User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
nav[contains(@class, 'nav-ce-stack nav-ce-stack__large-screen')]
header[@id='header']
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'main-prefix')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
div[contains(@class, 'view-medstat-quiz-listing-panes')]
div[contains(@class, 'pane-article-sidebar-latest-news')]
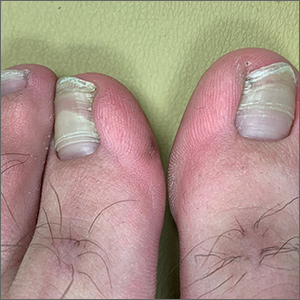
Elective surgery should be delayed 7 weeks after COVID-19 infection for unvaccinated patients, statement recommends
.
For patients fully vaccinated against COVID-19 with breakthrough infections, there is no consensus on how vaccination affects the time between COVID-19 infection and elective surgery. Clinicians should use their clinical judgment to schedule procedures, said Randall M. Clark, MD, president of the American Society of Anesthesiologists (ASA). “We need all physicians, anesthesiologists, surgeons, and others to base their decision to go ahead with elective surgery on the patient’s symptoms, their need for the procedure, and whether delays could cause other problems with their health,” he said in an interview.
Prior to these updated recommendations, which were published Feb. 22, the ASA and the APSF recommended a 4-week gap between COVID-19 diagnosis and elective surgery for asymptomatic or mild cases, regardless of a patient’s vaccination status.
Extending the wait time from 4 to 7 weeks was based on a multination study conducted in October 2020 following more than 140,000 surgical patients. Patients with previous COVID-19 infection had an increased risk for complications and death in elective surgery for up to 6 weeks following their diagnosis, compared with patients without COVID-19. Additional research in the United States found that patients with a preoperative COVID diagnosis were at higher risk for postoperative complications of respiratory failure for up to 4 weeks after diagnosis and postoperative pneumonia complications for up to 8 weeks after diagnosis.
Because these studies were conducted in unvaccinated populations or those with low vaccination rates, and preliminary data suggest vaccinated patients with breakthrough infections may have a lower risk for complications and death postinfection, “we felt that it was prudent to just make recommendations specific to unvaccinated patients,” Dr. Clark added.
Although this guidance is “very helpful” in that it summarizes the currently available research to give evidence-based recommendations, the 7-week wait time is a “very conservative estimate,” Brent Matthews, MD, surgeon-in-chief of the surgery care division of Atrium Health, Charlotte, N.C., told this news organization. At Atrium Health, surgery is scheduled at least 21 days after a patient’s COVID-19 diagnosis, regardless of their vaccination status, Dr. Matthews said.
The studies currently available were conducted earlier in the pandemic, when a different variant was prevalent, Dr. Matthews explained. The Omicron variant is currently the most prevalent COVID-19 variant and is less virulent than earlier strains of the virus. The joint statement does note that there is currently “no robust data” on patients infected with the Delta or Omicron variants of COVID-19, and that “the Omicron variant causes less severe disease and is more likely to reside in the oro- and nasopharynx without infiltration and damage to the lungs.”
Still, the new recommendations are a reminder to re-evaluate the potential complications from surgery for previously infected patients and to consider what comorbidities might make them more vulnerable, Dr. Matthews said. “The real power of the joint statement is to get people to ensure that they make an assessment of every patient that comes in front of them who has had a recent positive COVID test.”
A version of this article first appeared on Medscape.com.
.
For patients fully vaccinated against COVID-19 with breakthrough infections, there is no consensus on how vaccination affects the time between COVID-19 infection and elective surgery. Clinicians should use their clinical judgment to schedule procedures, said Randall M. Clark, MD, president of the American Society of Anesthesiologists (ASA). “We need all physicians, anesthesiologists, surgeons, and others to base their decision to go ahead with elective surgery on the patient’s symptoms, their need for the procedure, and whether delays could cause other problems with their health,” he said in an interview.
Prior to these updated recommendations, which were published Feb. 22, the ASA and the APSF recommended a 4-week gap between COVID-19 diagnosis and elective surgery for asymptomatic or mild cases, regardless of a patient’s vaccination status.
Extending the wait time from 4 to 7 weeks was based on a multination study conducted in October 2020 following more than 140,000 surgical patients. Patients with previous COVID-19 infection had an increased risk for complications and death in elective surgery for up to 6 weeks following their diagnosis, compared with patients without COVID-19. Additional research in the United States found that patients with a preoperative COVID diagnosis were at higher risk for postoperative complications of respiratory failure for up to 4 weeks after diagnosis and postoperative pneumonia complications for up to 8 weeks after diagnosis.
Because these studies were conducted in unvaccinated populations or those with low vaccination rates, and preliminary data suggest vaccinated patients with breakthrough infections may have a lower risk for complications and death postinfection, “we felt that it was prudent to just make recommendations specific to unvaccinated patients,” Dr. Clark added.
Although this guidance is “very helpful” in that it summarizes the currently available research to give evidence-based recommendations, the 7-week wait time is a “very conservative estimate,” Brent Matthews, MD, surgeon-in-chief of the surgery care division of Atrium Health, Charlotte, N.C., told this news organization. At Atrium Health, surgery is scheduled at least 21 days after a patient’s COVID-19 diagnosis, regardless of their vaccination status, Dr. Matthews said.
The studies currently available were conducted earlier in the pandemic, when a different variant was prevalent, Dr. Matthews explained. The Omicron variant is currently the most prevalent COVID-19 variant and is less virulent than earlier strains of the virus. The joint statement does note that there is currently “no robust data” on patients infected with the Delta or Omicron variants of COVID-19, and that “the Omicron variant causes less severe disease and is more likely to reside in the oro- and nasopharynx without infiltration and damage to the lungs.”
Still, the new recommendations are a reminder to re-evaluate the potential complications from surgery for previously infected patients and to consider what comorbidities might make them more vulnerable, Dr. Matthews said. “The real power of the joint statement is to get people to ensure that they make an assessment of every patient that comes in front of them who has had a recent positive COVID test.”
A version of this article first appeared on Medscape.com.
.
For patients fully vaccinated against COVID-19 with breakthrough infections, there is no consensus on how vaccination affects the time between COVID-19 infection and elective surgery. Clinicians should use their clinical judgment to schedule procedures, said Randall M. Clark, MD, president of the American Society of Anesthesiologists (ASA). “We need all physicians, anesthesiologists, surgeons, and others to base their decision to go ahead with elective surgery on the patient’s symptoms, their need for the procedure, and whether delays could cause other problems with their health,” he said in an interview.
Prior to these updated recommendations, which were published Feb. 22, the ASA and the APSF recommended a 4-week gap between COVID-19 diagnosis and elective surgery for asymptomatic or mild cases, regardless of a patient’s vaccination status.
Extending the wait time from 4 to 7 weeks was based on a multination study conducted in October 2020 following more than 140,000 surgical patients. Patients with previous COVID-19 infection had an increased risk for complications and death in elective surgery for up to 6 weeks following their diagnosis, compared with patients without COVID-19. Additional research in the United States found that patients with a preoperative COVID diagnosis were at higher risk for postoperative complications of respiratory failure for up to 4 weeks after diagnosis and postoperative pneumonia complications for up to 8 weeks after diagnosis.
Because these studies were conducted in unvaccinated populations or those with low vaccination rates, and preliminary data suggest vaccinated patients with breakthrough infections may have a lower risk for complications and death postinfection, “we felt that it was prudent to just make recommendations specific to unvaccinated patients,” Dr. Clark added.
Although this guidance is “very helpful” in that it summarizes the currently available research to give evidence-based recommendations, the 7-week wait time is a “very conservative estimate,” Brent Matthews, MD, surgeon-in-chief of the surgery care division of Atrium Health, Charlotte, N.C., told this news organization. At Atrium Health, surgery is scheduled at least 21 days after a patient’s COVID-19 diagnosis, regardless of their vaccination status, Dr. Matthews said.
The studies currently available were conducted earlier in the pandemic, when a different variant was prevalent, Dr. Matthews explained. The Omicron variant is currently the most prevalent COVID-19 variant and is less virulent than earlier strains of the virus. The joint statement does note that there is currently “no robust data” on patients infected with the Delta or Omicron variants of COVID-19, and that “the Omicron variant causes less severe disease and is more likely to reside in the oro- and nasopharynx without infiltration and damage to the lungs.”
Still, the new recommendations are a reminder to re-evaluate the potential complications from surgery for previously infected patients and to consider what comorbidities might make them more vulnerable, Dr. Matthews said. “The real power of the joint statement is to get people to ensure that they make an assessment of every patient that comes in front of them who has had a recent positive COVID test.”
A version of this article first appeared on Medscape.com.
How Lp(a) can help improve ASCVD risk assessment
A look back at a pair of large cohort studies suggests a telling relation between two distinct predictors of atherosclerotic cardiovascular disease (ASCVD) risk and may offer guidance on how to interpret them together.
Elevated levels of lipoprotein(a), or Lp(a), and high coronary artery calcium (CAC) scores were both predictive of ASCVD risk over 10 years, but independent of each other and a host of more traditional cardiovascular risk factors, for example, in the analysis of data from the MESA (Multi-Ethnic Study of Atherosclerosis) and DHS (Dallas Heart Study) longitudinal cohorts.
Notably, the risk when both Lp(a) and CAC scores were high far exceeded that associated with either marker alone. But when CAC scores were less than 100 Agatston units, predicted ASCVD risk wasn’t influenced by levels of Lp(a). Indeed, a CAC score of 0 predicted the lowest levels of ASCVD risk, even with elevated Lp(a).
That is, the findings suggest, the addition of Lp(a) makes a difference to the risk assessment only when CAC scores are high, at least 100 units, and elevated Lp(a) doesn’t mean increased ASCVD risk in the absence of coronary calcium.
“Our novel findings indicate that elevated Lp(a) drives ASCVD risk independent of the subclinical coronary atherosclerosis burden captured by CAC score,” concluded a report on the analysis, published in the Journal of the American College of Cardiology, with lead author Anurag Mehta, MD, Emory University, Atlanta.
There are no formal recommendations on how to interpret Lp(a) and CAC scores together, but the current findings “provide impetus for measuring Lp(a) in more individuals as part of the shared decision-making process,” the authors contended.
“Really, the calcium score carries the majority of the information in terms of risk, except in the highest CAC score group. That is, if you have a high Lp(a) and a high burden of calcium, your risk is significantly higher than if you just have the high calcium score and the normal Lp(a),” senior author Parag H. Joshi, MD, MHS, said in an interview.
“We thought we would see that the group with higher Lp(a) would have more events over 10 years, even among those who didn’t have coronary calcium,” said Dr. Joshi, of the University of Texas Southwestern Medical Center, Dallas. “But we really don’t see that, at least in a statistically significant way.”
A CAC score of 0 would at least support a more conservative approach in a patient with elevated Lp(a) “who is hesitant to be on a statin or to be more aggressive managing their risk,” Dr. Joshi said.
“This study should be very reassuring for a patient like that,” Ron Blankstein, MD, director of cardiac computed tomography at Brigham and Women’s Hospital, Boston, said in an interview.
“If you have a high Lp(a) and you’re concerned, I think this study really supports the role of calcium scoring for further risk assessment,” said Dr. Blankstein, who is not associated with the new report. “We often check Lp(a) in individuals who perhaps have a family history or who come to see us in a preventive cardiology clinic. If it is high and there is concern, a calcium score can be very helpful. If it’s zero, that really means a very low risk of events. And if it’s elevated, I think we’re going to be more concerned about that patient.”
The current analysis suggests “that, when a patient without clinical cardiovascular disease is identified with either CAC ≥100 or Lp(a) >50 mg/dL, the next step in the risk evaluation should be to measure either Lp(a) or CAC, respectively – if not already performed – to identify the patients at highest risk,” Sotirios Tsimikas, MD, director of vascular medicine at University of California, San Diego, wrote in an accompanying editorial.
“Both Lp(a) and CAC should be more broadly applied in clinical care settings in patients without prior ASCVD to identify those that most likely will benefit from more aggressive therapy and, in the future, from Lp(a)-lowering therapies,” he wrote.
The analyses were conducted separately on data from 4,512 initially asymptomatic patients in MESA and 2,078 from the DHS cohort, who were followed for ASCVD events an average of 13 years and 11 years, respectively. Such events included coronary heart disease–related death, nonfatal MI, and fatal or nonfatal stroke.
In the MESA cohort – 52% women, 36.8% White, 29.3% Black, 22.2% Hispanic, and 11.7% Chinese – elevated Lp(a) (quintile 5 vs. quintiles 1-4) and CAC scores of 1-99 and above 100 (both compared with 0) were each independently associated with increased risk for ASCVD events. The hazard ratio was 1.29 (P = .02) for elevated Lp(a), 1.68 (P < .01) for a CAC score of 1-99, and 2.66 (P < .01) for a CAC score of at least 100.
The corresponding HRs in the DHS cohort were 1.54 (P = .07) for Lp(a), 3.32 (P < .01) for a CAC score of 1-99, and 5.21 (P < .01) for a CAC score of at least 100.
Of note, the authors wrote, ASCVD risk among MESA participants with a CAC score of 0 was not significantly different in those with normal and elevated Lp(a).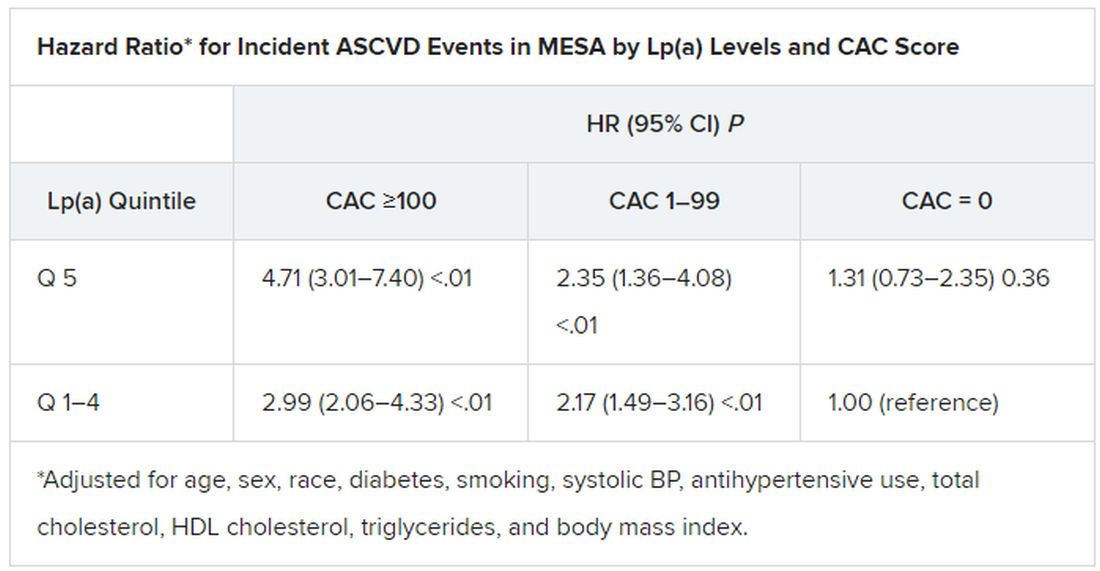
The findings were similar in the corresponding DHS analysis, the authors noted.
When both Lp(a) and CAC scores are considered as dichotomous variables, the highest 10-year ASCVD incidence in MESA was in participants with both elevated Lp(a) (≥50 mg/dL) and a high CAC score (≥100). The lowest risk was seen when Lp(a) was normal (<50 mg/dL) and the CAC score was no more than moderately high (<100).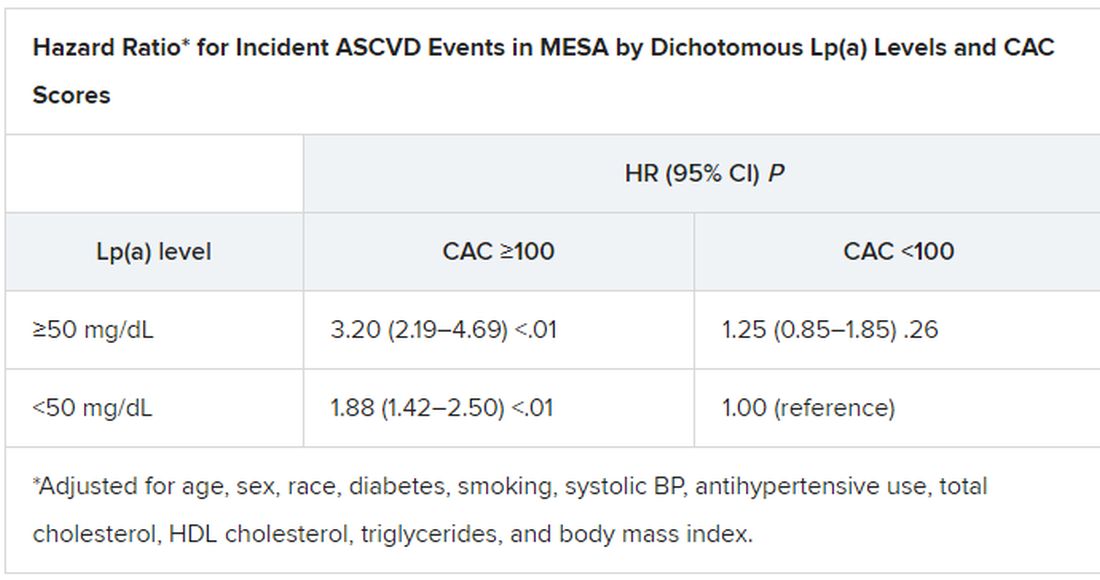
The results in the corresponding DHS analysis, according to the report, again mirrored those from MESA.
“This study has important implications for our patients and also potentially for future clinical trial design,” Dr. Blankstein noted. “A big part of developing a trial in this space is identifying the patients who are at higher risk,” and the current analysis supports CAC scores for identifying the highest-risk patient among those with elevated Lp(a).
Current wisdom is that, for the most part, Lp(a) levels are genetically mediated and are mostly unaffected by interventions such as diet management or exercise. It’s unknown whether reducing elevated Lp(a) levels pharmacologically will cut ASCVD risk, but there are a number of clinical trial programs currently aimed at learning just that. They include the Novartis-sponsored phase 3 HORIZON trial of the antisense agent pelacarsen (TQJ230), with an estimated enrollment of almost 7,700; a randomized, controlled dose-finding study of the small interfering RNA agent olpasiran (AMG890), with 290 patients and funded by Amgen; and an 88-patient phase 1 study of another siRNA agent, SLN360, supported by Silence Therapeutics.
Dr. Mehta reported no relevant relationships. Dr. Joshi has received grant support from Novo Nordisk and consulting income from Bayer and Regeneron; holds equity in G3 Therapeutics; and has served as site investigator for GlaxoSmithKline, Sanofi, AstraZeneca, and Novartis. Dr. Blankstein reported serving as a consultant to Amgen, Novartis, and Silence Therapeutics.
A version of this article first appeared on Medscape.com.
A look back at a pair of large cohort studies suggests a telling relation between two distinct predictors of atherosclerotic cardiovascular disease (ASCVD) risk and may offer guidance on how to interpret them together.
Elevated levels of lipoprotein(a), or Lp(a), and high coronary artery calcium (CAC) scores were both predictive of ASCVD risk over 10 years, but independent of each other and a host of more traditional cardiovascular risk factors, for example, in the analysis of data from the MESA (Multi-Ethnic Study of Atherosclerosis) and DHS (Dallas Heart Study) longitudinal cohorts.
Notably, the risk when both Lp(a) and CAC scores were high far exceeded that associated with either marker alone. But when CAC scores were less than 100 Agatston units, predicted ASCVD risk wasn’t influenced by levels of Lp(a). Indeed, a CAC score of 0 predicted the lowest levels of ASCVD risk, even with elevated Lp(a).
That is, the findings suggest, the addition of Lp(a) makes a difference to the risk assessment only when CAC scores are high, at least 100 units, and elevated Lp(a) doesn’t mean increased ASCVD risk in the absence of coronary calcium.
“Our novel findings indicate that elevated Lp(a) drives ASCVD risk independent of the subclinical coronary atherosclerosis burden captured by CAC score,” concluded a report on the analysis, published in the Journal of the American College of Cardiology, with lead author Anurag Mehta, MD, Emory University, Atlanta.
There are no formal recommendations on how to interpret Lp(a) and CAC scores together, but the current findings “provide impetus for measuring Lp(a) in more individuals as part of the shared decision-making process,” the authors contended.
“Really, the calcium score carries the majority of the information in terms of risk, except in the highest CAC score group. That is, if you have a high Lp(a) and a high burden of calcium, your risk is significantly higher than if you just have the high calcium score and the normal Lp(a),” senior author Parag H. Joshi, MD, MHS, said in an interview.
“We thought we would see that the group with higher Lp(a) would have more events over 10 years, even among those who didn’t have coronary calcium,” said Dr. Joshi, of the University of Texas Southwestern Medical Center, Dallas. “But we really don’t see that, at least in a statistically significant way.”
A CAC score of 0 would at least support a more conservative approach in a patient with elevated Lp(a) “who is hesitant to be on a statin or to be more aggressive managing their risk,” Dr. Joshi said.
“This study should be very reassuring for a patient like that,” Ron Blankstein, MD, director of cardiac computed tomography at Brigham and Women’s Hospital, Boston, said in an interview.
“If you have a high Lp(a) and you’re concerned, I think this study really supports the role of calcium scoring for further risk assessment,” said Dr. Blankstein, who is not associated with the new report. “We often check Lp(a) in individuals who perhaps have a family history or who come to see us in a preventive cardiology clinic. If it is high and there is concern, a calcium score can be very helpful. If it’s zero, that really means a very low risk of events. And if it’s elevated, I think we’re going to be more concerned about that patient.”
The current analysis suggests “that, when a patient without clinical cardiovascular disease is identified with either CAC ≥100 or Lp(a) >50 mg/dL, the next step in the risk evaluation should be to measure either Lp(a) or CAC, respectively – if not already performed – to identify the patients at highest risk,” Sotirios Tsimikas, MD, director of vascular medicine at University of California, San Diego, wrote in an accompanying editorial.
“Both Lp(a) and CAC should be more broadly applied in clinical care settings in patients without prior ASCVD to identify those that most likely will benefit from more aggressive therapy and, in the future, from Lp(a)-lowering therapies,” he wrote.
The analyses were conducted separately on data from 4,512 initially asymptomatic patients in MESA and 2,078 from the DHS cohort, who were followed for ASCVD events an average of 13 years and 11 years, respectively. Such events included coronary heart disease–related death, nonfatal MI, and fatal or nonfatal stroke.
In the MESA cohort – 52% women, 36.8% White, 29.3% Black, 22.2% Hispanic, and 11.7% Chinese – elevated Lp(a) (quintile 5 vs. quintiles 1-4) and CAC scores of 1-99 and above 100 (both compared with 0) were each independently associated with increased risk for ASCVD events. The hazard ratio was 1.29 (P = .02) for elevated Lp(a), 1.68 (P < .01) for a CAC score of 1-99, and 2.66 (P < .01) for a CAC score of at least 100.
The corresponding HRs in the DHS cohort were 1.54 (P = .07) for Lp(a), 3.32 (P < .01) for a CAC score of 1-99, and 5.21 (P < .01) for a CAC score of at least 100.
Of note, the authors wrote, ASCVD risk among MESA participants with a CAC score of 0 was not significantly different in those with normal and elevated Lp(a).
The findings were similar in the corresponding DHS analysis, the authors noted.
When both Lp(a) and CAC scores are considered as dichotomous variables, the highest 10-year ASCVD incidence in MESA was in participants with both elevated Lp(a) (≥50 mg/dL) and a high CAC score (≥100). The lowest risk was seen when Lp(a) was normal (<50 mg/dL) and the CAC score was no more than moderately high (<100).
The results in the corresponding DHS analysis, according to the report, again mirrored those from MESA.
“This study has important implications for our patients and also potentially for future clinical trial design,” Dr. Blankstein noted. “A big part of developing a trial in this space is identifying the patients who are at higher risk,” and the current analysis supports CAC scores for identifying the highest-risk patient among those with elevated Lp(a).
Current wisdom is that, for the most part, Lp(a) levels are genetically mediated and are mostly unaffected by interventions such as diet management or exercise. It’s unknown whether reducing elevated Lp(a) levels pharmacologically will cut ASCVD risk, but there are a number of clinical trial programs currently aimed at learning just that. They include the Novartis-sponsored phase 3 HORIZON trial of the antisense agent pelacarsen (TQJ230), with an estimated enrollment of almost 7,700; a randomized, controlled dose-finding study of the small interfering RNA agent olpasiran (AMG890), with 290 patients and funded by Amgen; and an 88-patient phase 1 study of another siRNA agent, SLN360, supported by Silence Therapeutics.
Dr. Mehta reported no relevant relationships. Dr. Joshi has received grant support from Novo Nordisk and consulting income from Bayer and Regeneron; holds equity in G3 Therapeutics; and has served as site investigator for GlaxoSmithKline, Sanofi, AstraZeneca, and Novartis. Dr. Blankstein reported serving as a consultant to Amgen, Novartis, and Silence Therapeutics.
A version of this article first appeared on Medscape.com.
A look back at a pair of large cohort studies suggests a telling relation between two distinct predictors of atherosclerotic cardiovascular disease (ASCVD) risk and may offer guidance on how to interpret them together.
Elevated levels of lipoprotein(a), or Lp(a), and high coronary artery calcium (CAC) scores were both predictive of ASCVD risk over 10 years, but independent of each other and a host of more traditional cardiovascular risk factors, for example, in the analysis of data from the MESA (Multi-Ethnic Study of Atherosclerosis) and DHS (Dallas Heart Study) longitudinal cohorts.
Notably, the risk when both Lp(a) and CAC scores were high far exceeded that associated with either marker alone. But when CAC scores were less than 100 Agatston units, predicted ASCVD risk wasn’t influenced by levels of Lp(a). Indeed, a CAC score of 0 predicted the lowest levels of ASCVD risk, even with elevated Lp(a).
That is, the findings suggest, the addition of Lp(a) makes a difference to the risk assessment only when CAC scores are high, at least 100 units, and elevated Lp(a) doesn’t mean increased ASCVD risk in the absence of coronary calcium.
“Our novel findings indicate that elevated Lp(a) drives ASCVD risk independent of the subclinical coronary atherosclerosis burden captured by CAC score,” concluded a report on the analysis, published in the Journal of the American College of Cardiology, with lead author Anurag Mehta, MD, Emory University, Atlanta.
There are no formal recommendations on how to interpret Lp(a) and CAC scores together, but the current findings “provide impetus for measuring Lp(a) in more individuals as part of the shared decision-making process,” the authors contended.
“Really, the calcium score carries the majority of the information in terms of risk, except in the highest CAC score group. That is, if you have a high Lp(a) and a high burden of calcium, your risk is significantly higher than if you just have the high calcium score and the normal Lp(a),” senior author Parag H. Joshi, MD, MHS, said in an interview.
“We thought we would see that the group with higher Lp(a) would have more events over 10 years, even among those who didn’t have coronary calcium,” said Dr. Joshi, of the University of Texas Southwestern Medical Center, Dallas. “But we really don’t see that, at least in a statistically significant way.”
A CAC score of 0 would at least support a more conservative approach in a patient with elevated Lp(a) “who is hesitant to be on a statin or to be more aggressive managing their risk,” Dr. Joshi said.
“This study should be very reassuring for a patient like that,” Ron Blankstein, MD, director of cardiac computed tomography at Brigham and Women’s Hospital, Boston, said in an interview.
“If you have a high Lp(a) and you’re concerned, I think this study really supports the role of calcium scoring for further risk assessment,” said Dr. Blankstein, who is not associated with the new report. “We often check Lp(a) in individuals who perhaps have a family history or who come to see us in a preventive cardiology clinic. If it is high and there is concern, a calcium score can be very helpful. If it’s zero, that really means a very low risk of events. And if it’s elevated, I think we’re going to be more concerned about that patient.”
The current analysis suggests “that, when a patient without clinical cardiovascular disease is identified with either CAC ≥100 or Lp(a) >50 mg/dL, the next step in the risk evaluation should be to measure either Lp(a) or CAC, respectively – if not already performed – to identify the patients at highest risk,” Sotirios Tsimikas, MD, director of vascular medicine at University of California, San Diego, wrote in an accompanying editorial.
“Both Lp(a) and CAC should be more broadly applied in clinical care settings in patients without prior ASCVD to identify those that most likely will benefit from more aggressive therapy and, in the future, from Lp(a)-lowering therapies,” he wrote.
The analyses were conducted separately on data from 4,512 initially asymptomatic patients in MESA and 2,078 from the DHS cohort, who were followed for ASCVD events an average of 13 years and 11 years, respectively. Such events included coronary heart disease–related death, nonfatal MI, and fatal or nonfatal stroke.
In the MESA cohort – 52% women, 36.8% White, 29.3% Black, 22.2% Hispanic, and 11.7% Chinese – elevated Lp(a) (quintile 5 vs. quintiles 1-4) and CAC scores of 1-99 and above 100 (both compared with 0) were each independently associated with increased risk for ASCVD events. The hazard ratio was 1.29 (P = .02) for elevated Lp(a), 1.68 (P < .01) for a CAC score of 1-99, and 2.66 (P < .01) for a CAC score of at least 100.
The corresponding HRs in the DHS cohort were 1.54 (P = .07) for Lp(a), 3.32 (P < .01) for a CAC score of 1-99, and 5.21 (P < .01) for a CAC score of at least 100.
Of note, the authors wrote, ASCVD risk among MESA participants with a CAC score of 0 was not significantly different in those with normal and elevated Lp(a).
The findings were similar in the corresponding DHS analysis, the authors noted.
When both Lp(a) and CAC scores are considered as dichotomous variables, the highest 10-year ASCVD incidence in MESA was in participants with both elevated Lp(a) (≥50 mg/dL) and a high CAC score (≥100). The lowest risk was seen when Lp(a) was normal (<50 mg/dL) and the CAC score was no more than moderately high (<100).
The results in the corresponding DHS analysis, according to the report, again mirrored those from MESA.
“This study has important implications for our patients and also potentially for future clinical trial design,” Dr. Blankstein noted. “A big part of developing a trial in this space is identifying the patients who are at higher risk,” and the current analysis supports CAC scores for identifying the highest-risk patient among those with elevated Lp(a).
Current wisdom is that, for the most part, Lp(a) levels are genetically mediated and are mostly unaffected by interventions such as diet management or exercise. It’s unknown whether reducing elevated Lp(a) levels pharmacologically will cut ASCVD risk, but there are a number of clinical trial programs currently aimed at learning just that. They include the Novartis-sponsored phase 3 HORIZON trial of the antisense agent pelacarsen (TQJ230), with an estimated enrollment of almost 7,700; a randomized, controlled dose-finding study of the small interfering RNA agent olpasiran (AMG890), with 290 patients and funded by Amgen; and an 88-patient phase 1 study of another siRNA agent, SLN360, supported by Silence Therapeutics.
Dr. Mehta reported no relevant relationships. Dr. Joshi has received grant support from Novo Nordisk and consulting income from Bayer and Regeneron; holds equity in G3 Therapeutics; and has served as site investigator for GlaxoSmithKline, Sanofi, AstraZeneca, and Novartis. Dr. Blankstein reported serving as a consultant to Amgen, Novartis, and Silence Therapeutics.
A version of this article first appeared on Medscape.com.
FROM THE JOURNAL OF THE AMERICAN COLLEGE OF CARDIOLOGY
New studies indicate COVID pandemic began in Wuhan market
Two preprint studies released on Feb. 26 offer additional evidence that the coronavirus pandemic started at a market in Wuhan, China.
By analyzing data from several sources, scientists concluded that the virus came from animals and spread to humans in late 2019 at the Huanan Seafood Market. They added that no evidence supported a theory that the virus came from a laboratory in Wuhan.
“When you look at all the evidence together, it’s an extraordinarily clear picture that the pandemic started at the Huanan market,” Michael Worobey, D.Phil., a co-author on both studies and an evolutionary biologist at the University of Arizona, told the New York Times.
The two reports haven’t yet been peer-reviewed or published in a scientific journal. They were posted on Zenodo, an open-access research repository operated by CERN.
In one study, researchers used spatial analysis to show that the earliest COVID-19 cases, which were diagnosed in December 2019, were linked to the market. Researchers also found that environmental samples that tested positive for the SARS-CoV-2 virus were associated with animal vendors.
In another study, researchers found that two major viral lineages of the coronavirus resulted from at least two events when the virus spread from animals into humans. The first transmission most likely happened in late November or early December 2019, they wrote, and the other likely happened a few weeks later.
Several of the researchers behind the new studies also published a review last summer that said the pandemic originated in an animal, likely at a wildlife market. At that time, they said the first known case was a vendor at the Huanan market.
The new findings provide the strongest evidence yet that the pandemic had animal-related origins, Dr. Worobey told CNN. He called the results a “game, set and match” for the theory that the pandemic began in a lab.
“It’s no longer something that makes sense to imagine that this started any other way,” he said.
In a separate line of research, scientists at the Chinese CDC conducted a new analysis of samples collected at the market in January. They found that the samples included the two main lineages of the coronavirus. They posted the results in a report on the Research Square preprint server Feb. 26.
“The beauty of it is how simply it all adds up now,” Jeremy Kamil, a virologist at Louisiana State University Health Sciences, who wasn’t involved with the new studies, told the New York Times.
The initial spread of the coronavirus was like a firework, Dr. Worobey told CNN, starting at the market and exploding outward. The “overwhelming majority” of cases were specifically linked to the western section of the market, where most of the live-mammal vendors were located, the study authors wrote. Then COVID-19 cases spread into the community from there, and the pattern of transmission changed by January or February 2020.
When researchers tested surfaces at the market for coronavirus genetic material, one stall had the most positives, including a cage where raccoon dogs had been kept.
The study authors said the findings highlight the urgent need to pay attention to situations where wild animals and humans interact closely on a daily basis.
“We need to do a better job of farming and regulating these wild animals,” Robert Garry, one of the co-authors and a professor of microbiology and immunology at the Tulane University School of Medicine, told CNN.
That could include better infrastructure in places like markets where viruses spill over from animals to humans, he said. Surveillance is also key in preventing future pandemics by detecting new respiratory diseases in humans, isolating patients, and sequencing new virus strains.
“This is not the last time this happens,” he said.
A version of this article first appeared on WebMD.com.
Two preprint studies released on Feb. 26 offer additional evidence that the coronavirus pandemic started at a market in Wuhan, China.
By analyzing data from several sources, scientists concluded that the virus came from animals and spread to humans in late 2019 at the Huanan Seafood Market. They added that no evidence supported a theory that the virus came from a laboratory in Wuhan.
“When you look at all the evidence together, it’s an extraordinarily clear picture that the pandemic started at the Huanan market,” Michael Worobey, D.Phil., a co-author on both studies and an evolutionary biologist at the University of Arizona, told the New York Times.
The two reports haven’t yet been peer-reviewed or published in a scientific journal. They were posted on Zenodo, an open-access research repository operated by CERN.
In one study, researchers used spatial analysis to show that the earliest COVID-19 cases, which were diagnosed in December 2019, were linked to the market. Researchers also found that environmental samples that tested positive for the SARS-CoV-2 virus were associated with animal vendors.
In another study, researchers found that two major viral lineages of the coronavirus resulted from at least two events when the virus spread from animals into humans. The first transmission most likely happened in late November or early December 2019, they wrote, and the other likely happened a few weeks later.
Several of the researchers behind the new studies also published a review last summer that said the pandemic originated in an animal, likely at a wildlife market. At that time, they said the first known case was a vendor at the Huanan market.
The new findings provide the strongest evidence yet that the pandemic had animal-related origins, Dr. Worobey told CNN. He called the results a “game, set and match” for the theory that the pandemic began in a lab.
“It’s no longer something that makes sense to imagine that this started any other way,” he said.
In a separate line of research, scientists at the Chinese CDC conducted a new analysis of samples collected at the market in January. They found that the samples included the two main lineages of the coronavirus. They posted the results in a report on the Research Square preprint server Feb. 26.
“The beauty of it is how simply it all adds up now,” Jeremy Kamil, a virologist at Louisiana State University Health Sciences, who wasn’t involved with the new studies, told the New York Times.
The initial spread of the coronavirus was like a firework, Dr. Worobey told CNN, starting at the market and exploding outward. The “overwhelming majority” of cases were specifically linked to the western section of the market, where most of the live-mammal vendors were located, the study authors wrote. Then COVID-19 cases spread into the community from there, and the pattern of transmission changed by January or February 2020.
When researchers tested surfaces at the market for coronavirus genetic material, one stall had the most positives, including a cage where raccoon dogs had been kept.
The study authors said the findings highlight the urgent need to pay attention to situations where wild animals and humans interact closely on a daily basis.
“We need to do a better job of farming and regulating these wild animals,” Robert Garry, one of the co-authors and a professor of microbiology and immunology at the Tulane University School of Medicine, told CNN.
That could include better infrastructure in places like markets where viruses spill over from animals to humans, he said. Surveillance is also key in preventing future pandemics by detecting new respiratory diseases in humans, isolating patients, and sequencing new virus strains.
“This is not the last time this happens,” he said.
A version of this article first appeared on WebMD.com.
Two preprint studies released on Feb. 26 offer additional evidence that the coronavirus pandemic started at a market in Wuhan, China.
By analyzing data from several sources, scientists concluded that the virus came from animals and spread to humans in late 2019 at the Huanan Seafood Market. They added that no evidence supported a theory that the virus came from a laboratory in Wuhan.
“When you look at all the evidence together, it’s an extraordinarily clear picture that the pandemic started at the Huanan market,” Michael Worobey, D.Phil., a co-author on both studies and an evolutionary biologist at the University of Arizona, told the New York Times.
The two reports haven’t yet been peer-reviewed or published in a scientific journal. They were posted on Zenodo, an open-access research repository operated by CERN.
In one study, researchers used spatial analysis to show that the earliest COVID-19 cases, which were diagnosed in December 2019, were linked to the market. Researchers also found that environmental samples that tested positive for the SARS-CoV-2 virus were associated with animal vendors.
In another study, researchers found that two major viral lineages of the coronavirus resulted from at least two events when the virus spread from animals into humans. The first transmission most likely happened in late November or early December 2019, they wrote, and the other likely happened a few weeks later.
Several of the researchers behind the new studies also published a review last summer that said the pandemic originated in an animal, likely at a wildlife market. At that time, they said the first known case was a vendor at the Huanan market.
The new findings provide the strongest evidence yet that the pandemic had animal-related origins, Dr. Worobey told CNN. He called the results a “game, set and match” for the theory that the pandemic began in a lab.
“It’s no longer something that makes sense to imagine that this started any other way,” he said.
In a separate line of research, scientists at the Chinese CDC conducted a new analysis of samples collected at the market in January. They found that the samples included the two main lineages of the coronavirus. They posted the results in a report on the Research Square preprint server Feb. 26.
“The beauty of it is how simply it all adds up now,” Jeremy Kamil, a virologist at Louisiana State University Health Sciences, who wasn’t involved with the new studies, told the New York Times.
The initial spread of the coronavirus was like a firework, Dr. Worobey told CNN, starting at the market and exploding outward. The “overwhelming majority” of cases were specifically linked to the western section of the market, where most of the live-mammal vendors were located, the study authors wrote. Then COVID-19 cases spread into the community from there, and the pattern of transmission changed by January or February 2020.
When researchers tested surfaces at the market for coronavirus genetic material, one stall had the most positives, including a cage where raccoon dogs had been kept.
The study authors said the findings highlight the urgent need to pay attention to situations where wild animals and humans interact closely on a daily basis.
“We need to do a better job of farming and regulating these wild animals,” Robert Garry, one of the co-authors and a professor of microbiology and immunology at the Tulane University School of Medicine, told CNN.
That could include better infrastructure in places like markets where viruses spill over from animals to humans, he said. Surveillance is also key in preventing future pandemics by detecting new respiratory diseases in humans, isolating patients, and sequencing new virus strains.
“This is not the last time this happens,” he said.
A version of this article first appeared on WebMD.com.
Autoantibodies may underpin clotting effects of COVID-19
Circulating antiphospholipid autoantibodies may contribute to endothelial cell activation and dysfunction in severe COVID-19, researchers report.
In 2020, the same researchers reported results from a preclinical study demonstrating that autoantibodies from patients with active COVID-19 caused clotting in mice.
The new study, published in Arthritis and Rheumatology, found higher-than-expected levels of antiphospholipid autoantibodies in the blood samples of 244 patients hospitalized with COVID-19.
“While endothelial dysfunction has been implicated in the widespread thromboinflammatory complications of COVID-19, the upstream mediators of endotheliopathy remain for the most part cryptic,” write Hui Shi, MD, PhD, and coauthors from the University of Michigan, Ann Arbor, and the National Heart, Lung, and Blood Institute.
When asked for comment on the study, Eline T. Luning Prak, MD, PhD, professor of pathology and laboratory medicine at the Hospital of the University of Pennsylvania in Philadelphia, said, “The autopsy cases for COVID-19 strongly point to thromboembolic complications in many individuals who succumbed to sequelae of the infection.
“Importantly, however, many factors can contribute to this pathology, including the inflammatory milieu, monocyte activation, neutrophil extracellular traps, immune complexes, complement, as well as effects on endothelial cells,” explained Dr. Luning Prak, who was not involved in the study.
“The findings in this paper nicely complement another study by Schmaier et al. that came out recently in JCI Insight that also suggests that endothelial cells can be activated by antibodies, she said.
‘Even stronger connection between autoantibody formation and clotting in COVID-19’
Dr. Shi and her team cultured human endothelial cells in serum or plasma from 244 patients hospitalized with COVID-19 and plasma from 100 patients with non-COVID sepsis. Using in-cell enzyme-linked immunosorbent assay, they measured levels of key cell adhesion molecules.
After analysis, the researchers found that serum from COVID-19 patients activated cultured endothelial cells to express surface adhesion molecules essential to inflammation and thrombosis, particularly E-selectin, ICAM-1, and VCAM-1.
“The presence of circulating antiphospholipid antibodies was a strong marker of the ability of COVID-19 serum to activate endothelium,” they explain.
Further analyses revealed that, for a subset of serum samples from patients with severe infection, this activation could be mitigated by depleting total immunoglobulin G.
In addition, supplementation of control serum with patient IgG was adequate to trigger endothelial activation.
On the basis of these results, the researchers hypothesize that antiphospholipid autoantibodies may characterize antibody profiles in severe COVID-19 that activate the endothelium and transition the usually quiescent blood-vessel wall interface toward inflammation and coagulation.
“[These findings] provide an even stronger connection between autoantibody formation and clotting in COVID-19,” Dr. Shi said in an accompanying press release.
Clinical implications
From a clinical perspective, Dr. Shi and her team question whether patients with severe COVID-19 should be tested for antiphospholipid antibodies to assess their risk of thrombosis and progression to respiratory failure.
Moreover, they question whether patients with high antiphospholipid antibody titers might benefit from therapies used in conventional cases of severe antiphospholipid syndrome, such as plasmapheresis, anticoagulation therapy, and complement inhibition, Dr. Shi added.
The researchers hope to answer these and other remaining questions in future studies. “Eventually, we may be able to repurpose treatments used in traditional cases of antiphospholipid syndrome for COVID-19.
“As we await definitive solutions to the pandemic, these findings add important context to the complex interplay between SARS-CoV-2 infection, the human immune system, and vascular immunobiology,” she concluded.
The study was supported by grants from the Rheumatology Research Foundation, the Michigan Medicine Frankel Cardiovascular Center, and the A. Alfred Taubman Medical Research Institute. One author is an inventor on an unrelated pending patent to the University of Michigan. The other authors and Dr. Luning Prak have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Circulating antiphospholipid autoantibodies may contribute to endothelial cell activation and dysfunction in severe COVID-19, researchers report.
In 2020, the same researchers reported results from a preclinical study demonstrating that autoantibodies from patients with active COVID-19 caused clotting in mice.
The new study, published in Arthritis and Rheumatology, found higher-than-expected levels of antiphospholipid autoantibodies in the blood samples of 244 patients hospitalized with COVID-19.
“While endothelial dysfunction has been implicated in the widespread thromboinflammatory complications of COVID-19, the upstream mediators of endotheliopathy remain for the most part cryptic,” write Hui Shi, MD, PhD, and coauthors from the University of Michigan, Ann Arbor, and the National Heart, Lung, and Blood Institute.
When asked for comment on the study, Eline T. Luning Prak, MD, PhD, professor of pathology and laboratory medicine at the Hospital of the University of Pennsylvania in Philadelphia, said, “The autopsy cases for COVID-19 strongly point to thromboembolic complications in many individuals who succumbed to sequelae of the infection.
“Importantly, however, many factors can contribute to this pathology, including the inflammatory milieu, monocyte activation, neutrophil extracellular traps, immune complexes, complement, as well as effects on endothelial cells,” explained Dr. Luning Prak, who was not involved in the study.
“The findings in this paper nicely complement another study by Schmaier et al. that came out recently in JCI Insight that also suggests that endothelial cells can be activated by antibodies, she said.
‘Even stronger connection between autoantibody formation and clotting in COVID-19’
Dr. Shi and her team cultured human endothelial cells in serum or plasma from 244 patients hospitalized with COVID-19 and plasma from 100 patients with non-COVID sepsis. Using in-cell enzyme-linked immunosorbent assay, they measured levels of key cell adhesion molecules.
After analysis, the researchers found that serum from COVID-19 patients activated cultured endothelial cells to express surface adhesion molecules essential to inflammation and thrombosis, particularly E-selectin, ICAM-1, and VCAM-1.
“The presence of circulating antiphospholipid antibodies was a strong marker of the ability of COVID-19 serum to activate endothelium,” they explain.
Further analyses revealed that, for a subset of serum samples from patients with severe infection, this activation could be mitigated by depleting total immunoglobulin G.
In addition, supplementation of control serum with patient IgG was adequate to trigger endothelial activation.
On the basis of these results, the researchers hypothesize that antiphospholipid autoantibodies may characterize antibody profiles in severe COVID-19 that activate the endothelium and transition the usually quiescent blood-vessel wall interface toward inflammation and coagulation.
“[These findings] provide an even stronger connection between autoantibody formation and clotting in COVID-19,” Dr. Shi said in an accompanying press release.
Clinical implications
From a clinical perspective, Dr. Shi and her team question whether patients with severe COVID-19 should be tested for antiphospholipid antibodies to assess their risk of thrombosis and progression to respiratory failure.
Moreover, they question whether patients with high antiphospholipid antibody titers might benefit from therapies used in conventional cases of severe antiphospholipid syndrome, such as plasmapheresis, anticoagulation therapy, and complement inhibition, Dr. Shi added.
The researchers hope to answer these and other remaining questions in future studies. “Eventually, we may be able to repurpose treatments used in traditional cases of antiphospholipid syndrome for COVID-19.
“As we await definitive solutions to the pandemic, these findings add important context to the complex interplay between SARS-CoV-2 infection, the human immune system, and vascular immunobiology,” she concluded.
The study was supported by grants from the Rheumatology Research Foundation, the Michigan Medicine Frankel Cardiovascular Center, and the A. Alfred Taubman Medical Research Institute. One author is an inventor on an unrelated pending patent to the University of Michigan. The other authors and Dr. Luning Prak have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Circulating antiphospholipid autoantibodies may contribute to endothelial cell activation and dysfunction in severe COVID-19, researchers report.
In 2020, the same researchers reported results from a preclinical study demonstrating that autoantibodies from patients with active COVID-19 caused clotting in mice.
The new study, published in Arthritis and Rheumatology, found higher-than-expected levels of antiphospholipid autoantibodies in the blood samples of 244 patients hospitalized with COVID-19.
“While endothelial dysfunction has been implicated in the widespread thromboinflammatory complications of COVID-19, the upstream mediators of endotheliopathy remain for the most part cryptic,” write Hui Shi, MD, PhD, and coauthors from the University of Michigan, Ann Arbor, and the National Heart, Lung, and Blood Institute.
When asked for comment on the study, Eline T. Luning Prak, MD, PhD, professor of pathology and laboratory medicine at the Hospital of the University of Pennsylvania in Philadelphia, said, “The autopsy cases for COVID-19 strongly point to thromboembolic complications in many individuals who succumbed to sequelae of the infection.
“Importantly, however, many factors can contribute to this pathology, including the inflammatory milieu, monocyte activation, neutrophil extracellular traps, immune complexes, complement, as well as effects on endothelial cells,” explained Dr. Luning Prak, who was not involved in the study.
“The findings in this paper nicely complement another study by Schmaier et al. that came out recently in JCI Insight that also suggests that endothelial cells can be activated by antibodies, she said.
‘Even stronger connection between autoantibody formation and clotting in COVID-19’
Dr. Shi and her team cultured human endothelial cells in serum or plasma from 244 patients hospitalized with COVID-19 and plasma from 100 patients with non-COVID sepsis. Using in-cell enzyme-linked immunosorbent assay, they measured levels of key cell adhesion molecules.
After analysis, the researchers found that serum from COVID-19 patients activated cultured endothelial cells to express surface adhesion molecules essential to inflammation and thrombosis, particularly E-selectin, ICAM-1, and VCAM-1.
“The presence of circulating antiphospholipid antibodies was a strong marker of the ability of COVID-19 serum to activate endothelium,” they explain.
Further analyses revealed that, for a subset of serum samples from patients with severe infection, this activation could be mitigated by depleting total immunoglobulin G.
In addition, supplementation of control serum with patient IgG was adequate to trigger endothelial activation.
On the basis of these results, the researchers hypothesize that antiphospholipid autoantibodies may characterize antibody profiles in severe COVID-19 that activate the endothelium and transition the usually quiescent blood-vessel wall interface toward inflammation and coagulation.
“[These findings] provide an even stronger connection between autoantibody formation and clotting in COVID-19,” Dr. Shi said in an accompanying press release.
Clinical implications
From a clinical perspective, Dr. Shi and her team question whether patients with severe COVID-19 should be tested for antiphospholipid antibodies to assess their risk of thrombosis and progression to respiratory failure.
Moreover, they question whether patients with high antiphospholipid antibody titers might benefit from therapies used in conventional cases of severe antiphospholipid syndrome, such as plasmapheresis, anticoagulation therapy, and complement inhibition, Dr. Shi added.
The researchers hope to answer these and other remaining questions in future studies. “Eventually, we may be able to repurpose treatments used in traditional cases of antiphospholipid syndrome for COVID-19.
“As we await definitive solutions to the pandemic, these findings add important context to the complex interplay between SARS-CoV-2 infection, the human immune system, and vascular immunobiology,” she concluded.
The study was supported by grants from the Rheumatology Research Foundation, the Michigan Medicine Frankel Cardiovascular Center, and the A. Alfred Taubman Medical Research Institute. One author is an inventor on an unrelated pending patent to the University of Michigan. The other authors and Dr. Luning Prak have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
COVID-19 vaccines do not trigger sudden hearing loss: Study
Anecdotal reports have linked the vaccines against COVID-19 to the sudden loss of hearing in some people. But a new study has found no evidence for such a connection with any of the three approved shots.
The analysis of data from the Centers for Disease Control and Prevention’s Vaccine Adverse Event Reporting System (VAERS) found that
“We’re not finding a signal,” said Eric J. Formeister, MD, a neurotology fellow at the Johns Hopkins University, Baltimore, and the first author of the U.S. study, which appeared Feb. 24 in JAMA Otolaryngology – Head and Neck Surgery.
Dr. Formeister and colleagues undertook the study in response to reports of hearing problems, including hearing loss and tinnitus, that occurred soon after COVID-19 vaccination.
They analyzed reports of sudden hearing loss, experienced within 21 days of vaccination, logged in VAERS. Anyone can report a potential event to the database, which does not require medical documentation in support of the adverse event. To minimize potential misdiagnoses, Dr. Formeister and colleagues reviewed only those reports that indicated that a doctor had diagnosed sudden hearing loss, leaving 555 cases (305 in women; mean age 54 years) between December 2020 and July 2021.
Dividing these reports by the total doses of vaccines administered in the United States during that period yielded an incidence rate of 0.6 cases of sudden hearing loss for every 100,000 people, Dr. Formeister and colleagues reported.
When the researchers divided all cases of hearing loss in the VAERS database (2,170) by the number of people who had received two doses of vaccine, the incidence rate increased to 28 per 100,000 people. For comparison, the authors reported, the incidence of sudden hearing loss within the United States population is between 11 and 77 per 100,000 people, depending on age.
“There was not an increase in cases of sudden [sensorineural] hearing loss associated with COVID-19 vaccination compared to previously published reports before the COVID-19 vaccination era,” study coauthor Elliott D. Kozin, MD, assistant professor of otolaryngology–head and neck surgery at Harvard Medical School, Boston, said in an interview.
Another reassuring sign: If hearing loss were linked to the vaccines, the researchers said, they would expect to see an increase in the number of complaints in lockstep with an increase in the number of doses administered. However, the opposite was true. “[T]he rate of reports per 100,000 doses decreased across the vaccination period, despite large concomitant increases in the absolute number of vaccine doses administered per week,” the researchers reported.
They also looked at case reports of 21 men and women who had experienced sudden hearing loss after having received COVID-19 vaccines, to see if they could discern any clinically relevant signs of people most likely to experience the adverse event. However, the group had a range of preexisting conditions and varying times after receiving a vaccine when their hearing loss occurred, leading Dr. Formeister’s team to conclude that they could find no clear markers of risk.
“When we examined patients across several institutions, there was no obvious pattern. The patient demographics and clinical findings were variable,” Dr. Kozin said. A provisional interpretation of this data, he added, is that no link exists between COVID-19 vaccination and predictable hearing deficits, although the analysis covered a small number of patients.
“Association does not necessarily imply a causal relationship,” said Michael Brenner, MD, FACS, associate professor of otolaryngology–head and neck surgery at the University of Michigan, Ann Arbor. Dr. Brenner, who was not involved in the study, said any hearing loss attributed to the COVID-19 vaccines could have had other causes besides the injections.
But a second study, also published in JAMA Otolaryngology – Head and Neck Surgery on Feb. 24, leaves open the possibility of a link. Researchers in Israel looked for increases in steroid prescriptions used to treat sudden hearing loss as vaccination with the Pfizer version of the shot became widespread in that country. Their conclusion: The vaccine might be associated with a slightly increased risk of sudden hearing loss, although if so, that risk is likely “very small” and the benefits of vaccination “outweigh its potential association” with the side effect.
Dr. Brenner agreed. “The evidence supports [the] clear public health benefit of COVID-19 vaccination, and the scale of those benefits dwarfs associations with hearing, which are of uncertain significance,” he said.
A version of this article first appeared on Medscape.com.
Anecdotal reports have linked the vaccines against COVID-19 to the sudden loss of hearing in some people. But a new study has found no evidence for such a connection with any of the three approved shots.
The analysis of data from the Centers for Disease Control and Prevention’s Vaccine Adverse Event Reporting System (VAERS) found that
“We’re not finding a signal,” said Eric J. Formeister, MD, a neurotology fellow at the Johns Hopkins University, Baltimore, and the first author of the U.S. study, which appeared Feb. 24 in JAMA Otolaryngology – Head and Neck Surgery.
Dr. Formeister and colleagues undertook the study in response to reports of hearing problems, including hearing loss and tinnitus, that occurred soon after COVID-19 vaccination.
They analyzed reports of sudden hearing loss, experienced within 21 days of vaccination, logged in VAERS. Anyone can report a potential event to the database, which does not require medical documentation in support of the adverse event. To minimize potential misdiagnoses, Dr. Formeister and colleagues reviewed only those reports that indicated that a doctor had diagnosed sudden hearing loss, leaving 555 cases (305 in women; mean age 54 years) between December 2020 and July 2021.
Dividing these reports by the total doses of vaccines administered in the United States during that period yielded an incidence rate of 0.6 cases of sudden hearing loss for every 100,000 people, Dr. Formeister and colleagues reported.
When the researchers divided all cases of hearing loss in the VAERS database (2,170) by the number of people who had received two doses of vaccine, the incidence rate increased to 28 per 100,000 people. For comparison, the authors reported, the incidence of sudden hearing loss within the United States population is between 11 and 77 per 100,000 people, depending on age.
“There was not an increase in cases of sudden [sensorineural] hearing loss associated with COVID-19 vaccination compared to previously published reports before the COVID-19 vaccination era,” study coauthor Elliott D. Kozin, MD, assistant professor of otolaryngology–head and neck surgery at Harvard Medical School, Boston, said in an interview.
Another reassuring sign: If hearing loss were linked to the vaccines, the researchers said, they would expect to see an increase in the number of complaints in lockstep with an increase in the number of doses administered. However, the opposite was true. “[T]he rate of reports per 100,000 doses decreased across the vaccination period, despite large concomitant increases in the absolute number of vaccine doses administered per week,” the researchers reported.
They also looked at case reports of 21 men and women who had experienced sudden hearing loss after having received COVID-19 vaccines, to see if they could discern any clinically relevant signs of people most likely to experience the adverse event. However, the group had a range of preexisting conditions and varying times after receiving a vaccine when their hearing loss occurred, leading Dr. Formeister’s team to conclude that they could find no clear markers of risk.
“When we examined patients across several institutions, there was no obvious pattern. The patient demographics and clinical findings were variable,” Dr. Kozin said. A provisional interpretation of this data, he added, is that no link exists between COVID-19 vaccination and predictable hearing deficits, although the analysis covered a small number of patients.
“Association does not necessarily imply a causal relationship,” said Michael Brenner, MD, FACS, associate professor of otolaryngology–head and neck surgery at the University of Michigan, Ann Arbor. Dr. Brenner, who was not involved in the study, said any hearing loss attributed to the COVID-19 vaccines could have had other causes besides the injections.
But a second study, also published in JAMA Otolaryngology – Head and Neck Surgery on Feb. 24, leaves open the possibility of a link. Researchers in Israel looked for increases in steroid prescriptions used to treat sudden hearing loss as vaccination with the Pfizer version of the shot became widespread in that country. Their conclusion: The vaccine might be associated with a slightly increased risk of sudden hearing loss, although if so, that risk is likely “very small” and the benefits of vaccination “outweigh its potential association” with the side effect.
Dr. Brenner agreed. “The evidence supports [the] clear public health benefit of COVID-19 vaccination, and the scale of those benefits dwarfs associations with hearing, which are of uncertain significance,” he said.
A version of this article first appeared on Medscape.com.
Anecdotal reports have linked the vaccines against COVID-19 to the sudden loss of hearing in some people. But a new study has found no evidence for such a connection with any of the three approved shots.
The analysis of data from the Centers for Disease Control and Prevention’s Vaccine Adverse Event Reporting System (VAERS) found that
“We’re not finding a signal,” said Eric J. Formeister, MD, a neurotology fellow at the Johns Hopkins University, Baltimore, and the first author of the U.S. study, which appeared Feb. 24 in JAMA Otolaryngology – Head and Neck Surgery.
Dr. Formeister and colleagues undertook the study in response to reports of hearing problems, including hearing loss and tinnitus, that occurred soon after COVID-19 vaccination.
They analyzed reports of sudden hearing loss, experienced within 21 days of vaccination, logged in VAERS. Anyone can report a potential event to the database, which does not require medical documentation in support of the adverse event. To minimize potential misdiagnoses, Dr. Formeister and colleagues reviewed only those reports that indicated that a doctor had diagnosed sudden hearing loss, leaving 555 cases (305 in women; mean age 54 years) between December 2020 and July 2021.
Dividing these reports by the total doses of vaccines administered in the United States during that period yielded an incidence rate of 0.6 cases of sudden hearing loss for every 100,000 people, Dr. Formeister and colleagues reported.
When the researchers divided all cases of hearing loss in the VAERS database (2,170) by the number of people who had received two doses of vaccine, the incidence rate increased to 28 per 100,000 people. For comparison, the authors reported, the incidence of sudden hearing loss within the United States population is between 11 and 77 per 100,000 people, depending on age.
“There was not an increase in cases of sudden [sensorineural] hearing loss associated with COVID-19 vaccination compared to previously published reports before the COVID-19 vaccination era,” study coauthor Elliott D. Kozin, MD, assistant professor of otolaryngology–head and neck surgery at Harvard Medical School, Boston, said in an interview.
Another reassuring sign: If hearing loss were linked to the vaccines, the researchers said, they would expect to see an increase in the number of complaints in lockstep with an increase in the number of doses administered. However, the opposite was true. “[T]he rate of reports per 100,000 doses decreased across the vaccination period, despite large concomitant increases in the absolute number of vaccine doses administered per week,” the researchers reported.
They also looked at case reports of 21 men and women who had experienced sudden hearing loss after having received COVID-19 vaccines, to see if they could discern any clinically relevant signs of people most likely to experience the adverse event. However, the group had a range of preexisting conditions and varying times after receiving a vaccine when their hearing loss occurred, leading Dr. Formeister’s team to conclude that they could find no clear markers of risk.
“When we examined patients across several institutions, there was no obvious pattern. The patient demographics and clinical findings were variable,” Dr. Kozin said. A provisional interpretation of this data, he added, is that no link exists between COVID-19 vaccination and predictable hearing deficits, although the analysis covered a small number of patients.
“Association does not necessarily imply a causal relationship,” said Michael Brenner, MD, FACS, associate professor of otolaryngology–head and neck surgery at the University of Michigan, Ann Arbor. Dr. Brenner, who was not involved in the study, said any hearing loss attributed to the COVID-19 vaccines could have had other causes besides the injections.
But a second study, also published in JAMA Otolaryngology – Head and Neck Surgery on Feb. 24, leaves open the possibility of a link. Researchers in Israel looked for increases in steroid prescriptions used to treat sudden hearing loss as vaccination with the Pfizer version of the shot became widespread in that country. Their conclusion: The vaccine might be associated with a slightly increased risk of sudden hearing loss, although if so, that risk is likely “very small” and the benefits of vaccination “outweigh its potential association” with the side effect.
Dr. Brenner agreed. “The evidence supports [the] clear public health benefit of COVID-19 vaccination, and the scale of those benefits dwarfs associations with hearing, which are of uncertain significance,” he said.
A version of this article first appeared on Medscape.com.
FROM JAMA OTOLARYNGOLOGY – HEAD AND NECK SURGERY
The importance of a post-COVID wellness program for medical staff
LAS VEGAS – , according to Jon A. Levenson, MD.
“We can learn from previous pandemics and epidemics, which will be important for us going forward from COVID-19,” Dr. Levenson, associate professor of psychiatry at Columbia University Irving Medical Center, New York, said during an annual psychopharmacology update held by the Nevada Psychiatric Association.
During the severe acute respiratory syndrome (SARS) epidemic in 2005, 68% of health care workers reported significant job-related stress, including increased workload, changing work duties, redeployment, shortage of medical supplies, concerns about insufficient personal protective equipment (PPE), lack of safety at work, absence of effective treatment protocols, inconsistent organizational support and information and misinformation from hospital management, and witnessing intense pain, isolation, and loss on a daily basis with few opportunities to take breaks (Psychiatr Serv. 2020 Oct 6. doi: 10.1176/appi.ps.202000274).
Personal concerns associated with psychopathological symptoms included spreading infection to family members; feeling responsibility for family members’ social isolation; self-isolating to avoid infecting family, which can lead to increased loneliness and sadness. “For those who were working remotely, this level of work is hard and challenging,” Dr. Levenson said. “For those who are parents, the 24-hour childcare responsibilities exist on top of work. They often found they can’t unwind with friends.”
Across SARS, MERS, Ebola, and swine flu, a wide range of prevalence in symptoms of distress, stress, anxiety, depressive symptoms, and substance use emerged, he continued. During COVID-19, at least three studies reported significant percentages of distress, depression, anxiety, insomnia, and PTSD among health care workers (JAMA Netw Open. 2020;3[3]:e203976, Front Psychol. 2020 Dec 8;11:608986., and Gen Hosp Psychiatry. Sep-Oct 2020;66:1-8).
“Who is at most-increased risk?” Dr. Levenson asked. “Women; those who are younger and have fewer years of work experience; those working on the front lines such as nurses and advanced practice professionals; and people with preexisting vulnerabilities to psychiatric disorders including anxiety, depression, obsessional symptoms, substance use, suicidal behavior, and impulse control disorders are likely to be especially vulnerable to stress-related symptoms.”
At CUIMC, there were certain “tipping points,” to the vulnerability of health care worker well-being in the early stage of the COVID-19 pandemic, he said, including the loss of an emergency medicine physician colleague from death by suicide. “On the national level there were so many other issues going on such as health care disparities of the COVID-19 infection itself, the murder of George Floyd in Minneapolis, other issues of racial injustice, a tense political climate with an upcoming election at the time, and other factors related to the natural climate concerns,” he said. This prompted several faculty members in the CUIMC department of psychiatry including Claude Ann Mellins, PhD, Laurel S. Mayer, MD, and Lourival Baptista-Neto, MD, to partner with ColumbiaDoctors and New York-Presbyterian Hospital and develop a model of care for health care workers known as CopeColumbia, a virtual program intended to address staff burnout and fatigue, with an emphasis on prevention and promotion of resilience.* It launched in March of 2020 and consists of 1:1 peer support, a peer support group program, town halls/webinars, and an active web site.
The 1:1 peer support sessions typically last 20-30 minutes and provide easy access for all distressed hospital and medical center staff. “We have a phone line staffed by Columbia psychiatrists and psychologists so that a distressed staff member can reach support directly,” he said. The format of these sessions includes a brief discussion of challenges and brainstorming around potential coping strategies. “This is not a psychotherapy session,” Dr. Levenson said. “Each session can be individualized to further assess the type of distress or to implement rating scales such as the Generalized Anxiety Disorder-7 scale to assess for signs and symptoms consistent with GAD. There are options to schedule a second or third peer support session, or a prompt referral within Columbia psychiatry when indicated.”
A typical peer support group meeting lasts about 30 minutes and comprises individual divisions or departments. Some goals of the peer groups are to discuss unique challenges of the work environment and to encourage the members of the group to come up with solutions; to promote team support and coping; to teach resilience-enhancing strategies from empirically based treatments such as CBT, “and to end each meeting with expressions of gratitude and of thanks within the group,” he said.
According to Dr. Levenson, sample questions CopeColumbia faculty use to facilitate coping, include “which coping skills are working for you?”; “Are you able to be present?”; “Have you honored loss with any specific ways or traditions?”; “Do you have any work buddies who support you and vice versa?”; “Can your work community build off each other’s individual strengths to help both the individual and the work group cope optimally?”; and “How can your work team help facilitate each other to best support each other?”
Other aspects of the CopeColumbia program include town halls/grand rounds that range from 30 to 60 minutes in length. “It may be a virtual presentation from a mental health professional on specific aspects of coping such as relaxation techniques,” he said. “The focus is how to manage stress, anxiety, trauma, loss, and grief. It also includes an active Q&A to engage staff participants. The advantage of this format is that you can reach many staff in an entire department.” The program also has an active web site for staff with both internal and external support links including mindfulness, meditation, exercise, parenting suggestions/caregiving, and other resources to promote well-being and resilience for staff and family.
To date, certain themes emerged from the 1:1 and peer support group sessions, including expressions of difficulty adapting to “such a new reality,” compared with the pre-COVID era. “Staff would often express anticipatory anxiety and uncertainty, such as is there going to be another surge of COVID-19 cases, and will there be a change in policies?” Dr. Levenson said. “There was a lot of expression of stress and frustration related to politicizing the virus and public containment strategies, both on a local and national level.”
Staff also mentioned the loss of usual coping strategies because of prolonged social isolation, especially for those doing remote work, and the loss of usual support resources that have helped them in the past. “They also reported delayed trauma and grief reactions, including symptoms of depression, anxiety, and posttraumatic stress,” he said. “Health care workers with children mentioned high levels of stress related to childcare, increased workload, and what seems like an impossible work-life balance.” Many reported exhaustion and irritability, “which could affect and cause tension within the work group and challenges to effective team cohesion,” he said. “There were also stressors related to the impact of racial injustices and the [presidential] election that could exacerbate the impact of COVID-19.”
Dr. Levenson hopes that CopeColumbia serves as a model for other health care systems looking for ways to support the mental well-being of their employees. “We want to promote the message that emotional health should have the same priority level as physical health,” he said. “The term that I like to use is total health. Addressing the well-being of health care workers is critical for a healthy workforce and for delivering high-quality patient care.”
He reported having no relevant financial disclosures related to his presentation.
Correction, 2/28/22: An earlier version of this article misstated Dr. Lourival Baptista-Neto's name.
LAS VEGAS – , according to Jon A. Levenson, MD.
“We can learn from previous pandemics and epidemics, which will be important for us going forward from COVID-19,” Dr. Levenson, associate professor of psychiatry at Columbia University Irving Medical Center, New York, said during an annual psychopharmacology update held by the Nevada Psychiatric Association.
During the severe acute respiratory syndrome (SARS) epidemic in 2005, 68% of health care workers reported significant job-related stress, including increased workload, changing work duties, redeployment, shortage of medical supplies, concerns about insufficient personal protective equipment (PPE), lack of safety at work, absence of effective treatment protocols, inconsistent organizational support and information and misinformation from hospital management, and witnessing intense pain, isolation, and loss on a daily basis with few opportunities to take breaks (Psychiatr Serv. 2020 Oct 6. doi: 10.1176/appi.ps.202000274).
Personal concerns associated with psychopathological symptoms included spreading infection to family members; feeling responsibility for family members’ social isolation; self-isolating to avoid infecting family, which can lead to increased loneliness and sadness. “For those who were working remotely, this level of work is hard and challenging,” Dr. Levenson said. “For those who are parents, the 24-hour childcare responsibilities exist on top of work. They often found they can’t unwind with friends.”
Across SARS, MERS, Ebola, and swine flu, a wide range of prevalence in symptoms of distress, stress, anxiety, depressive symptoms, and substance use emerged, he continued. During COVID-19, at least three studies reported significant percentages of distress, depression, anxiety, insomnia, and PTSD among health care workers (JAMA Netw Open. 2020;3[3]:e203976, Front Psychol. 2020 Dec 8;11:608986., and Gen Hosp Psychiatry. Sep-Oct 2020;66:1-8).
“Who is at most-increased risk?” Dr. Levenson asked. “Women; those who are younger and have fewer years of work experience; those working on the front lines such as nurses and advanced practice professionals; and people with preexisting vulnerabilities to psychiatric disorders including anxiety, depression, obsessional symptoms, substance use, suicidal behavior, and impulse control disorders are likely to be especially vulnerable to stress-related symptoms.”
At CUIMC, there were certain “tipping points,” to the vulnerability of health care worker well-being in the early stage of the COVID-19 pandemic, he said, including the loss of an emergency medicine physician colleague from death by suicide. “On the national level there were so many other issues going on such as health care disparities of the COVID-19 infection itself, the murder of George Floyd in Minneapolis, other issues of racial injustice, a tense political climate with an upcoming election at the time, and other factors related to the natural climate concerns,” he said. This prompted several faculty members in the CUIMC department of psychiatry including Claude Ann Mellins, PhD, Laurel S. Mayer, MD, and Lourival Baptista-Neto, MD, to partner with ColumbiaDoctors and New York-Presbyterian Hospital and develop a model of care for health care workers known as CopeColumbia, a virtual program intended to address staff burnout and fatigue, with an emphasis on prevention and promotion of resilience.* It launched in March of 2020 and consists of 1:1 peer support, a peer support group program, town halls/webinars, and an active web site.
The 1:1 peer support sessions typically last 20-30 minutes and provide easy access for all distressed hospital and medical center staff. “We have a phone line staffed by Columbia psychiatrists and psychologists so that a distressed staff member can reach support directly,” he said. The format of these sessions includes a brief discussion of challenges and brainstorming around potential coping strategies. “This is not a psychotherapy session,” Dr. Levenson said. “Each session can be individualized to further assess the type of distress or to implement rating scales such as the Generalized Anxiety Disorder-7 scale to assess for signs and symptoms consistent with GAD. There are options to schedule a second or third peer support session, or a prompt referral within Columbia psychiatry when indicated.”
A typical peer support group meeting lasts about 30 minutes and comprises individual divisions or departments. Some goals of the peer groups are to discuss unique challenges of the work environment and to encourage the members of the group to come up with solutions; to promote team support and coping; to teach resilience-enhancing strategies from empirically based treatments such as CBT, “and to end each meeting with expressions of gratitude and of thanks within the group,” he said.
According to Dr. Levenson, sample questions CopeColumbia faculty use to facilitate coping, include “which coping skills are working for you?”; “Are you able to be present?”; “Have you honored loss with any specific ways or traditions?”; “Do you have any work buddies who support you and vice versa?”; “Can your work community build off each other’s individual strengths to help both the individual and the work group cope optimally?”; and “How can your work team help facilitate each other to best support each other?”
Other aspects of the CopeColumbia program include town halls/grand rounds that range from 30 to 60 minutes in length. “It may be a virtual presentation from a mental health professional on specific aspects of coping such as relaxation techniques,” he said. “The focus is how to manage stress, anxiety, trauma, loss, and grief. It also includes an active Q&A to engage staff participants. The advantage of this format is that you can reach many staff in an entire department.” The program also has an active web site for staff with both internal and external support links including mindfulness, meditation, exercise, parenting suggestions/caregiving, and other resources to promote well-being and resilience for staff and family.
To date, certain themes emerged from the 1:1 and peer support group sessions, including expressions of difficulty adapting to “such a new reality,” compared with the pre-COVID era. “Staff would often express anticipatory anxiety and uncertainty, such as is there going to be another surge of COVID-19 cases, and will there be a change in policies?” Dr. Levenson said. “There was a lot of expression of stress and frustration related to politicizing the virus and public containment strategies, both on a local and national level.”
Staff also mentioned the loss of usual coping strategies because of prolonged social isolation, especially for those doing remote work, and the loss of usual support resources that have helped them in the past. “They also reported delayed trauma and grief reactions, including symptoms of depression, anxiety, and posttraumatic stress,” he said. “Health care workers with children mentioned high levels of stress related to childcare, increased workload, and what seems like an impossible work-life balance.” Many reported exhaustion and irritability, “which could affect and cause tension within the work group and challenges to effective team cohesion,” he said. “There were also stressors related to the impact of racial injustices and the [presidential] election that could exacerbate the impact of COVID-19.”
Dr. Levenson hopes that CopeColumbia serves as a model for other health care systems looking for ways to support the mental well-being of their employees. “We want to promote the message that emotional health should have the same priority level as physical health,” he said. “The term that I like to use is total health. Addressing the well-being of health care workers is critical for a healthy workforce and for delivering high-quality patient care.”
He reported having no relevant financial disclosures related to his presentation.
Correction, 2/28/22: An earlier version of this article misstated Dr. Lourival Baptista-Neto's name.
LAS VEGAS – , according to Jon A. Levenson, MD.
“We can learn from previous pandemics and epidemics, which will be important for us going forward from COVID-19,” Dr. Levenson, associate professor of psychiatry at Columbia University Irving Medical Center, New York, said during an annual psychopharmacology update held by the Nevada Psychiatric Association.
During the severe acute respiratory syndrome (SARS) epidemic in 2005, 68% of health care workers reported significant job-related stress, including increased workload, changing work duties, redeployment, shortage of medical supplies, concerns about insufficient personal protective equipment (PPE), lack of safety at work, absence of effective treatment protocols, inconsistent organizational support and information and misinformation from hospital management, and witnessing intense pain, isolation, and loss on a daily basis with few opportunities to take breaks (Psychiatr Serv. 2020 Oct 6. doi: 10.1176/appi.ps.202000274).
Personal concerns associated with psychopathological symptoms included spreading infection to family members; feeling responsibility for family members’ social isolation; self-isolating to avoid infecting family, which can lead to increased loneliness and sadness. “For those who were working remotely, this level of work is hard and challenging,” Dr. Levenson said. “For those who are parents, the 24-hour childcare responsibilities exist on top of work. They often found they can’t unwind with friends.”
Across SARS, MERS, Ebola, and swine flu, a wide range of prevalence in symptoms of distress, stress, anxiety, depressive symptoms, and substance use emerged, he continued. During COVID-19, at least three studies reported significant percentages of distress, depression, anxiety, insomnia, and PTSD among health care workers (JAMA Netw Open. 2020;3[3]:e203976, Front Psychol. 2020 Dec 8;11:608986., and Gen Hosp Psychiatry. Sep-Oct 2020;66:1-8).
“Who is at most-increased risk?” Dr. Levenson asked. “Women; those who are younger and have fewer years of work experience; those working on the front lines such as nurses and advanced practice professionals; and people with preexisting vulnerabilities to psychiatric disorders including anxiety, depression, obsessional symptoms, substance use, suicidal behavior, and impulse control disorders are likely to be especially vulnerable to stress-related symptoms.”
At CUIMC, there were certain “tipping points,” to the vulnerability of health care worker well-being in the early stage of the COVID-19 pandemic, he said, including the loss of an emergency medicine physician colleague from death by suicide. “On the national level there were so many other issues going on such as health care disparities of the COVID-19 infection itself, the murder of George Floyd in Minneapolis, other issues of racial injustice, a tense political climate with an upcoming election at the time, and other factors related to the natural climate concerns,” he said. This prompted several faculty members in the CUIMC department of psychiatry including Claude Ann Mellins, PhD, Laurel S. Mayer, MD, and Lourival Baptista-Neto, MD, to partner with ColumbiaDoctors and New York-Presbyterian Hospital and develop a model of care for health care workers known as CopeColumbia, a virtual program intended to address staff burnout and fatigue, with an emphasis on prevention and promotion of resilience.* It launched in March of 2020 and consists of 1:1 peer support, a peer support group program, town halls/webinars, and an active web site.
The 1:1 peer support sessions typically last 20-30 minutes and provide easy access for all distressed hospital and medical center staff. “We have a phone line staffed by Columbia psychiatrists and psychologists so that a distressed staff member can reach support directly,” he said. The format of these sessions includes a brief discussion of challenges and brainstorming around potential coping strategies. “This is not a psychotherapy session,” Dr. Levenson said. “Each session can be individualized to further assess the type of distress or to implement rating scales such as the Generalized Anxiety Disorder-7 scale to assess for signs and symptoms consistent with GAD. There are options to schedule a second or third peer support session, or a prompt referral within Columbia psychiatry when indicated.”
A typical peer support group meeting lasts about 30 minutes and comprises individual divisions or departments. Some goals of the peer groups are to discuss unique challenges of the work environment and to encourage the members of the group to come up with solutions; to promote team support and coping; to teach resilience-enhancing strategies from empirically based treatments such as CBT, “and to end each meeting with expressions of gratitude and of thanks within the group,” he said.
According to Dr. Levenson, sample questions CopeColumbia faculty use to facilitate coping, include “which coping skills are working for you?”; “Are you able to be present?”; “Have you honored loss with any specific ways or traditions?”; “Do you have any work buddies who support you and vice versa?”; “Can your work community build off each other’s individual strengths to help both the individual and the work group cope optimally?”; and “How can your work team help facilitate each other to best support each other?”
Other aspects of the CopeColumbia program include town halls/grand rounds that range from 30 to 60 minutes in length. “It may be a virtual presentation from a mental health professional on specific aspects of coping such as relaxation techniques,” he said. “The focus is how to manage stress, anxiety, trauma, loss, and grief. It also includes an active Q&A to engage staff participants. The advantage of this format is that you can reach many staff in an entire department.” The program also has an active web site for staff with both internal and external support links including mindfulness, meditation, exercise, parenting suggestions/caregiving, and other resources to promote well-being and resilience for staff and family.
To date, certain themes emerged from the 1:1 and peer support group sessions, including expressions of difficulty adapting to “such a new reality,” compared with the pre-COVID era. “Staff would often express anticipatory anxiety and uncertainty, such as is there going to be another surge of COVID-19 cases, and will there be a change in policies?” Dr. Levenson said. “There was a lot of expression of stress and frustration related to politicizing the virus and public containment strategies, both on a local and national level.”
Staff also mentioned the loss of usual coping strategies because of prolonged social isolation, especially for those doing remote work, and the loss of usual support resources that have helped them in the past. “They also reported delayed trauma and grief reactions, including symptoms of depression, anxiety, and posttraumatic stress,” he said. “Health care workers with children mentioned high levels of stress related to childcare, increased workload, and what seems like an impossible work-life balance.” Many reported exhaustion and irritability, “which could affect and cause tension within the work group and challenges to effective team cohesion,” he said. “There were also stressors related to the impact of racial injustices and the [presidential] election that could exacerbate the impact of COVID-19.”
Dr. Levenson hopes that CopeColumbia serves as a model for other health care systems looking for ways to support the mental well-being of their employees. “We want to promote the message that emotional health should have the same priority level as physical health,” he said. “The term that I like to use is total health. Addressing the well-being of health care workers is critical for a healthy workforce and for delivering high-quality patient care.”
He reported having no relevant financial disclosures related to his presentation.
Correction, 2/28/22: An earlier version of this article misstated Dr. Lourival Baptista-Neto's name.
FROM NPA 2022
Why pregnant people were left behind while vaccines moved at ‘warp speed’ to help the masses
Kia Slade was 7 months pregnant, unvaccinated, and fighting for breath, her oxygen levels plummeting, when her son came into the world last May.
A severe case of COVID-19 pneumonia had left Ms. Slade delirious. When the intensive care team tried to place an oxygen mask on her face, she snatched it away, she recalled. Her baby’s heart rate began to drop.
Ms. Slade’s doctor performed an emergency cesarean section at her bedside in the intensive care unit, delivering baby Tristan 10 weeks early. He weighed just 2 pounds, 14 ounces, about half the size of small full-term baby.
But Ms. Slade wouldn’t meet him until July. She was on a ventilator in a medically-induced coma for 8 weeks, and she developed a serious infection and blood clot while unconscious. It was only after a perilous 2½ months in the hospital, during which her heart stopped twice, that Ms. Slade was vaccinated against COVID-19.
“I wish I had gotten the vaccine earlier,” said Ms. Slade, 42, who remains too sick to return to work as a special education teacher in Baltimore. Doctors “kept pushing me to get vaccinated, but there just wasn’t enough information out there for me to do it.”
A year ago, there was little to no vaccine safety data for pregnant people like Ms. Slade, because they had been excluded from clinical trials run by Pfizer, Moderna, and other vaccine makers.
Lacking data, health experts were unsure and divided about how to advise expectant parents. Although U.S. health officials permitted pregnant people to be vaccinated, the World Health Organization in January 2021 actually discouraged them from doing so; it later reversed that recommendation.
The uncertainty led many women to delay vaccination, and only about two-thirds of the pregnant people who have been tracked by the Centers for Disease Control and Prevention were fully vaccinated as of Feb. 5, 2022, leaving many expectant moms at a high risk of infection and life-threatening complications.
More than 29,000 pregnant people have been hospitalized with COVID-19 and 274 have died, according to the CDC.
“There were surely women who were hospitalized because there wasn’t information available to them,” said Paul A. Offit, MD, director of the Vaccine Education Center at the Children’s Hospital of Philadelphia.
Vaccine developers say that pregnant people – who have special health needs and risks – were excluded from clinical trials to protect them from potential side effects of novel technologies, including the Pfizer and Moderna mRNA vaccines and formulations made with cold viruses, such as the Johnson & Johnson vaccine.
But a KHN analysis also shows that pregnant people were left behind because including them in vaccine studies would have complicated and potentially delayed the delivery of COVID-19 vaccines to the broader population.
A growing number of women’s health researchers and advocates say that excluding pregnant people – and the months-long delay in recommending that they be immunized – helped fuel widespread vaccine hesitancy in this vulnerable group.
“Women and their unborn fetuses are dying of COVID infection,” said Jane Van Dis, MD, an ob.gyn. at the University of Rochester (N.Y.) Medical Center who has treated many patients like Ms. Slade. “Our failure as a society to vaccinate women in pregnancy will be remembered by the children and families who lost their mothers to this disease.”
New technology, uncertain risks
At the time COVID-19 vaccines were being developed, scientists had very little experience using mRNA vaccines in pregnant women, said Jacqueline Miller, MD, a senior vice president involved in vaccine research at Moderna.
“When you study anything in pregnant women, you have two patients, the mom and the unborn child,” Dr. Miller said. “Until we had more safety data on the platform, it wasn’t something we wanted to undertake.”
But Dr. Offit noted that vaccines have a strong record of safety in pregnancy and he sees no reason to have excluded pregnant people. None of the vaccines currently in use – including the chickenpox and rubella vaccines, which contain live viruses – have been shown to harm fetuses, he said. Doctors routinely recommend that pregnant people receive pertussis and flu vaccinations.
Dr. Offit, the coinventor of a rotavirus vaccine, said that some concerns about vaccines stem from commercial, not medical, interests. Drug makers don’t want to risk that their product will be blamed for any problems occurring in pregnant people, even if coincidental, he said.
“These companies don’t want bad news,” Dr. Offit said.
In the United States, health officials typically would have told expectant mothers not to take a vaccine that was untested during pregnancy, said Dr. Offit, a member of a committee that advises the Food and Drug Administration on vaccines.
Due to the urgency of the pandemic, health agencies instead permitted pregnant people to make up their own minds about vaccines without recommending them.
Women’s medical associations were also hampered by the lack of data. Neither the American College of Obstetricians and Gynecologists nor the Society for Maternal-Fetal Medicine actively encouraged pregnant people to be vaccinated until July 30, 2021, after the first real-world vaccine studies had been published. The CDC followed suit in August of 2021.
“If we had had this data in the beginning, we would have been able to vaccinate more women,” said Kelli Burroughs, MD, the department chair of obstetrics and gynecology at Memorial Hermann Sugar Land Hospital near Houston.
Yet anti-vaccine groups wasted no time in scaring pregnant people, flooding social media with misinformation about impaired fertility and harm to the fetus.
In the first few months after the COVID-19 vaccines were approved, some doctors were ambivalent about recommending them, and some still advise pregnant patients against vaccination.
An estimated 67% of pregnant people today are fully vaccinated, compared with about 89% of people 65 and older, another high-risk group, and 65% of Americans overall. Vaccination rates are lower among minorities, with 65% of expectant Hispanic mothers and 53% of pregnant African Americans fully vaccinated, according to the CDC.
Vaccination is especially important during pregnancy, because of increased risks of hospitalization, ICU admission, and mechanical ventilation, Dr. Burroughs said. A study released in February from the National Institutes of Health found that pregnant people with a moderate to severe COVID-19 infection also were more likely to have a C-section, deliver preterm, or develop a postpartum hemorrhage.
Black moms such as Ms. Slade were already at higher risk of maternal and infant mortality before the pandemic, because of higher underlying risks, unequal access to health care, and other factors. COVID-19 has magnified those risks, said Dr. Burroughs, who has persuaded reluctant patients by revealing that she had a healthy pregnancy and child after being vaccinated.
Ms. Slade said she has never opposed vaccines and had no hesitation about receiving other vaccines while pregnant. But she said she “just wasn’t comfortable” with COVID-19 shots.
“If there had been data out there saying the COVID shot was safe, and that nothing would happen to my baby and there was no risk of birth defects, I would have taken it,” said Ms. Slade, who has had type 2 diabetes for 12 years.
Working at warp speed
Government scientists at the NIH were concerned about the risk of COVID-19 to pregnant people from the very beginning and knew that expectant moms needed vaccines as much or more than anyone else, said Larry Corey, MD, a leader of the COVID-19 Prevention Network, which coordinated COVID-19 vaccine trials for the federal government.
But including pregnant volunteers in the larger vaccine trials could have led to interruptions and delays, Dr. Corey said. Researchers would have had to enroll thousands of pregnant volunteers to achieve statistically robust results that weren’t due to chance, he said.
Pregnancy can bring on a wide range of complications: gestational diabetes, hypertension, anemia, bleeding, blood clots, or problems with the placenta, for example. Up to 20% of people who know they’re pregnant miscarry. Because researchers would have been obliged to investigate any medical problem to make sure it wasn’t caused by one of the COVID-19 vaccines, including pregnant people might have meant having to hit pause on those trials, Dr. Corey said.
With death tolls from the pandemic mounting, “we had a mission to do this as quickly and as thoroughly as possible,” Dr. Corey said. Making COVID-19 vaccines available within a year “saved hundreds of thousands of lives.”
The first data on COVID-19 vaccine safety in pregnancy was published in April of 2021 when the CDC released an analysis of nearly 36,000 vaccinated pregnant people who had enrolled in a registry called V-safe, which allows users to log the dates of their vaccinations and any subsequent symptoms.
Later research showed that COVID-19 vaccines weren’t associated with increased risk of miscarriage or premature delivery.
Brenna Hughes, MD, a maternal-fetal medicine specialist and member of the American College of Obstetricians and Gynecologists’ COVID-19 expert group, agrees that adding pregnant people to large-scale COVID-19 vaccine and drug trials may have been impractical. But researchers could have launched parallel trials of pregnant women, once early studies showed the vaccines were safe in humans, she said.
“Would it have been hard? Everything with COVID is hard,” Dr. Hughes said. “But it would have been feasible.”
The FDA requires that researchers perform additional animal studies – called developmental and reproductive toxicity studies – before testing vaccines in pregnant people. Although these studies are essential, they take 5-6 months, and weren’t completed until late 2020, around the time the first COVID-19 vaccines were authorized for adults, said Emily Erbelding, MD, director of microbiology and infectious diseases at the National Institute of Allergy and Infectious Diseases, part of the NIH.
Pregnancy studies “were an afterthought,” said Irina Burd, MD, director of Johns Hopkins’ Integrated Research Center for Fetal Medicine and a professor of gynecology and obstetrics. “They should have been done sooner.”
The NIH is conducting a study of pregnant and postpartum people who decided on their own to be vaccinated, Dr. Erbelding said. The study is due to be completed by July 2023.
Janssen and Moderna are also conducting studies in pregnant people, both due to be completed in 2024.
Pfizer scientists encountered problems when they initiated a clinical trial, which would have randomly assigned pregnant people to receive either a vaccine or placebo. Once vaccines were widely available, many patients weren’t willing to take a chance on being unvaccinated until after delivery.
Pfizer has stopped recruiting patients and has not said whether it will publicly report any data from the trial.
Dr. Hughes said vaccine developers need to include pregnant people from the very beginning.
“There is this notion of protecting pregnant people from research,” Dr. Hughes said. “But we should be protecting patients through research, not from research.”
Recovering physically and emotionally
Ms. Slade still regrets being deprived of time with her children while she fought the disease.
Being on a ventilator kept her from spending those early weeks with her newborn, or from seeing her 9-year-old daughter, Zoe.
Even when Ms. Slade was finally able to see her son, she wasn’t able to tell him she loved him or sing a lullaby, or even talk at all, because of a breathing tube in her throat.
Today, Ms. Slade is a strong advocate of COVID-19 vaccinations, urging her friends and family to get their shots to avoid suffering the way she has.
Ms. Slade had to relearn to walk after being bedridden for weeks. Her many weeks on a ventilator may have contributed to her stomach paralysis, which often causes intense pain, nausea, and even vomiting when she eats or drinks. Ms. Slade weighs 50 pounds less today than before she became pregnant and has resorted to going to the emergency department when the pain is unbearable. “Most days, I’m just miserable,” she said.
Her family suffered as well. Like many babies born prematurely, Tristan, now nearly 9 months old and crawling, receives physical therapy to strengthen his muscles. At 15 pounds, Tristan is largely healthy, although his doctor said he has symptoms of asthma.
Ms. Slade said she would like to attend family counseling with Zoe, who rarely complains and tends to keep her feelings to herself. Ms. Slade said she knows her illness must have been terrifying for her little girl.
“The other day she was talking to me,” Ms. Slade said, “and she said, ‘You know, I almost had to bury you.’ ”
KHN (Kaiser Health News) is a national newsroom that produces in-depth journalism about health issues. Together with Policy Analysis and Polling, KHN is one of the three major operating programs at KFF (Kaiser Family Foundation). KFF is an endowed nonprofit organization providing information on health issues to the nation.
Kia Slade was 7 months pregnant, unvaccinated, and fighting for breath, her oxygen levels plummeting, when her son came into the world last May.
A severe case of COVID-19 pneumonia had left Ms. Slade delirious. When the intensive care team tried to place an oxygen mask on her face, she snatched it away, she recalled. Her baby’s heart rate began to drop.
Ms. Slade’s doctor performed an emergency cesarean section at her bedside in the intensive care unit, delivering baby Tristan 10 weeks early. He weighed just 2 pounds, 14 ounces, about half the size of small full-term baby.
But Ms. Slade wouldn’t meet him until July. She was on a ventilator in a medically-induced coma for 8 weeks, and she developed a serious infection and blood clot while unconscious. It was only after a perilous 2½ months in the hospital, during which her heart stopped twice, that Ms. Slade was vaccinated against COVID-19.
“I wish I had gotten the vaccine earlier,” said Ms. Slade, 42, who remains too sick to return to work as a special education teacher in Baltimore. Doctors “kept pushing me to get vaccinated, but there just wasn’t enough information out there for me to do it.”
A year ago, there was little to no vaccine safety data for pregnant people like Ms. Slade, because they had been excluded from clinical trials run by Pfizer, Moderna, and other vaccine makers.
Lacking data, health experts were unsure and divided about how to advise expectant parents. Although U.S. health officials permitted pregnant people to be vaccinated, the World Health Organization in January 2021 actually discouraged them from doing so; it later reversed that recommendation.
The uncertainty led many women to delay vaccination, and only about two-thirds of the pregnant people who have been tracked by the Centers for Disease Control and Prevention were fully vaccinated as of Feb. 5, 2022, leaving many expectant moms at a high risk of infection and life-threatening complications.
More than 29,000 pregnant people have been hospitalized with COVID-19 and 274 have died, according to the CDC.
“There were surely women who were hospitalized because there wasn’t information available to them,” said Paul A. Offit, MD, director of the Vaccine Education Center at the Children’s Hospital of Philadelphia.
Vaccine developers say that pregnant people – who have special health needs and risks – were excluded from clinical trials to protect them from potential side effects of novel technologies, including the Pfizer and Moderna mRNA vaccines and formulations made with cold viruses, such as the Johnson & Johnson vaccine.
But a KHN analysis also shows that pregnant people were left behind because including them in vaccine studies would have complicated and potentially delayed the delivery of COVID-19 vaccines to the broader population.
A growing number of women’s health researchers and advocates say that excluding pregnant people – and the months-long delay in recommending that they be immunized – helped fuel widespread vaccine hesitancy in this vulnerable group.
“Women and their unborn fetuses are dying of COVID infection,” said Jane Van Dis, MD, an ob.gyn. at the University of Rochester (N.Y.) Medical Center who has treated many patients like Ms. Slade. “Our failure as a society to vaccinate women in pregnancy will be remembered by the children and families who lost their mothers to this disease.”
New technology, uncertain risks
At the time COVID-19 vaccines were being developed, scientists had very little experience using mRNA vaccines in pregnant women, said Jacqueline Miller, MD, a senior vice president involved in vaccine research at Moderna.
“When you study anything in pregnant women, you have two patients, the mom and the unborn child,” Dr. Miller said. “Until we had more safety data on the platform, it wasn’t something we wanted to undertake.”
But Dr. Offit noted that vaccines have a strong record of safety in pregnancy and he sees no reason to have excluded pregnant people. None of the vaccines currently in use – including the chickenpox and rubella vaccines, which contain live viruses – have been shown to harm fetuses, he said. Doctors routinely recommend that pregnant people receive pertussis and flu vaccinations.
Dr. Offit, the coinventor of a rotavirus vaccine, said that some concerns about vaccines stem from commercial, not medical, interests. Drug makers don’t want to risk that their product will be blamed for any problems occurring in pregnant people, even if coincidental, he said.
“These companies don’t want bad news,” Dr. Offit said.
In the United States, health officials typically would have told expectant mothers not to take a vaccine that was untested during pregnancy, said Dr. Offit, a member of a committee that advises the Food and Drug Administration on vaccines.
Due to the urgency of the pandemic, health agencies instead permitted pregnant people to make up their own minds about vaccines without recommending them.
Women’s medical associations were also hampered by the lack of data. Neither the American College of Obstetricians and Gynecologists nor the Society for Maternal-Fetal Medicine actively encouraged pregnant people to be vaccinated until July 30, 2021, after the first real-world vaccine studies had been published. The CDC followed suit in August of 2021.
“If we had had this data in the beginning, we would have been able to vaccinate more women,” said Kelli Burroughs, MD, the department chair of obstetrics and gynecology at Memorial Hermann Sugar Land Hospital near Houston.
Yet anti-vaccine groups wasted no time in scaring pregnant people, flooding social media with misinformation about impaired fertility and harm to the fetus.
In the first few months after the COVID-19 vaccines were approved, some doctors were ambivalent about recommending them, and some still advise pregnant patients against vaccination.
An estimated 67% of pregnant people today are fully vaccinated, compared with about 89% of people 65 and older, another high-risk group, and 65% of Americans overall. Vaccination rates are lower among minorities, with 65% of expectant Hispanic mothers and 53% of pregnant African Americans fully vaccinated, according to the CDC.
Vaccination is especially important during pregnancy, because of increased risks of hospitalization, ICU admission, and mechanical ventilation, Dr. Burroughs said. A study released in February from the National Institutes of Health found that pregnant people with a moderate to severe COVID-19 infection also were more likely to have a C-section, deliver preterm, or develop a postpartum hemorrhage.
Black moms such as Ms. Slade were already at higher risk of maternal and infant mortality before the pandemic, because of higher underlying risks, unequal access to health care, and other factors. COVID-19 has magnified those risks, said Dr. Burroughs, who has persuaded reluctant patients by revealing that she had a healthy pregnancy and child after being vaccinated.
Ms. Slade said she has never opposed vaccines and had no hesitation about receiving other vaccines while pregnant. But she said she “just wasn’t comfortable” with COVID-19 shots.
“If there had been data out there saying the COVID shot was safe, and that nothing would happen to my baby and there was no risk of birth defects, I would have taken it,” said Ms. Slade, who has had type 2 diabetes for 12 years.
Working at warp speed
Government scientists at the NIH were concerned about the risk of COVID-19 to pregnant people from the very beginning and knew that expectant moms needed vaccines as much or more than anyone else, said Larry Corey, MD, a leader of the COVID-19 Prevention Network, which coordinated COVID-19 vaccine trials for the federal government.
But including pregnant volunteers in the larger vaccine trials could have led to interruptions and delays, Dr. Corey said. Researchers would have had to enroll thousands of pregnant volunteers to achieve statistically robust results that weren’t due to chance, he said.
Pregnancy can bring on a wide range of complications: gestational diabetes, hypertension, anemia, bleeding, blood clots, or problems with the placenta, for example. Up to 20% of people who know they’re pregnant miscarry. Because researchers would have been obliged to investigate any medical problem to make sure it wasn’t caused by one of the COVID-19 vaccines, including pregnant people might have meant having to hit pause on those trials, Dr. Corey said.
With death tolls from the pandemic mounting, “we had a mission to do this as quickly and as thoroughly as possible,” Dr. Corey said. Making COVID-19 vaccines available within a year “saved hundreds of thousands of lives.”
The first data on COVID-19 vaccine safety in pregnancy was published in April of 2021 when the CDC released an analysis of nearly 36,000 vaccinated pregnant people who had enrolled in a registry called V-safe, which allows users to log the dates of their vaccinations and any subsequent symptoms.
Later research showed that COVID-19 vaccines weren’t associated with increased risk of miscarriage or premature delivery.
Brenna Hughes, MD, a maternal-fetal medicine specialist and member of the American College of Obstetricians and Gynecologists’ COVID-19 expert group, agrees that adding pregnant people to large-scale COVID-19 vaccine and drug trials may have been impractical. But researchers could have launched parallel trials of pregnant women, once early studies showed the vaccines were safe in humans, she said.
“Would it have been hard? Everything with COVID is hard,” Dr. Hughes said. “But it would have been feasible.”
The FDA requires that researchers perform additional animal studies – called developmental and reproductive toxicity studies – before testing vaccines in pregnant people. Although these studies are essential, they take 5-6 months, and weren’t completed until late 2020, around the time the first COVID-19 vaccines were authorized for adults, said Emily Erbelding, MD, director of microbiology and infectious diseases at the National Institute of Allergy and Infectious Diseases, part of the NIH.
Pregnancy studies “were an afterthought,” said Irina Burd, MD, director of Johns Hopkins’ Integrated Research Center for Fetal Medicine and a professor of gynecology and obstetrics. “They should have been done sooner.”
The NIH is conducting a study of pregnant and postpartum people who decided on their own to be vaccinated, Dr. Erbelding said. The study is due to be completed by July 2023.
Janssen and Moderna are also conducting studies in pregnant people, both due to be completed in 2024.
Pfizer scientists encountered problems when they initiated a clinical trial, which would have randomly assigned pregnant people to receive either a vaccine or placebo. Once vaccines were widely available, many patients weren’t willing to take a chance on being unvaccinated until after delivery.
Pfizer has stopped recruiting patients and has not said whether it will publicly report any data from the trial.
Dr. Hughes said vaccine developers need to include pregnant people from the very beginning.
“There is this notion of protecting pregnant people from research,” Dr. Hughes said. “But we should be protecting patients through research, not from research.”
Recovering physically and emotionally
Ms. Slade still regrets being deprived of time with her children while she fought the disease.
Being on a ventilator kept her from spending those early weeks with her newborn, or from seeing her 9-year-old daughter, Zoe.
Even when Ms. Slade was finally able to see her son, she wasn’t able to tell him she loved him or sing a lullaby, or even talk at all, because of a breathing tube in her throat.
Today, Ms. Slade is a strong advocate of COVID-19 vaccinations, urging her friends and family to get their shots to avoid suffering the way she has.
Ms. Slade had to relearn to walk after being bedridden for weeks. Her many weeks on a ventilator may have contributed to her stomach paralysis, which often causes intense pain, nausea, and even vomiting when she eats or drinks. Ms. Slade weighs 50 pounds less today than before she became pregnant and has resorted to going to the emergency department when the pain is unbearable. “Most days, I’m just miserable,” she said.
Her family suffered as well. Like many babies born prematurely, Tristan, now nearly 9 months old and crawling, receives physical therapy to strengthen his muscles. At 15 pounds, Tristan is largely healthy, although his doctor said he has symptoms of asthma.
Ms. Slade said she would like to attend family counseling with Zoe, who rarely complains and tends to keep her feelings to herself. Ms. Slade said she knows her illness must have been terrifying for her little girl.
“The other day she was talking to me,” Ms. Slade said, “and she said, ‘You know, I almost had to bury you.’ ”
KHN (Kaiser Health News) is a national newsroom that produces in-depth journalism about health issues. Together with Policy Analysis and Polling, KHN is one of the three major operating programs at KFF (Kaiser Family Foundation). KFF is an endowed nonprofit organization providing information on health issues to the nation.
Kia Slade was 7 months pregnant, unvaccinated, and fighting for breath, her oxygen levels plummeting, when her son came into the world last May.
A severe case of COVID-19 pneumonia had left Ms. Slade delirious. When the intensive care team tried to place an oxygen mask on her face, she snatched it away, she recalled. Her baby’s heart rate began to drop.
Ms. Slade’s doctor performed an emergency cesarean section at her bedside in the intensive care unit, delivering baby Tristan 10 weeks early. He weighed just 2 pounds, 14 ounces, about half the size of small full-term baby.
But Ms. Slade wouldn’t meet him until July. She was on a ventilator in a medically-induced coma for 8 weeks, and she developed a serious infection and blood clot while unconscious. It was only after a perilous 2½ months in the hospital, during which her heart stopped twice, that Ms. Slade was vaccinated against COVID-19.
“I wish I had gotten the vaccine earlier,” said Ms. Slade, 42, who remains too sick to return to work as a special education teacher in Baltimore. Doctors “kept pushing me to get vaccinated, but there just wasn’t enough information out there for me to do it.”
A year ago, there was little to no vaccine safety data for pregnant people like Ms. Slade, because they had been excluded from clinical trials run by Pfizer, Moderna, and other vaccine makers.
Lacking data, health experts were unsure and divided about how to advise expectant parents. Although U.S. health officials permitted pregnant people to be vaccinated, the World Health Organization in January 2021 actually discouraged them from doing so; it later reversed that recommendation.
The uncertainty led many women to delay vaccination, and only about two-thirds of the pregnant people who have been tracked by the Centers for Disease Control and Prevention were fully vaccinated as of Feb. 5, 2022, leaving many expectant moms at a high risk of infection and life-threatening complications.
More than 29,000 pregnant people have been hospitalized with COVID-19 and 274 have died, according to the CDC.
“There were surely women who were hospitalized because there wasn’t information available to them,” said Paul A. Offit, MD, director of the Vaccine Education Center at the Children’s Hospital of Philadelphia.
Vaccine developers say that pregnant people – who have special health needs and risks – were excluded from clinical trials to protect them from potential side effects of novel technologies, including the Pfizer and Moderna mRNA vaccines and formulations made with cold viruses, such as the Johnson & Johnson vaccine.
But a KHN analysis also shows that pregnant people were left behind because including them in vaccine studies would have complicated and potentially delayed the delivery of COVID-19 vaccines to the broader population.
A growing number of women’s health researchers and advocates say that excluding pregnant people – and the months-long delay in recommending that they be immunized – helped fuel widespread vaccine hesitancy in this vulnerable group.
“Women and their unborn fetuses are dying of COVID infection,” said Jane Van Dis, MD, an ob.gyn. at the University of Rochester (N.Y.) Medical Center who has treated many patients like Ms. Slade. “Our failure as a society to vaccinate women in pregnancy will be remembered by the children and families who lost their mothers to this disease.”
New technology, uncertain risks
At the time COVID-19 vaccines were being developed, scientists had very little experience using mRNA vaccines in pregnant women, said Jacqueline Miller, MD, a senior vice president involved in vaccine research at Moderna.
“When you study anything in pregnant women, you have two patients, the mom and the unborn child,” Dr. Miller said. “Until we had more safety data on the platform, it wasn’t something we wanted to undertake.”
But Dr. Offit noted that vaccines have a strong record of safety in pregnancy and he sees no reason to have excluded pregnant people. None of the vaccines currently in use – including the chickenpox and rubella vaccines, which contain live viruses – have been shown to harm fetuses, he said. Doctors routinely recommend that pregnant people receive pertussis and flu vaccinations.
Dr. Offit, the coinventor of a rotavirus vaccine, said that some concerns about vaccines stem from commercial, not medical, interests. Drug makers don’t want to risk that their product will be blamed for any problems occurring in pregnant people, even if coincidental, he said.
“These companies don’t want bad news,” Dr. Offit said.
In the United States, health officials typically would have told expectant mothers not to take a vaccine that was untested during pregnancy, said Dr. Offit, a member of a committee that advises the Food and Drug Administration on vaccines.
Due to the urgency of the pandemic, health agencies instead permitted pregnant people to make up their own minds about vaccines without recommending them.
Women’s medical associations were also hampered by the lack of data. Neither the American College of Obstetricians and Gynecologists nor the Society for Maternal-Fetal Medicine actively encouraged pregnant people to be vaccinated until July 30, 2021, after the first real-world vaccine studies had been published. The CDC followed suit in August of 2021.
“If we had had this data in the beginning, we would have been able to vaccinate more women,” said Kelli Burroughs, MD, the department chair of obstetrics and gynecology at Memorial Hermann Sugar Land Hospital near Houston.
Yet anti-vaccine groups wasted no time in scaring pregnant people, flooding social media with misinformation about impaired fertility and harm to the fetus.
In the first few months after the COVID-19 vaccines were approved, some doctors were ambivalent about recommending them, and some still advise pregnant patients against vaccination.
An estimated 67% of pregnant people today are fully vaccinated, compared with about 89% of people 65 and older, another high-risk group, and 65% of Americans overall. Vaccination rates are lower among minorities, with 65% of expectant Hispanic mothers and 53% of pregnant African Americans fully vaccinated, according to the CDC.
Vaccination is especially important during pregnancy, because of increased risks of hospitalization, ICU admission, and mechanical ventilation, Dr. Burroughs said. A study released in February from the National Institutes of Health found that pregnant people with a moderate to severe COVID-19 infection also were more likely to have a C-section, deliver preterm, or develop a postpartum hemorrhage.
Black moms such as Ms. Slade were already at higher risk of maternal and infant mortality before the pandemic, because of higher underlying risks, unequal access to health care, and other factors. COVID-19 has magnified those risks, said Dr. Burroughs, who has persuaded reluctant patients by revealing that she had a healthy pregnancy and child after being vaccinated.
Ms. Slade said she has never opposed vaccines and had no hesitation about receiving other vaccines while pregnant. But she said she “just wasn’t comfortable” with COVID-19 shots.
“If there had been data out there saying the COVID shot was safe, and that nothing would happen to my baby and there was no risk of birth defects, I would have taken it,” said Ms. Slade, who has had type 2 diabetes for 12 years.
Working at warp speed
Government scientists at the NIH were concerned about the risk of COVID-19 to pregnant people from the very beginning and knew that expectant moms needed vaccines as much or more than anyone else, said Larry Corey, MD, a leader of the COVID-19 Prevention Network, which coordinated COVID-19 vaccine trials for the federal government.
But including pregnant volunteers in the larger vaccine trials could have led to interruptions and delays, Dr. Corey said. Researchers would have had to enroll thousands of pregnant volunteers to achieve statistically robust results that weren’t due to chance, he said.
Pregnancy can bring on a wide range of complications: gestational diabetes, hypertension, anemia, bleeding, blood clots, or problems with the placenta, for example. Up to 20% of people who know they’re pregnant miscarry. Because researchers would have been obliged to investigate any medical problem to make sure it wasn’t caused by one of the COVID-19 vaccines, including pregnant people might have meant having to hit pause on those trials, Dr. Corey said.
With death tolls from the pandemic mounting, “we had a mission to do this as quickly and as thoroughly as possible,” Dr. Corey said. Making COVID-19 vaccines available within a year “saved hundreds of thousands of lives.”
The first data on COVID-19 vaccine safety in pregnancy was published in April of 2021 when the CDC released an analysis of nearly 36,000 vaccinated pregnant people who had enrolled in a registry called V-safe, which allows users to log the dates of their vaccinations and any subsequent symptoms.
Later research showed that COVID-19 vaccines weren’t associated with increased risk of miscarriage or premature delivery.
Brenna Hughes, MD, a maternal-fetal medicine specialist and member of the American College of Obstetricians and Gynecologists’ COVID-19 expert group, agrees that adding pregnant people to large-scale COVID-19 vaccine and drug trials may have been impractical. But researchers could have launched parallel trials of pregnant women, once early studies showed the vaccines were safe in humans, she said.
“Would it have been hard? Everything with COVID is hard,” Dr. Hughes said. “But it would have been feasible.”
The FDA requires that researchers perform additional animal studies – called developmental and reproductive toxicity studies – before testing vaccines in pregnant people. Although these studies are essential, they take 5-6 months, and weren’t completed until late 2020, around the time the first COVID-19 vaccines were authorized for adults, said Emily Erbelding, MD, director of microbiology and infectious diseases at the National Institute of Allergy and Infectious Diseases, part of the NIH.
Pregnancy studies “were an afterthought,” said Irina Burd, MD, director of Johns Hopkins’ Integrated Research Center for Fetal Medicine and a professor of gynecology and obstetrics. “They should have been done sooner.”
The NIH is conducting a study of pregnant and postpartum people who decided on their own to be vaccinated, Dr. Erbelding said. The study is due to be completed by July 2023.
Janssen and Moderna are also conducting studies in pregnant people, both due to be completed in 2024.
Pfizer scientists encountered problems when they initiated a clinical trial, which would have randomly assigned pregnant people to receive either a vaccine or placebo. Once vaccines were widely available, many patients weren’t willing to take a chance on being unvaccinated until after delivery.
Pfizer has stopped recruiting patients and has not said whether it will publicly report any data from the trial.
Dr. Hughes said vaccine developers need to include pregnant people from the very beginning.
“There is this notion of protecting pregnant people from research,” Dr. Hughes said. “But we should be protecting patients through research, not from research.”
Recovering physically and emotionally
Ms. Slade still regrets being deprived of time with her children while she fought the disease.
Being on a ventilator kept her from spending those early weeks with her newborn, or from seeing her 9-year-old daughter, Zoe.
Even when Ms. Slade was finally able to see her son, she wasn’t able to tell him she loved him or sing a lullaby, or even talk at all, because of a breathing tube in her throat.
Today, Ms. Slade is a strong advocate of COVID-19 vaccinations, urging her friends and family to get their shots to avoid suffering the way she has.
Ms. Slade had to relearn to walk after being bedridden for weeks. Her many weeks on a ventilator may have contributed to her stomach paralysis, which often causes intense pain, nausea, and even vomiting when she eats or drinks. Ms. Slade weighs 50 pounds less today than before she became pregnant and has resorted to going to the emergency department when the pain is unbearable. “Most days, I’m just miserable,” she said.
Her family suffered as well. Like many babies born prematurely, Tristan, now nearly 9 months old and crawling, receives physical therapy to strengthen his muscles. At 15 pounds, Tristan is largely healthy, although his doctor said he has symptoms of asthma.
Ms. Slade said she would like to attend family counseling with Zoe, who rarely complains and tends to keep her feelings to herself. Ms. Slade said she knows her illness must have been terrifying for her little girl.
“The other day she was talking to me,” Ms. Slade said, “and she said, ‘You know, I almost had to bury you.’ ”
KHN (Kaiser Health News) is a national newsroom that produces in-depth journalism about health issues. Together with Policy Analysis and Polling, KHN is one of the three major operating programs at KFF (Kaiser Family Foundation). KFF is an endowed nonprofit organization providing information on health issues to the nation.
Next-generation Dexcom CGM, G7, accurate and easier to use
The Dexcom G7 continuous glucose monitor (CGM) is as accurate or better than other currently marketed CGM devices for measuring glucose in those with diabetes, new data from a pivotal study suggest.
Currently under review by the U.S. Food and Drug Administration, the G7 is expected to be an improvement over the Dexcom G6 version in several ways.
The on-body size will be 60% smaller, “roughly the size of three stacked quarters,” according to the authors, and will incorporate the sensor with a single-use transmitter, as opposed to the current separate 3-month transmitter used with the G6 sensor. This will eliminate the need for using a transmitter across multiple sensor sessions (as is the case for G6).
The warm-up period after insertion is reduced from 2 hours to 27 minutes, and users are given an extra 12-hour “grace period” after the 10-day wear period to change the device before it stops displaying glucose data. Up to 24 hours of missed data can also be recaptured.
“The enhanced features of G7 may increase clinical adoption, encourage sustained use, and reduce the burden of diabetes management,” write Satish K. Garg, MD, of the University of Colorado, Aurora, and colleagues, in their article, published online Feb. 14 in Diabetes Technology & Therapeutics.
Several features of the G6 remain unchanged, including factory calibration, but also the capacity for optional user calibrations, use of Bluetooth to transmit data up to 20 feet (approximately 6 meters), and data displays on either a dedicated receiver or a variety of iOS and Android smart devices.
It will also allow for user-customized settings and alerts, as well as the option for real-time remote “share” monitoring with caregivers or providers. The G7 will also not be susceptible to interference by acetaminophen (paracetamol) or ascorbic acid.
And, like the G6, the G7 was submitted for approval to the FDA as an “integrated CGM,” meaning that it will be interoperable with other compatible devices, including insulin pumps, glucose meters, or other electronic devices used for diabetes management.
Accuracy shown on abdomen, arm
The prospective, multicenter, single-arm study reported by Dr. Garg and colleagues was conducted at 12 U.S. sites between February and June 2021.
In-clinic visits were conducted on days 1 or 2, 4 or 7, and one additional day for comparisons with a reference glucose measure (YSI 2300 Stat Plus glucose analyzer). Participants wore blinded G7 sensors concurrently on the upper arm and abdomen while continuing to use their own personal glucose monitoring method (CGM or fingerstick) at home.
A total of 316 insulin-using adults with type 1 or type 2 diabetes contributed data from 308 arm- and 311 abdomen-placed blinded devices, which generated 77,774 matched pairs of data within the blood glucose reportable range of 40-400 mg/dL.
The overall mean absolute relative difference (MARD) of each CGM-YSI pair (a standard metric for CGM analysis) was 8.2%, with 9.1% for the abdomen and 8.2% for the arm.
Accuracy remained high in both arm- and abdomen-placed sensors across the 10-day wear period through the 12-hour grace period and across glucose ranges. There were no significant differences between G7 accuracy by diabetes type, insulin regimen, or body mass index.
The highest agreement rates and lowest MARDs occurred when CGM readings were increasing or decreasing by no more than 1 mg/dL per minute. However, even at the highest rates of glucose concentration change, MARD values below 10% were found for arm-placed sensors and below 10.5% for abdomen-placed sensors, Dr. Garg and colleagues report.
When the hypoglycemia threshold alert was set to 55 mg/dL, true alert rates for detection of hypoglycemia below 70 mg/dL by sensors worn on the arm and abdomen were 91.3% and 85.2%, respectively. With hyperglycemia threshold alerts set to 300 mg/dL, the true alert rates for detection of hyperglycemia greater than 250 mg/dL by sensors worn on the arm and abdomen were 99.9% and 99.8% respectively.
The overall mean time lag for the sensors was 3.5 minutes, 3.6 minutes for the arm, and 3.4 minutes for the abdomen. There were no serious adverse events during the study.
The study excluded children and adolescents; data from these populations will be reported separately, the authors note.
Accuracy at least as good as prior Dexcom versions, competitors
The MARD values of 8.2% on the arm and 9.1% on the abdomen were similar to or better than accuracy measurements of other commercially available CGM systems, note Dr. Garg and colleagues, although they acknowledge that few head-to-head studies at different anatomic locations have been conducted.
A study of an older Dexcom version (G4 Platinum) found MARD estimates of 12.0% on the arm and 12.3% on the abdomen, the authors note.
The newly FDA-approved implantable Eversense E3 (Senseonics) CGM, worn on the upper arm, has a MARD of 9.1%, while the arm-placed Abbott FreeStyle Libre 2, approved in the U.S. in June 2020, has an overall MARD of 9.3%.
Lag-time differences between the reference and G7 were also similar to or better than time delays in prior-generation Dexcom CGMs, Dr. Garg and colleagues say.
Participants also completed a survey. “The redesigned applicator allows for sensor deployment with one hand, and most participants found G7 easier to insert than their prior CGM system,” the researchers say.
Finally, “unlike G6, G7 allows for temporary silencing of all audible alerts, including Urgent Low. Taken together, these attributes are anticipated to provide for a better end-user experience with G7 and help reduce diabetes burden,” they conclude.
The study was supported by a grant from Dexcom. Dr. Garg has reported receiving consultant fees from Medtronic, Novo Nordisk, Zealand, LifeScan, Roche, and Lilly, as well as research grants through the University of Colorado from Lilly, Novo Nordisk, Medtronic, Dexcom, T1D Exchange, Helmsley Trust, NIDDK, and JDRF.
A version of this article first appeared on Medscape.com.
The Dexcom G7 continuous glucose monitor (CGM) is as accurate or better than other currently marketed CGM devices for measuring glucose in those with diabetes, new data from a pivotal study suggest.
Currently under review by the U.S. Food and Drug Administration, the G7 is expected to be an improvement over the Dexcom G6 version in several ways.
The on-body size will be 60% smaller, “roughly the size of three stacked quarters,” according to the authors, and will incorporate the sensor with a single-use transmitter, as opposed to the current separate 3-month transmitter used with the G6 sensor. This will eliminate the need for using a transmitter across multiple sensor sessions (as is the case for G6).
The warm-up period after insertion is reduced from 2 hours to 27 minutes, and users are given an extra 12-hour “grace period” after the 10-day wear period to change the device before it stops displaying glucose data. Up to 24 hours of missed data can also be recaptured.
“The enhanced features of G7 may increase clinical adoption, encourage sustained use, and reduce the burden of diabetes management,” write Satish K. Garg, MD, of the University of Colorado, Aurora, and colleagues, in their article, published online Feb. 14 in Diabetes Technology & Therapeutics.
Several features of the G6 remain unchanged, including factory calibration, but also the capacity for optional user calibrations, use of Bluetooth to transmit data up to 20 feet (approximately 6 meters), and data displays on either a dedicated receiver or a variety of iOS and Android smart devices.
It will also allow for user-customized settings and alerts, as well as the option for real-time remote “share” monitoring with caregivers or providers. The G7 will also not be susceptible to interference by acetaminophen (paracetamol) or ascorbic acid.
And, like the G6, the G7 was submitted for approval to the FDA as an “integrated CGM,” meaning that it will be interoperable with other compatible devices, including insulin pumps, glucose meters, or other electronic devices used for diabetes management.
Accuracy shown on abdomen, arm
The prospective, multicenter, single-arm study reported by Dr. Garg and colleagues was conducted at 12 U.S. sites between February and June 2021.
In-clinic visits were conducted on days 1 or 2, 4 or 7, and one additional day for comparisons with a reference glucose measure (YSI 2300 Stat Plus glucose analyzer). Participants wore blinded G7 sensors concurrently on the upper arm and abdomen while continuing to use their own personal glucose monitoring method (CGM or fingerstick) at home.
A total of 316 insulin-using adults with type 1 or type 2 diabetes contributed data from 308 arm- and 311 abdomen-placed blinded devices, which generated 77,774 matched pairs of data within the blood glucose reportable range of 40-400 mg/dL.
The overall mean absolute relative difference (MARD) of each CGM-YSI pair (a standard metric for CGM analysis) was 8.2%, with 9.1% for the abdomen and 8.2% for the arm.
Accuracy remained high in both arm- and abdomen-placed sensors across the 10-day wear period through the 12-hour grace period and across glucose ranges. There were no significant differences between G7 accuracy by diabetes type, insulin regimen, or body mass index.
The highest agreement rates and lowest MARDs occurred when CGM readings were increasing or decreasing by no more than 1 mg/dL per minute. However, even at the highest rates of glucose concentration change, MARD values below 10% were found for arm-placed sensors and below 10.5% for abdomen-placed sensors, Dr. Garg and colleagues report.
When the hypoglycemia threshold alert was set to 55 mg/dL, true alert rates for detection of hypoglycemia below 70 mg/dL by sensors worn on the arm and abdomen were 91.3% and 85.2%, respectively. With hyperglycemia threshold alerts set to 300 mg/dL, the true alert rates for detection of hyperglycemia greater than 250 mg/dL by sensors worn on the arm and abdomen were 99.9% and 99.8% respectively.
The overall mean time lag for the sensors was 3.5 minutes, 3.6 minutes for the arm, and 3.4 minutes for the abdomen. There were no serious adverse events during the study.
The study excluded children and adolescents; data from these populations will be reported separately, the authors note.
Accuracy at least as good as prior Dexcom versions, competitors
The MARD values of 8.2% on the arm and 9.1% on the abdomen were similar to or better than accuracy measurements of other commercially available CGM systems, note Dr. Garg and colleagues, although they acknowledge that few head-to-head studies at different anatomic locations have been conducted.
A study of an older Dexcom version (G4 Platinum) found MARD estimates of 12.0% on the arm and 12.3% on the abdomen, the authors note.
The newly FDA-approved implantable Eversense E3 (Senseonics) CGM, worn on the upper arm, has a MARD of 9.1%, while the arm-placed Abbott FreeStyle Libre 2, approved in the U.S. in June 2020, has an overall MARD of 9.3%.
Lag-time differences between the reference and G7 were also similar to or better than time delays in prior-generation Dexcom CGMs, Dr. Garg and colleagues say.
Participants also completed a survey. “The redesigned applicator allows for sensor deployment with one hand, and most participants found G7 easier to insert than their prior CGM system,” the researchers say.
Finally, “unlike G6, G7 allows for temporary silencing of all audible alerts, including Urgent Low. Taken together, these attributes are anticipated to provide for a better end-user experience with G7 and help reduce diabetes burden,” they conclude.
The study was supported by a grant from Dexcom. Dr. Garg has reported receiving consultant fees from Medtronic, Novo Nordisk, Zealand, LifeScan, Roche, and Lilly, as well as research grants through the University of Colorado from Lilly, Novo Nordisk, Medtronic, Dexcom, T1D Exchange, Helmsley Trust, NIDDK, and JDRF.
A version of this article first appeared on Medscape.com.
The Dexcom G7 continuous glucose monitor (CGM) is as accurate or better than other currently marketed CGM devices for measuring glucose in those with diabetes, new data from a pivotal study suggest.
Currently under review by the U.S. Food and Drug Administration, the G7 is expected to be an improvement over the Dexcom G6 version in several ways.
The on-body size will be 60% smaller, “roughly the size of three stacked quarters,” according to the authors, and will incorporate the sensor with a single-use transmitter, as opposed to the current separate 3-month transmitter used with the G6 sensor. This will eliminate the need for using a transmitter across multiple sensor sessions (as is the case for G6).
The warm-up period after insertion is reduced from 2 hours to 27 minutes, and users are given an extra 12-hour “grace period” after the 10-day wear period to change the device before it stops displaying glucose data. Up to 24 hours of missed data can also be recaptured.
“The enhanced features of G7 may increase clinical adoption, encourage sustained use, and reduce the burden of diabetes management,” write Satish K. Garg, MD, of the University of Colorado, Aurora, and colleagues, in their article, published online Feb. 14 in Diabetes Technology & Therapeutics.
Several features of the G6 remain unchanged, including factory calibration, but also the capacity for optional user calibrations, use of Bluetooth to transmit data up to 20 feet (approximately 6 meters), and data displays on either a dedicated receiver or a variety of iOS and Android smart devices.
It will also allow for user-customized settings and alerts, as well as the option for real-time remote “share” monitoring with caregivers or providers. The G7 will also not be susceptible to interference by acetaminophen (paracetamol) or ascorbic acid.
And, like the G6, the G7 was submitted for approval to the FDA as an “integrated CGM,” meaning that it will be interoperable with other compatible devices, including insulin pumps, glucose meters, or other electronic devices used for diabetes management.
Accuracy shown on abdomen, arm
The prospective, multicenter, single-arm study reported by Dr. Garg and colleagues was conducted at 12 U.S. sites between February and June 2021.
In-clinic visits were conducted on days 1 or 2, 4 or 7, and one additional day for comparisons with a reference glucose measure (YSI 2300 Stat Plus glucose analyzer). Participants wore blinded G7 sensors concurrently on the upper arm and abdomen while continuing to use their own personal glucose monitoring method (CGM or fingerstick) at home.
A total of 316 insulin-using adults with type 1 or type 2 diabetes contributed data from 308 arm- and 311 abdomen-placed blinded devices, which generated 77,774 matched pairs of data within the blood glucose reportable range of 40-400 mg/dL.
The overall mean absolute relative difference (MARD) of each CGM-YSI pair (a standard metric for CGM analysis) was 8.2%, with 9.1% for the abdomen and 8.2% for the arm.
Accuracy remained high in both arm- and abdomen-placed sensors across the 10-day wear period through the 12-hour grace period and across glucose ranges. There were no significant differences between G7 accuracy by diabetes type, insulin regimen, or body mass index.
The highest agreement rates and lowest MARDs occurred when CGM readings were increasing or decreasing by no more than 1 mg/dL per minute. However, even at the highest rates of glucose concentration change, MARD values below 10% were found for arm-placed sensors and below 10.5% for abdomen-placed sensors, Dr. Garg and colleagues report.
When the hypoglycemia threshold alert was set to 55 mg/dL, true alert rates for detection of hypoglycemia below 70 mg/dL by sensors worn on the arm and abdomen were 91.3% and 85.2%, respectively. With hyperglycemia threshold alerts set to 300 mg/dL, the true alert rates for detection of hyperglycemia greater than 250 mg/dL by sensors worn on the arm and abdomen were 99.9% and 99.8% respectively.
The overall mean time lag for the sensors was 3.5 minutes, 3.6 minutes for the arm, and 3.4 minutes for the abdomen. There were no serious adverse events during the study.
The study excluded children and adolescents; data from these populations will be reported separately, the authors note.
Accuracy at least as good as prior Dexcom versions, competitors
The MARD values of 8.2% on the arm and 9.1% on the abdomen were similar to or better than accuracy measurements of other commercially available CGM systems, note Dr. Garg and colleagues, although they acknowledge that few head-to-head studies at different anatomic locations have been conducted.
A study of an older Dexcom version (G4 Platinum) found MARD estimates of 12.0% on the arm and 12.3% on the abdomen, the authors note.
The newly FDA-approved implantable Eversense E3 (Senseonics) CGM, worn on the upper arm, has a MARD of 9.1%, while the arm-placed Abbott FreeStyle Libre 2, approved in the U.S. in June 2020, has an overall MARD of 9.3%.
Lag-time differences between the reference and G7 were also similar to or better than time delays in prior-generation Dexcom CGMs, Dr. Garg and colleagues say.
Participants also completed a survey. “The redesigned applicator allows for sensor deployment with one hand, and most participants found G7 easier to insert than their prior CGM system,” the researchers say.
Finally, “unlike G6, G7 allows for temporary silencing of all audible alerts, including Urgent Low. Taken together, these attributes are anticipated to provide for a better end-user experience with G7 and help reduce diabetes burden,” they conclude.
The study was supported by a grant from Dexcom. Dr. Garg has reported receiving consultant fees from Medtronic, Novo Nordisk, Zealand, LifeScan, Roche, and Lilly, as well as research grants through the University of Colorado from Lilly, Novo Nordisk, Medtronic, Dexcom, T1D Exchange, Helmsley Trust, NIDDK, and JDRF.
A version of this article first appeared on Medscape.com.
Mask mandates ending in all but one state
As COVID-19 cases and hospitalizations continue to decline across the United States,
Retailers and cruises are following along, with Apple and Target stores lifting their own mask mandates this week. Cruise lines such as Norwegian and Royal Caribbean International have said mask requirements will be relaxed for vaccinated passengers, according to the Washington Post.
But guidance from the Centers for Disease Control and Prevention hasn’t changed even as the Omicron variant recedes across the country. Vaccinated people should wear masks when indoors in areas of “substantial or high transmission,” which still covers more than 95% of the country, according to a CDC map.
As daily cases continue to fall, the CDC is reviewing its recommendations, Rochelle Walensky, MD, the CDC director, said during a briefing last week.
“We want to give people a break from things like mask-wearing, when these metrics are better, and then have the ability to reach for them again should things worsen,” she said.
As states relax mask rules, county and city officials are now deciding what to do in their jurisdictions. Vaccinated residents in Los Angeles County may soon be able to go maskless in indoor settings that check for proof of vaccination, according to the Los Angeles Times.
Chicago will also end its mask and COVID-19 vaccine mandates for public places such as restaurants Feb. 28, according to the Chicago Tribune. Illinois will end a statewide indoor mask mandate on the same day. Masks will still be required in health care settings and public transmit.
State and local school boards are debating their mask policies as well. The Maryland State Board of Education voted Feb. 22 to allow local school districts to decide whether students must wear face coverings in school, according to the Associated Press. The update will take effect on March 1 if approved by a Maryland General Assembly committee that oversees the rule.
In New York, state officials have begun lifting mask rules. At the same time, 58% of New York voters want to see early March data before school mask mandates are ended, according to a new poll, released Feb. 22 by the Siena College Research Institute. About 45% of those polled said the state’s indoor public mask mandate should also still be in place.
The debate about wearing masks in schools will likely continue, especially as districts get caught between health authorities and parents, according to the Wall Street Journal. District officials in several states are receiving hundreds of emails daily from both sides, with parents calling for mask rules to end or saying that requirements should remain in place for now to keep kids safe.
A version of this article first appeared on WebMD.com.
As COVID-19 cases and hospitalizations continue to decline across the United States,
Retailers and cruises are following along, with Apple and Target stores lifting their own mask mandates this week. Cruise lines such as Norwegian and Royal Caribbean International have said mask requirements will be relaxed for vaccinated passengers, according to the Washington Post.
But guidance from the Centers for Disease Control and Prevention hasn’t changed even as the Omicron variant recedes across the country. Vaccinated people should wear masks when indoors in areas of “substantial or high transmission,” which still covers more than 95% of the country, according to a CDC map.
As daily cases continue to fall, the CDC is reviewing its recommendations, Rochelle Walensky, MD, the CDC director, said during a briefing last week.
“We want to give people a break from things like mask-wearing, when these metrics are better, and then have the ability to reach for them again should things worsen,” she said.
As states relax mask rules, county and city officials are now deciding what to do in their jurisdictions. Vaccinated residents in Los Angeles County may soon be able to go maskless in indoor settings that check for proof of vaccination, according to the Los Angeles Times.
Chicago will also end its mask and COVID-19 vaccine mandates for public places such as restaurants Feb. 28, according to the Chicago Tribune. Illinois will end a statewide indoor mask mandate on the same day. Masks will still be required in health care settings and public transmit.
State and local school boards are debating their mask policies as well. The Maryland State Board of Education voted Feb. 22 to allow local school districts to decide whether students must wear face coverings in school, according to the Associated Press. The update will take effect on March 1 if approved by a Maryland General Assembly committee that oversees the rule.
In New York, state officials have begun lifting mask rules. At the same time, 58% of New York voters want to see early March data before school mask mandates are ended, according to a new poll, released Feb. 22 by the Siena College Research Institute. About 45% of those polled said the state’s indoor public mask mandate should also still be in place.
The debate about wearing masks in schools will likely continue, especially as districts get caught between health authorities and parents, according to the Wall Street Journal. District officials in several states are receiving hundreds of emails daily from both sides, with parents calling for mask rules to end or saying that requirements should remain in place for now to keep kids safe.
A version of this article first appeared on WebMD.com.
As COVID-19 cases and hospitalizations continue to decline across the United States,
Retailers and cruises are following along, with Apple and Target stores lifting their own mask mandates this week. Cruise lines such as Norwegian and Royal Caribbean International have said mask requirements will be relaxed for vaccinated passengers, according to the Washington Post.
But guidance from the Centers for Disease Control and Prevention hasn’t changed even as the Omicron variant recedes across the country. Vaccinated people should wear masks when indoors in areas of “substantial or high transmission,” which still covers more than 95% of the country, according to a CDC map.
As daily cases continue to fall, the CDC is reviewing its recommendations, Rochelle Walensky, MD, the CDC director, said during a briefing last week.
“We want to give people a break from things like mask-wearing, when these metrics are better, and then have the ability to reach for them again should things worsen,” she said.
As states relax mask rules, county and city officials are now deciding what to do in their jurisdictions. Vaccinated residents in Los Angeles County may soon be able to go maskless in indoor settings that check for proof of vaccination, according to the Los Angeles Times.
Chicago will also end its mask and COVID-19 vaccine mandates for public places such as restaurants Feb. 28, according to the Chicago Tribune. Illinois will end a statewide indoor mask mandate on the same day. Masks will still be required in health care settings and public transmit.
State and local school boards are debating their mask policies as well. The Maryland State Board of Education voted Feb. 22 to allow local school districts to decide whether students must wear face coverings in school, according to the Associated Press. The update will take effect on March 1 if approved by a Maryland General Assembly committee that oversees the rule.
In New York, state officials have begun lifting mask rules. At the same time, 58% of New York voters want to see early March data before school mask mandates are ended, according to a new poll, released Feb. 22 by the Siena College Research Institute. About 45% of those polled said the state’s indoor public mask mandate should also still be in place.
The debate about wearing masks in schools will likely continue, especially as districts get caught between health authorities and parents, according to the Wall Street Journal. District officials in several states are receiving hundreds of emails daily from both sides, with parents calling for mask rules to end or saying that requirements should remain in place for now to keep kids safe.
A version of this article first appeared on WebMD.com.
Toenail ridges
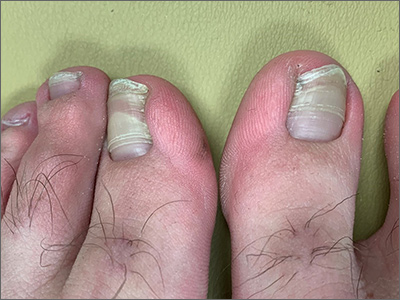
Transverse ridges that grow out with the nails are called Beau lines, also known as Beau’s ridges. This contrasts with Mees lines which are transverse white bands that grow out with the toenails, are nonpalpable, and are attributed to arsenic poisoning.
Beau lines are caused by a disruption in nail growth that can result from trauma, hypotension, or systemic or severe illness; they have also been reported in cases of COVID-19.1 Beau lines can occur on a single nail if the trauma or injury is isolated to 1 digit. If there was a systemic illness or stress, the lines can affect all 20 nails. The time of the inciting event can be approximated by how far the lines are from the cuticle. While there is some variability, it usually takes 12 to 18 months to grow an entirely new toenail. If the Beau lines have grown halfway out, then the stressor likely occurred 6 to 9 months earlier.
In this image, some asymmetry is visible between the right and left great toenails and there are some subtle distal changes, raising the possibility that there was more than 1 injury to this patient’s system (or prolonged difficulty). The patient said that to his knowledge, he had not been infected with COVID-19. However, hair and nail changes may be the only finding in some individuals who have been infected with COVID-19.1
This patient was counseled regarding the nature of this disorder and that without knowing what illness or injury caused the change, it was a benign finding. He was advised that it did not appear to be onychomycosis and did not require any medications or antifungal therapy. The patient was told to follow up if any changes developed.
Image courtesy of Daniel Stulberg, MD. Text courtesy of Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.
- Deng J, Ngo T, Zhu TH, Halverstam C. Telogen effluvium, Beau lines, and acral peeling associated with COVID-19 infection. JAAD Case Rep. 2021;13:138-140. doi: 10.1016/j.jdcr.2021.05.026

Transverse ridges that grow out with the nails are called Beau lines, also known as Beau’s ridges. This contrasts with Mees lines which are transverse white bands that grow out with the toenails, are nonpalpable, and are attributed to arsenic poisoning.
Beau lines are caused by a disruption in nail growth that can result from trauma, hypotension, or systemic or severe illness; they have also been reported in cases of COVID-19.1 Beau lines can occur on a single nail if the trauma or injury is isolated to 1 digit. If there was a systemic illness or stress, the lines can affect all 20 nails. The time of the inciting event can be approximated by how far the lines are from the cuticle. While there is some variability, it usually takes 12 to 18 months to grow an entirely new toenail. If the Beau lines have grown halfway out, then the stressor likely occurred 6 to 9 months earlier.
In this image, some asymmetry is visible between the right and left great toenails and there are some subtle distal changes, raising the possibility that there was more than 1 injury to this patient’s system (or prolonged difficulty). The patient said that to his knowledge, he had not been infected with COVID-19. However, hair and nail changes may be the only finding in some individuals who have been infected with COVID-19.1
This patient was counseled regarding the nature of this disorder and that without knowing what illness or injury caused the change, it was a benign finding. He was advised that it did not appear to be onychomycosis and did not require any medications or antifungal therapy. The patient was told to follow up if any changes developed.
Image courtesy of Daniel Stulberg, MD. Text courtesy of Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.

Transverse ridges that grow out with the nails are called Beau lines, also known as Beau’s ridges. This contrasts with Mees lines which are transverse white bands that grow out with the toenails, are nonpalpable, and are attributed to arsenic poisoning.
Beau lines are caused by a disruption in nail growth that can result from trauma, hypotension, or systemic or severe illness; they have also been reported in cases of COVID-19.1 Beau lines can occur on a single nail if the trauma or injury is isolated to 1 digit. If there was a systemic illness or stress, the lines can affect all 20 nails. The time of the inciting event can be approximated by how far the lines are from the cuticle. While there is some variability, it usually takes 12 to 18 months to grow an entirely new toenail. If the Beau lines have grown halfway out, then the stressor likely occurred 6 to 9 months earlier.
In this image, some asymmetry is visible between the right and left great toenails and there are some subtle distal changes, raising the possibility that there was more than 1 injury to this patient’s system (or prolonged difficulty). The patient said that to his knowledge, he had not been infected with COVID-19. However, hair and nail changes may be the only finding in some individuals who have been infected with COVID-19.1
This patient was counseled regarding the nature of this disorder and that without knowing what illness or injury caused the change, it was a benign finding. He was advised that it did not appear to be onychomycosis and did not require any medications or antifungal therapy. The patient was told to follow up if any changes developed.
Image courtesy of Daniel Stulberg, MD. Text courtesy of Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.
- Deng J, Ngo T, Zhu TH, Halverstam C. Telogen effluvium, Beau lines, and acral peeling associated with COVID-19 infection. JAAD Case Rep. 2021;13:138-140. doi: 10.1016/j.jdcr.2021.05.026
- Deng J, Ngo T, Zhu TH, Halverstam C. Telogen effluvium, Beau lines, and acral peeling associated with COVID-19 infection. JAAD Case Rep. 2021;13:138-140. doi: 10.1016/j.jdcr.2021.05.026