User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
Sulfites: The 2024 American Contact Dermatitis Society Allergen of the Year
The American Contact Dermatitis Society (ACDS) selected sulfites as the 2024 Allergen of the Year.1 Due to their preservative and antioxidant properties, sulfites are prevalent in a variety of foods, beverages, medications, and personal care products; however, sulfites also have been implicated as a potential contact allergen. In this article, we review common sources of sulfite exposure, clinical manifestations of allergic contact dermatitis (ACD) to sulfites, and patch testing considerations for this emerging allergen.
What Are Sulfites?
Sulfiting agents are compounds that contain the sulfite ion SO32-, including sulfur dioxide, sodium disulfite (sodium metabisulfite), and potassium metabisulfite.2 Sulfites occur naturally in the environment and commonly are used as preservatives, antibrowning agents, and antioxidants in various foods, beverages, medications, cosmetics, and skin care products. As antibrowning agents and antioxidants, sulfites help maintain the natural appearance of foods and other products and prevent premature spoiling by inactivating oxidative enzymes.3 It should be noted that sulfites and sulfates are distinct and unrelated compounds that do not cross-react.1
Common Sources of Sulfite Exposure
From a morning glass of juice to an evening shower, in the pharmacy and at the hair salon, sulfite exposure is ubiquitous in most daily routines. Sulfites are present in many foods and beverages, either as a byproduct of natural fermentation or as an additive to prevent spoiling and color change. The Table provides examples of foods with high sulfite content.1,4-6 In particular, dried fruit, bottled lemon juice, wine, grape juice, sauerkraut juice, and pickled onions have high sulfite content.

Topical medications and personal care products represent other potential sources of sulfite exposure. A number of reports have shown that sulfites may be included in topical steroids,7 antibiotics,8 antifungals,9 hemorrhoidal preparations,10 local anesthetics,11 and urinary catheterization gel,12 highlighting their many potential applications. In addition, a comprehensive ingredient analysis of 264 ophthalmic medications found that 3.8% of the products contained sodium disulfite.13 Sulfites may be found in personal care products, including facial and hand cleansers, shampoos, moisturizers, and toothpastes. Hair dyes also commonly contain sulfites,7 which are listed in as many as 90% of hair dye kits in the ACDS Contact Allergen Management Program database.1
Occupational exposures also are widespread, as sulfites are extensively utilized across diverse industries such as pharmaceuticals, health care, leather manufacturing, mineral extraction, food preparation, chemical manufacturing, textiles, alcohol brewing, and wine production.1
Sulfites also are used in the rubber industry—particularly in gloves—due to their anticoagulant and preservative properties.4 This is relevant to health care providers, who may use dozens of disposable gloves in a single day. In an experimental pilot study, researchers detected sulfites in 83% (5/6) of natural rubber latex gloves, 96% (23/24) of synthetic (nitrile) gloves, and 0% (0/5) of polyvinyl chloride gloves.14 While this study was limited to a small sample size, it demonstrates the common use of sulfites in certain rubber gloves and encourages future studies to determine whether there is a quantitative threshold to elicit allergic reactions.
Sulfite Allergy
In 1968, an early case report of ACD to sulfites was published involving a pharmaceutical worker who developed hand eczema after working at a factory for 3 months and had a positive patch test to potassium metabisulfite.15 There have been other cases published in the literature since then, including localized ACD as well as less common cases of systemic contact dermatitis following oral, injectable, and rectal sulfite exposures.16
The North American Contact Dermatitis Group found that, among 132 (2.7%) of 4885 patients with positive patch tests to sodium disulfite from 2017 to 2018, the most commonly involved body sites were the face (28.8%) and hands (20.5%) followed by a scattered/generalized distribution (13.6%). Involvement of the face and hands may correlate with the most frequent sources of exposure that were identified, including personal care products (particularly hair dyes)(18.9%), medications (9.1%), and foods (7.6%).17 A multicenter analysis of patch test results from Germany, Austria, and Switzerland from 1999 to 2013 showed that 357 (2.9%) of 12,156 patients had positive reactions to sodium disulfite, with the most commonly identified exposure sources being topical pharmaceutical agents (59.3%); cosmetics, creams, and sunscreens (13.6%); and systemic drugs (6.8%).18 However, it is not always possible to determine the clinical relevance of a positive patch test to sulfites.1
Other than the face and hands, there have been other unexpected anatomic locations for sulfite ACD (eg, the lower back), and systemic contact dermatitis has manifested with widespread rashes due to oral, rectal, and parenteral exposure.4,16,19 There is no definitive link between sulfite contact allergy and patient sex, but there seems to be a higher prevalence in patients older than 40 years, perhaps related to overall lifetime exposure.1
Immediate hypersensitivity reactions to sulfites also have been reported, including urticaria, angioedema, and anaphylaxis.4 Due to multiple cases of severe dermatologic and respiratory reactions to food products containing sulfites,20 the US Food and Drug Administration prohibited their use in fresh fruit and vegetables as antibrowning agents in 1986 and required labels on packaged foods that contained sulfites at more than 10 parts per million.21 However, food and drinks produced in restaurants, bakeries, and cafes as well as those that are distributed directly to consumers from the preparation site are exempt from these rules.17
In addition, consuming high amounts of dietary sulfites has been linked to headaches through unclear (ie, not necessarily allergic) mechanisms.4,22 One study found that wine with a higher sulfite concentration was associated with increased risk for headaches in participants who had a history of headaches related to wine consumption.22
Patch Testing to Sulfites
The North American Contact Dermatitis Group has tested sodium disulfite since 2017 and found an increased frequency of positive patch tests from 2.7% (N=4885) in 2017 and 201817 to 3.3% (N=4115) in 2019 and 202023 among patients referred for testing. Similarly, patch testing to sodium disulfite in nearly 40,000 patients in 9 European countries showed a pooled prevalence of reactions of 3.1%.17 However, this contact allergy may go unrecognized, as sulfites are not included in common patch test series, including the thin-layer rapid use epicutaneous test and the ACDS Core Allergen Series.24,25 The relatively high patch test positivity to sulfites along with the prevalence of daily exposures supports the addition of sulfites to more patch test screening series.
The recommended patch test concentration for sodium disulfite is 1% in petrolatum.5 Testing in aqueous solutions is not recommended because they can cause sulfites to break down, potentially producing false-positive or irritant patch test reactions.7,26,27
Recommendations for Patients With Sulfite Allergies
Individuals with contact allergies to sulfites should be counseled on exposure sources and should be given resources providing a list of safe products, such as the ACDS Contact Allergen Management Program (https://www.acdscamp.org/login) or SkinSAFE (https://www.skinsafeproducts.com/). Prescribers should be cognizant of sulfites that are present in prescription medications. Just because a patient has a positive patch test to sulfites does not automatically imply that they will need to modify their diet to avoid sulfite-containing foods; in the absence of cheilitis or a distribution suggestive of systemic contact dermatitis (eg, vesicular hand/foot dermatitis, intertriginous eruptions), this step may be unnecessary. On the other hand, individuals who have experienced immediate hypersensitivity reactions to sulfites should avoid sulfite-containing foods and carry an epinephrine autoinjector.
Final Interpretation
Sulfites are ubiquitous compounds found in various foods, beverages, medications, and personal care products in addition to a range of occupational exposures. The face and hands are the most common sites of sulfite ACD. Despite patch test positivity in as many as 3% of tested patients,17,23 sulfite allergy may be missed due to lack of routine testing on standard screening series.
- Ekstein SF, Warshaw EM. Sulfites: allergen of the year 2024. Dermatitis. 2024;35:6-12. doi:10.1089/derm.2023.0154
- Gunnison AF, Jacobsen DW. Sulfite hypersensitivity. a critical review. CRC Crit Rev Toxicol. 1987;17:185-214. doi:10.3109/10408448709071208
- Clough SR. Sodium sulfite. In: Wexler P, ed. Encyclopedia of Toxicology. 3rd ed. Academic Press; 2014: 341-343.
- Vally H, Misso NL, Madan V. Clinical effects of sulphite additives. Clin Exp Allergy. 2009;39:1643-1651. doi:10.1111/j.1365-2222.2009.03362.x
- Ralph N, Verma S, Merry S, et al. What is the relevance of contact allergy to sodium metabisulfite and which concentration of the allergen should we use? Dermatitis. 2015;26:162-165. doi:10.1097/der.0000000000000120
- Madan V, Walker SL, Beck MH. Sodium metabisulfite allergy is common but is it relevant? Contact Dermatitis. 2007;57:173-176. doi:10.1111/j.1600-0536.2007.01188.x
- García-Gavín J, Parente J, Goossens A. Allergic contact dermatitis caused by sodium metabisulfite: a challenging allergen. a case series and literature review. Contact Dermatitis. 2012;67:260-269. doi:10.1111/j.1600-0536.2012.02135.x
- Milpied B, van Wassenhove L, Larousse C, et al. Contact dermatitis from rifamycin. Contact Dermatitis. 1986;14:252-253. doi:10.1111/j.1600-0536.1986.tb01240.x
- Lodi A, Chiarelli G, Mancini LL, et al. Contact allergy to sodium sulfite contained in an antifungal preparation. Contact Dermatitis. 1993;29:97. doi:10.1111/j.1600-0536.1993.tb03493.x
- Sánchez-Pérez J, Abajo P, Córdoba S, et al. Allergic contact dermatitis from sodium metabisulfite in an antihemorrhoidal cream. Contact Dermatitis. 2000;42:176-177.
- Boyd AH, Warshaw EM. Sulfites: no longer a zebra? Dermatitis. 2017;28:364-366. doi:10.1097/der.0000000000000312
- Grosch E, Mahler V. Allergic contact dermatitis caused by a catheter system containing sodium metabisulfite. Contact Dermatitis. 2017;76:186-187. doi:10.1111/cod.12675
- Shaver RL, Warshaw EM. Contact allergens in prescription topical ophthalmic medications. Dermatitis. 2022;33:135-143. doi:10.1097/der.0000000000000751
- Dendooven E, Darrigade AS, Foubert K, et al. The presence of sulfites in ‘natural rubber latex’ and ‘synthetic’ rubber gloves: an experimental pilot study. Br J Dermatol. 2020;182:1054-1055. doi:10.1111/bjd.18608
- Nater JP. Allergic contact dermatitis caused by potassium metabisulfite. Dermatologica. 1968;136:477-478. doi:10.1159/000254143
- Borges AS, Valejo Coelho MM, Fernandes C, et al. Systemic allergic dermatitis caused by sodium metabisulfite in rectal enemas. Contact Dermatitis. 2018;78:429-430. doi:10.1111/cod.12971
- Warshaw EM, Buonomo M, DeKoven JG, et al. Patch testing with sodium disulfite: North American Contact Dermatitis Group experience, 2017 to 2018. Contact Dermatitis. 2021;85:285-296. doi:10.1111/cod.13860
- Häberle M, Geier J, Mahler V. Contact allergy to sulfites: clinical and occupational relevance—new data from the German Contact Dermatitis Research Group and the Information Network of Departments of Dermatology (IVDK). J Dtsch Dermatol Ges. 2016;14:938-941. doi:10.1111/ddg.13009
- Tan MG, Li HO, Pratt MD. Systemic allergic dermatitis to sodium metabisulfite in local anesthetic solution. Contact Dermatitis. 2022;86:120-121. doi:10.1111/cod.13978
- D’Amore T, Di Taranto A, Berardi G, et al. Sulfites in meat: occurrence, activity, toxicity, regulation, and detection. a comprehensive review. Compr Rev Food Sci Food Saf. 2020;19:2701-2720. doi:10.1111/1541-4337.12607
- Grotheer P, Marshall M, Simonne A. Sulfites: separating fact from fiction. May 11, 2022. UF IFAS Extension. University of Florida. Accessed October 4, 2024. https://edis.ifas.ufl.edu/publication/FY731
- Silva M, Gama J, Pinto N, et al. Sulfite concentration and the occurrence of headache in young adults: a prospective study. Eur J Clin Nutr. 2019;73:1316-1322. doi:10.1038/s41430-019-0420-2
- DeKoven JG, Warshaw EM, Reeder MJ, et al. North American Contact Dermatitis Group patch test results: 2019-2020. Dermatitis. 2023;34:90-104. doi:10.1089/derm.2022.29017.jdk
- T.R.U.E. Test. Thin-layer rapid use epicutaneous patch test. SmartPractice Dermatology Allergy. Accessed October 4, 2024. https://www.smartpractice.com/shop/category?id=581719&m=SPA
- Schalock PC, Dunnick CA, Nedorost, et al; American Contact Dermatitis Society Core Allergen Series Committee. American Contact Dermatitis Society Core Allergen Series: 2020 update. Dermatitis. 2020;31:279-282.
- Kaaman AC, Boman A, Wrangsjö K, et al. Contact allergy to sodium metabisulfite: an occupational problem. Contact Dermatitis. 2010;63:110-112. doi:10.1111/j.1600-0536.2010.01756.x
- Vena GA, Foti C, Angelini G. Sulfite contact allergy. Contact Dermatitis. 1994;31:172-175. doi:10.1111/j.1600-0536.1994.tb01959.x
The American Contact Dermatitis Society (ACDS) selected sulfites as the 2024 Allergen of the Year.1 Due to their preservative and antioxidant properties, sulfites are prevalent in a variety of foods, beverages, medications, and personal care products; however, sulfites also have been implicated as a potential contact allergen. In this article, we review common sources of sulfite exposure, clinical manifestations of allergic contact dermatitis (ACD) to sulfites, and patch testing considerations for this emerging allergen.
What Are Sulfites?
Sulfiting agents are compounds that contain the sulfite ion SO32-, including sulfur dioxide, sodium disulfite (sodium metabisulfite), and potassium metabisulfite.2 Sulfites occur naturally in the environment and commonly are used as preservatives, antibrowning agents, and antioxidants in various foods, beverages, medications, cosmetics, and skin care products. As antibrowning agents and antioxidants, sulfites help maintain the natural appearance of foods and other products and prevent premature spoiling by inactivating oxidative enzymes.3 It should be noted that sulfites and sulfates are distinct and unrelated compounds that do not cross-react.1
Common Sources of Sulfite Exposure
From a morning glass of juice to an evening shower, in the pharmacy and at the hair salon, sulfite exposure is ubiquitous in most daily routines. Sulfites are present in many foods and beverages, either as a byproduct of natural fermentation or as an additive to prevent spoiling and color change. The Table provides examples of foods with high sulfite content.1,4-6 In particular, dried fruit, bottled lemon juice, wine, grape juice, sauerkraut juice, and pickled onions have high sulfite content.

Topical medications and personal care products represent other potential sources of sulfite exposure. A number of reports have shown that sulfites may be included in topical steroids,7 antibiotics,8 antifungals,9 hemorrhoidal preparations,10 local anesthetics,11 and urinary catheterization gel,12 highlighting their many potential applications. In addition, a comprehensive ingredient analysis of 264 ophthalmic medications found that 3.8% of the products contained sodium disulfite.13 Sulfites may be found in personal care products, including facial and hand cleansers, shampoos, moisturizers, and toothpastes. Hair dyes also commonly contain sulfites,7 which are listed in as many as 90% of hair dye kits in the ACDS Contact Allergen Management Program database.1
Occupational exposures also are widespread, as sulfites are extensively utilized across diverse industries such as pharmaceuticals, health care, leather manufacturing, mineral extraction, food preparation, chemical manufacturing, textiles, alcohol brewing, and wine production.1
Sulfites also are used in the rubber industry—particularly in gloves—due to their anticoagulant and preservative properties.4 This is relevant to health care providers, who may use dozens of disposable gloves in a single day. In an experimental pilot study, researchers detected sulfites in 83% (5/6) of natural rubber latex gloves, 96% (23/24) of synthetic (nitrile) gloves, and 0% (0/5) of polyvinyl chloride gloves.14 While this study was limited to a small sample size, it demonstrates the common use of sulfites in certain rubber gloves and encourages future studies to determine whether there is a quantitative threshold to elicit allergic reactions.
Sulfite Allergy
In 1968, an early case report of ACD to sulfites was published involving a pharmaceutical worker who developed hand eczema after working at a factory for 3 months and had a positive patch test to potassium metabisulfite.15 There have been other cases published in the literature since then, including localized ACD as well as less common cases of systemic contact dermatitis following oral, injectable, and rectal sulfite exposures.16
The North American Contact Dermatitis Group found that, among 132 (2.7%) of 4885 patients with positive patch tests to sodium disulfite from 2017 to 2018, the most commonly involved body sites were the face (28.8%) and hands (20.5%) followed by a scattered/generalized distribution (13.6%). Involvement of the face and hands may correlate with the most frequent sources of exposure that were identified, including personal care products (particularly hair dyes)(18.9%), medications (9.1%), and foods (7.6%).17 A multicenter analysis of patch test results from Germany, Austria, and Switzerland from 1999 to 2013 showed that 357 (2.9%) of 12,156 patients had positive reactions to sodium disulfite, with the most commonly identified exposure sources being topical pharmaceutical agents (59.3%); cosmetics, creams, and sunscreens (13.6%); and systemic drugs (6.8%).18 However, it is not always possible to determine the clinical relevance of a positive patch test to sulfites.1
Other than the face and hands, there have been other unexpected anatomic locations for sulfite ACD (eg, the lower back), and systemic contact dermatitis has manifested with widespread rashes due to oral, rectal, and parenteral exposure.4,16,19 There is no definitive link between sulfite contact allergy and patient sex, but there seems to be a higher prevalence in patients older than 40 years, perhaps related to overall lifetime exposure.1
Immediate hypersensitivity reactions to sulfites also have been reported, including urticaria, angioedema, and anaphylaxis.4 Due to multiple cases of severe dermatologic and respiratory reactions to food products containing sulfites,20 the US Food and Drug Administration prohibited their use in fresh fruit and vegetables as antibrowning agents in 1986 and required labels on packaged foods that contained sulfites at more than 10 parts per million.21 However, food and drinks produced in restaurants, bakeries, and cafes as well as those that are distributed directly to consumers from the preparation site are exempt from these rules.17
In addition, consuming high amounts of dietary sulfites has been linked to headaches through unclear (ie, not necessarily allergic) mechanisms.4,22 One study found that wine with a higher sulfite concentration was associated with increased risk for headaches in participants who had a history of headaches related to wine consumption.22
Patch Testing to Sulfites
The North American Contact Dermatitis Group has tested sodium disulfite since 2017 and found an increased frequency of positive patch tests from 2.7% (N=4885) in 2017 and 201817 to 3.3% (N=4115) in 2019 and 202023 among patients referred for testing. Similarly, patch testing to sodium disulfite in nearly 40,000 patients in 9 European countries showed a pooled prevalence of reactions of 3.1%.17 However, this contact allergy may go unrecognized, as sulfites are not included in common patch test series, including the thin-layer rapid use epicutaneous test and the ACDS Core Allergen Series.24,25 The relatively high patch test positivity to sulfites along with the prevalence of daily exposures supports the addition of sulfites to more patch test screening series.
The recommended patch test concentration for sodium disulfite is 1% in petrolatum.5 Testing in aqueous solutions is not recommended because they can cause sulfites to break down, potentially producing false-positive or irritant patch test reactions.7,26,27
Recommendations for Patients With Sulfite Allergies
Individuals with contact allergies to sulfites should be counseled on exposure sources and should be given resources providing a list of safe products, such as the ACDS Contact Allergen Management Program (https://www.acdscamp.org/login) or SkinSAFE (https://www.skinsafeproducts.com/). Prescribers should be cognizant of sulfites that are present in prescription medications. Just because a patient has a positive patch test to sulfites does not automatically imply that they will need to modify their diet to avoid sulfite-containing foods; in the absence of cheilitis or a distribution suggestive of systemic contact dermatitis (eg, vesicular hand/foot dermatitis, intertriginous eruptions), this step may be unnecessary. On the other hand, individuals who have experienced immediate hypersensitivity reactions to sulfites should avoid sulfite-containing foods and carry an epinephrine autoinjector.
Final Interpretation
Sulfites are ubiquitous compounds found in various foods, beverages, medications, and personal care products in addition to a range of occupational exposures. The face and hands are the most common sites of sulfite ACD. Despite patch test positivity in as many as 3% of tested patients,17,23 sulfite allergy may be missed due to lack of routine testing on standard screening series.
The American Contact Dermatitis Society (ACDS) selected sulfites as the 2024 Allergen of the Year.1 Due to their preservative and antioxidant properties, sulfites are prevalent in a variety of foods, beverages, medications, and personal care products; however, sulfites also have been implicated as a potential contact allergen. In this article, we review common sources of sulfite exposure, clinical manifestations of allergic contact dermatitis (ACD) to sulfites, and patch testing considerations for this emerging allergen.
What Are Sulfites?
Sulfiting agents are compounds that contain the sulfite ion SO32-, including sulfur dioxide, sodium disulfite (sodium metabisulfite), and potassium metabisulfite.2 Sulfites occur naturally in the environment and commonly are used as preservatives, antibrowning agents, and antioxidants in various foods, beverages, medications, cosmetics, and skin care products. As antibrowning agents and antioxidants, sulfites help maintain the natural appearance of foods and other products and prevent premature spoiling by inactivating oxidative enzymes.3 It should be noted that sulfites and sulfates are distinct and unrelated compounds that do not cross-react.1
Common Sources of Sulfite Exposure
From a morning glass of juice to an evening shower, in the pharmacy and at the hair salon, sulfite exposure is ubiquitous in most daily routines. Sulfites are present in many foods and beverages, either as a byproduct of natural fermentation or as an additive to prevent spoiling and color change. The Table provides examples of foods with high sulfite content.1,4-6 In particular, dried fruit, bottled lemon juice, wine, grape juice, sauerkraut juice, and pickled onions have high sulfite content.

Topical medications and personal care products represent other potential sources of sulfite exposure. A number of reports have shown that sulfites may be included in topical steroids,7 antibiotics,8 antifungals,9 hemorrhoidal preparations,10 local anesthetics,11 and urinary catheterization gel,12 highlighting their many potential applications. In addition, a comprehensive ingredient analysis of 264 ophthalmic medications found that 3.8% of the products contained sodium disulfite.13 Sulfites may be found in personal care products, including facial and hand cleansers, shampoos, moisturizers, and toothpastes. Hair dyes also commonly contain sulfites,7 which are listed in as many as 90% of hair dye kits in the ACDS Contact Allergen Management Program database.1
Occupational exposures also are widespread, as sulfites are extensively utilized across diverse industries such as pharmaceuticals, health care, leather manufacturing, mineral extraction, food preparation, chemical manufacturing, textiles, alcohol brewing, and wine production.1
Sulfites also are used in the rubber industry—particularly in gloves—due to their anticoagulant and preservative properties.4 This is relevant to health care providers, who may use dozens of disposable gloves in a single day. In an experimental pilot study, researchers detected sulfites in 83% (5/6) of natural rubber latex gloves, 96% (23/24) of synthetic (nitrile) gloves, and 0% (0/5) of polyvinyl chloride gloves.14 While this study was limited to a small sample size, it demonstrates the common use of sulfites in certain rubber gloves and encourages future studies to determine whether there is a quantitative threshold to elicit allergic reactions.
Sulfite Allergy
In 1968, an early case report of ACD to sulfites was published involving a pharmaceutical worker who developed hand eczema after working at a factory for 3 months and had a positive patch test to potassium metabisulfite.15 There have been other cases published in the literature since then, including localized ACD as well as less common cases of systemic contact dermatitis following oral, injectable, and rectal sulfite exposures.16
The North American Contact Dermatitis Group found that, among 132 (2.7%) of 4885 patients with positive patch tests to sodium disulfite from 2017 to 2018, the most commonly involved body sites were the face (28.8%) and hands (20.5%) followed by a scattered/generalized distribution (13.6%). Involvement of the face and hands may correlate with the most frequent sources of exposure that were identified, including personal care products (particularly hair dyes)(18.9%), medications (9.1%), and foods (7.6%).17 A multicenter analysis of patch test results from Germany, Austria, and Switzerland from 1999 to 2013 showed that 357 (2.9%) of 12,156 patients had positive reactions to sodium disulfite, with the most commonly identified exposure sources being topical pharmaceutical agents (59.3%); cosmetics, creams, and sunscreens (13.6%); and systemic drugs (6.8%).18 However, it is not always possible to determine the clinical relevance of a positive patch test to sulfites.1
Other than the face and hands, there have been other unexpected anatomic locations for sulfite ACD (eg, the lower back), and systemic contact dermatitis has manifested with widespread rashes due to oral, rectal, and parenteral exposure.4,16,19 There is no definitive link between sulfite contact allergy and patient sex, but there seems to be a higher prevalence in patients older than 40 years, perhaps related to overall lifetime exposure.1
Immediate hypersensitivity reactions to sulfites also have been reported, including urticaria, angioedema, and anaphylaxis.4 Due to multiple cases of severe dermatologic and respiratory reactions to food products containing sulfites,20 the US Food and Drug Administration prohibited their use in fresh fruit and vegetables as antibrowning agents in 1986 and required labels on packaged foods that contained sulfites at more than 10 parts per million.21 However, food and drinks produced in restaurants, bakeries, and cafes as well as those that are distributed directly to consumers from the preparation site are exempt from these rules.17
In addition, consuming high amounts of dietary sulfites has been linked to headaches through unclear (ie, not necessarily allergic) mechanisms.4,22 One study found that wine with a higher sulfite concentration was associated with increased risk for headaches in participants who had a history of headaches related to wine consumption.22
Patch Testing to Sulfites
The North American Contact Dermatitis Group has tested sodium disulfite since 2017 and found an increased frequency of positive patch tests from 2.7% (N=4885) in 2017 and 201817 to 3.3% (N=4115) in 2019 and 202023 among patients referred for testing. Similarly, patch testing to sodium disulfite in nearly 40,000 patients in 9 European countries showed a pooled prevalence of reactions of 3.1%.17 However, this contact allergy may go unrecognized, as sulfites are not included in common patch test series, including the thin-layer rapid use epicutaneous test and the ACDS Core Allergen Series.24,25 The relatively high patch test positivity to sulfites along with the prevalence of daily exposures supports the addition of sulfites to more patch test screening series.
The recommended patch test concentration for sodium disulfite is 1% in petrolatum.5 Testing in aqueous solutions is not recommended because they can cause sulfites to break down, potentially producing false-positive or irritant patch test reactions.7,26,27
Recommendations for Patients With Sulfite Allergies
Individuals with contact allergies to sulfites should be counseled on exposure sources and should be given resources providing a list of safe products, such as the ACDS Contact Allergen Management Program (https://www.acdscamp.org/login) or SkinSAFE (https://www.skinsafeproducts.com/). Prescribers should be cognizant of sulfites that are present in prescription medications. Just because a patient has a positive patch test to sulfites does not automatically imply that they will need to modify their diet to avoid sulfite-containing foods; in the absence of cheilitis or a distribution suggestive of systemic contact dermatitis (eg, vesicular hand/foot dermatitis, intertriginous eruptions), this step may be unnecessary. On the other hand, individuals who have experienced immediate hypersensitivity reactions to sulfites should avoid sulfite-containing foods and carry an epinephrine autoinjector.
Final Interpretation
Sulfites are ubiquitous compounds found in various foods, beverages, medications, and personal care products in addition to a range of occupational exposures. The face and hands are the most common sites of sulfite ACD. Despite patch test positivity in as many as 3% of tested patients,17,23 sulfite allergy may be missed due to lack of routine testing on standard screening series.
- Ekstein SF, Warshaw EM. Sulfites: allergen of the year 2024. Dermatitis. 2024;35:6-12. doi:10.1089/derm.2023.0154
- Gunnison AF, Jacobsen DW. Sulfite hypersensitivity. a critical review. CRC Crit Rev Toxicol. 1987;17:185-214. doi:10.3109/10408448709071208
- Clough SR. Sodium sulfite. In: Wexler P, ed. Encyclopedia of Toxicology. 3rd ed. Academic Press; 2014: 341-343.
- Vally H, Misso NL, Madan V. Clinical effects of sulphite additives. Clin Exp Allergy. 2009;39:1643-1651. doi:10.1111/j.1365-2222.2009.03362.x
- Ralph N, Verma S, Merry S, et al. What is the relevance of contact allergy to sodium metabisulfite and which concentration of the allergen should we use? Dermatitis. 2015;26:162-165. doi:10.1097/der.0000000000000120
- Madan V, Walker SL, Beck MH. Sodium metabisulfite allergy is common but is it relevant? Contact Dermatitis. 2007;57:173-176. doi:10.1111/j.1600-0536.2007.01188.x
- García-Gavín J, Parente J, Goossens A. Allergic contact dermatitis caused by sodium metabisulfite: a challenging allergen. a case series and literature review. Contact Dermatitis. 2012;67:260-269. doi:10.1111/j.1600-0536.2012.02135.x
- Milpied B, van Wassenhove L, Larousse C, et al. Contact dermatitis from rifamycin. Contact Dermatitis. 1986;14:252-253. doi:10.1111/j.1600-0536.1986.tb01240.x
- Lodi A, Chiarelli G, Mancini LL, et al. Contact allergy to sodium sulfite contained in an antifungal preparation. Contact Dermatitis. 1993;29:97. doi:10.1111/j.1600-0536.1993.tb03493.x
- Sánchez-Pérez J, Abajo P, Córdoba S, et al. Allergic contact dermatitis from sodium metabisulfite in an antihemorrhoidal cream. Contact Dermatitis. 2000;42:176-177.
- Boyd AH, Warshaw EM. Sulfites: no longer a zebra? Dermatitis. 2017;28:364-366. doi:10.1097/der.0000000000000312
- Grosch E, Mahler V. Allergic contact dermatitis caused by a catheter system containing sodium metabisulfite. Contact Dermatitis. 2017;76:186-187. doi:10.1111/cod.12675
- Shaver RL, Warshaw EM. Contact allergens in prescription topical ophthalmic medications. Dermatitis. 2022;33:135-143. doi:10.1097/der.0000000000000751
- Dendooven E, Darrigade AS, Foubert K, et al. The presence of sulfites in ‘natural rubber latex’ and ‘synthetic’ rubber gloves: an experimental pilot study. Br J Dermatol. 2020;182:1054-1055. doi:10.1111/bjd.18608
- Nater JP. Allergic contact dermatitis caused by potassium metabisulfite. Dermatologica. 1968;136:477-478. doi:10.1159/000254143
- Borges AS, Valejo Coelho MM, Fernandes C, et al. Systemic allergic dermatitis caused by sodium metabisulfite in rectal enemas. Contact Dermatitis. 2018;78:429-430. doi:10.1111/cod.12971
- Warshaw EM, Buonomo M, DeKoven JG, et al. Patch testing with sodium disulfite: North American Contact Dermatitis Group experience, 2017 to 2018. Contact Dermatitis. 2021;85:285-296. doi:10.1111/cod.13860
- Häberle M, Geier J, Mahler V. Contact allergy to sulfites: clinical and occupational relevance—new data from the German Contact Dermatitis Research Group and the Information Network of Departments of Dermatology (IVDK). J Dtsch Dermatol Ges. 2016;14:938-941. doi:10.1111/ddg.13009
- Tan MG, Li HO, Pratt MD. Systemic allergic dermatitis to sodium metabisulfite in local anesthetic solution. Contact Dermatitis. 2022;86:120-121. doi:10.1111/cod.13978
- D’Amore T, Di Taranto A, Berardi G, et al. Sulfites in meat: occurrence, activity, toxicity, regulation, and detection. a comprehensive review. Compr Rev Food Sci Food Saf. 2020;19:2701-2720. doi:10.1111/1541-4337.12607
- Grotheer P, Marshall M, Simonne A. Sulfites: separating fact from fiction. May 11, 2022. UF IFAS Extension. University of Florida. Accessed October 4, 2024. https://edis.ifas.ufl.edu/publication/FY731
- Silva M, Gama J, Pinto N, et al. Sulfite concentration and the occurrence of headache in young adults: a prospective study. Eur J Clin Nutr. 2019;73:1316-1322. doi:10.1038/s41430-019-0420-2
- DeKoven JG, Warshaw EM, Reeder MJ, et al. North American Contact Dermatitis Group patch test results: 2019-2020. Dermatitis. 2023;34:90-104. doi:10.1089/derm.2022.29017.jdk
- T.R.U.E. Test. Thin-layer rapid use epicutaneous patch test. SmartPractice Dermatology Allergy. Accessed October 4, 2024. https://www.smartpractice.com/shop/category?id=581719&m=SPA
- Schalock PC, Dunnick CA, Nedorost, et al; American Contact Dermatitis Society Core Allergen Series Committee. American Contact Dermatitis Society Core Allergen Series: 2020 update. Dermatitis. 2020;31:279-282.
- Kaaman AC, Boman A, Wrangsjö K, et al. Contact allergy to sodium metabisulfite: an occupational problem. Contact Dermatitis. 2010;63:110-112. doi:10.1111/j.1600-0536.2010.01756.x
- Vena GA, Foti C, Angelini G. Sulfite contact allergy. Contact Dermatitis. 1994;31:172-175. doi:10.1111/j.1600-0536.1994.tb01959.x
- Ekstein SF, Warshaw EM. Sulfites: allergen of the year 2024. Dermatitis. 2024;35:6-12. doi:10.1089/derm.2023.0154
- Gunnison AF, Jacobsen DW. Sulfite hypersensitivity. a critical review. CRC Crit Rev Toxicol. 1987;17:185-214. doi:10.3109/10408448709071208
- Clough SR. Sodium sulfite. In: Wexler P, ed. Encyclopedia of Toxicology. 3rd ed. Academic Press; 2014: 341-343.
- Vally H, Misso NL, Madan V. Clinical effects of sulphite additives. Clin Exp Allergy. 2009;39:1643-1651. doi:10.1111/j.1365-2222.2009.03362.x
- Ralph N, Verma S, Merry S, et al. What is the relevance of contact allergy to sodium metabisulfite and which concentration of the allergen should we use? Dermatitis. 2015;26:162-165. doi:10.1097/der.0000000000000120
- Madan V, Walker SL, Beck MH. Sodium metabisulfite allergy is common but is it relevant? Contact Dermatitis. 2007;57:173-176. doi:10.1111/j.1600-0536.2007.01188.x
- García-Gavín J, Parente J, Goossens A. Allergic contact dermatitis caused by sodium metabisulfite: a challenging allergen. a case series and literature review. Contact Dermatitis. 2012;67:260-269. doi:10.1111/j.1600-0536.2012.02135.x
- Milpied B, van Wassenhove L, Larousse C, et al. Contact dermatitis from rifamycin. Contact Dermatitis. 1986;14:252-253. doi:10.1111/j.1600-0536.1986.tb01240.x
- Lodi A, Chiarelli G, Mancini LL, et al. Contact allergy to sodium sulfite contained in an antifungal preparation. Contact Dermatitis. 1993;29:97. doi:10.1111/j.1600-0536.1993.tb03493.x
- Sánchez-Pérez J, Abajo P, Córdoba S, et al. Allergic contact dermatitis from sodium metabisulfite in an antihemorrhoidal cream. Contact Dermatitis. 2000;42:176-177.
- Boyd AH, Warshaw EM. Sulfites: no longer a zebra? Dermatitis. 2017;28:364-366. doi:10.1097/der.0000000000000312
- Grosch E, Mahler V. Allergic contact dermatitis caused by a catheter system containing sodium metabisulfite. Contact Dermatitis. 2017;76:186-187. doi:10.1111/cod.12675
- Shaver RL, Warshaw EM. Contact allergens in prescription topical ophthalmic medications. Dermatitis. 2022;33:135-143. doi:10.1097/der.0000000000000751
- Dendooven E, Darrigade AS, Foubert K, et al. The presence of sulfites in ‘natural rubber latex’ and ‘synthetic’ rubber gloves: an experimental pilot study. Br J Dermatol. 2020;182:1054-1055. doi:10.1111/bjd.18608
- Nater JP. Allergic contact dermatitis caused by potassium metabisulfite. Dermatologica. 1968;136:477-478. doi:10.1159/000254143
- Borges AS, Valejo Coelho MM, Fernandes C, et al. Systemic allergic dermatitis caused by sodium metabisulfite in rectal enemas. Contact Dermatitis. 2018;78:429-430. doi:10.1111/cod.12971
- Warshaw EM, Buonomo M, DeKoven JG, et al. Patch testing with sodium disulfite: North American Contact Dermatitis Group experience, 2017 to 2018. Contact Dermatitis. 2021;85:285-296. doi:10.1111/cod.13860
- Häberle M, Geier J, Mahler V. Contact allergy to sulfites: clinical and occupational relevance—new data from the German Contact Dermatitis Research Group and the Information Network of Departments of Dermatology (IVDK). J Dtsch Dermatol Ges. 2016;14:938-941. doi:10.1111/ddg.13009
- Tan MG, Li HO, Pratt MD. Systemic allergic dermatitis to sodium metabisulfite in local anesthetic solution. Contact Dermatitis. 2022;86:120-121. doi:10.1111/cod.13978
- D’Amore T, Di Taranto A, Berardi G, et al. Sulfites in meat: occurrence, activity, toxicity, regulation, and detection. a comprehensive review. Compr Rev Food Sci Food Saf. 2020;19:2701-2720. doi:10.1111/1541-4337.12607
- Grotheer P, Marshall M, Simonne A. Sulfites: separating fact from fiction. May 11, 2022. UF IFAS Extension. University of Florida. Accessed October 4, 2024. https://edis.ifas.ufl.edu/publication/FY731
- Silva M, Gama J, Pinto N, et al. Sulfite concentration and the occurrence of headache in young adults: a prospective study. Eur J Clin Nutr. 2019;73:1316-1322. doi:10.1038/s41430-019-0420-2
- DeKoven JG, Warshaw EM, Reeder MJ, et al. North American Contact Dermatitis Group patch test results: 2019-2020. Dermatitis. 2023;34:90-104. doi:10.1089/derm.2022.29017.jdk
- T.R.U.E. Test. Thin-layer rapid use epicutaneous patch test. SmartPractice Dermatology Allergy. Accessed October 4, 2024. https://www.smartpractice.com/shop/category?id=581719&m=SPA
- Schalock PC, Dunnick CA, Nedorost, et al; American Contact Dermatitis Society Core Allergen Series Committee. American Contact Dermatitis Society Core Allergen Series: 2020 update. Dermatitis. 2020;31:279-282.
- Kaaman AC, Boman A, Wrangsjö K, et al. Contact allergy to sodium metabisulfite: an occupational problem. Contact Dermatitis. 2010;63:110-112. doi:10.1111/j.1600-0536.2010.01756.x
- Vena GA, Foti C, Angelini G. Sulfite contact allergy. Contact Dermatitis. 1994;31:172-175. doi:10.1111/j.1600-0536.1994.tb01959.x
Practice Points
- Sulfites are ubiquitous compounds that serve as preservatives and antioxidants in various foods, beverages, medications, and personal care products.
- Allergic contact dermatitis to sulfites most commonly affects the face and hands.
- Because sulfites are not included in most patch test screening series, contact allergy to sulfites may be missed unless expanded testing is performed.
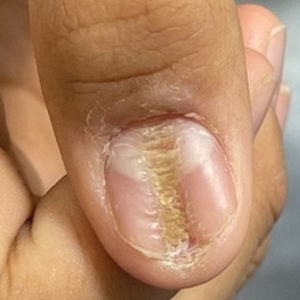
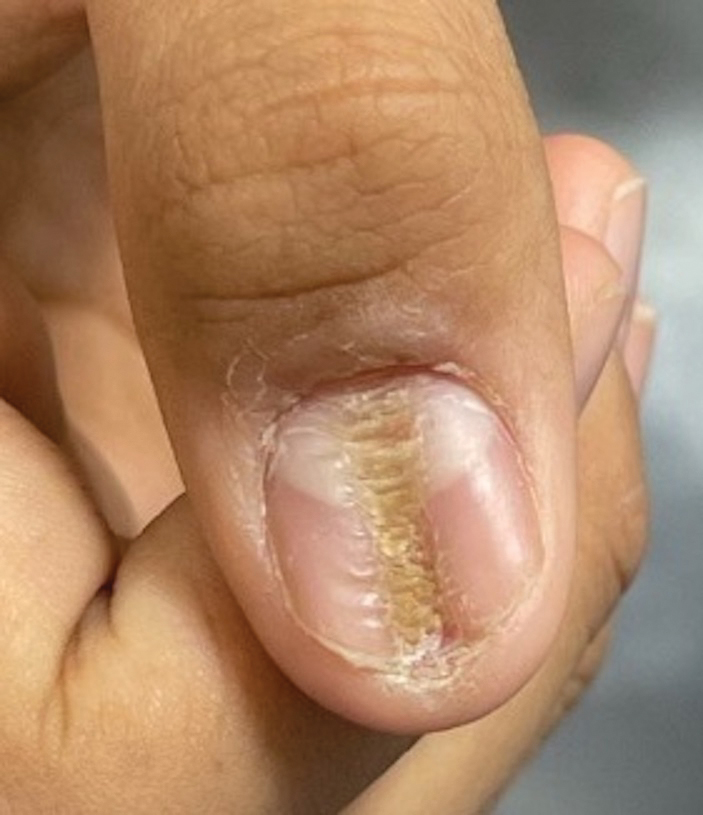
Longitudinal Depression on the Right Thumbnail
THE DIAGNOSIS: Habit-Tic Deformity
Habit-tic deformity is a cause of nail dystrophy that commonly arises in children and adults due to subconscious repetitive and self-injurious manipulation of the nail bed or cuticle, which ultimately damages the nail matrix.1,2 It can be considered a variant of onychotillomania.1
Characteristic features of habit-tic deformity include a longitudinal depression on the central nail plate with transverse ridges,1 which can be more prominent on the dominant hand.3 Patients typically note a long duration of nail deformity, often without insight into its etiology.2 Diagnosis relies on careful assessment of the clinical presentation and the patient’s history to rule out other differential diagnoses. Based on our patient’s clinical presentation and history, we excluded wart, squamous cell carcinoma, eczema, psoriasis, lichen planus, autoimmune connective tissue disease, onychomycosis, paronychia, pincer nail deformity, and Beau line as potential diagnoses. Biopsy also can be performed to exclude these diagnoses from the differential if the cause is unclear following clinical examination.
Treatment for habit-tic deformity involves identifying and addressing the underlying habit. Barrier methods such as bandages and cyanoacrylate adhesives that prevent further manipulation of the nail matrix are effective treatments for habit-tic deformity.2 A multidisciplinary approach with psychiatry may be optimal to identify underlying psychological comorbidities and break the habit through behavior interventions and medications.4 Nail dystrophy generally improves once the habit is disrupted; however, a younger age of onset may carry a worse prognosis.3 Patients should be counseled that the affected nail may never grow normally.
Our patient was advised to use fluocinonide ointment 0.05% to reduce inflammation of the proximal nail fold and to cover the thumbnail with a bandage to prevent picking. He also was counseled that the nail may show ongoing abnormal growth. Minimal improvement was noted after 6 months.
- Rieder EA, Tosti A. Onychotillomania: an underrecognized disorder. J Am Acad Dermatol. 2016;75:1245-1250.doi:10.1016/j.jaad.2016
- Ring DS. Inexpensive solution for habit-tic deformity. Arch Dermatol. 2010;146:1222-1223. doi:10.1001/archdermatol.2010.287
- Horne MI, Utzig JB, Rieder EA, et al. Alopecia areata and habit tic deformities. Skin Appendage Disord. 2018;4:323-325. doi:10.1159/000486540
- Sonthalia S, Sharma P, Kapoor J, et al. Habit tic deformity: need fora comprehensive approach. Skin Appendage Disord. 2019;5:117-118.doi:10.1159/000489320 .05.036
THE DIAGNOSIS: Habit-Tic Deformity
Habit-tic deformity is a cause of nail dystrophy that commonly arises in children and adults due to subconscious repetitive and self-injurious manipulation of the nail bed or cuticle, which ultimately damages the nail matrix.1,2 It can be considered a variant of onychotillomania.1
Characteristic features of habit-tic deformity include a longitudinal depression on the central nail plate with transverse ridges,1 which can be more prominent on the dominant hand.3 Patients typically note a long duration of nail deformity, often without insight into its etiology.2 Diagnosis relies on careful assessment of the clinical presentation and the patient’s history to rule out other differential diagnoses. Based on our patient’s clinical presentation and history, we excluded wart, squamous cell carcinoma, eczema, psoriasis, lichen planus, autoimmune connective tissue disease, onychomycosis, paronychia, pincer nail deformity, and Beau line as potential diagnoses. Biopsy also can be performed to exclude these diagnoses from the differential if the cause is unclear following clinical examination.
Treatment for habit-tic deformity involves identifying and addressing the underlying habit. Barrier methods such as bandages and cyanoacrylate adhesives that prevent further manipulation of the nail matrix are effective treatments for habit-tic deformity.2 A multidisciplinary approach with psychiatry may be optimal to identify underlying psychological comorbidities and break the habit through behavior interventions and medications.4 Nail dystrophy generally improves once the habit is disrupted; however, a younger age of onset may carry a worse prognosis.3 Patients should be counseled that the affected nail may never grow normally.
Our patient was advised to use fluocinonide ointment 0.05% to reduce inflammation of the proximal nail fold and to cover the thumbnail with a bandage to prevent picking. He also was counseled that the nail may show ongoing abnormal growth. Minimal improvement was noted after 6 months.
THE DIAGNOSIS: Habit-Tic Deformity
Habit-tic deformity is a cause of nail dystrophy that commonly arises in children and adults due to subconscious repetitive and self-injurious manipulation of the nail bed or cuticle, which ultimately damages the nail matrix.1,2 It can be considered a variant of onychotillomania.1
Characteristic features of habit-tic deformity include a longitudinal depression on the central nail plate with transverse ridges,1 which can be more prominent on the dominant hand.3 Patients typically note a long duration of nail deformity, often without insight into its etiology.2 Diagnosis relies on careful assessment of the clinical presentation and the patient’s history to rule out other differential diagnoses. Based on our patient’s clinical presentation and history, we excluded wart, squamous cell carcinoma, eczema, psoriasis, lichen planus, autoimmune connective tissue disease, onychomycosis, paronychia, pincer nail deformity, and Beau line as potential diagnoses. Biopsy also can be performed to exclude these diagnoses from the differential if the cause is unclear following clinical examination.
Treatment for habit-tic deformity involves identifying and addressing the underlying habit. Barrier methods such as bandages and cyanoacrylate adhesives that prevent further manipulation of the nail matrix are effective treatments for habit-tic deformity.2 A multidisciplinary approach with psychiatry may be optimal to identify underlying psychological comorbidities and break the habit through behavior interventions and medications.4 Nail dystrophy generally improves once the habit is disrupted; however, a younger age of onset may carry a worse prognosis.3 Patients should be counseled that the affected nail may never grow normally.
Our patient was advised to use fluocinonide ointment 0.05% to reduce inflammation of the proximal nail fold and to cover the thumbnail with a bandage to prevent picking. He also was counseled that the nail may show ongoing abnormal growth. Minimal improvement was noted after 6 months.
- Rieder EA, Tosti A. Onychotillomania: an underrecognized disorder. J Am Acad Dermatol. 2016;75:1245-1250.doi:10.1016/j.jaad.2016
- Ring DS. Inexpensive solution for habit-tic deformity. Arch Dermatol. 2010;146:1222-1223. doi:10.1001/archdermatol.2010.287
- Horne MI, Utzig JB, Rieder EA, et al. Alopecia areata and habit tic deformities. Skin Appendage Disord. 2018;4:323-325. doi:10.1159/000486540
- Sonthalia S, Sharma P, Kapoor J, et al. Habit tic deformity: need fora comprehensive approach. Skin Appendage Disord. 2019;5:117-118.doi:10.1159/000489320 .05.036
- Rieder EA, Tosti A. Onychotillomania: an underrecognized disorder. J Am Acad Dermatol. 2016;75:1245-1250.doi:10.1016/j.jaad.2016
- Ring DS. Inexpensive solution for habit-tic deformity. Arch Dermatol. 2010;146:1222-1223. doi:10.1001/archdermatol.2010.287
- Horne MI, Utzig JB, Rieder EA, et al. Alopecia areata and habit tic deformities. Skin Appendage Disord. 2018;4:323-325. doi:10.1159/000486540
- Sonthalia S, Sharma P, Kapoor J, et al. Habit tic deformity: need fora comprehensive approach. Skin Appendage Disord. 2019;5:117-118.doi:10.1159/000489320 .05.036
A healthy 13-year-old boy presented to the dermatology department with dystrophy of the right thumbnail of 3 to 4 years’ duration. A 5-mm-wide, depressed median longitudinal groove with a fir tree pattern was noted on the central nail plate. The patient noted that the groove had been gradually deepening. There was erythema, edema, and lichenification of the proximal nailfold without vascular changes, and the lunula was enlarged. No hyperkeratosis, subungual debris, erythematous nail folds, or inward curvature of the lateral aspects of the nail were noted. The patient denied any pruritus, pain, discomfort, or bleeding; he also denied any recent illness or trauma to the nail. None of the other nails were affected, and no other lesions or rashes were observed elsewhere on the body. The patient was unsure if he picked at the nail but acknowledged that he may have done so subconsciously. He had no history of eczema, psoriasis, or autoimmune connective tissue disorders.
Sea Buckthorn
A member of the Elaeagnaceae family, Hippophae rhamnoides, better known as sea buckthorn, is a high-altitude wild shrub endemic to Europe and Asia with edible fruits and a lengthy record of use in traditional Chinese medicine.1-6 Used as a health supplement and consumed in the diet throughout the world,5 sea buckthorn berries, seeds, and leaves have been used in traditional medicine to treat burns/injuries, edema, hypertension, inflammation, skin grafts, ulcers, and wounds.4,7
This hardy plant is associated with a wide range of biologic activities, including anti-atherogenic, anti-atopic dermatitis, antibacterial, anticancer, antifungal, anti-inflammatory, antimicrobial, antioxidant, anti-psoriasis, anti-sebum, anti-stress, anti-tumor, cytoprotective, hepatoprotective, immunomodulatory, neuroprotective, radioprotective, and tissue regenerative functions.4,5,8-11
Key Constituents
Functional constituents identified in sea buckthorn include alkaloids, carotenoids, flavonoids, lignans, organic acids, phenolic acids, proanthocyanidins, polyunsaturated acids (including omega-3, -6, -7, and -9), steroids, tannins, terpenoids, and volatile oils, as well as nutritional compounds such as minerals, proteins, and vitamins.4,5,11 Sea buckthorn pericarp oil contains copious amounts of saturated palmitic acid (29%-36%) and omega-7 unsaturated palmitoleic acid (36%-48%), which fosters cutaneous and mucosal epithelialization, as well as linoleic (10%-12%) and oleic (4%-6%) acids.12,6 Significant amounts of carotenoids as well as alpha‐linolenic fatty acid (38%), linoleic (36%), oleic (13%), and palmitic (7%) acids are present in sea buckthorn seed oil.6
Polysaccharides
In an expansive review on the pharmacological activities of sea buckthorn polysaccharides, Teng and colleagues reported in April 2024 that 20 diverse polysaccharides have been culled from sea buckthorn and exhibited various healthy activities, including antioxidant, anti-fatigue, anti-inflammatory, anti-obesity, anti-tumor, hepatoprotective, hypoglycemic, and immunoregulation, and regulation of intestinal flora activities.1
Proanthocyanidins and Anti-Aging
In 2023, Liu and colleagues investigated the anti–skin aging impact of sea buckthorn proanthocyanidins in D-galactose-induced aging in mice given the known free radical scavenging activity of these compounds. They found the proanthocyanidins mitigated D-galactose-induced aging and can augment the total antioxidant capacity of the body. Sea buckthorn proanthocyanidins can further attenuate the effects of skin aging by regulating the TGF-beta1/Smads pathway and MMPs/TIMP system, thus amplifying collagen I and tropoelastin content.13
A year earlier, many of the same investigators assessed the possible protective activity of sea buckthorn proanthocyanidins against cutaneous aging engendered by oxidative stress from hydrogen peroxide. The compounds amplified superoxide dismutase and glutathione antioxidant functions. The extracts also fostered collagen I production in aging human skin fibroblasts via the TGF-beta1/Smads pathway and hindered collagen I degradation by regulating the MMPs/TIMPs system, which maintained extracellular matrix integrity. Senescent cell migration was also promoted with 100 mcg/mL of sea buckthorn proanthocyanidins. The researchers concluded that this sets the stage for investigating how sea buckthorn proanthocyanidins can be incorporated in cosmetic formulations.14 In a separate study, Liu and colleagues demonstrated that sea buckthorn proanthocyanidins can attenuate oxidative damage and protect mitochondrial function.9
Acne and Barrier Functions
The extracts of H rhamnoides and Cassia fistula in a combined formulation were found to be effective in lowering skin sebum content in humans with grade I and grade II acne vulgaris in a 2014 single-blind, randomized, placebo-controlled, split-face study with two groups of 25 patients each (aged 18-37 years).15 Khan and colleagues have also reported that a sea buckthorn oil-in-water emulsion improved barrier function in human skin as tested by a tewameter and corneometer (noninvasive probes) in 13 healthy males with a mean age of 27 ± 4.8 years.16
Anti-Aging, Antioxidant, Antibacterial, Skin-Whitening Activity
Zaman and colleagues reported in 2011 that results from an in vivo study of the effects of a sea buckthorn fruit extract topical cream on stratum corneum water content and transepidermal water loss indicated that the formulation enhanced cell surface integrin expression thus facilitating collagen contraction.17
In 2012, Khan and colleagues reported amelioration in skin elasticity, thus achieving an anti-aging result, from the use of a water-in-oil–based hydroalcoholic cream loaded with fruit extract of H rhamnoides, as measured with a Cutometer.18 The previous year, some of the same researchers reported that the antioxidants and flavonoids found in a topical sea buckthorn formulation could decrease cutaneous melanin and erythema levels.
More recently, Gęgotek and colleagues found that sea buckthorn seed oil prevented redox balance and lipid metabolism disturbances in skin fibroblasts and keratinocytes caused by UVA or UVB. They suggested that such findings point to the potential of this natural agent to confer anti-inflammatory properties and photoprotection to the skin.19
In 2020, Ivanišová and colleagues investigated the antioxidant and antimicrobial activities of H rhamnoides 100% oil, 100% juice, dry berries, and tea (dry berries, leaves, and twigs). They found that all of the studied sea buckthorn products displayed high antioxidant activity (identified through DPPH radical scavenging and molybdenum reducing antioxidant power tests). Sea buckthorn juice contained the highest total content of polyphenols, flavonoids, and carotenoids. All of the tested products also exhibited substantial antibacterial activity against the tested microbes.20
Burns and Wound Healing
In a preclinical study of the effects of sea buckthorn leaf extracts on wound healing in albino rats using an excision-punch wound model in 2005, Gupta and colleagues found that twice daily topical application of the aqueous leaf extract fostered wound healing. This was indicated by higher hydroxyproline and protein levels, a diminished wound area, and lower lipid peroxide levels. The investigators suggested that sea buckthorn may facilitate wound healing at least in part because of elevated antioxidant activity in the granulation tissue.3
A year later, Wang and colleagues reported on observations of using H rhamnoides oil, a traditional Chinese herbal medicine derived from sea buckthorn fruit, as a burn treatment. In the study, 151 burn patients received an H rhamnoides oil dressing (changed every other day until wound healing) that was covered with a disinfecting dressing. The dressing reduced swelling and effusion, and alleviated pain, with patients receiving the sea buckthorn dressing experiencing greater apparent exudation reduction, pain reduction, and more rapid epithelial cell growth and wound healing than controls (treated only with Vaseline gauze). The difference between the two groups was statistically significant.21
Conclusion
Sea buckthorn has been used for hundreds if not thousands of years in traditional medical applications, including for dermatologic purposes. Emerging data appear to support the use of this dynamic plant for consideration in dermatologic applications. As is often the case, much more work is necessary in the form of randomized controlled trials to determine the effectiveness of sea buckthorn formulations as well as the most appropriate avenues of research or uses for dermatologic application of this traditionally used botanical agent.
Dr. Baumann is a private practice dermatologist, researcher, author, and entrepreneur in Miami. She founded the division of cosmetic dermatology at the University of Miami in 1997. The third edition of her bestselling textbook, “Cosmetic Dermatology,” was published in 2022. Dr. Baumann has received funding for advisory boards and/or clinical research trials from Allergan, Galderma, Johnson & Johnson, and Burt’s Bees. She is the CEO of Skin Type Solutions, a SaaS company used to generate skin care routines in office and as a e-commerce solution. Write to her at [email protected].
References
1. Teng H et al. J Ethnopharmacol. 2024 Apr 24;324:117809. doi: 10.1016/j.jep.2024.117809.
2. Wang Z et al. Int J Biol Macromol. 2024 Apr;263(Pt 1):130206. doi: 10.1016/j.ijbiomac.2024.130206.
3. Gupta A et al. Int J Low Extrem Wounds. 2005 Jun;4(2):88-92. doi: 10.1177/1534734605277401.
4. Pundir S et al. J Ethnopharmacol. 2021 Feb 10;266:113434. doi: 10.1016/j.jep.2020.113434.
5. Ma QG et al. J Agric Food Chem. 2023 Mar 29;71(12):4769-4788. doi: 10.1021/acs.jafc.2c06916.
6. Poljšak N et al. Phytother Res. 2020 Feb;34(2):254-269. doi: 10.1002/ptr.6524. doi: 10.1002/ptr.6524.
7. Upadhyay NK et al. Evid Based Complement Alternat Med. 2011;2011:659705. doi: 10.1093/ecam/nep189.
8. Suryakumar G, Gupta A. J Ethnopharmacol. 2011 Nov 18;138(2):268-78. doi: 10.1016/j.jep.2011.09.024.
9. Liu K et al. Front Pharmacol. 2022 Jul 8;13:914146. doi: 10.3389/fphar.2022.914146.
10. Akhtar N et al. J Pharm Bioallied Sci. 2010 Jan;2(1):13-7. doi: 10.4103/0975-7406.62698.
11. Ren R et al. RSC Adv. 2020 Dec 17;10(73):44654-44671. doi: 10.1039/d0ra06488b.
12. Ito H et al. Burns. 2014 May;40(3):511-9. doi: 10.1016/j.burns.2013.08.011.
13. Liu X et al. Food Sci Nutr. 2023 Dec 7;12(2):1082-1094. doi: 10.1002/fsn3.3823.
14. Liu X at al. Antioxidants (Basel). 2022 Sep 25;11(10):1900. doi: 10.3390/antiox11101900.
15. Khan BA, Akhtar N. Postepy Dermatol Alergol. 2014 Aug;31(4):229-234. doi: 10.5114/pdia.2014.40934.
16. Khan BA, Akhtar N. Pak J Pharm Sci. 2014 Nov;27(6):1919-22.
17. Khan AB et al. African J Pharm Pharmacol. 2011 Aug;5(8):1092-5.
18. Khan BA, Akhtar N, Braga VA. Trop J Pharm Res. 2012;11(6):955-62.
19. Gęgotek A et al. Antioxidants (Basel). 2018 Aug 23;7(9):110. doi: 10.3390/antiox7090110.
20. Ivanišová E et al. Acta Sci Pol Technol Aliment. 2020 Apr-Jun;19(2):195-205. doi: 10.17306/J.AFS.0809.
21. Wang ZY, Luo XL, He CP. Nan Fang Yi Ke Da Xue Xue Bao. 2006 Jan;26(1):124-5.
A member of the Elaeagnaceae family, Hippophae rhamnoides, better known as sea buckthorn, is a high-altitude wild shrub endemic to Europe and Asia with edible fruits and a lengthy record of use in traditional Chinese medicine.1-6 Used as a health supplement and consumed in the diet throughout the world,5 sea buckthorn berries, seeds, and leaves have been used in traditional medicine to treat burns/injuries, edema, hypertension, inflammation, skin grafts, ulcers, and wounds.4,7
This hardy plant is associated with a wide range of biologic activities, including anti-atherogenic, anti-atopic dermatitis, antibacterial, anticancer, antifungal, anti-inflammatory, antimicrobial, antioxidant, anti-psoriasis, anti-sebum, anti-stress, anti-tumor, cytoprotective, hepatoprotective, immunomodulatory, neuroprotective, radioprotective, and tissue regenerative functions.4,5,8-11
Key Constituents
Functional constituents identified in sea buckthorn include alkaloids, carotenoids, flavonoids, lignans, organic acids, phenolic acids, proanthocyanidins, polyunsaturated acids (including omega-3, -6, -7, and -9), steroids, tannins, terpenoids, and volatile oils, as well as nutritional compounds such as minerals, proteins, and vitamins.4,5,11 Sea buckthorn pericarp oil contains copious amounts of saturated palmitic acid (29%-36%) and omega-7 unsaturated palmitoleic acid (36%-48%), which fosters cutaneous and mucosal epithelialization, as well as linoleic (10%-12%) and oleic (4%-6%) acids.12,6 Significant amounts of carotenoids as well as alpha‐linolenic fatty acid (38%), linoleic (36%), oleic (13%), and palmitic (7%) acids are present in sea buckthorn seed oil.6
Polysaccharides
In an expansive review on the pharmacological activities of sea buckthorn polysaccharides, Teng and colleagues reported in April 2024 that 20 diverse polysaccharides have been culled from sea buckthorn and exhibited various healthy activities, including antioxidant, anti-fatigue, anti-inflammatory, anti-obesity, anti-tumor, hepatoprotective, hypoglycemic, and immunoregulation, and regulation of intestinal flora activities.1
Proanthocyanidins and Anti-Aging
In 2023, Liu and colleagues investigated the anti–skin aging impact of sea buckthorn proanthocyanidins in D-galactose-induced aging in mice given the known free radical scavenging activity of these compounds. They found the proanthocyanidins mitigated D-galactose-induced aging and can augment the total antioxidant capacity of the body. Sea buckthorn proanthocyanidins can further attenuate the effects of skin aging by regulating the TGF-beta1/Smads pathway and MMPs/TIMP system, thus amplifying collagen I and tropoelastin content.13
A year earlier, many of the same investigators assessed the possible protective activity of sea buckthorn proanthocyanidins against cutaneous aging engendered by oxidative stress from hydrogen peroxide. The compounds amplified superoxide dismutase and glutathione antioxidant functions. The extracts also fostered collagen I production in aging human skin fibroblasts via the TGF-beta1/Smads pathway and hindered collagen I degradation by regulating the MMPs/TIMPs system, which maintained extracellular matrix integrity. Senescent cell migration was also promoted with 100 mcg/mL of sea buckthorn proanthocyanidins. The researchers concluded that this sets the stage for investigating how sea buckthorn proanthocyanidins can be incorporated in cosmetic formulations.14 In a separate study, Liu and colleagues demonstrated that sea buckthorn proanthocyanidins can attenuate oxidative damage and protect mitochondrial function.9
Acne and Barrier Functions
The extracts of H rhamnoides and Cassia fistula in a combined formulation were found to be effective in lowering skin sebum content in humans with grade I and grade II acne vulgaris in a 2014 single-blind, randomized, placebo-controlled, split-face study with two groups of 25 patients each (aged 18-37 years).15 Khan and colleagues have also reported that a sea buckthorn oil-in-water emulsion improved barrier function in human skin as tested by a tewameter and corneometer (noninvasive probes) in 13 healthy males with a mean age of 27 ± 4.8 years.16
Anti-Aging, Antioxidant, Antibacterial, Skin-Whitening Activity
Zaman and colleagues reported in 2011 that results from an in vivo study of the effects of a sea buckthorn fruit extract topical cream on stratum corneum water content and transepidermal water loss indicated that the formulation enhanced cell surface integrin expression thus facilitating collagen contraction.17
In 2012, Khan and colleagues reported amelioration in skin elasticity, thus achieving an anti-aging result, from the use of a water-in-oil–based hydroalcoholic cream loaded with fruit extract of H rhamnoides, as measured with a Cutometer.18 The previous year, some of the same researchers reported that the antioxidants and flavonoids found in a topical sea buckthorn formulation could decrease cutaneous melanin and erythema levels.
More recently, Gęgotek and colleagues found that sea buckthorn seed oil prevented redox balance and lipid metabolism disturbances in skin fibroblasts and keratinocytes caused by UVA or UVB. They suggested that such findings point to the potential of this natural agent to confer anti-inflammatory properties and photoprotection to the skin.19
In 2020, Ivanišová and colleagues investigated the antioxidant and antimicrobial activities of H rhamnoides 100% oil, 100% juice, dry berries, and tea (dry berries, leaves, and twigs). They found that all of the studied sea buckthorn products displayed high antioxidant activity (identified through DPPH radical scavenging and molybdenum reducing antioxidant power tests). Sea buckthorn juice contained the highest total content of polyphenols, flavonoids, and carotenoids. All of the tested products also exhibited substantial antibacterial activity against the tested microbes.20
Burns and Wound Healing
In a preclinical study of the effects of sea buckthorn leaf extracts on wound healing in albino rats using an excision-punch wound model in 2005, Gupta and colleagues found that twice daily topical application of the aqueous leaf extract fostered wound healing. This was indicated by higher hydroxyproline and protein levels, a diminished wound area, and lower lipid peroxide levels. The investigators suggested that sea buckthorn may facilitate wound healing at least in part because of elevated antioxidant activity in the granulation tissue.3
A year later, Wang and colleagues reported on observations of using H rhamnoides oil, a traditional Chinese herbal medicine derived from sea buckthorn fruit, as a burn treatment. In the study, 151 burn patients received an H rhamnoides oil dressing (changed every other day until wound healing) that was covered with a disinfecting dressing. The dressing reduced swelling and effusion, and alleviated pain, with patients receiving the sea buckthorn dressing experiencing greater apparent exudation reduction, pain reduction, and more rapid epithelial cell growth and wound healing than controls (treated only with Vaseline gauze). The difference between the two groups was statistically significant.21
Conclusion
Sea buckthorn has been used for hundreds if not thousands of years in traditional medical applications, including for dermatologic purposes. Emerging data appear to support the use of this dynamic plant for consideration in dermatologic applications. As is often the case, much more work is necessary in the form of randomized controlled trials to determine the effectiveness of sea buckthorn formulations as well as the most appropriate avenues of research or uses for dermatologic application of this traditionally used botanical agent.
Dr. Baumann is a private practice dermatologist, researcher, author, and entrepreneur in Miami. She founded the division of cosmetic dermatology at the University of Miami in 1997. The third edition of her bestselling textbook, “Cosmetic Dermatology,” was published in 2022. Dr. Baumann has received funding for advisory boards and/or clinical research trials from Allergan, Galderma, Johnson & Johnson, and Burt’s Bees. She is the CEO of Skin Type Solutions, a SaaS company used to generate skin care routines in office and as a e-commerce solution. Write to her at [email protected].
References
1. Teng H et al. J Ethnopharmacol. 2024 Apr 24;324:117809. doi: 10.1016/j.jep.2024.117809.
2. Wang Z et al. Int J Biol Macromol. 2024 Apr;263(Pt 1):130206. doi: 10.1016/j.ijbiomac.2024.130206.
3. Gupta A et al. Int J Low Extrem Wounds. 2005 Jun;4(2):88-92. doi: 10.1177/1534734605277401.
4. Pundir S et al. J Ethnopharmacol. 2021 Feb 10;266:113434. doi: 10.1016/j.jep.2020.113434.
5. Ma QG et al. J Agric Food Chem. 2023 Mar 29;71(12):4769-4788. doi: 10.1021/acs.jafc.2c06916.
6. Poljšak N et al. Phytother Res. 2020 Feb;34(2):254-269. doi: 10.1002/ptr.6524. doi: 10.1002/ptr.6524.
7. Upadhyay NK et al. Evid Based Complement Alternat Med. 2011;2011:659705. doi: 10.1093/ecam/nep189.
8. Suryakumar G, Gupta A. J Ethnopharmacol. 2011 Nov 18;138(2):268-78. doi: 10.1016/j.jep.2011.09.024.
9. Liu K et al. Front Pharmacol. 2022 Jul 8;13:914146. doi: 10.3389/fphar.2022.914146.
10. Akhtar N et al. J Pharm Bioallied Sci. 2010 Jan;2(1):13-7. doi: 10.4103/0975-7406.62698.
11. Ren R et al. RSC Adv. 2020 Dec 17;10(73):44654-44671. doi: 10.1039/d0ra06488b.
12. Ito H et al. Burns. 2014 May;40(3):511-9. doi: 10.1016/j.burns.2013.08.011.
13. Liu X et al. Food Sci Nutr. 2023 Dec 7;12(2):1082-1094. doi: 10.1002/fsn3.3823.
14. Liu X at al. Antioxidants (Basel). 2022 Sep 25;11(10):1900. doi: 10.3390/antiox11101900.
15. Khan BA, Akhtar N. Postepy Dermatol Alergol. 2014 Aug;31(4):229-234. doi: 10.5114/pdia.2014.40934.
16. Khan BA, Akhtar N. Pak J Pharm Sci. 2014 Nov;27(6):1919-22.
17. Khan AB et al. African J Pharm Pharmacol. 2011 Aug;5(8):1092-5.
18. Khan BA, Akhtar N, Braga VA. Trop J Pharm Res. 2012;11(6):955-62.
19. Gęgotek A et al. Antioxidants (Basel). 2018 Aug 23;7(9):110. doi: 10.3390/antiox7090110.
20. Ivanišová E et al. Acta Sci Pol Technol Aliment. 2020 Apr-Jun;19(2):195-205. doi: 10.17306/J.AFS.0809.
21. Wang ZY, Luo XL, He CP. Nan Fang Yi Ke Da Xue Xue Bao. 2006 Jan;26(1):124-5.
A member of the Elaeagnaceae family, Hippophae rhamnoides, better known as sea buckthorn, is a high-altitude wild shrub endemic to Europe and Asia with edible fruits and a lengthy record of use in traditional Chinese medicine.1-6 Used as a health supplement and consumed in the diet throughout the world,5 sea buckthorn berries, seeds, and leaves have been used in traditional medicine to treat burns/injuries, edema, hypertension, inflammation, skin grafts, ulcers, and wounds.4,7
This hardy plant is associated with a wide range of biologic activities, including anti-atherogenic, anti-atopic dermatitis, antibacterial, anticancer, antifungal, anti-inflammatory, antimicrobial, antioxidant, anti-psoriasis, anti-sebum, anti-stress, anti-tumor, cytoprotective, hepatoprotective, immunomodulatory, neuroprotective, radioprotective, and tissue regenerative functions.4,5,8-11
Key Constituents
Functional constituents identified in sea buckthorn include alkaloids, carotenoids, flavonoids, lignans, organic acids, phenolic acids, proanthocyanidins, polyunsaturated acids (including omega-3, -6, -7, and -9), steroids, tannins, terpenoids, and volatile oils, as well as nutritional compounds such as minerals, proteins, and vitamins.4,5,11 Sea buckthorn pericarp oil contains copious amounts of saturated palmitic acid (29%-36%) and omega-7 unsaturated palmitoleic acid (36%-48%), which fosters cutaneous and mucosal epithelialization, as well as linoleic (10%-12%) and oleic (4%-6%) acids.12,6 Significant amounts of carotenoids as well as alpha‐linolenic fatty acid (38%), linoleic (36%), oleic (13%), and palmitic (7%) acids are present in sea buckthorn seed oil.6
Polysaccharides
In an expansive review on the pharmacological activities of sea buckthorn polysaccharides, Teng and colleagues reported in April 2024 that 20 diverse polysaccharides have been culled from sea buckthorn and exhibited various healthy activities, including antioxidant, anti-fatigue, anti-inflammatory, anti-obesity, anti-tumor, hepatoprotective, hypoglycemic, and immunoregulation, and regulation of intestinal flora activities.1
Proanthocyanidins and Anti-Aging
In 2023, Liu and colleagues investigated the anti–skin aging impact of sea buckthorn proanthocyanidins in D-galactose-induced aging in mice given the known free radical scavenging activity of these compounds. They found the proanthocyanidins mitigated D-galactose-induced aging and can augment the total antioxidant capacity of the body. Sea buckthorn proanthocyanidins can further attenuate the effects of skin aging by regulating the TGF-beta1/Smads pathway and MMPs/TIMP system, thus amplifying collagen I and tropoelastin content.13
A year earlier, many of the same investigators assessed the possible protective activity of sea buckthorn proanthocyanidins against cutaneous aging engendered by oxidative stress from hydrogen peroxide. The compounds amplified superoxide dismutase and glutathione antioxidant functions. The extracts also fostered collagen I production in aging human skin fibroblasts via the TGF-beta1/Smads pathway and hindered collagen I degradation by regulating the MMPs/TIMPs system, which maintained extracellular matrix integrity. Senescent cell migration was also promoted with 100 mcg/mL of sea buckthorn proanthocyanidins. The researchers concluded that this sets the stage for investigating how sea buckthorn proanthocyanidins can be incorporated in cosmetic formulations.14 In a separate study, Liu and colleagues demonstrated that sea buckthorn proanthocyanidins can attenuate oxidative damage and protect mitochondrial function.9
Acne and Barrier Functions
The extracts of H rhamnoides and Cassia fistula in a combined formulation were found to be effective in lowering skin sebum content in humans with grade I and grade II acne vulgaris in a 2014 single-blind, randomized, placebo-controlled, split-face study with two groups of 25 patients each (aged 18-37 years).15 Khan and colleagues have also reported that a sea buckthorn oil-in-water emulsion improved barrier function in human skin as tested by a tewameter and corneometer (noninvasive probes) in 13 healthy males with a mean age of 27 ± 4.8 years.16
Anti-Aging, Antioxidant, Antibacterial, Skin-Whitening Activity
Zaman and colleagues reported in 2011 that results from an in vivo study of the effects of a sea buckthorn fruit extract topical cream on stratum corneum water content and transepidermal water loss indicated that the formulation enhanced cell surface integrin expression thus facilitating collagen contraction.17
In 2012, Khan and colleagues reported amelioration in skin elasticity, thus achieving an anti-aging result, from the use of a water-in-oil–based hydroalcoholic cream loaded with fruit extract of H rhamnoides, as measured with a Cutometer.18 The previous year, some of the same researchers reported that the antioxidants and flavonoids found in a topical sea buckthorn formulation could decrease cutaneous melanin and erythema levels.
More recently, Gęgotek and colleagues found that sea buckthorn seed oil prevented redox balance and lipid metabolism disturbances in skin fibroblasts and keratinocytes caused by UVA or UVB. They suggested that such findings point to the potential of this natural agent to confer anti-inflammatory properties and photoprotection to the skin.19
In 2020, Ivanišová and colleagues investigated the antioxidant and antimicrobial activities of H rhamnoides 100% oil, 100% juice, dry berries, and tea (dry berries, leaves, and twigs). They found that all of the studied sea buckthorn products displayed high antioxidant activity (identified through DPPH radical scavenging and molybdenum reducing antioxidant power tests). Sea buckthorn juice contained the highest total content of polyphenols, flavonoids, and carotenoids. All of the tested products also exhibited substantial antibacterial activity against the tested microbes.20
Burns and Wound Healing
In a preclinical study of the effects of sea buckthorn leaf extracts on wound healing in albino rats using an excision-punch wound model in 2005, Gupta and colleagues found that twice daily topical application of the aqueous leaf extract fostered wound healing. This was indicated by higher hydroxyproline and protein levels, a diminished wound area, and lower lipid peroxide levels. The investigators suggested that sea buckthorn may facilitate wound healing at least in part because of elevated antioxidant activity in the granulation tissue.3
A year later, Wang and colleagues reported on observations of using H rhamnoides oil, a traditional Chinese herbal medicine derived from sea buckthorn fruit, as a burn treatment. In the study, 151 burn patients received an H rhamnoides oil dressing (changed every other day until wound healing) that was covered with a disinfecting dressing. The dressing reduced swelling and effusion, and alleviated pain, with patients receiving the sea buckthorn dressing experiencing greater apparent exudation reduction, pain reduction, and more rapid epithelial cell growth and wound healing than controls (treated only with Vaseline gauze). The difference between the two groups was statistically significant.21
Conclusion
Sea buckthorn has been used for hundreds if not thousands of years in traditional medical applications, including for dermatologic purposes. Emerging data appear to support the use of this dynamic plant for consideration in dermatologic applications. As is often the case, much more work is necessary in the form of randomized controlled trials to determine the effectiveness of sea buckthorn formulations as well as the most appropriate avenues of research or uses for dermatologic application of this traditionally used botanical agent.
Dr. Baumann is a private practice dermatologist, researcher, author, and entrepreneur in Miami. She founded the division of cosmetic dermatology at the University of Miami in 1997. The third edition of her bestselling textbook, “Cosmetic Dermatology,” was published in 2022. Dr. Baumann has received funding for advisory boards and/or clinical research trials from Allergan, Galderma, Johnson & Johnson, and Burt’s Bees. She is the CEO of Skin Type Solutions, a SaaS company used to generate skin care routines in office and as a e-commerce solution. Write to her at [email protected].
References
1. Teng H et al. J Ethnopharmacol. 2024 Apr 24;324:117809. doi: 10.1016/j.jep.2024.117809.
2. Wang Z et al. Int J Biol Macromol. 2024 Apr;263(Pt 1):130206. doi: 10.1016/j.ijbiomac.2024.130206.
3. Gupta A et al. Int J Low Extrem Wounds. 2005 Jun;4(2):88-92. doi: 10.1177/1534734605277401.
4. Pundir S et al. J Ethnopharmacol. 2021 Feb 10;266:113434. doi: 10.1016/j.jep.2020.113434.
5. Ma QG et al. J Agric Food Chem. 2023 Mar 29;71(12):4769-4788. doi: 10.1021/acs.jafc.2c06916.
6. Poljšak N et al. Phytother Res. 2020 Feb;34(2):254-269. doi: 10.1002/ptr.6524. doi: 10.1002/ptr.6524.
7. Upadhyay NK et al. Evid Based Complement Alternat Med. 2011;2011:659705. doi: 10.1093/ecam/nep189.
8. Suryakumar G, Gupta A. J Ethnopharmacol. 2011 Nov 18;138(2):268-78. doi: 10.1016/j.jep.2011.09.024.
9. Liu K et al. Front Pharmacol. 2022 Jul 8;13:914146. doi: 10.3389/fphar.2022.914146.
10. Akhtar N et al. J Pharm Bioallied Sci. 2010 Jan;2(1):13-7. doi: 10.4103/0975-7406.62698.
11. Ren R et al. RSC Adv. 2020 Dec 17;10(73):44654-44671. doi: 10.1039/d0ra06488b.
12. Ito H et al. Burns. 2014 May;40(3):511-9. doi: 10.1016/j.burns.2013.08.011.
13. Liu X et al. Food Sci Nutr. 2023 Dec 7;12(2):1082-1094. doi: 10.1002/fsn3.3823.
14. Liu X at al. Antioxidants (Basel). 2022 Sep 25;11(10):1900. doi: 10.3390/antiox11101900.
15. Khan BA, Akhtar N. Postepy Dermatol Alergol. 2014 Aug;31(4):229-234. doi: 10.5114/pdia.2014.40934.
16. Khan BA, Akhtar N. Pak J Pharm Sci. 2014 Nov;27(6):1919-22.
17. Khan AB et al. African J Pharm Pharmacol. 2011 Aug;5(8):1092-5.
18. Khan BA, Akhtar N, Braga VA. Trop J Pharm Res. 2012;11(6):955-62.
19. Gęgotek A et al. Antioxidants (Basel). 2018 Aug 23;7(9):110. doi: 10.3390/antiox7090110.
20. Ivanišová E et al. Acta Sci Pol Technol Aliment. 2020 Apr-Jun;19(2):195-205. doi: 10.17306/J.AFS.0809.
21. Wang ZY, Luo XL, He CP. Nan Fang Yi Ke Da Xue Xue Bao. 2006 Jan;26(1):124-5.
Skin Fungal Infections Increasing in the United States
TOPLINE:
. Tinea unguium, tinea pedis, and tinea corporis were among the most common infections.
METHODOLOGY:
- Researchers analyzed data from the National Ambulatory Medical Care Survey and National Hospital Ambulatory Medical Care Survey from 2005 to 2016, to evaluate trends in the prevalence of SCFIs during this period.
- The analysis included over 13 billion ambulatory visits to nonfederally funded community, office-based physician practices, and emergency or outpatient departments in the United States, with an estimated 1,104,258,333 annual average.
- The Jonckheere-Terpstra nonparametric test for trend was used to determine the pattern of SCFI prevalence over the 12-year period.
TAKEAWAY:
- SCFIs constituted approximately 0.54% of all annual ambulatory visits, with an estimated 6,001,852 visits for SCFIs per year and over 72 million total visits for the infections during the study period.
- Tinea unguium, tinea pedis, and tinea corporis were the most common infections, comprising 20.5%, 12.2%, and 12.0% of the total visits, respectively.
- Researchers noted an increasing trend in annual SCFIs (P = .03).
IN PRACTICE:
“We observed a high burden of SCFIs among outpatient visits in the United States and an increasing trend in their prevalence,” the authors wrote. These results, they added, “highlight the importance of healthcare providers being able to identify, treat, and, when necessary, refer patients with SCFIs, as a high burden of disease is associated with a significant negative impact on the individual and population levels.”
SOURCE:
The study was co-led by Sarah L. Spaulding, BS, and A. Mitchel Wride, BA, from the Yale School of Medicine, New Haven, Connecticut, and was published online October 30 in the Journal of the American Academy of Dermatology.
LIMITATIONS:
The authors did not list any study limitations.
DISCLOSURES:
The lead authors were supported by Yale School of Medicine Medical Student Research Fellowships. Two other authors declared receiving consulting fees, research funding, and licensing fees outside the submitted work and also served on a data and safety monitoring board for Advarra Inc.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
TOPLINE:
. Tinea unguium, tinea pedis, and tinea corporis were among the most common infections.
METHODOLOGY:
- Researchers analyzed data from the National Ambulatory Medical Care Survey and National Hospital Ambulatory Medical Care Survey from 2005 to 2016, to evaluate trends in the prevalence of SCFIs during this period.
- The analysis included over 13 billion ambulatory visits to nonfederally funded community, office-based physician practices, and emergency or outpatient departments in the United States, with an estimated 1,104,258,333 annual average.
- The Jonckheere-Terpstra nonparametric test for trend was used to determine the pattern of SCFI prevalence over the 12-year period.
TAKEAWAY:
- SCFIs constituted approximately 0.54% of all annual ambulatory visits, with an estimated 6,001,852 visits for SCFIs per year and over 72 million total visits for the infections during the study period.
- Tinea unguium, tinea pedis, and tinea corporis were the most common infections, comprising 20.5%, 12.2%, and 12.0% of the total visits, respectively.
- Researchers noted an increasing trend in annual SCFIs (P = .03).
IN PRACTICE:
“We observed a high burden of SCFIs among outpatient visits in the United States and an increasing trend in their prevalence,” the authors wrote. These results, they added, “highlight the importance of healthcare providers being able to identify, treat, and, when necessary, refer patients with SCFIs, as a high burden of disease is associated with a significant negative impact on the individual and population levels.”
SOURCE:
The study was co-led by Sarah L. Spaulding, BS, and A. Mitchel Wride, BA, from the Yale School of Medicine, New Haven, Connecticut, and was published online October 30 in the Journal of the American Academy of Dermatology.
LIMITATIONS:
The authors did not list any study limitations.
DISCLOSURES:
The lead authors were supported by Yale School of Medicine Medical Student Research Fellowships. Two other authors declared receiving consulting fees, research funding, and licensing fees outside the submitted work and also served on a data and safety monitoring board for Advarra Inc.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
TOPLINE:
. Tinea unguium, tinea pedis, and tinea corporis were among the most common infections.
METHODOLOGY:
- Researchers analyzed data from the National Ambulatory Medical Care Survey and National Hospital Ambulatory Medical Care Survey from 2005 to 2016, to evaluate trends in the prevalence of SCFIs during this period.
- The analysis included over 13 billion ambulatory visits to nonfederally funded community, office-based physician practices, and emergency or outpatient departments in the United States, with an estimated 1,104,258,333 annual average.
- The Jonckheere-Terpstra nonparametric test for trend was used to determine the pattern of SCFI prevalence over the 12-year period.
TAKEAWAY:
- SCFIs constituted approximately 0.54% of all annual ambulatory visits, with an estimated 6,001,852 visits for SCFIs per year and over 72 million total visits for the infections during the study period.
- Tinea unguium, tinea pedis, and tinea corporis were the most common infections, comprising 20.5%, 12.2%, and 12.0% of the total visits, respectively.
- Researchers noted an increasing trend in annual SCFIs (P = .03).
IN PRACTICE:
“We observed a high burden of SCFIs among outpatient visits in the United States and an increasing trend in their prevalence,” the authors wrote. These results, they added, “highlight the importance of healthcare providers being able to identify, treat, and, when necessary, refer patients with SCFIs, as a high burden of disease is associated with a significant negative impact on the individual and population levels.”
SOURCE:
The study was co-led by Sarah L. Spaulding, BS, and A. Mitchel Wride, BA, from the Yale School of Medicine, New Haven, Connecticut, and was published online October 30 in the Journal of the American Academy of Dermatology.
LIMITATIONS:
The authors did not list any study limitations.
DISCLOSURES:
The lead authors were supported by Yale School of Medicine Medical Student Research Fellowships. Two other authors declared receiving consulting fees, research funding, and licensing fees outside the submitted work and also served on a data and safety monitoring board for Advarra Inc.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
Study Finds Link to Increased Risk for Bulimia, Binge Eating and HS
“Clinicians should actively screen for eating disorders,” particularly bulimia nervosa and binge eating disorder, in patients with HS,” lead study author Christopher Guirguis, DMD, a student at Georgetown University School of Medicine, Washington, DC, told this news organization in advance of the annual Symposium on Hidradenitis Suppurative Advances, where the study was presented during an oral abstract session. “The significant psychological burden in these patients requires a holistic approach that integrates both dermatologic and psychosocial care. Addressing their mental health needs is essential for improving overall patient outcomes and quality of life,” he added.
In collaboration with fellow Georgetown medical student and first author Lauren Chin and Mikael Horissian, MD, a dermatologist and director of the HS Clinic at Gesinger Health System, Danville, Pennsylvania, Guirguis drew from the National Institutes of Health’s All of Us Research Program to identify 1653 individuals with a diagnosis of HS and a control group of 8265 individuals without a diagnosis of HS. They used the Observational Medical Outcomes Partnership to identify anorexia nervosa, bulimia nervosa, body dysmorphic disorder, binge eating disorder, and eating disorder, unspecified. Obsessive-compulsive disorder (OCD) was also included because of its association with bulimia. They used statistical models to compare cohorts and comorbidities. “What makes this work unique is its focus on the link between HS and eating disorders, a relationship previously underexplored,” he said.
The mean age of the overall study cohort was 46.8 years, and 78.6% were female. Univariate analysis revealed that, compared with controls, individuals in the HS cohort showed significantly increased diagnoses of bulimia, binge eating disorder, OCD, and eating disorder, unspecified, by 2.6, 5.48, 2.50, and 2.43 times, respectively (P < .05 for all associations). After adjusting for age, race, sex, and ethnicity, the researchers observed that patients with HS were 4.46 times as likely to have a diagnosis of binge eating disorder and 3.51 times as likely to have a diagnosis of bulimia as those who did not have HS (P < .05 for both associations).
Guirguis said that the absence of body dysmorphic disorder diagnoses in the HS cohort was unexpected. “Given HS’s known association with body image issues, we anticipated a higher prevalence of BDD,” he said. “This discrepancy may reflect underreporting or diagnostic overshadowing, where the physical symptoms of HS dominate clinical attention, potentially masking or complicating the identification of psychological conditions like BDD.”
He acknowledged certain limitations of the study, including the potential for variations in documentation practices in the database. “Additionally, there may be bias due to underrepresentation of certain demographic groups or underreporting of psychological comorbidities, which could influence the findings.”
Patricia M. Richey, MD, assistant professor of dermatology, at Boston University School of Medicine in Massachusetts, who was asked to comment on the study, said the results “should affect how physicians discuss lifestyle recommendations in those already at increased risk of psychiatric disease and disrupted body image.” The findings should also “prompt physicians to screen this patient population more thoroughly for eating disorders as we know they are an underrecognized and often undertreated entity,” she added.
Neither the study authors nor Richey reported having relevant disclosures.
A version of this article appeared on Medscape.com.
“Clinicians should actively screen for eating disorders,” particularly bulimia nervosa and binge eating disorder, in patients with HS,” lead study author Christopher Guirguis, DMD, a student at Georgetown University School of Medicine, Washington, DC, told this news organization in advance of the annual Symposium on Hidradenitis Suppurative Advances, where the study was presented during an oral abstract session. “The significant psychological burden in these patients requires a holistic approach that integrates both dermatologic and psychosocial care. Addressing their mental health needs is essential for improving overall patient outcomes and quality of life,” he added.
In collaboration with fellow Georgetown medical student and first author Lauren Chin and Mikael Horissian, MD, a dermatologist and director of the HS Clinic at Gesinger Health System, Danville, Pennsylvania, Guirguis drew from the National Institutes of Health’s All of Us Research Program to identify 1653 individuals with a diagnosis of HS and a control group of 8265 individuals without a diagnosis of HS. They used the Observational Medical Outcomes Partnership to identify anorexia nervosa, bulimia nervosa, body dysmorphic disorder, binge eating disorder, and eating disorder, unspecified. Obsessive-compulsive disorder (OCD) was also included because of its association with bulimia. They used statistical models to compare cohorts and comorbidities. “What makes this work unique is its focus on the link between HS and eating disorders, a relationship previously underexplored,” he said.
The mean age of the overall study cohort was 46.8 years, and 78.6% were female. Univariate analysis revealed that, compared with controls, individuals in the HS cohort showed significantly increased diagnoses of bulimia, binge eating disorder, OCD, and eating disorder, unspecified, by 2.6, 5.48, 2.50, and 2.43 times, respectively (P < .05 for all associations). After adjusting for age, race, sex, and ethnicity, the researchers observed that patients with HS were 4.46 times as likely to have a diagnosis of binge eating disorder and 3.51 times as likely to have a diagnosis of bulimia as those who did not have HS (P < .05 for both associations).
Guirguis said that the absence of body dysmorphic disorder diagnoses in the HS cohort was unexpected. “Given HS’s known association with body image issues, we anticipated a higher prevalence of BDD,” he said. “This discrepancy may reflect underreporting or diagnostic overshadowing, where the physical symptoms of HS dominate clinical attention, potentially masking or complicating the identification of psychological conditions like BDD.”
He acknowledged certain limitations of the study, including the potential for variations in documentation practices in the database. “Additionally, there may be bias due to underrepresentation of certain demographic groups or underreporting of psychological comorbidities, which could influence the findings.”
Patricia M. Richey, MD, assistant professor of dermatology, at Boston University School of Medicine in Massachusetts, who was asked to comment on the study, said the results “should affect how physicians discuss lifestyle recommendations in those already at increased risk of psychiatric disease and disrupted body image.” The findings should also “prompt physicians to screen this patient population more thoroughly for eating disorders as we know they are an underrecognized and often undertreated entity,” she added.
Neither the study authors nor Richey reported having relevant disclosures.
A version of this article appeared on Medscape.com.
“Clinicians should actively screen for eating disorders,” particularly bulimia nervosa and binge eating disorder, in patients with HS,” lead study author Christopher Guirguis, DMD, a student at Georgetown University School of Medicine, Washington, DC, told this news organization in advance of the annual Symposium on Hidradenitis Suppurative Advances, where the study was presented during an oral abstract session. “The significant psychological burden in these patients requires a holistic approach that integrates both dermatologic and psychosocial care. Addressing their mental health needs is essential for improving overall patient outcomes and quality of life,” he added.
In collaboration with fellow Georgetown medical student and first author Lauren Chin and Mikael Horissian, MD, a dermatologist and director of the HS Clinic at Gesinger Health System, Danville, Pennsylvania, Guirguis drew from the National Institutes of Health’s All of Us Research Program to identify 1653 individuals with a diagnosis of HS and a control group of 8265 individuals without a diagnosis of HS. They used the Observational Medical Outcomes Partnership to identify anorexia nervosa, bulimia nervosa, body dysmorphic disorder, binge eating disorder, and eating disorder, unspecified. Obsessive-compulsive disorder (OCD) was also included because of its association with bulimia. They used statistical models to compare cohorts and comorbidities. “What makes this work unique is its focus on the link between HS and eating disorders, a relationship previously underexplored,” he said.
The mean age of the overall study cohort was 46.8 years, and 78.6% were female. Univariate analysis revealed that, compared with controls, individuals in the HS cohort showed significantly increased diagnoses of bulimia, binge eating disorder, OCD, and eating disorder, unspecified, by 2.6, 5.48, 2.50, and 2.43 times, respectively (P < .05 for all associations). After adjusting for age, race, sex, and ethnicity, the researchers observed that patients with HS were 4.46 times as likely to have a diagnosis of binge eating disorder and 3.51 times as likely to have a diagnosis of bulimia as those who did not have HS (P < .05 for both associations).
Guirguis said that the absence of body dysmorphic disorder diagnoses in the HS cohort was unexpected. “Given HS’s known association with body image issues, we anticipated a higher prevalence of BDD,” he said. “This discrepancy may reflect underreporting or diagnostic overshadowing, where the physical symptoms of HS dominate clinical attention, potentially masking or complicating the identification of psychological conditions like BDD.”
He acknowledged certain limitations of the study, including the potential for variations in documentation practices in the database. “Additionally, there may be bias due to underrepresentation of certain demographic groups or underreporting of psychological comorbidities, which could influence the findings.”
Patricia M. Richey, MD, assistant professor of dermatology, at Boston University School of Medicine in Massachusetts, who was asked to comment on the study, said the results “should affect how physicians discuss lifestyle recommendations in those already at increased risk of psychiatric disease and disrupted body image.” The findings should also “prompt physicians to screen this patient population more thoroughly for eating disorders as we know they are an underrecognized and often undertreated entity,” she added.
Neither the study authors nor Richey reported having relevant disclosures.
A version of this article appeared on Medscape.com.
Effects of Bimekizumab Durable for HS Through One Year
AMSTERDAM — The monoclonal antibody according to new data from an open-label extension period.
“Efficacy and health-related quality-of-life outcomes were maintained through 2 years of treatment,” study presenter Christos C. Zouboulis, MD, professor of dermatology, venereology, and allergology, Brandenburg Medical School Theodor Fontane, Dessau, Germany, said at the European Academy of Dermatology and Venereology (EADV) 2024 Congress.
“No new safety signals were observed,” he added. “These data highlight the durability and consistency of bimekizumab treatment in patients with moderate to severe hidradenitis suppurativa,” Zouboulis concluded.
Efficacy Maintained
“This is the type of long-term data that clinicians hope to see in large phase 3 trials for hidradenitis suppurativa medications,” commented Jennifer L. Hsiao, MD, clinical associate professor of dermatology, University of Southern California, Los Angeles, who was not involved in the study.
She told this news organization that, beyond maintained improvement of patient-reported quality of life, the results are “raising the bar in terms of measuring treatment success,” with over three quarters of patients achieving a high level of response on the Hidradenitis Suppurativa Clinical Response (HiSCR) scale at the final 96-week follow-up.
“Clinicians and patients have struggled with maintaining treatment efficacy over time with the first [Food and Drug Administration]–approved class of biologics for hidradenitis suppurativa — TNF [tumor necrosis factor]–alpha antagonists,” Hsiao said. She emphasized that sustained treatment efficacy will reduce the need for continued treatment switching and “hopefully improve treatment adherence.”
“It was also helpful to see that, consistent with studies of bimekizumab in psoriasis, rates of oral candidiasis appear to decrease with prolonged exposure over 2 years, though as with any open-label extension study, study dropout is a limitation,” she said.
“The availability of long-term efficacy and safety data, such as those shown in this study, will help guide shared decision-making discussions with our patients.” Overall, Hsiao believes there is “much to be excited about in the field of hidradenitis suppurativa, with a robust pipeline of potential treatments.”
One-Year Extension Study
HS is a “chronic and debilitating inflammatory skin disease,” Zouboulis told the audience. He noted that interleukin (IL)–17F and IL-17A are highly expressed in lesional skin and play a role in the disease immunopathogenesis.
Bimekizumab is a humanized immunoglobulin G1 monoclonal antibody that selectively inhibits both IL-17F and IL-17A. It has previously demonstrated clinically meaningful improvements in patients with moderate to severe HS in the phase 3 BE HEARD I and BE HEARD II trials evaluating several dosing regimens.
Zouboulis said the current analysis combines data from the two phase 3 studies with the BE HEARD EXT open-label extension study, in which patients from both trials were continued on bimekizumab 320 mg every 2 weeks.
Of the 1014 patients initially enrolled in the two trials, 556 continued into the open-label extension. Their average age was 36.6 years, and 53.8% were women. The majority (80.6%) were White. Of the 556 patients enrolled in the extension, 446 completed the 1-year extension study.
The average draining tunnel count at baseline was 3.8, and 54.5% had Hurley stage II disease; the remaining 45.5% had stage III disease. The mean total Dermatology Life Quality Index (DLQI) score at baseline was 11.0, indicating the HS was having a very large impact on the patients’ lives.
After the 16-week initial treatment period and the maintenance treatment period out to 48 weeks, 64.0% of patients achieved HiSCR75, indicating at least a 75% reduction from baseline in the total abscess and inflammatory nodule count, rising to 77.1% at the end of the open-label extension, after a total follow-up of 96 weeks.
HiSCR100 scores, indicating a 100% reduction in total abscess and inflammatory nodule counts, were achieved by 30.2% of 556 patients after 48 weeks and 44.2% of 446 at the 96-week follow-up.
These findings were mirrored by substantial reductions on the International HS Severity Score System, with a 70.3% reduction over baseline at 48 weeks and a 79.8% reduction at the final follow-up.
There were also “clinically meaningful” reductions in the total draining tunnel count at 1 year that were further reduced at 2 years, Zouboulis reported, at a 57.5% reduction over baseline, increasing to 73.7% by 96 weeks. The mean draining tunnel count at the end of follow-up was 1.1.
Over the full 96 weeks, the mean DLQI score reduced from 11.0 to 4.7, with 33.9% of patients achieving a score of 0 or 1 on the scale, which he said is basically patients saying: “I don’t have disease now.”
Finally, the safety data showed that there were “no differences compared to what we knew before,” Zouboulis said, with the most common treatment-related adverse events being hidradenitis, coronavirus infection, and oral candidiasis. There were few serious and severe treatment-related adverse events, and few that led to treatment discontinuation.
The study was funded by UCB.Zouboulis declared relationships with AstraZeneca, Boehringer Ingelheim, Brandenburg Medical School Theodor Fontane, EAD, European Union, German Federal Ministry of Education and Research, GSK, InflaRx, MSD, Novartis, Relaxera, UCB, Almirall, Boehringer Ingelheim, Eli Lilly, Idorsia, Incyte, L’Oréal, NAOS-BIODERMA, Pfizer, PM, Sanofi. Hsiao is on the board of directors for the Hidradenitis Suppurativa Foundation and has declared relationships with AbbVie, Aclaris Therapeutics, Amgen, Boehringer Ingelheim, Incyte, Novartis, Sanofi-Regeneron, and UCB.
A version of this article appeared on Medscape.com.
AMSTERDAM — The monoclonal antibody according to new data from an open-label extension period.
“Efficacy and health-related quality-of-life outcomes were maintained through 2 years of treatment,” study presenter Christos C. Zouboulis, MD, professor of dermatology, venereology, and allergology, Brandenburg Medical School Theodor Fontane, Dessau, Germany, said at the European Academy of Dermatology and Venereology (EADV) 2024 Congress.
“No new safety signals were observed,” he added. “These data highlight the durability and consistency of bimekizumab treatment in patients with moderate to severe hidradenitis suppurativa,” Zouboulis concluded.
Efficacy Maintained
“This is the type of long-term data that clinicians hope to see in large phase 3 trials for hidradenitis suppurativa medications,” commented Jennifer L. Hsiao, MD, clinical associate professor of dermatology, University of Southern California, Los Angeles, who was not involved in the study.
She told this news organization that, beyond maintained improvement of patient-reported quality of life, the results are “raising the bar in terms of measuring treatment success,” with over three quarters of patients achieving a high level of response on the Hidradenitis Suppurativa Clinical Response (HiSCR) scale at the final 96-week follow-up.
“Clinicians and patients have struggled with maintaining treatment efficacy over time with the first [Food and Drug Administration]–approved class of biologics for hidradenitis suppurativa — TNF [tumor necrosis factor]–alpha antagonists,” Hsiao said. She emphasized that sustained treatment efficacy will reduce the need for continued treatment switching and “hopefully improve treatment adherence.”
“It was also helpful to see that, consistent with studies of bimekizumab in psoriasis, rates of oral candidiasis appear to decrease with prolonged exposure over 2 years, though as with any open-label extension study, study dropout is a limitation,” she said.
“The availability of long-term efficacy and safety data, such as those shown in this study, will help guide shared decision-making discussions with our patients.” Overall, Hsiao believes there is “much to be excited about in the field of hidradenitis suppurativa, with a robust pipeline of potential treatments.”
One-Year Extension Study
HS is a “chronic and debilitating inflammatory skin disease,” Zouboulis told the audience. He noted that interleukin (IL)–17F and IL-17A are highly expressed in lesional skin and play a role in the disease immunopathogenesis.
Bimekizumab is a humanized immunoglobulin G1 monoclonal antibody that selectively inhibits both IL-17F and IL-17A. It has previously demonstrated clinically meaningful improvements in patients with moderate to severe HS in the phase 3 BE HEARD I and BE HEARD II trials evaluating several dosing regimens.
Zouboulis said the current analysis combines data from the two phase 3 studies with the BE HEARD EXT open-label extension study, in which patients from both trials were continued on bimekizumab 320 mg every 2 weeks.
Of the 1014 patients initially enrolled in the two trials, 556 continued into the open-label extension. Their average age was 36.6 years, and 53.8% were women. The majority (80.6%) were White. Of the 556 patients enrolled in the extension, 446 completed the 1-year extension study.
The average draining tunnel count at baseline was 3.8, and 54.5% had Hurley stage II disease; the remaining 45.5% had stage III disease. The mean total Dermatology Life Quality Index (DLQI) score at baseline was 11.0, indicating the HS was having a very large impact on the patients’ lives.
After the 16-week initial treatment period and the maintenance treatment period out to 48 weeks, 64.0% of patients achieved HiSCR75, indicating at least a 75% reduction from baseline in the total abscess and inflammatory nodule count, rising to 77.1% at the end of the open-label extension, after a total follow-up of 96 weeks.
HiSCR100 scores, indicating a 100% reduction in total abscess and inflammatory nodule counts, were achieved by 30.2% of 556 patients after 48 weeks and 44.2% of 446 at the 96-week follow-up.
These findings were mirrored by substantial reductions on the International HS Severity Score System, with a 70.3% reduction over baseline at 48 weeks and a 79.8% reduction at the final follow-up.
There were also “clinically meaningful” reductions in the total draining tunnel count at 1 year that were further reduced at 2 years, Zouboulis reported, at a 57.5% reduction over baseline, increasing to 73.7% by 96 weeks. The mean draining tunnel count at the end of follow-up was 1.1.
Over the full 96 weeks, the mean DLQI score reduced from 11.0 to 4.7, with 33.9% of patients achieving a score of 0 or 1 on the scale, which he said is basically patients saying: “I don’t have disease now.”
Finally, the safety data showed that there were “no differences compared to what we knew before,” Zouboulis said, with the most common treatment-related adverse events being hidradenitis, coronavirus infection, and oral candidiasis. There were few serious and severe treatment-related adverse events, and few that led to treatment discontinuation.
The study was funded by UCB.Zouboulis declared relationships with AstraZeneca, Boehringer Ingelheim, Brandenburg Medical School Theodor Fontane, EAD, European Union, German Federal Ministry of Education and Research, GSK, InflaRx, MSD, Novartis, Relaxera, UCB, Almirall, Boehringer Ingelheim, Eli Lilly, Idorsia, Incyte, L’Oréal, NAOS-BIODERMA, Pfizer, PM, Sanofi. Hsiao is on the board of directors for the Hidradenitis Suppurativa Foundation and has declared relationships with AbbVie, Aclaris Therapeutics, Amgen, Boehringer Ingelheim, Incyte, Novartis, Sanofi-Regeneron, and UCB.
A version of this article appeared on Medscape.com.
AMSTERDAM — The monoclonal antibody according to new data from an open-label extension period.
“Efficacy and health-related quality-of-life outcomes were maintained through 2 years of treatment,” study presenter Christos C. Zouboulis, MD, professor of dermatology, venereology, and allergology, Brandenburg Medical School Theodor Fontane, Dessau, Germany, said at the European Academy of Dermatology and Venereology (EADV) 2024 Congress.
“No new safety signals were observed,” he added. “These data highlight the durability and consistency of bimekizumab treatment in patients with moderate to severe hidradenitis suppurativa,” Zouboulis concluded.
Efficacy Maintained
“This is the type of long-term data that clinicians hope to see in large phase 3 trials for hidradenitis suppurativa medications,” commented Jennifer L. Hsiao, MD, clinical associate professor of dermatology, University of Southern California, Los Angeles, who was not involved in the study.
She told this news organization that, beyond maintained improvement of patient-reported quality of life, the results are “raising the bar in terms of measuring treatment success,” with over three quarters of patients achieving a high level of response on the Hidradenitis Suppurativa Clinical Response (HiSCR) scale at the final 96-week follow-up.
“Clinicians and patients have struggled with maintaining treatment efficacy over time with the first [Food and Drug Administration]–approved class of biologics for hidradenitis suppurativa — TNF [tumor necrosis factor]–alpha antagonists,” Hsiao said. She emphasized that sustained treatment efficacy will reduce the need for continued treatment switching and “hopefully improve treatment adherence.”
“It was also helpful to see that, consistent with studies of bimekizumab in psoriasis, rates of oral candidiasis appear to decrease with prolonged exposure over 2 years, though as with any open-label extension study, study dropout is a limitation,” she said.
“The availability of long-term efficacy and safety data, such as those shown in this study, will help guide shared decision-making discussions with our patients.” Overall, Hsiao believes there is “much to be excited about in the field of hidradenitis suppurativa, with a robust pipeline of potential treatments.”
One-Year Extension Study
HS is a “chronic and debilitating inflammatory skin disease,” Zouboulis told the audience. He noted that interleukin (IL)–17F and IL-17A are highly expressed in lesional skin and play a role in the disease immunopathogenesis.
Bimekizumab is a humanized immunoglobulin G1 monoclonal antibody that selectively inhibits both IL-17F and IL-17A. It has previously demonstrated clinically meaningful improvements in patients with moderate to severe HS in the phase 3 BE HEARD I and BE HEARD II trials evaluating several dosing regimens.
Zouboulis said the current analysis combines data from the two phase 3 studies with the BE HEARD EXT open-label extension study, in which patients from both trials were continued on bimekizumab 320 mg every 2 weeks.
Of the 1014 patients initially enrolled in the two trials, 556 continued into the open-label extension. Their average age was 36.6 years, and 53.8% were women. The majority (80.6%) were White. Of the 556 patients enrolled in the extension, 446 completed the 1-year extension study.
The average draining tunnel count at baseline was 3.8, and 54.5% had Hurley stage II disease; the remaining 45.5% had stage III disease. The mean total Dermatology Life Quality Index (DLQI) score at baseline was 11.0, indicating the HS was having a very large impact on the patients’ lives.
After the 16-week initial treatment period and the maintenance treatment period out to 48 weeks, 64.0% of patients achieved HiSCR75, indicating at least a 75% reduction from baseline in the total abscess and inflammatory nodule count, rising to 77.1% at the end of the open-label extension, after a total follow-up of 96 weeks.
HiSCR100 scores, indicating a 100% reduction in total abscess and inflammatory nodule counts, were achieved by 30.2% of 556 patients after 48 weeks and 44.2% of 446 at the 96-week follow-up.
These findings were mirrored by substantial reductions on the International HS Severity Score System, with a 70.3% reduction over baseline at 48 weeks and a 79.8% reduction at the final follow-up.
There were also “clinically meaningful” reductions in the total draining tunnel count at 1 year that were further reduced at 2 years, Zouboulis reported, at a 57.5% reduction over baseline, increasing to 73.7% by 96 weeks. The mean draining tunnel count at the end of follow-up was 1.1.
Over the full 96 weeks, the mean DLQI score reduced from 11.0 to 4.7, with 33.9% of patients achieving a score of 0 or 1 on the scale, which he said is basically patients saying: “I don’t have disease now.”
Finally, the safety data showed that there were “no differences compared to what we knew before,” Zouboulis said, with the most common treatment-related adverse events being hidradenitis, coronavirus infection, and oral candidiasis. There were few serious and severe treatment-related adverse events, and few that led to treatment discontinuation.
The study was funded by UCB.Zouboulis declared relationships with AstraZeneca, Boehringer Ingelheim, Brandenburg Medical School Theodor Fontane, EAD, European Union, German Federal Ministry of Education and Research, GSK, InflaRx, MSD, Novartis, Relaxera, UCB, Almirall, Boehringer Ingelheim, Eli Lilly, Idorsia, Incyte, L’Oréal, NAOS-BIODERMA, Pfizer, PM, Sanofi. Hsiao is on the board of directors for the Hidradenitis Suppurativa Foundation and has declared relationships with AbbVie, Aclaris Therapeutics, Amgen, Boehringer Ingelheim, Incyte, Novartis, Sanofi-Regeneron, and UCB.
A version of this article appeared on Medscape.com.
FROM EADV 2024
Men Wanted: New Efforts to Attract Male Nurses
Only 12% of the nurses providing patient care at hospitals and health clinics today are men. Although the percentage of nurses has increased — men made up just 2.7% of nurses in 1970 — nursing is still considered a “pink collar” profession, a female-dominated field.
“We’ve made strides over the last couple of decades, but [the number of men pursuing nursing careers] is leveling out,” said Jason Dunne, DNP, MSN, RN, chief academic officer at the Arizona College of Nursing, Phoenix. “There continues to be persistent gender stereotypes that [have] discouraged men from entering the profession.”
“The nursing shortage is very real,” Dunne said. “We need to be highly focused on the shortage and look at opportunities to bring diversity into the profession, and one big way to solve it is bringing more men into nursing.”
Representation Matters
Colleges recognize the need to diversify their nursing student population and have turned their attention to increasing the number of men attending informational sessions and career days. Dunne believes, “There is a general lack of awareness of nursing as a career choice [for men].”
The Nursing Consortium of Florida hosts a “Day in the Life of a Nurse” program to introduce high school students to nursing careers, and the University of Virginia School of Nursing invites male nursing students to speak at educational events to promote workforce diversity.
“When I was growing up, the males wouldn’t have been included in those sessions,” said Melissa Gilbert Gomes, PhD, APRN, PMHNP-BC, FNAP, FAAN, associate dean for diversity, equity, and inclusion at the University of Virginia School of Nursing, Charlottesville, Virginia. “It was nice to see their interest and to have a male student there for them to ask questions and to help them see that this could be a place for them.”
Nursing schools have also engaged in other efforts to encourage more men to consider nursing careers, from highlighting male nurses in marketing materials and engaging with men at career fairs to updating course curriculum to include content on men’s health and connecting male nursing students with men in nursing faculty or clinical settings.
Focusing on nursing as a lucrative career choice could also attract more men to the profession. On average, male registered nurses (RNs) make $7300 per year more than their female counterparts due to the gender pay gap. The median wage for male RNs in acute care, cardiology, and perioperative specialties is $90,000 annually.
At the University of Virginia School of Nursing, which the American Association for Men in Nursing (AAMN) named “Best School for Men in Nursing” in 2023, 20% of nursing students are men.
The school has a Men Advancing Nursing club and is in the process of chartering a new AAMN chapter. The goal, according to Gomes, is to create an environment where male nursing students feel represented and supported.
“Valuing the perspective that men bring [to nursing] is important,” she said. “Coming together [and] having that camaraderie and intrinsic motivation to specifically speak to areas that impact men ... is important.”
Promoting Patient Care
Highlighting the diversity of career options within the nursing profession is also essential. RNs can pursue careers in specialties ranging from pediatrics, orthopedics, and occupational health to anesthesia, cardiology, and nephrology. The specialty with the highest number of male RNs tends to be acute care, which encompasses emergency/trauma and medical-surgical.
John Schmidt, DNP, MSN, BSN, faculty member and program lead for the acute care nurse practitioner program at Purdue Global School of Nursing, refers to these specialties as having a high excitement factor.
“Men gravitate to nursing to help people,” he said. “In critical care, there is instant gratification. You see patients get better. It’s the same in the [intensive care unit] and the emergency department. We take care of them and can see how we made a difference.”
When hospitals and health systems create environments that support men in nursing, patients also benefit. Research shows that patients often prefer nurses of the same gender, and a more diverse healthcare workforce has been linked to improved patient outcomes. Reducing gender inequities among nursing staff could also improve job satisfaction and retention rates for men in nursing.
“When you’re in a vulnerable space as a patient ... it’s important to know that your care provider understands you [and] having men as nurses is a part of that,” said Gomes. “Even though patients might not be used to having a male nurse at the bedside, once they have the experience, it challenges preconceived notions [and] that connection is important.”
Hospitals must proactively support men in nursing to achieve the benefits of greater gender diversity in the nursing workforce. Male nurses have fewer role models and report higher levels of loneliness, isolation, and role strain.
Groups such as NYC Men in Nursing and mentorship programs such as Men in Nursing at RUSH University College of Nursing and RUSH University Medical Center, and the North Carolina Healthcare Association Diverse Healthcare Leaders Mentorship Program were designed to provide coaching, education, and networking opportunities and connect men in nursing.
Male nurses, Dunne added, must be role models and must take the lead in changing the conversations about gender roles in nursing. Establishing support systems and mentorship opportunities is instrumental in inspiring men to pursue nursing careers and creating visibility into the profession and “would create a level of parity for men in the profession and encourage them to want to stay in nursing as a long-term career.”
He told this news organization that creating scholarships for men enrolled in nursing school, increasing the involvement of male nurse leaders in recruitment efforts, and updating curriculum to ensure men are reflected in the materials is also essential.
“We’ve got to be willing and open to having the conversations to end the stereotypes that have plagued the profession,” said Dunne. “And we’ve got to push men in nursing to be front and center so folks see that there are opportunities for men in nursing.”
A version of this article appeared on Medscape.com.
Only 12% of the nurses providing patient care at hospitals and health clinics today are men. Although the percentage of nurses has increased — men made up just 2.7% of nurses in 1970 — nursing is still considered a “pink collar” profession, a female-dominated field.
“We’ve made strides over the last couple of decades, but [the number of men pursuing nursing careers] is leveling out,” said Jason Dunne, DNP, MSN, RN, chief academic officer at the Arizona College of Nursing, Phoenix. “There continues to be persistent gender stereotypes that [have] discouraged men from entering the profession.”
“The nursing shortage is very real,” Dunne said. “We need to be highly focused on the shortage and look at opportunities to bring diversity into the profession, and one big way to solve it is bringing more men into nursing.”
Representation Matters
Colleges recognize the need to diversify their nursing student population and have turned their attention to increasing the number of men attending informational sessions and career days. Dunne believes, “There is a general lack of awareness of nursing as a career choice [for men].”
The Nursing Consortium of Florida hosts a “Day in the Life of a Nurse” program to introduce high school students to nursing careers, and the University of Virginia School of Nursing invites male nursing students to speak at educational events to promote workforce diversity.
“When I was growing up, the males wouldn’t have been included in those sessions,” said Melissa Gilbert Gomes, PhD, APRN, PMHNP-BC, FNAP, FAAN, associate dean for diversity, equity, and inclusion at the University of Virginia School of Nursing, Charlottesville, Virginia. “It was nice to see their interest and to have a male student there for them to ask questions and to help them see that this could be a place for them.”
Nursing schools have also engaged in other efforts to encourage more men to consider nursing careers, from highlighting male nurses in marketing materials and engaging with men at career fairs to updating course curriculum to include content on men’s health and connecting male nursing students with men in nursing faculty or clinical settings.
Focusing on nursing as a lucrative career choice could also attract more men to the profession. On average, male registered nurses (RNs) make $7300 per year more than their female counterparts due to the gender pay gap. The median wage for male RNs in acute care, cardiology, and perioperative specialties is $90,000 annually.
At the University of Virginia School of Nursing, which the American Association for Men in Nursing (AAMN) named “Best School for Men in Nursing” in 2023, 20% of nursing students are men.
The school has a Men Advancing Nursing club and is in the process of chartering a new AAMN chapter. The goal, according to Gomes, is to create an environment where male nursing students feel represented and supported.
“Valuing the perspective that men bring [to nursing] is important,” she said. “Coming together [and] having that camaraderie and intrinsic motivation to specifically speak to areas that impact men ... is important.”
Promoting Patient Care
Highlighting the diversity of career options within the nursing profession is also essential. RNs can pursue careers in specialties ranging from pediatrics, orthopedics, and occupational health to anesthesia, cardiology, and nephrology. The specialty with the highest number of male RNs tends to be acute care, which encompasses emergency/trauma and medical-surgical.
John Schmidt, DNP, MSN, BSN, faculty member and program lead for the acute care nurse practitioner program at Purdue Global School of Nursing, refers to these specialties as having a high excitement factor.
“Men gravitate to nursing to help people,” he said. “In critical care, there is instant gratification. You see patients get better. It’s the same in the [intensive care unit] and the emergency department. We take care of them and can see how we made a difference.”
When hospitals and health systems create environments that support men in nursing, patients also benefit. Research shows that patients often prefer nurses of the same gender, and a more diverse healthcare workforce has been linked to improved patient outcomes. Reducing gender inequities among nursing staff could also improve job satisfaction and retention rates for men in nursing.
“When you’re in a vulnerable space as a patient ... it’s important to know that your care provider understands you [and] having men as nurses is a part of that,” said Gomes. “Even though patients might not be used to having a male nurse at the bedside, once they have the experience, it challenges preconceived notions [and] that connection is important.”
Hospitals must proactively support men in nursing to achieve the benefits of greater gender diversity in the nursing workforce. Male nurses have fewer role models and report higher levels of loneliness, isolation, and role strain.
Groups such as NYC Men in Nursing and mentorship programs such as Men in Nursing at RUSH University College of Nursing and RUSH University Medical Center, and the North Carolina Healthcare Association Diverse Healthcare Leaders Mentorship Program were designed to provide coaching, education, and networking opportunities and connect men in nursing.
Male nurses, Dunne added, must be role models and must take the lead in changing the conversations about gender roles in nursing. Establishing support systems and mentorship opportunities is instrumental in inspiring men to pursue nursing careers and creating visibility into the profession and “would create a level of parity for men in the profession and encourage them to want to stay in nursing as a long-term career.”
He told this news organization that creating scholarships for men enrolled in nursing school, increasing the involvement of male nurse leaders in recruitment efforts, and updating curriculum to ensure men are reflected in the materials is also essential.
“We’ve got to be willing and open to having the conversations to end the stereotypes that have plagued the profession,” said Dunne. “And we’ve got to push men in nursing to be front and center so folks see that there are opportunities for men in nursing.”
A version of this article appeared on Medscape.com.
Only 12% of the nurses providing patient care at hospitals and health clinics today are men. Although the percentage of nurses has increased — men made up just 2.7% of nurses in 1970 — nursing is still considered a “pink collar” profession, a female-dominated field.
“We’ve made strides over the last couple of decades, but [the number of men pursuing nursing careers] is leveling out,” said Jason Dunne, DNP, MSN, RN, chief academic officer at the Arizona College of Nursing, Phoenix. “There continues to be persistent gender stereotypes that [have] discouraged men from entering the profession.”
“The nursing shortage is very real,” Dunne said. “We need to be highly focused on the shortage and look at opportunities to bring diversity into the profession, and one big way to solve it is bringing more men into nursing.”
Representation Matters
Colleges recognize the need to diversify their nursing student population and have turned their attention to increasing the number of men attending informational sessions and career days. Dunne believes, “There is a general lack of awareness of nursing as a career choice [for men].”
The Nursing Consortium of Florida hosts a “Day in the Life of a Nurse” program to introduce high school students to nursing careers, and the University of Virginia School of Nursing invites male nursing students to speak at educational events to promote workforce diversity.
“When I was growing up, the males wouldn’t have been included in those sessions,” said Melissa Gilbert Gomes, PhD, APRN, PMHNP-BC, FNAP, FAAN, associate dean for diversity, equity, and inclusion at the University of Virginia School of Nursing, Charlottesville, Virginia. “It was nice to see their interest and to have a male student there for them to ask questions and to help them see that this could be a place for them.”
Nursing schools have also engaged in other efforts to encourage more men to consider nursing careers, from highlighting male nurses in marketing materials and engaging with men at career fairs to updating course curriculum to include content on men’s health and connecting male nursing students with men in nursing faculty or clinical settings.
Focusing on nursing as a lucrative career choice could also attract more men to the profession. On average, male registered nurses (RNs) make $7300 per year more than their female counterparts due to the gender pay gap. The median wage for male RNs in acute care, cardiology, and perioperative specialties is $90,000 annually.
At the University of Virginia School of Nursing, which the American Association for Men in Nursing (AAMN) named “Best School for Men in Nursing” in 2023, 20% of nursing students are men.
The school has a Men Advancing Nursing club and is in the process of chartering a new AAMN chapter. The goal, according to Gomes, is to create an environment where male nursing students feel represented and supported.
“Valuing the perspective that men bring [to nursing] is important,” she said. “Coming together [and] having that camaraderie and intrinsic motivation to specifically speak to areas that impact men ... is important.”
Promoting Patient Care
Highlighting the diversity of career options within the nursing profession is also essential. RNs can pursue careers in specialties ranging from pediatrics, orthopedics, and occupational health to anesthesia, cardiology, and nephrology. The specialty with the highest number of male RNs tends to be acute care, which encompasses emergency/trauma and medical-surgical.
John Schmidt, DNP, MSN, BSN, faculty member and program lead for the acute care nurse practitioner program at Purdue Global School of Nursing, refers to these specialties as having a high excitement factor.
“Men gravitate to nursing to help people,” he said. “In critical care, there is instant gratification. You see patients get better. It’s the same in the [intensive care unit] and the emergency department. We take care of them and can see how we made a difference.”
When hospitals and health systems create environments that support men in nursing, patients also benefit. Research shows that patients often prefer nurses of the same gender, and a more diverse healthcare workforce has been linked to improved patient outcomes. Reducing gender inequities among nursing staff could also improve job satisfaction and retention rates for men in nursing.
“When you’re in a vulnerable space as a patient ... it’s important to know that your care provider understands you [and] having men as nurses is a part of that,” said Gomes. “Even though patients might not be used to having a male nurse at the bedside, once they have the experience, it challenges preconceived notions [and] that connection is important.”
Hospitals must proactively support men in nursing to achieve the benefits of greater gender diversity in the nursing workforce. Male nurses have fewer role models and report higher levels of loneliness, isolation, and role strain.
Groups such as NYC Men in Nursing and mentorship programs such as Men in Nursing at RUSH University College of Nursing and RUSH University Medical Center, and the North Carolina Healthcare Association Diverse Healthcare Leaders Mentorship Program were designed to provide coaching, education, and networking opportunities and connect men in nursing.
Male nurses, Dunne added, must be role models and must take the lead in changing the conversations about gender roles in nursing. Establishing support systems and mentorship opportunities is instrumental in inspiring men to pursue nursing careers and creating visibility into the profession and “would create a level of parity for men in the profession and encourage them to want to stay in nursing as a long-term career.”
He told this news organization that creating scholarships for men enrolled in nursing school, increasing the involvement of male nurse leaders in recruitment efforts, and updating curriculum to ensure men are reflected in the materials is also essential.
“We’ve got to be willing and open to having the conversations to end the stereotypes that have plagued the profession,” said Dunne. “And we’ve got to push men in nursing to be front and center so folks see that there are opportunities for men in nursing.”
A version of this article appeared on Medscape.com.
Lawmakers Rush to Stave Off Doctor Pay Cuts as Medicare Finalizes 2025 Rates
Federal lawmakers are rushing to soften the blow of Medicare’s 2025 effective pay cut for doctors in 2025, introducing a bill that could limit the cut. But they have little time to act.
In 2025, the conversion factor used to calculate payment to doctors and hospitals caring for Medicare patients will drop to $32.35, a nearly 3% decrease from the current level.
Congress likely will act before the cuts take effect, said Rep. Larry Bucshon, MD (R-IN), who specialized in cardiothoracic surgery before joining Congress. Lawmakers in past years have typically tinkered with the Medicare physician fee schedule at the last minute, tucking in fixes to December legislative packages and spending bills.
“I’m pretty optimistic that a good portion of the fee cuts will be mitigated and they won’t go through,” Bucshon told this news organization in an interview.
Bruce A. Scott, MD, president of the American Medical Association (AMA) said in a statement that CMS’ release of the final fee schedule on November 1 should trigger serious work on a change to the 2025 Medicare physician fee schedule.
“The fee schedule rule released [on November 1] starts the clock — with January 1 looming,” Scott said. “A legislative remedy will require hard work and compromise. The 66 million patients who rely on Medicare are counting on that.”
Both Bucshon and Scott also joined many lawmakers and medical associations in calling on Congress for a larger overhaul of the Medicare physician fee schedule, well beyond whatever temporary adjustment may be made in the months ahead to avoid or soften the 2025 cuts.
The physician fee schedule sets formulas and rules regarding how the largest US buyer of health services pays the almost 1.3 million clinicians who bill Medicare. Of these, 51% are physicians. The physician fee schedule also covers payments for nurse practitioners, physician assistants, physical therapists, and other health professionals.
Last Major Overhaul Unpopular
There’s broad dissatisfaction with Congress’ last major overhaul of the Medicare physician fee schedule. The 2015 Medicare Access and CHIP Reauthorization Act (MACRA) aimed to shift clinicians toward programs tying pay increases to quality measures. But the implementation of that aim through the Merit-based Incentive Payment System is widely considered a disappointment.
MACRA was intended to end the need for annual “doc fixes,” as Congress’ last-minute Medicare adjustments are known. Seventeen such tweaks passed before MACRA took effect.
But MACRA did not include a broad-based inflation adjuster, and some clinicians’ incomes are lagging as inflation rates — and practice costs — have risen. Scott said the Medicare Economic Index, which is a measure used to gauge increases in practice costs for clinicians, is expected to rise by 3.5%.
“To put it bluntly, Medicare plans to pay us less while costs go up. You don’t have to be an economist to know that is an unsustainable trend, though one that has been going on for decades,” Scott said. “For physician practices operating on small margins already, this means it is harder to acquire new equipment, harder to retain staff, harder to take on new Medicare patients, and harder to keep the doors open, particularly in rural and underserved areas.”
In a statement, Jen Brull, MD, president of the American Academy of Family Physicians, noted that this likely will be the fifth year in a row that Congress will need to do a patch to prevent cuts in pay to clinicians.
Bucshon, who will retire from the House in January, said he expects Congress to pass legislation tying Medicare payment rates to inflation — eventually.
“People want to find a way to fix this problem, but also do it in a way that does not cut benefits to anyone, and that’s the key,” Bucshon said. “We’re going to have to find a way to make sure that providers are properly reimbursed.”
A version of this article first appeared on Medscape.com.
Federal lawmakers are rushing to soften the blow of Medicare’s 2025 effective pay cut for doctors in 2025, introducing a bill that could limit the cut. But they have little time to act.
In 2025, the conversion factor used to calculate payment to doctors and hospitals caring for Medicare patients will drop to $32.35, a nearly 3% decrease from the current level.
Congress likely will act before the cuts take effect, said Rep. Larry Bucshon, MD (R-IN), who specialized in cardiothoracic surgery before joining Congress. Lawmakers in past years have typically tinkered with the Medicare physician fee schedule at the last minute, tucking in fixes to December legislative packages and spending bills.
“I’m pretty optimistic that a good portion of the fee cuts will be mitigated and they won’t go through,” Bucshon told this news organization in an interview.
Bruce A. Scott, MD, president of the American Medical Association (AMA) said in a statement that CMS’ release of the final fee schedule on November 1 should trigger serious work on a change to the 2025 Medicare physician fee schedule.
“The fee schedule rule released [on November 1] starts the clock — with January 1 looming,” Scott said. “A legislative remedy will require hard work and compromise. The 66 million patients who rely on Medicare are counting on that.”
Both Bucshon and Scott also joined many lawmakers and medical associations in calling on Congress for a larger overhaul of the Medicare physician fee schedule, well beyond whatever temporary adjustment may be made in the months ahead to avoid or soften the 2025 cuts.
The physician fee schedule sets formulas and rules regarding how the largest US buyer of health services pays the almost 1.3 million clinicians who bill Medicare. Of these, 51% are physicians. The physician fee schedule also covers payments for nurse practitioners, physician assistants, physical therapists, and other health professionals.
Last Major Overhaul Unpopular
There’s broad dissatisfaction with Congress’ last major overhaul of the Medicare physician fee schedule. The 2015 Medicare Access and CHIP Reauthorization Act (MACRA) aimed to shift clinicians toward programs tying pay increases to quality measures. But the implementation of that aim through the Merit-based Incentive Payment System is widely considered a disappointment.
MACRA was intended to end the need for annual “doc fixes,” as Congress’ last-minute Medicare adjustments are known. Seventeen such tweaks passed before MACRA took effect.
But MACRA did not include a broad-based inflation adjuster, and some clinicians’ incomes are lagging as inflation rates — and practice costs — have risen. Scott said the Medicare Economic Index, which is a measure used to gauge increases in practice costs for clinicians, is expected to rise by 3.5%.
“To put it bluntly, Medicare plans to pay us less while costs go up. You don’t have to be an economist to know that is an unsustainable trend, though one that has been going on for decades,” Scott said. “For physician practices operating on small margins already, this means it is harder to acquire new equipment, harder to retain staff, harder to take on new Medicare patients, and harder to keep the doors open, particularly in rural and underserved areas.”
In a statement, Jen Brull, MD, president of the American Academy of Family Physicians, noted that this likely will be the fifth year in a row that Congress will need to do a patch to prevent cuts in pay to clinicians.
Bucshon, who will retire from the House in January, said he expects Congress to pass legislation tying Medicare payment rates to inflation — eventually.
“People want to find a way to fix this problem, but also do it in a way that does not cut benefits to anyone, and that’s the key,” Bucshon said. “We’re going to have to find a way to make sure that providers are properly reimbursed.”
A version of this article first appeared on Medscape.com.
Federal lawmakers are rushing to soften the blow of Medicare’s 2025 effective pay cut for doctors in 2025, introducing a bill that could limit the cut. But they have little time to act.
In 2025, the conversion factor used to calculate payment to doctors and hospitals caring for Medicare patients will drop to $32.35, a nearly 3% decrease from the current level.
Congress likely will act before the cuts take effect, said Rep. Larry Bucshon, MD (R-IN), who specialized in cardiothoracic surgery before joining Congress. Lawmakers in past years have typically tinkered with the Medicare physician fee schedule at the last minute, tucking in fixes to December legislative packages and spending bills.
“I’m pretty optimistic that a good portion of the fee cuts will be mitigated and they won’t go through,” Bucshon told this news organization in an interview.
Bruce A. Scott, MD, president of the American Medical Association (AMA) said in a statement that CMS’ release of the final fee schedule on November 1 should trigger serious work on a change to the 2025 Medicare physician fee schedule.
“The fee schedule rule released [on November 1] starts the clock — with January 1 looming,” Scott said. “A legislative remedy will require hard work and compromise. The 66 million patients who rely on Medicare are counting on that.”
Both Bucshon and Scott also joined many lawmakers and medical associations in calling on Congress for a larger overhaul of the Medicare physician fee schedule, well beyond whatever temporary adjustment may be made in the months ahead to avoid or soften the 2025 cuts.
The physician fee schedule sets formulas and rules regarding how the largest US buyer of health services pays the almost 1.3 million clinicians who bill Medicare. Of these, 51% are physicians. The physician fee schedule also covers payments for nurse practitioners, physician assistants, physical therapists, and other health professionals.
Last Major Overhaul Unpopular
There’s broad dissatisfaction with Congress’ last major overhaul of the Medicare physician fee schedule. The 2015 Medicare Access and CHIP Reauthorization Act (MACRA) aimed to shift clinicians toward programs tying pay increases to quality measures. But the implementation of that aim through the Merit-based Incentive Payment System is widely considered a disappointment.
MACRA was intended to end the need for annual “doc fixes,” as Congress’ last-minute Medicare adjustments are known. Seventeen such tweaks passed before MACRA took effect.
But MACRA did not include a broad-based inflation adjuster, and some clinicians’ incomes are lagging as inflation rates — and practice costs — have risen. Scott said the Medicare Economic Index, which is a measure used to gauge increases in practice costs for clinicians, is expected to rise by 3.5%.
“To put it bluntly, Medicare plans to pay us less while costs go up. You don’t have to be an economist to know that is an unsustainable trend, though one that has been going on for decades,” Scott said. “For physician practices operating on small margins already, this means it is harder to acquire new equipment, harder to retain staff, harder to take on new Medicare patients, and harder to keep the doors open, particularly in rural and underserved areas.”
In a statement, Jen Brull, MD, president of the American Academy of Family Physicians, noted that this likely will be the fifth year in a row that Congress will need to do a patch to prevent cuts in pay to clinicians.
Bucshon, who will retire from the House in January, said he expects Congress to pass legislation tying Medicare payment rates to inflation — eventually.
“People want to find a way to fix this problem, but also do it in a way that does not cut benefits to anyone, and that’s the key,” Bucshon said. “We’re going to have to find a way to make sure that providers are properly reimbursed.”
A version of this article first appeared on Medscape.com.
Lichenoid Drug Eruption Secondary to Apalutamide Treatment
To the Editor:
Lichenoid drug eruptions are lichen planus–like hypersensitivity reactions induced by medications. These reactions are rare but cause irritation to the skin, as extreme pruritus is common. One review of 300 consecutive cases of drug eruptions submitted to dermatopathology revealed that 12% of cases were classified as lichenoid drug reactions.1 Lichenoid dermatitis is characterized by extremely pruritic, scaly, eczematous or psoriasiform papules, often along the extensor surfaces and trunk.2 The pruritic nature of the rash can negatively impact quality of life. Treatment typically involves discontinuation of the offending medication, although complete resolution can take months, even after the drug is stopped. Although there have been some data suggesting that topical and/or oral corticosteroids can help with resolution, the rash can persist even with steroid treatment.2
The histopathologic findings of lichenoid drug eruptions show lichen planus–like changes such as hyperkeratosis, irregular acanthosis, and lichenoid interface dermatitis. Accordingly, idiopathic lichen planus is an important differential diagnosis for lichenoid drug eruptions; however, compared to idiopathic lichen planus, lichenoid drug eruptions are more likely to be associated with eosinophils and parakeratosis.1,3 In some cases, the histopathologic distinction between the 2 conditions is impossible, and clinical history needs to be considered to make a diagnosis.1 Drugs known to cause lichenoid drug reactions more commonly include angiotensin-converting enzyme inhibitors, beta blockers, thiazides, gold, penicillamine, and antimalarials.2 Lichenoid drug eruptions also have been documented in patients taking the second-generation nonsteroidal androgen receptor antagonist enzalutamide, which is used for the treatment of prostate cancer.4 More recently, the newer second-generation nonsteroidal androgen receptor antagonist apalutamide has been implicated in several cases of lichenoid drug eruptions.5,6
We present a case of an apalutamide-induced lichenoid drug eruption that was resistant to dose reduction and required discontinuation of treatment due to the negative impact on the patient’s quality of life. Once the rash resolved, the patient transitioned to enzalutamide without any adverse events (AEs).
A 72-year-old man with a history of metastatic prostate cancer (stage IVB) presented to the dermatology clinic with a 4-month history of a dry itchy rash on the face, chest, back, and legs that had developed 2 to 3 months after oncology started him on apalutamide. The patient initially received apalutamide 240 mg/d, which was reduced by his oncologist 3 months later to 180 mg/d following the appearance of the rash. Then apalutamide was held as he awaited improvement of the rash.
One week after the apalutamide was held, the patient presented to dermatology. He reported that he had tried over-the-counter ammonium lactate 12% lotion twice daily when the rash first developed without improvement. When the apalutamide was held, oncology prescribed mupirocin ointment 2% 3 times daily which yielded minimal relief. On physical examination, widespread lichenified papules and plaques were noted on the face, chest, back, and legs (Figure 1). Dermatology initially prescribed triamcinolone ointment 0.1% twice daily. A 4-mm punch biopsy specimen of the upper back revealed a lichenoid interface dermatitis with numerous eosinophils compatible with a lichenoid hypersensitivity reaction (Figure 2). Considering the clinical and histologic findings, a diagnosis of lichenoid drug eruption secondary to apalutamide treatment was made.
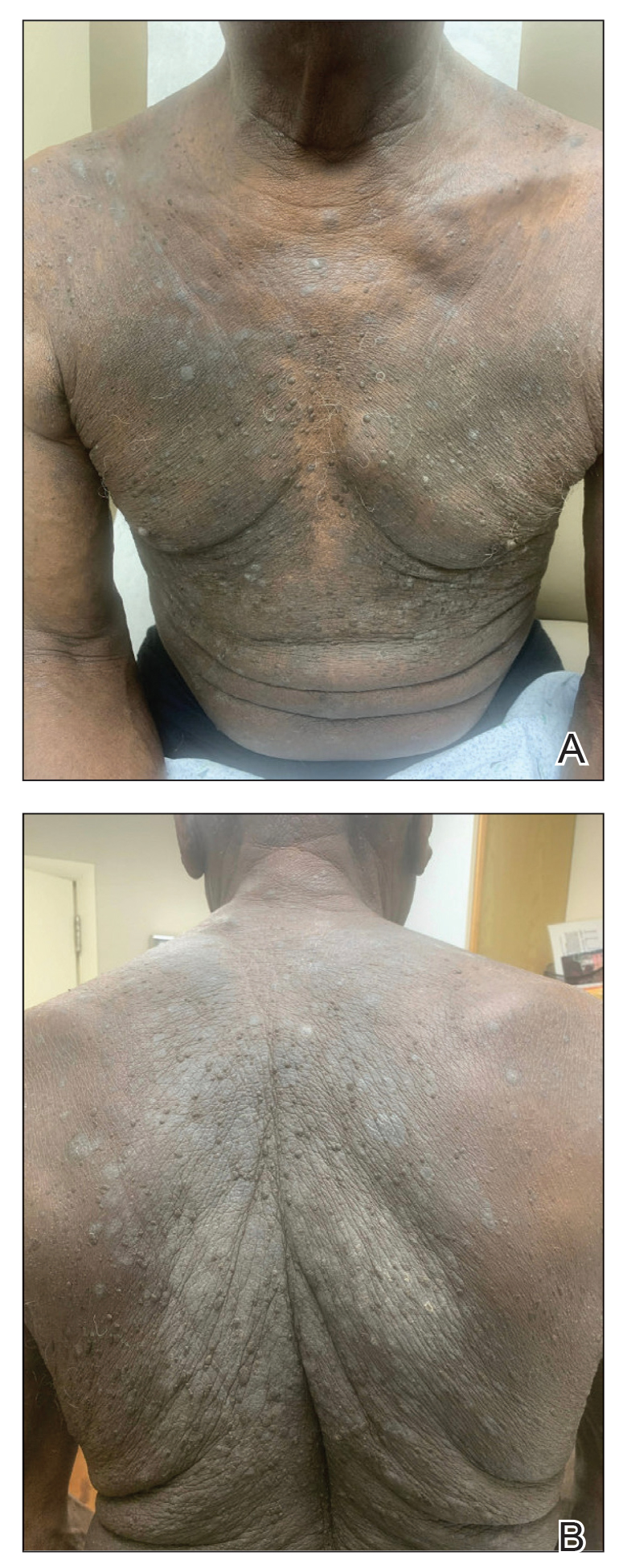
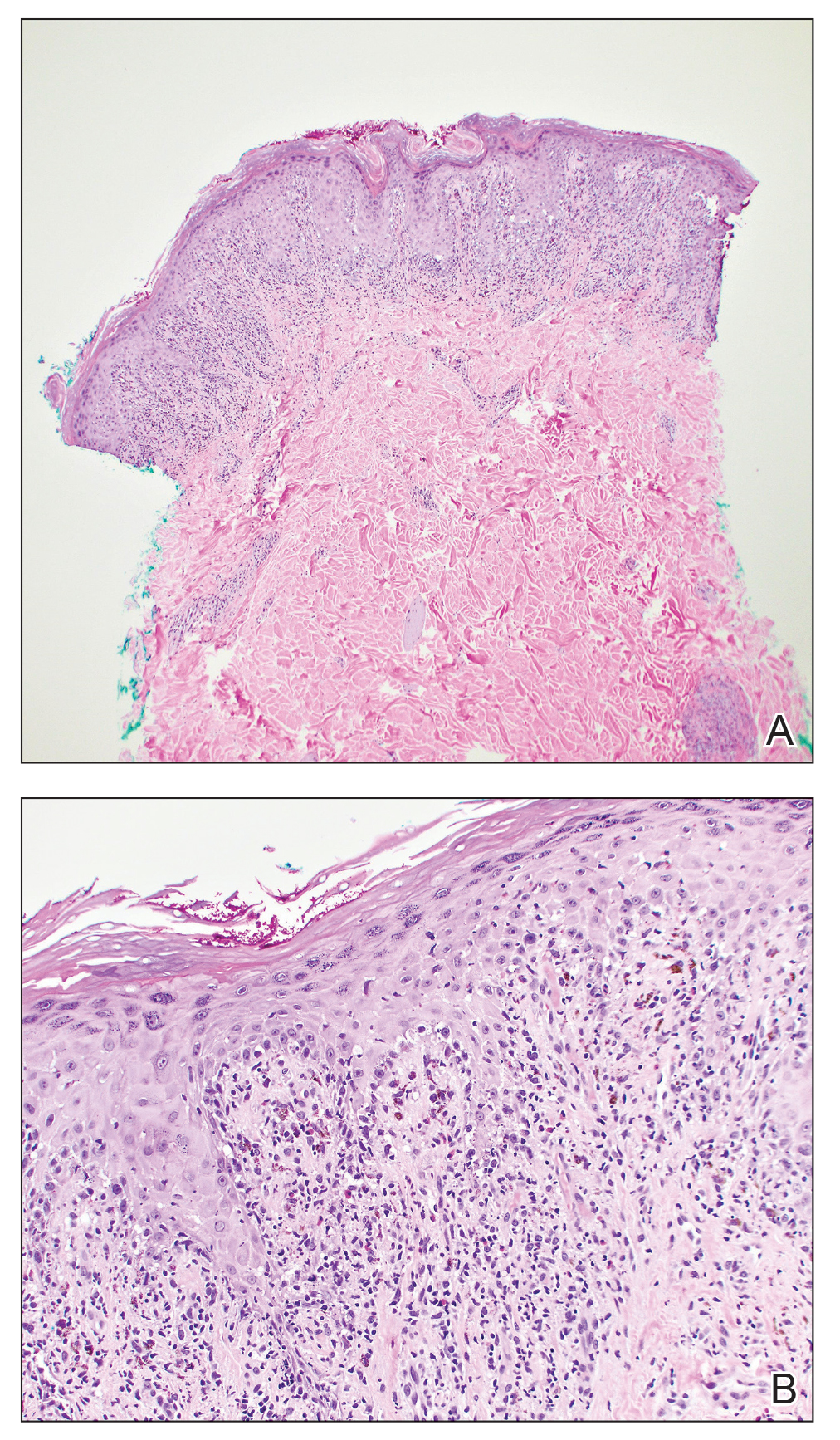
Two weeks after discontinuation of the medication, the rash improved, and the patient restarted apalutamide at a dosage of 120 mg/d; however, the rash re-emerged within 1 month and was resistant to the triamcinolone ointment 0.1%. Apalutamide was again discontinued, and oncology switched the patient to enzalutamide 160 mg/d in an effort to find a medication the patient could better tolerate. Two months after starting enzalutamide, the patient had resolution of the rash and no further dermatologic complications.
Apalutamide is a second-generation nonsteroidal androgen receptor antagonist used in the treatment of nonmetastatic castration-resistant prostate cancer (CRPC) and metastatic castration-sensitive prostate cancer (CSPC).7 It stops the spread and growth of prostate cancer cells by several different mechanisms, including competitively binding androgen receptors, preventing 5α-dihydrotestosterone from binding to androgen receptors, blocking androgen receptor nuclear translocation, impairing co-activator recruitment, and restraining androgen receptor DNA binding.7 The SPARTAN and TITAN phase 3 clinical trials demonstrated increased overall survival and time to progression with apalutamide in both nonmetastatic CRPC and metastatic CSPC. In both trials, the rash was shown to be an AE more commonly associated with apalutamide than placebo.8,9
Until recently, the characteristics of apalutamide-induced drug rashes have not been well described. One literature review reported 6 cases of cutaneous apalutamide-induced drug eruptions.5 Four (66.7%) of these eruptions were maculopapular rashes, only 2 of which were histologically classified as lichenoid in nature. The other 2 eruptions were classified as toxic epidermal necrosis.5 Another study of 303 patients with prostate cancer who were treated with apalutamide recorded the frequency and time to onset of dermatologic AEs.6 Seventy-one (23.4%) of the patients had dermatologic AEs, and of those, only 20 (28.2%) had AEs that resulted in interruptions in apalutamide therapy (with only 5 [25.0%] requiring medication discontinuation). Thirty-two (45.1%) patients were managed with topical or oral corticosteroids or dose modification. In this study, histopathology was examined in 8 cases (one of which had 2 biopsies for a total of 9 biopsies), 7 of which were consistent with lichenoid interface dermatitis.6
Lichenoid interface dermatitis is a rare manifestation of an apalutamide-induced drug eruption and also has been reported secondary to treatment with enzalutamide, another second-generation nonsteroidal androgen receptor antagonist.4 Enzalutamide was the first second-generation nonsteroidal androgen receptor antagonist approved for the treatment of prostate cancer. It originally was approved only for metastatic CRPC after docetaxel therapy in 2012, then later was expanded to metastatic and nonmetastatic CRPC in 2012 and 2018, respectively, as well as metastatic CSPC in 2019.7 Because enzalutamide is from the same medication class as apalutamide and has been on the market longer for the treatment of nonmetastatic CRPC and metastatic CSPC, it is not surprising that similar drug eruptions now are being reported secondary to apalutamide use as well.
It is important for providers to consider lichenoid drug eruptions in the differential diagnosis of pruritic rashes in patients taking second-generation nonsteroidal androgen receptor antagonists such as apalutamide or enzalutamide. Although dose reduction or treatment discontinuation have been the standard of care for patients with extremely pruritic lichenoid drug eruptions secondary to these medications, these are not ideal because they are important for cancer treatment. Interestingly, after our patient’s apalutamide-induced rash resolved and he was switched to enzalutamide, he did not develop any AEs. Based on our patient’s experience, physicians could consider switching their patients to another drug of the same class, as they may be able tolerate that medication. More research is needed to determine how commonly patients tolerate a different second-generation nonsteroidal androgen receptor antagonist after not tolerating another medication from the same class.
- Weyers W, Metze D. Histopathology of drug eruptions—general criteria, common patterns, and differential diagnosis. Dermatol Pract Concept. 2011;1:33-47. doi:10.5826/dpc.0101a09
- Cheraghlou S, Levy LL. Fixed drug eruption, bullous drug eruptions, and lichenoid drug eruptions. Clin Dermatol. 2020;38:679-692. doi:10.1016/j.clindermatol.2020.06.010
- Thompson DF, Skaehill PA. Drug-induced lichen planus. Pharmacotherapy. 1994;14:561-571.
- Khan S, Saizan AL, O’Brien K, et al. Diffuse hyperpigmented lichenoid drug eruption secondary to enzalutamide. Curr Probl Cancer Case Rep. 2022;5:100135. doi:10.1016/j.cpccr.2021.100135
- Katayama H, Saeki H, Osada S-I. Maculopapular drug eruption caused by apalutamide: case report and review of the literature. J Nippon Med Sch. 2022;89:550-554. doi:10.1272/jnms.JNMS.2022_89-503
- Pan A, Reingold RE, Zhao JL, et al. Dermatologic adverse events in prostate cancer patients treated with the androgen receptor inhibitor apalutamide. J Urol. 2022;207:1010-1019. doi:10.1097/JU.0000000000002425
- Rajaram P, Rivera A, Muthima K, et al. Second-generation androgen receptor antagonists as hormonal therapeutics for three forms of prostate cancer. Molecules. 2020;25:2448. doi:10.3390/molecules25102448
- Smith MR, Saad F, Chowdhury S, et al. Apalutamide treatment and metastasis-free survival in prostate cancer. N Engl J Med. 2018;378:1408-1418. doi:10.1056/NEJMoa1715546
- Chi KN, Agarwal N, Bjartell A, et al. Apalutamide for metastatic, castration-sensative prostate cancer. N Engl J Med. 2019;381:13-24. doi:10.1056/NEJMoa1903307
To the Editor:
Lichenoid drug eruptions are lichen planus–like hypersensitivity reactions induced by medications. These reactions are rare but cause irritation to the skin, as extreme pruritus is common. One review of 300 consecutive cases of drug eruptions submitted to dermatopathology revealed that 12% of cases were classified as lichenoid drug reactions.1 Lichenoid dermatitis is characterized by extremely pruritic, scaly, eczematous or psoriasiform papules, often along the extensor surfaces and trunk.2 The pruritic nature of the rash can negatively impact quality of life. Treatment typically involves discontinuation of the offending medication, although complete resolution can take months, even after the drug is stopped. Although there have been some data suggesting that topical and/or oral corticosteroids can help with resolution, the rash can persist even with steroid treatment.2
The histopathologic findings of lichenoid drug eruptions show lichen planus–like changes such as hyperkeratosis, irregular acanthosis, and lichenoid interface dermatitis. Accordingly, idiopathic lichen planus is an important differential diagnosis for lichenoid drug eruptions; however, compared to idiopathic lichen planus, lichenoid drug eruptions are more likely to be associated with eosinophils and parakeratosis.1,3 In some cases, the histopathologic distinction between the 2 conditions is impossible, and clinical history needs to be considered to make a diagnosis.1 Drugs known to cause lichenoid drug reactions more commonly include angiotensin-converting enzyme inhibitors, beta blockers, thiazides, gold, penicillamine, and antimalarials.2 Lichenoid drug eruptions also have been documented in patients taking the second-generation nonsteroidal androgen receptor antagonist enzalutamide, which is used for the treatment of prostate cancer.4 More recently, the newer second-generation nonsteroidal androgen receptor antagonist apalutamide has been implicated in several cases of lichenoid drug eruptions.5,6
We present a case of an apalutamide-induced lichenoid drug eruption that was resistant to dose reduction and required discontinuation of treatment due to the negative impact on the patient’s quality of life. Once the rash resolved, the patient transitioned to enzalutamide without any adverse events (AEs).
A 72-year-old man with a history of metastatic prostate cancer (stage IVB) presented to the dermatology clinic with a 4-month history of a dry itchy rash on the face, chest, back, and legs that had developed 2 to 3 months after oncology started him on apalutamide. The patient initially received apalutamide 240 mg/d, which was reduced by his oncologist 3 months later to 180 mg/d following the appearance of the rash. Then apalutamide was held as he awaited improvement of the rash.
One week after the apalutamide was held, the patient presented to dermatology. He reported that he had tried over-the-counter ammonium lactate 12% lotion twice daily when the rash first developed without improvement. When the apalutamide was held, oncology prescribed mupirocin ointment 2% 3 times daily which yielded minimal relief. On physical examination, widespread lichenified papules and plaques were noted on the face, chest, back, and legs (Figure 1). Dermatology initially prescribed triamcinolone ointment 0.1% twice daily. A 4-mm punch biopsy specimen of the upper back revealed a lichenoid interface dermatitis with numerous eosinophils compatible with a lichenoid hypersensitivity reaction (Figure 2). Considering the clinical and histologic findings, a diagnosis of lichenoid drug eruption secondary to apalutamide treatment was made.


Two weeks after discontinuation of the medication, the rash improved, and the patient restarted apalutamide at a dosage of 120 mg/d; however, the rash re-emerged within 1 month and was resistant to the triamcinolone ointment 0.1%. Apalutamide was again discontinued, and oncology switched the patient to enzalutamide 160 mg/d in an effort to find a medication the patient could better tolerate. Two months after starting enzalutamide, the patient had resolution of the rash and no further dermatologic complications.
Apalutamide is a second-generation nonsteroidal androgen receptor antagonist used in the treatment of nonmetastatic castration-resistant prostate cancer (CRPC) and metastatic castration-sensitive prostate cancer (CSPC).7 It stops the spread and growth of prostate cancer cells by several different mechanisms, including competitively binding androgen receptors, preventing 5α-dihydrotestosterone from binding to androgen receptors, blocking androgen receptor nuclear translocation, impairing co-activator recruitment, and restraining androgen receptor DNA binding.7 The SPARTAN and TITAN phase 3 clinical trials demonstrated increased overall survival and time to progression with apalutamide in both nonmetastatic CRPC and metastatic CSPC. In both trials, the rash was shown to be an AE more commonly associated with apalutamide than placebo.8,9
Until recently, the characteristics of apalutamide-induced drug rashes have not been well described. One literature review reported 6 cases of cutaneous apalutamide-induced drug eruptions.5 Four (66.7%) of these eruptions were maculopapular rashes, only 2 of which were histologically classified as lichenoid in nature. The other 2 eruptions were classified as toxic epidermal necrosis.5 Another study of 303 patients with prostate cancer who were treated with apalutamide recorded the frequency and time to onset of dermatologic AEs.6 Seventy-one (23.4%) of the patients had dermatologic AEs, and of those, only 20 (28.2%) had AEs that resulted in interruptions in apalutamide therapy (with only 5 [25.0%] requiring medication discontinuation). Thirty-two (45.1%) patients were managed with topical or oral corticosteroids or dose modification. In this study, histopathology was examined in 8 cases (one of which had 2 biopsies for a total of 9 biopsies), 7 of which were consistent with lichenoid interface dermatitis.6
Lichenoid interface dermatitis is a rare manifestation of an apalutamide-induced drug eruption and also has been reported secondary to treatment with enzalutamide, another second-generation nonsteroidal androgen receptor antagonist.4 Enzalutamide was the first second-generation nonsteroidal androgen receptor antagonist approved for the treatment of prostate cancer. It originally was approved only for metastatic CRPC after docetaxel therapy in 2012, then later was expanded to metastatic and nonmetastatic CRPC in 2012 and 2018, respectively, as well as metastatic CSPC in 2019.7 Because enzalutamide is from the same medication class as apalutamide and has been on the market longer for the treatment of nonmetastatic CRPC and metastatic CSPC, it is not surprising that similar drug eruptions now are being reported secondary to apalutamide use as well.
It is important for providers to consider lichenoid drug eruptions in the differential diagnosis of pruritic rashes in patients taking second-generation nonsteroidal androgen receptor antagonists such as apalutamide or enzalutamide. Although dose reduction or treatment discontinuation have been the standard of care for patients with extremely pruritic lichenoid drug eruptions secondary to these medications, these are not ideal because they are important for cancer treatment. Interestingly, after our patient’s apalutamide-induced rash resolved and he was switched to enzalutamide, he did not develop any AEs. Based on our patient’s experience, physicians could consider switching their patients to another drug of the same class, as they may be able tolerate that medication. More research is needed to determine how commonly patients tolerate a different second-generation nonsteroidal androgen receptor antagonist after not tolerating another medication from the same class.
To the Editor:
Lichenoid drug eruptions are lichen planus–like hypersensitivity reactions induced by medications. These reactions are rare but cause irritation to the skin, as extreme pruritus is common. One review of 300 consecutive cases of drug eruptions submitted to dermatopathology revealed that 12% of cases were classified as lichenoid drug reactions.1 Lichenoid dermatitis is characterized by extremely pruritic, scaly, eczematous or psoriasiform papules, often along the extensor surfaces and trunk.2 The pruritic nature of the rash can negatively impact quality of life. Treatment typically involves discontinuation of the offending medication, although complete resolution can take months, even after the drug is stopped. Although there have been some data suggesting that topical and/or oral corticosteroids can help with resolution, the rash can persist even with steroid treatment.2
The histopathologic findings of lichenoid drug eruptions show lichen planus–like changes such as hyperkeratosis, irregular acanthosis, and lichenoid interface dermatitis. Accordingly, idiopathic lichen planus is an important differential diagnosis for lichenoid drug eruptions; however, compared to idiopathic lichen planus, lichenoid drug eruptions are more likely to be associated with eosinophils and parakeratosis.1,3 In some cases, the histopathologic distinction between the 2 conditions is impossible, and clinical history needs to be considered to make a diagnosis.1 Drugs known to cause lichenoid drug reactions more commonly include angiotensin-converting enzyme inhibitors, beta blockers, thiazides, gold, penicillamine, and antimalarials.2 Lichenoid drug eruptions also have been documented in patients taking the second-generation nonsteroidal androgen receptor antagonist enzalutamide, which is used for the treatment of prostate cancer.4 More recently, the newer second-generation nonsteroidal androgen receptor antagonist apalutamide has been implicated in several cases of lichenoid drug eruptions.5,6
We present a case of an apalutamide-induced lichenoid drug eruption that was resistant to dose reduction and required discontinuation of treatment due to the negative impact on the patient’s quality of life. Once the rash resolved, the patient transitioned to enzalutamide without any adverse events (AEs).
A 72-year-old man with a history of metastatic prostate cancer (stage IVB) presented to the dermatology clinic with a 4-month history of a dry itchy rash on the face, chest, back, and legs that had developed 2 to 3 months after oncology started him on apalutamide. The patient initially received apalutamide 240 mg/d, which was reduced by his oncologist 3 months later to 180 mg/d following the appearance of the rash. Then apalutamide was held as he awaited improvement of the rash.
One week after the apalutamide was held, the patient presented to dermatology. He reported that he had tried over-the-counter ammonium lactate 12% lotion twice daily when the rash first developed without improvement. When the apalutamide was held, oncology prescribed mupirocin ointment 2% 3 times daily which yielded minimal relief. On physical examination, widespread lichenified papules and plaques were noted on the face, chest, back, and legs (Figure 1). Dermatology initially prescribed triamcinolone ointment 0.1% twice daily. A 4-mm punch biopsy specimen of the upper back revealed a lichenoid interface dermatitis with numerous eosinophils compatible with a lichenoid hypersensitivity reaction (Figure 2). Considering the clinical and histologic findings, a diagnosis of lichenoid drug eruption secondary to apalutamide treatment was made.


Two weeks after discontinuation of the medication, the rash improved, and the patient restarted apalutamide at a dosage of 120 mg/d; however, the rash re-emerged within 1 month and was resistant to the triamcinolone ointment 0.1%. Apalutamide was again discontinued, and oncology switched the patient to enzalutamide 160 mg/d in an effort to find a medication the patient could better tolerate. Two months after starting enzalutamide, the patient had resolution of the rash and no further dermatologic complications.
Apalutamide is a second-generation nonsteroidal androgen receptor antagonist used in the treatment of nonmetastatic castration-resistant prostate cancer (CRPC) and metastatic castration-sensitive prostate cancer (CSPC).7 It stops the spread and growth of prostate cancer cells by several different mechanisms, including competitively binding androgen receptors, preventing 5α-dihydrotestosterone from binding to androgen receptors, blocking androgen receptor nuclear translocation, impairing co-activator recruitment, and restraining androgen receptor DNA binding.7 The SPARTAN and TITAN phase 3 clinical trials demonstrated increased overall survival and time to progression with apalutamide in both nonmetastatic CRPC and metastatic CSPC. In both trials, the rash was shown to be an AE more commonly associated with apalutamide than placebo.8,9
Until recently, the characteristics of apalutamide-induced drug rashes have not been well described. One literature review reported 6 cases of cutaneous apalutamide-induced drug eruptions.5 Four (66.7%) of these eruptions were maculopapular rashes, only 2 of which were histologically classified as lichenoid in nature. The other 2 eruptions were classified as toxic epidermal necrosis.5 Another study of 303 patients with prostate cancer who were treated with apalutamide recorded the frequency and time to onset of dermatologic AEs.6 Seventy-one (23.4%) of the patients had dermatologic AEs, and of those, only 20 (28.2%) had AEs that resulted in interruptions in apalutamide therapy (with only 5 [25.0%] requiring medication discontinuation). Thirty-two (45.1%) patients were managed with topical or oral corticosteroids or dose modification. In this study, histopathology was examined in 8 cases (one of which had 2 biopsies for a total of 9 biopsies), 7 of which were consistent with lichenoid interface dermatitis.6
Lichenoid interface dermatitis is a rare manifestation of an apalutamide-induced drug eruption and also has been reported secondary to treatment with enzalutamide, another second-generation nonsteroidal androgen receptor antagonist.4 Enzalutamide was the first second-generation nonsteroidal androgen receptor antagonist approved for the treatment of prostate cancer. It originally was approved only for metastatic CRPC after docetaxel therapy in 2012, then later was expanded to metastatic and nonmetastatic CRPC in 2012 and 2018, respectively, as well as metastatic CSPC in 2019.7 Because enzalutamide is from the same medication class as apalutamide and has been on the market longer for the treatment of nonmetastatic CRPC and metastatic CSPC, it is not surprising that similar drug eruptions now are being reported secondary to apalutamide use as well.
It is important for providers to consider lichenoid drug eruptions in the differential diagnosis of pruritic rashes in patients taking second-generation nonsteroidal androgen receptor antagonists such as apalutamide or enzalutamide. Although dose reduction or treatment discontinuation have been the standard of care for patients with extremely pruritic lichenoid drug eruptions secondary to these medications, these are not ideal because they are important for cancer treatment. Interestingly, after our patient’s apalutamide-induced rash resolved and he was switched to enzalutamide, he did not develop any AEs. Based on our patient’s experience, physicians could consider switching their patients to another drug of the same class, as they may be able tolerate that medication. More research is needed to determine how commonly patients tolerate a different second-generation nonsteroidal androgen receptor antagonist after not tolerating another medication from the same class.
- Weyers W, Metze D. Histopathology of drug eruptions—general criteria, common patterns, and differential diagnosis. Dermatol Pract Concept. 2011;1:33-47. doi:10.5826/dpc.0101a09
- Cheraghlou S, Levy LL. Fixed drug eruption, bullous drug eruptions, and lichenoid drug eruptions. Clin Dermatol. 2020;38:679-692. doi:10.1016/j.clindermatol.2020.06.010
- Thompson DF, Skaehill PA. Drug-induced lichen planus. Pharmacotherapy. 1994;14:561-571.
- Khan S, Saizan AL, O’Brien K, et al. Diffuse hyperpigmented lichenoid drug eruption secondary to enzalutamide. Curr Probl Cancer Case Rep. 2022;5:100135. doi:10.1016/j.cpccr.2021.100135
- Katayama H, Saeki H, Osada S-I. Maculopapular drug eruption caused by apalutamide: case report and review of the literature. J Nippon Med Sch. 2022;89:550-554. doi:10.1272/jnms.JNMS.2022_89-503
- Pan A, Reingold RE, Zhao JL, et al. Dermatologic adverse events in prostate cancer patients treated with the androgen receptor inhibitor apalutamide. J Urol. 2022;207:1010-1019. doi:10.1097/JU.0000000000002425
- Rajaram P, Rivera A, Muthima K, et al. Second-generation androgen receptor antagonists as hormonal therapeutics for three forms of prostate cancer. Molecules. 2020;25:2448. doi:10.3390/molecules25102448
- Smith MR, Saad F, Chowdhury S, et al. Apalutamide treatment and metastasis-free survival in prostate cancer. N Engl J Med. 2018;378:1408-1418. doi:10.1056/NEJMoa1715546
- Chi KN, Agarwal N, Bjartell A, et al. Apalutamide for metastatic, castration-sensative prostate cancer. N Engl J Med. 2019;381:13-24. doi:10.1056/NEJMoa1903307
- Weyers W, Metze D. Histopathology of drug eruptions—general criteria, common patterns, and differential diagnosis. Dermatol Pract Concept. 2011;1:33-47. doi:10.5826/dpc.0101a09
- Cheraghlou S, Levy LL. Fixed drug eruption, bullous drug eruptions, and lichenoid drug eruptions. Clin Dermatol. 2020;38:679-692. doi:10.1016/j.clindermatol.2020.06.010
- Thompson DF, Skaehill PA. Drug-induced lichen planus. Pharmacotherapy. 1994;14:561-571.
- Khan S, Saizan AL, O’Brien K, et al. Diffuse hyperpigmented lichenoid drug eruption secondary to enzalutamide. Curr Probl Cancer Case Rep. 2022;5:100135. doi:10.1016/j.cpccr.2021.100135
- Katayama H, Saeki H, Osada S-I. Maculopapular drug eruption caused by apalutamide: case report and review of the literature. J Nippon Med Sch. 2022;89:550-554. doi:10.1272/jnms.JNMS.2022_89-503
- Pan A, Reingold RE, Zhao JL, et al. Dermatologic adverse events in prostate cancer patients treated with the androgen receptor inhibitor apalutamide. J Urol. 2022;207:1010-1019. doi:10.1097/JU.0000000000002425
- Rajaram P, Rivera A, Muthima K, et al. Second-generation androgen receptor antagonists as hormonal therapeutics for three forms of prostate cancer. Molecules. 2020;25:2448. doi:10.3390/molecules25102448
- Smith MR, Saad F, Chowdhury S, et al. Apalutamide treatment and metastasis-free survival in prostate cancer. N Engl J Med. 2018;378:1408-1418. doi:10.1056/NEJMoa1715546
- Chi KN, Agarwal N, Bjartell A, et al. Apalutamide for metastatic, castration-sensative prostate cancer. N Engl J Med. 2019;381:13-24. doi:10.1056/NEJMoa1903307
Practice Points
- Although it is rare, patients can develop lichenoid drug eruptions secondary to treatment with second-generation nonsteroidal androgen receptor antagonists such as apalutamide.
- If a patient develops a lichenoid drug eruption while taking a specific second-generation nonsteroidal androgen receptor antagonist, the entire class of medications should not be ruled out, as some patients can tolerate other drugs from that class.
Botulinum Toxin Injection for Treatment of Scleroderma-Related Anterior Neck Sclerosis
To the Editor:
Scleroderma is a chronic autoimmune connective tissue disease that results in excessive collagen deposition in the skin and other organs throughout the body. On its own or in the setting of mixed connective tissue disease, scleroderma can result in systemic or localized symptoms that can limit patients’ functional capabilities, cause pain and discomfort, and reduce self-esteem—all negatively impacting patients’ quality of life.1,2 Neck sclerosis is a common manifestation of scleroderma. There is no curative treatment for scleroderma; thus, therapy is focused on slowing disease progression and improving quality of life. We present a case of neck sclerosis in a 44-year-old woman with scleroderma that was successfully treated with botulinum toxin (BTX) type A injection, resulting in improved skin laxity and appearance with high patient satisfaction. Our case demonstrates the potential positive effects of BTX treatment in patients with features of sclerosis or fibrosis, particularly in the neck region.
A 44-year-old woman presented to the dermatology clinic for treatment of thickened neck skin with stiffness and tightness that had been present for months to years. She had a history of mixed connective tissue disease (MCTD)(positive anti-ribonucleoprotein, anti–Sjögren syndrome–related antigen, and anti-Smith antibodies) with features of scleroderma and polyarthritis. The patient currently was taking sulfasalazine for the polyarthritis; she previously had taken hydroxychloroquine but discontinued treatment due to ineffectiveness. She was not taking any topical or systemic medications for scleroderma. On physical examination, the skin on the anterior neck appeared thickened with shiny patches (Figure 1). Pinching the skin in the affected area demonstrated sclerosis with high tension.
The dermatologist (J.J.) discussed potential treatment options to help relax the tension in the skin of the anterior neck, including BTX injections. After receiving counsel on adverse effects, alternative treatments, and postprocedural care, the patient decided to proceed with the procedure. The anterior neck was cleansed with an alcohol swab and 37 units (range, 25–50 units) of incobotulinumtoxinA (reconstituted using 2.5-mL bacteriostatic normal saline per 100 units) was injected transdermally using a 9-point injection technique, with each injection placed approximately 1 cm apart. The approximate treatment area included the space between the sternocleidomastoid anterior edges and below the hyoid bone up to the cricothyroid membrane (anatomic zone II).
When the patient returned for follow-up 3 weeks later, she reported considerable improvement in the stiffness and appearance of the skin on the anterior neck. On physical examination, the skin of the neck appeared softened, and improved laxity was seen on pinching the skin compared to the initial presentation (Figure 2). The patient expressed satisfaction with the results and denied any adverse events following the procedure.
Mixed connective tissue disease manifests with a combination of features from various disorders—mainly lupus, scleroderma, polymyositis, and rheumatoid arthritis. It is most prevalent in females and often is diagnosed in the third decade of life.3 It is associated with positive antinuclear antibodies and human leukocyte antigen (HLA) II alleles (HLA-DR4, HLA-DR1, and HLA-DR2). Raynaud phenomenon (RP), one of the most common skin manifestations in both scleroderma and MCTD, is present in 75% to 90% of patients with MCTD.3
Scleroderma is a chronic connective tissue disorder that results in excessive collagen deposition in the skin and other organs throughout the body.4 Although the etiology is unknown, scleroderma develops when overactivation of the immune system leads to CD4+ T-lymphocyte infiltration in the skin, along with the release of profibrotic interleukins and growth factors, resulting in fibrosis.4 Subtypes include localized scleroderma (morphea), limited cutaneous systemic sclerosis (formerly known as CREST [calcinosis, RP, esophageal dysmotility, sclerodactyly, and telangiectasia] syndrome), diffuse cutaneous systemic sclerosis, and systemic sclerosis sine scleroderma.5 Scleroderma is associated with positive antinuclear antibodies and HLA II alleles (HLA-DR2 and HLA-DR5).
On its own or in the setting of MCTD, scleroderma can result in systemic or localized symptoms. Overall, the most common symptom is RP.5 Localized scleroderma and limited cutaneous systemic sclerosis manifest with symptoms of the skin and underlying tissues. Diffuse cutaneous systemic sclerosis involves cutaneous and visceral symptoms, including lung, esophageal, and vascular involvement.6 Similar to MCTD, scleroderma is most prevalent in middle-aged females,7 though it occurs at a higher rate and with a more severe disease course in Black patients.8
A highly sensitive and specific test for scleroderma that can aid in diagnosis is the neck sign—tightening of the skin of the neck when the head extends.9,10 In one study, the neck sign was positive in more than 90% of patients with scleroderma and negative for control patients and those with primary RP.9 Thus, neck sclerosis is a common manifestation of scleroderma for which patients may seek treatment.
While there is no curative treatment for scleroderma, skin manifestations can be treated with mycophenolate mofetil or methotrexate.5 Systemic treatments may be recommended if the patient has additional symptoms, such as azathioprine for myositis/arthritis and cyclophosphamide for interstitial lung disease.5 However, it is important to note that these medications are associated with risk for gastrointestinal upset, mouth sores, fatigue, or other complications.
Botulinum toxin is a bacterial protein toxin and neuromodulator that inhibits neurotransmitter release by cleaving SNARE proteins at peripheral nerve terminal junctions.11 It has been used in a variety of dermatologic and nondermatologic conditions, including migraines, hyperhidrosis, contractures, scars, and overactive bladder. It also has been used in aesthetics for facial rejuvenation and minimization of wrinkle appearance. Dermatologists and rheumatologists have successfully used BTX to treat primary and secondary RP—the most common symptom of scleroderma—due to its vasodilatation properties.12 Although our patient did not have RP, use of BTX to treat other features of scleroderma, including en coup de sabre, thoracic outlet syndrome, dyspareunia, gastroparesis, pterygium inversum unguis, and dysphagia has been documented.13-18 An in vivo mouse study that examined the possible mechanism for BTX as a treatment in scleroderma found that BTX injections significantly decreased dermal thickness and inflammation in fibrosis (P<.05). An analysis of oxidative stress and mRNA expression showed that BTX may treat fibrosis by suppressing oxidative stress and inflammatory cells, resulting in decreased apoptosis and oxidant-induced intracellular accumulation of reactive oxygen species.19 Another animal study demonstrated the positive effects of BTX treatment for fibrosis of the bladder in rats.20 In one case report, a female patient with scleroderma and facial fibrosis received perioral BTX injections for cosmetic purposes but also observed improvement in mouth constriction, demonstrating the potential efficacy of BTX for facial fibrosis.21
Our case demonstrates the potential positive effects of BTX treatment in patients with features of sclerosis or fibrosis, particularly in the neck region. We recommend assessing the efficacy of the initial BTX treatment after 2 to 3 weeks, with additional injections as needed to achieve the patient’s desired level of comfort and appearance at approximately 3-month intervals (aligning with the expected duration of efficacy of BTX).22 Our patient experienced considerable relief and high satisfaction with BTX treatment. Given the limitations of sclerosis treatments and the unwanted adverse-effect profile of systemic treatments, BTX injections may be a preferrable treatment option for cutaneous manifestations of scleroderma among patients. Future studies with larger patient populations and a control group are warranted to further explore the use of BTX for the dermatologic treatment of scleroderma.
- Lis-S´wie¸ty A, Skrzypek-Salamon A, Ranosz-Janicka I, et al. Health-related quality of life and its influencing factors in adult patients with localized scleroderma—a cross-sectional study. Health Qual Life Outcomes. 2020;18:133. doi:10.1186/s12955-020-01386-0
- Almeida C, Almeida I, Vasconcelos C. Quality of life in systemic sclerosis. Autoimmun Rev. 2015;14:1087-1096. doi:10.1016/j.autrev.2015.07.012
- Ortega-Hernandez OD, Shoenfeld Y. Mixed connective tissue disease: an overview of clinical manifestations, diagnosis and treatment. Best Pract Res Clin Rheumatol. 2012;26:61-72. doi:10.1016/j.berh.2012.01.009
- Rongioletti F, Ferreli C, Atzori L, et al. Scleroderma with an update about clinico-pathological correlation. G Ital Dermatol Venereol. 2018;153:208-215. doi:10.23736/S0392-0488.18.05922-9
- Fett N. Scleroderma: nomenclature, etiology, pathogenesis, prognosis, and treatments: facts and controversies. Clin Dermatol. 2013;31:432-437. doi:10.1016/j.clindermatol.2013.01.010
- Careta MF, Romiti R. Localized scleroderma: clinical spectrum and therapeutic update. An Bras Dermatol. 2015;90:62-73. doi:10.1590/abd1806-4841.20152890
- Calderon LM, Pope JE. Scleroderma epidemiology update. Curr Opin Rheumatol. 2021;33:122-127. doi:10.1097/BOR.0000000000000785
- Morgan ND, Gelber AC. African Americans and scleroderma: examining the root cause of the association. Arthritis Care Res (Hoboken). 2019;71:1151-1153. doi:10.1002/acr.23860
- Barnett AJ. The “neck sign” in scleroderma. Arthritis Rheum. 1989;32:209-211. doi:10.1002/anr.1780320215
- Barnett AJ, Miller M, Littlejohn GO. The diagnosis and classification of scleroderma (systemic sclerosis). Postgrad Med J. 1988;64:121-125. doi:10.1136/pgmj.64.748.121
- Rossetto O, Pirazzini M, Fabris F, et al. Botulinum neurotoxins: mechanism of action. Handb Exp Pharmacol. 2021;263:35-47.doi:10.1007/164_2020_355
- Ennis D, Ahmad Z, Anderson MA, et al. Botulinum toxin in the management of primary and secondary Raynaud’s phenomenon. Best Pract Res Clin Rheumatol. 2021;35:101684. doi:10.1016/j.berh.2021.101684
- Turkmani MG, Alnomair N. Enhancement of the aesthetic outcome of scleroderma en coup de sabre with botulinum toxin injection. JAAD Case Rep. 2018;4:579-581. doi:10.1016/j.jdcr.2018.03.023
- Le EN, Freischlag JA, Christo PJ, et al. Thoracic outlet syndrome secondary to localized scleroderma treated with botulinum toxin injection. Arthritis Care Res (Hoboken). 2010;62:430-433. doi:10.1002/acr.20099
- Mousty E, Rathat G, Rouleau C, et al. Botulinum toxin type A for treatment of dyspareunia caused by localized scleroderma. Acta Obstet Gynecol Scand. 2011;90:926-927. doi:10.1111/j.1600-0412.2011.01183.x
- Tang DM, Friedenberg FK. Gastroparesis: approach, diagnostic evaluation, and management. Dis Mon. 2011;57:74-101. doi:10.1016/j.disamonth.2010.12.007
- Katschinski M. [Diagnosis and treatment of esophageal motility disorders]. Ther Umsch. 2001;58:128-133. doi:10.1024/0040-5930.58.3.128
- Kim DJ, Odell ID. Improvement of pterygium inversum unguis and Raynaud phenomenon with interdigital botulinum toxin injections. JAAD Case Rep. 2022;26:79-81. doi:10.1016/j.jdcr.2022.06.009
- Baral H, Sekiguchi A, Uchiyama A, et al. Inhibition of skin fibrosis in systemic sclerosis by botulinum toxin B via the suppression of oxidative stress. J Dermatol. 2021;48:1052-1061. doi:10.1111/1346-8138.15888
- Jia C, Xing T, Shang Z, et al. Botulinum toxin A improves neurogenic bladder fibrosis by suppressing transforming growth factor β1 expression in rats. Transl Androl Urol. 2021;10:2000-2007. doi:10.21037/tau-21-62
- Hoverson K, Love T, Lam TK, et al. A novel treatment for limited mouth opening due to facial fibrosis: a case series. J Am Acad Dermatol. 2018;78:190-192. doi:10.1016/j.jaad.2017.07.006
- Kollewe K, Mohammadi B, Köhler S, et al. Blepharospasm: long-term treatment with either Botox®, Xeomin® or Dysport®. J Neural Transm (Vienna). 2015;122:427-431. doi:10.1007/s00702-014-1278-z
To the Editor:
Scleroderma is a chronic autoimmune connective tissue disease that results in excessive collagen deposition in the skin and other organs throughout the body. On its own or in the setting of mixed connective tissue disease, scleroderma can result in systemic or localized symptoms that can limit patients’ functional capabilities, cause pain and discomfort, and reduce self-esteem—all negatively impacting patients’ quality of life.1,2 Neck sclerosis is a common manifestation of scleroderma. There is no curative treatment for scleroderma; thus, therapy is focused on slowing disease progression and improving quality of life. We present a case of neck sclerosis in a 44-year-old woman with scleroderma that was successfully treated with botulinum toxin (BTX) type A injection, resulting in improved skin laxity and appearance with high patient satisfaction. Our case demonstrates the potential positive effects of BTX treatment in patients with features of sclerosis or fibrosis, particularly in the neck region.
A 44-year-old woman presented to the dermatology clinic for treatment of thickened neck skin with stiffness and tightness that had been present for months to years. She had a history of mixed connective tissue disease (MCTD)(positive anti-ribonucleoprotein, anti–Sjögren syndrome–related antigen, and anti-Smith antibodies) with features of scleroderma and polyarthritis. The patient currently was taking sulfasalazine for the polyarthritis; she previously had taken hydroxychloroquine but discontinued treatment due to ineffectiveness. She was not taking any topical or systemic medications for scleroderma. On physical examination, the skin on the anterior neck appeared thickened with shiny patches (Figure 1). Pinching the skin in the affected area demonstrated sclerosis with high tension.

The dermatologist (J.J.) discussed potential treatment options to help relax the tension in the skin of the anterior neck, including BTX injections. After receiving counsel on adverse effects, alternative treatments, and postprocedural care, the patient decided to proceed with the procedure. The anterior neck was cleansed with an alcohol swab and 37 units (range, 25–50 units) of incobotulinumtoxinA (reconstituted using 2.5-mL bacteriostatic normal saline per 100 units) was injected transdermally using a 9-point injection technique, with each injection placed approximately 1 cm apart. The approximate treatment area included the space between the sternocleidomastoid anterior edges and below the hyoid bone up to the cricothyroid membrane (anatomic zone II).
When the patient returned for follow-up 3 weeks later, she reported considerable improvement in the stiffness and appearance of the skin on the anterior neck. On physical examination, the skin of the neck appeared softened, and improved laxity was seen on pinching the skin compared to the initial presentation (Figure 2). The patient expressed satisfaction with the results and denied any adverse events following the procedure.

Mixed connective tissue disease manifests with a combination of features from various disorders—mainly lupus, scleroderma, polymyositis, and rheumatoid arthritis. It is most prevalent in females and often is diagnosed in the third decade of life.3 It is associated with positive antinuclear antibodies and human leukocyte antigen (HLA) II alleles (HLA-DR4, HLA-DR1, and HLA-DR2). Raynaud phenomenon (RP), one of the most common skin manifestations in both scleroderma and MCTD, is present in 75% to 90% of patients with MCTD.3
Scleroderma is a chronic connective tissue disorder that results in excessive collagen deposition in the skin and other organs throughout the body.4 Although the etiology is unknown, scleroderma develops when overactivation of the immune system leads to CD4+ T-lymphocyte infiltration in the skin, along with the release of profibrotic interleukins and growth factors, resulting in fibrosis.4 Subtypes include localized scleroderma (morphea), limited cutaneous systemic sclerosis (formerly known as CREST [calcinosis, RP, esophageal dysmotility, sclerodactyly, and telangiectasia] syndrome), diffuse cutaneous systemic sclerosis, and systemic sclerosis sine scleroderma.5 Scleroderma is associated with positive antinuclear antibodies and HLA II alleles (HLA-DR2 and HLA-DR5).
On its own or in the setting of MCTD, scleroderma can result in systemic or localized symptoms. Overall, the most common symptom is RP.5 Localized scleroderma and limited cutaneous systemic sclerosis manifest with symptoms of the skin and underlying tissues. Diffuse cutaneous systemic sclerosis involves cutaneous and visceral symptoms, including lung, esophageal, and vascular involvement.6 Similar to MCTD, scleroderma is most prevalent in middle-aged females,7 though it occurs at a higher rate and with a more severe disease course in Black patients.8
A highly sensitive and specific test for scleroderma that can aid in diagnosis is the neck sign—tightening of the skin of the neck when the head extends.9,10 In one study, the neck sign was positive in more than 90% of patients with scleroderma and negative for control patients and those with primary RP.9 Thus, neck sclerosis is a common manifestation of scleroderma for which patients may seek treatment.
While there is no curative treatment for scleroderma, skin manifestations can be treated with mycophenolate mofetil or methotrexate.5 Systemic treatments may be recommended if the patient has additional symptoms, such as azathioprine for myositis/arthritis and cyclophosphamide for interstitial lung disease.5 However, it is important to note that these medications are associated with risk for gastrointestinal upset, mouth sores, fatigue, or other complications.
Botulinum toxin is a bacterial protein toxin and neuromodulator that inhibits neurotransmitter release by cleaving SNARE proteins at peripheral nerve terminal junctions.11 It has been used in a variety of dermatologic and nondermatologic conditions, including migraines, hyperhidrosis, contractures, scars, and overactive bladder. It also has been used in aesthetics for facial rejuvenation and minimization of wrinkle appearance. Dermatologists and rheumatologists have successfully used BTX to treat primary and secondary RP—the most common symptom of scleroderma—due to its vasodilatation properties.12 Although our patient did not have RP, use of BTX to treat other features of scleroderma, including en coup de sabre, thoracic outlet syndrome, dyspareunia, gastroparesis, pterygium inversum unguis, and dysphagia has been documented.13-18 An in vivo mouse study that examined the possible mechanism for BTX as a treatment in scleroderma found that BTX injections significantly decreased dermal thickness and inflammation in fibrosis (P<.05). An analysis of oxidative stress and mRNA expression showed that BTX may treat fibrosis by suppressing oxidative stress and inflammatory cells, resulting in decreased apoptosis and oxidant-induced intracellular accumulation of reactive oxygen species.19 Another animal study demonstrated the positive effects of BTX treatment for fibrosis of the bladder in rats.20 In one case report, a female patient with scleroderma and facial fibrosis received perioral BTX injections for cosmetic purposes but also observed improvement in mouth constriction, demonstrating the potential efficacy of BTX for facial fibrosis.21
Our case demonstrates the potential positive effects of BTX treatment in patients with features of sclerosis or fibrosis, particularly in the neck region. We recommend assessing the efficacy of the initial BTX treatment after 2 to 3 weeks, with additional injections as needed to achieve the patient’s desired level of comfort and appearance at approximately 3-month intervals (aligning with the expected duration of efficacy of BTX).22 Our patient experienced considerable relief and high satisfaction with BTX treatment. Given the limitations of sclerosis treatments and the unwanted adverse-effect profile of systemic treatments, BTX injections may be a preferrable treatment option for cutaneous manifestations of scleroderma among patients. Future studies with larger patient populations and a control group are warranted to further explore the use of BTX for the dermatologic treatment of scleroderma.
To the Editor:
Scleroderma is a chronic autoimmune connective tissue disease that results in excessive collagen deposition in the skin and other organs throughout the body. On its own or in the setting of mixed connective tissue disease, scleroderma can result in systemic or localized symptoms that can limit patients’ functional capabilities, cause pain and discomfort, and reduce self-esteem—all negatively impacting patients’ quality of life.1,2 Neck sclerosis is a common manifestation of scleroderma. There is no curative treatment for scleroderma; thus, therapy is focused on slowing disease progression and improving quality of life. We present a case of neck sclerosis in a 44-year-old woman with scleroderma that was successfully treated with botulinum toxin (BTX) type A injection, resulting in improved skin laxity and appearance with high patient satisfaction. Our case demonstrates the potential positive effects of BTX treatment in patients with features of sclerosis or fibrosis, particularly in the neck region.
A 44-year-old woman presented to the dermatology clinic for treatment of thickened neck skin with stiffness and tightness that had been present for months to years. She had a history of mixed connective tissue disease (MCTD)(positive anti-ribonucleoprotein, anti–Sjögren syndrome–related antigen, and anti-Smith antibodies) with features of scleroderma and polyarthritis. The patient currently was taking sulfasalazine for the polyarthritis; she previously had taken hydroxychloroquine but discontinued treatment due to ineffectiveness. She was not taking any topical or systemic medications for scleroderma. On physical examination, the skin on the anterior neck appeared thickened with shiny patches (Figure 1). Pinching the skin in the affected area demonstrated sclerosis with high tension.

The dermatologist (J.J.) discussed potential treatment options to help relax the tension in the skin of the anterior neck, including BTX injections. After receiving counsel on adverse effects, alternative treatments, and postprocedural care, the patient decided to proceed with the procedure. The anterior neck was cleansed with an alcohol swab and 37 units (range, 25–50 units) of incobotulinumtoxinA (reconstituted using 2.5-mL bacteriostatic normal saline per 100 units) was injected transdermally using a 9-point injection technique, with each injection placed approximately 1 cm apart. The approximate treatment area included the space between the sternocleidomastoid anterior edges and below the hyoid bone up to the cricothyroid membrane (anatomic zone II).
When the patient returned for follow-up 3 weeks later, she reported considerable improvement in the stiffness and appearance of the skin on the anterior neck. On physical examination, the skin of the neck appeared softened, and improved laxity was seen on pinching the skin compared to the initial presentation (Figure 2). The patient expressed satisfaction with the results and denied any adverse events following the procedure.

Mixed connective tissue disease manifests with a combination of features from various disorders—mainly lupus, scleroderma, polymyositis, and rheumatoid arthritis. It is most prevalent in females and often is diagnosed in the third decade of life.3 It is associated with positive antinuclear antibodies and human leukocyte antigen (HLA) II alleles (HLA-DR4, HLA-DR1, and HLA-DR2). Raynaud phenomenon (RP), one of the most common skin manifestations in both scleroderma and MCTD, is present in 75% to 90% of patients with MCTD.3
Scleroderma is a chronic connective tissue disorder that results in excessive collagen deposition in the skin and other organs throughout the body.4 Although the etiology is unknown, scleroderma develops when overactivation of the immune system leads to CD4+ T-lymphocyte infiltration in the skin, along with the release of profibrotic interleukins and growth factors, resulting in fibrosis.4 Subtypes include localized scleroderma (morphea), limited cutaneous systemic sclerosis (formerly known as CREST [calcinosis, RP, esophageal dysmotility, sclerodactyly, and telangiectasia] syndrome), diffuse cutaneous systemic sclerosis, and systemic sclerosis sine scleroderma.5 Scleroderma is associated with positive antinuclear antibodies and HLA II alleles (HLA-DR2 and HLA-DR5).
On its own or in the setting of MCTD, scleroderma can result in systemic or localized symptoms. Overall, the most common symptom is RP.5 Localized scleroderma and limited cutaneous systemic sclerosis manifest with symptoms of the skin and underlying tissues. Diffuse cutaneous systemic sclerosis involves cutaneous and visceral symptoms, including lung, esophageal, and vascular involvement.6 Similar to MCTD, scleroderma is most prevalent in middle-aged females,7 though it occurs at a higher rate and with a more severe disease course in Black patients.8
A highly sensitive and specific test for scleroderma that can aid in diagnosis is the neck sign—tightening of the skin of the neck when the head extends.9,10 In one study, the neck sign was positive in more than 90% of patients with scleroderma and negative for control patients and those with primary RP.9 Thus, neck sclerosis is a common manifestation of scleroderma for which patients may seek treatment.
While there is no curative treatment for scleroderma, skin manifestations can be treated with mycophenolate mofetil or methotrexate.5 Systemic treatments may be recommended if the patient has additional symptoms, such as azathioprine for myositis/arthritis and cyclophosphamide for interstitial lung disease.5 However, it is important to note that these medications are associated with risk for gastrointestinal upset, mouth sores, fatigue, or other complications.
Botulinum toxin is a bacterial protein toxin and neuromodulator that inhibits neurotransmitter release by cleaving SNARE proteins at peripheral nerve terminal junctions.11 It has been used in a variety of dermatologic and nondermatologic conditions, including migraines, hyperhidrosis, contractures, scars, and overactive bladder. It also has been used in aesthetics for facial rejuvenation and minimization of wrinkle appearance. Dermatologists and rheumatologists have successfully used BTX to treat primary and secondary RP—the most common symptom of scleroderma—due to its vasodilatation properties.12 Although our patient did not have RP, use of BTX to treat other features of scleroderma, including en coup de sabre, thoracic outlet syndrome, dyspareunia, gastroparesis, pterygium inversum unguis, and dysphagia has been documented.13-18 An in vivo mouse study that examined the possible mechanism for BTX as a treatment in scleroderma found that BTX injections significantly decreased dermal thickness and inflammation in fibrosis (P<.05). An analysis of oxidative stress and mRNA expression showed that BTX may treat fibrosis by suppressing oxidative stress and inflammatory cells, resulting in decreased apoptosis and oxidant-induced intracellular accumulation of reactive oxygen species.19 Another animal study demonstrated the positive effects of BTX treatment for fibrosis of the bladder in rats.20 In one case report, a female patient with scleroderma and facial fibrosis received perioral BTX injections for cosmetic purposes but also observed improvement in mouth constriction, demonstrating the potential efficacy of BTX for facial fibrosis.21
Our case demonstrates the potential positive effects of BTX treatment in patients with features of sclerosis or fibrosis, particularly in the neck region. We recommend assessing the efficacy of the initial BTX treatment after 2 to 3 weeks, with additional injections as needed to achieve the patient’s desired level of comfort and appearance at approximately 3-month intervals (aligning with the expected duration of efficacy of BTX).22 Our patient experienced considerable relief and high satisfaction with BTX treatment. Given the limitations of sclerosis treatments and the unwanted adverse-effect profile of systemic treatments, BTX injections may be a preferrable treatment option for cutaneous manifestations of scleroderma among patients. Future studies with larger patient populations and a control group are warranted to further explore the use of BTX for the dermatologic treatment of scleroderma.
- Lis-S´wie¸ty A, Skrzypek-Salamon A, Ranosz-Janicka I, et al. Health-related quality of life and its influencing factors in adult patients with localized scleroderma—a cross-sectional study. Health Qual Life Outcomes. 2020;18:133. doi:10.1186/s12955-020-01386-0
- Almeida C, Almeida I, Vasconcelos C. Quality of life in systemic sclerosis. Autoimmun Rev. 2015;14:1087-1096. doi:10.1016/j.autrev.2015.07.012
- Ortega-Hernandez OD, Shoenfeld Y. Mixed connective tissue disease: an overview of clinical manifestations, diagnosis and treatment. Best Pract Res Clin Rheumatol. 2012;26:61-72. doi:10.1016/j.berh.2012.01.009
- Rongioletti F, Ferreli C, Atzori L, et al. Scleroderma with an update about clinico-pathological correlation. G Ital Dermatol Venereol. 2018;153:208-215. doi:10.23736/S0392-0488.18.05922-9
- Fett N. Scleroderma: nomenclature, etiology, pathogenesis, prognosis, and treatments: facts and controversies. Clin Dermatol. 2013;31:432-437. doi:10.1016/j.clindermatol.2013.01.010
- Careta MF, Romiti R. Localized scleroderma: clinical spectrum and therapeutic update. An Bras Dermatol. 2015;90:62-73. doi:10.1590/abd1806-4841.20152890
- Calderon LM, Pope JE. Scleroderma epidemiology update. Curr Opin Rheumatol. 2021;33:122-127. doi:10.1097/BOR.0000000000000785
- Morgan ND, Gelber AC. African Americans and scleroderma: examining the root cause of the association. Arthritis Care Res (Hoboken). 2019;71:1151-1153. doi:10.1002/acr.23860
- Barnett AJ. The “neck sign” in scleroderma. Arthritis Rheum. 1989;32:209-211. doi:10.1002/anr.1780320215
- Barnett AJ, Miller M, Littlejohn GO. The diagnosis and classification of scleroderma (systemic sclerosis). Postgrad Med J. 1988;64:121-125. doi:10.1136/pgmj.64.748.121
- Rossetto O, Pirazzini M, Fabris F, et al. Botulinum neurotoxins: mechanism of action. Handb Exp Pharmacol. 2021;263:35-47.doi:10.1007/164_2020_355
- Ennis D, Ahmad Z, Anderson MA, et al. Botulinum toxin in the management of primary and secondary Raynaud’s phenomenon. Best Pract Res Clin Rheumatol. 2021;35:101684. doi:10.1016/j.berh.2021.101684
- Turkmani MG, Alnomair N. Enhancement of the aesthetic outcome of scleroderma en coup de sabre with botulinum toxin injection. JAAD Case Rep. 2018;4:579-581. doi:10.1016/j.jdcr.2018.03.023
- Le EN, Freischlag JA, Christo PJ, et al. Thoracic outlet syndrome secondary to localized scleroderma treated with botulinum toxin injection. Arthritis Care Res (Hoboken). 2010;62:430-433. doi:10.1002/acr.20099
- Mousty E, Rathat G, Rouleau C, et al. Botulinum toxin type A for treatment of dyspareunia caused by localized scleroderma. Acta Obstet Gynecol Scand. 2011;90:926-927. doi:10.1111/j.1600-0412.2011.01183.x
- Tang DM, Friedenberg FK. Gastroparesis: approach, diagnostic evaluation, and management. Dis Mon. 2011;57:74-101. doi:10.1016/j.disamonth.2010.12.007
- Katschinski M. [Diagnosis and treatment of esophageal motility disorders]. Ther Umsch. 2001;58:128-133. doi:10.1024/0040-5930.58.3.128
- Kim DJ, Odell ID. Improvement of pterygium inversum unguis and Raynaud phenomenon with interdigital botulinum toxin injections. JAAD Case Rep. 2022;26:79-81. doi:10.1016/j.jdcr.2022.06.009
- Baral H, Sekiguchi A, Uchiyama A, et al. Inhibition of skin fibrosis in systemic sclerosis by botulinum toxin B via the suppression of oxidative stress. J Dermatol. 2021;48:1052-1061. doi:10.1111/1346-8138.15888
- Jia C, Xing T, Shang Z, et al. Botulinum toxin A improves neurogenic bladder fibrosis by suppressing transforming growth factor β1 expression in rats. Transl Androl Urol. 2021;10:2000-2007. doi:10.21037/tau-21-62
- Hoverson K, Love T, Lam TK, et al. A novel treatment for limited mouth opening due to facial fibrosis: a case series. J Am Acad Dermatol. 2018;78:190-192. doi:10.1016/j.jaad.2017.07.006
- Kollewe K, Mohammadi B, Köhler S, et al. Blepharospasm: long-term treatment with either Botox®, Xeomin® or Dysport®. J Neural Transm (Vienna). 2015;122:427-431. doi:10.1007/s00702-014-1278-z
- Lis-S´wie¸ty A, Skrzypek-Salamon A, Ranosz-Janicka I, et al. Health-related quality of life and its influencing factors in adult patients with localized scleroderma—a cross-sectional study. Health Qual Life Outcomes. 2020;18:133. doi:10.1186/s12955-020-01386-0
- Almeida C, Almeida I, Vasconcelos C. Quality of life in systemic sclerosis. Autoimmun Rev. 2015;14:1087-1096. doi:10.1016/j.autrev.2015.07.012
- Ortega-Hernandez OD, Shoenfeld Y. Mixed connective tissue disease: an overview of clinical manifestations, diagnosis and treatment. Best Pract Res Clin Rheumatol. 2012;26:61-72. doi:10.1016/j.berh.2012.01.009
- Rongioletti F, Ferreli C, Atzori L, et al. Scleroderma with an update about clinico-pathological correlation. G Ital Dermatol Venereol. 2018;153:208-215. doi:10.23736/S0392-0488.18.05922-9
- Fett N. Scleroderma: nomenclature, etiology, pathogenesis, prognosis, and treatments: facts and controversies. Clin Dermatol. 2013;31:432-437. doi:10.1016/j.clindermatol.2013.01.010
- Careta MF, Romiti R. Localized scleroderma: clinical spectrum and therapeutic update. An Bras Dermatol. 2015;90:62-73. doi:10.1590/abd1806-4841.20152890
- Calderon LM, Pope JE. Scleroderma epidemiology update. Curr Opin Rheumatol. 2021;33:122-127. doi:10.1097/BOR.0000000000000785
- Morgan ND, Gelber AC. African Americans and scleroderma: examining the root cause of the association. Arthritis Care Res (Hoboken). 2019;71:1151-1153. doi:10.1002/acr.23860
- Barnett AJ. The “neck sign” in scleroderma. Arthritis Rheum. 1989;32:209-211. doi:10.1002/anr.1780320215
- Barnett AJ, Miller M, Littlejohn GO. The diagnosis and classification of scleroderma (systemic sclerosis). Postgrad Med J. 1988;64:121-125. doi:10.1136/pgmj.64.748.121
- Rossetto O, Pirazzini M, Fabris F, et al. Botulinum neurotoxins: mechanism of action. Handb Exp Pharmacol. 2021;263:35-47.doi:10.1007/164_2020_355
- Ennis D, Ahmad Z, Anderson MA, et al. Botulinum toxin in the management of primary and secondary Raynaud’s phenomenon. Best Pract Res Clin Rheumatol. 2021;35:101684. doi:10.1016/j.berh.2021.101684
- Turkmani MG, Alnomair N. Enhancement of the aesthetic outcome of scleroderma en coup de sabre with botulinum toxin injection. JAAD Case Rep. 2018;4:579-581. doi:10.1016/j.jdcr.2018.03.023
- Le EN, Freischlag JA, Christo PJ, et al. Thoracic outlet syndrome secondary to localized scleroderma treated with botulinum toxin injection. Arthritis Care Res (Hoboken). 2010;62:430-433. doi:10.1002/acr.20099
- Mousty E, Rathat G, Rouleau C, et al. Botulinum toxin type A for treatment of dyspareunia caused by localized scleroderma. Acta Obstet Gynecol Scand. 2011;90:926-927. doi:10.1111/j.1600-0412.2011.01183.x
- Tang DM, Friedenberg FK. Gastroparesis: approach, diagnostic evaluation, and management. Dis Mon. 2011;57:74-101. doi:10.1016/j.disamonth.2010.12.007
- Katschinski M. [Diagnosis and treatment of esophageal motility disorders]. Ther Umsch. 2001;58:128-133. doi:10.1024/0040-5930.58.3.128
- Kim DJ, Odell ID. Improvement of pterygium inversum unguis and Raynaud phenomenon with interdigital botulinum toxin injections. JAAD Case Rep. 2022;26:79-81. doi:10.1016/j.jdcr.2022.06.009
- Baral H, Sekiguchi A, Uchiyama A, et al. Inhibition of skin fibrosis in systemic sclerosis by botulinum toxin B via the suppression of oxidative stress. J Dermatol. 2021;48:1052-1061. doi:10.1111/1346-8138.15888
- Jia C, Xing T, Shang Z, et al. Botulinum toxin A improves neurogenic bladder fibrosis by suppressing transforming growth factor β1 expression in rats. Transl Androl Urol. 2021;10:2000-2007. doi:10.21037/tau-21-62
- Hoverson K, Love T, Lam TK, et al. A novel treatment for limited mouth opening due to facial fibrosis: a case series. J Am Acad Dermatol. 2018;78:190-192. doi:10.1016/j.jaad.2017.07.006
- Kollewe K, Mohammadi B, Köhler S, et al. Blepharospasm: long-term treatment with either Botox®, Xeomin® or Dysport®. J Neural Transm (Vienna). 2015;122:427-431. doi:10.1007/s00702-014-1278-z
Practice Points
- Scleroderma is a chronic autoimmune connective tissue disease that results in excessive collagen deposition in the skin and other organs throughout the body.
- Although there is no curative treatment for scleroderma, there are options to slow disease progression and improve quality of life.
- Botulinum toxin injection may be a preferred treatment option in patients with features of sclerosis or fibrosis related to scleroderma, particularly in the neck region.