history of hypertension during pregnancy, or a personal history of premature birth,” the AHA states.
The scientific statement was prepared by the volunteer writing group on behalf of the AHA Council on Hypertension; the Council on the Kidney in Cardiovascular Disease; the Council on Arteriosclerosis, Thrombosis and Vascular Biology; the Council on Cardiovascular Radiology and Intervention; the Council on Lifelong Congenital Heart Disease and Heart Health in the Young; and the Stroke Council.
The authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Doctors lose jobs after speaking out about unsafe conditions
In April 2020, hospitalist Samantha Houston, MD, lost her job at Baptist Memorial Hospital–North, in Oxford, Miss., after she publicly campaigned to get donations of N95 masks for nurses. Dr. Houston filed a lawsuit against the hospital, saying she was improperly fired for speaking out. The lawsuit has not yet gone to trial.
In January 2017, emergency physician Raymond Brovont, MD, was fired by EmCare, an emergency physician staffing company, after reporting understaffing at hospitals with which it contracted in the Kansas City, Mo., area. Dr. Brovont sued EmCare, and the company lost the case. In February 2019, it was ordered to pay him $13.1 million in damages.
These are just two of several cases in recent years in which physicians have spoken out about problems involving patient care and have been sanctioned. Other physicians who see problems choose to stay silent.
Doctors often hesitate to speak out because of the prospect of losing their jobs. A 2013 study of emergency physicians found that nearly 20% reported a possible or real threat to their employment if they expressed concerns about quality of care.
When physicians do not speak openly about important medical issues, the quality of care in their institutions suffers, said a coauthor of the study, Larry D. Weiss, MD, JD, a retired professor of emergency medicine at the University of Maryland, Baltimore.
“Physicians can’t effectively represent patients if they are always thinking they can get fired for what they say,” Dr. Weiss said. “If you don’t have protections like due process, which is often the case, you are less likely to speak out.”
In the first few weeks, health care facilities were struggling to obtain personal protective equipment (PPE) and to create policies that would keep patients and caregivers safe.
Physicians such as Dr. Houston took the initiative to make sure their institutions were taking the right steps against COVID-19 and found themselves at loggerheads with administrators who were concerned that their organizations were being portrayed as unsafe.
The case of one physician who spoke out
One of the highest-profile cases of a physician speaking out and being removed from work during the pandemic is that of Ming Lin, MD, an emergency physician who lost a job he had held for 17 years at St. Joseph Medical Center, in Bellingham, Wash. Dr. Lin lost his job after he made a series of Facebook posts that criticized the hospital’s COVID-19 preparedness efforts.
In an interview, Dr. Lin discussed the details of his situation to a degree that rarely occurs in such cases. This is one of the most extensive interviews he has granted.
Postings on Facebook
Dr. Lin said that on the basis of an intense study of the virus at the onset of the pandemic, he developed many ideas as to what could be done to mitigate its spread. While working as a locum tenens physician on his time off, he could see how others dealt with COVID-19.
Dr. Lin said from past experiences he did not feel that he could present his ideas directly to administration and be heard, so he decided to air his ideas about how his hospital could handle COVID-19 on his Facebook page, which drew a large audience.
He said he was certain that hospital administrators were reading his posts. He said receptionists at this hospital were advised not to wear masks, evidently because it would alarm patients. Dr. Lin said he posted concerns about their safety and called for them to wear masks. Soon after, the hospital directed receptionists to wear masks.
Dr. Lin’s Facebook posts also criticized the hospital for taking what he felt was too long to get results on COVID-19 tests. “It was taking them up to 10 days to get test results, because samples were being sent to a lab in California,” he said. He suggested it would be faster to send samples to the University of Washington. Soon after, the hospital started sending samples there.
In just a couple of weeks, Dr. Lin said, he voiced almost a dozen concerns. Each time the hospital made changes in line with his recommendations. Although he didn’t get any direct acknowledgment from the hospital for his help, he said he felt he was making a positive impact.
How employers react to physicians who speak out
Physicians who speak out about conditions tend to deeply disturb administrators, said William P. Sullivan, DO, JD, an emergency physician and lawyer in Frankfort, Ill., who has written about physicians being terminated by hospitals.
“These physicians go to the news media or they use social media,” Dr. Sullivan said, “but hospital administrators don’t want the public to hear bad things about their hospital.”
Then the public might not come to the hospital, which is an administrator’s worst nightmare. Even if physicians think their criticisms are reasonable, administrators may still fear a resulting drop in patients.
Dr. Houston, for example, was helping her Mississippi hospital by collecting donations of N95 masks for nurses, but to administrators, it showed that the hospital did not have enough masks.
“It is not helpful to stoke fear and anxiety, even if the intent is sincere,” a spokesperson for the hospital said.
Administrator fires back
Dr. Lin’s posts were deeply concerning to Richard DeCarlo, chief operating officer of PeaceHealth, which runs St. Joseph Hospital. Mr. DeCarlo discussed his concerns in a video interview in April with the blogger Zubin Damania, MD, known as ZDoggMD.
Comments on Dr. Lin’s Facebook posts showed that people “were fearful to go to the hospital,” he told Dr. Damania. “They were concluding that they would need to drive to another hospital.”
Mr. DeCarlo said he was also unhappy that Dr. Lin did not directly contact administrators about his concerns. “He didn’t communicate with his medical director,” Mr. DeCarlo said in the interview. “The ED staff had been meeting three times a week with the chief medical officer to make sure they had everything they needed, but he only attended one of these meetings and didn’t ask any questions.”
Dr. Lin maintains he did ask questions at the first meeting but stopped attending because he felt he wasn’t being heeded. “I found their tone not very receptive,” he said.
Doctor allegedly offered “misinformation”
At the start of the pandemic, some hospitals made it clear what would happen to doctors who brought up lack of PPE or other problems to the media. For example, NYU Langone Medical Center in New York sent an email to staff warning that speaking to the media without permission “will be subject to disciplinary action, including termination.”
PeaceHealth took a different tack. “It’s not that we have a policy that says don’t ever talk to the media,” Mr. DeCarlo said in the ZDoggMD interview, but in Dr. Lin’s case, “what was at issue was the misinformation. His leader went to him and said, ‘Look, you’re posting things that aren’t accurate.’ ”
Dr. Lin disputes that he provided any misinformation. In the interview, Mr. DeCarlo cited just one example of alleged misinformation. He said Dr. Lin called for a tent outside the emergency department (ED) to protect patients entering the department from aerosol exposure to COVID-19. Mr. DeCarlo said the tent was not needed because fewer people were using the ED.
“To put it in an extreme way,” Mr. DeCarlo said of Dr. Lin’s posts, “it was like yelling fire in a theater where there is not a fire.”
Dr. Lin said the hospital did briefly erect a tent and then removed it, and he still insisted that a tent was a good idea. He added that Mr. DeCarlo never mentioned any of the other suggestions Dr. Lin made, nor did he state that the hospital adopted them.
Doctor gets a warning
Dr. Lin said that after he started posting his concerns, he got a call from the emergency department director who worked for TeamHealth, an emergency medicine staffing firm that contracted with PeaceHealth and employed Dr. Lin, too.
Dr. Lin said his immediate supervisor at TeamHealth told him the hospital was unhappy with his posts and that he should take them down and suggested he might be fired. Dr. Lin said the supervisor also asked him to apologize to the hospital administration for these posts, but he refused to do so.
“Retracting and apologizing was not only wrong but would have left me vulnerable to being terminated with no repercussions,” he said.
“At that point, I realized I had crossed the Rubicon,” Dr. Lin said. He thought he might well be fired, no matter what he did, so he took his story to The Seattle Times, which had a much wider platform than his Facebook page had.
Dr. Lin lost his job at St. Joseph a week after The Seattle Times story about him appeared. “About 10 minutes before my shift was supposed to start, I received a text message from TeamHealth saying that someone else would be taking the shift,” he said.
In a release, TeamHealth insisted Dr. Lin was not fired and that he was scheduled to be reassigned to work at other hospitals. Dr. Lin, however, said he was not told this at the time and that he found out later that the new assignment would involve a pay cut and a significant commute. He said he has not taken any new assignments from TeamHealth since he lost his job at St. Joseph.
Dr. Lin has filed a lawsuit against PeaceHealth, TeamHealth, and Mr. DeCarlo, asking for his job back and for an apology. He said he has not asked for any financial damages at this point.
Since leaving St. Joseph, Dr. Lin has been working as an administrator for the Indian Health Service in the upper plains states. He said he can do some of the work at home in Washington State, which allows him to be with his wife and three young children.
Dr. Lin no longer sees patients. “I feel I have lost my confidence as a clinician,” he said. “I’m not sure why, but I find it hard to make quick judgments when taking care of patients.”
He said many doctors have told him about their own troubles with speaking out, but they did not want to come forward and talk about it because they feared more repercussions.
Do doctors who speak out have any rights?
Because TeamHealth, Dr. Lin’s actual employer, asserts he was never actually terminated, Dr. Lin has not been able to appeal his case internally in accordance with due process, an option that allows doctors to get a fair hearing and to appeal decisions against them.
The American Academy of Emergency Medicine pointed out this problem. “Dr. Lin, as a member of the medical staff, is entitled to full due process and a fair hearing from his peers on the medical staff,” the academy said in a statement supporting him.
The Joint Commission, the hospital accreditor, requires that hospitals provide due process to doctors before they can be terminated. However, Dr. Sullivan said employers often make physicians waive their due process rights in the employment contract. “The result is that the employer can terminate doctors for no reason,” he said.
In the 2013 survey of emergency physicians, 62% reported that their employers could terminate them without full due process.
Dr. Weiss, the Maryland MD-JD, said that when he advises doctors on their contracts, he generally tells them to cross out the waiver language. The applicant, he says, may also tell the employer that the waivers are considered unethical by many physician professional societies. In some cases, he said, the hospital will back down.
Conclusion
To maintain quality of care, it is essential that physicians feel free to speak out about issues that concern them. They can improve their chances of being heard by working directly with management and attending meetings, but in some cases, management may be unwilling to listen.
A version of this article first appeared on Medscape.com.
In April 2020, hospitalist Samantha Houston, MD, lost her job at Baptist Memorial Hospital–North, in Oxford, Miss., after she publicly campaigned to get donations of N95 masks for nurses. Dr. Houston filed a lawsuit against the hospital, saying she was improperly fired for speaking out. The lawsuit has not yet gone to trial.
In January 2017, emergency physician Raymond Brovont, MD, was fired by EmCare, an emergency physician staffing company, after reporting understaffing at hospitals with which it contracted in the Kansas City, Mo., area. Dr. Brovont sued EmCare, and the company lost the case. In February 2019, it was ordered to pay him $13.1 million in damages.
These are just two of several cases in recent years in which physicians have spoken out about problems involving patient care and have been sanctioned. Other physicians who see problems choose to stay silent.
Doctors often hesitate to speak out because of the prospect of losing their jobs. A 2013 study of emergency physicians found that nearly 20% reported a possible or real threat to their employment if they expressed concerns about quality of care.
When physicians do not speak openly about important medical issues, the quality of care in their institutions suffers, said a coauthor of the study, Larry D. Weiss, MD, JD, a retired professor of emergency medicine at the University of Maryland, Baltimore.
“Physicians can’t effectively represent patients if they are always thinking they can get fired for what they say,” Dr. Weiss said. “If you don’t have protections like due process, which is often the case, you are less likely to speak out.”
In the first few weeks, health care facilities were struggling to obtain personal protective equipment (PPE) and to create policies that would keep patients and caregivers safe.
Physicians such as Dr. Houston took the initiative to make sure their institutions were taking the right steps against COVID-19 and found themselves at loggerheads with administrators who were concerned that their organizations were being portrayed as unsafe.
The case of one physician who spoke out
One of the highest-profile cases of a physician speaking out and being removed from work during the pandemic is that of Ming Lin, MD, an emergency physician who lost a job he had held for 17 years at St. Joseph Medical Center, in Bellingham, Wash. Dr. Lin lost his job after he made a series of Facebook posts that criticized the hospital’s COVID-19 preparedness efforts.
In an interview, Dr. Lin discussed the details of his situation to a degree that rarely occurs in such cases. This is one of the most extensive interviews he has granted.
Postings on Facebook
Dr. Lin said that on the basis of an intense study of the virus at the onset of the pandemic, he developed many ideas as to what could be done to mitigate its spread. While working as a locum tenens physician on his time off, he could see how others dealt with COVID-19.
Dr. Lin said from past experiences he did not feel that he could present his ideas directly to administration and be heard, so he decided to air his ideas about how his hospital could handle COVID-19 on his Facebook page, which drew a large audience.
He said he was certain that hospital administrators were reading his posts. He said receptionists at this hospital were advised not to wear masks, evidently because it would alarm patients. Dr. Lin said he posted concerns about their safety and called for them to wear masks. Soon after, the hospital directed receptionists to wear masks.
Dr. Lin’s Facebook posts also criticized the hospital for taking what he felt was too long to get results on COVID-19 tests. “It was taking them up to 10 days to get test results, because samples were being sent to a lab in California,” he said. He suggested it would be faster to send samples to the University of Washington. Soon after, the hospital started sending samples there.
In just a couple of weeks, Dr. Lin said, he voiced almost a dozen concerns. Each time the hospital made changes in line with his recommendations. Although he didn’t get any direct acknowledgment from the hospital for his help, he said he felt he was making a positive impact.
How employers react to physicians who speak out
Physicians who speak out about conditions tend to deeply disturb administrators, said William P. Sullivan, DO, JD, an emergency physician and lawyer in Frankfort, Ill., who has written about physicians being terminated by hospitals.
“These physicians go to the news media or they use social media,” Dr. Sullivan said, “but hospital administrators don’t want the public to hear bad things about their hospital.”
Then the public might not come to the hospital, which is an administrator’s worst nightmare. Even if physicians think their criticisms are reasonable, administrators may still fear a resulting drop in patients.
Dr. Houston, for example, was helping her Mississippi hospital by collecting donations of N95 masks for nurses, but to administrators, it showed that the hospital did not have enough masks.
“It is not helpful to stoke fear and anxiety, even if the intent is sincere,” a spokesperson for the hospital said.
Administrator fires back
Dr. Lin’s posts were deeply concerning to Richard DeCarlo, chief operating officer of PeaceHealth, which runs St. Joseph Hospital. Mr. DeCarlo discussed his concerns in a video interview in April with the blogger Zubin Damania, MD, known as ZDoggMD.
Comments on Dr. Lin’s Facebook posts showed that people “were fearful to go to the hospital,” he told Dr. Damania. “They were concluding that they would need to drive to another hospital.”
Mr. DeCarlo said he was also unhappy that Dr. Lin did not directly contact administrators about his concerns. “He didn’t communicate with his medical director,” Mr. DeCarlo said in the interview. “The ED staff had been meeting three times a week with the chief medical officer to make sure they had everything they needed, but he only attended one of these meetings and didn’t ask any questions.”
Dr. Lin maintains he did ask questions at the first meeting but stopped attending because he felt he wasn’t being heeded. “I found their tone not very receptive,” he said.
Doctor allegedly offered “misinformation”
At the start of the pandemic, some hospitals made it clear what would happen to doctors who brought up lack of PPE or other problems to the media. For example, NYU Langone Medical Center in New York sent an email to staff warning that speaking to the media without permission “will be subject to disciplinary action, including termination.”
PeaceHealth took a different tack. “It’s not that we have a policy that says don’t ever talk to the media,” Mr. DeCarlo said in the ZDoggMD interview, but in Dr. Lin’s case, “what was at issue was the misinformation. His leader went to him and said, ‘Look, you’re posting things that aren’t accurate.’ ”
Dr. Lin disputes that he provided any misinformation. In the interview, Mr. DeCarlo cited just one example of alleged misinformation. He said Dr. Lin called for a tent outside the emergency department (ED) to protect patients entering the department from aerosol exposure to COVID-19. Mr. DeCarlo said the tent was not needed because fewer people were using the ED.
“To put it in an extreme way,” Mr. DeCarlo said of Dr. Lin’s posts, “it was like yelling fire in a theater where there is not a fire.”
Dr. Lin said the hospital did briefly erect a tent and then removed it, and he still insisted that a tent was a good idea. He added that Mr. DeCarlo never mentioned any of the other suggestions Dr. Lin made, nor did he state that the hospital adopted them.
Doctor gets a warning
Dr. Lin said that after he started posting his concerns, he got a call from the emergency department director who worked for TeamHealth, an emergency medicine staffing firm that contracted with PeaceHealth and employed Dr. Lin, too.
Dr. Lin said his immediate supervisor at TeamHealth told him the hospital was unhappy with his posts and that he should take them down and suggested he might be fired. Dr. Lin said the supervisor also asked him to apologize to the hospital administration for these posts, but he refused to do so.
“Retracting and apologizing was not only wrong but would have left me vulnerable to being terminated with no repercussions,” he said.
“At that point, I realized I had crossed the Rubicon,” Dr. Lin said. He thought he might well be fired, no matter what he did, so he took his story to The Seattle Times, which had a much wider platform than his Facebook page had.
Dr. Lin lost his job at St. Joseph a week after The Seattle Times story about him appeared. “About 10 minutes before my shift was supposed to start, I received a text message from TeamHealth saying that someone else would be taking the shift,” he said.
In a release, TeamHealth insisted Dr. Lin was not fired and that he was scheduled to be reassigned to work at other hospitals. Dr. Lin, however, said he was not told this at the time and that he found out later that the new assignment would involve a pay cut and a significant commute. He said he has not taken any new assignments from TeamHealth since he lost his job at St. Joseph.
Dr. Lin has filed a lawsuit against PeaceHealth, TeamHealth, and Mr. DeCarlo, asking for his job back and for an apology. He said he has not asked for any financial damages at this point.
Since leaving St. Joseph, Dr. Lin has been working as an administrator for the Indian Health Service in the upper plains states. He said he can do some of the work at home in Washington State, which allows him to be with his wife and three young children.
Dr. Lin no longer sees patients. “I feel I have lost my confidence as a clinician,” he said. “I’m not sure why, but I find it hard to make quick judgments when taking care of patients.”
He said many doctors have told him about their own troubles with speaking out, but they did not want to come forward and talk about it because they feared more repercussions.
Do doctors who speak out have any rights?
Because TeamHealth, Dr. Lin’s actual employer, asserts he was never actually terminated, Dr. Lin has not been able to appeal his case internally in accordance with due process, an option that allows doctors to get a fair hearing and to appeal decisions against them.
The American Academy of Emergency Medicine pointed out this problem. “Dr. Lin, as a member of the medical staff, is entitled to full due process and a fair hearing from his peers on the medical staff,” the academy said in a statement supporting him.
The Joint Commission, the hospital accreditor, requires that hospitals provide due process to doctors before they can be terminated. However, Dr. Sullivan said employers often make physicians waive their due process rights in the employment contract. “The result is that the employer can terminate doctors for no reason,” he said.
In the 2013 survey of emergency physicians, 62% reported that their employers could terminate them without full due process.
Dr. Weiss, the Maryland MD-JD, said that when he advises doctors on their contracts, he generally tells them to cross out the waiver language. The applicant, he says, may also tell the employer that the waivers are considered unethical by many physician professional societies. In some cases, he said, the hospital will back down.
Conclusion
To maintain quality of care, it is essential that physicians feel free to speak out about issues that concern them. They can improve their chances of being heard by working directly with management and attending meetings, but in some cases, management may be unwilling to listen.
A version of this article first appeared on Medscape.com.
In April 2020, hospitalist Samantha Houston, MD, lost her job at Baptist Memorial Hospital–North, in Oxford, Miss., after she publicly campaigned to get donations of N95 masks for nurses. Dr. Houston filed a lawsuit against the hospital, saying she was improperly fired for speaking out. The lawsuit has not yet gone to trial.
In January 2017, emergency physician Raymond Brovont, MD, was fired by EmCare, an emergency physician staffing company, after reporting understaffing at hospitals with which it contracted in the Kansas City, Mo., area. Dr. Brovont sued EmCare, and the company lost the case. In February 2019, it was ordered to pay him $13.1 million in damages.
These are just two of several cases in recent years in which physicians have spoken out about problems involving patient care and have been sanctioned. Other physicians who see problems choose to stay silent.
Doctors often hesitate to speak out because of the prospect of losing their jobs. A 2013 study of emergency physicians found that nearly 20% reported a possible or real threat to their employment if they expressed concerns about quality of care.
When physicians do not speak openly about important medical issues, the quality of care in their institutions suffers, said a coauthor of the study, Larry D. Weiss, MD, JD, a retired professor of emergency medicine at the University of Maryland, Baltimore.
“Physicians can’t effectively represent patients if they are always thinking they can get fired for what they say,” Dr. Weiss said. “If you don’t have protections like due process, which is often the case, you are less likely to speak out.”
In the first few weeks, health care facilities were struggling to obtain personal protective equipment (PPE) and to create policies that would keep patients and caregivers safe.
Physicians such as Dr. Houston took the initiative to make sure their institutions were taking the right steps against COVID-19 and found themselves at loggerheads with administrators who were concerned that their organizations were being portrayed as unsafe.
The case of one physician who spoke out
One of the highest-profile cases of a physician speaking out and being removed from work during the pandemic is that of Ming Lin, MD, an emergency physician who lost a job he had held for 17 years at St. Joseph Medical Center, in Bellingham, Wash. Dr. Lin lost his job after he made a series of Facebook posts that criticized the hospital’s COVID-19 preparedness efforts.
In an interview, Dr. Lin discussed the details of his situation to a degree that rarely occurs in such cases. This is one of the most extensive interviews he has granted.
Postings on Facebook
Dr. Lin said that on the basis of an intense study of the virus at the onset of the pandemic, he developed many ideas as to what could be done to mitigate its spread. While working as a locum tenens physician on his time off, he could see how others dealt with COVID-19.
Dr. Lin said from past experiences he did not feel that he could present his ideas directly to administration and be heard, so he decided to air his ideas about how his hospital could handle COVID-19 on his Facebook page, which drew a large audience.
He said he was certain that hospital administrators were reading his posts. He said receptionists at this hospital were advised not to wear masks, evidently because it would alarm patients. Dr. Lin said he posted concerns about their safety and called for them to wear masks. Soon after, the hospital directed receptionists to wear masks.
Dr. Lin’s Facebook posts also criticized the hospital for taking what he felt was too long to get results on COVID-19 tests. “It was taking them up to 10 days to get test results, because samples were being sent to a lab in California,” he said. He suggested it would be faster to send samples to the University of Washington. Soon after, the hospital started sending samples there.
In just a couple of weeks, Dr. Lin said, he voiced almost a dozen concerns. Each time the hospital made changes in line with his recommendations. Although he didn’t get any direct acknowledgment from the hospital for his help, he said he felt he was making a positive impact.
How employers react to physicians who speak out
Physicians who speak out about conditions tend to deeply disturb administrators, said William P. Sullivan, DO, JD, an emergency physician and lawyer in Frankfort, Ill., who has written about physicians being terminated by hospitals.
“These physicians go to the news media or they use social media,” Dr. Sullivan said, “but hospital administrators don’t want the public to hear bad things about their hospital.”
Then the public might not come to the hospital, which is an administrator’s worst nightmare. Even if physicians think their criticisms are reasonable, administrators may still fear a resulting drop in patients.
Dr. Houston, for example, was helping her Mississippi hospital by collecting donations of N95 masks for nurses, but to administrators, it showed that the hospital did not have enough masks.
“It is not helpful to stoke fear and anxiety, even if the intent is sincere,” a spokesperson for the hospital said.
Administrator fires back
Dr. Lin’s posts were deeply concerning to Richard DeCarlo, chief operating officer of PeaceHealth, which runs St. Joseph Hospital. Mr. DeCarlo discussed his concerns in a video interview in April with the blogger Zubin Damania, MD, known as ZDoggMD.
Comments on Dr. Lin’s Facebook posts showed that people “were fearful to go to the hospital,” he told Dr. Damania. “They were concluding that they would need to drive to another hospital.”
Mr. DeCarlo said he was also unhappy that Dr. Lin did not directly contact administrators about his concerns. “He didn’t communicate with his medical director,” Mr. DeCarlo said in the interview. “The ED staff had been meeting three times a week with the chief medical officer to make sure they had everything they needed, but he only attended one of these meetings and didn’t ask any questions.”
Dr. Lin maintains he did ask questions at the first meeting but stopped attending because he felt he wasn’t being heeded. “I found their tone not very receptive,” he said.
Doctor allegedly offered “misinformation”
At the start of the pandemic, some hospitals made it clear what would happen to doctors who brought up lack of PPE or other problems to the media. For example, NYU Langone Medical Center in New York sent an email to staff warning that speaking to the media without permission “will be subject to disciplinary action, including termination.”
PeaceHealth took a different tack. “It’s not that we have a policy that says don’t ever talk to the media,” Mr. DeCarlo said in the ZDoggMD interview, but in Dr. Lin’s case, “what was at issue was the misinformation. His leader went to him and said, ‘Look, you’re posting things that aren’t accurate.’ ”
Dr. Lin disputes that he provided any misinformation. In the interview, Mr. DeCarlo cited just one example of alleged misinformation. He said Dr. Lin called for a tent outside the emergency department (ED) to protect patients entering the department from aerosol exposure to COVID-19. Mr. DeCarlo said the tent was not needed because fewer people were using the ED.
“To put it in an extreme way,” Mr. DeCarlo said of Dr. Lin’s posts, “it was like yelling fire in a theater where there is not a fire.”
Dr. Lin said the hospital did briefly erect a tent and then removed it, and he still insisted that a tent was a good idea. He added that Mr. DeCarlo never mentioned any of the other suggestions Dr. Lin made, nor did he state that the hospital adopted them.
Doctor gets a warning
Dr. Lin said that after he started posting his concerns, he got a call from the emergency department director who worked for TeamHealth, an emergency medicine staffing firm that contracted with PeaceHealth and employed Dr. Lin, too.
Dr. Lin said his immediate supervisor at TeamHealth told him the hospital was unhappy with his posts and that he should take them down and suggested he might be fired. Dr. Lin said the supervisor also asked him to apologize to the hospital administration for these posts, but he refused to do so.
“Retracting and apologizing was not only wrong but would have left me vulnerable to being terminated with no repercussions,” he said.
“At that point, I realized I had crossed the Rubicon,” Dr. Lin said. He thought he might well be fired, no matter what he did, so he took his story to The Seattle Times, which had a much wider platform than his Facebook page had.
Dr. Lin lost his job at St. Joseph a week after The Seattle Times story about him appeared. “About 10 minutes before my shift was supposed to start, I received a text message from TeamHealth saying that someone else would be taking the shift,” he said.
In a release, TeamHealth insisted Dr. Lin was not fired and that he was scheduled to be reassigned to work at other hospitals. Dr. Lin, however, said he was not told this at the time and that he found out later that the new assignment would involve a pay cut and a significant commute. He said he has not taken any new assignments from TeamHealth since he lost his job at St. Joseph.
Dr. Lin has filed a lawsuit against PeaceHealth, TeamHealth, and Mr. DeCarlo, asking for his job back and for an apology. He said he has not asked for any financial damages at this point.
Since leaving St. Joseph, Dr. Lin has been working as an administrator for the Indian Health Service in the upper plains states. He said he can do some of the work at home in Washington State, which allows him to be with his wife and three young children.
Dr. Lin no longer sees patients. “I feel I have lost my confidence as a clinician,” he said. “I’m not sure why, but I find it hard to make quick judgments when taking care of patients.”
He said many doctors have told him about their own troubles with speaking out, but they did not want to come forward and talk about it because they feared more repercussions.
Do doctors who speak out have any rights?
Because TeamHealth, Dr. Lin’s actual employer, asserts he was never actually terminated, Dr. Lin has not been able to appeal his case internally in accordance with due process, an option that allows doctors to get a fair hearing and to appeal decisions against them.
The American Academy of Emergency Medicine pointed out this problem. “Dr. Lin, as a member of the medical staff, is entitled to full due process and a fair hearing from his peers on the medical staff,” the academy said in a statement supporting him.
The Joint Commission, the hospital accreditor, requires that hospitals provide due process to doctors before they can be terminated. However, Dr. Sullivan said employers often make physicians waive their due process rights in the employment contract. “The result is that the employer can terminate doctors for no reason,” he said.
In the 2013 survey of emergency physicians, 62% reported that their employers could terminate them without full due process.
Dr. Weiss, the Maryland MD-JD, said that when he advises doctors on their contracts, he generally tells them to cross out the waiver language. The applicant, he says, may also tell the employer that the waivers are considered unethical by many physician professional societies. In some cases, he said, the hospital will back down.
Conclusion
To maintain quality of care, it is essential that physicians feel free to speak out about issues that concern them. They can improve their chances of being heard by working directly with management and attending meetings, but in some cases, management may be unwilling to listen.
A version of this article first appeared on Medscape.com.
AHA guidance on blood clots linked to COVID-19 vaccine
A newly released report is offering guidance concerning rare conditions associated with COVID-19 as well as vaccines against the virus.
The report was released April 29, 2021, by the American Heart Association/American Stroke Association Stroke Council Leadership in answer to the decision April 23 by the Centers for Disease Control and Prevention and the Food and Drug Administration to lift an earlier “pause” in administration of the Johnson & Johnson (Janssen) vaccine.
That pause had been put in place after reports were received of a possible association between the J&J vaccine and cerebral venous sinus thrombosis (CVST) and thrombosis-thrombocytopenia syndrome (TTS, blood clots plus low blood platelets). CVST and TTS were also linked to patients in Europe and Canada who received the AstraZeneca COVID-19 vaccine.
However, the new report noted that these conditions are very rare.
“The risk of CVST due to infection with COVID-19 is 8-10 times higher than the risk of CVST after receiving a COVID-19 vaccine,” lead author Karen L. Furie, MD, chair of the department of neurology at Brown University, Providence, R.I., said in a press release.
“The public can be reassured by the CDC’s and FDA’s investigation and these statistics – the likelihood of developing CVST after a COVID-19 vaccine is extremely low,” said Dr. Furie, adding that the authors “urge all adults to receive any of the approved COVID-19 vaccines.”
The new guidance, which was published online April 29, 2021, in Stroke, discusses signs and symptoms of CVST and TTS, as well as vaccine-induced immune thrombotic thrombocytopenia (VITT). It also recommends best options for treating these conditions.
Assessing 81 million patients
In their analysis, the investigators assessed a database of 59 health care organizations and 81 million patients, 98% of whom were in the United States.
Of almost 514,000 patients diagnosed with COVID-19 between January 2020 and March 2021, 20 also received a diagnosis of CVST.
Among about 490,000 adults who received either the Pfizer or Moderna vaccines, there were no diagnosed cases of thrombocytopenia.
Dr. Furie reiterated that CVST blood clots “are very rare adverse events,” but recommended that any patient in the ED with a suspected clot should be screened immediately to determine if they received a COVID vaccine during the previous few weeks.
For those who have recently received the COVID-19 vaccine, a suspected clot should be treated with nonheparin anticoagulants, Dr. Furie said.
“No heparin products in any dose should be given for suspected CVST, TTS, or VITT. With the right treatment, most patients can have a full recovery,” she added. The report includes additional, detailed treatment recommendations if one of these conditions are suspected.
Rare events
The authors noted that cases of TTS/VITT occurred up to 2.5 weeks after receiving the J&J vaccine in the United States and up to 3.5 weeks after receiving the AstraZeneca vaccine in Europe.
An April 23 report from the CDC and FDA noted that, out of almost 7 million adults who received the J&J vaccine, the agencies investigated only 15 reported cases of TTS.
An April 7 report from the European Medicines Agency noted that, out of more than 25 million people who received the AstraZeneca vaccine in the European Union, it found 62 cases of CVST.
A statement put out by the American Heart Association/American Stroke Association urges “everyone to receive a COVID-19 vaccine” as soon as possible.
“We are confident the benefits of vaccination far exceed the very small, rare risks,” the organizations said. “The risks of vaccination are also far smaller than the risk of COVID-19 and its potentially fatal consequences.”
A version of this article first appeared on Medscape.com.
A newly released report is offering guidance concerning rare conditions associated with COVID-19 as well as vaccines against the virus.
The report was released April 29, 2021, by the American Heart Association/American Stroke Association Stroke Council Leadership in answer to the decision April 23 by the Centers for Disease Control and Prevention and the Food and Drug Administration to lift an earlier “pause” in administration of the Johnson & Johnson (Janssen) vaccine.
That pause had been put in place after reports were received of a possible association between the J&J vaccine and cerebral venous sinus thrombosis (CVST) and thrombosis-thrombocytopenia syndrome (TTS, blood clots plus low blood platelets). CVST and TTS were also linked to patients in Europe and Canada who received the AstraZeneca COVID-19 vaccine.
However, the new report noted that these conditions are very rare.
“The risk of CVST due to infection with COVID-19 is 8-10 times higher than the risk of CVST after receiving a COVID-19 vaccine,” lead author Karen L. Furie, MD, chair of the department of neurology at Brown University, Providence, R.I., said in a press release.
“The public can be reassured by the CDC’s and FDA’s investigation and these statistics – the likelihood of developing CVST after a COVID-19 vaccine is extremely low,” said Dr. Furie, adding that the authors “urge all adults to receive any of the approved COVID-19 vaccines.”
The new guidance, which was published online April 29, 2021, in Stroke, discusses signs and symptoms of CVST and TTS, as well as vaccine-induced immune thrombotic thrombocytopenia (VITT). It also recommends best options for treating these conditions.
Assessing 81 million patients
In their analysis, the investigators assessed a database of 59 health care organizations and 81 million patients, 98% of whom were in the United States.
Of almost 514,000 patients diagnosed with COVID-19 between January 2020 and March 2021, 20 also received a diagnosis of CVST.
Among about 490,000 adults who received either the Pfizer or Moderna vaccines, there were no diagnosed cases of thrombocytopenia.
Dr. Furie reiterated that CVST blood clots “are very rare adverse events,” but recommended that any patient in the ED with a suspected clot should be screened immediately to determine if they received a COVID vaccine during the previous few weeks.
For those who have recently received the COVID-19 vaccine, a suspected clot should be treated with nonheparin anticoagulants, Dr. Furie said.
“No heparin products in any dose should be given for suspected CVST, TTS, or VITT. With the right treatment, most patients can have a full recovery,” she added. The report includes additional, detailed treatment recommendations if one of these conditions are suspected.
Rare events
The authors noted that cases of TTS/VITT occurred up to 2.5 weeks after receiving the J&J vaccine in the United States and up to 3.5 weeks after receiving the AstraZeneca vaccine in Europe.
An April 23 report from the CDC and FDA noted that, out of almost 7 million adults who received the J&J vaccine, the agencies investigated only 15 reported cases of TTS.
An April 7 report from the European Medicines Agency noted that, out of more than 25 million people who received the AstraZeneca vaccine in the European Union, it found 62 cases of CVST.
A statement put out by the American Heart Association/American Stroke Association urges “everyone to receive a COVID-19 vaccine” as soon as possible.
“We are confident the benefits of vaccination far exceed the very small, rare risks,” the organizations said. “The risks of vaccination are also far smaller than the risk of COVID-19 and its potentially fatal consequences.”
A version of this article first appeared on Medscape.com.
A newly released report is offering guidance concerning rare conditions associated with COVID-19 as well as vaccines against the virus.
The report was released April 29, 2021, by the American Heart Association/American Stroke Association Stroke Council Leadership in answer to the decision April 23 by the Centers for Disease Control and Prevention and the Food and Drug Administration to lift an earlier “pause” in administration of the Johnson & Johnson (Janssen) vaccine.
That pause had been put in place after reports were received of a possible association between the J&J vaccine and cerebral venous sinus thrombosis (CVST) and thrombosis-thrombocytopenia syndrome (TTS, blood clots plus low blood platelets). CVST and TTS were also linked to patients in Europe and Canada who received the AstraZeneca COVID-19 vaccine.
However, the new report noted that these conditions are very rare.
“The risk of CVST due to infection with COVID-19 is 8-10 times higher than the risk of CVST after receiving a COVID-19 vaccine,” lead author Karen L. Furie, MD, chair of the department of neurology at Brown University, Providence, R.I., said in a press release.
“The public can be reassured by the CDC’s and FDA’s investigation and these statistics – the likelihood of developing CVST after a COVID-19 vaccine is extremely low,” said Dr. Furie, adding that the authors “urge all adults to receive any of the approved COVID-19 vaccines.”
The new guidance, which was published online April 29, 2021, in Stroke, discusses signs and symptoms of CVST and TTS, as well as vaccine-induced immune thrombotic thrombocytopenia (VITT). It also recommends best options for treating these conditions.
Assessing 81 million patients
In their analysis, the investigators assessed a database of 59 health care organizations and 81 million patients, 98% of whom were in the United States.
Of almost 514,000 patients diagnosed with COVID-19 between January 2020 and March 2021, 20 also received a diagnosis of CVST.
Among about 490,000 adults who received either the Pfizer or Moderna vaccines, there were no diagnosed cases of thrombocytopenia.
Dr. Furie reiterated that CVST blood clots “are very rare adverse events,” but recommended that any patient in the ED with a suspected clot should be screened immediately to determine if they received a COVID vaccine during the previous few weeks.
For those who have recently received the COVID-19 vaccine, a suspected clot should be treated with nonheparin anticoagulants, Dr. Furie said.
“No heparin products in any dose should be given for suspected CVST, TTS, or VITT. With the right treatment, most patients can have a full recovery,” she added. The report includes additional, detailed treatment recommendations if one of these conditions are suspected.
Rare events
The authors noted that cases of TTS/VITT occurred up to 2.5 weeks after receiving the J&J vaccine in the United States and up to 3.5 weeks after receiving the AstraZeneca vaccine in Europe.
An April 23 report from the CDC and FDA noted that, out of almost 7 million adults who received the J&J vaccine, the agencies investigated only 15 reported cases of TTS.
An April 7 report from the European Medicines Agency noted that, out of more than 25 million people who received the AstraZeneca vaccine in the European Union, it found 62 cases of CVST.
A statement put out by the American Heart Association/American Stroke Association urges “everyone to receive a COVID-19 vaccine” as soon as possible.
“We are confident the benefits of vaccination far exceed the very small, rare risks,” the organizations said. “The risks of vaccination are also far smaller than the risk of COVID-19 and its potentially fatal consequences.”
A version of this article first appeared on Medscape.com.
FDA class I recall for some Cordis carotid stent systems
Cordis, part of Cardinal Health, has recalled certain lots of its Precise PRO Rx carotid stent system because of a risk of separation of the distal tip of the sheathed delivery system during use.
The Food and Drug Administration has classified this recall as class I, the most serious type, because of the potential for serious injury or death.
“If the device separates during use this may cause serious adverse events such as removal of the separated tip from the carotid artery, embolization distally, or stroke,” noted the recall notice posted on the FDA website.
To date, there have been seven complaints, including five reported injuries, related to this device issue. No deaths have been reported.
The Precise PRO Rx stent system is used in patients with stenotic lesions of the carotid arteries. The system includes a metal (nitinol) self-expanding stent preloaded on a delivery catheter used to place the stent.
The recall covers 7,300 devices made between October 2019 and August 2020 and distributed between Dec. 6, 2019, to Feb. 8, 2021.
The FDA has a complete list of product and lot numbers for the recalled devices on their website.
The company sent an urgent medical device recall letter to all affected customers asking them to check inventories and providing instructions on how to return any recalled product they have on hand.
Health care providers with questions about this recall can contact the company by email at [email protected] or by phone at 786-313-2087.
Health care providers can report adverse reactions or quality problems they experience using these devices to the FDA’s MedWatch program.
A version of this article first appeared on Medscape.com.
Cordis, part of Cardinal Health, has recalled certain lots of its Precise PRO Rx carotid stent system because of a risk of separation of the distal tip of the sheathed delivery system during use.
The Food and Drug Administration has classified this recall as class I, the most serious type, because of the potential for serious injury or death.
“If the device separates during use this may cause serious adverse events such as removal of the separated tip from the carotid artery, embolization distally, or stroke,” noted the recall notice posted on the FDA website.
To date, there have been seven complaints, including five reported injuries, related to this device issue. No deaths have been reported.
The Precise PRO Rx stent system is used in patients with stenotic lesions of the carotid arteries. The system includes a metal (nitinol) self-expanding stent preloaded on a delivery catheter used to place the stent.
The recall covers 7,300 devices made between October 2019 and August 2020 and distributed between Dec. 6, 2019, to Feb. 8, 2021.
The FDA has a complete list of product and lot numbers for the recalled devices on their website.
The company sent an urgent medical device recall letter to all affected customers asking them to check inventories and providing instructions on how to return any recalled product they have on hand.
Health care providers with questions about this recall can contact the company by email at [email protected] or by phone at 786-313-2087.
Health care providers can report adverse reactions or quality problems they experience using these devices to the FDA’s MedWatch program.
A version of this article first appeared on Medscape.com.
Cordis, part of Cardinal Health, has recalled certain lots of its Precise PRO Rx carotid stent system because of a risk of separation of the distal tip of the sheathed delivery system during use.
The Food and Drug Administration has classified this recall as class I, the most serious type, because of the potential for serious injury or death.
“If the device separates during use this may cause serious adverse events such as removal of the separated tip from the carotid artery, embolization distally, or stroke,” noted the recall notice posted on the FDA website.
To date, there have been seven complaints, including five reported injuries, related to this device issue. No deaths have been reported.
The Precise PRO Rx stent system is used in patients with stenotic lesions of the carotid arteries. The system includes a metal (nitinol) self-expanding stent preloaded on a delivery catheter used to place the stent.
The recall covers 7,300 devices made between October 2019 and August 2020 and distributed between Dec. 6, 2019, to Feb. 8, 2021.
The FDA has a complete list of product and lot numbers for the recalled devices on their website.
The company sent an urgent medical device recall letter to all affected customers asking them to check inventories and providing instructions on how to return any recalled product they have on hand.
Health care providers with questions about this recall can contact the company by email at [email protected] or by phone at 786-313-2087.
Health care providers can report adverse reactions or quality problems they experience using these devices to the FDA’s MedWatch program.
A version of this article first appeared on Medscape.com.
Higher MI shock survival with NCSI protocol: Final results
What started as an attempt to standardize care for acute myocardial infarction with cardiogenic shock at a handful of Detroit-area hospitals has led to markedly better survival rates than the traditional flip of a coin, in a nationwide analysis.
Final results from the National Cardiogenic Shock Initiative (NCSI) show 71% of patients survived to discharge and 68% were alive at 30 days.
Patients presenting in stage C or D shock, who comprised the bulk of patients in previous trials, had survival rates of 79% and 77%, respectively.
Among stage E patients, who are in extremis and have typical survival rates of less than 20%, survival was 54% at discharge and 49% at 30 days, co–principal investigator Babar Basir, DO, Henry Ford Hospital, Detroit, reported at the Society for Cardiovascular Angiography and Interventions (SCAI) annual scientific sessions, held virtually.
“This is the first push to really be able to consistently get survival rates over 50%, particularly in those patients who presented in stage C and D shock,” he said. “Really, it’s important to emphasize here the hard work it’s taken to get to this point and all the research that’s been done.”
The NCSI protocol emphasizes rapid identification and support of cardiogenic shock (door to support time <90 minutes), early placement of the Impella (Abiomed) ventricular assist device prior to percutaneous coronary intervention (PCI), and right heart monitoring to reduce the use of inotropes and vasopressors.
Co–principal investigator William O’Neill, MD, also from Henry Ford, previously reported results from the pilot study showing 84% of 30 patients survived to discharge.
The present analysis was based on outcomes of 406 consecutive acute MI patients (mean age, 63.7 years; 24% female) who presented with cardiogenic shock at 32 academic and 48 community hospitals in 29 states and the District of Columbia.
Dr. Basir emphasized that this is the largest prospective North American acute MI cardiogenic shock study in 20 years and recruited “one of the sickest cohorts ever studied.” The average blood pressure among the patients was 77/50 mm Hg; 77% had a lactate of at least 2 mmol/L (mean, 4.8 mmol/L), and 25% were in stage E shock.
One-quarter of patients were transferred from other institutions, 82% presented with ST-segment elevation MI, two-thirds had multivessel disease, and 13% had a left main culprit lesion.
Right heart catheterization was used in 90% of patients, an Impella CP device in 92%, an Impella 2.5 device in 5%, femoral access PCI in 78%, and aspiration thrombectomy in a full 27%.
Despite this sick cohort, survival at 30 days was better than in any previous study of cardiogenic shock, Dr. Basir said. In comparison, 30-day survival rates were 53%, 60%, and 49% in the SHOCK, IABP SHOCK, and CULPRIT SHOCK trials, respectively.
That said, survival over the course of the first year fell to 53% in the entire cohort, 62% in patients with stage C or D shock, and 31% in those in stage E shock.
“One-year mortality continues to be a problem for these patients and emphasizes the need for goal-directed medical therapy, early advanced heart failure follow-up, and novel therapies such as what we are planning with the evaluation of [supersaturated oxygen] SSO2 to reduce infarct sizes in the ISO-SHOCK trial,” set to begin later this year, Dr. Basir said.
Given the promising results in the NCSI, the randomized controlled RECOVER IV trial is planned to begin in 2022, he noted. It will assess whether Impella pre-PCI is superior to PCI without Impella in patients with inclusion criteria similar to that of the NCSI. The DanGer Shock randomized trial is ongoing in Denmark and Germany and assessing all-cause mortality at 6 months with the Impella CP device compared with standard of care.
“We hypothesize that greater utilization of this protocol, and refinement of the escalation strategies will consistently lead to a survival rate greater than 80%,” Dr. Basir concluded.
Past SCAI president Kirk Garratt, MD, Christiana Care, Newark, Del., who moderated a press conference where the data were highlighted, noted that late complications led to a roughly 20% absolute mortality increase from discharge to 1 year, and questioned what percentage could be attributed to the mechanical support offered.
Dr. Basir said that information was not specifically tracked but that many patients presented with multiorgan failure and, irrespective of that, the majority died from ongoing heart failure.
During the formal presentation, panelist Ron Waksman, MD, MedStar Heart Institute, Washington, questioned whether results were different between academic and community centers, but also pointed to the lack of a comparator in the single-arm study.
“It’s very hard to do any comparison historically; we do need to have a control group,” he said. “If you would have opened it to any treatment at the time of the initiative, which is great, but not just limit it to use of the Impella devices, we would have better understanding if there is really a differentiation between one device versus the other devices.”
Dr. Basir replied, “I think that is a very reasonable comment and, in regard to your question, it is always difficult to differentiate between academic and community centers, but these were large community programs that have all of the technologies available in an academic center.”
NCIS is funded in part by unrestricted grants from Abiomed and Chiesi. Dr. Basir reported consulting for Abbott Vascular, Abiomed, Cardiovascular Systems, Chiesi, Procyrion, and Zoll.
A version of this article first appeared on Medscape.com.
What started as an attempt to standardize care for acute myocardial infarction with cardiogenic shock at a handful of Detroit-area hospitals has led to markedly better survival rates than the traditional flip of a coin, in a nationwide analysis.
Final results from the National Cardiogenic Shock Initiative (NCSI) show 71% of patients survived to discharge and 68% were alive at 30 days.
Patients presenting in stage C or D shock, who comprised the bulk of patients in previous trials, had survival rates of 79% and 77%, respectively.
Among stage E patients, who are in extremis and have typical survival rates of less than 20%, survival was 54% at discharge and 49% at 30 days, co–principal investigator Babar Basir, DO, Henry Ford Hospital, Detroit, reported at the Society for Cardiovascular Angiography and Interventions (SCAI) annual scientific sessions, held virtually.
“This is the first push to really be able to consistently get survival rates over 50%, particularly in those patients who presented in stage C and D shock,” he said. “Really, it’s important to emphasize here the hard work it’s taken to get to this point and all the research that’s been done.”
The NCSI protocol emphasizes rapid identification and support of cardiogenic shock (door to support time <90 minutes), early placement of the Impella (Abiomed) ventricular assist device prior to percutaneous coronary intervention (PCI), and right heart monitoring to reduce the use of inotropes and vasopressors.
Co–principal investigator William O’Neill, MD, also from Henry Ford, previously reported results from the pilot study showing 84% of 30 patients survived to discharge.
The present analysis was based on outcomes of 406 consecutive acute MI patients (mean age, 63.7 years; 24% female) who presented with cardiogenic shock at 32 academic and 48 community hospitals in 29 states and the District of Columbia.
Dr. Basir emphasized that this is the largest prospective North American acute MI cardiogenic shock study in 20 years and recruited “one of the sickest cohorts ever studied.” The average blood pressure among the patients was 77/50 mm Hg; 77% had a lactate of at least 2 mmol/L (mean, 4.8 mmol/L), and 25% were in stage E shock.
One-quarter of patients were transferred from other institutions, 82% presented with ST-segment elevation MI, two-thirds had multivessel disease, and 13% had a left main culprit lesion.
Right heart catheterization was used in 90% of patients, an Impella CP device in 92%, an Impella 2.5 device in 5%, femoral access PCI in 78%, and aspiration thrombectomy in a full 27%.
Despite this sick cohort, survival at 30 days was better than in any previous study of cardiogenic shock, Dr. Basir said. In comparison, 30-day survival rates were 53%, 60%, and 49% in the SHOCK, IABP SHOCK, and CULPRIT SHOCK trials, respectively.
That said, survival over the course of the first year fell to 53% in the entire cohort, 62% in patients with stage C or D shock, and 31% in those in stage E shock.
“One-year mortality continues to be a problem for these patients and emphasizes the need for goal-directed medical therapy, early advanced heart failure follow-up, and novel therapies such as what we are planning with the evaluation of [supersaturated oxygen] SSO2 to reduce infarct sizes in the ISO-SHOCK trial,” set to begin later this year, Dr. Basir said.
Given the promising results in the NCSI, the randomized controlled RECOVER IV trial is planned to begin in 2022, he noted. It will assess whether Impella pre-PCI is superior to PCI without Impella in patients with inclusion criteria similar to that of the NCSI. The DanGer Shock randomized trial is ongoing in Denmark and Germany and assessing all-cause mortality at 6 months with the Impella CP device compared with standard of care.
“We hypothesize that greater utilization of this protocol, and refinement of the escalation strategies will consistently lead to a survival rate greater than 80%,” Dr. Basir concluded.
Past SCAI president Kirk Garratt, MD, Christiana Care, Newark, Del., who moderated a press conference where the data were highlighted, noted that late complications led to a roughly 20% absolute mortality increase from discharge to 1 year, and questioned what percentage could be attributed to the mechanical support offered.
Dr. Basir said that information was not specifically tracked but that many patients presented with multiorgan failure and, irrespective of that, the majority died from ongoing heart failure.
During the formal presentation, panelist Ron Waksman, MD, MedStar Heart Institute, Washington, questioned whether results were different between academic and community centers, but also pointed to the lack of a comparator in the single-arm study.
“It’s very hard to do any comparison historically; we do need to have a control group,” he said. “If you would have opened it to any treatment at the time of the initiative, which is great, but not just limit it to use of the Impella devices, we would have better understanding if there is really a differentiation between one device versus the other devices.”
Dr. Basir replied, “I think that is a very reasonable comment and, in regard to your question, it is always difficult to differentiate between academic and community centers, but these were large community programs that have all of the technologies available in an academic center.”
NCIS is funded in part by unrestricted grants from Abiomed and Chiesi. Dr. Basir reported consulting for Abbott Vascular, Abiomed, Cardiovascular Systems, Chiesi, Procyrion, and Zoll.
A version of this article first appeared on Medscape.com.
What started as an attempt to standardize care for acute myocardial infarction with cardiogenic shock at a handful of Detroit-area hospitals has led to markedly better survival rates than the traditional flip of a coin, in a nationwide analysis.
Final results from the National Cardiogenic Shock Initiative (NCSI) show 71% of patients survived to discharge and 68% were alive at 30 days.
Patients presenting in stage C or D shock, who comprised the bulk of patients in previous trials, had survival rates of 79% and 77%, respectively.
Among stage E patients, who are in extremis and have typical survival rates of less than 20%, survival was 54% at discharge and 49% at 30 days, co–principal investigator Babar Basir, DO, Henry Ford Hospital, Detroit, reported at the Society for Cardiovascular Angiography and Interventions (SCAI) annual scientific sessions, held virtually.
“This is the first push to really be able to consistently get survival rates over 50%, particularly in those patients who presented in stage C and D shock,” he said. “Really, it’s important to emphasize here the hard work it’s taken to get to this point and all the research that’s been done.”
The NCSI protocol emphasizes rapid identification and support of cardiogenic shock (door to support time <90 minutes), early placement of the Impella (Abiomed) ventricular assist device prior to percutaneous coronary intervention (PCI), and right heart monitoring to reduce the use of inotropes and vasopressors.
Co–principal investigator William O’Neill, MD, also from Henry Ford, previously reported results from the pilot study showing 84% of 30 patients survived to discharge.
The present analysis was based on outcomes of 406 consecutive acute MI patients (mean age, 63.7 years; 24% female) who presented with cardiogenic shock at 32 academic and 48 community hospitals in 29 states and the District of Columbia.
Dr. Basir emphasized that this is the largest prospective North American acute MI cardiogenic shock study in 20 years and recruited “one of the sickest cohorts ever studied.” The average blood pressure among the patients was 77/50 mm Hg; 77% had a lactate of at least 2 mmol/L (mean, 4.8 mmol/L), and 25% were in stage E shock.
One-quarter of patients were transferred from other institutions, 82% presented with ST-segment elevation MI, two-thirds had multivessel disease, and 13% had a left main culprit lesion.
Right heart catheterization was used in 90% of patients, an Impella CP device in 92%, an Impella 2.5 device in 5%, femoral access PCI in 78%, and aspiration thrombectomy in a full 27%.
Despite this sick cohort, survival at 30 days was better than in any previous study of cardiogenic shock, Dr. Basir said. In comparison, 30-day survival rates were 53%, 60%, and 49% in the SHOCK, IABP SHOCK, and CULPRIT SHOCK trials, respectively.
That said, survival over the course of the first year fell to 53% in the entire cohort, 62% in patients with stage C or D shock, and 31% in those in stage E shock.
“One-year mortality continues to be a problem for these patients and emphasizes the need for goal-directed medical therapy, early advanced heart failure follow-up, and novel therapies such as what we are planning with the evaluation of [supersaturated oxygen] SSO2 to reduce infarct sizes in the ISO-SHOCK trial,” set to begin later this year, Dr. Basir said.
Given the promising results in the NCSI, the randomized controlled RECOVER IV trial is planned to begin in 2022, he noted. It will assess whether Impella pre-PCI is superior to PCI without Impella in patients with inclusion criteria similar to that of the NCSI. The DanGer Shock randomized trial is ongoing in Denmark and Germany and assessing all-cause mortality at 6 months with the Impella CP device compared with standard of care.
“We hypothesize that greater utilization of this protocol, and refinement of the escalation strategies will consistently lead to a survival rate greater than 80%,” Dr. Basir concluded.
Past SCAI president Kirk Garratt, MD, Christiana Care, Newark, Del., who moderated a press conference where the data were highlighted, noted that late complications led to a roughly 20% absolute mortality increase from discharge to 1 year, and questioned what percentage could be attributed to the mechanical support offered.
Dr. Basir said that information was not specifically tracked but that many patients presented with multiorgan failure and, irrespective of that, the majority died from ongoing heart failure.
During the formal presentation, panelist Ron Waksman, MD, MedStar Heart Institute, Washington, questioned whether results were different between academic and community centers, but also pointed to the lack of a comparator in the single-arm study.
“It’s very hard to do any comparison historically; we do need to have a control group,” he said. “If you would have opened it to any treatment at the time of the initiative, which is great, but not just limit it to use of the Impella devices, we would have better understanding if there is really a differentiation between one device versus the other devices.”
Dr. Basir replied, “I think that is a very reasonable comment and, in regard to your question, it is always difficult to differentiate between academic and community centers, but these were large community programs that have all of the technologies available in an academic center.”
NCIS is funded in part by unrestricted grants from Abiomed and Chiesi. Dr. Basir reported consulting for Abbott Vascular, Abiomed, Cardiovascular Systems, Chiesi, Procyrion, and Zoll.
A version of this article first appeared on Medscape.com.
Modest clinical gain for AF screening of asymptomatic elderly: STROKESTOP
Some, perhaps many, previously unrecognized cases of atrial fibrillation (AF) will come to light in a screening program aimed at older asymptomatic adults. The key question is whether the challenges of such systematic but age-restricted AF screening in the community, with oral anticoagulation (OAC) offered to those found to have the arrhythmia, is worthwhile in preventing events such as death or stroke.
Now there is evidence supporting such a clinical benefit from a large, prospective, randomized trial. A screening program restricted to people 75 or 76 years of age in two Swedish communities, which called on them to use a handheld single-lead ECG system at home intermittently for 2 weeks, was followed by a slight drop in clinical events over about 7 years.
The 4% decline in risk (P = .045) in the STROKESTOP trial’s “intention-to-treat” (ITT) analysis yielded a number needed to treat of 91; that is, that many people had to be targeted by the screening program to prevent one primary-endpoint clinical event.
Those included ischemic stroke, systemic thromboembolism, hospitalization for severe bleeding, and death from any cause, investigators reported April 23 during the virtual European Heart Rhythm Association (EHRA) 2021 Congress.
If that benefit and its significance seem marginal, some secondary findings might be reassuring. Half the population of the target age in the two communities – 13,979 randomly selected people – were invited to join the trial and follow the screening protocol, comprising the ITT cohort. The other half, numbering 13,996, was not invited and served as control subjects.
However, only 51% of the ITT cohort accepted the invitation and participated in the trial; they represented the “as-treated” cohort, observed Emma Svennberg, MD, PhD, Karolinska Institute, Danderyd Hospital, Stockholm, who presented the analysis at the EHRA sessions.
The screening protocol identified untreated AF, whether previously known or unknown, in about 5% of the 7,165 as-treated screening participants; OAC was initiated in about three-fourths of those cases.
The as-treated group, on their own, benefited with a 24% drop in the prospectively defined secondary endpoint of ischemic stroke, compared with the entire control group.
The clinical benefit in the ITT population was “small but significant,” but over the same period in the as-treated cohort, there was a highly significant drop in risk for ischemic stroke, Dr. Svennberg said in an interview.
The trial’s lead message, she said, is that “screening for atrial fibrillation in an elderly population reduces the risk of death and ischemic stroke without increasing the risk of bleeding.”
Caveats: As-treated vs. ITT
But there are caveats that complicate interpretation of the trial and, Dr. Svennberg proposed, point to the importance of that interpretation of both the ITT and as-treated analyses.
“We detected significantly more atrial fibrillation in the group that was randomized to screening. A major strength of our study was that we referred all of those individuals for a structured follow-up within the study,” she said. “Although the focus of the follow-up was oral anticoagulant therapy, other risk factors were also assessed and managed, such as hypertension and diabetes.”
It’s possible that increased detection of AF followed by such structured management contributed to the observed benefit, Dr. Svennberg proposed.
However, the exclusion of those in the prespecified ITT population who declined to be screened or otherwise didn’t participate left an as-treated cohort that was healthier than the ITT population or the control group.
Indeed, the nonparticipating invitees were sicker, with significantly more diabetes, vascular disease, hypertension, and heart failure, and higher CHA2DS2VASc stroke risk scores than those who agreed to participate.
“We took a more difficult route in setting up this study, in that we identified all individuals aged 75 to 76 residing in our two regions and excluded no one,” Dr. Svennberg said in an interview. “That means even individuals with end-stage disease, severe dementia, bedridden in nursing homes, et cetera, were also randomized but perhaps not likely or eligible to participate.”
Therefore, some invitees were unable to join the study even as others might have declined “out of low interest” or other personal reasons, she said. “We believe that this mimics how a population-based screening program would be performed if done in our country.”
In the ITT analysis, screening successfully identified previously unknown or untreated cases of AF, which led to expanded OAC use and intensified risk-factor management, “which was key to a successful outcome.”
In the as-treated analysis, Dr. Svennberg said, “I think a combination of the intervention and the population being overall more healthy was driving the secondary endpoint.”
Systematic vs. opportunistic screening
Although “opportunistic screening in individuals aged 65 and older” is recommended by current European Society of Cardiology guidelines, systematic screening, such as that used in STROKESTOP, has a much weaker evidence base, observed Renate B. Schnabel, MD, PhD, University Heart & Vascular Center, Hamburg, Germany, as the invited discussant after the STROKESTOP presentation.
STROKESTOP “is one of the first studies, if not the first study,” to show a clinical benefit from screening for AF, Dr. Schnabel said.
Fewer-than-projected primary outcome events were seen during the trial, and event curves for screened and control participants didn’t start to separate until about 4 years into the study, she said. It therefore might take a long time for the screened elderly to realize the clinical benefits of screening.
Studies such as the recent SCREEN-AF and mSTOPS have amply shown that AF screening in the asymptomatic elderly can reveal previously unrecognized AF far more often than would be detected in routine practice, allowing them the opportunity to go on OAC. But the trials weren’t able to show whether the benefits of such management outweigh the risks or costs.
Indeed, on April 20, the U.S. Preventive Services Task Force (USPSTF) released a draft recommendation statement concluding that “the current evidence is insufficient to assess the balance of benefits and harms” associated with AF screening in asymptomatic people at least 50 years of age.
In STROKESTOP, however, benefit for the primary outcome reached significance in the prespecified ITT analysis and “appeared to be driven by the reduction in ischemic stroke incidence,” Dr. Schnabel said.
“The future guidelines have gained strong evidence to judge on systematic atrial fibrillation screening” as it was performed in the trial, she said. “How to implement atrial fibrillation screening, including systematic screening in health care systems across Europe and beyond, remains an open question.”
A randomized population
STROKESTOP considered all 75- and 76-year-olds living in Sweden’s Stockholm County (n = 23,888) and the Halland region (n = 4,880) and randomly assigned them to the ITT group or a control group, with stratification by sex, birth year, and geographic region. In both groups, 54.6% were female and the mean CHA2DS2VASc score was 3.5.
People assigned to the ITT cohort were invited to be screened and followed. Those who agreed to participate underwent a baseline ECG assessment to detect or rule out permanent AF. Guideline-based OAC and follow-up was offered to those found with the arrhythmia. Those in sinus rhythm with no history of AF used a handheld single-lead ECG recorder (Zenicor) for 30 seconds twice daily for 14 days.
Structured management, including OAC, was offered to anyone demonstrating sufficient AF, that is, at least one bout without p waves in one 30-second recording or at least two such episodes lasting 10-29 seconds during the 2-week screening period.
In the ITT analysis, the hazard ratio (HR) for the composite clinical primary endpoint was 0.96 (95% confidence interval, 0.920-0.999; P = .045), but in the as-treated analysis, the HR for ischemic stroke was 0.76 (95% CI, 0.68-0.87; P < .001).
“I believe that this will likely be generalizable to most countries’ elderly residents,” Dr. Svennberg said. “I think if we can find a significant difference in our elderly population in Sweden, most countries will be able to do so, or find even more significant results.”
That’s because “baseline detection of AF in Sweden is high,” she said, “so new detection is likely more difficult.” Also, in Sweden, “care can be sought without monetary concern, and prescriptions are provided at low costs to the patients.” Therefore, patients newly identified with AF, whether in studies or not, “would likely be started on therapy.”
It will be important to know whether the screening strategy is cost-effective, Dr. Schnabel said, because “the overall effect, with a hazard ratio of 0.96, is not too big, and costs incurred by systematic screening are comparatively high.”
STROKESTOP “now provides sound information for cost-effectiveness analyses, which to date have largely relied on assumptions.”
STROKESTOP was partially supported by Carl Bennet AB, Boehringer-Ingelheim, Bayer, Bristol-Meyers Squibb, and Pfizer. Dr. Svennberg disclosed receiving fees for lectures or consulting from Bayer, Bristol-Meyers Squibb, Pfizer, Boehringer-Ingelheim, Merck Sharp & Dohme, and Sanofi; and institutional grants from Roche Diagnostics and Carl Bennett Ltd.
A version of this article first appeared on Medscape.com.
Some, perhaps many, previously unrecognized cases of atrial fibrillation (AF) will come to light in a screening program aimed at older asymptomatic adults. The key question is whether the challenges of such systematic but age-restricted AF screening in the community, with oral anticoagulation (OAC) offered to those found to have the arrhythmia, is worthwhile in preventing events such as death or stroke.
Now there is evidence supporting such a clinical benefit from a large, prospective, randomized trial. A screening program restricted to people 75 or 76 years of age in two Swedish communities, which called on them to use a handheld single-lead ECG system at home intermittently for 2 weeks, was followed by a slight drop in clinical events over about 7 years.
The 4% decline in risk (P = .045) in the STROKESTOP trial’s “intention-to-treat” (ITT) analysis yielded a number needed to treat of 91; that is, that many people had to be targeted by the screening program to prevent one primary-endpoint clinical event.
Those included ischemic stroke, systemic thromboembolism, hospitalization for severe bleeding, and death from any cause, investigators reported April 23 during the virtual European Heart Rhythm Association (EHRA) 2021 Congress.
If that benefit and its significance seem marginal, some secondary findings might be reassuring. Half the population of the target age in the two communities – 13,979 randomly selected people – were invited to join the trial and follow the screening protocol, comprising the ITT cohort. The other half, numbering 13,996, was not invited and served as control subjects.
However, only 51% of the ITT cohort accepted the invitation and participated in the trial; they represented the “as-treated” cohort, observed Emma Svennberg, MD, PhD, Karolinska Institute, Danderyd Hospital, Stockholm, who presented the analysis at the EHRA sessions.
The screening protocol identified untreated AF, whether previously known or unknown, in about 5% of the 7,165 as-treated screening participants; OAC was initiated in about three-fourths of those cases.
The as-treated group, on their own, benefited with a 24% drop in the prospectively defined secondary endpoint of ischemic stroke, compared with the entire control group.
The clinical benefit in the ITT population was “small but significant,” but over the same period in the as-treated cohort, there was a highly significant drop in risk for ischemic stroke, Dr. Svennberg said in an interview.
The trial’s lead message, she said, is that “screening for atrial fibrillation in an elderly population reduces the risk of death and ischemic stroke without increasing the risk of bleeding.”
Caveats: As-treated vs. ITT
But there are caveats that complicate interpretation of the trial and, Dr. Svennberg proposed, point to the importance of that interpretation of both the ITT and as-treated analyses.
“We detected significantly more atrial fibrillation in the group that was randomized to screening. A major strength of our study was that we referred all of those individuals for a structured follow-up within the study,” she said. “Although the focus of the follow-up was oral anticoagulant therapy, other risk factors were also assessed and managed, such as hypertension and diabetes.”
It’s possible that increased detection of AF followed by such structured management contributed to the observed benefit, Dr. Svennberg proposed.
However, the exclusion of those in the prespecified ITT population who declined to be screened or otherwise didn’t participate left an as-treated cohort that was healthier than the ITT population or the control group.
Indeed, the nonparticipating invitees were sicker, with significantly more diabetes, vascular disease, hypertension, and heart failure, and higher CHA2DS2VASc stroke risk scores than those who agreed to participate.
“We took a more difficult route in setting up this study, in that we identified all individuals aged 75 to 76 residing in our two regions and excluded no one,” Dr. Svennberg said in an interview. “That means even individuals with end-stage disease, severe dementia, bedridden in nursing homes, et cetera, were also randomized but perhaps not likely or eligible to participate.”
Therefore, some invitees were unable to join the study even as others might have declined “out of low interest” or other personal reasons, she said. “We believe that this mimics how a population-based screening program would be performed if done in our country.”
In the ITT analysis, screening successfully identified previously unknown or untreated cases of AF, which led to expanded OAC use and intensified risk-factor management, “which was key to a successful outcome.”
In the as-treated analysis, Dr. Svennberg said, “I think a combination of the intervention and the population being overall more healthy was driving the secondary endpoint.”
Systematic vs. opportunistic screening
Although “opportunistic screening in individuals aged 65 and older” is recommended by current European Society of Cardiology guidelines, systematic screening, such as that used in STROKESTOP, has a much weaker evidence base, observed Renate B. Schnabel, MD, PhD, University Heart & Vascular Center, Hamburg, Germany, as the invited discussant after the STROKESTOP presentation.
STROKESTOP “is one of the first studies, if not the first study,” to show a clinical benefit from screening for AF, Dr. Schnabel said.
Fewer-than-projected primary outcome events were seen during the trial, and event curves for screened and control participants didn’t start to separate until about 4 years into the study, she said. It therefore might take a long time for the screened elderly to realize the clinical benefits of screening.
Studies such as the recent SCREEN-AF and mSTOPS have amply shown that AF screening in the asymptomatic elderly can reveal previously unrecognized AF far more often than would be detected in routine practice, allowing them the opportunity to go on OAC. But the trials weren’t able to show whether the benefits of such management outweigh the risks or costs.
Indeed, on April 20, the U.S. Preventive Services Task Force (USPSTF) released a draft recommendation statement concluding that “the current evidence is insufficient to assess the balance of benefits and harms” associated with AF screening in asymptomatic people at least 50 years of age.
In STROKESTOP, however, benefit for the primary outcome reached significance in the prespecified ITT analysis and “appeared to be driven by the reduction in ischemic stroke incidence,” Dr. Schnabel said.
“The future guidelines have gained strong evidence to judge on systematic atrial fibrillation screening” as it was performed in the trial, she said. “How to implement atrial fibrillation screening, including systematic screening in health care systems across Europe and beyond, remains an open question.”
A randomized population
STROKESTOP considered all 75- and 76-year-olds living in Sweden’s Stockholm County (n = 23,888) and the Halland region (n = 4,880) and randomly assigned them to the ITT group or a control group, with stratification by sex, birth year, and geographic region. In both groups, 54.6% were female and the mean CHA2DS2VASc score was 3.5.
People assigned to the ITT cohort were invited to be screened and followed. Those who agreed to participate underwent a baseline ECG assessment to detect or rule out permanent AF. Guideline-based OAC and follow-up was offered to those found with the arrhythmia. Those in sinus rhythm with no history of AF used a handheld single-lead ECG recorder (Zenicor) for 30 seconds twice daily for 14 days.
Structured management, including OAC, was offered to anyone demonstrating sufficient AF, that is, at least one bout without p waves in one 30-second recording or at least two such episodes lasting 10-29 seconds during the 2-week screening period.
In the ITT analysis, the hazard ratio (HR) for the composite clinical primary endpoint was 0.96 (95% confidence interval, 0.920-0.999; P = .045), but in the as-treated analysis, the HR for ischemic stroke was 0.76 (95% CI, 0.68-0.87; P < .001).
“I believe that this will likely be generalizable to most countries’ elderly residents,” Dr. Svennberg said. “I think if we can find a significant difference in our elderly population in Sweden, most countries will be able to do so, or find even more significant results.”
That’s because “baseline detection of AF in Sweden is high,” she said, “so new detection is likely more difficult.” Also, in Sweden, “care can be sought without monetary concern, and prescriptions are provided at low costs to the patients.” Therefore, patients newly identified with AF, whether in studies or not, “would likely be started on therapy.”
It will be important to know whether the screening strategy is cost-effective, Dr. Schnabel said, because “the overall effect, with a hazard ratio of 0.96, is not too big, and costs incurred by systematic screening are comparatively high.”
STROKESTOP “now provides sound information for cost-effectiveness analyses, which to date have largely relied on assumptions.”
STROKESTOP was partially supported by Carl Bennet AB, Boehringer-Ingelheim, Bayer, Bristol-Meyers Squibb, and Pfizer. Dr. Svennberg disclosed receiving fees for lectures or consulting from Bayer, Bristol-Meyers Squibb, Pfizer, Boehringer-Ingelheim, Merck Sharp & Dohme, and Sanofi; and institutional grants from Roche Diagnostics and Carl Bennett Ltd.
A version of this article first appeared on Medscape.com.
Some, perhaps many, previously unrecognized cases of atrial fibrillation (AF) will come to light in a screening program aimed at older asymptomatic adults. The key question is whether the challenges of such systematic but age-restricted AF screening in the community, with oral anticoagulation (OAC) offered to those found to have the arrhythmia, is worthwhile in preventing events such as death or stroke.
Now there is evidence supporting such a clinical benefit from a large, prospective, randomized trial. A screening program restricted to people 75 or 76 years of age in two Swedish communities, which called on them to use a handheld single-lead ECG system at home intermittently for 2 weeks, was followed by a slight drop in clinical events over about 7 years.
The 4% decline in risk (P = .045) in the STROKESTOP trial’s “intention-to-treat” (ITT) analysis yielded a number needed to treat of 91; that is, that many people had to be targeted by the screening program to prevent one primary-endpoint clinical event.
Those included ischemic stroke, systemic thromboembolism, hospitalization for severe bleeding, and death from any cause, investigators reported April 23 during the virtual European Heart Rhythm Association (EHRA) 2021 Congress.
If that benefit and its significance seem marginal, some secondary findings might be reassuring. Half the population of the target age in the two communities – 13,979 randomly selected people – were invited to join the trial and follow the screening protocol, comprising the ITT cohort. The other half, numbering 13,996, was not invited and served as control subjects.
However, only 51% of the ITT cohort accepted the invitation and participated in the trial; they represented the “as-treated” cohort, observed Emma Svennberg, MD, PhD, Karolinska Institute, Danderyd Hospital, Stockholm, who presented the analysis at the EHRA sessions.
The screening protocol identified untreated AF, whether previously known or unknown, in about 5% of the 7,165 as-treated screening participants; OAC was initiated in about three-fourths of those cases.
The as-treated group, on their own, benefited with a 24% drop in the prospectively defined secondary endpoint of ischemic stroke, compared with the entire control group.
The clinical benefit in the ITT population was “small but significant,” but over the same period in the as-treated cohort, there was a highly significant drop in risk for ischemic stroke, Dr. Svennberg said in an interview.
The trial’s lead message, she said, is that “screening for atrial fibrillation in an elderly population reduces the risk of death and ischemic stroke without increasing the risk of bleeding.”
Caveats: As-treated vs. ITT
But there are caveats that complicate interpretation of the trial and, Dr. Svennberg proposed, point to the importance of that interpretation of both the ITT and as-treated analyses.
“We detected significantly more atrial fibrillation in the group that was randomized to screening. A major strength of our study was that we referred all of those individuals for a structured follow-up within the study,” she said. “Although the focus of the follow-up was oral anticoagulant therapy, other risk factors were also assessed and managed, such as hypertension and diabetes.”
It’s possible that increased detection of AF followed by such structured management contributed to the observed benefit, Dr. Svennberg proposed.
However, the exclusion of those in the prespecified ITT population who declined to be screened or otherwise didn’t participate left an as-treated cohort that was healthier than the ITT population or the control group.
Indeed, the nonparticipating invitees were sicker, with significantly more diabetes, vascular disease, hypertension, and heart failure, and higher CHA2DS2VASc stroke risk scores than those who agreed to participate.
“We took a more difficult route in setting up this study, in that we identified all individuals aged 75 to 76 residing in our two regions and excluded no one,” Dr. Svennberg said in an interview. “That means even individuals with end-stage disease, severe dementia, bedridden in nursing homes, et cetera, were also randomized but perhaps not likely or eligible to participate.”
Therefore, some invitees were unable to join the study even as others might have declined “out of low interest” or other personal reasons, she said. “We believe that this mimics how a population-based screening program would be performed if done in our country.”
In the ITT analysis, screening successfully identified previously unknown or untreated cases of AF, which led to expanded OAC use and intensified risk-factor management, “which was key to a successful outcome.”
In the as-treated analysis, Dr. Svennberg said, “I think a combination of the intervention and the population being overall more healthy was driving the secondary endpoint.”
Systematic vs. opportunistic screening
Although “opportunistic screening in individuals aged 65 and older” is recommended by current European Society of Cardiology guidelines, systematic screening, such as that used in STROKESTOP, has a much weaker evidence base, observed Renate B. Schnabel, MD, PhD, University Heart & Vascular Center, Hamburg, Germany, as the invited discussant after the STROKESTOP presentation.
STROKESTOP “is one of the first studies, if not the first study,” to show a clinical benefit from screening for AF, Dr. Schnabel said.
Fewer-than-projected primary outcome events were seen during the trial, and event curves for screened and control participants didn’t start to separate until about 4 years into the study, she said. It therefore might take a long time for the screened elderly to realize the clinical benefits of screening.
Studies such as the recent SCREEN-AF and mSTOPS have amply shown that AF screening in the asymptomatic elderly can reveal previously unrecognized AF far more often than would be detected in routine practice, allowing them the opportunity to go on OAC. But the trials weren’t able to show whether the benefits of such management outweigh the risks or costs.
Indeed, on April 20, the U.S. Preventive Services Task Force (USPSTF) released a draft recommendation statement concluding that “the current evidence is insufficient to assess the balance of benefits and harms” associated with AF screening in asymptomatic people at least 50 years of age.
In STROKESTOP, however, benefit for the primary outcome reached significance in the prespecified ITT analysis and “appeared to be driven by the reduction in ischemic stroke incidence,” Dr. Schnabel said.
“The future guidelines have gained strong evidence to judge on systematic atrial fibrillation screening” as it was performed in the trial, she said. “How to implement atrial fibrillation screening, including systematic screening in health care systems across Europe and beyond, remains an open question.”
A randomized population
STROKESTOP considered all 75- and 76-year-olds living in Sweden’s Stockholm County (n = 23,888) and the Halland region (n = 4,880) and randomly assigned them to the ITT group or a control group, with stratification by sex, birth year, and geographic region. In both groups, 54.6% were female and the mean CHA2DS2VASc score was 3.5.
People assigned to the ITT cohort were invited to be screened and followed. Those who agreed to participate underwent a baseline ECG assessment to detect or rule out permanent AF. Guideline-based OAC and follow-up was offered to those found with the arrhythmia. Those in sinus rhythm with no history of AF used a handheld single-lead ECG recorder (Zenicor) for 30 seconds twice daily for 14 days.
Structured management, including OAC, was offered to anyone demonstrating sufficient AF, that is, at least one bout without p waves in one 30-second recording or at least two such episodes lasting 10-29 seconds during the 2-week screening period.
In the ITT analysis, the hazard ratio (HR) for the composite clinical primary endpoint was 0.96 (95% confidence interval, 0.920-0.999; P = .045), but in the as-treated analysis, the HR for ischemic stroke was 0.76 (95% CI, 0.68-0.87; P < .001).
“I believe that this will likely be generalizable to most countries’ elderly residents,” Dr. Svennberg said. “I think if we can find a significant difference in our elderly population in Sweden, most countries will be able to do so, or find even more significant results.”
That’s because “baseline detection of AF in Sweden is high,” she said, “so new detection is likely more difficult.” Also, in Sweden, “care can be sought without monetary concern, and prescriptions are provided at low costs to the patients.” Therefore, patients newly identified with AF, whether in studies or not, “would likely be started on therapy.”
It will be important to know whether the screening strategy is cost-effective, Dr. Schnabel said, because “the overall effect, with a hazard ratio of 0.96, is not too big, and costs incurred by systematic screening are comparatively high.”
STROKESTOP “now provides sound information for cost-effectiveness analyses, which to date have largely relied on assumptions.”
STROKESTOP was partially supported by Carl Bennet AB, Boehringer-Ingelheim, Bayer, Bristol-Meyers Squibb, and Pfizer. Dr. Svennberg disclosed receiving fees for lectures or consulting from Bayer, Bristol-Meyers Squibb, Pfizer, Boehringer-Ingelheim, Merck Sharp & Dohme, and Sanofi; and institutional grants from Roche Diagnostics and Carl Bennett Ltd.
A version of this article first appeared on Medscape.com.
FDA moves to ban menthol in cigarettes
The Food and Drug Administration said that within a year it will ban menthol in cigarettes and ban all flavors including menthol in cigars.
Menthol makes it easier to start smoking, and also enhances the effects of nicotine, making it more addictive and harder to quit, the FDA said in announcing its actions on Thursday.
Nineteen organizations – including the American Academy of Pediatrics, American Cancer Society, American College of Chest Physicians, American Medical Association, American Heart Association, and the National Medical Association – have pushed the FDA to ban menthol for years. The agency banned all flavors in cigarettes in 2009 but did not take any action against menthol. In 2013, the groups filed a petition demanding that the FDA ban menthol, too. The agency responded months later with a notice that it would start the process.
But it never took any action. Action on Smoking and Health and the African American Tobacco Control Leadership Council, later joined by the AMA and the NMA, sued in 2020 to compel the agency to do something. Now it has finally agreed to act.
The African American Tobacco Control Leadership Council welcomed the move but said the fight is not over and encouraged tobacco control activists to fight to ban menthol tobacco products at the local, state and federal level. “We know that this rule-making process could take years and we know that the tobacco industry will continue to do everything in their power to derail any attempt to remove their deadly products from the market,” Phillip Gardiner, MD, council cochair, said in a statement.
The AMA is urging the FDA to quickly implement the ban and remove the products “without further delay,” AMA President Susan R. Bailey, MD, said in a statement.
“FDA’s long-awaited decision to take action to eliminate menthol flavoring in cigarettes and all flavors in cigars ends a decades-long deference to the tobacco industry, which has repeatedly demonstrated its willingness to profit from products that result in death,” Lisa Lacasse, president of the American Cancer Society Cancer Action Network, said in her own statement.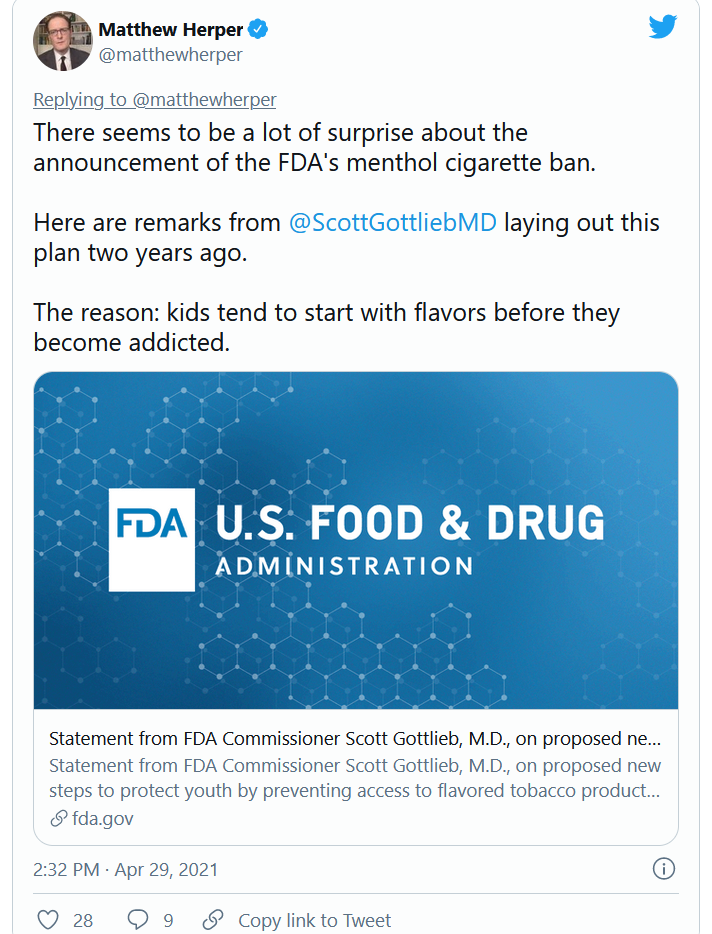
Ms. Lacasse said banning menthol will help eliminate health disparities. She said 86% of Black people who smoke use menthol cigarettes, compared with 46% of Hispanic people who smoke, 39% of Asian people who smoke, and 29% of White people who smoke. “FDA’s actions today send a clear message that Big Tobacco’s strategy to profit off addicting Black communities will no longer be tolerated,” she said.
Not all groups are on board, however. The American Civil Liberties Union and several other organizations wrote to the country’s top health officials urging them to reconsider.
“Such a ban will trigger criminal penalties which will disproportionately impact people of color, as well as prioritize criminalization over public health and harm reduction,” the letter says. “A ban will also lead to unconstitutional policing and other negative interactions with local law enforcement.”
The letter calls the proposed ban “well intentioned,” but said any effort to reduce death and disease from tobacco “must avoid solutions that will create yet another reason for armed police to engage citizens on the street based on pretext or conduct that does not pose a threat to public safety.”
Instead of a ban, the organizations said, policy makers should consider increased education for adults and minors, stop-smoking programs, and increased funding for health centers in communities of color.
The Biden administration, however, pressed the point that banning menthol will bring many positives. Acting FDA Commissioner Janet Woodcock, MD said in a statement that banning menthol “will help significantly reduce youth initiation, increase the chances of smoking cessation among current smokers, and address health disparities experienced by communities of color, low-income populations, and LGBTQ-plus individuals, all of whom are far more likely to use these tobacco products.”
The FDA cited data showing that, in the first year or so after a ban goes into effect, an additional 923,000 smokers would quit, including 230,000 African Americans. Another study suggests that 633,000 deaths would be averted, including 237,000 Black Americans.
Dr. Woodcock added that, “armed with strong scientific evidence, and with full support from the [Biden] administration, we believe these actions will launch us on a trajectory toward ending tobacco-related disease and death in the U.S.”
The FDA estimates that 18.6 million Americans who are current smokers use menthol cigarettes, with a disproportionately high number being Black people. Menthol cigarette use among Black and Hispanic youth increased from 2011 to 2018, but declined for non-Hispanic White youth.
Flavored mass-produced cigars and cigarillos are disproportionately popular among youth, especially non-Hispanic Black high school students, who in 2020 reported past 30-day cigar smoking at levels twice as high as their White counterparts, said the FDA. Three-quarters of 12- to 17-year-olds reported they smoke cigars because they like the flavors. In 2020, more young people tried a cigar every day than tried a cigarette, reports the agency.
“This long-overdue decision will protect future generations of young people from nicotine addiction, especially Black children and communities, which have disproportionately suffered from menthol tobacco use due to targeted efforts from the tobacco industry,” Lee Savio Beers, MD, president of the American Academy of Pediatrics, said in a statement.
The FDA’s announcement “is only a first step that must be followed with urgent, comprehensive action to remove these flavored products from the market,” he said.
A version of this article first appeared on WebMD.com.
The Food and Drug Administration said that within a year it will ban menthol in cigarettes and ban all flavors including menthol in cigars.
Menthol makes it easier to start smoking, and also enhances the effects of nicotine, making it more addictive and harder to quit, the FDA said in announcing its actions on Thursday.
Nineteen organizations – including the American Academy of Pediatrics, American Cancer Society, American College of Chest Physicians, American Medical Association, American Heart Association, and the National Medical Association – have pushed the FDA to ban menthol for years. The agency banned all flavors in cigarettes in 2009 but did not take any action against menthol. In 2013, the groups filed a petition demanding that the FDA ban menthol, too. The agency responded months later with a notice that it would start the process.
But it never took any action. Action on Smoking and Health and the African American Tobacco Control Leadership Council, later joined by the AMA and the NMA, sued in 2020 to compel the agency to do something. Now it has finally agreed to act.
The African American Tobacco Control Leadership Council welcomed the move but said the fight is not over and encouraged tobacco control activists to fight to ban menthol tobacco products at the local, state and federal level. “We know that this rule-making process could take years and we know that the tobacco industry will continue to do everything in their power to derail any attempt to remove their deadly products from the market,” Phillip Gardiner, MD, council cochair, said in a statement.
The AMA is urging the FDA to quickly implement the ban and remove the products “without further delay,” AMA President Susan R. Bailey, MD, said in a statement.
“FDA’s long-awaited decision to take action to eliminate menthol flavoring in cigarettes and all flavors in cigars ends a decades-long deference to the tobacco industry, which has repeatedly demonstrated its willingness to profit from products that result in death,” Lisa Lacasse, president of the American Cancer Society Cancer Action Network, said in her own statement.
Ms. Lacasse said banning menthol will help eliminate health disparities. She said 86% of Black people who smoke use menthol cigarettes, compared with 46% of Hispanic people who smoke, 39% of Asian people who smoke, and 29% of White people who smoke. “FDA’s actions today send a clear message that Big Tobacco’s strategy to profit off addicting Black communities will no longer be tolerated,” she said.
Not all groups are on board, however. The American Civil Liberties Union and several other organizations wrote to the country’s top health officials urging them to reconsider.
“Such a ban will trigger criminal penalties which will disproportionately impact people of color, as well as prioritize criminalization over public health and harm reduction,” the letter says. “A ban will also lead to unconstitutional policing and other negative interactions with local law enforcement.”
The letter calls the proposed ban “well intentioned,” but said any effort to reduce death and disease from tobacco “must avoid solutions that will create yet another reason for armed police to engage citizens on the street based on pretext or conduct that does not pose a threat to public safety.”
Instead of a ban, the organizations said, policy makers should consider increased education for adults and minors, stop-smoking programs, and increased funding for health centers in communities of color.
The Biden administration, however, pressed the point that banning menthol will bring many positives. Acting FDA Commissioner Janet Woodcock, MD said in a statement that banning menthol “will help significantly reduce youth initiation, increase the chances of smoking cessation among current smokers, and address health disparities experienced by communities of color, low-income populations, and LGBTQ-plus individuals, all of whom are far more likely to use these tobacco products.”
The FDA cited data showing that, in the first year or so after a ban goes into effect, an additional 923,000 smokers would quit, including 230,000 African Americans. Another study suggests that 633,000 deaths would be averted, including 237,000 Black Americans.
Dr. Woodcock added that, “armed with strong scientific evidence, and with full support from the [Biden] administration, we believe these actions will launch us on a trajectory toward ending tobacco-related disease and death in the U.S.”
The FDA estimates that 18.6 million Americans who are current smokers use menthol cigarettes, with a disproportionately high number being Black people. Menthol cigarette use among Black and Hispanic youth increased from 2011 to 2018, but declined for non-Hispanic White youth.
Flavored mass-produced cigars and cigarillos are disproportionately popular among youth, especially non-Hispanic Black high school students, who in 2020 reported past 30-day cigar smoking at levels twice as high as their White counterparts, said the FDA. Three-quarters of 12- to 17-year-olds reported they smoke cigars because they like the flavors. In 2020, more young people tried a cigar every day than tried a cigarette, reports the agency.
“This long-overdue decision will protect future generations of young people from nicotine addiction, especially Black children and communities, which have disproportionately suffered from menthol tobacco use due to targeted efforts from the tobacco industry,” Lee Savio Beers, MD, president of the American Academy of Pediatrics, said in a statement.
The FDA’s announcement “is only a first step that must be followed with urgent, comprehensive action to remove these flavored products from the market,” he said.
A version of this article first appeared on WebMD.com.
The Food and Drug Administration said that within a year it will ban menthol in cigarettes and ban all flavors including menthol in cigars.
Menthol makes it easier to start smoking, and also enhances the effects of nicotine, making it more addictive and harder to quit, the FDA said in announcing its actions on Thursday.
Nineteen organizations – including the American Academy of Pediatrics, American Cancer Society, American College of Chest Physicians, American Medical Association, American Heart Association, and the National Medical Association – have pushed the FDA to ban menthol for years. The agency banned all flavors in cigarettes in 2009 but did not take any action against menthol. In 2013, the groups filed a petition demanding that the FDA ban menthol, too. The agency responded months later with a notice that it would start the process.
But it never took any action. Action on Smoking and Health and the African American Tobacco Control Leadership Council, later joined by the AMA and the NMA, sued in 2020 to compel the agency to do something. Now it has finally agreed to act.
The African American Tobacco Control Leadership Council welcomed the move but said the fight is not over and encouraged tobacco control activists to fight to ban menthol tobacco products at the local, state and federal level. “We know that this rule-making process could take years and we know that the tobacco industry will continue to do everything in their power to derail any attempt to remove their deadly products from the market,” Phillip Gardiner, MD, council cochair, said in a statement.
The AMA is urging the FDA to quickly implement the ban and remove the products “without further delay,” AMA President Susan R. Bailey, MD, said in a statement.
“FDA’s long-awaited decision to take action to eliminate menthol flavoring in cigarettes and all flavors in cigars ends a decades-long deference to the tobacco industry, which has repeatedly demonstrated its willingness to profit from products that result in death,” Lisa Lacasse, president of the American Cancer Society Cancer Action Network, said in her own statement.
Ms. Lacasse said banning menthol will help eliminate health disparities. She said 86% of Black people who smoke use menthol cigarettes, compared with 46% of Hispanic people who smoke, 39% of Asian people who smoke, and 29% of White people who smoke. “FDA’s actions today send a clear message that Big Tobacco’s strategy to profit off addicting Black communities will no longer be tolerated,” she said.
Not all groups are on board, however. The American Civil Liberties Union and several other organizations wrote to the country’s top health officials urging them to reconsider.
“Such a ban will trigger criminal penalties which will disproportionately impact people of color, as well as prioritize criminalization over public health and harm reduction,” the letter says. “A ban will also lead to unconstitutional policing and other negative interactions with local law enforcement.”
The letter calls the proposed ban “well intentioned,” but said any effort to reduce death and disease from tobacco “must avoid solutions that will create yet another reason for armed police to engage citizens on the street based on pretext or conduct that does not pose a threat to public safety.”
Instead of a ban, the organizations said, policy makers should consider increased education for adults and minors, stop-smoking programs, and increased funding for health centers in communities of color.
The Biden administration, however, pressed the point that banning menthol will bring many positives. Acting FDA Commissioner Janet Woodcock, MD said in a statement that banning menthol “will help significantly reduce youth initiation, increase the chances of smoking cessation among current smokers, and address health disparities experienced by communities of color, low-income populations, and LGBTQ-plus individuals, all of whom are far more likely to use these tobacco products.”
The FDA cited data showing that, in the first year or so after a ban goes into effect, an additional 923,000 smokers would quit, including 230,000 African Americans. Another study suggests that 633,000 deaths would be averted, including 237,000 Black Americans.
Dr. Woodcock added that, “armed with strong scientific evidence, and with full support from the [Biden] administration, we believe these actions will launch us on a trajectory toward ending tobacco-related disease and death in the U.S.”
The FDA estimates that 18.6 million Americans who are current smokers use menthol cigarettes, with a disproportionately high number being Black people. Menthol cigarette use among Black and Hispanic youth increased from 2011 to 2018, but declined for non-Hispanic White youth.
Flavored mass-produced cigars and cigarillos are disproportionately popular among youth, especially non-Hispanic Black high school students, who in 2020 reported past 30-day cigar smoking at levels twice as high as their White counterparts, said the FDA. Three-quarters of 12- to 17-year-olds reported they smoke cigars because they like the flavors. In 2020, more young people tried a cigar every day than tried a cigarette, reports the agency.
“This long-overdue decision will protect future generations of young people from nicotine addiction, especially Black children and communities, which have disproportionately suffered from menthol tobacco use due to targeted efforts from the tobacco industry,” Lee Savio Beers, MD, president of the American Academy of Pediatrics, said in a statement.
The FDA’s announcement “is only a first step that must be followed with urgent, comprehensive action to remove these flavored products from the market,” he said.
A version of this article first appeared on WebMD.com.
CDC guidelines coming on long COVID
The Centers for Disease Control and Prevention is finalizing new guidelines to help clinicians diagnose and manage long COVID, or postacute sequelae of SARS-CoV-2 infection.
In a day-long congressional hearing on April 28, John Brooks, MD, a medical epidemiologist at the CDC’s division of HIV/AIDS prevention, testified that the guidelines were going through the clearance process at the agency, but would be forthcoming.
“They should be coming out very shortly,” Dr. Brooks said.
The guidelines, which were developed in collaboration with newly established long-COVID clinics and patient advocacy groups, will “illustrate how to diagnose and begin to pull together what we know about management,” of the complex condition, he said.
For many doctors and patients who are struggling to understand symptoms that persist for months after the initial viral infection, the guidelines can’t come soon enough.
National Institutes of Health Director Francis Collins, MD, PhD, who also testified at the hearing, estimated that as many as 3 million people could be left with chronic health problems after even mild COVID infections.
“I can’t overstate how serious this issue is for the health of our nation,” he said.
Dr. Collins said his estimate was based on studies showing that roughly 10% of people who get COVID could be affected by this and whose “long-term course is uncertain,” he said. So far, more than 32 million Americans are known to have been infected with the new coronavirus.
“We need to make sure we put our arms around them and bring answers and care to them,” said Rep. Anna Eshoo (D-Calif.), chairwoman of the Subcommittee on Health.
Jennifer Possick, MD, who directs the post-COVID recovery program at Yale New Haven (Conn.) Hospital, testified that the tidal wave of patients she and her colleagues were seeing was overwhelming.
“We are a well-resourced program at an academic medical center, but we are swamped by the need in our community. This year, we have seen more patients with post COVID-19 conditions in our clinic alone than we have new cases of asthma and COPD combined,” she said. “The magnitude of the challenge is daunting.”
Dr. Possick estimated that there are “over 60” clinics in the United States that have started to treat long-COVID patients, but said they are grassroots efforts and all very different from each other.
“Whoever had the resources, had the time, [and] was able to take the initiative and forge to the relationships because most of them are multidisciplinary, did so,” she said.
Patients testify
Several representatives shared moving personal stories of loved ones or staffers who remained ill months after a COVID diagnosis.
Rep. Ann Kuster, from New Hampshire, talked about her 34-year-old niece, a member of the U.S. Ski Team, who had COVID just over a year ago and “continues to struggle with everything, even the simplest activities of daily living” she said. “She has to choose between taking a shower or making dinner. I’m so proud of her for hanging in there.”
Long-COVID patients invited to testify by the subcommittee described months of disability that left them with soaring medical bills and no ability to work to pay them.
“I am now a poor, Black, disabled woman, living with long COVID,” said Chimere Smith, who said she had been a school teacher in Baltimore. “Saying it aloud makes it no more easy to accept.”
She said COVID had affected her ability to think clearly and caused debilitating fatigue, which prevented her from working. She said she lost her vision for almost 5 months because doctors misdiagnosed a cataract caused by long COVID as dry eye.
“If I did not have a loving family, I [would] be speaking to you today [from] my car, the only property I now own.”
Ms. Smith said that long-COVID clinics, which are mostly housed within academic medical centers, were not going to be accessible for all long-haulers, who are disproportionately women of color. She has started a clinic, based out of her church, to help other patients from her community.
“No one wants to hear that long COVID has decimated my life or the lives of other black women in less than a year,” Ms. Smith said. “We’ve just been waiting and hoping for compassionate doctors and politicians who would acknowledge us.”
A version of this article first appeared on Medscape.com.
The Centers for Disease Control and Prevention is finalizing new guidelines to help clinicians diagnose and manage long COVID, or postacute sequelae of SARS-CoV-2 infection.
In a day-long congressional hearing on April 28, John Brooks, MD, a medical epidemiologist at the CDC’s division of HIV/AIDS prevention, testified that the guidelines were going through the clearance process at the agency, but would be forthcoming.
“They should be coming out very shortly,” Dr. Brooks said.
The guidelines, which were developed in collaboration with newly established long-COVID clinics and patient advocacy groups, will “illustrate how to diagnose and begin to pull together what we know about management,” of the complex condition, he said.
For many doctors and patients who are struggling to understand symptoms that persist for months after the initial viral infection, the guidelines can’t come soon enough.
National Institutes of Health Director Francis Collins, MD, PhD, who also testified at the hearing, estimated that as many as 3 million people could be left with chronic health problems after even mild COVID infections.
“I can’t overstate how serious this issue is for the health of our nation,” he said.
Dr. Collins said his estimate was based on studies showing that roughly 10% of people who get COVID could be affected by this and whose “long-term course is uncertain,” he said. So far, more than 32 million Americans are known to have been infected with the new coronavirus.
“We need to make sure we put our arms around them and bring answers and care to them,” said Rep. Anna Eshoo (D-Calif.), chairwoman of the Subcommittee on Health.
Jennifer Possick, MD, who directs the post-COVID recovery program at Yale New Haven (Conn.) Hospital, testified that the tidal wave of patients she and her colleagues were seeing was overwhelming.
“We are a well-resourced program at an academic medical center, but we are swamped by the need in our community. This year, we have seen more patients with post COVID-19 conditions in our clinic alone than we have new cases of asthma and COPD combined,” she said. “The magnitude of the challenge is daunting.”
Dr. Possick estimated that there are “over 60” clinics in the United States that have started to treat long-COVID patients, but said they are grassroots efforts and all very different from each other.
“Whoever had the resources, had the time, [and] was able to take the initiative and forge to the relationships because most of them are multidisciplinary, did so,” she said.
Patients testify
Several representatives shared moving personal stories of loved ones or staffers who remained ill months after a COVID diagnosis.
Rep. Ann Kuster, from New Hampshire, talked about her 34-year-old niece, a member of the U.S. Ski Team, who had COVID just over a year ago and “continues to struggle with everything, even the simplest activities of daily living” she said. “She has to choose between taking a shower or making dinner. I’m so proud of her for hanging in there.”
Long-COVID patients invited to testify by the subcommittee described months of disability that left them with soaring medical bills and no ability to work to pay them.
“I am now a poor, Black, disabled woman, living with long COVID,” said Chimere Smith, who said she had been a school teacher in Baltimore. “Saying it aloud makes it no more easy to accept.”
She said COVID had affected her ability to think clearly and caused debilitating fatigue, which prevented her from working. She said she lost her vision for almost 5 months because doctors misdiagnosed a cataract caused by long COVID as dry eye.
“If I did not have a loving family, I [would] be speaking to you today [from] my car, the only property I now own.”
Ms. Smith said that long-COVID clinics, which are mostly housed within academic medical centers, were not going to be accessible for all long-haulers, who are disproportionately women of color. She has started a clinic, based out of her church, to help other patients from her community.
“No one wants to hear that long COVID has decimated my life or the lives of other black women in less than a year,” Ms. Smith said. “We’ve just been waiting and hoping for compassionate doctors and politicians who would acknowledge us.”
A version of this article first appeared on Medscape.com.
The Centers for Disease Control and Prevention is finalizing new guidelines to help clinicians diagnose and manage long COVID, or postacute sequelae of SARS-CoV-2 infection.
In a day-long congressional hearing on April 28, John Brooks, MD, a medical epidemiologist at the CDC’s division of HIV/AIDS prevention, testified that the guidelines were going through the clearance process at the agency, but would be forthcoming.
“They should be coming out very shortly,” Dr. Brooks said.
The guidelines, which were developed in collaboration with newly established long-COVID clinics and patient advocacy groups, will “illustrate how to diagnose and begin to pull together what we know about management,” of the complex condition, he said.
For many doctors and patients who are struggling to understand symptoms that persist for months after the initial viral infection, the guidelines can’t come soon enough.
National Institutes of Health Director Francis Collins, MD, PhD, who also testified at the hearing, estimated that as many as 3 million people could be left with chronic health problems after even mild COVID infections.
“I can’t overstate how serious this issue is for the health of our nation,” he said.
Dr. Collins said his estimate was based on studies showing that roughly 10% of people who get COVID could be affected by this and whose “long-term course is uncertain,” he said. So far, more than 32 million Americans are known to have been infected with the new coronavirus.
“We need to make sure we put our arms around them and bring answers and care to them,” said Rep. Anna Eshoo (D-Calif.), chairwoman of the Subcommittee on Health.
Jennifer Possick, MD, who directs the post-COVID recovery program at Yale New Haven (Conn.) Hospital, testified that the tidal wave of patients she and her colleagues were seeing was overwhelming.
“We are a well-resourced program at an academic medical center, but we are swamped by the need in our community. This year, we have seen more patients with post COVID-19 conditions in our clinic alone than we have new cases of asthma and COPD combined,” she said. “The magnitude of the challenge is daunting.”
Dr. Possick estimated that there are “over 60” clinics in the United States that have started to treat long-COVID patients, but said they are grassroots efforts and all very different from each other.
“Whoever had the resources, had the time, [and] was able to take the initiative and forge to the relationships because most of them are multidisciplinary, did so,” she said.
Patients testify
Several representatives shared moving personal stories of loved ones or staffers who remained ill months after a COVID diagnosis.
Rep. Ann Kuster, from New Hampshire, talked about her 34-year-old niece, a member of the U.S. Ski Team, who had COVID just over a year ago and “continues to struggle with everything, even the simplest activities of daily living” she said. “She has to choose between taking a shower or making dinner. I’m so proud of her for hanging in there.”
Long-COVID patients invited to testify by the subcommittee described months of disability that left them with soaring medical bills and no ability to work to pay them.
“I am now a poor, Black, disabled woman, living with long COVID,” said Chimere Smith, who said she had been a school teacher in Baltimore. “Saying it aloud makes it no more easy to accept.”
She said COVID had affected her ability to think clearly and caused debilitating fatigue, which prevented her from working. She said she lost her vision for almost 5 months because doctors misdiagnosed a cataract caused by long COVID as dry eye.
“If I did not have a loving family, I [would] be speaking to you today [from] my car, the only property I now own.”
Ms. Smith said that long-COVID clinics, which are mostly housed within academic medical centers, were not going to be accessible for all long-haulers, who are disproportionately women of color. She has started a clinic, based out of her church, to help other patients from her community.
“No one wants to hear that long COVID has decimated my life or the lives of other black women in less than a year,” Ms. Smith said. “We’ve just been waiting and hoping for compassionate doctors and politicians who would acknowledge us.”
A version of this article first appeared on Medscape.com.
Being overweight ups risk of severe COVID-19 in hospital
In a global meta-analysis of more than 7,000 patients who were hospitalized with COVID-19, individuals with overweight or obesity were more likely to need respiratory support but were not more likely to die in the hospital, compared to individuals of normal weight.
Compared to patients without diabetes, those with diabetes had higher odds of needing invasive respiratory support (with intubation) but not for needing noninvasive respiratory support or of dying in the hospital.
“Surprisingly,” among patients with diabetes, being overweight or having obesity did not further increase the odds of any of these outcomes, the researchers wrote. The finding needs to be confirmed in larger studies, they said, because the sample sizes in these subanalyses were small and the confidence intervals were large.
The study by Danielle K. Longmore, PhD, of Murdoch Children’s Research Institute (MCRI), Melbourne, and colleagues from the International BMI-COVID consortium, was published online April 15 in Diabetes Care.
This new research “adds to the known data on the associations between obesity and severe COVID-19 disease and extends these findings” to patients who are overweight and/or have diabetes, Dr. Longmore, a pediatric endocrinologist with a clinical and research interest in childhood and youth obesity, said in an interview.
Immunologist Siroon Bekkering, PhD, of Radboud University Medical Center, Nijmegen, the Netherlands, explained that never before have so much data of different types regarding obesity been combined in one large study. Dr. Bekkering is a coauthor of the article and was a principal investigator.
“Several national and international observations already showed the important role of overweight and obesity in a more severe COVID-19 course. This study adds to those observations by combining data from several countries with the possibility to look at the risk factors separately,” she said in a statement from her institution.
“Regardless of other risk factors (such as heart disease or diabetes), we now see that too high a BMI [body mass index] can actually lead to a more severe course in [coronavirus] infection,” she said.
Study implications: Data show that overweight, obesity add to risk
These latest findings highlight the urgent need to develop public health policies to address socioeconomic and psychological drivers of obesity, Dr. Longmore said.
“Although taking steps to address obesity in the short term is unlikely to have an immediate impact in the COVID-19 pandemic, it will likely reduce the disease burden in future viral pandemics and reduce risks of complications like heart disease and stroke,” she observed in a statement issued by MCRI.
Coauthor Kirsty R. Short, PhD, a research fellow at the University of Queensland, Brisbane, Australia, noted that “obesity is associated with numerous poor health outcomes, including increased risk of cardiometabolic and respiratory disease and more severe viral disease including influenza, dengue, and SARS-CoV-1.
“Given the large scale of this study,” she said, “we have conclusively shown that being overweight or obese are independent risk factors for worse outcomes in adults hospitalized with COVID-19.”
“At the moment, the World Health Organization has not had enough high-quality data to include being overweight or obese as a risk factor for severe COVID-19 disease,” added another author, David P. Burgner, PhD, a pediatric infectious diseases clinician scientist from MCRI.
“Our study should help inform decisions about which higher-risk groups should be vaccinated as a priority,” he observed.
Does being overweight up risk of worse COVID-19 outcomes?
About 13% of the world’s population are overweight, and 40% have obesity. There are wide between-country variations in these data, and about 90% of patients with type 2 diabetes are overweight or obese, the researchers noted.
The Organisation for Economic Co-operation and Development reported that the prevalence of obesity in 2016-2017 was 5.7% to 8.9% in Asia, 9.8% to 16.8% in Europe, 26.5% in South Africa, and 40.0% in the United States, they added.
Obesity is common and has emerged as an important risk factor for severe COVID-19. However, most previous studies of COVID-19 and elevated BMI were conducted in single centers and did not focus on patients with overweight.
To investigate, the researchers identified 7,244 patients (two-thirds were overweight or obese) who were hospitalized with COVID-19 in 69 hospitals (18 sites) in 11 countries from Jan. 17, 2020, to June 2, 2020.
Most patients were hospitalized with COVID-19 in the Netherlands (2,260), followed by New York City (1,682), Switzerland (920), St. Louis (805), Norway, Italy, China, South Africa, Indonesia, Denmark, Los Angeles, Austria, and Singapore.
Just over half (60%) of the individuals were male, and 52% were older than 65.
Overall, 34.8% were overweight, and 30.8% had obesity, but the average weight varied considerably between countries and sites.
Increased need for respiratory support, same mortality risk
Compared with patients with normal weight, patients who were overweight had a 44% increased risk of needing supplemental oxygen/noninvasive ventilation, and those with obesity had a 75% increased risk of this, after adjustment for age (< 65, ≥ 65), sex, hypertension, diabetes, or preexisting cardiovascular disease or respiratory conditions.
Patients who were overweight had a 22% increased risk of needing invasive (mechanical) ventilation, and those with obesity had a 73% increased risk of this, after multivariable adjustment.
Being overweight or having obesity was not associated with a significantly increased risk of dying in the hospital, however.
“In other viral respiratory infections, such as influenza, there is a similar pattern of increased requirement for ventilatory support but lower in-hospital mortality among individuals with obesity, when compared to those with normal range BMI,” Dr. Longmore noted. She said that larger studies are needed to further explore this finding regarding COVID-19.
Compared to patients without diabetes, those with diabetes had a 21% increased risk of requiring invasive ventilation, but they did not have an increased risk of needing noninvasive ventilation or of dying in the hospital.
As in previous studies, individuals who had cardiovascular and preexisting respiratory diseases were not at greater risk of needing oxygen or mechanical ventilation but were at increased risk for in-hospital death. Men had a greater risk of needing invasive mechanical ventilation, and individuals who were older than 65 had an increased risk of requiring oxygen or of dying in the hospital.
A living meta-analysis, call for more collaborators
“We consider this a ‘living meta-analysis’ and invite other centers to join us,” Dr. Longmore said. “We hope to update the analyses as more data are contributed.”
No specific project funded the study. The authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
In a global meta-analysis of more than 7,000 patients who were hospitalized with COVID-19, individuals with overweight or obesity were more likely to need respiratory support but were not more likely to die in the hospital, compared to individuals of normal weight.
Compared to patients without diabetes, those with diabetes had higher odds of needing invasive respiratory support (with intubation) but not for needing noninvasive respiratory support or of dying in the hospital.
“Surprisingly,” among patients with diabetes, being overweight or having obesity did not further increase the odds of any of these outcomes, the researchers wrote. The finding needs to be confirmed in larger studies, they said, because the sample sizes in these subanalyses were small and the confidence intervals were large.
The study by Danielle K. Longmore, PhD, of Murdoch Children’s Research Institute (MCRI), Melbourne, and colleagues from the International BMI-COVID consortium, was published online April 15 in Diabetes Care.
This new research “adds to the known data on the associations between obesity and severe COVID-19 disease and extends these findings” to patients who are overweight and/or have diabetes, Dr. Longmore, a pediatric endocrinologist with a clinical and research interest in childhood and youth obesity, said in an interview.
Immunologist Siroon Bekkering, PhD, of Radboud University Medical Center, Nijmegen, the Netherlands, explained that never before have so much data of different types regarding obesity been combined in one large study. Dr. Bekkering is a coauthor of the article and was a principal investigator.
“Several national and international observations already showed the important role of overweight and obesity in a more severe COVID-19 course. This study adds to those observations by combining data from several countries with the possibility to look at the risk factors separately,” she said in a statement from her institution.
“Regardless of other risk factors (such as heart disease or diabetes), we now see that too high a BMI [body mass index] can actually lead to a more severe course in [coronavirus] infection,” she said.
Study implications: Data show that overweight, obesity add to risk
These latest findings highlight the urgent need to develop public health policies to address socioeconomic and psychological drivers of obesity, Dr. Longmore said.
“Although taking steps to address obesity in the short term is unlikely to have an immediate impact in the COVID-19 pandemic, it will likely reduce the disease burden in future viral pandemics and reduce risks of complications like heart disease and stroke,” she observed in a statement issued by MCRI.
Coauthor Kirsty R. Short, PhD, a research fellow at the University of Queensland, Brisbane, Australia, noted that “obesity is associated with numerous poor health outcomes, including increased risk of cardiometabolic and respiratory disease and more severe viral disease including influenza, dengue, and SARS-CoV-1.
“Given the large scale of this study,” she said, “we have conclusively shown that being overweight or obese are independent risk factors for worse outcomes in adults hospitalized with COVID-19.”
“At the moment, the World Health Organization has not had enough high-quality data to include being overweight or obese as a risk factor for severe COVID-19 disease,” added another author, David P. Burgner, PhD, a pediatric infectious diseases clinician scientist from MCRI.
“Our study should help inform decisions about which higher-risk groups should be vaccinated as a priority,” he observed.
Does being overweight up risk of worse COVID-19 outcomes?
About 13% of the world’s population are overweight, and 40% have obesity. There are wide between-country variations in these data, and about 90% of patients with type 2 diabetes are overweight or obese, the researchers noted.
The Organisation for Economic Co-operation and Development reported that the prevalence of obesity in 2016-2017 was 5.7% to 8.9% in Asia, 9.8% to 16.8% in Europe, 26.5% in South Africa, and 40.0% in the United States, they added.
Obesity is common and has emerged as an important risk factor for severe COVID-19. However, most previous studies of COVID-19 and elevated BMI were conducted in single centers and did not focus on patients with overweight.
To investigate, the researchers identified 7,244 patients (two-thirds were overweight or obese) who were hospitalized with COVID-19 in 69 hospitals (18 sites) in 11 countries from Jan. 17, 2020, to June 2, 2020.
Most patients were hospitalized with COVID-19 in the Netherlands (2,260), followed by New York City (1,682), Switzerland (920), St. Louis (805), Norway, Italy, China, South Africa, Indonesia, Denmark, Los Angeles, Austria, and Singapore.
Just over half (60%) of the individuals were male, and 52% were older than 65.
Overall, 34.8% were overweight, and 30.8% had obesity, but the average weight varied considerably between countries and sites.
Increased need for respiratory support, same mortality risk
Compared with patients with normal weight, patients who were overweight had a 44% increased risk of needing supplemental oxygen/noninvasive ventilation, and those with obesity had a 75% increased risk of this, after adjustment for age (< 65, ≥ 65), sex, hypertension, diabetes, or preexisting cardiovascular disease or respiratory conditions.
Patients who were overweight had a 22% increased risk of needing invasive (mechanical) ventilation, and those with obesity had a 73% increased risk of this, after multivariable adjustment.
Being overweight or having obesity was not associated with a significantly increased risk of dying in the hospital, however.
“In other viral respiratory infections, such as influenza, there is a similar pattern of increased requirement for ventilatory support but lower in-hospital mortality among individuals with obesity, when compared to those with normal range BMI,” Dr. Longmore noted. She said that larger studies are needed to further explore this finding regarding COVID-19.
Compared to patients without diabetes, those with diabetes had a 21% increased risk of requiring invasive ventilation, but they did not have an increased risk of needing noninvasive ventilation or of dying in the hospital.
As in previous studies, individuals who had cardiovascular and preexisting respiratory diseases were not at greater risk of needing oxygen or mechanical ventilation but were at increased risk for in-hospital death. Men had a greater risk of needing invasive mechanical ventilation, and individuals who were older than 65 had an increased risk of requiring oxygen or of dying in the hospital.
A living meta-analysis, call for more collaborators
“We consider this a ‘living meta-analysis’ and invite other centers to join us,” Dr. Longmore said. “We hope to update the analyses as more data are contributed.”
No specific project funded the study. The authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
In a global meta-analysis of more than 7,000 patients who were hospitalized with COVID-19, individuals with overweight or obesity were more likely to need respiratory support but were not more likely to die in the hospital, compared to individuals of normal weight.
Compared to patients without diabetes, those with diabetes had higher odds of needing invasive respiratory support (with intubation) but not for needing noninvasive respiratory support or of dying in the hospital.
“Surprisingly,” among patients with diabetes, being overweight or having obesity did not further increase the odds of any of these outcomes, the researchers wrote. The finding needs to be confirmed in larger studies, they said, because the sample sizes in these subanalyses were small and the confidence intervals were large.
The study by Danielle K. Longmore, PhD, of Murdoch Children’s Research Institute (MCRI), Melbourne, and colleagues from the International BMI-COVID consortium, was published online April 15 in Diabetes Care.
This new research “adds to the known data on the associations between obesity and severe COVID-19 disease and extends these findings” to patients who are overweight and/or have diabetes, Dr. Longmore, a pediatric endocrinologist with a clinical and research interest in childhood and youth obesity, said in an interview.
Immunologist Siroon Bekkering, PhD, of Radboud University Medical Center, Nijmegen, the Netherlands, explained that never before have so much data of different types regarding obesity been combined in one large study. Dr. Bekkering is a coauthor of the article and was a principal investigator.
“Several national and international observations already showed the important role of overweight and obesity in a more severe COVID-19 course. This study adds to those observations by combining data from several countries with the possibility to look at the risk factors separately,” she said in a statement from her institution.
“Regardless of other risk factors (such as heart disease or diabetes), we now see that too high a BMI [body mass index] can actually lead to a more severe course in [coronavirus] infection,” she said.
Study implications: Data show that overweight, obesity add to risk
These latest findings highlight the urgent need to develop public health policies to address socioeconomic and psychological drivers of obesity, Dr. Longmore said.
“Although taking steps to address obesity in the short term is unlikely to have an immediate impact in the COVID-19 pandemic, it will likely reduce the disease burden in future viral pandemics and reduce risks of complications like heart disease and stroke,” she observed in a statement issued by MCRI.
Coauthor Kirsty R. Short, PhD, a research fellow at the University of Queensland, Brisbane, Australia, noted that “obesity is associated with numerous poor health outcomes, including increased risk of cardiometabolic and respiratory disease and more severe viral disease including influenza, dengue, and SARS-CoV-1.
“Given the large scale of this study,” she said, “we have conclusively shown that being overweight or obese are independent risk factors for worse outcomes in adults hospitalized with COVID-19.”
“At the moment, the World Health Organization has not had enough high-quality data to include being overweight or obese as a risk factor for severe COVID-19 disease,” added another author, David P. Burgner, PhD, a pediatric infectious diseases clinician scientist from MCRI.
“Our study should help inform decisions about which higher-risk groups should be vaccinated as a priority,” he observed.
Does being overweight up risk of worse COVID-19 outcomes?
About 13% of the world’s population are overweight, and 40% have obesity. There are wide between-country variations in these data, and about 90% of patients with type 2 diabetes are overweight or obese, the researchers noted.
The Organisation for Economic Co-operation and Development reported that the prevalence of obesity in 2016-2017 was 5.7% to 8.9% in Asia, 9.8% to 16.8% in Europe, 26.5% in South Africa, and 40.0% in the United States, they added.
Obesity is common and has emerged as an important risk factor for severe COVID-19. However, most previous studies of COVID-19 and elevated BMI were conducted in single centers and did not focus on patients with overweight.
To investigate, the researchers identified 7,244 patients (two-thirds were overweight or obese) who were hospitalized with COVID-19 in 69 hospitals (18 sites) in 11 countries from Jan. 17, 2020, to June 2, 2020.
Most patients were hospitalized with COVID-19 in the Netherlands (2,260), followed by New York City (1,682), Switzerland (920), St. Louis (805), Norway, Italy, China, South Africa, Indonesia, Denmark, Los Angeles, Austria, and Singapore.
Just over half (60%) of the individuals were male, and 52% were older than 65.
Overall, 34.8% were overweight, and 30.8% had obesity, but the average weight varied considerably between countries and sites.
Increased need for respiratory support, same mortality risk
Compared with patients with normal weight, patients who were overweight had a 44% increased risk of needing supplemental oxygen/noninvasive ventilation, and those with obesity had a 75% increased risk of this, after adjustment for age (< 65, ≥ 65), sex, hypertension, diabetes, or preexisting cardiovascular disease or respiratory conditions.
Patients who were overweight had a 22% increased risk of needing invasive (mechanical) ventilation, and those with obesity had a 73% increased risk of this, after multivariable adjustment.
Being overweight or having obesity was not associated with a significantly increased risk of dying in the hospital, however.
“In other viral respiratory infections, such as influenza, there is a similar pattern of increased requirement for ventilatory support but lower in-hospital mortality among individuals with obesity, when compared to those with normal range BMI,” Dr. Longmore noted. She said that larger studies are needed to further explore this finding regarding COVID-19.
Compared to patients without diabetes, those with diabetes had a 21% increased risk of requiring invasive ventilation, but they did not have an increased risk of needing noninvasive ventilation or of dying in the hospital.
As in previous studies, individuals who had cardiovascular and preexisting respiratory diseases were not at greater risk of needing oxygen or mechanical ventilation but were at increased risk for in-hospital death. Men had a greater risk of needing invasive mechanical ventilation, and individuals who were older than 65 had an increased risk of requiring oxygen or of dying in the hospital.
A living meta-analysis, call for more collaborators
“We consider this a ‘living meta-analysis’ and invite other centers to join us,” Dr. Longmore said. “We hope to update the analyses as more data are contributed.”
No specific project funded the study. The authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.





