User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
nav[contains(@class, 'nav-ce-stack nav-ce-stack__large-screen')]
header[@id='header']
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'main-prefix')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
div[contains(@class, 'view-medstat-quiz-listing-panes')]
div[contains(@class, 'pane-article-sidebar-latest-news')]
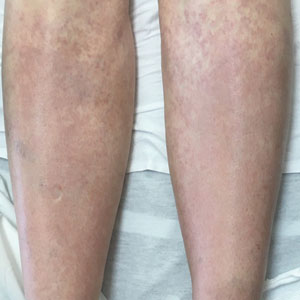
COVID tied to spike in deaths in chronic liver disease with diabetes
The COVID-19 pandemic fueled a sharp uptick in deaths related to chronic liver disease and cirrhosis among people with diabetes, largely owing to nonalcoholic fatty liver disease (NAFLD) and alcohol-related liver disease (ALD), new data show.
“Our observations confirm that COVID-19 had a higher likelihood of impacting vulnerable populations with pre-existing chronic liver diseases and diabetes, with a death rate as high as 10% in individuals with co-existing chronic liver disease and diabetes,” write the authors.
“The inability to attend regular outpatient clinics for close monitoring and treatment accompanied by diversion of health care resources to COVID-19 care may have resulted in the suboptimal or delayed clinical care of individuals with diabetes and chronic liver disease during the COVID-19 pandemic,” they add.
Donghee Kim, MD, PhD, with the Division of Gastroenterology and Hepatology, Stanford (Calif.) University School of Medicine, and colleagues report their findings in the journal Digestive and Liver Disease.
Vulnerable group
The researchers used U.S. national mortality data (2017-2020) to estimate chronic liver disease–related mortality trends among individuals with diabetes before and during the COVID-19 pandemic.
Before the pandemic, the quarterly mortality for chronic liver disease remained stable (quarterly percentage change, 0.6%) but then sharply increased during the pandemic (QPC, 8.6%).
A similar trend was seen with cirrhosis-related mortality (QPC, 0.3% before the pandemic vs. 8.4% during the pandemic).
NAFLD and ALD mortality among individuals with diabetes was steadily increasing before the pandemic (QPC, 4.2% and 3.5%, respectively) but showed a more rapid increase during the pandemic (QPC, 9.6% and 7.7%, respectively).
ALD-related mortality in men was more than threefold higher than in women, while NAFLD-related mortality in women was more than twofold higher than in men.
Mortality for hepatitis C virus infection declined before the pandemic (QPC, −3.3%) and remained stable during the pandemic.
COVID-19–related mortality among adults with chronic liver disease and diabetes also rose sharply during the pandemic – from 0.4% in the first quarter of 2020 to 12.9% in the last quarter of 2020 – with no considerable difference between men and women.
Blame it on lockdowns?
Dr. Kim and colleagues say research is needed to better understand the direct and indirect influence of COVID-19 on coexisting chronic liver disease and diabetes.
“It is plausible that psychosocial stress and a higher predisposition to psychiatric disorders during the COVID-19 pandemic can increase the risk of alcohol use disorder and ALD,” they write.
“Furthermore, it is prudent to suspect that COVID-19–related lockdowns may increase the risk of obesity, leading to a higher risk of insulin resistance and metabolic complications, including diabetes and NAFLD. Future studies are needed to improve our understanding of these possible pathogenetic links. More importantly, emergency preparedness or contingency plans must be in place to continue and provide uninterrupted care for chronic ailments during times of disaster,” they add.
The study had no specific funding. The authors report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The COVID-19 pandemic fueled a sharp uptick in deaths related to chronic liver disease and cirrhosis among people with diabetes, largely owing to nonalcoholic fatty liver disease (NAFLD) and alcohol-related liver disease (ALD), new data show.
“Our observations confirm that COVID-19 had a higher likelihood of impacting vulnerable populations with pre-existing chronic liver diseases and diabetes, with a death rate as high as 10% in individuals with co-existing chronic liver disease and diabetes,” write the authors.
“The inability to attend regular outpatient clinics for close monitoring and treatment accompanied by diversion of health care resources to COVID-19 care may have resulted in the suboptimal or delayed clinical care of individuals with diabetes and chronic liver disease during the COVID-19 pandemic,” they add.
Donghee Kim, MD, PhD, with the Division of Gastroenterology and Hepatology, Stanford (Calif.) University School of Medicine, and colleagues report their findings in the journal Digestive and Liver Disease.
Vulnerable group
The researchers used U.S. national mortality data (2017-2020) to estimate chronic liver disease–related mortality trends among individuals with diabetes before and during the COVID-19 pandemic.
Before the pandemic, the quarterly mortality for chronic liver disease remained stable (quarterly percentage change, 0.6%) but then sharply increased during the pandemic (QPC, 8.6%).
A similar trend was seen with cirrhosis-related mortality (QPC, 0.3% before the pandemic vs. 8.4% during the pandemic).
NAFLD and ALD mortality among individuals with diabetes was steadily increasing before the pandemic (QPC, 4.2% and 3.5%, respectively) but showed a more rapid increase during the pandemic (QPC, 9.6% and 7.7%, respectively).
ALD-related mortality in men was more than threefold higher than in women, while NAFLD-related mortality in women was more than twofold higher than in men.
Mortality for hepatitis C virus infection declined before the pandemic (QPC, −3.3%) and remained stable during the pandemic.
COVID-19–related mortality among adults with chronic liver disease and diabetes also rose sharply during the pandemic – from 0.4% in the first quarter of 2020 to 12.9% in the last quarter of 2020 – with no considerable difference between men and women.
Blame it on lockdowns?
Dr. Kim and colleagues say research is needed to better understand the direct and indirect influence of COVID-19 on coexisting chronic liver disease and diabetes.
“It is plausible that psychosocial stress and a higher predisposition to psychiatric disorders during the COVID-19 pandemic can increase the risk of alcohol use disorder and ALD,” they write.
“Furthermore, it is prudent to suspect that COVID-19–related lockdowns may increase the risk of obesity, leading to a higher risk of insulin resistance and metabolic complications, including diabetes and NAFLD. Future studies are needed to improve our understanding of these possible pathogenetic links. More importantly, emergency preparedness or contingency plans must be in place to continue and provide uninterrupted care for chronic ailments during times of disaster,” they add.
The study had no specific funding. The authors report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The COVID-19 pandemic fueled a sharp uptick in deaths related to chronic liver disease and cirrhosis among people with diabetes, largely owing to nonalcoholic fatty liver disease (NAFLD) and alcohol-related liver disease (ALD), new data show.
“Our observations confirm that COVID-19 had a higher likelihood of impacting vulnerable populations with pre-existing chronic liver diseases and diabetes, with a death rate as high as 10% in individuals with co-existing chronic liver disease and diabetes,” write the authors.
“The inability to attend regular outpatient clinics for close monitoring and treatment accompanied by diversion of health care resources to COVID-19 care may have resulted in the suboptimal or delayed clinical care of individuals with diabetes and chronic liver disease during the COVID-19 pandemic,” they add.
Donghee Kim, MD, PhD, with the Division of Gastroenterology and Hepatology, Stanford (Calif.) University School of Medicine, and colleagues report their findings in the journal Digestive and Liver Disease.
Vulnerable group
The researchers used U.S. national mortality data (2017-2020) to estimate chronic liver disease–related mortality trends among individuals with diabetes before and during the COVID-19 pandemic.
Before the pandemic, the quarterly mortality for chronic liver disease remained stable (quarterly percentage change, 0.6%) but then sharply increased during the pandemic (QPC, 8.6%).
A similar trend was seen with cirrhosis-related mortality (QPC, 0.3% before the pandemic vs. 8.4% during the pandemic).
NAFLD and ALD mortality among individuals with diabetes was steadily increasing before the pandemic (QPC, 4.2% and 3.5%, respectively) but showed a more rapid increase during the pandemic (QPC, 9.6% and 7.7%, respectively).
ALD-related mortality in men was more than threefold higher than in women, while NAFLD-related mortality in women was more than twofold higher than in men.
Mortality for hepatitis C virus infection declined before the pandemic (QPC, −3.3%) and remained stable during the pandemic.
COVID-19–related mortality among adults with chronic liver disease and diabetes also rose sharply during the pandemic – from 0.4% in the first quarter of 2020 to 12.9% in the last quarter of 2020 – with no considerable difference between men and women.
Blame it on lockdowns?
Dr. Kim and colleagues say research is needed to better understand the direct and indirect influence of COVID-19 on coexisting chronic liver disease and diabetes.
“It is plausible that psychosocial stress and a higher predisposition to psychiatric disorders during the COVID-19 pandemic can increase the risk of alcohol use disorder and ALD,” they write.
“Furthermore, it is prudent to suspect that COVID-19–related lockdowns may increase the risk of obesity, leading to a higher risk of insulin resistance and metabolic complications, including diabetes and NAFLD. Future studies are needed to improve our understanding of these possible pathogenetic links. More importantly, emergency preparedness or contingency plans must be in place to continue and provide uninterrupted care for chronic ailments during times of disaster,” they add.
The study had no specific funding. The authors report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Sexual issues common for GI patients, but docs often avoid topic
VIENNA – Sexual dysfunction in patients with gastrointestinal disorders is undermanaged, with a lack of clinician education, time constraints, and embarrassment preventing constructive discussions to improve patient care and quality of life, according to a new survey.
Overall, 71% of gastroenterologists do not ask their patients about sexual dysfunction, the survey finds.
“While patients with gastrointestinal disorders often experience sexual dysfunction, discussions around the matter are not routine in gastroenterological care,” said Marco Romano, MD, from the University of Campania “Luigi Vanvitelli,” Naples, Italy.
Romano presented the survey findings at this year’s United European Gastroenterology Week meeting.
The research shows not only a clear need for better awareness but also a need to build gastroenterologists’ confidence in addressing sexual dysfunction with their patients, Dr. Romano added.
“Most felt that sexual medicine education and improvement of communication skills within the context of their residency training might be important in order to increase the awareness of sexual dysfunction, to overcome barriers, and to improve care and quality of life for their patients,” reported Dr. Romano. “This will lead to prompt diagnosis and treatment of any sexual problems.”
Respectfully asking the patients if their gastrointestinal disorders interfere with their intimate relationships “is often considered a relief to patients who find that the gastrointestinal problem and the sexual dysfunction are interlinked,” he added.
The findings
The survey was needed because the question of whether gastroenterologists inquire about their patients’ sexual issues had never been assessed, Dr. Romano said.
The researchers sent a cross-sectional, anonymous online survey to members of the Italian Society of Gastroenterology and Digestive Endoscopy. The questionnaire, designed and informed by a literature review, consisted of 29 single multiple-choice and open-ended questions.
A total of 426 surveys were returned: 335 from experienced gastroenterologists and 91 from residents (less experienced). Of all respondents, 54.7% were men and 45.3% were women.
Even though most gastroenterologists do not ask their patients about sexual dysfunction, the majority want to learn how to manage the issue, the survey found. Of the survey respondents, 80% agreed that it would be useful for gastroenterologists to attend courses dedicated to the problem of sexual dysfunction.
Only 4% of patients report (initiate a dialogue about) the problem, the survey found. Among women aged 40-50 years, the most common complaint reported was dyspareunia (pain on intercourse). In men, the most frequent complaints reported were in the over-40s age group, with 75% citing erectile dysfunction and 45% reporting loss of libido.
The most common gastrointestinal disorders associated with sexual dysfunction are inflammatory bowel diseases (37% of cases), chronic liver diseases (28%), and irritable bowel syndrome (26%), according to the survey.
On the question of whether medications played a role in patients’ sexual dysfunction, nearly 15% of respondents said that prokinetic agents were involved, and 18% thought proton pump inhibitors affect sexual function. Both drug classes are considered responsible for sexual disturbances.
Few gastroenterologists prescribe phosphodiesterase type 5 inhibitors (PDE5i), e.g., Viagra, to treat sexual dysfunction, the survey found. Approximately 90% of respondents said that they never prescribed this class of drugs, preferring to refer patients to an andrologist. Of those who did prescribe PDE5i, significantly fewer residents did compared with experienced gastroenterologists (1.1% vs. 8.8%, respectively; P = .01).
Finally, the biggest reasons why gastroenterologists do not discuss sexual dysfunction are lack of knowledge (80%), insufficient experience (58%), time (44%), and embarrassment (30%).
Practice experience matters
There were some differences among respondents in the experienced group vs. the residents. More men were in the experienced group compared with residents (57.6% vs. 44%, respectively); mean age was 47 years vs. 29 years, respectively; and 71% had 5 or more years of experience in the experienced gastroenterologist group, whereas 78% had 1-5 years of experience among residents.
The survey found that more residents than experienced gastroenterologists “never discussed sexual dysfunction” (38.5% vs. 21.3%, respectively; P = .001) and that more residents than experienced gastroenterologists reported that “patients did not relate their sexual dysfunction to the prescribed therapy” (47.8% vs. 32.5%, respectively; P = .007).
The two groups varied regarding prescription drugs’ role in sexual dysfunction. More experienced gastroenterologists than residents felt that proton pump inhibitors (5.8% vs. 0%, respectively; P = .018) or prokinetics (19.8% vs. 9.5%, respectively; P = .028) might be responsible for some degree of sexual dysfunction.
More residents than experienced doctors felt that other (nongastroenterologic) drugs might contribute to sexual dysfunction in their patients (57.1% vs. 44.7%, respectively; P = .043).
Dr. Romano reported that fewer residents than experienced gastroenterologists referred male patients with sexual dysfunction to an andrologist (frequently/always: 28.1% vs. 44.4%, respectively; P = .004). However, more residents than experienced gastroenterologists disagreed that discussing sexual dysfunction with patients pertains only to specialists (andrologists and gynecologists; 83.5% vs. 71.2%, respectively; P = .018).
Time to step up
Asma Fikree, BMBCh, PhD, of Royal London Hospital, Barts Health NHS Trust, London, moderated the session. The survey highlights that asking patients about sexual dysfunction is an area for improvement for gastroenterologists, she said.
“We might do it in men and ask about erectile dysfunction, but we are very poor about asking in women,” Dr. Fikree noted.
The pros and cons of different medications should be discussed with patients, she said.
Gastroenterologists need to do a better job of considering how medications can lead to sexual dysfunction and interfere with quality of life, and training would help, she added.
“Some patients might not be very bothered by sexual dysfunction, but others might consider it very important,” Dr. Fikree said. “We should be considering this as part of their treatment and care.”
Dr. Romano and Dr. Fikree report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
VIENNA – Sexual dysfunction in patients with gastrointestinal disorders is undermanaged, with a lack of clinician education, time constraints, and embarrassment preventing constructive discussions to improve patient care and quality of life, according to a new survey.
Overall, 71% of gastroenterologists do not ask their patients about sexual dysfunction, the survey finds.
“While patients with gastrointestinal disorders often experience sexual dysfunction, discussions around the matter are not routine in gastroenterological care,” said Marco Romano, MD, from the University of Campania “Luigi Vanvitelli,” Naples, Italy.
Romano presented the survey findings at this year’s United European Gastroenterology Week meeting.
The research shows not only a clear need for better awareness but also a need to build gastroenterologists’ confidence in addressing sexual dysfunction with their patients, Dr. Romano added.
“Most felt that sexual medicine education and improvement of communication skills within the context of their residency training might be important in order to increase the awareness of sexual dysfunction, to overcome barriers, and to improve care and quality of life for their patients,” reported Dr. Romano. “This will lead to prompt diagnosis and treatment of any sexual problems.”
Respectfully asking the patients if their gastrointestinal disorders interfere with their intimate relationships “is often considered a relief to patients who find that the gastrointestinal problem and the sexual dysfunction are interlinked,” he added.
The findings
The survey was needed because the question of whether gastroenterologists inquire about their patients’ sexual issues had never been assessed, Dr. Romano said.
The researchers sent a cross-sectional, anonymous online survey to members of the Italian Society of Gastroenterology and Digestive Endoscopy. The questionnaire, designed and informed by a literature review, consisted of 29 single multiple-choice and open-ended questions.
A total of 426 surveys were returned: 335 from experienced gastroenterologists and 91 from residents (less experienced). Of all respondents, 54.7% were men and 45.3% were women.
Even though most gastroenterologists do not ask their patients about sexual dysfunction, the majority want to learn how to manage the issue, the survey found. Of the survey respondents, 80% agreed that it would be useful for gastroenterologists to attend courses dedicated to the problem of sexual dysfunction.
Only 4% of patients report (initiate a dialogue about) the problem, the survey found. Among women aged 40-50 years, the most common complaint reported was dyspareunia (pain on intercourse). In men, the most frequent complaints reported were in the over-40s age group, with 75% citing erectile dysfunction and 45% reporting loss of libido.
The most common gastrointestinal disorders associated with sexual dysfunction are inflammatory bowel diseases (37% of cases), chronic liver diseases (28%), and irritable bowel syndrome (26%), according to the survey.
On the question of whether medications played a role in patients’ sexual dysfunction, nearly 15% of respondents said that prokinetic agents were involved, and 18% thought proton pump inhibitors affect sexual function. Both drug classes are considered responsible for sexual disturbances.
Few gastroenterologists prescribe phosphodiesterase type 5 inhibitors (PDE5i), e.g., Viagra, to treat sexual dysfunction, the survey found. Approximately 90% of respondents said that they never prescribed this class of drugs, preferring to refer patients to an andrologist. Of those who did prescribe PDE5i, significantly fewer residents did compared with experienced gastroenterologists (1.1% vs. 8.8%, respectively; P = .01).
Finally, the biggest reasons why gastroenterologists do not discuss sexual dysfunction are lack of knowledge (80%), insufficient experience (58%), time (44%), and embarrassment (30%).
Practice experience matters
There were some differences among respondents in the experienced group vs. the residents. More men were in the experienced group compared with residents (57.6% vs. 44%, respectively); mean age was 47 years vs. 29 years, respectively; and 71% had 5 or more years of experience in the experienced gastroenterologist group, whereas 78% had 1-5 years of experience among residents.
The survey found that more residents than experienced gastroenterologists “never discussed sexual dysfunction” (38.5% vs. 21.3%, respectively; P = .001) and that more residents than experienced gastroenterologists reported that “patients did not relate their sexual dysfunction to the prescribed therapy” (47.8% vs. 32.5%, respectively; P = .007).
The two groups varied regarding prescription drugs’ role in sexual dysfunction. More experienced gastroenterologists than residents felt that proton pump inhibitors (5.8% vs. 0%, respectively; P = .018) or prokinetics (19.8% vs. 9.5%, respectively; P = .028) might be responsible for some degree of sexual dysfunction.
More residents than experienced doctors felt that other (nongastroenterologic) drugs might contribute to sexual dysfunction in their patients (57.1% vs. 44.7%, respectively; P = .043).
Dr. Romano reported that fewer residents than experienced gastroenterologists referred male patients with sexual dysfunction to an andrologist (frequently/always: 28.1% vs. 44.4%, respectively; P = .004). However, more residents than experienced gastroenterologists disagreed that discussing sexual dysfunction with patients pertains only to specialists (andrologists and gynecologists; 83.5% vs. 71.2%, respectively; P = .018).
Time to step up
Asma Fikree, BMBCh, PhD, of Royal London Hospital, Barts Health NHS Trust, London, moderated the session. The survey highlights that asking patients about sexual dysfunction is an area for improvement for gastroenterologists, she said.
“We might do it in men and ask about erectile dysfunction, but we are very poor about asking in women,” Dr. Fikree noted.
The pros and cons of different medications should be discussed with patients, she said.
Gastroenterologists need to do a better job of considering how medications can lead to sexual dysfunction and interfere with quality of life, and training would help, she added.
“Some patients might not be very bothered by sexual dysfunction, but others might consider it very important,” Dr. Fikree said. “We should be considering this as part of their treatment and care.”
Dr. Romano and Dr. Fikree report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
VIENNA – Sexual dysfunction in patients with gastrointestinal disorders is undermanaged, with a lack of clinician education, time constraints, and embarrassment preventing constructive discussions to improve patient care and quality of life, according to a new survey.
Overall, 71% of gastroenterologists do not ask their patients about sexual dysfunction, the survey finds.
“While patients with gastrointestinal disorders often experience sexual dysfunction, discussions around the matter are not routine in gastroenterological care,” said Marco Romano, MD, from the University of Campania “Luigi Vanvitelli,” Naples, Italy.
Romano presented the survey findings at this year’s United European Gastroenterology Week meeting.
The research shows not only a clear need for better awareness but also a need to build gastroenterologists’ confidence in addressing sexual dysfunction with their patients, Dr. Romano added.
“Most felt that sexual medicine education and improvement of communication skills within the context of their residency training might be important in order to increase the awareness of sexual dysfunction, to overcome barriers, and to improve care and quality of life for their patients,” reported Dr. Romano. “This will lead to prompt diagnosis and treatment of any sexual problems.”
Respectfully asking the patients if their gastrointestinal disorders interfere with their intimate relationships “is often considered a relief to patients who find that the gastrointestinal problem and the sexual dysfunction are interlinked,” he added.
The findings
The survey was needed because the question of whether gastroenterologists inquire about their patients’ sexual issues had never been assessed, Dr. Romano said.
The researchers sent a cross-sectional, anonymous online survey to members of the Italian Society of Gastroenterology and Digestive Endoscopy. The questionnaire, designed and informed by a literature review, consisted of 29 single multiple-choice and open-ended questions.
A total of 426 surveys were returned: 335 from experienced gastroenterologists and 91 from residents (less experienced). Of all respondents, 54.7% were men and 45.3% were women.
Even though most gastroenterologists do not ask their patients about sexual dysfunction, the majority want to learn how to manage the issue, the survey found. Of the survey respondents, 80% agreed that it would be useful for gastroenterologists to attend courses dedicated to the problem of sexual dysfunction.
Only 4% of patients report (initiate a dialogue about) the problem, the survey found. Among women aged 40-50 years, the most common complaint reported was dyspareunia (pain on intercourse). In men, the most frequent complaints reported were in the over-40s age group, with 75% citing erectile dysfunction and 45% reporting loss of libido.
The most common gastrointestinal disorders associated with sexual dysfunction are inflammatory bowel diseases (37% of cases), chronic liver diseases (28%), and irritable bowel syndrome (26%), according to the survey.
On the question of whether medications played a role in patients’ sexual dysfunction, nearly 15% of respondents said that prokinetic agents were involved, and 18% thought proton pump inhibitors affect sexual function. Both drug classes are considered responsible for sexual disturbances.
Few gastroenterologists prescribe phosphodiesterase type 5 inhibitors (PDE5i), e.g., Viagra, to treat sexual dysfunction, the survey found. Approximately 90% of respondents said that they never prescribed this class of drugs, preferring to refer patients to an andrologist. Of those who did prescribe PDE5i, significantly fewer residents did compared with experienced gastroenterologists (1.1% vs. 8.8%, respectively; P = .01).
Finally, the biggest reasons why gastroenterologists do not discuss sexual dysfunction are lack of knowledge (80%), insufficient experience (58%), time (44%), and embarrassment (30%).
Practice experience matters
There were some differences among respondents in the experienced group vs. the residents. More men were in the experienced group compared with residents (57.6% vs. 44%, respectively); mean age was 47 years vs. 29 years, respectively; and 71% had 5 or more years of experience in the experienced gastroenterologist group, whereas 78% had 1-5 years of experience among residents.
The survey found that more residents than experienced gastroenterologists “never discussed sexual dysfunction” (38.5% vs. 21.3%, respectively; P = .001) and that more residents than experienced gastroenterologists reported that “patients did not relate their sexual dysfunction to the prescribed therapy” (47.8% vs. 32.5%, respectively; P = .007).
The two groups varied regarding prescription drugs’ role in sexual dysfunction. More experienced gastroenterologists than residents felt that proton pump inhibitors (5.8% vs. 0%, respectively; P = .018) or prokinetics (19.8% vs. 9.5%, respectively; P = .028) might be responsible for some degree of sexual dysfunction.
More residents than experienced doctors felt that other (nongastroenterologic) drugs might contribute to sexual dysfunction in their patients (57.1% vs. 44.7%, respectively; P = .043).
Dr. Romano reported that fewer residents than experienced gastroenterologists referred male patients with sexual dysfunction to an andrologist (frequently/always: 28.1% vs. 44.4%, respectively; P = .004). However, more residents than experienced gastroenterologists disagreed that discussing sexual dysfunction with patients pertains only to specialists (andrologists and gynecologists; 83.5% vs. 71.2%, respectively; P = .018).
Time to step up
Asma Fikree, BMBCh, PhD, of Royal London Hospital, Barts Health NHS Trust, London, moderated the session. The survey highlights that asking patients about sexual dysfunction is an area for improvement for gastroenterologists, she said.
“We might do it in men and ask about erectile dysfunction, but we are very poor about asking in women,” Dr. Fikree noted.
The pros and cons of different medications should be discussed with patients, she said.
Gastroenterologists need to do a better job of considering how medications can lead to sexual dysfunction and interfere with quality of life, and training would help, she added.
“Some patients might not be very bothered by sexual dysfunction, but others might consider it very important,” Dr. Fikree said. “We should be considering this as part of their treatment and care.”
Dr. Romano and Dr. Fikree report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
AT UEG WEEK 2022
Preexisting mental illness symptoms spiked during pandemic
“Those with preexisting mental health conditions may be particularly vulnerable to these effects because they are more susceptible to experiencing high levels of stress during a crisis and are more likely to experience isolation/despair during confinement compared to the general population,” wrote Danna Ramirez of The Menninger Clinic, Houston, and colleagues.
In a study published in Psychiatry Research , the investigators compared data from 142 adolescents aged 12-17 years and 470 adults aged 18-79 years who were admitted to an inpatient psychiatric hospital in Houston. Of these, 65 adolescents and 235 adults were admitted before the pandemic, and 77 adolescents and 235 adults were admitted during the pandemic.
Clinical outcomes were scores on the Generalized Anxiety Disorder Scale (GAD-7), the Patient Health Questionnaire (PHQ-9), the Patient Health Questionnaire for Adolescents (PHQ-A), the Difficulties in Emotion Regulation Scale–Short Form (DERS-SF), the World Health Organization Disability Assessment Scale (WHODAS), the World Health Organization Alcohol, Smoking, and Substance Involvement Screening Test (WHOASSIST), the Pittsburgh Sleep Quality Index (PSQI), the Disturbing Dream and Nightmare Severity Index (DDNSI), and the Suicide Behaviors Questionnaire–Revised (SBQ-R).
Overall, adults admitted during the pandemic had significantly higher levels of anxiety, depression, emotional dysregulation, and disability (P < .001 for all) as well as nightmares (P = .013) compared to those admitted prior to the pandemic.
Among adolescents, measures of anxiety, depression, and sleep quality were significantly higher at admission during the pandemic compared to prior to the pandemic (P = .005, P = .005, and P = .011, respectively)
Reasons for the increase in symptom severity remain unclear, but include the possibility that individuals with preexisting mental illness simply became more ill; or that individuals with symptoms delayed hospital admission out of fear of exposure to COVID-19, which resulted in more severe symptoms at admission, the researchers wrote in their discussion.
The findings were limited by several factors, including the primarily White population and the reliance on self-reports, the researchers noted. Another limitation was the lack of differentiation between patients who may have had COVID-19 before hospitalization and those who did not, so the researchers could not determine whether the virus itself played a biological role in symptom severity.
However, the results support data from previous studies and identify increased psychiatry symptom severity for patients admitted for inpatient psychiatry care during the pandemic, they said. Although resources are scarce, the findings emphasize that mental health needs, especially for those with preexisting conditions, should not be overlooked, and continuity and expansion of access to mental health care for all should be prioritized, they concluded.
The study was supported by The Menninger Clinic and The Menninger Clinic Foundation. The researchers had no financial conflicts to disclose.
“Those with preexisting mental health conditions may be particularly vulnerable to these effects because they are more susceptible to experiencing high levels of stress during a crisis and are more likely to experience isolation/despair during confinement compared to the general population,” wrote Danna Ramirez of The Menninger Clinic, Houston, and colleagues.
In a study published in Psychiatry Research , the investigators compared data from 142 adolescents aged 12-17 years and 470 adults aged 18-79 years who were admitted to an inpatient psychiatric hospital in Houston. Of these, 65 adolescents and 235 adults were admitted before the pandemic, and 77 adolescents and 235 adults were admitted during the pandemic.
Clinical outcomes were scores on the Generalized Anxiety Disorder Scale (GAD-7), the Patient Health Questionnaire (PHQ-9), the Patient Health Questionnaire for Adolescents (PHQ-A), the Difficulties in Emotion Regulation Scale–Short Form (DERS-SF), the World Health Organization Disability Assessment Scale (WHODAS), the World Health Organization Alcohol, Smoking, and Substance Involvement Screening Test (WHOASSIST), the Pittsburgh Sleep Quality Index (PSQI), the Disturbing Dream and Nightmare Severity Index (DDNSI), and the Suicide Behaviors Questionnaire–Revised (SBQ-R).
Overall, adults admitted during the pandemic had significantly higher levels of anxiety, depression, emotional dysregulation, and disability (P < .001 for all) as well as nightmares (P = .013) compared to those admitted prior to the pandemic.
Among adolescents, measures of anxiety, depression, and sleep quality were significantly higher at admission during the pandemic compared to prior to the pandemic (P = .005, P = .005, and P = .011, respectively)
Reasons for the increase in symptom severity remain unclear, but include the possibility that individuals with preexisting mental illness simply became more ill; or that individuals with symptoms delayed hospital admission out of fear of exposure to COVID-19, which resulted in more severe symptoms at admission, the researchers wrote in their discussion.
The findings were limited by several factors, including the primarily White population and the reliance on self-reports, the researchers noted. Another limitation was the lack of differentiation between patients who may have had COVID-19 before hospitalization and those who did not, so the researchers could not determine whether the virus itself played a biological role in symptom severity.
However, the results support data from previous studies and identify increased psychiatry symptom severity for patients admitted for inpatient psychiatry care during the pandemic, they said. Although resources are scarce, the findings emphasize that mental health needs, especially for those with preexisting conditions, should not be overlooked, and continuity and expansion of access to mental health care for all should be prioritized, they concluded.
The study was supported by The Menninger Clinic and The Menninger Clinic Foundation. The researchers had no financial conflicts to disclose.
“Those with preexisting mental health conditions may be particularly vulnerable to these effects because they are more susceptible to experiencing high levels of stress during a crisis and are more likely to experience isolation/despair during confinement compared to the general population,” wrote Danna Ramirez of The Menninger Clinic, Houston, and colleagues.
In a study published in Psychiatry Research , the investigators compared data from 142 adolescents aged 12-17 years and 470 adults aged 18-79 years who were admitted to an inpatient psychiatric hospital in Houston. Of these, 65 adolescents and 235 adults were admitted before the pandemic, and 77 adolescents and 235 adults were admitted during the pandemic.
Clinical outcomes were scores on the Generalized Anxiety Disorder Scale (GAD-7), the Patient Health Questionnaire (PHQ-9), the Patient Health Questionnaire for Adolescents (PHQ-A), the Difficulties in Emotion Regulation Scale–Short Form (DERS-SF), the World Health Organization Disability Assessment Scale (WHODAS), the World Health Organization Alcohol, Smoking, and Substance Involvement Screening Test (WHOASSIST), the Pittsburgh Sleep Quality Index (PSQI), the Disturbing Dream and Nightmare Severity Index (DDNSI), and the Suicide Behaviors Questionnaire–Revised (SBQ-R).
Overall, adults admitted during the pandemic had significantly higher levels of anxiety, depression, emotional dysregulation, and disability (P < .001 for all) as well as nightmares (P = .013) compared to those admitted prior to the pandemic.
Among adolescents, measures of anxiety, depression, and sleep quality were significantly higher at admission during the pandemic compared to prior to the pandemic (P = .005, P = .005, and P = .011, respectively)
Reasons for the increase in symptom severity remain unclear, but include the possibility that individuals with preexisting mental illness simply became more ill; or that individuals with symptoms delayed hospital admission out of fear of exposure to COVID-19, which resulted in more severe symptoms at admission, the researchers wrote in their discussion.
The findings were limited by several factors, including the primarily White population and the reliance on self-reports, the researchers noted. Another limitation was the lack of differentiation between patients who may have had COVID-19 before hospitalization and those who did not, so the researchers could not determine whether the virus itself played a biological role in symptom severity.
However, the results support data from previous studies and identify increased psychiatry symptom severity for patients admitted for inpatient psychiatry care during the pandemic, they said. Although resources are scarce, the findings emphasize that mental health needs, especially for those with preexisting conditions, should not be overlooked, and continuity and expansion of access to mental health care for all should be prioritized, they concluded.
The study was supported by The Menninger Clinic and The Menninger Clinic Foundation. The researchers had no financial conflicts to disclose.
FROM PSYCHIATRY RESEARCH
People of color more likely to be hospitalized for influenza, CDC report finds
Black Americans are 80% more likely to be hospitalized for the flu, compared with White Americans, according to new federal data.
Black, Hispanic, and American Indian/Alaska Native (AI/AN) adults in the United States also have had lower influenza vaccination rates, compared with their White counterparts, since 2010, researchers at the Centers for Disease Control and Prevention (CDC) revealed in a report.
The inequalities are the result of barriers to care, distrust of the medical system, and misinformation, the report said.
“We have many of the tools we need to address inequities and flu vaccination coverage and outcomes,” said CDC Acting Principal Deputy Director Debra Houry, MD, MPH, in a press call; “however, we must acknowledge that inequities in access to care continue to exist. To improve vaccine uptake, we must address the root causes of these ongoing disparities.”
The CDC has already reported early increases in flu activity in the United States, with the highest activity in the southeastern and south-central parts of the country. Experts also warn of a potentially more severe influenza season than in the previous 2 years. CDC officials emphasized that vaccination is the best protection against severe illness, hospitalization, and death from the flu. “Everyone should get vaccinated against flu today and encourage others and their community to get a flu vaccine for the best protection against flu this fall and winter,” Dr. Houry said.
In the recent report on disparities by community published October 18 in CDC Vital Signs, researchers looked at hospitalization rates from 2009 to 2022 and vaccination rates from 2010 to 2022 based on race and ethnicity using two national databases, the Influenza-Associated Hospitalization Surveillance Network and the Behavioral Risk Factor Surveillance System. All individuals included in the analysis were aged 18 years or older, and the 2020-2021 flu season was excluded from the analysis because of insufficient data.
Compared with those for White adults, hospitalization rates were 80% higher for Black adults, 30% higher for Hispanic adults, and 20% higher for AI/AN adults. While flu vaccination rates were similar in White and Asian adults (about 54%), coverage was lower in Black (42%), Hispanic (38%), AI/AN (41%), and other/multiracial (43%) adults. This disparity persisted even among individuals who had medical insurance, a personal health care provider, and a routine checkup within the last year.
Carla Black, PhD, MPH, an epidemiologist at the CDC’s Immunization Services Division, said during the press call. While flu vaccines may not always prevent infection, people who do get sick after being vaccinated tend to have better outcomes, she added. The report noted that building trust, increasing access to vaccination services, and combating misinformation are important steps to increasing vaccine coverage in minority groups.
While social distancing measures such as masking have made it difficult for the flu to spread, the relaxation of these safety measures could also lead to higher case counts. “We’ve had two mild flu seasons, and this means we might be ripe for a severe season,” Dr. Black said. “People haven’t had natural disease in 2 years, so there’s less natural immunity out there. People are going back to work. People are traveling again. All of these factors could contribute to us having a more severe flu season.”
A version of this article first appeared on Medscape.com.
Black Americans are 80% more likely to be hospitalized for the flu, compared with White Americans, according to new federal data.
Black, Hispanic, and American Indian/Alaska Native (AI/AN) adults in the United States also have had lower influenza vaccination rates, compared with their White counterparts, since 2010, researchers at the Centers for Disease Control and Prevention (CDC) revealed in a report.
The inequalities are the result of barriers to care, distrust of the medical system, and misinformation, the report said.
“We have many of the tools we need to address inequities and flu vaccination coverage and outcomes,” said CDC Acting Principal Deputy Director Debra Houry, MD, MPH, in a press call; “however, we must acknowledge that inequities in access to care continue to exist. To improve vaccine uptake, we must address the root causes of these ongoing disparities.”
The CDC has already reported early increases in flu activity in the United States, with the highest activity in the southeastern and south-central parts of the country. Experts also warn of a potentially more severe influenza season than in the previous 2 years. CDC officials emphasized that vaccination is the best protection against severe illness, hospitalization, and death from the flu. “Everyone should get vaccinated against flu today and encourage others and their community to get a flu vaccine for the best protection against flu this fall and winter,” Dr. Houry said.
In the recent report on disparities by community published October 18 in CDC Vital Signs, researchers looked at hospitalization rates from 2009 to 2022 and vaccination rates from 2010 to 2022 based on race and ethnicity using two national databases, the Influenza-Associated Hospitalization Surveillance Network and the Behavioral Risk Factor Surveillance System. All individuals included in the analysis were aged 18 years or older, and the 2020-2021 flu season was excluded from the analysis because of insufficient data.
Compared with those for White adults, hospitalization rates were 80% higher for Black adults, 30% higher for Hispanic adults, and 20% higher for AI/AN adults. While flu vaccination rates were similar in White and Asian adults (about 54%), coverage was lower in Black (42%), Hispanic (38%), AI/AN (41%), and other/multiracial (43%) adults. This disparity persisted even among individuals who had medical insurance, a personal health care provider, and a routine checkup within the last year.
Carla Black, PhD, MPH, an epidemiologist at the CDC’s Immunization Services Division, said during the press call. While flu vaccines may not always prevent infection, people who do get sick after being vaccinated tend to have better outcomes, she added. The report noted that building trust, increasing access to vaccination services, and combating misinformation are important steps to increasing vaccine coverage in minority groups.
While social distancing measures such as masking have made it difficult for the flu to spread, the relaxation of these safety measures could also lead to higher case counts. “We’ve had two mild flu seasons, and this means we might be ripe for a severe season,” Dr. Black said. “People haven’t had natural disease in 2 years, so there’s less natural immunity out there. People are going back to work. People are traveling again. All of these factors could contribute to us having a more severe flu season.”
A version of this article first appeared on Medscape.com.
Black Americans are 80% more likely to be hospitalized for the flu, compared with White Americans, according to new federal data.
Black, Hispanic, and American Indian/Alaska Native (AI/AN) adults in the United States also have had lower influenza vaccination rates, compared with their White counterparts, since 2010, researchers at the Centers for Disease Control and Prevention (CDC) revealed in a report.
The inequalities are the result of barriers to care, distrust of the medical system, and misinformation, the report said.
“We have many of the tools we need to address inequities and flu vaccination coverage and outcomes,” said CDC Acting Principal Deputy Director Debra Houry, MD, MPH, in a press call; “however, we must acknowledge that inequities in access to care continue to exist. To improve vaccine uptake, we must address the root causes of these ongoing disparities.”
The CDC has already reported early increases in flu activity in the United States, with the highest activity in the southeastern and south-central parts of the country. Experts also warn of a potentially more severe influenza season than in the previous 2 years. CDC officials emphasized that vaccination is the best protection against severe illness, hospitalization, and death from the flu. “Everyone should get vaccinated against flu today and encourage others and their community to get a flu vaccine for the best protection against flu this fall and winter,” Dr. Houry said.
In the recent report on disparities by community published October 18 in CDC Vital Signs, researchers looked at hospitalization rates from 2009 to 2022 and vaccination rates from 2010 to 2022 based on race and ethnicity using two national databases, the Influenza-Associated Hospitalization Surveillance Network and the Behavioral Risk Factor Surveillance System. All individuals included in the analysis were aged 18 years or older, and the 2020-2021 flu season was excluded from the analysis because of insufficient data.
Compared with those for White adults, hospitalization rates were 80% higher for Black adults, 30% higher for Hispanic adults, and 20% higher for AI/AN adults. While flu vaccination rates were similar in White and Asian adults (about 54%), coverage was lower in Black (42%), Hispanic (38%), AI/AN (41%), and other/multiracial (43%) adults. This disparity persisted even among individuals who had medical insurance, a personal health care provider, and a routine checkup within the last year.
Carla Black, PhD, MPH, an epidemiologist at the CDC’s Immunization Services Division, said during the press call. While flu vaccines may not always prevent infection, people who do get sick after being vaccinated tend to have better outcomes, she added. The report noted that building trust, increasing access to vaccination services, and combating misinformation are important steps to increasing vaccine coverage in minority groups.
While social distancing measures such as masking have made it difficult for the flu to spread, the relaxation of these safety measures could also lead to higher case counts. “We’ve had two mild flu seasons, and this means we might be ripe for a severe season,” Dr. Black said. “People haven’t had natural disease in 2 years, so there’s less natural immunity out there. People are going back to work. People are traveling again. All of these factors could contribute to us having a more severe flu season.”
A version of this article first appeared on Medscape.com.
Nonhormonal drug fezolinetant found safe for hot flashes in yearlong study
The drug fezolinetant, a selective neurokinin-3 receptor antagonist under investigation for treatment of menopausal vasomotor symptoms, showed acceptable long-term safety and tolerability during a 1-year phase 3 randomized controlled trial, according to data presented at the annual meeting of the North American Menopause Society. The study, called SKYLIGHT 4, examined fezolinetant treatment, especially in terms of endometrial health.
The findings mean that fezolinetant “may help bridge a gap in the management of vasomotor symptoms,” according to lead author Genevieve Neal-Perry, MD, PhD, chair of the department of obstetrics and gynecology at the University of North Carolina at Chapel Hill.
This study was an important step in fezolinetant’s path toward potential approval by the Food and Drug Administration for vasomotor symptoms.
”Moderate and severe vasomotor symptoms can adversely affect quality of life of those affected and result in sleep disruption as well as increased risk for heart disease and other high-risk medical problems,” Dr. Neal-Perry said. “Although menopausal hormone therapy significantly improves vasomotor symptoms, it may not be desired or it may not be safe for some women,” resulting in gaps in care and a need for targeted, nonhormonal therapies for hot flashes. A planned study will also assess the safety of the drug in patients with a diagnosis of hormone-sensitive cancer and disorders that increase the risk for blood clots.
”Fezolinetant has a low side effect profile, it is a nonhormonal option, and it is selective for the neurons that trigger and mediate hot flashes,” Dr. Neal-Perry said.
Hot flashes are caused by kisspeptin, neurokinin B, and dynorphin neurons located in the hypothalamus. Fezolinetant works by selectively blocking the neurokinin 3 receptor (NK3R), which regulates a person’s sense of temperature, Dr. Neal-Perry explained. Overactivation of NK3R, resulting from low estrogen levels, plays a role in the hot flashes and cold sweats women experience during menopause.
Drug development for hot flashes ”has been hampered by a lack of knowledge regarding the biological cause,” Dr. Neal-Perry said. “Now that we have a robust understanding of the basic biology of hot flashes, we can develop novel, highly effective, and targeted therapy.”
This safety study involved 1,830 women, ages 40-65, who were experiencing menopausal vasomotor symptoms and were randomly assigned to one of three arms for 52 weeks: 45 mg of fezolinetant, 30 mg of fezolinetant, or a placebo once daily.
The primary endpoints included the percentage of women with endometrial hyperplasia, the percentage of women with endometrial cancer, and the frequency and severity of treatment-emergent adverse events (TEAEs). To meet the primary safety endpoint, no more than 1% of participants could have hyperplasia or malignancy, with an upper confidence interval boundary not greater than 4%. Women who met prespecified criteria for their endometrial health to be assessed, underwent endometrial biopsies at baseline and at the end of the study. Three independent pathologists analyzed the tissue without knowledge of which study arm each sample came from. Among the 599 endometrial biopsy samples, 0.5% of the 203 participants taking 45 mg fezolinetant had hyperplasia while none of the women in the other two arms did. Among the 210 women taking 30 mg of fezolinetant, 0.5% had a malignancy; no malignancies occurred in the other two arms.
Overall adverse events were similar across all three arms, including rates of adverse events leading to discontinuation. The most common adverse events were headache and COVID-19. TEAEs related to the drug were 18.1% in the 45-mg arm, 15.4% in the 30-mg arm, and 17.4% in the placebo arm. Serious adverse events were similar across all three arms, and only 0.5% of participants in the 45-mg arm experienced drug-related serious adverse events, compared with none of the women in the 30-mg arm and 0.2% of women in the placebo group.
”The frequency of transaminase elevations was low, and these TEAEs were generally isolated, transient, and resolved on treatment or with discontinuation,” the authors reported.
The next steps for fezolinetant will be to assess its effect on mood and quality of life measures related to vasomotor symptoms, Dr. Neal-Perry said.
Samantha Dunham, MD, a NAMS-certified menopause practitioner and an associate professor of obstetrics and gynecology at New York University, suggested the drug’s safety in the study is encouraging.
”As a medication that treats menopausal symptoms, the study confirmed there are no issues with the endometrium, or lining of the uterus, not that one would expect issues given the mechanism of action,” Dr. Dunham, also codirector of NYU Langone’s Center for Midlife Health and Menopause, said in an interview. Dr. Dunham was not involved in the study.
”Earlier versions of medication in this class have caused liver enzyme elevation.” The trial of this medication showed that there were only transient elevations in liver enzymes, which resolved upon cessation of the medication. Dr. Dunham said. ”If the medicine proves to be safe over long periods of time in different populations, this will be a very significant medication for treating menopausal vasomotor symptoms.”
The research was funded by Astellas Pharma. Dr. Dunham had no disclosures. Dr. Neal-Perry is a scientific advisory board member for Astellas and Ferring Pharmaceuticals, and has received research funding from Merck and Overa.
The drug fezolinetant, a selective neurokinin-3 receptor antagonist under investigation for treatment of menopausal vasomotor symptoms, showed acceptable long-term safety and tolerability during a 1-year phase 3 randomized controlled trial, according to data presented at the annual meeting of the North American Menopause Society. The study, called SKYLIGHT 4, examined fezolinetant treatment, especially in terms of endometrial health.
The findings mean that fezolinetant “may help bridge a gap in the management of vasomotor symptoms,” according to lead author Genevieve Neal-Perry, MD, PhD, chair of the department of obstetrics and gynecology at the University of North Carolina at Chapel Hill.
This study was an important step in fezolinetant’s path toward potential approval by the Food and Drug Administration for vasomotor symptoms.
”Moderate and severe vasomotor symptoms can adversely affect quality of life of those affected and result in sleep disruption as well as increased risk for heart disease and other high-risk medical problems,” Dr. Neal-Perry said. “Although menopausal hormone therapy significantly improves vasomotor symptoms, it may not be desired or it may not be safe for some women,” resulting in gaps in care and a need for targeted, nonhormonal therapies for hot flashes. A planned study will also assess the safety of the drug in patients with a diagnosis of hormone-sensitive cancer and disorders that increase the risk for blood clots.
”Fezolinetant has a low side effect profile, it is a nonhormonal option, and it is selective for the neurons that trigger and mediate hot flashes,” Dr. Neal-Perry said.
Hot flashes are caused by kisspeptin, neurokinin B, and dynorphin neurons located in the hypothalamus. Fezolinetant works by selectively blocking the neurokinin 3 receptor (NK3R), which regulates a person’s sense of temperature, Dr. Neal-Perry explained. Overactivation of NK3R, resulting from low estrogen levels, plays a role in the hot flashes and cold sweats women experience during menopause.
Drug development for hot flashes ”has been hampered by a lack of knowledge regarding the biological cause,” Dr. Neal-Perry said. “Now that we have a robust understanding of the basic biology of hot flashes, we can develop novel, highly effective, and targeted therapy.”
This safety study involved 1,830 women, ages 40-65, who were experiencing menopausal vasomotor symptoms and were randomly assigned to one of three arms for 52 weeks: 45 mg of fezolinetant, 30 mg of fezolinetant, or a placebo once daily.
The primary endpoints included the percentage of women with endometrial hyperplasia, the percentage of women with endometrial cancer, and the frequency and severity of treatment-emergent adverse events (TEAEs). To meet the primary safety endpoint, no more than 1% of participants could have hyperplasia or malignancy, with an upper confidence interval boundary not greater than 4%. Women who met prespecified criteria for their endometrial health to be assessed, underwent endometrial biopsies at baseline and at the end of the study. Three independent pathologists analyzed the tissue without knowledge of which study arm each sample came from. Among the 599 endometrial biopsy samples, 0.5% of the 203 participants taking 45 mg fezolinetant had hyperplasia while none of the women in the other two arms did. Among the 210 women taking 30 mg of fezolinetant, 0.5% had a malignancy; no malignancies occurred in the other two arms.
Overall adverse events were similar across all three arms, including rates of adverse events leading to discontinuation. The most common adverse events were headache and COVID-19. TEAEs related to the drug were 18.1% in the 45-mg arm, 15.4% in the 30-mg arm, and 17.4% in the placebo arm. Serious adverse events were similar across all three arms, and only 0.5% of participants in the 45-mg arm experienced drug-related serious adverse events, compared with none of the women in the 30-mg arm and 0.2% of women in the placebo group.
”The frequency of transaminase elevations was low, and these TEAEs were generally isolated, transient, and resolved on treatment or with discontinuation,” the authors reported.
The next steps for fezolinetant will be to assess its effect on mood and quality of life measures related to vasomotor symptoms, Dr. Neal-Perry said.
Samantha Dunham, MD, a NAMS-certified menopause practitioner and an associate professor of obstetrics and gynecology at New York University, suggested the drug’s safety in the study is encouraging.
”As a medication that treats menopausal symptoms, the study confirmed there are no issues with the endometrium, or lining of the uterus, not that one would expect issues given the mechanism of action,” Dr. Dunham, also codirector of NYU Langone’s Center for Midlife Health and Menopause, said in an interview. Dr. Dunham was not involved in the study.
”Earlier versions of medication in this class have caused liver enzyme elevation.” The trial of this medication showed that there were only transient elevations in liver enzymes, which resolved upon cessation of the medication. Dr. Dunham said. ”If the medicine proves to be safe over long periods of time in different populations, this will be a very significant medication for treating menopausal vasomotor symptoms.”
The research was funded by Astellas Pharma. Dr. Dunham had no disclosures. Dr. Neal-Perry is a scientific advisory board member for Astellas and Ferring Pharmaceuticals, and has received research funding from Merck and Overa.
The drug fezolinetant, a selective neurokinin-3 receptor antagonist under investigation for treatment of menopausal vasomotor symptoms, showed acceptable long-term safety and tolerability during a 1-year phase 3 randomized controlled trial, according to data presented at the annual meeting of the North American Menopause Society. The study, called SKYLIGHT 4, examined fezolinetant treatment, especially in terms of endometrial health.
The findings mean that fezolinetant “may help bridge a gap in the management of vasomotor symptoms,” according to lead author Genevieve Neal-Perry, MD, PhD, chair of the department of obstetrics and gynecology at the University of North Carolina at Chapel Hill.
This study was an important step in fezolinetant’s path toward potential approval by the Food and Drug Administration for vasomotor symptoms.
”Moderate and severe vasomotor symptoms can adversely affect quality of life of those affected and result in sleep disruption as well as increased risk for heart disease and other high-risk medical problems,” Dr. Neal-Perry said. “Although menopausal hormone therapy significantly improves vasomotor symptoms, it may not be desired or it may not be safe for some women,” resulting in gaps in care and a need for targeted, nonhormonal therapies for hot flashes. A planned study will also assess the safety of the drug in patients with a diagnosis of hormone-sensitive cancer and disorders that increase the risk for blood clots.
”Fezolinetant has a low side effect profile, it is a nonhormonal option, and it is selective for the neurons that trigger and mediate hot flashes,” Dr. Neal-Perry said.
Hot flashes are caused by kisspeptin, neurokinin B, and dynorphin neurons located in the hypothalamus. Fezolinetant works by selectively blocking the neurokinin 3 receptor (NK3R), which regulates a person’s sense of temperature, Dr. Neal-Perry explained. Overactivation of NK3R, resulting from low estrogen levels, plays a role in the hot flashes and cold sweats women experience during menopause.
Drug development for hot flashes ”has been hampered by a lack of knowledge regarding the biological cause,” Dr. Neal-Perry said. “Now that we have a robust understanding of the basic biology of hot flashes, we can develop novel, highly effective, and targeted therapy.”
This safety study involved 1,830 women, ages 40-65, who were experiencing menopausal vasomotor symptoms and were randomly assigned to one of three arms for 52 weeks: 45 mg of fezolinetant, 30 mg of fezolinetant, or a placebo once daily.
The primary endpoints included the percentage of women with endometrial hyperplasia, the percentage of women with endometrial cancer, and the frequency and severity of treatment-emergent adverse events (TEAEs). To meet the primary safety endpoint, no more than 1% of participants could have hyperplasia or malignancy, with an upper confidence interval boundary not greater than 4%. Women who met prespecified criteria for their endometrial health to be assessed, underwent endometrial biopsies at baseline and at the end of the study. Three independent pathologists analyzed the tissue without knowledge of which study arm each sample came from. Among the 599 endometrial biopsy samples, 0.5% of the 203 participants taking 45 mg fezolinetant had hyperplasia while none of the women in the other two arms did. Among the 210 women taking 30 mg of fezolinetant, 0.5% had a malignancy; no malignancies occurred in the other two arms.
Overall adverse events were similar across all three arms, including rates of adverse events leading to discontinuation. The most common adverse events were headache and COVID-19. TEAEs related to the drug were 18.1% in the 45-mg arm, 15.4% in the 30-mg arm, and 17.4% in the placebo arm. Serious adverse events were similar across all three arms, and only 0.5% of participants in the 45-mg arm experienced drug-related serious adverse events, compared with none of the women in the 30-mg arm and 0.2% of women in the placebo group.
”The frequency of transaminase elevations was low, and these TEAEs were generally isolated, transient, and resolved on treatment or with discontinuation,” the authors reported.
The next steps for fezolinetant will be to assess its effect on mood and quality of life measures related to vasomotor symptoms, Dr. Neal-Perry said.
Samantha Dunham, MD, a NAMS-certified menopause practitioner and an associate professor of obstetrics and gynecology at New York University, suggested the drug’s safety in the study is encouraging.
”As a medication that treats menopausal symptoms, the study confirmed there are no issues with the endometrium, or lining of the uterus, not that one would expect issues given the mechanism of action,” Dr. Dunham, also codirector of NYU Langone’s Center for Midlife Health and Menopause, said in an interview. Dr. Dunham was not involved in the study.
”Earlier versions of medication in this class have caused liver enzyme elevation.” The trial of this medication showed that there were only transient elevations in liver enzymes, which resolved upon cessation of the medication. Dr. Dunham said. ”If the medicine proves to be safe over long periods of time in different populations, this will be a very significant medication for treating menopausal vasomotor symptoms.”
The research was funded by Astellas Pharma. Dr. Dunham had no disclosures. Dr. Neal-Perry is a scientific advisory board member for Astellas and Ferring Pharmaceuticals, and has received research funding from Merck and Overa.
FROM NAMS 2022
Islet transplants in type 1 diabetes durable up to 8 years
Transplantation of cadaveric pancreatic islet cells resulted in graft survival and function with acceptable safety for up to 8 years in selected individuals with type 1 diabetes, new research finds.
The study is a long-term follow-up of two phase 3 pivotal trials from the Clinical Islet Transplantation Consortium of a purified human pancreatic islet cell product for treating people with type 1 diabetes.
One trial involved islet transplantation in 48 people who experienced severe hypoglycemia and hypoglycemic unawareness, and the other trial included 24 people who also experienced those complications and were already receiving immunosuppression following kidney transplant. The trials, both registered with the U.S. Food and Drug Administration (FDA), met their primary efficacy and safety endpoints at 2- and 3-year timepoints.
The follow-up data have now been published in Diabetes Care by Michael Rickels, MD, and colleagues.
The procedure involved infusion through the hepatic portal vein of one or more purified human pancreatic islet products under standardized immunosuppression using methods that Dr. Rickels and colleagues have been developing since 2004. The approach involves multiple modalities to protect the islets prior to transplantation.
Among the 34 islet-alone and eight islet-after–kidney transplant recipients who entered the extended follow-up, durable graft survival allowing for achievement of glycemic targets occurred without severe hypoglycemia or adverse effects from immunosuppression.
The primary outcome, actuarial survival of graft islet function, was 56% at the maximum follow-up of 8.3 years for the islet-only transplantation group and 49% at 7.3 years for the islet-after–kidney transplantation group (P = .004).
The findings suggest that “in the long run, islet transplantation has efficacy, including among those who have had kidney transplants ... Most type 1 diabetes patients are improved tremendously with current insulin delivery systems ... but for those having the most difficulty controlling their blood sugar – and those whose diabetes has already been complicated by needing a kidney transplant – the outcomes we saw in this study are what we’ve been hoping to achieve for more than 20 years,” said Dr. Rickels in a statement from his institution, the University of Pennsylvania, Philadelphia.
In the initial trials at day 75 after the initial transplant, 87.5% of the islet-alone and 71% of the islet-after–kidney transplant group achieved hemoglobin A1c under 7%, and 85% and 54%, respectively, achieved A1c at or under 6.5%. At the end of maximal follow-up, 49% of islet-only transplant recipients maintained A1c under 7%, although none had A1c at or under 6.5%. For the islet-after–kidney transplant group, these proportions were 35% and 17%, respectively (P = .0017 for A1c under 7.0% and P < .0001 for A1c ≤ 6.5%, respectively, between the groups).
There were 12 severe hypoglycemic episodes in five patients (three islet-alone and two islet-after–kidney transplant group) during the initial trials, but no additional episodes occurred in either group during long-term follow-up.
Overall, 53 individuals – 37 in the islet-alone and 16 in the islet-after–kidney transplant group – or 74% of the total, achieved a period of insulin independence with A1c under 7%, ranging from 36 to 481 days. The range of time to achieving insulin independence reflects individuals who received one, two, or three islet infusions.
The fact that most patients achieved insulin independence following just one (n = 20) or two (n = 30) infusions and only three patients required three infusions was notable, Dr. Rickels said.
“Currently, around the world, there’s an expectation of two to three donor pancreases being needed. Here, it’s one, maybe two. It’s a much more efficient protocol and opens up access for more islet transplantation as a hoped-for alternative to pancreas transplants.”
Of those who achieved insulin independence, 30 (57%) remained insulin-independent throughout follow-up (20 of 37 islet-alone and 10 of 16 islet-after–kidney transplant patients), with no difference in duration of insulin independence between the groups.
There were no deaths during post-transplant follow-up. Rates of serious adverse events were 0.31 and 0.43 per patient-year for the islet-after–kidney and islet-alone transplant groups, respectively. Of a total of 104 serious adverse events, 65 occurred during the initial trials and had been previously reported. Of the additional 39 serious adverse events that occurred during long-term follow-up, 11 were possibly due to immunosuppression and 27 were deemed unrelated to the procedures.
According to Dr. Rickels, “These are the most seriously affected patients, and you’d be expecting to see some hospitalizations in a population managed on immunosuppression therapy ... It’s important to note that none of the adverse events were related to the actual islet product. Also, kidney function remained stable during long-term follow-up in both cohorts, in fact, improving in those who had kidney transplants.”
Overall, he said, “This is a much less invasive procedure that opens itself up to significantly fewer complications than what many of these patients would otherwise require, a pancreas transplant, which involves major abdominal surgery.”
The investigators plan to submit these data as part of a biologic license application (BLA) to the FDA.
The research was supported by grants from JDRF, the National Institute of Diabetes and Digestive and Kidney Diseases, and the National Institute of Allergy and Infectious Diseases. Dr. Rickels has reported receiving consulting fees from Sernova and Vertex Pharmaceuticals.
A version of this article first appeared on Medscape.com.
Transplantation of cadaveric pancreatic islet cells resulted in graft survival and function with acceptable safety for up to 8 years in selected individuals with type 1 diabetes, new research finds.
The study is a long-term follow-up of two phase 3 pivotal trials from the Clinical Islet Transplantation Consortium of a purified human pancreatic islet cell product for treating people with type 1 diabetes.
One trial involved islet transplantation in 48 people who experienced severe hypoglycemia and hypoglycemic unawareness, and the other trial included 24 people who also experienced those complications and were already receiving immunosuppression following kidney transplant. The trials, both registered with the U.S. Food and Drug Administration (FDA), met their primary efficacy and safety endpoints at 2- and 3-year timepoints.
The follow-up data have now been published in Diabetes Care by Michael Rickels, MD, and colleagues.
The procedure involved infusion through the hepatic portal vein of one or more purified human pancreatic islet products under standardized immunosuppression using methods that Dr. Rickels and colleagues have been developing since 2004. The approach involves multiple modalities to protect the islets prior to transplantation.
Among the 34 islet-alone and eight islet-after–kidney transplant recipients who entered the extended follow-up, durable graft survival allowing for achievement of glycemic targets occurred without severe hypoglycemia or adverse effects from immunosuppression.
The primary outcome, actuarial survival of graft islet function, was 56% at the maximum follow-up of 8.3 years for the islet-only transplantation group and 49% at 7.3 years for the islet-after–kidney transplantation group (P = .004).
The findings suggest that “in the long run, islet transplantation has efficacy, including among those who have had kidney transplants ... Most type 1 diabetes patients are improved tremendously with current insulin delivery systems ... but for those having the most difficulty controlling their blood sugar – and those whose diabetes has already been complicated by needing a kidney transplant – the outcomes we saw in this study are what we’ve been hoping to achieve for more than 20 years,” said Dr. Rickels in a statement from his institution, the University of Pennsylvania, Philadelphia.
In the initial trials at day 75 after the initial transplant, 87.5% of the islet-alone and 71% of the islet-after–kidney transplant group achieved hemoglobin A1c under 7%, and 85% and 54%, respectively, achieved A1c at or under 6.5%. At the end of maximal follow-up, 49% of islet-only transplant recipients maintained A1c under 7%, although none had A1c at or under 6.5%. For the islet-after–kidney transplant group, these proportions were 35% and 17%, respectively (P = .0017 for A1c under 7.0% and P < .0001 for A1c ≤ 6.5%, respectively, between the groups).
There were 12 severe hypoglycemic episodes in five patients (three islet-alone and two islet-after–kidney transplant group) during the initial trials, but no additional episodes occurred in either group during long-term follow-up.
Overall, 53 individuals – 37 in the islet-alone and 16 in the islet-after–kidney transplant group – or 74% of the total, achieved a period of insulin independence with A1c under 7%, ranging from 36 to 481 days. The range of time to achieving insulin independence reflects individuals who received one, two, or three islet infusions.
The fact that most patients achieved insulin independence following just one (n = 20) or two (n = 30) infusions and only three patients required three infusions was notable, Dr. Rickels said.
“Currently, around the world, there’s an expectation of two to three donor pancreases being needed. Here, it’s one, maybe two. It’s a much more efficient protocol and opens up access for more islet transplantation as a hoped-for alternative to pancreas transplants.”
Of those who achieved insulin independence, 30 (57%) remained insulin-independent throughout follow-up (20 of 37 islet-alone and 10 of 16 islet-after–kidney transplant patients), with no difference in duration of insulin independence between the groups.
There were no deaths during post-transplant follow-up. Rates of serious adverse events were 0.31 and 0.43 per patient-year for the islet-after–kidney and islet-alone transplant groups, respectively. Of a total of 104 serious adverse events, 65 occurred during the initial trials and had been previously reported. Of the additional 39 serious adverse events that occurred during long-term follow-up, 11 were possibly due to immunosuppression and 27 were deemed unrelated to the procedures.
According to Dr. Rickels, “These are the most seriously affected patients, and you’d be expecting to see some hospitalizations in a population managed on immunosuppression therapy ... It’s important to note that none of the adverse events were related to the actual islet product. Also, kidney function remained stable during long-term follow-up in both cohorts, in fact, improving in those who had kidney transplants.”
Overall, he said, “This is a much less invasive procedure that opens itself up to significantly fewer complications than what many of these patients would otherwise require, a pancreas transplant, which involves major abdominal surgery.”
The investigators plan to submit these data as part of a biologic license application (BLA) to the FDA.
The research was supported by grants from JDRF, the National Institute of Diabetes and Digestive and Kidney Diseases, and the National Institute of Allergy and Infectious Diseases. Dr. Rickels has reported receiving consulting fees from Sernova and Vertex Pharmaceuticals.
A version of this article first appeared on Medscape.com.
Transplantation of cadaveric pancreatic islet cells resulted in graft survival and function with acceptable safety for up to 8 years in selected individuals with type 1 diabetes, new research finds.
The study is a long-term follow-up of two phase 3 pivotal trials from the Clinical Islet Transplantation Consortium of a purified human pancreatic islet cell product for treating people with type 1 diabetes.
One trial involved islet transplantation in 48 people who experienced severe hypoglycemia and hypoglycemic unawareness, and the other trial included 24 people who also experienced those complications and were already receiving immunosuppression following kidney transplant. The trials, both registered with the U.S. Food and Drug Administration (FDA), met their primary efficacy and safety endpoints at 2- and 3-year timepoints.
The follow-up data have now been published in Diabetes Care by Michael Rickels, MD, and colleagues.
The procedure involved infusion through the hepatic portal vein of one or more purified human pancreatic islet products under standardized immunosuppression using methods that Dr. Rickels and colleagues have been developing since 2004. The approach involves multiple modalities to protect the islets prior to transplantation.
Among the 34 islet-alone and eight islet-after–kidney transplant recipients who entered the extended follow-up, durable graft survival allowing for achievement of glycemic targets occurred without severe hypoglycemia or adverse effects from immunosuppression.
The primary outcome, actuarial survival of graft islet function, was 56% at the maximum follow-up of 8.3 years for the islet-only transplantation group and 49% at 7.3 years for the islet-after–kidney transplantation group (P = .004).
The findings suggest that “in the long run, islet transplantation has efficacy, including among those who have had kidney transplants ... Most type 1 diabetes patients are improved tremendously with current insulin delivery systems ... but for those having the most difficulty controlling their blood sugar – and those whose diabetes has already been complicated by needing a kidney transplant – the outcomes we saw in this study are what we’ve been hoping to achieve for more than 20 years,” said Dr. Rickels in a statement from his institution, the University of Pennsylvania, Philadelphia.
In the initial trials at day 75 after the initial transplant, 87.5% of the islet-alone and 71% of the islet-after–kidney transplant group achieved hemoglobin A1c under 7%, and 85% and 54%, respectively, achieved A1c at or under 6.5%. At the end of maximal follow-up, 49% of islet-only transplant recipients maintained A1c under 7%, although none had A1c at or under 6.5%. For the islet-after–kidney transplant group, these proportions were 35% and 17%, respectively (P = .0017 for A1c under 7.0% and P < .0001 for A1c ≤ 6.5%, respectively, between the groups).
There were 12 severe hypoglycemic episodes in five patients (three islet-alone and two islet-after–kidney transplant group) during the initial trials, but no additional episodes occurred in either group during long-term follow-up.
Overall, 53 individuals – 37 in the islet-alone and 16 in the islet-after–kidney transplant group – or 74% of the total, achieved a period of insulin independence with A1c under 7%, ranging from 36 to 481 days. The range of time to achieving insulin independence reflects individuals who received one, two, or three islet infusions.
The fact that most patients achieved insulin independence following just one (n = 20) or two (n = 30) infusions and only three patients required three infusions was notable, Dr. Rickels said.
“Currently, around the world, there’s an expectation of two to three donor pancreases being needed. Here, it’s one, maybe two. It’s a much more efficient protocol and opens up access for more islet transplantation as a hoped-for alternative to pancreas transplants.”
Of those who achieved insulin independence, 30 (57%) remained insulin-independent throughout follow-up (20 of 37 islet-alone and 10 of 16 islet-after–kidney transplant patients), with no difference in duration of insulin independence between the groups.
There were no deaths during post-transplant follow-up. Rates of serious adverse events were 0.31 and 0.43 per patient-year for the islet-after–kidney and islet-alone transplant groups, respectively. Of a total of 104 serious adverse events, 65 occurred during the initial trials and had been previously reported. Of the additional 39 serious adverse events that occurred during long-term follow-up, 11 were possibly due to immunosuppression and 27 were deemed unrelated to the procedures.
According to Dr. Rickels, “These are the most seriously affected patients, and you’d be expecting to see some hospitalizations in a population managed on immunosuppression therapy ... It’s important to note that none of the adverse events were related to the actual islet product. Also, kidney function remained stable during long-term follow-up in both cohorts, in fact, improving in those who had kidney transplants.”
Overall, he said, “This is a much less invasive procedure that opens itself up to significantly fewer complications than what many of these patients would otherwise require, a pancreas transplant, which involves major abdominal surgery.”
The investigators plan to submit these data as part of a biologic license application (BLA) to the FDA.
The research was supported by grants from JDRF, the National Institute of Diabetes and Digestive and Kidney Diseases, and the National Institute of Allergy and Infectious Diseases. Dr. Rickels has reported receiving consulting fees from Sernova and Vertex Pharmaceuticals.
A version of this article first appeared on Medscape.com.
FROM DIABETES CARE
Nonblanching Rash on the Legs and Chest
The Diagnosis: Leukemia Cutis
Hematoxylin and eosin staining revealed an infiltration of monomorphic atypical myeloid cells with cleaved nuclei within the dermis, with a relatively uninvolved epidermis (Figure, A). The cells formed aggregates in single-file lines along dermal collagen bundles. Occasional Auer rods, which are crystal aggregates of the enzyme myeloperoxidase, a marker unique to cells of the myeloid lineage (Figure, B) were appreciated.
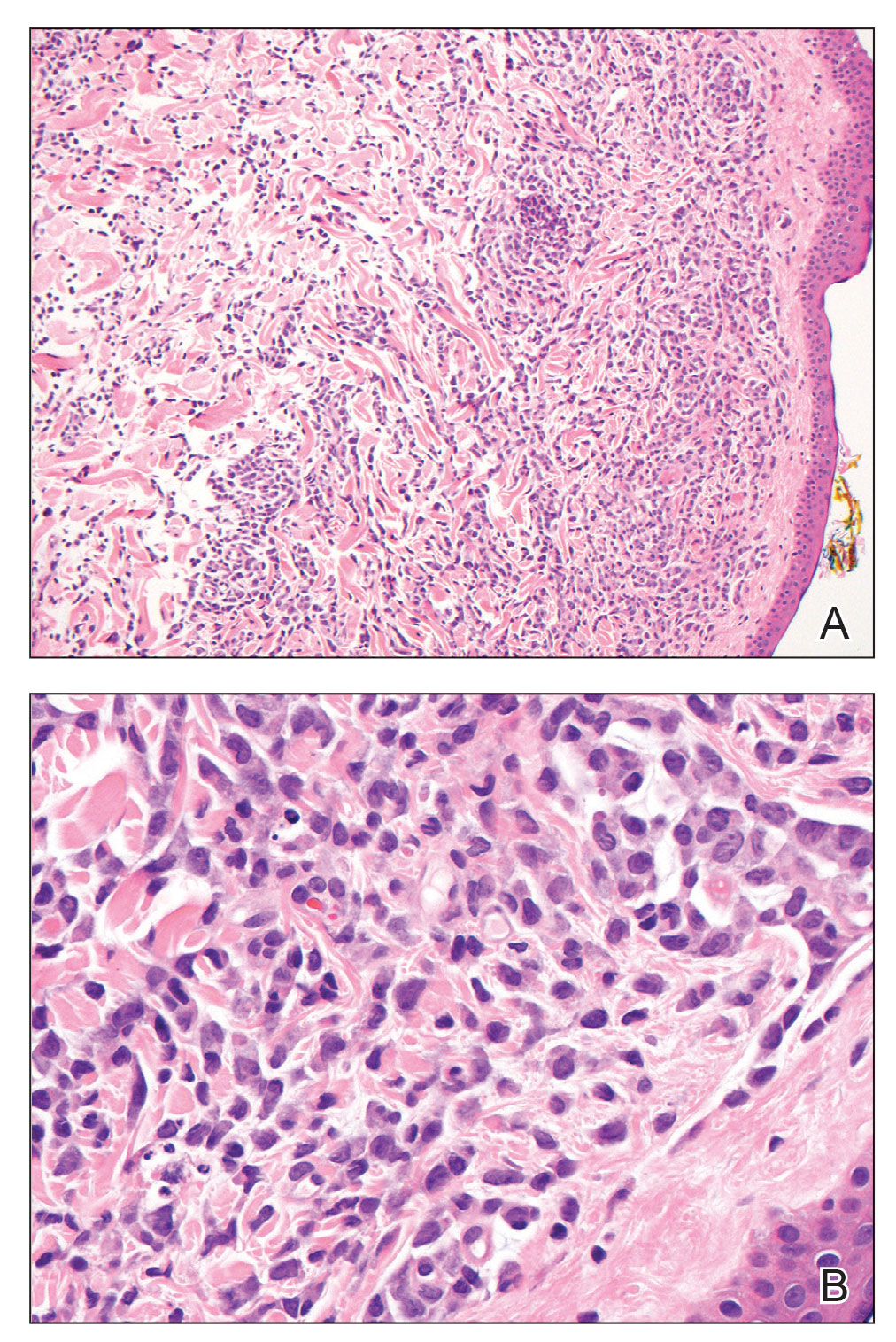
Immunohistochemical staining for myeloperoxidase was weakly positive; however, flow cytometric evaluation of the bone marrow aspirate revealed that approximately 20% of all CD45+ cells were myeloid blasts. These findings confirmed the diagnosis of recurrent acute myeloid leukemia (AML). The diagnosis of AML can be confirmed with a bone marrow biopsy demonstrating more than 20% of the total cells in blast form as well as evidence that the cells are of myeloid origin, which can be inferred by the presence of Auer rods, positive myeloperoxidase staining, or immunophenotyping. In our patient, the Auer rods, myeloperoxidase staining, and atypical myeloid cells on skin biopsy, in conjunction with the bone marrow biopsy results, confirmed leukemia cutis.
Leukemia cutis is the infiltration of neoplastic proliferating leukocytes in the epidermis, dermis, or subcutis from a primary or more commonly metastatic malignancy. Leukemic cutaneous involvement is seen in up to 13% of leukemia patients and most commonly is seen in monocytic or myelomonocytic forms of AML.1 It may present anywhere on the body but mostly is found on the back, trunk, and head. It also may have a predilection for areas with a history of trauma or inflammation. The lesions most often are firm, erythematous to violaceous papules and nodules, though leukemia cutis can present with hemorrhagic ulcers, purpura, or other cutaneous manifestations of concomitant thrombocytopenia such as petechiae and ecchymoses.2 Involvement of the lower extremities mimicking venous stasis dermatitis has been described.3,4
Treatment of leukemia cutis requires targeting the underlying leukemia2 under the guidance of hematology and oncology as well as the use of chemotherapeutic agents.5 The presence of leukemia cutis is a poor prognostic sign, and a discussion regarding goals of care often is appropriate. Our patient initially responded to FLAG (fludarabine, cytarabine, filgrastim) chemotherapy induction and consolidation, which was followed by midostaurin maintenance. However, she ultimately regressed, requiring decitabine and gilteritinib treatment, and died 9 months later from the course of the disease.
Although typically asymptomatic and presenting on the lower limbs, capillaritis (also known as the pigmented purpuric dermatoses) consists of a set of cutaneous conditions that often are chronic and relapsing in nature, as opposed to our patient’s subacute presentation. These benign conditions have several distinct morphologies; some are characterized by pigmented macules or pinpoint red-brown petechiae that most often are found on the legs but also are seen on the trunk and upper extremities.6 Of the various clinical presentations of capillaritis, our patient’s skin findings may be most consistent with pigmented purpuric lichenoid dermatitis of Gougerot and Blum, in which purpuric red-brown papules coalesce into plaques, though her lesions were not raised. The other pigmented purpuric dermatoses can present with cayenne pepper–colored petechiae, golden-brown macules, pruritic purpuric patches, or red-brown annular patches,6 which were not seen in our patient.
Venous stasis dermatitis also favors the lower extremities7; however, it classically includes the medial malleolus and often presents with scaling and hyperpigmentation from hemosiderin deposition.8 It often is associated with pruritus, as opposed to the nonpruritic nonpainful lesions in leukemia cutis. Other signs of venous insufficiency also may be appreciated, including edema or varicose veins,7 which were not evident in our patient.
Leukocytoclastic vasculitis, a small vessel vasculitis, also appears as palpable or macular purpura, which classically is asymptomatic and erupts on the shins approximately 1 week after an inciting exposure,9 such as medications, pathogens, or autoimmune diseases. One of the least distinctive vasculitides is polyarteritis nodosa, a form of medium vessel vasculitis, which presents most often with palpable purpura or painful nodules on the lower extremities and may be accompanied by livedo reticularis or digital necrosis.9 Acute leukemia may be accompanied by inflammatory paraneoplastic conditions including vasculitis, which is thought to be due to leukemic cells infiltrating and damaging blood vessels.10
Pretibial myxedema is closely associated with Graves disease and shares some features seen in the presentation of our patient’s leukemia cutis. It is asymptomatic, classically affects the pretibial regions, and most commonly affects older adults and women.11,12 Pretibial myxedema presents with thick indurated plaques rather than patches. Our patient did not demonstrate ophthalmopathy, which nearly always precedes pretibial myxedema.12 The most common form of pretibial myxedema is nonpitting, though nodular, plaquelike, polypoid, and elephantiasic forms also exist.11 Pretibial myxedema classically favors the shins; however, it also can affect the ankles, dorsal aspects of the feet, and toes. The characteristic induration of the skin is believed to be the result of excess fibroblast production of glycosaminoglycans in the dermis and subcutis likely triggered by stimulation of fibroblast thyroid stimulating hormone receptors.11
- Bakst RL, Tallman MS, Douer D, et al. How I treat extramedullary acute myeloid leukemia. Blood. 2011;118:3785-3793.
- Bolognia JL, Schaffer JV, Duncan KO, et al. Other lymphoproliferative and myeloproliferative diseases. In: Bolognia JL, Schaffer JV, Duncan KO, et al, eds. Dermatology Essentials. 2nd ed. Elsevier; 2014:973-977.
- Papadavid E, Panayiotides I, Katoulis A, et al. Stasis dermatitis-like leukaemic infiltration in a patient with myelodysplastic syndrome. Clin Exp Dermatol. 2008;33:298-300.
- Chang HY, Wong KM, Bosenberg M, et al. Myelogenous leukemia cutis resembling stasis dermatitis. J Am Acad Dermatol. 2003;49:128-129.
- Aguilera SB, Zarraga M, Rosen L. Leukemia cutis in a patient with acute myelogenous leukemia: a case report and review of the literature. Cutis. 2010;85:31-36.
- Kim DH, Seo SH, Ahn HH, et al. Characteristics and clinical manifestations of pigmented purpuric dermatosis. Ann Dermatol. 2015;27:404-410.
- Bolognia JL, Schaffer JV, Duncan KO, et al. Other eczematous eruptions. In: Bolognia JL, Schaffer JV, Duncan KO, et al, eds. Dermatology Essentials. 2nd ed. Elsevier; 2014:103-108.
- Krooks JA, Weatherall AG. Leukemia cutis in acute myeloid leukemia signifies a poor prognosis. Cutis. 2018;102:266, 271-272.
- Wetter DA, Dutz JP, Shinkai K, et al. Cutaneous vasculitis. In: Bolognia JL, Schaffer JV, Lorenzo C, eds. Dermatology. 4th ed. Elsevier; 2018:409-439.
- Jones D, Dorfman DM, Barnhill RL, et al. Leukemic vasculitis: a feature of leukemia cutis in some patients. Am J Clin Pathol. 1997;107:637-642.
- Fatourechi V. Pretibial myxedema: pathophysiology and treatment options. Am J Clin Dermatol. 2005;6:295-309.
- Fatourechi V, Pajouhi M, Fransway AF. Dermopathy of Graves disease (pretibial myxedema). review of 150 cases. Medicine (Baltimore). 1994;73:1-7.
The Diagnosis: Leukemia Cutis
Hematoxylin and eosin staining revealed an infiltration of monomorphic atypical myeloid cells with cleaved nuclei within the dermis, with a relatively uninvolved epidermis (Figure, A). The cells formed aggregates in single-file lines along dermal collagen bundles. Occasional Auer rods, which are crystal aggregates of the enzyme myeloperoxidase, a marker unique to cells of the myeloid lineage (Figure, B) were appreciated.

Immunohistochemical staining for myeloperoxidase was weakly positive; however, flow cytometric evaluation of the bone marrow aspirate revealed that approximately 20% of all CD45+ cells were myeloid blasts. These findings confirmed the diagnosis of recurrent acute myeloid leukemia (AML). The diagnosis of AML can be confirmed with a bone marrow biopsy demonstrating more than 20% of the total cells in blast form as well as evidence that the cells are of myeloid origin, which can be inferred by the presence of Auer rods, positive myeloperoxidase staining, or immunophenotyping. In our patient, the Auer rods, myeloperoxidase staining, and atypical myeloid cells on skin biopsy, in conjunction with the bone marrow biopsy results, confirmed leukemia cutis.
Leukemia cutis is the infiltration of neoplastic proliferating leukocytes in the epidermis, dermis, or subcutis from a primary or more commonly metastatic malignancy. Leukemic cutaneous involvement is seen in up to 13% of leukemia patients and most commonly is seen in monocytic or myelomonocytic forms of AML.1 It may present anywhere on the body but mostly is found on the back, trunk, and head. It also may have a predilection for areas with a history of trauma or inflammation. The lesions most often are firm, erythematous to violaceous papules and nodules, though leukemia cutis can present with hemorrhagic ulcers, purpura, or other cutaneous manifestations of concomitant thrombocytopenia such as petechiae and ecchymoses.2 Involvement of the lower extremities mimicking venous stasis dermatitis has been described.3,4
Treatment of leukemia cutis requires targeting the underlying leukemia2 under the guidance of hematology and oncology as well as the use of chemotherapeutic agents.5 The presence of leukemia cutis is a poor prognostic sign, and a discussion regarding goals of care often is appropriate. Our patient initially responded to FLAG (fludarabine, cytarabine, filgrastim) chemotherapy induction and consolidation, which was followed by midostaurin maintenance. However, she ultimately regressed, requiring decitabine and gilteritinib treatment, and died 9 months later from the course of the disease.
Although typically asymptomatic and presenting on the lower limbs, capillaritis (also known as the pigmented purpuric dermatoses) consists of a set of cutaneous conditions that often are chronic and relapsing in nature, as opposed to our patient’s subacute presentation. These benign conditions have several distinct morphologies; some are characterized by pigmented macules or pinpoint red-brown petechiae that most often are found on the legs but also are seen on the trunk and upper extremities.6 Of the various clinical presentations of capillaritis, our patient’s skin findings may be most consistent with pigmented purpuric lichenoid dermatitis of Gougerot and Blum, in which purpuric red-brown papules coalesce into plaques, though her lesions were not raised. The other pigmented purpuric dermatoses can present with cayenne pepper–colored petechiae, golden-brown macules, pruritic purpuric patches, or red-brown annular patches,6 which were not seen in our patient.
Venous stasis dermatitis also favors the lower extremities7; however, it classically includes the medial malleolus and often presents with scaling and hyperpigmentation from hemosiderin deposition.8 It often is associated with pruritus, as opposed to the nonpruritic nonpainful lesions in leukemia cutis. Other signs of venous insufficiency also may be appreciated, including edema or varicose veins,7 which were not evident in our patient.
Leukocytoclastic vasculitis, a small vessel vasculitis, also appears as palpable or macular purpura, which classically is asymptomatic and erupts on the shins approximately 1 week after an inciting exposure,9 such as medications, pathogens, or autoimmune diseases. One of the least distinctive vasculitides is polyarteritis nodosa, a form of medium vessel vasculitis, which presents most often with palpable purpura or painful nodules on the lower extremities and may be accompanied by livedo reticularis or digital necrosis.9 Acute leukemia may be accompanied by inflammatory paraneoplastic conditions including vasculitis, which is thought to be due to leukemic cells infiltrating and damaging blood vessels.10
Pretibial myxedema is closely associated with Graves disease and shares some features seen in the presentation of our patient’s leukemia cutis. It is asymptomatic, classically affects the pretibial regions, and most commonly affects older adults and women.11,12 Pretibial myxedema presents with thick indurated plaques rather than patches. Our patient did not demonstrate ophthalmopathy, which nearly always precedes pretibial myxedema.12 The most common form of pretibial myxedema is nonpitting, though nodular, plaquelike, polypoid, and elephantiasic forms also exist.11 Pretibial myxedema classically favors the shins; however, it also can affect the ankles, dorsal aspects of the feet, and toes. The characteristic induration of the skin is believed to be the result of excess fibroblast production of glycosaminoglycans in the dermis and subcutis likely triggered by stimulation of fibroblast thyroid stimulating hormone receptors.11
The Diagnosis: Leukemia Cutis
Hematoxylin and eosin staining revealed an infiltration of monomorphic atypical myeloid cells with cleaved nuclei within the dermis, with a relatively uninvolved epidermis (Figure, A). The cells formed aggregates in single-file lines along dermal collagen bundles. Occasional Auer rods, which are crystal aggregates of the enzyme myeloperoxidase, a marker unique to cells of the myeloid lineage (Figure, B) were appreciated.

Immunohistochemical staining for myeloperoxidase was weakly positive; however, flow cytometric evaluation of the bone marrow aspirate revealed that approximately 20% of all CD45+ cells were myeloid blasts. These findings confirmed the diagnosis of recurrent acute myeloid leukemia (AML). The diagnosis of AML can be confirmed with a bone marrow biopsy demonstrating more than 20% of the total cells in blast form as well as evidence that the cells are of myeloid origin, which can be inferred by the presence of Auer rods, positive myeloperoxidase staining, or immunophenotyping. In our patient, the Auer rods, myeloperoxidase staining, and atypical myeloid cells on skin biopsy, in conjunction with the bone marrow biopsy results, confirmed leukemia cutis.
Leukemia cutis is the infiltration of neoplastic proliferating leukocytes in the epidermis, dermis, or subcutis from a primary or more commonly metastatic malignancy. Leukemic cutaneous involvement is seen in up to 13% of leukemia patients and most commonly is seen in monocytic or myelomonocytic forms of AML.1 It may present anywhere on the body but mostly is found on the back, trunk, and head. It also may have a predilection for areas with a history of trauma or inflammation. The lesions most often are firm, erythematous to violaceous papules and nodules, though leukemia cutis can present with hemorrhagic ulcers, purpura, or other cutaneous manifestations of concomitant thrombocytopenia such as petechiae and ecchymoses.2 Involvement of the lower extremities mimicking venous stasis dermatitis has been described.3,4
Treatment of leukemia cutis requires targeting the underlying leukemia2 under the guidance of hematology and oncology as well as the use of chemotherapeutic agents.5 The presence of leukemia cutis is a poor prognostic sign, and a discussion regarding goals of care often is appropriate. Our patient initially responded to FLAG (fludarabine, cytarabine, filgrastim) chemotherapy induction and consolidation, which was followed by midostaurin maintenance. However, she ultimately regressed, requiring decitabine and gilteritinib treatment, and died 9 months later from the course of the disease.
Although typically asymptomatic and presenting on the lower limbs, capillaritis (also known as the pigmented purpuric dermatoses) consists of a set of cutaneous conditions that often are chronic and relapsing in nature, as opposed to our patient’s subacute presentation. These benign conditions have several distinct morphologies; some are characterized by pigmented macules or pinpoint red-brown petechiae that most often are found on the legs but also are seen on the trunk and upper extremities.6 Of the various clinical presentations of capillaritis, our patient’s skin findings may be most consistent with pigmented purpuric lichenoid dermatitis of Gougerot and Blum, in which purpuric red-brown papules coalesce into plaques, though her lesions were not raised. The other pigmented purpuric dermatoses can present with cayenne pepper–colored petechiae, golden-brown macules, pruritic purpuric patches, or red-brown annular patches,6 which were not seen in our patient.
Venous stasis dermatitis also favors the lower extremities7; however, it classically includes the medial malleolus and often presents with scaling and hyperpigmentation from hemosiderin deposition.8 It often is associated with pruritus, as opposed to the nonpruritic nonpainful lesions in leukemia cutis. Other signs of venous insufficiency also may be appreciated, including edema or varicose veins,7 which were not evident in our patient.
Leukocytoclastic vasculitis, a small vessel vasculitis, also appears as palpable or macular purpura, which classically is asymptomatic and erupts on the shins approximately 1 week after an inciting exposure,9 such as medications, pathogens, or autoimmune diseases. One of the least distinctive vasculitides is polyarteritis nodosa, a form of medium vessel vasculitis, which presents most often with palpable purpura or painful nodules on the lower extremities and may be accompanied by livedo reticularis or digital necrosis.9 Acute leukemia may be accompanied by inflammatory paraneoplastic conditions including vasculitis, which is thought to be due to leukemic cells infiltrating and damaging blood vessels.10
Pretibial myxedema is closely associated with Graves disease and shares some features seen in the presentation of our patient’s leukemia cutis. It is asymptomatic, classically affects the pretibial regions, and most commonly affects older adults and women.11,12 Pretibial myxedema presents with thick indurated plaques rather than patches. Our patient did not demonstrate ophthalmopathy, which nearly always precedes pretibial myxedema.12 The most common form of pretibial myxedema is nonpitting, though nodular, plaquelike, polypoid, and elephantiasic forms also exist.11 Pretibial myxedema classically favors the shins; however, it also can affect the ankles, dorsal aspects of the feet, and toes. The characteristic induration of the skin is believed to be the result of excess fibroblast production of glycosaminoglycans in the dermis and subcutis likely triggered by stimulation of fibroblast thyroid stimulating hormone receptors.11
- Bakst RL, Tallman MS, Douer D, et al. How I treat extramedullary acute myeloid leukemia. Blood. 2011;118:3785-3793.
- Bolognia JL, Schaffer JV, Duncan KO, et al. Other lymphoproliferative and myeloproliferative diseases. In: Bolognia JL, Schaffer JV, Duncan KO, et al, eds. Dermatology Essentials. 2nd ed. Elsevier; 2014:973-977.
- Papadavid E, Panayiotides I, Katoulis A, et al. Stasis dermatitis-like leukaemic infiltration in a patient with myelodysplastic syndrome. Clin Exp Dermatol. 2008;33:298-300.
- Chang HY, Wong KM, Bosenberg M, et al. Myelogenous leukemia cutis resembling stasis dermatitis. J Am Acad Dermatol. 2003;49:128-129.
- Aguilera SB, Zarraga M, Rosen L. Leukemia cutis in a patient with acute myelogenous leukemia: a case report and review of the literature. Cutis. 2010;85:31-36.
- Kim DH, Seo SH, Ahn HH, et al. Characteristics and clinical manifestations of pigmented purpuric dermatosis. Ann Dermatol. 2015;27:404-410.
- Bolognia JL, Schaffer JV, Duncan KO, et al. Other eczematous eruptions. In: Bolognia JL, Schaffer JV, Duncan KO, et al, eds. Dermatology Essentials. 2nd ed. Elsevier; 2014:103-108.
- Krooks JA, Weatherall AG. Leukemia cutis in acute myeloid leukemia signifies a poor prognosis. Cutis. 2018;102:266, 271-272.
- Wetter DA, Dutz JP, Shinkai K, et al. Cutaneous vasculitis. In: Bolognia JL, Schaffer JV, Lorenzo C, eds. Dermatology. 4th ed. Elsevier; 2018:409-439.
- Jones D, Dorfman DM, Barnhill RL, et al. Leukemic vasculitis: a feature of leukemia cutis in some patients. Am J Clin Pathol. 1997;107:637-642.
- Fatourechi V. Pretibial myxedema: pathophysiology and treatment options. Am J Clin Dermatol. 2005;6:295-309.
- Fatourechi V, Pajouhi M, Fransway AF. Dermopathy of Graves disease (pretibial myxedema). review of 150 cases. Medicine (Baltimore). 1994;73:1-7.
- Bakst RL, Tallman MS, Douer D, et al. How I treat extramedullary acute myeloid leukemia. Blood. 2011;118:3785-3793.
- Bolognia JL, Schaffer JV, Duncan KO, et al. Other lymphoproliferative and myeloproliferative diseases. In: Bolognia JL, Schaffer JV, Duncan KO, et al, eds. Dermatology Essentials. 2nd ed. Elsevier; 2014:973-977.
- Papadavid E, Panayiotides I, Katoulis A, et al. Stasis dermatitis-like leukaemic infiltration in a patient with myelodysplastic syndrome. Clin Exp Dermatol. 2008;33:298-300.
- Chang HY, Wong KM, Bosenberg M, et al. Myelogenous leukemia cutis resembling stasis dermatitis. J Am Acad Dermatol. 2003;49:128-129.
- Aguilera SB, Zarraga M, Rosen L. Leukemia cutis in a patient with acute myelogenous leukemia: a case report and review of the literature. Cutis. 2010;85:31-36.
- Kim DH, Seo SH, Ahn HH, et al. Characteristics and clinical manifestations of pigmented purpuric dermatosis. Ann Dermatol. 2015;27:404-410.
- Bolognia JL, Schaffer JV, Duncan KO, et al. Other eczematous eruptions. In: Bolognia JL, Schaffer JV, Duncan KO, et al, eds. Dermatology Essentials. 2nd ed. Elsevier; 2014:103-108.
- Krooks JA, Weatherall AG. Leukemia cutis in acute myeloid leukemia signifies a poor prognosis. Cutis. 2018;102:266, 271-272.
- Wetter DA, Dutz JP, Shinkai K, et al. Cutaneous vasculitis. In: Bolognia JL, Schaffer JV, Lorenzo C, eds. Dermatology. 4th ed. Elsevier; 2018:409-439.
- Jones D, Dorfman DM, Barnhill RL, et al. Leukemic vasculitis: a feature of leukemia cutis in some patients. Am J Clin Pathol. 1997;107:637-642.
- Fatourechi V. Pretibial myxedema: pathophysiology and treatment options. Am J Clin Dermatol. 2005;6:295-309.
- Fatourechi V, Pajouhi M, Fransway AF. Dermopathy of Graves disease (pretibial myxedema). review of 150 cases. Medicine (Baltimore). 1994;73:1-7.
A 67-year-old woman with history of atrial fibrillation and leukemia presented with a nonpruritic nonpainful rash of 10 days' duration that began on the distal lower extremities (top) and then spread superiorly. She reported having a sore throat and mouth, cough, night sweats, unintentional weight loss, and lymphadenopathy. Physical examination revealed pink-purple nonblanching macules and patches on the lower extremities extending from the ankles to the knees. She also had firm pink papules on the chest (bottom) and back. Punch biopsies of the skin on the chest and leg were obtained for histologic examination and immunohistochemical staining.
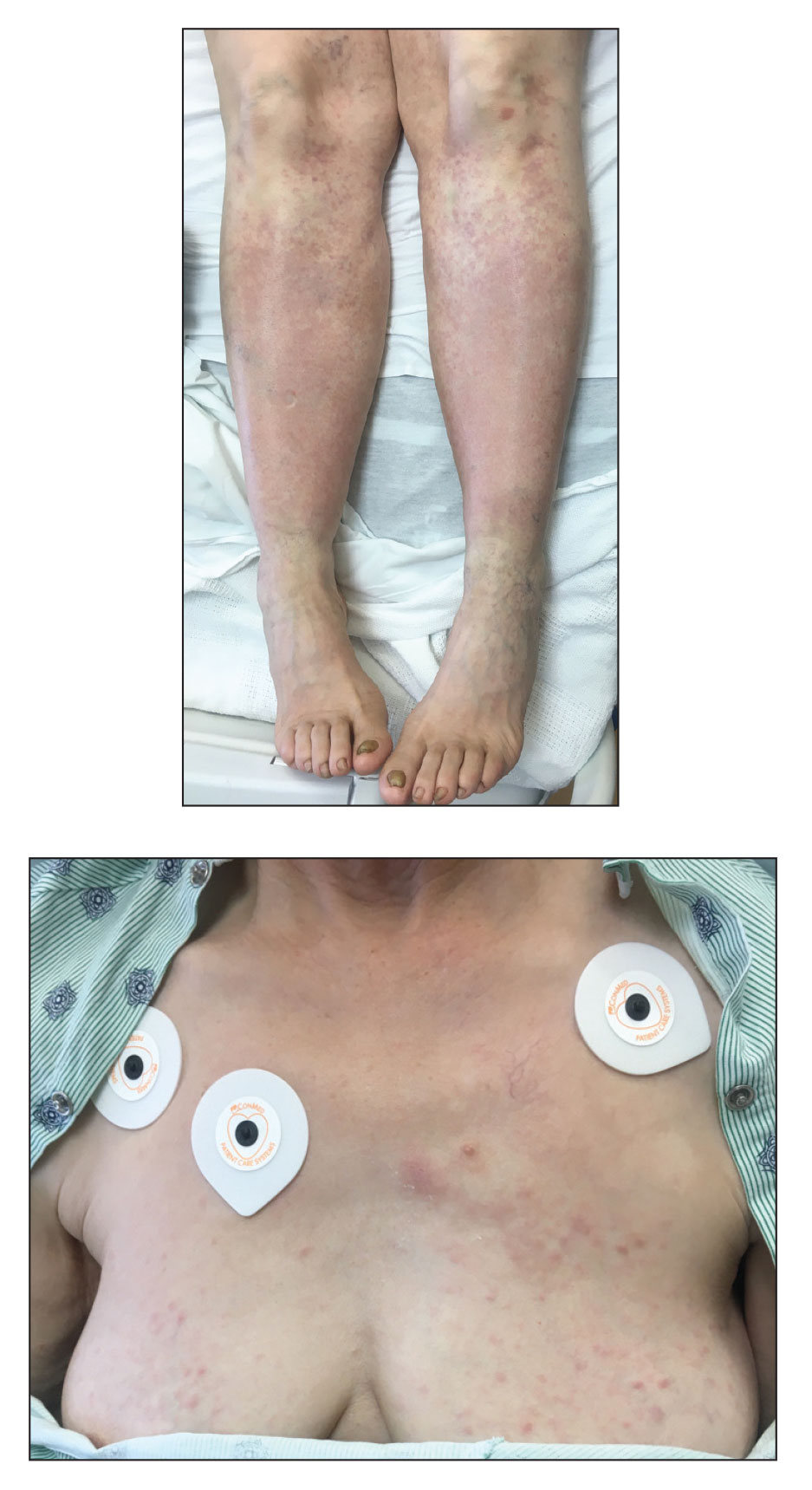
Diabetes becoming less potent risk factor for CVD events
Diabetes persists as a risk factor for cardiovascular events, but where it once meant the same risk of heart attack or stroke as cardiovascular disease itself, a large Canadian population study reports that’s no longer the case. Thanks to advances in diabetes management over the past quarter century, diabetes is no longer considered equivalent to CVD as a risk factor for cardiovascular events, researchers from the University of Toronto reported.
The retrospective, population-based study used administrative data from Ontario’s provincial universal health care system. The researchers created five population-based cohorts of adults at 5-year intervals from 1994 to 2014, consisting of 1.87 million adults in the first cohort and 1.5 million in the last. In that 20-year span, the prevalence of diabetes in this population tripled, from 3.1% to 9%.
“In the last 25 years we’ve seen wholesale changes in the way people approach diabetes,” lead study author Calvin Ke, MD, PhD, an endocrinologist and assistant professor at the University of Toronto, said in an interview. “Part of the findings show that diabetes and cardiovascular disease were equivalent for risk of cardiovascular events in 1994, but by 2014 that was not the case.”
However, Dr. Ke added, “Diabetes is still a very strong cardiovascular risk factor.”
The investigators for the study, reported as a research letter in JAMA, analyzed the risk of cardiovascular events in four subgroups: those who had both diabetes and CVD, CVD only, diabetes only, and no CVD or diabetes.
Between 1994 and 2014, the cardiovascular event rates declined significantly among people with diabetes alone, compared with people with no disease: from 28.4 to 12.7 per 1,000 person-years, or an absolute risk increase (ARI) of 4.4% and a relative risk (RR) more than double (2.06), in 1994 to 14 vs. 8 per 1,000 person-years, and an ARI of 2% and RR less than double (1.58) 20 years later.
Among people with CVD only, those values shifted from 36.1 per 1,000 person-years, ARI of 5.1% and RR of 2.16 in 1994 to 23.9, ARI of 3.7% and RR still more than double (2.06) in 2014.
People with both CVD and diabetes had the highest CVD event rates across all 5-year cohorts: 74 per 1,000 person-years, ARI of 12% and RR almost four times greater (3.81) in 1994 than people with no disease. By 2014, the ARI in this group was 7.6% and the RR 3.10.
The investigators calculated that event rates from 1994 to 2014 declined across all four subgroups, with rate ratios of 0.49 for diabetes only, 0.66 for CVD only, 0.60 for both diabetes and CVD, and 0.63 for neither disease.
Shift in practice
The study noted that the shift in diabetes as a risk factor for heart attack and stroke is “a change that likely reflects the use of modern, multifactorial approaches to diabetes.”
“A number of changes have occurred in practice that really focus on this idea of a multifactorial approach to diabetes: more aggressive management of blood sugar, blood pressure, and lipids,” Dr. Ke said. “We know from the statin trials that statins can reduce the risk of heart disease significantly, and the use of statins increased from 28.4% in 1999 to 56.3% in 2018 in the United States,” Dr. Ke said. He added that statin use in Canada in adults ages 40 and older went from 1.2% in 1994 to 58.4% in 2010-2015. Use of ACE inhibitors and angiotensin receptor blockers for hypertension followed similar trends, contributing further to reducing risks for heart attack and stroke, Dr. Ke said.
Dr. Ke also noted that the evolution of guidelines and advances in treatments for both CVD and diabetes since 1994 have contributed to improving risks for people with diabetes. SGLT2 inhibitors have been linked to a 2%-6% reduction in hemoglobin A1c, he said. “All of these factors combined have had a major effect on the reduced risk of cardiovascular events.”
Prakash Deedwania, MD, professor at the University of California, San Francisco, Fresno, said that this study confirms a trend that others have reported regarding the risk of CVD in diabetes. The large database covering millions of adults is a study strength, he said.
And the findings, Dr. Deedwania added, underscore what’s been published in clinical guidelines, notably the American Heart Association scientific statement for managing CVD risk in patients with diabetes. “This means that, from observations made 20-plus years ago, when most people were not being treated for diabetes or heart disease, the pendulum has swung,” he said.
However, he added, “The authors state clearly that it does not mean that diabetes is not associated with a higher risk of cardiovascular events; it just means it is no longer equivalent to CVD.”
Managing diabetes continues to be “particularly important,” Dr. Deedwania said, because the prevalence of diabetes continues to rise. “This is a phenomenal risk, and it emphasizes that, to really conquer or control diabetes, we should make every effort to prevent diabetes,” he said.
Dr. Ke and Dr. Deedwania have no relevant financial relationships to disclose.
Diabetes persists as a risk factor for cardiovascular events, but where it once meant the same risk of heart attack or stroke as cardiovascular disease itself, a large Canadian population study reports that’s no longer the case. Thanks to advances in diabetes management over the past quarter century, diabetes is no longer considered equivalent to CVD as a risk factor for cardiovascular events, researchers from the University of Toronto reported.
The retrospective, population-based study used administrative data from Ontario’s provincial universal health care system. The researchers created five population-based cohorts of adults at 5-year intervals from 1994 to 2014, consisting of 1.87 million adults in the first cohort and 1.5 million in the last. In that 20-year span, the prevalence of diabetes in this population tripled, from 3.1% to 9%.
“In the last 25 years we’ve seen wholesale changes in the way people approach diabetes,” lead study author Calvin Ke, MD, PhD, an endocrinologist and assistant professor at the University of Toronto, said in an interview. “Part of the findings show that diabetes and cardiovascular disease were equivalent for risk of cardiovascular events in 1994, but by 2014 that was not the case.”
However, Dr. Ke added, “Diabetes is still a very strong cardiovascular risk factor.”
The investigators for the study, reported as a research letter in JAMA, analyzed the risk of cardiovascular events in four subgroups: those who had both diabetes and CVD, CVD only, diabetes only, and no CVD or diabetes.
Between 1994 and 2014, the cardiovascular event rates declined significantly among people with diabetes alone, compared with people with no disease: from 28.4 to 12.7 per 1,000 person-years, or an absolute risk increase (ARI) of 4.4% and a relative risk (RR) more than double (2.06), in 1994 to 14 vs. 8 per 1,000 person-years, and an ARI of 2% and RR less than double (1.58) 20 years later.
Among people with CVD only, those values shifted from 36.1 per 1,000 person-years, ARI of 5.1% and RR of 2.16 in 1994 to 23.9, ARI of 3.7% and RR still more than double (2.06) in 2014.
People with both CVD and diabetes had the highest CVD event rates across all 5-year cohorts: 74 per 1,000 person-years, ARI of 12% and RR almost four times greater (3.81) in 1994 than people with no disease. By 2014, the ARI in this group was 7.6% and the RR 3.10.
The investigators calculated that event rates from 1994 to 2014 declined across all four subgroups, with rate ratios of 0.49 for diabetes only, 0.66 for CVD only, 0.60 for both diabetes and CVD, and 0.63 for neither disease.
Shift in practice
The study noted that the shift in diabetes as a risk factor for heart attack and stroke is “a change that likely reflects the use of modern, multifactorial approaches to diabetes.”
“A number of changes have occurred in practice that really focus on this idea of a multifactorial approach to diabetes: more aggressive management of blood sugar, blood pressure, and lipids,” Dr. Ke said. “We know from the statin trials that statins can reduce the risk of heart disease significantly, and the use of statins increased from 28.4% in 1999 to 56.3% in 2018 in the United States,” Dr. Ke said. He added that statin use in Canada in adults ages 40 and older went from 1.2% in 1994 to 58.4% in 2010-2015. Use of ACE inhibitors and angiotensin receptor blockers for hypertension followed similar trends, contributing further to reducing risks for heart attack and stroke, Dr. Ke said.
Dr. Ke also noted that the evolution of guidelines and advances in treatments for both CVD and diabetes since 1994 have contributed to improving risks for people with diabetes. SGLT2 inhibitors have been linked to a 2%-6% reduction in hemoglobin A1c, he said. “All of these factors combined have had a major effect on the reduced risk of cardiovascular events.”
Prakash Deedwania, MD, professor at the University of California, San Francisco, Fresno, said that this study confirms a trend that others have reported regarding the risk of CVD in diabetes. The large database covering millions of adults is a study strength, he said.
And the findings, Dr. Deedwania added, underscore what’s been published in clinical guidelines, notably the American Heart Association scientific statement for managing CVD risk in patients with diabetes. “This means that, from observations made 20-plus years ago, when most people were not being treated for diabetes or heart disease, the pendulum has swung,” he said.
However, he added, “The authors state clearly that it does not mean that diabetes is not associated with a higher risk of cardiovascular events; it just means it is no longer equivalent to CVD.”
Managing diabetes continues to be “particularly important,” Dr. Deedwania said, because the prevalence of diabetes continues to rise. “This is a phenomenal risk, and it emphasizes that, to really conquer or control diabetes, we should make every effort to prevent diabetes,” he said.
Dr. Ke and Dr. Deedwania have no relevant financial relationships to disclose.
Diabetes persists as a risk factor for cardiovascular events, but where it once meant the same risk of heart attack or stroke as cardiovascular disease itself, a large Canadian population study reports that’s no longer the case. Thanks to advances in diabetes management over the past quarter century, diabetes is no longer considered equivalent to CVD as a risk factor for cardiovascular events, researchers from the University of Toronto reported.
The retrospective, population-based study used administrative data from Ontario’s provincial universal health care system. The researchers created five population-based cohorts of adults at 5-year intervals from 1994 to 2014, consisting of 1.87 million adults in the first cohort and 1.5 million in the last. In that 20-year span, the prevalence of diabetes in this population tripled, from 3.1% to 9%.
“In the last 25 years we’ve seen wholesale changes in the way people approach diabetes,” lead study author Calvin Ke, MD, PhD, an endocrinologist and assistant professor at the University of Toronto, said in an interview. “Part of the findings show that diabetes and cardiovascular disease were equivalent for risk of cardiovascular events in 1994, but by 2014 that was not the case.”
However, Dr. Ke added, “Diabetes is still a very strong cardiovascular risk factor.”
The investigators for the study, reported as a research letter in JAMA, analyzed the risk of cardiovascular events in four subgroups: those who had both diabetes and CVD, CVD only, diabetes only, and no CVD or diabetes.
Between 1994 and 2014, the cardiovascular event rates declined significantly among people with diabetes alone, compared with people with no disease: from 28.4 to 12.7 per 1,000 person-years, or an absolute risk increase (ARI) of 4.4% and a relative risk (RR) more than double (2.06), in 1994 to 14 vs. 8 per 1,000 person-years, and an ARI of 2% and RR less than double (1.58) 20 years later.
Among people with CVD only, those values shifted from 36.1 per 1,000 person-years, ARI of 5.1% and RR of 2.16 in 1994 to 23.9, ARI of 3.7% and RR still more than double (2.06) in 2014.
People with both CVD and diabetes had the highest CVD event rates across all 5-year cohorts: 74 per 1,000 person-years, ARI of 12% and RR almost four times greater (3.81) in 1994 than people with no disease. By 2014, the ARI in this group was 7.6% and the RR 3.10.
The investigators calculated that event rates from 1994 to 2014 declined across all four subgroups, with rate ratios of 0.49 for diabetes only, 0.66 for CVD only, 0.60 for both diabetes and CVD, and 0.63 for neither disease.
Shift in practice
The study noted that the shift in diabetes as a risk factor for heart attack and stroke is “a change that likely reflects the use of modern, multifactorial approaches to diabetes.”
“A number of changes have occurred in practice that really focus on this idea of a multifactorial approach to diabetes: more aggressive management of blood sugar, blood pressure, and lipids,” Dr. Ke said. “We know from the statin trials that statins can reduce the risk of heart disease significantly, and the use of statins increased from 28.4% in 1999 to 56.3% in 2018 in the United States,” Dr. Ke said. He added that statin use in Canada in adults ages 40 and older went from 1.2% in 1994 to 58.4% in 2010-2015. Use of ACE inhibitors and angiotensin receptor blockers for hypertension followed similar trends, contributing further to reducing risks for heart attack and stroke, Dr. Ke said.
Dr. Ke also noted that the evolution of guidelines and advances in treatments for both CVD and diabetes since 1994 have contributed to improving risks for people with diabetes. SGLT2 inhibitors have been linked to a 2%-6% reduction in hemoglobin A1c, he said. “All of these factors combined have had a major effect on the reduced risk of cardiovascular events.”
Prakash Deedwania, MD, professor at the University of California, San Francisco, Fresno, said that this study confirms a trend that others have reported regarding the risk of CVD in diabetes. The large database covering millions of adults is a study strength, he said.
And the findings, Dr. Deedwania added, underscore what’s been published in clinical guidelines, notably the American Heart Association scientific statement for managing CVD risk in patients with diabetes. “This means that, from observations made 20-plus years ago, when most people were not being treated for diabetes or heart disease, the pendulum has swung,” he said.
However, he added, “The authors state clearly that it does not mean that diabetes is not associated with a higher risk of cardiovascular events; it just means it is no longer equivalent to CVD.”
Managing diabetes continues to be “particularly important,” Dr. Deedwania said, because the prevalence of diabetes continues to rise. “This is a phenomenal risk, and it emphasizes that, to really conquer or control diabetes, we should make every effort to prevent diabetes,” he said.
Dr. Ke and Dr. Deedwania have no relevant financial relationships to disclose.
FROM JAMA
Large trial of home-based COPD rehab shows positive results
NASHVILLE – The first multicenter randomized controlled trial of a home-based rehabilitation program for patients with chronic obstructive pulmonary disease (COPD) showed highly positive results, according to findings presented at the annual meeting of the American College of Chest Physicians (CHEST).
At the end of 12 weeks, those randomly assigned to the intervention had a significant and clinically meaningful improvement in all domains of the Chronic Respiratory Questionnaire (CRQ), including activity levels and emotional well-being, reported Roberto P. Benzo, MD, a consultant in the division of pulmonary and critical care medicine, Mayo Clinic, Rochester, Minn.
Presenting soon-to-be-published data, Dr. Benzo said that the intervention is based on a tablet-based app. The tablet is programmed to upload data captured from an activity monitor and pulse oximeter. Along with documentation of app usage, this information can then be downloaded for the remote coach to review with the patient.
The primary outcome of the randomized study were the physical and emotional domains of the CRQ quality of life, but a long list of secondary outcomes – including physical activity, symptoms of depression, sleep quality, and health care utilization, such as emergency room visits – was also analyzed.
In addition to the significant benefit on the primary outcomes, the home-based rehabilitation program relative to a wait list for intervention was associated with benefit or a trend for benefit on essentially every outcome measured. Health care utilization was a possible exception, but even then, the absolute number of visits was lower in the treatment arm.
“With a study period of only 12 weeks, we were limited to our ability to show a difference in emergency room visits,” said Dr. Benzo, who also noted that the study was conducted during the COVID-19 pandemic, when hospital visits were already occurring at a lower than usual rate. Based on the other findings, he suspects that a reduction in health care utilization could also be shown in more typical circumstances, particularly with a longer follow-up.
In the study, 375 patients with COPD were randomly assigned to a home health care regimen delivered by an app with remote coaching or to a wait list and usual care. The median age was 69 years. Fifty-nine percent were women. The median FEV1 at enrollment was 45% of predicted.
The patients were able to access their own data to monitor their progress at any time, not just at the time of coaching, but contact with the remote coach occurred on a weekly basis. Patients rated their level of energy, how they felt generally, and their progress toward daily goals, which was also captured on the app and could be discussed with the coach during the review of the previous week’s activity.
At 12 weeks, the favorable 0.54-point change (P < .001) and 0.51 change (P < .001) in the physical and emotional summary scores, respectively, met the criteria for a clinically meaningful change, Dr. Benzo reported. There were also significantly favorable changes from baseline and relative to controls in CRQ domains of self-management, sleep quality, and depression (all P ≤ .01).
Other data collected are supportive. For example, Dr. Benzo reported that those in the rehabilitation group took 624 more steps on average per day than those in the control group. The experimental group also spent nearly an hour more performing moderate or greater levels of activity.
“The app promotes behavioral change,” said Dr. Benzo, who said that this “completely home-based model” of rehabilitation is likely to be cost-effective given the relatively low costs of remote coaching and reasonable costs of the activity monitor, tablet, and other equipment.
Importantly, home-based rehabilitation is a billable practice under currently available CPT codes, according to Dr. Benzo, who believes this approach is not only effective but “feasible and practical.”
Two clinicians active in the care of patients with COPD believe this approach could fulfill an unmet need if further validated. Andrew Berman, MD, professor of medicine, New Jersey Medical School, Newark, thinks the premise is sound.
“Digital competency is still a big issue as is access to adequate quality Internet, but this could be a very useful approach for many individuals, and it avoids visits to a center, which could be a big advantage for patients,” Dr. Berman said.
Abebaw M. Johannes, PhD, a professor of physical therapy at Azusa Pacific University, Azusa, Calif., agreed. He said that home-based remote coaching could be a way of overcoming the current hurdles of participating in institutional-based programs
“This is clearly an unmet need in COPD,” he said.
The development of more effective and patient-friendly programs is what was driving this research, according to Dr. Benzo. He cited data suggesting that only about 30% of patients with COPD are participating in rehabilitation programs once discharged from the hospital despite the evidence that they can improve quality of life. For many of these patients, a home-based program might be the answer.
Dr. Benzo, Dr. Berman, and Dr. Johannes reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
NASHVILLE – The first multicenter randomized controlled trial of a home-based rehabilitation program for patients with chronic obstructive pulmonary disease (COPD) showed highly positive results, according to findings presented at the annual meeting of the American College of Chest Physicians (CHEST).
At the end of 12 weeks, those randomly assigned to the intervention had a significant and clinically meaningful improvement in all domains of the Chronic Respiratory Questionnaire (CRQ), including activity levels and emotional well-being, reported Roberto P. Benzo, MD, a consultant in the division of pulmonary and critical care medicine, Mayo Clinic, Rochester, Minn.
Presenting soon-to-be-published data, Dr. Benzo said that the intervention is based on a tablet-based app. The tablet is programmed to upload data captured from an activity monitor and pulse oximeter. Along with documentation of app usage, this information can then be downloaded for the remote coach to review with the patient.
The primary outcome of the randomized study were the physical and emotional domains of the CRQ quality of life, but a long list of secondary outcomes – including physical activity, symptoms of depression, sleep quality, and health care utilization, such as emergency room visits – was also analyzed.
In addition to the significant benefit on the primary outcomes, the home-based rehabilitation program relative to a wait list for intervention was associated with benefit or a trend for benefit on essentially every outcome measured. Health care utilization was a possible exception, but even then, the absolute number of visits was lower in the treatment arm.
“With a study period of only 12 weeks, we were limited to our ability to show a difference in emergency room visits,” said Dr. Benzo, who also noted that the study was conducted during the COVID-19 pandemic, when hospital visits were already occurring at a lower than usual rate. Based on the other findings, he suspects that a reduction in health care utilization could also be shown in more typical circumstances, particularly with a longer follow-up.
In the study, 375 patients with COPD were randomly assigned to a home health care regimen delivered by an app with remote coaching or to a wait list and usual care. The median age was 69 years. Fifty-nine percent were women. The median FEV1 at enrollment was 45% of predicted.
The patients were able to access their own data to monitor their progress at any time, not just at the time of coaching, but contact with the remote coach occurred on a weekly basis. Patients rated their level of energy, how they felt generally, and their progress toward daily goals, which was also captured on the app and could be discussed with the coach during the review of the previous week’s activity.
At 12 weeks, the favorable 0.54-point change (P < .001) and 0.51 change (P < .001) in the physical and emotional summary scores, respectively, met the criteria for a clinically meaningful change, Dr. Benzo reported. There were also significantly favorable changes from baseline and relative to controls in CRQ domains of self-management, sleep quality, and depression (all P ≤ .01).
Other data collected are supportive. For example, Dr. Benzo reported that those in the rehabilitation group took 624 more steps on average per day than those in the control group. The experimental group also spent nearly an hour more performing moderate or greater levels of activity.
“The app promotes behavioral change,” said Dr. Benzo, who said that this “completely home-based model” of rehabilitation is likely to be cost-effective given the relatively low costs of remote coaching and reasonable costs of the activity monitor, tablet, and other equipment.
Importantly, home-based rehabilitation is a billable practice under currently available CPT codes, according to Dr. Benzo, who believes this approach is not only effective but “feasible and practical.”
Two clinicians active in the care of patients with COPD believe this approach could fulfill an unmet need if further validated. Andrew Berman, MD, professor of medicine, New Jersey Medical School, Newark, thinks the premise is sound.
“Digital competency is still a big issue as is access to adequate quality Internet, but this could be a very useful approach for many individuals, and it avoids visits to a center, which could be a big advantage for patients,” Dr. Berman said.
Abebaw M. Johannes, PhD, a professor of physical therapy at Azusa Pacific University, Azusa, Calif., agreed. He said that home-based remote coaching could be a way of overcoming the current hurdles of participating in institutional-based programs
“This is clearly an unmet need in COPD,” he said.
The development of more effective and patient-friendly programs is what was driving this research, according to Dr. Benzo. He cited data suggesting that only about 30% of patients with COPD are participating in rehabilitation programs once discharged from the hospital despite the evidence that they can improve quality of life. For many of these patients, a home-based program might be the answer.
Dr. Benzo, Dr. Berman, and Dr. Johannes reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
NASHVILLE – The first multicenter randomized controlled trial of a home-based rehabilitation program for patients with chronic obstructive pulmonary disease (COPD) showed highly positive results, according to findings presented at the annual meeting of the American College of Chest Physicians (CHEST).
At the end of 12 weeks, those randomly assigned to the intervention had a significant and clinically meaningful improvement in all domains of the Chronic Respiratory Questionnaire (CRQ), including activity levels and emotional well-being, reported Roberto P. Benzo, MD, a consultant in the division of pulmonary and critical care medicine, Mayo Clinic, Rochester, Minn.
Presenting soon-to-be-published data, Dr. Benzo said that the intervention is based on a tablet-based app. The tablet is programmed to upload data captured from an activity monitor and pulse oximeter. Along with documentation of app usage, this information can then be downloaded for the remote coach to review with the patient.
The primary outcome of the randomized study were the physical and emotional domains of the CRQ quality of life, but a long list of secondary outcomes – including physical activity, symptoms of depression, sleep quality, and health care utilization, such as emergency room visits – was also analyzed.
In addition to the significant benefit on the primary outcomes, the home-based rehabilitation program relative to a wait list for intervention was associated with benefit or a trend for benefit on essentially every outcome measured. Health care utilization was a possible exception, but even then, the absolute number of visits was lower in the treatment arm.
“With a study period of only 12 weeks, we were limited to our ability to show a difference in emergency room visits,” said Dr. Benzo, who also noted that the study was conducted during the COVID-19 pandemic, when hospital visits were already occurring at a lower than usual rate. Based on the other findings, he suspects that a reduction in health care utilization could also be shown in more typical circumstances, particularly with a longer follow-up.
In the study, 375 patients with COPD were randomly assigned to a home health care regimen delivered by an app with remote coaching or to a wait list and usual care. The median age was 69 years. Fifty-nine percent were women. The median FEV1 at enrollment was 45% of predicted.
The patients were able to access their own data to monitor their progress at any time, not just at the time of coaching, but contact with the remote coach occurred on a weekly basis. Patients rated their level of energy, how they felt generally, and their progress toward daily goals, which was also captured on the app and could be discussed with the coach during the review of the previous week’s activity.
At 12 weeks, the favorable 0.54-point change (P < .001) and 0.51 change (P < .001) in the physical and emotional summary scores, respectively, met the criteria for a clinically meaningful change, Dr. Benzo reported. There were also significantly favorable changes from baseline and relative to controls in CRQ domains of self-management, sleep quality, and depression (all P ≤ .01).
Other data collected are supportive. For example, Dr. Benzo reported that those in the rehabilitation group took 624 more steps on average per day than those in the control group. The experimental group also spent nearly an hour more performing moderate or greater levels of activity.
“The app promotes behavioral change,” said Dr. Benzo, who said that this “completely home-based model” of rehabilitation is likely to be cost-effective given the relatively low costs of remote coaching and reasonable costs of the activity monitor, tablet, and other equipment.
Importantly, home-based rehabilitation is a billable practice under currently available CPT codes, according to Dr. Benzo, who believes this approach is not only effective but “feasible and practical.”
Two clinicians active in the care of patients with COPD believe this approach could fulfill an unmet need if further validated. Andrew Berman, MD, professor of medicine, New Jersey Medical School, Newark, thinks the premise is sound.
“Digital competency is still a big issue as is access to adequate quality Internet, but this could be a very useful approach for many individuals, and it avoids visits to a center, which could be a big advantage for patients,” Dr. Berman said.
Abebaw M. Johannes, PhD, a professor of physical therapy at Azusa Pacific University, Azusa, Calif., agreed. He said that home-based remote coaching could be a way of overcoming the current hurdles of participating in institutional-based programs
“This is clearly an unmet need in COPD,” he said.
The development of more effective and patient-friendly programs is what was driving this research, according to Dr. Benzo. He cited data suggesting that only about 30% of patients with COPD are participating in rehabilitation programs once discharged from the hospital despite the evidence that they can improve quality of life. For many of these patients, a home-based program might be the answer.
Dr. Benzo, Dr. Berman, and Dr. Johannes reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM CHEST 2022
FMT in IBS: ‘We’ve been targeting the wrong part of the intestine’
VIENNA – , vs. it being administered into the large intestine, according to a new study.
Patients also reported an improvement in symptoms and quality of life with repeated doses of FMT (two doses, given 1 week apart), compared with a single dose in the small intestine, although statistical significance was not met.
“Administering a fecal transplant to the small intestine leads to long-term – up to 1 year in this analysis – colonization of beneficial bacteria, whereas administrating the fecal transplant to the large intestine results in the effect only lasting for the first 3 months,” said Magdy El-Salhy, MD, from the University of Bergen, Norway.
Dr. El-Salhy presented the results at the annual United European Gastroenterology Week meeting.
“It seems that bacteria in the small intestine play a more central role in IBS, as well as its associated fatigue, than bacteria in the large intestine,” Dr. El-Salhy said in an interview.
“Until now, we’ve been targeting the wrong part of the intestine,” he said.
The findings are the first to show that the small intestine is a more effective location for administering FMT than the large intestine for IBS. “It would be worthwhile doing similar [studies] in other diseases, especially in inflammatory bowel diseases,” said Dr. El-Salhy.
Researchers also didn’t expect the repeated dose to improve symptoms for a longer duration. “It really was revolutionary to see,” he added.
Some of Dr. El-Salhy’s patients have had up to 5 years of follow-up, although these results were not presented at this year’s UEG, he said.
“Around 75% of my patients have shown duration of response up to 3 years, and a few up to 5 years, on a 60-g dose from an earlier study group,” he said. “It’s an incredible result after a 10-minute treatment.”
In Dr. El-Salhy’s previous work, he found that increasing the dose from 30 g to 60 g increased the response from about 75% to about 90%. However, in this study presented, he found that increasing the dose to 90 g did not further increase the response. He also noted that while repeating the FMT dose improved symptoms and quality of life more than a single transplantation, it did not increase the response.
Targeting the small intestine
FMT has been widely investigated for the treatment of such conditions as psoriatic arthritis, Clostridioides difficile infection, and ulcerative colitis.
In this study, Dr. El-Salhy built on prior work (seven randomized controlled studies with varied outcomes) by asking whether the transplant dose increases FMT efficacy, which route of administration is more effective, and whether repeating FMT increases efficacy in patients with IBS.
A total of 186 patients were randomized to one of three groups: 90 g of frozen transplant into the large intestine (n = 62), 90 g of frozen transplant into the small intestine (n = 62), or 90 g of frozen transplant into the small intestine twice (with a 1-week interval; n = 62). FMT was administered via nasoduodenal tube and colonoscopy into the small and large intestines, respectively.
Outcomes were measured at 3, 6, and 12 months. The 12-month analysis of outcomes via patient questionnaire included 60, 61, and 60 patients, respectively.
The patient questionnaires included in the study were the IBS-SSS (a composite score of abdominal pain, duration of abdominal pain, bloating/distention, satisfaction with bowel habits, and IBS-related quality of life), the Birmingham IBS Symptom questionnaire, the Fatigue Assessment Scale questionnaire, the IBS-Quality of Life assessment, and the Short-Form Nepean Dyspepsia Index.
Fecal samples were taken and tested for bacterial loads. The bacterial profile and dysbiosis index were determined using the 16S rRNA gene.
At 3 months, patients had similar response rates, around 80%, across single dose in large intestine, single dose in small intestine, and repeat doses in small intestine.
At 6 months, the differences in response rates started to become noticeable, with 67.9% for single dose in large intestine, 71.4% for single dose in small intestine, and 86% for repeat doses in small intestine.
By 12 months, the difference in response rate between the single dose in the large and small intestines was statistically significant at 51.9% and 75.5%, respectively. The response rate to the repeat doses in the small intestine at 12 months (80.9%) was similar to that at 3 months (80.8%).
Side effects, including mild abdominal pain, diarrhea, and constipation, after FMT were seen for the first 5 days after treatment. “People who generally suffer from constipation get diarrhea after FMT and vice versa,” Dr. El-Salhy reported.
“Long-term side effects, as monitored up to 3 years, were not observed,” he added.
Treatment reduced IBS symptoms in all patient groups as measured by IBS-SSS scores. By 12 months, the score fell from around 350 to around 220 in patients who received a single dose in the large intestine, from around 300 to around 200 in patients who received a single dose in the small intestine, and from around 350 to around 170 in patients who received repeat doses in the small intestine.
Quality of life showed a statistically significant difference at 3 months between single and repeated doses in the small intestine and similarly at 6 and 12 months.
Chronic fatigue, experienced by many patients with IBS, was substantially reduced after FMT, Dr. El-Salhy noted. “This surge in energy is often more important to them than the gastrointestinal symptoms.”
Location affects bacterial success
Certain beneficial bacteria were found to thrive more when the donor transplant was administered to the small intestine than to the large intestine.
Of note, Lactobacillus species and Holdemanella biformis grew and then dropped off sharply after 3 months in patients who received a single-dose fecal transplant in the large intestine, while they grew after 3 months and continued to grow after 6 and 12 months in the groups who received a fecal transplant in the small intestine.
“We think bacteria in the small intestine have different characteristics to those in the large intestine,” Dr. El-Salhy said. “This is relatively new, because many years ago it was thought that bile acids prevented bacterial survival. Now we know lots can thrive in the small intestine.”
“It might be viral or some other component that is most effective here. We don’t know yet, but so far we have identified 11 bacteria of interest,” he added.
Broader questions
“Rather than focusing on a specific, single strain microbe as a predictor of success in a disease, the global equilibrium of microbiota is more important, and microbial ecology parameters would be interesting to assess,” remarked Gianluca Ianiro, MD, from the Università Cattolica del Sacro Cuore, Rome, who comoderated the session. “Selected survival of some bacteria through the gut may be the response.”
FMT emerged in response to the challenges posed by recurrent C. difficile infections, noted Alexander Khoruts, MD, a professor of medicine in the division of gastroenterology, hepatology, and nutrition at the University of Minnesota, Minneapolis, who was not involved in the research.
“It is much harder to achieve remodeling of the gut microbiome in non–C. difficile conditions where there is an intact and resilient indigenous microbiota,” he said in an interview. “Therefore, regimens using antibiotic preconditioning and repeated administrations of microbiota are generally more efficacious in achieving this objective.”
The specificity of the bacteria according to disease type targeted was important, said Dr. Khoruts, who has a special interest in gut microbiota.
“The big question in non–C. difficile indications is the composition of donor microbiota. It is critical that we understand the mechanisms involved in each target disease to design appropriate microbiota-based therapeutics,” he said.
Dr. Khoruts sounded a note of caution with respect to establishing the pharmacokinetic and dynamic data related to FMT, which is classified as a drug in the United States.
“It’s imperative that we develop the pharmacology discipline appropriate for this class of therapeutics, including their pharmacokinetics and pharmacodynamics, and an understanding of their potential toxicity and drug-drug interactions,” he said.
Drug distribution data are needed to determine host-microbiota interactions.
“This includes the small bowel microbiome, which continues to be woefully understudied,” Dr. Khoruts said.
Dr. El-Salhy reports no relevant financial relationships. Dr. Ianiro reports receiving personal fees for acting as speaker for Biocodex, Sofar, Malesci, and Tillotts Pharma, and for acting as consultant/advisor for Ferring Therapeutics, Biocodex, Tillotts Pharma, and Zambon. Dr. Khoruts reports he has patents pertaining to fecal microbiota separation from stool and their cryopreservation and lyopreservation.
A version of this article first appeared on Medscape.com.
VIENNA – , vs. it being administered into the large intestine, according to a new study.
Patients also reported an improvement in symptoms and quality of life with repeated doses of FMT (two doses, given 1 week apart), compared with a single dose in the small intestine, although statistical significance was not met.
“Administering a fecal transplant to the small intestine leads to long-term – up to 1 year in this analysis – colonization of beneficial bacteria, whereas administrating the fecal transplant to the large intestine results in the effect only lasting for the first 3 months,” said Magdy El-Salhy, MD, from the University of Bergen, Norway.
Dr. El-Salhy presented the results at the annual United European Gastroenterology Week meeting.
“It seems that bacteria in the small intestine play a more central role in IBS, as well as its associated fatigue, than bacteria in the large intestine,” Dr. El-Salhy said in an interview.
“Until now, we’ve been targeting the wrong part of the intestine,” he said.
The findings are the first to show that the small intestine is a more effective location for administering FMT than the large intestine for IBS. “It would be worthwhile doing similar [studies] in other diseases, especially in inflammatory bowel diseases,” said Dr. El-Salhy.
Researchers also didn’t expect the repeated dose to improve symptoms for a longer duration. “It really was revolutionary to see,” he added.
Some of Dr. El-Salhy’s patients have had up to 5 years of follow-up, although these results were not presented at this year’s UEG, he said.
“Around 75% of my patients have shown duration of response up to 3 years, and a few up to 5 years, on a 60-g dose from an earlier study group,” he said. “It’s an incredible result after a 10-minute treatment.”
In Dr. El-Salhy’s previous work, he found that increasing the dose from 30 g to 60 g increased the response from about 75% to about 90%. However, in this study presented, he found that increasing the dose to 90 g did not further increase the response. He also noted that while repeating the FMT dose improved symptoms and quality of life more than a single transplantation, it did not increase the response.
Targeting the small intestine
FMT has been widely investigated for the treatment of such conditions as psoriatic arthritis, Clostridioides difficile infection, and ulcerative colitis.
In this study, Dr. El-Salhy built on prior work (seven randomized controlled studies with varied outcomes) by asking whether the transplant dose increases FMT efficacy, which route of administration is more effective, and whether repeating FMT increases efficacy in patients with IBS.
A total of 186 patients were randomized to one of three groups: 90 g of frozen transplant into the large intestine (n = 62), 90 g of frozen transplant into the small intestine (n = 62), or 90 g of frozen transplant into the small intestine twice (with a 1-week interval; n = 62). FMT was administered via nasoduodenal tube and colonoscopy into the small and large intestines, respectively.
Outcomes were measured at 3, 6, and 12 months. The 12-month analysis of outcomes via patient questionnaire included 60, 61, and 60 patients, respectively.
The patient questionnaires included in the study were the IBS-SSS (a composite score of abdominal pain, duration of abdominal pain, bloating/distention, satisfaction with bowel habits, and IBS-related quality of life), the Birmingham IBS Symptom questionnaire, the Fatigue Assessment Scale questionnaire, the IBS-Quality of Life assessment, and the Short-Form Nepean Dyspepsia Index.
Fecal samples were taken and tested for bacterial loads. The bacterial profile and dysbiosis index were determined using the 16S rRNA gene.
At 3 months, patients had similar response rates, around 80%, across single dose in large intestine, single dose in small intestine, and repeat doses in small intestine.
At 6 months, the differences in response rates started to become noticeable, with 67.9% for single dose in large intestine, 71.4% for single dose in small intestine, and 86% for repeat doses in small intestine.
By 12 months, the difference in response rate between the single dose in the large and small intestines was statistically significant at 51.9% and 75.5%, respectively. The response rate to the repeat doses in the small intestine at 12 months (80.9%) was similar to that at 3 months (80.8%).
Side effects, including mild abdominal pain, diarrhea, and constipation, after FMT were seen for the first 5 days after treatment. “People who generally suffer from constipation get diarrhea after FMT and vice versa,” Dr. El-Salhy reported.
“Long-term side effects, as monitored up to 3 years, were not observed,” he added.
Treatment reduced IBS symptoms in all patient groups as measured by IBS-SSS scores. By 12 months, the score fell from around 350 to around 220 in patients who received a single dose in the large intestine, from around 300 to around 200 in patients who received a single dose in the small intestine, and from around 350 to around 170 in patients who received repeat doses in the small intestine.
Quality of life showed a statistically significant difference at 3 months between single and repeated doses in the small intestine and similarly at 6 and 12 months.
Chronic fatigue, experienced by many patients with IBS, was substantially reduced after FMT, Dr. El-Salhy noted. “This surge in energy is often more important to them than the gastrointestinal symptoms.”
Location affects bacterial success
Certain beneficial bacteria were found to thrive more when the donor transplant was administered to the small intestine than to the large intestine.
Of note, Lactobacillus species and Holdemanella biformis grew and then dropped off sharply after 3 months in patients who received a single-dose fecal transplant in the large intestine, while they grew after 3 months and continued to grow after 6 and 12 months in the groups who received a fecal transplant in the small intestine.
“We think bacteria in the small intestine have different characteristics to those in the large intestine,” Dr. El-Salhy said. “This is relatively new, because many years ago it was thought that bile acids prevented bacterial survival. Now we know lots can thrive in the small intestine.”
“It might be viral or some other component that is most effective here. We don’t know yet, but so far we have identified 11 bacteria of interest,” he added.
Broader questions
“Rather than focusing on a specific, single strain microbe as a predictor of success in a disease, the global equilibrium of microbiota is more important, and microbial ecology parameters would be interesting to assess,” remarked Gianluca Ianiro, MD, from the Università Cattolica del Sacro Cuore, Rome, who comoderated the session. “Selected survival of some bacteria through the gut may be the response.”
FMT emerged in response to the challenges posed by recurrent C. difficile infections, noted Alexander Khoruts, MD, a professor of medicine in the division of gastroenterology, hepatology, and nutrition at the University of Minnesota, Minneapolis, who was not involved in the research.
“It is much harder to achieve remodeling of the gut microbiome in non–C. difficile conditions where there is an intact and resilient indigenous microbiota,” he said in an interview. “Therefore, regimens using antibiotic preconditioning and repeated administrations of microbiota are generally more efficacious in achieving this objective.”
The specificity of the bacteria according to disease type targeted was important, said Dr. Khoruts, who has a special interest in gut microbiota.
“The big question in non–C. difficile indications is the composition of donor microbiota. It is critical that we understand the mechanisms involved in each target disease to design appropriate microbiota-based therapeutics,” he said.
Dr. Khoruts sounded a note of caution with respect to establishing the pharmacokinetic and dynamic data related to FMT, which is classified as a drug in the United States.
“It’s imperative that we develop the pharmacology discipline appropriate for this class of therapeutics, including their pharmacokinetics and pharmacodynamics, and an understanding of their potential toxicity and drug-drug interactions,” he said.
Drug distribution data are needed to determine host-microbiota interactions.
“This includes the small bowel microbiome, which continues to be woefully understudied,” Dr. Khoruts said.
Dr. El-Salhy reports no relevant financial relationships. Dr. Ianiro reports receiving personal fees for acting as speaker for Biocodex, Sofar, Malesci, and Tillotts Pharma, and for acting as consultant/advisor for Ferring Therapeutics, Biocodex, Tillotts Pharma, and Zambon. Dr. Khoruts reports he has patents pertaining to fecal microbiota separation from stool and their cryopreservation and lyopreservation.
A version of this article first appeared on Medscape.com.
VIENNA – , vs. it being administered into the large intestine, according to a new study.
Patients also reported an improvement in symptoms and quality of life with repeated doses of FMT (two doses, given 1 week apart), compared with a single dose in the small intestine, although statistical significance was not met.
“Administering a fecal transplant to the small intestine leads to long-term – up to 1 year in this analysis – colonization of beneficial bacteria, whereas administrating the fecal transplant to the large intestine results in the effect only lasting for the first 3 months,” said Magdy El-Salhy, MD, from the University of Bergen, Norway.
Dr. El-Salhy presented the results at the annual United European Gastroenterology Week meeting.
“It seems that bacteria in the small intestine play a more central role in IBS, as well as its associated fatigue, than bacteria in the large intestine,” Dr. El-Salhy said in an interview.
“Until now, we’ve been targeting the wrong part of the intestine,” he said.
The findings are the first to show that the small intestine is a more effective location for administering FMT than the large intestine for IBS. “It would be worthwhile doing similar [studies] in other diseases, especially in inflammatory bowel diseases,” said Dr. El-Salhy.
Researchers also didn’t expect the repeated dose to improve symptoms for a longer duration. “It really was revolutionary to see,” he added.
Some of Dr. El-Salhy’s patients have had up to 5 years of follow-up, although these results were not presented at this year’s UEG, he said.
“Around 75% of my patients have shown duration of response up to 3 years, and a few up to 5 years, on a 60-g dose from an earlier study group,” he said. “It’s an incredible result after a 10-minute treatment.”
In Dr. El-Salhy’s previous work, he found that increasing the dose from 30 g to 60 g increased the response from about 75% to about 90%. However, in this study presented, he found that increasing the dose to 90 g did not further increase the response. He also noted that while repeating the FMT dose improved symptoms and quality of life more than a single transplantation, it did not increase the response.
Targeting the small intestine
FMT has been widely investigated for the treatment of such conditions as psoriatic arthritis, Clostridioides difficile infection, and ulcerative colitis.
In this study, Dr. El-Salhy built on prior work (seven randomized controlled studies with varied outcomes) by asking whether the transplant dose increases FMT efficacy, which route of administration is more effective, and whether repeating FMT increases efficacy in patients with IBS.
A total of 186 patients were randomized to one of three groups: 90 g of frozen transplant into the large intestine (n = 62), 90 g of frozen transplant into the small intestine (n = 62), or 90 g of frozen transplant into the small intestine twice (with a 1-week interval; n = 62). FMT was administered via nasoduodenal tube and colonoscopy into the small and large intestines, respectively.
Outcomes were measured at 3, 6, and 12 months. The 12-month analysis of outcomes via patient questionnaire included 60, 61, and 60 patients, respectively.
The patient questionnaires included in the study were the IBS-SSS (a composite score of abdominal pain, duration of abdominal pain, bloating/distention, satisfaction with bowel habits, and IBS-related quality of life), the Birmingham IBS Symptom questionnaire, the Fatigue Assessment Scale questionnaire, the IBS-Quality of Life assessment, and the Short-Form Nepean Dyspepsia Index.
Fecal samples were taken and tested for bacterial loads. The bacterial profile and dysbiosis index were determined using the 16S rRNA gene.
At 3 months, patients had similar response rates, around 80%, across single dose in large intestine, single dose in small intestine, and repeat doses in small intestine.
At 6 months, the differences in response rates started to become noticeable, with 67.9% for single dose in large intestine, 71.4% for single dose in small intestine, and 86% for repeat doses in small intestine.
By 12 months, the difference in response rate between the single dose in the large and small intestines was statistically significant at 51.9% and 75.5%, respectively. The response rate to the repeat doses in the small intestine at 12 months (80.9%) was similar to that at 3 months (80.8%).
Side effects, including mild abdominal pain, diarrhea, and constipation, after FMT were seen for the first 5 days after treatment. “People who generally suffer from constipation get diarrhea after FMT and vice versa,” Dr. El-Salhy reported.
“Long-term side effects, as monitored up to 3 years, were not observed,” he added.
Treatment reduced IBS symptoms in all patient groups as measured by IBS-SSS scores. By 12 months, the score fell from around 350 to around 220 in patients who received a single dose in the large intestine, from around 300 to around 200 in patients who received a single dose in the small intestine, and from around 350 to around 170 in patients who received repeat doses in the small intestine.
Quality of life showed a statistically significant difference at 3 months between single and repeated doses in the small intestine and similarly at 6 and 12 months.
Chronic fatigue, experienced by many patients with IBS, was substantially reduced after FMT, Dr. El-Salhy noted. “This surge in energy is often more important to them than the gastrointestinal symptoms.”
Location affects bacterial success
Certain beneficial bacteria were found to thrive more when the donor transplant was administered to the small intestine than to the large intestine.
Of note, Lactobacillus species and Holdemanella biformis grew and then dropped off sharply after 3 months in patients who received a single-dose fecal transplant in the large intestine, while they grew after 3 months and continued to grow after 6 and 12 months in the groups who received a fecal transplant in the small intestine.
“We think bacteria in the small intestine have different characteristics to those in the large intestine,” Dr. El-Salhy said. “This is relatively new, because many years ago it was thought that bile acids prevented bacterial survival. Now we know lots can thrive in the small intestine.”
“It might be viral or some other component that is most effective here. We don’t know yet, but so far we have identified 11 bacteria of interest,” he added.
Broader questions
“Rather than focusing on a specific, single strain microbe as a predictor of success in a disease, the global equilibrium of microbiota is more important, and microbial ecology parameters would be interesting to assess,” remarked Gianluca Ianiro, MD, from the Università Cattolica del Sacro Cuore, Rome, who comoderated the session. “Selected survival of some bacteria through the gut may be the response.”
FMT emerged in response to the challenges posed by recurrent C. difficile infections, noted Alexander Khoruts, MD, a professor of medicine in the division of gastroenterology, hepatology, and nutrition at the University of Minnesota, Minneapolis, who was not involved in the research.
“It is much harder to achieve remodeling of the gut microbiome in non–C. difficile conditions where there is an intact and resilient indigenous microbiota,” he said in an interview. “Therefore, regimens using antibiotic preconditioning and repeated administrations of microbiota are generally more efficacious in achieving this objective.”
The specificity of the bacteria according to disease type targeted was important, said Dr. Khoruts, who has a special interest in gut microbiota.
“The big question in non–C. difficile indications is the composition of donor microbiota. It is critical that we understand the mechanisms involved in each target disease to design appropriate microbiota-based therapeutics,” he said.
Dr. Khoruts sounded a note of caution with respect to establishing the pharmacokinetic and dynamic data related to FMT, which is classified as a drug in the United States.
“It’s imperative that we develop the pharmacology discipline appropriate for this class of therapeutics, including their pharmacokinetics and pharmacodynamics, and an understanding of their potential toxicity and drug-drug interactions,” he said.
Drug distribution data are needed to determine host-microbiota interactions.
“This includes the small bowel microbiome, which continues to be woefully understudied,” Dr. Khoruts said.
Dr. El-Salhy reports no relevant financial relationships. Dr. Ianiro reports receiving personal fees for acting as speaker for Biocodex, Sofar, Malesci, and Tillotts Pharma, and for acting as consultant/advisor for Ferring Therapeutics, Biocodex, Tillotts Pharma, and Zambon. Dr. Khoruts reports he has patents pertaining to fecal microbiota separation from stool and their cryopreservation and lyopreservation.
A version of this article first appeared on Medscape.com.
AT UEG WEEK 2022