User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
nav[contains(@class, 'nav-ce-stack nav-ce-stack__large-screen')]
header[@id='header']
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'main-prefix')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
div[contains(@class, 'view-medstat-quiz-listing-panes')]
div[contains(@class, 'pane-article-sidebar-latest-news')]
Apixaban outmatches rivaroxaban in patients with AFib and valvular heart disease
Compared with rivaroxaban, apixaban cut risks nearly in half, suggesting that clinicians should consider these new data when choosing an anticoagulant, reported lead author Ghadeer K. Dawwas, PhD, of the University of Pennsylvania, Philadelphia, and colleagues.
In the new retrospective study involving almost 20,000 patients, Dr. Dawwas and her colleagues “emulated a target trial” using private insurance claims from Optum’s deidentified Clinformatics Data Mart Database. The cohort was narrowed from a screened population of 58,210 patients with concurrent AFib and VHD to 9,947 new apixaban users who could be closely matched with 9,947 new rivaroxaban users. Covariates included provider specialty, type of VHD, demographic characteristics, measures of health care use, baseline use of medications, and baseline comorbidities.
The primary effectiveness outcome was a composite of systemic embolism and ischemic stroke, while the primary safety outcome was a composite of intracranial or gastrointestinal bleeding.
“Although several ongoing trials aim to compare apixaban with warfarin in patients with AFib and VHD, none of these trials will directly compare apixaban and rivaroxaban,” the investigators wrote. Their report is in Annals of Internal Medicine.
Dr. Dawwas and colleagues previously showed that direct oral anticoagulants (DOACs) were safer and more effective than warfarin in the same patient population. Comparing apixaban and rivaroxaban – the two most common DOACs – was the next logical step, Dr. Dawwas said in an interview.
Study results
Compared with rivaroxaban, patients who received apixaban had a 43% reduced risk of stroke or embolism (hazard ratio [HR], 0.57; 95% confidence interval [CI], 0.40-0.80). Apixaban’s ability to protect against bleeding appeared even more pronounced, with a 49% reduced risk over rivaroxaban (HR, 0.51; 95% CI, 0.41-0.62).
Comparing the two agents on an absolute basis, apixaban reduced risk of embolism or stroke by 0.2% within the first 6 months of treatment initiation, and 1.1% within the first year of initiation. At the same time points, absolute risk reductions for bleeding were 1.2% and 1.9%, respectively.
The investigators noted that their results held consistent in an alternative analysis that considered separate types of VHD.
“Based on the results from our analysis, we showed that apixaban is effective and safe in patients with atrial fibrillation and valvular heart diseases,” Dr. Dawwas said.
Head-to-head trial needed to change practice
Christopher M. Bianco, DO, associate professor of medicine at West Virginia University Heart and Vascular Institute, Morgantown, said the findings “add to the growing body of literature,” but “a head-to-head trial would be necessary to make a definitive change to clinical practice.”
Dr. Bianco, who recently conducted a retrospective analysis of apixaban and rivaroxaban that found no difference in safety and efficacy among a different patient population, said these kinds of studies are helpful in generating hypotheses, but they can’t account for all relevant clinical factors.
“There are just so many things that go into the decision-making process of [prescribing] apixaban and rivaroxaban,” he said. “Even though [Dr. Dawwas and colleagues] used propensity matching, you’re never going to be able to sort that out with a retrospective analysis.”
Specifically, Dr. Bianco noted that the findings did not include dose data. This is a key gap, he said, considering how often real-world datasets have shown that providers underdose DOACs for a number of unaccountable reasons, and how frequently patients exhibit poor adherence.
The study also lacked detail concerning the degree of renal dysfunction, which can determine drug eligibility, Dr. Bianco said. Furthermore, attempts to stratify patients based on thrombosis and bleeding risk were likely “insufficient,” he added.
Dr. Bianco also cautioned that the investigators defined valvular heart disease as any valve-related disease of any severity. In contrast, previous studies have generally restricted valvular heart disease to patients with mitral stenosis or prosthetic valves.
“This is definitely not the traditional definition of valvular heart disease, so the title is a little bit misleading in that sense, although they certainly do disclose that in the methods,” Dr. Bianco said.
On a more positive note, he highlighted the size of the patient population, and the real-world data, which included many patients who would be excluded from clinical trials.
More broadly, the study helps drive research forward, Dr. Bianco concluded; namely, by attracting financial support for a more powerful head-to-head trial that drug makers are unlikely to fund due to inherent market risk.
This study was supported by the National Institutes of Health. The investigators disclosed additional relationships with Takeda, Spark, Sanofi, and others. Dr. Bianco disclosed no conflicts of interest.
Compared with rivaroxaban, apixaban cut risks nearly in half, suggesting that clinicians should consider these new data when choosing an anticoagulant, reported lead author Ghadeer K. Dawwas, PhD, of the University of Pennsylvania, Philadelphia, and colleagues.
In the new retrospective study involving almost 20,000 patients, Dr. Dawwas and her colleagues “emulated a target trial” using private insurance claims from Optum’s deidentified Clinformatics Data Mart Database. The cohort was narrowed from a screened population of 58,210 patients with concurrent AFib and VHD to 9,947 new apixaban users who could be closely matched with 9,947 new rivaroxaban users. Covariates included provider specialty, type of VHD, demographic characteristics, measures of health care use, baseline use of medications, and baseline comorbidities.
The primary effectiveness outcome was a composite of systemic embolism and ischemic stroke, while the primary safety outcome was a composite of intracranial or gastrointestinal bleeding.
“Although several ongoing trials aim to compare apixaban with warfarin in patients with AFib and VHD, none of these trials will directly compare apixaban and rivaroxaban,” the investigators wrote. Their report is in Annals of Internal Medicine.
Dr. Dawwas and colleagues previously showed that direct oral anticoagulants (DOACs) were safer and more effective than warfarin in the same patient population. Comparing apixaban and rivaroxaban – the two most common DOACs – was the next logical step, Dr. Dawwas said in an interview.
Study results
Compared with rivaroxaban, patients who received apixaban had a 43% reduced risk of stroke or embolism (hazard ratio [HR], 0.57; 95% confidence interval [CI], 0.40-0.80). Apixaban’s ability to protect against bleeding appeared even more pronounced, with a 49% reduced risk over rivaroxaban (HR, 0.51; 95% CI, 0.41-0.62).
Comparing the two agents on an absolute basis, apixaban reduced risk of embolism or stroke by 0.2% within the first 6 months of treatment initiation, and 1.1% within the first year of initiation. At the same time points, absolute risk reductions for bleeding were 1.2% and 1.9%, respectively.
The investigators noted that their results held consistent in an alternative analysis that considered separate types of VHD.
“Based on the results from our analysis, we showed that apixaban is effective and safe in patients with atrial fibrillation and valvular heart diseases,” Dr. Dawwas said.
Head-to-head trial needed to change practice
Christopher M. Bianco, DO, associate professor of medicine at West Virginia University Heart and Vascular Institute, Morgantown, said the findings “add to the growing body of literature,” but “a head-to-head trial would be necessary to make a definitive change to clinical practice.”
Dr. Bianco, who recently conducted a retrospective analysis of apixaban and rivaroxaban that found no difference in safety and efficacy among a different patient population, said these kinds of studies are helpful in generating hypotheses, but they can’t account for all relevant clinical factors.
“There are just so many things that go into the decision-making process of [prescribing] apixaban and rivaroxaban,” he said. “Even though [Dr. Dawwas and colleagues] used propensity matching, you’re never going to be able to sort that out with a retrospective analysis.”
Specifically, Dr. Bianco noted that the findings did not include dose data. This is a key gap, he said, considering how often real-world datasets have shown that providers underdose DOACs for a number of unaccountable reasons, and how frequently patients exhibit poor adherence.
The study also lacked detail concerning the degree of renal dysfunction, which can determine drug eligibility, Dr. Bianco said. Furthermore, attempts to stratify patients based on thrombosis and bleeding risk were likely “insufficient,” he added.
Dr. Bianco also cautioned that the investigators defined valvular heart disease as any valve-related disease of any severity. In contrast, previous studies have generally restricted valvular heart disease to patients with mitral stenosis or prosthetic valves.
“This is definitely not the traditional definition of valvular heart disease, so the title is a little bit misleading in that sense, although they certainly do disclose that in the methods,” Dr. Bianco said.
On a more positive note, he highlighted the size of the patient population, and the real-world data, which included many patients who would be excluded from clinical trials.
More broadly, the study helps drive research forward, Dr. Bianco concluded; namely, by attracting financial support for a more powerful head-to-head trial that drug makers are unlikely to fund due to inherent market risk.
This study was supported by the National Institutes of Health. The investigators disclosed additional relationships with Takeda, Spark, Sanofi, and others. Dr. Bianco disclosed no conflicts of interest.
Compared with rivaroxaban, apixaban cut risks nearly in half, suggesting that clinicians should consider these new data when choosing an anticoagulant, reported lead author Ghadeer K. Dawwas, PhD, of the University of Pennsylvania, Philadelphia, and colleagues.
In the new retrospective study involving almost 20,000 patients, Dr. Dawwas and her colleagues “emulated a target trial” using private insurance claims from Optum’s deidentified Clinformatics Data Mart Database. The cohort was narrowed from a screened population of 58,210 patients with concurrent AFib and VHD to 9,947 new apixaban users who could be closely matched with 9,947 new rivaroxaban users. Covariates included provider specialty, type of VHD, demographic characteristics, measures of health care use, baseline use of medications, and baseline comorbidities.
The primary effectiveness outcome was a composite of systemic embolism and ischemic stroke, while the primary safety outcome was a composite of intracranial or gastrointestinal bleeding.
“Although several ongoing trials aim to compare apixaban with warfarin in patients with AFib and VHD, none of these trials will directly compare apixaban and rivaroxaban,” the investigators wrote. Their report is in Annals of Internal Medicine.
Dr. Dawwas and colleagues previously showed that direct oral anticoagulants (DOACs) were safer and more effective than warfarin in the same patient population. Comparing apixaban and rivaroxaban – the two most common DOACs – was the next logical step, Dr. Dawwas said in an interview.
Study results
Compared with rivaroxaban, patients who received apixaban had a 43% reduced risk of stroke or embolism (hazard ratio [HR], 0.57; 95% confidence interval [CI], 0.40-0.80). Apixaban’s ability to protect against bleeding appeared even more pronounced, with a 49% reduced risk over rivaroxaban (HR, 0.51; 95% CI, 0.41-0.62).
Comparing the two agents on an absolute basis, apixaban reduced risk of embolism or stroke by 0.2% within the first 6 months of treatment initiation, and 1.1% within the first year of initiation. At the same time points, absolute risk reductions for bleeding were 1.2% and 1.9%, respectively.
The investigators noted that their results held consistent in an alternative analysis that considered separate types of VHD.
“Based on the results from our analysis, we showed that apixaban is effective and safe in patients with atrial fibrillation and valvular heart diseases,” Dr. Dawwas said.
Head-to-head trial needed to change practice
Christopher M. Bianco, DO, associate professor of medicine at West Virginia University Heart and Vascular Institute, Morgantown, said the findings “add to the growing body of literature,” but “a head-to-head trial would be necessary to make a definitive change to clinical practice.”
Dr. Bianco, who recently conducted a retrospective analysis of apixaban and rivaroxaban that found no difference in safety and efficacy among a different patient population, said these kinds of studies are helpful in generating hypotheses, but they can’t account for all relevant clinical factors.
“There are just so many things that go into the decision-making process of [prescribing] apixaban and rivaroxaban,” he said. “Even though [Dr. Dawwas and colleagues] used propensity matching, you’re never going to be able to sort that out with a retrospective analysis.”
Specifically, Dr. Bianco noted that the findings did not include dose data. This is a key gap, he said, considering how often real-world datasets have shown that providers underdose DOACs for a number of unaccountable reasons, and how frequently patients exhibit poor adherence.
The study also lacked detail concerning the degree of renal dysfunction, which can determine drug eligibility, Dr. Bianco said. Furthermore, attempts to stratify patients based on thrombosis and bleeding risk were likely “insufficient,” he added.
Dr. Bianco also cautioned that the investigators defined valvular heart disease as any valve-related disease of any severity. In contrast, previous studies have generally restricted valvular heart disease to patients with mitral stenosis or prosthetic valves.
“This is definitely not the traditional definition of valvular heart disease, so the title is a little bit misleading in that sense, although they certainly do disclose that in the methods,” Dr. Bianco said.
On a more positive note, he highlighted the size of the patient population, and the real-world data, which included many patients who would be excluded from clinical trials.
More broadly, the study helps drive research forward, Dr. Bianco concluded; namely, by attracting financial support for a more powerful head-to-head trial that drug makers are unlikely to fund due to inherent market risk.
This study was supported by the National Institutes of Health. The investigators disclosed additional relationships with Takeda, Spark, Sanofi, and others. Dr. Bianco disclosed no conflicts of interest.
FROM ANNALS OF INTERNAL MEDICINE
‘Too good to be true’? Ultrasound safely treats kidney stones
Treatment with transcutaneous, focused ultrasound safely led to the repositioning and rupture of a majority of stones in the ureter when tested in 29 people at two U.S. centers in the first human feasibility study of the technology.*
potentially relieving pain and facilitating passage in awake patients,” write M. Kennedy Hall, MD, an emergency medicine physician at the University of Washington School of Medicine, Seattle, and co-authors in a report published in the Journal of Urology.
“This is the first human trial in awake subjects” of this method for nonsurgically facilitating ureteral stone clearance, and the results of limited patient discomfort and stone motion in 66% of treated patients seem “almost too good to be true,” comments Karen L. Stern, MD, a urologist at the Mayo Clinic, Phoenix, in an accompanying editorial.
However, Dr. Stern notes two study limitations. First, to correctly target the focused ultrasound clinicians first need to visualize a stone with ultrasound. However, “most urologists are not experienced in ultrasound,” a limitation that could mean the treatment may be more likely performed by emergency physicians or radiologists in the future.
Second, the study had no control patients, and Dr. Stern cautions that the clinical significance of BWL cannot be definitively assessed without a randomized trial that directly compares the new method with either spontaneous stone passage or medical expulsion therapy.
“The authors showed a distal ureteral stone passage [rate] of 81%, but those stones passed within days, not minutes, of the procedure, and that rate is not too far off published data on spontaneous passage,” Dr. Stern notes in her editorial.*
Despite these caveats, Dr. Stern calls the potential for BWL “immense.”
Ultrasound for 10 minutes or less
The study enrolled adults who presented to the University of Washington emergency department or endo-urology clinic with a proximal or distal ureteral stone. Twenty-nine awake, unanesthetized patients in whom clinicians had an unobstructed view of a stone in the focal zone received ultrasound treatment performed by trained personnel.
Treatment involved ultrasound bursts of up to 3 seconds for stone propulsion and 30 seconds at a time for BWL. Total ultrasound exposure could not exceed 10 minutes, and no patient underwent more than one treatment. Sixteen patients received treatment for propulsion only, and 13 received both propulsion and BWL treatments.
The primary outcome was stone motion, which occurred in 19 of the 29 patients (66%). A prespecified secondary outcome was passage of the stone in the 26 patients with distal stones. Among the 21 patients with at least 14 days of follow-up, passage occurred in 18 (86%) after an average of 3.9 days following treatment. The researchers also confirmed stone fragmentation in five of the 13 patients (38%) who received BWL treatment.
The researchers infer that ultrasound treatment facilitated stone passage by stone propulsion, fragmentation, and peristalsis.
Mathew D. Sorensen, MD, a study co-investigator, likened the effect of ultrasound on the stones to a leaf blower on garden debris.
“Essentially, we use the acoustic energy of ultrasound, which gets focused on the stone and creates movement, like a blower,” said Dr. Sorensen, a urologist at the University of Washington School of Medicine. “A push of energy lasts a second or two, sort of a sweeping movement to try to get fragments to move out of what’s usually the bottom of the kidney, toward the exit. Because the energy moves in one way, away from the probe, it’s sort of like playing pool,” he explained.
All 29 treated patients in the current study tolerated the procedure, and none experienced unanticipated events. No patients needed to visit the emergency department or an intervention because of the treatment, and there were no observed ureter injuries.
Pain scores dropped significantly, procedure is ‘nearly painless’
Assessment of pain scores by each patient before and after treatment showed overall significant decreases in both mean and median scores, with 10 patients reporting decreased pain and two reporting increased pain.
When discomfort occurred during the procedure it was described by patients as either similar to a pinprick or as a referred sensation to pass urine and the stone. These effects occurred during 18 of 820 (2%) total propulsive bursts of ultrasound administered.
“It’s nearly painless, and you can do it while the patient is awake, and without sedation, which is critical,” said Dr. Hall in a statement from the University of Washington School of Medicine. He envisions eventually performing the procedure in a clinic or emergency department setting.
All three patients with a proximal stone and three patients with a distal stone who did not pass their stone after treatment underwent surgical stone removal.
The researchers acknowledge that a lack of a control group was a study limitation. They cite a historical, spontaneous stone passage rate of 54% included in a 2016 guideline from the American Urological Association and based on a meta-analysis of 27 studies with 1,205 total patients.
The study was funded by the United States National Aeronautics and Space Administration, which is interested in having a technology available to treat ureteral stones that may develop during prolonged space travel.
The study received no commercial funding. Four of the study’s 23 authors, including Dr. Sorensen, are consultants to and hold equity in SonoMotion, a company that has licensed the tested technology from the University of Washington for commercial development. The report did not identify a specific manufacturer of the ultrasound equipment used for treatment.
A version of this article first appeared on Medscape.com.
*Correction, 10/21/22: An earlier version of this article misstated the location of the stones. They were in the ureter.
Treatment with transcutaneous, focused ultrasound safely led to the repositioning and rupture of a majority of stones in the ureter when tested in 29 people at two U.S. centers in the first human feasibility study of the technology.*
potentially relieving pain and facilitating passage in awake patients,” write M. Kennedy Hall, MD, an emergency medicine physician at the University of Washington School of Medicine, Seattle, and co-authors in a report published in the Journal of Urology.
“This is the first human trial in awake subjects” of this method for nonsurgically facilitating ureteral stone clearance, and the results of limited patient discomfort and stone motion in 66% of treated patients seem “almost too good to be true,” comments Karen L. Stern, MD, a urologist at the Mayo Clinic, Phoenix, in an accompanying editorial.
However, Dr. Stern notes two study limitations. First, to correctly target the focused ultrasound clinicians first need to visualize a stone with ultrasound. However, “most urologists are not experienced in ultrasound,” a limitation that could mean the treatment may be more likely performed by emergency physicians or radiologists in the future.
Second, the study had no control patients, and Dr. Stern cautions that the clinical significance of BWL cannot be definitively assessed without a randomized trial that directly compares the new method with either spontaneous stone passage or medical expulsion therapy.
“The authors showed a distal ureteral stone passage [rate] of 81%, but those stones passed within days, not minutes, of the procedure, and that rate is not too far off published data on spontaneous passage,” Dr. Stern notes in her editorial.*
Despite these caveats, Dr. Stern calls the potential for BWL “immense.”
Ultrasound for 10 minutes or less
The study enrolled adults who presented to the University of Washington emergency department or endo-urology clinic with a proximal or distal ureteral stone. Twenty-nine awake, unanesthetized patients in whom clinicians had an unobstructed view of a stone in the focal zone received ultrasound treatment performed by trained personnel.
Treatment involved ultrasound bursts of up to 3 seconds for stone propulsion and 30 seconds at a time for BWL. Total ultrasound exposure could not exceed 10 minutes, and no patient underwent more than one treatment. Sixteen patients received treatment for propulsion only, and 13 received both propulsion and BWL treatments.
The primary outcome was stone motion, which occurred in 19 of the 29 patients (66%). A prespecified secondary outcome was passage of the stone in the 26 patients with distal stones. Among the 21 patients with at least 14 days of follow-up, passage occurred in 18 (86%) after an average of 3.9 days following treatment. The researchers also confirmed stone fragmentation in five of the 13 patients (38%) who received BWL treatment.
The researchers infer that ultrasound treatment facilitated stone passage by stone propulsion, fragmentation, and peristalsis.
Mathew D. Sorensen, MD, a study co-investigator, likened the effect of ultrasound on the stones to a leaf blower on garden debris.
“Essentially, we use the acoustic energy of ultrasound, which gets focused on the stone and creates movement, like a blower,” said Dr. Sorensen, a urologist at the University of Washington School of Medicine. “A push of energy lasts a second or two, sort of a sweeping movement to try to get fragments to move out of what’s usually the bottom of the kidney, toward the exit. Because the energy moves in one way, away from the probe, it’s sort of like playing pool,” he explained.
All 29 treated patients in the current study tolerated the procedure, and none experienced unanticipated events. No patients needed to visit the emergency department or an intervention because of the treatment, and there were no observed ureter injuries.
Pain scores dropped significantly, procedure is ‘nearly painless’
Assessment of pain scores by each patient before and after treatment showed overall significant decreases in both mean and median scores, with 10 patients reporting decreased pain and two reporting increased pain.
When discomfort occurred during the procedure it was described by patients as either similar to a pinprick or as a referred sensation to pass urine and the stone. These effects occurred during 18 of 820 (2%) total propulsive bursts of ultrasound administered.
“It’s nearly painless, and you can do it while the patient is awake, and without sedation, which is critical,” said Dr. Hall in a statement from the University of Washington School of Medicine. He envisions eventually performing the procedure in a clinic or emergency department setting.
All three patients with a proximal stone and three patients with a distal stone who did not pass their stone after treatment underwent surgical stone removal.
The researchers acknowledge that a lack of a control group was a study limitation. They cite a historical, spontaneous stone passage rate of 54% included in a 2016 guideline from the American Urological Association and based on a meta-analysis of 27 studies with 1,205 total patients.
The study was funded by the United States National Aeronautics and Space Administration, which is interested in having a technology available to treat ureteral stones that may develop during prolonged space travel.
The study received no commercial funding. Four of the study’s 23 authors, including Dr. Sorensen, are consultants to and hold equity in SonoMotion, a company that has licensed the tested technology from the University of Washington for commercial development. The report did not identify a specific manufacturer of the ultrasound equipment used for treatment.
A version of this article first appeared on Medscape.com.
*Correction, 10/21/22: An earlier version of this article misstated the location of the stones. They were in the ureter.
Treatment with transcutaneous, focused ultrasound safely led to the repositioning and rupture of a majority of stones in the ureter when tested in 29 people at two U.S. centers in the first human feasibility study of the technology.*
potentially relieving pain and facilitating passage in awake patients,” write M. Kennedy Hall, MD, an emergency medicine physician at the University of Washington School of Medicine, Seattle, and co-authors in a report published in the Journal of Urology.
“This is the first human trial in awake subjects” of this method for nonsurgically facilitating ureteral stone clearance, and the results of limited patient discomfort and stone motion in 66% of treated patients seem “almost too good to be true,” comments Karen L. Stern, MD, a urologist at the Mayo Clinic, Phoenix, in an accompanying editorial.
However, Dr. Stern notes two study limitations. First, to correctly target the focused ultrasound clinicians first need to visualize a stone with ultrasound. However, “most urologists are not experienced in ultrasound,” a limitation that could mean the treatment may be more likely performed by emergency physicians or radiologists in the future.
Second, the study had no control patients, and Dr. Stern cautions that the clinical significance of BWL cannot be definitively assessed without a randomized trial that directly compares the new method with either spontaneous stone passage or medical expulsion therapy.
“The authors showed a distal ureteral stone passage [rate] of 81%, but those stones passed within days, not minutes, of the procedure, and that rate is not too far off published data on spontaneous passage,” Dr. Stern notes in her editorial.*
Despite these caveats, Dr. Stern calls the potential for BWL “immense.”
Ultrasound for 10 minutes or less
The study enrolled adults who presented to the University of Washington emergency department or endo-urology clinic with a proximal or distal ureteral stone. Twenty-nine awake, unanesthetized patients in whom clinicians had an unobstructed view of a stone in the focal zone received ultrasound treatment performed by trained personnel.
Treatment involved ultrasound bursts of up to 3 seconds for stone propulsion and 30 seconds at a time for BWL. Total ultrasound exposure could not exceed 10 minutes, and no patient underwent more than one treatment. Sixteen patients received treatment for propulsion only, and 13 received both propulsion and BWL treatments.
The primary outcome was stone motion, which occurred in 19 of the 29 patients (66%). A prespecified secondary outcome was passage of the stone in the 26 patients with distal stones. Among the 21 patients with at least 14 days of follow-up, passage occurred in 18 (86%) after an average of 3.9 days following treatment. The researchers also confirmed stone fragmentation in five of the 13 patients (38%) who received BWL treatment.
The researchers infer that ultrasound treatment facilitated stone passage by stone propulsion, fragmentation, and peristalsis.
Mathew D. Sorensen, MD, a study co-investigator, likened the effect of ultrasound on the stones to a leaf blower on garden debris.
“Essentially, we use the acoustic energy of ultrasound, which gets focused on the stone and creates movement, like a blower,” said Dr. Sorensen, a urologist at the University of Washington School of Medicine. “A push of energy lasts a second or two, sort of a sweeping movement to try to get fragments to move out of what’s usually the bottom of the kidney, toward the exit. Because the energy moves in one way, away from the probe, it’s sort of like playing pool,” he explained.
All 29 treated patients in the current study tolerated the procedure, and none experienced unanticipated events. No patients needed to visit the emergency department or an intervention because of the treatment, and there were no observed ureter injuries.
Pain scores dropped significantly, procedure is ‘nearly painless’
Assessment of pain scores by each patient before and after treatment showed overall significant decreases in both mean and median scores, with 10 patients reporting decreased pain and two reporting increased pain.
When discomfort occurred during the procedure it was described by patients as either similar to a pinprick or as a referred sensation to pass urine and the stone. These effects occurred during 18 of 820 (2%) total propulsive bursts of ultrasound administered.
“It’s nearly painless, and you can do it while the patient is awake, and without sedation, which is critical,” said Dr. Hall in a statement from the University of Washington School of Medicine. He envisions eventually performing the procedure in a clinic or emergency department setting.
All three patients with a proximal stone and three patients with a distal stone who did not pass their stone after treatment underwent surgical stone removal.
The researchers acknowledge that a lack of a control group was a study limitation. They cite a historical, spontaneous stone passage rate of 54% included in a 2016 guideline from the American Urological Association and based on a meta-analysis of 27 studies with 1,205 total patients.
The study was funded by the United States National Aeronautics and Space Administration, which is interested in having a technology available to treat ureteral stones that may develop during prolonged space travel.
The study received no commercial funding. Four of the study’s 23 authors, including Dr. Sorensen, are consultants to and hold equity in SonoMotion, a company that has licensed the tested technology from the University of Washington for commercial development. The report did not identify a specific manufacturer of the ultrasound equipment used for treatment.
A version of this article first appeared on Medscape.com.
*Correction, 10/21/22: An earlier version of this article misstated the location of the stones. They were in the ureter.
FROM THE JOURNAL OF UROLOGY
Screening gaps miss childhood heart problems
People with a rare genetic condition that causes extremely elevated levels of low-density lipoprotein cholesterol (LDL-C) may miss out on decades of treatment because of a lack of lipid screening in childhood, researchers reported at the annual meeting of the American Academy of Pediatrics.
The condition, homozygous familial hypercholesterolemia (FH), raises the risk for atherosclerotic cardiovascular disease (ASCVD) as early as the first decade of life.
Routine screening for FH is uncommon, however, the researchers said. Lack of familiarity with guidelines and limited access to lipid specialists have been cited as possible reasons for inconsistent screening practices.
“These findings and recent improvement in lipid lowering therapies make a compelling case for rigorous compliance with AAP’s guidelines on lipid screening for children with a family history of FH or ASCVD at age 2,” study coauthor Mary P. McGowan, MD, chief medical officer of the Family Heart Foundation, said in a statement about the new study.
Early consequences
To characterize patients with homozygous FH, Dr. McGowan and her colleagues examined data from 67 participants in the CASCADE-FH registry. The Family Heart Foundation created the registry in 2013, and 40 medical centers in the United States contribute data to the repository. The researchers had access to data about patients with homozygous FH from 20 centers in the registry.
Dr. McGowan’s group compared 16 patients with homozygous FH who enrolled in the registry when they were children and 51 patients who were adults at the time of their enrollment.
Patients enrolled as children had a median age at diagnosis of 2 years (interquartile range [IQR], 2-3.5), whereas patients enrolled as adults had a median age at diagnosis of 12.6 years (IQR, 4.1-26.5).
The median untreated level of LDL-C in those enrolled as children was 776 mg/dL (IQR, 704-892). Among those enrolled as adults, it was 533 mg/dL (IQR, 467-702).
Approximately 19% of those enrolled as children had evidence of aortic valve stenosis, and 43.8% had evidence of ASCVD. The median age at onset of ASCVD was 8.9 years. One child was diagnosed with ASCVD at age 2 years and underwent liver transplant at age 4 years. Another was diagnosed with the condition at age 3 years and underwent liver transplant at age 8 years. Two children underwent coronary artery bypass grafting at ages 6 years and 14 years. Five participants underwent liver transplant before age 18 years.
About 56% of participants who enrolled as children had xanthomas, or fat deposits in tendons, and none had corneal arcus — a gray-white line of fat deposits around the edge of the cornea, both of which can indicate homozygous FH in children.
Treatment reduced LDL-C substantially, but only 25% of children achieved goal levels of cholesterol, the researchers reported. Patients who received more lipid-lowering therapies had a better chance of reaching their target levels, they found.
The data raise “the possibility that only children with the most severe phenotypes are diagnosed before adulthood,” the researchers said.
Clinical diagnosis of homozygous FH can be based on LDL-C levels, family history, and the presence of xanthomas, the researchers noted. Many children do not have physical findings, however, and a lipid panel or genetic testing may be necessary.
“There is a clear need to implement universal screening” to identify all children with homozygous FH and heterozygous FH, a less severe and more common form of FH, Dr. McGowan said.
Possible missed cases
As many as 1 in 250 people may have heterozygous FH, and 1 in 300,000 people may have homozygous FH, according to estimates. Patients with homozygous FH have two FH genes, one from each parent. In patients with homozygous FH, levels of LDL-C levels typically range between 400 and 1,000 mg/dL without treatment, which is four to 10 times higher than normal concentrations of the blood fat, according to the Family Heart Foundation.
“This study adds to a growing body of literature – including our own work – demonstrating that recommended universal screening occurs in barely 1 in 5 children. This means some patients are not being recognized as having treatable diseases,” said Justin H. Berger, MD, PhD, a pediatric cardiologist at Children’s Hospital of Philadelphia.
Even among children who are at the highest risk for early onset adult-type heart disease, only a quarter to two-thirds receive recommended screening, said Dr. Berger, who was not a member of the study team.
While Dr. Berger advocates universal lipid screening, improving screening rates in practice probably isn’t as simple as telling clinicians to screen more, he said. “Increasing testing will increase health care spending and the burden on busy primary care providers without addressing who will subsequently evaluate and manage children with abnormal lipid screening results,” Dr. Berger said.
Instead, clinicians may want to focus on screening patients who are at risk, which “could have dramatic benefits for their life-long cardiovascular health,” he said.
Dr. McGowan disclosed ties to Abbott and Regeneron, and her coauthors disclosed ties to Esperion Therapeutics and research funding from Regeneron and REGENXBIO. Dr. Berger disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
People with a rare genetic condition that causes extremely elevated levels of low-density lipoprotein cholesterol (LDL-C) may miss out on decades of treatment because of a lack of lipid screening in childhood, researchers reported at the annual meeting of the American Academy of Pediatrics.
The condition, homozygous familial hypercholesterolemia (FH), raises the risk for atherosclerotic cardiovascular disease (ASCVD) as early as the first decade of life.
Routine screening for FH is uncommon, however, the researchers said. Lack of familiarity with guidelines and limited access to lipid specialists have been cited as possible reasons for inconsistent screening practices.
“These findings and recent improvement in lipid lowering therapies make a compelling case for rigorous compliance with AAP’s guidelines on lipid screening for children with a family history of FH or ASCVD at age 2,” study coauthor Mary P. McGowan, MD, chief medical officer of the Family Heart Foundation, said in a statement about the new study.
Early consequences
To characterize patients with homozygous FH, Dr. McGowan and her colleagues examined data from 67 participants in the CASCADE-FH registry. The Family Heart Foundation created the registry in 2013, and 40 medical centers in the United States contribute data to the repository. The researchers had access to data about patients with homozygous FH from 20 centers in the registry.
Dr. McGowan’s group compared 16 patients with homozygous FH who enrolled in the registry when they were children and 51 patients who were adults at the time of their enrollment.
Patients enrolled as children had a median age at diagnosis of 2 years (interquartile range [IQR], 2-3.5), whereas patients enrolled as adults had a median age at diagnosis of 12.6 years (IQR, 4.1-26.5).
The median untreated level of LDL-C in those enrolled as children was 776 mg/dL (IQR, 704-892). Among those enrolled as adults, it was 533 mg/dL (IQR, 467-702).
Approximately 19% of those enrolled as children had evidence of aortic valve stenosis, and 43.8% had evidence of ASCVD. The median age at onset of ASCVD was 8.9 years. One child was diagnosed with ASCVD at age 2 years and underwent liver transplant at age 4 years. Another was diagnosed with the condition at age 3 years and underwent liver transplant at age 8 years. Two children underwent coronary artery bypass grafting at ages 6 years and 14 years. Five participants underwent liver transplant before age 18 years.
About 56% of participants who enrolled as children had xanthomas, or fat deposits in tendons, and none had corneal arcus — a gray-white line of fat deposits around the edge of the cornea, both of which can indicate homozygous FH in children.
Treatment reduced LDL-C substantially, but only 25% of children achieved goal levels of cholesterol, the researchers reported. Patients who received more lipid-lowering therapies had a better chance of reaching their target levels, they found.
The data raise “the possibility that only children with the most severe phenotypes are diagnosed before adulthood,” the researchers said.
Clinical diagnosis of homozygous FH can be based on LDL-C levels, family history, and the presence of xanthomas, the researchers noted. Many children do not have physical findings, however, and a lipid panel or genetic testing may be necessary.
“There is a clear need to implement universal screening” to identify all children with homozygous FH and heterozygous FH, a less severe and more common form of FH, Dr. McGowan said.
Possible missed cases
As many as 1 in 250 people may have heterozygous FH, and 1 in 300,000 people may have homozygous FH, according to estimates. Patients with homozygous FH have two FH genes, one from each parent. In patients with homozygous FH, levels of LDL-C levels typically range between 400 and 1,000 mg/dL without treatment, which is four to 10 times higher than normal concentrations of the blood fat, according to the Family Heart Foundation.
“This study adds to a growing body of literature – including our own work – demonstrating that recommended universal screening occurs in barely 1 in 5 children. This means some patients are not being recognized as having treatable diseases,” said Justin H. Berger, MD, PhD, a pediatric cardiologist at Children’s Hospital of Philadelphia.
Even among children who are at the highest risk for early onset adult-type heart disease, only a quarter to two-thirds receive recommended screening, said Dr. Berger, who was not a member of the study team.
While Dr. Berger advocates universal lipid screening, improving screening rates in practice probably isn’t as simple as telling clinicians to screen more, he said. “Increasing testing will increase health care spending and the burden on busy primary care providers without addressing who will subsequently evaluate and manage children with abnormal lipid screening results,” Dr. Berger said.
Instead, clinicians may want to focus on screening patients who are at risk, which “could have dramatic benefits for their life-long cardiovascular health,” he said.
Dr. McGowan disclosed ties to Abbott and Regeneron, and her coauthors disclosed ties to Esperion Therapeutics and research funding from Regeneron and REGENXBIO. Dr. Berger disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
People with a rare genetic condition that causes extremely elevated levels of low-density lipoprotein cholesterol (LDL-C) may miss out on decades of treatment because of a lack of lipid screening in childhood, researchers reported at the annual meeting of the American Academy of Pediatrics.
The condition, homozygous familial hypercholesterolemia (FH), raises the risk for atherosclerotic cardiovascular disease (ASCVD) as early as the first decade of life.
Routine screening for FH is uncommon, however, the researchers said. Lack of familiarity with guidelines and limited access to lipid specialists have been cited as possible reasons for inconsistent screening practices.
“These findings and recent improvement in lipid lowering therapies make a compelling case for rigorous compliance with AAP’s guidelines on lipid screening for children with a family history of FH or ASCVD at age 2,” study coauthor Mary P. McGowan, MD, chief medical officer of the Family Heart Foundation, said in a statement about the new study.
Early consequences
To characterize patients with homozygous FH, Dr. McGowan and her colleagues examined data from 67 participants in the CASCADE-FH registry. The Family Heart Foundation created the registry in 2013, and 40 medical centers in the United States contribute data to the repository. The researchers had access to data about patients with homozygous FH from 20 centers in the registry.
Dr. McGowan’s group compared 16 patients with homozygous FH who enrolled in the registry when they were children and 51 patients who were adults at the time of their enrollment.
Patients enrolled as children had a median age at diagnosis of 2 years (interquartile range [IQR], 2-3.5), whereas patients enrolled as adults had a median age at diagnosis of 12.6 years (IQR, 4.1-26.5).
The median untreated level of LDL-C in those enrolled as children was 776 mg/dL (IQR, 704-892). Among those enrolled as adults, it was 533 mg/dL (IQR, 467-702).
Approximately 19% of those enrolled as children had evidence of aortic valve stenosis, and 43.8% had evidence of ASCVD. The median age at onset of ASCVD was 8.9 years. One child was diagnosed with ASCVD at age 2 years and underwent liver transplant at age 4 years. Another was diagnosed with the condition at age 3 years and underwent liver transplant at age 8 years. Two children underwent coronary artery bypass grafting at ages 6 years and 14 years. Five participants underwent liver transplant before age 18 years.
About 56% of participants who enrolled as children had xanthomas, or fat deposits in tendons, and none had corneal arcus — a gray-white line of fat deposits around the edge of the cornea, both of which can indicate homozygous FH in children.
Treatment reduced LDL-C substantially, but only 25% of children achieved goal levels of cholesterol, the researchers reported. Patients who received more lipid-lowering therapies had a better chance of reaching their target levels, they found.
The data raise “the possibility that only children with the most severe phenotypes are diagnosed before adulthood,” the researchers said.
Clinical diagnosis of homozygous FH can be based on LDL-C levels, family history, and the presence of xanthomas, the researchers noted. Many children do not have physical findings, however, and a lipid panel or genetic testing may be necessary.
“There is a clear need to implement universal screening” to identify all children with homozygous FH and heterozygous FH, a less severe and more common form of FH, Dr. McGowan said.
Possible missed cases
As many as 1 in 250 people may have heterozygous FH, and 1 in 300,000 people may have homozygous FH, according to estimates. Patients with homozygous FH have two FH genes, one from each parent. In patients with homozygous FH, levels of LDL-C levels typically range between 400 and 1,000 mg/dL without treatment, which is four to 10 times higher than normal concentrations of the blood fat, according to the Family Heart Foundation.
“This study adds to a growing body of literature – including our own work – demonstrating that recommended universal screening occurs in barely 1 in 5 children. This means some patients are not being recognized as having treatable diseases,” said Justin H. Berger, MD, PhD, a pediatric cardiologist at Children’s Hospital of Philadelphia.
Even among children who are at the highest risk for early onset adult-type heart disease, only a quarter to two-thirds receive recommended screening, said Dr. Berger, who was not a member of the study team.
While Dr. Berger advocates universal lipid screening, improving screening rates in practice probably isn’t as simple as telling clinicians to screen more, he said. “Increasing testing will increase health care spending and the burden on busy primary care providers without addressing who will subsequently evaluate and manage children with abnormal lipid screening results,” Dr. Berger said.
Instead, clinicians may want to focus on screening patients who are at risk, which “could have dramatic benefits for their life-long cardiovascular health,” he said.
Dr. McGowan disclosed ties to Abbott and Regeneron, and her coauthors disclosed ties to Esperion Therapeutics and research funding from Regeneron and REGENXBIO. Dr. Berger disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM AAP 2022
No effect of diet on dementia risk?
Contrary to some prior studies,
After adjusting for relevant demographic and other lifestyle measures, there was no association between adherence to healthy dietary advice or the Mediterranean diet on the future risk of dementia or amyloid-beta (Abeta) accumulation.
“While our study does not rule out a possible association between diet and dementia, we did not find a link in our study, which had a long follow-up period, included younger participants than some other studies and did not require people to remember what foods they had eaten regularly years before,” study investigator Isabelle Glans, MD, of Lund (Sweden) University, said in a news release.
The findings were published online in Neurology.
No risk reduction
Several studies have investigated how dietary habits affect dementia risk, with inconsistent results.
The new findings are based on 28,025 adults (61% women; mean age, 58 years at baseline) who were free of dementia at baseline and were followed over a 20-year period as part of the Swedish Malmö Diet and Cancer Study. Dietary habits were assessed with a 7-day food diary, detailed food frequency questionnaire, and in-person interview.
During follow-up, 1,943 individuals (6.9%) developed dementia.
Compared with those who did not develop dementia, those who did develop dementia during follow-up were older and had a lower level of education and more cardiovascular risk factors and comorbidities at baseline.
Individuals who adhered to conventional healthy dietary recommendations did not have a lower risk of developing all-cause dementia (hazard ratio comparing worst with best adherence, 0.93; 95% confidence interval, 0.81-1.08), Alzheimer’s disease (HR, 1.03; 95% CI, 0.85-1.23) or vascular dementia (HR, 0.93; 95% CI, 0.69-1.26).
Adherence to the modified Mediterranean diet also did not appear to lower the risk of all-cause dementia (HR, 0.93; 95% CI, 0.75-1.15), Alzheimer’s disease (HR, 0.90; 95% CI, 0.68-1.19), or vascular dementia (HR, 1.00; 95% CI, 0.65-1.55).
There was also no significant association between diet and Alzheimer’s disease–related pathology, as measured by cerebrospinal fluid analysis of Abeta42 in a subgroup of 738 participants. Various sensitivity analyses yielded similar results.
Diet still matters
The authors of an accompanying editorial noted that diet as a “singular factor may not have a strong enough effect on cognition, but is more likely to be considered as one factor embedded with various others, the sum of which may influence the course of cognitive function (diet, regular exercise, vascular risk factor control, avoiding cigarette smoking, drinking alcohol in moderation, etc).
“Diet should not be forgotten and it still matters” but should be regarded as “one part of a multidomain intervention with respect to cognitive performance,” wrote Nils Peters, MD, with the University of Basel (Switzerland), and Benedetta Nacmias, PhD, with the University of Florence (Italy)).
“Key questions that remain include how to provide evidence for promoting the implications of dietary habits on cognition? Overall, dietary strategies will most likely be implicated either in order to reduce the increasing number of older subjects with dementia, or to extend healthy life expectancy, or both,” Dr. Peters and Dr. Nacmias said.
The study had no commercial funding. Dr. Glans, Dr. Peters, and Dr. Nacmias disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Contrary to some prior studies,
After adjusting for relevant demographic and other lifestyle measures, there was no association between adherence to healthy dietary advice or the Mediterranean diet on the future risk of dementia or amyloid-beta (Abeta) accumulation.
“While our study does not rule out a possible association between diet and dementia, we did not find a link in our study, which had a long follow-up period, included younger participants than some other studies and did not require people to remember what foods they had eaten regularly years before,” study investigator Isabelle Glans, MD, of Lund (Sweden) University, said in a news release.
The findings were published online in Neurology.
No risk reduction
Several studies have investigated how dietary habits affect dementia risk, with inconsistent results.
The new findings are based on 28,025 adults (61% women; mean age, 58 years at baseline) who were free of dementia at baseline and were followed over a 20-year period as part of the Swedish Malmö Diet and Cancer Study. Dietary habits were assessed with a 7-day food diary, detailed food frequency questionnaire, and in-person interview.
During follow-up, 1,943 individuals (6.9%) developed dementia.
Compared with those who did not develop dementia, those who did develop dementia during follow-up were older and had a lower level of education and more cardiovascular risk factors and comorbidities at baseline.
Individuals who adhered to conventional healthy dietary recommendations did not have a lower risk of developing all-cause dementia (hazard ratio comparing worst with best adherence, 0.93; 95% confidence interval, 0.81-1.08), Alzheimer’s disease (HR, 1.03; 95% CI, 0.85-1.23) or vascular dementia (HR, 0.93; 95% CI, 0.69-1.26).
Adherence to the modified Mediterranean diet also did not appear to lower the risk of all-cause dementia (HR, 0.93; 95% CI, 0.75-1.15), Alzheimer’s disease (HR, 0.90; 95% CI, 0.68-1.19), or vascular dementia (HR, 1.00; 95% CI, 0.65-1.55).
There was also no significant association between diet and Alzheimer’s disease–related pathology, as measured by cerebrospinal fluid analysis of Abeta42 in a subgroup of 738 participants. Various sensitivity analyses yielded similar results.
Diet still matters
The authors of an accompanying editorial noted that diet as a “singular factor may not have a strong enough effect on cognition, but is more likely to be considered as one factor embedded with various others, the sum of which may influence the course of cognitive function (diet, regular exercise, vascular risk factor control, avoiding cigarette smoking, drinking alcohol in moderation, etc).
“Diet should not be forgotten and it still matters” but should be regarded as “one part of a multidomain intervention with respect to cognitive performance,” wrote Nils Peters, MD, with the University of Basel (Switzerland), and Benedetta Nacmias, PhD, with the University of Florence (Italy)).
“Key questions that remain include how to provide evidence for promoting the implications of dietary habits on cognition? Overall, dietary strategies will most likely be implicated either in order to reduce the increasing number of older subjects with dementia, or to extend healthy life expectancy, or both,” Dr. Peters and Dr. Nacmias said.
The study had no commercial funding. Dr. Glans, Dr. Peters, and Dr. Nacmias disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Contrary to some prior studies,
After adjusting for relevant demographic and other lifestyle measures, there was no association between adherence to healthy dietary advice or the Mediterranean diet on the future risk of dementia or amyloid-beta (Abeta) accumulation.
“While our study does not rule out a possible association between diet and dementia, we did not find a link in our study, which had a long follow-up period, included younger participants than some other studies and did not require people to remember what foods they had eaten regularly years before,” study investigator Isabelle Glans, MD, of Lund (Sweden) University, said in a news release.
The findings were published online in Neurology.
No risk reduction
Several studies have investigated how dietary habits affect dementia risk, with inconsistent results.
The new findings are based on 28,025 adults (61% women; mean age, 58 years at baseline) who were free of dementia at baseline and were followed over a 20-year period as part of the Swedish Malmö Diet and Cancer Study. Dietary habits were assessed with a 7-day food diary, detailed food frequency questionnaire, and in-person interview.
During follow-up, 1,943 individuals (6.9%) developed dementia.
Compared with those who did not develop dementia, those who did develop dementia during follow-up were older and had a lower level of education and more cardiovascular risk factors and comorbidities at baseline.
Individuals who adhered to conventional healthy dietary recommendations did not have a lower risk of developing all-cause dementia (hazard ratio comparing worst with best adherence, 0.93; 95% confidence interval, 0.81-1.08), Alzheimer’s disease (HR, 1.03; 95% CI, 0.85-1.23) or vascular dementia (HR, 0.93; 95% CI, 0.69-1.26).
Adherence to the modified Mediterranean diet also did not appear to lower the risk of all-cause dementia (HR, 0.93; 95% CI, 0.75-1.15), Alzheimer’s disease (HR, 0.90; 95% CI, 0.68-1.19), or vascular dementia (HR, 1.00; 95% CI, 0.65-1.55).
There was also no significant association between diet and Alzheimer’s disease–related pathology, as measured by cerebrospinal fluid analysis of Abeta42 in a subgroup of 738 participants. Various sensitivity analyses yielded similar results.
Diet still matters
The authors of an accompanying editorial noted that diet as a “singular factor may not have a strong enough effect on cognition, but is more likely to be considered as one factor embedded with various others, the sum of which may influence the course of cognitive function (diet, regular exercise, vascular risk factor control, avoiding cigarette smoking, drinking alcohol in moderation, etc).
“Diet should not be forgotten and it still matters” but should be regarded as “one part of a multidomain intervention with respect to cognitive performance,” wrote Nils Peters, MD, with the University of Basel (Switzerland), and Benedetta Nacmias, PhD, with the University of Florence (Italy)).
“Key questions that remain include how to provide evidence for promoting the implications of dietary habits on cognition? Overall, dietary strategies will most likely be implicated either in order to reduce the increasing number of older subjects with dementia, or to extend healthy life expectancy, or both,” Dr. Peters and Dr. Nacmias said.
The study had no commercial funding. Dr. Glans, Dr. Peters, and Dr. Nacmias disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM NEUROLOGY
New COVID variant gaining traction in U.S.
, according to the CDC’s latest data.
Just 1 month ago, the variant accounted for less than 1% of cases.
“When you get variants like that, you look at what their rate of increase is as a relative proportion of the variants, and this has a pretty troublesome doubling time,” Anthony Fauci, MD, said in an interview with CBS News. Dr. Fauci is the director of the National Institute of Allergy and Infectious Diseases and also the chief medical adviser to President Joe Biden.
There are also concerning features of the BQ.1 variant, which include mutations that could potentially escape vaccines and treatments for COVID-19.
Currently, the most widespread variant in the U.S. is the Omicron subvariant known as BA.5, which accounts for 68% of all infections. One of the go-to treatments for BA.5 infections is monoclonal antibodies, which may not be as effective when fighting the up-and-coming strains of BQ.1 and its descendant BQ.1.1, according to experts.
“That’s the reason why people are concerned about BQ.1.1, for the double reason of its doubling time and the fact that it seems to elude important monoclonal antibodies,” Dr. Fauci told CBS News.
Currently, BQ.1 and BQ.1.1 appear most widespread in the New York and New Jersey region, accounting for nearly 20% of infections there, according to the CDC.
But because the new variant is a descendant of Omicron, Dr. Fauci said the currently available booster shots are still the best first line of protection against this up-and-coming threat.
“The bad news is that there’s a new variant that’s emerging and that has qualities or characteristics that could evade some of the interventions we have. But, the somewhat encouraging news is that it’s a BA.5 sub-lineage, so there is almost certainly going to be some cross-protection that you can boost up,” he said.
A version of this article first appeared on WebMD.com.
, according to the CDC’s latest data.
Just 1 month ago, the variant accounted for less than 1% of cases.
“When you get variants like that, you look at what their rate of increase is as a relative proportion of the variants, and this has a pretty troublesome doubling time,” Anthony Fauci, MD, said in an interview with CBS News. Dr. Fauci is the director of the National Institute of Allergy and Infectious Diseases and also the chief medical adviser to President Joe Biden.
There are also concerning features of the BQ.1 variant, which include mutations that could potentially escape vaccines and treatments for COVID-19.
Currently, the most widespread variant in the U.S. is the Omicron subvariant known as BA.5, which accounts for 68% of all infections. One of the go-to treatments for BA.5 infections is monoclonal antibodies, which may not be as effective when fighting the up-and-coming strains of BQ.1 and its descendant BQ.1.1, according to experts.
“That’s the reason why people are concerned about BQ.1.1, for the double reason of its doubling time and the fact that it seems to elude important monoclonal antibodies,” Dr. Fauci told CBS News.
Currently, BQ.1 and BQ.1.1 appear most widespread in the New York and New Jersey region, accounting for nearly 20% of infections there, according to the CDC.
But because the new variant is a descendant of Omicron, Dr. Fauci said the currently available booster shots are still the best first line of protection against this up-and-coming threat.
“The bad news is that there’s a new variant that’s emerging and that has qualities or characteristics that could evade some of the interventions we have. But, the somewhat encouraging news is that it’s a BA.5 sub-lineage, so there is almost certainly going to be some cross-protection that you can boost up,” he said.
A version of this article first appeared on WebMD.com.
, according to the CDC’s latest data.
Just 1 month ago, the variant accounted for less than 1% of cases.
“When you get variants like that, you look at what their rate of increase is as a relative proportion of the variants, and this has a pretty troublesome doubling time,” Anthony Fauci, MD, said in an interview with CBS News. Dr. Fauci is the director of the National Institute of Allergy and Infectious Diseases and also the chief medical adviser to President Joe Biden.
There are also concerning features of the BQ.1 variant, which include mutations that could potentially escape vaccines and treatments for COVID-19.
Currently, the most widespread variant in the U.S. is the Omicron subvariant known as BA.5, which accounts for 68% of all infections. One of the go-to treatments for BA.5 infections is monoclonal antibodies, which may not be as effective when fighting the up-and-coming strains of BQ.1 and its descendant BQ.1.1, according to experts.
“That’s the reason why people are concerned about BQ.1.1, for the double reason of its doubling time and the fact that it seems to elude important monoclonal antibodies,” Dr. Fauci told CBS News.
Currently, BQ.1 and BQ.1.1 appear most widespread in the New York and New Jersey region, accounting for nearly 20% of infections there, according to the CDC.
But because the new variant is a descendant of Omicron, Dr. Fauci said the currently available booster shots are still the best first line of protection against this up-and-coming threat.
“The bad news is that there’s a new variant that’s emerging and that has qualities or characteristics that could evade some of the interventions we have. But, the somewhat encouraging news is that it’s a BA.5 sub-lineage, so there is almost certainly going to be some cross-protection that you can boost up,” he said.
A version of this article first appeared on WebMD.com.
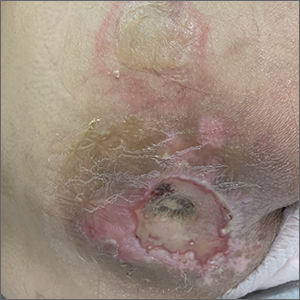
Sacral blistering
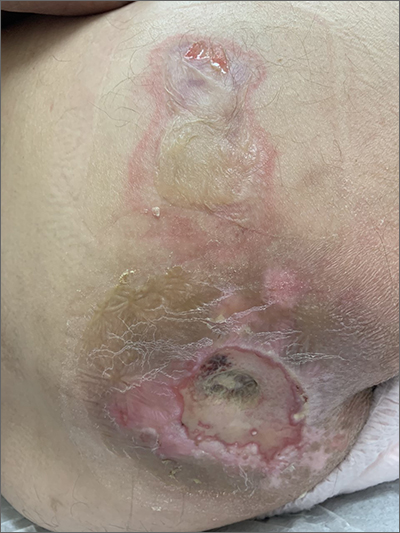
This patient had sustained multiple pressure injuries. The superior aspect of this image shows bullous change with intact dermis, which would classify that area of injury as a Stage 2 pressure injury.1 An older injury in the coccygeal area was through the dermis (Stage 3), with some eschar seen at the base, making that area unstageable.1 (There may have been deeper injury under the eschar.)
This patient was at heightened risk for pressure injury because of his paraplegia.2 Fortunately, he had some preserved sensation. However, his rotator cuff surgery made it harder for him to smoothly transfer to and from the wheelchair, leading to sheer forces against his skin. Social determinates of health care pose an additional risk for pressure injuries. Without a properly fitted wheelchair and cushion, there is an increased risk of localized pressure over both bony prominences and parts of the body that come into contact with mechanical elements of the wheelchair.
Treatment for all pressure injuries includes relief of pressure on the affected area. In this patient’s case, he had to stay in bed (and out of the wheelchair) so that he could heal.
This patient was very knowledgeable about his condition and pressure injuries. He had already arranged for a wheelchair fitting and a visit with the wound care team. His Stage 2 injury had a bullous change instead of absent epithelium, so rather than an adherent hydrocolloid dressing (which would likely remove the loosened epithelium), he was provided with a nonadherent dressing to the area, then an absorbent foam overdressing.
An image of the patient’s deeper sacral injury was shared with the wound care team, which recommended filling the area with a silver rope dressing for its absorptive filler and antibacterial properties. The area was then covered with an absorbent foam. The patient planned to follow up with the Wound Care Clinic for reevaluation and ongoing treatment.
Images and text courtesy of Daniel Stulberg, MD, FAAFP, Professor and Chair, Department of Family and Community Medicine, Western Michigan University Homer Stryker, MD School of Medicine Kalamazoo.
1. 2019 Guideline. The National Pressure Injury Advisory Panel. Accessed October 9, 2022. https://npiap.com/page/2019Guideline
2. Ricci JA, Bayer LR, Orgill DP. Evidence-based medicine: the evaluation and treatment of pressure injuries. Plast Reconstr Surg. 2017;139:275e-286e. doi: 10.1097/PRS.0000000000002850

This patient had sustained multiple pressure injuries. The superior aspect of this image shows bullous change with intact dermis, which would classify that area of injury as a Stage 2 pressure injury.1 An older injury in the coccygeal area was through the dermis (Stage 3), with some eschar seen at the base, making that area unstageable.1 (There may have been deeper injury under the eschar.)
This patient was at heightened risk for pressure injury because of his paraplegia.2 Fortunately, he had some preserved sensation. However, his rotator cuff surgery made it harder for him to smoothly transfer to and from the wheelchair, leading to sheer forces against his skin. Social determinates of health care pose an additional risk for pressure injuries. Without a properly fitted wheelchair and cushion, there is an increased risk of localized pressure over both bony prominences and parts of the body that come into contact with mechanical elements of the wheelchair.
Treatment for all pressure injuries includes relief of pressure on the affected area. In this patient’s case, he had to stay in bed (and out of the wheelchair) so that he could heal.
This patient was very knowledgeable about his condition and pressure injuries. He had already arranged for a wheelchair fitting and a visit with the wound care team. His Stage 2 injury had a bullous change instead of absent epithelium, so rather than an adherent hydrocolloid dressing (which would likely remove the loosened epithelium), he was provided with a nonadherent dressing to the area, then an absorbent foam overdressing.
An image of the patient’s deeper sacral injury was shared with the wound care team, which recommended filling the area with a silver rope dressing for its absorptive filler and antibacterial properties. The area was then covered with an absorbent foam. The patient planned to follow up with the Wound Care Clinic for reevaluation and ongoing treatment.
Images and text courtesy of Daniel Stulberg, MD, FAAFP, Professor and Chair, Department of Family and Community Medicine, Western Michigan University Homer Stryker, MD School of Medicine Kalamazoo.

This patient had sustained multiple pressure injuries. The superior aspect of this image shows bullous change with intact dermis, which would classify that area of injury as a Stage 2 pressure injury.1 An older injury in the coccygeal area was through the dermis (Stage 3), with some eschar seen at the base, making that area unstageable.1 (There may have been deeper injury under the eschar.)
This patient was at heightened risk for pressure injury because of his paraplegia.2 Fortunately, he had some preserved sensation. However, his rotator cuff surgery made it harder for him to smoothly transfer to and from the wheelchair, leading to sheer forces against his skin. Social determinates of health care pose an additional risk for pressure injuries. Without a properly fitted wheelchair and cushion, there is an increased risk of localized pressure over both bony prominences and parts of the body that come into contact with mechanical elements of the wheelchair.
Treatment for all pressure injuries includes relief of pressure on the affected area. In this patient’s case, he had to stay in bed (and out of the wheelchair) so that he could heal.
This patient was very knowledgeable about his condition and pressure injuries. He had already arranged for a wheelchair fitting and a visit with the wound care team. His Stage 2 injury had a bullous change instead of absent epithelium, so rather than an adherent hydrocolloid dressing (which would likely remove the loosened epithelium), he was provided with a nonadherent dressing to the area, then an absorbent foam overdressing.
An image of the patient’s deeper sacral injury was shared with the wound care team, which recommended filling the area with a silver rope dressing for its absorptive filler and antibacterial properties. The area was then covered with an absorbent foam. The patient planned to follow up with the Wound Care Clinic for reevaluation and ongoing treatment.
Images and text courtesy of Daniel Stulberg, MD, FAAFP, Professor and Chair, Department of Family and Community Medicine, Western Michigan University Homer Stryker, MD School of Medicine Kalamazoo.
1. 2019 Guideline. The National Pressure Injury Advisory Panel. Accessed October 9, 2022. https://npiap.com/page/2019Guideline
2. Ricci JA, Bayer LR, Orgill DP. Evidence-based medicine: the evaluation and treatment of pressure injuries. Plast Reconstr Surg. 2017;139:275e-286e. doi: 10.1097/PRS.0000000000002850
1. 2019 Guideline. The National Pressure Injury Advisory Panel. Accessed October 9, 2022. https://npiap.com/page/2019Guideline
2. Ricci JA, Bayer LR, Orgill DP. Evidence-based medicine: the evaluation and treatment of pressure injuries. Plast Reconstr Surg. 2017;139:275e-286e. doi: 10.1097/PRS.0000000000002850
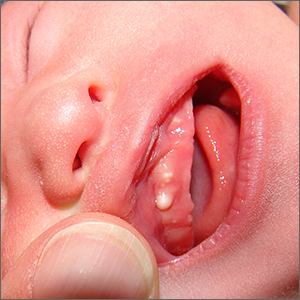
Newborn with white oral lesions
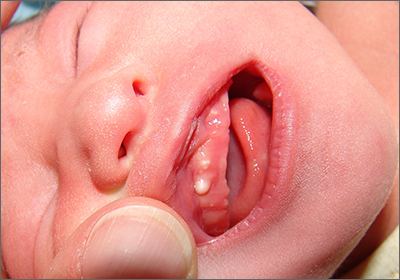
These lesions, called Bohn nodules, manifest on the buccal or lingual portion of the maxillary alveolar ridge and, less frequently, on the mandibular alveolar ridge. Because Bohn nodules are white and have a firm consistency, they are often confused with teeth. They can be differentiated by location, as teeth usually erupt from the distal aspect of the alveolar ridge.
Bohn nodules are epithelial cysts that are filled with keratin, which gives them their white color. They are caused by portions of epithelium that get trapped under surrounding epithelial cells. Bohn nodules usually resolve when the overlying epithelium ruptures and releases the keratinaceous material (usually by the time the child is 3 months of age).1
These nodules can be confused with neonatal or supernumerary teeth. Neonatal teeth can be abnormally small and pointed; they are true deciduous teeth that have erupted early. If they are removed, the child will not replace them until the time of their adult tooth eruption. Additionally, there are supernumerary teeth, which are extra teeth that are often abnormally shaped and loosely adherent. These abnormal teeth warrant extraction to avoid trauma to the tongue or aspiration.2
In this case, the family was advised regarding the benign nature of the nodules and the expectation that they would spontaneously resolve.
Images and text courtesy of Daniel Stulberg, MD, FAAFP, Professor and Chair, Department of Family and Community Medicine, Western Michigan University Homer Stryker, MD School of Medicine Kalamazoo.
1. Gupta N, Ramji S. Bohn's nodules: an under-recognised entity. Arch Dis Child Fetal Neonatal Ed. 2013;98:F464. doi: 10.1136/archdischild-2012-302922
2. DeSeta M, Holden E, Siddik D, et al. Natal and neonatal teeth: a review and case series. Br Dent J. 2022;232:449-453. doi: 10.1038/s41415-022-4091-3

These lesions, called Bohn nodules, manifest on the buccal or lingual portion of the maxillary alveolar ridge and, less frequently, on the mandibular alveolar ridge. Because Bohn nodules are white and have a firm consistency, they are often confused with teeth. They can be differentiated by location, as teeth usually erupt from the distal aspect of the alveolar ridge.
Bohn nodules are epithelial cysts that are filled with keratin, which gives them their white color. They are caused by portions of epithelium that get trapped under surrounding epithelial cells. Bohn nodules usually resolve when the overlying epithelium ruptures and releases the keratinaceous material (usually by the time the child is 3 months of age).1
These nodules can be confused with neonatal or supernumerary teeth. Neonatal teeth can be abnormally small and pointed; they are true deciduous teeth that have erupted early. If they are removed, the child will not replace them until the time of their adult tooth eruption. Additionally, there are supernumerary teeth, which are extra teeth that are often abnormally shaped and loosely adherent. These abnormal teeth warrant extraction to avoid trauma to the tongue or aspiration.2
In this case, the family was advised regarding the benign nature of the nodules and the expectation that they would spontaneously resolve.
Images and text courtesy of Daniel Stulberg, MD, FAAFP, Professor and Chair, Department of Family and Community Medicine, Western Michigan University Homer Stryker, MD School of Medicine Kalamazoo.

These lesions, called Bohn nodules, manifest on the buccal or lingual portion of the maxillary alveolar ridge and, less frequently, on the mandibular alveolar ridge. Because Bohn nodules are white and have a firm consistency, they are often confused with teeth. They can be differentiated by location, as teeth usually erupt from the distal aspect of the alveolar ridge.
Bohn nodules are epithelial cysts that are filled with keratin, which gives them their white color. They are caused by portions of epithelium that get trapped under surrounding epithelial cells. Bohn nodules usually resolve when the overlying epithelium ruptures and releases the keratinaceous material (usually by the time the child is 3 months of age).1
These nodules can be confused with neonatal or supernumerary teeth. Neonatal teeth can be abnormally small and pointed; they are true deciduous teeth that have erupted early. If they are removed, the child will not replace them until the time of their adult tooth eruption. Additionally, there are supernumerary teeth, which are extra teeth that are often abnormally shaped and loosely adherent. These abnormal teeth warrant extraction to avoid trauma to the tongue or aspiration.2
In this case, the family was advised regarding the benign nature of the nodules and the expectation that they would spontaneously resolve.
Images and text courtesy of Daniel Stulberg, MD, FAAFP, Professor and Chair, Department of Family and Community Medicine, Western Michigan University Homer Stryker, MD School of Medicine Kalamazoo.
1. Gupta N, Ramji S. Bohn's nodules: an under-recognised entity. Arch Dis Child Fetal Neonatal Ed. 2013;98:F464. doi: 10.1136/archdischild-2012-302922
2. DeSeta M, Holden E, Siddik D, et al. Natal and neonatal teeth: a review and case series. Br Dent J. 2022;232:449-453. doi: 10.1038/s41415-022-4091-3
1. Gupta N, Ramji S. Bohn's nodules: an under-recognised entity. Arch Dis Child Fetal Neonatal Ed. 2013;98:F464. doi: 10.1136/archdischild-2012-302922
2. DeSeta M, Holden E, Siddik D, et al. Natal and neonatal teeth: a review and case series. Br Dent J. 2022;232:449-453. doi: 10.1038/s41415-022-4091-3
Finerenone benefits T2D across spectrum of renal function
Treatment with finerenone produced roughly similar reductions in heart failure–related outcomes in people with type 2 diabetes and chronic kidney disease (CKD) across the spectrum of kidney function, compared with placebo, including those who had albuminuria but a preserved estimated glomerular filtration rate (eGFR), in a post hoc analysis of pooled data from more than 13,000 people.
The findings, from the two pivotal trials for the agent, “reinforce the importance of routine eGFR and UACR [urinary albumin-to-creatinine ratio] screening” in people with type 2 diabetes to identify new candidates for treatment with finerenone (Kerendia), Gerasimos Filippatos, MD, and coauthors said in a report published online in JACC: Heart Failure.
Among the 13,026 patients in the two combined trials, 40% had a preserved eGFR of greater than 60 mL/min per 1.73 m2 despite also having albuminuria with a UACR of at least 30 mg/g, showing how often this combination occurs. But many clinicians “do not follow the guidelines” and fail to measure the UACR in these patients in routine practice, noted Dr. Filippatos at the annual congress of the European Society of Cardiology in August.
“We now have something to do for these patients,” treat them with finerenone, said Dr. Filippatos, professor and director of heart failure at the Attikon University Hospital, Athens.
The availability of finerenone following its U.S. approval in 2021 means clinicians “must get used to measuring UACR” in people with type 2 diabetes even when their eGFR is normal, especially people with type 2 diabetes plus high cardiovascular disease risk, he said.
The Food and Drug Administration approved finerenone, a nonsteroidal mineralocorticoid receptor antagonist, for treating people with type 2 diabetes and CKD in July 2021, but its uptake has been slow, experts say. In a talk in September 2022 during the annual meeting of the European Association for the Study of Diabetes, Jennifer B. Green, MD, estimated that U.S. uptake of finerenone for appropriate people with type 2 diabetes had not advanced beyond 10%.
A recent review also noted that uptake of screening for elevated UACR in U.S. patients with type 2 diabetes was in the range of 10%-40% during 2017-2019, a “shockingly low rate,” said Dr. Green, a professor and diabetes specialist at Duke University, Durham, N.C.
A new reason to screen for albuminuria
“It’s an extremely important message,” Johann Bauersachs, MD, commented in an interview. Results from “many studies have shown that albuminuria is an excellent additional marker for cardiovascular disease risk. But measurement of albuminuria is not widely done, despite guidelines that recommend annual albuminuria testing in people with type 2 diabetes,” said Dr. Bauersachs, professor and head of the department of cardiology at Hannover (Germany) Medical School.
“Even before there was finerenone, there were reasons to measure UACR, but I hope adding finerenone will help, and more clinicians will incorporate UACR into their routine practice,” said Dr. Bauersachs, who was not involved with the finerenone studies.
The analyses reported by Dr. Filippatos and coauthors used data from two related trials of finerenone, FIDELIO-DKD and FIGARO-DKD, combined by prespecified design into a single dataset, FIDELITY, with a total of 13,026 participants eligible for analysis and followed for a median of 3 years. All had type 2 diabetes and CKD based on having a UACR of at least 30 mg/g. Their eGFR levels could run as high as 74 mL/min per 1.73 m2 in FIDELIO-DKD, and as high as 90 mL/min/1.73m2 in FIGARO-DKD. The two trials excluded people with heart failure with reduced ejection fraction, and those with a serum potassium greater than 4.8 mmol/L.
In the FIDELITY dataset treatment with finerenone led to a significant 17% reduction in the combined incidence of cardiovascular death or first hospitalization for heart failure relative to those who received placebo. This relative risk reduction was not affected by either eGFR or UACR values at baseline, the new analysis showed.
The analysis also demonstrated a nonsignificant trend toward greater reductions in heart failure–related outcomes among study participants who began with an eGFR in the normal range of at least 60 mL/min per 1.73 m2. The researchers also found a nonsignificant trend to a greater reduction in heart failure–related events among those with a UACR of less than 300 mg/g.
Finerenone favors patients with less advanced CKD
In short “the magnitude of the treatment benefit tended to favor patients with less advanced CKD,” concluded the researchers, suggesting that “earlier intervention [with finerenone] in the CKD course is likely to provide the greatest long-term benefit on heart failure–related outcomes.” This led them to further infer “the importance of not only routine assessing eGFR, but also perhaps more importantly, routinely screening for UACR to facilitate early diagnosis and early intervention in patients with type 2 diabetes.”
Findings from FIDELIO-DKD and FIGARO-DKD led to recent guideline additions for finerenone by several medical groups. In August 2022, the American Association of Clinical Endocrinologists released an update to its guideline for managing people with diabetes that recommended treating people with type 2 diabetes with finerenone when they have a UACR of at least 30 mg/g if they are already treated with a maximum-tolerated dose of a renin-angiotensin system inhibitor, have a normal serum potassium level, and have an eGFR of at least 25 mL/min per 1.73 m2. The identical recommendation also appeared in a Consensus Report from the American Diabetes Association and KDIGO, an international organization promoting evidence-based management of patients with CKD.
“Finerenone provides a very important contribution because it improves prognosis even in very well managed patients” with type 2 diabetes, commented Lars Rydén, MD, professor of cardiology at the Karolinska Institute in Stockholm, as designated discussant for the report by Dr. Filippatos at the ESC congress.
The findings from the FIDELITY analysis are “trustworthy, and clinically important,” Dr. Rydén said. When left untreated, diabetic kidney disease “reduces life expectancy by an average of 16 years.”
The finerenone trials were sponsored by Bayer, which markets finerenone (Kerendia). Dr. Filippatos has received lecture fees from Bayer as well as from Amgen, Medtronic, Novartis, Servier, and Vifor. Dr. Green has financial ties to Bayer as well as to Anji, AstraZeneca, Boehringer Ingelheim/Lilly, Hawthorne Effect/Omada, Merck, Novo Nordisk, Pfizer, Roche, Sanofi/Lexicon, and Valo. Dr. Bauersachs has been a consultant to Bayer as well as to Amgen, AstraZeneca, Boehringer Ingelheim, Cardior, Cervia, CVRx, Novartis, Pfizer, and Vifor, and he has received research funding from Abiomed. Dr. Rydén has financial ties to Bayer, Boehringer Ingelheim, Eli Lilly, and Novo Nordisk.
Treatment with finerenone produced roughly similar reductions in heart failure–related outcomes in people with type 2 diabetes and chronic kidney disease (CKD) across the spectrum of kidney function, compared with placebo, including those who had albuminuria but a preserved estimated glomerular filtration rate (eGFR), in a post hoc analysis of pooled data from more than 13,000 people.
The findings, from the two pivotal trials for the agent, “reinforce the importance of routine eGFR and UACR [urinary albumin-to-creatinine ratio] screening” in people with type 2 diabetes to identify new candidates for treatment with finerenone (Kerendia), Gerasimos Filippatos, MD, and coauthors said in a report published online in JACC: Heart Failure.
Among the 13,026 patients in the two combined trials, 40% had a preserved eGFR of greater than 60 mL/min per 1.73 m2 despite also having albuminuria with a UACR of at least 30 mg/g, showing how often this combination occurs. But many clinicians “do not follow the guidelines” and fail to measure the UACR in these patients in routine practice, noted Dr. Filippatos at the annual congress of the European Society of Cardiology in August.
“We now have something to do for these patients,” treat them with finerenone, said Dr. Filippatos, professor and director of heart failure at the Attikon University Hospital, Athens.
The availability of finerenone following its U.S. approval in 2021 means clinicians “must get used to measuring UACR” in people with type 2 diabetes even when their eGFR is normal, especially people with type 2 diabetes plus high cardiovascular disease risk, he said.
The Food and Drug Administration approved finerenone, a nonsteroidal mineralocorticoid receptor antagonist, for treating people with type 2 diabetes and CKD in July 2021, but its uptake has been slow, experts say. In a talk in September 2022 during the annual meeting of the European Association for the Study of Diabetes, Jennifer B. Green, MD, estimated that U.S. uptake of finerenone for appropriate people with type 2 diabetes had not advanced beyond 10%.
A recent review also noted that uptake of screening for elevated UACR in U.S. patients with type 2 diabetes was in the range of 10%-40% during 2017-2019, a “shockingly low rate,” said Dr. Green, a professor and diabetes specialist at Duke University, Durham, N.C.
A new reason to screen for albuminuria
“It’s an extremely important message,” Johann Bauersachs, MD, commented in an interview. Results from “many studies have shown that albuminuria is an excellent additional marker for cardiovascular disease risk. But measurement of albuminuria is not widely done, despite guidelines that recommend annual albuminuria testing in people with type 2 diabetes,” said Dr. Bauersachs, professor and head of the department of cardiology at Hannover (Germany) Medical School.
“Even before there was finerenone, there were reasons to measure UACR, but I hope adding finerenone will help, and more clinicians will incorporate UACR into their routine practice,” said Dr. Bauersachs, who was not involved with the finerenone studies.
The analyses reported by Dr. Filippatos and coauthors used data from two related trials of finerenone, FIDELIO-DKD and FIGARO-DKD, combined by prespecified design into a single dataset, FIDELITY, with a total of 13,026 participants eligible for analysis and followed for a median of 3 years. All had type 2 diabetes and CKD based on having a UACR of at least 30 mg/g. Their eGFR levels could run as high as 74 mL/min per 1.73 m2 in FIDELIO-DKD, and as high as 90 mL/min/1.73m2 in FIGARO-DKD. The two trials excluded people with heart failure with reduced ejection fraction, and those with a serum potassium greater than 4.8 mmol/L.
In the FIDELITY dataset treatment with finerenone led to a significant 17% reduction in the combined incidence of cardiovascular death or first hospitalization for heart failure relative to those who received placebo. This relative risk reduction was not affected by either eGFR or UACR values at baseline, the new analysis showed.
The analysis also demonstrated a nonsignificant trend toward greater reductions in heart failure–related outcomes among study participants who began with an eGFR in the normal range of at least 60 mL/min per 1.73 m2. The researchers also found a nonsignificant trend to a greater reduction in heart failure–related events among those with a UACR of less than 300 mg/g.
Finerenone favors patients with less advanced CKD
In short “the magnitude of the treatment benefit tended to favor patients with less advanced CKD,” concluded the researchers, suggesting that “earlier intervention [with finerenone] in the CKD course is likely to provide the greatest long-term benefit on heart failure–related outcomes.” This led them to further infer “the importance of not only routine assessing eGFR, but also perhaps more importantly, routinely screening for UACR to facilitate early diagnosis and early intervention in patients with type 2 diabetes.”
Findings from FIDELIO-DKD and FIGARO-DKD led to recent guideline additions for finerenone by several medical groups. In August 2022, the American Association of Clinical Endocrinologists released an update to its guideline for managing people with diabetes that recommended treating people with type 2 diabetes with finerenone when they have a UACR of at least 30 mg/g if they are already treated with a maximum-tolerated dose of a renin-angiotensin system inhibitor, have a normal serum potassium level, and have an eGFR of at least 25 mL/min per 1.73 m2. The identical recommendation also appeared in a Consensus Report from the American Diabetes Association and KDIGO, an international organization promoting evidence-based management of patients with CKD.
“Finerenone provides a very important contribution because it improves prognosis even in very well managed patients” with type 2 diabetes, commented Lars Rydén, MD, professor of cardiology at the Karolinska Institute in Stockholm, as designated discussant for the report by Dr. Filippatos at the ESC congress.
The findings from the FIDELITY analysis are “trustworthy, and clinically important,” Dr. Rydén said. When left untreated, diabetic kidney disease “reduces life expectancy by an average of 16 years.”
The finerenone trials were sponsored by Bayer, which markets finerenone (Kerendia). Dr. Filippatos has received lecture fees from Bayer as well as from Amgen, Medtronic, Novartis, Servier, and Vifor. Dr. Green has financial ties to Bayer as well as to Anji, AstraZeneca, Boehringer Ingelheim/Lilly, Hawthorne Effect/Omada, Merck, Novo Nordisk, Pfizer, Roche, Sanofi/Lexicon, and Valo. Dr. Bauersachs has been a consultant to Bayer as well as to Amgen, AstraZeneca, Boehringer Ingelheim, Cardior, Cervia, CVRx, Novartis, Pfizer, and Vifor, and he has received research funding from Abiomed. Dr. Rydén has financial ties to Bayer, Boehringer Ingelheim, Eli Lilly, and Novo Nordisk.
Treatment with finerenone produced roughly similar reductions in heart failure–related outcomes in people with type 2 diabetes and chronic kidney disease (CKD) across the spectrum of kidney function, compared with placebo, including those who had albuminuria but a preserved estimated glomerular filtration rate (eGFR), in a post hoc analysis of pooled data from more than 13,000 people.
The findings, from the two pivotal trials for the agent, “reinforce the importance of routine eGFR and UACR [urinary albumin-to-creatinine ratio] screening” in people with type 2 diabetes to identify new candidates for treatment with finerenone (Kerendia), Gerasimos Filippatos, MD, and coauthors said in a report published online in JACC: Heart Failure.
Among the 13,026 patients in the two combined trials, 40% had a preserved eGFR of greater than 60 mL/min per 1.73 m2 despite also having albuminuria with a UACR of at least 30 mg/g, showing how often this combination occurs. But many clinicians “do not follow the guidelines” and fail to measure the UACR in these patients in routine practice, noted Dr. Filippatos at the annual congress of the European Society of Cardiology in August.
“We now have something to do for these patients,” treat them with finerenone, said Dr. Filippatos, professor and director of heart failure at the Attikon University Hospital, Athens.
The availability of finerenone following its U.S. approval in 2021 means clinicians “must get used to measuring UACR” in people with type 2 diabetes even when their eGFR is normal, especially people with type 2 diabetes plus high cardiovascular disease risk, he said.
The Food and Drug Administration approved finerenone, a nonsteroidal mineralocorticoid receptor antagonist, for treating people with type 2 diabetes and CKD in July 2021, but its uptake has been slow, experts say. In a talk in September 2022 during the annual meeting of the European Association for the Study of Diabetes, Jennifer B. Green, MD, estimated that U.S. uptake of finerenone for appropriate people with type 2 diabetes had not advanced beyond 10%.
A recent review also noted that uptake of screening for elevated UACR in U.S. patients with type 2 diabetes was in the range of 10%-40% during 2017-2019, a “shockingly low rate,” said Dr. Green, a professor and diabetes specialist at Duke University, Durham, N.C.
A new reason to screen for albuminuria
“It’s an extremely important message,” Johann Bauersachs, MD, commented in an interview. Results from “many studies have shown that albuminuria is an excellent additional marker for cardiovascular disease risk. But measurement of albuminuria is not widely done, despite guidelines that recommend annual albuminuria testing in people with type 2 diabetes,” said Dr. Bauersachs, professor and head of the department of cardiology at Hannover (Germany) Medical School.
“Even before there was finerenone, there were reasons to measure UACR, but I hope adding finerenone will help, and more clinicians will incorporate UACR into their routine practice,” said Dr. Bauersachs, who was not involved with the finerenone studies.
The analyses reported by Dr. Filippatos and coauthors used data from two related trials of finerenone, FIDELIO-DKD and FIGARO-DKD, combined by prespecified design into a single dataset, FIDELITY, with a total of 13,026 participants eligible for analysis and followed for a median of 3 years. All had type 2 diabetes and CKD based on having a UACR of at least 30 mg/g. Their eGFR levels could run as high as 74 mL/min per 1.73 m2 in FIDELIO-DKD, and as high as 90 mL/min/1.73m2 in FIGARO-DKD. The two trials excluded people with heart failure with reduced ejection fraction, and those with a serum potassium greater than 4.8 mmol/L.
In the FIDELITY dataset treatment with finerenone led to a significant 17% reduction in the combined incidence of cardiovascular death or first hospitalization for heart failure relative to those who received placebo. This relative risk reduction was not affected by either eGFR or UACR values at baseline, the new analysis showed.
The analysis also demonstrated a nonsignificant trend toward greater reductions in heart failure–related outcomes among study participants who began with an eGFR in the normal range of at least 60 mL/min per 1.73 m2. The researchers also found a nonsignificant trend to a greater reduction in heart failure–related events among those with a UACR of less than 300 mg/g.
Finerenone favors patients with less advanced CKD
In short “the magnitude of the treatment benefit tended to favor patients with less advanced CKD,” concluded the researchers, suggesting that “earlier intervention [with finerenone] in the CKD course is likely to provide the greatest long-term benefit on heart failure–related outcomes.” This led them to further infer “the importance of not only routine assessing eGFR, but also perhaps more importantly, routinely screening for UACR to facilitate early diagnosis and early intervention in patients with type 2 diabetes.”
Findings from FIDELIO-DKD and FIGARO-DKD led to recent guideline additions for finerenone by several medical groups. In August 2022, the American Association of Clinical Endocrinologists released an update to its guideline for managing people with diabetes that recommended treating people with type 2 diabetes with finerenone when they have a UACR of at least 30 mg/g if they are already treated with a maximum-tolerated dose of a renin-angiotensin system inhibitor, have a normal serum potassium level, and have an eGFR of at least 25 mL/min per 1.73 m2. The identical recommendation also appeared in a Consensus Report from the American Diabetes Association and KDIGO, an international organization promoting evidence-based management of patients with CKD.
“Finerenone provides a very important contribution because it improves prognosis even in very well managed patients” with type 2 diabetes, commented Lars Rydén, MD, professor of cardiology at the Karolinska Institute in Stockholm, as designated discussant for the report by Dr. Filippatos at the ESC congress.
The findings from the FIDELITY analysis are “trustworthy, and clinically important,” Dr. Rydén said. When left untreated, diabetic kidney disease “reduces life expectancy by an average of 16 years.”
The finerenone trials were sponsored by Bayer, which markets finerenone (Kerendia). Dr. Filippatos has received lecture fees from Bayer as well as from Amgen, Medtronic, Novartis, Servier, and Vifor. Dr. Green has financial ties to Bayer as well as to Anji, AstraZeneca, Boehringer Ingelheim/Lilly, Hawthorne Effect/Omada, Merck, Novo Nordisk, Pfizer, Roche, Sanofi/Lexicon, and Valo. Dr. Bauersachs has been a consultant to Bayer as well as to Amgen, AstraZeneca, Boehringer Ingelheim, Cardior, Cervia, CVRx, Novartis, Pfizer, and Vifor, and he has received research funding from Abiomed. Dr. Rydén has financial ties to Bayer, Boehringer Ingelheim, Eli Lilly, and Novo Nordisk.
FROM JACC: HEART FAILURE
Climate change magnifies health effects of wildfire smoke in care deserts
DRESSLERVILLE, NEV. – Smoke began billowing into the skies of northwestern Nevada in September, clouding the mountains, dimming the sun – and quashing residents’ hopes that they would be spared from wildfires and the awful air quality the blazes produce.
The lung-irritating particles were blowing in from burning forests in California and settling in Douglas County, Nevada, home to nearly 50,000 people, prompting warnings that air quality had reached hazardous levels.
Those levels meant the air was very unhealthy, bad enough to raise alarms about people’s immediate health care needs and questions about whether worsening pollution could result in long-term health issues. People could increasingly face such risks as climate change makes wildfires, drought, dust storms, and floods more frequent across the United States and the world.
Some people simply feel powerless.
“There’s not much we could do about it,” said Serrell Smokey, chairman of the Washoe Tribe of Nevada and California. The tribe’s land straddles the border between California and Nevada near Lake Tahoe and extends into Douglas County, about 60 miles south of Reno.
Tribe members and other area residents are among millions of people nationwide who this year will experience poor air quality because of wildfires. In September, as smoke settled over Nevada, fire-related air quality alerts were dispatched in six other states: California, Idaho, Montana, Oregon, Washington, and Wyoming.
Yet, by one measure, people who live in Douglas County are better off than those in some other hard-hit areas. Douglas County residents must drive 30 minutes, on average, for medical care from lung specialists called pulmonologists. In other parts of the West and Upper Midwest, however, patients must drive an hour or more, according to data analyzed by GoodRx, a website that tracks prescription drug prices and conducts research.
Specifically, the research found that about 5.5 million Americans live in the 488 counties where drive times to pulmonologists are an hour or more. Much of Nevada and large parts of Montana fall into those gaps between specialists – places that have recently grappled with wildfires that fill the air with smoke and ash, which can cause lung problems or exacerbate existing ones.
Data from the Association of American Medical Colleges shows the number of pulmonary disease specialists in the United States dropped nearly 11% from 2014 to 2019. The group, which is based in Washington, D.C., and represents the academic medicine community, noted that the decline might not be as high as it appears because some physicians are opting to practice pulmonary critical care rather than just pulmonology. Many of those types of pulmonologists work in hospital intensive care units.
About 15,000 pulmonologists are practicing in the United States, according to the GoodRx report. Yet vast swaths of the country have few or none.
“New Mexico has one pulmonologist for the entire southeastern part of state, not counting Las Cruces, which is closer to El Paso,” said Victor Test, MD, a pulmonologist at Texas Tech Physicians.
Dr. Test, one of 13 pulmonologists in the Lubbock, Tex., region, said that his patients from within Texas sometimes drive 4 hours for an appointment and that other people travel from “New Mexico, Oklahoma, even far western Kansas.”
Increases in wildfires and their intensity will likely expand the need for pulmonologists.
“Climate change is going to affect lung disease,” said Nicholas Kenyon, MD, a professor of pulmonary, critical care, and sleep medicine at the University of California, Davis, where he and several other researchers are tracking the effects of wildfires. At his Sacramento practice, Dr. Kenyon said, he sees patients from far northern parts of California, including Eureka, a 5-hour drive from the state capital.
The short-term effects of breathing smoke are pretty well known. People show up in emergency rooms with asthma attacks, exacerbation of COPD, bronchitis, and even pneumonia, Dr. Kenyon said. Some have chest pain or other cardiac concerns.
“But we have very little understanding of what happens over the longer term,” he said. “If people get 2 or 3 weeks of wildfire exposure for 2 or 3 years, does that lead to worsening of asthma or COPD? We just don’t know.”
Fires release multiple pollutants, including carbon dioxide, carbon monoxide, and chemicals like benzene. All fires send particles into the air. Health researchers and air quality experts are most concerned about tiny pieces referred to as particulate matter 2.5. Far smaller than a human hair, the particles can lodge deep in the lungs and have been linked to heart and lung conditions.
Increases in those tiny particles are associated with a greater risk of death from all causes, excluding accidents, homicides, and other nonaccidental causes, for up to 4 days after a population is exposed, according to a 2020 New England Journal of Medicine overview.
The concentration of fine particulate matter is one of five gauges used to calculate the Air Quality Index, a numerical and color-coded index used to let the public know about local air pollution levels. Green denotes good air quality and is given if the total index is 50 or less. When the measurement exceeds 100, the air quality gets an orange label and may be bad for certain groups. Levels over 200 get a red label and are considered unhealthy for everyone.
Government agencies track those levels, as do people who use apps or websites to determine whether it’s safe to go outside.
When the AQI rises above 150, Farah Madhani-Lovely, MD, a pulmonologist, said, Renown Regional Medical Center in Reno shuts its outpatient pulmonary rehabilitation clinic because it doesn’t want to encourage patients to drive in. Some patients from Douglas County opt for care near home, about an hour away. “We don’t want these patients exposed outside because just 1 minute of exposure to the smoke can trigger an exacerbation of their chronic disease,” Dr. Madhani-Lovely said.
Mr. Smokey said connecting with pulmonologists can be difficult for Washoe Tribe members, particularly those who live on the California side of the reservation. “We cannot find providers for them,” he said. “We end up referring them out and sending them hundreds of miles out of their way just to get care that we should be able to provide here.”
Recruiting specialists to rural areas or smaller cities has long been difficult. For one thing, a specialist might be the only one for miles around, “so there’s a tremendous burden in terms of coverage and days off,” Dr. Test said.
Another concern is that physicians tend to train in larger cities and often want to practice in similar places. Even recruiting pulmonary physicians to Lubbock, a city of 260,000 in West Texas, is a challenge, Dr. Test said.
“I love Lubbock,” he said. “But I tell people who have never been here, I say, ‘It’s really flat.’ They don’t understand flat until they get here.”
In Nevada, on days when the air quality is bad, Washoe tribal members try to protect themselves with makeshift air purifiers created from fans, duct tape, and air filters, Mr. Smokey said.
Longer term, Mr. Smokey and other tribal leaders are pushing the Indian Health Service to establish a specialty care hospital in northern Nevada. The closest specialty care hospital for Washoe tribal members is more than 700 miles away, in Phoenix.
It’s difficult because “there’s a need we should be taking care of,” Mr. Smokey said. “But we have to fight for it. And sometimes that fight takes years, years, and years to accomplish.”
A version of this article first appeared on Medscape.com.
DRESSLERVILLE, NEV. – Smoke began billowing into the skies of northwestern Nevada in September, clouding the mountains, dimming the sun – and quashing residents’ hopes that they would be spared from wildfires and the awful air quality the blazes produce.
The lung-irritating particles were blowing in from burning forests in California and settling in Douglas County, Nevada, home to nearly 50,000 people, prompting warnings that air quality had reached hazardous levels.
Those levels meant the air was very unhealthy, bad enough to raise alarms about people’s immediate health care needs and questions about whether worsening pollution could result in long-term health issues. People could increasingly face such risks as climate change makes wildfires, drought, dust storms, and floods more frequent across the United States and the world.
Some people simply feel powerless.
“There’s not much we could do about it,” said Serrell Smokey, chairman of the Washoe Tribe of Nevada and California. The tribe’s land straddles the border between California and Nevada near Lake Tahoe and extends into Douglas County, about 60 miles south of Reno.
Tribe members and other area residents are among millions of people nationwide who this year will experience poor air quality because of wildfires. In September, as smoke settled over Nevada, fire-related air quality alerts were dispatched in six other states: California, Idaho, Montana, Oregon, Washington, and Wyoming.
Yet, by one measure, people who live in Douglas County are better off than those in some other hard-hit areas. Douglas County residents must drive 30 minutes, on average, for medical care from lung specialists called pulmonologists. In other parts of the West and Upper Midwest, however, patients must drive an hour or more, according to data analyzed by GoodRx, a website that tracks prescription drug prices and conducts research.
Specifically, the research found that about 5.5 million Americans live in the 488 counties where drive times to pulmonologists are an hour or more. Much of Nevada and large parts of Montana fall into those gaps between specialists – places that have recently grappled with wildfires that fill the air with smoke and ash, which can cause lung problems or exacerbate existing ones.
Data from the Association of American Medical Colleges shows the number of pulmonary disease specialists in the United States dropped nearly 11% from 2014 to 2019. The group, which is based in Washington, D.C., and represents the academic medicine community, noted that the decline might not be as high as it appears because some physicians are opting to practice pulmonary critical care rather than just pulmonology. Many of those types of pulmonologists work in hospital intensive care units.
About 15,000 pulmonologists are practicing in the United States, according to the GoodRx report. Yet vast swaths of the country have few or none.
“New Mexico has one pulmonologist for the entire southeastern part of state, not counting Las Cruces, which is closer to El Paso,” said Victor Test, MD, a pulmonologist at Texas Tech Physicians.
Dr. Test, one of 13 pulmonologists in the Lubbock, Tex., region, said that his patients from within Texas sometimes drive 4 hours for an appointment and that other people travel from “New Mexico, Oklahoma, even far western Kansas.”
Increases in wildfires and their intensity will likely expand the need for pulmonologists.
“Climate change is going to affect lung disease,” said Nicholas Kenyon, MD, a professor of pulmonary, critical care, and sleep medicine at the University of California, Davis, where he and several other researchers are tracking the effects of wildfires. At his Sacramento practice, Dr. Kenyon said, he sees patients from far northern parts of California, including Eureka, a 5-hour drive from the state capital.
The short-term effects of breathing smoke are pretty well known. People show up in emergency rooms with asthma attacks, exacerbation of COPD, bronchitis, and even pneumonia, Dr. Kenyon said. Some have chest pain or other cardiac concerns.
“But we have very little understanding of what happens over the longer term,” he said. “If people get 2 or 3 weeks of wildfire exposure for 2 or 3 years, does that lead to worsening of asthma or COPD? We just don’t know.”
Fires release multiple pollutants, including carbon dioxide, carbon monoxide, and chemicals like benzene. All fires send particles into the air. Health researchers and air quality experts are most concerned about tiny pieces referred to as particulate matter 2.5. Far smaller than a human hair, the particles can lodge deep in the lungs and have been linked to heart and lung conditions.
Increases in those tiny particles are associated with a greater risk of death from all causes, excluding accidents, homicides, and other nonaccidental causes, for up to 4 days after a population is exposed, according to a 2020 New England Journal of Medicine overview.
The concentration of fine particulate matter is one of five gauges used to calculate the Air Quality Index, a numerical and color-coded index used to let the public know about local air pollution levels. Green denotes good air quality and is given if the total index is 50 or less. When the measurement exceeds 100, the air quality gets an orange label and may be bad for certain groups. Levels over 200 get a red label and are considered unhealthy for everyone.
Government agencies track those levels, as do people who use apps or websites to determine whether it’s safe to go outside.
When the AQI rises above 150, Farah Madhani-Lovely, MD, a pulmonologist, said, Renown Regional Medical Center in Reno shuts its outpatient pulmonary rehabilitation clinic because it doesn’t want to encourage patients to drive in. Some patients from Douglas County opt for care near home, about an hour away. “We don’t want these patients exposed outside because just 1 minute of exposure to the smoke can trigger an exacerbation of their chronic disease,” Dr. Madhani-Lovely said.
Mr. Smokey said connecting with pulmonologists can be difficult for Washoe Tribe members, particularly those who live on the California side of the reservation. “We cannot find providers for them,” he said. “We end up referring them out and sending them hundreds of miles out of their way just to get care that we should be able to provide here.”
Recruiting specialists to rural areas or smaller cities has long been difficult. For one thing, a specialist might be the only one for miles around, “so there’s a tremendous burden in terms of coverage and days off,” Dr. Test said.
Another concern is that physicians tend to train in larger cities and often want to practice in similar places. Even recruiting pulmonary physicians to Lubbock, a city of 260,000 in West Texas, is a challenge, Dr. Test said.
“I love Lubbock,” he said. “But I tell people who have never been here, I say, ‘It’s really flat.’ They don’t understand flat until they get here.”
In Nevada, on days when the air quality is bad, Washoe tribal members try to protect themselves with makeshift air purifiers created from fans, duct tape, and air filters, Mr. Smokey said.
Longer term, Mr. Smokey and other tribal leaders are pushing the Indian Health Service to establish a specialty care hospital in northern Nevada. The closest specialty care hospital for Washoe tribal members is more than 700 miles away, in Phoenix.
It’s difficult because “there’s a need we should be taking care of,” Mr. Smokey said. “But we have to fight for it. And sometimes that fight takes years, years, and years to accomplish.”
A version of this article first appeared on Medscape.com.
DRESSLERVILLE, NEV. – Smoke began billowing into the skies of northwestern Nevada in September, clouding the mountains, dimming the sun – and quashing residents’ hopes that they would be spared from wildfires and the awful air quality the blazes produce.
The lung-irritating particles were blowing in from burning forests in California and settling in Douglas County, Nevada, home to nearly 50,000 people, prompting warnings that air quality had reached hazardous levels.
Those levels meant the air was very unhealthy, bad enough to raise alarms about people’s immediate health care needs and questions about whether worsening pollution could result in long-term health issues. People could increasingly face such risks as climate change makes wildfires, drought, dust storms, and floods more frequent across the United States and the world.
Some people simply feel powerless.
“There’s not much we could do about it,” said Serrell Smokey, chairman of the Washoe Tribe of Nevada and California. The tribe’s land straddles the border between California and Nevada near Lake Tahoe and extends into Douglas County, about 60 miles south of Reno.
Tribe members and other area residents are among millions of people nationwide who this year will experience poor air quality because of wildfires. In September, as smoke settled over Nevada, fire-related air quality alerts were dispatched in six other states: California, Idaho, Montana, Oregon, Washington, and Wyoming.
Yet, by one measure, people who live in Douglas County are better off than those in some other hard-hit areas. Douglas County residents must drive 30 minutes, on average, for medical care from lung specialists called pulmonologists. In other parts of the West and Upper Midwest, however, patients must drive an hour or more, according to data analyzed by GoodRx, a website that tracks prescription drug prices and conducts research.
Specifically, the research found that about 5.5 million Americans live in the 488 counties where drive times to pulmonologists are an hour or more. Much of Nevada and large parts of Montana fall into those gaps between specialists – places that have recently grappled with wildfires that fill the air with smoke and ash, which can cause lung problems or exacerbate existing ones.
Data from the Association of American Medical Colleges shows the number of pulmonary disease specialists in the United States dropped nearly 11% from 2014 to 2019. The group, which is based in Washington, D.C., and represents the academic medicine community, noted that the decline might not be as high as it appears because some physicians are opting to practice pulmonary critical care rather than just pulmonology. Many of those types of pulmonologists work in hospital intensive care units.
About 15,000 pulmonologists are practicing in the United States, according to the GoodRx report. Yet vast swaths of the country have few or none.
“New Mexico has one pulmonologist for the entire southeastern part of state, not counting Las Cruces, which is closer to El Paso,” said Victor Test, MD, a pulmonologist at Texas Tech Physicians.
Dr. Test, one of 13 pulmonologists in the Lubbock, Tex., region, said that his patients from within Texas sometimes drive 4 hours for an appointment and that other people travel from “New Mexico, Oklahoma, even far western Kansas.”
Increases in wildfires and their intensity will likely expand the need for pulmonologists.
“Climate change is going to affect lung disease,” said Nicholas Kenyon, MD, a professor of pulmonary, critical care, and sleep medicine at the University of California, Davis, where he and several other researchers are tracking the effects of wildfires. At his Sacramento practice, Dr. Kenyon said, he sees patients from far northern parts of California, including Eureka, a 5-hour drive from the state capital.
The short-term effects of breathing smoke are pretty well known. People show up in emergency rooms with asthma attacks, exacerbation of COPD, bronchitis, and even pneumonia, Dr. Kenyon said. Some have chest pain or other cardiac concerns.
“But we have very little understanding of what happens over the longer term,” he said. “If people get 2 or 3 weeks of wildfire exposure for 2 or 3 years, does that lead to worsening of asthma or COPD? We just don’t know.”
Fires release multiple pollutants, including carbon dioxide, carbon monoxide, and chemicals like benzene. All fires send particles into the air. Health researchers and air quality experts are most concerned about tiny pieces referred to as particulate matter 2.5. Far smaller than a human hair, the particles can lodge deep in the lungs and have been linked to heart and lung conditions.
Increases in those tiny particles are associated with a greater risk of death from all causes, excluding accidents, homicides, and other nonaccidental causes, for up to 4 days after a population is exposed, according to a 2020 New England Journal of Medicine overview.
The concentration of fine particulate matter is one of five gauges used to calculate the Air Quality Index, a numerical and color-coded index used to let the public know about local air pollution levels. Green denotes good air quality and is given if the total index is 50 or less. When the measurement exceeds 100, the air quality gets an orange label and may be bad for certain groups. Levels over 200 get a red label and are considered unhealthy for everyone.
Government agencies track those levels, as do people who use apps or websites to determine whether it’s safe to go outside.
When the AQI rises above 150, Farah Madhani-Lovely, MD, a pulmonologist, said, Renown Regional Medical Center in Reno shuts its outpatient pulmonary rehabilitation clinic because it doesn’t want to encourage patients to drive in. Some patients from Douglas County opt for care near home, about an hour away. “We don’t want these patients exposed outside because just 1 minute of exposure to the smoke can trigger an exacerbation of their chronic disease,” Dr. Madhani-Lovely said.
Mr. Smokey said connecting with pulmonologists can be difficult for Washoe Tribe members, particularly those who live on the California side of the reservation. “We cannot find providers for them,” he said. “We end up referring them out and sending them hundreds of miles out of their way just to get care that we should be able to provide here.”
Recruiting specialists to rural areas or smaller cities has long been difficult. For one thing, a specialist might be the only one for miles around, “so there’s a tremendous burden in terms of coverage and days off,” Dr. Test said.
Another concern is that physicians tend to train in larger cities and often want to practice in similar places. Even recruiting pulmonary physicians to Lubbock, a city of 260,000 in West Texas, is a challenge, Dr. Test said.
“I love Lubbock,” he said. “But I tell people who have never been here, I say, ‘It’s really flat.’ They don’t understand flat until they get here.”
In Nevada, on days when the air quality is bad, Washoe tribal members try to protect themselves with makeshift air purifiers created from fans, duct tape, and air filters, Mr. Smokey said.
Longer term, Mr. Smokey and other tribal leaders are pushing the Indian Health Service to establish a specialty care hospital in northern Nevada. The closest specialty care hospital for Washoe tribal members is more than 700 miles away, in Phoenix.
It’s difficult because “there’s a need we should be taking care of,” Mr. Smokey said. “But we have to fight for it. And sometimes that fight takes years, years, and years to accomplish.”
A version of this article first appeared on Medscape.com.
You and the skeptical patient: Who’s the doctor here?
“I spoke to him on many occasions about the dangers of COVID, but he just didn’t believe me,” said Dr. Hood, an internist in Lexington, Ky. “He just didn’t give me enough time to help him. He waited to let me know he was ill with COVID and took days to pick up the medicine. Unfortunately, he then passed away.”
The rise of the skeptical patient
It can be extremely frustrating for doctors when patients question or disbelieve their physician’s medical advice and explanations. And many physicians resent the amount of time they spend trying to explain or make their case, especially during a busy day. But patients’ skepticism about the validity of some treatments seems to be increasing.
“Patients are now more likely to have their own medical explanation for their complaint than they used to, and that can be bad for their health,” Dr. Hood said.
Dr. Hood sees medical cynicism as part of Americans’ growing distrust of experts, leveraged by easy access to the internet. “When people Google, they tend to look for support of their opinions, rather than arrive at a fully educated decision.”
Only about half of patients believe their physicians “provide fair and accurate treatment information all or most of the time,” according to a 2019 survey by the Pew Research Center.
Patients’ distrust has become more obvious during the COVID-19 pandemic, said John Schumann, MD, an internist with Oak Street Health, a practice with more than 500 physicians and other providers in 20 states, treating almost exclusively Medicare patients.
“The skeptics became more entrenched during the pandemic,” said Dr. Schumann, who is based in Tulsa, Okla. “They may think the COVID vaccines were approved too quickly, or believe the pandemic itself is a hoax.”
“There’s a lot of antiscience rhetoric now,” Dr. Schumann added. “I’d say about half of my patients are comfortable with science-based decisions and the other half are not.”
What are patients mistrustful about?
Patients’ suspicions of certain therapies began long before the pandemic. In dermatology, for example, some patients refuse to take topical steroids, said Steven R. Feldman, MD, a dermatologist in Winston-Salem, N.C.
“Their distrust is usually based on anecdotal stories they read about,” he noted. “Patients in other specialties are dead set against vaccinations.”
In addition to refusing treatments and inoculations, some patients ask for questionable regimens mentioned in the news. “Some patients have demanded hydroxychloroquine or Noromectin, drugs that are unproven in the treatment of COVID,” Dr. Schumann said. “We refuse to prescribe them.”
Dr. Hood said patients’ reluctance to follow medical advice can often be based on cost. “I have a patient who was more willing to save $20 than to save his life. But when the progression of his test results fit my predictions, he became more willing to take treatments. I had to wait for the opportune moment to convince him.”
Many naysayer patients keep their views to themselves, and physicians may be unaware that the patients are stonewalling. A 2006 study estimated that about 10%-16% of primary care patients actively resist medical authority.
Dr. Schumann cited patients who don’t want to hear an upsetting diagnosis. “Some patients might refuse to take a biopsy to see if they have cancer because they don’t want to know,” he said. “In many cases, they simply won’t get the biopsy and won’t tell the doctor that they didn’t.”
Sometimes skeptics’ arguments have merit
Some patients’ concerns can be valid, such as when they refuse to go on statins, said Zain Hakeem, DO, a physician in Austin, Tex.
“In some cases, I feel that statins are not necessary,” he said. “The science on statins for primary prevention is not strong, although they should be used for exceedingly high-risk patients.”
Certain patients, especially those with chronic conditions, do a great deal of research, using legitimate sources on the Web, and their research is well supported.
However, these patients can be overconfident in their conclusions. Several studies have shown that with just a little experience, people can replace beginners’ caution with a false sense of competence.
For example, “Patients may not weigh the risks correctly,” Dr. Hakeem said. “They can be more concerned about the risk of having their colon perforated during a colonoscopy, while the risk of cancer if they don’t have a colonoscopy is much higher.”
Some highly successful people may be more likely to trust their own medical instincts. When Steve Jobs, the founder of Apple, was diagnosed with pancreatic cancer in 2003, he put off surgery for 9 months while he tried to cure his disease with a vegan diet, acupuncture, herbs, bowel cleansings, and other remedies he read about. He died in 2011. Some experts believe that delay hastened his death.
Of course, not all physicians’ diagnoses or treatments are correct. One study indicated doctors’ diagnostic error rate could be as high as 15%. And just as patients can be overconfident in their conclusions, so can doctors. Another study found that physicians’ stated confidence in their diagnosis was only slightly affected by the inaccuracy of that diagnosis or the difficulty of the case.
Best ways to deal with cynical patients
Patients’ skepticism can frustrate doctors, reduce the efficiency of care delivery, and interfere with recovery. What can doctors do to deal with these problems?
1. Build the patient’s trust in you. “Getting patients to adhere to your advice involves making sure they feel they have a caring doctor whom they trust,” Dr. Feldman said.
“I want to show patients that I am entirely focused on them,” he added. “For example, I may rush to the door of the exam room from my last appointment, but I open the door very slowly and deliberately, because I want the patient to see that I won’t hurry with them.”
2. Spend time with the patient. Familiarity builds trust. Dr. Schumann said doctors at Oak Street Health see their patients an average of six to eight times a year, an unusually high number. “The more patients see their physicians, the more likely they are to trust them.”
3. Keep up to date. “I make sure I’m up to date with the literature, and I try to present a truthful message,” Dr. Hood said. “For instance, my research showed that inflammation played a strong role in developing complications from COVID, so I wrote a detailed treatment protocol aimed at the inflammation and the immune response, which has been very effective.”
4. Confront patients tactfully. Patients who do research on the Web don’t want to be scolded, Dr. Feldman said. In fact, he praises them, even if he doesn’t agree with their findings. “I might say: ‘What a relief to finally find patients who’ve taken the time to educate themselves before coming here.’ ”
Dr. Feldman is careful not to dispute patients’ conclusions. “Debating the issues is not an effective approach to get patients to trust you. The last thing you want to tell a patient is: ‘Listen to me! I’m an expert.’ People just dig in.”
However, it does help to give patients feedback. “I’m a big fan of patients arguing with me,” Dr. Hakeem said. “It means you can straighten out misunderstandings and improve decision-making.”
5. Explain your reasoning. “You need to communicate clearly and show them your thinking,” Dr. Hood said. “For instance, I’ll explain why a patient has a strong risk for heart attack.”
6. Acknowledge uncertainties. “The doctor may present the science as far more certain than it is,” Dr. Hakeem said. “If you don’t acknowledge the uncertainties, you could break the patient’s trust in you.”
7. Don’t use a lot of numbers. “Data is not a good tool to convince patients,” Dr. Feldman said. “The human brain isn’t designed to work that way.”
If you want to use numbers to show clinical risk, Dr. Hakeem advisd using natural frequencies, such as 10 out of 10,000, which is less confusing to the patient than the equivalent percentage of 0.1%.
It can be helpful to refer to familiar concepts. One way to understand a risk is to compare it with risks in daily life, such as the dangers of driving or falling in the shower, Dr. Hakeem added.
Dr. Feldman often refers to another person’s experience when presenting his medical advice. “I might say to the patient: ‘You remind me of another patient I had. They were sitting in the same chair you’re sitting in. They did really well on this drug, and I think it’s probably the best choice for you, too.’ ”
8. Adopt shared decision-making. This approach involves empowering the patient to become an equal partner in medical decisions. The patient is given information through portals and is encouraged to do research. Critics, however, say that most patients don’t want this degree of empowerment and would rather depend on the doctor’s advice.
Conclusion
It’s often impossible to get through to a skeptical patient, which can be disheartening for doctors. “Physicians want to do what is best for the patient, so when the patient doesn’t listen, they may take it personally,” Dr. Hood said. “But you always have to remember, the patient is the one with disease, and it’s up to the patient to open the door.”
Still, some skeptical patients ultimately change their minds. Dr. Schumann said patients who initially declined the COVID vaccine eventually decided to get it. “It often took them more than a year. but it’s never too late.”
A version of this article first appeared on Medscape.com.
“I spoke to him on many occasions about the dangers of COVID, but he just didn’t believe me,” said Dr. Hood, an internist in Lexington, Ky. “He just didn’t give me enough time to help him. He waited to let me know he was ill with COVID and took days to pick up the medicine. Unfortunately, he then passed away.”
The rise of the skeptical patient
It can be extremely frustrating for doctors when patients question or disbelieve their physician’s medical advice and explanations. And many physicians resent the amount of time they spend trying to explain or make their case, especially during a busy day. But patients’ skepticism about the validity of some treatments seems to be increasing.
“Patients are now more likely to have their own medical explanation for their complaint than they used to, and that can be bad for their health,” Dr. Hood said.
Dr. Hood sees medical cynicism as part of Americans’ growing distrust of experts, leveraged by easy access to the internet. “When people Google, they tend to look for support of their opinions, rather than arrive at a fully educated decision.”
Only about half of patients believe their physicians “provide fair and accurate treatment information all or most of the time,” according to a 2019 survey by the Pew Research Center.
Patients’ distrust has become more obvious during the COVID-19 pandemic, said John Schumann, MD, an internist with Oak Street Health, a practice with more than 500 physicians and other providers in 20 states, treating almost exclusively Medicare patients.
“The skeptics became more entrenched during the pandemic,” said Dr. Schumann, who is based in Tulsa, Okla. “They may think the COVID vaccines were approved too quickly, or believe the pandemic itself is a hoax.”
“There’s a lot of antiscience rhetoric now,” Dr. Schumann added. “I’d say about half of my patients are comfortable with science-based decisions and the other half are not.”
What are patients mistrustful about?
Patients’ suspicions of certain therapies began long before the pandemic. In dermatology, for example, some patients refuse to take topical steroids, said Steven R. Feldman, MD, a dermatologist in Winston-Salem, N.C.
“Their distrust is usually based on anecdotal stories they read about,” he noted. “Patients in other specialties are dead set against vaccinations.”
In addition to refusing treatments and inoculations, some patients ask for questionable regimens mentioned in the news. “Some patients have demanded hydroxychloroquine or Noromectin, drugs that are unproven in the treatment of COVID,” Dr. Schumann said. “We refuse to prescribe them.”
Dr. Hood said patients’ reluctance to follow medical advice can often be based on cost. “I have a patient who was more willing to save $20 than to save his life. But when the progression of his test results fit my predictions, he became more willing to take treatments. I had to wait for the opportune moment to convince him.”
Many naysayer patients keep their views to themselves, and physicians may be unaware that the patients are stonewalling. A 2006 study estimated that about 10%-16% of primary care patients actively resist medical authority.
Dr. Schumann cited patients who don’t want to hear an upsetting diagnosis. “Some patients might refuse to take a biopsy to see if they have cancer because they don’t want to know,” he said. “In many cases, they simply won’t get the biopsy and won’t tell the doctor that they didn’t.”
Sometimes skeptics’ arguments have merit
Some patients’ concerns can be valid, such as when they refuse to go on statins, said Zain Hakeem, DO, a physician in Austin, Tex.
“In some cases, I feel that statins are not necessary,” he said. “The science on statins for primary prevention is not strong, although they should be used for exceedingly high-risk patients.”
Certain patients, especially those with chronic conditions, do a great deal of research, using legitimate sources on the Web, and their research is well supported.
However, these patients can be overconfident in their conclusions. Several studies have shown that with just a little experience, people can replace beginners’ caution with a false sense of competence.
For example, “Patients may not weigh the risks correctly,” Dr. Hakeem said. “They can be more concerned about the risk of having their colon perforated during a colonoscopy, while the risk of cancer if they don’t have a colonoscopy is much higher.”
Some highly successful people may be more likely to trust their own medical instincts. When Steve Jobs, the founder of Apple, was diagnosed with pancreatic cancer in 2003, he put off surgery for 9 months while he tried to cure his disease with a vegan diet, acupuncture, herbs, bowel cleansings, and other remedies he read about. He died in 2011. Some experts believe that delay hastened his death.
Of course, not all physicians’ diagnoses or treatments are correct. One study indicated doctors’ diagnostic error rate could be as high as 15%. And just as patients can be overconfident in their conclusions, so can doctors. Another study found that physicians’ stated confidence in their diagnosis was only slightly affected by the inaccuracy of that diagnosis or the difficulty of the case.
Best ways to deal with cynical patients
Patients’ skepticism can frustrate doctors, reduce the efficiency of care delivery, and interfere with recovery. What can doctors do to deal with these problems?
1. Build the patient’s trust in you. “Getting patients to adhere to your advice involves making sure they feel they have a caring doctor whom they trust,” Dr. Feldman said.
“I want to show patients that I am entirely focused on them,” he added. “For example, I may rush to the door of the exam room from my last appointment, but I open the door very slowly and deliberately, because I want the patient to see that I won’t hurry with them.”
2. Spend time with the patient. Familiarity builds trust. Dr. Schumann said doctors at Oak Street Health see their patients an average of six to eight times a year, an unusually high number. “The more patients see their physicians, the more likely they are to trust them.”
3. Keep up to date. “I make sure I’m up to date with the literature, and I try to present a truthful message,” Dr. Hood said. “For instance, my research showed that inflammation played a strong role in developing complications from COVID, so I wrote a detailed treatment protocol aimed at the inflammation and the immune response, which has been very effective.”
4. Confront patients tactfully. Patients who do research on the Web don’t want to be scolded, Dr. Feldman said. In fact, he praises them, even if he doesn’t agree with their findings. “I might say: ‘What a relief to finally find patients who’ve taken the time to educate themselves before coming here.’ ”
Dr. Feldman is careful not to dispute patients’ conclusions. “Debating the issues is not an effective approach to get patients to trust you. The last thing you want to tell a patient is: ‘Listen to me! I’m an expert.’ People just dig in.”
However, it does help to give patients feedback. “I’m a big fan of patients arguing with me,” Dr. Hakeem said. “It means you can straighten out misunderstandings and improve decision-making.”
5. Explain your reasoning. “You need to communicate clearly and show them your thinking,” Dr. Hood said. “For instance, I’ll explain why a patient has a strong risk for heart attack.”
6. Acknowledge uncertainties. “The doctor may present the science as far more certain than it is,” Dr. Hakeem said. “If you don’t acknowledge the uncertainties, you could break the patient’s trust in you.”
7. Don’t use a lot of numbers. “Data is not a good tool to convince patients,” Dr. Feldman said. “The human brain isn’t designed to work that way.”
If you want to use numbers to show clinical risk, Dr. Hakeem advisd using natural frequencies, such as 10 out of 10,000, which is less confusing to the patient than the equivalent percentage of 0.1%.
It can be helpful to refer to familiar concepts. One way to understand a risk is to compare it with risks in daily life, such as the dangers of driving or falling in the shower, Dr. Hakeem added.
Dr. Feldman often refers to another person’s experience when presenting his medical advice. “I might say to the patient: ‘You remind me of another patient I had. They were sitting in the same chair you’re sitting in. They did really well on this drug, and I think it’s probably the best choice for you, too.’ ”
8. Adopt shared decision-making. This approach involves empowering the patient to become an equal partner in medical decisions. The patient is given information through portals and is encouraged to do research. Critics, however, say that most patients don’t want this degree of empowerment and would rather depend on the doctor’s advice.
Conclusion
It’s often impossible to get through to a skeptical patient, which can be disheartening for doctors. “Physicians want to do what is best for the patient, so when the patient doesn’t listen, they may take it personally,” Dr. Hood said. “But you always have to remember, the patient is the one with disease, and it’s up to the patient to open the door.”
Still, some skeptical patients ultimately change their minds. Dr. Schumann said patients who initially declined the COVID vaccine eventually decided to get it. “It often took them more than a year. but it’s never too late.”
A version of this article first appeared on Medscape.com.
“I spoke to him on many occasions about the dangers of COVID, but he just didn’t believe me,” said Dr. Hood, an internist in Lexington, Ky. “He just didn’t give me enough time to help him. He waited to let me know he was ill with COVID and took days to pick up the medicine. Unfortunately, he then passed away.”
The rise of the skeptical patient
It can be extremely frustrating for doctors when patients question or disbelieve their physician’s medical advice and explanations. And many physicians resent the amount of time they spend trying to explain or make their case, especially during a busy day. But patients’ skepticism about the validity of some treatments seems to be increasing.
“Patients are now more likely to have their own medical explanation for their complaint than they used to, and that can be bad for their health,” Dr. Hood said.
Dr. Hood sees medical cynicism as part of Americans’ growing distrust of experts, leveraged by easy access to the internet. “When people Google, they tend to look for support of their opinions, rather than arrive at a fully educated decision.”
Only about half of patients believe their physicians “provide fair and accurate treatment information all or most of the time,” according to a 2019 survey by the Pew Research Center.
Patients’ distrust has become more obvious during the COVID-19 pandemic, said John Schumann, MD, an internist with Oak Street Health, a practice with more than 500 physicians and other providers in 20 states, treating almost exclusively Medicare patients.
“The skeptics became more entrenched during the pandemic,” said Dr. Schumann, who is based in Tulsa, Okla. “They may think the COVID vaccines were approved too quickly, or believe the pandemic itself is a hoax.”
“There’s a lot of antiscience rhetoric now,” Dr. Schumann added. “I’d say about half of my patients are comfortable with science-based decisions and the other half are not.”
What are patients mistrustful about?
Patients’ suspicions of certain therapies began long before the pandemic. In dermatology, for example, some patients refuse to take topical steroids, said Steven R. Feldman, MD, a dermatologist in Winston-Salem, N.C.
“Their distrust is usually based on anecdotal stories they read about,” he noted. “Patients in other specialties are dead set against vaccinations.”
In addition to refusing treatments and inoculations, some patients ask for questionable regimens mentioned in the news. “Some patients have demanded hydroxychloroquine or Noromectin, drugs that are unproven in the treatment of COVID,” Dr. Schumann said. “We refuse to prescribe them.”
Dr. Hood said patients’ reluctance to follow medical advice can often be based on cost. “I have a patient who was more willing to save $20 than to save his life. But when the progression of his test results fit my predictions, he became more willing to take treatments. I had to wait for the opportune moment to convince him.”
Many naysayer patients keep their views to themselves, and physicians may be unaware that the patients are stonewalling. A 2006 study estimated that about 10%-16% of primary care patients actively resist medical authority.
Dr. Schumann cited patients who don’t want to hear an upsetting diagnosis. “Some patients might refuse to take a biopsy to see if they have cancer because they don’t want to know,” he said. “In many cases, they simply won’t get the biopsy and won’t tell the doctor that they didn’t.”
Sometimes skeptics’ arguments have merit
Some patients’ concerns can be valid, such as when they refuse to go on statins, said Zain Hakeem, DO, a physician in Austin, Tex.
“In some cases, I feel that statins are not necessary,” he said. “The science on statins for primary prevention is not strong, although they should be used for exceedingly high-risk patients.”
Certain patients, especially those with chronic conditions, do a great deal of research, using legitimate sources on the Web, and their research is well supported.
However, these patients can be overconfident in their conclusions. Several studies have shown that with just a little experience, people can replace beginners’ caution with a false sense of competence.
For example, “Patients may not weigh the risks correctly,” Dr. Hakeem said. “They can be more concerned about the risk of having their colon perforated during a colonoscopy, while the risk of cancer if they don’t have a colonoscopy is much higher.”
Some highly successful people may be more likely to trust their own medical instincts. When Steve Jobs, the founder of Apple, was diagnosed with pancreatic cancer in 2003, he put off surgery for 9 months while he tried to cure his disease with a vegan diet, acupuncture, herbs, bowel cleansings, and other remedies he read about. He died in 2011. Some experts believe that delay hastened his death.
Of course, not all physicians’ diagnoses or treatments are correct. One study indicated doctors’ diagnostic error rate could be as high as 15%. And just as patients can be overconfident in their conclusions, so can doctors. Another study found that physicians’ stated confidence in their diagnosis was only slightly affected by the inaccuracy of that diagnosis or the difficulty of the case.
Best ways to deal with cynical patients
Patients’ skepticism can frustrate doctors, reduce the efficiency of care delivery, and interfere with recovery. What can doctors do to deal with these problems?
1. Build the patient’s trust in you. “Getting patients to adhere to your advice involves making sure they feel they have a caring doctor whom they trust,” Dr. Feldman said.
“I want to show patients that I am entirely focused on them,” he added. “For example, I may rush to the door of the exam room from my last appointment, but I open the door very slowly and deliberately, because I want the patient to see that I won’t hurry with them.”
2. Spend time with the patient. Familiarity builds trust. Dr. Schumann said doctors at Oak Street Health see their patients an average of six to eight times a year, an unusually high number. “The more patients see their physicians, the more likely they are to trust them.”
3. Keep up to date. “I make sure I’m up to date with the literature, and I try to present a truthful message,” Dr. Hood said. “For instance, my research showed that inflammation played a strong role in developing complications from COVID, so I wrote a detailed treatment protocol aimed at the inflammation and the immune response, which has been very effective.”
4. Confront patients tactfully. Patients who do research on the Web don’t want to be scolded, Dr. Feldman said. In fact, he praises them, even if he doesn’t agree with their findings. “I might say: ‘What a relief to finally find patients who’ve taken the time to educate themselves before coming here.’ ”
Dr. Feldman is careful not to dispute patients’ conclusions. “Debating the issues is not an effective approach to get patients to trust you. The last thing you want to tell a patient is: ‘Listen to me! I’m an expert.’ People just dig in.”
However, it does help to give patients feedback. “I’m a big fan of patients arguing with me,” Dr. Hakeem said. “It means you can straighten out misunderstandings and improve decision-making.”
5. Explain your reasoning. “You need to communicate clearly and show them your thinking,” Dr. Hood said. “For instance, I’ll explain why a patient has a strong risk for heart attack.”
6. Acknowledge uncertainties. “The doctor may present the science as far more certain than it is,” Dr. Hakeem said. “If you don’t acknowledge the uncertainties, you could break the patient’s trust in you.”
7. Don’t use a lot of numbers. “Data is not a good tool to convince patients,” Dr. Feldman said. “The human brain isn’t designed to work that way.”
If you want to use numbers to show clinical risk, Dr. Hakeem advisd using natural frequencies, such as 10 out of 10,000, which is less confusing to the patient than the equivalent percentage of 0.1%.
It can be helpful to refer to familiar concepts. One way to understand a risk is to compare it with risks in daily life, such as the dangers of driving or falling in the shower, Dr. Hakeem added.
Dr. Feldman often refers to another person’s experience when presenting his medical advice. “I might say to the patient: ‘You remind me of another patient I had. They were sitting in the same chair you’re sitting in. They did really well on this drug, and I think it’s probably the best choice for you, too.’ ”
8. Adopt shared decision-making. This approach involves empowering the patient to become an equal partner in medical decisions. The patient is given information through portals and is encouraged to do research. Critics, however, say that most patients don’t want this degree of empowerment and would rather depend on the doctor’s advice.
Conclusion
It’s often impossible to get through to a skeptical patient, which can be disheartening for doctors. “Physicians want to do what is best for the patient, so when the patient doesn’t listen, they may take it personally,” Dr. Hood said. “But you always have to remember, the patient is the one with disease, and it’s up to the patient to open the door.”
Still, some skeptical patients ultimately change their minds. Dr. Schumann said patients who initially declined the COVID vaccine eventually decided to get it. “It often took them more than a year. but it’s never too late.”
A version of this article first appeared on Medscape.com.