User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
nav[contains(@class, 'nav-ce-stack nav-ce-stack__large-screen')]
header[@id='header']
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'main-prefix')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
div[contains(@class, 'view-medstat-quiz-listing-panes')]
div[contains(@class, 'pane-article-sidebar-latest-news')]
Antibody mix may prevent COVID symptoms in some asymptomatic people
over 28 days, new research shows.
Results of the study by Meagan P. O’Brien, MD, from Regeneron Pharmaceuticals and one of the study’s funders, and coauthors were published online Jan. 14, 2022, in an original investigation in JAMA.
The results suggest new potential for monoclonal antibodies currently used for postexposure prophylaxis and treatment of symptomatic SARS-CoV-2. It has not been clear whether monoclonal antibodies can benefit people with asymptomatic SARS-CoV-2 infection.
The trial included 314 participants (mean age, 41 years; 51.6% women). Of the participants, 310 (99.7%) completed the efficacy assessment period, and 204 were asymptomatic and tested negative at baseline and were included in the primary efficacy analysis.
The subcutaneous combination of casirivimab and imdevimab, 1,200 mg (600 mg each), significantly prevented progression to symptomatic disease (29/100 [29.0%] vs. 44/104 [42.3%] with placebo; odds ratio, 0.54 [95% confidence interval, 0.30-0.97]; P = .04; absolute risk difference, −13.3% [95% CI, −26.3% to −0.3%]).
These results were part of a randomized, double-blind, placebo-controlled, phase 3 trial of close household contacts of a SARS-CoV-2–infected person at 112 sites in the United States, Romania, and Moldova. They were enrolled between July 13, 2020, and Jan. 28, 2021; follow-up ended March 11, 2021.
Asymptomatic people at least 12 years old were eligible if identified within 96 hours of index case positive test collection and were randomly assigned 1:1 to receive one dose of subcutaneous casirivimab and imdevimab (n = 158), or placebo (n = 156).
COVID-19 vaccination was prohibited before enrollment but was allowed after completing the 28-day efficacy assessment period.
Caution warranted
In an accompanying editorial, however, Jonathan Z. Li, MD, Brigham and Women’s Hospital and Harvard Medical School, both in Boston, and Rajesh T. Gandhi, MD, Massachusetts General Hospital, Boston, and Harvard Medical School, urged caution in interpreting the results.
They wrote that, although monoclonal antibodies are generally used in individuals at high risk for severe COVID-19, this study population was less vulnerable, with an average age of 41, and 30% had no risk for the disease.
“Of the remainder, the most common risk factor was being overweight (which confers less risk than other factors),” the editorialists wrote.
They pointed out, as did the study authors, that enrollment came before the emergence of the Delta and Omicron variants, and that both casirivimab and imdevimab maintain their activity against Delta but not against Omicron.
“While prevention of symptomatic infection has benefits,” they wrote, “the primary goal of monoclonal antibody therapy is to prevent progression to severe disease; however, this trial was unable to assess this outcome because there were only three hospitalizations (all in the placebo group). Also, this study was conducted prior to widespread COVID-19 vaccination; whether monoclonal antibodies have the same benefit in people who have breakthrough infection after vaccination is not known.”
The editorialists highlighted the subcutaneous delivery in this study.
They wrote that Dr. O’Brien and coauthors provide evidence that subcutaneous administration is effective in infected individuals. “However, high serum monoclonal antibody levels are achieved more quickly after intravenous administration than following subcutaneous injection; it is unknown whether intravenous administration might have led to even greater efficacy for individuals with asymptomatic SARS-CoV-2 infection.”
The authors of the study also add that, despite efforts to recruit non-White participants, relatively few non-White people were enrolled. Additionally, few adolescents were enrolled.
The sample size was also relatively small, they acknowledge, because of a study design in which the infection status of asymptomatic participants was not confirmed at inclusion.
Several of the authors are employees/stockholders of Regeneron, and have a patent pending, which has been licensed and is receiving royalties. The study was supported by Regeneron and F. Hoffmann–La Roche. This trial was conducted jointly with the National Institute of Allergy and Infectious Diseases and the National Institutes of Health. The CoVPN (COVID-19 Prevention Network) is supported by cooperative agreement awards from the NIAID and NIH.
A version of this article first appeared on Medscape.com.
over 28 days, new research shows.
Results of the study by Meagan P. O’Brien, MD, from Regeneron Pharmaceuticals and one of the study’s funders, and coauthors were published online Jan. 14, 2022, in an original investigation in JAMA.
The results suggest new potential for monoclonal antibodies currently used for postexposure prophylaxis and treatment of symptomatic SARS-CoV-2. It has not been clear whether monoclonal antibodies can benefit people with asymptomatic SARS-CoV-2 infection.
The trial included 314 participants (mean age, 41 years; 51.6% women). Of the participants, 310 (99.7%) completed the efficacy assessment period, and 204 were asymptomatic and tested negative at baseline and were included in the primary efficacy analysis.
The subcutaneous combination of casirivimab and imdevimab, 1,200 mg (600 mg each), significantly prevented progression to symptomatic disease (29/100 [29.0%] vs. 44/104 [42.3%] with placebo; odds ratio, 0.54 [95% confidence interval, 0.30-0.97]; P = .04; absolute risk difference, −13.3% [95% CI, −26.3% to −0.3%]).
These results were part of a randomized, double-blind, placebo-controlled, phase 3 trial of close household contacts of a SARS-CoV-2–infected person at 112 sites in the United States, Romania, and Moldova. They were enrolled between July 13, 2020, and Jan. 28, 2021; follow-up ended March 11, 2021.
Asymptomatic people at least 12 years old were eligible if identified within 96 hours of index case positive test collection and were randomly assigned 1:1 to receive one dose of subcutaneous casirivimab and imdevimab (n = 158), or placebo (n = 156).
COVID-19 vaccination was prohibited before enrollment but was allowed after completing the 28-day efficacy assessment period.
Caution warranted
In an accompanying editorial, however, Jonathan Z. Li, MD, Brigham and Women’s Hospital and Harvard Medical School, both in Boston, and Rajesh T. Gandhi, MD, Massachusetts General Hospital, Boston, and Harvard Medical School, urged caution in interpreting the results.
They wrote that, although monoclonal antibodies are generally used in individuals at high risk for severe COVID-19, this study population was less vulnerable, with an average age of 41, and 30% had no risk for the disease.
“Of the remainder, the most common risk factor was being overweight (which confers less risk than other factors),” the editorialists wrote.
They pointed out, as did the study authors, that enrollment came before the emergence of the Delta and Omicron variants, and that both casirivimab and imdevimab maintain their activity against Delta but not against Omicron.
“While prevention of symptomatic infection has benefits,” they wrote, “the primary goal of monoclonal antibody therapy is to prevent progression to severe disease; however, this trial was unable to assess this outcome because there were only three hospitalizations (all in the placebo group). Also, this study was conducted prior to widespread COVID-19 vaccination; whether monoclonal antibodies have the same benefit in people who have breakthrough infection after vaccination is not known.”
The editorialists highlighted the subcutaneous delivery in this study.
They wrote that Dr. O’Brien and coauthors provide evidence that subcutaneous administration is effective in infected individuals. “However, high serum monoclonal antibody levels are achieved more quickly after intravenous administration than following subcutaneous injection; it is unknown whether intravenous administration might have led to even greater efficacy for individuals with asymptomatic SARS-CoV-2 infection.”
The authors of the study also add that, despite efforts to recruit non-White participants, relatively few non-White people were enrolled. Additionally, few adolescents were enrolled.
The sample size was also relatively small, they acknowledge, because of a study design in which the infection status of asymptomatic participants was not confirmed at inclusion.
Several of the authors are employees/stockholders of Regeneron, and have a patent pending, which has been licensed and is receiving royalties. The study was supported by Regeneron and F. Hoffmann–La Roche. This trial was conducted jointly with the National Institute of Allergy and Infectious Diseases and the National Institutes of Health. The CoVPN (COVID-19 Prevention Network) is supported by cooperative agreement awards from the NIAID and NIH.
A version of this article first appeared on Medscape.com.
over 28 days, new research shows.
Results of the study by Meagan P. O’Brien, MD, from Regeneron Pharmaceuticals and one of the study’s funders, and coauthors were published online Jan. 14, 2022, in an original investigation in JAMA.
The results suggest new potential for monoclonal antibodies currently used for postexposure prophylaxis and treatment of symptomatic SARS-CoV-2. It has not been clear whether monoclonal antibodies can benefit people with asymptomatic SARS-CoV-2 infection.
The trial included 314 participants (mean age, 41 years; 51.6% women). Of the participants, 310 (99.7%) completed the efficacy assessment period, and 204 were asymptomatic and tested negative at baseline and were included in the primary efficacy analysis.
The subcutaneous combination of casirivimab and imdevimab, 1,200 mg (600 mg each), significantly prevented progression to symptomatic disease (29/100 [29.0%] vs. 44/104 [42.3%] with placebo; odds ratio, 0.54 [95% confidence interval, 0.30-0.97]; P = .04; absolute risk difference, −13.3% [95% CI, −26.3% to −0.3%]).
These results were part of a randomized, double-blind, placebo-controlled, phase 3 trial of close household contacts of a SARS-CoV-2–infected person at 112 sites in the United States, Romania, and Moldova. They were enrolled between July 13, 2020, and Jan. 28, 2021; follow-up ended March 11, 2021.
Asymptomatic people at least 12 years old were eligible if identified within 96 hours of index case positive test collection and were randomly assigned 1:1 to receive one dose of subcutaneous casirivimab and imdevimab (n = 158), or placebo (n = 156).
COVID-19 vaccination was prohibited before enrollment but was allowed after completing the 28-day efficacy assessment period.
Caution warranted
In an accompanying editorial, however, Jonathan Z. Li, MD, Brigham and Women’s Hospital and Harvard Medical School, both in Boston, and Rajesh T. Gandhi, MD, Massachusetts General Hospital, Boston, and Harvard Medical School, urged caution in interpreting the results.
They wrote that, although monoclonal antibodies are generally used in individuals at high risk for severe COVID-19, this study population was less vulnerable, with an average age of 41, and 30% had no risk for the disease.
“Of the remainder, the most common risk factor was being overweight (which confers less risk than other factors),” the editorialists wrote.
They pointed out, as did the study authors, that enrollment came before the emergence of the Delta and Omicron variants, and that both casirivimab and imdevimab maintain their activity against Delta but not against Omicron.
“While prevention of symptomatic infection has benefits,” they wrote, “the primary goal of monoclonal antibody therapy is to prevent progression to severe disease; however, this trial was unable to assess this outcome because there were only three hospitalizations (all in the placebo group). Also, this study was conducted prior to widespread COVID-19 vaccination; whether monoclonal antibodies have the same benefit in people who have breakthrough infection after vaccination is not known.”
The editorialists highlighted the subcutaneous delivery in this study.
They wrote that Dr. O’Brien and coauthors provide evidence that subcutaneous administration is effective in infected individuals. “However, high serum monoclonal antibody levels are achieved more quickly after intravenous administration than following subcutaneous injection; it is unknown whether intravenous administration might have led to even greater efficacy for individuals with asymptomatic SARS-CoV-2 infection.”
The authors of the study also add that, despite efforts to recruit non-White participants, relatively few non-White people were enrolled. Additionally, few adolescents were enrolled.
The sample size was also relatively small, they acknowledge, because of a study design in which the infection status of asymptomatic participants was not confirmed at inclusion.
Several of the authors are employees/stockholders of Regeneron, and have a patent pending, which has been licensed and is receiving royalties. The study was supported by Regeneron and F. Hoffmann–La Roche. This trial was conducted jointly with the National Institute of Allergy and Infectious Diseases and the National Institutes of Health. The CoVPN (COVID-19 Prevention Network) is supported by cooperative agreement awards from the NIAID and NIH.
A version of this article first appeared on Medscape.com.
FROM JAMA
Chronic respiratory conditions occur more often in RSV vs. flu
Hospitalized intensive care patients with respiratory syncytial virus were significantly more likely to be immunocompromised and to have chronic respiratory conditions than those with influenza infections, but in-hospital mortality rates were similar, based on data from 618 adults.
Respiratory syncytial virus is common in adults, but characteristics of RSV patients requiring ICU care have not been explored, despite routine testing for RSV in critically ill patients in many institutions, Julien Coussement, PhD, of Université Libre de Bruxelles, Brussels, and colleagues wrote.
“Influenza is another respiratory virus routinely tested for in ICU patients with respiratory symptoms because of its well-known morbidity and mortality, but there are no data specifically comparing RSV and influenza infections in adult ICU patients,” they noted.
In a retrospective, multicenter study published in the journal CHEST, the researchers analyzed data from 309 adult ICU patients with RSV infection and 309 with influenza infection between November 2011 and April 2018 from 17 sites in France and Belgium. Each RSV patient was matched to a flu patient according to institution and date of diagnosis.
The primary objective was a comparison of in-hospital mortality between the groups, defined as death from any cause during an index hospital stay in acute care. Secondary objectives were comparisons of the clinical and biological characteristics of patients with RSV versus flu.
Overall, in-hospital mortality was not significantly different between the RSV and influenza groups (23.9% vs. 25.6%, P = .63).
However, patients with RSV infection were significantly more likely than those with flu to have an underlying chronic respiratory condition (60.2% vs. 40.1%, P < .001) and to be immunocompromised (35% vs. 26.2%, P = .02). Very few of the patients overall (39 patients, 6.3%) were considered young and healthy prior to hospitalization; and significantly fewer of these were in the RSV group than in the influenza group (9 patients and 30 patients, respectively).
Airway obstruction at the time of diagnosis was significantly more common in the RSV patients than in influenza patients (49.5% vs. 39.5%, P = .01), but influenza patients were significantly more likely to have acute respiratory distress syndrome at the time of diagnosis (21.7% vs. 14.6%, P = .02). Rates of coinfections were similar between the groups, and approximately 60% of coinfected patients received at least 72 hours of therapeutic antibiotics. Overall length of hospital stay, ICU stay, and duration of mechanical ventilation were similar between the groups.
The results show that severe RSV occurs mainly in older patients with comorbidities, and these results reflect data from previous studies, the researchers wrote in their discussion. In addition, “patients with influenza infection were significantly more likely to have fever, myalgia, increased CPK level, thrombocytopenia and transaminitis at diagnosis than were those with RSV infection. Whether these differences may be used to guide patient management remains to be determined.”
The study findings were limited by several factors including the retrospective design, and testing for respiratory viruses on symptomatic patients only, rather than all ICU patients, the researchers noted. Other limitations include the inability to show a causal link between viral infections and patient outcomes and the heterogenous management of patients among different centers.
However, the results were strengthened by the large sample size and multivariate analysis, and support the need for interventions to prevent and treat severe RSV, they concluded.
The study received no outside funding. Lead author Dr. Coussement disclosed serving on advisory board for Sanofi.
Hospitalized intensive care patients with respiratory syncytial virus were significantly more likely to be immunocompromised and to have chronic respiratory conditions than those with influenza infections, but in-hospital mortality rates were similar, based on data from 618 adults.
Respiratory syncytial virus is common in adults, but characteristics of RSV patients requiring ICU care have not been explored, despite routine testing for RSV in critically ill patients in many institutions, Julien Coussement, PhD, of Université Libre de Bruxelles, Brussels, and colleagues wrote.
“Influenza is another respiratory virus routinely tested for in ICU patients with respiratory symptoms because of its well-known morbidity and mortality, but there are no data specifically comparing RSV and influenza infections in adult ICU patients,” they noted.
In a retrospective, multicenter study published in the journal CHEST, the researchers analyzed data from 309 adult ICU patients with RSV infection and 309 with influenza infection between November 2011 and April 2018 from 17 sites in France and Belgium. Each RSV patient was matched to a flu patient according to institution and date of diagnosis.
The primary objective was a comparison of in-hospital mortality between the groups, defined as death from any cause during an index hospital stay in acute care. Secondary objectives were comparisons of the clinical and biological characteristics of patients with RSV versus flu.
Overall, in-hospital mortality was not significantly different between the RSV and influenza groups (23.9% vs. 25.6%, P = .63).
However, patients with RSV infection were significantly more likely than those with flu to have an underlying chronic respiratory condition (60.2% vs. 40.1%, P < .001) and to be immunocompromised (35% vs. 26.2%, P = .02). Very few of the patients overall (39 patients, 6.3%) were considered young and healthy prior to hospitalization; and significantly fewer of these were in the RSV group than in the influenza group (9 patients and 30 patients, respectively).
Airway obstruction at the time of diagnosis was significantly more common in the RSV patients than in influenza patients (49.5% vs. 39.5%, P = .01), but influenza patients were significantly more likely to have acute respiratory distress syndrome at the time of diagnosis (21.7% vs. 14.6%, P = .02). Rates of coinfections were similar between the groups, and approximately 60% of coinfected patients received at least 72 hours of therapeutic antibiotics. Overall length of hospital stay, ICU stay, and duration of mechanical ventilation were similar between the groups.
The results show that severe RSV occurs mainly in older patients with comorbidities, and these results reflect data from previous studies, the researchers wrote in their discussion. In addition, “patients with influenza infection were significantly more likely to have fever, myalgia, increased CPK level, thrombocytopenia and transaminitis at diagnosis than were those with RSV infection. Whether these differences may be used to guide patient management remains to be determined.”
The study findings were limited by several factors including the retrospective design, and testing for respiratory viruses on symptomatic patients only, rather than all ICU patients, the researchers noted. Other limitations include the inability to show a causal link between viral infections and patient outcomes and the heterogenous management of patients among different centers.
However, the results were strengthened by the large sample size and multivariate analysis, and support the need for interventions to prevent and treat severe RSV, they concluded.
The study received no outside funding. Lead author Dr. Coussement disclosed serving on advisory board for Sanofi.
Hospitalized intensive care patients with respiratory syncytial virus were significantly more likely to be immunocompromised and to have chronic respiratory conditions than those with influenza infections, but in-hospital mortality rates were similar, based on data from 618 adults.
Respiratory syncytial virus is common in adults, but characteristics of RSV patients requiring ICU care have not been explored, despite routine testing for RSV in critically ill patients in many institutions, Julien Coussement, PhD, of Université Libre de Bruxelles, Brussels, and colleagues wrote.
“Influenza is another respiratory virus routinely tested for in ICU patients with respiratory symptoms because of its well-known morbidity and mortality, but there are no data specifically comparing RSV and influenza infections in adult ICU patients,” they noted.
In a retrospective, multicenter study published in the journal CHEST, the researchers analyzed data from 309 adult ICU patients with RSV infection and 309 with influenza infection between November 2011 and April 2018 from 17 sites in France and Belgium. Each RSV patient was matched to a flu patient according to institution and date of diagnosis.
The primary objective was a comparison of in-hospital mortality between the groups, defined as death from any cause during an index hospital stay in acute care. Secondary objectives were comparisons of the clinical and biological characteristics of patients with RSV versus flu.
Overall, in-hospital mortality was not significantly different between the RSV and influenza groups (23.9% vs. 25.6%, P = .63).
However, patients with RSV infection were significantly more likely than those with flu to have an underlying chronic respiratory condition (60.2% vs. 40.1%, P < .001) and to be immunocompromised (35% vs. 26.2%, P = .02). Very few of the patients overall (39 patients, 6.3%) were considered young and healthy prior to hospitalization; and significantly fewer of these were in the RSV group than in the influenza group (9 patients and 30 patients, respectively).
Airway obstruction at the time of diagnosis was significantly more common in the RSV patients than in influenza patients (49.5% vs. 39.5%, P = .01), but influenza patients were significantly more likely to have acute respiratory distress syndrome at the time of diagnosis (21.7% vs. 14.6%, P = .02). Rates of coinfections were similar between the groups, and approximately 60% of coinfected patients received at least 72 hours of therapeutic antibiotics. Overall length of hospital stay, ICU stay, and duration of mechanical ventilation were similar between the groups.
The results show that severe RSV occurs mainly in older patients with comorbidities, and these results reflect data from previous studies, the researchers wrote in their discussion. In addition, “patients with influenza infection were significantly more likely to have fever, myalgia, increased CPK level, thrombocytopenia and transaminitis at diagnosis than were those with RSV infection. Whether these differences may be used to guide patient management remains to be determined.”
The study findings were limited by several factors including the retrospective design, and testing for respiratory viruses on symptomatic patients only, rather than all ICU patients, the researchers noted. Other limitations include the inability to show a causal link between viral infections and patient outcomes and the heterogenous management of patients among different centers.
However, the results were strengthened by the large sample size and multivariate analysis, and support the need for interventions to prevent and treat severe RSV, they concluded.
The study received no outside funding. Lead author Dr. Coussement disclosed serving on advisory board for Sanofi.
FROM CHEST
‘Lucky genes’ may protect against some obesity-related diseases
in a large new genetics study.
That is, people with unfavorable adiposity gene variants had fat stored under the skin throughout the body, but they also had more ectopic fat (fat in the “wrong place”) surrounding the pancreas and liver, which is associated with a higher risk of metabolic diseases such as heart disease and type 2 diabetes.
In contrast, people with favorable adiposity gene variants had more subcutaneous fat (such as a paunch or a double chin).
The study by Susan Martin, PhD, a postdoctoral research associate at the University of Exeter (England) and colleagues, was recently published in eLife.
“Some people have ‘unlucky fat genes,’ meaning they store higher levels of fat everywhere, including under the skin [and around the] liver and pancreas. That’s associated with a higher risk of diseases such as type 2 diabetes,” senior author Hanieh Yaghootkar, MD, PhD, summarized in a press release from the University of Exeter.
“Others are luckier and have genes that mean higher fat under the skin but lower liver fat and a lower risk of diseases like type 2 diabetes,” added Dr. Yaghootkar, from Brunel University London.
Among 37 chronic diseases that are associated with obesity, the researchers found the metabolic effects of adiposity are likely the main cause of the following 11: type 2 diabetes, polycystic ovary syndrome, coronary artery disease, peripheral artery disease, hypertension, stroke, heart failure, atrial fibrillation, chronic kidney disease, renal cancer, and gout.
On the other hand, excess weight itself (such as a heavy load on the joints) rather than a metabolic effect is associated with nine other obesity-related diseases: osteoarthritis, rheumatoid arthritis, osteoporosis, gastro-esophageal reflux disease, gallstones, adult-onset asthma, psoriasis, deep vein thrombosis, and venous thromboembolism.
Good genes no substitute for a healthy lifestyle
“People with more favorable adiposity gene variants are still at risk of the nine diseases” that are not caused by metabolic effects – such as osteoarthritis – but are caused by the effect of excess weight on the joints, another author, Timothy M. Frayling, PhD, stressed.
“People with obesity and unfavorable adiposity gene variants are at higher risk of all 20 diseases because they have the double hit of the excess mechanical effects and the adverse metabolic effects,” Dr. Frayling of the University of Exeter, told this news organization in an email.
The main clinical message, he said, is that “this research helps inform which conditions may respond better to therapies that lower the adverse effects” of risk factors such as high cholesterol and blood glucose levels, “and high blood pressure, even with no weight loss.”
“In contrast, other conditions really require the weight loss.”
“These results emphasize that many people in the community who are of higher body mass index are at risk of multiple chronic conditions that can severely impair their quality of life or cause morbidity or mortality, even if their metabolic parameters appear relatively normal,” the researchers conclude.
“Whilst it’s important that we identify the causes of obesity-related disease, good genes [are] still no substitute for a healthy lifestyle,” Dr. Martin stressed.
“A favorable adiposity will only go so far. If you’re obese, the advice is to still try and shift the excess weight where you can,” she said.
“The authors have conducted a robust and very comprehensive study using Mendelian randomization to disentangle metabolic and nonmetabolic effects of overweight on a long list of disease outcomes,” reviewing editor Edward D. Janus, MD, PhD, of the University of Melbourne summarized.
“This is an important topic and can help us better understand how overweight influences risk of several important outcomes.”
Metabolic and nonmetabolic diseases caused by obesity
The researchers aimed to investigate the effects of adiposity on metabolic and nonmetabolic diseases caused by obesity.
They used data from 176,899 individuals in the FinnGen project in Finland and from over 500,000 individuals in the UK Biobank database.
They performed Mendelian randomization studies to investigate the causal association between BMI, body fat percentage, favorable adiposity alleles, and unfavorable adiposity alleles with 37 disease outcomes.
Of these 37 chronic diseases associated with obesity, 11 diseases were directly related to the metabolic effect of adiposity (where favorable adiposity or unfavorable adiposity gene variants had opposite effects). Nine other diseases were unrelated to the metabolic effects of adiposity.
For most of the remaining diseases – for example, Alzheimer’s disease and different cancers – it was difficult to draw firm conclusions about the respective roles of favorable adiposity and unfavorable adiposity gene variants.
The study was funded by Diabetes UK, the UK Medical Research Council, the World Cancer Research Fund, and the National Cancer Institute. Author disclosures are listed with the article.
A version of this article first appeared on Medscape.com.
in a large new genetics study.
That is, people with unfavorable adiposity gene variants had fat stored under the skin throughout the body, but they also had more ectopic fat (fat in the “wrong place”) surrounding the pancreas and liver, which is associated with a higher risk of metabolic diseases such as heart disease and type 2 diabetes.
In contrast, people with favorable adiposity gene variants had more subcutaneous fat (such as a paunch or a double chin).
The study by Susan Martin, PhD, a postdoctoral research associate at the University of Exeter (England) and colleagues, was recently published in eLife.
“Some people have ‘unlucky fat genes,’ meaning they store higher levels of fat everywhere, including under the skin [and around the] liver and pancreas. That’s associated with a higher risk of diseases such as type 2 diabetes,” senior author Hanieh Yaghootkar, MD, PhD, summarized in a press release from the University of Exeter.
“Others are luckier and have genes that mean higher fat under the skin but lower liver fat and a lower risk of diseases like type 2 diabetes,” added Dr. Yaghootkar, from Brunel University London.
Among 37 chronic diseases that are associated with obesity, the researchers found the metabolic effects of adiposity are likely the main cause of the following 11: type 2 diabetes, polycystic ovary syndrome, coronary artery disease, peripheral artery disease, hypertension, stroke, heart failure, atrial fibrillation, chronic kidney disease, renal cancer, and gout.
On the other hand, excess weight itself (such as a heavy load on the joints) rather than a metabolic effect is associated with nine other obesity-related diseases: osteoarthritis, rheumatoid arthritis, osteoporosis, gastro-esophageal reflux disease, gallstones, adult-onset asthma, psoriasis, deep vein thrombosis, and venous thromboembolism.
Good genes no substitute for a healthy lifestyle
“People with more favorable adiposity gene variants are still at risk of the nine diseases” that are not caused by metabolic effects – such as osteoarthritis – but are caused by the effect of excess weight on the joints, another author, Timothy M. Frayling, PhD, stressed.
“People with obesity and unfavorable adiposity gene variants are at higher risk of all 20 diseases because they have the double hit of the excess mechanical effects and the adverse metabolic effects,” Dr. Frayling of the University of Exeter, told this news organization in an email.
The main clinical message, he said, is that “this research helps inform which conditions may respond better to therapies that lower the adverse effects” of risk factors such as high cholesterol and blood glucose levels, “and high blood pressure, even with no weight loss.”
“In contrast, other conditions really require the weight loss.”
“These results emphasize that many people in the community who are of higher body mass index are at risk of multiple chronic conditions that can severely impair their quality of life or cause morbidity or mortality, even if their metabolic parameters appear relatively normal,” the researchers conclude.
“Whilst it’s important that we identify the causes of obesity-related disease, good genes [are] still no substitute for a healthy lifestyle,” Dr. Martin stressed.
“A favorable adiposity will only go so far. If you’re obese, the advice is to still try and shift the excess weight where you can,” she said.
“The authors have conducted a robust and very comprehensive study using Mendelian randomization to disentangle metabolic and nonmetabolic effects of overweight on a long list of disease outcomes,” reviewing editor Edward D. Janus, MD, PhD, of the University of Melbourne summarized.
“This is an important topic and can help us better understand how overweight influences risk of several important outcomes.”
Metabolic and nonmetabolic diseases caused by obesity
The researchers aimed to investigate the effects of adiposity on metabolic and nonmetabolic diseases caused by obesity.
They used data from 176,899 individuals in the FinnGen project in Finland and from over 500,000 individuals in the UK Biobank database.
They performed Mendelian randomization studies to investigate the causal association between BMI, body fat percentage, favorable adiposity alleles, and unfavorable adiposity alleles with 37 disease outcomes.
Of these 37 chronic diseases associated with obesity, 11 diseases were directly related to the metabolic effect of adiposity (where favorable adiposity or unfavorable adiposity gene variants had opposite effects). Nine other diseases were unrelated to the metabolic effects of adiposity.
For most of the remaining diseases – for example, Alzheimer’s disease and different cancers – it was difficult to draw firm conclusions about the respective roles of favorable adiposity and unfavorable adiposity gene variants.
The study was funded by Diabetes UK, the UK Medical Research Council, the World Cancer Research Fund, and the National Cancer Institute. Author disclosures are listed with the article.
A version of this article first appeared on Medscape.com.
in a large new genetics study.
That is, people with unfavorable adiposity gene variants had fat stored under the skin throughout the body, but they also had more ectopic fat (fat in the “wrong place”) surrounding the pancreas and liver, which is associated with a higher risk of metabolic diseases such as heart disease and type 2 diabetes.
In contrast, people with favorable adiposity gene variants had more subcutaneous fat (such as a paunch or a double chin).
The study by Susan Martin, PhD, a postdoctoral research associate at the University of Exeter (England) and colleagues, was recently published in eLife.
“Some people have ‘unlucky fat genes,’ meaning they store higher levels of fat everywhere, including under the skin [and around the] liver and pancreas. That’s associated with a higher risk of diseases such as type 2 diabetes,” senior author Hanieh Yaghootkar, MD, PhD, summarized in a press release from the University of Exeter.
“Others are luckier and have genes that mean higher fat under the skin but lower liver fat and a lower risk of diseases like type 2 diabetes,” added Dr. Yaghootkar, from Brunel University London.
Among 37 chronic diseases that are associated with obesity, the researchers found the metabolic effects of adiposity are likely the main cause of the following 11: type 2 diabetes, polycystic ovary syndrome, coronary artery disease, peripheral artery disease, hypertension, stroke, heart failure, atrial fibrillation, chronic kidney disease, renal cancer, and gout.
On the other hand, excess weight itself (such as a heavy load on the joints) rather than a metabolic effect is associated with nine other obesity-related diseases: osteoarthritis, rheumatoid arthritis, osteoporosis, gastro-esophageal reflux disease, gallstones, adult-onset asthma, psoriasis, deep vein thrombosis, and venous thromboembolism.
Good genes no substitute for a healthy lifestyle
“People with more favorable adiposity gene variants are still at risk of the nine diseases” that are not caused by metabolic effects – such as osteoarthritis – but are caused by the effect of excess weight on the joints, another author, Timothy M. Frayling, PhD, stressed.
“People with obesity and unfavorable adiposity gene variants are at higher risk of all 20 diseases because they have the double hit of the excess mechanical effects and the adverse metabolic effects,” Dr. Frayling of the University of Exeter, told this news organization in an email.
The main clinical message, he said, is that “this research helps inform which conditions may respond better to therapies that lower the adverse effects” of risk factors such as high cholesterol and blood glucose levels, “and high blood pressure, even with no weight loss.”
“In contrast, other conditions really require the weight loss.”
“These results emphasize that many people in the community who are of higher body mass index are at risk of multiple chronic conditions that can severely impair their quality of life or cause morbidity or mortality, even if their metabolic parameters appear relatively normal,” the researchers conclude.
“Whilst it’s important that we identify the causes of obesity-related disease, good genes [are] still no substitute for a healthy lifestyle,” Dr. Martin stressed.
“A favorable adiposity will only go so far. If you’re obese, the advice is to still try and shift the excess weight where you can,” she said.
“The authors have conducted a robust and very comprehensive study using Mendelian randomization to disentangle metabolic and nonmetabolic effects of overweight on a long list of disease outcomes,” reviewing editor Edward D. Janus, MD, PhD, of the University of Melbourne summarized.
“This is an important topic and can help us better understand how overweight influences risk of several important outcomes.”
Metabolic and nonmetabolic diseases caused by obesity
The researchers aimed to investigate the effects of adiposity on metabolic and nonmetabolic diseases caused by obesity.
They used data from 176,899 individuals in the FinnGen project in Finland and from over 500,000 individuals in the UK Biobank database.
They performed Mendelian randomization studies to investigate the causal association between BMI, body fat percentage, favorable adiposity alleles, and unfavorable adiposity alleles with 37 disease outcomes.
Of these 37 chronic diseases associated with obesity, 11 diseases were directly related to the metabolic effect of adiposity (where favorable adiposity or unfavorable adiposity gene variants had opposite effects). Nine other diseases were unrelated to the metabolic effects of adiposity.
For most of the remaining diseases – for example, Alzheimer’s disease and different cancers – it was difficult to draw firm conclusions about the respective roles of favorable adiposity and unfavorable adiposity gene variants.
The study was funded by Diabetes UK, the UK Medical Research Council, the World Cancer Research Fund, and the National Cancer Institute. Author disclosures are listed with the article.
A version of this article first appeared on Medscape.com.
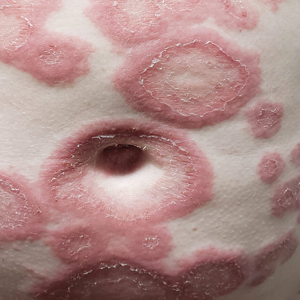
Diffuse Urticarial Rash in a Pregnant Patient
The Diagnosis: Pemphigoid Gestationis
A lesional biopsy showed a subepidermal split with eosinophils and neutrophils. Perilesional biopsy for direct immunofluorescence (DIF) showed linear deposition of 3+ C3 along the basement membrane zone. The clinical, histopathologic, and immunofluorescent findings were consistent with pemphigoid gestationis (PG). Prednisone 1 mg/kg daily was initiated. Her condition continued to worsen, and cyclosporine 250 mg daily was added while prednisone was tapered, with remission of disease.
Pemphigoid gestationis is an autoimmune bullous dermatosis that occurs in the second or third trimester of pregnancy, with an incidence of 1 in 50,000 to 60,000 pregnancies.1 In terms of pathogenesis, aberrant expression of major histocompatibility complex class II molecules on placental tissues causes the loss of immune tolerance of the placenta, which leads to the production of antibodies against the placental protein bullous pemphigoid 180.2 Bullous pemphigoid 180 also is a hemidesmosomal protein found in the skin of the mother; therefore, the presence of the circulating antibodies leads to separation at the dermoepidermal junction and vesiculation.
Pemphigoid gestationis is characterized by the sudden eruption of intensely pruritic urticarial papules and plaques, classically with periumbilical involvement. Tense vesicles and bullae can develop. Women with PG have an increased risk for development of Graves disease. Histopathology shows subepidermal vesiculation with a predominance of eosinophils. Direct immunofluorescence classically shows linear deposition of C3 along the basement membrane zone. Fetal complications include prematurity and small size for gestational age. Additionally, blisters can be seen in 5% to 10% of neonates due to placental transmission of autoantibodies.3
Frequently PG flares shortly postpartum. Pemphigoid gestationis resolves within 6 months postdelivery but frequently reoccurs in subsequent pregnancies. Mild disease can be treated with mid- to high-potency topical corticosteroids. Severe disease is managed with oral corticosteroids, most commonly prednisone. Refractory disease is managed with azathioprine, cyclosporine, intravenous immunoglobulin, or plasmapheresis.
The differential diagnosis of PG includes other pregnancy-associated dermatoses such as atopic eruption of pregnancy, impetigo herpetiformis, intrahepatic cholestasis of pregnancy, and polymorphous eruption of pregnancy. Atopic eruption of pregnancy is the most common dermatosis of pregnancy and is characterized by an eczematous eruption in patients with an atopic history, typically in the first trimester. Blisters are not seen, and DIF is negative. Impetigo herpetiformis, or pustular psoriasis of pregnancy, is a variant of generalized pustular psoriasis that occurs during pregnancy. Diffuse erythematous patches studded with pustules, rather than vesicles, are seen; DIF is negative. Intrahepatic cholestasis of pregnancy presents without primary skin findings and severe pruritus predominantly on the palms and soles, often with secondary excoriations. Polymorphous eruption of pregnancy presents as a polymorphous eruption of urticarial to erythematous papules and plaques commonly originating in striae. In contrast to PG, there is periumbilical sparing, vesiculation is rare, and DIF is negative.
- Shornick JK, Bangert JL, Freeman RG, et al. Herpes gestationis: clinical and histologic features of twenty-eight cases. J Am Acad Dermatol. 1983;8:214-224.
- Sadik CD, Lima AL, Zillikens D. Pemphigoid gestationis: toward a better understanding of the etiopathogenesis. Clin Dermatol. 2016;34:378-382.
- Shornick JK, Black MM. Fetal risks in herpes gestationis. J Am Acad Dermatol. 1992;26:63-68.
The Diagnosis: Pemphigoid Gestationis
A lesional biopsy showed a subepidermal split with eosinophils and neutrophils. Perilesional biopsy for direct immunofluorescence (DIF) showed linear deposition of 3+ C3 along the basement membrane zone. The clinical, histopathologic, and immunofluorescent findings were consistent with pemphigoid gestationis (PG). Prednisone 1 mg/kg daily was initiated. Her condition continued to worsen, and cyclosporine 250 mg daily was added while prednisone was tapered, with remission of disease.
Pemphigoid gestationis is an autoimmune bullous dermatosis that occurs in the second or third trimester of pregnancy, with an incidence of 1 in 50,000 to 60,000 pregnancies.1 In terms of pathogenesis, aberrant expression of major histocompatibility complex class II molecules on placental tissues causes the loss of immune tolerance of the placenta, which leads to the production of antibodies against the placental protein bullous pemphigoid 180.2 Bullous pemphigoid 180 also is a hemidesmosomal protein found in the skin of the mother; therefore, the presence of the circulating antibodies leads to separation at the dermoepidermal junction and vesiculation.
Pemphigoid gestationis is characterized by the sudden eruption of intensely pruritic urticarial papules and plaques, classically with periumbilical involvement. Tense vesicles and bullae can develop. Women with PG have an increased risk for development of Graves disease. Histopathology shows subepidermal vesiculation with a predominance of eosinophils. Direct immunofluorescence classically shows linear deposition of C3 along the basement membrane zone. Fetal complications include prematurity and small size for gestational age. Additionally, blisters can be seen in 5% to 10% of neonates due to placental transmission of autoantibodies.3
Frequently PG flares shortly postpartum. Pemphigoid gestationis resolves within 6 months postdelivery but frequently reoccurs in subsequent pregnancies. Mild disease can be treated with mid- to high-potency topical corticosteroids. Severe disease is managed with oral corticosteroids, most commonly prednisone. Refractory disease is managed with azathioprine, cyclosporine, intravenous immunoglobulin, or plasmapheresis.
The differential diagnosis of PG includes other pregnancy-associated dermatoses such as atopic eruption of pregnancy, impetigo herpetiformis, intrahepatic cholestasis of pregnancy, and polymorphous eruption of pregnancy. Atopic eruption of pregnancy is the most common dermatosis of pregnancy and is characterized by an eczematous eruption in patients with an atopic history, typically in the first trimester. Blisters are not seen, and DIF is negative. Impetigo herpetiformis, or pustular psoriasis of pregnancy, is a variant of generalized pustular psoriasis that occurs during pregnancy. Diffuse erythematous patches studded with pustules, rather than vesicles, are seen; DIF is negative. Intrahepatic cholestasis of pregnancy presents without primary skin findings and severe pruritus predominantly on the palms and soles, often with secondary excoriations. Polymorphous eruption of pregnancy presents as a polymorphous eruption of urticarial to erythematous papules and plaques commonly originating in striae. In contrast to PG, there is periumbilical sparing, vesiculation is rare, and DIF is negative.
The Diagnosis: Pemphigoid Gestationis
A lesional biopsy showed a subepidermal split with eosinophils and neutrophils. Perilesional biopsy for direct immunofluorescence (DIF) showed linear deposition of 3+ C3 along the basement membrane zone. The clinical, histopathologic, and immunofluorescent findings were consistent with pemphigoid gestationis (PG). Prednisone 1 mg/kg daily was initiated. Her condition continued to worsen, and cyclosporine 250 mg daily was added while prednisone was tapered, with remission of disease.
Pemphigoid gestationis is an autoimmune bullous dermatosis that occurs in the second or third trimester of pregnancy, with an incidence of 1 in 50,000 to 60,000 pregnancies.1 In terms of pathogenesis, aberrant expression of major histocompatibility complex class II molecules on placental tissues causes the loss of immune tolerance of the placenta, which leads to the production of antibodies against the placental protein bullous pemphigoid 180.2 Bullous pemphigoid 180 also is a hemidesmosomal protein found in the skin of the mother; therefore, the presence of the circulating antibodies leads to separation at the dermoepidermal junction and vesiculation.
Pemphigoid gestationis is characterized by the sudden eruption of intensely pruritic urticarial papules and plaques, classically with periumbilical involvement. Tense vesicles and bullae can develop. Women with PG have an increased risk for development of Graves disease. Histopathology shows subepidermal vesiculation with a predominance of eosinophils. Direct immunofluorescence classically shows linear deposition of C3 along the basement membrane zone. Fetal complications include prematurity and small size for gestational age. Additionally, blisters can be seen in 5% to 10% of neonates due to placental transmission of autoantibodies.3
Frequently PG flares shortly postpartum. Pemphigoid gestationis resolves within 6 months postdelivery but frequently reoccurs in subsequent pregnancies. Mild disease can be treated with mid- to high-potency topical corticosteroids. Severe disease is managed with oral corticosteroids, most commonly prednisone. Refractory disease is managed with azathioprine, cyclosporine, intravenous immunoglobulin, or plasmapheresis.
The differential diagnosis of PG includes other pregnancy-associated dermatoses such as atopic eruption of pregnancy, impetigo herpetiformis, intrahepatic cholestasis of pregnancy, and polymorphous eruption of pregnancy. Atopic eruption of pregnancy is the most common dermatosis of pregnancy and is characterized by an eczematous eruption in patients with an atopic history, typically in the first trimester. Blisters are not seen, and DIF is negative. Impetigo herpetiformis, or pustular psoriasis of pregnancy, is a variant of generalized pustular psoriasis that occurs during pregnancy. Diffuse erythematous patches studded with pustules, rather than vesicles, are seen; DIF is negative. Intrahepatic cholestasis of pregnancy presents without primary skin findings and severe pruritus predominantly on the palms and soles, often with secondary excoriations. Polymorphous eruption of pregnancy presents as a polymorphous eruption of urticarial to erythematous papules and plaques commonly originating in striae. In contrast to PG, there is periumbilical sparing, vesiculation is rare, and DIF is negative.
- Shornick JK, Bangert JL, Freeman RG, et al. Herpes gestationis: clinical and histologic features of twenty-eight cases. J Am Acad Dermatol. 1983;8:214-224.
- Sadik CD, Lima AL, Zillikens D. Pemphigoid gestationis: toward a better understanding of the etiopathogenesis. Clin Dermatol. 2016;34:378-382.
- Shornick JK, Black MM. Fetal risks in herpes gestationis. J Am Acad Dermatol. 1992;26:63-68.
- Shornick JK, Bangert JL, Freeman RG, et al. Herpes gestationis: clinical and histologic features of twenty-eight cases. J Am Acad Dermatol. 1983;8:214-224.
- Sadik CD, Lima AL, Zillikens D. Pemphigoid gestationis: toward a better understanding of the etiopathogenesis. Clin Dermatol. 2016;34:378-382.
- Shornick JK, Black MM. Fetal risks in herpes gestationis. J Am Acad Dermatol. 1992;26:63-68.
A 29-year-old pregnant woman at 18 weeks and 5 days of gestation presented with a diffuse, pruritic, blistering rash of 5 weeks’ duration that started on the forearms and generalized to affect the trunk, legs, palms, and soles. Physical examination showed diffuse urticarial papules and plaques with small tense vesicles with an annular configuration on the abdomen and marked periumbilical involvement.
Children and COVID-19: The Omicron tide may have turned
The Omicron-fueled surge appears to have peaked as new cases of COVID-19 in U.S. children dropped for the first time since late November 2021, dipping back below the 1 million mark for the week, according to the American Academy of Pediatrics and the Children’s Hospital Association.
The total number of cases in children was up to 11.4 million as of Jan. 27, with children representing 18.6% of all cases reported since the pandemic started, the AAP and CHA said in their weekly COVID-19 report.
As children remain the largest reservoir of unvaccinated Americans, their share of the COVID case load continues to rise quickly. Just 2 weeks ago, children made up 17.8% of the cumulative number of cases, and at the end of December it was 17.4%, the AAP/CHA data show.
The latest data from the Centers for Disease Control and Prevention show that trends for admissions and emergency department visits reflect the decline in new cases. New admissions of children aged 0-17 years with diagnosed COVID-19 peaked at 1.25 per 100,000 population on Jan. 15 and were down to 0.95 per 100,000 on Jan. 29.
Daily ED visits for COVID-19, measured as a percentage of all ED visits, peaked at 13.9% on Jan. 14 for children aged 0-11 years and on Jan. 9 for both 12- to 15-year-olds (14.1%) and 16- to 17-year-olds (13.8%). By Jan. 28, the rates were down to 5.6% (0-11), 3.1% (12-15), and 3.3% (16-17), the CDC reported based on data from the National Syndromic Surveillance Program.
Trends involving more severe illness support observations that Omicron is milder than earlier variants. Children hospitalized with COVID-19 were less likely to be admitted to an intensive care unit over the last 2 months than during the Delta surge in the late summer and early fall or during the winter of 2020-2021, the CDC said based on data from the BD Insights Research Database, which includes 229,000 patients and 267 hospitals.
Those data show that the highest monthly rate occurred early on, in May of 2020, when 27.8% of children with COVID-19 ended up in the ICU. The rates for December 2021 and January 2022, by comparison, were 11.0% and 11.3%, respectively, the CDC said.
Vaccination lags in younger children
As reports surface about Pfizer-BioNTech filing an emergency use request to extend vaccine coverage to children aged 6 months to 5 years, it does appear that prevention efforts could use the proverbial shot in the arm.
As of Jan. 30, just 30.4% of children aged 5-11 have received at least one dose of the COVID-19 vaccine, and only 21.6% are fully vaccinated. At a comparable point in their timeline – just short of 3 months after approval – the respective numbers for children aged 12-15 were about 42% and 31%, CDC data show.
In the younger group, both initial doses and completions rose slightly in the first 2 weeks of January but then dropped in each of the last 2 weeks. There was a more significant surge in interest among the 12- to 17-year-olds in mid-January, but the last full week of the month brought declines of more than 50% in both measures, according to a separate AAP analysis.
The Omicron-fueled surge appears to have peaked as new cases of COVID-19 in U.S. children dropped for the first time since late November 2021, dipping back below the 1 million mark for the week, according to the American Academy of Pediatrics and the Children’s Hospital Association.
The total number of cases in children was up to 11.4 million as of Jan. 27, with children representing 18.6% of all cases reported since the pandemic started, the AAP and CHA said in their weekly COVID-19 report.
As children remain the largest reservoir of unvaccinated Americans, their share of the COVID case load continues to rise quickly. Just 2 weeks ago, children made up 17.8% of the cumulative number of cases, and at the end of December it was 17.4%, the AAP/CHA data show.
The latest data from the Centers for Disease Control and Prevention show that trends for admissions and emergency department visits reflect the decline in new cases. New admissions of children aged 0-17 years with diagnosed COVID-19 peaked at 1.25 per 100,000 population on Jan. 15 and were down to 0.95 per 100,000 on Jan. 29.
Daily ED visits for COVID-19, measured as a percentage of all ED visits, peaked at 13.9% on Jan. 14 for children aged 0-11 years and on Jan. 9 for both 12- to 15-year-olds (14.1%) and 16- to 17-year-olds (13.8%). By Jan. 28, the rates were down to 5.6% (0-11), 3.1% (12-15), and 3.3% (16-17), the CDC reported based on data from the National Syndromic Surveillance Program.
Trends involving more severe illness support observations that Omicron is milder than earlier variants. Children hospitalized with COVID-19 were less likely to be admitted to an intensive care unit over the last 2 months than during the Delta surge in the late summer and early fall or during the winter of 2020-2021, the CDC said based on data from the BD Insights Research Database, which includes 229,000 patients and 267 hospitals.
Those data show that the highest monthly rate occurred early on, in May of 2020, when 27.8% of children with COVID-19 ended up in the ICU. The rates for December 2021 and January 2022, by comparison, were 11.0% and 11.3%, respectively, the CDC said.
Vaccination lags in younger children
As reports surface about Pfizer-BioNTech filing an emergency use request to extend vaccine coverage to children aged 6 months to 5 years, it does appear that prevention efforts could use the proverbial shot in the arm.
As of Jan. 30, just 30.4% of children aged 5-11 have received at least one dose of the COVID-19 vaccine, and only 21.6% are fully vaccinated. At a comparable point in their timeline – just short of 3 months after approval – the respective numbers for children aged 12-15 were about 42% and 31%, CDC data show.
In the younger group, both initial doses and completions rose slightly in the first 2 weeks of January but then dropped in each of the last 2 weeks. There was a more significant surge in interest among the 12- to 17-year-olds in mid-January, but the last full week of the month brought declines of more than 50% in both measures, according to a separate AAP analysis.
The Omicron-fueled surge appears to have peaked as new cases of COVID-19 in U.S. children dropped for the first time since late November 2021, dipping back below the 1 million mark for the week, according to the American Academy of Pediatrics and the Children’s Hospital Association.
The total number of cases in children was up to 11.4 million as of Jan. 27, with children representing 18.6% of all cases reported since the pandemic started, the AAP and CHA said in their weekly COVID-19 report.
As children remain the largest reservoir of unvaccinated Americans, their share of the COVID case load continues to rise quickly. Just 2 weeks ago, children made up 17.8% of the cumulative number of cases, and at the end of December it was 17.4%, the AAP/CHA data show.
The latest data from the Centers for Disease Control and Prevention show that trends for admissions and emergency department visits reflect the decline in new cases. New admissions of children aged 0-17 years with diagnosed COVID-19 peaked at 1.25 per 100,000 population on Jan. 15 and were down to 0.95 per 100,000 on Jan. 29.
Daily ED visits for COVID-19, measured as a percentage of all ED visits, peaked at 13.9% on Jan. 14 for children aged 0-11 years and on Jan. 9 for both 12- to 15-year-olds (14.1%) and 16- to 17-year-olds (13.8%). By Jan. 28, the rates were down to 5.6% (0-11), 3.1% (12-15), and 3.3% (16-17), the CDC reported based on data from the National Syndromic Surveillance Program.
Trends involving more severe illness support observations that Omicron is milder than earlier variants. Children hospitalized with COVID-19 were less likely to be admitted to an intensive care unit over the last 2 months than during the Delta surge in the late summer and early fall or during the winter of 2020-2021, the CDC said based on data from the BD Insights Research Database, which includes 229,000 patients and 267 hospitals.
Those data show that the highest monthly rate occurred early on, in May of 2020, when 27.8% of children with COVID-19 ended up in the ICU. The rates for December 2021 and January 2022, by comparison, were 11.0% and 11.3%, respectively, the CDC said.
Vaccination lags in younger children
As reports surface about Pfizer-BioNTech filing an emergency use request to extend vaccine coverage to children aged 6 months to 5 years, it does appear that prevention efforts could use the proverbial shot in the arm.
As of Jan. 30, just 30.4% of children aged 5-11 have received at least one dose of the COVID-19 vaccine, and only 21.6% are fully vaccinated. At a comparable point in their timeline – just short of 3 months after approval – the respective numbers for children aged 12-15 were about 42% and 31%, CDC data show.
In the younger group, both initial doses and completions rose slightly in the first 2 weeks of January but then dropped in each of the last 2 weeks. There was a more significant surge in interest among the 12- to 17-year-olds in mid-January, but the last full week of the month brought declines of more than 50% in both measures, according to a separate AAP analysis.
CDC issues new pneumococcal vaccine recommendations for adults
The recommendations, voted on by the CDC’s Advisory Committee on Immunization Practices (ACIP) in October and made final in January with publication in the agency’s Morbidity and Mortality Weekly Report (MMWR), call for use of the 15-valent pneumococcal conjugate vaccine (PCV15; Vaxneuvance, Merck Sharp & Dohme) or 20-valent PCV (PREVNAR20; Wyeth Pharmaceuticals).
The recommendations apply to PCV-naive adults in the United States who are either aged 65 years or older, or who are aged 19-64 years and have underlying conditions such as diabetes, chronic heart or liver disease, or HIV, and have not previously received a PCV or whose previous vaccination history is unknown.
If the PCV15 vaccine is used, a subsequent dose of the 23-valent pneumococcal polysaccharide vaccine (PPSV23; Pneumovax23, Merck Sharp & Dohme) should be provided, typically at least 1 year later, under the recommendations.
As reported by this news organization, PCV15 and PREVNAR20 received approval from the Food and Drug Administration last July.
Those approvals provided an impetus for the revised recommendations, “offer[ing] an opportunity to review the existing recommendations and available data,” Miwako Kobayashi, MD, first author of the MMWR report and a medical epidemiologist with the National Center for Immunization and Respiratory Diseases, CDC, in Atlanta, said in an interview.
“As part of that process, ACIP strived to simplify the recommendations,” she said.
The previous recommendations called for the PCV13 vaccine and the PPSV23 and had varying conditions (depending on certain age and risk groups) that added complexity to the process. Under the new approach, the same recommendation applies regardless of specific medical conditions or other risk factors.
“With the simplified recommendation for adults 19 through 64, we expect coverage may increase among this population,” Dr. Kobayashi said.
Compared with the PCV13 vaccine, PREVNAR20 protects against seven additional serotypes involved in cases of invasive pneumococcal disease (IPD) and pneumonia, which are responsible for up to 40% of all cases of pneumococcal disease and related deaths in the United States.
While the PREVNAR20 includes five more pneumococcal serotypes than PCV15, the
CDC does not recommend one over the other, Dr. Kobayashi noted.
More than 90% of cases of adult IPD involve older adults and adults with chronic medical conditions or immunocompromising conditions, cerebrospinal fluid leaks, or cochlear implants, the MMWR report notes.
Commenting on the recommendations, Amit A. Shah, MD, a geriatrician with the Mayo Clinic in Phoenix, Ariz., underscored the need for clinicians to be proactive in recommending the vaccines to those patients.
“Despite only needing one vaccine dose after turning 65 to be considered vaccinated, only about 70% of people in this group have received any pneumococcal vaccination,” he said in an interview. “This percentage has not increased much over the past several years.”
The new approach should help change that, he said.
“These new recommendations are a significant simplification from the prior confusing and challenging-to-implement recommendations from 2019,” Dr. Shah explained.
Among the 2019 recommendations was a stipulation for “shared decision-making” with PCV13, and a conversation that often only complicated matters, he noted.
“Patients and providers alike had confusion about this since it was not a clear-cut ‘yes, give it’ or ‘no, do not give it any longer’ recommendation.”
“Now that this new recommendation will require no extra time for a discussion in the clinic, and just a simple ‘it’s time for your pneumonia shot’ offer, this may become more feasible,” Dr. Shah added. “In addition, removal of the shared decision-making stipulation allows for this immunization to be easily protocolized in the clinic, similar to automatic offers to the flu vaccine for patients each year.”
According to the CDC, pneumococcal pneumonia causes an estimated 150,000 hospitalizations each year in the United States, while pneumococcal meningitis and bacteremia killed approximately 3,250 people in the United States in 2019.
“Clinicians are patients’ most trusted resource when it comes to vaccine recommendations,” Dr. Kobayashi said. “We encourage all clinicians to recommend pneumococcal vaccines when indicated.”
Dr. Kobayashi and Dr. Shah have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The recommendations, voted on by the CDC’s Advisory Committee on Immunization Practices (ACIP) in October and made final in January with publication in the agency’s Morbidity and Mortality Weekly Report (MMWR), call for use of the 15-valent pneumococcal conjugate vaccine (PCV15; Vaxneuvance, Merck Sharp & Dohme) or 20-valent PCV (PREVNAR20; Wyeth Pharmaceuticals).
The recommendations apply to PCV-naive adults in the United States who are either aged 65 years or older, or who are aged 19-64 years and have underlying conditions such as diabetes, chronic heart or liver disease, or HIV, and have not previously received a PCV or whose previous vaccination history is unknown.
If the PCV15 vaccine is used, a subsequent dose of the 23-valent pneumococcal polysaccharide vaccine (PPSV23; Pneumovax23, Merck Sharp & Dohme) should be provided, typically at least 1 year later, under the recommendations.
As reported by this news organization, PCV15 and PREVNAR20 received approval from the Food and Drug Administration last July.
Those approvals provided an impetus for the revised recommendations, “offer[ing] an opportunity to review the existing recommendations and available data,” Miwako Kobayashi, MD, first author of the MMWR report and a medical epidemiologist with the National Center for Immunization and Respiratory Diseases, CDC, in Atlanta, said in an interview.
“As part of that process, ACIP strived to simplify the recommendations,” she said.
The previous recommendations called for the PCV13 vaccine and the PPSV23 and had varying conditions (depending on certain age and risk groups) that added complexity to the process. Under the new approach, the same recommendation applies regardless of specific medical conditions or other risk factors.
“With the simplified recommendation for adults 19 through 64, we expect coverage may increase among this population,” Dr. Kobayashi said.
Compared with the PCV13 vaccine, PREVNAR20 protects against seven additional serotypes involved in cases of invasive pneumococcal disease (IPD) and pneumonia, which are responsible for up to 40% of all cases of pneumococcal disease and related deaths in the United States.
While the PREVNAR20 includes five more pneumococcal serotypes than PCV15, the
CDC does not recommend one over the other, Dr. Kobayashi noted.
More than 90% of cases of adult IPD involve older adults and adults with chronic medical conditions or immunocompromising conditions, cerebrospinal fluid leaks, or cochlear implants, the MMWR report notes.
Commenting on the recommendations, Amit A. Shah, MD, a geriatrician with the Mayo Clinic in Phoenix, Ariz., underscored the need for clinicians to be proactive in recommending the vaccines to those patients.
“Despite only needing one vaccine dose after turning 65 to be considered vaccinated, only about 70% of people in this group have received any pneumococcal vaccination,” he said in an interview. “This percentage has not increased much over the past several years.”
The new approach should help change that, he said.
“These new recommendations are a significant simplification from the prior confusing and challenging-to-implement recommendations from 2019,” Dr. Shah explained.
Among the 2019 recommendations was a stipulation for “shared decision-making” with PCV13, and a conversation that often only complicated matters, he noted.
“Patients and providers alike had confusion about this since it was not a clear-cut ‘yes, give it’ or ‘no, do not give it any longer’ recommendation.”
“Now that this new recommendation will require no extra time for a discussion in the clinic, and just a simple ‘it’s time for your pneumonia shot’ offer, this may become more feasible,” Dr. Shah added. “In addition, removal of the shared decision-making stipulation allows for this immunization to be easily protocolized in the clinic, similar to automatic offers to the flu vaccine for patients each year.”
According to the CDC, pneumococcal pneumonia causes an estimated 150,000 hospitalizations each year in the United States, while pneumococcal meningitis and bacteremia killed approximately 3,250 people in the United States in 2019.
“Clinicians are patients’ most trusted resource when it comes to vaccine recommendations,” Dr. Kobayashi said. “We encourage all clinicians to recommend pneumococcal vaccines when indicated.”
Dr. Kobayashi and Dr. Shah have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The recommendations, voted on by the CDC’s Advisory Committee on Immunization Practices (ACIP) in October and made final in January with publication in the agency’s Morbidity and Mortality Weekly Report (MMWR), call for use of the 15-valent pneumococcal conjugate vaccine (PCV15; Vaxneuvance, Merck Sharp & Dohme) or 20-valent PCV (PREVNAR20; Wyeth Pharmaceuticals).
The recommendations apply to PCV-naive adults in the United States who are either aged 65 years or older, or who are aged 19-64 years and have underlying conditions such as diabetes, chronic heart or liver disease, or HIV, and have not previously received a PCV or whose previous vaccination history is unknown.
If the PCV15 vaccine is used, a subsequent dose of the 23-valent pneumococcal polysaccharide vaccine (PPSV23; Pneumovax23, Merck Sharp & Dohme) should be provided, typically at least 1 year later, under the recommendations.
As reported by this news organization, PCV15 and PREVNAR20 received approval from the Food and Drug Administration last July.
Those approvals provided an impetus for the revised recommendations, “offer[ing] an opportunity to review the existing recommendations and available data,” Miwako Kobayashi, MD, first author of the MMWR report and a medical epidemiologist with the National Center for Immunization and Respiratory Diseases, CDC, in Atlanta, said in an interview.
“As part of that process, ACIP strived to simplify the recommendations,” she said.
The previous recommendations called for the PCV13 vaccine and the PPSV23 and had varying conditions (depending on certain age and risk groups) that added complexity to the process. Under the new approach, the same recommendation applies regardless of specific medical conditions or other risk factors.
“With the simplified recommendation for adults 19 through 64, we expect coverage may increase among this population,” Dr. Kobayashi said.
Compared with the PCV13 vaccine, PREVNAR20 protects against seven additional serotypes involved in cases of invasive pneumococcal disease (IPD) and pneumonia, which are responsible for up to 40% of all cases of pneumococcal disease and related deaths in the United States.
While the PREVNAR20 includes five more pneumococcal serotypes than PCV15, the
CDC does not recommend one over the other, Dr. Kobayashi noted.
More than 90% of cases of adult IPD involve older adults and adults with chronic medical conditions or immunocompromising conditions, cerebrospinal fluid leaks, or cochlear implants, the MMWR report notes.
Commenting on the recommendations, Amit A. Shah, MD, a geriatrician with the Mayo Clinic in Phoenix, Ariz., underscored the need for clinicians to be proactive in recommending the vaccines to those patients.
“Despite only needing one vaccine dose after turning 65 to be considered vaccinated, only about 70% of people in this group have received any pneumococcal vaccination,” he said in an interview. “This percentage has not increased much over the past several years.”
The new approach should help change that, he said.
“These new recommendations are a significant simplification from the prior confusing and challenging-to-implement recommendations from 2019,” Dr. Shah explained.
Among the 2019 recommendations was a stipulation for “shared decision-making” with PCV13, and a conversation that often only complicated matters, he noted.
“Patients and providers alike had confusion about this since it was not a clear-cut ‘yes, give it’ or ‘no, do not give it any longer’ recommendation.”
“Now that this new recommendation will require no extra time for a discussion in the clinic, and just a simple ‘it’s time for your pneumonia shot’ offer, this may become more feasible,” Dr. Shah added. “In addition, removal of the shared decision-making stipulation allows for this immunization to be easily protocolized in the clinic, similar to automatic offers to the flu vaccine for patients each year.”
According to the CDC, pneumococcal pneumonia causes an estimated 150,000 hospitalizations each year in the United States, while pneumococcal meningitis and bacteremia killed approximately 3,250 people in the United States in 2019.
“Clinicians are patients’ most trusted resource when it comes to vaccine recommendations,” Dr. Kobayashi said. “We encourage all clinicians to recommend pneumococcal vaccines when indicated.”
Dr. Kobayashi and Dr. Shah have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM THE MMWR
Researchers eye cannabis for gynecologic pain
Many women use cannabis to help manage gynecologic pain conditions.
When patients ask or tell clinicians about this treatment approach, however, few if any controlled trials exist to inform medical guidance.
A recent review of studies in this area presents a “thorough analysis of this very relevant topic,” said Erin A. Blake, MD, of Presbyterian Cancer Care, Rio Rancho, N.M..
The findings “are consistent with my anecdotal clinical findings as well as the results of my own research,” Dr. Blake said. “Cannabis products represent an underutilized but likely effective modality to relieve pain and other symptoms experienced by our patients.”
Mostly in the dark
Cannabis products “are unregulated and the data we have surrounding them is extremely limited due to outdated federal laws,” said Dr. Blake, who in 2019 described nonprescription cannabis use for symptom management by women with gynecologic malignancies. “Our ability to practice evidence-based medicine related to cannabis products will be limited until we are legally and financially able to design trials to evaluate them in a controlled fashion.”
For the new review, Jenell S. Coleman, MD, MPH, with Johns Hopkins University, Baltimore, and colleagues, identified 16 studies since 1990, including Dr. Blake’s, that examined the use of cannabinoids for managing pain from gynecologic conditions.
Dr. Coleman and her coauthors, Angela L. Liang and Erin L. Gingher, analyzed eight cross-sectional studies, six prospective studies, and two randomized controlled trials.
Patients who used cannabis tended to do so “multiple times per week, and they used a variety of delivery methods and a wide range of doses,” the authors said. “One of the most common reasons for cannabis use was pain management, and all the cross-sectional studies found that most women reported pain relief with cannabis use, especially among women who used a combination of CBD plus THC compared with either cannabinoid alone.”
Cross-sectional studies included patients with chronic pelvic pain (in two of the studies), vulvodynia (one), endometriosis (four), and gynecologic malignancy (two). These studies included between 36 and 3,426 participants and were conducted in the United States, Canada, Australia, and New Zealand.
In one Australian study, for example, Armour and colleagues asked 484 patients with endometriosis to rate the effectiveness of self-management strategies, including cannabis, heat, diet, and exercise, for reducing pelvic pain. Cannabis was used by 13% of the participants and had the highest average effectiveness rating: 7.6 on a 10-point scale.
In some cases, patients who use cannabis may decrease their use of other pain medications, the review found.
Cannabis side effects may include dry mouth, sleepiness, increased appetite, palpitations, and a “high” associated with THC.
Enhancing endogenous cannabinoids
The six prospective cohort studies and two randomized controlled trials examined the effectiveness of compounds – including palmitoylethanolamide (PEA) and a fatty acid amide hydrolase inhibitor – that can enhance endogenous cannabinoids.
Studies of PEA combined with antioxidants showed that these treatments significantly decreased pain from primary dysmenorrhea, pelvic pain, and interstitial cystitis. PEA-combination medications were well tolerated, with nausea and spotting as potential side effects.
On the other hand, a study that assessed a fatty acid amide hydrolase inhibitor found that it did not decrease pain from interstitial cystitis.
Dr. Coleman began reviewing the endocannabinoid system and cannabis research after hearing from patients who were using cannabis for pelvic pain.
Seeing various preclinical data that suggest cannabis could be useful for pain conditions came as a surprise.
Still, the existing evidence base for clinical effectiveness is poor quality, Dr. Coleman said in an interview. Rigorous trials are needed.
“It is a whole field that is just waiting for the U.S. to do something in terms of legalization so that we can actually study to see, does this make sense?” Dr. Coleman said.
Cannabis should not be used while pregnant
In a recent meta-analysis based on data from nearly 60,000 individuals, women who used marijuana during pregnancy were at increased risk for adverse neonatal outcomes such as low birth weight and preterm birth. Study author Greg J. Marchand, MD, of the Marchand Institute for Minimally Invasive Surgery, Mesa, Ariz., noted that the results will force some difficult decisions for mothers who use marijuana to treat medical problems, and that there may not be good substitute treatments for some of these conditions, especially chronic pain and anxiety.
Dr. Coleman disclosed investments in a cannabis exchange-traded fund. Dr. Blake and Dr. Marchand had no relevant financial disclosures.
Many women use cannabis to help manage gynecologic pain conditions.
When patients ask or tell clinicians about this treatment approach, however, few if any controlled trials exist to inform medical guidance.
A recent review of studies in this area presents a “thorough analysis of this very relevant topic,” said Erin A. Blake, MD, of Presbyterian Cancer Care, Rio Rancho, N.M..
The findings “are consistent with my anecdotal clinical findings as well as the results of my own research,” Dr. Blake said. “Cannabis products represent an underutilized but likely effective modality to relieve pain and other symptoms experienced by our patients.”
Mostly in the dark
Cannabis products “are unregulated and the data we have surrounding them is extremely limited due to outdated federal laws,” said Dr. Blake, who in 2019 described nonprescription cannabis use for symptom management by women with gynecologic malignancies. “Our ability to practice evidence-based medicine related to cannabis products will be limited until we are legally and financially able to design trials to evaluate them in a controlled fashion.”
For the new review, Jenell S. Coleman, MD, MPH, with Johns Hopkins University, Baltimore, and colleagues, identified 16 studies since 1990, including Dr. Blake’s, that examined the use of cannabinoids for managing pain from gynecologic conditions.
Dr. Coleman and her coauthors, Angela L. Liang and Erin L. Gingher, analyzed eight cross-sectional studies, six prospective studies, and two randomized controlled trials.
Patients who used cannabis tended to do so “multiple times per week, and they used a variety of delivery methods and a wide range of doses,” the authors said. “One of the most common reasons for cannabis use was pain management, and all the cross-sectional studies found that most women reported pain relief with cannabis use, especially among women who used a combination of CBD plus THC compared with either cannabinoid alone.”
Cross-sectional studies included patients with chronic pelvic pain (in two of the studies), vulvodynia (one), endometriosis (four), and gynecologic malignancy (two). These studies included between 36 and 3,426 participants and were conducted in the United States, Canada, Australia, and New Zealand.
In one Australian study, for example, Armour and colleagues asked 484 patients with endometriosis to rate the effectiveness of self-management strategies, including cannabis, heat, diet, and exercise, for reducing pelvic pain. Cannabis was used by 13% of the participants and had the highest average effectiveness rating: 7.6 on a 10-point scale.
In some cases, patients who use cannabis may decrease their use of other pain medications, the review found.
Cannabis side effects may include dry mouth, sleepiness, increased appetite, palpitations, and a “high” associated with THC.
Enhancing endogenous cannabinoids
The six prospective cohort studies and two randomized controlled trials examined the effectiveness of compounds – including palmitoylethanolamide (PEA) and a fatty acid amide hydrolase inhibitor – that can enhance endogenous cannabinoids.
Studies of PEA combined with antioxidants showed that these treatments significantly decreased pain from primary dysmenorrhea, pelvic pain, and interstitial cystitis. PEA-combination medications were well tolerated, with nausea and spotting as potential side effects.
On the other hand, a study that assessed a fatty acid amide hydrolase inhibitor found that it did not decrease pain from interstitial cystitis.
Dr. Coleman began reviewing the endocannabinoid system and cannabis research after hearing from patients who were using cannabis for pelvic pain.
Seeing various preclinical data that suggest cannabis could be useful for pain conditions came as a surprise.
Still, the existing evidence base for clinical effectiveness is poor quality, Dr. Coleman said in an interview. Rigorous trials are needed.
“It is a whole field that is just waiting for the U.S. to do something in terms of legalization so that we can actually study to see, does this make sense?” Dr. Coleman said.
Cannabis should not be used while pregnant
In a recent meta-analysis based on data from nearly 60,000 individuals, women who used marijuana during pregnancy were at increased risk for adverse neonatal outcomes such as low birth weight and preterm birth. Study author Greg J. Marchand, MD, of the Marchand Institute for Minimally Invasive Surgery, Mesa, Ariz., noted that the results will force some difficult decisions for mothers who use marijuana to treat medical problems, and that there may not be good substitute treatments for some of these conditions, especially chronic pain and anxiety.
Dr. Coleman disclosed investments in a cannabis exchange-traded fund. Dr. Blake and Dr. Marchand had no relevant financial disclosures.
Many women use cannabis to help manage gynecologic pain conditions.
When patients ask or tell clinicians about this treatment approach, however, few if any controlled trials exist to inform medical guidance.
A recent review of studies in this area presents a “thorough analysis of this very relevant topic,” said Erin A. Blake, MD, of Presbyterian Cancer Care, Rio Rancho, N.M..
The findings “are consistent with my anecdotal clinical findings as well as the results of my own research,” Dr. Blake said. “Cannabis products represent an underutilized but likely effective modality to relieve pain and other symptoms experienced by our patients.”
Mostly in the dark
Cannabis products “are unregulated and the data we have surrounding them is extremely limited due to outdated federal laws,” said Dr. Blake, who in 2019 described nonprescription cannabis use for symptom management by women with gynecologic malignancies. “Our ability to practice evidence-based medicine related to cannabis products will be limited until we are legally and financially able to design trials to evaluate them in a controlled fashion.”
For the new review, Jenell S. Coleman, MD, MPH, with Johns Hopkins University, Baltimore, and colleagues, identified 16 studies since 1990, including Dr. Blake’s, that examined the use of cannabinoids for managing pain from gynecologic conditions.
Dr. Coleman and her coauthors, Angela L. Liang and Erin L. Gingher, analyzed eight cross-sectional studies, six prospective studies, and two randomized controlled trials.
Patients who used cannabis tended to do so “multiple times per week, and they used a variety of delivery methods and a wide range of doses,” the authors said. “One of the most common reasons for cannabis use was pain management, and all the cross-sectional studies found that most women reported pain relief with cannabis use, especially among women who used a combination of CBD plus THC compared with either cannabinoid alone.”
Cross-sectional studies included patients with chronic pelvic pain (in two of the studies), vulvodynia (one), endometriosis (four), and gynecologic malignancy (two). These studies included between 36 and 3,426 participants and were conducted in the United States, Canada, Australia, and New Zealand.
In one Australian study, for example, Armour and colleagues asked 484 patients with endometriosis to rate the effectiveness of self-management strategies, including cannabis, heat, diet, and exercise, for reducing pelvic pain. Cannabis was used by 13% of the participants and had the highest average effectiveness rating: 7.6 on a 10-point scale.
In some cases, patients who use cannabis may decrease their use of other pain medications, the review found.
Cannabis side effects may include dry mouth, sleepiness, increased appetite, palpitations, and a “high” associated with THC.
Enhancing endogenous cannabinoids
The six prospective cohort studies and two randomized controlled trials examined the effectiveness of compounds – including palmitoylethanolamide (PEA) and a fatty acid amide hydrolase inhibitor – that can enhance endogenous cannabinoids.
Studies of PEA combined with antioxidants showed that these treatments significantly decreased pain from primary dysmenorrhea, pelvic pain, and interstitial cystitis. PEA-combination medications were well tolerated, with nausea and spotting as potential side effects.
On the other hand, a study that assessed a fatty acid amide hydrolase inhibitor found that it did not decrease pain from interstitial cystitis.
Dr. Coleman began reviewing the endocannabinoid system and cannabis research after hearing from patients who were using cannabis for pelvic pain.
Seeing various preclinical data that suggest cannabis could be useful for pain conditions came as a surprise.
Still, the existing evidence base for clinical effectiveness is poor quality, Dr. Coleman said in an interview. Rigorous trials are needed.
“It is a whole field that is just waiting for the U.S. to do something in terms of legalization so that we can actually study to see, does this make sense?” Dr. Coleman said.
Cannabis should not be used while pregnant
In a recent meta-analysis based on data from nearly 60,000 individuals, women who used marijuana during pregnancy were at increased risk for adverse neonatal outcomes such as low birth weight and preterm birth. Study author Greg J. Marchand, MD, of the Marchand Institute for Minimally Invasive Surgery, Mesa, Ariz., noted that the results will force some difficult decisions for mothers who use marijuana to treat medical problems, and that there may not be good substitute treatments for some of these conditions, especially chronic pain and anxiety.
Dr. Coleman disclosed investments in a cannabis exchange-traded fund. Dr. Blake and Dr. Marchand had no relevant financial disclosures.
FROM OBSTETRICS & GYNECOLOGY
Vitamin D shows no survival benefit in nondeficient elderly
, including mortality linked to cardiovascular disease, new results from a large, placebo-controlled trial show.
“The take-home message is that routine vitamin D supplementation, irrespective of the dosing regimen, is unlikely to be beneficial in a population with a low prevalence of vitamin D deficiency,” first author Rachel E. Neale, PhD, of the Population Health Department, QIMR Berghofer Medical Research Institute, in Brisbane, Australia, told this news organization.
Despite extensive previous research on vitamin D supplementation, “mortality has not been the primary outcome in any previous large trial of high-dose vitamin D supplementation,” Dr. Neale and coauthors noted. The results, published online in Lancet Diabetes & Endocrinology, are from the D-Health trial.
With more than 20,000 participants, this is the largest intermittent-dosing trial to date, the authors noted. The primary outcome was all-cause mortality.
In an accompanying editorial, Inez Schoenmakers, PhD, noted that “the findings [are] highly relevant for population policy, owing to the study’s population-based design, large scale, and long duration.”
This new “research contributes to the concept that improving vitamin D status with supplementation in a mostly vitamin D-replete older population does not influence all-cause mortality,” Dr. Schoenmakers, of the Faculty of Medicine and Health Sciences, University of East Anglia, Norwich, England, said in an interview.
“This is not dissimilar to research with many other nutrients showing that increasing intake above the adequate intake has no further health benefits,” she added.
D-Health Trial
The D-Health Trial involved 21,315 participants in Australia, enrolled between February 2014 and June 2015, who had not been screened for vitamin D deficiency but were largely considered to be vitamin D replete. They were a mean age of 69.3 years and 54% were men.
Participants were randomized 1:1 to a once-monthly oral vitamin D3 supplementation of 60,000 IU (n = 10,662) or a placebo capsule (n = 10,653).
They were permitted to take up to 2,000 IU/day of supplemental vitamin D in addition to the study protocol and had no history of kidney stones, hypercalcemia, hyperparathyroidism, osteomalacia, or sarcoidosis.
Over a median follow-up of 5.7 years, there were 1,100 deaths: 562 in the vitamin D group (5.3%) and 538 in the placebo group (5.1%). With a hazard ratio (HR) for all-cause mortality of 1.04, the difference was not significant (P = .47).
There were also no significant differences in terms of mortality from cardiovascular disease (HR, 0.96; P = .77), cancer (HR, 1.15; P = .13), or other causes (HR, 0.83; P = .15).
Rates of total adverse events between the two groups, including hypercalcemia and kidney stones, were similar.
An exploratory analysis excluding the first 2 years of follow-up in fact showed a numerically higher hazard ratio for cancer mortality in the vitamin D group versus no supplementation (HR, 1.24; P = .05). However, the authors noted that the effect was “not apparent when the analysis was restricted to deaths that were coded by the study team and not officially coded.”
Nevertheless, “our findings, from a large study in an unscreened population, give pause to earlier reports that vitamin D supplements might reduce cancer mortality,” they underscored.
Retention and adherence in the study were high, each exceeding 80%. Although blood samples were not collected at baseline, samples from 3,943 randomly sampled participants during follow-up showed mean serum 25-hydroxy-vitamin D concentrations of 77 nmol/L in the placebo group and 115 nmol/L in the vitamin D group, both within the normal range of 50-125 nmol/L.
Findings supported by previous research
The trial results are consistent with those of prior large studies and meta-analyses of older adults with a low prevalence of vitamin D deficiency showing that vitamin D3 supplementation, regardless of whether taken daily or monthly, is not likely to have an effect on all-cause mortality.
In the US VITAL trial, recently published in the New England Journal of Medicine, among 25,871 participants administered 2,000 IU/day of vitamin D3 for a median of 5.3 years, there was no reduction in all-cause mortality.
The ViDA trial of 5,110 older adults in New Zealand, published in 2019 in the Journal of Endocrinological Investigation, also showed monthly vitamin D3 supplementation of 100,000 IU for a median of 3.3 years was not associated with a benefit in people who were not deficient.
“In total, the results from the large trials and meta-analyses suggest that routine supplementation of older adults in populations with a low prevalence of vitamin D deficiency is unlikely to reduce the rate of all-cause mortality,” Dr. Neale and colleagues concluded.
Longer-term supplementation beneficial?
The population was limited to older adults and the study had a relatively short follow-up period, which Dr. Neale noted was necessary for pragmatic reasons.
“Our primary outcome was all-cause mortality, so to have sufficient deaths we either needed to study older adults or a much larger sample of younger adults,” she explained.
“However, we felt that [the former] ... had biological justification, as there is evidence that vitamin D plays a role later in the course of a number of diseases, with potential impacts on mortality.”
She noted that recent studies evaluating genetically predicted concentrations of serum 25(OH)D have further shown no link between those levels and all-cause mortality, stroke, or coronary heart disease.
“This confirms the statement that vitamin D is unlikely to be beneficial in people who are not vitamin D deficient, irrespective of whether supplementation occurs over the short or longer term,” Dr. Neale said.
The source of vitamin D, itself, is another consideration, with ongoing speculation of differences in benefits between dietary or supplementation sources versus sunlight exposure.
“Exposure to ultraviolet radiation, for which serum 25(OH)D concentration is a good marker, might confer benefits not mediated by vitamin D,” Dr. Neale and coauthors noted.
They added that the results in the older Australian population “cannot be generalized to populations with a higher prevalence of vitamin D deficiency, or with a greater proportion of people not of White ancestry, than the study population.”
Ten-year mortality rates from the D-Health trial are expected to be reported in the future.
Strategies still needed to address vitamin D deficiency
Further commenting on the findings, Dr. Schoenmakers underscored that “vitamin D deficiency is very common worldwide, [and] more should be done to develop strategies to address the needs of those groups and populations that are at risk of the consequences of vitamin D deficiency.”
That said, the D-Health study is important in helping to distinguish when supplementation may – and may not – be of benefit, she noted.
“This and other research in the past 15 years have contributed to our understanding [of] what the ranges of vitamin D status are [in which] health consequences may be anticipated.”
The D-Health Trial was funded by the National Health and Medical Research Council. Dr. Neale and Dr. Schoenmakers have reported no relevant financial relationships.
version of this article first appeared on Medscape.com.
, including mortality linked to cardiovascular disease, new results from a large, placebo-controlled trial show.
“The take-home message is that routine vitamin D supplementation, irrespective of the dosing regimen, is unlikely to be beneficial in a population with a low prevalence of vitamin D deficiency,” first author Rachel E. Neale, PhD, of the Population Health Department, QIMR Berghofer Medical Research Institute, in Brisbane, Australia, told this news organization.
Despite extensive previous research on vitamin D supplementation, “mortality has not been the primary outcome in any previous large trial of high-dose vitamin D supplementation,” Dr. Neale and coauthors noted. The results, published online in Lancet Diabetes & Endocrinology, are from the D-Health trial.
With more than 20,000 participants, this is the largest intermittent-dosing trial to date, the authors noted. The primary outcome was all-cause mortality.
In an accompanying editorial, Inez Schoenmakers, PhD, noted that “the findings [are] highly relevant for population policy, owing to the study’s population-based design, large scale, and long duration.”
This new “research contributes to the concept that improving vitamin D status with supplementation in a mostly vitamin D-replete older population does not influence all-cause mortality,” Dr. Schoenmakers, of the Faculty of Medicine and Health Sciences, University of East Anglia, Norwich, England, said in an interview.
“This is not dissimilar to research with many other nutrients showing that increasing intake above the adequate intake has no further health benefits,” she added.
D-Health Trial
The D-Health Trial involved 21,315 participants in Australia, enrolled between February 2014 and June 2015, who had not been screened for vitamin D deficiency but were largely considered to be vitamin D replete. They were a mean age of 69.3 years and 54% were men.
Participants were randomized 1:1 to a once-monthly oral vitamin D3 supplementation of 60,000 IU (n = 10,662) or a placebo capsule (n = 10,653).
They were permitted to take up to 2,000 IU/day of supplemental vitamin D in addition to the study protocol and had no history of kidney stones, hypercalcemia, hyperparathyroidism, osteomalacia, or sarcoidosis.
Over a median follow-up of 5.7 years, there were 1,100 deaths: 562 in the vitamin D group (5.3%) and 538 in the placebo group (5.1%). With a hazard ratio (HR) for all-cause mortality of 1.04, the difference was not significant (P = .47).
There were also no significant differences in terms of mortality from cardiovascular disease (HR, 0.96; P = .77), cancer (HR, 1.15; P = .13), or other causes (HR, 0.83; P = .15).
Rates of total adverse events between the two groups, including hypercalcemia and kidney stones, were similar.
An exploratory analysis excluding the first 2 years of follow-up in fact showed a numerically higher hazard ratio for cancer mortality in the vitamin D group versus no supplementation (HR, 1.24; P = .05). However, the authors noted that the effect was “not apparent when the analysis was restricted to deaths that were coded by the study team and not officially coded.”
Nevertheless, “our findings, from a large study in an unscreened population, give pause to earlier reports that vitamin D supplements might reduce cancer mortality,” they underscored.
Retention and adherence in the study were high, each exceeding 80%. Although blood samples were not collected at baseline, samples from 3,943 randomly sampled participants during follow-up showed mean serum 25-hydroxy-vitamin D concentrations of 77 nmol/L in the placebo group and 115 nmol/L in the vitamin D group, both within the normal range of 50-125 nmol/L.
Findings supported by previous research
The trial results are consistent with those of prior large studies and meta-analyses of older adults with a low prevalence of vitamin D deficiency showing that vitamin D3 supplementation, regardless of whether taken daily or monthly, is not likely to have an effect on all-cause mortality.
In the US VITAL trial, recently published in the New England Journal of Medicine, among 25,871 participants administered 2,000 IU/day of vitamin D3 for a median of 5.3 years, there was no reduction in all-cause mortality.
The ViDA trial of 5,110 older adults in New Zealand, published in 2019 in the Journal of Endocrinological Investigation, also showed monthly vitamin D3 supplementation of 100,000 IU for a median of 3.3 years was not associated with a benefit in people who were not deficient.
“In total, the results from the large trials and meta-analyses suggest that routine supplementation of older adults in populations with a low prevalence of vitamin D deficiency is unlikely to reduce the rate of all-cause mortality,” Dr. Neale and colleagues concluded.
Longer-term supplementation beneficial?
The population was limited to older adults and the study had a relatively short follow-up period, which Dr. Neale noted was necessary for pragmatic reasons.
“Our primary outcome was all-cause mortality, so to have sufficient deaths we either needed to study older adults or a much larger sample of younger adults,” she explained.
“However, we felt that [the former] ... had biological justification, as there is evidence that vitamin D plays a role later in the course of a number of diseases, with potential impacts on mortality.”
She noted that recent studies evaluating genetically predicted concentrations of serum 25(OH)D have further shown no link between those levels and all-cause mortality, stroke, or coronary heart disease.
“This confirms the statement that vitamin D is unlikely to be beneficial in people who are not vitamin D deficient, irrespective of whether supplementation occurs over the short or longer term,” Dr. Neale said.
The source of vitamin D, itself, is another consideration, with ongoing speculation of differences in benefits between dietary or supplementation sources versus sunlight exposure.
“Exposure to ultraviolet radiation, for which serum 25(OH)D concentration is a good marker, might confer benefits not mediated by vitamin D,” Dr. Neale and coauthors noted.
They added that the results in the older Australian population “cannot be generalized to populations with a higher prevalence of vitamin D deficiency, or with a greater proportion of people not of White ancestry, than the study population.”
Ten-year mortality rates from the D-Health trial are expected to be reported in the future.
Strategies still needed to address vitamin D deficiency
Further commenting on the findings, Dr. Schoenmakers underscored that “vitamin D deficiency is very common worldwide, [and] more should be done to develop strategies to address the needs of those groups and populations that are at risk of the consequences of vitamin D deficiency.”
That said, the D-Health study is important in helping to distinguish when supplementation may – and may not – be of benefit, she noted.
“This and other research in the past 15 years have contributed to our understanding [of] what the ranges of vitamin D status are [in which] health consequences may be anticipated.”
The D-Health Trial was funded by the National Health and Medical Research Council. Dr. Neale and Dr. Schoenmakers have reported no relevant financial relationships.
version of this article first appeared on Medscape.com.
, including mortality linked to cardiovascular disease, new results from a large, placebo-controlled trial show.
“The take-home message is that routine vitamin D supplementation, irrespective of the dosing regimen, is unlikely to be beneficial in a population with a low prevalence of vitamin D deficiency,” first author Rachel E. Neale, PhD, of the Population Health Department, QIMR Berghofer Medical Research Institute, in Brisbane, Australia, told this news organization.
Despite extensive previous research on vitamin D supplementation, “mortality has not been the primary outcome in any previous large trial of high-dose vitamin D supplementation,” Dr. Neale and coauthors noted. The results, published online in Lancet Diabetes & Endocrinology, are from the D-Health trial.
With more than 20,000 participants, this is the largest intermittent-dosing trial to date, the authors noted. The primary outcome was all-cause mortality.
In an accompanying editorial, Inez Schoenmakers, PhD, noted that “the findings [are] highly relevant for population policy, owing to the study’s population-based design, large scale, and long duration.”
This new “research contributes to the concept that improving vitamin D status with supplementation in a mostly vitamin D-replete older population does not influence all-cause mortality,” Dr. Schoenmakers, of the Faculty of Medicine and Health Sciences, University of East Anglia, Norwich, England, said in an interview.
“This is not dissimilar to research with many other nutrients showing that increasing intake above the adequate intake has no further health benefits,” she added.
D-Health Trial
The D-Health Trial involved 21,315 participants in Australia, enrolled between February 2014 and June 2015, who had not been screened for vitamin D deficiency but were largely considered to be vitamin D replete. They were a mean age of 69.3 years and 54% were men.
Participants were randomized 1:1 to a once-monthly oral vitamin D3 supplementation of 60,000 IU (n = 10,662) or a placebo capsule (n = 10,653).
They were permitted to take up to 2,000 IU/day of supplemental vitamin D in addition to the study protocol and had no history of kidney stones, hypercalcemia, hyperparathyroidism, osteomalacia, or sarcoidosis.
Over a median follow-up of 5.7 years, there were 1,100 deaths: 562 in the vitamin D group (5.3%) and 538 in the placebo group (5.1%). With a hazard ratio (HR) for all-cause mortality of 1.04, the difference was not significant (P = .47).
There were also no significant differences in terms of mortality from cardiovascular disease (HR, 0.96; P = .77), cancer (HR, 1.15; P = .13), or other causes (HR, 0.83; P = .15).
Rates of total adverse events between the two groups, including hypercalcemia and kidney stones, were similar.
An exploratory analysis excluding the first 2 years of follow-up in fact showed a numerically higher hazard ratio for cancer mortality in the vitamin D group versus no supplementation (HR, 1.24; P = .05). However, the authors noted that the effect was “not apparent when the analysis was restricted to deaths that were coded by the study team and not officially coded.”
Nevertheless, “our findings, from a large study in an unscreened population, give pause to earlier reports that vitamin D supplements might reduce cancer mortality,” they underscored.
Retention and adherence in the study were high, each exceeding 80%. Although blood samples were not collected at baseline, samples from 3,943 randomly sampled participants during follow-up showed mean serum 25-hydroxy-vitamin D concentrations of 77 nmol/L in the placebo group and 115 nmol/L in the vitamin D group, both within the normal range of 50-125 nmol/L.
Findings supported by previous research
The trial results are consistent with those of prior large studies and meta-analyses of older adults with a low prevalence of vitamin D deficiency showing that vitamin D3 supplementation, regardless of whether taken daily or monthly, is not likely to have an effect on all-cause mortality.
In the US VITAL trial, recently published in the New England Journal of Medicine, among 25,871 participants administered 2,000 IU/day of vitamin D3 for a median of 5.3 years, there was no reduction in all-cause mortality.
The ViDA trial of 5,110 older adults in New Zealand, published in 2019 in the Journal of Endocrinological Investigation, also showed monthly vitamin D3 supplementation of 100,000 IU for a median of 3.3 years was not associated with a benefit in people who were not deficient.
“In total, the results from the large trials and meta-analyses suggest that routine supplementation of older adults in populations with a low prevalence of vitamin D deficiency is unlikely to reduce the rate of all-cause mortality,” Dr. Neale and colleagues concluded.
Longer-term supplementation beneficial?
The population was limited to older adults and the study had a relatively short follow-up period, which Dr. Neale noted was necessary for pragmatic reasons.
“Our primary outcome was all-cause mortality, so to have sufficient deaths we either needed to study older adults or a much larger sample of younger adults,” she explained.
“However, we felt that [the former] ... had biological justification, as there is evidence that vitamin D plays a role later in the course of a number of diseases, with potential impacts on mortality.”
She noted that recent studies evaluating genetically predicted concentrations of serum 25(OH)D have further shown no link between those levels and all-cause mortality, stroke, or coronary heart disease.
“This confirms the statement that vitamin D is unlikely to be beneficial in people who are not vitamin D deficient, irrespective of whether supplementation occurs over the short or longer term,” Dr. Neale said.
The source of vitamin D, itself, is another consideration, with ongoing speculation of differences in benefits between dietary or supplementation sources versus sunlight exposure.
“Exposure to ultraviolet radiation, for which serum 25(OH)D concentration is a good marker, might confer benefits not mediated by vitamin D,” Dr. Neale and coauthors noted.
They added that the results in the older Australian population “cannot be generalized to populations with a higher prevalence of vitamin D deficiency, or with a greater proportion of people not of White ancestry, than the study population.”
Ten-year mortality rates from the D-Health trial are expected to be reported in the future.
Strategies still needed to address vitamin D deficiency
Further commenting on the findings, Dr. Schoenmakers underscored that “vitamin D deficiency is very common worldwide, [and] more should be done to develop strategies to address the needs of those groups and populations that are at risk of the consequences of vitamin D deficiency.”
That said, the D-Health study is important in helping to distinguish when supplementation may – and may not – be of benefit, she noted.
“This and other research in the past 15 years have contributed to our understanding [of] what the ranges of vitamin D status are [in which] health consequences may be anticipated.”
The D-Health Trial was funded by the National Health and Medical Research Council. Dr. Neale and Dr. Schoenmakers have reported no relevant financial relationships.
version of this article first appeared on Medscape.com.
FROM THE LANCET DIABETES & ENDOCRINOLOGY
Omicron subvariant 1.5 times more contagious than Omicron
according to CNBC.
The Statens Serum Institut, which monitors infectious diseases in Denmark, said that BA.2 is more contagious, but it doesn’t appear to increase hospitalizations or reduce how well the vaccine works.
BA.2 overtook BA.1 as the primary variant in Denmark within a few weeks, Troels Lillebaek, director of the institute, told CNBC. The subvariant has five unique mutations on a key part of the spike protein, which is what the coronavirus uses to invade human cells. This often means a higher rate of spreading.
The Omicron subvariant has been detected in at least 29 states in the United States and 56 countries, according to the latest update from Outbreak.info. The United States has detected 188 infections, with the worldwide total nearing 25,000.
Denmark has reported the highest number of cases, followed by the United Kingdom and India. Both Denmark and India have reported that BA.2 now accounts for about half of new COVID-19 cases in those countries.
On Jan. 28, the U.K. Health Security Agency said BA.2 has a “substantial” growth advantage over the original Omicron strain. The subvariant has spread faster in all regions of England where there were enough cases to conduct an analysis, the agency said in a report.
A preliminary evaluation found that BA.2 doesn’t appear to change how well the vaccine works compared to the original Omicron strain, the agency said. A booster dose was 70% effective at preventing symptomatic illness for BA.2, compared with 63% for the original Omicron strain.
The Centers for Disease Control and Prevention also said on Jan. 28 that, although the subvariant has become more common in some countries, it is currently at a low level in the United States and doesn’t appear to be more serious.
“Currently there is no evidence that the BA.2 lineage is more severe than the BA.1 lineage,” Kristen Nordlund, a CDC spokesperson, told CNBC.
The World Health Organization hasn’t labeled BA.2 a “variant of concern” so far but will continue to monitor it. WHO officials have said that new variants will arise as Omicron spreads across the world.
“The next variant of concern will be more fit, and what we mean by that is it will be more transmissible because it will have to overtake what is currently circulating,” Maria Van Kerkhove, the WHO’s COVID-19 technical lead, said during a livestream on Jan. 25.
“The big question is whether or not future variants will be more or less severe,” she said.
A version of this article first appeared on WebMD.com.
according to CNBC.
The Statens Serum Institut, which monitors infectious diseases in Denmark, said that BA.2 is more contagious, but it doesn’t appear to increase hospitalizations or reduce how well the vaccine works.
BA.2 overtook BA.1 as the primary variant in Denmark within a few weeks, Troels Lillebaek, director of the institute, told CNBC. The subvariant has five unique mutations on a key part of the spike protein, which is what the coronavirus uses to invade human cells. This often means a higher rate of spreading.
The Omicron subvariant has been detected in at least 29 states in the United States and 56 countries, according to the latest update from Outbreak.info. The United States has detected 188 infections, with the worldwide total nearing 25,000.
Denmark has reported the highest number of cases, followed by the United Kingdom and India. Both Denmark and India have reported that BA.2 now accounts for about half of new COVID-19 cases in those countries.
On Jan. 28, the U.K. Health Security Agency said BA.2 has a “substantial” growth advantage over the original Omicron strain. The subvariant has spread faster in all regions of England where there were enough cases to conduct an analysis, the agency said in a report.
A preliminary evaluation found that BA.2 doesn’t appear to change how well the vaccine works compared to the original Omicron strain, the agency said. A booster dose was 70% effective at preventing symptomatic illness for BA.2, compared with 63% for the original Omicron strain.
The Centers for Disease Control and Prevention also said on Jan. 28 that, although the subvariant has become more common in some countries, it is currently at a low level in the United States and doesn’t appear to be more serious.
“Currently there is no evidence that the BA.2 lineage is more severe than the BA.1 lineage,” Kristen Nordlund, a CDC spokesperson, told CNBC.
The World Health Organization hasn’t labeled BA.2 a “variant of concern” so far but will continue to monitor it. WHO officials have said that new variants will arise as Omicron spreads across the world.
“The next variant of concern will be more fit, and what we mean by that is it will be more transmissible because it will have to overtake what is currently circulating,” Maria Van Kerkhove, the WHO’s COVID-19 technical lead, said during a livestream on Jan. 25.
“The big question is whether or not future variants will be more or less severe,” she said.
A version of this article first appeared on WebMD.com.
according to CNBC.
The Statens Serum Institut, which monitors infectious diseases in Denmark, said that BA.2 is more contagious, but it doesn’t appear to increase hospitalizations or reduce how well the vaccine works.
BA.2 overtook BA.1 as the primary variant in Denmark within a few weeks, Troels Lillebaek, director of the institute, told CNBC. The subvariant has five unique mutations on a key part of the spike protein, which is what the coronavirus uses to invade human cells. This often means a higher rate of spreading.
The Omicron subvariant has been detected in at least 29 states in the United States and 56 countries, according to the latest update from Outbreak.info. The United States has detected 188 infections, with the worldwide total nearing 25,000.
Denmark has reported the highest number of cases, followed by the United Kingdom and India. Both Denmark and India have reported that BA.2 now accounts for about half of new COVID-19 cases in those countries.
On Jan. 28, the U.K. Health Security Agency said BA.2 has a “substantial” growth advantage over the original Omicron strain. The subvariant has spread faster in all regions of England where there were enough cases to conduct an analysis, the agency said in a report.
A preliminary evaluation found that BA.2 doesn’t appear to change how well the vaccine works compared to the original Omicron strain, the agency said. A booster dose was 70% effective at preventing symptomatic illness for BA.2, compared with 63% for the original Omicron strain.
The Centers for Disease Control and Prevention also said on Jan. 28 that, although the subvariant has become more common in some countries, it is currently at a low level in the United States and doesn’t appear to be more serious.
“Currently there is no evidence that the BA.2 lineage is more severe than the BA.1 lineage,” Kristen Nordlund, a CDC spokesperson, told CNBC.
The World Health Organization hasn’t labeled BA.2 a “variant of concern” so far but will continue to monitor it. WHO officials have said that new variants will arise as Omicron spreads across the world.
“The next variant of concern will be more fit, and what we mean by that is it will be more transmissible because it will have to overtake what is currently circulating,” Maria Van Kerkhove, the WHO’s COVID-19 technical lead, said during a livestream on Jan. 25.
“The big question is whether or not future variants will be more or less severe,” she said.
A version of this article first appeared on WebMD.com.
Pandemic pushed death rates to historic highs
Excess mortality is a way of quantifying the impact of a pandemic, based on overall mortality from nonpandemic periods. Mortality data over long periods of time are not available for many countries, but Switzerland, Sweden, and Spain have accumulated death count data for an uninterrupted period of more than 100 years.
In a study published in the Annals of Internal Medicine, Kaspar Staub, PhD, of the University of Zurich led a team of researchers in reviewing data on monthly excess deaths from all causes for Switzerland, Sweden, and Spain for 2020 to 2021. Dr. Staub and colleagues also compared these numbers to other pandemic and nonpandemic periods since the end of the 19th century. The starting years were 1877 for Switzerland, 1851 for Sweden, and 1908 for Spain.
The researchers collected data for monthly all-cause deaths from the statistical offices of each country and determined excess mortality by comparing these numbers to population size and age structure.
They found that 2020 showed the highest number of excess deaths since 1918, with relative excess of deaths of 12.5% in Switzerland, 8.5% in Sweden, and 17.3 % in Spain.
To put it another way, the number of excess deaths per 100,000 people was 100 for Switzerland, 75 for Sweden, and 155 for Spain.
“Our findings suggest that the pandemic led to the second-largest mortality disaster driven by a viral infection in more than 100 years in the three countries we studied, second only to the 1918 influenza pandemic,” the researchers wrote.
They explained that the excess mortality for the year 1918 was six to seven times higher than the 2020 numbers, but that the 2020 numbers might have been higher without the strong public health interventions taken worldwide to mitigate the impact of the COVID-19 pandemic.
“Early estimates suggest that vaccination prevented approximately 470,000 deaths in persons aged 60 years or older across 33 European countries between December 2019 and November 2021,” they wrote. However, because the COVID-19 pandemic is ongoing, “a more conclusive assessment will have to wait,” they added.
The 2020 numbers also were higher than most mortality rates since 1918, including peak years of previous influenza pandemics that occurred in 1957, 1968, 1977, and, most recently, the swine flu pandemic of 2009 which was caused by a novel strain of the H1N1 influenza virus.
The study findings had some limitations. For example, only three countries were included. Also, monthly death numbers according to sex, age, and cause of death were available only for the past 60 years, and data from years before the 20th century may not be reliable, the researchers said.
The new study does not account for the long-term effects of patients suffering from long COVID, they noted.
Study findings support strong public health response
“With the COVID-19 pandemic ongoing, this study reinforces the historic magnitude of the problem in terms of mortality and could add to the justification for ongoing public health measures such as vaccination drives and vaccine mandates to curb deaths,” said Suman Pal, MD, an internal medicine physician at the University of New Mexico, Albuquerque, in an interview.
“The results are surprising because when we view the rapid advancement in medical science over the last few decades, which have led to a decline in mortality from many previously fatal diseases, the scale of excess mortality from COVID-19 seems to have offset many such gains in the past 2 years.”
Prior studies of United States mortality data have estimated that excess deaths in the United States in 2020 exceeded the deaths attributed to COVID-19, said Dr. Pal. “The findings of this study could help clinicians in their discussion of the need for COVID-19 prevention measures with their patients” and inform discussions between doctors and patients about prevention strategies, he explained.
“Emphasizing that this pandemic is the second-largest cause of death due to a viral infection in a century could help patients understand the need for public health measures that may be viewed as unprecedented, such as government-imposed lockdowns, contact tracing, mask requirements, restrictions on travel, and vaccine mandates,” Dr. Pal noted. Better understanding of the evidence behind such measures may decrease the public’s resistance to following them, he added.As for additional research, “region-specific analysis of excess deaths may help estimate the impact of COVID-19 better, especially in regions where data reporting may be unreliable.”
Dr. F. Perry Wilson's take on study
“All-cause mortality is a key metric to assess the impact of the pandemic, because each death is treated equally,” said F. Perry Wilson, MD, of Yale University, in an interview. “With this type of analysis, there is no vague definition of a death from COVID or with COVID,” he explained. “A death is a death, and more deaths than expected is, of course, a bad thing. These analyses give a high-level view of the true human cost of the pandemic,” he said.
Dr. Wilson said he was not surprised by the findings. “There have been multiple studies, across multiple countries including the United States, which show similar findings—that observed deaths during this pandemic are substantially higher than expected,” he said. The current study findings are unique in that they compare the current pandemic to death rates in a nearly unbroken chain into the last century using data that only a few countries can provide, he noted.
The mortality data are “quite similar to what we see in the United States, with the exception that Spain was particularly hard-hit in the first COVID-19 wave in April 2020, said Dr. Wilson. By contrast, “the U.S. had substantially more excess deaths in the recent Delta wave, presumably due to lower vaccination uptake,” he added.
The current study is important for clinicians and their patients, said Dr. Wilson. “Data like these can help cut through some of the misinformation, such as the idea that only people who would have died anyway die of COVID, or that COVID is not severe,” he emphasized. “Overall death data are quite clear that far more people, millions more people, died over the last 22 months than could possibly be explained except by a global-level mortality event,” he said.
“One thing this study reminds us of is the value of high-quality data,” said Dr. Wilson. “Few countries have near complete vital statistics records on their entire populations and these can be so crucial to understand the true impact of pandemics and other disasters,” he explained. Of course, mortality data also serve as a reminder “that COVID is a serious disease: a once-in-a-century (we hope) pandemic,” he added.
The current study showed that excess death rates were similar, but not the same, from country to country, Dr. Wilson noted. “Moving forward, we need to learn what factors, from vaccination to social distancing strategies,” saved lives around the world,” he said.
The study was supported by the Foundation for Research in Science and the Humanities at the University of Zurich, the Swiss National Science Foundation, and the U.S. National Institute of Allergy and Infectious Diseases. The researchers, Dr. Pal, and Dr. Wilson had no financial conflicts.
*This article was updated on 2/1/2022.
Excess mortality is a way of quantifying the impact of a pandemic, based on overall mortality from nonpandemic periods. Mortality data over long periods of time are not available for many countries, but Switzerland, Sweden, and Spain have accumulated death count data for an uninterrupted period of more than 100 years.
In a study published in the Annals of Internal Medicine, Kaspar Staub, PhD, of the University of Zurich led a team of researchers in reviewing data on monthly excess deaths from all causes for Switzerland, Sweden, and Spain for 2020 to 2021. Dr. Staub and colleagues also compared these numbers to other pandemic and nonpandemic periods since the end of the 19th century. The starting years were 1877 for Switzerland, 1851 for Sweden, and 1908 for Spain.
The researchers collected data for monthly all-cause deaths from the statistical offices of each country and determined excess mortality by comparing these numbers to population size and age structure.
They found that 2020 showed the highest number of excess deaths since 1918, with relative excess of deaths of 12.5% in Switzerland, 8.5% in Sweden, and 17.3 % in Spain.
To put it another way, the number of excess deaths per 100,000 people was 100 for Switzerland, 75 for Sweden, and 155 for Spain.
“Our findings suggest that the pandemic led to the second-largest mortality disaster driven by a viral infection in more than 100 years in the three countries we studied, second only to the 1918 influenza pandemic,” the researchers wrote.
They explained that the excess mortality for the year 1918 was six to seven times higher than the 2020 numbers, but that the 2020 numbers might have been higher without the strong public health interventions taken worldwide to mitigate the impact of the COVID-19 pandemic.
“Early estimates suggest that vaccination prevented approximately 470,000 deaths in persons aged 60 years or older across 33 European countries between December 2019 and November 2021,” they wrote. However, because the COVID-19 pandemic is ongoing, “a more conclusive assessment will have to wait,” they added.
The 2020 numbers also were higher than most mortality rates since 1918, including peak years of previous influenza pandemics that occurred in 1957, 1968, 1977, and, most recently, the swine flu pandemic of 2009 which was caused by a novel strain of the H1N1 influenza virus.
The study findings had some limitations. For example, only three countries were included. Also, monthly death numbers according to sex, age, and cause of death were available only for the past 60 years, and data from years before the 20th century may not be reliable, the researchers said.
The new study does not account for the long-term effects of patients suffering from long COVID, they noted.
Study findings support strong public health response
“With the COVID-19 pandemic ongoing, this study reinforces the historic magnitude of the problem in terms of mortality and could add to the justification for ongoing public health measures such as vaccination drives and vaccine mandates to curb deaths,” said Suman Pal, MD, an internal medicine physician at the University of New Mexico, Albuquerque, in an interview.
“The results are surprising because when we view the rapid advancement in medical science over the last few decades, which have led to a decline in mortality from many previously fatal diseases, the scale of excess mortality from COVID-19 seems to have offset many such gains in the past 2 years.”
Prior studies of United States mortality data have estimated that excess deaths in the United States in 2020 exceeded the deaths attributed to COVID-19, said Dr. Pal. “The findings of this study could help clinicians in their discussion of the need for COVID-19 prevention measures with their patients” and inform discussions between doctors and patients about prevention strategies, he explained.
“Emphasizing that this pandemic is the second-largest cause of death due to a viral infection in a century could help patients understand the need for public health measures that may be viewed as unprecedented, such as government-imposed lockdowns, contact tracing, mask requirements, restrictions on travel, and vaccine mandates,” Dr. Pal noted. Better understanding of the evidence behind such measures may decrease the public’s resistance to following them, he added.As for additional research, “region-specific analysis of excess deaths may help estimate the impact of COVID-19 better, especially in regions where data reporting may be unreliable.”
Dr. F. Perry Wilson's take on study
“All-cause mortality is a key metric to assess the impact of the pandemic, because each death is treated equally,” said F. Perry Wilson, MD, of Yale University, in an interview. “With this type of analysis, there is no vague definition of a death from COVID or with COVID,” he explained. “A death is a death, and more deaths than expected is, of course, a bad thing. These analyses give a high-level view of the true human cost of the pandemic,” he said.
Dr. Wilson said he was not surprised by the findings. “There have been multiple studies, across multiple countries including the United States, which show similar findings—that observed deaths during this pandemic are substantially higher than expected,” he said. The current study findings are unique in that they compare the current pandemic to death rates in a nearly unbroken chain into the last century using data that only a few countries can provide, he noted.
The mortality data are “quite similar to what we see in the United States, with the exception that Spain was particularly hard-hit in the first COVID-19 wave in April 2020, said Dr. Wilson. By contrast, “the U.S. had substantially more excess deaths in the recent Delta wave, presumably due to lower vaccination uptake,” he added.
The current study is important for clinicians and their patients, said Dr. Wilson. “Data like these can help cut through some of the misinformation, such as the idea that only people who would have died anyway die of COVID, or that COVID is not severe,” he emphasized. “Overall death data are quite clear that far more people, millions more people, died over the last 22 months than could possibly be explained except by a global-level mortality event,” he said.
“One thing this study reminds us of is the value of high-quality data,” said Dr. Wilson. “Few countries have near complete vital statistics records on their entire populations and these can be so crucial to understand the true impact of pandemics and other disasters,” he explained. Of course, mortality data also serve as a reminder “that COVID is a serious disease: a once-in-a-century (we hope) pandemic,” he added.
The current study showed that excess death rates were similar, but not the same, from country to country, Dr. Wilson noted. “Moving forward, we need to learn what factors, from vaccination to social distancing strategies,” saved lives around the world,” he said.
The study was supported by the Foundation for Research in Science and the Humanities at the University of Zurich, the Swiss National Science Foundation, and the U.S. National Institute of Allergy and Infectious Diseases. The researchers, Dr. Pal, and Dr. Wilson had no financial conflicts.
*This article was updated on 2/1/2022.
Excess mortality is a way of quantifying the impact of a pandemic, based on overall mortality from nonpandemic periods. Mortality data over long periods of time are not available for many countries, but Switzerland, Sweden, and Spain have accumulated death count data for an uninterrupted period of more than 100 years.
In a study published in the Annals of Internal Medicine, Kaspar Staub, PhD, of the University of Zurich led a team of researchers in reviewing data on monthly excess deaths from all causes for Switzerland, Sweden, and Spain for 2020 to 2021. Dr. Staub and colleagues also compared these numbers to other pandemic and nonpandemic periods since the end of the 19th century. The starting years were 1877 for Switzerland, 1851 for Sweden, and 1908 for Spain.
The researchers collected data for monthly all-cause deaths from the statistical offices of each country and determined excess mortality by comparing these numbers to population size and age structure.
They found that 2020 showed the highest number of excess deaths since 1918, with relative excess of deaths of 12.5% in Switzerland, 8.5% in Sweden, and 17.3 % in Spain.
To put it another way, the number of excess deaths per 100,000 people was 100 for Switzerland, 75 for Sweden, and 155 for Spain.
“Our findings suggest that the pandemic led to the second-largest mortality disaster driven by a viral infection in more than 100 years in the three countries we studied, second only to the 1918 influenza pandemic,” the researchers wrote.
They explained that the excess mortality for the year 1918 was six to seven times higher than the 2020 numbers, but that the 2020 numbers might have been higher without the strong public health interventions taken worldwide to mitigate the impact of the COVID-19 pandemic.
“Early estimates suggest that vaccination prevented approximately 470,000 deaths in persons aged 60 years or older across 33 European countries between December 2019 and November 2021,” they wrote. However, because the COVID-19 pandemic is ongoing, “a more conclusive assessment will have to wait,” they added.
The 2020 numbers also were higher than most mortality rates since 1918, including peak years of previous influenza pandemics that occurred in 1957, 1968, 1977, and, most recently, the swine flu pandemic of 2009 which was caused by a novel strain of the H1N1 influenza virus.
The study findings had some limitations. For example, only three countries were included. Also, monthly death numbers according to sex, age, and cause of death were available only for the past 60 years, and data from years before the 20th century may not be reliable, the researchers said.
The new study does not account for the long-term effects of patients suffering from long COVID, they noted.
Study findings support strong public health response
“With the COVID-19 pandemic ongoing, this study reinforces the historic magnitude of the problem in terms of mortality and could add to the justification for ongoing public health measures such as vaccination drives and vaccine mandates to curb deaths,” said Suman Pal, MD, an internal medicine physician at the University of New Mexico, Albuquerque, in an interview.
“The results are surprising because when we view the rapid advancement in medical science over the last few decades, which have led to a decline in mortality from many previously fatal diseases, the scale of excess mortality from COVID-19 seems to have offset many such gains in the past 2 years.”
Prior studies of United States mortality data have estimated that excess deaths in the United States in 2020 exceeded the deaths attributed to COVID-19, said Dr. Pal. “The findings of this study could help clinicians in their discussion of the need for COVID-19 prevention measures with their patients” and inform discussions between doctors and patients about prevention strategies, he explained.
“Emphasizing that this pandemic is the second-largest cause of death due to a viral infection in a century could help patients understand the need for public health measures that may be viewed as unprecedented, such as government-imposed lockdowns, contact tracing, mask requirements, restrictions on travel, and vaccine mandates,” Dr. Pal noted. Better understanding of the evidence behind such measures may decrease the public’s resistance to following them, he added.As for additional research, “region-specific analysis of excess deaths may help estimate the impact of COVID-19 better, especially in regions where data reporting may be unreliable.”
Dr. F. Perry Wilson's take on study
“All-cause mortality is a key metric to assess the impact of the pandemic, because each death is treated equally,” said F. Perry Wilson, MD, of Yale University, in an interview. “With this type of analysis, there is no vague definition of a death from COVID or with COVID,” he explained. “A death is a death, and more deaths than expected is, of course, a bad thing. These analyses give a high-level view of the true human cost of the pandemic,” he said.
Dr. Wilson said he was not surprised by the findings. “There have been multiple studies, across multiple countries including the United States, which show similar findings—that observed deaths during this pandemic are substantially higher than expected,” he said. The current study findings are unique in that they compare the current pandemic to death rates in a nearly unbroken chain into the last century using data that only a few countries can provide, he noted.
The mortality data are “quite similar to what we see in the United States, with the exception that Spain was particularly hard-hit in the first COVID-19 wave in April 2020, said Dr. Wilson. By contrast, “the U.S. had substantially more excess deaths in the recent Delta wave, presumably due to lower vaccination uptake,” he added.
The current study is important for clinicians and their patients, said Dr. Wilson. “Data like these can help cut through some of the misinformation, such as the idea that only people who would have died anyway die of COVID, or that COVID is not severe,” he emphasized. “Overall death data are quite clear that far more people, millions more people, died over the last 22 months than could possibly be explained except by a global-level mortality event,” he said.
“One thing this study reminds us of is the value of high-quality data,” said Dr. Wilson. “Few countries have near complete vital statistics records on their entire populations and these can be so crucial to understand the true impact of pandemics and other disasters,” he explained. Of course, mortality data also serve as a reminder “that COVID is a serious disease: a once-in-a-century (we hope) pandemic,” he added.
The current study showed that excess death rates were similar, but not the same, from country to country, Dr. Wilson noted. “Moving forward, we need to learn what factors, from vaccination to social distancing strategies,” saved lives around the world,” he said.
The study was supported by the Foundation for Research in Science and the Humanities at the University of Zurich, the Swiss National Science Foundation, and the U.S. National Institute of Allergy and Infectious Diseases. The researchers, Dr. Pal, and Dr. Wilson had no financial conflicts.
*This article was updated on 2/1/2022.
FROM ANNALS OF INTERNAL MEDICINE