User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
nav[contains(@class, 'nav-ce-stack nav-ce-stack__large-screen')]
header[@id='header']
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'main-prefix')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
div[contains(@class, 'view-medstat-quiz-listing-panes')]
div[contains(@class, 'pane-article-sidebar-latest-news')]
CDC says 44% of people hospitalized with COVID had third dose or booster
, the Centers for Disease Control and Prevention says.
Unvaccinated adults were 3.4 times more likely to be hospitalized with COVID than those who were vaccinated, the CDC said.
The CDC report considered hospitalization numbers from March 20 to May 31, when the omicron subvariant BA.2 was the dominant strain. Researchers found 39.1% of patients had received a primary vaccination series and at least one booster or additional dose; 5% were fully vaccinated with two boosters.
“Adults should stay up to date with COVID-19 vaccination, including booster doses,” the CDC said. “Multiple nonpharmaceutical and medical prevention measures should be used to protect persons at high risk for severe SARS-CoV-2, regardless of vaccination status.”
Older adults and people with underlying medical conditions who become infected with the coronavirus are more likely to be hospitalized.
The study also found that hospitalization rates among people over 65 increased threefold over the study period. Rates among people under 65 rose 1.7 times.
A version of this article first appeared on WebMD.com.
, the Centers for Disease Control and Prevention says.
Unvaccinated adults were 3.4 times more likely to be hospitalized with COVID than those who were vaccinated, the CDC said.
The CDC report considered hospitalization numbers from March 20 to May 31, when the omicron subvariant BA.2 was the dominant strain. Researchers found 39.1% of patients had received a primary vaccination series and at least one booster or additional dose; 5% were fully vaccinated with two boosters.
“Adults should stay up to date with COVID-19 vaccination, including booster doses,” the CDC said. “Multiple nonpharmaceutical and medical prevention measures should be used to protect persons at high risk for severe SARS-CoV-2, regardless of vaccination status.”
Older adults and people with underlying medical conditions who become infected with the coronavirus are more likely to be hospitalized.
The study also found that hospitalization rates among people over 65 increased threefold over the study period. Rates among people under 65 rose 1.7 times.
A version of this article first appeared on WebMD.com.
, the Centers for Disease Control and Prevention says.
Unvaccinated adults were 3.4 times more likely to be hospitalized with COVID than those who were vaccinated, the CDC said.
The CDC report considered hospitalization numbers from March 20 to May 31, when the omicron subvariant BA.2 was the dominant strain. Researchers found 39.1% of patients had received a primary vaccination series and at least one booster or additional dose; 5% were fully vaccinated with two boosters.
“Adults should stay up to date with COVID-19 vaccination, including booster doses,” the CDC said. “Multiple nonpharmaceutical and medical prevention measures should be used to protect persons at high risk for severe SARS-CoV-2, regardless of vaccination status.”
Older adults and people with underlying medical conditions who become infected with the coronavirus are more likely to be hospitalized.
The study also found that hospitalization rates among people over 65 increased threefold over the study period. Rates among people under 65 rose 1.7 times.
A version of this article first appeared on WebMD.com.
FROM MMWR
After index colonoscopy, what’s the CRC risk in 40- to 49-year-olds vs. 50- to 59-year-olds?
New data suggest that for individuals who do not have an adenoma detected on an index colonoscopy, the risk of developing an advanced neoplasia (AN) and colorectal cancer (CRC) is lower in those who are aged 40-49 years, compared with those who are 50-59 years old.
However, there is no difference between the two age groups in detection rates of nonadvanced adenoma (NAA) or advanced adenoma (AA), the same study found.
“The primary goal of this study was to investigate the risk of metachronous AN associated with conventional adenoma detected on the index colonoscopy,” explain the authors, led by Gene Ma, MD, Kaiser Permanente Northern California, San Jose.
“The lack of good-quality evidence to inform surveillance in the 40-49 year old population has resulted in inconsistent surveillance patterns in clinical practice, leading to variation in the quality of care, including both inadequate and excessive colonoscopic surveillance,” Dr. Ma and colleagues observe.
The findings from this study “expand our understanding of the risk of AN and CRC in younger individuals and suggest that the current multi-society guidelines for surveillance may be applicable for individuals 40-49 years of age,” the authors conclude.
The study was published online in The American Journal of Gastroenterology, and included 2,396 individuals between 40 and 49 years of age and 8,978 individuals between 50 and 59 years of age.
The colonoscopy was carried out for screening in 40.2% in the younger age group versus 34.8% in the older age group and was prompted by a positive fecal immunochemical test in 3.3% of the younger age group versus 32% of the older age group.
The median follow-up for both age groups was roughly 7 years.
“When comparing the 40-49 years group to the 50-59 years group, index colonoscopy detected no adenoma in 62.9% versus 40.1% (P < .0001); NAA in 25.4% versus 39.0% (P <.001), and AA in 11.6% versus 21.0% (P < .0001), respectively,” Dr. Ma and colleagues report.
When the two age groups were compared for surveillance colonoscopy, no adenoma was detected in 67% of the younger age group versus 54.7% of the older age group (P < .0001), whereas NAA was detected in 25.4% of the 40- to 49-year-olds versus 38.4% of the 50- to 59-year-olds (P < .0001). AA was detected in 3.5% versus 6.95 (P < .0001) of persons in each of the two age groups, respectively.
AN was detected on surveillance colonoscopy after index colonoscopy in 2.2% of the younger age group and twice that percentage, at 4.4%, in the older age group (P = .0003). On surveillance colonoscopy, NAA was found in 4.6% of the younger age group, compared with 7% of the older age group (P = .03), whereas AA was found in 7.9% of the 40- to 49-year-olds, compared with 11.7% of the 50- to 59-year-olds (P = .06).
The median time until surveillance colonoscopy was similar in both age groups when either NAA or AA was found on index colonoscopy, the authors note. In addition, the median time until the detection of AN was similar whether NAA or AA was detected on index colonoscopy, they add.
The overall crude cumulative incidence of AN was lower in the younger age group when no adenoma was detected on index colonoscopy (P = .0003) as well as when NAA was detected, which would be consistent with recommendations from current guidelines for surveillance colonoscopy after adenoma detection. However, there was no difference between the two age groups in the overall cumulative incidence of AN when AA was detected on index colonoscopy.
Overall, the risk for metachronous AN in persons aged 40-49 years was lower when no adenoma was detected on index colonoscopy, but there was no difference between the two age groups when NAA or AA was detected again on index colonoscopy. Similarly, those aged 40-49 years of age had a lower risk for AA or CRC when no adenoma was detected on index colonoscopy – but again, there was no difference in the risk for AA or CRC when either NAA or AA was detected on index colonoscopy.
The authors have no conflicts of interest to report.
A version of this article first appeared on Medscape.com.
New data suggest that for individuals who do not have an adenoma detected on an index colonoscopy, the risk of developing an advanced neoplasia (AN) and colorectal cancer (CRC) is lower in those who are aged 40-49 years, compared with those who are 50-59 years old.
However, there is no difference between the two age groups in detection rates of nonadvanced adenoma (NAA) or advanced adenoma (AA), the same study found.
“The primary goal of this study was to investigate the risk of metachronous AN associated with conventional adenoma detected on the index colonoscopy,” explain the authors, led by Gene Ma, MD, Kaiser Permanente Northern California, San Jose.
“The lack of good-quality evidence to inform surveillance in the 40-49 year old population has resulted in inconsistent surveillance patterns in clinical practice, leading to variation in the quality of care, including both inadequate and excessive colonoscopic surveillance,” Dr. Ma and colleagues observe.
The findings from this study “expand our understanding of the risk of AN and CRC in younger individuals and suggest that the current multi-society guidelines for surveillance may be applicable for individuals 40-49 years of age,” the authors conclude.
The study was published online in The American Journal of Gastroenterology, and included 2,396 individuals between 40 and 49 years of age and 8,978 individuals between 50 and 59 years of age.
The colonoscopy was carried out for screening in 40.2% in the younger age group versus 34.8% in the older age group and was prompted by a positive fecal immunochemical test in 3.3% of the younger age group versus 32% of the older age group.
The median follow-up for both age groups was roughly 7 years.
“When comparing the 40-49 years group to the 50-59 years group, index colonoscopy detected no adenoma in 62.9% versus 40.1% (P < .0001); NAA in 25.4% versus 39.0% (P <.001), and AA in 11.6% versus 21.0% (P < .0001), respectively,” Dr. Ma and colleagues report.
When the two age groups were compared for surveillance colonoscopy, no adenoma was detected in 67% of the younger age group versus 54.7% of the older age group (P < .0001), whereas NAA was detected in 25.4% of the 40- to 49-year-olds versus 38.4% of the 50- to 59-year-olds (P < .0001). AA was detected in 3.5% versus 6.95 (P < .0001) of persons in each of the two age groups, respectively.
AN was detected on surveillance colonoscopy after index colonoscopy in 2.2% of the younger age group and twice that percentage, at 4.4%, in the older age group (P = .0003). On surveillance colonoscopy, NAA was found in 4.6% of the younger age group, compared with 7% of the older age group (P = .03), whereas AA was found in 7.9% of the 40- to 49-year-olds, compared with 11.7% of the 50- to 59-year-olds (P = .06).
The median time until surveillance colonoscopy was similar in both age groups when either NAA or AA was found on index colonoscopy, the authors note. In addition, the median time until the detection of AN was similar whether NAA or AA was detected on index colonoscopy, they add.
The overall crude cumulative incidence of AN was lower in the younger age group when no adenoma was detected on index colonoscopy (P = .0003) as well as when NAA was detected, which would be consistent with recommendations from current guidelines for surveillance colonoscopy after adenoma detection. However, there was no difference between the two age groups in the overall cumulative incidence of AN when AA was detected on index colonoscopy.
Overall, the risk for metachronous AN in persons aged 40-49 years was lower when no adenoma was detected on index colonoscopy, but there was no difference between the two age groups when NAA or AA was detected again on index colonoscopy. Similarly, those aged 40-49 years of age had a lower risk for AA or CRC when no adenoma was detected on index colonoscopy – but again, there was no difference in the risk for AA or CRC when either NAA or AA was detected on index colonoscopy.
The authors have no conflicts of interest to report.
A version of this article first appeared on Medscape.com.
New data suggest that for individuals who do not have an adenoma detected on an index colonoscopy, the risk of developing an advanced neoplasia (AN) and colorectal cancer (CRC) is lower in those who are aged 40-49 years, compared with those who are 50-59 years old.
However, there is no difference between the two age groups in detection rates of nonadvanced adenoma (NAA) or advanced adenoma (AA), the same study found.
“The primary goal of this study was to investigate the risk of metachronous AN associated with conventional adenoma detected on the index colonoscopy,” explain the authors, led by Gene Ma, MD, Kaiser Permanente Northern California, San Jose.
“The lack of good-quality evidence to inform surveillance in the 40-49 year old population has resulted in inconsistent surveillance patterns in clinical practice, leading to variation in the quality of care, including both inadequate and excessive colonoscopic surveillance,” Dr. Ma and colleagues observe.
The findings from this study “expand our understanding of the risk of AN and CRC in younger individuals and suggest that the current multi-society guidelines for surveillance may be applicable for individuals 40-49 years of age,” the authors conclude.
The study was published online in The American Journal of Gastroenterology, and included 2,396 individuals between 40 and 49 years of age and 8,978 individuals between 50 and 59 years of age.
The colonoscopy was carried out for screening in 40.2% in the younger age group versus 34.8% in the older age group and was prompted by a positive fecal immunochemical test in 3.3% of the younger age group versus 32% of the older age group.
The median follow-up for both age groups was roughly 7 years.
“When comparing the 40-49 years group to the 50-59 years group, index colonoscopy detected no adenoma in 62.9% versus 40.1% (P < .0001); NAA in 25.4% versus 39.0% (P <.001), and AA in 11.6% versus 21.0% (P < .0001), respectively,” Dr. Ma and colleagues report.
When the two age groups were compared for surveillance colonoscopy, no adenoma was detected in 67% of the younger age group versus 54.7% of the older age group (P < .0001), whereas NAA was detected in 25.4% of the 40- to 49-year-olds versus 38.4% of the 50- to 59-year-olds (P < .0001). AA was detected in 3.5% versus 6.95 (P < .0001) of persons in each of the two age groups, respectively.
AN was detected on surveillance colonoscopy after index colonoscopy in 2.2% of the younger age group and twice that percentage, at 4.4%, in the older age group (P = .0003). On surveillance colonoscopy, NAA was found in 4.6% of the younger age group, compared with 7% of the older age group (P = .03), whereas AA was found in 7.9% of the 40- to 49-year-olds, compared with 11.7% of the 50- to 59-year-olds (P = .06).
The median time until surveillance colonoscopy was similar in both age groups when either NAA or AA was found on index colonoscopy, the authors note. In addition, the median time until the detection of AN was similar whether NAA or AA was detected on index colonoscopy, they add.
The overall crude cumulative incidence of AN was lower in the younger age group when no adenoma was detected on index colonoscopy (P = .0003) as well as when NAA was detected, which would be consistent with recommendations from current guidelines for surveillance colonoscopy after adenoma detection. However, there was no difference between the two age groups in the overall cumulative incidence of AN when AA was detected on index colonoscopy.
Overall, the risk for metachronous AN in persons aged 40-49 years was lower when no adenoma was detected on index colonoscopy, but there was no difference between the two age groups when NAA or AA was detected again on index colonoscopy. Similarly, those aged 40-49 years of age had a lower risk for AA or CRC when no adenoma was detected on index colonoscopy – but again, there was no difference in the risk for AA or CRC when either NAA or AA was detected on index colonoscopy.
The authors have no conflicts of interest to report.
A version of this article first appeared on Medscape.com.
FROM THE AMERICAN JOURNAL OF GASTROENTEROLOGY
Isotretinoin prescribers need better education on emergency contraception
, in a survey of 57 clinicians.
Pregnancies among patients on isotretinoin have declined since the iPLEDGE risk management program was introduced in 2005, but from 2011 to 2017, 210 to 310 pregnancies were reported to the Food and Drug Administration every year, wrote Catherine E. Smiley of Penn State University, Hershey, Pa., and coauthors Melissa Butt, DrPH, and Andrea L. Zaenglein, MD, of Penn State.
For patients on isotretinoin, EC “becomes critical when abstinence fails or contraception is not used properly,” but EC merits only a brief mention in iPLEDGE materials for patients and providers, they noted.
Patients on isotretinoin who choose abstinence as their form of birth control are the group at greatest risk for pregnancy, Dr. Zaenglein, professor of dermatology and pediatric dermatology, Penn State University, said in an interview. “However, the iPLEDGE program fails to educate patients adequately on emergency contraception,” she explained.
To assess pediatric dermatologists’ understanding of EC and their contraception counseling practices for isotretinoin patients, the researchers surveyed 57 pediatric dermatologists who prescribed isotretinoin as part of their practices. The findings were published in Pediatric Dermatology.Respondents included 53 practicing dermatologists, 2 residents, and 2 fellows. Approximately one-third (31.6%) had been in practice for 6-10 years, almost 23% had been in practice for 3-5 years, and almost 20% had been in practice for 21 or more years. Almost two-thirds practiced pediatric dermatology only.
Overall, 58% of the respondents strongly agreed that they provided contraception counseling to patients at their initial visit for isotretinoin, but only 7% and 3.5% reported providing EC counseling at initial and follow-up visits, respectively. More than half (58%) said they did not counsel patients on the side effects of EC.
As for provider education, 7.1% of respondents said they had received formal education on EC counseling, 25% reported receiving informal education on EC counseling, and 68% said they received no education on EC counseling.
A total of 32% of respondents said they were at least somewhat confident in how to obtain EC in their state.
EC is an effective form of contraception if used after unprotected intercourse, and discounts can reduce the price to as low as $9.69, the researchers wrote in their discussion. “Given that most providers in this study did not receive formal education on EC, and most do not provide EC counseling to their patients of reproductive potential on isotretinoin, EC education should be a core competency in dermatology residency education on isotretinoin prescribing,” the researchers noted. In addition, EC counseling in the iPLEDGE program should be improved by including more information in education materials and reminding patients that EC is an option, they said.
The study findings were limited by several factors including the small sample size and the multiple-choice format that prevented respondents to share rationales for their responses, the researchers noted.
However, the results highlight the need to improve EC education among pediatric dermatologists to better inform patients considering isotretinoin, especially those choosing abstinence as a method of birth control, they emphasized.
“This study is very important at this specific time for two reasons,” Dr. Zaenglein said in an interview. “The first is that with the recent disastrous rollout of the new iPLEDGE changes, there have been many calls to reform the REMS program. For the first time in the 22-year history of the program, the isotretinoin manufacturers, who manage the iPLEDGE program as an unidentified group (the IPMG), have been forced by the FDA to meet with the AAD iPLEDGE Task Force,” said Dr. Zaenglein, a member of the task force.
“The task force is currently advocating for common sense changes to iPLEDGE and I think enhancing education on emergency contraception is vital to the goal of the program, stated as ‘to manage the risk of isotretinoin’s teratogenicity and to minimize fetal exposure,’ ” she added. For many patients who previously became pregnant on isotretinoin, Plan B, an over-the-counter, FDA-approved form of contraception, might have prevented that pregnancy if the patients received adequate education on EC, she said.
The current study is especially relevant now, said Dr. Zaenglein. “With the reversal of Roe v. Wade, access to abortion is restricted or completely banned in many states, which makes educating our patients on how to prevent pregnancy even more important.”
Dr. Zaenglein said she was “somewhat surprised” by how many respondents were not educating their isotretinoin patients on EC. “However, these results follow a known trend among dermatologists. Only 50% of dermatologists prescribe oral contraceptives for acne, despite its being an FDA-approved treatment for the most common dermatologic condition we see in adolescents and young adults,” she noted.
“In general, dermatologists, and subsequently dermatology residents, are poorly educated on issues of reproductive health and how they are relevant to dermatologic care,” she added.
Dr. Zaenglein’s take home message: “Dermatologists should educate all patients of childbearing potential taking isotretinoin on how to acquire and use emergency contraception at every visit.” As for additional research, she said that since the study was conducted with pediatric dermatologists, “it would be very interesting to see if general dermatologists had the same lack of comfort in educating patients on emergency contraception and what their standard counseling practices are.”
The study received no outside funding. Dr. Zaenglein is a member of the AAD’s iPLEDGE Work Group and serves as an editor-in-chief of Pediatric Dermatology.
, in a survey of 57 clinicians.
Pregnancies among patients on isotretinoin have declined since the iPLEDGE risk management program was introduced in 2005, but from 2011 to 2017, 210 to 310 pregnancies were reported to the Food and Drug Administration every year, wrote Catherine E. Smiley of Penn State University, Hershey, Pa., and coauthors Melissa Butt, DrPH, and Andrea L. Zaenglein, MD, of Penn State.
For patients on isotretinoin, EC “becomes critical when abstinence fails or contraception is not used properly,” but EC merits only a brief mention in iPLEDGE materials for patients and providers, they noted.
Patients on isotretinoin who choose abstinence as their form of birth control are the group at greatest risk for pregnancy, Dr. Zaenglein, professor of dermatology and pediatric dermatology, Penn State University, said in an interview. “However, the iPLEDGE program fails to educate patients adequately on emergency contraception,” she explained.
To assess pediatric dermatologists’ understanding of EC and their contraception counseling practices for isotretinoin patients, the researchers surveyed 57 pediatric dermatologists who prescribed isotretinoin as part of their practices. The findings were published in Pediatric Dermatology.Respondents included 53 practicing dermatologists, 2 residents, and 2 fellows. Approximately one-third (31.6%) had been in practice for 6-10 years, almost 23% had been in practice for 3-5 years, and almost 20% had been in practice for 21 or more years. Almost two-thirds practiced pediatric dermatology only.
Overall, 58% of the respondents strongly agreed that they provided contraception counseling to patients at their initial visit for isotretinoin, but only 7% and 3.5% reported providing EC counseling at initial and follow-up visits, respectively. More than half (58%) said they did not counsel patients on the side effects of EC.
As for provider education, 7.1% of respondents said they had received formal education on EC counseling, 25% reported receiving informal education on EC counseling, and 68% said they received no education on EC counseling.
A total of 32% of respondents said they were at least somewhat confident in how to obtain EC in their state.
EC is an effective form of contraception if used after unprotected intercourse, and discounts can reduce the price to as low as $9.69, the researchers wrote in their discussion. “Given that most providers in this study did not receive formal education on EC, and most do not provide EC counseling to their patients of reproductive potential on isotretinoin, EC education should be a core competency in dermatology residency education on isotretinoin prescribing,” the researchers noted. In addition, EC counseling in the iPLEDGE program should be improved by including more information in education materials and reminding patients that EC is an option, they said.
The study findings were limited by several factors including the small sample size and the multiple-choice format that prevented respondents to share rationales for their responses, the researchers noted.
However, the results highlight the need to improve EC education among pediatric dermatologists to better inform patients considering isotretinoin, especially those choosing abstinence as a method of birth control, they emphasized.
“This study is very important at this specific time for two reasons,” Dr. Zaenglein said in an interview. “The first is that with the recent disastrous rollout of the new iPLEDGE changes, there have been many calls to reform the REMS program. For the first time in the 22-year history of the program, the isotretinoin manufacturers, who manage the iPLEDGE program as an unidentified group (the IPMG), have been forced by the FDA to meet with the AAD iPLEDGE Task Force,” said Dr. Zaenglein, a member of the task force.
“The task force is currently advocating for common sense changes to iPLEDGE and I think enhancing education on emergency contraception is vital to the goal of the program, stated as ‘to manage the risk of isotretinoin’s teratogenicity and to minimize fetal exposure,’ ” she added. For many patients who previously became pregnant on isotretinoin, Plan B, an over-the-counter, FDA-approved form of contraception, might have prevented that pregnancy if the patients received adequate education on EC, she said.
The current study is especially relevant now, said Dr. Zaenglein. “With the reversal of Roe v. Wade, access to abortion is restricted or completely banned in many states, which makes educating our patients on how to prevent pregnancy even more important.”
Dr. Zaenglein said she was “somewhat surprised” by how many respondents were not educating their isotretinoin patients on EC. “However, these results follow a known trend among dermatologists. Only 50% of dermatologists prescribe oral contraceptives for acne, despite its being an FDA-approved treatment for the most common dermatologic condition we see in adolescents and young adults,” she noted.
“In general, dermatologists, and subsequently dermatology residents, are poorly educated on issues of reproductive health and how they are relevant to dermatologic care,” she added.
Dr. Zaenglein’s take home message: “Dermatologists should educate all patients of childbearing potential taking isotretinoin on how to acquire and use emergency contraception at every visit.” As for additional research, she said that since the study was conducted with pediatric dermatologists, “it would be very interesting to see if general dermatologists had the same lack of comfort in educating patients on emergency contraception and what their standard counseling practices are.”
The study received no outside funding. Dr. Zaenglein is a member of the AAD’s iPLEDGE Work Group and serves as an editor-in-chief of Pediatric Dermatology.
, in a survey of 57 clinicians.
Pregnancies among patients on isotretinoin have declined since the iPLEDGE risk management program was introduced in 2005, but from 2011 to 2017, 210 to 310 pregnancies were reported to the Food and Drug Administration every year, wrote Catherine E. Smiley of Penn State University, Hershey, Pa., and coauthors Melissa Butt, DrPH, and Andrea L. Zaenglein, MD, of Penn State.
For patients on isotretinoin, EC “becomes critical when abstinence fails or contraception is not used properly,” but EC merits only a brief mention in iPLEDGE materials for patients and providers, they noted.
Patients on isotretinoin who choose abstinence as their form of birth control are the group at greatest risk for pregnancy, Dr. Zaenglein, professor of dermatology and pediatric dermatology, Penn State University, said in an interview. “However, the iPLEDGE program fails to educate patients adequately on emergency contraception,” she explained.
To assess pediatric dermatologists’ understanding of EC and their contraception counseling practices for isotretinoin patients, the researchers surveyed 57 pediatric dermatologists who prescribed isotretinoin as part of their practices. The findings were published in Pediatric Dermatology.Respondents included 53 practicing dermatologists, 2 residents, and 2 fellows. Approximately one-third (31.6%) had been in practice for 6-10 years, almost 23% had been in practice for 3-5 years, and almost 20% had been in practice for 21 or more years. Almost two-thirds practiced pediatric dermatology only.
Overall, 58% of the respondents strongly agreed that they provided contraception counseling to patients at their initial visit for isotretinoin, but only 7% and 3.5% reported providing EC counseling at initial and follow-up visits, respectively. More than half (58%) said they did not counsel patients on the side effects of EC.
As for provider education, 7.1% of respondents said they had received formal education on EC counseling, 25% reported receiving informal education on EC counseling, and 68% said they received no education on EC counseling.
A total of 32% of respondents said they were at least somewhat confident in how to obtain EC in their state.
EC is an effective form of contraception if used after unprotected intercourse, and discounts can reduce the price to as low as $9.69, the researchers wrote in their discussion. “Given that most providers in this study did not receive formal education on EC, and most do not provide EC counseling to their patients of reproductive potential on isotretinoin, EC education should be a core competency in dermatology residency education on isotretinoin prescribing,” the researchers noted. In addition, EC counseling in the iPLEDGE program should be improved by including more information in education materials and reminding patients that EC is an option, they said.
The study findings were limited by several factors including the small sample size and the multiple-choice format that prevented respondents to share rationales for their responses, the researchers noted.
However, the results highlight the need to improve EC education among pediatric dermatologists to better inform patients considering isotretinoin, especially those choosing abstinence as a method of birth control, they emphasized.
“This study is very important at this specific time for two reasons,” Dr. Zaenglein said in an interview. “The first is that with the recent disastrous rollout of the new iPLEDGE changes, there have been many calls to reform the REMS program. For the first time in the 22-year history of the program, the isotretinoin manufacturers, who manage the iPLEDGE program as an unidentified group (the IPMG), have been forced by the FDA to meet with the AAD iPLEDGE Task Force,” said Dr. Zaenglein, a member of the task force.
“The task force is currently advocating for common sense changes to iPLEDGE and I think enhancing education on emergency contraception is vital to the goal of the program, stated as ‘to manage the risk of isotretinoin’s teratogenicity and to minimize fetal exposure,’ ” she added. For many patients who previously became pregnant on isotretinoin, Plan B, an over-the-counter, FDA-approved form of contraception, might have prevented that pregnancy if the patients received adequate education on EC, she said.
The current study is especially relevant now, said Dr. Zaenglein. “With the reversal of Roe v. Wade, access to abortion is restricted or completely banned in many states, which makes educating our patients on how to prevent pregnancy even more important.”
Dr. Zaenglein said she was “somewhat surprised” by how many respondents were not educating their isotretinoin patients on EC. “However, these results follow a known trend among dermatologists. Only 50% of dermatologists prescribe oral contraceptives for acne, despite its being an FDA-approved treatment for the most common dermatologic condition we see in adolescents and young adults,” she noted.
“In general, dermatologists, and subsequently dermatology residents, are poorly educated on issues of reproductive health and how they are relevant to dermatologic care,” she added.
Dr. Zaenglein’s take home message: “Dermatologists should educate all patients of childbearing potential taking isotretinoin on how to acquire and use emergency contraception at every visit.” As for additional research, she said that since the study was conducted with pediatric dermatologists, “it would be very interesting to see if general dermatologists had the same lack of comfort in educating patients on emergency contraception and what their standard counseling practices are.”
The study received no outside funding. Dr. Zaenglein is a member of the AAD’s iPLEDGE Work Group and serves as an editor-in-chief of Pediatric Dermatology.
FROM PEDIATRIC DERMATOLOGY
New study supports safety of COVID-19 boosters during pregnancy
Doctors and health professionals continue to recommend COVID-19 vaccine boosters or third doses for adolescents and adults more than 5 months after their initial vaccinations with the Pfizer-BioNTech BNT162b2 or Moderna mRNA-1273 primary vaccine series or more than 2 months after receiving the Janssen JNJ-78436735 vaccine, Alisa Kachikis, MD, of the University of Washington, Seattle, and colleagues wrote in JAMA Network Open.
Although multiple studies have shown that the COVID-19 primary series is safe and well tolerated in pregnant and lactating women, information on the safety and tolerability of boosters are lacking, the researchers noted.
“COVID-19 will be with us for a while, and it is important to continue to provide data on COVID-19 vaccines in these groups, particularly because there still are many questions about the vaccine, and because pregnant individuals have been, understandably, more hesitant to receive COVID-19 vaccines,” Dr. Kachikis said in an interview. “The findings of this study that COVID-19 booster doses are well tolerated among pregnant and lactating individuals are especially pertinent with the new COVID-19 boosters available this fall.”
In the new study, the researchers reviewed data from 17,014 participants who were part of an ongoing online prospective study of COVID-19 vaccines in pregnant and lactating individuals. Data were collected between October 2021 and April 2022 through an online survey.
The study population included 2,009 participants (11.8%) who were pregnant at the time of their booster or third dose, 10,279 (60.4%) who were lactating, and 4,726 (27.8%) who were neither pregnant nor lactating. The mean age of the participants was 33.3 years; 92.1% self-identified as White, 94.5% self-identified as non-Hispanic, and 99.7% self-identified as female.
The receipt of a booster was similar across trimesters; 26.4%, 36.5%, and 37.1% of participants received boosters or third doses in the first, second, and third trimester, respectively. The primary outcome was self-reported vaccine reactions within 24 hours of the dose.
Overall, 82.8% of the respondents reported a reaction at the site of the injection, such as redness, pain, or swelling, and 67.9% reported at least one systemic symptom, such as aches and pains, headache, chills, or fever. The most frequently reported symptoms across all groups were injection-site pain (82.2%) and fatigue (54.4%).
The pregnant women were significantly more likely than nonpregnant or nonlactating individuals to report any local reaction at the injection site (adjusted odds ratio, 1.2; P = .01), but less likely to report any systemic reaction (aOR, 0.7; P < .001).
The majority (97.6%) of the pregnant respondents and 96.0% of those lactating reported no obstetric or lactation concerns after vaccination.
Overall, a majority of the respondents reported that recommendations from public health authorities were helpful in their decision to receive a COVID-19 booster or third dose (90.0% of pregnant respondents, 89.9% of lactating respondents, and 88.1% of those neither pregnant nor lactating).
Although vaccine uptake in the current study population was high (91.1% overall and 95.0% of those pregnant), “the importance of the health care professional’s recommendation is pertinent given the ongoing increased vaccine hesitancy among pregnant individuals in the context of the COVID-19 vaccine,” the researchers emphasized.
The study findings were limited by several factors including the reliance on self-reports and a convenience sample composed mainly of health care workers because of their vaccine eligibility at the time the study started, which limits generalizability, the researchers noted. Analyses on the pregnancy outcomes of those who were pregnant when vaccinated are in progress.
The results were strengthened by the large study population that included participants from all 50 states and several territories, and ability to compare results between pregnant and lactating individuals with those who were neither pregnant nor lactating, but were of childbearing age, they said.
The results support the safety of COVID-19 boosters for pregnant and breastfeeding individuals, and these data are important to inform discussions between patients and clinicians to boost vaccine uptake and acceptance in this population, they concluded.
“Our earlier data analysis showed that pregnant and lactating individuals did very well with the initial COVID-19 vaccine series, so it was not very surprising that they also did well with COVID-19 booster or third doses,” Dr. Kachikis said in an interview.
There are two takeaway messages for clinicians, she said: “First, pregnant and lactating individuals tolerated the COVID-19 booster well. The second is that clinicians are very important when it comes to vaccine acceptance.”
“In our study, we found that, while pregnant participants were more likely to report that they were hesitant to receive the booster, they also were more likely to have discussed the COVID-19 booster with their health care provider, and to have received a recommendation to receive the booster. So, spending a little bit of extra time with patients discussing COVID-19 boosters and recommending them can make a significant difference,” she said.
The message of the study is highly reassuring for pregnant and lactating individuals, Dr. Kachikis added. “Most of the participants reported that they had fewer symptoms with the COVID-19 booster compared to the primary vaccine series, which is good news, especially since a new COVID-19 booster is being recommended for the fall.”
Reassuring findings for doctors and patients
The current study is especially timely, as updated COVID-19 boosters have now been recommended for most individuals by the Centers for Disease Control and Prevention, Martina L. Badell, MD, a maternal-fetal medicine specialist at Emory University, Atlanta, said in an interview.
The findings support previous studies on the tolerability of COVID-19 vaccinations in pregnant and lactating persons, said Dr. Badell, who was not involved in the study.
The reassuring message for clinicians is that COVID-19 booster vaccinations are similarly well tolerated in pregnancy and lactation as they are in nonpregnant individuals, said Dr. Badell. “Given the risks of COVID infections in pregnancy and neonates, reassuring data on the tolerability and safety of vaccination in this population is very important.” Also, the researchers found that all three cohorts reported that recommendations from public or medical health authorities helped them make a decision about vaccination; “thus the more data to support these recommendations, the better,” she emphasized.
If you are pregnant or breastfeeding, the message from the study is that COVID-19 booster vaccinations are similarly well tolerated by those who are pregnant or breastfeeding and those who are not, said Dr. Badell.
“This study provides additional support for the strong recommendation to encourage not only COVID-19 vaccination in pregnancy and lactation, but booster vaccinations specifically,” and pregnant and breastfeeding individuals should not be excluded from the new CDC recommendations for COVID-19 boosters, she said.
Future research suggestions
Next steps for research include evaluating the obstetrical and neonatal outcomes in pregnancy and lactation following COVID- 19 boosters, Dr. Badell added.
Dr. Kachikis suggested studies try to answer the remaining questions about COVID-19 vaccines and the immunity of pregnant and lactating persons, particularly since they were excluded from the early clinical trials in 2020.
The study was supported by the National Institute of Allergy and Infectious Diseases, a Women’s Reproductive Health Research Award, and the National Center for Advancing Translational Sciences of the National Institutes of Health. \Dr. Kachikis disclosed serving as a research consultant for Pfizer and GlaxoSmithKline and as an unpaid consultant for GlaxoSmithKline unrelated to the current study, as well as grant support from Merck and Pfizer unrelated to the current study. Dr. Badell had no financial conflicts to disclose.
Doctors and health professionals continue to recommend COVID-19 vaccine boosters or third doses for adolescents and adults more than 5 months after their initial vaccinations with the Pfizer-BioNTech BNT162b2 or Moderna mRNA-1273 primary vaccine series or more than 2 months after receiving the Janssen JNJ-78436735 vaccine, Alisa Kachikis, MD, of the University of Washington, Seattle, and colleagues wrote in JAMA Network Open.
Although multiple studies have shown that the COVID-19 primary series is safe and well tolerated in pregnant and lactating women, information on the safety and tolerability of boosters are lacking, the researchers noted.
“COVID-19 will be with us for a while, and it is important to continue to provide data on COVID-19 vaccines in these groups, particularly because there still are many questions about the vaccine, and because pregnant individuals have been, understandably, more hesitant to receive COVID-19 vaccines,” Dr. Kachikis said in an interview. “The findings of this study that COVID-19 booster doses are well tolerated among pregnant and lactating individuals are especially pertinent with the new COVID-19 boosters available this fall.”
In the new study, the researchers reviewed data from 17,014 participants who were part of an ongoing online prospective study of COVID-19 vaccines in pregnant and lactating individuals. Data were collected between October 2021 and April 2022 through an online survey.
The study population included 2,009 participants (11.8%) who were pregnant at the time of their booster or third dose, 10,279 (60.4%) who were lactating, and 4,726 (27.8%) who were neither pregnant nor lactating. The mean age of the participants was 33.3 years; 92.1% self-identified as White, 94.5% self-identified as non-Hispanic, and 99.7% self-identified as female.
The receipt of a booster was similar across trimesters; 26.4%, 36.5%, and 37.1% of participants received boosters or third doses in the first, second, and third trimester, respectively. The primary outcome was self-reported vaccine reactions within 24 hours of the dose.
Overall, 82.8% of the respondents reported a reaction at the site of the injection, such as redness, pain, or swelling, and 67.9% reported at least one systemic symptom, such as aches and pains, headache, chills, or fever. The most frequently reported symptoms across all groups were injection-site pain (82.2%) and fatigue (54.4%).
The pregnant women were significantly more likely than nonpregnant or nonlactating individuals to report any local reaction at the injection site (adjusted odds ratio, 1.2; P = .01), but less likely to report any systemic reaction (aOR, 0.7; P < .001).
The majority (97.6%) of the pregnant respondents and 96.0% of those lactating reported no obstetric or lactation concerns after vaccination.
Overall, a majority of the respondents reported that recommendations from public health authorities were helpful in their decision to receive a COVID-19 booster or third dose (90.0% of pregnant respondents, 89.9% of lactating respondents, and 88.1% of those neither pregnant nor lactating).
Although vaccine uptake in the current study population was high (91.1% overall and 95.0% of those pregnant), “the importance of the health care professional’s recommendation is pertinent given the ongoing increased vaccine hesitancy among pregnant individuals in the context of the COVID-19 vaccine,” the researchers emphasized.
The study findings were limited by several factors including the reliance on self-reports and a convenience sample composed mainly of health care workers because of their vaccine eligibility at the time the study started, which limits generalizability, the researchers noted. Analyses on the pregnancy outcomes of those who were pregnant when vaccinated are in progress.
The results were strengthened by the large study population that included participants from all 50 states and several territories, and ability to compare results between pregnant and lactating individuals with those who were neither pregnant nor lactating, but were of childbearing age, they said.
The results support the safety of COVID-19 boosters for pregnant and breastfeeding individuals, and these data are important to inform discussions between patients and clinicians to boost vaccine uptake and acceptance in this population, they concluded.
“Our earlier data analysis showed that pregnant and lactating individuals did very well with the initial COVID-19 vaccine series, so it was not very surprising that they also did well with COVID-19 booster or third doses,” Dr. Kachikis said in an interview.
There are two takeaway messages for clinicians, she said: “First, pregnant and lactating individuals tolerated the COVID-19 booster well. The second is that clinicians are very important when it comes to vaccine acceptance.”
“In our study, we found that, while pregnant participants were more likely to report that they were hesitant to receive the booster, they also were more likely to have discussed the COVID-19 booster with their health care provider, and to have received a recommendation to receive the booster. So, spending a little bit of extra time with patients discussing COVID-19 boosters and recommending them can make a significant difference,” she said.
The message of the study is highly reassuring for pregnant and lactating individuals, Dr. Kachikis added. “Most of the participants reported that they had fewer symptoms with the COVID-19 booster compared to the primary vaccine series, which is good news, especially since a new COVID-19 booster is being recommended for the fall.”
Reassuring findings for doctors and patients
The current study is especially timely, as updated COVID-19 boosters have now been recommended for most individuals by the Centers for Disease Control and Prevention, Martina L. Badell, MD, a maternal-fetal medicine specialist at Emory University, Atlanta, said in an interview.
The findings support previous studies on the tolerability of COVID-19 vaccinations in pregnant and lactating persons, said Dr. Badell, who was not involved in the study.
The reassuring message for clinicians is that COVID-19 booster vaccinations are similarly well tolerated in pregnancy and lactation as they are in nonpregnant individuals, said Dr. Badell. “Given the risks of COVID infections in pregnancy and neonates, reassuring data on the tolerability and safety of vaccination in this population is very important.” Also, the researchers found that all three cohorts reported that recommendations from public or medical health authorities helped them make a decision about vaccination; “thus the more data to support these recommendations, the better,” she emphasized.
If you are pregnant or breastfeeding, the message from the study is that COVID-19 booster vaccinations are similarly well tolerated by those who are pregnant or breastfeeding and those who are not, said Dr. Badell.
“This study provides additional support for the strong recommendation to encourage not only COVID-19 vaccination in pregnancy and lactation, but booster vaccinations specifically,” and pregnant and breastfeeding individuals should not be excluded from the new CDC recommendations for COVID-19 boosters, she said.
Future research suggestions
Next steps for research include evaluating the obstetrical and neonatal outcomes in pregnancy and lactation following COVID- 19 boosters, Dr. Badell added.
Dr. Kachikis suggested studies try to answer the remaining questions about COVID-19 vaccines and the immunity of pregnant and lactating persons, particularly since they were excluded from the early clinical trials in 2020.
The study was supported by the National Institute of Allergy and Infectious Diseases, a Women’s Reproductive Health Research Award, and the National Center for Advancing Translational Sciences of the National Institutes of Health. \Dr. Kachikis disclosed serving as a research consultant for Pfizer and GlaxoSmithKline and as an unpaid consultant for GlaxoSmithKline unrelated to the current study, as well as grant support from Merck and Pfizer unrelated to the current study. Dr. Badell had no financial conflicts to disclose.
Doctors and health professionals continue to recommend COVID-19 vaccine boosters or third doses for adolescents and adults more than 5 months after their initial vaccinations with the Pfizer-BioNTech BNT162b2 or Moderna mRNA-1273 primary vaccine series or more than 2 months after receiving the Janssen JNJ-78436735 vaccine, Alisa Kachikis, MD, of the University of Washington, Seattle, and colleagues wrote in JAMA Network Open.
Although multiple studies have shown that the COVID-19 primary series is safe and well tolerated in pregnant and lactating women, information on the safety and tolerability of boosters are lacking, the researchers noted.
“COVID-19 will be with us for a while, and it is important to continue to provide data on COVID-19 vaccines in these groups, particularly because there still are many questions about the vaccine, and because pregnant individuals have been, understandably, more hesitant to receive COVID-19 vaccines,” Dr. Kachikis said in an interview. “The findings of this study that COVID-19 booster doses are well tolerated among pregnant and lactating individuals are especially pertinent with the new COVID-19 boosters available this fall.”
In the new study, the researchers reviewed data from 17,014 participants who were part of an ongoing online prospective study of COVID-19 vaccines in pregnant and lactating individuals. Data were collected between October 2021 and April 2022 through an online survey.
The study population included 2,009 participants (11.8%) who were pregnant at the time of their booster or third dose, 10,279 (60.4%) who were lactating, and 4,726 (27.8%) who were neither pregnant nor lactating. The mean age of the participants was 33.3 years; 92.1% self-identified as White, 94.5% self-identified as non-Hispanic, and 99.7% self-identified as female.
The receipt of a booster was similar across trimesters; 26.4%, 36.5%, and 37.1% of participants received boosters or third doses in the first, second, and third trimester, respectively. The primary outcome was self-reported vaccine reactions within 24 hours of the dose.
Overall, 82.8% of the respondents reported a reaction at the site of the injection, such as redness, pain, or swelling, and 67.9% reported at least one systemic symptom, such as aches and pains, headache, chills, or fever. The most frequently reported symptoms across all groups were injection-site pain (82.2%) and fatigue (54.4%).
The pregnant women were significantly more likely than nonpregnant or nonlactating individuals to report any local reaction at the injection site (adjusted odds ratio, 1.2; P = .01), but less likely to report any systemic reaction (aOR, 0.7; P < .001).
The majority (97.6%) of the pregnant respondents and 96.0% of those lactating reported no obstetric or lactation concerns after vaccination.
Overall, a majority of the respondents reported that recommendations from public health authorities were helpful in their decision to receive a COVID-19 booster or third dose (90.0% of pregnant respondents, 89.9% of lactating respondents, and 88.1% of those neither pregnant nor lactating).
Although vaccine uptake in the current study population was high (91.1% overall and 95.0% of those pregnant), “the importance of the health care professional’s recommendation is pertinent given the ongoing increased vaccine hesitancy among pregnant individuals in the context of the COVID-19 vaccine,” the researchers emphasized.
The study findings were limited by several factors including the reliance on self-reports and a convenience sample composed mainly of health care workers because of their vaccine eligibility at the time the study started, which limits generalizability, the researchers noted. Analyses on the pregnancy outcomes of those who were pregnant when vaccinated are in progress.
The results were strengthened by the large study population that included participants from all 50 states and several territories, and ability to compare results between pregnant and lactating individuals with those who were neither pregnant nor lactating, but were of childbearing age, they said.
The results support the safety of COVID-19 boosters for pregnant and breastfeeding individuals, and these data are important to inform discussions between patients and clinicians to boost vaccine uptake and acceptance in this population, they concluded.
“Our earlier data analysis showed that pregnant and lactating individuals did very well with the initial COVID-19 vaccine series, so it was not very surprising that they also did well with COVID-19 booster or third doses,” Dr. Kachikis said in an interview.
There are two takeaway messages for clinicians, she said: “First, pregnant and lactating individuals tolerated the COVID-19 booster well. The second is that clinicians are very important when it comes to vaccine acceptance.”
“In our study, we found that, while pregnant participants were more likely to report that they were hesitant to receive the booster, they also were more likely to have discussed the COVID-19 booster with their health care provider, and to have received a recommendation to receive the booster. So, spending a little bit of extra time with patients discussing COVID-19 boosters and recommending them can make a significant difference,” she said.
The message of the study is highly reassuring for pregnant and lactating individuals, Dr. Kachikis added. “Most of the participants reported that they had fewer symptoms with the COVID-19 booster compared to the primary vaccine series, which is good news, especially since a new COVID-19 booster is being recommended for the fall.”
Reassuring findings for doctors and patients
The current study is especially timely, as updated COVID-19 boosters have now been recommended for most individuals by the Centers for Disease Control and Prevention, Martina L. Badell, MD, a maternal-fetal medicine specialist at Emory University, Atlanta, said in an interview.
The findings support previous studies on the tolerability of COVID-19 vaccinations in pregnant and lactating persons, said Dr. Badell, who was not involved in the study.
The reassuring message for clinicians is that COVID-19 booster vaccinations are similarly well tolerated in pregnancy and lactation as they are in nonpregnant individuals, said Dr. Badell. “Given the risks of COVID infections in pregnancy and neonates, reassuring data on the tolerability and safety of vaccination in this population is very important.” Also, the researchers found that all three cohorts reported that recommendations from public or medical health authorities helped them make a decision about vaccination; “thus the more data to support these recommendations, the better,” she emphasized.
If you are pregnant or breastfeeding, the message from the study is that COVID-19 booster vaccinations are similarly well tolerated by those who are pregnant or breastfeeding and those who are not, said Dr. Badell.
“This study provides additional support for the strong recommendation to encourage not only COVID-19 vaccination in pregnancy and lactation, but booster vaccinations specifically,” and pregnant and breastfeeding individuals should not be excluded from the new CDC recommendations for COVID-19 boosters, she said.
Future research suggestions
Next steps for research include evaluating the obstetrical and neonatal outcomes in pregnancy and lactation following COVID- 19 boosters, Dr. Badell added.
Dr. Kachikis suggested studies try to answer the remaining questions about COVID-19 vaccines and the immunity of pregnant and lactating persons, particularly since they were excluded from the early clinical trials in 2020.
The study was supported by the National Institute of Allergy and Infectious Diseases, a Women’s Reproductive Health Research Award, and the National Center for Advancing Translational Sciences of the National Institutes of Health. \Dr. Kachikis disclosed serving as a research consultant for Pfizer and GlaxoSmithKline and as an unpaid consultant for GlaxoSmithKline unrelated to the current study, as well as grant support from Merck and Pfizer unrelated to the current study. Dr. Badell had no financial conflicts to disclose.
FROM JAMA NETWORK OPEN
Unvaccinated 10 times more likely to be hospitalized for Omicron
The data, which included almost 200,000 COVID-19–associated hospitalizations across 13 states, also showed that vaccinated, hospitalized patients were more often older and already dealing with other health conditions, compared with unvaccinated, hospitalized patients, reported lead author Fiona P. Havers, MD, of the CDC, Atlanta.
“Unlike previously published reports and web pages … this study reports hospitalization rates by vaccination status and clinical and demographic characteristics of hospitalized patients, beginning with the period when vaccines first became available, and includes comparisons of unvaccinated persons, persons vaccinated with a primary series without a booster dose, and those vaccinated with a primary series and at least 1 booster dose,” the investigators wrote in JAMA Internal Medicine.
In total, the investigators reviewed 192,509 hospitalizations involving patients 18 years and older. The study period spanned from Jan. 1, 2021, to April 30, 2022. Data were reported month by month, showing that the relative monthly hospitalization rate peaked in May 2021, when it was 17.7 times higher for unvaccinated versus vaccinated individuals (with or without a booster).
To account for differences in clinical course between Delta and Omicron, the investigators also analyzed data sorted into two time periods: July-December 2021 (Delta predominant) and January-April 2022 (Omicron BA.1 predominant). These analyses revealed the greater hospitalization risk presented by Delta. Specifically, unvaccinated people were 12.2 times more likely to be hospitalized for Delta than vaccinated people, with or without a booster, versus 6.8 times for Omicron BA.1.
Study shows power of the booster
A closer look at the Omicron BA.1 data showed the power of a booster dose. From January to April 2022, individuals who were fully vaccinated with a booster dose were 10.5 times less likely than unvaccinated individuals to be hospitalized for Omicron BA.1. Plus, boosted people were 2.5 times less likely to be hospitalized for Omicron BA.1 than people who got vaccinated but skipped the booster.
“The high hospitalization rates in unvaccinated compared with vaccinated persons with and without a booster dose underscores the importance of COVID-19 vaccinations in preventing hospitalizations and suggests that increasing vaccination coverage, including booster dose coverage, can prevent hospitalizations, serious illness, and death,” the investigators wrote.
The study also revealed that vaccinated hospitalized patients were significantly older, on average, than unvaccinated hospitalized patients (median, 70 vs. 58 years; P < .001). They were also significantly more likely to have three or more underlying medical conditions (77.8% vs. 51.6%; P < .001)
“A greater proportion of hospitalized cases among vaccinated persons occurred in individuals with medical fragility who were older, more likely to reside in long-term care facilities, and have three or more underlying medical conditions, including immunosuppressive conditions,” the investigators wrote.
New variants outpacing data, vaccines remain essential
While data from April 2022 alone showed a 3.5-fold higher rate of hospitalization among unvaccinated versus vaccinated individuals with or without a booster, newer data suggest that emerging strains of Omicron are putting more people in the hospital.
A recent report by the CDC showed weekly hospitalization rates climbing from March 20 to May 31, 2022, which coincided with predominance of the newer Omicron BA.2 variant. While unvaccinated people were still around 3.5 times more likely to be hospitalized than vaccinated people, overall hospitalization rates jumped 3-fold for people 65 years and older, and 1.7-fold for adults younger than 65. Adding further complexity to this constantly evolving situation is that Omicron BA.2 has since been joined by the BA.4 and BA.5 lineages, for which vaccines are now available.
In the paper published in JAMA Internal Medicine, the CDC report, and in a comment for this article, the CDC offered the same take-home message: Get vaccinated.
“These findings reinforce previous research illustrating how vaccination provides protection from hospitalization due to COVID-19,” a CDC spokesperson said. “COVID-19 vaccines are proven to help prevent serious COVID-19 illness, and everyone ages 6 months and older should stay up to date with COVID-19 vaccines.”
The study published in JAMA Internal Medicine was supported by the CDC. The investigators disclosed additional relationships with Sanofi, GSK, MedImmune, and others.
The data, which included almost 200,000 COVID-19–associated hospitalizations across 13 states, also showed that vaccinated, hospitalized patients were more often older and already dealing with other health conditions, compared with unvaccinated, hospitalized patients, reported lead author Fiona P. Havers, MD, of the CDC, Atlanta.
“Unlike previously published reports and web pages … this study reports hospitalization rates by vaccination status and clinical and demographic characteristics of hospitalized patients, beginning with the period when vaccines first became available, and includes comparisons of unvaccinated persons, persons vaccinated with a primary series without a booster dose, and those vaccinated with a primary series and at least 1 booster dose,” the investigators wrote in JAMA Internal Medicine.
In total, the investigators reviewed 192,509 hospitalizations involving patients 18 years and older. The study period spanned from Jan. 1, 2021, to April 30, 2022. Data were reported month by month, showing that the relative monthly hospitalization rate peaked in May 2021, when it was 17.7 times higher for unvaccinated versus vaccinated individuals (with or without a booster).
To account for differences in clinical course between Delta and Omicron, the investigators also analyzed data sorted into two time periods: July-December 2021 (Delta predominant) and January-April 2022 (Omicron BA.1 predominant). These analyses revealed the greater hospitalization risk presented by Delta. Specifically, unvaccinated people were 12.2 times more likely to be hospitalized for Delta than vaccinated people, with or without a booster, versus 6.8 times for Omicron BA.1.
Study shows power of the booster
A closer look at the Omicron BA.1 data showed the power of a booster dose. From January to April 2022, individuals who were fully vaccinated with a booster dose were 10.5 times less likely than unvaccinated individuals to be hospitalized for Omicron BA.1. Plus, boosted people were 2.5 times less likely to be hospitalized for Omicron BA.1 than people who got vaccinated but skipped the booster.
“The high hospitalization rates in unvaccinated compared with vaccinated persons with and without a booster dose underscores the importance of COVID-19 vaccinations in preventing hospitalizations and suggests that increasing vaccination coverage, including booster dose coverage, can prevent hospitalizations, serious illness, and death,” the investigators wrote.
The study also revealed that vaccinated hospitalized patients were significantly older, on average, than unvaccinated hospitalized patients (median, 70 vs. 58 years; P < .001). They were also significantly more likely to have three or more underlying medical conditions (77.8% vs. 51.6%; P < .001)
“A greater proportion of hospitalized cases among vaccinated persons occurred in individuals with medical fragility who were older, more likely to reside in long-term care facilities, and have three or more underlying medical conditions, including immunosuppressive conditions,” the investigators wrote.
New variants outpacing data, vaccines remain essential
While data from April 2022 alone showed a 3.5-fold higher rate of hospitalization among unvaccinated versus vaccinated individuals with or without a booster, newer data suggest that emerging strains of Omicron are putting more people in the hospital.
A recent report by the CDC showed weekly hospitalization rates climbing from March 20 to May 31, 2022, which coincided with predominance of the newer Omicron BA.2 variant. While unvaccinated people were still around 3.5 times more likely to be hospitalized than vaccinated people, overall hospitalization rates jumped 3-fold for people 65 years and older, and 1.7-fold for adults younger than 65. Adding further complexity to this constantly evolving situation is that Omicron BA.2 has since been joined by the BA.4 and BA.5 lineages, for which vaccines are now available.
In the paper published in JAMA Internal Medicine, the CDC report, and in a comment for this article, the CDC offered the same take-home message: Get vaccinated.
“These findings reinforce previous research illustrating how vaccination provides protection from hospitalization due to COVID-19,” a CDC spokesperson said. “COVID-19 vaccines are proven to help prevent serious COVID-19 illness, and everyone ages 6 months and older should stay up to date with COVID-19 vaccines.”
The study published in JAMA Internal Medicine was supported by the CDC. The investigators disclosed additional relationships with Sanofi, GSK, MedImmune, and others.
The data, which included almost 200,000 COVID-19–associated hospitalizations across 13 states, also showed that vaccinated, hospitalized patients were more often older and already dealing with other health conditions, compared with unvaccinated, hospitalized patients, reported lead author Fiona P. Havers, MD, of the CDC, Atlanta.
“Unlike previously published reports and web pages … this study reports hospitalization rates by vaccination status and clinical and demographic characteristics of hospitalized patients, beginning with the period when vaccines first became available, and includes comparisons of unvaccinated persons, persons vaccinated with a primary series without a booster dose, and those vaccinated with a primary series and at least 1 booster dose,” the investigators wrote in JAMA Internal Medicine.
In total, the investigators reviewed 192,509 hospitalizations involving patients 18 years and older. The study period spanned from Jan. 1, 2021, to April 30, 2022. Data were reported month by month, showing that the relative monthly hospitalization rate peaked in May 2021, when it was 17.7 times higher for unvaccinated versus vaccinated individuals (with or without a booster).
To account for differences in clinical course between Delta and Omicron, the investigators also analyzed data sorted into two time periods: July-December 2021 (Delta predominant) and January-April 2022 (Omicron BA.1 predominant). These analyses revealed the greater hospitalization risk presented by Delta. Specifically, unvaccinated people were 12.2 times more likely to be hospitalized for Delta than vaccinated people, with or without a booster, versus 6.8 times for Omicron BA.1.
Study shows power of the booster
A closer look at the Omicron BA.1 data showed the power of a booster dose. From January to April 2022, individuals who were fully vaccinated with a booster dose were 10.5 times less likely than unvaccinated individuals to be hospitalized for Omicron BA.1. Plus, boosted people were 2.5 times less likely to be hospitalized for Omicron BA.1 than people who got vaccinated but skipped the booster.
“The high hospitalization rates in unvaccinated compared with vaccinated persons with and without a booster dose underscores the importance of COVID-19 vaccinations in preventing hospitalizations and suggests that increasing vaccination coverage, including booster dose coverage, can prevent hospitalizations, serious illness, and death,” the investigators wrote.
The study also revealed that vaccinated hospitalized patients were significantly older, on average, than unvaccinated hospitalized patients (median, 70 vs. 58 years; P < .001). They were also significantly more likely to have three or more underlying medical conditions (77.8% vs. 51.6%; P < .001)
“A greater proportion of hospitalized cases among vaccinated persons occurred in individuals with medical fragility who were older, more likely to reside in long-term care facilities, and have three or more underlying medical conditions, including immunosuppressive conditions,” the investigators wrote.
New variants outpacing data, vaccines remain essential
While data from April 2022 alone showed a 3.5-fold higher rate of hospitalization among unvaccinated versus vaccinated individuals with or without a booster, newer data suggest that emerging strains of Omicron are putting more people in the hospital.
A recent report by the CDC showed weekly hospitalization rates climbing from March 20 to May 31, 2022, which coincided with predominance of the newer Omicron BA.2 variant. While unvaccinated people were still around 3.5 times more likely to be hospitalized than vaccinated people, overall hospitalization rates jumped 3-fold for people 65 years and older, and 1.7-fold for adults younger than 65. Adding further complexity to this constantly evolving situation is that Omicron BA.2 has since been joined by the BA.4 and BA.5 lineages, for which vaccines are now available.
In the paper published in JAMA Internal Medicine, the CDC report, and in a comment for this article, the CDC offered the same take-home message: Get vaccinated.
“These findings reinforce previous research illustrating how vaccination provides protection from hospitalization due to COVID-19,” a CDC spokesperson said. “COVID-19 vaccines are proven to help prevent serious COVID-19 illness, and everyone ages 6 months and older should stay up to date with COVID-19 vaccines.”
The study published in JAMA Internal Medicine was supported by the CDC. The investigators disclosed additional relationships with Sanofi, GSK, MedImmune, and others.
FROM JAMA INTERNAL MEDICINE
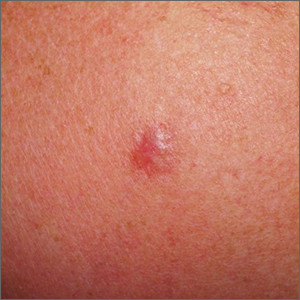
Shoulder lesion
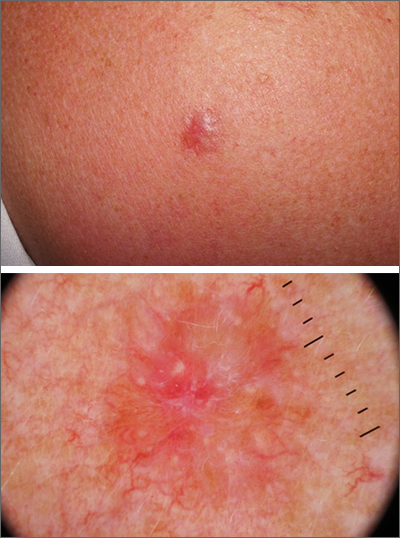
A punch biopsy of the lesion was performed and the results were consistent with a dermatofibroma, which is a benign growth.
Dermatofibromas may manifest as a pink papule on fair-skinned individuals or a darker brown papule in patients of color. Clinically, the texture can be helpful to discern an etiology—dermatofibromas may dimple when pinched laterally, while melanocytic nevi or melanomas tend to be somewhat softer on palpation. Cutaneous sarcoma, while exceedingly rare, may be firmer and chaotic, and varied with multiple colors and topographical changes.
The dermoscopic pattern of a dermatofibroma includes central scar-like areas, a peripheral pigment network, occasional shiny white lines, and confluent circular brown macules. Other less frequent dermoscopic structures may also be seen. A prospective study of the dermoscopic morphology of 412 dermatofibromas found 10 distinct dermoscopic patterns, but also noted that 25% of the dermatofibromas exhibited an atypical pattern.1 Atypical pigment, multiple scar-like areas, and dotted vessels can occur in a dermatofibroma, as well as in a Spitz nevus, and melanoma. Thus, such findings should prompt a biopsy.
Dermatofibromas are safe to observe, but they can be surgically excised if they cause pain or cosmetic concerns.
This patient was reassured to know that the lesion would not require surgical intervention and was unlikely to enlarge or change significantly over time.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained). Dr. Karnes is the medical director of MDFMR Dermatology Services, Augusta, ME.
1. Zaballos P, Puig S, Llambrich A, Malvehy J. Dermoscopy of dermatofibromas: a prospective morphological study of 412 cases. Arch Dermatol. 2008;144:75-83. doi: 10.1001/archdermatol.2007.8

A punch biopsy of the lesion was performed and the results were consistent with a dermatofibroma, which is a benign growth.
Dermatofibromas may manifest as a pink papule on fair-skinned individuals or a darker brown papule in patients of color. Clinically, the texture can be helpful to discern an etiology—dermatofibromas may dimple when pinched laterally, while melanocytic nevi or melanomas tend to be somewhat softer on palpation. Cutaneous sarcoma, while exceedingly rare, may be firmer and chaotic, and varied with multiple colors and topographical changes.
The dermoscopic pattern of a dermatofibroma includes central scar-like areas, a peripheral pigment network, occasional shiny white lines, and confluent circular brown macules. Other less frequent dermoscopic structures may also be seen. A prospective study of the dermoscopic morphology of 412 dermatofibromas found 10 distinct dermoscopic patterns, but also noted that 25% of the dermatofibromas exhibited an atypical pattern.1 Atypical pigment, multiple scar-like areas, and dotted vessels can occur in a dermatofibroma, as well as in a Spitz nevus, and melanoma. Thus, such findings should prompt a biopsy.
Dermatofibromas are safe to observe, but they can be surgically excised if they cause pain or cosmetic concerns.
This patient was reassured to know that the lesion would not require surgical intervention and was unlikely to enlarge or change significantly over time.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained). Dr. Karnes is the medical director of MDFMR Dermatology Services, Augusta, ME.

A punch biopsy of the lesion was performed and the results were consistent with a dermatofibroma, which is a benign growth.
Dermatofibromas may manifest as a pink papule on fair-skinned individuals or a darker brown papule in patients of color. Clinically, the texture can be helpful to discern an etiology—dermatofibromas may dimple when pinched laterally, while melanocytic nevi or melanomas tend to be somewhat softer on palpation. Cutaneous sarcoma, while exceedingly rare, may be firmer and chaotic, and varied with multiple colors and topographical changes.
The dermoscopic pattern of a dermatofibroma includes central scar-like areas, a peripheral pigment network, occasional shiny white lines, and confluent circular brown macules. Other less frequent dermoscopic structures may also be seen. A prospective study of the dermoscopic morphology of 412 dermatofibromas found 10 distinct dermoscopic patterns, but also noted that 25% of the dermatofibromas exhibited an atypical pattern.1 Atypical pigment, multiple scar-like areas, and dotted vessels can occur in a dermatofibroma, as well as in a Spitz nevus, and melanoma. Thus, such findings should prompt a biopsy.
Dermatofibromas are safe to observe, but they can be surgically excised if they cause pain or cosmetic concerns.
This patient was reassured to know that the lesion would not require surgical intervention and was unlikely to enlarge or change significantly over time.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained). Dr. Karnes is the medical director of MDFMR Dermatology Services, Augusta, ME.
1. Zaballos P, Puig S, Llambrich A, Malvehy J. Dermoscopy of dermatofibromas: a prospective morphological study of 412 cases. Arch Dermatol. 2008;144:75-83. doi: 10.1001/archdermatol.2007.8
1. Zaballos P, Puig S, Llambrich A, Malvehy J. Dermoscopy of dermatofibromas: a prospective morphological study of 412 cases. Arch Dermatol. 2008;144:75-83. doi: 10.1001/archdermatol.2007.8
Where a child eats breakfast is important
We’ve been told for decades that a child who doesn’t start the day with a good breakfast is entering school at a serious disadvantage. The brain needs a good supply of energy to learn optimally. So the standard wisdom goes. Subsidized school breakfast programs have been built around this chestnut. But, is there solid evidence to support the notion that simply adding a morning meal to a child’s schedule will improve his or her school performance? It sounds like common sense, but is it just one of those old grandmother’s nuggets that doesn’t stand up under close scrutiny?
A recent study from Spain suggests that the relationship between breakfast and school performance is not merely related to the nutritional needs of a growing brain. Using data from nearly 4,000 Spanish children aged 4-14 collected in a 2017 national health survey, the investigators found “skipping breakfast and eating breakfast out of the home were linked to greater odds of psychosocial behavioral problems than eating breakfast at home.” And, we already know that, in general, children who misbehave in school don’t thrive academically.
There were also associations between the absence or presence of certain food groups in the morning meal with behavioral problems. But the data lacked the granularity to draw any firm conclusions – although the authors felt that what they consider a healthy Spanish diet may have had a positive influence on behavior.
The findings in this study may simply be another example of the many positive influences that have been associated with family meals and have little to do with what is actually consumed. The association may not have much to do with the family gathering together at a single Norman Rockwell sitting, a reality that I suspect seldom occurs. The apparent positive influence of breakfast may be that it reflects a family’s priorities: that food is important, that sleep is important, and that school is important – so important that scheduling the morning should focus on sending the child off well prepared. The child who is allowed to stay up to an unhealthy hour is likely to be difficult to arouse in the morning for breakfast and getting off to school.
It may be that the child’s behavior problems are so disruptive and taxing for the family that even with their best efforts, the parents can’t find the time and energy to provide a breakfast in the home.
On the other hand, the study doesn’t tell us how many children aren’t offered breakfast at home because their families simply can’t afford it. Obviously, the answer depends on the socioeconomic mix of a given community. In some localities this may represent a sizable percentage of the population.
So where does this leave us? Unfortunately, as I read through the discussion at the end of this paper I felt that the authors were leaning too much toward further research based on the potential associations between behavior and specific food groups their data suggested.
For me, the take-home message from this paper is that our existing efforts to improve academic success with food offered in school should also include strategies that promote eating breakfast at home. For example, the backpack take-home food distribution programs that seem to have been effective could include breakfast-targeted items packaged in a way that encourage families to provide breakfast at home.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at [email protected].
We’ve been told for decades that a child who doesn’t start the day with a good breakfast is entering school at a serious disadvantage. The brain needs a good supply of energy to learn optimally. So the standard wisdom goes. Subsidized school breakfast programs have been built around this chestnut. But, is there solid evidence to support the notion that simply adding a morning meal to a child’s schedule will improve his or her school performance? It sounds like common sense, but is it just one of those old grandmother’s nuggets that doesn’t stand up under close scrutiny?
A recent study from Spain suggests that the relationship between breakfast and school performance is not merely related to the nutritional needs of a growing brain. Using data from nearly 4,000 Spanish children aged 4-14 collected in a 2017 national health survey, the investigators found “skipping breakfast and eating breakfast out of the home were linked to greater odds of psychosocial behavioral problems than eating breakfast at home.” And, we already know that, in general, children who misbehave in school don’t thrive academically.
There were also associations between the absence or presence of certain food groups in the morning meal with behavioral problems. But the data lacked the granularity to draw any firm conclusions – although the authors felt that what they consider a healthy Spanish diet may have had a positive influence on behavior.
The findings in this study may simply be another example of the many positive influences that have been associated with family meals and have little to do with what is actually consumed. The association may not have much to do with the family gathering together at a single Norman Rockwell sitting, a reality that I suspect seldom occurs. The apparent positive influence of breakfast may be that it reflects a family’s priorities: that food is important, that sleep is important, and that school is important – so important that scheduling the morning should focus on sending the child off well prepared. The child who is allowed to stay up to an unhealthy hour is likely to be difficult to arouse in the morning for breakfast and getting off to school.
It may be that the child’s behavior problems are so disruptive and taxing for the family that even with their best efforts, the parents can’t find the time and energy to provide a breakfast in the home.
On the other hand, the study doesn’t tell us how many children aren’t offered breakfast at home because their families simply can’t afford it. Obviously, the answer depends on the socioeconomic mix of a given community. In some localities this may represent a sizable percentage of the population.
So where does this leave us? Unfortunately, as I read through the discussion at the end of this paper I felt that the authors were leaning too much toward further research based on the potential associations between behavior and specific food groups their data suggested.
For me, the take-home message from this paper is that our existing efforts to improve academic success with food offered in school should also include strategies that promote eating breakfast at home. For example, the backpack take-home food distribution programs that seem to have been effective could include breakfast-targeted items packaged in a way that encourage families to provide breakfast at home.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at [email protected].
We’ve been told for decades that a child who doesn’t start the day with a good breakfast is entering school at a serious disadvantage. The brain needs a good supply of energy to learn optimally. So the standard wisdom goes. Subsidized school breakfast programs have been built around this chestnut. But, is there solid evidence to support the notion that simply adding a morning meal to a child’s schedule will improve his or her school performance? It sounds like common sense, but is it just one of those old grandmother’s nuggets that doesn’t stand up under close scrutiny?
A recent study from Spain suggests that the relationship between breakfast and school performance is not merely related to the nutritional needs of a growing brain. Using data from nearly 4,000 Spanish children aged 4-14 collected in a 2017 national health survey, the investigators found “skipping breakfast and eating breakfast out of the home were linked to greater odds of psychosocial behavioral problems than eating breakfast at home.” And, we already know that, in general, children who misbehave in school don’t thrive academically.
There were also associations between the absence or presence of certain food groups in the morning meal with behavioral problems. But the data lacked the granularity to draw any firm conclusions – although the authors felt that what they consider a healthy Spanish diet may have had a positive influence on behavior.
The findings in this study may simply be another example of the many positive influences that have been associated with family meals and have little to do with what is actually consumed. The association may not have much to do with the family gathering together at a single Norman Rockwell sitting, a reality that I suspect seldom occurs. The apparent positive influence of breakfast may be that it reflects a family’s priorities: that food is important, that sleep is important, and that school is important – so important that scheduling the morning should focus on sending the child off well prepared. The child who is allowed to stay up to an unhealthy hour is likely to be difficult to arouse in the morning for breakfast and getting off to school.
It may be that the child’s behavior problems are so disruptive and taxing for the family that even with their best efforts, the parents can’t find the time and energy to provide a breakfast in the home.
On the other hand, the study doesn’t tell us how many children aren’t offered breakfast at home because their families simply can’t afford it. Obviously, the answer depends on the socioeconomic mix of a given community. In some localities this may represent a sizable percentage of the population.
So where does this leave us? Unfortunately, as I read through the discussion at the end of this paper I felt that the authors were leaning too much toward further research based on the potential associations between behavior and specific food groups their data suggested.
For me, the take-home message from this paper is that our existing efforts to improve academic success with food offered in school should also include strategies that promote eating breakfast at home. For example, the backpack take-home food distribution programs that seem to have been effective could include breakfast-targeted items packaged in a way that encourage families to provide breakfast at home.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at [email protected].
Biomarker-guided steroid therapy shown safe for COPD
Eosinophil-guided corticosteroid therapy for patients with chronic obstructive pulmonary disease (COPD) is equivalent in efficacy to standard of care therapy, but the eosinophil-guided therapy may help mitigate the harmful side effects associated with even short courses of corticosteroids, investigators said in a primary care–based randomized trial.
Among patients in 14 primary care practices in the United Kingdom who experienced COPD exacerbations, the proportion of patients who experienced treatment failure at day 28 was 27% for those who were randomized to receive prednisolone only when blood eosinophil counts on a point-of-care assay equaled or exceeded 2%, compared with 34% of all patients randomized to standard of care.
The relative risk for treatment failure using the eosinophil-guided approach was 0.82, which did not reach statistical significance, but indicated noninferiority for the biomarker-based dosing method, Mona Bafadhel, MD, of King’s College London, reported on behalf of colleagues in the Stratified Treatment to Reduce Risk in COPD (STARR2) trial.
This is the largest primary care multicenter trial, and probably adds another 20% to the literature base for exacerbations in COPD,” she said in an oral abstract presentation at the European Respiratory Society 2022 Congress.
“A personalized endotype-based treatment with oral prednisolone is possible in patients with COPD and I think should be now part of clinical guidelines,” she added.
Too much of a good thing
Although systemic corticosteroids are the universal treatment for COPD exacerbations, the drugs are also known to increase harm, with studies showing that cumulative doses of oral corticosteroids in COPD patients is associated with an increased risk for death. In addition, systemic corticosteroids are the third most common cause of adverse events leading to hospitalization, behind only chemotherapy and antibiotic use leading to Clostridioides difficile infections, Dr. Bafadhel said.
“And of course, corticosteroids are associated with significant harmful effects, including a five-times increased risk of sepsis, three-times increased risk of [venous thromboembolism], and a twice-increased risk of fracture,” she said.
Dr. Bafadhel and colleagues had previously shown in the single-center BEAT-COPD study that peripheral blood eosinophils at the time of a moderate COPD exacerbation could be used to safely direct oral corticosteroid therapy. She also pointed to a 2019 multicenter open-label study showing that eosinophil-guided care was noninferior to standard prescribing of oral corticosteroids for patients with severe exacerbations.
Primary care study
The investigators conducted the current study to test whether eosinophil-guided therapy at the point of care in a primary practice setting was efficacious, with the ultimate goal of encouraging changes in guidelines.
They recruited patients with COPD exacerbations from 14 general practices in Oxfordshire and Buckinghamshire in the Thames Valley.
The patients were randomly assigned to receive either standard of care or the biomarker-guided intervention for 14 days. In this arm, patients with eosinophil counts of 2% or greater received matched prednisolone, while patients with counts below 2% received placebo. The patients were blinded to the assigned drug.
A total of 203 exacerbations among 152 patients were evenly allocated to treatment or control groups. The mean patient age was 71. Of the 102 exacerbations allocated to eosinophil-guided therapy, 34 were treated with placebo.
As noted before, in the intention-to-treat analysis the primary outcome of the treatment failure rate, defined as any need for antibiotics and/or steroids at one month, was 27% in the biomarker-guided arm and 34% in the standard care arm.
“In the per-protocol analysis we also demonstrated that there was a suggestion that there is possible superiority of using blood eosinophil-directed oral corticosteroid prescriptions at the time of acute exacerbation using the point-of-care eosinophil test,” Dr. Bafadhel said.
There were no significant differences in the secondary outcomes of mean change in forced expiratory volume in 1 second (FEV1), COPD Assessment Test scores from exacerbation to follow-up, and symptoms according to a visual analog scale.
Invited discussant Dave Singh, MD, of the University of Manchester, England, asked Dr. Bafadhel how the data she presented supported her conclusions about the potential benefits of eosinophil-guided therapy, given that the P values were nonsignificant.
“The primary outcome was powered on noninferiority, and of course what we’ve shown is that it’s not any worse, it’s not any better, but of course it’s the effect of how many courses of steroids you can reduce in that population,” Dr. Bafadhel replied.
She noted that although the investigators have not performed an economic analysis to determine how many adverse events might be avoided using the biomarker-guided approach, “we do know that some of these patients who are given prednisolone, their comorbidities of diabetes worsened, for example.”
In the online Q&A for the presentation, Sohail Ansari, MD, from the Mid and South Essex NHS Foundation Trust in the United Kingdom, said that many patients in primary care practices receive “rescue packs” containing antibiotics and steroids, but may not be equipped to know when they should use the steroids and therefore may overuse them.
“Perhaps community-based, adequately resourced respiratory teams [may] be a way forward, but it will need adequate investment and commitment,” he wrote.
The trial was supported by the University of Oxford and National Institute for Health and Care Research, UK. Dr. Bafadhel reported grant and research support from the National Institute for Health and Care Research, Asthma & Lung UK, AstraZeneca, and Roche, and honoraria or fees from others. Dr. Singh reported speaking fees, honoraria, and research grants from multiple companies. Dr. Ansari reported no conflicts of interest.
A version of this article first appeared on Medscape.com.
Eosinophil-guided corticosteroid therapy for patients with chronic obstructive pulmonary disease (COPD) is equivalent in efficacy to standard of care therapy, but the eosinophil-guided therapy may help mitigate the harmful side effects associated with even short courses of corticosteroids, investigators said in a primary care–based randomized trial.
Among patients in 14 primary care practices in the United Kingdom who experienced COPD exacerbations, the proportion of patients who experienced treatment failure at day 28 was 27% for those who were randomized to receive prednisolone only when blood eosinophil counts on a point-of-care assay equaled or exceeded 2%, compared with 34% of all patients randomized to standard of care.
The relative risk for treatment failure using the eosinophil-guided approach was 0.82, which did not reach statistical significance, but indicated noninferiority for the biomarker-based dosing method, Mona Bafadhel, MD, of King’s College London, reported on behalf of colleagues in the Stratified Treatment to Reduce Risk in COPD (STARR2) trial.
This is the largest primary care multicenter trial, and probably adds another 20% to the literature base for exacerbations in COPD,” she said in an oral abstract presentation at the European Respiratory Society 2022 Congress.
“A personalized endotype-based treatment with oral prednisolone is possible in patients with COPD and I think should be now part of clinical guidelines,” she added.
Too much of a good thing
Although systemic corticosteroids are the universal treatment for COPD exacerbations, the drugs are also known to increase harm, with studies showing that cumulative doses of oral corticosteroids in COPD patients is associated with an increased risk for death. In addition, systemic corticosteroids are the third most common cause of adverse events leading to hospitalization, behind only chemotherapy and antibiotic use leading to Clostridioides difficile infections, Dr. Bafadhel said.
“And of course, corticosteroids are associated with significant harmful effects, including a five-times increased risk of sepsis, three-times increased risk of [venous thromboembolism], and a twice-increased risk of fracture,” she said.
Dr. Bafadhel and colleagues had previously shown in the single-center BEAT-COPD study that peripheral blood eosinophils at the time of a moderate COPD exacerbation could be used to safely direct oral corticosteroid therapy. She also pointed to a 2019 multicenter open-label study showing that eosinophil-guided care was noninferior to standard prescribing of oral corticosteroids for patients with severe exacerbations.
Primary care study
The investigators conducted the current study to test whether eosinophil-guided therapy at the point of care in a primary practice setting was efficacious, with the ultimate goal of encouraging changes in guidelines.
They recruited patients with COPD exacerbations from 14 general practices in Oxfordshire and Buckinghamshire in the Thames Valley.
The patients were randomly assigned to receive either standard of care or the biomarker-guided intervention for 14 days. In this arm, patients with eosinophil counts of 2% or greater received matched prednisolone, while patients with counts below 2% received placebo. The patients were blinded to the assigned drug.
A total of 203 exacerbations among 152 patients were evenly allocated to treatment or control groups. The mean patient age was 71. Of the 102 exacerbations allocated to eosinophil-guided therapy, 34 were treated with placebo.
As noted before, in the intention-to-treat analysis the primary outcome of the treatment failure rate, defined as any need for antibiotics and/or steroids at one month, was 27% in the biomarker-guided arm and 34% in the standard care arm.
“In the per-protocol analysis we also demonstrated that there was a suggestion that there is possible superiority of using blood eosinophil-directed oral corticosteroid prescriptions at the time of acute exacerbation using the point-of-care eosinophil test,” Dr. Bafadhel said.
There were no significant differences in the secondary outcomes of mean change in forced expiratory volume in 1 second (FEV1), COPD Assessment Test scores from exacerbation to follow-up, and symptoms according to a visual analog scale.
Invited discussant Dave Singh, MD, of the University of Manchester, England, asked Dr. Bafadhel how the data she presented supported her conclusions about the potential benefits of eosinophil-guided therapy, given that the P values were nonsignificant.
“The primary outcome was powered on noninferiority, and of course what we’ve shown is that it’s not any worse, it’s not any better, but of course it’s the effect of how many courses of steroids you can reduce in that population,” Dr. Bafadhel replied.
She noted that although the investigators have not performed an economic analysis to determine how many adverse events might be avoided using the biomarker-guided approach, “we do know that some of these patients who are given prednisolone, their comorbidities of diabetes worsened, for example.”
In the online Q&A for the presentation, Sohail Ansari, MD, from the Mid and South Essex NHS Foundation Trust in the United Kingdom, said that many patients in primary care practices receive “rescue packs” containing antibiotics and steroids, but may not be equipped to know when they should use the steroids and therefore may overuse them.
“Perhaps community-based, adequately resourced respiratory teams [may] be a way forward, but it will need adequate investment and commitment,” he wrote.
The trial was supported by the University of Oxford and National Institute for Health and Care Research, UK. Dr. Bafadhel reported grant and research support from the National Institute for Health and Care Research, Asthma & Lung UK, AstraZeneca, and Roche, and honoraria or fees from others. Dr. Singh reported speaking fees, honoraria, and research grants from multiple companies. Dr. Ansari reported no conflicts of interest.
A version of this article first appeared on Medscape.com.
Eosinophil-guided corticosteroid therapy for patients with chronic obstructive pulmonary disease (COPD) is equivalent in efficacy to standard of care therapy, but the eosinophil-guided therapy may help mitigate the harmful side effects associated with even short courses of corticosteroids, investigators said in a primary care–based randomized trial.
Among patients in 14 primary care practices in the United Kingdom who experienced COPD exacerbations, the proportion of patients who experienced treatment failure at day 28 was 27% for those who were randomized to receive prednisolone only when blood eosinophil counts on a point-of-care assay equaled or exceeded 2%, compared with 34% of all patients randomized to standard of care.
The relative risk for treatment failure using the eosinophil-guided approach was 0.82, which did not reach statistical significance, but indicated noninferiority for the biomarker-based dosing method, Mona Bafadhel, MD, of King’s College London, reported on behalf of colleagues in the Stratified Treatment to Reduce Risk in COPD (STARR2) trial.
This is the largest primary care multicenter trial, and probably adds another 20% to the literature base for exacerbations in COPD,” she said in an oral abstract presentation at the European Respiratory Society 2022 Congress.
“A personalized endotype-based treatment with oral prednisolone is possible in patients with COPD and I think should be now part of clinical guidelines,” she added.
Too much of a good thing
Although systemic corticosteroids are the universal treatment for COPD exacerbations, the drugs are also known to increase harm, with studies showing that cumulative doses of oral corticosteroids in COPD patients is associated with an increased risk for death. In addition, systemic corticosteroids are the third most common cause of adverse events leading to hospitalization, behind only chemotherapy and antibiotic use leading to Clostridioides difficile infections, Dr. Bafadhel said.
“And of course, corticosteroids are associated with significant harmful effects, including a five-times increased risk of sepsis, three-times increased risk of [venous thromboembolism], and a twice-increased risk of fracture,” she said.
Dr. Bafadhel and colleagues had previously shown in the single-center BEAT-COPD study that peripheral blood eosinophils at the time of a moderate COPD exacerbation could be used to safely direct oral corticosteroid therapy. She also pointed to a 2019 multicenter open-label study showing that eosinophil-guided care was noninferior to standard prescribing of oral corticosteroids for patients with severe exacerbations.
Primary care study
The investigators conducted the current study to test whether eosinophil-guided therapy at the point of care in a primary practice setting was efficacious, with the ultimate goal of encouraging changes in guidelines.
They recruited patients with COPD exacerbations from 14 general practices in Oxfordshire and Buckinghamshire in the Thames Valley.
The patients were randomly assigned to receive either standard of care or the biomarker-guided intervention for 14 days. In this arm, patients with eosinophil counts of 2% or greater received matched prednisolone, while patients with counts below 2% received placebo. The patients were blinded to the assigned drug.
A total of 203 exacerbations among 152 patients were evenly allocated to treatment or control groups. The mean patient age was 71. Of the 102 exacerbations allocated to eosinophil-guided therapy, 34 were treated with placebo.
As noted before, in the intention-to-treat analysis the primary outcome of the treatment failure rate, defined as any need for antibiotics and/or steroids at one month, was 27% in the biomarker-guided arm and 34% in the standard care arm.
“In the per-protocol analysis we also demonstrated that there was a suggestion that there is possible superiority of using blood eosinophil-directed oral corticosteroid prescriptions at the time of acute exacerbation using the point-of-care eosinophil test,” Dr. Bafadhel said.
There were no significant differences in the secondary outcomes of mean change in forced expiratory volume in 1 second (FEV1), COPD Assessment Test scores from exacerbation to follow-up, and symptoms according to a visual analog scale.
Invited discussant Dave Singh, MD, of the University of Manchester, England, asked Dr. Bafadhel how the data she presented supported her conclusions about the potential benefits of eosinophil-guided therapy, given that the P values were nonsignificant.
“The primary outcome was powered on noninferiority, and of course what we’ve shown is that it’s not any worse, it’s not any better, but of course it’s the effect of how many courses of steroids you can reduce in that population,” Dr. Bafadhel replied.
She noted that although the investigators have not performed an economic analysis to determine how many adverse events might be avoided using the biomarker-guided approach, “we do know that some of these patients who are given prednisolone, their comorbidities of diabetes worsened, for example.”
In the online Q&A for the presentation, Sohail Ansari, MD, from the Mid and South Essex NHS Foundation Trust in the United Kingdom, said that many patients in primary care practices receive “rescue packs” containing antibiotics and steroids, but may not be equipped to know when they should use the steroids and therefore may overuse them.
“Perhaps community-based, adequately resourced respiratory teams [may] be a way forward, but it will need adequate investment and commitment,” he wrote.
The trial was supported by the University of Oxford and National Institute for Health and Care Research, UK. Dr. Bafadhel reported grant and research support from the National Institute for Health and Care Research, Asthma & Lung UK, AstraZeneca, and Roche, and honoraria or fees from others. Dr. Singh reported speaking fees, honoraria, and research grants from multiple companies. Dr. Ansari reported no conflicts of interest.
A version of this article first appeared on Medscape.com.
FROM ERS 2022 CONGRESS
RETHINC takes air out of COPD-like therapy for smokers
Current or former smokers who have clinically significant respiratory symptoms but no spirometric evidence of airway obstruction are often treated with dual bronchodilators commonly prescribed for patients with chronic obstructive pulmonary disease (COPD).
But as results of the randomized RETHINC (Redefining Therapy In Early COPD for the Pulmonary Trials Cooperative) trial showed, bronchodilator therapy was no better than placebo at reducing respiratory symptoms in smokers, reported MeiLan K. Han, MD, from the University of Michigan, Ann Arbor, on behalf of colleagues in the RETHINC study group.
“Many tobacco-exposed symptomatic individuals are currently being treated. We don’t know if this is because physicians just aren’t doing spirometry and assuming COPD or they strongly believe that there’s a benefit, but the bottom line is that we really need to do spirometry to understand who benefits from bronchodilators, and we need further research to understand how to treat this specific group of patients because there truly is pathogenesis and disease burden,” Dr. Han said in an oral abstract presentation at the annual congress of the European Respiratory Society.
The study results were also published online in the New England Journal of Medicine to coincide with the presentation.
In an editorial accompanying the study, Don D. Sin, MD, MPH, from the University of British Columbia, Vancouver, commented that these medications should most likely be reserved for patients with COPD who have clinically significant airflow limitation,” and that “respiratory symptoms in tobacco-exposed persons are common but are highly variable over time.”
Dave Singh, MD, from the University of Manchester (England), the invited discussant, called it “a very important negative study.”
Not up to GOLD standard
Current or former smokers who are symptomatic, with COPD Assessment Test (CAT) scores of at least 10 despite having preserved function on spirometry, have been shown to have higher prospective rates of respiratory disease exacerbations and increased sputum total mucin concentrations. Approximately 43% of such patients are treated with bronchodilators, and 23% are treated with inhaled corticosteroids (ICS), Dr. Han noted.
Her group hypothesized that ever-smokers with spirometric values that fall within the normal range – that is, a postbronchodilator FEV1/FVC ratio of 70 or greater – would still derive benefit from long-acting bronchodilator therapy, even though these patients are currently excluded from Global Initiative for Chronic Obstructive Lung Disease (GOLD) recommendations.
To test this, they conducted a 12-week, multicenter, randomized, parallel-group study in which patients were assigned to receive either indacaterol (27.5 mcg) and glycopyrrolate (15.6 mcg) inhaled twice daily or placebo.
They enrolled adults aged 40-80 years with a minimum of 10 pack-years of smoking history, postbronchodilator FEV1/FVC ratio of 70 or greater, and CAT scores of 10 or greater. Patients with known concomitant lung disease, a primary diagnosis of asthma, or body mass index lower than 15 or higher than 40 and those being concomitantly treated with long-acting beta2-agonists or muscarinic antagonists or a short-acting combination were excluded, although patients were allowed to be on a short-acting beta-agonist.
A total of 535 participants were randomized, but COVID-19 pandemic–imposed obstacles resulted in a modified intention-to-treat population of 277 patients assigned to receive the active treatment and 244 assigned to receive placebo.
There was no difference between the groups for the primary outcome of an at least 4-point decrease in St. George’s Respiratory Questionnaire scores in patients who did not experience treatment failure, defined as an increase in respiratory symptoms requiring treatment with active long-acting bronchodilators or ICS.
The primary endpoint was seen in 56.4% of patients in the bronchodilator group, and 59% of controls.
Although there was greater improvement in pulmonary function from baseline in the treatment group, compared with the placebo group, the improvements did not correlate with similar improvements in symptoms, Dr. Han said.
There were 4 serious adverse events in the bronchodilator group and 11 in the placebo group, but none of the events were deemed to be related to the assigned treatments.
Dr. Han acknowledged limitations of the study, which may have included symptoms driven by other factors such as cardiac disease, suggesting that if such patients had been identified and excluded, a stronger effect might have been seen for the active treatment.
In addition, the study was underpowered to look at the subgroup of participants with chronic bronchitis, and the 12 weeks of the study may have been too short to see improvements in symptoms.
In his editorial, Dr. Sin noted that the study showed that cough and sputum production rather than exertion dyspnea are the primary symptoms among ever-smokers.
“Although bronchodilators are effective in ameliorating breathlessness and improving exercise tolerance, they are generally ineffective for cough,” he wrote. “Existing drugs for the treatment of COPD, such as inhaled glucocorticoids or phosphodiesterase-4 inhibitors, or new therapeutics such as P2X3 receptor antagonists may be more effective for the treatment of cough and sputum production related to smoking and could be considered for future evaluations in this patient population.”
The study was supported by the National Heart, Lung, and Blood Institute, the National Center for Advancing Translational Sciences, and Sunovion Pharmaceuticals. Novartis Pharmaceuticals donated the trial medication and placebo. Dr. Han disclosed grant/research support and honoraria or consulting fees from various companies. Dr. Singh reported speaking fees, honoraria, and research grants from multiple companies. Dr. Sin reported having no conflicts of interest to disclose.
A version of this article first appeared on Medscape.com.
Current or former smokers who have clinically significant respiratory symptoms but no spirometric evidence of airway obstruction are often treated with dual bronchodilators commonly prescribed for patients with chronic obstructive pulmonary disease (COPD).
But as results of the randomized RETHINC (Redefining Therapy In Early COPD for the Pulmonary Trials Cooperative) trial showed, bronchodilator therapy was no better than placebo at reducing respiratory symptoms in smokers, reported MeiLan K. Han, MD, from the University of Michigan, Ann Arbor, on behalf of colleagues in the RETHINC study group.
“Many tobacco-exposed symptomatic individuals are currently being treated. We don’t know if this is because physicians just aren’t doing spirometry and assuming COPD or they strongly believe that there’s a benefit, but the bottom line is that we really need to do spirometry to understand who benefits from bronchodilators, and we need further research to understand how to treat this specific group of patients because there truly is pathogenesis and disease burden,” Dr. Han said in an oral abstract presentation at the annual congress of the European Respiratory Society.
The study results were also published online in the New England Journal of Medicine to coincide with the presentation.
In an editorial accompanying the study, Don D. Sin, MD, MPH, from the University of British Columbia, Vancouver, commented that these medications should most likely be reserved for patients with COPD who have clinically significant airflow limitation,” and that “respiratory symptoms in tobacco-exposed persons are common but are highly variable over time.”
Dave Singh, MD, from the University of Manchester (England), the invited discussant, called it “a very important negative study.”
Not up to GOLD standard
Current or former smokers who are symptomatic, with COPD Assessment Test (CAT) scores of at least 10 despite having preserved function on spirometry, have been shown to have higher prospective rates of respiratory disease exacerbations and increased sputum total mucin concentrations. Approximately 43% of such patients are treated with bronchodilators, and 23% are treated with inhaled corticosteroids (ICS), Dr. Han noted.
Her group hypothesized that ever-smokers with spirometric values that fall within the normal range – that is, a postbronchodilator FEV1/FVC ratio of 70 or greater – would still derive benefit from long-acting bronchodilator therapy, even though these patients are currently excluded from Global Initiative for Chronic Obstructive Lung Disease (GOLD) recommendations.
To test this, they conducted a 12-week, multicenter, randomized, parallel-group study in which patients were assigned to receive either indacaterol (27.5 mcg) and glycopyrrolate (15.6 mcg) inhaled twice daily or placebo.
They enrolled adults aged 40-80 years with a minimum of 10 pack-years of smoking history, postbronchodilator FEV1/FVC ratio of 70 or greater, and CAT scores of 10 or greater. Patients with known concomitant lung disease, a primary diagnosis of asthma, or body mass index lower than 15 or higher than 40 and those being concomitantly treated with long-acting beta2-agonists or muscarinic antagonists or a short-acting combination were excluded, although patients were allowed to be on a short-acting beta-agonist.
A total of 535 participants were randomized, but COVID-19 pandemic–imposed obstacles resulted in a modified intention-to-treat population of 277 patients assigned to receive the active treatment and 244 assigned to receive placebo.
There was no difference between the groups for the primary outcome of an at least 4-point decrease in St. George’s Respiratory Questionnaire scores in patients who did not experience treatment failure, defined as an increase in respiratory symptoms requiring treatment with active long-acting bronchodilators or ICS.
The primary endpoint was seen in 56.4% of patients in the bronchodilator group, and 59% of controls.
Although there was greater improvement in pulmonary function from baseline in the treatment group, compared with the placebo group, the improvements did not correlate with similar improvements in symptoms, Dr. Han said.
There were 4 serious adverse events in the bronchodilator group and 11 in the placebo group, but none of the events were deemed to be related to the assigned treatments.
Dr. Han acknowledged limitations of the study, which may have included symptoms driven by other factors such as cardiac disease, suggesting that if such patients had been identified and excluded, a stronger effect might have been seen for the active treatment.
In addition, the study was underpowered to look at the subgroup of participants with chronic bronchitis, and the 12 weeks of the study may have been too short to see improvements in symptoms.
In his editorial, Dr. Sin noted that the study showed that cough and sputum production rather than exertion dyspnea are the primary symptoms among ever-smokers.
“Although bronchodilators are effective in ameliorating breathlessness and improving exercise tolerance, they are generally ineffective for cough,” he wrote. “Existing drugs for the treatment of COPD, such as inhaled glucocorticoids or phosphodiesterase-4 inhibitors, or new therapeutics such as P2X3 receptor antagonists may be more effective for the treatment of cough and sputum production related to smoking and could be considered for future evaluations in this patient population.”
The study was supported by the National Heart, Lung, and Blood Institute, the National Center for Advancing Translational Sciences, and Sunovion Pharmaceuticals. Novartis Pharmaceuticals donated the trial medication and placebo. Dr. Han disclosed grant/research support and honoraria or consulting fees from various companies. Dr. Singh reported speaking fees, honoraria, and research grants from multiple companies. Dr. Sin reported having no conflicts of interest to disclose.
A version of this article first appeared on Medscape.com.
Current or former smokers who have clinically significant respiratory symptoms but no spirometric evidence of airway obstruction are often treated with dual bronchodilators commonly prescribed for patients with chronic obstructive pulmonary disease (COPD).
But as results of the randomized RETHINC (Redefining Therapy In Early COPD for the Pulmonary Trials Cooperative) trial showed, bronchodilator therapy was no better than placebo at reducing respiratory symptoms in smokers, reported MeiLan K. Han, MD, from the University of Michigan, Ann Arbor, on behalf of colleagues in the RETHINC study group.
“Many tobacco-exposed symptomatic individuals are currently being treated. We don’t know if this is because physicians just aren’t doing spirometry and assuming COPD or they strongly believe that there’s a benefit, but the bottom line is that we really need to do spirometry to understand who benefits from bronchodilators, and we need further research to understand how to treat this specific group of patients because there truly is pathogenesis and disease burden,” Dr. Han said in an oral abstract presentation at the annual congress of the European Respiratory Society.
The study results were also published online in the New England Journal of Medicine to coincide with the presentation.
In an editorial accompanying the study, Don D. Sin, MD, MPH, from the University of British Columbia, Vancouver, commented that these medications should most likely be reserved for patients with COPD who have clinically significant airflow limitation,” and that “respiratory symptoms in tobacco-exposed persons are common but are highly variable over time.”
Dave Singh, MD, from the University of Manchester (England), the invited discussant, called it “a very important negative study.”
Not up to GOLD standard
Current or former smokers who are symptomatic, with COPD Assessment Test (CAT) scores of at least 10 despite having preserved function on spirometry, have been shown to have higher prospective rates of respiratory disease exacerbations and increased sputum total mucin concentrations. Approximately 43% of such patients are treated with bronchodilators, and 23% are treated with inhaled corticosteroids (ICS), Dr. Han noted.
Her group hypothesized that ever-smokers with spirometric values that fall within the normal range – that is, a postbronchodilator FEV1/FVC ratio of 70 or greater – would still derive benefit from long-acting bronchodilator therapy, even though these patients are currently excluded from Global Initiative for Chronic Obstructive Lung Disease (GOLD) recommendations.
To test this, they conducted a 12-week, multicenter, randomized, parallel-group study in which patients were assigned to receive either indacaterol (27.5 mcg) and glycopyrrolate (15.6 mcg) inhaled twice daily or placebo.
They enrolled adults aged 40-80 years with a minimum of 10 pack-years of smoking history, postbronchodilator FEV1/FVC ratio of 70 or greater, and CAT scores of 10 or greater. Patients with known concomitant lung disease, a primary diagnosis of asthma, or body mass index lower than 15 or higher than 40 and those being concomitantly treated with long-acting beta2-agonists or muscarinic antagonists or a short-acting combination were excluded, although patients were allowed to be on a short-acting beta-agonist.
A total of 535 participants were randomized, but COVID-19 pandemic–imposed obstacles resulted in a modified intention-to-treat population of 277 patients assigned to receive the active treatment and 244 assigned to receive placebo.
There was no difference between the groups for the primary outcome of an at least 4-point decrease in St. George’s Respiratory Questionnaire scores in patients who did not experience treatment failure, defined as an increase in respiratory symptoms requiring treatment with active long-acting bronchodilators or ICS.
The primary endpoint was seen in 56.4% of patients in the bronchodilator group, and 59% of controls.
Although there was greater improvement in pulmonary function from baseline in the treatment group, compared with the placebo group, the improvements did not correlate with similar improvements in symptoms, Dr. Han said.
There were 4 serious adverse events in the bronchodilator group and 11 in the placebo group, but none of the events were deemed to be related to the assigned treatments.
Dr. Han acknowledged limitations of the study, which may have included symptoms driven by other factors such as cardiac disease, suggesting that if such patients had been identified and excluded, a stronger effect might have been seen for the active treatment.
In addition, the study was underpowered to look at the subgroup of participants with chronic bronchitis, and the 12 weeks of the study may have been too short to see improvements in symptoms.
In his editorial, Dr. Sin noted that the study showed that cough and sputum production rather than exertion dyspnea are the primary symptoms among ever-smokers.
“Although bronchodilators are effective in ameliorating breathlessness and improving exercise tolerance, they are generally ineffective for cough,” he wrote. “Existing drugs for the treatment of COPD, such as inhaled glucocorticoids or phosphodiesterase-4 inhibitors, or new therapeutics such as P2X3 receptor antagonists may be more effective for the treatment of cough and sputum production related to smoking and could be considered for future evaluations in this patient population.”
The study was supported by the National Heart, Lung, and Blood Institute, the National Center for Advancing Translational Sciences, and Sunovion Pharmaceuticals. Novartis Pharmaceuticals donated the trial medication and placebo. Dr. Han disclosed grant/research support and honoraria or consulting fees from various companies. Dr. Singh reported speaking fees, honoraria, and research grants from multiple companies. Dr. Sin reported having no conflicts of interest to disclose.
A version of this article first appeared on Medscape.com.
FROM ERS 2022
Children and COVID: Weekly cases close out August with a second straight increase
New cases rose by 4.6% for the week of Aug. 26 to Sept. 1, following a week in which cases increased by almost 9%, as the second half of August basically reversed the two consecutive weeks of decreases during the first half of the month, based on the AAP/CHA data collected from state and territorial health departments.
Similar trends can be seen for emergency department visits, with the exception of children aged 0-11 years, whose ED visit rates have continued to fall since late July. Children aged 12-15, however, had a 7-day average of 4.4% of ED visits with diagnosed COVID on Aug. 25, compared with 3.1% for Aug. 12. Children aged 16-17 years were at 3.4% on Aug. 27, compared with 3.1% as late as Aug. 15, the Centers for Disease Control and Prevention reported.
Hospital admissions with confirmed COVID-19, reported only for children aged 0-17 years, also reflect the late-August trend of increased cases. New hospitalizations dropped from 0.46 per 100,000 population on July 30 to 0.40 per 100,000 on Aug. 19 but have since risen to 0.44 per 100,000 as of Aug. 27, the CDC said on its COVID Data Tracker.
Initial vaccinations, meanwhile, have declined since early August for all children, according to a separate report from the AAP. A look at CDC data for two specific days – the first and last Mondays of the month – shows that those aged under 5 received 12,982 doses on Aug. 1, compared with 5,824 doses on Aug. 29. Over that same time, initial vaccinations in 5- to 11-year-olds went from 9,058 to 2,879, while among those aged 12-17 they dropped from 4,245 to 1,226.
Cumulatively, 5.5% of all children under age 5 had received at least one dose and 1.3% were fully vaccinated by Aug. 30, compared with 38.1% and 30.7%, respectively, of those aged 5-11 and 70.7% and 60.5% of 12- to 17-year-olds, the CDC said.
New cases rose by 4.6% for the week of Aug. 26 to Sept. 1, following a week in which cases increased by almost 9%, as the second half of August basically reversed the two consecutive weeks of decreases during the first half of the month, based on the AAP/CHA data collected from state and territorial health departments.
Similar trends can be seen for emergency department visits, with the exception of children aged 0-11 years, whose ED visit rates have continued to fall since late July. Children aged 12-15, however, had a 7-day average of 4.4% of ED visits with diagnosed COVID on Aug. 25, compared with 3.1% for Aug. 12. Children aged 16-17 years were at 3.4% on Aug. 27, compared with 3.1% as late as Aug. 15, the Centers for Disease Control and Prevention reported.
Hospital admissions with confirmed COVID-19, reported only for children aged 0-17 years, also reflect the late-August trend of increased cases. New hospitalizations dropped from 0.46 per 100,000 population on July 30 to 0.40 per 100,000 on Aug. 19 but have since risen to 0.44 per 100,000 as of Aug. 27, the CDC said on its COVID Data Tracker.
Initial vaccinations, meanwhile, have declined since early August for all children, according to a separate report from the AAP. A look at CDC data for two specific days – the first and last Mondays of the month – shows that those aged under 5 received 12,982 doses on Aug. 1, compared with 5,824 doses on Aug. 29. Over that same time, initial vaccinations in 5- to 11-year-olds went from 9,058 to 2,879, while among those aged 12-17 they dropped from 4,245 to 1,226.
Cumulatively, 5.5% of all children under age 5 had received at least one dose and 1.3% were fully vaccinated by Aug. 30, compared with 38.1% and 30.7%, respectively, of those aged 5-11 and 70.7% and 60.5% of 12- to 17-year-olds, the CDC said.
New cases rose by 4.6% for the week of Aug. 26 to Sept. 1, following a week in which cases increased by almost 9%, as the second half of August basically reversed the two consecutive weeks of decreases during the first half of the month, based on the AAP/CHA data collected from state and territorial health departments.
Similar trends can be seen for emergency department visits, with the exception of children aged 0-11 years, whose ED visit rates have continued to fall since late July. Children aged 12-15, however, had a 7-day average of 4.4% of ED visits with diagnosed COVID on Aug. 25, compared with 3.1% for Aug. 12. Children aged 16-17 years were at 3.4% on Aug. 27, compared with 3.1% as late as Aug. 15, the Centers for Disease Control and Prevention reported.
Hospital admissions with confirmed COVID-19, reported only for children aged 0-17 years, also reflect the late-August trend of increased cases. New hospitalizations dropped from 0.46 per 100,000 population on July 30 to 0.40 per 100,000 on Aug. 19 but have since risen to 0.44 per 100,000 as of Aug. 27, the CDC said on its COVID Data Tracker.
Initial vaccinations, meanwhile, have declined since early August for all children, according to a separate report from the AAP. A look at CDC data for two specific days – the first and last Mondays of the month – shows that those aged under 5 received 12,982 doses on Aug. 1, compared with 5,824 doses on Aug. 29. Over that same time, initial vaccinations in 5- to 11-year-olds went from 9,058 to 2,879, while among those aged 12-17 they dropped from 4,245 to 1,226.
Cumulatively, 5.5% of all children under age 5 had received at least one dose and 1.3% were fully vaccinated by Aug. 30, compared with 38.1% and 30.7%, respectively, of those aged 5-11 and 70.7% and 60.5% of 12- to 17-year-olds, the CDC said.